User login
A pandemic silver lining? Dramatic drop in teen drug use
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.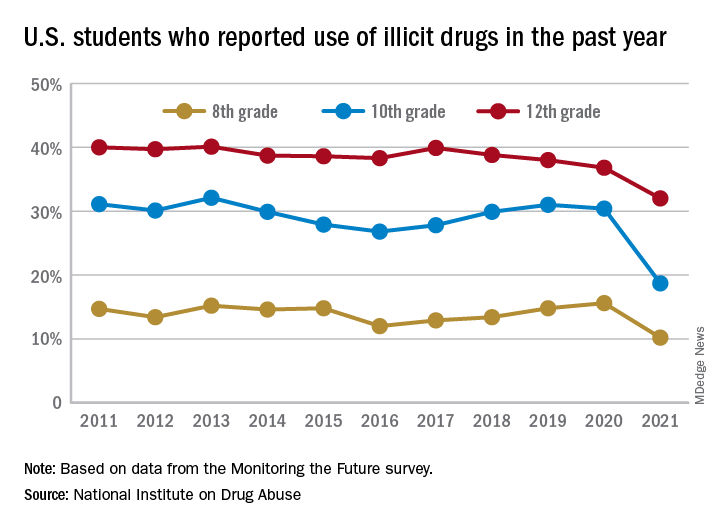
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Not All Pulmonary Nodules in Smokers are Lung Cancer
Identification of pulmonary nodules in older adults who smoke immediately brings concern for malignancy in the mind of clinicians. This is particularly the case in patients with significant smoking history. According to the National Cancer Institute in 2019, 12.9% of all new cancer cases were lung cancers.1 Screening for lung cancer, especially in patients with increased risk from smoking, is imperative to early detection and treatment. However, 20% of patients will be overdiagnosed by lung cancer-screening techniques.2 The rate of malignancy noted on a patient’s first screening computed tomography (CT) scan was between 3.7% and 5.5%.3
Rheumatoid arthritis (RA) is an autoimmune inflammatory condition that mainly affects the joints. Extraarticular manifestations can arise in various locations throughout the body, however. These manifestations are commonly observed in the skin, heart, and lungs.4 Prevalence of pulmonary rheumatoid nodules ranges from < 0.4% in radiologic studies to 32% in lung biopsies of patients with RA and nodules.5
Furthermore, there is a strong association between the risk of rheumatoid nodules in patients with positive serum rheumatoid factor (RF) and smoking history.6 Solitary pulmonary nodules in patients with RA can coexist with bronchogenic carcinoma, making their diagnosis more important.7
Case Presentation
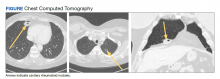
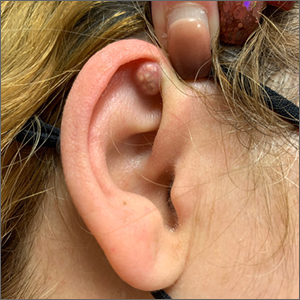
A 54-year-old woman with a 30 pack-year smoking history and history of RA initially presented to the emergency department for cough and dyspnea for 5-day duration. Her initial diagnosis was bronchitis based on presenting symptom profile. A chest CT demonstrated 3 cavitary pulmonary nodules, 1 measuring 2.4 x 2.0 cm in the right middle lobe, and 2 additional nodules, measuring 1.8 x 1.4 and 1.5 x 1.4 in the left upper lobe (Figure). She had no improvement of symptoms after a 7-day course of doxycycline. The patient was taking methotrexate 15 mg weekly and golimumab 50 mg subcutaneously every 4 weeks as treatment for RA, prescribed by her rheumatologist.
Pulmonology was consulted and a positron emission tomography-CT (PET-CT) confirmed several cavitary pulmonary nodules involving both lungs with no suspicious fluorodeoxyglucose (FDG) uptake. The largest lesion was in the right middle lobe with FDG uptake of 1.9. Additional nodules were found in the left upper lobe, measuring 1.8 x 1.4 cm with FDG of 4.01, and in the left lung apex, measuring 1.5 x 1.4 cm with uptake of 3.53. CTguided percutaneous fine needle aspiration (PFNA) of the right middle lobe lung nodule demonstrated granuloma with central inflammatory debris. Grocott methenamine silver (GMS) stain was negative for fungal organism, acid-fast bacteria (AFB) stain was negative for acid-fast bacilli, and CD20 and CD3 immunostaining demonstrated mixed B- and T-cell populations. There was no evidence of atypia or malignancy. The biopsy demonstrated granuloma with central inflammatory debris on a background of densely fibrotic tissue and lympho-plasmatic inflammation. This finding confirmed the diagnosis of RA with pulmonary involvement.
Outpatient follow-up was established with a pulmonologist and rheumatologist. Methotrexate 15 mg weekly and golimumab subcutaneously 50 mg every 4 weeks were prescribed for the patient. The nodules are being monitored based on Fleischer guidelines with CT imaging 3 to 6 months following initial presentation. Further imaging will be considered at 18 to 24 months as well to further assess stability of the nodules and monitor for changes in size, shape, and necrosis. The patient also was encouraged to quit smoking. Her clinical course since the diagnosis has been stable.
Discussion
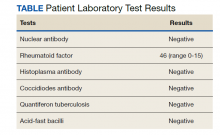
The differential diagnosis for new multiple pulmonary nodules on imaging studies is broad and includes infectious processes, such as tuberculosis, as well as other mycobacterial, fungal, and bacterial infections. Noninfectious causes of lung disease are an even broader category of consideration. Noninfectious pulmonary nodules differential includes sarcoidosis, granulomatous with polyangiitis, hypersensitivity pneumonitis, methotrexate drug reaction, pulmonary manifestations of systemic conditions, such as RA chronic granulomatous disease and malignancy.8 Bronchogenic carcinoma was suspected in this patient due to her smoking history. Squamous cell carcinoma was also considered as the lesion was cavitary. AFB and GMS stains were negative for fungi. Langerhans cell histiocytosis were considered but ruled out as these lesions contain larger numbers of eosinophils than described in the pathology report. Histoplasma and coccidiosis laboratory tests were obtained as the patient lived in a region endemic to both these fungi but were negative (Table). A diagnosis of rheumatoid nodule was made based on the clinical setting, typical radiographic, histopathology features, and negative cultures.
This case is unique due to the quality and location of the rheumatoid nodules within the lungs. Pulmonary manifestations of RA are usually subcutaneous or subpleural, solid, and peripherally located.9 This patient’s nodules were necrobiotic and located within the lung parenchyma. There was significant cavitation. These factors are atypical features of pulmonary RA.
Pulmonary RA can have many associated symptoms and remains an important factor in patient mortality. Estimates demonstrate that 10 to 20% of RA-related deaths are secondary to pulmonary manifestations.10 There are a wide array of symptoms and presentations to be aware of clinically. These symptoms are often nondescript, widely sensitive to many disease processes, and nonspecific to pulmonary RA. These symptoms include dyspnea, wheezing, and nonproductive cough.10 Bronchiectasis is a common symptom as well as small airway obstruction.10 Consolidated necrobiotic lesions are present in up to 20% of pulmonary RA cases.10 Generally these lesions are asymptomatic but can also be associated with pneumothorax, hemoptysis, and airway obstruction.10 Awareness of these symptoms is important for diagnosis and monitoring clinical improvement in patients.
Further workup is necessary to differentiate malignancy-related pulmonary nodules and other causes; if the index of suspicion is high for malignancy as in our case, the workup should be more aggressive. Biopsy is mandatory in such cases to rule out infections and malignancy, as it is highly sensitive and specific. The main problem hindering management is when a clinician fails to include this in their differential diagnosis. This further elucidates the importance of awareness of this diagnosis. Suspicious lesions in a proper clinical setting should be followed up by imaging studies and confirmatory histopathological diagnosis. Typical follow-up is 3 months after initial presentation to assess stability and possibly 18 to 24 months as well based on Fleischer guidelines.
Various treatment modalities have been tried as per literature, including tocilizumab and rituximab. 11,12 Our patient is currently being treated with golimumab based on outpatient rheumatologist recommendations.
Conclusions
This case demonstrates the importance of a careful workup to narrow a broad differential. Medical diagnosis of pulmonary nodules requires an in-depth workup, including clinical evaluation, laboratory and pulmonary functions tests, as well as various imaging studies.
1. Lung and Bronchus Cancer - Cancer Stat Facts. SEER. Accessed February 2, 2020. https://seer.cancer.gov /statfacts/html/lungb.html
2. Shaughnessy AF. One in Five Patients Overdiagnosed with Lung Cancer Screening. Am Fam Physician. 2014 Jul 15;90(2):112.
3. McWilliams A, Tammemagi MC, Mayo JR, et al. Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med. 2013;369;910-919. doi:10.1056/NEJMoa1214726
4. Stamp LK, Cleland LG. Rheumatoid arthritis. In: Thompson LU, Ward WE, eds. Optimizing Women’s Health through Nutrition. CRC Press; 2008; 279-320.
5. Yousem SA, Colby TV, Carrington CB. Lung biopsy in rheumatoid arthritis. Am Rev Respir Dis. 1985;131(5):770-777. doi:10.1164/arrd.1985.131.5.770
6. Nyhäll-Wåhlin BM, Jacobsson LT, Petersson IF, Turesson C; BARFOT study group. Smoking is a strong risk factor for rheumatoid nodules in early rheumatoid arthritis. Ann Rheum Dis. 2006;65(5):601-606. doi:10.1136/ard.2005.039172
7. Shenberger KN, Schned AR, Taylor TH. Rheumatoid disease and bronchogenic carcinoma—case report and review of the literature. J Rheumatol. 1984;11:226–228.
8. Mukhopadhyay S, Wilcox BE, Myers JL, et al. Pulmonary necrotizing granulomas of unknown cause clinical and pathologic analysis of 131 patients with completely resected nodules. Chest. 2013;144(3):813-824. doi:10.1378/chest.12-2113
9. Ohshimo S, Guzman J, Costabel U, Bonella F. Differential diagnosis of granulomatous lung disease: clues and pitfalls: Number 4 in the Series “Pathology for the clinician.” Edited by Peter Dorfmüller and Alberto Cavazza. Eur Respir Rev. 2017;26(145):170012. Published 2017 Aug 9. doi:10.1183/16000617.0012-2017
10. Brown KK. Rheumatoid lung disease. Proc Am Thorac Soc. 2007;4(5):443-448. doi:10.1513/pats.200703-045MS
11. Braun MG, Wagener P. Regression von peripheren und pulmonalen Rheumaknoten unter Rituximab-Therapie [Regression of peripheral and pulmonary rheumatoid nodules under therapy with rituximab]. Z Rheumatol. 2013;72(2):166-171. doi:10.1007/s00393-012-1054-0
12. Andres M, Vela P, Romera C. Marked improvement of lung rheumatoid nodules after treatment with tocilizumab. Rheumatology (Oxford). 2012;51(6):1132-1134. doi:10.1093/rheumatology/ker455
Identification of pulmonary nodules in older adults who smoke immediately brings concern for malignancy in the mind of clinicians. This is particularly the case in patients with significant smoking history. According to the National Cancer Institute in 2019, 12.9% of all new cancer cases were lung cancers.1 Screening for lung cancer, especially in patients with increased risk from smoking, is imperative to early detection and treatment. However, 20% of patients will be overdiagnosed by lung cancer-screening techniques.2 The rate of malignancy noted on a patient’s first screening computed tomography (CT) scan was between 3.7% and 5.5%.3
Rheumatoid arthritis (RA) is an autoimmune inflammatory condition that mainly affects the joints. Extraarticular manifestations can arise in various locations throughout the body, however. These manifestations are commonly observed in the skin, heart, and lungs.4 Prevalence of pulmonary rheumatoid nodules ranges from < 0.4% in radiologic studies to 32% in lung biopsies of patients with RA and nodules.5
Furthermore, there is a strong association between the risk of rheumatoid nodules in patients with positive serum rheumatoid factor (RF) and smoking history.6 Solitary pulmonary nodules in patients with RA can coexist with bronchogenic carcinoma, making their diagnosis more important.7
Case Presentation
A 54-year-old woman with a 30 pack-year smoking history and history of RA initially presented to the emergency department for cough and dyspnea for 5-day duration. Her initial diagnosis was bronchitis based on presenting symptom profile. A chest CT demonstrated 3 cavitary pulmonary nodules, 1 measuring 2.4 x 2.0 cm in the right middle lobe, and 2 additional nodules, measuring 1.8 x 1.4 and 1.5 x 1.4 in the left upper lobe (Figure). She had no improvement of symptoms after a 7-day course of doxycycline. The patient was taking methotrexate 15 mg weekly and golimumab 50 mg subcutaneously every 4 weeks as treatment for RA, prescribed by her rheumatologist.
Pulmonology was consulted and a positron emission tomography-CT (PET-CT) confirmed several cavitary pulmonary nodules involving both lungs with no suspicious fluorodeoxyglucose (FDG) uptake. The largest lesion was in the right middle lobe with FDG uptake of 1.9. Additional nodules were found in the left upper lobe, measuring 1.8 x 1.4 cm with FDG of 4.01, and in the left lung apex, measuring 1.5 x 1.4 cm with uptake of 3.53. CTguided percutaneous fine needle aspiration (PFNA) of the right middle lobe lung nodule demonstrated granuloma with central inflammatory debris. Grocott methenamine silver (GMS) stain was negative for fungal organism, acid-fast bacteria (AFB) stain was negative for acid-fast bacilli, and CD20 and CD3 immunostaining demonstrated mixed B- and T-cell populations. There was no evidence of atypia or malignancy. The biopsy demonstrated granuloma with central inflammatory debris on a background of densely fibrotic tissue and lympho-plasmatic inflammation. This finding confirmed the diagnosis of RA with pulmonary involvement.
Outpatient follow-up was established with a pulmonologist and rheumatologist. Methotrexate 15 mg weekly and golimumab subcutaneously 50 mg every 4 weeks were prescribed for the patient. The nodules are being monitored based on Fleischer guidelines with CT imaging 3 to 6 months following initial presentation. Further imaging will be considered at 18 to 24 months as well to further assess stability of the nodules and monitor for changes in size, shape, and necrosis. The patient also was encouraged to quit smoking. Her clinical course since the diagnosis has been stable.
Discussion
The differential diagnosis for new multiple pulmonary nodules on imaging studies is broad and includes infectious processes, such as tuberculosis, as well as other mycobacterial, fungal, and bacterial infections. Noninfectious causes of lung disease are an even broader category of consideration. Noninfectious pulmonary nodules differential includes sarcoidosis, granulomatous with polyangiitis, hypersensitivity pneumonitis, methotrexate drug reaction, pulmonary manifestations of systemic conditions, such as RA chronic granulomatous disease and malignancy.8 Bronchogenic carcinoma was suspected in this patient due to her smoking history. Squamous cell carcinoma was also considered as the lesion was cavitary. AFB and GMS stains were negative for fungi. Langerhans cell histiocytosis were considered but ruled out as these lesions contain larger numbers of eosinophils than described in the pathology report. Histoplasma and coccidiosis laboratory tests were obtained as the patient lived in a region endemic to both these fungi but were negative (Table). A diagnosis of rheumatoid nodule was made based on the clinical setting, typical radiographic, histopathology features, and negative cultures.
This case is unique due to the quality and location of the rheumatoid nodules within the lungs. Pulmonary manifestations of RA are usually subcutaneous or subpleural, solid, and peripherally located.9 This patient’s nodules were necrobiotic and located within the lung parenchyma. There was significant cavitation. These factors are atypical features of pulmonary RA.
Pulmonary RA can have many associated symptoms and remains an important factor in patient mortality. Estimates demonstrate that 10 to 20% of RA-related deaths are secondary to pulmonary manifestations.10 There are a wide array of symptoms and presentations to be aware of clinically. These symptoms are often nondescript, widely sensitive to many disease processes, and nonspecific to pulmonary RA. These symptoms include dyspnea, wheezing, and nonproductive cough.10 Bronchiectasis is a common symptom as well as small airway obstruction.10 Consolidated necrobiotic lesions are present in up to 20% of pulmonary RA cases.10 Generally these lesions are asymptomatic but can also be associated with pneumothorax, hemoptysis, and airway obstruction.10 Awareness of these symptoms is important for diagnosis and monitoring clinical improvement in patients.
