User login
Lower Transfusion Threshold for Sepsis Equals Fewer Transfusions, No Effect on Mortality
Clinical question
Does a lower transfusion threshold for critically ill patients with septic shock affect outcomes?
Bottom line
Using a lower threshold for transfusion for patients with septic shock in the intensive care unit (ICU) decreases the number of transfusions received without affecting mortality.
Reference
Study design
Randomized controlled trial (nonblinded); (LOE: 1b)
Setting
Inpatient (ICU only)
Synopsis
Using partial blinding and concealed allocation, these investigators randomized ICU patients with septic shock and a hemoglobin level of less than 9 g/dL to receive red blood cell transfusions at either a higher threshold (< 9 g/dL) or a lower threshold (< 7 g/dL). The intervention continued for the entire ICU stay, to a maximum of 90 days. The 2 groups were similar at baseline with an average age of 67 years and a median Sepsis-Related Organ Failure Assessment (SOFA) score of 10 out of 24. Analysis was by intention to treat. Not suprisingly, patients in the higher threshold group received twice as many transfusions as those in the lower threshold group (3088 transfusions vs 1545; P < .001). Notably, one third of the patients in the lower-threshold group required no transfusions at all compared with only 1% in the higher-threshold group (P < .001). For the primary outcome of death at 90 days, there was no significant difference detected between the 2 groups. The per-protocol analysis, which excluded patients with major protocol violations, also showed the same result. Secondary outcomes, including the use of life support and the number of ischemic events in the ICU (eg, acute myocardial or cerebral ischemia), were also similar in the 2 groups.
Dr. Kulkarni is an assistant professor of hospital medicine at Northwestern University in Chicago.
Clinical question
Does a lower transfusion threshold for critically ill patients with septic shock affect outcomes?
Bottom line
Using a lower threshold for transfusion for patients with septic shock in the intensive care unit (ICU) decreases the number of transfusions received without affecting mortality.
Reference
Study design
Randomized controlled trial (nonblinded); (LOE: 1b)
Setting
Inpatient (ICU only)
Synopsis
Using partial blinding and concealed allocation, these investigators randomized ICU patients with septic shock and a hemoglobin level of less than 9 g/dL to receive red blood cell transfusions at either a higher threshold (< 9 g/dL) or a lower threshold (< 7 g/dL). The intervention continued for the entire ICU stay, to a maximum of 90 days. The 2 groups were similar at baseline with an average age of 67 years and a median Sepsis-Related Organ Failure Assessment (SOFA) score of 10 out of 24. Analysis was by intention to treat. Not suprisingly, patients in the higher threshold group received twice as many transfusions as those in the lower threshold group (3088 transfusions vs 1545; P < .001). Notably, one third of the patients in the lower-threshold group required no transfusions at all compared with only 1% in the higher-threshold group (P < .001). For the primary outcome of death at 90 days, there was no significant difference detected between the 2 groups. The per-protocol analysis, which excluded patients with major protocol violations, also showed the same result. Secondary outcomes, including the use of life support and the number of ischemic events in the ICU (eg, acute myocardial or cerebral ischemia), were also similar in the 2 groups.
Dr. Kulkarni is an assistant professor of hospital medicine at Northwestern University in Chicago.
Clinical question
Does a lower transfusion threshold for critically ill patients with septic shock affect outcomes?
Bottom line
Using a lower threshold for transfusion for patients with septic shock in the intensive care unit (ICU) decreases the number of transfusions received without affecting mortality.
Reference
Study design
Randomized controlled trial (nonblinded); (LOE: 1b)
Setting
Inpatient (ICU only)
Synopsis
Using partial blinding and concealed allocation, these investigators randomized ICU patients with septic shock and a hemoglobin level of less than 9 g/dL to receive red blood cell transfusions at either a higher threshold (< 9 g/dL) or a lower threshold (< 7 g/dL). The intervention continued for the entire ICU stay, to a maximum of 90 days. The 2 groups were similar at baseline with an average age of 67 years and a median Sepsis-Related Organ Failure Assessment (SOFA) score of 10 out of 24. Analysis was by intention to treat. Not suprisingly, patients in the higher threshold group received twice as many transfusions as those in the lower threshold group (3088 transfusions vs 1545; P < .001). Notably, one third of the patients in the lower-threshold group required no transfusions at all compared with only 1% in the higher-threshold group (P < .001). For the primary outcome of death at 90 days, there was no significant difference detected between the 2 groups. The per-protocol analysis, which excluded patients with major protocol violations, also showed the same result. Secondary outcomes, including the use of life support and the number of ischemic events in the ICU (eg, acute myocardial or cerebral ischemia), were also similar in the 2 groups.
Dr. Kulkarni is an assistant professor of hospital medicine at Northwestern University in Chicago.
Targeted Therapy for Chronic Lymphocytic Leukemia
This presentation by Adrian Wiestner, MD, PhD, from the 2014 AVAHO Meeting in Portland, Oregon, provides an overview of new insights into the pathogenesis and treatment of CLL, how to interpret molecular targets during treatment, and the advantages and disadvantages of these treatment options for patients.
"The standard of care today is really chemo-immunotherapy," Wiestner said. "Ideally, we would like to have a more disease-directed therapy that is tolerable and active."
This presentation by Adrian Wiestner, MD, PhD, from the 2014 AVAHO Meeting in Portland, Oregon, provides an overview of new insights into the pathogenesis and treatment of CLL, how to interpret molecular targets during treatment, and the advantages and disadvantages of these treatment options for patients.
"The standard of care today is really chemo-immunotherapy," Wiestner said. "Ideally, we would like to have a more disease-directed therapy that is tolerable and active."
This presentation by Adrian Wiestner, MD, PhD, from the 2014 AVAHO Meeting in Portland, Oregon, provides an overview of new insights into the pathogenesis and treatment of CLL, how to interpret molecular targets during treatment, and the advantages and disadvantages of these treatment options for patients.
"The standard of care today is really chemo-immunotherapy," Wiestner said. "Ideally, we would like to have a more disease-directed therapy that is tolerable and active."
The Rural Surgeon: A new column
Surgeons who treat patients in rural areas are a unique group, distinct from other surgeons because of scope of practice, environment, and resources. These surgeons address a vast range of surgical problems, work in relative professional isolation, and have fewer resources available to get the job done. Nevertheless, these physicians play critical roles in their profession and are irreplaceable assets in their communities.
Although poorly defined, often misunderstood, arguably unfairly characterized, and inadequately documented, surgery has a long distinguished history and tradition in rural communities.
What are the main challenges faced by rural surgeons and what can be done to strengthen their practices, professional lives, and longevity in the field? Rural patients are more likely than urban patients to be elderly and poor and have chronic illnesses (J. Am Coll. Surg. 2014;219:814-8). The average age of rural surgeons suggests that many will retire in the coming decade. Who will replace them and care for 60 million citizens who reside in rural America? How will the gradual decline of residents choosing general surgery and rural practice affect patient care?
This new monthly column, Rural Surgeons Speak, focuses on these questions and searches for answers. Rural Surgeons Speak will introduce to all readers the voices, concerns, questions, and opinions of surgeons practicing in the small towns and rural regions of the United States. Although many of the challenges faced by rural surgeons are unique to this group, there is overlap with issues encountered by urban surgeons and those in academic settings. In spite of problems ranging from a shift from independent practice to hospital employment and to treating the poor with pronounced chronic illnesses, rural surgeons are dedicated and committed. Although these matters are common to all surgeons, rural surgeons’ experiences tend to be singular because of the high percentages of such patients and fewer institutional resources.
I have practiced surgery in small and large rural locations for 37 years. Rural surgery is not a specialty. My case log, filled with endoscopies, laparoscopies, laparotomies, and breast operations, may be even more varied than the logs of urban colleagues. In my early years of practice, I did orthopaedic surgery and gynecology. As a conscious choice, I practiced surgery where I wanted, and how I wanted. My choice was not by default. When I started, I sought a location that would allow me to “have a more complete life with less tension, excellent schools, opportunity for economical living,” and a fulfilling surgery practice. Unknown to me, Dr. Edwin F. Cave, ACS president, made those statements about rural surgery in the Daily Clinical Bulletin for the 37th Annual Clinical Congress decades before I started.
Like other rural surgeons, I wanted to embrace and be embraced by my community. I also saw opportunities for personal advancement (involvement with ACS), and for the development of my surgical talents (lasers, laparoscopies, endoscopies). I live just a mile from the hospital. I know my patients by their first names. They speak to me on the telephone. My number is in the book. When they meet me in church or at the grocery store, I am “Doc.” Occasionally a PEx is performed in such locations. It is personal. It is comfortable. There is a real sense of community and of belonging. I am involved. I provide an invaluable service “right at home,” where my patients want to be treated. In return, I am valued and supported.
After becoming a FACS in 1979, I involved myself in many activities of the ACS. Most recently, I participated in the rural surgery renaissance from its beginning. As part of my participation, I routinely contacted rural surgeons around the United States and gained intimate and detailed knowledge about their practices, successes, and concerns. I bring insights from my background to the writing of this column.
While glamour is at a minimum in rural America, personal satisfaction for many of these surgeons is at a maximum. Unfortunately, isolation is a given in most rural practices, and therein lies the problem. In spite of offering much to their patients and, in turn, to the profession, rural surgeons are easy targets of negative assessments, ill-conceived policy changes, and misunderstandings.
Despite their vital role in treating patients, rural surgeons often regard themselves as unrecognized and unappreciated by their peers. There is a subtle bias in the profession against a surgeon who would choose this life of relative isolation, alleged nonspecialized surgery, and overwork in communities with fewer cultural resources and fewer employment options for spouses. Yet their work is essential to the health of millions of people who live in rural areas. Thankfully, the profession as a whole has stopped ignoring rural surgery in recent years as the ACS leadership has stepped in and begun to increase support for rural surgeons.
In May 2011, during the 5th Annual Rural Surgery Symposium and Workshop, I presented a talk, “The ACS and the Rural Surgeon.” Past President Dr. LaMar McGinnis acknowledged the educational value of the presentation, which then went on to wider distribution. The Board of Regents received another talk about rural surgery in February 2012: “Us vs. Them.” Based on the talk and because of their keen insights, the Regents formed the Advisory Council on Rural Surgery – the first new advisory council in 50 years. Subsequently, a College-developed rural listserv became the most highly successful communications program in ACS history and led to the creation of the rural surgery community. In the following 2 years, rural surgeons exchanged 9 million emails on numerous, varied topics. An article in the July 2014 ACS Bulletin describes the program (http://bulletin.facs.org/2014/07/acs-rural-listserv-an-underdog-success-story/). Finally, in his inaugural address at the 2012 Clinical Congress, Dr. Brent Eastman identified rural surgery as one of the four fundamental components of surgery for the next 100 years (http://bulletin.facs.org/2012/12/presidential-address/). Recognition of rural surgery obviously has increased.
My experience of moderating the rural listserv with nearly 1,000 subscribers has revealed the amazing diversity, passion, commitment, and perseverance of rural surgeons who have united in a true community. Their perspectives on their professional lives and the lives of their patients are well worth closer acquaintance by surgeons from all sectors. They have a lot to teach us all. To learn about rural surgery will be to learn about all surgery. Understand rural surgery in 2014 and, consequently, understand all surgery. Recognize the benefits of rural surgery and then instill them in all practices. Supporting rural surgery today supports all surgery.
The column aims to offer commentary on diverse topics confronting rural surgeons today. Opinions, editorials, letters, economics, and clinical matters, all from the rural perspective, will be included. Over 60 individual “threads” appeared on the listserv this year and the subjects of many of these threads could be covered in future columns. Uncommon subjects such as YKYAARSI (“You know you are a rural surgeon if…”) should be entertaining and informative. Guest authors who are experts on varying subjects will contribute articles.
While its roots are in the rural surgery community, the column’s scope will touch all surgical practices. Rural surgery will be better defined, positively characterized, and documented. In partnership with the ACS to achieve the general goal of supporting rural surgery, Rural Surgeons Speak aims to enable all ACS Fellows to realize that they belong to a community of all surgeons, regardless of location or practice type.
Dr. Caropreso is a general surgeon at Keokuk (Iowa) Area Hospital and Clinical Professor of Surgery at the University of Iowa Carver College of Medicine. He has practiced surgery in the rural communities of Mason City, Iowa; Keokuk, Iowa; and Carthage, Ill., for 37 years.
Surgeons who treat patients in rural areas are a unique group, distinct from other surgeons because of scope of practice, environment, and resources. These surgeons address a vast range of surgical problems, work in relative professional isolation, and have fewer resources available to get the job done. Nevertheless, these physicians play critical roles in their profession and are irreplaceable assets in their communities.
Although poorly defined, often misunderstood, arguably unfairly characterized, and inadequately documented, surgery has a long distinguished history and tradition in rural communities.
What are the main challenges faced by rural surgeons and what can be done to strengthen their practices, professional lives, and longevity in the field? Rural patients are more likely than urban patients to be elderly and poor and have chronic illnesses (J. Am Coll. Surg. 2014;219:814-8). The average age of rural surgeons suggests that many will retire in the coming decade. Who will replace them and care for 60 million citizens who reside in rural America? How will the gradual decline of residents choosing general surgery and rural practice affect patient care?
This new monthly column, Rural Surgeons Speak, focuses on these questions and searches for answers. Rural Surgeons Speak will introduce to all readers the voices, concerns, questions, and opinions of surgeons practicing in the small towns and rural regions of the United States. Although many of the challenges faced by rural surgeons are unique to this group, there is overlap with issues encountered by urban surgeons and those in academic settings. In spite of problems ranging from a shift from independent practice to hospital employment and to treating the poor with pronounced chronic illnesses, rural surgeons are dedicated and committed. Although these matters are common to all surgeons, rural surgeons’ experiences tend to be singular because of the high percentages of such patients and fewer institutional resources.
I have practiced surgery in small and large rural locations for 37 years. Rural surgery is not a specialty. My case log, filled with endoscopies, laparoscopies, laparotomies, and breast operations, may be even more varied than the logs of urban colleagues. In my early years of practice, I did orthopaedic surgery and gynecology. As a conscious choice, I practiced surgery where I wanted, and how I wanted. My choice was not by default. When I started, I sought a location that would allow me to “have a more complete life with less tension, excellent schools, opportunity for economical living,” and a fulfilling surgery practice. Unknown to me, Dr. Edwin F. Cave, ACS president, made those statements about rural surgery in the Daily Clinical Bulletin for the 37th Annual Clinical Congress decades before I started.
Like other rural surgeons, I wanted to embrace and be embraced by my community. I also saw opportunities for personal advancement (involvement with ACS), and for the development of my surgical talents (lasers, laparoscopies, endoscopies). I live just a mile from the hospital. I know my patients by their first names. They speak to me on the telephone. My number is in the book. When they meet me in church or at the grocery store, I am “Doc.” Occasionally a PEx is performed in such locations. It is personal. It is comfortable. There is a real sense of community and of belonging. I am involved. I provide an invaluable service “right at home,” where my patients want to be treated. In return, I am valued and supported.
After becoming a FACS in 1979, I involved myself in many activities of the ACS. Most recently, I participated in the rural surgery renaissance from its beginning. As part of my participation, I routinely contacted rural surgeons around the United States and gained intimate and detailed knowledge about their practices, successes, and concerns. I bring insights from my background to the writing of this column.
While glamour is at a minimum in rural America, personal satisfaction for many of these surgeons is at a maximum. Unfortunately, isolation is a given in most rural practices, and therein lies the problem. In spite of offering much to their patients and, in turn, to the profession, rural surgeons are easy targets of negative assessments, ill-conceived policy changes, and misunderstandings.
Despite their vital role in treating patients, rural surgeons often regard themselves as unrecognized and unappreciated by their peers. There is a subtle bias in the profession against a surgeon who would choose this life of relative isolation, alleged nonspecialized surgery, and overwork in communities with fewer cultural resources and fewer employment options for spouses. Yet their work is essential to the health of millions of people who live in rural areas. Thankfully, the profession as a whole has stopped ignoring rural surgery in recent years as the ACS leadership has stepped in and begun to increase support for rural surgeons.
In May 2011, during the 5th Annual Rural Surgery Symposium and Workshop, I presented a talk, “The ACS and the Rural Surgeon.” Past President Dr. LaMar McGinnis acknowledged the educational value of the presentation, which then went on to wider distribution. The Board of Regents received another talk about rural surgery in February 2012: “Us vs. Them.” Based on the talk and because of their keen insights, the Regents formed the Advisory Council on Rural Surgery – the first new advisory council in 50 years. Subsequently, a College-developed rural listserv became the most highly successful communications program in ACS history and led to the creation of the rural surgery community. In the following 2 years, rural surgeons exchanged 9 million emails on numerous, varied topics. An article in the July 2014 ACS Bulletin describes the program (http://bulletin.facs.org/2014/07/acs-rural-listserv-an-underdog-success-story/). Finally, in his inaugural address at the 2012 Clinical Congress, Dr. Brent Eastman identified rural surgery as one of the four fundamental components of surgery for the next 100 years (http://bulletin.facs.org/2012/12/presidential-address/). Recognition of rural surgery obviously has increased.
My experience of moderating the rural listserv with nearly 1,000 subscribers has revealed the amazing diversity, passion, commitment, and perseverance of rural surgeons who have united in a true community. Their perspectives on their professional lives and the lives of their patients are well worth closer acquaintance by surgeons from all sectors. They have a lot to teach us all. To learn about rural surgery will be to learn about all surgery. Understand rural surgery in 2014 and, consequently, understand all surgery. Recognize the benefits of rural surgery and then instill them in all practices. Supporting rural surgery today supports all surgery.
The column aims to offer commentary on diverse topics confronting rural surgeons today. Opinions, editorials, letters, economics, and clinical matters, all from the rural perspective, will be included. Over 60 individual “threads” appeared on the listserv this year and the subjects of many of these threads could be covered in future columns. Uncommon subjects such as YKYAARSI (“You know you are a rural surgeon if…”) should be entertaining and informative. Guest authors who are experts on varying subjects will contribute articles.
While its roots are in the rural surgery community, the column’s scope will touch all surgical practices. Rural surgery will be better defined, positively characterized, and documented. In partnership with the ACS to achieve the general goal of supporting rural surgery, Rural Surgeons Speak aims to enable all ACS Fellows to realize that they belong to a community of all surgeons, regardless of location or practice type.
Dr. Caropreso is a general surgeon at Keokuk (Iowa) Area Hospital and Clinical Professor of Surgery at the University of Iowa Carver College of Medicine. He has practiced surgery in the rural communities of Mason City, Iowa; Keokuk, Iowa; and Carthage, Ill., for 37 years.
Surgeons who treat patients in rural areas are a unique group, distinct from other surgeons because of scope of practice, environment, and resources. These surgeons address a vast range of surgical problems, work in relative professional isolation, and have fewer resources available to get the job done. Nevertheless, these physicians play critical roles in their profession and are irreplaceable assets in their communities.
Although poorly defined, often misunderstood, arguably unfairly characterized, and inadequately documented, surgery has a long distinguished history and tradition in rural communities.
What are the main challenges faced by rural surgeons and what can be done to strengthen their practices, professional lives, and longevity in the field? Rural patients are more likely than urban patients to be elderly and poor and have chronic illnesses (J. Am Coll. Surg. 2014;219:814-8). The average age of rural surgeons suggests that many will retire in the coming decade. Who will replace them and care for 60 million citizens who reside in rural America? How will the gradual decline of residents choosing general surgery and rural practice affect patient care?
This new monthly column, Rural Surgeons Speak, focuses on these questions and searches for answers. Rural Surgeons Speak will introduce to all readers the voices, concerns, questions, and opinions of surgeons practicing in the small towns and rural regions of the United States. Although many of the challenges faced by rural surgeons are unique to this group, there is overlap with issues encountered by urban surgeons and those in academic settings. In spite of problems ranging from a shift from independent practice to hospital employment and to treating the poor with pronounced chronic illnesses, rural surgeons are dedicated and committed. Although these matters are common to all surgeons, rural surgeons’ experiences tend to be singular because of the high percentages of such patients and fewer institutional resources.
I have practiced surgery in small and large rural locations for 37 years. Rural surgery is not a specialty. My case log, filled with endoscopies, laparoscopies, laparotomies, and breast operations, may be even more varied than the logs of urban colleagues. In my early years of practice, I did orthopaedic surgery and gynecology. As a conscious choice, I practiced surgery where I wanted, and how I wanted. My choice was not by default. When I started, I sought a location that would allow me to “have a more complete life with less tension, excellent schools, opportunity for economical living,” and a fulfilling surgery practice. Unknown to me, Dr. Edwin F. Cave, ACS president, made those statements about rural surgery in the Daily Clinical Bulletin for the 37th Annual Clinical Congress decades before I started.
Like other rural surgeons, I wanted to embrace and be embraced by my community. I also saw opportunities for personal advancement (involvement with ACS), and for the development of my surgical talents (lasers, laparoscopies, endoscopies). I live just a mile from the hospital. I know my patients by their first names. They speak to me on the telephone. My number is in the book. When they meet me in church or at the grocery store, I am “Doc.” Occasionally a PEx is performed in such locations. It is personal. It is comfortable. There is a real sense of community and of belonging. I am involved. I provide an invaluable service “right at home,” where my patients want to be treated. In return, I am valued and supported.
After becoming a FACS in 1979, I involved myself in many activities of the ACS. Most recently, I participated in the rural surgery renaissance from its beginning. As part of my participation, I routinely contacted rural surgeons around the United States and gained intimate and detailed knowledge about their practices, successes, and concerns. I bring insights from my background to the writing of this column.
While glamour is at a minimum in rural America, personal satisfaction for many of these surgeons is at a maximum. Unfortunately, isolation is a given in most rural practices, and therein lies the problem. In spite of offering much to their patients and, in turn, to the profession, rural surgeons are easy targets of negative assessments, ill-conceived policy changes, and misunderstandings.
Despite their vital role in treating patients, rural surgeons often regard themselves as unrecognized and unappreciated by their peers. There is a subtle bias in the profession against a surgeon who would choose this life of relative isolation, alleged nonspecialized surgery, and overwork in communities with fewer cultural resources and fewer employment options for spouses. Yet their work is essential to the health of millions of people who live in rural areas. Thankfully, the profession as a whole has stopped ignoring rural surgery in recent years as the ACS leadership has stepped in and begun to increase support for rural surgeons.
In May 2011, during the 5th Annual Rural Surgery Symposium and Workshop, I presented a talk, “The ACS and the Rural Surgeon.” Past President Dr. LaMar McGinnis acknowledged the educational value of the presentation, which then went on to wider distribution. The Board of Regents received another talk about rural surgery in February 2012: “Us vs. Them.” Based on the talk and because of their keen insights, the Regents formed the Advisory Council on Rural Surgery – the first new advisory council in 50 years. Subsequently, a College-developed rural listserv became the most highly successful communications program in ACS history and led to the creation of the rural surgery community. In the following 2 years, rural surgeons exchanged 9 million emails on numerous, varied topics. An article in the July 2014 ACS Bulletin describes the program (http://bulletin.facs.org/2014/07/acs-rural-listserv-an-underdog-success-story/). Finally, in his inaugural address at the 2012 Clinical Congress, Dr. Brent Eastman identified rural surgery as one of the four fundamental components of surgery for the next 100 years (http://bulletin.facs.org/2012/12/presidential-address/). Recognition of rural surgery obviously has increased.
My experience of moderating the rural listserv with nearly 1,000 subscribers has revealed the amazing diversity, passion, commitment, and perseverance of rural surgeons who have united in a true community. Their perspectives on their professional lives and the lives of their patients are well worth closer acquaintance by surgeons from all sectors. They have a lot to teach us all. To learn about rural surgery will be to learn about all surgery. Understand rural surgery in 2014 and, consequently, understand all surgery. Recognize the benefits of rural surgery and then instill them in all practices. Supporting rural surgery today supports all surgery.
The column aims to offer commentary on diverse topics confronting rural surgeons today. Opinions, editorials, letters, economics, and clinical matters, all from the rural perspective, will be included. Over 60 individual “threads” appeared on the listserv this year and the subjects of many of these threads could be covered in future columns. Uncommon subjects such as YKYAARSI (“You know you are a rural surgeon if…”) should be entertaining and informative. Guest authors who are experts on varying subjects will contribute articles.
While its roots are in the rural surgery community, the column’s scope will touch all surgical practices. Rural surgery will be better defined, positively characterized, and documented. In partnership with the ACS to achieve the general goal of supporting rural surgery, Rural Surgeons Speak aims to enable all ACS Fellows to realize that they belong to a community of all surgeons, regardless of location or practice type.
Dr. Caropreso is a general surgeon at Keokuk (Iowa) Area Hospital and Clinical Professor of Surgery at the University of Iowa Carver College of Medicine. He has practiced surgery in the rural communities of Mason City, Iowa; Keokuk, Iowa; and Carthage, Ill., for 37 years.
More isn’t always better with daunorubicin induction in AML

