User login
TEST TEST
test
test
test
Periorbital Changes Induced by Prostaglandin Eye Drops
To the Editor:
A 42-year man presented with hollowing of the upper eyelid and skin discoloration of the left periorbital area of 10 years’ duration. He was a professional mixed martial arts fighter with a history of 2 surgeries for retinal detachment of the left eye 13 years prior to the current presentation. The patient also has macular scarring in the left eye. He denied a history of facial fracture, reconstructive surgery, or other medical conditions. His visual acuity was unknown; however, he did not require corrective glasses. He used 3 prescription ophthalmic eye drops—dorzolamide hydrochloride plus timolol maleate, 10 mL; brimonidine tartrate ophthalmic solution 0.15%, 5 mL; and latanoprost ophthalmic solution 0.005%, 125 μg/2.5 mL—in the left eye to lower intraocular pressure, as therapy for glaucoma. If left untreated, glaucoma can lead to vision loss.
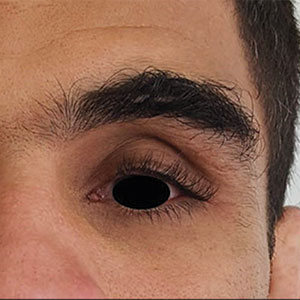
Physical examination revealed periorbital hyperpigmentation on the left side; hypertrichosis and eyelash trichomegaly compared to the right side; and a deep left upper orbital sulcus compared to the right side (Figure). The patient was alert and oriented to person, place, and time. Extraocular movement was intact bilaterally, and his pupillary reflex was symmetric. No tenderness was noted over the affected area on palpation; no subcutaneous masses or lesions were observed or palpated. There was no ocular discharge, the conjunctiva was pink, and the sclera was white bilaterally.
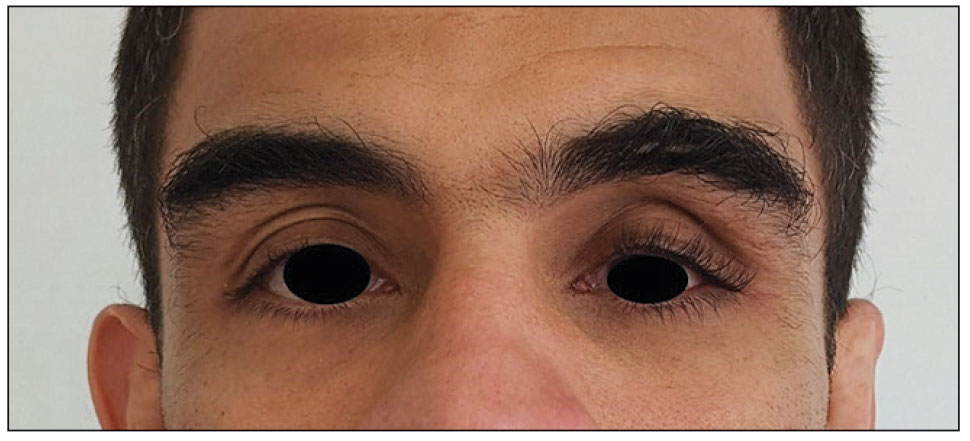
The differential diagnosis included professional trauma-induced orbital changes, nevus of Ota (oculomucodermal melanocytosis), prostaglandin-associated periorbitopathy (PAP), and melasma. Although the patient sustained an injury that caused retinal detachment, he never experienced an orbital bone fracture; additionally, a fracture would not explain the skin discoloration or longer eyelashes. Periorbital nevus of Ota most commonly manifests as a unilateral scleral and brown-bluish skin discoloration but does not cause hollowing of the orbital sulcus or affect the length and thickness of eyelashes. Melasma—bilateral skin hyperpigmentation that most commonly affects women—can be induced by oral contraceptives, antibiotics, heat, sun exposure, and pregnancy. It does not affect the color of the iris or the depth of the scleral sulcus, and it does not increase the length and thickness of eyelashes. Based on the clinical presentation and a review of the eye drops used, he was diagnosed with PAP due to prolonged use of latanoprost ophthalmic solution. The patient was referred to an ophthalmologist for consideration of a switch to a different class of medication.
Of the 3 eye drops used by this patient, latanoprost, a prostaglandin analog, decreases intraocular pressure and is known to cause PAP. This condition comprises a constellation of changes, including upper eyelid ptosis, deepening of the upper eyelid sulcus, involution of dermatochalasis, periorbital fat atrophy, mild enophthalmos (sunken eye), inferior scleral show, increased prominence of eyelid vessels, and tight eyelids.1 Latanoprost most often produces these findings, but all prostaglandin ophthalmic medications can, including the dual-indication bimatoprost, which was approved by the US Food and Drug Administration to reduce elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension but also is used to grow darker, thicker, and longer eyelashes. Clinicians who prescribe bimatoprost for this cosmetic indication should be mindful of the potential for PAP and discuss it with patients.
The prescribing information (PI) for bimatoprost (Latisse; Allergan) does not list PAP as an adverse reaction observed in the 4-month multicenter, double-blind, randomized, vehicle-controlled study of bimatoprost (as Latisse) in 278 adults.2 The PI does list “periorbital and lid changes associated with periorbital fat atrophy and skin tightness resulting in deepening of eyelid sulcus and eyelid ptosis” as an adverse reaction in postmarketing experience. However, according to the PI, the frequency of these adverse reactions cannot be established, as the reporting of such incidents was voluntary and the size of the treated population was uncertain.2
Prostaglandins can cause periorbitopathy by several mechanisms; one speculated cause is that this group of medications might provoke smooth muscle contraction. Prostaglandin medications also have an affinity for fat cells1; atrophy of fat cells can lead to enophthalmos and deepening upper eyelid sulcus. In an observational study of 105 participants who were using a prostaglandin in 1 eye for longer than 1 month (the other eye was used as a control), the overall frequency of prostaglandin-associated periorbitopathy was 93.3% in the bimatoprost group, 41.4% in the latanoprost group, and 70% in the travoprost group, while the frequency of deepening of the upper eyelid sulcus was 80% in the bimatoprost group, 15.7% in the latanoprost group, and 45% in the travoprost group.3 These changes may not be as striking when a patient is using a prostaglandin ophthalmic medication in both eyes and may not be noticed even by the patient. It is prudent for the clinician to take a baseline photograph of the patient when these medications are prescribed to observe for early signs of periorbitopathy. These adverse effects may not be reversible when the medication is discontinued4 and have been observed as early as 4 to 6 weeks after the start of treatment.5
Our patient was counseled that his constellation of PAP findings potentially could be partially reversed over months or even a year or longer if the offending agent was discontinued. However, he was cautioned that cessation of latanoprost first needed to be discussed with his ophthalmologist, who would determine if there was a suitable alternative to a prostaglandin analog for him. The patient’s only concern was the aesthetic appearance of the left periorbital area. A hyaluronic acid filler or fat grafting can be considered for correction of orbital sulcus hollowing; however, we could not locate any long-term studies in which such corrective treatments were applied for PAP. Our patient continues to use latanoprost with no change in the frequency of use. There have been no further changes or progression in the physical appearance of the left eye or periorbital area. The patient has not undergone any corrective treatments.
- Berke SJ. PAP: new concerns for prostaglandin use. Rev Ophthalmol. 2012;19:70.
- Latisse (bimatoprost ophthalmic solution 0.03%). Package insert. Allergan; 2021. Accessed April 11, 2024. https://www.rxabbvie.com/pdf/latisse_pi.pdf
- Kucukevcilioglu M, Bayer A, Uysal Y, et al. Prostaglandin associated periorbitopathy in patients using bimatoprost, latanoprost and travoprost. Clin Exp Ophthalmol. 2014;42:126-131. doi:10.1111/ceo.12163
- Filippopoulos T, Paula JS, Torun N, et al. Periorbital changes associated with topical bimatoprost. Ophthalmic Plast Reconstr Surg. 2008;24:302-307. doi:10.1097/IOP.0b013e31817d81df
- Peplinski LS, Smith KA. Deepening of lid sulcus from topical bimatoprost therapy. Optom Vis Sci. 2004;81:574-577. doi:10.1097/01.opx.0000141791.16683.4a
To the Editor:
A 42-year man presented with hollowing of the upper eyelid and skin discoloration of the left periorbital area of 10 years’ duration. He was a professional mixed martial arts fighter with a history of 2 surgeries for retinal detachment of the left eye 13 years prior to the current presentation. The patient also has macular scarring in the left eye. He denied a history of facial fracture, reconstructive surgery, or other medical conditions. His visual acuity was unknown; however, he did not require corrective glasses. He used 3 prescription ophthalmic eye drops—dorzolamide hydrochloride plus timolol maleate, 10 mL; brimonidine tartrate ophthalmic solution 0.15%, 5 mL; and latanoprost ophthalmic solution 0.005%, 125 μg/2.5 mL—in the left eye to lower intraocular pressure, as therapy for glaucoma. If left untreated, glaucoma can lead to vision loss.
Physical examination revealed periorbital hyperpigmentation on the left side; hypertrichosis and eyelash trichomegaly compared to the right side; and a deep left upper orbital sulcus compared to the right side (Figure). The patient was alert and oriented to person, place, and time. Extraocular movement was intact bilaterally, and his pupillary reflex was symmetric. No tenderness was noted over the affected area on palpation; no subcutaneous masses or lesions were observed or palpated. There was no ocular discharge, the conjunctiva was pink, and the sclera was white bilaterally.

The differential diagnosis included professional trauma-induced orbital changes, nevus of Ota (oculomucodermal melanocytosis), prostaglandin-associated periorbitopathy (PAP), and melasma. Although the patient sustained an injury that caused retinal detachment, he never experienced an orbital bone fracture; additionally, a fracture would not explain the skin discoloration or longer eyelashes. Periorbital nevus of Ota most commonly manifests as a unilateral scleral and brown-bluish skin discoloration but does not cause hollowing of the orbital sulcus or affect the length and thickness of eyelashes. Melasma—bilateral skin hyperpigmentation that most commonly affects women—can be induced by oral contraceptives, antibiotics, heat, sun exposure, and pregnancy. It does not affect the color of the iris or the depth of the scleral sulcus, and it does not increase the length and thickness of eyelashes. Based on the clinical presentation and a review of the eye drops used, he was diagnosed with PAP due to prolonged use of latanoprost ophthalmic solution. The patient was referred to an ophthalmologist for consideration of a switch to a different class of medication.
Of the 3 eye drops used by this patient, latanoprost, a prostaglandin analog, decreases intraocular pressure and is known to cause PAP. This condition comprises a constellation of changes, including upper eyelid ptosis, deepening of the upper eyelid sulcus, involution of dermatochalasis, periorbital fat atrophy, mild enophthalmos (sunken eye), inferior scleral show, increased prominence of eyelid vessels, and tight eyelids.1 Latanoprost most often produces these findings, but all prostaglandin ophthalmic medications can, including the dual-indication bimatoprost, which was approved by the US Food and Drug Administration to reduce elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension but also is used to grow darker, thicker, and longer eyelashes. Clinicians who prescribe bimatoprost for this cosmetic indication should be mindful of the potential for PAP and discuss it with patients.
The prescribing information (PI) for bimatoprost (Latisse; Allergan) does not list PAP as an adverse reaction observed in the 4-month multicenter, double-blind, randomized, vehicle-controlled study of bimatoprost (as Latisse) in 278 adults.2 The PI does list “periorbital and lid changes associated with periorbital fat atrophy and skin tightness resulting in deepening of eyelid sulcus and eyelid ptosis” as an adverse reaction in postmarketing experience. However, according to the PI, the frequency of these adverse reactions cannot be established, as the reporting of such incidents was voluntary and the size of the treated population was uncertain.2
Prostaglandins can cause periorbitopathy by several mechanisms; one speculated cause is that this group of medications might provoke smooth muscle contraction. Prostaglandin medications also have an affinity for fat cells1; atrophy of fat cells can lead to enophthalmos and deepening upper eyelid sulcus. In an observational study of 105 participants who were using a prostaglandin in 1 eye for longer than 1 month (the other eye was used as a control), the overall frequency of prostaglandin-associated periorbitopathy was 93.3% in the bimatoprost group, 41.4% in the latanoprost group, and 70% in the travoprost group, while the frequency of deepening of the upper eyelid sulcus was 80% in the bimatoprost group, 15.7% in the latanoprost group, and 45% in the travoprost group.3 These changes may not be as striking when a patient is using a prostaglandin ophthalmic medication in both eyes and may not be noticed even by the patient. It is prudent for the clinician to take a baseline photograph of the patient when these medications are prescribed to observe for early signs of periorbitopathy. These adverse effects may not be reversible when the medication is discontinued4 and have been observed as early as 4 to 6 weeks after the start of treatment.5
Our patient was counseled that his constellation of PAP findings potentially could be partially reversed over months or even a year or longer if the offending agent was discontinued. However, he was cautioned that cessation of latanoprost first needed to be discussed with his ophthalmologist, who would determine if there was a suitable alternative to a prostaglandin analog for him. The patient’s only concern was the aesthetic appearance of the left periorbital area. A hyaluronic acid filler or fat grafting can be considered for correction of orbital sulcus hollowing; however, we could not locate any long-term studies in which such corrective treatments were applied for PAP. Our patient continues to use latanoprost with no change in the frequency of use. There have been no further changes or progression in the physical appearance of the left eye or periorbital area. The patient has not undergone any corrective treatments.
To the Editor:
A 42-year man presented with hollowing of the upper eyelid and skin discoloration of the left periorbital area of 10 years’ duration. He was a professional mixed martial arts fighter with a history of 2 surgeries for retinal detachment of the left eye 13 years prior to the current presentation. The patient also has macular scarring in the left eye. He denied a history of facial fracture, reconstructive surgery, or other medical conditions. His visual acuity was unknown; however, he did not require corrective glasses. He used 3 prescription ophthalmic eye drops—dorzolamide hydrochloride plus timolol maleate, 10 mL; brimonidine tartrate ophthalmic solution 0.15%, 5 mL; and latanoprost ophthalmic solution 0.005%, 125 μg/2.5 mL—in the left eye to lower intraocular pressure, as therapy for glaucoma. If left untreated, glaucoma can lead to vision loss.
Physical examination revealed periorbital hyperpigmentation on the left side; hypertrichosis and eyelash trichomegaly compared to the right side; and a deep left upper orbital sulcus compared to the right side (Figure). The patient was alert and oriented to person, place, and time. Extraocular movement was intact bilaterally, and his pupillary reflex was symmetric. No tenderness was noted over the affected area on palpation; no subcutaneous masses or lesions were observed or palpated. There was no ocular discharge, the conjunctiva was pink, and the sclera was white bilaterally.

The differential diagnosis included professional trauma-induced orbital changes, nevus of Ota (oculomucodermal melanocytosis), prostaglandin-associated periorbitopathy (PAP), and melasma. Although the patient sustained an injury that caused retinal detachment, he never experienced an orbital bone fracture; additionally, a fracture would not explain the skin discoloration or longer eyelashes. Periorbital nevus of Ota most commonly manifests as a unilateral scleral and brown-bluish skin discoloration but does not cause hollowing of the orbital sulcus or affect the length and thickness of eyelashes. Melasma—bilateral skin hyperpigmentation that most commonly affects women—can be induced by oral contraceptives, antibiotics, heat, sun exposure, and pregnancy. It does not affect the color of the iris or the depth of the scleral sulcus, and it does not increase the length and thickness of eyelashes. Based on the clinical presentation and a review of the eye drops used, he was diagnosed with PAP due to prolonged use of latanoprost ophthalmic solution. The patient was referred to an ophthalmologist for consideration of a switch to a different class of medication.
