User login
Point/Counterpoint: Should all suspected mucinous cystic neoplasms be resected?
Dr. Pawlik is a pretty clever guy and a strong adversary. Mucinous cystic neoplasm is literally the only topic in hepato-pancreato-biliary surgery he has not written about – yet.1
But I will argue that all suspected mucinous cystic neoplasms (MCNs) should be surgically removed. Reasons include: the natural history of a benign MCN in a typical 45-year-old remains unknown, clinical features are not always reliable, and it’s difficult to distinguish benign from malignant neoplasms without surgery.
Although many MCNs are benign when resected, all have malignant potential.
The prevalence of cancer at time of diagnosis with an MCN is fairly low, about 15%. This means most of these cysts are benign when resected. However, discrimination between benign and malignant is difficult without surgery, and the degree of epithelial dysplasia at time of resection ranges from mild to severe with invasive carcinoma.
When cancer is present, patients tend to do poorly. In one study where 44 out of 344 patients developed invasive cancer, the group with cancer had a 3-year overall survival rate of 59%.2
In addition, aspiration of cyst fluid is often of limited utility. It is poor at distinguishing whether a cyst is benign or malignant. For example, in a series of 55 patients with MCNs that underwent fine-needle aspiration of cyst epithelium, 71% of assays were nondiagnostic.3 So this diagnostic test is very insensitive and may miss at least half of cancers – another point in favor of surgical resection for all MCNs.
It is true that evidence in the literature associates certain clinical factors with a higher risk for malignancy in suspected MCNs. These include male gender, larger cysts, and location in the pancreatic head or neck or larger cyst diameter and presence of nodules.2,4 However, use of clinical features is not perfect.
In another study, of 163 resected MCNs, those with invasive cancer were often – but not always – larger than 4 cm with nodules.5 These same series revealed that those with invasive cancer were often, but again not always, larger than 4 cm.
We see this in our practice as well – that a large cyst or presence of nodules is not a foolproof indicator of malignancy. We had a 38-year-old woman with a large cyst and nodules who did not have cancer. In contrast, a 45-year-old woman at our institution presented with small cysts and no cancer on fine-needle aspiration. However, final pathology in her case revealed high-grade dysplasia.
Also, we only know what happens to cysts that have been resected. We know nothing about duration or other best practices for following patients who do not undergo surgery. For this reason, we really don’t know what these tumors will do if left alone.
Surgery is curative and carries a pretty low risk. Following resection, these patients do well. We’re talking about curative operations.
Dr. Katz is an Associate Professor, Department of Surgical Oncology, Division of Surgery at the University of Texas MD Anderson Cancer Center in Houston. He is also Chief of the Pancreas Surgery Service at MD Anderson. Dr. Katz noted he was asked to provide the pro side of the argument, and he may not necessarily uphold these positions in his own practice. Dr. Katz had no relevant financial disclosures.
References:
1. Dr. Pawlik’s list of selected publications: http://pathology.jhu.edu/liver/pawlik.cfm.
2. JAMA Surg. 2017;152:19-25.
3. Cancer. 2017;125:169-77.
4. Pancreas. 2011;40:67-71.
5. Ann Surg. 2008;247:571-9.
Some mucinous cystic neoplasms can be safely followed.
Data in the literature suggest some of these suspected mucinous cystic neoplasms can be followed; surgery may not be indicated solely because “as surgeons we tend to take all masses out” and because operative complications occur. Therefore, resection is not a benign procedure.
Dr. Katz, you were done before you got started. The evidence shows that some of the MCNs can be followed rather than resected.
Most of the published studies focus on IPMN, but I think the topic of MCNs is becoming increasingly important; they follow IPMNs as the second most common type of cystic neoplasms. So we’re going to be finding more small MCNs. In this debate, we are not talking about an 8-cm MCN, but rather what do we do when we see a 2- to 3-cm MCN. Do these patients all need to undergo resection?
What Dr. Katz is concerned about is that we are going to miss a cancer. We should operate on all patients because – at least as Dr. Katz’s argues – the surgery can always be done without complications.
However, even at the very, very experienced centers, morbidity was 30%-50%, and a pancreatic fistula developed for 1 in 10 patients. So it’s not a benign procedure.1
As surgeons, we tend to take every mass out. Although operative mortality is low, it is still in a measurable range, 1%-2%, and that is even at expert centers. Many of these small cysts are being found at smaller and community hospitals, and undue morbidity and mortality may weigh more heavily in these settings.2
Rather than “being a hammer and seeing everything as a nail,” we need a more rational approach. For example, we should identify a subgroup that will the most benefit from resection.
Investigators from Memorial Sloan Kettering Cancer Center reported that certain clinical factors identify patients at higher risk for mortality, such as nodules.3 Patients with invasive cancer almost all had nodules. And all patients with invasive cancer without nodules had a tumor larger than 4 cm.
Maybe using both presence of nodules and size is the right approach to identifying underling malignancy in suspected MCNs. Again, size is important, as is male gender, presence of solid nodules, and duct dilatation.4
We are arguing whether all MCNs should be resected. I’m positing that many 2-cm to 3-cm MCNs can be followed. There is only a small chance they will grow over time, and it’s unlikely they are harboring a malignancy.
Dr. Pawlik is Chair of Surgery at the Ohio State Wexner Medical Center in Columbus. He is also the Urban Meyer III and Shelley Meyer Chair for Cancer Research at Ohio State. He has no disclosures.
References
1. HPB (Oxford). 2007;9:8-15.
2. Diagnosis and Management of Cystic Lesions of the Pancreas. Diagnostic and Therapeutic Endoscopy Volume 2011 (2011), Article ID 478913.
3. JAMA Surg. 2017;152:19-25.
4. Ann Surg. 2006;244:572-82.
This Point/Counterpoint feature is based on comments Dr. Katz and Dr. Pawlik made during a debate at AHPBA 2017, the annual meeting of the Americas Hepato-Pancreato-Biliary Association.
Dr. Pawlik is a pretty clever guy and a strong adversary. Mucinous cystic neoplasm is literally the only topic in hepato-pancreato-biliary surgery he has not written about – yet.1
But I will argue that all suspected mucinous cystic neoplasms (MCNs) should be surgically removed. Reasons include: the natural history of a benign MCN in a typical 45-year-old remains unknown, clinical features are not always reliable, and it’s difficult to distinguish benign from malignant neoplasms without surgery.
Although many MCNs are benign when resected, all have malignant potential.
The prevalence of cancer at time of diagnosis with an MCN is fairly low, about 15%. This means most of these cysts are benign when resected. However, discrimination between benign and malignant is difficult without surgery, and the degree of epithelial dysplasia at time of resection ranges from mild to severe with invasive carcinoma.
When cancer is present, patients tend to do poorly. In one study where 44 out of 344 patients developed invasive cancer, the group with cancer had a 3-year overall survival rate of 59%.2
In addition, aspiration of cyst fluid is often of limited utility. It is poor at distinguishing whether a cyst is benign or malignant. For example, in a series of 55 patients with MCNs that underwent fine-needle aspiration of cyst epithelium, 71% of assays were nondiagnostic.3 So this diagnostic test is very insensitive and may miss at least half of cancers – another point in favor of surgical resection for all MCNs.
It is true that evidence in the literature associates certain clinical factors with a higher risk for malignancy in suspected MCNs. These include male gender, larger cysts, and location in the pancreatic head or neck or larger cyst diameter and presence of nodules.2,4 However, use of clinical features is not perfect.
In another study, of 163 resected MCNs, those with invasive cancer were often – but not always – larger than 4 cm with nodules.5 These same series revealed that those with invasive cancer were often, but again not always, larger than 4 cm.
We see this in our practice as well – that a large cyst or presence of nodules is not a foolproof indicator of malignancy. We had a 38-year-old woman with a large cyst and nodules who did not have cancer. In contrast, a 45-year-old woman at our institution presented with small cysts and no cancer on fine-needle aspiration. However, final pathology in her case revealed high-grade dysplasia.
Also, we only know what happens to cysts that have been resected. We know nothing about duration or other best practices for following patients who do not undergo surgery. For this reason, we really don’t know what these tumors will do if left alone.
Surgery is curative and carries a pretty low risk. Following resection, these patients do well. We’re talking about curative operations.
Dr. Katz is an Associate Professor, Department of Surgical Oncology, Division of Surgery at the University of Texas MD Anderson Cancer Center in Houston. He is also Chief of the Pancreas Surgery Service at MD Anderson. Dr. Katz noted he was asked to provide the pro side of the argument, and he may not necessarily uphold these positions in his own practice. Dr. Katz had no relevant financial disclosures.
References:
1. Dr. Pawlik’s list of selected publications: http://pathology.jhu.edu/liver/pawlik.cfm.
2. JAMA Surg. 2017;152:19-25.
3. Cancer. 2017;125:169-77.
4. Pancreas. 2011;40:67-71.
5. Ann Surg. 2008;247:571-9.
Some mucinous cystic neoplasms can be safely followed.