Photo courtesy of ASH
SAN FRANCISCO—Results regarding daunorubicin escalation in induction for patients with acute myeloid leukemia (AML) have varied among different studies.
And a 90 mg/m2 dose has been shown to be more effective than 45. Now, results of the UK NCRI AML17 trial have added yet another dimension to the discussion—the use of 60 mg/m2 compared to 90.
Alan K. Burnett, MD, of Cardiff University in the UK, presented the data as abstract 7 at the 2014 ASH Annual Meeting.
He explained that ECOG E1900 had demonstrated superior remission and overall survival for a 90-mg dose of daunorubicin compared to a 45-mg dose in adults younger than 60.
The HOVON trial showed superior remission but no difference in overall survival for the higher dose of daunorubicin in patients older than 60.
The Korean trial demonstrated superior remission and survival rates for the 90-mg dose in patients younger than 60.
And in the GOELAMS trial, investigators found no difference between a 90-mg and a 60-mg dose level.
So the UK AML Study Group undertook to clarify the issue by comparing 90 mg to 60 mg in induction.
The investigators randomized 1206 patients younger than 60 with de novo or secondary AML or high-risk myelodysplastic syndromes (MDS) to receive 90 or 60 mg of daunorubicin on days 1, 3, and 5 of their first induction course, followed by 50 mg/m2 on days 1, 3, and 5 in course 2.
All patients received ara-C during courses 1 and 2. Patients had to have LVEF of 45% or greater to be included in the trial.
The median follow-up was 29 months. Patient characteristics were comparable between the 2 groups.
The median age was 53 in both groups (range, 16-72 years), and slightly more than half of patients were male. Eighty-five percent and 84% had de novo AML in the 60-mg and 90-mg arms, respectively. Ten percent in each group had secondary AML. And 5% and 6%, respectively, had high-risk MDS.
Eleven percent in the 60-mg arm had favorable cytogenetics, compared with 9% in the 90-mg arm. Eighteen percent in each arm had mutant FLT3-ITD, and 40% in each arm were poor risk.
The investigators found no significant difference in response between the two arms—84% in the 60-mg arm and 81% in the 90-mg arm.
However, they observed a trend for increased 30-day mortality in the 90-mg arm and a significant difference in the 60-day mortality rate, which was 5% in the 60-mg arm and 10% in the 90-mg arm (P=0.001).
Twenty-nine people died by day 60 in the 60-mg arm compared with 58 in the 90-mg arm.
The main reasons for 60-day mortality in the 60-mg and 90-mg dose groups, respectively, included infection (11 vs 25 deaths), hemorrhage (3 vs 5 deaths), infection plus hemorrhage (3 vs 1 death), and resistant disease (2 vs 14 deaths), among other causes.
At 24 months, overall survival between the 2 groups was comparable, at 60% in the 60-mg arm and 59% in the 90-mg arm.
The cumulative incidence of relapse at 24 months from complete response was 41% in the 60-mg arm and 37% in the 90-mg arm.
One hundred ninety-seven patients in the 60-mg arm and 169 patients in the 90-mg arm went on to receive a stem cell transplant.
When survival was censored at transplant, there was also no difference between the arms, at 60% for the 90-mg group and 61% for the 60-mg group.
The investigators conducted subgroup analyses and found no significant benefit for 90 mg/m2 in any subgroup. Dr Burnett noted, however, that there could be a potential late benefit for FLT3-ITD mutated patients who receive a 90-mg dose.
FLT3 mutated patients had a non-significant survival benefit (HR 0.74 [0.47-1.17] P=0.2) with a 90-mg dose. However, survival was significantly worse for FLT3 wild type patients receiving 90 mg (HR 1.31 [1.03-1.67] P=0.03).
Otherwise, the group found that there is no evidence to suggest that 90 mg is superior to 60 mg. ![]()

Photo courtesy of ASH
SAN FRANCISCO—Results regarding daunorubicin escalation in induction for patients with acute myeloid leukemia (AML) have varied among different studies.
And a 90 mg/m2 dose has been shown to be more effective than 45. Now, results of the UK NCRI AML17 trial have added yet another dimension to the discussion—the use of 60 mg/m2 compared to 90.
Alan K. Burnett, MD, of Cardiff University in the UK, presented the data as abstract 7 at the 2014 ASH Annual Meeting.
He explained that ECOG E1900 had demonstrated superior remission and overall survival for a 90-mg dose of daunorubicin compared to a 45-mg dose in adults younger than 60.
The HOVON trial showed superior remission but no difference in overall survival for the higher dose of daunorubicin in patients older than 60.
The Korean trial demonstrated superior remission and survival rates for the 90-mg dose in patients younger than 60.
And in the GOELAMS trial, investigators found no difference between a 90-mg and a 60-mg dose level.
So the UK AML Study Group undertook to clarify the issue by comparing 90 mg to 60 mg in induction.
The investigators randomized 1206 patients younger than 60 with de novo or secondary AML or high-risk myelodysplastic syndromes (MDS) to receive 90 or 60 mg of daunorubicin on days 1, 3, and 5 of their first induction course, followed by 50 mg/m2 on days 1, 3, and 5 in course 2.
All patients received ara-C during courses 1 and 2. Patients had to have LVEF of 45% or greater to be included in the trial.
The median follow-up was 29 months. Patient characteristics were comparable between the 2 groups.
The median age was 53 in both groups (range, 16-72 years), and slightly more than half of patients were male. Eighty-five percent and 84% had de novo AML in the 60-mg and 90-mg arms, respectively. Ten percent in each group had secondary AML. And 5% and 6%, respectively, had high-risk MDS.
Eleven percent in the 60-mg arm had favorable cytogenetics, compared with 9% in the 90-mg arm. Eighteen percent in each arm had mutant FLT3-ITD, and 40% in each arm were poor risk.
The investigators found no significant difference in response between the two arms—84% in the 60-mg arm and 81% in the 90-mg arm.
However, they observed a trend for increased 30-day mortality in the 90-mg arm and a significant difference in the 60-day mortality rate, which was 5% in the 60-mg arm and 10% in the 90-mg arm (P=0.001).
Twenty-nine people died by day 60 in the 60-mg arm compared with 58 in the 90-mg arm.
The main reasons for 60-day mortality in the 60-mg and 90-mg dose groups, respectively, included infection (11 vs 25 deaths), hemorrhage (3 vs 5 deaths), infection plus hemorrhage (3 vs 1 death), and resistant disease (2 vs 14 deaths), among other causes.
At 24 months, overall survival between the 2 groups was comparable, at 60% in the 60-mg arm and 59% in the 90-mg arm.
The cumulative incidence of relapse at 24 months from complete response was 41% in the 60-mg arm and 37% in the 90-mg arm.
One hundred ninety-seven patients in the 60-mg arm and 169 patients in the 90-mg arm went on to receive a stem cell transplant.
When survival was censored at transplant, there was also no difference between the arms, at 60% for the 90-mg group and 61% for the 60-mg group.
The investigators conducted subgroup analyses and found no significant benefit for 90 mg/m2 in any subgroup. Dr Burnett noted, however, that there could be a potential late benefit for FLT3-ITD mutated patients who receive a 90-mg dose.
FLT3 mutated patients had a non-significant survival benefit (HR 0.74 [0.47-1.17] P=0.2) with a 90-mg dose. However, survival was significantly worse for FLT3 wild type patients receiving 90 mg (HR 1.31 [1.03-1.67] P=0.03).
Otherwise, the group found that there is no evidence to suggest that 90 mg is superior to 60 mg. ![]()

Photo courtesy of ASH
SAN FRANCISCO—Results regarding daunorubicin escalation in induction for patients with acute myeloid leukemia (AML) have varied among different studies.
And a 90 mg/m2 dose has been shown to be more effective than 45. Now, results of the UK NCRI AML17 trial have added yet another dimension to the discussion—the use of 60 mg/m2 compared to 90.
Alan K. Burnett, MD, of Cardiff University in the UK, presented the data as abstract 7 at the 2014 ASH Annual Meeting.
He explained that ECOG E1900 had demonstrated superior remission and overall survival for a 90-mg dose of daunorubicin compared to a 45-mg dose in adults younger than 60.
The HOVON trial showed superior remission but no difference in overall survival for the higher dose of daunorubicin in patients older than 60.
The Korean trial demonstrated superior remission and survival rates for the 90-mg dose in patients younger than 60.
And in the GOELAMS trial, investigators found no difference between a 90-mg and a 60-mg dose level.
So the UK AML Study Group undertook to clarify the issue by comparing 90 mg to 60 mg in induction.
The investigators randomized 1206 patients younger than 60 with de novo or secondary AML or high-risk myelodysplastic syndromes (MDS) to receive 90 or 60 mg of daunorubicin on days 1, 3, and 5 of their first induction course, followed by 50 mg/m2 on days 1, 3, and 5 in course 2.
All patients received ara-C during courses 1 and 2. Patients had to have LVEF of 45% or greater to be included in the trial.
The median follow-up was 29 months. Patient characteristics were comparable between the 2 groups.
The median age was 53 in both groups (range, 16-72 years), and slightly more than half of patients were male. Eighty-five percent and 84% had de novo AML in the 60-mg and 90-mg arms, respectively. Ten percent in each group had secondary AML. And 5% and 6%, respectively, had high-risk MDS.
Eleven percent in the 60-mg arm had favorable cytogenetics, compared with 9% in the 90-mg arm. Eighteen percent in each arm had mutant FLT3-ITD, and 40% in each arm were poor risk.
The investigators found no significant difference in response between the two arms—84% in the 60-mg arm and 81% in the 90-mg arm.
However, they observed a trend for increased 30-day mortality in the 90-mg arm and a significant difference in the 60-day mortality rate, which was 5% in the 60-mg arm and 10% in the 90-mg arm (P=0.001).
Twenty-nine people died by day 60 in the 60-mg arm compared with 58 in the 90-mg arm.
The main reasons for 60-day mortality in the 60-mg and 90-mg dose groups, respectively, included infection (11 vs 25 deaths), hemorrhage (3 vs 5 deaths), infection plus hemorrhage (3 vs 1 death), and resistant disease (2 vs 14 deaths), among other causes.
At 24 months, overall survival between the 2 groups was comparable, at 60% in the 60-mg arm and 59% in the 90-mg arm.
The cumulative incidence of relapse at 24 months from complete response was 41% in the 60-mg arm and 37% in the 90-mg arm.
One hundred ninety-seven patients in the 60-mg arm and 169 patients in the 90-mg arm went on to receive a stem cell transplant.
When survival was censored at transplant, there was also no difference between the arms, at 60% for the 90-mg group and 61% for the 60-mg group.
The investigators conducted subgroup analyses and found no significant benefit for 90 mg/m2 in any subgroup. Dr Burnett noted, however, that there could be a potential late benefit for FLT3-ITD mutated patients who receive a 90-mg dose.
FLT3 mutated patients had a non-significant survival benefit (HR 0.74 [0.47-1.17] P=0.2) with a 90-mg dose. However, survival was significantly worse for FLT3 wild type patients receiving 90 mg (HR 1.31 [1.03-1.67] P=0.03).
Otherwise, the group found that there is no evidence to suggest that 90 mg is superior to 60 mg. ![]()
Encapsulating doxorubicin can reduce heart damage

Credit: USDA
VIENNA—Encapsulating the anthracycline doxorubicin in a liposome can reduce the risk of developing heart damage, according to a study presented at EuroEcho-Imaging 2014.
Researchers administered doxorubicin encased in a liposome to a small group of pigs and compared cardiac outcomes to those in pigs that received unmanipulated doxorubicin or epirubicin.
Pigs that received encapsulated doxorubicin still developed cardiotoxicity, but at lower rates than pigs that received traditional doxorubicin.
Pigs that received epirubicin were excluded due to low survival rates.
“[M]any chemotherapies—in particular, anthracyclines—cause cardiac side effects that can lead to cardiomyopathy and severe heart failure,” said study investigator Jutta Bergler-Klein, MD, of the Medical University of Vienna in Austria. “Cardiotoxicity can occur acutely or up to 30 years after chemotherapy and is the second most common cause of death in cancer patients, after secondary malignancy in childhood cancer survivors.”
“Liposomal encapsulation is a new technique which wraps the chemotherapy drug in a fatty cover called a liposome. More of the drug reaches the cancer cells because there is less degradation, and there are fewer side effects on healthy cells because the fat cover acts as a barrier.”
“The drug stays in the bloodstream longer, allowing higher cumulative doses to be given. We tested whether non-pegylated liposome encapsulation of the anthracycline doxorubicin (called Myocet) could decrease its cardiotoxicity compared to conventional doxorubicin or epirubicin, another anthracycline.”
The study included 24 pigs that were randomized to receive the human dose-equivalent of Myocet, conventional doxorubicin, or epirubicin in 3 cycles. The epirubicin group was excluded from the final analyses because of low survival levels.
The researchers assessed cardiac function by echocardiography and MRI at baseline and follow-up (after about 3 months). Laboratory follow-up included hematology, renal function, and measurement of the cardiac enzymes troponin and BNP.
“The dose, imaging methodology, and blood parameters simulate the monitoring that patients on this treatment would receive and produces valuable translational data,” Dr Bergler-Klein said.
The researchers found that the group receiving Myocet had better diastolic and systolic function in the left and right ventricles, compared to conventional doxorubicin. The Myocet group also had less fibrosis in the myocardium, as shown by MRI and histology staining.
“Our study shows that doxorubicin encapsulated in a liposome had fewer cardiac side effects than doxorubicin given in the conventional way,” Dr Bergler-Klein said.
“We did find cardiac toxicity in the Myocet group as well, despite the fact that the pigs were young, healthy, and received anthracyclines for only a short period. This emphasizes how important it is for all cancer patients taking anthracyclines to receive cardiac monitoring using echocardiography and biomarkers, and MRI where indicated.”
“Many patients who recover after chemotherapy have asymptomatic heart damage, which can become symptomatic as they get older. When heart problems are picked up early, patients can be given preventive treatment, including ACE inhibitors, angiotensin receptor blockers, or beta-blockers, to prevent the progression to overt heart failure.”
The researchers are now conducting gene-expression profiling on the histology samples, hoping to explain the better outcome and cardiac function after Myocet therapy. They have found differences in the expression of genes that control energy use and the metabolic state, with better regulation in the Myocet group. ![]()

Credit: USDA
VIENNA—Encapsulating the anthracycline doxorubicin in a liposome can reduce the risk of developing heart damage, according to a study presented at EuroEcho-Imaging 2014.
Researchers administered doxorubicin encased in a liposome to a small group of pigs and compared cardiac outcomes to those in pigs that received unmanipulated doxorubicin or epirubicin.
Pigs that received encapsulated doxorubicin still developed cardiotoxicity, but at lower rates than pigs that received traditional doxorubicin.
Pigs that received epirubicin were excluded due to low survival rates.
“[M]any chemotherapies—in particular, anthracyclines—cause cardiac side effects that can lead to cardiomyopathy and severe heart failure,” said study investigator Jutta Bergler-Klein, MD, of the Medical University of Vienna in Austria. “Cardiotoxicity can occur acutely or up to 30 years after chemotherapy and is the second most common cause of death in cancer patients, after secondary malignancy in childhood cancer survivors.”
“Liposomal encapsulation is a new technique which wraps the chemotherapy drug in a fatty cover called a liposome. More of the drug reaches the cancer cells because there is less degradation, and there are fewer side effects on healthy cells because the fat cover acts as a barrier.”
“The drug stays in the bloodstream longer, allowing higher cumulative doses to be given. We tested whether non-pegylated liposome encapsulation of the anthracycline doxorubicin (called Myocet) could decrease its cardiotoxicity compared to conventional doxorubicin or epirubicin, another anthracycline.”
The study included 24 pigs that were randomized to receive the human dose-equivalent of Myocet, conventional doxorubicin, or epirubicin in 3 cycles. The epirubicin group was excluded from the final analyses because of low survival levels.
The researchers assessed cardiac function by echocardiography and MRI at baseline and follow-up (after about 3 months). Laboratory follow-up included hematology, renal function, and measurement of the cardiac enzymes troponin and BNP.
“The dose, imaging methodology, and blood parameters simulate the monitoring that patients on this treatment would receive and produces valuable translational data,” Dr Bergler-Klein said.
The researchers found that the group receiving Myocet had better diastolic and systolic function in the left and right ventricles, compared to conventional doxorubicin. The Myocet group also had less fibrosis in the myocardium, as shown by MRI and histology staining.
“Our study shows that doxorubicin encapsulated in a liposome had fewer cardiac side effects than doxorubicin given in the conventional way,” Dr Bergler-Klein said.
“We did find cardiac toxicity in the Myocet group as well, despite the fact that the pigs were young, healthy, and received anthracyclines for only a short period. This emphasizes how important it is for all cancer patients taking anthracyclines to receive cardiac monitoring using echocardiography and biomarkers, and MRI where indicated.”
“Many patients who recover after chemotherapy have asymptomatic heart damage, which can become symptomatic as they get older. When heart problems are picked up early, patients can be given preventive treatment, including ACE inhibitors, angiotensin receptor blockers, or beta-blockers, to prevent the progression to overt heart failure.”
The researchers are now conducting gene-expression profiling on the histology samples, hoping to explain the better outcome and cardiac function after Myocet therapy. They have found differences in the expression of genes that control energy use and the metabolic state, with better regulation in the Myocet group. ![]()

Credit: USDA
VIENNA—Encapsulating the anthracycline doxorubicin in a liposome can reduce the risk of developing heart damage, according to a study presented at EuroEcho-Imaging 2014.
Researchers administered doxorubicin encased in a liposome to a small group of pigs and compared cardiac outcomes to those in pigs that received unmanipulated doxorubicin or epirubicin.
Pigs that received encapsulated doxorubicin still developed cardiotoxicity, but at lower rates than pigs that received traditional doxorubicin.
Pigs that received epirubicin were excluded due to low survival rates.
“[M]any chemotherapies—in particular, anthracyclines—cause cardiac side effects that can lead to cardiomyopathy and severe heart failure,” said study investigator Jutta Bergler-Klein, MD, of the Medical University of Vienna in Austria. “Cardiotoxicity can occur acutely or up to 30 years after chemotherapy and is the second most common cause of death in cancer patients, after secondary malignancy in childhood cancer survivors.”
“Liposomal encapsulation is a new technique which wraps the chemotherapy drug in a fatty cover called a liposome. More of the drug reaches the cancer cells because there is less degradation, and there are fewer side effects on healthy cells because the fat cover acts as a barrier.”
“The drug stays in the bloodstream longer, allowing higher cumulative doses to be given. We tested whether non-pegylated liposome encapsulation of the anthracycline doxorubicin (called Myocet) could decrease its cardiotoxicity compared to conventional doxorubicin or epirubicin, another anthracycline.”
The study included 24 pigs that were randomized to receive the human dose-equivalent of Myocet, conventional doxorubicin, or epirubicin in 3 cycles. The epirubicin group was excluded from the final analyses because of low survival levels.
The researchers assessed cardiac function by echocardiography and MRI at baseline and follow-up (after about 3 months). Laboratory follow-up included hematology, renal function, and measurement of the cardiac enzymes troponin and BNP.
“The dose, imaging methodology, and blood parameters simulate the monitoring that patients on this treatment would receive and produces valuable translational data,” Dr Bergler-Klein said.
The researchers found that the group receiving Myocet had better diastolic and systolic function in the left and right ventricles, compared to conventional doxorubicin. The Myocet group also had less fibrosis in the myocardium, as shown by MRI and histology staining.
“Our study shows that doxorubicin encapsulated in a liposome had fewer cardiac side effects than doxorubicin given in the conventional way,” Dr Bergler-Klein said.
“We did find cardiac toxicity in the Myocet group as well, despite the fact that the pigs were young, healthy, and received anthracyclines for only a short period. This emphasizes how important it is for all cancer patients taking anthracyclines to receive cardiac monitoring using echocardiography and biomarkers, and MRI where indicated.”
“Many patients who recover after chemotherapy have asymptomatic heart damage, which can become symptomatic as they get older. When heart problems are picked up early, patients can be given preventive treatment, including ACE inhibitors, angiotensin receptor blockers, or beta-blockers, to prevent the progression to overt heart failure.”
The researchers are now conducting gene-expression profiling on the histology samples, hoping to explain the better outcome and cardiac function after Myocet therapy. They have found differences in the expression of genes that control energy use and the metabolic state, with better regulation in the Myocet group. ![]()
The Best Times to Try Abiraterone
Until recently, there have been few treatment options for advanced prostate cancer that is resistant to androgen-directed therapies. Newer treatments that target residual androgen production offer some hope of prolonging the interval before chemotherapy, with fewer adverse effects (AEs) and better efficacy. One of those is abiraterone, which blocks extragonadal, testicular, and tumor androgen biosynthesis.
An ongoing multinational phase 3 study is evaluating the clinical benefits of abiraterone plus prednisone vs prednisone alone in patients with progressive metastatic castration-resistant prostate cancer (mCRPC). Follow-up for the study has now exceeded 27 months, giving a good opportunity to evaluate safety and efficacy. Thus, after having reviewed outcomes so far, the independent data-monitoring committee recommended that the study be unblinded and patients be allowed to cross over from prednisone to abiraterone. The researchers reported the results of the third interim analysis, with updated analysis.
Patients were stratified by Eastern Cooperative Oncology Group performance status (ECOG-PS) and randomly assigned to receive abiraterone 1,000 mg plus prednisone 5 mg twice daily or placebo plus prednisone.
Patients who received abiraterone had, compared with those on prednisone, statistically significant improvement in radiographic progression-free survival (PFS), with a median time to disease progression or death of 16.5 months, vs 8.2 months (95% CI, 0.45-0.61).
Overall survival also lengthened, from a median of 35.3 months vs 30.1 months (95% CI, 0.66-0.95).
All secondary endpoints also favored abiraterone over prednisone. For instance, abiraterone treatment delayed the time to the need for opiates for cancer-related pain and the time to initiation of chemotherapy. Abiraterone also delayed the time to deterioration in ECOG-PS and prostate-specific antigen (PSA) progression. Abiraterone more than doubled the PSA response rate: 68% vs 29% with prednisone.
Patients reported more pain relief. Those receiving abiraterone had statistically significant improvement in pain interference (P = .005), although the improvement in mean pain intensity was not significant.
Adverse effects leading to dose modifications or interruption of treatment were reported in 21% of patients on abiraterone, compared with 12% of the prednisone group. Six patients (1%) in each group died of drug-related treatment-emergent AEs. The AEs of “special interest,” the researchers say, included events related to mineralocorticoid excess, such as hypertension, hypokalemia, and fluid retention—all unsurprising, given the known mechanism of action of abiraterone. Grade 3 or 4 AEs with increased alanine aminotransferase and aspartate aminotransferase were more common in the abiraterone group.
The most common subsequent therapy for patients who terminated the study was docetaxel. However, another recent study, from Johns Hopkins researchers in Baltimore, Maryland, indicates the transition warrants caution: The findings suggest a potential cross-resistance between docetaxel and abiraterone.
Their study compared outcomes in 24 men who received abiraterone before docetaxel with 95 who were abiraterone-naïve. Men who were on abiraterone were less likely to achieve a PSA response, and their cancer was more likely to progress.
The researchers concede that their study groups were small; they also say it is possible that differences in disease severity may have influenced the time to progression. However, they say the fact that PSA-PFS was significantly different between the 2 groups (P = .002) supports their initial hypothesis—that is, that abiraterone pretreatment reduces responsiveness to docetaxel.
In spite of its limitations, the researchers say their study represents the only comparative analysis of PSA-PFS and PFS after docetaxel treatment for patients who have or have not received prior abiraterone. Their report, they add, offers the “strongest available evidence to date” of a clinically meaningful cross-resistance between abiraterone and docetaxel. They conclude that their findings provide “valuable information” about which patients are likely to derive the most benefit from docetaxel.
Sources
Rathkopf DE, Smith MR, de Bono JS, et al. Eur Urol. 2014;66(5):815-825.
doi: 10.1016/j.eururo.2014.02.056.
Schewizer MT, Zhou XC, Wang H, et al. Eur Urol. 2014;66(4):646-652.
doi: 10.1016/j.eururo.2014.01.018.
Until recently, there have been few treatment options for advanced prostate cancer that is resistant to androgen-directed therapies. Newer treatments that target residual androgen production offer some hope of prolonging the interval before chemotherapy, with fewer adverse effects (AEs) and better efficacy. One of those is abiraterone, which blocks extragonadal, testicular, and tumor androgen biosynthesis.
An ongoing multinational phase 3 study is evaluating the clinical benefits of abiraterone plus prednisone vs prednisone alone in patients with progressive metastatic castration-resistant prostate cancer (mCRPC). Follow-up for the study has now exceeded 27 months, giving a good opportunity to evaluate safety and efficacy. Thus, after having reviewed outcomes so far, the independent data-monitoring committee recommended that the study be unblinded and patients be allowed to cross over from prednisone to abiraterone. The researchers reported the results of the third interim analysis, with updated analysis.
Patients were stratified by Eastern Cooperative Oncology Group performance status (ECOG-PS) and randomly assigned to receive abiraterone 1,000 mg plus prednisone 5 mg twice daily or placebo plus prednisone.
Patients who received abiraterone had, compared with those on prednisone, statistically significant improvement in radiographic progression-free survival (PFS), with a median time to disease progression or death of 16.5 months, vs 8.2 months (95% CI, 0.45-0.61).
Overall survival also lengthened, from a median of 35.3 months vs 30.1 months (95% CI, 0.66-0.95).
All secondary endpoints also favored abiraterone over prednisone. For instance, abiraterone treatment delayed the time to the need for opiates for cancer-related pain and the time to initiation of chemotherapy. Abiraterone also delayed the time to deterioration in ECOG-PS and prostate-specific antigen (PSA) progression. Abiraterone more than doubled the PSA response rate: 68% vs 29% with prednisone.
Patients reported more pain relief. Those receiving abiraterone had statistically significant improvement in pain interference (P = .005), although the improvement in mean pain intensity was not significant.
Adverse effects leading to dose modifications or interruption of treatment were reported in 21% of patients on abiraterone, compared with 12% of the prednisone group. Six patients (1%) in each group died of drug-related treatment-emergent AEs. The AEs of “special interest,” the researchers say, included events related to mineralocorticoid excess, such as hypertension, hypokalemia, and fluid retention—all unsurprising, given the known mechanism of action of abiraterone. Grade 3 or 4 AEs with increased alanine aminotransferase and aspartate aminotransferase were more common in the abiraterone group.
The most common subsequent therapy for patients who terminated the study was docetaxel. However, another recent study, from Johns Hopkins researchers in Baltimore, Maryland, indicates the transition warrants caution: The findings suggest a potential cross-resistance between docetaxel and abiraterone.
Their study compared outcomes in 24 men who received abiraterone before docetaxel with 95 who were abiraterone-naïve. Men who were on abiraterone were less likely to achieve a PSA response, and their cancer was more likely to progress.
The researchers concede that their study groups were small; they also say it is possible that differences in disease severity may have influenced the time to progression. However, they say the fact that PSA-PFS was significantly different between the 2 groups (P = .002) supports their initial hypothesis—that is, that abiraterone pretreatment reduces responsiveness to docetaxel.
In spite of its limitations, the researchers say their study represents the only comparative analysis of PSA-PFS and PFS after docetaxel treatment for patients who have or have not received prior abiraterone. Their report, they add, offers the “strongest available evidence to date” of a clinically meaningful cross-resistance between abiraterone and docetaxel. They conclude that their findings provide “valuable information” about which patients are likely to derive the most benefit from docetaxel.
Sources
Rathkopf DE, Smith MR, de Bono JS, et al. Eur Urol. 2014;66(5):815-825.
doi: 10.1016/j.eururo.2014.02.056.
Schewizer MT, Zhou XC, Wang H, et al. Eur Urol. 2014;66(4):646-652.
doi: 10.1016/j.eururo.2014.01.018.
Until recently, there have been few treatment options for advanced prostate cancer that is resistant to androgen-directed therapies. Newer treatments that target residual androgen production offer some hope of prolonging the interval before chemotherapy, with fewer adverse effects (AEs) and better efficacy. One of those is abiraterone, which blocks extragonadal, testicular, and tumor androgen biosynthesis.
An ongoing multinational phase 3 study is evaluating the clinical benefits of abiraterone plus prednisone vs prednisone alone in patients with progressive metastatic castration-resistant prostate cancer (mCRPC). Follow-up for the study has now exceeded 27 months, giving a good opportunity to evaluate safety and efficacy. Thus, after having reviewed outcomes so far, the independent data-monitoring committee recommended that the study be unblinded and patients be allowed to cross over from prednisone to abiraterone. The researchers reported the results of the third interim analysis, with updated analysis.
Patients were stratified by Eastern Cooperative Oncology Group performance status (ECOG-PS) and randomly assigned to receive abiraterone 1,000 mg plus prednisone 5 mg twice daily or placebo plus prednisone.
Patients who received abiraterone had, compared with those on prednisone, statistically significant improvement in radiographic progression-free survival (PFS), with a median time to disease progression or death of 16.5 months, vs 8.2 months (95% CI, 0.45-0.61).
Overall survival also lengthened, from a median of 35.3 months vs 30.1 months (95% CI, 0.66-0.95).
All secondary endpoints also favored abiraterone over prednisone. For instance, abiraterone treatment delayed the time to the need for opiates for cancer-related pain and the time to initiation of chemotherapy. Abiraterone also delayed the time to deterioration in ECOG-PS and prostate-specific antigen (PSA) progression. Abiraterone more than doubled the PSA response rate: 68% vs 29% with prednisone.
Patients reported more pain relief. Those receiving abiraterone had statistically significant improvement in pain interference (P = .005), although the improvement in mean pain intensity was not significant.
Adverse effects leading to dose modifications or interruption of treatment were reported in 21% of patients on abiraterone, compared with 12% of the prednisone group. Six patients (1%) in each group died of drug-related treatment-emergent AEs. The AEs of “special interest,” the researchers say, included events related to mineralocorticoid excess, such as hypertension, hypokalemia, and fluid retention—all unsurprising, given the known mechanism of action of abiraterone. Grade 3 or 4 AEs with increased alanine aminotransferase and aspartate aminotransferase were more common in the abiraterone group.
The most common subsequent therapy for patients who terminated the study was docetaxel. However, another recent study, from Johns Hopkins researchers in Baltimore, Maryland, indicates the transition warrants caution: The findings suggest a potential cross-resistance between docetaxel and abiraterone.
Their study compared outcomes in 24 men who received abiraterone before docetaxel with 95 who were abiraterone-naïve. Men who were on abiraterone were less likely to achieve a PSA response, and their cancer was more likely to progress.
The researchers concede that their study groups were small; they also say it is possible that differences in disease severity may have influenced the time to progression. However, they say the fact that PSA-PFS was significantly different between the 2 groups (P = .002) supports their initial hypothesis—that is, that abiraterone pretreatment reduces responsiveness to docetaxel.
In spite of its limitations, the researchers say their study represents the only comparative analysis of PSA-PFS and PFS after docetaxel treatment for patients who have or have not received prior abiraterone. Their report, they add, offers the “strongest available evidence to date” of a clinically meaningful cross-resistance between abiraterone and docetaxel. They conclude that their findings provide “valuable information” about which patients are likely to derive the most benefit from docetaxel.
Sources
Rathkopf DE, Smith MR, de Bono JS, et al. Eur Urol. 2014;66(5):815-825.
doi: 10.1016/j.eururo.2014.02.056.
Schewizer MT, Zhou XC, Wang H, et al. Eur Urol. 2014;66(4):646-652.
doi: 10.1016/j.eururo.2014.01.018.
Disordered methylation compromises CLL treatment

