User login
Treatment of opioid use disorder with buprenorphine and methadone effective but underutilized
Background: Opioid use disorder (OUD) is a chronic disease with a high health care and societal burden from overdose and complications requiring hospitalization. Though clinical trials demonstrate effectiveness of methadone and buprenorphine, most patients do not have access to these medications.
Study design: Retrospective comparative effectiveness study.
Setting: Nationwide claims database of commercial and Medicare Advantage Enrollees.
Synopsis: A total of 40,885 individuals aged 16 years or older with OUD were studied in an intent-to-treat analysis of six unique treatment pathways. Though used in just 12.5% of patients, only treatment with buprenorphine or methadone was protective against overdose at 3 and 12 months, compared with no treatment. Additionally, these medications and nonintensive behavioral health counseling were associated with lower incidence of acute care episodes from complications of opioid use. Notably, those treated with buprenorphine or methadone for more than 6 months received the greatest benefit. With use of only health care encounters, the results may underestimate incidence of complications of ongoing opioid misuse.
Bottom line: Buprenorphine and methadone for OUD were associated with reduced overdose and opioid-related morbidity, compared with opioid antagonist therapy, inpatient treatment, or intensive outpatient behavioral interventions and should be considered a first-line treatment.
Citation: Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
Dr. Inofuentes is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: Opioid use disorder (OUD) is a chronic disease with a high health care and societal burden from overdose and complications requiring hospitalization. Though clinical trials demonstrate effectiveness of methadone and buprenorphine, most patients do not have access to these medications.
Study design: Retrospective comparative effectiveness study.
Setting: Nationwide claims database of commercial and Medicare Advantage Enrollees.
Synopsis: A total of 40,885 individuals aged 16 years or older with OUD were studied in an intent-to-treat analysis of six unique treatment pathways. Though used in just 12.5% of patients, only treatment with buprenorphine or methadone was protective against overdose at 3 and 12 months, compared with no treatment. Additionally, these medications and nonintensive behavioral health counseling were associated with lower incidence of acute care episodes from complications of opioid use. Notably, those treated with buprenorphine or methadone for more than 6 months received the greatest benefit. With use of only health care encounters, the results may underestimate incidence of complications of ongoing opioid misuse.
Bottom line: Buprenorphine and methadone for OUD were associated with reduced overdose and opioid-related morbidity, compared with opioid antagonist therapy, inpatient treatment, or intensive outpatient behavioral interventions and should be considered a first-line treatment.
Citation: Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
Dr. Inofuentes is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
Background: Opioid use disorder (OUD) is a chronic disease with a high health care and societal burden from overdose and complications requiring hospitalization. Though clinical trials demonstrate effectiveness of methadone and buprenorphine, most patients do not have access to these medications.
Study design: Retrospective comparative effectiveness study.
Setting: Nationwide claims database of commercial and Medicare Advantage Enrollees.
Synopsis: A total of 40,885 individuals aged 16 years or older with OUD were studied in an intent-to-treat analysis of six unique treatment pathways. Though used in just 12.5% of patients, only treatment with buprenorphine or methadone was protective against overdose at 3 and 12 months, compared with no treatment. Additionally, these medications and nonintensive behavioral health counseling were associated with lower incidence of acute care episodes from complications of opioid use. Notably, those treated with buprenorphine or methadone for more than 6 months received the greatest benefit. With use of only health care encounters, the results may underestimate incidence of complications of ongoing opioid misuse.
Bottom line: Buprenorphine and methadone for OUD were associated with reduced overdose and opioid-related morbidity, compared with opioid antagonist therapy, inpatient treatment, or intensive outpatient behavioral interventions and should be considered a first-line treatment.
Citation: Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
Dr. Inofuentes is assistant professor of medicine, section of hospital medicine, at the University of Virginia School of Medicine, Charlottesville.
‘I did nothing wrong’: MDs used their own sperm for fertility patients
Martin D. Greenberg, MD, was sued in May for secretly using his own sperm to inseminate one of his infertility patients 38 years earlier. The patient’s daughter found out last year when she used a DNA test from 23andme to learn about her family history. The 77-year-old New York gynecologist is retired in Florida.
All but one of the cases took place before 1990. Most of them came to light in the past few years, when biological offspring found out from home DNA tests.
“It is a gross betrayal of the trust that a patient puts in her doctor. It is an absolute perversion of the practice of medicine,” said Dev Sethi, a plaintiff attorney who sued a Tucson, Ariz., physician who inseminated at least 10 patients with his own sperm. “The hubris of a doctor to impregnate his own patient, in some effort to either save money or populate the world with his offspring, is striking.”
Why would these physicians use their own sperm and then keep it secret? Why were there so many of them? When their offspring now try to communicate with them, do they want to have a relationship? And how do they react when they’re found out?
The doctors’ behavior mystifies Sigal Klipstein, MD, a reproductive endocrinologist in Hoffman Estates, Ill., who is chair of the ethics committee of the American Society for Reproductive Medicine.
“These doctors lived with secrets for many years. How do you live with that as a doctor?” said Dr. Klipstein, who was still in high school when most of these cases occurred. “It surprises me that anybody would do this.”
Lack of training and lots of secrecy
Were these physicians particularly selfish or egotistical? Or was expedience the prime motivation?
At the time, there was little training in the techniques and ethics of infertility care, said Jody Madeira, JD, PhD, a law professor at Indiana University, Bloomington, who has closely studied the doctors.
“Many of them were ob.gyns., but they did not take CME courses for this work,” she said. The subspecialty of reproductive endocrinology and infertility was just beginning in the early 1970s, according an ASRM spokesman.
Treatment of infertility was a rather hush-hush topic at that time, which made it easier to be deceptive. In 1955, an Illinois court held that artificial insemination constituted adultery. “The social stigma resulting from the practice forces the parents to keep secret the infant’s origin,” a law review article from 1955 stated.
“In the 1950s and 1960s and even into the 1970s and 1980s, infertility treatments were considered shameful, and patients were often advised to keep their treatment to themselves,” Dr. Madeira said. “With everything so secret, it was easy to be deceptive.”
The field has become more sophisticated since then, Dr. Klipstein said. “For known donors, there is a legal contract between the recipient and donor. And it is no longer possible to be an anonymous donor. People can find you through DNA tests.”
Owing to changes in the field as well as the growing likelihood of being caught through DNA tests, most experts believe that rampant infertility fraud ended long ago.
How they were found out
When the doctors were active, there was little risk of being exposed. In those times, paternity tests were based on broad factors such as blood type and were unreliable. More accurate DNA tests were underway, but the doctors’ offspring did not think of using them because they suspected nothing.
Most of the doctors’ deeds only came to light with the rise of a new industry – home DNA testing for people who are curious about their family background. First came 23andme in 2007, then Ancestry.com in 2015. The number of people being tested reached almost 2 million in 2016, 7 million in 2017, and 30 million in 2020.
As more people entered company databases, it became easier to pinpoint biological fathers through other relatives. This explains how doctors who had not taken a home DNA test were identified.
The home tests have been shown to be highly accurate. None of the results for doctors accused of using their own sperm have proven to be false, and courts recognize similar DNA tests as proof of paternity.
But when found out, many of the physicians disputed the results and acted as if they could still keep their secret. “I don’t deny it; I don’t admit it,” Paul Brennan Jones, MD, a Colorado doctor, said when he was accused of siring eight children through his infertility patients decades before. Asked whether he would provide a DNA sample, the 80-year-old doctor responded: “No ... because I don’t want to have any incriminating evidence against me.”
How often did it happen?
Donor Deceived, a website that monitors these cases, reports 32 cases of physicians surreptitiously providing sperm to their patients. Eleven of the doctors are linked to 1 known offspring, two are linked to more than 75 offspring, one to 15, one to 10, three to 9, three to 7, and two to 5.
“It’s unlikely that any of the doctors did it just once,” said Adam B. Wolf, a San Francisco attorney who is representing the plaintiff in the Greenberg case. “It’s happened before. When doctors get the idea to do something crazy, they do it multiple times.”
Mr. Wolf believes that, because most people haven’t taken a DNA test, there are many more biological children of infertility doctors who have yet to come forward.
Many of the doctors who were found out have negotiated settlements with patients, under which they pay undisclosed sums of money in exchange for the patient’s keeping silent. Mr. Wolf said that, of the two dozen victims of sperm-donor doctors his law firm has represented, all but three have settled.
“We give an opportunity to the doctor to resolve the claims without having to publicly out this person for using his own sperm in his patients,” Mr. Wolf said. “Most doctors jump at the opportunity to not be known as the kind of person who would do that.”
Cases about to go to trial have been withdrawn because of being settled. In May, a case against Gerald E. Mortimer, MD, in Idaho, was dismissed after 3 years of litigation. The judge had made some key decisions that made it less likely that Dr. Mortimer would win. Dr. Mortimer’s biological daughter filed the initial case. She alleged medical negligence, failure to obtain informed consent, fraud, battery, intentional infliction of emotional distress, and several other causes of action.
Dr. Madeira objects to the use of confidential settlements, because other offspring cannot be alerted. But she also believes that, as more people find out about their parentage through DNA tests, it will be harder for accused doctors to make confidential settlements with all of them, and the doctors will eventually be identified.
In settlements, offspring ask for the medical histories of these doctors. So far, offspring have linked the development of Tay-Sachs disease, cystic fibrosis, and ovarian cancer with these doctors.
Denial: Physicians’ most frequent reaction
Once identified, most of the doctors denied the charge. When Gary Phillip Wood, MD, of Arkansas, was tracked down by his biological son, Dr. Wood insisted he had had a vasectomy years before the man was born but still would not agree to a DNA test. He died in April 2021.
None of the identified sperm doctors were interested in having a relationship with their newly identified offspring. When Gary Vandenberg, MD, of California, was contacted by his biological daughter, he abruptly ended the conversation, wishing her “good luck in life,” she recalled. “When I first found out, I was very suicidal. I did not want this existence. I still have those days. My husband had to take off work and stay home quite a bit to make sure I didn’t do anything to myself.”
When Gary Don Davis, MD, of Idaho, was asked about his paternity, he replied: “Let me check on that. Goodbye.” He could not be reached after that, and he died a few months later.
The accused doctors often have no medical records of their work. Dr. Wood said that all his records had been destroyed, and Dr. Greenberg said he did not have any records on his accuser and doubted that he had ever treated her. A 1977 survey found that more than half of infertility doctors did not keep any medical records so as to preserve the donor’s anonymity.
Many of the accused doctors said they used their own sperm because they were deeply committed to helping their patients. At one physician’s trial, his defense attorney said: “If Cecil made any mistakes, it was in losing his objectivity and trying so hard to get patients pregnant.”
Was it really ethically wrong?
Many of the doctors don’t accept that they did any harm, says Julie D. Cantor, MD, JD, a former adjunct professor at the University of California, Los Angeles. “These doctors seemed to be thinking: ‘The patient wanted to get pregnant and have a baby, and that’s what happened, so no harm done.’ But the entire interaction is based on a lie.”
The doctors also had the problem of having to use fresh sperm rather than frozen sperm, as is used today. Sperm had to be used within hours of being produced. If the donor did not show up at the time of the appointment, the doctor might decide to keep the appointment with the patient anyway and provide his own sperm.
However, “these doctors didn’t have to use their own sperm,” Mr. Wolf said. “They could have rescheduled the appointment until a new donor could be found.”
Some say that the doctors seemed to have had a very high opinion of themselves and their own sperm. “Some of them had savior complexes,” Dr. Madeira said. “They seemed to be thinking: ‘I’m giving the gift of life, and I’m the only one who can really do it, because I have great genes.’ ”
When Kim McMorries, MD, of Texas, was confronted with the fact that he had donated sperm 33 years before, he insisted that it was ethical at the time. “When this occurred, it was not considered wrong,” he wrote in an email to his biological daughter.
Today, doctors are bound by the doctrine of informed consent, which holds that patients should be informed about all steps taken in their care. The term was coined by a judge in 1960, and it took some time for some in the medical world to fully accept informed consent. Still, Dr. Madeira asserts it was always unethical to secretly fertilize patients.
“Even in the more paternalistic era of the 1970s and 1980s, it was not right to lie to your patients about such an important part of their lives,” she said.
Some sperm doctors insisted that they had received informed consent when the patient agreed to use an anonymous donor. “Dr. Kiken did that which he was asked to do,” wrote the attorneys for Michael S. Kiken, MD, of Virginia. “Anonymous donor meant that the patient would not know the donor’s identity, he would be anonymous to her.”
Dr. Madeira does not accept this argument either. “The doctor may have thought it was understood that he could be the anonymous person, but the patients did not see it that way,” she said. “They were not expecting the anonymous donor would be their own doctor.”
“I think what happened is a crime,” said Dr. Klipstein. “It’s an ethical violation, a fracture in the trust between doctor and patient.”
Existing laws, however, don’t make it easy to prosecute the doctors. When lawsuits are filed against these doctors, “you have to shoehorn existing statutes to fit the facts, and that may not be a terrific fit,” Dr. Cantor said.
The doctors have been charged with battery, fraud, negligence, breach of duty, unjust enrichment, and rape. But none of them have been found guilty specifically of secretly using their own sperm. Two of the doctors were convicted, but for other offenses, such as perjury for denying their involvement.
Since 2019, five states – Arizona, Colorado, Florida, Indiana, and Texas – have passed statutes specifically outlawing infertility fraud. In addition, a 1995 California law requires identifying the sperm donor.
It may be difficult, however, to apply these new laws to offenses by aging sperm doctors that happened decades ago. “Some states have inflexible limits on the amount of time in which you can sue, even if you didn’t know about the problem until recently,” Dr. Madeira said. “Texas, for example, allows civil lawsuits only up to 10 years after commission.”
Before the fertility fraud physicians can be brought to justice, many of them might just fade away.
A version of this article first appeared on Medscape.com.
Martin D. Greenberg, MD, was sued in May for secretly using his own sperm to inseminate one of his infertility patients 38 years earlier. The patient’s daughter found out last year when she used a DNA test from 23andme to learn about her family history. The 77-year-old New York gynecologist is retired in Florida.
All but one of the cases took place before 1990. Most of them came to light in the past few years, when biological offspring found out from home DNA tests.
“It is a gross betrayal of the trust that a patient puts in her doctor. It is an absolute perversion of the practice of medicine,” said Dev Sethi, a plaintiff attorney who sued a Tucson, Ariz., physician who inseminated at least 10 patients with his own sperm. “The hubris of a doctor to impregnate his own patient, in some effort to either save money or populate the world with his offspring, is striking.”
Why would these physicians use their own sperm and then keep it secret? Why were there so many of them? When their offspring now try to communicate with them, do they want to have a relationship? And how do they react when they’re found out?
The doctors’ behavior mystifies Sigal Klipstein, MD, a reproductive endocrinologist in Hoffman Estates, Ill., who is chair of the ethics committee of the American Society for Reproductive Medicine.
“These doctors lived with secrets for many years. How do you live with that as a doctor?” said Dr. Klipstein, who was still in high school when most of these cases occurred. “It surprises me that anybody would do this.”
Lack of training and lots of secrecy
Were these physicians particularly selfish or egotistical? Or was expedience the prime motivation?
At the time, there was little training in the techniques and ethics of infertility care, said Jody Madeira, JD, PhD, a law professor at Indiana University, Bloomington, who has closely studied the doctors.
“Many of them were ob.gyns., but they did not take CME courses for this work,” she said. The subspecialty of reproductive endocrinology and infertility was just beginning in the early 1970s, according an ASRM spokesman.
Treatment of infertility was a rather hush-hush topic at that time, which made it easier to be deceptive. In 1955, an Illinois court held that artificial insemination constituted adultery. “The social stigma resulting from the practice forces the parents to keep secret the infant’s origin,” a law review article from 1955 stated.
“In the 1950s and 1960s and even into the 1970s and 1980s, infertility treatments were considered shameful, and patients were often advised to keep their treatment to themselves,” Dr. Madeira said. “With everything so secret, it was easy to be deceptive.”
The field has become more sophisticated since then, Dr. Klipstein said. “For known donors, there is a legal contract between the recipient and donor. And it is no longer possible to be an anonymous donor. People can find you through DNA tests.”
Owing to changes in the field as well as the growing likelihood of being caught through DNA tests, most experts believe that rampant infertility fraud ended long ago.
