User login
Blood thinning with bioprosthetic valves
Clinical question: Does anticoagulation prevent thromboembolic events in patients undergoing bioprosthetic valve implantation?
Background: The main advantage of bioprosthetic valves, compared with mechanical valves, is the avoidance of long-term anticoagulation. Current guidelines recommend the use of vitamin K antagonist (VKA) during the first 3 months after surgery, which remains controversial. Two randomized controlled trials (RCTs) showed no benefit of using VKA in the first 3 months; however, other studies have reported conflicting results.
Study design: Meta-analysis and systematic review.
Setting: Multicenter.
Synopsis: This meta-analysis included two RCTs and 12 observational studies that compared the outcomes in group I (VKA) versus group II (antiplatelet therapy/no treatment). There was no difference in thromboembolic events between group I (1%) and group II (1.5%), but there were more bleeding events in group I (2.6%) versus group II (1.1%). In addition, no differences in all-cause of mortality rate and need for redo surgery were found between the two groups.
Bottom line: The use of VKA in the first 3 months after a bioprosthetic valve implantation does not decrease the rate of thromboembolic events or mortality, but it is associated with increased risk of major bleeding.
Citation: Masri A, Gillinov M, Johnston DM, et al. Anticoagulation versus antiplatelet or no therapy in patients undergoing bioprosthetic valve implantation: a systematic review and meta-analysis [published online ahead of print Aug. 3, 2016]. Heart. doi: 10.1136/heartjnl-2016-309630
Dr. Florindez is an assistant professor at the University of Miami Miller School of Medicine and a hospitalist at University of Miami Hospital and Jackson Memorial Hospital.
Clinical question: Does anticoagulation prevent thromboembolic events in patients undergoing bioprosthetic valve implantation?
Background: The main advantage of bioprosthetic valves, compared with mechanical valves, is the avoidance of long-term anticoagulation. Current guidelines recommend the use of vitamin K antagonist (VKA) during the first 3 months after surgery, which remains controversial. Two randomized controlled trials (RCTs) showed no benefit of using VKA in the first 3 months; however, other studies have reported conflicting results.
Study design: Meta-analysis and systematic review.
Setting: Multicenter.
Synopsis: This meta-analysis included two RCTs and 12 observational studies that compared the outcomes in group I (VKA) versus group II (antiplatelet therapy/no treatment). There was no difference in thromboembolic events between group I (1%) and group II (1.5%), but there were more bleeding events in group I (2.6%) versus group II (1.1%). In addition, no differences in all-cause of mortality rate and need for redo surgery were found between the two groups.
Bottom line: The use of VKA in the first 3 months after a bioprosthetic valve implantation does not decrease the rate of thromboembolic events or mortality, but it is associated with increased risk of major bleeding.
Citation: Masri A, Gillinov M, Johnston DM, et al. Anticoagulation versus antiplatelet or no therapy in patients undergoing bioprosthetic valve implantation: a systematic review and meta-analysis [published online ahead of print Aug. 3, 2016]. Heart. doi: 10.1136/heartjnl-2016-309630
Dr. Florindez is an assistant professor at the University of Miami Miller School of Medicine and a hospitalist at University of Miami Hospital and Jackson Memorial Hospital.
Clinical question: Does anticoagulation prevent thromboembolic events in patients undergoing bioprosthetic valve implantation?
Background: The main advantage of bioprosthetic valves, compared with mechanical valves, is the avoidance of long-term anticoagulation. Current guidelines recommend the use of vitamin K antagonist (VKA) during the first 3 months after surgery, which remains controversial. Two randomized controlled trials (RCTs) showed no benefit of using VKA in the first 3 months; however, other studies have reported conflicting results.
Study design: Meta-analysis and systematic review.
Setting: Multicenter.
Synopsis: This meta-analysis included two RCTs and 12 observational studies that compared the outcomes in group I (VKA) versus group II (antiplatelet therapy/no treatment). There was no difference in thromboembolic events between group I (1%) and group II (1.5%), but there were more bleeding events in group I (2.6%) versus group II (1.1%). In addition, no differences in all-cause of mortality rate and need for redo surgery were found between the two groups.
Bottom line: The use of VKA in the first 3 months after a bioprosthetic valve implantation does not decrease the rate of thromboembolic events or mortality, but it is associated with increased risk of major bleeding.
Citation: Masri A, Gillinov M, Johnston DM, et al. Anticoagulation versus antiplatelet or no therapy in patients undergoing bioprosthetic valve implantation: a systematic review and meta-analysis [published online ahead of print Aug. 3, 2016]. Heart. doi: 10.1136/heartjnl-2016-309630
Dr. Florindez is an assistant professor at the University of Miami Miller School of Medicine and a hospitalist at University of Miami Hospital and Jackson Memorial Hospital.
The Barbershop Study: Hypertension causes nocturia
NEW ORLEANS – Uncontrolled systolic hypertension is a strong independent determinant of nocturia in middle-aged African American men, O’Neil Mason, MD, reported at the American Heart Association scientific sessions.
This finding from the ongoing National Heart, Lung, and Blood Institute–sponsored Barbershop Study challenges the traditional notion of hypertension as an asymptomatic disease. It also provides a novel health promotion message aimed at improving compliance with blood pressure medication.
The Barbershop Study is a hypertension intervention trial that’s being conducted in Los Angeles barbershops frequented by black men. In the initial screening phase for study eligibility, 2,577 African American men aged 35-79 years underwent highly accurate blood pressure measurements using an average of three readings taken via an oscillometric monitor.
The mean age of the men was 53 years. It was an obese group, with a mean body mass index of 30 kg/m2. Fifty percent of the men had hypertension, and among that cohort fully one-third weren’t on antihypertensive medication and another 28% were treated but uncontrolled, with on-treatment blood pressures of 140/90 mm Hg or more. Thus, only 39% of these middle-aged African American men with high blood pressure were treated and controlled at baseline.
Seventy-seven percent of the screened men reported awakening once or more per night to urinate. A progressive increase in nocturia severity was seen with increasing systolic blood pressure. The prevalence of nocturia ranged from 68% among normotensive men to 91% among those with treated but uncontrolled hypertension, Dr. Mason reported.
In a multivariate logistic regression analysis controlling for the standard risk factors for nocturia – including advancing age, an enlarged prostate, and diabetes, which was present in 16% of the men – stage 1 systolic hypertension in the range of 140-159 mm Hg was independently associated with a 1.57-fold increased likelihood of nocturia, compared with normotensive subjects. Stage 2 hypertension, with a systolic blood pressure of 160 mm Hg or more, was associated with a 2.32-fold increased risk; that’s in the same ballpark as having an enlarged prostate, which carried a 2.1-fold increased risk. Prehypertension – that is, a systolic pressure of 120-139 mm Hg – was associated with a nonsignificant 1.18-fold risk.
Diastolic blood pressure wasn’t an independent determinant of nocturia.
In a similar multivariate analysis focused on severe nocturia, defined as three or more episodes per night, stage 1 systolic hypertension was independently associated with a 2.29-fold increased risk, compared with normotension, and stage 2 systolic hypertension carried a 2.77-fold increased risk.
Audience members were clearly intrigued by this novel finding. They were quick to speculate as to potential underlying pathophysiologic mechanisms, including atrial stretch, increased renal blood flow, or perhaps a side effect of diuretic therapy. However, Dr. Mason and his coinvestigators favor another possibility: “African Americans have more salt-sensitive hypertension and they have less nocturnal blood pressure dipping,” he noted. “So if nighttime blood pressure is high it could lead through increased pressure natriuresis to increased urine production. More activity in getting up to go to the bathroom increases the blood pressure and creates a cycle that begets more urine.”
Asked if uncontrolled systolic hypertension is also a determinant of nocturia in African American women, Dr. Mason replied that he would assume so. But that question hasn’t ever been studied. The Barbershop Study is restricted to African American men with hypertension because studies have shown they have a particularly low rate of controlled hypertension. In contrast, the controlled hypertension rate among hypertensive African American women is comparable with their white counterparts.
In the next phase of the Barbershop Study, participants’ use of various classes of antihypertensive medication will be prospectively tracked. Among other things, this will enable investigators to determine whether diuretics contribute to nocturia.
Dr. Mason reported having no conflicts of interest regarding the study.
NEW ORLEANS – Uncontrolled systolic hypertension is a strong independent determinant of nocturia in middle-aged African American men, O’Neil Mason, MD, reported at the American Heart Association scientific sessions.
This finding from the ongoing National Heart, Lung, and Blood Institute–sponsored Barbershop Study challenges the traditional notion of hypertension as an asymptomatic disease. It also provides a novel health promotion message aimed at improving compliance with blood pressure medication.
The Barbershop Study is a hypertension intervention trial that’s being conducted in Los Angeles barbershops frequented by black men. In the initial screening phase for study eligibility, 2,577 African American men aged 35-79 years underwent highly accurate blood pressure measurements using an average of three readings taken via an oscillometric monitor.
The mean age of the men was 53 years. It was an obese group, with a mean body mass index of 30 kg/m2. Fifty percent of the men had hypertension, and among that cohort fully one-third weren’t on antihypertensive medication and another 28% were treated but uncontrolled, with on-treatment blood pressures of 140/90 mm Hg or more. Thus, only 39% of these middle-aged African American men with high blood pressure were treated and controlled at baseline.
Seventy-seven percent of the screened men reported awakening once or more per night to urinate. A progressive increase in nocturia severity was seen with increasing systolic blood pressure. The prevalence of nocturia ranged from 68% among normotensive men to 91% among those with treated but uncontrolled hypertension, Dr. Mason reported.
In a multivariate logistic regression analysis controlling for the standard risk factors for nocturia – including advancing age, an enlarged prostate, and diabetes, which was present in 16% of the men – stage 1 systolic hypertension in the range of 140-159 mm Hg was independently associated with a 1.57-fold increased likelihood of nocturia, compared with normotensive subjects. Stage 2 hypertension, with a systolic blood pressure of 160 mm Hg or more, was associated with a 2.32-fold increased risk; that’s in the same ballpark as having an enlarged prostate, which carried a 2.1-fold increased risk. Prehypertension – that is, a systolic pressure of 120-139 mm Hg – was associated with a nonsignificant 1.18-fold risk.
Diastolic blood pressure wasn’t an independent determinant of nocturia.
In a similar multivariate analysis focused on severe nocturia, defined as three or more episodes per night, stage 1 systolic hypertension was independently associated with a 2.29-fold increased risk, compared with normotension, and stage 2 systolic hypertension carried a 2.77-fold increased risk.
Audience members were clearly intrigued by this novel finding. They were quick to speculate as to potential underlying pathophysiologic mechanisms, including atrial stretch, increased renal blood flow, or perhaps a side effect of diuretic therapy. However, Dr. Mason and his coinvestigators favor another possibility: “African Americans have more salt-sensitive hypertension and they have less nocturnal blood pressure dipping,” he noted. “So if nighttime blood pressure is high it could lead through increased pressure natriuresis to increased urine production. More activity in getting up to go to the bathroom increases the blood pressure and creates a cycle that begets more urine.”
Asked if uncontrolled systolic hypertension is also a determinant of nocturia in African American women, Dr. Mason replied that he would assume so. But that question hasn’t ever been studied. The Barbershop Study is restricted to African American men with hypertension because studies have shown they have a particularly low rate of controlled hypertension. In contrast, the controlled hypertension rate among hypertensive African American women is comparable with their white counterparts.
In the next phase of the Barbershop Study, participants’ use of various classes of antihypertensive medication will be prospectively tracked. Among other things, this will enable investigators to determine whether diuretics contribute to nocturia.
Dr. Mason reported having no conflicts of interest regarding the study.
NEW ORLEANS – Uncontrolled systolic hypertension is a strong independent determinant of nocturia in middle-aged African American men, O’Neil Mason, MD, reported at the American Heart Association scientific sessions.
This finding from the ongoing National Heart, Lung, and Blood Institute–sponsored Barbershop Study challenges the traditional notion of hypertension as an asymptomatic disease. It also provides a novel health promotion message aimed at improving compliance with blood pressure medication.
The Barbershop Study is a hypertension intervention trial that’s being conducted in Los Angeles barbershops frequented by black men. In the initial screening phase for study eligibility, 2,577 African American men aged 35-79 years underwent highly accurate blood pressure measurements using an average of three readings taken via an oscillometric monitor.
The mean age of the men was 53 years. It was an obese group, with a mean body mass index of 30 kg/m2. Fifty percent of the men had hypertension, and among that cohort fully one-third weren’t on antihypertensive medication and another 28% were treated but uncontrolled, with on-treatment blood pressures of 140/90 mm Hg or more. Thus, only 39% of these middle-aged African American men with high blood pressure were treated and controlled at baseline.
Seventy-seven percent of the screened men reported awakening once or more per night to urinate. A progressive increase in nocturia severity was seen with increasing systolic blood pressure. The prevalence of nocturia ranged from 68% among normotensive men to 91% among those with treated but uncontrolled hypertension, Dr. Mason reported.
In a multivariate logistic regression analysis controlling for the standard risk factors for nocturia – including advancing age, an enlarged prostate, and diabetes, which was present in 16% of the men – stage 1 systolic hypertension in the range of 140-159 mm Hg was independently associated with a 1.57-fold increased likelihood of nocturia, compared with normotensive subjects. Stage 2 hypertension, with a systolic blood pressure of 160 mm Hg or more, was associated with a 2.32-fold increased risk; that’s in the same ballpark as having an enlarged prostate, which carried a 2.1-fold increased risk. Prehypertension – that is, a systolic pressure of 120-139 mm Hg – was associated with a nonsignificant 1.18-fold risk.
Diastolic blood pressure wasn’t an independent determinant of nocturia.
In a similar multivariate analysis focused on severe nocturia, defined as three or more episodes per night, stage 1 systolic hypertension was independently associated with a 2.29-fold increased risk, compared with normotension, and stage 2 systolic hypertension carried a 2.77-fold increased risk.
Audience members were clearly intrigued by this novel finding. They were quick to speculate as to potential underlying pathophysiologic mechanisms, including atrial stretch, increased renal blood flow, or perhaps a side effect of diuretic therapy. However, Dr. Mason and his coinvestigators favor another possibility: “African Americans have more salt-sensitive hypertension and they have less nocturnal blood pressure dipping,” he noted. “So if nighttime blood pressure is high it could lead through increased pressure natriuresis to increased urine production. More activity in getting up to go to the bathroom increases the blood pressure and creates a cycle that begets more urine.”
Asked if uncontrolled systolic hypertension is also a determinant of nocturia in African American women, Dr. Mason replied that he would assume so. But that question hasn’t ever been studied. The Barbershop Study is restricted to African American men with hypertension because studies have shown they have a particularly low rate of controlled hypertension. In contrast, the controlled hypertension rate among hypertensive African American women is comparable with their white counterparts.
In the next phase of the Barbershop Study, participants’ use of various classes of antihypertensive medication will be prospectively tracked. Among other things, this will enable investigators to determine whether diuretics contribute to nocturia.
Dr. Mason reported having no conflicts of interest regarding the study.
Key clinical point:
Major finding: A systolic blood pressure of 140-159 mm Hg was independently associated with a 2.29-fold increased risk of severe nocturia, compared with normotension in middle-aged African American men, while a pressure of 160 mm Hg or more conferred a 2.77-fold increased risk.
Data source: A report on the initial cross-sectional screening phase of the Barbershop Study, in which 2,577 middle-aged African American men underwent blood pressure measurements in Los Angeles barbershops.
Disclosures: The Barbershop Study is funded by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts.
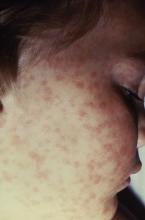
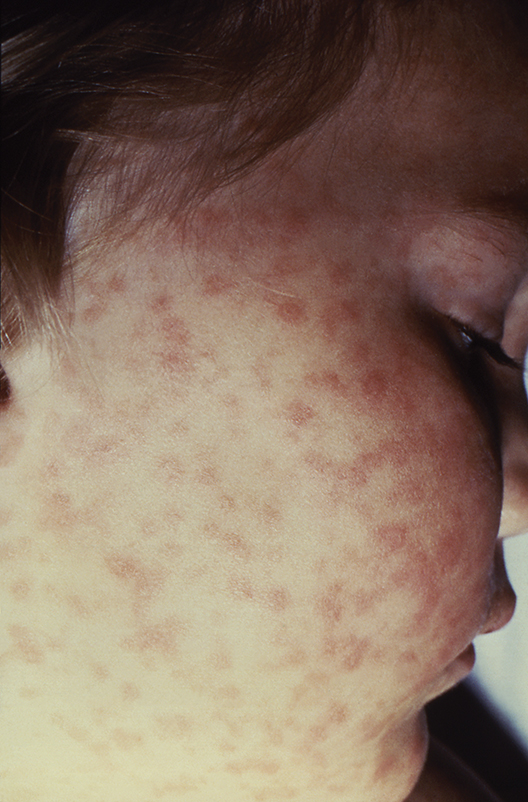
Lab values poor surrogate for detecting pediatric Rocky Mountain spotted fever in children
The three fatalities observed in a retrospective analysis of six cases of Rocky Mountain spotted fever (RMSF) in children were associated with either a delayed diagnosis pending laboratory findings or delayed antirickettsia treatment.
“The fact that all fatal cases died before the convalescent period emphasizes that diagnosis should be based on clinical findings instead of RMSF serologic and histologic testing,” wrote the authors of a study published online in Pediatric Dermatology (2016 Dec 19. doi: 10.1111/pde.13053).
Two of the fatal cases involved delayed antirickettsial therapy after the patients were misdiagnosed with group A streptococcus. None of the six children were initially evaluated for R. rickettsii; they averaged three encounters with their clinician before being admitted for acute inpatient care where they received intravenous doxycycline after nearly a week of symptoms.
“All fatal cases were complicated by neurologic manifestations, including seizures, obtundation, and uncal herniation,” a finding that is consistent with the literature, the authors said.
Although the high fatality rate might be the result of the small study size, Ms. Tull and her coinvestigators concluded that the disease should be considered in all differential diagnoses for children who present with a fever and rash during the summer months in endemic areas, particularly since pediatric cases of the disease are associated with poorer outcomes than in adult cases.
Given that RMSF often remains subclinical in its early stages, and typically presents with nonspecific symptoms of fever, rash, headache, and abdominal pain when it does emerge, physicians might be tempted to defer treatment until after serologic and histologic results are in, as is the standard method. Concerns over doxycycline’s tendency to stain teeth and cause enamel hypoplasia are also common. However, empirical administration could mean the difference between life and death, since treatment within the first 5 days following infection is associated with better outcomes – an algorithm complicated by the fact that symptoms caused by R. rickettsii have been known to take as long as 21 days to appear.
In the study, Ms. Tull and her colleagues found that the average time between exposure to the tick and the onset of symptoms was 6.6 days (range, 1-21 days).
Currently, there are no diagnostic tests “that reliably diagnose RMSF during the first 7 days of illness,” and most patients “do not develop detectable antibodies until the second week of illness,” the investigators reported. Even then, sensitivity of indirect fluorescent antibody serum testing after the second week of illness is only between 86% and 94%, they noted. Further, the sensitivity of immunohistochemical (IHC) tissue staining has been reported at 70%, and false-negative IHC results are common in acute disease when antibody response is harder to detect.
Ms. Tull and her colleagues found that five of the six patients in their study had negative IHC testing; two of the six had positive serum antibody titers. For this reason, they concluded that Rocky Mountain spotted fever diagnosis should be based on “clinical history, examination, and laboratory abnormalities” rather than laboratory testing, and urged that “prompt treatment should be instituted empirically.”
The authors did not have any relevant financial disclosures.
[email protected]
On Twitter @whitneymcknight
The three fatalities observed in a retrospective analysis of six cases of Rocky Mountain spotted fever (RMSF) in children were associated with either a delayed diagnosis pending laboratory findings or delayed antirickettsia treatment.
“The fact that all fatal cases died before the convalescent period emphasizes that diagnosis should be based on clinical findings instead of RMSF serologic and histologic testing,” wrote the authors of a study published online in Pediatric Dermatology (2016 Dec 19. doi: 10.1111/pde.13053).
Two of the fatal cases involved delayed antirickettsial therapy after the patients were misdiagnosed with group A streptococcus. None of the six children were initially evaluated for R. rickettsii; they averaged three encounters with their clinician before being admitted for acute inpatient care where they received intravenous doxycycline after nearly a week of symptoms.
“All fatal cases were complicated by neurologic manifestations, including seizures, obtundation, and uncal herniation,” a finding that is consistent with the literature, the authors said.
Although the high fatality rate might be the result of the small study size, Ms. Tull and her coinvestigators concluded that the disease should be considered in all differential diagnoses for children who present with a fever and rash during the summer months in endemic areas, particularly since pediatric cases of the disease are associated with poorer outcomes than in adult cases.
Given that RMSF often remains subclinical in its early stages, and typically presents with nonspecific symptoms of fever, rash, headache, and abdominal pain when it does emerge, physicians might be tempted to defer treatment until after serologic and histologic results are in, as is the standard method. Concerns over doxycycline’s tendency to stain teeth and cause enamel hypoplasia are also common. However, empirical administration could mean the difference between life and death, since treatment within the first 5 days following infection is associated with better outcomes – an algorithm complicated by the fact that symptoms caused by R. rickettsii have been known to take as long as 21 days to appear.
In the study, Ms. Tull and her colleagues found that the average time between exposure to the tick and the onset of symptoms was 6.6 days (range, 1-21 days).
Currently, there are no diagnostic tests “that reliably diagnose RMSF during the first 7 days of illness,” and most patients “do not develop detectable antibodies until the second week of illness,” the investigators reported. Even then, sensitivity of indirect fluorescent antibody serum testing after the second week of illness is only between 86% and 94%, they noted. Further, the sensitivity of immunohistochemical (IHC) tissue staining has been reported at 70%, and false-negative IHC results are common in acute disease when antibody response is harder to detect.
Ms. Tull and her colleagues found that five of the six patients in their study had negative IHC testing; two of the six had positive serum antibody titers. For this reason, they concluded that Rocky Mountain spotted fever diagnosis should be based on “clinical history, examination, and laboratory abnormalities” rather than laboratory testing, and urged that “prompt treatment should be instituted empirically.”
The authors did not have any relevant financial disclosures.
[email protected]
On Twitter @whitneymcknight
The three fatalities observed in a retrospective analysis of six cases of Rocky Mountain spotted fever (RMSF) in children were associated with either a delayed diagnosis pending laboratory findings or delayed antirickettsia treatment.
“The fact that all fatal cases died before the convalescent period emphasizes that diagnosis should be based on clinical findings instead of RMSF serologic and histologic testing,” wrote the authors of a study published online in Pediatric Dermatology (2016 Dec 19. doi: 10.1111/pde.13053).
Two of the fatal cases involved delayed antirickettsial therapy after the patients were misdiagnosed with group A streptococcus. None of the six children were initially evaluated for R. rickettsii; they averaged three encounters with their clinician before being admitted for acute inpatient care where they received intravenous doxycycline after nearly a week of symptoms.
