User login
Editorial Board Biographies
Matthew J. Matava, MD
Associate Editor for Professional Sports
Dr. Matava is a professor of Orthopedic Surgery and Physical Therapy, Chief of the Sports Medicine Service, and the Head Team Physician for the varsity athletic program at Washington University in St. Louis. He is also a team physician for the National Hockey League’s St. Louis Blues. Formerly, he was the Head Team Physician for the St. Louis Rams, and was President of the National Football League Physicians Society (NFLPS) from 2013-2015. Dr. Matava earned his Medical Degree from the University of Missouri-Kansas City. He completed his internship and orthopedic surgery residency at Emory University in Atlanta, GA, followed by a fellowship in sports medicine and arthroscopic surgery at the Cincinnati Sports Medicine and Orthopedic Center. He is the recipient of several research awards from Emory University, is a member of the Alpha Omega Medical Honor Society, and received the Palma Chironis Award for Excellence in Teaching from the Washington University Department of Orthopedic Surgery in 2012. Dr. Matava has been listed as a “Best Doctor in America” since 2005, and was recently hailed by Orthopedics This Week as one of the top 28 sports knee surgeons in the nation.
Jeffrey Sawyer, MD
Associate Editor for Pediatrics
Dr. Sawyer is a professor of Orthopaedic Surgery and the Pediatric Orthopaedic Fellowship Director at the University of Tennessee-Campbell Clinic. He also serves as a reviewer/editor for the Journal of Pediatric Orthopaedics and Orthopedic Clinics of North America. He graduated from the University of Rochester School of Medicine and completed his residency at the University of Pennsylvania, prior to completing his Pediatric Orthopaedic Fellowship at the University of Tennessee-Campbell Clinic. Dr. Sawyer has held numerous leadership positions in the Pediatric Orthopaedic Society of North America (POSNA). He also was a POSNA Traveling Fellow and won the POSNA Special Achievement Award for his work on the Pediatric Orthopaedic Workforce. He is a national authority on pediatric orthopedic trauma, and is on the Executive Committee of the Children’s Spine Foundation.
Brian K. Vickaryous, MD
Associate Editor for Trauma
Dr. Vickaryous is a specialist in orthopedic traumatology at the Florida Hospital Orthopedic Institute in Orlando, Florida, and has an additional subspecialty board certification in sports medicine. He attended the University of Miami, Florida through the combined degree Medical
Honors Program and completed his residency at the William Beaumont Army Medical Center/Texas Tech University of the Health Sciences. Dr. Vickaryous has also deployed overseas as Commander of the Trauma Unit, the 8th Forward Surgical Team, in Iraq in support of Operation Iraqi Freedom. He currently is a member of the American Academy of Orthopaedic
Surgeons (AAOS) and the Orthopaedic Trauma Association (OTA).
Michael B. Gerhardt, MD
Associate Editor for Sports Medicine
Dr. Gerhardt is a sports medicine specialist at the Kerlan-Jobe Institute and Santa Monica Orthopaedic Group in Los Angeles, CA. He also serves as faculty in the Department of Orthopaedic Surgery at Cedars-Sinai Medical Center. Dr. Gerhardt earned his undergraduate degree from UC San Diego and graduated medical school with honors from the Medical College of Pennsylvania. He received the Leonard Marmur Award for excellence in research and education during his orthopedic residency at the University of Southern California, prior to completing a Sports Medicine Fellowship in 2003. He received further training in hip arthroscopy at the Nashville Orthopaedic Sports Medicine and Orthopaedic Clinic, and maintains a leadership role in the area of sports medicine and hip preservation on a national and international level. Currently, he serves as Team Physician for the US Soccer Men’s National Team, the Los Angeles Galaxy, and Pepperdine University.
Matthew J. Matava, MD
Associate Editor for Professional Sports
Dr. Matava is a professor of Orthopedic Surgery and Physical Therapy, Chief of the Sports Medicine Service, and the Head Team Physician for the varsity athletic program at Washington University in St. Louis. He is also a team physician for the National Hockey League’s St. Louis Blues. Formerly, he was the Head Team Physician for the St. Louis Rams, and was President of the National Football League Physicians Society (NFLPS) from 2013-2015. Dr. Matava earned his Medical Degree from the University of Missouri-Kansas City. He completed his internship and orthopedic surgery residency at Emory University in Atlanta, GA, followed by a fellowship in sports medicine and arthroscopic surgery at the Cincinnati Sports Medicine and Orthopedic Center. He is the recipient of several research awards from Emory University, is a member of the Alpha Omega Medical Honor Society, and received the Palma Chironis Award for Excellence in Teaching from the Washington University Department of Orthopedic Surgery in 2012. Dr. Matava has been listed as a “Best Doctor in America” since 2005, and was recently hailed by Orthopedics This Week as one of the top 28 sports knee surgeons in the nation.
Jeffrey Sawyer, MD
Associate Editor for Pediatrics
Dr. Sawyer is a professor of Orthopaedic Surgery and the Pediatric Orthopaedic Fellowship Director at the University of Tennessee-Campbell Clinic. He also serves as a reviewer/editor for the Journal of Pediatric Orthopaedics and Orthopedic Clinics of North America. He graduated from the University of Rochester School of Medicine and completed his residency at the University of Pennsylvania, prior to completing his Pediatric Orthopaedic Fellowship at the University of Tennessee-Campbell Clinic. Dr. Sawyer has held numerous leadership positions in the Pediatric Orthopaedic Society of North America (POSNA). He also was a POSNA Traveling Fellow and won the POSNA Special Achievement Award for his work on the Pediatric Orthopaedic Workforce. He is a national authority on pediatric orthopedic trauma, and is on the Executive Committee of the Children’s Spine Foundation.
Brian K. Vickaryous, MD
Associate Editor for Trauma
Dr. Vickaryous is a specialist in orthopedic traumatology at the Florida Hospital Orthopedic Institute in Orlando, Florida, and has an additional subspecialty board certification in sports medicine. He attended the University of Miami, Florida through the combined degree Medical
Honors Program and completed his residency at the William Beaumont Army Medical Center/Texas Tech University of the Health Sciences. Dr. Vickaryous has also deployed overseas as Commander of the Trauma Unit, the 8th Forward Surgical Team, in Iraq in support of Operation Iraqi Freedom. He currently is a member of the American Academy of Orthopaedic
Surgeons (AAOS) and the Orthopaedic Trauma Association (OTA).
Michael B. Gerhardt, MD
Associate Editor for Sports Medicine
Dr. Gerhardt is a sports medicine specialist at the Kerlan-Jobe Institute and Santa Monica Orthopaedic Group in Los Angeles, CA. He also serves as faculty in the Department of Orthopaedic Surgery at Cedars-Sinai Medical Center. Dr. Gerhardt earned his undergraduate degree from UC San Diego and graduated medical school with honors from the Medical College of Pennsylvania. He received the Leonard Marmur Award for excellence in research and education during his orthopedic residency at the University of Southern California, prior to completing a Sports Medicine Fellowship in 2003. He received further training in hip arthroscopy at the Nashville Orthopaedic Sports Medicine and Orthopaedic Clinic, and maintains a leadership role in the area of sports medicine and hip preservation on a national and international level. Currently, he serves as Team Physician for the US Soccer Men’s National Team, the Los Angeles Galaxy, and Pepperdine University.
Matthew J. Matava, MD
Associate Editor for Professional Sports
Dr. Matava is a professor of Orthopedic Surgery and Physical Therapy, Chief of the Sports Medicine Service, and the Head Team Physician for the varsity athletic program at Washington University in St. Louis. He is also a team physician for the National Hockey League’s St. Louis Blues. Formerly, he was the Head Team Physician for the St. Louis Rams, and was President of the National Football League Physicians Society (NFLPS) from 2013-2015. Dr. Matava earned his Medical Degree from the University of Missouri-Kansas City. He completed his internship and orthopedic surgery residency at Emory University in Atlanta, GA, followed by a fellowship in sports medicine and arthroscopic surgery at the Cincinnati Sports Medicine and Orthopedic Center. He is the recipient of several research awards from Emory University, is a member of the Alpha Omega Medical Honor Society, and received the Palma Chironis Award for Excellence in Teaching from the Washington University Department of Orthopedic Surgery in 2012. Dr. Matava has been listed as a “Best Doctor in America” since 2005, and was recently hailed by Orthopedics This Week as one of the top 28 sports knee surgeons in the nation.
Jeffrey Sawyer, MD
Associate Editor for Pediatrics
Dr. Sawyer is a professor of Orthopaedic Surgery and the Pediatric Orthopaedic Fellowship Director at the University of Tennessee-Campbell Clinic. He also serves as a reviewer/editor for the Journal of Pediatric Orthopaedics and Orthopedic Clinics of North America. He graduated from the University of Rochester School of Medicine and completed his residency at the University of Pennsylvania, prior to completing his Pediatric Orthopaedic Fellowship at the University of Tennessee-Campbell Clinic. Dr. Sawyer has held numerous leadership positions in the Pediatric Orthopaedic Society of North America (POSNA). He also was a POSNA Traveling Fellow and won the POSNA Special Achievement Award for his work on the Pediatric Orthopaedic Workforce. He is a national authority on pediatric orthopedic trauma, and is on the Executive Committee of the Children’s Spine Foundation.
Brian K. Vickaryous, MD
Associate Editor for Trauma
Dr. Vickaryous is a specialist in orthopedic traumatology at the Florida Hospital Orthopedic Institute in Orlando, Florida, and has an additional subspecialty board certification in sports medicine. He attended the University of Miami, Florida through the combined degree Medical
Honors Program and completed his residency at the William Beaumont Army Medical Center/Texas Tech University of the Health Sciences. Dr. Vickaryous has also deployed overseas as Commander of the Trauma Unit, the 8th Forward Surgical Team, in Iraq in support of Operation Iraqi Freedom. He currently is a member of the American Academy of Orthopaedic
Surgeons (AAOS) and the Orthopaedic Trauma Association (OTA).
Michael B. Gerhardt, MD
Associate Editor for Sports Medicine
Dr. Gerhardt is a sports medicine specialist at the Kerlan-Jobe Institute and Santa Monica Orthopaedic Group in Los Angeles, CA. He also serves as faculty in the Department of Orthopaedic Surgery at Cedars-Sinai Medical Center. Dr. Gerhardt earned his undergraduate degree from UC San Diego and graduated medical school with honors from the Medical College of Pennsylvania. He received the Leonard Marmur Award for excellence in research and education during his orthopedic residency at the University of Southern California, prior to completing a Sports Medicine Fellowship in 2003. He received further training in hip arthroscopy at the Nashville Orthopaedic Sports Medicine and Orthopaedic Clinic, and maintains a leadership role in the area of sports medicine and hip preservation on a national and international level. Currently, he serves as Team Physician for the US Soccer Men’s National Team, the Los Angeles Galaxy, and Pepperdine University.
Neuroscience-based Nomenclature: Classifying psychotropics by mechanism of action rather than indication
An important new initiative to reclassify psychiatric medications is underway. Currently, psychotropic drugs are named primarily for their clinical use, usually as a member of 1 of 6 classes: antipsychotic, anti‑depressant, mood stabilizer, stimulant, anxiolytic, and hypnotic.1,2
This naming system creates confusion because so-called antidepressants commonly are used as anxiolytics, antipsychotics increasingly are used as antidepressants, and so on.1,2
Vocabulary based on clinical indications also leads to difficulty in classifying new agents, especially those with novel mechanisms of action or clinical uses. Therefore, there is a need to make the names of psychotropic drugs more rational and scientifically based, rather than indication-based. A task force of experts from major psychopharmacology societies around the world is developing an alternative naming system that is increasingly being accepted by the major experts and journals throughout the world, called Neuroscience-based Nomenclature (NbN).3-5
So, what is NbN?