Further workup is necessary to differentiate malignancy-related pulmonary nodules and other causes; if the index of suspicion is high for malignancy as in our case, the workup should be more aggressive. Biopsy is mandatory in such cases to rule out infections and malignancy, as it is highly sensitive and specific. The main problem hindering management is when a clinician fails to include this in their differential diagnosis. This further elucidates the importance of awareness of this diagnosis. Suspicious lesions in a proper clinical setting should be followed up by imaging studies and confirmatory histopathological diagnosis. Typical follow-up is 3 months after initial presentation to assess stability and possibly 18 to 24 months as well based on Fleischer guidelines.
Various treatment modalities have been tried as per literature, including tocilizumab and rituximab. 11,12 Our patient is currently being treated with golimumab based on outpatient rheumatologist recommendations.
Conclusions
This case demonstrates the importance of a careful workup to narrow a broad differential. Medical diagnosis of pulmonary nodules requires an in-depth workup, including clinical evaluation, laboratory and pulmonary functions tests, as well as various imaging studies.
Identification of pulmonary nodules in older adults who smoke immediately brings concern for malignancy in the mind of clinicians. This is particularly the case in patients with significant smoking history. According to the National Cancer Institute in 2019, 12.9% of all new cancer cases were lung cancers.1 Screening for lung cancer, especially in patients with increased risk from smoking, is imperative to early detection and treatment. However, 20% of patients will be overdiagnosed by lung cancer-screening techniques.2 The rate of malignancy noted on a patient’s first screening computed tomography (CT) scan was between 3.7% and 5.5%.3
Rheumatoid arthritis (RA) is an autoimmune inflammatory condition that mainly affects the joints. Extraarticular manifestations can arise in various locations throughout the body, however. These manifestations are commonly observed in the skin, heart, and lungs.4 Prevalence of pulmonary rheumatoid nodules ranges from < 0.4% in radiologic studies to 32% in lung biopsies of patients with RA and nodules.5
Furthermore, there is a strong association between the risk of rheumatoid nodules in patients with positive serum rheumatoid factor (RF) and smoking history.6 Solitary pulmonary nodules in patients with RA can coexist with bronchogenic carcinoma, making their diagnosis more important.7
Case Presentation
A 54-year-old woman with a 30 pack-year smoking history and history of RA initially presented to the emergency department for cough and dyspnea for 5-day duration. Her initial diagnosis was bronchitis based on presenting symptom profile. A chest CT demonstrated 3 cavitary pulmonary nodules, 1 measuring 2.4 x 2.0 cm in the right middle lobe, and 2 additional nodules, measuring 1.8 x 1.4 and 1.5 x 1.4 in the left upper lobe (Figure). She had no improvement of symptoms after a 7-day course of doxycycline. The patient was taking methotrexate 15 mg weekly and golimumab 50 mg subcutaneously every 4 weeks as treatment for RA, prescribed by her rheumatologist.
Pulmonology was consulted and a positron emission tomography-CT (PET-CT) confirmed several cavitary pulmonary nodules involving both lungs with no suspicious fluorodeoxyglucose (FDG) uptake. The largest lesion was in the right middle lobe with FDG uptake of 1.9. Additional nodules were found in the left upper lobe, measuring 1.8 x 1.4 cm with FDG of 4.01, and in the left lung apex, measuring 1.5 x 1.4 cm with uptake of 3.53. CTguided percutaneous fine needle aspiration (PFNA) of the right middle lobe lung nodule demonstrated granuloma with central inflammatory debris. Grocott methenamine silver (GMS) stain was negative for fungal organism, acid-fast bacteria (AFB) stain was negative for acid-fast bacilli, and CD20 and CD3 immunostaining demonstrated mixed B- and T-cell populations. There was no evidence of atypia or malignancy. The biopsy demonstrated granuloma with central inflammatory debris on a background of densely fibrotic tissue and lympho-plasmatic inflammation. This finding confirmed the diagnosis of RA with pulmonary involvement.
Outpatient follow-up was established with a pulmonologist and rheumatologist. Methotrexate 15 mg weekly and golimumab subcutaneously 50 mg every 4 weeks were prescribed for the patient. The nodules are being monitored based on Fleischer guidelines with CT imaging 3 to 6 months following initial presentation. Further imaging will be considered at 18 to 24 months as well to further assess stability of the nodules and monitor for changes in size, shape, and necrosis. The patient also was encouraged to quit smoking. Her clinical course since the diagnosis has been stable.
Discussion
The differential diagnosis for new multiple pulmonary nodules on imaging studies is broad and includes infectious processes, such as tuberculosis, as well as other mycobacterial, fungal, and bacterial infections. Noninfectious causes of lung disease are an even broader category of consideration. Noninfectious pulmonary nodules differential includes sarcoidosis, granulomatous with polyangiitis, hypersensitivity pneumonitis, methotrexate drug reaction, pulmonary manifestations of systemic conditions, such as RA chronic granulomatous disease and malignancy.8 Bronchogenic carcinoma was suspected in this patient due to her smoking history. Squamous cell carcinoma was also considered as the lesion was cavitary. AFB and GMS stains were negative for fungi. Langerhans cell histiocytosis were considered but ruled out as these lesions contain larger numbers of eosinophils than described in the pathology report. Histoplasma and coccidiosis laboratory tests were obtained as the patient lived in a region endemic to both these fungi but were negative (Table). A diagnosis of rheumatoid nodule was made based on the clinical setting, typical radiographic, histopathology features, and negative cultures.
This case is unique due to the quality and location of the rheumatoid nodules within the lungs. Pulmonary manifestations of RA are usually subcutaneous or subpleural, solid, and peripherally located.9 This patient’s nodules were necrobiotic and located within the lung parenchyma. There was significant cavitation. These factors are atypical features of pulmonary RA.
Pulmonary RA can have many associated symptoms and remains an important factor in patient mortality. Estimates demonstrate that 10 to 20% of RA-related deaths are secondary to pulmonary manifestations.10 There are a wide array of symptoms and presentations to be aware of clinically. These symptoms are often nondescript, widely sensitive to many disease processes, and nonspecific to pulmonary RA. These symptoms include dyspnea, wheezing, and nonproductive cough.10 Bronchiectasis is a common symptom as well as small airway obstruction.10 Consolidated necrobiotic lesions are present in up to 20% of pulmonary RA cases.10 Generally these lesions are asymptomatic but can also be associated with pneumothorax, hemoptysis, and airway obstruction.10 Awareness of these symptoms is important for diagnosis and monitoring clinical improvement in patients.
Further workup is necessary to differentiate malignancy-related pulmonary nodules and other causes; if the index of suspicion is high for malignancy as in our case, the workup should be more aggressive. Biopsy is mandatory in such cases to rule out infections and malignancy, as it is highly sensitive and specific. The main problem hindering management is when a clinician fails to include this in their differential diagnosis. This further elucidates the importance of awareness of this diagnosis. Suspicious lesions in a proper clinical setting should be followed up by imaging studies and confirmatory histopathological diagnosis. Typical follow-up is 3 months after initial presentation to assess stability and possibly 18 to 24 months as well based on Fleischer guidelines.
Various treatment modalities have been tried as per literature, including tocilizumab and rituximab. 11,12 Our patient is currently being treated with golimumab based on outpatient rheumatologist recommendations.
Conclusions
This case demonstrates the importance of a careful workup to narrow a broad differential. Medical diagnosis of pulmonary nodules requires an in-depth workup, including clinical evaluation, laboratory and pulmonary functions tests, as well as various imaging studies.
1. Lung and Bronchus Cancer - Cancer Stat Facts. SEER. Accessed February 2, 2020. https://seer.cancer.gov /statfacts/html/lungb.html
2. Shaughnessy AF. One in Five Patients Overdiagnosed with Lung Cancer Screening. Am Fam Physician. 2014 Jul 15;90(2):112.
3. McWilliams A, Tammemagi MC, Mayo JR, et al. Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med. 2013;369;910-919. doi:10.1056/NEJMoa1214726
4. Stamp LK, Cleland LG. Rheumatoid arthritis. In: Thompson LU, Ward WE, eds. Optimizing Women’s Health through Nutrition. CRC Press; 2008; 279-320.
5. Yousem SA, Colby TV, Carrington CB. Lung biopsy in rheumatoid arthritis. Am Rev Respir Dis. 1985;131(5):770-777. doi:10.1164/arrd.1985.131.5.770
6. Nyhäll-Wåhlin BM, Jacobsson LT, Petersson IF, Turesson C; BARFOT study group. Smoking is a strong risk factor for rheumatoid nodules in early rheumatoid arthritis. Ann Rheum Dis. 2006;65(5):601-606. doi:10.1136/ard.2005.039172
7. Shenberger KN, Schned AR, Taylor TH. Rheumatoid disease and bronchogenic carcinoma—case report and review of the literature. J Rheumatol. 1984;11:226–228.
8. Mukhopadhyay S, Wilcox BE, Myers JL, et al. Pulmonary necrotizing granulomas of unknown cause clinical and pathologic analysis of 131 patients with completely resected nodules. Chest. 2013;144(3):813-824. doi:10.1378/chest.12-2113
9. Ohshimo S, Guzman J, Costabel U, Bonella F. Differential diagnosis of granulomatous lung disease: clues and pitfalls: Number 4 in the Series “Pathology for the clinician.” Edited by Peter Dorfmüller and Alberto Cavazza. Eur Respir Rev. 2017;26(145):170012. Published 2017 Aug 9. doi:10.1183/16000617.0012-2017
10. Brown KK. Rheumatoid lung disease. Proc Am Thorac Soc. 2007;4(5):443-448. doi:10.1513/pats.200703-045MS
11. Braun MG, Wagener P. Regression von peripheren und pulmonalen Rheumaknoten unter Rituximab-Therapie [Regression of peripheral and pulmonary rheumatoid nodules under therapy with rituximab]. Z Rheumatol. 2013;72(2):166-171. doi:10.1007/s00393-012-1054-0
12. Andres M, Vela P, Romera C. Marked improvement of lung rheumatoid nodules after treatment with tocilizumab. Rheumatology (Oxford). 2012;51(6):1132-1134. doi:10.1093/rheumatology/ker455
1. Lung and Bronchus Cancer - Cancer Stat Facts. SEER. Accessed February 2, 2020. https://seer.cancer.gov /statfacts/html/lungb.html
2. Shaughnessy AF. One in Five Patients Overdiagnosed with Lung Cancer Screening. Am Fam Physician. 2014 Jul 15;90(2):112.
3. McWilliams A, Tammemagi MC, Mayo JR, et al. Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med. 2013;369;910-919. doi:10.1056/NEJMoa1214726
4. Stamp LK, Cleland LG. Rheumatoid arthritis. In: Thompson LU, Ward WE, eds. Optimizing Women’s Health through Nutrition. CRC Press; 2008; 279-320.
5. Yousem SA, Colby TV, Carrington CB. Lung biopsy in rheumatoid arthritis. Am Rev Respir Dis. 1985;131(5):770-777. doi:10.1164/arrd.1985.131.5.770
6. Nyhäll-Wåhlin BM, Jacobsson LT, Petersson IF, Turesson C; BARFOT study group. Smoking is a strong risk factor for rheumatoid nodules in early rheumatoid arthritis. Ann Rheum Dis. 2006;65(5):601-606. doi:10.1136/ard.2005.039172
7. Shenberger KN, Schned AR, Taylor TH. Rheumatoid disease and bronchogenic carcinoma—case report and review of the literature. J Rheumatol. 1984;11:226–228.
8. Mukhopadhyay S, Wilcox BE, Myers JL, et al. Pulmonary necrotizing granulomas of unknown cause clinical and pathologic analysis of 131 patients with completely resected nodules. Chest. 2013;144(3):813-824. doi:10.1378/chest.12-2113
9. Ohshimo S, Guzman J, Costabel U, Bonella F. Differential diagnosis of granulomatous lung disease: clues and pitfalls: Number 4 in the Series “Pathology for the clinician.” Edited by Peter Dorfmüller and Alberto Cavazza. Eur Respir Rev. 2017;26(145):170012. Published 2017 Aug 9. doi:10.1183/16000617.0012-2017
10. Brown KK. Rheumatoid lung disease. Proc Am Thorac Soc. 2007;4(5):443-448. doi:10.1513/pats.200703-045MS
11. Braun MG, Wagener P. Regression von peripheren und pulmonalen Rheumaknoten unter Rituximab-Therapie [Regression of peripheral and pulmonary rheumatoid nodules under therapy with rituximab]. Z Rheumatol. 2013;72(2):166-171. doi:10.1007/s00393-012-1054-0
12. Andres M, Vela P, Romera C. Marked improvement of lung rheumatoid nodules after treatment with tocilizumab. Rheumatology (Oxford). 2012;51(6):1132-1134. doi:10.1093/rheumatology/ker455
Medicare insulin negotiations seen saving $17 billion
Medicare could have saved more than $16.7 billion on three kinds of insulin products from 2011 to 2017 if it had secured the same discounts other federal health programs get through negotiations, House Democrats argue in a new report.
On Dec. 10, Democrats on the House Committee on Oversight and Reform released a final majority staff report, which they say is the culmination of an almost 3-year investigation into pharmaceutical pricing and business practices. The report draws from 1.5 million pages of internal company documents, the committee says.
Documents from insulin makers Eli Lilly, Novo Nordisk, and Sanofi indicate these firms “raised their prices in lockstep in order to maintain ‘pricing parity’,” with senior executives encouraging the practice, the committee staff writes in the report.
“In a discussion among Novo Nordisk employees about an Eli Lilly price increase for a different diabetes product on Dec. 24, 2015, a Novo Nordisk pricing analyst remarked, ‘[M]aybe Sanofi will wait until tomorrow morning to announce their price increase ... that’s all I want for Christmas,’” the report states.
House Democrats are seeking to use the report findings to aid their Senate colleagues’ attempt to pass the sweeping Build Back Better bill, which includes many provisions addressing drug costs.
It’s still unclear when the Senate will act on the measure. The House passed the Build Back Better bill, 220-213, in November. It includes a provision that would allow Medicare to negotiate the prices of certain drugs covered by Part D pharmacy plans.
That would mark a reversal of the stance taken when Congress created the pharmacy benefit in a 2003 law, which left negotiations to insurers that cover Part D plans.
Republicans have long argued insurers get the best deals on drugs for people on Medicare. Democrats say this approach sacrifices much of Medicare’s bargaining clout, scattering it among plans.
“This fight has been going on since the Medicare Part D legislation which gave away the store” to drugmakers, said Speaker Nancy Pelosi (D-CA) at a Dec. 10 press conference about the House Oversight report. “And they got used to having the store to themselves.”
The Endocrine Society is urging the Senate to protect the insulin affordability provisions included in the Build Back Better Act and move quickly to pass this crucial legislation.
“We implore all Senators to ensure these provisions are not scaled back. The Build Back Better Act represents the best opportunity to address the price of insulin. Millions of Americans cannot wait any longer for a solution,” it said in a statement issued Dec. 14.
Better deals for military, medicaid programs
Medicare is unusual among federal programs in that it doesn’t directly leverage its clout to lower drug costs.
Total Part D expenditures were approximately $105 billion last year, according to Medicare’s board of trustees. This spending is divided among the many insurers that run Part D plans, which then make a myriad of decisions about formularies and other factors that affect pricing.
For drugs administered by clinicians, and thus covered by Medicare Part B, the program pays a premium of the reported average sales price. Part B drug spending was $39 billion in 2019, an increase of about 11.6% from the previous year, according to the Medicare Payment Advisory Commission.
In contrast, federal law calls for steep reductions in drug prices for people on Medicaid.
The Department of Veterans Affairs (VA) and the Defense Department (DoD)’s Tricare program use several bargaining strategies to lower prices. To control costs, VA and DoD often use formularies of preferred drugs, steer patients to lower-cost drugs, and buy drugs in large volumes, “all of which increase their leverage with drug manufacturers,” the staff of the Congressional Budget Office (CBO) wrote in a Feb. 2021 report.