How they were found out
When the doctors were active, there was little risk of being exposed. In those times, paternity tests were based on broad factors such as blood type and were unreliable. More accurate DNA tests were underway, but the doctors’ offspring did not think of using them because they suspected nothing.
Most of the doctors’ deeds only came to light with the rise of a new industry – home DNA testing for people who are curious about their family background. First came 23andme in 2007, then Ancestry.com in 2015. The number of people being tested reached almost 2 million in 2016, 7 million in 2017, and 30 million in 2020.
As more people entered company databases, it became easier to pinpoint biological fathers through other relatives. This explains how doctors who had not taken a home DNA test were identified.
The home tests have been shown to be highly accurate. None of the results for doctors accused of using their own sperm have proven to be false, and courts recognize similar DNA tests as proof of paternity.
But when found out, many of the physicians disputed the results and acted as if they could still keep their secret. “I don’t deny it; I don’t admit it,” Paul Brennan Jones, MD, a Colorado doctor, said when he was accused of siring eight children through his infertility patients decades before. Asked whether he would provide a DNA sample, the 80-year-old doctor responded: “No ... because I don’t want to have any incriminating evidence against me.”
How often did it happen?
Donor Deceived, a website that monitors these cases, reports 32 cases of physicians surreptitiously providing sperm to their patients. Eleven of the doctors are linked to 1 known offspring, two are linked to more than 75 offspring, one to 15, one to 10, three to 9, three to 7, and two to 5.
“It’s unlikely that any of the doctors did it just once,” said Adam B. Wolf, a San Francisco attorney who is representing the plaintiff in the Greenberg case. “It’s happened before. When doctors get the idea to do something crazy, they do it multiple times.”
Mr. Wolf believes that, because most people haven’t taken a DNA test, there are many more biological children of infertility doctors who have yet to come forward.
Many of the doctors who were found out have negotiated settlements with patients, under which they pay undisclosed sums of money in exchange for the patient’s keeping silent. Mr. Wolf said that, of the two dozen victims of sperm-donor doctors his law firm has represented, all but three have settled.
“We give an opportunity to the doctor to resolve the claims without having to publicly out this person for using his own sperm in his patients,” Mr. Wolf said. “Most doctors jump at the opportunity to not be known as the kind of person who would do that.”
Cases about to go to trial have been withdrawn because of being settled. In May, a case against Gerald E. Mortimer, MD, in Idaho, was dismissed after 3 years of litigation. The judge had made some key decisions that made it less likely that Dr. Mortimer would win. Dr. Mortimer’s biological daughter filed the initial case. She alleged medical negligence, failure to obtain informed consent, fraud, battery, intentional infliction of emotional distress, and several other causes of action.
Dr. Madeira objects to the use of confidential settlements, because other offspring cannot be alerted. But she also believes that, as more people find out about their parentage through DNA tests, it will be harder for accused doctors to make confidential settlements with all of them, and the doctors will eventually be identified.
In settlements, offspring ask for the medical histories of these doctors. So far, offspring have linked the development of Tay-Sachs disease, cystic fibrosis, and ovarian cancer with these doctors.
Denial: Physicians’ most frequent reaction
Once identified, most of the doctors denied the charge. When Gary Phillip Wood, MD, of Arkansas, was tracked down by his biological son, Dr. Wood insisted he had had a vasectomy years before the man was born but still would not agree to a DNA test. He died in April 2021.
None of the identified sperm doctors were interested in having a relationship with their newly identified offspring. When Gary Vandenberg, MD, of California, was contacted by his biological daughter, he abruptly ended the conversation, wishing her “good luck in life,” she recalled. “When I first found out, I was very suicidal. I did not want this existence. I still have those days. My husband had to take off work and stay home quite a bit to make sure I didn’t do anything to myself.”
When Gary Don Davis, MD, of Idaho, was asked about his paternity, he replied: “Let me check on that. Goodbye.” He could not be reached after that, and he died a few months later.
The accused doctors often have no medical records of their work. Dr. Wood said that all his records had been destroyed, and Dr. Greenberg said he did not have any records on his accuser and doubted that he had ever treated her. A 1977 survey found that more than half of infertility doctors did not keep any medical records so as to preserve the donor’s anonymity.
Many of the accused doctors said they used their own sperm because they were deeply committed to helping their patients. At one physician’s trial, his defense attorney said: “If Cecil made any mistakes, it was in losing his objectivity and trying so hard to get patients pregnant.”
Was it really ethically wrong?
Many of the doctors don’t accept that they did any harm, says Julie D. Cantor, MD, JD, a former adjunct professor at the University of California, Los Angeles. “These doctors seemed to be thinking: ‘The patient wanted to get pregnant and have a baby, and that’s what happened, so no harm done.’ But the entire interaction is based on a lie.”
The doctors also had the problem of having to use fresh sperm rather than frozen sperm, as is used today. Sperm had to be used within hours of being produced. If the donor did not show up at the time of the appointment, the doctor might decide to keep the appointment with the patient anyway and provide his own sperm.
However, “these doctors didn’t have to use their own sperm,” Mr. Wolf said. “They could have rescheduled the appointment until a new donor could be found.”
Some say that the doctors seemed to have had a very high opinion of themselves and their own sperm. “Some of them had savior complexes,” Dr. Madeira said. “They seemed to be thinking: ‘I’m giving the gift of life, and I’m the only one who can really do it, because I have great genes.’ ”
When Kim McMorries, MD, of Texas, was confronted with the fact that he had donated sperm 33 years before, he insisted that it was ethical at the time. “When this occurred, it was not considered wrong,” he wrote in an email to his biological daughter.
Today, doctors are bound by the doctrine of informed consent, which holds that patients should be informed about all steps taken in their care. The term was coined by a judge in 1960, and it took some time for some in the medical world to fully accept informed consent. Still, Dr. Madeira asserts it was always unethical to secretly fertilize patients.
“Even in the more paternalistic era of the 1970s and 1980s, it was not right to lie to your patients about such an important part of their lives,” she said.
Some sperm doctors insisted that they had received informed consent when the patient agreed to use an anonymous donor. “Dr. Kiken did that which he was asked to do,” wrote the attorneys for Michael S. Kiken, MD, of Virginia. “Anonymous donor meant that the patient would not know the donor’s identity, he would be anonymous to her.”
Dr. Madeira does not accept this argument either. “The doctor may have thought it was understood that he could be the anonymous person, but the patients did not see it that way,” she said. “They were not expecting the anonymous donor would be their own doctor.”
“I think what happened is a crime,” said Dr. Klipstein. “It’s an ethical violation, a fracture in the trust between doctor and patient.”
Existing laws, however, don’t make it easy to prosecute the doctors. When lawsuits are filed against these doctors, “you have to shoehorn existing statutes to fit the facts, and that may not be a terrific fit,” Dr. Cantor said.
The doctors have been charged with battery, fraud, negligence, breach of duty, unjust enrichment, and rape. But none of them have been found guilty specifically of secretly using their own sperm. Two of the doctors were convicted, but for other offenses, such as perjury for denying their involvement.
Since 2019, five states – Arizona, Colorado, Florida, Indiana, and Texas – have passed statutes specifically outlawing infertility fraud. In addition, a 1995 California law requires identifying the sperm donor.
It may be difficult, however, to apply these new laws to offenses by aging sperm doctors that happened decades ago. “Some states have inflexible limits on the amount of time in which you can sue, even if you didn’t know about the problem until recently,” Dr. Madeira said. “Texas, for example, allows civil lawsuits only up to 10 years after commission.”
Before the fertility fraud physicians can be brought to justice, many of them might just fade away.
A version of this article first appeared on Medscape.com.
Martin D. Greenberg, MD, was sued in May for secretly using his own sperm to inseminate one of his infertility patients 38 years earlier. The patient’s daughter found out last year when she used a DNA test from 23andme to learn about her family history. The 77-year-old New York gynecologist is retired in Florida.
All but one of the cases took place before 1990. Most of them came to light in the past few years, when biological offspring found out from home DNA tests.
“It is a gross betrayal of the trust that a patient puts in her doctor. It is an absolute perversion of the practice of medicine,” said Dev Sethi, a plaintiff attorney who sued a Tucson, Ariz., physician who inseminated at least 10 patients with his own sperm. “The hubris of a doctor to impregnate his own patient, in some effort to either save money or populate the world with his offspring, is striking.”
Why would these physicians use their own sperm and then keep it secret? Why were there so many of them? When their offspring now try to communicate with them, do they want to have a relationship? And how do they react when they’re found out?
The doctors’ behavior mystifies Sigal Klipstein, MD, a reproductive endocrinologist in Hoffman Estates, Ill., who is chair of the ethics committee of the American Society for Reproductive Medicine.
“These doctors lived with secrets for many years. How do you live with that as a doctor?” said Dr. Klipstein, who was still in high school when most of these cases occurred. “It surprises me that anybody would do this.”
Lack of training and lots of secrecy
Were these physicians particularly selfish or egotistical? Or was expedience the prime motivation?
At the time, there was little training in the techniques and ethics of infertility care, said Jody Madeira, JD, PhD, a law professor at Indiana University, Bloomington, who has closely studied the doctors.
“Many of them were ob.gyns., but they did not take CME courses for this work,” she said. The subspecialty of reproductive endocrinology and infertility was just beginning in the early 1970s, according an ASRM spokesman.
Treatment of infertility was a rather hush-hush topic at that time, which made it easier to be deceptive. In 1955, an Illinois court held that artificial insemination constituted adultery. “The social stigma resulting from the practice forces the parents to keep secret the infant’s origin,” a law review article from 1955 stated.
“In the 1950s and 1960s and even into the 1970s and 1980s, infertility treatments were considered shameful, and patients were often advised to keep their treatment to themselves,” Dr. Madeira said. “With everything so secret, it was easy to be deceptive.”
The field has become more sophisticated since then, Dr. Klipstein said. “For known donors, there is a legal contract between the recipient and donor. And it is no longer possible to be an anonymous donor. People can find you through DNA tests.”
Owing to changes in the field as well as the growing likelihood of being caught through DNA tests, most experts believe that rampant infertility fraud ended long ago.
How they were found out
When the doctors were active, there was little risk of being exposed. In those times, paternity tests were based on broad factors such as blood type and were unreliable. More accurate DNA tests were underway, but the doctors’ offspring did not think of using them because they suspected nothing.
Most of the doctors’ deeds only came to light with the rise of a new industry – home DNA testing for people who are curious about their family background. First came 23andme in 2007, then Ancestry.com in 2015. The number of people being tested reached almost 2 million in 2016, 7 million in 2017, and 30 million in 2020.
As more people entered company databases, it became easier to pinpoint biological fathers through other relatives. This explains how doctors who had not taken a home DNA test were identified.
The home tests have been shown to be highly accurate. None of the results for doctors accused of using their own sperm have proven to be false, and courts recognize similar DNA tests as proof of paternity.
But when found out, many of the physicians disputed the results and acted as if they could still keep their secret. “I don’t deny it; I don’t admit it,” Paul Brennan Jones, MD, a Colorado doctor, said when he was accused of siring eight children through his infertility patients decades before. Asked whether he would provide a DNA sample, the 80-year-old doctor responded: “No ... because I don’t want to have any incriminating evidence against me.”
How often did it happen?
Donor Deceived, a website that monitors these cases, reports 32 cases of physicians surreptitiously providing sperm to their patients. Eleven of the doctors are linked to 1 known offspring, two are linked to more than 75 offspring, one to 15, one to 10, three to 9, three to 7, and two to 5.
“It’s unlikely that any of the doctors did it just once,” said Adam B. Wolf, a San Francisco attorney who is representing the plaintiff in the Greenberg case. “It’s happened before. When doctors get the idea to do something crazy, they do it multiple times.”
Mr. Wolf believes that, because most people haven’t taken a DNA test, there are many more biological children of infertility doctors who have yet to come forward.
Many of the doctors who were found out have negotiated settlements with patients, under which they pay undisclosed sums of money in exchange for the patient’s keeping silent. Mr. Wolf said that, of the two dozen victims of sperm-donor doctors his law firm has represented, all but three have settled.
“We give an opportunity to the doctor to resolve the claims without having to publicly out this person for using his own sperm in his patients,” Mr. Wolf said. “Most doctors jump at the opportunity to not be known as the kind of person who would do that.”
Cases about to go to trial have been withdrawn because of being settled. In May, a case against Gerald E. Mortimer, MD, in Idaho, was dismissed after 3 years of litigation. The judge had made some key decisions that made it less likely that Dr. Mortimer would win. Dr. Mortimer’s biological daughter filed the initial case. She alleged medical negligence, failure to obtain informed consent, fraud, battery, intentional infliction of emotional distress, and several other causes of action.
Dr. Madeira objects to the use of confidential settlements, because other offspring cannot be alerted. But she also believes that, as more people find out about their parentage through DNA tests, it will be harder for accused doctors to make confidential settlements with all of them, and the doctors will eventually be identified.
In settlements, offspring ask for the medical histories of these doctors. So far, offspring have linked the development of Tay-Sachs disease, cystic fibrosis, and ovarian cancer with these doctors.
Denial: Physicians’ most frequent reaction
Once identified, most of the doctors denied the charge. When Gary Phillip Wood, MD, of Arkansas, was tracked down by his biological son, Dr. Wood insisted he had had a vasectomy years before the man was born but still would not agree to a DNA test. He died in April 2021.
None of the identified sperm doctors were interested in having a relationship with their newly identified offspring. When Gary Vandenberg, MD, of California, was contacted by his biological daughter, he abruptly ended the conversation, wishing her “good luck in life,” she recalled. “When I first found out, I was very suicidal. I did not want this existence. I still have those days. My husband had to take off work and stay home quite a bit to make sure I didn’t do anything to myself.”
When Gary Don Davis, MD, of Idaho, was asked about his paternity, he replied: “Let me check on that. Goodbye.” He could not be reached after that, and he died a few months later.
The accused doctors often have no medical records of their work. Dr. Wood said that all his records had been destroyed, and Dr. Greenberg said he did not have any records on his accuser and doubted that he had ever treated her. A 1977 survey found that more than half of infertility doctors did not keep any medical records so as to preserve the donor’s anonymity.
Many of the accused doctors said they used their own sperm because they were deeply committed to helping their patients. At one physician’s trial, his defense attorney said: “If Cecil made any mistakes, it was in losing his objectivity and trying so hard to get patients pregnant.”
Was it really ethically wrong?
Many of the doctors don’t accept that they did any harm, says Julie D. Cantor, MD, JD, a former adjunct professor at the University of California, Los Angeles. “These doctors seemed to be thinking: ‘The patient wanted to get pregnant and have a baby, and that’s what happened, so no harm done.’ But the entire interaction is based on a lie.”
The doctors also had the problem of having to use fresh sperm rather than frozen sperm, as is used today. Sperm had to be used within hours of being produced. If the donor did not show up at the time of the appointment, the doctor might decide to keep the appointment with the patient anyway and provide his own sperm.
However, “these doctors didn’t have to use their own sperm,” Mr. Wolf said. “They could have rescheduled the appointment until a new donor could be found.”
Some say that the doctors seemed to have had a very high opinion of themselves and their own sperm. “Some of them had savior complexes,” Dr. Madeira said. “They seemed to be thinking: ‘I’m giving the gift of life, and I’m the only one who can really do it, because I have great genes.’ ”
When Kim McMorries, MD, of Texas, was confronted with the fact that he had donated sperm 33 years before, he insisted that it was ethical at the time. “When this occurred, it was not considered wrong,” he wrote in an email to his biological daughter.
Today, doctors are bound by the doctrine of informed consent, which holds that patients should be informed about all steps taken in their care. The term was coined by a judge in 1960, and it took some time for some in the medical world to fully accept informed consent. Still, Dr. Madeira asserts it was always unethical to secretly fertilize patients.
“Even in the more paternalistic era of the 1970s and 1980s, it was not right to lie to your patients about such an important part of their lives,” she said.
Some sperm doctors insisted that they had received informed consent when the patient agreed to use an anonymous donor. “Dr. Kiken did that which he was asked to do,” wrote the attorneys for Michael S. Kiken, MD, of Virginia. “Anonymous donor meant that the patient would not know the donor’s identity, he would be anonymous to her.”