Data in the literature suggest some of these suspected mucinous cystic neoplasms can be followed; surgery may not be indicated solely because “as surgeons we tend to take all masses out” and because operative complications occur. Therefore, resection is not a benign procedure.
Dr. Katz, you were done before you got started. The evidence shows that some of the MCNs can be followed rather than resected.
Most of the published studies focus on IPMN, but I think the topic of MCNs is becoming increasingly important; they follow IPMNs as the second most common type of cystic neoplasms. So we’re going to be finding more small MCNs. In this debate, we are not talking about an 8-cm MCN, but rather what do we do when we see a 2- to 3-cm MCN. Do these patients all need to undergo resection?
What Dr. Katz is concerned about is that we are going to miss a cancer. We should operate on all patients because – at least as Dr. Katz’s argues – the surgery can always be done without complications.
However, even at the very, very experienced centers, morbidity was 30%-50%, and a pancreatic fistula developed for 1 in 10 patients. So it’s not a benign procedure.1
As surgeons, we tend to take every mass out. Although operative mortality is low, it is still in a measurable range, 1%-2%, and that is even at expert centers. Many of these small cysts are being found at smaller and community hospitals, and undue morbidity and mortality may weigh more heavily in these settings.2
Rather than “being a hammer and seeing everything as a nail,” we need a more rational approach. For example, we should identify a subgroup that will the most benefit from resection.
Investigators from Memorial Sloan Kettering Cancer Center reported that certain clinical factors identify patients at higher risk for mortality, such as nodules.3 Patients with invasive cancer almost all had nodules. And all patients with invasive cancer without nodules had a tumor larger than 4 cm.
Maybe using both presence of nodules and size is the right approach to identifying underling malignancy in suspected MCNs. Again, size is important, as is male gender, presence of solid nodules, and duct dilatation.4
We are arguing whether all MCNs should be resected. I’m positing that many 2-cm to 3-cm MCNs can be followed. There is only a small chance they will grow over time, and it’s unlikely they are harboring a malignancy.
Dr. Pawlik is Chair of Surgery at the Ohio State Wexner Medical Center in Columbus. He is also the Urban Meyer III and Shelley Meyer Chair for Cancer Research at Ohio State. He has no disclosures.
References
1. HPB (Oxford). 2007;9:8-15.
2. Diagnosis and Management of Cystic Lesions of the Pancreas. Diagnostic and Therapeutic Endoscopy Volume 2011 (2011), Article ID 478913.
3. JAMA Surg. 2017;152:19-25.
4. Ann Surg. 2006;244:572-82.
This Point/Counterpoint feature is based on comments Dr. Katz and Dr. Pawlik made during a debate at AHPBA 2017, the annual meeting of the Americas Hepato-Pancreato-Biliary Association.
Dr. Pawlik is a pretty clever guy and a strong adversary. Mucinous cystic neoplasm is literally the only topic in hepato-pancreato-biliary surgery he has not written about – yet.1
But I will argue that all suspected mucinous cystic neoplasms (MCNs) should be surgically removed. Reasons include: the natural history of a benign MCN in a typical 45-year-old remains unknown, clinical features are not always reliable, and it’s difficult to distinguish benign from malignant neoplasms without surgery.
Although many MCNs are benign when resected, all have malignant potential.
The prevalence of cancer at time of diagnosis with an MCN is fairly low, about 15%. This means most of these cysts are benign when resected. However, discrimination between benign and malignant is difficult without surgery, and the degree of epithelial dysplasia at time of resection ranges from mild to severe with invasive carcinoma.
When cancer is present, patients tend to do poorly. In one study where 44 out of 344 patients developed invasive cancer, the group with cancer had a 3-year overall survival rate of 59%.2
In addition, aspiration of cyst fluid is often of limited utility. It is poor at distinguishing whether a cyst is benign or malignant. For example, in a series of 55 patients with MCNs that underwent fine-needle aspiration of cyst epithelium, 71% of assays were nondiagnostic.3 So this diagnostic test is very insensitive and may miss at least half of cancers – another point in favor of surgical resection for all MCNs.
It is true that evidence in the literature associates certain clinical factors with a higher risk for malignancy in suspected MCNs. These include male gender, larger cysts, and location in the pancreatic head or neck or larger cyst diameter and presence of nodules.2,4 However, use of clinical features is not perfect.
In another study, of 163 resected MCNs, those with invasive cancer were often – but not always – larger than 4 cm with nodules.5 These same series revealed that those with invasive cancer were often, but again not always, larger than 4 cm.
We see this in our practice as well – that a large cyst or presence of nodules is not a foolproof indicator of malignancy. We had a 38-year-old woman with a large cyst and nodules who did not have cancer. In contrast, a 45-year-old woman at our institution presented with small cysts and no cancer on fine-needle aspiration. However, final pathology in her case revealed high-grade dysplasia.
Also, we only know what happens to cysts that have been resected. We know nothing about duration or other best practices for following patients who do not undergo surgery. For this reason, we really don’t know what these tumors will do if left alone.
Surgery is curative and carries a pretty low risk. Following resection, these patients do well. We’re talking about curative operations.
Dr. Katz is an Associate Professor, Department of Surgical Oncology, Division of Surgery at the University of Texas MD Anderson Cancer Center in Houston. He is also Chief of the Pancreas Surgery Service at MD Anderson. Dr. Katz noted he was asked to provide the pro side of the argument, and he may not necessarily uphold these positions in his own practice. Dr. Katz had no relevant financial disclosures.
References:
1. Dr. Pawlik’s list of selected publications: http://pathology.jhu.edu/liver/pawlik.cfm.
2. JAMA Surg. 2017;152:19-25.
3. Cancer. 2017;125:169-77.
4. Pancreas. 2011;40:67-71.
5. Ann Surg. 2008;247:571-9.
Some mucinous cystic neoplasms can be safely followed.
Data in the literature suggest some of these suspected mucinous cystic neoplasms can be followed; surgery may not be indicated solely because “as surgeons we tend to take all masses out” and because operative complications occur. Therefore, resection is not a benign procedure.
Dr. Katz, you were done before you got started. The evidence shows that some of the MCNs can be followed rather than resected.
Most of the published studies focus on IPMN, but I think the topic of MCNs is becoming increasingly important; they follow IPMNs as the second most common type of cystic neoplasms. So we’re going to be finding more small MCNs. In this debate, we are not talking about an 8-cm MCN, but rather what do we do when we see a 2- to 3-cm MCN. Do these patients all need to undergo resection?
What Dr. Katz is concerned about is that we are going to miss a cancer. We should operate on all patients because – at least as Dr. Katz’s argues – the surgery can always be done without complications.
However, even at the very, very experienced centers, morbidity was 30%-50%, and a pancreatic fistula developed for 1 in 10 patients. So it’s not a benign procedure.1
As surgeons, we tend to take every mass out. Although operative mortality is low, it is still in a measurable range, 1%-2%, and that is even at expert centers. Many of these small cysts are being found at smaller and community hospitals, and undue morbidity and mortality may weigh more heavily in these settings.2
Rather than “being a hammer and seeing everything as a nail,” we need a more rational approach. For example, we should identify a subgroup that will the most benefit from resection.
Investigators from Memorial Sloan Kettering Cancer Center reported that certain clinical factors identify patients at higher risk for mortality, such as nodules.3 Patients with invasive cancer almost all had nodules. And all patients with invasive cancer without nodules had a tumor larger than 4 cm.
Maybe using both presence of nodules and size is the right approach to identifying underling malignancy in suspected MCNs. Again, size is important, as is male gender, presence of solid nodules, and duct dilatation.4
We are arguing whether all MCNs should be resected. I’m positing that many 2-cm to 3-cm MCNs can be followed. There is only a small chance they will grow over time, and it’s unlikely they are harboring a malignancy.
Dr. Pawlik is Chair of Surgery at the Ohio State Wexner Medical Center in Columbus. He is also the Urban Meyer III and Shelley Meyer Chair for Cancer Research at Ohio State. He has no disclosures.
References
1. HPB (Oxford). 2007;9:8-15.
2. Diagnosis and Management of Cystic Lesions of the Pancreas. Diagnostic and Therapeutic Endoscopy Volume 2011 (2011), Article ID 478913.
3. JAMA Surg. 2017;152:19-25.
4. Ann Surg. 2006;244:572-82.
This Point/Counterpoint feature is based on comments Dr. Katz and Dr. Pawlik made during a debate at AHPBA 2017, the annual meeting of the Americas Hepato-Pancreato-Biliary Association.
Editorial Note
The first Skin of Color column, "Diversity in Dermatology: A Society Devoted to Skin of Color," produced in collaboration with the Skin of Color Society appears on page 322. This column will be published quarterly and will serve to increase the knowledge available to dermatologists to help improve delivery of care to this underserved population.
