User login
Menstrual migraines: Which options and when?
› Consider recommending that patients with menstrual migraines try using prophylactic triptans 2 days before the onset of menses. B
› Advise against estrogen-containing contraception for women who have menstrual migraines with aura, who smoke, or are over 35, due to the increased risk of stroke (absolute contraindication). A
› Consider estrogen-containing contraception if the benefits outweigh the risks for women with migraines who are under 35 and do not have aura (relative contraindication). A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE › Mary, a 34-year-old woman, is a new patient to your practice after moving to the area for a job. She has a history of migraine headaches triggered by her menstrual periods. She has been taking combined oral contraceptives (COCs) since she was 17, with a few years off when she had 2 children. Her migraines improved when she was pregnant, but worsened postpartum with each of her daughters to a point where she had to stop breastfeeding at 4 months to go back on the pills.
On the COCs, she gets one or 2 mild-to-moderate headaches a month. She uses sumatriptan for abortive treatment with good relief. She has not missed work in the past 4 years because of her migraines. During the 6 months she was off COCs when trying to get pregnant, she routinely missed 2 to 3 workdays per month due to migraines. She knows when she is going to get a headache because she sees flashing lights in her left visual field. She has no other neurologic symptoms with the headaches, and the character of the headaches has not changed. She is a non-smoker, has normal blood pressure and lipid levels, and no other vascular risk factors.
You review her history and talk to her about the risk of stroke with migraines and with COCs. She is almost 35 years of age and you recommend stopping the COCs due to the risk. She feels strongly that she wants to continue taking the COCs, saying her quality of life is poor when she is off the pills. What should you do?
Migraine headaches are 2 to 3 times more prevalent in women than in men,1 with a lifetime risk of 43% vs 18%, respectively.2 Women account for about 80% of the $1 billion spent each year in the United States in medical expenses and lost work productivity related to migraines.1,2
Clinical patterns suggestive of menstrual migraine. About half of women affected by migraine have menstrually-related migraines (MRM); 3% to 12% have pure menstrual migraines (PMM).3 MRM and PMM are both characterized by the presence of symptoms in at least 2 to 3 consecutive cycles, with symptoms occurring from between 2 days before to 3 days after the onset of menstruation. However, in PMM, symptoms do not occur at any other time of the menstrual cycle; in MRM, symptoms can occur at other times of the cycle. PMM is more likely to respond to hormone therapy than is MRM.
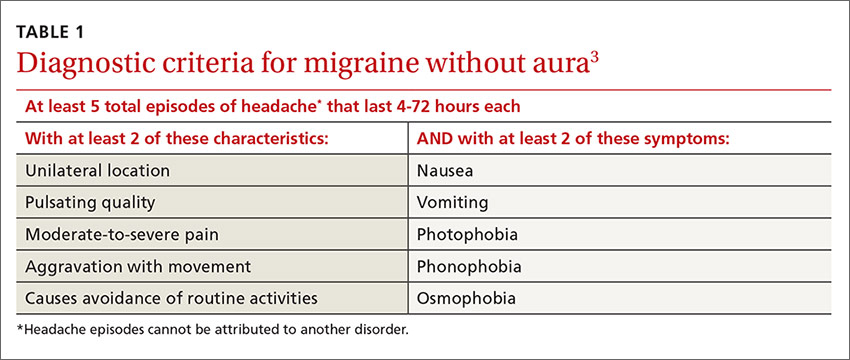
Multiple studies in the United States, Europe, and Asia have noted that migraines related to menses typically last longer, are more severe, less likely to be associated with aura, and more likely to be recurrent and recalcitrant to treatment than non-menstrual migraines.1 TABLE 13 describes diagnostic criteria for migraine without aura.
Possible mechanisms of MRM and PMM. The etiology of migraine is not well understood and is likely multifactorial.4 Incidence of menstrual migraines is related to cyclic changes in female hormones—specifically, the decreasing levels of estrogen that typically happen the week before onset of menses.1 The mechanism is not yet clear, though it is thought that a decline in estrogen levels triggers a decline in serotonin levels, which may lead to cranial vasodilation and sensitization of the trigeminal nerve.5,6 Estrogen decline has also been linked to increased cranial nociception as well as decreased endogenous opioid activity. A study using positron emission tomography found increased activity of serotonergic neurons in migraineurs.7 The evidence that triptans and serotonin receptor agonists are effective in the treatment of migraine also supports the theory that serotonin neurohormonal signaling pathways play a critical role in the pathogenesis of migraines.7
Prevalence patterns point to the role of estrogen. The prevalence of migraines in women increases around puberty, peaks between ages 30 and 40, and decreases after natural menopause.6 Migraine prevalence increases during the first week postpartum, when levels of estrogen and progesterone decrease suddenly and significantly.1 Migraine frequency and intensity decrease in the second and third trimesters of pregnancy and after menopause, when estrogen levels fluctuate significantly less.1 In the Women’s Health Initiative study, women who used hormone replacement therapy (HRT) had a 42% increased risk of migraines compared with women in the study who had never used HRT.8
The association of migraine with female hormones was further supported by a Dutch study of male-to-female transgender patients on estrogen therapy, who had a 26% incidence of migraine, equivalent to the 25% prevalence in natal female controls in this study, compared with just 7.5% in male controls.9 The association between migraine and estrogen withdrawal was investigated in studies performed more than 40 years ago, when women experiencing migraines around the time of menses were given intramuscular estradiol and experienced a delay in symptom onset.10
Abortive and prophylactic treatments: Factors that guide selection
In considering probable menstrual migraine, take a detailed history, review headache diaries if available to determine association of headaches with menses, and perform a thorough neurologic examination. If a diagnosis of menstrual migraine is established, discuss the benefits of different treatment options, both abortive and prophylactic.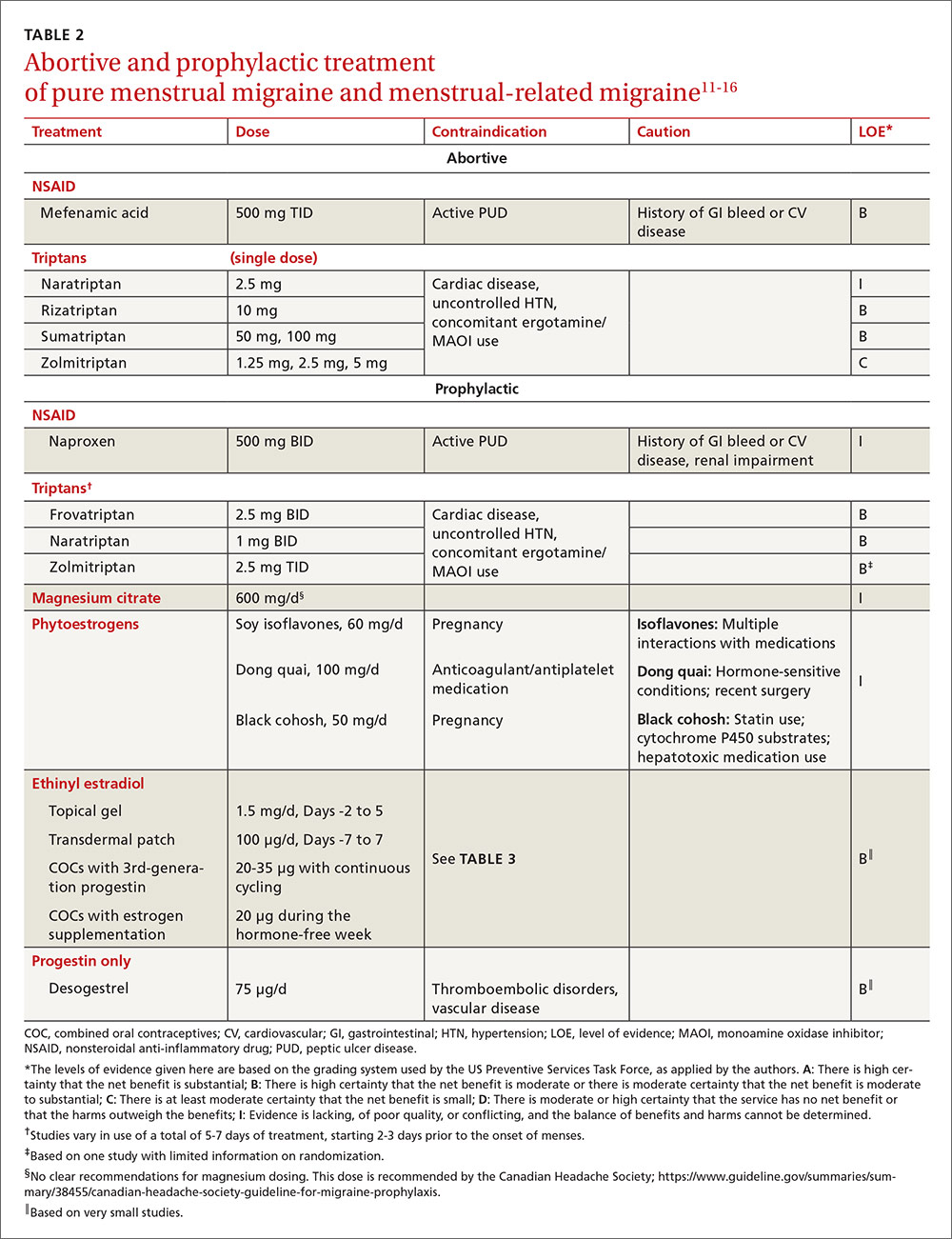
For the patient with MRM, take into account frequency of symptoms, predictability of menstruation, medication costs, and comorbidities. Both triptans and nonsteroidal anti-inflammatory drugs (NSAIDs) can be effective treatments for MRM.11 Abortive therapy may be appropriate if a patient prefers to take medication intermittently, if her menses are unpredictable, or if she does not get migraine headaches with every menses. Mefenamic acid, sumatriptan, and rizatriptan have category B recommendations for abortive treatment for menstrual migraines (TABLE 211-16). (For the patient who has regular MRM but unpredictable menses, ovulation predictor kits can be used to help predict the onset of menses, although this would involve additional cost.)
For the patient who has predictable menses and regularly occurring menstrual migraine, some data show that a short-term prophylactic regimen with triptans started 2 to 3 days before the onset of menses and continued for 5 to 7 days total can reduce the incidence of menstrual migraine (TABLE 211-16). At least one high-quality randomized controlled trial (RCT) showed a significant reduction in the incidence of MRM when women were treated prophylactically with frovatriptan, a long-acting triptan with a half-life of approximately 26 hours. Participants received frovatriptan 2.5 mg once a day or twice a day or placebo in the perimenstrual period (day -2 to +3). The incidence of MRM was 52%, 41%, and 67%, respectively (P<.0001).11,17
Another RCT of fair quality examined the effect of naratriptan (half-life 6-8 hours) on the median number of menstrual migraines over 4 menstrual cycles. Women who received 1 mg of naratriptan BID for 2 to 3 days before menses had 2 MRM episodes over the 4 cycles compared with 4 MRM episodes in women who received placebo over the same time period (P<.05).11,18 A third RCT, also of fair quality, compared 2 different regimens of zolmitriptan (half-life 3 hours) with placebo and found that women who received 2.5 mg of zolmitriptan either BID or TID 2 to 3 days prior to menses had a reduction both in frequency of menstrual migraines and in the mean number of breakthrough headaches per menstrual cycle, as well as a reduction in the need for rescue medications.12,19 Triptans are contraindicated in women with a history of cardiac disease or uncontrolled hypertension. Also, triptans can be expensive, precluding their use for some patients.
Evidence is insufficient to recommend for or against the use of NSAIDs as prophylaxis for MRM.11 NSAIDs may be contraindicated in women with a history of peptic ulcer disease or gastrointestinal bleeding. That said, if NSAIDs are not contraindicated, a trial may be reasonable given their low cost.
Data are sparse on the use of vitamins and supplements in treating and preventing PMM or MRM. In one very small double-blind, placebo-controlled study in 1991 (N=24, with efficacy data for 20), participants received a 2-week course of oral magnesium premenstrually. There was a statistically significant reduction in the number of days with headache per month (from 4.7±3.1 days to 2.4±2.2 days; P<.01) and in the total pain index (P<.03).20 A number of studies have demonstrated a correlation between hypomagnesemia and migraine headaches.5,21 The exact mechanism for this relationship is unclear.
Some recent evidence-based reviews have examined the efficacy of nutraceuticals such as magnesium, feverfew, butterbur, coenzyme Q10, and riboflavin on typical migraine, but it is not clear if these results are translatable to the treatment and prophylaxis of menstrual migraine.11,22 A multicenter, single-blind, RCT is underway to examine the efficacy of acupuncture as prophylaxis for MRM.23
Estrogen: Prescribing criteria are strict
Most studies examining the role of exogenous estrogen in reducing menstrual migraines have used topical estrogen (either in patch or gel formulations) in the perimenstrual window (TABLE 211-16). The topical estrogen route has been examined, in particular, as it is presumed to confer less risk of hypercoagulability by avoiding first-pass metabolism. However, there is conflicting evidence on this issue, in particular regarding premenopausal women.13,25 Additionally, many of the studies of estrogen supplementation show a trend toward increased headache once estrogen is discontinued, presumably due to estrogen withdrawal.10,24
That said, one study by MacGregor, et al demonstrates that the use of estradiol gel in the perimenstrual window leads to a 22% reduction in migraine days as well as less severe migrainous symptoms.26 This trend has been demonstrated in other studies examining estrogen supplementation. Of note, the estrogen studies generally are small, older, and of fair to poor quality.11 These studies have used higher doses of estrogen than are commonly used for contraception today because lower doses of estrogen seem not to have the same impact on migraine.5,24
As for COCs, with either normal or extended cycling, data are more mixed than for estrogen supplementation alone; equivalent numbers of women experience improvement, no change, or worsening of their headache pattern. Many women have continuing or worsening migraines in the hormone-free week, and thus most studies have examined the use of extended cycling COCs.5 Sulak, et al demonstrated a statistically significant reduction in headache frequency using extended-cycling COCs, though they did not examine MRM in particular.27 The efficacy of extended-cycling COCs for reduction of MRM was confirmed by Coffee and colleagues with a small but statistically significant decrease in daily headache scores.28
Adverse effects. All estrogen therapies pose the risk of adverse effects (deep vein thrombosis, hypertension, breast tenderness, nausea, etc). Additionally, estrogen supplementation may actually trigger migraines in some women if, when it is discontinued, the blood estrogen level does not remain above a threshold concentration.5,10,24 Estrogen may also trigger migraine in previously headache-free women and may convert migraine without aura into migraine with aura. In either case, therapy should be stopped.5,24
There is promising evidence from 2 small RCTs and one observational trial that progestin-only contraceptive pills (POP) may reduce the frequency and severity of menstrual migraines (TABLE 211-16). More prospective data are needed to confirm this reduction, as there have not been specific studies examining other progesterone-only preparations to prevent menstrual migraines.