Dr. Madeira does not accept this argument either. “The doctor may have thought it was understood that he could be the anonymous person, but the patients did not see it that way,” she said. “They were not expecting the anonymous donor would be their own doctor.”
“I think what happened is a crime,” said Dr. Klipstein. “It’s an ethical violation, a fracture in the trust between doctor and patient.”
Existing laws, however, don’t make it easy to prosecute the doctors. When lawsuits are filed against these doctors, “you have to shoehorn existing statutes to fit the facts, and that may not be a terrific fit,” Dr. Cantor said.
The doctors have been charged with battery, fraud, negligence, breach of duty, unjust enrichment, and rape. But none of them have been found guilty specifically of secretly using their own sperm. Two of the doctors were convicted, but for other offenses, such as perjury for denying their involvement.
Since 2019, five states – Arizona, Colorado, Florida, Indiana, and Texas – have passed statutes specifically outlawing infertility fraud. In addition, a 1995 California law requires identifying the sperm donor.
It may be difficult, however, to apply these new laws to offenses by aging sperm doctors that happened decades ago. “Some states have inflexible limits on the amount of time in which you can sue, even if you didn’t know about the problem until recently,” Dr. Madeira said. “Texas, for example, allows civil lawsuits only up to 10 years after commission.”
Before the fertility fraud physicians can be brought to justice, many of them might just fade away.
A version of this article first appeared on Medscape.com.
The ADA and hearing-impaired patients
A recent claim against a New Jersey physician attracted considerable attention in both the medical and legal communities, not only because it resulted in a substantial jury award, but because that award was not covered by malpractice insurance.
It is a good reminder for the rest of us: This case involved a charge of discrimination against a hearing-impaired patient – which meant the physician not only had to fund his own defense, but was personally responsible for the $400,000 award against him.
The Americans with Disabilities Act (ADA) was designed to protect individuals with various disabilities against discrimination in various public situations – including, specifically, “the professional office of a health care professional.”
When the disability is impaired hearing, the law requires physicians to provide any “auxiliary aids and services” that might be necessary to insure clear communication between doctor and patient. In the vast majority of such situations, a pad and pencil will satisfy that requirement. But occasionally it does not, particularly when complex medical concepts are involved; and in such cases, as the New Jersey trial demonstrated, failure to make the necessary extra effort can be very expensive.
The claim involved a hearing-impaired patient with lupus erythematosus under treatment by a rheumatologist. For almost 2 years the patient’s partner and her daughter provided translation; but that arrangement was inadequate, she testified, because her partner and daughter were unfamiliar with medical terminology and she was “unable to understand and participate in her care,” which left her “unaware of risks and available alternatives.”
She repeatedly requested that the rheumatologist provide an American Sign Language interpreter for her office visits. He refused on grounds that the cost of an interpreter would exceed the payment he would receive for the visits, which made it an “undue financial burden,” and therefore exempt from ADA requirements.
But the undue-burden exemption is not automatic; it must be demonstrated in court. And the jury decided the rheumatologist’s annual income of $425,000 rendered the cost of an interpreter quite affordable.
The lessons are clear: Physicians must take antidiscrimination laws seriously, particularly when uninsurable issues are involved; and we must be constantly aware of the needs of disabled patients, to be sure their care is not substantially different from that of any other patient.
In the case of hearing-impaired or deaf patients, it is important to remember that forms of communication that are quite adequate for most are not appropriate for some. Lip reading, written notes, and the use of family members as interpreters may be perfectly acceptable to one patient and unsuitable for another.
If the patient agrees to written notes and lip reading, as most do, you need to remember to speak slowly, and to write down critical information to avoid any miscommunications. And as always, it is crucial to document all communication, as well as the methods used for that communication – specifically including the fact that the patient agreed to those forms of communication. Documentation, as I’ve often said, is like garlic: There is no such thing as too much of it.
Should a patient not agree that written notes are sufficient, other alternatives can be offered: computer transcription, assistive listening devices, videotext displays (often available in hospitals), and telecommunication devices such as TTY and TDD. But if the patient rejects all of those options and continues to insist on a professional interpreter, the precedent set by the New Jersey case suggests that you need to acquiesce, even if the interpreter’s fee exceeds the visit reimbursement – and the ADA prohibits you from passing your cost along to the patient. But any such cost will be far less than a noninsured judgment against you.
If you must go that route, make sure the interpreter you hire is familiar with medical terminology, and is not acquainted or related to the patient (for HIPAA reasons). Your state may have an online registry of available interpreters, or your hospital may have a sign language interpreter on its staff that they might allow you to “borrow.”
The good news is several states have responded to this issue by introducing legislation that would require health insurance carriers to pay for the cost of interpreters, although none, as of this writing, have yet become law.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
A recent claim against a New Jersey physician attracted considerable attention in both the medical and legal communities, not only because it resulted in a substantial jury award, but because that award was not covered by malpractice insurance.
It is a good reminder for the rest of us: This case involved a charge of discrimination against a hearing-impaired patient – which meant the physician not only had to fund his own defense, but was personally responsible for the $400,000 award against him.
The Americans with Disabilities Act (ADA) was designed to protect individuals with various disabilities against discrimination in various public situations – including, specifically, “the professional office of a health care professional.”
When the disability is impaired hearing, the law requires physicians to provide any “auxiliary aids and services” that might be necessary to insure clear communication between doctor and patient. In the vast majority of such situations, a pad and pencil will satisfy that requirement. But occasionally it does not, particularly when complex medical concepts are involved; and in such cases, as the New Jersey trial demonstrated, failure to make the necessary extra effort can be very expensive.
The claim involved a hearing-impaired patient with lupus erythematosus under treatment by a rheumatologist. For almost 2 years the patient’s partner and her daughter provided translation; but that arrangement was inadequate, she testified, because her partner and daughter were unfamiliar with medical terminology and she was “unable to understand and participate in her care,” which left her “unaware of risks and available alternatives.”
She repeatedly requested that the rheumatologist provide an American Sign Language interpreter for her office visits. He refused on grounds that the cost of an interpreter would exceed the payment he would receive for the visits, which made it an “undue financial burden,” and therefore exempt from ADA requirements.
But the undue-burden exemption is not automatic; it must be demonstrated in court. And the jury decided the rheumatologist’s annual income of $425,000 rendered the cost of an interpreter quite affordable.
The lessons are clear: Physicians must take antidiscrimination laws seriously, particularly when uninsurable issues are involved; and we must be constantly aware of the needs of disabled patients, to be sure their care is not substantially different from that of any other patient.
In the case of hearing-impaired or deaf patients, it is important to remember that forms of communication that are quite adequate for most are not appropriate for some. Lip reading, written notes, and the use of family members as interpreters may be perfectly acceptable to one patient and unsuitable for another.
If the patient agrees to written notes and lip reading, as most do, you need to remember to speak slowly, and to write down critical information to avoid any miscommunications. And as always, it is crucial to document all communication, as well as the methods used for that communication – specifically including the fact that the patient agreed to those forms of communication. Documentation, as I’ve often said, is like garlic: There is no such thing as too much of it.
Should a patient not agree that written notes are sufficient, other alternatives can be offered: computer transcription, assistive listening devices, videotext displays (often available in hospitals), and telecommunication devices such as TTY and TDD. But if the patient rejects all of those options and continues to insist on a professional interpreter, the precedent set by the New Jersey case suggests that you need to acquiesce, even if the interpreter’s fee exceeds the visit reimbursement – and the ADA prohibits you from passing your cost along to the patient. But any such cost will be far less than a noninsured judgment against you.
If you must go that route, make sure the interpreter you hire is familiar with medical terminology, and is not acquainted or related to the patient (for HIPAA reasons). Your state may have an online registry of available interpreters, or your hospital may have a sign language interpreter on its staff that they might allow you to “borrow.”
The good news is several states have responded to this issue by introducing legislation that would require health insurance carriers to pay for the cost of interpreters, although none, as of this writing, have yet become law.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
A recent claim against a New Jersey physician attracted considerable attention in both the medical and legal communities, not only because it resulted in a substantial jury award, but because that award was not covered by malpractice insurance.
It is a good reminder for the rest of us: This case involved a charge of discrimination against a hearing-impaired patient – which meant the physician not only had to fund his own defense, but was personally responsible for the $400,000 award against him.
The Americans with Disabilities Act (ADA) was designed to protect individuals with various disabilities against discrimination in various public situations – including, specifically, “the professional office of a health care professional.”
When the disability is impaired hearing, the law requires physicians to provide any “auxiliary aids and services” that might be necessary to insure clear communication between doctor and patient. In the vast majority of such situations, a pad and pencil will satisfy that requirement. But occasionally it does not, particularly when complex medical concepts are involved; and in such cases, as the New Jersey trial demonstrated, failure to make the necessary extra effort can be very expensive.
The claim involved a hearing-impaired patient with lupus erythematosus under treatment by a rheumatologist. For almost 2 years the patient’s partner and her daughter provided translation; but that arrangement was inadequate, she testified, because her partner and daughter were unfamiliar with medical terminology and she was “unable to understand and participate in her care,” which left her “unaware of risks and available alternatives.”
She repeatedly requested that the rheumatologist provide an American Sign Language interpreter for her office visits. He refused on grounds that the cost of an interpreter would exceed the payment he would receive for the visits, which made it an “undue financial burden,” and therefore exempt from ADA requirements.
But the undue-burden exemption is not automatic; it must be demonstrated in court. And the jury decided the rheumatologist’s annual income of $425,000 rendered the cost of an interpreter quite affordable.
The lessons are clear: Physicians must take antidiscrimination laws seriously, particularly when uninsurable issues are involved; and we must be constantly aware of the needs of disabled patients, to be sure their care is not substantially different from that of any other patient.
In the case of hearing-impaired or deaf patients, it is important to remember that forms of communication that are quite adequate for most are not appropriate for some. Lip reading, written notes, and the use of family members as interpreters may be perfectly acceptable to one patient and unsuitable for another.
If the patient agrees to written notes and lip reading, as most do, you need to remember to speak slowly, and to write down critical information to avoid any miscommunications. And as always, it is crucial to document all communication, as well as the methods used for that communication – specifically including the fact that the patient agreed to those forms of communication. Documentation, as I’ve often said, is like garlic: There is no such thing as too much of it.
Should a patient not agree that written notes are sufficient, other alternatives can be offered: computer transcription, assistive listening devices, videotext displays (often available in hospitals), and telecommunication devices such as TTY and TDD. But if the patient rejects all of those options and continues to insist on a professional interpreter, the precedent set by the New Jersey case suggests that you need to acquiesce, even if the interpreter’s fee exceeds the visit reimbursement – and the ADA prohibits you from passing your cost along to the patient. But any such cost will be far less than a noninsured judgment against you.
If you must go that route, make sure the interpreter you hire is familiar with medical terminology, and is not acquainted or related to the patient (for HIPAA reasons). Your state may have an online registry of available interpreters, or your hospital may have a sign language interpreter on its staff that they might allow you to “borrow.”
The good news is several states have responded to this issue by introducing legislation that would require health insurance carriers to pay for the cost of interpreters, although none, as of this writing, have yet become law.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Widely prescribed meds ineffective for low back pain?
Results of a large systematic review and meta-analysis of randomized controlled trials show very “low certainty evidence” that non-benzodiazepine antispasmodics provide meaningful improvement in pain intensity in patients with low back pain – and may actually increase adverse event risk.
“We found that muscle relaxants might reduce pain in the short term, but on average, the effect is probably too small to be important, and most patients wouldn’t be able to feel any difference in their pain compared to taking a placebo,” study investigator Aidan Cashin, PhD, with the Center for Pain IMPACT, Neuroscience Research Australia, and University of New South Wales, Sydney, told this news organization. “There is also an increased risk of side effects,” he added.
The study was published online July 7 in The BMJ.
Global problem
Low back pain is a major global public health problem that burdens individuals, health care systems, and societies.
“Most people, around 80%, will have at least one episode of low back pain during their life,” Dr. Cashin noted.
Muscle relaxants, a broad class of drugs that include non-benzodiazepine antispasmodics and antispastics, are often prescribed for low back pain. In 2020 alone, prescriptions exceeded 1.3 million in England and topped 30 million in the United States.
“However, clinical practice guidelines have provided conflicting recommendations for the use of muscle relaxants to treat low back pain,” Dr. Cashin said.
To assess the efficacy and safety of muscle relaxants, the researchers conducted a detailed analysis of 31 randomized controlled trials that compared muscle relaxants with placebo, usual care, or no treatment in a total of 6,505 adults with nonspecific low back pain.
For acute low back pain, they found “very low certainty evidence” that non-benzodiazepine antispasmodics might reduce pain intensity at 2 weeks or less, but the effect is small – less than 8 points on a 0 to 100 point scale – and not clinically meaningful.
They found little to no effect of non-benzodiazepine antispasmodics on pain intensity at 3 to 13 weeks or on disability at any follow-up time points. None of the trials assessed the effect of muscle relaxants on long-term outcomes.
There was also low-certainty and very-low-certainty evidence that non-benzodiazepine antispasmodics might increase the risk of an adverse event, commonly dizziness, drowsiness, headache, and nausea (relative risk 1.6; 95% confidence interval, 1.2-2.0).
Better research needed
“We were surprised by the findings, as earlier research suggested that muscle relaxants did reduce pain intensity. But when we included all of the most up-to-date research, the results became much less certain,” said Dr. Cashin.
“We were also surprised to see that so much of the research wasn’t done very well, which means that we can’t be very certain in the results. There is a clear need to improve how research is done for low back pain so that we better understand whether medicines can help people or not,” Dr. Cashin said.
“We would encourage clinicians to discuss this uncertainty in the efficacy and safety of muscle relaxants with patients, sharing information about the possibility for a worthwhile benefit in pain reduction but increased risk of experiencing a nonserious adverse event, to allow them to make informed treatment decisions,” corresponding author James McAuley, PhD, University of New South Wales, said in an interview.
“We know that no matter what medicines people with low back pain are taking, they should avoid staying in bed, and they should try to be active and continue with their usual activities, including work, as much as they can. High-quality research shows that people who do this are more likely to recover faster and more completely,” said Dr. McAuley.
A symptom, not a diagnosis
Reached for comment, Andrew Hecht, MD, chief of spine surgery at Mount Sinai Health System, New York, noted that acute low back pain is “a symptom, not a diagnosis, and most episodes of acute low back pain without leg pain will resolve within a few weeks no matter what you do.”
“For people who have an episode of acute low back pain, we typically use anti-inflammatory medications, combined with a short, low dose course of a muscle relaxant if necessary, depending on the severity of symptoms, to help get you over the worst part of it,” Dr. Hecht said.
“We are trying to help the patient feel better in the short term and get more physically strong with therapy to try to reduce the frequency of these attacks in the future,” he added.
“But each patient is different. It’s not one-size-fits-all, and we don’t give prolonged courses of muscle relaxants because they have some side effects, like sedation,” Dr. Hecht cautioned.
The study had no specific funding. Dr. Cashin, Dr. McAuley, and Dr. Hecht have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large systematic review and meta-analysis of randomized controlled trials show very “low certainty evidence” that non-benzodiazepine antispasmodics provide meaningful improvement in pain intensity in patients with low back pain – and may actually increase adverse event risk.
“We found that muscle relaxants might reduce pain in the short term, but on average, the effect is probably too small to be important, and most patients wouldn’t be able to feel any difference in their pain compared to taking a placebo,” study investigator Aidan Cashin, PhD, with the Center for Pain IMPACT, Neuroscience Research Australia, and University of New South Wales, Sydney, told this news organization. “There is also an increased risk of side effects,” he added.
The study was published online July 7 in The BMJ.
Global problem
Low back pain is a major global public health problem that burdens individuals, health care systems, and societies.
“Most people, around 80%, will have at least one episode of low back pain during their life,” Dr. Cashin noted.
Muscle relaxants, a broad class of drugs that include non-benzodiazepine antispasmodics and antispastics, are often prescribed for low back pain. In 2020 alone, prescriptions exceeded 1.3 million in England and topped 30 million in the United States.
“However, clinical practice guidelines have provided conflicting recommendations for the use of muscle relaxants to treat low back pain,” Dr. Cashin said.
