User login
Mandatory reporting laws
Question: You are moonlighting in the emergency department and have just finished treating a 5-year-old boy with an apparent Colles’ fracture, who was accompanied by his mother with bruises on her face. Her exam revealed additional bruises over her abdominal wall. The mother said they accidentally tripped and fell down the stairs, and spontaneously denied any acts of violence in the family.
Given this scenario, which of the following is best?
A. You suspect both child and spousal abuse, but lack sufficient evidence to report the incident.
B. Failure to report based on reasonable suspicion alone may amount to a criminal offense punishable by possible imprisonment.
C. You may face a potential malpractice lawsuit if subsequent injuries caused by abuse could have been prevented had you reported.
D. Mandatory reporting laws apply not only to abuse of children and spouses, but also of the elderly and other vulnerable adults.
E. All are correct except A.
Answer: E. All doctors, especially those working in emergency departments, treat injuries on a regular basis. Accidents probably account for the majority of these injuries, but the most pernicious are those caused by willful abuse or neglect. Such conduct, believed to be widespread and underrecognized, victimizes children, women, the elderly, and other vulnerable groups.
Mandatory reporting laws arose from the need to identify and prevent these activities that cause serious harm and loss of lives. Physicians and other health care workers are in a prime position to diagnose or raise the suspicion of abuse and neglect. This article focuses on laws that mandate physician reporting of such behavior. Not addressed are other reportable situations such as certain infectious diseases, gunshot wounds, threats to third parties, and so on.
Child abuse
The best-known example of a mandatory reporting law relates to child abuse, which is broadly defined as when a parent or caretaker emotionally, physically, or sexually abuses, neglects, or abandons a child. Child abuse laws are intended to protect children from serious harm without abridging parental discipline of their children.
Cases of child abuse are pervasive; four or five children are tragically killed by abuse or neglect every day, and each year, some 6 million children are reported as victims of child abuse. Henry Kempe’s studies on the “battered child syndrome” in 1962 served to underscore the physician’s role in exposing child maltreatment, and 1973 saw the enactment of the Child Abuse Prevention and Treatment Act, which set standards for mandatory reporting as a condition for federal funding.
All U.S. states have statutes identifying persons who are required to report suspected child maltreatment to an appropriate agency, such as child protective services. Reasonable suspicion, without need for proof, is sufficient to trigger the mandatory reporting duty. A summary of the general reporting requirements, as well as each state’s key statutory features, are available at Child Welfare Information Gateway.1
Bruises, fractures, and burns are recurring examples of injuries resulting from child abuse, but there are many others, including severe emotional harm, which is an important consequence. Clues to abuse include a child’s fearful and anxious demeanor, wearing clothes to hide injuries, and inappropriate sexual conduct.2 The perpetrators and/or complicit parties typically blame an innocent home accident for the victim’s injuries to mislead the health care provider.
Elder abuse
Elder abuse is broadly construed to include physical, sexual, and psychological abuse, as well as financial exploitation and caregiver neglect.3 It is a serious problem in the United States, estimated in 2008 to affect 1 in 10 elders. The figure is likely an underestimate, because many elderly victims are afraid or unwilling to lodge a complaint against the abuser whom they love and may depend upon.4
The law, which protects the “elderly” (e.g., those aged 62 years or older in Hawaii), may also be extended to other younger vulnerable adults, who because of an impairment, are unable to 1) communicate or make responsible decisions to manage one’s own care or resources, 2) carry out or arrange for essential activities of daily living, or 3) protect one’s self from abuse.5
The law mandates reporting where there is reason to believe abuse has occurred or the vulnerable adult is in danger of abuse if immediate action is not taken. Reporting statutes for elder abuse vary somewhat on the identity of mandated reporters (health care providers are always included), the victim’s mental capacity, dwelling place (home or in an assisted-living facility), and type of purported activity that warrants reporting.
Domestic violence
As defined by the National Coalition Against Domestic Violence, “Domestic violence is the willful intimidation, physical assault, battery, sexual assault, and/or other abusive behavior as part of a systematic pattern of power and control perpetrated by one intimate partner against another. ... The frequency and severity of domestic violence can vary dramatically; however, the one constant component of domestic violence is one partner’s consistent efforts to maintain power and control over the other.”6 Domestic violence is said to have reached epidemic proportions, with one in four women experiencing it at some point in her life.
Virtually all states mandate the reporting of domestic violence by health care providers if there is a reasonable suspicion that observed patient injuries are the result of physical abuse.7 California, for example, requires the provider to call local law enforcement as soon as possible or to send in a written report within 48 hours.
There may be exceptions to required reporting, as when an adult victim withholds consent but accepts victim referral services. State laws encourage but do not always require that the health care provider inform the patient about the report, but federal law dictates otherwise unless this puts the patient at risk. Hawaii’s domestic violence laws were originally enacted to deter spousal abuse, but they now also protect other household members.8
Any individual who assumes a duty or responsibility pursuant to all of these reporting laws is immunized from criminal or civil liability. On the other hand, a mandated reporter who knowingly fails to report an incident or who willfully prevents another person from reporting such an incident commits a criminal offence.
In the case of a physician, there is the added risk of a malpractice lawsuit based on “violation of statute” (breach of a legal duty), should another injury occur down the road that was arguably preventable by his or her failure to report.
Experts generally believe that mandatory reporting laws are important in identifying child maltreatment. However, it has been asserted that despite a 5-decade history of mandatory reporting, no clear endpoints attest to the efficacy of this approach, and it is argued that no data exist to demonstrate that incremental increases in reporting have contributed to child safety.
Particularly challenging are attempts at impact comparisons between states with different policies. A number of countries, including the United Kingdom, do not have mandatory reporting laws and regulate reporting by professional societies.9
In addition, some critics of mandatory reporting raise concerns surrounding law enforcement showing up at the victim’s house to question the family about abuse, or to make an arrest or issue warnings. They posit that when the behavior of an abuser is under scrutiny, this can paradoxically create a potentially more dangerous environment for the patient-victim, whom the perpetrator now considers to have betrayed his or her trust. Others bemoan that revealing patient confidences violates the physician’s ethical code.
However, the intolerable incidence of violence against the vulnerable has properly made mandatory reporting the law of the land. Although the criminal penalty is currently light for failure to report, there is a move toward increasing its severity. Hawaii, for example, recently introduced Senate Bill 2477 that makes nonreporting by those required to do so a Class C felony punishable by up to 5 years in prison. The offense currently is a petty misdemeanor punishable by up to 30 days in jail.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Child Welfare Information Gateway (2016). Mandatory reporters of child abuse and neglect. Washington, D.C.: U.S. Department of Health and Human Services, Children’s Bureau. Available at www.childwelfare.gov; email: [email protected]; phone: 800-394-3366.
2. Available at www.childwelfare.gov/topics/can.
3. Available at www.justice.gov/elderjustice/elder-justice-statutes-0.
4. Available at www.cdc.gov/violenceprevention/elderabuse/index.html.
5. Hawaii Revised Statutes, Sec. 346-222, 346-224, 346-250, 412:3-114.5.
6. Available at ncadv.org.
7. Ann Emerg Med. 2002 Jan;39(1):56-60.
8. Hawaii Revised Statutes, Sec. 709-906.
9. Pediatrics. 2017 Apr;139(4). pii: e20163511.
Question: You are moonlighting in the emergency department and have just finished treating a 5-year-old boy with an apparent Colles’ fracture, who was accompanied by his mother with bruises on her face. Her exam revealed additional bruises over her abdominal wall. The mother said they accidentally tripped and fell down the stairs, and spontaneously denied any acts of violence in the family.
Given this scenario, which of the following is best?
A. You suspect both child and spousal abuse, but lack sufficient evidence to report the incident.
B. Failure to report based on reasonable suspicion alone may amount to a criminal offense punishable by possible imprisonment.
C. You may face a potential malpractice lawsuit if subsequent injuries caused by abuse could have been prevented had you reported.
D. Mandatory reporting laws apply not only to abuse of children and spouses, but also of the elderly and other vulnerable adults.
E. All are correct except A.
Answer: E. All doctors, especially those working in emergency departments, treat injuries on a regular basis. Accidents probably account for the majority of these injuries, but the most pernicious are those caused by willful abuse or neglect. Such conduct, believed to be widespread and underrecognized, victimizes children, women, the elderly, and other vulnerable groups.
Mandatory reporting laws arose from the need to identify and prevent these activities that cause serious harm and loss of lives. Physicians and other health care workers are in a prime position to diagnose or raise the suspicion of abuse and neglect. This article focuses on laws that mandate physician reporting of such behavior. Not addressed are other reportable situations such as certain infectious diseases, gunshot wounds, threats to third parties, and so on.
Child abuse
The best-known example of a mandatory reporting law relates to child abuse, which is broadly defined as when a parent or caretaker emotionally, physically, or sexually abuses, neglects, or abandons a child. Child abuse laws are intended to protect children from serious harm without abridging parental discipline of their children.
Cases of child abuse are pervasive; four or five children are tragically killed by abuse or neglect every day, and each year, some 6 million children are reported as victims of child abuse. Henry Kempe’s studies on the “battered child syndrome” in 1962 served to underscore the physician’s role in exposing child maltreatment, and 1973 saw the enactment of the Child Abuse Prevention and Treatment Act, which set standards for mandatory reporting as a condition for federal funding.
All U.S. states have statutes identifying persons who are required to report suspected child maltreatment to an appropriate agency, such as child protective services. Reasonable suspicion, without need for proof, is sufficient to trigger the mandatory reporting duty. A summary of the general reporting requirements, as well as each state’s key statutory features, are available at Child Welfare Information Gateway.1
Bruises, fractures, and burns are recurring examples of injuries resulting from child abuse, but there are many others, including severe emotional harm, which is an important consequence. Clues to abuse include a child’s fearful and anxious demeanor, wearing clothes to hide injuries, and inappropriate sexual conduct.2 The perpetrators and/or complicit parties typically blame an innocent home accident for the victim’s injuries to mislead the health care provider.
Elder abuse
Elder abuse is broadly construed to include physical, sexual, and psychological abuse, as well as financial exploitation and caregiver neglect.3 It is a serious problem in the United States, estimated in 2008 to affect 1 in 10 elders. The figure is likely an underestimate, because many elderly victims are afraid or unwilling to lodge a complaint against the abuser whom they love and may depend upon.4
The law, which protects the “elderly” (e.g., those aged 62 years or older in Hawaii), may also be extended to other younger vulnerable adults, who because of an impairment, are unable to 1) communicate or make responsible decisions to manage one’s own care or resources, 2) carry out or arrange for essential activities of daily living, or 3) protect one’s self from abuse.5
The law mandates reporting where there is reason to believe abuse has occurred or the vulnerable adult is in danger of abuse if immediate action is not taken. Reporting statutes for elder abuse vary somewhat on the identity of mandated reporters (health care providers are always included), the victim’s mental capacity, dwelling place (home or in an assisted-living facility), and type of purported activity that warrants reporting.
Domestic violence
As defined by the National Coalition Against Domestic Violence, “Domestic violence is the willful intimidation, physical assault, battery, sexual assault, and/or other abusive behavior as part of a systematic pattern of power and control perpetrated by one intimate partner against another. ... The frequency and severity of domestic violence can vary dramatically; however, the one constant component of domestic violence is one partner’s consistent efforts to maintain power and control over the other.”6 Domestic violence is said to have reached epidemic proportions, with one in four women experiencing it at some point in her life.
Virtually all states mandate the reporting of domestic violence by health care providers if there is a reasonable suspicion that observed patient injuries are the result of physical abuse.7 California, for example, requires the provider to call local law enforcement as soon as possible or to send in a written report within 48 hours.
There may be exceptions to required reporting, as when an adult victim withholds consent but accepts victim referral services. State laws encourage but do not always require that the health care provider inform the patient about the report, but federal law dictates otherwise unless this puts the patient at risk. Hawaii’s domestic violence laws were originally enacted to deter spousal abuse, but they now also protect other household members.8
Any individual who assumes a duty or responsibility pursuant to all of these reporting laws is immunized from criminal or civil liability. On the other hand, a mandated reporter who knowingly fails to report an incident or who willfully prevents another person from reporting such an incident commits a criminal offence.
In the case of a physician, there is the added risk of a malpractice lawsuit based on “violation of statute” (breach of a legal duty), should another injury occur down the road that was arguably preventable by his or her failure to report.
Experts generally believe that mandatory reporting laws are important in identifying child maltreatment. However, it has been asserted that despite a 5-decade history of mandatory reporting, no clear endpoints attest to the efficacy of this approach, and it is argued that no data exist to demonstrate that incremental increases in reporting have contributed to child safety.
Particularly challenging are attempts at impact comparisons between states with different policies. A number of countries, including the United Kingdom, do not have mandatory reporting laws and regulate reporting by professional societies.9
In addition, some critics of mandatory reporting raise concerns surrounding law enforcement showing up at the victim’s house to question the family about abuse, or to make an arrest or issue warnings. They posit that when the behavior of an abuser is under scrutiny, this can paradoxically create a potentially more dangerous environment for the patient-victim, whom the perpetrator now considers to have betrayed his or her trust. Others bemoan that revealing patient confidences violates the physician’s ethical code.
However, the intolerable incidence of violence against the vulnerable has properly made mandatory reporting the law of the land. Although the criminal penalty is currently light for failure to report, there is a move toward increasing its severity. Hawaii, for example, recently introduced Senate Bill 2477 that makes nonreporting by those required to do so a Class C felony punishable by up to 5 years in prison. The offense currently is a petty misdemeanor punishable by up to 30 days in jail.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Child Welfare Information Gateway (2016). Mandatory reporters of child abuse and neglect. Washington, D.C.: U.S. Department of Health and Human Services, Children’s Bureau. Available at www.childwelfare.gov; email: [email protected]; phone: 800-394-3366.