Credit: Christoph Bock
New research suggests disordered methylation is one of the defining characteristics of cancer and helps tumors adapt to changing circumstances.
The study, published in Cancer Cell, showed that disordered methylation has a direct bearing on the effectiveness of cancer therapy.
In patients with chronic lymphocytic leukemia (CLL), researchers found that treatment produced shorter remissions if the tumor tissue showed signs of highly disordered methylation.
The findings indicate that such disorganization can actually benefit tumors and render them less vulnerable to anticancer drugs.
“The behavior of a cancer cell is dictated not only by genetics . . . but also by epigenetics,” said study author Catherine Wu, MD, of the Dana-Farber Cancer Institute in Boston.
“We know that tumors are composed of many subgroups of cells, each with its own array of gene mutations. In this study, we wanted to see if that type of genetic diversity coincides with epigenetic diversity. In other words, does the range of methylation patterns mirror the genetic variety we find in tumors?”
To find out, the researchers used bisulfite sequencing, which allows scientists to track the presence or absence of methyl groups at specific rungs on the DNA ladder.
They also devised a simple measure called PDR—percent discordant reads—for quantifying the extent of irregular methylation within a tissue sample. The higher the PDR, the more variability in how the methyl groups are arranged.
They measured the PDR and the amount of genetic diversity in 104 CLL samples and 27 samples of normal B cells.
“We thought the epigenetic structure would map right onto the genetic structure,” said study author Alexander Meissner, PhD, of the Broad Institute of MIT and Harvard in Cambridge, Massachusetts.
“That is, the degree of genetic diversity in each sample would match the variation in methylation marks in an organized fashion.”
To the researchers’ surprise, the methylation patterns showed a tremendous degree of random disarray.
“We know that individual tumors are checkered with genetically distinct groups of cells,” Dr Meissner explained. “Bisulfite sequencing enabled us to see that the placement of methyl groups across tumor cell DNA also varies substantially among cells in the same tumor. In fact, disorderly methylation pervades the entire tumor.”
The results revealed that the diversity within individual tumors apparently proceeds along two independent, yet interrelated tracks: one resulting in a genetic hodgepodge of cell groups, the other resulting in haphazard methylation.
The methylation irregularities, technically known as “local methylation disorder,” were highly evident in CLL and other types of cancer.
Because methyl groups control the expression of genes, disorderly methylation might be expected to cause wildly inconsistent gene activity even within a single tumor. This, in fact, is what the researchers found.
The disruption of methylation machinery might seem hazardous to tumor survival, but the researchers theorize that tumors can turn the disorderliness to their own advantage.
“Just as in the case of genetic heterogeneity within tumors, increased random variation of the epigenetic profile may augment the diversity of malignant cells,” said study author Dan Landau, MD, PhD, of Dana-Farber and the Broad Institute.
“The ability of cancers to maintain high levels of diversity is an effective hedging strategy, enabling them to better adapt to therapy, as well as enhancing the ‘trial and error’ process in search of better evolutionary trajectories.”
“Cancer survives through some wildly inventive ways,” Dr Wu added. “Methylation disorder is one of the ways it creates the conditions that enable it to adapt.” ![]()

Credit: Christoph Bock
New research suggests disordered methylation is one of the defining characteristics of cancer and helps tumors adapt to changing circumstances.
The study, published in Cancer Cell, showed that disordered methylation has a direct bearing on the effectiveness of cancer therapy.
In patients with chronic lymphocytic leukemia (CLL), researchers found that treatment produced shorter remissions if the tumor tissue showed signs of highly disordered methylation.
The findings indicate that such disorganization can actually benefit tumors and render them less vulnerable to anticancer drugs.
“The behavior of a cancer cell is dictated not only by genetics . . . but also by epigenetics,” said study author Catherine Wu, MD, of the Dana-Farber Cancer Institute in Boston.
“We know that tumors are composed of many subgroups of cells, each with its own array of gene mutations. In this study, we wanted to see if that type of genetic diversity coincides with epigenetic diversity. In other words, does the range of methylation patterns mirror the genetic variety we find in tumors?”
To find out, the researchers used bisulfite sequencing, which allows scientists to track the presence or absence of methyl groups at specific rungs on the DNA ladder.
They also devised a simple measure called PDR—percent discordant reads—for quantifying the extent of irregular methylation within a tissue sample. The higher the PDR, the more variability in how the methyl groups are arranged.
They measured the PDR and the amount of genetic diversity in 104 CLL samples and 27 samples of normal B cells.
“We thought the epigenetic structure would map right onto the genetic structure,” said study author Alexander Meissner, PhD, of the Broad Institute of MIT and Harvard in Cambridge, Massachusetts.
“That is, the degree of genetic diversity in each sample would match the variation in methylation marks in an organized fashion.”
To the researchers’ surprise, the methylation patterns showed a tremendous degree of random disarray.
“We know that individual tumors are checkered with genetically distinct groups of cells,” Dr Meissner explained. “Bisulfite sequencing enabled us to see that the placement of methyl groups across tumor cell DNA also varies substantially among cells in the same tumor. In fact, disorderly methylation pervades the entire tumor.”
The results revealed that the diversity within individual tumors apparently proceeds along two independent, yet interrelated tracks: one resulting in a genetic hodgepodge of cell groups, the other resulting in haphazard methylation.
The methylation irregularities, technically known as “local methylation disorder,” were highly evident in CLL and other types of cancer.
Because methyl groups control the expression of genes, disorderly methylation might be expected to cause wildly inconsistent gene activity even within a single tumor. This, in fact, is what the researchers found.
The disruption of methylation machinery might seem hazardous to tumor survival, but the researchers theorize that tumors can turn the disorderliness to their own advantage.
“Just as in the case of genetic heterogeneity within tumors, increased random variation of the epigenetic profile may augment the diversity of malignant cells,” said study author Dan Landau, MD, PhD, of Dana-Farber and the Broad Institute.
“The ability of cancers to maintain high levels of diversity is an effective hedging strategy, enabling them to better adapt to therapy, as well as enhancing the ‘trial and error’ process in search of better evolutionary trajectories.”
“Cancer survives through some wildly inventive ways,” Dr Wu added. “Methylation disorder is one of the ways it creates the conditions that enable it to adapt.” ![]()

Credit: Christoph Bock
New research suggests disordered methylation is one of the defining characteristics of cancer and helps tumors adapt to changing circumstances.
The study, published in Cancer Cell, showed that disordered methylation has a direct bearing on the effectiveness of cancer therapy.
In patients with chronic lymphocytic leukemia (CLL), researchers found that treatment produced shorter remissions if the tumor tissue showed signs of highly disordered methylation.
The findings indicate that such disorganization can actually benefit tumors and render them less vulnerable to anticancer drugs.
“The behavior of a cancer cell is dictated not only by genetics . . . but also by epigenetics,” said study author Catherine Wu, MD, of the Dana-Farber Cancer Institute in Boston.
“We know that tumors are composed of many subgroups of cells, each with its own array of gene mutations. In this study, we wanted to see if that type of genetic diversity coincides with epigenetic diversity. In other words, does the range of methylation patterns mirror the genetic variety we find in tumors?”
To find out, the researchers used bisulfite sequencing, which allows scientists to track the presence or absence of methyl groups at specific rungs on the DNA ladder.
They also devised a simple measure called PDR—percent discordant reads—for quantifying the extent of irregular methylation within a tissue sample. The higher the PDR, the more variability in how the methyl groups are arranged.
They measured the PDR and the amount of genetic diversity in 104 CLL samples and 27 samples of normal B cells.
“We thought the epigenetic structure would map right onto the genetic structure,” said study author Alexander Meissner, PhD, of the Broad Institute of MIT and Harvard in Cambridge, Massachusetts.
“That is, the degree of genetic diversity in each sample would match the variation in methylation marks in an organized fashion.”
To the researchers’ surprise, the methylation patterns showed a tremendous degree of random disarray.
“We know that individual tumors are checkered with genetically distinct groups of cells,” Dr Meissner explained. “Bisulfite sequencing enabled us to see that the placement of methyl groups across tumor cell DNA also varies substantially among cells in the same tumor. In fact, disorderly methylation pervades the entire tumor.”
The results revealed that the diversity within individual tumors apparently proceeds along two independent, yet interrelated tracks: one resulting in a genetic hodgepodge of cell groups, the other resulting in haphazard methylation.
The methylation irregularities, technically known as “local methylation disorder,” were highly evident in CLL and other types of cancer.
Because methyl groups control the expression of genes, disorderly methylation might be expected to cause wildly inconsistent gene activity even within a single tumor. This, in fact, is what the researchers found.
The disruption of methylation machinery might seem hazardous to tumor survival, but the researchers theorize that tumors can turn the disorderliness to their own advantage.
“Just as in the case of genetic heterogeneity within tumors, increased random variation of the epigenetic profile may augment the diversity of malignant cells,” said study author Dan Landau, MD, PhD, of Dana-Farber and the Broad Institute.
“The ability of cancers to maintain high levels of diversity is an effective hedging strategy, enabling them to better adapt to therapy, as well as enhancing the ‘trial and error’ process in search of better evolutionary trajectories.”
“Cancer survives through some wildly inventive ways,” Dr Wu added. “Methylation disorder is one of the ways it creates the conditions that enable it to adapt.” ![]()
CLL drug can fight AML too, study suggests

Credit: FDA
SAN FRANCISCO—A BCL2 inhibitor that previously proved active against chronic lymphocytic leukemia has shown activity in certain patients with acute myelogenous leukemia (AML) as well.
This phase 2 trial was the first use of the inhibitor, ABT-199 (or venetoclax), in patients with relapsed or refractory AML.
Five of 32 patients treated with ABT-199 achieved a complete response (CR) or CR with incomplete blood count recovery (CRi), and several more had stable disease.
The drug appeared to be particularly active in patients with IDH mutations.
Marina Konopleva, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, presented these results at the 2014 ASH Annual Meeting (abstract 118). The research was funded by AbbVie, Inc., the company developing ABT-199.
The trial was launched on the basis of preclinical studies showing that ABT-199 could kill AML cell lines, patients’ AML cells, and patient-derived AML cells implanted in mice.
The researchers enrolled 32 AML patients, 30 of whom had relapsed or refractory disease. Patients had a median age of 71 (range, 19 to 84), and half were male.
The overall response rate was 15.5%, with 1 patient achieving a CR and 4 patients achieving a CRi. The researchers noted that 3 of the patients who had a CR/CRi had IDH mutations. Two of these patients also achieved minimal residual disease negativity.
The team said these results suggest single-agent ABT-199 can have considerable clinical activity in patients with relapsed or refractory AML, and patients with mutations in IDH genes may be particularly sensitive to the drug.
The researchers also found the median bone marrow blast count in evaluable patients decreased 36% after treatment with ABT-199. And 6 patients (19%) had at least a 50% reduction in bone marrow blasts.
Common adverse events following treatment (occurring in at least 25% of patients) included nausea, diarrhea, fatigue, neutropenia, and vomiting. Grade 3 and 4 adverse events (occurring in 3 or more patients) included febrile neutropenia, anemia, and pneumonia.
No patients died as a result of treatment-related adverse events.
Furthermore, the maximum-tolerated dose was not reached, leaving open the possibility of higher doses in further trials. The next step is to carry out trials combining ABT-199 with other agents. These trials are currently opening at several sites.
AbbVie said ABT-199 will be studied in combination with common AML treatments, and the company is developing ABT-199 for, and evaluating the drug in, several hematologic malignancies. ![]()

Credit: FDA
SAN FRANCISCO—A BCL2 inhibitor that previously proved active against chronic lymphocytic leukemia has shown activity in certain patients with acute myelogenous leukemia (AML) as well.
This phase 2 trial was the first use of the inhibitor, ABT-199 (or venetoclax), in patients with relapsed or refractory AML.
Five of 32 patients treated with ABT-199 achieved a complete response (CR) or CR with incomplete blood count recovery (CRi), and several more had stable disease.
The drug appeared to be particularly active in patients with IDH mutations.
Marina Konopleva, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, presented these results at the 2014 ASH Annual Meeting (abstract 118). The research was funded by AbbVie, Inc., the company developing ABT-199.
The trial was launched on the basis of preclinical studies showing that ABT-199 could kill AML cell lines, patients’ AML cells, and patient-derived AML cells implanted in mice.
The researchers enrolled 32 AML patients, 30 of whom had relapsed or refractory disease. Patients had a median age of 71 (range, 19 to 84), and half were male.
The overall response rate was 15.5%, with 1 patient achieving a CR and 4 patients achieving a CRi. The researchers noted that 3 of the patients who had a CR/CRi had IDH mutations. Two of these patients also achieved minimal residual disease negativity.
The team said these results suggest single-agent ABT-199 can have considerable clinical activity in patients with relapsed or refractory AML, and patients with mutations in IDH genes may be particularly sensitive to the drug.
The researchers also found the median bone marrow blast count in evaluable patients decreased 36% after treatment with ABT-199. And 6 patients (19%) had at least a 50% reduction in bone marrow blasts.
Common adverse events following treatment (occurring in at least 25% of patients) included nausea, diarrhea, fatigue, neutropenia, and vomiting. Grade 3 and 4 adverse events (occurring in 3 or more patients) included febrile neutropenia, anemia, and pneumonia.
No patients died as a result of treatment-related adverse events.
Furthermore, the maximum-tolerated dose was not reached, leaving open the possibility of higher doses in further trials. The next step is to carry out trials combining ABT-199 with other agents. These trials are currently opening at several sites.
AbbVie said ABT-199 will be studied in combination with common AML treatments, and the company is developing ABT-199 for, and evaluating the drug in, several hematologic malignancies. ![]()

