User login
Gender and Sports: Can Science Enable Fair Competition?
The allegations against Algerian boxer Imane Khelif at the Paris Olympics raised the questions of intersexuality and its implications in competitive sports. This news organization has decided to delve into the topic to assist doctors who suspect a similar condition in their patients. No certain clinical data about Ms. Khelif have been made public, so this article does not concern the boxer but rather takes inspiration from the media controversy.
What Is Intersexuality?
Intersexuality encompasses a spectrum of variations in sexual development that lead to the simultaneous presence of typical male and female characteristics. As reiterated by the United Nations Office of the High Commissioner for Human Rights, the medical definition does not affect the patient’s self-identification of gender or sexual orientation.
“The percentage of people who fall within the intersexuality spectrum is less than 0.5 per thousand of the general population, but there are no precise statistics, given the difficulty of definition,” said Roberto Lala, MD, pediatric endocrinologist and president of the Federation of Rare Childhood Diseases.
Indeed, there is not only a strict definition of intersexuality that involves a significant presence of these mixed physical characteristics in a way that conditions the self-image of the subject but also a broad definition, said Dr. Lala. “For example, clitoral hypertrophy in a female otherwise conforming to the female gender, which does not raise doubts about identity,” he said.
Chromosomes, Genes, and Hormones
“The human body is, so to speak, programmed to take on female appearances in development and shifts toward male ones only if exposed to testosterone and other factors. For this to happen, testosterone must be produced during embryonic development, and it must function properly,” said Paolo Moghetti, full professor of endocrinology at the University of Verona, Italy.
The protein encoded by SRY, which is located on the Y chromosome, determines the development of the testicles from undifferentiated tissue of the embryonic gonads. The testicles of the embryo then produce testosterone. The absence of the Y chromosome is a common characteristic of most female individuals. However, there are individuals with a female phenotype who have X and Y chromosomes but lack SRY or have a variant of it that is not entirely functional.
Numerous other chromosomal or genetic variations can lead to alterations in sexual differentiation. “In phenotypically male adult subjects (with a chromosomal makeup of 46XY) with complete androgen insensitivity (so-called Morris syndrome), testosterone levels in the blood are elevated, above normal even for a male, but the hormone is totally ineffective, and the phenotype is totally female at birth, with completely female development of secondary sexual characteristics at puberty,” said Dr. Moghetti.
This means that affected individuals have well-developed breasts and a complete lack or extremely reduced presence of hair, including underarm and pubic hair. Menstruation is also completely absent because there is no uterus, and there are testes, not visible because they are considered in the abdomen.
“There are syndromes that are currently considered congenital but not genetic, of which a genetic origin will probably be identified in the future,” said Dr. Lala.
Some variations in sexual development can be diagnosed prenatally, such as an alteration of the number of sex chromosomes or a discordance between the morphologic characteristics highlighted by ultrasound and the genotype detected by amniocentesis. Some variations are evident at birth because of atypical anatomical characteristics. Others are diagnosed during puberty or later in adulthood, in the presence of infertility. The Italian National Institute of Health details these variations on its website, describing the characteristics that determine diagnosis and treatment.
Pathologies or Variations?
Some anomalies in sexual development negatively affect the patient’s physical health. One example is congenital adrenal hyperplasia. “It results from an inherited defect of the adrenal glands, which reduces cortisol production while increasing testosterone production,” said Dr. Lala. “In addition to the appearance of male characteristics in females, in more severe forms, it carries the risk of collapse and shock and requires pharmacological treatment.” It is undoubtedly a pathology.
Other variations in sexual characteristics do not affect the patient’s physical health negatively. They may, however, have a psychologic effect, sometimes a significant one, because of the lack of social acceptance of a person who cannot be classified within the binary classification of sexes.
“Conditions in which mixed male and female aspects are clearly evident have been and are still pathologized by the family, the treating physician, and society,” said Dr. Lala. “In the late 1970s, when a child was born with intersexual anatomical characteristics, it was common practice to surgically intervene, making them female, because it was technically easier.”
Over the years, patients who, as they grew up, were dissatisfied with the solution adopted at birth began to make their voices heard, Dr. Lala added. Scientific societies and international organizations have spoken out against subjecting intersexual newborns to surgical interventions that are not medically necessary. “Nowadays, decisions are made on a case-by-case basis, taking into account the families’ wishes. Interventions are justified with medical reasons, which are often very nuanced,” Dr. Lala concluded.
Implications for Sports
Traditionally, athletes participating in competitions in certain sports have been divided into male and female categories to ensure a certain equity and uniformity in performance. Over the years, the emergence of new information about sexual development has made it necessary to update the criteria used in this division.
The main factor responsible for the performance diversity between males and females is the action of testosterone on the male and female organism. “Testosterone has important effects on muscle mass and enhances training results,” said Dr. Moghetti. “As a demonstration of this fact, before puberty, the best performances in athletics or swimming by males and females are similar, then males gain a significant advantage of around 10%-20%.”
A few years ago, the World Athletics Federation conducted widespread screening of athletes participating in its world championships. “It identified a small group of individuals with potentially abnormal testosterone levels for the female sex,” said Dr. Moghetti. “Some were found to be doping, others had genetic defects, and for some, an interpretation was not even possible.”
Some of the individuals had a male genotype but a defect in 5-alpha-reductase, an enzyme essential for the formation of male genitals and hair growth. An athlete with these characteristics, assigned female sex at birth, has a male level of testosterone that stimulates the accumulation of muscle mass, Dr. Moghetti explained. Therefore, the individual has a considerable advantage in performances influenced by this hormone.
“In the end, the Federation decided to set limits on the testosterone levels of athletes participating in certain types of races, especially those in middle distance, that appeared to be more sensitive to differences in hormone levels,” said Dr. Moghetti. “The limitation does not apply to athletes with Morris syndrome, ie, with a male genotype and complete resistance to testosterone, for whom the high level of this hormone does not provide any advantage.” Given the complexity of the problem, he hopes for a case-by-case policy that considers the needs of patients with genetic alterations and those of athletes who have to compete with them.
Not the First Time
A recent incident underscored the difficulty of regulating such complex issues. The World Athletics Federation excluded South African middle-distance runner Caster Semenya from competitions years ago because of excessively high testosterone levels.
“The Federation’s regulations recommend that athletes in these cases reduce hormone levels to values below the threshold of 5 nmol/L of blood for a period of at least 6 months before the race by using hormonal contraceptives. The use of such drugs does not pose a health risk, as they are substances normally taken by women for contraception purposes,” said Amelia Filippelli, a pharmacologist at the University of Salerno in Italy. The South African middle-distance runner refused the drug and appealed to the Court of Arbitration for Sport and later to the Swiss Federal Court. Both rejected her appeal. Finally, Ms. Semenya appealed to the European Court of Human Rights, which in 2023 recognized a violation of her rights but does not have the authority to order a change in the Federation’s regulations.
Beyond the ideologic positions of nonexperts, therefore, the issue is still the subject of debate in the scientific community, which is evaluating not only its medical aspects but also its ethical implications.
This story was translated from Univadis Italy, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The allegations against Algerian boxer Imane Khelif at the Paris Olympics raised the questions of intersexuality and its implications in competitive sports. This news organization has decided to delve into the topic to assist doctors who suspect a similar condition in their patients. No certain clinical data about Ms. Khelif have been made public, so this article does not concern the boxer but rather takes inspiration from the media controversy.
What Is Intersexuality?
Intersexuality encompasses a spectrum of variations in sexual development that lead to the simultaneous presence of typical male and female characteristics. As reiterated by the United Nations Office of the High Commissioner for Human Rights, the medical definition does not affect the patient’s self-identification of gender or sexual orientation.
“The percentage of people who fall within the intersexuality spectrum is less than 0.5 per thousand of the general population, but there are no precise statistics, given the difficulty of definition,” said Roberto Lala, MD, pediatric endocrinologist and president of the Federation of Rare Childhood Diseases.
Indeed, there is not only a strict definition of intersexuality that involves a significant presence of these mixed physical characteristics in a way that conditions the self-image of the subject but also a broad definition, said Dr. Lala. “For example, clitoral hypertrophy in a female otherwise conforming to the female gender, which does not raise doubts about identity,” he said.
Chromosomes, Genes, and Hormones
“The human body is, so to speak, programmed to take on female appearances in development and shifts toward male ones only if exposed to testosterone and other factors. For this to happen, testosterone must be produced during embryonic development, and it must function properly,” said Paolo Moghetti, full professor of endocrinology at the University of Verona, Italy.
The protein encoded by SRY, which is located on the Y chromosome, determines the development of the testicles from undifferentiated tissue of the embryonic gonads. The testicles of the embryo then produce testosterone. The absence of the Y chromosome is a common characteristic of most female individuals. However, there are individuals with a female phenotype who have X and Y chromosomes but lack SRY or have a variant of it that is not entirely functional.
Numerous other chromosomal or genetic variations can lead to alterations in sexual differentiation. “In phenotypically male adult subjects (with a chromosomal makeup of 46XY) with complete androgen insensitivity (so-called Morris syndrome), testosterone levels in the blood are elevated, above normal even for a male, but the hormone is totally ineffective, and the phenotype is totally female at birth, with completely female development of secondary sexual characteristics at puberty,” said Dr. Moghetti.
This means that affected individuals have well-developed breasts and a complete lack or extremely reduced presence of hair, including underarm and pubic hair. Menstruation is also completely absent because there is no uterus, and there are testes, not visible because they are considered in the abdomen.
“There are syndromes that are currently considered congenital but not genetic, of which a genetic origin will probably be identified in the future,” said Dr. Lala.
Some variations in sexual development can be diagnosed prenatally, such as an alteration of the number of sex chromosomes or a discordance between the morphologic characteristics highlighted by ultrasound and the genotype detected by amniocentesis. Some variations are evident at birth because of atypical anatomical characteristics. Others are diagnosed during puberty or later in adulthood, in the presence of infertility. The Italian National Institute of Health details these variations on its website, describing the characteristics that determine diagnosis and treatment.
Pathologies or Variations?
Some anomalies in sexual development negatively affect the patient’s physical health. One example is congenital adrenal hyperplasia. “It results from an inherited defect of the adrenal glands, which reduces cortisol production while increasing testosterone production,” said Dr. Lala. “In addition to the appearance of male characteristics in females, in more severe forms, it carries the risk of collapse and shock and requires pharmacological treatment.” It is undoubtedly a pathology.
Other variations in sexual characteristics do not affect the patient’s physical health negatively. They may, however, have a psychologic effect, sometimes a significant one, because of the lack of social acceptance of a person who cannot be classified within the binary classification of sexes.
“Conditions in which mixed male and female aspects are clearly evident have been and are still pathologized by the family, the treating physician, and society,” said Dr. Lala. “In the late 1970s, when a child was born with intersexual anatomical characteristics, it was common practice to surgically intervene, making them female, because it was technically easier.”
Over the years, patients who, as they grew up, were dissatisfied with the solution adopted at birth began to make their voices heard, Dr. Lala added. Scientific societies and international organizations have spoken out against subjecting intersexual newborns to surgical interventions that are not medically necessary. “Nowadays, decisions are made on a case-by-case basis, taking into account the families’ wishes. Interventions are justified with medical reasons, which are often very nuanced,” Dr. Lala concluded.
Implications for Sports
Traditionally, athletes participating in competitions in certain sports have been divided into male and female categories to ensure a certain equity and uniformity in performance. Over the years, the emergence of new information about sexual development has made it necessary to update the criteria used in this division.
The main factor responsible for the performance diversity between males and females is the action of testosterone on the male and female organism. “Testosterone has important effects on muscle mass and enhances training results,” said Dr. Moghetti. “As a demonstration of this fact, before puberty, the best performances in athletics or swimming by males and females are similar, then males gain a significant advantage of around 10%-20%.”
A few years ago, the World Athletics Federation conducted widespread screening of athletes participating in its world championships. “It identified a small group of individuals with potentially abnormal testosterone levels for the female sex,” said Dr. Moghetti. “Some were found to be doping, others had genetic defects, and for some, an interpretation was not even possible.”
Some of the individuals had a male genotype but a defect in 5-alpha-reductase, an enzyme essential for the formation of male genitals and hair growth. An athlete with these characteristics, assigned female sex at birth, has a male level of testosterone that stimulates the accumulation of muscle mass, Dr. Moghetti explained. Therefore, the individual has a considerable advantage in performances influenced by this hormone.
“In the end, the Federation decided to set limits on the testosterone levels of athletes participating in certain types of races, especially those in middle distance, that appeared to be more sensitive to differences in hormone levels,” said Dr. Moghetti. “The limitation does not apply to athletes with Morris syndrome, ie, with a male genotype and complete resistance to testosterone, for whom the high level of this hormone does not provide any advantage.” Given the complexity of the problem, he hopes for a case-by-case policy that considers the needs of patients with genetic alterations and those of athletes who have to compete with them.
Not the First Time
A recent incident underscored the difficulty of regulating such complex issues. The World Athletics Federation excluded South African middle-distance runner Caster Semenya from competitions years ago because of excessively high testosterone levels.
“The Federation’s regulations recommend that athletes in these cases reduce hormone levels to values below the threshold of 5 nmol/L of blood for a period of at least 6 months before the race by using hormonal contraceptives. The use of such drugs does not pose a health risk, as they are substances normally taken by women for contraception purposes,” said Amelia Filippelli, a pharmacologist at the University of Salerno in Italy. The South African middle-distance runner refused the drug and appealed to the Court of Arbitration for Sport and later to the Swiss Federal Court. Both rejected her appeal. Finally, Ms. Semenya appealed to the European Court of Human Rights, which in 2023 recognized a violation of her rights but does not have the authority to order a change in the Federation’s regulations.
Beyond the ideologic positions of nonexperts, therefore, the issue is still the subject of debate in the scientific community, which is evaluating not only its medical aspects but also its ethical implications.
This story was translated from Univadis Italy, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The allegations against Algerian boxer Imane Khelif at the Paris Olympics raised the questions of intersexuality and its implications in competitive sports. This news organization has decided to delve into the topic to assist doctors who suspect a similar condition in their patients. No certain clinical data about Ms. Khelif have been made public, so this article does not concern the boxer but rather takes inspiration from the media controversy.
What Is Intersexuality?
Intersexuality encompasses a spectrum of variations in sexual development that lead to the simultaneous presence of typical male and female characteristics. As reiterated by the United Nations Office of the High Commissioner for Human Rights, the medical definition does not affect the patient’s self-identification of gender or sexual orientation.
“The percentage of people who fall within the intersexuality spectrum is less than 0.5 per thousand of the general population, but there are no precise statistics, given the difficulty of definition,” said Roberto Lala, MD, pediatric endocrinologist and president of the Federation of Rare Childhood Diseases.
Indeed, there is not only a strict definition of intersexuality that involves a significant presence of these mixed physical characteristics in a way that conditions the self-image of the subject but also a broad definition, said Dr. Lala. “For example, clitoral hypertrophy in a female otherwise conforming to the female gender, which does not raise doubts about identity,” he said.
Chromosomes, Genes, and Hormones
“The human body is, so to speak, programmed to take on female appearances in development and shifts toward male ones only if exposed to testosterone and other factors. For this to happen, testosterone must be produced during embryonic development, and it must function properly,” said Paolo Moghetti, full professor of endocrinology at the University of Verona, Italy.
The protein encoded by SRY, which is located on the Y chromosome, determines the development of the testicles from undifferentiated tissue of the embryonic gonads. The testicles of the embryo then produce testosterone. The absence of the Y chromosome is a common characteristic of most female individuals. However, there are individuals with a female phenotype who have X and Y chromosomes but lack SRY or have a variant of it that is not entirely functional.
Numerous other chromosomal or genetic variations can lead to alterations in sexual differentiation. “In phenotypically male adult subjects (with a chromosomal makeup of 46XY) with complete androgen insensitivity (so-called Morris syndrome), testosterone levels in the blood are elevated, above normal even for a male, but the hormone is totally ineffective, and the phenotype is totally female at birth, with completely female development of secondary sexual characteristics at puberty,” said Dr. Moghetti.
This means that affected individuals have well-developed breasts and a complete lack or extremely reduced presence of hair, including underarm and pubic hair. Menstruation is also completely absent because there is no uterus, and there are testes, not visible because they are considered in the abdomen.
“There are syndromes that are currently considered congenital but not genetic, of which a genetic origin will probably be identified in the future,” said Dr. Lala.
Some variations in sexual development can be diagnosed prenatally, such as an alteration of the number of sex chromosomes or a discordance between the morphologic characteristics highlighted by ultrasound and the genotype detected by amniocentesis. Some variations are evident at birth because of atypical anatomical characteristics. Others are diagnosed during puberty or later in adulthood, in the presence of infertility. The Italian National Institute of Health details these variations on its website, describing the characteristics that determine diagnosis and treatment.
Pathologies or Variations?
Some anomalies in sexual development negatively affect the patient’s physical health. One example is congenital adrenal hyperplasia. “It results from an inherited defect of the adrenal glands, which reduces cortisol production while increasing testosterone production,” said Dr. Lala. “In addition to the appearance of male characteristics in females, in more severe forms, it carries the risk of collapse and shock and requires pharmacological treatment.” It is undoubtedly a pathology.
Other variations in sexual characteristics do not affect the patient’s physical health negatively. They may, however, have a psychologic effect, sometimes a significant one, because of the lack of social acceptance of a person who cannot be classified within the binary classification of sexes.
“Conditions in which mixed male and female aspects are clearly evident have been and are still pathologized by the family, the treating physician, and society,” said Dr. Lala. “In the late 1970s, when a child was born with intersexual anatomical characteristics, it was common practice to surgically intervene, making them female, because it was technically easier.”
Over the years, patients who, as they grew up, were dissatisfied with the solution adopted at birth began to make their voices heard, Dr. Lala added. Scientific societies and international organizations have spoken out against subjecting intersexual newborns to surgical interventions that are not medically necessary. “Nowadays, decisions are made on a case-by-case basis, taking into account the families’ wishes. Interventions are justified with medical reasons, which are often very nuanced,” Dr. Lala concluded.
Implications for Sports
Traditionally, athletes participating in competitions in certain sports have been divided into male and female categories to ensure a certain equity and uniformity in performance. Over the years, the emergence of new information about sexual development has made it necessary to update the criteria used in this division.
The main factor responsible for the performance diversity between males and females is the action of testosterone on the male and female organism. “Testosterone has important effects on muscle mass and enhances training results,” said Dr. Moghetti. “As a demonstration of this fact, before puberty, the best performances in athletics or swimming by males and females are similar, then males gain a significant advantage of around 10%-20%.”
A few years ago, the World Athletics Federation conducted widespread screening of athletes participating in its world championships. “It identified a small group of individuals with potentially abnormal testosterone levels for the female sex,” said Dr. Moghetti. “Some were found to be doping, others had genetic defects, and for some, an interpretation was not even possible.”
Some of the individuals had a male genotype but a defect in 5-alpha-reductase, an enzyme essential for the formation of male genitals and hair growth. An athlete with these characteristics, assigned female sex at birth, has a male level of testosterone that stimulates the accumulation of muscle mass, Dr. Moghetti explained. Therefore, the individual has a considerable advantage in performances influenced by this hormone.
“In the end, the Federation decided to set limits on the testosterone levels of athletes participating in certain types of races, especially those in middle distance, that appeared to be more sensitive to differences in hormone levels,” said Dr. Moghetti. “The limitation does not apply to athletes with Morris syndrome, ie, with a male genotype and complete resistance to testosterone, for whom the high level of this hormone does not provide any advantage.” Given the complexity of the problem, he hopes for a case-by-case policy that considers the needs of patients with genetic alterations and those of athletes who have to compete with them.
Not the First Time
A recent incident underscored the difficulty of regulating such complex issues. The World Athletics Federation excluded South African middle-distance runner Caster Semenya from competitions years ago because of excessively high testosterone levels.
“The Federation’s regulations recommend that athletes in these cases reduce hormone levels to values below the threshold of 5 nmol/L of blood for a period of at least 6 months before the race by using hormonal contraceptives. The use of such drugs does not pose a health risk, as they are substances normally taken by women for contraception purposes,” said Amelia Filippelli, a pharmacologist at the University of Salerno in Italy. The South African middle-distance runner refused the drug and appealed to the Court of Arbitration for Sport and later to the Swiss Federal Court. Both rejected her appeal. Finally, Ms. Semenya appealed to the European Court of Human Rights, which in 2023 recognized a violation of her rights but does not have the authority to order a change in the Federation’s regulations.
Beyond the ideologic positions of nonexperts, therefore, the issue is still the subject of debate in the scientific community, which is evaluating not only its medical aspects but also its ethical implications.
This story was translated from Univadis Italy, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Hand Sanitizers and Alcohol Abuse: A Dangerous Cocktail
Since the COVID-19 pandemic, alcoholic hand sanitizers have become widely accessible nationwide. They can pose a problem, especially for emergency departments, when alcohol-dependent patients start drinking them. One example that demonstrates the challenge of diagnosing alcohol abuse is the medical history of a young man, as reported by Mahmoud El Hussein, MD, and colleagues from Hôpital Lariboisière in Paris, France.
Presentation and History
A 26-year-old man presented with severe abdominal pain at the emergency department. Upon arrival, he was hemodynamically stable but nervous and verbally aggressive at times. The patient reported no relevant preexisting conditions and was not taking any medications.
Findings
Upon initial physical examination, the patient had a soft, diffusely tender abdomen; tachycardia; and notably poor hygiene. The patient was afebrile. An ECG confirmed the tachycardia but showed no signs of ischemia. Blood work, except for slightly elevated liver values, did not reveal any abnormalities, particularly ruling out bleeding or kidney disease.