The CBO report examines how those different federal agencies’ approaches played out in terms of prices, net of applicable rebates, and discounts of 176 top-selling brand-name drugs in Medicare Part D.
The average price for this group of drugs was $118 in Medicaid. And for VA and DoD, the average prices were $190 and $184, respectively, for drugs dispensed at the agencies’ medical facilities or by mail.
But for Medicare Part D, the average price was $343, CBO said in the report, which was one of the sources consulted by House Oversight staff when developing their report released on Dec. 10.
Insulin still of interest, 100 years after its discovery
The House Oversight report runs to almost 270 pages. It addresses several issues with drug prices, including strategies pharmaceutical companies have used to thwart generic competition. On Monday, the trade group America’s Health Insurance Plans separately released its own report looking at patents and delays to the introduction of generic drugs.
Yet, much of the debate on drug prices has focused on one of the oldest widely produced prescription drugs, insulin.
Even with the allowance of generic competition for the essential medicine, branded versions of insulin have been some of the costliest products for Medicare in recent years. Eli Lilly, Novo Nordisk, and Sanofi dominate the insulin market.
Medicare Part D spent about $2.5 billion in 2019 on Sanofi’s Lantus Solostar insulin, or about $2,585 per person in the program using it. The program also paid about $1.1 billion for another form of Lantus, or about $2,746 per patient.
Medicare Part D also spent about $1.84 billion in 2019 on Novo Nordisk’s NovoLog FlexPen, or about $3,063 per person.
Medicare Part D’s drug spending dashboard also lists eight versions of Lilly’s Humalog, with combined 2019 spending of more than $2 billion. The cost per patient in Medicare Part D ranges from $5,619 to $1,462.
“Over the past 20 years, they have repeatedly and dramatically raised the list prices of their rapid-acting and long-acting insulins and reaped billions of dollars in revenues,” write the House Oversight staff in their report.
Republicans on the House Oversight and Reform Committee disagree with their Democratic colleagues on many points in the debate on drug prices, but they also looked at insulin as a cause for concern.
GOP members of the committee released a separate report on Dec. 10. They call for greater clarity into the role middlemen in the drug-supply chain – known as pharmaceutical benefit managers – may play in the rising costs of medicines. The GOP report notes that there are bills pending in the House that would seek to steer any discounts offered on insulin within the supply chain toward consumers (Insulin Price Reduction Act H.R. 4906, Insulin Cost Reduction Act H.R. 5623).
Democratic staff in the committee’s report seek to draw attention to how manufacturers priced their insulin products, including the comment by the Novo Nordisk employee about wishing for a price hike for a competitor’s product.
In a statement provided to this news organization, Novo Nordisk said the committee’s report reflects “a limited picture of the efforts put forth by our company and other companies to manage formulary access.”
“This glimpse into the complexity of pricing, formularies, and the health care system demonstrates why Novo Nordisk continues to advocate for comprehensive solutions,” Denmark’s Novo Nordisk said in the statement.
$35 a month for insulin?
Paris-based Sanofi said it makes insulin-pricing decisions independently from competitors. Sanofi said the net price of its insulins has declined by 53% since 2012, arguing the high prices charged to patients reflect decisions made elsewhere in the supply chain.
“Over the same period, the net price for commercial and Medicare Part D plans of Lantus has fallen 44.9%, while average out-of-pocket costs for patients with commercial insurance and Medicare Part D has risen approximately 82%,” Sanofi said.
“For all the focus on the growth of list prices, today, the average net price of Lantus is below 2006 levels. That is why we support policy reforms to require health plans to share negotiated savings with patients by requiring patient cost-sharing be tied to the net prices.”
Indianapolis-based Lilly offered a similar response in a statement to this news organization.
“Lilly, like other companies, monitors competitor list-price changes that are available through publicly available services,” the company said. “However, any changes we make to our list prices are independent decisions, and to the extent they consider competitors they are informed only through publicly available data.”
Despite rising insurance deductibles, the average monthly out-of-pocket cost for Lilly insulin has dropped 27% to $28.05 over the past 4 years, the company said in an interview. Lilly also noted that there are “several affordability options now available” allowing people to purchase their monthly prescription of its insulin for $35, “whether they are uninsured or use commercial insurance, Medicaid, or a participating Medicare Part D plan.”
In 2020, Lilly had announced that people with commercial insurance and those without insurance would be able to get monthly prescriptions of Lilly insulin for $35.
The Build Back Better Act would require insurers, including Medicare Part D plans and private group or individual health plans, to charge patient cost-sharing of no more than $35 per month for insulin products, said the staff of the nonprofit Kaiser Family Foundation (KFF) in a review of the bill.
“Private group or individual plans would not be required to cover all insulin products, just one of each dosage form (vial, pen) and insulin type (rapid-acting, short-acting, intermediate-acting, and long-acting), for no more than $35,” the KFF staff state in the report.
People enrolled in Medicare can already choose to enroll in a Part D plan participating in a federal test program that can secure certain insulin products for them at a monthly copayment of $35. In 2022, a total of 2,159 Part D plans will participate in this model, a 32% increase in participating plans since 2021, KFF said.
A version of this article first appeared on Medscape.com.
Medicare could have saved more than $16.7 billion on three kinds of insulin products from 2011 to 2017 if it had secured the same discounts other federal health programs get through negotiations, House Democrats argue in a new report.
On Dec. 10, Democrats on the House Committee on Oversight and Reform released a final majority staff report, which they say is the culmination of an almost 3-year investigation into pharmaceutical pricing and business practices. The report draws from 1.5 million pages of internal company documents, the committee says.
Documents from insulin makers Eli Lilly, Novo Nordisk, and Sanofi indicate these firms “raised their prices in lockstep in order to maintain ‘pricing parity’,” with senior executives encouraging the practice, the committee staff writes in the report.
“In a discussion among Novo Nordisk employees about an Eli Lilly price increase for a different diabetes product on Dec. 24, 2015, a Novo Nordisk pricing analyst remarked, ‘[M]aybe Sanofi will wait until tomorrow morning to announce their price increase ... that’s all I want for Christmas,’” the report states.
House Democrats are seeking to use the report findings to aid their Senate colleagues’ attempt to pass the sweeping Build Back Better bill, which includes many provisions addressing drug costs.
It’s still unclear when the Senate will act on the measure. The House passed the Build Back Better bill, 220-213, in November. It includes a provision that would allow Medicare to negotiate the prices of certain drugs covered by Part D pharmacy plans.
That would mark a reversal of the stance taken when Congress created the pharmacy benefit in a 2003 law, which left negotiations to insurers that cover Part D plans.
Republicans have long argued insurers get the best deals on drugs for people on Medicare. Democrats say this approach sacrifices much of Medicare’s bargaining clout, scattering it among plans.
“This fight has been going on since the Medicare Part D legislation which gave away the store” to drugmakers, said Speaker Nancy Pelosi (D-CA) at a Dec. 10 press conference about the House Oversight report. “And they got used to having the store to themselves.”
The Endocrine Society is urging the Senate to protect the insulin affordability provisions included in the Build Back Better Act and move quickly to pass this crucial legislation.
“We implore all Senators to ensure these provisions are not scaled back. The Build Back Better Act represents the best opportunity to address the price of insulin. Millions of Americans cannot wait any longer for a solution,” it said in a statement issued Dec. 14.
Better deals for military, medicaid programs
Medicare is unusual among federal programs in that it doesn’t directly leverage its clout to lower drug costs.
Total Part D expenditures were approximately $105 billion last year, according to Medicare’s board of trustees. This spending is divided among the many insurers that run Part D plans, which then make a myriad of decisions about formularies and other factors that affect pricing.
For drugs administered by clinicians, and thus covered by Medicare Part B, the program pays a premium of the reported average sales price. Part B drug spending was $39 billion in 2019, an increase of about 11.6% from the previous year, according to the Medicare Payment Advisory Commission.
In contrast, federal law calls for steep reductions in drug prices for people on Medicaid.
The Department of Veterans Affairs (VA) and the Defense Department (DoD)’s Tricare program use several bargaining strategies to lower prices. To control costs, VA and DoD often use formularies of preferred drugs, steer patients to lower-cost drugs, and buy drugs in large volumes, “all of which increase their leverage with drug manufacturers,” the staff of the Congressional Budget Office (CBO) wrote in a Feb. 2021 report.
The CBO report examines how those different federal agencies’ approaches played out in terms of prices, net of applicable rebates, and discounts of 176 top-selling brand-name drugs in Medicare Part D.
The average price for this group of drugs was $118 in Medicaid. And for VA and DoD, the average prices were $190 and $184, respectively, for drugs dispensed at the agencies’ medical facilities or by mail.
But for Medicare Part D, the average price was $343, CBO said in the report, which was one of the sources consulted by House Oversight staff when developing their report released on Dec. 10.
Insulin still of interest, 100 years after its discovery
The House Oversight report runs to almost 270 pages. It addresses several issues with drug prices, including strategies pharmaceutical companies have used to thwart generic competition. On Monday, the trade group America’s Health Insurance Plans separately released its own report looking at patents and delays to the introduction of generic drugs.
Yet, much of the debate on drug prices has focused on one of the oldest widely produced prescription drugs, insulin.
Even with the allowance of generic competition for the essential medicine, branded versions of insulin have been some of the costliest products for Medicare in recent years. Eli Lilly, Novo Nordisk, and Sanofi dominate the insulin market.
Medicare Part D spent about $2.5 billion in 2019 on Sanofi’s Lantus Solostar insulin, or about $2,585 per person in the program using it. The program also paid about $1.1 billion for another form of Lantus, or about $2,746 per patient.
Medicare Part D also spent about $1.84 billion in 2019 on Novo Nordisk’s NovoLog FlexPen, or about $3,063 per person.
Medicare Part D’s drug spending dashboard also lists eight versions of Lilly’s Humalog, with combined 2019 spending of more than $2 billion. The cost per patient in Medicare Part D ranges from $5,619 to $1,462.
“Over the past 20 years, they have repeatedly and dramatically raised the list prices of their rapid-acting and long-acting insulins and reaped billions of dollars in revenues,” write the House Oversight staff in their report.
Republicans on the House Oversight and Reform Committee disagree with their Democratic colleagues on many points in the debate on drug prices, but they also looked at insulin as a cause for concern.
GOP members of the committee released a separate report on Dec. 10. They call for greater clarity into the role middlemen in the drug-supply chain – known as pharmaceutical benefit managers – may play in the rising costs of medicines. The GOP report notes that there are bills pending in the House that would seek to steer any discounts offered on insulin within the supply chain toward consumers (Insulin Price Reduction Act H.R. 4906, Insulin Cost Reduction Act H.R. 5623).
Democratic staff in the committee’s report seek to draw attention to how manufacturers priced their insulin products, including the comment by the Novo Nordisk employee about wishing for a price hike for a competitor’s product.
In a statement provided to this news organization, Novo Nordisk said the committee’s report reflects “a limited picture of the efforts put forth by our company and other companies to manage formulary access.”
“This glimpse into the complexity of pricing, formularies, and the health care system demonstrates why Novo Nordisk continues to advocate for comprehensive solutions,” Denmark’s Novo Nordisk said in the statement.
$35 a month for insulin?
Paris-based Sanofi said it makes insulin-pricing decisions independently from competitors. Sanofi said the net price of its insulins has declined by 53% since 2012, arguing the high prices charged to patients reflect decisions made elsewhere in the supply chain.
“Over the same period, the net price for commercial and Medicare Part D plans of Lantus has fallen 44.9%, while average out-of-pocket costs for patients with commercial insurance and Medicare Part D has risen approximately 82%,” Sanofi said.
“For all the focus on the growth of list prices, today, the average net price of Lantus is below 2006 levels. That is why we support policy reforms to require health plans to share negotiated savings with patients by requiring patient cost-sharing be tied to the net prices.”
Indianapolis-based Lilly offered a similar response in a statement to this news organization.
“Lilly, like other companies, monitors competitor list-price changes that are available through publicly available services,” the company said. “However, any changes we make to our list prices are independent decisions, and to the extent they consider competitors they are informed only through publicly available data.”
Despite rising insurance deductibles, the average monthly out-of-pocket cost for Lilly insulin has dropped 27% to $28.05 over the past 4 years, the company said in an interview. Lilly also noted that there are “several affordability options now available” allowing people to purchase their monthly prescription of its insulin for $35, “whether they are uninsured or use commercial insurance, Medicaid, or a participating Medicare Part D plan.”
In 2020, Lilly had announced that people with commercial insurance and those without insurance would be able to get monthly prescriptions of Lilly insulin for $35.
The Build Back Better Act would require insurers, including Medicare Part D plans and private group or individual health plans, to charge patient cost-sharing of no more than $35 per month for insulin products, said the staff of the nonprofit Kaiser Family Foundation (KFF) in a review of the bill.
“Private group or individual plans would not be required to cover all insulin products, just one of each dosage form (vial, pen) and insulin type (rapid-acting, short-acting, intermediate-acting, and long-acting), for no more than $35,” the KFF staff state in the report.
People enrolled in Medicare can already choose to enroll in a Part D plan participating in a federal test program that can secure certain insulin products for them at a monthly copayment of $35. In 2022, a total of 2,159 Part D plans will participate in this model, a 32% increase in participating plans since 2021, KFF said.
A version of this article first appeared on Medscape.com.
Medicare could have saved more than $16.7 billion on three kinds of insulin products from 2011 to 2017 if it had secured the same discounts other federal health programs get through negotiations, House Democrats argue in a new report.
On Dec. 10, Democrats on the House Committee on Oversight and Reform released a final majority staff report, which they say is the culmination of an almost 3-year investigation into pharmaceutical pricing and business practices. The report draws from 1.5 million pages of internal company documents, the committee says.
Documents from insulin makers Eli Lilly, Novo Nordisk, and Sanofi indicate these firms “raised their prices in lockstep in order to maintain ‘pricing parity’,” with senior executives encouraging the practice, the committee staff writes in the report.
“In a discussion among Novo Nordisk employees about an Eli Lilly price increase for a different diabetes product on Dec. 24, 2015, a Novo Nordisk pricing analyst remarked, ‘[M]aybe Sanofi will wait until tomorrow morning to announce their price increase ... that’s all I want for Christmas,’” the report states.
House Democrats are seeking to use the report findings to aid their Senate colleagues’ attempt to pass the sweeping Build Back Better bill, which includes many provisions addressing drug costs.
It’s still unclear when the Senate will act on the measure. The House passed the Build Back Better bill, 220-213, in November. It includes a provision that would allow Medicare to negotiate the prices of certain drugs covered by Part D pharmacy plans.
That would mark a reversal of the stance taken when Congress created the pharmacy benefit in a 2003 law, which left negotiations to insurers that cover Part D plans.
Republicans have long argued insurers get the best deals on drugs for people on Medicare. Democrats say this approach sacrifices much of Medicare’s bargaining clout, scattering it among plans.
“This fight has been going on since the Medicare Part D legislation which gave away the store” to drugmakers, said Speaker Nancy Pelosi (D-CA) at a Dec. 10 press conference about the House Oversight report. “And they got used to having the store to themselves.”
The Endocrine Society is urging the Senate to protect the insulin affordability provisions included in the Build Back Better Act and move quickly to pass this crucial legislation.
“We implore all Senators to ensure these provisions are not scaled back. The Build Back Better Act represents the best opportunity to address the price of insulin. Millions of Americans cannot wait any longer for a solution,” it said in a statement issued Dec. 14.
Better deals for military, medicaid programs
Medicare is unusual among federal programs in that it doesn’t directly leverage its clout to lower drug costs.
Total Part D expenditures were approximately $105 billion last year, according to Medicare’s board of trustees. This spending is divided among the many insurers that run Part D plans, which then make a myriad of decisions about formularies and other factors that affect pricing.
For drugs administered by clinicians, and thus covered by Medicare Part B, the program pays a premium of the reported average sales price. Part B drug spending was $39 billion in 2019, an increase of about 11.6% from the previous year, according to the Medicare Payment Advisory Commission.
In contrast, federal law calls for steep reductions in drug prices for people on Medicaid.