Credit: FDA
SAN FRANCISCO—A BCL2 inhibitor that previously proved active against chronic lymphocytic leukemia has shown activity in certain patients with acute myelogenous leukemia (AML) as well.
This phase 2 trial was the first use of the inhibitor, ABT-199 (or venetoclax), in patients with relapsed or refractory AML.
Five of 32 patients treated with ABT-199 achieved a complete response (CR) or CR with incomplete blood count recovery (CRi), and several more had stable disease.
The drug appeared to be particularly active in patients with IDH mutations.
Marina Konopleva, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, presented these results at the 2014 ASH Annual Meeting (abstract 118). The research was funded by AbbVie, Inc., the company developing ABT-199.
The trial was launched on the basis of preclinical studies showing that ABT-199 could kill AML cell lines, patients’ AML cells, and patient-derived AML cells implanted in mice.
The researchers enrolled 32 AML patients, 30 of whom had relapsed or refractory disease. Patients had a median age of 71 (range, 19 to 84), and half were male.
The overall response rate was 15.5%, with 1 patient achieving a CR and 4 patients achieving a CRi. The researchers noted that 3 of the patients who had a CR/CRi had IDH mutations. Two of these patients also achieved minimal residual disease negativity.
The team said these results suggest single-agent ABT-199 can have considerable clinical activity in patients with relapsed or refractory AML, and patients with mutations in IDH genes may be particularly sensitive to the drug.
The researchers also found the median bone marrow blast count in evaluable patients decreased 36% after treatment with ABT-199. And 6 patients (19%) had at least a 50% reduction in bone marrow blasts.
Common adverse events following treatment (occurring in at least 25% of patients) included nausea, diarrhea, fatigue, neutropenia, and vomiting. Grade 3 and 4 adverse events (occurring in 3 or more patients) included febrile neutropenia, anemia, and pneumonia.
No patients died as a result of treatment-related adverse events.
Furthermore, the maximum-tolerated dose was not reached, leaving open the possibility of higher doses in further trials. The next step is to carry out trials combining ABT-199 with other agents. These trials are currently opening at several sites.
AbbVie said ABT-199 will be studied in combination with common AML treatments, and the company is developing ABT-199 for, and evaluating the drug in, several hematologic malignancies. ![]()
Crisis Mode and Information Exchange
Using electronic health records to improve the continuity of care between hospital units does not replace the need for interpersonal communication to improve transitions of care. Hospital personnel play a critical role in accurately exchanging patient information during patient transfers, a process requiring accurate communication between hospital units to prevent system failures.[1] Because poor communication contributes to preventable adverse events,[2] and effective communication during handoffs decreases medical errors and readmissions,[3] hospitals need to ensure their work environments are conducive to effective communication.
Individuals working under time constraints and heavy workloads could potentially be at high risk of misinterpreting or delivering inaccurate information,[4] partially due to limited ability to accurately process and communicate information under stressful circumstances. Furthermore, because time‐constrained decision makers tend to use less information and less rigorous decision strategies,[5] work climates characterized by staff members doing too many things too quickly could cause patient health information to be lost during transitions of care across hospital units.
Current studies illustrate scenarios in which demanding or time‐constrained work environments caused information exchange errors. One study found that the increased rate of prescribing errors was partially attributed to a high‐demand work environment characterized by working after hours and multitasking.[6] Other studies found that clinicians' limited time to relay and respond to information and ask clarifying questions during patient handoffs was partially attributed to the fast‐faced and chaotic environment of the emergency room.[7, 8] These studies are consistent with another study that found patient handoffs between emergency departments and inpatient wards were inadequate, partially due to less interactive and more rushed communication.[9] The fact that communication breakdowns are widely cited as barriers to patient handoffs[7, 8, 10] and facilitators of medical errors,[7, 8] further underscores the detrimental effect that crisis mode work climates could have on exchanging patient information during transitions of care.
The objective of this analysis was to evaluate the extent to which a crisis mode work climate impacts the occurrence of patient information exchange problems. Estimating associations between hospital staff members' perceptions of crisis mode work climates and perceptions of information exchange problems provide insights as to whether high‐demand and time‐constrained work climates negatively impact the exchange of patient information. Because hospital staff members working under time constraints and heavy workloads could potentially be at high risk of misinterpreting or delivering inaccurate information, we hypothesized that higher levels of a perceived crisis mode work climate would be associated with higher levels of perceived problems with information exchange across hospital units.
METHODS
Data Source
Data originated from the Agency of Healthcare Research and Quality 2010 Hospital Survey on Patient Safety Culture. This validated survey, designed to assess the safety climate within acute‐care settings, remains an important annual survey deployed each year to track changes and factors impacting patient safety.[11] We included only those respondents who self‐reported their position as a nurse, physician, pharmacist, dietician, therapist, technician, patient care assistant, or hospital unit secretary, all of whom are likely responsible for exchanging patient information across hospital units. For this reason, we excluded respondents who self‐reported their position as administrative or miscellaneous. Applying these exclusion criteria resulted in 247,104 respondents across 884 hospitals.
Conceptual Framework
The relationship between perceived crisis mode work climates and patient information exchange problems is likely influenced by staff skill levels, work climate, and infrastructure (Figure 1). With respect to skill levels, hospital staff members with many years of experience compared to those with fewer years may be relatively desensitized to chaotic work environments and consequently have higher thresholds for perceiving crisis modes. Number of hours worked per week likely impacts perceived crisis mode as illustrated in 1 study finding that full‐time nurses reported a significantly lower work pace compared to part‐time nurses.[12] Years of experience likely impacts perception of information exchange problems, particularly if staff members with many years of experience are familiar enough with hospital systems or protocols to easily detect exchange errors or mishaps.
With respect to work climates, workers' perception of cooperation, coordination and patient safety, and specific hospital unit likely impact perceptions of crisis work mode and information exchange problems. For example, hospital staff members reporting high levels of cooperation, coordination, and patient safety likely perceive fewer crisis modes and information exchange problems compared to those in less‐cooperative hospital units. Furthermore, the heterogeneity of work cultures across departments within a hospital results in department‐specific perceptions of crisis mode climates and information exchange problems. Infrastructure factors, such as hospital size, teaching and ownership status, and census region, likely impact the amount of resources available for staffing and infrastructure, which in turn could impact work pace and information exchange accuracy.
Variable Definitions
We defined our predictor as the perceived presence of a crisis mode work climate as captured from the survey questionnaire item: We work in crisis mode trying to do too much, too quickly. This question item had a Likert response scale comprised of the following 5 answer choices: (1) strongly disagree, (2) disagree, (3) neutral, (4) agree, (5) strongly agree. We created a 3‐level response variable by aggregating the agree and disagree responses, respectively, as the first 2 levels, and retaining the neutral response as the third level. Consequently, those responding strongly disagree or disagree were classified as working in lowcrisis mode work climates and those responding strongly agree or agree were classified as working in high crisis mode work climates. We defined our outcome measure as the presence of patient information exchange problems as captured from the survey questionnaire item: Problems often occur in the exchange of information across hospital units. Because this question item had a Likert response scale similar to the crisis mode question predictor, we also created a 3‐level categorical variable in the same fashion. Consequently, those responding strongly disagree or disagree were classified as perceiving no problems exchanging patient information, and those responding strongly agree or agree were classified as perceiving problems exchanging patient information. For the fewer than 10% of the respondents with missing data on either the predictor our outcome variables, the mode measure of central tendency was imputed, a methodology validated in a previous study.[13]
We also included questionnaire items that captured staff skill levels, work climate, and infrastructure as covariates to account for potential confounders (Figure 1). The staff skill levels domain included years of experience working in the hospital, specialty, and unit; current staff position; and extent of patient contact. The work climate domain included respondent perceptions of coordination and cooperation, patient safety, and primary work area or unit in which the provider reported working. The hospital infrastructure domain included bed size, census region, teaching status, and government ownership status. For the fewer than 10% of the respondents with missing data on any of the categorical variables, the mode measure of central tendency was imputed, a methodology validated in a previous study.[13]
Analytic Approach
We used multivariable ordinal regressions to estimate the likelihood of perceived problems in patient information exchange conditional upon perceptions of a crisis mode work climate, controlling for staff skill levels, work climate, and hospital infrastructure. Our estimates therefore reflect the likelihood of hospital staff responding strongly agree or agree to the question Problems often occur in the exchange of information across hospital units conditional upon responding strongly agree or agree to the question We work in crisis mode trying to do too much, too quickly. In addition to controlling for hospital‐specific response rates, we also adjusted our standard errors to account for the clustering of respondents within hospitals. All analyses were conducted in SAS version 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
The hospital sample averaged 279 respondents per hospital with a 56% response rate. Most hospitals were located in the Central region of the United States, and 32% and 19% were teaching and government‐owned hospitals, respectively. Forty‐three percent and 44% of the hospitals in the sample were designated as small and medium hospitals, respectively (Table 1).
| Characteristics | % |
|---|---|
| |
| Hospital characteristics, N=884 | |
| Bed size | |
| Small, 199 | 43.5 |
| Medium, 100399 | 43.8 |
| Large, 400 plus | 12.7 |
| Teaching status | |
| Yes | 32.2 |
| No | 67.8 |
| Government ownership | |
| Yes | 19.5 |
| No | 80.5 |
| Census region | |
| Mid‐Atlantic and New England | 8.7 |
| South Atlantic | 14.8 |
| Central | 57.2 |
| Mountain | 7.7 |
| Pacific | 11.5 |
| Response rate, mean (SD) | 0.56 (0.28) |
| Respondents per hospital, mean (SD) | 279 (358) |
| Respondent characteristics, N=274,140 | |
| How long have you worked in your current specialty or profession? | |
| <1 year | 5.8 |
| 15 years | 32.8 |
| 610 years | 16.2 |
| 1115 years | 12.0 |
| 1620 years | 10.6 |
| 21 years | 22.7 |
| How long have you worked in this hospital? | |
| <1 year | 9.8 |
| 15 years | 42.8 |
| 610 years | 17.8 |
| 1115 years | 9.0 |
| 1620 years | 8.2 |
| 21 years | 12.4 |
| How long have you worked in your current hospital work area/unit? | |
| <1 year | 13.1 |
| 15 years | 48.0 |
| 610 years | 18.1 |
| 1115 years | 8.1 |
| 1620 years | 6.0 |
| 21 years | 6.7 |
| Typically, how many hours per week do you work in this hospital? | |
| <20 hours | 4.8 |
| 2039 hours | 39.9 |
| 4059 hours | 48.8 |
| 6079 hours | 4.2 |
| 8099 hours | 2.1 |
| 100 hours | 0.11 |
| What is your staff position in this hospital? | |
| Registered nurse | 51.2 |
| Technician (eg, ECG, lab, radiology) | 14.1 |
| Unit assistant/clerk/secretary | 8.5 |
| Patient care assistant/hospital aide/care partner | 7.4 |
| Physical, occupational, or speech therapist | 3.7 |
| Attending/staff physician | 3.5 |
| LVN/LPN | 3.0 |
| Respiratory therapist | 2.9 |
| Pharmacist | 2.2 |
| Physician assistant/nurse practitioner | 1.4 |
| Resident physician/physician in training | 1.2 |
| Dietician | 0.83 |
| In your staff position, do you typically have direct interaction or contact with patients? | |
| Yes | 86.6 |
| No | 13.4 |
| What is your primary work area or unit in this hospital? | |
| Other | 27.7 |
| Medicine (nonsurgical) | 11.1 |
| Surgery | 10.0 |
| Intensive care unit (any type) | 8.6 |
| Many different hospital units/no specific unit | 6.8 |
| Radiology | 6.2 |
| Emergency department | 5.8 |
| Obstetrics | 4.9 |
| Laboratory | 4.9 |
| Rehabilitation | 4.2 |
| Pediatrics | 3.8 |
| Pharmacy | 3.2 |
| Psychiatry/mental health | 2.1 |
| Anesthesiology | 0.55 |
Thirty‐seven percent of the respondents have worked in their current specialty or profession for 5 years or less (Table 1). Over half of the respondents have worked in their current hospital for 5 years or less, whereas 61% have worked in their current unit within the hospital for 5 years or less. Forty‐nine percent work at least 40 hours per week. Registered nurses and technicians represented the 2 largest subgroups of staff positions, comprising 51% and 14% of the sample, respectively. Dieticians and resident physicians, on the other hand, represented the 2 smallest subgroups of staff positions, comprising 0.83% and 1.2% of the sample, respectively. Eighty‐seven percent of the respondents have direct interaction or contact with patients. Apart from those responding other as their hospital unit, nonsurgical medicine and surgery represented the largest subgroup primary work areas, comprising 11% and 10% of the sample, respectively. In contrast, psychiatry and anesthesiology represented the 2 smallest subgroups of primary work areas, comprising 2.1% and 0.55% of the sample, respectively (Table 1).
Respondents scored relatively high with regard to teamwork and helping each other out under hurried or busy circumstances. For example, 85% agreed or strongly agreed that their unit worked together as a team to get work done when a lot of work needed to be completed quickly, and 68% agreed or strongly agreed that individuals within their unit helped out when an area in their unit became busy (Table 1). Despite this cooperation, 31% agreed or strongly agreed that hospital units did not coordinate well together. Paradoxically, 57% agreed or strongly agreed that there was good cooperation among hospital units that needed to work together. Seventy‐five percent of the respondents reported excellent or very good patient safety levels within their unit, although 53% agreed or strongly agreed that staff worked longer hours than was best for patient care (Table 1).
With regard to perceived crisis mode work climate, 32% and 47% reported agreeing and disagreeing, respectively, that their work unit worked in crisis mode trying to do too much too quickly (Table 2). With regard to perceived problems with patient information exchange, 27% and 36% reported agreeing and disagreeing, respectively, that information exchange problems occurred across hospital units (Table 2).
| Perceptions | % |
|---|---|
| We work in crisis mode trying to do too much, too quickly | |
| Strongly disagree | 8.1 |
| Disagree | 39.2 |
| Neutral | 21.0 |
| Agree | 24.3 |
| Strongly agree | 7.5 |
| Problems often occur in the exchange of information across hospital units | |
| Strongly disagree | 4.6 |
| Disagree | 31.3 |
| Neutral | 37.3 |
| Agree | 24.0 |
| Strongly agree | 2.7 |
| When a lot of work needs to be done quickly, we work together as a team to get the work done. | |
| Strongly disagree | 1.5 |
| Disagree | 6.1 |
| Neutral | 7.5 |
| Agree | 53.6 |
| Strongly agree | 31.2 |
| When one area in this unit gets really busy, others help out. | |
| Strongly disagree | 3.9 |
| Disagree | 13.9 |
| Neutral | 13.7 |
| Agree | 52.6 |
| Strongly agree | 15.8 |
| Hospital units do not coordinate well with each other. | |
| Strongly disagree | 5.6 |
| Disagree | 38.8 |
| Neutral | 23.7 |
| Agree | 25.3 |
| Strongly agree | 6.6 |
| There is good cooperation among hospital units that need to work together. | |
| Strongly disagree | 2.7 |
| Disagree | 15.1 |
| Neutral | 24.7 |
| Agree | 51.1 |
| Strongly agree | 6.3 |
| Please give your work area/unit in this hospital an overall grade on patient safety. | |
| Excellent | 23.0 |
| Very good | 49.8 |
| Acceptable | 21.8 |
| Poor | 4.6 |
| Failing | 0.76 |
| Staff in this unit work longer hours than is best for patient care. | |
| Strongly disagree | 11.5 |
| Disagree | 42.2 |
| Neutral | 23.6 |
| Agree | 18.4 |
| Strongly agree | 6.3 |
In the unadjusted analyses, crisis mode perceptions and information exchange problem perceptions were significantly associated. Among those who agreed that their work unit worked in crisis mode, a larger proportion of respondents agreed (41%) versus disagreed (24%) that problems often occurred in exchanging patient information across units (Table 3). In contrast, among those who disagreed that their work unit worked in crisis mode, a larger proportion of respondents disagreed (47%) versus agreed (19%) that problems often occurred in exchanging patient information across units (Table 3).
| Problems Often Occur in Exchange of Information Across Hospital Units | |||
|---|---|---|---|
| Agree (N=66,115), Row %* | Neutral (N=92,228), Row % | Disagree (N=88,797), Row % | |
| |||
| Crisis Mode Work Climate | |||
| Agree (N=78,253) | 40.8 | 35.4 | 23.8 |
| Neutral (N=51,836)‖ | 22.9 | 48.9 | 28.2 |
| Disagree (N=116,781) | 19.0 | 33.5 | 47.5 |
In the multivariable ordinal regression, compared to those who disagreed that their unit worked in crisis mode, those who agreed were 1.6 times more likely to report that problems often occurred in exchanging patient information across units (odds ratio [OR]: 1.6, 95% confidence interval [CI]: 1.58‐1.65) (Table 4). Additionally, some key covariates were independently associated with perceptions of information exchange problems. Two of these covariates measured workplace coordination. Those who reported that hospital units did not cooperate well together were more likely to report problematic information exchange compared to those who reported that hospital units did cooperate well (OR: 4.7, 95% CI: 4.35.0). Relatedly, those who reported that hospital units did coordinate well were less likely to report problematic information exchange compared to those who reported that hospital units did not coordinate well (OR: 0.10, 95% CI: 0.10‐0.11). Two other covariates measured patient contact and perceptions about long working hours. Those who reported having direct interaction or contact with patients were less likely to report problematic information exchange compared to those who reported not having direct interaction or contact with patients (OR: 0.85, 95% CI: 0.83‐0.87). Those who reported that staff did not work longer hours than was better for patient care were less likely to report problematic information exchange compared to those who did report working longer hours than was better for patient care (OR: 0.76, 95% CI: 0.73 0.79). One covariate measured hospital size. Those who reported working in smaller hospitals were less likely to report problematic information exchange compared to those reporting working in large hospitals (OR: 0.66, 95% CI 0.59‐0.75) (Table 4).
| Characteristic | Unadjusted OR (95% CI) | Adjusted OR* (95% CI) |
|---|---|---|
| ||
| Primary predictor of interest | ||
| Crisis mode work climate | ||
| Agree | 3.0 (2.9‐3.1) | 1.6 (1.5‐1.6) |
| Neutral | 1.8 (1.7‐1.8) | 1.3 (1.2‐1.3) |
| Disagree | Reference | Reference |
| Hospital characteristics | ||
| Bed Size | ||
| Small, 624 | 0.51 (0.44‐0.59) | 0.66 (0.59‐0.75) |
| Small, 249 | 0.59 (0.53‐0.66) | 0.77 (0.70‐0.84) |
| Small, 5099 | 0.65 (0.58‐0.73) | 0.78 (0.71‐0.84) |
| Medium, 100199 | 0.85 (0.77‐0.95) | 0.92 (0.86‐1.0) |
| Medium, 200299 | 1.0 (0.98‐1.1) | 0.97 (0.90‐1.0) |
| Medium, 300399 | 0.96 (0.85‐1.1) | 1.0 (0.92‐1.1) |
| Large, 400499 | 0.99 (0.86‐1.1) | 0.96 (0.87‐1.0) |
| Large, 500 plus | Reference | Reference |
| Teaching status | ||
| No | 0.81 (0.76‐0.87) | 1.0 (0.95‐1.0) |
| Yes | Reference | Reference |
| Government ownership | ||
| No | 1.1 (1.01.2) | 1.0 (0.98‐1.1) |
| Yes | Reference | Reference |
| Census region | ||
| Mid‐Atlantic and New England | 1.0 (0.88‐1.1) | 0.91 (0.84‐0.99) |
| South Atlantic | 0.95 (0.85‐1.1) | 1.0 (0.95‐1.1) |
| Central 1 | 0.95 (0.85‐1.0) | 0.95 (0.89‐1.0) |
| Central 2 | 0.71 (0.62‐0.81) | 0.91 (0.83‐0.99) |
| Central 3 | 0.80 (0.71‐0.91) | 0.97 (0.90‐1.0) |
| Central 4 | 0.76 (0.68‐0.86) | 0.93 (0.85‐1.0) |
| Mountain | 0.84 (0.73‐0.96) | 0.98 (0.90‐1.1) |
| Pacific | Reference | Reference |
| Average survey response rate within hospital | 0.65 (0.58‐0.72) | 0.93 (0.82‐1.0) |
| Respondent characteristics | ||
| How long have you worked in your current specialty or profession? | ||
| <1 year | 0.75 (0.73‐0.78) | 1.03 (0.99‐1.1) |
| 15 years | 0.99 (0.97‐1.0) | 1.1 (1.1‐1.1) |
| 610 years | 1.0 (1.01.1) | 0.99 (0.96‐1.0) |
| 1115 years | 1.0 (1.01.1) | 1.0 (0.97‐1.0) |
| 1620 years | 1.0 (0.98‐1.0) | 0.97 (0.94‐1.0) |
| 21 years | Reference | Reference |
| How long have you worked in this hospital? | ||
| <1 year | 0.75 (0.73‐0.77) | 0.90 (0.85‐0.90) |
| 15 years | 1.03 (1.001.05) | 0.99 (0.95‐1.0) |
| 610 years | 1.1 (1.1‐1.1) | 0.99 (0.95‐1.0) |
| 1115 years | 1.1 (1. 01.1) | 1.0 (0.96‐1.0) |
| 1620 years | 1.1 (1.01.1) | 0.98 (0.94‐1.0) |
| 21 years | Reference | Reference |
| How long have you worked in your current hospital work area/unit? | ||
| <1 year | 0.79 (0.76‐0.82) | 0.98 (0.93‐1.0) |
| 15 years | 1.0 (1.01.1) | 1.0 (0.99‐1.1) |
| 610 years | 1.1 (1.1‐1.1) | 1.0 (1.01.1) |
| 1115 years | 1.1 (1.01.1) | 1.0 (0.99‐1.1) |
| 1620 years | 1.1 (1.01.1) | 1.1 (1.01.1) |
| 21 years | Reference | Reference |
| Typically, how many hours per week do you work in this hospital? | ||
| <20 | 0.63 (0.50‐0.79) | 0.91 (0.72‐1.2) |
| 2039 | 0.75 (0.59‐0.94) | 0.90 (0.71‐1.1) |
| 4059 | 0.87 (0.69‐1.1) | 1.1 (0.85‐1.4) |
| 6079 | 0.95 (0.75‐1.2) | 1.0 (0.82‐1.3) |
| 8099 | 0.99 (0.78‐1.2) | 1.1 (0.86‐1.4) |
| 100 | Reference | Reference |
| What is your staff position in this hospital? | ||
| Registered nurse | 0.92 (0.90‐0.94) | 1.1 (0.98‐1.0) |
| Technician (eg, ECG, lab, radiology) | Reference | Reference |
| Unit assistant/clerk/secretary | 0.79 (0.76‐0.81) | 0.94 (0.80‐0.96) |
| Patient care assistant/hospital aide/care partner | 0.78 (0.75‐0.81) | 0.96 (0.90‐0.98) |
| Physical, occupational, or speech therapist | 0.88 (0.84‐0.92) | 1.2 (1.1‐1.2) |
| Attending/staff physician | 1.0 (0.97‐1.1) | 1.3 (1.2‐1.3) |
| LVN/LPN | 0.89 (0.85‐0.94) | 1.0 (0.92‐1.0) |
| Respiratory therapist | 0.84 (0.80‐0.88) | 0.97 (0.89‐1.0) |
| Pharmacist | 1.5 (1.4‐1.6) | 1.3 (1.1‐1.3) |
| Physician assistant/nurse practitioner | 0.93 (0.87‐1.0) | 1.2 (1.1‐1.2) |
| Resident physician/physician in training | 0.96 (0.89‐1.0) | 1.3 (1.2‐1.4) |
| Dietician | 0.86 (0.79‐0.94) | 1.2 (1.1‐1.3) |
| In your staff position, do you typically have direct interaction or contact with patients? | ||
| Yes | 0.83 (0.82‐0.85) | 0.85 (0.83‐0.87) |
| No | Reference | Reference |
| What is your primary work area or unit in this hospital? | ||
| Other | Reference | Reference |
| Medicine (nonsurgical) | 1.1 (1.01.1) | 0.84 (0.82‐0.89) |
| Surgery | 1.1 (1.1‐1.2) | 0.88 (0.86‐0.91) |
| Intensive care unit (any type) | 0.93 (0.90‐0.96) | 0.78 (0.76‐0.81) |
| Many different hospital units/no specific unit | 1.2 (1.1‐1.2) | 1.0 (0.98‐ 1.0) |
| Radiology | 1.1 (1.1‐1.1) | 1.0 (1.01.1) |
| Emergency department | 1.0 (0.97‐1.0) | 0.57 (0.55‐0.60) |
| Obstetrics | 0.76 (0.73‐0.79) | 0.66 (0.63‐0.69) |
| Laboratory | 1.2 (1.2‐1.3) | 1.0 (1.01.1) |
| Rehabilitation | 1.0 (0.97‐1.0) | 1.0 (0.98‐1.1) |
| Pediatrics | 0.90 (0.86‐0.94) | 0.83 (0.80‐0.87) |
| Pharmacy | 1.6 (1.5‐1.7) | 1.1 (1.01.2) |
| Psychiatry/mental health | 1.2 (1.1‐1.2) | 0.96 (0.90‐1.0) |
| Anesthesiology | 1.1 (1.01.3) | 0.93 (0.83‐1.0) |
| Respondent perceptions | ||
| When a lot of work needs to be done quickly, we work together as a team to get the work done. | ||
| Strongly disagree | 3.2 (3.03.4) | 1.0 (0.98‐1.1) |
| Disagree | 3.2 (3.13.3) | 1.0 (1.01.1) |
| Neutral | 2.3 (2.2‐2.4) | 0.98 (0.94‐1.0) |
| Agree | 2.3 (2.2‐2.4) | 1.0 (1.002‐1.04) |
| Strongly agree | Reference | Reference |
| Staff in this unit work longer hours than is best for patient care. | ||
| Strongly disagree | 0.51 (0.48‐0.53) | 0.76 (0.73‐0.79) |
| Disagree | 0.68 (0.67‐0.70) | 0.81 (0.78‐0.84) |
| Neutral | 0.94 (0.91‐0.97) | 0.93 (0.90‐0.97) |
| Agree | 1.0 (0.99‐1.1) | 0.94 (0.91‐0.98) |
| Strongly agree | Reference | Reference |
| When 1 area in this unit gets really busy, others help out. | ||
| Strongly disagree | 3.8 (3.7 ‐ 4.0) | 1.0 (0.96‐1.1) |
| Disagree | 3.0 (2.9‐3.1) | 1.0 (0.99‐1.1) |
| Neutral | 2.2 (2.12.3) | 1.0 (0.97‐1.0) |
| Agree | 1.5 (1.5‐1.6) | 0.99 (0.96‐1.0) |
| Strongly agree | Reference | Reference |
| Hospital units do not coordinate well with each other. | ||
| Strongly disagree | 0.03 (0.03‐0.04) | 0.10 (0.10‐0.11) |
| Disagree | 0.08 (0.08‐0.08) | 0.18 (0.17‐0.19) |
| Neutral | 0.21 (0.20‐0.22) | 0.32 (0.30‐0.33) |
| Agree | 0.50 (0.48‐0.52) | 0.61 (0.58‐0.63) |
| Strongly agree | Reference | Reference |
| There is good cooperation among hospital units that need to work together. | ||
| Strongly disagree | 20.1 (18.921.5) | 4.7 (4.35.0) |
| Disagree | 14.2 (13.614.9) | 4.2 (4.14.5) |
| Neutral | 6.7 (6.47.0) | 2.7 (2.6‐2.8) |
| Agree | 2.4 (2.3‐2.5) | 1.6 (1.6‐1.7) |
| Strongly agree | Reference | Reference |
| Please give your work area/unit in this hospital an overall grade on patient safety | ||
| Excellent | 0.13 (0.12‐0.14) | 0.47 (0.42‐0.52) |
| Very good | 0.24 (0.21‐0.26) | 0.63 (0.57‐0.70) |
| Acceptable | 0.49 (0.45‐0.54) | 0.79 (0.72‐0.88) |
| Poor | 0.83 (0.75‐0.92) | 0.92 (0.83‐1.03) |
| Failing | Reference | Reference |
DISCUSSION
Our results illustrate that when hospital staff agree that their hospital works in crisis mode, they are more likely to agree that their hospital unit had frequent problems exchanging patient information across units. Because hospital staff working under time constraints and heavy workloads could potentially be at risk of misinterpreting or delivering inaccurate information, these results imply that crisis mode work climates increase the risk of problematic health information exchange. An equally plausible interpretation could be that problematic patient health information exchange increases the risk of hospital staff perceiving crisis mode work climates. Given that information gaps are associated with patient handoff errors,[14] and that patient handoff errors are associated with adverse events,[2, 3, 6, 8] an urgent need exists to implement information exchange systems that prevent information gaps from harming patients. Consequently, hospitals need to implement workflow strategies that prevent information gaps from undermining patient safety during transitions of care.
Other factors affect information exchange apart from crisis mode work climate, as illustrated by the significant associations of key covariates in the multivariate model. The effect found between perceived coordination and information exchange implies that improving information exchange requires good cooperation and coordination. The effect found between patient contact and information exchange implies that working directly with patients improves either the accuracy or the perception of information exchange. Finally, the effect found between hospital size and information exchange suggests that small hospitals are less likely than large hospitals to have information exchange problem. The geographical dispersion and the complexity of larger institutions could result in information exchange problems due to more confusion and less in‐person communication.
Because problematic patient information exchanges are associated with hospital size, coordination, and patient contact, in addition to crisis mode work climate, multifaceted solutions are necessary to resolve the problem. For example, hospital interventions designed to improve coordination could in turn attenuate perceived crisis modes. Furthermore, tailoring these interventions to hospitals that belong to complex geographically dispersed provider networks would likely decrease errors during transitions of care. Because multiple factors cause information exchange problems, implementing interventions that improve both coordination and crisis mode work climates would likely result in a greater net improvement compared to interventions focused solely on decreasing crisis mode work climates.
Some limitations of our paper are worth noting. First, we did not have information on the volume of data exchanged or the functionality levels of the electronic health record systems, both of which likely impact the accuracy of patient information exchange. For example, hospitals with smaller versus larger amounts of data exchanged could be less prone to error. On the other hand, this risk of error could be reduced even further by implementing robust health information technology (IT) systems that improve the accuracy of information transfer. This is consistent with studies showing that hospitals without computerized provider order entry (CPOE) systems have been shown to have higher medication error rates compared to those hospitals with CPOE systems.[15] Therefore, omitting data volume and health IT capabilities from the multivariate model could introduce unobserved heterogeneity, resulting in biased associations between perceived crisis mode work climate and perceived information exchange problems. Second, the cross‐sectional design limits our ability to infer causality because we are not certain whether the perceived crisis mode occurred before, after, or simultaneously to perceived information exchange problems. Third, the self‐reported nature of the questionnaire items does not provide information on observed levels of crisis mode and exchange problems, which could be inconsistent with perceived levels. Fourth, the relatively low within‐hospital response rate decreases the external validity of our findings. For example, if responders' perceptions of crisis mode or information exchange problems significantly differed from nonresponders, our results would not be generalizable to the larger population of acute‐care hospitals across the United States. Therefore, conclusions should be viewed with caution if applying these results to hospitals with respondents significantly differing from those contained within our sample.
Despite these limitations, the large sample size in conjunction with the use of data from a survey having acceptable psychometric properties[16] strengthens the external and internal validity of our findings. Although questionnaire items measuring perceptions are relatively subjective in nature compared to using metrics that capture observed problems or crisis modes, we argue that staff perception data are equally informative, as they guide organization leaders on how to improve workplace performance. Because a core concept of high reliability organizations (HROs) is to preserve constant awareness by key leaders and staff of the state of the systems and processes that affect patient care,[17] HROs could benefit from knowing the extent to which staff perceptions impact patient care. From a methods perspective, the multivariable ordinal regressions enabled us to control for potential confounders that if omitted could have resulted in biased estimates. Furthermore, low levels of multicollinearity as illustrated by low variation inflation factors enabled us to isolate the independent effect of crisis mode perceptions. Including hospital size and hospital work unit as covariates was an additional methodological strength helping account for the unobserved heterogeneity caused by excluding volume of data exchanged or health IT system capability. For example, because larger compared to smaller hospitals usually have more sophisticated health IT systems,[15] including bed size in the model theoretically captures some of the variation that would have been captured if we were able to include a covariate measuring health IT capability. Last, using ordinal regression facilitates interpretation of the findings because the questionnaire items for the predictor and outcome were originally captured on a Likert scale.
Our findings underscore the significant impact that work climate has on accurate information exchange, and ultimately patient safety. Improving patient safety is imperative for hospitals, especially within the context of recent regulations stemming from the Affordable Care Act that incentivize hospitals to reduce readmissions[18] and improve transitions of care.[19] Because accurate health information exchange is a critical component of patient care, resolving barriers that decrease the accuracy of this exchange is essential. Therefore, future studies need to continue examining these associations within the context of study designs that incorporate longitudinal data and datasets that include objective measures capturing crisis mode work climates and information exchange problems. Because effective communication during handoffs is associated with decreases in medical errors and readmissions, hospitals need to continually ensure that work environments are conducive to effective patient information exchange.
Disclosures
Nothing to report
- , , . Safety and complexity: inter‐departmental relationships as a threat to patient safety in the operating department. J Health Organ Manag. 2006;20(2–3):227–242.
- , , , , . Communication loads on clinical staff in the emergency department. Med J Aust. 2002;176(9):415–418.
- , , , et al. Rates of medical errors and preventable adverse events among hospitalized children following implementation of a resident handoff bundle. JAMA. 2013;310(21):2262–2270.
- , , . The Handbook of Group Communication Theory and Research. Thousand Oaks, CA: Sage Publications; 1999.
- , , . Judgment and decision making under time pressure. In: Svenson O, Maule AJ, eds. Time Pressure and Stress in Human Judgment and Decision Making. New York, NY: Plenum Press; 1993:27–40.
- , , , , . Learning from error: identifying contributory causes of medication errors in an Australian hospital. Med J Aust. 2008;188(5):276–279.
- , , . Communicating in the “gray zone”: perceptions about emergency physician hospitalist handoffs and patient safety. Acad Emerg Med. 2007;14(10):884–894.
- , . Emergency physician intershift handovers: an analysis of our transitional care. Pediatr Emerg Care. 2006;22(10):751–754.
- , , , , , . Dropping the baton: a qualitative analysis of failures during the transition from emergency department to inpatient care. Ann Emerg Med. 2009;53(6):701–710.
- , , , . Lost in translation: challenges and opportunities in physician‐to‐physician communication during patient handoffs. Acad Med. 2005;80(12):1094–1099.
- , , , et al. Hospital Survey on Patient Safety Culture: 2010 user comparative database report. (Prepared by Westat, Rockville, MD, under Contract No. HHSA 290200710024C). Rockville, MD: Agency for Healthcare Research and Quality; February 2010. AHRQ Publication No. 10‐0026.
- , , , , . Physiological and behavioural response patterns at work among hospital nurses. J Nurs Manag. 2011;19(1):57–68.
- , . The treatment of missing values and its effect in the classifier accuracy. In: Banks D, House L, McMorris FR, Arabie P, Gaul W, eds. Classification, Clustering and Data Mining Applications. Berlin, Germany: Springer‐Verlag; 2004.
- , . A systematic review of failures in handoff communication during intrahospital transfers. Jt Comm J Qual Patient Saf. 2011;37(6):274–284.
- , , , et al. Reduction in medication errors in hospitals due to adoption of computerized provider order entry systems. J Am Med Inform Assoc. 2013;20(3):470–476.
- , . Multilevel psychometric properties of the AHRQ hospital survey on patient safety culture. BMC Health Serv Res. 2010;10:199.
- , , , et al. Becoming a High Reliability Organization: Operational Advice for Hospital Leaders. Prepared by the Lewin Group under contract no. 290‐04‐0011. AHRQ publication no. 08–0022. Rockville, MD: Agency for Healthcare Research and Quality; 2008.
- Health policy brief: Medicare hospital readmissions reduction program. Health Affairs. November 12, 2013. Available at: http://www.healthaffairs.org/healthpolicybriefs/brief.php?brief_id=102. Accessed August 11, 2014.
- , , , , . The care span: the importance of transitional care in achieving health reform. Health Aff (Millwood). 2011;30(4):746–754.
Using electronic health records to improve the continuity of care between hospital units does not replace the need for interpersonal communication to improve transitions of care. Hospital personnel play a critical role in accurately exchanging patient information during patient transfers, a process requiring accurate communication between hospital units to prevent system failures.[1] Because poor communication contributes to preventable adverse events,[2] and effective communication during handoffs decreases medical errors and readmissions,[3] hospitals need to ensure their work environments are conducive to effective communication.
Individuals working under time constraints and heavy workloads could potentially be at high risk of misinterpreting or delivering inaccurate information,[4] partially due to limited ability to accurately process and communicate information under stressful circumstances. Furthermore, because time‐constrained decision makers tend to use less information and less rigorous decision strategies,[5] work climates characterized by staff members doing too many things too quickly could cause patient health information to be lost during transitions of care across hospital units.
Current studies illustrate scenarios in which demanding or time‐constrained work environments caused information exchange errors. One study found that the increased rate of prescribing errors was partially attributed to a high‐demand work environment characterized by working after hours and multitasking.[6] Other studies found that clinicians' limited time to relay and respond to information and ask clarifying questions during patient handoffs was partially attributed to the fast‐faced and chaotic environment of the emergency room.[7, 8] These studies are consistent with another study that found patient handoffs between emergency departments and inpatient wards were inadequate, partially due to less interactive and more rushed communication.[9] The fact that communication breakdowns are widely cited as barriers to patient handoffs[7, 8, 10] and facilitators of medical errors,[7, 8] further underscores the detrimental effect that crisis mode work climates could have on exchanging patient information during transitions of care.
The objective of this analysis was to evaluate the extent to which a crisis mode work climate impacts the occurrence of patient information exchange problems. Estimating associations between hospital staff members' perceptions of crisis mode work climates and perceptions of information exchange problems provide insights as to whether high‐demand and time‐constrained work climates negatively impact the exchange of patient information. Because hospital staff members working under time constraints and heavy workloads could potentially be at high risk of misinterpreting or delivering inaccurate information, we hypothesized that higher levels of a perceived crisis mode work climate would be associated with higher levels of perceived problems with information exchange across hospital units.
METHODS
Data Source
Data originated from the Agency of Healthcare Research and Quality 2010 Hospital Survey on Patient Safety Culture. This validated survey, designed to assess the safety climate within acute‐care settings, remains an important annual survey deployed each year to track changes and factors impacting patient safety.[11] We included only those respondents who self‐reported their position as a nurse, physician, pharmacist, dietician, therapist, technician, patient care assistant, or hospital unit secretary, all of whom are likely responsible for exchanging patient information across hospital units. For this reason, we excluded respondents who self‐reported their position as administrative or miscellaneous. Applying these exclusion criteria resulted in 247,104 respondents across 884 hospitals.
Conceptual Framework
The relationship between perceived crisis mode work climates and patient information exchange problems is likely influenced by staff skill levels, work climate, and infrastructure (Figure 1). With respect to skill levels, hospital staff members with many years of experience compared to those with fewer years may be relatively desensitized to chaotic work environments and consequently have higher thresholds for perceiving crisis modes. Number of hours worked per week likely impacts perceived crisis mode as illustrated in 1 study finding that full‐time nurses reported a significantly lower work pace compared to part‐time nurses.[12] Years of experience likely impacts perception of information exchange problems, particularly if staff members with many years of experience are familiar enough with hospital systems or protocols to easily detect exchange errors or mishaps.