A urease rapid test to rule out kidney stones also showed no pathologic findings. In consultation with the surgical department, a CT scan of the abdomen was performed to rule out organ perforation, volvulus, or mesenteric ischemia. Only signs of fatty liver were found.
A Neighbor’s Tip
During all examinations, the patient’s abdomen was repeatedly palpated to promptly detect signs of an acute abdomen. However, there was never any defense tension at any point.
Intravenous analgesics and proton pump inhibitors (ie, paracetamol, phloroglucin, and pantoprazole) did not relieve the patient’s symptoms. Morphine was administered intravenously for sedation.
Only after a frustrating diagnostic process did a neighbor of the patient inform a nurse that he suspected the patient of stealing and consuming hand sanitizer. With the patient’s consent, a blood alcohol test was performed, revealing a blood alcohol concentration of 0.2% (2 g/L). A urine test, also conducted with the patient’s consent, tested positive for tetrahydrocannabinol. Additional tests showed the following results:
- Venous pH: 7.29 (normal, 7.32-7.38)
- Anion gap (mEq/L): 14 (normal, 3-9)
- Ketone bodies (mmol/L): 0.2 (normal, < 0.6)
- Calculated serum osmolality (mOsm/kg): 292 (normal, 285-295)
- Measured serum osmolality (mOsm/kg): 320 (normal, 285-295)
- Osmolality gap (mOsm/kg): 2 (normal, < 10)
The patient was informed of the test results and confessed to feigning abdominal pain. He was dependent on alcohol and experiencing withdrawal symptoms. The patient had stolen seven 475-mL bottles of hand sanitizer and consumed one and a half in the past 4-6 hours. According to the authors, the sanitizer consisted of 80% ethanol, 1.45% glycerol, and 0.13% hydrogen peroxide.
Discussion
In Germany, alcohol consumption results in approximately €57 billion in direct economic costs annually, according to data from the Federal Ministry of Health. In 2021, about 7.9 million people aged 18-64 years consumed alcohol in a risky manner (approximately 9.6% of the German population). About 9 million people (approximately 11%) were classified as alcoholics.
Therefore, staff in emergency departments, where potential abusers encounter a wide range of potential abuse items, should exercise caution.
Although hand sanitizers are mainly composed of ethanol, they may also contain isopropanol, methanol, or acetone. Methanol poisoning can cause abdominal pain, visual disturbances, central nervous system damage, and death. Other alcohols such as ethylene glycol, commonly found in antifreeze, can distort blood values (lactate) and complicate a correct diagnosis.
Physicians working in emergency departments should proceed with caution when suspecting alcohol abuse. Questioning the patient’s environment and determining additional laboratory parameters (such as osmolality gap in the case report) can help unmask substance abuse if it is in doubt.
This story was translated from Univadis Germany, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Since the COVID-19 pandemic, alcoholic hand sanitizers have become widely accessible nationwide. They can pose a problem, especially for emergency departments, when alcohol-dependent patients start drinking them. One example that demonstrates the challenge of diagnosing alcohol abuse is the medical history of a young man, as reported by Mahmoud El Hussein, MD, and colleagues from Hôpital Lariboisière in Paris, France.
Presentation and History
A 26-year-old man presented with severe abdominal pain at the emergency department. Upon arrival, he was hemodynamically stable but nervous and verbally aggressive at times. The patient reported no relevant preexisting conditions and was not taking any medications.
Findings
Upon initial physical examination, the patient had a soft, diffusely tender abdomen; tachycardia; and notably poor hygiene. The patient was afebrile. An ECG confirmed the tachycardia but showed no signs of ischemia. Blood work, except for slightly elevated liver values, did not reveal any abnormalities, particularly ruling out bleeding or kidney disease.
A urease rapid test to rule out kidney stones also showed no pathologic findings. In consultation with the surgical department, a CT scan of the abdomen was performed to rule out organ perforation, volvulus, or mesenteric ischemia. Only signs of fatty liver were found.
A Neighbor’s Tip
During all examinations, the patient’s abdomen was repeatedly palpated to promptly detect signs of an acute abdomen. However, there was never any defense tension at any point.
Intravenous analgesics and proton pump inhibitors (ie, paracetamol, phloroglucin, and pantoprazole) did not relieve the patient’s symptoms. Morphine was administered intravenously for sedation.
Only after a frustrating diagnostic process did a neighbor of the patient inform a nurse that he suspected the patient of stealing and consuming hand sanitizer. With the patient’s consent, a blood alcohol test was performed, revealing a blood alcohol concentration of 0.2% (2 g/L). A urine test, also conducted with the patient’s consent, tested positive for tetrahydrocannabinol. Additional tests showed the following results:
- Venous pH: 7.29 (normal, 7.32-7.38)
- Anion gap (mEq/L): 14 (normal, 3-9)
- Ketone bodies (mmol/L): 0.2 (normal, < 0.6)
- Calculated serum osmolality (mOsm/kg): 292 (normal, 285-295)
- Measured serum osmolality (mOsm/kg): 320 (normal, 285-295)
- Osmolality gap (mOsm/kg): 2 (normal, < 10)
The patient was informed of the test results and confessed to feigning abdominal pain. He was dependent on alcohol and experiencing withdrawal symptoms. The patient had stolen seven 475-mL bottles of hand sanitizer and consumed one and a half in the past 4-6 hours. According to the authors, the sanitizer consisted of 80% ethanol, 1.45% glycerol, and 0.13% hydrogen peroxide.
Discussion
In Germany, alcohol consumption results in approximately €57 billion in direct economic costs annually, according to data from the Federal Ministry of Health. In 2021, about 7.9 million people aged 18-64 years consumed alcohol in a risky manner (approximately 9.6% of the German population). About 9 million people (approximately 11%) were classified as alcoholics.
Therefore, staff in emergency departments, where potential abusers encounter a wide range of potential abuse items, should exercise caution.
Although hand sanitizers are mainly composed of ethanol, they may also contain isopropanol, methanol, or acetone. Methanol poisoning can cause abdominal pain, visual disturbances, central nervous system damage, and death. Other alcohols such as ethylene glycol, commonly found in antifreeze, can distort blood values (lactate) and complicate a correct diagnosis.
Physicians working in emergency departments should proceed with caution when suspecting alcohol abuse. Questioning the patient’s environment and determining additional laboratory parameters (such as osmolality gap in the case report) can help unmask substance abuse if it is in doubt.
This story was translated from Univadis Germany, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Since the COVID-19 pandemic, alcoholic hand sanitizers have become widely accessible nationwide. They can pose a problem, especially for emergency departments, when alcohol-dependent patients start drinking them. One example that demonstrates the challenge of diagnosing alcohol abuse is the medical history of a young man, as reported by Mahmoud El Hussein, MD, and colleagues from Hôpital Lariboisière in Paris, France.
Presentation and History
A 26-year-old man presented with severe abdominal pain at the emergency department. Upon arrival, he was hemodynamically stable but nervous and verbally aggressive at times. The patient reported no relevant preexisting conditions and was not taking any medications.
Findings
Upon initial physical examination, the patient had a soft, diffusely tender abdomen; tachycardia; and notably poor hygiene. The patient was afebrile. An ECG confirmed the tachycardia but showed no signs of ischemia. Blood work, except for slightly elevated liver values, did not reveal any abnormalities, particularly ruling out bleeding or kidney disease.
A urease rapid test to rule out kidney stones also showed no pathologic findings. In consultation with the surgical department, a CT scan of the abdomen was performed to rule out organ perforation, volvulus, or mesenteric ischemia. Only signs of fatty liver were found.
A Neighbor’s Tip
During all examinations, the patient’s abdomen was repeatedly palpated to promptly detect signs of an acute abdomen. However, there was never any defense tension at any point.
Intravenous analgesics and proton pump inhibitors (ie, paracetamol, phloroglucin, and pantoprazole) did not relieve the patient’s symptoms. Morphine was administered intravenously for sedation.
Only after a frustrating diagnostic process did a neighbor of the patient inform a nurse that he suspected the patient of stealing and consuming hand sanitizer. With the patient’s consent, a blood alcohol test was performed, revealing a blood alcohol concentration of 0.2% (2 g/L). A urine test, also conducted with the patient’s consent, tested positive for tetrahydrocannabinol. Additional tests showed the following results:
- Venous pH: 7.29 (normal, 7.32-7.38)
- Anion gap (mEq/L): 14 (normal, 3-9)
- Ketone bodies (mmol/L): 0.2 (normal, < 0.6)
- Calculated serum osmolality (mOsm/kg): 292 (normal, 285-295)
- Measured serum osmolality (mOsm/kg): 320 (normal, 285-295)
- Osmolality gap (mOsm/kg): 2 (normal, < 10)
The patient was informed of the test results and confessed to feigning abdominal pain. He was dependent on alcohol and experiencing withdrawal symptoms. The patient had stolen seven 475-mL bottles of hand sanitizer and consumed one and a half in the past 4-6 hours. According to the authors, the sanitizer consisted of 80% ethanol, 1.45% glycerol, and 0.13% hydrogen peroxide.
Discussion
In Germany, alcohol consumption results in approximately €57 billion in direct economic costs annually, according to data from the Federal Ministry of Health. In 2021, about 7.9 million people aged 18-64 years consumed alcohol in a risky manner (approximately 9.6% of the German population). About 9 million people (approximately 11%) were classified as alcoholics.
Therefore, staff in emergency departments, where potential abusers encounter a wide range of potential abuse items, should exercise caution.
Although hand sanitizers are mainly composed of ethanol, they may also contain isopropanol, methanol, or acetone. Methanol poisoning can cause abdominal pain, visual disturbances, central nervous system damage, and death. Other alcohols such as ethylene glycol, commonly found in antifreeze, can distort blood values (lactate) and complicate a correct diagnosis.
Physicians working in emergency departments should proceed with caution when suspecting alcohol abuse. Questioning the patient’s environment and determining additional laboratory parameters (such as osmolality gap in the case report) can help unmask substance abuse if it is in doubt.
This story was translated from Univadis Germany, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Private Equity in Medicine: Cardiology in the Crosshairs
This transcript has been edited for clarity.
Robert A. Harrington, MD: I’m continuing my series of conversations with leaders in the field of cardiovascular medicine who are working on interesting projects and making contributions in the science and policy space. We have three guests joining us today who have recently written two papers in the Journal of the American College of Cardiology. One is an original research paper dealing with the issue of private equity’s acquisition of outpatient cardiology practices. And the second is an editorial that really tries to get at why this is happening. Is it a problem? Is it a solution to a problem?
Fortunately, I have all three as guests to think about this important issue that has implications for clinical care, reimbursement, physician wellness, and clinician wellness, and it has implications regarding public policy and how we should be thinking about the practice of medicine in this country.
Dr. Victoria L. Bartlett is an internal medicine resident at Brigham and Women’s Hospital in Boston, and a research fellow in the Smith Center at the Beth Israel Lahey medical center in Boston. Dr Rishi K. Wadhera is the senior author of the paper written by Dr. Bartlett. Dr. Rishi is associate professor of medicine at Harvard Medical School, and he is the associate director of the Smith Center at Beth Israel Lahey.
Rishi K. Wadhera, MD, MPP, MPhil: Thanks for having us, Bob.
Dr. Harrington: The editorialist, Dr. Ed Fry, is the national service line leader for cardiovascular medicine, for Ascension Health. Dr. Frey is a recent past president of the American College of Cardiology (ACC).
Edward T. A. Fry, MD: Great to be here. Thanks.
What is private equity? Why the interest in medicine?
Dr. Harrington: I was intrigued by the papers and it caused me to do a deeper dive into some of the earlier works that you have referenced about this growing topic of private equity making its way into medical practice. Rishi, I’ll start with you. For the casual reader like myself, what is the business of private equity?
Dr. Wadhera: Private equity firms basically used pooled investments from multiple sources. These can be individual and institutional investors, pension funds, endowments, and they use those funds to invest in private companies that have the potential to return a profit. The goal really is to turn a profit for institutional investors over a shorter time horizon.
Dr. Harrington: How do they do that? I can understand, you buy a factory and you want to make the factory a little more efficient, and you think that perhaps, by combining some technologies, etc., that you might have in other factories, you can drive more value out of the one you just invested in in a short period of time. What’s the general business sense of how they’re going to do that in a cardiology practice? Is it all about making us more efficient?
Dr. Wadhera: Operational efficiency is the overarching theme here. One could argue that perhaps, private equity firms have the expertise to bring that kind of organizational know-how and operational efficiency to medicine. But there’s evidence that the way that private equity firms maximize their margin is maybe through mechanisms that aren’t necessarily good for patient care, such as reduced nursing staffing. When private equity acquires hospitals or practices in the same location, they have greater negotiating power at the payer table, to have higher prices for the services they deliver. There’s a lot of discussion about whether the sort of changes that private equity firms tend to implement are good or bad for patient care and also for clinicians.
Dr. Harrington: Great summary. Ed. Why is this happening in medicine? What did we do in medicine that made us ripe for investment by private equity? When you and I started out years ago, I don’t think we ever would have thought that this was in the future.
Dr. Fry: I think number one, as we know, is that medicine represents about 20% of our economy. There are huge amounts of money involved in these considerations. If players in this space can access even a small fraction of that money, it’s a lot of money and a lot of incentive for them.
In medicine in general, and then maybe more specifically, in cardiology, we’ve seen a shift away from private practice into employed practice. When people made those decisions over the past 10-15 years, there were certainly positives and risks that they took. I think for some, along the way, they realized that perhaps they gave up more than they thought in terms of control and running their own business and the opportunity to shape that themselves and be rewarded for that as they were in private practice. In cardiology, more specifically, we’ve seen this shift to the outpatient space: moving diagnostics and even therapeutics into ambulatory surgical centers and outpatient-based labs, and that is another potential source of revenue for these private equity companies.
As I wrote in the editorial, there are certainly a lot of pressures and frustrations that the day-to-day clinician feels, and maybe, this move to private equity is more of a symptom of those concerns and that this could be an opportunity to take the bull by the horns again in cardiology. We’ve evolved from a predominantly hospital-based acute care specialty into one of diagnosis, chronic disease management, and longitudinal care punctuated by diagnostics and therapeutics, which are, again, I think, attractive to private equity firms as potential sources for revenue.
Dr. Harrington: Ed, why cardiology? What’s happened over the years that has led to that appearance, if you will, of private equity and cardiovascular medicine?
Dr. Fry: Some of the earlier specialties were dermatology, ophthalmology, and gastroenterology, in particular. And interestingly, those tend to be specialties that have less chronic disease management and are more based on procedures and things like that. Within cardiology, obviously, the big driver is that our population is aging: 11,000 people turn 65 every day and become eligible for Medicare. With that, we see a rise in disease prevalence and then the rise in risk factors, obviously, with obesity and diabetes driving that, so there are more people who are going to have an illness that requires evaluation, diagnostics, and procedures. Because of that, it is a very target rich environment for private equity.
Dr. Harrington: That’s great background. Now, let’s dive into what you did, Victoria. What got you interested in the question? And give us some background on the literature that you were trying to build upon when you asked your series of questions.
Victoria L. Bartlett, MD: There’s been a lot of interest in private equity acquisitions and healthcare. A lot of the existing literature has been around hospital acquisitions and what happens there. There’s some literature, as you’ve mentioned, in outpatient practices, in certain specialties, where private equity has existed a little bit longer than in cardiology. They’ve been asking really similar questions to what we have been asking about cardiology, which is what happens when practices are acquired.
A kind of overview is that many of those studies have found increased costs to payers, to patients, and many have also found evidence of decreased quality. The evidence for the latter is honestly more difficult to figure out, but there has been evidence of decreased nursing ratios in nursing homes. There’s been evidence of changing the mix in clinics to more advanced practice providers than physicians. There’s been some evidence in hospitals that maybe quality doesn’t change too much. But the deeper layer under that is that these private equity–acquired hospitals may be selecting certain patients that are less sick, that are not going to negatively affect their metrics as much. That’s the environment that we had been reading about and starting to ask: Are we seeing that in cardiology too?
Dr. Harrington: Share with the audience what you did. You took what I would call a descriptive approach to try to understand the current landscape in cardiovascular medicine. As Ed already pointed out, a lot of the earlier data does not concern cardiology practices. My read of your paper is that you were trying to at least lay the groundwork for us to understand as a community what’s going on out there. Is that a fair interpretation?
Dr. Bartlett: Absolutely. Even that initial question of what’s happening is more challenging than it seems it might be to answer, partly because with private equity, these are private transactions. They don’t have to publicly report anything. So there’s a lot of manual work to gather these data. Our first questions were: What are these transactions? When are they happening? Where are they happening? What are the clinics that private equity is interested in? What are the community characteristics of those clinics? And what could that tell us about what’s going on?
Who Is Getting Acquired?
Dr. Harrington: Tell the audience broadly what you found. What are those clinics? And how often does this happen?
Dr. Bartlett: We looked at acquisitions between 2013 and 2023, and in that 10-year span, we found 41 acquisitions of outpatient cardiology practices, which corresponded to 342 acquisitions of clinics. The vast majority of these, pretty much 95%, occurred between 2021 and 2023. We calculated that about 3% of cardiology clinics in the US are owned by private equity. The states with the highest number of acquisitions were Florida, Texas, and Arizona, and particularly the urban areas in those states, ie, Jacksonville, Houston, Dallas. And interestingly, that mirrors what we’ve seen before in anesthesia and dermatology.
Our last question was around community characteristics, we looked at several that had a statistically significant association with private equity acquisition, and we found that private equity firms were less likely to acquire clinics in the highest poverty communities. Within the communities, we looked at the proportion of adults over 65, the proportion of racial and ethnic minorities, educational level, rurality, and didn’t find any significant associations between private equity acquisition and those characteristics.
Dr. Harrington: Thank you. Rishi, do you want to interpret why private equity was targeting certain areas?
Dr. Wadhera: Private equity goes where they can actually acquire practices. Those states, in particular, have more independent practices than, say, Massachusetts does. Then there’s the target population available in those states. Building on what Ed said earlier, why all of a sudden? Because Victoria just pointed out that the vast majority of these acquisitions happened between 2020 and 2023 and you see the surge, and I expect that surge to continue over the next several years. And the question is why?
We know with the rise in cardiometabolic risk factors at a population level, that the cardiovascular disease is only going to become more common. Cardiac procedures are very well reimbursed. There’s likely a lot of appeal in entering a specialty with a highly profitable service line. Over the past decade, federal policymakers very intentionally have created incentives to shift the delivery of cardiac procedures to nonhospital settings. We see that with the rise of ambulatory surgical centers and more cardiac procedures are being reimbursed in these types of settings. And I think that private equity firms may see this as an opportunity to maximize profits.
Victoria created this beautiful map in our study that showed how concentrated these acquisitions are. They really concentrated in specific markets. And I think that parallels what we’re seeing with health systems more broadly, this consolidation, and concentration is the ultimate goal. These different stakeholders, it’s not just private equity, have more market power, so that when they go to insurers, they can demand higher prices for procedures and services.
Dr. Harrington: It’s hard to look at the dates of 2021 or 2020 to 2023, and not wonder if there is a COVID effect. Victoria, do you think there’s a COVID effect, or is it just true, true, unrelated?
Dr. Bartlett: COVID definitely put a lot of financial pressure on providers, and particularly small independent practices. They would have felt that the most, and I certainly think is a piece of the picture but may not be all the picture.
Dr. Harrington: That’s what I would have guessed. We were all under financial pressures, but the small, independent practices didn’t have the big health system behind them to backstop things. Ed, as a former leader of the ACC, and the ACC very much works at the local level, are you hearing from the governors of these states that this is an issue, and not hearing from other states?
Dr. Fry: Certainly this activity is concentrated in the states that Victoria and Rishi described for the reasons that they outlined. This is still a very small number and probably will remain relatively small if we consider that 85% of cardiologists are employed, and the bar to exit an employment arrangement and enter into a private equity situation is pretty darn high. There’s a lot of costs associated with that. So it may have a finite cap to it, and that may be part of what buffers some of the response.
I would like to go back and address other reasons why this is happening. Particularly because of the aging population of cardiovascular patients, we’ve also seen the rise of Medicare Advantage, which is a type of value, if you consider it a type of value-based care. There are incentives built into Medicare Advantage to manage costs and to do various things so there is certainly a reward incentive. I am not wearing my hat as a representative of the ACC nor Ascension, and I will probably be a consumer of these services before I’m ever a participant, but I would say that private equity in some respects, is acting as a disruptor in this entire process. One of the positive outcomes from this is for a reevaluation of the role of clinicians in the overall delivery of care for health systems and academic medical centers. I think that can be a positive; I always try to look at the bright side of things too.
Patient and Clinician Satisfaction
Dr. Harrington: To your last comment. Ed, maybe I’ll ask you Rishi or Victoria, any insights into clinician wellness, how people feel when their practice has been bought by private equity? Are there any data out there?
Dr. Wadhera: Not that I know of. I will say that we have a study under review right now that doesn’t answer your question directly, Bob, but that looks at how private equity acquisitions of US hospitals affect the patient care experience. And what we found, using a rigorous, quasi experimental study design comparing private equity–acquired hospitals to neighboring control hospitals, is that private equity acquisition leads to a pretty marked decrease in patient care experience and satisfaction.
That’s capturing another dimension of quality that mortality and readmissions don’t necessarily reflect. It doesn’t answer your question directly, but I think an important area for future research is understanding the effects on the clinician experience as well as, most importantly, the patient experience.