To assess the efficacy and safety of muscle relaxants, the researchers conducted a detailed analysis of 31 randomized controlled trials that compared muscle relaxants with placebo, usual care, or no treatment in a total of 6,505 adults with nonspecific low back pain.
For acute low back pain, they found “very low certainty evidence” that non-benzodiazepine antispasmodics might reduce pain intensity at 2 weeks or less, but the effect is small – less than 8 points on a 0 to 100 point scale – and not clinically meaningful.
They found little to no effect of non-benzodiazepine antispasmodics on pain intensity at 3 to 13 weeks or on disability at any follow-up time points. None of the trials assessed the effect of muscle relaxants on long-term outcomes.
There was also low-certainty and very-low-certainty evidence that non-benzodiazepine antispasmodics might increase the risk of an adverse event, commonly dizziness, drowsiness, headache, and nausea (relative risk 1.6; 95% confidence interval, 1.2-2.0).
Better research needed
“We were surprised by the findings, as earlier research suggested that muscle relaxants did reduce pain intensity. But when we included all of the most up-to-date research, the results became much less certain,” said Dr. Cashin.
“We were also surprised to see that so much of the research wasn’t done very well, which means that we can’t be very certain in the results. There is a clear need to improve how research is done for low back pain so that we better understand whether medicines can help people or not,” Dr. Cashin said.
“We would encourage clinicians to discuss this uncertainty in the efficacy and safety of muscle relaxants with patients, sharing information about the possibility for a worthwhile benefit in pain reduction but increased risk of experiencing a nonserious adverse event, to allow them to make informed treatment decisions,” corresponding author James McAuley, PhD, University of New South Wales, said in an interview.
“We know that no matter what medicines people with low back pain are taking, they should avoid staying in bed, and they should try to be active and continue with their usual activities, including work, as much as they can. High-quality research shows that people who do this are more likely to recover faster and more completely,” said Dr. McAuley.
A symptom, not a diagnosis
Reached for comment, Andrew Hecht, MD, chief of spine surgery at Mount Sinai Health System, New York, noted that acute low back pain is “a symptom, not a diagnosis, and most episodes of acute low back pain without leg pain will resolve within a few weeks no matter what you do.”
“For people who have an episode of acute low back pain, we typically use anti-inflammatory medications, combined with a short, low dose course of a muscle relaxant if necessary, depending on the severity of symptoms, to help get you over the worst part of it,” Dr. Hecht said.
“We are trying to help the patient feel better in the short term and get more physically strong with therapy to try to reduce the frequency of these attacks in the future,” he added.
“But each patient is different. It’s not one-size-fits-all, and we don’t give prolonged courses of muscle relaxants because they have some side effects, like sedation,” Dr. Hecht cautioned.
The study had no specific funding. Dr. Cashin, Dr. McAuley, and Dr. Hecht have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large systematic review and meta-analysis of randomized controlled trials show very “low certainty evidence” that non-benzodiazepine antispasmodics provide meaningful improvement in pain intensity in patients with low back pain – and may actually increase adverse event risk.
“We found that muscle relaxants might reduce pain in the short term, but on average, the effect is probably too small to be important, and most patients wouldn’t be able to feel any difference in their pain compared to taking a placebo,” study investigator Aidan Cashin, PhD, with the Center for Pain IMPACT, Neuroscience Research Australia, and University of New South Wales, Sydney, told this news organization. “There is also an increased risk of side effects,” he added.
The study was published online July 7 in The BMJ.
Global problem
Low back pain is a major global public health problem that burdens individuals, health care systems, and societies.
“Most people, around 80%, will have at least one episode of low back pain during their life,” Dr. Cashin noted.
Muscle relaxants, a broad class of drugs that include non-benzodiazepine antispasmodics and antispastics, are often prescribed for low back pain. In 2020 alone, prescriptions exceeded 1.3 million in England and topped 30 million in the United States.
“However, clinical practice guidelines have provided conflicting recommendations for the use of muscle relaxants to treat low back pain,” Dr. Cashin said.
To assess the efficacy and safety of muscle relaxants, the researchers conducted a detailed analysis of 31 randomized controlled trials that compared muscle relaxants with placebo, usual care, or no treatment in a total of 6,505 adults with nonspecific low back pain.
For acute low back pain, they found “very low certainty evidence” that non-benzodiazepine antispasmodics might reduce pain intensity at 2 weeks or less, but the effect is small – less than 8 points on a 0 to 100 point scale – and not clinically meaningful.
They found little to no effect of non-benzodiazepine antispasmodics on pain intensity at 3 to 13 weeks or on disability at any follow-up time points. None of the trials assessed the effect of muscle relaxants on long-term outcomes.
There was also low-certainty and very-low-certainty evidence that non-benzodiazepine antispasmodics might increase the risk of an adverse event, commonly dizziness, drowsiness, headache, and nausea (relative risk 1.6; 95% confidence interval, 1.2-2.0).
Better research needed
“We were surprised by the findings, as earlier research suggested that muscle relaxants did reduce pain intensity. But when we included all of the most up-to-date research, the results became much less certain,” said Dr. Cashin.
“We were also surprised to see that so much of the research wasn’t done very well, which means that we can’t be very certain in the results. There is a clear need to improve how research is done for low back pain so that we better understand whether medicines can help people or not,” Dr. Cashin said.
“We would encourage clinicians to discuss this uncertainty in the efficacy and safety of muscle relaxants with patients, sharing information about the possibility for a worthwhile benefit in pain reduction but increased risk of experiencing a nonserious adverse event, to allow them to make informed treatment decisions,” corresponding author James McAuley, PhD, University of New South Wales, said in an interview.
“We know that no matter what medicines people with low back pain are taking, they should avoid staying in bed, and they should try to be active and continue with their usual activities, including work, as much as they can. High-quality research shows that people who do this are more likely to recover faster and more completely,” said Dr. McAuley.
A symptom, not a diagnosis
Reached for comment, Andrew Hecht, MD, chief of spine surgery at Mount Sinai Health System, New York, noted that acute low back pain is “a symptom, not a diagnosis, and most episodes of acute low back pain without leg pain will resolve within a few weeks no matter what you do.”
“For people who have an episode of acute low back pain, we typically use anti-inflammatory medications, combined with a short, low dose course of a muscle relaxant if necessary, depending on the severity of symptoms, to help get you over the worst part of it,” Dr. Hecht said.
“We are trying to help the patient feel better in the short term and get more physically strong with therapy to try to reduce the frequency of these attacks in the future,” he added.
“But each patient is different. It’s not one-size-fits-all, and we don’t give prolonged courses of muscle relaxants because they have some side effects, like sedation,” Dr. Hecht cautioned.
The study had no specific funding. Dr. Cashin, Dr. McAuley, and Dr. Hecht have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Drinking coffee not linked to increased arrhythmia risk in new study
In fact, an adjusted analysis found that “each additional cup of coffee intake was associated with a 3% lower risk of incident arrhythmia,” Eun-jeong Kim, MD, of the division of cardiology at the University of California, San Francisco, and colleagues reported in JAMA Internal Medicine.
In addition, genetic differences that affect caffeine metabolism did not significantly influence the odds of arrhythmias, the researchers found.
Still, these findings should not necessarily encourage people to start drinking coffee if they don’t already, or to guzzle additional cups with abandon, they said.
“We certainly don’t want to say drink coffee and it will reduce your risk of arrhythmias,” study author Gregory M. Marcus, MD, MAS, associate chief of cardiology for research at UCSF Health, said in an interview. “But rather, we think the main point is that a blanket prohibition against coffee or caffeine to reduce the risk of arrhythmias among patients who have a diagnosis of arrhythmias is likely unwarranted. And given some evidence that coffee consumption may actually have other benefits regarding diabetes, mood, and perhaps overall mortality, it may be problematic to admonish patients to avoid coffee or caffeine when it is not really warranted.”
Methods and results
The conventional wisdom that caffeine increases arrhythmic risk has not been well substantiated. To further examine whether moderate, habitual coffee drinking relates to arrhythmia risk, and whether certain genetic variants influence the association, Dr. Kim and colleagues analyzed data from the UK Biobank. They focused on longitudinal data collected between 2006 and 2018 from 386,258 people who did not have a prior diagnosis of arrhythmia.
Participants had an average age of 56 years, and about 52% were female. They provided information about their coffee consumption, and the researchers grouped the participants into eight categories based on their daily coffee intake: 0, less than 1, 1, 2, 3, 4, 5, and 6 or more cups per day.
Over an average follow-up of 4.5 years, 16,979 participants developed an incident arrhythmia. After adjusting for demographic characteristics, comorbid conditions, and lifestyle habits, the decreased risk with each cup of coffee was similar for atrial fibrillation or flutter (hazard ratio, 0.97) and supraventricular tachycardia (HR, 0.96).
Taking into account genetic variations that relate to caffeine metabolism did not modify the findings. Mendelian randomization analyses that used a polygenic score of inherited caffeine metabolism patterns “failed to provide evidence that caffeine consumption leads to a greater risk of arrhythmias,” the researchers said.
Professional society guidelines have suggested staying away from caffeinated products to reduce the risk of arrhythmia, but this guidance has “relied on assumed mechanisms and a small observational study from 1980,” the authors wrote. Subsequent research has indicated that coffee’s reputation of increasing the risk of arrhythmia may be undeserved.
“The investigators should be commended on performing a high-quality observational study to try to further understand the association between coffee consumption and arrhythmias, or the lack of one,” commented Zachary D. Goldberger, MD, MS, with the division of cardiovascular medicine at the University of Wisconsin–Madison, who was not involved in the study. “This is not a randomized, controlled trial, and coffee consumption was self-reported, but the methods employed are rigorous, despite these and other important limitations. However, we need to be extremely cautious in how we interpret these findings, and not use these data as a prescription for more coffee. It’s important to recognize that this study is not telling us to drink more coffee, or start drinking coffee, to protect against developing arrhythmias. However, it should offer more reassurance that moderate coffee consumption is not necessarily harmful, and will not always lead to arrhythmias. This is important, given the widespread notion that coffee is universally proarrhythmic.”
A call for personalized guidance
“As the investigators note, there are definitely biologically plausible reasons how coffee and caffeine may not cause arrhythmias, and may be possibly protective in some, despite being a stimulant,” Dr. Goldberger said. “However, if your patient is reporting palpitations or symptoms of an arrhythmia, and feels they be related to coffee or caffeine, we should not use this study to tell them that coffee may not be the culprit. We need to listen to our patients, and the decision to reduce coffee consumption to reduce these symptoms needs to be personalized.”
The effect size was small, and only about 4% of the participants developed an arrhythmia, Dr. Goldberger and Rodney A. Hayward, MD, wrote in an invited commentary on the study in JAMA Internal Medicine. Dr. Hayward is a professor of public health and internal medicine at the University of Michigan, Ann Arbor, and a senior investigator at the Ann Arbor Veterans Affairs Center for Clinical Management Research.
“Unfortunately, coffee consumption was self-reported at a single time point. Not only can this lead to recall bias, but subsequent and substantial changes in coffee consumption are also possible, including reductions due to new signs or symptoms,” they said.
No evidence that coffee ups risk for developing arrhythmias
Another recent study suggests that people may alter their coffee consumption depending on their baseline cardiovascular health, according to the commentary.
Overall, the results “strengthen the evidence that caffeine is not proarrhythmic, but they should not be taken as proving that coffee is an antiarrhythmic—this distinction is of paramount importance,” Dr. Goldberger and Dr. Hayward wrote. “Health care professionals can reassure patients that there is no evidence that drinking coffee increases the risk for developing arrhythmias. This is particularly important for the many patients with benign palpitations who are devastated when they think, or are told, that they have to stop drinking coffee. Given current evidence, this is entirely a patient-preference decision, not a medical one.”
Dr. Marcus, a cardiac electrophysiologist, sees patients with arrhythmias all the time. They tend to “come in fairly convinced that caffeine is to be avoided when they have arrhythmias,” he said. “Often, they been told by their primary care physician or their general cardiologist to avoid caffeine because they have an arrhythmia.
“What I suggest to my patients is that they feel free to go ahead and experiment and try coffee,” Dr. Marcus said.
Still, Dr. Marcus suspects that there are some individuals in whom caffeine is a trigger for the arrhythmia. But evidence indicates these cases likely are rare, and avoiding caffeine need not apply to the general population, particularly “given the potential health benefits of benefits of coffee and also, frankly, just the enhanced quality of life that people can enjoy drinking a good cup of coffee.”
The research was conducted using the UK Biobank resource, which was established by the Wellcome Trust, the Medical Research Council, the U.K. Department of Health, and the Scottish government. The UK Biobank has received funding from other agencies and foundations as well. Dr. Marcus disclosed grants from Baylis, Medtronic, and Eight Sleep outside the submitted work. In addition, he reported consulting for Johnson & Johnson and InCarda, and holding equity in InCarda. A coauthor received salary support from the National Institutes of Health during the study. Dr. Goldberger and Dr. Hayward disclosed no conflicts of interest.
In fact, an adjusted analysis found that “each additional cup of coffee intake was associated with a 3% lower risk of incident arrhythmia,” Eun-jeong Kim, MD, of the division of cardiology at the University of California, San Francisco, and colleagues reported in JAMA Internal Medicine.
In addition, genetic differences that affect caffeine metabolism did not significantly influence the odds of arrhythmias, the researchers found.
Still, these findings should not necessarily encourage people to start drinking coffee if they don’t already, or to guzzle additional cups with abandon, they said.
“We certainly don’t want to say drink coffee and it will reduce your risk of arrhythmias,” study author Gregory M. Marcus, MD, MAS, associate chief of cardiology for research at UCSF Health, said in an interview. “But rather, we think the main point is that a blanket prohibition against coffee or caffeine to reduce the risk of arrhythmias among patients who have a diagnosis of arrhythmias is likely unwarranted. And given some evidence that coffee consumption may actually have other benefits regarding diabetes, mood, and perhaps overall mortality, it may be problematic to admonish patients to avoid coffee or caffeine when it is not really warranted.”
Methods and results
The conventional wisdom that caffeine increases arrhythmic risk has not been well substantiated. To further examine whether moderate, habitual coffee drinking relates to arrhythmia risk, and whether certain genetic variants influence the association, Dr. Kim and colleagues analyzed data from the UK Biobank. They focused on longitudinal data collected between 2006 and 2018 from 386,258 people who did not have a prior diagnosis of arrhythmia.
Participants had an average age of 56 years, and about 52% were female. They provided information about their coffee consumption, and the researchers grouped the participants into eight categories based on their daily coffee intake: 0, less than 1, 1, 2, 3, 4, 5, and 6 or more cups per day.
Over an average follow-up of 4.5 years, 16,979 participants developed an incident arrhythmia. After adjusting for demographic characteristics, comorbid conditions, and lifestyle habits, the decreased risk with each cup of coffee was similar for atrial fibrillation or flutter (hazard ratio, 0.97) and supraventricular tachycardia (HR, 0.96).
Taking into account genetic variations that relate to caffeine metabolism did not modify the findings. Mendelian randomization analyses that used a polygenic score of inherited caffeine metabolism patterns “failed to provide evidence that caffeine consumption leads to a greater risk of arrhythmias,” the researchers said.
Professional society guidelines have suggested staying away from caffeinated products to reduce the risk of arrhythmia, but this guidance has “relied on assumed mechanisms and a small observational study from 1980,” the authors wrote. Subsequent research has indicated that coffee’s reputation of increasing the risk of arrhythmia may be undeserved.
“The investigators should be commended on performing a high-quality observational study to try to further understand the association between coffee consumption and arrhythmias, or the lack of one,” commented Zachary D. Goldberger, MD, MS, with the division of cardiovascular medicine at the University of Wisconsin–Madison, who was not involved in the study. “This is not a randomized, controlled trial, and coffee consumption was self-reported, but the methods employed are rigorous, despite these and other important limitations. However, we need to be extremely cautious in how we interpret these findings, and not use these data as a prescription for more coffee. It’s important to recognize that this study is not telling us to drink more coffee, or start drinking coffee, to protect against developing arrhythmias. However, it should offer more reassurance that moderate coffee consumption is not necessarily harmful, and will not always lead to arrhythmias. This is important, given the widespread notion that coffee is universally proarrhythmic.”