The Department of Veterans Affairs (VA) and the Defense Department (DoD)’s Tricare program use several bargaining strategies to lower prices. To control costs, VA and DoD often use formularies of preferred drugs, steer patients to lower-cost drugs, and buy drugs in large volumes, “all of which increase their leverage with drug manufacturers,” the staff of the Congressional Budget Office (CBO) wrote in a Feb. 2021 report.
The CBO report examines how those different federal agencies’ approaches played out in terms of prices, net of applicable rebates, and discounts of 176 top-selling brand-name drugs in Medicare Part D.
The average price for this group of drugs was $118 in Medicaid. And for VA and DoD, the average prices were $190 and $184, respectively, for drugs dispensed at the agencies’ medical facilities or by mail.
But for Medicare Part D, the average price was $343, CBO said in the report, which was one of the sources consulted by House Oversight staff when developing their report released on Dec. 10.
Insulin still of interest, 100 years after its discovery
The House Oversight report runs to almost 270 pages. It addresses several issues with drug prices, including strategies pharmaceutical companies have used to thwart generic competition. On Monday, the trade group America’s Health Insurance Plans separately released its own report looking at patents and delays to the introduction of generic drugs.
Yet, much of the debate on drug prices has focused on one of the oldest widely produced prescription drugs, insulin.
Even with the allowance of generic competition for the essential medicine, branded versions of insulin have been some of the costliest products for Medicare in recent years. Eli Lilly, Novo Nordisk, and Sanofi dominate the insulin market.
Medicare Part D spent about $2.5 billion in 2019 on Sanofi’s Lantus Solostar insulin, or about $2,585 per person in the program using it. The program also paid about $1.1 billion for another form of Lantus, or about $2,746 per patient.
Medicare Part D also spent about $1.84 billion in 2019 on Novo Nordisk’s NovoLog FlexPen, or about $3,063 per person.
Medicare Part D’s drug spending dashboard also lists eight versions of Lilly’s Humalog, with combined 2019 spending of more than $2 billion. The cost per patient in Medicare Part D ranges from $5,619 to $1,462.
“Over the past 20 years, they have repeatedly and dramatically raised the list prices of their rapid-acting and long-acting insulins and reaped billions of dollars in revenues,” write the House Oversight staff in their report.
Republicans on the House Oversight and Reform Committee disagree with their Democratic colleagues on many points in the debate on drug prices, but they also looked at insulin as a cause for concern.
GOP members of the committee released a separate report on Dec. 10. They call for greater clarity into the role middlemen in the drug-supply chain – known as pharmaceutical benefit managers – may play in the rising costs of medicines. The GOP report notes that there are bills pending in the House that would seek to steer any discounts offered on insulin within the supply chain toward consumers (Insulin Price Reduction Act H.R. 4906, Insulin Cost Reduction Act H.R. 5623).
Democratic staff in the committee’s report seek to draw attention to how manufacturers priced their insulin products, including the comment by the Novo Nordisk employee about wishing for a price hike for a competitor’s product.
In a statement provided to this news organization, Novo Nordisk said the committee’s report reflects “a limited picture of the efforts put forth by our company and other companies to manage formulary access.”
“This glimpse into the complexity of pricing, formularies, and the health care system demonstrates why Novo Nordisk continues to advocate for comprehensive solutions,” Denmark’s Novo Nordisk said in the statement.
$35 a month for insulin?
Paris-based Sanofi said it makes insulin-pricing decisions independently from competitors. Sanofi said the net price of its insulins has declined by 53% since 2012, arguing the high prices charged to patients reflect decisions made elsewhere in the supply chain.
“Over the same period, the net price for commercial and Medicare Part D plans of Lantus has fallen 44.9%, while average out-of-pocket costs for patients with commercial insurance and Medicare Part D has risen approximately 82%,” Sanofi said.
“For all the focus on the growth of list prices, today, the average net price of Lantus is below 2006 levels. That is why we support policy reforms to require health plans to share negotiated savings with patients by requiring patient cost-sharing be tied to the net prices.”
Indianapolis-based Lilly offered a similar response in a statement to this news organization.
“Lilly, like other companies, monitors competitor list-price changes that are available through publicly available services,” the company said. “However, any changes we make to our list prices are independent decisions, and to the extent they consider competitors they are informed only through publicly available data.”
Despite rising insurance deductibles, the average monthly out-of-pocket cost for Lilly insulin has dropped 27% to $28.05 over the past 4 years, the company said in an interview. Lilly also noted that there are “several affordability options now available” allowing people to purchase their monthly prescription of its insulin for $35, “whether they are uninsured or use commercial insurance, Medicaid, or a participating Medicare Part D plan.”
In 2020, Lilly had announced that people with commercial insurance and those without insurance would be able to get monthly prescriptions of Lilly insulin for $35.
The Build Back Better Act would require insurers, including Medicare Part D plans and private group or individual health plans, to charge patient cost-sharing of no more than $35 per month for insulin products, said the staff of the nonprofit Kaiser Family Foundation (KFF) in a review of the bill.
“Private group or individual plans would not be required to cover all insulin products, just one of each dosage form (vial, pen) and insulin type (rapid-acting, short-acting, intermediate-acting, and long-acting), for no more than $35,” the KFF staff state in the report.
People enrolled in Medicare can already choose to enroll in a Part D plan participating in a federal test program that can secure certain insulin products for them at a monthly copayment of $35. In 2022, a total of 2,159 Part D plans will participate in this model, a 32% increase in participating plans since 2021, KFF said.
A version of this article first appeared on Medscape.com.
Even COVID-19 can’t stop a true optimist
Squeezing a little lemonade out of COVID-19
We like to think of ourselves as optimists here at LOTME. A glass is half full, the sky is partly sunny, and our motto is “Always look on the bright side of insanity.” Then again, our motto before that was “LOTME: Where science meets stupid,” so what do we know?
Anyway, it’s that upbeat, can-do attitude that allows us to say something positive – two somethings, actually – about the insanity that is COVID-19.
Our journey to the bright side begins, oddly enough, in the courtroom. Seems that our old friend, the face mask, is something of a lie-detector aid for juries. The authors of a recent literature review of studies on deception “found that facial expressions and other forms of nonverbal behaviour are an unreliable indicator of deceit,” according to a statement from the University of Portsmouth, where the analysis was conducted.
The one study that directly examined the role of face coverings in court proceedings showed that, “by taking away the distraction of nonverbal behaviours, observers had to rely on speech content, which turned out to be better for detecting lies,” the university said.
The second stage of our positivity trek brings us to the National Trends in Disability Employment monthly update, where we see a fourth consecutive month of gains for people with disabilities despite the larger trend of declines among those without disabilities.
Here are some numbers from the Kessler Foundation and the University of New Hampshire’s Institute on Disability to tell the story: From October to November, the employment-to-population ratio increased 4.2% for working-age people with disabilities, compared with 0.4% for people without disabilities. At the same time, the labor force participation rate rose 2.4% for working-age people with disabilities and just 0.1% for working-age people without disabilities.
Both indicators surpassed their historic highs, Andrew Houtenville, PhD, director of the Institute on Disability, said in the update. “These gains suggest that the restructuring resulting from the pandemic may be benefiting people with disabilities. Ironically, it may have taken a pandemic to shake the labor market loose for people with disabilities.”
And that is how a world-class optimist turns one gigantic lemon into lemonade.
Cut the cheese for better sleep
So, we’ve already talked about the TikTok lettuce tea hack that’s supposed to help us sleep better. Well, there’s another food that could have the opposite effect.
According to an article from the BBC, cheese has something of a reputation. Ever since the 1960s, when a researcher noted that one patient’s nightmares stopped after he quit eating an ounce or two of cheddar each night, there’s been speculation that cheese gives you weird dreams. Another study in 2005 suggested certain types of cheese cause certain types of dreams. Blue cheese for vivid dreams and cheddar cheese for celebrity cameos.
But is there any truth to it at all?
Regardless of what we eat, going to bed hungry could cause vivid dreams, according to research by Tore Nielsen, director of the University of Montreal’s dream and nightmare lab. The 2015 study showed that high lactose could have an effect on dreams.
In that study, 17% of participants said their dreams were influenced by what they ate, but the kicker was that dairy products were the foods most reported as causing the weird dreams, the BBC noted.
“It’s likely an indirect effect in that lactose produces symptoms like gas, bloating and diarrhoea and influences dreams, as dreams draw on somatic sources like this. And if you have certain kinds of intolerances, you still may be likely to eat those foods sometimes,” Mr. Nielsen told the BBC.
There’s also the theory that it’s all in the timing of consumption. Are you the type of person to sneak a slice of cheese from the fridge late at night? (Nods.) Same.
“One reason cheese and nightmares come about is that eating later before bed is more likely to disrupt sleep, and cheese can be hard to digest,” said Charlotte Gupta, a research fellow at Central Queensland University in Australia and a coauthor of a 2020 review on how diet affects our sleep.
So as tempting as it is, maybe skip sprinkling Parmesan cheese shreds into your mouth at the open fridge before bed.
Teeing up against Parkinson’s
For the nearly 1 million people in the United States with Parkinson’s disease, tai chi is one of the best ways to alleviate the symptoms. The average Parkinson’s patient, however, is going to be on the older side and more likely to view the martial art as some sort of communist plot. And would you participate in a communist plot? We don’t think so.
One group of researchers saw that patients weren’t keeping up with their therapy and decided to try a different activity, something that older people would be more likely to stick with. Something a bit more stereotypical. No, not shuffleboard. They tried golf.
“Golf is popular – the most popular sport for people over the age of 55 – which might encourage people to try it and stick with it,” study author Anne-Marie A. Wills, MD, of Massachusetts General Hospital, Boston, said in a Study Finds report.
In a small study, the investigators had a group of patients with Parkinson’s regularly go to a driving range for 10 weeks to hit golf balls (all expenses paid too, and that’s a big deal for golf), while another group continued with their tai chi.
At the end of the study, the 8 patients who went to the driving range had significantly better results in a Parkinson’s mobility test than those of the 12 patients in the tai chi group. In addition, the golf-group participants said they were more likely to continue with their therapy than were those who did tai chi.
Despite the small size of the study, the research team said the results certainly warrant further research. After all, the best sort of therapy is the kind that actually gets done. And golf just gets in your head. The eternal quest to add distance, to straighten out that annoying slice, to stop thinning half your chips, to make those annoying 4-footers. ... Maybe that’s just us.
Squeezing a little lemonade out of COVID-19
We like to think of ourselves as optimists here at LOTME. A glass is half full, the sky is partly sunny, and our motto is “Always look on the bright side of insanity.” Then again, our motto before that was “LOTME: Where science meets stupid,” so what do we know?
Anyway, it’s that upbeat, can-do attitude that allows us to say something positive – two somethings, actually – about the insanity that is COVID-19.
Our journey to the bright side begins, oddly enough, in the courtroom. Seems that our old friend, the face mask, is something of a lie-detector aid for juries. The authors of a recent literature review of studies on deception “found that facial expressions and other forms of nonverbal behaviour are an unreliable indicator of deceit,” according to a statement from the University of Portsmouth, where the analysis was conducted.
The one study that directly examined the role of face coverings in court proceedings showed that, “by taking away the distraction of nonverbal behaviours, observers had to rely on speech content, which turned out to be better for detecting lies,” the university said.
The second stage of our positivity trek brings us to the National Trends in Disability Employment monthly update, where we see a fourth consecutive month of gains for people with disabilities despite the larger trend of declines among those without disabilities.
Here are some numbers from the Kessler Foundation and the University of New Hampshire’s Institute on Disability to tell the story: From October to November, the employment-to-population ratio increased 4.2% for working-age people with disabilities, compared with 0.4% for people without disabilities. At the same time, the labor force participation rate rose 2.4% for working-age people with disabilities and just 0.1% for working-age people without disabilities.
Both indicators surpassed their historic highs, Andrew Houtenville, PhD, director of the Institute on Disability, said in the update. “These gains suggest that the restructuring resulting from the pandemic may be benefiting people with disabilities. Ironically, it may have taken a pandemic to shake the labor market loose for people with disabilities.”
And that is how a world-class optimist turns one gigantic lemon into lemonade.
Cut the cheese for better sleep
So, we’ve already talked about the TikTok lettuce tea hack that’s supposed to help us sleep better. Well, there’s another food that could have the opposite effect.
According to an article from the BBC, cheese has something of a reputation. Ever since the 1960s, when a researcher noted that one patient’s nightmares stopped after he quit eating an ounce or two of cheddar each night, there’s been speculation that cheese gives you weird dreams. Another study in 2005 suggested certain types of cheese cause certain types of dreams. Blue cheese for vivid dreams and cheddar cheese for celebrity cameos.
But is there any truth to it at all?
Regardless of what we eat, going to bed hungry could cause vivid dreams, according to research by Tore Nielsen, director of the University of Montreal’s dream and nightmare lab. The 2015 study showed that high lactose could have an effect on dreams.
In that study, 17% of participants said their dreams were influenced by what they ate, but the kicker was that dairy products were the foods most reported as causing the weird dreams, the BBC noted.
“It’s likely an indirect effect in that lactose produces symptoms like gas, bloating and diarrhoea and influences dreams, as dreams draw on somatic sources like this. And if you have certain kinds of intolerances, you still may be likely to eat those foods sometimes,” Mr. Nielsen told the BBC.
There’s also the theory that it’s all in the timing of consumption. Are you the type of person to sneak a slice of cheese from the fridge late at night? (Nods.) Same.
“One reason cheese and nightmares come about is that eating later before bed is more likely to disrupt sleep, and cheese can be hard to digest,” said Charlotte Gupta, a research fellow at Central Queensland University in Australia and a coauthor of a 2020 review on how diet affects our sleep.
So as tempting as it is, maybe skip sprinkling Parmesan cheese shreds into your mouth at the open fridge before bed.
Teeing up against Parkinson’s
For the nearly 1 million people in the United States with Parkinson’s disease, tai chi is one of the best ways to alleviate the symptoms. The average Parkinson’s patient, however, is going to be on the older side and more likely to view the martial art as some sort of communist plot. And would you participate in a communist plot? We don’t think so.
One group of researchers saw that patients weren’t keeping up with their therapy and decided to try a different activity, something that older people would be more likely to stick with. Something a bit more stereotypical. No, not shuffleboard. They tried golf.
“Golf is popular – the most popular sport for people over the age of 55 – which might encourage people to try it and stick with it,” study author Anne-Marie A. Wills, MD, of Massachusetts General Hospital, Boston, said in a Study Finds report.
In a small study, the investigators had a group of patients with Parkinson’s regularly go to a driving range for 10 weeks to hit golf balls (all expenses paid too, and that’s a big deal for golf), while another group continued with their tai chi.
At the end of the study, the 8 patients who went to the driving range had significantly better results in a Parkinson’s mobility test than those of the 12 patients in the tai chi group. In addition, the golf-group participants said they were more likely to continue with their therapy than were those who did tai chi.
Despite the small size of the study, the research team said the results certainly warrant further research. After all, the best sort of therapy is the kind that actually gets done. And golf just gets in your head. The eternal quest to add distance, to straighten out that annoying slice, to stop thinning half your chips, to make those annoying 4-footers. ... Maybe that’s just us.
Squeezing a little lemonade out of COVID-19
We like to think of ourselves as optimists here at LOTME. A glass is half full, the sky is partly sunny, and our motto is “Always look on the bright side of insanity.” Then again, our motto before that was “LOTME: Where science meets stupid,” so what do we know?
Anyway, it’s that upbeat, can-do attitude that allows us to say something positive – two somethings, actually – about the insanity that is COVID-19.
Our journey to the bright side begins, oddly enough, in the courtroom. Seems that our old friend, the face mask, is something of a lie-detector aid for juries. The authors of a recent literature review of studies on deception “found that facial expressions and other forms of nonverbal behaviour are an unreliable indicator of deceit,” according to a statement from the University of Portsmouth, where the analysis was conducted.
The one study that directly examined the role of face coverings in court proceedings showed that, “by taking away the distraction of nonverbal behaviours, observers had to rely on speech content, which turned out to be better for detecting lies,” the university said.
The second stage of our positivity trek brings us to the National Trends in Disability Employment monthly update, where we see a fourth consecutive month of gains for people with disabilities despite the larger trend of declines among those without disabilities.
