User login
In Case You Missed It: COVID
New challenge for docs: End of COVID federal public health emergency
The Biden administration intends to end by May 11 certain COVID-19 emergency measures used to aid in the response to the pandemic, while many others will remain in place.
A separate declaration covers the Food and Drug Administration’s emergency use authorizations (EUAs) for COVID medicines and tests. That would not be affected by the May 11 deadline, the FDA said. In addition, Congress and state lawmakers have extended some COVID response measures.
The result is a patchwork of emergency COVID-19 measures with different end dates.
The American Medical Association and the American Academy of Family Physicians (AAFP) are assessing how best to advise their members about the end of the public health emergency.
Several waivers regarding copays and coverage and policies regarding controlled substances will expire, Claire Ernst, director of government affairs at the Medical Group Management Association, told this news organization.
The impact of the unwinding “will vary based on some factors, such as what state the practice resides in,” Ms. Ernst said. “Fortunately, Congress provided some predictability for practices by extending many of the telehealth waivers through the end of 2024.”
The AAFP told this news organization that it has joined several other groups in calling for the release of proposed Drug Enforcement Administration (DEA) regulations meant to permanently allow prescriptions of buprenorphine treatment for opioid use disorder via telehealth. The AAFP and other groups want to review these proposals and, if needed, urge the DEA to modify or finalize before there are any disruptions in access to medications for opioid use disorder.
Patients’ questions
Clinicians can expect to field patients’ questions about their insurance coverage and what they need to pay, said Nancy Foster, vice president for quality and patient safety policy at the American Hospital Association (AHA).
“Your doctor’s office, that clinic you typically get care at, that is the face of medicine to you,” Ms. Foster told this news organization. “Many doctors and their staff will be asked, ‘What’s happening with Medicaid?’ ‘What about my Medicare coverage?’ ‘Can I still access care in the same way that I did before?’ ”
Physicians will need to be ready to answers those question, or point patients to where they can get answers, Ms. Foster said.
For example, Medicaid will no longer cover postpartum care for some enrollees after giving birth, said Taylor Platt, health policy manager for the American College of Obstetricians and Gynecologists.
The federal response to the pandemic created “a de facto postpartum coverage extension for Medicaid enrollees,” which will be lost in some states, Ms. Platt told this news organization. However, 28 states and the District of Columbia have taken separate measures to extend postpartum coverage to 1 year.
“This coverage has been critical for postpartum individuals to address health needs like substance use and mental health treatment and chronic conditions,” Ms. Platt said.
States significantly changed Medicaid policy to expand access to care during the pandemic.
All 50 states and the District of Columbia, for example, expanded coverage or access to telehealth services in Medicaid during the pandemic, according to a Jan. 31 report from the Kaiser Family Foundation (KFF). These expansions expire under various deadlines, although most states have made or are planning to make some Medicaid telehealth flexibilities permanent, KFF said.
The KFF report notes that all states and the District of Columbia temporarily waived some aspects of state licensure requirements, so that clinicians with equivalent licenses in other states could practice via telehealth.
In some states, these waivers are still active and are tied to the end of the federal emergency declaration. In others, they expired, with some states allowing for long-term or permanent interstate telemedicine, KFF said. (The Federation of State Medical Boards has a detailed summary of these modifications.)
The end of free COVID vaccines, testing for some patients
The AAFP has also raised concerns about continued access to COVID-19 vaccines, particularly for uninsured adults. Ashish Jha, MD, MPH, the White House COVID-19 Response Coordinator, said in a tweet that this transition, however, wouldn’t happen until a few months after the public health emergency ends.
After those few months, there will be a transition from U.S. government–distributed vaccines and treatments to ones purchased through the regular health care system, the “way we do for every other vaccine and treatment,” Dr. Jha added.
But that raises the same kind of difficult questions that permeate U.S. health care, with a potential to keep COVID active, said Patricia Jackson, RN, president of the Association for Professionals in Infection Control and Epidemiology (APIC).
People who don’t have insurance may lose access to COVID testing and vaccines.
“Will that lead to increases in transmission? Who knows,” Ms. Jackson told this news organization. “We will have to see. There are some health equity issues that potentially arise.”
Future FDA actions
Biden’s May 11 deadline applies to emergency provisions made under a Section 319 declaration, which allow the Department of Health and Human Services to respond to crises.
But a separate flexibility, known as a Section 564 declaration, covers the FDA’s EUAs, which can remain in effect even as the other declarations end.
The best-known EUAs for the pandemic were used to bring COVID vaccines and treatments to market. Many of these have since been converted to normal approvals as companies presented more evidence to support the initial emergency approvals. In other cases, EUAs have been withdrawn owing to disappointing research results, changing virus strains, and evolving medical treatments.
The FDA also used many EUAs to cover new uses of ventilators and other hospital equipment and expand these supplies in response to the pandemic, said Mark Howell, AHA’s director of policy and patient safety.
The FDA should examine the EUAs issued during the pandemic to see what greater flexibilities might be used to deal with future serious shortages of critical supplies. International incidents such as the war in Ukraine show how fragile the supply chain can be. The FDA should consider its recent experience with EUAs to address this, Mr. Howell said.
“What do we do coming out of the pandemic? And how do we think about being more proactive in this space to ensure that our supply doesn’t bottleneck, that we continue to make sure that providers have access to supply that’s not only safe and effective, but that they can use?” Mr. Howell told this news organization.
Such planning might also help prepare the country for the next pandemic, which is a near certainty, APIC’s Ms. Jackson said. The nation needs a nimbler response to the next major outbreak of an infectious disease, she said.
“There is going to be a next time,” Ms. Jackson said. “We are going to have another pandemic.”
A version of this article first appeared on Medscape.com.
The Biden administration intends to end by May 11 certain COVID-19 emergency measures used to aid in the response to the pandemic, while many others will remain in place.
A separate declaration covers the Food and Drug Administration’s emergency use authorizations (EUAs) for COVID medicines and tests. That would not be affected by the May 11 deadline, the FDA said. In addition, Congress and state lawmakers have extended some COVID response measures.
The result is a patchwork of emergency COVID-19 measures with different end dates.
The American Medical Association and the American Academy of Family Physicians (AAFP) are assessing how best to advise their members about the end of the public health emergency.
Several waivers regarding copays and coverage and policies regarding controlled substances will expire, Claire Ernst, director of government affairs at the Medical Group Management Association, told this news organization.
The impact of the unwinding “will vary based on some factors, such as what state the practice resides in,” Ms. Ernst said. “Fortunately, Congress provided some predictability for practices by extending many of the telehealth waivers through the end of 2024.”
The AAFP told this news organization that it has joined several other groups in calling for the release of proposed Drug Enforcement Administration (DEA) regulations meant to permanently allow prescriptions of buprenorphine treatment for opioid use disorder via telehealth. The AAFP and other groups want to review these proposals and, if needed, urge the DEA to modify or finalize before there are any disruptions in access to medications for opioid use disorder.
Patients’ questions
Clinicians can expect to field patients’ questions about their insurance coverage and what they need to pay, said Nancy Foster, vice president for quality and patient safety policy at the American Hospital Association (AHA).
“Your doctor’s office, that clinic you typically get care at, that is the face of medicine to you,” Ms. Foster told this news organization. “Many doctors and their staff will be asked, ‘What’s happening with Medicaid?’ ‘What about my Medicare coverage?’ ‘Can I still access care in the same way that I did before?’ ”
Physicians will need to be ready to answers those question, or point patients to where they can get answers, Ms. Foster said.
For example, Medicaid will no longer cover postpartum care for some enrollees after giving birth, said Taylor Platt, health policy manager for the American College of Obstetricians and Gynecologists.
The federal response to the pandemic created “a de facto postpartum coverage extension for Medicaid enrollees,” which will be lost in some states, Ms. Platt told this news organization. However, 28 states and the District of Columbia have taken separate measures to extend postpartum coverage to 1 year.
“This coverage has been critical for postpartum individuals to address health needs like substance use and mental health treatment and chronic conditions,” Ms. Platt said.
States significantly changed Medicaid policy to expand access to care during the pandemic.
All 50 states and the District of Columbia, for example, expanded coverage or access to telehealth services in Medicaid during the pandemic, according to a Jan. 31 report from the Kaiser Family Foundation (KFF). These expansions expire under various deadlines, although most states have made or are planning to make some Medicaid telehealth flexibilities permanent, KFF said.
The KFF report notes that all states and the District of Columbia temporarily waived some aspects of state licensure requirements, so that clinicians with equivalent licenses in other states could practice via telehealth.
In some states, these waivers are still active and are tied to the end of the federal emergency declaration. In others, they expired, with some states allowing for long-term or permanent interstate telemedicine, KFF said. (The Federation of State Medical Boards has a detailed summary of these modifications.)
The end of free COVID vaccines, testing for some patients
The AAFP has also raised concerns about continued access to COVID-19 vaccines, particularly for uninsured adults. Ashish Jha, MD, MPH, the White House COVID-19 Response Coordinator, said in a tweet that this transition, however, wouldn’t happen until a few months after the public health emergency ends.
After those few months, there will be a transition from U.S. government–distributed vaccines and treatments to ones purchased through the regular health care system, the “way we do for every other vaccine and treatment,” Dr. Jha added.
But that raises the same kind of difficult questions that permeate U.S. health care, with a potential to keep COVID active, said Patricia Jackson, RN, president of the Association for Professionals in Infection Control and Epidemiology (APIC).
People who don’t have insurance may lose access to COVID testing and vaccines.
“Will that lead to increases in transmission? Who knows,” Ms. Jackson told this news organization. “We will have to see. There are some health equity issues that potentially arise.”
Future FDA actions
Biden’s May 11 deadline applies to emergency provisions made under a Section 319 declaration, which allow the Department of Health and Human Services to respond to crises.
But a separate flexibility, known as a Section 564 declaration, covers the FDA’s EUAs, which can remain in effect even as the other declarations end.
The best-known EUAs for the pandemic were used to bring COVID vaccines and treatments to market. Many of these have since been converted to normal approvals as companies presented more evidence to support the initial emergency approvals. In other cases, EUAs have been withdrawn owing to disappointing research results, changing virus strains, and evolving medical treatments.
The FDA also used many EUAs to cover new uses of ventilators and other hospital equipment and expand these supplies in response to the pandemic, said Mark Howell, AHA’s director of policy and patient safety.
The FDA should examine the EUAs issued during the pandemic to see what greater flexibilities might be used to deal with future serious shortages of critical supplies. International incidents such as the war in Ukraine show how fragile the supply chain can be. The FDA should consider its recent experience with EUAs to address this, Mr. Howell said.
“What do we do coming out of the pandemic? And how do we think about being more proactive in this space to ensure that our supply doesn’t bottleneck, that we continue to make sure that providers have access to supply that’s not only safe and effective, but that they can use?” Mr. Howell told this news organization.
Such planning might also help prepare the country for the next pandemic, which is a near certainty, APIC’s Ms. Jackson said. The nation needs a nimbler response to the next major outbreak of an infectious disease, she said.
“There is going to be a next time,” Ms. Jackson said. “We are going to have another pandemic.”
A version of this article first appeared on Medscape.com.
The Biden administration intends to end by May 11 certain COVID-19 emergency measures used to aid in the response to the pandemic, while many others will remain in place.
A separate declaration covers the Food and Drug Administration’s emergency use authorizations (EUAs) for COVID medicines and tests. That would not be affected by the May 11 deadline, the FDA said. In addition, Congress and state lawmakers have extended some COVID response measures.
The result is a patchwork of emergency COVID-19 measures with different end dates.
The American Medical Association and the American Academy of Family Physicians (AAFP) are assessing how best to advise their members about the end of the public health emergency.
Several waivers regarding copays and coverage and policies regarding controlled substances will expire, Claire Ernst, director of government affairs at the Medical Group Management Association, told this news organization.
The impact of the unwinding “will vary based on some factors, such as what state the practice resides in,” Ms. Ernst said. “Fortunately, Congress provided some predictability for practices by extending many of the telehealth waivers through the end of 2024.”
The AAFP told this news organization that it has joined several other groups in calling for the release of proposed Drug Enforcement Administration (DEA) regulations meant to permanently allow prescriptions of buprenorphine treatment for opioid use disorder via telehealth. The AAFP and other groups want to review these proposals and, if needed, urge the DEA to modify or finalize before there are any disruptions in access to medications for opioid use disorder.
Patients’ questions
Clinicians can expect to field patients’ questions about their insurance coverage and what they need to pay, said Nancy Foster, vice president for quality and patient safety policy at the American Hospital Association (AHA).
“Your doctor’s office, that clinic you typically get care at, that is the face of medicine to you,” Ms. Foster told this news organization. “Many doctors and their staff will be asked, ‘What’s happening with Medicaid?’ ‘What about my Medicare coverage?’ ‘Can I still access care in the same way that I did before?’ ”
Physicians will need to be ready to answers those question, or point patients to where they can get answers, Ms. Foster said.
For example, Medicaid will no longer cover postpartum care for some enrollees after giving birth, said Taylor Platt, health policy manager for the American College of Obstetricians and Gynecologists.
The federal response to the pandemic created “a de facto postpartum coverage extension for Medicaid enrollees,” which will be lost in some states, Ms. Platt told this news organization. However, 28 states and the District of Columbia have taken separate measures to extend postpartum coverage to 1 year.
“This coverage has been critical for postpartum individuals to address health needs like substance use and mental health treatment and chronic conditions,” Ms. Platt said.
States significantly changed Medicaid policy to expand access to care during the pandemic.
All 50 states and the District of Columbia, for example, expanded coverage or access to telehealth services in Medicaid during the pandemic, according to a Jan. 31 report from the Kaiser Family Foundation (KFF). These expansions expire under various deadlines, although most states have made or are planning to make some Medicaid telehealth flexibilities permanent, KFF said.
The KFF report notes that all states and the District of Columbia temporarily waived some aspects of state licensure requirements, so that clinicians with equivalent licenses in other states could practice via telehealth.
In some states, these waivers are still active and are tied to the end of the federal emergency declaration. In others, they expired, with some states allowing for long-term or permanent interstate telemedicine, KFF said. (The Federation of State Medical Boards has a detailed summary of these modifications.)
The end of free COVID vaccines, testing for some patients
The AAFP has also raised concerns about continued access to COVID-19 vaccines, particularly for uninsured adults. Ashish Jha, MD, MPH, the White House COVID-19 Response Coordinator, said in a tweet that this transition, however, wouldn’t happen until a few months after the public health emergency ends.
After those few months, there will be a transition from U.S. government–distributed vaccines and treatments to ones purchased through the regular health care system, the “way we do for every other vaccine and treatment,” Dr. Jha added.
But that raises the same kind of difficult questions that permeate U.S. health care, with a potential to keep COVID active, said Patricia Jackson, RN, president of the Association for Professionals in Infection Control and Epidemiology (APIC).
People who don’t have insurance may lose access to COVID testing and vaccines.
“Will that lead to increases in transmission? Who knows,” Ms. Jackson told this news organization. “We will have to see. There are some health equity issues that potentially arise.”
Future FDA actions
Biden’s May 11 deadline applies to emergency provisions made under a Section 319 declaration, which allow the Department of Health and Human Services to respond to crises.
But a separate flexibility, known as a Section 564 declaration, covers the FDA’s EUAs, which can remain in effect even as the other declarations end.
The best-known EUAs for the pandemic were used to bring COVID vaccines and treatments to market. Many of these have since been converted to normal approvals as companies presented more evidence to support the initial emergency approvals. In other cases, EUAs have been withdrawn owing to disappointing research results, changing virus strains, and evolving medical treatments.
The FDA also used many EUAs to cover new uses of ventilators and other hospital equipment and expand these supplies in response to the pandemic, said Mark Howell, AHA’s director of policy and patient safety.
The FDA should examine the EUAs issued during the pandemic to see what greater flexibilities might be used to deal with future serious shortages of critical supplies. International incidents such as the war in Ukraine show how fragile the supply chain can be. The FDA should consider its recent experience with EUAs to address this, Mr. Howell said.
“What do we do coming out of the pandemic? And how do we think about being more proactive in this space to ensure that our supply doesn’t bottleneck, that we continue to make sure that providers have access to supply that’s not only safe and effective, but that they can use?” Mr. Howell told this news organization.
Such planning might also help prepare the country for the next pandemic, which is a near certainty, APIC’s Ms. Jackson said. The nation needs a nimbler response to the next major outbreak of an infectious disease, she said.
“There is going to be a next time,” Ms. Jackson said. “We are going to have another pandemic.”
A version of this article first appeared on Medscape.com.
New report says suicide rates rising among young Black people
Significant increases in suicide occurred among Native American, Black and Hispanic people, with a startling rise among young Black people. Meanwhile, the rate of suicide among older people declined between 2018 and 2021, the Centers for Disease Control and Prevention has reported.
In 2021, 48,183 people died by suicide in the United States, which equates to a suicide rate of 14.1 per 100,000 people. That level equals the 2018 suicide rate, which had seen a peak that was followed by declines associated with the pandemic.
Experts said rebounding suicide rates are common following times of crisis, such as the COVID-19 pandemic. Suicide declines have also occurred during times of war and natural disaster, when psychological resilience tends to increase and people work together to overcome shared adversity.
“That will wane, and then you will see rebounding in suicide rates. That is, in fact, what we feared would happen. And it has happened, at least in 2021,” Christine Moutier, MD, chief medical officer of the American Foundation for Suicide Prevention, told the New York Times.
The new CDC report found that the largest increase was among Black people aged 10-24 years, who experienced a 36.6% increase in suicide rate between 2018 and 2021. While Black people experience mental illness at the same rates as that of the general population, historically they have disproportionately limited access to mental health care, according to the American Psychiatric Association.
CDC report authors noted that some of the biggest increases in suicide rates occurred among groups most affected by the pandemic.
From 2018 to 2021, the suicide rate for people aged 25-44 increased among Native Americans by 33.7% and among Black people by 22.9%. Suicide increased among multiracial people by 20.6% and among Hispanic or Latinx people by 19.4%. Among White people of all ages, the suicide rate declined or remained steady.
