User login
Pediatric ENT Complaints: An Update
Among all of the causes of ear, nose, and throat (ENT) complaints, acute otitis media (AOM), bacterial sinusitis, and streptococcal pharyngitis (SP) are the most common infections prompting pediatric presentation to the ED. Through a series of case scenarios, along with key questions to help guide the clinician’s work-up, this review covers the proper evaluation and management of pediatric ENT complaints.
Case Scenario 1
A 13-month-old girl presented to the ED with a 1-day history of fever and runny nose. According to her parents, the child had been continually pulling on her ears in apparent discomfort. During history-taking, the parents further informed the emergency physician (EP) that the patient started daycare 4 months earlier and had two elementary school-aged siblings. The patient’s medical history was significant for otitis media, but the parents stated she had not been on antibiotics for over 4 months.
On physical examination, the patient’s vital signs were: blood pressure (BP), 75/50 mm Hg; temperature (T), 101.3°F; slight tachycardia; and normal age-adjusted respiratory rate (RR). Oxygen saturation was 100% on room air. The lungs were clear to auscultation and heart sounds were normal and without murmur. The otolaryngologic examination revealed copious yellow discharge from both nostrils, non-erythematous posterior oropharynx, and erythema to the right tympanic membrane (TM). Questions to Guide the Work-Up: (1) What physical examination findings should be present for accurate diagnosis of otitis media? (2) Will this patient require antibiotics immediately, or is a “wait-and-see” approach indicated? (3) If treatment with antibiotic therapy is warranted, what are the appropriate therapeutic regimen and duration of therapy?
Otitis Media
Acute otitis media is one of the most common presentations in young children. Defined as the rapid onset of signs and symptoms of middle ear inflammation, in conjunction with middle ear effusion (MEE), AOM can develop secondary to a viral or bacterial infection. It is estimated that more than 80% of the pediatric population will experience at least one episode of AOM by age 3 years.1-3
Risk factors for AOM include upper respiratory infection (URI), daycare attendance, siblings, parental smoking, and formula-feeding versus breastfeeding. The patient’s history may include rapid-onset otalgia, fever, irritability, anorexia, and concurrent URI symptoms, as well as other nonspecific symptoms (eg, ear rubbing and/or pulling, crying, changes in behavior and sleep patterns).2-4 In general, otalgia and ear-rubbing in the nonverbal patient seem to have the best predictive value for AOM.3
Signs and Symptoms
A normal TM should be translucent and pearly gray, with visible landmarks of the manubrium of malleus and pars flaccida. A TM that is bulging, cloudy, and immobile is the most consistent finding in AOM, with bulging having a specificity of 97%. Redness of the tympanic membrane is not a useful predictor of AOM as this finding is noted in upward of 30% of pediatric patients on general examination but in <1% of AOM diagnoses in the absence of a bulging TM.
Diagnosis
Pneumatic otoscopy is the gold standard for diagnosing for MEE; however, this examination can be difficult in younger, often uncooperative, patients. A TM that does not perceptibly move with either positive or negative insufflation pressure greatly enhances the diagnostic accuracy for MEE over the use of visible eardrum characteristics alone.2-5
Acute otitis media is a clinical diagnosis and does not require imaging studies or laboratory evaluation unless more serious processes, such as skull fracture, mastoiditis, or intracranial abscess, are being considered.2,3
Treatment and Management
Analgesia. The first step in managing patients with AOM is to provide analgesia. In most cases, acetaminophen in patients over 2 months of age, or ibuprofen in patients over 6 months of age, are adequate choices for managing pain. When either of these analgesics is administered in the clinic/ED setting, patients should be monitored to assure adequate pain relief prior to discharge.
While topical agents such as combination antipyrine-benzocaine suspensions were commonly given in the past to alleviate the pain associated with AOM, there are limited data to support their effectiveness. As such, in July 2015, the US Food and Drug Administration ordered manufacturers to halt production on these unapproved prescription products.3,4,6 There are also no randomized controlled trials (RCTs) to support the use of decongestants or antihistamines for resolution of AOM or otalgia.3,7
Antibiotic Therapy. The most common bacteria associated with AOM are Streptococcus pneumonia, nontypeable Hemophilus influenza, and Moraxella catarrhalis. In 30% of patients, the causative etiology is viral. When the decision is made to treat AOM, high-dose amoxicillin is still considered the first-line treatment, despite ever evolving susceptibilities of bacteria.
When a child is noted to have been treated with amoxicillin within a 30-day period or who has concurrent conjunctivitis, amoxicillin-clavulanate is considered the first-line treatment.2-4,7,8 The current American Academy of Pediatrics (AAP) guidelines recommend 10 days of antibiotic therapy for children younger than age 2 years, and 5 to 7 days for children older than age 2 years who have uncomplicated AOM. Intramuscular (IM) ceftriaxone is an acceptable first-line agent in a child who is unable to tolerate oral medications or who is suffering persistent emesis. Intramuscular ceftriaxone can be given as a single dose of 50 mg/kg, though the patient should be followed closely as studies show that a second dose may be necessary 5 to 7 days later to prevent infection recurrence. The IM dose of ceftriaxone 50 mg/kg can also be given if treatment with other antibiotics fails to resolve the AOM (failure is defined as no improvement in the patient’s condition 48 to 72 hours from treatment). In such cases, ceftriaxone is given in three consecutive doses.3,4,7
Wait-and-See Approach. Studies of patients whose AOM was confirmed via culture (19% were positive for S pneumoniae, 48% for H influenza, and 78% for M catarrhalis) showed bacterial clearance without antibiotic intervention.4 Based on these findings, the 2013 revised AAP evidence-based clinical practice guidelines indicate an initial watching-and-waiting period combined with pain management for patients older than 6 months of age who are diagnosed with unilateral AOM in the absence of severe symptoms (ie, fever is lower than 102.2˚F or patient has severe otalgia).4 A period of observation prior to treatment is also endorsed for children older than age 2 years who exhibit nonsevere symptoms—even if they have bilateral disease.4
Conversely, all patients younger than age 6 months and all children with severe symptoms should be treated with antibiotics at diagnosis.3,4 The wait-and-see approach, recommends an observation period of 24 to 48 hours for children in the lower risk group prior to antibiotic administration. Delayed antibiotic administration can be performed by a physician in an office/ED follow-up or as a safety-net antibiotic prescription (SNAP) sent home with the family on the initial ED encounter.2-4,8,9
Case 1 Resolution
Given this patient’s unilateral and nonsevere symptoms (minor otalgia, fever <102.2°F), age older than 6 months, and no recent antibiotic use), she was treated with oral ibuprofen. At discharge, the parents were given a 10-day SNAP prescription of high-dose amoxicillin (90 mg/kg/d, divided into two daily doses) and instructed to fill the prescription only if the patient’s otalgia did not improve in 1 or 2 days.
Case Scenario 2
A 5-year-old boy was presented for evaluation by his parents, who stated that their son had been sick since he had started kindergarten in the fall. The patient had a 10-day history of cough, thick runny nose, and facial pain, and a 1-day history of new-onset fever and headache. His parents further noted that the patient had been seen by his pediatrician several times over the past week. At each of these visits, the pediatrician had informed them that their son had a virus.
Vital signs on examination were: BP, 100/60 mm Hg; heart rate (HR), 112 beats/min; normal age-adjusted RR; and T, 102.6oF. Oxygen saturation was 100% on room air. The patient did not appear toxic, his lungs were clear on auscultation, and there were no other clinical signs suggestive of meningitis. The otolaryngologic examination revealed bilateral thick mucoid drainage and visible edema and erythema of the nasal turbinates. The patient was noted to have some facial pain in the maxillary area bilaterally.
Questions to Guide the Work-Up: (1) Does the patient have a prolonged URI or pediatric sinusitis, and what differentiates the two conditions? (2) What sinuses are present in a 5-year-old patient? (3) What treatment modalities are available for sinusitis? (4) Is imaging of the sinuses helpful in confirming the diagnosis?
Acute Bacterial Sinusitis
Rhinosinusitis is an inflammation of the mucosal lining of the nasal passages and paranasal sinuses. Most cases occur secondary to a viral URI and resolve spontaneously in 99% of the pediatric population.10,11
Acute bacterial sinusitis (ABS) is an inflammation of the same mucosal lining of the nasal passages secondary to bacterial overgrowth that lasts more than 10 days, with complete resolution by 30 days.12,13 When evaluating a pediatric patient for ABS, it is important to consider the sinus growth and development: If the sinus is not yet formed, it therefore cannot be the location of an ABS.13 The ethmoid and maxillary sinuses are present at birth, aerated within 4 months of life, and are fully developed by age 12 years. The sphenoid sinuses begin development around age 3 years, are aerated by age 7 or 8 years, and are fully developed by age 18 to 20 years. The frontal sinuses begin development around age 8 years and are aerated and fully developed by age 12 to 15 years.10,13,14 While most guidelines focus on children older than age 1 year (due to very small infantile sinuses), ABS does occur in children younger than age 1 year.12,14
Signs and Symptoms
Differentiation between a viral URI/rhinosinusitis and ABS is a challenge and can be based upon severity of symptoms as well as length of illness. Symptoms of ABS are typically present and persistent for more than 10 days, without improvement. Continuing illness and worsening of symptoms are identifying features of ABS given most viral URIs gradually resolve within a 10-day timeframe. Other common symptoms include milky/thick nasal discharge, fever, predominantly nocturnal cough, and headache. Other less common symptoms include facial pain, toothache, malodorous breath, and periorbital edema. On physical examination, erythema and edema of the turbinates, as well as reproducible pain over aerated sinuses, are suggestive of ABS.10-14
Diagnosis
In the acute care setting, diagnosis of ABS should be clinical in nature. Neither imaging nor laboratory work-up is generally required secondary to their poor diagnostic specificity for ABS. The bacteria involved in ABS are similar to those associated with AOM, with S pneumonia, nontypeable H influenza, and M catarrhalis being the predominant organisms.10-15
Treatment and Management
Treatment of ABS is generally recommended once the diagnosis is made, though this is based largely on expert opinion as there are limited RCTs available.13 However, available studies do show a more rapid improvement in children on antibiotic therapy than those on placebo.15,16
Antibiotic Therapy. Amoxicillin remains the antimicrobial agent of choice for first-line treatment of uncomplicated ABS forsituations in which antimicrobial resistance is not suspected. In communities with a high prevalence of nonsusceptible S pneumoniae (>10%, including intermediate- and high-level resistance), treatment may be initiated at 80 to 90 mg/kg/d in two divided doses, with a maximum of 2 g per dose.
Patients presenting with moderate to severe illness, as well as those who are younger than 2 years, attend childcare, or have recently been treated with an antimicrobial, may receive high-dose amoxicillin-clavulanate as initial therapy given the elevated beta-lactamase production of the common bacteria that cause ABS.
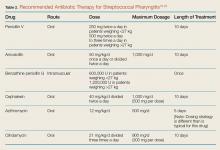
Second-line alternatives include azithromycin, cefdinir, and sulfamethoxizole-trimethoprim (Table 1). There are data to suggest higher rates of decreased susceptibility of S pneumonia and H influenza to third-generation cephalosporins, and the addition of clindamycin may be warranted when utilizing those medications. Treatment is recommended for 10 to 14 days, though improvement should be noted within 1 to 3 days.10-12,14-17
Adjuvant Therapy. Additional therapies include nasal irrigation, decongestants, antihistamines, and intranasal steroids; however, there are only anecdotal reports of their efficacy in providing symptom relief. Therefore, there are insufficient evidence-based data to support or refute the role of these adjuvant therapies in treating pediatric patients with ABS.9,13
Case 2 Resolution
The prolonged duration and severity of symptoms (high fever and headache) and the gradual worsening of the clinical course (ie, late-onset fever) in this patient all suggest ABS rather than a simple prolonged URI. The physical examination findings of inflamed turbinates and facial pain further increase the specificity for ABS. The patient was started on oral amoxicillin-clavulanate with planned treatment for 14 days. At discharge, his parents were instructed to follow-up with the patient’s pediatrician in 3 days to ensure a degree of clinical resolution.
Case Scenario 3
A 4-year-old boy was presented by his parents for evaluation of a 2-day history of a persistent and unimproved sore throat. The patient’s mother indicated that the child’s oral T upon returning home earlier from preschool was 101.2oF. She further noted that her 17-month-old daughter and 8-year-old son also experienced similar symptoms which had self-resolved. Triage vital signs were: T, 100.8oF, orally; BP, HR, and RR were all within normal limits. Oxygen saturation was 100% on room air.
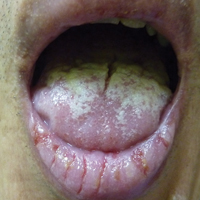
On physical examination, the child was noted to have anterior cervical lymph nodes bilaterally and an erythematous oropharynx with exudate noted on both tonsils. There were no cutaneous abnormalities, nasal edema, erythema, or drainage. Based on the clinical examination, the EP was suspicious for SP.
Questions to Guide the Work-Up: (1) Is SP diagnosed based on clinical findings alone in this patient’s age group? (2) At what age in the pediatric population is it appropriate to perform a rapid streptococcal antigen test? (3) Are there medications other than antibiotics that are beneficial in treating symptomatic SP?
Streptococcal Pharyngitis
Streptococcal pharyngitis is a clinical condition caused by group A beta-hemolytic S pyogens. This bacterium is responsible for multiple conditions, including pharyngitis, skin infections, poststreptococcal glomerulonephritis, and rheumatic fever, as well as invasive syndromes. (This case focuses solely on SP).
Pharyngitis can occur secondary to a viral or bacterial infection, and SP is the most common cause of pediatric bacterial pharyngitis. It is estimated that children aged 5 to 15 years are more commonly diagnosed with SP, although approximately 24% of children younger than age 5 years with pharyngitis symptoms will be ultimately diagnosed with SP.
Signs and Symptoms
Typical symptoms include fever, pharyngitis, generalized abdominal pain, nausea, vomiting, headache, and absence of viral URI symptoms (eg, cough, nasal discharge). However, younger patients with SP may have clinical findings of prolonged nasal drainage and excoriated nares. Examination findings may include swollen and tender anterior cervical lymph nodes; generalized edema and erythema of the posterior pharynx; tonsillar exudates; and palatal petechiae.
Diagnosis
Centor Criteria. The Centor criteria were developed to assist practitioners in identifying patients with potential SP. Criteria for patients older than age 15 years include fever, absence of cough, tonsillar exudates, and tender anterior cervical lymphadenopathy. A modified Centor criteria was later established to include children older than age 5 years, with children between ages 5 and 15 years being the fifth variable in the modified score. In general, patients with a score of 4 or 5 (presence of each variable = 1 point) are most likely to test positive for SP on rapid antigen testing (RAT) or culture.18-20
Swab, Rapid Antigen Testing, and Culture. Swabbing the throat and RAT and/or culture should be performed in most children with suspected SP because the clinical features alone do not reliably discriminate SP from viral pharyngitis. Rapid antigen testing is only specific for group A beta-hemolytic streptococcal species, which is the only streptococcal species that is routinely treated with antibiotics in the setting of acute pharyngitis. It is unlikely for a patient with a score of 0 or 1 to have SP, and several sources suggest neither testing nor treating this cohort, but rather to consider an alternative diagnosis.18-20
Within the population of children and young adolescents, due to a RAT sensitivity of 70% to 90%, a negative result should always be backed-up by a throat culture, and treatment initiated if results of the culture are later found to be positive. As the current generation of RAT tests have a high specificity, a positive RAT does not necessitate a back-up culture, and treatment is indicated without further investigation.19,20
Routine RAT is not recommended in children younger than age 3 years as patients in this age group are at low-risk of developing rheumatic fever. One notable exception for these very young children would be if there are siblings in the home with confirmed SP, in which case, RAT should be considered in the clinical context of SP.21 Adolescents over age 15 years are another cohort with a low likelihood of developing rheumatic fever, though they can develop other poststreptococcal complications, such as glomerulonephritis.
The US Centers for Disease Control and Prevention/American Academy of Family Practitioners (AAFP) guidelines suggest that pharyngitis in older adolescents can be approached in a similar fashion to adults, with empiric therapy for a Centor score of 3 or 4, RAT (without the need for follow-up culture) for Centor score of 2, and neither testing nor treating patients with a score of 0 or 1.19
Treatment and Management
Streptococcal pharyngitis is treated mainly to prevent the poststreptococcal complications of rheumatic fever, though it will not prevent poststreptococcal glomerulonephritis. Treatment of SP also facilitates resolution of symptoms and return to baseline activities.
Antibiotic Therapy. Patients who have a positive RAT or a follow-up throat culture positive for group A streptococcus should be given antibiotics. The gold standard treatment is penicillin V orally for 10 days.
Corticosteroid Therapy. The use of corticosteroids for symptom control of SP in pediatric patients is controversial. Although the Infectious Disease Society of America does not recommend corticosteroid therapy in the treatment of SP, several studies show such therapy (namely dexamethasone), improves pain in children and adolescents diagnosed with SP, but without significant change to the overall disease course.21,23-26
Case 3 Resolution
The patient had a modified Centor criteria score of 4, as well as siblings with similar symptoms. In following current guidelines, the EP performed a RAT and back-up culture. The RAT was negative in the ED, but the back-up culture was subsequently positive, and the child was started on a 10-day course of oral amoxicillin.
Conclusion
When evaluating pediatric patients presenting with ENT signs and symptoms such as ear pain and erythema, fever, sore throat, nasal congestion and discharge, a thorough physical examination and history-taking—including recent illness of any siblings—along with testing when indicated, is essential to guide the diagnosis and determine appropriate treatment and management. In addition to administering antibiotic therapy when such is warranted, the EP should provide appropriate analgesia to manage the patient’s pain and assure relief prior to discharge.
1. Rosenfeld RM, Shin JJ, Schwartz SR, et al. Clinical practice guideline: otitis media with effusion (Update). Otolaryngol Head Neck Surg. 2016;154(1 Suppl):S1-S41. doi:10.1177/0194599815623467.
2. Acute Otitis Media Guideline Team, Cincinnati Children’s Hospital Medical Center. Evidence-based care guideline for medical management of acute otitis media in children 2 months to 13 years of age. http://f.i-md.com/medinfo/material/4f4/4eb132ba44ae4ffe12a814f4/4eb132d744ae4ffe12a814f7.pdf. August 2006. Accessed December 29, 2016.
3. Nesbit CE, Powers MC. An evidence-based approach to managing acute otitis media. Pediatr Emerg Med Pract. 2013;10(4):1-26; quiz 26-27.
4. Lieberthal AS, Carroll AE, Chonmaitree T, et al. The diagnosis and management of acute otitis media. Pediatrics. 2013;131(3):e964-e999. doi:10.1542/peds.2012-3488.
5. American Academy of Family Physicians; American Academy of Otolaryngology-Head and Neck Surgery; American Academy of Pediatrics Subcommittee on Otitis Media With Effusion. Otitis media with effusion. Pediatrics. 2004;113(5):1412-1429.
6. US Food and Drug Administration Web site. FDA: Use only approved prescription ear drops. http://www.fda.gov/ForConsumers/ConsumerUpdates/-ucm453087.htm. Updated July 10, 2015. Accessed December 15, 2016.
7. Sack F. An evidence based approach to the management of uncomplicated acute otitis media in children. Int Pediatrics. 2005;20(1):44-46.
8. Johnson NC, Holger JS. Pediatric acute otitis media: the case for delayed antibiotic treatment. J Emerg Med. 2007;32(3):279-284. doi:10.1016/j.jemermed.2006.07.029.
9. Spiro DM, Tay KY, Arnold DH, Dziura JD, Baker MD, Shapiro ED. Wait-and-see prescription for the treatment of acute otitis media: a randomized controlled trial. JAMA. 2006;296(10):1235-1241. doi:10.1001/jama.296.10.1235.
10. Brook I. Management of acute rhinosinusitis in pediatric patients. Pediatr Emerg Med Pract. 2012;9(5):1-24.
11. Ferdman RM, Linzer JF Jr. The runny nose in the emergency department: rhinitis and sinusitis. Clin Pediatr Emerg Med. 2007;8(2):123-130.
12. Acute Bacterial Sinusitis Guideline Team, Cincinnati Children’s Hospital Medical Center: Evidence-based care guideline for medical management of acute bacterial sinusitis in children 1 through 18 years of age. http://www.antibioticos.msssi.gob.es/PDF/sinusitisguideline.pdf. July 7, 2006. Accessed December 29, 2016
13. Holt KR, Murdoch Cuenca M, Cuenca PJ, Johnston GM. acute pediatric sinusitis and “the 10-day rule.” Pediatr Emerg Med Pract. 2006;3(2):1-16.
14. American Academy of Pediatrics. Subcommittee on Management of Sinusitis and Committee on Quality Improvement. Clinical practice guideline: management of sinusitis. Pediatrics. 2001;108(3):798-808.
15. Wald ER, Nash D, Eickhoff J. Effectiveness of amoxicillin/clavulanate potassium in the treatment of acute bacterial sinusitis in children. Pediatrics. 2009;124(1):9-15. doi:10.1542/peds.2008-2902.
16. Arroll B, Kenealy T. Are antibiotics effective for acute purulent rhinitis? Systematic review and meta-analysis of placebo controlled randomised trials. BMJ. 2006;333(7562):279. doi:10.1136/bmj.38891.681215.AE.
17. McQuillan L, Crane LA, Kempe A. Diagnosis and management of acute sinusitis by pediatricians. Pediatrics. 2009;123(2):e193-e198.
18. Singer JI, Fontanette R. Recognizable and suspected group A beta-hemolytic streptococcal syndromes. Pediatr Emerg Med Rep. 2010;15(11):129-144.
19. Weglowski J. An evidence-based approach to the evaluation and treatment of pharyngitis in children. Pediatr Emerg Med Pract. 2011;8(12):1-28.
20. Gerber MA, Baltimore RS, Eaton CB, et al. Prevention of rheumatic fever and diagnosis and treatment of acute Streptococcal pharyngitis: a scientific statement from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, the Interdisciplinary Council on Functional Genomics and Translational Biology, and the Interdisciplinary Council on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation. 2009;119(11):1541-1551. doi:10.1161/CIRCULATIONAHA.109.191959.
21. Shulman ST, Bisno AL, Clegg HW, et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis. 2012;55(10):1279-1282. doi:10.1093/cid/cis847.
22. Clegg HW, Ryan AG, Dallas SD, et al. Treatment of streptococcal pharyngitis with once-daily compared with twice-daily amoxicillin: a noninferiority trial. Pediatr Infect Dis J. 2006;25(9):761-767. doi:10.1097/01.inf.0000235678.46805.92.
23. Bulloch B, Kabani A, Tenenbein M. Oral dexamethasone for the treatment of pain in children with acute pharyngitis: a randomized, double-blind, placebo-controlled trial. Ann Emerg Med. 2003;41(5):601-608. doi:10.1067/mem.2003.136.
24. Niland ML, Bonsu BK, Nuss KE, Goodman DG. A pilot study of 1 versus 3 days of dexamethasone as add-on therapy in children with streptococcal pharyngitis. Pediatr Infect Dis J. 2006;25(6):477-481. doi:10.1097/01.inf.0000219469.95772.3f.
25. Wei JL, Kasperbauer JL, Weaver AL, Boggust AJ. Efficacy of single-dose dexamethasone as adjuvant therapy for acute pharyngitis. Laryngoscope. 2002;112(1):87-93. doi:10.1097/00005537-200201000-00016.
26. Hayward G, Thompson M, Heneghan C, Perera R, Del Mar C, Glasziou P. Corticosteroids for pain relief in sore throat: systematic review and meta-analysis. BMJ. 2009;339:b2976. doi:10.1136/bmj.b2976.
Among all of the causes of ear, nose, and throat (ENT) complaints, acute otitis media (AOM), bacterial sinusitis, and streptococcal pharyngitis (SP) are the most common infections prompting pediatric presentation to the ED. Through a series of case scenarios, along with key questions to help guide the clinician’s work-up, this review covers the proper evaluation and management of pediatric ENT complaints.
Case Scenario 1
A 13-month-old girl presented to the ED with a 1-day history of fever and runny nose. According to her parents, the child had been continually pulling on her ears in apparent discomfort. During history-taking, the parents further informed the emergency physician (EP) that the patient started daycare 4 months earlier and had two elementary school-aged siblings. The patient’s medical history was significant for otitis media, but the parents stated she had not been on antibiotics for over 4 months.
On physical examination, the patient’s vital signs were: blood pressure (BP), 75/50 mm Hg; temperature (T), 101.3°F; slight tachycardia; and normal age-adjusted respiratory rate (RR). Oxygen saturation was 100% on room air. The lungs were clear to auscultation and heart sounds were normal and without murmur. The otolaryngologic examination revealed copious yellow discharge from both nostrils, non-erythematous posterior oropharynx, and erythema to the right tympanic membrane (TM). Questions to Guide the Work-Up: (1) What physical examination findings should be present for accurate diagnosis of otitis media? (2) Will this patient require antibiotics immediately, or is a “wait-and-see” approach indicated? (3) If treatment with antibiotic therapy is warranted, what are the appropriate therapeutic regimen and duration of therapy?
Otitis Media
Acute otitis media is one of the most common presentations in young children. Defined as the rapid onset of signs and symptoms of middle ear inflammation, in conjunction with middle ear effusion (MEE), AOM can develop secondary to a viral or bacterial infection. It is estimated that more than 80% of the pediatric population will experience at least one episode of AOM by age 3 years.1-3
Risk factors for AOM include upper respiratory infection (URI), daycare attendance, siblings, parental smoking, and formula-feeding versus breastfeeding. The patient’s history may include rapid-onset otalgia, fever, irritability, anorexia, and concurrent URI symptoms, as well as other nonspecific symptoms (eg, ear rubbing and/or pulling, crying, changes in behavior and sleep patterns).2-4 In general, otalgia and ear-rubbing in the nonverbal patient seem to have the best predictive value for AOM.3
Signs and Symptoms
A normal TM should be translucent and pearly gray, with visible landmarks of the manubrium of malleus and pars flaccida. A TM that is bulging, cloudy, and immobile is the most consistent finding in AOM, with bulging having a specificity of 97%. Redness of the tympanic membrane is not a useful predictor of AOM as this finding is noted in upward of 30% of pediatric patients on general examination but in <1% of AOM diagnoses in the absence of a bulging TM.