With respect to work climates, workers' perception of cooperation, coordination and patient safety, and specific hospital unit likely impact perceptions of crisis work mode and information exchange problems. For example, hospital staff members reporting high levels of cooperation, coordination, and patient safety likely perceive fewer crisis modes and information exchange problems compared to those in less‐cooperative hospital units. Furthermore, the heterogeneity of work cultures across departments within a hospital results in department‐specific perceptions of crisis mode climates and information exchange problems. Infrastructure factors, such as hospital size, teaching and ownership status, and census region, likely impact the amount of resources available for staffing and infrastructure, which in turn could impact work pace and information exchange accuracy.
Variable Definitions
We defined our predictor as the perceived presence of a crisis mode work climate as captured from the survey questionnaire item: We work in crisis mode trying to do too much, too quickly. This question item had a Likert response scale comprised of the following 5 answer choices: (1) strongly disagree, (2) disagree, (3) neutral, (4) agree, (5) strongly agree. We created a 3‐level response variable by aggregating the agree and disagree responses, respectively, as the first 2 levels, and retaining the neutral response as the third level. Consequently, those responding strongly disagree or disagree were classified as working in lowcrisis mode work climates and those responding strongly agree or agree were classified as working in high crisis mode work climates. We defined our outcome measure as the presence of patient information exchange problems as captured from the survey questionnaire item: Problems often occur in the exchange of information across hospital units. Because this question item had a Likert response scale similar to the crisis mode question predictor, we also created a 3‐level categorical variable in the same fashion. Consequently, those responding strongly disagree or disagree were classified as perceiving no problems exchanging patient information, and those responding strongly agree or agree were classified as perceiving problems exchanging patient information. For the fewer than 10% of the respondents with missing data on either the predictor our outcome variables, the mode measure of central tendency was imputed, a methodology validated in a previous study.[13]
We also included questionnaire items that captured staff skill levels, work climate, and infrastructure as covariates to account for potential confounders (Figure 1). The staff skill levels domain included years of experience working in the hospital, specialty, and unit; current staff position; and extent of patient contact. The work climate domain included respondent perceptions of coordination and cooperation, patient safety, and primary work area or unit in which the provider reported working. The hospital infrastructure domain included bed size, census region, teaching status, and government ownership status. For the fewer than 10% of the respondents with missing data on any of the categorical variables, the mode measure of central tendency was imputed, a methodology validated in a previous study.[13]
Analytic Approach
We used multivariable ordinal regressions to estimate the likelihood of perceived problems in patient information exchange conditional upon perceptions of a crisis mode work climate, controlling for staff skill levels, work climate, and hospital infrastructure. Our estimates therefore reflect the likelihood of hospital staff responding strongly agree or agree to the question Problems often occur in the exchange of information across hospital units conditional upon responding strongly agree or agree to the question We work in crisis mode trying to do too much, too quickly. In addition to controlling for hospital‐specific response rates, we also adjusted our standard errors to account for the clustering of respondents within hospitals. All analyses were conducted in SAS version 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
The hospital sample averaged 279 respondents per hospital with a 56% response rate. Most hospitals were located in the Central region of the United States, and 32% and 19% were teaching and government‐owned hospitals, respectively. Forty‐three percent and 44% of the hospitals in the sample were designated as small and medium hospitals, respectively (Table 1).
| Characteristics | % |
|---|---|
| |
| Hospital characteristics, N=884 | |
| Bed size | |
| Small, 199 | 43.5 |
| Medium, 100399 | 43.8 |
| Large, 400 plus | 12.7 |
| Teaching status | |
| Yes | 32.2 |
| No | 67.8 |
| Government ownership | |
| Yes | 19.5 |
| No | 80.5 |
| Census region | |
| Mid‐Atlantic and New England | 8.7 |
| South Atlantic | 14.8 |
| Central | 57.2 |
| Mountain | 7.7 |
| Pacific | 11.5 |
| Response rate, mean (SD) | 0.56 (0.28) |
| Respondents per hospital, mean (SD) | 279 (358) |
| Respondent characteristics, N=274,140 | |
| How long have you worked in your current specialty or profession? | |
| <1 year | 5.8 |
| 15 years | 32.8 |
| 610 years | 16.2 |
| 1115 years | 12.0 |
| 1620 years | 10.6 |
| 21 years | 22.7 |
| How long have you worked in this hospital? | |
| <1 year | 9.8 |
| 15 years | 42.8 |
| 610 years | 17.8 |
| 1115 years | 9.0 |
| 1620 years | 8.2 |
| 21 years | 12.4 |
| How long have you worked in your current hospital work area/unit? | |
| <1 year | 13.1 |
| 15 years | 48.0 |
| 610 years | 18.1 |
| 1115 years | 8.1 |
| 1620 years | 6.0 |
| 21 years | 6.7 |
| Typically, how many hours per week do you work in this hospital? | |
| <20 hours | 4.8 |
| 2039 hours | 39.9 |
| 4059 hours | 48.8 |
| 6079 hours | 4.2 |
| 8099 hours | 2.1 |
| 100 hours | 0.11 |
| What is your staff position in this hospital? | |
| Registered nurse | 51.2 |
| Technician (eg, ECG, lab, radiology) | 14.1 |
| Unit assistant/clerk/secretary | 8.5 |
| Patient care assistant/hospital aide/care partner | 7.4 |
| Physical, occupational, or speech therapist | 3.7 |
| Attending/staff physician | 3.5 |
| LVN/LPN | 3.0 |
| Respiratory therapist | 2.9 |
| Pharmacist | 2.2 |
| Physician assistant/nurse practitioner | 1.4 |
| Resident physician/physician in training | 1.2 |
| Dietician | 0.83 |
| In your staff position, do you typically have direct interaction or contact with patients? | |
| Yes | 86.6 |
| No | 13.4 |
| What is your primary work area or unit in this hospital? | |
| Other | 27.7 |
| Medicine (nonsurgical) | 11.1 |
| Surgery | 10.0 |
| Intensive care unit (any type) | 8.6 |
| Many different hospital units/no specific unit | 6.8 |
| Radiology | 6.2 |
| Emergency department | 5.8 |
| Obstetrics | 4.9 |
| Laboratory | 4.9 |
| Rehabilitation | 4.2 |
| Pediatrics | 3.8 |
| Pharmacy | 3.2 |
| Psychiatry/mental health | 2.1 |
| Anesthesiology | 0.55 |
Thirty‐seven percent of the respondents have worked in their current specialty or profession for 5 years or less (Table 1). Over half of the respondents have worked in their current hospital for 5 years or less, whereas 61% have worked in their current unit within the hospital for 5 years or less. Forty‐nine percent work at least 40 hours per week. Registered nurses and technicians represented the 2 largest subgroups of staff positions, comprising 51% and 14% of the sample, respectively. Dieticians and resident physicians, on the other hand, represented the 2 smallest subgroups of staff positions, comprising 0.83% and 1.2% of the sample, respectively. Eighty‐seven percent of the respondents have direct interaction or contact with patients. Apart from those responding other as their hospital unit, nonsurgical medicine and surgery represented the largest subgroup primary work areas, comprising 11% and 10% of the sample, respectively. In contrast, psychiatry and anesthesiology represented the 2 smallest subgroups of primary work areas, comprising 2.1% and 0.55% of the sample, respectively (Table 1).
Respondents scored relatively high with regard to teamwork and helping each other out under hurried or busy circumstances. For example, 85% agreed or strongly agreed that their unit worked together as a team to get work done when a lot of work needed to be completed quickly, and 68% agreed or strongly agreed that individuals within their unit helped out when an area in their unit became busy (Table 1). Despite this cooperation, 31% agreed or strongly agreed that hospital units did not coordinate well together. Paradoxically, 57% agreed or strongly agreed that there was good cooperation among hospital units that needed to work together. Seventy‐five percent of the respondents reported excellent or very good patient safety levels within their unit, although 53% agreed or strongly agreed that staff worked longer hours than was best for patient care (Table 1).
With regard to perceived crisis mode work climate, 32% and 47% reported agreeing and disagreeing, respectively, that their work unit worked in crisis mode trying to do too much too quickly (Table 2). With regard to perceived problems with patient information exchange, 27% and 36% reported agreeing and disagreeing, respectively, that information exchange problems occurred across hospital units (Table 2).
| Perceptions | % |
|---|---|
| We work in crisis mode trying to do too much, too quickly | |
| Strongly disagree | 8.1 |
| Disagree | 39.2 |
| Neutral | 21.0 |
| Agree | 24.3 |
| Strongly agree | 7.5 |
| Problems often occur in the exchange of information across hospital units | |
| Strongly disagree | 4.6 |
| Disagree | 31.3 |
| Neutral | 37.3 |
| Agree | 24.0 |
| Strongly agree | 2.7 |
| When a lot of work needs to be done quickly, we work together as a team to get the work done. | |
| Strongly disagree | 1.5 |
| Disagree | 6.1 |
| Neutral | 7.5 |
| Agree | 53.6 |
| Strongly agree | 31.2 |
| When one area in this unit gets really busy, others help out. | |
| Strongly disagree | 3.9 |
| Disagree | 13.9 |
| Neutral | 13.7 |
| Agree | 52.6 |
| Strongly agree | 15.8 |
| Hospital units do not coordinate well with each other. | |
| Strongly disagree | 5.6 |
| Disagree | 38.8 |
| Neutral | 23.7 |
| Agree | 25.3 |
| Strongly agree | 6.6 |
| There is good cooperation among hospital units that need to work together. | |
| Strongly disagree | 2.7 |
| Disagree | 15.1 |
| Neutral | 24.7 |
| Agree | 51.1 |
| Strongly agree | 6.3 |
| Please give your work area/unit in this hospital an overall grade on patient safety. | |
| Excellent | 23.0 |
| Very good | 49.8 |
| Acceptable | 21.8 |
| Poor | 4.6 |
| Failing | 0.76 |
| Staff in this unit work longer hours than is best for patient care. | |
| Strongly disagree | 11.5 |
| Disagree | 42.2 |
| Neutral | 23.6 |
| Agree | 18.4 |
| Strongly agree | 6.3 |
In the unadjusted analyses, crisis mode perceptions and information exchange problem perceptions were significantly associated. Among those who agreed that their work unit worked in crisis mode, a larger proportion of respondents agreed (41%) versus disagreed (24%) that problems often occurred in exchanging patient information across units (Table 3). In contrast, among those who disagreed that their work unit worked in crisis mode, a larger proportion of respondents disagreed (47%) versus agreed (19%) that problems often occurred in exchanging patient information across units (Table 3).
| Problems Often Occur in Exchange of Information Across Hospital Units | |||
|---|---|---|---|
| Agree (N=66,115), Row %* | Neutral (N=92,228), Row % | Disagree (N=88,797), Row % | |
| |||
| Crisis Mode Work Climate | |||
| Agree (N=78,253) | 40.8 | 35.4 | 23.8 |
| Neutral (N=51,836)‖ | 22.9 | 48.9 | 28.2 |
| Disagree (N=116,781) | 19.0 | 33.5 | 47.5 |
In the multivariable ordinal regression, compared to those who disagreed that their unit worked in crisis mode, those who agreed were 1.6 times more likely to report that problems often occurred in exchanging patient information across units (odds ratio [OR]: 1.6, 95% confidence interval [CI]: 1.58‐1.65) (Table 4). Additionally, some key covariates were independently associated with perceptions of information exchange problems. Two of these covariates measured workplace coordination. Those who reported that hospital units did not cooperate well together were more likely to report problematic information exchange compared to those who reported that hospital units did cooperate well (OR: 4.7, 95% CI: 4.35.0). Relatedly, those who reported that hospital units did coordinate well were less likely to report problematic information exchange compared to those who reported that hospital units did not coordinate well (OR: 0.10, 95% CI: 0.10‐0.11). Two other covariates measured patient contact and perceptions about long working hours. Those who reported having direct interaction or contact with patients were less likely to report problematic information exchange compared to those who reported not having direct interaction or contact with patients (OR: 0.85, 95% CI: 0.83‐0.87). Those who reported that staff did not work longer hours than was better for patient care were less likely to report problematic information exchange compared to those who did report working longer hours than was better for patient care (OR: 0.76, 95% CI: 0.73 0.79). One covariate measured hospital size. Those who reported working in smaller hospitals were less likely to report problematic information exchange compared to those reporting working in large hospitals (OR: 0.66, 95% CI 0.59‐0.75) (Table 4).
| Characteristic | Unadjusted OR (95% CI) | Adjusted OR* (95% CI) |
|---|---|---|
| ||
| Primary predictor of interest | ||
| Crisis mode work climate | ||
| Agree | 3.0 (2.9‐3.1) | 1.6 (1.5‐1.6) |
| Neutral | 1.8 (1.7‐1.8) | 1.3 (1.2‐1.3) |
| Disagree | Reference | Reference |
| Hospital characteristics | ||
| Bed Size | ||
| Small, 624 | 0.51 (0.44‐0.59) | 0.66 (0.59‐0.75) |
| Small, 249 | 0.59 (0.53‐0.66) | 0.77 (0.70‐0.84) |
| Small, 5099 | 0.65 (0.58‐0.73) | 0.78 (0.71‐0.84) |
| Medium, 100199 | 0.85 (0.77‐0.95) | 0.92 (0.86‐1.0) |
| Medium, 200299 | 1.0 (0.98‐1.1) | 0.97 (0.90‐1.0) |
| Medium, 300399 | 0.96 (0.85‐1.1) | 1.0 (0.92‐1.1) |
| Large, 400499 | 0.99 (0.86‐1.1) | 0.96 (0.87‐1.0) |
| Large, 500 plus | Reference | Reference |
| Teaching status | ||
| No | 0.81 (0.76‐0.87) | 1.0 (0.95‐1.0) |
| Yes | Reference | Reference |
| Government ownership | ||
| No | 1.1 (1.01.2) | 1.0 (0.98‐1.1) |
| Yes | Reference | Reference |
| Census region | ||
| Mid‐Atlantic and New England | 1.0 (0.88‐1.1) | 0.91 (0.84‐0.99) |
| South Atlantic | 0.95 (0.85‐1.1) | 1.0 (0.95‐1.1) |
| Central 1 | 0.95 (0.85‐1.0) | 0.95 (0.89‐1.0) |
| Central 2 | 0.71 (0.62‐0.81) | 0.91 (0.83‐0.99) |
| Central 3 | 0.80 (0.71‐0.91) | 0.97 (0.90‐1.0) |
| Central 4 | 0.76 (0.68‐0.86) | 0.93 (0.85‐1.0) |
| Mountain | 0.84 (0.73‐0.96) | 0.98 (0.90‐1.1) |
| Pacific | Reference | Reference |
| Average survey response rate within hospital | 0.65 (0.58‐0.72) | 0.93 (0.82‐1.0) |
| Respondent characteristics | ||
| How long have you worked in your current specialty or profession? | ||
| <1 year | 0.75 (0.73‐0.78) | 1.03 (0.99‐1.1) |
| 15 years | 0.99 (0.97‐1.0) | 1.1 (1.1‐1.1) |
| 610 years | 1.0 (1.01.1) | 0.99 (0.96‐1.0) |
| 1115 years | 1.0 (1.01.1) | 1.0 (0.97‐1.0) |
| 1620 years | 1.0 (0.98‐1.0) | 0.97 (0.94‐1.0) |
| 21 years | Reference | Reference |
| How long have you worked in this hospital? | ||
| <1 year | 0.75 (0.73‐0.77) | 0.90 (0.85‐0.90) |
| 15 years | 1.03 (1.001.05) | 0.99 (0.95‐1.0) |
| 610 years | 1.1 (1.1‐1.1) | 0.99 (0.95‐1.0) |
| 1115 years | 1.1 (1. 01.1) | 1.0 (0.96‐1.0) |
| 1620 years | 1.1 (1.01.1) | 0.98 (0.94‐1.0) |
| 21 years | Reference | Reference |
| How long have you worked in your current hospital work area/unit? | ||
| <1 year | 0.79 (0.76‐0.82) | 0.98 (0.93‐1.0) |
| 15 years | 1.0 (1.01.1) | 1.0 (0.99‐1.1) |
| 610 years | 1.1 (1.1‐1.1) | 1.0 (1.01.1) |
| 1115 years | 1.1 (1.01.1) | 1.0 (0.99‐1.1) |
| 1620 years | 1.1 (1.01.1) | 1.1 (1.01.1) |
| 21 years | Reference | Reference |
| Typically, how many hours per week do you work in this hospital? | ||
| <20 | 0.63 (0.50‐0.79) | 0.91 (0.72‐1.2) |
| 2039 | 0.75 (0.59‐0.94) | 0.90 (0.71‐1.1) |
| 4059 | 0.87 (0.69‐1.1) | 1.1 (0.85‐1.4) |
| 6079 | 0.95 (0.75‐1.2) | 1.0 (0.82‐1.3) |
| 8099 | 0.99 (0.78‐1.2) | 1.1 (0.86‐1.4) |
| 100 | Reference | Reference |
| What is your staff position in this hospital? | ||
| Registered nurse | 0.92 (0.90‐0.94) | 1.1 (0.98‐1.0) |
| Technician (eg, ECG, lab, radiology) | Reference | Reference |
| Unit assistant/clerk/secretary | 0.79 (0.76‐0.81) | 0.94 (0.80‐0.96) |
| Patient care assistant/hospital aide/care partner | 0.78 (0.75‐0.81) | 0.96 (0.90‐0.98) |
| Physical, occupational, or speech therapist | 0.88 (0.84‐0.92) | 1.2 (1.1‐1.2) |
| Attending/staff physician | 1.0 (0.97‐1.1) | 1.3 (1.2‐1.3) |
| LVN/LPN | 0.89 (0.85‐0.94) | 1.0 (0.92‐1.0) |
| Respiratory therapist | 0.84 (0.80‐0.88) | 0.97 (0.89‐1.0) |
| Pharmacist | 1.5 (1.4‐1.6) | 1.3 (1.1‐1.3) |
| Physician assistant/nurse practitioner | 0.93 (0.87‐1.0) | 1.2 (1.1‐1.2) |
| Resident physician/physician in training | 0.96 (0.89‐1.0) | 1.3 (1.2‐1.4) |
| Dietician | 0.86 (0.79‐0.94) | 1.2 (1.1‐1.3) |
| In your staff position, do you typically have direct interaction or contact with patients? | ||
| Yes | 0.83 (0.82‐0.85) | 0.85 (0.83‐0.87) |
| No | Reference | Reference |
| What is your primary work area or unit in this hospital? | ||
| Other | Reference | Reference |
| Medicine (nonsurgical) | 1.1 (1.01.1) | 0.84 (0.82‐0.89) |
| Surgery | 1.1 (1.1‐1.2) | 0.88 (0.86‐0.91) |
| Intensive care unit (any type) | 0.93 (0.90‐0.96) | 0.78 (0.76‐0.81) |
| Many different hospital units/no specific unit | 1.2 (1.1‐1.2) | 1.0 (0.98‐ 1.0) |
| Radiology | 1.1 (1.1‐1.1) | 1.0 (1.01.1) |
| Emergency department | 1.0 (0.97‐1.0) | 0.57 (0.55‐0.60) |
| Obstetrics | 0.76 (0.73‐0.79) | 0.66 (0.63‐0.69) |
| Laboratory | 1.2 (1.2‐1.3) | 1.0 (1.01.1) |
| Rehabilitation | 1.0 (0.97‐1.0) | 1.0 (0.98‐1.1) |
| Pediatrics | 0.90 (0.86‐0.94) | 0.83 (0.80‐0.87) |
| Pharmacy | 1.6 (1.5‐1.7) | 1.1 (1.01.2) |
| Psychiatry/mental health | 1.2 (1.1‐1.2) | 0.96 (0.90‐1.0) |
| Anesthesiology | 1.1 (1.01.3) | 0.93 (0.83‐1.0) |
| Respondent perceptions | ||
| When a lot of work needs to be done quickly, we work together as a team to get the work done. | ||
| Strongly disagree | 3.2 (3.03.4) | 1.0 (0.98‐1.1) |
| Disagree | 3.2 (3.13.3) | 1.0 (1.01.1) |
| Neutral | 2.3 (2.2‐2.4) | 0.98 (0.94‐1.0) |
| Agree | 2.3 (2.2‐2.4) | 1.0 (1.002‐1.04) |
| Strongly agree | Reference | Reference |
| Staff in this unit work longer hours than is best for patient care. | ||
| Strongly disagree | 0.51 (0.48‐0.53) | 0.76 (0.73‐0.79) |
| Disagree | 0.68 (0.67‐0.70) | 0.81 (0.78‐0.84) |
| Neutral | 0.94 (0.91‐0.97) | 0.93 (0.90‐0.97) |
| Agree | 1.0 (0.99‐1.1) | 0.94 (0.91‐0.98) |
| Strongly agree | Reference | Reference |
| When 1 area in this unit gets really busy, others help out. | ||
| Strongly disagree | 3.8 (3.7 ‐ 4.0) | 1.0 (0.96‐1.1) |
| Disagree | 3.0 (2.9‐3.1) | 1.0 (0.99‐1.1) |
| Neutral | 2.2 (2.12.3) | 1.0 (0.97‐1.0) |
| Agree | 1.5 (1.5‐1.6) | 0.99 (0.96‐1.0) |
| Strongly agree | Reference | Reference |
| Hospital units do not coordinate well with each other. | ||
| Strongly disagree | 0.03 (0.03‐0.04) | 0.10 (0.10‐0.11) |
| Disagree | 0.08 (0.08‐0.08) | 0.18 (0.17‐0.19) |
| Neutral | 0.21 (0.20‐0.22) | 0.32 (0.30‐0.33) |
| Agree | 0.50 (0.48‐0.52) | 0.61 (0.58‐0.63) |
| Strongly agree | Reference | Reference |
| There is good cooperation among hospital units that need to work together. | ||
| Strongly disagree | 20.1 (18.921.5) | 4.7 (4.35.0) |
| Disagree | 14.2 (13.614.9) | 4.2 (4.14.5) |
| Neutral | 6.7 (6.47.0) | 2.7 (2.6‐2.8) |
| Agree | 2.4 (2.3‐2.5) | 1.6 (1.6‐1.7) |
| Strongly agree | Reference | Reference |
| Please give your work area/unit in this hospital an overall grade on patient safety | ||
| Excellent | 0.13 (0.12‐0.14) | 0.47 (0.42‐0.52) |
| Very good | 0.24 (0.21‐0.26) | 0.63 (0.57‐0.70) |
| Acceptable | 0.49 (0.45‐0.54) | 0.79 (0.72‐0.88) |
| Poor | 0.83 (0.75‐0.92) | 0.92 (0.83‐1.03) |
| Failing | Reference | Reference |
DISCUSSION
Our results illustrate that when hospital staff agree that their hospital works in crisis mode, they are more likely to agree that their hospital unit had frequent problems exchanging patient information across units. Because hospital staff working under time constraints and heavy workloads could potentially be at risk of misinterpreting or delivering inaccurate information, these results imply that crisis mode work climates increase the risk of problematic health information exchange. An equally plausible interpretation could be that problematic patient health information exchange increases the risk of hospital staff perceiving crisis mode work climates. Given that information gaps are associated with patient handoff errors,[14] and that patient handoff errors are associated with adverse events,[2, 3, 6, 8] an urgent need exists to implement information exchange systems that prevent information gaps from harming patients. Consequently, hospitals need to implement workflow strategies that prevent information gaps from undermining patient safety during transitions of care.
Other factors affect information exchange apart from crisis mode work climate, as illustrated by the significant associations of key covariates in the multivariate model. The effect found between perceived coordination and information exchange implies that improving information exchange requires good cooperation and coordination. The effect found between patient contact and information exchange implies that working directly with patients improves either the accuracy or the perception of information exchange. Finally, the effect found between hospital size and information exchange suggests that small hospitals are less likely than large hospitals to have information exchange problem. The geographical dispersion and the complexity of larger institutions could result in information exchange problems due to more confusion and less in‐person communication.
Because problematic patient information exchanges are associated with hospital size, coordination, and patient contact, in addition to crisis mode work climate, multifaceted solutions are necessary to resolve the problem. For example, hospital interventions designed to improve coordination could in turn attenuate perceived crisis modes. Furthermore, tailoring these interventions to hospitals that belong to complex geographically dispersed provider networks would likely decrease errors during transitions of care. Because multiple factors cause information exchange problems, implementing interventions that improve both coordination and crisis mode work climates would likely result in a greater net improvement compared to interventions focused solely on decreasing crisis mode work climates.
Some limitations of our paper are worth noting. First, we did not have information on the volume of data exchanged or the functionality levels of the electronic health record systems, both of which likely impact the accuracy of patient information exchange. For example, hospitals with smaller versus larger amounts of data exchanged could be less prone to error. On the other hand, this risk of error could be reduced even further by implementing robust health information technology (IT) systems that improve the accuracy of information transfer. This is consistent with studies showing that hospitals without computerized provider order entry (CPOE) systems have been shown to have higher medication error rates compared to those hospitals with CPOE systems.[15] Therefore, omitting data volume and health IT capabilities from the multivariate model could introduce unobserved heterogeneity, resulting in biased associations between perceived crisis mode work climate and perceived information exchange problems. Second, the cross‐sectional design limits our ability to infer causality because we are not certain whether the perceived crisis mode occurred before, after, or simultaneously to perceived information exchange problems. Third, the self‐reported nature of the questionnaire items does not provide information on observed levels of crisis mode and exchange problems, which could be inconsistent with perceived levels. Fourth, the relatively low within‐hospital response rate decreases the external validity of our findings. For example, if responders' perceptions of crisis mode or information exchange problems significantly differed from nonresponders, our results would not be generalizable to the larger population of acute‐care hospitals across the United States. Therefore, conclusions should be viewed with caution if applying these results to hospitals with respondents significantly differing from those contained within our sample.
Despite these limitations, the large sample size in conjunction with the use of data from a survey having acceptable psychometric properties[16] strengthens the external and internal validity of our findings. Although questionnaire items measuring perceptions are relatively subjective in nature compared to using metrics that capture observed problems or crisis modes, we argue that staff perception data are equally informative, as they guide organization leaders on how to improve workplace performance. Because a core concept of high reliability organizations (HROs) is to preserve constant awareness by key leaders and staff of the state of the systems and processes that affect patient care,[17] HROs could benefit from knowing the extent to which staff perceptions impact patient care. From a methods perspective, the multivariable ordinal regressions enabled us to control for potential confounders that if omitted could have resulted in biased estimates. Furthermore, low levels of multicollinearity as illustrated by low variation inflation factors enabled us to isolate the independent effect of crisis mode perceptions. Including hospital size and hospital work unit as covariates was an additional methodological strength helping account for the unobserved heterogeneity caused by excluding volume of data exchanged or health IT system capability. For example, because larger compared to smaller hospitals usually have more sophisticated health IT systems,[15] including bed size in the model theoretically captures some of the variation that would have been captured if we were able to include a covariate measuring health IT capability. Last, using ordinal regression facilitates interpretation of the findings because the questionnaire items for the predictor and outcome were originally captured on a Likert scale.
Our findings underscore the significant impact that work climate has on accurate information exchange, and ultimately patient safety. Improving patient safety is imperative for hospitals, especially within the context of recent regulations stemming from the Affordable Care Act that incentivize hospitals to reduce readmissions[18] and improve transitions of care.[19] Because accurate health information exchange is a critical component of patient care, resolving barriers that decrease the accuracy of this exchange is essential. Therefore, future studies need to continue examining these associations within the context of study designs that incorporate longitudinal data and datasets that include objective measures capturing crisis mode work climates and information exchange problems. Because effective communication during handoffs is associated with decreases in medical errors and readmissions, hospitals need to continually ensure that work environments are conducive to effective patient information exchange.
Disclosures
Nothing to report
Using electronic health records to improve the continuity of care between hospital units does not replace the need for interpersonal communication to improve transitions of care. Hospital personnel play a critical role in accurately exchanging patient information during patient transfers, a process requiring accurate communication between hospital units to prevent system failures.[1] Because poor communication contributes to preventable adverse events,[2] and effective communication during handoffs decreases medical errors and readmissions,[3] hospitals need to ensure their work environments are conducive to effective communication.
Individuals working under time constraints and heavy workloads could potentially be at high risk of misinterpreting or delivering inaccurate information,[4] partially due to limited ability to accurately process and communicate information under stressful circumstances. Furthermore, because time‐constrained decision makers tend to use less information and less rigorous decision strategies,[5] work climates characterized by staff members doing too many things too quickly could cause patient health information to be lost during transitions of care across hospital units.
Current studies illustrate scenarios in which demanding or time‐constrained work environments caused information exchange errors. One study found that the increased rate of prescribing errors was partially attributed to a high‐demand work environment characterized by working after hours and multitasking.[6] Other studies found that clinicians' limited time to relay and respond to information and ask clarifying questions during patient handoffs was partially attributed to the fast‐faced and chaotic environment of the emergency room.[7, 8] These studies are consistent with another study that found patient handoffs between emergency departments and inpatient wards were inadequate, partially due to less interactive and more rushed communication.[9] The fact that communication breakdowns are widely cited as barriers to patient handoffs[7, 8, 10] and facilitators of medical errors,[7, 8] further underscores the detrimental effect that crisis mode work climates could have on exchanging patient information during transitions of care.
The objective of this analysis was to evaluate the extent to which a crisis mode work climate impacts the occurrence of patient information exchange problems. Estimating associations between hospital staff members' perceptions of crisis mode work climates and perceptions of information exchange problems provide insights as to whether high‐demand and time‐constrained work climates negatively impact the exchange of patient information. Because hospital staff members working under time constraints and heavy workloads could potentially be at high risk of misinterpreting or delivering inaccurate information, we hypothesized that higher levels of a perceived crisis mode work climate would be associated with higher levels of perceived problems with information exchange across hospital units.
METHODS
Data Source
Data originated from the Agency of Healthcare Research and Quality 2010 Hospital Survey on Patient Safety Culture. This validated survey, designed to assess the safety climate within acute‐care settings, remains an important annual survey deployed each year to track changes and factors impacting patient safety.[11] We included only those respondents who self‐reported their position as a nurse, physician, pharmacist, dietician, therapist, technician, patient care assistant, or hospital unit secretary, all of whom are likely responsible for exchanging patient information across hospital units. For this reason, we excluded respondents who self‐reported their position as administrative or miscellaneous. Applying these exclusion criteria resulted in 247,104 respondents across 884 hospitals.
Conceptual Framework
The relationship between perceived crisis mode work climates and patient information exchange problems is likely influenced by staff skill levels, work climate, and infrastructure (Figure 1). With respect to skill levels, hospital staff members with many years of experience compared to those with fewer years may be relatively desensitized to chaotic work environments and consequently have higher thresholds for perceiving crisis modes. Number of hours worked per week likely impacts perceived crisis mode as illustrated in 1 study finding that full‐time nurses reported a significantly lower work pace compared to part‐time nurses.[12] Years of experience likely impacts perception of information exchange problems, particularly if staff members with many years of experience are familiar enough with hospital systems or protocols to easily detect exchange errors or mishaps.