“All fatal cases were complicated by neurologic manifestations, including seizures, obtundation, and uncal herniation,” a finding that is consistent with the literature, the authors said.
Although the high fatality rate might be the result of the small study size, Ms. Tull and her coinvestigators concluded that the disease should be considered in all differential diagnoses for children who present with a fever and rash during the summer months in endemic areas, particularly since pediatric cases of the disease are associated with poorer outcomes than in adult cases.
Given that RMSF often remains subclinical in its early stages, and typically presents with nonspecific symptoms of fever, rash, headache, and abdominal pain when it does emerge, physicians might be tempted to defer treatment until after serologic and histologic results are in, as is the standard method. Concerns over doxycycline’s tendency to stain teeth and cause enamel hypoplasia are also common. However, empirical administration could mean the difference between life and death, since treatment within the first 5 days following infection is associated with better outcomes – an algorithm complicated by the fact that symptoms caused by R. rickettsii have been known to take as long as 21 days to appear.
In the study, Ms. Tull and her colleagues found that the average time between exposure to the tick and the onset of symptoms was 6.6 days (range, 1-21 days).
Currently, there are no diagnostic tests “that reliably diagnose RMSF during the first 7 days of illness,” and most patients “do not develop detectable antibodies until the second week of illness,” the investigators reported. Even then, sensitivity of indirect fluorescent antibody serum testing after the second week of illness is only between 86% and 94%, they noted. Further, the sensitivity of immunohistochemical (IHC) tissue staining has been reported at 70%, and false-negative IHC results are common in acute disease when antibody response is harder to detect.
Ms. Tull and her colleagues found that five of the six patients in their study had negative IHC testing; two of the six had positive serum antibody titers. For this reason, they concluded that Rocky Mountain spotted fever diagnosis should be based on “clinical history, examination, and laboratory abnormalities” rather than laboratory testing, and urged that “prompt treatment should be instituted empirically.”
The authors did not have any relevant financial disclosures.
[email protected]
On Twitter @whitneymcknight
FROM PEDIATRIC DERMATOLOGY
Key clinical point:
Major finding: Half of pediatric patients diagnosed with Rocky Mountain spotted fever died after treatment was delayed.
Data source: A retrospective analysis of 6 pediatric RMSF cases among 3,912 inpatient dermatology consultations over a period of 10 years at a tertiary care center.
Disclosures: The authors did not have any relevant financial disclosures. .
Medicare payments set for infliximab biosimilar Inflectra
Payment for the infliximab biosimilar drug Inflectra will now be covered by Medicare, the drug’s manufacturer, Pfizer, said in an announcement.
The Centers for Medicare & Medicaid Services (CMS) included Inflectra (infliximab-dyyb) in its January 2017 Average Sales Price pricing file, which went into effect Jan. 1, 2017. Pfizer said that Inflectra is priced at a 15% discount to the current wholesale acquisition cost for the infliximab originator Remicade, but this price does not include discounts to payers, providers, distributors, and other purchasing organizations.
For the first quarter of 2017, the payment limit set by the CMS for Inflectra is $100.306 per 10-mg unit and $82.218 for Remicade.
Various national and regional wholesalers across the country began receiving shipments of Inflectra in November 2016, according to Pfizer.
In conjunction with the availability of Inflectra, Pfizer announced its enCompass program, “a comprehensive reimbursement service and patient support program offering coding and reimbursement support for providers, copay assistance to eligible patients who have commercial insurance that covers Inflectra, and financial assistance for eligible uninsured and underinsured patients.”
The FDA approved Inflectra in April 2016 for all of the same indications as Remicade: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, plaque psoriasis, and ulcerative colitis.
Payment for the infliximab biosimilar drug Inflectra will now be covered by Medicare, the drug’s manufacturer, Pfizer, said in an announcement.
The Centers for Medicare & Medicaid Services (CMS) included Inflectra (infliximab-dyyb) in its January 2017 Average Sales Price pricing file, which went into effect Jan. 1, 2017. Pfizer said that Inflectra is priced at a 15% discount to the current wholesale acquisition cost for the infliximab originator Remicade, but this price does not include discounts to payers, providers, distributors, and other purchasing organizations.
For the first quarter of 2017, the payment limit set by the CMS for Inflectra is $100.306 per 10-mg unit and $82.218 for Remicade.
Various national and regional wholesalers across the country began receiving shipments of Inflectra in November 2016, according to Pfizer.
In conjunction with the availability of Inflectra, Pfizer announced its enCompass program, “a comprehensive reimbursement service and patient support program offering coding and reimbursement support for providers, copay assistance to eligible patients who have commercial insurance that covers Inflectra, and financial assistance for eligible uninsured and underinsured patients.”
The FDA approved Inflectra in April 2016 for all of the same indications as Remicade: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, plaque psoriasis, and ulcerative colitis.
Payment for the infliximab biosimilar drug Inflectra will now be covered by Medicare, the drug’s manufacturer, Pfizer, said in an announcement.
The Centers for Medicare & Medicaid Services (CMS) included Inflectra (infliximab-dyyb) in its January 2017 Average Sales Price pricing file, which went into effect Jan. 1, 2017. Pfizer said that Inflectra is priced at a 15% discount to the current wholesale acquisition cost for the infliximab originator Remicade, but this price does not include discounts to payers, providers, distributors, and other purchasing organizations.
For the first quarter of 2017, the payment limit set by the CMS for Inflectra is $100.306 per 10-mg unit and $82.218 for Remicade.
Various national and regional wholesalers across the country began receiving shipments of Inflectra in November 2016, according to Pfizer.
In conjunction with the availability of Inflectra, Pfizer announced its enCompass program, “a comprehensive reimbursement service and patient support program offering coding and reimbursement support for providers, copay assistance to eligible patients who have commercial insurance that covers Inflectra, and financial assistance for eligible uninsured and underinsured patients.”
The FDA approved Inflectra in April 2016 for all of the same indications as Remicade: rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, plaque psoriasis, and ulcerative colitis.
High-intensity statins cut amputations and mortality in PAD
NEW ORLEANS – High-intensity statin therapy in patients with peripheral artery disease was associated with significant reductions in amputations as well as mortality during up to 5 years of follow-up in the first large study to examine the relationship, Shipra Arya, MD, reported at the American Heart Association scientific sessions.
Low- or moderate-intensity statin therapy also improved survival compared to no statin, albeit to a significantly lesser magnitude than high-intensity therapy. But high- and low/intermediate-intensity statins were similarly effective in reducing amputation risk, according to Dr. Arya, a vascular surgeon at Emory University in Atlanta.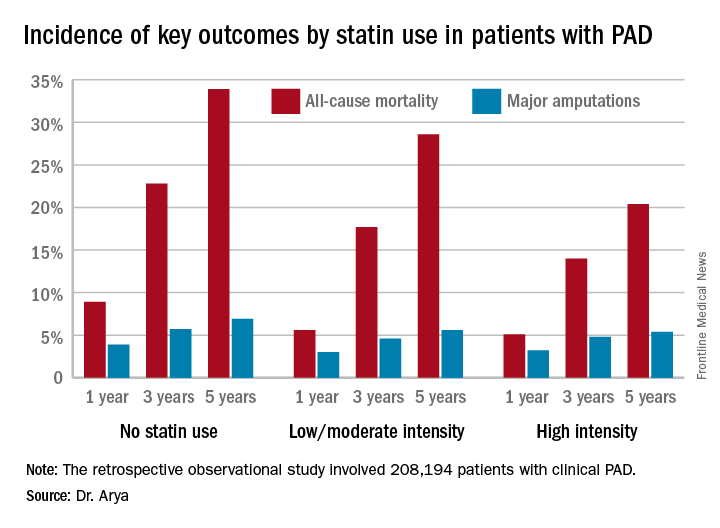
The 2013 AHA/American College of Cardiology treatment guidelines recommend high-intensity statins for all patients with clinical atherosclerotic disease, including those with PAD (Circulation. 2014 Jun 24;129[25 Suppl 2]:S1-45). (Updated PAD guidelines unveiled at the AHA meeting strongly recommend statin medication for all patients with PAD [Circulation. 2016 Nov 13. doi: 10.1161/CIR.0000000000000470]).However, the bulk of patients in Dr. Arya’s study were captured in the database prior to release of the 2013 guidelines. That may account for the sparse use of high-intensity statin therapy in the study cohort. Indeed, only 11.3% of the PAD patients were on a high-intensity statin. Another 36.2% were on moderate-intensity statin therapy, 3.5% were on low-intensity therapy, and 27.6% weren’t on a statin at all.
The relationship between statin therapy and mortality was strongly dose-dependent.
This study was funded by the AHA and the Atlanta Veterans Affairs Medical Center. Dr. Arya reported having no financial conflicts of interest.
NEW ORLEANS – High-intensity statin therapy in patients with peripheral artery disease was associated with significant reductions in amputations as well as mortality during up to 5 years of follow-up in the first large study to examine the relationship, Shipra Arya, MD, reported at the American Heart Association scientific sessions.
Low- or moderate-intensity statin therapy also improved survival compared to no statin, albeit to a significantly lesser magnitude than high-intensity therapy. But high- and low/intermediate-intensity statins were similarly effective in reducing amputation risk, according to Dr. Arya, a vascular surgeon at Emory University in Atlanta.
The 2013 AHA/American College of Cardiology treatment guidelines recommend high-intensity statins for all patients with clinical atherosclerotic disease, including those with PAD (Circulation. 2014 Jun 24;129[25 Suppl 2]:S1-45). (Updated PAD guidelines unveiled at the AHA meeting strongly recommend statin medication for all patients with PAD [Circulation. 2016 Nov 13. doi: 10.1161/CIR.0000000000000470]).However, the bulk of patients in Dr. Arya’s study were captured in the database prior to release of the 2013 guidelines. That may account for the sparse use of high-intensity statin therapy in the study cohort. Indeed, only 11.3% of the PAD patients were on a high-intensity statin. Another 36.2% were on moderate-intensity statin therapy, 3.5% were on low-intensity therapy, and 27.6% weren’t on a statin at all.
The relationship between statin therapy and mortality was strongly dose-dependent.
This study was funded by the AHA and the Atlanta Veterans Affairs Medical Center. Dr. Arya reported having no financial conflicts of interest.
NEW ORLEANS – High-intensity statin therapy in patients with peripheral artery disease was associated with significant reductions in amputations as well as mortality during up to 5 years of follow-up in the first large study to examine the relationship, Shipra Arya, MD, reported at the American Heart Association scientific sessions.
Low- or moderate-intensity statin therapy also improved survival compared to no statin, albeit to a significantly lesser magnitude than high-intensity therapy. But high- and low/intermediate-intensity statins were similarly effective in reducing amputation risk, according to Dr. Arya, a vascular surgeon at Emory University in Atlanta.
The 2013 AHA/American College of Cardiology treatment guidelines recommend high-intensity statins for all patients with clinical atherosclerotic disease, including those with PAD (Circulation. 2014 Jun 24;129[25 Suppl 2]:S1-45). (Updated PAD guidelines unveiled at the AHA meeting strongly recommend statin medication for all patients with PAD [Circulation. 2016 Nov 13. doi: 10.1161/CIR.0000000000000470]).However, the bulk of patients in Dr. Arya’s study were captured in the database prior to release of the 2013 guidelines. That may account for the sparse use of high-intensity statin therapy in the study cohort. Indeed, only 11.3% of the PAD patients were on a high-intensity statin. Another 36.2% were on moderate-intensity statin therapy, 3.5% were on low-intensity therapy, and 27.6% weren’t on a statin at all.
The relationship between statin therapy and mortality was strongly dose-dependent.
This study was funded by the AHA and the Atlanta Veterans Affairs Medical Center. Dr. Arya reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The 5-year all-cause mortality rate after diagnosis of peripheral artery disease was 20.4% in patients on high-intensity statin therapy, 28.6% in those on a low- or moderate-intensity statin, and 33.9% in patients not on a statin.
Data source: A retrospective observational study of 208,194 patients with clinical peripheral artery disease in the national Veterans Affairs database for 2003-2014.
Disclosures: The AHA and the Atlanta Veterans Affairs Medical Center funded the study. The presenter reported having no financial conflicts of interest.
USPSTF reaffirms need for folic acid supplements in pregnancy
The U.S. Preventive Services Task Force continues to recommend that all women planning or capable of pregnancy should take a daily supplement of 0.4-0.8 mg of folic acid to prevent neural tube defects in their offspring.
The task force “concludes with high certainty” that the benefits of such supplementation are substantial and the harms are minimal, according to the recommendation statement published online Jan. 10 in JAMA (2017;317[2]:183-9). The group based its updated recommendation on a systematic review of 24 studies performed since 2009 and involving 58,860 women. Although some newer studies have suggested that supplementation is no longer needed in this era of folic acid fortification of foods, “the USPSTF found no new substantial evidence ... that would lead to a change in its recommendation from 2009,” the researchers wrote.
The most recent data estimate that folic acid supplementation prevents neural tube defects in approximately 1,300 births each year.
This updated USPSTF recommendation is in accord with recommendations from the Health and Medicine Division of the National Academies (formerly the Institute of Medicine), the American College of Obstetricians and Gynecologists, the American Academy of Family Physicians, the U.S. Public Health Service, the Centers for Disease Control and Prevention, the American Academy of Pediatrics, the American Academy of Neurology, and the American College of Medical Genetics and Genomics.
This work was supported solely by the USPSTF, an independent voluntary group mandated by Congress to assess preventive care services and funded by the Agency for Healthcare Research and Quality.
The USPSTF recommendation that all women of childbearing age take folic acid supplements is a prudent one. Ideally, it will educate all women who are planning or capable of pregnancy to follow this recommendation and thereby reduce the risk of these severe birth defects in their infants.
Should the USPSTF recommendation be rejected because fortified food is already providing sufficient folic acid to prevent neural tube defects? No. Too little is known about how folic acid prevents neural tube defects. For example, it is not known whether the tissue stores of folate in the developing embryo or the availability of folate in the serum during the all-important few days of neural tube closure is most important. Habitual use of folic acid supplements is a more reliable method of ensuring adequate levels than is diet. In theory, a woman might not consume sufficient enriched cereal grains during the critical period of approximately 1 week when the neural tube is closing. Exactly when folate must be available also is not known. In addition, some popular diets, such as low carbohydrate or gluten free, may reduce exposure to grains, limiting folic acid intake.
James L. Mills, MD, is in the Division of Intramural Population Health Research at the Eunice Kennedy Shriver National Institute of Child Health and Human Development in Bethesda, Md. He reported having no relevant financial disclosures. These comments are adapted from an editorial accompanying the USPSTF report (JAMA. 2017;317 [2]:144-5).
The USPSTF recommendation that all women of childbearing age take folic acid supplements is a prudent one. Ideally, it will educate all women who are planning or capable of pregnancy to follow this recommendation and thereby reduce the risk of these severe birth defects in their infants.
Should the USPSTF recommendation be rejected because fortified food is already providing sufficient folic acid to prevent neural tube defects? No. Too little is known about how folic acid prevents neural tube defects. For example, it is not known whether the tissue stores of folate in the developing embryo or the availability of folate in the serum during the all-important few days of neural tube closure is most important. Habitual use of folic acid supplements is a more reliable method of ensuring adequate levels than is diet. In theory, a woman might not consume sufficient enriched cereal grains during the critical period of approximately 1 week when the neural tube is closing. Exactly when folate must be available also is not known. In addition, some popular diets, such as low carbohydrate or gluten free, may reduce exposure to grains, limiting folic acid intake.
James L. Mills, MD, is in the Division of Intramural Population Health Research at the Eunice Kennedy Shriver National Institute of Child Health and Human Development in Bethesda, Md. He reported having no relevant financial disclosures. These comments are adapted from an editorial accompanying the USPSTF report (JAMA. 2017;317 [2]:144-5).
The USPSTF recommendation that all women of childbearing age take folic acid supplements is a prudent one. Ideally, it will educate all women who are planning or capable of pregnancy to follow this recommendation and thereby reduce the risk of these severe birth defects in their infants.
Should the USPSTF recommendation be rejected because fortified food is already providing sufficient folic acid to prevent neural tube defects? No. Too little is known about how folic acid prevents neural tube defects. For example, it is not known whether the tissue stores of folate in the developing embryo or the availability of folate in the serum during the all-important few days of neural tube closure is most important. Habitual use of folic acid supplements is a more reliable method of ensuring adequate levels than is diet. In theory, a woman might not consume sufficient enriched cereal grains during the critical period of approximately 1 week when the neural tube is closing. Exactly when folate must be available also is not known. In addition, some popular diets, such as low carbohydrate or gluten free, may reduce exposure to grains, limiting folic acid intake.
James L. Mills, MD, is in the Division of Intramural Population Health Research at the Eunice Kennedy Shriver National Institute of Child Health and Human Development in Bethesda, Md. He reported having no relevant financial disclosures. These comments are adapted from an editorial accompanying the USPSTF report (JAMA. 2017;317 [2]:144-5).
The U.S. Preventive Services Task Force continues to recommend that all women planning or capable of pregnancy should take a daily supplement of 0.4-0.8 mg of folic acid to prevent neural tube defects in their offspring.
The task force “concludes with high certainty” that the benefits of such supplementation are substantial and the harms are minimal, according to the recommendation statement published online Jan. 10 in JAMA (2017;317[2]:183-9). The group based its updated recommendation on a systematic review of 24 studies performed since 2009 and involving 58,860 women. Although some newer studies have suggested that supplementation is no longer needed in this era of folic acid fortification of foods, “the USPSTF found no new substantial evidence ... that would lead to a change in its recommendation from 2009,” the researchers wrote.
The most recent data estimate that folic acid supplementation prevents neural tube defects in approximately 1,300 births each year.
This updated USPSTF recommendation is in accord with recommendations from the Health and Medicine Division of the National Academies (formerly the Institute of Medicine), the American College of Obstetricians and Gynecologists, the American Academy of Family Physicians, the U.S. Public Health Service, the Centers for Disease Control and Prevention, the American Academy of Pediatrics, the American Academy of Neurology, and the American College of Medical Genetics and Genomics.
This work was supported solely by the USPSTF, an independent voluntary group mandated by Congress to assess preventive care services and funded by the Agency for Healthcare Research and Quality.
The U.S. Preventive Services Task Force continues to recommend that all women planning or capable of pregnancy should take a daily supplement of 0.4-0.8 mg of folic acid to prevent neural tube defects in their offspring.
The task force “concludes with high certainty” that the benefits of such supplementation are substantial and the harms are minimal, according to the recommendation statement published online Jan. 10 in JAMA (2017;317[2]:183-9). The group based its updated recommendation on a systematic review of 24 studies performed since 2009 and involving 58,860 women. Although some newer studies have suggested that supplementation is no longer needed in this era of folic acid fortification of foods, “the USPSTF found no new substantial evidence ... that would lead to a change in its recommendation from 2009,” the researchers wrote.
The most recent data estimate that folic acid supplementation prevents neural tube defects in approximately 1,300 births each year.
This updated USPSTF recommendation is in accord with recommendations from the Health and Medicine Division of the National Academies (formerly the Institute of Medicine), the American College of Obstetricians and Gynecologists, the American Academy of Family Physicians, the U.S. Public Health Service, the Centers for Disease Control and Prevention, the American Academy of Pediatrics, the American Academy of Neurology, and the American College of Medical Genetics and Genomics.
This work was supported solely by the USPSTF, an independent voluntary group mandated by Congress to assess preventive care services and funded by the Agency for Healthcare Research and Quality.
FROM JAMA
Key clinical point:
Major finding: Folic acid supplementation prevents neural tube defects in an estimated 1,300 births each year in the United States.
Data source: A systematic review of 24 studies (involving 58,860 women) that were performed since 2009 regarding the benefits and harms of folic acid supplementation.
Disclosures: This work was supported solely by the USPSTF, an independent voluntary group mandated by Congress to assess preventive care services and funded by the Agency for Healthcare Research and Quality.
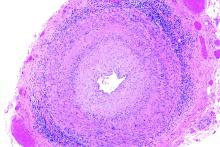
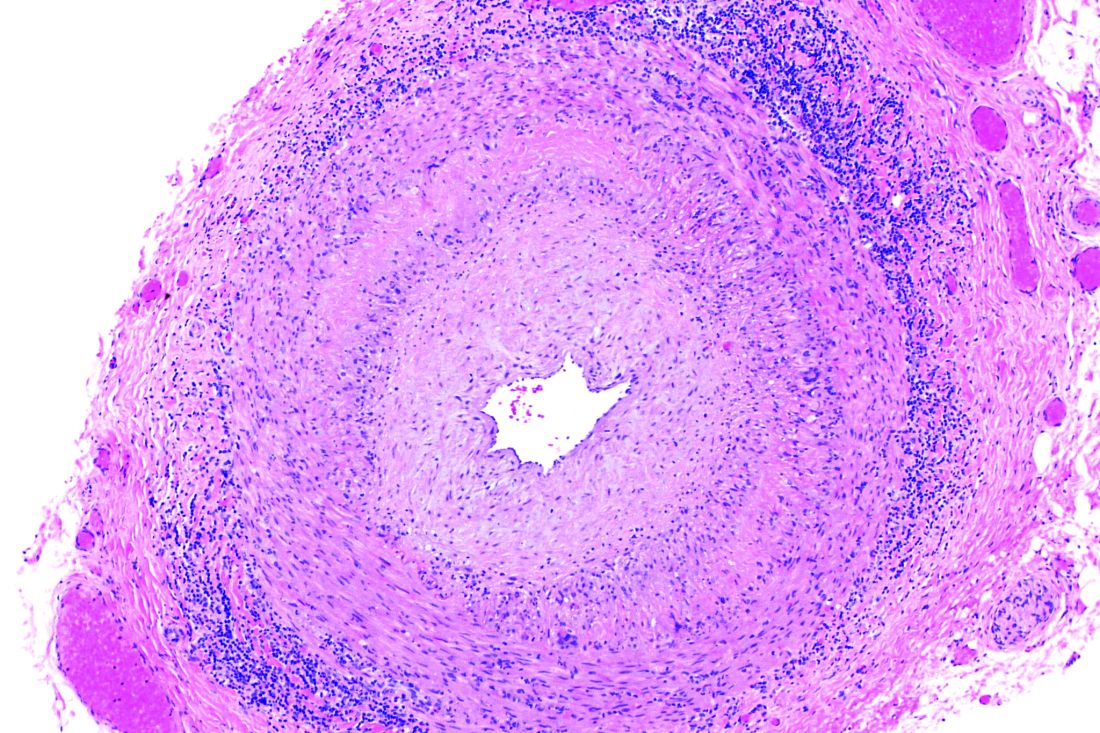
Giant cell arteritis independently raises risk for venous thromboembolism
The risk of venous thromboembolism increases markedly shortly before the diagnosis of giant cell arteritis regardless of glucocorticoid exposure, peaks at the time of diagnosis, and then progressively declines, according to a matched cohort review involving more than 6,000 arteritis patients.