Of the 3 eye drops used by this patient, latanoprost, a prostaglandin analog, decreases intraocular pressure and is known to cause PAP. This condition comprises a constellation of changes, including upper eyelid ptosis, deepening of the upper eyelid sulcus, involution of dermatochalasis, periorbital fat atrophy, mild enophthalmos (sunken eye), inferior scleral show, increased prominence of eyelid vessels, and tight eyelids.1 Latanoprost most often produces these findings, but all prostaglandin ophthalmic medications can, including the dual-indication bimatoprost, which was approved by the US Food and Drug Administration to reduce elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension but also is used to grow darker, thicker, and longer eyelashes. Clinicians who prescribe bimatoprost for this cosmetic indication should be mindful of the potential for PAP and discuss it with patients.
The prescribing information (PI) for bimatoprost (Latisse; Allergan) does not list PAP as an adverse reaction observed in the 4-month multicenter, double-blind, randomized, vehicle-controlled study of bimatoprost (as Latisse) in 278 adults.2 The PI does list “periorbital and lid changes associated with periorbital fat atrophy and skin tightness resulting in deepening of eyelid sulcus and eyelid ptosis” as an adverse reaction in postmarketing experience. However, according to the PI, the frequency of these adverse reactions cannot be established, as the reporting of such incidents was voluntary and the size of the treated population was uncertain.2
Prostaglandins can cause periorbitopathy by several mechanisms; one speculated cause is that this group of medications might provoke smooth muscle contraction. Prostaglandin medications also have an affinity for fat cells1; atrophy of fat cells can lead to enophthalmos and deepening upper eyelid sulcus. In an observational study of 105 participants who were using a prostaglandin in 1 eye for longer than 1 month (the other eye was used as a control), the overall frequency of prostaglandin-associated periorbitopathy was 93.3% in the bimatoprost group, 41.4% in the latanoprost group, and 70% in the travoprost group, while the frequency of deepening of the upper eyelid sulcus was 80% in the bimatoprost group, 15.7% in the latanoprost group, and 45% in the travoprost group.3 These changes may not be as striking when a patient is using a prostaglandin ophthalmic medication in both eyes and may not be noticed even by the patient. It is prudent for the clinician to take a baseline photograph of the patient when these medications are prescribed to observe for early signs of periorbitopathy. These adverse effects may not be reversible when the medication is discontinued4 and have been observed as early as 4 to 6 weeks after the start of treatment.5
Our patient was counseled that his constellation of PAP findings potentially could be partially reversed over months or even a year or longer if the offending agent was discontinued. However, he was cautioned that cessation of latanoprost first needed to be discussed with his ophthalmologist, who would determine if there was a suitable alternative to a prostaglandin analog for him. The patient’s only concern was the aesthetic appearance of the left periorbital area. A hyaluronic acid filler or fat grafting can be considered for correction of orbital sulcus hollowing; however, we could not locate any long-term studies in which such corrective treatments were applied for PAP. Our patient continues to use latanoprost with no change in the frequency of use. There have been no further changes or progression in the physical appearance of the left eye or periorbital area. The patient has not undergone any corrective treatments.
- Berke SJ. PAP: new concerns for prostaglandin use. Rev Ophthalmol. 2012;19:70.
- Latisse (bimatoprost ophthalmic solution 0.03%). Package insert. Allergan; 2021. Accessed April 11, 2024. https://www.rxabbvie.com/pdf/latisse_pi.pdf
- Kucukevcilioglu M, Bayer A, Uysal Y, et al. Prostaglandin associated periorbitopathy in patients using bimatoprost, latanoprost and travoprost. Clin Exp Ophthalmol. 2014;42:126-131. doi:10.1111/ceo.12163
- Filippopoulos T, Paula JS, Torun N, et al. Periorbital changes associated with topical bimatoprost. Ophthalmic Plast Reconstr Surg. 2008;24:302-307. doi:10.1097/IOP.0b013e31817d81df
- Peplinski LS, Smith KA. Deepening of lid sulcus from topical bimatoprost therapy. Optom Vis Sci. 2004;81:574-577. doi:10.1097/01.opx.0000141791.16683.4a
- Berke SJ. PAP: new concerns for prostaglandin use. Rev Ophthalmol. 2012;19:70.
- Latisse (bimatoprost ophthalmic solution 0.03%). Package insert. Allergan; 2021. Accessed April 11, 2024. https://www.rxabbvie.com/pdf/latisse_pi.pdf
- Kucukevcilioglu M, Bayer A, Uysal Y, et al. Prostaglandin associated periorbitopathy in patients using bimatoprost, latanoprost and travoprost. Clin Exp Ophthalmol. 2014;42:126-131. doi:10.1111/ceo.12163
- Filippopoulos T, Paula JS, Torun N, et al. Periorbital changes associated with topical bimatoprost. Ophthalmic Plast Reconstr Surg. 2008;24:302-307. doi:10.1097/IOP.0b013e31817d81df
- Peplinski LS, Smith KA. Deepening of lid sulcus from topical bimatoprost therapy. Optom Vis Sci. 2004;81:574-577. doi:10.1097/01.opx.0000141791.16683.4a
PRACTICE POINTS
- Ask patients to provide photographs taken prior to noticed changes to assess progression if they are new to your practice.
- Take photographs of patients in good light against a solid-colored background to have a baseline. It may be helpful to update patient images annually.
- Discuss with patients the aesthetic changes that may occur with the use of prescription medications. Provide pamphlets with images to educate them on what to expect.
Late-Stage Incidence Rates Support CRC Screening From Age 45
, a cross-sectional study of stage-stratified CRC found.
It is well known that CRC is becoming more prevalent generally in the under 50-year population, but stage-related analyses have not been done.
Staging analysis in this age group is important, however, as an increasing burden of advance-staged disease would provide further evidence for earlier screening initiation, wrote Eric M. Montminy, MD, a gastroenterologist at John H. Stroger Hospital of County Cook, Chicago, Illinois, and colleagues in JAMA Network Open.
The United States Preventive Services Task Force (USPSTF) has recommended that average-risk screening begin at 45 years of age, as do the American Gastroenterological Association and other GI societies, although the American College of Physicians last year published clinical guidance recommending 50 years as the age to start screening for CRC for patients with average risk.
“Patients aged 46-49 may become confused on which guideline to follow, similar to confusion occurring with prior breast cancer screening changes,” Dr. Montminy said in an interview. “We wanted to demonstrate incidence rates with stage stratification to help clarify the incidence trends in this age group. Stage stratification is a key because it provides insight into the relationship between time and cancer incidence, ie, is screening finding early cancer or not?”
A 2020 study in JAMA Network Open demonstrated a 46.1% increase in CRC incidence rates (IRs) in persons aged 49-50 years. This steep increase is consistent with the presence of a large preexisting and undetected case burden.
“Our results demonstrate that adults aged 46-49 years, who are between now-conflicting guidelines on whether to start screening at age 45 or 50 years, have an increasing burden of more advanced-stage CRC and thus may be at an increased risk if screening is not initiated at age 45 years,” Dr. Montminy’s group wrote.
Using incidence data per 100,000 population from the National Cancer Institute’s Surveillance, Epidemiology, and End Results registry, the investigators observed the following IRs for early-onset CRC in the age group of 46-49 years:
- Distant adenocarcinoma IRs increased faster than other stages: annual percentage change (APC), 2.2 (95% CI, 1.8-2.6).
- Regional IRs also significantly increased: APC, 1.3 (95% CI, 0.8-1.7).
- Absolute regional IRs of CRC in the age bracket of 46-49 years are similar to total pancreatic cancer IRs in all ages and all stages combined (13.2 of 100,000) over similar years. When distant IRs for CRC are included with regional IRs, those for IRs for CRC are double those for pancreatic cancer of all stages combined.
- The only decrease was seen in localized IRs: APC, -0.6 (95% CI, -1 to -0.2).
“My best advice for clinicians is to provide the facts from the data to patients so they can make an informed health decision,” Dr. Montminy said. “This includes taking an appropriate personal and family history and having the patient factor this aspect into their decision on when and how they want to perform colon cancer screening.”
His institution adheres to the USPSTF recommendation of initiation of CRC screening at age 45 years.
Findings From 2000 to 2020
During 2000-2020 period, 26,887 CRCs were diagnosed in adults aged 46-49 years (54.5% in men).
As of 2020, the localized adenocarcinoma IR decreased to 7.7 of 100,000, but regional adenocarcinoma IR increased to 13.4 of 100,000 and distant adenocarcinoma IR increased to 9.0 of 100,000.
Regional adenocarcinoma IR remained the highest of all stages in 2000-2020. From 2014 to 2020, distant IRs became similar to localized IRs, except in 2017 when distant IRs were significantly higher than localized.
Why the CRC Uptick?
“It remains an enigma at this time as to why we’re seeing this shift,” Dr. Montminy said, noting that etiologies from the colonic microbiome to cellphones have been postulated. “To date, no theory has substantially provided causality. But whatever the source is, it is affecting Western countries in unison with data demonstrating a birth cohort effect as well,” he added. “We additionally know, based on the current epidemiologic data, that current screening practices are failing, and a unified discussion must occur in order to prevent young patients from developing advanced colon cancer.”
Offering his perspective on the findings, Joshua Meyer, MD, vice chair of translational research in the Department of Radiation Oncology at Fox Chase Cancer Center in Philadelphia, said the findings reinforce the practice of offering screening to average-risk individuals starting at age 45 years, the threshold at his institution. “There are previously published data demonstrating an increase in advanced stage at the time of screening initiation, and these data support that,” said Dr. Meyer, who was not involved in the present analysis.
More research needs to be done, he continued, not just on optimal age but also on the effect of multiple other factors impacting risk. “These may include family history and genetic risk as well as the role of blood- and stool-based screening assays in an integrated strategy to screen for colorectal cancer.”
There are multiple screening tests, and while colonoscopy, the gold standard, is very safe, it is not completely without risks, Dr. Meyer added. “And the question of the appropriate allocation of limited societal resources continues to be discussed on a broader level and largely explains the difference between the two guidelines.”
This study received no specific funding. Co-author Jordan J. Karlitz, MD, reported personal fees from GRAIL (senior medical director) and an equity position from Gastro Girl/GI On Demand outside f the submitted work. Dr. Meyer disclosed no conflicts of interest relevant to his comments.
, a cross-sectional study of stage-stratified CRC found.
It is well known that CRC is becoming more prevalent generally in the under 50-year population, but stage-related analyses have not been done.
Staging analysis in this age group is important, however, as an increasing burden of advance-staged disease would provide further evidence for earlier screening initiation, wrote Eric M. Montminy, MD, a gastroenterologist at John H. Stroger Hospital of County Cook, Chicago, Illinois, and colleagues in JAMA Network Open.
The United States Preventive Services Task Force (USPSTF) has recommended that average-risk screening begin at 45 years of age, as do the American Gastroenterological Association and other GI societies, although the American College of Physicians last year published clinical guidance recommending 50 years as the age to start screening for CRC for patients with average risk.
“Patients aged 46-49 may become confused on which guideline to follow, similar to confusion occurring with prior breast cancer screening changes,” Dr. Montminy said in an interview. “We wanted to demonstrate incidence rates with stage stratification to help clarify the incidence trends in this age group. Stage stratification is a key because it provides insight into the relationship between time and cancer incidence, ie, is screening finding early cancer or not?”
A 2020 study in JAMA Network Open demonstrated a 46.1% increase in CRC incidence rates (IRs) in persons aged 49-50 years. This steep increase is consistent with the presence of a large preexisting and undetected case burden.
“Our results demonstrate that adults aged 46-49 years, who are between now-conflicting guidelines on whether to start screening at age 45 or 50 years, have an increasing burden of more advanced-stage CRC and thus may be at an increased risk if screening is not initiated at age 45 years,” Dr. Montminy’s group wrote.
Using incidence data per 100,000 population from the National Cancer Institute’s Surveillance, Epidemiology, and End Results registry, the investigators observed the following IRs for early-onset CRC in the age group of 46-49 years:
- Distant adenocarcinoma IRs increased faster than other stages: annual percentage change (APC), 2.2 (95% CI, 1.8-2.6).
- Regional IRs also significantly increased: APC, 1.3 (95% CI, 0.8-1.7).
- Absolute regional IRs of CRC in the age bracket of 46-49 years are similar to total pancreatic cancer IRs in all ages and all stages combined (13.2 of 100,000) over similar years. When distant IRs for CRC are included with regional IRs, those for IRs for CRC are double those for pancreatic cancer of all stages combined.
- The only decrease was seen in localized IRs: APC, -0.6 (95% CI, -1 to -0.2).
“My best advice for clinicians is to provide the facts from the data to patients so they can make an informed health decision,” Dr. Montminy said. “This includes taking an appropriate personal and family history and having the patient factor this aspect into their decision on when and how they want to perform colon cancer screening.”
His institution adheres to the USPSTF recommendation of initiation of CRC screening at age 45 years.
Findings From 2000 to 2020
During 2000-2020 period, 26,887 CRCs were diagnosed in adults aged 46-49 years (54.5% in men).
As of 2020, the localized adenocarcinoma IR decreased to 7.7 of 100,000, but regional adenocarcinoma IR increased to 13.4 of 100,000 and distant adenocarcinoma IR increased to 9.0 of 100,000.
Regional adenocarcinoma IR remained the highest of all stages in 2000-2020. From 2014 to 2020, distant IRs became similar to localized IRs, except in 2017 when distant IRs were significantly higher than localized.
Why the CRC Uptick?
“It remains an enigma at this time as to why we’re seeing this shift,” Dr. Montminy said, noting that etiologies from the colonic microbiome to cellphones have been postulated. “To date, no theory has substantially provided causality. But whatever the source is, it is affecting Western countries in unison with data demonstrating a birth cohort effect as well,” he added. “We additionally know, based on the current epidemiologic data, that current screening practices are failing, and a unified discussion must occur in order to prevent young patients from developing advanced colon cancer.”
Offering his perspective on the findings, Joshua Meyer, MD, vice chair of translational research in the Department of Radiation Oncology at Fox Chase Cancer Center in Philadelphia, said the findings reinforce the practice of offering screening to average-risk individuals starting at age 45 years, the threshold at his institution. “There are previously published data demonstrating an increase in advanced stage at the time of screening initiation, and these data support that,” said Dr. Meyer, who was not involved in the present analysis.
More research needs to be done, he continued, not just on optimal age but also on the effect of multiple other factors impacting risk. “These may include family history and genetic risk as well as the role of blood- and stool-based screening assays in an integrated strategy to screen for colorectal cancer.”
There are multiple screening tests, and while colonoscopy, the gold standard, is very safe, it is not completely without risks, Dr. Meyer added. “And the question of the appropriate allocation of limited societal resources continues to be discussed on a broader level and largely explains the difference between the two guidelines.”
This study received no specific funding. Co-author Jordan J. Karlitz, MD, reported personal fees from GRAIL (senior medical director) and an equity position from Gastro Girl/GI On Demand outside f the submitted work. Dr. Meyer disclosed no conflicts of interest relevant to his comments.
, a cross-sectional study of stage-stratified CRC found.