Diagnosis
Pneumatic otoscopy is the gold standard for diagnosing for MEE; however, this examination can be difficult in younger, often uncooperative, patients. A TM that does not perceptibly move with either positive or negative insufflation pressure greatly enhances the diagnostic accuracy for MEE over the use of visible eardrum characteristics alone.2-5
Acute otitis media is a clinical diagnosis and does not require imaging studies or laboratory evaluation unless more serious processes, such as skull fracture, mastoiditis, or intracranial abscess, are being considered.2,3
Treatment and Management
Analgesia. The first step in managing patients with AOM is to provide analgesia. In most cases, acetaminophen in patients over 2 months of age, or ibuprofen in patients over 6 months of age, are adequate choices for managing pain. When either of these analgesics is administered in the clinic/ED setting, patients should be monitored to assure adequate pain relief prior to discharge.
While topical agents such as combination antipyrine-benzocaine suspensions were commonly given in the past to alleviate the pain associated with AOM, there are limited data to support their effectiveness. As such, in July 2015, the US Food and Drug Administration ordered manufacturers to halt production on these unapproved prescription products.3,4,6 There are also no randomized controlled trials (RCTs) to support the use of decongestants or antihistamines for resolution of AOM or otalgia.3,7
Antibiotic Therapy. The most common bacteria associated with AOM are Streptococcus pneumonia, nontypeable Hemophilus influenza, and Moraxella catarrhalis. In 30% of patients, the causative etiology is viral. When the decision is made to treat AOM, high-dose amoxicillin is still considered the first-line treatment, despite ever evolving susceptibilities of bacteria.
When a child is noted to have been treated with amoxicillin within a 30-day period or who has concurrent conjunctivitis, amoxicillin-clavulanate is considered the first-line treatment.2-4,7,8 The current American Academy of Pediatrics (AAP) guidelines recommend 10 days of antibiotic therapy for children younger than age 2 years, and 5 to 7 days for children older than age 2 years who have uncomplicated AOM. Intramuscular (IM) ceftriaxone is an acceptable first-line agent in a child who is unable to tolerate oral medications or who is suffering persistent emesis. Intramuscular ceftriaxone can be given as a single dose of 50 mg/kg, though the patient should be followed closely as studies show that a second dose may be necessary 5 to 7 days later to prevent infection recurrence. The IM dose of ceftriaxone 50 mg/kg can also be given if treatment with other antibiotics fails to resolve the AOM (failure is defined as no improvement in the patient’s condition 48 to 72 hours from treatment). In such cases, ceftriaxone is given in three consecutive doses.3,4,7
Wait-and-See Approach. Studies of patients whose AOM was confirmed via culture (19% were positive for S pneumoniae, 48% for H influenza, and 78% for M catarrhalis) showed bacterial clearance without antibiotic intervention.4 Based on these findings, the 2013 revised AAP evidence-based clinical practice guidelines indicate an initial watching-and-waiting period combined with pain management for patients older than 6 months of age who are diagnosed with unilateral AOM in the absence of severe symptoms (ie, fever is lower than 102.2˚F or patient has severe otalgia).4 A period of observation prior to treatment is also endorsed for children older than age 2 years who exhibit nonsevere symptoms—even if they have bilateral disease.4
Conversely, all patients younger than age 6 months and all children with severe symptoms should be treated with antibiotics at diagnosis.3,4 The wait-and-see approach, recommends an observation period of 24 to 48 hours for children in the lower risk group prior to antibiotic administration. Delayed antibiotic administration can be performed by a physician in an office/ED follow-up or as a safety-net antibiotic prescription (SNAP) sent home with the family on the initial ED encounter.2-4,8,9
Case 1 Resolution
Given this patient’s unilateral and nonsevere symptoms (minor otalgia, fever <102.2°F), age older than 6 months, and no recent antibiotic use), she was treated with oral ibuprofen. At discharge, the parents were given a 10-day SNAP prescription of high-dose amoxicillin (90 mg/kg/d, divided into two daily doses) and instructed to fill the prescription only if the patient’s otalgia did not improve in 1 or 2 days.
Case Scenario 2
A 5-year-old boy was presented for evaluation by his parents, who stated that their son had been sick since he had started kindergarten in the fall. The patient had a 10-day history of cough, thick runny nose, and facial pain, and a 1-day history of new-onset fever and headache. His parents further noted that the patient had been seen by his pediatrician several times over the past week. At each of these visits, the pediatrician had informed them that their son had a virus.
Vital signs on examination were: BP, 100/60 mm Hg; heart rate (HR), 112 beats/min; normal age-adjusted RR; and T, 102.6oF. Oxygen saturation was 100% on room air. The patient did not appear toxic, his lungs were clear on auscultation, and there were no other clinical signs suggestive of meningitis. The otolaryngologic examination revealed bilateral thick mucoid drainage and visible edema and erythema of the nasal turbinates. The patient was noted to have some facial pain in the maxillary area bilaterally.
Questions to Guide the Work-Up: (1) Does the patient have a prolonged URI or pediatric sinusitis, and what differentiates the two conditions? (2) What sinuses are present in a 5-year-old patient? (3) What treatment modalities are available for sinusitis? (4) Is imaging of the sinuses helpful in confirming the diagnosis?
Acute Bacterial Sinusitis
Rhinosinusitis is an inflammation of the mucosal lining of the nasal passages and paranasal sinuses. Most cases occur secondary to a viral URI and resolve spontaneously in 99% of the pediatric population.10,11
Acute bacterial sinusitis (ABS) is an inflammation of the same mucosal lining of the nasal passages secondary to bacterial overgrowth that lasts more than 10 days, with complete resolution by 30 days.12,13 When evaluating a pediatric patient for ABS, it is important to consider the sinus growth and development: If the sinus is not yet formed, it therefore cannot be the location of an ABS.13 The ethmoid and maxillary sinuses are present at birth, aerated within 4 months of life, and are fully developed by age 12 years. The sphenoid sinuses begin development around age 3 years, are aerated by age 7 or 8 years, and are fully developed by age 18 to 20 years. The frontal sinuses begin development around age 8 years and are aerated and fully developed by age 12 to 15 years.10,13,14 While most guidelines focus on children older than age 1 year (due to very small infantile sinuses), ABS does occur in children younger than age 1 year.12,14
Signs and Symptoms
Differentiation between a viral URI/rhinosinusitis and ABS is a challenge and can be based upon severity of symptoms as well as length of illness. Symptoms of ABS are typically present and persistent for more than 10 days, without improvement. Continuing illness and worsening of symptoms are identifying features of ABS given most viral URIs gradually resolve within a 10-day timeframe. Other common symptoms include milky/thick nasal discharge, fever, predominantly nocturnal cough, and headache. Other less common symptoms include facial pain, toothache, malodorous breath, and periorbital edema. On physical examination, erythema and edema of the turbinates, as well as reproducible pain over aerated sinuses, are suggestive of ABS.10-14
Diagnosis
In the acute care setting, diagnosis of ABS should be clinical in nature. Neither imaging nor laboratory work-up is generally required secondary to their poor diagnostic specificity for ABS. The bacteria involved in ABS are similar to those associated with AOM, with S pneumonia, nontypeable H influenza, and M catarrhalis being the predominant organisms.10-15
Treatment and Management
Treatment of ABS is generally recommended once the diagnosis is made, though this is based largely on expert opinion as there are limited RCTs available.13 However, available studies do show a more rapid improvement in children on antibiotic therapy than those on placebo.15,16
Antibiotic Therapy. Amoxicillin remains the antimicrobial agent of choice for first-line treatment of uncomplicated ABS forsituations in which antimicrobial resistance is not suspected. In communities with a high prevalence of nonsusceptible S pneumoniae (>10%, including intermediate- and high-level resistance), treatment may be initiated at 80 to 90 mg/kg/d in two divided doses, with a maximum of 2 g per dose.
Patients presenting with moderate to severe illness, as well as those who are younger than 2 years, attend childcare, or have recently been treated with an antimicrobial, may receive high-dose amoxicillin-clavulanate as initial therapy given the elevated beta-lactamase production of the common bacteria that cause ABS.
Second-line alternatives include azithromycin, cefdinir, and sulfamethoxizole-trimethoprim (Table 1). There are data to suggest higher rates of decreased susceptibility of S pneumonia and H influenza to third-generation cephalosporins, and the addition of clindamycin may be warranted when utilizing those medications. Treatment is recommended for 10 to 14 days, though improvement should be noted within 1 to 3 days.10-12,14-17
Adjuvant Therapy. Additional therapies include nasal irrigation, decongestants, antihistamines, and intranasal steroids; however, there are only anecdotal reports of their efficacy in providing symptom relief. Therefore, there are insufficient evidence-based data to support or refute the role of these adjuvant therapies in treating pediatric patients with ABS.9,13
Case 2 Resolution
The prolonged duration and severity of symptoms (high fever and headache) and the gradual worsening of the clinical course (ie, late-onset fever) in this patient all suggest ABS rather than a simple prolonged URI. The physical examination findings of inflamed turbinates and facial pain further increase the specificity for ABS. The patient was started on oral amoxicillin-clavulanate with planned treatment for 14 days. At discharge, his parents were instructed to follow-up with the patient’s pediatrician in 3 days to ensure a degree of clinical resolution.
Case Scenario 3
A 4-year-old boy was presented by his parents for evaluation of a 2-day history of a persistent and unimproved sore throat. The patient’s mother indicated that the child’s oral T upon returning home earlier from preschool was 101.2oF. She further noted that her 17-month-old daughter and 8-year-old son also experienced similar symptoms which had self-resolved. Triage vital signs were: T, 100.8oF, orally; BP, HR, and RR were all within normal limits. Oxygen saturation was 100% on room air.
On physical examination, the child was noted to have anterior cervical lymph nodes bilaterally and an erythematous oropharynx with exudate noted on both tonsils. There were no cutaneous abnormalities, nasal edema, erythema, or drainage. Based on the clinical examination, the EP was suspicious for SP.
Questions to Guide the Work-Up: (1) Is SP diagnosed based on clinical findings alone in this patient’s age group? (2) At what age in the pediatric population is it appropriate to perform a rapid streptococcal antigen test? (3) Are there medications other than antibiotics that are beneficial in treating symptomatic SP?
Streptococcal Pharyngitis
Streptococcal pharyngitis is a clinical condition caused by group A beta-hemolytic S pyogens. This bacterium is responsible for multiple conditions, including pharyngitis, skin infections, poststreptococcal glomerulonephritis, and rheumatic fever, as well as invasive syndromes. (This case focuses solely on SP).
Pharyngitis can occur secondary to a viral or bacterial infection, and SP is the most common cause of pediatric bacterial pharyngitis. It is estimated that children aged 5 to 15 years are more commonly diagnosed with SP, although approximately 24% of children younger than age 5 years with pharyngitis symptoms will be ultimately diagnosed with SP.
Signs and Symptoms
Typical symptoms include fever, pharyngitis, generalized abdominal pain, nausea, vomiting, headache, and absence of viral URI symptoms (eg, cough, nasal discharge). However, younger patients with SP may have clinical findings of prolonged nasal drainage and excoriated nares. Examination findings may include swollen and tender anterior cervical lymph nodes; generalized edema and erythema of the posterior pharynx; tonsillar exudates; and palatal petechiae.
Diagnosis
Centor Criteria. The Centor criteria were developed to assist practitioners in identifying patients with potential SP. Criteria for patients older than age 15 years include fever, absence of cough, tonsillar exudates, and tender anterior cervical lymphadenopathy. A modified Centor criteria was later established to include children older than age 5 years, with children between ages 5 and 15 years being the fifth variable in the modified score. In general, patients with a score of 4 or 5 (presence of each variable = 1 point) are most likely to test positive for SP on rapid antigen testing (RAT) or culture.18-20
Swab, Rapid Antigen Testing, and Culture. Swabbing the throat and RAT and/or culture should be performed in most children with suspected SP because the clinical features alone do not reliably discriminate SP from viral pharyngitis. Rapid antigen testing is only specific for group A beta-hemolytic streptococcal species, which is the only streptococcal species that is routinely treated with antibiotics in the setting of acute pharyngitis. It is unlikely for a patient with a score of 0 or 1 to have SP, and several sources suggest neither testing nor treating this cohort, but rather to consider an alternative diagnosis.18-20
Within the population of children and young adolescents, due to a RAT sensitivity of 70% to 90%, a negative result should always be backed-up by a throat culture, and treatment initiated if results of the culture are later found to be positive. As the current generation of RAT tests have a high specificity, a positive RAT does not necessitate a back-up culture, and treatment is indicated without further investigation.19,20
Routine RAT is not recommended in children younger than age 3 years as patients in this age group are at low-risk of developing rheumatic fever. One notable exception for these very young children would be if there are siblings in the home with confirmed SP, in which case, RAT should be considered in the clinical context of SP.21 Adolescents over age 15 years are another cohort with a low likelihood of developing rheumatic fever, though they can develop other poststreptococcal complications, such as glomerulonephritis.
The US Centers for Disease Control and Prevention/American Academy of Family Practitioners (AAFP) guidelines suggest that pharyngitis in older adolescents can be approached in a similar fashion to adults, with empiric therapy for a Centor score of 3 or 4, RAT (without the need for follow-up culture) for Centor score of 2, and neither testing nor treating patients with a score of 0 or 1.19
Treatment and Management
Streptococcal pharyngitis is treated mainly to prevent the poststreptococcal complications of rheumatic fever, though it will not prevent poststreptococcal glomerulonephritis. Treatment of SP also facilitates resolution of symptoms and return to baseline activities.
Antibiotic Therapy. Patients who have a positive RAT or a follow-up throat culture positive for group A streptococcus should be given antibiotics. The gold standard treatment is penicillin V orally for 10 days.
Corticosteroid Therapy. The use of corticosteroids for symptom control of SP in pediatric patients is controversial. Although the Infectious Disease Society of America does not recommend corticosteroid therapy in the treatment of SP, several studies show such therapy (namely dexamethasone), improves pain in children and adolescents diagnosed with SP, but without significant change to the overall disease course.21,23-26
Case 3 Resolution
The patient had a modified Centor criteria score of 4, as well as siblings with similar symptoms. In following current guidelines, the EP performed a RAT and back-up culture. The RAT was negative in the ED, but the back-up culture was subsequently positive, and the child was started on a 10-day course of oral amoxicillin.
Conclusion
When evaluating pediatric patients presenting with ENT signs and symptoms such as ear pain and erythema, fever, sore throat, nasal congestion and discharge, a thorough physical examination and history-taking—including recent illness of any siblings—along with testing when indicated, is essential to guide the diagnosis and determine appropriate treatment and management. In addition to administering antibiotic therapy when such is warranted, the EP should provide appropriate analgesia to manage the patient’s pain and assure relief prior to discharge.
Among all of the causes of ear, nose, and throat (ENT) complaints, acute otitis media (AOM), bacterial sinusitis, and streptococcal pharyngitis (SP) are the most common infections prompting pediatric presentation to the ED. Through a series of case scenarios, along with key questions to help guide the clinician’s work-up, this review covers the proper evaluation and management of pediatric ENT complaints.
Case Scenario 1
A 13-month-old girl presented to the ED with a 1-day history of fever and runny nose. According to her parents, the child had been continually pulling on her ears in apparent discomfort. During history-taking, the parents further informed the emergency physician (EP) that the patient started daycare 4 months earlier and had two elementary school-aged siblings. The patient’s medical history was significant for otitis media, but the parents stated she had not been on antibiotics for over 4 months.
On physical examination, the patient’s vital signs were: blood pressure (BP), 75/50 mm Hg; temperature (T), 101.3°F; slight tachycardia; and normal age-adjusted respiratory rate (RR). Oxygen saturation was 100% on room air. The lungs were clear to auscultation and heart sounds were normal and without murmur. The otolaryngologic examination revealed copious yellow discharge from both nostrils, non-erythematous posterior oropharynx, and erythema to the right tympanic membrane (TM). Questions to Guide the Work-Up: (1) What physical examination findings should be present for accurate diagnosis of otitis media? (2) Will this patient require antibiotics immediately, or is a “wait-and-see” approach indicated? (3) If treatment with antibiotic therapy is warranted, what are the appropriate therapeutic regimen and duration of therapy?
Otitis Media
Acute otitis media is one of the most common presentations in young children. Defined as the rapid onset of signs and symptoms of middle ear inflammation, in conjunction with middle ear effusion (MEE), AOM can develop secondary to a viral or bacterial infection. It is estimated that more than 80% of the pediatric population will experience at least one episode of AOM by age 3 years.1-3
Risk factors for AOM include upper respiratory infection (URI), daycare attendance, siblings, parental smoking, and formula-feeding versus breastfeeding. The patient’s history may include rapid-onset otalgia, fever, irritability, anorexia, and concurrent URI symptoms, as well as other nonspecific symptoms (eg, ear rubbing and/or pulling, crying, changes in behavior and sleep patterns).2-4 In general, otalgia and ear-rubbing in the nonverbal patient seem to have the best predictive value for AOM.3
Signs and Symptoms
A normal TM should be translucent and pearly gray, with visible landmarks of the manubrium of malleus and pars flaccida. A TM that is bulging, cloudy, and immobile is the most consistent finding in AOM, with bulging having a specificity of 97%. Redness of the tympanic membrane is not a useful predictor of AOM as this finding is noted in upward of 30% of pediatric patients on general examination but in <1% of AOM diagnoses in the absence of a bulging TM.
Diagnosis
Pneumatic otoscopy is the gold standard for diagnosing for MEE; however, this examination can be difficult in younger, often uncooperative, patients. A TM that does not perceptibly move with either positive or negative insufflation pressure greatly enhances the diagnostic accuracy for MEE over the use of visible eardrum characteristics alone.2-5
Acute otitis media is a clinical diagnosis and does not require imaging studies or laboratory evaluation unless more serious processes, such as skull fracture, mastoiditis, or intracranial abscess, are being considered.2,3
Treatment and Management
Analgesia. The first step in managing patients with AOM is to provide analgesia. In most cases, acetaminophen in patients over 2 months of age, or ibuprofen in patients over 6 months of age, are adequate choices for managing pain. When either of these analgesics is administered in the clinic/ED setting, patients should be monitored to assure adequate pain relief prior to discharge.
While topical agents such as combination antipyrine-benzocaine suspensions were commonly given in the past to alleviate the pain associated with AOM, there are limited data to support their effectiveness. As such, in July 2015, the US Food and Drug Administration ordered manufacturers to halt production on these unapproved prescription products.3,4,6 There are also no randomized controlled trials (RCTs) to support the use of decongestants or antihistamines for resolution of AOM or otalgia.3,7
Antibiotic Therapy. The most common bacteria associated with AOM are Streptococcus pneumonia, nontypeable Hemophilus influenza, and Moraxella catarrhalis. In 30% of patients, the causative etiology is viral. When the decision is made to treat AOM, high-dose amoxicillin is still considered the first-line treatment, despite ever evolving susceptibilities of bacteria.
When a child is noted to have been treated with amoxicillin within a 30-day period or who has concurrent conjunctivitis, amoxicillin-clavulanate is considered the first-line treatment.2-4,7,8 The current American Academy of Pediatrics (AAP) guidelines recommend 10 days of antibiotic therapy for children younger than age 2 years, and 5 to 7 days for children older than age 2 years who have uncomplicated AOM. Intramuscular (IM) ceftriaxone is an acceptable first-line agent in a child who is unable to tolerate oral medications or who is suffering persistent emesis. Intramuscular ceftriaxone can be given as a single dose of 50 mg/kg, though the patient should be followed closely as studies show that a second dose may be necessary 5 to 7 days later to prevent infection recurrence. The IM dose of ceftriaxone 50 mg/kg can also be given if treatment with other antibiotics fails to resolve the AOM (failure is defined as no improvement in the patient’s condition 48 to 72 hours from treatment). In such cases, ceftriaxone is given in three consecutive doses.3,4,7
Wait-and-See Approach. Studies of patients whose AOM was confirmed via culture (19% were positive for S pneumoniae, 48% for H influenza, and 78% for M catarrhalis) showed bacterial clearance without antibiotic intervention.4 Based on these findings, the 2013 revised AAP evidence-based clinical practice guidelines indicate an initial watching-and-waiting period combined with pain management for patients older than 6 months of age who are diagnosed with unilateral AOM in the absence of severe symptoms (ie, fever is lower than 102.2˚F or patient has severe otalgia).4 A period of observation prior to treatment is also endorsed for children older than age 2 years who exhibit nonsevere symptoms—even if they have bilateral disease.4
Conversely, all patients younger than age 6 months and all children with severe symptoms should be treated with antibiotics at diagnosis.3,4 The wait-and-see approach, recommends an observation period of 24 to 48 hours for children in the lower risk group prior to antibiotic administration. Delayed antibiotic administration can be performed by a physician in an office/ED follow-up or as a safety-net antibiotic prescription (SNAP) sent home with the family on the initial ED encounter.2-4,8,9
Case 1 Resolution
Given this patient’s unilateral and nonsevere symptoms (minor otalgia, fever <102.2°F), age older than 6 months, and no recent antibiotic use), she was treated with oral ibuprofen. At discharge, the parents were given a 10-day SNAP prescription of high-dose amoxicillin (90 mg/kg/d, divided into two daily doses) and instructed to fill the prescription only if the patient’s otalgia did not improve in 1 or 2 days.
Case Scenario 2
A 5-year-old boy was presented for evaluation by his parents, who stated that their son had been sick since he had started kindergarten in the fall. The patient had a 10-day history of cough, thick runny nose, and facial pain, and a 1-day history of new-onset fever and headache. His parents further noted that the patient had been seen by his pediatrician several times over the past week. At each of these visits, the pediatrician had informed them that their son had a virus.
Vital signs on examination were: BP, 100/60 mm Hg; heart rate (HR), 112 beats/min; normal age-adjusted RR; and T, 102.6oF. Oxygen saturation was 100% on room air. The patient did not appear toxic, his lungs were clear on auscultation, and there were no other clinical signs suggestive of meningitis. The otolaryngologic examination revealed bilateral thick mucoid drainage and visible edema and erythema of the nasal turbinates. The patient was noted to have some facial pain in the maxillary area bilaterally.
Questions to Guide the Work-Up: (1) Does the patient have a prolonged URI or pediatric sinusitis, and what differentiates the two conditions? (2) What sinuses are present in a 5-year-old patient? (3) What treatment modalities are available for sinusitis? (4) Is imaging of the sinuses helpful in confirming the diagnosis?
Acute Bacterial Sinusitis
Rhinosinusitis is an inflammation of the mucosal lining of the nasal passages and paranasal sinuses. Most cases occur secondary to a viral URI and resolve spontaneously in 99% of the pediatric population.10,11
Acute bacterial sinusitis (ABS) is an inflammation of the same mucosal lining of the nasal passages secondary to bacterial overgrowth that lasts more than 10 days, with complete resolution by 30 days.12,13 When evaluating a pediatric patient for ABS, it is important to consider the sinus growth and development: If the sinus is not yet formed, it therefore cannot be the location of an ABS.13 The ethmoid and maxillary sinuses are present at birth, aerated within 4 months of life, and are fully developed by age 12 years. The sphenoid sinuses begin development around age 3 years, are aerated by age 7 or 8 years, and are fully developed by age 18 to 20 years. The frontal sinuses begin development around age 8 years and are aerated and fully developed by age 12 to 15 years.10,13,14 While most guidelines focus on children older than age 1 year (due to very small infantile sinuses), ABS does occur in children younger than age 1 year.12,14
Signs and Symptoms
Differentiation between a viral URI/rhinosinusitis and ABS is a challenge and can be based upon severity of symptoms as well as length of illness. Symptoms of ABS are typically present and persistent for more than 10 days, without improvement. Continuing illness and worsening of symptoms are identifying features of ABS given most viral URIs gradually resolve within a 10-day timeframe. Other common symptoms include milky/thick nasal discharge, fever, predominantly nocturnal cough, and headache. Other less common symptoms include facial pain, toothache, malodorous breath, and periorbital edema. On physical examination, erythema and edema of the turbinates, as well as reproducible pain over aerated sinuses, are suggestive of ABS.10-14
Diagnosis
In the acute care setting, diagnosis of ABS should be clinical in nature. Neither imaging nor laboratory work-up is generally required secondary to their poor diagnostic specificity for ABS. The bacteria involved in ABS are similar to those associated with AOM, with S pneumonia, nontypeable H influenza, and M catarrhalis being the predominant organisms.10-15
Treatment and Management
Treatment of ABS is generally recommended once the diagnosis is made, though this is based largely on expert opinion as there are limited RCTs available.13 However, available studies do show a more rapid improvement in children on antibiotic therapy than those on placebo.15,16
Antibiotic Therapy. Amoxicillin remains the antimicrobial agent of choice for first-line treatment of uncomplicated ABS forsituations in which antimicrobial resistance is not suspected. In communities with a high prevalence of nonsusceptible S pneumoniae (>10%, including intermediate- and high-level resistance), treatment may be initiated at 80 to 90 mg/kg/d in two divided doses, with a maximum of 2 g per dose.
Patients presenting with moderate to severe illness, as well as those who are younger than 2 years, attend childcare, or have recently been treated with an antimicrobial, may receive high-dose amoxicillin-clavulanate as initial therapy given the elevated beta-lactamase production of the common bacteria that cause ABS.
Second-line alternatives include azithromycin, cefdinir, and sulfamethoxizole-trimethoprim (Table 1). There are data to suggest higher rates of decreased susceptibility of S pneumonia and H influenza to third-generation cephalosporins, and the addition of clindamycin may be warranted when utilizing those medications. Treatment is recommended for 10 to 14 days, though improvement should be noted within 1 to 3 days.10-12,14-17
Adjuvant Therapy. Additional therapies include nasal irrigation, decongestants, antihistamines, and intranasal steroids; however, there are only anecdotal reports of their efficacy in providing symptom relief. Therefore, there are insufficient evidence-based data to support or refute the role of these adjuvant therapies in treating pediatric patients with ABS.9,13
Case 2 Resolution
The prolonged duration and severity of symptoms (high fever and headache) and the gradual worsening of the clinical course (ie, late-onset fever) in this patient all suggest ABS rather than a simple prolonged URI. The physical examination findings of inflamed turbinates and facial pain further increase the specificity for ABS. The patient was started on oral amoxicillin-clavulanate with planned treatment for 14 days. At discharge, his parents were instructed to follow-up with the patient’s pediatrician in 3 days to ensure a degree of clinical resolution.