2. Available at www.childwelfare.gov/topics/can.
3. Available at www.justice.gov/elderjustice/elder-justice-statutes-0.
4. Available at www.cdc.gov/violenceprevention/elderabuse/index.html.
5. Hawaii Revised Statutes, Sec. 346-222, 346-224, 346-250, 412:3-114.5.
6. Available at ncadv.org.
7. Ann Emerg Med. 2002 Jan;39(1):56-60.
8. Hawaii Revised Statutes, Sec. 709-906.
9. Pediatrics. 2017 Apr;139(4). pii: e20163511.
Question: You are moonlighting in the emergency department and have just finished treating a 5-year-old boy with an apparent Colles’ fracture, who was accompanied by his mother with bruises on her face. Her exam revealed additional bruises over her abdominal wall. The mother said they accidentally tripped and fell down the stairs, and spontaneously denied any acts of violence in the family.
Given this scenario, which of the following is best?
A. You suspect both child and spousal abuse, but lack sufficient evidence to report the incident.
B. Failure to report based on reasonable suspicion alone may amount to a criminal offense punishable by possible imprisonment.
C. You may face a potential malpractice lawsuit if subsequent injuries caused by abuse could have been prevented had you reported.
D. Mandatory reporting laws apply not only to abuse of children and spouses, but also of the elderly and other vulnerable adults.
E. All are correct except A.
Answer: E. All doctors, especially those working in emergency departments, treat injuries on a regular basis. Accidents probably account for the majority of these injuries, but the most pernicious are those caused by willful abuse or neglect. Such conduct, believed to be widespread and underrecognized, victimizes children, women, the elderly, and other vulnerable groups.
Mandatory reporting laws arose from the need to identify and prevent these activities that cause serious harm and loss of lives. Physicians and other health care workers are in a prime position to diagnose or raise the suspicion of abuse and neglect. This article focuses on laws that mandate physician reporting of such behavior. Not addressed are other reportable situations such as certain infectious diseases, gunshot wounds, threats to third parties, and so on.
Child abuse
The best-known example of a mandatory reporting law relates to child abuse, which is broadly defined as when a parent or caretaker emotionally, physically, or sexually abuses, neglects, or abandons a child. Child abuse laws are intended to protect children from serious harm without abridging parental discipline of their children.
Cases of child abuse are pervasive; four or five children are tragically killed by abuse or neglect every day, and each year, some 6 million children are reported as victims of child abuse. Henry Kempe’s studies on the “battered child syndrome” in 1962 served to underscore the physician’s role in exposing child maltreatment, and 1973 saw the enactment of the Child Abuse Prevention and Treatment Act, which set standards for mandatory reporting as a condition for federal funding.
All U.S. states have statutes identifying persons who are required to report suspected child maltreatment to an appropriate agency, such as child protective services. Reasonable suspicion, without need for proof, is sufficient to trigger the mandatory reporting duty. A summary of the general reporting requirements, as well as each state’s key statutory features, are available at Child Welfare Information Gateway.1
Bruises, fractures, and burns are recurring examples of injuries resulting from child abuse, but there are many others, including severe emotional harm, which is an important consequence. Clues to abuse include a child’s fearful and anxious demeanor, wearing clothes to hide injuries, and inappropriate sexual conduct.2 The perpetrators and/or complicit parties typically blame an innocent home accident for the victim’s injuries to mislead the health care provider.
Elder abuse
Elder abuse is broadly construed to include physical, sexual, and psychological abuse, as well as financial exploitation and caregiver neglect.3 It is a serious problem in the United States, estimated in 2008 to affect 1 in 10 elders. The figure is likely an underestimate, because many elderly victims are afraid or unwilling to lodge a complaint against the abuser whom they love and may depend upon.4
The law, which protects the “elderly” (e.g., those aged 62 years or older in Hawaii), may also be extended to other younger vulnerable adults, who because of an impairment, are unable to 1) communicate or make responsible decisions to manage one’s own care or resources, 2) carry out or arrange for essential activities of daily living, or 3) protect one’s self from abuse.5
The law mandates reporting where there is reason to believe abuse has occurred or the vulnerable adult is in danger of abuse if immediate action is not taken. Reporting statutes for elder abuse vary somewhat on the identity of mandated reporters (health care providers are always included), the victim’s mental capacity, dwelling place (home or in an assisted-living facility), and type of purported activity that warrants reporting.
Domestic violence
As defined by the National Coalition Against Domestic Violence, “Domestic violence is the willful intimidation, physical assault, battery, sexual assault, and/or other abusive behavior as part of a systematic pattern of power and control perpetrated by one intimate partner against another. ... The frequency and severity of domestic violence can vary dramatically; however, the one constant component of domestic violence is one partner’s consistent efforts to maintain power and control over the other.”6 Domestic violence is said to have reached epidemic proportions, with one in four women experiencing it at some point in her life.
Virtually all states mandate the reporting of domestic violence by health care providers if there is a reasonable suspicion that observed patient injuries are the result of physical abuse.7 California, for example, requires the provider to call local law enforcement as soon as possible or to send in a written report within 48 hours.
There may be exceptions to required reporting, as when an adult victim withholds consent but accepts victim referral services. State laws encourage but do not always require that the health care provider inform the patient about the report, but federal law dictates otherwise unless this puts the patient at risk. Hawaii’s domestic violence laws were originally enacted to deter spousal abuse, but they now also protect other household members.8
Any individual who assumes a duty or responsibility pursuant to all of these reporting laws is immunized from criminal or civil liability. On the other hand, a mandated reporter who knowingly fails to report an incident or who willfully prevents another person from reporting such an incident commits a criminal offence.
In the case of a physician, there is the added risk of a malpractice lawsuit based on “violation of statute” (breach of a legal duty), should another injury occur down the road that was arguably preventable by his or her failure to report.
Experts generally believe that mandatory reporting laws are important in identifying child maltreatment. However, it has been asserted that despite a 5-decade history of mandatory reporting, no clear endpoints attest to the efficacy of this approach, and it is argued that no data exist to demonstrate that incremental increases in reporting have contributed to child safety.
Particularly challenging are attempts at impact comparisons between states with different policies. A number of countries, including the United Kingdom, do not have mandatory reporting laws and regulate reporting by professional societies.9
In addition, some critics of mandatory reporting raise concerns surrounding law enforcement showing up at the victim’s house to question the family about abuse, or to make an arrest or issue warnings. They posit that when the behavior of an abuser is under scrutiny, this can paradoxically create a potentially more dangerous environment for the patient-victim, whom the perpetrator now considers to have betrayed his or her trust. Others bemoan that revealing patient confidences violates the physician’s ethical code.
However, the intolerable incidence of violence against the vulnerable has properly made mandatory reporting the law of the land. Although the criminal penalty is currently light for failure to report, there is a move toward increasing its severity. Hawaii, for example, recently introduced Senate Bill 2477 that makes nonreporting by those required to do so a Class C felony punishable by up to 5 years in prison. The offense currently is a petty misdemeanor punishable by up to 30 days in jail.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Child Welfare Information Gateway (2016). Mandatory reporters of child abuse and neglect. Washington, D.C.: U.S. Department of Health and Human Services, Children’s Bureau. Available at www.childwelfare.gov; email: [email protected]; phone: 800-394-3366.
2. Available at www.childwelfare.gov/topics/can.
3. Available at www.justice.gov/elderjustice/elder-justice-statutes-0.
4. Available at www.cdc.gov/violenceprevention/elderabuse/index.html.
5. Hawaii Revised Statutes, Sec. 346-222, 346-224, 346-250, 412:3-114.5.
6. Available at ncadv.org.
7. Ann Emerg Med. 2002 Jan;39(1):56-60.
8. Hawaii Revised Statutes, Sec. 709-906.
9. Pediatrics. 2017 Apr;139(4). pii: e20163511.
Obesity paradox applies to post-stroke mortality
CHICAGO – Overweight and obese military veterans who experienced an in-hospital stroke had a lower 30-day and 1-year all-cause mortality than did those who were normal weight in a large national study, Lauren Costa reported at the American Heart Association scientific sessions.
Underweight patients had a significantly increased mortality risk, added Ms. Costa of the VA Boston Healthcare System.
It’s yet another instance of what is known as the obesity paradox, which has also been described in patients with heart failure, acute coronary syndrome, MI, chronic obstructive pulmonary disease, and other conditions.
Ms. Costa presented a retrospective study of 26,267 patients in the Veterans Health Administration database who had a first stroke in-hospital during 2002-2012. There were subsequently 14,166 deaths, including 2,473 within the first 30 days and 5,854 in the first year post stroke.
Each patient’s body mass index was calculated based on the average of all BMI measurements obtained 1-24 months prior to the stroke. The analysis of the relationship between BMI and poststroke mortality included extensive statistical adjustment for potential confounders, including age, sex, smoking, cancer, dementia, peripheral artery disease, diabetes, coronary heart disease, atrial fibrillation, chronic kidney disease, use of statins, and antihypertensive therapy.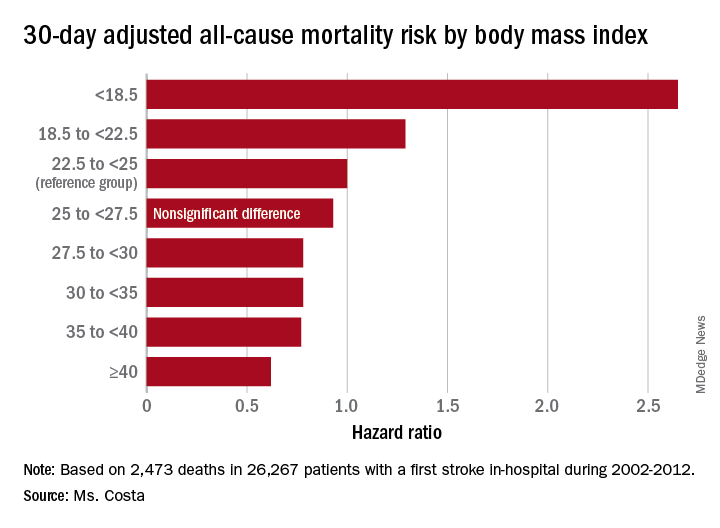
Breaking down the study population into eight BMI categories, Ms. Costa found that the adjusted risk of 30-day all-cause mortality post stroke was reduced by 22%-38% in patients in the overweight or obese groupings, compared with the reference population with a normal-weight BMI of 22.5 to less than 25 kg/m2.
One-year, all-cause mortality showed the same pattern of BMI-based significant differences.
Of deaths within 30 days post stroke, 34% were stroke-related. In an analysis restricted to that group, the evidence of an obesity paradox was attenuated. Indeed, the only BMI group with an adjusted 30-day stroke-related mortality significantly different from the normal-weight reference group were patients with Class III obesity, defined as a BMI of 40 or more. Their risk was reduced by 45%.
The obesity paradox remains a controversial issue among epidemiologists. The increased mortality associated with being underweight among patients with diseases where the obesity paradox has been documented is widely thought to be caused by frailty and/or an underlying illness not adjusted for in analyses. But the mechanism for the reduced mortality risk in overweight and obese patients seen in the VA stroke study and other studies remains unknown despite much speculation.
Ms. Costa reported having no financial conflicts regarding her study, which was supported by the Department of Veterans Affairs.
SOURCE: Costa L. Circulation. 2018;138(suppl 1): Abstract 14288.
CHICAGO – Overweight and obese military veterans who experienced an in-hospital stroke had a lower 30-day and 1-year all-cause mortality than did those who were normal weight in a large national study, Lauren Costa reported at the American Heart Association scientific sessions.
Underweight patients had a significantly increased mortality risk, added Ms. Costa of the VA Boston Healthcare System.
It’s yet another instance of what is known as the obesity paradox, which has also been described in patients with heart failure, acute coronary syndrome, MI, chronic obstructive pulmonary disease, and other conditions.
Ms. Costa presented a retrospective study of 26,267 patients in the Veterans Health Administration database who had a first stroke in-hospital during 2002-2012. There were subsequently 14,166 deaths, including 2,473 within the first 30 days and 5,854 in the first year post stroke.
Each patient’s body mass index was calculated based on the average of all BMI measurements obtained 1-24 months prior to the stroke. The analysis of the relationship between BMI and poststroke mortality included extensive statistical adjustment for potential confounders, including age, sex, smoking, cancer, dementia, peripheral artery disease, diabetes, coronary heart disease, atrial fibrillation, chronic kidney disease, use of statins, and antihypertensive therapy.
Breaking down the study population into eight BMI categories, Ms. Costa found that the adjusted risk of 30-day all-cause mortality post stroke was reduced by 22%-38% in patients in the overweight or obese groupings, compared with the reference population with a normal-weight BMI of 22.5 to less than 25 kg/m2.
One-year, all-cause mortality showed the same pattern of BMI-based significant differences.
Of deaths within 30 days post stroke, 34% were stroke-related. In an analysis restricted to that group, the evidence of an obesity paradox was attenuated. Indeed, the only BMI group with an adjusted 30-day stroke-related mortality significantly different from the normal-weight reference group were patients with Class III obesity, defined as a BMI of 40 or more. Their risk was reduced by 45%.
The obesity paradox remains a controversial issue among epidemiologists. The increased mortality associated with being underweight among patients with diseases where the obesity paradox has been documented is widely thought to be caused by frailty and/or an underlying illness not adjusted for in analyses. But the mechanism for the reduced mortality risk in overweight and obese patients seen in the VA stroke study and other studies remains unknown despite much speculation.
Ms. Costa reported having no financial conflicts regarding her study, which was supported by the Department of Veterans Affairs.
SOURCE: Costa L. Circulation. 2018;138(suppl 1): Abstract 14288.