Dr. Harrington: Nicely said, it seems like a good time to think about mixed qualitative methods such as focus groups, etc., coupled with the more quantitative research methods. Victoria, I suspect you talked to people in acquired practices. Any insight into whether it’s observational or rigorous data on the clinician experience?
Dr. Bartlett: Not that I have seen. I imagine it’s probably mixed because as we’ve been saying, there’s a lot of financial pressure on practices, small, independent practices, and it can become overwhelming to run them. Private equity firms offer a very attractive value proposition or can. But I think it’s a great point that should be highlighted.
Dr. Harrington: Ed, taking off your cardiovascular leadership hat, not representing any specific organization, what are the policy things that we should be thinking about?
Dr. Fry: There’s an opportunity to combine these conversations around research, collecting more data, and the advocacy issues related to that. One of the things that perhaps differentiates cardiology in this space from other specialties, or subspecialties, surgical subspecialties, is the plethora of data that we already have with well-established registry tools. We have good benchmarks. From a professional society standpoint, we have an obligation to make sure that the care that is provided in whatever environment meets the standards and is measurable, reportable, and provides a level of consumerism to patients and payers to be able to look at that. I think we have an obligation to advocate for the use of well-validated registry tools to track the data, to have objective data, to be able to demonstrate outcomes.
Interestingly, there’s an ACC/American Heart Association policy document from 2020 on professionalism and ethics in cardiology. And it calls for the obligation of the profession to make sure that in alternative sites of care, that we are achieving at least as good a result, if not better. We have to be true to that.
Dr. Harrington: I was actually a coauthor on that paper on professionalism and talking about some of the research and education issues within the academic medical centers. You’re spot on. And I love the comment about the importance of long-standing registries, whether maintained by the ACC, the Heart Association, or the Society of Thoracic Surgeons, where we can get insights into the quality issues.
We need more work done on the patient experience, the clinician experience, but I also take the positive, Ed, that this may be a disruptor that could lend itself to some positive change in other areas that need to change.
This has been a fantastic conversation on the appearance, if you will, of private equity in cardiovascular medicine and some of the observations made by colleagues at the Smith Center at the Beth Israel Lahey, with great commentary by Ed Fry on whether this is a symptom or a solution and what we should be thinking about from a broader societal perspective. I want to thank my three guests today, Victoria, Ed, and Rishi, for joining us here.
Dr. Harrington is the Stephen and Suzanne Weiss Dean of Weill Cornell Medicine and provost for medical affairs of Cornell University, as well as a former president of the American Heart Association. He disclosed ties with several companies. Dr. Bartlett is resident physician, Department of Internal Medicine, Brigham & women’s Hospital, Boston, and has disclosed no relevant financial relationships. Dr. Fry is chair, Ascension National Cardiovascular Service Line, Ascension St. Vincent Heart Center in Indianapolis, Indiana. Dr. Wadhera is associate professor, Harvard Medical School, and associate director, Richard A. and Susan F. Smith Center for Outcomes Research, Beth Israel Deaconess Medical Center, both in Boston. Dr. Wadhera disclosed ties with Abbott, ChamberCardio, CVS Health, the National Institutes of Health, American Heart Association, and the Donaghue Foundation.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Robert A. Harrington, MD: I’m continuing my series of conversations with leaders in the field of cardiovascular medicine who are working on interesting projects and making contributions in the science and policy space. We have three guests joining us today who have recently written two papers in the Journal of the American College of Cardiology. One is an original research paper dealing with the issue of private equity’s acquisition of outpatient cardiology practices. And the second is an editorial that really tries to get at why this is happening. Is it a problem? Is it a solution to a problem?
Fortunately, I have all three as guests to think about this important issue that has implications for clinical care, reimbursement, physician wellness, and clinician wellness, and it has implications regarding public policy and how we should be thinking about the practice of medicine in this country.
Dr. Victoria L. Bartlett is an internal medicine resident at Brigham and Women’s Hospital in Boston, and a research fellow in the Smith Center at the Beth Israel Lahey medical center in Boston. Dr Rishi K. Wadhera is the senior author of the paper written by Dr. Bartlett. Dr. Rishi is associate professor of medicine at Harvard Medical School, and he is the associate director of the Smith Center at Beth Israel Lahey.
Rishi K. Wadhera, MD, MPP, MPhil: Thanks for having us, Bob.
Dr. Harrington: The editorialist, Dr. Ed Fry, is the national service line leader for cardiovascular medicine, for Ascension Health. Dr. Frey is a recent past president of the American College of Cardiology (ACC).
Edward T. A. Fry, MD: Great to be here. Thanks.
What is private equity? Why the interest in medicine?
Dr. Harrington: I was intrigued by the papers and it caused me to do a deeper dive into some of the earlier works that you have referenced about this growing topic of private equity making its way into medical practice. Rishi, I’ll start with you. For the casual reader like myself, what is the business of private equity?
Dr. Wadhera: Private equity firms basically used pooled investments from multiple sources. These can be individual and institutional investors, pension funds, endowments, and they use those funds to invest in private companies that have the potential to return a profit. The goal really is to turn a profit for institutional investors over a shorter time horizon.
Dr. Harrington: How do they do that? I can understand, you buy a factory and you want to make the factory a little more efficient, and you think that perhaps, by combining some technologies, etc., that you might have in other factories, you can drive more value out of the one you just invested in in a short period of time. What’s the general business sense of how they’re going to do that in a cardiology practice? Is it all about making us more efficient?
Dr. Wadhera: Operational efficiency is the overarching theme here. One could argue that perhaps, private equity firms have the expertise to bring that kind of organizational know-how and operational efficiency to medicine. But there’s evidence that the way that private equity firms maximize their margin is maybe through mechanisms that aren’t necessarily good for patient care, such as reduced nursing staffing. When private equity acquires hospitals or practices in the same location, they have greater negotiating power at the payer table, to have higher prices for the services they deliver. There’s a lot of discussion about whether the sort of changes that private equity firms tend to implement are good or bad for patient care and also for clinicians.
Dr. Harrington: Great summary. Ed. Why is this happening in medicine? What did we do in medicine that made us ripe for investment by private equity? When you and I started out years ago, I don’t think we ever would have thought that this was in the future.
Dr. Fry: I think number one, as we know, is that medicine represents about 20% of our economy. There are huge amounts of money involved in these considerations. If players in this space can access even a small fraction of that money, it’s a lot of money and a lot of incentive for them.
In medicine in general, and then maybe more specifically, in cardiology, we’ve seen a shift away from private practice into employed practice. When people made those decisions over the past 10-15 years, there were certainly positives and risks that they took. I think for some, along the way, they realized that perhaps they gave up more than they thought in terms of control and running their own business and the opportunity to shape that themselves and be rewarded for that as they were in private practice. In cardiology, more specifically, we’ve seen this shift to the outpatient space: moving diagnostics and even therapeutics into ambulatory surgical centers and outpatient-based labs, and that is another potential source of revenue for these private equity companies.
As I wrote in the editorial, there are certainly a lot of pressures and frustrations that the day-to-day clinician feels, and maybe, this move to private equity is more of a symptom of those concerns and that this could be an opportunity to take the bull by the horns again in cardiology. We’ve evolved from a predominantly hospital-based acute care specialty into one of diagnosis, chronic disease management, and longitudinal care punctuated by diagnostics and therapeutics, which are, again, I think, attractive to private equity firms as potential sources for revenue.
Dr. Harrington: Ed, why cardiology? What’s happened over the years that has led to that appearance, if you will, of private equity and cardiovascular medicine?
Dr. Fry: Some of the earlier specialties were dermatology, ophthalmology, and gastroenterology, in particular. And interestingly, those tend to be specialties that have less chronic disease management and are more based on procedures and things like that. Within cardiology, obviously, the big driver is that our population is aging: 11,000 people turn 65 every day and become eligible for Medicare. With that, we see a rise in disease prevalence and then the rise in risk factors, obviously, with obesity and diabetes driving that, so there are more people who are going to have an illness that requires evaluation, diagnostics, and procedures. Because of that, it is a very target rich environment for private equity.
Dr. Harrington: That’s great background. Now, let’s dive into what you did, Victoria. What got you interested in the question? And give us some background on the literature that you were trying to build upon when you asked your series of questions.
Victoria L. Bartlett, MD: There’s been a lot of interest in private equity acquisitions and healthcare. A lot of the existing literature has been around hospital acquisitions and what happens there. There’s some literature, as you’ve mentioned, in outpatient practices, in certain specialties, where private equity has existed a little bit longer than in cardiology. They’ve been asking really similar questions to what we have been asking about cardiology, which is what happens when practices are acquired.
A kind of overview is that many of those studies have found increased costs to payers, to patients, and many have also found evidence of decreased quality. The evidence for the latter is honestly more difficult to figure out, but there has been evidence of decreased nursing ratios in nursing homes. There’s been evidence of changing the mix in clinics to more advanced practice providers than physicians. There’s been some evidence in hospitals that maybe quality doesn’t change too much. But the deeper layer under that is that these private equity–acquired hospitals may be selecting certain patients that are less sick, that are not going to negatively affect their metrics as much. That’s the environment that we had been reading about and starting to ask: Are we seeing that in cardiology too?
Dr. Harrington: Share with the audience what you did. You took what I would call a descriptive approach to try to understand the current landscape in cardiovascular medicine. As Ed already pointed out, a lot of the earlier data does not concern cardiology practices. My read of your paper is that you were trying to at least lay the groundwork for us to understand as a community what’s going on out there. Is that a fair interpretation?
Dr. Bartlett: Absolutely. Even that initial question of what’s happening is more challenging than it seems it might be to answer, partly because with private equity, these are private transactions. They don’t have to publicly report anything. So there’s a lot of manual work to gather these data. Our first questions were: What are these transactions? When are they happening? Where are they happening? What are the clinics that private equity is interested in? What are the community characteristics of those clinics? And what could that tell us about what’s going on?
Who Is Getting Acquired?
Dr. Harrington: Tell the audience broadly what you found. What are those clinics? And how often does this happen?
Dr. Bartlett: We looked at acquisitions between 2013 and 2023, and in that 10-year span, we found 41 acquisitions of outpatient cardiology practices, which corresponded to 342 acquisitions of clinics. The vast majority of these, pretty much 95%, occurred between 2021 and 2023. We calculated that about 3% of cardiology clinics in the US are owned by private equity. The states with the highest number of acquisitions were Florida, Texas, and Arizona, and particularly the urban areas in those states, ie, Jacksonville, Houston, Dallas. And interestingly, that mirrors what we’ve seen before in anesthesia and dermatology.
Our last question was around community characteristics, we looked at several that had a statistically significant association with private equity acquisition, and we found that private equity firms were less likely to acquire clinics in the highest poverty communities. Within the communities, we looked at the proportion of adults over 65, the proportion of racial and ethnic minorities, educational level, rurality, and didn’t find any significant associations between private equity acquisition and those characteristics.
Dr. Harrington: Thank you. Rishi, do you want to interpret why private equity was targeting certain areas?
Dr. Wadhera: Private equity goes where they can actually acquire practices. Those states, in particular, have more independent practices than, say, Massachusetts does. Then there’s the target population available in those states. Building on what Ed said earlier, why all of a sudden? Because Victoria just pointed out that the vast majority of these acquisitions happened between 2020 and 2023 and you see the surge, and I expect that surge to continue over the next several years. And the question is why?
We know with the rise in cardiometabolic risk factors at a population level, that the cardiovascular disease is only going to become more common. Cardiac procedures are very well reimbursed. There’s likely a lot of appeal in entering a specialty with a highly profitable service line. Over the past decade, federal policymakers very intentionally have created incentives to shift the delivery of cardiac procedures to nonhospital settings. We see that with the rise of ambulatory surgical centers and more cardiac procedures are being reimbursed in these types of settings. And I think that private equity firms may see this as an opportunity to maximize profits.
Victoria created this beautiful map in our study that showed how concentrated these acquisitions are. They really concentrated in specific markets. And I think that parallels what we’re seeing with health systems more broadly, this consolidation, and concentration is the ultimate goal. These different stakeholders, it’s not just private equity, have more market power, so that when they go to insurers, they can demand higher prices for procedures and services.
Dr. Harrington: It’s hard to look at the dates of 2021 or 2020 to 2023, and not wonder if there is a COVID effect. Victoria, do you think there’s a COVID effect, or is it just true, true, unrelated?
Dr. Bartlett: COVID definitely put a lot of financial pressure on providers, and particularly small independent practices. They would have felt that the most, and I certainly think is a piece of the picture but may not be all the picture.
Dr. Harrington: That’s what I would have guessed. We were all under financial pressures, but the small, independent practices didn’t have the big health system behind them to backstop things. Ed, as a former leader of the ACC, and the ACC very much works at the local level, are you hearing from the governors of these states that this is an issue, and not hearing from other states?
Dr. Fry: Certainly this activity is concentrated in the states that Victoria and Rishi described for the reasons that they outlined. This is still a very small number and probably will remain relatively small if we consider that 85% of cardiologists are employed, and the bar to exit an employment arrangement and enter into a private equity situation is pretty darn high. There’s a lot of costs associated with that. So it may have a finite cap to it, and that may be part of what buffers some of the response.
I would like to go back and address other reasons why this is happening. Particularly because of the aging population of cardiovascular patients, we’ve also seen the rise of Medicare Advantage, which is a type of value, if you consider it a type of value-based care. There are incentives built into Medicare Advantage to manage costs and to do various things so there is certainly a reward incentive. I am not wearing my hat as a representative of the ACC nor Ascension, and I will probably be a consumer of these services before I’m ever a participant, but I would say that private equity in some respects, is acting as a disruptor in this entire process. One of the positive outcomes from this is for a reevaluation of the role of clinicians in the overall delivery of care for health systems and academic medical centers. I think that can be a positive; I always try to look at the bright side of things too.
Patient and Clinician Satisfaction
Dr. Harrington: To your last comment. Ed, maybe I’ll ask you Rishi or Victoria, any insights into clinician wellness, how people feel when their practice has been bought by private equity? Are there any data out there?
Dr. Wadhera: Not that I know of. I will say that we have a study under review right now that doesn’t answer your question directly, Bob, but that looks at how private equity acquisitions of US hospitals affect the patient care experience. And what we found, using a rigorous, quasi experimental study design comparing private equity–acquired hospitals to neighboring control hospitals, is that private equity acquisition leads to a pretty marked decrease in patient care experience and satisfaction.
That’s capturing another dimension of quality that mortality and readmissions don’t necessarily reflect. It doesn’t answer your question directly, but I think an important area for future research is understanding the effects on the clinician experience as well as, most importantly, the patient experience.
Dr. Harrington: Nicely said, it seems like a good time to think about mixed qualitative methods such as focus groups, etc., coupled with the more quantitative research methods. Victoria, I suspect you talked to people in acquired practices. Any insight into whether it’s observational or rigorous data on the clinician experience?
Dr. Bartlett: Not that I have seen. I imagine it’s probably mixed because as we’ve been saying, there’s a lot of financial pressure on practices, small, independent practices, and it can become overwhelming to run them. Private equity firms offer a very attractive value proposition or can. But I think it’s a great point that should be highlighted.
Dr. Harrington: Ed, taking off your cardiovascular leadership hat, not representing any specific organization, what are the policy things that we should be thinking about?
Dr. Fry: There’s an opportunity to combine these conversations around research, collecting more data, and the advocacy issues related to that. One of the things that perhaps differentiates cardiology in this space from other specialties, or subspecialties, surgical subspecialties, is the plethora of data that we already have with well-established registry tools. We have good benchmarks. From a professional society standpoint, we have an obligation to make sure that the care that is provided in whatever environment meets the standards and is measurable, reportable, and provides a level of consumerism to patients and payers to be able to look at that. I think we have an obligation to advocate for the use of well-validated registry tools to track the data, to have objective data, to be able to demonstrate outcomes.
Interestingly, there’s an ACC/American Heart Association policy document from 2020 on professionalism and ethics in cardiology. And it calls for the obligation of the profession to make sure that in alternative sites of care, that we are achieving at least as good a result, if not better. We have to be true to that.
Dr. Harrington: I was actually a coauthor on that paper on professionalism and talking about some of the research and education issues within the academic medical centers. You’re spot on. And I love the comment about the importance of long-standing registries, whether maintained by the ACC, the Heart Association, or the Society of Thoracic Surgeons, where we can get insights into the quality issues.
We need more work done on the patient experience, the clinician experience, but I also take the positive, Ed, that this may be a disruptor that could lend itself to some positive change in other areas that need to change.
This has been a fantastic conversation on the appearance, if you will, of private equity in cardiovascular medicine and some of the observations made by colleagues at the Smith Center at the Beth Israel Lahey, with great commentary by Ed Fry on whether this is a symptom or a solution and what we should be thinking about from a broader societal perspective. I want to thank my three guests today, Victoria, Ed, and Rishi, for joining us here.
Dr. Harrington is the Stephen and Suzanne Weiss Dean of Weill Cornell Medicine and provost for medical affairs of Cornell University, as well as a former president of the American Heart Association. He disclosed ties with several companies. Dr. Bartlett is resident physician, Department of Internal Medicine, Brigham & women’s Hospital, Boston, and has disclosed no relevant financial relationships. Dr. Fry is chair, Ascension National Cardiovascular Service Line, Ascension St. Vincent Heart Center in Indianapolis, Indiana. Dr. Wadhera is associate professor, Harvard Medical School, and associate director, Richard A. and Susan F. Smith Center for Outcomes Research, Beth Israel Deaconess Medical Center, both in Boston. Dr. Wadhera disclosed ties with Abbott, ChamberCardio, CVS Health, the National Institutes of Health, American Heart Association, and the Donaghue Foundation.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Robert A. Harrington, MD: I’m continuing my series of conversations with leaders in the field of cardiovascular medicine who are working on interesting projects and making contributions in the science and policy space. We have three guests joining us today who have recently written two papers in the Journal of the American College of Cardiology. One is an original research paper dealing with the issue of private equity’s acquisition of outpatient cardiology practices. And the second is an editorial that really tries to get at why this is happening. Is it a problem? Is it a solution to a problem?
Fortunately, I have all three as guests to think about this important issue that has implications for clinical care, reimbursement, physician wellness, and clinician wellness, and it has implications regarding public policy and how we should be thinking about the practice of medicine in this country.
Dr. Victoria L. Bartlett is an internal medicine resident at Brigham and Women’s Hospital in Boston, and a research fellow in the Smith Center at the Beth Israel Lahey medical center in Boston. Dr Rishi K. Wadhera is the senior author of the paper written by Dr. Bartlett. Dr. Rishi is associate professor of medicine at Harvard Medical School, and he is the associate director of the Smith Center at Beth Israel Lahey.
Rishi K. Wadhera, MD, MPP, MPhil: Thanks for having us, Bob.
Dr. Harrington: The editorialist, Dr. Ed Fry, is the national service line leader for cardiovascular medicine, for Ascension Health. Dr. Frey is a recent past president of the American College of Cardiology (ACC).
Edward T. A. Fry, MD: Great to be here. Thanks.
What is private equity? Why the interest in medicine?
Dr. Harrington: I was intrigued by the papers and it caused me to do a deeper dive into some of the earlier works that you have referenced about this growing topic of private equity making its way into medical practice. Rishi, I’ll start with you. For the casual reader like myself, what is the business of private equity?
Dr. Wadhera: Private equity firms basically used pooled investments from multiple sources. These can be individual and institutional investors, pension funds, endowments, and they use those funds to invest in private companies that have the potential to return a profit. The goal really is to turn a profit for institutional investors over a shorter time horizon.
Dr. Harrington: How do they do that? I can understand, you buy a factory and you want to make the factory a little more efficient, and you think that perhaps, by combining some technologies, etc., that you might have in other factories, you can drive more value out of the one you just invested in in a short period of time. What’s the general business sense of how they’re going to do that in a cardiology practice? Is it all about making us more efficient?
Dr. Wadhera: Operational efficiency is the overarching theme here. One could argue that perhaps, private equity firms have the expertise to bring that kind of organizational know-how and operational efficiency to medicine. But there’s evidence that the way that private equity firms maximize their margin is maybe through mechanisms that aren’t necessarily good for patient care, such as reduced nursing staffing. When private equity acquires hospitals or practices in the same location, they have greater negotiating power at the payer table, to have higher prices for the services they deliver. There’s a lot of discussion about whether the sort of changes that private equity firms tend to implement are good or bad for patient care and also for clinicians.
Dr. Harrington: Great summary. Ed. Why is this happening in medicine? What did we do in medicine that made us ripe for investment by private equity? When you and I started out years ago, I don’t think we ever would have thought that this was in the future.
Dr. Fry: I think number one, as we know, is that medicine represents about 20% of our economy. There are huge amounts of money involved in these considerations. If players in this space can access even a small fraction of that money, it’s a lot of money and a lot of incentive for them.
In medicine in general, and then maybe more specifically, in cardiology, we’ve seen a shift away from private practice into employed practice. When people made those decisions over the past 10-15 years, there were certainly positives and risks that they took. I think for some, along the way, they realized that perhaps they gave up more than they thought in terms of control and running their own business and the opportunity to shape that themselves and be rewarded for that as they were in private practice. In cardiology, more specifically, we’ve seen this shift to the outpatient space: moving diagnostics and even therapeutics into ambulatory surgical centers and outpatient-based labs, and that is another potential source of revenue for these private equity companies.
As I wrote in the editorial, there are certainly a lot of pressures and frustrations that the day-to-day clinician feels, and maybe, this move to private equity is more of a symptom of those concerns and that this could be an opportunity to take the bull by the horns again in cardiology. We’ve evolved from a predominantly hospital-based acute care specialty into one of diagnosis, chronic disease management, and longitudinal care punctuated by diagnostics and therapeutics, which are, again, I think, attractive to private equity firms as potential sources for revenue.