Here are some numbers from the Kessler Foundation and the University of New Hampshire’s Institute on Disability to tell the story: From October to November, the employment-to-population ratio increased 4.2% for working-age people with disabilities, compared with 0.4% for people without disabilities. At the same time, the labor force participation rate rose 2.4% for working-age people with disabilities and just 0.1% for working-age people without disabilities.
Both indicators surpassed their historic highs, Andrew Houtenville, PhD, director of the Institute on Disability, said in the update. “These gains suggest that the restructuring resulting from the pandemic may be benefiting people with disabilities. Ironically, it may have taken a pandemic to shake the labor market loose for people with disabilities.”
And that is how a world-class optimist turns one gigantic lemon into lemonade.
Cut the cheese for better sleep
So, we’ve already talked about the TikTok lettuce tea hack that’s supposed to help us sleep better. Well, there’s another food that could have the opposite effect.
According to an article from the BBC, cheese has something of a reputation. Ever since the 1960s, when a researcher noted that one patient’s nightmares stopped after he quit eating an ounce or two of cheddar each night, there’s been speculation that cheese gives you weird dreams. Another study in 2005 suggested certain types of cheese cause certain types of dreams. Blue cheese for vivid dreams and cheddar cheese for celebrity cameos.
But is there any truth to it at all?
Regardless of what we eat, going to bed hungry could cause vivid dreams, according to research by Tore Nielsen, director of the University of Montreal’s dream and nightmare lab. The 2015 study showed that high lactose could have an effect on dreams.
In that study, 17% of participants said their dreams were influenced by what they ate, but the kicker was that dairy products were the foods most reported as causing the weird dreams, the BBC noted.
“It’s likely an indirect effect in that lactose produces symptoms like gas, bloating and diarrhoea and influences dreams, as dreams draw on somatic sources like this. And if you have certain kinds of intolerances, you still may be likely to eat those foods sometimes,” Mr. Nielsen told the BBC.
There’s also the theory that it’s all in the timing of consumption. Are you the type of person to sneak a slice of cheese from the fridge late at night? (Nods.) Same.
“One reason cheese and nightmares come about is that eating later before bed is more likely to disrupt sleep, and cheese can be hard to digest,” said Charlotte Gupta, a research fellow at Central Queensland University in Australia and a coauthor of a 2020 review on how diet affects our sleep.
So as tempting as it is, maybe skip sprinkling Parmesan cheese shreds into your mouth at the open fridge before bed.
Teeing up against Parkinson’s
For the nearly 1 million people in the United States with Parkinson’s disease, tai chi is one of the best ways to alleviate the symptoms. The average Parkinson’s patient, however, is going to be on the older side and more likely to view the martial art as some sort of communist plot. And would you participate in a communist plot? We don’t think so.
One group of researchers saw that patients weren’t keeping up with their therapy and decided to try a different activity, something that older people would be more likely to stick with. Something a bit more stereotypical. No, not shuffleboard. They tried golf.
“Golf is popular – the most popular sport for people over the age of 55 – which might encourage people to try it and stick with it,” study author Anne-Marie A. Wills, MD, of Massachusetts General Hospital, Boston, said in a Study Finds report.
In a small study, the investigators had a group of patients with Parkinson’s regularly go to a driving range for 10 weeks to hit golf balls (all expenses paid too, and that’s a big deal for golf), while another group continued with their tai chi.
At the end of the study, the 8 patients who went to the driving range had significantly better results in a Parkinson’s mobility test than those of the 12 patients in the tai chi group. In addition, the golf-group participants said they were more likely to continue with their therapy than were those who did tai chi.
Despite the small size of the study, the research team said the results certainly warrant further research. After all, the best sort of therapy is the kind that actually gets done. And golf just gets in your head. The eternal quest to add distance, to straighten out that annoying slice, to stop thinning half your chips, to make those annoying 4-footers. ... Maybe that’s just us.
Cold snare polypectomy works for larger colorectal polyps too
Large colorectal polyps up to 15 mm in size can be safely and effectively removed with cold snare polypectomy (CSP), with a low incomplete resection rate, according to a single-center, observational study from China.
Multiple current guidelines recommend CSP for removing diminutive (≤5 mm) and small (6-9 mm) polyps, citing clinical data demonstrating that such an approach leads to high complete resection rates and a good safety profile.
However, the use of CSP for removing larger colorectal polyps (≥10 mm) remains controversial, with only limited safety and efficacy data.
Yuqi He, MD, from the department of gastroenterology at the Seventh Medical Center of Chinese PLA General Hospital in Beijing, and colleagues evaluated 440 neoplastic polyps removed by CSP from 261 patients (mean age, 56.6 years; 166 men).
Indications for colonoscopy were screening (53%), diagnostic (17%), polyp surveillance (24%), and other (6%).
Of the 440 polyps, 353 (80%) were small (5-9 mm) and 87 (20%) were large (10-15 mm); 379 (86%) were adenomas, 59 (13%) were sessile serrated lesions (SSL), and 2 (<1%) were high-grade dysplasia.
For all polyps (5-15 mm), the incomplete resection rate (primary outcome) was 2.27%. Incomplete resection was more common for large polyps (3.45%) compared with small polyps (1.98%), but the difference was not statistically significant (P = .411).
In univariate analysis, factors associated with incomplete resection were SSL, piecemeal resection, and prolonged resection time.
In multivariate regression analysis, independent risk factors for incomplete resection were SSL (odds ratio, 6.45; 95% CI, 1.48-28.03; P = .013) and prolonged resection time (OR, 7.39; 95% CI, 1.48-36.96; P = .015).
Immediate bleeding was more common with resection of large polyps (6.9% vs. 1.42%, P = .003).
There were no recurrences on follow-up colonoscopy in 37 cases with large polyps, further supporting the efficacy of CSP for this size group, the researchers say.
Their study was published online Nov. 16 in Clinical Gastroenterology and Hepatology.
Important study, lingering questions, experts say
Reached for comment, Rajesh N. Keswani, MD, of Northwestern University Feinberg School of Medicine, Chicago, said, “It is standard practice to remove polyps <10 mm using cold techniques. We generally advocate for use of a cold snare for polyps 3-9 mm in size; either large cold forceps or a cold snare can be used for polyps 1-2 mm in size,” a recommendation also conveyed in the American Gastroenterological Association’s recent clinical practice update.
“It is still unclear whether cold polypectomy techniques are appropriate for larger lesions,” Dr. Keswani, who wasn’t involved in the study, told this news organization.
“For larger polyps that require piecemeal technique (generally >2 cm), there are multiple small studies showing that resection using cold snare piecemeal technique may be safe and effective, at minimum for serrated lesions. There has been enough interest in this approach that is being formally studied in a large multicenter trial,” he noted.
“Based on the safety and efficacy data thus far for cold snare polypectomy in larger serrated lesions, it would be difficult to think that it would not be a safe choice for serrated lesions 10-20 mm,” Dr. Keswani commented.
However, the data remain “unclear for adenomatous lesions 10-20 mm in size. Unfortunately, in this study only 87 polyps 10-15 mm [in size] were removed, and a proportion were serrated and a proportion were adenomas,” he said in an interview.
“Thus, I don’t think this study is able to answer one of our key remaining polypectomy questions, which is whether cold snare techniques are the optimal approach for adenomas >10 mm in size,” said Dr. Keswani.
Also weighing in was Emre Gorgun, MD, with the Cleveland Clinic, who said it’s “an important study in the sense it raises awareness to the role of cold snaring techniques, especially since it is less expensive, quicker, and safe.”
“Late complications related to possibly using energy might be also eliminated. However, results for expanding the practice to larger polyps up to 15 mm should be taken cautiously into consideration,” Dr. Gorgun told this news organization.
“The threat of bleeding in high-risk patients, for example with a history of anticoagulation use, were not reported. Other patient-related factors can increase these risks as well. Additionally, the long-term follow-up is not reported,” commented Dr. Gorgun, who was not associated with the research.
“This study triggers the idea of using cold snaring in larger polyps; however, the results should be further reproduced and verified for long-term consequences and safety,” he concluded.
The authors have disclosed no relevant financial relationships. Dr. Keswani is a consultant for Boston Scientific and Neptune Medical and receives research support from Virgo. Dr. Gorgun is a consultant for Boston Scientific. Support for the study was provided b y grants from Project of Army Special Care and Beijing Municipal Science and Technology Commission.
A version of this article first appeared on Medscape.com.
Large colorectal polyps up to 15 mm in size can be safely and effectively removed with cold snare polypectomy (CSP), with a low incomplete resection rate, according to a single-center, observational study from China.
Multiple current guidelines recommend CSP for removing diminutive (≤5 mm) and small (6-9 mm) polyps, citing clinical data demonstrating that such an approach leads to high complete resection rates and a good safety profile.
However, the use of CSP for removing larger colorectal polyps (≥10 mm) remains controversial, with only limited safety and efficacy data.
Yuqi He, MD, from the department of gastroenterology at the Seventh Medical Center of Chinese PLA General Hospital in Beijing, and colleagues evaluated 440 neoplastic polyps removed by CSP from 261 patients (mean age, 56.6 years; 166 men).
Indications for colonoscopy were screening (53%), diagnostic (17%), polyp surveillance (24%), and other (6%).
Of the 440 polyps, 353 (80%) were small (5-9 mm) and 87 (20%) were large (10-15 mm); 379 (86%) were adenomas, 59 (13%) were sessile serrated lesions (SSL), and 2 (<1%) were high-grade dysplasia.
For all polyps (5-15 mm), the incomplete resection rate (primary outcome) was 2.27%. Incomplete resection was more common for large polyps (3.45%) compared with small polyps (1.98%), but the difference was not statistically significant (P = .411).
In univariate analysis, factors associated with incomplete resection were SSL, piecemeal resection, and prolonged resection time.
In multivariate regression analysis, independent risk factors for incomplete resection were SSL (odds ratio, 6.45; 95% CI, 1.48-28.03; P = .013) and prolonged resection time (OR, 7.39; 95% CI, 1.48-36.96; P = .015).
Immediate bleeding was more common with resection of large polyps (6.9% vs. 1.42%, P = .003).
There were no recurrences on follow-up colonoscopy in 37 cases with large polyps, further supporting the efficacy of CSP for this size group, the researchers say.
Their study was published online Nov. 16 in Clinical Gastroenterology and Hepatology.
Important study, lingering questions, experts say
Reached for comment, Rajesh N. Keswani, MD, of Northwestern University Feinberg School of Medicine, Chicago, said, “It is standard practice to remove polyps <10 mm using cold techniques. We generally advocate for use of a cold snare for polyps 3-9 mm in size; either large cold forceps or a cold snare can be used for polyps 1-2 mm in size,” a recommendation also conveyed in the American Gastroenterological Association’s recent clinical practice update.
“It is still unclear whether cold polypectomy techniques are appropriate for larger lesions,” Dr. Keswani, who wasn’t involved in the study, told this news organization.
“For larger polyps that require piecemeal technique (generally >2 cm), there are multiple small studies showing that resection using cold snare piecemeal technique may be safe and effective, at minimum for serrated lesions. There has been enough interest in this approach that is being formally studied in a large multicenter trial,” he noted.
“Based on the safety and efficacy data thus far for cold snare polypectomy in larger serrated lesions, it would be difficult to think that it would not be a safe choice for serrated lesions 10-20 mm,” Dr. Keswani commented.
However, the data remain “unclear for adenomatous lesions 10-20 mm in size. Unfortunately, in this study only 87 polyps 10-15 mm [in size] were removed, and a proportion were serrated and a proportion were adenomas,” he said in an interview.
“Thus, I don’t think this study is able to answer one of our key remaining polypectomy questions, which is whether cold snare techniques are the optimal approach for adenomas >10 mm in size,” said Dr. Keswani.
Also weighing in was Emre Gorgun, MD, with the Cleveland Clinic, who said it’s “an important study in the sense it raises awareness to the role of cold snaring techniques, especially since it is less expensive, quicker, and safe.”
“Late complications related to possibly using energy might be also eliminated. However, results for expanding the practice to larger polyps up to 15 mm should be taken cautiously into consideration,” Dr. Gorgun told this news organization.
“The threat of bleeding in high-risk patients, for example with a history of anticoagulation use, were not reported. Other patient-related factors can increase these risks as well. Additionally, the long-term follow-up is not reported,” commented Dr. Gorgun, who was not associated with the research.
“This study triggers the idea of using cold snaring in larger polyps; however, the results should be further reproduced and verified for long-term consequences and safety,” he concluded.
The authors have disclosed no relevant financial relationships. Dr. Keswani is a consultant for Boston Scientific and Neptune Medical and receives research support from Virgo. Dr. Gorgun is a consultant for Boston Scientific. Support for the study was provided b y grants from Project of Army Special Care and Beijing Municipal Science and Technology Commission.
A version of this article first appeared on Medscape.com.
Large colorectal polyps up to 15 mm in size can be safely and effectively removed with cold snare polypectomy (CSP), with a low incomplete resection rate, according to a single-center, observational study from China.
Multiple current guidelines recommend CSP for removing diminutive (≤5 mm) and small (6-9 mm) polyps, citing clinical data demonstrating that such an approach leads to high complete resection rates and a good safety profile.
However, the use of CSP for removing larger colorectal polyps (≥10 mm) remains controversial, with only limited safety and efficacy data.
Yuqi He, MD, from the department of gastroenterology at the Seventh Medical Center of Chinese PLA General Hospital in Beijing, and colleagues evaluated 440 neoplastic polyps removed by CSP from 261 patients (mean age, 56.6 years; 166 men).
Indications for colonoscopy were screening (53%), diagnostic (17%), polyp surveillance (24%), and other (6%).
Of the 440 polyps, 353 (80%) were small (5-9 mm) and 87 (20%) were large (10-15 mm); 379 (86%) were adenomas, 59 (13%) were sessile serrated lesions (SSL), and 2 (<1%) were high-grade dysplasia.
For all polyps (5-15 mm), the incomplete resection rate (primary outcome) was 2.27%. Incomplete resection was more common for large polyps (3.45%) compared with small polyps (1.98%), but the difference was not statistically significant (P = .411).
In univariate analysis, factors associated with incomplete resection were SSL, piecemeal resection, and prolonged resection time.
In multivariate regression analysis, independent risk factors for incomplete resection were SSL (odds ratio, 6.45; 95% CI, 1.48-28.03; P = .013) and prolonged resection time (OR, 7.39; 95% CI, 1.48-36.96; P = .015).
Immediate bleeding was more common with resection of large polyps (6.9% vs. 1.42%, P = .003).
There were no recurrences on follow-up colonoscopy in 37 cases with large polyps, further supporting the efficacy of CSP for this size group, the researchers say.
Their study was published online Nov. 16 in Clinical Gastroenterology and Hepatology.
Important study, lingering questions, experts say
Reached for comment, Rajesh N. Keswani, MD, of Northwestern University Feinberg School of Medicine, Chicago, said, “It is standard practice to remove polyps <10 mm using cold techniques. We generally advocate for use of a cold snare for polyps 3-9 mm in size; either large cold forceps or a cold snare can be used for polyps 1-2 mm in size,” a recommendation also conveyed in the American Gastroenterological Association’s recent clinical practice update.
“It is still unclear whether cold polypectomy techniques are appropriate for larger lesions,” Dr. Keswani, who wasn’t involved in the study, told this news organization.
“For larger polyps that require piecemeal technique (generally >2 cm), there are multiple small studies showing that resection using cold snare piecemeal technique may be safe and effective, at minimum for serrated lesions. There has been enough interest in this approach that is being formally studied in a large multicenter trial,” he noted.
“Based on the safety and efficacy data thus far for cold snare polypectomy in larger serrated lesions, it would be difficult to think that it would not be a safe choice for serrated lesions 10-20 mm,” Dr. Keswani commented.
However, the data remain “unclear for adenomatous lesions 10-20 mm in size. Unfortunately, in this study only 87 polyps 10-15 mm [in size] were removed, and a proportion were serrated and a proportion were adenomas,” he said in an interview.
“Thus, I don’t think this study is able to answer one of our key remaining polypectomy questions, which is whether cold snare techniques are the optimal approach for adenomas >10 mm in size,” said Dr. Keswani.