“As the nation continues to respond to the short- and long-term impacts of the COVID-19 pandemic, remaining vigilant in prevention efforts is critical, especially among disproportionately affected populations where longer-term impacts might compound preexisting inequities in suicide risk,” the CDC researchers wrote.
A version of this article first appeared on WebMD.com.
Significant increases in suicide occurred among Native American, Black and Hispanic people, with a startling rise among young Black people. Meanwhile, the rate of suicide among older people declined between 2018 and 2021, the Centers for Disease Control and Prevention has reported.
In 2021, 48,183 people died by suicide in the United States, which equates to a suicide rate of 14.1 per 100,000 people. That level equals the 2018 suicide rate, which had seen a peak that was followed by declines associated with the pandemic.
Experts said rebounding suicide rates are common following times of crisis, such as the COVID-19 pandemic. Suicide declines have also occurred during times of war and natural disaster, when psychological resilience tends to increase and people work together to overcome shared adversity.
“That will wane, and then you will see rebounding in suicide rates. That is, in fact, what we feared would happen. And it has happened, at least in 2021,” Christine Moutier, MD, chief medical officer of the American Foundation for Suicide Prevention, told the New York Times.
The new CDC report found that the largest increase was among Black people aged 10-24 years, who experienced a 36.6% increase in suicide rate between 2018 and 2021. While Black people experience mental illness at the same rates as that of the general population, historically they have disproportionately limited access to mental health care, according to the American Psychiatric Association.
CDC report authors noted that some of the biggest increases in suicide rates occurred among groups most affected by the pandemic.
From 2018 to 2021, the suicide rate for people aged 25-44 increased among Native Americans by 33.7% and among Black people by 22.9%. Suicide increased among multiracial people by 20.6% and among Hispanic or Latinx people by 19.4%. Among White people of all ages, the suicide rate declined or remained steady.
“As the nation continues to respond to the short- and long-term impacts of the COVID-19 pandemic, remaining vigilant in prevention efforts is critical, especially among disproportionately affected populations where longer-term impacts might compound preexisting inequities in suicide risk,” the CDC researchers wrote.
A version of this article first appeared on WebMD.com.
Significant increases in suicide occurred among Native American, Black and Hispanic people, with a startling rise among young Black people. Meanwhile, the rate of suicide among older people declined between 2018 and 2021, the Centers for Disease Control and Prevention has reported.
In 2021, 48,183 people died by suicide in the United States, which equates to a suicide rate of 14.1 per 100,000 people. That level equals the 2018 suicide rate, which had seen a peak that was followed by declines associated with the pandemic.
Experts said rebounding suicide rates are common following times of crisis, such as the COVID-19 pandemic. Suicide declines have also occurred during times of war and natural disaster, when psychological resilience tends to increase and people work together to overcome shared adversity.
“That will wane, and then you will see rebounding in suicide rates. That is, in fact, what we feared would happen. And it has happened, at least in 2021,” Christine Moutier, MD, chief medical officer of the American Foundation for Suicide Prevention, told the New York Times.
The new CDC report found that the largest increase was among Black people aged 10-24 years, who experienced a 36.6% increase in suicide rate between 2018 and 2021. While Black people experience mental illness at the same rates as that of the general population, historically they have disproportionately limited access to mental health care, according to the American Psychiatric Association.
CDC report authors noted that some of the biggest increases in suicide rates occurred among groups most affected by the pandemic.
From 2018 to 2021, the suicide rate for people aged 25-44 increased among Native Americans by 33.7% and among Black people by 22.9%. Suicide increased among multiracial people by 20.6% and among Hispanic or Latinx people by 19.4%. Among White people of all ages, the suicide rate declined or remained steady.
“As the nation continues to respond to the short- and long-term impacts of the COVID-19 pandemic, remaining vigilant in prevention efforts is critical, especially among disproportionately affected populations where longer-term impacts might compound preexisting inequities in suicide risk,” the CDC researchers wrote.
A version of this article first appeared on WebMD.com.
Prehospital COVID therapy effective in rheumatic disease patients
Outpatient COVID-19 treatment with monoclonal antibodies or antiretroviral medications such as nirmatrelvir-ritonavir (Paxlovid) administered to patients with systemic autoimmune rheumatic disease led to lower odds of having severe outcomes when compared with similar patients who received no outpatient treatment in a real-world, retrospective analysis of cases.
The investigators found that there were nine hospitalizations or deaths (2.1%) among 426 patients who received outpatient treatment, compared with 49 (17.6%) among 278 who did not receive outpatient treatment, yielding an odds ratio of 0.12 (95% confidence interval, 0.05-0.25), after adjusting for age, sex, race, comorbidities, and kidney function. The study was published in Lancet Rheumatology.
“Across the board, there was a really strong association with receiving outpatient treatment and lower risk of severe COVID-19,” senior author Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine, Harvard Medical School and Brigham and Women’s Hospital, Boston, said in an interview. “It is pretty powerful evidence that, in this high-risk group, that treatment still matters related to preventing severe COVID. We found almost all patients who had severe COVID-19, either hospitalized or who had died, were in the untreated group.”
Early outpatient treatment an important tool in patients with rheumatic disease
Dr. Sparks noted that he and his coinvestigators conducted the study because the benefit of outpatient COVID-19 treatments in individuals with systemic autoimmune rheumatic disease was not adequately determined in clinical trials because they had infrequent enrollment of such patients.
The analysis included 704 patients with a mean age of 58.4 years who were seen at Mass General Brigham Integrated Health Care System, a multicenter health care system that includes 14 hospitals and primary care or specialty outpatient centers in the Boston area. A majority were female (76%) and White (84%). Nearly half had rheumatoid arthritis. Of the 704, 426 (61%) received outpatient treatment, which included nirmatrelvir-ritonavir (n = 307), monoclonal antibodies (n = 105), molnupiravir (n = 5), remdesivir (n = 3), and combination treatment (n = 6).
The findings underline the need to individualize approaches to outpatient treatment in those who test positive for SARS-CoV-2 to fend off severe COVID-19, according to Dr. Sparks. “It seems if you are vaccinated and in the general population that you are way less likely to have severe COVID-19 in the current environment, but that doesn’t necessarily apply to some high-risk groups like patients on immunosuppression. There are still patients at risk of severe COVID-19, and some of them are in this group of rheumatic patients. This should be part of the discussion related to deciding whether or not to treat.”
Dr. Sparks noted that vaccination against COVID-19 confers protection against developing severe COVID-19 in patients with rheumatic disease as it does in the general population, but patients with rheumatic diseases remain at increased risk for severe presentation. “Certainly, the vaccines really help our patients too, but there’s still a bit of a gap between the risk for our patients with rheumatic diseases and the general population” in developing severe COVID-19.
Dr. Sparks said he hopes the results represent a “call to action” that even among vaccinated patients there are still some who have poor outcomes, and that early outpatient treatment appears to be an important tool in the fight against poor outcomes from SARS-CoV-2 infection.
COVID-19 rebound
The study also reported on the phenomenon of COVID-19 rebound (recurrence of symptoms and test positivity after regimen completion) after oral outpatient SARS-CoV-2 treatment. “This [COVID-19 rebound] is a downside to treatment,” he said. COVID rebound was not infrequent: A total of 25 (8%) of 318 patients who received oral outpatient treatment had documented COVID-19 rebound.
“It was reassuring because we found no one who had rebound progressed to have severe COVID-19,” Dr. Sparks said. “On the other hand, [rebound] happened pretty frequently in our data, as 8% of patients are documented to have it.”
Dr. Sparks said he and coinvestigators speculate that more patients in the cohort may have experienced COVID-19 rebound but did not communicate this to their health care providers, and, as such, it was not documented in the medical record. The potential development of COVID-19 rebound “is something to counsel your patients about.” COVID-19 rebound is a phenomenon that is being most commonly observed with nirmatrelvir-ritonavir as outpatient treatment.
Possible confounding factors in study
Katie Bechman, MBChB, clinical lecturer in rheumatology at King’s College London, who coauthored an accompanying editorial about the study and its findings, pointed out that the study is limited by its observational design.
“With any study that looks at the efficacy of treatment, especially in an observational cohort, you’re going to have to consider the unmeasured confounding and the difference between these two groups,” Dr. Bechman said. “I know that they did try to adjust for that in this study, but there’s always going to be factors that we can’t [control for]. That is something that needs to be considered. I think that’s always something we need to consider when we’re looking at observational data.”
In lieu of a randomized, controlled trial, Dr. Bechman noted that the study and its associated findings serve as “the best data we have,” and she described the results as “very informative and positive.”
She added that the large number of patients represents a strength of the study, as does the robust method employed for identifying which patients had COVID-19.
The learnings from this study with respect to outpatient treatment can be applied to more common illnesses that patients with rheumatic disease may develop, such as the flu, according to Dr. Bechman.
“One of the positive aspects from this pandemic is that we’ve learned a huge amount about how best to treat certain viruses and prevent them in patients,” she said. “It would be worth thinking towards the future, what we can do for illnesses that we see very commonly in these populations. There may be treatment regimens that we haven’t really considered until now. You could hypothesize that in the next couple of years, if we have an influenza breakout, that we should be providing some prehospital antiviral treatment to patients, especially the ones that are at high risk.”
The study was conducted without outside funding. Dr. Sparks has received research support from Bristol-Myers Squibb and consulted for AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer unrelated to this work. Dr. Bechman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Outpatient COVID-19 treatment with monoclonal antibodies or antiretroviral medications such as nirmatrelvir-ritonavir (Paxlovid) administered to patients with systemic autoimmune rheumatic disease led to lower odds of having severe outcomes when compared with similar patients who received no outpatient treatment in a real-world, retrospective analysis of cases.
The investigators found that there were nine hospitalizations or deaths (2.1%) among 426 patients who received outpatient treatment, compared with 49 (17.6%) among 278 who did not receive outpatient treatment, yielding an odds ratio of 0.12 (95% confidence interval, 0.05-0.25), after adjusting for age, sex, race, comorbidities, and kidney function. The study was published in Lancet Rheumatology.
“Across the board, there was a really strong association with receiving outpatient treatment and lower risk of severe COVID-19,” senior author Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine, Harvard Medical School and Brigham and Women’s Hospital, Boston, said in an interview. “It is pretty powerful evidence that, in this high-risk group, that treatment still matters related to preventing severe COVID. We found almost all patients who had severe COVID-19, either hospitalized or who had died, were in the untreated group.”
Early outpatient treatment an important tool in patients with rheumatic disease
Dr. Sparks noted that he and his coinvestigators conducted the study because the benefit of outpatient COVID-19 treatments in individuals with systemic autoimmune rheumatic disease was not adequately determined in clinical trials because they had infrequent enrollment of such patients.
The analysis included 704 patients with a mean age of 58.4 years who were seen at Mass General Brigham Integrated Health Care System, a multicenter health care system that includes 14 hospitals and primary care or specialty outpatient centers in the Boston area. A majority were female (76%) and White (84%). Nearly half had rheumatoid arthritis. Of the 704, 426 (61%) received outpatient treatment, which included nirmatrelvir-ritonavir (n = 307), monoclonal antibodies (n = 105), molnupiravir (n = 5), remdesivir (n = 3), and combination treatment (n = 6).
The findings underline the need to individualize approaches to outpatient treatment in those who test positive for SARS-CoV-2 to fend off severe COVID-19, according to Dr. Sparks. “It seems if you are vaccinated and in the general population that you are way less likely to have severe COVID-19 in the current environment, but that doesn’t necessarily apply to some high-risk groups like patients on immunosuppression. There are still patients at risk of severe COVID-19, and some of them are in this group of rheumatic patients. This should be part of the discussion related to deciding whether or not to treat.”
Dr. Sparks noted that vaccination against COVID-19 confers protection against developing severe COVID-19 in patients with rheumatic disease as it does in the general population, but patients with rheumatic diseases remain at increased risk for severe presentation. “Certainly, the vaccines really help our patients too, but there’s still a bit of a gap between the risk for our patients with rheumatic diseases and the general population” in developing severe COVID-19.
Dr. Sparks said he hopes the results represent a “call to action” that even among vaccinated patients there are still some who have poor outcomes, and that early outpatient treatment appears to be an important tool in the fight against poor outcomes from SARS-CoV-2 infection.
COVID-19 rebound
The study also reported on the phenomenon of COVID-19 rebound (recurrence of symptoms and test positivity after regimen completion) after oral outpatient SARS-CoV-2 treatment. “This [COVID-19 rebound] is a downside to treatment,” he said. COVID rebound was not infrequent: A total of 25 (8%) of 318 patients who received oral outpatient treatment had documented COVID-19 rebound.
“It was reassuring because we found no one who had rebound progressed to have severe COVID-19,” Dr. Sparks said. “On the other hand, [rebound] happened pretty frequently in our data, as 8% of patients are documented to have it.”
Dr. Sparks said he and coinvestigators speculate that more patients in the cohort may have experienced COVID-19 rebound but did not communicate this to their health care providers, and, as such, it was not documented in the medical record. The potential development of COVID-19 rebound “is something to counsel your patients about.” COVID-19 rebound is a phenomenon that is being most commonly observed with nirmatrelvir-ritonavir as outpatient treatment.
Possible confounding factors in study
Katie Bechman, MBChB, clinical lecturer in rheumatology at King’s College London, who coauthored an accompanying editorial about the study and its findings, pointed out that the study is limited by its observational design.
“With any study that looks at the efficacy of treatment, especially in an observational cohort, you’re going to have to consider the unmeasured confounding and the difference between these two groups,” Dr. Bechman said. “I know that they did try to adjust for that in this study, but there’s always going to be factors that we can’t [control for]. That is something that needs to be considered. I think that’s always something we need to consider when we’re looking at observational data.”
In lieu of a randomized, controlled trial, Dr. Bechman noted that the study and its associated findings serve as “the best data we have,” and she described the results as “very informative and positive.”
She added that the large number of patients represents a strength of the study, as does the robust method employed for identifying which patients had COVID-19.
The learnings from this study with respect to outpatient treatment can be applied to more common illnesses that patients with rheumatic disease may develop, such as the flu, according to Dr. Bechman.
“One of the positive aspects from this pandemic is that we’ve learned a huge amount about how best to treat certain viruses and prevent them in patients,” she said. “It would be worth thinking towards the future, what we can do for illnesses that we see very commonly in these populations. There may be treatment regimens that we haven’t really considered until now. You could hypothesize that in the next couple of years, if we have an influenza breakout, that we should be providing some prehospital antiviral treatment to patients, especially the ones that are at high risk.”
The study was conducted without outside funding. Dr. Sparks has received research support from Bristol-Myers Squibb and consulted for AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer unrelated to this work. Dr. Bechman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Outpatient COVID-19 treatment with monoclonal antibodies or antiretroviral medications such as nirmatrelvir-ritonavir (Paxlovid) administered to patients with systemic autoimmune rheumatic disease led to lower odds of having severe outcomes when compared with similar patients who received no outpatient treatment in a real-world, retrospective analysis of cases.
The investigators found that there were nine hospitalizations or deaths (2.1%) among 426 patients who received outpatient treatment, compared with 49 (17.6%) among 278 who did not receive outpatient treatment, yielding an odds ratio of 0.12 (95% confidence interval, 0.05-0.25), after adjusting for age, sex, race, comorbidities, and kidney function. The study was published in Lancet Rheumatology.
“Across the board, there was a really strong association with receiving outpatient treatment and lower risk of severe COVID-19,” senior author Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine, Harvard Medical School and Brigham and Women’s Hospital, Boston, said in an interview. “It is pretty powerful evidence that, in this high-risk group, that treatment still matters related to preventing severe COVID. We found almost all patients who had severe COVID-19, either hospitalized or who had died, were in the untreated group.”
Early outpatient treatment an important tool in patients with rheumatic disease
Dr. Sparks noted that he and his coinvestigators conducted the study because the benefit of outpatient COVID-19 treatments in individuals with systemic autoimmune rheumatic disease was not adequately determined in clinical trials because they had infrequent enrollment of such patients.
The analysis included 704 patients with a mean age of 58.4 years who were seen at Mass General Brigham Integrated Health Care System, a multicenter health care system that includes 14 hospitals and primary care or specialty outpatient centers in the Boston area. A majority were female (76%) and White (84%). Nearly half had rheumatoid arthritis. Of the 704, 426 (61%) received outpatient treatment, which included nirmatrelvir-ritonavir (n = 307), monoclonal antibodies (n = 105), molnupiravir (n = 5), remdesivir (n = 3), and combination treatment (n = 6).
The findings underline the need to individualize approaches to outpatient treatment in those who test positive for SARS-CoV-2 to fend off severe COVID-19, according to Dr. Sparks. “It seems if you are vaccinated and in the general population that you are way less likely to have severe COVID-19 in the current environment, but that doesn’t necessarily apply to some high-risk groups like patients on immunosuppression. There are still patients at risk of severe COVID-19, and some of them are in this group of rheumatic patients. This should be part of the discussion related to deciding whether or not to treat.”
Dr. Sparks noted that vaccination against COVID-19 confers protection against developing severe COVID-19 in patients with rheumatic disease as it does in the general population, but patients with rheumatic diseases remain at increased risk for severe presentation. “Certainly, the vaccines really help our patients too, but there’s still a bit of a gap between the risk for our patients with rheumatic diseases and the general population” in developing severe COVID-19.
Dr. Sparks said he hopes the results represent a “call to action” that even among vaccinated patients there are still some who have poor outcomes, and that early outpatient treatment appears to be an important tool in the fight against poor outcomes from SARS-CoV-2 infection.
COVID-19 rebound
The study also reported on the phenomenon of COVID-19 rebound (recurrence of symptoms and test positivity after regimen completion) after oral outpatient SARS-CoV-2 treatment. “This [COVID-19 rebound] is a downside to treatment,” he said. COVID rebound was not infrequent: A total of 25 (8%) of 318 patients who received oral outpatient treatment had documented COVID-19 rebound.
“It was reassuring because we found no one who had rebound progressed to have severe COVID-19,” Dr. Sparks said. “On the other hand, [rebound] happened pretty frequently in our data, as 8% of patients are documented to have it.”