CHICAGO – Overweight and obese military veterans who experienced an in-hospital stroke had a lower 30-day and 1-year all-cause mortality than did those who were normal weight in a large national study, Lauren Costa reported at the American Heart Association scientific sessions.
Underweight patients had a significantly increased mortality risk, added Ms. Costa of the VA Boston Healthcare System.
It’s yet another instance of what is known as the obesity paradox, which has also been described in patients with heart failure, acute coronary syndrome, MI, chronic obstructive pulmonary disease, and other conditions.
Ms. Costa presented a retrospective study of 26,267 patients in the Veterans Health Administration database who had a first stroke in-hospital during 2002-2012. There were subsequently 14,166 deaths, including 2,473 within the first 30 days and 5,854 in the first year post stroke.
Each patient’s body mass index was calculated based on the average of all BMI measurements obtained 1-24 months prior to the stroke. The analysis of the relationship between BMI and poststroke mortality included extensive statistical adjustment for potential confounders, including age, sex, smoking, cancer, dementia, peripheral artery disease, diabetes, coronary heart disease, atrial fibrillation, chronic kidney disease, use of statins, and antihypertensive therapy.
Breaking down the study population into eight BMI categories, Ms. Costa found that the adjusted risk of 30-day all-cause mortality post stroke was reduced by 22%-38% in patients in the overweight or obese groupings, compared with the reference population with a normal-weight BMI of 22.5 to less than 25 kg/m2.
One-year, all-cause mortality showed the same pattern of BMI-based significant differences.
Of deaths within 30 days post stroke, 34% were stroke-related. In an analysis restricted to that group, the evidence of an obesity paradox was attenuated. Indeed, the only BMI group with an adjusted 30-day stroke-related mortality significantly different from the normal-weight reference group were patients with Class III obesity, defined as a BMI of 40 or more. Their risk was reduced by 45%.
The obesity paradox remains a controversial issue among epidemiologists. The increased mortality associated with being underweight among patients with diseases where the obesity paradox has been documented is widely thought to be caused by frailty and/or an underlying illness not adjusted for in analyses. But the mechanism for the reduced mortality risk in overweight and obese patients seen in the VA stroke study and other studies remains unknown despite much speculation.
Ms. Costa reported having no financial conflicts regarding her study, which was supported by the Department of Veterans Affairs.
SOURCE: Costa L. Circulation. 2018;138(suppl 1): Abstract 14288.
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: Heavier stroke patients have lower 30-day and 1-year all-cause mortality.
Major finding: The 30-day stroke-related mortality rate after in-hospital stroke was reduced by 45% in VA patients with Class III obesity.
Study details: This retrospective study looked at the relationship between body mass index and post-stroke mortality in more than 26,000 veterans who had an inpatient stroke, with extensive adjustments made for potential confounders.
Disclosures: The presenter reported having no financial conflicts regarding the study, which was sponsored by the Department of Veterans Affairs.
Source: Costa L. Circulation. 2018;138(suppl 1): Abstract 14288.
Participate in a Retirement Plan
"Tranche" dates -- for completing all the paperwork for the Kai-Zen retirement accelerator program offered through the SVS Affinity Program of expanded benefits -- have been set for 2019. The first dates are Jan. 28, March 11 and May 6. Generally, paperwork can take approximately 30 or more days, but someone who has already started could meet the Jan. 28 date. Kai-Zen combines financing and life insurance, with premiums jointly funded by lenders and the participant (or an employer). It can help members speed investment growth in their life insurance policies, using leverage that provides up to 60 percent more death benefit protection and 60 percent more supplemental retirement income. See a video of how Kai-Zen works here. For more information contact Mark Blocker at [email protected] or at 312-291-4472.
"Tranche" dates -- for completing all the paperwork for the Kai-Zen retirement accelerator program offered through the SVS Affinity Program of expanded benefits -- have been set for 2019. The first dates are Jan. 28, March 11 and May 6. Generally, paperwork can take approximately 30 or more days, but someone who has already started could meet the Jan. 28 date. Kai-Zen combines financing and life insurance, with premiums jointly funded by lenders and the participant (or an employer). It can help members speed investment growth in their life insurance policies, using leverage that provides up to 60 percent more death benefit protection and 60 percent more supplemental retirement income. See a video of how Kai-Zen works here. For more information contact Mark Blocker at [email protected] or at 312-291-4472.
"Tranche" dates -- for completing all the paperwork for the Kai-Zen retirement accelerator program offered through the SVS Affinity Program of expanded benefits -- have been set for 2019. The first dates are Jan. 28, March 11 and May 6. Generally, paperwork can take approximately 30 or more days, but someone who has already started could meet the Jan. 28 date. Kai-Zen combines financing and life insurance, with premiums jointly funded by lenders and the participant (or an employer). It can help members speed investment growth in their life insurance policies, using leverage that provides up to 60 percent more death benefit protection and 60 percent more supplemental retirement income. See a video of how Kai-Zen works here. For more information contact Mark Blocker at [email protected] or at 312-291-4472.
Emerging Roles of Social Media in Dermatology
As the residents on the podium ran through case presentations at the Texas Dermatological Society meeting this past fall (September 21-22, 2018; Galveston, Texas), I discretely surveyed the room. To no surprise, perhaps half of the attendees at some point during the hour-long presentation glanced down at their smartphones, and 2018 statistics suggest that approximately 74% of these Internet glances were made by engagers of social media sites.1 My FOMO (fear of missing out) kicked in. What was everyone looking at? I opened Instagram on my smartphone and plastered at the top of my home page were Texas Dermatological Society–related “stories” posted by other dermatology residents from across the state, one story featuring the very presentation I was attending. I peeked 2 rows ahead to find the social media “influencer” I have been following on Instagram for months in real life for the first time.
It is not just the younger population glued to their social media accounts. In fact, Facebook boasted a more than 80% increase in users 55 years and older between 2011 and 2014 and a 41% increase in users aged 35 to 54 years.2 In total, there were 3.2 billion social media users globally in 2018.3 With such a large portion of the population engaged in social media, it is no wonder that it has become a rapidly emerging presence within the field of dermatology.
#Ad
Social media has become a powerful marketing tool for the practicing dermatologist. In a recent survey, 41% of social media users reported that social media influenced their choice of a particular physician, facility, or medical practice.4 Corresponding to this behavior, dermatology practices also have used social media to educate patients on services offered, acquire new patients, engage existing patients, create brand loyalty, become a trusted source of medical information in a sea of digital misinformation, and facilitate positive word-of-mouth opportunities.5 In fact, 53% of physician practices in the United States operate a Facebook page.6 For these physicians, marketing through social media carries the advantages of low cost and rapid transmission of information to a wide audience.7 Furthermore, the development of business insights and statistics by some social media platforms, such as those available to users on business profiles on Instagram, enables practices and marketers to target their audiences and optimize reach.
#DermLife
The role of social media in dermatology extends far beyond marketing. Lifestyle blogs centered on daily life as a medical provider, even within the field of dermatology, are gaining popularity. Dermatology-centered lifestyle blogs often incorporate the root derm in their handle, enabling other users to identify the account holder and interact in meaningful ways. According to a post from one popular Instagram influencer Dr. Audrey Sue Cruz (@dr.audreyxsue), such profiles may serve to prevent burnout, provide a creative outlet, share life as a resident, develop a supportive community, provide mentorship, and spread inspiration.
#Hashtag
Another interesting utility of social media is the use of standardized hashtags to facilitate scientific and clinical dialogue among medical professionals. Standardization of hashtag ontology on Twitter and Instagram has been adopted by the urology and gastroenterology fields to filter out “noise” by individuals not intending to join academic discussion.8 In dermatology, standardized hashtags have not been adopted, to my knowledge; however, a search for esoteric dermatologic terms such as #dermatopathology or #mohssurgery directs users to specialty-specific discussions.
#DontFryDay
Another role of social media in dermatology is dissemination of information. One notable example is the reach on Twitter of the “Don’t Fry Day” campaign, an annual campaign by the National Council on Skin Cancer Prevention to promote sun safety awareness and sun protection behaviors. In a recent study by Nguyen et al,9 the hashtag #DontFryDay was tracked on Twitter to assess the reach of the campaign. They found that this campaign had an impressive reach of approximately 1200 contributors, resulting in more than 16.5 million impressions; 18 celebrities and verified individuals accounted for 8,735,549 impressions.9
Despite the large potential for dissemination of information on social media, in 2014 none of the top 10 dermatologic journals or professional dermatologic organizations maintained an Instagram account. Only one of the top 10 patient advocate groups related to dermatology conditions—the Melanoma Research Foundation—was found on Instagram as of 2014.10 Furthermore, none of the top 10 most popular dermatology journals, professional dermatology organizations, or dermatology-related patient advocate groups could be found on Tumblr as of 2014.11 Although some of the aforementioned organizations have since adopted social media accounts, such as Cutis and Dermatology News (@mdedgederm) on Instagram in 2018, these social media platforms remain largely untapped outlets for dissemination of information to the public by reputable sources.
#VerifyHealthcare
Although social media has offered many advantages to the field of dermatology, it also has brought about unique challenges such as blind authorship, lack of source citation, and presentation of opinion as fact.7 To compound the challenge, 90% of millennials aged 18 to 24 years reportedly trust health care information shared by others on social media.12 Do we, as dermatologists, have a duty to take to social media to provide reputable health information? In an effort to address this emerging problem, popular Instagram influencer Dr. Austin Chiang (@austinchiangmd) initiated the #VerifyHealthcare movement, which called for physicians active on social media to practice transparency by providing users with their credentials.13 The goal of the movement is to help users differentiate medical information disseminated by trained medical professionals from misinformation by disreputable sources.
Final Thoughts
Despite its shortcomings, the emerging roles of social media in dermatology have proven to be a prominent force here to stay, providing new and innovative opportunities to dermatologists for social networking, dissemination of health information, motivation and inspiration, and marketing.
- Warden C. 30 statistics on social media and healthcare. Referral MD website. https://getreferralmd.com/2017/01/30-facts-statistics-on-social-media-and-healthcare/. Accessed January 16, 2019.
- Saul DJ. 3 million teens leave Facebook in 3 years: the Facebook demographic report. ISL website. https://isl.co/2014/01/3-million-teens-leave-facebook-in-3-years-the-2014-facebook-demographic-report/. Published January 15, 2014. Accessed January 9, 2019.
- Chaffey D. Global social media research summary 2018. Smart Insights website. https://www.smartinsights.com/social-media-marketing/social-media-strategy/new-global-social-media-research/. Published November 23, 2018. Accessed January 3, 2019.
- Ottenhoff M. Infographic: rising use of social and mobile in healthcare. The Spark Report. December 17, 2012. http://thesparkreport.com/infographic-social-mobile-healthcare/. Accessed January 9, 2019.
- Benabio J. The value of social media for dermatologists. Cutis. 2013;91:269-270.
- The healthcare social media shakeup. CDW Healthcare website. http://www.cdwcommunit.com/resources/infographic/social-media/. Accessed January 9, 2019.
- Vance K, Howe W, Dellavelle RP. Social internet sites as a source of public health information. Dermatol Clin. 2009;27:133-136.
- Chang AL, Vartabedian B, Spiegel B. Harnessing the hashtag: a standard approach to GI dialogue on social media. Am J Gastroeneterol. 2016;111:1082-1084.
- Nguyen JL, Heckman C, Perna F. Analysis of the Twitter “Don’t Fry Day” campaign. JAMA Dermatol. 2018;154:961-962.
- Karimkhani C, Connett J, Boyars L, et al. Dermatology on Instagram. Dermatol Online J. 2014;20. pii:13030/qt71g178w9.
- Correnti C, Boyars L, Karimkhani C, et al. Dermatology on Tumblr. Dermatol Online J. 2014;20:22642.
- Honigman B. 24 outstanding statistics on how social media has impacted healthcare. Referral MD website. https://getreferralmd.com/2013/09/healthcare-social-media-statistics. Accessed January 16, 2019.
- Oliver E. #VerifyHealthcare campaign seeks to increase social media transparency—5 insights. Becker’s GI & Endoscopy website. https://www.beckersasc.com/gastroenterology-and-endoscopy/verifyhealthcare-campaign-seeks-to-increase-social-media-transparency-5-insights.html. Published September 24, 2018. Accessed January 16, 2019.
As the residents on the podium ran through case presentations at the Texas Dermatological Society meeting this past fall (September 21-22, 2018; Galveston, Texas), I discretely surveyed the room. To no surprise, perhaps half of the attendees at some point during the hour-long presentation glanced down at their smartphones, and 2018 statistics suggest that approximately 74% of these Internet glances were made by engagers of social media sites.1 My FOMO (fear of missing out) kicked in. What was everyone looking at? I opened Instagram on my smartphone and plastered at the top of my home page were Texas Dermatological Society–related “stories” posted by other dermatology residents from across the state, one story featuring the very presentation I was attending. I peeked 2 rows ahead to find the social media “influencer” I have been following on Instagram for months in real life for the first time.
It is not just the younger population glued to their social media accounts. In fact, Facebook boasted a more than 80% increase in users 55 years and older between 2011 and 2014 and a 41% increase in users aged 35 to 54 years.2 In total, there were 3.2 billion social media users globally in 2018.3 With such a large portion of the population engaged in social media, it is no wonder that it has become a rapidly emerging presence within the field of dermatology.
#Ad
Social media has become a powerful marketing tool for the practicing dermatologist. In a recent survey, 41% of social media users reported that social media influenced their choice of a particular physician, facility, or medical practice.4 Corresponding to this behavior, dermatology practices also have used social media to educate patients on services offered, acquire new patients, engage existing patients, create brand loyalty, become a trusted source of medical information in a sea of digital misinformation, and facilitate positive word-of-mouth opportunities.5 In fact, 53% of physician practices in the United States operate a Facebook page.6 For these physicians, marketing through social media carries the advantages of low cost and rapid transmission of information to a wide audience.7 Furthermore, the development of business insights and statistics by some social media platforms, such as those available to users on business profiles on Instagram, enables practices and marketers to target their audiences and optimize reach.