Case Scenario 3
A 4-year-old boy was presented by his parents for evaluation of a 2-day history of a persistent and unimproved sore throat. The patient’s mother indicated that the child’s oral T upon returning home earlier from preschool was 101.2oF. She further noted that her 17-month-old daughter and 8-year-old son also experienced similar symptoms which had self-resolved. Triage vital signs were: T, 100.8oF, orally; BP, HR, and RR were all within normal limits. Oxygen saturation was 100% on room air.
On physical examination, the child was noted to have anterior cervical lymph nodes bilaterally and an erythematous oropharynx with exudate noted on both tonsils. There were no cutaneous abnormalities, nasal edema, erythema, or drainage. Based on the clinical examination, the EP was suspicious for SP.
Questions to Guide the Work-Up: (1) Is SP diagnosed based on clinical findings alone in this patient’s age group? (2) At what age in the pediatric population is it appropriate to perform a rapid streptococcal antigen test? (3) Are there medications other than antibiotics that are beneficial in treating symptomatic SP?
Streptococcal Pharyngitis
Streptococcal pharyngitis is a clinical condition caused by group A beta-hemolytic S pyogens. This bacterium is responsible for multiple conditions, including pharyngitis, skin infections, poststreptococcal glomerulonephritis, and rheumatic fever, as well as invasive syndromes. (This case focuses solely on SP).
Pharyngitis can occur secondary to a viral or bacterial infection, and SP is the most common cause of pediatric bacterial pharyngitis. It is estimated that children aged 5 to 15 years are more commonly diagnosed with SP, although approximately 24% of children younger than age 5 years with pharyngitis symptoms will be ultimately diagnosed with SP.
Signs and Symptoms
Typical symptoms include fever, pharyngitis, generalized abdominal pain, nausea, vomiting, headache, and absence of viral URI symptoms (eg, cough, nasal discharge). However, younger patients with SP may have clinical findings of prolonged nasal drainage and excoriated nares. Examination findings may include swollen and tender anterior cervical lymph nodes; generalized edema and erythema of the posterior pharynx; tonsillar exudates; and palatal petechiae.
Diagnosis
Centor Criteria. The Centor criteria were developed to assist practitioners in identifying patients with potential SP. Criteria for patients older than age 15 years include fever, absence of cough, tonsillar exudates, and tender anterior cervical lymphadenopathy. A modified Centor criteria was later established to include children older than age 5 years, with children between ages 5 and 15 years being the fifth variable in the modified score. In general, patients with a score of 4 or 5 (presence of each variable = 1 point) are most likely to test positive for SP on rapid antigen testing (RAT) or culture.18-20
Swab, Rapid Antigen Testing, and Culture. Swabbing the throat and RAT and/or culture should be performed in most children with suspected SP because the clinical features alone do not reliably discriminate SP from viral pharyngitis. Rapid antigen testing is only specific for group A beta-hemolytic streptococcal species, which is the only streptococcal species that is routinely treated with antibiotics in the setting of acute pharyngitis. It is unlikely for a patient with a score of 0 or 1 to have SP, and several sources suggest neither testing nor treating this cohort, but rather to consider an alternative diagnosis.18-20
Within the population of children and young adolescents, due to a RAT sensitivity of 70% to 90%, a negative result should always be backed-up by a throat culture, and treatment initiated if results of the culture are later found to be positive. As the current generation of RAT tests have a high specificity, a positive RAT does not necessitate a back-up culture, and treatment is indicated without further investigation.19,20
Routine RAT is not recommended in children younger than age 3 years as patients in this age group are at low-risk of developing rheumatic fever. One notable exception for these very young children would be if there are siblings in the home with confirmed SP, in which case, RAT should be considered in the clinical context of SP.21 Adolescents over age 15 years are another cohort with a low likelihood of developing rheumatic fever, though they can develop other poststreptococcal complications, such as glomerulonephritis.
The US Centers for Disease Control and Prevention/American Academy of Family Practitioners (AAFP) guidelines suggest that pharyngitis in older adolescents can be approached in a similar fashion to adults, with empiric therapy for a Centor score of 3 or 4, RAT (without the need for follow-up culture) for Centor score of 2, and neither testing nor treating patients with a score of 0 or 1.19
Treatment and Management
Streptococcal pharyngitis is treated mainly to prevent the poststreptococcal complications of rheumatic fever, though it will not prevent poststreptococcal glomerulonephritis. Treatment of SP also facilitates resolution of symptoms and return to baseline activities.
Antibiotic Therapy. Patients who have a positive RAT or a follow-up throat culture positive for group A streptococcus should be given antibiotics. The gold standard treatment is penicillin V orally for 10 days.
Corticosteroid Therapy. The use of corticosteroids for symptom control of SP in pediatric patients is controversial. Although the Infectious Disease Society of America does not recommend corticosteroid therapy in the treatment of SP, several studies show such therapy (namely dexamethasone), improves pain in children and adolescents diagnosed with SP, but without significant change to the overall disease course.21,23-26
Case 3 Resolution
The patient had a modified Centor criteria score of 4, as well as siblings with similar symptoms. In following current guidelines, the EP performed a RAT and back-up culture. The RAT was negative in the ED, but the back-up culture was subsequently positive, and the child was started on a 10-day course of oral amoxicillin.
Conclusion
When evaluating pediatric patients presenting with ENT signs and symptoms such as ear pain and erythema, fever, sore throat, nasal congestion and discharge, a thorough physical examination and history-taking—including recent illness of any siblings—along with testing when indicated, is essential to guide the diagnosis and determine appropriate treatment and management. In addition to administering antibiotic therapy when such is warranted, the EP should provide appropriate analgesia to manage the patient’s pain and assure relief prior to discharge.
1. Rosenfeld RM, Shin JJ, Schwartz SR, et al. Clinical practice guideline: otitis media with effusion (Update). Otolaryngol Head Neck Surg. 2016;154(1 Suppl):S1-S41. doi:10.1177/0194599815623467.
2. Acute Otitis Media Guideline Team, Cincinnati Children’s Hospital Medical Center. Evidence-based care guideline for medical management of acute otitis media in children 2 months to 13 years of age. http://f.i-md.com/medinfo/material/4f4/4eb132ba44ae4ffe12a814f4/4eb132d744ae4ffe12a814f7.pdf. August 2006. Accessed December 29, 2016.
3. Nesbit CE, Powers MC. An evidence-based approach to managing acute otitis media. Pediatr Emerg Med Pract. 2013;10(4):1-26; quiz 26-27.
4. Lieberthal AS, Carroll AE, Chonmaitree T, et al. The diagnosis and management of acute otitis media. Pediatrics. 2013;131(3):e964-e999. doi:10.1542/peds.2012-3488.
5. American Academy of Family Physicians; American Academy of Otolaryngology-Head and Neck Surgery; American Academy of Pediatrics Subcommittee on Otitis Media With Effusion. Otitis media with effusion. Pediatrics. 2004;113(5):1412-1429.
6. US Food and Drug Administration Web site. FDA: Use only approved prescription ear drops. http://www.fda.gov/ForConsumers/ConsumerUpdates/-ucm453087.htm. Updated July 10, 2015. Accessed December 15, 2016.
7. Sack F. An evidence based approach to the management of uncomplicated acute otitis media in children. Int Pediatrics. 2005;20(1):44-46.
8. Johnson NC, Holger JS. Pediatric acute otitis media: the case for delayed antibiotic treatment. J Emerg Med. 2007;32(3):279-284. doi:10.1016/j.jemermed.2006.07.029.
9. Spiro DM, Tay KY, Arnold DH, Dziura JD, Baker MD, Shapiro ED. Wait-and-see prescription for the treatment of acute otitis media: a randomized controlled trial. JAMA. 2006;296(10):1235-1241. doi:10.1001/jama.296.10.1235.
10. Brook I. Management of acute rhinosinusitis in pediatric patients. Pediatr Emerg Med Pract. 2012;9(5):1-24.
11. Ferdman RM, Linzer JF Jr. The runny nose in the emergency department: rhinitis and sinusitis. Clin Pediatr Emerg Med. 2007;8(2):123-130.
12. Acute Bacterial Sinusitis Guideline Team, Cincinnati Children’s Hospital Medical Center: Evidence-based care guideline for medical management of acute bacterial sinusitis in children 1 through 18 years of age. http://www.antibioticos.msssi.gob.es/PDF/sinusitisguideline.pdf. July 7, 2006. Accessed December 29, 2016
13. Holt KR, Murdoch Cuenca M, Cuenca PJ, Johnston GM. acute pediatric sinusitis and “the 10-day rule.” Pediatr Emerg Med Pract. 2006;3(2):1-16.
14. American Academy of Pediatrics. Subcommittee on Management of Sinusitis and Committee on Quality Improvement. Clinical practice guideline: management of sinusitis. Pediatrics. 2001;108(3):798-808.
15. Wald ER, Nash D, Eickhoff J. Effectiveness of amoxicillin/clavulanate potassium in the treatment of acute bacterial sinusitis in children. Pediatrics. 2009;124(1):9-15. doi:10.1542/peds.2008-2902.
16. Arroll B, Kenealy T. Are antibiotics effective for acute purulent rhinitis? Systematic review and meta-analysis of placebo controlled randomised trials. BMJ. 2006;333(7562):279. doi:10.1136/bmj.38891.681215.AE.
17. McQuillan L, Crane LA, Kempe A. Diagnosis and management of acute sinusitis by pediatricians. Pediatrics. 2009;123(2):e193-e198.
18. Singer JI, Fontanette R. Recognizable and suspected group A beta-hemolytic streptococcal syndromes. Pediatr Emerg Med Rep. 2010;15(11):129-144.
19. Weglowski J. An evidence-based approach to the evaluation and treatment of pharyngitis in children. Pediatr Emerg Med Pract. 2011;8(12):1-28.
20. Gerber MA, Baltimore RS, Eaton CB, et al. Prevention of rheumatic fever and diagnosis and treatment of acute Streptococcal pharyngitis: a scientific statement from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, the Interdisciplinary Council on Functional Genomics and Translational Biology, and the Interdisciplinary Council on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation. 2009;119(11):1541-1551. doi:10.1161/CIRCULATIONAHA.109.191959.
21. Shulman ST, Bisno AL, Clegg HW, et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis. 2012;55(10):1279-1282. doi:10.1093/cid/cis847.
22. Clegg HW, Ryan AG, Dallas SD, et al. Treatment of streptococcal pharyngitis with once-daily compared with twice-daily amoxicillin: a noninferiority trial. Pediatr Infect Dis J. 2006;25(9):761-767. doi:10.1097/01.inf.0000235678.46805.92.
23. Bulloch B, Kabani A, Tenenbein M. Oral dexamethasone for the treatment of pain in children with acute pharyngitis: a randomized, double-blind, placebo-controlled trial. Ann Emerg Med. 2003;41(5):601-608. doi:10.1067/mem.2003.136.
24. Niland ML, Bonsu BK, Nuss KE, Goodman DG. A pilot study of 1 versus 3 days of dexamethasone as add-on therapy in children with streptococcal pharyngitis. Pediatr Infect Dis J. 2006;25(6):477-481. doi:10.1097/01.inf.0000219469.95772.3f.
25. Wei JL, Kasperbauer JL, Weaver AL, Boggust AJ. Efficacy of single-dose dexamethasone as adjuvant therapy for acute pharyngitis. Laryngoscope. 2002;112(1):87-93. doi:10.1097/00005537-200201000-00016.
26. Hayward G, Thompson M, Heneghan C, Perera R, Del Mar C, Glasziou P. Corticosteroids for pain relief in sore throat: systematic review and meta-analysis. BMJ. 2009;339:b2976. doi:10.1136/bmj.b2976.
1. Rosenfeld RM, Shin JJ, Schwartz SR, et al. Clinical practice guideline: otitis media with effusion (Update). Otolaryngol Head Neck Surg. 2016;154(1 Suppl):S1-S41. doi:10.1177/0194599815623467.
2. Acute Otitis Media Guideline Team, Cincinnati Children’s Hospital Medical Center. Evidence-based care guideline for medical management of acute otitis media in children 2 months to 13 years of age. http://f.i-md.com/medinfo/material/4f4/4eb132ba44ae4ffe12a814f4/4eb132d744ae4ffe12a814f7.pdf. August 2006. Accessed December 29, 2016.
3. Nesbit CE, Powers MC. An evidence-based approach to managing acute otitis media. Pediatr Emerg Med Pract. 2013;10(4):1-26; quiz 26-27.
4. Lieberthal AS, Carroll AE, Chonmaitree T, et al. The diagnosis and management of acute otitis media. Pediatrics. 2013;131(3):e964-e999. doi:10.1542/peds.2012-3488.
5. American Academy of Family Physicians; American Academy of Otolaryngology-Head and Neck Surgery; American Academy of Pediatrics Subcommittee on Otitis Media With Effusion. Otitis media with effusion. Pediatrics. 2004;113(5):1412-1429.
6. US Food and Drug Administration Web site. FDA: Use only approved prescription ear drops. http://www.fda.gov/ForConsumers/ConsumerUpdates/-ucm453087.htm. Updated July 10, 2015. Accessed December 15, 2016.
7. Sack F. An evidence based approach to the management of uncomplicated acute otitis media in children. Int Pediatrics. 2005;20(1):44-46.
8. Johnson NC, Holger JS. Pediatric acute otitis media: the case for delayed antibiotic treatment. J Emerg Med. 2007;32(3):279-284. doi:10.1016/j.jemermed.2006.07.029.
9. Spiro DM, Tay KY, Arnold DH, Dziura JD, Baker MD, Shapiro ED. Wait-and-see prescription for the treatment of acute otitis media: a randomized controlled trial. JAMA. 2006;296(10):1235-1241. doi:10.1001/jama.296.10.1235.
10. Brook I. Management of acute rhinosinusitis in pediatric patients. Pediatr Emerg Med Pract. 2012;9(5):1-24.
11. Ferdman RM, Linzer JF Jr. The runny nose in the emergency department: rhinitis and sinusitis. Clin Pediatr Emerg Med. 2007;8(2):123-130.
12. Acute Bacterial Sinusitis Guideline Team, Cincinnati Children’s Hospital Medical Center: Evidence-based care guideline for medical management of acute bacterial sinusitis in children 1 through 18 years of age. http://www.antibioticos.msssi.gob.es/PDF/sinusitisguideline.pdf. July 7, 2006. Accessed December 29, 2016
13. Holt KR, Murdoch Cuenca M, Cuenca PJ, Johnston GM. acute pediatric sinusitis and “the 10-day rule.” Pediatr Emerg Med Pract. 2006;3(2):1-16.
14. American Academy of Pediatrics. Subcommittee on Management of Sinusitis and Committee on Quality Improvement. Clinical practice guideline: management of sinusitis. Pediatrics. 2001;108(3):798-808.
15. Wald ER, Nash D, Eickhoff J. Effectiveness of amoxicillin/clavulanate potassium in the treatment of acute bacterial sinusitis in children. Pediatrics. 2009;124(1):9-15. doi:10.1542/peds.2008-2902.
16. Arroll B, Kenealy T. Are antibiotics effective for acute purulent rhinitis? Systematic review and meta-analysis of placebo controlled randomised trials. BMJ. 2006;333(7562):279. doi:10.1136/bmj.38891.681215.AE.
17. McQuillan L, Crane LA, Kempe A. Diagnosis and management of acute sinusitis by pediatricians. Pediatrics. 2009;123(2):e193-e198.
18. Singer JI, Fontanette R. Recognizable and suspected group A beta-hemolytic streptococcal syndromes. Pediatr Emerg Med Rep. 2010;15(11):129-144.
19. Weglowski J. An evidence-based approach to the evaluation and treatment of pharyngitis in children. Pediatr Emerg Med Pract. 2011;8(12):1-28.
20. Gerber MA, Baltimore RS, Eaton CB, et al. Prevention of rheumatic fever and diagnosis and treatment of acute Streptococcal pharyngitis: a scientific statement from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, the Interdisciplinary Council on Functional Genomics and Translational Biology, and the Interdisciplinary Council on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation. 2009;119(11):1541-1551. doi:10.1161/CIRCULATIONAHA.109.191959.
21. Shulman ST, Bisno AL, Clegg HW, et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis. 2012;55(10):1279-1282. doi:10.1093/cid/cis847.
22. Clegg HW, Ryan AG, Dallas SD, et al. Treatment of streptococcal pharyngitis with once-daily compared with twice-daily amoxicillin: a noninferiority trial. Pediatr Infect Dis J. 2006;25(9):761-767. doi:10.1097/01.inf.0000235678.46805.92.
23. Bulloch B, Kabani A, Tenenbein M. Oral dexamethasone for the treatment of pain in children with acute pharyngitis: a randomized, double-blind, placebo-controlled trial. Ann Emerg Med. 2003;41(5):601-608. doi:10.1067/mem.2003.136.
24. Niland ML, Bonsu BK, Nuss KE, Goodman DG. A pilot study of 1 versus 3 days of dexamethasone as add-on therapy in children with streptococcal pharyngitis. Pediatr Infect Dis J. 2006;25(6):477-481. doi:10.1097/01.inf.0000219469.95772.3f.
25. Wei JL, Kasperbauer JL, Weaver AL, Boggust AJ. Efficacy of single-dose dexamethasone as adjuvant therapy for acute pharyngitis. Laryngoscope. 2002;112(1):87-93. doi:10.1097/00005537-200201000-00016.
26. Hayward G, Thompson M, Heneghan C, Perera R, Del Mar C, Glasziou P. Corticosteroids for pain relief in sore throat: systematic review and meta-analysis. BMJ. 2009;339:b2976. doi:10.1136/bmj.b2976.
First-trimester blood glucose predicts congenital heart disease risk
NEW ORLEANS – A single, random, first-trimester maternal plasma glucose measurement is superior to an oral glucose tolerance test later in pregnancy as a predictor of congenital heart disease in newborns, Emmi Helle, MD, reported at the American Heart Association scientific sessions.
This finding from a large retrospective study, if confirmed in a prospective data set, is likely to be practice changing. At present, a 1-hour oral glucose tolerance test in the second or third trimester is considered the best means of identifying pregnant women who ought to undergo fetal echocardiography for prenatal diagnosis of congenital heart disease, noted Dr. Helle of Stanford (Calif.) University.
An elevated random plasma glucose value in the first trimester was broadly predictive of increased risk for a variety of congenital heart anomalies, not just, for example, cyanotic conditions.
Fetal heart development is completed during the first trimester, Dr. Helle observed.
Her study received a warm reception. Michael A. Portman, MD, singled it out in his final-day wrap-up of the meeting’s highlights in the field of congenital heart disease.
Several studies have demonstrated that prenatal diagnosis of congenital heart disease results in improved surgical outcomes in newborns. The question is, how to get the right women – those at increased risk – to diagnostic fetal echocardiography. Guidelines suggest but don’t mandate on the basis of weak evidence that an oral glucose tolerance test performed in the second or early third trimester may be a useful means of screening mothers for fetal imaging. Dr. Helle’s study points to a better way.
“Hopefully we can change our guidelines and make them more scientific for identification of mothers who should undergo fetal echocardiography,” said Dr. Portman, professor of pediatrics at the University of Washington, Seattle, and director of pediatric cardiovascular research at Seattle Children’s Hospital.
Dr. Helle and Dr. Portman reported having no relevant financial interests.
NEW ORLEANS – A single, random, first-trimester maternal plasma glucose measurement is superior to an oral glucose tolerance test later in pregnancy as a predictor of congenital heart disease in newborns, Emmi Helle, MD, reported at the American Heart Association scientific sessions.
This finding from a large retrospective study, if confirmed in a prospective data set, is likely to be practice changing. At present, a 1-hour oral glucose tolerance test in the second or third trimester is considered the best means of identifying pregnant women who ought to undergo fetal echocardiography for prenatal diagnosis of congenital heart disease, noted Dr. Helle of Stanford (Calif.) University.
An elevated random plasma glucose value in the first trimester was broadly predictive of increased risk for a variety of congenital heart anomalies, not just, for example, cyanotic conditions.
Fetal heart development is completed during the first trimester, Dr. Helle observed.
Her study received a warm reception. Michael A. Portman, MD, singled it out in his final-day wrap-up of the meeting’s highlights in the field of congenital heart disease.
Several studies have demonstrated that prenatal diagnosis of congenital heart disease results in improved surgical outcomes in newborns. The question is, how to get the right women – those at increased risk – to diagnostic fetal echocardiography. Guidelines suggest but don’t mandate on the basis of weak evidence that an oral glucose tolerance test performed in the second or early third trimester may be a useful means of screening mothers for fetal imaging. Dr. Helle’s study points to a better way.
“Hopefully we can change our guidelines and make them more scientific for identification of mothers who should undergo fetal echocardiography,” said Dr. Portman, professor of pediatrics at the University of Washington, Seattle, and director of pediatric cardiovascular research at Seattle Children’s Hospital.
Dr. Helle and Dr. Portman reported having no relevant financial interests.
NEW ORLEANS – A single, random, first-trimester maternal plasma glucose measurement is superior to an oral glucose tolerance test later in pregnancy as a predictor of congenital heart disease in newborns, Emmi Helle, MD, reported at the American Heart Association scientific sessions.
This finding from a large retrospective study, if confirmed in a prospective data set, is likely to be practice changing. At present, a 1-hour oral glucose tolerance test in the second or third trimester is considered the best means of identifying pregnant women who ought to undergo fetal echocardiography for prenatal diagnosis of congenital heart disease, noted Dr. Helle of Stanford (Calif.) University.
An elevated random plasma glucose value in the first trimester was broadly predictive of increased risk for a variety of congenital heart anomalies, not just, for example, cyanotic conditions.
Fetal heart development is completed during the first trimester, Dr. Helle observed.
Her study received a warm reception. Michael A. Portman, MD, singled it out in his final-day wrap-up of the meeting’s highlights in the field of congenital heart disease.
Several studies have demonstrated that prenatal diagnosis of congenital heart disease results in improved surgical outcomes in newborns. The question is, how to get the right women – those at increased risk – to diagnostic fetal echocardiography. Guidelines suggest but don’t mandate on the basis of weak evidence that an oral glucose tolerance test performed in the second or early third trimester may be a useful means of screening mothers for fetal imaging. Dr. Helle’s study points to a better way.
“Hopefully we can change our guidelines and make them more scientific for identification of mothers who should undergo fetal echocardiography,” said Dr. Portman, professor of pediatrics at the University of Washington, Seattle, and director of pediatric cardiovascular research at Seattle Children’s Hospital.
Dr. Helle and Dr. Portman reported having no relevant financial interests.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: For every 10-mg/dL increase in maternal plasma glucose on a random first-trimester measurement, the risk of giving birth to a baby with congenital heart disease rose by 8%.
Data source: A retrospective study of 19,197 pregnancies, 811 of which resulted in congenital heart disease in the offspring.
Disclosures: The presenter reported having no financial conflicts of interest regarding the study.
Fewer people having problems with medical bills
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
Sorafenib survival benefit in HCC depends on etiology
The impact of sorafenib in treating patients with advanced hepatocellular cancer may be dependent on their hepatitis status, according to a meta-analysis of patient-level data from three large prospective clinical trials using sorafenib as the control treatment.
The analysis indicated that sorafenib had a favorable effect on overall survival for hepatocellular carcinoma patients who were positive for hepatitis C (HCV), but negative for hepatitis B (HBV) only.
For HCV-positive/HBV-negative patients with advanced unresectable hepatocellular carcinoma (aHCC) who received sorafenib, median unadjusted survival was 12.6 months, compared to 10.2 months for patients who received other treatments, yielding a log hazard ratio of –0.27 (95% confidence interval [CI] –0.46 to –0.06).
Though the study did not shed light on the reasons for this difference in overall survival (OS), the results were seen consistently in data from all trials, and sorafenib did not confer any significant survival benefit for individuals who were not HCV positive and HBV negative. “Irrespective of the mechanism, our data suggest that in future trials in aHCC, particularly where sorafenib is the control arm, there should be stratification according to etiology,” wrote Richard Jackson, MSc, and his coauthors (J Clin Oncol. 2017 Jan 3:JCO2016695197 [Epub ahead of print]).
Mr. Jackson, a medical statistician with the Institute of Translational Medicine at the University of Liverpool, England, and his collaborators examined data from 3,256 patients with aHCC. Of these, 1,643 (50%) received sorafenib. The remainder was aggregated into an “other treatment” group, pooling data from patients who received brivanib, sunitinib, and linifanib.
All patients were also divided into four etiologic subgroups: HBV-negative/HCV-negative; HBV-negative/HCV-positive; HBV-positive/HCV-negative; and HBV-positive/HCV-positive. Mr. Jackson and his colleagues then examined the pooled data to see what effect treatment type (sorafenib versus pooled comparator treatments) had on overall survival for each etiologic subgroup. They found no statistically significant survival benefit for sorafenib in the three etiologic subgroups that were not HCV positive/HBV negative, though the data showed a statistically insignificant trend favoring sorafenib. Race, when examined as a potential confounding factor, was not associated with a difference in OS benefit for sorafenib.
The sponsors of three large clinical trials of treatments for aHCC that used sorafenib as the control arm provided deidentified patient-level data to the study’s authors, who then undertook an individual patient data (IPD) meta-analysis. “IPD meta-analyses have a major advantage over aggregate meta-analyses in that they ensure consistent analytic techniques and allow for detailed inspection of interaction of subgroup effects that are not available in published evidence,” wrote Mr. Jackson and his coauthors.
In discussing their findings, the researchers called for more of the data-sharing that allowed their IPD meta-analysis, citing a proposal on the topic from the International Committee of Medical Journal Editors. “[O]ur study showed how the benefits of access to completed trial data are not necessarily confined to reanalysis of the original hypothesis tested by the trial. Here, by a meta-analysis, we arrive at an answer to a question that was not considered when the trials were conceived and could not have been answered by any of the trials individually,” they wrote.