It’s not been clear until now if the recently recognized risk of venous thromboembolism (VTE) in giant cell arteritis (GCA) was due to the disease itself, or the glucocorticoids used to treat it. “Because inflammation in GCA spares the venous circulation, our finding that patients are at greatest risk of VTE in the period surrounding GCA diagnosis (when inflammation is at its highest level), and the demonstration that this risk is not associated with the use of glucocorticoids, suggest that immunothrombosis could play a pathogenic role,” said investigators led by Sebastian Unizony, MD, of Massachusetts General Hospital, Boston (Arthritis Rheumatol. 2017 Jan;69[1]:176-84).
The report was short on advice about what to do to prevent VTE in GCA, but the investigators did recommend “adequate monitoring ... for early recognition of this potentially serious complication.”
The team used a British medical record database covering 1990-2013 to compare 6,441 patients with new-onset GCA to 63,985 controls without GCA matched for age, sex, and date of study entry. VTE was defined as pulmonary embolism and/or deep vein thrombosis.
The incidence of VTE shortly before diagnosis was 4.2 cases per 1,000 person-years in the GCA group, but 2.3 cases per 1,000 person-years among controls. It was about the same when the analysis was limited to GCA patients not exposed to oral glucocorticoids before diagnosis: 4.0 cases versus 2.2 cases in the control group per 1,000 person-years. The finding was key to the conclusion that GCA is an independent VTE risk factor.
During the 12, 9, 6, and 3 months leading up to GCA diagnosis, the relative risks for VTE among patients not treated with glucocorticoids – versus controls – were 1.8, 2.2, 2.4, and 3.6. In the first 3, 6, 12, 24, 48, and 96 months after GCA diagnosis, when virtually all patients were on glucocorticoids at least for the first 6 months, the relative risks for VTE were 9.9, 7.7, 5.9, 4.4, 3.3, 2.4; the last risk score of 2.4 indicated that GCA patients were still slightly more likely than controls to have a VTE even 8 years after diagnosis.
The mean age of patients in the study was 73 years, and 70% of the subjects were women. GCA patients were more likely than were controls to be smokers and to have cardiovascular disease. Also, a greater proportion of GCA patients used aspirin and had recent surgery and hospitalizations. There was no difference in body mass index (mean in both groups 27 kg/m2) or the prevalence of fracture, trauma, or cancer between the groups.
The National Institutes of Health funded the work. There was no disclosure information in the report.
Whether these findings have implications for treatment is unclear. Should a patient with GCA who sustains a VTE early in the course of disease receive anticoagulation short term, with the thought that the VTE was provoked by a risk factor that has been neutralized? Or should treatment be long term, out of concern that the risk factor is still present? Does this finding bear on the controversial question of whether a patient with GCA should receive aspirin?
[A] robust finding of the analysis is that the risk of a first VTE declines steadily over at least the first 2 years after diagnosis of GCA. However, the problem of distinguishing the effects of disease from the effects of treatment has returned. All patients with GCA are now receiving corticosteroids, at least during the period of very high risk in the first 6 months after diagnosis, and one can expect that the average severity of inflammation and average dose of prednisone/prednisolone will decline in parallel. The steadily declining risk of VTE for at least 1 year after diagnosis suggests that both GCA and corticosteroids increase the risk of VTE. It remains impossible to prove or disprove that hypothesis or to estimate the independent risks conferred by the disease and its treatment.
Having GCA probably increases the risk of VTE at least for the first 24 months after diagnosis and the beginning of treatment, but after 24 months, it is unclear. The clinician will still need to make a guess regarding duration of anticoagulation.
Paul Monach, MD, PhD, is a vasculitis specialist at Boston University. He made his comments in an accompanying editorial (Arthritis Rheumatol. 2017 Jan;69[1]:3-5).
Whether these findings have implications for treatment is unclear. Should a patient with GCA who sustains a VTE early in the course of disease receive anticoagulation short term, with the thought that the VTE was provoked by a risk factor that has been neutralized? Or should treatment be long term, out of concern that the risk factor is still present? Does this finding bear on the controversial question of whether a patient with GCA should receive aspirin?
[A] robust finding of the analysis is that the risk of a first VTE declines steadily over at least the first 2 years after diagnosis of GCA. However, the problem of distinguishing the effects of disease from the effects of treatment has returned. All patients with GCA are now receiving corticosteroids, at least during the period of very high risk in the first 6 months after diagnosis, and one can expect that the average severity of inflammation and average dose of prednisone/prednisolone will decline in parallel. The steadily declining risk of VTE for at least 1 year after diagnosis suggests that both GCA and corticosteroids increase the risk of VTE. It remains impossible to prove or disprove that hypothesis or to estimate the independent risks conferred by the disease and its treatment.
Having GCA probably increases the risk of VTE at least for the first 24 months after diagnosis and the beginning of treatment, but after 24 months, it is unclear. The clinician will still need to make a guess regarding duration of anticoagulation.
Paul Monach, MD, PhD, is a vasculitis specialist at Boston University. He made his comments in an accompanying editorial (Arthritis Rheumatol. 2017 Jan;69[1]:3-5).
Whether these findings have implications for treatment is unclear. Should a patient with GCA who sustains a VTE early in the course of disease receive anticoagulation short term, with the thought that the VTE was provoked by a risk factor that has been neutralized? Or should treatment be long term, out of concern that the risk factor is still present? Does this finding bear on the controversial question of whether a patient with GCA should receive aspirin?
[A] robust finding of the analysis is that the risk of a first VTE declines steadily over at least the first 2 years after diagnosis of GCA. However, the problem of distinguishing the effects of disease from the effects of treatment has returned. All patients with GCA are now receiving corticosteroids, at least during the period of very high risk in the first 6 months after diagnosis, and one can expect that the average severity of inflammation and average dose of prednisone/prednisolone will decline in parallel. The steadily declining risk of VTE for at least 1 year after diagnosis suggests that both GCA and corticosteroids increase the risk of VTE. It remains impossible to prove or disprove that hypothesis or to estimate the independent risks conferred by the disease and its treatment.
Having GCA probably increases the risk of VTE at least for the first 24 months after diagnosis and the beginning of treatment, but after 24 months, it is unclear. The clinician will still need to make a guess regarding duration of anticoagulation.
Paul Monach, MD, PhD, is a vasculitis specialist at Boston University. He made his comments in an accompanying editorial (Arthritis Rheumatol. 2017 Jan;69[1]:3-5).
The risk of venous thromboembolism increases markedly shortly before the diagnosis of giant cell arteritis regardless of glucocorticoid exposure, peaks at the time of diagnosis, and then progressively declines, according to a matched cohort review involving more than 6,000 arteritis patients.
It’s not been clear until now if the recently recognized risk of venous thromboembolism (VTE) in giant cell arteritis (GCA) was due to the disease itself, or the glucocorticoids used to treat it. “Because inflammation in GCA spares the venous circulation, our finding that patients are at greatest risk of VTE in the period surrounding GCA diagnosis (when inflammation is at its highest level), and the demonstration that this risk is not associated with the use of glucocorticoids, suggest that immunothrombosis could play a pathogenic role,” said investigators led by Sebastian Unizony, MD, of Massachusetts General Hospital, Boston (Arthritis Rheumatol. 2017 Jan;69[1]:176-84).
The report was short on advice about what to do to prevent VTE in GCA, but the investigators did recommend “adequate monitoring ... for early recognition of this potentially serious complication.”
The team used a British medical record database covering 1990-2013 to compare 6,441 patients with new-onset GCA to 63,985 controls without GCA matched for age, sex, and date of study entry. VTE was defined as pulmonary embolism and/or deep vein thrombosis.
The incidence of VTE shortly before diagnosis was 4.2 cases per 1,000 person-years in the GCA group, but 2.3 cases per 1,000 person-years among controls. It was about the same when the analysis was limited to GCA patients not exposed to oral glucocorticoids before diagnosis: 4.0 cases versus 2.2 cases in the control group per 1,000 person-years. The finding was key to the conclusion that GCA is an independent VTE risk factor.
During the 12, 9, 6, and 3 months leading up to GCA diagnosis, the relative risks for VTE among patients not treated with glucocorticoids – versus controls – were 1.8, 2.2, 2.4, and 3.6. In the first 3, 6, 12, 24, 48, and 96 months after GCA diagnosis, when virtually all patients were on glucocorticoids at least for the first 6 months, the relative risks for VTE were 9.9, 7.7, 5.9, 4.4, 3.3, 2.4; the last risk score of 2.4 indicated that GCA patients were still slightly more likely than controls to have a VTE even 8 years after diagnosis.
The mean age of patients in the study was 73 years, and 70% of the subjects were women. GCA patients were more likely than were controls to be smokers and to have cardiovascular disease. Also, a greater proportion of GCA patients used aspirin and had recent surgery and hospitalizations. There was no difference in body mass index (mean in both groups 27 kg/m2) or the prevalence of fracture, trauma, or cancer between the groups.
The National Institutes of Health funded the work. There was no disclosure information in the report.
The risk of venous thromboembolism increases markedly shortly before the diagnosis of giant cell arteritis regardless of glucocorticoid exposure, peaks at the time of diagnosis, and then progressively declines, according to a matched cohort review involving more than 6,000 arteritis patients.
It’s not been clear until now if the recently recognized risk of venous thromboembolism (VTE) in giant cell arteritis (GCA) was due to the disease itself, or the glucocorticoids used to treat it. “Because inflammation in GCA spares the venous circulation, our finding that patients are at greatest risk of VTE in the period surrounding GCA diagnosis (when inflammation is at its highest level), and the demonstration that this risk is not associated with the use of glucocorticoids, suggest that immunothrombosis could play a pathogenic role,” said investigators led by Sebastian Unizony, MD, of Massachusetts General Hospital, Boston (Arthritis Rheumatol. 2017 Jan;69[1]:176-84).
The report was short on advice about what to do to prevent VTE in GCA, but the investigators did recommend “adequate monitoring ... for early recognition of this potentially serious complication.”
The team used a British medical record database covering 1990-2013 to compare 6,441 patients with new-onset GCA to 63,985 controls without GCA matched for age, sex, and date of study entry. VTE was defined as pulmonary embolism and/or deep vein thrombosis.
The incidence of VTE shortly before diagnosis was 4.2 cases per 1,000 person-years in the GCA group, but 2.3 cases per 1,000 person-years among controls. It was about the same when the analysis was limited to GCA patients not exposed to oral glucocorticoids before diagnosis: 4.0 cases versus 2.2 cases in the control group per 1,000 person-years. The finding was key to the conclusion that GCA is an independent VTE risk factor.
During the 12, 9, 6, and 3 months leading up to GCA diagnosis, the relative risks for VTE among patients not treated with glucocorticoids – versus controls – were 1.8, 2.2, 2.4, and 3.6. In the first 3, 6, 12, 24, 48, and 96 months after GCA diagnosis, when virtually all patients were on glucocorticoids at least for the first 6 months, the relative risks for VTE were 9.9, 7.7, 5.9, 4.4, 3.3, 2.4; the last risk score of 2.4 indicated that GCA patients were still slightly more likely than controls to have a VTE even 8 years after diagnosis.
The mean age of patients in the study was 73 years, and 70% of the subjects were women. GCA patients were more likely than were controls to be smokers and to have cardiovascular disease. Also, a greater proportion of GCA patients used aspirin and had recent surgery and hospitalizations. There was no difference in body mass index (mean in both groups 27 kg/m2) or the prevalence of fracture, trauma, or cancer between the groups.
The National Institutes of Health funded the work. There was no disclosure information in the report.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point:
Major finding: In the 12, 9, 6, and 3 months before GCA diagnosis, the relative risks for VTE among patients not treated with glucocorticoids – versus controls without GCA – were 1.8, 2.2, 2.4, and 3.6.
Data source: Matched cohort review involving more than 6,000 arteritis patients.
Disclosures: The National Institutes of Health funded the work. There was no disclosure information in the report.
Medicare failed to recover up to $125 million in overpayments, records show
Six years ago, federal health officials were confident they could save taxpayers hundreds of millions of dollars annually by auditing private Medicare Advantage insurance plans that allegedly overcharged the government for medical services.
An initial round of audits found that Medicare had potentially overpaid five of the health plans $128 million in 2007 alone, according to confidential government documents released recently in response to a public records request and lawsuit.
But officials never recovered most of that money. Under intense pressure from the health insurance industry, the Centers for Medicare and Medicaid Services quietly backed off their repayment demands and settled the audits in 2012 for just under $3.4 million – shortchanging taxpayers by up to $125 million in possible overcharges just for 2007.
Medicare Advantage is a popular alternative to traditional Medicare. The privately run health plans have enrolled more than 17 million elderly and disabled people – about a third of those eligible for Medicare – at a cost to taxpayers of more than $150 billion a year. And while the plans generally enjoy strong support in Congress, there are critics.
“It’s unclear why the Obama Administration allowed CMS to overpromise and under-deliver so badly on collecting these overpayments,” Sen. Chuck Grassley, R-Iowa, told Kaiser Health News in an email response to the findings.
He said CMS “should account for why this process seems to be so broken and why it can’t seem to fix it, despite recommendations to do so. The taxpayers depend on getting this process right.”
The failure to collect also alarmed Steve Ellis, vice president of the budget watchdog group Taxpayers for Common Sense in Washington.
“They need to put up a bigger and stronger fight to make sure these programs are operated on the straight and narrow,” Mr. Ellis said.
Yet outside of public view, federal officials have been losing a high-stakes battle to curb widespread billing errors by Medicare Advantage plans, according to the records obtained through a Freedom of Information Act lawsuit filed by the Center for Public Integrity.
The Center for Public Integrity first disclosed in 2014 that billions of tax dollars are wasted annually partly because some health plans appear to exaggerate how sick their patients are, a practice known in health care circles as “upcoding.”
Last August, the investigative journalism group reported that 35 of 37 health plans CMS has audited overcharged Medicare, often by overstating the severity of medical conditions such as diabetes and depression.
The newly released CMS records identify the companies chosen for the initial 2007 audits as a Florida Humana plan, a Washington state subsidiary of United Healthcare called PacifiCare, an Aetna plan in New Jersey, and an Independence Blue Cross plan in the Philadelphia area.
The fifth one focused on a Lovelace Medicare plan in New Mexico, which has since been acquired by Blue Cross.
Each of the five audits, which took more than 2 years to complete, unearthed significant – and costly – billing mistakes, though the plans disputed them.
For example, auditors couldn’t confirm that one-third of the diseases the health plans had been paid to treat actually existed, mostly because patient records lacked “sufficient documentation of a diagnosis.”
Overall, Medicare paid the wrong amount for nearly two-thirds of patients whose records were examined; all five plans were far more likely to charge too much than too little. For one in five patients, the overcharges were $5,000 or more for the year, according to the audits. None of the plans would discuss the findings.
As preliminary results of the audits started to roll in, CMS officials outlined steps to recover more than $128 million from the five plans at a confidential agency briefing in August 2010, according to a policy memo prepared for the meeting. The records don’t indicate who attended.
That day, CMS set Humana’s payment error at $33.5 million, PacifiCare at $20.2 million, Aetna at $27.6 million, Independence Blue Cross at nearly $34 million and Lovelace at just under $13 million. Those estimates were based on extrapolation of a sample of cases examined at each plan.
CMS “has developed a process for moving forward with payment recovery,” according to a briefing paper from the 2010 meeting.
But that process fizzled after 2 years of haggling with the plans and insurance industry representatives, who argued the audits were flawed and the results unreliable. In August 2012, CMS gave in and notified the plans it would settle for a few cents on the dollar.
“Given this was a new process, the decision was made at the time to tie repayments to the actual claims reviewed as part of the 2007 pilot audit,” said CMS spokesman Aaron Albright. “For subsequent audits, we said we intended to determine repayments by extrapolating the error rate of the sample of claims reviewed to all claims under the contract.” Mr. Albright said more of the audits are underway. Allowing the insurers to dodge liability dealt a serious blow to the government’s efforts to crack down on billing abuses – a setback one taxpayer advocate called alarming.
“That’s a very bad way to operate the system.” said Patrick Burns, acting executive director and president of Taxpayers Against Fraud in Washington, on hearing of the outcome. “Nobody is held accountable.”
Indeed, CMS kept the settlement terms under wraps until 2015, after an inquiry by Sen. Grassley. The senator had requested details about Medicare Advantage fraud controls in response to articles published by the Center for Public Integrity.
In a July 31, 2015 letter to Sen. Grassley, CMS Acting Administrator Andy Slavitt attached a table that showed the five plans repaid just under $3.4 million. The letter didn’t mention the earlier estimate that the government was due $128 million. Sen. Grassley said it should not have taken the FOIA lawsuit to make that information available to the public.
“Perhaps adding insult to injury, these numbers might never have seen the light of day without a lengthy lawsuit,” Sen. Grassley said this week.
Paying based on risk scores
When Congress created the current Medicare Advantage program in 2003, it devised a new way to pay the health plans.
The method, phased in starting in 2004, seemed simple enough: Pay higher rates for sicker patients and less for people in good health using a formula called a risk score.
But CMS officials soon realized that risk scores rose much faster at some plans than others, a possible sign of upcoding, or other billing irregularities, records show. These overcharges topped $4 billion in 2005, one CMS study found.
The special audits, called Risk Adjustment Data Validation, or RADV, were designed to identify, and hold accountable, health plans that couldn’t justify their fees with supporting medical evidence.
Until these audits, CMS “pretty much went on the honor system with the plans,” an unnamed agency official wrote in an undated presentation.
In the five 2007 pilot audits, two sets of auditors inspected medical records for a random sample of 201 patients at each plan. If the medical chart didn’t properly document that a patient had the illnesses the plan had reported, Medicare wanted a refund. Auditors gave the plans the benefit of the doubt when auditors couldn’t agree, according to the CMS briefing paper.
Finally, CMS applied a standard technique used in fraud investigations in which the payment error rate is extrapolated across the entire health plan, which greatly multiplies the amount due. CMS said it was conservative in assessing the penalties and allowed the plans to appeal.
Appeals or no, the health plans recoiled at the prospect they could be on the hook for millions of dollars they hadn’t budgeted for and didn’t believe they owed. The actual 2007 overage for the 201 Humana patients, for example, was $477,235. Once extrapolated, it soared to $33.5 million.
Michael S. Adelberg, a former CMS official who is now an industry consultant in Washington, said that in retrospect the audit process was “probably rushed.”
Mr. Adelberg said the audits “raised strong industry concerns” on a variety of fronts, from whether CMS had the legal authority to conduct them to the soundness of their methods. CMS stands by its audit techniques and has defended RADV as the only way it can assure plans bill honestly.
Yet agency records released through the FOIA case suggest CMS lacked the will to press ahead with extrapolated audits for Medicare Advantage plans given the fierce industry backlash – even though they do so in overpayment cases targeting other types of medical providers.
One confidential CMS presentation dated March 30, 2011, notes that officials had received more than 500 comments expressing “significant resistance” to the RADV audits.
The presentation goes on to say the audit program’s success depended on its “ability to address the challenges raised.”
CMS didn’t overcome those challenges. Instead, it agreed to settle the five initial audits for $3.4 million, just what it found in the patient files it reviewed – without the extrapolations. And the center did the same for 32 additional 2007 audits, which officials had predicted would refund up to $800 million to the federal treasury. In the end, CMS wound up with $10.3 million from the 32 plans.
The RADV program’s shortcomings, though little known to the public, haven’t gone totally unnoticed. The program was the target of a sharply critical May 2016 report by the Government Accountability Office, which noted that Medicare Advantage plans have overbilled the government by billions of dollars, but rarely been forced to repay the money or face other consequences.
The GAO, the watchdog arm of Congress, called for “fundamental improvements.” The watchdogs also found that CMS has spent about $117 million on the audits, but recouped just under $14 million.
Government officials didn’t dispute that the RADV process had taken far too long and yielded way too little. But while CMS has resumed extrapolated audits, there’s little evidence it is speeding things up.
CMS expected to complete extrapolated audits for payments made in 2011 and finish the job in early 2014, agency records show.
But it has yet to do so. In late December, an agency spokesman said he had no new information about when the 2011 audits would be finished or how much the government would collect.
While the industry awaits the results, it has hardly warmed to the process.
America’s Health Insurance Plans, an industry trade group, argued in a June 2016 position paper that RADV was “not yet stable and reliable,” adding that the audits “could disrupt the care being provided by plans that are working hard to meet the needs of their enrollees.”
John Gorman, a former government health care official and current industry consultant, said he expects RADV to forge ahead under the incoming administration. But he predicted efforts to collect overpayments will “slow down” because the Trump team will prove to be “more sympathetic” to business interests than the Obama administration. The Trump transition office did not respond to a request for comment.
Mr. Gorman said that while career civil servants at CMS decide which plans get audited, how much to assess the health plans as a result rests with “political appointees” who are susceptible to industry lobbying, which he termed “the old Potomac two- step.”
But Sen. Grassley said he is determined to keep a close eye on the audit program. “I intend to press the incoming administration on holding CMS accountable for overpayments that harm taxpayers,” he said.
Taxpayer advocate Mr. Ellis said with so much public money at stake, the government needs to step up its game.
“You can presume that the more people get away with overpayments the more they are going to take,” he said. “As the program gets bigger the problem get bigger.”
This story is a collaboration between Kaiser Health News, a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation, and the Center for Public Integrity.
Six years ago, federal health officials were confident they could save taxpayers hundreds of millions of dollars annually by auditing private Medicare Advantage insurance plans that allegedly overcharged the government for medical services.
An initial round of audits found that Medicare had potentially overpaid five of the health plans $128 million in 2007 alone, according to confidential government documents released recently in response to a public records request and lawsuit.
But officials never recovered most of that money. Under intense pressure from the health insurance industry, the Centers for Medicare and Medicaid Services quietly backed off their repayment demands and settled the audits in 2012 for just under $3.4 million – shortchanging taxpayers by up to $125 million in possible overcharges just for 2007.
Medicare Advantage is a popular alternative to traditional Medicare. The privately run health plans have enrolled more than 17 million elderly and disabled people – about a third of those eligible for Medicare – at a cost to taxpayers of more than $150 billion a year. And while the plans generally enjoy strong support in Congress, there are critics.
“It’s unclear why the Obama Administration allowed CMS to overpromise and under-deliver so badly on collecting these overpayments,” Sen. Chuck Grassley, R-Iowa, told Kaiser Health News in an email response to the findings.
He said CMS “should account for why this process seems to be so broken and why it can’t seem to fix it, despite recommendations to do so. The taxpayers depend on getting this process right.”
The failure to collect also alarmed Steve Ellis, vice president of the budget watchdog group Taxpayers for Common Sense in Washington.
“They need to put up a bigger and stronger fight to make sure these programs are operated on the straight and narrow,” Mr. Ellis said.
Yet outside of public view, federal officials have been losing a high-stakes battle to curb widespread billing errors by Medicare Advantage plans, according to the records obtained through a Freedom of Information Act lawsuit filed by the Center for Public Integrity.
The Center for Public Integrity first disclosed in 2014 that billions of tax dollars are wasted annually partly because some health plans appear to exaggerate how sick their patients are, a practice known in health care circles as “upcoding.”
Last August, the investigative journalism group reported that 35 of 37 health plans CMS has audited overcharged Medicare, often by overstating the severity of medical conditions such as diabetes and depression.