Risk of ischemic stroke. Unfortunately, there are population data showing that second-generation and, to a smaller degree, third-generation progestins, which include the desogestrel used in the above studies, may increase the risk of ischemic stroke. This is a particular concern in women who experience migraine.29 Second-generation progestins include levonorgestrel, which is in the levonorgestrel IUD; however, there is no direct evidence for increased ischemic stroke in this particular preparation, and the circulating plasma levels are low. Etonorgestrel, the active ingredient in the contraceptive implant, is a third-generation progestin, though there is no direct evidence of increased ischemic stroke with use of the etonorgestrel implant.
There is a 2- to 4-fold increased risk of ischemic stroke in women who experience migraine.1,5,30 As stated above, this risk may be further increased by some progesterone formulations. But there is also a demonstrable increase in ischemic stroke risk with the use of estrogen, particularly at the higher concentrations that have been shown to prevent MRM.31,32 The overall incidence of ischemic stroke in menstrual-age women is low, which has limited the number of studies with enough power to quantify the absolute increased risk of stroke in conjunction with estrogen use. Nevertheless, exogenous estrogen is thought to increase the risk of ischemic stroke an additional 2- to 4-fold.1,5,29,30,32-34
Women who experience aura. MRM, as it is defined, typically excludes women who experience aura; however, the number of women who experience aura with migraine either in proximity to their menses or throughout the month has not been well documented. The risk of ischemic stroke is higher for women who experience migraine with aura than those with migraine alone, possibly because aura is associated with reduced regional vascular flow leading to hypoperfusion, which sets the stage for a possible ischemic event.4,5,35 The risk of ischemic stroke is amplified further for women who are over 35, who smoke, or who have additional vascular risk factors (eg, uncontrolled hypertension, diabetes, or known vascular or cardiac disease).1,5,34 This array of evidence serves as the basis for the US Medical Eligibility Criteria (USMEC) recommendations36 for hormonal contraceptive use, in particular the absolute contraindication for estrogen use in women who experience migraine with aura (TABLE 336-38).
CASE › Given Mary’s experience of aura with migraine, you talk with her at length about the risk of ischemic stroke and the USMEC recommendation that she absolutely should not be taking COCs. You suggest a progestin-only method of contraception such as depot medroxyprogesterone acetate, a progestin intrauterine device, or a hormonal implant, which may suppress ovulation and decrease her headaches. You discuss that while some women may have headaches with these progestin-only methods, stroke risk is significantly reduced. You also suggest a trial of prophylactic triptans as another possible option.
She says she understands the increased risk of stroke but is still unwilling to try anything else right now due to worries about her quality of life. You decide jointly to refill COCs for 3 months, and you document the shared decision process in the chart. After advising the patient that you will not continue to prescribe COCs for an extended period of time, you also schedule a follow-up appointment to further discuss risks and benefits of migraine treatment and means of reducing other risk factors for stroke.
CORRESPONDENCE
Sarina Schrager, MD, MS, University of Wisconsin, Department of Family Medicine, 1100 Delaplaine Ct, Madison, WI 53715; [email protected].
1. MacGregor EA, Rosenberg JD, Kurth T. Sex-related differences in epidemiological and clinic-based headache studies. Headache. 2011;51:843-859.
2. Stewart WF, Wood C, Reed ML, et al. Cumulative lifetime migraine incidence in women and men. Cephalalgia. 2008;28:1170-1178.
3. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd ed. Cephalalgia. 2013;33:629-808.
4. Garza I, Swanson JW, Cheshire WP Jr, et al. Headache and other craniofacial pain. In: Daroff RB, Fenichel GM, Jankovic J, et al, eds. Bradley’s Neurology in Clinical Practice. 6th ed. Philadelphia, PA: Elsevier Saunders; 2012:1703-1744.
5. Martin VT, Behbehani M. Ovarian hormones and migraine headache: understanding mechanisms and pathogenesis – part 2. Headache. 2006;46:365-386.
6. Brandes JL. The influence of estrogen on migraine: a systematic review. JAMA. 2006; 295:1824-1830.
7. Loder EW. Menstrual migraine: pathophysiology, diagnosis and impact. Headache. 2006;46 (Suppl 2):S55-S60.
8. Misakian AL, Langer RD, Bensenor IM, et al. Postmenopausal hormone therapy and migraine headache. J Women’s Health (Larchmt). 2003;12:1027-1036.
9. Pringsheim T, Gooren L. Migraine prevalence in male to female transsexuals on hormone therapy. Neurology. 2004;63:593-594.
10. Somerville BW. The role of estradiol withdrawal in the etiology of menstrual migraine. Neurology. 1972;22:355-365.
11. Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70:1555-1563.
12. Hu Y, Guan X, Fan L, et al. Triptans in prevention of menstrual migraine: a systematic review with meta-analysis. J Headache Pain. 2013;14:7.
13. Canonico M, Plu-Bureau G, Lowe GD, et al. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ. 2008;336:1227-1231.
14. Merki-Feld GS, Imthurn B, Langner R, et al. Headache frequency and intensity in female migraineurs using desogestrel-only contraception: a retrospective pilot diary study. Cephalalgia. 2013;33:340-346.
15.
16. Morotti M, Remorgida V, Venturini PL, et al. Progestin-only contraception compared with extended combined oral contraceptive in women with migraine without aura: a retrospective pilot study. Eur J Obstet Gynecol Reprod Biol. 2014;183:178-182.
17. Silberstein SD, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63:261-269.
18. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized double-blind, placebo-controlled study. Headache. 2001;41:248-256.
19. Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs. 2008;22:877-886.
20. Facchinetti F, Sances G, Borella P, et al. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31:298-301.
21. Teigen L, Boes CJ. An evidence-based review of oral magnesium supplementation in the preventive treatment of migraine. Cephalalgia. 2014;35:912-922.
22. Taylor FR. Nutraceuticals and headache: the biological basis. Headache. 2011;51:484-501.
23. Zhang XZ, Zhang L, Guo J, et al. Acupuncture as prophylaxis for menstrual-related migraine: study protocol for a multicenter randomized controlled trial. Trials. 2013;14:374.
24. MacGregor EA. Oestrogen and attacks of migraine with and without aura. Lancet Neurol. 2004;3:354-361.
25. Cole JA, Norman H, Doherty M, et al. Venous thromboembolism, myocardial infarction, and stroke among transdermal contraceptive system users. Obstet Gynecol. 2007;109:339-346.
26. MacGregor EA, Frith A, Ellis J, et al. Prevention of menstrual attacks of migraine: a double blind placebo-controlled crossover study. Neurology. 2006;67:2159-2163.
27. Sulak P, Willis S, Kuehl T, et al. Headaches and oral contraceptives: impact of eliminating the standard 7-day placebo interval. Headache. 2007;47:27-37.
28. Coffee AL, Sulak PJ, Hill AJ, et al. Extended cycle combined oral contraceptives and prophylactic frovatriptan during the hormone-free interval in women with menstrual-related migraines. J Womens Health. 2014;23:310-317.
29. Lidegaard Ø, Kreiner S. Contraceptives and cerebral thrombosis: a five-year national case-control study. Contraception. 2002;65:197-205.
30. Bousser MG. Estrogen, migraine, and stroke. Stroke. 2004;35(Suppl 1):2652-2656.
31. Gillum LA, Mamidipudi SK, Johnston SC. Ischemic stroke risk with oral contraceptives: A meta-analysis. JAMA. 2000;284:72-78.
32. Donaghy M, Chang CL, Poulter N. Duration, frequency, recency, and type of migraine and the risk of ischaemic stroke in women of childbearing age. J Neurol Neurosurg Psychiatry. 2002;73:747-750.
33. Sacco S, Ricci S, Degan D. Migraine in women: the role of hormones and their impact on vascular diseases. J Headache Pain. 2012;12:177-189.
34. Merikangas KR, Fenton BT, Cheng SH, et al. Association between migraine and stroke in a large-scale epidemiological study of the United States. Arch Neurol. 1997;54:362-368.
35. MacClellan LR, Giles W, Cole J, et al. Probable migraine with visual aura and risk of ischemic stroke: the stroke prevention in young women study. Stroke. 2007;38:2438-2445.
36. Centers for Disease Control and Prevention. U.S. Medical Eligibility Criteria for Contraceptive Use, 2010. MMWR Recomm Rep
37. Chang CL, Donaghy M, Poulter N. Migraine and stroke in young women: case-control study. The World Health Organisation Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception. BMJ. 1999;318:13-18.
38. Tzourio C, Tehindrazanarivelo A, Iglésias S, et al. Case-control study of migraine and risk of ischaemic stroke in young women. BMJ. 1995;310:830-833.
› Consider recommending that patients with menstrual migraines try using prophylactic triptans 2 days before the onset of menses. B
› Advise against estrogen-containing contraception for women who have menstrual migraines with aura, who smoke, or are over 35, due to the increased risk of stroke (absolute contraindication). A
› Consider estrogen-containing contraception if the benefits outweigh the risks for women with migraines who are under 35 and do not have aura (relative contraindication). A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE › Mary, a 34-year-old woman, is a new patient to your practice after moving to the area for a job. She has a history of migraine headaches triggered by her menstrual periods. She has been taking combined oral contraceptives (COCs) since she was 17, with a few years off when she had 2 children. Her migraines improved when she was pregnant, but worsened postpartum with each of her daughters to a point where she had to stop breastfeeding at 4 months to go back on the pills.
On the COCs, she gets one or 2 mild-to-moderate headaches a month. She uses sumatriptan for abortive treatment with good relief. She has not missed work in the past 4 years because of her migraines. During the 6 months she was off COCs when trying to get pregnant, she routinely missed 2 to 3 workdays per month due to migraines. She knows when she is going to get a headache because she sees flashing lights in her left visual field. She has no other neurologic symptoms with the headaches, and the character of the headaches has not changed. She is a non-smoker, has normal blood pressure and lipid levels, and no other vascular risk factors.
You review her history and talk to her about the risk of stroke with migraines and with COCs. She is almost 35 years of age and you recommend stopping the COCs due to the risk. She feels strongly that she wants to continue taking the COCs, saying her quality of life is poor when she is off the pills. What should you do?
Migraine headaches are 2 to 3 times more prevalent in women than in men,1 with a lifetime risk of 43% vs 18%, respectively.2 Women account for about 80% of the $1 billion spent each year in the United States in medical expenses and lost work productivity related to migraines.1,2
Clinical patterns suggestive of menstrual migraine. About half of women affected by migraine have menstrually-related migraines (MRM); 3% to 12% have pure menstrual migraines (PMM).3 MRM and PMM are both characterized by the presence of symptoms in at least 2 to 3 consecutive cycles, with symptoms occurring from between 2 days before to 3 days after the onset of menstruation. However, in PMM, symptoms do not occur at any other time of the menstrual cycle; in MRM, symptoms can occur at other times of the cycle. PMM is more likely to respond to hormone therapy than is MRM.
Multiple studies in the United States, Europe, and Asia have noted that migraines related to menses typically last longer, are more severe, less likely to be associated with aura, and more likely to be recurrent and recalcitrant to treatment than non-menstrual migraines.1 TABLE 13 describes diagnostic criteria for migraine without aura.

Possible mechanisms of MRM and PMM. The etiology of migraine is not well understood and is likely multifactorial.4 Incidence of menstrual migraines is related to cyclic changes in female hormones—specifically, the decreasing levels of estrogen that typically happen the week before onset of menses.1 The mechanism is not yet clear, though it is thought that a decline in estrogen levels triggers a decline in serotonin levels, which may lead to cranial vasodilation and sensitization of the trigeminal nerve.5,6 Estrogen decline has also been linked to increased cranial nociception as well as decreased endogenous opioid activity. A study using positron emission tomography found increased activity of serotonergic neurons in migraineurs.7 The evidence that triptans and serotonin receptor agonists are effective in the treatment of migraine also supports the theory that serotonin neurohormonal signaling pathways play a critical role in the pathogenesis of migraines.7
Prevalence patterns point to the role of estrogen. The prevalence of migraines in women increases around puberty, peaks between ages 30 and 40, and decreases after natural menopause.6 Migraine prevalence increases during the first week postpartum, when levels of estrogen and progesterone decrease suddenly and significantly.1 Migraine frequency and intensity decrease in the second and third trimesters of pregnancy and after menopause, when estrogen levels fluctuate significantly less.1 In the Women’s Health Initiative study, women who used hormone replacement therapy (HRT) had a 42% increased risk of migraines compared with women in the study who had never used HRT.8
The association of migraine with female hormones was further supported by a Dutch study of male-to-female transgender patients on estrogen therapy, who had a 26% incidence of migraine, equivalent to the 25% prevalence in natal female controls in this study, compared with just 7.5% in male controls.9 The association between migraine and estrogen withdrawal was investigated in studies performed more than 40 years ago, when women experiencing migraines around the time of menses were given intramuscular estradiol and experienced a delay in symptom onset.10
Abortive and prophylactic treatments: Factors that guide selection
In considering probable menstrual migraine, take a detailed history, review headache diaries if available to determine association of headaches with menses, and perform a thorough neurologic examination. If a diagnosis of menstrual migraine is established, discuss the benefits of different treatment options, both abortive and prophylactic.