The study data were provided by Bristol-Myers Squibb, Pfizer, and AbbVie from studies they sponsored. Mr. Jackson reported no conflicts of interest.
[email protected]
On Twitter @karioakes
The impact of sorafenib in treating patients with advanced hepatocellular cancer may be dependent on their hepatitis status, according to a meta-analysis of patient-level data from three large prospective clinical trials using sorafenib as the control treatment.
The analysis indicated that sorafenib had a favorable effect on overall survival for hepatocellular carcinoma patients who were positive for hepatitis C (HCV), but negative for hepatitis B (HBV) only.
For HCV-positive/HBV-negative patients with advanced unresectable hepatocellular carcinoma (aHCC) who received sorafenib, median unadjusted survival was 12.6 months, compared to 10.2 months for patients who received other treatments, yielding a log hazard ratio of –0.27 (95% confidence interval [CI] –0.46 to –0.06).
Though the study did not shed light on the reasons for this difference in overall survival (OS), the results were seen consistently in data from all trials, and sorafenib did not confer any significant survival benefit for individuals who were not HCV positive and HBV negative. “Irrespective of the mechanism, our data suggest that in future trials in aHCC, particularly where sorafenib is the control arm, there should be stratification according to etiology,” wrote Richard Jackson, MSc, and his coauthors (J Clin Oncol. 2017 Jan 3:JCO2016695197 [Epub ahead of print]).
Mr. Jackson, a medical statistician with the Institute of Translational Medicine at the University of Liverpool, England, and his collaborators examined data from 3,256 patients with aHCC. Of these, 1,643 (50%) received sorafenib. The remainder was aggregated into an “other treatment” group, pooling data from patients who received brivanib, sunitinib, and linifanib.
All patients were also divided into four etiologic subgroups: HBV-negative/HCV-negative; HBV-negative/HCV-positive; HBV-positive/HCV-negative; and HBV-positive/HCV-positive. Mr. Jackson and his colleagues then examined the pooled data to see what effect treatment type (sorafenib versus pooled comparator treatments) had on overall survival for each etiologic subgroup. They found no statistically significant survival benefit for sorafenib in the three etiologic subgroups that were not HCV positive/HBV negative, though the data showed a statistically insignificant trend favoring sorafenib. Race, when examined as a potential confounding factor, was not associated with a difference in OS benefit for sorafenib.
The sponsors of three large clinical trials of treatments for aHCC that used sorafenib as the control arm provided deidentified patient-level data to the study’s authors, who then undertook an individual patient data (IPD) meta-analysis. “IPD meta-analyses have a major advantage over aggregate meta-analyses in that they ensure consistent analytic techniques and allow for detailed inspection of interaction of subgroup effects that are not available in published evidence,” wrote Mr. Jackson and his coauthors.
In discussing their findings, the researchers called for more of the data-sharing that allowed their IPD meta-analysis, citing a proposal on the topic from the International Committee of Medical Journal Editors. “[O]ur study showed how the benefits of access to completed trial data are not necessarily confined to reanalysis of the original hypothesis tested by the trial. Here, by a meta-analysis, we arrive at an answer to a question that was not considered when the trials were conceived and could not have been answered by any of the trials individually,” they wrote.
The study data were provided by Bristol-Myers Squibb, Pfizer, and AbbVie from studies they sponsored. Mr. Jackson reported no conflicts of interest.
[email protected]
On Twitter @karioakes
The impact of sorafenib in treating patients with advanced hepatocellular cancer may be dependent on their hepatitis status, according to a meta-analysis of patient-level data from three large prospective clinical trials using sorafenib as the control treatment.
The analysis indicated that sorafenib had a favorable effect on overall survival for hepatocellular carcinoma patients who were positive for hepatitis C (HCV), but negative for hepatitis B (HBV) only.
For HCV-positive/HBV-negative patients with advanced unresectable hepatocellular carcinoma (aHCC) who received sorafenib, median unadjusted survival was 12.6 months, compared to 10.2 months for patients who received other treatments, yielding a log hazard ratio of –0.27 (95% confidence interval [CI] –0.46 to –0.06).
Though the study did not shed light on the reasons for this difference in overall survival (OS), the results were seen consistently in data from all trials, and sorafenib did not confer any significant survival benefit for individuals who were not HCV positive and HBV negative. “Irrespective of the mechanism, our data suggest that in future trials in aHCC, particularly where sorafenib is the control arm, there should be stratification according to etiology,” wrote Richard Jackson, MSc, and his coauthors (J Clin Oncol. 2017 Jan 3:JCO2016695197 [Epub ahead of print]).
Mr. Jackson, a medical statistician with the Institute of Translational Medicine at the University of Liverpool, England, and his collaborators examined data from 3,256 patients with aHCC. Of these, 1,643 (50%) received sorafenib. The remainder was aggregated into an “other treatment” group, pooling data from patients who received brivanib, sunitinib, and linifanib.
All patients were also divided into four etiologic subgroups: HBV-negative/HCV-negative; HBV-negative/HCV-positive; HBV-positive/HCV-negative; and HBV-positive/HCV-positive. Mr. Jackson and his colleagues then examined the pooled data to see what effect treatment type (sorafenib versus pooled comparator treatments) had on overall survival for each etiologic subgroup. They found no statistically significant survival benefit for sorafenib in the three etiologic subgroups that were not HCV positive/HBV negative, though the data showed a statistically insignificant trend favoring sorafenib. Race, when examined as a potential confounding factor, was not associated with a difference in OS benefit for sorafenib.
The sponsors of three large clinical trials of treatments for aHCC that used sorafenib as the control arm provided deidentified patient-level data to the study’s authors, who then undertook an individual patient data (IPD) meta-analysis. “IPD meta-analyses have a major advantage over aggregate meta-analyses in that they ensure consistent analytic techniques and allow for detailed inspection of interaction of subgroup effects that are not available in published evidence,” wrote Mr. Jackson and his coauthors.
In discussing their findings, the researchers called for more of the data-sharing that allowed their IPD meta-analysis, citing a proposal on the topic from the International Committee of Medical Journal Editors. “[O]ur study showed how the benefits of access to completed trial data are not necessarily confined to reanalysis of the original hypothesis tested by the trial. Here, by a meta-analysis, we arrive at an answer to a question that was not considered when the trials were conceived and could not have been answered by any of the trials individually,” they wrote.
The study data were provided by Bristol-Myers Squibb, Pfizer, and AbbVie from studies they sponsored. Mr. Jackson reported no conflicts of interest.
[email protected]
On Twitter @karioakes
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: Median unadjusted survival time for HCV-positive/HBV-negative patients was 12.6 months on sorafenib, compared to 10.2 months for these patients who received other treatments.
Data source: Individual patient data meta-analysis of three randomized phase III clinical trials, examining data from 3,256 patients.
Disclosures: The study data were provided by Bristol-Myers Squibb, Pfizer, and AbbVie. Mr. Jackson reported no conflicts of interest.
Gastric cancer yields to growth hormone antagonist in lab
It sounds counterintuitive, but targeting a neuropeptide hormone produced in the hypothalamus may be an effective strategy for treating gastric cancer, the second most common cause of cancer deaths worldwide, investigators from China and the United States contend.
Growth hormone–releasing hormone (GHRH) and its receptor (GHRH-R) are found primarily in the anterior pituitary gland, but are also present in gastric cancers, other solid tumors, and lymphomas. Increased levels of GHRH-R in tumor samples from patients with gastric cancer are associated with poor outcomes, noted Andrew V. Schally, PhD, MD, DSc, of the University of Miami, and his colleagues at the Shantou (China) University Medical College.
Furthermore, an experimental peptide drug labeled MIA-602 that targets GHRH-R inhibited the growth of gastric cancer cell lines and human tumor xenografts in mice, the investigators reported in the journal PNAS.
“The GHRH receptor is both a biomarker that can confirm prognosis and a therapeutic target,” Dr. Schally said in a statement.
Elevated GHRH-R expression in tumors
GHRH-R antagonists such as MIA-602 work through downregulation of the p21-activated kinase 1 (PAK1)–mediated signal transducer and activator of transcription 3 (STAT3)/nuclear factor–kappaB (NF-kappaB) inflammatory pathway. This pathway is involved in the interplay between inflammatory processes and intracellular signaling thought to be the cause of gastric cancer tumorigenesis and progression, the investigators explained.
They first looked for GHRH-R expression in gastric cancer samples from 106 patients, using immunohistochemistry staining of primary tumors and adjacent normal tissues. They found that gastric cancer tissues “exhibited robust expression of GHRH-R, compared with normal tissues.”
In 50 samples, GHRH-R was determined to be overexpressed, and this overexpression was significantly associated with both greater tumor size (P = .031) and high pathologic tumor stage (P = .001). Increasing expression of GHRH-R was also significantly associated with worse overall survival (P less than .001).
They confirmed these findings in samples from a multinational cohort of patients, which again showed that the highest levels of GHRH-R expression were associated with poor overall survival (P less than .001).
The authors also looked at messenger RNA expression and gene copy number in 65 gastric cancer samples and 19 adjacent normal tissue samples, and found that GHRH-R mRNA was significantly higher in tumor tissues than normal control tissues (P less than .001).
MAI-602 in vitro and in vivo
To see whether MAI-602 could inhibit the growth of gastric cancer cells, the investigators tried it at various doses in three human gastric cancer cell lines, and found that it inhibited cells in a dose-dependent fashion, compared with vehicle used as a control (P less than .001).
In addition, the experimental agent “exhibited remarkable inhibitory effects on tumor growth in vivo” in mice with human tumor xenografts (P less than .001).
Finally, they showed that the cancer suppression effects of MAI-602 work through inhibition of STAT3/NF-kappaB inflammatory signaling. In vitro and in vivo, MAI-602 decreased the expression of both GHRH and GHRH-R, whereas as a GHRH-R agonist increased levels of both the hormone and its receptor. They also demonstrated that PAK1 appears to be a critical mediator of STAT3/NF-kappaB activity, and that MAI-602 works primarily by blocking PAK1-mediated inflammatory signaling.
“MIA-602 remarkably inhibits the growth of human in vitro and in vivo through the suppression of PAK1–STAT3/NF-kappaB signaling. Our study strongly highlights the therapeutic potential of GHRH-R antagonists in the treatment of gastric cancer patients. Knowledge gained in our study will shed light on how to select the appropriate patients for personalized cancer therapy using GHRH-R antagonists,” Dr. Schally and his coauthors wrote.
The study was supported by the Li Ka Shing Foundation, Chinese foundation, and government grants to individual researchers, as well as support from the the Medical Research Service of the U.S. Department of Veterans Affairs, South Florida Veterans Affairs Foundation for Research and Education, and the University of Miami.
It sounds counterintuitive, but targeting a neuropeptide hormone produced in the hypothalamus may be an effective strategy for treating gastric cancer, the second most common cause of cancer deaths worldwide, investigators from China and the United States contend.
Growth hormone–releasing hormone (GHRH) and its receptor (GHRH-R) are found primarily in the anterior pituitary gland, but are also present in gastric cancers, other solid tumors, and lymphomas. Increased levels of GHRH-R in tumor samples from patients with gastric cancer are associated with poor outcomes, noted Andrew V. Schally, PhD, MD, DSc, of the University of Miami, and his colleagues at the Shantou (China) University Medical College.
Furthermore, an experimental peptide drug labeled MIA-602 that targets GHRH-R inhibited the growth of gastric cancer cell lines and human tumor xenografts in mice, the investigators reported in the journal PNAS.
“The GHRH receptor is both a biomarker that can confirm prognosis and a therapeutic target,” Dr. Schally said in a statement.
Elevated GHRH-R expression in tumors
GHRH-R antagonists such as MIA-602 work through downregulation of the p21-activated kinase 1 (PAK1)–mediated signal transducer and activator of transcription 3 (STAT3)/nuclear factor–kappaB (NF-kappaB) inflammatory pathway. This pathway is involved in the interplay between inflammatory processes and intracellular signaling thought to be the cause of gastric cancer tumorigenesis and progression, the investigators explained.
They first looked for GHRH-R expression in gastric cancer samples from 106 patients, using immunohistochemistry staining of primary tumors and adjacent normal tissues. They found that gastric cancer tissues “exhibited robust expression of GHRH-R, compared with normal tissues.”
In 50 samples, GHRH-R was determined to be overexpressed, and this overexpression was significantly associated with both greater tumor size (P = .031) and high pathologic tumor stage (P = .001). Increasing expression of GHRH-R was also significantly associated with worse overall survival (P less than .001).
They confirmed these findings in samples from a multinational cohort of patients, which again showed that the highest levels of GHRH-R expression were associated with poor overall survival (P less than .001).
The authors also looked at messenger RNA expression and gene copy number in 65 gastric cancer samples and 19 adjacent normal tissue samples, and found that GHRH-R mRNA was significantly higher in tumor tissues than normal control tissues (P less than .001).
MAI-602 in vitro and in vivo
To see whether MAI-602 could inhibit the growth of gastric cancer cells, the investigators tried it at various doses in three human gastric cancer cell lines, and found that it inhibited cells in a dose-dependent fashion, compared with vehicle used as a control (P less than .001).
In addition, the experimental agent “exhibited remarkable inhibitory effects on tumor growth in vivo” in mice with human tumor xenografts (P less than .001).
Finally, they showed that the cancer suppression effects of MAI-602 work through inhibition of STAT3/NF-kappaB inflammatory signaling. In vitro and in vivo, MAI-602 decreased the expression of both GHRH and GHRH-R, whereas as a GHRH-R agonist increased levels of both the hormone and its receptor. They also demonstrated that PAK1 appears to be a critical mediator of STAT3/NF-kappaB activity, and that MAI-602 works primarily by blocking PAK1-mediated inflammatory signaling.
“MIA-602 remarkably inhibits the growth of human in vitro and in vivo through the suppression of PAK1–STAT3/NF-kappaB signaling. Our study strongly highlights the therapeutic potential of GHRH-R antagonists in the treatment of gastric cancer patients. Knowledge gained in our study will shed light on how to select the appropriate patients for personalized cancer therapy using GHRH-R antagonists,” Dr. Schally and his coauthors wrote.
The study was supported by the Li Ka Shing Foundation, Chinese foundation, and government grants to individual researchers, as well as support from the the Medical Research Service of the U.S. Department of Veterans Affairs, South Florida Veterans Affairs Foundation for Research and Education, and the University of Miami.
It sounds counterintuitive, but targeting a neuropeptide hormone produced in the hypothalamus may be an effective strategy for treating gastric cancer, the second most common cause of cancer deaths worldwide, investigators from China and the United States contend.
Growth hormone–releasing hormone (GHRH) and its receptor (GHRH-R) are found primarily in the anterior pituitary gland, but are also present in gastric cancers, other solid tumors, and lymphomas. Increased levels of GHRH-R in tumor samples from patients with gastric cancer are associated with poor outcomes, noted Andrew V. Schally, PhD, MD, DSc, of the University of Miami, and his colleagues at the Shantou (China) University Medical College.
Furthermore, an experimental peptide drug labeled MIA-602 that targets GHRH-R inhibited the growth of gastric cancer cell lines and human tumor xenografts in mice, the investigators reported in the journal PNAS.
“The GHRH receptor is both a biomarker that can confirm prognosis and a therapeutic target,” Dr. Schally said in a statement.
Elevated GHRH-R expression in tumors
GHRH-R antagonists such as MIA-602 work through downregulation of the p21-activated kinase 1 (PAK1)–mediated signal transducer and activator of transcription 3 (STAT3)/nuclear factor–kappaB (NF-kappaB) inflammatory pathway. This pathway is involved in the interplay between inflammatory processes and intracellular signaling thought to be the cause of gastric cancer tumorigenesis and progression, the investigators explained.
They first looked for GHRH-R expression in gastric cancer samples from 106 patients, using immunohistochemistry staining of primary tumors and adjacent normal tissues. They found that gastric cancer tissues “exhibited robust expression of GHRH-R, compared with normal tissues.”
In 50 samples, GHRH-R was determined to be overexpressed, and this overexpression was significantly associated with both greater tumor size (P = .031) and high pathologic tumor stage (P = .001). Increasing expression of GHRH-R was also significantly associated with worse overall survival (P less than .001).
They confirmed these findings in samples from a multinational cohort of patients, which again showed that the highest levels of GHRH-R expression were associated with poor overall survival (P less than .001).
The authors also looked at messenger RNA expression and gene copy number in 65 gastric cancer samples and 19 adjacent normal tissue samples, and found that GHRH-R mRNA was significantly higher in tumor tissues than normal control tissues (P less than .001).
MAI-602 in vitro and in vivo
To see whether MAI-602 could inhibit the growth of gastric cancer cells, the investigators tried it at various doses in three human gastric cancer cell lines, and found that it inhibited cells in a dose-dependent fashion, compared with vehicle used as a control (P less than .001).
In addition, the experimental agent “exhibited remarkable inhibitory effects on tumor growth in vivo” in mice with human tumor xenografts (P less than .001).
Finally, they showed that the cancer suppression effects of MAI-602 work through inhibition of STAT3/NF-kappaB inflammatory signaling. In vitro and in vivo, MAI-602 decreased the expression of both GHRH and GHRH-R, whereas as a GHRH-R agonist increased levels of both the hormone and its receptor. They also demonstrated that PAK1 appears to be a critical mediator of STAT3/NF-kappaB activity, and that MAI-602 works primarily by blocking PAK1-mediated inflammatory signaling.
“MIA-602 remarkably inhibits the growth of human in vitro and in vivo through the suppression of PAK1–STAT3/NF-kappaB signaling. Our study strongly highlights the therapeutic potential of GHRH-R antagonists in the treatment of gastric cancer patients. Knowledge gained in our study will shed light on how to select the appropriate patients for personalized cancer therapy using GHRH-R antagonists,” Dr. Schally and his coauthors wrote.
The study was supported by the Li Ka Shing Foundation, Chinese foundation, and government grants to individual researchers, as well as support from the the Medical Research Service of the U.S. Department of Veterans Affairs, South Florida Veterans Affairs Foundation for Research and Education, and the University of Miami.
FROM PNAS
Key clinical point:
Major finding: The experimental GHRH-R antagonist MAI-602 inhibited gastric cancer growth in cell lines and human tumor xenograft models.
Data source: Proof of concept experiments showing the relationship between GHRH-R and gastric cancer, and elucidation of a method for targeting GHRH-R with an investigational peptide compound.
Disclosures: The study was supported by the Li Ka Shing Foundation, Chinese foundation, and government grants to individual researchers, as well as support from the the Medical Research Service of the U.S. Department of Veterans Affairs, South Florida Veterans Affairs Foundation for Research and Education, and the University of Miami.
Painful Oral and Genital Ulcers
The Diagnosis: Pemphigus Vegetans
Pemphigus vegetans is a rare variant of pemphigus vulgaris. Clinically, pemphigus vegetans is characterized by vegetative lesions over the flexures, but any area of the skin may be involved. There have been case reports involving the scalp,1,2 mouth,3 and foot.4 There are 2 clinical subtypes: the Neumann type and the Hallopeau type.5 The Hallopeau type is relatively benign, requires lower doses of systemic corticosteroids, and has a prolonged remission, while the Neumann type necessitates higher doses of systemic corticosteroids and often presents with relapses and remissions.
The diagnosis of pemphigus vegetans is based on clinical suspicion and confirmed by histological examination and immunological findings. The diagnosis may be difficult, as its presentation varies and histopathological findings may resemble other conditions.
Systemic corticosteroids are the well-established drug of choice for treating pemphigus vegetans to induce remission and maintain healing before cautiously tapering down the dosage approximately 50% every 2 weeks.6 Adjuvant drugs used in conjunction with steroids for steroid-sparing purpose include azathioprine, cyclophosphamide, mycophenolate mofetil, methotrexate, and cyclosporine.6 Pulsed intravenous steroids,7 intravenous immunoglobulins,8 pulsed dexamethasone cyclophosphamide,9 and extracorporeal photopheresis10 are given for severe and recalcitrant disease.
Laboratory investigations of our patient showed a normal complete blood cell count and a normal renal and liver profile. Herpes simplex virus serology was positive for type 1 and type 2 IgM and IgG. Urethral swab was dry and negative for gonorrhea. Serology for chlamydia, toxoplasma, amoebiasis, and leishmaniasis was negative. Human immunodeficiency virus serology, hepatitis screening, rapid plasma reagin, Treponema pallidum hemagglutination, rheumatoid factor, and antinuclear antibody all were negative. The patient was given a course of oral acyclovir 400 mg 3 times daily and empirical treatment with oral doxycycline 100 mg twice daily for a week with no clinical response.
Two biopsies from the perianal ulcers showed inflamed squamous papillomata with no Donovan bodies. A third biopsy from an intact blister showed acantholytic cells in the suprabasal bullae with eosinophilic and lymphocytic infiltrates at the upper dermis. Direct immunofluorescence demonstrated intercellular C3 and IgG deposits.
The patient was started on oral prednisolone at 1 mg/kg daily and oral azathioprine 50 mg daily with resolution of the perianal, penile, and oral ulcers (Figures 1 and 2). He achieved good suppression of further eruption. At the patient's most recent follow-up (2.5 years after the initial presentation), he was in remission and was currently taking oral azathioprine 100 mg once daily and no oral corticosteroids.
- Danopoulou I, Stavropoulos P, Stratigos A, et al. Pemphigus vegetans confined to the scalp. Int J Dermatol. 2006;45:1008-1009.
- Mori M, Mariotti G, Grandi V, et al. Pemphigus vegetans of the scalp [published online October 22,2014]. J Eur Acad Dermatol Venereol. 2016;30:368-370.
- Augusto de Oliveira M, Martins E Martins F, Lourenço S, et al. Oral pemphigus vegetans: a case report. Dermatol Online J. 2012;18:10.
- Ma DL, Fang K. Hallopeau type of pemphigus vegetans confined to the right foot: case report. Chin Med J (Engl). 2009;122:588-590.
- Ahmed AR, Blose DA. Pemphigus vegetans. Neumann type and Hallopeau type. Int J Dermatol. 1984;23:135-141.
- Harman KE, Albert S, Black MM, et al. Guidelines for the management of pemphigus vulgaris. Br J Dermatol. 2003;149:926-937.
- Chryssomallis F, Dimitriades A, Chaidemenos GC, et al. Steroid-pulse therapy in pemphigus vulgaris long term follow-up. Int J Dermatol. 1995;34:438-442.
- Ahmed AR. Intravenous immunoglobulin therapy in the treatment of patients with pemphigus vulgaris unresponsive to conventional immunosuppressive treatment. J Am Acad Dermatol. 2001;45:679-690.
- Pasricha JS, Khaitan BK, Raman RS, et al. Dexamethasone-cyclophosphamide pulse therapy for pemphigus. Int J Dermatol. 1995;34:875-882.
- Rook AH, Jegasothy BV, Heald P, et al. Extracorporeal photochemotherapy for drug-resistant pemphigus vulgaris. Ann Int Med. 1990;112:303-305.
The Diagnosis: Pemphigus Vegetans
Pemphigus vegetans is a rare variant of pemphigus vulgaris. Clinically, pemphigus vegetans is characterized by vegetative lesions over the flexures, but any area of the skin may be involved. There have been case reports involving the scalp,1,2 mouth,3 and foot.4 There are 2 clinical subtypes: the Neumann type and the Hallopeau type.5 The Hallopeau type is relatively benign, requires lower doses of systemic corticosteroids, and has a prolonged remission, while the Neumann type necessitates higher doses of systemic corticosteroids and often presents with relapses and remissions.
The diagnosis of pemphigus vegetans is based on clinical suspicion and confirmed by histological examination and immunological findings. The diagnosis may be difficult, as its presentation varies and histopathological findings may resemble other conditions.
Systemic corticosteroids are the well-established drug of choice for treating pemphigus vegetans to induce remission and maintain healing before cautiously tapering down the dosage approximately 50% every 2 weeks.6 Adjuvant drugs used in conjunction with steroids for steroid-sparing purpose include azathioprine, cyclophosphamide, mycophenolate mofetil, methotrexate, and cyclosporine.6 Pulsed intravenous steroids,7 intravenous immunoglobulins,8 pulsed dexamethasone cyclophosphamide,9 and extracorporeal photopheresis10 are given for severe and recalcitrant disease.
Laboratory investigations of our patient showed a normal complete blood cell count and a normal renal and liver profile. Herpes simplex virus serology was positive for type 1 and type 2 IgM and IgG. Urethral swab was dry and negative for gonorrhea. Serology for chlamydia, toxoplasma, amoebiasis, and leishmaniasis was negative. Human immunodeficiency virus serology, hepatitis screening, rapid plasma reagin, Treponema pallidum hemagglutination, rheumatoid factor, and antinuclear antibody all were negative. The patient was given a course of oral acyclovir 400 mg 3 times daily and empirical treatment with oral doxycycline 100 mg twice daily for a week with no clinical response.
Two biopsies from the perianal ulcers showed inflamed squamous papillomata with no Donovan bodies. A third biopsy from an intact blister showed acantholytic cells in the suprabasal bullae with eosinophilic and lymphocytic infiltrates at the upper dermis. Direct immunofluorescence demonstrated intercellular C3 and IgG deposits.
The patient was started on oral prednisolone at 1 mg/kg daily and oral azathioprine 50 mg daily with resolution of the perianal, penile, and oral ulcers (Figures 1 and 2). He achieved good suppression of further eruption. At the patient's most recent follow-up (2.5 years after the initial presentation), he was in remission and was currently taking oral azathioprine 100 mg once daily and no oral corticosteroids.


The Diagnosis: Pemphigus Vegetans
Pemphigus vegetans is a rare variant of pemphigus vulgaris. Clinically, pemphigus vegetans is characterized by vegetative lesions over the flexures, but any area of the skin may be involved. There have been case reports involving the scalp,1,2 mouth,3 and foot.4 There are 2 clinical subtypes: the Neumann type and the Hallopeau type.5 The Hallopeau type is relatively benign, requires lower doses of systemic corticosteroids, and has a prolonged remission, while the Neumann type necessitates higher doses of systemic corticosteroids and often presents with relapses and remissions.