#DermLife
The role of social media in dermatology extends far beyond marketing. Lifestyle blogs centered on daily life as a medical provider, even within the field of dermatology, are gaining popularity. Dermatology-centered lifestyle blogs often incorporate the root derm in their handle, enabling other users to identify the account holder and interact in meaningful ways. According to a post from one popular Instagram influencer Dr. Audrey Sue Cruz (@dr.audreyxsue), such profiles may serve to prevent burnout, provide a creative outlet, share life as a resident, develop a supportive community, provide mentorship, and spread inspiration.
#Hashtag
Another interesting utility of social media is the use of standardized hashtags to facilitate scientific and clinical dialogue among medical professionals. Standardization of hashtag ontology on Twitter and Instagram has been adopted by the urology and gastroenterology fields to filter out “noise” by individuals not intending to join academic discussion.8 In dermatology, standardized hashtags have not been adopted, to my knowledge; however, a search for esoteric dermatologic terms such as #dermatopathology or #mohssurgery directs users to specialty-specific discussions.
#DontFryDay
Another role of social media in dermatology is dissemination of information. One notable example is the reach on Twitter of the “Don’t Fry Day” campaign, an annual campaign by the National Council on Skin Cancer Prevention to promote sun safety awareness and sun protection behaviors. In a recent study by Nguyen et al,9 the hashtag #DontFryDay was tracked on Twitter to assess the reach of the campaign. They found that this campaign had an impressive reach of approximately 1200 contributors, resulting in more than 16.5 million impressions; 18 celebrities and verified individuals accounted for 8,735,549 impressions.9
Despite the large potential for dissemination of information on social media, in 2014 none of the top 10 dermatologic journals or professional dermatologic organizations maintained an Instagram account. Only one of the top 10 patient advocate groups related to dermatology conditions—the Melanoma Research Foundation—was found on Instagram as of 2014.10 Furthermore, none of the top 10 most popular dermatology journals, professional dermatology organizations, or dermatology-related patient advocate groups could be found on Tumblr as of 2014.11 Although some of the aforementioned organizations have since adopted social media accounts, such as Cutis and Dermatology News (@mdedgederm) on Instagram in 2018, these social media platforms remain largely untapped outlets for dissemination of information to the public by reputable sources.
#VerifyHealthcare
Although social media has offered many advantages to the field of dermatology, it also has brought about unique challenges such as blind authorship, lack of source citation, and presentation of opinion as fact.7 To compound the challenge, 90% of millennials aged 18 to 24 years reportedly trust health care information shared by others on social media.12 Do we, as dermatologists, have a duty to take to social media to provide reputable health information? In an effort to address this emerging problem, popular Instagram influencer Dr. Austin Chiang (@austinchiangmd) initiated the #VerifyHealthcare movement, which called for physicians active on social media to practice transparency by providing users with their credentials.13 The goal of the movement is to help users differentiate medical information disseminated by trained medical professionals from misinformation by disreputable sources.
Final Thoughts
Despite its shortcomings, the emerging roles of social media in dermatology have proven to be a prominent force here to stay, providing new and innovative opportunities to dermatologists for social networking, dissemination of health information, motivation and inspiration, and marketing.
As the residents on the podium ran through case presentations at the Texas Dermatological Society meeting this past fall (September 21-22, 2018; Galveston, Texas), I discretely surveyed the room. To no surprise, perhaps half of the attendees at some point during the hour-long presentation glanced down at their smartphones, and 2018 statistics suggest that approximately 74% of these Internet glances were made by engagers of social media sites.1 My FOMO (fear of missing out) kicked in. What was everyone looking at? I opened Instagram on my smartphone and plastered at the top of my home page were Texas Dermatological Society–related “stories” posted by other dermatology residents from across the state, one story featuring the very presentation I was attending. I peeked 2 rows ahead to find the social media “influencer” I have been following on Instagram for months in real life for the first time.
It is not just the younger population glued to their social media accounts. In fact, Facebook boasted a more than 80% increase in users 55 years and older between 2011 and 2014 and a 41% increase in users aged 35 to 54 years.2 In total, there were 3.2 billion social media users globally in 2018.3 With such a large portion of the population engaged in social media, it is no wonder that it has become a rapidly emerging presence within the field of dermatology.
#Ad
Social media has become a powerful marketing tool for the practicing dermatologist. In a recent survey, 41% of social media users reported that social media influenced their choice of a particular physician, facility, or medical practice.4 Corresponding to this behavior, dermatology practices also have used social media to educate patients on services offered, acquire new patients, engage existing patients, create brand loyalty, become a trusted source of medical information in a sea of digital misinformation, and facilitate positive word-of-mouth opportunities.5 In fact, 53% of physician practices in the United States operate a Facebook page.6 For these physicians, marketing through social media carries the advantages of low cost and rapid transmission of information to a wide audience.7 Furthermore, the development of business insights and statistics by some social media platforms, such as those available to users on business profiles on Instagram, enables practices and marketers to target their audiences and optimize reach.
#DermLife
The role of social media in dermatology extends far beyond marketing. Lifestyle blogs centered on daily life as a medical provider, even within the field of dermatology, are gaining popularity. Dermatology-centered lifestyle blogs often incorporate the root derm in their handle, enabling other users to identify the account holder and interact in meaningful ways. According to a post from one popular Instagram influencer Dr. Audrey Sue Cruz (@dr.audreyxsue), such profiles may serve to prevent burnout, provide a creative outlet, share life as a resident, develop a supportive community, provide mentorship, and spread inspiration.
#Hashtag
Another interesting utility of social media is the use of standardized hashtags to facilitate scientific and clinical dialogue among medical professionals. Standardization of hashtag ontology on Twitter and Instagram has been adopted by the urology and gastroenterology fields to filter out “noise” by individuals not intending to join academic discussion.8 In dermatology, standardized hashtags have not been adopted, to my knowledge; however, a search for esoteric dermatologic terms such as #dermatopathology or #mohssurgery directs users to specialty-specific discussions.
#DontFryDay
Another role of social media in dermatology is dissemination of information. One notable example is the reach on Twitter of the “Don’t Fry Day” campaign, an annual campaign by the National Council on Skin Cancer Prevention to promote sun safety awareness and sun protection behaviors. In a recent study by Nguyen et al,9 the hashtag #DontFryDay was tracked on Twitter to assess the reach of the campaign. They found that this campaign had an impressive reach of approximately 1200 contributors, resulting in more than 16.5 million impressions; 18 celebrities and verified individuals accounted for 8,735,549 impressions.9
Despite the large potential for dissemination of information on social media, in 2014 none of the top 10 dermatologic journals or professional dermatologic organizations maintained an Instagram account. Only one of the top 10 patient advocate groups related to dermatology conditions—the Melanoma Research Foundation—was found on Instagram as of 2014.10 Furthermore, none of the top 10 most popular dermatology journals, professional dermatology organizations, or dermatology-related patient advocate groups could be found on Tumblr as of 2014.11 Although some of the aforementioned organizations have since adopted social media accounts, such as Cutis and Dermatology News (@mdedgederm) on Instagram in 2018, these social media platforms remain largely untapped outlets for dissemination of information to the public by reputable sources.
#VerifyHealthcare
Although social media has offered many advantages to the field of dermatology, it also has brought about unique challenges such as blind authorship, lack of source citation, and presentation of opinion as fact.7 To compound the challenge, 90% of millennials aged 18 to 24 years reportedly trust health care information shared by others on social media.12 Do we, as dermatologists, have a duty to take to social media to provide reputable health information? In an effort to address this emerging problem, popular Instagram influencer Dr. Austin Chiang (@austinchiangmd) initiated the #VerifyHealthcare movement, which called for physicians active on social media to practice transparency by providing users with their credentials.13 The goal of the movement is to help users differentiate medical information disseminated by trained medical professionals from misinformation by disreputable sources.
Final Thoughts
Despite its shortcomings, the emerging roles of social media in dermatology have proven to be a prominent force here to stay, providing new and innovative opportunities to dermatologists for social networking, dissemination of health information, motivation and inspiration, and marketing.
- Warden C. 30 statistics on social media and healthcare. Referral MD website. https://getreferralmd.com/2017/01/30-facts-statistics-on-social-media-and-healthcare/. Accessed January 16, 2019.
- Saul DJ. 3 million teens leave Facebook in 3 years: the Facebook demographic report. ISL website. https://isl.co/2014/01/3-million-teens-leave-facebook-in-3-years-the-2014-facebook-demographic-report/. Published January 15, 2014. Accessed January 9, 2019.
- Chaffey D. Global social media research summary 2018. Smart Insights website. https://www.smartinsights.com/social-media-marketing/social-media-strategy/new-global-social-media-research/. Published November 23, 2018. Accessed January 3, 2019.
- Ottenhoff M. Infographic: rising use of social and mobile in healthcare. The Spark Report. December 17, 2012. http://thesparkreport.com/infographic-social-mobile-healthcare/. Accessed January 9, 2019.
- Benabio J. The value of social media for dermatologists. Cutis. 2013;91:269-270.
- The healthcare social media shakeup. CDW Healthcare website. http://www.cdwcommunit.com/resources/infographic/social-media/. Accessed January 9, 2019.
- Vance K, Howe W, Dellavelle RP. Social internet sites as a source of public health information. Dermatol Clin. 2009;27:133-136.
- Chang AL, Vartabedian B, Spiegel B. Harnessing the hashtag: a standard approach to GI dialogue on social media. Am J Gastroeneterol. 2016;111:1082-1084.
- Nguyen JL, Heckman C, Perna F. Analysis of the Twitter “Don’t Fry Day” campaign. JAMA Dermatol. 2018;154:961-962.
- Karimkhani C, Connett J, Boyars L, et al. Dermatology on Instagram. Dermatol Online J. 2014;20. pii:13030/qt71g178w9.
- Correnti C, Boyars L, Karimkhani C, et al. Dermatology on Tumblr. Dermatol Online J. 2014;20:22642.
- Honigman B. 24 outstanding statistics on how social media has impacted healthcare. Referral MD website. https://getreferralmd.com/2013/09/healthcare-social-media-statistics. Accessed January 16, 2019.
- Oliver E. #VerifyHealthcare campaign seeks to increase social media transparency—5 insights. Becker’s GI & Endoscopy website. https://www.beckersasc.com/gastroenterology-and-endoscopy/verifyhealthcare-campaign-seeks-to-increase-social-media-transparency-5-insights.html. Published September 24, 2018. Accessed January 16, 2019.
- Warden C. 30 statistics on social media and healthcare. Referral MD website. https://getreferralmd.com/2017/01/30-facts-statistics-on-social-media-and-healthcare/. Accessed January 16, 2019.
- Saul DJ. 3 million teens leave Facebook in 3 years: the Facebook demographic report. ISL website. https://isl.co/2014/01/3-million-teens-leave-facebook-in-3-years-the-2014-facebook-demographic-report/. Published January 15, 2014. Accessed January 9, 2019.
- Chaffey D. Global social media research summary 2018. Smart Insights website. https://www.smartinsights.com/social-media-marketing/social-media-strategy/new-global-social-media-research/. Published November 23, 2018. Accessed January 3, 2019.
- Ottenhoff M. Infographic: rising use of social and mobile in healthcare. The Spark Report. December 17, 2012. http://thesparkreport.com/infographic-social-mobile-healthcare/. Accessed January 9, 2019.
- Benabio J. The value of social media for dermatologists. Cutis. 2013;91:269-270.
- The healthcare social media shakeup. CDW Healthcare website. http://www.cdwcommunit.com/resources/infographic/social-media/. Accessed January 9, 2019.
- Vance K, Howe W, Dellavelle RP. Social internet sites as a source of public health information. Dermatol Clin. 2009;27:133-136.
- Chang AL, Vartabedian B, Spiegel B. Harnessing the hashtag: a standard approach to GI dialogue on social media. Am J Gastroeneterol. 2016;111:1082-1084.
- Nguyen JL, Heckman C, Perna F. Analysis of the Twitter “Don’t Fry Day” campaign. JAMA Dermatol. 2018;154:961-962.
- Karimkhani C, Connett J, Boyars L, et al. Dermatology on Instagram. Dermatol Online J. 2014;20. pii:13030/qt71g178w9.
- Correnti C, Boyars L, Karimkhani C, et al. Dermatology on Tumblr. Dermatol Online J. 2014;20:22642.
- Honigman B. 24 outstanding statistics on how social media has impacted healthcare. Referral MD website. https://getreferralmd.com/2013/09/healthcare-social-media-statistics. Accessed January 16, 2019.
- Oliver E. #VerifyHealthcare campaign seeks to increase social media transparency—5 insights. Becker’s GI & Endoscopy website. https://www.beckersasc.com/gastroenterology-and-endoscopy/verifyhealthcare-campaign-seeks-to-increase-social-media-transparency-5-insights.html. Published September 24, 2018. Accessed January 16, 2019.
Resident Pearl
- The emerging presence of social media in dermatology provides opportunities for dermatologists to participate in dissemination and consumption of reliable health information, marketing, social networking among colleagues, and motivation and inspiration. It has been proposed that participation may serve to prevent resident burnout.
Getting serious about post-EVAR aortic neck dilation
CHICAGO – Clinically significant aortic neck dilation occurs in one-quarter of patients after endovascular abdominal aortic aneurysm repair and is associated with sharply increased risk for type Ia endoleak, stent migration at the proximal seal zone, continued aneurysmal sack enlargement, and even aneurysm rupture, Jason T. Lee, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
He cited what he considers to be the best-quality systematic review of the literature regarding proximal neck dilation after endovascular aneurysm repair (EVAR), which was conducted by investigators at Paracelsus Medical University in Nuremberg, Germany. The data, he said, speak to the importance of trying to minimize the chances of aortic neck dilation (AND).
The review, which included 26 published studies through 2015 and nearly 10,000 EVAR patients, concluded that AND occurred in 24.6% of these patients. The investigators defined AND as more than 3 mm of dilation, which with an average neck diameter of about 30 mm in all comers, would represent a 10% aortic neck-diameter expansion. The incidence of the composite adverse outcome of type I endoleak, stent migration, and reintervention during a maximum of 9 years of follow-up was 29-fold greater in the AND group than in AND-free patients (J Endovasc Ther. 2017;24[1]:59-67).
Continuing controversy exists regarding the extent to which post-EVAR AND is a manifestation of the underlying aneurysmal process as opposed to an adverse effect caused by the outward radial force applied by the stent. That being said, there are several factors related to AND that are potentially within the vascular interventionalist’s control. Topping the list is aggressive oversizing of self-expanding stent grafts in an effort to obtain an excellent seal, according to Dr. Lee, a professor of surgery at Stanford (Calif.) University.
“The data from multiple series definitely suggests that more than 20% oversizing is correlated with a higher incidence of AND. So maybe the answer is oversizing by more like 10%-15%,” the vascular surgeon said.
Another factor contributing to AND is the overwhelming popularity of self-expanding aortic stent grafts, now utilized in 95% of all EVARS done in the United States. Balloon-expandable stent grafts require much less oversizing – less than 5% – to maintain their diameter post deployment, but they are rarely used. Indeed, the only commercially available balloon-expandable device in the United States is the TriVascular Ovation stent system, which seals the stent graft to the wall of the aorta via polyethylene glycol rather than by radial force.
Intriguingly, Italian investigators have reported that, in a multicenter series of 161 patients who underwent EVAR with the Ovation endograft, no AND occurred at CT scanning done after a minimum of 24 months of follow-up (J Vasc Surg. 2016;63[1]:8-15).
Previous studies of the implications of AND after EVAR have been limited to comparisons of early-generation devices, so Dr. Lee and his coinvestigators conducted a retrospective review of a prospective Stanford database that included 86 patients who underwent elective, uncomplicated infrarenal endovascular abdominal aortic aneurysm repairs using a variety of contemporary stent grafts. Eighty-six percent of patients experienced AND during a median radiologic follow-up of 21.9 months, with a mean 1.3-mm increase at 30 days and 3.3 mm at most recent follow-up. The degree of AND correlated with the amount of oversizing. However, AND didn’t vary significantly by device type, which included the Cook Zenith, Gore Excluder, Medtronic Endurant, and Endologix Powerlink self-expanding stent grafts (Ann Vasc Surg. 2017;43:115-20).
Aortic anatomy also plays a role in AND. Shorter necks with severe angulation have been found to pose a higher risk. Open repair, which entails much less AND than does EVAR, may make more sense in that challenging anatomic situation, according to Dr. Lee.
He reported current research funding from Cook, Gore, and Medtronic.
CHICAGO – Clinically significant aortic neck dilation occurs in one-quarter of patients after endovascular abdominal aortic aneurysm repair and is associated with sharply increased risk for type Ia endoleak, stent migration at the proximal seal zone, continued aneurysmal sack enlargement, and even aneurysm rupture, Jason T. Lee, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
He cited what he considers to be the best-quality systematic review of the literature regarding proximal neck dilation after endovascular aneurysm repair (EVAR), which was conducted by investigators at Paracelsus Medical University in Nuremberg, Germany. The data, he said, speak to the importance of trying to minimize the chances of aortic neck dilation (AND).
The review, which included 26 published studies through 2015 and nearly 10,000 EVAR patients, concluded that AND occurred in 24.6% of these patients. The investigators defined AND as more than 3 mm of dilation, which with an average neck diameter of about 30 mm in all comers, would represent a 10% aortic neck-diameter expansion. The incidence of the composite adverse outcome of type I endoleak, stent migration, and reintervention during a maximum of 9 years of follow-up was 29-fold greater in the AND group than in AND-free patients (J Endovasc Ther. 2017;24[1]:59-67).
Continuing controversy exists regarding the extent to which post-EVAR AND is a manifestation of the underlying aneurysmal process as opposed to an adverse effect caused by the outward radial force applied by the stent. That being said, there are several factors related to AND that are potentially within the vascular interventionalist’s control. Topping the list is aggressive oversizing of self-expanding stent grafts in an effort to obtain an excellent seal, according to Dr. Lee, a professor of surgery at Stanford (Calif.) University.
“The data from multiple series definitely suggests that more than 20% oversizing is correlated with a higher incidence of AND. So maybe the answer is oversizing by more like 10%-15%,” the vascular surgeon said.
Another factor contributing to AND is the overwhelming popularity of self-expanding aortic stent grafts, now utilized in 95% of all EVARS done in the United States. Balloon-expandable stent grafts require much less oversizing – less than 5% – to maintain their diameter post deployment, but they are rarely used. Indeed, the only commercially available balloon-expandable device in the United States is the TriVascular Ovation stent system, which seals the stent graft to the wall of the aorta via polyethylene glycol rather than by radial force.
Intriguingly, Italian investigators have reported that, in a multicenter series of 161 patients who underwent EVAR with the Ovation endograft, no AND occurred at CT scanning done after a minimum of 24 months of follow-up (J Vasc Surg. 2016;63[1]:8-15).
Previous studies of the implications of AND after EVAR have been limited to comparisons of early-generation devices, so Dr. Lee and his coinvestigators conducted a retrospective review of a prospective Stanford database that included 86 patients who underwent elective, uncomplicated infrarenal endovascular abdominal aortic aneurysm repairs using a variety of contemporary stent grafts. Eighty-six percent of patients experienced AND during a median radiologic follow-up of 21.9 months, with a mean 1.3-mm increase at 30 days and 3.3 mm at most recent follow-up. The degree of AND correlated with the amount of oversizing. However, AND didn’t vary significantly by device type, which included the Cook Zenith, Gore Excluder, Medtronic Endurant, and Endologix Powerlink self-expanding stent grafts (Ann Vasc Surg. 2017;43:115-20).
Aortic anatomy also plays a role in AND. Shorter necks with severe angulation have been found to pose a higher risk. Open repair, which entails much less AND than does EVAR, may make more sense in that challenging anatomic situation, according to Dr. Lee.
He reported current research funding from Cook, Gore, and Medtronic.
CHICAGO – Clinically significant aortic neck dilation occurs in one-quarter of patients after endovascular abdominal aortic aneurysm repair and is associated with sharply increased risk for type Ia endoleak, stent migration at the proximal seal zone, continued aneurysmal sack enlargement, and even aneurysm rupture, Jason T. Lee, MD, said at a symposium on vascular surgery sponsored by Northwestern University.
He cited what he considers to be the best-quality systematic review of the literature regarding proximal neck dilation after endovascular aneurysm repair (EVAR), which was conducted by investigators at Paracelsus Medical University in Nuremberg, Germany. The data, he said, speak to the importance of trying to minimize the chances of aortic neck dilation (AND).
The review, which included 26 published studies through 2015 and nearly 10,000 EVAR patients, concluded that AND occurred in 24.6% of these patients. The investigators defined AND as more than 3 mm of dilation, which with an average neck diameter of about 30 mm in all comers, would represent a 10% aortic neck-diameter expansion. The incidence of the composite adverse outcome of type I endoleak, stent migration, and reintervention during a maximum of 9 years of follow-up was 29-fold greater in the AND group than in AND-free patients (J Endovasc Ther. 2017;24[1]:59-67).
Continuing controversy exists regarding the extent to which post-EVAR AND is a manifestation of the underlying aneurysmal process as opposed to an adverse effect caused by the outward radial force applied by the stent. That being said, there are several factors related to AND that are potentially within the vascular interventionalist’s control. Topping the list is aggressive oversizing of self-expanding stent grafts in an effort to obtain an excellent seal, according to Dr. Lee, a professor of surgery at Stanford (Calif.) University.
“The data from multiple series definitely suggests that more than 20% oversizing is correlated with a higher incidence of AND. So maybe the answer is oversizing by more like 10%-15%,” the vascular surgeon said.
Another factor contributing to AND is the overwhelming popularity of self-expanding aortic stent grafts, now utilized in 95% of all EVARS done in the United States. Balloon-expandable stent grafts require much less oversizing – less than 5% – to maintain their diameter post deployment, but they are rarely used. Indeed, the only commercially available balloon-expandable device in the United States is the TriVascular Ovation stent system, which seals the stent graft to the wall of the aorta via polyethylene glycol rather than by radial force.
Intriguingly, Italian investigators have reported that, in a multicenter series of 161 patients who underwent EVAR with the Ovation endograft, no AND occurred at CT scanning done after a minimum of 24 months of follow-up (J Vasc Surg. 2016;63[1]:8-15).
Previous studies of the implications of AND after EVAR have been limited to comparisons of early-generation devices, so Dr. Lee and his coinvestigators conducted a retrospective review of a prospective Stanford database that included 86 patients who underwent elective, uncomplicated infrarenal endovascular abdominal aortic aneurysm repairs using a variety of contemporary stent grafts. Eighty-six percent of patients experienced AND during a median radiologic follow-up of 21.9 months, with a mean 1.3-mm increase at 30 days and 3.3 mm at most recent follow-up. The degree of AND correlated with the amount of oversizing. However, AND didn’t vary significantly by device type, which included the Cook Zenith, Gore Excluder, Medtronic Endurant, and Endologix Powerlink self-expanding stent grafts (Ann Vasc Surg. 2017;43:115-20).
Aortic anatomy also plays a role in AND. Shorter necks with severe angulation have been found to pose a higher risk. Open repair, which entails much less AND than does EVAR, may make more sense in that challenging anatomic situation, according to Dr. Lee.
He reported current research funding from Cook, Gore, and Medtronic.
EXPERT ANALYSIS FROM THE NORTHWESTERN VASCULAR SYMPOSIUM
MMF proves viable as alternative option in moderate ANCA-associated vasculitis
Mycophenolate mofetil was noninferior to pulsed cyclophosphamide for remission induction in ANCA-associated vasculitis, but there were more relapses, in a randomized, unblinded trial of 140 patients with non–life-threatening disease.
“Our results demonstrate that MMF [mycophenolate mofetil] represents an alternative to CYC [cyclophosphamide] for remission induction in AAV [ANCA-associated vasculitis],” said investigators led by Rachel B. Jones, MD, of the department of renal medicine at Addenbrooke’s Hospital, Cambridge, England.
“While treatment with MMF may be associated with a higher risk of relapse compared with pulsed CYC, this increased risk may be acceptable to avoid the potential adverse effects of CYC” – infections, malignancies, and infertility – “particularly when the baseline risk of relapse is low,” as with the elderly or myeloperoxidase (MPO)-ANCA disease, “or if rituximab is unavailable,” Dr. Jones and her colleagues wrote in Annals of the Rheumatic Diseases.
Since enrollment began in 2007, they also noted that “it has become common to use rituximab as an alternative to CYC induction therapy ... However, rituximab is expensive, and its use is restricted in many countries ... Alternative effective low-cost induction therapies” – such as MMF – “may be required in some cases.”
The work, which was done on behalf of the European Vasculitis Study Group, was the largest randomized trial to date of MMF for AAV remission induction, and the first to include children; patients were enrolled at 21 sites in Europe, Australia, and New Zealand.
They were randomly assigned to MMF or pulsed CYC, with 66 adults and 4 children ranging in age from 10 to 16 years in each group. In addition to those with life-threatening disease, patients younger than 6 years old and those on dialysis or with an estimated glomerular filtration rate below 15 mL/min/m2 were excluded. Following remission, subjects were switched to oral azathioprine and a prednisone taper.
The primary outcome was remission by 6 months, defined as the absence of disease activity on two consecutive occasions at least 1 month apart, plus adherence to the taper. Baseline ANCA subtype, disease activity, and organ involvement were similar between the groups.
Overall, 47 patients in the in the MMF group (67%), including 1 child, reached the primary endpoint, versus 43 patients (61%), again including 1 child, in the CYC group (risk difference = 5.7%; 90% confidence interval, –7.5% to 19%), which established noninferiority. The median time to remission was about 90 days in both arms. “Compliance was a contributory factor to the lower remission rate[s] in children,” the authors noted.
More relapses occurred in the MMF group (33% vs. 19% with CYC). Among MPO-ANCA patients, the relapse rate was 15% versus 12% with CYC. In proteinase 3 (PR3)-ANCA patients, almost half in the MMF group relapsed (48% versus 24%).
There were no significant differences in serious infections (26% with MMF versus 17% with CYC; P = .3), malignancies, thromboembolisms, or death (four CYC patients and five MMF patients at 6 months).
Adult patients in the MMF group received MMF 2 g/day, with dose increases to 3 g/day permitted for uncontrolled disease at 4 weeks. Patients younger than 17 years old were dosed according to body surface area. The CYC group received intravenous pulsed CYC 15 mg/kg every 2-3 weeks with reductions for age and renal function. A total of 58 CYC patients (83%) received at least six pulses.
After remission, azathioprine was dosed at 2 mg/kg per day. Oral prednisolone started at 1 mg/kg per day and was reduced to 5 mg/day by 6 months.
Plasma exchange or additional methylprednisolone were allowed at entry; there were no differences in their use between the groups.
The trial was sponsored by Cambridge University Hospitals NHS Foundation Trust. Vifor Pharma provided a research grant to cover the trial and MMF costs. Dr. Jones and the other investigators reported ties to several companies, including GlaxoSmithKline, Genentech/Roche, ChemoCentryx, and Genzyme/Sanofi. Vifor holds an equity stake in ChemoCentryx.
SOURCE: Jones RB et al. Ann Rheum Dis. 2019 Jan 5. doi: 10.1136/annrheumdis-2018-214245.
We sometimes encounter patients for whom access to rituximab is a problem, for financial, logistical, or tolerability reasons. Also, some patients are really reluctant about an infusion, and there is still some skittishness about rituximab. I have had some patients who, despite my having strongly reiterated how rare progressive multifocal leukoencephalopathy is, have been unable to get past the fact that that’s in the package insert. It’s helpful to me as a clinician to know that mycophenolate is something we can turn to in these situations. As I discuss the ups and down of different potential strategies with patients, this is something that can be part of the discussion.
This was a really well done study, and they were honest about the limitations. They excluded patients with the most severe disease, so you are still not talking about your patients on dialysis getting mycophenolate. Also, there are questions in terms of it’s not being as effective at sustaining remission, but that’s a bridge you can cross once they are in remission, in terms of how vigilant you are about follow-up. You also can re-induce remission with mycophenolate if you have to.
Robert F. Spiera, MD, is director of the vasculitis and scleroderma program at the Hospital for Special Surgery in New York. He had no relevant disclosures, and is on the editorial advisory board of MDedge Rheumatology.
We sometimes encounter patients for whom access to rituximab is a problem, for financial, logistical, or tolerability reasons. Also, some patients are really reluctant about an infusion, and there is still some skittishness about rituximab. I have had some patients who, despite my having strongly reiterated how rare progressive multifocal leukoencephalopathy is, have been unable to get past the fact that that’s in the package insert. It’s helpful to me as a clinician to know that mycophenolate is something we can turn to in these situations. As I discuss the ups and down of different potential strategies with patients, this is something that can be part of the discussion.
This was a really well done study, and they were honest about the limitations. They excluded patients with the most severe disease, so you are still not talking about your patients on dialysis getting mycophenolate. Also, there are questions in terms of it’s not being as effective at sustaining remission, but that’s a bridge you can cross once they are in remission, in terms of how vigilant you are about follow-up. You also can re-induce remission with mycophenolate if you have to.
Robert F. Spiera, MD, is director of the vasculitis and scleroderma program at the Hospital for Special Surgery in New York. He had no relevant disclosures, and is on the editorial advisory board of MDedge Rheumatology.
We sometimes encounter patients for whom access to rituximab is a problem, for financial, logistical, or tolerability reasons. Also, some patients are really reluctant about an infusion, and there is still some skittishness about rituximab. I have had some patients who, despite my having strongly reiterated how rare progressive multifocal leukoencephalopathy is, have been unable to get past the fact that that’s in the package insert. It’s helpful to me as a clinician to know that mycophenolate is something we can turn to in these situations. As I discuss the ups and down of different potential strategies with patients, this is something that can be part of the discussion.
This was a really well done study, and they were honest about the limitations. They excluded patients with the most severe disease, so you are still not talking about your patients on dialysis getting mycophenolate. Also, there are questions in terms of it’s not being as effective at sustaining remission, but that’s a bridge you can cross once they are in remission, in terms of how vigilant you are about follow-up. You also can re-induce remission with mycophenolate if you have to.
Robert F. Spiera, MD, is director of the vasculitis and scleroderma program at the Hospital for Special Surgery in New York. He had no relevant disclosures, and is on the editorial advisory board of MDedge Rheumatology.
Mycophenolate mofetil was noninferior to pulsed cyclophosphamide for remission induction in ANCA-associated vasculitis, but there were more relapses, in a randomized, unblinded trial of 140 patients with non–life-threatening disease.
“Our results demonstrate that MMF [mycophenolate mofetil] represents an alternative to CYC [cyclophosphamide] for remission induction in AAV [ANCA-associated vasculitis],” said investigators led by Rachel B. Jones, MD, of the department of renal medicine at Addenbrooke’s Hospital, Cambridge, England.
“While treatment with MMF may be associated with a higher risk of relapse compared with pulsed CYC, this increased risk may be acceptable to avoid the potential adverse effects of CYC” – infections, malignancies, and infertility – “particularly when the baseline risk of relapse is low,” as with the elderly or myeloperoxidase (MPO)-ANCA disease, “or if rituximab is unavailable,” Dr. Jones and her colleagues wrote in Annals of the Rheumatic Diseases.
Since enrollment began in 2007, they also noted that “it has become common to use rituximab as an alternative to CYC induction therapy ... However, rituximab is expensive, and its use is restricted in many countries ... Alternative effective low-cost induction therapies” – such as MMF – “may be required in some cases.”
The work, which was done on behalf of the European Vasculitis Study Group, was the largest randomized trial to date of MMF for AAV remission induction, and the first to include children; patients were enrolled at 21 sites in Europe, Australia, and New Zealand.
They were randomly assigned to MMF or pulsed CYC, with 66 adults and 4 children ranging in age from 10 to 16 years in each group. In addition to those with life-threatening disease, patients younger than 6 years old and those on dialysis or with an estimated glomerular filtration rate below 15 mL/min/m2 were excluded. Following remission, subjects were switched to oral azathioprine and a prednisone taper.
The primary outcome was remission by 6 months, defined as the absence of disease activity on two consecutive occasions at least 1 month apart, plus adherence to the taper. Baseline ANCA subtype, disease activity, and organ involvement were similar between the groups.
Overall, 47 patients in the in the MMF group (67%), including 1 child, reached the primary endpoint, versus 43 patients (61%), again including 1 child, in the CYC group (risk difference = 5.7%; 90% confidence interval, –7.5% to 19%), which established noninferiority. The median time to remission was about 90 days in both arms. “Compliance was a contributory factor to the lower remission rate[s] in children,” the authors noted.
More relapses occurred in the MMF group (33% vs. 19% with CYC). Among MPO-ANCA patients, the relapse rate was 15% versus 12% with CYC. In proteinase 3 (PR3)-ANCA patients, almost half in the MMF group relapsed (48% versus 24%).
There were no significant differences in serious infections (26% with MMF versus 17% with CYC; P = .3), malignancies, thromboembolisms, or death (four CYC patients and five MMF patients at 6 months).
Adult patients in the MMF group received MMF 2 g/day, with dose increases to 3 g/day permitted for uncontrolled disease at 4 weeks. Patients younger than 17 years old were dosed according to body surface area. The CYC group received intravenous pulsed CYC 15 mg/kg every 2-3 weeks with reductions for age and renal function. A total of 58 CYC patients (83%) received at least six pulses.
After remission, azathioprine was dosed at 2 mg/kg per day. Oral prednisolone started at 1 mg/kg per day and was reduced to 5 mg/day by 6 months.
Plasma exchange or additional methylprednisolone were allowed at entry; there were no differences in their use between the groups.
The trial was sponsored by Cambridge University Hospitals NHS Foundation Trust. Vifor Pharma provided a research grant to cover the trial and MMF costs. Dr. Jones and the other investigators reported ties to several companies, including GlaxoSmithKline, Genentech/Roche, ChemoCentryx, and Genzyme/Sanofi. Vifor holds an equity stake in ChemoCentryx.
SOURCE: Jones RB et al. Ann Rheum Dis. 2019 Jan 5. doi: 10.1136/annrheumdis-2018-214245.
Mycophenolate mofetil was noninferior to pulsed cyclophosphamide for remission induction in ANCA-associated vasculitis, but there were more relapses, in a randomized, unblinded trial of 140 patients with non–life-threatening disease.
“Our results demonstrate that MMF [mycophenolate mofetil] represents an alternative to CYC [cyclophosphamide] for remission induction in AAV [ANCA-associated vasculitis],” said investigators led by Rachel B. Jones, MD, of the department of renal medicine at Addenbrooke’s Hospital, Cambridge, England.
“While treatment with MMF may be associated with a higher risk of relapse compared with pulsed CYC, this increased risk may be acceptable to avoid the potential adverse effects of CYC” – infections, malignancies, and infertility – “particularly when the baseline risk of relapse is low,” as with the elderly or myeloperoxidase (MPO)-ANCA disease, “or if rituximab is unavailable,” Dr. Jones and her colleagues wrote in Annals of the Rheumatic Diseases.
Since enrollment began in 2007, they also noted that “it has become common to use rituximab as an alternative to CYC induction therapy ... However, rituximab is expensive, and its use is restricted in many countries ... Alternative effective low-cost induction therapies” – such as MMF – “may be required in some cases.”
The work, which was done on behalf of the European Vasculitis Study Group, was the largest randomized trial to date of MMF for AAV remission induction, and the first to include children; patients were enrolled at 21 sites in Europe, Australia, and New Zealand.
They were randomly assigned to MMF or pulsed CYC, with 66 adults and 4 children ranging in age from 10 to 16 years in each group. In addition to those with life-threatening disease, patients younger than 6 years old and those on dialysis or with an estimated glomerular filtration rate below 15 mL/min/m2 were excluded. Following remission, subjects were switched to oral azathioprine and a prednisone taper.
The primary outcome was remission by 6 months, defined as the absence of disease activity on two consecutive occasions at least 1 month apart, plus adherence to the taper. Baseline ANCA subtype, disease activity, and organ involvement were similar between the groups.
Overall, 47 patients in the in the MMF group (67%), including 1 child, reached the primary endpoint, versus 43 patients (61%), again including 1 child, in the CYC group (risk difference = 5.7%; 90% confidence interval, –7.5% to 19%), which established noninferiority. The median time to remission was about 90 days in both arms. “Compliance was a contributory factor to the lower remission rate[s] in children,” the authors noted.
More relapses occurred in the MMF group (33% vs. 19% with CYC). Among MPO-ANCA patients, the relapse rate was 15% versus 12% with CYC. In proteinase 3 (PR3)-ANCA patients, almost half in the MMF group relapsed (48% versus 24%).
There were no significant differences in serious infections (26% with MMF versus 17% with CYC; P = .3), malignancies, thromboembolisms, or death (four CYC patients and five MMF patients at 6 months).
Adult patients in the MMF group received MMF 2 g/day, with dose increases to 3 g/day permitted for uncontrolled disease at 4 weeks. Patients younger than 17 years old were dosed according to body surface area. The CYC group received intravenous pulsed CYC 15 mg/kg every 2-3 weeks with reductions for age and renal function. A total of 58 CYC patients (83%) received at least six pulses.
After remission, azathioprine was dosed at 2 mg/kg per day. Oral prednisolone started at 1 mg/kg per day and was reduced to 5 mg/day by 6 months.
Plasma exchange or additional methylprednisolone were allowed at entry; there were no differences in their use between the groups.
The trial was sponsored by Cambridge University Hospitals NHS Foundation Trust. Vifor Pharma provided a research grant to cover the trial and MMF costs. Dr. Jones and the other investigators reported ties to several companies, including GlaxoSmithKline, Genentech/Roche, ChemoCentryx, and Genzyme/Sanofi. Vifor holds an equity stake in ChemoCentryx.
SOURCE: Jones RB et al. Ann Rheum Dis. 2019 Jan 5. doi: 10.1136/annrheumdis-2018-214245.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point:
Major finding: Overall, 47 patients in the in the MMF group (67%), including 1 child, reached remission by 6 months, versus 43 subjects (61%), again including 1 child, in the CYC group (risk difference = 5.7%; 90% CI, –7.5% to 19%), which established noninferiority.
Study details: Randomized, multisite trial with 140 patients.
Disclosures: The Cambridge University Hospitals NHS Foundation Trust sponsored the study. Vifor Pharma provided a research grant to cover the trial and MMF costs. Dr. Jones and the other investigators reported ties to several companies, including GlaxoSmithKline, Genentech/Roche, and ChemoCentryx, which is owned in part by Vifor.
Source: Jones RB et al. Ann Rheum Dis. 2019 Jan 5. doi: 10.1136/annrheumdis-2018-214245.
Carbs vs. fats for CVD
Also today, from MDedge Pediatrics, a consult on influenza and pneumonia, brodalumab beats ustekinumab to PASI 100, and a novel agent cuts LDL in patients who are intolerant to statins.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, from MDedge Pediatrics, a consult on influenza and pneumonia, brodalumab beats ustekinumab to PASI 100, and a novel agent cuts LDL in patients who are intolerant to statins.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, from MDedge Pediatrics, a consult on influenza and pneumonia, brodalumab beats ustekinumab to PASI 100, and a novel agent cuts LDL in patients who are intolerant to statins.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Family estrangement: Would mutual respect make a difference?
Families are the bedrock of the lives of many, but some people opt to separate permanently from family members. A recent episode of the NPR program “1A” explored why families sever ties.
For journalist Harriet Brown, author of “Shadow Daughter: A Memoir of Estrangement,” (Da Capo Press, 2018), the decision to end her relationship with her mother came after she blamed Ms. Brown in “a blistering email” for the relapsed anorexia of Ms. Brown’s daughter.
“I was done with her,” Ms. Brown said on the program. “I told her I was done. That was it. And I never talked to her again. I think for both of us it felt final in some way.”
She said a lot of shame and stigma comes from having a bad relationship with a parent. “I really wanted to make it clear that ... sometimes walking away is really the best thing to do.”
Kristina M. Scharp, PhD, who has studied the estrangement phenomenon, said no national data exist on family estrangement. “About 12% of mothers and research would suggest even more fathers would report being estranged from one of their children,” said Dr. Scharp, who is with the University of Washington, Seattle. “It’s fairly common.”
Estrangement might be more common today because times have changed, said Joshua Coleman, PhD, author of “When Parents Hurt: Compassionate Strategies When you and Your Grown Child Don’t get Along” (HarperCollins, 2008). “Today’s adult children don’t view their relationships with their parents the way their folks did with their parents … the principles of obligation, duty, and respect that baby boomers and generations before them had for their elders aren’t necessarily there anymore,” Dr. Coleman said in a previous interview with the Chicago Tribune.
For her part, Ms. Brown said, the relationship with her mother – who is now deceased – might have been healed with respect. she said. “Honestly, it was that basic with my mother.”