For the patient with MRM, take into account frequency of symptoms, predictability of menstruation, medication costs, and comorbidities. Both triptans and nonsteroidal anti-inflammatory drugs (NSAIDs) can be effective treatments for MRM.11 Abortive therapy may be appropriate if a patient prefers to take medication intermittently, if her menses are unpredictable, or if she does not get migraine headaches with every menses. Mefenamic acid, sumatriptan, and rizatriptan have category B recommendations for abortive treatment for menstrual migraines (TABLE 211-16). (For the patient who has regular MRM but unpredictable menses, ovulation predictor kits can be used to help predict the onset of menses, although this would involve additional cost.)
For the patient who has predictable menses and regularly occurring menstrual migraine, some data show that a short-term prophylactic regimen with triptans started 2 to 3 days before the onset of menses and continued for 5 to 7 days total can reduce the incidence of menstrual migraine (TABLE 211-16). At least one high-quality randomized controlled trial (RCT) showed a significant reduction in the incidence of MRM when women were treated prophylactically with frovatriptan, a long-acting triptan with a half-life of approximately 26 hours. Participants received frovatriptan 2.5 mg once a day or twice a day or placebo in the perimenstrual period (day -2 to +3). The incidence of MRM was 52%, 41%, and 67%, respectively (P<.0001).11,17
Another RCT of fair quality examined the effect of naratriptan (half-life 6-8 hours) on the median number of menstrual migraines over 4 menstrual cycles. Women who received 1 mg of naratriptan BID for 2 to 3 days before menses had 2 MRM episodes over the 4 cycles compared with 4 MRM episodes in women who received placebo over the same time period (P<.05).11,18 A third RCT, also of fair quality, compared 2 different regimens of zolmitriptan (half-life 3 hours) with placebo and found that women who received 2.5 mg of zolmitriptan either BID or TID 2 to 3 days prior to menses had a reduction both in frequency of menstrual migraines and in the mean number of breakthrough headaches per menstrual cycle, as well as a reduction in the need for rescue medications.12,19 Triptans are contraindicated in women with a history of cardiac disease or uncontrolled hypertension. Also, triptans can be expensive, precluding their use for some patients.
Evidence is insufficient to recommend for or against the use of NSAIDs as prophylaxis for MRM.11 NSAIDs may be contraindicated in women with a history of peptic ulcer disease or gastrointestinal bleeding. That said, if NSAIDs are not contraindicated, a trial may be reasonable given their low cost.
Data are sparse on the use of vitamins and supplements in treating and preventing PMM or MRM. In one very small double-blind, placebo-controlled study in 1991 (N=24, with efficacy data for 20), participants received a 2-week course of oral magnesium premenstrually. There was a statistically significant reduction in the number of days with headache per month (from 4.7±3.1 days to 2.4±2.2 days; P<.01) and in the total pain index (P<.03).20 A number of studies have demonstrated a correlation between hypomagnesemia and migraine headaches.5,21 The exact mechanism for this relationship is unclear.
Some recent evidence-based reviews have examined the efficacy of nutraceuticals such as magnesium, feverfew, butterbur, coenzyme Q10, and riboflavin on typical migraine, but it is not clear if these results are translatable to the treatment and prophylaxis of menstrual migraine.11,22 A multicenter, single-blind, RCT is underway to examine the efficacy of acupuncture as prophylaxis for MRM.23
Estrogen: Prescribing criteria are strict
Most studies examining the role of exogenous estrogen in reducing menstrual migraines have used topical estrogen (either in patch or gel formulations) in the perimenstrual window (TABLE 211-16). The topical estrogen route has been examined, in particular, as it is presumed to confer less risk of hypercoagulability by avoiding first-pass metabolism. However, there is conflicting evidence on this issue, in particular regarding premenopausal women.13,25 Additionally, many of the studies of estrogen supplementation show a trend toward increased headache once estrogen is discontinued, presumably due to estrogen withdrawal.10,24
That said, one study by MacGregor, et al demonstrates that the use of estradiol gel in the perimenstrual window leads to a 22% reduction in migraine days as well as less severe migrainous symptoms.26 This trend has been demonstrated in other studies examining estrogen supplementation. Of note, the estrogen studies generally are small, older, and of fair to poor quality.11 These studies have used higher doses of estrogen than are commonly used for contraception today because lower doses of estrogen seem not to have the same impact on migraine.5,24
As for COCs, with either normal or extended cycling, data are more mixed than for estrogen supplementation alone; equivalent numbers of women experience improvement, no change, or worsening of their headache pattern. Many women have continuing or worsening migraines in the hormone-free week, and thus most studies have examined the use of extended cycling COCs.5 Sulak, et al demonstrated a statistically significant reduction in headache frequency using extended-cycling COCs, though they did not examine MRM in particular.27 The efficacy of extended-cycling COCs for reduction of MRM was confirmed by Coffee and colleagues with a small but statistically significant decrease in daily headache scores.28
Adverse effects. All estrogen therapies pose the risk of adverse effects (deep vein thrombosis, hypertension, breast tenderness, nausea, etc). Additionally, estrogen supplementation may actually trigger migraines in some women if, when it is discontinued, the blood estrogen level does not remain above a threshold concentration.5,10,24 Estrogen may also trigger migraine in previously headache-free women and may convert migraine without aura into migraine with aura. In either case, therapy should be stopped.5,24
There is promising evidence from 2 small RCTs and one observational trial that progestin-only contraceptive pills (POP) may reduce the frequency and severity of menstrual migraines (TABLE 211-16). More prospective data are needed to confirm this reduction, as there have not been specific studies examining other progesterone-only preparations to prevent menstrual migraines.
Risk of ischemic stroke. Unfortunately, there are population data showing that second-generation and, to a smaller degree, third-generation progestins, which include the desogestrel used in the above studies, may increase the risk of ischemic stroke. This is a particular concern in women who experience migraine.29 Second-generation progestins include levonorgestrel, which is in the levonorgestrel IUD; however, there is no direct evidence for increased ischemic stroke in this particular preparation, and the circulating plasma levels are low. Etonorgestrel, the active ingredient in the contraceptive implant, is a third-generation progestin, though there is no direct evidence of increased ischemic stroke with use of the etonorgestrel implant.
There is a 2- to 4-fold increased risk of ischemic stroke in women who experience migraine.1,5,30 As stated above, this risk may be further increased by some progesterone formulations. But there is also a demonstrable increase in ischemic stroke risk with the use of estrogen, particularly at the higher concentrations that have been shown to prevent MRM.31,32 The overall incidence of ischemic stroke in menstrual-age women is low, which has limited the number of studies with enough power to quantify the absolute increased risk of stroke in conjunction with estrogen use. Nevertheless, exogenous estrogen is thought to increase the risk of ischemic stroke an additional 2- to 4-fold.1,5,29,30,32-34
Women who experience aura. MRM, as it is defined, typically excludes women who experience aura; however, the number of women who experience aura with migraine either in proximity to their menses or throughout the month has not been well documented. The risk of ischemic stroke is higher for women who experience migraine with aura than those with migraine alone, possibly because aura is associated with reduced regional vascular flow leading to hypoperfusion, which sets the stage for a possible ischemic event.4,5,35 The risk of ischemic stroke is amplified further for women who are over 35, who smoke, or who have additional vascular risk factors (eg, uncontrolled hypertension, diabetes, or known vascular or cardiac disease).1,5,34 This array of evidence serves as the basis for the US Medical Eligibility Criteria (USMEC) recommendations36 for hormonal contraceptive use, in particular the absolute contraindication for estrogen use in women who experience migraine with aura (TABLE 336-38).
CASE › Given Mary’s experience of aura with migraine, you talk with her at length about the risk of ischemic stroke and the USMEC recommendation that she absolutely should not be taking COCs. You suggest a progestin-only method of contraception such as depot medroxyprogesterone acetate, a progestin intrauterine device, or a hormonal implant, which may suppress ovulation and decrease her headaches. You discuss that while some women may have headaches with these progestin-only methods, stroke risk is significantly reduced. You also suggest a trial of prophylactic triptans as another possible option.
She says she understands the increased risk of stroke but is still unwilling to try anything else right now due to worries about her quality of life. You decide jointly to refill COCs for 3 months, and you document the shared decision process in the chart. After advising the patient that you will not continue to prescribe COCs for an extended period of time, you also schedule a follow-up appointment to further discuss risks and benefits of migraine treatment and means of reducing other risk factors for stroke.
CORRESPONDENCE
Sarina Schrager, MD, MS, University of Wisconsin, Department of Family Medicine, 1100 Delaplaine Ct, Madison, WI 53715; [email protected].
› Consider recommending that patients with menstrual migraines try using prophylactic triptans 2 days before the onset of menses. B
› Advise against estrogen-containing contraception for women who have menstrual migraines with aura, who smoke, or are over 35, due to the increased risk of stroke (absolute contraindication). A
› Consider estrogen-containing contraception if the benefits outweigh the risks for women with migraines who are under 35 and do not have aura (relative contraindication). A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE › Mary, a 34-year-old woman, is a new patient to your practice after moving to the area for a job. She has a history of migraine headaches triggered by her menstrual periods. She has been taking combined oral contraceptives (COCs) since she was 17, with a few years off when she had 2 children. Her migraines improved when she was pregnant, but worsened postpartum with each of her daughters to a point where she had to stop breastfeeding at 4 months to go back on the pills.
On the COCs, she gets one or 2 mild-to-moderate headaches a month. She uses sumatriptan for abortive treatment with good relief. She has not missed work in the past 4 years because of her migraines. During the 6 months she was off COCs when trying to get pregnant, she routinely missed 2 to 3 workdays per month due to migraines. She knows when she is going to get a headache because she sees flashing lights in her left visual field. She has no other neurologic symptoms with the headaches, and the character of the headaches has not changed. She is a non-smoker, has normal blood pressure and lipid levels, and no other vascular risk factors.
You review her history and talk to her about the risk of stroke with migraines and with COCs. She is almost 35 years of age and you recommend stopping the COCs due to the risk. She feels strongly that she wants to continue taking the COCs, saying her quality of life is poor when she is off the pills. What should you do?
Migraine headaches are 2 to 3 times more prevalent in women than in men,1 with a lifetime risk of 43% vs 18%, respectively.2 Women account for about 80% of the $1 billion spent each year in the United States in medical expenses and lost work productivity related to migraines.1,2
Clinical patterns suggestive of menstrual migraine. About half of women affected by migraine have menstrually-related migraines (MRM); 3% to 12% have pure menstrual migraines (PMM).3 MRM and PMM are both characterized by the presence of symptoms in at least 2 to 3 consecutive cycles, with symptoms occurring from between 2 days before to 3 days after the onset of menstruation. However, in PMM, symptoms do not occur at any other time of the menstrual cycle; in MRM, symptoms can occur at other times of the cycle. PMM is more likely to respond to hormone therapy than is MRM.
Multiple studies in the United States, Europe, and Asia have noted that migraines related to menses typically last longer, are more severe, less likely to be associated with aura, and more likely to be recurrent and recalcitrant to treatment than non-menstrual migraines.1 TABLE 13 describes diagnostic criteria for migraine without aura.

Possible mechanisms of MRM and PMM. The etiology of migraine is not well understood and is likely multifactorial.4 Incidence of menstrual migraines is related to cyclic changes in female hormones—specifically, the decreasing levels of estrogen that typically happen the week before onset of menses.1 The mechanism is not yet clear, though it is thought that a decline in estrogen levels triggers a decline in serotonin levels, which may lead to cranial vasodilation and sensitization of the trigeminal nerve.5,6 Estrogen decline has also been linked to increased cranial nociception as well as decreased endogenous opioid activity. A study using positron emission tomography found increased activity of serotonergic neurons in migraineurs.7 The evidence that triptans and serotonin receptor agonists are effective in the treatment of migraine also supports the theory that serotonin neurohormonal signaling pathways play a critical role in the pathogenesis of migraines.7
Prevalence patterns point to the role of estrogen. The prevalence of migraines in women increases around puberty, peaks between ages 30 and 40, and decreases after natural menopause.6 Migraine prevalence increases during the first week postpartum, when levels of estrogen and progesterone decrease suddenly and significantly.1 Migraine frequency and intensity decrease in the second and third trimesters of pregnancy and after menopause, when estrogen levels fluctuate significantly less.1 In the Women’s Health Initiative study, women who used hormone replacement therapy (HRT) had a 42% increased risk of migraines compared with women in the study who had never used HRT.8
The association of migraine with female hormones was further supported by a Dutch study of male-to-female transgender patients on estrogen therapy, who had a 26% incidence of migraine, equivalent to the 25% prevalence in natal female controls in this study, compared with just 7.5% in male controls.9 The association between migraine and estrogen withdrawal was investigated in studies performed more than 40 years ago, when women experiencing migraines around the time of menses were given intramuscular estradiol and experienced a delay in symptom onset.10
Abortive and prophylactic treatments: Factors that guide selection
In considering probable menstrual migraine, take a detailed history, review headache diaries if available to determine association of headaches with menses, and perform a thorough neurologic examination. If a diagnosis of menstrual migraine is established, discuss the benefits of different treatment options, both abortive and prophylactic.