A call for personalized guidance
“As the investigators note, there are definitely biologically plausible reasons how coffee and caffeine may not cause arrhythmias, and may be possibly protective in some, despite being a stimulant,” Dr. Goldberger said. “However, if your patient is reporting palpitations or symptoms of an arrhythmia, and feels they be related to coffee or caffeine, we should not use this study to tell them that coffee may not be the culprit. We need to listen to our patients, and the decision to reduce coffee consumption to reduce these symptoms needs to be personalized.”
The effect size was small, and only about 4% of the participants developed an arrhythmia, Dr. Goldberger and Rodney A. Hayward, MD, wrote in an invited commentary on the study in JAMA Internal Medicine. Dr. Hayward is a professor of public health and internal medicine at the University of Michigan, Ann Arbor, and a senior investigator at the Ann Arbor Veterans Affairs Center for Clinical Management Research.
“Unfortunately, coffee consumption was self-reported at a single time point. Not only can this lead to recall bias, but subsequent and substantial changes in coffee consumption are also possible, including reductions due to new signs or symptoms,” they said.
No evidence that coffee ups risk for developing arrhythmias
Another recent study suggests that people may alter their coffee consumption depending on their baseline cardiovascular health, according to the commentary.
Overall, the results “strengthen the evidence that caffeine is not proarrhythmic, but they should not be taken as proving that coffee is an antiarrhythmic—this distinction is of paramount importance,” Dr. Goldberger and Dr. Hayward wrote. “Health care professionals can reassure patients that there is no evidence that drinking coffee increases the risk for developing arrhythmias. This is particularly important for the many patients with benign palpitations who are devastated when they think, or are told, that they have to stop drinking coffee. Given current evidence, this is entirely a patient-preference decision, not a medical one.”
Dr. Marcus, a cardiac electrophysiologist, sees patients with arrhythmias all the time. They tend to “come in fairly convinced that caffeine is to be avoided when they have arrhythmias,” he said. “Often, they been told by their primary care physician or their general cardiologist to avoid caffeine because they have an arrhythmia.
“What I suggest to my patients is that they feel free to go ahead and experiment and try coffee,” Dr. Marcus said.
Still, Dr. Marcus suspects that there are some individuals in whom caffeine is a trigger for the arrhythmia. But evidence indicates these cases likely are rare, and avoiding caffeine need not apply to the general population, particularly “given the potential health benefits of benefits of coffee and also, frankly, just the enhanced quality of life that people can enjoy drinking a good cup of coffee.”
The research was conducted using the UK Biobank resource, which was established by the Wellcome Trust, the Medical Research Council, the U.K. Department of Health, and the Scottish government. The UK Biobank has received funding from other agencies and foundations as well. Dr. Marcus disclosed grants from Baylis, Medtronic, and Eight Sleep outside the submitted work. In addition, he reported consulting for Johnson & Johnson and InCarda, and holding equity in InCarda. A coauthor received salary support from the National Institutes of Health during the study. Dr. Goldberger and Dr. Hayward disclosed no conflicts of interest.
In fact, an adjusted analysis found that “each additional cup of coffee intake was associated with a 3% lower risk of incident arrhythmia,” Eun-jeong Kim, MD, of the division of cardiology at the University of California, San Francisco, and colleagues reported in JAMA Internal Medicine.
In addition, genetic differences that affect caffeine metabolism did not significantly influence the odds of arrhythmias, the researchers found.
Still, these findings should not necessarily encourage people to start drinking coffee if they don’t already, or to guzzle additional cups with abandon, they said.
“We certainly don’t want to say drink coffee and it will reduce your risk of arrhythmias,” study author Gregory M. Marcus, MD, MAS, associate chief of cardiology for research at UCSF Health, said in an interview. “But rather, we think the main point is that a blanket prohibition against coffee or caffeine to reduce the risk of arrhythmias among patients who have a diagnosis of arrhythmias is likely unwarranted. And given some evidence that coffee consumption may actually have other benefits regarding diabetes, mood, and perhaps overall mortality, it may be problematic to admonish patients to avoid coffee or caffeine when it is not really warranted.”
Methods and results
The conventional wisdom that caffeine increases arrhythmic risk has not been well substantiated. To further examine whether moderate, habitual coffee drinking relates to arrhythmia risk, and whether certain genetic variants influence the association, Dr. Kim and colleagues analyzed data from the UK Biobank. They focused on longitudinal data collected between 2006 and 2018 from 386,258 people who did not have a prior diagnosis of arrhythmia.
Participants had an average age of 56 years, and about 52% were female. They provided information about their coffee consumption, and the researchers grouped the participants into eight categories based on their daily coffee intake: 0, less than 1, 1, 2, 3, 4, 5, and 6 or more cups per day.
Over an average follow-up of 4.5 years, 16,979 participants developed an incident arrhythmia. After adjusting for demographic characteristics, comorbid conditions, and lifestyle habits, the decreased risk with each cup of coffee was similar for atrial fibrillation or flutter (hazard ratio, 0.97) and supraventricular tachycardia (HR, 0.96).
Taking into account genetic variations that relate to caffeine metabolism did not modify the findings. Mendelian randomization analyses that used a polygenic score of inherited caffeine metabolism patterns “failed to provide evidence that caffeine consumption leads to a greater risk of arrhythmias,” the researchers said.
Professional society guidelines have suggested staying away from caffeinated products to reduce the risk of arrhythmia, but this guidance has “relied on assumed mechanisms and a small observational study from 1980,” the authors wrote. Subsequent research has indicated that coffee’s reputation of increasing the risk of arrhythmia may be undeserved.
“The investigators should be commended on performing a high-quality observational study to try to further understand the association between coffee consumption and arrhythmias, or the lack of one,” commented Zachary D. Goldberger, MD, MS, with the division of cardiovascular medicine at the University of Wisconsin–Madison, who was not involved in the study. “This is not a randomized, controlled trial, and coffee consumption was self-reported, but the methods employed are rigorous, despite these and other important limitations. However, we need to be extremely cautious in how we interpret these findings, and not use these data as a prescription for more coffee. It’s important to recognize that this study is not telling us to drink more coffee, or start drinking coffee, to protect against developing arrhythmias. However, it should offer more reassurance that moderate coffee consumption is not necessarily harmful, and will not always lead to arrhythmias. This is important, given the widespread notion that coffee is universally proarrhythmic.”
A call for personalized guidance
“As the investigators note, there are definitely biologically plausible reasons how coffee and caffeine may not cause arrhythmias, and may be possibly protective in some, despite being a stimulant,” Dr. Goldberger said. “However, if your patient is reporting palpitations or symptoms of an arrhythmia, and feels they be related to coffee or caffeine, we should not use this study to tell them that coffee may not be the culprit. We need to listen to our patients, and the decision to reduce coffee consumption to reduce these symptoms needs to be personalized.”
The effect size was small, and only about 4% of the participants developed an arrhythmia, Dr. Goldberger and Rodney A. Hayward, MD, wrote in an invited commentary on the study in JAMA Internal Medicine. Dr. Hayward is a professor of public health and internal medicine at the University of Michigan, Ann Arbor, and a senior investigator at the Ann Arbor Veterans Affairs Center for Clinical Management Research.
“Unfortunately, coffee consumption was self-reported at a single time point. Not only can this lead to recall bias, but subsequent and substantial changes in coffee consumption are also possible, including reductions due to new signs or symptoms,” they said.
No evidence that coffee ups risk for developing arrhythmias
Another recent study suggests that people may alter their coffee consumption depending on their baseline cardiovascular health, according to the commentary.
Overall, the results “strengthen the evidence that caffeine is not proarrhythmic, but they should not be taken as proving that coffee is an antiarrhythmic—this distinction is of paramount importance,” Dr. Goldberger and Dr. Hayward wrote. “Health care professionals can reassure patients that there is no evidence that drinking coffee increases the risk for developing arrhythmias. This is particularly important for the many patients with benign palpitations who are devastated when they think, or are told, that they have to stop drinking coffee. Given current evidence, this is entirely a patient-preference decision, not a medical one.”
Dr. Marcus, a cardiac electrophysiologist, sees patients with arrhythmias all the time. They tend to “come in fairly convinced that caffeine is to be avoided when they have arrhythmias,” he said. “Often, they been told by their primary care physician or their general cardiologist to avoid caffeine because they have an arrhythmia.
“What I suggest to my patients is that they feel free to go ahead and experiment and try coffee,” Dr. Marcus said.
Still, Dr. Marcus suspects that there are some individuals in whom caffeine is a trigger for the arrhythmia. But evidence indicates these cases likely are rare, and avoiding caffeine need not apply to the general population, particularly “given the potential health benefits of benefits of coffee and also, frankly, just the enhanced quality of life that people can enjoy drinking a good cup of coffee.”
The research was conducted using the UK Biobank resource, which was established by the Wellcome Trust, the Medical Research Council, the U.K. Department of Health, and the Scottish government. The UK Biobank has received funding from other agencies and foundations as well. Dr. Marcus disclosed grants from Baylis, Medtronic, and Eight Sleep outside the submitted work. In addition, he reported consulting for Johnson & Johnson and InCarda, and holding equity in InCarda. A coauthor received salary support from the National Institutes of Health during the study. Dr. Goldberger and Dr. Hayward disclosed no conflicts of interest.
FROM JAMA INTERNAL MEDICINE
Levothyroxine overprescribing common, consistent over time
Most U.S. prescriptions for the thyroid hormone replacement drug levothyroxine are not appropriate for patients with mild subclinical hypothyroidism, a trend that has remained steady for a decade despite evidence showing no significant benefits for those patients, new research shows.
“These results suggest substantial overuse of levothyroxine during the entire duration of the study, suggesting opportunities to improve care,” wrote the authors of the study published in JAMA Internal Medicine.
“There have been previous reports of increased levothyroxine overuse in the U.S., but this is the first paper to describe the nature of the drivers of the overuse,” first author Juan P. Brito, MD, of the division of endocrinology, diabetes, metabolism and nutrition, department of internal medicine, Mayo Clinic, Rochester, Minn., said in an interview.
The findings underscore the need to improve awareness of the ongoing overuse, said the authors of an accompanying editorial.
“We hope [this study] resonates as a call to action for clinicians to stop treating patients with mild subclinical hypothyroidism,” they wrote.
Only 8% of those receiving levothyroxine had overt hypothyroidism
For the study, Dr. Brito and colleagues analyzed data of adults enrolled in Medicare Advantage who filled levothyroxine prescriptions between January 2008 and December 2018 and had thyrotropin levels measured within 3 months prior to the prescription. Patients with a history of thyroid surgery, thyroid cancer, central hypothyroidism, or who were pregnant, were excluded from the study.
In the 110,842 patients who started levothyroxine during the study period, there were no significant changes in median thyrotropin levels at the time of treatment initiation, with a median level in 2008 of 5.8 mIU/L and a level in 2018 of 5.3 mIU/L (P = .79).
In a subanalysis of 58,706 patients for whom thyrotropin as well as free thyroxine (FT4 or T4) levels were available – which allowed for the determination of the level of hypothyroidism – levothyroxine was initiated for overt hypothyroidism in only 8.4% of cases.
In as many as 61.0% of cases, patients had subclinical hypothyroidism, and in 30.5% of cases, patients had normal thyroid levels.
While the proportion of adults with overt hypothyroidism initiated on levothyroxine significantly increased over the 10 years (7.6% to 8.4%; P = .02), rates of those with subclinical hypothyroidism remained unchanged (59.3% to 65.7%; P = .36), as did the proportion with normal thyroid function (32.9% to 26.2%; P = .84).
A closer look at patients specifically with subclinical hypothyroidism showed there were also no changes in the proportion with mild subclinical hypothyroidism (thyrotropin level of 4.5 mIU/L to <10 mIU/L with normal FT4 or T4) between the beginning and end of the study period (48.2% vs. 57.9%; P = .73). Rates of moderate subclinical hypothyroidism (thyrotropin level 10-19.9 mIU/L) were also similar (8.5% to 6.4%; P = .16).
No significant benefit, but ample undesirable effects
The authors underscore that levothyroxine has been shown time and again to offer no significant benefit to patients with subclinical hypothyroidism of any type, emphasized in a 2018 meta-analysis of 21 randomized, controlled trials.
“Frequent initiation of levothyroxine in these patients is at odds with evidence demonstrating no significant association of levothyroxine replacement with measures of health-related quality of life, thyroid-related symptoms, depressive symptoms, fatigue, or cognitive function,” they explained.
In addition to showing no benefit for subclinical hypothyroidism, levothyroxine is associated with a host of unwanted side effects, noted editorialists William K. Silverstein, MD, of Sunnybrook Health Sciences Centre, department of medicine, University of Toronto, and Deborah Grady, MD, of the department of medicine, University of California, San Francisco.
Some studies have shown a link between long-term levothyroxine therapy and an increased risk of cardiovascular disease, cardiac dysrhythmias, osteoporosis, and fractures, they explained.
In addition, unnecessary treatment “increases pill burden and costs, necessitates routine physician visits and blood work, and requires modification of daily routines so that patients can take medications on an empty stomach,” the editorialists wrote.
Importantly, evidence shows that once levothyroxine treatment for subclinical hypothyroidism is started, most patients will continue the therapy for life, they added.
The fact that levothyroxine is among the most commonly prescribed drugs in the United States, with about 7% of the population estimated to have an active prescription when overt hypothyroidism affects only about 0.2%-2% of the population, underscores the extent of levothyroxine overuse, Dr. Silverstein said in an interview.
“The really notable surprise was how pervasive inappropriate use of levothyroxine was,” he said. “The fact that only 8% of patients had a biochemical indication for treatment is striking.”
Potential solutions: ‘Shift the conversation’
In terms of potential solutions to the problem, Dr. Silverstein suggested laboratories change reference ranges so that only thyrotropin values greater than 10 mIU/L are reported as abnormal.
“Studies have shown that changing the thyrotropin reference range is associated with clinicians’ prescribing patterns,” he noted.
Dr. Brito agreed, noting that “there are many guidelines with different hypothyroidism thresholds, so we need to be more consistent about the message to clinicians.
“In addition, we have to come up with different approaches to symptoms that have nothing to do with levothyroxine,” Dr. Brito said.
“I try to explain to patients that it’s very unlikely that subclinical hypothyroidism would be driving significant symptoms like fatigue, weight gain, and hair loss,” Dr. Brito said. “So, one approach is to shift the conversation from how your thyroid is causing this to ‘how are we going to treat the symptoms?’ ”
The study was supported by the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery. Dr. Silverstein has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Most U.S. prescriptions for the thyroid hormone replacement drug levothyroxine are not appropriate for patients with mild subclinical hypothyroidism, a trend that has remained steady for a decade despite evidence showing no significant benefits for those patients, new research shows.
“These results suggest substantial overuse of levothyroxine during the entire duration of the study, suggesting opportunities to improve care,” wrote the authors of the study published in JAMA Internal Medicine.
“There have been previous reports of increased levothyroxine overuse in the U.S., but this is the first paper to describe the nature of the drivers of the overuse,” first author Juan P. Brito, MD, of the division of endocrinology, diabetes, metabolism and nutrition, department of internal medicine, Mayo Clinic, Rochester, Minn., said in an interview.
The findings underscore the need to improve awareness of the ongoing overuse, said the authors of an accompanying editorial.
“We hope [this study] resonates as a call to action for clinicians to stop treating patients with mild subclinical hypothyroidism,” they wrote.
Only 8% of those receiving levothyroxine had overt hypothyroidism
For the study, Dr. Brito and colleagues analyzed data of adults enrolled in Medicare Advantage who filled levothyroxine prescriptions between January 2008 and December 2018 and had thyrotropin levels measured within 3 months prior to the prescription. Patients with a history of thyroid surgery, thyroid cancer, central hypothyroidism, or who were pregnant, were excluded from the study.
In the 110,842 patients who started levothyroxine during the study period, there were no significant changes in median thyrotropin levels at the time of treatment initiation, with a median level in 2008 of 5.8 mIU/L and a level in 2018 of 5.3 mIU/L (P = .79).
In a subanalysis of 58,706 patients for whom thyrotropin as well as free thyroxine (FT4 or T4) levels were available – which allowed for the determination of the level of hypothyroidism – levothyroxine was initiated for overt hypothyroidism in only 8.4% of cases.
In as many as 61.0% of cases, patients had subclinical hypothyroidism, and in 30.5% of cases, patients had normal thyroid levels.