Dr. Harrington: Ed, why cardiology? What’s happened over the years that has led to that appearance, if you will, of private equity and cardiovascular medicine?
Dr. Fry: Some of the earlier specialties were dermatology, ophthalmology, and gastroenterology, in particular. And interestingly, those tend to be specialties that have less chronic disease management and are more based on procedures and things like that. Within cardiology, obviously, the big driver is that our population is aging: 11,000 people turn 65 every day and become eligible for Medicare. With that, we see a rise in disease prevalence and then the rise in risk factors, obviously, with obesity and diabetes driving that, so there are more people who are going to have an illness that requires evaluation, diagnostics, and procedures. Because of that, it is a very target rich environment for private equity.
Dr. Harrington: That’s great background. Now, let’s dive into what you did, Victoria. What got you interested in the question? And give us some background on the literature that you were trying to build upon when you asked your series of questions.
Victoria L. Bartlett, MD: There’s been a lot of interest in private equity acquisitions and healthcare. A lot of the existing literature has been around hospital acquisitions and what happens there. There’s some literature, as you’ve mentioned, in outpatient practices, in certain specialties, where private equity has existed a little bit longer than in cardiology. They’ve been asking really similar questions to what we have been asking about cardiology, which is what happens when practices are acquired.
A kind of overview is that many of those studies have found increased costs to payers, to patients, and many have also found evidence of decreased quality. The evidence for the latter is honestly more difficult to figure out, but there has been evidence of decreased nursing ratios in nursing homes. There’s been evidence of changing the mix in clinics to more advanced practice providers than physicians. There’s been some evidence in hospitals that maybe quality doesn’t change too much. But the deeper layer under that is that these private equity–acquired hospitals may be selecting certain patients that are less sick, that are not going to negatively affect their metrics as much. That’s the environment that we had been reading about and starting to ask: Are we seeing that in cardiology too?
Dr. Harrington: Share with the audience what you did. You took what I would call a descriptive approach to try to understand the current landscape in cardiovascular medicine. As Ed already pointed out, a lot of the earlier data does not concern cardiology practices. My read of your paper is that you were trying to at least lay the groundwork for us to understand as a community what’s going on out there. Is that a fair interpretation?
Dr. Bartlett: Absolutely. Even that initial question of what’s happening is more challenging than it seems it might be to answer, partly because with private equity, these are private transactions. They don’t have to publicly report anything. So there’s a lot of manual work to gather these data. Our first questions were: What are these transactions? When are they happening? Where are they happening? What are the clinics that private equity is interested in? What are the community characteristics of those clinics? And what could that tell us about what’s going on?
Who Is Getting Acquired?
Dr. Harrington: Tell the audience broadly what you found. What are those clinics? And how often does this happen?
Dr. Bartlett: We looked at acquisitions between 2013 and 2023, and in that 10-year span, we found 41 acquisitions of outpatient cardiology practices, which corresponded to 342 acquisitions of clinics. The vast majority of these, pretty much 95%, occurred between 2021 and 2023. We calculated that about 3% of cardiology clinics in the US are owned by private equity. The states with the highest number of acquisitions were Florida, Texas, and Arizona, and particularly the urban areas in those states, ie, Jacksonville, Houston, Dallas. And interestingly, that mirrors what we’ve seen before in anesthesia and dermatology.
Our last question was around community characteristics, we looked at several that had a statistically significant association with private equity acquisition, and we found that private equity firms were less likely to acquire clinics in the highest poverty communities. Within the communities, we looked at the proportion of adults over 65, the proportion of racial and ethnic minorities, educational level, rurality, and didn’t find any significant associations between private equity acquisition and those characteristics.
Dr. Harrington: Thank you. Rishi, do you want to interpret why private equity was targeting certain areas?
Dr. Wadhera: Private equity goes where they can actually acquire practices. Those states, in particular, have more independent practices than, say, Massachusetts does. Then there’s the target population available in those states. Building on what Ed said earlier, why all of a sudden? Because Victoria just pointed out that the vast majority of these acquisitions happened between 2020 and 2023 and you see the surge, and I expect that surge to continue over the next several years. And the question is why?
We know with the rise in cardiometabolic risk factors at a population level, that the cardiovascular disease is only going to become more common. Cardiac procedures are very well reimbursed. There’s likely a lot of appeal in entering a specialty with a highly profitable service line. Over the past decade, federal policymakers very intentionally have created incentives to shift the delivery of cardiac procedures to nonhospital settings. We see that with the rise of ambulatory surgical centers and more cardiac procedures are being reimbursed in these types of settings. And I think that private equity firms may see this as an opportunity to maximize profits.
Victoria created this beautiful map in our study that showed how concentrated these acquisitions are. They really concentrated in specific markets. And I think that parallels what we’re seeing with health systems more broadly, this consolidation, and concentration is the ultimate goal. These different stakeholders, it’s not just private equity, have more market power, so that when they go to insurers, they can demand higher prices for procedures and services.
Dr. Harrington: It’s hard to look at the dates of 2021 or 2020 to 2023, and not wonder if there is a COVID effect. Victoria, do you think there’s a COVID effect, or is it just true, true, unrelated?
Dr. Bartlett: COVID definitely put a lot of financial pressure on providers, and particularly small independent practices. They would have felt that the most, and I certainly think is a piece of the picture but may not be all the picture.
Dr. Harrington: That’s what I would have guessed. We were all under financial pressures, but the small, independent practices didn’t have the big health system behind them to backstop things. Ed, as a former leader of the ACC, and the ACC very much works at the local level, are you hearing from the governors of these states that this is an issue, and not hearing from other states?
Dr. Fry: Certainly this activity is concentrated in the states that Victoria and Rishi described for the reasons that they outlined. This is still a very small number and probably will remain relatively small if we consider that 85% of cardiologists are employed, and the bar to exit an employment arrangement and enter into a private equity situation is pretty darn high. There’s a lot of costs associated with that. So it may have a finite cap to it, and that may be part of what buffers some of the response.
I would like to go back and address other reasons why this is happening. Particularly because of the aging population of cardiovascular patients, we’ve also seen the rise of Medicare Advantage, which is a type of value, if you consider it a type of value-based care. There are incentives built into Medicare Advantage to manage costs and to do various things so there is certainly a reward incentive. I am not wearing my hat as a representative of the ACC nor Ascension, and I will probably be a consumer of these services before I’m ever a participant, but I would say that private equity in some respects, is acting as a disruptor in this entire process. One of the positive outcomes from this is for a reevaluation of the role of clinicians in the overall delivery of care for health systems and academic medical centers. I think that can be a positive; I always try to look at the bright side of things too.
Patient and Clinician Satisfaction
Dr. Harrington: To your last comment. Ed, maybe I’ll ask you Rishi or Victoria, any insights into clinician wellness, how people feel when their practice has been bought by private equity? Are there any data out there?
Dr. Wadhera: Not that I know of. I will say that we have a study under review right now that doesn’t answer your question directly, Bob, but that looks at how private equity acquisitions of US hospitals affect the patient care experience. And what we found, using a rigorous, quasi experimental study design comparing private equity–acquired hospitals to neighboring control hospitals, is that private equity acquisition leads to a pretty marked decrease in patient care experience and satisfaction.
That’s capturing another dimension of quality that mortality and readmissions don’t necessarily reflect. It doesn’t answer your question directly, but I think an important area for future research is understanding the effects on the clinician experience as well as, most importantly, the patient experience.
Dr. Harrington: Nicely said, it seems like a good time to think about mixed qualitative methods such as focus groups, etc., coupled with the more quantitative research methods. Victoria, I suspect you talked to people in acquired practices. Any insight into whether it’s observational or rigorous data on the clinician experience?
Dr. Bartlett: Not that I have seen. I imagine it’s probably mixed because as we’ve been saying, there’s a lot of financial pressure on practices, small, independent practices, and it can become overwhelming to run them. Private equity firms offer a very attractive value proposition or can. But I think it’s a great point that should be highlighted.
Dr. Harrington: Ed, taking off your cardiovascular leadership hat, not representing any specific organization, what are the policy things that we should be thinking about?
Dr. Fry: There’s an opportunity to combine these conversations around research, collecting more data, and the advocacy issues related to that. One of the things that perhaps differentiates cardiology in this space from other specialties, or subspecialties, surgical subspecialties, is the plethora of data that we already have with well-established registry tools. We have good benchmarks. From a professional society standpoint, we have an obligation to make sure that the care that is provided in whatever environment meets the standards and is measurable, reportable, and provides a level of consumerism to patients and payers to be able to look at that. I think we have an obligation to advocate for the use of well-validated registry tools to track the data, to have objective data, to be able to demonstrate outcomes.
Interestingly, there’s an ACC/American Heart Association policy document from 2020 on professionalism and ethics in cardiology. And it calls for the obligation of the profession to make sure that in alternative sites of care, that we are achieving at least as good a result, if not better. We have to be true to that.
Dr. Harrington: I was actually a coauthor on that paper on professionalism and talking about some of the research and education issues within the academic medical centers. You’re spot on. And I love the comment about the importance of long-standing registries, whether maintained by the ACC, the Heart Association, or the Society of Thoracic Surgeons, where we can get insights into the quality issues.
We need more work done on the patient experience, the clinician experience, but I also take the positive, Ed, that this may be a disruptor that could lend itself to some positive change in other areas that need to change.
This has been a fantastic conversation on the appearance, if you will, of private equity in cardiovascular medicine and some of the observations made by colleagues at the Smith Center at the Beth Israel Lahey, with great commentary by Ed Fry on whether this is a symptom or a solution and what we should be thinking about from a broader societal perspective. I want to thank my three guests today, Victoria, Ed, and Rishi, for joining us here.
Dr. Harrington is the Stephen and Suzanne Weiss Dean of Weill Cornell Medicine and provost for medical affairs of Cornell University, as well as a former president of the American Heart Association. He disclosed ties with several companies. Dr. Bartlett is resident physician, Department of Internal Medicine, Brigham & women’s Hospital, Boston, and has disclosed no relevant financial relationships. Dr. Fry is chair, Ascension National Cardiovascular Service Line, Ascension St. Vincent Heart Center in Indianapolis, Indiana. Dr. Wadhera is associate professor, Harvard Medical School, and associate director, Richard A. and Susan F. Smith Center for Outcomes Research, Beth Israel Deaconess Medical Center, both in Boston. Dr. Wadhera disclosed ties with Abbott, ChamberCardio, CVS Health, the National Institutes of Health, American Heart Association, and the Donaghue Foundation.
A version of this article first appeared on Medscape.com.
Storybooks Can Help Children Deal with Skin Conditions
TORONTO —
So far, “the study demonstrates that these books have value to patients and families,” one of the study authors, Sonia Havele, MD, a pediatrician and dermatology resident at Children’s Mercy Hospital Kansas City, Kansas City, Missouri, said in an interview.
“There are tools to help kids cope with their skin conditions, but we’re underutilizing them,” she added. “And part of the reason we’re underutilizing storybooks is that we just don’t know what’s out there.” For the study, the researchers received funding to purchase 18 “creative and thoughtful” storybooks related to pediatric skin conditions, reviewed by at least two pediatric dermatologists before being selected, which are just a sample of related books that are available.
The study results were presented as a poster at the annual meeting of the Society for Pediatric Dermatology.
Children with visible skin conditions, which can include port-wine stains, capillary malformations, and congenital moles, may be subjected to teasing or bullying at school, and the conditions can also affect their quality of life.
Beauty and the Birthmark
The books include one titled “Beauty with a Birthmark” and another, “My Hair Went on Vacation.” An illustrated book, “Just Ask: Be Different, Be Brave, Be You,” by US Supreme Court Justice Sonia Sotomayor, offers tips on how to answer common questions about someone’s appearance.
Dr. Havele said that Justice Sotomayor’s book “empowers kids, their siblings, their classmates ... to ask questions, and it teaches patients not to be afraid of those questions, and to really lean into educating their peers, and their family members.”
“Kids are really just curious,” she added. “They’ll make comments like: ‘Hey, what’s that spot on your face?’ Or, they’ll ask about vitiligo because they’ve never seen somebody with it before.”
To evaluate the psychosocial impact of these types of books for children with visible skin conditions, Dr. Havele and colleagues designed a study that includes patients aged 2-12 years dealing with issues related to self-esteem, acceptance, coping, or bullying. Parents are provided with a relevant storybook to read at home with their child in a “safe and comfortable space” and “at their own pace and their own time,” said Dr. Havele.
Inside the book is a QR code to access the validated Children’s Dermatology Life Quality Index (CDLQI). Families complete the survey at baseline and provide feedback after reading the book. Researchers collect information about demographics, age, gender, and skin conditions, which included atopic dermatitis, alopecia areata, vitiligo, hemangioma, and port-wine stain.
The response rate so far is 34%, and close to 80 parents have completed the survey with their child, Dr. Havele said.
At baseline, many of the children were either moderately or severely affected in terms of their quality of life (45% scored ≥ 6 on the CDLQI).
After reading the book, about 80% of parents reported it had a positive impact, and about 20% said it had a somewhat positive impact on their child’s self-image or confidence. Almost 80% agreed, and the remainder somewhat agreed it encouraged their child to embrace differences.
Most respondents also said the book helped the parent and child cope with the child’s condition. “So really, it was overall a positive response,” said Dr. Havele. “We are able to demonstrate that these books have value in a more scientific or objective way.”
This may not be surprising. Dr. Havele referred to more formal bibliotherapy (book therapy), which has been studied in other pediatric populations, including patients with cancer and those who have experienced trauma.
Awesome Space
Pediatric dermatologists are perfectly positioned to play a role in improving the lives of their patients with skin issues. “We see the impact of visible skin disease on children all the time,” said Dr. Havele. “The dermatology visit is an awesome space and opportunity to introduce these books to families and potentially help them talk about the skin condition with their child.”
In addition to prescribing therapies, “we’re also with these kids through an emotional journey, and I think giving them tools for that emotional journey is very helpful,” she added.
Such books would have been a great help to Dr. Havele herself. Growing up, she had severe atopic dermatitis covering much of her body. “Having such a resource would have helped me better cope with my reality of being different than everyone else.”
She hopes a database will be established to house these resources so other providers can refer patients to the list of books. Other books include “The Itchy-saurus: The Dino with an itch that can’t be scratched,” “Hair in My Brush,” and “I am Unique!”
Dr. Havele had no relevant disclosures.
A version of this article first appeared on Medscape.com.
TORONTO —
So far, “the study demonstrates that these books have value to patients and families,” one of the study authors, Sonia Havele, MD, a pediatrician and dermatology resident at Children’s Mercy Hospital Kansas City, Kansas City, Missouri, said in an interview.
“There are tools to help kids cope with their skin conditions, but we’re underutilizing them,” she added. “And part of the reason we’re underutilizing storybooks is that we just don’t know what’s out there.” For the study, the researchers received funding to purchase 18 “creative and thoughtful” storybooks related to pediatric skin conditions, reviewed by at least two pediatric dermatologists before being selected, which are just a sample of related books that are available.
The study results were presented as a poster at the annual meeting of the Society for Pediatric Dermatology.
Children with visible skin conditions, which can include port-wine stains, capillary malformations, and congenital moles, may be subjected to teasing or bullying at school, and the conditions can also affect their quality of life.
Beauty and the Birthmark
The books include one titled “Beauty with a Birthmark” and another, “My Hair Went on Vacation.” An illustrated book, “Just Ask: Be Different, Be Brave, Be You,” by US Supreme Court Justice Sonia Sotomayor, offers tips on how to answer common questions about someone’s appearance.
Dr. Havele said that Justice Sotomayor’s book “empowers kids, their siblings, their classmates ... to ask questions, and it teaches patients not to be afraid of those questions, and to really lean into educating their peers, and their family members.”
“Kids are really just curious,” she added. “They’ll make comments like: ‘Hey, what’s that spot on your face?’ Or, they’ll ask about vitiligo because they’ve never seen somebody with it before.”
To evaluate the psychosocial impact of these types of books for children with visible skin conditions, Dr. Havele and colleagues designed a study that includes patients aged 2-12 years dealing with issues related to self-esteem, acceptance, coping, or bullying. Parents are provided with a relevant storybook to read at home with their child in a “safe and comfortable space” and “at their own pace and their own time,” said Dr. Havele.
Inside the book is a QR code to access the validated Children’s Dermatology Life Quality Index (CDLQI). Families complete the survey at baseline and provide feedback after reading the book. Researchers collect information about demographics, age, gender, and skin conditions, which included atopic dermatitis, alopecia areata, vitiligo, hemangioma, and port-wine stain.
The response rate so far is 34%, and close to 80 parents have completed the survey with their child, Dr. Havele said.
At baseline, many of the children were either moderately or severely affected in terms of their quality of life (45% scored ≥ 6 on the CDLQI).
After reading the book, about 80% of parents reported it had a positive impact, and about 20% said it had a somewhat positive impact on their child’s self-image or confidence. Almost 80% agreed, and the remainder somewhat agreed it encouraged their child to embrace differences.
Most respondents also said the book helped the parent and child cope with the child’s condition. “So really, it was overall a positive response,” said Dr. Havele. “We are able to demonstrate that these books have value in a more scientific or objective way.”
This may not be surprising. Dr. Havele referred to more formal bibliotherapy (book therapy), which has been studied in other pediatric populations, including patients with cancer and those who have experienced trauma.
Awesome Space
Pediatric dermatologists are perfectly positioned to play a role in improving the lives of their patients with skin issues. “We see the impact of visible skin disease on children all the time,” said Dr. Havele. “The dermatology visit is an awesome space and opportunity to introduce these books to families and potentially help them talk about the skin condition with their child.”
In addition to prescribing therapies, “we’re also with these kids through an emotional journey, and I think giving them tools for that emotional journey is very helpful,” she added.
Such books would have been a great help to Dr. Havele herself. Growing up, she had severe atopic dermatitis covering much of her body. “Having such a resource would have helped me better cope with my reality of being different than everyone else.”
She hopes a database will be established to house these resources so other providers can refer patients to the list of books. Other books include “The Itchy-saurus: The Dino with an itch that can’t be scratched,” “Hair in My Brush,” and “I am Unique!”
Dr. Havele had no relevant disclosures.
A version of this article first appeared on Medscape.com.
TORONTO —
So far, “the study demonstrates that these books have value to patients and families,” one of the study authors, Sonia Havele, MD, a pediatrician and dermatology resident at Children’s Mercy Hospital Kansas City, Kansas City, Missouri, said in an interview.
“There are tools to help kids cope with their skin conditions, but we’re underutilizing them,” she added. “And part of the reason we’re underutilizing storybooks is that we just don’t know what’s out there.” For the study, the researchers received funding to purchase 18 “creative and thoughtful” storybooks related to pediatric skin conditions, reviewed by at least two pediatric dermatologists before being selected, which are just a sample of related books that are available.
The study results were presented as a poster at the annual meeting of the Society for Pediatric Dermatology.
Children with visible skin conditions, which can include port-wine stains, capillary malformations, and congenital moles, may be subjected to teasing or bullying at school, and the conditions can also affect their quality of life.
Beauty and the Birthmark
The books include one titled “Beauty with a Birthmark” and another, “My Hair Went on Vacation.” An illustrated book, “Just Ask: Be Different, Be Brave, Be You,” by US Supreme Court Justice Sonia Sotomayor, offers tips on how to answer common questions about someone’s appearance.
Dr. Havele said that Justice Sotomayor’s book “empowers kids, their siblings, their classmates ... to ask questions, and it teaches patients not to be afraid of those questions, and to really lean into educating their peers, and their family members.”
“Kids are really just curious,” she added. “They’ll make comments like: ‘Hey, what’s that spot on your face?’ Or, they’ll ask about vitiligo because they’ve never seen somebody with it before.”
To evaluate the psychosocial impact of these types of books for children with visible skin conditions, Dr. Havele and colleagues designed a study that includes patients aged 2-12 years dealing with issues related to self-esteem, acceptance, coping, or bullying. Parents are provided with a relevant storybook to read at home with their child in a “safe and comfortable space” and “at their own pace and their own time,” said Dr. Havele.
Inside the book is a QR code to access the validated Children’s Dermatology Life Quality Index (CDLQI). Families complete the survey at baseline and provide feedback after reading the book. Researchers collect information about demographics, age, gender, and skin conditions, which included atopic dermatitis, alopecia areata, vitiligo, hemangioma, and port-wine stain.
The response rate so far is 34%, and close to 80 parents have completed the survey with their child, Dr. Havele said.
At baseline, many of the children were either moderately or severely affected in terms of their quality of life (45% scored ≥ 6 on the CDLQI).
After reading the book, about 80% of parents reported it had a positive impact, and about 20% said it had a somewhat positive impact on their child’s self-image or confidence. Almost 80% agreed, and the remainder somewhat agreed it encouraged their child to embrace differences.
Most respondents also said the book helped the parent and child cope with the child’s condition. “So really, it was overall a positive response,” said Dr. Havele. “We are able to demonstrate that these books have value in a more scientific or objective way.”
This may not be surprising. Dr. Havele referred to more formal bibliotherapy (book therapy), which has been studied in other pediatric populations, including patients with cancer and those who have experienced trauma.
Awesome Space
Pediatric dermatologists are perfectly positioned to play a role in improving the lives of their patients with skin issues. “We see the impact of visible skin disease on children all the time,” said Dr. Havele. “The dermatology visit is an awesome space and opportunity to introduce these books to families and potentially help them talk about the skin condition with their child.”
In addition to prescribing therapies, “we’re also with these kids through an emotional journey, and I think giving them tools for that emotional journey is very helpful,” she added.