For the patient with MRM, take into account frequency of symptoms, predictability of menstruation, medication costs, and comorbidities. Both triptans and nonsteroidal anti-inflammatory drugs (NSAIDs) can be effective treatments for MRM.11 Abortive therapy may be appropriate if a patient prefers to take medication intermittently, if her menses are unpredictable, or if she does not get migraine headaches with every menses. Mefenamic acid, sumatriptan, and rizatriptan have category B recommendations for abortive treatment for menstrual migraines (TABLE 211-16). (For the patient who has regular MRM but unpredictable menses, ovulation predictor kits can be used to help predict the onset of menses, although this would involve additional cost.)
For the patient who has predictable menses and regularly occurring menstrual migraine, some data show that a short-term prophylactic regimen with triptans started 2 to 3 days before the onset of menses and continued for 5 to 7 days total can reduce the incidence of menstrual migraine (TABLE 211-16). At least one high-quality randomized controlled trial (RCT) showed a significant reduction in the incidence of MRM when women were treated prophylactically with frovatriptan, a long-acting triptan with a half-life of approximately 26 hours. Participants received frovatriptan 2.5 mg once a day or twice a day or placebo in the perimenstrual period (day -2 to +3). The incidence of MRM was 52%, 41%, and 67%, respectively (P<.0001).11,17
Another RCT of fair quality examined the effect of naratriptan (half-life 6-8 hours) on the median number of menstrual migraines over 4 menstrual cycles. Women who received 1 mg of naratriptan BID for 2 to 3 days before menses had 2 MRM episodes over the 4 cycles compared with 4 MRM episodes in women who received placebo over the same time period (P<.05).11,18 A third RCT, also of fair quality, compared 2 different regimens of zolmitriptan (half-life 3 hours) with placebo and found that women who received 2.5 mg of zolmitriptan either BID or TID 2 to 3 days prior to menses had a reduction both in frequency of menstrual migraines and in the mean number of breakthrough headaches per menstrual cycle, as well as a reduction in the need for rescue medications.12,19 Triptans are contraindicated in women with a history of cardiac disease or uncontrolled hypertension. Also, triptans can be expensive, precluding their use for some patients.
Evidence is insufficient to recommend for or against the use of NSAIDs as prophylaxis for MRM.11 NSAIDs may be contraindicated in women with a history of peptic ulcer disease or gastrointestinal bleeding. That said, if NSAIDs are not contraindicated, a trial may be reasonable given their low cost.
Data are sparse on the use of vitamins and supplements in treating and preventing PMM or MRM. In one very small double-blind, placebo-controlled study in 1991 (N=24, with efficacy data for 20), participants received a 2-week course of oral magnesium premenstrually. There was a statistically significant reduction in the number of days with headache per month (from 4.7±3.1 days to 2.4±2.2 days; P<.01) and in the total pain index (P<.03).20 A number of studies have demonstrated a correlation between hypomagnesemia and migraine headaches.5,21 The exact mechanism for this relationship is unclear.
Some recent evidence-based reviews have examined the efficacy of nutraceuticals such as magnesium, feverfew, butterbur, coenzyme Q10, and riboflavin on typical migraine, but it is not clear if these results are translatable to the treatment and prophylaxis of menstrual migraine.11,22 A multicenter, single-blind, RCT is underway to examine the efficacy of acupuncture as prophylaxis for MRM.23
Estrogen: Prescribing criteria are strict
Most studies examining the role of exogenous estrogen in reducing menstrual migraines have used topical estrogen (either in patch or gel formulations) in the perimenstrual window (TABLE 211-16). The topical estrogen route has been examined, in particular, as it is presumed to confer less risk of hypercoagulability by avoiding first-pass metabolism. However, there is conflicting evidence on this issue, in particular regarding premenopausal women.13,25 Additionally, many of the studies of estrogen supplementation show a trend toward increased headache once estrogen is discontinued, presumably due to estrogen withdrawal.10,24
That said, one study by MacGregor, et al demonstrates that the use of estradiol gel in the perimenstrual window leads to a 22% reduction in migraine days as well as less severe migrainous symptoms.26 This trend has been demonstrated in other studies examining estrogen supplementation. Of note, the estrogen studies generally are small, older, and of fair to poor quality.11 These studies have used higher doses of estrogen than are commonly used for contraception today because lower doses of estrogen seem not to have the same impact on migraine.5,24
As for COCs, with either normal or extended cycling, data are more mixed than for estrogen supplementation alone; equivalent numbers of women experience improvement, no change, or worsening of their headache pattern. Many women have continuing or worsening migraines in the hormone-free week, and thus most studies have examined the use of extended cycling COCs.5 Sulak, et al demonstrated a statistically significant reduction in headache frequency using extended-cycling COCs, though they did not examine MRM in particular.27 The efficacy of extended-cycling COCs for reduction of MRM was confirmed by Coffee and colleagues with a small but statistically significant decrease in daily headache scores.28
Adverse effects. All estrogen therapies pose the risk of adverse effects (deep vein thrombosis, hypertension, breast tenderness, nausea, etc). Additionally, estrogen supplementation may actually trigger migraines in some women if, when it is discontinued, the blood estrogen level does not remain above a threshold concentration.5,10,24 Estrogen may also trigger migraine in previously headache-free women and may convert migraine without aura into migraine with aura. In either case, therapy should be stopped.5,24
There is promising evidence from 2 small RCTs and one observational trial that progestin-only contraceptive pills (POP) may reduce the frequency and severity of menstrual migraines (TABLE 211-16). More prospective data are needed to confirm this reduction, as there have not been specific studies examining other progesterone-only preparations to prevent menstrual migraines.
Risk of ischemic stroke. Unfortunately, there are population data showing that second-generation and, to a smaller degree, third-generation progestins, which include the desogestrel used in the above studies, may increase the risk of ischemic stroke. This is a particular concern in women who experience migraine.29 Second-generation progestins include levonorgestrel, which is in the levonorgestrel IUD; however, there is no direct evidence for increased ischemic stroke in this particular preparation, and the circulating plasma levels are low. Etonorgestrel, the active ingredient in the contraceptive implant, is a third-generation progestin, though there is no direct evidence of increased ischemic stroke with use of the etonorgestrel implant.
There is a 2- to 4-fold increased risk of ischemic stroke in women who experience migraine.1,5,30 As stated above, this risk may be further increased by some progesterone formulations. But there is also a demonstrable increase in ischemic stroke risk with the use of estrogen, particularly at the higher concentrations that have been shown to prevent MRM.31,32 The overall incidence of ischemic stroke in menstrual-age women is low, which has limited the number of studies with enough power to quantify the absolute increased risk of stroke in conjunction with estrogen use. Nevertheless, exogenous estrogen is thought to increase the risk of ischemic stroke an additional 2- to 4-fold.1,5,29,30,32-34
Women who experience aura. MRM, as it is defined, typically excludes women who experience aura; however, the number of women who experience aura with migraine either in proximity to their menses or throughout the month has not been well documented. The risk of ischemic stroke is higher for women who experience migraine with aura than those with migraine alone, possibly because aura is associated with reduced regional vascular flow leading to hypoperfusion, which sets the stage for a possible ischemic event.4,5,35 The risk of ischemic stroke is amplified further for women who are over 35, who smoke, or who have additional vascular risk factors (eg, uncontrolled hypertension, diabetes, or known vascular or cardiac disease).1,5,34 This array of evidence serves as the basis for the US Medical Eligibility Criteria (USMEC) recommendations36 for hormonal contraceptive use, in particular the absolute contraindication for estrogen use in women who experience migraine with aura (TABLE 336-38).
CASE › Given Mary’s experience of aura with migraine, you talk with her at length about the risk of ischemic stroke and the USMEC recommendation that she absolutely should not be taking COCs. You suggest a progestin-only method of contraception such as depot medroxyprogesterone acetate, a progestin intrauterine device, or a hormonal implant, which may suppress ovulation and decrease her headaches. You discuss that while some women may have headaches with these progestin-only methods, stroke risk is significantly reduced. You also suggest a trial of prophylactic triptans as another possible option.
She says she understands the increased risk of stroke but is still unwilling to try anything else right now due to worries about her quality of life. You decide jointly to refill COCs for 3 months, and you document the shared decision process in the chart. After advising the patient that you will not continue to prescribe COCs for an extended period of time, you also schedule a follow-up appointment to further discuss risks and benefits of migraine treatment and means of reducing other risk factors for stroke.
CORRESPONDENCE
Sarina Schrager, MD, MS, University of Wisconsin, Department of Family Medicine, 1100 Delaplaine Ct, Madison, WI 53715; [email protected].
1. MacGregor EA, Rosenberg JD, Kurth T. Sex-related differences in epidemiological and clinic-based headache studies. Headache. 2011;51:843-859.
2. Stewart WF, Wood C, Reed ML, et al. Cumulative lifetime migraine incidence in women and men. Cephalalgia. 2008;28:1170-1178.
3. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd ed. Cephalalgia. 2013;33:629-808.
4. Garza I, Swanson JW, Cheshire WP Jr, et al. Headache and other craniofacial pain. In: Daroff RB, Fenichel GM, Jankovic J, et al, eds. Bradley’s Neurology in Clinical Practice. 6th ed. Philadelphia, PA: Elsevier Saunders; 2012:1703-1744.
5. Martin VT, Behbehani M. Ovarian hormones and migraine headache: understanding mechanisms and pathogenesis – part 2. Headache. 2006;46:365-386.
6. Brandes JL. The influence of estrogen on migraine: a systematic review. JAMA. 2006; 295:1824-1830.
7. Loder EW. Menstrual migraine: pathophysiology, diagnosis and impact. Headache. 2006;46 (Suppl 2):S55-S60.
8. Misakian AL, Langer RD, Bensenor IM, et al. Postmenopausal hormone therapy and migraine headache. J Women’s Health (Larchmt). 2003;12:1027-1036.
9. Pringsheim T, Gooren L. Migraine prevalence in male to female transsexuals on hormone therapy. Neurology. 2004;63:593-594.
10. Somerville BW. The role of estradiol withdrawal in the etiology of menstrual migraine. Neurology. 1972;22:355-365.
11. Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70:1555-1563.
12. Hu Y, Guan X, Fan L, et al. Triptans in prevention of menstrual migraine: a systematic review with meta-analysis. J Headache Pain. 2013;14:7.
13. Canonico M, Plu-Bureau G, Lowe GD, et al. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ. 2008;336:1227-1231.
14. Merki-Feld GS, Imthurn B, Langner R, et al. Headache frequency and intensity in female migraineurs using desogestrel-only contraception: a retrospective pilot diary study. Cephalalgia. 2013;33:340-346.
15.
16. Morotti M, Remorgida V, Venturini PL, et al. Progestin-only contraception compared with extended combined oral contraceptive in women with migraine without aura: a retrospective pilot study. Eur J Obstet Gynecol Reprod Biol. 2014;183:178-182.
17. Silberstein SD, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63:261-269.
18. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized double-blind, placebo-controlled study. Headache. 2001;41:248-256.
19. Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs. 2008;22:877-886.
20. Facchinetti F, Sances G, Borella P, et al. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31:298-301.
21. Teigen L, Boes CJ. An evidence-based review of oral magnesium supplementation in the preventive treatment of migraine. Cephalalgia. 2014;35:912-922.
22. Taylor FR. Nutraceuticals and headache: the biological basis. Headache. 2011;51:484-501.
23. Zhang XZ, Zhang L, Guo J, et al. Acupuncture as prophylaxis for menstrual-related migraine: study protocol for a multicenter randomized controlled trial. Trials. 2013;14:374.
24. MacGregor EA. Oestrogen and attacks of migraine with and without aura. Lancet Neurol. 2004;3:354-361.
25. Cole JA, Norman H, Doherty M, et al. Venous thromboembolism, myocardial infarction, and stroke among transdermal contraceptive system users. Obstet Gynecol. 2007;109:339-346.
26. MacGregor EA, Frith A, Ellis J, et al. Prevention of menstrual attacks of migraine: a double blind placebo-controlled crossover study. Neurology. 2006;67:2159-2163.
27. Sulak P, Willis S, Kuehl T, et al. Headaches and oral contraceptives: impact of eliminating the standard 7-day placebo interval. Headache. 2007;47:27-37.
28. Coffee AL, Sulak PJ, Hill AJ, et al. Extended cycle combined oral contraceptives and prophylactic frovatriptan during the hormone-free interval in women with menstrual-related migraines. J Womens Health. 2014;23:310-317.
29. Lidegaard Ø, Kreiner S. Contraceptives and cerebral thrombosis: a five-year national case-control study. Contraception. 2002;65:197-205.
30. Bousser MG. Estrogen, migraine, and stroke. Stroke. 2004;35(Suppl 1):2652-2656.
31. Gillum LA, Mamidipudi SK, Johnston SC. Ischemic stroke risk with oral contraceptives: A meta-analysis. JAMA. 2000;284:72-78.
32. Donaghy M, Chang CL, Poulter N. Duration, frequency, recency, and type of migraine and the risk of ischaemic stroke in women of childbearing age. J Neurol Neurosurg Psychiatry. 2002;73:747-750.
33. Sacco S, Ricci S, Degan D. Migraine in women: the role of hormones and their impact on vascular diseases. J Headache Pain. 2012;12:177-189.