The diagnosis of pemphigus vegetans is based on clinical suspicion and confirmed by histological examination and immunological findings. The diagnosis may be difficult, as its presentation varies and histopathological findings may resemble other conditions.
Systemic corticosteroids are the well-established drug of choice for treating pemphigus vegetans to induce remission and maintain healing before cautiously tapering down the dosage approximately 50% every 2 weeks.6 Adjuvant drugs used in conjunction with steroids for steroid-sparing purpose include azathioprine, cyclophosphamide, mycophenolate mofetil, methotrexate, and cyclosporine.6 Pulsed intravenous steroids,7 intravenous immunoglobulins,8 pulsed dexamethasone cyclophosphamide,9 and extracorporeal photopheresis10 are given for severe and recalcitrant disease.
Laboratory investigations of our patient showed a normal complete blood cell count and a normal renal and liver profile. Herpes simplex virus serology was positive for type 1 and type 2 IgM and IgG. Urethral swab was dry and negative for gonorrhea. Serology for chlamydia, toxoplasma, amoebiasis, and leishmaniasis was negative. Human immunodeficiency virus serology, hepatitis screening, rapid plasma reagin, Treponema pallidum hemagglutination, rheumatoid factor, and antinuclear antibody all were negative. The patient was given a course of oral acyclovir 400 mg 3 times daily and empirical treatment with oral doxycycline 100 mg twice daily for a week with no clinical response.
Two biopsies from the perianal ulcers showed inflamed squamous papillomata with no Donovan bodies. A third biopsy from an intact blister showed acantholytic cells in the suprabasal bullae with eosinophilic and lymphocytic infiltrates at the upper dermis. Direct immunofluorescence demonstrated intercellular C3 and IgG deposits.
The patient was started on oral prednisolone at 1 mg/kg daily and oral azathioprine 50 mg daily with resolution of the perianal, penile, and oral ulcers (Figures 1 and 2). He achieved good suppression of further eruption. At the patient's most recent follow-up (2.5 years after the initial presentation), he was in remission and was currently taking oral azathioprine 100 mg once daily and no oral corticosteroids.


- Danopoulou I, Stavropoulos P, Stratigos A, et al. Pemphigus vegetans confined to the scalp. Int J Dermatol. 2006;45:1008-1009.
- Mori M, Mariotti G, Grandi V, et al. Pemphigus vegetans of the scalp [published online October 22,2014]. J Eur Acad Dermatol Venereol. 2016;30:368-370.
- Augusto de Oliveira M, Martins E Martins F, Lourenço S, et al. Oral pemphigus vegetans: a case report. Dermatol Online J. 2012;18:10.
- Ma DL, Fang K. Hallopeau type of pemphigus vegetans confined to the right foot: case report. Chin Med J (Engl). 2009;122:588-590.
- Ahmed AR, Blose DA. Pemphigus vegetans. Neumann type and Hallopeau type. Int J Dermatol. 1984;23:135-141.
- Harman KE, Albert S, Black MM, et al. Guidelines for the management of pemphigus vulgaris. Br J Dermatol. 2003;149:926-937.
- Chryssomallis F, Dimitriades A, Chaidemenos GC, et al. Steroid-pulse therapy in pemphigus vulgaris long term follow-up. Int J Dermatol. 1995;34:438-442.
- Ahmed AR. Intravenous immunoglobulin therapy in the treatment of patients with pemphigus vulgaris unresponsive to conventional immunosuppressive treatment. J Am Acad Dermatol. 2001;45:679-690.
- Pasricha JS, Khaitan BK, Raman RS, et al. Dexamethasone-cyclophosphamide pulse therapy for pemphigus. Int J Dermatol. 1995;34:875-882.
- Rook AH, Jegasothy BV, Heald P, et al. Extracorporeal photochemotherapy for drug-resistant pemphigus vulgaris. Ann Int Med. 1990;112:303-305.
- Danopoulou I, Stavropoulos P, Stratigos A, et al. Pemphigus vegetans confined to the scalp. Int J Dermatol. 2006;45:1008-1009.
- Mori M, Mariotti G, Grandi V, et al. Pemphigus vegetans of the scalp [published online October 22,2014]. J Eur Acad Dermatol Venereol. 2016;30:368-370.
- Augusto de Oliveira M, Martins E Martins F, Lourenço S, et al. Oral pemphigus vegetans: a case report. Dermatol Online J. 2012;18:10.
- Ma DL, Fang K. Hallopeau type of pemphigus vegetans confined to the right foot: case report. Chin Med J (Engl). 2009;122:588-590.
- Ahmed AR, Blose DA. Pemphigus vegetans. Neumann type and Hallopeau type. Int J Dermatol. 1984;23:135-141.
- Harman KE, Albert S, Black MM, et al. Guidelines for the management of pemphigus vulgaris. Br J Dermatol. 2003;149:926-937.
- Chryssomallis F, Dimitriades A, Chaidemenos GC, et al. Steroid-pulse therapy in pemphigus vulgaris long term follow-up. Int J Dermatol. 1995;34:438-442.
- Ahmed AR. Intravenous immunoglobulin therapy in the treatment of patients with pemphigus vulgaris unresponsive to conventional immunosuppressive treatment. J Am Acad Dermatol. 2001;45:679-690.
- Pasricha JS, Khaitan BK, Raman RS, et al. Dexamethasone-cyclophosphamide pulse therapy for pemphigus. Int J Dermatol. 1995;34:875-882.
- Rook AH, Jegasothy BV, Heald P, et al. Extracorporeal photochemotherapy for drug-resistant pemphigus vulgaris. Ann Int Med. 1990;112:303-305.
A 52-year-old man presented with persistent painful oral ulcers and penile and perianal erosions of 6 months' duration. He strongly denied engaging in high-risk sexual activities and had lost 10 kg over the last 6 months. He did not report taking any over-the-counter or alternative medications. On physical examination there were multiple fissures on the lower lip with erosive white plaques on the tongue and buccal mucosa. There were erosions over the foreskin and glans penis and a few erosive plaques on the perianal skin. Bilateral inguinal lymph nodes were enlarged.
Diagnostic laparoscopy identifies ovarian cancers amenable to PCS
For women with suspected advanced epithelial ovarian cancer, diagnostic laparoscopy can help to distinguish between patients who could benefit from primary cytoreductive surgery (PCS) and those who might have better outcomes with neoadjuvant chemotherapy and interval cytoreductive surgery, according to investigators in the Netherlands.
In a randomized controlled trial exploring whether initial diagnostic laparoscopy could spare some patients from undergoing futile PCS, the investigators found that only 10% of patients assigned to diagnostic laparoscopy prior to PCS underwent a subsequent futile laparotomy, defined as residual disease greater than 1 cm following surgery. In contrast, 39% of women assigned to primary PCS had disease that might have been better treated by chemotherapy and interval surgery,
“In women with a plan for PCS, these data suggest that performance of diagnostic laparoscopy first is reasonable and that if cytoreduction to [less than] 1 cm of residual disease seems feasible, to proceed with PCS,” wrote Marrije R. Buist, MD of Academic Medical Center, Amsterdam, and colleagues.
Among women with International Federation of Gynecology and Obstetrics (FIGO) stage IIIC to IV epithelial ovarian cancer, survival depends largely on the ability of surgery to either completely remove disease, or to leave at best less than 1 cm of residual disease. However, aggressive surgery in patients with more extensive disease is associated with significant morbidities, the authors noted.
“If at PCS, extensive disease is present, surgery could be ceased, and neoadjuvant chemotherapy with interval surgery could be a good alternative treatment. Therefore, the identification of patients with extensive disease who are likely to have [more than] 1 cm of residual tumor after PCS, defined as a futile laparotomy, is important,” they wrote.
To test this idea, the investigators, from eight cancer centers in the Netherlands, enrolled 201 patients with suspected FIGO stage IIB ovarian cancer or higher, and randomly assigned them to undergo either initial diagnostic laparoscopy or PCS.
They found that 10 of the 102 patients (10%) assigned to diagnostic laparoscopy went on to undergo PCS that revealed residual disease greater than 1 cm, compared with 39 of the 99 patients (39%) assigned to PCS. This difference translated into a relative risk for futile laparotomy of 0.25 for diagnostic laparoscopy compared with PCS (P less than .001).
Only 3 (3%) patients in the diagnostic laparoscopy group went on to have both PCS and interval surgery, compared with 28 (28%) patients initially assigned to PCS (P less than .001).
The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
For women with suspected advanced epithelial ovarian cancer, diagnostic laparoscopy can help to distinguish between patients who could benefit from primary cytoreductive surgery (PCS) and those who might have better outcomes with neoadjuvant chemotherapy and interval cytoreductive surgery, according to investigators in the Netherlands.
In a randomized controlled trial exploring whether initial diagnostic laparoscopy could spare some patients from undergoing futile PCS, the investigators found that only 10% of patients assigned to diagnostic laparoscopy prior to PCS underwent a subsequent futile laparotomy, defined as residual disease greater than 1 cm following surgery. In contrast, 39% of women assigned to primary PCS had disease that might have been better treated by chemotherapy and interval surgery,
“In women with a plan for PCS, these data suggest that performance of diagnostic laparoscopy first is reasonable and that if cytoreduction to [less than] 1 cm of residual disease seems feasible, to proceed with PCS,” wrote Marrije R. Buist, MD of Academic Medical Center, Amsterdam, and colleagues.
Among women with International Federation of Gynecology and Obstetrics (FIGO) stage IIIC to IV epithelial ovarian cancer, survival depends largely on the ability of surgery to either completely remove disease, or to leave at best less than 1 cm of residual disease. However, aggressive surgery in patients with more extensive disease is associated with significant morbidities, the authors noted.
“If at PCS, extensive disease is present, surgery could be ceased, and neoadjuvant chemotherapy with interval surgery could be a good alternative treatment. Therefore, the identification of patients with extensive disease who are likely to have [more than] 1 cm of residual tumor after PCS, defined as a futile laparotomy, is important,” they wrote.
To test this idea, the investigators, from eight cancer centers in the Netherlands, enrolled 201 patients with suspected FIGO stage IIB ovarian cancer or higher, and randomly assigned them to undergo either initial diagnostic laparoscopy or PCS.
They found that 10 of the 102 patients (10%) assigned to diagnostic laparoscopy went on to undergo PCS that revealed residual disease greater than 1 cm, compared with 39 of the 99 patients (39%) assigned to PCS. This difference translated into a relative risk for futile laparotomy of 0.25 for diagnostic laparoscopy compared with PCS (P less than .001).
Only 3 (3%) patients in the diagnostic laparoscopy group went on to have both PCS and interval surgery, compared with 28 (28%) patients initially assigned to PCS (P less than .001).
The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
For women with suspected advanced epithelial ovarian cancer, diagnostic laparoscopy can help to distinguish between patients who could benefit from primary cytoreductive surgery (PCS) and those who might have better outcomes with neoadjuvant chemotherapy and interval cytoreductive surgery, according to investigators in the Netherlands.
In a randomized controlled trial exploring whether initial diagnostic laparoscopy could spare some patients from undergoing futile PCS, the investigators found that only 10% of patients assigned to diagnostic laparoscopy prior to PCS underwent a subsequent futile laparotomy, defined as residual disease greater than 1 cm following surgery. In contrast, 39% of women assigned to primary PCS had disease that might have been better treated by chemotherapy and interval surgery,
“In women with a plan for PCS, these data suggest that performance of diagnostic laparoscopy first is reasonable and that if cytoreduction to [less than] 1 cm of residual disease seems feasible, to proceed with PCS,” wrote Marrije R. Buist, MD of Academic Medical Center, Amsterdam, and colleagues.
Among women with International Federation of Gynecology and Obstetrics (FIGO) stage IIIC to IV epithelial ovarian cancer, survival depends largely on the ability of surgery to either completely remove disease, or to leave at best less than 1 cm of residual disease. However, aggressive surgery in patients with more extensive disease is associated with significant morbidities, the authors noted.
“If at PCS, extensive disease is present, surgery could be ceased, and neoadjuvant chemotherapy with interval surgery could be a good alternative treatment. Therefore, the identification of patients with extensive disease who are likely to have [more than] 1 cm of residual tumor after PCS, defined as a futile laparotomy, is important,” they wrote.
To test this idea, the investigators, from eight cancer centers in the Netherlands, enrolled 201 patients with suspected FIGO stage IIB ovarian cancer or higher, and randomly assigned them to undergo either initial diagnostic laparoscopy or PCS.
They found that 10 of the 102 patients (10%) assigned to diagnostic laparoscopy went on to undergo PCS that revealed residual disease greater than 1 cm, compared with 39 of the 99 patients (39%) assigned to PCS. This difference translated into a relative risk for futile laparotomy of 0.25 for diagnostic laparoscopy compared with PCS (P less than .001).
Only 3 (3%) patients in the diagnostic laparoscopy group went on to have both PCS and interval surgery, compared with 28 (28%) patients initially assigned to PCS (P less than .001).
The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
Key clinical point: Diagnostic laparoscopy can help to identify patients with advanced ovarian cancer who can best benefit from primary surgery or chemotherapy.
Major finding: Ten percent of women assigned to diagnostic laparoscopy underwent futile laparotomy, vs. 39% assigned to primary cytoreductive surgery.
Data source: Randomized controlled trial of 201 women with suspected FIGO stage IIB or greater disease.
Disclosures The Dutch Organization for Health Research and Development supported the study. All but one coauthor reported having no potential conflicts of interest.
Law & Medicine: How case law shapes EMTALA
(This is the second installment of a three-part series.)
Question: Emergency Medical Treatment and Labor Act (EMTALA) litigation has yielded which of the following rules of law?
A. The statute is applicable only when a patient is physically in the hospital’s emergency department (ED).
B. Directing an ambulance away from the ED is a violation of EMTALA.
C. All patients presenting to the ED must have an appropriate medical screening exam conforming to customary standard of care.
D. It is not what is performed in any medical screening exam, but whether it is applied evenly to all patients similarly situated.
E. The U.S. Supreme Court has held that an improper motive behind an unstable transfer is a prerequisite to an EMTALA violation.
Answer: D. In 1986, Congress enacted the Emergency Medical Treatment and Labor Act to ensure that all patients who present themselves to the emergency department are appropriately screened for an emergent medical condition, and if one is present, that they be stabilized prior to transfer or discharge.
In the 3 decades since its enactment, the statute has, as expected, spawned numerous lawsuits. Parts two and three of this series on EMTALA summarize the salient findings and rules of law in several interesting and impactful cases. These cases are neither encyclopedic nor necessarily representative of the types of litigation commonly encountered.
EMTALA is about events in the emergency department. They begin with the patient coming to the ED seeking treatment, and the statute specifically refers to “any individual ... [who] comes to the emergency department and a request is made on the individual’s behalf for examination or treatment for a medical condition.”
But what if the patient has yet to arrive, e.g., in an ambulance en route, and was diverted elsewhere in an unstable condition? In Hawaii’s case of Arrington v. Wong (237 F.3d 1066 [9th Cir. 2001]), the court was faced with whether the requirement that a patient must first “come to” the hospital means his or her literal physical presence in the hospital.
On May 5, 1996, Harold Arrington developed dyspnea while driving to his job as a security guard. En route to the closest medical facility, the Queen’s Medical Center, the ambulance personnel contacted Dr. Norbert Wong, the physician on call, describing the patient as being in severe respiratory distress, speaking one to two words at a time, and breathing about 50 times a minute.
Although Queen’s was not on diversionary status at the time, Dr. Wong thought it was okay for the ambulance to go instead to Tripler Hospital, a more distant hospital, as the patient’s doctor worked there. Unfortunately, by the time the ambulance arrived at Tripler, Mr. Arrington’s condition had deteriorated, and he was pronounced dead shortly after arrival.
The lower court ruled for the defendant, holding that the statute demanded an actual physical presence in the ED, but the 9th Circuit Court of Appeals reversed. It held that under EMTALA, a hospital may divert an ambulance that has contacted its emergency department and is on the way to that hospital only if the hospital is in diversionary status, because the diverting hospital then has a valid, treatment-related reason for doing so.
Such an interpretation of the law works no hardship on the hospital and is consistent with the Centers for Medicare & Medicaid Services’ regulation that “only requires hospitals that offer emergency services to provide screening and stabilizing treatment within the scope of their capabilities.” The 9th Circuit felt that this was consistent with the purpose and language of the EMTALA statute.
The next case, Summers v. Baptist Medical Center (91 F. 3d 1132 [8th Cir. 1996]), addressed the screening aspect of EMTALA, specifically on the distinction between disparity and adequacy in screening procedures. In Summers, the plaintiff fell from a tree while hunting and sustained bilateral hemothoraces, vertebral, rib, and sternal fractures. Incredibly, the diagnosis in the first hospital ED was muscle spasms, and the diagnoses only became clear when he checked into a second hospital 2 days later.
Still, the court ruled that there was no EMTALA violation, and that allegations of substandard care should be addressed under a negligence theory in state courts and not under EMTALA. The court reasoned that under the statute, an “inappropriate” screening is one that is performed in a disparate manner to similarly situated patients, and the hospital itself is usually left to define for itself what is within its capabilities. It is up to the hospital itself to determine what its screening procedures will be and to apply them alike to all patients with comparable complaints.
Likewise, in Vickers v. Nash (78 F.3d 139 [4th Cir. 1996]), an intoxicated patient who sustained a head injury following a fight died 4 days later from an epidural hematoma that was missed. He did have his head laceration treated in the ED, and was observed for 11 hours before discharge. The court ruled that the plaintiffs had failed to prove there was disparate treatment, and that hospitals can only be expected to stabilize emergency medical conditions known to them at the time.
Finally, in its first and thus far only EMTALA case, the U.S. Supreme Court in 1999 looked at whether an improper motive was a prerequisite for a finding of an EMTALA violation regarding stabilization and transfer.
In Roberts v. Galen of Virginia (119 S. Court 685 [1999]), the patient, injured in a truck accident, required a splenectomy and ventilator support. After a prolonged hospital stay, she was about to be moved to a nearby nursing home when she developed a high fever from an infection, and had to be transferred to an acute care facility. Her guardian, Roberts, brought suit, asserting violations of EMTALA’s stabilization and transfer requirements. The hospital argued that no material deterioration of the condition was likely to result from or occur during the transfer, and the district court determined her transfer was not prompted by an improper motive.
On appeal, the 6th Circuit Court of Appeals affirmed, extending its earlier holding that a showing of improper motive was required to make out an inadequate screening claim under EMTALA.
The hospital raised a number of important defenses, which included the physician lacking actual knowledge that the patient had an emergency medical condition, that EMTALA did not apply to in-hospital treatment and discharge decisions, and denying that EMTALA imposes minimum substantive standards of medical care.
The U.S. Supreme Court granted certiorari on the single issue whether the improper motive test should apply to an allegedly wrongful transfer. Overturning the appeals court, it held that Section 1395dd(b) (stabilization and transfer) contained no express or implied “improper motive” requirement. The Supreme Court declined to resolve broader issues under the statute.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at [email protected].
(This is the second installment of a three-part series.)
Question: Emergency Medical Treatment and Labor Act (EMTALA) litigation has yielded which of the following rules of law?
A. The statute is applicable only when a patient is physically in the hospital’s emergency department (ED).
B. Directing an ambulance away from the ED is a violation of EMTALA.
C. All patients presenting to the ED must have an appropriate medical screening exam conforming to customary standard of care.
D. It is not what is performed in any medical screening exam, but whether it is applied evenly to all patients similarly situated.
E. The U.S. Supreme Court has held that an improper motive behind an unstable transfer is a prerequisite to an EMTALA violation.
Answer: D. In 1986, Congress enacted the Emergency Medical Treatment and Labor Act to ensure that all patients who present themselves to the emergency department are appropriately screened for an emergent medical condition, and if one is present, that they be stabilized prior to transfer or discharge.
In the 3 decades since its enactment, the statute has, as expected, spawned numerous lawsuits. Parts two and three of this series on EMTALA summarize the salient findings and rules of law in several interesting and impactful cases. These cases are neither encyclopedic nor necessarily representative of the types of litigation commonly encountered.
EMTALA is about events in the emergency department. They begin with the patient coming to the ED seeking treatment, and the statute specifically refers to “any individual ... [who] comes to the emergency department and a request is made on the individual’s behalf for examination or treatment for a medical condition.”
But what if the patient has yet to arrive, e.g., in an ambulance en route, and was diverted elsewhere in an unstable condition? In Hawaii’s case of Arrington v. Wong (237 F.3d 1066 [9th Cir. 2001]), the court was faced with whether the requirement that a patient must first “come to” the hospital means his or her literal physical presence in the hospital.
On May 5, 1996, Harold Arrington developed dyspnea while driving to his job as a security guard. En route to the closest medical facility, the Queen’s Medical Center, the ambulance personnel contacted Dr. Norbert Wong, the physician on call, describing the patient as being in severe respiratory distress, speaking one to two words at a time, and breathing about 50 times a minute.
Although Queen’s was not on diversionary status at the time, Dr. Wong thought it was okay for the ambulance to go instead to Tripler Hospital, a more distant hospital, as the patient’s doctor worked there. Unfortunately, by the time the ambulance arrived at Tripler, Mr. Arrington’s condition had deteriorated, and he was pronounced dead shortly after arrival.
The lower court ruled for the defendant, holding that the statute demanded an actual physical presence in the ED, but the 9th Circuit Court of Appeals reversed. It held that under EMTALA, a hospital may divert an ambulance that has contacted its emergency department and is on the way to that hospital only if the hospital is in diversionary status, because the diverting hospital then has a valid, treatment-related reason for doing so.
Such an interpretation of the law works no hardship on the hospital and is consistent with the Centers for Medicare & Medicaid Services’ regulation that “only requires hospitals that offer emergency services to provide screening and stabilizing treatment within the scope of their capabilities.” The 9th Circuit felt that this was consistent with the purpose and language of the EMTALA statute.
The next case, Summers v. Baptist Medical Center (91 F. 3d 1132 [8th Cir. 1996]), addressed the screening aspect of EMTALA, specifically on the distinction between disparity and adequacy in screening procedures. In Summers, the plaintiff fell from a tree while hunting and sustained bilateral hemothoraces, vertebral, rib, and sternal fractures. Incredibly, the diagnosis in the first hospital ED was muscle spasms, and the diagnoses only became clear when he checked into a second hospital 2 days later.
Still, the court ruled that there was no EMTALA violation, and that allegations of substandard care should be addressed under a negligence theory in state courts and not under EMTALA. The court reasoned that under the statute, an “inappropriate” screening is one that is performed in a disparate manner to similarly situated patients, and the hospital itself is usually left to define for itself what is within its capabilities. It is up to the hospital itself to determine what its screening procedures will be and to apply them alike to all patients with comparable complaints.
Likewise, in Vickers v. Nash (78 F.3d 139 [4th Cir. 1996]), an intoxicated patient who sustained a head injury following a fight died 4 days later from an epidural hematoma that was missed. He did have his head laceration treated in the ED, and was observed for 11 hours before discharge. The court ruled that the plaintiffs had failed to prove there was disparate treatment, and that hospitals can only be expected to stabilize emergency medical conditions known to them at the time.
Finally, in its first and thus far only EMTALA case, the U.S. Supreme Court in 1999 looked at whether an improper motive was a prerequisite for a finding of an EMTALA violation regarding stabilization and transfer.
In Roberts v. Galen of Virginia (119 S. Court 685 [1999]), the patient, injured in a truck accident, required a splenectomy and ventilator support. After a prolonged hospital stay, she was about to be moved to a nearby nursing home when she developed a high fever from an infection, and had to be transferred to an acute care facility. Her guardian, Roberts, brought suit, asserting violations of EMTALA’s stabilization and transfer requirements. The hospital argued that no material deterioration of the condition was likely to result from or occur during the transfer, and the district court determined her transfer was not prompted by an improper motive.
On appeal, the 6th Circuit Court of Appeals affirmed, extending its earlier holding that a showing of improper motive was required to make out an inadequate screening claim under EMTALA.
The hospital raised a number of important defenses, which included the physician lacking actual knowledge that the patient had an emergency medical condition, that EMTALA did not apply to in-hospital treatment and discharge decisions, and denying that EMTALA imposes minimum substantive standards of medical care.
The U.S. Supreme Court granted certiorari on the single issue whether the improper motive test should apply to an allegedly wrongful transfer. Overturning the appeals court, it held that Section 1395dd(b) (stabilization and transfer) contained no express or implied “improper motive” requirement. The Supreme Court declined to resolve broader issues under the statute.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at [email protected].
(This is the second installment of a three-part series.)
Question: Emergency Medical Treatment and Labor Act (EMTALA) litigation has yielded which of the following rules of law?
A. The statute is applicable only when a patient is physically in the hospital’s emergency department (ED).
B. Directing an ambulance away from the ED is a violation of EMTALA.
C. All patients presenting to the ED must have an appropriate medical screening exam conforming to customary standard of care.
D. It is not what is performed in any medical screening exam, but whether it is applied evenly to all patients similarly situated.
E. The U.S. Supreme Court has held that an improper motive behind an unstable transfer is a prerequisite to an EMTALA violation.
Answer: D. In 1986, Congress enacted the Emergency Medical Treatment and Labor Act to ensure that all patients who present themselves to the emergency department are appropriately screened for an emergent medical condition, and if one is present, that they be stabilized prior to transfer or discharge.
In the 3 decades since its enactment, the statute has, as expected, spawned numerous lawsuits. Parts two and three of this series on EMTALA summarize the salient findings and rules of law in several interesting and impactful cases. These cases are neither encyclopedic nor necessarily representative of the types of litigation commonly encountered.
EMTALA is about events in the emergency department. They begin with the patient coming to the ED seeking treatment, and the statute specifically refers to “any individual ... [who] comes to the emergency department and a request is made on the individual’s behalf for examination or treatment for a medical condition.”
But what if the patient has yet to arrive, e.g., in an ambulance en route, and was diverted elsewhere in an unstable condition? In Hawaii’s case of Arrington v. Wong (237 F.3d 1066 [9th Cir. 2001]), the court was faced with whether the requirement that a patient must first “come to” the hospital means his or her literal physical presence in the hospital.