Dr. Sparks said he and coinvestigators speculate that more patients in the cohort may have experienced COVID-19 rebound but did not communicate this to their health care providers, and, as such, it was not documented in the medical record. The potential development of COVID-19 rebound “is something to counsel your patients about.” COVID-19 rebound is a phenomenon that is being most commonly observed with nirmatrelvir-ritonavir as outpatient treatment.
Possible confounding factors in study
Katie Bechman, MBChB, clinical lecturer in rheumatology at King’s College London, who coauthored an accompanying editorial about the study and its findings, pointed out that the study is limited by its observational design.
“With any study that looks at the efficacy of treatment, especially in an observational cohort, you’re going to have to consider the unmeasured confounding and the difference between these two groups,” Dr. Bechman said. “I know that they did try to adjust for that in this study, but there’s always going to be factors that we can’t [control for]. That is something that needs to be considered. I think that’s always something we need to consider when we’re looking at observational data.”
In lieu of a randomized, controlled trial, Dr. Bechman noted that the study and its associated findings serve as “the best data we have,” and she described the results as “very informative and positive.”
She added that the large number of patients represents a strength of the study, as does the robust method employed for identifying which patients had COVID-19.
The learnings from this study with respect to outpatient treatment can be applied to more common illnesses that patients with rheumatic disease may develop, such as the flu, according to Dr. Bechman.
“One of the positive aspects from this pandemic is that we’ve learned a huge amount about how best to treat certain viruses and prevent them in patients,” she said. “It would be worth thinking towards the future, what we can do for illnesses that we see very commonly in these populations. There may be treatment regimens that we haven’t really considered until now. You could hypothesize that in the next couple of years, if we have an influenza breakout, that we should be providing some prehospital antiviral treatment to patients, especially the ones that are at high risk.”
The study was conducted without outside funding. Dr. Sparks has received research support from Bristol-Myers Squibb and consulted for AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer unrelated to this work. Dr. Bechman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE LANCET RHEUMATOLOGY
Risk of infections low among kids receiving systemic meds for psoriasis, study finds
.
Those are key findings from what is believed to be the largest cohort study of its kind to estimate the 6-month rate of infections among children with psoriasis who started treatment with ustekinumab, etanercept, or methotrexate.
“Clinical trials have demonstrated high efficacy of new immunomodulatory agents in treating children with psoriasis,” lead author Maria C. Schneeweiss, MD, of the division of pharmacoepidemiology in the departments of medicine and dermatology at Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues wrote in the article, which was published online in JAMA Dermatology. “However, the risk of infections in clinical practice has not been fully characterized by comparing these medications against each other in pairwise comparisons.”
Drawing from two large U.S. insurance claims databases, the researchers identified 2,338 patients aged 17 years and younger who were receiving treatment with a topical medication for psoriasis and started new treatment with ustekinumab, etanercept, or methotrexate. They stratified their analysis by the time before pediatric labeling (2009-2015) and after pediatric approval (2016-2021), and their follow-up of patients started 1 day after initiating treatment and ended at 6 months.
Of the 2,338 patients, 1,368 (58%) were girls. From 2009 through 2021, 379 patients began treatment with ustekinumab, 779 patients began treatment with etanercept, and 1,180 patients began treatment with methotrexate. The propensity score–adjusted incidence rate of serious infection was 18.4 per 1,000 person-years (3 events) for those who used ustekinumab, 25.6 per 1,000 person-years (9 events) for those who used etanercept, and 14.9 per 1,000 person-years (8 events) for those who used methotrexate. The adjusted rate of outpatient infections was 254.9 per 1,000 person-years (39 events) for those who used ustekinumab, 435.7 per 1,000 person-years (139 events) for those who used etanercept, and 433.6 per 1,000 person-years (209 events) for those who used methotrexate. Meanwhile, the adjusted rate ratio of outpatient infections was 0.58 for ustekinumab vs. etanercept, 0.66 for ustekinumab vs. methotrexate, and 0.95 for etanercept vs. methotrexate. The researchers found that ratios were similar during the off-label use era and after pediatric labeling.
Anna L. Grossberg, MD, director of pediatric dermatology at the Johns Hopkins Children’s Center, Baltimore, who was asked to comment on the work, told this news organization that the data on outpatient infections in ustekinumab users “demonstrated that they may have a decreased risk of infection compared to pediatric psoriasis patients treated with methotrexate or the TNF-alpha inhibitor etanercept. This is previously unreported and reflects my personal experience with this medication in my own pediatric psoriasis patients.” She added the study’s overall findings lend further support to the safety of biologic medications and nonbiologic systemic immunomodulatory treatments for management of psoriasis. “This study will help guide pediatric dermatologists in counseling patients and their families about these risks [of infection], and in general providing reassurance that these risks appear to be quite low,” Dr. Grossberg said. “In particular, ustekinumab, a newer biologic medication that was recently FDA-approved for children 6 years and older for pediatric psoriasis, was not associated with higher infection rates than the other agents analyzed in this study, and in fact appears to carry a reduced risk compared to both etanercept and methotrexate.”
She noted certain limitations of the study, including its reliance on insurance claims data, “which can be limiting because information on possible confounding variables may not be known,” she said. “For example, the authors point out that environmental and behavioral risk factors for serious infection could not be evaluated or adjusted for, nor could the severity of the patients’ psoriasis. Additionally, this study only reported on outpatient infections that resulted in an antibiotic or other medications being prescribed and filled. It therefore may have missed children who presented with certain viral infections (examples could include the common cold and uncomplicated ear infections), which often will not require a prescription medication. Furthermore, it would fail to capture those who may have been seen for an infection but failed to fill the intended prescription.”
Dr. Schneeweiss reported receiving grants from AbbVie and UCB to Brigham and Women’s Hospital unrelated to the topic of this study and outside the submitted work. The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Grossberg reported having no financial disclosures.
.
Those are key findings from what is believed to be the largest cohort study of its kind to estimate the 6-month rate of infections among children with psoriasis who started treatment with ustekinumab, etanercept, or methotrexate.
“Clinical trials have demonstrated high efficacy of new immunomodulatory agents in treating children with psoriasis,” lead author Maria C. Schneeweiss, MD, of the division of pharmacoepidemiology in the departments of medicine and dermatology at Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues wrote in the article, which was published online in JAMA Dermatology. “However, the risk of infections in clinical practice has not been fully characterized by comparing these medications against each other in pairwise comparisons.”
Drawing from two large U.S. insurance claims databases, the researchers identified 2,338 patients aged 17 years and younger who were receiving treatment with a topical medication for psoriasis and started new treatment with ustekinumab, etanercept, or methotrexate. They stratified their analysis by the time before pediatric labeling (2009-2015) and after pediatric approval (2016-2021), and their follow-up of patients started 1 day after initiating treatment and ended at 6 months.
Of the 2,338 patients, 1,368 (58%) were girls. From 2009 through 2021, 379 patients began treatment with ustekinumab, 779 patients began treatment with etanercept, and 1,180 patients began treatment with methotrexate. The propensity score–adjusted incidence rate of serious infection was 18.4 per 1,000 person-years (3 events) for those who used ustekinumab, 25.6 per 1,000 person-years (9 events) for those who used etanercept, and 14.9 per 1,000 person-years (8 events) for those who used methotrexate. The adjusted rate of outpatient infections was 254.9 per 1,000 person-years (39 events) for those who used ustekinumab, 435.7 per 1,000 person-years (139 events) for those who used etanercept, and 433.6 per 1,000 person-years (209 events) for those who used methotrexate. Meanwhile, the adjusted rate ratio of outpatient infections was 0.58 for ustekinumab vs. etanercept, 0.66 for ustekinumab vs. methotrexate, and 0.95 for etanercept vs. methotrexate. The researchers found that ratios were similar during the off-label use era and after pediatric labeling.
Anna L. Grossberg, MD, director of pediatric dermatology at the Johns Hopkins Children’s Center, Baltimore, who was asked to comment on the work, told this news organization that the data on outpatient infections in ustekinumab users “demonstrated that they may have a decreased risk of infection compared to pediatric psoriasis patients treated with methotrexate or the TNF-alpha inhibitor etanercept. This is previously unreported and reflects my personal experience with this medication in my own pediatric psoriasis patients.” She added the study’s overall findings lend further support to the safety of biologic medications and nonbiologic systemic immunomodulatory treatments for management of psoriasis. “This study will help guide pediatric dermatologists in counseling patients and their families about these risks [of infection], and in general providing reassurance that these risks appear to be quite low,” Dr. Grossberg said. “In particular, ustekinumab, a newer biologic medication that was recently FDA-approved for children 6 years and older for pediatric psoriasis, was not associated with higher infection rates than the other agents analyzed in this study, and in fact appears to carry a reduced risk compared to both etanercept and methotrexate.”
She noted certain limitations of the study, including its reliance on insurance claims data, “which can be limiting because information on possible confounding variables may not be known,” she said. “For example, the authors point out that environmental and behavioral risk factors for serious infection could not be evaluated or adjusted for, nor could the severity of the patients’ psoriasis. Additionally, this study only reported on outpatient infections that resulted in an antibiotic or other medications being prescribed and filled. It therefore may have missed children who presented with certain viral infections (examples could include the common cold and uncomplicated ear infections), which often will not require a prescription medication. Furthermore, it would fail to capture those who may have been seen for an infection but failed to fill the intended prescription.”
Dr. Schneeweiss reported receiving grants from AbbVie and UCB to Brigham and Women’s Hospital unrelated to the topic of this study and outside the submitted work. The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Grossberg reported having no financial disclosures.
.
Those are key findings from what is believed to be the largest cohort study of its kind to estimate the 6-month rate of infections among children with psoriasis who started treatment with ustekinumab, etanercept, or methotrexate.
“Clinical trials have demonstrated high efficacy of new immunomodulatory agents in treating children with psoriasis,” lead author Maria C. Schneeweiss, MD, of the division of pharmacoepidemiology in the departments of medicine and dermatology at Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues wrote in the article, which was published online in JAMA Dermatology. “However, the risk of infections in clinical practice has not been fully characterized by comparing these medications against each other in pairwise comparisons.”
Drawing from two large U.S. insurance claims databases, the researchers identified 2,338 patients aged 17 years and younger who were receiving treatment with a topical medication for psoriasis and started new treatment with ustekinumab, etanercept, or methotrexate. They stratified their analysis by the time before pediatric labeling (2009-2015) and after pediatric approval (2016-2021), and their follow-up of patients started 1 day after initiating treatment and ended at 6 months.
Of the 2,338 patients, 1,368 (58%) were girls. From 2009 through 2021, 379 patients began treatment with ustekinumab, 779 patients began treatment with etanercept, and 1,180 patients began treatment with methotrexate. The propensity score–adjusted incidence rate of serious infection was 18.4 per 1,000 person-years (3 events) for those who used ustekinumab, 25.6 per 1,000 person-years (9 events) for those who used etanercept, and 14.9 per 1,000 person-years (8 events) for those who used methotrexate. The adjusted rate of outpatient infections was 254.9 per 1,000 person-years (39 events) for those who used ustekinumab, 435.7 per 1,000 person-years (139 events) for those who used etanercept, and 433.6 per 1,000 person-years (209 events) for those who used methotrexate. Meanwhile, the adjusted rate ratio of outpatient infections was 0.58 for ustekinumab vs. etanercept, 0.66 for ustekinumab vs. methotrexate, and 0.95 for etanercept vs. methotrexate. The researchers found that ratios were similar during the off-label use era and after pediatric labeling.
Anna L. Grossberg, MD, director of pediatric dermatology at the Johns Hopkins Children’s Center, Baltimore, who was asked to comment on the work, told this news organization that the data on outpatient infections in ustekinumab users “demonstrated that they may have a decreased risk of infection compared to pediatric psoriasis patients treated with methotrexate or the TNF-alpha inhibitor etanercept. This is previously unreported and reflects my personal experience with this medication in my own pediatric psoriasis patients.” She added the study’s overall findings lend further support to the safety of biologic medications and nonbiologic systemic immunomodulatory treatments for management of psoriasis. “This study will help guide pediatric dermatologists in counseling patients and their families about these risks [of infection], and in general providing reassurance that these risks appear to be quite low,” Dr. Grossberg said. “In particular, ustekinumab, a newer biologic medication that was recently FDA-approved for children 6 years and older for pediatric psoriasis, was not associated with higher infection rates than the other agents analyzed in this study, and in fact appears to carry a reduced risk compared to both etanercept and methotrexate.”
She noted certain limitations of the study, including its reliance on insurance claims data, “which can be limiting because information on possible confounding variables may not be known,” she said. “For example, the authors point out that environmental and behavioral risk factors for serious infection could not be evaluated or adjusted for, nor could the severity of the patients’ psoriasis. Additionally, this study only reported on outpatient infections that resulted in an antibiotic or other medications being prescribed and filled. It therefore may have missed children who presented with certain viral infections (examples could include the common cold and uncomplicated ear infections), which often will not require a prescription medication. Furthermore, it would fail to capture those who may have been seen for an infection but failed to fill the intended prescription.”
Dr. Schneeweiss reported receiving grants from AbbVie and UCB to Brigham and Women’s Hospital unrelated to the topic of this study and outside the submitted work. The study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Dr. Grossberg reported having no financial disclosures.
FROM JAMA DERMATOLOGY
Must-read acute care medicine articles from 2022
When 2022 began, we started seeing some light at the end of the COVID-19 tunnel. Vaccines were widely available, and even with new variants of the virus still occasionally emerging, the rates of severe morbidity and mortality appeared to be decreasing.
Expectedly, journals appeared to start moving more toward mainstream topics and publications rather than what seemed like a major focus on COVID-19 publications. The resulting literature was fantastic.
Several of those topics were discussed in a prior Emergency Medicine Viewpoint from this news organization, and many more of the research advances of 2022 will be discussed in the near future. However, in this Viewpoint, I would like to present my annual review of my three “must-read” articles of the past year.
As in past years, I am choosing reviews of the literature rather than original research articles (which, all too often, become outdated or debunked within a few years). I choose these articles in the hopes that readers will not simply settle for my brief reviews of the key points but instead will feel compelled to download and read the entire articles. These publications address common conditions and quandaries we face in the daily practice of emergency medicine and are practice-changing.
Myocardial dysfunction after cardiac arrest: Tips and pitfalls
The management of post–cardiac arrest patients remains a hot topic in the resuscitation literature as we continue to understand that the immediate post-arrest period is critical to patient outcome.
Ortuno and colleagues reviewed the current literature on post-arrest care and wrote an outstanding summary of how to optimally care for these patients. More specifically, they focused on post-arrest patients who demonstrate continued shock, or “post–cardiac arrest myocardial dysfunction” (PCAMD).
They propose three mechanisms for the pathogenesis of PCAMD: ischemia reperfusion phenomenon, systemic inflammatory response, and increased catecholamine release
I will skip through the details of the pathophysiology that they describe in the article, but I certainly do recommend that everyone review their descriptions.
Management of these patients begins with a good hemodynamic assessment, which includes clinical markers of perfusion (blood pressure, capillary refill), ECG, and point-of-care ultrasound (POCUS). If the initial assessment reveals an obvious cause of the cardiac arrest (e.g., massive pulmonary embolism, myocardial infarction, pericardial tamponade), then the underlying cause should be treated expeditiously.
In the absence of an obvious treatable cause of the shock, the fluid status and cardiac function should be addressed with POCUS. If the patient is hypovolemic, intravenous fluids should be administered. If the fluid status is adequate, POCUS should be used to estimate the patient’s ventricular function. If the ventricle appears to be hyperdynamic with good contractility, shock should be treated with norepinephrine. On the other hand, if the ventricle is hypodynamic, dobutamine should be substituted for norepinephrine or, more often, added to norepinephrine.
The above represents a simplified summary of the critical points, but the authors do delve into further detail and also discuss some other options for therapies, including steroids, coronary revascularization, extracorporeal membrane oxygenation, and so on. The review is very thoughtful, thorough, and definitely worth a full read.
Top myths of diagnosis and management of infectious diseases in hospital medicine
Most, if not all of us in medicine, have heard the saying that 50% of what we learn in medical school (or residency) will turn out to be wrong. I certainly believe in this concept and consequently, like many of you, I enjoy reading about myths and misconceptions that we have been taught. With that in mind, I have to say that I love this article because it seems to have been written specifically to address what I was taught!
This author group, consisting mostly of clinical PharmDs who are experts in antibiotic use, provide us with an evidence-based discussion of myths and pitfalls in how antibiotics are often used in current clinical practice. The authors review their top 10 myths involving the use of antibiotics in treating infections in the hospital setting. A few of these relate more to the inpatient setting, but here are my favorite emergency department (ED)–related myths that they address:
- “Antibiotics do no harm.” The authors address the risk-benefit of antibiotics based on assumed vs. confirmed infections, including a brief discussion of adverse drug effects.
- “Antibiotic durations of 7, 14, or 21 days are typically necessary.” The authors address appropriate duration of antibiotic use and the fact that unnecessarily long durations of use can lead to resistance. They also provide reassurance that some infections can be treated with quite short durations of antibiotics.
- “If one drug is good, two (or more!) is better.” The use of multiple antibiotics, often with overlapping bacterial coverage, is rampant in medicine and further increases the risk for adverse drug effects and resistance.
- “Oral antibiotics are not as good as intravenous antibiotics for hospitalized patients.” This is definitely a myth that I learned. I recall being taught by many senior physicians that anyone sick enough for admission should be treated with intravenous antibiotics. As it turns out, absorption and effectiveness of most oral antibiotics is just as good as intravenous antibiotics, and the oral formulations are often safer.
- “A history of a penicillin allergy means the patient can never receive a beta-lactam antibiotic.” This is a myth that was debunked quite a few years ago, but it seems that many clinicians still need a reminder.
The authors included five more myths that are worth the read. This is an article that needs to be disseminated among all hospital clinicians.
Guidelines for low-risk, recurrent abdominal pain in the emergency department
The Society for Academic Emergency Medicine (SAEM) recently initiated a program focused on creating evidence-based approaches to challenging chief complaints and presentations in the emergency department (ED). In 2021, they published an approach to managing patients with recurrent, low-risk chest pain in the ED. This past year, they published their second guideline, focused on the management of patients with low-risk, recurrent abdominal pain in the ED.