With respect to work climates, workers' perception of cooperation, coordination and patient safety, and specific hospital unit likely impact perceptions of crisis work mode and information exchange problems. For example, hospital staff members reporting high levels of cooperation, coordination, and patient safety likely perceive fewer crisis modes and information exchange problems compared to those in less‐cooperative hospital units. Furthermore, the heterogeneity of work cultures across departments within a hospital results in department‐specific perceptions of crisis mode climates and information exchange problems. Infrastructure factors, such as hospital size, teaching and ownership status, and census region, likely impact the amount of resources available for staffing and infrastructure, which in turn could impact work pace and information exchange accuracy.
Variable Definitions
We defined our predictor as the perceived presence of a crisis mode work climate as captured from the survey questionnaire item: We work in crisis mode trying to do too much, too quickly. This question item had a Likert response scale comprised of the following 5 answer choices: (1) strongly disagree, (2) disagree, (3) neutral, (4) agree, (5) strongly agree. We created a 3‐level response variable by aggregating the agree and disagree responses, respectively, as the first 2 levels, and retaining the neutral response as the third level. Consequently, those responding strongly disagree or disagree were classified as working in lowcrisis mode work climates and those responding strongly agree or agree were classified as working in high crisis mode work climates. We defined our outcome measure as the presence of patient information exchange problems as captured from the survey questionnaire item: Problems often occur in the exchange of information across hospital units. Because this question item had a Likert response scale similar to the crisis mode question predictor, we also created a 3‐level categorical variable in the same fashion. Consequently, those responding strongly disagree or disagree were classified as perceiving no problems exchanging patient information, and those responding strongly agree or agree were classified as perceiving problems exchanging patient information. For the fewer than 10% of the respondents with missing data on either the predictor our outcome variables, the mode measure of central tendency was imputed, a methodology validated in a previous study.[13]
We also included questionnaire items that captured staff skill levels, work climate, and infrastructure as covariates to account for potential confounders (Figure 1). The staff skill levels domain included years of experience working in the hospital, specialty, and unit; current staff position; and extent of patient contact. The work climate domain included respondent perceptions of coordination and cooperation, patient safety, and primary work area or unit in which the provider reported working. The hospital infrastructure domain included bed size, census region, teaching status, and government ownership status. For the fewer than 10% of the respondents with missing data on any of the categorical variables, the mode measure of central tendency was imputed, a methodology validated in a previous study.[13]
Analytic Approach
We used multivariable ordinal regressions to estimate the likelihood of perceived problems in patient information exchange conditional upon perceptions of a crisis mode work climate, controlling for staff skill levels, work climate, and hospital infrastructure. Our estimates therefore reflect the likelihood of hospital staff responding strongly agree or agree to the question Problems often occur in the exchange of information across hospital units conditional upon responding strongly agree or agree to the question We work in crisis mode trying to do too much, too quickly. In addition to controlling for hospital‐specific response rates, we also adjusted our standard errors to account for the clustering of respondents within hospitals. All analyses were conducted in SAS version 9.2 (SAS Institute Inc., Cary, NC).
RESULTS
The hospital sample averaged 279 respondents per hospital with a 56% response rate. Most hospitals were located in the Central region of the United States, and 32% and 19% were teaching and government‐owned hospitals, respectively. Forty‐three percent and 44% of the hospitals in the sample were designated as small and medium hospitals, respectively (Table 1).
| Characteristics | % |
|---|---|
| |
| Hospital characteristics, N=884 | |
| Bed size | |
| Small, 199 | 43.5 |
| Medium, 100399 | 43.8 |
| Large, 400 plus | 12.7 |
| Teaching status | |
| Yes | 32.2 |
| No | 67.8 |
| Government ownership | |
| Yes | 19.5 |
| No | 80.5 |
| Census region | |
| Mid‐Atlantic and New England | 8.7 |
| South Atlantic | 14.8 |
| Central | 57.2 |
| Mountain | 7.7 |
| Pacific | 11.5 |
| Response rate, mean (SD) | 0.56 (0.28) |
| Respondents per hospital, mean (SD) | 279 (358) |
| Respondent characteristics, N=274,140 | |
| How long have you worked in your current specialty or profession? | |
| <1 year | 5.8 |
| 15 years | 32.8 |
| 610 years | 16.2 |
| 1115 years | 12.0 |
| 1620 years | 10.6 |
| 21 years | 22.7 |
| How long have you worked in this hospital? | |
| <1 year | 9.8 |
| 15 years | 42.8 |
| 610 years | 17.8 |
| 1115 years | 9.0 |
| 1620 years | 8.2 |
| 21 years | 12.4 |
| How long have you worked in your current hospital work area/unit? | |
| <1 year | 13.1 |
| 15 years | 48.0 |
| 610 years | 18.1 |
| 1115 years | 8.1 |
| 1620 years | 6.0 |
| 21 years | 6.7 |
| Typically, how many hours per week do you work in this hospital? | |
| <20 hours | 4.8 |
| 2039 hours | 39.9 |
| 4059 hours | 48.8 |
| 6079 hours | 4.2 |
| 8099 hours | 2.1 |
| 100 hours | 0.11 |
| What is your staff position in this hospital? | |
| Registered nurse | 51.2 |
| Technician (eg, ECG, lab, radiology) | 14.1 |
| Unit assistant/clerk/secretary | 8.5 |
| Patient care assistant/hospital aide/care partner | 7.4 |
| Physical, occupational, or speech therapist | 3.7 |
| Attending/staff physician | 3.5 |
| LVN/LPN | 3.0 |
| Respiratory therapist | 2.9 |
| Pharmacist | 2.2 |
| Physician assistant/nurse practitioner | 1.4 |
| Resident physician/physician in training | 1.2 |
| Dietician | 0.83 |
| In your staff position, do you typically have direct interaction or contact with patients? | |
| Yes | 86.6 |
| No | 13.4 |
| What is your primary work area or unit in this hospital? | |
| Other | 27.7 |
| Medicine (nonsurgical) | 11.1 |
| Surgery | 10.0 |
| Intensive care unit (any type) | 8.6 |
| Many different hospital units/no specific unit | 6.8 |
| Radiology | 6.2 |
| Emergency department | 5.8 |
| Obstetrics | 4.9 |
| Laboratory | 4.9 |
| Rehabilitation | 4.2 |
| Pediatrics | 3.8 |
| Pharmacy | 3.2 |
| Psychiatry/mental health | 2.1 |
| Anesthesiology | 0.55 |
Thirty‐seven percent of the respondents have worked in their current specialty or profession for 5 years or less (Table 1). Over half of the respondents have worked in their current hospital for 5 years or less, whereas 61% have worked in their current unit within the hospital for 5 years or less. Forty‐nine percent work at least 40 hours per week. Registered nurses and technicians represented the 2 largest subgroups of staff positions, comprising 51% and 14% of the sample, respectively. Dieticians and resident physicians, on the other hand, represented the 2 smallest subgroups of staff positions, comprising 0.83% and 1.2% of the sample, respectively. Eighty‐seven percent of the respondents have direct interaction or contact with patients. Apart from those responding other as their hospital unit, nonsurgical medicine and surgery represented the largest subgroup primary work areas, comprising 11% and 10% of the sample, respectively. In contrast, psychiatry and anesthesiology represented the 2 smallest subgroups of primary work areas, comprising 2.1% and 0.55% of the sample, respectively (Table 1).
Respondents scored relatively high with regard to teamwork and helping each other out under hurried or busy circumstances. For example, 85% agreed or strongly agreed that their unit worked together as a team to get work done when a lot of work needed to be completed quickly, and 68% agreed or strongly agreed that individuals within their unit helped out when an area in their unit became busy (Table 1). Despite this cooperation, 31% agreed or strongly agreed that hospital units did not coordinate well together. Paradoxically, 57% agreed or strongly agreed that there was good cooperation among hospital units that needed to work together. Seventy‐five percent of the respondents reported excellent or very good patient safety levels within their unit, although 53% agreed or strongly agreed that staff worked longer hours than was best for patient care (Table 1).
With regard to perceived crisis mode work climate, 32% and 47% reported agreeing and disagreeing, respectively, that their work unit worked in crisis mode trying to do too much too quickly (Table 2). With regard to perceived problems with patient information exchange, 27% and 36% reported agreeing and disagreeing, respectively, that information exchange problems occurred across hospital units (Table 2).
| Perceptions | % |
|---|---|
| We work in crisis mode trying to do too much, too quickly | |
| Strongly disagree | 8.1 |
| Disagree | 39.2 |
| Neutral | 21.0 |
| Agree | 24.3 |
| Strongly agree | 7.5 |
| Problems often occur in the exchange of information across hospital units | |
| Strongly disagree | 4.6 |
| Disagree | 31.3 |
| Neutral | 37.3 |
| Agree | 24.0 |
| Strongly agree | 2.7 |
| When a lot of work needs to be done quickly, we work together as a team to get the work done. | |
| Strongly disagree | 1.5 |
| Disagree | 6.1 |
| Neutral | 7.5 |
| Agree | 53.6 |
| Strongly agree | 31.2 |
| When one area in this unit gets really busy, others help out. | |
| Strongly disagree | 3.9 |
| Disagree | 13.9 |
| Neutral | 13.7 |
| Agree | 52.6 |
| Strongly agree | 15.8 |
| Hospital units do not coordinate well with each other. | |
| Strongly disagree | 5.6 |
| Disagree | 38.8 |
| Neutral | 23.7 |
| Agree | 25.3 |
| Strongly agree | 6.6 |
| There is good cooperation among hospital units that need to work together. | |
| Strongly disagree | 2.7 |
| Disagree | 15.1 |
| Neutral | 24.7 |
| Agree | 51.1 |
| Strongly agree | 6.3 |
| Please give your work area/unit in this hospital an overall grade on patient safety. | |
| Excellent | 23.0 |
| Very good | 49.8 |
| Acceptable | 21.8 |
| Poor | 4.6 |
| Failing | 0.76 |
| Staff in this unit work longer hours than is best for patient care. | |
| Strongly disagree | 11.5 |
| Disagree | 42.2 |
| Neutral | 23.6 |
| Agree | 18.4 |
| Strongly agree | 6.3 |
In the unadjusted analyses, crisis mode perceptions and information exchange problem perceptions were significantly associated. Among those who agreed that their work unit worked in crisis mode, a larger proportion of respondents agreed (41%) versus disagreed (24%) that problems often occurred in exchanging patient information across units (Table 3). In contrast, among those who disagreed that their work unit worked in crisis mode, a larger proportion of respondents disagreed (47%) versus agreed (19%) that problems often occurred in exchanging patient information across units (Table 3).
| Problems Often Occur in Exchange of Information Across Hospital Units | |||
|---|---|---|---|
| Agree (N=66,115), Row %* | Neutral (N=92,228), Row % | Disagree (N=88,797), Row % | |
| |||
| Crisis Mode Work Climate | |||
| Agree (N=78,253) | 40.8 | 35.4 | 23.8 |
| Neutral (N=51,836)‖ | 22.9 | 48.9 | 28.2 |
| Disagree (N=116,781) | 19.0 | 33.5 | 47.5 |
In the multivariable ordinal regression, compared to those who disagreed that their unit worked in crisis mode, those who agreed were 1.6 times more likely to report that problems often occurred in exchanging patient information across units (odds ratio [OR]: 1.6, 95% confidence interval [CI]: 1.58‐1.65) (Table 4). Additionally, some key covariates were independently associated with perceptions of information exchange problems. Two of these covariates measured workplace coordination. Those who reported that hospital units did not cooperate well together were more likely to report problematic information exchange compared to those who reported that hospital units did cooperate well (OR: 4.7, 95% CI: 4.35.0). Relatedly, those who reported that hospital units did coordinate well were less likely to report problematic information exchange compared to those who reported that hospital units did not coordinate well (OR: 0.10, 95% CI: 0.10‐0.11). Two other covariates measured patient contact and perceptions about long working hours. Those who reported having direct interaction or contact with patients were less likely to report problematic information exchange compared to those who reported not having direct interaction or contact with patients (OR: 0.85, 95% CI: 0.83‐0.87). Those who reported that staff did not work longer hours than was better for patient care were less likely to report problematic information exchange compared to those who did report working longer hours than was better for patient care (OR: 0.76, 95% CI: 0.73 0.79). One covariate measured hospital size. Those who reported working in smaller hospitals were less likely to report problematic information exchange compared to those reporting working in large hospitals (OR: 0.66, 95% CI 0.59‐0.75) (Table 4).
| Characteristic | Unadjusted OR (95% CI) | Adjusted OR* (95% CI) |
|---|---|---|
| ||
| Primary predictor of interest | ||
| Crisis mode work climate | ||
| Agree | 3.0 (2.9‐3.1) | 1.6 (1.5‐1.6) |
| Neutral | 1.8 (1.7‐1.8) | 1.3 (1.2‐1.3) |
| Disagree | Reference | Reference |
| Hospital characteristics | ||
| Bed Size | ||
| Small, 624 | 0.51 (0.44‐0.59) | 0.66 (0.59‐0.75) |
| Small, 249 | 0.59 (0.53‐0.66) | 0.77 (0.70‐0.84) |
| Small, 5099 | 0.65 (0.58‐0.73) | 0.78 (0.71‐0.84) |
| Medium, 100199 | 0.85 (0.77‐0.95) | 0.92 (0.86‐1.0) |
| Medium, 200299 | 1.0 (0.98‐1.1) | 0.97 (0.90‐1.0) |
| Medium, 300399 | 0.96 (0.85‐1.1) | 1.0 (0.92‐1.1) |
| Large, 400499 | 0.99 (0.86‐1.1) | 0.96 (0.87‐1.0) |
| Large, 500 plus | Reference | Reference |
| Teaching status | ||
| No | 0.81 (0.76‐0.87) | 1.0 (0.95‐1.0) |
| Yes | Reference | Reference |
| Government ownership | ||
| No | 1.1 (1.01.2) | 1.0 (0.98‐1.1) |
| Yes | Reference | Reference |
| Census region | ||
| Mid‐Atlantic and New England | 1.0 (0.88‐1.1) | 0.91 (0.84‐0.99) |
| South Atlantic | 0.95 (0.85‐1.1) | 1.0 (0.95‐1.1) |
| Central 1 | 0.95 (0.85‐1.0) | 0.95 (0.89‐1.0) |
| Central 2 | 0.71 (0.62‐0.81) | 0.91 (0.83‐0.99) |
| Central 3 | 0.80 (0.71‐0.91) | 0.97 (0.90‐1.0) |
| Central 4 | 0.76 (0.68‐0.86) | 0.93 (0.85‐1.0) |
| Mountain | 0.84 (0.73‐0.96) | 0.98 (0.90‐1.1) |
| Pacific | Reference | Reference |
| Average survey response rate within hospital | 0.65 (0.58‐0.72) | 0.93 (0.82‐1.0) |
| Respondent characteristics | ||
| How long have you worked in your current specialty or profession? | ||
| <1 year | 0.75 (0.73‐0.78) | 1.03 (0.99‐1.1) |
| 15 years | 0.99 (0.97‐1.0) | 1.1 (1.1‐1.1) |
| 610 years | 1.0 (1.01.1) | 0.99 (0.96‐1.0) |
| 1115 years | 1.0 (1.01.1) | 1.0 (0.97‐1.0) |
| 1620 years | 1.0 (0.98‐1.0) | 0.97 (0.94‐1.0) |
| 21 years | Reference | Reference |
| How long have you worked in this hospital? | ||
| <1 year | 0.75 (0.73‐0.77) | 0.90 (0.85‐0.90) |
| 15 years | 1.03 (1.001.05) | 0.99 (0.95‐1.0) |
| 610 years | 1.1 (1.1‐1.1) | 0.99 (0.95‐1.0) |
| 1115 years | 1.1 (1. 01.1) | 1.0 (0.96‐1.0) |
| 1620 years | 1.1 (1.01.1) | 0.98 (0.94‐1.0) |
| 21 years | Reference | Reference |
| How long have you worked in your current hospital work area/unit? | ||
| <1 year | 0.79 (0.76‐0.82) | 0.98 (0.93‐1.0) |
| 15 years | 1.0 (1.01.1) | 1.0 (0.99‐1.1) |
| 610 years | 1.1 (1.1‐1.1) | 1.0 (1.01.1) |
| 1115 years | 1.1 (1.01.1) | 1.0 (0.99‐1.1) |
| 1620 years | 1.1 (1.01.1) | 1.1 (1.01.1) |
| 21 years | Reference | Reference |
| Typically, how many hours per week do you work in this hospital? | ||
| <20 | 0.63 (0.50‐0.79) | 0.91 (0.72‐1.2) |
| 2039 | 0.75 (0.59‐0.94) | 0.90 (0.71‐1.1) |
| 4059 | 0.87 (0.69‐1.1) | 1.1 (0.85‐1.4) |
| 6079 | 0.95 (0.75‐1.2) | 1.0 (0.82‐1.3) |
| 8099 | 0.99 (0.78‐1.2) | 1.1 (0.86‐1.4) |
| 100 | Reference | Reference |
| What is your staff position in this hospital? | ||
| Registered nurse | 0.92 (0.90‐0.94) | 1.1 (0.98‐1.0) |
| Technician (eg, ECG, lab, radiology) | Reference | Reference |
| Unit assistant/clerk/secretary | 0.79 (0.76‐0.81) | 0.94 (0.80‐0.96) |
| Patient care assistant/hospital aide/care partner | 0.78 (0.75‐0.81) | 0.96 (0.90‐0.98) |
| Physical, occupational, or speech therapist | 0.88 (0.84‐0.92) | 1.2 (1.1‐1.2) |
| Attending/staff physician | 1.0 (0.97‐1.1) | 1.3 (1.2‐1.3) |
| LVN/LPN | 0.89 (0.85‐0.94) | 1.0 (0.92‐1.0) |
| Respiratory therapist | 0.84 (0.80‐0.88) | 0.97 (0.89‐1.0) |
| Pharmacist | 1.5 (1.4‐1.6) | 1.3 (1.1‐1.3) |
| Physician assistant/nurse practitioner | 0.93 (0.87‐1.0) | 1.2 (1.1‐1.2) |
| Resident physician/physician in training | 0.96 (0.89‐1.0) | 1.3 (1.2‐1.4) |
| Dietician | 0.86 (0.79‐0.94) | 1.2 (1.1‐1.3) |
| In your staff position, do you typically have direct interaction or contact with patients? | ||
| Yes | 0.83 (0.82‐0.85) | 0.85 (0.83‐0.87) |
| No | Reference | Reference |
| What is your primary work area or unit in this hospital? | ||
| Other | Reference | Reference |
| Medicine (nonsurgical) | 1.1 (1.01.1) | 0.84 (0.82‐0.89) |
| Surgery | 1.1 (1.1‐1.2) | 0.88 (0.86‐0.91) |
| Intensive care unit (any type) | 0.93 (0.90‐0.96) | 0.78 (0.76‐0.81) |
| Many different hospital units/no specific unit | 1.2 (1.1‐1.2) | 1.0 (0.98‐ 1.0) |
| Radiology | 1.1 (1.1‐1.1) | 1.0 (1.01.1) |
| Emergency department | 1.0 (0.97‐1.0) | 0.57 (0.55‐0.60) |
| Obstetrics | 0.76 (0.73‐0.79) | 0.66 (0.63‐0.69) |
| Laboratory | 1.2 (1.2‐1.3) | 1.0 (1.01.1) |
| Rehabilitation | 1.0 (0.97‐1.0) | 1.0 (0.98‐1.1) |
| Pediatrics | 0.90 (0.86‐0.94) | 0.83 (0.80‐0.87) |
| Pharmacy | 1.6 (1.5‐1.7) | 1.1 (1.01.2) |
| Psychiatry/mental health | 1.2 (1.1‐1.2) | 0.96 (0.90‐1.0) |
| Anesthesiology | 1.1 (1.01.3) | 0.93 (0.83‐1.0) |
| Respondent perceptions | ||
| When a lot of work needs to be done quickly, we work together as a team to get the work done. | ||
| Strongly disagree | 3.2 (3.03.4) | 1.0 (0.98‐1.1) |
| Disagree | 3.2 (3.13.3) | 1.0 (1.01.1) |
| Neutral | 2.3 (2.2‐2.4) | 0.98 (0.94‐1.0) |
| Agree | 2.3 (2.2‐2.4) | 1.0 (1.002‐1.04) |
| Strongly agree | Reference | Reference |
| Staff in this unit work longer hours than is best for patient care. | ||
| Strongly disagree | 0.51 (0.48‐0.53) | 0.76 (0.73‐0.79) |
| Disagree | 0.68 (0.67‐0.70) | 0.81 (0.78‐0.84) |
| Neutral | 0.94 (0.91‐0.97) | 0.93 (0.90‐0.97) |
| Agree | 1.0 (0.99‐1.1) | 0.94 (0.91‐0.98) |
| Strongly agree | Reference | Reference |
| When 1 area in this unit gets really busy, others help out. | ||
| Strongly disagree | 3.8 (3.7 ‐ 4.0) | 1.0 (0.96‐1.1) |
| Disagree | 3.0 (2.9‐3.1) | 1.0 (0.99‐1.1) |
| Neutral | 2.2 (2.12.3) | 1.0 (0.97‐1.0) |
| Agree | 1.5 (1.5‐1.6) | 0.99 (0.96‐1.0) |
| Strongly agree | Reference | Reference |
| Hospital units do not coordinate well with each other. | ||
| Strongly disagree | 0.03 (0.03‐0.04) | 0.10 (0.10‐0.11) |
| Disagree | 0.08 (0.08‐0.08) | 0.18 (0.17‐0.19) |
| Neutral | 0.21 (0.20‐0.22) | 0.32 (0.30‐0.33) |
| Agree | 0.50 (0.48‐0.52) | 0.61 (0.58‐0.63) |
| Strongly agree | Reference | Reference |
| There is good cooperation among hospital units that need to work together. | ||
| Strongly disagree | 20.1 (18.921.5) | 4.7 (4.35.0) |
| Disagree | 14.2 (13.614.9) | 4.2 (4.14.5) |
| Neutral | 6.7 (6.47.0) | 2.7 (2.6‐2.8) |
| Agree | 2.4 (2.3‐2.5) | 1.6 (1.6‐1.7) |
| Strongly agree | Reference | Reference |
| Please give your work area/unit in this hospital an overall grade on patient safety | ||
| Excellent | 0.13 (0.12‐0.14) | 0.47 (0.42‐0.52) |
| Very good | 0.24 (0.21‐0.26) | 0.63 (0.57‐0.70) |
| Acceptable | 0.49 (0.45‐0.54) | 0.79 (0.72‐0.88) |
| Poor | 0.83 (0.75‐0.92) | 0.92 (0.83‐1.03) |
| Failing | Reference | Reference |
DISCUSSION
Our results illustrate that when hospital staff agree that their hospital works in crisis mode, they are more likely to agree that their hospital unit had frequent problems exchanging patient information across units. Because hospital staff working under time constraints and heavy workloads could potentially be at risk of misinterpreting or delivering inaccurate information, these results imply that crisis mode work climates increase the risk of problematic health information exchange. An equally plausible interpretation could be that problematic patient health information exchange increases the risk of hospital staff perceiving crisis mode work climates. Given that information gaps are associated with patient handoff errors,[14] and that patient handoff errors are associated with adverse events,[2, 3, 6, 8] an urgent need exists to implement information exchange systems that prevent information gaps from harming patients. Consequently, hospitals need to implement workflow strategies that prevent information gaps from undermining patient safety during transitions of care.
Other factors affect information exchange apart from crisis mode work climate, as illustrated by the significant associations of key covariates in the multivariate model. The effect found between perceived coordination and information exchange implies that improving information exchange requires good cooperation and coordination. The effect found between patient contact and information exchange implies that working directly with patients improves either the accuracy or the perception of information exchange. Finally, the effect found between hospital size and information exchange suggests that small hospitals are less likely than large hospitals to have information exchange problem. The geographical dispersion and the complexity of larger institutions could result in information exchange problems due to more confusion and less in‐person communication.
Because problematic patient information exchanges are associated with hospital size, coordination, and patient contact, in addition to crisis mode work climate, multifaceted solutions are necessary to resolve the problem. For example, hospital interventions designed to improve coordination could in turn attenuate perceived crisis modes. Furthermore, tailoring these interventions to hospitals that belong to complex geographically dispersed provider networks would likely decrease errors during transitions of care. Because multiple factors cause information exchange problems, implementing interventions that improve both coordination and crisis mode work climates would likely result in a greater net improvement compared to interventions focused solely on decreasing crisis mode work climates.
Some limitations of our paper are worth noting. First, we did not have information on the volume of data exchanged or the functionality levels of the electronic health record systems, both of which likely impact the accuracy of patient information exchange. For example, hospitals with smaller versus larger amounts of data exchanged could be less prone to error. On the other hand, this risk of error could be reduced even further by implementing robust health information technology (IT) systems that improve the accuracy of information transfer. This is consistent with studies showing that hospitals without computerized provider order entry (CPOE) systems have been shown to have higher medication error rates compared to those hospitals with CPOE systems.[15] Therefore, omitting data volume and health IT capabilities from the multivariate model could introduce unobserved heterogeneity, resulting in biased associations between perceived crisis mode work climate and perceived information exchange problems. Second, the cross‐sectional design limits our ability to infer causality because we are not certain whether the perceived crisis mode occurred before, after, or simultaneously to perceived information exchange problems. Third, the self‐reported nature of the questionnaire items does not provide information on observed levels of crisis mode and exchange problems, which could be inconsistent with perceived levels. Fourth, the relatively low within‐hospital response rate decreases the external validity of our findings. For example, if responders' perceptions of crisis mode or information exchange problems significantly differed from nonresponders, our results would not be generalizable to the larger population of acute‐care hospitals across the United States. Therefore, conclusions should be viewed with caution if applying these results to hospitals with respondents significantly differing from those contained within our sample.
Despite these limitations, the large sample size in conjunction with the use of data from a survey having acceptable psychometric properties[16] strengthens the external and internal validity of our findings. Although questionnaire items measuring perceptions are relatively subjective in nature compared to using metrics that capture observed problems or crisis modes, we argue that staff perception data are equally informative, as they guide organization leaders on how to improve workplace performance. Because a core concept of high reliability organizations (HROs) is to preserve constant awareness by key leaders and staff of the state of the systems and processes that affect patient care,[17] HROs could benefit from knowing the extent to which staff perceptions impact patient care. From a methods perspective, the multivariable ordinal regressions enabled us to control for potential confounders that if omitted could have resulted in biased estimates. Furthermore, low levels of multicollinearity as illustrated by low variation inflation factors enabled us to isolate the independent effect of crisis mode perceptions. Including hospital size and hospital work unit as covariates was an additional methodological strength helping account for the unobserved heterogeneity caused by excluding volume of data exchanged or health IT system capability. For example, because larger compared to smaller hospitals usually have more sophisticated health IT systems,[15] including bed size in the model theoretically captures some of the variation that would have been captured if we were able to include a covariate measuring health IT capability. Last, using ordinal regression facilitates interpretation of the findings because the questionnaire items for the predictor and outcome were originally captured on a Likert scale.
Our findings underscore the significant impact that work climate has on accurate information exchange, and ultimately patient safety. Improving patient safety is imperative for hospitals, especially within the context of recent regulations stemming from the Affordable Care Act that incentivize hospitals to reduce readmissions[18] and improve transitions of care.[19] Because accurate health information exchange is a critical component of patient care, resolving barriers that decrease the accuracy of this exchange is essential. Therefore, future studies need to continue examining these associations within the context of study designs that incorporate longitudinal data and datasets that include objective measures capturing crisis mode work climates and information exchange problems. Because effective communication during handoffs is associated with decreases in medical errors and readmissions, hospitals need to continually ensure that work environments are conducive to effective patient information exchange.
Disclosures
Nothing to report
- , , . Safety and complexity: inter‐departmental relationships as a threat to patient safety in the operating department. J Health Organ Manag. 2006;20(2–3):227–242.
- , , , , . Communication loads on clinical staff in the emergency department. Med J Aust. 2002;176(9):415–418.
- , , , et al. Rates of medical errors and preventable adverse events among hospitalized children following implementation of a resident handoff bundle. JAMA. 2013;310(21):2262–2270.
- , , . The Handbook of Group Communication Theory and Research. Thousand Oaks, CA: Sage Publications; 1999.
- , , . Judgment and decision making under time pressure. In: Svenson O, Maule AJ, eds. Time Pressure and Stress in Human Judgment and Decision Making. New York, NY: Plenum Press; 1993:27–40.
- , , , , . Learning from error: identifying contributory causes of medication errors in an Australian hospital. Med J Aust. 2008;188(5):276–279.
- , , . Communicating in the “gray zone”: perceptions about emergency physician hospitalist handoffs and patient safety. Acad Emerg Med. 2007;14(10):884–894.
- , . Emergency physician intershift handovers: an analysis of our transitional care. Pediatr Emerg Care. 2006;22(10):751–754.
- , , , , , . Dropping the baton: a qualitative analysis of failures during the transition from emergency department to inpatient care. Ann Emerg Med. 2009;53(6):701–710.
- , , , . Lost in translation: challenges and opportunities in physician‐to‐physician communication during patient handoffs. Acad Med. 2005;80(12):1094–1099.
- , , , et al. Hospital Survey on Patient Safety Culture: 2010 user comparative database report. (Prepared by Westat, Rockville, MD, under Contract No. HHSA 290200710024C). Rockville, MD: Agency for Healthcare Research and Quality; February 2010. AHRQ Publication No. 10‐0026.
- , , , , . Physiological and behavioural response patterns at work among hospital nurses. J Nurs Manag. 2011;19(1):57–68.
- , . The treatment of missing values and its effect in the classifier accuracy. In: Banks D, House L, McMorris FR, Arabie P, Gaul W, eds. Classification, Clustering and Data Mining Applications. Berlin, Germany: Springer‐Verlag; 2004.
- , . A systematic review of failures in handoff communication during intrahospital transfers. Jt Comm J Qual Patient Saf. 2011;37(6):274–284.
- , , , et al. Reduction in medication errors in hospitals due to adoption of computerized provider order entry systems. J Am Med Inform Assoc. 2013;20(3):470–476.
- , . Multilevel psychometric properties of the AHRQ hospital survey on patient safety culture. BMC Health Serv Res. 2010;10:199.
- , , , et al. Becoming a High Reliability Organization: Operational Advice for Hospital Leaders. Prepared by the Lewin Group under contract no. 290‐04‐0011. AHRQ publication no. 08–0022. Rockville, MD: Agency for Healthcare Research and Quality; 2008.
- Health policy brief: Medicare hospital readmissions reduction program. Health Affairs. November 12, 2013. Available at: http://www.healthaffairs.org/healthpolicybriefs/brief.php?brief_id=102. Accessed August 11, 2014.
- , , , , . The care span: the importance of transitional care in achieving health reform. Health Aff (Millwood). 2011;30(4):746–754.
- , , . Safety and complexity: inter‐departmental relationships as a threat to patient safety in the operating department. J Health Organ Manag. 2006;20(2–3):227–242.
- , , , , . Communication loads on clinical staff in the emergency department. Med J Aust. 2002;176(9):415–418.
- , , , et al. Rates of medical errors and preventable adverse events among hospitalized children following implementation of a resident handoff bundle. JAMA. 2013;310(21):2262–2270.
- , , . The Handbook of Group Communication Theory and Research. Thousand Oaks, CA: Sage Publications; 1999.
- , , . Judgment and decision making under time pressure. In: Svenson O, Maule AJ, eds. Time Pressure and Stress in Human Judgment and Decision Making. New York, NY: Plenum Press; 1993:27–40.
- , , , , . Learning from error: identifying contributory causes of medication errors in an Australian hospital. Med J Aust. 2008;188(5):276–279.
- , , . Communicating in the “gray zone”: perceptions about emergency physician hospitalist handoffs and patient safety. Acad Emerg Med. 2007;14(10):884–894.
- , . Emergency physician intershift handovers: an analysis of our transitional care. Pediatr Emerg Care. 2006;22(10):751–754.
- , , , , , . Dropping the baton: a qualitative analysis of failures during the transition from emergency department to inpatient care. Ann Emerg Med. 2009;53(6):701–710.
- , , , . Lost in translation: challenges and opportunities in physician‐to‐physician communication during patient handoffs. Acad Med. 2005;80(12):1094–1099.
- , , , et al. Hospital Survey on Patient Safety Culture: 2010 user comparative database report. (Prepared by Westat, Rockville, MD, under Contract No. HHSA 290200710024C). Rockville, MD: Agency for Healthcare Research and Quality; February 2010. AHRQ Publication No. 10‐0026.
- , , , , . Physiological and behavioural response patterns at work among hospital nurses. J Nurs Manag. 2011;19(1):57–68.
- , . The treatment of missing values and its effect in the classifier accuracy. In: Banks D, House L, McMorris FR, Arabie P, Gaul W, eds. Classification, Clustering and Data Mining Applications. Berlin, Germany: Springer‐Verlag; 2004.
- , . A systematic review of failures in handoff communication during intrahospital transfers. Jt Comm J Qual Patient Saf. 2011;37(6):274–284.
- , , , et al. Reduction in medication errors in hospitals due to adoption of computerized provider order entry systems. J Am Med Inform Assoc. 2013;20(3):470–476.
- , . Multilevel psychometric properties of the AHRQ hospital survey on patient safety culture. BMC Health Serv Res. 2010;10:199.
- , , , et al. Becoming a High Reliability Organization: Operational Advice for Hospital Leaders. Prepared by the Lewin Group under contract no. 290‐04‐0011. AHRQ publication no. 08–0022. Rockville, MD: Agency for Healthcare Research and Quality; 2008.
- Health policy brief: Medicare hospital readmissions reduction program. Health Affairs. November 12, 2013. Available at: http://www.healthaffairs.org/healthpolicybriefs/brief.php?brief_id=102. Accessed August 11, 2014.
- , , , , . The care span: the importance of transitional care in achieving health reform. Health Aff (Millwood). 2011;30(4):746–754.
© 2014 Society of Hospital Medicine
Furuncular Myiasis in 2 American Travelers Returning From Senegal
Case Reports
Patient 1
A 16-year-old adolescent boy presented to the emergency department with painful, pruritic, erythematous nodules on the bilateral legs of 1 week’s duration. The lesions had developed 1 week after returning from a monthlong trip to Senegal with a volunteer youth group. He did not recall sustaining any painful insect bites or illnesses while traveling in Africa and only noticed the erythematous papules on the legs when he returned home to the United States. After consulting with his primary care physician and a local dermatologist, the patient began taking oral cephalexin for suspected bacterial furunculosis with no considerable improvement. Over the course of 1 week, the lesions became increasingly painful and pruritic, prompting a visit to the emergency department. Prior to his arrival, the patient reported squeezing a live worm from one of the lesions on the right ankle.
On presentation, the patient was afebrile (temperature, 36.7°C) and his vital signs revealed no abnormalities. Physical examination revealed tender erythematous nodules on the bilateral heels, ankles, and shins with pinpoint puncta noted at the center of many of the lesions (Figure 1). The nodules were warm and indurated and no pulsatile movement was appreciated. The legs appeared to be well perfused with intact sensation and motor function. The patient brought in the live mobile larva that he extruded from the lesion on the right ankle. Both the departments of infectious diseases and dermatology were consulted and a preliminary diagnosis of furuncular myiasis was made.
The lesions were occluded with petroleum jelly and the patient was instructed to follow-up with the dermatology department later that same day. On follow-up in the dermatology clinic, the tips of intact larvae were appreciated at the central puncta of some of the lesions (Figure 2). Lidocaine adrenaline tetracaine gel was applied to lesions on the legs for 40 minutes, then lidocaine gel 1% was injected into each lesion. On injection, immobile larvae were ejected from the central puncta of most of the lesions; the remaining lesions were treated via 3-mm punch biopsy as a means of extraction. Each nodule contained only a single larva, all of which were dead at the time of removal (Figure 3). The wounds were left open and the patient was instructed to continue treatment with cephalexin with leg elevation and rest. Pathologic examination of deep dermal skin sections revealed larval fragments encased by a thick chitinous cuticle with spines that were consistent with furuncular myiasis (Figures 4 and 5). Given the patient’s recent history of travel to Africa along with the morphology of the extracted specimens, the larvae were identified as Cordylobia anthropophaga, a common cause of furuncular myiasis in that region.
Patient 2
The next week, a 17-year-old adolescent girl who had been on the same trip to Senegal as patient 1 presented with 2 similar erythematous nodules with central crusts on the left inner thigh and buttock. On noticing the lesions approximately 3 days prior to presentation, the patient applied topical antibiotic ointment to each nodule, which incited the evacuation of white tube-shaped structures that were presented for examination. On presentation, the nodules were healing well. Given the patient’s travel history and physical examination, a presumptive diagnosis of furuncular myiasis from C anthropophaga also was made.
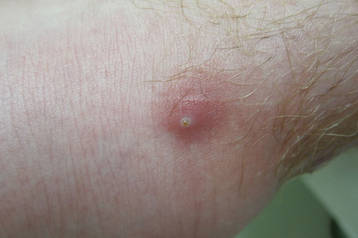