Look for Skin of Color columns in upcoming issues of Cutis.
The first Skin of Color column, "Diversity in Dermatology: A Society Devoted to Skin of Color," produced in collaboration with the Skin of Color Society appears on page 322. This column will be published quarterly and will serve to increase the knowledge available to dermatologists to help improve delivery of care to this underserved population.
Look for Skin of Color columns in upcoming issues of Cutis.
The first Skin of Color column, "Diversity in Dermatology: A Society Devoted to Skin of Color," produced in collaboration with the Skin of Color Society appears on page 322. This column will be published quarterly and will serve to increase the knowledge available to dermatologists to help improve delivery of care to this underserved population.
Look for Skin of Color columns in upcoming issues of Cutis.
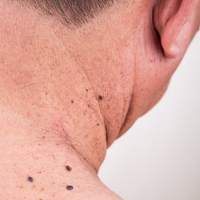
Skin Cancer Mortality in Patients With Skin of Color
Skin cancers in patients with skin of color are less prevalent but have a higher morbidity and mortality compared to white patients. Challenges to early detection, including clinical differences in presentation, low public awareness, lower index of suspicion among health care providers, and access to specialty care, likely contribute to observed differences in prognosis between skin of color and white populations.
Skin cancer is the most common malignancy in the United States, accounting for approximately 40% of all neoplasms in white patients but only 1% to 4% in Asian American and black patients.1,2 Largely due to the photoprotective effects of increased constitutive epidermal melanin, melanoma is approximately 10 to 20 times less frequent in black patients and 3 to 7 times less common in Hispanics than age-matched whites.1 Nonmelanoma skin cancers including squamous cell carcinoma (SCC) and basal cell carcinoma also are less prevalent in darker skin types.3,4
In the United States, Hispanic, American Indian
Similar to melanoma, the mortality from SCC is disproportionately increased in skin of color populations, ranging from 18% to 29% in black patients.3,10,11 There is a paucity of population-based studies in the United States looking at mortality rates of nonmelanoma skin cancers and their trends over time, but a 1993 study suggests that mortality rates are declining less consistently in black patients than white patients.11
Factors that may contribute to higher mortality rates in patients with skin of color include a greater propensity for inherently aggressive skin cancers (eg, higher risk of SCC) and delays in diagnosis (eg, late-stage diagnosis of melanoma).1,4 For melanoma, increased mortality has been attributed to a predominance of acral lentiginous melanomas, which are more frequently diagnosed at more advanced stages than other melanoma subtypes.6,12,13 Black patients, Hispanics, Asians, and Pacific Islanders are all more likely to present with thicker tumors and metastases on initial presentation than their white counterparts (P<.001).2,8,9,12-14 The higher risk of death from SCC results from the predominance of lesions on non–sun-exposed areas, particularly the legs and anogenital areas, and within sites of chronic scarring or inflammation.4 Unlike sun-induced SCC, the most commonly observed type of SCC in lighter skin types, SCCs that develop in association with chronic inflammatory or ulcerative processes are aggressive and invasive, and they metastasize to distant sites in 20% to 40% of cases (versus 1%–4% in sun-induced SCC).1,3,4 For all skin cancers, poor access to medical care, patients’ unawareness of their skin cancer risk, lack of adequate skin examinations, and prevalence of lesions on uncommon sites that may be inconspicuous or overlooked have all been suggested to delay diagnosis.1,15,16 Given that more advanced disease is associated with worse outcomes, the implications of this delay are enormous and remain a cause for concern.
The alarming skin cancer mortality rates in patients with skin of color are a call to action for the medical community. The consistent use of full-body skin examinations including close inspection of mucosal, acral, and genital areas for all patients independent of skin type and racial/ethnic background is paramount. Advancing skin cancer education in skin of color populations, such as through distribution of patient-directed educational materials produced by organizations such as the American Academy of Dermatology, Skin Cancer Foundation, and Skin of Color Society, is an important step toward increased public awareness.16 Use of social and traditional media outlets as well as community-directed health outreach campaigns also are important strategies to change the common misconception that darker-skinned individuals do not get skin cancer. We hope that with a multipronged approach, disparities in skin cancer mortality will steadily be eliminated.
- Gloster HM Jr, Neal K. Skin cancer in skin of color. J Am Acad Dermatol. 2006;55:741-760; quiz 761-764.
- Cormier JN, Xing Y, Ding M, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med. 2006;166:1907-1914.
- Mora RG, Perniciaro C. Cancer of the skin in blacks: I. a review of 163 black patients with cutaneous squamous cell carcinoma. J Am Acad Dermatol. 1981;5:535-543.
- Halder RM, Bridgeman-Shah S. Skin cancer in African Americans. Cancer. 1995;75:667-673.
- Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2013. Bethesda, MD: National Cancer Institute; April 2016. http://seer.cancer.gov/csr/1975_2013/. Updated September 12, 2016. Accessed April 7, 2017.
- Bellows CF, Belafsky P, Fortgang IS, et al. Melanoma in African-Americans: trends in biological behavior and clinical characteristics over two decades. J Surg Oncol. 2001;78:10-16.
- Chen L, Jin S. Trends in mortality rates of cutaneous melanoma in East Asian populations. Peer J. 2014;4:e2809.
- Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California Cancer Registry data. Cancer Causes Control. 1997;8:246-252.
- Johnson DS, Yamane S, Morita S, et al. Malignant melanoma in non-Caucasians: experience from Hawaii. Surg Clin N Am. 2003;83:275-282.
- Fleming ID, Barnawell JR, Burlison PE, et al. Skin cancer in black patients. Cancer. 1975;35:600-605.
- Weinstock MA. Nonmelanoma skin cancer mortality in the United States, 1969 through 1988. Arch Dermatol. 1993;129:1286-1290.
- Byrd KM, Wilson DC, Hoyler SS. Advanced presentation of melanoma in African Americans. J Am Acad Dermatol. 2004;50:142-143.
- Hu S, Parmet Y, Allen G, et al. Disparity in melanoma: a trend analysis of melanoma incidence and stage at diagnosis among whites, Hispanics, and blacks in Florida. Arch Dermatol. 2009;145:1369-1374.
- Black WC, Goldhahn RT, Wiggins C. Melanoma within a southwestern Hispanic population. Arch Dermatol. 1987;123:1331-1334.
- Harvey VM, Oldfield CW, Chen JT, et al. Melanoma disparities among US Hispanics: use of the social ecological model to contextualize reasons for inequitable outcomes and frame a research agenda [published online August 29, 2016]. J Skin Cancer. 2016;2016:4635740.
- Robinson JK, Joshi KM, Ortiz S, et al. Melanoma knowledge, perception, and awareness in ethnic minorities in Chicago: recommendations regarding education. Psychooncology. 2011;20:313-320.
Skin cancers in patients with skin of color are less prevalent but have a higher morbidity and mortality compared to white patients. Challenges to early detection, including clinical differences in presentation, low public awareness, lower index of suspicion among health care providers, and access to specialty care, likely contribute to observed differences in prognosis between skin of color and white populations.
Skin cancer is the most common malignancy in the United States, accounting for approximately 40% of all neoplasms in white patients but only 1% to 4% in Asian American and black patients.1,2 Largely due to the photoprotective effects of increased constitutive epidermal melanin, melanoma is approximately 10 to 20 times less frequent in black patients and 3 to 7 times less common in Hispanics than age-matched whites.1 Nonmelanoma skin cancers including squamous cell carcinoma (SCC) and basal cell carcinoma also are less prevalent in darker skin types.3,4
In the United States, Hispanic, American Indian
Similar to melanoma, the mortality from SCC is disproportionately increased in skin of color populations, ranging from 18% to 29% in black patients.3,10,11 There is a paucity of population-based studies in the United States looking at mortality rates of nonmelanoma skin cancers and their trends over time, but a 1993 study suggests that mortality rates are declining less consistently in black patients than white patients.11
Factors that may contribute to higher mortality rates in patients with skin of color include a greater propensity for inherently aggressive skin cancers (eg, higher risk of SCC) and delays in diagnosis (eg, late-stage diagnosis of melanoma).1,4 For melanoma, increased mortality has been attributed to a predominance of acral lentiginous melanomas, which are more frequently diagnosed at more advanced stages than other melanoma subtypes.6,12,13 Black patients, Hispanics, Asians, and Pacific Islanders are all more likely to present with thicker tumors and metastases on initial presentation than their white counterparts (P<.001).2,8,9,12-14 The higher risk of death from SCC results from the predominance of lesions on non–sun-exposed areas, particularly the legs and anogenital areas, and within sites of chronic scarring or inflammation.4 Unlike sun-induced SCC, the most commonly observed type of SCC in lighter skin types, SCCs that develop in association with chronic inflammatory or ulcerative processes are aggressive and invasive, and they metastasize to distant sites in 20% to 40% of cases (versus 1%–4% in sun-induced SCC).1,3,4 For all skin cancers, poor access to medical care, patients’ unawareness of their skin cancer risk, lack of adequate skin examinations, and prevalence of lesions on uncommon sites that may be inconspicuous or overlooked have all been suggested to delay diagnosis.1,15,16 Given that more advanced disease is associated with worse outcomes, the implications of this delay are enormous and remain a cause for concern.