The newly released CMS records identify the companies chosen for the initial 2007 audits as a Florida Humana plan, a Washington state subsidiary of United Healthcare called PacifiCare, an Aetna plan in New Jersey, and an Independence Blue Cross plan in the Philadelphia area.
The fifth one focused on a Lovelace Medicare plan in New Mexico, which has since been acquired by Blue Cross.
Each of the five audits, which took more than 2 years to complete, unearthed significant – and costly – billing mistakes, though the plans disputed them.
For example, auditors couldn’t confirm that one-third of the diseases the health plans had been paid to treat actually existed, mostly because patient records lacked “sufficient documentation of a diagnosis.”
Overall, Medicare paid the wrong amount for nearly two-thirds of patients whose records were examined; all five plans were far more likely to charge too much than too little. For one in five patients, the overcharges were $5,000 or more for the year, according to the audits. None of the plans would discuss the findings.
As preliminary results of the audits started to roll in, CMS officials outlined steps to recover more than $128 million from the five plans at a confidential agency briefing in August 2010, according to a policy memo prepared for the meeting. The records don’t indicate who attended.
That day, CMS set Humana’s payment error at $33.5 million, PacifiCare at $20.2 million, Aetna at $27.6 million, Independence Blue Cross at nearly $34 million and Lovelace at just under $13 million. Those estimates were based on extrapolation of a sample of cases examined at each plan.
CMS “has developed a process for moving forward with payment recovery,” according to a briefing paper from the 2010 meeting.
But that process fizzled after 2 years of haggling with the plans and insurance industry representatives, who argued the audits were flawed and the results unreliable. In August 2012, CMS gave in and notified the plans it would settle for a few cents on the dollar.
“Given this was a new process, the decision was made at the time to tie repayments to the actual claims reviewed as part of the 2007 pilot audit,” said CMS spokesman Aaron Albright. “For subsequent audits, we said we intended to determine repayments by extrapolating the error rate of the sample of claims reviewed to all claims under the contract.” Mr. Albright said more of the audits are underway. Allowing the insurers to dodge liability dealt a serious blow to the government’s efforts to crack down on billing abuses – a setback one taxpayer advocate called alarming.
“That’s a very bad way to operate the system.” said Patrick Burns, acting executive director and president of Taxpayers Against Fraud in Washington, on hearing of the outcome. “Nobody is held accountable.”
Indeed, CMS kept the settlement terms under wraps until 2015, after an inquiry by Sen. Grassley. The senator had requested details about Medicare Advantage fraud controls in response to articles published by the Center for Public Integrity.
In a July 31, 2015 letter to Sen. Grassley, CMS Acting Administrator Andy Slavitt attached a table that showed the five plans repaid just under $3.4 million. The letter didn’t mention the earlier estimate that the government was due $128 million. Sen. Grassley said it should not have taken the FOIA lawsuit to make that information available to the public.
“Perhaps adding insult to injury, these numbers might never have seen the light of day without a lengthy lawsuit,” Sen. Grassley said this week.
Paying based on risk scores
When Congress created the current Medicare Advantage program in 2003, it devised a new way to pay the health plans.
The method, phased in starting in 2004, seemed simple enough: Pay higher rates for sicker patients and less for people in good health using a formula called a risk score.
But CMS officials soon realized that risk scores rose much faster at some plans than others, a possible sign of upcoding, or other billing irregularities, records show. These overcharges topped $4 billion in 2005, one CMS study found.
The special audits, called Risk Adjustment Data Validation, or RADV, were designed to identify, and hold accountable, health plans that couldn’t justify their fees with supporting medical evidence.
Until these audits, CMS “pretty much went on the honor system with the plans,” an unnamed agency official wrote in an undated presentation.
In the five 2007 pilot audits, two sets of auditors inspected medical records for a random sample of 201 patients at each plan. If the medical chart didn’t properly document that a patient had the illnesses the plan had reported, Medicare wanted a refund. Auditors gave the plans the benefit of the doubt when auditors couldn’t agree, according to the CMS briefing paper.
Finally, CMS applied a standard technique used in fraud investigations in which the payment error rate is extrapolated across the entire health plan, which greatly multiplies the amount due. CMS said it was conservative in assessing the penalties and allowed the plans to appeal.
Appeals or no, the health plans recoiled at the prospect they could be on the hook for millions of dollars they hadn’t budgeted for and didn’t believe they owed. The actual 2007 overage for the 201 Humana patients, for example, was $477,235. Once extrapolated, it soared to $33.5 million.
Michael S. Adelberg, a former CMS official who is now an industry consultant in Washington, said that in retrospect the audit process was “probably rushed.”
Mr. Adelberg said the audits “raised strong industry concerns” on a variety of fronts, from whether CMS had the legal authority to conduct them to the soundness of their methods. CMS stands by its audit techniques and has defended RADV as the only way it can assure plans bill honestly.
Yet agency records released through the FOIA case suggest CMS lacked the will to press ahead with extrapolated audits for Medicare Advantage plans given the fierce industry backlash – even though they do so in overpayment cases targeting other types of medical providers.
One confidential CMS presentation dated March 30, 2011, notes that officials had received more than 500 comments expressing “significant resistance” to the RADV audits.
The presentation goes on to say the audit program’s success depended on its “ability to address the challenges raised.”
CMS didn’t overcome those challenges. Instead, it agreed to settle the five initial audits for $3.4 million, just what it found in the patient files it reviewed – without the extrapolations. And the center did the same for 32 additional 2007 audits, which officials had predicted would refund up to $800 million to the federal treasury. In the end, CMS wound up with $10.3 million from the 32 plans.
The RADV program’s shortcomings, though little known to the public, haven’t gone totally unnoticed. The program was the target of a sharply critical May 2016 report by the Government Accountability Office, which noted that Medicare Advantage plans have overbilled the government by billions of dollars, but rarely been forced to repay the money or face other consequences.
The GAO, the watchdog arm of Congress, called for “fundamental improvements.” The watchdogs also found that CMS has spent about $117 million on the audits, but recouped just under $14 million.
Government officials didn’t dispute that the RADV process had taken far too long and yielded way too little. But while CMS has resumed extrapolated audits, there’s little evidence it is speeding things up.
CMS expected to complete extrapolated audits for payments made in 2011 and finish the job in early 2014, agency records show.
But it has yet to do so. In late December, an agency spokesman said he had no new information about when the 2011 audits would be finished or how much the government would collect.
While the industry awaits the results, it has hardly warmed to the process.
America’s Health Insurance Plans, an industry trade group, argued in a June 2016 position paper that RADV was “not yet stable and reliable,” adding that the audits “could disrupt the care being provided by plans that are working hard to meet the needs of their enrollees.”
John Gorman, a former government health care official and current industry consultant, said he expects RADV to forge ahead under the incoming administration. But he predicted efforts to collect overpayments will “slow down” because the Trump team will prove to be “more sympathetic” to business interests than the Obama administration. The Trump transition office did not respond to a request for comment.
Mr. Gorman said that while career civil servants at CMS decide which plans get audited, how much to assess the health plans as a result rests with “political appointees” who are susceptible to industry lobbying, which he termed “the old Potomac two- step.”
But Sen. Grassley said he is determined to keep a close eye on the audit program. “I intend to press the incoming administration on holding CMS accountable for overpayments that harm taxpayers,” he said.
Taxpayer advocate Mr. Ellis said with so much public money at stake, the government needs to step up its game.
“You can presume that the more people get away with overpayments the more they are going to take,” he said. “As the program gets bigger the problem get bigger.”
This story is a collaboration between Kaiser Health News, a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation, and the Center for Public Integrity.
Six years ago, federal health officials were confident they could save taxpayers hundreds of millions of dollars annually by auditing private Medicare Advantage insurance plans that allegedly overcharged the government for medical services.
An initial round of audits found that Medicare had potentially overpaid five of the health plans $128 million in 2007 alone, according to confidential government documents released recently in response to a public records request and lawsuit.
But officials never recovered most of that money. Under intense pressure from the health insurance industry, the Centers for Medicare and Medicaid Services quietly backed off their repayment demands and settled the audits in 2012 for just under $3.4 million – shortchanging taxpayers by up to $125 million in possible overcharges just for 2007.
Medicare Advantage is a popular alternative to traditional Medicare. The privately run health plans have enrolled more than 17 million elderly and disabled people – about a third of those eligible for Medicare – at a cost to taxpayers of more than $150 billion a year. And while the plans generally enjoy strong support in Congress, there are critics.
“It’s unclear why the Obama Administration allowed CMS to overpromise and under-deliver so badly on collecting these overpayments,” Sen. Chuck Grassley, R-Iowa, told Kaiser Health News in an email response to the findings.
He said CMS “should account for why this process seems to be so broken and why it can’t seem to fix it, despite recommendations to do so. The taxpayers depend on getting this process right.”
The failure to collect also alarmed Steve Ellis, vice president of the budget watchdog group Taxpayers for Common Sense in Washington.
“They need to put up a bigger and stronger fight to make sure these programs are operated on the straight and narrow,” Mr. Ellis said.
Yet outside of public view, federal officials have been losing a high-stakes battle to curb widespread billing errors by Medicare Advantage plans, according to the records obtained through a Freedom of Information Act lawsuit filed by the Center for Public Integrity.
The Center for Public Integrity first disclosed in 2014 that billions of tax dollars are wasted annually partly because some health plans appear to exaggerate how sick their patients are, a practice known in health care circles as “upcoding.”
Last August, the investigative journalism group reported that 35 of 37 health plans CMS has audited overcharged Medicare, often by overstating the severity of medical conditions such as diabetes and depression.
The newly released CMS records identify the companies chosen for the initial 2007 audits as a Florida Humana plan, a Washington state subsidiary of United Healthcare called PacifiCare, an Aetna plan in New Jersey, and an Independence Blue Cross plan in the Philadelphia area.
The fifth one focused on a Lovelace Medicare plan in New Mexico, which has since been acquired by Blue Cross.
Each of the five audits, which took more than 2 years to complete, unearthed significant – and costly – billing mistakes, though the plans disputed them.
For example, auditors couldn’t confirm that one-third of the diseases the health plans had been paid to treat actually existed, mostly because patient records lacked “sufficient documentation of a diagnosis.”
Overall, Medicare paid the wrong amount for nearly two-thirds of patients whose records were examined; all five plans were far more likely to charge too much than too little. For one in five patients, the overcharges were $5,000 or more for the year, according to the audits. None of the plans would discuss the findings.
As preliminary results of the audits started to roll in, CMS officials outlined steps to recover more than $128 million from the five plans at a confidential agency briefing in August 2010, according to a policy memo prepared for the meeting. The records don’t indicate who attended.
That day, CMS set Humana’s payment error at $33.5 million, PacifiCare at $20.2 million, Aetna at $27.6 million, Independence Blue Cross at nearly $34 million and Lovelace at just under $13 million. Those estimates were based on extrapolation of a sample of cases examined at each plan.
CMS “has developed a process for moving forward with payment recovery,” according to a briefing paper from the 2010 meeting.
But that process fizzled after 2 years of haggling with the plans and insurance industry representatives, who argued the audits were flawed and the results unreliable. In August 2012, CMS gave in and notified the plans it would settle for a few cents on the dollar.
“Given this was a new process, the decision was made at the time to tie repayments to the actual claims reviewed as part of the 2007 pilot audit,” said CMS spokesman Aaron Albright. “For subsequent audits, we said we intended to determine repayments by extrapolating the error rate of the sample of claims reviewed to all claims under the contract.” Mr. Albright said more of the audits are underway. Allowing the insurers to dodge liability dealt a serious blow to the government’s efforts to crack down on billing abuses – a setback one taxpayer advocate called alarming.
“That’s a very bad way to operate the system.” said Patrick Burns, acting executive director and president of Taxpayers Against Fraud in Washington, on hearing of the outcome. “Nobody is held accountable.”
Indeed, CMS kept the settlement terms under wraps until 2015, after an inquiry by Sen. Grassley. The senator had requested details about Medicare Advantage fraud controls in response to articles published by the Center for Public Integrity.
In a July 31, 2015 letter to Sen. Grassley, CMS Acting Administrator Andy Slavitt attached a table that showed the five plans repaid just under $3.4 million. The letter didn’t mention the earlier estimate that the government was due $128 million. Sen. Grassley said it should not have taken the FOIA lawsuit to make that information available to the public.
“Perhaps adding insult to injury, these numbers might never have seen the light of day without a lengthy lawsuit,” Sen. Grassley said this week.
Paying based on risk scores
When Congress created the current Medicare Advantage program in 2003, it devised a new way to pay the health plans.
The method, phased in starting in 2004, seemed simple enough: Pay higher rates for sicker patients and less for people in good health using a formula called a risk score.
But CMS officials soon realized that risk scores rose much faster at some plans than others, a possible sign of upcoding, or other billing irregularities, records show. These overcharges topped $4 billion in 2005, one CMS study found.
The special audits, called Risk Adjustment Data Validation, or RADV, were designed to identify, and hold accountable, health plans that couldn’t justify their fees with supporting medical evidence.
Until these audits, CMS “pretty much went on the honor system with the plans,” an unnamed agency official wrote in an undated presentation.
In the five 2007 pilot audits, two sets of auditors inspected medical records for a random sample of 201 patients at each plan. If the medical chart didn’t properly document that a patient had the illnesses the plan had reported, Medicare wanted a refund. Auditors gave the plans the benefit of the doubt when auditors couldn’t agree, according to the CMS briefing paper.
Finally, CMS applied a standard technique used in fraud investigations in which the payment error rate is extrapolated across the entire health plan, which greatly multiplies the amount due. CMS said it was conservative in assessing the penalties and allowed the plans to appeal.
Appeals or no, the health plans recoiled at the prospect they could be on the hook for millions of dollars they hadn’t budgeted for and didn’t believe they owed. The actual 2007 overage for the 201 Humana patients, for example, was $477,235. Once extrapolated, it soared to $33.5 million.
Michael S. Adelberg, a former CMS official who is now an industry consultant in Washington, said that in retrospect the audit process was “probably rushed.”
Mr. Adelberg said the audits “raised strong industry concerns” on a variety of fronts, from whether CMS had the legal authority to conduct them to the soundness of their methods. CMS stands by its audit techniques and has defended RADV as the only way it can assure plans bill honestly.
Yet agency records released through the FOIA case suggest CMS lacked the will to press ahead with extrapolated audits for Medicare Advantage plans given the fierce industry backlash – even though they do so in overpayment cases targeting other types of medical providers.
One confidential CMS presentation dated March 30, 2011, notes that officials had received more than 500 comments expressing “significant resistance” to the RADV audits.
The presentation goes on to say the audit program’s success depended on its “ability to address the challenges raised.”
CMS didn’t overcome those challenges. Instead, it agreed to settle the five initial audits for $3.4 million, just what it found in the patient files it reviewed – without the extrapolations. And the center did the same for 32 additional 2007 audits, which officials had predicted would refund up to $800 million to the federal treasury. In the end, CMS wound up with $10.3 million from the 32 plans.
The RADV program’s shortcomings, though little known to the public, haven’t gone totally unnoticed. The program was the target of a sharply critical May 2016 report by the Government Accountability Office, which noted that Medicare Advantage plans have overbilled the government by billions of dollars, but rarely been forced to repay the money or face other consequences.
The GAO, the watchdog arm of Congress, called for “fundamental improvements.” The watchdogs also found that CMS has spent about $117 million on the audits, but recouped just under $14 million.
Government officials didn’t dispute that the RADV process had taken far too long and yielded way too little. But while CMS has resumed extrapolated audits, there’s little evidence it is speeding things up.
CMS expected to complete extrapolated audits for payments made in 2011 and finish the job in early 2014, agency records show.
But it has yet to do so. In late December, an agency spokesman said he had no new information about when the 2011 audits would be finished or how much the government would collect.
While the industry awaits the results, it has hardly warmed to the process.
America’s Health Insurance Plans, an industry trade group, argued in a June 2016 position paper that RADV was “not yet stable and reliable,” adding that the audits “could disrupt the care being provided by plans that are working hard to meet the needs of their enrollees.”
John Gorman, a former government health care official and current industry consultant, said he expects RADV to forge ahead under the incoming administration. But he predicted efforts to collect overpayments will “slow down” because the Trump team will prove to be “more sympathetic” to business interests than the Obama administration. The Trump transition office did not respond to a request for comment.
Mr. Gorman said that while career civil servants at CMS decide which plans get audited, how much to assess the health plans as a result rests with “political appointees” who are susceptible to industry lobbying, which he termed “the old Potomac two- step.”
But Sen. Grassley said he is determined to keep a close eye on the audit program. “I intend to press the incoming administration on holding CMS accountable for overpayments that harm taxpayers,” he said.
Taxpayer advocate Mr. Ellis said with so much public money at stake, the government needs to step up its game.
“You can presume that the more people get away with overpayments the more they are going to take,” he said. “As the program gets bigger the problem get bigger.”
This story is a collaboration between Kaiser Health News, a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation, and the Center for Public Integrity.
Historic HIV vaccine efficacy study underway in South Africa
.
According to an announcement by the National Institute of Allergy and Infectious Diseases, a cofunder of the trial and part of the U.S. National Institutes of Health, the study (called HVTN 702) involves a new version of the only HIV vaccine candidate ever shown to provide some protection against the virus. HVTN 702 intends to enroll 5,400 men and women, which would make it the largest and most advanced HIV vaccine clinical trial to take place in South Africa.
“If deployed alongside our current armory of proven HIV prevention tools, a safe and effective vaccine could be the final nail in the coffin for HIV,” Anthony S. Fauci, MD, director of NIAID, said in a statement. “Even a moderately effective vaccine would significantly decrease the burden of HIV disease over time in countries and populations with high rates of HIV infection, such as South Africa.”
The experimental vaccine regimen tested in the Thai trial was found to be 31.2 % effective at preventing HIV infection over the 3.5-year follow-up after vaccination. In the HVTN 702 study, the design, schedule, and components of the RV144 vaccine regimen have been modified in an attempt to increase the magnitude and duration of vaccine-elicited protective immune responses.
NIAID is responsible for all operational aspects of this phase IIb/III trial, which is enrolling HIV-uninfected, sexually active men and women aged 18-35 years. The NIAID-funded HIV Vaccine Trials Network is conducting the trial at 15 sites across South Africa, and expects results in late 2020. The study volunteers are being randomized to receive either the investigational vaccine regimen or a placebo. All study participants will receive a total of five injections over 1 year.
“If an HIV vaccine were found to work in South Africa, it could dramatically alter the course of the pandemic,” said HVTN 702 protocol chair Glenda Gray, MBBCH, president and CEO of the South African Medical Research Council and research professor of pediatrics at the University of the Witwatersrand, Johannesburg.
The HVTN 702 study begins just months after interim results were reported for HVTN 100, a predecessor clinical trial that found that the new vaccine regimen was safe for the 252 study participants and induced comparable immune responses to those reported in RV144, according to NIAID.
Both HVTN 100 and HVTN 702 are part of a larger HIV vaccine research endeavor led by the Pox-Protein Public-Private Partnership (P5), which includes NIAID, the Bill & Melinda Gates Foundation, the South African Medical Research Council, HVTN, Sanofi Pasteur, GSK, and the U.S. Military HIV Research Program.
NIAID said the HVTN 702 vaccine regimen consists of two experimental vaccines: a canarypox vector–based vaccine called ALVAC-HIV and a two-component gp120 protein subunit vaccine with an adjuvant to enhance the body’s immune response to the vaccine. Both ALVAC-HIV and the protein vaccine have been modified from the versions used in RV144 to be specific to HIV subtype C, the predominant HIV subtype in southern Africa.
In addition, NIAID said the protein subunit vaccine in HVTN 702 is combined with MF59, a different adjuvant than the one used in RV144, in the hope of generating a more robust immune response. The HVTN 702 vaccine regimen includes booster shots at the 1-year mark in an effort to prolong the early protective effect observed in RV144.
[email protected]
On Twitter @richpizzi
.
According to an announcement by the National Institute of Allergy and Infectious Diseases, a cofunder of the trial and part of the U.S. National Institutes of Health, the study (called HVTN 702) involves a new version of the only HIV vaccine candidate ever shown to provide some protection against the virus. HVTN 702 intends to enroll 5,400 men and women, which would make it the largest and most advanced HIV vaccine clinical trial to take place in South Africa.
“If deployed alongside our current armory of proven HIV prevention tools, a safe and effective vaccine could be the final nail in the coffin for HIV,” Anthony S. Fauci, MD, director of NIAID, said in a statement. “Even a moderately effective vaccine would significantly decrease the burden of HIV disease over time in countries and populations with high rates of HIV infection, such as South Africa.”
The experimental vaccine regimen tested in the Thai trial was found to be 31.2 % effective at preventing HIV infection over the 3.5-year follow-up after vaccination. In the HVTN 702 study, the design, schedule, and components of the RV144 vaccine regimen have been modified in an attempt to increase the magnitude and duration of vaccine-elicited protective immune responses.
NIAID is responsible for all operational aspects of this phase IIb/III trial, which is enrolling HIV-uninfected, sexually active men and women aged 18-35 years. The NIAID-funded HIV Vaccine Trials Network is conducting the trial at 15 sites across South Africa, and expects results in late 2020. The study volunteers are being randomized to receive either the investigational vaccine regimen or a placebo. All study participants will receive a total of five injections over 1 year.
“If an HIV vaccine were found to work in South Africa, it could dramatically alter the course of the pandemic,” said HVTN 702 protocol chair Glenda Gray, MBBCH, president and CEO of the South African Medical Research Council and research professor of pediatrics at the University of the Witwatersrand, Johannesburg.
The HVTN 702 study begins just months after interim results were reported for HVTN 100, a predecessor clinical trial that found that the new vaccine regimen was safe for the 252 study participants and induced comparable immune responses to those reported in RV144, according to NIAID.
Both HVTN 100 and HVTN 702 are part of a larger HIV vaccine research endeavor led by the Pox-Protein Public-Private Partnership (P5), which includes NIAID, the Bill & Melinda Gates Foundation, the South African Medical Research Council, HVTN, Sanofi Pasteur, GSK, and the U.S. Military HIV Research Program.
NIAID said the HVTN 702 vaccine regimen consists of two experimental vaccines: a canarypox vector–based vaccine called ALVAC-HIV and a two-component gp120 protein subunit vaccine with an adjuvant to enhance the body’s immune response to the vaccine. Both ALVAC-HIV and the protein vaccine have been modified from the versions used in RV144 to be specific to HIV subtype C, the predominant HIV subtype in southern Africa.
In addition, NIAID said the protein subunit vaccine in HVTN 702 is combined with MF59, a different adjuvant than the one used in RV144, in the hope of generating a more robust immune response. The HVTN 702 vaccine regimen includes booster shots at the 1-year mark in an effort to prolong the early protective effect observed in RV144.
[email protected]
On Twitter @richpizzi
.
According to an announcement by the National Institute of Allergy and Infectious Diseases, a cofunder of the trial and part of the U.S. National Institutes of Health, the study (called HVTN 702) involves a new version of the only HIV vaccine candidate ever shown to provide some protection against the virus. HVTN 702 intends to enroll 5,400 men and women, which would make it the largest and most advanced HIV vaccine clinical trial to take place in South Africa.