Also weighing in was Emre Gorgun, MD, with the Cleveland Clinic, who said it’s “an important study in the sense it raises awareness to the role of cold snaring techniques, especially since it is less expensive, quicker, and safe.”
“Late complications related to possibly using energy might be also eliminated. However, results for expanding the practice to larger polyps up to 15 mm should be taken cautiously into consideration,” Dr. Gorgun told this news organization.
“The threat of bleeding in high-risk patients, for example with a history of anticoagulation use, were not reported. Other patient-related factors can increase these risks as well. Additionally, the long-term follow-up is not reported,” commented Dr. Gorgun, who was not associated with the research.
“This study triggers the idea of using cold snaring in larger polyps; however, the results should be further reproduced and verified for long-term consequences and safety,” he concluded.
The authors have disclosed no relevant financial relationships. Dr. Keswani is a consultant for Boston Scientific and Neptune Medical and receives research support from Virgo. Dr. Gorgun is a consultant for Boston Scientific. Support for the study was provided b y grants from Project of Army Special Care and Beijing Municipal Science and Technology Commission.
A version of this article first appeared on Medscape.com.
A mass on the ear
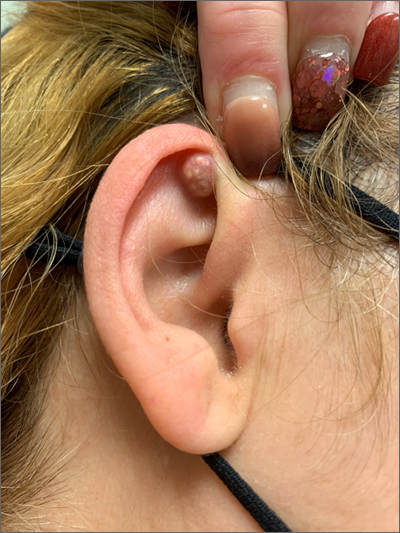
Pathology indicated a proliferation of basaloid cells with matrical differentiation in transition and “shadow” cells, pointing to a diagnosis of pilomatricoma.
Pilomatricoma, also known as pilomatrixoma, is a benign skin tumor associated with hair follicles. The lesions are most often found on the neck or head area but can occur on the arms, legs, or torso. They are usually slow growing, solitary, and painless. The frequency of occurrence is rare, accounting for less than 1% of all benign skin tumors.1
A mutation in the Catenin beta-1 (CTNNB1) gene is the most common cause of isolated pilomatricoma and is a somatic defect, meaning it is acquired, not inherited. The mutation of the CTNNB1 gene causes disruption of normal function and maturation of the hair follicle. This leads to rapid cell growth and uncontrolled division, resulting in the formation of the pilomatricoma.1
A comprehensive review performed in 2018 noted that only 16% of pilomatricomas were accurately diagnosed on clinical exam.1 Clues that point to the diagnosis of pilomatricoma are the irregular, whitish yellow spots just under the skin. In contrast, epidermoid cysts usually have a central pore and a ballotable feel. The expression of calcification and gritty material from the lesion in this case ruled out a diagnosis of an epidermoid cyst. The most common method of treatment is surgical removal.1
This patient was counseled regarding her diagnosis and given the option of a plastic surgery referral to excise the affected tissue in its entirety. She opted to wait and see if the growth would scar down and not return.
Image courtesy of Edward A. Jackson, MD. Text courtesy of Edward A. Jackson, MD, FAAFP, Advent Health Medical Group Family Medicine at East Orlando, FL, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Jones CD, Ho W, Robertson BF, et al. Pilomatrixoma: a comprehensive review of the literature. Am J Dermatopathol. 2018;40:631-641. doi: 10.1097/DAD.0000000000001118

Pathology indicated a proliferation of basaloid cells with matrical differentiation in transition and “shadow” cells, pointing to a diagnosis of pilomatricoma.
Pilomatricoma, also known as pilomatrixoma, is a benign skin tumor associated with hair follicles. The lesions are most often found on the neck or head area but can occur on the arms, legs, or torso. They are usually slow growing, solitary, and painless. The frequency of occurrence is rare, accounting for less than 1% of all benign skin tumors.1
A mutation in the Catenin beta-1 (CTNNB1) gene is the most common cause of isolated pilomatricoma and is a somatic defect, meaning it is acquired, not inherited. The mutation of the CTNNB1 gene causes disruption of normal function and maturation of the hair follicle. This leads to rapid cell growth and uncontrolled division, resulting in the formation of the pilomatricoma.1
A comprehensive review performed in 2018 noted that only 16% of pilomatricomas were accurately diagnosed on clinical exam.1 Clues that point to the diagnosis of pilomatricoma are the irregular, whitish yellow spots just under the skin. In contrast, epidermoid cysts usually have a central pore and a ballotable feel. The expression of calcification and gritty material from the lesion in this case ruled out a diagnosis of an epidermoid cyst. The most common method of treatment is surgical removal.1
This patient was counseled regarding her diagnosis and given the option of a plastic surgery referral to excise the affected tissue in its entirety. She opted to wait and see if the growth would scar down and not return.
Image courtesy of Edward A. Jackson, MD. Text courtesy of Edward A. Jackson, MD, FAAFP, Advent Health Medical Group Family Medicine at East Orlando, FL, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Pathology indicated a proliferation of basaloid cells with matrical differentiation in transition and “shadow” cells, pointing to a diagnosis of pilomatricoma.
Pilomatricoma, also known as pilomatrixoma, is a benign skin tumor associated with hair follicles. The lesions are most often found on the neck or head area but can occur on the arms, legs, or torso. They are usually slow growing, solitary, and painless. The frequency of occurrence is rare, accounting for less than 1% of all benign skin tumors.1
A mutation in the Catenin beta-1 (CTNNB1) gene is the most common cause of isolated pilomatricoma and is a somatic defect, meaning it is acquired, not inherited. The mutation of the CTNNB1 gene causes disruption of normal function and maturation of the hair follicle. This leads to rapid cell growth and uncontrolled division, resulting in the formation of the pilomatricoma.1
A comprehensive review performed in 2018 noted that only 16% of pilomatricomas were accurately diagnosed on clinical exam.1 Clues that point to the diagnosis of pilomatricoma are the irregular, whitish yellow spots just under the skin. In contrast, epidermoid cysts usually have a central pore and a ballotable feel. The expression of calcification and gritty material from the lesion in this case ruled out a diagnosis of an epidermoid cyst. The most common method of treatment is surgical removal.1
This patient was counseled regarding her diagnosis and given the option of a plastic surgery referral to excise the affected tissue in its entirety. She opted to wait and see if the growth would scar down and not return.
Image courtesy of Edward A. Jackson, MD. Text courtesy of Edward A. Jackson, MD, FAAFP, Advent Health Medical Group Family Medicine at East Orlando, FL, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Jones CD, Ho W, Robertson BF, et al. Pilomatrixoma: a comprehensive review of the literature. Am J Dermatopathol. 2018;40:631-641. doi: 10.1097/DAD.0000000000001118
1. Jones CD, Ho W, Robertson BF, et al. Pilomatrixoma: a comprehensive review of the literature. Am J Dermatopathol. 2018;40:631-641. doi: 10.1097/DAD.0000000000001118
Discharge within 24 hours of PCI can be safe in select STEMI
Highly selected low-risk patients can be safely sent home about 24 hours after successful percutaneous coronary intervention (PCI) for ST-segment elevation myocardial infarction (STEMI) when supported by intense, multidisciplinary virtual follow-up, a prospective study suggests for the first time.
The risk for major adverse cardiac events (MACE) in STEMI patients following an early hospital discharge (EHD) pathway was similar at 9 months to that seen for propensity-matched historic control subjects who met the same EHD criteria but were discharged later than 48 hours.
The stay in almost half (48%) the early discharge group was 24 hours or less, according to the study, published Dec. 13 in the Journal of the American College of Cardiology.
“We’ve shown that if we use appropriate risk criteria and instigate the appropriate, safe follow-up that it’s safe to select and discharge low-risk patients at an earlier time period, such as 24 hours,” senior author Daniel A. Jones, PhD, Barts Heart Centre, London, this news organization.
“Obviously, it’s one center in one city in the world,” he said. “Whether it’s applicable at other heart site centers, I believe it is, but I think we need more data to be able to change guidelines.”
Current European Society of Cardiology guidelines say that select patients should be considered for early discharge 48 to 72 hours after STEMI, but the COVID-19 pandemic incentivized the team to try and push that window.
“The COVID pandemic essentially brought a focus on resources, on minimizing the risk to our patient population in terms of catching COVID within hospital,” he said. “It became clear that to maintain the heart site service, we probably needed to get people out a bit quicker than we did before, so we came up with this pathway.”
Between March 2020 and June 2021, 600 patients presenting with STEMI were entered into the EHD pathway if they met the following pre-existing criteria for 48- to 72-hour discharge:
- Left ventricular ejection fraction 40% or greater
- Successful primary PCI with TIMI flow grade 3
- Absence of bystander disease requiring inpatient revascularization
- No recurrent ischemic symptoms
- No heart failure
- No significant arrhythmias
- No hemodynamic instability
- No significant comorbidity
- Suitable social circumstances for early discharge
The patients were given cardiac rehabilitation counseling over the phone within 48 hours and blood pressure machines if not available at home. At weeks 2 and 8, they spoke virtually with a dedicated cardiology advanced care practitioner who up-titrated medications and answered any questions. At week 12, they were seen by an interventional cardiologist or at a high-risk prevention clinic.
Their mean age was 59.2 years, 86% were male, the median symptom-to-balloon time was 80 minutes, and median door-to-balloon time was 50 minutes.
The early discharge patients were compared with 700 historic control subjects who met the EHD criteria and were discharged after 48 hours from Oct. 2018 to June 2021 and 560 patients discharged on standard-care pathways between April 2020 and June 2021.
Those discharged after 48 hours were more likely to have an anterior MI, multivessel disease, and multivessel PCI.
Comparable outcomes
The median length of stay was 24.6 hours (minimum 17 hours, maximum 40 hours) for the EHD group, 56.1 hours for historic control subjects, and 78.9 hours for the standard-care group.
The introduction of the EHD pathway significantly reduced the overall length of stay for all STEMI patients compared with the pre-pathway period of Oct. 2018 to March 2020 (median, 3 vs. 2 days; P < .0001).
Length of stay varied among patients; however, 420 patients stayed 1 less night in the hospital with the remaining patients staying about 8 to 12 fewer hours, resulting in approximate savings of £450,000, the authors note.
Over a median follow-up of 271 days, there were no cardiovascular deaths, two deaths from COVID-19, and a MACE rate of 1.2% (two deaths, three unscheduled revascularizations, and two further MI presentations) in the EHD group. That compares with a 0.7% mortality and 1.9% rate of MACE among historic control subjects, neither of which were significantly different.
There was also no difference in mortality (0.34% vs. 0.69%; P = .410) or MACE (1.2% vs. 1.9%; P = .342) among 560 pairs of propensity-matched EHD patients and historic control subjects.
Mortality was 4.1% in the standard-care group; cardiovascular mortality was 2.2%, and the rate of MACE was 8.6%.
When patients were surveyed, 85% were “satisfied” or “very satisfied” with the EHD pathway, whereas 73% of control and standard-care patients were satisfied with their care. Three-fourths of EHD patients also reported saving money and 62.5% saved time off work because of the virtual follow-up.
Judgment calls
“They didn’t really tell us much about the patients who didn’t qualify into this ultra–low-risk group but, obviously, it’s highly selected,” Cindy Grines, MD, Northside Hospital Cardiovascular Institute, Lawrenceville, Georgia, said in an interview. “In the U.S., you don’t get those chest pain onset-to-reperfusion in 80 minutes. So that was really kind of shocking.”
It also suggests that early discharge was applied to patients who may have had minimal myocardial damage from the STEMI, she suggested. “Even in their own hospital system, a lot of patients who met the criteria on paper were kept longer than 48 hours. So a lot of it’s a judgment call.”
Additional red flags where physicians may overrule the early discharge protocol are very late perfusion, advanced age, severe renal insufficiency, profound anemia, cardiac arrest requiring more than brief resuscitation, bleeding complications, or symptomatic coronavirus, Dr. Grines and J. Jeffrey Marshall, MD, also from Northside, observe in an accompanying editorial.
About 60% of patients were suitable for the EHD pathway, Dr. Jones said. “Typically, they are quite low risk, but we still had four in 10 anterior infarct, and about 25% had left ventricular function between 40% and 45%. So even though the majority are low risk, there are patients in there that you would consider to have had a decent infarct.”
“I think this is applicable to patients at most centers, and probably anywhere between a third to a fifth of all patients presenting to heart centers would be suitable for this discharge pathway,” he said.
Dr. Grines said the pathway is “definitely feasible” but there aren’t enough patients studied to know with 100% certainty whether it’s safe. A single observational study also isn’t enough to change guidelines, which in the United States do not comment on length of stay.
“In the ultra-low-risk patients – such as the ones where you got them in very early and you almost aborted the infarct or if it was a very small infarct – you can kind of treat them like an unstable angina patient, where you can do the PCI and potentially discharge them in 24 hours,” Dr. Grines said. “I think most of us might agree on that.”
“The other thing you have to weigh is the risk/benefit ratio,” she said. “If you have no beds available, you end up rationing care to some extent. So if you have a patient that’s otherwise doing well after a very small MI and have an emergency room full of people that need to be admitted and they’re sicker, then you end up making those judgment calls.”
Dr. Jones pointed out that current guidelines are based largely on observational data and that the team is planning to pilot the EHD pathway at five to 10 centers around the United Kingdom or potentially in Europe or the United States.
“This is an area where a [randomized controlled trial] RCT would be expensive, whereas a well-coordinated multicenter registry would probably provide enough information to change guidelines,” he said. “We’re not suggesting that every STEMI patient is suitable, but people that are low risk that you would already be considering for early discharge I think can go a bit quicker.”
Dr. Jones has received funding from the Barts Charity and financial support for blood pressure machines from the Barts Guild. First author Krishnaraj Rathod has received funding from the National Institute for Health and Research in the form of an Academic Clinical Lectureship. All other authors, Dr. Grines, and Dr. Marshall report having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Highly selected low-risk patients can be safely sent home about 24 hours after successful percutaneous coronary intervention (PCI) for ST-segment elevation myocardial infarction (STEMI) when supported by intense, multidisciplinary virtual follow-up, a prospective study suggests for the first time.
The risk for major adverse cardiac events (MACE) in STEMI patients following an early hospital discharge (EHD) pathway was similar at 9 months to that seen for propensity-matched historic control subjects who met the same EHD criteria but were discharged later than 48 hours.
The stay in almost half (48%) the early discharge group was 24 hours or less, according to the study, published Dec. 13 in the Journal of the American College of Cardiology.
“We’ve shown that if we use appropriate risk criteria and instigate the appropriate, safe follow-up that it’s safe to select and discharge low-risk patients at an earlier time period, such as 24 hours,” senior author Daniel A. Jones, PhD, Barts Heart Centre, London, this news organization.
“Obviously, it’s one center in one city in the world,” he said. “Whether it’s applicable at other heart site centers, I believe it is, but I think we need more data to be able to change guidelines.”
Current European Society of Cardiology guidelines say that select patients should be considered for early discharge 48 to 72 hours after STEMI, but the COVID-19 pandemic incentivized the team to try and push that window.
“The COVID pandemic essentially brought a focus on resources, on minimizing the risk to our patient population in terms of catching COVID within hospital,” he said. “It became clear that to maintain the heart site service, we probably needed to get people out a bit quicker than we did before, so we came up with this pathway.”
Between March 2020 and June 2021, 600 patients presenting with STEMI were entered into the EHD pathway if they met the following pre-existing criteria for 48- to 72-hour discharge:
- Left ventricular ejection fraction 40% or greater
- Successful primary PCI with TIMI flow grade 3
- Absence of bystander disease requiring inpatient revascularization
- No recurrent ischemic symptoms
- No heart failure
- No significant arrhythmias
- No hemodynamic instability
- No significant comorbidity
- Suitable social circumstances for early discharge
The patients were given cardiac rehabilitation counseling over the phone within 48 hours and blood pressure machines if not available at home. At weeks 2 and 8, they spoke virtually with a dedicated cardiology advanced care practitioner who up-titrated medications and answered any questions. At week 12, they were seen by an interventional cardiologist or at a high-risk prevention clinic.