While the proportion of adults with overt hypothyroidism initiated on levothyroxine significantly increased over the 10 years (7.6% to 8.4%; P = .02), rates of those with subclinical hypothyroidism remained unchanged (59.3% to 65.7%; P = .36), as did the proportion with normal thyroid function (32.9% to 26.2%; P = .84).
A closer look at patients specifically with subclinical hypothyroidism showed there were also no changes in the proportion with mild subclinical hypothyroidism (thyrotropin level of 4.5 mIU/L to <10 mIU/L with normal FT4 or T4) between the beginning and end of the study period (48.2% vs. 57.9%; P = .73). Rates of moderate subclinical hypothyroidism (thyrotropin level 10-19.9 mIU/L) were also similar (8.5% to 6.4%; P = .16).
No significant benefit, but ample undesirable effects
The authors underscore that levothyroxine has been shown time and again to offer no significant benefit to patients with subclinical hypothyroidism of any type, emphasized in a 2018 meta-analysis of 21 randomized, controlled trials.
“Frequent initiation of levothyroxine in these patients is at odds with evidence demonstrating no significant association of levothyroxine replacement with measures of health-related quality of life, thyroid-related symptoms, depressive symptoms, fatigue, or cognitive function,” they explained.
In addition to showing no benefit for subclinical hypothyroidism, levothyroxine is associated with a host of unwanted side effects, noted editorialists William K. Silverstein, MD, of Sunnybrook Health Sciences Centre, department of medicine, University of Toronto, and Deborah Grady, MD, of the department of medicine, University of California, San Francisco.
Some studies have shown a link between long-term levothyroxine therapy and an increased risk of cardiovascular disease, cardiac dysrhythmias, osteoporosis, and fractures, they explained.
In addition, unnecessary treatment “increases pill burden and costs, necessitates routine physician visits and blood work, and requires modification of daily routines so that patients can take medications on an empty stomach,” the editorialists wrote.
Importantly, evidence shows that once levothyroxine treatment for subclinical hypothyroidism is started, most patients will continue the therapy for life, they added.
The fact that levothyroxine is among the most commonly prescribed drugs in the United States, with about 7% of the population estimated to have an active prescription when overt hypothyroidism affects only about 0.2%-2% of the population, underscores the extent of levothyroxine overuse, Dr. Silverstein said in an interview.
“The really notable surprise was how pervasive inappropriate use of levothyroxine was,” he said. “The fact that only 8% of patients had a biochemical indication for treatment is striking.”
Potential solutions: ‘Shift the conversation’
In terms of potential solutions to the problem, Dr. Silverstein suggested laboratories change reference ranges so that only thyrotropin values greater than 10 mIU/L are reported as abnormal.
“Studies have shown that changing the thyrotropin reference range is associated with clinicians’ prescribing patterns,” he noted.
Dr. Brito agreed, noting that “there are many guidelines with different hypothyroidism thresholds, so we need to be more consistent about the message to clinicians.
“In addition, we have to come up with different approaches to symptoms that have nothing to do with levothyroxine,” Dr. Brito said.
“I try to explain to patients that it’s very unlikely that subclinical hypothyroidism would be driving significant symptoms like fatigue, weight gain, and hair loss,” Dr. Brito said. “So, one approach is to shift the conversation from how your thyroid is causing this to ‘how are we going to treat the symptoms?’ ”
The study was supported by the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery. Dr. Silverstein has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Most U.S. prescriptions for the thyroid hormone replacement drug levothyroxine are not appropriate for patients with mild subclinical hypothyroidism, a trend that has remained steady for a decade despite evidence showing no significant benefits for those patients, new research shows.
“These results suggest substantial overuse of levothyroxine during the entire duration of the study, suggesting opportunities to improve care,” wrote the authors of the study published in JAMA Internal Medicine.
“There have been previous reports of increased levothyroxine overuse in the U.S., but this is the first paper to describe the nature of the drivers of the overuse,” first author Juan P. Brito, MD, of the division of endocrinology, diabetes, metabolism and nutrition, department of internal medicine, Mayo Clinic, Rochester, Minn., said in an interview.
The findings underscore the need to improve awareness of the ongoing overuse, said the authors of an accompanying editorial.
“We hope [this study] resonates as a call to action for clinicians to stop treating patients with mild subclinical hypothyroidism,” they wrote.
Only 8% of those receiving levothyroxine had overt hypothyroidism
For the study, Dr. Brito and colleagues analyzed data of adults enrolled in Medicare Advantage who filled levothyroxine prescriptions between January 2008 and December 2018 and had thyrotropin levels measured within 3 months prior to the prescription. Patients with a history of thyroid surgery, thyroid cancer, central hypothyroidism, or who were pregnant, were excluded from the study.
In the 110,842 patients who started levothyroxine during the study period, there were no significant changes in median thyrotropin levels at the time of treatment initiation, with a median level in 2008 of 5.8 mIU/L and a level in 2018 of 5.3 mIU/L (P = .79).
In a subanalysis of 58,706 patients for whom thyrotropin as well as free thyroxine (FT4 or T4) levels were available – which allowed for the determination of the level of hypothyroidism – levothyroxine was initiated for overt hypothyroidism in only 8.4% of cases.
In as many as 61.0% of cases, patients had subclinical hypothyroidism, and in 30.5% of cases, patients had normal thyroid levels.
While the proportion of adults with overt hypothyroidism initiated on levothyroxine significantly increased over the 10 years (7.6% to 8.4%; P = .02), rates of those with subclinical hypothyroidism remained unchanged (59.3% to 65.7%; P = .36), as did the proportion with normal thyroid function (32.9% to 26.2%; P = .84).
A closer look at patients specifically with subclinical hypothyroidism showed there were also no changes in the proportion with mild subclinical hypothyroidism (thyrotropin level of 4.5 mIU/L to <10 mIU/L with normal FT4 or T4) between the beginning and end of the study period (48.2% vs. 57.9%; P = .73). Rates of moderate subclinical hypothyroidism (thyrotropin level 10-19.9 mIU/L) were also similar (8.5% to 6.4%; P = .16).
No significant benefit, but ample undesirable effects
The authors underscore that levothyroxine has been shown time and again to offer no significant benefit to patients with subclinical hypothyroidism of any type, emphasized in a 2018 meta-analysis of 21 randomized, controlled trials.
“Frequent initiation of levothyroxine in these patients is at odds with evidence demonstrating no significant association of levothyroxine replacement with measures of health-related quality of life, thyroid-related symptoms, depressive symptoms, fatigue, or cognitive function,” they explained.
In addition to showing no benefit for subclinical hypothyroidism, levothyroxine is associated with a host of unwanted side effects, noted editorialists William K. Silverstein, MD, of Sunnybrook Health Sciences Centre, department of medicine, University of Toronto, and Deborah Grady, MD, of the department of medicine, University of California, San Francisco.
Some studies have shown a link between long-term levothyroxine therapy and an increased risk of cardiovascular disease, cardiac dysrhythmias, osteoporosis, and fractures, they explained.
In addition, unnecessary treatment “increases pill burden and costs, necessitates routine physician visits and blood work, and requires modification of daily routines so that patients can take medications on an empty stomach,” the editorialists wrote.
Importantly, evidence shows that once levothyroxine treatment for subclinical hypothyroidism is started, most patients will continue the therapy for life, they added.
The fact that levothyroxine is among the most commonly prescribed drugs in the United States, with about 7% of the population estimated to have an active prescription when overt hypothyroidism affects only about 0.2%-2% of the population, underscores the extent of levothyroxine overuse, Dr. Silverstein said in an interview.
“The really notable surprise was how pervasive inappropriate use of levothyroxine was,” he said. “The fact that only 8% of patients had a biochemical indication for treatment is striking.”
Potential solutions: ‘Shift the conversation’
In terms of potential solutions to the problem, Dr. Silverstein suggested laboratories change reference ranges so that only thyrotropin values greater than 10 mIU/L are reported as abnormal.
“Studies have shown that changing the thyrotropin reference range is associated with clinicians’ prescribing patterns,” he noted.
Dr. Brito agreed, noting that “there are many guidelines with different hypothyroidism thresholds, so we need to be more consistent about the message to clinicians.
“In addition, we have to come up with different approaches to symptoms that have nothing to do with levothyroxine,” Dr. Brito said.
“I try to explain to patients that it’s very unlikely that subclinical hypothyroidism would be driving significant symptoms like fatigue, weight gain, and hair loss,” Dr. Brito said. “So, one approach is to shift the conversation from how your thyroid is causing this to ‘how are we going to treat the symptoms?’ ”
The study was supported by the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery. Dr. Silverstein has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Does early delivery for FGR affect school outcomes?
Iatrogenic delivery for suspected fetal growth restriction (FGR) may be associated with an increased likelihood of poorer school outcomes among infants born severely small for gestational age, a study of children in Australia suggests.
researchers reported in JAMA.
“It raises the question: in our efforts to improve outcomes in babies that are small, are we potentially doing more harm than good?” said Robert M. Silver, MD, of the department of obstetrics and gynecology at the University of Utah, Salt Lake City, who was not involved in the study. “I think that is a very important question to ask.”
However, “we can’t make that conclusion based on this one study,” he said in an interview. It could be that, in cases where severely small infants were delivered early, there may have been testing that indicated acute risks, and these infants may have tended to be sicker overall. “It may have been that if those babies weren’t delivered, they would have suffered a stillbirth or major brain injury,” Dr. Silver said. “It is really important that we acknowledge that we shouldn’t change our clinical practice” based on this one study.”
At the same time, the study underscores questions and challenges that surround the definition, identification, and management of suspected FGR, Dr. Silver said.
The study authors described their research as exploratory. In a related editorial Dr. Silver and Nathan R. Blue, MD said the findings should be considered hypothesis generating.
For the study, Roshan John Selvaratnam, BMedSc(Hons), a researcher affiliated with Monash University, Melbourne, and colleagues analyzed data from 181,902 children with developmental outcomes and 425,717 children with educational outcomes in Australia. They included children born at 32 weeks’ or more gestation between 2003 and 2013.
Severely small infants delivered early for suspected FGR had an average gestation of 37.9 weeks, whereas those not suspected of having FGR had an average gestation of 39.4 weeks.
Among infants who were severely small for gestational age, those delivered early for suspected FGR were more likely to be in the bottom 10th percentile on at least two developmental domains when they started school, compared with those not suspected of having FGR (16.2% vs. 12.7%; adjusted odds ratio, 1.36). They also were more likely to have low test scores in subsequent years. In grade 7, for example, the adjusted odds ratio for scoring below the national minimum standard on at least two educational domains was 1.33 (13.4% vs. 10.5%).
The researchers defined severely small for gestational age as birth weight below the third percentile. Among infants with normal growth, defined as birth weight at the 10th percentile or greater, school outcomes did not significantly differ between those with early delivery for suspected FGR and those not suspected of having FGR. Approximately 8% of the infants with normal growth had poor developmental outcomes.
The study authors described the dilemma that clinicians face with suspected FGR: “Either intervene early to prevent a small risk of stillbirth but potentially cause immediate and lifelong harm to the child or accept the increasing risk of stillbirth associated with prolonging the pregnancy to avoid more common neonatal and longer-term morbidities.”
It could be that severely small infants with suspected FGR in the study were “more compromised than those not suspected of having FGR,” which might explain the outcomes, Mr. Selvaratnam and coauthors wrote.
Another more plausible explanation is that “iatrogenic prematurity was harmful,” they said.
The researchers were unable to adjust for many factors that may influence academic success, including smoking and alcohol use during pregnancy, maternal body mass index, and breastfeeding, they noted. They also lacked information about the etiology for FGR and whether children had genetic abnormalities.
The study also does not take into account neonatal, infant, and childhood complications, Dr. Silver and Dr. Blue wrote in their editorial. “Nonetheless, these data are a welcome contribution given the knowledge gaps with regard to the optimal obstetric management of FGR.”
The establishment of a diagnostic standard for FGR is needed to properly investigate ways to improve risk stratification, diagnosis, and management, Dr. Silver and Dr. Blue added.
“What we have to do is get better at predicting which babies are at very high risk for continuing the pregnancy and which babies are at low risk for continuing the pregnancy so that we can better decide which babies would benefit from slightly early delivery,” Dr. Silver said.
Improved detection and management of FGR may be on the horizon. “Our ability to image the placental function has gotten a lot better, and I think that is really going to help us,” Dr. Silver said. Studies that aim to further improve the ability to assess whether babies are getting adequate blood flow during pregnancy are ongoing, which could further help doctors evaluate risks.
The study investigators and Dr. Silver had no conflict of interest disclosures. Dr. Blue disclosed grants from Samsung Medison and personal fees from Elsevier. The study was supported by a grant from the Australian government’s National Health and Medical Research Council Program, and Mr. Selvaratnam is supported by scholarships from an Australian government research training program and the National Centre of Research Excellence in Stillbirth.
Iatrogenic delivery for suspected fetal growth restriction (FGR) may be associated with an increased likelihood of poorer school outcomes among infants born severely small for gestational age, a study of children in Australia suggests.
researchers reported in JAMA.
“It raises the question: in our efforts to improve outcomes in babies that are small, are we potentially doing more harm than good?” said Robert M. Silver, MD, of the department of obstetrics and gynecology at the University of Utah, Salt Lake City, who was not involved in the study. “I think that is a very important question to ask.”
However, “we can’t make that conclusion based on this one study,” he said in an interview. It could be that, in cases where severely small infants were delivered early, there may have been testing that indicated acute risks, and these infants may have tended to be sicker overall. “It may have been that if those babies weren’t delivered, they would have suffered a stillbirth or major brain injury,” Dr. Silver said. “It is really important that we acknowledge that we shouldn’t change our clinical practice” based on this one study.”
At the same time, the study underscores questions and challenges that surround the definition, identification, and management of suspected FGR, Dr. Silver said.
The study authors described their research as exploratory. In a related editorial Dr. Silver and Nathan R. Blue, MD said the findings should be considered hypothesis generating.
For the study, Roshan John Selvaratnam, BMedSc(Hons), a researcher affiliated with Monash University, Melbourne, and colleagues analyzed data from 181,902 children with developmental outcomes and 425,717 children with educational outcomes in Australia. They included children born at 32 weeks’ or more gestation between 2003 and 2013.
Severely small infants delivered early for suspected FGR had an average gestation of 37.9 weeks, whereas those not suspected of having FGR had an average gestation of 39.4 weeks.
Among infants who were severely small for gestational age, those delivered early for suspected FGR were more likely to be in the bottom 10th percentile on at least two developmental domains when they started school, compared with those not suspected of having FGR (16.2% vs. 12.7%; adjusted odds ratio, 1.36). They also were more likely to have low test scores in subsequent years. In grade 7, for example, the adjusted odds ratio for scoring below the national minimum standard on at least two educational domains was 1.33 (13.4% vs. 10.5%).
The researchers defined severely small for gestational age as birth weight below the third percentile. Among infants with normal growth, defined as birth weight at the 10th percentile or greater, school outcomes did not significantly differ between those with early delivery for suspected FGR and those not suspected of having FGR. Approximately 8% of the infants with normal growth had poor developmental outcomes.
The study authors described the dilemma that clinicians face with suspected FGR: “Either intervene early to prevent a small risk of stillbirth but potentially cause immediate and lifelong harm to the child or accept the increasing risk of stillbirth associated with prolonging the pregnancy to avoid more common neonatal and longer-term morbidities.”
It could be that severely small infants with suspected FGR in the study were “more compromised than those not suspected of having FGR,” which might explain the outcomes, Mr. Selvaratnam and coauthors wrote.
Another more plausible explanation is that “iatrogenic prematurity was harmful,” they said.
The researchers were unable to adjust for many factors that may influence academic success, including smoking and alcohol use during pregnancy, maternal body mass index, and breastfeeding, they noted. They also lacked information about the etiology for FGR and whether children had genetic abnormalities.
The study also does not take into account neonatal, infant, and childhood complications, Dr. Silver and Dr. Blue wrote in their editorial. “Nonetheless, these data are a welcome contribution given the knowledge gaps with regard to the optimal obstetric management of FGR.”
The establishment of a diagnostic standard for FGR is needed to properly investigate ways to improve risk stratification, diagnosis, and management, Dr. Silver and Dr. Blue added.