“If deployed alongside our current armory of proven HIV prevention tools, a safe and effective vaccine could be the final nail in the coffin for HIV,” Anthony S. Fauci, MD, director of NIAID, said in a statement. “Even a moderately effective vaccine would significantly decrease the burden of HIV disease over time in countries and populations with high rates of HIV infection, such as South Africa.”
The experimental vaccine regimen tested in the Thai trial was found to be 31.2 % effective at preventing HIV infection over the 3.5-year follow-up after vaccination. In the HVTN 702 study, the design, schedule, and components of the RV144 vaccine regimen have been modified in an attempt to increase the magnitude and duration of vaccine-elicited protective immune responses.
NIAID is responsible for all operational aspects of this phase IIb/III trial, which is enrolling HIV-uninfected, sexually active men and women aged 18-35 years. The NIAID-funded HIV Vaccine Trials Network is conducting the trial at 15 sites across South Africa, and expects results in late 2020. The study volunteers are being randomized to receive either the investigational vaccine regimen or a placebo. All study participants will receive a total of five injections over 1 year.
“If an HIV vaccine were found to work in South Africa, it could dramatically alter the course of the pandemic,” said HVTN 702 protocol chair Glenda Gray, MBBCH, president and CEO of the South African Medical Research Council and research professor of pediatrics at the University of the Witwatersrand, Johannesburg.
The HVTN 702 study begins just months after interim results were reported for HVTN 100, a predecessor clinical trial that found that the new vaccine regimen was safe for the 252 study participants and induced comparable immune responses to those reported in RV144, according to NIAID.
Both HVTN 100 and HVTN 702 are part of a larger HIV vaccine research endeavor led by the Pox-Protein Public-Private Partnership (P5), which includes NIAID, the Bill & Melinda Gates Foundation, the South African Medical Research Council, HVTN, Sanofi Pasteur, GSK, and the U.S. Military HIV Research Program.
NIAID said the HVTN 702 vaccine regimen consists of two experimental vaccines: a canarypox vector–based vaccine called ALVAC-HIV and a two-component gp120 protein subunit vaccine with an adjuvant to enhance the body’s immune response to the vaccine. Both ALVAC-HIV and the protein vaccine have been modified from the versions used in RV144 to be specific to HIV subtype C, the predominant HIV subtype in southern Africa.
In addition, NIAID said the protein subunit vaccine in HVTN 702 is combined with MF59, a different adjuvant than the one used in RV144, in the hope of generating a more robust immune response. The HVTN 702 vaccine regimen includes booster shots at the 1-year mark in an effort to prolong the early protective effect observed in RV144.
[email protected]
On Twitter @richpizzi
Racial Differences in Adherence to Prescribed Analgesia in Cancer Patients: An Integrated Review of Quantitative Research
From the University of Pennsylvania School of Nursing, Philadelphia, PA.
Abstract
- Background: Racial/ethnic disparities in analgesic treatment for pain have been widely documented in the United States. However, the connection between race/ethnicity and adherence to prescribed analgesics has not been described.
- Objectives: To review and synthesize quantitative research documenting racial/ethnic differences in adherence to prescribed analgesia in cancer patients.
- Methods: We performed a systematic search of quantitative, primary studies in Scopus, CINAHL, PubMed, Ovid, PsychInfo, and EMBASE. The title and abstract of each article was reviewed for relevance and whether inclusion criteria were met. Evidence was examined for relevant outcomes, data collection methods, variables studied in relation to adherence, and the magnitude of association between race/ethnicity and adherence.
- Results: Seven studies met inclusion criteria. Reported rates of adherence varied in studies among Hispanic/Latinos, African Americans, Asians, and whites based on variation in measurement tools, research questions, populations from which participants were recruited, and predictive variables analyzed. Most existing studies of analgesic adherence used self-report to measure adherence. Only 1 study used a validated, real-time electronic instrument to monitor prescribed opioid adherence and had a longitudinal study design.
- Conclusion: Limited research has examined relationships between adherence to prescribed analgesic regimens and racial disparities. Existing studies point to the clinical and socioeconomic factors that may interact with race/ethnicity in explaining analgesic and opioid adherence outcomes in cancer patients.
Key words: race, ethnicity, adherence, opiates, analgesics, pain management, cancer, pain treatment disparities.
The ongoing opioid epidemic and recent development of the Centers for Disease Control and Prevention (CDC) guidelines for chronic pain management have shaped a national conversation on opioid prescription and utilization [1]. The CDC delineates provider recommendations for opioid prescription. This focus on prescribed medication regimens is inadequate without an understanding of how patients take or adhere to prescribed medications. Cancer patients are a unique group. Moderate to severe pain in cancer patients is usually treated with opioids, and adherence to analgesia has been conceptualized a key mediator of cancer pain outcomes. For instance, a recent study found that patterns of analgesic adherence, specifically, inconsistent adherence to strong opioids (World Health Organization step 3), is one of the strongest predictors of health care utilization among outpatients with cancer pain [2]. Approximately 67% to 77% of cancer patients experience pain that requires management with analgesia [3], especially in the absence of access to nonpharmacologic pain treatments [2]. Thus, barriers in relation to adequate pain management can result in poor pain treatment outcomes and impaired quality of life for cancer patients.
Insufficient pain management has been found to have a negative impact on the quality of life and physical and mental functions of patients with cancer [4]. Patients who experience severe cancer pain are significantly more likely to experience multiple other symptoms such as depression, fatigue, and insomnia, resulting in diminished physical function [5], social role function [6], and greater out of pocket cost of managing pain and asso-ciated symptoms [7]. Minority populations, however, disproportionately carry the burden of undertreated pain [4,8–11,13–16]. Evidence suggests that blacks/African Americans are more likely to experience unrelieved cancer pain [4,8–11,13–16]. They are also less likely than their white counterparts to receive analgesic treatment for cancer pain [8–11,13,15,16]. Little is known, however, about racial disparities in relation to adherence to analgesia for cancer pain when providers prescribe analgesics.
The purpose of this paper is to review the published literature that has addressed the associations between disparities and adherence to analgesia among cancer patients. Evidence was examined for outcomes studied, data collection methods, variables studied in relation to adherence, and the magnitude of association based on race and adherence.
Methods
We performed a systematic search of studies published between 1990 and the present in Scopus, CINAHL, PubMed, Ovid, PsychInfo, and the EMBASE databases. The inclusion criteria consisted of published articles in the aforementioned databases that were (1) set in the United States, (2) primary studies, (3) employed quantitative design, (4) assessed adherence or compliance to analgesics or adequacy of pain management using the Pain Management Index (PMI), (5) sample was exclusively minority or may have had a comparative group. The title and abstract of each article in the the search results was reviewed for relevance to study aims and inclusion and exclusion criteria, and any duplicates were eliminated. A total of 6 studies were found using this method (Table 1), and an additional study was found in the reference list of 1 of these 6.
Results
The 7 included studies were observational in nature; 4 were cross-sectional [4,12,15,16], 2 were retrospective [3,14], and 1 was prospective and used objective measures of analgesic adherence [13] (Table 2).
Defining and Operationalizing Adherence
Meghani and Bruner [16] point out that analgesic adherence is a “heterogeneous construct that lends itself to varied results and interpretations depending on the measurements used or dimensions studied.” Adherence to analgesia was explicitly defined in all 7 studies (Table 3). One study reported an adherence rate that was the total dose over 24 hours divided by the dose prescribed then multiplied by 100 [4]. The total dose over 24 hours was used in another study but was converted to an equianalgesic calculation [12]. Another set of studies used a similar definition but specified percentages based on medication or type of prescription, such as an around-the-clock(ATC) regimen [13,15,16]. In 2 studies, adherence was measured based on chart review of yes/no questions posed about whether or not patients had taken medications as prescribed [3,15].
The measurements of adherence differed between studies. Four studies [4,12,14,16] used adherence as a primary outcome and the rest employed adherence as a facet of pain management [3,13,15]. The most frequent measure of adherence was self-report. The widely validated Morisky Medication Adherence Scale (MMAS) instrument was used in 3 of 7 studies [12,13,15]. Meghani and Bruner [15] utilized the modified MMAS plus a previously validated visual analog scale for doses of medication to assess adherence over week- and month-long intervals. One study used patient interviews to capture self-reporting of opioid prescription and opioid use. Additionally, the study used MMAS to further characterize the adherence measurements [12]. Using a more objective method, Meghani et al [13] employed a microprocessor in the medication cap to determine the percentage of the total number of prescribed doses that were actually taken [13]. The processor sensed when the bottle was open, which served as a proxy for taking medications at appropriate times.
Analgesic Adherence Rate
To report the analgesic adherence rates, 6 studies presented a percentage [3,4,12,13,15] and all but 1 highlighted the barriers associated with poor adherence [3,4,12,13,15,16].
The results of a pilot study exploring intentional and unintentional adherence revealed that 85.5% of patients took the prescribed medications in the previous week. Further analysis using visual analogue scale for dose adherence found that that 51% took up to 60% of the prescribed medications [15]. In an exclusively African-American sample, the adherence rate was reported as 46% [4]. Another study by Meghani et al compared adherence to prescribed ATC analgesics between African Americans and whites with cancer-related pain using an electronic monitoring system [13]. The overall adherence rate for African Americans was 53% and 74% for whites [13]. The authors concluded that there was a significant difference between the analgesic adherence rates between African Americans and whites in this study. On sub-analysis, analgesic adherence rates for African Americans were much lower for weak opioids (34%) and higher for long-acting opioids (63%).
In a study of individuals from an outpatient supportive care center with a majority white sample (74% Caucasian), overall 9.6% of patients deviated from the opioid regimen, while approximately 90% reported high adherence [12]. It is important to note that a convenience sample was used here. Of the total 19 patients that deviated from the regimen, 11 used less opioids than prescribed and 8 used higher doses. Upon analysis, the opioid deviation was more frequent in males and non-whites. However, statistical analyses of the magnitude of deviation from prescribed dose and non-white racial/ethnic background were not reported. Within the “non-whites” category, the race/ethnicity is defined as African American (16%, n = 32) and “other” (9%, n = 18). The authors contend that this strong adherence resulted from a strong understanding of the regimen as evidenced by a high agreement between the prescribed dose and the patient reported prescription [12]. Nguyen et al [12] argue that the literature shows that lower adherence rates for minority patients may be explained by the presence of comorbidities and lack of insurance.
Two other studies reported adherence rates for separate insurance cohorts [3,14]. The Medicaid cohort was younger and had a higher percentage of African-American individuals. However, in the self-pay/charity care group, the majority was Hispanic [3]. In the pilot study, the differences between the groups on adherence with prescribed medication regimens did not achieve statistical significance. The data were summarized to suggest that nonadherence was more likely in the self-pay/charity care group and more follow-up visits occurred after discharge [3]. During the larger retrospective study there was no difference in number of patients adhering to the regimen at each follow-up visit in each benefit group. The study concluded that the long-acting opiate adherence was influenced only by the benefits of use and that race/ethnicity was not a statistically significant predictor [14].
Factors Associated with Adherence
Multiple studies investigated factors underlying reported analgesic adherence rates for the ethnic and racial groups studied. Both clinical and sociodemographic variables were associated with analgesic adherence (Table 4). These included cancer type and disease stage [3,4,13,14], pain intensity [3,4,13–16], side effects [13,15], type of analgesic prescribed [3,4,13–16], income/socioeconomic status [3,13,14], behavioral history [3,12,13], gender [3,4,12–16], and perceived barriers [3,4,13,15,16].
Cancer Type and Stage
Most studies did not find significant associations between analgesic adherence rates and cancer type and stage [3,12,14]. However, 1 study that sought to identify unique factors underlying analgesic adherence for African Americans and whites found that whites reported higher analgesic adherence in relation to “time since cancer diagnosis,” possibly indicating disease severity and progression [13]. In another study that involved a majority of African-American patients, individuals with colon and rectal cancer had lower adherence rates [4]. In this study, patients with colon and rectal cancer had more analgesic prescriptions (2.5 +/– 2.3 analgesics) compared to patients with other cancer diagnoses. The authors concluded that an increased medication burden might have contributed to a decreased adherence rate. Overall, other cancer types did not correlate with adherence rates [4].
Pain Intensity
Six studies examined pain intensity and duration [3,4,13–16]. Three studies found a difference in reported pain intensity between racial/ethnic groups [3,13,16], 1 found no correlation between pain intensity and race/ethnicity [14], and 3 concluded that pain intensity was a significant predictor of adherence rates [3,13,15].
Meghani and Bruner’s pilot study explored possible correlates associated with intentional and unintentional nonadherence [15]. Overall, individuals were more likely to report forgetfulness (unintentional nonadherence) and to stop taking pain medicine when feeling “worse” (intentional nonadherence) if they believed that it was easier to deal with pain than with the side effects of analgesia [15]. Further, forgetfulness was negatively associated with the need for “stronger” pain medication. Concern about using too much pain medication was positively correlated with both forgetfulness and carelessness. The need for stronger pain medication was also correlated with significantly higher pain levels and lower pain relief [15].
In a comparative study of African Americans and whites, African Americans reported greater cancer pain and lower pain relief on the Brief Pain Inventory (BPI) and had a negative PMI. The PMI measure is a simple index linking the usual severity of cancer pain with the category of medication prescribed to treat it. PMI is calculated by subtracting patient’s pain levels (“pain worst” score from the BPI coded as mild, moderate, or severe) from the most potent analgesia prescribed. A negative PMI implies inadequate analgesic prescription relative to the reported pain level. Pain intensity was a significant factor related to increased adherence in whites but not African Americans. For African Americans, analgesic adherence was predicted by socioeconomic status, provider communication factors, and side effects. Similarly, in another study that compared African Americans and Hispanics, African Americans were more likely to have a negative PMI than Hispanics and were less likely to report that pain medication relieved pain [16]. In a pilot study that compared Medicaid recipients to self-pay/charity care patients, African-American participants had lower reported pain scores than Hispanics and Caucasians [3]. In the larger follow-up study, however, ethnicity did not prove a significant predictor for pain levels [14].
In a study with exclusively African-American patients, a significant correlation was found between pain intensity and adherence; specifically, as intensity increased, adherence increased [4]. Results for the entire African-American cohort indicated that 90% of patients had analgesic prescriptions for cancer-related pain, but 86% continued to report having moderate to severe worst pain [4]. A study that compared African Americans and whites showed that lower pain relief with analgesics was associated with lower adherence to analgesia for cancer pain among whites [13]. For every unit increase in “least pain” scores (indicating lower pain relief) on the BPI item, dose adherence decreased by 2.88%. Pain levels and relief did not explain adherence rates among African Americans. Whites were also more likely to make decisions on analgesic use based on the amount of relief anticipated from the use of analgesics [13] whereas African Americans were more likely to make analgesic use decisions based on analgesic side effects.
Side Effects
In a pilot study that explored the intricacies of adherence, some individuals felt it was easier to deal with pain than with the side effects of pain medications. These individuals were also more likely to report forgetfulness and to stop taking medications if feeling “worse” [15]. One study, which included African-American and white cohorts, found that an increase in the severity of side effects was associated with lower adherence to analgesia for African Americans but not whites. Furthermore, African Americans reported a greater number of analgesic side effects at baseline. African Americans were also more likely to make analgesic decisions based on side effects in comparison to whites participants, who made decisions based on expectation of pain relief [13]. In a study with exclusively African-American patients, patients with concerns about pain medication possibly causing confusion were more likely to have poor adherence [4].
Type of Analgesic Prescribed
In the analyses, 3 studies found a difference between analgesic prescriptions among ethnic groups [12,13,16], 3 found that there was a statistical significance between type of prescription and adherence [4,13,16], and 2 studies [3,14] found no statistical correlation between type of analgesic prescribed and adherence.
In a study of African Americans and Hispanics, both groups took analgesics on an “as-needed” basis despite the guidelines for cancer pain management [16]. However, African Americans reported taking analgesics less than twice daily. Overall, only a small percentage of patients took sustained-release analgesics that require fewer doses per day [16]. Similarly, in another study that compared adherence between African Americans and whites, the overall analgesic adherence rate was different on sub-analysis for specific analgesic prescriptions. The analgesic adherence rates for African Americans ranged from 34% for weak opioids to 63% for long-acting opioids. In comparison, the analgesic adherence rates for whites ranged from 55% for weak opioids to 78% for long-acting opioids [13]. In conclusion, patients on long-acting opioids were more likely to have higher adherence. Adherence rates for African Americans were found in another study. The adherence rate for adjuvant analgesics was highest at 65%, step 2 opioids at 44% and step 3 opioids at 43% [4].
In a study with exclusively African-American patients, poor adherence was significantly correlated with step 3 opioids [4]. Another study that explored the correlation between type of analgesic and adherence found that intentional nonadherence was less likely in individuals that were prescribed step 3 opioids [15]. Specifically, individuals with this behavior were also more likely to report lower pain levels and chose to stop the use of analgesics when feeling better [15].
Within a pilot study that compared benefit programs and payor groups, the differences in the prescription of long-acting opiates did not reach statistical significance [3]. However, in the larger, definitive study, the comparison revealed that patients in the self-pay/charity care group were less likely to receive a prescription for long-acting opiates. The data further revealed that Hispanic and Asian patients were prescribed long-acting opiates at a lower rate compared to the larger sample. Further, African Americans and Caucasians were prescribed long-acting opiates at a higher rate than the larger sample. In another analysis, with benefits and race/ethnicity, benefits were the only statistically significant predictor. While statistically controlling for race/ethnicity, Medicaid patients were 2.4 times more likely to receive a prescription for long-acting opioids than the self-pay/charity care patients [14].
Income/Socioeconomic Status
Three studies in this analysis [3,13,14] found that income and socioeconomic status were significant predictors of analgesic adherence for cancer pain. In a comparison between African Americans and whites, income was the strongest predictor of analgesic adherence for cancer pain in African Americans [13]; specifically, individuals with a household income of less than $10,000 a year had a 41.83% lower percentage of dose adherence. Among whites, income did not have a significant correlation with analgesic rates [13].
A pilot study and larger definitive study [3,14] were conducted to compare the effects of prescription benefits. The prescription benefits included were Medicaid and self-pay/charity care. Through comparison, none of the Medicaid patients reported financial barriers but the self-pay/charity care patients were more likely to report financial barriers to adherence [3]. In the larger study, the findings indicated that there was significant association of adherence by benefits and race/ethnicity. As mentioned above, benefits were a dominant predictor of long acting opiate use and further adherence [14].
Gender
Apart from ethnicity or race as a variable associated with adherence, association of analgesic adherence and gender were observed in 4 studies [3,13–15] and evaluated in 2 studies. One study [4] found that a patient’s gender and education level did not correlate with adherence rates. However, in another study [12] men were more likely to deviate from the prescribed dose. Overall, within the entire cohort [12] men and minority patients were most likely to deviate from the prescribed dosing regimen in comparison to all other patient demographic factors.
Attitudes and Barriers
Five of the 7 studies investigated perceived barriers to analgesic adherence [3,4,13,15,16]. Four used the Barriers Questionnaire II (BQ-II) [18] to further understand patients’ beliefs about cancer pain management [3,12,13,15]. Using this validated tool, 1 study found that non-white individuals had higher scores on the BQ-II than white patients [12]. Within the non-white group in the above study, the mean score on the BQ-II for African Americans was 1.76 (± 0.81) and the mean score for “other” was 2.16 (± 0.93) [12]. Further, low MMAS scores were significantly associated with higher BQ-II scores. Similarly, higher BQ-II scores correlated with opioid deviation toward higher than prescribed dose [12].
Another study with a primarily African-American cohort did not use the BQ-II but asked specific questions in regards to perceived barriers to analgesics. Within the cohort, 87% reported a fear of addiction to pain medicine. Further, 77% had a fear of injection, 75% were concerned about a tolerance for analgesics, and side effects were a major concern. Overall, nausea was the greatest reported concern followed by potential for confusion, which was negatively associated with taking analgesics. Distracting the doctor from curing their illness was a predictor of improved adherence; however, individuals were more likely to take Tylenol for pain relief. Similarly, no significant barrier items affected adherence to NSAIDs. In relation to step 2 opioids, patients who felt it was important to be strong by not talking about pain were more likely to have better adherence [4]. Similar results with African Americans were identified in another study [13]. In the comparison between African Americans and whites, African Americans had more subjective barriers compared to whites. Particularly for African Americans, each unit increase in concern about distracting the doctor from curing the disease, the percentage of dose adherence decreased by 7.44 [13].
In a study that compared payer groups, a questionnaire elicited reasons for nonadherence [3]. Similar reasons for nonadherence emerged including financial, fear of addiction or increased medication use, and running out medication.
Behavioral History
Only 1 study used CAGE (Cut down, Annoyed, Guilty, and Eye-opener), an alcohol-screening questionnaire, to determine a possible relationship with analgesic adherence. In this study, there were 19 cases of opioid deviation, 16% of which were CAGE positive and had severe deviation toward less than the prescribed doses [12]. In further analysis, no association was found between CAGE positively and opioid deviation to higher intake [12]. Two other studies gathered data on history of depression, substance use, and alcohol use but no significant correlation was found [3,13].
Discussion
Previous literature has reported overall analgesic adherence rates among oncology patients ranging from 62% to 72% [23]. Factors at the provider and system level have been considered in past research, but the patient perspective is poorly represented in the literature [13]. A majority of studies on analgesic adherence have been completed with cohorts made up predominantly of white individuals [13,23,24], while others focus on racially homogenous and/or ethnically different populations in other countries [21,25,26].
This review confirms that there is a paucity of well-designed studies that describe the associations between racial and ethnic disparities and adherence to opioids among patients with cancer pain. This is despite the fact that moderate to severe cancer pain in the U.S. is managed mainly with analgesics and specifically with opioids [19]. In addition, cancer patients with health insurance have both more pharmacy claims as well as more claims for higher doses of opioids [20] compared to noncancer patients. The lack of attention to analgesic and opioid adherence among cancer patients is surprising in the light of the recent high-profile initiatives to reduce opioid misuse [31].
Multiple studies highlighted the importance of pain management education and adequate pain assessment for effective analgesic use [4,16]. In the study in the palliative care setting, the authors concluded that patients who are educated, counseled, and monitored by a palliative or supportive care team have less episodes of opioid deviation and trends toward lower opioid use [12]. A systematic review and meta-analysis confirmed findings that educational interventions for patients improved knowledge about cancer pain management, however, most did not improve reported adherence to analgesics [27,28]. These findings emphasize the need for further research on interventions to improve racial/ethnic disparities in analgesic adherence for cancer pain.
Limitations
The findings of this review should be evaluated in the context of the following limitations. First, adherence to a prescribed regimen is a difficult outcome to measure and a majority of studies in this review used subjective measures to assess analgesic adherence for cancer pain. Of note, self-report was the primary measurement employed. Studies in non–cancer pain settings that have evaluated various methodological approaches to adherence measurement found that patients are likely to over-report adherence when using self-report or a diary format in comparison to an electronic monitoring system. Only 1 study in this review used an objective measure of adherence [13]. Some previous studies contend that self-report in comparison to other, objective measurements of medication adherence are accurate [23]. Further research is needed to determine the most accurate measurement of analgesic adherence in cancer patients.