Their mean age was 59.2 years, 86% were male, the median symptom-to-balloon time was 80 minutes, and median door-to-balloon time was 50 minutes.
The early discharge patients were compared with 700 historic control subjects who met the EHD criteria and were discharged after 48 hours from Oct. 2018 to June 2021 and 560 patients discharged on standard-care pathways between April 2020 and June 2021.
Those discharged after 48 hours were more likely to have an anterior MI, multivessel disease, and multivessel PCI.
Comparable outcomes
The median length of stay was 24.6 hours (minimum 17 hours, maximum 40 hours) for the EHD group, 56.1 hours for historic control subjects, and 78.9 hours for the standard-care group.
The introduction of the EHD pathway significantly reduced the overall length of stay for all STEMI patients compared with the pre-pathway period of Oct. 2018 to March 2020 (median, 3 vs. 2 days; P < .0001).
Length of stay varied among patients; however, 420 patients stayed 1 less night in the hospital with the remaining patients staying about 8 to 12 fewer hours, resulting in approximate savings of £450,000, the authors note.
Over a median follow-up of 271 days, there were no cardiovascular deaths, two deaths from COVID-19, and a MACE rate of 1.2% (two deaths, three unscheduled revascularizations, and two further MI presentations) in the EHD group. That compares with a 0.7% mortality and 1.9% rate of MACE among historic control subjects, neither of which were significantly different.
There was also no difference in mortality (0.34% vs. 0.69%; P = .410) or MACE (1.2% vs. 1.9%; P = .342) among 560 pairs of propensity-matched EHD patients and historic control subjects.
Mortality was 4.1% in the standard-care group; cardiovascular mortality was 2.2%, and the rate of MACE was 8.6%.
When patients were surveyed, 85% were “satisfied” or “very satisfied” with the EHD pathway, whereas 73% of control and standard-care patients were satisfied with their care. Three-fourths of EHD patients also reported saving money and 62.5% saved time off work because of the virtual follow-up.
Judgment calls
“They didn’t really tell us much about the patients who didn’t qualify into this ultra–low-risk group but, obviously, it’s highly selected,” Cindy Grines, MD, Northside Hospital Cardiovascular Institute, Lawrenceville, Georgia, said in an interview. “In the U.S., you don’t get those chest pain onset-to-reperfusion in 80 minutes. So that was really kind of shocking.”
It also suggests that early discharge was applied to patients who may have had minimal myocardial damage from the STEMI, she suggested. “Even in their own hospital system, a lot of patients who met the criteria on paper were kept longer than 48 hours. So a lot of it’s a judgment call.”
Additional red flags where physicians may overrule the early discharge protocol are very late perfusion, advanced age, severe renal insufficiency, profound anemia, cardiac arrest requiring more than brief resuscitation, bleeding complications, or symptomatic coronavirus, Dr. Grines and J. Jeffrey Marshall, MD, also from Northside, observe in an accompanying editorial.
About 60% of patients were suitable for the EHD pathway, Dr. Jones said. “Typically, they are quite low risk, but we still had four in 10 anterior infarct, and about 25% had left ventricular function between 40% and 45%. So even though the majority are low risk, there are patients in there that you would consider to have had a decent infarct.”
“I think this is applicable to patients at most centers, and probably anywhere between a third to a fifth of all patients presenting to heart centers would be suitable for this discharge pathway,” he said.
Dr. Grines said the pathway is “definitely feasible” but there aren’t enough patients studied to know with 100% certainty whether it’s safe. A single observational study also isn’t enough to change guidelines, which in the United States do not comment on length of stay.
“In the ultra-low-risk patients – such as the ones where you got them in very early and you almost aborted the infarct or if it was a very small infarct – you can kind of treat them like an unstable angina patient, where you can do the PCI and potentially discharge them in 24 hours,” Dr. Grines said. “I think most of us might agree on that.”
“The other thing you have to weigh is the risk/benefit ratio,” she said. “If you have no beds available, you end up rationing care to some extent. So if you have a patient that’s otherwise doing well after a very small MI and have an emergency room full of people that need to be admitted and they’re sicker, then you end up making those judgment calls.”
Dr. Jones pointed out that current guidelines are based largely on observational data and that the team is planning to pilot the EHD pathway at five to 10 centers around the United Kingdom or potentially in Europe or the United States.
“This is an area where a [randomized controlled trial] RCT would be expensive, whereas a well-coordinated multicenter registry would probably provide enough information to change guidelines,” he said. “We’re not suggesting that every STEMI patient is suitable, but people that are low risk that you would already be considering for early discharge I think can go a bit quicker.”
Dr. Jones has received funding from the Barts Charity and financial support for blood pressure machines from the Barts Guild. First author Krishnaraj Rathod has received funding from the National Institute for Health and Research in the form of an Academic Clinical Lectureship. All other authors, Dr. Grines, and Dr. Marshall report having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Highly selected low-risk patients can be safely sent home about 24 hours after successful percutaneous coronary intervention (PCI) for ST-segment elevation myocardial infarction (STEMI) when supported by intense, multidisciplinary virtual follow-up, a prospective study suggests for the first time.
The risk for major adverse cardiac events (MACE) in STEMI patients following an early hospital discharge (EHD) pathway was similar at 9 months to that seen for propensity-matched historic control subjects who met the same EHD criteria but were discharged later than 48 hours.
The stay in almost half (48%) the early discharge group was 24 hours or less, according to the study, published Dec. 13 in the Journal of the American College of Cardiology.
“We’ve shown that if we use appropriate risk criteria and instigate the appropriate, safe follow-up that it’s safe to select and discharge low-risk patients at an earlier time period, such as 24 hours,” senior author Daniel A. Jones, PhD, Barts Heart Centre, London, this news organization.
“Obviously, it’s one center in one city in the world,” he said. “Whether it’s applicable at other heart site centers, I believe it is, but I think we need more data to be able to change guidelines.”
Current European Society of Cardiology guidelines say that select patients should be considered for early discharge 48 to 72 hours after STEMI, but the COVID-19 pandemic incentivized the team to try and push that window.
“The COVID pandemic essentially brought a focus on resources, on minimizing the risk to our patient population in terms of catching COVID within hospital,” he said. “It became clear that to maintain the heart site service, we probably needed to get people out a bit quicker than we did before, so we came up with this pathway.”
Between March 2020 and June 2021, 600 patients presenting with STEMI were entered into the EHD pathway if they met the following pre-existing criteria for 48- to 72-hour discharge:
- Left ventricular ejection fraction 40% or greater
- Successful primary PCI with TIMI flow grade 3
- Absence of bystander disease requiring inpatient revascularization
- No recurrent ischemic symptoms
- No heart failure
- No significant arrhythmias
- No hemodynamic instability
- No significant comorbidity
- Suitable social circumstances for early discharge
The patients were given cardiac rehabilitation counseling over the phone within 48 hours and blood pressure machines if not available at home. At weeks 2 and 8, they spoke virtually with a dedicated cardiology advanced care practitioner who up-titrated medications and answered any questions. At week 12, they were seen by an interventional cardiologist or at a high-risk prevention clinic.
Their mean age was 59.2 years, 86% were male, the median symptom-to-balloon time was 80 minutes, and median door-to-balloon time was 50 minutes.
The early discharge patients were compared with 700 historic control subjects who met the EHD criteria and were discharged after 48 hours from Oct. 2018 to June 2021 and 560 patients discharged on standard-care pathways between April 2020 and June 2021.
Those discharged after 48 hours were more likely to have an anterior MI, multivessel disease, and multivessel PCI.
Comparable outcomes
The median length of stay was 24.6 hours (minimum 17 hours, maximum 40 hours) for the EHD group, 56.1 hours for historic control subjects, and 78.9 hours for the standard-care group.
The introduction of the EHD pathway significantly reduced the overall length of stay for all STEMI patients compared with the pre-pathway period of Oct. 2018 to March 2020 (median, 3 vs. 2 days; P < .0001).
Length of stay varied among patients; however, 420 patients stayed 1 less night in the hospital with the remaining patients staying about 8 to 12 fewer hours, resulting in approximate savings of £450,000, the authors note.
Over a median follow-up of 271 days, there were no cardiovascular deaths, two deaths from COVID-19, and a MACE rate of 1.2% (two deaths, three unscheduled revascularizations, and two further MI presentations) in the EHD group. That compares with a 0.7% mortality and 1.9% rate of MACE among historic control subjects, neither of which were significantly different.
There was also no difference in mortality (0.34% vs. 0.69%; P = .410) or MACE (1.2% vs. 1.9%; P = .342) among 560 pairs of propensity-matched EHD patients and historic control subjects.
Mortality was 4.1% in the standard-care group; cardiovascular mortality was 2.2%, and the rate of MACE was 8.6%.
When patients were surveyed, 85% were “satisfied” or “very satisfied” with the EHD pathway, whereas 73% of control and standard-care patients were satisfied with their care. Three-fourths of EHD patients also reported saving money and 62.5% saved time off work because of the virtual follow-up.
Judgment calls
“They didn’t really tell us much about the patients who didn’t qualify into this ultra–low-risk group but, obviously, it’s highly selected,” Cindy Grines, MD, Northside Hospital Cardiovascular Institute, Lawrenceville, Georgia, said in an interview. “In the U.S., you don’t get those chest pain onset-to-reperfusion in 80 minutes. So that was really kind of shocking.”
It also suggests that early discharge was applied to patients who may have had minimal myocardial damage from the STEMI, she suggested. “Even in their own hospital system, a lot of patients who met the criteria on paper were kept longer than 48 hours. So a lot of it’s a judgment call.”
Additional red flags where physicians may overrule the early discharge protocol are very late perfusion, advanced age, severe renal insufficiency, profound anemia, cardiac arrest requiring more than brief resuscitation, bleeding complications, or symptomatic coronavirus, Dr. Grines and J. Jeffrey Marshall, MD, also from Northside, observe in an accompanying editorial.
About 60% of patients were suitable for the EHD pathway, Dr. Jones said. “Typically, they are quite low risk, but we still had four in 10 anterior infarct, and about 25% had left ventricular function between 40% and 45%. So even though the majority are low risk, there are patients in there that you would consider to have had a decent infarct.”
“I think this is applicable to patients at most centers, and probably anywhere between a third to a fifth of all patients presenting to heart centers would be suitable for this discharge pathway,” he said.
Dr. Grines said the pathway is “definitely feasible” but there aren’t enough patients studied to know with 100% certainty whether it’s safe. A single observational study also isn’t enough to change guidelines, which in the United States do not comment on length of stay.
“In the ultra-low-risk patients – such as the ones where you got them in very early and you almost aborted the infarct or if it was a very small infarct – you can kind of treat them like an unstable angina patient, where you can do the PCI and potentially discharge them in 24 hours,” Dr. Grines said. “I think most of us might agree on that.”
“The other thing you have to weigh is the risk/benefit ratio,” she said. “If you have no beds available, you end up rationing care to some extent. So if you have a patient that’s otherwise doing well after a very small MI and have an emergency room full of people that need to be admitted and they’re sicker, then you end up making those judgment calls.”
Dr. Jones pointed out that current guidelines are based largely on observational data and that the team is planning to pilot the EHD pathway at five to 10 centers around the United Kingdom or potentially in Europe or the United States.
“This is an area where a [randomized controlled trial] RCT would be expensive, whereas a well-coordinated multicenter registry would probably provide enough information to change guidelines,” he said. “We’re not suggesting that every STEMI patient is suitable, but people that are low risk that you would already be considering for early discharge I think can go a bit quicker.”
Dr. Jones has received funding from the Barts Charity and financial support for blood pressure machines from the Barts Guild. First author Krishnaraj Rathod has received funding from the National Institute for Health and Research in the form of an Academic Clinical Lectureship. All other authors, Dr. Grines, and Dr. Marshall report having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
New update on perforation management
One of the most devastating complications in any endoscopic procedure is a perforation that may result in a life-threatening situation for the patient and abrupt termination of the intended procedure. The new AGA Clinical Practice Update on Endoscopic Management of Perforations in Gastrointestinal Tract: Expert Review offers a practical approach to prevent GI perforations, as well as detect subtle signs of and endoscopically manage them.
Best practice advice
- The area of perforation should be kept clean to prevent any spillage of gastrointestinal contents into the perforation by aspirating liquids and, if necessary, changing the patient position to bring the perforation into a nondependent location while minimizing insufflation of carbon dioxide to avoid compartment syndrome.
- Use of carbon dioxide for insufflation is encouraged for all endoscopic procedures, especially any endoscopic procedure with increased risk of perforation. If available, carbon dioxide should be used for all endoscopic procedures.
- All endoscopists should be aware of the procedures that carry an increased risk for perforation such as any dilation, foreign body removal, any per oral endoscopic myotomy (Zenker’s, esophageal, pyloric), stricture incision, thermal coagulation for hemostasis or tumor ablation, percutaneous endoscopic gastrostomy, ampullectomy, endoscopic mucosal resectio, endoscopic submucosal dissection, endoluminal stenting with self-expanding metal stent, full-thickness endoscopic resection, endoscopic retrograde cholangiopancreatography in surgically altered anatomy, endoscopic ultrasound (EUS)–guided biliary and pancreatic access, EUS-guided cystogastrostomy, and endoscopic gastroenterostomy using a lumen apposing metal stent.
- Urgent surgical consultation should be highly considered in all cases with perforation even when endoscopic repair is technically successful.
One of the most devastating complications in any endoscopic procedure is a perforation that may result in a life-threatening situation for the patient and abrupt termination of the intended procedure. The new AGA Clinical Practice Update on Endoscopic Management of Perforations in Gastrointestinal Tract: Expert Review offers a practical approach to prevent GI perforations, as well as detect subtle signs of and endoscopically manage them.
Best practice advice
- The area of perforation should be kept clean to prevent any spillage of gastrointestinal contents into the perforation by aspirating liquids and, if necessary, changing the patient position to bring the perforation into a nondependent location while minimizing insufflation of carbon dioxide to avoid compartment syndrome.
- Use of carbon dioxide for insufflation is encouraged for all endoscopic procedures, especially any endoscopic procedure with increased risk of perforation. If available, carbon dioxide should be used for all endoscopic procedures.
- All endoscopists should be aware of the procedures that carry an increased risk for perforation such as any dilation, foreign body removal, any per oral endoscopic myotomy (Zenker’s, esophageal, pyloric), stricture incision, thermal coagulation for hemostasis or tumor ablation, percutaneous endoscopic gastrostomy, ampullectomy, endoscopic mucosal resectio, endoscopic submucosal dissection, endoluminal stenting with self-expanding metal stent, full-thickness endoscopic resection, endoscopic retrograde cholangiopancreatography in surgically altered anatomy, endoscopic ultrasound (EUS)–guided biliary and pancreatic access, EUS-guided cystogastrostomy, and endoscopic gastroenterostomy using a lumen apposing metal stent.
- Urgent surgical consultation should be highly considered in all cases with perforation even when endoscopic repair is technically successful.
One of the most devastating complications in any endoscopic procedure is a perforation that may result in a life-threatening situation for the patient and abrupt termination of the intended procedure. The new AGA Clinical Practice Update on Endoscopic Management of Perforations in Gastrointestinal Tract: Expert Review offers a practical approach to prevent GI perforations, as well as detect subtle signs of and endoscopically manage them.
Best practice advice
- The area of perforation should be kept clean to prevent any spillage of gastrointestinal contents into the perforation by aspirating liquids and, if necessary, changing the patient position to bring the perforation into a nondependent location while minimizing insufflation of carbon dioxide to avoid compartment syndrome.
- Use of carbon dioxide for insufflation is encouraged for all endoscopic procedures, especially any endoscopic procedure with increased risk of perforation. If available, carbon dioxide should be used for all endoscopic procedures.