The alarming skin cancer mortality rates in patients with skin of color are a call to action for the medical community. The consistent use of full-body skin examinations including close inspection of mucosal, acral, and genital areas for all patients independent of skin type and racial/ethnic background is paramount. Advancing skin cancer education in skin of color populations, such as through distribution of patient-directed educational materials produced by organizations such as the American Academy of Dermatology, Skin Cancer Foundation, and Skin of Color Society, is an important step toward increased public awareness.16 Use of social and traditional media outlets as well as community-directed health outreach campaigns also are important strategies to change the common misconception that darker-skinned individuals do not get skin cancer. We hope that with a multipronged approach, disparities in skin cancer mortality will steadily be eliminated.
Skin cancers in patients with skin of color are less prevalent but have a higher morbidity and mortality compared to white patients. Challenges to early detection, including clinical differences in presentation, low public awareness, lower index of suspicion among health care providers, and access to specialty care, likely contribute to observed differences in prognosis between skin of color and white populations.
Skin cancer is the most common malignancy in the United States, accounting for approximately 40% of all neoplasms in white patients but only 1% to 4% in Asian American and black patients.1,2 Largely due to the photoprotective effects of increased constitutive epidermal melanin, melanoma is approximately 10 to 20 times less frequent in black patients and 3 to 7 times less common in Hispanics than age-matched whites.1 Nonmelanoma skin cancers including squamous cell carcinoma (SCC) and basal cell carcinoma also are less prevalent in darker skin types.3,4
In the United States, Hispanic, American Indian
Similar to melanoma, the mortality from SCC is disproportionately increased in skin of color populations, ranging from 18% to 29% in black patients.3,10,11 There is a paucity of population-based studies in the United States looking at mortality rates of nonmelanoma skin cancers and their trends over time, but a 1993 study suggests that mortality rates are declining less consistently in black patients than white patients.11
Factors that may contribute to higher mortality rates in patients with skin of color include a greater propensity for inherently aggressive skin cancers (eg, higher risk of SCC) and delays in diagnosis (eg, late-stage diagnosis of melanoma).1,4 For melanoma, increased mortality has been attributed to a predominance of acral lentiginous melanomas, which are more frequently diagnosed at more advanced stages than other melanoma subtypes.6,12,13 Black patients, Hispanics, Asians, and Pacific Islanders are all more likely to present with thicker tumors and metastases on initial presentation than their white counterparts (P<.001).2,8,9,12-14 The higher risk of death from SCC results from the predominance of lesions on non–sun-exposed areas, particularly the legs and anogenital areas, and within sites of chronic scarring or inflammation.4 Unlike sun-induced SCC, the most commonly observed type of SCC in lighter skin types, SCCs that develop in association with chronic inflammatory or ulcerative processes are aggressive and invasive, and they metastasize to distant sites in 20% to 40% of cases (versus 1%–4% in sun-induced SCC).1,3,4 For all skin cancers, poor access to medical care, patients’ unawareness of their skin cancer risk, lack of adequate skin examinations, and prevalence of lesions on uncommon sites that may be inconspicuous or overlooked have all been suggested to delay diagnosis.1,15,16 Given that more advanced disease is associated with worse outcomes, the implications of this delay are enormous and remain a cause for concern.
The alarming skin cancer mortality rates in patients with skin of color are a call to action for the medical community. The consistent use of full-body skin examinations including close inspection of mucosal, acral, and genital areas for all patients independent of skin type and racial/ethnic background is paramount. Advancing skin cancer education in skin of color populations, such as through distribution of patient-directed educational materials produced by organizations such as the American Academy of Dermatology, Skin Cancer Foundation, and Skin of Color Society, is an important step toward increased public awareness.16 Use of social and traditional media outlets as well as community-directed health outreach campaigns also are important strategies to change the common misconception that darker-skinned individuals do not get skin cancer. We hope that with a multipronged approach, disparities in skin cancer mortality will steadily be eliminated.
- Gloster HM Jr, Neal K. Skin cancer in skin of color. J Am Acad Dermatol. 2006;55:741-760; quiz 761-764.
- Cormier JN, Xing Y, Ding M, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med. 2006;166:1907-1914.
- Mora RG, Perniciaro C. Cancer of the skin in blacks: I. a review of 163 black patients with cutaneous squamous cell carcinoma. J Am Acad Dermatol. 1981;5:535-543.
- Halder RM, Bridgeman-Shah S. Skin cancer in African Americans. Cancer. 1995;75:667-673.
- Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2013. Bethesda, MD: National Cancer Institute; April 2016. http://seer.cancer.gov/csr/1975_2013/. Updated September 12, 2016. Accessed April 7, 2017.
- Bellows CF, Belafsky P, Fortgang IS, et al. Melanoma in African-Americans: trends in biological behavior and clinical characteristics over two decades. J Surg Oncol. 2001;78:10-16.
- Chen L, Jin S. Trends in mortality rates of cutaneous melanoma in East Asian populations. Peer J. 2014;4:e2809.
- Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California Cancer Registry data. Cancer Causes Control. 1997;8:246-252.
- Johnson DS, Yamane S, Morita S, et al. Malignant melanoma in non-Caucasians: experience from Hawaii. Surg Clin N Am. 2003;83:275-282.
- Fleming ID, Barnawell JR, Burlison PE, et al. Skin cancer in black patients. Cancer. 1975;35:600-605.
- Weinstock MA. Nonmelanoma skin cancer mortality in the United States, 1969 through 1988. Arch Dermatol. 1993;129:1286-1290.
- Byrd KM, Wilson DC, Hoyler SS. Advanced presentation of melanoma in African Americans. J Am Acad Dermatol. 2004;50:142-143.
- Hu S, Parmet Y, Allen G, et al. Disparity in melanoma: a trend analysis of melanoma incidence and stage at diagnosis among whites, Hispanics, and blacks in Florida. Arch Dermatol. 2009;145:1369-1374.
- Black WC, Goldhahn RT, Wiggins C. Melanoma within a southwestern Hispanic population. Arch Dermatol. 1987;123:1331-1334.
- Harvey VM, Oldfield CW, Chen JT, et al. Melanoma disparities among US Hispanics: use of the social ecological model to contextualize reasons for inequitable outcomes and frame a research agenda [published online August 29, 2016]. J Skin Cancer. 2016;2016:4635740.
- Robinson JK, Joshi KM, Ortiz S, et al. Melanoma knowledge, perception, and awareness in ethnic minorities in Chicago: recommendations regarding education. Psychooncology. 2011;20:313-320.
- Gloster HM Jr, Neal K. Skin cancer in skin of color. J Am Acad Dermatol. 2006;55:741-760; quiz 761-764.
- Cormier JN, Xing Y, Ding M, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med. 2006;166:1907-1914.
- Mora RG, Perniciaro C. Cancer of the skin in blacks: I. a review of 163 black patients with cutaneous squamous cell carcinoma. J Am Acad Dermatol. 1981;5:535-543.
- Halder RM, Bridgeman-Shah S. Skin cancer in African Americans. Cancer. 1995;75:667-673.
- Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2013. Bethesda, MD: National Cancer Institute; April 2016. http://seer.cancer.gov/csr/1975_2013/. Updated September 12, 2016. Accessed April 7, 2017.
- Bellows CF, Belafsky P, Fortgang IS, et al. Melanoma in African-Americans: trends in biological behavior and clinical characteristics over two decades. J Surg Oncol. 2001;78:10-16.
- Chen L, Jin S. Trends in mortality rates of cutaneous melanoma in East Asian populations. Peer J. 2014;4:e2809.
- Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California Cancer Registry data. Cancer Causes Control. 1997;8:246-252.
- Johnson DS, Yamane S, Morita S, et al. Malignant melanoma in non-Caucasians: experience from Hawaii. Surg Clin N Am. 2003;83:275-282.
- Fleming ID, Barnawell JR, Burlison PE, et al. Skin cancer in black patients. Cancer. 1975;35:600-605.
- Weinstock MA. Nonmelanoma skin cancer mortality in the United States, 1969 through 1988. Arch Dermatol. 1993;129:1286-1290.
- Byrd KM, Wilson DC, Hoyler SS. Advanced presentation of melanoma in African Americans. J Am Acad Dermatol. 2004;50:142-143.
- Hu S, Parmet Y, Allen G, et al. Disparity in melanoma: a trend analysis of melanoma incidence and stage at diagnosis among whites, Hispanics, and blacks in Florida. Arch Dermatol. 2009;145:1369-1374.
- Black WC, Goldhahn RT, Wiggins C. Melanoma within a southwestern Hispanic population. Arch Dermatol. 1987;123:1331-1334.
- Harvey VM, Oldfield CW, Chen JT, et al. Melanoma disparities among US Hispanics: use of the social ecological model to contextualize reasons for inequitable outcomes and frame a research agenda [published online August 29, 2016]. J Skin Cancer. 2016;2016:4635740.
- Robinson JK, Joshi KM, Ortiz S, et al. Melanoma knowledge, perception, and awareness in ethnic minorities in Chicago: recommendations regarding education. Psychooncology. 2011;20:313-320.
Elder Abuse: A New Old Problem
Emergency physicians (EPs) are trained to recognize and treat conditions that most severely affect patients at the extremes of age. For decades, the recognition and management of child neglect and abuse has been part of emergency medicine (EM) residency training and most states now require physicians to complete a child abuse course for licensing. In this issue, “Recognizing and Managing Elder Abuse in the Emergency Department” by Rosen et al describes abuse at the other age extreme. The thorough discussion following an illustrative case presentation raises serious concerns that the occurrence of elder neglect and abuse may be increasing in frequency in a rapidly aging population.
Child abuse and elder abuse share several common features along with some notable differences. When a member of either age group presents to the ED with fractures and/or soft tissue injuries, EPs must maintain a high index of suspicion for abuse, obtain a carefully directed history, and be able to recognize the characteristic patterns of injury. Hallmarks of both child abuse and elder abuse include a history provided by the caregiver that is not consistent with the actual injuries; an often-unobtainable independent history from an infant or baby not yet able to speak or an older adult with dementia; and a physical exam revealing bruises in areas that are not over bony prominences. A radiographic skeletal survey may show multiple fractures in various stages of healing, and laboratory testing may reveal nutritional evidence of neglect, medication over- or underdosing, or the presence of medications that have not been prescribed for the patient.
Patterns of abuse injuries in the two age groups differ. As noted by Colbourne and Clarke in Tintinalli’s Emergency Medicine (8th ed, p. 1001), nonaccidental bruises in children are more common on the torso, neck, ears, cheeks, buttocks, and back; appear in clusters; are frequently symmetrical; and tend to be larger and more numerous than accidental injuries. Hand or implement patterns on the skin may be observed. Rib and metaphyseal fractures are unusual in children, as are all fractures at a very young age.
In the midst of an epidemic of elderly fall injuries, abuse injuries, as described in the pages ahead, most commonly occur on the head, neck, and upper extremities, and include large bruises on the face, lateral arm, or posterior torso. Based on preliminary results from an ongoing study, left periorbital, neck, and ulnar forearm injuries appear to be particularly indicative of abuse rather than accidental. An elderly person may be abused by an adult-child or relative living in the same household attempting to gain control over the victim’s wealth or residence.
Interventional resources required for both types of abuse, as well as for intimate partner abuse, are also similar and include safe facilities for extended treatment and separation from a suspected abuser; hospital security, legal, and administrative support; social services; law enforcement; psychiatric evaluation of adult capacity; and child or adult protective services, which, as Rosen et al note, operate very differently from one another. All states, except one, now require reporting of both child abuse and elder abuse.*
None of these comparisons of child abuse and elder abuse are meant to suggest equivalency—moral or otherwise. Children are not “little adults,” and the frail elderly are not truly “child-like.” Each incident of a child “slipping through the cracks” of the protective measures currently in place underscores the need for sufficient resources to deal with child abuse alone, and an increasing number of elder abuse cases should not compete with these needs. But implementing greater awareness, preventive measures, and physical and human resources to address these problems at both extremes of age cannot be put off for the future.
When I started the first geriatric emergency medicine fellowship in the country in 2005, elder abuse was not even on my radar screen. Now it must be considered a serious and growing problem by all.
*New York State alone does not require reporting of elder abuse.
Emergency physicians (EPs) are trained to recognize and treat conditions that most severely affect patients at the extremes of age. For decades, the recognition and management of child neglect and abuse has been part of emergency medicine (EM) residency training and most states now require physicians to complete a child abuse course for licensing. In this issue, “Recognizing and Managing Elder Abuse in the Emergency Department” by Rosen et al describes abuse at the other age extreme. The thorough discussion following an illustrative case presentation raises serious concerns that the occurrence of elder neglect and abuse may be increasing in frequency in a rapidly aging population.
Child abuse and elder abuse share several common features along with some notable differences. When a member of either age group presents to the ED with fractures and/or soft tissue injuries, EPs must maintain a high index of suspicion for abuse, obtain a carefully directed history, and be able to recognize the characteristic patterns of injury. Hallmarks of both child abuse and elder abuse include a history provided by the caregiver that is not consistent with the actual injuries; an often-unobtainable independent history from an infant or baby not yet able to speak or an older adult with dementia; and a physical exam revealing bruises in areas that are not over bony prominences. A radiographic skeletal survey may show multiple fractures in various stages of healing, and laboratory testing may reveal nutritional evidence of neglect, medication over- or underdosing, or the presence of medications that have not been prescribed for the patient.
Patterns of abuse injuries in the two age groups differ. As noted by Colbourne and Clarke in Tintinalli’s Emergency Medicine (8th ed, p. 1001), nonaccidental bruises in children are more common on the torso, neck, ears, cheeks, buttocks, and back; appear in clusters; are frequently symmetrical; and tend to be larger and more numerous than accidental injuries. Hand or implement patterns on the skin may be observed. Rib and metaphyseal fractures are unusual in children, as are all fractures at a very young age.
In the midst of an epidemic of elderly fall injuries, abuse injuries, as described in the pages ahead, most commonly occur on the head, neck, and upper extremities, and include large bruises on the face, lateral arm, or posterior torso. Based on preliminary results from an ongoing study, left periorbital, neck, and ulnar forearm injuries appear to be particularly indicative of abuse rather than accidental. An elderly person may be abused by an adult-child or relative living in the same household attempting to gain control over the victim’s wealth or residence.
Interventional resources required for both types of abuse, as well as for intimate partner abuse, are also similar and include safe facilities for extended treatment and separation from a suspected abuser; hospital security, legal, and administrative support; social services; law enforcement; psychiatric evaluation of adult capacity; and child or adult protective services, which, as Rosen et al note, operate very differently from one another. All states, except one, now require reporting of both child abuse and elder abuse.*
None of these comparisons of child abuse and elder abuse are meant to suggest equivalency—moral or otherwise. Children are not “little adults,” and the frail elderly are not truly “child-like.” Each incident of a child “slipping through the cracks” of the protective measures currently in place underscores the need for sufficient resources to deal with child abuse alone, and an increasing number of elder abuse cases should not compete with these needs. But implementing greater awareness, preventive measures, and physical and human resources to address these problems at both extremes of age cannot be put off for the future.
When I started the first geriatric emergency medicine fellowship in the country in 2005, elder abuse was not even on my radar screen. Now it must be considered a serious and growing problem by all.
*New York State alone does not require reporting of elder abuse.
Emergency physicians (EPs) are trained to recognize and treat conditions that most severely affect patients at the extremes of age. For decades, the recognition and management of child neglect and abuse has been part of emergency medicine (EM) residency training and most states now require physicians to complete a child abuse course for licensing. In this issue, “Recognizing and Managing Elder Abuse in the Emergency Department” by Rosen et al describes abuse at the other age extreme. The thorough discussion following an illustrative case presentation raises serious concerns that the occurrence of elder neglect and abuse may be increasing in frequency in a rapidly aging population.
Child abuse and elder abuse share several common features along with some notable differences. When a member of either age group presents to the ED with fractures and/or soft tissue injuries, EPs must maintain a high index of suspicion for abuse, obtain a carefully directed history, and be able to recognize the characteristic patterns of injury. Hallmarks of both child abuse and elder abuse include a history provided by the caregiver that is not consistent with the actual injuries; an often-unobtainable independent history from an infant or baby not yet able to speak or an older adult with dementia; and a physical exam revealing bruises in areas that are not over bony prominences. A radiographic skeletal survey may show multiple fractures in various stages of healing, and laboratory testing may reveal nutritional evidence of neglect, medication over- or underdosing, or the presence of medications that have not been prescribed for the patient.
Patterns of abuse injuries in the two age groups differ. As noted by Colbourne and Clarke in Tintinalli’s Emergency Medicine (8th ed, p. 1001), nonaccidental bruises in children are more common on the torso, neck, ears, cheeks, buttocks, and back; appear in clusters; are frequently symmetrical; and tend to be larger and more numerous than accidental injuries. Hand or implement patterns on the skin may be observed. Rib and metaphyseal fractures are unusual in children, as are all fractures at a very young age.
In the midst of an epidemic of elderly fall injuries, abuse injuries, as described in the pages ahead, most commonly occur on the head, neck, and upper extremities, and include large bruises on the face, lateral arm, or posterior torso. Based on preliminary results from an ongoing study, left periorbital, neck, and ulnar forearm injuries appear to be particularly indicative of abuse rather than accidental. An elderly person may be abused by an adult-child or relative living in the same household attempting to gain control over the victim’s wealth or residence.
Interventional resources required for both types of abuse, as well as for intimate partner abuse, are also similar and include safe facilities for extended treatment and separation from a suspected abuser; hospital security, legal, and administrative support; social services; law enforcement; psychiatric evaluation of adult capacity; and child or adult protective services, which, as Rosen et al note, operate very differently from one another. All states, except one, now require reporting of both child abuse and elder abuse.*
None of these comparisons of child abuse and elder abuse are meant to suggest equivalency—moral or otherwise. Children are not “little adults,” and the frail elderly are not truly “child-like.” Each incident of a child “slipping through the cracks” of the protective measures currently in place underscores the need for sufficient resources to deal with child abuse alone, and an increasing number of elder abuse cases should not compete with these needs. But implementing greater awareness, preventive measures, and physical and human resources to address these problems at both extremes of age cannot be put off for the future.
When I started the first geriatric emergency medicine fellowship in the country in 2005, elder abuse was not even on my radar screen. Now it must be considered a serious and growing problem by all.
*New York State alone does not require reporting of elder abuse.
Providing Mental Health Care to All Veterans Regardless of Discharge Status
During his testimony before the House Committee on Veterans Affairs on March 7, 2017, Secretary of Veterans Affairs David J. Shulkin, MD, expressed his intent to remove the administrative barrier prohibiting other-than-honorably (OTH) discharged service members from receiving VHA mental health care. This is the first time in VA history to integrate those veterans whose OTH discharge status had previously disenfranchised them.
In his comments to Congress, Dr. Shulkin voiced his gratitude to Rep. Mike Coffman (R-CO) for helping him to “better understand the urgency of getting this right.” In March 2016, Rep. Coffman introduced the
Veterans Fairness Act, which would permit OTH discharged combat veterans to obtain emergency mental health services. Rep. Coffman cited that 22,000 U.S. Army veterans were discharged for misconduct since 2009, most with a traumatic brain injury (TBI) or mental illness.1 Veterans often refer to OTH discharges as having “bad paper.” In 2013, National Public Radio produced a series on OTH discharged service members that underscored their struggles.2 Those reports estimated that more than 100,000 veterans left the service with OTH discharges in the decade before the story.2
These individuals, many of whom have already lost a great deal as a result of their military service, lose much more when they are OTH discharged. They are unable to apply for the GI Bill, which enables them to further their education and livelihood; they cannot get a VA home loan to help them house their families; and they are ineligible for disability even for combat-related conditions like posttraumatic stress disorder (PTSD) and TBI. Most damaging of all, until Dr. Shulkin’s historic announcement, they also could not get VA health care. In effect, OTH discharge status creates a second class of service men and women, even though the discharge may have been the result of injury and illness related to their time in uniform. That consequence makes Dr. Shulkin’s proposal not only an administrative change, but also an ethical decision regarding the civil and human rights of service members, which is the reason most major veterans service and advocacy organizations have long endorsed it.
Although research on OTH discharged veterans has been limited, studies have found a high rate of mental health problems. The OTH discharged service members are significantly represented in the cohorts who face some of the most serious public health problems that the VA has tried to address through new programs that were initiated during the prior administration and continued by the current one, such as ending homelessness and preventing suicide.
A 2017 study compared rates of mental illness and substance use among veterans with routine discharges with those who had nonroutine separations from the military.3 The results showed that there was a higher rate of almost every psychiatric diagnosis in the nonroutine discharges; the rates were particularly high for those discharged for misconduct.3 Because of the established correlation of multiple deployments to Afghanistan and Iraq and incidence of TBI, PTSD, and substance use and the association of these disorders with behaviors that contribute to OTH discharge status, a clear duty to care for these men and women emerges.
Similarly, the ethical principle of nonmalfeasance provides persuasive justification for Dr. Shulkin’s proposed change in VA eligibility for mental health care. The study also found that even if not previously entitled to VA services, these veterans share the increased risk of suicide found in all those who have worn a uniform for their country and similarly need compassionate, competent veteran-centered care.3
Recent research showed that patients who receive mental health care within the VA have lower rates of suicide than that of those who receive care in the community.4 The results of this study contribute to the ethical imperative to grant these former service members access to potentially life-saving mental health treatment more urgent.
The elevated suicide risk of those veterans who do not have VA mental health services makes this extension of care clinically and ethically imperative and urgent. In his testimony at the hearing, Dr. Shulkin underscored this rationale, “The President and I have made it clear that suicide prevention is one of our top priorities,” Shulkin added. “We know the rate of death by suicide among veterans who do not use VA care is increasing at a greater rate than veterans who use VA care. This is a national emergency that requires bold action. We must and we will do all that we can to help former service members who may be at risk. When we say even one veteran suicide is one too many, we mean it.
The downstream consequences of OTH discharge status are the most detrimental to the veteran and have negative effects on the veteran’s family and community. Nonroutinely discharged veterans are more likely to be homeless. The new initiative would open a variety of VA mental health services to OTH discharged service members, including those available in VA emergency departments, Vet Centers, and the Veterans Crisis Line. In developing the plan to expand coverage to OTH discharged veterans, Dr. Shulkin indicated that he would consult with Veterans Service Organizations and the DoD.
We can hope that additional services will be opened to OTH discharged service members, such as case management and housing assistance, which have proven so successful in reintegrating those service members with routine discharges.
1. Roeder T. Rep. Coffman renews push for mental health care for all veterans who need it. http://gazette.com/rep.-coffman-renews-push-for-mental-health-care-for-all-veterans-who-need-it/article/1596355. Updated February 9, 2017. Access April 10, 2017.
2. Peñaloza M, Lawrence Q. Veterans and otherthan-honorable discharges. http://www.npr.orgseries/250013036/veterans-and-other-than-honorable-discharges. Published December 12, 2013. Accessed April 10, 2017.
3. Brignone E, Fargo JD, Blais RK, Carter ME, Samore MH, Gundlapalli AV. Non-routine discharge from military service: mental illness, substance use disorders, and suicidality. Am J Prev Med. 2017;52(5):557-565.
4. Hoffmire CA, Kemp Je, Bossarte RM. Changes in suicide mortality for veterans and non-veterans by gender and history of VHA service use, 2000-2010. Psychiatr Serv. 2015;66(9):959-965.
During his testimony before the House Committee on Veterans Affairs on March 7, 2017, Secretary of Veterans Affairs David J. Shulkin, MD, expressed his intent to remove the administrative barrier prohibiting other-than-honorably (OTH) discharged service members from receiving VHA mental health care. This is the first time in VA history to integrate those veterans whose OTH discharge status had previously disenfranchised them.
In his comments to Congress, Dr. Shulkin voiced his gratitude to Rep. Mike Coffman (R-CO) for helping him to “better understand the urgency of getting this right.” In March 2016, Rep. Coffman introduced the
Veterans Fairness Act, which would permit OTH discharged combat veterans to obtain emergency mental health services. Rep. Coffman cited that 22,000 U.S. Army veterans were discharged for misconduct since 2009, most with a traumatic brain injury (TBI) or mental illness.1 Veterans often refer to OTH discharges as having “bad paper.” In 2013, National Public Radio produced a series on OTH discharged service members that underscored their struggles.2 Those reports estimated that more than 100,000 veterans left the service with OTH discharges in the decade before the story.2
These individuals, many of whom have already lost a great deal as a result of their military service, lose much more when they are OTH discharged. They are unable to apply for the GI Bill, which enables them to further their education and livelihood; they cannot get a VA home loan to help them house their families; and they are ineligible for disability even for combat-related conditions like posttraumatic stress disorder (PTSD) and TBI. Most damaging of all, until Dr. Shulkin’s historic announcement, they also could not get VA health care. In effect, OTH discharge status creates a second class of service men and women, even though the discharge may have been the result of injury and illness related to their time in uniform. That consequence makes Dr. Shulkin’s proposal not only an administrative change, but also an ethical decision regarding the civil and human rights of service members, which is the reason most major veterans service and advocacy organizations have long endorsed it.
Although research on OTH discharged veterans has been limited, studies have found a high rate of mental health problems. The OTH discharged service members are significantly represented in the cohorts who face some of the most serious public health problems that the VA has tried to address through new programs that were initiated during the prior administration and continued by the current one, such as ending homelessness and preventing suicide.
A 2017 study compared rates of mental illness and substance use among veterans with routine discharges with those who had nonroutine separations from the military.3 The results showed that there was a higher rate of almost every psychiatric diagnosis in the nonroutine discharges; the rates were particularly high for those discharged for misconduct.3 Because of the established correlation of multiple deployments to Afghanistan and Iraq and incidence of TBI, PTSD, and substance use and the association of these disorders with behaviors that contribute to OTH discharge status, a clear duty to care for these men and women emerges.
Similarly, the ethical principle of nonmalfeasance provides persuasive justification for Dr. Shulkin’s proposed change in VA eligibility for mental health care. The study also found that even if not previously entitled to VA services, these veterans share the increased risk of suicide found in all those who have worn a uniform for their country and similarly need compassionate, competent veteran-centered care.3
Recent research showed that patients who receive mental health care within the VA have lower rates of suicide than that of those who receive care in the community.4 The results of this study contribute to the ethical imperative to grant these former service members access to potentially life-saving mental health treatment more urgent.
The elevated suicide risk of those veterans who do not have VA mental health services makes this extension of care clinically and ethically imperative and urgent. In his testimony at the hearing, Dr. Shulkin underscored this rationale, “The President and I have made it clear that suicide prevention is one of our top priorities,” Shulkin added. “We know the rate of death by suicide among veterans who do not use VA care is increasing at a greater rate than veterans who use VA care. This is a national emergency that requires bold action. We must and we will do all that we can to help former service members who may be at risk. When we say even one veteran suicide is one too many, we mean it.
The downstream consequences of OTH discharge status are the most detrimental to the veteran and have negative effects on the veteran’s family and community. Nonroutinely discharged veterans are more likely to be homeless. The new initiative would open a variety of VA mental health services to OTH discharged service members, including those available in VA emergency departments, Vet Centers, and the Veterans Crisis Line. In developing the plan to expand coverage to OTH discharged veterans, Dr. Shulkin indicated that he would consult with Veterans Service Organizations and the DoD.
We can hope that additional services will be opened to OTH discharged service members, such as case management and housing assistance, which have proven so successful in reintegrating those service members with routine discharges.
During his testimony before the House Committee on Veterans Affairs on March 7, 2017, Secretary of Veterans Affairs David J. Shulkin, MD, expressed his intent to remove the administrative barrier prohibiting other-than-honorably (OTH) discharged service members from receiving VHA mental health care. This is the first time in VA history to integrate those veterans whose OTH discharge status had previously disenfranchised them.
In his comments to Congress, Dr. Shulkin voiced his gratitude to Rep. Mike Coffman (R-CO) for helping him to “better understand the urgency of getting this right.” In March 2016, Rep. Coffman introduced the
Veterans Fairness Act, which would permit OTH discharged combat veterans to obtain emergency mental health services. Rep. Coffman cited that 22,000 U.S. Army veterans were discharged for misconduct since 2009, most with a traumatic brain injury (TBI) or mental illness.1 Veterans often refer to OTH discharges as having “bad paper.” In 2013, National Public Radio produced a series on OTH discharged service members that underscored their struggles.2 Those reports estimated that more than 100,000 veterans left the service with OTH discharges in the decade before the story.2
These individuals, many of whom have already lost a great deal as a result of their military service, lose much more when they are OTH discharged. They are unable to apply for the GI Bill, which enables them to further their education and livelihood; they cannot get a VA home loan to help them house their families; and they are ineligible for disability even for combat-related conditions like posttraumatic stress disorder (PTSD) and TBI. Most damaging of all, until Dr. Shulkin’s historic announcement, they also could not get VA health care. In effect, OTH discharge status creates a second class of service men and women, even though the discharge may have been the result of injury and illness related to their time in uniform. That consequence makes Dr. Shulkin’s proposal not only an administrative change, but also an ethical decision regarding the civil and human rights of service members, which is the reason most major veterans service and advocacy organizations have long endorsed it.
Although research on OTH discharged veterans has been limited, studies have found a high rate of mental health problems. The OTH discharged service members are significantly represented in the cohorts who face some of the most serious public health problems that the VA has tried to address through new programs that were initiated during the prior administration and continued by the current one, such as ending homelessness and preventing suicide.
A 2017 study compared rates of mental illness and substance use among veterans with routine discharges with those who had nonroutine separations from the military.3 The results showed that there was a higher rate of almost every psychiatric diagnosis in the nonroutine discharges; the rates were particularly high for those discharged for misconduct.3 Because of the established correlation of multiple deployments to Afghanistan and Iraq and incidence of TBI, PTSD, and substance use and the association of these disorders with behaviors that contribute to OTH discharge status, a clear duty to care for these men and women emerges.
Similarly, the ethical principle of nonmalfeasance provides persuasive justification for Dr. Shulkin’s proposed change in VA eligibility for mental health care. The study also found that even if not previously entitled to VA services, these veterans share the increased risk of suicide found in all those who have worn a uniform for their country and similarly need compassionate, competent veteran-centered care.3
Recent research showed that patients who receive mental health care within the VA have lower rates of suicide than that of those who receive care in the community.4 The results of this study contribute to the ethical imperative to grant these former service members access to potentially life-saving mental health treatment more urgent.
The elevated suicide risk of those veterans who do not have VA mental health services makes this extension of care clinically and ethically imperative and urgent. In his testimony at the hearing, Dr. Shulkin underscored this rationale, “The President and I have made it clear that suicide prevention is one of our top priorities,” Shulkin added. “We know the rate of death by suicide among veterans who do not use VA care is increasing at a greater rate than veterans who use VA care. This is a national emergency that requires bold action. We must and we will do all that we can to help former service members who may be at risk. When we say even one veteran suicide is one too many, we mean it.
The downstream consequences of OTH discharge status are the most detrimental to the veteran and have negative effects on the veteran’s family and community. Nonroutinely discharged veterans are more likely to be homeless. The new initiative would open a variety of VA mental health services to OTH discharged service members, including those available in VA emergency departments, Vet Centers, and the Veterans Crisis Line. In developing the plan to expand coverage to OTH discharged veterans, Dr. Shulkin indicated that he would consult with Veterans Service Organizations and the DoD.
We can hope that additional services will be opened to OTH discharged service members, such as case management and housing assistance, which have proven so successful in reintegrating those service members with routine discharges.
1. Roeder T. Rep. Coffman renews push for mental health care for all veterans who need it. http://gazette.com/rep.-coffman-renews-push-for-mental-health-care-for-all-veterans-who-need-it/article/1596355. Updated February 9, 2017. Access April 10, 2017.
2. Peñaloza M, Lawrence Q. Veterans and otherthan-honorable discharges. http://www.npr.orgseries/250013036/veterans-and-other-than-honorable-discharges. Published December 12, 2013. Accessed April 10, 2017.
3. Brignone E, Fargo JD, Blais RK, Carter ME, Samore MH, Gundlapalli AV. Non-routine discharge from military service: mental illness, substance use disorders, and suicidality. Am J Prev Med. 2017;52(5):557-565.
4. Hoffmire CA, Kemp Je, Bossarte RM. Changes in suicide mortality for veterans and non-veterans by gender and history of VHA service use, 2000-2010. Psychiatr Serv. 2015;66(9):959-965.
1. Roeder T. Rep. Coffman renews push for mental health care for all veterans who need it. http://gazette.com/rep.-coffman-renews-push-for-mental-health-care-for-all-veterans-who-need-it/article/1596355. Updated February 9, 2017. Access April 10, 2017.
2. Peñaloza M, Lawrence Q. Veterans and otherthan-honorable discharges. http://www.npr.orgseries/250013036/veterans-and-other-than-honorable-discharges. Published December 12, 2013. Accessed April 10, 2017.
3. Brignone E, Fargo JD, Blais RK, Carter ME, Samore MH, Gundlapalli AV. Non-routine discharge from military service: mental illness, substance use disorders, and suicidality. Am J Prev Med. 2017;52(5):557-565.
4. Hoffmire CA, Kemp Je, Bossarte RM. Changes in suicide mortality for veterans and non-veterans by gender and history of VHA service use, 2000-2010. Psychiatr Serv. 2015;66(9):959-965.
Clearing the Air
The month of May marks peak season for asthma and allergies. Coincidentally, May is also Asthma and Allergy Awareness and Clean Air month. Seems a perfect match, don’t you agree? Air quality plays a menacing role in the manifestation of asthma and allergies. Let’s take a closer look at that synergy of poor air quality and respiratory sequelae and discuss approaches to mitigate these problems.
Clean air has a natural balance of gases (ie, oxygen, nitrogen, carbon dioxide) and does not contain pollutants or allergens.1 The Environmental Protection Agency (EPA), whose mission is to protect human health and the environment, identifies six common air pollutants: particulate matter (eg, dust, dirt, soot, smoke), photochemical oxidants (including ozone), carbon monoxide, sulfur dioxide, nitrogen dioxide, and lead.2 The EPA has five goals in their strategic plan, the first of which is “Addressing Climate Change and Improving Air Quality.”3 The agency is working with a multitude of stakeholders to achieve its mission of reducing air pollution. One major way we can contribute to this goal is to use public transportation, walk, or ride a bike instead of driving a car.