Such books would have been a great help to Dr. Havele herself. Growing up, she had severe atopic dermatitis covering much of her body. “Having such a resource would have helped me better cope with my reality of being different than everyone else.”
She hopes a database will be established to house these resources so other providers can refer patients to the list of books. Other books include “The Itchy-saurus: The Dino with an itch that can’t be scratched,” “Hair in My Brush,” and “I am Unique!”
Dr. Havele had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM SPD 2024
Few Severe Toxicities After SBRT in Oligometastatic Cancer
TOPLINE:
according to a large real-world analysis.
METHODOLOGY:
- Advances in cancer imaging have helped identify more patients with oligometastatic disease. Although the standard treatment approach typically involves systemic therapy such as chemotherapy and immunotherapy, SBRT has increasingly become an option for these patients. However, the toxicities associated with SBRT remain less clear.
- OligoCare, a European, prospective, registry-based, single-arm observational study, aims to provide real-world outcomes among patients with oligometastatic cancer who received SBRT. In this analysis, the researchers evaluated early toxicities among 1468 patients with different primary cancers — non–small cell lung cancer (NSCLC; 19.7%), colorectal cancer (20%), breast cancer (15.5%), and prostate cancer (44.8%).
- The primary outcome was acute toxicities, including new malignancies and deaths, within 6 months of initiating SBRT.
- Overall, 527 (35.9%) patients received concomitant systemic treatment and 828 (56%) had de novo oligometastatic disease.
TAKEAWAY:
- Overall, though, only eight patients (0.5%) experienced acute SBRT-related toxicity of grade 3 and above within 6 months; two events, however, were fatal (pneumonitis and cerebral hemorrhage), and both occurred in patients with NSCLC.
- The other six grade 3 events included one instance of each of the following: empyema, pneumonia, radiation pneumonitis, radiation skin injury, decreased appetite, and bone pain. Two of these events occurred in patients with NSCLC, two in patients with breast cancer, one in patients with colorectal cancer, and one in patients with prostate cancer.
- New primary malignancies were reported in 13 (0.9%) patients, which included bladder cancer (n = 3), nonmelanoma skin cancer (n = 3), and leukemia (n = 1).
- Overall, 43 (2.9%) patients died within 6 months, most from their primary cancer (58.1%).
IN PRACTICE:
Low rates of early acute toxicities reported in this real-world study help confirm the safety of SBRT in the treatment of oligometastases, the authors concluded. However, “some anatomical sites might be associated with an increased risk of even severe or fatal toxicities.”
SOURCE:
The study, led by Filippo Alongi, Advanced Radiation Oncology Department, IRCCS Sacro Cuore Don Calabria Hospital, Cancer Care Center, Negrar di Valpolicella, Italy, and University of Brescia, also in Italy, was published online in Radiotherapy & Oncology .
LIMITATIONS:
Some limitations of the study include the nonrandomized design and potential variability in patient selection criteria, treatment doses, and schedules.
DISCLOSURES:
The study did not receive any funding support. Two authors declared receiving speaker or lecture honoraria or consultation fees from various sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
according to a large real-world analysis.
METHODOLOGY:
- Advances in cancer imaging have helped identify more patients with oligometastatic disease. Although the standard treatment approach typically involves systemic therapy such as chemotherapy and immunotherapy, SBRT has increasingly become an option for these patients. However, the toxicities associated with SBRT remain less clear.
- OligoCare, a European, prospective, registry-based, single-arm observational study, aims to provide real-world outcomes among patients with oligometastatic cancer who received SBRT. In this analysis, the researchers evaluated early toxicities among 1468 patients with different primary cancers — non–small cell lung cancer (NSCLC; 19.7%), colorectal cancer (20%), breast cancer (15.5%), and prostate cancer (44.8%).
- The primary outcome was acute toxicities, including new malignancies and deaths, within 6 months of initiating SBRT.
- Overall, 527 (35.9%) patients received concomitant systemic treatment and 828 (56%) had de novo oligometastatic disease.
TAKEAWAY:
- Overall, though, only eight patients (0.5%) experienced acute SBRT-related toxicity of grade 3 and above within 6 months; two events, however, were fatal (pneumonitis and cerebral hemorrhage), and both occurred in patients with NSCLC.
- The other six grade 3 events included one instance of each of the following: empyema, pneumonia, radiation pneumonitis, radiation skin injury, decreased appetite, and bone pain. Two of these events occurred in patients with NSCLC, two in patients with breast cancer, one in patients with colorectal cancer, and one in patients with prostate cancer.
- New primary malignancies were reported in 13 (0.9%) patients, which included bladder cancer (n = 3), nonmelanoma skin cancer (n = 3), and leukemia (n = 1).
- Overall, 43 (2.9%) patients died within 6 months, most from their primary cancer (58.1%).
IN PRACTICE:
Low rates of early acute toxicities reported in this real-world study help confirm the safety of SBRT in the treatment of oligometastases, the authors concluded. However, “some anatomical sites might be associated with an increased risk of even severe or fatal toxicities.”
SOURCE:
The study, led by Filippo Alongi, Advanced Radiation Oncology Department, IRCCS Sacro Cuore Don Calabria Hospital, Cancer Care Center, Negrar di Valpolicella, Italy, and University of Brescia, also in Italy, was published online in Radiotherapy & Oncology .
LIMITATIONS:
Some limitations of the study include the nonrandomized design and potential variability in patient selection criteria, treatment doses, and schedules.
DISCLOSURES:
The study did not receive any funding support. Two authors declared receiving speaker or lecture honoraria or consultation fees from various sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
according to a large real-world analysis.
METHODOLOGY:
- Advances in cancer imaging have helped identify more patients with oligometastatic disease. Although the standard treatment approach typically involves systemic therapy such as chemotherapy and immunotherapy, SBRT has increasingly become an option for these patients. However, the toxicities associated with SBRT remain less clear.
- OligoCare, a European, prospective, registry-based, single-arm observational study, aims to provide real-world outcomes among patients with oligometastatic cancer who received SBRT. In this analysis, the researchers evaluated early toxicities among 1468 patients with different primary cancers — non–small cell lung cancer (NSCLC; 19.7%), colorectal cancer (20%), breast cancer (15.5%), and prostate cancer (44.8%).
- The primary outcome was acute toxicities, including new malignancies and deaths, within 6 months of initiating SBRT.
- Overall, 527 (35.9%) patients received concomitant systemic treatment and 828 (56%) had de novo oligometastatic disease.
TAKEAWAY:
- Overall, though, only eight patients (0.5%) experienced acute SBRT-related toxicity of grade 3 and above within 6 months; two events, however, were fatal (pneumonitis and cerebral hemorrhage), and both occurred in patients with NSCLC.
- The other six grade 3 events included one instance of each of the following: empyema, pneumonia, radiation pneumonitis, radiation skin injury, decreased appetite, and bone pain. Two of these events occurred in patients with NSCLC, two in patients with breast cancer, one in patients with colorectal cancer, and one in patients with prostate cancer.
- New primary malignancies were reported in 13 (0.9%) patients, which included bladder cancer (n = 3), nonmelanoma skin cancer (n = 3), and leukemia (n = 1).
- Overall, 43 (2.9%) patients died within 6 months, most from their primary cancer (58.1%).
IN PRACTICE:
Low rates of early acute toxicities reported in this real-world study help confirm the safety of SBRT in the treatment of oligometastases, the authors concluded. However, “some anatomical sites might be associated with an increased risk of even severe or fatal toxicities.”
SOURCE:
The study, led by Filippo Alongi, Advanced Radiation Oncology Department, IRCCS Sacro Cuore Don Calabria Hospital, Cancer Care Center, Negrar di Valpolicella, Italy, and University of Brescia, also in Italy, was published online in Radiotherapy & Oncology .
LIMITATIONS:
Some limitations of the study include the nonrandomized design and potential variability in patient selection criteria, treatment doses, and schedules.
DISCLOSURES:
The study did not receive any funding support. Two authors declared receiving speaker or lecture honoraria or consultation fees from various sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Experts Highlight Challenges That Remain for AI Devices in Triaging Skin Cancer
Emerging according to researchers and dermatologists investigating AI.
While some AI-integrated devices designed to triage skin lesions have emerged, including one that received Food and Drug Administration (FDA) clearance earlier in 2024, it may be some time before AI has a meaningful clinical impact in dermatology and, more specifically, the diagnosis of skin cancer, Ivy Lee, MD, a dermatologist in Pasadena, California, and chair of the American Academy of Dermatology’s augmented intelligence committee, told this news organization.
“It hasn’t really translated into clinical practice yet,” Dr. Lee said of AI in dermatology. “There have been significant advances in terms of the technical possibility and feasibility of these tools, but the translation and integration of AI into actual clinical work flows to benefit patients beyond academic research studies has been limited.” More studies and more “easily accessible and digestible information” are needed to evaluate AI tools in dermatologic practice.
“In dermatology, we’re on a cusp with AI,” said Rebecca Hartman, MD, MPH, chief of dermatology at the VA Boston Healthcare System and director of melanoma epidemiology at Brigham and Women’s Hospital, Boston, Massachusetts. “I think it’s going to come and change what we do,” which is especially true for any image-based specialty,” including radiology and pathology, in addition to dermatology.
Dr. Hartman led a study of one of these emerging technologies, the handheld elastic scattering spectroscopy device DermaSensor, which was cleared by the FDA in January for evaluating skin lesions suggestive of skin cancer.
Early AI Devices for Skin Cancer Detection
At the American Society for Laser Medicine and Surgery (ASLMS) meeting in April, a panel explored a number of algorithms with dermatologic applications that use AI to triage skin lesions, including DermaSensor.
Raman spectroscopy, which contains a handheld Raman probe, a diode laser, and a detecting spectrograph. A laser beam — which at 1.56 W/cm2 is below the maximum permissible exposure — focuses on the skin target with a 3.5-mm spot, gathers data on the target, and feeds it back into the unit that houses the algorithm that evaluates the spot analysis. It’s still in the investigative phase. A clinical trial, published almost 5 years ago, demonstrated a sensitivity of 90%-99% and a specificity of 24%-66% for skin cancer.
A dermatoscope called Sklip clips onto a smartphone and performs what company cofounder Alexander Witkowski, MD, PhD, described as an “optical painless virtual biopsy” for at-home use. The device uploads the captured image to an AI platform for analysis. It received FDA breakthrough device designation in 2022. At the ASLMS meeting, Dr. Witkowski said that clinical performance showed the device had a 97% sensitivity and 30% specificity for skin cancer.
DermaSensor, described in the study conducted by Dr. Hartman and others as a noninvasive, point-and-click spectrometer, is a wireless handheld piece that weighs about 10 ounces. The unit captures five recordings to generate a spectral reading, which an algorithm in the software unit analyzes. The study found a sensitivity of 95.5% and specificity of 32.5% for melanoma detection with the device.
The target market for DermaSensor is primary care physicians, and, according to the FDA announcement in January, it is indicated for evaluating skin lesions “suggestive” of melanoma, basal cell carcinoma (BCC), and/or squamous cell carcinoma (SCC) in patients aged 40 and older to “assist healthcare providers in determining whether to refer a patient to a dermatologist.”
So Many Cases, So Few Dermatologists
In dermatology, AI devices have the potential to streamline the crushing burden of diagnosing skin cancer, said Yun Liu, PhD, a senior staff scientist at Google Research, Mountain View, California, who’s worked on developing machine-learning tools in dermatology among other medical fields. “Many people cannot access dermatology expertise when they most need it, ie, without waiting a long time. This causes substantial morbidity for patients,” Dr. Liu said in an interview.
His own research of an AI-based tool to help primary care physicians and nurse practitioners in teledermatology practices diagnose skin conditions documented the shortage of dermatologists to triage lesions, including a finding that only about one quarter of skin conditions are seen by a specialist and that nonspecialists play a pivotal role in the management of skin lesions.
The Centers for Disease Control and Prevention reports that about 6.1 million adults are treated for BCC and SCCs each year. The American Medical Association estimates that 13,200 active dermatologists practice in the United States.
Overcoming Barriers to AI in Dermatology
Before AI makes significant inroads in dermatology, clinicians need to see more verifiable data, said Roxana Daneshjou, MD, PhD, assistant professor of biomedical data science and dermatology at Stanford University, Stanford, California. “One of the challenges is having the availability of models that actually improve clinical care because we have some very early prospective trials on different devices, but we don’t have large-scale randomized clinical trials of AI devices showing definitive behaviors such as improved patient outcomes, that it helps curb skin cancer, or it catches it like dermatologists but helps reduce the biopsy load,” she said. “You need good data.”
Another challenge she noted was overcoming biases built into medicine. “A lot of the image-based models are built on datasets depicting skin disease on White skin, and those models don’t work so well on people with brown and black skin, who have historically had worse outcomes and also have been underrepresented in dermatology,” said Dr. Daneshjou, an associate editor of NEJM AI.
There’s also the challenge of getting verified AI models into the clinic. “Similar to many medical AI endeavors, developing a proof-of-concept or research prototype is far easier and faster than bringing the development to real users,” Dr. Liu said. “In particular, it is important to conduct thorough validation studies on various patient populations and settings and understand how these AI tools can best fit into the workflow or patient journey.”
A study published in 2023 documented progress Google made in deploying AI models in retina specialty clinics in India and Thailand, Dr. Liu noted.
Another challenge is to avoid overdiagnosis with these new technologies, Dr. Hartman said. Her group’s study showed the DermaSensor had a positive predictive value of 16% and a negative predictive value of 98.5%. “I think there’s some question about how this will factor into overdiagnosis. Could this actually bombard dermatologists more if the positive predictive value’s only 16%?”
One key to dermatologists accepting AI tools is having a transparent process for validating them, Dr. Lee said. “Even with FDA clearance, we don’t have the transparency we need as clinicians, researchers, and advocates of machine learning and AI in healthcare.”
But, Dr. Lee noted, the FDA in June took a step toward illuminating its validation process when it adopted guiding principles for transparency for machine learning–enabled devices. “Once we can get more access to this information and have more transparency, that’s where we can think about actually about making the decision to implement or not implement into local healthcare settings,” she said. The process was further enabled by a White House executive order in October 2023 on the safe, secure, and trustworthy development and use of AI.
The experience with telehealth during the COVID-19 pandemic, when patients and providers quickly embraced the technology to stay connected, serves as a potential template for AI, Dr. Lee noted. “As we’d seen with telehealth through the pandemic, you also need the cultural evolution and the development of the infrastructure around it to actually make sure this is a sustainable implementation and a scalable implementation in healthcare.”
Dr. Lee had no relevant relationships to disclose. Dr. Hartman received funding from DermaSensor for a study. Dr. Witkowski is a cofounder of Sklip. Dr. Liu is an employee of Google Research. Dr. Daneshjou reported financial relationships with MD Algorithms, Revea, and L’Oreal.
A version of this article first appeared on Medscape.com.
Emerging according to researchers and dermatologists investigating AI.
While some AI-integrated devices designed to triage skin lesions have emerged, including one that received Food and Drug Administration (FDA) clearance earlier in 2024, it may be some time before AI has a meaningful clinical impact in dermatology and, more specifically, the diagnosis of skin cancer, Ivy Lee, MD, a dermatologist in Pasadena, California, and chair of the American Academy of Dermatology’s augmented intelligence committee, told this news organization.
“It hasn’t really translated into clinical practice yet,” Dr. Lee said of AI in dermatology. “There have been significant advances in terms of the technical possibility and feasibility of these tools, but the translation and integration of AI into actual clinical work flows to benefit patients beyond academic research studies has been limited.” More studies and more “easily accessible and digestible information” are needed to evaluate AI tools in dermatologic practice.
“In dermatology, we’re on a cusp with AI,” said Rebecca Hartman, MD, MPH, chief of dermatology at the VA Boston Healthcare System and director of melanoma epidemiology at Brigham and Women’s Hospital, Boston, Massachusetts. “I think it’s going to come and change what we do,” which is especially true for any image-based specialty,” including radiology and pathology, in addition to dermatology.
Dr. Hartman led a study of one of these emerging technologies, the handheld elastic scattering spectroscopy device DermaSensor, which was cleared by the FDA in January for evaluating skin lesions suggestive of skin cancer.
Early AI Devices for Skin Cancer Detection
At the American Society for Laser Medicine and Surgery (ASLMS) meeting in April, a panel explored a number of algorithms with dermatologic applications that use AI to triage skin lesions, including DermaSensor.
Raman spectroscopy, which contains a handheld Raman probe, a diode laser, and a detecting spectrograph. A laser beam — which at 1.56 W/cm2 is below the maximum permissible exposure — focuses on the skin target with a 3.5-mm spot, gathers data on the target, and feeds it back into the unit that houses the algorithm that evaluates the spot analysis. It’s still in the investigative phase. A clinical trial, published almost 5 years ago, demonstrated a sensitivity of 90%-99% and a specificity of 24%-66% for skin cancer.
A dermatoscope called Sklip clips onto a smartphone and performs what company cofounder Alexander Witkowski, MD, PhD, described as an “optical painless virtual biopsy” for at-home use. The device uploads the captured image to an AI platform for analysis. It received FDA breakthrough device designation in 2022. At the ASLMS meeting, Dr. Witkowski said that clinical performance showed the device had a 97% sensitivity and 30% specificity for skin cancer.
DermaSensor, described in the study conducted by Dr. Hartman and others as a noninvasive, point-and-click spectrometer, is a wireless handheld piece that weighs about 10 ounces. The unit captures five recordings to generate a spectral reading, which an algorithm in the software unit analyzes. The study found a sensitivity of 95.5% and specificity of 32.5% for melanoma detection with the device.
The target market for DermaSensor is primary care physicians, and, according to the FDA announcement in January, it is indicated for evaluating skin lesions “suggestive” of melanoma, basal cell carcinoma (BCC), and/or squamous cell carcinoma (SCC) in patients aged 40 and older to “assist healthcare providers in determining whether to refer a patient to a dermatologist.”
So Many Cases, So Few Dermatologists
In dermatology, AI devices have the potential to streamline the crushing burden of diagnosing skin cancer, said Yun Liu, PhD, a senior staff scientist at Google Research, Mountain View, California, who’s worked on developing machine-learning tools in dermatology among other medical fields. “Many people cannot access dermatology expertise when they most need it, ie, without waiting a long time. This causes substantial morbidity for patients,” Dr. Liu said in an interview.
His own research of an AI-based tool to help primary care physicians and nurse practitioners in teledermatology practices diagnose skin conditions documented the shortage of dermatologists to triage lesions, including a finding that only about one quarter of skin conditions are seen by a specialist and that nonspecialists play a pivotal role in the management of skin lesions.
The Centers for Disease Control and Prevention reports that about 6.1 million adults are treated for BCC and SCCs each year. The American Medical Association estimates that 13,200 active dermatologists practice in the United States.
Overcoming Barriers to AI in Dermatology
Before AI makes significant inroads in dermatology, clinicians need to see more verifiable data, said Roxana Daneshjou, MD, PhD, assistant professor of biomedical data science and dermatology at Stanford University, Stanford, California. “One of the challenges is having the availability of models that actually improve clinical care because we have some very early prospective trials on different devices, but we don’t have large-scale randomized clinical trials of AI devices showing definitive behaviors such as improved patient outcomes, that it helps curb skin cancer, or it catches it like dermatologists but helps reduce the biopsy load,” she said. “You need good data.”
Another challenge she noted was overcoming biases built into medicine. “A lot of the image-based models are built on datasets depicting skin disease on White skin, and those models don’t work so well on people with brown and black skin, who have historically had worse outcomes and also have been underrepresented in dermatology,” said Dr. Daneshjou, an associate editor of NEJM AI.
There’s also the challenge of getting verified AI models into the clinic. “Similar to many medical AI endeavors, developing a proof-of-concept or research prototype is far easier and faster than bringing the development to real users,” Dr. Liu said. “In particular, it is important to conduct thorough validation studies on various patient populations and settings and understand how these AI tools can best fit into the workflow or patient journey.”
A study published in 2023 documented progress Google made in deploying AI models in retina specialty clinics in India and Thailand, Dr. Liu noted.
Another challenge is to avoid overdiagnosis with these new technologies, Dr. Hartman said. Her group’s study showed the DermaSensor had a positive predictive value of 16% and a negative predictive value of 98.5%. “I think there’s some question about how this will factor into overdiagnosis. Could this actually bombard dermatologists more if the positive predictive value’s only 16%?”
One key to dermatologists accepting AI tools is having a transparent process for validating them, Dr. Lee said. “Even with FDA clearance, we don’t have the transparency we need as clinicians, researchers, and advocates of machine learning and AI in healthcare.”
But, Dr. Lee noted, the FDA in June took a step toward illuminating its validation process when it adopted guiding principles for transparency for machine learning–enabled devices. “Once we can get more access to this information and have more transparency, that’s where we can think about actually about making the decision to implement or not implement into local healthcare settings,” she said. The process was further enabled by a White House executive order in October 2023 on the safe, secure, and trustworthy development and use of AI.
The experience with telehealth during the COVID-19 pandemic, when patients and providers quickly embraced the technology to stay connected, serves as a potential template for AI, Dr. Lee noted. “As we’d seen with telehealth through the pandemic, you also need the cultural evolution and the development of the infrastructure around it to actually make sure this is a sustainable implementation and a scalable implementation in healthcare.”
Dr. Lee had no relevant relationships to disclose. Dr. Hartman received funding from DermaSensor for a study. Dr. Witkowski is a cofounder of Sklip. Dr. Liu is an employee of Google Research. Dr. Daneshjou reported financial relationships with MD Algorithms, Revea, and L’Oreal.