It is well known that CRC is becoming more prevalent generally in the under 50-year population, but stage-related analyses have not been done.
Staging analysis in this age group is important, however, as an increasing burden of advance-staged disease would provide further evidence for earlier screening initiation, wrote Eric M. Montminy, MD, a gastroenterologist at John H. Stroger Hospital of County Cook, Chicago, Illinois, and colleagues in JAMA Network Open.
The United States Preventive Services Task Force (USPSTF) has recommended that average-risk screening begin at 45 years of age, as do the American Gastroenterological Association and other GI societies, although the American College of Physicians last year published clinical guidance recommending 50 years as the age to start screening for CRC for patients with average risk.
“Patients aged 46-49 may become confused on which guideline to follow, similar to confusion occurring with prior breast cancer screening changes,” Dr. Montminy said in an interview. “We wanted to demonstrate incidence rates with stage stratification to help clarify the incidence trends in this age group. Stage stratification is a key because it provides insight into the relationship between time and cancer incidence, ie, is screening finding early cancer or not?”
A 2020 study in JAMA Network Open demonstrated a 46.1% increase in CRC incidence rates (IRs) in persons aged 49-50 years. This steep increase is consistent with the presence of a large preexisting and undetected case burden.
“Our results demonstrate that adults aged 46-49 years, who are between now-conflicting guidelines on whether to start screening at age 45 or 50 years, have an increasing burden of more advanced-stage CRC and thus may be at an increased risk if screening is not initiated at age 45 years,” Dr. Montminy’s group wrote.
Using incidence data per 100,000 population from the National Cancer Institute’s Surveillance, Epidemiology, and End Results registry, the investigators observed the following IRs for early-onset CRC in the age group of 46-49 years:
- Distant adenocarcinoma IRs increased faster than other stages: annual percentage change (APC), 2.2 (95% CI, 1.8-2.6).
- Regional IRs also significantly increased: APC, 1.3 (95% CI, 0.8-1.7).
- Absolute regional IRs of CRC in the age bracket of 46-49 years are similar to total pancreatic cancer IRs in all ages and all stages combined (13.2 of 100,000) over similar years. When distant IRs for CRC are included with regional IRs, those for IRs for CRC are double those for pancreatic cancer of all stages combined.
- The only decrease was seen in localized IRs: APC, -0.6 (95% CI, -1 to -0.2).
“My best advice for clinicians is to provide the facts from the data to patients so they can make an informed health decision,” Dr. Montminy said. “This includes taking an appropriate personal and family history and having the patient factor this aspect into their decision on when and how they want to perform colon cancer screening.”
His institution adheres to the USPSTF recommendation of initiation of CRC screening at age 45 years.
Findings From 2000 to 2020
During 2000-2020 period, 26,887 CRCs were diagnosed in adults aged 46-49 years (54.5% in men).
As of 2020, the localized adenocarcinoma IR decreased to 7.7 of 100,000, but regional adenocarcinoma IR increased to 13.4 of 100,000 and distant adenocarcinoma IR increased to 9.0 of 100,000.
Regional adenocarcinoma IR remained the highest of all stages in 2000-2020. From 2014 to 2020, distant IRs became similar to localized IRs, except in 2017 when distant IRs were significantly higher than localized.
Why the CRC Uptick?
“It remains an enigma at this time as to why we’re seeing this shift,” Dr. Montminy said, noting that etiologies from the colonic microbiome to cellphones have been postulated. “To date, no theory has substantially provided causality. But whatever the source is, it is affecting Western countries in unison with data demonstrating a birth cohort effect as well,” he added. “We additionally know, based on the current epidemiologic data, that current screening practices are failing, and a unified discussion must occur in order to prevent young patients from developing advanced colon cancer.”
Offering his perspective on the findings, Joshua Meyer, MD, vice chair of translational research in the Department of Radiation Oncology at Fox Chase Cancer Center in Philadelphia, said the findings reinforce the practice of offering screening to average-risk individuals starting at age 45 years, the threshold at his institution. “There are previously published data demonstrating an increase in advanced stage at the time of screening initiation, and these data support that,” said Dr. Meyer, who was not involved in the present analysis.
More research needs to be done, he continued, not just on optimal age but also on the effect of multiple other factors impacting risk. “These may include family history and genetic risk as well as the role of blood- and stool-based screening assays in an integrated strategy to screen for colorectal cancer.”
There are multiple screening tests, and while colonoscopy, the gold standard, is very safe, it is not completely without risks, Dr. Meyer added. “And the question of the appropriate allocation of limited societal resources continues to be discussed on a broader level and largely explains the difference between the two guidelines.”
This study received no specific funding. Co-author Jordan J. Karlitz, MD, reported personal fees from GRAIL (senior medical director) and an equity position from Gastro Girl/GI On Demand outside f the submitted work. Dr. Meyer disclosed no conflicts of interest relevant to his comments.
FROM JAMA NETWORK OPEN
Commentary: Diet and Lifestyle in Migraine, May 2024
Migraine and other headache types are common ailments, and there are many stereotypes and stigmas associated with these conditions. One of the prevailing beliefs about headaches and migraines is that they are linked with internalizing mental health conditions — anxiety and depression. These associations can affect pediatric migraine patients and their parents in complicated ways, potentially hindering adequate diagnosis and treatment. Results of a recent prospective study, published in the journal Headache, provided results that challenge the widespread belief that people who have migraines have a higher-than-average rate of internalizing mental health disorders. The authors provided a discussion and data to explain that their initial hypothesis of a relationship between migraine and mental health was disproven. The study included 123 participants age 8-18 years who had been previously diagnosed with migraine. The patients, who were seen in a pediatric neurology clinic, completed headache questionnaires and validated measures of anxiety and depressive symptoms. The final analysis showed no significant association between migraines or headaches with anxiety or depression.
Why does this matter? Stigma can prevent patients and parents from seeking care if parents feel that they will be judged as bad parents for contributing to their children's anxiety, depression, headaches, and migraines. In fact, beyond mental health stigma, children who have migraine can be blamed for having an unhealthy lifestyle.[1] While advice to get enough sleep, eat healthy, and stay active is worthwhile, there can be an implication that pediatric migraine patients are causing their migraines by living an unhealthy lifestyle.[1] Additionally, the implication that parents are not properly taking care of their children's health can inhibit an accurate symptom history. Releasing pediatric migraine patients and their parents from myths about migraines and headaches can be a beneficial component of doctor-patient communication regarding migraine care.
It is possible that dietary adjustments or supplements could help improve migraine frequency and severity. Maintaining a healthy diet is a frequent recommendation for people who have headaches, but it can be frustrating for patients to receive general recommendations to follow a healthy lifestyle. Specific direction regarding which foods to avoid and which foods to add to a diet can be helpful for patients as they try to navigate the challenge of adopting migraine-friendly lifestyle changes.
Eicosapentaenoic acid (EPA) is one of the omega-3 fatty acids. A recent study, with results published in Brain, Behavior, and Immunity, examined the effects of EPA on migraines. The 12-week randomized, double-blind, placebo-controlled trial included 70 participants who had been diagnosed with episodic migraine. Participants were randomly assigned to either EPA (2 g fish oil with 1.8 g of EPA/day) or placebo (2 g soybean oil/day). Migraine frequency and severity were assessed using standardized scales. According to the authors, the high-dose-EPA group had significantly reduced migraine frequency and severity, fewer number of days using acute treatment, reduced migraine-associated disability, improved anxiety and depression, and improved quality of life in comparison to the placebo group. The EPA group did not experience notable adverse events. To provide a sense of scale regarding dietary EPA, 3 oz of cooked wild salmon has 0.35 g of EPA, 3 oz of cooked shrimp has 0.2 g of EPA, and 3 oz of light canned tuna has 0.02 g of EPA.[2] Thus, it's important to note that the amount of EPA used in this study was higher than what would be expected of dietary EPA.
An observational prospective study published in Scientific Reports examined the effects of dietary phytochemical index (DPI) on migraine. DPI is defined as the proportion of daily energy intake derived from foods rich in phytochemicals. Consumption of phytochemical-rich foods has been associated with cardiovascular and metabolic diseases prevention in various populations. These foods include fruits, vegetables, whole grains, seeds, nuts, and legumes. The study included 265 adults age 20-50 who had a diagnosis of migraine. Participants were asked to fill out a questionnaire, which was used to evaluate their diet in the preceding year, and they were asked to complete a diary to track their migraine symptoms. The results showed an inverse relationship between DPI index and migraine frequency. Participants who had the highest DPI had the lowest migraine frequency.[3] While the authors found the results to be statistically significant, they did not point to a cause and effect. Migraine-associated symptoms such as nausea can have an effect on dietary choices, so patients who experience migraine symptoms may avoid certain foods before, during, or after a migraine episode. They also may consistently avoid foods that they have experienced as migraine triggers.
Diet and lifestyle can have an effect on migraine frequency, severity, and overall migraine-associated quality of life. Beyond general recommendations, however, it is not yet well established which foods or supplements could potentially help alleviate migraines. Advice to maintain a healthy lifestyle is definitely worthwhile for migraine patients, but it is important to avoid conveying blame or stigma when it comes to communication about the effect of lifestyle on migraine. This is especially important for pediatric migraine patients because the stigma extends beyond children to parents and could potentially interfere with clear communication and adequate care.
Additional References
1. Gelfand AA, Irwin SL. Lifestyle advice for pediatric migraine: Blaming the patient, or evidence based? Semin Neurol. 2020;40:277-285. doi: 10.1055/s-0040-1708868 Source
2. National Institutes of Health. Office of Dietary Supplements. Omega-3 fatty acids. Updated February 15, 2023. Source
3. Hamedi-Shahraki S, Jowshan M-R, Zolghadrpour M-A, et al. Dietary phytochemical index is favorably associated with oxidative stress status and cardiovascular risk factors in adults with obesity. Sci Rep. 2023;13:7035. doi: 10.1038/s41598-023-34064-4 Source
Migraine and other headache types are common ailments, and there are many stereotypes and stigmas associated with these conditions. One of the prevailing beliefs about headaches and migraines is that they are linked with internalizing mental health conditions — anxiety and depression. These associations can affect pediatric migraine patients and their parents in complicated ways, potentially hindering adequate diagnosis and treatment. Results of a recent prospective study, published in the journal Headache, provided results that challenge the widespread belief that people who have migraines have a higher-than-average rate of internalizing mental health disorders. The authors provided a discussion and data to explain that their initial hypothesis of a relationship between migraine and mental health was disproven. The study included 123 participants age 8-18 years who had been previously diagnosed with migraine. The patients, who were seen in a pediatric neurology clinic, completed headache questionnaires and validated measures of anxiety and depressive symptoms. The final analysis showed no significant association between migraines or headaches with anxiety or depression.
Why does this matter? Stigma can prevent patients and parents from seeking care if parents feel that they will be judged as bad parents for contributing to their children's anxiety, depression, headaches, and migraines. In fact, beyond mental health stigma, children who have migraine can be blamed for having an unhealthy lifestyle.[1] While advice to get enough sleep, eat healthy, and stay active is worthwhile, there can be an implication that pediatric migraine patients are causing their migraines by living an unhealthy lifestyle.[1] Additionally, the implication that parents are not properly taking care of their children's health can inhibit an accurate symptom history. Releasing pediatric migraine patients and their parents from myths about migraines and headaches can be a beneficial component of doctor-patient communication regarding migraine care.
It is possible that dietary adjustments or supplements could help improve migraine frequency and severity. Maintaining a healthy diet is a frequent recommendation for people who have headaches, but it can be frustrating for patients to receive general recommendations to follow a healthy lifestyle. Specific direction regarding which foods to avoid and which foods to add to a diet can be helpful for patients as they try to navigate the challenge of adopting migraine-friendly lifestyle changes.
Eicosapentaenoic acid (EPA) is one of the omega-3 fatty acids. A recent study, with results published in Brain, Behavior, and Immunity, examined the effects of EPA on migraines. The 12-week randomized, double-blind, placebo-controlled trial included 70 participants who had been diagnosed with episodic migraine. Participants were randomly assigned to either EPA (2 g fish oil with 1.8 g of EPA/day) or placebo (2 g soybean oil/day). Migraine frequency and severity were assessed using standardized scales. According to the authors, the high-dose-EPA group had significantly reduced migraine frequency and severity, fewer number of days using acute treatment, reduced migraine-associated disability, improved anxiety and depression, and improved quality of life in comparison to the placebo group. The EPA group did not experience notable adverse events. To provide a sense of scale regarding dietary EPA, 3 oz of cooked wild salmon has 0.35 g of EPA, 3 oz of cooked shrimp has 0.2 g of EPA, and 3 oz of light canned tuna has 0.02 g of EPA.[2] Thus, it's important to note that the amount of EPA used in this study was higher than what would be expected of dietary EPA.
An observational prospective study published in Scientific Reports examined the effects of dietary phytochemical index (DPI) on migraine. DPI is defined as the proportion of daily energy intake derived from foods rich in phytochemicals. Consumption of phytochemical-rich foods has been associated with cardiovascular and metabolic diseases prevention in various populations. These foods include fruits, vegetables, whole grains, seeds, nuts, and legumes. The study included 265 adults age 20-50 who had a diagnosis of migraine. Participants were asked to fill out a questionnaire, which was used to evaluate their diet in the preceding year, and they were asked to complete a diary to track their migraine symptoms. The results showed an inverse relationship between DPI index and migraine frequency. Participants who had the highest DPI had the lowest migraine frequency.[3] While the authors found the results to be statistically significant, they did not point to a cause and effect. Migraine-associated symptoms such as nausea can have an effect on dietary choices, so patients who experience migraine symptoms may avoid certain foods before, during, or after a migraine episode. They also may consistently avoid foods that they have experienced as migraine triggers.
Diet and lifestyle can have an effect on migraine frequency, severity, and overall migraine-associated quality of life. Beyond general recommendations, however, it is not yet well established which foods or supplements could potentially help alleviate migraines. Advice to maintain a healthy lifestyle is definitely worthwhile for migraine patients, but it is important to avoid conveying blame or stigma when it comes to communication about the effect of lifestyle on migraine. This is especially important for pediatric migraine patients because the stigma extends beyond children to parents and could potentially interfere with clear communication and adequate care.
Additional References
1. Gelfand AA, Irwin SL. Lifestyle advice for pediatric migraine: Blaming the patient, or evidence based? Semin Neurol. 2020;40:277-285. doi: 10.1055/s-0040-1708868 Source
2. National Institutes of Health. Office of Dietary Supplements. Omega-3 fatty acids. Updated February 15, 2023. Source
3. Hamedi-Shahraki S, Jowshan M-R, Zolghadrpour M-A, et al. Dietary phytochemical index is favorably associated with oxidative stress status and cardiovascular risk factors in adults with obesity. Sci Rep. 2023;13:7035. doi: 10.1038/s41598-023-34064-4 Source
Migraine and other headache types are common ailments, and there are many stereotypes and stigmas associated with these conditions. One of the prevailing beliefs about headaches and migraines is that they are linked with internalizing mental health conditions — anxiety and depression. These associations can affect pediatric migraine patients and their parents in complicated ways, potentially hindering adequate diagnosis and treatment. Results of a recent prospective study, published in the journal Headache, provided results that challenge the widespread belief that people who have migraines have a higher-than-average rate of internalizing mental health disorders. The authors provided a discussion and data to explain that their initial hypothesis of a relationship between migraine and mental health was disproven. The study included 123 participants age 8-18 years who had been previously diagnosed with migraine. The patients, who were seen in a pediatric neurology clinic, completed headache questionnaires and validated measures of anxiety and depressive symptoms. The final analysis showed no significant association between migraines or headaches with anxiety or depression.