- All endoscopists should be aware of the procedures that carry an increased risk for perforation such as any dilation, foreign body removal, any per oral endoscopic myotomy (Zenker’s, esophageal, pyloric), stricture incision, thermal coagulation for hemostasis or tumor ablation, percutaneous endoscopic gastrostomy, ampullectomy, endoscopic mucosal resectio, endoscopic submucosal dissection, endoluminal stenting with self-expanding metal stent, full-thickness endoscopic resection, endoscopic retrograde cholangiopancreatography in surgically altered anatomy, endoscopic ultrasound (EUS)–guided biliary and pancreatic access, EUS-guided cystogastrostomy, and endoscopic gastroenterostomy using a lumen apposing metal stent.
- Urgent surgical consultation should be highly considered in all cases with perforation even when endoscopic repair is technically successful.
How to be charitable this year
After an unexpected and challenging year and as the new year begins, there are some ways you can make a difference, without giving up assets you may need today.
- Beneficiary designation. Adding the AGA Research Foundation as a beneficiary of your retirement plan or other assets is simple and you don’t need to leave your home to complete the gift. Since the funds are granted after your lifetime, you can maintain your family budget today.
- Gift in your will. With as little as one sentence, you can create a brighter tomorrow at the AGA Research Foundation without parting with assets today. You can designate the AGA Research Foundation as the beneficiary of a specific asset or, as many of our donors do to ensure that their family is protected, as the recipient of a percentage of the total estate.
- Grant from your donor advised fund (DAF). This popular one-stop giving solution lets you care for multiple causes and organizations with minimal paperwork. Consider it a charitable savings account where money waits until you’re ready to distribute it. And when you use your existing DAF to recommend a grant, it means you can invest in our future without impacting your budget today.
- Distribution from your IRA. If you are 70½ years or older, you can use your IRA to make a gift directly to the AGA Research Foundation without having to pay income tax on the distribution. Beginning in the year you turn 72, you must take your required minimum distribution (RMD). You can use a gift from your IRA to satisfy all or part of your RMD.
Learn more at https://gastro.planmylegacy.org.
After an unexpected and challenging year and as the new year begins, there are some ways you can make a difference, without giving up assets you may need today.
- Beneficiary designation. Adding the AGA Research Foundation as a beneficiary of your retirement plan or other assets is simple and you don’t need to leave your home to complete the gift. Since the funds are granted after your lifetime, you can maintain your family budget today.
- Gift in your will. With as little as one sentence, you can create a brighter tomorrow at the AGA Research Foundation without parting with assets today. You can designate the AGA Research Foundation as the beneficiary of a specific asset or, as many of our donors do to ensure that their family is protected, as the recipient of a percentage of the total estate.
- Grant from your donor advised fund (DAF). This popular one-stop giving solution lets you care for multiple causes and organizations with minimal paperwork. Consider it a charitable savings account where money waits until you’re ready to distribute it. And when you use your existing DAF to recommend a grant, it means you can invest in our future without impacting your budget today.
- Distribution from your IRA. If you are 70½ years or older, you can use your IRA to make a gift directly to the AGA Research Foundation without having to pay income tax on the distribution. Beginning in the year you turn 72, you must take your required minimum distribution (RMD). You can use a gift from your IRA to satisfy all or part of your RMD.
Learn more at https://gastro.planmylegacy.org.
After an unexpected and challenging year and as the new year begins, there are some ways you can make a difference, without giving up assets you may need today.
- Beneficiary designation. Adding the AGA Research Foundation as a beneficiary of your retirement plan or other assets is simple and you don’t need to leave your home to complete the gift. Since the funds are granted after your lifetime, you can maintain your family budget today.
- Gift in your will. With as little as one sentence, you can create a brighter tomorrow at the AGA Research Foundation without parting with assets today. You can designate the AGA Research Foundation as the beneficiary of a specific asset or, as many of our donors do to ensure that their family is protected, as the recipient of a percentage of the total estate.
- Grant from your donor advised fund (DAF). This popular one-stop giving solution lets you care for multiple causes and organizations with minimal paperwork. Consider it a charitable savings account where money waits until you’re ready to distribute it. And when you use your existing DAF to recommend a grant, it means you can invest in our future without impacting your budget today.
- Distribution from your IRA. If you are 70½ years or older, you can use your IRA to make a gift directly to the AGA Research Foundation without having to pay income tax on the distribution. Beginning in the year you turn 72, you must take your required minimum distribution (RMD). You can use a gift from your IRA to satisfy all or part of your RMD.
Learn more at https://gastro.planmylegacy.org.
How does atopic dermatitis present in skin of color?
“We see very heterogenous and broad clinical presentations across the diverse patient populations that we see,” Andrew F. Alexis, MD, MPH, said at the Revolutionizing Atopic Dermatitis symposium. “Some of these differences might be related to population variations in skin barrier function, immunologic factors, genetic factors, and environmental factors, which all interplay to produce variations in the clinical presentation and overall impact of AD. Many nongenetic factors also contribute to differences that we see, including some socioeconomic and other factors that feed into health disparities.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, discussed four main clinical features of AD in skin of color.
Erythema is less visible because it is masked by pigment
“There can be some masking of the redness and alteration of that color such that it doesn’t look bright red as it would in the background of lightly pigmented skin,” Dr. Alexis said. “Instead, the [AD lesions] have shades of grayish-red or grayish-brown or reddish-brown. It’s important to recognize this clinical presentation and look carefully and assess the patient – not just visually but with palpation and take into consideration symptomatology so that you don’t fall into the trap of calling an AD lesion postinflammatory hyperpigmentation. It’s also helpful to isolate the islands of normal or nonlesional skin and contrast that with the areas of lesional skin, to get a sense of how active and inflamed the areas are. Palpation really helps to appreciate the elevation of the lesions that are involved.”
Follicular accentuation
Morphological variants common in skin of color include the follicular variant or micropapular variant of AD. “You might just see a collection of papules that are 1-2 mm in size and pruritic and in typical sites of predilection [for] eczema,” he said. Prurigo nodularis–like lesions or prurigo nodularis in association with AD are also seen more frequently in skin of color.
Lichenification
The lichenoid variant of AD is characterized by a violaceous hue and other features that resemble lichen planus and has been reported to be more common in individuals of African descent. A prospective study of about 1,000 patients with AD seen over 2 years at a dermatology clinic in southeastern Nigeria found that 54% of patients had papular lichenoid lesions. In addition, 51% had elevated blood eosinophil counts, especially those with severe disease.
Dr. Alexis added that psoriasiform features have been reported in studies of East Asian populations with AD. These plaques may be more well demarcated and have clinical and histologic features that resemble psoriasis.
Dyspigmentation
One common feature across the spectrum of patients with skin of color “is the risk of longstanding pigmentary sequelae in the form of hyperpigmentation or hypopigmentation,” said Dr. Alexis, who is also vice chair for diversity and inclusion for the department of dermatology at Weill Cornell Medicine. “In very severe longstanding areas with chronic excoriation to the point of breaking of the skin, eroding of the skin, causing permanent damage to the melanocytes, dyspigmentation that resembles vitiligo can be seen. We can also see hypopigmentation as a consequence of topical corticosteroids, particularly those that are class I or class II and are used for prolonged periods of time.”
Dr. Alexis noted that delays in treatment and undertreatment can contribute to a higher risk of pigmentary and other long-term sequelae. “New therapies show promise in improving outcomes in AD patients with skin of color. When it comes to therapeutic responses, there are some post hoc studies that have investigated potential differences in safety and efficacy of the agents that have been recently approved. We clearly need more data to better understand if there are potential racial or ethnic differences.”
Dr. Alexis reported no relevant financial relationships.
Commentary by Lawrence F. Eichenfield, MD
Atopic dermatitis (AD) is highly heterogenous, with tremendous variations in extent, qualities of eczema, symptom complex, and physical presentation. Prior studies have reported disparities of care delivered to racial and ethnic minorities in the United States, as well as higher susceptibility to AD and odds of persistent disease into adulthood from child-onset AD. Recognizing some differences in presentation of AD in patients with skin of color is important as we select our therapeutic interventions, including assessing new treatments being added to our armamentarium. Erythema may be harder to notice in darker skin, but attempting to blanch the skin with pressure can help to assess the color and inflammation. Appreciating lichenoid changes, including papular and “micropapular” AD, and psoriasiform-like thickening in certain patients (reportedly more common in East Asian populations) are important as well. And dyspigmentation is an important aspect of the disease presentation and patient and parental concern, given both hypopigmentaton and hyperpigmentation commonly seen over the course of AD.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
“We see very heterogenous and broad clinical presentations across the diverse patient populations that we see,” Andrew F. Alexis, MD, MPH, said at the Revolutionizing Atopic Dermatitis symposium. “Some of these differences might be related to population variations in skin barrier function, immunologic factors, genetic factors, and environmental factors, which all interplay to produce variations in the clinical presentation and overall impact of AD. Many nongenetic factors also contribute to differences that we see, including some socioeconomic and other factors that feed into health disparities.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, discussed four main clinical features of AD in skin of color.
Erythema is less visible because it is masked by pigment
“There can be some masking of the redness and alteration of that color such that it doesn’t look bright red as it would in the background of lightly pigmented skin,” Dr. Alexis said. “Instead, the [AD lesions] have shades of grayish-red or grayish-brown or reddish-brown. It’s important to recognize this clinical presentation and look carefully and assess the patient – not just visually but with palpation and take into consideration symptomatology so that you don’t fall into the trap of calling an AD lesion postinflammatory hyperpigmentation. It’s also helpful to isolate the islands of normal or nonlesional skin and contrast that with the areas of lesional skin, to get a sense of how active and inflamed the areas are. Palpation really helps to appreciate the elevation of the lesions that are involved.”
Follicular accentuation
Morphological variants common in skin of color include the follicular variant or micropapular variant of AD. “You might just see a collection of papules that are 1-2 mm in size and pruritic and in typical sites of predilection [for] eczema,” he said. Prurigo nodularis–like lesions or prurigo nodularis in association with AD are also seen more frequently in skin of color.
Lichenification
The lichenoid variant of AD is characterized by a violaceous hue and other features that resemble lichen planus and has been reported to be more common in individuals of African descent. A prospective study of about 1,000 patients with AD seen over 2 years at a dermatology clinic in southeastern Nigeria found that 54% of patients had papular lichenoid lesions. In addition, 51% had elevated blood eosinophil counts, especially those with severe disease.
Dr. Alexis added that psoriasiform features have been reported in studies of East Asian populations with AD. These plaques may be more well demarcated and have clinical and histologic features that resemble psoriasis.
Dyspigmentation
One common feature across the spectrum of patients with skin of color “is the risk of longstanding pigmentary sequelae in the form of hyperpigmentation or hypopigmentation,” said Dr. Alexis, who is also vice chair for diversity and inclusion for the department of dermatology at Weill Cornell Medicine. “In very severe longstanding areas with chronic excoriation to the point of breaking of the skin, eroding of the skin, causing permanent damage to the melanocytes, dyspigmentation that resembles vitiligo can be seen. We can also see hypopigmentation as a consequence of topical corticosteroids, particularly those that are class I or class II and are used for prolonged periods of time.”
Dr. Alexis noted that delays in treatment and undertreatment can contribute to a higher risk of pigmentary and other long-term sequelae. “New therapies show promise in improving outcomes in AD patients with skin of color. When it comes to therapeutic responses, there are some post hoc studies that have investigated potential differences in safety and efficacy of the agents that have been recently approved. We clearly need more data to better understand if there are potential racial or ethnic differences.”
Dr. Alexis reported no relevant financial relationships.
Commentary by Lawrence F. Eichenfield, MD
Atopic dermatitis (AD) is highly heterogenous, with tremendous variations in extent, qualities of eczema, symptom complex, and physical presentation. Prior studies have reported disparities of care delivered to racial and ethnic minorities in the United States, as well as higher susceptibility to AD and odds of persistent disease into adulthood from child-onset AD. Recognizing some differences in presentation of AD in patients with skin of color is important as we select our therapeutic interventions, including assessing new treatments being added to our armamentarium. Erythema may be harder to notice in darker skin, but attempting to blanch the skin with pressure can help to assess the color and inflammation. Appreciating lichenoid changes, including papular and “micropapular” AD, and psoriasiform-like thickening in certain patients (reportedly more common in East Asian populations) are important as well. And dyspigmentation is an important aspect of the disease presentation and patient and parental concern, given both hypopigmentaton and hyperpigmentation commonly seen over the course of AD.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
“We see very heterogenous and broad clinical presentations across the diverse patient populations that we see,” Andrew F. Alexis, MD, MPH, said at the Revolutionizing Atopic Dermatitis symposium. “Some of these differences might be related to population variations in skin barrier function, immunologic factors, genetic factors, and environmental factors, which all interplay to produce variations in the clinical presentation and overall impact of AD. Many nongenetic factors also contribute to differences that we see, including some socioeconomic and other factors that feed into health disparities.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, discussed four main clinical features of AD in skin of color.
Erythema is less visible because it is masked by pigment
“There can be some masking of the redness and alteration of that color such that it doesn’t look bright red as it would in the background of lightly pigmented skin,” Dr. Alexis said. “Instead, the [AD lesions] have shades of grayish-red or grayish-brown or reddish-brown. It’s important to recognize this clinical presentation and look carefully and assess the patient – not just visually but with palpation and take into consideration symptomatology so that you don’t fall into the trap of calling an AD lesion postinflammatory hyperpigmentation. It’s also helpful to isolate the islands of normal or nonlesional skin and contrast that with the areas of lesional skin, to get a sense of how active and inflamed the areas are. Palpation really helps to appreciate the elevation of the lesions that are involved.”
Follicular accentuation
Morphological variants common in skin of color include the follicular variant or micropapular variant of AD. “You might just see a collection of papules that are 1-2 mm in size and pruritic and in typical sites of predilection [for] eczema,” he said. Prurigo nodularis–like lesions or prurigo nodularis in association with AD are also seen more frequently in skin of color.
Lichenification
The lichenoid variant of AD is characterized by a violaceous hue and other features that resemble lichen planus and has been reported to be more common in individuals of African descent. A prospective study of about 1,000 patients with AD seen over 2 years at a dermatology clinic in southeastern Nigeria found that 54% of patients had papular lichenoid lesions. In addition, 51% had elevated blood eosinophil counts, especially those with severe disease.
Dr. Alexis added that psoriasiform features have been reported in studies of East Asian populations with AD. These plaques may be more well demarcated and have clinical and histologic features that resemble psoriasis.
Dyspigmentation
One common feature across the spectrum of patients with skin of color “is the risk of longstanding pigmentary sequelae in the form of hyperpigmentation or hypopigmentation,” said Dr. Alexis, who is also vice chair for diversity and inclusion for the department of dermatology at Weill Cornell Medicine. “In very severe longstanding areas with chronic excoriation to the point of breaking of the skin, eroding of the skin, causing permanent damage to the melanocytes, dyspigmentation that resembles vitiligo can be seen. We can also see hypopigmentation as a consequence of topical corticosteroids, particularly those that are class I or class II and are used for prolonged periods of time.”
Dr. Alexis noted that delays in treatment and undertreatment can contribute to a higher risk of pigmentary and other long-term sequelae. “New therapies show promise in improving outcomes in AD patients with skin of color. When it comes to therapeutic responses, there are some post hoc studies that have investigated potential differences in safety and efficacy of the agents that have been recently approved. We clearly need more data to better understand if there are potential racial or ethnic differences.”
Dr. Alexis reported no relevant financial relationships.
Commentary by Lawrence F. Eichenfield, MD
Atopic dermatitis (AD) is highly heterogenous, with tremendous variations in extent, qualities of eczema, symptom complex, and physical presentation. Prior studies have reported disparities of care delivered to racial and ethnic minorities in the United States, as well as higher susceptibility to AD and odds of persistent disease into adulthood from child-onset AD. Recognizing some differences in presentation of AD in patients with skin of color is important as we select our therapeutic interventions, including assessing new treatments being added to our armamentarium. Erythema may be harder to notice in darker skin, but attempting to blanch the skin with pressure can help to assess the color and inflammation. Appreciating lichenoid changes, including papular and “micropapular” AD, and psoriasiform-like thickening in certain patients (reportedly more common in East Asian populations) are important as well. And dyspigmentation is an important aspect of the disease presentation and patient and parental concern, given both hypopigmentaton and hyperpigmentation commonly seen over the course of AD.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM REVOLUTIONIZING AD 2021