Recurrent low-risk abdominal pain is a common and vexing presentation to EDs around the world, and there is little prior published guidance. Do all of these patients need repeat imaging? How do we manage their pain? Are there nonabdominal conditions that should be considered?
Broder and colleagues did a fantastic review of the current literature and, on behalf of SAEM, have provided a rational approach to optimal management of these patients. The four major questions they addressed, with brief summaries of their recommendations, are:
- Should adult ED patients with low-risk, recurrent and previously undifferentiated abdominal pain receive a repeat CT abdomen-pelvis (CTAP) after a negative CTAP within the past 12 months? This is a typical question that we all ponder when managing these patients. Unfortunately, the writing group found insufficient evidence to definitively identify populations in whom CTAP was recommended vs could be safely withheld. It is a bit disappointing that there is no definite answer to the question. On the other hand, it is reassuring to know that the world’s best evidence essentially says that it is perfectly appropriate to use your own good clinical judgment.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain with a negative CTAP receive additional imaging with abdominal ultrasound? In this case, the writing group found enough evidence, though low-level, to suggest against routine ultrasound in the absence of concern specifically for pelvic or hepatobiliary pathology. Like most tests, ultrasound is best used when there are specific concerns rather than being used in an undifferentiated fashion.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain receive screening for depression/anxiety? The writing group found enough evidence, though low-level again, to suggest that screening for depression and/or anxiety be performed during the ED evaluation. This could lead to successful therapy for the abdominal pain.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain receive nonopioid and/or nonpharmacologic analgesics? The writing group found little evidence to suggest for or against these analgesics, but they made a consensus recommendation suggesting an opioid-minimizing strategy for pain control.
Although the final recommendations of the writing group were not definitive or based on the strongest level of evidence, I find it helpful to have this guidance, nevertheless, on behalf of a major national organization. I also find it helpful to know that even with the best evidence available, optimal patient care will often boil down to physician experience and gestalt. I should also add that the overall article is chock-full of pearls and helpful information that will further inform the readers’ decisions, and so the full version is definitely worth the read.
In summary
There you have it – my three favorite practice-changing articles of 2022. Although I have tried to provide key points here, the full discussions of those key points in the published articles will provide a great deal more education than I can offer in this brief write-up, and so I strongly encourage everyone to read the full versions. Please be sure to include in the comments section your own pick for favorite or must-read articles from the past year.
Amal Mattu, MD, is a professor, vice chair of education, and codirector of the emergency cardiology fellowship in the department of emergency medicine at the University of Maryland, Baltimore. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
When 2022 began, we started seeing some light at the end of the COVID-19 tunnel. Vaccines were widely available, and even with new variants of the virus still occasionally emerging, the rates of severe morbidity and mortality appeared to be decreasing.
Expectedly, journals appeared to start moving more toward mainstream topics and publications rather than what seemed like a major focus on COVID-19 publications. The resulting literature was fantastic.
Several of those topics were discussed in a prior Emergency Medicine Viewpoint from this news organization, and many more of the research advances of 2022 will be discussed in the near future. However, in this Viewpoint, I would like to present my annual review of my three “must-read” articles of the past year.
As in past years, I am choosing reviews of the literature rather than original research articles (which, all too often, become outdated or debunked within a few years). I choose these articles in the hopes that readers will not simply settle for my brief reviews of the key points but instead will feel compelled to download and read the entire articles. These publications address common conditions and quandaries we face in the daily practice of emergency medicine and are practice-changing.
Myocardial dysfunction after cardiac arrest: Tips and pitfalls
The management of post–cardiac arrest patients remains a hot topic in the resuscitation literature as we continue to understand that the immediate post-arrest period is critical to patient outcome.
Ortuno and colleagues reviewed the current literature on post-arrest care and wrote an outstanding summary of how to optimally care for these patients. More specifically, they focused on post-arrest patients who demonstrate continued shock, or “post–cardiac arrest myocardial dysfunction” (PCAMD).
They propose three mechanisms for the pathogenesis of PCAMD: ischemia reperfusion phenomenon, systemic inflammatory response, and increased catecholamine release
I will skip through the details of the pathophysiology that they describe in the article, but I certainly do recommend that everyone review their descriptions.
Management of these patients begins with a good hemodynamic assessment, which includes clinical markers of perfusion (blood pressure, capillary refill), ECG, and point-of-care ultrasound (POCUS). If the initial assessment reveals an obvious cause of the cardiac arrest (e.g., massive pulmonary embolism, myocardial infarction, pericardial tamponade), then the underlying cause should be treated expeditiously.
In the absence of an obvious treatable cause of the shock, the fluid status and cardiac function should be addressed with POCUS. If the patient is hypovolemic, intravenous fluids should be administered. If the fluid status is adequate, POCUS should be used to estimate the patient’s ventricular function. If the ventricle appears to be hyperdynamic with good contractility, shock should be treated with norepinephrine. On the other hand, if the ventricle is hypodynamic, dobutamine should be substituted for norepinephrine or, more often, added to norepinephrine.
The above represents a simplified summary of the critical points, but the authors do delve into further detail and also discuss some other options for therapies, including steroids, coronary revascularization, extracorporeal membrane oxygenation, and so on. The review is very thoughtful, thorough, and definitely worth a full read.
Top myths of diagnosis and management of infectious diseases in hospital medicine
Most, if not all of us in medicine, have heard the saying that 50% of what we learn in medical school (or residency) will turn out to be wrong. I certainly believe in this concept and consequently, like many of you, I enjoy reading about myths and misconceptions that we have been taught. With that in mind, I have to say that I love this article because it seems to have been written specifically to address what I was taught!
This author group, consisting mostly of clinical PharmDs who are experts in antibiotic use, provide us with an evidence-based discussion of myths and pitfalls in how antibiotics are often used in current clinical practice. The authors review their top 10 myths involving the use of antibiotics in treating infections in the hospital setting. A few of these relate more to the inpatient setting, but here are my favorite emergency department (ED)–related myths that they address:
- “Antibiotics do no harm.” The authors address the risk-benefit of antibiotics based on assumed vs. confirmed infections, including a brief discussion of adverse drug effects.
- “Antibiotic durations of 7, 14, or 21 days are typically necessary.” The authors address appropriate duration of antibiotic use and the fact that unnecessarily long durations of use can lead to resistance. They also provide reassurance that some infections can be treated with quite short durations of antibiotics.
- “If one drug is good, two (or more!) is better.” The use of multiple antibiotics, often with overlapping bacterial coverage, is rampant in medicine and further increases the risk for adverse drug effects and resistance.
- “Oral antibiotics are not as good as intravenous antibiotics for hospitalized patients.” This is definitely a myth that I learned. I recall being taught by many senior physicians that anyone sick enough for admission should be treated with intravenous antibiotics. As it turns out, absorption and effectiveness of most oral antibiotics is just as good as intravenous antibiotics, and the oral formulations are often safer.
- “A history of a penicillin allergy means the patient can never receive a beta-lactam antibiotic.” This is a myth that was debunked quite a few years ago, but it seems that many clinicians still need a reminder.
The authors included five more myths that are worth the read. This is an article that needs to be disseminated among all hospital clinicians.
Guidelines for low-risk, recurrent abdominal pain in the emergency department
The Society for Academic Emergency Medicine (SAEM) recently initiated a program focused on creating evidence-based approaches to challenging chief complaints and presentations in the emergency department (ED). In 2021, they published an approach to managing patients with recurrent, low-risk chest pain in the ED. This past year, they published their second guideline, focused on the management of patients with low-risk, recurrent abdominal pain in the ED.
Recurrent low-risk abdominal pain is a common and vexing presentation to EDs around the world, and there is little prior published guidance. Do all of these patients need repeat imaging? How do we manage their pain? Are there nonabdominal conditions that should be considered?
Broder and colleagues did a fantastic review of the current literature and, on behalf of SAEM, have provided a rational approach to optimal management of these patients. The four major questions they addressed, with brief summaries of their recommendations, are:
- Should adult ED patients with low-risk, recurrent and previously undifferentiated abdominal pain receive a repeat CT abdomen-pelvis (CTAP) after a negative CTAP within the past 12 months? This is a typical question that we all ponder when managing these patients. Unfortunately, the writing group found insufficient evidence to definitively identify populations in whom CTAP was recommended vs could be safely withheld. It is a bit disappointing that there is no definite answer to the question. On the other hand, it is reassuring to know that the world’s best evidence essentially says that it is perfectly appropriate to use your own good clinical judgment.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain with a negative CTAP receive additional imaging with abdominal ultrasound? In this case, the writing group found enough evidence, though low-level, to suggest against routine ultrasound in the absence of concern specifically for pelvic or hepatobiliary pathology. Like most tests, ultrasound is best used when there are specific concerns rather than being used in an undifferentiated fashion.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain receive screening for depression/anxiety? The writing group found enough evidence, though low-level again, to suggest that screening for depression and/or anxiety be performed during the ED evaluation. This could lead to successful therapy for the abdominal pain.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain receive nonopioid and/or nonpharmacologic analgesics? The writing group found little evidence to suggest for or against these analgesics, but they made a consensus recommendation suggesting an opioid-minimizing strategy for pain control.
Although the final recommendations of the writing group were not definitive or based on the strongest level of evidence, I find it helpful to have this guidance, nevertheless, on behalf of a major national organization. I also find it helpful to know that even with the best evidence available, optimal patient care will often boil down to physician experience and gestalt. I should also add that the overall article is chock-full of pearls and helpful information that will further inform the readers’ decisions, and so the full version is definitely worth the read.
In summary
There you have it – my three favorite practice-changing articles of 2022. Although I have tried to provide key points here, the full discussions of those key points in the published articles will provide a great deal more education than I can offer in this brief write-up, and so I strongly encourage everyone to read the full versions. Please be sure to include in the comments section your own pick for favorite or must-read articles from the past year.
Amal Mattu, MD, is a professor, vice chair of education, and codirector of the emergency cardiology fellowship in the department of emergency medicine at the University of Maryland, Baltimore. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
When 2022 began, we started seeing some light at the end of the COVID-19 tunnel. Vaccines were widely available, and even with new variants of the virus still occasionally emerging, the rates of severe morbidity and mortality appeared to be decreasing.
Expectedly, journals appeared to start moving more toward mainstream topics and publications rather than what seemed like a major focus on COVID-19 publications. The resulting literature was fantastic.
Several of those topics were discussed in a prior Emergency Medicine Viewpoint from this news organization, and many more of the research advances of 2022 will be discussed in the near future. However, in this Viewpoint, I would like to present my annual review of my three “must-read” articles of the past year.
As in past years, I am choosing reviews of the literature rather than original research articles (which, all too often, become outdated or debunked within a few years). I choose these articles in the hopes that readers will not simply settle for my brief reviews of the key points but instead will feel compelled to download and read the entire articles. These publications address common conditions and quandaries we face in the daily practice of emergency medicine and are practice-changing.
Myocardial dysfunction after cardiac arrest: Tips and pitfalls
The management of post–cardiac arrest patients remains a hot topic in the resuscitation literature as we continue to understand that the immediate post-arrest period is critical to patient outcome.
Ortuno and colleagues reviewed the current literature on post-arrest care and wrote an outstanding summary of how to optimally care for these patients. More specifically, they focused on post-arrest patients who demonstrate continued shock, or “post–cardiac arrest myocardial dysfunction” (PCAMD).
They propose three mechanisms for the pathogenesis of PCAMD: ischemia reperfusion phenomenon, systemic inflammatory response, and increased catecholamine release
I will skip through the details of the pathophysiology that they describe in the article, but I certainly do recommend that everyone review their descriptions.
Management of these patients begins with a good hemodynamic assessment, which includes clinical markers of perfusion (blood pressure, capillary refill), ECG, and point-of-care ultrasound (POCUS). If the initial assessment reveals an obvious cause of the cardiac arrest (e.g., massive pulmonary embolism, myocardial infarction, pericardial tamponade), then the underlying cause should be treated expeditiously.
In the absence of an obvious treatable cause of the shock, the fluid status and cardiac function should be addressed with POCUS. If the patient is hypovolemic, intravenous fluids should be administered. If the fluid status is adequate, POCUS should be used to estimate the patient’s ventricular function. If the ventricle appears to be hyperdynamic with good contractility, shock should be treated with norepinephrine. On the other hand, if the ventricle is hypodynamic, dobutamine should be substituted for norepinephrine or, more often, added to norepinephrine.
The above represents a simplified summary of the critical points, but the authors do delve into further detail and also discuss some other options for therapies, including steroids, coronary revascularization, extracorporeal membrane oxygenation, and so on. The review is very thoughtful, thorough, and definitely worth a full read.
Top myths of diagnosis and management of infectious diseases in hospital medicine
Most, if not all of us in medicine, have heard the saying that 50% of what we learn in medical school (or residency) will turn out to be wrong. I certainly believe in this concept and consequently, like many of you, I enjoy reading about myths and misconceptions that we have been taught. With that in mind, I have to say that I love this article because it seems to have been written specifically to address what I was taught!
This author group, consisting mostly of clinical PharmDs who are experts in antibiotic use, provide us with an evidence-based discussion of myths and pitfalls in how antibiotics are often used in current clinical practice. The authors review their top 10 myths involving the use of antibiotics in treating infections in the hospital setting. A few of these relate more to the inpatient setting, but here are my favorite emergency department (ED)–related myths that they address:
- “Antibiotics do no harm.” The authors address the risk-benefit of antibiotics based on assumed vs. confirmed infections, including a brief discussion of adverse drug effects.
- “Antibiotic durations of 7, 14, or 21 days are typically necessary.” The authors address appropriate duration of antibiotic use and the fact that unnecessarily long durations of use can lead to resistance. They also provide reassurance that some infections can be treated with quite short durations of antibiotics.
- “If one drug is good, two (or more!) is better.” The use of multiple antibiotics, often with overlapping bacterial coverage, is rampant in medicine and further increases the risk for adverse drug effects and resistance.
- “Oral antibiotics are not as good as intravenous antibiotics for hospitalized patients.” This is definitely a myth that I learned. I recall being taught by many senior physicians that anyone sick enough for admission should be treated with intravenous antibiotics. As it turns out, absorption and effectiveness of most oral antibiotics is just as good as intravenous antibiotics, and the oral formulations are often safer.
- “A history of a penicillin allergy means the patient can never receive a beta-lactam antibiotic.” This is a myth that was debunked quite a few years ago, but it seems that many clinicians still need a reminder.
The authors included five more myths that are worth the read. This is an article that needs to be disseminated among all hospital clinicians.
Guidelines for low-risk, recurrent abdominal pain in the emergency department
The Society for Academic Emergency Medicine (SAEM) recently initiated a program focused on creating evidence-based approaches to challenging chief complaints and presentations in the emergency department (ED). In 2021, they published an approach to managing patients with recurrent, low-risk chest pain in the ED. This past year, they published their second guideline, focused on the management of patients with low-risk, recurrent abdominal pain in the ED.
Recurrent low-risk abdominal pain is a common and vexing presentation to EDs around the world, and there is little prior published guidance. Do all of these patients need repeat imaging? How do we manage their pain? Are there nonabdominal conditions that should be considered?
Broder and colleagues did a fantastic review of the current literature and, on behalf of SAEM, have provided a rational approach to optimal management of these patients. The four major questions they addressed, with brief summaries of their recommendations, are:
- Should adult ED patients with low-risk, recurrent and previously undifferentiated abdominal pain receive a repeat CT abdomen-pelvis (CTAP) after a negative CTAP within the past 12 months? This is a typical question that we all ponder when managing these patients. Unfortunately, the writing group found insufficient evidence to definitively identify populations in whom CTAP was recommended vs could be safely withheld. It is a bit disappointing that there is no definite answer to the question. On the other hand, it is reassuring to know that the world’s best evidence essentially says that it is perfectly appropriate to use your own good clinical judgment.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain with a negative CTAP receive additional imaging with abdominal ultrasound? In this case, the writing group found enough evidence, though low-level, to suggest against routine ultrasound in the absence of concern specifically for pelvic or hepatobiliary pathology. Like most tests, ultrasound is best used when there are specific concerns rather than being used in an undifferentiated fashion.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain receive screening for depression/anxiety? The writing group found enough evidence, though low-level again, to suggest that screening for depression and/or anxiety be performed during the ED evaluation. This could lead to successful therapy for the abdominal pain.
- Should adult ED patients with low-risk, recurrent, and previously undifferentiated abdominal pain receive nonopioid and/or nonpharmacologic analgesics? The writing group found little evidence to suggest for or against these analgesics, but they made a consensus recommendation suggesting an opioid-minimizing strategy for pain control.
Although the final recommendations of the writing group were not definitive or based on the strongest level of evidence, I find it helpful to have this guidance, nevertheless, on behalf of a major national organization. I also find it helpful to know that even with the best evidence available, optimal patient care will often boil down to physician experience and gestalt. I should also add that the overall article is chock-full of pearls and helpful information that will further inform the readers’ decisions, and so the full version is definitely worth the read.
In summary
There you have it – my three favorite practice-changing articles of 2022. Although I have tried to provide key points here, the full discussions of those key points in the published articles will provide a great deal more education than I can offer in this brief write-up, and so I strongly encourage everyone to read the full versions. Please be sure to include in the comments section your own pick for favorite or must-read articles from the past year.
Amal Mattu, MD, is a professor, vice chair of education, and codirector of the emergency cardiology fellowship in the department of emergency medicine at the University of Maryland, Baltimore. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The five biggest changes in the 2023 adult vaccine schedules
This transcript has been edited for clarity.
Hello. I’m Dr Sandra Fryhofer. Welcome to Medicine Matters.
It’s a new year, which means a new ACIP adult immunization schedule – a valuable resource collating ACIP’s most up-to-date vaccination recommendations.