34. Merikangas KR, Fenton BT, Cheng SH, et al. Association between migraine and stroke in a large-scale epidemiological study of the United States. Arch Neurol. 1997;54:362-368.
35. MacClellan LR, Giles W, Cole J, et al. Probable migraine with visual aura and risk of ischemic stroke: the stroke prevention in young women study. Stroke. 2007;38:2438-2445.
36. Centers for Disease Control and Prevention. U.S. Medical Eligibility Criteria for Contraceptive Use, 2010. MMWR Recomm Rep
37. Chang CL, Donaghy M, Poulter N. Migraine and stroke in young women: case-control study. The World Health Organisation Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception. BMJ. 1999;318:13-18.
38. Tzourio C, Tehindrazanarivelo A, Iglésias S, et al. Case-control study of migraine and risk of ischaemic stroke in young women. BMJ. 1995;310:830-833.
1. MacGregor EA, Rosenberg JD, Kurth T. Sex-related differences in epidemiological and clinic-based headache studies. Headache. 2011;51:843-859.
2. Stewart WF, Wood C, Reed ML, et al. Cumulative lifetime migraine incidence in women and men. Cephalalgia. 2008;28:1170-1178.
3. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd ed. Cephalalgia. 2013;33:629-808.
4. Garza I, Swanson JW, Cheshire WP Jr, et al. Headache and other craniofacial pain. In: Daroff RB, Fenichel GM, Jankovic J, et al, eds. Bradley’s Neurology in Clinical Practice. 6th ed. Philadelphia, PA: Elsevier Saunders; 2012:1703-1744.
5. Martin VT, Behbehani M. Ovarian hormones and migraine headache: understanding mechanisms and pathogenesis – part 2. Headache. 2006;46:365-386.
6. Brandes JL. The influence of estrogen on migraine: a systematic review. JAMA. 2006; 295:1824-1830.
7. Loder EW. Menstrual migraine: pathophysiology, diagnosis and impact. Headache. 2006;46 (Suppl 2):S55-S60.
8. Misakian AL, Langer RD, Bensenor IM, et al. Postmenopausal hormone therapy and migraine headache. J Women’s Health (Larchmt). 2003;12:1027-1036.
9. Pringsheim T, Gooren L. Migraine prevalence in male to female transsexuals on hormone therapy. Neurology. 2004;63:593-594.
10. Somerville BW. The role of estradiol withdrawal in the etiology of menstrual migraine. Neurology. 1972;22:355-365.
11. Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70:1555-1563.
12. Hu Y, Guan X, Fan L, et al. Triptans in prevention of menstrual migraine: a systematic review with meta-analysis. J Headache Pain. 2013;14:7.
13. Canonico M, Plu-Bureau G, Lowe GD, et al. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ. 2008;336:1227-1231.
14. Merki-Feld GS, Imthurn B, Langner R, et al. Headache frequency and intensity in female migraineurs using desogestrel-only contraception: a retrospective pilot diary study. Cephalalgia. 2013;33:340-346.
15.
16. Morotti M, Remorgida V, Venturini PL, et al. Progestin-only contraception compared with extended combined oral contraceptive in women with migraine without aura: a retrospective pilot study. Eur J Obstet Gynecol Reprod Biol. 2014;183:178-182.
17. Silberstein SD, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63:261-269.
18. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized double-blind, placebo-controlled study. Headache. 2001;41:248-256.
19. Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs. 2008;22:877-886.
20. Facchinetti F, Sances G, Borella P, et al. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31:298-301.
21. Teigen L, Boes CJ. An evidence-based review of oral magnesium supplementation in the preventive treatment of migraine. Cephalalgia. 2014;35:912-922.
22. Taylor FR. Nutraceuticals and headache: the biological basis. Headache. 2011;51:484-501.
23. Zhang XZ, Zhang L, Guo J, et al. Acupuncture as prophylaxis for menstrual-related migraine: study protocol for a multicenter randomized controlled trial. Trials. 2013;14:374.
24. MacGregor EA. Oestrogen and attacks of migraine with and without aura. Lancet Neurol. 2004;3:354-361.
25. Cole JA, Norman H, Doherty M, et al. Venous thromboembolism, myocardial infarction, and stroke among transdermal contraceptive system users. Obstet Gynecol. 2007;109:339-346.
26. MacGregor EA, Frith A, Ellis J, et al. Prevention of menstrual attacks of migraine: a double blind placebo-controlled crossover study. Neurology. 2006;67:2159-2163.
27. Sulak P, Willis S, Kuehl T, et al. Headaches and oral contraceptives: impact of eliminating the standard 7-day placebo interval. Headache. 2007;47:27-37.
28. Coffee AL, Sulak PJ, Hill AJ, et al. Extended cycle combined oral contraceptives and prophylactic frovatriptan during the hormone-free interval in women with menstrual-related migraines. J Womens Health. 2014;23:310-317.
29. Lidegaard Ø, Kreiner S. Contraceptives and cerebral thrombosis: a five-year national case-control study. Contraception. 2002;65:197-205.
30. Bousser MG. Estrogen, migraine, and stroke. Stroke. 2004;35(Suppl 1):2652-2656.
31. Gillum LA, Mamidipudi SK, Johnston SC. Ischemic stroke risk with oral contraceptives: A meta-analysis. JAMA. 2000;284:72-78.
32. Donaghy M, Chang CL, Poulter N. Duration, frequency, recency, and type of migraine and the risk of ischaemic stroke in women of childbearing age. J Neurol Neurosurg Psychiatry. 2002;73:747-750.
33. Sacco S, Ricci S, Degan D. Migraine in women: the role of hormones and their impact on vascular diseases. J Headache Pain. 2012;12:177-189.
34. Merikangas KR, Fenton BT, Cheng SH, et al. Association between migraine and stroke in a large-scale epidemiological study of the United States. Arch Neurol. 1997;54:362-368.
35. MacClellan LR, Giles W, Cole J, et al. Probable migraine with visual aura and risk of ischemic stroke: the stroke prevention in young women study. Stroke. 2007;38:2438-2445.
36. Centers for Disease Control and Prevention. U.S. Medical Eligibility Criteria for Contraceptive Use, 2010. MMWR Recomm Rep
37. Chang CL, Donaghy M, Poulter N. Migraine and stroke in young women: case-control study. The World Health Organisation Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception. BMJ. 1999;318:13-18.
38. Tzourio C, Tehindrazanarivelo A, Iglésias S, et al. Case-control study of migraine and risk of ischaemic stroke in young women. BMJ. 1995;310:830-833.
Letter to the Editor: Menopause and HT
“2016 UPDATE ON MENOPAUSE”
ANDREW M. KAUNITZ, MD (JULY 2016)
Menopause and hormone therapy
As a long-term believer (proven!) of the value of the old comment, “estrogen forever,” I was pleased to see all the positive comments about estrogen in Dr. Kaunitz’s article. I was disappointed, however, in the comments in the box (page 39), “What this evidence means for practice.”
While my prejudice, statistically supported, is old fashioned, omission of the newer and marvelous way to counteract the only bad effects of estrogen (endometrial stimulation leading to endometrial adenocarcinoma) seems to be a major oversight. The new and least (if any) side-effect method means a levonorgestrel-releasing intrauterine device (LNG-IUD) yielding local progesterone counteraction to this major side effect of estrogen therapy.
Arthur A. Fleisher II, MD
Northridge, California
Dr. Kaunitz responds
I thank Dr. Fleisher for his interest in my 2016 Update on Menopause. I agree that off-label use of the LNG-IUD represents an appropriate alternative to systemic progestin when using estrogen to treat menopausal symptoms in women with an intact uterus.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
“2016 UPDATE ON MENOPAUSE”
ANDREW M. KAUNITZ, MD (JULY 2016)
Menopause and hormone therapy
As a long-term believer (proven!) of the value of the old comment, “estrogen forever,” I was pleased to see all the positive comments about estrogen in Dr. Kaunitz’s article. I was disappointed, however, in the comments in the box (page 39), “What this evidence means for practice.”
While my prejudice, statistically supported, is old fashioned, omission of the newer and marvelous way to counteract the only bad effects of estrogen (endometrial stimulation leading to endometrial adenocarcinoma) seems to be a major oversight. The new and least (if any) side-effect method means a levonorgestrel-releasing intrauterine device (LNG-IUD) yielding local progesterone counteraction to this major side effect of estrogen therapy.
Arthur A. Fleisher II, MD
Northridge, California
Dr. Kaunitz responds
I thank Dr. Fleisher for his interest in my 2016 Update on Menopause. I agree that off-label use of the LNG-IUD represents an appropriate alternative to systemic progestin when using estrogen to treat menopausal symptoms in women with an intact uterus.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
“2016 UPDATE ON MENOPAUSE”
ANDREW M. KAUNITZ, MD (JULY 2016)
Menopause and hormone therapy
As a long-term believer (proven!) of the value of the old comment, “estrogen forever,” I was pleased to see all the positive comments about estrogen in Dr. Kaunitz’s article. I was disappointed, however, in the comments in the box (page 39), “What this evidence means for practice.”
While my prejudice, statistically supported, is old fashioned, omission of the newer and marvelous way to counteract the only bad effects of estrogen (endometrial stimulation leading to endometrial adenocarcinoma) seems to be a major oversight. The new and least (if any) side-effect method means a levonorgestrel-releasing intrauterine device (LNG-IUD) yielding local progesterone counteraction to this major side effect of estrogen therapy.
Arthur A. Fleisher II, MD
Northridge, California
Dr. Kaunitz responds
I thank Dr. Fleisher for his interest in my 2016 Update on Menopause. I agree that off-label use of the LNG-IUD represents an appropriate alternative to systemic progestin when using estrogen to treat menopausal symptoms in women with an intact uterus.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Letter to the Editor: Tubal occlusion device removal
VIDEO: “LAPAROSCOPIC SALPINGECTOMY AND CORNUAL RESECTION REPURPOSED: A NOVEL APPROACH TO TUBAL OCCLUSION DEVICE REMOVAL”
MICHELLE PACIS, MD, MPH (JULY 2016)
Easier technique for removing tubal occlusion devices?
My patient’s rheumatologist recently asked me to remove the tubal occlusion device (Essure) inserts that I had placed approximately 5 years ago. I think the technique I used was a little easier than the one shown in the video by Dr. Pacis and featured by Dr. Advincula in his video series. I started with a standard salpingectomy from the fimbriated end, as did the technique in the video. Then I made a circumferential incision of the tubal serosa at the junction of the tube as it enters the cornua, taking care to not cut the device insert, which could be visualized and felt with cold shears. The proximal end of the device insert, including the post and coil, then easily pulled out with some elongation of the coil. Since I did not need to resect the cornua, I was able to easily seal off the small defect without need to suture.
Alexander Lin, MD
Chicago, Illinois
Dr. Pacis responds
Thank you for sharing your method for tubal occlusion device removal. Your technique would certainly work for devices that reside predominantly in the tube. We have found that many of the devices become quite anchored and adherent to the tubal mucosa. While there are many surgical approaches to device removal, our preference is to perform salpingectomy with cornual resection, so as to avoid traction on the microinsert, and remove the device intact. We are then able to give the specimen, which contains the insert, to pathology so they can comment on the status of the device.
VIDEO: “LAPAROSCOPIC SALPINGECTOMY AND CORNUAL RESECTION REPURPOSED: A NOVEL APPROACH TO TUBAL OCCLUSION DEVICE REMOVAL”
MICHELLE PACIS, MD, MPH (JULY 2016)
Easier technique for removing tubal occlusion devices?
My patient’s rheumatologist recently asked me to remove the tubal occlusion device (Essure) inserts that I had placed approximately 5 years ago. I think the technique I used was a little easier than the one shown in the video by Dr. Pacis and featured by Dr. Advincula in his video series. I started with a standard salpingectomy from the fimbriated end, as did the technique in the video. Then I made a circumferential incision of the tubal serosa at the junction of the tube as it enters the cornua, taking care to not cut the device insert, which could be visualized and felt with cold shears. The proximal end of the device insert, including the post and coil, then easily pulled out with some elongation of the coil. Since I did not need to resect the cornua, I was able to easily seal off the small defect without need to suture.
Alexander Lin, MD
Chicago, Illinois
Dr. Pacis responds
Thank you for sharing your method for tubal occlusion device removal. Your technique would certainly work for devices that reside predominantly in the tube. We have found that many of the devices become quite anchored and adherent to the tubal mucosa. While there are many surgical approaches to device removal, our preference is to perform salpingectomy with cornual resection, so as to avoid traction on the microinsert, and remove the device intact. We are then able to give the specimen, which contains the insert, to pathology so they can comment on the status of the device.
VIDEO: “LAPAROSCOPIC SALPINGECTOMY AND CORNUAL RESECTION REPURPOSED: A NOVEL APPROACH TO TUBAL OCCLUSION DEVICE REMOVAL”
MICHELLE PACIS, MD, MPH (JULY 2016)
Easier technique for removing tubal occlusion devices?
My patient’s rheumatologist recently asked me to remove the tubal occlusion device (Essure) inserts that I had placed approximately 5 years ago. I think the technique I used was a little easier than the one shown in the video by Dr. Pacis and featured by Dr. Advincula in his video series. I started with a standard salpingectomy from the fimbriated end, as did the technique in the video. Then I made a circumferential incision of the tubal serosa at the junction of the tube as it enters the cornua, taking care to not cut the device insert, which could be visualized and felt with cold shears. The proximal end of the device insert, including the post and coil, then easily pulled out with some elongation of the coil. Since I did not need to resect the cornua, I was able to easily seal off the small defect without need to suture.