Why does this matter? Stigma can prevent patients and parents from seeking care if parents feel that they will be judged as bad parents for contributing to their children's anxiety, depression, headaches, and migraines. In fact, beyond mental health stigma, children who have migraine can be blamed for having an unhealthy lifestyle.[1] While advice to get enough sleep, eat healthy, and stay active is worthwhile, there can be an implication that pediatric migraine patients are causing their migraines by living an unhealthy lifestyle.[1] Additionally, the implication that parents are not properly taking care of their children's health can inhibit an accurate symptom history. Releasing pediatric migraine patients and their parents from myths about migraines and headaches can be a beneficial component of doctor-patient communication regarding migraine care.
It is possible that dietary adjustments or supplements could help improve migraine frequency and severity. Maintaining a healthy diet is a frequent recommendation for people who have headaches, but it can be frustrating for patients to receive general recommendations to follow a healthy lifestyle. Specific direction regarding which foods to avoid and which foods to add to a diet can be helpful for patients as they try to navigate the challenge of adopting migraine-friendly lifestyle changes.
Eicosapentaenoic acid (EPA) is one of the omega-3 fatty acids. A recent study, with results published in Brain, Behavior, and Immunity, examined the effects of EPA on migraines. The 12-week randomized, double-blind, placebo-controlled trial included 70 participants who had been diagnosed with episodic migraine. Participants were randomly assigned to either EPA (2 g fish oil with 1.8 g of EPA/day) or placebo (2 g soybean oil/day). Migraine frequency and severity were assessed using standardized scales. According to the authors, the high-dose-EPA group had significantly reduced migraine frequency and severity, fewer number of days using acute treatment, reduced migraine-associated disability, improved anxiety and depression, and improved quality of life in comparison to the placebo group. The EPA group did not experience notable adverse events. To provide a sense of scale regarding dietary EPA, 3 oz of cooked wild salmon has 0.35 g of EPA, 3 oz of cooked shrimp has 0.2 g of EPA, and 3 oz of light canned tuna has 0.02 g of EPA.[2] Thus, it's important to note that the amount of EPA used in this study was higher than what would be expected of dietary EPA.
An observational prospective study published in Scientific Reports examined the effects of dietary phytochemical index (DPI) on migraine. DPI is defined as the proportion of daily energy intake derived from foods rich in phytochemicals. Consumption of phytochemical-rich foods has been associated with cardiovascular and metabolic diseases prevention in various populations. These foods include fruits, vegetables, whole grains, seeds, nuts, and legumes. The study included 265 adults age 20-50 who had a diagnosis of migraine. Participants were asked to fill out a questionnaire, which was used to evaluate their diet in the preceding year, and they were asked to complete a diary to track their migraine symptoms. The results showed an inverse relationship between DPI index and migraine frequency. Participants who had the highest DPI had the lowest migraine frequency.[3] While the authors found the results to be statistically significant, they did not point to a cause and effect. Migraine-associated symptoms such as nausea can have an effect on dietary choices, so patients who experience migraine symptoms may avoid certain foods before, during, or after a migraine episode. They also may consistently avoid foods that they have experienced as migraine triggers.
Diet and lifestyle can have an effect on migraine frequency, severity, and overall migraine-associated quality of life. Beyond general recommendations, however, it is not yet well established which foods or supplements could potentially help alleviate migraines. Advice to maintain a healthy lifestyle is definitely worthwhile for migraine patients, but it is important to avoid conveying blame or stigma when it comes to communication about the effect of lifestyle on migraine. This is especially important for pediatric migraine patients because the stigma extends beyond children to parents and could potentially interfere with clear communication and adequate care.
Additional References
1. Gelfand AA, Irwin SL. Lifestyle advice for pediatric migraine: Blaming the patient, or evidence based? Semin Neurol. 2020;40:277-285. doi: 10.1055/s-0040-1708868 Source
2. National Institutes of Health. Office of Dietary Supplements. Omega-3 fatty acids. Updated February 15, 2023. Source
3. Hamedi-Shahraki S, Jowshan M-R, Zolghadrpour M-A, et al. Dietary phytochemical index is favorably associated with oxidative stress status and cardiovascular risk factors in adults with obesity. Sci Rep. 2023;13:7035. doi: 10.1038/s41598-023-34064-4 Source
Could Bedside Training Help End the US Neurologist Shortage?
DENVER — , a new report suggested.
Bedside Rounding Alliance for Internal Medicine and Neurology Residents (BRAINs) moves training from the lecture hall to the bedside, offering instruction on obtaining a focused neurologic history and performing a focused neurologic physical exam for common neurologic symptoms.
Almost 100% of trainees surveyed gave the program a favorable rating, citing patient exposure and bedside training from neurology educators as keys to its success.
As internal medicine providers are often “the first to lay eyes” on patients with a neurology complaint, it’s important they “have a basic level of comfort” in addressing patients’ common questions and concerns, study author Prashanth Rajarajan, MD, PhD, a resident in the Department of Neurology at Brigham and Women’s Hospital, Boston, told this news organization.
The findings were presented at the 2024 annual meeting of the American Academy of Neurology.
Addressing ‘Neurophobia’
Neurology is often viewed by medical trainees as the most difficult subspecialty, Dr. Rajarajan said. Many have what he calls “neurophobia,” which he defines as “a discomfort with assessing and treating neurologic complaints.”
A survey at his institution showed 62% of internal medicine residents lacked the confidence to diagnose and treat neurologic diseases, he reported.
BRAINs is a structured neurology trainee-led, inpatient bedside teaching session for internal medicine residents, medical students, and others that aims to increase trainees’ confidence in assessing patients with common neurologic symptoms.
The program includes a biweekly 45-minute session. Most of the session is spent at the bedside and involves demonstrations and practice of a focused neurologic history and physical exam.
Participants receive feedback from educators, typically neurology residents or fellows in epilepsy, stroke, or some other neurology subspecialty. It also includes a short discussion on pertinent diagnostics, management, and other topics.
Surveys evaluating the program and teaching skill development were completed by 59 residents and 15 neurology educators who participated in BRAINs between 2022 and 2024.
Over 90% of trainees (54) agreed BRAINs sessions met the program’s objective (5 were neutral); 49 agreed it increased confidence in taking a neuro history (9 were neutral and 1 disagreed); 56 felt it boosted their confidence in doing a neuro exam (3 were neutral); and 56 said BRAINs is more effective than traditional lecture-based didactics (3 were neutral).
All the residents rated the material covered as appropriate for their level of training; 88% considered the 45-minute session length appropriate; and 98% had a favorable impression of the program as a whole.
When asked to identify the most helpful aspect of the program, 82% cited more patient exposure and 81% more bedside teaching.
All educators reported that the sessions were an effective way to practice near-peer teaching skills. Most (87%) felt the experience was more effective at accomplishing learning objectives than preparing and giving traditional didactic lectures, and 80% agreed it also gave them an opportunity to get to know their medical colleagues.
Use It or Lose It
Dr. Rajarajan noted that the program doesn’t require significant planning or extra staff, is not resource-intensive, and can be adapted to different services such as emergency departments and other learner populations.
But time will tell if the newfound confidence of those taking the program actually lasts.
“You have to keep using it,” he said. “You use it or lose it when comes to these skills.”
Commenting on the initiative, Denney Zimmerman, DO, Neurocritical Care Faculty, Blount Memorial Hospital, Maryville, Tennessee, and cochair of the AAN session featuring the study, called the program a good example of one way to counteract “neurophobia” and address the widespread neurologist shortage in the United States.
A 2019 AAN report showed that by 2025, almost every state in the United States will have a mismatch between the number of practicing neurologists and the demand from patients with neurologic conditions. The report offered several ways to address the shortage, including more neurology-focused training for internal medicine doctors during their residency.
“They’re usually on the front line, both in the hospital and in the clinics, and can help expedite patients who need to be seen by neurology sooner rather than later,” Dr. Zimmerman said.
Dr. Zimmerman noted that the study assessed how well participants perceived the program but not whether it improved their skills.
He pointed out that different groups may assess different diseases during their training session. “I think it’s important to ensure you’re hitting all the major topics.”
The study received funding from MGB Centers of Expertise Education Grant. Drs. Rajarajan and Zimmerman reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
DENVER — , a new report suggested.
Bedside Rounding Alliance for Internal Medicine and Neurology Residents (BRAINs) moves training from the lecture hall to the bedside, offering instruction on obtaining a focused neurologic history and performing a focused neurologic physical exam for common neurologic symptoms.
Almost 100% of trainees surveyed gave the program a favorable rating, citing patient exposure and bedside training from neurology educators as keys to its success.
As internal medicine providers are often “the first to lay eyes” on patients with a neurology complaint, it’s important they “have a basic level of comfort” in addressing patients’ common questions and concerns, study author Prashanth Rajarajan, MD, PhD, a resident in the Department of Neurology at Brigham and Women’s Hospital, Boston, told this news organization.
The findings were presented at the 2024 annual meeting of the American Academy of Neurology.
Addressing ‘Neurophobia’
Neurology is often viewed by medical trainees as the most difficult subspecialty, Dr. Rajarajan said. Many have what he calls “neurophobia,” which he defines as “a discomfort with assessing and treating neurologic complaints.”
A survey at his institution showed 62% of internal medicine residents lacked the confidence to diagnose and treat neurologic diseases, he reported.
BRAINs is a structured neurology trainee-led, inpatient bedside teaching session for internal medicine residents, medical students, and others that aims to increase trainees’ confidence in assessing patients with common neurologic symptoms.
The program includes a biweekly 45-minute session. Most of the session is spent at the bedside and involves demonstrations and practice of a focused neurologic history and physical exam.
Participants receive feedback from educators, typically neurology residents or fellows in epilepsy, stroke, or some other neurology subspecialty. It also includes a short discussion on pertinent diagnostics, management, and other topics.
Surveys evaluating the program and teaching skill development were completed by 59 residents and 15 neurology educators who participated in BRAINs between 2022 and 2024.
Over 90% of trainees (54) agreed BRAINs sessions met the program’s objective (5 were neutral); 49 agreed it increased confidence in taking a neuro history (9 were neutral and 1 disagreed); 56 felt it boosted their confidence in doing a neuro exam (3 were neutral); and 56 said BRAINs is more effective than traditional lecture-based didactics (3 were neutral).
All the residents rated the material covered as appropriate for their level of training; 88% considered the 45-minute session length appropriate; and 98% had a favorable impression of the program as a whole.
When asked to identify the most helpful aspect of the program, 82% cited more patient exposure and 81% more bedside teaching.
All educators reported that the sessions were an effective way to practice near-peer teaching skills. Most (87%) felt the experience was more effective at accomplishing learning objectives than preparing and giving traditional didactic lectures, and 80% agreed it also gave them an opportunity to get to know their medical colleagues.
Use It or Lose It
Dr. Rajarajan noted that the program doesn’t require significant planning or extra staff, is not resource-intensive, and can be adapted to different services such as emergency departments and other learner populations.
But time will tell if the newfound confidence of those taking the program actually lasts.
“You have to keep using it,” he said. “You use it or lose it when comes to these skills.”
Commenting on the initiative, Denney Zimmerman, DO, Neurocritical Care Faculty, Blount Memorial Hospital, Maryville, Tennessee, and cochair of the AAN session featuring the study, called the program a good example of one way to counteract “neurophobia” and address the widespread neurologist shortage in the United States.
A 2019 AAN report showed that by 2025, almost every state in the United States will have a mismatch between the number of practicing neurologists and the demand from patients with neurologic conditions. The report offered several ways to address the shortage, including more neurology-focused training for internal medicine doctors during their residency.
“They’re usually on the front line, both in the hospital and in the clinics, and can help expedite patients who need to be seen by neurology sooner rather than later,” Dr. Zimmerman said.
Dr. Zimmerman noted that the study assessed how well participants perceived the program but not whether it improved their skills.
He pointed out that different groups may assess different diseases during their training session. “I think it’s important to ensure you’re hitting all the major topics.”
The study received funding from MGB Centers of Expertise Education Grant. Drs. Rajarajan and Zimmerman reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
DENVER — , a new report suggested.
Bedside Rounding Alliance for Internal Medicine and Neurology Residents (BRAINs) moves training from the lecture hall to the bedside, offering instruction on obtaining a focused neurologic history and performing a focused neurologic physical exam for common neurologic symptoms.
Almost 100% of trainees surveyed gave the program a favorable rating, citing patient exposure and bedside training from neurology educators as keys to its success.
As internal medicine providers are often “the first to lay eyes” on patients with a neurology complaint, it’s important they “have a basic level of comfort” in addressing patients’ common questions and concerns, study author Prashanth Rajarajan, MD, PhD, a resident in the Department of Neurology at Brigham and Women’s Hospital, Boston, told this news organization.
The findings were presented at the 2024 annual meeting of the American Academy of Neurology.
Addressing ‘Neurophobia’
Neurology is often viewed by medical trainees as the most difficult subspecialty, Dr. Rajarajan said. Many have what he calls “neurophobia,” which he defines as “a discomfort with assessing and treating neurologic complaints.”
A survey at his institution showed 62% of internal medicine residents lacked the confidence to diagnose and treat neurologic diseases, he reported.
BRAINs is a structured neurology trainee-led, inpatient bedside teaching session for internal medicine residents, medical students, and others that aims to increase trainees’ confidence in assessing patients with common neurologic symptoms.
The program includes a biweekly 45-minute session. Most of the session is spent at the bedside and involves demonstrations and practice of a focused neurologic history and physical exam.
Participants receive feedback from educators, typically neurology residents or fellows in epilepsy, stroke, or some other neurology subspecialty. It also includes a short discussion on pertinent diagnostics, management, and other topics.
Surveys evaluating the program and teaching skill development were completed by 59 residents and 15 neurology educators who participated in BRAINs between 2022 and 2024.
Over 90% of trainees (54) agreed BRAINs sessions met the program’s objective (5 were neutral); 49 agreed it increased confidence in taking a neuro history (9 were neutral and 1 disagreed); 56 felt it boosted their confidence in doing a neuro exam (3 were neutral); and 56 said BRAINs is more effective than traditional lecture-based didactics (3 were neutral).
All the residents rated the material covered as appropriate for their level of training; 88% considered the 45-minute session length appropriate; and 98% had a favorable impression of the program as a whole.
When asked to identify the most helpful aspect of the program, 82% cited more patient exposure and 81% more bedside teaching.
All educators reported that the sessions were an effective way to practice near-peer teaching skills. Most (87%) felt the experience was more effective at accomplishing learning objectives than preparing and giving traditional didactic lectures, and 80% agreed it also gave them an opportunity to get to know their medical colleagues.
Use It or Lose It
Dr. Rajarajan noted that the program doesn’t require significant planning or extra staff, is not resource-intensive, and can be adapted to different services such as emergency departments and other learner populations.
But time will tell if the newfound confidence of those taking the program actually lasts.
“You have to keep using it,” he said. “You use it or lose it when comes to these skills.”
Commenting on the initiative, Denney Zimmerman, DO, Neurocritical Care Faculty, Blount Memorial Hospital, Maryville, Tennessee, and cochair of the AAN session featuring the study, called the program a good example of one way to counteract “neurophobia” and address the widespread neurologist shortage in the United States.