Here are this year’s five most important changes:
- COVID vaccines now front and center
- New emphasis on polio vaccination
- Inclusion of some nonvaccine products (such as monoclonal antibody products)
- Pharmacists group has approved the schedule for the first time
- New shared clinical decision-making option for pneumococcal vaccines
The schedule’s organization remains the same. It still has four sections:
- Table 1: vaccinations by age
- Table 2: vaccinations by medical condition and other indications
- The Notes section (alphabetically ordered by vaccine type)
- Appendix listing of vaccine-specific contraindications and precautions
But what’s unique this year is that some of the abbreviations have historical implications. The first change is no big surprise in light of what we’ve gone through in the past few years. COVID vaccines are listed first on the cover page by brand name for those authorized and by company name for those still under US emergency use authorization. They’re also listed first on the graphics and in the notes.
COVID and mRNA and protein-based vaccines have now been assigned official abbreviations based on vaccine platform and valency.
- 1vCOV-mRNA: Comirnaty/Pfizer-BioNTech and Spikevax Moderna COVID-19 vaccines
- 2vCOV-mRNA: Pfizer-BioNTech and Moderna bivalent COVID-19 vaccines
- 1vCOV-aPS: Novavax COVID-19 vaccine
Also remarkable is the absence of COVID viral vector vaccines on the list. However, the viral vector COVID vaccine (which has been available but is not preferred) does have a CDC website link in the Notes section.
A sad but necessary inclusion was triggered by recent polio cases in New York. Polio was believed to be eradicated, and we thought adults no longer needed to be vaccinated against polio. In the new schedule, the polio vaccine is listed on the cover page but is not included in the tables. Current polio vaccination recommendations are now in the Notes section.
Also of historical significance and something that may set a precedent is the inclusion of nonvaccine products. The value of COVID preexposure prophylaxis with products including monoclonal antibodies (such as Evusheld) for people who are moderately or severely immunocompromised is mentioned in the Notes section.
For the first time ever, the schedule has been approved by the American Pharmacists Association, which validates pharmacists as established partners in vaccine administration.
Color-code key
One aspect of the schedule that has not changed is the color-code key:
- Yellow: Recommended if the patient meets the age requirement
- Purple: Indicated for those with additional risk factors or another indication
- Blue: Recommended based on shared clinical decision-making
- Orange: Precaution
- Red: Contraindicated or not recommended; the vaccine should not be administered. Overlays on the red more precisely clarify whether a vaccine is really contraindicated or just not recommended. An asterisk on red means vaccinate after pregnancy if indicated.
- Gray: No recommendation or not applicable
Vaccinations by age
Table 1 lists recommended vaccinations by age. There is one major change. COVID vaccines are on the first row of the graphic, with the need for both a primary series and boosters emphasized on the overlay. The notes have hyperlinks to the most up-to-date COVID vaccination recommendations.
Pneumococcal vaccination. Pneumococcal vaccination is routinely recommended starting at age 65. Current recommendations for those not previously vaccinated have not changed since last year. But on Table 1, the bottom half of the row for those 65 or older is now blue (and that’s new). This new color blue means shared clinical decision-making and applies to people who were previously considered fully vaccinated with the now extinct combination of PCV13 and PPSV23. These patients now have the option of getting a dose of PCV20 five years after completing their PCV13-PPSV23 combo series. This option is blue because the decision is up to you and your patient.
Check the notes for more pneumococcal vaccination details. For example, for those partially vaccinated using lower valency vaccines, there’s an option of substituting PCV20 for PPSV23 to broaden and increase durability of protection.
The pneumococcal vaccination recommendation options are complicated. A new pneumococcal vaccination app can help.
Hepatitis B. For adults under age 60, the color code for the hepatitis B vaccine is yellow, meaning it’s indicated for all. For older patients, the color code is purple. If a patient who is age 60 or older wants the hepatitis B vaccine, they can have it even in the absence of additional risk indications.
Vaccinations by medical condition or other indications
Other than a few minor word changes on the overlay, the only thing that’s new is the COVID vaccine row.
This table is helpful for matching vaccine recommendations with specific medical conditions, including pregnancy, immunocompromise, HIV (with specifics according to CD4 count), asplenia, complement deficiencies, heart disease, lung disease, alcoholism, chronic liver disease, diabetes, health care personnel, and men who have sex with men.
Use this table to dot the i’s and cross the t’s when it comes to vaccination recommendations. For example, take a look at the pregnancy column. Live virus vaccines, including LAIV, MMR, and varicella, are contraindicated and color-coded red. MMR and varicella also have an asterisk, meaning vaccinate after pregnancy if indicated. HPV vaccines are not live virus vaccines, but the overlay says they are not recommended during pregnancy. The asterisk indicates that you can vaccinate after pregnancy.
Vaccine notes
The notes are in alphabetical order, and their organization (routine, special situations, and shared clinical decision-making when indicated) has not changed. They are concise and succinct, but sometimes they’re not enough. That’s why vaccine-specific links to more complete recommendations are so convenient.
Notes for hepatitis B contain nuances on specific dosing for vaccinating patients on dialysis, as well as a reminder that newer hepatitis C vaccines such as Heplisav and PreHevbrio are not recommended during pregnancy due to lack of safety data.
For influenza, everyone 6 months or older still needs yearly flu vaccination with an age- and health-appropriate flu vaccine. But for those aged 65 or older, the notes specify the three vaccine versions now preferred: high-dose, recombinant, or adjuvanted versions. However, if these aren’t available, it’s better to get any flu vaccine than to go without.
Under meningococcal vaccines, the notes for MenACWY and MenB are combined. For MenB, trade names Bexsero and Trumenba are specified because the products are not interchangeable. Booster intervals for those still at risk are different for each vaccine type: every 5 years for MenACWY boosters, and every 2-3 years for boosts of MenB.
The recent polio cases in New York have put polio vaccination in the spotlight. ACIP has now reinstated its Polio Vaccine Work Group. The new schedule lists polio vaccines on the cover page. Current recommendations have been added to the notes section. Routine vaccination for adults is not necessary, at least for now. However, those at increased risk for exposure to polio fall in the special-situation category. For those at increased risk who have completed a polio vaccine series, a single lifetime IPV booster can be given. For those at increased risk who have not completed their polio vaccine series, now would be the time to finish the series.
Appendix
The final step in using the new schedule is checking the appendix and its list of vaccine-specific contraindications and precautions.
I hope this review of the new ACIP adult immunization schedule has been helpful. For Medicine Matters, I’m Dr. Sandra Fryhofer.
Dr. Fryhofer is clinical associate professor of medicine, Emory University, Atlanta. She reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hello. I’m Dr Sandra Fryhofer. Welcome to Medicine Matters.
It’s a new year, which means a new ACIP adult immunization schedule – a valuable resource collating ACIP’s most up-to-date vaccination recommendations.
Here are this year’s five most important changes:
- COVID vaccines now front and center
- New emphasis on polio vaccination
- Inclusion of some nonvaccine products (such as monoclonal antibody products)
- Pharmacists group has approved the schedule for the first time
- New shared clinical decision-making option for pneumococcal vaccines
The schedule’s organization remains the same. It still has four sections:
- Table 1: vaccinations by age
- Table 2: vaccinations by medical condition and other indications
- The Notes section (alphabetically ordered by vaccine type)
- Appendix listing of vaccine-specific contraindications and precautions
But what’s unique this year is that some of the abbreviations have historical implications. The first change is no big surprise in light of what we’ve gone through in the past few years. COVID vaccines are listed first on the cover page by brand name for those authorized and by company name for those still under US emergency use authorization. They’re also listed first on the graphics and in the notes.
COVID and mRNA and protein-based vaccines have now been assigned official abbreviations based on vaccine platform and valency.
- 1vCOV-mRNA: Comirnaty/Pfizer-BioNTech and Spikevax Moderna COVID-19 vaccines
- 2vCOV-mRNA: Pfizer-BioNTech and Moderna bivalent COVID-19 vaccines
- 1vCOV-aPS: Novavax COVID-19 vaccine
Also remarkable is the absence of COVID viral vector vaccines on the list. However, the viral vector COVID vaccine (which has been available but is not preferred) does have a CDC website link in the Notes section.
A sad but necessary inclusion was triggered by recent polio cases in New York. Polio was believed to be eradicated, and we thought adults no longer needed to be vaccinated against polio. In the new schedule, the polio vaccine is listed on the cover page but is not included in the tables. Current polio vaccination recommendations are now in the Notes section.
Also of historical significance and something that may set a precedent is the inclusion of nonvaccine products. The value of COVID preexposure prophylaxis with products including monoclonal antibodies (such as Evusheld) for people who are moderately or severely immunocompromised is mentioned in the Notes section.
For the first time ever, the schedule has been approved by the American Pharmacists Association, which validates pharmacists as established partners in vaccine administration.
Color-code key
One aspect of the schedule that has not changed is the color-code key:
- Yellow: Recommended if the patient meets the age requirement
- Purple: Indicated for those with additional risk factors or another indication
- Blue: Recommended based on shared clinical decision-making
- Orange: Precaution
- Red: Contraindicated or not recommended; the vaccine should not be administered. Overlays on the red more precisely clarify whether a vaccine is really contraindicated or just not recommended. An asterisk on red means vaccinate after pregnancy if indicated.
- Gray: No recommendation or not applicable
Vaccinations by age
Table 1 lists recommended vaccinations by age. There is one major change. COVID vaccines are on the first row of the graphic, with the need for both a primary series and boosters emphasized on the overlay. The notes have hyperlinks to the most up-to-date COVID vaccination recommendations.
Pneumococcal vaccination. Pneumococcal vaccination is routinely recommended starting at age 65. Current recommendations for those not previously vaccinated have not changed since last year. But on Table 1, the bottom half of the row for those 65 or older is now blue (and that’s new). This new color blue means shared clinical decision-making and applies to people who were previously considered fully vaccinated with the now extinct combination of PCV13 and PPSV23. These patients now have the option of getting a dose of PCV20 five years after completing their PCV13-PPSV23 combo series. This option is blue because the decision is up to you and your patient.
Check the notes for more pneumococcal vaccination details. For example, for those partially vaccinated using lower valency vaccines, there’s an option of substituting PCV20 for PPSV23 to broaden and increase durability of protection.
The pneumococcal vaccination recommendation options are complicated. A new pneumococcal vaccination app can help.
Hepatitis B. For adults under age 60, the color code for the hepatitis B vaccine is yellow, meaning it’s indicated for all. For older patients, the color code is purple. If a patient who is age 60 or older wants the hepatitis B vaccine, they can have it even in the absence of additional risk indications.
Vaccinations by medical condition or other indications
Other than a few minor word changes on the overlay, the only thing that’s new is the COVID vaccine row.
This table is helpful for matching vaccine recommendations with specific medical conditions, including pregnancy, immunocompromise, HIV (with specifics according to CD4 count), asplenia, complement deficiencies, heart disease, lung disease, alcoholism, chronic liver disease, diabetes, health care personnel, and men who have sex with men.
Use this table to dot the i’s and cross the t’s when it comes to vaccination recommendations. For example, take a look at the pregnancy column. Live virus vaccines, including LAIV, MMR, and varicella, are contraindicated and color-coded red. MMR and varicella also have an asterisk, meaning vaccinate after pregnancy if indicated. HPV vaccines are not live virus vaccines, but the overlay says they are not recommended during pregnancy. The asterisk indicates that you can vaccinate after pregnancy.
Vaccine notes
The notes are in alphabetical order, and their organization (routine, special situations, and shared clinical decision-making when indicated) has not changed. They are concise and succinct, but sometimes they’re not enough. That’s why vaccine-specific links to more complete recommendations are so convenient.
Notes for hepatitis B contain nuances on specific dosing for vaccinating patients on dialysis, as well as a reminder that newer hepatitis C vaccines such as Heplisav and PreHevbrio are not recommended during pregnancy due to lack of safety data.
For influenza, everyone 6 months or older still needs yearly flu vaccination with an age- and health-appropriate flu vaccine. But for those aged 65 or older, the notes specify the three vaccine versions now preferred: high-dose, recombinant, or adjuvanted versions. However, if these aren’t available, it’s better to get any flu vaccine than to go without.
Under meningococcal vaccines, the notes for MenACWY and MenB are combined. For MenB, trade names Bexsero and Trumenba are specified because the products are not interchangeable. Booster intervals for those still at risk are different for each vaccine type: every 5 years for MenACWY boosters, and every 2-3 years for boosts of MenB.
The recent polio cases in New York have put polio vaccination in the spotlight. ACIP has now reinstated its Polio Vaccine Work Group. The new schedule lists polio vaccines on the cover page. Current recommendations have been added to the notes section. Routine vaccination for adults is not necessary, at least for now. However, those at increased risk for exposure to polio fall in the special-situation category. For those at increased risk who have completed a polio vaccine series, a single lifetime IPV booster can be given. For those at increased risk who have not completed their polio vaccine series, now would be the time to finish the series.
Appendix
The final step in using the new schedule is checking the appendix and its list of vaccine-specific contraindications and precautions.
I hope this review of the new ACIP adult immunization schedule has been helpful. For Medicine Matters, I’m Dr. Sandra Fryhofer.
Dr. Fryhofer is clinical associate professor of medicine, Emory University, Atlanta. She reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hello. I’m Dr Sandra Fryhofer. Welcome to Medicine Matters.
It’s a new year, which means a new ACIP adult immunization schedule – a valuable resource collating ACIP’s most up-to-date vaccination recommendations.
Here are this year’s five most important changes:
- COVID vaccines now front and center
- New emphasis on polio vaccination
- Inclusion of some nonvaccine products (such as monoclonal antibody products)
- Pharmacists group has approved the schedule for the first time
- New shared clinical decision-making option for pneumococcal vaccines
The schedule’s organization remains the same. It still has four sections:
- Table 1: vaccinations by age
- Table 2: vaccinations by medical condition and other indications
- The Notes section (alphabetically ordered by vaccine type)
- Appendix listing of vaccine-specific contraindications and precautions
But what’s unique this year is that some of the abbreviations have historical implications. The first change is no big surprise in light of what we’ve gone through in the past few years. COVID vaccines are listed first on the cover page by brand name for those authorized and by company name for those still under US emergency use authorization. They’re also listed first on the graphics and in the notes.
COVID and mRNA and protein-based vaccines have now been assigned official abbreviations based on vaccine platform and valency.
- 1vCOV-mRNA: Comirnaty/Pfizer-BioNTech and Spikevax Moderna COVID-19 vaccines
- 2vCOV-mRNA: Pfizer-BioNTech and Moderna bivalent COVID-19 vaccines
- 1vCOV-aPS: Novavax COVID-19 vaccine
Also remarkable is the absence of COVID viral vector vaccines on the list. However, the viral vector COVID vaccine (which has been available but is not preferred) does have a CDC website link in the Notes section.
A sad but necessary inclusion was triggered by recent polio cases in New York. Polio was believed to be eradicated, and we thought adults no longer needed to be vaccinated against polio. In the new schedule, the polio vaccine is listed on the cover page but is not included in the tables. Current polio vaccination recommendations are now in the Notes section.
Also of historical significance and something that may set a precedent is the inclusion of nonvaccine products. The value of COVID preexposure prophylaxis with products including monoclonal antibodies (such as Evusheld) for people who are moderately or severely immunocompromised is mentioned in the Notes section.
For the first time ever, the schedule has been approved by the American Pharmacists Association, which validates pharmacists as established partners in vaccine administration.
Color-code key
One aspect of the schedule that has not changed is the color-code key:
- Yellow: Recommended if the patient meets the age requirement
- Purple: Indicated for those with additional risk factors or another indication
- Blue: Recommended based on shared clinical decision-making
- Orange: Precaution
- Red: Contraindicated or not recommended; the vaccine should not be administered. Overlays on the red more precisely clarify whether a vaccine is really contraindicated or just not recommended. An asterisk on red means vaccinate after pregnancy if indicated.
- Gray: No recommendation or not applicable
Vaccinations by age
Table 1 lists recommended vaccinations by age. There is one major change. COVID vaccines are on the first row of the graphic, with the need for both a primary series and boosters emphasized on the overlay. The notes have hyperlinks to the most up-to-date COVID vaccination recommendations.
Pneumococcal vaccination. Pneumococcal vaccination is routinely recommended starting at age 65. Current recommendations for those not previously vaccinated have not changed since last year. But on Table 1, the bottom half of the row for those 65 or older is now blue (and that’s new). This new color blue means shared clinical decision-making and applies to people who were previously considered fully vaccinated with the now extinct combination of PCV13 and PPSV23. These patients now have the option of getting a dose of PCV20 five years after completing their PCV13-PPSV23 combo series. This option is blue because the decision is up to you and your patient.
Check the notes for more pneumococcal vaccination details. For example, for those partially vaccinated using lower valency vaccines, there’s an option of substituting PCV20 for PPSV23 to broaden and increase durability of protection.
The pneumococcal vaccination recommendation options are complicated. A new pneumococcal vaccination app can help.
Hepatitis B. For adults under age 60, the color code for the hepatitis B vaccine is yellow, meaning it’s indicated for all. For older patients, the color code is purple. If a patient who is age 60 or older wants the hepatitis B vaccine, they can have it even in the absence of additional risk indications.
Vaccinations by medical condition or other indications
Other than a few minor word changes on the overlay, the only thing that’s new is the COVID vaccine row.
This table is helpful for matching vaccine recommendations with specific medical conditions, including pregnancy, immunocompromise, HIV (with specifics according to CD4 count), asplenia, complement deficiencies, heart disease, lung disease, alcoholism, chronic liver disease, diabetes, health care personnel, and men who have sex with men.
Use this table to dot the i’s and cross the t’s when it comes to vaccination recommendations. For example, take a look at the pregnancy column. Live virus vaccines, including LAIV, MMR, and varicella, are contraindicated and color-coded red. MMR and varicella also have an asterisk, meaning vaccinate after pregnancy if indicated. HPV vaccines are not live virus vaccines, but the overlay says they are not recommended during pregnancy. The asterisk indicates that you can vaccinate after pregnancy.
Vaccine notes
The notes are in alphabetical order, and their organization (routine, special situations, and shared clinical decision-making when indicated) has not changed. They are concise and succinct, but sometimes they’re not enough. That’s why vaccine-specific links to more complete recommendations are so convenient.