Alexander Lin, MD
Chicago, Illinois
Dr. Pacis responds
Thank you for sharing your method for tubal occlusion device removal. Your technique would certainly work for devices that reside predominantly in the tube. We have found that many of the devices become quite anchored and adherent to the tubal mucosa. While there are many surgical approaches to device removal, our preference is to perform salpingectomy with cornual resection, so as to avoid traction on the microinsert, and remove the device intact. We are then able to give the specimen, which contains the insert, to pathology so they can comment on the status of the device.
Letter to the Editor: Therapeutic hypothermia for newborns
“PROTECTING THE NEWBORN BRAIN—THE FINAL FRONTIER IN OBSTETRIC AND NEONATAL CARE”
ROBERT L. BARBIERI, MD (AUGUST 2016)
Therapeutic hypothermia
I practice in a small community hospital without a neonatal intensive care unit (NICU). We have always paid attention to warming neonates. Although we cannot start neonatal therapeutic hypothermia, as Dr. Barbieri discusses in his August Editorial, would there be any benefit to avoiding purposefully warming infants who are depressed at birth? NICU care requires a pediatric transport team, which takes at least an hour to arrive.
Jane Dawson, MD
Maryville, Missouri
Dr. Barbieri responds
I thank Dr. Dawson for her observations and query. I agree that at a hospital without a NICU, pending the arrival of a pediatric transport team, clinicians should strive to prevent hyperthermia in a newborn with encephalopathy because hyperthermia might exacerbate the ischemic injury. It may make sense to avoid aggressive warming of the newborn to permit the core temperature to decrease in order to begin the hypothermia process.
“PROTECTING THE NEWBORN BRAIN—THE FINAL FRONTIER IN OBSTETRIC AND NEONATAL CARE”
ROBERT L. BARBIERI, MD (AUGUST 2016)
Therapeutic hypothermia
I practice in a small community hospital without a neonatal intensive care unit (NICU). We have always paid attention to warming neonates. Although we cannot start neonatal therapeutic hypothermia, as Dr. Barbieri discusses in his August Editorial, would there be any benefit to avoiding purposefully warming infants who are depressed at birth? NICU care requires a pediatric transport team, which takes at least an hour to arrive.
Jane Dawson, MD
Maryville, Missouri
Dr. Barbieri responds
I thank Dr. Dawson for her observations and query. I agree that at a hospital without a NICU, pending the arrival of a pediatric transport team, clinicians should strive to prevent hyperthermia in a newborn with encephalopathy because hyperthermia might exacerbate the ischemic injury. It may make sense to avoid aggressive warming of the newborn to permit the core temperature to decrease in order to begin the hypothermia process.
“PROTECTING THE NEWBORN BRAIN—THE FINAL FRONTIER IN OBSTETRIC AND NEONATAL CARE”
ROBERT L. BARBIERI, MD (AUGUST 2016)
Therapeutic hypothermia
I practice in a small community hospital without a neonatal intensive care unit (NICU). We have always paid attention to warming neonates. Although we cannot start neonatal therapeutic hypothermia, as Dr. Barbieri discusses in his August Editorial, would there be any benefit to avoiding purposefully warming infants who are depressed at birth? NICU care requires a pediatric transport team, which takes at least an hour to arrive.
Jane Dawson, MD
Maryville, Missouri
Dr. Barbieri responds
I thank Dr. Dawson for her observations and query. I agree that at a hospital without a NICU, pending the arrival of a pediatric transport team, clinicians should strive to prevent hyperthermia in a newborn with encephalopathy because hyperthermia might exacerbate the ischemic injury. It may make sense to avoid aggressive warming of the newborn to permit the core temperature to decrease in order to begin the hypothermia process.
Letters to the Editor: Managing impacted fetal head at cesarean
“STOP USING INSTRUMENTS TO ASSIST WITH DELIVERY OF THE HEAD AT CESAREAN; START DISENGAGING THE HEAD PRIOR TO SURGERY”
ERROL R. NORWITZ, MD, PHD, MBA (AUGUST 2016)
Patient positioning helps in managing impacted fetal head
As a general practice ObGyn, I have seen an increasing incidence of difficult cesareans as a result of prolonged second stage of labor. Dr. Norwitz cites this increase in his article. I have found that trying to elevate the fetal head prior to the start of surgery has been remarkably ineffective. In my practice, I place all my patients with second-stage arrest in low lithotomy stirrups (“blue fins”); this allows the nurses easier access to the vagina to elevate the head at surgery while I am reaching down from above. Usually, this facilitates delivery. It also allows better assessment of blood loss through the vagina as the cesarean progresses, and it makes placement of a Bakri balloon easier if necessary. If stirrups are not available, the patient can be placed in frog leg positioning so that my assistant can reach down and elevate the head if necessary. I find that in a patient with a very small pelvis, it is hard to get my hand down to the baby’s head. I have not yet done a breech extraction, but I know it is possible. I would probably try nitroglycerin first.
I think that difficult cesarean delivery is much more common than difficult shoulder dystocia, and we should develop standard procedures for addressing the issue and use simulation models to practice. In my time-out prior to surgery, I discuss my concerns so that everyone is ready for it, including the anesthesiologist/CRNA, and we have nitroglycerin available to relax the uterus if necessary. I hope that the American College of Obstetricians and Gynecologists (ACOG) will develop a committee opinion about this very important issue.
Marguerite P. Cohen, MD
Portland, Oregon
Assistant is key in disengaging fetal head
Disengaging the head by an assistant during a cesarean delivery is probably the most successful and useful method for managing an impacted fetal head at cesarean. The disengagement of the head prior to cesarean is practiced routinely in Europe, where forceps delivery is frequently performed. However, the disengagement should be done in the operating room (OR) just prior to or during the cesarean. To perform this in the delivery room, as suggested in Dr. Norwitz’s article, risks the associated fetal bradycardia due to head compression that might compromise an already compromised fetus. In addition, there is a risk of cord prolapse or release of excessive amniotic fluid resulting in cord compression. Also, in many hospitals in the United States, there is some delay to perform the cesarean because the OR is on a different floor from the labor and delivery room and the OR staff come from home.
Vacuum extraction can be safely used for the extraction of the head if it is not possible to deliver it manually. However, the head should be manually disimpacted and rotated to occiput anterior prior to application of the vacuum. But the presence of caput might pose some difficulty with proper application and traction.
It is important to remember that the risk factors for an impacted fetal head are also risk factors for postoperative infection. Therefore, vaginal preparation with antiseptic solution should be considered prior to cesarean delivery for all patients in labor.1
Raymond Michael, MD
Marshall, Minnesota
Reference
- Haas DM, Morgan Al Darei S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2010;3:CD007892.
“STOP USING INSTRUMENTS TO ASSIST WITH DELIVERY OF THE HEAD AT CESAREAN; START DISENGAGING THE HEAD PRIOR TO SURGERY”
ERROL R. NORWITZ, MD, PHD, MBA (AUGUST 2016)
Patient positioning helps in managing impacted fetal head
As a general practice ObGyn, I have seen an increasing incidence of difficult cesareans as a result of prolonged second stage of labor. Dr. Norwitz cites this increase in his article. I have found that trying to elevate the fetal head prior to the start of surgery has been remarkably ineffective. In my practice, I place all my patients with second-stage arrest in low lithotomy stirrups (“blue fins”); this allows the nurses easier access to the vagina to elevate the head at surgery while I am reaching down from above. Usually, this facilitates delivery. It also allows better assessment of blood loss through the vagina as the cesarean progresses, and it makes placement of a Bakri balloon easier if necessary. If stirrups are not available, the patient can be placed in frog leg positioning so that my assistant can reach down and elevate the head if necessary. I find that in a patient with a very small pelvis, it is hard to get my hand down to the baby’s head. I have not yet done a breech extraction, but I know it is possible. I would probably try nitroglycerin first.
I think that difficult cesarean delivery is much more common than difficult shoulder dystocia, and we should develop standard procedures for addressing the issue and use simulation models to practice. In my time-out prior to surgery, I discuss my concerns so that everyone is ready for it, including the anesthesiologist/CRNA, and we have nitroglycerin available to relax the uterus if necessary. I hope that the American College of Obstetricians and Gynecologists (ACOG) will develop a committee opinion about this very important issue.
Marguerite P. Cohen, MD
Portland, Oregon
Assistant is key in disengaging fetal head
Disengaging the head by an assistant during a cesarean delivery is probably the most successful and useful method for managing an impacted fetal head at cesarean. The disengagement of the head prior to cesarean is practiced routinely in Europe, where forceps delivery is frequently performed. However, the disengagement should be done in the operating room (OR) just prior to or during the cesarean. To perform this in the delivery room, as suggested in Dr. Norwitz’s article, risks the associated fetal bradycardia due to head compression that might compromise an already compromised fetus. In addition, there is a risk of cord prolapse or release of excessive amniotic fluid resulting in cord compression. Also, in many hospitals in the United States, there is some delay to perform the cesarean because the OR is on a different floor from the labor and delivery room and the OR staff come from home.
Vacuum extraction can be safely used for the extraction of the head if it is not possible to deliver it manually. However, the head should be manually disimpacted and rotated to occiput anterior prior to application of the vacuum. But the presence of caput might pose some difficulty with proper application and traction.
It is important to remember that the risk factors for an impacted fetal head are also risk factors for postoperative infection. Therefore, vaginal preparation with antiseptic solution should be considered prior to cesarean delivery for all patients in labor.1
Raymond Michael, MD
Marshall, Minnesota
Reference
- Haas DM, Morgan Al Darei S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2010;3:CD007892.
“STOP USING INSTRUMENTS TO ASSIST WITH DELIVERY OF THE HEAD AT CESAREAN; START DISENGAGING THE HEAD PRIOR TO SURGERY”
ERROL R. NORWITZ, MD, PHD, MBA (AUGUST 2016)
Patient positioning helps in managing impacted fetal head
As a general practice ObGyn, I have seen an increasing incidence of difficult cesareans as a result of prolonged second stage of labor. Dr. Norwitz cites this increase in his article. I have found that trying to elevate the fetal head prior to the start of surgery has been remarkably ineffective. In my practice, I place all my patients with second-stage arrest in low lithotomy stirrups (“blue fins”); this allows the nurses easier access to the vagina to elevate the head at surgery while I am reaching down from above. Usually, this facilitates delivery. It also allows better assessment of blood loss through the vagina as the cesarean progresses, and it makes placement of a Bakri balloon easier if necessary. If stirrups are not available, the patient can be placed in frog leg positioning so that my assistant can reach down and elevate the head if necessary. I find that in a patient with a very small pelvis, it is hard to get my hand down to the baby’s head. I have not yet done a breech extraction, but I know it is possible. I would probably try nitroglycerin first.
I think that difficult cesarean delivery is much more common than difficult shoulder dystocia, and we should develop standard procedures for addressing the issue and use simulation models to practice. In my time-out prior to surgery, I discuss my concerns so that everyone is ready for it, including the anesthesiologist/CRNA, and we have nitroglycerin available to relax the uterus if necessary. I hope that the American College of Obstetricians and Gynecologists (ACOG) will develop a committee opinion about this very important issue.
Marguerite P. Cohen, MD
Portland, Oregon
Assistant is key in disengaging fetal head
Disengaging the head by an assistant during a cesarean delivery is probably the most successful and useful method for managing an impacted fetal head at cesarean. The disengagement of the head prior to cesarean is practiced routinely in Europe, where forceps delivery is frequently performed. However, the disengagement should be done in the operating room (OR) just prior to or during the cesarean. To perform this in the delivery room, as suggested in Dr. Norwitz’s article, risks the associated fetal bradycardia due to head compression that might compromise an already compromised fetus. In addition, there is a risk of cord prolapse or release of excessive amniotic fluid resulting in cord compression. Also, in many hospitals in the United States, there is some delay to perform the cesarean because the OR is on a different floor from the labor and delivery room and the OR staff come from home.
Vacuum extraction can be safely used for the extraction of the head if it is not possible to deliver it manually. However, the head should be manually disimpacted and rotated to occiput anterior prior to application of the vacuum. But the presence of caput might pose some difficulty with proper application and traction.
It is important to remember that the risk factors for an impacted fetal head are also risk factors for postoperative infection. Therefore, vaginal preparation with antiseptic solution should be considered prior to cesarean delivery for all patients in labor.1
Raymond Michael, MD
Marshall, Minnesota
Reference
- Haas DM, Morgan Al Darei S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2010;3:CD007892.
Product Update: SureSound+ device; LILETTA single-handed inserter
PRECISE PREABLATION MEASUREMENT
The SureSound + device is designed to allow a physician to measure cavity length with one click, unlike traditional sounding methods that require multiple calculations, says Hologic. Hologic indicates that the device is inserted through the cervical canal, the Malecot anchor is deployed on the internal os, and the inner probe’s polyethylene tip is extended to the fundus to gauge uterine cavity length. The single-use device also may reduce the risk of contamination, says Hologic.
The manufacturer indicates the NovaSure procedure is appropriate for treating premenopausal women with heavy periods due to benign causes who have finished childbearing.
FOR MORE INFORMATION, VISIT: http://www.novasure.com/hcp/suresound_plus
NEW LILETTA INSERTER
The companies indicate that the LILETTA IUD is for use by women wishing to prevent pregnancy for up to 3 years regardless of parity or body mass index, with a cumulative 3-year efficacy rate of 99.45%. Approval of the new LILETTA single-handed inserter was based on the ACCESS IUS study that enrolled 1,751 US women and also led to LILETTA’s approval. Product marketing materials state that, during the ACCESS IUS study, the success rate for insertions with the new inserter was 99.2%. The original inserter requires a 2-handed technique.