A 2019 AAN report showed that by 2025, almost every state in the United States will have a mismatch between the number of practicing neurologists and the demand from patients with neurologic conditions. The report offered several ways to address the shortage, including more neurology-focused training for internal medicine doctors during their residency.
“They’re usually on the front line, both in the hospital and in the clinics, and can help expedite patients who need to be seen by neurology sooner rather than later,” Dr. Zimmerman said.
Dr. Zimmerman noted that the study assessed how well participants perceived the program but not whether it improved their skills.
He pointed out that different groups may assess different diseases during their training session. “I think it’s important to ensure you’re hitting all the major topics.”
The study received funding from MGB Centers of Expertise Education Grant. Drs. Rajarajan and Zimmerman reported no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM AAN 2024
Risk for COVID-19 Infection in Patients With Vitiligo
To the Editor:
Vitiligo is a depigmentation disorder that results from the loss of melanocytes in the epidermis.1 The most widely accepted pathophysiology for melanocyte destruction in vitiligo is an autoimmune process involving dysregulated cytokine production and autoreactive T-cell activation.1 Individuals with cutaneous autoinflammatory conditions currently are vital patient populations warranting research, as their susceptibility to COVID-19 infection may differ from the general population. We previously found a small increased risk for COVID-19 infection in patients with psoriasis,2 which suggests that other dermatologic conditions also may impact COVID-19 risk. The risk for COVID-19 infection in patients with vitiligo remains largely unknown. In this retrospective cohort study, we investigated the risk for COVID-19 infection in patients with vitiligo compared with those without vitiligo utilizing claims data from the COVID-19 Research Database (https://covid19researchdatabase.org/).
Claims were evaluated for patients aged 3 years and older with a vitiligo diagnosis (International Classification of Diseases, Tenth Revision [ICD-10] code L80) that was made between January 1, 2016, and January 1, 2020. Individuals without a vitiligo diagnosis during the same period were placed (4:1 ratio) in the control group and were matched with study group patients for age and sex. All comorbidity variables and vitiligo diagnoses were extracted from ICD-10 codes that were given prior to a diagnosis of COVID-19. We then constructed multivariable logistic regression models adjusting for measured confounders to evaluate if vitiligo was associated with higher risk for COVID-19 infection after January 1, 2020.
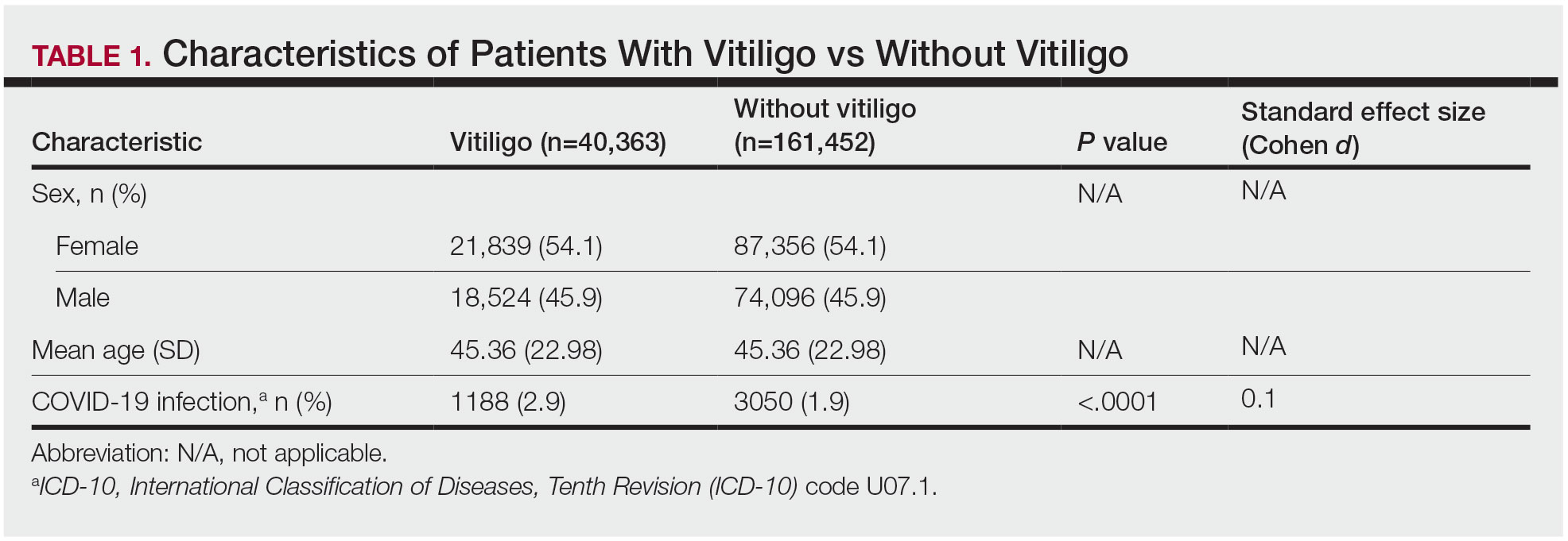
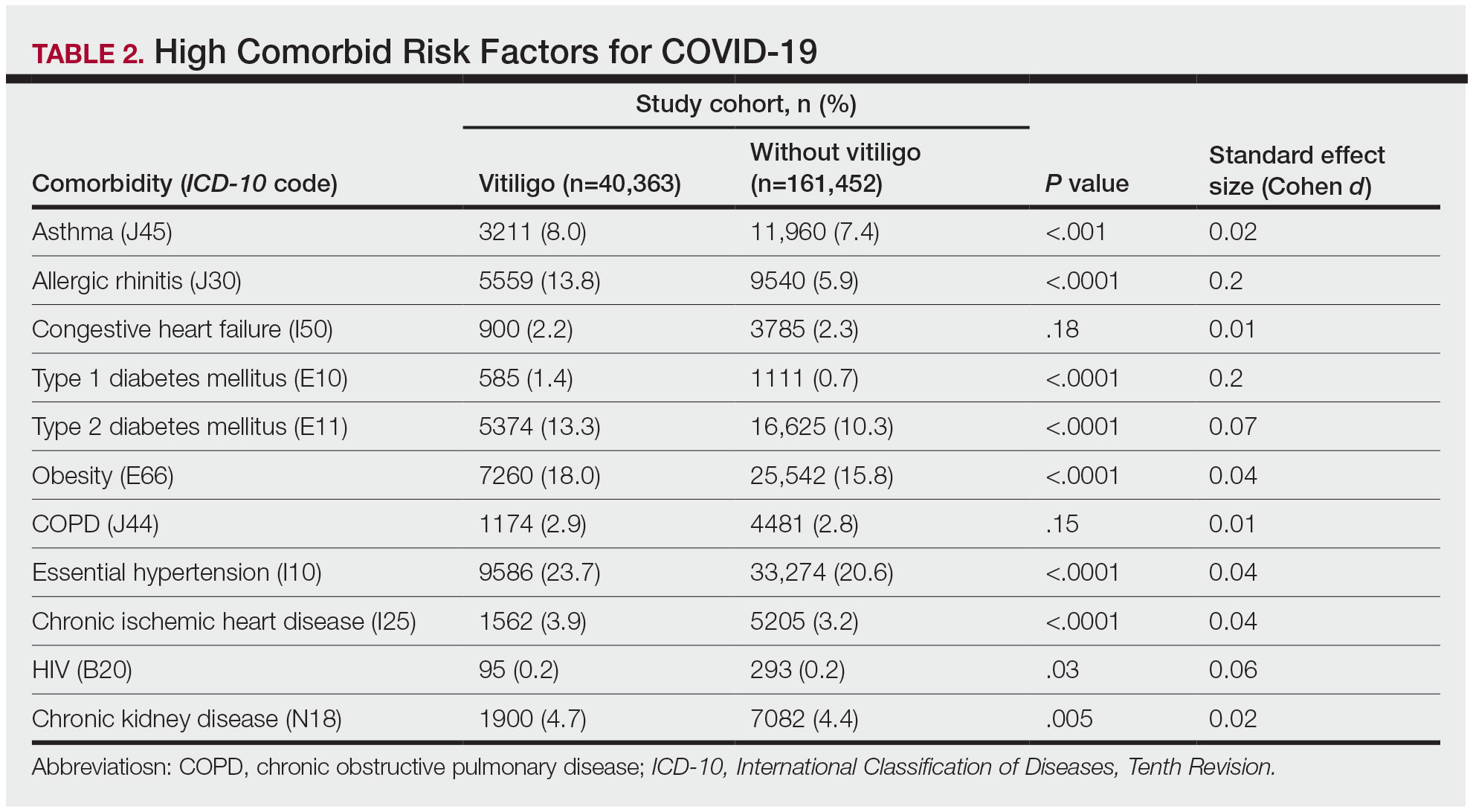
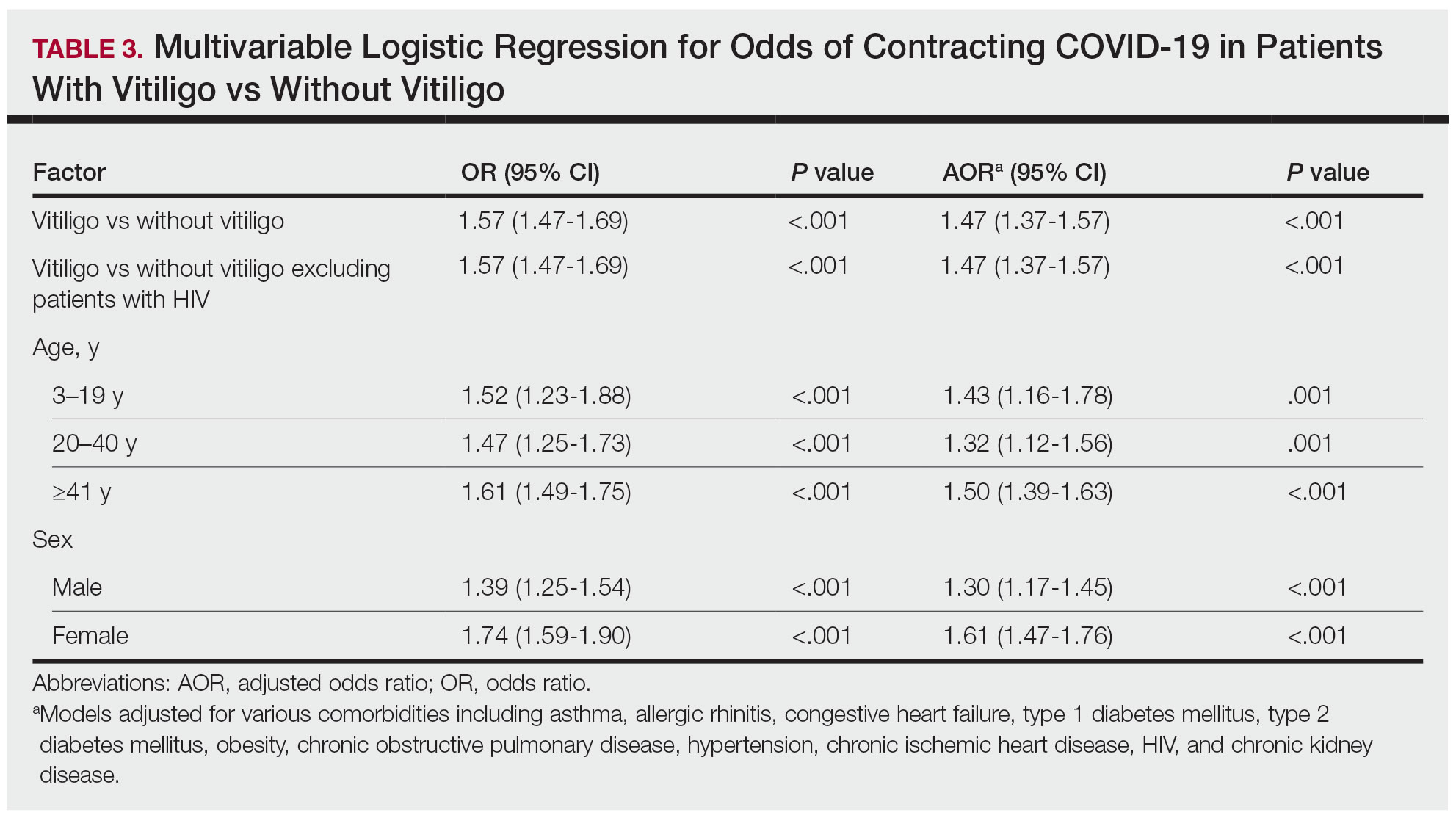
The vitiligo and nonvitiligo cohorts included 40,363 and 161,452 patients, respectively (Table 1). Logistic regression analysis with adjustment for confounding variables, including high comorbid risk factors (Table 2) revealed that patients with a diagnosis of vitiligo had significantly increased odds of COVID-19 infection compared with patients without vitiligo (adjusted odds ratio [AOR], 1.47; 95% CI, 1.37-1.57; P<.001)(Table 3). Additionally, subgroup logistic analyses for sex, age, and exclusion of patients who were HIV positive revealed that females with vitiligo had higher odds of contracting COVID-19 than males with vitiligo (Table 3).
Our results showed that patients with vitiligo had a higher relative risk for contracting COVID-19 than individuals without vitiligo. It has been reported that the prevalence of COVID-19 is higher among patients with autoimmune diseases compared to the general population.3 Additionally, a handful of vitiligo patients are managed with immunosuppressive agents that may further weaken their immune response.1 Moreover, survey results from dermatologists managing vitiligo patients revealed that physicians were fairly comfortable prescribing immunosuppressants and encouraging in-office phototherapy during the COVID-19 pandemic.4 As a result, more patients may have been attending in-office visits for their phototherapy, which may have increased their risk for COVID-19. Although these factors play a role in COVID-19 infection rates, the underlying immune dysregulation in vitiligo in relation to COVID-19 remains unknown and should be further explored.
Our findings are limited by the use of ICD-10 codes, the inability to control for all potential confounding variables, the lack of data regarding the stage of vitiligo, and the absence of data for undiagnosed COVID-19 infections. In addition, patients with vitiligo may be more likely to seek care, potentially increasing their rates of COVID-19 testing. The inability to identify the stage of vitiligo during enrollment in the database may have altered our results, as individuals with active disease have increased levels of IFN-γ. Increased secretion of IFN-γ also potentially helps in the clearance of COVID-19 infection.1 Future studies should investigate this relationship via planned COVID-19 testing, identification of vitiligo stage, and controlling for other associated comorbidities.
- Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi:10.1016/j.det.2016.11.014
- Wu JJ, Liu J, Thatiparthi A, et al. The risk of COVID-19 in patients with psoriasis—a retrospective cohort study [published online September 20, 2022]. J Am Acad Dermatol. doi:10.1016/j.jaad.2022.07.040
- Zhong J, Shen G, Yang H, et al. COVID-19 in patients with rheumatic disease in Hubei province, China: a multicentre retrospective observational study. Lancet Rheumatol. 2020;2:E557-E564. doi:10.1016/S2665-9913(20)30227-7
- Chatterjee M, Das A. Management of vitiligo amidst the COVID-19 pandemic: a survey and resulting consensus. Indian J Dermatol. 2021;66:479-483. doi:10.4103/ijd.ijd_859_20
To the Editor:
Vitiligo is a depigmentation disorder that results from the loss of melanocytes in the epidermis.1 The most widely accepted pathophysiology for melanocyte destruction in vitiligo is an autoimmune process involving dysregulated cytokine production and autoreactive T-cell activation.1 Individuals with cutaneous autoinflammatory conditions currently are vital patient populations warranting research, as their susceptibility to COVID-19 infection may differ from the general population. We previously found a small increased risk for COVID-19 infection in patients with psoriasis,2 which suggests that other dermatologic conditions also may impact COVID-19 risk. The risk for COVID-19 infection in patients with vitiligo remains largely unknown. In this retrospective cohort study, we investigated the risk for COVID-19 infection in patients with vitiligo compared with those without vitiligo utilizing claims data from the COVID-19 Research Database (https://covid19researchdatabase.org/).
Claims were evaluated for patients aged 3 years and older with a vitiligo diagnosis (International Classification of Diseases, Tenth Revision [ICD-10] code L80) that was made between January 1, 2016, and January 1, 2020. Individuals without a vitiligo diagnosis during the same period were placed (4:1 ratio) in the control group and were matched with study group patients for age and sex. All comorbidity variables and vitiligo diagnoses were extracted from ICD-10 codes that were given prior to a diagnosis of COVID-19. We then constructed multivariable logistic regression models adjusting for measured confounders to evaluate if vitiligo was associated with higher risk for COVID-19 infection after January 1, 2020.
The vitiligo and nonvitiligo cohorts included 40,363 and 161,452 patients, respectively (Table 1). Logistic regression analysis with adjustment for confounding variables, including high comorbid risk factors (Table 2) revealed that patients with a diagnosis of vitiligo had significantly increased odds of COVID-19 infection compared with patients without vitiligo (adjusted odds ratio [AOR], 1.47; 95% CI, 1.37-1.57; P<.001)(Table 3). Additionally, subgroup logistic analyses for sex, age, and exclusion of patients who were HIV positive revealed that females with vitiligo had higher odds of contracting COVID-19 than males with vitiligo (Table 3).

Our results showed that patients with vitiligo had a higher relative risk for contracting COVID-19 than individuals without vitiligo. It has been reported that the prevalence of COVID-19 is higher among patients with autoimmune diseases compared to the general population.3 Additionally, a handful of vitiligo patients are managed with immunosuppressive agents that may further weaken their immune response.1 Moreover, survey results from dermatologists managing vitiligo patients revealed that physicians were fairly comfortable prescribing immunosuppressants and encouraging in-office phototherapy during the COVID-19 pandemic.4 As a result, more patients may have been attending in-office visits for their phototherapy, which may have increased their risk for COVID-19. Although these factors play a role in COVID-19 infection rates, the underlying immune dysregulation in vitiligo in relation to COVID-19 remains unknown and should be further explored.

Our findings are limited by the use of ICD-10 codes, the inability to control for all potential confounding variables, the lack of data regarding the stage of vitiligo, and the absence of data for undiagnosed COVID-19 infections. In addition, patients with vitiligo may be more likely to seek care, potentially increasing their rates of COVID-19 testing. The inability to identify the stage of vitiligo during enrollment in the database may have altered our results, as individuals with active disease have increased levels of IFN-γ. Increased secretion of IFN-γ also potentially helps in the clearance of COVID-19 infection.1 Future studies should investigate this relationship via planned COVID-19 testing, identification of vitiligo stage, and controlling for other associated comorbidities.

To the Editor:
Vitiligo is a depigmentation disorder that results from the loss of melanocytes in the epidermis.1 The most widely accepted pathophysiology for melanocyte destruction in vitiligo is an autoimmune process involving dysregulated cytokine production and autoreactive T-cell activation.1 Individuals with cutaneous autoinflammatory conditions currently are vital patient populations warranting research, as their susceptibility to COVID-19 infection may differ from the general population. We previously found a small increased risk for COVID-19 infection in patients with psoriasis,2 which suggests that other dermatologic conditions also may impact COVID-19 risk. The risk for COVID-19 infection in patients with vitiligo remains largely unknown. In this retrospective cohort study, we investigated the risk for COVID-19 infection in patients with vitiligo compared with those without vitiligo utilizing claims data from the COVID-19 Research Database (https://covid19researchdatabase.org/).
Claims were evaluated for patients aged 3 years and older with a vitiligo diagnosis (International Classification of Diseases, Tenth Revision [ICD-10] code L80) that was made between January 1, 2016, and January 1, 2020. Individuals without a vitiligo diagnosis during the same period were placed (4:1 ratio) in the control group and were matched with study group patients for age and sex. All comorbidity variables and vitiligo diagnoses were extracted from ICD-10 codes that were given prior to a diagnosis of COVID-19. We then constructed multivariable logistic regression models adjusting for measured confounders to evaluate if vitiligo was associated with higher risk for COVID-19 infection after January 1, 2020.
The vitiligo and nonvitiligo cohorts included 40,363 and 161,452 patients, respectively (Table 1). Logistic regression analysis with adjustment for confounding variables, including high comorbid risk factors (Table 2) revealed that patients with a diagnosis of vitiligo had significantly increased odds of COVID-19 infection compared with patients without vitiligo (adjusted odds ratio [AOR], 1.47; 95% CI, 1.37-1.57; P<.001)(Table 3). Additionally, subgroup logistic analyses for sex, age, and exclusion of patients who were HIV positive revealed that females with vitiligo had higher odds of contracting COVID-19 than males with vitiligo (Table 3).

Our results showed that patients with vitiligo had a higher relative risk for contracting COVID-19 than individuals without vitiligo. It has been reported that the prevalence of COVID-19 is higher among patients with autoimmune diseases compared to the general population.3 Additionally, a handful of vitiligo patients are managed with immunosuppressive agents that may further weaken their immune response.1 Moreover, survey results from dermatologists managing vitiligo patients revealed that physicians were fairly comfortable prescribing immunosuppressants and encouraging in-office phototherapy during the COVID-19 pandemic.4 As a result, more patients may have been attending in-office visits for their phototherapy, which may have increased their risk for COVID-19. Although these factors play a role in COVID-19 infection rates, the underlying immune dysregulation in vitiligo in relation to COVID-19 remains unknown and should be further explored.

Our findings are limited by the use of ICD-10 codes, the inability to control for all potential confounding variables, the lack of data regarding the stage of vitiligo, and the absence of data for undiagnosed COVID-19 infections. In addition, patients with vitiligo may be more likely to seek care, potentially increasing their rates of COVID-19 testing. The inability to identify the stage of vitiligo during enrollment in the database may have altered our results, as individuals with active disease have increased levels of IFN-γ. Increased secretion of IFN-γ also potentially helps in the clearance of COVID-19 infection.1 Future studies should investigate this relationship via planned COVID-19 testing, identification of vitiligo stage, and controlling for other associated comorbidities.

- Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi:10.1016/j.det.2016.11.014
- Wu JJ, Liu J, Thatiparthi A, et al. The risk of COVID-19 in patients with psoriasis—a retrospective cohort study [published online September 20, 2022]. J Am Acad Dermatol. doi:10.1016/j.jaad.2022.07.040
- Zhong J, Shen G, Yang H, et al. COVID-19 in patients with rheumatic disease in Hubei province, China: a multicentre retrospective observational study. Lancet Rheumatol. 2020;2:E557-E564. doi:10.1016/S2665-9913(20)30227-7
- Chatterjee M, Das A. Management of vitiligo amidst the COVID-19 pandemic: a survey and resulting consensus. Indian J Dermatol. 2021;66:479-483. doi:10.4103/ijd.ijd_859_20
- Rashighi M, Harris JE. Vitiligo pathogenesis and emerging treatments. Dermatol Clin. 2017;35:257-265. doi:10.1016/j.det.2016.11.014
- Wu JJ, Liu J, Thatiparthi A, et al. The risk of COVID-19 in patients with psoriasis—a retrospective cohort study [published online September 20, 2022]. J Am Acad Dermatol. doi:10.1016/j.jaad.2022.07.040
- Zhong J, Shen G, Yang H, et al. COVID-19 in patients with rheumatic disease in Hubei province, China: a multicentre retrospective observational study. Lancet Rheumatol. 2020;2:E557-E564. doi:10.1016/S2665-9913(20)30227-7
- Chatterjee M, Das A. Management of vitiligo amidst the COVID-19 pandemic: a survey and resulting consensus. Indian J Dermatol. 2021;66:479-483. doi:10.4103/ijd.ijd_859_20
Practice Points
- The underlying autoimmune process in vitiligo can result in various changes to the immune system.
- A diagnosis of vitiligo may alter the body’s immune response to COVID-19 infection.
Few Cancer Survivors Meet ACS Nutrition, Exercise Guidelines
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
Erythrodermic Pityriasis Rubra Pilaris Following COVID-19 Vaccination
To the Editor:
A 32-year-old man presented to our clinic with acute-onset erythroderma associated with severe itching of 1 month’s duration. The patient developed the eruption after receiving the second dose of the Sinopharm BBIBP COVID-19 vaccine (BBIBP-CorV) 2 weeks prior to presentation. His medical history was unremarkable. There was no personal or family history of skin disease and no history of drug intake. Physical examination revealed
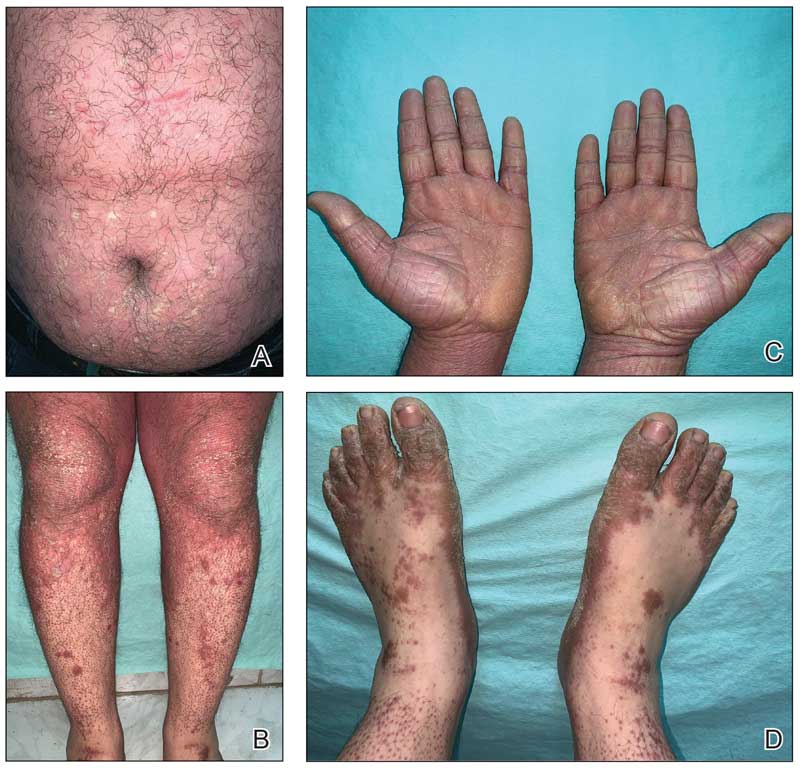
Histopathology of two 4-mm punch biopsies of the skin on the trunk and lower limb showed
Pityriasis rubra pilaris is a rare papulosquamous skin disease of unknown etiology with several theories including genetic factors, aberrant metabolism of vitamin A, infection, drug reaction, autoimmune disease, and malignancy.1 Clinically, there are 6 types of PRP: type I (classical adult), type II (atypical adult), type III (classical juvenile), type IV (circumscribed juvenile), type V (atypical juvenile), and type VI (HIV associated). Classic features include orange-red keratotic follicular papules that coalesce into plaques with characteristic islands of sparing.1
Pityriasis rubra pilaris is a rare sequela following administration of certain vaccines, including diphtheria, pertussis, and tetanus; measles-mumps-rubella; and polio vaccines.2,3 Among the various skin reactions that have been reported following COVID-19 vaccination, PRP has been reported in 19 patients: 7 (36.8%) after AstraZeneca vaccination, 3 (15.8%) after CoronaVac, 3 (15.8%) after Moderna, 5 (26.3%) after Pfizer-BioNTech,4 and 1 (5.3%) after Sinopharm.5 Our patient represents an additional case of a reaction after the Sinopharm vaccine. The condition developed after the first dose of vaccine in 11 patients, after the second dose in 6 patients, and after the third dose in 2 patients.
Other papulosquamous skin reactions have been reported after
Pityriasis rubra pilaris can be self-limited in some cases and may not require treatment. Topical therapies such as keratolytics, emollients, and vitamin D may be utilized, especially for localized disease. Systemic therapy may be needed for refractory cases, including retinoids or immunosuppressive medications such as methotrexate, which is considered a second-line treatment for refractory PRP (after retinoids) and was used in our case. Azathioprine and cyclosporine also may be used. Phototherapy may play a role in PRP treatment, but the response is variable.7
Pityriasis rubra pilaris should be added to the list of cutaneous adverse reactions that can occur following vaccination with the Sinopharm BBIBP-CorV vaccine. Dermatologists must be aware of the possibility of vaccine-induced PRP, especially in de novo cases.
- Wang D, Chong VC-L, Chong W-S, et al. A review on pityriasis rubra pilaris. Am J Clin Dermatol. 2018;19:377-390. doi:10.1007/s40257-017-0338-1
- Mohamed M, Belhadjali H, Hammedi F, et al. Pityriasis rubra pilaris occurring after vaccination with diphtheria-pertussis-tetanus and oral poliovirus vaccines [letter]. Indian J Dermatol Venereol Leprol. 2015;81:618-620. doi:10.4103/0378-6323.168326
- Naciri Bennani B, Cheikh Rouhou H, Waton J, et al. Pityriasis rubra pilaris after vaccination. Ann Dermatol Venereol. 2011;138:753-756. doi:10.1016/j.annder.2011.01.049
- Liu YA, Dai J, Nagarajan P, et al. Pityriasis rubra pilaris after Moderna COVID-19 vaccination: a case report and literature review. Am J Dermatopathol. 2023;45:185-188. doi:10.1097/DAD.0000000000002369.
- Samarasinghe KH, Janani T, Gunasekera CN. Pityriasis rubra pilaris like eruption following Sinopharm-SARS COVID-19 vaccine. Sri Lanka J Dermatol. 2021;22:99-100.
- Shakoei S, Kalantari Y, Nasimi M, et al. Cutaneous manifestations following COVID-19 vaccination: a report of 25 cases. Dermatol Ther. 2022;35:E15651. doi:10.1111/dth.15651
- Moretta G, De Luca EV, Di Stefani A. Management of refractory pityriasis rubra pilaris: challenges and solutions. Clin Cosmet Investig Dermatol. 2017;10:451-457. doi:10.2147/CCID.S124351.