Notes for hepatitis B contain nuances on specific dosing for vaccinating patients on dialysis, as well as a reminder that newer hepatitis C vaccines such as Heplisav and PreHevbrio are not recommended during pregnancy due to lack of safety data.
For influenza, everyone 6 months or older still needs yearly flu vaccination with an age- and health-appropriate flu vaccine. But for those aged 65 or older, the notes specify the three vaccine versions now preferred: high-dose, recombinant, or adjuvanted versions. However, if these aren’t available, it’s better to get any flu vaccine than to go without.
Under meningococcal vaccines, the notes for MenACWY and MenB are combined. For MenB, trade names Bexsero and Trumenba are specified because the products are not interchangeable. Booster intervals for those still at risk are different for each vaccine type: every 5 years for MenACWY boosters, and every 2-3 years for boosts of MenB.
The recent polio cases in New York have put polio vaccination in the spotlight. ACIP has now reinstated its Polio Vaccine Work Group. The new schedule lists polio vaccines on the cover page. Current recommendations have been added to the notes section. Routine vaccination for adults is not necessary, at least for now. However, those at increased risk for exposure to polio fall in the special-situation category. For those at increased risk who have completed a polio vaccine series, a single lifetime IPV booster can be given. For those at increased risk who have not completed their polio vaccine series, now would be the time to finish the series.
Appendix
The final step in using the new schedule is checking the appendix and its list of vaccine-specific contraindications and precautions.
I hope this review of the new ACIP adult immunization schedule has been helpful. For Medicine Matters, I’m Dr. Sandra Fryhofer.
Dr. Fryhofer is clinical associate professor of medicine, Emory University, Atlanta. She reported numerous conflicts of interest.
A version of this article first appeared on Medscape.com.
AAP approves 2023 child and adolescent immunization schedule
The American Academy of Pediatrics said it supports the Recommended Childhood and Adolescent Immunization Schedule: United States, 2023.
In a policy statement published online in the journal Pediatrics, the AAP said the updated recommendations do not include major changes from those released in 2022 by the Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention.
In one small shift, COVID-19 is now addressed in the main text instead of being relegated to the notes section.
“And a new vaccine – Priorix [GlaxoSmithKline] – has been added for MMR [measles, mumps, rubella], so now there are two available,” Sean T. O’Leary, MD, MPH, chair of the AAP’s Committee on Infectious Diseases, told this news organization. “There’s also a second pneumococcal conjugate vaccine listed, PCV15, and this and PCV13 can essentially be used interchangeably.”
Minor updates to the schedule, reflected on the cover page, relate to vaccines for COVID-19, dengue fever, and pneumococcal disease, added Dr. O’Leary, a professor of pediatrics at the University of Colorado Anschutz Medical Campus and Children’s Hospital Colorado, Aurora.
The committee also changed layouts to improve the usability of the schedule. Updated annually, the guidance provides a table on recommended pediatric immunizations from birth to age 18 years, and catch-up recommendations for children aged 4 months to 18 years who start their vaccinations late or are more than 1 month behind the recommended age for vaccine administration.
“We hope this annual update will encourage clinicians to make sure all their patients are up to date on their routine vaccinations,” Dr. O’Leary said. “It’s an opportunity to develop strategies to improve vaccination rates.”
The 2023 schedule follows news from the CDC that kindergarten vaccination rates declined during the 2021-2022 school year. Only 93% of kindergarteners obtained full vaccinations, representing a drop of 1 percentage point from the year before and 2 percentage points from the 2019-2020 school year.
The dip in coverage has been attributed to disruptions caused by the COVID-19 pandemic. AAP advises health care professionals to urge families to make sure their child’s vaccines are current.
Among other additions:
In Table 1
- MMR: Second vaccine added (Priorix, GlaxoSmithKline Biologicals)
- Pneumococcal disease: second conjugate vaccine, PCV15, added (Vaxneuvance, Merck Sharp & Dohme).
- COVID-19: New row added.
- Dengue: Text changed from “Seropositive in endemic areas only” to “Seropositive in endemic dengue areas.”
- Inactivated polio vaccine: “See Notes” added to the column for children aged 18 years.
In Table 2
- PCV: Dose 3 to dose 4 interval revised to align with ACIP’s recommendation for dose 4. This dose is necessary only for children ages 12-59 months regardless of risk, or age 60-71 months with any risk who received three doses before age 12 months.
A parent-friendly vaccine schedule for children and adolescents is available on the CDC’s website.
“Vaccines are essential for the health of our whole society, including children and adolescents,” Dr. O’Leary said in a press release from AAP. “These schedules provide a road map [that] parents and pediatricians can follow to help children get the vaccines they need so their immune systems will be ready to recognize and resist diseases.”
As previously, the 2023 schedule was adjusted to ensure consistency between the formats of the childhood/adolescent and adult immunization guidance. A meeting of stakeholder organizations in October 2022 harmonized the two formats.
A version of this article first appeared on Medscape.com.
The American Academy of Pediatrics said it supports the Recommended Childhood and Adolescent Immunization Schedule: United States, 2023.
In a policy statement published online in the journal Pediatrics, the AAP said the updated recommendations do not include major changes from those released in 2022 by the Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention.
In one small shift, COVID-19 is now addressed in the main text instead of being relegated to the notes section.
“And a new vaccine – Priorix [GlaxoSmithKline] – has been added for MMR [measles, mumps, rubella], so now there are two available,” Sean T. O’Leary, MD, MPH, chair of the AAP’s Committee on Infectious Diseases, told this news organization. “There’s also a second pneumococcal conjugate vaccine listed, PCV15, and this and PCV13 can essentially be used interchangeably.”
Minor updates to the schedule, reflected on the cover page, relate to vaccines for COVID-19, dengue fever, and pneumococcal disease, added Dr. O’Leary, a professor of pediatrics at the University of Colorado Anschutz Medical Campus and Children’s Hospital Colorado, Aurora.
The committee also changed layouts to improve the usability of the schedule. Updated annually, the guidance provides a table on recommended pediatric immunizations from birth to age 18 years, and catch-up recommendations for children aged 4 months to 18 years who start their vaccinations late or are more than 1 month behind the recommended age for vaccine administration.
“We hope this annual update will encourage clinicians to make sure all their patients are up to date on their routine vaccinations,” Dr. O’Leary said. “It’s an opportunity to develop strategies to improve vaccination rates.”
The 2023 schedule follows news from the CDC that kindergarten vaccination rates declined during the 2021-2022 school year. Only 93% of kindergarteners obtained full vaccinations, representing a drop of 1 percentage point from the year before and 2 percentage points from the 2019-2020 school year.
The dip in coverage has been attributed to disruptions caused by the COVID-19 pandemic. AAP advises health care professionals to urge families to make sure their child’s vaccines are current.
Among other additions:
In Table 1
- MMR: Second vaccine added (Priorix, GlaxoSmithKline Biologicals)
- Pneumococcal disease: second conjugate vaccine, PCV15, added (Vaxneuvance, Merck Sharp & Dohme).
- COVID-19: New row added.
- Dengue: Text changed from “Seropositive in endemic areas only” to “Seropositive in endemic dengue areas.”
- Inactivated polio vaccine: “See Notes” added to the column for children aged 18 years.
In Table 2
- PCV: Dose 3 to dose 4 interval revised to align with ACIP’s recommendation for dose 4. This dose is necessary only for children ages 12-59 months regardless of risk, or age 60-71 months with any risk who received three doses before age 12 months.
A parent-friendly vaccine schedule for children and adolescents is available on the CDC’s website.
“Vaccines are essential for the health of our whole society, including children and adolescents,” Dr. O’Leary said in a press release from AAP. “These schedules provide a road map [that] parents and pediatricians can follow to help children get the vaccines they need so their immune systems will be ready to recognize and resist diseases.”
As previously, the 2023 schedule was adjusted to ensure consistency between the formats of the childhood/adolescent and adult immunization guidance. A meeting of stakeholder organizations in October 2022 harmonized the two formats.
A version of this article first appeared on Medscape.com.
The American Academy of Pediatrics said it supports the Recommended Childhood and Adolescent Immunization Schedule: United States, 2023.
In a policy statement published online in the journal Pediatrics, the AAP said the updated recommendations do not include major changes from those released in 2022 by the Advisory Committee on Immunization Practices of the Centers for Disease Control and Prevention.
In one small shift, COVID-19 is now addressed in the main text instead of being relegated to the notes section.
“And a new vaccine – Priorix [GlaxoSmithKline] – has been added for MMR [measles, mumps, rubella], so now there are two available,” Sean T. O’Leary, MD, MPH, chair of the AAP’s Committee on Infectious Diseases, told this news organization. “There’s also a second pneumococcal conjugate vaccine listed, PCV15, and this and PCV13 can essentially be used interchangeably.”
Minor updates to the schedule, reflected on the cover page, relate to vaccines for COVID-19, dengue fever, and pneumococcal disease, added Dr. O’Leary, a professor of pediatrics at the University of Colorado Anschutz Medical Campus and Children’s Hospital Colorado, Aurora.
The committee also changed layouts to improve the usability of the schedule. Updated annually, the guidance provides a table on recommended pediatric immunizations from birth to age 18 years, and catch-up recommendations for children aged 4 months to 18 years who start their vaccinations late or are more than 1 month behind the recommended age for vaccine administration.
“We hope this annual update will encourage clinicians to make sure all their patients are up to date on their routine vaccinations,” Dr. O’Leary said. “It’s an opportunity to develop strategies to improve vaccination rates.”
The 2023 schedule follows news from the CDC that kindergarten vaccination rates declined during the 2021-2022 school year. Only 93% of kindergarteners obtained full vaccinations, representing a drop of 1 percentage point from the year before and 2 percentage points from the 2019-2020 school year.
The dip in coverage has been attributed to disruptions caused by the COVID-19 pandemic. AAP advises health care professionals to urge families to make sure their child’s vaccines are current.
Among other additions:
In Table 1
- MMR: Second vaccine added (Priorix, GlaxoSmithKline Biologicals)
- Pneumococcal disease: second conjugate vaccine, PCV15, added (Vaxneuvance, Merck Sharp & Dohme).
- COVID-19: New row added.
- Dengue: Text changed from “Seropositive in endemic areas only” to “Seropositive in endemic dengue areas.”
- Inactivated polio vaccine: “See Notes” added to the column for children aged 18 years.
In Table 2
- PCV: Dose 3 to dose 4 interval revised to align with ACIP’s recommendation for dose 4. This dose is necessary only for children ages 12-59 months regardless of risk, or age 60-71 months with any risk who received three doses before age 12 months.
A parent-friendly vaccine schedule for children and adolescents is available on the CDC’s website.
“Vaccines are essential for the health of our whole society, including children and adolescents,” Dr. O’Leary said in a press release from AAP. “These schedules provide a road map [that] parents and pediatricians can follow to help children get the vaccines they need so their immune systems will be ready to recognize and resist diseases.”
As previously, the 2023 schedule was adjusted to ensure consistency between the formats of the childhood/adolescent and adult immunization guidance. A meeting of stakeholder organizations in October 2022 harmonized the two formats.
A version of this article first appeared on Medscape.com.
FROM PEDIATRICS
A new (old) drug joins the COVID fray, and guess what? It works
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
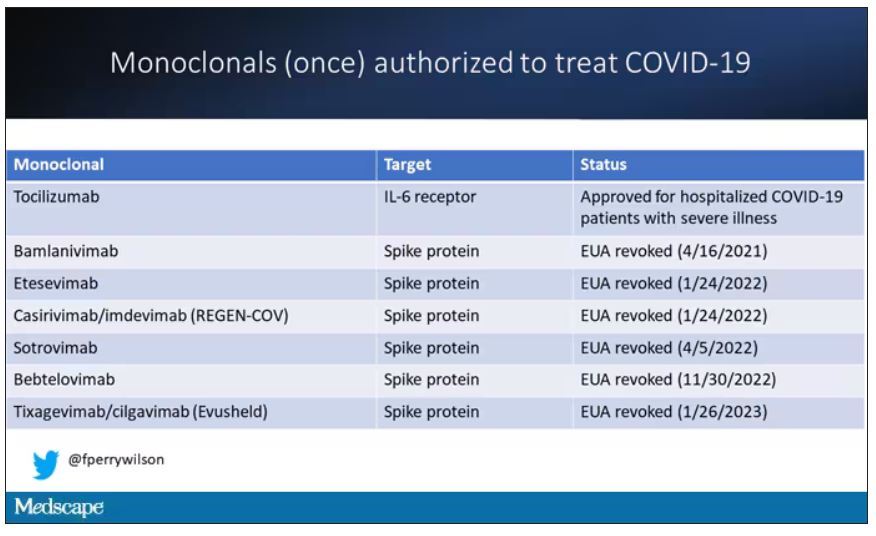
At this point, with the monoclonals found to be essentially useless, we are left with remdesivir with its modest efficacy and Paxlovid, which, for some reason, people don’t seem to be taking.
Part of the reason the monoclonals have failed lately is because of their specificity; they are homogeneous antibodies targeted toward a very specific epitope that may change from variant to variant. We need a broader therapeutic, one that has activity across all variants — maybe even one that has activity against all viruses? We’ve got one. Interferon.
The first mention of interferon as a potential COVID therapy was at the very start of the pandemic, so I’m sort of surprised that the first large, randomized trial is only being reported now in the New England Journal of Medicine.
Before we dig into the results, let’s talk mechanism. This is a trial of interferon-lambda, also known as interleukin-29.
The lambda interferons were only discovered in 2003. They differ from the more familiar interferons only in their cellular receptors; the downstream effects seem quite similar. As opposed to the cellular receptors for interferon alfa, which are widely expressed, the receptors for lambda are restricted to epithelial tissues. This makes it a good choice as a COVID treatment, since the virus also preferentially targets those epithelial cells.
In this study, 1,951 participants from Brazil and Canada, but mostly Brazil, with new COVID infections who were not yet hospitalized were randomized to receive 180 mcg of interferon lambda or placebo.
This was a relatively current COVID trial, as you can see from the participant characteristics. The majority had been vaccinated, and nearly half of the infections were during the Omicron phase of the pandemic.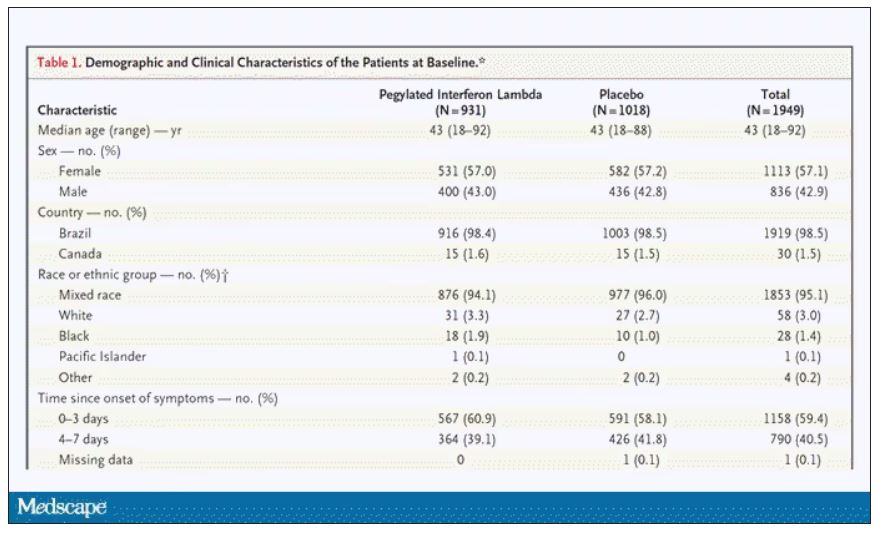
If you just want to cut to the chase, interferon worked.