According to Allergan and Medicines360, the new inserter includes: an ergonomic design allowing for single-handed insertion that can be used with either hand, a bendable tube to accommodate the anatomy of the patient during insertion, and the ability to reload the device if needed before insertion.
FOR MORE INFORMATION, VISIT: https://www.liletta.com
PRECISE PREABLATION MEASUREMENT
The SureSound + device is designed to allow a physician to measure cavity length with one click, unlike traditional sounding methods that require multiple calculations, says Hologic. Hologic indicates that the device is inserted through the cervical canal, the Malecot anchor is deployed on the internal os, and the inner probe’s polyethylene tip is extended to the fundus to gauge uterine cavity length. The single-use device also may reduce the risk of contamination, says Hologic.
The manufacturer indicates the NovaSure procedure is appropriate for treating premenopausal women with heavy periods due to benign causes who have finished childbearing.
FOR MORE INFORMATION, VISIT: http://www.novasure.com/hcp/suresound_plus
NEW LILETTA INSERTER
The companies indicate that the LILETTA IUD is for use by women wishing to prevent pregnancy for up to 3 years regardless of parity or body mass index, with a cumulative 3-year efficacy rate of 99.45%. Approval of the new LILETTA single-handed inserter was based on the ACCESS IUS study that enrolled 1,751 US women and also led to LILETTA’s approval. Product marketing materials state that, during the ACCESS IUS study, the success rate for insertions with the new inserter was 99.2%. The original inserter requires a 2-handed technique.
According to Allergan and Medicines360, the new inserter includes: an ergonomic design allowing for single-handed insertion that can be used with either hand, a bendable tube to accommodate the anatomy of the patient during insertion, and the ability to reload the device if needed before insertion.
FOR MORE INFORMATION, VISIT: https://www.liletta.com
PRECISE PREABLATION MEASUREMENT
The SureSound + device is designed to allow a physician to measure cavity length with one click, unlike traditional sounding methods that require multiple calculations, says Hologic. Hologic indicates that the device is inserted through the cervical canal, the Malecot anchor is deployed on the internal os, and the inner probe’s polyethylene tip is extended to the fundus to gauge uterine cavity length. The single-use device also may reduce the risk of contamination, says Hologic.
The manufacturer indicates the NovaSure procedure is appropriate for treating premenopausal women with heavy periods due to benign causes who have finished childbearing.
FOR MORE INFORMATION, VISIT: http://www.novasure.com/hcp/suresound_plus
NEW LILETTA INSERTER
The companies indicate that the LILETTA IUD is for use by women wishing to prevent pregnancy for up to 3 years regardless of parity or body mass index, with a cumulative 3-year efficacy rate of 99.45%. Approval of the new LILETTA single-handed inserter was based on the ACCESS IUS study that enrolled 1,751 US women and also led to LILETTA’s approval. Product marketing materials state that, during the ACCESS IUS study, the success rate for insertions with the new inserter was 99.2%. The original inserter requires a 2-handed technique.
According to Allergan and Medicines360, the new inserter includes: an ergonomic design allowing for single-handed insertion that can be used with either hand, a bendable tube to accommodate the anatomy of the patient during insertion, and the ability to reload the device if needed before insertion.
FOR MORE INFORMATION, VISIT: https://www.liletta.com
FDA approves first automated insulin delivery system
The Food and Drug Administration has approved the first automated insulin delivery device for patients aged 14 years and older with type 1 diabetes.
The MiniMed 670G by Medtronic – an “artificial pancreas” – is a hybrid closed-loop system designed to automatically monitor glucose and deliver appropriate basal insulin doses. The system was shown in a study of 123 individuals with type 1 diabetes to be safe for those aged 14 and older; no serious adverse events, diabetic ketoacidosis, or severe hypoglycemia occurred during a 3-month period when the system was used as frequently as possible by study subjects.
“This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin,” Jeffrey Shuren, MD, director of the FDA’s Center for Devices and Radiological Health, said in a press statement.
As part of its approval of the device, the FDA is requiring a postmarket study of its performance in real-world settings, and although the approved version of the device is unsafe for use in children aged 6 years and younger and in those who require fewer than eight units of insulin daily, Medtronic is currently evaluating the safety and efficacy of the device in children aged 7-13 years.
The Food and Drug Administration has approved the first automated insulin delivery device for patients aged 14 years and older with type 1 diabetes.
The MiniMed 670G by Medtronic – an “artificial pancreas” – is a hybrid closed-loop system designed to automatically monitor glucose and deliver appropriate basal insulin doses. The system was shown in a study of 123 individuals with type 1 diabetes to be safe for those aged 14 and older; no serious adverse events, diabetic ketoacidosis, or severe hypoglycemia occurred during a 3-month period when the system was used as frequently as possible by study subjects.
“This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin,” Jeffrey Shuren, MD, director of the FDA’s Center for Devices and Radiological Health, said in a press statement.
As part of its approval of the device, the FDA is requiring a postmarket study of its performance in real-world settings, and although the approved version of the device is unsafe for use in children aged 6 years and younger and in those who require fewer than eight units of insulin daily, Medtronic is currently evaluating the safety and efficacy of the device in children aged 7-13 years.
The Food and Drug Administration has approved the first automated insulin delivery device for patients aged 14 years and older with type 1 diabetes.
The MiniMed 670G by Medtronic – an “artificial pancreas” – is a hybrid closed-loop system designed to automatically monitor glucose and deliver appropriate basal insulin doses. The system was shown in a study of 123 individuals with type 1 diabetes to be safe for those aged 14 and older; no serious adverse events, diabetic ketoacidosis, or severe hypoglycemia occurred during a 3-month period when the system was used as frequently as possible by study subjects.
“This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin,” Jeffrey Shuren, MD, director of the FDA’s Center for Devices and Radiological Health, said in a press statement.
As part of its approval of the device, the FDA is requiring a postmarket study of its performance in real-world settings, and although the approved version of the device is unsafe for use in children aged 6 years and younger and in those who require fewer than eight units of insulin daily, Medtronic is currently evaluating the safety and efficacy of the device in children aged 7-13 years.
Long-term breast cancer studies yield encouraging data for recurrence, survival
Four pivotal breast cancer trials were presented at the 2016 annual meeting of the American Society of Clinical Oncology in Chicago. The MA.17R trial, which was the plenary talk by Dr Paul Goss, looked at extending adjuvant aromatase inhibitors to 10 years or beyond in postmenopausal women; two presentations reported on mutations after progression in metastatic breast cancer, one on first-line AIs and the other on prior endocrine therapy (PALOMA-3); and results from the Z0011 trial showed that sentinel lymph node dissection without axillary lymph node dissection might show promising 10-year loco-regional control and survival outcomes.
Click on the PDF icon at the top of this introduction to read the full article.
Four pivotal breast cancer trials were presented at the 2016 annual meeting of the American Society of Clinical Oncology in Chicago. The MA.17R trial, which was the plenary talk by Dr Paul Goss, looked at extending adjuvant aromatase inhibitors to 10 years or beyond in postmenopausal women; two presentations reported on mutations after progression in metastatic breast cancer, one on first-line AIs and the other on prior endocrine therapy (PALOMA-3); and results from the Z0011 trial showed that sentinel lymph node dissection without axillary lymph node dissection might show promising 10-year loco-regional control and survival outcomes.
Click on the PDF icon at the top of this introduction to read the full article.
Four pivotal breast cancer trials were presented at the 2016 annual meeting of the American Society of Clinical Oncology in Chicago. The MA.17R trial, which was the plenary talk by Dr Paul Goss, looked at extending adjuvant aromatase inhibitors to 10 years or beyond in postmenopausal women; two presentations reported on mutations after progression in metastatic breast cancer, one on first-line AIs and the other on prior endocrine therapy (PALOMA-3); and results from the Z0011 trial showed that sentinel lymph node dissection without axillary lymph node dissection might show promising 10-year loco-regional control and survival outcomes.
Click on the PDF icon at the top of this introduction to read the full article.
Anticipate, treat GI issues in scleroderma
LAS VEGAS – Pay attention to GI symptoms in your scleroderma patients. Gut involvement in scleroderma, a result of vasculopathy and fibrosis affecting the GI tract, is common and can be debilitating, said Daniel Furst, MD.
Speaking at the annual Perspectives in Rheumatologic Diseases presented by the Global Academy for Medical Education, Dr. Furst said that the progression often begins at the mouth and esophagus, and progresses through the digestive system, eventually reaching the rectum and anus.
“You think about motility issues early on, in the esophagus,” and early oral symptoms can include mouth dryness, said Dr. Furst, who is associated with the University of California, Los Angeles, the University of Washington, Seattle, and the University of Florence (Italy).
As scleroderma begins to affect the midgut, Dr. Furst said that the secondary results of the decrease in motility are symptoms such as heartburn, nausea, vomiting, and early satiety.
“When you have a decrease in motility, then the normal ... housekeeping waves of the midgut and the colon are decreased,” he said. Bacteria from the colon can then invade the small bowel, causing overgrowth of the midgut by species not normally seen there. Not only gas, but also malnutrition can eventually result, he said.
When scleroderma affects the lower gut, patients can have bloating, diarrhea, and constipation, and finally, incontinence of the bowel, a condition with often devastating psychosocial consequences.
The choice of promotility agents depends on the area affected; erythromycin and metoclopramide help in the upper GI tract, while tegaserod can be helpful in the lower gut.
Global Academy for Medical Education and this news organization are owned by the same parent company.
On Twitter @karioakes
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Pay attention to GI symptoms in your scleroderma patients. Gut involvement in scleroderma, a result of vasculopathy and fibrosis affecting the GI tract, is common and can be debilitating, said Daniel Furst, MD.
Speaking at the annual Perspectives in Rheumatologic Diseases presented by the Global Academy for Medical Education, Dr. Furst said that the progression often begins at the mouth and esophagus, and progresses through the digestive system, eventually reaching the rectum and anus.
“You think about motility issues early on, in the esophagus,” and early oral symptoms can include mouth dryness, said Dr. Furst, who is associated with the University of California, Los Angeles, the University of Washington, Seattle, and the University of Florence (Italy).
As scleroderma begins to affect the midgut, Dr. Furst said that the secondary results of the decrease in motility are symptoms such as heartburn, nausea, vomiting, and early satiety.
“When you have a decrease in motility, then the normal ... housekeeping waves of the midgut and the colon are decreased,” he said. Bacteria from the colon can then invade the small bowel, causing overgrowth of the midgut by species not normally seen there. Not only gas, but also malnutrition can eventually result, he said.
When scleroderma affects the lower gut, patients can have bloating, diarrhea, and constipation, and finally, incontinence of the bowel, a condition with often devastating psychosocial consequences.
The choice of promotility agents depends on the area affected; erythromycin and metoclopramide help in the upper GI tract, while tegaserod can be helpful in the lower gut.
Global Academy for Medical Education and this news organization are owned by the same parent company.
On Twitter @karioakes
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Pay attention to GI symptoms in your scleroderma patients. Gut involvement in scleroderma, a result of vasculopathy and fibrosis affecting the GI tract, is common and can be debilitating, said Daniel Furst, MD.
Speaking at the annual Perspectives in Rheumatologic Diseases presented by the Global Academy for Medical Education, Dr. Furst said that the progression often begins at the mouth and esophagus, and progresses through the digestive system, eventually reaching the rectum and anus.
“You think about motility issues early on, in the esophagus,” and early oral symptoms can include mouth dryness, said Dr. Furst, who is associated with the University of California, Los Angeles, the University of Washington, Seattle, and the University of Florence (Italy).
As scleroderma begins to affect the midgut, Dr. Furst said that the secondary results of the decrease in motility are symptoms such as heartburn, nausea, vomiting, and early satiety.
“When you have a decrease in motility, then the normal ... housekeeping waves of the midgut and the colon are decreased,” he said. Bacteria from the colon can then invade the small bowel, causing overgrowth of the midgut by species not normally seen there. Not only gas, but also malnutrition can eventually result, he said.
When scleroderma affects the lower gut, patients can have bloating, diarrhea, and constipation, and finally, incontinence of the bowel, a condition with often devastating psychosocial consequences.
The choice of promotility agents depends on the area affected; erythromycin and metoclopramide help in the upper GI tract, while tegaserod can be helpful in the lower gut.
Global Academy for Medical Education and this news organization are owned by the same parent company.
On Twitter @karioakes
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE ANNUAL PERSPECTIVES IN RHEUMATIC DISEASES
Cannabis use after first-episode psychosis may raise relapse risk
Does habitual cannabis use trigger psychosis, or does a tendency toward psychosis predict increased cannabis use?
“[Our] results suggest that it is more likely than not that continued cannabis use after onset of psychosis is causally associated with increased risk of relapse of psychosis, resulting in psychiatric hospitalization,” reported the authors of a study published online Sept. 28 in JAMA Psychiatry.
It is well established that cannabis use after first-episode psychosis is associated with poor outcomes; what is not so well established is why. The three most prominent theories so far are that cannabis use and psychosis share inherent genetic and/or environmental vulnerabilities, impending psychosis impels increased cannabis use, and conversely, cannabis use has a causal effect on psychosis (Lancet Psychiatry. 2016:3[5]:401) and (Lancet Psychiatry. 2015;3[5]401-2).
After conducting a 10-year, prospective cohort investigation of 220 people who presented to psychiatric services at numerous sites in London between April 2002 and July 2013, Tabea Schoeler and her associates in the current study found that the odds of a relapse of psychosis during periods of cannabis use was increased by 1.13, compared with periods of no cannabis use (95% confidence interval, 1.03-1.24) (JAMA Psychiatry. 2016 Sep 28. doi: 10.1001/jamapsychiatry.2016.2427). Further, those who used cannabis continually after first-episode psychosis were found to have a 59.1% higher risk of relapsing psychosis in that 10-year period, compared with a 28.5% increased risk in those who quit using cannabis entirely (P less than .001).