“What we have to do is get better at predicting which babies are at very high risk for continuing the pregnancy and which babies are at low risk for continuing the pregnancy so that we can better decide which babies would benefit from slightly early delivery,” Dr. Silver said.
Improved detection and management of FGR may be on the horizon. “Our ability to image the placental function has gotten a lot better, and I think that is really going to help us,” Dr. Silver said. Studies that aim to further improve the ability to assess whether babies are getting adequate blood flow during pregnancy are ongoing, which could further help doctors evaluate risks.
The study investigators and Dr. Silver had no conflict of interest disclosures. Dr. Blue disclosed grants from Samsung Medison and personal fees from Elsevier. The study was supported by a grant from the Australian government’s National Health and Medical Research Council Program, and Mr. Selvaratnam is supported by scholarships from an Australian government research training program and the National Centre of Research Excellence in Stillbirth.
Iatrogenic delivery for suspected fetal growth restriction (FGR) may be associated with an increased likelihood of poorer school outcomes among infants born severely small for gestational age, a study of children in Australia suggests.
researchers reported in JAMA.
“It raises the question: in our efforts to improve outcomes in babies that are small, are we potentially doing more harm than good?” said Robert M. Silver, MD, of the department of obstetrics and gynecology at the University of Utah, Salt Lake City, who was not involved in the study. “I think that is a very important question to ask.”
However, “we can’t make that conclusion based on this one study,” he said in an interview. It could be that, in cases where severely small infants were delivered early, there may have been testing that indicated acute risks, and these infants may have tended to be sicker overall. “It may have been that if those babies weren’t delivered, they would have suffered a stillbirth or major brain injury,” Dr. Silver said. “It is really important that we acknowledge that we shouldn’t change our clinical practice” based on this one study.”
At the same time, the study underscores questions and challenges that surround the definition, identification, and management of suspected FGR, Dr. Silver said.
The study authors described their research as exploratory. In a related editorial Dr. Silver and Nathan R. Blue, MD said the findings should be considered hypothesis generating.
For the study, Roshan John Selvaratnam, BMedSc(Hons), a researcher affiliated with Monash University, Melbourne, and colleagues analyzed data from 181,902 children with developmental outcomes and 425,717 children with educational outcomes in Australia. They included children born at 32 weeks’ or more gestation between 2003 and 2013.
Severely small infants delivered early for suspected FGR had an average gestation of 37.9 weeks, whereas those not suspected of having FGR had an average gestation of 39.4 weeks.
Among infants who were severely small for gestational age, those delivered early for suspected FGR were more likely to be in the bottom 10th percentile on at least two developmental domains when they started school, compared with those not suspected of having FGR (16.2% vs. 12.7%; adjusted odds ratio, 1.36). They also were more likely to have low test scores in subsequent years. In grade 7, for example, the adjusted odds ratio for scoring below the national minimum standard on at least two educational domains was 1.33 (13.4% vs. 10.5%).
The researchers defined severely small for gestational age as birth weight below the third percentile. Among infants with normal growth, defined as birth weight at the 10th percentile or greater, school outcomes did not significantly differ between those with early delivery for suspected FGR and those not suspected of having FGR. Approximately 8% of the infants with normal growth had poor developmental outcomes.
The study authors described the dilemma that clinicians face with suspected FGR: “Either intervene early to prevent a small risk of stillbirth but potentially cause immediate and lifelong harm to the child or accept the increasing risk of stillbirth associated with prolonging the pregnancy to avoid more common neonatal and longer-term morbidities.”
It could be that severely small infants with suspected FGR in the study were “more compromised than those not suspected of having FGR,” which might explain the outcomes, Mr. Selvaratnam and coauthors wrote.
Another more plausible explanation is that “iatrogenic prematurity was harmful,” they said.
The researchers were unable to adjust for many factors that may influence academic success, including smoking and alcohol use during pregnancy, maternal body mass index, and breastfeeding, they noted. They also lacked information about the etiology for FGR and whether children had genetic abnormalities.
The study also does not take into account neonatal, infant, and childhood complications, Dr. Silver and Dr. Blue wrote in their editorial. “Nonetheless, these data are a welcome contribution given the knowledge gaps with regard to the optimal obstetric management of FGR.”
The establishment of a diagnostic standard for FGR is needed to properly investigate ways to improve risk stratification, diagnosis, and management, Dr. Silver and Dr. Blue added.
“What we have to do is get better at predicting which babies are at very high risk for continuing the pregnancy and which babies are at low risk for continuing the pregnancy so that we can better decide which babies would benefit from slightly early delivery,” Dr. Silver said.
Improved detection and management of FGR may be on the horizon. “Our ability to image the placental function has gotten a lot better, and I think that is really going to help us,” Dr. Silver said. Studies that aim to further improve the ability to assess whether babies are getting adequate blood flow during pregnancy are ongoing, which could further help doctors evaluate risks.
The study investigators and Dr. Silver had no conflict of interest disclosures. Dr. Blue disclosed grants from Samsung Medison and personal fees from Elsevier. The study was supported by a grant from the Australian government’s National Health and Medical Research Council Program, and Mr. Selvaratnam is supported by scholarships from an Australian government research training program and the National Centre of Research Excellence in Stillbirth.
FROM JAMA
Why Size (of Your Differential) Matters
ANSWER
The correct answer is all of the above (choice “f”).
DISCUSSION
The most likely diagnostic explanation for this patient’s presentation is erythema annulare centrifugum (EAC; choice “c”). However, this diagnosis is often difficult to establish, in part because of the broad differential and also because the very existence of EAC is far from well established.
The overwhelming consensus is that EAC represents a hypersensitivity reaction to an unknown antigen. It can be triggered by a wide variety of micro-organisms, stress, or even pregnancy.
In this case, there was no clinical or historical reason to suspect an underlying cancer, Lyme disease, or lupus, nor did the biopsy results suggest any of these.
The key takeaway here is to urge providers to avoid jumping onto a diagnostic bandwagon before considering a wider differential. Indeed, size matters when it relates to the length of one’s differential diagnosis list. If you don’t consider it, you can’t diagnose it.
TREATMENT
Fortunately, EAC nearly always resolves, with or without treatment. This patient received reassurance as such but was scheduled to return for a check of his lesions in 2 months. At that point, they had resolved.
ANSWER
The correct answer is all of the above (choice “f”).
DISCUSSION
The most likely diagnostic explanation for this patient’s presentation is erythema annulare centrifugum (EAC; choice “c”). However, this diagnosis is often difficult to establish, in part because of the broad differential and also because the very existence of EAC is far from well established.
The overwhelming consensus is that EAC represents a hypersensitivity reaction to an unknown antigen. It can be triggered by a wide variety of micro-organisms, stress, or even pregnancy.
In this case, there was no clinical or historical reason to suspect an underlying cancer, Lyme disease, or lupus, nor did the biopsy results suggest any of these.
The key takeaway here is to urge providers to avoid jumping onto a diagnostic bandwagon before considering a wider differential. Indeed, size matters when it relates to the length of one’s differential diagnosis list. If you don’t consider it, you can’t diagnose it.
TREATMENT
Fortunately, EAC nearly always resolves, with or without treatment. This patient received reassurance as such but was scheduled to return for a check of his lesions in 2 months. At that point, they had resolved.
ANSWER
The correct answer is all of the above (choice “f”).
DISCUSSION
The most likely diagnostic explanation for this patient’s presentation is erythema annulare centrifugum (EAC; choice “c”). However, this diagnosis is often difficult to establish, in part because of the broad differential and also because the very existence of EAC is far from well established.
The overwhelming consensus is that EAC represents a hypersensitivity reaction to an unknown antigen. It can be triggered by a wide variety of micro-organisms, stress, or even pregnancy.
In this case, there was no clinical or historical reason to suspect an underlying cancer, Lyme disease, or lupus, nor did the biopsy results suggest any of these.
The key takeaway here is to urge providers to avoid jumping onto a diagnostic bandwagon before considering a wider differential. Indeed, size matters when it relates to the length of one’s differential diagnosis list. If you don’t consider it, you can’t diagnose it.
TREATMENT
Fortunately, EAC nearly always resolves, with or without treatment. This patient received reassurance as such but was scheduled to return for a check of his lesions in 2 months. At that point, they had resolved.
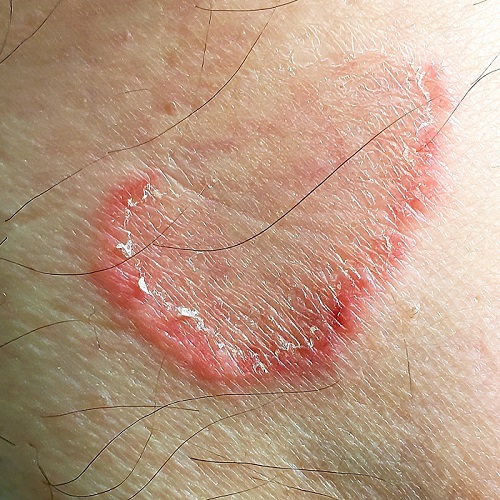
A 50-year-old man is astonished when his “fungal infection” fails to respond to an unknown OTC topical cream a friend advised him to use.
For several weeks, he’s been plagued by the slightly itchy lesions that appeared on his leg without known cause. His friend assured him that the shape and configuration of the lesions could only represent one thing: “ringworm,” that is, fungal infection.
However, in clinic, the patient denies any contact with animals or children and reports that his job is inside only, never involving the great outdoors. He has felt fine throughout the lesions’ tenure, going so far as to say he is “in perfect health.” He has been in a mutually monogamous relationship for many years.
The lesions (4 in total) are located on the medial aspect of the left leg, extending into the popliteal area. At first glance, they appear to be peripherally scaly, pink, and annular. Close inspection reveals that most of the scaling is not on the outer edge; it is confined to the inside of the border, a phenomenon termed trailing or centripetal scaling. The lesions all show an arciform morphology in the shape of a “C.”
KOH examination of the scale shows no fungal elements. Shave biopsy reveals a dense perivascular lymphocytic infiltrate, moderate parakeratosis, and perhaps most importantly, no fungal elements in the stratum corneum.
There are no palpable lymph nodes in the groin on the affected side.
Syphilis prevalence in MSM 15 times higher than in general population
Worldwide, nearly 8% of men who have sex with men (MSM) may have syphilis, a new systematic review and meta-analysis suggests. This estimate, generated from 275 studies across 77 countries, is 15 times greater than the most recent estimates of syphilis prevalence in men in a general population.
“That disparity is absolutely unacceptable,” Matthew Chico, PhD, associate professor at the London School of Hygiene and Tropical Medicine, and senior author of the review, said in an interview.
Although the World Health Organization (WHO) aims to reduce the global prevalence of syphilis by 90% by 2030, an ambitious goal set in 2016, recent research suggests syphilis numbers are moving in the opposite direction. Cases in the United States rose 74% between 2015 and 2019, and other nations, such as Australia, South Korea, and the United Kingdom, are seeing similar trends.
Syphilis prevalence is generally higher in MSM, largely in subpopulations of men who have multiple sexual partners, Kenneth Mayer, MD, said in an interview. Dr. Mayer is medical research director at the Fenway Institute, Boston, and was not involved with the study.
Health literacy, lack of access to care, and medical mistrust can all be challenges to screening, identifying, and treating the infection in this population.
Reducing syphilis cases will require focusing interventions on higher-risk groups such as MSM, said Dr. Chico; however, there was “a real dearth in knowledge about the most likely prevalence of syphilis among MSM on a global level,” he said.
To help fill in the gaps, Dr. Chico and his research team collected studies that included syphilis prevalence data for MSM published between Jan. 1, 2000, and Feb. 1, 2020. Researchers excluded studies that included only MSM living with HIV, injection drug users, patients who routinely visit sexually transmitted infection (STI) clinics, and people seeking care only for STIs or other genital symptoms, because these studies would have skewed global syphilis prevalence estimates higher.
Their review, published July 8 in The Lancet Global Health, found that the pooled global prevalence of syphilis from 2000-2020 in MSM was 7.5%. It ranged from 1.9% in Australia and New Zealand to 10.6% in Latin America and the Caribbean. In comparison, the WHO estimates that globally, 0.5% of men in a general population have syphilis, a 15-fold difference.
This elevated estimate is not surprising, and the review provides a more international view of syphilis. Earlier attempts to estimate the prevalence of syphilis among MSM were generally conducted in higher-income countries such as the United States, Dr. Mayer said. “It’s important that clinicians recognize that this is a global health issue, so they can do the appropriate screening.”
The review found that regions in which the prevalence of HIV was above 5% had higher rates of syphilis (8.7%) compared to regions in which the prevalence of HIV was below 5% (6.6%). Pooled syphilis prevalence estimates were also higher for lower-middle-income and upper-middle-income countries (8.7% and 8.6%, respectively).
Global syphilis prevalence dipped from 8.9% in studies from 2000 to 2009 to 6.6% in studies from 2010 to 2020. In Europe, Northern America, Latin America, the Caribbean, and Oceania (excluding Australia and New Zealand), syphilis prevalence estimates for 2015-2020 were higher compared with 2010-2014.
The authors acknowledged that there were some limitations to the study, particularly that regions of Eastern and Southeastern Asia contributed more than half (54.5%) of the global data points used in the study and accounted for more than 82% of the study’s participants. This highlights the lack of data from other regions around the world, Dr. Chico said.
Dr. Chico said these findings “serve as a clarion call to action” to focus interventions on groups at higher risk for syphilis, such as MSM, in the effort to drastically reduce syphilis cases around the world. Dr. Mayer agrees. “[Syphilis] is a readily diagnosable and treatable infection,” he said. “It definitely is something that we should be able to get a handle on, but that requires paying attention to the different subgroups who have particularly high rates of the infection.”
A version of this article first appeared on Medscape.com.
Worldwide, nearly 8% of men who have sex with men (MSM) may have syphilis, a new systematic review and meta-analysis suggests. This estimate, generated from 275 studies across 77 countries, is 15 times greater than the most recent estimates of syphilis prevalence in men in a general population.
“That disparity is absolutely unacceptable,” Matthew Chico, PhD, associate professor at the London School of Hygiene and Tropical Medicine, and senior author of the review, said in an interview.
Although the World Health Organization (WHO) aims to reduce the global prevalence of syphilis by 90% by 2030, an ambitious goal set in 2016, recent research suggests syphilis numbers are moving in the opposite direction. Cases in the United States rose 74% between 2015 and 2019, and other nations, such as Australia, South Korea, and the United Kingdom, are seeing similar trends.
Syphilis prevalence is generally higher in MSM, largely in subpopulations of men who have multiple sexual partners, Kenneth Mayer, MD, said in an interview. Dr. Mayer is medical research director at the Fenway Institute, Boston, and was not involved with the study.
Health literacy, lack of access to care, and medical mistrust can all be challenges to screening, identifying, and treating the infection in this population.
Reducing syphilis cases will require focusing interventions on higher-risk groups such as MSM, said Dr. Chico; however, there was “a real dearth in knowledge about the most likely prevalence of syphilis among MSM on a global level,” he said.
To help fill in the gaps, Dr. Chico and his research team collected studies that included syphilis prevalence data for MSM published between Jan. 1, 2000, and Feb. 1, 2020. Researchers excluded studies that included only MSM living with HIV, injection drug users, patients who routinely visit sexually transmitted infection (STI) clinics, and people seeking care only for STIs or other genital symptoms, because these studies would have skewed global syphilis prevalence estimates higher.
Their review, published July 8 in The Lancet Global Health, found that the pooled global prevalence of syphilis from 2000-2020 in MSM was 7.5%. It ranged from 1.9% in Australia and New Zealand to 10.6% in Latin America and the Caribbean. In comparison, the WHO estimates that globally, 0.5% of men in a general population have syphilis, a 15-fold difference.
This elevated estimate is not surprising, and the review provides a more international view of syphilis. Earlier attempts to estimate the prevalence of syphilis among MSM were generally conducted in higher-income countries such as the United States, Dr. Mayer said. “It’s important that clinicians recognize that this is a global health issue, so they can do the appropriate screening.”
The review found that regions in which the prevalence of HIV was above 5% had higher rates of syphilis (8.7%) compared to regions in which the prevalence of HIV was below 5% (6.6%). Pooled syphilis prevalence estimates were also higher for lower-middle-income and upper-middle-income countries (8.7% and 8.6%, respectively).