A version of this article first appeared on Medscape.com.
Emerging according to researchers and dermatologists investigating AI.
While some AI-integrated devices designed to triage skin lesions have emerged, including one that received Food and Drug Administration (FDA) clearance earlier in 2024, it may be some time before AI has a meaningful clinical impact in dermatology and, more specifically, the diagnosis of skin cancer, Ivy Lee, MD, a dermatologist in Pasadena, California, and chair of the American Academy of Dermatology’s augmented intelligence committee, told this news organization.
“It hasn’t really translated into clinical practice yet,” Dr. Lee said of AI in dermatology. “There have been significant advances in terms of the technical possibility and feasibility of these tools, but the translation and integration of AI into actual clinical work flows to benefit patients beyond academic research studies has been limited.” More studies and more “easily accessible and digestible information” are needed to evaluate AI tools in dermatologic practice.
“In dermatology, we’re on a cusp with AI,” said Rebecca Hartman, MD, MPH, chief of dermatology at the VA Boston Healthcare System and director of melanoma epidemiology at Brigham and Women’s Hospital, Boston, Massachusetts. “I think it’s going to come and change what we do,” which is especially true for any image-based specialty,” including radiology and pathology, in addition to dermatology.
Dr. Hartman led a study of one of these emerging technologies, the handheld elastic scattering spectroscopy device DermaSensor, which was cleared by the FDA in January for evaluating skin lesions suggestive of skin cancer.
Early AI Devices for Skin Cancer Detection
At the American Society for Laser Medicine and Surgery (ASLMS) meeting in April, a panel explored a number of algorithms with dermatologic applications that use AI to triage skin lesions, including DermaSensor.
Raman spectroscopy, which contains a handheld Raman probe, a diode laser, and a detecting spectrograph. A laser beam — which at 1.56 W/cm2 is below the maximum permissible exposure — focuses on the skin target with a 3.5-mm spot, gathers data on the target, and feeds it back into the unit that houses the algorithm that evaluates the spot analysis. It’s still in the investigative phase. A clinical trial, published almost 5 years ago, demonstrated a sensitivity of 90%-99% and a specificity of 24%-66% for skin cancer.
A dermatoscope called Sklip clips onto a smartphone and performs what company cofounder Alexander Witkowski, MD, PhD, described as an “optical painless virtual biopsy” for at-home use. The device uploads the captured image to an AI platform for analysis. It received FDA breakthrough device designation in 2022. At the ASLMS meeting, Dr. Witkowski said that clinical performance showed the device had a 97% sensitivity and 30% specificity for skin cancer.
DermaSensor, described in the study conducted by Dr. Hartman and others as a noninvasive, point-and-click spectrometer, is a wireless handheld piece that weighs about 10 ounces. The unit captures five recordings to generate a spectral reading, which an algorithm in the software unit analyzes. The study found a sensitivity of 95.5% and specificity of 32.5% for melanoma detection with the device.
The target market for DermaSensor is primary care physicians, and, according to the FDA announcement in January, it is indicated for evaluating skin lesions “suggestive” of melanoma, basal cell carcinoma (BCC), and/or squamous cell carcinoma (SCC) in patients aged 40 and older to “assist healthcare providers in determining whether to refer a patient to a dermatologist.”
So Many Cases, So Few Dermatologists
In dermatology, AI devices have the potential to streamline the crushing burden of diagnosing skin cancer, said Yun Liu, PhD, a senior staff scientist at Google Research, Mountain View, California, who’s worked on developing machine-learning tools in dermatology among other medical fields. “Many people cannot access dermatology expertise when they most need it, ie, without waiting a long time. This causes substantial morbidity for patients,” Dr. Liu said in an interview.
His own research of an AI-based tool to help primary care physicians and nurse practitioners in teledermatology practices diagnose skin conditions documented the shortage of dermatologists to triage lesions, including a finding that only about one quarter of skin conditions are seen by a specialist and that nonspecialists play a pivotal role in the management of skin lesions.
The Centers for Disease Control and Prevention reports that about 6.1 million adults are treated for BCC and SCCs each year. The American Medical Association estimates that 13,200 active dermatologists practice in the United States.
Overcoming Barriers to AI in Dermatology
Before AI makes significant inroads in dermatology, clinicians need to see more verifiable data, said Roxana Daneshjou, MD, PhD, assistant professor of biomedical data science and dermatology at Stanford University, Stanford, California. “One of the challenges is having the availability of models that actually improve clinical care because we have some very early prospective trials on different devices, but we don’t have large-scale randomized clinical trials of AI devices showing definitive behaviors such as improved patient outcomes, that it helps curb skin cancer, or it catches it like dermatologists but helps reduce the biopsy load,” she said. “You need good data.”
Another challenge she noted was overcoming biases built into medicine. “A lot of the image-based models are built on datasets depicting skin disease on White skin, and those models don’t work so well on people with brown and black skin, who have historically had worse outcomes and also have been underrepresented in dermatology,” said Dr. Daneshjou, an associate editor of NEJM AI.
There’s also the challenge of getting verified AI models into the clinic. “Similar to many medical AI endeavors, developing a proof-of-concept or research prototype is far easier and faster than bringing the development to real users,” Dr. Liu said. “In particular, it is important to conduct thorough validation studies on various patient populations and settings and understand how these AI tools can best fit into the workflow or patient journey.”
A study published in 2023 documented progress Google made in deploying AI models in retina specialty clinics in India and Thailand, Dr. Liu noted.
Another challenge is to avoid overdiagnosis with these new technologies, Dr. Hartman said. Her group’s study showed the DermaSensor had a positive predictive value of 16% and a negative predictive value of 98.5%. “I think there’s some question about how this will factor into overdiagnosis. Could this actually bombard dermatologists more if the positive predictive value’s only 16%?”
One key to dermatologists accepting AI tools is having a transparent process for validating them, Dr. Lee said. “Even with FDA clearance, we don’t have the transparency we need as clinicians, researchers, and advocates of machine learning and AI in healthcare.”
But, Dr. Lee noted, the FDA in June took a step toward illuminating its validation process when it adopted guiding principles for transparency for machine learning–enabled devices. “Once we can get more access to this information and have more transparency, that’s where we can think about actually about making the decision to implement or not implement into local healthcare settings,” she said. The process was further enabled by a White House executive order in October 2023 on the safe, secure, and trustworthy development and use of AI.
The experience with telehealth during the COVID-19 pandemic, when patients and providers quickly embraced the technology to stay connected, serves as a potential template for AI, Dr. Lee noted. “As we’d seen with telehealth through the pandemic, you also need the cultural evolution and the development of the infrastructure around it to actually make sure this is a sustainable implementation and a scalable implementation in healthcare.”
Dr. Lee had no relevant relationships to disclose. Dr. Hartman received funding from DermaSensor for a study. Dr. Witkowski is a cofounder of Sklip. Dr. Liu is an employee of Google Research. Dr. Daneshjou reported financial relationships with MD Algorithms, Revea, and L’Oreal.
A version of this article first appeared on Medscape.com.
Who’s Behind Cosmetic Procedures at MedSpas?
CARLSBAD, CALIFORNIA — according to Sara Hogan, MD.
“I’m not anti-MedSpa; I’m pro-patient safety,” Dr. Hogan, clinical assistant professor of dermatology at George Washington University, Washington, DC, said at the Controversies & Conversations in Laser & Cosmetic Surgery symposium. “The MedSpa industry is booming; it brought in $17 billion in 2023. There are as many MedSpas in the United States as there are practicing dermatologists, and that number is set to exceed the number of dermatologists.”
According to industry data from the American Med Spa Association, 63% of member MedSpas have non-MD ownership. Among MedSpas owned by physicians, 80% are of a non–core specialty, meaning a specialty other than dermatology, plastic surgery, otorhinolaryngology, or ophthalmology. Of MedSpa medical directors, 69% are from non–core physician specialties. “There’s an increasing amount of data that shows a relatively higher incidence of complications from cosmetic procedures that are delivered at MedSpas,” Dr. Hogan said. “A 2020 study suggested that this is likely due to improper training, improper technique, and/or improper device settings.”
Dr. Hogan also cited adverse effects linked to counterfeit or mishandled botulinum toxin injections that prompted the Centers for Disease Control and Prevention to issue an alert to clinicians in April 2024. Clusters of 22 people in 11 states reported adverse effects after receiving injections with counterfeit botulinum toxin or injections administered by unlicensed or untrained individuals or in non-healthcare settings, such as homes or spas.
To better understand who performs cosmetic procedures, provides medical supervision, and follows safety protocols at MedSpas, Dr. Hogan and colleagues conducted a “truth in advertising” study of 127 MedSpas in the greater Chicago area. They chose this geographic location because an analysis published in 2021 identified Chicago as having the third highest number of aesthetic physicians and the fifth highest number of MedSpas in the United States. The researchers enlisted help from “secret shoppers” who contacted the MedSpas by telephone to ask about the level of training, if patients underwent a review of medical history, the level of on-site physician supervision, and the protocol for complications.
The top five cosmetic procedures offered by the 127 surveyed MedSpas were facials (85.0%), hair removal (85.0%), botulinum toxin injections (83.5%), dermal fillers (82.7%), and chemical peels (76.4%). About two thirds of cosmetic procedures were performed by aestheticians (66.9%), followed by registered nurses or licensed practical nurses (52.8%), board-certified physicians (48.8%, mostly plastic and reconstructive surgeons), nurse practitioners (27.6%), and physician assistants (9.4%).
In the realm of supervision, 16.5% of MedSpas surveyed reported that a medical director or supervising physician is always on site. “If not located on site, when asked where the physicians are, the majority of the time they were at the physician’s primary practice, clinic, or hospital,” Dr. Hogan said. “Only 65% of the MedSpas surveyed stated that they informed the patient that the supervising physician is not on site. In addition, a patient’s medical history is reviewed at only 40% of the MedSpas. To give context, in Illinois, a physician can only deliver care after a physician-patient relationship has been established, meaning that a good faith exam has been performed. And if they are to delegate any type of service, they must always be on site to provide assistance.”
Dr. Hogan noted that there are no federal statutes or agencies that regulate or oversee MedSpas. “Regulation and oversight are often delegated to state licensing agencies that are overwhelmed and often stretched thin regarding personnel and budgets,” she said. To raise awareness of this issue, the American Society for Dermatologic Surgery Association (ASDSA) launched the Medical Spa Safety Coalition, which aims to promote model legislation for states known as the Medical Spa Safety Act. Highlights of the bill include clear definitions of medical spa and medical director, as well as the requirement of an on-site medical director who must be a physician trained in all procedures performed at the MedSpa. Coalition members include 16 state dermatology boards as well as the ASDSA, the American Academy of Dermatology Association, the American Society for Laser Medicine & Surgery, and the American Society of Plastic Surgeons.
The ASDSA provided funding to support the published study. Dr. Hogan reported having no financial disclosures.
A version of this article appeared on Medscape.com.
CARLSBAD, CALIFORNIA — according to Sara Hogan, MD.
“I’m not anti-MedSpa; I’m pro-patient safety,” Dr. Hogan, clinical assistant professor of dermatology at George Washington University, Washington, DC, said at the Controversies & Conversations in Laser & Cosmetic Surgery symposium. “The MedSpa industry is booming; it brought in $17 billion in 2023. There are as many MedSpas in the United States as there are practicing dermatologists, and that number is set to exceed the number of dermatologists.”
According to industry data from the American Med Spa Association, 63% of member MedSpas have non-MD ownership. Among MedSpas owned by physicians, 80% are of a non–core specialty, meaning a specialty other than dermatology, plastic surgery, otorhinolaryngology, or ophthalmology. Of MedSpa medical directors, 69% are from non–core physician specialties. “There’s an increasing amount of data that shows a relatively higher incidence of complications from cosmetic procedures that are delivered at MedSpas,” Dr. Hogan said. “A 2020 study suggested that this is likely due to improper training, improper technique, and/or improper device settings.”
Dr. Hogan also cited adverse effects linked to counterfeit or mishandled botulinum toxin injections that prompted the Centers for Disease Control and Prevention to issue an alert to clinicians in April 2024. Clusters of 22 people in 11 states reported adverse effects after receiving injections with counterfeit botulinum toxin or injections administered by unlicensed or untrained individuals or in non-healthcare settings, such as homes or spas.
To better understand who performs cosmetic procedures, provides medical supervision, and follows safety protocols at MedSpas, Dr. Hogan and colleagues conducted a “truth in advertising” study of 127 MedSpas in the greater Chicago area. They chose this geographic location because an analysis published in 2021 identified Chicago as having the third highest number of aesthetic physicians and the fifth highest number of MedSpas in the United States. The researchers enlisted help from “secret shoppers” who contacted the MedSpas by telephone to ask about the level of training, if patients underwent a review of medical history, the level of on-site physician supervision, and the protocol for complications.
The top five cosmetic procedures offered by the 127 surveyed MedSpas were facials (85.0%), hair removal (85.0%), botulinum toxin injections (83.5%), dermal fillers (82.7%), and chemical peels (76.4%). About two thirds of cosmetic procedures were performed by aestheticians (66.9%), followed by registered nurses or licensed practical nurses (52.8%), board-certified physicians (48.8%, mostly plastic and reconstructive surgeons), nurse practitioners (27.6%), and physician assistants (9.4%).
In the realm of supervision, 16.5% of MedSpas surveyed reported that a medical director or supervising physician is always on site. “If not located on site, when asked where the physicians are, the majority of the time they were at the physician’s primary practice, clinic, or hospital,” Dr. Hogan said. “Only 65% of the MedSpas surveyed stated that they informed the patient that the supervising physician is not on site. In addition, a patient’s medical history is reviewed at only 40% of the MedSpas. To give context, in Illinois, a physician can only deliver care after a physician-patient relationship has been established, meaning that a good faith exam has been performed. And if they are to delegate any type of service, they must always be on site to provide assistance.”
Dr. Hogan noted that there are no federal statutes or agencies that regulate or oversee MedSpas. “Regulation and oversight are often delegated to state licensing agencies that are overwhelmed and often stretched thin regarding personnel and budgets,” she said. To raise awareness of this issue, the American Society for Dermatologic Surgery Association (ASDSA) launched the Medical Spa Safety Coalition, which aims to promote model legislation for states known as the Medical Spa Safety Act. Highlights of the bill include clear definitions of medical spa and medical director, as well as the requirement of an on-site medical director who must be a physician trained in all procedures performed at the MedSpa. Coalition members include 16 state dermatology boards as well as the ASDSA, the American Academy of Dermatology Association, the American Society for Laser Medicine & Surgery, and the American Society of Plastic Surgeons.
The ASDSA provided funding to support the published study. Dr. Hogan reported having no financial disclosures.
A version of this article appeared on Medscape.com.
CARLSBAD, CALIFORNIA — according to Sara Hogan, MD.
“I’m not anti-MedSpa; I’m pro-patient safety,” Dr. Hogan, clinical assistant professor of dermatology at George Washington University, Washington, DC, said at the Controversies & Conversations in Laser & Cosmetic Surgery symposium. “The MedSpa industry is booming; it brought in $17 billion in 2023. There are as many MedSpas in the United States as there are practicing dermatologists, and that number is set to exceed the number of dermatologists.”
According to industry data from the American Med Spa Association, 63% of member MedSpas have non-MD ownership. Among MedSpas owned by physicians, 80% are of a non–core specialty, meaning a specialty other than dermatology, plastic surgery, otorhinolaryngology, or ophthalmology. Of MedSpa medical directors, 69% are from non–core physician specialties. “There’s an increasing amount of data that shows a relatively higher incidence of complications from cosmetic procedures that are delivered at MedSpas,” Dr. Hogan said. “A 2020 study suggested that this is likely due to improper training, improper technique, and/or improper device settings.”
Dr. Hogan also cited adverse effects linked to counterfeit or mishandled botulinum toxin injections that prompted the Centers for Disease Control and Prevention to issue an alert to clinicians in April 2024. Clusters of 22 people in 11 states reported adverse effects after receiving injections with counterfeit botulinum toxin or injections administered by unlicensed or untrained individuals or in non-healthcare settings, such as homes or spas.
To better understand who performs cosmetic procedures, provides medical supervision, and follows safety protocols at MedSpas, Dr. Hogan and colleagues conducted a “truth in advertising” study of 127 MedSpas in the greater Chicago area. They chose this geographic location because an analysis published in 2021 identified Chicago as having the third highest number of aesthetic physicians and the fifth highest number of MedSpas in the United States. The researchers enlisted help from “secret shoppers” who contacted the MedSpas by telephone to ask about the level of training, if patients underwent a review of medical history, the level of on-site physician supervision, and the protocol for complications.
The top five cosmetic procedures offered by the 127 surveyed MedSpas were facials (85.0%), hair removal (85.0%), botulinum toxin injections (83.5%), dermal fillers (82.7%), and chemical peels (76.4%). About two thirds of cosmetic procedures were performed by aestheticians (66.9%), followed by registered nurses or licensed practical nurses (52.8%), board-certified physicians (48.8%, mostly plastic and reconstructive surgeons), nurse practitioners (27.6%), and physician assistants (9.4%).
In the realm of supervision, 16.5% of MedSpas surveyed reported that a medical director or supervising physician is always on site. “If not located on site, when asked where the physicians are, the majority of the time they were at the physician’s primary practice, clinic, or hospital,” Dr. Hogan said. “Only 65% of the MedSpas surveyed stated that they informed the patient that the supervising physician is not on site. In addition, a patient’s medical history is reviewed at only 40% of the MedSpas. To give context, in Illinois, a physician can only deliver care after a physician-patient relationship has been established, meaning that a good faith exam has been performed. And if they are to delegate any type of service, they must always be on site to provide assistance.”
Dr. Hogan noted that there are no federal statutes or agencies that regulate or oversee MedSpas. “Regulation and oversight are often delegated to state licensing agencies that are overwhelmed and often stretched thin regarding personnel and budgets,” she said. To raise awareness of this issue, the American Society for Dermatologic Surgery Association (ASDSA) launched the Medical Spa Safety Coalition, which aims to promote model legislation for states known as the Medical Spa Safety Act. Highlights of the bill include clear definitions of medical spa and medical director, as well as the requirement of an on-site medical director who must be a physician trained in all procedures performed at the MedSpa. Coalition members include 16 state dermatology boards as well as the ASDSA, the American Academy of Dermatology Association, the American Society for Laser Medicine & Surgery, and the American Society of Plastic Surgeons.
The ASDSA provided funding to support the published study. Dr. Hogan reported having no financial disclosures.
A version of this article appeared on Medscape.com.
Hip Fractures in Patients With Dementia: To Operate or Not?
In the case of a hip fracture, operating on a patient with dementia can be a difficult decision to make. Indeed, surgery exposes patients with dementia to a higher mortality rate, more delirium and postoperative complications, and a greater loss of mobility than patients of the same age without cognitive impairments. For patients with dementia in institutional settings, survival is better for those who undergo surgery than for those who do not. But what about the prognosis of surgery vs no surgery for patients with dementia who live at home?
To answer this question, researchers in the United States conducted a cohort study using Medicare data. This retrospective study included patients aged 66 years and older with dementia who were living at home and not placed in institutions and who had a hip fracture between January 2017 and June 2018. Patients with incomplete observations, particularly regarding the location and type of residence (home/institution), were excluded from the analysis. Fractures were categorized as (i) fractures of the head and neck, (ii) pertrochanteric, (iii) subtrochanteric, and (iv) multiple/complex. The type and severity (mild, moderate, or severe) of dementia were identified using the diagnostic code list in the International Statistical Classification of Diseases, Tenth Revision.
Examining Mortality
The primary outcome was mortality at 30, 90, and 180 days. Secondary outcomes included hospital delirium; the need for subsequent hospitalization (within 6 months) or home health services (within 10 days); and intensive care interventions such as dialysis, intubation, resuscitation maneuvers, mechanical ventilation, or the insertion of a feeding tube.
Postoperative medical facility admissions were distinguished according to whether there was a plan to return home. To compare the surgery and nonsurgery groups, an inverse propensity score analysis was conducted within subgroups determined by fracture type, comorbidities (using the Elixhauser score), the person or entity responsible for admission (ie, physician, clinic, hospital, etc.), dual eligibility (Medicare and Medicaid), place of residence (ie, urban or rural), race, and sex. Dementia severity was estimated using a frailty index on the basis of evaluation tests that were systematically collected in the Medicare database.
Results Favored Surgery
Among 56,209 patients with dementia who were admitted for a hip fracture (73.0% women; mean age, 86.4 ± 7.0 years), 33,142 (59.0%) underwent surgery and 23,067 (41.0%) did not. Among surgically treated patients, 73.3% had fractures of the head or neck of the femur, and 40.2% had moderate to severe dementia. The nonsurgically treated fractures were 78.5% pertrochanteric. Comorbidities were evenly distributed between the two groups.
At 180 days, mortality was 31.8% in the surgery group compared with 45.7% in the nonsurgery group, resulting in a significant reduction in the unadjusted relative risk (RR) for death in favor of surgery (RR, 0.67; 95% CI, 0.60-0.76; P < .001). Among patients with mild dementia and a fracture of the head or neck of the femur, mortality at 180 days was 26.5% among surgical patients and 34.9% among nonsurgical patients (RR, 0.67; 95% CI, 0.60-0.76; P < .001). After the investigators adjusted for risk according to propensity score, the benefit of surgery remained significant at 30, 90, and 180 days, regardless of dementia severity. There was no significant difference in mortality for other types of hip fractures between the surgery and nonsurgery groups, however.