The only true way to measure causality in this area would be to administer cannabis to participants in a randomized, clinically controlled trial, thus challenging the ethics of clinical study. Instead, Ms. Schoeler of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London and her associates – including Sir Robin M. Murray, MD, a psychiatrist who has been a leading voice in favor of the view that cannabis use and psychotic episodes are often linked – used patient self-reports to assess changes in cannabis use over time. This allowed the investigators to more clearly differentiate between confounders that cannot be readily observed, and that are not likely to change over time, such as any shared genetic or environmental factors, and confounders that can be observed and that do have the potential to change, such as the change in cannabis use status over time, including nonuser vs. user status, and the pattern and frequency of use after onset of illness.
The data were collected in two separate assessments, one was conducted near the time of illness onset, and again at least 2 years after the first episode of psychosis. If interviews were not possible to complete in person, they were conducted over the phone. These data were combined with patient clinical records across the study period. Frequency of cannabis use was measured based on the Cannabis Experience Questionnaire. A patient was considered to have relapsed if he or she was admitted to an inpatient psychiatric facility for exacerbated symptoms of psychosis within 2 years of the first episode of psychosis.
As for the study population, 59.1% of whom were men with an average age of 29 years, there were no significant differences as to whether they had been diagnosed with either affective or nonaffective psychosis. However, there were significant differences between the risk of relapse and the frequency of cannabis use 2 years after onset of illness. In the nonuser group, 28.5% of patients relapsed. In the intermittent user group, 36% relapsed. In the continuous use group, 59.1% relapsed (P less than .001).
Differences also were found across the three groups in medication adherence rates. Among the nonusers, 47.7% reportedly complied with treatment. The rate was 32% among intermittent users and 20.5% among habitual users (P = .02). Illicit use of other drugs was also higher in the continuous cannabis use group: 27.3% vs. 4% in the cannabis nonuse group and 12% in the intermittent use group (P less than .001).
Also noteworthy was the age of psychosis onset across the groups. While the average age of onset across the study was 28.62 years, for habitual users the average age of illness onset was 25.44 years, compared with 29.52 in nonusers and 28.79 in intermittent users (P = .02).
These results should be viewed in light of their retrospective and self-reported nature – neither of which take into account either premorbid cannabis use or age of illness onset as either predictors or moderators – but the data suggest a dose-response relationship between cannabis use and psychosis, according to the investigators. If so, the implications of the data could reach beyond patient outcomes.
“Because cannabis use is a potentially modifiable risk factor that has an adverse influence on the risk of relapse of psychosis and hospitalization in a given individual, with limited efficacy of existing interventions, these results underscore the importance of developing novel intervention strategies and demand urgent attention from clinicians and health care policy makers,” the authors wrote.
Whether the study conclusively demonstrates that it is cannabis use – and not genetic liability or reverse causation – that drives relapse of psychosis is still not clear. By the investigators’ own admission, the effect of discontinuing cannabis use on patient outcomes has not been tested, leaving the question of a definite causal relationship between continual cannabis use and relapse of psychosis still open for debate.
Ms. Schoeler and the other investigators – with the exception of Dr. Murray – reported no conflicts of interest. Dr. Murray reported that he has received honoraria from Janssen, Sunovion, Otsuka, and Lundbeck. The study was funded in large part by the U.K.’s National Institute for Health Research and by King’s College London.
On Twitter @whitneymcknight
Does habitual cannabis use trigger psychosis, or does a tendency toward psychosis predict increased cannabis use?
“[Our] results suggest that it is more likely than not that continued cannabis use after onset of psychosis is causally associated with increased risk of relapse of psychosis, resulting in psychiatric hospitalization,” reported the authors of a study published online Sept. 28 in JAMA Psychiatry.
It is well established that cannabis use after first-episode psychosis is associated with poor outcomes; what is not so well established is why. The three most prominent theories so far are that cannabis use and psychosis share inherent genetic and/or environmental vulnerabilities, impending psychosis impels increased cannabis use, and conversely, cannabis use has a causal effect on psychosis (Lancet Psychiatry. 2016:3[5]:401) and (Lancet Psychiatry. 2015;3[5]401-2).
After conducting a 10-year, prospective cohort investigation of 220 people who presented to psychiatric services at numerous sites in London between April 2002 and July 2013, Tabea Schoeler and her associates in the current study found that the odds of a relapse of psychosis during periods of cannabis use was increased by 1.13, compared with periods of no cannabis use (95% confidence interval, 1.03-1.24) (JAMA Psychiatry. 2016 Sep 28. doi: 10.1001/jamapsychiatry.2016.2427). Further, those who used cannabis continually after first-episode psychosis were found to have a 59.1% higher risk of relapsing psychosis in that 10-year period, compared with a 28.5% increased risk in those who quit using cannabis entirely (P less than .001).
The only true way to measure causality in this area would be to administer cannabis to participants in a randomized, clinically controlled trial, thus challenging the ethics of clinical study. Instead, Ms. Schoeler of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London and her associates – including Sir Robin M. Murray, MD, a psychiatrist who has been a leading voice in favor of the view that cannabis use and psychotic episodes are often linked – used patient self-reports to assess changes in cannabis use over time. This allowed the investigators to more clearly differentiate between confounders that cannot be readily observed, and that are not likely to change over time, such as any shared genetic or environmental factors, and confounders that can be observed and that do have the potential to change, such as the change in cannabis use status over time, including nonuser vs. user status, and the pattern and frequency of use after onset of illness.
The data were collected in two separate assessments, one was conducted near the time of illness onset, and again at least 2 years after the first episode of psychosis. If interviews were not possible to complete in person, they were conducted over the phone. These data were combined with patient clinical records across the study period. Frequency of cannabis use was measured based on the Cannabis Experience Questionnaire. A patient was considered to have relapsed if he or she was admitted to an inpatient psychiatric facility for exacerbated symptoms of psychosis within 2 years of the first episode of psychosis.
As for the study population, 59.1% of whom were men with an average age of 29 years, there were no significant differences as to whether they had been diagnosed with either affective or nonaffective psychosis. However, there were significant differences between the risk of relapse and the frequency of cannabis use 2 years after onset of illness. In the nonuser group, 28.5% of patients relapsed. In the intermittent user group, 36% relapsed. In the continuous use group, 59.1% relapsed (P less than .001).
Differences also were found across the three groups in medication adherence rates. Among the nonusers, 47.7% reportedly complied with treatment. The rate was 32% among intermittent users and 20.5% among habitual users (P = .02). Illicit use of other drugs was also higher in the continuous cannabis use group: 27.3% vs. 4% in the cannabis nonuse group and 12% in the intermittent use group (P less than .001).
Also noteworthy was the age of psychosis onset across the groups. While the average age of onset across the study was 28.62 years, for habitual users the average age of illness onset was 25.44 years, compared with 29.52 in nonusers and 28.79 in intermittent users (P = .02).
These results should be viewed in light of their retrospective and self-reported nature – neither of which take into account either premorbid cannabis use or age of illness onset as either predictors or moderators – but the data suggest a dose-response relationship between cannabis use and psychosis, according to the investigators. If so, the implications of the data could reach beyond patient outcomes.
“Because cannabis use is a potentially modifiable risk factor that has an adverse influence on the risk of relapse of psychosis and hospitalization in a given individual, with limited efficacy of existing interventions, these results underscore the importance of developing novel intervention strategies and demand urgent attention from clinicians and health care policy makers,” the authors wrote.
Whether the study conclusively demonstrates that it is cannabis use – and not genetic liability or reverse causation – that drives relapse of psychosis is still not clear. By the investigators’ own admission, the effect of discontinuing cannabis use on patient outcomes has not been tested, leaving the question of a definite causal relationship between continual cannabis use and relapse of psychosis still open for debate.
Ms. Schoeler and the other investigators – with the exception of Dr. Murray – reported no conflicts of interest. Dr. Murray reported that he has received honoraria from Janssen, Sunovion, Otsuka, and Lundbeck. The study was funded in large part by the U.K.’s National Institute for Health Research and by King’s College London.
On Twitter @whitneymcknight
Does habitual cannabis use trigger psychosis, or does a tendency toward psychosis predict increased cannabis use?
“[Our] results suggest that it is more likely than not that continued cannabis use after onset of psychosis is causally associated with increased risk of relapse of psychosis, resulting in psychiatric hospitalization,” reported the authors of a study published online Sept. 28 in JAMA Psychiatry.
It is well established that cannabis use after first-episode psychosis is associated with poor outcomes; what is not so well established is why. The three most prominent theories so far are that cannabis use and psychosis share inherent genetic and/or environmental vulnerabilities, impending psychosis impels increased cannabis use, and conversely, cannabis use has a causal effect on psychosis (Lancet Psychiatry. 2016:3[5]:401) and (Lancet Psychiatry. 2015;3[5]401-2).
After conducting a 10-year, prospective cohort investigation of 220 people who presented to psychiatric services at numerous sites in London between April 2002 and July 2013, Tabea Schoeler and her associates in the current study found that the odds of a relapse of psychosis during periods of cannabis use was increased by 1.13, compared with periods of no cannabis use (95% confidence interval, 1.03-1.24) (JAMA Psychiatry. 2016 Sep 28. doi: 10.1001/jamapsychiatry.2016.2427). Further, those who used cannabis continually after first-episode psychosis were found to have a 59.1% higher risk of relapsing psychosis in that 10-year period, compared with a 28.5% increased risk in those who quit using cannabis entirely (P less than .001).
The only true way to measure causality in this area would be to administer cannabis to participants in a randomized, clinically controlled trial, thus challenging the ethics of clinical study. Instead, Ms. Schoeler of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London and her associates – including Sir Robin M. Murray, MD, a psychiatrist who has been a leading voice in favor of the view that cannabis use and psychotic episodes are often linked – used patient self-reports to assess changes in cannabis use over time. This allowed the investigators to more clearly differentiate between confounders that cannot be readily observed, and that are not likely to change over time, such as any shared genetic or environmental factors, and confounders that can be observed and that do have the potential to change, such as the change in cannabis use status over time, including nonuser vs. user status, and the pattern and frequency of use after onset of illness.
The data were collected in two separate assessments, one was conducted near the time of illness onset, and again at least 2 years after the first episode of psychosis. If interviews were not possible to complete in person, they were conducted over the phone. These data were combined with patient clinical records across the study period. Frequency of cannabis use was measured based on the Cannabis Experience Questionnaire. A patient was considered to have relapsed if he or she was admitted to an inpatient psychiatric facility for exacerbated symptoms of psychosis within 2 years of the first episode of psychosis.
As for the study population, 59.1% of whom were men with an average age of 29 years, there were no significant differences as to whether they had been diagnosed with either affective or nonaffective psychosis. However, there were significant differences between the risk of relapse and the frequency of cannabis use 2 years after onset of illness. In the nonuser group, 28.5% of patients relapsed. In the intermittent user group, 36% relapsed. In the continuous use group, 59.1% relapsed (P less than .001).
Differences also were found across the three groups in medication adherence rates. Among the nonusers, 47.7% reportedly complied with treatment. The rate was 32% among intermittent users and 20.5% among habitual users (P = .02). Illicit use of other drugs was also higher in the continuous cannabis use group: 27.3% vs. 4% in the cannabis nonuse group and 12% in the intermittent use group (P less than .001).
Also noteworthy was the age of psychosis onset across the groups. While the average age of onset across the study was 28.62 years, for habitual users the average age of illness onset was 25.44 years, compared with 29.52 in nonusers and 28.79 in intermittent users (P = .02).
These results should be viewed in light of their retrospective and self-reported nature – neither of which take into account either premorbid cannabis use or age of illness onset as either predictors or moderators – but the data suggest a dose-response relationship between cannabis use and psychosis, according to the investigators. If so, the implications of the data could reach beyond patient outcomes.
“Because cannabis use is a potentially modifiable risk factor that has an adverse influence on the risk of relapse of psychosis and hospitalization in a given individual, with limited efficacy of existing interventions, these results underscore the importance of developing novel intervention strategies and demand urgent attention from clinicians and health care policy makers,” the authors wrote.
Whether the study conclusively demonstrates that it is cannabis use – and not genetic liability or reverse causation – that drives relapse of psychosis is still not clear. By the investigators’ own admission, the effect of discontinuing cannabis use on patient outcomes has not been tested, leaving the question of a definite causal relationship between continual cannabis use and relapse of psychosis still open for debate.
Ms. Schoeler and the other investigators – with the exception of Dr. Murray – reported no conflicts of interest. Dr. Murray reported that he has received honoraria from Janssen, Sunovion, Otsuka, and Lundbeck. The study was funded in large part by the U.K.’s National Institute for Health Research and by King’s College London.
On Twitter @whitneymcknight
FROM JAMA PSYCHIATRY
Key clinical point: Frequent cannabis use in people with psychosis could lead to hospitalization for psychosis relapse.
Major finding: Continuing to use cannabis after first-episode psychosis was associated with 1.13 greater odds of psychosis relapse, compared with psychosis relapse rates during periods of no cannabis use (95% confidence interval, 1.03-1.24).
Data source: A prospective U.K. cohort study of 220 people who presented to psychiatric services with first-episode psychosis between 2002 and 2013.
Disclosures: Ms. Schoeler and the other investigators – with the exception of Dr. Murray – reported no conflicts of interest. Dr. Murray reported that he has received honoraria from Janssen, Sunovion, Otsuka, and Lundbeck. The study was funded in large part by the U.K.’s National Institute for Health Research and by King’s College London.