The primary outcome – hospitalization or a prolonged emergency room visit for COVID – was 50% lower in the interferon group.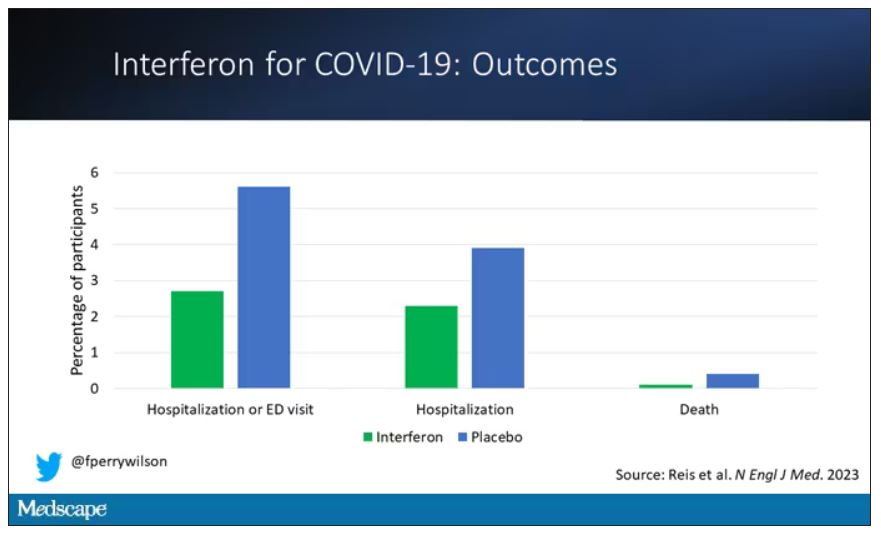
Key secondary outcomes, including death from COVID, were lower in the interferon group as well. These effects persisted across most of the subgroups I was looking out for.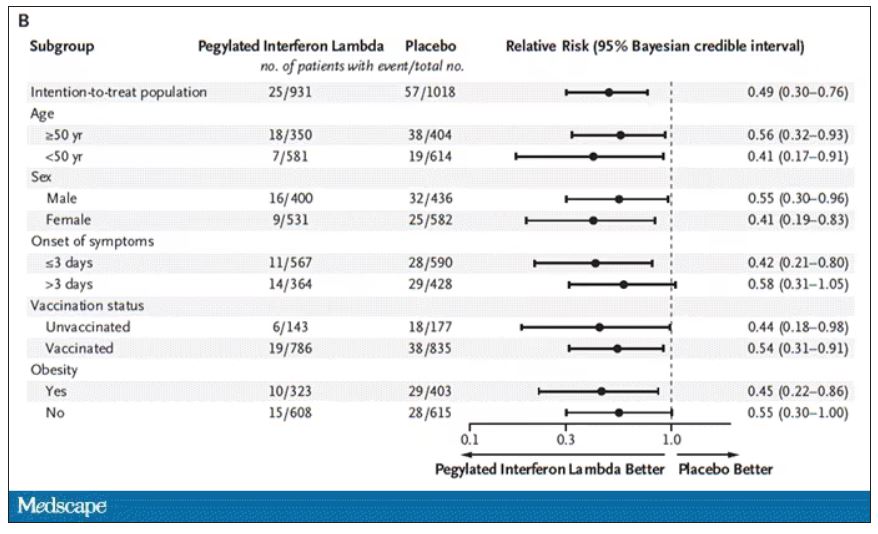
Interferon seemed to help those who were already vaccinated and those who were unvaccinated. There’s a hint that it works better within the first few days of symptoms, which isn’t surprising; we’ve seen this for many of the therapeutics, including Paxlovid. Time is of the essence. Encouragingly, the effect was a bit more pronounced among those infected with Omicron.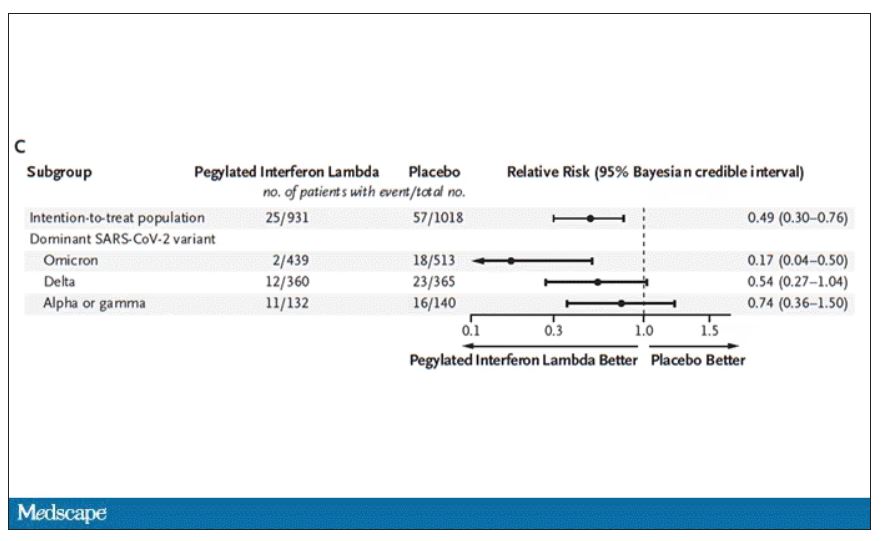
Of course, if you have any experience with interferon, you know that the side effects can be pretty rough. In the bad old days when we treated hepatitis C infection with interferon, patients would get their injections on Friday in anticipation of being essentially out of commission with flu-like symptoms through the weekend. But we don’t see much evidence of adverse events in this trial, maybe due to the greater specificity of interferon lambda.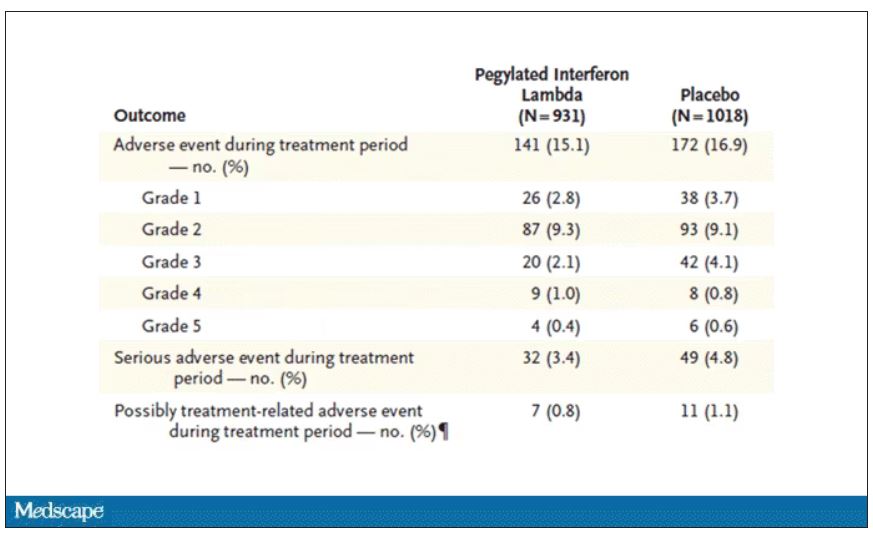
Putting it all together, the state of play for interferons in COVID may be changing. To date, the FDA has not recommended the use of interferon alfa or -beta for COVID-19, citing some data that they are ineffective or even harmful in hospitalized patients with COVID. Interferon lambda is not FDA approved and thus not even available in the United States. But the reason it has not been approved is that there has not been a large, well-conducted interferon lambda trial. Now there is. Will this study be enough to prompt an emergency use authorization? The elephant in the room, of course, is Paxlovid, which at this point has a longer safety track record and, importantly, is oral. I’d love to see a head-to-head trial. Short of that, I tend to be in favor of having more options on the table.
Dr. Perry Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.

At this point, with the monoclonals found to be essentially useless, we are left with remdesivir with its modest efficacy and Paxlovid, which, for some reason, people don’t seem to be taking.
Part of the reason the monoclonals have failed lately is because of their specificity; they are homogeneous antibodies targeted toward a very specific epitope that may change from variant to variant. We need a broader therapeutic, one that has activity across all variants — maybe even one that has activity against all viruses? We’ve got one. Interferon.
The first mention of interferon as a potential COVID therapy was at the very start of the pandemic, so I’m sort of surprised that the first large, randomized trial is only being reported now in the New England Journal of Medicine.
Before we dig into the results, let’s talk mechanism. This is a trial of interferon-lambda, also known as interleukin-29.
The lambda interferons were only discovered in 2003. They differ from the more familiar interferons only in their cellular receptors; the downstream effects seem quite similar. As opposed to the cellular receptors for interferon alfa, which are widely expressed, the receptors for lambda are restricted to epithelial tissues. This makes it a good choice as a COVID treatment, since the virus also preferentially targets those epithelial cells.
In this study, 1,951 participants from Brazil and Canada, but mostly Brazil, with new COVID infections who were not yet hospitalized were randomized to receive 180 mcg of interferon lambda or placebo.
This was a relatively current COVID trial, as you can see from the participant characteristics. The majority had been vaccinated, and nearly half of the infections were during the Omicron phase of the pandemic.
If you just want to cut to the chase, interferon worked.
The primary outcome – hospitalization or a prolonged emergency room visit for COVID – was 50% lower in the interferon group.
Key secondary outcomes, including death from COVID, were lower in the interferon group as well. These effects persisted across most of the subgroups I was looking out for.
Interferon seemed to help those who were already vaccinated and those who were unvaccinated. There’s a hint that it works better within the first few days of symptoms, which isn’t surprising; we’ve seen this for many of the therapeutics, including Paxlovid. Time is of the essence. Encouragingly, the effect was a bit more pronounced among those infected with Omicron.
Of course, if you have any experience with interferon, you know that the side effects can be pretty rough. In the bad old days when we treated hepatitis C infection with interferon, patients would get their injections on Friday in anticipation of being essentially out of commission with flu-like symptoms through the weekend. But we don’t see much evidence of adverse events in this trial, maybe due to the greater specificity of interferon lambda.
Putting it all together, the state of play for interferons in COVID may be changing. To date, the FDA has not recommended the use of interferon alfa or -beta for COVID-19, citing some data that they are ineffective or even harmful in hospitalized patients with COVID. Interferon lambda is not FDA approved and thus not even available in the United States. But the reason it has not been approved is that there has not been a large, well-conducted interferon lambda trial. Now there is. Will this study be enough to prompt an emergency use authorization? The elephant in the room, of course, is Paxlovid, which at this point has a longer safety track record and, importantly, is oral. I’d love to see a head-to-head trial. Short of that, I tend to be in favor of having more options on the table.
Dr. Perry Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.

At this point, with the monoclonals found to be essentially useless, we are left with remdesivir with its modest efficacy and Paxlovid, which, for some reason, people don’t seem to be taking.
Part of the reason the monoclonals have failed lately is because of their specificity; they are homogeneous antibodies targeted toward a very specific epitope that may change from variant to variant. We need a broader therapeutic, one that has activity across all variants — maybe even one that has activity against all viruses? We’ve got one. Interferon.
The first mention of interferon as a potential COVID therapy was at the very start of the pandemic, so I’m sort of surprised that the first large, randomized trial is only being reported now in the New England Journal of Medicine.
Before we dig into the results, let’s talk mechanism. This is a trial of interferon-lambda, also known as interleukin-29.
The lambda interferons were only discovered in 2003. They differ from the more familiar interferons only in their cellular receptors; the downstream effects seem quite similar. As opposed to the cellular receptors for interferon alfa, which are widely expressed, the receptors for lambda are restricted to epithelial tissues. This makes it a good choice as a COVID treatment, since the virus also preferentially targets those epithelial cells.
In this study, 1,951 participants from Brazil and Canada, but mostly Brazil, with new COVID infections who were not yet hospitalized were randomized to receive 180 mcg of interferon lambda or placebo.
This was a relatively current COVID trial, as you can see from the participant characteristics. The majority had been vaccinated, and nearly half of the infections were during the Omicron phase of the pandemic.
If you just want to cut to the chase, interferon worked.
The primary outcome – hospitalization or a prolonged emergency room visit for COVID – was 50% lower in the interferon group.
Key secondary outcomes, including death from COVID, were lower in the interferon group as well. These effects persisted across most of the subgroups I was looking out for.
Interferon seemed to help those who were already vaccinated and those who were unvaccinated. There’s a hint that it works better within the first few days of symptoms, which isn’t surprising; we’ve seen this for many of the therapeutics, including Paxlovid. Time is of the essence. Encouragingly, the effect was a bit more pronounced among those infected with Omicron.
Of course, if you have any experience with interferon, you know that the side effects can be pretty rough. In the bad old days when we treated hepatitis C infection with interferon, patients would get their injections on Friday in anticipation of being essentially out of commission with flu-like symptoms through the weekend. But we don’t see much evidence of adverse events in this trial, maybe due to the greater specificity of interferon lambda.
Putting it all together, the state of play for interferons in COVID may be changing. To date, the FDA has not recommended the use of interferon alfa or -beta for COVID-19, citing some data that they are ineffective or even harmful in hospitalized patients with COVID. Interferon lambda is not FDA approved and thus not even available in the United States. But the reason it has not been approved is that there has not been a large, well-conducted interferon lambda trial. Now there is. Will this study be enough to prompt an emergency use authorization? The elephant in the room, of course, is Paxlovid, which at this point has a longer safety track record and, importantly, is oral. I’d love to see a head-to-head trial. Short of that, I tend to be in favor of having more options on the table.
Dr. Perry Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Maternal COVID-19 vaccine curbs infant infection
a new study shows.
Previous research has confirmed that COVID-19 neutralizing antibodies following maternal vaccination or maternal COVID-19 infection are present in umbilical cord blood, breast milk, and infant serum specimens, wrote Sarah C.J. Jorgensen, PharmD, MPH, of the University of Toronto, and colleagues in their article published in The BMJ.
In the study, the researchers identified maternal and newborn pairs using administrative databases from Canada. The study population included 8,809 infants aged younger than 6 months who were born between May 7, 2021, and March 31, 2022, and who underwent testing for COVID-19 between May 7, 2021, and September 5, 2022.
Maternal vaccination with the primary COVID-19 mRNA monovalent vaccine series was defined as two vaccine doses administered up to 14 days before delivery, with at least one of the doses after the conception date.
Maternal vaccination with the primary series plus one booster was defined as three doses administered up to 14 days before delivery, with at least one of these doses after the conception date.
The primary outcome was the presence of delta or omicron COVID-19 infection or hospital admission of the infants.
The study population included 99 COVID-19 cases with the delta variant (with 4,365 controls) and 1,501 cases with the omicron variant (with 4,847 controls).
Overall, the vaccine effectiveness of maternal doses was 95% against delta infection and 45% against omicron.
The effectiveness against hospital admission in cases of delta and omicron variants were 97% and 53%, respectively.
The effectiveness of three doses was 73% against omicron infant infection and 80% against omicron-related infant hospitalization. Data were not available for the effectiveness of three doses against the delta variant.
The effectiveness of two doses of vaccine against infant omicron infection was highest when mothers received the second dose during the third trimester of pregnancy, compared with during the first trimester or second trimester (53% vs. 47% and 53% vs. 37%, respectively).
Vaccine effectiveness with two doses against infant infection from omicron was highest in the first 8 weeks of life (57%), then decreased to 40% among infants after 16 weeks of age.
Although the study was not designed to assess the mechanism of action of the impact of maternal vaccination on infants, the current study results were consistent with other recent studies showing a reduction in infections and hospitalizations among infants whose mothers received COVID-19 vaccines during pregnancy, the researchers wrote in their discussion.
The findings were limited by several factors including the potential unmeasured confounders not available in databases, such as whether infants were breastfed, the researchers noted. Other limitations included a lack of data on home test results and the inability to assess the waning impact of the vaccine effectiveness against the delta variant because of the small number of delta cases, they said. However, the results suggest that the mRNA COVID-19 vaccine during pregnancy was moderately to highly effective for protection against omicron and delta infection and infection-related hospitalization – especially during the first 8 weeks of life.
Effectiveness is encouraging, but updates are needed
The effectiveness of maternal vaccination to prevent COVID-19 infection and related hospitalizations in infants is promising, especially since those younger than 6 months have no other source of vaccine protection against COVID-19 infection, wrote Dana Danino, MD, of Soroka University Medical Center, Israel, and Ilan Youngster, MD, of Shamir Medical Center, Israel, in an accompanying editorial also published in The BMJ.
They also noted that maternal vaccination during pregnancy is an established method of protecting infants from infections such as influenza and pertussis.
Data from previous studies show that most infants whose mothers were vaccinated against COVID-19 during pregnancy retained maternal antibodies at 6 months, “but evidence for protection against neonatal COVID-19 infection has been deficient,” they said.
The current study findings support the value of vaccination during pregnancy, and the findings were strengthened by the large study population, the editorialists wrote. However, whether the same effectiveness holds for other COVID-19 strains such as BQ.1, BQ.1.1, BF.7, XBB, and XBB.1 remains unknown, they said.
Other areas in need of exploration include the optimal timing of vaccination during pregnancy, the protective effects of a bivalent mRNA vaccine (vs. the primary monovalent vaccine in the current study), and the potential benefits of additional boosters, they added.
“Although Jorgenson and colleagues’ study reinforces the value of maternal vaccination against COVID-19 during pregnancy, more studies are needed to better inform vaccination recommendations in an evolving landscape of new SARS-CoV-2 strains and novel vaccines,” the editorialists concluded.
The study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-term Care; the study also received funding from the Canadian Immunization Research Network and the Public Health Agency of Canada. Dr. Jorgensen and the editorialists had no financial conflicts to disclose.
*This article was updated on 3/2/2023.
a new study shows.
Previous research has confirmed that COVID-19 neutralizing antibodies following maternal vaccination or maternal COVID-19 infection are present in umbilical cord blood, breast milk, and infant serum specimens, wrote Sarah C.J. Jorgensen, PharmD, MPH, of the University of Toronto, and colleagues in their article published in The BMJ.
In the study, the researchers identified maternal and newborn pairs using administrative databases from Canada. The study population included 8,809 infants aged younger than 6 months who were born between May 7, 2021, and March 31, 2022, and who underwent testing for COVID-19 between May 7, 2021, and September 5, 2022.
Maternal vaccination with the primary COVID-19 mRNA monovalent vaccine series was defined as two vaccine doses administered up to 14 days before delivery, with at least one of the doses after the conception date.
Maternal vaccination with the primary series plus one booster was defined as three doses administered up to 14 days before delivery, with at least one of these doses after the conception date.
The primary outcome was the presence of delta or omicron COVID-19 infection or hospital admission of the infants.
The study population included 99 COVID-19 cases with the delta variant (with 4,365 controls) and 1,501 cases with the omicron variant (with 4,847 controls).
Overall, the vaccine effectiveness of maternal doses was 95% against delta infection and 45% against omicron.
The effectiveness against hospital admission in cases of delta and omicron variants were 97% and 53%, respectively.
The effectiveness of three doses was 73% against omicron infant infection and 80% against omicron-related infant hospitalization. Data were not available for the effectiveness of three doses against the delta variant.
The effectiveness of two doses of vaccine against infant omicron infection was highest when mothers received the second dose during the third trimester of pregnancy, compared with during the first trimester or second trimester (53% vs. 47% and 53% vs. 37%, respectively).
Vaccine effectiveness with two doses against infant infection from omicron was highest in the first 8 weeks of life (57%), then decreased to 40% among infants after 16 weeks of age.
Although the study was not designed to assess the mechanism of action of the impact of maternal vaccination on infants, the current study results were consistent with other recent studies showing a reduction in infections and hospitalizations among infants whose mothers received COVID-19 vaccines during pregnancy, the researchers wrote in their discussion.
The findings were limited by several factors including the potential unmeasured confounders not available in databases, such as whether infants were breastfed, the researchers noted. Other limitations included a lack of data on home test results and the inability to assess the waning impact of the vaccine effectiveness against the delta variant because of the small number of delta cases, they said. However, the results suggest that the mRNA COVID-19 vaccine during pregnancy was moderately to highly effective for protection against omicron and delta infection and infection-related hospitalization – especially during the first 8 weeks of life.
Effectiveness is encouraging, but updates are needed
The effectiveness of maternal vaccination to prevent COVID-19 infection and related hospitalizations in infants is promising, especially since those younger than 6 months have no other source of vaccine protection against COVID-19 infection, wrote Dana Danino, MD, of Soroka University Medical Center, Israel, and Ilan Youngster, MD, of Shamir Medical Center, Israel, in an accompanying editorial also published in The BMJ.