|
Comment
The term myiasis stems from the Greek term for fly and is used to describe the infestation of fly larvae in living vertebrates.1 Myiasis has many classifications, the 3 most common being furuncular, migratory, and wound myiasis, which are differentiated by the different fly species found in distinct regions of the world. Furuncular myiasis is the most benign form, usually affecting only a localized region of the skin; migratory myiasis is characterized by larvae traveling substantial distances from one anatomic site to another within the lower layers of the epidermis; and wound myiasis involves rapid reproduction of larvae in necrotic tissue with subsequent tissue destruction.2
The clinical presentation of the lesions noted in our patients suggested a diagnosis of furuncular myiasis, which commonly is caused by Dermatobia hominis, C anthropophaga, Cuterebra species, Wohlfahrtia vigil, and Wohlfahrtia opaca larvae.3Dermatobia hominis is the most common cause of furuncular myiasis and usually is found in Central and South America. Our patients likely developed an infestation of C anthropophaga (also known as the tumbu fly), a yellow-brown, 7- to 12-mm blowfly commonly found throughout tropical Africa.3 Although C anthropophaga is historically limited to sub-Saharan Africa, there has been a report of a case acquired in Portugal.4
In a review of the literature, C anthropophaga myiasis was documented in Italian travelers returning from Senegal5-7; our cases are unique because they represent North American travelers returning from Senegal with furuncular myiasis. Furuncular myiasis from C anthropophaga has been reported in travelers returning to North America from other African countries, including Angola,8 Tanzania,9-11 Kenya,9 Sierra Leone,12 and Ivory Coast.13 Several cases of ocular myiasis from D hominis and Oestrus ovis have been reported in European travelers returning from Tunisia.14,15
Tumbu fly infestations typically affect dogs and rodents but can arise in human hosts.3 Children may be affected by C anthropophaga furuncular myiasis more often than adults because they have thinner skin and less immunity to the larvae.2
|
There are 2 mechanisms by which infestation of human hosts by C anthropophaga can occur. Most commonly, female flies lay eggs in shady areas in soil that is contaminated by feces or urine. The hatched larvae can survive in the ground for up to 2 weeks and later attach to a host when prompted by heat or movement.3 Therefore, clothing set out to dry may be contaminated by this soil. Alternatively, female flies can lay eggs directly onto clothing that is contaminated by feces or urine and the larvae subsequently hatch outside the soil with easy access to human skin once the clothing is worn.2
Common penetration sites are the head, neck, and back, as well as areas covered by contaminated or infested clothing.2,3 Penetration of the human skin occurs instantly and is a painless process that is rarely noticed by the human host.3 The larvae burrow into the skin for 8 to 12 days, resulting in a furuncle that occasionally secretes a serous fluid.2 Within the first 2 days of infestation, the host may experience symptoms ranging from local pruritus to severe pain. Six days following initial onset, an intense inflammatory response may result in local lymphadenopathy along with fever and fatigue.2 The larvae use their posterior spiracles to create openings in the skin to create air holes that allow them to breathe.3 On physical examination, the spiracles generally appear as 1- to 3-mm dark linear streaks within furuncles, which is important in the diagnosis of C anthropophaga furuncular myiasis.1,3 If spiracles are not appreciated on initial examination, diagnosis can be made by submerging the affected areas in water or saliva to look for air bubbles arising from the central puncta of the lesions.1
All causes of furuncular myiasis are characterized by a ratio of 1 larva to 1 furuncle.16 Although most of these types of larvae that can cause furuncular myiasis result in single lesions, C anthropophaga infestation often produces several furuncles that may coalesce into plaques.1,2 The differential diagnosis for C anthropophaga furuncular myiasis includes pyoderma, impetigo, staphylococcal furunculosis, cutaneous leishmaniasis, infected cyst, retained foreign body, and facticial disease.2,3 Dracunculiasis also may be considered, which occurs after ingestion of contaminated water.2 Ultrasonography may be helpful for the diagnosis of furuncular myiasis, as it can facilitate identification of foreign bodies, abscesses, and even larvae in some cases.17 Definitive diagnosis of any type of myiasis involves extraction of the larva and identification of the family, genus, and species by a parasitologist.1 Some experts suggest rearing preserved live larvae with raw meat after extraction because adult specimens are more reliable than larvae for species diagnosis.1
Treatment of furuncular myiasis involves occlusion and extraction of the larvae from the skin. Suffocation of the larvae by occlusion of air holes with petroleum jelly, paraffin oil, bacon fat, glue, and other obstructing substances forces the larvae to emerge in search of oxygen, though immature larvae may be more reluctant than mature ones.2,3 Definitive treatment involves the direct removal of the larvae by surgery or expulsion by pressure, though it is recommended that lesions are pretreated with occlusive techniques.1,3 Other reported methods of extraction include injection of lidocaine and the use of a commercial venom extractor.1 It should be noted that rupture and incomplete extraction of larvae can lead to secondary infections and allergic reactions. Lesions can be pretreated with lidocaine gel prior to extraction, and antibiotics should be used in cases of secondary bacterial infection. Ivermectin also has been reported as a treatment of furuncular myiasis and other types of myiasis.1 Prevention of infestation by C anthropophaga includes avoidance of endemic areas, maintaining good hygiene, and ironing clothing or drying it in sunny locations.1,2 Overall, furuncular myiasis has a good prognosis with rapid recovery and a low incidence of complications.1
Conclusion
We present 2 cases of travelers returning to North America from Senegal with C anthropophaga furuncular myiasis. Careful review of travel history, physical examination, and identification of fly larvae are important for diagnosis. Individuals traveling to sub-Saharan Africa should avoid drying clothes in shady places and lying on the ground. They also are urged to iron their clothing before wearing it.
1. Caissie R, Beaulieu F, Giroux M, et al. Cutaneous myiasis: diagnosis, treatment, and prevention. J Oral Maxillofac Surg. 2008;66:560-568.
2. McGraw TA, Turiansky GW. Cutaneous myiasis. J Am Acad Dermatol. 2008;58:907-926.
3. Robbins K, Khachemoune A. Cutaneous myiasis: a review of the common types of myiasis. Int J Dermatol. 2010;49:1092-1098.
4. Curtis SJ, Edwards C, Athulathmuda C, et al. Case of the month: cutaneous myiasis in a returning traveller from the Algarve: first report of tumbu maggots, Cordylobia anthropophaga, acquired in Portugal. Emerg Med J. 2006;23:236-237.
5. Veraldi S, Brusasco A, Süss L. Cutaneous myiasis caused by larvae of Cordylobia anthropophaga (Blanchard). Int J Dermatol. 1993;32:184-187.
6. Cultrera R, Dettori G, Calderaro A, et al. Cutaneous myiasis caused by Cordylobia anthropophaga (Blanchard 1872): description of 5 cases from costal regions of Senegal [in Italian]. Parassitologia. 1993;35:47-49.
7. Fusco FM, Nardiello S, Brancaccio G, et al. Cutaneous myiasis from Cordylobia anthropophaga in a traveller returning from Senegal: a case study [in Italian]. Infez Med. 2005;13:109-111.
8. Lee EJ, Robinson F. Furuncular myiasis of the face caused by larva of the tumbu fly (Cordylobia anthropophaga)[published online ahead of print July 21, 2006]. Eye (Lond). 2007;21:268-269.
9. Rice PL, Gleason N. Two cases of myiasis in the United States by the African tumbu fly, Cordylobia anthropophaga (Diptera, Calliphoridae). Am J Trop Med Hyg. 1972;21:62-65.
10. March CH. A case of “ver du Cayor” in Manhattan. Arch Dermatol. 1964;90:32-33.
11. Schorr WF. Tumbu-fly myiasis in Marshfield, Wis. Arch Dermatol. 1967;95:61-62.
12. Potter TS, Dorman MA, Ghaemi M, et al. Inflammatory papules on the back of a traveling businessman. tumbu
fly myiasis. Arch Dermatol. 1995;131:951, 954.
13. Ockenhouse CF, Samlaska CP, Benson PM, et al. Cutaneous myiasis caused by the African tumbu fly (Cordylobia anthropophaga). Arch Dermatol. 1990;126:199-202.
14. Kaouech E, Kallel K, Belhadj S, et al. Dermatobia hominis furuncular myiasis in a man returning from Latin America: first imported case in Tunisia [in French]. Med Trop (Mars). 2010;70:135-136.
15. Zayani A, Chaabouni M, Gouiaa R, et al. Conjuctival myiasis. 23 cases in the Tunisian Sahel [in French]. Arch Inst Pasteur Tunis. 1989;66:289-292.
16. Latorre M, Ullate JV, Sanchez J, et al. A case of myiasis due to Dermatobia hominis. Eur J Clin Microbiol Infect Dis. 1993;12:968-969.
17. Mahal JJ, Sperling JD. Furuncular myiasis from Dermatobia hominis: a case of human botfly infestation [published online ahead of print February 1, 2010]. J Emerg Med. 2012;43:618-621.
Case Reports
Patient 1
A 16-year-old adolescent boy presented to the emergency department with painful, pruritic, erythematous nodules on the bilateral legs of 1 week’s duration. The lesions had developed 1 week after returning from a monthlong trip to Senegal with a volunteer youth group. He did not recall sustaining any painful insect bites or illnesses while traveling in Africa and only noticed the erythematous papules on the legs when he returned home to the United States. After consulting with his primary care physician and a local dermatologist, the patient began taking oral cephalexin for suspected bacterial furunculosis with no considerable improvement. Over the course of 1 week, the lesions became increasingly painful and pruritic, prompting a visit to the emergency department. Prior to his arrival, the patient reported squeezing a live worm from one of the lesions on the right ankle.
On presentation, the patient was afebrile (temperature, 36.7°C) and his vital signs revealed no abnormalities. Physical examination revealed tender erythematous nodules on the bilateral heels, ankles, and shins with pinpoint puncta noted at the center of many of the lesions (Figure 1). The nodules were warm and indurated and no pulsatile movement was appreciated. The legs appeared to be well perfused with intact sensation and motor function. The patient brought in the live mobile larva that he extruded from the lesion on the right ankle. Both the departments of infectious diseases and dermatology were consulted and a preliminary diagnosis of furuncular myiasis was made.
The lesions were occluded with petroleum jelly and the patient was instructed to follow-up with the dermatology department later that same day. On follow-up in the dermatology clinic, the tips of intact larvae were appreciated at the central puncta of some of the lesions (Figure 2). Lidocaine adrenaline tetracaine gel was applied to lesions on the legs for 40 minutes, then lidocaine gel 1% was injected into each lesion. On injection, immobile larvae were ejected from the central puncta of most of the lesions; the remaining lesions were treated via 3-mm punch biopsy as a means of extraction. Each nodule contained only a single larva, all of which were dead at the time of removal (Figure 3). The wounds were left open and the patient was instructed to continue treatment with cephalexin with leg elevation and rest. Pathologic examination of deep dermal skin sections revealed larval fragments encased by a thick chitinous cuticle with spines that were consistent with furuncular myiasis (Figures 4 and 5). Given the patient’s recent history of travel to Africa along with the morphology of the extracted specimens, the larvae were identified as Cordylobia anthropophaga, a common cause of furuncular myiasis in that region.
Patient 2
The next week, a 17-year-old adolescent girl who had been on the same trip to Senegal as patient 1 presented with 2 similar erythematous nodules with central crusts on the left inner thigh and buttock. On noticing the lesions approximately 3 days prior to presentation, the patient applied topical antibiotic ointment to each nodule, which incited the evacuation of white tube-shaped structures that were presented for examination. On presentation, the nodules were healing well. Given the patient’s travel history and physical examination, a presumptive diagnosis of furuncular myiasis from C anthropophaga also was made.