The adjusted RR for in-hospital delirium was 1.23 (P = .008), which was significant for the surgery group, but only for those with moderate to severe dementia. There were also fewer permanent placements (P < .001) among the surgically treated patients, and fewer patients with mild dementia required nurse care at home. There was no difference in resuscitation maneuvers between surgery and nonsurgery patients, whether the dementia was mild or not. For patients with a fracture of the head or neck of the femur, there was no difference in the likelihood of rehabilitation admission within 180 days, whether they were operated on or not.
Ethical Considerations
This study can inform discussions among healthcare professionals, patients, and patients’ families about which goals to set and which strategy to choose. The main interest of this study lies in its comparison of outcomes between patients with dementia who were operated on and those who were not, rather than comparing patients with and without dementia. Among patients with dementia living at home with a fracture of the head or neck of the femur, those who underwent surgery had a lower risk for death than those who did not, regardless of the severity of dementia.
It is noteworthy that less than two thirds of patients with dementia underwent surgery, which contradicts recommendations for almost routine surgery for patients with dementia. This observation raises questions about respecting patient wishes and advance directives when known, possible detrimental delays in referrals, and legal-medical issues.
Furthermore, the treatment choices of American surgeons are clearly influenced by the type of hip fracture. Fractures of the head and neck of the femur are typically treated with prosthetic arthroplasty, which simplifies postoperative care, compared with osteosynthesis. The latter procedure is more often used for extra-articular hip fractures but entails higher risks. While survival is an apparently more easily achievable goal through surgery, ethical considerations about other treatment objectives such as pain control, functional recovery, and treatment adequacy cannot be overlooked. It is worth noting that the French National Authority for Health issued recommendations in 2018 regarding the care pathway for patients hospitalized for a hip fracture within an orthogeriatric organization.
This story was translated from JIM, which is part of the Medscape Medical News professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
In the case of a hip fracture, operating on a patient with dementia can be a difficult decision to make. Indeed, surgery exposes patients with dementia to a higher mortality rate, more delirium and postoperative complications, and a greater loss of mobility than patients of the same age without cognitive impairments. For patients with dementia in institutional settings, survival is better for those who undergo surgery than for those who do not. But what about the prognosis of surgery vs no surgery for patients with dementia who live at home?
To answer this question, researchers in the United States conducted a cohort study using Medicare data. This retrospective study included patients aged 66 years and older with dementia who were living at home and not placed in institutions and who had a hip fracture between January 2017 and June 2018. Patients with incomplete observations, particularly regarding the location and type of residence (home/institution), were excluded from the analysis. Fractures were categorized as (i) fractures of the head and neck, (ii) pertrochanteric, (iii) subtrochanteric, and (iv) multiple/complex. The type and severity (mild, moderate, or severe) of dementia were identified using the diagnostic code list in the International Statistical Classification of Diseases, Tenth Revision.
Examining Mortality
The primary outcome was mortality at 30, 90, and 180 days. Secondary outcomes included hospital delirium; the need for subsequent hospitalization (within 6 months) or home health services (within 10 days); and intensive care interventions such as dialysis, intubation, resuscitation maneuvers, mechanical ventilation, or the insertion of a feeding tube.
Postoperative medical facility admissions were distinguished according to whether there was a plan to return home. To compare the surgery and nonsurgery groups, an inverse propensity score analysis was conducted within subgroups determined by fracture type, comorbidities (using the Elixhauser score), the person or entity responsible for admission (ie, physician, clinic, hospital, etc.), dual eligibility (Medicare and Medicaid), place of residence (ie, urban or rural), race, and sex. Dementia severity was estimated using a frailty index on the basis of evaluation tests that were systematically collected in the Medicare database.
Results Favored Surgery
Among 56,209 patients with dementia who were admitted for a hip fracture (73.0% women; mean age, 86.4 ± 7.0 years), 33,142 (59.0%) underwent surgery and 23,067 (41.0%) did not. Among surgically treated patients, 73.3% had fractures of the head or neck of the femur, and 40.2% had moderate to severe dementia. The nonsurgically treated fractures were 78.5% pertrochanteric. Comorbidities were evenly distributed between the two groups.
At 180 days, mortality was 31.8% in the surgery group compared with 45.7% in the nonsurgery group, resulting in a significant reduction in the unadjusted relative risk (RR) for death in favor of surgery (RR, 0.67; 95% CI, 0.60-0.76; P < .001). Among patients with mild dementia and a fracture of the head or neck of the femur, mortality at 180 days was 26.5% among surgical patients and 34.9% among nonsurgical patients (RR, 0.67; 95% CI, 0.60-0.76; P < .001). After the investigators adjusted for risk according to propensity score, the benefit of surgery remained significant at 30, 90, and 180 days, regardless of dementia severity. There was no significant difference in mortality for other types of hip fractures between the surgery and nonsurgery groups, however.
The adjusted RR for in-hospital delirium was 1.23 (P = .008), which was significant for the surgery group, but only for those with moderate to severe dementia. There were also fewer permanent placements (P < .001) among the surgically treated patients, and fewer patients with mild dementia required nurse care at home. There was no difference in resuscitation maneuvers between surgery and nonsurgery patients, whether the dementia was mild or not. For patients with a fracture of the head or neck of the femur, there was no difference in the likelihood of rehabilitation admission within 180 days, whether they were operated on or not.
Ethical Considerations
This study can inform discussions among healthcare professionals, patients, and patients’ families about which goals to set and which strategy to choose. The main interest of this study lies in its comparison of outcomes between patients with dementia who were operated on and those who were not, rather than comparing patients with and without dementia. Among patients with dementia living at home with a fracture of the head or neck of the femur, those who underwent surgery had a lower risk for death than those who did not, regardless of the severity of dementia.
It is noteworthy that less than two thirds of patients with dementia underwent surgery, which contradicts recommendations for almost routine surgery for patients with dementia. This observation raises questions about respecting patient wishes and advance directives when known, possible detrimental delays in referrals, and legal-medical issues.
Furthermore, the treatment choices of American surgeons are clearly influenced by the type of hip fracture. Fractures of the head and neck of the femur are typically treated with prosthetic arthroplasty, which simplifies postoperative care, compared with osteosynthesis. The latter procedure is more often used for extra-articular hip fractures but entails higher risks. While survival is an apparently more easily achievable goal through surgery, ethical considerations about other treatment objectives such as pain control, functional recovery, and treatment adequacy cannot be overlooked. It is worth noting that the French National Authority for Health issued recommendations in 2018 regarding the care pathway for patients hospitalized for a hip fracture within an orthogeriatric organization.
This story was translated from JIM, which is part of the Medscape Medical News professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
In the case of a hip fracture, operating on a patient with dementia can be a difficult decision to make. Indeed, surgery exposes patients with dementia to a higher mortality rate, more delirium and postoperative complications, and a greater loss of mobility than patients of the same age without cognitive impairments. For patients with dementia in institutional settings, survival is better for those who undergo surgery than for those who do not. But what about the prognosis of surgery vs no surgery for patients with dementia who live at home?
To answer this question, researchers in the United States conducted a cohort study using Medicare data. This retrospective study included patients aged 66 years and older with dementia who were living at home and not placed in institutions and who had a hip fracture between January 2017 and June 2018. Patients with incomplete observations, particularly regarding the location and type of residence (home/institution), were excluded from the analysis. Fractures were categorized as (i) fractures of the head and neck, (ii) pertrochanteric, (iii) subtrochanteric, and (iv) multiple/complex. The type and severity (mild, moderate, or severe) of dementia were identified using the diagnostic code list in the International Statistical Classification of Diseases, Tenth Revision.
Examining Mortality
The primary outcome was mortality at 30, 90, and 180 days. Secondary outcomes included hospital delirium; the need for subsequent hospitalization (within 6 months) or home health services (within 10 days); and intensive care interventions such as dialysis, intubation, resuscitation maneuvers, mechanical ventilation, or the insertion of a feeding tube.
Postoperative medical facility admissions were distinguished according to whether there was a plan to return home. To compare the surgery and nonsurgery groups, an inverse propensity score analysis was conducted within subgroups determined by fracture type, comorbidities (using the Elixhauser score), the person or entity responsible for admission (ie, physician, clinic, hospital, etc.), dual eligibility (Medicare and Medicaid), place of residence (ie, urban or rural), race, and sex. Dementia severity was estimated using a frailty index on the basis of evaluation tests that were systematically collected in the Medicare database.
Results Favored Surgery
Among 56,209 patients with dementia who were admitted for a hip fracture (73.0% women; mean age, 86.4 ± 7.0 years), 33,142 (59.0%) underwent surgery and 23,067 (41.0%) did not. Among surgically treated patients, 73.3% had fractures of the head or neck of the femur, and 40.2% had moderate to severe dementia. The nonsurgically treated fractures were 78.5% pertrochanteric. Comorbidities were evenly distributed between the two groups.
At 180 days, mortality was 31.8% in the surgery group compared with 45.7% in the nonsurgery group, resulting in a significant reduction in the unadjusted relative risk (RR) for death in favor of surgery (RR, 0.67; 95% CI, 0.60-0.76; P < .001). Among patients with mild dementia and a fracture of the head or neck of the femur, mortality at 180 days was 26.5% among surgical patients and 34.9% among nonsurgical patients (RR, 0.67; 95% CI, 0.60-0.76; P < .001). After the investigators adjusted for risk according to propensity score, the benefit of surgery remained significant at 30, 90, and 180 days, regardless of dementia severity. There was no significant difference in mortality for other types of hip fractures between the surgery and nonsurgery groups, however.
The adjusted RR for in-hospital delirium was 1.23 (P = .008), which was significant for the surgery group, but only for those with moderate to severe dementia. There were also fewer permanent placements (P < .001) among the surgically treated patients, and fewer patients with mild dementia required nurse care at home. There was no difference in resuscitation maneuvers between surgery and nonsurgery patients, whether the dementia was mild or not. For patients with a fracture of the head or neck of the femur, there was no difference in the likelihood of rehabilitation admission within 180 days, whether they were operated on or not.
Ethical Considerations
This study can inform discussions among healthcare professionals, patients, and patients’ families about which goals to set and which strategy to choose. The main interest of this study lies in its comparison of outcomes between patients with dementia who were operated on and those who were not, rather than comparing patients with and without dementia. Among patients with dementia living at home with a fracture of the head or neck of the femur, those who underwent surgery had a lower risk for death than those who did not, regardless of the severity of dementia.
It is noteworthy that less than two thirds of patients with dementia underwent surgery, which contradicts recommendations for almost routine surgery for patients with dementia. This observation raises questions about respecting patient wishes and advance directives when known, possible detrimental delays in referrals, and legal-medical issues.
Furthermore, the treatment choices of American surgeons are clearly influenced by the type of hip fracture. Fractures of the head and neck of the femur are typically treated with prosthetic arthroplasty, which simplifies postoperative care, compared with osteosynthesis. The latter procedure is more often used for extra-articular hip fractures but entails higher risks. While survival is an apparently more easily achievable goal through surgery, ethical considerations about other treatment objectives such as pain control, functional recovery, and treatment adequacy cannot be overlooked. It is worth noting that the French National Authority for Health issued recommendations in 2018 regarding the care pathway for patients hospitalized for a hip fracture within an orthogeriatric organization.
This story was translated from JIM, which is part of the Medscape Medical News professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Predicting RSV’s Role in the Upcoming Winter Respiratory Season
For children younger than 5 years old, RSV is the main drive — approximately 2,000,000 outpatient/ED visits and about 75,000 hospitalizations annually. RSV disease ranges from upper respiratory tract infections, eg, in older children and healthy adults, to more severe lower tract disease in young children and the elderly. Premature infants and high-risk groups are particularly prone to severe disease.1 Up to 300 pediatric RSV deaths occur yearly. “Normal” RSV seasons start in mid-November, peak in late December-January, and end after April. Note: More drawn out seasons occur in southern latitudes, eg Texas or Florida. But lately RSV seasons have been anything but normal.
2015-2016 to 2022-2023
RSV data from the Pediatric Health Information System (PHIS), collected at over 49 US children’s hospitals during 2015 to early 2023, show how crazy RSV seasons have been lately.2 The involved months, intensity, and duration of four prepandemic seasons were pretty “normal” (Figure 1). The 2019-2020 season started normally, peaked in January 2020, and was slowing as expected by February. But when SARS-Cov-2 restrictions kicked in during mid-March, RSV detections tanked to almost nothing (ditto other respiratory viruses). A near 14-month RSV hiatus meant that the 2020-2021 RSV season never materialized. However, RSV was not done with us in 2021. It rebounded in May with weekly hospitalizations peaking in late July; this “rebound season” lasted 9 months, not dropping to baseline until February 2022 (Figure 1).
I guess we should have expected a post-pandemic “disturbance in the Force,” as Yoda once said; but I sure didn’t see a prolonged summer/fall/early winter RSV season coming. It was like two “normal” seasons mashed up into one late-but-long season. Not to be outdone, the 2022-2023 RSV season started early (September) and hospitalizations skyrocketed to peak in November at over twice the peak number from any year since 2015, overloading hospitals (influenza and SARS-Cov-2 seasons were co-circulating). The season terminated early though (March 2023).
Okay, so RSV seasonality/intensity were weird post pandemic, but was anything else different? Some 2021-2023 data suggest more RSV disease in older children, rather than the usual younger than 18 month-olds going through their first winter.3 More medically attended RSV in older ages (2-4 years of life) may have been due to the pandemic year without RSV circulation distorting herd immunity, ie older children remained RSV naive. Other data suggest the apparent increase was really just more frequent multiplex viral testing in older children triggered by SARS-CoV-2 co-circulation.4 More data are needed to decide.
CDC 2023-2024 RESP-NET data
The 2023-2024 winter surge (Figure 2), as measured by RESP-NET’s cumulative RSV, influenza and SARS-CoV-2 hospitalization rates for 0- to 5-year-olds,5 shows that all three viruses’ seasonal months were normal-ish: late October 2023 start, late December-early January peak, and mid-May 2024 return to baseline. RSV season was approximately 22% less severe by area-under-the-curve calculations compared with 2022-2023, but still worse than prepandemic years.6 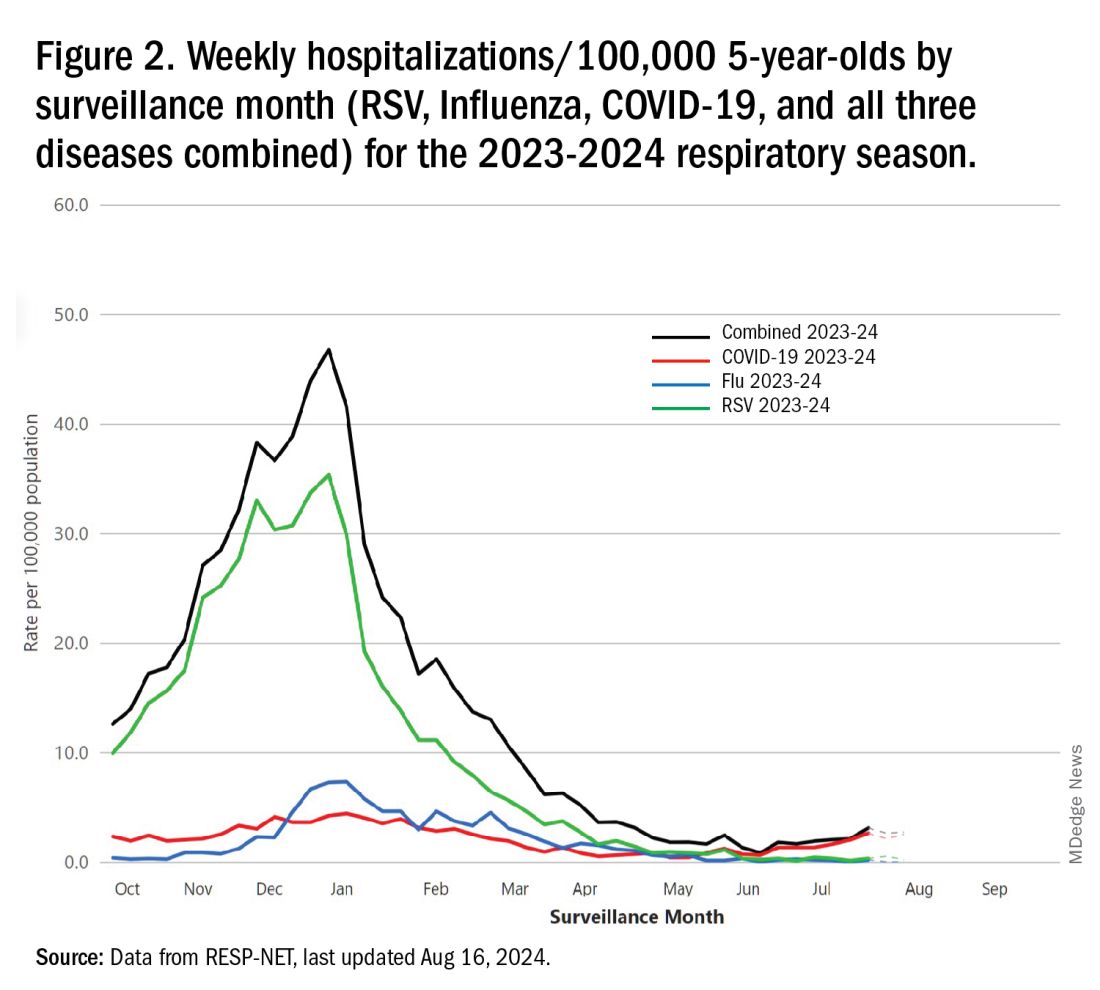
One wonders if the 2022-2023 RSV season might have been worse but for use of the limited supply of nirsevimab.7
Viral Parade
Now we ready ourselves for the 2024-2025 respiratory surge, wondering what nature has in store for us. Will the usual “respiratory virus parade” occur? Will rhinovirus and parainfluenza prevalence bump after a few weeks of schools being in session, adding to the now-usual summer/fall SARS-CoV-2 surge? Note: Twenty-seven states as of Aug. 16 had high SARS-CoV-2 detection in wastewater. Will RSV and influenza start sometime in October/November, peak in January (along with rising SARS-CoV2 activity), followed by a second parainfluenza bump as SARS-CoV-2, influenza, and RSV drop off in April/May? Further, will RSV and influenza seasons be more or less severe than the last 2 years?
Prediction
The overall 2024-2025 respiratory season will be less severe than the past 2 years and hopefully than recent prepandemic years. What is the blueprint for a milder season? First, herd immunity to non-RSV and non-influenza viruses (parainfluenza, rhinovirus, metapneumovirus, adenovirus) in older children should be normalized after 2 years back to usual social activity. So, I expect no mega-seasons from them. The emerging SARS-CoV-2 virus (LB.1) is immunologically close to its recent still-circulating ancestors (KP.2, KP.2.3, KP.3 and KP.3.1.1), so existing SARS-CoV2 herd immunity along with recommended booster vaccine uptake should keep the lid on SARS-CoV2.
Influenza Could Be the Bad News
Which type will dominate? Will a drift/shift occur or vaccine-mismatch reduce vaccine effectiveness? Can we get at least half the population influenza vaccinated, given the vaccine fatigue permeating the US population? The influenza season now underway in the Southern Hemisphere usually helps us predict our season. The Australian May-August 2024 experience (still on an upward trajectory for severity in mid-August) saw no drift/shift or vaccine mismatch. However, this 2024 season has been as severe as 2022 (their worst in a decade). That said, more than 95% has been type A (mostly H1N1 but H3N2 increased in July). So, if our overall 2024-2025 respiratory season is not milder, influenza is the most likely culprit. To reduce chances of influenza being the fly-in-the-ointment, we need to be particularly proactive with seasonal influenza vaccine which is back to the traditional trivalent formulation (one H1N1, one H3N2, and one B type).8 All of this could go out the window if avian influenza becomes more transmissible, but that seems unlikely at present.
Mild RSV Season?