They also noted that maternal vaccination during pregnancy is an established method of protecting infants from infections such as influenza and pertussis.
Data from previous studies show that most infants whose mothers were vaccinated against COVID-19 during pregnancy retained maternal antibodies at 6 months, “but evidence for protection against neonatal COVID-19 infection has been deficient,” they said.
The current study findings support the value of vaccination during pregnancy, and the findings were strengthened by the large study population, the editorialists wrote. However, whether the same effectiveness holds for other COVID-19 strains such as BQ.1, BQ.1.1, BF.7, XBB, and XBB.1 remains unknown, they said.
Other areas in need of exploration include the optimal timing of vaccination during pregnancy, the protective effects of a bivalent mRNA vaccine (vs. the primary monovalent vaccine in the current study), and the potential benefits of additional boosters, they added.
“Although Jorgenson and colleagues’ study reinforces the value of maternal vaccination against COVID-19 during pregnancy, more studies are needed to better inform vaccination recommendations in an evolving landscape of new SARS-CoV-2 strains and novel vaccines,” the editorialists concluded.
The study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-term Care; the study also received funding from the Canadian Immunization Research Network and the Public Health Agency of Canada. Dr. Jorgensen and the editorialists had no financial conflicts to disclose.
*This article was updated on 3/2/2023.
a new study shows.
Previous research has confirmed that COVID-19 neutralizing antibodies following maternal vaccination or maternal COVID-19 infection are present in umbilical cord blood, breast milk, and infant serum specimens, wrote Sarah C.J. Jorgensen, PharmD, MPH, of the University of Toronto, and colleagues in their article published in The BMJ.
In the study, the researchers identified maternal and newborn pairs using administrative databases from Canada. The study population included 8,809 infants aged younger than 6 months who were born between May 7, 2021, and March 31, 2022, and who underwent testing for COVID-19 between May 7, 2021, and September 5, 2022.
Maternal vaccination with the primary COVID-19 mRNA monovalent vaccine series was defined as two vaccine doses administered up to 14 days before delivery, with at least one of the doses after the conception date.
Maternal vaccination with the primary series plus one booster was defined as three doses administered up to 14 days before delivery, with at least one of these doses after the conception date.
The primary outcome was the presence of delta or omicron COVID-19 infection or hospital admission of the infants.
The study population included 99 COVID-19 cases with the delta variant (with 4,365 controls) and 1,501 cases with the omicron variant (with 4,847 controls).
Overall, the vaccine effectiveness of maternal doses was 95% against delta infection and 45% against omicron.
The effectiveness against hospital admission in cases of delta and omicron variants were 97% and 53%, respectively.
The effectiveness of three doses was 73% against omicron infant infection and 80% against omicron-related infant hospitalization. Data were not available for the effectiveness of three doses against the delta variant.
The effectiveness of two doses of vaccine against infant omicron infection was highest when mothers received the second dose during the third trimester of pregnancy, compared with during the first trimester or second trimester (53% vs. 47% and 53% vs. 37%, respectively).
Vaccine effectiveness with two doses against infant infection from omicron was highest in the first 8 weeks of life (57%), then decreased to 40% among infants after 16 weeks of age.
Although the study was not designed to assess the mechanism of action of the impact of maternal vaccination on infants, the current study results were consistent with other recent studies showing a reduction in infections and hospitalizations among infants whose mothers received COVID-19 vaccines during pregnancy, the researchers wrote in their discussion.
The findings were limited by several factors including the potential unmeasured confounders not available in databases, such as whether infants were breastfed, the researchers noted. Other limitations included a lack of data on home test results and the inability to assess the waning impact of the vaccine effectiveness against the delta variant because of the small number of delta cases, they said. However, the results suggest that the mRNA COVID-19 vaccine during pregnancy was moderately to highly effective for protection against omicron and delta infection and infection-related hospitalization – especially during the first 8 weeks of life.
Effectiveness is encouraging, but updates are needed
The effectiveness of maternal vaccination to prevent COVID-19 infection and related hospitalizations in infants is promising, especially since those younger than 6 months have no other source of vaccine protection against COVID-19 infection, wrote Dana Danino, MD, of Soroka University Medical Center, Israel, and Ilan Youngster, MD, of Shamir Medical Center, Israel, in an accompanying editorial also published in The BMJ.
They also noted that maternal vaccination during pregnancy is an established method of protecting infants from infections such as influenza and pertussis.
Data from previous studies show that most infants whose mothers were vaccinated against COVID-19 during pregnancy retained maternal antibodies at 6 months, “but evidence for protection against neonatal COVID-19 infection has been deficient,” they said.
The current study findings support the value of vaccination during pregnancy, and the findings were strengthened by the large study population, the editorialists wrote. However, whether the same effectiveness holds for other COVID-19 strains such as BQ.1, BQ.1.1, BF.7, XBB, and XBB.1 remains unknown, they said.
Other areas in need of exploration include the optimal timing of vaccination during pregnancy, the protective effects of a bivalent mRNA vaccine (vs. the primary monovalent vaccine in the current study), and the potential benefits of additional boosters, they added.
“Although Jorgenson and colleagues’ study reinforces the value of maternal vaccination against COVID-19 during pregnancy, more studies are needed to better inform vaccination recommendations in an evolving landscape of new SARS-CoV-2 strains and novel vaccines,” the editorialists concluded.
The study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-term Care; the study also received funding from the Canadian Immunization Research Network and the Public Health Agency of Canada. Dr. Jorgensen and the editorialists had no financial conflicts to disclose.
*This article was updated on 3/2/2023.
FROM THE BMJ
Acute cardiac events common during COVID hospitalization
particularly among those with underlying heart disease, and are associated with more severe disease outcomes, a new study suggests.
“We expected to see acute cardiac events occurring among adults hospitalized with COVID-19 but were surprised by how frequently they occurred,” Rebecca C. Woodruff, PhD, MPH, of the U.S. Centers for Disease Control and Prevention, Atlanta, told this news organization.
Overall, she said, “about 1 in 10 adults experienced an acute cardiac event – including heart attacks and acute heart failure – while hospitalized with COVID-19, and this included people with no preexisting heart disease.”
However, she added, “about a quarter of those with underlying heart disease had an acute cardiac event. These patients tended to experience more severe disease outcomes relative to patients hospitalized with COVID-19 who did not experience an acute cardiac event.”
The findings might be relevant to hospitalizations for other viral diseases, “though we can’t say for sure,” she noted. “This study was modeled off a previous study conducted before the COVID-19 pandemic among adults hospitalized with influenza. About 11.7% of [those] adults experienced an acute cardiac event, which was a similar percentage as what we found among patients hospitalized with COVID-19.”
The study was published online in the Journal of the American College of Cardiology.
Underlying cardiac disease key
Dr. Woodruff and colleagues analyzed medical records on a probability sample of 8,460 adults hospitalized with SARS-CoV-2 infection identified from 99 U.S. counties in 14 U.S. states (about 10% of the United States population) from January to November 2021.
Among participants, 11.4% had an acute cardiac event during their hospitalization. The median age was 69 years; 56.5% were men; 48.7%, non-Hispanic White; 33.6%, non-Hispanic Black; 7.4%, Hispanic; and 7.1%, non-Hispanic Asian or Pacific Islander.
As indicated, the prevalence was higher among those with underlying cardiac disease (23.4%), compared with those without (6.2%).
Acute ischemic heart disease (5.5%) and acute heart failure (5.4%) were the most prevalent events; 0.3% of participants had acute myocarditis or pericarditis.
Risk factors varied, depending on underlying cardiac disease status. Those who experienced one or more acute cardiac events had a greater risk for intensive care unit admission (adjusted risk ratio,1.9) and in-hospital death (aRR, 1.7) versus those who did not.
In multivariable analyses, the risk of experiencing acute heart failure was significantly greater among men (aRR, 1.5) and among those with a history of congestive heart failure (aRR, 13.5), atrial fibrillation (aRR, 1.6) or hypertension (aRR,1.3).
Among patients who experienced one or more acute cardiac events, 39.2% required an intensive care unit stay for a median of 5 days. Approximately 22.4% required invasive mechanical ventilation or extracorporeal membrane oxygenation, and 21.1% died while hospitalized.
“Persons at greater risk for experiencing acute cardiac events during COVID-19–associated hospitalizations might benefit from more intensive clinical evaluation and monitoring during hospitalization,” the authors conclude.
The team currently is taking a closer look at acute myocarditis among patients hospitalized with COVID-19, Dr. Woodruff said. Preliminary results were presented at the 2022 annual scientific sessions of the American Heart Association and a paper is forthcoming.
Contemporary data needed
James A. de Lemos, MD, co-chair of the American Heart Association’s COVID-19 CVD Registry Steering Committee and professor of medicine at the University of Texas Southwestern Medical Center, Dallas, said the findings mirror his team’s clinical experience in 2020 and 2021 and echo what was seen in the AHA COVID registry: that is, a 0.3% rate of myocarditis.
“The major caveat is that [the findings] may not be generalizable to contemporary COVID infection, both due to changing viral variants and higher levels of immunity in the population,” he said.
“Rates of COVID hospitalization are markedly lower with the current dominant variants, and we would expect the cardiac risk to be lower as well. I would like to see more contemporary data with current variants, particularly focused on higher risk patients with cardiovascular disease,” Dr. de Lemos added.
In a related editorial, George A. Mensa, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Md., and colleagues suggest that the broader impact of the COVID-19 pandemic on human health remains “incompletely examined.”
“The impact of COVID-19 on cardiovascular mortality, in particular, appears to have varied widely, with no large increases seen in a number of the most developed countries but marked increases in hypertensive heart disease mortality seen in the United States in 2021,” they conclude. “The potential contribution of COVID-19 to these deaths, either directly or indirectly, remains to be determined.”
No commercial funding or relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
particularly among those with underlying heart disease, and are associated with more severe disease outcomes, a new study suggests.
“We expected to see acute cardiac events occurring among adults hospitalized with COVID-19 but were surprised by how frequently they occurred,” Rebecca C. Woodruff, PhD, MPH, of the U.S. Centers for Disease Control and Prevention, Atlanta, told this news organization.
Overall, she said, “about 1 in 10 adults experienced an acute cardiac event – including heart attacks and acute heart failure – while hospitalized with COVID-19, and this included people with no preexisting heart disease.”
However, she added, “about a quarter of those with underlying heart disease had an acute cardiac event. These patients tended to experience more severe disease outcomes relative to patients hospitalized with COVID-19 who did not experience an acute cardiac event.”
The findings might be relevant to hospitalizations for other viral diseases, “though we can’t say for sure,” she noted. “This study was modeled off a previous study conducted before the COVID-19 pandemic among adults hospitalized with influenza. About 11.7% of [those] adults experienced an acute cardiac event, which was a similar percentage as what we found among patients hospitalized with COVID-19.”
The study was published online in the Journal of the American College of Cardiology.
Underlying cardiac disease key
Dr. Woodruff and colleagues analyzed medical records on a probability sample of 8,460 adults hospitalized with SARS-CoV-2 infection identified from 99 U.S. counties in 14 U.S. states (about 10% of the United States population) from January to November 2021.
Among participants, 11.4% had an acute cardiac event during their hospitalization. The median age was 69 years; 56.5% were men; 48.7%, non-Hispanic White; 33.6%, non-Hispanic Black; 7.4%, Hispanic; and 7.1%, non-Hispanic Asian or Pacific Islander.
As indicated, the prevalence was higher among those with underlying cardiac disease (23.4%), compared with those without (6.2%).
Acute ischemic heart disease (5.5%) and acute heart failure (5.4%) were the most prevalent events; 0.3% of participants had acute myocarditis or pericarditis.
Risk factors varied, depending on underlying cardiac disease status. Those who experienced one or more acute cardiac events had a greater risk for intensive care unit admission (adjusted risk ratio,1.9) and in-hospital death (aRR, 1.7) versus those who did not.
In multivariable analyses, the risk of experiencing acute heart failure was significantly greater among men (aRR, 1.5) and among those with a history of congestive heart failure (aRR, 13.5), atrial fibrillation (aRR, 1.6) or hypertension (aRR,1.3).
Among patients who experienced one or more acute cardiac events, 39.2% required an intensive care unit stay for a median of 5 days. Approximately 22.4% required invasive mechanical ventilation or extracorporeal membrane oxygenation, and 21.1% died while hospitalized.
“Persons at greater risk for experiencing acute cardiac events during COVID-19–associated hospitalizations might benefit from more intensive clinical evaluation and monitoring during hospitalization,” the authors conclude.
The team currently is taking a closer look at acute myocarditis among patients hospitalized with COVID-19, Dr. Woodruff said. Preliminary results were presented at the 2022 annual scientific sessions of the American Heart Association and a paper is forthcoming.
Contemporary data needed
James A. de Lemos, MD, co-chair of the American Heart Association’s COVID-19 CVD Registry Steering Committee and professor of medicine at the University of Texas Southwestern Medical Center, Dallas, said the findings mirror his team’s clinical experience in 2020 and 2021 and echo what was seen in the AHA COVID registry: that is, a 0.3% rate of myocarditis.
“The major caveat is that [the findings] may not be generalizable to contemporary COVID infection, both due to changing viral variants and higher levels of immunity in the population,” he said.
“Rates of COVID hospitalization are markedly lower with the current dominant variants, and we would expect the cardiac risk to be lower as well. I would like to see more contemporary data with current variants, particularly focused on higher risk patients with cardiovascular disease,” Dr. de Lemos added.
In a related editorial, George A. Mensa, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Md., and colleagues suggest that the broader impact of the COVID-19 pandemic on human health remains “incompletely examined.”
“The impact of COVID-19 on cardiovascular mortality, in particular, appears to have varied widely, with no large increases seen in a number of the most developed countries but marked increases in hypertensive heart disease mortality seen in the United States in 2021,” they conclude. “The potential contribution of COVID-19 to these deaths, either directly or indirectly, remains to be determined.”
No commercial funding or relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
particularly among those with underlying heart disease, and are associated with more severe disease outcomes, a new study suggests.
“We expected to see acute cardiac events occurring among adults hospitalized with COVID-19 but were surprised by how frequently they occurred,” Rebecca C. Woodruff, PhD, MPH, of the U.S. Centers for Disease Control and Prevention, Atlanta, told this news organization.
Overall, she said, “about 1 in 10 adults experienced an acute cardiac event – including heart attacks and acute heart failure – while hospitalized with COVID-19, and this included people with no preexisting heart disease.”
However, she added, “about a quarter of those with underlying heart disease had an acute cardiac event. These patients tended to experience more severe disease outcomes relative to patients hospitalized with COVID-19 who did not experience an acute cardiac event.”
The findings might be relevant to hospitalizations for other viral diseases, “though we can’t say for sure,” she noted. “This study was modeled off a previous study conducted before the COVID-19 pandemic among adults hospitalized with influenza. About 11.7% of [those] adults experienced an acute cardiac event, which was a similar percentage as what we found among patients hospitalized with COVID-19.”
The study was published online in the Journal of the American College of Cardiology.
Underlying cardiac disease key
Dr. Woodruff and colleagues analyzed medical records on a probability sample of 8,460 adults hospitalized with SARS-CoV-2 infection identified from 99 U.S. counties in 14 U.S. states (about 10% of the United States population) from January to November 2021.
Among participants, 11.4% had an acute cardiac event during their hospitalization. The median age was 69 years; 56.5% were men; 48.7%, non-Hispanic White; 33.6%, non-Hispanic Black; 7.4%, Hispanic; and 7.1%, non-Hispanic Asian or Pacific Islander.
As indicated, the prevalence was higher among those with underlying cardiac disease (23.4%), compared with those without (6.2%).
Acute ischemic heart disease (5.5%) and acute heart failure (5.4%) were the most prevalent events; 0.3% of participants had acute myocarditis or pericarditis.
Risk factors varied, depending on underlying cardiac disease status. Those who experienced one or more acute cardiac events had a greater risk for intensive care unit admission (adjusted risk ratio,1.9) and in-hospital death (aRR, 1.7) versus those who did not.
In multivariable analyses, the risk of experiencing acute heart failure was significantly greater among men (aRR, 1.5) and among those with a history of congestive heart failure (aRR, 13.5), atrial fibrillation (aRR, 1.6) or hypertension (aRR,1.3).
Among patients who experienced one or more acute cardiac events, 39.2% required an intensive care unit stay for a median of 5 days. Approximately 22.4% required invasive mechanical ventilation or extracorporeal membrane oxygenation, and 21.1% died while hospitalized.
“Persons at greater risk for experiencing acute cardiac events during COVID-19–associated hospitalizations might benefit from more intensive clinical evaluation and monitoring during hospitalization,” the authors conclude.
The team currently is taking a closer look at acute myocarditis among patients hospitalized with COVID-19, Dr. Woodruff said. Preliminary results were presented at the 2022 annual scientific sessions of the American Heart Association and a paper is forthcoming.
Contemporary data needed
James A. de Lemos, MD, co-chair of the American Heart Association’s COVID-19 CVD Registry Steering Committee and professor of medicine at the University of Texas Southwestern Medical Center, Dallas, said the findings mirror his team’s clinical experience in 2020 and 2021 and echo what was seen in the AHA COVID registry: that is, a 0.3% rate of myocarditis.
“The major caveat is that [the findings] may not be generalizable to contemporary COVID infection, both due to changing viral variants and higher levels of immunity in the population,” he said.
“Rates of COVID hospitalization are markedly lower with the current dominant variants, and we would expect the cardiac risk to be lower as well. I would like to see more contemporary data with current variants, particularly focused on higher risk patients with cardiovascular disease,” Dr. de Lemos added.
In a related editorial, George A. Mensa, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Md., and colleagues suggest that the broader impact of the COVID-19 pandemic on human health remains “incompletely examined.”
“The impact of COVID-19 on cardiovascular mortality, in particular, appears to have varied widely, with no large increases seen in a number of the most developed countries but marked increases in hypertensive heart disease mortality seen in the United States in 2021,” they conclude. “The potential contribution of COVID-19 to these deaths, either directly or indirectly, remains to be determined.”
No commercial funding or relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY




