User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
What do I have? How to tell patients you’re not sure
Physicians often struggle with telling patients when they are unsure about a diagnosis. In the absence of clarity, doctors may fear losing a patient’s trust by appearing unsure.
Yet diagnostic uncertainty is an inevitable part of medicine.
“It’s often uncertain what is really going on. People have lots of unspecific symptoms,” said Gordon D. Schiff, MD, a patient safety researcher at Harvard Medical School and Brigham and Women’s Hospital in Boston.
By one estimate, more than one-third of patients are discharged from an emergency department without a clear diagnosis. Physicians may order more tests to try to resolve uncertainty, but this method is not foolproof and may lead to increased health care costs. Physicians can use an uncertain diagnosis as an opportunity to improve conversations with patients, Dr. Schiff said.
“How do you talk to patients about that? How do you convey that?” Dr. Schiff asked.
To begin to answer these questions, The scenarios included an enlarged lymph node in a patient in remission for lymphoma, which could suggest recurrence of the disease but not necessarily; a patient with a new-onset headache; and another patient with an unexplained fever and a respiratory tract infection.
For each vignette, the researchers also asked patient advocates – many of whom had experienced receiving an incorrect diagnosis – for their thoughts on how the conversation should go.
Almost 70 people were consulted (24 primary care physicians, 40 patients, and five experts in informatics and quality and safety). Dr. Schiff and his colleagues produced six standardized elements that should be part of a conversation whenever a diagnosis is unclear.
- The most likely diagnosis, along with any alternatives if this isn’t certain, with phrases such as, “Sometimes we don’t have the answers, but we will keep trying to figure out what is going on.”
- Next steps – lab tests, return visits, etc.
- Expected time frame for patient’s improvement and recovery.
- Full disclosure of the limitations of the physical examination or any lab tests.
- Ways to contact the physician going forward.
- Patient insights on their experience and reaction to what they just heard.
The researchers, who published their findings in JAMA Network Open, recommend that the conversation be transcribed in real time using voice recognition software and a microphone, and then printed for the patient to take home. The physician should make eye contact with the patient during the conversation, they suggested.
“Patients felt it was a conversation, that they actually understood what was said. Most patients felt like they were partners during the encounter,” said Maram Khazen, PhD, a coauthor of the paper, who studies communication dynamics. Dr. Khazen was a visiting postdoctoral fellow with Dr. Schiff during the study, and is now a lecturer at the Max Stern Yezreel Valley College in Israel.
Hardeep Singh, MD, MPH, a patient safety researcher at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine in Houston, called the new work “a great start,” but said that the complexity of the field warrants more research into the tool. Dr. Singh was not involved in the study.
Dr. Singh pointed out that many of the patient voices came from spokespeople for advocacy groups, and that these participants are not necessarily representative of actual people with unclear diagnoses.
“The choice of words really matters,” said Dr. Singh, who led a 2018 study that showed that people reacted more negatively when physicians bluntly acknowledged uncertainty than when they walked patients through different possible diagnoses. Dr. Schiff and Dr. Khazen’s framework offers good principles for discussing uncertainty, he added, but further research is needed on the optimal language to use during conversations.
“It’s really encouraging that we’re seeing high-quality research like this, that leverages patient engagement principles,” said Dimitrios Papanagnou, MD, MPH, an emergency medicine physician and vice dean of medicine at Thomas Jefferson University in Philadelphia.
Dr. Papanagnou, who was not part of the study, called for diverse patients to be part of conversations about diagnostic uncertainty.
“Are we having patients from diverse experiences, from underrepresented groups, participate in this kind of work?” Dr. Papanagnou asked. Dr. Schiff and Dr. Khazen said they agree that the tool needs to be tested in larger samples of diverse patients.
Some common themes about how to communicate diagnostic uncertainty are emerging in multiple areas of medicine. Dr. Papanagnou helped develop an uncertainty communication checklist for discharging patients from an emergency department to home, with principles similar to those that Dr. Schiff and Dr. Khazen recommend for primary care providers.
The study was funded by Harvard Hospitals’ malpractice insurer, the Controlled Risk Insurance Company. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians often struggle with telling patients when they are unsure about a diagnosis. In the absence of clarity, doctors may fear losing a patient’s trust by appearing unsure.
Yet diagnostic uncertainty is an inevitable part of medicine.
“It’s often uncertain what is really going on. People have lots of unspecific symptoms,” said Gordon D. Schiff, MD, a patient safety researcher at Harvard Medical School and Brigham and Women’s Hospital in Boston.
By one estimate, more than one-third of patients are discharged from an emergency department without a clear diagnosis. Physicians may order more tests to try to resolve uncertainty, but this method is not foolproof and may lead to increased health care costs. Physicians can use an uncertain diagnosis as an opportunity to improve conversations with patients, Dr. Schiff said.
“How do you talk to patients about that? How do you convey that?” Dr. Schiff asked.
To begin to answer these questions, The scenarios included an enlarged lymph node in a patient in remission for lymphoma, which could suggest recurrence of the disease but not necessarily; a patient with a new-onset headache; and another patient with an unexplained fever and a respiratory tract infection.
For each vignette, the researchers also asked patient advocates – many of whom had experienced receiving an incorrect diagnosis – for their thoughts on how the conversation should go.
Almost 70 people were consulted (24 primary care physicians, 40 patients, and five experts in informatics and quality and safety). Dr. Schiff and his colleagues produced six standardized elements that should be part of a conversation whenever a diagnosis is unclear.
- The most likely diagnosis, along with any alternatives if this isn’t certain, with phrases such as, “Sometimes we don’t have the answers, but we will keep trying to figure out what is going on.”
- Next steps – lab tests, return visits, etc.
- Expected time frame for patient’s improvement and recovery.
- Full disclosure of the limitations of the physical examination or any lab tests.
- Ways to contact the physician going forward.
- Patient insights on their experience and reaction to what they just heard.
The researchers, who published their findings in JAMA Network Open, recommend that the conversation be transcribed in real time using voice recognition software and a microphone, and then printed for the patient to take home. The physician should make eye contact with the patient during the conversation, they suggested.
“Patients felt it was a conversation, that they actually understood what was said. Most patients felt like they were partners during the encounter,” said Maram Khazen, PhD, a coauthor of the paper, who studies communication dynamics. Dr. Khazen was a visiting postdoctoral fellow with Dr. Schiff during the study, and is now a lecturer at the Max Stern Yezreel Valley College in Israel.
Hardeep Singh, MD, MPH, a patient safety researcher at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine in Houston, called the new work “a great start,” but said that the complexity of the field warrants more research into the tool. Dr. Singh was not involved in the study.
Dr. Singh pointed out that many of the patient voices came from spokespeople for advocacy groups, and that these participants are not necessarily representative of actual people with unclear diagnoses.
“The choice of words really matters,” said Dr. Singh, who led a 2018 study that showed that people reacted more negatively when physicians bluntly acknowledged uncertainty than when they walked patients through different possible diagnoses. Dr. Schiff and Dr. Khazen’s framework offers good principles for discussing uncertainty, he added, but further research is needed on the optimal language to use during conversations.
“It’s really encouraging that we’re seeing high-quality research like this, that leverages patient engagement principles,” said Dimitrios Papanagnou, MD, MPH, an emergency medicine physician and vice dean of medicine at Thomas Jefferson University in Philadelphia.
Dr. Papanagnou, who was not part of the study, called for diverse patients to be part of conversations about diagnostic uncertainty.
“Are we having patients from diverse experiences, from underrepresented groups, participate in this kind of work?” Dr. Papanagnou asked. Dr. Schiff and Dr. Khazen said they agree that the tool needs to be tested in larger samples of diverse patients.
Some common themes about how to communicate diagnostic uncertainty are emerging in multiple areas of medicine. Dr. Papanagnou helped develop an uncertainty communication checklist for discharging patients from an emergency department to home, with principles similar to those that Dr. Schiff and Dr. Khazen recommend for primary care providers.
The study was funded by Harvard Hospitals’ malpractice insurer, the Controlled Risk Insurance Company. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physicians often struggle with telling patients when they are unsure about a diagnosis. In the absence of clarity, doctors may fear losing a patient’s trust by appearing unsure.
Yet diagnostic uncertainty is an inevitable part of medicine.
“It’s often uncertain what is really going on. People have lots of unspecific symptoms,” said Gordon D. Schiff, MD, a patient safety researcher at Harvard Medical School and Brigham and Women’s Hospital in Boston.
By one estimate, more than one-third of patients are discharged from an emergency department without a clear diagnosis. Physicians may order more tests to try to resolve uncertainty, but this method is not foolproof and may lead to increased health care costs. Physicians can use an uncertain diagnosis as an opportunity to improve conversations with patients, Dr. Schiff said.
“How do you talk to patients about that? How do you convey that?” Dr. Schiff asked.
To begin to answer these questions, The scenarios included an enlarged lymph node in a patient in remission for lymphoma, which could suggest recurrence of the disease but not necessarily; a patient with a new-onset headache; and another patient with an unexplained fever and a respiratory tract infection.
For each vignette, the researchers also asked patient advocates – many of whom had experienced receiving an incorrect diagnosis – for their thoughts on how the conversation should go.
Almost 70 people were consulted (24 primary care physicians, 40 patients, and five experts in informatics and quality and safety). Dr. Schiff and his colleagues produced six standardized elements that should be part of a conversation whenever a diagnosis is unclear.
- The most likely diagnosis, along with any alternatives if this isn’t certain, with phrases such as, “Sometimes we don’t have the answers, but we will keep trying to figure out what is going on.”
- Next steps – lab tests, return visits, etc.
- Expected time frame for patient’s improvement and recovery.
- Full disclosure of the limitations of the physical examination or any lab tests.
- Ways to contact the physician going forward.
- Patient insights on their experience and reaction to what they just heard.
The researchers, who published their findings in JAMA Network Open, recommend that the conversation be transcribed in real time using voice recognition software and a microphone, and then printed for the patient to take home. The physician should make eye contact with the patient during the conversation, they suggested.
“Patients felt it was a conversation, that they actually understood what was said. Most patients felt like they were partners during the encounter,” said Maram Khazen, PhD, a coauthor of the paper, who studies communication dynamics. Dr. Khazen was a visiting postdoctoral fellow with Dr. Schiff during the study, and is now a lecturer at the Max Stern Yezreel Valley College in Israel.
Hardeep Singh, MD, MPH, a patient safety researcher at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine in Houston, called the new work “a great start,” but said that the complexity of the field warrants more research into the tool. Dr. Singh was not involved in the study.
Dr. Singh pointed out that many of the patient voices came from spokespeople for advocacy groups, and that these participants are not necessarily representative of actual people with unclear diagnoses.
“The choice of words really matters,” said Dr. Singh, who led a 2018 study that showed that people reacted more negatively when physicians bluntly acknowledged uncertainty than when they walked patients through different possible diagnoses. Dr. Schiff and Dr. Khazen’s framework offers good principles for discussing uncertainty, he added, but further research is needed on the optimal language to use during conversations.
“It’s really encouraging that we’re seeing high-quality research like this, that leverages patient engagement principles,” said Dimitrios Papanagnou, MD, MPH, an emergency medicine physician and vice dean of medicine at Thomas Jefferson University in Philadelphia.
Dr. Papanagnou, who was not part of the study, called for diverse patients to be part of conversations about diagnostic uncertainty.
“Are we having patients from diverse experiences, from underrepresented groups, participate in this kind of work?” Dr. Papanagnou asked. Dr. Schiff and Dr. Khazen said they agree that the tool needs to be tested in larger samples of diverse patients.
Some common themes about how to communicate diagnostic uncertainty are emerging in multiple areas of medicine. Dr. Papanagnou helped develop an uncertainty communication checklist for discharging patients from an emergency department to home, with principles similar to those that Dr. Schiff and Dr. Khazen recommend for primary care providers.
The study was funded by Harvard Hospitals’ malpractice insurer, the Controlled Risk Insurance Company. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Spinosad: New kid on the block for treating scabies
HONOLULU – , Anthony J. Mancini, MD, said during a presentation at the Hawaii Dermatology Seminar provided by MedscapeLIVE!
In April 2021, spinosad topical suspension 0.9%, was approved by the Food and Drug Administration for treating scabies infestations in adult and pediatric patients 4 years of age and older – a first-in-class drug and the first new scabicide approved in 31 years. It was also approved for treating head lice in adults and children aged 6 months of age and older.
“Scabies has been described as the worst itch one can experience,” said Dr. Mancini, professor of pediatrics and dermatology at Northwestern University, Chicago. “It’s a hallmark of the disease, it can persist for weeks, it’s most intense at night, and patients report various sensations. It’s believed to be a both type I and type IV hypersensitivity reaction.”
The microscopic scabies mite burrows into the upper layer of the skin where it lives and lays its eggs. Besides intense itching, the classic presentation consists of a skin rash composed of inflammatory papules, linear burrows and crusted papules (especially on the hands, feet, and groin), and at times, larger red nodules. “Scabies nodules can persist for many months,” he said.
The Global Burden of Disease Study 2015 cited scabies as having the greatest burden of disease in tropical regions, especially among children, adolescents, and the elderly. The greatest burden of disability-adjusted life years (DALYs) occurred in East and Southeast Asia, Oceana, and tropical South America, but in North America, there was a 24% increase in the DALY rate between 1990 and 2015.
In addition, the World Health Organization designated scabies as a neglected tropical disease in 2017 and included it in its 10-year road map for neglected tropical diseases 2021-2030 with goals of promoting disease awareness and encouraging research and achieving global control.
“In our country, we typically see scabies treated successfully without complications, but there can be complications, especially in underdeveloped areas, like Staph aureus and Group A beta-hemolytic streptococcal infections,” which can be fatal, said Dr. Mancini, who is also head of pediatric dermatology at Ann & Robert H. Lurie Children’s Hospital of Chicago.
Permethrin 5% cream is typically offered as first-line topical therapy in the United States for the treatment of scabies. However, in vitro studies and small investigator-initiated in vivo studies have reported that efficacy appears to be decreasing. In one of the trials, Italian researchers enrolled 155 patients who were treated with permethrin 5% for 8 hours for 2 consecutive days and repeated the treatment 5 days later . Following the course of permethrin, only 34 responded, 96 failed treatment, and 25 were lost to follow-up.
“The study authors concluded that mite resistance to permethrin 5% seems to be increasing, following a path like other ectoparasite resistance,” said Dr. Mancini, who was not involved with the study. “We may even be seeing more ivermectin resistance in some geographic locations, as well.”
According to new scabicide efficacy criteria established by the FDA in 2016, complete cure is now defined as meeting both clinical and confirmatory criteria. A clinical cure means that all signs and symptoms of scabies have completely resolved, including burrows, inflammatory/noninflammatory lesions, and pruritus. A confirmatory cure means there is an absence of mites, eggs, scybala (feces), and burrows via microscopy or dermoscopy.
Enter spinosad, which is derived from a naturally occurring soil microorganism known as Saccharopolyspora spinosa and is composed of two active molecules: spinosyn A and spinosyn D. According to Dr. Mancini, spinosad’s mechanism of action is unique from other medications used to treat ectoparasites. It activates nicotinic and GABA-gated sodium channels, leads to sodium influx in the insect nerves, hyperexcitation, then paralysis and death. Cross-resistance to other insecticides has not been reported, he added, and there is no known evidence of resistance to its active compound.
Approval of the drug was based on data from two phase 3 randomized clinical trials involving 551 index cases and household contacts. In the intent-to-treat population, with the two trials combined, complete cure was achieved in 78.1% of the spinosad-treated group, compared with 39.6% in the vehicle group (P < .0001), clinical cure was achieved in 79.6% of the spinosad group, compared with 41.2% in the vehicle group (P < .001), and microscopic cure occurred in 85.9% of the spinosad group, compared with 52.6% in the vehicle group (P < .001).
Of the 306 participants in the study, the only adverse events reported by more than one patient each included abdominal pain, back pain, cough, headache, neck pain, and decreased weight in two patients each (0.8%), which investigators believed were not attributable to the study drug. Adverse events that investigators considered to be potentially related to the study drug included burning sensation in two participants (0.7%) and dry skin in another (0.3%). In clinical trials reported in the prescribing information, adverse events occurring in greater than 1% of subjects included application-site irritation (3% spinosad vs. 0% vehicle) and dry skin (2% spinosad vs. 0% vehicle).
“Spinosad met the FDA’s new stringent criteria, with all signs and symptoms of scabies completely resolved and confirmed via microscopy or dermoscopy,” said Dr. Mancini, who was not involved in the trials. “The patented formulation drives the active compound to the stratum corneum, where mites live and breed. It’s a single full-body application, without any resistance observed to date. This is an exciting newer option for treating our scabies patients.”
In an interview at the meeting, John S. Barbieri, MD, MBA, of the department of dermatology, Brigham and Women’s Hospital, Boston, said that, while he has no clinical experience with spinosad for scabies, he welcomes a new option for the condition. “The fact that it has a different mechanism of action than permethrin is a good thing,” he said.
Dr. Mancini disclosed that he is a consultant or an adviser for ParaPRO, the manufacturer of spinosad, and Cassiopea, Castle Creek, Novan, Novartis, and Verrica. He was not involved in clinical trials of spinosad. Dr. Barbieri disclosed that he receives consulting fees from Dexcel.
Medscape and this news organization are owned by the same parent company.
HONOLULU – , Anthony J. Mancini, MD, said during a presentation at the Hawaii Dermatology Seminar provided by MedscapeLIVE!
In April 2021, spinosad topical suspension 0.9%, was approved by the Food and Drug Administration for treating scabies infestations in adult and pediatric patients 4 years of age and older – a first-in-class drug and the first new scabicide approved in 31 years. It was also approved for treating head lice in adults and children aged 6 months of age and older.
“Scabies has been described as the worst itch one can experience,” said Dr. Mancini, professor of pediatrics and dermatology at Northwestern University, Chicago. “It’s a hallmark of the disease, it can persist for weeks, it’s most intense at night, and patients report various sensations. It’s believed to be a both type I and type IV hypersensitivity reaction.”
The microscopic scabies mite burrows into the upper layer of the skin where it lives and lays its eggs. Besides intense itching, the classic presentation consists of a skin rash composed of inflammatory papules, linear burrows and crusted papules (especially on the hands, feet, and groin), and at times, larger red nodules. “Scabies nodules can persist for many months,” he said.
The Global Burden of Disease Study 2015 cited scabies as having the greatest burden of disease in tropical regions, especially among children, adolescents, and the elderly. The greatest burden of disability-adjusted life years (DALYs) occurred in East and Southeast Asia, Oceana, and tropical South America, but in North America, there was a 24% increase in the DALY rate between 1990 and 2015.
In addition, the World Health Organization designated scabies as a neglected tropical disease in 2017 and included it in its 10-year road map for neglected tropical diseases 2021-2030 with goals of promoting disease awareness and encouraging research and achieving global control.
“In our country, we typically see scabies treated successfully without complications, but there can be complications, especially in underdeveloped areas, like Staph aureus and Group A beta-hemolytic streptococcal infections,” which can be fatal, said Dr. Mancini, who is also head of pediatric dermatology at Ann & Robert H. Lurie Children’s Hospital of Chicago.
Permethrin 5% cream is typically offered as first-line topical therapy in the United States for the treatment of scabies. However, in vitro studies and small investigator-initiated in vivo studies have reported that efficacy appears to be decreasing. In one of the trials, Italian researchers enrolled 155 patients who were treated with permethrin 5% for 8 hours for 2 consecutive days and repeated the treatment 5 days later . Following the course of permethrin, only 34 responded, 96 failed treatment, and 25 were lost to follow-up.
“The study authors concluded that mite resistance to permethrin 5% seems to be increasing, following a path like other ectoparasite resistance,” said Dr. Mancini, who was not involved with the study. “We may even be seeing more ivermectin resistance in some geographic locations, as well.”
According to new scabicide efficacy criteria established by the FDA in 2016, complete cure is now defined as meeting both clinical and confirmatory criteria. A clinical cure means that all signs and symptoms of scabies have completely resolved, including burrows, inflammatory/noninflammatory lesions, and pruritus. A confirmatory cure means there is an absence of mites, eggs, scybala (feces), and burrows via microscopy or dermoscopy.
Enter spinosad, which is derived from a naturally occurring soil microorganism known as Saccharopolyspora spinosa and is composed of two active molecules: spinosyn A and spinosyn D. According to Dr. Mancini, spinosad’s mechanism of action is unique from other medications used to treat ectoparasites. It activates nicotinic and GABA-gated sodium channels, leads to sodium influx in the insect nerves, hyperexcitation, then paralysis and death. Cross-resistance to other insecticides has not been reported, he added, and there is no known evidence of resistance to its active compound.
Approval of the drug was based on data from two phase 3 randomized clinical trials involving 551 index cases and household contacts. In the intent-to-treat population, with the two trials combined, complete cure was achieved in 78.1% of the spinosad-treated group, compared with 39.6% in the vehicle group (P < .0001), clinical cure was achieved in 79.6% of the spinosad group, compared with 41.2% in the vehicle group (P < .001), and microscopic cure occurred in 85.9% of the spinosad group, compared with 52.6% in the vehicle group (P < .001).
Of the 306 participants in the study, the only adverse events reported by more than one patient each included abdominal pain, back pain, cough, headache, neck pain, and decreased weight in two patients each (0.8%), which investigators believed were not attributable to the study drug. Adverse events that investigators considered to be potentially related to the study drug included burning sensation in two participants (0.7%) and dry skin in another (0.3%). In clinical trials reported in the prescribing information, adverse events occurring in greater than 1% of subjects included application-site irritation (3% spinosad vs. 0% vehicle) and dry skin (2% spinosad vs. 0% vehicle).
“Spinosad met the FDA’s new stringent criteria, with all signs and symptoms of scabies completely resolved and confirmed via microscopy or dermoscopy,” said Dr. Mancini, who was not involved in the trials. “The patented formulation drives the active compound to the stratum corneum, where mites live and breed. It’s a single full-body application, without any resistance observed to date. This is an exciting newer option for treating our scabies patients.”
In an interview at the meeting, John S. Barbieri, MD, MBA, of the department of dermatology, Brigham and Women’s Hospital, Boston, said that, while he has no clinical experience with spinosad for scabies, he welcomes a new option for the condition. “The fact that it has a different mechanism of action than permethrin is a good thing,” he said.
Dr. Mancini disclosed that he is a consultant or an adviser for ParaPRO, the manufacturer of spinosad, and Cassiopea, Castle Creek, Novan, Novartis, and Verrica. He was not involved in clinical trials of spinosad. Dr. Barbieri disclosed that he receives consulting fees from Dexcel.
Medscape and this news organization are owned by the same parent company.
HONOLULU – , Anthony J. Mancini, MD, said during a presentation at the Hawaii Dermatology Seminar provided by MedscapeLIVE!
In April 2021, spinosad topical suspension 0.9%, was approved by the Food and Drug Administration for treating scabies infestations in adult and pediatric patients 4 years of age and older – a first-in-class drug and the first new scabicide approved in 31 years. It was also approved for treating head lice in adults and children aged 6 months of age and older.
“Scabies has been described as the worst itch one can experience,” said Dr. Mancini, professor of pediatrics and dermatology at Northwestern University, Chicago. “It’s a hallmark of the disease, it can persist for weeks, it’s most intense at night, and patients report various sensations. It’s believed to be a both type I and type IV hypersensitivity reaction.”
The microscopic scabies mite burrows into the upper layer of the skin where it lives and lays its eggs. Besides intense itching, the classic presentation consists of a skin rash composed of inflammatory papules, linear burrows and crusted papules (especially on the hands, feet, and groin), and at times, larger red nodules. “Scabies nodules can persist for many months,” he said.
The Global Burden of Disease Study 2015 cited scabies as having the greatest burden of disease in tropical regions, especially among children, adolescents, and the elderly. The greatest burden of disability-adjusted life years (DALYs) occurred in East and Southeast Asia, Oceana, and tropical South America, but in North America, there was a 24% increase in the DALY rate between 1990 and 2015.
In addition, the World Health Organization designated scabies as a neglected tropical disease in 2017 and included it in its 10-year road map for neglected tropical diseases 2021-2030 with goals of promoting disease awareness and encouraging research and achieving global control.
“In our country, we typically see scabies treated successfully without complications, but there can be complications, especially in underdeveloped areas, like Staph aureus and Group A beta-hemolytic streptococcal infections,” which can be fatal, said Dr. Mancini, who is also head of pediatric dermatology at Ann & Robert H. Lurie Children’s Hospital of Chicago.
Permethrin 5% cream is typically offered as first-line topical therapy in the United States for the treatment of scabies. However, in vitro studies and small investigator-initiated in vivo studies have reported that efficacy appears to be decreasing. In one of the trials, Italian researchers enrolled 155 patients who were treated with permethrin 5% for 8 hours for 2 consecutive days and repeated the treatment 5 days later . Following the course of permethrin, only 34 responded, 96 failed treatment, and 25 were lost to follow-up.
“The study authors concluded that mite resistance to permethrin 5% seems to be increasing, following a path like other ectoparasite resistance,” said Dr. Mancini, who was not involved with the study. “We may even be seeing more ivermectin resistance in some geographic locations, as well.”
According to new scabicide efficacy criteria established by the FDA in 2016, complete cure is now defined as meeting both clinical and confirmatory criteria. A clinical cure means that all signs and symptoms of scabies have completely resolved, including burrows, inflammatory/noninflammatory lesions, and pruritus. A confirmatory cure means there is an absence of mites, eggs, scybala (feces), and burrows via microscopy or dermoscopy.
Enter spinosad, which is derived from a naturally occurring soil microorganism known as Saccharopolyspora spinosa and is composed of two active molecules: spinosyn A and spinosyn D. According to Dr. Mancini, spinosad’s mechanism of action is unique from other medications used to treat ectoparasites. It activates nicotinic and GABA-gated sodium channels, leads to sodium influx in the insect nerves, hyperexcitation, then paralysis and death. Cross-resistance to other insecticides has not been reported, he added, and there is no known evidence of resistance to its active compound.
Approval of the drug was based on data from two phase 3 randomized clinical trials involving 551 index cases and household contacts. In the intent-to-treat population, with the two trials combined, complete cure was achieved in 78.1% of the spinosad-treated group, compared with 39.6% in the vehicle group (P < .0001), clinical cure was achieved in 79.6% of the spinosad group, compared with 41.2% in the vehicle group (P < .001), and microscopic cure occurred in 85.9% of the spinosad group, compared with 52.6% in the vehicle group (P < .001).
Of the 306 participants in the study, the only adverse events reported by more than one patient each included abdominal pain, back pain, cough, headache, neck pain, and decreased weight in two patients each (0.8%), which investigators believed were not attributable to the study drug. Adverse events that investigators considered to be potentially related to the study drug included burning sensation in two participants (0.7%) and dry skin in another (0.3%). In clinical trials reported in the prescribing information, adverse events occurring in greater than 1% of subjects included application-site irritation (3% spinosad vs. 0% vehicle) and dry skin (2% spinosad vs. 0% vehicle).
“Spinosad met the FDA’s new stringent criteria, with all signs and symptoms of scabies completely resolved and confirmed via microscopy or dermoscopy,” said Dr. Mancini, who was not involved in the trials. “The patented formulation drives the active compound to the stratum corneum, where mites live and breed. It’s a single full-body application, without any resistance observed to date. This is an exciting newer option for treating our scabies patients.”
In an interview at the meeting, John S. Barbieri, MD, MBA, of the department of dermatology, Brigham and Women’s Hospital, Boston, said that, while he has no clinical experience with spinosad for scabies, he welcomes a new option for the condition. “The fact that it has a different mechanism of action than permethrin is a good thing,” he said.
Dr. Mancini disclosed that he is a consultant or an adviser for ParaPRO, the manufacturer of spinosad, and Cassiopea, Castle Creek, Novan, Novartis, and Verrica. He was not involved in clinical trials of spinosad. Dr. Barbieri disclosed that he receives consulting fees from Dexcel.
Medscape and this news organization are owned by the same parent company.
AT THE MEDSCAPELIVE! HAWAII DERMATOLOGY SEMINAR
Notes on direct admission of pediatric patients
Scenario: Yesterday you saw a 6-month-old infant with what appeared to be viral gastroenteritis and mild dehydration. When you called his parents today to check on his condition he was not improving despite your recommendations about his diet and oral rehydration. Should you have him brought to your office for a reevaluation, have his parents take him to the local emergency department for evaluation and probable hospital admission, or ask his parents to take him to the hospital telling them that you will call and arrange for a direct admission.
Obviously, I haven’t given you enough background information to allow you to give me an answer you are comfortable with. What time of day is it? Is it a holiday weekend? What’s the weather like? How far is it from the patient’s home to your office? To the emergency department? How is the local ED staffed? Are there hospitalists? What is their training?
Whether or not you choose to see the patient first in the office, is direct admission to the hospital an option that you are likely to choose? What steps do you take to see that it happens smoothly?
At least one-quarter of the unscheduled pediatric hospitalizations begin with a direct admission, meaning that the patients are not first evaluated in that hospital’s ED. In a recent policy statement, the American Academy of Pediatrics Committee on Hospital Care explored the pluses and minuses of direct admission and issued a list of seven recommendations. Among the concerns raised by the authors are “potential delays in initial evaluation and treatment, inconsistent admission processes, and difficulties in determining the appropriateness of patients for direct admission.” The committee makes it clear that they understand each community has it own strengths and challenges and the unique needs of each patient make it difficult to define a set of recommendations that fits all.
However, as I read through the committee’s seven recommendations, one leapt off the screen as a unifying concept that should apply in every situation. Recommendation No. 2 reads, “[There should be] clear systems of communication between members of the health care team and with families of children requiring admission.”
First, who is on this “health care team”? Are you a team member with the hospital folks – the ED nurses and doctors, the hospitalists, the floor nurses? Do you share an employer? Are you in the same town? Have your ever met them face to face? Do you do so regularly?
I assume you call the ED or the pediatric floor to arrange a direct admit? Maybe you don’t. I can recall working in situations where several infamous “local docs” would just send the patients in with a scribbled note (or not) and no phone call. Will you be speaking to folks who are even vaguely familiar with you or even your name? Do you get to speak with people who will be hands on with the patient?
Obviously, where I’m going with this is that, if you and the hospital staff are truly on the same health care team, communication should flow freely among the members and having some familiarity allows this to happen more smoothly. It can start on our end as the referring physician by making the call personally. Likewise, the receiving hospital must make frontline people available so you can speak with staff who will be working with the patient. Do you have enough information to tell the family what to expect?
Of course legible and complete records are a must. But nothing beats personal contact and a name. If you can tell a parent “I spoke to Martha, the nurse who will meet you on the floor,” that can be a giant first step forward in the healing process.
Most of us trained at hospitals that accepted direct admit patients and can remember the challenges. And most of us recall EDs that weren’t pediatric friendly. Whether our local situation favors direct admission or ED preadmission evaluation, it is our job to make the communication flow with the patient’s safety and the family’s comfort in mind.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Scenario: Yesterday you saw a 6-month-old infant with what appeared to be viral gastroenteritis and mild dehydration. When you called his parents today to check on his condition he was not improving despite your recommendations about his diet and oral rehydration. Should you have him brought to your office for a reevaluation, have his parents take him to the local emergency department for evaluation and probable hospital admission, or ask his parents to take him to the hospital telling them that you will call and arrange for a direct admission.
Obviously, I haven’t given you enough background information to allow you to give me an answer you are comfortable with. What time of day is it? Is it a holiday weekend? What’s the weather like? How far is it from the patient’s home to your office? To the emergency department? How is the local ED staffed? Are there hospitalists? What is their training?
Whether or not you choose to see the patient first in the office, is direct admission to the hospital an option that you are likely to choose? What steps do you take to see that it happens smoothly?
At least one-quarter of the unscheduled pediatric hospitalizations begin with a direct admission, meaning that the patients are not first evaluated in that hospital’s ED. In a recent policy statement, the American Academy of Pediatrics Committee on Hospital Care explored the pluses and minuses of direct admission and issued a list of seven recommendations. Among the concerns raised by the authors are “potential delays in initial evaluation and treatment, inconsistent admission processes, and difficulties in determining the appropriateness of patients for direct admission.” The committee makes it clear that they understand each community has it own strengths and challenges and the unique needs of each patient make it difficult to define a set of recommendations that fits all.
However, as I read through the committee’s seven recommendations, one leapt off the screen as a unifying concept that should apply in every situation. Recommendation No. 2 reads, “[There should be] clear systems of communication between members of the health care team and with families of children requiring admission.”
First, who is on this “health care team”? Are you a team member with the hospital folks – the ED nurses and doctors, the hospitalists, the floor nurses? Do you share an employer? Are you in the same town? Have your ever met them face to face? Do you do so regularly?
I assume you call the ED or the pediatric floor to arrange a direct admit? Maybe you don’t. I can recall working in situations where several infamous “local docs” would just send the patients in with a scribbled note (or not) and no phone call. Will you be speaking to folks who are even vaguely familiar with you or even your name? Do you get to speak with people who will be hands on with the patient?
Obviously, where I’m going with this is that, if you and the hospital staff are truly on the same health care team, communication should flow freely among the members and having some familiarity allows this to happen more smoothly. It can start on our end as the referring physician by making the call personally. Likewise, the receiving hospital must make frontline people available so you can speak with staff who will be working with the patient. Do you have enough information to tell the family what to expect?
Of course legible and complete records are a must. But nothing beats personal contact and a name. If you can tell a parent “I spoke to Martha, the nurse who will meet you on the floor,” that can be a giant first step forward in the healing process.
Most of us trained at hospitals that accepted direct admit patients and can remember the challenges. And most of us recall EDs that weren’t pediatric friendly. Whether our local situation favors direct admission or ED preadmission evaluation, it is our job to make the communication flow with the patient’s safety and the family’s comfort in mind.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Scenario: Yesterday you saw a 6-month-old infant with what appeared to be viral gastroenteritis and mild dehydration. When you called his parents today to check on his condition he was not improving despite your recommendations about his diet and oral rehydration. Should you have him brought to your office for a reevaluation, have his parents take him to the local emergency department for evaluation and probable hospital admission, or ask his parents to take him to the hospital telling them that you will call and arrange for a direct admission.
Obviously, I haven’t given you enough background information to allow you to give me an answer you are comfortable with. What time of day is it? Is it a holiday weekend? What’s the weather like? How far is it from the patient’s home to your office? To the emergency department? How is the local ED staffed? Are there hospitalists? What is their training?
Whether or not you choose to see the patient first in the office, is direct admission to the hospital an option that you are likely to choose? What steps do you take to see that it happens smoothly?
At least one-quarter of the unscheduled pediatric hospitalizations begin with a direct admission, meaning that the patients are not first evaluated in that hospital’s ED. In a recent policy statement, the American Academy of Pediatrics Committee on Hospital Care explored the pluses and minuses of direct admission and issued a list of seven recommendations. Among the concerns raised by the authors are “potential delays in initial evaluation and treatment, inconsistent admission processes, and difficulties in determining the appropriateness of patients for direct admission.” The committee makes it clear that they understand each community has it own strengths and challenges and the unique needs of each patient make it difficult to define a set of recommendations that fits all.
However, as I read through the committee’s seven recommendations, one leapt off the screen as a unifying concept that should apply in every situation. Recommendation No. 2 reads, “[There should be] clear systems of communication between members of the health care team and with families of children requiring admission.”
First, who is on this “health care team”? Are you a team member with the hospital folks – the ED nurses and doctors, the hospitalists, the floor nurses? Do you share an employer? Are you in the same town? Have your ever met them face to face? Do you do so regularly?
I assume you call the ED or the pediatric floor to arrange a direct admit? Maybe you don’t. I can recall working in situations where several infamous “local docs” would just send the patients in with a scribbled note (or not) and no phone call. Will you be speaking to folks who are even vaguely familiar with you or even your name? Do you get to speak with people who will be hands on with the patient?
Obviously, where I’m going with this is that, if you and the hospital staff are truly on the same health care team, communication should flow freely among the members and having some familiarity allows this to happen more smoothly. It can start on our end as the referring physician by making the call personally. Likewise, the receiving hospital must make frontline people available so you can speak with staff who will be working with the patient. Do you have enough information to tell the family what to expect?
Of course legible and complete records are a must. But nothing beats personal contact and a name. If you can tell a parent “I spoke to Martha, the nurse who will meet you on the floor,” that can be a giant first step forward in the healing process.
Most of us trained at hospitals that accepted direct admit patients and can remember the challenges. And most of us recall EDs that weren’t pediatric friendly. Whether our local situation favors direct admission or ED preadmission evaluation, it is our job to make the communication flow with the patient’s safety and the family’s comfort in mind.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Pandemic hit Black children harder, study shows
Black children had almost three times as many COVID-related deaths as White children and about twice as many hospitalizations, according to a new study.
The study said that 1,556 children have died from the start of the pandemic until Nov. 30, 2022, with 593 of those children being 4 and under. Black children died of COVID-related causes 2.7 times more often than White children and were hospitalized 2.2 times more often than White children, the study said.
Lower vaccination rates for Black people may be a factor. The study said 43.6% of White children have received two or more vaccinations, compared with 40.2% of Black children.
“First and foremost, this study repudiates the misunderstanding that COVID-19 has not been of consequence to children who have had more than 15.5 million reported cases, representing 18 percent of all cases in the United States,” Reed Tuckson, MD, a member of the Black Coalition Against COVID board of directors and former District of Columbia public health commissioner, said in a news release.
“And second, our research shows that like their adult counterparts, Black and other children of color have shouldered more of the burden of COVID-19 than the White population.”
The study was commissioned by BCAC and conducted by the Satcher Health Leadership Institute of the Morehouse School of Medicine, Atlanta. It’s based on studies conducted by other agencies over 2 years.
Black and Hispanic children also had more severe COVID cases, the study said. Among 281 pediatric patients in New York, New Jersey, and Connecticut, 23.3% of severe cases were Black and 51% of severe cases were Hispanic.
The study says 1 in 310 Black children lost a parent or caregiver to COVID between April 2020 and June 2012, compared with 1 in 738 White children.
Economic and health-related hardships were experienced by 31% of Black households, 29% of Latino households, and 16% of White households, the study said.
“Children with COVID-19 in communities of color were sicker, [were] hospitalized and died at higher rates than White children,” Sandra Harris-Hooker, the interim executive director at the Satcher Health Leadership Institute of Morehouse School, said in the release. “We can now fully understand the devastating impact the virus had on communities of color across generations.”
The study recommends several changes, such as modifying eligibility requirements for the Children’s Health Insurance Program to help more children who fall into coverage gaps and expanding the Child Tax Credit.
A version of this article first appeared on WebMD.com.
Black children had almost three times as many COVID-related deaths as White children and about twice as many hospitalizations, according to a new study.
The study said that 1,556 children have died from the start of the pandemic until Nov. 30, 2022, with 593 of those children being 4 and under. Black children died of COVID-related causes 2.7 times more often than White children and were hospitalized 2.2 times more often than White children, the study said.
Lower vaccination rates for Black people may be a factor. The study said 43.6% of White children have received two or more vaccinations, compared with 40.2% of Black children.
“First and foremost, this study repudiates the misunderstanding that COVID-19 has not been of consequence to children who have had more than 15.5 million reported cases, representing 18 percent of all cases in the United States,” Reed Tuckson, MD, a member of the Black Coalition Against COVID board of directors and former District of Columbia public health commissioner, said in a news release.
“And second, our research shows that like their adult counterparts, Black and other children of color have shouldered more of the burden of COVID-19 than the White population.”
The study was commissioned by BCAC and conducted by the Satcher Health Leadership Institute of the Morehouse School of Medicine, Atlanta. It’s based on studies conducted by other agencies over 2 years.
Black and Hispanic children also had more severe COVID cases, the study said. Among 281 pediatric patients in New York, New Jersey, and Connecticut, 23.3% of severe cases were Black and 51% of severe cases were Hispanic.
The study says 1 in 310 Black children lost a parent or caregiver to COVID between April 2020 and June 2012, compared with 1 in 738 White children.
Economic and health-related hardships were experienced by 31% of Black households, 29% of Latino households, and 16% of White households, the study said.
“Children with COVID-19 in communities of color were sicker, [were] hospitalized and died at higher rates than White children,” Sandra Harris-Hooker, the interim executive director at the Satcher Health Leadership Institute of Morehouse School, said in the release. “We can now fully understand the devastating impact the virus had on communities of color across generations.”
The study recommends several changes, such as modifying eligibility requirements for the Children’s Health Insurance Program to help more children who fall into coverage gaps and expanding the Child Tax Credit.
A version of this article first appeared on WebMD.com.
Black children had almost three times as many COVID-related deaths as White children and about twice as many hospitalizations, according to a new study.
The study said that 1,556 children have died from the start of the pandemic until Nov. 30, 2022, with 593 of those children being 4 and under. Black children died of COVID-related causes 2.7 times more often than White children and were hospitalized 2.2 times more often than White children, the study said.
Lower vaccination rates for Black people may be a factor. The study said 43.6% of White children have received two or more vaccinations, compared with 40.2% of Black children.
“First and foremost, this study repudiates the misunderstanding that COVID-19 has not been of consequence to children who have had more than 15.5 million reported cases, representing 18 percent of all cases in the United States,” Reed Tuckson, MD, a member of the Black Coalition Against COVID board of directors and former District of Columbia public health commissioner, said in a news release.
“And second, our research shows that like their adult counterparts, Black and other children of color have shouldered more of the burden of COVID-19 than the White population.”
The study was commissioned by BCAC and conducted by the Satcher Health Leadership Institute of the Morehouse School of Medicine, Atlanta. It’s based on studies conducted by other agencies over 2 years.
Black and Hispanic children also had more severe COVID cases, the study said. Among 281 pediatric patients in New York, New Jersey, and Connecticut, 23.3% of severe cases were Black and 51% of severe cases were Hispanic.
The study says 1 in 310 Black children lost a parent or caregiver to COVID between April 2020 and June 2012, compared with 1 in 738 White children.
Economic and health-related hardships were experienced by 31% of Black households, 29% of Latino households, and 16% of White households, the study said.
“Children with COVID-19 in communities of color were sicker, [were] hospitalized and died at higher rates than White children,” Sandra Harris-Hooker, the interim executive director at the Satcher Health Leadership Institute of Morehouse School, said in the release. “We can now fully understand the devastating impact the virus had on communities of color across generations.”
The study recommends several changes, such as modifying eligibility requirements for the Children’s Health Insurance Program to help more children who fall into coverage gaps and expanding the Child Tax Credit.
A version of this article first appeared on WebMD.com.
FDA OKs first drug for Rett syndrome
Rett syndrome is a rare, genetic neurodevelopmental disorder that affects about 6,000-9,000 people in the United States, mostly females.
Symptoms typically present between 6 and 18 months of age, with patients experiencing a rapid decline with loss of fine motor and communication skills.
Trofinetide is a synthetic analogue of the amino-terminal tripeptide of insulinlike growth factor-1 (IGF-1), which occurs naturally in the brain. The drug is designed to treat the core symptoms of Rett syndrome by potentially reducing neuroinflammation and supporting synaptic function.
The approval of trofinetide was supported by results from the pivotal phase 3 LAVENDER study that tested the efficacy and safety of trofinetide vs. placebo in 187 female patients with Rett syndrome, aged 5-20 years.
A total of 93 participants were randomly assigned to twice-daily oral trofinetide, and 94 received placebo for 12 weeks.
After 12 weeks, trofinetide showed a statistically significant improvement from baseline, compared with placebo, on both the caregiver-assessed Rett Syndrome Behavior Questionnaire (RSBQ) and 7-point Clinical Global Impression-Improvement (CGI-I) scale.
The drug also outperformed placebo at 12 weeks in a key secondary endpoint: the composite score on the Communication and Symbolic Behavior Scales Developmental Profile Infant-Toddler Checklist-Social (CSBS-DP-IT Social), a scale on which caregivers assess nonverbal communication.
The most common adverse events with trofinetide treatment were diarrhea and vomiting. Almost all these events were considered mild or moderate.
‘Historic day’
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” Melissa Kennedy, MHA, chief executive officer of the International Rett Syndrome Foundation, said in a news release issued by Acadia.
“Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones,” Ms. Kennedy said.
Trofinetide is expected to be available in the United States by the end of April.
A version of this article first appeared on Medscape.com.
Rett syndrome is a rare, genetic neurodevelopmental disorder that affects about 6,000-9,000 people in the United States, mostly females.
Symptoms typically present between 6 and 18 months of age, with patients experiencing a rapid decline with loss of fine motor and communication skills.
Trofinetide is a synthetic analogue of the amino-terminal tripeptide of insulinlike growth factor-1 (IGF-1), which occurs naturally in the brain. The drug is designed to treat the core symptoms of Rett syndrome by potentially reducing neuroinflammation and supporting synaptic function.
The approval of trofinetide was supported by results from the pivotal phase 3 LAVENDER study that tested the efficacy and safety of trofinetide vs. placebo in 187 female patients with Rett syndrome, aged 5-20 years.
A total of 93 participants were randomly assigned to twice-daily oral trofinetide, and 94 received placebo for 12 weeks.
After 12 weeks, trofinetide showed a statistically significant improvement from baseline, compared with placebo, on both the caregiver-assessed Rett Syndrome Behavior Questionnaire (RSBQ) and 7-point Clinical Global Impression-Improvement (CGI-I) scale.
The drug also outperformed placebo at 12 weeks in a key secondary endpoint: the composite score on the Communication and Symbolic Behavior Scales Developmental Profile Infant-Toddler Checklist-Social (CSBS-DP-IT Social), a scale on which caregivers assess nonverbal communication.
The most common adverse events with trofinetide treatment were diarrhea and vomiting. Almost all these events were considered mild or moderate.
‘Historic day’
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” Melissa Kennedy, MHA, chief executive officer of the International Rett Syndrome Foundation, said in a news release issued by Acadia.
“Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones,” Ms. Kennedy said.
Trofinetide is expected to be available in the United States by the end of April.
A version of this article first appeared on Medscape.com.
Rett syndrome is a rare, genetic neurodevelopmental disorder that affects about 6,000-9,000 people in the United States, mostly females.
Symptoms typically present between 6 and 18 months of age, with patients experiencing a rapid decline with loss of fine motor and communication skills.
Trofinetide is a synthetic analogue of the amino-terminal tripeptide of insulinlike growth factor-1 (IGF-1), which occurs naturally in the brain. The drug is designed to treat the core symptoms of Rett syndrome by potentially reducing neuroinflammation and supporting synaptic function.
The approval of trofinetide was supported by results from the pivotal phase 3 LAVENDER study that tested the efficacy and safety of trofinetide vs. placebo in 187 female patients with Rett syndrome, aged 5-20 years.
A total of 93 participants were randomly assigned to twice-daily oral trofinetide, and 94 received placebo for 12 weeks.
After 12 weeks, trofinetide showed a statistically significant improvement from baseline, compared with placebo, on both the caregiver-assessed Rett Syndrome Behavior Questionnaire (RSBQ) and 7-point Clinical Global Impression-Improvement (CGI-I) scale.
The drug also outperformed placebo at 12 weeks in a key secondary endpoint: the composite score on the Communication and Symbolic Behavior Scales Developmental Profile Infant-Toddler Checklist-Social (CSBS-DP-IT Social), a scale on which caregivers assess nonverbal communication.
The most common adverse events with trofinetide treatment were diarrhea and vomiting. Almost all these events were considered mild or moderate.
‘Historic day’
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” Melissa Kennedy, MHA, chief executive officer of the International Rett Syndrome Foundation, said in a news release issued by Acadia.
“Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones,” Ms. Kennedy said.
Trofinetide is expected to be available in the United States by the end of April.
A version of this article first appeared on Medscape.com.
Children and COVID: A look back as the fourth year begins
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?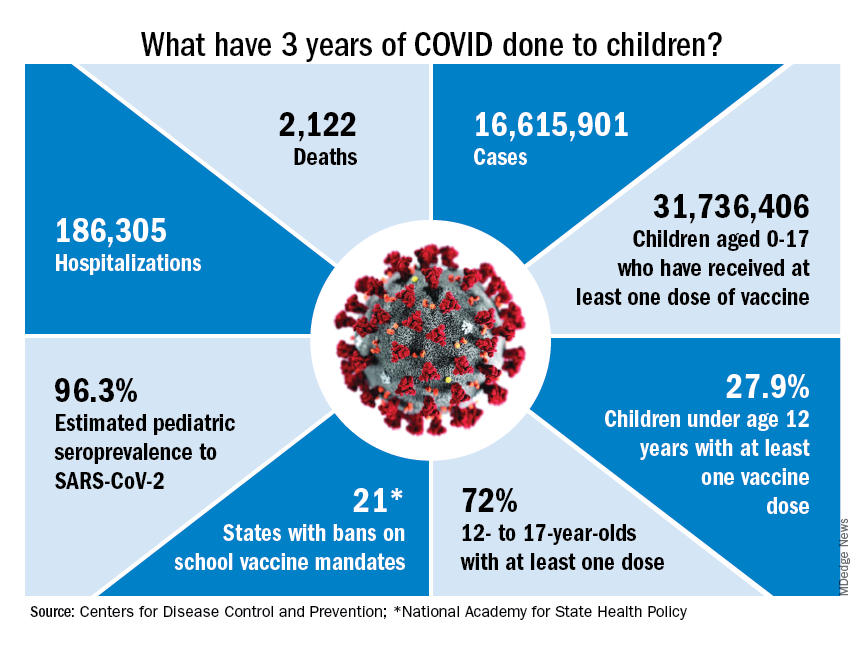
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
Will new guidelines widen the gap in treating childhood obesity?
In the United States, the Centers for Disease Control and Prevention estimates that nearly one in five children have obesity. Since the 1980s, the number of children with obesity has been increasing, with each generation reaching higher rates and greater weights at earlier ages. Even with extensive efforts from parents, clinicians, educators, and policymakers to limit the excessive weight gain among children, the number of obesity and severe obesity diagnoses keeps rising.
In response to this critical public health challenge, the American Academy of Pediatrics (AAP) introduced new clinical practice guidelines for the evaluation and management of obesity in children and adolescents. Developed by an expert panel, the new AAP guidelines present a departure in the conceptualization of obesity, recognizing the role that social determinants of health play in contributing to excessive weight gain.
As a community health researcher who investigates disparities in childhood obesity, I applaud the paradigm shift from the AAP. I specifically endorse the recognition that obesity is a very serious metabolic disease that won’t go away unless we introduce systemic changes and effective treatments.
However, I, like so many of my colleagues and anyone aware of the access barriers to the recommended treatments, worry about the consequences that the new guidelines will have in the context of current and future health disparities.
A recent study, published in Pediatrics, showed that childhood obesity disparities are widening. Younger generations of children are reaching higher weights at younger ages. These alarming trends are greater among Black children and children growing up with the greatest socioeconomic disadvantages. The new AAP guidelines – even while driven by good intentions – can exacerbate these differences and set children who are able to live healthy lives further apart from those with disproportionate obesity risks, who lack access to the treatments recommended by the AAP.
Rather than “watchful waiting,” to see if children outgrow obesity, the new guidelines call for “aggressive treatment,” as reported by this news organization. At least 26 hours of in-person intensive health behavior and lifestyle counseling and treatment are recommended for children aged 2 years old or older who meet the obesity criteria. For children aged 12 years or older, the AAP recommends complementing lifestyle counseling with pharmacotherapy. This breakthrough welcomes the use of promising antiobesity medications (for example, orlistat, Wegovy [semaglutide], Saxenda [liraglutide], Qsymia (phentermine and topiramate]) approved by the Food and Drug Administration for long-term use in children aged 12 and up. For children 13 years or older with severe obesity, bariatric surgery should be considered.
Will cost barriers continue to increase disparity?
The very promising semaglutide (Wegovy) is a GLP-1–based medication currently offered for about $1,000 per month. As with other chronic diseases, children should be prepared to take obesity medications for prolonged periods of time. A study conducted in adults found that when the medication is suspended, any weight loss can be regained. The costs of bariatric surgery total over $20,000.
In the U.S. health care system, at current prices, very few of the children in need of the medications or surgical treatments have access to them. Most private health insurance companies and Medicaid reject coverage for childhood obesity treatments. Barriers to treatment access are greater for Black and Hispanic children, children growing up in poverty, and children living in the U.S. South region, all of whom are more likely to develop obesity earlier in life than their White and wealthier counterparts.
The AAP recognized that a substantial time and financial commitment is required to follow the new treatment recommendations. Members of the AAP Expert Committee that developed the guidelines stated that they are “aware of the multitude of barriers to treatment that patients and their families face.”
Nevertheless, the recognition of the role of social determinants of health in the development of childhood obesity didn’t motivate the introduction of treatment options that aren’t unattainable for most U.S. families.
It’s important to step away from the conclusion that because of the price tag, at the population level, the new AAP guidelines will be inconsequential. This conclusion fails to recognize the potential harm that the guidelines may introduce. In the context of childhood obesity disparities, the new treatment recommendations probably will widen the childhood obesity prevalence gap between the haves – who will benefit from the options available to reduce childhood obesity – and the have-nots, whose obesity rates will continue with their growth.
We live in a world of the haves and have-nots. This applies to financial resources as well as obesity rates. In the long term, the optimists hope that the GLP-1 medications will become ubiquitous, generics will be developed, and insurance companies will expand coverage and grant access to most children in need of effective obesity treatment options. Until this happens, unless intentional policies are promptly introduced, childhood obesity disparities will continue to widen.
To avoid the increasing disparities, brave and intentional actions are required. A lack of attention dealt to this known problem will result in a lost opportunity for the AAP, legislators, and others in a position to help U.S. children.
Liliana Aguayo, PhD, MPH, is assistant professor, Clinical Research Track, Hubert Department of Global Health, Emory University, Atlanta. A version of this article first appeared on Medscape.com.
In the United States, the Centers for Disease Control and Prevention estimates that nearly one in five children have obesity. Since the 1980s, the number of children with obesity has been increasing, with each generation reaching higher rates and greater weights at earlier ages. Even with extensive efforts from parents, clinicians, educators, and policymakers to limit the excessive weight gain among children, the number of obesity and severe obesity diagnoses keeps rising.
In response to this critical public health challenge, the American Academy of Pediatrics (AAP) introduced new clinical practice guidelines for the evaluation and management of obesity in children and adolescents. Developed by an expert panel, the new AAP guidelines present a departure in the conceptualization of obesity, recognizing the role that social determinants of health play in contributing to excessive weight gain.
As a community health researcher who investigates disparities in childhood obesity, I applaud the paradigm shift from the AAP. I specifically endorse the recognition that obesity is a very serious metabolic disease that won’t go away unless we introduce systemic changes and effective treatments.
However, I, like so many of my colleagues and anyone aware of the access barriers to the recommended treatments, worry about the consequences that the new guidelines will have in the context of current and future health disparities.
A recent study, published in Pediatrics, showed that childhood obesity disparities are widening. Younger generations of children are reaching higher weights at younger ages. These alarming trends are greater among Black children and children growing up with the greatest socioeconomic disadvantages. The new AAP guidelines – even while driven by good intentions – can exacerbate these differences and set children who are able to live healthy lives further apart from those with disproportionate obesity risks, who lack access to the treatments recommended by the AAP.
Rather than “watchful waiting,” to see if children outgrow obesity, the new guidelines call for “aggressive treatment,” as reported by this news organization. At least 26 hours of in-person intensive health behavior and lifestyle counseling and treatment are recommended for children aged 2 years old or older who meet the obesity criteria. For children aged 12 years or older, the AAP recommends complementing lifestyle counseling with pharmacotherapy. This breakthrough welcomes the use of promising antiobesity medications (for example, orlistat, Wegovy [semaglutide], Saxenda [liraglutide], Qsymia (phentermine and topiramate]) approved by the Food and Drug Administration for long-term use in children aged 12 and up. For children 13 years or older with severe obesity, bariatric surgery should be considered.
Will cost barriers continue to increase disparity?
The very promising semaglutide (Wegovy) is a GLP-1–based medication currently offered for about $1,000 per month. As with other chronic diseases, children should be prepared to take obesity medications for prolonged periods of time. A study conducted in adults found that when the medication is suspended, any weight loss can be regained. The costs of bariatric surgery total over $20,000.
In the U.S. health care system, at current prices, very few of the children in need of the medications or surgical treatments have access to them. Most private health insurance companies and Medicaid reject coverage for childhood obesity treatments. Barriers to treatment access are greater for Black and Hispanic children, children growing up in poverty, and children living in the U.S. South region, all of whom are more likely to develop obesity earlier in life than their White and wealthier counterparts.
The AAP recognized that a substantial time and financial commitment is required to follow the new treatment recommendations. Members of the AAP Expert Committee that developed the guidelines stated that they are “aware of the multitude of barriers to treatment that patients and their families face.”
Nevertheless, the recognition of the role of social determinants of health in the development of childhood obesity didn’t motivate the introduction of treatment options that aren’t unattainable for most U.S. families.
It’s important to step away from the conclusion that because of the price tag, at the population level, the new AAP guidelines will be inconsequential. This conclusion fails to recognize the potential harm that the guidelines may introduce. In the context of childhood obesity disparities, the new treatment recommendations probably will widen the childhood obesity prevalence gap between the haves – who will benefit from the options available to reduce childhood obesity – and the have-nots, whose obesity rates will continue with their growth.
We live in a world of the haves and have-nots. This applies to financial resources as well as obesity rates. In the long term, the optimists hope that the GLP-1 medications will become ubiquitous, generics will be developed, and insurance companies will expand coverage and grant access to most children in need of effective obesity treatment options. Until this happens, unless intentional policies are promptly introduced, childhood obesity disparities will continue to widen.
To avoid the increasing disparities, brave and intentional actions are required. A lack of attention dealt to this known problem will result in a lost opportunity for the AAP, legislators, and others in a position to help U.S. children.
Liliana Aguayo, PhD, MPH, is assistant professor, Clinical Research Track, Hubert Department of Global Health, Emory University, Atlanta. A version of this article first appeared on Medscape.com.
In the United States, the Centers for Disease Control and Prevention estimates that nearly one in five children have obesity. Since the 1980s, the number of children with obesity has been increasing, with each generation reaching higher rates and greater weights at earlier ages. Even with extensive efforts from parents, clinicians, educators, and policymakers to limit the excessive weight gain among children, the number of obesity and severe obesity diagnoses keeps rising.
In response to this critical public health challenge, the American Academy of Pediatrics (AAP) introduced new clinical practice guidelines for the evaluation and management of obesity in children and adolescents. Developed by an expert panel, the new AAP guidelines present a departure in the conceptualization of obesity, recognizing the role that social determinants of health play in contributing to excessive weight gain.
As a community health researcher who investigates disparities in childhood obesity, I applaud the paradigm shift from the AAP. I specifically endorse the recognition that obesity is a very serious metabolic disease that won’t go away unless we introduce systemic changes and effective treatments.
However, I, like so many of my colleagues and anyone aware of the access barriers to the recommended treatments, worry about the consequences that the new guidelines will have in the context of current and future health disparities.
A recent study, published in Pediatrics, showed that childhood obesity disparities are widening. Younger generations of children are reaching higher weights at younger ages. These alarming trends are greater among Black children and children growing up with the greatest socioeconomic disadvantages. The new AAP guidelines – even while driven by good intentions – can exacerbate these differences and set children who are able to live healthy lives further apart from those with disproportionate obesity risks, who lack access to the treatments recommended by the AAP.
Rather than “watchful waiting,” to see if children outgrow obesity, the new guidelines call for “aggressive treatment,” as reported by this news organization. At least 26 hours of in-person intensive health behavior and lifestyle counseling and treatment are recommended for children aged 2 years old or older who meet the obesity criteria. For children aged 12 years or older, the AAP recommends complementing lifestyle counseling with pharmacotherapy. This breakthrough welcomes the use of promising antiobesity medications (for example, orlistat, Wegovy [semaglutide], Saxenda [liraglutide], Qsymia (phentermine and topiramate]) approved by the Food and Drug Administration for long-term use in children aged 12 and up. For children 13 years or older with severe obesity, bariatric surgery should be considered.
Will cost barriers continue to increase disparity?
The very promising semaglutide (Wegovy) is a GLP-1–based medication currently offered for about $1,000 per month. As with other chronic diseases, children should be prepared to take obesity medications for prolonged periods of time. A study conducted in adults found that when the medication is suspended, any weight loss can be regained. The costs of bariatric surgery total over $20,000.
In the U.S. health care system, at current prices, very few of the children in need of the medications or surgical treatments have access to them. Most private health insurance companies and Medicaid reject coverage for childhood obesity treatments. Barriers to treatment access are greater for Black and Hispanic children, children growing up in poverty, and children living in the U.S. South region, all of whom are more likely to develop obesity earlier in life than their White and wealthier counterparts.
The AAP recognized that a substantial time and financial commitment is required to follow the new treatment recommendations. Members of the AAP Expert Committee that developed the guidelines stated that they are “aware of the multitude of barriers to treatment that patients and their families face.”
Nevertheless, the recognition of the role of social determinants of health in the development of childhood obesity didn’t motivate the introduction of treatment options that aren’t unattainable for most U.S. families.
It’s important to step away from the conclusion that because of the price tag, at the population level, the new AAP guidelines will be inconsequential. This conclusion fails to recognize the potential harm that the guidelines may introduce. In the context of childhood obesity disparities, the new treatment recommendations probably will widen the childhood obesity prevalence gap between the haves – who will benefit from the options available to reduce childhood obesity – and the have-nots, whose obesity rates will continue with their growth.
We live in a world of the haves and have-nots. This applies to financial resources as well as obesity rates. In the long term, the optimists hope that the GLP-1 medications will become ubiquitous, generics will be developed, and insurance companies will expand coverage and grant access to most children in need of effective obesity treatment options. Until this happens, unless intentional policies are promptly introduced, childhood obesity disparities will continue to widen.
To avoid the increasing disparities, brave and intentional actions are required. A lack of attention dealt to this known problem will result in a lost opportunity for the AAP, legislators, and others in a position to help U.S. children.
Liliana Aguayo, PhD, MPH, is assistant professor, Clinical Research Track, Hubert Department of Global Health, Emory University, Atlanta. A version of this article first appeared on Medscape.com.
Firing patients
One might assume that, just as patients are free to accept or reject their doctors, physicians have an equal right to reject their patients; to a certain extent, that is true. There are no specific laws prohibiting a provider from terminating a patient relationship for any reason, other than a discriminatory one – race, nationality, religion, age, gender, sexual orientation, and so on. However, the evolution of ever-larger practice environments has raised new questions.
While verbal abuse, inappropriate treatment demands (particularly for controlled substances), refusal to adhere to mutually agreed treatment plans, and failure to keep appointments or pay bills remain the most common reasons for dismissal, evolving practice environments may require us to modify our responses.
What happens, for example, when a patient is banned from a large clinic that employs most of that community’s physicians, or is the only practice in town with the specialists required by that patient? The medical profession does have an obligation to not exclude such patients from care.
In a large cross-specialty system or consolidated specialist practice, firing a patient has a very different level of consequences than in a small office. There must be a balance between separating patients and doctors who don’t get along and seeing that the patient in question receives competent treatment. The physician, as the professional, has a higher standard to live up to with respect to handling this kind of situation.
If the problem is a personality conflict, the solution may be as simple as transferring the patient to another caregiver within the practice. While it does not make sense for a patient to continue seeing a doctor who does not want to see them, it also does not make sense to ban a patient from a large system where there could well be one or more other doctors who would be a good match. If a patient is unable to pay outstanding bills, a large clinic might prohibit them from making new appointments until they have worked out a payment plan rather than firing them outright.
If you are part of a large practice, take the time to research your group’s official policies for dealing with such situations. If there is no written policy, you might want to start that discussion with your colleagues.
The point is that in any practice, large or small, firing a patient should be a last resort. Try to make every effort to resolve the problem amicably. Communicate with the patient in question, explain your concerns, and discuss options for resolution. Take time to listen to the patient, as they may have an explanation (rational or not) for their objectionable behavior.
You can also send a letter, repeating your concerns and proposed solutions, as further documentation of your efforts to achieve an amicable resolution. All verbal and written warnings must, of course, be documented. If the patient has a managed care policy, we review the managed care contract, which sometimes includes specific requirements for dismissal of its patients.
When such efforts fail, we send the patient two letters – one certified with return receipt, the other by conventional first class, in case the patient refuses the certified copy – explaining the reason for dismissal, and that care will be discontinued in 30 days from the letter’s date. (Most attorneys and medical associations agree that 30 days is sufficient reasonable notice.) We offer to provide care during the interim period, include a list of names and contact information for potential alternate providers, and offer to transfer records after receiving written permission.
Following these precautions will usually protect you from charges of “patient abandonment,” which is generally defined as the unilateral severance by the physician of the physician-patient relationship without giving the patient sufficient advance notice to obtain the services of another practitioner, and at a time when the patient still requires medical attention.
Some states have their own unique definitions of patient abandonment. You should check with your state’s health department, and your attorney, for any unusual requirements in your state, because violating them could lead to intervention by your state licensing board. There is also the risk of civil litigation, which is typically not covered by malpractice policies, and may not be covered by your general liability policy either.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
One might assume that, just as patients are free to accept or reject their doctors, physicians have an equal right to reject their patients; to a certain extent, that is true. There are no specific laws prohibiting a provider from terminating a patient relationship for any reason, other than a discriminatory one – race, nationality, religion, age, gender, sexual orientation, and so on. However, the evolution of ever-larger practice environments has raised new questions.
While verbal abuse, inappropriate treatment demands (particularly for controlled substances), refusal to adhere to mutually agreed treatment plans, and failure to keep appointments or pay bills remain the most common reasons for dismissal, evolving practice environments may require us to modify our responses.
What happens, for example, when a patient is banned from a large clinic that employs most of that community’s physicians, or is the only practice in town with the specialists required by that patient? The medical profession does have an obligation to not exclude such patients from care.
In a large cross-specialty system or consolidated specialist practice, firing a patient has a very different level of consequences than in a small office. There must be a balance between separating patients and doctors who don’t get along and seeing that the patient in question receives competent treatment. The physician, as the professional, has a higher standard to live up to with respect to handling this kind of situation.
If the problem is a personality conflict, the solution may be as simple as transferring the patient to another caregiver within the practice. While it does not make sense for a patient to continue seeing a doctor who does not want to see them, it also does not make sense to ban a patient from a large system where there could well be one or more other doctors who would be a good match. If a patient is unable to pay outstanding bills, a large clinic might prohibit them from making new appointments until they have worked out a payment plan rather than firing them outright.
If you are part of a large practice, take the time to research your group’s official policies for dealing with such situations. If there is no written policy, you might want to start that discussion with your colleagues.
The point is that in any practice, large or small, firing a patient should be a last resort. Try to make every effort to resolve the problem amicably. Communicate with the patient in question, explain your concerns, and discuss options for resolution. Take time to listen to the patient, as they may have an explanation (rational or not) for their objectionable behavior.
You can also send a letter, repeating your concerns and proposed solutions, as further documentation of your efforts to achieve an amicable resolution. All verbal and written warnings must, of course, be documented. If the patient has a managed care policy, we review the managed care contract, which sometimes includes specific requirements for dismissal of its patients.
When such efforts fail, we send the patient two letters – one certified with return receipt, the other by conventional first class, in case the patient refuses the certified copy – explaining the reason for dismissal, and that care will be discontinued in 30 days from the letter’s date. (Most attorneys and medical associations agree that 30 days is sufficient reasonable notice.) We offer to provide care during the interim period, include a list of names and contact information for potential alternate providers, and offer to transfer records after receiving written permission.
Following these precautions will usually protect you from charges of “patient abandonment,” which is generally defined as the unilateral severance by the physician of the physician-patient relationship without giving the patient sufficient advance notice to obtain the services of another practitioner, and at a time when the patient still requires medical attention.
Some states have their own unique definitions of patient abandonment. You should check with your state’s health department, and your attorney, for any unusual requirements in your state, because violating them could lead to intervention by your state licensing board. There is also the risk of civil litigation, which is typically not covered by malpractice policies, and may not be covered by your general liability policy either.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
One might assume that, just as patients are free to accept or reject their doctors, physicians have an equal right to reject their patients; to a certain extent, that is true. There are no specific laws prohibiting a provider from terminating a patient relationship for any reason, other than a discriminatory one – race, nationality, religion, age, gender, sexual orientation, and so on. However, the evolution of ever-larger practice environments has raised new questions.
While verbal abuse, inappropriate treatment demands (particularly for controlled substances), refusal to adhere to mutually agreed treatment plans, and failure to keep appointments or pay bills remain the most common reasons for dismissal, evolving practice environments may require us to modify our responses.
What happens, for example, when a patient is banned from a large clinic that employs most of that community’s physicians, or is the only practice in town with the specialists required by that patient? The medical profession does have an obligation to not exclude such patients from care.
In a large cross-specialty system or consolidated specialist practice, firing a patient has a very different level of consequences than in a small office. There must be a balance between separating patients and doctors who don’t get along and seeing that the patient in question receives competent treatment. The physician, as the professional, has a higher standard to live up to with respect to handling this kind of situation.
If the problem is a personality conflict, the solution may be as simple as transferring the patient to another caregiver within the practice. While it does not make sense for a patient to continue seeing a doctor who does not want to see them, it also does not make sense to ban a patient from a large system where there could well be one or more other doctors who would be a good match. If a patient is unable to pay outstanding bills, a large clinic might prohibit them from making new appointments until they have worked out a payment plan rather than firing them outright.
If you are part of a large practice, take the time to research your group’s official policies for dealing with such situations. If there is no written policy, you might want to start that discussion with your colleagues.
The point is that in any practice, large or small, firing a patient should be a last resort. Try to make every effort to resolve the problem amicably. Communicate with the patient in question, explain your concerns, and discuss options for resolution. Take time to listen to the patient, as they may have an explanation (rational or not) for their objectionable behavior.
You can also send a letter, repeating your concerns and proposed solutions, as further documentation of your efforts to achieve an amicable resolution. All verbal and written warnings must, of course, be documented. If the patient has a managed care policy, we review the managed care contract, which sometimes includes specific requirements for dismissal of its patients.
When such efforts fail, we send the patient two letters – one certified with return receipt, the other by conventional first class, in case the patient refuses the certified copy – explaining the reason for dismissal, and that care will be discontinued in 30 days from the letter’s date. (Most attorneys and medical associations agree that 30 days is sufficient reasonable notice.) We offer to provide care during the interim period, include a list of names and contact information for potential alternate providers, and offer to transfer records after receiving written permission.
Following these precautions will usually protect you from charges of “patient abandonment,” which is generally defined as the unilateral severance by the physician of the physician-patient relationship without giving the patient sufficient advance notice to obtain the services of another practitioner, and at a time when the patient still requires medical attention.
Some states have their own unique definitions of patient abandonment. You should check with your state’s health department, and your attorney, for any unusual requirements in your state, because violating them could lead to intervention by your state licensing board. There is also the risk of civil litigation, which is typically not covered by malpractice policies, and may not be covered by your general liability policy either.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Factors linked with increased VTE risk in COVID outpatients
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
Though VTE risk is well studied and significant in those hospitalized with COVID, little is known about the risk in the outpatient setting, said the authors of the new research published online in JAMA Network Open.
The study was conducted at two integrated health care delivery systems in northern and southern California. Data were gathered from the Kaiser Permanente Virtual Data Warehouse and electronic health records.
Nearly 400,000 patients studied
Researchers, led by Margaret Fang, MD, with the division of hospital medicine, University of California, San Francisco, identified 398,530 outpatients with COVID-19 from Jan. 1, 2020, through Jan. 31, 2021.
VTE risk was low overall for ambulatory COVID patients.
“It is a reassuring study,” Dr. Fang said in an interview.
The researchers found that the risk is highest in the first 30 days after COVID-19 diagnosis (unadjusted rate, 0.58; 95% confidence interval, 0.51-0.67 per 100 person-years vs. 0.09; 95% CI, 0.08-0.11 per 100 person-years after 30 days).
Factors linked with high VTE risk
They also found that several factors were linked with a higher risk of blood clots in the study population, including being at least 55 years old; being male; having a history of blood clots or thrombophilia; and a body mass index (BMI) of at least 30 kg/m2.
The authors write, “These findings may help identify subsets of patients with COVID-19 who could benefit from VTE preventive strategies and more intensive short-term surveillance.”
Are routine anticoagulants justified?
Previously, randomized clinical trials have found that hospitalized patients with moderate COVID-19 may benefit from therapeutically dosed heparin anticoagulants but that therapeutic anticoagulation had no net benefit – and perhaps could even harm – patients who were critically ill with COVID.
“[M]uch less is known about the optimal thromboprophylaxis strategy for people with milder presentations of COVID-19 who do not require hospitalization,” they write.
Mild COVID VTE risk similar to general population
The authors note that rates of blood clots linked with COVID-19 are not much higher than the average blood clot rate in the general population, which is about 0.1-0.2 per 100 person-years.
Therefore, the results don’t justify routine administration of anticoagulation given the costs, inconvenience, and bleeding risks, they acknowledge.
Dr. Fang told this publication that it’s hard to know what to tell patients, given the overall low VTE risk. She said their study wasn’t designed to advise when to give prophylaxis.
Physicians should inform patients of their higher risk
“We should tell our patients who fall into these risk categories that blood clot is a concern after the development of COVID, especially in those first 30 days. And some people might benefit from increased surveillance,” Dr. Fang said.
”I think this study would support ongoing studies that look at whether selected patients benefit from VTE prophylaxis, for example low-dose anticoagulants,” she said.
Dr. Fang said the subgroup factors they found increased risk of blood clots for all patients, not just COVID-19 patients. It’s not clear why factors such as being male may increase blood clot risk, though that is consistent with previous literature, but higher risk with higher BMI might be related to a combination of inflammation or decreased mobility, she said.
Unanswered questions
Robert H. Hopkins Jr., MD, says the study helps answer a couple of important questions – that the VTE risk in nonhospitalized COVID-19 patients is low and when and for which patients risk may be highest.
However, there are several unanswered questions that argue against routine initiation of anticoagulants, notes the professor of internal medicine and pediatrics chief, division of general internal medicine, at University of Arkansas for Medical Sciences, Little Rock.
One is the change in the COVID variant landscape.
“We do not know whether rates of VTE are same or lower or higher with current circulating variants,” Dr. Hopkins said.
The authors acknowledge this as a limitation. Study data predate Omicron and subvariants, which appear to lower clinical severity, so it’s unclear whether VTE risk is different in this Omicron era.
Dr. Hopkins added another unknown: “We do not know whether vaccination affects rates of VTE in ambulatory breakthrough infection.”
Dr. Hopkins and the authors also note the lack of a control group in the study, to better compare risk.
Coauthor Dr. Prasad reports consultant fees from EpiExcellence LLC outside the submitted work. Coauthor Dr. Go reports grants paid to the division of research, Kaiser Permanente Northern California, from CSL Behring, Novartis, Bristol Meyers Squibb/Pfizer Alliance, and Janssen outside the submitted work.
The research was funded through Patient-Centered Outcomes Research Institute.
Dr. Hopkins reports no relevant financial relationships.
FROM JAMA NETWORK OPEN
Can particles in dairy and beef cause cancer and MS?
In Western diets, dairy and beef are ubiquitous: Milk goes with coffee, melted cheese with pizza, and chili with rice. But what if dairy products and beef contained a new kind of pathogen that could infect you as a child and trigger cancer or multiple sclerosis (MS) 40-70 years later?
However, in two joint statements, the German Federal Institute for Risk Assessment (BfR) and the Max Rubner Institute (MRI) have rejected such theories.
In 2008, Harald zur Hausen, MD, DSc, received the Nobel Prize in Medicine for his discovery that human papillomaviruses cause cervical cancer. His starting point was the observation that sexually abstinent women, such as nuns, rarely develop this cancer. So it was possible to draw the conclusion that pathogens are transmitted during sexual intercourse, explain Dr. zur Hausen and his wife Ethel-Michele de Villiers, PhD, both of DKFZ Heidelberg.
Papillomaviruses, as well as human herpes and Epstein-Barr viruses (EBV), polyomaviruses, and retroviruses, cause cancer in a direct way: by inserting their genes into the DNA of human cells. With a latency of a few years to a few decades, the proteins formed through expression stimulate malignant growth by altering the regulating host gene.
Acid radicals
However, viruses – just like bacteria and parasites – can also indirectly trigger cancer. One mechanism for this triggering is the disruption of immune defenses, as shown by the sometimes drastically increased tumor incidence with AIDS or with immunosuppressants after transplants. Chronic inflammation is a second mechanism that generates acid radicals and thereby causes random mutations in replicating cells. Examples include stomach cancer caused by Helicobacter pylori and liver cancer caused by Schistosoma, liver fluke, and hepatitis B and C viruses.
According to Dr. de Villiers and Dr. zur Hausen, there are good reasons to believe that other pathogens could cause chronic inflammation and thereby lead to cancer. Epidemiologic data suggest that dairy and meat products from European cows (Bos taurus) are a potential source. This is because colon cancer and breast cancer commonly occur in places where these foods are heavily consumed (that is, in North America, Argentina, Europe, and Australia). In contrast, the rate is low in India, where cows are revered as holy animals. Also noteworthy is that women with a lactose intolerance rarely develop breast cancer.
Viral progeny
In fact, the researchers found single-stranded DNA rings that originated in viruses, which they named bovine meat and milk factors (BMMF), in the intestines of patients with colon cancer. They reported, “This new class of pathogen deserves, in our opinion at least, to become the focus of cancer development and further chronic diseases.” They also detected elevated levels of acid radicals in these areas (that is, oxidative stress), which is typical for chronic inflammation.
The researchers assume that infants, whose immune system is not yet fully matured, ingest the BMMF as soon as they have dairy. Therefore, there is no need for adults to avoid dairy or beef because everyone is infected anyway, said Dr. zur Hausen.
‘Breast milk is healthy’
Dr. De Villiers and Dr. zur Hausen outlined more evidence of cancer-triggering pathogens. Mothers who have breastfed are less likely, especially after multiple pregnancies, to develop tumors in various organs or to have MS and type 2 diabetes. The authors attribute the protective effect to oligosaccharides in breast milk, which begin to be formed midway through the pregnancy. They bind to lectin receptors and, in so doing, mask the terminal molecule onto which the viruses need to dock. As a result, their port of entry into the cells is blocked.
The oligosaccharides also protect the baby against life-threatening infections by blocking access by rotaviruses and noroviruses. In this way, especially if breastfeeding lasts a long time – around 1 year – the period of incomplete immunocompetence is bridged.
Colon cancer
To date, it has been assumed that around 20% of all cancerous diseases globally are caused by infections, said the researchers. But if the suspected BMMF cases are included, this figure rises to 50%, even to around 80%, for colon cancer. If the suspicion is confirmed, the consequences for prevention and therapy would be significant.
The voice of a Nobel prize winner undoubtedly carries weight, but at the time, Dr. zur Hausen still had to convince a host of skeptics with his discovery that a viral infection is a major cause of cervical cancer. Nonetheless, some indicators suggest that he and his wife have found a dead end this time.
Institutional skepticism
When his working group made the results public in February 2019, the DKFZ felt the need to give an all-clear signal in response to alarmed press reports. There is no reason to see dairy and meat consumption as something negative. Similarly, in their first joint statement, the BfR and the MRI judged the data to be insufficient and called for further studies. Multiple research teams began to focus on BMMF as a result. In what foods can they be found? Are they more common in patients with cancer than in healthy people? Are they infectious? Do they cause inflammation and cancer?
The findings presented in a second statement by the BfR and MRI at the end of November 2022 contradicted the claims made by the DKFZ scientists across the board. In no way do BMMF represent new pathogens. They are variants of already known DNA sequences. In addition, they are present in numerous animal-based and plant-based foods, including pork, fish, fruit, vegetables, and nuts.
BMMF do not possess the ability to infect human cells, the institutes said. The proof that they are damaging to one’s health was also absent. It is true that the incidence of intestinal tumors correlates positively with the consumption of red and processed meat – which in no way signifies causality – but dairy products are linked to a reduced risk. On the other hand, breast cancer cannot be associated with the consumption of beef or dairy.
Therefore, both institutes recommend continuing to use these products as supplementary diet for infants because of their micronutrients. They further stated that the products are safe for people of all ages.
Association with MS?
Unperturbed, Dr. de Villiers and Dr. zur Hausen went one step further in their current article. They posited that MS is also associated with the consumption of dairy products and beef. Here too geographic distribution prompted the idea to look for BMMF in the brain lesions of patients with MS. The researchers isolated ring-shaped DNA molecules that proved to be closely related to BMMF from dairy and cattle blood. “The result was electrifying for us.”
However, there are several other factors to consider, such as vitamin D3 deficiency. This is because the incidence of MS decreases the further you travel from the poles toward the equator (that is, as solar radiation increases). Also, EBV clearly plays a role because patients with MS display increased titers of EBV antibodies. One study also showed that people in Antarctica excreted reactivated EBV in their saliva during winter and that vitamin D3 stopped the viral secretion.
Under these conditions, the researchers hypothesized that MS is caused by a double infection of brain cells by EBV and BMMF. EBV is reactivated by a lack of vitamin D3, and the BMMF multiply and are eventually converted into proteins. A focal immunoreaction causes the Schwann cells and oligodendrocytes to malfunction, which leads to the destruction of the myelin sheaths around the nerve fibers.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
In Western diets, dairy and beef are ubiquitous: Milk goes with coffee, melted cheese with pizza, and chili with rice. But what if dairy products and beef contained a new kind of pathogen that could infect you as a child and trigger cancer or multiple sclerosis (MS) 40-70 years later?
However, in two joint statements, the German Federal Institute for Risk Assessment (BfR) and the Max Rubner Institute (MRI) have rejected such theories.
In 2008, Harald zur Hausen, MD, DSc, received the Nobel Prize in Medicine for his discovery that human papillomaviruses cause cervical cancer. His starting point was the observation that sexually abstinent women, such as nuns, rarely develop this cancer. So it was possible to draw the conclusion that pathogens are transmitted during sexual intercourse, explain Dr. zur Hausen and his wife Ethel-Michele de Villiers, PhD, both of DKFZ Heidelberg.
Papillomaviruses, as well as human herpes and Epstein-Barr viruses (EBV), polyomaviruses, and retroviruses, cause cancer in a direct way: by inserting their genes into the DNA of human cells. With a latency of a few years to a few decades, the proteins formed through expression stimulate malignant growth by altering the regulating host gene.
Acid radicals
However, viruses – just like bacteria and parasites – can also indirectly trigger cancer. One mechanism for this triggering is the disruption of immune defenses, as shown by the sometimes drastically increased tumor incidence with AIDS or with immunosuppressants after transplants. Chronic inflammation is a second mechanism that generates acid radicals and thereby causes random mutations in replicating cells. Examples include stomach cancer caused by Helicobacter pylori and liver cancer caused by Schistosoma, liver fluke, and hepatitis B and C viruses.
According to Dr. de Villiers and Dr. zur Hausen, there are good reasons to believe that other pathogens could cause chronic inflammation and thereby lead to cancer. Epidemiologic data suggest that dairy and meat products from European cows (Bos taurus) are a potential source. This is because colon cancer and breast cancer commonly occur in places where these foods are heavily consumed (that is, in North America, Argentina, Europe, and Australia). In contrast, the rate is low in India, where cows are revered as holy animals. Also noteworthy is that women with a lactose intolerance rarely develop breast cancer.
Viral progeny
In fact, the researchers found single-stranded DNA rings that originated in viruses, which they named bovine meat and milk factors (BMMF), in the intestines of patients with colon cancer. They reported, “This new class of pathogen deserves, in our opinion at least, to become the focus of cancer development and further chronic diseases.” They also detected elevated levels of acid radicals in these areas (that is, oxidative stress), which is typical for chronic inflammation.
The researchers assume that infants, whose immune system is not yet fully matured, ingest the BMMF as soon as they have dairy. Therefore, there is no need for adults to avoid dairy or beef because everyone is infected anyway, said Dr. zur Hausen.
‘Breast milk is healthy’
Dr. De Villiers and Dr. zur Hausen outlined more evidence of cancer-triggering pathogens. Mothers who have breastfed are less likely, especially after multiple pregnancies, to develop tumors in various organs or to have MS and type 2 diabetes. The authors attribute the protective effect to oligosaccharides in breast milk, which begin to be formed midway through the pregnancy. They bind to lectin receptors and, in so doing, mask the terminal molecule onto which the viruses need to dock. As a result, their port of entry into the cells is blocked.
The oligosaccharides also protect the baby against life-threatening infections by blocking access by rotaviruses and noroviruses. In this way, especially if breastfeeding lasts a long time – around 1 year – the period of incomplete immunocompetence is bridged.
Colon cancer
To date, it has been assumed that around 20% of all cancerous diseases globally are caused by infections, said the researchers. But if the suspected BMMF cases are included, this figure rises to 50%, even to around 80%, for colon cancer. If the suspicion is confirmed, the consequences for prevention and therapy would be significant.
The voice of a Nobel prize winner undoubtedly carries weight, but at the time, Dr. zur Hausen still had to convince a host of skeptics with his discovery that a viral infection is a major cause of cervical cancer. Nonetheless, some indicators suggest that he and his wife have found a dead end this time.
Institutional skepticism
When his working group made the results public in February 2019, the DKFZ felt the need to give an all-clear signal in response to alarmed press reports. There is no reason to see dairy and meat consumption as something negative. Similarly, in their first joint statement, the BfR and the MRI judged the data to be insufficient and called for further studies. Multiple research teams began to focus on BMMF as a result. In what foods can they be found? Are they more common in patients with cancer than in healthy people? Are they infectious? Do they cause inflammation and cancer?
The findings presented in a second statement by the BfR and MRI at the end of November 2022 contradicted the claims made by the DKFZ scientists across the board. In no way do BMMF represent new pathogens. They are variants of already known DNA sequences. In addition, they are present in numerous animal-based and plant-based foods, including pork, fish, fruit, vegetables, and nuts.
BMMF do not possess the ability to infect human cells, the institutes said. The proof that they are damaging to one’s health was also absent. It is true that the incidence of intestinal tumors correlates positively with the consumption of red and processed meat – which in no way signifies causality – but dairy products are linked to a reduced risk. On the other hand, breast cancer cannot be associated with the consumption of beef or dairy.
Therefore, both institutes recommend continuing to use these products as supplementary diet for infants because of their micronutrients. They further stated that the products are safe for people of all ages.
Association with MS?
Unperturbed, Dr. de Villiers and Dr. zur Hausen went one step further in their current article. They posited that MS is also associated with the consumption of dairy products and beef. Here too geographic distribution prompted the idea to look for BMMF in the brain lesions of patients with MS. The researchers isolated ring-shaped DNA molecules that proved to be closely related to BMMF from dairy and cattle blood. “The result was electrifying for us.”
However, there are several other factors to consider, such as vitamin D3 deficiency. This is because the incidence of MS decreases the further you travel from the poles toward the equator (that is, as solar radiation increases). Also, EBV clearly plays a role because patients with MS display increased titers of EBV antibodies. One study also showed that people in Antarctica excreted reactivated EBV in their saliva during winter and that vitamin D3 stopped the viral secretion.
Under these conditions, the researchers hypothesized that MS is caused by a double infection of brain cells by EBV and BMMF. EBV is reactivated by a lack of vitamin D3, and the BMMF multiply and are eventually converted into proteins. A focal immunoreaction causes the Schwann cells and oligodendrocytes to malfunction, which leads to the destruction of the myelin sheaths around the nerve fibers.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
In Western diets, dairy and beef are ubiquitous: Milk goes with coffee, melted cheese with pizza, and chili with rice. But what if dairy products and beef contained a new kind of pathogen that could infect you as a child and trigger cancer or multiple sclerosis (MS) 40-70 years later?
However, in two joint statements, the German Federal Institute for Risk Assessment (BfR) and the Max Rubner Institute (MRI) have rejected such theories.
In 2008, Harald zur Hausen, MD, DSc, received the Nobel Prize in Medicine for his discovery that human papillomaviruses cause cervical cancer. His starting point was the observation that sexually abstinent women, such as nuns, rarely develop this cancer. So it was possible to draw the conclusion that pathogens are transmitted during sexual intercourse, explain Dr. zur Hausen and his wife Ethel-Michele de Villiers, PhD, both of DKFZ Heidelberg.
Papillomaviruses, as well as human herpes and Epstein-Barr viruses (EBV), polyomaviruses, and retroviruses, cause cancer in a direct way: by inserting their genes into the DNA of human cells. With a latency of a few years to a few decades, the proteins formed through expression stimulate malignant growth by altering the regulating host gene.
Acid radicals
However, viruses – just like bacteria and parasites – can also indirectly trigger cancer. One mechanism for this triggering is the disruption of immune defenses, as shown by the sometimes drastically increased tumor incidence with AIDS or with immunosuppressants after transplants. Chronic inflammation is a second mechanism that generates acid radicals and thereby causes random mutations in replicating cells. Examples include stomach cancer caused by Helicobacter pylori and liver cancer caused by Schistosoma, liver fluke, and hepatitis B and C viruses.
According to Dr. de Villiers and Dr. zur Hausen, there are good reasons to believe that other pathogens could cause chronic inflammation and thereby lead to cancer. Epidemiologic data suggest that dairy and meat products from European cows (Bos taurus) are a potential source. This is because colon cancer and breast cancer commonly occur in places where these foods are heavily consumed (that is, in North America, Argentina, Europe, and Australia). In contrast, the rate is low in India, where cows are revered as holy animals. Also noteworthy is that women with a lactose intolerance rarely develop breast cancer.
Viral progeny
In fact, the researchers found single-stranded DNA rings that originated in viruses, which they named bovine meat and milk factors (BMMF), in the intestines of patients with colon cancer. They reported, “This new class of pathogen deserves, in our opinion at least, to become the focus of cancer development and further chronic diseases.” They also detected elevated levels of acid radicals in these areas (that is, oxidative stress), which is typical for chronic inflammation.
The researchers assume that infants, whose immune system is not yet fully matured, ingest the BMMF as soon as they have dairy. Therefore, there is no need for adults to avoid dairy or beef because everyone is infected anyway, said Dr. zur Hausen.
‘Breast milk is healthy’
Dr. De Villiers and Dr. zur Hausen outlined more evidence of cancer-triggering pathogens. Mothers who have breastfed are less likely, especially after multiple pregnancies, to develop tumors in various organs or to have MS and type 2 diabetes. The authors attribute the protective effect to oligosaccharides in breast milk, which begin to be formed midway through the pregnancy. They bind to lectin receptors and, in so doing, mask the terminal molecule onto which the viruses need to dock. As a result, their port of entry into the cells is blocked.
The oligosaccharides also protect the baby against life-threatening infections by blocking access by rotaviruses and noroviruses. In this way, especially if breastfeeding lasts a long time – around 1 year – the period of incomplete immunocompetence is bridged.
Colon cancer
To date, it has been assumed that around 20% of all cancerous diseases globally are caused by infections, said the researchers. But if the suspected BMMF cases are included, this figure rises to 50%, even to around 80%, for colon cancer. If the suspicion is confirmed, the consequences for prevention and therapy would be significant.
The voice of a Nobel prize winner undoubtedly carries weight, but at the time, Dr. zur Hausen still had to convince a host of skeptics with his discovery that a viral infection is a major cause of cervical cancer. Nonetheless, some indicators suggest that he and his wife have found a dead end this time.
Institutional skepticism
When his working group made the results public in February 2019, the DKFZ felt the need to give an all-clear signal in response to alarmed press reports. There is no reason to see dairy and meat consumption as something negative. Similarly, in their first joint statement, the BfR and the MRI judged the data to be insufficient and called for further studies. Multiple research teams began to focus on BMMF as a result. In what foods can they be found? Are they more common in patients with cancer than in healthy people? Are they infectious? Do they cause inflammation and cancer?
The findings presented in a second statement by the BfR and MRI at the end of November 2022 contradicted the claims made by the DKFZ scientists across the board. In no way do BMMF represent new pathogens. They are variants of already known DNA sequences. In addition, they are present in numerous animal-based and plant-based foods, including pork, fish, fruit, vegetables, and nuts.
BMMF do not possess the ability to infect human cells, the institutes said. The proof that they are damaging to one’s health was also absent. It is true that the incidence of intestinal tumors correlates positively with the consumption of red and processed meat – which in no way signifies causality – but dairy products are linked to a reduced risk. On the other hand, breast cancer cannot be associated with the consumption of beef or dairy.
Therefore, both institutes recommend continuing to use these products as supplementary diet for infants because of their micronutrients. They further stated that the products are safe for people of all ages.
Association with MS?
Unperturbed, Dr. de Villiers and Dr. zur Hausen went one step further in their current article. They posited that MS is also associated with the consumption of dairy products and beef. Here too geographic distribution prompted the idea to look for BMMF in the brain lesions of patients with MS. The researchers isolated ring-shaped DNA molecules that proved to be closely related to BMMF from dairy and cattle blood. “The result was electrifying for us.”
However, there are several other factors to consider, such as vitamin D3 deficiency. This is because the incidence of MS decreases the further you travel from the poles toward the equator (that is, as solar radiation increases). Also, EBV clearly plays a role because patients with MS display increased titers of EBV antibodies. One study also showed that people in Antarctica excreted reactivated EBV in their saliva during winter and that vitamin D3 stopped the viral secretion.
Under these conditions, the researchers hypothesized that MS is caused by a double infection of brain cells by EBV and BMMF. EBV is reactivated by a lack of vitamin D3, and the BMMF multiply and are eventually converted into proteins. A focal immunoreaction causes the Schwann cells and oligodendrocytes to malfunction, which leads to the destruction of the myelin sheaths around the nerve fibers.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.