Phelps gets mental health advocacy award
In the pool, Michael Phelps was golden, with 28 Olympics medals, 23 of them gold, hanging around his neck by the end of his swimming career. But the release from the water to real life after the 2016 Summer Olympics left no outlet for troubles that had dogged him for years. Drunk driving convictions and a ban from competition during his competitive years had failed to stop his downward spiral of depression. His thoughts turned to suicide.
But his story has a bright ending. With his realization that he had hit rock bottom, and with the help of his wife and therapists, he accepted his depression and learned to live with the reality that his life is, for the most part, pretty good.
His openness about his struggles with depression has been influential. The latest recognition came in early January, when he received the Morton E. Ruderman Award in Inclusion from the Ruderman Family Foundation. As reported in the Boston Globe, Mr. Phelps was recognized for his advocacy for people with disabilities and “his own journey with mental health.”
“I do like who I am and I’m comfortable with who I am. I couldn’t say that a few years ago. So I’m in a very good place and just living life one day at a time,” Mr. Phelps remarked in an interview with CNN last year.
Mr. Phelps is now a paid spokesperson for TalkSpace, an online therapy company.
“I’d like to make a difference. I’d like to be able to save a life if I can. You know, for me that’s more important than winning a gold medal. The stuff that I’m doing now is very exciting. It’s hard, it’s challenging but it’s fun for me. That’s what drives me to get out of bed every morning,” he said.
Offenders with mental illness get a break
A law included as part of budget legislation that was signed by then-Gov. Jerry Brown of California in June 2018 has offered people with mental health troubles who have been charged with a crime the opportunity for treatment instead of jail time.
As reported in the Los Angeles Times, the law provides judges with the discretion to order offenders into treatment rather than sentencing them. Success in treatment can lead to charges being dropped.
The law has been praised by some mental health advocates but panned by law enforcement officials and prosecutors with a harder view of criminal justice. The opposition stems largely from a December 2018 ruling by the 4th District Court of Appeal that the law could be applied to retroactively address the case of a man imprisoned for 29 years in 2017 for multiple felony charges that included domestic violence and assault.
The law does not extend to those charged with murder, manslaughter, rape, and child sexual abuse. To date, citing mental illness in seeking diversion of sentencing has not proven successful in most cases.
Schools get mental health allocation
The Orleans Parish School Board, which serves all of New Orleans, will allocate $1.3 million to the Center for Resilience, a local mental health day treatment program, beginning this year. The new program will expand mental health help to children in grades 9-12, according to a report in the Times-Picayune.
The funding will enable the center to expand an existing program that helps students with behavioral issues. Such help is not available in the traditional school system. By offsetting part of the price tag for the mental health care, the initiative “[helps to make] this critical service more available for our students most in need,” said Dominique Ellis, a spokesperson for the school board. “Mental health day treatment programs like the Center for Resilience typically cost between the ranges of $80,000-$100,000 per student to operate effectively.”
The development brings New Orleans level with money spent on similar programs in other school boards nationwide. It’s a service that is sorely needed. According to statistics supplied by the Center for Resilience, 60% of New Orleans children suffer from PTSD and are 4.5 times more likely to suffer from hyperactivity, aggression, and social withdrawal than similarly aged children elsewhere in the United States.
Bill addressing opioid crisis a “no-brainer”
Legislation put forward in the current session of the Texas Legislature would require pharmacists to identify all prescription opioids with a red cap and a hard-to-miss warning of the addictive risk of the medications. In addition, pharmacists would have to explain the risk in person to those receiving the medications and get signed acknowledgment of the conversation before dispensing the drugs.
As described in an article in the Austin American-Stateman, nearly 3,000 people in Texas died from drug overdoses in 2017. The deaths tied to opioid overdoses are not certain, but other data from 2015 suggest that one-third is a reasonable estimate.
“Losing a loved one to an opioid overdose is a tragedy that far too many Texas families experience,” said Rep. Shawn Thierry last month, when she introduced the three bills. “These distinctive red caps will serve as a clear notice to Texans that opioids are unlike milder forms of prescription pain relievers and have life-altering risks that must be considered before taking them.”
Warning labels on everything from food to tobacco products, and including them on prescription opioids is a “no-brainer,” Ms. Thierry said. “The more we can educate our residents the less likely they will be to misuse these medications.”
A report issued in November 2018 chronicled the drug crisis in Texas and offered recommendations. Many of the 100 recommendations involved the absence of treatment resources in the state.
Families are the bedrock of the lives of many, but some people opt to separate permanently from family members. A recent episode of the NPR program “1A” explored why families sever ties.
For journalist Harriet Brown, author of “Shadow Daughter: A Memoir of Estrangement,” (Da Capo Press, 2018), the decision to end her relationship with her mother came after she blamed Ms. Brown in “a blistering email” for the relapsed anorexia of Ms. Brown’s daughter.
“I was done with her,” Ms. Brown said on the program. “I told her I was done. That was it. And I never talked to her again. I think for both of us it felt final in some way.”
She said a lot of shame and stigma comes from having a bad relationship with a parent. “I really wanted to make it clear that ... sometimes walking away is really the best thing to do.”
Kristina M. Scharp, PhD, who has studied the estrangement phenomenon, said no national data exist on family estrangement. “About 12% of mothers and research would suggest even more fathers would report being estranged from one of their children,” said Dr. Scharp, who is with the University of Washington, Seattle. “It’s fairly common.”
Estrangement might be more common today because times have changed, said Joshua Coleman, PhD, author of “When Parents Hurt: Compassionate Strategies When you and Your Grown Child Don’t get Along” (HarperCollins, 2008). “Today’s adult children don’t view their relationships with their parents the way their folks did with their parents … the principles of obligation, duty, and respect that baby boomers and generations before them had for their elders aren’t necessarily there anymore,” Dr. Coleman said in a previous interview with the Chicago Tribune.
For her part, Ms. Brown said, the relationship with her mother – who is now deceased – might have been healed with respect. she said. “Honestly, it was that basic with my mother.”
Phelps gets mental health advocacy award
In the pool, Michael Phelps was golden, with 28 Olympics medals, 23 of them gold, hanging around his neck by the end of his swimming career. But the release from the water to real life after the 2016 Summer Olympics left no outlet for troubles that had dogged him for years. Drunk driving convictions and a ban from competition during his competitive years had failed to stop his downward spiral of depression. His thoughts turned to suicide.
But his story has a bright ending. With his realization that he had hit rock bottom, and with the help of his wife and therapists, he accepted his depression and learned to live with the reality that his life is, for the most part, pretty good.
His openness about his struggles with depression has been influential. The latest recognition came in early January, when he received the Morton E. Ruderman Award in Inclusion from the Ruderman Family Foundation. As reported in the Boston Globe, Mr. Phelps was recognized for his advocacy for people with disabilities and “his own journey with mental health.”
“I do like who I am and I’m comfortable with who I am. I couldn’t say that a few years ago. So I’m in a very good place and just living life one day at a time,” Mr. Phelps remarked in an interview with CNN last year.
Mr. Phelps is now a paid spokesperson for TalkSpace, an online therapy company.
“I’d like to make a difference. I’d like to be able to save a life if I can. You know, for me that’s more important than winning a gold medal. The stuff that I’m doing now is very exciting. It’s hard, it’s challenging but it’s fun for me. That’s what drives me to get out of bed every morning,” he said.
Offenders with mental illness get a break
A law included as part of budget legislation that was signed by then-Gov. Jerry Brown of California in June 2018 has offered people with mental health troubles who have been charged with a crime the opportunity for treatment instead of jail time.
As reported in the Los Angeles Times, the law provides judges with the discretion to order offenders into treatment rather than sentencing them. Success in treatment can lead to charges being dropped.
The law has been praised by some mental health advocates but panned by law enforcement officials and prosecutors with a harder view of criminal justice. The opposition stems largely from a December 2018 ruling by the 4th District Court of Appeal that the law could be applied to retroactively address the case of a man imprisoned for 29 years in 2017 for multiple felony charges that included domestic violence and assault.
The law does not extend to those charged with murder, manslaughter, rape, and child sexual abuse. To date, citing mental illness in seeking diversion of sentencing has not proven successful in most cases.
Schools get mental health allocation
The Orleans Parish School Board, which serves all of New Orleans, will allocate $1.3 million to the Center for Resilience, a local mental health day treatment program, beginning this year. The new program will expand mental health help to children in grades 9-12, according to a report in the Times-Picayune.
The funding will enable the center to expand an existing program that helps students with behavioral issues. Such help is not available in the traditional school system. By offsetting part of the price tag for the mental health care, the initiative “[helps to make] this critical service more available for our students most in need,” said Dominique Ellis, a spokesperson for the school board. “Mental health day treatment programs like the Center for Resilience typically cost between the ranges of $80,000-$100,000 per student to operate effectively.”
The development brings New Orleans level with money spent on similar programs in other school boards nationwide. It’s a service that is sorely needed. According to statistics supplied by the Center for Resilience, 60% of New Orleans children suffer from PTSD and are 4.5 times more likely to suffer from hyperactivity, aggression, and social withdrawal than similarly aged children elsewhere in the United States.
Bill addressing opioid crisis a “no-brainer”
Legislation put forward in the current session of the Texas Legislature would require pharmacists to identify all prescription opioids with a red cap and a hard-to-miss warning of the addictive risk of the medications. In addition, pharmacists would have to explain the risk in person to those receiving the medications and get signed acknowledgment of the conversation before dispensing the drugs.
As described in an article in the Austin American-Stateman, nearly 3,000 people in Texas died from drug overdoses in 2017. The deaths tied to opioid overdoses are not certain, but other data from 2015 suggest that one-third is a reasonable estimate.
“Losing a loved one to an opioid overdose is a tragedy that far too many Texas families experience,” said Rep. Shawn Thierry last month, when she introduced the three bills. “These distinctive red caps will serve as a clear notice to Texans that opioids are unlike milder forms of prescription pain relievers and have life-altering risks that must be considered before taking them.”
Warning labels on everything from food to tobacco products, and including them on prescription opioids is a “no-brainer,” Ms. Thierry said. “The more we can educate our residents the less likely they will be to misuse these medications.”
A report issued in November 2018 chronicled the drug crisis in Texas and offered recommendations. Many of the 100 recommendations involved the absence of treatment resources in the state.
Families are the bedrock of the lives of many, but some people opt to separate permanently from family members. A recent episode of the NPR program “1A” explored why families sever ties.
For journalist Harriet Brown, author of “Shadow Daughter: A Memoir of Estrangement,” (Da Capo Press, 2018), the decision to end her relationship with her mother came after she blamed Ms. Brown in “a blistering email” for the relapsed anorexia of Ms. Brown’s daughter.
“I was done with her,” Ms. Brown said on the program. “I told her I was done. That was it. And I never talked to her again. I think for both of us it felt final in some way.”
She said a lot of shame and stigma comes from having a bad relationship with a parent. “I really wanted to make it clear that ... sometimes walking away is really the best thing to do.”
Kristina M. Scharp, PhD, who has studied the estrangement phenomenon, said no national data exist on family estrangement. “About 12% of mothers and research would suggest even more fathers would report being estranged from one of their children,” said Dr. Scharp, who is with the University of Washington, Seattle. “It’s fairly common.”
Estrangement might be more common today because times have changed, said Joshua Coleman, PhD, author of “When Parents Hurt: Compassionate Strategies When you and Your Grown Child Don’t get Along” (HarperCollins, 2008). “Today’s adult children don’t view their relationships with their parents the way their folks did with their parents … the principles of obligation, duty, and respect that baby boomers and generations before them had for their elders aren’t necessarily there anymore,” Dr. Coleman said in a previous interview with the Chicago Tribune.
For her part, Ms. Brown said, the relationship with her mother – who is now deceased – might have been healed with respect. she said. “Honestly, it was that basic with my mother.”
Phelps gets mental health advocacy award
In the pool, Michael Phelps was golden, with 28 Olympics medals, 23 of them gold, hanging around his neck by the end of his swimming career. But the release from the water to real life after the 2016 Summer Olympics left no outlet for troubles that had dogged him for years. Drunk driving convictions and a ban from competition during his competitive years had failed to stop his downward spiral of depression. His thoughts turned to suicide.
But his story has a bright ending. With his realization that he had hit rock bottom, and with the help of his wife and therapists, he accepted his depression and learned to live with the reality that his life is, for the most part, pretty good.
His openness about his struggles with depression has been influential. The latest recognition came in early January, when he received the Morton E. Ruderman Award in Inclusion from the Ruderman Family Foundation. As reported in the Boston Globe, Mr. Phelps was recognized for his advocacy for people with disabilities and “his own journey with mental health.”
“I do like who I am and I’m comfortable with who I am. I couldn’t say that a few years ago. So I’m in a very good place and just living life one day at a time,” Mr. Phelps remarked in an interview with CNN last year.
Mr. Phelps is now a paid spokesperson for TalkSpace, an online therapy company.
“I’d like to make a difference. I’d like to be able to save a life if I can. You know, for me that’s more important than winning a gold medal. The stuff that I’m doing now is very exciting. It’s hard, it’s challenging but it’s fun for me. That’s what drives me to get out of bed every morning,” he said.
Offenders with mental illness get a break
A law included as part of budget legislation that was signed by then-Gov. Jerry Brown of California in June 2018 has offered people with mental health troubles who have been charged with a crime the opportunity for treatment instead of jail time.
As reported in the Los Angeles Times, the law provides judges with the discretion to order offenders into treatment rather than sentencing them. Success in treatment can lead to charges being dropped.
The law has been praised by some mental health advocates but panned by law enforcement officials and prosecutors with a harder view of criminal justice. The opposition stems largely from a December 2018 ruling by the 4th District Court of Appeal that the law could be applied to retroactively address the case of a man imprisoned for 29 years in 2017 for multiple felony charges that included domestic violence and assault.