On May 5, 1996, Harold Arrington developed dyspnea while driving to his job as a security guard. En route to the closest medical facility, the Queen’s Medical Center, the ambulance personnel contacted Dr. Norbert Wong, the physician on call, describing the patient as being in severe respiratory distress, speaking one to two words at a time, and breathing about 50 times a minute.
Although Queen’s was not on diversionary status at the time, Dr. Wong thought it was okay for the ambulance to go instead to Tripler Hospital, a more distant hospital, as the patient’s doctor worked there. Unfortunately, by the time the ambulance arrived at Tripler, Mr. Arrington’s condition had deteriorated, and he was pronounced dead shortly after arrival.
The lower court ruled for the defendant, holding that the statute demanded an actual physical presence in the ED, but the 9th Circuit Court of Appeals reversed. It held that under EMTALA, a hospital may divert an ambulance that has contacted its emergency department and is on the way to that hospital only if the hospital is in diversionary status, because the diverting hospital then has a valid, treatment-related reason for doing so.
Such an interpretation of the law works no hardship on the hospital and is consistent with the Centers for Medicare & Medicaid Services’ regulation that “only requires hospitals that offer emergency services to provide screening and stabilizing treatment within the scope of their capabilities.” The 9th Circuit felt that this was consistent with the purpose and language of the EMTALA statute.
The next case, Summers v. Baptist Medical Center (91 F. 3d 1132 [8th Cir. 1996]), addressed the screening aspect of EMTALA, specifically on the distinction between disparity and adequacy in screening procedures. In Summers, the plaintiff fell from a tree while hunting and sustained bilateral hemothoraces, vertebral, rib, and sternal fractures. Incredibly, the diagnosis in the first hospital ED was muscle spasms, and the diagnoses only became clear when he checked into a second hospital 2 days later.
Still, the court ruled that there was no EMTALA violation, and that allegations of substandard care should be addressed under a negligence theory in state courts and not under EMTALA. The court reasoned that under the statute, an “inappropriate” screening is one that is performed in a disparate manner to similarly situated patients, and the hospital itself is usually left to define for itself what is within its capabilities. It is up to the hospital itself to determine what its screening procedures will be and to apply them alike to all patients with comparable complaints.
Likewise, in Vickers v. Nash (78 F.3d 139 [4th Cir. 1996]), an intoxicated patient who sustained a head injury following a fight died 4 days later from an epidural hematoma that was missed. He did have his head laceration treated in the ED, and was observed for 11 hours before discharge. The court ruled that the plaintiffs had failed to prove there was disparate treatment, and that hospitals can only be expected to stabilize emergency medical conditions known to them at the time.
Finally, in its first and thus far only EMTALA case, the U.S. Supreme Court in 1999 looked at whether an improper motive was a prerequisite for a finding of an EMTALA violation regarding stabilization and transfer.
In Roberts v. Galen of Virginia (119 S. Court 685 [1999]), the patient, injured in a truck accident, required a splenectomy and ventilator support. After a prolonged hospital stay, she was about to be moved to a nearby nursing home when she developed a high fever from an infection, and had to be transferred to an acute care facility. Her guardian, Roberts, brought suit, asserting violations of EMTALA’s stabilization and transfer requirements. The hospital argued that no material deterioration of the condition was likely to result from or occur during the transfer, and the district court determined her transfer was not prompted by an improper motive.
On appeal, the 6th Circuit Court of Appeals affirmed, extending its earlier holding that a showing of improper motive was required to make out an inadequate screening claim under EMTALA.
The hospital raised a number of important defenses, which included the physician lacking actual knowledge that the patient had an emergency medical condition, that EMTALA did not apply to in-hospital treatment and discharge decisions, and denying that EMTALA imposes minimum substantive standards of medical care.
The U.S. Supreme Court granted certiorari on the single issue whether the improper motive test should apply to an allegedly wrongful transfer. Overturning the appeals court, it held that Section 1395dd(b) (stabilization and transfer) contained no express or implied “improper motive” requirement. The Supreme Court declined to resolve broader issues under the statute.
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, and currently directs the St. Francis International Center for Healthcare Ethics in Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. Some of the articles in this series are adapted from the author’s 2006 book, “Medical Malpractice: Understanding the Law, Managing the Risk,” and his 2012 Halsbury treatise, “Medical Negligence and Professional Misconduct.” For additional information, readers may contact the author at [email protected].
What’s New for Federal Practitioner in 2017?
It has been a long time since a new year has brought as much uncertainty as 2017 promises to bring, making some federal employees excited and others apprehensive. Rumors abound of how the federal health care sector may change: Hiring freezes, manpower cuts, and privatization are all concerns of Federal Practitioner readers. As in the past, we will keep you up-to-date with in-depth interviews of leaders in federal health care, intelligent coverage of news stories impacting your practice, and clinical and research articles about new programs and initiatives.
Also this year, we are pleased to announce several new regular columns that we hope will inform and entertain you. The first is a column on mental health and traumatic brain injury in the DoD and the VA. We are privileged to have U.S. Army COL (Ret) Elspeth Cameron Ritchie, MD, MPH, edit this column. She is widely known and respected and brings her vast experience to the column as an active-duty psychiatrist coupled with her current position as a VA physician. Dr. Ritchie will author articles as well as edit those of her VA and DoD colleagues. Mental health touches almost every aspect of federal practice, and we all will learn our contributions and challenges in this rapidly moving specialty.
Whereas the mental health column looks toward the scientific future, the second column looks back to the humanistic past. We are thrilled that 2 physician-historians of military medicine, Robert Hierholzer, MD, a VA psychiatrist, and John Pierce, MD, a retired U.S. Army pediatrician share the writing and editing for this column, which will debut this spring.
Have you ever wondered who or how VA and military hospitals were named? These 2 historical writers have a wealth of interesting anecdotes and stories about VA and military medical centers. We hope you will enjoy reading the stories of military and veteran health care: the war heroes, devoted clinicians, and groundbreaking researchers who have left their mark on DoD and VA health care.
We also will be launching a new pilot study feature for clinicians and researchers who have a novel or valuable idea but have only a small number of participants or preliminary results. This will be a great way for new investigators, trainees, and young health care practitioners to present their work to the medical community.
These new editorial offerings are just a start—we also want to invite you, your colleagues, and learners to start your own new tradition of writing for Federal Practitioner. For those who have submitted articles in the past, please keep up the habit.We are eager to receive original research, review articles, and clinical cases from DoD, PHS, and VA mid-career and senior clinicians and researchers as well as articles describing innovative programs and modes of health care treatment and delivery. With a print circulation of more than 35,000 readers and very active online presence, consider Federal Practitioner for your next article!
This year my New Year’s resolution as editor-in-chief is to encourage readers to contact either Editor Reid Paul or me if you have an idea for an article you would like to write, a column you would like to see, or if you have an interest in serving as a peer reviewer or joining our Editorial Advisory Association. We want to hear from you about what you want and need from Federal Practitioner.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies.
It has been a long time since a new year has brought as much uncertainty as 2017 promises to bring, making some federal employees excited and others apprehensive. Rumors abound of how the federal health care sector may change: Hiring freezes, manpower cuts, and privatization are all concerns of Federal Practitioner readers. As in the past, we will keep you up-to-date with in-depth interviews of leaders in federal health care, intelligent coverage of news stories impacting your practice, and clinical and research articles about new programs and initiatives.
Also this year, we are pleased to announce several new regular columns that we hope will inform and entertain you. The first is a column on mental health and traumatic brain injury in the DoD and the VA. We are privileged to have U.S. Army COL (Ret) Elspeth Cameron Ritchie, MD, MPH, edit this column. She is widely known and respected and brings her vast experience to the column as an active-duty psychiatrist coupled with her current position as a VA physician. Dr. Ritchie will author articles as well as edit those of her VA and DoD colleagues. Mental health touches almost every aspect of federal practice, and we all will learn our contributions and challenges in this rapidly moving specialty.
Whereas the mental health column looks toward the scientific future, the second column looks back to the humanistic past. We are thrilled that 2 physician-historians of military medicine, Robert Hierholzer, MD, a VA psychiatrist, and John Pierce, MD, a retired U.S. Army pediatrician share the writing and editing for this column, which will debut this spring.
Have you ever wondered who or how VA and military hospitals were named? These 2 historical writers have a wealth of interesting anecdotes and stories about VA and military medical centers. We hope you will enjoy reading the stories of military and veteran health care: the war heroes, devoted clinicians, and groundbreaking researchers who have left their mark on DoD and VA health care.
We also will be launching a new pilot study feature for clinicians and researchers who have a novel or valuable idea but have only a small number of participants or preliminary results. This will be a great way for new investigators, trainees, and young health care practitioners to present their work to the medical community.
These new editorial offerings are just a start—we also want to invite you, your colleagues, and learners to start your own new tradition of writing for Federal Practitioner. For those who have submitted articles in the past, please keep up the habit.We are eager to receive original research, review articles, and clinical cases from DoD, PHS, and VA mid-career and senior clinicians and researchers as well as articles describing innovative programs and modes of health care treatment and delivery. With a print circulation of more than 35,000 readers and very active online presence, consider Federal Practitioner for your next article!
This year my New Year’s resolution as editor-in-chief is to encourage readers to contact either Editor Reid Paul or me if you have an idea for an article you would like to write, a column you would like to see, or if you have an interest in serving as a peer reviewer or joining our Editorial Advisory Association. We want to hear from you about what you want and need from Federal Practitioner.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies.
It has been a long time since a new year has brought as much uncertainty as 2017 promises to bring, making some federal employees excited and others apprehensive. Rumors abound of how the federal health care sector may change: Hiring freezes, manpower cuts, and privatization are all concerns of Federal Practitioner readers. As in the past, we will keep you up-to-date with in-depth interviews of leaders in federal health care, intelligent coverage of news stories impacting your practice, and clinical and research articles about new programs and initiatives.
Also this year, we are pleased to announce several new regular columns that we hope will inform and entertain you. The first is a column on mental health and traumatic brain injury in the DoD and the VA. We are privileged to have U.S. Army COL (Ret) Elspeth Cameron Ritchie, MD, MPH, edit this column. She is widely known and respected and brings her vast experience to the column as an active-duty psychiatrist coupled with her current position as a VA physician. Dr. Ritchie will author articles as well as edit those of her VA and DoD colleagues. Mental health touches almost every aspect of federal practice, and we all will learn our contributions and challenges in this rapidly moving specialty.
Whereas the mental health column looks toward the scientific future, the second column looks back to the humanistic past. We are thrilled that 2 physician-historians of military medicine, Robert Hierholzer, MD, a VA psychiatrist, and John Pierce, MD, a retired U.S. Army pediatrician share the writing and editing for this column, which will debut this spring.
Have you ever wondered who or how VA and military hospitals were named? These 2 historical writers have a wealth of interesting anecdotes and stories about VA and military medical centers. We hope you will enjoy reading the stories of military and veteran health care: the war heroes, devoted clinicians, and groundbreaking researchers who have left their mark on DoD and VA health care.
We also will be launching a new pilot study feature for clinicians and researchers who have a novel or valuable idea but have only a small number of participants or preliminary results. This will be a great way for new investigators, trainees, and young health care practitioners to present their work to the medical community.
These new editorial offerings are just a start—we also want to invite you, your colleagues, and learners to start your own new tradition of writing for Federal Practitioner. For those who have submitted articles in the past, please keep up the habit.We are eager to receive original research, review articles, and clinical cases from DoD, PHS, and VA mid-career and senior clinicians and researchers as well as articles describing innovative programs and modes of health care treatment and delivery. With a print circulation of more than 35,000 readers and very active online presence, consider Federal Practitioner for your next article!
This year my New Year’s resolution as editor-in-chief is to encourage readers to contact either Editor Reid Paul or me if you have an idea for an article you would like to write, a column you would like to see, or if you have an interest in serving as a peer reviewer or joining our Editorial Advisory Association. We want to hear from you about what you want and need from Federal Practitioner.
Author disclosures
The author reports no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the author and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies.
Discharges Against Medical Advice
Patients leave the hospital against medical advice (AMA) for a variety of reasons. The AMA rate is approximately 1% nationally but substantially higher at safety-net hospitals and has rapidly increased over the past decade.1-5 The principle that patients have the right to make choices about their healthcare, up to and including whether to leave the hospital against the advice of medical staff, is well-established law and a foundation of medical ethics.6 In practice, however, AMA discharges are often emotionally charged for both patients and providers, and, in the high-stress setting of AMA discharge, providers may be confused about their roles.7-9
The demographics of patients who leave AMA have been well described. Compared with conventionally discharged patients, AMA patients are younger, more likely to be male, and more likely a marginalized ethnic or racial minority.10-14 Patients with mental illnesses and addiction issues are overrepresented in AMA discharges, and complicated capacity assessments and limited resources may strain providers.7,8,15,16 Studies have repeatedly shown higher rates of readmission and mortality for AMA patients than for conventionally discharged patients.17-21 Whether AMA discharge is a marker for other prognostic factors that bode poorly for patients or contributes to negative outcomes, data suggest this group of patients is vulnerable, having mortality rates up to 40% higher 1 year after discharge, relative to conventionally discharged patients.12
Several models of standardized best practice approaches for AMA have been proposed by bioethicists.6,22,23 Although details of these approaches vary, all involve assessing the patient’s decision-making capacity, clarifying the risks of AMA discharge, addressing factors that might be prompting the discharge, formulating an alternative outpatient treatment plan or “next best” option, and documenting extensively. A recent study found patients often gave advance warning of an AMA discharge, but physicians rarely prepared by arranging follow-up care.8 The investigators hypothesized that providers might not have known what they were permitted to arrange for AMA patients, or might have thought that providing “second best” options went against their principles. The investigators noted that nurses might have become aware of AMA risk sooner than physicians did but could not act on this awareness by preparing medications and arranging follow-up.
Translating models of best practice care for AMA patients into clinical practice requires buy-in from bedside providers, not just bioethicists. Given the study findings that providers have misconceptions about their roles in the AMA discharge,7 it is prudent to investigate providers’ current practices, beliefs, and concerns about AMA discharges before introducing a new approach.
The present authors conducted a mixed-methods cross-sectional study of the state of AMA discharges at Highland Hospital (Oakland, California), a 236-bed county hospital and trauma center serving a primarily underserved urban patient population. The aim of this study was to assess current provider practices for AMA discharges and provider perceptions and knowledge about AMA discharges, ultimately to help direct future educational interventions with medical providers or hospital policy changes needed to improve the quality of AMA discharges.
METHODS
Phase 1 of this study involved identifying AMA patients through a review of data from Highland Hospital’s electronic medical records for 2014. These data included discharge status (eg, AMA vs other discharge types). The hospital’s floor clerk distinguishes between absent without official leave (AWOL; the patient leaves without notifying a provider) and AMA discharge. Discharges designated AWOL were excluded from the analyses.
In phase 2, a structured chart review (Appendix A) was performed for all patients identified during phase 1 as being discharged AMA in 2014. In these reviews, further assessment was made of patient and visit characteristics in hospitalizations that ended in AMA discharge, and of providers’ documentation of AMA discharges—that is, whether several factors were documented (capacity; predischarge indication that patient might leave AMA; reason for AMA; and indications that discharge medications, transportation, and follow-up were arranged). These visit factors were reviewed because the literature has identified them as being important markers for AMA discharge safety.6,8 Two research assistants, under the guidance of Dr. Stearns, reviewed the charts. To ensure agreement across chart reviews with respect to subjective questions (eg, whether capacity was adequately documented), the group reviewed the first 10 consecutive charts together; there was full agreement on how to classify the data of interest. Throughout the study, whenever a research assistant asked how to classify particular patient data, Dr. Stearns reviewed the data, and the research team made a decision together. Additional data, for AMA patients and for all patients admitted to Highland Hospital, were obtained from the hospital’s data warehouse, which pools data from within the health system.
Phase 3 involved surveying healthcare providers who were involved in patient care on the internal medicine and trauma surgery services at the hospital. These providers were selected because chart review revealed that the vast majority of patients who left AMA in 2014 were on one of these services. Surveys (Appendix B) asked participant providers to identify their role at the hospital, to provide a self-assessment of competence in various aspects of AMA discharge, to voice opinions about provider responsibilities in arranging follow-up for AMA patients, and to make suggestions about the AMA process. The authors designed these surveys, which included questions about aspects of care that have been highlighted in the AMA discharge literature as being important for AMA discharge safety.6,8,22,23 Surveys were distributed to providers at internal medicine and trauma surgery department meetings and nursing conferences. Data (without identifying information) were analyzed, and survey responses kept anonymous.
The Alameda Health System Institutional Review Board approved this project. Providers were given the option of writing their name and contact information at the top of the survey in order to be entered into a drawing to receive a prize for completion.
We performed statistical analyses of the patient charts and physician survey data using Stata (version 14.0, Stata Corp., College Station, Texas). We analyzed both patient- and encounter-level data. In demographic analyses, this approach prevented duplicate counting of patients who left AMA multiple times. Patient-level analyses compared the demographic characteristics of AMA patients and patients discharged conventionally from the hospital in 2014. In addition, patients with either 1 or multiple AMA discharges were compared to identify characteristics that might be linked to highest risk of recurrent AMA discharge in the hope that early identification of these patients might facilitate providers’ early awareness and preparation for follow-up care or hospitalization alternatives. We used ANOVAs for continuous variables and tests of proportions for categorical variables. On the encounter level, analyses examined data about each admission (eg, AMA forms signed, follow-up arrangements made, capacity documented, etc.) for all AMA discharges. We employed chi square tests to identify variations in healthcare provider survey responses. A P value < 0.05 was used as the significance cut-off point.
Staged logistic regression analyses, adjusted for demographic characteristics, were performed to assess the association between risk of leaving AMA (yes or no) and demographic characteristics and the association between risk of leaving AMA more than once (yes or no) and health-related characteristics.
RESULTS
Demographic, Clinical, and Utilization Characteristics
Of the 12,036 Highland Hospital admissions in 2014, 319 (2.7%) ended with an AMA discharge. Of the 8207 individual patients discharged, 268 left AMA once, and 29 left AMA multiple times. Further review of the Admissions, Discharges, and Transfers Report generated from the electronic medical record revealed that 15 AWOL discharges were misclassified as AMA discharges.
Compared with patients discharged conventionally, AMA patients were significantly younger; more likely to be male, to self-identify as Black/African American, and to be English-speaking; and less likely to self-identify as Asian/Pacific Islander or Hispanic/Latino or to be Chinese- or Spanish-speaking (Table 1). They were also more likely than all patients admitted to Highland to be homeless (15.7% vs 8.7%; P < 0.01). Multivariate regression analysis revealed persistent age and sex disparities, but racial disparities were mitigated in adjusted analyses (Appendix C). Language disparities persisted only for Spanish speakers, who had a significantly lower rate of AMA discharge, even in adjusted analyses.
The majority of AMA patients were on the internal medicine service (63.5%) or the trauma surgery service (24.8%). Regarding admission diagnosis, 17.2% of AMA patients were admitted for infections, 5.0% for drug or alcohol intoxication or withdrawal, 38.9% for acute noninfectious illnesses, 16.7% for decompensation of chronic disease, 18.4% for injuries or trauma, and 3.8% for pregnancy complications or labor. Compared with patients who left AMA once, patients who left AMA multiple times had higher rates of heavy alcohol use (53.9% vs 30.9%; P = 0.01) and illicit drug use (88.5% vs 53.7%; P < 0.001) (Table 2). In multivariate analyses, the increased odds of leaving AMA more than once persisted for current heavy illicit drug users compared with patients who had never engaged in illicit drug use.
Discharge Characteristics and Documentation
Providers documented a patient’s plan to leave AMA before actual discharge 17.3% of the time. The documented plan to leave had to indicate that the patient was actually considering leaving. For example, “Patient is eager to go home” was not enough to qualify as a plan, but “Patient is thinking of leaving” qualified. For 84.3% of AMA discharges, the hospital’s AMA form was signed and was included in the medical record. Documentation showed that medications were prescribed for AMA patients 21.4% of the time, follow-up was arranged 25.7% of the time, and follow-up was pending arrangement 14.8% of the time. The majority of AMA patients (71.4%) left during daytime hours. In 29.6% of AMA discharges, providers documented AMA patients had decision-making capacity.
Readmission After AMA Discharge
Of the 268 AMA patients, 67.7% were not readmitted within the 6 months after AMA, 24.5% had 1 or 2 readmissions, and the rest had 3 or more readmissions (1 patient had 15). In addition, 35.8% returned to the emergency department within 30 days, and 16.4% were readmitted within 30 days. In 2014, the hospital’s overall 30-day readmission rate was 10.8%. Of the patients readmitted within 6 months after AMA, 23.5% left AMA again at the next visit, 9.4% left AWOL, and 67.1% were discharged conventionally.
Drivers of Premature Discharge
Qualitative analysis of the 35.5% of patient charts documenting a reason for leaving the hospital revealed 3 broad, interrelated themes (Figure 1). The first theme, dissatisfaction with hospital care, included chart notations such as “His wife couldn’t sleep in the hospital room” and “Not satisfied with all-liquid diet.” The second theme, urgent personal issues, included comments such as “He has a very important court date for his children” and “He needed to take care of immigration forms.” The third theme, mental health and substance abuse issues, included notations such as “He wants to go smoke” and “Severe anxiety and prison flashbacks.”
Provider Self-Assessment and Beliefs
The survey was completed by 178 healthcare providers: 49.4% registered nurses, 19.1% trainee physicians, 20.8% attending physicians, and 10.7% other providers, including chaplains, social workers, and clerks. Regarding self-assessment of competency in AMA discharges, 94% of providers agreed they were comfortable assessing capacity, and 94% agreed they were comfortable talking with patients about the risks of leaving AMA (Figure 2). Nurses were more likely than trainee physicians to agree they knew what to do for patients who lacked capacity (74% vs 49%; P = 0.02). Most providers (70%) agreed they usually knew why their patients were leaving AMA; in this self-assessment, there were no significant differences between types of providers.
Regarding follow-up, attending physicians and trainee physicians demonstrated more agreement than nurses that AMA patients should receive medications and follow-up (94% and 84% vs 64%; P < 0.05). Nurses were more likely than attending physicians to say patients should lose their rights to hospital follow-up because of leaving AMA (38% vs 6%; P < 0.01). A minority of providers (37%) agreed transportation should be arranged. Addiction was the most common driver of AMA discharge (35%), followed by familial obligations (19%), dissatisfaction with hospital care (16%), and financial concerns (15%).
DISCUSSION
The demographic characteristics of AMA patients in this study are similar to those identified in other studies, showing overrepresentation of young male patients.12,14 Homeless patients were also overrepresented in the AMA discharge population at Highland Hospital—a finding that has not been consistently reported in prior studies, and that warrants further examination. In adjusted analyses, Spanish speakers had a lower rate of AMA discharge, and there were no racial variations. This is consistent with another study’s finding: that racial disparities in AMA discharge rates were largely attributable to confounders.24 Language differences may result from failure of staff to fully explain the option of AMA discharge to non-English speakers, or from fear of immigration consequences after AMA discharge. Further investigation of patient experiences is needed to identify factors that contribute to demographic variations in AMA discharge rates.25,26
Of the patients who left AMA multiple times, nearly all were actively using illicit drugs. In a recent study conducted at a safety-net hospital in Vancouver, Canada, 43% of patients with illicit drug use and at least 1 hospitalization left AMA at least once during the 6-year study period.11 Many factors might explain this correlation—addiction itself, poor pain control for patients with addiction issues, fears about incarceration, and poor treatment of drug users by healthcare staff.15 Although the medical literature highlights deficits in pain control for patients addicted to opiates, proposed solutions are sparse and focus on perioperative pain control and physician prescribing practices.27,28 At safety-net hospitals in which addiction is a factor in many hospitalizations, there is opportunity for new research in inpatient pain control for patients with substance dependence. In addition, harm reduction strategies—such as methadone maintenance for hospitalized patients with opiate dependence and abscess clinics as hospitalization alternatives for injection-associated infection treatment—may be key in improving safety for patients.11,15,29
Comparing the provider survey and chart review results highlights discordance between provider beliefs and clinical practice. Healthcare providers at Highland Hospital considered themselves competent in assessing capacity and talking with patients about the risks of AMA discharge. In practice, however, capacity was documented in less than a third of AMA discharges. Although the majority of providers thought medications and follow-up should be arranged for patients, arrangements were seldom made. This may be partially attributable to limited resources for making these arrangements. Average time to “third next available” primary care appointment within the county health system that includes Highland was 44.6 days for established patients during the period of study; for new primary care patients, the average wait for an appointment was 2 to 3 months. Highland has a same-day clinic, but inpatient providers are discouraged from using it as a postdischarge clinic for patients who would be better served in primary care. Medications and transportation are easily arranged during daytime hours but are not immediately available at night. In addition, some of this discrepancy may be attributable to the limited documentation rather than to provider failure to achieve their own benchmarks of quality care for AMA patients.
Documentation in AMA discharges is key for multiple reasons. Most AMA patients in this study signed an AMA form, and it could be that the rate of documenting decision-making capacity was low because providers thought a signed AMA form was adequate documentation of capacity and informed consent. In numerous court cases, however, these forms were found to be insufficient evidence of informed consent (lacking other supportive documentation) and possibly to go against the public good.30 In addition, high rates of repeat emergency department visits and readmissions for AMA patients, demonstrated here and in other studies, highlight the importance of careful documentation in informing subsequent providers about hospital returnees’ ongoing issues.17-19
This study also demonstrated differences between nurses and physicians in their beliefs about arranging follow-up for AMA patients. Nurses were less likely than physicians to think follow-up arrangements should be made for AMA patients and more likely to say these patients should lose the right to follow-up because of the AMA discharge. For conventional discharges, nurses provide patients with significantly more discharge education than interns or hospitalists do.31 This discrepancy highlights an urgent need for the education and involvement of nurses as stakeholders in the challenging AMA discharge process. Although the percentage of physicians who thought they were not obligated to provide medications and arrange follow-up for AMA patients was lower than the percentage of nurses, these beliefs contradict best practice guidelines for AMA discharges,22,23 and this finding calls attention to the need for interventions to improve adherence to professional and ethical guidelines in this aspect of clinical practice.
Providers showed a lack of familiarity with practice guidelines regarding certain aspects of the AMA discharge process. For example, most providers thought they should not have to arrange transportation for AMA patients, even though both the California Hospital Association Guidelines and the Highland Hospital internal policy on AMA discharges recommend arranging appropriate transportation.32 This finding suggests a need for educational interventions to ensure providers are informed about state and hospital policies, and a need to include both physicians and nurses in policymaking so theory can be tied to practice.
This study was limited to a single center with healthcare provider and patient populations that might not be generalizable to other settings. In the retrospective chart review, the authors were limited to information documented in the medical record, which might not accurately reflect the AMA discharge process. As they surveyed a limited number of social workers, case managers, and others who play an important role in the AMA discharge process, their data may lack varying viewpoints.
Overall, these data suggest providers at this county hospital generally agreed in principle with the best practice guidelines proposed by bioethicists for AMA discharges. In practice, however, providers were not reliably following these guidelines. Future interventions—including provider education on best practice guidelines for AMA discharge, provider involvement in policymaking, supportive templates for guiding documentation of AMA discharges, and improving access to follow-up care—will be key in improving the safety and health outcomes of AMA patients.
Acknowledgments
The authors thank Kelly Aguilar, Kethia Chheng, Irene Yen, and the Research Advancement and Coordination Initiative at Alameda Health System for important contributions to this project.
Disclosures
Highland Hospital Department of Medicine internal grant 2015.23 helped fund this research. A portion of the data was presented as a poster at the University of California San Francisco Health Disparities Symposium; October 2015; San Francisco, CA. Two posters from the data were presented at Hospital Medicine 2016, March 2016; San Diego, CA.
1. Southern WN, Nahvi S, Arnsten JH. Increased risk of mortality and readmission among patients discharged against medical advice. Am J Med. 2012;125(6):594-602. PubMed
2. Stranges E, Wier L, Merrill C, Steiner C. Hospitalizations in which Patients Leave the Hospital against Medical Advice (AMA), 2007. HCUP Statistical Brief #78. August 2009. Agency for Healthcare Research and Quality, Rockville, MD. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb78.pdf. Accessed November 30, 2016. PubMed
3. Devitt PJ, Devitt AC, Dewan M. Does identifying a discharge as “against medical advice” confer legal protection? J Fam Pract. 2000;49(3):224-227. PubMed
4. O’Hara D, Hart W, McDonald I. Leaving hospital against medical advice. J Qual Clin Pract. 1996;16(3):157-164. PubMed
5. Ibrahim SA, Kwoh CK, Krishnan E. Factors associated with patients who leave acute-care hospitals against medical advice. Am J Public Health. 2007;97(12):2204-2208. PubMed
6. Clark MA, Abbott JT, Adyanthaya T. Ethics seminars: a best-practice approach to navigating the against-medical-advice discharge. Acad Emerg Med. 2014;21(9):1050-1057. PubMed
7. Windish DM, Ratanawongsa N. Providers’ perceptions of relationships and professional roles when caring for patients who leave the hospital against medical advice. J Gen Intern Med. 2008;23(10):1698-1707. PubMed
8. Edwards J, Markert R, Bricker D. Discharge against medical advice: how often do we intervene? J Hosp Med. 2013;8(10):574-577. PubMed
9. Alfandre DJ. “I’m going home”: discharges against medical advice. Mayo Clin Proc. 2009;84(3):255-260. PubMed
10. Katzenellenbogen JM, Sanfilippo FM, Hobbs MS, et al. Voting with their feet—predictors of discharge against medical advice in Aboriginal and non-Aboriginal ischaemic heart disease inpatients in Western Australia: an analytic study using data linkage. BMC Health Serv Res. 2013;13:330. PubMed
11. Ti L, Milloy MJ, Buxton J, et al. Factors associated with leaving hospital against medical advice among people who use illicit drugs in Vancouver, Canada. PLoS One. 2015;10(10):e0141594. PubMed
12. Yong TY, Fok JS, Hakendorf P, Ben-Tovim D, Thompson CH, Li JY. Characteristics and outcomes of discharges against medical advice among hospitalised patients. Intern Med J. 2013;43(7):798-802. PubMed
13. Tabatabaei SM, Sargazi Moakhar Z, Behmanesh Pour F, Shaare Mollashahi S, Zaboli M. Hospitalized pregnant women who leave against medical advice: attributes and reasons. Matern Child Health J. 2016;20(1):128-138. PubMed
14. Aliyu ZY. Discharge against medical advice: sociodemographic, clinical and financial perspectives. Int J Clin Pract. 2002;56(5):325-327. PubMed
15. Ti L, Ti L. Leaving the hospital against medical advice among people who use illicit drugs: a systematic review. Am J Public Health. 2015;105(12):e53-e59. PubMed
16. Targum SD, Capodanno AE, Hoffman HA, Foudraine C. An intervention to reduce the rate of hospital discharges against medical advice. Am J Psychiatry. 1982;139(5):657-659. PubMed
17. Choi M, Kim H, Qian H, Palepu A. Readmission rates of patients discharged against medical advice: a matched cohort study. PLoS One. 2011;6(9):e24459. PubMed
18. Glasgow JM, Vaughn-Sarrazin M, Kaboli PJ. Leaving against medical advice (AMA): risk of 30-day mortality and hospital readmission. J Gen Intern Med. 2010;25(9):926-929. PubMed
19. Garland A, Ramsey CD, Fransoo R, et al. Rates of readmission and death associated with leaving hospital against medical advice: a population-based study. CMAJ. 2013;185(14):1207-1214. PubMed
20. Hwang SW, Li J, Gupta R, Chien V, Martin RE. What happens to patients who leave hospital against medical advice? CMAJ. 2003;168(4):417-420. PubMed
21. Onukwugha E, Mullins CD, Loh FE, Saunders E, Shaya FT, Weir MR. Readmissions after unauthorized discharges in the cardiovascular setting. Med Care. 2011;49(2):215-224. PubMed
22. Alfandre D. Reconsidering against medical advice discharges: embracing patient-centeredness to promote high quality care and a renewed research agenda. J Gen Intern Med. 2013;28(12):1657-1662. PubMed
23. Berger JT. Discharge against medical advice: ethical considerations and professional obligations. J Hosp Med. 2008;3(5):403-408. PubMed
24. Franks P, Meldrum S, Fiscella K. Discharges against medical advice: are race/ethnicity predictors? J Gen Intern Med. 2006;21(9):955-960. PubMed
25. Hicks LS, Ayanian JZ, Orav EJ, et al. Is hospital service associated with racial and ethnic disparities in experiences with hospital care? Am J Med. 2005;118(5):529-535. PubMed
26. Hicks LS, Tovar DA, Orav EJ, Johnson PA. Experiences with hospital care: perspectives of black and Hispanic patients. J Gen Intern Med. 2008;23(8):1234-1240. PubMed
27. McCreaddie M, Lyons I, Watt D, et al. Routines and rituals: a grounded theory of the pain management of drug users in acute care settings. J Clin Nurs. 2010;19(19-20):2730-2740. PubMed
28. Carroll IR, Angst MS, Clark JD. Management of perioperative pain in patients chronically consuming opioids. Reg Anesth Pain Med. 2004;29(6):576-591. PubMed
29. Chan AC, Palepu A, Guh DP, et al. HIV-positive injection drug users who leave the hospital against medical advice: the mitigating role of methadone and social support. J Acquir Immune Defic Syndr. 2004;35(1):56-59. PubMed
30. Levy F, Mareiniss DP, Iacovelli C. The importance of a proper against-medical-advice (AMA) discharge: how signing out AMA may create significant liability protection for providers. J Emerg Med. 2012;43(3):516-520. PubMed
31. Ashbrook L, Mourad M, Sehgal N. Communicating discharge instructions to patients: a survey of nurse, intern, and hospitalist practices. J Hosp Med. 2013;8(1):36-41. PubMed
32. Joint Commission on Accreditation of Healthcare Organizations. Title 22, California Code of Regulations, §70707.3.
Patients leave the hospital against medical advice (AMA) for a variety of reasons. The AMA rate is approximately 1% nationally but substantially higher at safety-net hospitals and has rapidly increased over the past decade.1-5 The principle that patients have the right to make choices about their healthcare, up to and including whether to leave the hospital against the advice of medical staff, is well-established law and a foundation of medical ethics.6 In practice, however, AMA discharges are often emotionally charged for both patients and providers, and, in the high-stress setting of AMA discharge, providers may be confused about their roles.7-9
The demographics of patients who leave AMA have been well described. Compared with conventionally discharged patients, AMA patients are younger, more likely to be male, and more likely a marginalized ethnic or racial minority.10-14 Patients with mental illnesses and addiction issues are overrepresented in AMA discharges, and complicated capacity assessments and limited resources may strain providers.7,8,15,16 Studies have repeatedly shown higher rates of readmission and mortality for AMA patients than for conventionally discharged patients.17-21 Whether AMA discharge is a marker for other prognostic factors that bode poorly for patients or contributes to negative outcomes, data suggest this group of patients is vulnerable, having mortality rates up to 40% higher 1 year after discharge, relative to conventionally discharged patients.12
Several models of standardized best practice approaches for AMA have been proposed by bioethicists.6,22,23 Although details of these approaches vary, all involve assessing the patient’s decision-making capacity, clarifying the risks of AMA discharge, addressing factors that might be prompting the discharge, formulating an alternative outpatient treatment plan or “next best” option, and documenting extensively. A recent study found patients often gave advance warning of an AMA discharge, but physicians rarely prepared by arranging follow-up care.8 The investigators hypothesized that providers might not have known what they were permitted to arrange for AMA patients, or might have thought that providing “second best” options went against their principles. The investigators noted that nurses might have become aware of AMA risk sooner than physicians did but could not act on this awareness by preparing medications and arranging follow-up.
Translating models of best practice care for AMA patients into clinical practice requires buy-in from bedside providers, not just bioethicists. Given the study findings that providers have misconceptions about their roles in the AMA discharge,7 it is prudent to investigate providers’ current practices, beliefs, and concerns about AMA discharges before introducing a new approach.
The present authors conducted a mixed-methods cross-sectional study of the state of AMA discharges at Highland Hospital (Oakland, California), a 236-bed county hospital and trauma center serving a primarily underserved urban patient population. The aim of this study was to assess current provider practices for AMA discharges and provider perceptions and knowledge about AMA discharges, ultimately to help direct future educational interventions with medical providers or hospital policy changes needed to improve the quality of AMA discharges.
METHODS
Phase 1 of this study involved identifying AMA patients through a review of data from Highland Hospital’s electronic medical records for 2014. These data included discharge status (eg, AMA vs other discharge types). The hospital’s floor clerk distinguishes between absent without official leave (AWOL; the patient leaves without notifying a provider) and AMA discharge. Discharges designated AWOL were excluded from the analyses.
In phase 2, a structured chart review (Appendix A) was performed for all patients identified during phase 1 as being discharged AMA in 2014. In these reviews, further assessment was made of patient and visit characteristics in hospitalizations that ended in AMA discharge, and of providers’ documentation of AMA discharges—that is, whether several factors were documented (capacity; predischarge indication that patient might leave AMA; reason for AMA; and indications that discharge medications, transportation, and follow-up were arranged). These visit factors were reviewed because the literature has identified them as being important markers for AMA discharge safety.6,8 Two research assistants, under the guidance of Dr. Stearns, reviewed the charts. To ensure agreement across chart reviews with respect to subjective questions (eg, whether capacity was adequately documented), the group reviewed the first 10 consecutive charts together; there was full agreement on how to classify the data of interest. Throughout the study, whenever a research assistant asked how to classify particular patient data, Dr. Stearns reviewed the data, and the research team made a decision together. Additional data, for AMA patients and for all patients admitted to Highland Hospital, were obtained from the hospital’s data warehouse, which pools data from within the health system.
Phase 3 involved surveying healthcare providers who were involved in patient care on the internal medicine and trauma surgery services at the hospital. These providers were selected because chart review revealed that the vast majority of patients who left AMA in 2014 were on one of these services. Surveys (Appendix B) asked participant providers to identify their role at the hospital, to provide a self-assessment of competence in various aspects of AMA discharge, to voice opinions about provider responsibilities in arranging follow-up for AMA patients, and to make suggestions about the AMA process. The authors designed these surveys, which included questions about aspects of care that have been highlighted in the AMA discharge literature as being important for AMA discharge safety.6,8,22,23 Surveys were distributed to providers at internal medicine and trauma surgery department meetings and nursing conferences. Data (without identifying information) were analyzed, and survey responses kept anonymous.
The Alameda Health System Institutional Review Board approved this project. Providers were given the option of writing their name and contact information at the top of the survey in order to be entered into a drawing to receive a prize for completion.
We performed statistical analyses of the patient charts and physician survey data using Stata (version 14.0, Stata Corp., College Station, Texas). We analyzed both patient- and encounter-level data. In demographic analyses, this approach prevented duplicate counting of patients who left AMA multiple times. Patient-level analyses compared the demographic characteristics of AMA patients and patients discharged conventionally from the hospital in 2014. In addition, patients with either 1 or multiple AMA discharges were compared to identify characteristics that might be linked to highest risk of recurrent AMA discharge in the hope that early identification of these patients might facilitate providers’ early awareness and preparation for follow-up care or hospitalization alternatives. We used ANOVAs for continuous variables and tests of proportions for categorical variables. On the encounter level, analyses examined data about each admission (eg, AMA forms signed, follow-up arrangements made, capacity documented, etc.) for all AMA discharges. We employed chi square tests to identify variations in healthcare provider survey responses. A P value < 0.05 was used as the significance cut-off point.
Staged logistic regression analyses, adjusted for demographic characteristics, were performed to assess the association between risk of leaving AMA (yes or no) and demographic characteristics and the association between risk of leaving AMA more than once (yes or no) and health-related characteristics.
RESULTS
Demographic, Clinical, and Utilization Characteristics
Of the 12,036 Highland Hospital admissions in 2014, 319 (2.7%) ended with an AMA discharge. Of the 8207 individual patients discharged, 268 left AMA once, and 29 left AMA multiple times. Further review of the Admissions, Discharges, and Transfers Report generated from the electronic medical record revealed that 15 AWOL discharges were misclassified as AMA discharges.
Compared with patients discharged conventionally, AMA patients were significantly younger; more likely to be male, to self-identify as Black/African American, and to be English-speaking; and less likely to self-identify as Asian/Pacific Islander or Hispanic/Latino or to be Chinese- or Spanish-speaking (Table 1). They were also more likely than all patients admitted to Highland to be homeless (15.7% vs 8.7%; P < 0.01). Multivariate regression analysis revealed persistent age and sex disparities, but racial disparities were mitigated in adjusted analyses (Appendix C). Language disparities persisted only for Spanish speakers, who had a significantly lower rate of AMA discharge, even in adjusted analyses.

The majority of AMA patients were on the internal medicine service (63.5%) or the trauma surgery service (24.8%). Regarding admission diagnosis, 17.2% of AMA patients were admitted for infections, 5.0% for drug or alcohol intoxication or withdrawal, 38.9% for acute noninfectious illnesses, 16.7% for decompensation of chronic disease, 18.4% for injuries or trauma, and 3.8% for pregnancy complications or labor. Compared with patients who left AMA once, patients who left AMA multiple times had higher rates of heavy alcohol use (53.9% vs 30.9%; P = 0.01) and illicit drug use (88.5% vs 53.7%; P < 0.001) (Table 2). In multivariate analyses, the increased odds of leaving AMA more than once persisted for current heavy illicit drug users compared with patients who had never engaged in illicit drug use.
Discharge Characteristics and Documentation
Providers documented a patient’s plan to leave AMA before actual discharge 17.3% of the time. The documented plan to leave had to indicate that the patient was actually considering leaving. For example, “Patient is eager to go home” was not enough to qualify as a plan, but “Patient is thinking of leaving” qualified. For 84.3% of AMA discharges, the hospital’s AMA form was signed and was included in the medical record. Documentation showed that medications were prescribed for AMA patients 21.4% of the time, follow-up was arranged 25.7% of the time, and follow-up was pending arrangement 14.8% of the time. The majority of AMA patients (71.4%) left during daytime hours. In 29.6% of AMA discharges, providers documented AMA patients had decision-making capacity.
Readmission After AMA Discharge
Of the 268 AMA patients, 67.7% were not readmitted within the 6 months after AMA, 24.5% had 1 or 2 readmissions, and the rest had 3 or more readmissions (1 patient had 15). In addition, 35.8% returned to the emergency department within 30 days, and 16.4% were readmitted within 30 days. In 2014, the hospital’s overall 30-day readmission rate was 10.8%. Of the patients readmitted within 6 months after AMA, 23.5% left AMA again at the next visit, 9.4% left AWOL, and 67.1% were discharged conventionally.
Drivers of Premature Discharge
Qualitative analysis of the 35.5% of patient charts documenting a reason for leaving the hospital revealed 3 broad, interrelated themes (Figure 1). The first theme, dissatisfaction with hospital care, included chart notations such as “His wife couldn’t sleep in the hospital room” and “Not satisfied with all-liquid diet.” The second theme, urgent personal issues, included comments such as “He has a very important court date for his children” and “He needed to take care of immigration forms.” The third theme, mental health and substance abuse issues, included notations such as “He wants to go smoke” and “Severe anxiety and prison flashbacks.”
Provider Self-Assessment and Beliefs
The survey was completed by 178 healthcare providers: 49.4% registered nurses, 19.1% trainee physicians, 20.8% attending physicians, and 10.7% other providers, including chaplains, social workers, and clerks. Regarding self-assessment of competency in AMA discharges, 94% of providers agreed they were comfortable assessing capacity, and 94% agreed they were comfortable talking with patients about the risks of leaving AMA (Figure 2). Nurses were more likely than trainee physicians to agree they knew what to do for patients who lacked capacity (74% vs 49%; P = 0.02). Most providers (70%) agreed they usually knew why their patients were leaving AMA; in this self-assessment, there were no significant differences between types of providers.
Regarding follow-up, attending physicians and trainee physicians demonstrated more agreement than nurses that AMA patients should receive medications and follow-up (94% and 84% vs 64%; P < 0.05). Nurses were more likely than attending physicians to say patients should lose their rights to hospital follow-up because of leaving AMA (38% vs 6%; P < 0.01). A minority of providers (37%) agreed transportation should be arranged. Addiction was the most common driver of AMA discharge (35%), followed by familial obligations (19%), dissatisfaction with hospital care (16%), and financial concerns (15%).
DISCUSSION
The demographic characteristics of AMA patients in this study are similar to those identified in other studies, showing overrepresentation of young male patients.12,14 Homeless patients were also overrepresented in the AMA discharge population at Highland Hospital—a finding that has not been consistently reported in prior studies, and that warrants further examination. In adjusted analyses, Spanish speakers had a lower rate of AMA discharge, and there were no racial variations. This is consistent with another study’s finding: that racial disparities in AMA discharge rates were largely attributable to confounders.24 Language differences may result from failure of staff to fully explain the option of AMA discharge to non-English speakers, or from fear of immigration consequences after AMA discharge. Further investigation of patient experiences is needed to identify factors that contribute to demographic variations in AMA discharge rates.25,26
Of the patients who left AMA multiple times, nearly all were actively using illicit drugs. In a recent study conducted at a safety-net hospital in Vancouver, Canada, 43% of patients with illicit drug use and at least 1 hospitalization left AMA at least once during the 6-year study period.11 Many factors might explain this correlation—addiction itself, poor pain control for patients with addiction issues, fears about incarceration, and poor treatment of drug users by healthcare staff.15 Although the medical literature highlights deficits in pain control for patients addicted to opiates, proposed solutions are sparse and focus on perioperative pain control and physician prescribing practices.27,28 At safety-net hospitals in which addiction is a factor in many hospitalizations, there is opportunity for new research in inpatient pain control for patients with substance dependence. In addition, harm reduction strategies—such as methadone maintenance for hospitalized patients with opiate dependence and abscess clinics as hospitalization alternatives for injection-associated infection treatment—may be key in improving safety for patients.11,15,29
Comparing the provider survey and chart review results highlights discordance between provider beliefs and clinical practice. Healthcare providers at Highland Hospital considered themselves competent in assessing capacity and talking with patients about the risks of AMA discharge. In practice, however, capacity was documented in less than a third of AMA discharges. Although the majority of providers thought medications and follow-up should be arranged for patients, arrangements were seldom made. This may be partially attributable to limited resources for making these arrangements. Average time to “third next available” primary care appointment within the county health system that includes Highland was 44.6 days for established patients during the period of study; for new primary care patients, the average wait for an appointment was 2 to 3 months. Highland has a same-day clinic, but inpatient providers are discouraged from using it as a postdischarge clinic for patients who would be better served in primary care. Medications and transportation are easily arranged during daytime hours but are not immediately available at night. In addition, some of this discrepancy may be attributable to the limited documentation rather than to provider failure to achieve their own benchmarks of quality care for AMA patients.
Documentation in AMA discharges is key for multiple reasons. Most AMA patients in this study signed an AMA form, and it could be that the rate of documenting decision-making capacity was low because providers thought a signed AMA form was adequate documentation of capacity and informed consent. In numerous court cases, however, these forms were found to be insufficient evidence of informed consent (lacking other supportive documentation) and possibly to go against the public good.30 In addition, high rates of repeat emergency department visits and readmissions for AMA patients, demonstrated here and in other studies, highlight the importance of careful documentation in informing subsequent providers about hospital returnees’ ongoing issues.17-19
This study also demonstrated differences between nurses and physicians in their beliefs about arranging follow-up for AMA patients. Nurses were less likely than physicians to think follow-up arrangements should be made for AMA patients and more likely to say these patients should lose the right to follow-up because of the AMA discharge. For conventional discharges, nurses provide patients with significantly more discharge education than interns or hospitalists do.31 This discrepancy highlights an urgent need for the education and involvement of nurses as stakeholders in the challenging AMA discharge process. Although the percentage of physicians who thought they were not obligated to provide medications and arrange follow-up for AMA patients was lower than the percentage of nurses, these beliefs contradict best practice guidelines for AMA discharges,22,23 and this finding calls attention to the need for interventions to improve adherence to professional and ethical guidelines in this aspect of clinical practice.
Providers showed a lack of familiarity with practice guidelines regarding certain aspects of the AMA discharge process. For example, most providers thought they should not have to arrange transportation for AMA patients, even though both the California Hospital Association Guidelines and the Highland Hospital internal policy on AMA discharges recommend arranging appropriate transportation.32 This finding suggests a need for educational interventions to ensure providers are informed about state and hospital policies, and a need to include both physicians and nurses in policymaking so theory can be tied to practice.
This study was limited to a single center with healthcare provider and patient populations that might not be generalizable to other settings. In the retrospective chart review, the authors were limited to information documented in the medical record, which might not accurately reflect the AMA discharge process. As they surveyed a limited number of social workers, case managers, and others who play an important role in the AMA discharge process, their data may lack varying viewpoints.
Overall, these data suggest providers at this county hospital generally agreed in principle with the best practice guidelines proposed by bioethicists for AMA discharges. In practice, however, providers were not reliably following these guidelines. Future interventions—including provider education on best practice guidelines for AMA discharge, provider involvement in policymaking, supportive templates for guiding documentation of AMA discharges, and improving access to follow-up care—will be key in improving the safety and health outcomes of AMA patients.
Acknowledgments
The authors thank Kelly Aguilar, Kethia Chheng, Irene Yen, and the Research Advancement and Coordination Initiative at Alameda Health System for important contributions to this project.
Disclosures
Highland Hospital Department of Medicine internal grant 2015.23 helped fund this research. A portion of the data was presented as a poster at the University of California San Francisco Health Disparities Symposium; October 2015; San Francisco, CA. Two posters from the data were presented at Hospital Medicine 2016, March 2016; San Diego, CA.
Patients leave the hospital against medical advice (AMA) for a variety of reasons. The AMA rate is approximately 1% nationally but substantially higher at safety-net hospitals and has rapidly increased over the past decade.1-5 The principle that patients have the right to make choices about their healthcare, up to and including whether to leave the hospital against the advice of medical staff, is well-established law and a foundation of medical ethics.6 In practice, however, AMA discharges are often emotionally charged for both patients and providers, and, in the high-stress setting of AMA discharge, providers may be confused about their roles.7-9
The demographics of patients who leave AMA have been well described. Compared with conventionally discharged patients, AMA patients are younger, more likely to be male, and more likely a marginalized ethnic or racial minority.10-14 Patients with mental illnesses and addiction issues are overrepresented in AMA discharges, and complicated capacity assessments and limited resources may strain providers.7,8,15,16 Studies have repeatedly shown higher rates of readmission and mortality for AMA patients than for conventionally discharged patients.17-21 Whether AMA discharge is a marker for other prognostic factors that bode poorly for patients or contributes to negative outcomes, data suggest this group of patients is vulnerable, having mortality rates up to 40% higher 1 year after discharge, relative to conventionally discharged patients.12
Several models of standardized best practice approaches for AMA have been proposed by bioethicists.6,22,23 Although details of these approaches vary, all involve assessing the patient’s decision-making capacity, clarifying the risks of AMA discharge, addressing factors that might be prompting the discharge, formulating an alternative outpatient treatment plan or “next best” option, and documenting extensively. A recent study found patients often gave advance warning of an AMA discharge, but physicians rarely prepared by arranging follow-up care.8 The investigators hypothesized that providers might not have known what they were permitted to arrange for AMA patients, or might have thought that providing “second best” options went against their principles. The investigators noted that nurses might have become aware of AMA risk sooner than physicians did but could not act on this awareness by preparing medications and arranging follow-up.
Translating models of best practice care for AMA patients into clinical practice requires buy-in from bedside providers, not just bioethicists. Given the study findings that providers have misconceptions about their roles in the AMA discharge,7 it is prudent to investigate providers’ current practices, beliefs, and concerns about AMA discharges before introducing a new approach.
The present authors conducted a mixed-methods cross-sectional study of the state of AMA discharges at Highland Hospital (Oakland, California), a 236-bed county hospital and trauma center serving a primarily underserved urban patient population. The aim of this study was to assess current provider practices for AMA discharges and provider perceptions and knowledge about AMA discharges, ultimately to help direct future educational interventions with medical providers or hospital policy changes needed to improve the quality of AMA discharges.