To the Editor:
A 32-year-old man presented to our clinic with acute-onset erythroderma associated with severe itching of 1 month’s duration. The patient developed the eruption after receiving the second dose of the Sinopharm BBIBP COVID-19 vaccine (BBIBP-CorV) 2 weeks prior to presentation. His medical history was unremarkable. There was no personal or family history of skin disease and no history of drug intake. Physical examination revealed

Histopathology of two 4-mm punch biopsies of the skin on the trunk and lower limb showed

Pityriasis rubra pilaris is a rare papulosquamous skin disease of unknown etiology with several theories including genetic factors, aberrant metabolism of vitamin A, infection, drug reaction, autoimmune disease, and malignancy.1 Clinically, there are 6 types of PRP: type I (classical adult), type II (atypical adult), type III (classical juvenile), type IV (circumscribed juvenile), type V (atypical juvenile), and type VI (HIV associated). Classic features include orange-red keratotic follicular papules that coalesce into plaques with characteristic islands of sparing.1
Pityriasis rubra pilaris is a rare sequela following administration of certain vaccines, including diphtheria, pertussis, and tetanus; measles-mumps-rubella; and polio vaccines.2,3 Among the various skin reactions that have been reported following COVID-19 vaccination, PRP has been reported in 19 patients: 7 (36.8%) after AstraZeneca vaccination, 3 (15.8%) after CoronaVac, 3 (15.8%) after Moderna, 5 (26.3%) after Pfizer-BioNTech,4 and 1 (5.3%) after Sinopharm.5 Our patient represents an additional case of a reaction after the Sinopharm vaccine. The condition developed after the first dose of vaccine in 11 patients, after the second dose in 6 patients, and after the third dose in 2 patients.
Other papulosquamous skin reactions have been reported after
Pityriasis rubra pilaris can be self-limited in some cases and may not require treatment. Topical therapies such as keratolytics, emollients, and vitamin D may be utilized, especially for localized disease. Systemic therapy may be needed for refractory cases, including retinoids or immunosuppressive medications such as methotrexate, which is considered a second-line treatment for refractory PRP (after retinoids) and was used in our case. Azathioprine and cyclosporine also may be used. Phototherapy may play a role in PRP treatment, but the response is variable.7
Pityriasis rubra pilaris should be added to the list of cutaneous adverse reactions that can occur following vaccination with the Sinopharm BBIBP-CorV vaccine. Dermatologists must be aware of the possibility of vaccine-induced PRP, especially in de novo cases.
To the Editor:
A 32-year-old man presented to our clinic with acute-onset erythroderma associated with severe itching of 1 month’s duration. The patient developed the eruption after receiving the second dose of the Sinopharm BBIBP COVID-19 vaccine (BBIBP-CorV) 2 weeks prior to presentation. His medical history was unremarkable. There was no personal or family history of skin disease and no history of drug intake. Physical examination revealed

Histopathology of two 4-mm punch biopsies of the skin on the trunk and lower limb showed

Pityriasis rubra pilaris is a rare papulosquamous skin disease of unknown etiology with several theories including genetic factors, aberrant metabolism of vitamin A, infection, drug reaction, autoimmune disease, and malignancy.1 Clinically, there are 6 types of PRP: type I (classical adult), type II (atypical adult), type III (classical juvenile), type IV (circumscribed juvenile), type V (atypical juvenile), and type VI (HIV associated). Classic features include orange-red keratotic follicular papules that coalesce into plaques with characteristic islands of sparing.1
Pityriasis rubra pilaris is a rare sequela following administration of certain vaccines, including diphtheria, pertussis, and tetanus; measles-mumps-rubella; and polio vaccines.2,3 Among the various skin reactions that have been reported following COVID-19 vaccination, PRP has been reported in 19 patients: 7 (36.8%) after AstraZeneca vaccination, 3 (15.8%) after CoronaVac, 3 (15.8%) after Moderna, 5 (26.3%) after Pfizer-BioNTech,4 and 1 (5.3%) after Sinopharm.5 Our patient represents an additional case of a reaction after the Sinopharm vaccine. The condition developed after the first dose of vaccine in 11 patients, after the second dose in 6 patients, and after the third dose in 2 patients.
Other papulosquamous skin reactions have been reported after
Pityriasis rubra pilaris can be self-limited in some cases and may not require treatment. Topical therapies such as keratolytics, emollients, and vitamin D may be utilized, especially for localized disease. Systemic therapy may be needed for refractory cases, including retinoids or immunosuppressive medications such as methotrexate, which is considered a second-line treatment for refractory PRP (after retinoids) and was used in our case. Azathioprine and cyclosporine also may be used. Phototherapy may play a role in PRP treatment, but the response is variable.7
Pityriasis rubra pilaris should be added to the list of cutaneous adverse reactions that can occur following vaccination with the Sinopharm BBIBP-CorV vaccine. Dermatologists must be aware of the possibility of vaccine-induced PRP, especially in de novo cases.
- Wang D, Chong VC-L, Chong W-S, et al. A review on pityriasis rubra pilaris. Am J Clin Dermatol. 2018;19:377-390. doi:10.1007/s40257-017-0338-1
- Mohamed M, Belhadjali H, Hammedi F, et al. Pityriasis rubra pilaris occurring after vaccination with diphtheria-pertussis-tetanus and oral poliovirus vaccines [letter]. Indian J Dermatol Venereol Leprol. 2015;81:618-620. doi:10.4103/0378-6323.168326
- Naciri Bennani B, Cheikh Rouhou H, Waton J, et al. Pityriasis rubra pilaris after vaccination. Ann Dermatol Venereol. 2011;138:753-756. doi:10.1016/j.annder.2011.01.049
- Liu YA, Dai J, Nagarajan P, et al. Pityriasis rubra pilaris after Moderna COVID-19 vaccination: a case report and literature review. Am J Dermatopathol. 2023;45:185-188. doi:10.1097/DAD.0000000000002369.
- Samarasinghe KH, Janani T, Gunasekera CN. Pityriasis rubra pilaris like eruption following Sinopharm-SARS COVID-19 vaccine. Sri Lanka J Dermatol. 2021;22:99-100.
- Shakoei S, Kalantari Y, Nasimi M, et al. Cutaneous manifestations following COVID-19 vaccination: a report of 25 cases. Dermatol Ther. 2022;35:E15651. doi:10.1111/dth.15651
- Moretta G, De Luca EV, Di Stefani A. Management of refractory pityriasis rubra pilaris: challenges and solutions. Clin Cosmet Investig Dermatol. 2017;10:451-457. doi:10.2147/CCID.S124351.
- Wang D, Chong VC-L, Chong W-S, et al. A review on pityriasis rubra pilaris. Am J Clin Dermatol. 2018;19:377-390. doi:10.1007/s40257-017-0338-1
- Mohamed M, Belhadjali H, Hammedi F, et al. Pityriasis rubra pilaris occurring after vaccination with diphtheria-pertussis-tetanus and oral poliovirus vaccines [letter]. Indian J Dermatol Venereol Leprol. 2015;81:618-620. doi:10.4103/0378-6323.168326
- Naciri Bennani B, Cheikh Rouhou H, Waton J, et al. Pityriasis rubra pilaris after vaccination. Ann Dermatol Venereol. 2011;138:753-756. doi:10.1016/j.annder.2011.01.049
- Liu YA, Dai J, Nagarajan P, et al. Pityriasis rubra pilaris after Moderna COVID-19 vaccination: a case report and literature review. Am J Dermatopathol. 2023;45:185-188. doi:10.1097/DAD.0000000000002369.
- Samarasinghe KH, Janani T, Gunasekera CN. Pityriasis rubra pilaris like eruption following Sinopharm-SARS COVID-19 vaccine. Sri Lanka J Dermatol. 2021;22:99-100.
- Shakoei S, Kalantari Y, Nasimi M, et al. Cutaneous manifestations following COVID-19 vaccination: a report of 25 cases. Dermatol Ther. 2022;35:E15651. doi:10.1111/dth.15651
- Moretta G, De Luca EV, Di Stefani A. Management of refractory pityriasis rubra pilaris: challenges and solutions. Clin Cosmet Investig Dermatol. 2017;10:451-457. doi:10.2147/CCID.S124351.
Practice Points
- Dermatologists must be aware of the possibility of COVID-19 vaccine–induced pityriasis rubra pilaris (PRP), especially in de novo cases.
- Management of these cases usually follows similar standards for PRP cases.
New British Behçet’s Disease Guidelines Emphasize Multidisciplinary Management
LIVERPOOL, ENGLAND — The British Society for Rheumatology (BSR) and the British Association of Dermatologists (BAD) have joined forces for the first time to develop the first British guidelines for the management of people living with Behçet’s disease.
The guidelines will also be the first “living guidelines” produced by either society, which means they will be regularly revised and updated when new evidence emerges that warrants inclusion.
With more than 90 recommendations being made, the new guidelines promise to be the most comprehensive and most up-to-date yet for what is regarded as a rare disease. Robert Moots, MBBS, PhD, provided a “sneak peek” of the guidelines at the annual meeting of the British Society for Rheumatology.
Dr. Moots, professor of rheumatology at the University of Liverpool and a consultant rheumatologist for Liverpool University Hospitals NHS Foundation Trust in England, noted that while the European Alliance of Associations for Rheumatology has produced a guideline for Behçet’s disease, this was last updated in 2018 and is not specific for the population for patients that is seen in the United Kingdom.
The British recommendations will cover all possible manifestations of Behçet’s disease and give practical advice on how to manage everything from the most common presentations such as skin lesions, mouth ulcers, and genital ulcers, as well as the potentially more serious eye, neurological, and vascular involvement.
Importance of Raising Awareness
“Joint and musculoskeletal problems are actually one of the least complained of symptoms in people with Behçet’s, and they often can’t understand why a rheumatologist is seeing them,” Dr. Moot said. “But of course, people do get joint problems, they can get enthesitis and arthralgia.”
Dr. Moots has been leading one of the three National Health Service (NHS) Centres of Excellence for Behçet’s Syndrome in England for more than a decade and told this news organization that diagnosing patients could be challenging. It can take up to 10 years from the first symptoms appearing to getting a diagnosis, so part of the job of the NHS Centres of Excellence is to raise awareness among both the healthcare profession and the general public.
“It’s a condition that people learn about at medical school. Most doctors will have come across it, but because it was thought to be really rare in the UK, nobody perhaps really expects to see it,” Dr. Moot said.
“But we all have these patients,” he added. “In Liverpool, we’re commissioned to be looking after an anticipated 150 people with Behçet’s — we’ve got 700. With more awareness, there’s more diagnoses being made, and people are being looked after better.”
Patient Perspective
Tony Thornburn, OBE, chair of the patient advocacy group Behçet’s UK, agreed in a separate interview that raising awareness of the syndrome was key to improving its management.
“Patients have said that it is a bit like having arthritis, lupus, MS [multiple sclerosis], and Crohn’s [disease] all at once,” Mr. Thorburn said. “So what we need is a guideline to ensure that people know what they’re looking at.”
Mr. Thorburn added, “Guidelines are important for raising awareness but also providing the detailed information that clinicians and GPs [general practitioners] need to have to treat a patient when they come in with this multifaceted condition.”
Multifaceted Means Multidisciplinary Management
Because there can be so many different aspects to managing someone with Behçet’s disease, a multispecialty team that was convened to develop the guidelines agreed that multidisciplinary management should be an overarching theme.
“The guideline development group consisted of all the specialties that you would need for a complex multisystem disease like Behçet’s,” Dr. Moot said. He highlighted that working alongside the consultants in adult and pediatric rheumatology were specialists in dermatology, gastroenterology, neurology, ophthalmology, obstetrics and gynecology, and psychology.
“We’re actually looking at psychological interactions and their impact for the first time,” Dr. Moot said, noting that clinicians needed to “take it seriously, and ask about it.”
Management of Manifestations
One of the general principles of the guidelines is to assess the involvement of each organ system and target treatment accordingly.
“One of the problems is that the evidence base to tell us what to do is pretty low,” Dr. Moots acknowledged. There have been few good quality randomized trials, so “treatment tends to be eminence-based rather than evidence-based.”
The recommendation wording bears this in mind, stating whether a treatment should or should not be offered, or just considered if there is no strong evidence to back up its use.
With regard to musculoskeletal manifestations, the recommendations say that colchicine should be offered, perhaps as a first-line option, or an intraarticular steroid injection in the case of monoarthritis. An intramuscular depot steroid may also be appropriate to offer, and there was good evidence to offer azathioprine or, as an alternative in refractory cases, a tumor necrosis factor (TNF) inhibitor. Nonsteroidal anti-inflammatory drugs, methotrexate, apremilast, secukinumab, and referral to a physiotherapist could only be considered, however, based on weaker levels of evidence for their use.
To treat mucocutaneous disease, the guidelines advise offering topical steroids in the form of ointment for genital ulcers or mouthwash or ointment for oral ulcers. For skin lesions, it is recommended to offer colchicine, azathioprine, mycophenolate mofetil, or TNF inhibitor and to consider the use of apremilast, secukinumab, or dapsone.
Future Work and Revision
“One of the key things we would like to see developing is a national registry,” Dr. Moots said. This would include biobanking samples for future research and possible genomic and phenotyping studies.
More work needs to be done in conducting clinical trials in children and young people with Behçet’s disease, studies to find prognostic factors for neurological disease, and clinical trials of potential new drug approaches such as Janus kinase inhibitors. Importantly, an auditing process needs to be set up to see what effect, if any, the guidelines will actually have onpatient management.
“It’s taken 5 years to today” to develop the guidelines, Dr. Moot said. What form the process of updating them will take still has to be decided, he said in the interview. It is likely that the necessary literature searches will be performed every 6 months or so, but it will be a compromise between the ideal situation and having the staffing time to do it.
“It’s a big ask,” Dr. Moot acknowledged, adding that even if updates were only once a year, it would still be much faster than the 5- or 6-year cycle that it traditionally takes for most guidelines to be updated.
The BSR and BAD’s processes for developing guidelines are accredited by the National Institute for Health and Care Excellence in England. Dr. Moots is the chief investigator for the Secukinumab in Behçet’s trial, which is sponsored by the Liverpool University Hospitals NHS Foundation Trust via grant funding from Novartis.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — The British Society for Rheumatology (BSR) and the British Association of Dermatologists (BAD) have joined forces for the first time to develop the first British guidelines for the management of people living with Behçet’s disease.
The guidelines will also be the first “living guidelines” produced by either society, which means they will be regularly revised and updated when new evidence emerges that warrants inclusion.
With more than 90 recommendations being made, the new guidelines promise to be the most comprehensive and most up-to-date yet for what is regarded as a rare disease. Robert Moots, MBBS, PhD, provided a “sneak peek” of the guidelines at the annual meeting of the British Society for Rheumatology.
Dr. Moots, professor of rheumatology at the University of Liverpool and a consultant rheumatologist for Liverpool University Hospitals NHS Foundation Trust in England, noted that while the European Alliance of Associations for Rheumatology has produced a guideline for Behçet’s disease, this was last updated in 2018 and is not specific for the population for patients that is seen in the United Kingdom.
The British recommendations will cover all possible manifestations of Behçet’s disease and give practical advice on how to manage everything from the most common presentations such as skin lesions, mouth ulcers, and genital ulcers, as well as the potentially more serious eye, neurological, and vascular involvement.
Importance of Raising Awareness
“Joint and musculoskeletal problems are actually one of the least complained of symptoms in people with Behçet’s, and they often can’t understand why a rheumatologist is seeing them,” Dr. Moot said. “But of course, people do get joint problems, they can get enthesitis and arthralgia.”
Dr. Moots has been leading one of the three National Health Service (NHS) Centres of Excellence for Behçet’s Syndrome in England for more than a decade and told this news organization that diagnosing patients could be challenging. It can take up to 10 years from the first symptoms appearing to getting a diagnosis, so part of the job of the NHS Centres of Excellence is to raise awareness among both the healthcare profession and the general public.