RSV season should be blunted because of the increased use of both the remarkably effective CDC-recommended maternal RSV vaccine9 (one dose during pregnancy weeks 32 through 36, administered September through January) and of nirsevimab (up to 90% reduction in hospitalizations and ED visits).10 (See Figure 3.) 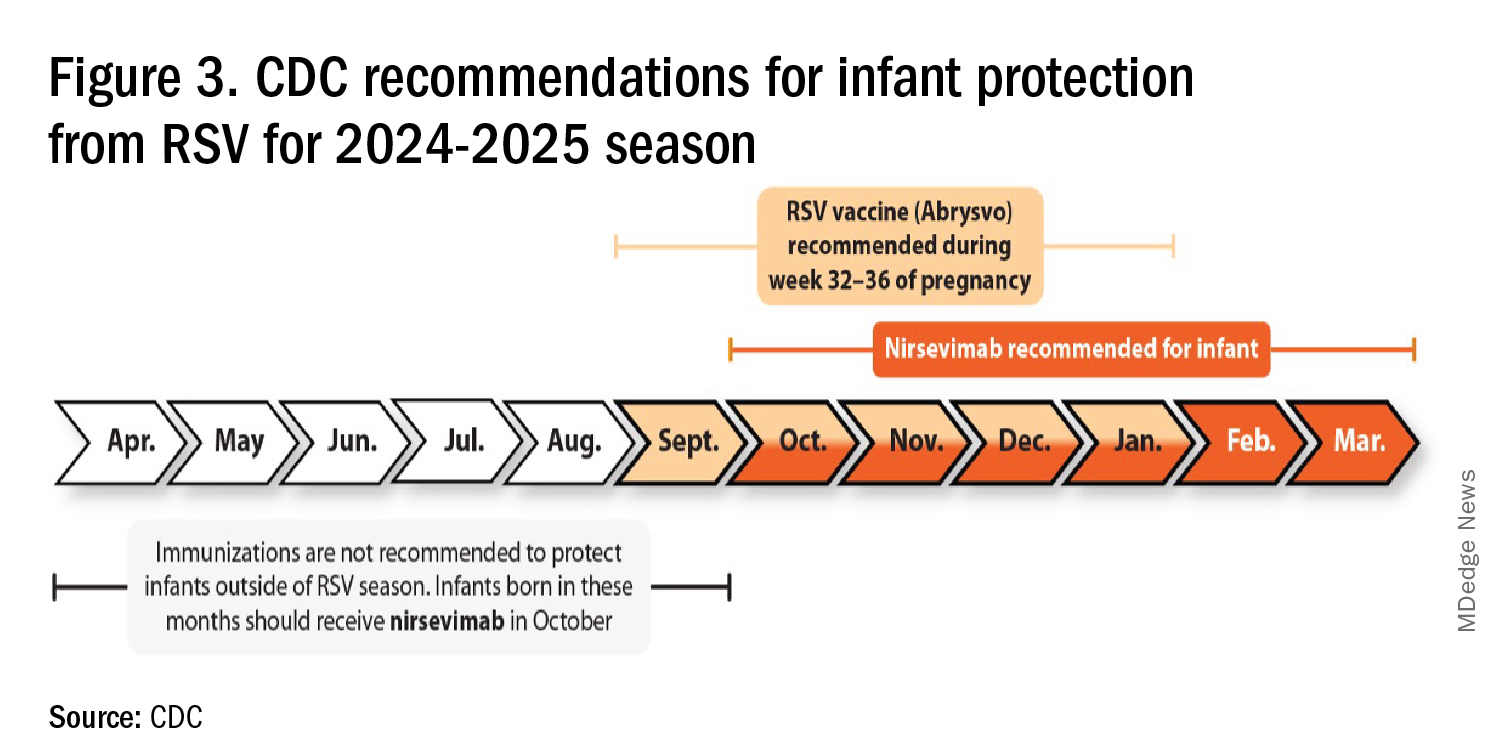
I also expect residual disease to occur mostly in younger than 18 month-olds (the “normal” aged population experiencing their first winter), who received no passive immunity (mother RSV unvaccinated and child did not receive nirsevimab). Some disease will still occur in high-risk infants/children. However, unlike active vaccination strategies, a competent immune system is not required to benefit from passive antibody, whether transplacental or directly administered.
Deep Thought
What if the traditional RSV seasonal hospitalization surge fails to materialize this season? It could happen. If we could get high acceptance/uptake of maternal vaccine and infant nirsevimab, RSV season could resemble the dramatic drop in rotavirus disease the second year after rotavirus vaccine introduction. We could be asking ourselves — “What happened to RSV?”
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. CDC. RSV in Infants and Young Children. Respiratory Syncytial Virus Infection (RSV). June 18, 2024. https://www.cdc.gov/rsv/infants-young-children/index.html.
2. Suss RJ and Simões EAF. Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Years, 2015-2022. JAMA Netw Open. 2024;7(4):e247125. doi:10.1001/jamanetworkopen.2024.7125.
3. Winthrop ZA et al. Pediatric Respiratory Syncytial Virus Hospitalizations and Respiratory Support After the COVID-19 Pandemic. JAMA Netw Open. 2024;7(6):e2416852. doi:10.1001/jamanetworkopen.2024.16852.
4. Petros BA et al. Increased Pediatric RSV Case Counts Following the Emergence of SARS-CoV-2 Are Attributable to Increased Testing. medRxiv [Preprint]. 2024 Feb 12:2024.02.06.24302387. doi: 10.1101/2024.02.06.24302387.
5. Rates of Laboratory-Confirmed RSV, COVID-19, and Flu Hospitalizations from the RESP-NET Surveillance Systems. Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/Rates-of-Laboratory-Confirmed-RSV-COVID-19-and-Flu/kvib-3txy/about_data.
6. CDC. Evaluating the 2023-2024 Respiratory Disease Season Outlook. CFA: Qualitative Assessments. August 14, 2024. https://www.cdc.gov/cfa-qualitative-assessments/php/data-research/2023-2024-season-outlook-retro.html.
7. Health Alert Network (HAN). Limited Availability of Nirsevimab in the United States—Interim CDC Recommendations to Protect Infants from Respiratory Syncytial Virus (RSV) during the 2023–2024 Respiratory Virus Season. October 23, 2023. https://emergency.cdc.gov/han/2023/han00499.asp.
8. CDC. Information for the 2024-2025 Flu Season. Centers for Disease Control and Prevention. March 14, 2024. https://www.cdc.gov/flu/season/faq-flu-season-2024-2025.htm.
9. Kampmann B et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023 Apr 20;388(16):1451-1464. doi: 10.1056/NEJMoa2216480.
10. Moline HL. Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus–Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season — New Vaccine Surveillance Network, October 2023–February 2024. MMWR Morb Mortal Wkly Rep. 2024;73. doi: 10.15585/mmwr.mm7309a4.
For children younger than 5 years old, RSV is the main drive — approximately 2,000,000 outpatient/ED visits and about 75,000 hospitalizations annually. RSV disease ranges from upper respiratory tract infections, eg, in older children and healthy adults, to more severe lower tract disease in young children and the elderly. Premature infants and high-risk groups are particularly prone to severe disease.1 Up to 300 pediatric RSV deaths occur yearly. “Normal” RSV seasons start in mid-November, peak in late December-January, and end after April. Note: More drawn out seasons occur in southern latitudes, eg Texas or Florida. But lately RSV seasons have been anything but normal.
2015-2016 to 2022-2023
RSV data from the Pediatric Health Information System (PHIS), collected at over 49 US children’s hospitals during 2015 to early 2023, show how crazy RSV seasons have been lately.2 The involved months, intensity, and duration of four prepandemic seasons were pretty “normal” (Figure 1). The 2019-2020 season started normally, peaked in January 2020, and was slowing as expected by February. But when SARS-Cov-2 restrictions kicked in during mid-March, RSV detections tanked to almost nothing (ditto other respiratory viruses). A near 14-month RSV hiatus meant that the 2020-2021 RSV season never materialized. However, RSV was not done with us in 2021. It rebounded in May with weekly hospitalizations peaking in late July; this “rebound season” lasted 9 months, not dropping to baseline until February 2022 (Figure 1).
I guess we should have expected a post-pandemic “disturbance in the Force,” as Yoda once said; but I sure didn’t see a prolonged summer/fall/early winter RSV season coming. It was like two “normal” seasons mashed up into one late-but-long season. Not to be outdone, the 2022-2023 RSV season started early (September) and hospitalizations skyrocketed to peak in November at over twice the peak number from any year since 2015, overloading hospitals (influenza and SARS-Cov-2 seasons were co-circulating). The season terminated early though (March 2023).
Okay, so RSV seasonality/intensity were weird post pandemic, but was anything else different? Some 2021-2023 data suggest more RSV disease in older children, rather than the usual younger than 18 month-olds going through their first winter.3 More medically attended RSV in older ages (2-4 years of life) may have been due to the pandemic year without RSV circulation distorting herd immunity, ie older children remained RSV naive. Other data suggest the apparent increase was really just more frequent multiplex viral testing in older children triggered by SARS-CoV-2 co-circulation.4 More data are needed to decide.
CDC 2023-2024 RESP-NET data
The 2023-2024 winter surge (Figure 2), as measured by RESP-NET’s cumulative RSV, influenza and SARS-CoV-2 hospitalization rates for 0- to 5-year-olds,5 shows that all three viruses’ seasonal months were normal-ish: late October 2023 start, late December-early January peak, and mid-May 2024 return to baseline. RSV season was approximately 22% less severe by area-under-the-curve calculations compared with 2022-2023, but still worse than prepandemic years.6 
One wonders if the 2022-2023 RSV season might have been worse but for use of the limited supply of nirsevimab.7
Viral Parade
Now we ready ourselves for the 2024-2025 respiratory surge, wondering what nature has in store for us. Will the usual “respiratory virus parade” occur? Will rhinovirus and parainfluenza prevalence bump after a few weeks of schools being in session, adding to the now-usual summer/fall SARS-CoV-2 surge? Note: Twenty-seven states as of Aug. 16 had high SARS-CoV-2 detection in wastewater. Will RSV and influenza start sometime in October/November, peak in January (along with rising SARS-CoV2 activity), followed by a second parainfluenza bump as SARS-CoV-2, influenza, and RSV drop off in April/May? Further, will RSV and influenza seasons be more or less severe than the last 2 years?
Prediction
The overall 2024-2025 respiratory season will be less severe than the past 2 years and hopefully than recent prepandemic years. What is the blueprint for a milder season? First, herd immunity to non-RSV and non-influenza viruses (parainfluenza, rhinovirus, metapneumovirus, adenovirus) in older children should be normalized after 2 years back to usual social activity. So, I expect no mega-seasons from them. The emerging SARS-CoV-2 virus (LB.1) is immunologically close to its recent still-circulating ancestors (KP.2, KP.2.3, KP.3 and KP.3.1.1), so existing SARS-CoV2 herd immunity along with recommended booster vaccine uptake should keep the lid on SARS-CoV2.
Influenza Could Be the Bad News
Which type will dominate? Will a drift/shift occur or vaccine-mismatch reduce vaccine effectiveness? Can we get at least half the population influenza vaccinated, given the vaccine fatigue permeating the US population? The influenza season now underway in the Southern Hemisphere usually helps us predict our season. The Australian May-August 2024 experience (still on an upward trajectory for severity in mid-August) saw no drift/shift or vaccine mismatch. However, this 2024 season has been as severe as 2022 (their worst in a decade). That said, more than 95% has been type A (mostly H1N1 but H3N2 increased in July). So, if our overall 2024-2025 respiratory season is not milder, influenza is the most likely culprit. To reduce chances of influenza being the fly-in-the-ointment, we need to be particularly proactive with seasonal influenza vaccine which is back to the traditional trivalent formulation (one H1N1, one H3N2, and one B type).8 All of this could go out the window if avian influenza becomes more transmissible, but that seems unlikely at present.
Mild RSV Season?
RSV season should be blunted because of the increased use of both the remarkably effective CDC-recommended maternal RSV vaccine9 (one dose during pregnancy weeks 32 through 36, administered September through January) and of nirsevimab (up to 90% reduction in hospitalizations and ED visits).10 (See Figure 3.) 
I also expect residual disease to occur mostly in younger than 18 month-olds (the “normal” aged population experiencing their first winter), who received no passive immunity (mother RSV unvaccinated and child did not receive nirsevimab). Some disease will still occur in high-risk infants/children. However, unlike active vaccination strategies, a competent immune system is not required to benefit from passive antibody, whether transplacental or directly administered.
Deep Thought
What if the traditional RSV seasonal hospitalization surge fails to materialize this season? It could happen. If we could get high acceptance/uptake of maternal vaccine and infant nirsevimab, RSV season could resemble the dramatic drop in rotavirus disease the second year after rotavirus vaccine introduction. We could be asking ourselves — “What happened to RSV?”
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. CDC. RSV in Infants and Young Children. Respiratory Syncytial Virus Infection (RSV). June 18, 2024. https://www.cdc.gov/rsv/infants-young-children/index.html.
2. Suss RJ and Simões EAF. Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Years, 2015-2022. JAMA Netw Open. 2024;7(4):e247125. doi:10.1001/jamanetworkopen.2024.7125.
3. Winthrop ZA et al. Pediatric Respiratory Syncytial Virus Hospitalizations and Respiratory Support After the COVID-19 Pandemic. JAMA Netw Open. 2024;7(6):e2416852. doi:10.1001/jamanetworkopen.2024.16852.
4. Petros BA et al. Increased Pediatric RSV Case Counts Following the Emergence of SARS-CoV-2 Are Attributable to Increased Testing. medRxiv [Preprint]. 2024 Feb 12:2024.02.06.24302387. doi: 10.1101/2024.02.06.24302387.
5. Rates of Laboratory-Confirmed RSV, COVID-19, and Flu Hospitalizations from the RESP-NET Surveillance Systems. Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/Rates-of-Laboratory-Confirmed-RSV-COVID-19-and-Flu/kvib-3txy/about_data.
6. CDC. Evaluating the 2023-2024 Respiratory Disease Season Outlook. CFA: Qualitative Assessments. August 14, 2024. https://www.cdc.gov/cfa-qualitative-assessments/php/data-research/2023-2024-season-outlook-retro.html.
7. Health Alert Network (HAN). Limited Availability of Nirsevimab in the United States—Interim CDC Recommendations to Protect Infants from Respiratory Syncytial Virus (RSV) during the 2023–2024 Respiratory Virus Season. October 23, 2023. https://emergency.cdc.gov/han/2023/han00499.asp.
8. CDC. Information for the 2024-2025 Flu Season. Centers for Disease Control and Prevention. March 14, 2024. https://www.cdc.gov/flu/season/faq-flu-season-2024-2025.htm.
9. Kampmann B et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023 Apr 20;388(16):1451-1464. doi: 10.1056/NEJMoa2216480.
10. Moline HL. Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus–Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season — New Vaccine Surveillance Network, October 2023–February 2024. MMWR Morb Mortal Wkly Rep. 2024;73. doi: 10.15585/mmwr.mm7309a4.
For children younger than 5 years old, RSV is the main drive — approximately 2,000,000 outpatient/ED visits and about 75,000 hospitalizations annually. RSV disease ranges from upper respiratory tract infections, eg, in older children and healthy adults, to more severe lower tract disease in young children and the elderly. Premature infants and high-risk groups are particularly prone to severe disease.1 Up to 300 pediatric RSV deaths occur yearly. “Normal” RSV seasons start in mid-November, peak in late December-January, and end after April. Note: More drawn out seasons occur in southern latitudes, eg Texas or Florida. But lately RSV seasons have been anything but normal.
2015-2016 to 2022-2023
RSV data from the Pediatric Health Information System (PHIS), collected at over 49 US children’s hospitals during 2015 to early 2023, show how crazy RSV seasons have been lately.2 The involved months, intensity, and duration of four prepandemic seasons were pretty “normal” (Figure 1). The 2019-2020 season started normally, peaked in January 2020, and was slowing as expected by February. But when SARS-Cov-2 restrictions kicked in during mid-March, RSV detections tanked to almost nothing (ditto other respiratory viruses). A near 14-month RSV hiatus meant that the 2020-2021 RSV season never materialized. However, RSV was not done with us in 2021. It rebounded in May with weekly hospitalizations peaking in late July; this “rebound season” lasted 9 months, not dropping to baseline until February 2022 (Figure 1).
I guess we should have expected a post-pandemic “disturbance in the Force,” as Yoda once said; but I sure didn’t see a prolonged summer/fall/early winter RSV season coming. It was like two “normal” seasons mashed up into one late-but-long season. Not to be outdone, the 2022-2023 RSV season started early (September) and hospitalizations skyrocketed to peak in November at over twice the peak number from any year since 2015, overloading hospitals (influenza and SARS-Cov-2 seasons were co-circulating). The season terminated early though (March 2023).
Okay, so RSV seasonality/intensity were weird post pandemic, but was anything else different? Some 2021-2023 data suggest more RSV disease in older children, rather than the usual younger than 18 month-olds going through their first winter.3 More medically attended RSV in older ages (2-4 years of life) may have been due to the pandemic year without RSV circulation distorting herd immunity, ie older children remained RSV naive. Other data suggest the apparent increase was really just more frequent multiplex viral testing in older children triggered by SARS-CoV-2 co-circulation.4 More data are needed to decide.
CDC 2023-2024 RESP-NET data
The 2023-2024 winter surge (Figure 2), as measured by RESP-NET’s cumulative RSV, influenza and SARS-CoV-2 hospitalization rates for 0- to 5-year-olds,5 shows that all three viruses’ seasonal months were normal-ish: late October 2023 start, late December-early January peak, and mid-May 2024 return to baseline. RSV season was approximately 22% less severe by area-under-the-curve calculations compared with 2022-2023, but still worse than prepandemic years.6 
One wonders if the 2022-2023 RSV season might have been worse but for use of the limited supply of nirsevimab.7
Viral Parade
Now we ready ourselves for the 2024-2025 respiratory surge, wondering what nature has in store for us. Will the usual “respiratory virus parade” occur? Will rhinovirus and parainfluenza prevalence bump after a few weeks of schools being in session, adding to the now-usual summer/fall SARS-CoV-2 surge? Note: Twenty-seven states as of Aug. 16 had high SARS-CoV-2 detection in wastewater. Will RSV and influenza start sometime in October/November, peak in January (along with rising SARS-CoV2 activity), followed by a second parainfluenza bump as SARS-CoV-2, influenza, and RSV drop off in April/May? Further, will RSV and influenza seasons be more or less severe than the last 2 years?
Prediction
The overall 2024-2025 respiratory season will be less severe than the past 2 years and hopefully than recent prepandemic years. What is the blueprint for a milder season? First, herd immunity to non-RSV and non-influenza viruses (parainfluenza, rhinovirus, metapneumovirus, adenovirus) in older children should be normalized after 2 years back to usual social activity. So, I expect no mega-seasons from them. The emerging SARS-CoV-2 virus (LB.1) is immunologically close to its recent still-circulating ancestors (KP.2, KP.2.3, KP.3 and KP.3.1.1), so existing SARS-CoV2 herd immunity along with recommended booster vaccine uptake should keep the lid on SARS-CoV2.
Influenza Could Be the Bad News
Which type will dominate? Will a drift/shift occur or vaccine-mismatch reduce vaccine effectiveness? Can we get at least half the population influenza vaccinated, given the vaccine fatigue permeating the US population? The influenza season now underway in the Southern Hemisphere usually helps us predict our season. The Australian May-August 2024 experience (still on an upward trajectory for severity in mid-August) saw no drift/shift or vaccine mismatch. However, this 2024 season has been as severe as 2022 (their worst in a decade). That said, more than 95% has been type A (mostly H1N1 but H3N2 increased in July). So, if our overall 2024-2025 respiratory season is not milder, influenza is the most likely culprit. To reduce chances of influenza being the fly-in-the-ointment, we need to be particularly proactive with seasonal influenza vaccine which is back to the traditional trivalent formulation (one H1N1, one H3N2, and one B type).8 All of this could go out the window if avian influenza becomes more transmissible, but that seems unlikely at present.
Mild RSV Season?
RSV season should be blunted because of the increased use of both the remarkably effective CDC-recommended maternal RSV vaccine9 (one dose during pregnancy weeks 32 through 36, administered September through January) and of nirsevimab (up to 90% reduction in hospitalizations and ED visits).10 (See Figure 3.) 
I also expect residual disease to occur mostly in younger than 18 month-olds (the “normal” aged population experiencing their first winter), who received no passive immunity (mother RSV unvaccinated and child did not receive nirsevimab). Some disease will still occur in high-risk infants/children. However, unlike active vaccination strategies, a competent immune system is not required to benefit from passive antibody, whether transplacental or directly administered.
Deep Thought
What if the traditional RSV seasonal hospitalization surge fails to materialize this season? It could happen. If we could get high acceptance/uptake of maternal vaccine and infant nirsevimab, RSV season could resemble the dramatic drop in rotavirus disease the second year after rotavirus vaccine introduction. We could be asking ourselves — “What happened to RSV?”
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. CDC. RSV in Infants and Young Children. Respiratory Syncytial Virus Infection (RSV). June 18, 2024. https://www.cdc.gov/rsv/infants-young-children/index.html.
2. Suss RJ and Simões EAF. Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Respiratory Syncytial Virus Hospital-Based Burden of Disease in Children Younger Than 5 Years, 2015-2022. JAMA Netw Open. 2024;7(4):e247125. doi:10.1001/jamanetworkopen.2024.7125.
3. Winthrop ZA et al. Pediatric Respiratory Syncytial Virus Hospitalizations and Respiratory Support After the COVID-19 Pandemic. JAMA Netw Open. 2024;7(6):e2416852. doi:10.1001/jamanetworkopen.2024.16852.
4. Petros BA et al. Increased Pediatric RSV Case Counts Following the Emergence of SARS-CoV-2 Are Attributable to Increased Testing. medRxiv [Preprint]. 2024 Feb 12:2024.02.06.24302387. doi: 10.1101/2024.02.06.24302387.
5. Rates of Laboratory-Confirmed RSV, COVID-19, and Flu Hospitalizations from the RESP-NET Surveillance Systems. Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/Rates-of-Laboratory-Confirmed-RSV-COVID-19-and-Flu/kvib-3txy/about_data.
6. CDC. Evaluating the 2023-2024 Respiratory Disease Season Outlook. CFA: Qualitative Assessments. August 14, 2024. https://www.cdc.gov/cfa-qualitative-assessments/php/data-research/2023-2024-season-outlook-retro.html.
7. Health Alert Network (HAN). Limited Availability of Nirsevimab in the United States—Interim CDC Recommendations to Protect Infants from Respiratory Syncytial Virus (RSV) during the 2023–2024 Respiratory Virus Season. October 23, 2023. https://emergency.cdc.gov/han/2023/han00499.asp.
8. CDC. Information for the 2024-2025 Flu Season. Centers for Disease Control and Prevention. March 14, 2024. https://www.cdc.gov/flu/season/faq-flu-season-2024-2025.htm.
9. Kampmann B et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N Engl J Med. 2023 Apr 20;388(16):1451-1464. doi: 10.1056/NEJMoa2216480.
10. Moline HL. Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus–Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season — New Vaccine Surveillance Network, October 2023–February 2024. MMWR Morb Mortal Wkly Rep. 2024;73. doi: 10.15585/mmwr.mm7309a4.