METHODS
Phase 1 of this study involved identifying AMA patients through a review of data from Highland Hospital’s electronic medical records for 2014. These data included discharge status (eg, AMA vs other discharge types). The hospital’s floor clerk distinguishes between absent without official leave (AWOL; the patient leaves without notifying a provider) and AMA discharge. Discharges designated AWOL were excluded from the analyses.
In phase 2, a structured chart review (Appendix A) was performed for all patients identified during phase 1 as being discharged AMA in 2014. In these reviews, further assessment was made of patient and visit characteristics in hospitalizations that ended in AMA discharge, and of providers’ documentation of AMA discharges—that is, whether several factors were documented (capacity; predischarge indication that patient might leave AMA; reason for AMA; and indications that discharge medications, transportation, and follow-up were arranged). These visit factors were reviewed because the literature has identified them as being important markers for AMA discharge safety.6,8 Two research assistants, under the guidance of Dr. Stearns, reviewed the charts. To ensure agreement across chart reviews with respect to subjective questions (eg, whether capacity was adequately documented), the group reviewed the first 10 consecutive charts together; there was full agreement on how to classify the data of interest. Throughout the study, whenever a research assistant asked how to classify particular patient data, Dr. Stearns reviewed the data, and the research team made a decision together. Additional data, for AMA patients and for all patients admitted to Highland Hospital, were obtained from the hospital’s data warehouse, which pools data from within the health system.
Phase 3 involved surveying healthcare providers who were involved in patient care on the internal medicine and trauma surgery services at the hospital. These providers were selected because chart review revealed that the vast majority of patients who left AMA in 2014 were on one of these services. Surveys (Appendix B) asked participant providers to identify their role at the hospital, to provide a self-assessment of competence in various aspects of AMA discharge, to voice opinions about provider responsibilities in arranging follow-up for AMA patients, and to make suggestions about the AMA process. The authors designed these surveys, which included questions about aspects of care that have been highlighted in the AMA discharge literature as being important for AMA discharge safety.6,8,22,23 Surveys were distributed to providers at internal medicine and trauma surgery department meetings and nursing conferences. Data (without identifying information) were analyzed, and survey responses kept anonymous.
The Alameda Health System Institutional Review Board approved this project. Providers were given the option of writing their name and contact information at the top of the survey in order to be entered into a drawing to receive a prize for completion.
We performed statistical analyses of the patient charts and physician survey data using Stata (version 14.0, Stata Corp., College Station, Texas). We analyzed both patient- and encounter-level data. In demographic analyses, this approach prevented duplicate counting of patients who left AMA multiple times. Patient-level analyses compared the demographic characteristics of AMA patients and patients discharged conventionally from the hospital in 2014. In addition, patients with either 1 or multiple AMA discharges were compared to identify characteristics that might be linked to highest risk of recurrent AMA discharge in the hope that early identification of these patients might facilitate providers’ early awareness and preparation for follow-up care or hospitalization alternatives. We used ANOVAs for continuous variables and tests of proportions for categorical variables. On the encounter level, analyses examined data about each admission (eg, AMA forms signed, follow-up arrangements made, capacity documented, etc.) for all AMA discharges. We employed chi square tests to identify variations in healthcare provider survey responses. A P value < 0.05 was used as the significance cut-off point.
Staged logistic regression analyses, adjusted for demographic characteristics, were performed to assess the association between risk of leaving AMA (yes or no) and demographic characteristics and the association between risk of leaving AMA more than once (yes or no) and health-related characteristics.
RESULTS
Demographic, Clinical, and Utilization Characteristics
Of the 12,036 Highland Hospital admissions in 2014, 319 (2.7%) ended with an AMA discharge. Of the 8207 individual patients discharged, 268 left AMA once, and 29 left AMA multiple times. Further review of the Admissions, Discharges, and Transfers Report generated from the electronic medical record revealed that 15 AWOL discharges were misclassified as AMA discharges.
Compared with patients discharged conventionally, AMA patients were significantly younger; more likely to be male, to self-identify as Black/African American, and to be English-speaking; and less likely to self-identify as Asian/Pacific Islander or Hispanic/Latino or to be Chinese- or Spanish-speaking (Table 1). They were also more likely than all patients admitted to Highland to be homeless (15.7% vs 8.7%; P < 0.01). Multivariate regression analysis revealed persistent age and sex disparities, but racial disparities were mitigated in adjusted analyses (Appendix C). Language disparities persisted only for Spanish speakers, who had a significantly lower rate of AMA discharge, even in adjusted analyses.

The majority of AMA patients were on the internal medicine service (63.5%) or the trauma surgery service (24.8%). Regarding admission diagnosis, 17.2% of AMA patients were admitted for infections, 5.0% for drug or alcohol intoxication or withdrawal, 38.9% for acute noninfectious illnesses, 16.7% for decompensation of chronic disease, 18.4% for injuries or trauma, and 3.8% for pregnancy complications or labor. Compared with patients who left AMA once, patients who left AMA multiple times had higher rates of heavy alcohol use (53.9% vs 30.9%; P = 0.01) and illicit drug use (88.5% vs 53.7%; P < 0.001) (Table 2). In multivariate analyses, the increased odds of leaving AMA more than once persisted for current heavy illicit drug users compared with patients who had never engaged in illicit drug use.
Discharge Characteristics and Documentation
Providers documented a patient’s plan to leave AMA before actual discharge 17.3% of the time. The documented plan to leave had to indicate that the patient was actually considering leaving. For example, “Patient is eager to go home” was not enough to qualify as a plan, but “Patient is thinking of leaving” qualified. For 84.3% of AMA discharges, the hospital’s AMA form was signed and was included in the medical record. Documentation showed that medications were prescribed for AMA patients 21.4% of the time, follow-up was arranged 25.7% of the time, and follow-up was pending arrangement 14.8% of the time. The majority of AMA patients (71.4%) left during daytime hours. In 29.6% of AMA discharges, providers documented AMA patients had decision-making capacity.
Readmission After AMA Discharge
Of the 268 AMA patients, 67.7% were not readmitted within the 6 months after AMA, 24.5% had 1 or 2 readmissions, and the rest had 3 or more readmissions (1 patient had 15). In addition, 35.8% returned to the emergency department within 30 days, and 16.4% were readmitted within 30 days. In 2014, the hospital’s overall 30-day readmission rate was 10.8%. Of the patients readmitted within 6 months after AMA, 23.5% left AMA again at the next visit, 9.4% left AWOL, and 67.1% were discharged conventionally.
Drivers of Premature Discharge
Qualitative analysis of the 35.5% of patient charts documenting a reason for leaving the hospital revealed 3 broad, interrelated themes (Figure 1). The first theme, dissatisfaction with hospital care, included chart notations such as “His wife couldn’t sleep in the hospital room” and “Not satisfied with all-liquid diet.” The second theme, urgent personal issues, included comments such as “He has a very important court date for his children” and “He needed to take care of immigration forms.” The third theme, mental health and substance abuse issues, included notations such as “He wants to go smoke” and “Severe anxiety and prison flashbacks.”
Provider Self-Assessment and Beliefs
The survey was completed by 178 healthcare providers: 49.4% registered nurses, 19.1% trainee physicians, 20.8% attending physicians, and 10.7% other providers, including chaplains, social workers, and clerks. Regarding self-assessment of competency in AMA discharges, 94% of providers agreed they were comfortable assessing capacity, and 94% agreed they were comfortable talking with patients about the risks of leaving AMA (Figure 2). Nurses were more likely than trainee physicians to agree they knew what to do for patients who lacked capacity (74% vs 49%; P = 0.02). Most providers (70%) agreed they usually knew why their patients were leaving AMA; in this self-assessment, there were no significant differences between types of providers.
Regarding follow-up, attending physicians and trainee physicians demonstrated more agreement than nurses that AMA patients should receive medications and follow-up (94% and 84% vs 64%; P < 0.05). Nurses were more likely than attending physicians to say patients should lose their rights to hospital follow-up because of leaving AMA (38% vs 6%; P < 0.01). A minority of providers (37%) agreed transportation should be arranged. Addiction was the most common driver of AMA discharge (35%), followed by familial obligations (19%), dissatisfaction with hospital care (16%), and financial concerns (15%).
DISCUSSION
The demographic characteristics of AMA patients in this study are similar to those identified in other studies, showing overrepresentation of young male patients.12,14 Homeless patients were also overrepresented in the AMA discharge population at Highland Hospital—a finding that has not been consistently reported in prior studies, and that warrants further examination. In adjusted analyses, Spanish speakers had a lower rate of AMA discharge, and there were no racial variations. This is consistent with another study’s finding: that racial disparities in AMA discharge rates were largely attributable to confounders.24 Language differences may result from failure of staff to fully explain the option of AMA discharge to non-English speakers, or from fear of immigration consequences after AMA discharge. Further investigation of patient experiences is needed to identify factors that contribute to demographic variations in AMA discharge rates.25,26
Of the patients who left AMA multiple times, nearly all were actively using illicit drugs. In a recent study conducted at a safety-net hospital in Vancouver, Canada, 43% of patients with illicit drug use and at least 1 hospitalization left AMA at least once during the 6-year study period.11 Many factors might explain this correlation—addiction itself, poor pain control for patients with addiction issues, fears about incarceration, and poor treatment of drug users by healthcare staff.15 Although the medical literature highlights deficits in pain control for patients addicted to opiates, proposed solutions are sparse and focus on perioperative pain control and physician prescribing practices.27,28 At safety-net hospitals in which addiction is a factor in many hospitalizations, there is opportunity for new research in inpatient pain control for patients with substance dependence. In addition, harm reduction strategies—such as methadone maintenance for hospitalized patients with opiate dependence and abscess clinics as hospitalization alternatives for injection-associated infection treatment—may be key in improving safety for patients.11,15,29
Comparing the provider survey and chart review results highlights discordance between provider beliefs and clinical practice. Healthcare providers at Highland Hospital considered themselves competent in assessing capacity and talking with patients about the risks of AMA discharge. In practice, however, capacity was documented in less than a third of AMA discharges. Although the majority of providers thought medications and follow-up should be arranged for patients, arrangements were seldom made. This may be partially attributable to limited resources for making these arrangements. Average time to “third next available” primary care appointment within the county health system that includes Highland was 44.6 days for established patients during the period of study; for new primary care patients, the average wait for an appointment was 2 to 3 months. Highland has a same-day clinic, but inpatient providers are discouraged from using it as a postdischarge clinic for patients who would be better served in primary care. Medications and transportation are easily arranged during daytime hours but are not immediately available at night. In addition, some of this discrepancy may be attributable to the limited documentation rather than to provider failure to achieve their own benchmarks of quality care for AMA patients.
Documentation in AMA discharges is key for multiple reasons. Most AMA patients in this study signed an AMA form, and it could be that the rate of documenting decision-making capacity was low because providers thought a signed AMA form was adequate documentation of capacity and informed consent. In numerous court cases, however, these forms were found to be insufficient evidence of informed consent (lacking other supportive documentation) and possibly to go against the public good.30 In addition, high rates of repeat emergency department visits and readmissions for AMA patients, demonstrated here and in other studies, highlight the importance of careful documentation in informing subsequent providers about hospital returnees’ ongoing issues.17-19
This study also demonstrated differences between nurses and physicians in their beliefs about arranging follow-up for AMA patients. Nurses were less likely than physicians to think follow-up arrangements should be made for AMA patients and more likely to say these patients should lose the right to follow-up because of the AMA discharge. For conventional discharges, nurses provide patients with significantly more discharge education than interns or hospitalists do.31 This discrepancy highlights an urgent need for the education and involvement of nurses as stakeholders in the challenging AMA discharge process. Although the percentage of physicians who thought they were not obligated to provide medications and arrange follow-up for AMA patients was lower than the percentage of nurses, these beliefs contradict best practice guidelines for AMA discharges,22,23 and this finding calls attention to the need for interventions to improve adherence to professional and ethical guidelines in this aspect of clinical practice.
Providers showed a lack of familiarity with practice guidelines regarding certain aspects of the AMA discharge process. For example, most providers thought they should not have to arrange transportation for AMA patients, even though both the California Hospital Association Guidelines and the Highland Hospital internal policy on AMA discharges recommend arranging appropriate transportation.32 This finding suggests a need for educational interventions to ensure providers are informed about state and hospital policies, and a need to include both physicians and nurses in policymaking so theory can be tied to practice.
This study was limited to a single center with healthcare provider and patient populations that might not be generalizable to other settings. In the retrospective chart review, the authors were limited to information documented in the medical record, which might not accurately reflect the AMA discharge process. As they surveyed a limited number of social workers, case managers, and others who play an important role in the AMA discharge process, their data may lack varying viewpoints.
Overall, these data suggest providers at this county hospital generally agreed in principle with the best practice guidelines proposed by bioethicists for AMA discharges. In practice, however, providers were not reliably following these guidelines. Future interventions—including provider education on best practice guidelines for AMA discharge, provider involvement in policymaking, supportive templates for guiding documentation of AMA discharges, and improving access to follow-up care—will be key in improving the safety and health outcomes of AMA patients.
Acknowledgments
The authors thank Kelly Aguilar, Kethia Chheng, Irene Yen, and the Research Advancement and Coordination Initiative at Alameda Health System for important contributions to this project.
Disclosures
Highland Hospital Department of Medicine internal grant 2015.23 helped fund this research. A portion of the data was presented as a poster at the University of California San Francisco Health Disparities Symposium; October 2015; San Francisco, CA. Two posters from the data were presented at Hospital Medicine 2016, March 2016; San Diego, CA.
1. Southern WN, Nahvi S, Arnsten JH. Increased risk of mortality and readmission among patients discharged against medical advice. Am J Med. 2012;125(6):594-602. PubMed
2. Stranges E, Wier L, Merrill C, Steiner C. Hospitalizations in which Patients Leave the Hospital against Medical Advice (AMA), 2007. HCUP Statistical Brief #78. August 2009. Agency for Healthcare Research and Quality, Rockville, MD. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb78.pdf. Accessed November 30, 2016. PubMed
3. Devitt PJ, Devitt AC, Dewan M. Does identifying a discharge as “against medical advice” confer legal protection? J Fam Pract. 2000;49(3):224-227. PubMed
4. O’Hara D, Hart W, McDonald I. Leaving hospital against medical advice. J Qual Clin Pract. 1996;16(3):157-164. PubMed
5. Ibrahim SA, Kwoh CK, Krishnan E. Factors associated with patients who leave acute-care hospitals against medical advice. Am J Public Health. 2007;97(12):2204-2208. PubMed
6. Clark MA, Abbott JT, Adyanthaya T. Ethics seminars: a best-practice approach to navigating the against-medical-advice discharge. Acad Emerg Med. 2014;21(9):1050-1057. PubMed
7. Windish DM, Ratanawongsa N. Providers’ perceptions of relationships and professional roles when caring for patients who leave the hospital against medical advice. J Gen Intern Med. 2008;23(10):1698-1707. PubMed
8. Edwards J, Markert R, Bricker D. Discharge against medical advice: how often do we intervene? J Hosp Med. 2013;8(10):574-577. PubMed
9. Alfandre DJ. “I’m going home”: discharges against medical advice. Mayo Clin Proc. 2009;84(3):255-260. PubMed
10. Katzenellenbogen JM, Sanfilippo FM, Hobbs MS, et al. Voting with their feet—predictors of discharge against medical advice in Aboriginal and non-Aboriginal ischaemic heart disease inpatients in Western Australia: an analytic study using data linkage. BMC Health Serv Res. 2013;13:330. PubMed
11. Ti L, Milloy MJ, Buxton J, et al. Factors associated with leaving hospital against medical advice among people who use illicit drugs in Vancouver, Canada. PLoS One. 2015;10(10):e0141594. PubMed
12. Yong TY, Fok JS, Hakendorf P, Ben-Tovim D, Thompson CH, Li JY. Characteristics and outcomes of discharges against medical advice among hospitalised patients. Intern Med J. 2013;43(7):798-802. PubMed
13. Tabatabaei SM, Sargazi Moakhar Z, Behmanesh Pour F, Shaare Mollashahi S, Zaboli M. Hospitalized pregnant women who leave against medical advice: attributes and reasons. Matern Child Health J. 2016;20(1):128-138. PubMed
14. Aliyu ZY. Discharge against medical advice: sociodemographic, clinical and financial perspectives. Int J Clin Pract. 2002;56(5):325-327. PubMed
15. Ti L, Ti L. Leaving the hospital against medical advice among people who use illicit drugs: a systematic review. Am J Public Health. 2015;105(12):e53-e59. PubMed
16. Targum SD, Capodanno AE, Hoffman HA, Foudraine C. An intervention to reduce the rate of hospital discharges against medical advice. Am J Psychiatry. 1982;139(5):657-659. PubMed
17. Choi M, Kim H, Qian H, Palepu A. Readmission rates of patients discharged against medical advice: a matched cohort study. PLoS One. 2011;6(9):e24459. PubMed
18. Glasgow JM, Vaughn-Sarrazin M, Kaboli PJ. Leaving against medical advice (AMA): risk of 30-day mortality and hospital readmission. J Gen Intern Med. 2010;25(9):926-929. PubMed
19. Garland A, Ramsey CD, Fransoo R, et al. Rates of readmission and death associated with leaving hospital against medical advice: a population-based study. CMAJ. 2013;185(14):1207-1214. PubMed
20. Hwang SW, Li J, Gupta R, Chien V, Martin RE. What happens to patients who leave hospital against medical advice? CMAJ. 2003;168(4):417-420. PubMed
21. Onukwugha E, Mullins CD, Loh FE, Saunders E, Shaya FT, Weir MR. Readmissions after unauthorized discharges in the cardiovascular setting. Med Care. 2011;49(2):215-224. PubMed
22. Alfandre D. Reconsidering against medical advice discharges: embracing patient-centeredness to promote high quality care and a renewed research agenda. J Gen Intern Med. 2013;28(12):1657-1662. PubMed
23. Berger JT. Discharge against medical advice: ethical considerations and professional obligations. J Hosp Med. 2008;3(5):403-408. PubMed
24. Franks P, Meldrum S, Fiscella K. Discharges against medical advice: are race/ethnicity predictors? J Gen Intern Med. 2006;21(9):955-960. PubMed
25. Hicks LS, Ayanian JZ, Orav EJ, et al. Is hospital service associated with racial and ethnic disparities in experiences with hospital care? Am J Med. 2005;118(5):529-535. PubMed
26. Hicks LS, Tovar DA, Orav EJ, Johnson PA. Experiences with hospital care: perspectives of black and Hispanic patients. J Gen Intern Med. 2008;23(8):1234-1240. PubMed
27. McCreaddie M, Lyons I, Watt D, et al. Routines and rituals: a grounded theory of the pain management of drug users in acute care settings. J Clin Nurs. 2010;19(19-20):2730-2740. PubMed
28. Carroll IR, Angst MS, Clark JD. Management of perioperative pain in patients chronically consuming opioids. Reg Anesth Pain Med. 2004;29(6):576-591. PubMed
29. Chan AC, Palepu A, Guh DP, et al. HIV-positive injection drug users who leave the hospital against medical advice: the mitigating role of methadone and social support. J Acquir Immune Defic Syndr. 2004;35(1):56-59. PubMed
30. Levy F, Mareiniss DP, Iacovelli C. The importance of a proper against-medical-advice (AMA) discharge: how signing out AMA may create significant liability protection for providers. J Emerg Med. 2012;43(3):516-520. PubMed
31. Ashbrook L, Mourad M, Sehgal N. Communicating discharge instructions to patients: a survey of nurse, intern, and hospitalist practices. J Hosp Med. 2013;8(1):36-41. PubMed
32. Joint Commission on Accreditation of Healthcare Organizations. Title 22, California Code of Regulations, §70707.3.
1. Southern WN, Nahvi S, Arnsten JH. Increased risk of mortality and readmission among patients discharged against medical advice. Am J Med. 2012;125(6):594-602. PubMed
2. Stranges E, Wier L, Merrill C, Steiner C. Hospitalizations in which Patients Leave the Hospital against Medical Advice (AMA), 2007. HCUP Statistical Brief #78. August 2009. Agency for Healthcare Research and Quality, Rockville, MD. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb78.pdf. Accessed November 30, 2016. PubMed
3. Devitt PJ, Devitt AC, Dewan M. Does identifying a discharge as “against medical advice” confer legal protection? J Fam Pract. 2000;49(3):224-227. PubMed
4. O’Hara D, Hart W, McDonald I. Leaving hospital against medical advice. J Qual Clin Pract. 1996;16(3):157-164. PubMed
5. Ibrahim SA, Kwoh CK, Krishnan E. Factors associated with patients who leave acute-care hospitals against medical advice. Am J Public Health. 2007;97(12):2204-2208. PubMed
6. Clark MA, Abbott JT, Adyanthaya T. Ethics seminars: a best-practice approach to navigating the against-medical-advice discharge. Acad Emerg Med. 2014;21(9):1050-1057. PubMed
7. Windish DM, Ratanawongsa N. Providers’ perceptions of relationships and professional roles when caring for patients who leave the hospital against medical advice. J Gen Intern Med. 2008;23(10):1698-1707. PubMed
8. Edwards J, Markert R, Bricker D. Discharge against medical advice: how often do we intervene? J Hosp Med. 2013;8(10):574-577. PubMed
9. Alfandre DJ. “I’m going home”: discharges against medical advice. Mayo Clin Proc. 2009;84(3):255-260. PubMed
10. Katzenellenbogen JM, Sanfilippo FM, Hobbs MS, et al. Voting with their feet—predictors of discharge against medical advice in Aboriginal and non-Aboriginal ischaemic heart disease inpatients in Western Australia: an analytic study using data linkage. BMC Health Serv Res. 2013;13:330. PubMed
11. Ti L, Milloy MJ, Buxton J, et al. Factors associated with leaving hospital against medical advice among people who use illicit drugs in Vancouver, Canada. PLoS One. 2015;10(10):e0141594. PubMed
12. Yong TY, Fok JS, Hakendorf P, Ben-Tovim D, Thompson CH, Li JY. Characteristics and outcomes of discharges against medical advice among hospitalised patients. Intern Med J. 2013;43(7):798-802. PubMed
13. Tabatabaei SM, Sargazi Moakhar Z, Behmanesh Pour F, Shaare Mollashahi S, Zaboli M. Hospitalized pregnant women who leave against medical advice: attributes and reasons. Matern Child Health J. 2016;20(1):128-138. PubMed
14. Aliyu ZY. Discharge against medical advice: sociodemographic, clinical and financial perspectives. Int J Clin Pract. 2002;56(5):325-327. PubMed
15. Ti L, Ti L. Leaving the hospital against medical advice among people who use illicit drugs: a systematic review. Am J Public Health. 2015;105(12):e53-e59. PubMed
16. Targum SD, Capodanno AE, Hoffman HA, Foudraine C. An intervention to reduce the rate of hospital discharges against medical advice. Am J Psychiatry. 1982;139(5):657-659. PubMed
17. Choi M, Kim H, Qian H, Palepu A. Readmission rates of patients discharged against medical advice: a matched cohort study. PLoS One. 2011;6(9):e24459. PubMed
18. Glasgow JM, Vaughn-Sarrazin M, Kaboli PJ. Leaving against medical advice (AMA): risk of 30-day mortality and hospital readmission. J Gen Intern Med. 2010;25(9):926-929. PubMed
19. Garland A, Ramsey CD, Fransoo R, et al. Rates of readmission and death associated with leaving hospital against medical advice: a population-based study. CMAJ. 2013;185(14):1207-1214. PubMed
20. Hwang SW, Li J, Gupta R, Chien V, Martin RE. What happens to patients who leave hospital against medical advice? CMAJ. 2003;168(4):417-420. PubMed
21. Onukwugha E, Mullins CD, Loh FE, Saunders E, Shaya FT, Weir MR. Readmissions after unauthorized discharges in the cardiovascular setting. Med Care. 2011;49(2):215-224. PubMed
22. Alfandre D. Reconsidering against medical advice discharges: embracing patient-centeredness to promote high quality care and a renewed research agenda. J Gen Intern Med. 2013;28(12):1657-1662. PubMed
23. Berger JT. Discharge against medical advice: ethical considerations and professional obligations. J Hosp Med. 2008;3(5):403-408. PubMed
24. Franks P, Meldrum S, Fiscella K. Discharges against medical advice: are race/ethnicity predictors? J Gen Intern Med. 2006;21(9):955-960. PubMed
25. Hicks LS, Ayanian JZ, Orav EJ, et al. Is hospital service associated with racial and ethnic disparities in experiences with hospital care? Am J Med. 2005;118(5):529-535. PubMed
26. Hicks LS, Tovar DA, Orav EJ, Johnson PA. Experiences with hospital care: perspectives of black and Hispanic patients. J Gen Intern Med. 2008;23(8):1234-1240. PubMed
27. McCreaddie M, Lyons I, Watt D, et al. Routines and rituals: a grounded theory of the pain management of drug users in acute care settings. J Clin Nurs. 2010;19(19-20):2730-2740. PubMed
28. Carroll IR, Angst MS, Clark JD. Management of perioperative pain in patients chronically consuming opioids. Reg Anesth Pain Med. 2004;29(6):576-591. PubMed
29. Chan AC, Palepu A, Guh DP, et al. HIV-positive injection drug users who leave the hospital against medical advice: the mitigating role of methadone and social support. J Acquir Immune Defic Syndr. 2004;35(1):56-59. PubMed
30. Levy F, Mareiniss DP, Iacovelli C. The importance of a proper against-medical-advice (AMA) discharge: how signing out AMA may create significant liability protection for providers. J Emerg Med. 2012;43(3):516-520. PubMed
31. Ashbrook L, Mourad M, Sehgal N. Communicating discharge instructions to patients: a survey of nurse, intern, and hospitalist practices. J Hosp Med. 2013;8(1):36-41. PubMed
32. Joint Commission on Accreditation of Healthcare Organizations. Title 22, California Code of Regulations, §70707.3.
© 2017 Society of Hospital Medicine