Also, invariably the studies employed an English-speaking sample, which excludes an understanding of analgesic adherence for cancer pain in linguistically diverse Americans. In addition, most studies included patients who were either white Americans or African Americans and some studies lumped several racial ethnic minority subgroups as “nonwhites” or “other.”
A majority of studies were cross-sectional [4,12,15,16]. For instance, studies used a 24-hour time period to assess ATC medication as well as as-needed regimens, which may not capture the information needed to understand adherence to as-needed regimens [4]. With longitudinal studies, a greater understanding of adherence can be determined. However, there is potential bias with studies that track patients primarily at follow-up appointments. Individuals who are compliant with follow-up appointments may present with different analgesic adherence compared to those who do not attend follow-up appointments. This potential bias should be evaluated in longitudinal studies with various sensitivity analyses or using tools that identify healthy user bias.
Most studies recruited patients from outpatient oncology clinics, however, 1 study was conducted with a sample from an outpatient supportive care center managed by a palliative care team [12]. Due to the goals of palliative care, which include specialized treatment for individuals with serious illness and a focus on symptom management and relief, patients in this setting may have a different attitude toward using opioids.
Conclusion
Although data remain limited, our review suggests that while overuse of opioids has been a well-cited concern in patients with chronic non-cancer pain [21,33], cancer patients demonstrate considerable underuse and inconsistent use of prescribed analgesics. This is important as a recent study found that inconsistent adherence to prescribed around-the-clock analgesics, specifically the interaction of strong opioids and inconsistent adherence, is a strong risk factor for hospitalization among cancer outpatients who are prescribed analgesics for pain [1]. Of note, adherence to opioids in patients with cancer may be driven by a unique set of factors and these factors may differ for minorities and non-minority patients. For instance, studies in this review indicate that income is a strong predictor of analgesic adherence for African Americans but not for whites. This is because race and socioeconomic status frequently overlap in the United States [29]. In addition, like cancer pain, analgesic side effects may also be poorly managed among African Americans and other minorities. For example, in 1 study, Meghani et al used a trade-off analysis technique (conjoint analysis) to understand trade-offs African Americans and whites employ in using analgesics for cancer pain [30]. The authors found that African Americans were more likely to make analgesic adherence decisions based on side effects whereas whites were more likely to make adherence decisions based on pain relief [30]. In subsequent analysis, these authors showed that the race effect found in their previous studies was mediated by the type of analgesics prescribed to African Americans vs. whites [31]. African Americans with cancer pain were prescribed analgesics that had a worse side effect profile after statistically adjusting for insurance type and clinical risks such as renal insufficiency [31].
Together, the available evidence indicates that both patients’ socioeconomic status and clinician treatment bias contributes to racial and ethnic disparities in analgesic adherence for cancer pain and subsequent cancer pain outcomes. Thus, future research should investigate interventions for improving analgesic adherence among low-income minorities. Also, there is a need for clinician-level interventions focusing on cognitive bias modification related to cancer pain and side effects management, which appears to relate to analgesic nonadherence among racial/ethnic minorities. In addition, further research is needed to (1) rigorously describe analgesic and opioid adherence for cancer pain, (2) elucidate racial/ethnic and other socioeconomic and clinical disparities in analgesic and opioid adherence for cancer pain; (3) and clarify the role of analgesic and opioid adherence for cancer patients including outcomes for the patients and the health care system.
Corresponding author: Salimah H. Meghani, PhD, MBE, RN, University of Pennsylvania School of Nursing, Room 337, Fagin Hall, 418 Curie Blvd, Philadelphia, PA 19104, [email protected].
Financial disclosures: None.
1. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain - United States, 2016. JAMA 2016;315:1624–45.
2. Meghani SH, Knafl GJ. Patterns of analgesic adherence predict health care utilization among outpatients with cancer pain. Patient Prefer Adher 2016;10:81–98.
3. Bryan M, De La Rosa N, Hill AM, et al. Influence of prescription benefits on reported pain in cancer patients. Pain Med 2008;9:1148–57.
4. Rhee YO, Kim E, Kim B. Assessment of pain and analgesic use in African American cancer patients: Factors related to adherence to analgesics. J Immigr Minor Health 2012;14:1045–51.
5. Laird BJ, Scott AC, Colvin LA, et al. Pain, depression, and fatigue as a symptom cluster in advanced cancer. J Pain Symptom Manage 2011;42:1–11.
6. Ferreira KA, Kimura M, Teixeira MJ, et al. Impact of cancer-related symptom synergisms on health-related quality of life and performance status. J Pain Symptom Manage 2008;35:604–16.
7. Craig BM, Strassels SA. Out-of-pocket prices of opioid analgesics in the United States, 1999-2004. Pain Med 2010;11:240–47.
8. Institute of Medicine: Relieving pain in america: a blueprint for transforming prevention, care, education, and research. Washington, DC: National Academies Press; 2011.
9. Institutes of Medicine. Unequal treatment: confronting racial and ethnic disparities in health care. Washington, DC: National Academies Press; 2003.
10. Meghani SH, Byun E, Gallagher RM. Time to take stock: A meta-analysis and systematic review of analgesic treatment disparities for pain in the United States. Pain Med 2012;13:150–74.
11. Cleeland CS, Gonin R, Baez L, Loehrer P, Pandya KJ. Pain and treatment of pain in minority patients with cancer. The Eastern Cooperative Oncology Group Minority Outpatient Pain Study. Ann Intern Med 1997;127:813–6.
12. Nguyen LMT, Rhondali W, De la Cruz M, et al. Frequency and predictors of patient deviation from prescribed opioids and barriers to opioid pain management in patients with advanced cancer. J Pain Symptom Manage 2013;45:506–16.
13. Meghani SH, Thompson AML, Chittams J, et al. Adherence to analgesics for cancer pain: A comparative study of African Americans and whites using an electronic monitoring device. J Pain 2015;16:825–35.
14. Weider R, DeLaRosa N, Bryan M, et al. Prescription coverage in indigent patients affects the use of long acting opioids in management of cancer pain. Pain Med 2014;15:42–51.
15. Meghani SH, Brune DW. A pilot study to identify correlates of intentional versus unintentional nonadherence. Pain Manag Nurs 2013;14:e22-30.
16. Anderson KO, Mendoza TR, Valero V, et al. Minority cancer patients and their providers: pain management attitudes and practice. Cancer 2000;88:1929–38.
17. Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health 1998;52:377–84.
18. Ward SE, Goldberg N, Miller-McCauley V, et al. Patient-related barriers to management of cancer pain. Pain 1993;52:319–24.
19. Glare PA, Davies PS, Finlay E, et al. Pain in cancer survivors. J Clin Oncol 2014;32:1739–47.
20. van den Beuken-van Everdingen MH, de Rijke JM, Kessels AG, et al. Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol 2007;18:1437–49.
21. Jacobsen R, Samsanaviciene J, Liubarskiene Z, et al. Barriers to cancer pain management in Danish and Lithuanian patients treated in pain and palliative care units. Pain Manag Nurs 2014; 15:51–8.
22. National Institutes of Health. Pathways to prevention: the role of opioids in the treatment of chronic pain. September 29–30, 2014. Executive summary: final report. Accessed 10 Sep 2015 at https://prevention.nih.gov/docs/programs/p2p/ODPPainPanelStatementFinal_10-02-14.pdf.
23. Miaskowski C, Dodd MJ, West C, et al. Lack of adherence with the analgesic regimen: A significant barrier to effective cancer pain management. J Clin Oncol 2001;19:4275–79.
24. Yoong J, Traeger LN, Gallagher ER, et al. A pilot study to investigate adherence to long-acting opioids among patients with advanced lung cancer. J Palliat Med 2013;16:391–6.
25. Lai YH, Keefe FJ, Sun WZ, et al. Relationship between pain-specific beliefs and adherence to analgesic regimens in Taiwanese cancer patients: A preliminary study. J Pain Symptom Manage 2002;24:415–22.
26. Cohen MZ, Musgrave CF, McGuire DB, et al. The cancer pain experience of Israeli adults 65 years and older: the influence of pain interference, symptom severity, and knowledge and attitudes on pain and pain control. Support Care Cancer 2005;13:708–14.
27. Bennett MI, Bagnall AM, Jose Closs S. How effective are patient-based educational interventions in the management of cancer pain? Systematic review and meta analysis. Pain 2009;143:192–9.
28. Oldenmenger WH, Sillevis Smitt PA, van Dooren S, et al. A systematic review on barriers hindering adequate cancer pain management and interventions to reduce them: a critical appraisal. Eur J Cancer 2009;45:1370–80.
29. Meghani SH, Chittams J. Controlling for socioeconomic status in pain disparities research: all-else-equal analysis when “all else” is not equal. Pain Med 2015;16:2222–5.
30. Meghani SH, Chittams J, Hanlon A, Curry J. Measuring preferences for analgesic treatment for cancer pain: how do African Americans and whites perform on choice-based conjoint analysis experiments? BMC Med Inform Decis Mak 2013;13:118.
31. Meghani SH, Kang Y, Chittams J, et al. African Americans with cancer pain are more likely to receive an analgesic with toxic metabolite despite clinical risks: a mediation analysis study. J Clin Oncol 2014;32:2773–9.
32. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids forchronic pain - United States, 2016. MMWR Recomm Rep 2016 Mar 18;65:1–49.
33. Chapman CR, Lipschitz DL, Angst MS, et al. Opioid pharmacotherapy for chronic non-cancer pain in the United States: a research guideline for developing an evidence-base. J Pain 2010;11:807–29.
From the University of Pennsylvania School of Nursing, Philadelphia, PA.
Abstract
- Background: Racial/ethnic disparities in analgesic treatment for pain have been widely documented in the United States. However, the connection between race/ethnicity and adherence to prescribed analgesics has not been described.
- Objectives: To review and synthesize quantitative research documenting racial/ethnic differences in adherence to prescribed analgesia in cancer patients.
- Methods: We performed a systematic search of quantitative, primary studies in Scopus, CINAHL, PubMed, Ovid, PsychInfo, and EMBASE. The title and abstract of each article was reviewed for relevance and whether inclusion criteria were met. Evidence was examined for relevant outcomes, data collection methods, variables studied in relation to adherence, and the magnitude of association between race/ethnicity and adherence.
- Results: Seven studies met inclusion criteria. Reported rates of adherence varied in studies among Hispanic/Latinos, African Americans, Asians, and whites based on variation in measurement tools, research questions, populations from which participants were recruited, and predictive variables analyzed. Most existing studies of analgesic adherence used self-report to measure adherence. Only 1 study used a validated, real-time electronic instrument to monitor prescribed opioid adherence and had a longitudinal study design.
- Conclusion: Limited research has examined relationships between adherence to prescribed analgesic regimens and racial disparities. Existing studies point to the clinical and socioeconomic factors that may interact with race/ethnicity in explaining analgesic and opioid adherence outcomes in cancer patients.
Key words: race, ethnicity, adherence, opiates, analgesics, pain management, cancer, pain treatment disparities.
The ongoing opioid epidemic and recent development of the Centers for Disease Control and Prevention (CDC) guidelines for chronic pain management have shaped a national conversation on opioid prescription and utilization [1]. The CDC delineates provider recommendations for opioid prescription. This focus on prescribed medication regimens is inadequate without an understanding of how patients take or adhere to prescribed medications. Cancer patients are a unique group. Moderate to severe pain in cancer patients is usually treated with opioids, and adherence to analgesia has been conceptualized a key mediator of cancer pain outcomes. For instance, a recent study found that patterns of analgesic adherence, specifically, inconsistent adherence to strong opioids (World Health Organization step 3), is one of the strongest predictors of health care utilization among outpatients with cancer pain [2]. Approximately 67% to 77% of cancer patients experience pain that requires management with analgesia [3], especially in the absence of access to nonpharmacologic pain treatments [2]. Thus, barriers in relation to adequate pain management can result in poor pain treatment outcomes and impaired quality of life for cancer patients.
Insufficient pain management has been found to have a negative impact on the quality of life and physical and mental functions of patients with cancer [4]. Patients who experience severe cancer pain are significantly more likely to experience multiple other symptoms such as depression, fatigue, and insomnia, resulting in diminished physical function [5], social role function [6], and greater out of pocket cost of managing pain and asso-ciated symptoms [7]. Minority populations, however, disproportionately carry the burden of undertreated pain [4,8–11,13–16]. Evidence suggests that blacks/African Americans are more likely to experience unrelieved cancer pain [4,8–11,13–16]. They are also less likely than their white counterparts to receive analgesic treatment for cancer pain [8–11,13,15,16]. Little is known, however, about racial disparities in relation to adherence to analgesia for cancer pain when providers prescribe analgesics.
The purpose of this paper is to review the published literature that has addressed the associations between disparities and adherence to analgesia among cancer patients. Evidence was examined for outcomes studied, data collection methods, variables studied in relation to adherence, and the magnitude of association based on race and adherence.
Methods
We performed a systematic search of studies published between 1990 and the present in Scopus, CINAHL, PubMed, Ovid, PsychInfo, and the EMBASE databases. The inclusion criteria consisted of published articles in the aforementioned databases that were (1) set in the United States, (2) primary studies, (3) employed quantitative design, (4) assessed adherence or compliance to analgesics or adequacy of pain management using the Pain Management Index (PMI), (5) sample was exclusively minority or may have had a comparative group. The title and abstract of each article in the the search results was reviewed for relevance to study aims and inclusion and exclusion criteria, and any duplicates were eliminated. A total of 6 studies were found using this method (Table 1), and an additional study was found in the reference list of 1 of these 6.
Results
The 7 included studies were observational in nature; 4 were cross-sectional [4,12,15,16], 2 were retrospective [3,14], and 1 was prospective and used objective measures of analgesic adherence [13] (Table 2).
Defining and Operationalizing Adherence
Meghani and Bruner [16] point out that analgesic adherence is a “heterogeneous construct that lends itself to varied results and interpretations depending on the measurements used or dimensions studied.” Adherence to analgesia was explicitly defined in all 7 studies (Table 3). One study reported an adherence rate that was the total dose over 24 hours divided by the dose prescribed then multiplied by 100 [4]. The total dose over 24 hours was used in another study but was converted to an equianalgesic calculation [12]. Another set of studies used a similar definition but specified percentages based on medication or type of prescription, such as an around-the-clock(ATC) regimen [13,15,16]. In 2 studies, adherence was measured based on chart review of yes/no questions posed about whether or not patients had taken medications as prescribed [3,15].
The measurements of adherence differed between studies. Four studies [4,12,14,16] used adherence as a primary outcome and the rest employed adherence as a facet of pain management [3,13,15]. The most frequent measure of adherence was self-report. The widely validated Morisky Medication Adherence Scale (MMAS) instrument was used in 3 of 7 studies [12,13,15]. Meghani and Bruner [15] utilized the modified MMAS plus a previously validated visual analog scale for doses of medication to assess adherence over week- and month-long intervals. One study used patient interviews to capture self-reporting of opioid prescription and opioid use. Additionally, the study used MMAS to further characterize the adherence measurements [12]. Using a more objective method, Meghani et al [13] employed a microprocessor in the medication cap to determine the percentage of the total number of prescribed doses that were actually taken [13]. The processor sensed when the bottle was open, which served as a proxy for taking medications at appropriate times.
Analgesic Adherence Rate
To report the analgesic adherence rates, 6 studies presented a percentage [3,4,12,13,15] and all but 1 highlighted the barriers associated with poor adherence [3,4,12,13,15,16].
The results of a pilot study exploring intentional and unintentional adherence revealed that 85.5% of patients took the prescribed medications in the previous week. Further analysis using visual analogue scale for dose adherence found that that 51% took up to 60% of the prescribed medications [15]. In an exclusively African-American sample, the adherence rate was reported as 46% [4]. Another study by Meghani et al compared adherence to prescribed ATC analgesics between African Americans and whites with cancer-related pain using an electronic monitoring system [13]. The overall adherence rate for African Americans was 53% and 74% for whites [13]. The authors concluded that there was a significant difference between the analgesic adherence rates between African Americans and whites in this study. On sub-analysis, analgesic adherence rates for African Americans were much lower for weak opioids (34%) and higher for long-acting opioids (63%).
In a study of individuals from an outpatient supportive care center with a majority white sample (74% Caucasian), overall 9.6% of patients deviated from the opioid regimen, while approximately 90% reported high adherence [12]. It is important to note that a convenience sample was used here. Of the total 19 patients that deviated from the regimen, 11 used less opioids than prescribed and 8 used higher doses. Upon analysis, the opioid deviation was more frequent in males and non-whites. However, statistical analyses of the magnitude of deviation from prescribed dose and non-white racial/ethnic background were not reported. Within the “non-whites” category, the race/ethnicity is defined as African American (16%, n = 32) and “other” (9%, n = 18). The authors contend that this strong adherence resulted from a strong understanding of the regimen as evidenced by a high agreement between the prescribed dose and the patient reported prescription [12]. Nguyen et al [12] argue that the literature shows that lower adherence rates for minority patients may be explained by the presence of comorbidities and lack of insurance.
Two other studies reported adherence rates for separate insurance cohorts [3,14]. The Medicaid cohort was younger and had a higher percentage of African-American individuals. However, in the self-pay/charity care group, the majority was Hispanic [3]. In the pilot study, the differences between the groups on adherence with prescribed medication regimens did not achieve statistical significance. The data were summarized to suggest that nonadherence was more likely in the self-pay/charity care group and more follow-up visits occurred after discharge [3]. During the larger retrospective study there was no difference in number of patients adhering to the regimen at each follow-up visit in each benefit group. The study concluded that the long-acting opiate adherence was influenced only by the benefits of use and that race/ethnicity was not a statistically significant predictor [14].
Factors Associated with Adherence
Multiple studies investigated factors underlying reported analgesic adherence rates for the ethnic and racial groups studied. Both clinical and sociodemographic variables were associated with analgesic adherence (Table 4). These included cancer type and disease stage [3,4,13,14], pain intensity [3,4,13–16], side effects [13,15], type of analgesic prescribed [3,4,13–16], income/socioeconomic status [3,13,14], behavioral history [3,12,13], gender [3,4,12–16], and perceived barriers [3,4,13,15,16].
Cancer Type and Stage
Most studies did not find significant associations between analgesic adherence rates and cancer type and stage [3,12,14]. However, 1 study that sought to identify unique factors underlying analgesic adherence for African Americans and whites found that whites reported higher analgesic adherence in relation to “time since cancer diagnosis,” possibly indicating disease severity and progression [13]. In another study that involved a majority of African-American patients, individuals with colon and rectal cancer had lower adherence rates [4]. In this study, patients with colon and rectal cancer had more analgesic prescriptions (2.5 +/– 2.3 analgesics) compared to patients with other cancer diagnoses. The authors concluded that an increased medication burden might have contributed to a decreased adherence rate. Overall, other cancer types did not correlate with adherence rates [4].
Pain Intensity
Six studies examined pain intensity and duration [3,4,13–16]. Three studies found a difference in reported pain intensity between racial/ethnic groups [3,13,16], 1 found no correlation between pain intensity and race/ethnicity [14], and 3 concluded that pain intensity was a significant predictor of adherence rates [3,13,15].
Meghani and Bruner’s pilot study explored possible correlates associated with intentional and unintentional nonadherence [15]. Overall, individuals were more likely to report forgetfulness (unintentional nonadherence) and to stop taking pain medicine when feeling “worse” (intentional nonadherence) if they believed that it was easier to deal with pain than with the side effects of analgesia [15]. Further, forgetfulness was negatively associated with the need for “stronger” pain medication. Concern about using too much pain medication was positively correlated with both forgetfulness and carelessness. The need for stronger pain medication was also correlated with significantly higher pain levels and lower pain relief [15].
In a comparative study of African Americans and whites, African Americans reported greater cancer pain and lower pain relief on the Brief Pain Inventory (BPI) and had a negative PMI. The PMI measure is a simple index linking the usual severity of cancer pain with the category of medication prescribed to treat it. PMI is calculated by subtracting patient’s pain levels (“pain worst” score from the BPI coded as mild, moderate, or severe) from the most potent analgesia prescribed. A negative PMI implies inadequate analgesic prescription relative to the reported pain level. Pain intensity was a significant factor related to increased adherence in whites but not African Americans. For African Americans, analgesic adherence was predicted by socioeconomic status, provider communication factors, and side effects. Similarly, in another study that compared African Americans and Hispanics, African Americans were more likely to have a negative PMI than Hispanics and were less likely to report that pain medication relieved pain [16]. In a pilot study that compared Medicaid recipients to self-pay/charity care patients, African-American participants had lower reported pain scores than Hispanics and Caucasians [3]. In the larger follow-up study, however, ethnicity did not prove a significant predictor for pain levels [14].
In a study with exclusively African-American patients, a significant correlation was found between pain intensity and adherence; specifically, as intensity increased, adherence increased [4]. Results for the entire African-American cohort indicated that 90% of patients had analgesic prescriptions for cancer-related pain, but 86% continued to report having moderate to severe worst pain [4]. A study that compared African Americans and whites showed that lower pain relief with analgesics was associated with lower adherence to analgesia for cancer pain among whites [13]. For every unit increase in “least pain” scores (indicating lower pain relief) on the BPI item, dose adherence decreased by 2.88%. Pain levels and relief did not explain adherence rates among African Americans. Whites were also more likely to make decisions on analgesic use based on the amount of relief anticipated from the use of analgesics [13] whereas African Americans were more likely to make analgesic use decisions based on analgesic side effects.
Side Effects
In a pilot study that explored the intricacies of adherence, some individuals felt it was easier to deal with pain than with the side effects of pain medications. These individuals were also more likely to report forgetfulness and to stop taking medications if feeling “worse” [15]. One study, which included African-American and white cohorts, found that an increase in the severity of side effects was associated with lower adherence to analgesia for African Americans but not whites. Furthermore, African Americans reported a greater number of analgesic side effects at baseline. African Americans were also more likely to make analgesic decisions based on side effects in comparison to whites participants, who made decisions based on expectation of pain relief [13]. In a study with exclusively African-American patients, patients with concerns about pain medication possibly causing confusion were more likely to have poor adherence [4].
Type of Analgesic Prescribed
In the analyses, 3 studies found a difference between analgesic prescriptions among ethnic groups [12,13,16], 3 found that there was a statistical significance between type of prescription and adherence [4,13,16], and 2 studies [3,14] found no statistical correlation between type of analgesic prescribed and adherence.
In a study of African Americans and Hispanics, both groups took analgesics on an “as-needed” basis despite the guidelines for cancer pain management [16]. However, African Americans reported taking analgesics less than twice daily. Overall, only a small percentage of patients took sustained-release analgesics that require fewer doses per day [16]. Similarly, in another study that compared adherence between African Americans and whites, the overall analgesic adherence rate was different on sub-analysis for specific analgesic prescriptions. The analgesic adherence rates for African Americans ranged from 34% for weak opioids to 63% for long-acting opioids. In comparison, the analgesic adherence rates for whites ranged from 55% for weak opioids to 78% for long-acting opioids [13]. In conclusion, patients on long-acting opioids were more likely to have higher adherence. Adherence rates for African Americans were found in another study. The adherence rate for adjuvant analgesics was highest at 65%, step 2 opioids at 44% and step 3 opioids at 43% [4].