The law does not extend to those charged with murder, manslaughter, rape, and child sexual abuse. To date, citing mental illness in seeking diversion of sentencing has not proven successful in most cases.
Schools get mental health allocation
The Orleans Parish School Board, which serves all of New Orleans, will allocate $1.3 million to the Center for Resilience, a local mental health day treatment program, beginning this year. The new program will expand mental health help to children in grades 9-12, according to a report in the Times-Picayune.
The funding will enable the center to expand an existing program that helps students with behavioral issues. Such help is not available in the traditional school system. By offsetting part of the price tag for the mental health care, the initiative “[helps to make] this critical service more available for our students most in need,” said Dominique Ellis, a spokesperson for the school board. “Mental health day treatment programs like the Center for Resilience typically cost between the ranges of $80,000-$100,000 per student to operate effectively.”
The development brings New Orleans level with money spent on similar programs in other school boards nationwide. It’s a service that is sorely needed. According to statistics supplied by the Center for Resilience, 60% of New Orleans children suffer from PTSD and are 4.5 times more likely to suffer from hyperactivity, aggression, and social withdrawal than similarly aged children elsewhere in the United States.
Bill addressing opioid crisis a “no-brainer”
Legislation put forward in the current session of the Texas Legislature would require pharmacists to identify all prescription opioids with a red cap and a hard-to-miss warning of the addictive risk of the medications. In addition, pharmacists would have to explain the risk in person to those receiving the medications and get signed acknowledgment of the conversation before dispensing the drugs.
As described in an article in the Austin American-Stateman, nearly 3,000 people in Texas died from drug overdoses in 2017. The deaths tied to opioid overdoses are not certain, but other data from 2015 suggest that one-third is a reasonable estimate.
“Losing a loved one to an opioid overdose is a tragedy that far too many Texas families experience,” said Rep. Shawn Thierry last month, when she introduced the three bills. “These distinctive red caps will serve as a clear notice to Texans that opioids are unlike milder forms of prescription pain relievers and have life-altering risks that must be considered before taking them.”
Warning labels on everything from food to tobacco products, and including them on prescription opioids is a “no-brainer,” Ms. Thierry said. “The more we can educate our residents the less likely they will be to misuse these medications.”
A report issued in November 2018 chronicled the drug crisis in Texas and offered recommendations. Many of the 100 recommendations involved the absence of treatment resources in the state.
RNA expression may hold key to hemophilia A severity
A decrease in the expression of two long noncoding RNAs in the factor VIII gene may explain the development of severe hemophilia, according to an analysis recently published in Hematology.
Researchers at the Iranian Comprehensive Hemophilia Care Center in Tehran identified two long noncoding RNAs for investigation – NONHSAT139219.2 and NONHSAT139215.2. They collected 5 mL of venous blood from 50 males with severe hemophilia A and 50 healthy male donors and analyzed the RNA expression.
The mean of the transcript levels of two long noncoding RNAs was significantly lower in the hemophilia A samples, compared with the normal samples – 5.52 for controls versus 1.25 for hemophilia A for NONHSAT139215 and 4.86 for controls versus 2.14 for hemophilia A samples for NONHSAT139219 (P less than .05).
Low expression levels of long noncoding RNAs in severe hemophilia A cases may be linked with the reduction of factor VIII levels, according to the investigators. “It is possible that the transcription of [long noncoding] RNAs leads to gene silencing or activation.”
This provides a potential biomarker with application in both prognosis and therapeutics; long noncoding RNA functional analysis should be performed in future studies, they wrote.
There was no outside funding for the study, and no potential conflict of interest was reported by the authors.
SOURCE: Niloofar N et al. Hematology. 2019;24(1):255-62.
A decrease in the expression of two long noncoding RNAs in the factor VIII gene may explain the development of severe hemophilia, according to an analysis recently published in Hematology.
Researchers at the Iranian Comprehensive Hemophilia Care Center in Tehran identified two long noncoding RNAs for investigation – NONHSAT139219.2 and NONHSAT139215.2. They collected 5 mL of venous blood from 50 males with severe hemophilia A and 50 healthy male donors and analyzed the RNA expression.
The mean of the transcript levels of two long noncoding RNAs was significantly lower in the hemophilia A samples, compared with the normal samples – 5.52 for controls versus 1.25 for hemophilia A for NONHSAT139215 and 4.86 for controls versus 2.14 for hemophilia A samples for NONHSAT139219 (P less than .05).
Low expression levels of long noncoding RNAs in severe hemophilia A cases may be linked with the reduction of factor VIII levels, according to the investigators. “It is possible that the transcription of [long noncoding] RNAs leads to gene silencing or activation.”
This provides a potential biomarker with application in both prognosis and therapeutics; long noncoding RNA functional analysis should be performed in future studies, they wrote.
There was no outside funding for the study, and no potential conflict of interest was reported by the authors.
SOURCE: Niloofar N et al. Hematology. 2019;24(1):255-62.
A decrease in the expression of two long noncoding RNAs in the factor VIII gene may explain the development of severe hemophilia, according to an analysis recently published in Hematology.
Researchers at the Iranian Comprehensive Hemophilia Care Center in Tehran identified two long noncoding RNAs for investigation – NONHSAT139219.2 and NONHSAT139215.2. They collected 5 mL of venous blood from 50 males with severe hemophilia A and 50 healthy male donors and analyzed the RNA expression.
The mean of the transcript levels of two long noncoding RNAs was significantly lower in the hemophilia A samples, compared with the normal samples – 5.52 for controls versus 1.25 for hemophilia A for NONHSAT139215 and 4.86 for controls versus 2.14 for hemophilia A samples for NONHSAT139219 (P less than .05).
Low expression levels of long noncoding RNAs in severe hemophilia A cases may be linked with the reduction of factor VIII levels, according to the investigators. “It is possible that the transcription of [long noncoding] RNAs leads to gene silencing or activation.”
This provides a potential biomarker with application in both prognosis and therapeutics; long noncoding RNA functional analysis should be performed in future studies, they wrote.
There was no outside funding for the study, and no potential conflict of interest was reported by the authors.
SOURCE: Niloofar N et al. Hematology. 2019;24(1):255-62.
FROM HEMATOLOGY
Key clinical point:
Major finding: Transcript levels of two long noncoding RNAs were significantly lower in severe hemophilia A patients, compared with healthy controls (P less than .05). Study details: An analysis of RNA expression levels in 50 patients with severe hemophilia A and 50 healthy controls at a single center in Iran.
Disclosures: There was no outside funding for the study, and no potential conflict of interest information was reported by the authors.
Source: Niloofar N et al. Hematology. 2019;24(1):255-62.
Intrapartum molecular GBS screening reduced newborn early-onset disease, antibiotic use
Point-of-care intrapartum molecular screening of group B Streptococcus reduced the incidence of early-onset disease cases and antibiotic use, according to research published in Obstetrics & Gynecology.
Najoua El Helali, PharmD, from the Service de Microbiologie Clinique at Groupe Hospitalier Paris Saint-Joseph, and her colleagues measured the rate of early-onset disease group B Streptococcus (GBS) in a single-center study analyzing antenatal culture screening for 4 years prior to implementation (2006-2009) of polymerase chain reaction (PCR) screening (2010-2015). There were 11,226 deliveries (11,818 live births) during the antenatal screening period and 18,835 deliveries (18,980 live births) during the PCR screening period. Overall, 4% of deliveries during the antenatal period and 0.1% of deliveries during the intrapartum period were not screened for GBS (P less than .001).
During 2006-2015, the rate of early-onset disease of GBS decreased to 0.21/1,000 cases from 1.01/1,000 cases (risk ratio, 0.25; 95% confidence interval, 0.14-0.43; P = .026), while the rate of probable early-onset disease GBS decreased to 0.73/1,000 cases from 2.8/1,000 cases (RR, 0.25; (95% CI, 0.14-0.43; P less than .001).
For patients with early-onset GBS, length of stay in hospital decreased by 64%, and antibiotic therapy decreased by 60%, but there was no significant difference in average length of stay or duration of antibiotic therapy during the study period. There was a reduction in annual delivery- and treatment-associated costs of early-onset disease GBS from $41,875 to $11,945, while the estimated extra cost of PCR screening to avoid one additional case of early-onset disease GBS was $5,819 and a cost increase of $49 per newborn.
“The additional PCR costs were offset in part by the reduction in early-onset GBS disease treatment costs,” the investigators said.
“A randomized, controlled multicenter study is probably needed to evaluate the cost-effectiveness of this prevention strategy and demonstrate a better efficacy in populations where poorly followed women are of unknown GBS status at presentation for delivery,” the researchers said. “In term newborns, however, using infection rate as an endpoint is problematic given the sample size needed.”
The researchers said their study was potentially limited by lack of a control group and population selection, and described mothers in their center as “mostly well-informed and well-monitored during their pregnancy.”
The authors reported no relevant conflicts of interest.
SOURCE: El Helali N et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003057.
Point-of-care intrapartum molecular screening of group B Streptococcus reduced the incidence of early-onset disease cases and antibiotic use, according to research published in Obstetrics & Gynecology.
Najoua El Helali, PharmD, from the Service de Microbiologie Clinique at Groupe Hospitalier Paris Saint-Joseph, and her colleagues measured the rate of early-onset disease group B Streptococcus (GBS) in a single-center study analyzing antenatal culture screening for 4 years prior to implementation (2006-2009) of polymerase chain reaction (PCR) screening (2010-2015). There were 11,226 deliveries (11,818 live births) during the antenatal screening period and 18,835 deliveries (18,980 live births) during the PCR screening period. Overall, 4% of deliveries during the antenatal period and 0.1% of deliveries during the intrapartum period were not screened for GBS (P less than .001).
During 2006-2015, the rate of early-onset disease of GBS decreased to 0.21/1,000 cases from 1.01/1,000 cases (risk ratio, 0.25; 95% confidence interval, 0.14-0.43; P = .026), while the rate of probable early-onset disease GBS decreased to 0.73/1,000 cases from 2.8/1,000 cases (RR, 0.25; (95% CI, 0.14-0.43; P less than .001).
For patients with early-onset GBS, length of stay in hospital decreased by 64%, and antibiotic therapy decreased by 60%, but there was no significant difference in average length of stay or duration of antibiotic therapy during the study period. There was a reduction in annual delivery- and treatment-associated costs of early-onset disease GBS from $41,875 to $11,945, while the estimated extra cost of PCR screening to avoid one additional case of early-onset disease GBS was $5,819 and a cost increase of $49 per newborn.
“The additional PCR costs were offset in part by the reduction in early-onset GBS disease treatment costs,” the investigators said.
“A randomized, controlled multicenter study is probably needed to evaluate the cost-effectiveness of this prevention strategy and demonstrate a better efficacy in populations where poorly followed women are of unknown GBS status at presentation for delivery,” the researchers said. “In term newborns, however, using infection rate as an endpoint is problematic given the sample size needed.”
The researchers said their study was potentially limited by lack of a control group and population selection, and described mothers in their center as “mostly well-informed and well-monitored during their pregnancy.”
The authors reported no relevant conflicts of interest.
SOURCE: El Helali N et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003057.
Point-of-care intrapartum molecular screening of group B Streptococcus reduced the incidence of early-onset disease cases and antibiotic use, according to research published in Obstetrics & Gynecology.
Najoua El Helali, PharmD, from the Service de Microbiologie Clinique at Groupe Hospitalier Paris Saint-Joseph, and her colleagues measured the rate of early-onset disease group B Streptococcus (GBS) in a single-center study analyzing antenatal culture screening for 4 years prior to implementation (2006-2009) of polymerase chain reaction (PCR) screening (2010-2015). There were 11,226 deliveries (11,818 live births) during the antenatal screening period and 18,835 deliveries (18,980 live births) during the PCR screening period. Overall, 4% of deliveries during the antenatal period and 0.1% of deliveries during the intrapartum period were not screened for GBS (P less than .001).
During 2006-2015, the rate of early-onset disease of GBS decreased to 0.21/1,000 cases from 1.01/1,000 cases (risk ratio, 0.25; 95% confidence interval, 0.14-0.43; P = .026), while the rate of probable early-onset disease GBS decreased to 0.73/1,000 cases from 2.8/1,000 cases (RR, 0.25; (95% CI, 0.14-0.43; P less than .001).
For patients with early-onset GBS, length of stay in hospital decreased by 64%, and antibiotic therapy decreased by 60%, but there was no significant difference in average length of stay or duration of antibiotic therapy during the study period. There was a reduction in annual delivery- and treatment-associated costs of early-onset disease GBS from $41,875 to $11,945, while the estimated extra cost of PCR screening to avoid one additional case of early-onset disease GBS was $5,819 and a cost increase of $49 per newborn.
“The additional PCR costs were offset in part by the reduction in early-onset GBS disease treatment costs,” the investigators said.
“A randomized, controlled multicenter study is probably needed to evaluate the cost-effectiveness of this prevention strategy and demonstrate a better efficacy in populations where poorly followed women are of unknown GBS status at presentation for delivery,” the researchers said. “In term newborns, however, using infection rate as an endpoint is problematic given the sample size needed.”
The researchers said their study was potentially limited by lack of a control group and population selection, and described mothers in their center as “mostly well-informed and well-monitored during their pregnancy.”
The authors reported no relevant conflicts of interest.
SOURCE: El Helali N et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003057.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point:
Major finding: The rate of early-onset disease group B Streptococcus decreased from 1.01/1,000 cases to 0.21/1,000 cases across the antenatal and intrapartum periods.
Study details: A single-center study of antenatal culture screening for 11,226 deliveries during 2006-2009 and intrapartum PCR screening for 18,835 deliveries during 2010-2015.
Disclosures: The authors reported no relevant conflicts of interest.
Source: El Helali N et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003057.