First and foremost, NbN renames the >100 known psychotropic drugs by 1 of the 11 principle pharmacological domains that include well-known terms such as serotonin dopamine, acetylcholine, and GABA (Table 1). Also included in NbN are 9 familiar modes of action, such as agonist, antagonist, reuptake inhibitor, and enzyme inhibitors (Table 2).3-5
NbN has 4 additional dimensions or layers3-5: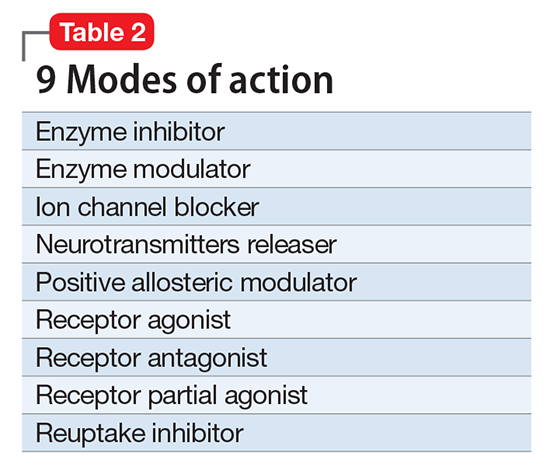
- The first layer enumerates the official indications as recognized by the regulatory agencies (ie, the FDA and other government organizations).
- The second layer states efficacy based on randomized controlled trials or substantial, evidence-based clinical data, as well as side effects (not the exhaustive list provided in manufacturers’ package inserts, but only the most common ones).
- The third layer is comprised of practical notes, highlighting potentially important drug interactions, metabolic issues, and specific warnings.
- The fourth section summarizes the neurobiological effects in laboratory animals and humans.
Specific dosages and titration regimens are not provided because they can vary among different countries, and NbN is intended for nomenclature and classification, not as a prescribing guide.
How does it work in practice?
Major journals in the field have begun adapting NbN for their published papers and
What is the current status?
Two international organizations endorse NbN, and the chief editors of nearly 3 dozen scientific journals, including
Clinicians should start adopting the NbN for the psychotropic drugs they prescribe every day. It is more scientific and consistent with the mechanism of action than with a specific disorder because many psychotropic medications have been found to be useful in >1 psychiatric disorder.
1. Nutt DJ. Beyond psychoanaleptics - can we improve antidepressant drug nomenclature? J Psychopharmacol. 2009;23(4):343-345.
2. Stahl SM. Classifying psychotropic drugs by mode of action not by target disorders. CNS Spectr. 2013;18(3):113-117.
3. Zohar J, Stahl S, Moller HJ, et al. A review of the current nomenclature for psychotropic agents and an introduction to the Neuroscience-based Nomenclature. Eur Neuropsychopharmacol. 2015; 25(12):2318-2325.
4. Zohar J, Stahl S, Moller HJ, et al. Neuroscience based nomenclature. Cambridge, United Kingdom: Cambridge University Press; 2014:254.
5. Neuroscience-based nomenclature. http://nbnomenclature.org. Accessed April 12, 2017.
An important new initiative to reclassify psychiatric medications is underway. Currently, psychotropic drugs are named primarily for their clinical use, usually as a member of 1 of 6 classes: antipsychotic, anti‑depressant, mood stabilizer, stimulant, anxiolytic, and hypnotic.1,2
This naming system creates confusion because so-called antidepressants commonly are used as anxiolytics, antipsychotics increasingly are used as antidepressants, and so on.1,2
Vocabulary based on clinical indications also leads to difficulty in classifying new agents, especially those with novel mechanisms of action or clinical uses. Therefore, there is a need to make the names of psychotropic drugs more rational and scientifically based, rather than indication-based. A task force of experts from major psychopharmacology societies around the world is developing an alternative naming system that is increasingly being accepted by the major experts and journals throughout the world, called Neuroscience-based Nomenclature (NbN).3-5
So, what is NbN?


First and foremost, NbN renames the >100 known psychotropic drugs by 1 of the 11 principle pharmacological domains that include well-known terms such as serotonin dopamine, acetylcholine, and GABA (Table 1). Also included in NbN are 9 familiar modes of action, such as agonist, antagonist, reuptake inhibitor, and enzyme inhibitors (Table 2).3-5
NbN has 4 additional dimensions or layers3-5:
- The first layer enumerates the official indications as recognized by the regulatory agencies (ie, the FDA and other government organizations).
- The second layer states efficacy based on randomized controlled trials or substantial, evidence-based clinical data, as well as side effects (not the exhaustive list provided in manufacturers’ package inserts, but only the most common ones).
- The third layer is comprised of practical notes, highlighting potentially important drug interactions, metabolic issues, and specific warnings.
- The fourth section summarizes the neurobiological effects in laboratory animals and humans.
Specific dosages and titration regimens are not provided because they can vary among different countries, and NbN is intended for nomenclature and classification, not as a prescribing guide.
How does it work in practice?
Major journals in the field have begun adapting NbN for their published papers and
What is the current status?
Two international organizations endorse NbN, and the chief editors of nearly 3 dozen scientific journals, including
Clinicians should start adopting the NbN for the psychotropic drugs they prescribe every day. It is more scientific and consistent with the mechanism of action than with a specific disorder because many psychotropic medications have been found to be useful in >1 psychiatric disorder.
An important new initiative to reclassify psychiatric medications is underway. Currently, psychotropic drugs are named primarily for their clinical use, usually as a member of 1 of 6 classes: antipsychotic, anti‑depressant, mood stabilizer, stimulant, anxiolytic, and hypnotic.1,2
This naming system creates confusion because so-called antidepressants commonly are used as anxiolytics, antipsychotics increasingly are used as antidepressants, and so on.1,2
Vocabulary based on clinical indications also leads to difficulty in classifying new agents, especially those with novel mechanisms of action or clinical uses. Therefore, there is a need to make the names of psychotropic drugs more rational and scientifically based, rather than indication-based. A task force of experts from major psychopharmacology societies around the world is developing an alternative naming system that is increasingly being accepted by the major experts and journals throughout the world, called Neuroscience-based Nomenclature (NbN).3-5
So, what is NbN?


First and foremost, NbN renames the >100 known psychotropic drugs by 1 of the 11 principle pharmacological domains that include well-known terms such as serotonin dopamine, acetylcholine, and GABA (Table 1). Also included in NbN are 9 familiar modes of action, such as agonist, antagonist, reuptake inhibitor, and enzyme inhibitors (Table 2).3-5
NbN has 4 additional dimensions or layers3-5:
- The first layer enumerates the official indications as recognized by the regulatory agencies (ie, the FDA and other government organizations).
- The second layer states efficacy based on randomized controlled trials or substantial, evidence-based clinical data, as well as side effects (not the exhaustive list provided in manufacturers’ package inserts, but only the most common ones).
- The third layer is comprised of practical notes, highlighting potentially important drug interactions, metabolic issues, and specific warnings.
- The fourth section summarizes the neurobiological effects in laboratory animals and humans.
Specific dosages and titration regimens are not provided because they can vary among different countries, and NbN is intended for nomenclature and classification, not as a prescribing guide.
How does it work in practice?
Major journals in the field have begun adapting NbN for their published papers and
What is the current status?
Two international organizations endorse NbN, and the chief editors of nearly 3 dozen scientific journals, including
Clinicians should start adopting the NbN for the psychotropic drugs they prescribe every day. It is more scientific and consistent with the mechanism of action than with a specific disorder because many psychotropic medications have been found to be useful in >1 psychiatric disorder.
1. Nutt DJ. Beyond psychoanaleptics - can we improve antidepressant drug nomenclature? J Psychopharmacol. 2009;23(4):343-345.
2. Stahl SM. Classifying psychotropic drugs by mode of action not by target disorders. CNS Spectr. 2013;18(3):113-117.
3. Zohar J, Stahl S, Moller HJ, et al. A review of the current nomenclature for psychotropic agents and an introduction to the Neuroscience-based Nomenclature. Eur Neuropsychopharmacol. 2015; 25(12):2318-2325.
4. Zohar J, Stahl S, Moller HJ, et al. Neuroscience based nomenclature. Cambridge, United Kingdom: Cambridge University Press; 2014:254.
5. Neuroscience-based nomenclature. http://nbnomenclature.org. Accessed April 12, 2017.
1. Nutt DJ. Beyond psychoanaleptics - can we improve antidepressant drug nomenclature? J Psychopharmacol. 2009;23(4):343-345.
2. Stahl SM. Classifying psychotropic drugs by mode of action not by target disorders. CNS Spectr. 2013;18(3):113-117.
3. Zohar J, Stahl S, Moller HJ, et al. A review of the current nomenclature for psychotropic agents and an introduction to the Neuroscience-based Nomenclature. Eur Neuropsychopharmacol. 2015; 25(12):2318-2325.
4. Zohar J, Stahl S, Moller HJ, et al. Neuroscience based nomenclature. Cambridge, United Kingdom: Cambridge University Press; 2014:254.
5. Neuroscience-based nomenclature. http://nbnomenclature.org. Accessed April 12, 2017.
These agents do double duty by reducing CV risk in diabetes
In this issue of JFP, Skolnik et al discuss ways we can assist patients with type 2 diabetes mellitus (T2DM) in lowering their cardiovascular (CV) risk. It is well established that the main predictors of the development of CV disease in patients with T2DM are blood pressure (BP) and lipid levels. Many randomized controlled trials (RCTs) have demonstrated the benefit of lowering BP and lipid levels on reducing CV disease in these patients.
The problem has been that other than a modest CV benefit from metformin, no glucose-lowering drug has been shown to have a significant effect on CV outcomes—until recently. Now there is solid evidence from RCTs that treatment with one of 3 newer agents—empagliflozin (a sodium-glucose cotransporter [SGLT]-2 inhibitor), liraglutide, and semaglutide (both glucagon-like peptide [GLP]-1 receptor agonists)—is associated with reductions in CV morbidity and mortality for patients with T2DM who have established, or are at high risk for, CV disease. (Of note: Semaglutide is not yet on the market. Its manufacturer submitted a New Drug Application late last year.)
For empagliflozin, an RCT involving more than 7000 patients calculated that the number needed to treat (NNT) over a 3-year period to prevent one CV event was 63 and the NNT to prevent one death from any cause was 38.1 For liraglutide, a double-blind trial involving over 9000 patients reported the NNT to prevent one CV event in 3 years was 53, and the NNT to prevent one death from any cause was 71.2 The RCT for semaglutide involved more than 3000 patients and reported the NNT to prevent one major CVD event was 43, but there was no significant difference in CV mortality between the semaglutide and placebo groups in that clinical trial.3
1. Zinman B, Wanner C, Lachin JM, et al, for the EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117-2128.
2. Marso SP, Daniels GH, Brown-Frandsen K, et al, for the LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
3. Marso SP, Bain SC, Consoli A, et al, for the SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
4. Ebell MH, Siwek J, Weiss BD, et al. Simplifying the language of evidence to improve patient care: Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in medical literature. J Fam Pract. 2004;53:111-120.
In this issue of JFP, Skolnik et al discuss ways we can assist patients with type 2 diabetes mellitus (T2DM) in lowering their cardiovascular (CV) risk. It is well established that the main predictors of the development of CV disease in patients with T2DM are blood pressure (BP) and lipid levels. Many randomized controlled trials (RCTs) have demonstrated the benefit of lowering BP and lipid levels on reducing CV disease in these patients.
The problem has been that other than a modest CV benefit from metformin, no glucose-lowering drug has been shown to have a significant effect on CV outcomes—until recently. Now there is solid evidence from RCTs that treatment with one of 3 newer agents—empagliflozin (a sodium-glucose cotransporter [SGLT]-2 inhibitor), liraglutide, and semaglutide (both glucagon-like peptide [GLP]-1 receptor agonists)—is associated with reductions in CV morbidity and mortality for patients with T2DM who have established, or are at high risk for, CV disease. (Of note: Semaglutide is not yet on the market. Its manufacturer submitted a New Drug Application late last year.)
For empagliflozin, an RCT involving more than 7000 patients calculated that the number needed to treat (NNT) over a 3-year period to prevent one CV event was 63 and the NNT to prevent one death from any cause was 38.1 For liraglutide, a double-blind trial involving over 9000 patients reported the NNT to prevent one CV event in 3 years was 53, and the NNT to prevent one death from any cause was 71.2 The RCT for semaglutide involved more than 3000 patients and reported the NNT to prevent one major CVD event was 43, but there was no significant difference in CV mortality between the semaglutide and placebo groups in that clinical trial.3
In this issue of JFP, Skolnik et al discuss ways we can assist patients with type 2 diabetes mellitus (T2DM) in lowering their cardiovascular (CV) risk. It is well established that the main predictors of the development of CV disease in patients with T2DM are blood pressure (BP) and lipid levels. Many randomized controlled trials (RCTs) have demonstrated the benefit of lowering BP and lipid levels on reducing CV disease in these patients.
The problem has been that other than a modest CV benefit from metformin, no glucose-lowering drug has been shown to have a significant effect on CV outcomes—until recently. Now there is solid evidence from RCTs that treatment with one of 3 newer agents—empagliflozin (a sodium-glucose cotransporter [SGLT]-2 inhibitor), liraglutide, and semaglutide (both glucagon-like peptide [GLP]-1 receptor agonists)—is associated with reductions in CV morbidity and mortality for patients with T2DM who have established, or are at high risk for, CV disease. (Of note: Semaglutide is not yet on the market. Its manufacturer submitted a New Drug Application late last year.)
For empagliflozin, an RCT involving more than 7000 patients calculated that the number needed to treat (NNT) over a 3-year period to prevent one CV event was 63 and the NNT to prevent one death from any cause was 38.1 For liraglutide, a double-blind trial involving over 9000 patients reported the NNT to prevent one CV event in 3 years was 53, and the NNT to prevent one death from any cause was 71.2 The RCT for semaglutide involved more than 3000 patients and reported the NNT to prevent one major CVD event was 43, but there was no significant difference in CV mortality between the semaglutide and placebo groups in that clinical trial.3
1. Zinman B, Wanner C, Lachin JM, et al, for the EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117-2128.
2. Marso SP, Daniels GH, Brown-Frandsen K, et al, for the LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
3. Marso SP, Bain SC, Consoli A, et al, for the SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
4. Ebell MH, Siwek J, Weiss BD, et al. Simplifying the language of evidence to improve patient care: Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in medical literature. J Fam Pract. 2004;53:111-120.
1. Zinman B, Wanner C, Lachin JM, et al, for the EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117-2128.
2. Marso SP, Daniels GH, Brown-Frandsen K, et al, for the LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311-322.
3. Marso SP, Bain SC, Consoli A, et al, for the SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-1844.
4. Ebell MH, Siwek J, Weiss BD, et al. Simplifying the language of evidence to improve patient care: Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in medical literature. J Fam Pract. 2004;53:111-120.
The EHR Report: Communication, social media, and legal vulnerability
Social media is now a part of everyday life. From Twitter, with its 140 character limit, to Facebook to Linkedin, there is a world of possibilities for communicating with friends, family, colleagues, and others online. Communication is good, but electronic media is a minefield for medical professionals who do not think carefully before they post.
The stories in the news about health care professionals who have posted obviously inflammatory material online, perhaps in a fit of rage, and have had their careers impacted or ended are just the tip of the iceberg. HIPAA violations have received a good deal of attention, with a well-known example being the doctor who was accused of posting a selfie with Joan Rivers, who was unconscious on the operating table. These examples, however, represent obvious violations of HIPAA and are infractions that most physicians would readily identify. Other examples may not be as obvious.
We know of one case where a nurse on the staff of a physicians’ office posted on Facebook that work was grueling that day because he felt under the weather with suspected flu. This may seem, at first, to be an innocuous communication. And that’s all it was, until, the son of an immunosuppressed man who had an appointment at that doctor’s office was flabbergasted to hear from a mutual friend that one of the nurses in the office was at work despite having the flu. He demanded to speak with the office manager and made sure that his father was not seen by that nurse. It may seem like an unlikely coincidence, but, in medical-legal circles, unlikely events occur all the time.
Many people who use social media will check in or post when they are out with friends or colleagues blowing off steam. For example, you might post something on social media about a holiday party you are attending. But, consider what happens if, at work the next day, something goes wrong, your care is called into question, and a lawsuit ensues. Your post may be innocent, but it now falls into the hands of the patient’s attorney. When you are having your deposition taken, the lawyer pulls your social media post out of a stack of papers and grills you about where you were, what you were doing, how late you stayed out, whether you were drinking, how much, and so on. Maybe you explain to him that you were only at the holiday party for an hour and did not have a single drink. That attorney, however, is not required to take your word for it and can ask you who you were with. All of a sudden, your friends and colleagues are being served with subpoenas for their depositions and being examined about what you did that night. Possibly, the lawyer is sending a subpoena for your credit card receipt and the restaurant’s billing records to determine what you ordered that night.
You should never rely on the false assumption that even “private” messages sent directly to other people will truly remain private. One of us was recently involved in a case where this worked to our advantage. A 30-year-old woman claimed that her family doctor never recommended that she see a gastroenterologist. A friend of the patient testified in a deposition that the two of them had discussed her medical care in private messages on Facebook. After the court ordered that the patient turn over her private Facebook messages, we learned that she told her friend that the doctor had indeed made the recommendation for her to see that specialist.
This cautionary tale doesn’t just apply to social media. Keep in mind that, if you are involved in litigation, attorneys can subpoena the records from your cellular phone provider. All cell phone text message are archived by the cellular provider and can be retrieved under subpoena. You may innocently be blowing off steam to a spouse or friend about a difficult patient or bad outcome but later have those text messages used against you in litigation.
The various social media platforms can be great tools for all kinds of professionals to share interesting information and further their professional development. However, everybody, especially the medical professional, needs to think before they post or send a message. We must also remember that, once information is out in cyberspace, it remains there and can never be truly erased. In other words, you can never unring the proverbial bell. It is important to think about the potential impact of that communication before posting and electronically communicating. Only communicate something that you would be comfortable defending in court.
Dr. Skolnik is professor of family and community medicine at Jefferson Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Mr. Shear is an associate attorney in the health care department at Marshall Dennehey Warner Coleman & Goggin in Pittsburgh. He represents physicians, medical professionals, and hospitals in medical malpractice actions.
Social media is now a part of everyday life. From Twitter, with its 140 character limit, to Facebook to Linkedin, there is a world of possibilities for communicating with friends, family, colleagues, and others online. Communication is good, but electronic media is a minefield for medical professionals who do not think carefully before they post.
The stories in the news about health care professionals who have posted obviously inflammatory material online, perhaps in a fit of rage, and have had their careers impacted or ended are just the tip of the iceberg. HIPAA violations have received a good deal of attention, with a well-known example being the doctor who was accused of posting a selfie with Joan Rivers, who was unconscious on the operating table. These examples, however, represent obvious violations of HIPAA and are infractions that most physicians would readily identify. Other examples may not be as obvious.
We know of one case where a nurse on the staff of a physicians’ office posted on Facebook that work was grueling that day because he felt under the weather with suspected flu. This may seem, at first, to be an innocuous communication. And that’s all it was, until, the son of an immunosuppressed man who had an appointment at that doctor’s office was flabbergasted to hear from a mutual friend that one of the nurses in the office was at work despite having the flu. He demanded to speak with the office manager and made sure that his father was not seen by that nurse. It may seem like an unlikely coincidence, but, in medical-legal circles, unlikely events occur all the time.
Many people who use social media will check in or post when they are out with friends or colleagues blowing off steam. For example, you might post something on social media about a holiday party you are attending. But, consider what happens if, at work the next day, something goes wrong, your care is called into question, and a lawsuit ensues. Your post may be innocent, but it now falls into the hands of the patient’s attorney. When you are having your deposition taken, the lawyer pulls your social media post out of a stack of papers and grills you about where you were, what you were doing, how late you stayed out, whether you were drinking, how much, and so on. Maybe you explain to him that you were only at the holiday party for an hour and did not have a single drink. That attorney, however, is not required to take your word for it and can ask you who you were with. All of a sudden, your friends and colleagues are being served with subpoenas for their depositions and being examined about what you did that night. Possibly, the lawyer is sending a subpoena for your credit card receipt and the restaurant’s billing records to determine what you ordered that night.
You should never rely on the false assumption that even “private” messages sent directly to other people will truly remain private. One of us was recently involved in a case where this worked to our advantage. A 30-year-old woman claimed that her family doctor never recommended that she see a gastroenterologist. A friend of the patient testified in a deposition that the two of them had discussed her medical care in private messages on Facebook. After the court ordered that the patient turn over her private Facebook messages, we learned that she told her friend that the doctor had indeed made the recommendation for her to see that specialist.
This cautionary tale doesn’t just apply to social media. Keep in mind that, if you are involved in litigation, attorneys can subpoena the records from your cellular phone provider. All cell phone text message are archived by the cellular provider and can be retrieved under subpoena. You may innocently be blowing off steam to a spouse or friend about a difficult patient or bad outcome but later have those text messages used against you in litigation.
The various social media platforms can be great tools for all kinds of professionals to share interesting information and further their professional development. However, everybody, especially the medical professional, needs to think before they post or send a message. We must also remember that, once information is out in cyberspace, it remains there and can never be truly erased. In other words, you can never unring the proverbial bell. It is important to think about the potential impact of that communication before posting and electronically communicating. Only communicate something that you would be comfortable defending in court.
Dr. Skolnik is professor of family and community medicine at Jefferson Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Mr. Shear is an associate attorney in the health care department at Marshall Dennehey Warner Coleman & Goggin in Pittsburgh. He represents physicians, medical professionals, and hospitals in medical malpractice actions.
Social media is now a part of everyday life. From Twitter, with its 140 character limit, to Facebook to Linkedin, there is a world of possibilities for communicating with friends, family, colleagues, and others online. Communication is good, but electronic media is a minefield for medical professionals who do not think carefully before they post.
The stories in the news about health care professionals who have posted obviously inflammatory material online, perhaps in a fit of rage, and have had their careers impacted or ended are just the tip of the iceberg. HIPAA violations have received a good deal of attention, with a well-known example being the doctor who was accused of posting a selfie with Joan Rivers, who was unconscious on the operating table. These examples, however, represent obvious violations of HIPAA and are infractions that most physicians would readily identify. Other examples may not be as obvious.
We know of one case where a nurse on the staff of a physicians’ office posted on Facebook that work was grueling that day because he felt under the weather with suspected flu. This may seem, at first, to be an innocuous communication. And that’s all it was, until, the son of an immunosuppressed man who had an appointment at that doctor’s office was flabbergasted to hear from a mutual friend that one of the nurses in the office was at work despite having the flu. He demanded to speak with the office manager and made sure that his father was not seen by that nurse. It may seem like an unlikely coincidence, but, in medical-legal circles, unlikely events occur all the time.
Many people who use social media will check in or post when they are out with friends or colleagues blowing off steam. For example, you might post something on social media about a holiday party you are attending. But, consider what happens if, at work the next day, something goes wrong, your care is called into question, and a lawsuit ensues. Your post may be innocent, but it now falls into the hands of the patient’s attorney. When you are having your deposition taken, the lawyer pulls your social media post out of a stack of papers and grills you about where you were, what you were doing, how late you stayed out, whether you were drinking, how much, and so on. Maybe you explain to him that you were only at the holiday party for an hour and did not have a single drink. That attorney, however, is not required to take your word for it and can ask you who you were with. All of a sudden, your friends and colleagues are being served with subpoenas for their depositions and being examined about what you did that night. Possibly, the lawyer is sending a subpoena for your credit card receipt and the restaurant’s billing records to determine what you ordered that night.
You should never rely on the false assumption that even “private” messages sent directly to other people will truly remain private. One of us was recently involved in a case where this worked to our advantage. A 30-year-old woman claimed that her family doctor never recommended that she see a gastroenterologist. A friend of the patient testified in a deposition that the two of them had discussed her medical care in private messages on Facebook. After the court ordered that the patient turn over her private Facebook messages, we learned that she told her friend that the doctor had indeed made the recommendation for her to see that specialist.
This cautionary tale doesn’t just apply to social media. Keep in mind that, if you are involved in litigation, attorneys can subpoena the records from your cellular phone provider. All cell phone text message are archived by the cellular provider and can be retrieved under subpoena. You may innocently be blowing off steam to a spouse or friend about a difficult patient or bad outcome but later have those text messages used against you in litigation.
The various social media platforms can be great tools for all kinds of professionals to share interesting information and further their professional development. However, everybody, especially the medical professional, needs to think before they post or send a message. We must also remember that, once information is out in cyberspace, it remains there and can never be truly erased. In other words, you can never unring the proverbial bell. It is important to think about the potential impact of that communication before posting and electronically communicating. Only communicate something that you would be comfortable defending in court.
Dr. Skolnik is professor of family and community medicine at Jefferson Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Jefferson Health. Mr. Shear is an associate attorney in the health care department at Marshall Dennehey Warner Coleman & Goggin in Pittsburgh. He represents physicians, medical professionals, and hospitals in medical malpractice actions.
What, you never filled the prescription?!
How many times do you come out of the exam room after seeing a patient in follow-up and heave a sigh because the parents did not give their child the medicine as you prescribed it?
Without adherence to the medication plan, a lot of suboptimal outcomes can and do occur. A urinary tract infection may come back partially treated, requiring a more extensive work-up. A strep infection may spread to family members. Inflammatory bowel disease may require bowel resection. Asthma may simmer with long-term inflammation and pulmonary compromise as well as concurrent activity limitations. Often children with asthma are given less than 50% of prescribed controller medicines. In one pediatric study, medication adherence was not even asked about in 66% of cases. In adults, 20%-30% of prescriptions are never filled.
As physicians, we are carefully schooled in making complex diagnoses, sorting out and prioritizing the laboratory work-up, and memorizing the latest and most effective treatment regimens. What is rarely taught, however, is how to conduct the conversation needed to optimize subsequent adherence to the medication plan.
Problem-solving counseling is an evidence-based method to improve medication adherence. This is a semistructured form of cognitive-behavioral intervention designed to engage the responsible person (parent or child) in shared decision making about whether and how to take medication, and which one to take. After all, for good or for bad, it is really their choice!
1. Problem definition. This step involves developing a clear and specific definition of the problem. Educating families about a medical condition has to start with asking what they already know. This often includes sagas of bad outcomes in relatives. Ask: Who do you know with asthma? How was it for them? The family needs to know symptoms, simple pathophysiology (such as inflammation you can’t see or feel), course, and prognosis. They also need to know where their child’s condition falls on the continuum. And they need to understand the essential prevention aspect of controllers in what appears to be an asymptomatic child. Failure to communicate this is a common reason for nonadherence in asthma.
2. Generation of alternatives. This involves brainstorming to identify multiple and creative solutions. This step will reveal past experiences as well as things the family learned on the Internet that may be true and relevant, or true but irrelevant, or false. Ask: What have you heard about treatments for asthma? What do you think would be best for your child? Generic handouts with a sampling of medicines, advantages and disadvantages, side effects, and costs of the main choices have been shown to be helpful guides that enhance adherence through empowering the family in their choice and reassuring family members that you have been thorough. It can be a balancing act to describe possible side effects without scaring the family into shutting down and being unable to make any choice at all. However, failure to discuss common effects they may notice – such as a racing heart from rescue medications – but that you think of as trivial, may also lead to nonadherence. A way to communicate about perceived side effects and manage them has to be part of an effective plan. Planning a phone or email check-in can make a big difference.
3. Decision making. This step involves evaluating all the solutions to identify the most effective and feasible option. Once the family understands the problem and the alternatives, it is crucial for you, as the physician, to not only ask their preference but be ready to suggest what you think would be best. While not wanting to be patronized, families want your opinion. I like to have family members close their eyes and visualize carrying out the selected routine. This is a good hypnotic technique for future remembering, but you also may discover important facts by this simple exercise, such as that the child gets up alone for school, making morning dosing unreliable. Shared decision making is not a way to abdicate your expert opinion, just to incorporate family preferences and factors.
4. Solution implementation. This step involves carrying out the plan. There is no substitute for a real life trial! There may be surprising issues: Autistic children may be afraid of a nebulizer machine. Sensitive children may refuse the flavor of some inhalers.
5. Solution verification. Evaluate the effectiveness of the solution and modify the plan as necessary. Follow-up contact is crucial, especially at the start of a new chronic medication plan. When families know that the plan can be changed if things do not go well or they change their minds, they will be less fearful of giving it a try and more honest about barriers they perceive or encounter rather than simply showing up at the next visit with the child’s condition out of control.
Although using problem-solving counseling may sound complicated, it is intended to be focused and brief, and has been shown to be feasibly done in the clinic by primary care providers, without lengthening the visit. CHADIS even has teleprompter text specific to parent-reported barriers to help you.
Even when family members understand and agrees to a medication plan during the visit, there are a variety of reasons they may not adhere to it. They may forget to give the medicine, be unable to afford it once prescribed, experience unpleasant side effects, encounter resistance from the child, or get unanticipated push back from family members. All of these issues can be addressed if you know that they happen. You just have to ask! Recommending smartphone reminders or reminder apps (such as Medisafe), using GoodRx to find cheaper sources, suggesting candy as a chaser, recommending behavior strategies for feisty kids, and providing written materials (or a phone call) for reluctant relatives are strategies you can prepare in advance to have in your quiver and are well worth your time.
If it weren’t enough to address adherence to optimize outcomes, asthma management and control will likely be a Clinical Quality Measure, determining how we will be paid starting this year. Now you have a tool to do it!
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to Frontline Medical News.
How many times do you come out of the exam room after seeing a patient in follow-up and heave a sigh because the parents did not give their child the medicine as you prescribed it?
Without adherence to the medication plan, a lot of suboptimal outcomes can and do occur. A urinary tract infection may come back partially treated, requiring a more extensive work-up. A strep infection may spread to family members. Inflammatory bowel disease may require bowel resection. Asthma may simmer with long-term inflammation and pulmonary compromise as well as concurrent activity limitations. Often children with asthma are given less than 50% of prescribed controller medicines. In one pediatric study, medication adherence was not even asked about in 66% of cases. In adults, 20%-30% of prescriptions are never filled.
As physicians, we are carefully schooled in making complex diagnoses, sorting out and prioritizing the laboratory work-up, and memorizing the latest and most effective treatment regimens. What is rarely taught, however, is how to conduct the conversation needed to optimize subsequent adherence to the medication plan.
Problem-solving counseling is an evidence-based method to improve medication adherence. This is a semistructured form of cognitive-behavioral intervention designed to engage the responsible person (parent or child) in shared decision making about whether and how to take medication, and which one to take. After all, for good or for bad, it is really their choice!
1. Problem definition. This step involves developing a clear and specific definition of the problem. Educating families about a medical condition has to start with asking what they already know. This often includes sagas of bad outcomes in relatives. Ask: Who do you know with asthma? How was it for them? The family needs to know symptoms, simple pathophysiology (such as inflammation you can’t see or feel), course, and prognosis. They also need to know where their child’s condition falls on the continuum. And they need to understand the essential prevention aspect of controllers in what appears to be an asymptomatic child. Failure to communicate this is a common reason for nonadherence in asthma.
2. Generation of alternatives. This involves brainstorming to identify multiple and creative solutions. This step will reveal past experiences as well as things the family learned on the Internet that may be true and relevant, or true but irrelevant, or false. Ask: What have you heard about treatments for asthma? What do you think would be best for your child? Generic handouts with a sampling of medicines, advantages and disadvantages, side effects, and costs of the main choices have been shown to be helpful guides that enhance adherence through empowering the family in their choice and reassuring family members that you have been thorough. It can be a balancing act to describe possible side effects without scaring the family into shutting down and being unable to make any choice at all. However, failure to discuss common effects they may notice – such as a racing heart from rescue medications – but that you think of as trivial, may also lead to nonadherence. A way to communicate about perceived side effects and manage them has to be part of an effective plan. Planning a phone or email check-in can make a big difference.
3. Decision making. This step involves evaluating all the solutions to identify the most effective and feasible option. Once the family understands the problem and the alternatives, it is crucial for you, as the physician, to not only ask their preference but be ready to suggest what you think would be best. While not wanting to be patronized, families want your opinion. I like to have family members close their eyes and visualize carrying out the selected routine. This is a good hypnotic technique for future remembering, but you also may discover important facts by this simple exercise, such as that the child gets up alone for school, making morning dosing unreliable. Shared decision making is not a way to abdicate your expert opinion, just to incorporate family preferences and factors.
4. Solution implementation. This step involves carrying out the plan. There is no substitute for a real life trial! There may be surprising issues: Autistic children may be afraid of a nebulizer machine. Sensitive children may refuse the flavor of some inhalers.
5. Solution verification. Evaluate the effectiveness of the solution and modify the plan as necessary. Follow-up contact is crucial, especially at the start of a new chronic medication plan. When families know that the plan can be changed if things do not go well or they change their minds, they will be less fearful of giving it a try and more honest about barriers they perceive or encounter rather than simply showing up at the next visit with the child’s condition out of control.
Although using problem-solving counseling may sound complicated, it is intended to be focused and brief, and has been shown to be feasibly done in the clinic by primary care providers, without lengthening the visit. CHADIS even has teleprompter text specific to parent-reported barriers to help you.
Even when family members understand and agrees to a medication plan during the visit, there are a variety of reasons they may not adhere to it. They may forget to give the medicine, be unable to afford it once prescribed, experience unpleasant side effects, encounter resistance from the child, or get unanticipated push back from family members. All of these issues can be addressed if you know that they happen. You just have to ask! Recommending smartphone reminders or reminder apps (such as Medisafe), using GoodRx to find cheaper sources, suggesting candy as a chaser, recommending behavior strategies for feisty kids, and providing written materials (or a phone call) for reluctant relatives are strategies you can prepare in advance to have in your quiver and are well worth your time.
If it weren’t enough to address adherence to optimize outcomes, asthma management and control will likely be a Clinical Quality Measure, determining how we will be paid starting this year. Now you have a tool to do it!
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to Frontline Medical News.
How many times do you come out of the exam room after seeing a patient in follow-up and heave a sigh because the parents did not give their child the medicine as you prescribed it?
Without adherence to the medication plan, a lot of suboptimal outcomes can and do occur. A urinary tract infection may come back partially treated, requiring a more extensive work-up. A strep infection may spread to family members. Inflammatory bowel disease may require bowel resection. Asthma may simmer with long-term inflammation and pulmonary compromise as well as concurrent activity limitations. Often children with asthma are given less than 50% of prescribed controller medicines. In one pediatric study, medication adherence was not even asked about in 66% of cases. In adults, 20%-30% of prescriptions are never filled.
As physicians, we are carefully schooled in making complex diagnoses, sorting out and prioritizing the laboratory work-up, and memorizing the latest and most effective treatment regimens. What is rarely taught, however, is how to conduct the conversation needed to optimize subsequent adherence to the medication plan.
Problem-solving counseling is an evidence-based method to improve medication adherence. This is a semistructured form of cognitive-behavioral intervention designed to engage the responsible person (parent or child) in shared decision making about whether and how to take medication, and which one to take. After all, for good or for bad, it is really their choice!
1. Problem definition. This step involves developing a clear and specific definition of the problem. Educating families about a medical condition has to start with asking what they already know. This often includes sagas of bad outcomes in relatives. Ask: Who do you know with asthma? How was it for them? The family needs to know symptoms, simple pathophysiology (such as inflammation you can’t see or feel), course, and prognosis. They also need to know where their child’s condition falls on the continuum. And they need to understand the essential prevention aspect of controllers in what appears to be an asymptomatic child. Failure to communicate this is a common reason for nonadherence in asthma.
2. Generation of alternatives. This involves brainstorming to identify multiple and creative solutions. This step will reveal past experiences as well as things the family learned on the Internet that may be true and relevant, or true but irrelevant, or false. Ask: What have you heard about treatments for asthma? What do you think would be best for your child? Generic handouts with a sampling of medicines, advantages and disadvantages, side effects, and costs of the main choices have been shown to be helpful guides that enhance adherence through empowering the family in their choice and reassuring family members that you have been thorough. It can be a balancing act to describe possible side effects without scaring the family into shutting down and being unable to make any choice at all. However, failure to discuss common effects they may notice – such as a racing heart from rescue medications – but that you think of as trivial, may also lead to nonadherence. A way to communicate about perceived side effects and manage them has to be part of an effective plan. Planning a phone or email check-in can make a big difference.
3. Decision making. This step involves evaluating all the solutions to identify the most effective and feasible option. Once the family understands the problem and the alternatives, it is crucial for you, as the physician, to not only ask their preference but be ready to suggest what you think would be best. While not wanting to be patronized, families want your opinion. I like to have family members close their eyes and visualize carrying out the selected routine. This is a good hypnotic technique for future remembering, but you also may discover important facts by this simple exercise, such as that the child gets up alone for school, making morning dosing unreliable. Shared decision making is not a way to abdicate your expert opinion, just to incorporate family preferences and factors.
4. Solution implementation. This step involves carrying out the plan. There is no substitute for a real life trial! There may be surprising issues: Autistic children may be afraid of a nebulizer machine. Sensitive children may refuse the flavor of some inhalers.
5. Solution verification. Evaluate the effectiveness of the solution and modify the plan as necessary. Follow-up contact is crucial, especially at the start of a new chronic medication plan. When families know that the plan can be changed if things do not go well or they change their minds, they will be less fearful of giving it a try and more honest about barriers they perceive or encounter rather than simply showing up at the next visit with the child’s condition out of control.
Although using problem-solving counseling may sound complicated, it is intended to be focused and brief, and has been shown to be feasibly done in the clinic by primary care providers, without lengthening the visit. CHADIS even has teleprompter text specific to parent-reported barriers to help you.
Even when family members understand and agrees to a medication plan during the visit, there are a variety of reasons they may not adhere to it. They may forget to give the medicine, be unable to afford it once prescribed, experience unpleasant side effects, encounter resistance from the child, or get unanticipated push back from family members. All of these issues can be addressed if you know that they happen. You just have to ask! Recommending smartphone reminders or reminder apps (such as Medisafe), using GoodRx to find cheaper sources, suggesting candy as a chaser, recommending behavior strategies for feisty kids, and providing written materials (or a phone call) for reluctant relatives are strategies you can prepare in advance to have in your quiver and are well worth your time.
If it weren’t enough to address adherence to optimize outcomes, asthma management and control will likely be a Clinical Quality Measure, determining how we will be paid starting this year. Now you have a tool to do it!
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to Frontline Medical News.
Medicine’s revenge against traveler’s diarrhea
Traveler’s diarrhea (TD) is the most common illness in people traveling from resource-advantaged countries to resource-limited regions of the globe. Approximately 50% of these types of travelers will contract diarrhea.
I knew of a group of infectious disease experts traveling to India together – presumably well-versed in how to avoid such things – and one-half of the group developed it.
I have many patients sending me electronic messages asking me for their standard 3-day ciprofloxacin prescriptions, “just in case.” I am guilty of having provided this, along with warnings that we could make matters worse by giving them Clostridium difficile colitis. Antibiotics may also increase the risk for post-TD irritable bowel syndrome, which can occur in up to 15% of patients.
Mild TD is defined as passage of one or two unformed stools in 24 hours without nausea, vomiting, abdominal pain, fever, or blood in stools. What is the evidence for the effectiveness of antibiotics, compared with other interventions such as loperamide, for mild TD?
Tinja Lääveri, MD, of the University of Helsinki, and colleagues conducted a systematic review on the efficacy and safety of loperamide for TD (Travel Med Infect Dis. 2016 Jul-Aug;14[4]:299-312). Fifteen articles were retrieved.
Loperamide was observed to be noninferior to antibiotics for the treatment of TD. In one study, loperamide was observed to be superior to bismuth, which is commonly recommended to prevent TD. Adverse events with loperamide occurred predominantly among patients with bloody diarrhea.
The authors remind us that loperamide is different from antimotility agents such as diphenoxylate with atropine, as loperamide has an antisecretory effect at lower doses and decreases motility only at higher doses.
If you subscribe to the idea that diarrhea helps rid the body of toxins, be reminded that secretory diarrhea is exploited by the organism to spread the infestation to as many humans as possible.
The recommended regimen for loperamide is a 4-mg loading dose, followed by 2 mg after every episode of diarrhea. Tell your patients not to use loperamide if they are having fever or bloody diarrhea, or if you suspect they could have C. difficile colitis (that is, recent antibiotics or other risk factors).
Happy travels.
Dr. Ebbert is professor of medicine, a general internist at the Mayo Clinic in Rochester, Minn., and a diplomate of the American Board of Addiction Medicine. The opinions expressed are those of the author and do not necessarily represent the views and opinions of the Mayo Clinic. The opinions expressed in this article should not be used to diagnose or treat any medical condition, nor should they be used as a substitute for medical advice from a qualified, board-certified practicing clinician. Dr. Ebbert has no relevant financial disclosures about this article.
Traveler’s diarrhea (TD) is the most common illness in people traveling from resource-advantaged countries to resource-limited regions of the globe. Approximately 50% of these types of travelers will contract diarrhea.
I knew of a group of infectious disease experts traveling to India together – presumably well-versed in how to avoid such things – and one-half of the group developed it.
I have many patients sending me electronic messages asking me for their standard 3-day ciprofloxacin prescriptions, “just in case.” I am guilty of having provided this, along with warnings that we could make matters worse by giving them Clostridium difficile colitis. Antibiotics may also increase the risk for post-TD irritable bowel syndrome, which can occur in up to 15% of patients.
Mild TD is defined as passage of one or two unformed stools in 24 hours without nausea, vomiting, abdominal pain, fever, or blood in stools. What is the evidence for the effectiveness of antibiotics, compared with other interventions such as loperamide, for mild TD?
Tinja Lääveri, MD, of the University of Helsinki, and colleagues conducted a systematic review on the efficacy and safety of loperamide for TD (Travel Med Infect Dis. 2016 Jul-Aug;14[4]:299-312). Fifteen articles were retrieved.
Loperamide was observed to be noninferior to antibiotics for the treatment of TD. In one study, loperamide was observed to be superior to bismuth, which is commonly recommended to prevent TD. Adverse events with loperamide occurred predominantly among patients with bloody diarrhea.
The authors remind us that loperamide is different from antimotility agents such as diphenoxylate with atropine, as loperamide has an antisecretory effect at lower doses and decreases motility only at higher doses.
If you subscribe to the idea that diarrhea helps rid the body of toxins, be reminded that secretory diarrhea is exploited by the organism to spread the infestation to as many humans as possible.
The recommended regimen for loperamide is a 4-mg loading dose, followed by 2 mg after every episode of diarrhea. Tell your patients not to use loperamide if they are having fever or bloody diarrhea, or if you suspect they could have C. difficile colitis (that is, recent antibiotics or other risk factors).
Happy travels.
Dr. Ebbert is professor of medicine, a general internist at the Mayo Clinic in Rochester, Minn., and a diplomate of the American Board of Addiction Medicine. The opinions expressed are those of the author and do not necessarily represent the views and opinions of the Mayo Clinic. The opinions expressed in this article should not be used to diagnose or treat any medical condition, nor should they be used as a substitute for medical advice from a qualified, board-certified practicing clinician. Dr. Ebbert has no relevant financial disclosures about this article.
Traveler’s diarrhea (TD) is the most common illness in people traveling from resource-advantaged countries to resource-limited regions of the globe. Approximately 50% of these types of travelers will contract diarrhea.
I knew of a group of infectious disease experts traveling to India together – presumably well-versed in how to avoid such things – and one-half of the group developed it.
I have many patients sending me electronic messages asking me for their standard 3-day ciprofloxacin prescriptions, “just in case.” I am guilty of having provided this, along with warnings that we could make matters worse by giving them Clostridium difficile colitis. Antibiotics may also increase the risk for post-TD irritable bowel syndrome, which can occur in up to 15% of patients.
Mild TD is defined as passage of one or two unformed stools in 24 hours without nausea, vomiting, abdominal pain, fever, or blood in stools. What is the evidence for the effectiveness of antibiotics, compared with other interventions such as loperamide, for mild TD?
Tinja Lääveri, MD, of the University of Helsinki, and colleagues conducted a systematic review on the efficacy and safety of loperamide for TD (Travel Med Infect Dis. 2016 Jul-Aug;14[4]:299-312). Fifteen articles were retrieved.
Loperamide was observed to be noninferior to antibiotics for the treatment of TD. In one study, loperamide was observed to be superior to bismuth, which is commonly recommended to prevent TD. Adverse events with loperamide occurred predominantly among patients with bloody diarrhea.
The authors remind us that loperamide is different from antimotility agents such as diphenoxylate with atropine, as loperamide has an antisecretory effect at lower doses and decreases motility only at higher doses.
If you subscribe to the idea that diarrhea helps rid the body of toxins, be reminded that secretory diarrhea is exploited by the organism to spread the infestation to as many humans as possible.
The recommended regimen for loperamide is a 4-mg loading dose, followed by 2 mg after every episode of diarrhea. Tell your patients not to use loperamide if they are having fever or bloody diarrhea, or if you suspect they could have C. difficile colitis (that is, recent antibiotics or other risk factors).
Happy travels.
Dr. Ebbert is professor of medicine, a general internist at the Mayo Clinic in Rochester, Minn., and a diplomate of the American Board of Addiction Medicine. The opinions expressed are those of the author and do not necessarily represent the views and opinions of the Mayo Clinic. The opinions expressed in this article should not be used to diagnose or treat any medical condition, nor should they be used as a substitute for medical advice from a qualified, board-certified practicing clinician. Dr. Ebbert has no relevant financial disclosures about this article.
International travel vaccination updates
There are several things you should know about necessary vaccinations, and sometimes potential supply problems, if your families will be traveling internationally.
Yellow fever and vaccine supply
Yellow fever is caused by a Flavivirus transmitted by the bite of an infected mosquito. It occurs in sub-Saharan Africa and in tropical areas in South America. Multiple factors determine a traveler’s risk for acquisition, including destination, season, duration of potential exposure, activities, and the local transmission rate. The majority of those infected are asymptomatic or have minimal clinical symptoms. The incubation period is 3-6 days, which is then followed by an influenza-like illness. Approximately 15% of infected individuals develop more serious symptoms including jaundice, hemorrhagic symptoms, shock, and, ultimately, multiorgan system failure with a fatality rate of 90%. There is no specific treatment.
Previously, vaccine boosters were required every 10 years. However, the duration of immunity was extensively reviewed by the World Health Organization and effective July 11, 2016, boosters are no longer required. A single dose of vaccine is now valid for the lifetime of the individual. This includes those persons vaccinated prior to July 11, 2016. Since it is a live vaccine, administration is contraindicated in certain individuals. Exemption letters are provided for those who have a medical contraindication.
Caution is advised in persons receiving their initial dose of YF-VAX who are older than 60 years of age because they have an increased risk of serious side effects. This is not a concern for the pediatrician. The vaccine can only be administered at state approved facilities. It is one vaccine that is not only recommended, but may be required for entry into certain countries. Go to www.cdc.gov/yellowfever for a complete list.
Sanofi Pasteur is the only U.S. manufacturer of YF-VAX. Production has ceased until mid-2018, when a new manufacturing facility will open. Current supplies are anticipated to be depleted by mid-2017, and orders have been limited to 5 doses per month. Sanofi Pasteur, in conjunction with the Food and Drug Administration, will make Stamaril – a yellow fever vaccine manufactured by the company in France and licensed in over 70 countries – available to U.S. travelers through an Expanded Access Investigational New Drug Application. Details on how and when this program will be operational are forthcoming. What is known is that, nationwide, there will be a limited number of sites administering Stamaril. Once finalized, a list of locations will be posted on the CDC Yellow Fever site.
How does this affect your patients? If travel to a yellow fever risk area is anticipated, they should not delay in seeking pretravel advice and immunizations until the last minute. Individual clinic inventories will not be stable. Postponing a trip or changing destinations is preferred if the vaccine is not available. Yellow fever exemption letters are only provided for those persons who have a medical contraindication to receive YF-VAX.
Zika, dengue, and chikungunya
These three Flaviviruses all are transmitted by mosquitoes and can present with fever, rash, and headache. Their distribution is overlapping in several parts of the world. Most infected people are asymptomatic. If symptoms develop, they usually are self-limited. Disease prevention is by mosquito avoidance. There are no preventive vaccines.
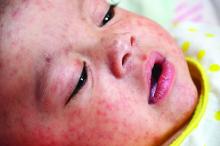
Zika virus is the only one associated with a congenital syndrome. It is characterized by brain abnormalities with or without microcephaly, neural tube defects, and ocular abnormalities.
Guidelines for the evaluation and management of Zika virus–exposed infants were initially published in January, 2016, with the most recent update published in August 2016 (MMWR Morb Mortal Wkly Rep. 2016 Aug 26;65[33]:870-8).
Preliminary data from the U.S. Zika pregnancy registry of 442 completed pregnancies between Jan. 15 to Sept. 22, 2016, identified birth defects in 26 fetuses/ infants (6%). There were 21 infants with birth defects among 395 live births and 5 fetuses with birth defects among 47 pregnancy losses. Birth defects were reported for 16 of 271 (6%) asymptomatic and 10 of 167 (6%) symptomatic women. There were no birth defects in infants when exposure occurred after the first trimester. Of the 26 affected infants, 4 had microcephaly and no neuroimaging and 3 (12%) had no fetal or infant testing. Approximately 41% (82/442) of infants did not have Zika virus testing (JAMA. 2017 Jan 3;317[1]:59-68).
It is unclear why testing was not performed. One concern is that the pediatrician may not have been aware of the maternal Zika virus exposure or test results. It may behoove us to begin asking questions about parental international travel to provide optimal management for our patients. We also should be familiar with the current guidelines for evaluating any potentially exposed infants, which include postnatal neuroimaging, Zika virus testing, a comprehensive newborn examination including neurologic exam, and a standard newborn hearing screen prior to hospital discharge.
Regardless of maternal Zika virus test results, infants with any clinical findings suggestive of congenital Zika virus syndrome and possible maternal exposure based on epidemiologic link also should be tested. Zika virus travel alerts and the most up to date information can be found on the Centers for Disease Control and Prevention website (www. cdc.gov/Zika).
Measles
Although endemic measles was eliminated in the United States in 2000, it is still common in many countries in Europe, Africa, and the Pacific. Most cases in the United States occur in unvaccinated individuals, with 78 cases reported in 2016. As of March 25, 2017, 28 cases have been reported. At least 10 countries – including Belgium, France, Italy, Germany, Portugal, and Thailand – have reported outbreaks of measles since April 2017. As reminder, all children aged 6-11 months should receive one dose of MMR and those 12 months or older should receive two doses of MMR at least 28 days apart if international travel is planned. Adults born after 1956 also should have received two doses of MMR prior to international travel.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She reported having no relevant financial disclosures.
There are several things you should know about necessary vaccinations, and sometimes potential supply problems, if your families will be traveling internationally.
Yellow fever and vaccine supply
Yellow fever is caused by a Flavivirus transmitted by the bite of an infected mosquito. It occurs in sub-Saharan Africa and in tropical areas in South America. Multiple factors determine a traveler’s risk for acquisition, including destination, season, duration of potential exposure, activities, and the local transmission rate. The majority of those infected are asymptomatic or have minimal clinical symptoms. The incubation period is 3-6 days, which is then followed by an influenza-like illness. Approximately 15% of infected individuals develop more serious symptoms including jaundice, hemorrhagic symptoms, shock, and, ultimately, multiorgan system failure with a fatality rate of 90%. There is no specific treatment.
Previously, vaccine boosters were required every 10 years. However, the duration of immunity was extensively reviewed by the World Health Organization and effective July 11, 2016, boosters are no longer required. A single dose of vaccine is now valid for the lifetime of the individual. This includes those persons vaccinated prior to July 11, 2016. Since it is a live vaccine, administration is contraindicated in certain individuals. Exemption letters are provided for those who have a medical contraindication.
Caution is advised in persons receiving their initial dose of YF-VAX who are older than 60 years of age because they have an increased risk of serious side effects. This is not a concern for the pediatrician. The vaccine can only be administered at state approved facilities. It is one vaccine that is not only recommended, but may be required for entry into certain countries. Go to www.cdc.gov/yellowfever for a complete list.
Sanofi Pasteur is the only U.S. manufacturer of YF-VAX. Production has ceased until mid-2018, when a new manufacturing facility will open. Current supplies are anticipated to be depleted by mid-2017, and orders have been limited to 5 doses per month. Sanofi Pasteur, in conjunction with the Food and Drug Administration, will make Stamaril – a yellow fever vaccine manufactured by the company in France and licensed in over 70 countries – available to U.S. travelers through an Expanded Access Investigational New Drug Application. Details on how and when this program will be operational are forthcoming. What is known is that, nationwide, there will be a limited number of sites administering Stamaril. Once finalized, a list of locations will be posted on the CDC Yellow Fever site.
How does this affect your patients? If travel to a yellow fever risk area is anticipated, they should not delay in seeking pretravel advice and immunizations until the last minute. Individual clinic inventories will not be stable. Postponing a trip or changing destinations is preferred if the vaccine is not available. Yellow fever exemption letters are only provided for those persons who have a medical contraindication to receive YF-VAX.
Zika, dengue, and chikungunya
These three Flaviviruses all are transmitted by mosquitoes and can present with fever, rash, and headache. Their distribution is overlapping in several parts of the world. Most infected people are asymptomatic. If symptoms develop, they usually are self-limited. Disease prevention is by mosquito avoidance. There are no preventive vaccines.
Zika virus is the only one associated with a congenital syndrome. It is characterized by brain abnormalities with or without microcephaly, neural tube defects, and ocular abnormalities.
Guidelines for the evaluation and management of Zika virus–exposed infants were initially published in January, 2016, with the most recent update published in August 2016 (MMWR Morb Mortal Wkly Rep. 2016 Aug 26;65[33]:870-8).
Preliminary data from the U.S. Zika pregnancy registry of 442 completed pregnancies between Jan. 15 to Sept. 22, 2016, identified birth defects in 26 fetuses/ infants (6%). There were 21 infants with birth defects among 395 live births and 5 fetuses with birth defects among 47 pregnancy losses. Birth defects were reported for 16 of 271 (6%) asymptomatic and 10 of 167 (6%) symptomatic women. There were no birth defects in infants when exposure occurred after the first trimester. Of the 26 affected infants, 4 had microcephaly and no neuroimaging and 3 (12%) had no fetal or infant testing. Approximately 41% (82/442) of infants did not have Zika virus testing (JAMA. 2017 Jan 3;317[1]:59-68).
It is unclear why testing was not performed. One concern is that the pediatrician may not have been aware of the maternal Zika virus exposure or test results. It may behoove us to begin asking questions about parental international travel to provide optimal management for our patients. We also should be familiar with the current guidelines for evaluating any potentially exposed infants, which include postnatal neuroimaging, Zika virus testing, a comprehensive newborn examination including neurologic exam, and a standard newborn hearing screen prior to hospital discharge.
Regardless of maternal Zika virus test results, infants with any clinical findings suggestive of congenital Zika virus syndrome and possible maternal exposure based on epidemiologic link also should be tested. Zika virus travel alerts and the most up to date information can be found on the Centers for Disease Control and Prevention website (www. cdc.gov/Zika).
Measles
Although endemic measles was eliminated in the United States in 2000, it is still common in many countries in Europe, Africa, and the Pacific. Most cases in the United States occur in unvaccinated individuals, with 78 cases reported in 2016. As of March 25, 2017, 28 cases have been reported. At least 10 countries – including Belgium, France, Italy, Germany, Portugal, and Thailand – have reported outbreaks of measles since April 2017. As reminder, all children aged 6-11 months should receive one dose of MMR and those 12 months or older should receive two doses of MMR at least 28 days apart if international travel is planned. Adults born after 1956 also should have received two doses of MMR prior to international travel.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She reported having no relevant financial disclosures.
There are several things you should know about necessary vaccinations, and sometimes potential supply problems, if your families will be traveling internationally.
Yellow fever and vaccine supply
Yellow fever is caused by a Flavivirus transmitted by the bite of an infected mosquito. It occurs in sub-Saharan Africa and in tropical areas in South America. Multiple factors determine a traveler’s risk for acquisition, including destination, season, duration of potential exposure, activities, and the local transmission rate. The majority of those infected are asymptomatic or have minimal clinical symptoms. The incubation period is 3-6 days, which is then followed by an influenza-like illness. Approximately 15% of infected individuals develop more serious symptoms including jaundice, hemorrhagic symptoms, shock, and, ultimately, multiorgan system failure with a fatality rate of 90%. There is no specific treatment.
Previously, vaccine boosters were required every 10 years. However, the duration of immunity was extensively reviewed by the World Health Organization and effective July 11, 2016, boosters are no longer required. A single dose of vaccine is now valid for the lifetime of the individual. This includes those persons vaccinated prior to July 11, 2016. Since it is a live vaccine, administration is contraindicated in certain individuals. Exemption letters are provided for those who have a medical contraindication.
Caution is advised in persons receiving their initial dose of YF-VAX who are older than 60 years of age because they have an increased risk of serious side effects. This is not a concern for the pediatrician. The vaccine can only be administered at state approved facilities. It is one vaccine that is not only recommended, but may be required for entry into certain countries. Go to www.cdc.gov/yellowfever for a complete list.
Sanofi Pasteur is the only U.S. manufacturer of YF-VAX. Production has ceased until mid-2018, when a new manufacturing facility will open. Current supplies are anticipated to be depleted by mid-2017, and orders have been limited to 5 doses per month. Sanofi Pasteur, in conjunction with the Food and Drug Administration, will make Stamaril – a yellow fever vaccine manufactured by the company in France and licensed in over 70 countries – available to U.S. travelers through an Expanded Access Investigational New Drug Application. Details on how and when this program will be operational are forthcoming. What is known is that, nationwide, there will be a limited number of sites administering Stamaril. Once finalized, a list of locations will be posted on the CDC Yellow Fever site.
How does this affect your patients? If travel to a yellow fever risk area is anticipated, they should not delay in seeking pretravel advice and immunizations until the last minute. Individual clinic inventories will not be stable. Postponing a trip or changing destinations is preferred if the vaccine is not available. Yellow fever exemption letters are only provided for those persons who have a medical contraindication to receive YF-VAX.
Zika, dengue, and chikungunya
These three Flaviviruses all are transmitted by mosquitoes and can present with fever, rash, and headache. Their distribution is overlapping in several parts of the world. Most infected people are asymptomatic. If symptoms develop, they usually are self-limited. Disease prevention is by mosquito avoidance. There are no preventive vaccines.
Zika virus is the only one associated with a congenital syndrome. It is characterized by brain abnormalities with or without microcephaly, neural tube defects, and ocular abnormalities.
Guidelines for the evaluation and management of Zika virus–exposed infants were initially published in January, 2016, with the most recent update published in August 2016 (MMWR Morb Mortal Wkly Rep. 2016 Aug 26;65[33]:870-8).
Preliminary data from the U.S. Zika pregnancy registry of 442 completed pregnancies between Jan. 15 to Sept. 22, 2016, identified birth defects in 26 fetuses/ infants (6%). There were 21 infants with birth defects among 395 live births and 5 fetuses with birth defects among 47 pregnancy losses. Birth defects were reported for 16 of 271 (6%) asymptomatic and 10 of 167 (6%) symptomatic women. There were no birth defects in infants when exposure occurred after the first trimester. Of the 26 affected infants, 4 had microcephaly and no neuroimaging and 3 (12%) had no fetal or infant testing. Approximately 41% (82/442) of infants did not have Zika virus testing (JAMA. 2017 Jan 3;317[1]:59-68).
It is unclear why testing was not performed. One concern is that the pediatrician may not have been aware of the maternal Zika virus exposure or test results. It may behoove us to begin asking questions about parental international travel to provide optimal management for our patients. We also should be familiar with the current guidelines for evaluating any potentially exposed infants, which include postnatal neuroimaging, Zika virus testing, a comprehensive newborn examination including neurologic exam, and a standard newborn hearing screen prior to hospital discharge.
Regardless of maternal Zika virus test results, infants with any clinical findings suggestive of congenital Zika virus syndrome and possible maternal exposure based on epidemiologic link also should be tested. Zika virus travel alerts and the most up to date information can be found on the Centers for Disease Control and Prevention website (www. cdc.gov/Zika).
Measles
Although endemic measles was eliminated in the United States in 2000, it is still common in many countries in Europe, Africa, and the Pacific. Most cases in the United States occur in unvaccinated individuals, with 78 cases reported in 2016. As of March 25, 2017, 28 cases have been reported. At least 10 countries – including Belgium, France, Italy, Germany, Portugal, and Thailand – have reported outbreaks of measles since April 2017. As reminder, all children aged 6-11 months should receive one dose of MMR and those 12 months or older should receive two doses of MMR at least 28 days apart if international travel is planned. Adults born after 1956 also should have received two doses of MMR prior to international travel.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She reported having no relevant financial disclosures.
Pes planus: To treat or not to treat
Pes planus, or flat feet, is a common concern addressed during well-child visits. Many parents express concern more because of the appearance of the feet than actual symptomatology. But what is within the realm of normal and what is beyond?
Ninety percent of clinic visits for foot problems are for flat feet.1 Historically, the treatment of flat feet was wearing orthopedic shoes that were unappealing, so parents fear that is the fate of their child. Although the exact incidence rate of flat feet has not been determined, it clearly is quite common. Other contributing factors are joint hypermobility, obesity, and age. All children under the age of 2 years have flat feet because of the fat pad that is present. By the age of 10 years, this fat pad regresses and the normal arch is formed.
Given that the arch does not fully form until the end of the first decade of life, neither diagnosis nor treatment should be given until after that time. Evaluating for flat feet in the clinic is usually limited to inspection in weight bearing and non-weight bearing. There are more formal procedures using foot prints and x-rays, but practically speaking those are not necessary. It is key to examine the patient while he or she is standing, to see if the arch is present and to gauge the orientation of the talus bone. When the patient is supine, again note if the arch is present and check the degree of dorsiflexion and plantarflexion.
Determine whether the talus bone is straight or in a valgus position, and whether there is a shortened Achilles tendon. This is crucial in predicting whether symptoms will emerge if they haven’t already. Patients with rigid flat feet or flexible flat feet with shortened Achilles tendon usually begin to complain of discomfort with activity in the second or third decade of life. Those symptoms may be limited to pain in the foot near the head of the talus, or can be complaints of knee, hip, or back pain.1
For the asymptomatic patient, no intervention is needed. Although many clinicians recommend orthotics, studies have shown that after years of wearing orthotics, the feet remain flat. For symptomatic patients with flexible flat feet, there is increased intrinsic muscle activity, which can result in soreness and achiness of the feet. Orthotics can offer some relief, but for patients with shortened Achilles tendons, it potentially can cause more discomfort.2 Both OTC and hard custom orthotics have been shown to relieve pain without significant increase in the height of the arch. There is little information to support using one over the other.2
Heel cord stretching is another reasonable intervention to improve any discomfort. It is important to note that the knee must be extended and the subtalar joint must be in the neutral position for the stretch to be effective.
Surgery is reserved for flexible and rigid flat feet that are symptomatic despite conservative treatment. Bone reconstruction and tendon lengthening have shown reduction in symptoms.
In summary, the vast majority of patients with flat feet do not need any intervention. Proper categorization is important to determine if intervention will be needed. The use of orthotics should be reserved for symptomatic patients, but will not alter the height of the arch. Surgery is indicated for those patients with significant symptoms that have not improved with conservative measures. It has been found to be effective if all components of the deformity have been addressed.
Dr. Pearce is a pediatrician in Frankfort, Ill. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Iran J Pediatr. 2013 Jun;23(3):247-60.
2. J Child Orthop. 2010;4(2):107-21.
Pes planus, or flat feet, is a common concern addressed during well-child visits. Many parents express concern more because of the appearance of the feet than actual symptomatology. But what is within the realm of normal and what is beyond?
Ninety percent of clinic visits for foot problems are for flat feet.1 Historically, the treatment of flat feet was wearing orthopedic shoes that were unappealing, so parents fear that is the fate of their child. Although the exact incidence rate of flat feet has not been determined, it clearly is quite common. Other contributing factors are joint hypermobility, obesity, and age. All children under the age of 2 years have flat feet because of the fat pad that is present. By the age of 10 years, this fat pad regresses and the normal arch is formed.
Given that the arch does not fully form until the end of the first decade of life, neither diagnosis nor treatment should be given until after that time. Evaluating for flat feet in the clinic is usually limited to inspection in weight bearing and non-weight bearing. There are more formal procedures using foot prints and x-rays, but practically speaking those are not necessary. It is key to examine the patient while he or she is standing, to see if the arch is present and to gauge the orientation of the talus bone. When the patient is supine, again note if the arch is present and check the degree of dorsiflexion and plantarflexion.
Determine whether the talus bone is straight or in a valgus position, and whether there is a shortened Achilles tendon. This is crucial in predicting whether symptoms will emerge if they haven’t already. Patients with rigid flat feet or flexible flat feet with shortened Achilles tendon usually begin to complain of discomfort with activity in the second or third decade of life. Those symptoms may be limited to pain in the foot near the head of the talus, or can be complaints of knee, hip, or back pain.1
For the asymptomatic patient, no intervention is needed. Although many clinicians recommend orthotics, studies have shown that after years of wearing orthotics, the feet remain flat. For symptomatic patients with flexible flat feet, there is increased intrinsic muscle activity, which can result in soreness and achiness of the feet. Orthotics can offer some relief, but for patients with shortened Achilles tendons, it potentially can cause more discomfort.2 Both OTC and hard custom orthotics have been shown to relieve pain without significant increase in the height of the arch. There is little information to support using one over the other.2
Heel cord stretching is another reasonable intervention to improve any discomfort. It is important to note that the knee must be extended and the subtalar joint must be in the neutral position for the stretch to be effective.
Surgery is reserved for flexible and rigid flat feet that are symptomatic despite conservative treatment. Bone reconstruction and tendon lengthening have shown reduction in symptoms.
In summary, the vast majority of patients with flat feet do not need any intervention. Proper categorization is important to determine if intervention will be needed. The use of orthotics should be reserved for symptomatic patients, but will not alter the height of the arch. Surgery is indicated for those patients with significant symptoms that have not improved with conservative measures. It has been found to be effective if all components of the deformity have been addressed.
Dr. Pearce is a pediatrician in Frankfort, Ill. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Iran J Pediatr. 2013 Jun;23(3):247-60.
2. J Child Orthop. 2010;4(2):107-21.
Pes planus, or flat feet, is a common concern addressed during well-child visits. Many parents express concern more because of the appearance of the feet than actual symptomatology. But what is within the realm of normal and what is beyond?
Ninety percent of clinic visits for foot problems are for flat feet.1 Historically, the treatment of flat feet was wearing orthopedic shoes that were unappealing, so parents fear that is the fate of their child. Although the exact incidence rate of flat feet has not been determined, it clearly is quite common. Other contributing factors are joint hypermobility, obesity, and age. All children under the age of 2 years have flat feet because of the fat pad that is present. By the age of 10 years, this fat pad regresses and the normal arch is formed.
Given that the arch does not fully form until the end of the first decade of life, neither diagnosis nor treatment should be given until after that time. Evaluating for flat feet in the clinic is usually limited to inspection in weight bearing and non-weight bearing. There are more formal procedures using foot prints and x-rays, but practically speaking those are not necessary. It is key to examine the patient while he or she is standing, to see if the arch is present and to gauge the orientation of the talus bone. When the patient is supine, again note if the arch is present and check the degree of dorsiflexion and plantarflexion.
Determine whether the talus bone is straight or in a valgus position, and whether there is a shortened Achilles tendon. This is crucial in predicting whether symptoms will emerge if they haven’t already. Patients with rigid flat feet or flexible flat feet with shortened Achilles tendon usually begin to complain of discomfort with activity in the second or third decade of life. Those symptoms may be limited to pain in the foot near the head of the talus, or can be complaints of knee, hip, or back pain.1
For the asymptomatic patient, no intervention is needed. Although many clinicians recommend orthotics, studies have shown that after years of wearing orthotics, the feet remain flat. For symptomatic patients with flexible flat feet, there is increased intrinsic muscle activity, which can result in soreness and achiness of the feet. Orthotics can offer some relief, but for patients with shortened Achilles tendons, it potentially can cause more discomfort.2 Both OTC and hard custom orthotics have been shown to relieve pain without significant increase in the height of the arch. There is little information to support using one over the other.2
Heel cord stretching is another reasonable intervention to improve any discomfort. It is important to note that the knee must be extended and the subtalar joint must be in the neutral position for the stretch to be effective.
Surgery is reserved for flexible and rigid flat feet that are symptomatic despite conservative treatment. Bone reconstruction and tendon lengthening have shown reduction in symptoms.
In summary, the vast majority of patients with flat feet do not need any intervention. Proper categorization is important to determine if intervention will be needed. The use of orthotics should be reserved for symptomatic patients, but will not alter the height of the arch. Surgery is indicated for those patients with significant symptoms that have not improved with conservative measures. It has been found to be effective if all components of the deformity have been addressed.
Dr. Pearce is a pediatrician in Frankfort, Ill. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Iran J Pediatr. 2013 Jun;23(3):247-60.
2. J Child Orthop. 2010;4(2):107-21.
Why state and school policies matter
Recently North Carolina proposed a bill (House Bill 780) that will ban same-sex marriage in the state, even though the U.S. Supreme Court ruled in 2015 that no states can ban same-sex marriage because doing so violates the 14th Amendment. Although many mainly will argue that H.B. 780 is unconstitutional, this bill also can be detrimental to the health of lesbian, gay, and bisexual (LGB) youth.
In March 2017, JAMA Pediatrics published a study on the association between same-sex marriage laws and the rates of suicide attempts.1 Using data from the Youth Risk Behavior Surveillance System, they analyzed the relationship between state policies that permitted same-sex marriage and self-report of suicide attempts within the last 12 months. Thirty percent of LGB youth (13% of the survey population) reported a suicide attempt in the past year prior to any state policies that permitted same-sex marriage, compared with about 9% of the general population. After states implemented pro–same-sex marriage policies, LGB youth suicide attempts dropped to about 26% – a 14% relative decline. This was not limited to LGB youth. The general youth suicide rates declined from 8.6% to 8% – a 7% relative decline. Although the change in suicide attempts was small, the authors determined that the likelihood it occurred by chance was very slim, and concluded that policies enabling same-sex marriage may be associated with an improvement in population health, especially for LGB youth.