In a study with exclusively African-American patients, poor adherence was significantly correlated with step 3 opioids [4]. Another study that explored the correlation between type of analgesic and adherence found that intentional nonadherence was less likely in individuals that were prescribed step 3 opioids [15]. Specifically, individuals with this behavior were also more likely to report lower pain levels and chose to stop the use of analgesics when feeling better [15].
Within a pilot study that compared benefit programs and payor groups, the differences in the prescription of long-acting opiates did not reach statistical significance [3]. However, in the larger, definitive study, the comparison revealed that patients in the self-pay/charity care group were less likely to receive a prescription for long-acting opiates. The data further revealed that Hispanic and Asian patients were prescribed long-acting opiates at a lower rate compared to the larger sample. Further, African Americans and Caucasians were prescribed long-acting opiates at a higher rate than the larger sample. In another analysis, with benefits and race/ethnicity, benefits were the only statistically significant predictor. While statistically controlling for race/ethnicity, Medicaid patients were 2.4 times more likely to receive a prescription for long-acting opioids than the self-pay/charity care patients [14].
Income/Socioeconomic Status
Three studies in this analysis [3,13,14] found that income and socioeconomic status were significant predictors of analgesic adherence for cancer pain. In a comparison between African Americans and whites, income was the strongest predictor of analgesic adherence for cancer pain in African Americans [13]; specifically, individuals with a household income of less than $10,000 a year had a 41.83% lower percentage of dose adherence. Among whites, income did not have a significant correlation with analgesic rates [13].
A pilot study and larger definitive study [3,14] were conducted to compare the effects of prescription benefits. The prescription benefits included were Medicaid and self-pay/charity care. Through comparison, none of the Medicaid patients reported financial barriers but the self-pay/charity care patients were more likely to report financial barriers to adherence [3]. In the larger study, the findings indicated that there was significant association of adherence by benefits and race/ethnicity. As mentioned above, benefits were a dominant predictor of long acting opiate use and further adherence [14].
Gender
Apart from ethnicity or race as a variable associated with adherence, association of analgesic adherence and gender were observed in 4 studies [3,13–15] and evaluated in 2 studies. One study [4] found that a patient’s gender and education level did not correlate with adherence rates. However, in another study [12] men were more likely to deviate from the prescribed dose. Overall, within the entire cohort [12] men and minority patients were most likely to deviate from the prescribed dosing regimen in comparison to all other patient demographic factors.
Attitudes and Barriers
Five of the 7 studies investigated perceived barriers to analgesic adherence [3,4,13,15,16]. Four used the Barriers Questionnaire II (BQ-II) [18] to further understand patients’ beliefs about cancer pain management [3,12,13,15]. Using this validated tool, 1 study found that non-white individuals had higher scores on the BQ-II than white patients [12]. Within the non-white group in the above study, the mean score on the BQ-II for African Americans was 1.76 (± 0.81) and the mean score for “other” was 2.16 (± 0.93) [12]. Further, low MMAS scores were significantly associated with higher BQ-II scores. Similarly, higher BQ-II scores correlated with opioid deviation toward higher than prescribed dose [12].
Another study with a primarily African-American cohort did not use the BQ-II but asked specific questions in regards to perceived barriers to analgesics. Within the cohort, 87% reported a fear of addiction to pain medicine. Further, 77% had a fear of injection, 75% were concerned about a tolerance for analgesics, and side effects were a major concern. Overall, nausea was the greatest reported concern followed by potential for confusion, which was negatively associated with taking analgesics. Distracting the doctor from curing their illness was a predictor of improved adherence; however, individuals were more likely to take Tylenol for pain relief. Similarly, no significant barrier items affected adherence to NSAIDs. In relation to step 2 opioids, patients who felt it was important to be strong by not talking about pain were more likely to have better adherence [4]. Similar results with African Americans were identified in another study [13]. In the comparison between African Americans and whites, African Americans had more subjective barriers compared to whites. Particularly for African Americans, each unit increase in concern about distracting the doctor from curing the disease, the percentage of dose adherence decreased by 7.44 [13].
In a study that compared payer groups, a questionnaire elicited reasons for nonadherence [3]. Similar reasons for nonadherence emerged including financial, fear of addiction or increased medication use, and running out medication.
Behavioral History
Only 1 study used CAGE (Cut down, Annoyed, Guilty, and Eye-opener), an alcohol-screening questionnaire, to determine a possible relationship with analgesic adherence. In this study, there were 19 cases of opioid deviation, 16% of which were CAGE positive and had severe deviation toward less than the prescribed doses [12]. In further analysis, no association was found between CAGE positively and opioid deviation to higher intake [12]. Two other studies gathered data on history of depression, substance use, and alcohol use but no significant correlation was found [3,13].
Discussion
Previous literature has reported overall analgesic adherence rates among oncology patients ranging from 62% to 72% [23]. Factors at the provider and system level have been considered in past research, but the patient perspective is poorly represented in the literature [13]. A majority of studies on analgesic adherence have been completed with cohorts made up predominantly of white individuals [13,23,24], while others focus on racially homogenous and/or ethnically different populations in other countries [21,25,26].
This review confirms that there is a paucity of well-designed studies that describe the associations between racial and ethnic disparities and adherence to opioids among patients with cancer pain. This is despite the fact that moderate to severe cancer pain in the U.S. is managed mainly with analgesics and specifically with opioids [19]. In addition, cancer patients with health insurance have both more pharmacy claims as well as more claims for higher doses of opioids [20] compared to noncancer patients. The lack of attention to analgesic and opioid adherence among cancer patients is surprising in the light of the recent high-profile initiatives to reduce opioid misuse [31].
Multiple studies highlighted the importance of pain management education and adequate pain assessment for effective analgesic use [4,16]. In the study in the palliative care setting, the authors concluded that patients who are educated, counseled, and monitored by a palliative or supportive care team have less episodes of opioid deviation and trends toward lower opioid use [12]. A systematic review and meta-analysis confirmed findings that educational interventions for patients improved knowledge about cancer pain management, however, most did not improve reported adherence to analgesics [27,28]. These findings emphasize the need for further research on interventions to improve racial/ethnic disparities in analgesic adherence for cancer pain.
Limitations
The findings of this review should be evaluated in the context of the following limitations. First, adherence to a prescribed regimen is a difficult outcome to measure and a majority of studies in this review used subjective measures to assess analgesic adherence for cancer pain. Of note, self-report was the primary measurement employed. Studies in non–cancer pain settings that have evaluated various methodological approaches to adherence measurement found that patients are likely to over-report adherence when using self-report or a diary format in comparison to an electronic monitoring system. Only 1 study in this review used an objective measure of adherence [13]. Some previous studies contend that self-report in comparison to other, objective measurements of medication adherence are accurate [23]. Further research is needed to determine the most accurate measurement of analgesic adherence in cancer patients.
Also, invariably the studies employed an English-speaking sample, which excludes an understanding of analgesic adherence for cancer pain in linguistically diverse Americans. In addition, most studies included patients who were either white Americans or African Americans and some studies lumped several racial ethnic minority subgroups as “nonwhites” or “other.”
A majority of studies were cross-sectional [4,12,15,16]. For instance, studies used a 24-hour time period to assess ATC medication as well as as-needed regimens, which may not capture the information needed to understand adherence to as-needed regimens [4]. With longitudinal studies, a greater understanding of adherence can be determined. However, there is potential bias with studies that track patients primarily at follow-up appointments. Individuals who are compliant with follow-up appointments may present with different analgesic adherence compared to those who do not attend follow-up appointments. This potential bias should be evaluated in longitudinal studies with various sensitivity analyses or using tools that identify healthy user bias.
Most studies recruited patients from outpatient oncology clinics, however, 1 study was conducted with a sample from an outpatient supportive care center managed by a palliative care team [12]. Due to the goals of palliative care, which include specialized treatment for individuals with serious illness and a focus on symptom management and relief, patients in this setting may have a different attitude toward using opioids.
Conclusion
Although data remain limited, our review suggests that while overuse of opioids has been a well-cited concern in patients with chronic non-cancer pain [21,33], cancer patients demonstrate considerable underuse and inconsistent use of prescribed analgesics. This is important as a recent study found that inconsistent adherence to prescribed around-the-clock analgesics, specifically the interaction of strong opioids and inconsistent adherence, is a strong risk factor for hospitalization among cancer outpatients who are prescribed analgesics for pain [1]. Of note, adherence to opioids in patients with cancer may be driven by a unique set of factors and these factors may differ for minorities and non-minority patients. For instance, studies in this review indicate that income is a strong predictor of analgesic adherence for African Americans but not for whites. This is because race and socioeconomic status frequently overlap in the United States [29]. In addition, like cancer pain, analgesic side effects may also be poorly managed among African Americans and other minorities. For example, in 1 study, Meghani et al used a trade-off analysis technique (conjoint analysis) to understand trade-offs African Americans and whites employ in using analgesics for cancer pain [30]. The authors found that African Americans were more likely to make analgesic adherence decisions based on side effects whereas whites were more likely to make adherence decisions based on pain relief [30]. In subsequent analysis, these authors showed that the race effect found in their previous studies was mediated by the type of analgesics prescribed to African Americans vs. whites [31]. African Americans with cancer pain were prescribed analgesics that had a worse side effect profile after statistically adjusting for insurance type and clinical risks such as renal insufficiency [31].
Together, the available evidence indicates that both patients’ socioeconomic status and clinician treatment bias contributes to racial and ethnic disparities in analgesic adherence for cancer pain and subsequent cancer pain outcomes. Thus, future research should investigate interventions for improving analgesic adherence among low-income minorities. Also, there is a need for clinician-level interventions focusing on cognitive bias modification related to cancer pain and side effects management, which appears to relate to analgesic nonadherence among racial/ethnic minorities. In addition, further research is needed to (1) rigorously describe analgesic and opioid adherence for cancer pain, (2) elucidate racial/ethnic and other socioeconomic and clinical disparities in analgesic and opioid adherence for cancer pain; (3) and clarify the role of analgesic and opioid adherence for cancer patients including outcomes for the patients and the health care system.
Corresponding author: Salimah H. Meghani, PhD, MBE, RN, University of Pennsylvania School of Nursing, Room 337, Fagin Hall, 418 Curie Blvd, Philadelphia, PA 19104, [email protected].
Financial disclosures: None.
From the University of Pennsylvania School of Nursing, Philadelphia, PA.
Abstract
- Background: Racial/ethnic disparities in analgesic treatment for pain have been widely documented in the United States. However, the connection between race/ethnicity and adherence to prescribed analgesics has not been described.
- Objectives: To review and synthesize quantitative research documenting racial/ethnic differences in adherence to prescribed analgesia in cancer patients.
- Methods: We performed a systematic search of quantitative, primary studies in Scopus, CINAHL, PubMed, Ovid, PsychInfo, and EMBASE. The title and abstract of each article was reviewed for relevance and whether inclusion criteria were met. Evidence was examined for relevant outcomes, data collection methods, variables studied in relation to adherence, and the magnitude of association between race/ethnicity and adherence.
- Results: Seven studies met inclusion criteria. Reported rates of adherence varied in studies among Hispanic/Latinos, African Americans, Asians, and whites based on variation in measurement tools, research questions, populations from which participants were recruited, and predictive variables analyzed. Most existing studies of analgesic adherence used self-report to measure adherence. Only 1 study used a validated, real-time electronic instrument to monitor prescribed opioid adherence and had a longitudinal study design.
- Conclusion: Limited research has examined relationships between adherence to prescribed analgesic regimens and racial disparities. Existing studies point to the clinical and socioeconomic factors that may interact with race/ethnicity in explaining analgesic and opioid adherence outcomes in cancer patients.
Key words: race, ethnicity, adherence, opiates, analgesics, pain management, cancer, pain treatment disparities.
The ongoing opioid epidemic and recent development of the Centers for Disease Control and Prevention (CDC) guidelines for chronic pain management have shaped a national conversation on opioid prescription and utilization [1]. The CDC delineates provider recommendations for opioid prescription. This focus on prescribed medication regimens is inadequate without an understanding of how patients take or adhere to prescribed medications. Cancer patients are a unique group. Moderate to severe pain in cancer patients is usually treated with opioids, and adherence to analgesia has been conceptualized a key mediator of cancer pain outcomes. For instance, a recent study found that patterns of analgesic adherence, specifically, inconsistent adherence to strong opioids (World Health Organization step 3), is one of the strongest predictors of health care utilization among outpatients with cancer pain [2]. Approximately 67% to 77% of cancer patients experience pain that requires management with analgesia [3], especially in the absence of access to nonpharmacologic pain treatments [2]. Thus, barriers in relation to adequate pain management can result in poor pain treatment outcomes and impaired quality of life for cancer patients.
Insufficient pain management has been found to have a negative impact on the quality of life and physical and mental functions of patients with cancer [4]. Patients who experience severe cancer pain are significantly more likely to experience multiple other symptoms such as depression, fatigue, and insomnia, resulting in diminished physical function [5], social role function [6], and greater out of pocket cost of managing pain and asso-ciated symptoms [7]. Minority populations, however, disproportionately carry the burden of undertreated pain [4,8–11,13–16]. Evidence suggests that blacks/African Americans are more likely to experience unrelieved cancer pain [4,8–11,13–16]. They are also less likely than their white counterparts to receive analgesic treatment for cancer pain [8–11,13,15,16]. Little is known, however, about racial disparities in relation to adherence to analgesia for cancer pain when providers prescribe analgesics.
The purpose of this paper is to review the published literature that has addressed the associations between disparities and adherence to analgesia among cancer patients. Evidence was examined for outcomes studied, data collection methods, variables studied in relation to adherence, and the magnitude of association based on race and adherence.
Methods
We performed a systematic search of studies published between 1990 and the present in Scopus, CINAHL, PubMed, Ovid, PsychInfo, and the EMBASE databases. The inclusion criteria consisted of published articles in the aforementioned databases that were (1) set in the United States, (2) primary studies, (3) employed quantitative design, (4) assessed adherence or compliance to analgesics or adequacy of pain management using the Pain Management Index (PMI), (5) sample was exclusively minority or may have had a comparative group. The title and abstract of each article in the the search results was reviewed for relevance to study aims and inclusion and exclusion criteria, and any duplicates were eliminated. A total of 6 studies were found using this method (Table 1), and an additional study was found in the reference list of 1 of these 6.
Results
The 7 included studies were observational in nature; 4 were cross-sectional [4,12,15,16], 2 were retrospective [3,14], and 1 was prospective and used objective measures of analgesic adherence [13] (Table 2).
Defining and Operationalizing Adherence
Meghani and Bruner [16] point out that analgesic adherence is a “heterogeneous construct that lends itself to varied results and interpretations depending on the measurements used or dimensions studied.” Adherence to analgesia was explicitly defined in all 7 studies (Table 3). One study reported an adherence rate that was the total dose over 24 hours divided by the dose prescribed then multiplied by 100 [4]. The total dose over 24 hours was used in another study but was converted to an equianalgesic calculation [12]. Another set of studies used a similar definition but specified percentages based on medication or type of prescription, such as an around-the-clock(ATC) regimen [13,15,16]. In 2 studies, adherence was measured based on chart review of yes/no questions posed about whether or not patients had taken medications as prescribed [3,15].
The measurements of adherence differed between studies. Four studies [4,12,14,16] used adherence as a primary outcome and the rest employed adherence as a facet of pain management [3,13,15]. The most frequent measure of adherence was self-report. The widely validated Morisky Medication Adherence Scale (MMAS) instrument was used in 3 of 7 studies [12,13,15]. Meghani and Bruner [15] utilized the modified MMAS plus a previously validated visual analog scale for doses of medication to assess adherence over week- and month-long intervals. One study used patient interviews to capture self-reporting of opioid prescription and opioid use. Additionally, the study used MMAS to further characterize the adherence measurements [12]. Using a more objective method, Meghani et al [13] employed a microprocessor in the medication cap to determine the percentage of the total number of prescribed doses that were actually taken [13]. The processor sensed when the bottle was open, which served as a proxy for taking medications at appropriate times.
Analgesic Adherence Rate
To report the analgesic adherence rates, 6 studies presented a percentage [3,4,12,13,15] and all but 1 highlighted the barriers associated with poor adherence [3,4,12,13,15,16].
The results of a pilot study exploring intentional and unintentional adherence revealed that 85.5% of patients took the prescribed medications in the previous week. Further analysis using visual analogue scale for dose adherence found that that 51% took up to 60% of the prescribed medications [15]. In an exclusively African-American sample, the adherence rate was reported as 46% [4]. Another study by Meghani et al compared adherence to prescribed ATC analgesics between African Americans and whites with cancer-related pain using an electronic monitoring system [13]. The overall adherence rate for African Americans was 53% and 74% for whites [13]. The authors concluded that there was a significant difference between the analgesic adherence rates between African Americans and whites in this study. On sub-analysis, analgesic adherence rates for African Americans were much lower for weak opioids (34%) and higher for long-acting opioids (63%).
In a study of individuals from an outpatient supportive care center with a majority white sample (74% Caucasian), overall 9.6% of patients deviated from the opioid regimen, while approximately 90% reported high adherence [12]. It is important to note that a convenience sample was used here. Of the total 19 patients that deviated from the regimen, 11 used less opioids than prescribed and 8 used higher doses. Upon analysis, the opioid deviation was more frequent in males and non-whites. However, statistical analyses of the magnitude of deviation from prescribed dose and non-white racial/ethnic background were not reported. Within the “non-whites” category, the race/ethnicity is defined as African American (16%, n = 32) and “other” (9%, n = 18). The authors contend that this strong adherence resulted from a strong understanding of the regimen as evidenced by a high agreement between the prescribed dose and the patient reported prescription [12]. Nguyen et al [12] argue that the literature shows that lower adherence rates for minority patients may be explained by the presence of comorbidities and lack of insurance.
Two other studies reported adherence rates for separate insurance cohorts [3,14]. The Medicaid cohort was younger and had a higher percentage of African-American individuals. However, in the self-pay/charity care group, the majority was Hispanic [3]. In the pilot study, the differences between the groups on adherence with prescribed medication regimens did not achieve statistical significance. The data were summarized to suggest that nonadherence was more likely in the self-pay/charity care group and more follow-up visits occurred after discharge [3]. During the larger retrospective study there was no difference in number of patients adhering to the regimen at each follow-up visit in each benefit group. The study concluded that the long-acting opiate adherence was influenced only by the benefits of use and that race/ethnicity was not a statistically significant predictor [14].
Factors Associated with Adherence
Multiple studies investigated factors underlying reported analgesic adherence rates for the ethnic and racial groups studied. Both clinical and sociodemographic variables were associated with analgesic adherence (Table 4). These included cancer type and disease stage [3,4,13,14], pain intensity [3,4,13–16], side effects [13,15], type of analgesic prescribed [3,4,13–16], income/socioeconomic status [3,13,14], behavioral history [3,12,13], gender [3,4,12–16], and perceived barriers [3,4,13,15,16].
Cancer Type and Stage
Most studies did not find significant associations between analgesic adherence rates and cancer type and stage [3,12,14]. However, 1 study that sought to identify unique factors underlying analgesic adherence for African Americans and whites found that whites reported higher analgesic adherence in relation to “time since cancer diagnosis,” possibly indicating disease severity and progression [13]. In another study that involved a majority of African-American patients, individuals with colon and rectal cancer had lower adherence rates [4]. In this study, patients with colon and rectal cancer had more analgesic prescriptions (2.5 +/– 2.3 analgesics) compared to patients with other cancer diagnoses. The authors concluded that an increased medication burden might have contributed to a decreased adherence rate. Overall, other cancer types did not correlate with adherence rates [4].
Pain Intensity
Six studies examined pain intensity and duration [3,4,13–16]. Three studies found a difference in reported pain intensity between racial/ethnic groups [3,13,16], 1 found no correlation between pain intensity and race/ethnicity [14], and 3 concluded that pain intensity was a significant predictor of adherence rates [3,13,15].
Meghani and Bruner’s pilot study explored possible correlates associated with intentional and unintentional nonadherence [15]. Overall, individuals were more likely to report forgetfulness (unintentional nonadherence) and to stop taking pain medicine when feeling “worse” (intentional nonadherence) if they believed that it was easier to deal with pain than with the side effects of analgesia [15]. Further, forgetfulness was negatively associated with the need for “stronger” pain medication. Concern about using too much pain medication was positively correlated with both forgetfulness and carelessness. The need for stronger pain medication was also correlated with significantly higher pain levels and lower pain relief [15].
In a comparative study of African Americans and whites, African Americans reported greater cancer pain and lower pain relief on the Brief Pain Inventory (BPI) and had a negative PMI. The PMI measure is a simple index linking the usual severity of cancer pain with the category of medication prescribed to treat it. PMI is calculated by subtracting patient’s pain levels (“pain worst” score from the BPI coded as mild, moderate, or severe) from the most potent analgesia prescribed. A negative PMI implies inadequate analgesic prescription relative to the reported pain level. Pain intensity was a significant factor related to increased adherence in whites but not African Americans. For African Americans, analgesic adherence was predicted by socioeconomic status, provider communication factors, and side effects. Similarly, in another study that compared African Americans and Hispanics, African Americans were more likely to have a negative PMI than Hispanics and were less likely to report that pain medication relieved pain [16]. In a pilot study that compared Medicaid recipients to self-pay/charity care patients, African-American participants had lower reported pain scores than Hispanics and Caucasians [3]. In the larger follow-up study, however, ethnicity did not prove a significant predictor for pain levels [14].
In a study with exclusively African-American patients, a significant correlation was found between pain intensity and adherence; specifically, as intensity increased, adherence increased [4]. Results for the entire African-American cohort indicated that 90% of patients had analgesic prescriptions for cancer-related pain, but 86% continued to report having moderate to severe worst pain [4]. A study that compared African Americans and whites showed that lower pain relief with analgesics was associated with lower adherence to analgesia for cancer pain among whites [13]. For every unit increase in “least pain” scores (indicating lower pain relief) on the BPI item, dose adherence decreased by 2.88%. Pain levels and relief did not explain adherence rates among African Americans. Whites were also more likely to make decisions on analgesic use based on the amount of relief anticipated from the use of analgesics [13] whereas African Americans were more likely to make analgesic use decisions based on analgesic side effects.
Side Effects
In a pilot study that explored the intricacies of adherence, some individuals felt it was easier to deal with pain than with the side effects of pain medications. These individuals were also more likely to report forgetfulness and to stop taking medications if feeling “worse” [15]. One study, which included African-American and white cohorts, found that an increase in the severity of side effects was associated with lower adherence to analgesia for African Americans but not whites. Furthermore, African Americans reported a greater number of analgesic side effects at baseline. African Americans were also more likely to make analgesic decisions based on side effects in comparison to whites participants, who made decisions based on expectation of pain relief [13]. In a study with exclusively African-American patients, patients with concerns about pain medication possibly causing confusion were more likely to have poor adherence [4].