“It’s a condition that people learn about at medical school. Most doctors will have come across it, but because it was thought to be really rare in the UK, nobody perhaps really expects to see it,” Dr. Moot said.
“But we all have these patients,” he added. “In Liverpool, we’re commissioned to be looking after an anticipated 150 people with Behçet’s — we’ve got 700. With more awareness, there’s more diagnoses being made, and people are being looked after better.”
Patient Perspective
Tony Thornburn, OBE, chair of the patient advocacy group Behçet’s UK, agreed in a separate interview that raising awareness of the syndrome was key to improving its management.
“Patients have said that it is a bit like having arthritis, lupus, MS [multiple sclerosis], and Crohn’s [disease] all at once,” Mr. Thorburn said. “So what we need is a guideline to ensure that people know what they’re looking at.”
Mr. Thorburn added, “Guidelines are important for raising awareness but also providing the detailed information that clinicians and GPs [general practitioners] need to have to treat a patient when they come in with this multifaceted condition.”
Multifaceted Means Multidisciplinary Management
Because there can be so many different aspects to managing someone with Behçet’s disease, a multispecialty team that was convened to develop the guidelines agreed that multidisciplinary management should be an overarching theme.
“The guideline development group consisted of all the specialties that you would need for a complex multisystem disease like Behçet’s,” Dr. Moot said. He highlighted that working alongside the consultants in adult and pediatric rheumatology were specialists in dermatology, gastroenterology, neurology, ophthalmology, obstetrics and gynecology, and psychology.
“We’re actually looking at psychological interactions and their impact for the first time,” Dr. Moot said, noting that clinicians needed to “take it seriously, and ask about it.”
Management of Manifestations
One of the general principles of the guidelines is to assess the involvement of each organ system and target treatment accordingly.
“One of the problems is that the evidence base to tell us what to do is pretty low,” Dr. Moots acknowledged. There have been few good quality randomized trials, so “treatment tends to be eminence-based rather than evidence-based.”
The recommendation wording bears this in mind, stating whether a treatment should or should not be offered, or just considered if there is no strong evidence to back up its use.
With regard to musculoskeletal manifestations, the recommendations say that colchicine should be offered, perhaps as a first-line option, or an intraarticular steroid injection in the case of monoarthritis. An intramuscular depot steroid may also be appropriate to offer, and there was good evidence to offer azathioprine or, as an alternative in refractory cases, a tumor necrosis factor (TNF) inhibitor. Nonsteroidal anti-inflammatory drugs, methotrexate, apremilast, secukinumab, and referral to a physiotherapist could only be considered, however, based on weaker levels of evidence for their use.
To treat mucocutaneous disease, the guidelines advise offering topical steroids in the form of ointment for genital ulcers or mouthwash or ointment for oral ulcers. For skin lesions, it is recommended to offer colchicine, azathioprine, mycophenolate mofetil, or TNF inhibitor and to consider the use of apremilast, secukinumab, or dapsone.
Future Work and Revision
“One of the key things we would like to see developing is a national registry,” Dr. Moots said. This would include biobanking samples for future research and possible genomic and phenotyping studies.
More work needs to be done in conducting clinical trials in children and young people with Behçet’s disease, studies to find prognostic factors for neurological disease, and clinical trials of potential new drug approaches such as Janus kinase inhibitors. Importantly, an auditing process needs to be set up to see what effect, if any, the guidelines will actually have onpatient management.
“It’s taken 5 years to today” to develop the guidelines, Dr. Moot said. What form the process of updating them will take still has to be decided, he said in the interview. It is likely that the necessary literature searches will be performed every 6 months or so, but it will be a compromise between the ideal situation and having the staffing time to do it.
“It’s a big ask,” Dr. Moot acknowledged, adding that even if updates were only once a year, it would still be much faster than the 5- or 6-year cycle that it traditionally takes for most guidelines to be updated.
The BSR and BAD’s processes for developing guidelines are accredited by the National Institute for Health and Care Excellence in England. Dr. Moots is the chief investigator for the Secukinumab in Behçet’s trial, which is sponsored by the Liverpool University Hospitals NHS Foundation Trust via grant funding from Novartis.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — The British Society for Rheumatology (BSR) and the British Association of Dermatologists (BAD) have joined forces for the first time to develop the first British guidelines for the management of people living with Behçet’s disease.
The guidelines will also be the first “living guidelines” produced by either society, which means they will be regularly revised and updated when new evidence emerges that warrants inclusion.
With more than 90 recommendations being made, the new guidelines promise to be the most comprehensive and most up-to-date yet for what is regarded as a rare disease. Robert Moots, MBBS, PhD, provided a “sneak peek” of the guidelines at the annual meeting of the British Society for Rheumatology.
Dr. Moots, professor of rheumatology at the University of Liverpool and a consultant rheumatologist for Liverpool University Hospitals NHS Foundation Trust in England, noted that while the European Alliance of Associations for Rheumatology has produced a guideline for Behçet’s disease, this was last updated in 2018 and is not specific for the population for patients that is seen in the United Kingdom.
The British recommendations will cover all possible manifestations of Behçet’s disease and give practical advice on how to manage everything from the most common presentations such as skin lesions, mouth ulcers, and genital ulcers, as well as the potentially more serious eye, neurological, and vascular involvement.
Importance of Raising Awareness
“Joint and musculoskeletal problems are actually one of the least complained of symptoms in people with Behçet’s, and they often can’t understand why a rheumatologist is seeing them,” Dr. Moot said. “But of course, people do get joint problems, they can get enthesitis and arthralgia.”
Dr. Moots has been leading one of the three National Health Service (NHS) Centres of Excellence for Behçet’s Syndrome in England for more than a decade and told this news organization that diagnosing patients could be challenging. It can take up to 10 years from the first symptoms appearing to getting a diagnosis, so part of the job of the NHS Centres of Excellence is to raise awareness among both the healthcare profession and the general public.
“It’s a condition that people learn about at medical school. Most doctors will have come across it, but because it was thought to be really rare in the UK, nobody perhaps really expects to see it,” Dr. Moot said.
“But we all have these patients,” he added. “In Liverpool, we’re commissioned to be looking after an anticipated 150 people with Behçet’s — we’ve got 700. With more awareness, there’s more diagnoses being made, and people are being looked after better.”
Patient Perspective
Tony Thornburn, OBE, chair of the patient advocacy group Behçet’s UK, agreed in a separate interview that raising awareness of the syndrome was key to improving its management.
“Patients have said that it is a bit like having arthritis, lupus, MS [multiple sclerosis], and Crohn’s [disease] all at once,” Mr. Thorburn said. “So what we need is a guideline to ensure that people know what they’re looking at.”
Mr. Thorburn added, “Guidelines are important for raising awareness but also providing the detailed information that clinicians and GPs [general practitioners] need to have to treat a patient when they come in with this multifaceted condition.”
Multifaceted Means Multidisciplinary Management
Because there can be so many different aspects to managing someone with Behçet’s disease, a multispecialty team that was convened to develop the guidelines agreed that multidisciplinary management should be an overarching theme.
“The guideline development group consisted of all the specialties that you would need for a complex multisystem disease like Behçet’s,” Dr. Moot said. He highlighted that working alongside the consultants in adult and pediatric rheumatology were specialists in dermatology, gastroenterology, neurology, ophthalmology, obstetrics and gynecology, and psychology.
“We’re actually looking at psychological interactions and their impact for the first time,” Dr. Moot said, noting that clinicians needed to “take it seriously, and ask about it.”
Management of Manifestations
One of the general principles of the guidelines is to assess the involvement of each organ system and target treatment accordingly.
“One of the problems is that the evidence base to tell us what to do is pretty low,” Dr. Moots acknowledged. There have been few good quality randomized trials, so “treatment tends to be eminence-based rather than evidence-based.”
The recommendation wording bears this in mind, stating whether a treatment should or should not be offered, or just considered if there is no strong evidence to back up its use.
With regard to musculoskeletal manifestations, the recommendations say that colchicine should be offered, perhaps as a first-line option, or an intraarticular steroid injection in the case of monoarthritis. An intramuscular depot steroid may also be appropriate to offer, and there was good evidence to offer azathioprine or, as an alternative in refractory cases, a tumor necrosis factor (TNF) inhibitor. Nonsteroidal anti-inflammatory drugs, methotrexate, apremilast, secukinumab, and referral to a physiotherapist could only be considered, however, based on weaker levels of evidence for their use.
To treat mucocutaneous disease, the guidelines advise offering topical steroids in the form of ointment for genital ulcers or mouthwash or ointment for oral ulcers. For skin lesions, it is recommended to offer colchicine, azathioprine, mycophenolate mofetil, or TNF inhibitor and to consider the use of apremilast, secukinumab, or dapsone.
Future Work and Revision
“One of the key things we would like to see developing is a national registry,” Dr. Moots said. This would include biobanking samples for future research and possible genomic and phenotyping studies.
More work needs to be done in conducting clinical trials in children and young people with Behçet’s disease, studies to find prognostic factors for neurological disease, and clinical trials of potential new drug approaches such as Janus kinase inhibitors. Importantly, an auditing process needs to be set up to see what effect, if any, the guidelines will actually have onpatient management.
“It’s taken 5 years to today” to develop the guidelines, Dr. Moot said. What form the process of updating them will take still has to be decided, he said in the interview. It is likely that the necessary literature searches will be performed every 6 months or so, but it will be a compromise between the ideal situation and having the staffing time to do it.
“It’s a big ask,” Dr. Moot acknowledged, adding that even if updates were only once a year, it would still be much faster than the 5- or 6-year cycle that it traditionally takes for most guidelines to be updated.
The BSR and BAD’s processes for developing guidelines are accredited by the National Institute for Health and Care Excellence in England. Dr. Moots is the chief investigator for the Secukinumab in Behçet’s trial, which is sponsored by the Liverpool University Hospitals NHS Foundation Trust via grant funding from Novartis.
A version of this article appeared on Medscape.com.
FROM BSR 2024
FDA Approves Second Gene Therapy for Hemophilia B
Patients are eligible for a one-time infusion of Pfizer’s gene therapy if they are currently using clotting factor IX prophylaxis therapy; have current or historical life-threatening hemorrhages; or have repeated, serious spontaneous bleeding episodes.
Beqvez is the second gene therapy the agency has approved for hemophilia B, a deficiency in clotting factor IX because of a faulty gene that occurs mostly in males. The FDA approved the first gene therapy, etranacogene dezaparvovec (Hemgenix), in November 2022.
Both therapies deliver a functional copy of the factor IX gene to liver cells via a viral vector.
Pfizer said the list price of Beqvez will be $3.5 million — the same price as Hemgenix. The argument for this hefty price tag is that these gene therapies offer the possibility of a cure whereas ongoing factor IX infusions can cost more than $20 million over a patient’s lifetime. Uptake of Hemgenix, however, has been slow, given the cost and concerns about the therapy’s durability and safety.
Beqvez was approved on the basis of the phase 3 BENEGENE-2 trial in 45 men with moderate to severe hemophilia B. These men had been on factor IX prophylaxis for at least 6 months and had tested negative for antibodies against the viral delivery vector.
The annualized bleeding rate fell from a mean of 4.5 events during the pretreatment period of at least 6 months to a mean of 2.5 events between week 12 and data cutoff (median, 1.8 years of follow-up), according to Pfizer’s press release. Overall, bleeding events were eliminated in 60% of patients who received the one-time infusion vs 29% of patients on factor IX prophylaxis therapy.
Overall, Pfizer reported that the gene therapy was “generally well-tolerated,” with an increase in transaminases reported as the most common adverse event. No deaths, serious infusion reactions, thrombotic events, or development of factor IX antibodies occurred.
Pfizer has said it will continue to monitor patients to assess the therapy’s long-term durability and safety over a 15-year period.
A version of this article appeared on Medscape.com.
Patients are eligible for a one-time infusion of Pfizer’s gene therapy if they are currently using clotting factor IX prophylaxis therapy; have current or historical life-threatening hemorrhages; or have repeated, serious spontaneous bleeding episodes.
Beqvez is the second gene therapy the agency has approved for hemophilia B, a deficiency in clotting factor IX because of a faulty gene that occurs mostly in males. The FDA approved the first gene therapy, etranacogene dezaparvovec (Hemgenix), in November 2022.
Both therapies deliver a functional copy of the factor IX gene to liver cells via a viral vector.
Pfizer said the list price of Beqvez will be $3.5 million — the same price as Hemgenix. The argument for this hefty price tag is that these gene therapies offer the possibility of a cure whereas ongoing factor IX infusions can cost more than $20 million over a patient’s lifetime. Uptake of Hemgenix, however, has been slow, given the cost and concerns about the therapy’s durability and safety.
Beqvez was approved on the basis of the phase 3 BENEGENE-2 trial in 45 men with moderate to severe hemophilia B. These men had been on factor IX prophylaxis for at least 6 months and had tested negative for antibodies against the viral delivery vector.
The annualized bleeding rate fell from a mean of 4.5 events during the pretreatment period of at least 6 months to a mean of 2.5 events between week 12 and data cutoff (median, 1.8 years of follow-up), according to Pfizer’s press release. Overall, bleeding events were eliminated in 60% of patients who received the one-time infusion vs 29% of patients on factor IX prophylaxis therapy.
Overall, Pfizer reported that the gene therapy was “generally well-tolerated,” with an increase in transaminases reported as the most common adverse event. No deaths, serious infusion reactions, thrombotic events, or development of factor IX antibodies occurred.
Pfizer has said it will continue to monitor patients to assess the therapy’s long-term durability and safety over a 15-year period.
A version of this article appeared on Medscape.com.
Patients are eligible for a one-time infusion of Pfizer’s gene therapy if they are currently using clotting factor IX prophylaxis therapy; have current or historical life-threatening hemorrhages; or have repeated, serious spontaneous bleeding episodes.
Beqvez is the second gene therapy the agency has approved for hemophilia B, a deficiency in clotting factor IX because of a faulty gene that occurs mostly in males. The FDA approved the first gene therapy, etranacogene dezaparvovec (Hemgenix), in November 2022.
Both therapies deliver a functional copy of the factor IX gene to liver cells via a viral vector.
Pfizer said the list price of Beqvez will be $3.5 million — the same price as Hemgenix. The argument for this hefty price tag is that these gene therapies offer the possibility of a cure whereas ongoing factor IX infusions can cost more than $20 million over a patient’s lifetime. Uptake of Hemgenix, however, has been slow, given the cost and concerns about the therapy’s durability and safety.
Beqvez was approved on the basis of the phase 3 BENEGENE-2 trial in 45 men with moderate to severe hemophilia B. These men had been on factor IX prophylaxis for at least 6 months and had tested negative for antibodies against the viral delivery vector.
The annualized bleeding rate fell from a mean of 4.5 events during the pretreatment period of at least 6 months to a mean of 2.5 events between week 12 and data cutoff (median, 1.8 years of follow-up), according to Pfizer’s press release. Overall, bleeding events were eliminated in 60% of patients who received the one-time infusion vs 29% of patients on factor IX prophylaxis therapy.
Overall, Pfizer reported that the gene therapy was “generally well-tolerated,” with an increase in transaminases reported as the most common adverse event. No deaths, serious infusion reactions, thrombotic events, or development of factor IX antibodies occurred.
Pfizer has said it will continue to monitor patients to assess the therapy’s long-term durability and safety over a 15-year period.
A version of this article appeared on Medscape.com.