Global syphilis prevalence dipped from 8.9% in studies from 2000 to 2009 to 6.6% in studies from 2010 to 2020. In Europe, Northern America, Latin America, the Caribbean, and Oceania (excluding Australia and New Zealand), syphilis prevalence estimates for 2015-2020 were higher compared with 2010-2014.
The authors acknowledged that there were some limitations to the study, particularly that regions of Eastern and Southeastern Asia contributed more than half (54.5%) of the global data points used in the study and accounted for more than 82% of the study’s participants. This highlights the lack of data from other regions around the world, Dr. Chico said.
Dr. Chico said these findings “serve as a clarion call to action” to focus interventions on groups at higher risk for syphilis, such as MSM, in the effort to drastically reduce syphilis cases around the world. Dr. Mayer agrees. “[Syphilis] is a readily diagnosable and treatable infection,” he said. “It definitely is something that we should be able to get a handle on, but that requires paying attention to the different subgroups who have particularly high rates of the infection.”
A version of this article first appeared on Medscape.com.
Worldwide, nearly 8% of men who have sex with men (MSM) may have syphilis, a new systematic review and meta-analysis suggests. This estimate, generated from 275 studies across 77 countries, is 15 times greater than the most recent estimates of syphilis prevalence in men in a general population.
“That disparity is absolutely unacceptable,” Matthew Chico, PhD, associate professor at the London School of Hygiene and Tropical Medicine, and senior author of the review, said in an interview.
Although the World Health Organization (WHO) aims to reduce the global prevalence of syphilis by 90% by 2030, an ambitious goal set in 2016, recent research suggests syphilis numbers are moving in the opposite direction. Cases in the United States rose 74% between 2015 and 2019, and other nations, such as Australia, South Korea, and the United Kingdom, are seeing similar trends.
Syphilis prevalence is generally higher in MSM, largely in subpopulations of men who have multiple sexual partners, Kenneth Mayer, MD, said in an interview. Dr. Mayer is medical research director at the Fenway Institute, Boston, and was not involved with the study.
Health literacy, lack of access to care, and medical mistrust can all be challenges to screening, identifying, and treating the infection in this population.
Reducing syphilis cases will require focusing interventions on higher-risk groups such as MSM, said Dr. Chico; however, there was “a real dearth in knowledge about the most likely prevalence of syphilis among MSM on a global level,” he said.
To help fill in the gaps, Dr. Chico and his research team collected studies that included syphilis prevalence data for MSM published between Jan. 1, 2000, and Feb. 1, 2020. Researchers excluded studies that included only MSM living with HIV, injection drug users, patients who routinely visit sexually transmitted infection (STI) clinics, and people seeking care only for STIs or other genital symptoms, because these studies would have skewed global syphilis prevalence estimates higher.
Their review, published July 8 in The Lancet Global Health, found that the pooled global prevalence of syphilis from 2000-2020 in MSM was 7.5%. It ranged from 1.9% in Australia and New Zealand to 10.6% in Latin America and the Caribbean. In comparison, the WHO estimates that globally, 0.5% of men in a general population have syphilis, a 15-fold difference.
This elevated estimate is not surprising, and the review provides a more international view of syphilis. Earlier attempts to estimate the prevalence of syphilis among MSM were generally conducted in higher-income countries such as the United States, Dr. Mayer said. “It’s important that clinicians recognize that this is a global health issue, so they can do the appropriate screening.”
The review found that regions in which the prevalence of HIV was above 5% had higher rates of syphilis (8.7%) compared to regions in which the prevalence of HIV was below 5% (6.6%). Pooled syphilis prevalence estimates were also higher for lower-middle-income and upper-middle-income countries (8.7% and 8.6%, respectively).
Global syphilis prevalence dipped from 8.9% in studies from 2000 to 2009 to 6.6% in studies from 2010 to 2020. In Europe, Northern America, Latin America, the Caribbean, and Oceania (excluding Australia and New Zealand), syphilis prevalence estimates for 2015-2020 were higher compared with 2010-2014.
The authors acknowledged that there were some limitations to the study, particularly that regions of Eastern and Southeastern Asia contributed more than half (54.5%) of the global data points used in the study and accounted for more than 82% of the study’s participants. This highlights the lack of data from other regions around the world, Dr. Chico said.
Dr. Chico said these findings “serve as a clarion call to action” to focus interventions on groups at higher risk for syphilis, such as MSM, in the effort to drastically reduce syphilis cases around the world. Dr. Mayer agrees. “[Syphilis] is a readily diagnosable and treatable infection,” he said. “It definitely is something that we should be able to get a handle on, but that requires paying attention to the different subgroups who have particularly high rates of the infection.”
A version of this article first appeared on Medscape.com.
Targeted outreach increases Black COVID-19 vaccination rates
Thoughtful, targeted approaches are needed to increase COVID-19 vaccination rates in Black and Latinx communities, which often distrust the health care system and face barriers to vaccine access, new data show.
“Black, Latinx, and Native American individuals represent about a combined 60% of COVID-19 deaths, despite comprising significantly less of the United States population,” said Jacinda C. Abdul-Mutakabbir, PharmD, from Loma Linda (Calif.) University.
“To put this into perspective, Black individuals represent 13.4% of the United States population, while Native Americans represent 1.6%, clearly showing the disproportionate outcomes here,” she explained during her online presentation at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
The vaccine creates an opportunity to change the disproportionate way COVID-19 has affected racial and ethnic communities, said Dr. Abdul-Mutakabbir, but “a long history of mistreatment within the U.S. health care system decreases their trust for the system to use fair practices when delivering these vaccines.”
For people in minority communities, often “the fear of cost associated with health care keeps them from being vaccinated,” she said. “Also, there is a lack of vaccines actually allocated to these communities, or inconsistent computer-based sign-ups that make WiFi mandatory, which in turn has created additional barriers for vaccination access.”
Loma Linda University maintains the largest mass-vaccination site in San Bernardino County, the fourth-largest county in southern California. However, only 3.0% of the people vaccinated there have been Black. And although 8% of the state’s population is Black, only 2.7% of the Black population has been vaccinated.
In contrast, Black Californians have accounted for about 20% of COVID-19 cases in the state, and 20%-30% of COVID-19 deaths.
To promote equitable access to COVID-19 vaccines, Dr. Abdul-Mutakabbir and colleagues developed a “three-tiered approach.” First, they had local Black faith leaders hold summits ahead of the vaccination clinics. Next, at those summits, they had a Black pharmacist educate attendees about the vaccines. And finally, they held a low-barrier community vaccination clinic in a Black community, where the pharmacist oversaw the transport and preparation of the vaccines.
Because access, transportation, and internet are all barriers to vaccination, the clinic used paper-based registration and was held as a pop-up clinic at a local Black church. The team held two clinics for the first Moderna dose, one clinic for the second Moderna dose, and one clinic for the Johnson & Johnson one-dose vaccine.
During the Moderna first-dose clinics, 673 vaccinations were administered, and during the second-dose clinic, 366 were administered. Early data showed a return rate of 87%, but the team has yet to update the final numbers, Dr. Abdul-Mutakabbir reported. During the Johnson & Johnson clinic, 314 vaccinations were administered, nearly half to Black people. After the community vaccination clinics, the mass vaccination site saw a 0.6% increase in vaccinations for Black people.
Dr. Abdul-Mutakabbir’s team also held three community clinics in Latinx communities. During the first-dose Moderna pop-up clinic, 258 vaccinations were administered, and during the second-dose clinic, 253 were, for a 98% return rate. Approximately 92% of those vaccinated were Latinx.
The study findings are not surprising, said Rhea Boyd, MD, director of equity and justice for California Children’s Trust.
“The barriers to vaccination are known and clear,” she said in an interview. “Mobile clinics with paper appointments address a number of those barriers head on, like transportation, internet access, and accessibility. Having Black providers leading the effort and church leaders involved also has been shown to increase confidence in the operations and process.”
Information campaigns can help counter online disinformation. Ultimately, however, “the main barrier to vaccination is access,” Dr. Boyd said. “Address access and rates will increase.”
The health inequities seen in vaccination rates among Black and Latinx people “are a product of structural and systemic racism,” Dr. Abdul-Mutakabbir said. “To create equitable processes, it is essential that we evaluate how we approach each of these different minoritized groups.”
Dr. Abdul-Mutakabbir disclosed no relevant financial relationships. Dr. Boyd codeveloped THE CONVERSATION, a national campaign to bring credible information about the COVID vaccines to Black and Latinx communities in partnership with KFF, BCAC, and Unidos US.
A version of this article first appeared on Medscape.com.
Thoughtful, targeted approaches are needed to increase COVID-19 vaccination rates in Black and Latinx communities, which often distrust the health care system and face barriers to vaccine access, new data show.
“Black, Latinx, and Native American individuals represent about a combined 60% of COVID-19 deaths, despite comprising significantly less of the United States population,” said Jacinda C. Abdul-Mutakabbir, PharmD, from Loma Linda (Calif.) University.
“To put this into perspective, Black individuals represent 13.4% of the United States population, while Native Americans represent 1.6%, clearly showing the disproportionate outcomes here,” she explained during her online presentation at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
The vaccine creates an opportunity to change the disproportionate way COVID-19 has affected racial and ethnic communities, said Dr. Abdul-Mutakabbir, but “a long history of mistreatment within the U.S. health care system decreases their trust for the system to use fair practices when delivering these vaccines.”
For people in minority communities, often “the fear of cost associated with health care keeps them from being vaccinated,” she said. “Also, there is a lack of vaccines actually allocated to these communities, or inconsistent computer-based sign-ups that make WiFi mandatory, which in turn has created additional barriers for vaccination access.”
Loma Linda University maintains the largest mass-vaccination site in San Bernardino County, the fourth-largest county in southern California. However, only 3.0% of the people vaccinated there have been Black. And although 8% of the state’s population is Black, only 2.7% of the Black population has been vaccinated.
In contrast, Black Californians have accounted for about 20% of COVID-19 cases in the state, and 20%-30% of COVID-19 deaths.
To promote equitable access to COVID-19 vaccines, Dr. Abdul-Mutakabbir and colleagues developed a “three-tiered approach.” First, they had local Black faith leaders hold summits ahead of the vaccination clinics. Next, at those summits, they had a Black pharmacist educate attendees about the vaccines. And finally, they held a low-barrier community vaccination clinic in a Black community, where the pharmacist oversaw the transport and preparation of the vaccines.
Because access, transportation, and internet are all barriers to vaccination, the clinic used paper-based registration and was held as a pop-up clinic at a local Black church. The team held two clinics for the first Moderna dose, one clinic for the second Moderna dose, and one clinic for the Johnson & Johnson one-dose vaccine.
During the Moderna first-dose clinics, 673 vaccinations were administered, and during the second-dose clinic, 366 were administered. Early data showed a return rate of 87%, but the team has yet to update the final numbers, Dr. Abdul-Mutakabbir reported. During the Johnson & Johnson clinic, 314 vaccinations were administered, nearly half to Black people. After the community vaccination clinics, the mass vaccination site saw a 0.6% increase in vaccinations for Black people.
Dr. Abdul-Mutakabbir’s team also held three community clinics in Latinx communities. During the first-dose Moderna pop-up clinic, 258 vaccinations were administered, and during the second-dose clinic, 253 were, for a 98% return rate. Approximately 92% of those vaccinated were Latinx.
The study findings are not surprising, said Rhea Boyd, MD, director of equity and justice for California Children’s Trust.
“The barriers to vaccination are known and clear,” she said in an interview. “Mobile clinics with paper appointments address a number of those barriers head on, like transportation, internet access, and accessibility. Having Black providers leading the effort and church leaders involved also has been shown to increase confidence in the operations and process.”
Information campaigns can help counter online disinformation. Ultimately, however, “the main barrier to vaccination is access,” Dr. Boyd said. “Address access and rates will increase.”
The health inequities seen in vaccination rates among Black and Latinx people “are a product of structural and systemic racism,” Dr. Abdul-Mutakabbir said. “To create equitable processes, it is essential that we evaluate how we approach each of these different minoritized groups.”
Dr. Abdul-Mutakabbir disclosed no relevant financial relationships. Dr. Boyd codeveloped THE CONVERSATION, a national campaign to bring credible information about the COVID vaccines to Black and Latinx communities in partnership with KFF, BCAC, and Unidos US.
A version of this article first appeared on Medscape.com.
Thoughtful, targeted approaches are needed to increase COVID-19 vaccination rates in Black and Latinx communities, which often distrust the health care system and face barriers to vaccine access, new data show.
“Black, Latinx, and Native American individuals represent about a combined 60% of COVID-19 deaths, despite comprising significantly less of the United States population,” said Jacinda C. Abdul-Mutakabbir, PharmD, from Loma Linda (Calif.) University.
“To put this into perspective, Black individuals represent 13.4% of the United States population, while Native Americans represent 1.6%, clearly showing the disproportionate outcomes here,” she explained during her online presentation at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
The vaccine creates an opportunity to change the disproportionate way COVID-19 has affected racial and ethnic communities, said Dr. Abdul-Mutakabbir, but “a long history of mistreatment within the U.S. health care system decreases their trust for the system to use fair practices when delivering these vaccines.”
For people in minority communities, often “the fear of cost associated with health care keeps them from being vaccinated,” she said. “Also, there is a lack of vaccines actually allocated to these communities, or inconsistent computer-based sign-ups that make WiFi mandatory, which in turn has created additional barriers for vaccination access.”
Loma Linda University maintains the largest mass-vaccination site in San Bernardino County, the fourth-largest county in southern California. However, only 3.0% of the people vaccinated there have been Black. And although 8% of the state’s population is Black, only 2.7% of the Black population has been vaccinated.
In contrast, Black Californians have accounted for about 20% of COVID-19 cases in the state, and 20%-30% of COVID-19 deaths.
To promote equitable access to COVID-19 vaccines, Dr. Abdul-Mutakabbir and colleagues developed a “three-tiered approach.” First, they had local Black faith leaders hold summits ahead of the vaccination clinics. Next, at those summits, they had a Black pharmacist educate attendees about the vaccines. And finally, they held a low-barrier community vaccination clinic in a Black community, where the pharmacist oversaw the transport and preparation of the vaccines.
Because access, transportation, and internet are all barriers to vaccination, the clinic used paper-based registration and was held as a pop-up clinic at a local Black church. The team held two clinics for the first Moderna dose, one clinic for the second Moderna dose, and one clinic for the Johnson & Johnson one-dose vaccine.
During the Moderna first-dose clinics, 673 vaccinations were administered, and during the second-dose clinic, 366 were administered. Early data showed a return rate of 87%, but the team has yet to update the final numbers, Dr. Abdul-Mutakabbir reported. During the Johnson & Johnson clinic, 314 vaccinations were administered, nearly half to Black people. After the community vaccination clinics, the mass vaccination site saw a 0.6% increase in vaccinations for Black people.
Dr. Abdul-Mutakabbir’s team also held three community clinics in Latinx communities. During the first-dose Moderna pop-up clinic, 258 vaccinations were administered, and during the second-dose clinic, 253 were, for a 98% return rate. Approximately 92% of those vaccinated were Latinx.
The study findings are not surprising, said Rhea Boyd, MD, director of equity and justice for California Children’s Trust.
“The barriers to vaccination are known and clear,” she said in an interview. “Mobile clinics with paper appointments address a number of those barriers head on, like transportation, internet access, and accessibility. Having Black providers leading the effort and church leaders involved also has been shown to increase confidence in the operations and process.”
Information campaigns can help counter online disinformation. Ultimately, however, “the main barrier to vaccination is access,” Dr. Boyd said. “Address access and rates will increase.”
The health inequities seen in vaccination rates among Black and Latinx people “are a product of structural and systemic racism,” Dr. Abdul-Mutakabbir said. “To create equitable processes, it is essential that we evaluate how we approach each of these different minoritized groups.”
Dr. Abdul-Mutakabbir disclosed no relevant financial relationships. Dr. Boyd codeveloped THE CONVERSATION, a national campaign to bring credible information about the COVID vaccines to Black and Latinx communities in partnership with KFF, BCAC, and Unidos US.
A version of this article first appeared on Medscape.com.