Type of Analgesic Prescribed
In the analyses, 3 studies found a difference between analgesic prescriptions among ethnic groups [12,13,16], 3 found that there was a statistical significance between type of prescription and adherence [4,13,16], and 2 studies [3,14] found no statistical correlation between type of analgesic prescribed and adherence.
In a study of African Americans and Hispanics, both groups took analgesics on an “as-needed” basis despite the guidelines for cancer pain management [16]. However, African Americans reported taking analgesics less than twice daily. Overall, only a small percentage of patients took sustained-release analgesics that require fewer doses per day [16]. Similarly, in another study that compared adherence between African Americans and whites, the overall analgesic adherence rate was different on sub-analysis for specific analgesic prescriptions. The analgesic adherence rates for African Americans ranged from 34% for weak opioids to 63% for long-acting opioids. In comparison, the analgesic adherence rates for whites ranged from 55% for weak opioids to 78% for long-acting opioids [13]. In conclusion, patients on long-acting opioids were more likely to have higher adherence. Adherence rates for African Americans were found in another study. The adherence rate for adjuvant analgesics was highest at 65%, step 2 opioids at 44% and step 3 opioids at 43% [4].
In a study with exclusively African-American patients, poor adherence was significantly correlated with step 3 opioids [4]. Another study that explored the correlation between type of analgesic and adherence found that intentional nonadherence was less likely in individuals that were prescribed step 3 opioids [15]. Specifically, individuals with this behavior were also more likely to report lower pain levels and chose to stop the use of analgesics when feeling better [15].
Within a pilot study that compared benefit programs and payor groups, the differences in the prescription of long-acting opiates did not reach statistical significance [3]. However, in the larger, definitive study, the comparison revealed that patients in the self-pay/charity care group were less likely to receive a prescription for long-acting opiates. The data further revealed that Hispanic and Asian patients were prescribed long-acting opiates at a lower rate compared to the larger sample. Further, African Americans and Caucasians were prescribed long-acting opiates at a higher rate than the larger sample. In another analysis, with benefits and race/ethnicity, benefits were the only statistically significant predictor. While statistically controlling for race/ethnicity, Medicaid patients were 2.4 times more likely to receive a prescription for long-acting opioids than the self-pay/charity care patients [14].
Income/Socioeconomic Status
Three studies in this analysis [3,13,14] found that income and socioeconomic status were significant predictors of analgesic adherence for cancer pain. In a comparison between African Americans and whites, income was the strongest predictor of analgesic adherence for cancer pain in African Americans [13]; specifically, individuals with a household income of less than $10,000 a year had a 41.83% lower percentage of dose adherence. Among whites, income did not have a significant correlation with analgesic rates [13].
A pilot study and larger definitive study [3,14] were conducted to compare the effects of prescription benefits. The prescription benefits included were Medicaid and self-pay/charity care. Through comparison, none of the Medicaid patients reported financial barriers but the self-pay/charity care patients were more likely to report financial barriers to adherence [3]. In the larger study, the findings indicated that there was significant association of adherence by benefits and race/ethnicity. As mentioned above, benefits were a dominant predictor of long acting opiate use and further adherence [14].
Gender
Apart from ethnicity or race as a variable associated with adherence, association of analgesic adherence and gender were observed in 4 studies [3,13–15] and evaluated in 2 studies. One study [4] found that a patient’s gender and education level did not correlate with adherence rates. However, in another study [12] men were more likely to deviate from the prescribed dose. Overall, within the entire cohort [12] men and minority patients were most likely to deviate from the prescribed dosing regimen in comparison to all other patient demographic factors.
Attitudes and Barriers
Five of the 7 studies investigated perceived barriers to analgesic adherence [3,4,13,15,16]. Four used the Barriers Questionnaire II (BQ-II) [18] to further understand patients’ beliefs about cancer pain management [3,12,13,15]. Using this validated tool, 1 study found that non-white individuals had higher scores on the BQ-II than white patients [12]. Within the non-white group in the above study, the mean score on the BQ-II for African Americans was 1.76 (± 0.81) and the mean score for “other” was 2.16 (± 0.93) [12]. Further, low MMAS scores were significantly associated with higher BQ-II scores. Similarly, higher BQ-II scores correlated with opioid deviation toward higher than prescribed dose [12].
Another study with a primarily African-American cohort did not use the BQ-II but asked specific questions in regards to perceived barriers to analgesics. Within the cohort, 87% reported a fear of addiction to pain medicine. Further, 77% had a fear of injection, 75% were concerned about a tolerance for analgesics, and side effects were a major concern. Overall, nausea was the greatest reported concern followed by potential for confusion, which was negatively associated with taking analgesics. Distracting the doctor from curing their illness was a predictor of improved adherence; however, individuals were more likely to take Tylenol for pain relief. Similarly, no significant barrier items affected adherence to NSAIDs. In relation to step 2 opioids, patients who felt it was important to be strong by not talking about pain were more likely to have better adherence [4]. Similar results with African Americans were identified in another study [13]. In the comparison between African Americans and whites, African Americans had more subjective barriers compared to whites. Particularly for African Americans, each unit increase in concern about distracting the doctor from curing the disease, the percentage of dose adherence decreased by 7.44 [13].
In a study that compared payer groups, a questionnaire elicited reasons for nonadherence [3]. Similar reasons for nonadherence emerged including financial, fear of addiction or increased medication use, and running out medication.
Behavioral History
Only 1 study used CAGE (Cut down, Annoyed, Guilty, and Eye-opener), an alcohol-screening questionnaire, to determine a possible relationship with analgesic adherence. In this study, there were 19 cases of opioid deviation, 16% of which were CAGE positive and had severe deviation toward less than the prescribed doses [12]. In further analysis, no association was found between CAGE positively and opioid deviation to higher intake [12]. Two other studies gathered data on history of depression, substance use, and alcohol use but no significant correlation was found [3,13].
Discussion
Previous literature has reported overall analgesic adherence rates among oncology patients ranging from 62% to 72% [23]. Factors at the provider and system level have been considered in past research, but the patient perspective is poorly represented in the literature [13]. A majority of studies on analgesic adherence have been completed with cohorts made up predominantly of white individuals [13,23,24], while others focus on racially homogenous and/or ethnically different populations in other countries [21,25,26].
This review confirms that there is a paucity of well-designed studies that describe the associations between racial and ethnic disparities and adherence to opioids among patients with cancer pain. This is despite the fact that moderate to severe cancer pain in the U.S. is managed mainly with analgesics and specifically with opioids [19]. In addition, cancer patients with health insurance have both more pharmacy claims as well as more claims for higher doses of opioids [20] compared to noncancer patients. The lack of attention to analgesic and opioid adherence among cancer patients is surprising in the light of the recent high-profile initiatives to reduce opioid misuse [31].
Multiple studies highlighted the importance of pain management education and adequate pain assessment for effective analgesic use [4,16]. In the study in the palliative care setting, the authors concluded that patients who are educated, counseled, and monitored by a palliative or supportive care team have less episodes of opioid deviation and trends toward lower opioid use [12]. A systematic review and meta-analysis confirmed findings that educational interventions for patients improved knowledge about cancer pain management, however, most did not improve reported adherence to analgesics [27,28]. These findings emphasize the need for further research on interventions to improve racial/ethnic disparities in analgesic adherence for cancer pain.
Limitations
The findings of this review should be evaluated in the context of the following limitations. First, adherence to a prescribed regimen is a difficult outcome to measure and a majority of studies in this review used subjective measures to assess analgesic adherence for cancer pain. Of note, self-report was the primary measurement employed. Studies in non–cancer pain settings that have evaluated various methodological approaches to adherence measurement found that patients are likely to over-report adherence when using self-report or a diary format in comparison to an electronic monitoring system. Only 1 study in this review used an objective measure of adherence [13]. Some previous studies contend that self-report in comparison to other, objective measurements of medication adherence are accurate [23]. Further research is needed to determine the most accurate measurement of analgesic adherence in cancer patients.
Also, invariably the studies employed an English-speaking sample, which excludes an understanding of analgesic adherence for cancer pain in linguistically diverse Americans. In addition, most studies included patients who were either white Americans or African Americans and some studies lumped several racial ethnic minority subgroups as “nonwhites” or “other.”
A majority of studies were cross-sectional [4,12,15,16]. For instance, studies used a 24-hour time period to assess ATC medication as well as as-needed regimens, which may not capture the information needed to understand adherence to as-needed regimens [4]. With longitudinal studies, a greater understanding of adherence can be determined. However, there is potential bias with studies that track patients primarily at follow-up appointments. Individuals who are compliant with follow-up appointments may present with different analgesic adherence compared to those who do not attend follow-up appointments. This potential bias should be evaluated in longitudinal studies with various sensitivity analyses or using tools that identify healthy user bias.
Most studies recruited patients from outpatient oncology clinics, however, 1 study was conducted with a sample from an outpatient supportive care center managed by a palliative care team [12]. Due to the goals of palliative care, which include specialized treatment for individuals with serious illness and a focus on symptom management and relief, patients in this setting may have a different attitude toward using opioids.
Conclusion
Although data remain limited, our review suggests that while overuse of opioids has been a well-cited concern in patients with chronic non-cancer pain [21,33], cancer patients demonstrate considerable underuse and inconsistent use of prescribed analgesics. This is important as a recent study found that inconsistent adherence to prescribed around-the-clock analgesics, specifically the interaction of strong opioids and inconsistent adherence, is a strong risk factor for hospitalization among cancer outpatients who are prescribed analgesics for pain [1]. Of note, adherence to opioids in patients with cancer may be driven by a unique set of factors and these factors may differ for minorities and non-minority patients. For instance, studies in this review indicate that income is a strong predictor of analgesic adherence for African Americans but not for whites. This is because race and socioeconomic status frequently overlap in the United States [29]. In addition, like cancer pain, analgesic side effects may also be poorly managed among African Americans and other minorities. For example, in 1 study, Meghani et al used a trade-off analysis technique (conjoint analysis) to understand trade-offs African Americans and whites employ in using analgesics for cancer pain [30]. The authors found that African Americans were more likely to make analgesic adherence decisions based on side effects whereas whites were more likely to make adherence decisions based on pain relief [30]. In subsequent analysis, these authors showed that the race effect found in their previous studies was mediated by the type of analgesics prescribed to African Americans vs. whites [31]. African Americans with cancer pain were prescribed analgesics that had a worse side effect profile after statistically adjusting for insurance type and clinical risks such as renal insufficiency [31].
Together, the available evidence indicates that both patients’ socioeconomic status and clinician treatment bias contributes to racial and ethnic disparities in analgesic adherence for cancer pain and subsequent cancer pain outcomes. Thus, future research should investigate interventions for improving analgesic adherence among low-income minorities. Also, there is a need for clinician-level interventions focusing on cognitive bias modification related to cancer pain and side effects management, which appears to relate to analgesic nonadherence among racial/ethnic minorities. In addition, further research is needed to (1) rigorously describe analgesic and opioid adherence for cancer pain, (2) elucidate racial/ethnic and other socioeconomic and clinical disparities in analgesic and opioid adherence for cancer pain; (3) and clarify the role of analgesic and opioid adherence for cancer patients including outcomes for the patients and the health care system.
Corresponding author: Salimah H. Meghani, PhD, MBE, RN, University of Pennsylvania School of Nursing, Room 337, Fagin Hall, 418 Curie Blvd, Philadelphia, PA 19104, [email protected].
Financial disclosures: None.
1. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain - United States, 2016. JAMA 2016;315:1624–45.
2. Meghani SH, Knafl GJ. Patterns of analgesic adherence predict health care utilization among outpatients with cancer pain. Patient Prefer Adher 2016;10:81–98.
3. Bryan M, De La Rosa N, Hill AM, et al. Influence of prescription benefits on reported pain in cancer patients. Pain Med 2008;9:1148–57.
4. Rhee YO, Kim E, Kim B. Assessment of pain and analgesic use in African American cancer patients: Factors related to adherence to analgesics. J Immigr Minor Health 2012;14:1045–51.
5. Laird BJ, Scott AC, Colvin LA, et al. Pain, depression, and fatigue as a symptom cluster in advanced cancer. J Pain Symptom Manage 2011;42:1–11.
6. Ferreira KA, Kimura M, Teixeira MJ, et al. Impact of cancer-related symptom synergisms on health-related quality of life and performance status. J Pain Symptom Manage 2008;35:604–16.
7. Craig BM, Strassels SA. Out-of-pocket prices of opioid analgesics in the United States, 1999-2004. Pain Med 2010;11:240–47.
8. Institute of Medicine: Relieving pain in america: a blueprint for transforming prevention, care, education, and research. Washington, DC: National Academies Press; 2011.
9. Institutes of Medicine. Unequal treatment: confronting racial and ethnic disparities in health care. Washington, DC: National Academies Press; 2003.
10. Meghani SH, Byun E, Gallagher RM. Time to take stock: A meta-analysis and systematic review of analgesic treatment disparities for pain in the United States. Pain Med 2012;13:150–74.
11. Cleeland CS, Gonin R, Baez L, Loehrer P, Pandya KJ. Pain and treatment of pain in minority patients with cancer. The Eastern Cooperative Oncology Group Minority Outpatient Pain Study. Ann Intern Med 1997;127:813–6.
12. Nguyen LMT, Rhondali W, De la Cruz M, et al. Frequency and predictors of patient deviation from prescribed opioids and barriers to opioid pain management in patients with advanced cancer. J Pain Symptom Manage 2013;45:506–16.
13. Meghani SH, Thompson AML, Chittams J, et al. Adherence to analgesics for cancer pain: A comparative study of African Americans and whites using an electronic monitoring device. J Pain 2015;16:825–35.
14. Weider R, DeLaRosa N, Bryan M, et al. Prescription coverage in indigent patients affects the use of long acting opioids in management of cancer pain. Pain Med 2014;15:42–51.
15. Meghani SH, Brune DW. A pilot study to identify correlates of intentional versus unintentional nonadherence. Pain Manag Nurs 2013;14:e22-30.
16. Anderson KO, Mendoza TR, Valero V, et al. Minority cancer patients and their providers: pain management attitudes and practice. Cancer 2000;88:1929–38.
17. Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health 1998;52:377–84.
18. Ward SE, Goldberg N, Miller-McCauley V, et al. Patient-related barriers to management of cancer pain. Pain 1993;52:319–24.
19. Glare PA, Davies PS, Finlay E, et al. Pain in cancer survivors. J Clin Oncol 2014;32:1739–47.
20. van den Beuken-van Everdingen MH, de Rijke JM, Kessels AG, et al. Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol 2007;18:1437–49.
21. Jacobsen R, Samsanaviciene J, Liubarskiene Z, et al. Barriers to cancer pain management in Danish and Lithuanian patients treated in pain and palliative care units. Pain Manag Nurs 2014; 15:51–8.
22. National Institutes of Health. Pathways to prevention: the role of opioids in the treatment of chronic pain. September 29–30, 2014. Executive summary: final report. Accessed 10 Sep 2015 at https://prevention.nih.gov/docs/programs/p2p/ODPPainPanelStatementFinal_10-02-14.pdf.
23. Miaskowski C, Dodd MJ, West C, et al. Lack of adherence with the analgesic regimen: A significant barrier to effective cancer pain management. J Clin Oncol 2001;19:4275–79.
24. Yoong J, Traeger LN, Gallagher ER, et al. A pilot study to investigate adherence to long-acting opioids among patients with advanced lung cancer. J Palliat Med 2013;16:391–6.
25. Lai YH, Keefe FJ, Sun WZ, et al. Relationship between pain-specific beliefs and adherence to analgesic regimens in Taiwanese cancer patients: A preliminary study. J Pain Symptom Manage 2002;24:415–22.
26. Cohen MZ, Musgrave CF, McGuire DB, et al. The cancer pain experience of Israeli adults 65 years and older: the influence of pain interference, symptom severity, and knowledge and attitudes on pain and pain control. Support Care Cancer 2005;13:708–14.
27. Bennett MI, Bagnall AM, Jose Closs S. How effective are patient-based educational interventions in the management of cancer pain? Systematic review and meta analysis. Pain 2009;143:192–9.
28. Oldenmenger WH, Sillevis Smitt PA, van Dooren S, et al. A systematic review on barriers hindering adequate cancer pain management and interventions to reduce them: a critical appraisal. Eur J Cancer 2009;45:1370–80.
29. Meghani SH, Chittams J. Controlling for socioeconomic status in pain disparities research: all-else-equal analysis when “all else” is not equal. Pain Med 2015;16:2222–5.
30. Meghani SH, Chittams J, Hanlon A, Curry J. Measuring preferences for analgesic treatment for cancer pain: how do African Americans and whites perform on choice-based conjoint analysis experiments? BMC Med Inform Decis Mak 2013;13:118.
31. Meghani SH, Kang Y, Chittams J, et al. African Americans with cancer pain are more likely to receive an analgesic with toxic metabolite despite clinical risks: a mediation analysis study. J Clin Oncol 2014;32:2773–9.
32. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids forchronic pain - United States, 2016. MMWR Recomm Rep 2016 Mar 18;65:1–49.
33. Chapman CR, Lipschitz DL, Angst MS, et al. Opioid pharmacotherapy for chronic non-cancer pain in the United States: a research guideline for developing an evidence-base. J Pain 2010;11:807–29.
1. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain - United States, 2016. JAMA 2016;315:1624–45.
2. Meghani SH, Knafl GJ. Patterns of analgesic adherence predict health care utilization among outpatients with cancer pain. Patient Prefer Adher 2016;10:81–98.
3. Bryan M, De La Rosa N, Hill AM, et al. Influence of prescription benefits on reported pain in cancer patients. Pain Med 2008;9:1148–57.
4. Rhee YO, Kim E, Kim B. Assessment of pain and analgesic use in African American cancer patients: Factors related to adherence to analgesics. J Immigr Minor Health 2012;14:1045–51.
5. Laird BJ, Scott AC, Colvin LA, et al. Pain, depression, and fatigue as a symptom cluster in advanced cancer. J Pain Symptom Manage 2011;42:1–11.
6. Ferreira KA, Kimura M, Teixeira MJ, et al. Impact of cancer-related symptom synergisms on health-related quality of life and performance status. J Pain Symptom Manage 2008;35:604–16.
7. Craig BM, Strassels SA. Out-of-pocket prices of opioid analgesics in the United States, 1999-2004. Pain Med 2010;11:240–47.
8. Institute of Medicine: Relieving pain in america: a blueprint for transforming prevention, care, education, and research. Washington, DC: National Academies Press; 2011.
9. Institutes of Medicine. Unequal treatment: confronting racial and ethnic disparities in health care. Washington, DC: National Academies Press; 2003.
10. Meghani SH, Byun E, Gallagher RM. Time to take stock: A meta-analysis and systematic review of analgesic treatment disparities for pain in the United States. Pain Med 2012;13:150–74.
11. Cleeland CS, Gonin R, Baez L, Loehrer P, Pandya KJ. Pain and treatment of pain in minority patients with cancer. The Eastern Cooperative Oncology Group Minority Outpatient Pain Study. Ann Intern Med 1997;127:813–6.
12. Nguyen LMT, Rhondali W, De la Cruz M, et al. Frequency and predictors of patient deviation from prescribed opioids and barriers to opioid pain management in patients with advanced cancer. J Pain Symptom Manage 2013;45:506–16.
13. Meghani SH, Thompson AML, Chittams J, et al. Adherence to analgesics for cancer pain: A comparative study of African Americans and whites using an electronic monitoring device. J Pain 2015;16:825–35.
14. Weider R, DeLaRosa N, Bryan M, et al. Prescription coverage in indigent patients affects the use of long acting opioids in management of cancer pain. Pain Med 2014;15:42–51.
15. Meghani SH, Brune DW. A pilot study to identify correlates of intentional versus unintentional nonadherence. Pain Manag Nurs 2013;14:e22-30.
16. Anderson KO, Mendoza TR, Valero V, et al. Minority cancer patients and their providers: pain management attitudes and practice. Cancer 2000;88:1929–38.
17. Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health 1998;52:377–84.
18. Ward SE, Goldberg N, Miller-McCauley V, et al. Patient-related barriers to management of cancer pain. Pain 1993;52:319–24.
19. Glare PA, Davies PS, Finlay E, et al. Pain in cancer survivors. J Clin Oncol 2014;32:1739–47.
20. van den Beuken-van Everdingen MH, de Rijke JM, Kessels AG, et al. Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol 2007;18:1437–49.
21. Jacobsen R, Samsanaviciene J, Liubarskiene Z, et al. Barriers to cancer pain management in Danish and Lithuanian patients treated in pain and palliative care units. Pain Manag Nurs 2014; 15:51–8.
22. National Institutes of Health. Pathways to prevention: the role of opioids in the treatment of chronic pain. September 29–30, 2014. Executive summary: final report. Accessed 10 Sep 2015 at https://prevention.nih.gov/docs/programs/p2p/ODPPainPanelStatementFinal_10-02-14.pdf.
23. Miaskowski C, Dodd MJ, West C, et al. Lack of adherence with the analgesic regimen: A significant barrier to effective cancer pain management. J Clin Oncol 2001;19:4275–79.
24. Yoong J, Traeger LN, Gallagher ER, et al. A pilot study to investigate adherence to long-acting opioids among patients with advanced lung cancer. J Palliat Med 2013;16:391–6.
25. Lai YH, Keefe FJ, Sun WZ, et al. Relationship between pain-specific beliefs and adherence to analgesic regimens in Taiwanese cancer patients: A preliminary study. J Pain Symptom Manage 2002;24:415–22.
26. Cohen MZ, Musgrave CF, McGuire DB, et al. The cancer pain experience of Israeli adults 65 years and older: the influence of pain interference, symptom severity, and knowledge and attitudes on pain and pain control. Support Care Cancer 2005;13:708–14.
27. Bennett MI, Bagnall AM, Jose Closs S. How effective are patient-based educational interventions in the management of cancer pain? Systematic review and meta analysis. Pain 2009;143:192–9.
28. Oldenmenger WH, Sillevis Smitt PA, van Dooren S, et al. A systematic review on barriers hindering adequate cancer pain management and interventions to reduce them: a critical appraisal. Eur J Cancer 2009;45:1370–80.
29. Meghani SH, Chittams J. Controlling for socioeconomic status in pain disparities research: all-else-equal analysis when “all else” is not equal. Pain Med 2015;16:2222–5.
30. Meghani SH, Chittams J, Hanlon A, Curry J. Measuring preferences for analgesic treatment for cancer pain: how do African Americans and whites perform on choice-based conjoint analysis experiments? BMC Med Inform Decis Mak 2013;13:118.
31. Meghani SH, Kang Y, Chittams J, et al. African Americans with cancer pain are more likely to receive an analgesic with toxic metabolite despite clinical risks: a mediation analysis study. J Clin Oncol 2014;32:2773–9.
32. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids forchronic pain - United States, 2016. MMWR Recomm Rep 2016 Mar 18;65:1–49.
33. Chapman CR, Lipschitz DL, Angst MS, et al. Opioid pharmacotherapy for chronic non-cancer pain in the United States: a research guideline for developing an evidence-base. J Pain 2010;11:807–29.