This was not the first study examining the relationship between policies and the health of LGB youth. Mark Hatzenbuehler, PhD, of Columbia University, New York, and his associates have published multiple studies on this topic. Four years earlier, Hatzenbuehler et al. published a study on the relationship between antibullying policies and suicide among LGB youth. Using data from the Oregon Healthy Teen survey (2006-2008), he found that LGB youth who live in counties where there are fewer districts with antibullying policies are more than twice as likely to attempt suicide, compared with LGB youth who live in counties where more districts had antibullying policies. Furthermore, the type of antibullying policy mattered. If a district’s antibullying policy did not prohibit bullying based on sexual orientation, then it had no impact on the suicide attempt rates of LGB youth in that area.2 Similar results were found when the relationship between anti–homophobic bullying policies and LGB suicide attempts was examined in the larger Youth Risk Behavior Surveillance System.3
State and local policies also influence other health outcomes among LGB youth. Hatzenbuehler et al. did another study that examined community-level determinants of tobacco use among LGB youth. Again using the data from the Oregon Healthy Teen Survey, they found that LGB youth living in communities that were more supportive of LGB youth (i.e., communities with a high proportion of same-sex couples living in the area, a high proportion of gay-straight alliances at schools, and LGB-specific antibullying policies) were less likely to smoke cigarettes, compared with LGB youth living in communities that were less supportive.4 A similar study in Canada by Konishi et al. found that schools with gay-straight alliances and anti–homophobic bullying policies were less likely to have LGB youth engaging in risky alcohol or illicit drug use.5
Why do these policies matter? A common theme among these policies is that they can either cause or alleviate stress for LGB youth. States that restrict same-sex marriages or schools that do not have any gay-straight alliances may signal to LGB youth that they are not valued or welcomed, or at the very worse, are despised. This creates a hostile and stressful environment for LGB youth, which raises the risk for mental health problems, such as depression and anxiety, which in turn, raises the risk for substance use and suicide.6 Conversely, the presence of a gay-straight alliance at school or a state that allows same-sex marriage may indicate to LGB youth that they are welcomed, if not just tolerated, and may alleviate this risk. Furthermore, antibullying policies seem to reduce the stress associated with bullying among LGB youth because it may serve as a deterrent to bullying based on sexual orientation. Although passing pro-LGB policies will not solve all the health problems among LGB youth, these policies certainly have an impact.
There might be trepidation among some pediatricians about being vocal on a politically charged policy proposal such as H.B. 780. However, suicide and substance use are major concerns for all physicians – especially pediatricians. Policy makers considering passing laws that can affect their LGB constituents should read these studies to see what kind of influence these proposals can have on the health and well-being of LGB youth. Moreover, health care providers should use their expertise, influence, and standing in their community to support policies that encourage protection for LGB youth and oppose policies that can harm LGB youth. These leaders are responsible for ensuring the health of the people they serve.
Dr. Montano is clinical instructor of pediatrics at the University of Pittsburgh and an adolescent medicine physician at Children’s Hospital of Pittsburgh of UPMC.
References
1. JAMA Pediatr. 2017. doi: 10.1001/jamapediatrics.2016.4529.
2. J Adolesc Health. 2013:S21-6. doi: 10.1016/j.jadohealth.2012.08.010.
3. Am J Public Health. 2014. doi: 10.2105/AJPH.2013.301508.
4. Arch Pediatr Adolesc Med. 2011. doi: 10.1001/archpediatrics.2011.64.
5. Prev Med. 2013. doi: 10.1016/j.ypmed.2013.06.031.
6. Psychol Bull. 2003. doi: 10.1037/0033-2909.129.5.674.
Recently North Carolina proposed a bill (House Bill 780) that will ban same-sex marriage in the state, even though the U.S. Supreme Court ruled in 2015 that no states can ban same-sex marriage because doing so violates the 14th Amendment. Although many mainly will argue that H.B. 780 is unconstitutional, this bill also can be detrimental to the health of lesbian, gay, and bisexual (LGB) youth.
In March 2017, JAMA Pediatrics published a study on the association between same-sex marriage laws and the rates of suicide attempts.1 Using data from the Youth Risk Behavior Surveillance System, they analyzed the relationship between state policies that permitted same-sex marriage and self-report of suicide attempts within the last 12 months. Thirty percent of LGB youth (13% of the survey population) reported a suicide attempt in the past year prior to any state policies that permitted same-sex marriage, compared with about 9% of the general population. After states implemented pro–same-sex marriage policies, LGB youth suicide attempts dropped to about 26% – a 14% relative decline. This was not limited to LGB youth. The general youth suicide rates declined from 8.6% to 8% – a 7% relative decline. Although the change in suicide attempts was small, the authors determined that the likelihood it occurred by chance was very slim, and concluded that policies enabling same-sex marriage may be associated with an improvement in population health, especially for LGB youth.
This was not the first study examining the relationship between policies and the health of LGB youth. Mark Hatzenbuehler, PhD, of Columbia University, New York, and his associates have published multiple studies on this topic. Four years earlier, Hatzenbuehler et al. published a study on the relationship between antibullying policies and suicide among LGB youth. Using data from the Oregon Healthy Teen survey (2006-2008), he found that LGB youth who live in counties where there are fewer districts with antibullying policies are more than twice as likely to attempt suicide, compared with LGB youth who live in counties where more districts had antibullying policies. Furthermore, the type of antibullying policy mattered. If a district’s antibullying policy did not prohibit bullying based on sexual orientation, then it had no impact on the suicide attempt rates of LGB youth in that area.2 Similar results were found when the relationship between anti–homophobic bullying policies and LGB suicide attempts was examined in the larger Youth Risk Behavior Surveillance System.3
State and local policies also influence other health outcomes among LGB youth. Hatzenbuehler et al. did another study that examined community-level determinants of tobacco use among LGB youth. Again using the data from the Oregon Healthy Teen Survey, they found that LGB youth living in communities that were more supportive of LGB youth (i.e., communities with a high proportion of same-sex couples living in the area, a high proportion of gay-straight alliances at schools, and LGB-specific antibullying policies) were less likely to smoke cigarettes, compared with LGB youth living in communities that were less supportive.4 A similar study in Canada by Konishi et al. found that schools with gay-straight alliances and anti–homophobic bullying policies were less likely to have LGB youth engaging in risky alcohol or illicit drug use.5
Why do these policies matter? A common theme among these policies is that they can either cause or alleviate stress for LGB youth. States that restrict same-sex marriages or schools that do not have any gay-straight alliances may signal to LGB youth that they are not valued or welcomed, or at the very worse, are despised. This creates a hostile and stressful environment for LGB youth, which raises the risk for mental health problems, such as depression and anxiety, which in turn, raises the risk for substance use and suicide.6 Conversely, the presence of a gay-straight alliance at school or a state that allows same-sex marriage may indicate to LGB youth that they are welcomed, if not just tolerated, and may alleviate this risk. Furthermore, antibullying policies seem to reduce the stress associated with bullying among LGB youth because it may serve as a deterrent to bullying based on sexual orientation. Although passing pro-LGB policies will not solve all the health problems among LGB youth, these policies certainly have an impact.
There might be trepidation among some pediatricians about being vocal on a politically charged policy proposal such as H.B. 780. However, suicide and substance use are major concerns for all physicians – especially pediatricians. Policy makers considering passing laws that can affect their LGB constituents should read these studies to see what kind of influence these proposals can have on the health and well-being of LGB youth. Moreover, health care providers should use their expertise, influence, and standing in their community to support policies that encourage protection for LGB youth and oppose policies that can harm LGB youth. These leaders are responsible for ensuring the health of the people they serve.
Dr. Montano is clinical instructor of pediatrics at the University of Pittsburgh and an adolescent medicine physician at Children’s Hospital of Pittsburgh of UPMC.
References
1. JAMA Pediatr. 2017. doi: 10.1001/jamapediatrics.2016.4529.
2. J Adolesc Health. 2013:S21-6. doi: 10.1016/j.jadohealth.2012.08.010.
3. Am J Public Health. 2014. doi: 10.2105/AJPH.2013.301508.
4. Arch Pediatr Adolesc Med. 2011. doi: 10.1001/archpediatrics.2011.64.
5. Prev Med. 2013. doi: 10.1016/j.ypmed.2013.06.031.
6. Psychol Bull. 2003. doi: 10.1037/0033-2909.129.5.674.
Recently North Carolina proposed a bill (House Bill 780) that will ban same-sex marriage in the state, even though the U.S. Supreme Court ruled in 2015 that no states can ban same-sex marriage because doing so violates the 14th Amendment. Although many mainly will argue that H.B. 780 is unconstitutional, this bill also can be detrimental to the health of lesbian, gay, and bisexual (LGB) youth.
In March 2017, JAMA Pediatrics published a study on the association between same-sex marriage laws and the rates of suicide attempts.1 Using data from the Youth Risk Behavior Surveillance System, they analyzed the relationship between state policies that permitted same-sex marriage and self-report of suicide attempts within the last 12 months. Thirty percent of LGB youth (13% of the survey population) reported a suicide attempt in the past year prior to any state policies that permitted same-sex marriage, compared with about 9% of the general population. After states implemented pro–same-sex marriage policies, LGB youth suicide attempts dropped to about 26% – a 14% relative decline. This was not limited to LGB youth. The general youth suicide rates declined from 8.6% to 8% – a 7% relative decline. Although the change in suicide attempts was small, the authors determined that the likelihood it occurred by chance was very slim, and concluded that policies enabling same-sex marriage may be associated with an improvement in population health, especially for LGB youth.
This was not the first study examining the relationship between policies and the health of LGB youth. Mark Hatzenbuehler, PhD, of Columbia University, New York, and his associates have published multiple studies on this topic. Four years earlier, Hatzenbuehler et al. published a study on the relationship between antibullying policies and suicide among LGB youth. Using data from the Oregon Healthy Teen survey (2006-2008), he found that LGB youth who live in counties where there are fewer districts with antibullying policies are more than twice as likely to attempt suicide, compared with LGB youth who live in counties where more districts had antibullying policies. Furthermore, the type of antibullying policy mattered. If a district’s antibullying policy did not prohibit bullying based on sexual orientation, then it had no impact on the suicide attempt rates of LGB youth in that area.2 Similar results were found when the relationship between anti–homophobic bullying policies and LGB suicide attempts was examined in the larger Youth Risk Behavior Surveillance System.3
State and local policies also influence other health outcomes among LGB youth. Hatzenbuehler et al. did another study that examined community-level determinants of tobacco use among LGB youth. Again using the data from the Oregon Healthy Teen Survey, they found that LGB youth living in communities that were more supportive of LGB youth (i.e., communities with a high proportion of same-sex couples living in the area, a high proportion of gay-straight alliances at schools, and LGB-specific antibullying policies) were less likely to smoke cigarettes, compared with LGB youth living in communities that were less supportive.4 A similar study in Canada by Konishi et al. found that schools with gay-straight alliances and anti–homophobic bullying policies were less likely to have LGB youth engaging in risky alcohol or illicit drug use.5
Why do these policies matter? A common theme among these policies is that they can either cause or alleviate stress for LGB youth. States that restrict same-sex marriages or schools that do not have any gay-straight alliances may signal to LGB youth that they are not valued or welcomed, or at the very worse, are despised. This creates a hostile and stressful environment for LGB youth, which raises the risk for mental health problems, such as depression and anxiety, which in turn, raises the risk for substance use and suicide.6 Conversely, the presence of a gay-straight alliance at school or a state that allows same-sex marriage may indicate to LGB youth that they are welcomed, if not just tolerated, and may alleviate this risk. Furthermore, antibullying policies seem to reduce the stress associated with bullying among LGB youth because it may serve as a deterrent to bullying based on sexual orientation. Although passing pro-LGB policies will not solve all the health problems among LGB youth, these policies certainly have an impact.
There might be trepidation among some pediatricians about being vocal on a politically charged policy proposal such as H.B. 780. However, suicide and substance use are major concerns for all physicians – especially pediatricians. Policy makers considering passing laws that can affect their LGB constituents should read these studies to see what kind of influence these proposals can have on the health and well-being of LGB youth. Moreover, health care providers should use their expertise, influence, and standing in their community to support policies that encourage protection for LGB youth and oppose policies that can harm LGB youth. These leaders are responsible for ensuring the health of the people they serve.
Dr. Montano is clinical instructor of pediatrics at the University of Pittsburgh and an adolescent medicine physician at Children’s Hospital of Pittsburgh of UPMC.
References
1. JAMA Pediatr. 2017. doi: 10.1001/jamapediatrics.2016.4529.
2. J Adolesc Health. 2013:S21-6. doi: 10.1016/j.jadohealth.2012.08.010.
3. Am J Public Health. 2014. doi: 10.2105/AJPH.2013.301508.
4. Arch Pediatr Adolesc Med. 2011. doi: 10.1001/archpediatrics.2011.64.
5. Prev Med. 2013. doi: 10.1016/j.ypmed.2013.06.031.
6. Psychol Bull. 2003. doi: 10.1037/0033-2909.129.5.674.