|
Comment
The term myiasis stems from the Greek term for fly and is used to describe the infestation of fly larvae in living vertebrates.1 Myiasis has many classifications, the 3 most common being furuncular, migratory, and wound myiasis, which are differentiated by the different fly species found in distinct regions of the world. Furuncular myiasis is the most benign form, usually affecting only a localized region of the skin; migratory myiasis is characterized by larvae traveling substantial distances from one anatomic site to another within the lower layers of the epidermis; and wound myiasis involves rapid reproduction of larvae in necrotic tissue with subsequent tissue destruction.2
The clinical presentation of the lesions noted in our patients suggested a diagnosis of furuncular myiasis, which commonly is caused by Dermatobia hominis, C anthropophaga, Cuterebra species, Wohlfahrtia vigil, and Wohlfahrtia opaca larvae.3Dermatobia hominis is the most common cause of furuncular myiasis and usually is found in Central and South America. Our patients likely developed an infestation of C anthropophaga (also known as the tumbu fly), a yellow-brown, 7- to 12-mm blowfly commonly found throughout tropical Africa.3 Although C anthropophaga is historically limited to sub-Saharan Africa, there has been a report of a case acquired in Portugal.4
In a review of the literature, C anthropophaga myiasis was documented in Italian travelers returning from Senegal5-7; our cases are unique because they represent North American travelers returning from Senegal with furuncular myiasis. Furuncular myiasis from C anthropophaga has been reported in travelers returning to North America from other African countries, including Angola,8 Tanzania,9-11 Kenya,9 Sierra Leone,12 and Ivory Coast.13 Several cases of ocular myiasis from D hominis and Oestrus ovis have been reported in European travelers returning from Tunisia.14,15
Tumbu fly infestations typically affect dogs and rodents but can arise in human hosts.3 Children may be affected by C anthropophaga furuncular myiasis more often than adults because they have thinner skin and less immunity to the larvae.2
|
There are 2 mechanisms by which infestation of human hosts by C anthropophaga can occur. Most commonly, female flies lay eggs in shady areas in soil that is contaminated by feces or urine. The hatched larvae can survive in the ground for up to 2 weeks and later attach to a host when prompted by heat or movement.3 Therefore, clothing set out to dry may be contaminated by this soil. Alternatively, female flies can lay eggs directly onto clothing that is contaminated by feces or urine and the larvae subsequently hatch outside the soil with easy access to human skin once the clothing is worn.2
Common penetration sites are the head, neck, and back, as well as areas covered by contaminated or infested clothing.2,3 Penetration of the human skin occurs instantly and is a painless process that is rarely noticed by the human host.3 The larvae burrow into the skin for 8 to 12 days, resulting in a furuncle that occasionally secretes a serous fluid.2 Within the first 2 days of infestation, the host may experience symptoms ranging from local pruritus to severe pain. Six days following initial onset, an intense inflammatory response may result in local lymphadenopathy along with fever and fatigue.2 The larvae use their posterior spiracles to create openings in the skin to create air holes that allow them to breathe.3 On physical examination, the spiracles generally appear as 1- to 3-mm dark linear streaks within furuncles, which is important in the diagnosis of C anthropophaga furuncular myiasis.1,3 If spiracles are not appreciated on initial examination, diagnosis can be made by submerging the affected areas in water or saliva to look for air bubbles arising from the central puncta of the lesions.1
All causes of furuncular myiasis are characterized by a ratio of 1 larva to 1 furuncle.16 Although most of these types of larvae that can cause furuncular myiasis result in single lesions, C anthropophaga infestation often produces several furuncles that may coalesce into plaques.1,2 The differential diagnosis for C anthropophaga furuncular myiasis includes pyoderma, impetigo, staphylococcal furunculosis, cutaneous leishmaniasis, infected cyst, retained foreign body, and facticial disease.2,3 Dracunculiasis also may be considered, which occurs after ingestion of contaminated water.2 Ultrasonography may be helpful for the diagnosis of furuncular myiasis, as it can facilitate identification of foreign bodies, abscesses, and even larvae in some cases.17 Definitive diagnosis of any type of myiasis involves extraction of the larva and identification of the family, genus, and species by a parasitologist.1 Some experts suggest rearing preserved live larvae with raw meat after extraction because adult specimens are more reliable than larvae for species diagnosis.1
Treatment of furuncular myiasis involves occlusion and extraction of the larvae from the skin. Suffocation of the larvae by occlusion of air holes with petroleum jelly, paraffin oil, bacon fat, glue, and other obstructing substances forces the larvae to emerge in search of oxygen, though immature larvae may be more reluctant than mature ones.2,3 Definitive treatment involves the direct removal of the larvae by surgery or expulsion by pressure, though it is recommended that lesions are pretreated with occlusive techniques.1,3 Other reported methods of extraction include injection of lidocaine and the use of a commercial venom extractor.1 It should be noted that rupture and incomplete extraction of larvae can lead to secondary infections and allergic reactions. Lesions can be pretreated with lidocaine gel prior to extraction, and antibiotics should be used in cases of secondary bacterial infection. Ivermectin also has been reported as a treatment of furuncular myiasis and other types of myiasis.1 Prevention of infestation by C anthropophaga includes avoidance of endemic areas, maintaining good hygiene, and ironing clothing or drying it in sunny locations.1,2 Overall, furuncular myiasis has a good prognosis with rapid recovery and a low incidence of complications.1
Conclusion
We present 2 cases of travelers returning to North America from Senegal with C anthropophaga furuncular myiasis. Careful review of travel history, physical examination, and identification of fly larvae are important for diagnosis. Individuals traveling to sub-Saharan Africa should avoid drying clothes in shady places and lying on the ground. They also are urged to iron their clothing before wearing it.
Case Reports
Patient 1
A 16-year-old adolescent boy presented to the emergency department with painful, pruritic, erythematous nodules on the bilateral legs of 1 week’s duration. The lesions had developed 1 week after returning from a monthlong trip to Senegal with a volunteer youth group. He did not recall sustaining any painful insect bites or illnesses while traveling in Africa and only noticed the erythematous papules on the legs when he returned home to the United States. After consulting with his primary care physician and a local dermatologist, the patient began taking oral cephalexin for suspected bacterial furunculosis with no considerable improvement. Over the course of 1 week, the lesions became increasingly painful and pruritic, prompting a visit to the emergency department. Prior to his arrival, the patient reported squeezing a live worm from one of the lesions on the right ankle.
On presentation, the patient was afebrile (temperature, 36.7°C) and his vital signs revealed no abnormalities. Physical examination revealed tender erythematous nodules on the bilateral heels, ankles, and shins with pinpoint puncta noted at the center of many of the lesions (Figure 1). The nodules were warm and indurated and no pulsatile movement was appreciated. The legs appeared to be well perfused with intact sensation and motor function. The patient brought in the live mobile larva that he extruded from the lesion on the right ankle. Both the departments of infectious diseases and dermatology were consulted and a preliminary diagnosis of furuncular myiasis was made.
The lesions were occluded with petroleum jelly and the patient was instructed to follow-up with the dermatology department later that same day. On follow-up in the dermatology clinic, the tips of intact larvae were appreciated at the central puncta of some of the lesions (Figure 2). Lidocaine adrenaline tetracaine gel was applied to lesions on the legs for 40 minutes, then lidocaine gel 1% was injected into each lesion. On injection, immobile larvae were ejected from the central puncta of most of the lesions; the remaining lesions were treated via 3-mm punch biopsy as a means of extraction. Each nodule contained only a single larva, all of which were dead at the time of removal (Figure 3). The wounds were left open and the patient was instructed to continue treatment with cephalexin with leg elevation and rest. Pathologic examination of deep dermal skin sections revealed larval fragments encased by a thick chitinous cuticle with spines that were consistent with furuncular myiasis (Figures 4 and 5). Given the patient’s recent history of travel to Africa along with the morphology of the extracted specimens, the larvae were identified as Cordylobia anthropophaga, a common cause of furuncular myiasis in that region.
Patient 2
The next week, a 17-year-old adolescent girl who had been on the same trip to Senegal as patient 1 presented with 2 similar erythematous nodules with central crusts on the left inner thigh and buttock. On noticing the lesions approximately 3 days prior to presentation, the patient applied topical antibiotic ointment to each nodule, which incited the evacuation of white tube-shaped structures that were presented for examination. On presentation, the nodules were healing well. Given the patient’s travel history and physical examination, a presumptive diagnosis of furuncular myiasis from C anthropophaga also was made.


|
Comment
The term myiasis stems from the Greek term for fly and is used to describe the infestation of fly larvae in living vertebrates.1 Myiasis has many classifications, the 3 most common being furuncular, migratory, and wound myiasis, which are differentiated by the different fly species found in distinct regions of the world. Furuncular myiasis is the most benign form, usually affecting only a localized region of the skin; migratory myiasis is characterized by larvae traveling substantial distances from one anatomic site to another within the lower layers of the epidermis; and wound myiasis involves rapid reproduction of larvae in necrotic tissue with subsequent tissue destruction.2
The clinical presentation of the lesions noted in our patients suggested a diagnosis of furuncular myiasis, which commonly is caused by Dermatobia hominis, C anthropophaga, Cuterebra species, Wohlfahrtia vigil, and Wohlfahrtia opaca larvae.3Dermatobia hominis is the most common cause of furuncular myiasis and usually is found in Central and South America. Our patients likely developed an infestation of C anthropophaga (also known as the tumbu fly), a yellow-brown, 7- to 12-mm blowfly commonly found throughout tropical Africa.3 Although C anthropophaga is historically limited to sub-Saharan Africa, there has been a report of a case acquired in Portugal.4
In a review of the literature, C anthropophaga myiasis was documented in Italian travelers returning from Senegal5-7; our cases are unique because they represent North American travelers returning from Senegal with furuncular myiasis. Furuncular myiasis from C anthropophaga has been reported in travelers returning to North America from other African countries, including Angola,8 Tanzania,9-11 Kenya,9 Sierra Leone,12 and Ivory Coast.13 Several cases of ocular myiasis from D hominis and Oestrus ovis have been reported in European travelers returning from Tunisia.14,15
Tumbu fly infestations typically affect dogs and rodents but can arise in human hosts.3 Children may be affected by C anthropophaga furuncular myiasis more often than adults because they have thinner skin and less immunity to the larvae.2
|
There are 2 mechanisms by which infestation of human hosts by C anthropophaga can occur. Most commonly, female flies lay eggs in shady areas in soil that is contaminated by feces or urine. The hatched larvae can survive in the ground for up to 2 weeks and later attach to a host when prompted by heat or movement.3 Therefore, clothing set out to dry may be contaminated by this soil. Alternatively, female flies can lay eggs directly onto clothing that is contaminated by feces or urine and the larvae subsequently hatch outside the soil with easy access to human skin once the clothing is worn.2
Common penetration sites are the head, neck, and back, as well as areas covered by contaminated or infested clothing.2,3 Penetration of the human skin occurs instantly and is a painless process that is rarely noticed by the human host.3 The larvae burrow into the skin for 8 to 12 days, resulting in a furuncle that occasionally secretes a serous fluid.2 Within the first 2 days of infestation, the host may experience symptoms ranging from local pruritus to severe pain. Six days following initial onset, an intense inflammatory response may result in local lymphadenopathy along with fever and fatigue.2 The larvae use their posterior spiracles to create openings in the skin to create air holes that allow them to breathe.3 On physical examination, the spiracles generally appear as 1- to 3-mm dark linear streaks within furuncles, which is important in the diagnosis of C anthropophaga furuncular myiasis.1,3 If spiracles are not appreciated on initial examination, diagnosis can be made by submerging the affected areas in water or saliva to look for air bubbles arising from the central puncta of the lesions.1
All causes of furuncular myiasis are characterized by a ratio of 1 larva to 1 furuncle.16 Although most of these types of larvae that can cause furuncular myiasis result in single lesions, C anthropophaga infestation often produces several furuncles that may coalesce into plaques.1,2 The differential diagnosis for C anthropophaga furuncular myiasis includes pyoderma, impetigo, staphylococcal furunculosis, cutaneous leishmaniasis, infected cyst, retained foreign body, and facticial disease.2,3 Dracunculiasis also may be considered, which occurs after ingestion of contaminated water.2 Ultrasonography may be helpful for the diagnosis of furuncular myiasis, as it can facilitate identification of foreign bodies, abscesses, and even larvae in some cases.17 Definitive diagnosis of any type of myiasis involves extraction of the larva and identification of the family, genus, and species by a parasitologist.1 Some experts suggest rearing preserved live larvae with raw meat after extraction because adult specimens are more reliable than larvae for species diagnosis.1
Treatment of furuncular myiasis involves occlusion and extraction of the larvae from the skin. Suffocation of the larvae by occlusion of air holes with petroleum jelly, paraffin oil, bacon fat, glue, and other obstructing substances forces the larvae to emerge in search of oxygen, though immature larvae may be more reluctant than mature ones.2,3 Definitive treatment involves the direct removal of the larvae by surgery or expulsion by pressure, though it is recommended that lesions are pretreated with occlusive techniques.1,3 Other reported methods of extraction include injection of lidocaine and the use of a commercial venom extractor.1 It should be noted that rupture and incomplete extraction of larvae can lead to secondary infections and allergic reactions. Lesions can be pretreated with lidocaine gel prior to extraction, and antibiotics should be used in cases of secondary bacterial infection. Ivermectin also has been reported as a treatment of furuncular myiasis and other types of myiasis.1 Prevention of infestation by C anthropophaga includes avoidance of endemic areas, maintaining good hygiene, and ironing clothing or drying it in sunny locations.1,2 Overall, furuncular myiasis has a good prognosis with rapid recovery and a low incidence of complications.1
Conclusion
We present 2 cases of travelers returning to North America from Senegal with C anthropophaga furuncular myiasis. Careful review of travel history, physical examination, and identification of fly larvae are important for diagnosis. Individuals traveling to sub-Saharan Africa should avoid drying clothes in shady places and lying on the ground. They also are urged to iron their clothing before wearing it.
1. Caissie R, Beaulieu F, Giroux M, et al. Cutaneous myiasis: diagnosis, treatment, and prevention. J Oral Maxillofac Surg. 2008;66:560-568.
2. McGraw TA, Turiansky GW. Cutaneous myiasis. J Am Acad Dermatol. 2008;58:907-926.
3. Robbins K, Khachemoune A. Cutaneous myiasis: a review of the common types of myiasis. Int J Dermatol. 2010;49:1092-1098.
4. Curtis SJ, Edwards C, Athulathmuda C, et al. Case of the month: cutaneous myiasis in a returning traveller from the Algarve: first report of tumbu maggots, Cordylobia anthropophaga, acquired in Portugal. Emerg Med J. 2006;23:236-237.
5. Veraldi S, Brusasco A, Süss L. Cutaneous myiasis caused by larvae of Cordylobia anthropophaga (Blanchard). Int J Dermatol. 1993;32:184-187.
6. Cultrera R, Dettori G, Calderaro A, et al. Cutaneous myiasis caused by Cordylobia anthropophaga (Blanchard 1872): description of 5 cases from costal regions of Senegal [in Italian]. Parassitologia. 1993;35:47-49.
7. Fusco FM, Nardiello S, Brancaccio G, et al. Cutaneous myiasis from Cordylobia anthropophaga in a traveller returning from Senegal: a case study [in Italian]. Infez Med. 2005;13:109-111.
8. Lee EJ, Robinson F. Furuncular myiasis of the face caused by larva of the tumbu fly (Cordylobia anthropophaga)[published online ahead of print July 21, 2006]. Eye (Lond). 2007;21:268-269.
9. Rice PL, Gleason N. Two cases of myiasis in the United States by the African tumbu fly, Cordylobia anthropophaga (Diptera, Calliphoridae). Am J Trop Med Hyg. 1972;21:62-65.
10. March CH. A case of “ver du Cayor” in Manhattan. Arch Dermatol. 1964;90:32-33.
11. Schorr WF. Tumbu-fly myiasis in Marshfield, Wis. Arch Dermatol. 1967;95:61-62.
12. Potter TS, Dorman MA, Ghaemi M, et al. Inflammatory papules on the back of a traveling businessman. tumbu
fly myiasis. Arch Dermatol. 1995;131:951, 954.
13. Ockenhouse CF, Samlaska CP, Benson PM, et al. Cutaneous myiasis caused by the African tumbu fly (Cordylobia anthropophaga). Arch Dermatol. 1990;126:199-202.
14. Kaouech E, Kallel K, Belhadj S, et al. Dermatobia hominis furuncular myiasis in a man returning from Latin America: first imported case in Tunisia [in French]. Med Trop (Mars). 2010;70:135-136.
15. Zayani A, Chaabouni M, Gouiaa R, et al. Conjuctival myiasis. 23 cases in the Tunisian Sahel [in French]. Arch Inst Pasteur Tunis. 1989;66:289-292.
16. Latorre M, Ullate JV, Sanchez J, et al. A case of myiasis due to Dermatobia hominis. Eur J Clin Microbiol Infect Dis. 1993;12:968-969.
17. Mahal JJ, Sperling JD. Furuncular myiasis from Dermatobia hominis: a case of human botfly infestation [published online ahead of print February 1, 2010]. J Emerg Med. 2012;43:618-621.
1. Caissie R, Beaulieu F, Giroux M, et al. Cutaneous myiasis: diagnosis, treatment, and prevention. J Oral Maxillofac Surg. 2008;66:560-568.
2. McGraw TA, Turiansky GW. Cutaneous myiasis. J Am Acad Dermatol. 2008;58:907-926.
3. Robbins K, Khachemoune A. Cutaneous myiasis: a review of the common types of myiasis. Int J Dermatol. 2010;49:1092-1098.
4. Curtis SJ, Edwards C, Athulathmuda C, et al. Case of the month: cutaneous myiasis in a returning traveller from the Algarve: first report of tumbu maggots, Cordylobia anthropophaga, acquired in Portugal. Emerg Med J. 2006;23:236-237.
5. Veraldi S, Brusasco A, Süss L. Cutaneous myiasis caused by larvae of Cordylobia anthropophaga (Blanchard). Int J Dermatol. 1993;32:184-187.
6. Cultrera R, Dettori G, Calderaro A, et al. Cutaneous myiasis caused by Cordylobia anthropophaga (Blanchard 1872): description of 5 cases from costal regions of Senegal [in Italian]. Parassitologia. 1993;35:47-49.
7. Fusco FM, Nardiello S, Brancaccio G, et al. Cutaneous myiasis from Cordylobia anthropophaga in a traveller returning from Senegal: a case study [in Italian]. Infez Med. 2005;13:109-111.
8. Lee EJ, Robinson F. Furuncular myiasis of the face caused by larva of the tumbu fly (Cordylobia anthropophaga)[published online ahead of print July 21, 2006]. Eye (Lond). 2007;21:268-269.
9. Rice PL, Gleason N. Two cases of myiasis in the United States by the African tumbu fly, Cordylobia anthropophaga (Diptera, Calliphoridae). Am J Trop Med Hyg. 1972;21:62-65.
10. March CH. A case of “ver du Cayor” in Manhattan. Arch Dermatol. 1964;90:32-33.
11. Schorr WF. Tumbu-fly myiasis in Marshfield, Wis. Arch Dermatol. 1967;95:61-62.
12. Potter TS, Dorman MA, Ghaemi M, et al. Inflammatory papules on the back of a traveling businessman. tumbu
fly myiasis. Arch Dermatol. 1995;131:951, 954.
13. Ockenhouse CF, Samlaska CP, Benson PM, et al. Cutaneous myiasis caused by the African tumbu fly (Cordylobia anthropophaga). Arch Dermatol. 1990;126:199-202.
14. Kaouech E, Kallel K, Belhadj S, et al. Dermatobia hominis furuncular myiasis in a man returning from Latin America: first imported case in Tunisia [in French]. Med Trop (Mars). 2010;70:135-136.
15. Zayani A, Chaabouni M, Gouiaa R, et al. Conjuctival myiasis. 23 cases in the Tunisian Sahel [in French]. Arch Inst Pasteur Tunis. 1989;66:289-292.
16. Latorre M, Ullate JV, Sanchez J, et al. A case of myiasis due to Dermatobia hominis. Eur J Clin Microbiol Infect Dis. 1993;12:968-969.
17. Mahal JJ, Sperling JD. Furuncular myiasis from Dermatobia hominis: a case of human botfly infestation [published online ahead of print February 1, 2010]. J Emerg Med. 2012;43:618-621.
Practice Points
- Cutaneous myiasis is caused by an infestation of fly larvae and can present as furuncles (furuncular myiasis), migratory inflammatory linear plaques (migratory myiasis), and worsening tissue destruction in existing wounds (wound myiasis).
- Furuncular myiasis should be included in the differential diagnosis in patients with furuncular skin lesions who have recently traveled to Central America, South America, or sub-Saharan Africa.
- Furuncular myiasis may be treated by both occlusive and extraction techniques.