As we know, pollutants have detrimental consequences not only on air quality, but the environment as a whole. Pollutants infiltrate our lakes and rivers and can also damage plants and trees. Recall from your early science classes the role that plants and trees have as natural air cleaners—scrubbers, if you will. They rid the air of carbon dioxide and can also remove formaldehyde, benzene, and a host of other toxins. Trees can eradicate gaseous pollutants and airborne particles.4 But these natural air scrubbers are slowly being destroyed by the overwhelming amount of pollutants—and our short- and long-term health will ultimately pay the price. These pollutants contribute to the risk for cardiovascular and respiratory diseases, primarily asthma.5
Surveillance data show the deadly, disruptive, and costly impact asthma has on the nation.6 Asthma is a serious, potentially life-threatening chronic respiratory disease that affects quality of life in more than 24 million Americans, including an estimated six million children.7 Moreover, it is one of the most common diseases associated with poor air quality. Environmental asthma triggers include pollen, chemicals, extreme weather changes, and smoke. Educating patients with asthma on how to avoid these triggers will help them manage their disease and lessen the disruption it can cause. Additionally, a concerted effort to minimize our contribution to air pollution will assist in reducing the incidence of asthma caused by external factors.
Creating an “asthma-friendly environment” is recommended by the American Lung Association. Some might view this as a major undertaking, but simple steps such as avoiding use of aerosol products, changing the air filter in HVAC units, and dusting and vacuuming the house frequently are low-cost, effective approaches to clean the air. Avoiding perfumes and perfumed products is another way to reduce indoor triggers.
In addition to trigger avoidance, proper management of asthma is essential. Vigilance in screening for early indicators of asthma and prompt initiation of treatment can decrease or eliminate the development of long-term consequences, such as COPD and asthma-COPD overlap syndrome.8
Allergies, which affect more than 50 million people in the US, can also result from poor air quality.9 In some cases, allergies are food-based and therefore fairly easy to evade. But others are induced by pollen, pet dander, and other air-based allergens. How do we manage these pesky allergens so we can breathe easy? While the field of allergy research continues to grow, we rely heavily on allergen-specific immunotherapy as a potentially curative treat
In order for a house to be allergen-free, it should also be plant-free, right? Contrary to popular belief, this is not the case! Not all plants are problematic. A houseplant can remove formaldehyde, benzene, and a multitude of other toxins that typically reside in indoor air. Plants such as areca palm, pothos (known as Mother-in-Law’s Tongue), and the Money Plant have been shown to improve air quality.10 Additionally, decreasing or eliminating the use of room deodorizers and other allergen-containing products can reduce flare-ups. More and more frequently, we see notices in offices and at conferences to avoid the use of perfumes; this is an effective measure for the safety and comfort of those with allergies.
Alas, outdoor allergens are a bit more difficult to manage. Airborne allergens exist in such high quantities that they are nearly impossible to elude when in season.11 Keeping windows closed, wiping down surfaces where pollen and dust collects, and avoiding flowering plants can help reduce contact with allergens.
Lowering the incidence of air pollution and the symptoms of allergies and asthma can improve quality of life. By implementing these approaches to contribute to cleaner air and reduce triggers, we can help our patients and ourselves.
1. Clean air month 2017. www.whathealth.com/awareness/event/cleanairmonth.html. Accessed April 4, 2017.
2. United States Environmental Protection Agency (EPA). Particulate matter (PM) pollution. www.epa.gov/pm-pollution. Accessed April 19, 2017.
3. United States Environmental Protection Agency (EPA). EPA strategic plan. www.epa.gov/planandbudget/strategicplan. Accessed April 4, 2017.
4. Downing A. Air: what’s a tree got to do with it? www.ecology.com/2011/09/02/air-and-trees. Accessed April 4, 2017.
5. World Health Organization. Ambient (outdoor) air quality and health. www.who.int/mediacentre/factsheets/fs313/en. Ac-cessed April 4, 2017.
6. CDC. Asthma’s impact on the nation. www.cdc.gov/asthma/impacts_nation/default.htm. Accessed April 4, 2017.
7. CDC. Asthma. www.cdc.gov/asthma/default.htm. Accessed April 4, 2017.
8. Bui DS, Burgess JA, Lowe AJ, et al. Childhood lung function predicts adult COPD and asthma-COPD overlap syndrome (ACOS). Am J Respir Crit Care Med. 2017 Feb 1. [Epub ahead of print]
9. Doheny K. Living with severe allergies. www.webmd.com/allergies/features/severe-allergies#1. Accessed April 4, 2017.
10. Gromicko N, Tarasenko K. Plants and indoor air quality. www.nachi.org/plants-indoor-air-quality.htm. Accessed April 4, 2017.
11. Asthma and Allergy Foundation of America. Fight the cause of allergy. http://fightthecauseofallergy.org/page/fight. Accessed April 4, 2017.
The month of May marks peak season for asthma and allergies. Coincidentally, May is also Asthma and Allergy Awareness and Clean Air month. Seems a perfect match, don’t you agree? Air quality plays a menacing role in the manifestation of asthma and allergies. Let’s take a closer look at that synergy of poor air quality and respiratory sequelae and discuss approaches to mitigate these problems.
Clean air has a natural balance of gases (ie, oxygen, nitrogen, carbon dioxide) and does not contain pollutants or allergens.1 The Environmental Protection Agency (EPA), whose mission is to protect human health and the environment, identifies six common air pollutants: particulate matter (eg, dust, dirt, soot, smoke), photochemical oxidants (including ozone), carbon monoxide, sulfur dioxide, nitrogen dioxide, and lead.2 The EPA has five goals in their strategic plan, the first of which is “Addressing Climate Change and Improving Air Quality.”3 The agency is working with a multitude of stakeholders to achieve its mission of reducing air pollution. One major way we can contribute to this goal is to use public transportation, walk, or ride a bike instead of driving a car.
As we know, pollutants have detrimental consequences not only on air quality, but the environment as a whole. Pollutants infiltrate our lakes and rivers and can also damage plants and trees. Recall from your early science classes the role that plants and trees have as natural air cleaners—scrubbers, if you will. They rid the air of carbon dioxide and can also remove formaldehyde, benzene, and a host of other toxins. Trees can eradicate gaseous pollutants and airborne particles.4 But these natural air scrubbers are slowly being destroyed by the overwhelming amount of pollutants—and our short- and long-term health will ultimately pay the price. These pollutants contribute to the risk for cardiovascular and respiratory diseases, primarily asthma.5
Surveillance data show the deadly, disruptive, and costly impact asthma has on the nation.6 Asthma is a serious, potentially life-threatening chronic respiratory disease that affects quality of life in more than 24 million Americans, including an estimated six million children.7 Moreover, it is one of the most common diseases associated with poor air quality. Environmental asthma triggers include pollen, chemicals, extreme weather changes, and smoke. Educating patients with asthma on how to avoid these triggers will help them manage their disease and lessen the disruption it can cause. Additionally, a concerted effort to minimize our contribution to air pollution will assist in reducing the incidence of asthma caused by external factors.
Creating an “asthma-friendly environment” is recommended by the American Lung Association. Some might view this as a major undertaking, but simple steps such as avoiding use of aerosol products, changing the air filter in HVAC units, and dusting and vacuuming the house frequently are low-cost, effective approaches to clean the air. Avoiding perfumes and perfumed products is another way to reduce indoor triggers.
In addition to trigger avoidance, proper management of asthma is essential. Vigilance in screening for early indicators of asthma and prompt initiation of treatment can decrease or eliminate the development of long-term consequences, such as COPD and asthma-COPD overlap syndrome.8
Allergies, which affect more than 50 million people in the US, can also result from poor air quality.9 In some cases, allergies are food-based and therefore fairly easy to evade. But others are induced by pollen, pet dander, and other air-based allergens. How do we manage these pesky allergens so we can breathe easy? While the field of allergy research continues to grow, we rely heavily on allergen-specific immunotherapy as a potentially curative treat
In order for a house to be allergen-free, it should also be plant-free, right? Contrary to popular belief, this is not the case! Not all plants are problematic. A houseplant can remove formaldehyde, benzene, and a multitude of other toxins that typically reside in indoor air. Plants such as areca palm, pothos (known as Mother-in-Law’s Tongue), and the Money Plant have been shown to improve air quality.10 Additionally, decreasing or eliminating the use of room deodorizers and other allergen-containing products can reduce flare-ups. More and more frequently, we see notices in offices and at conferences to avoid the use of perfumes; this is an effective measure for the safety and comfort of those with allergies.
Alas, outdoor allergens are a bit more difficult to manage. Airborne allergens exist in such high quantities that they are nearly impossible to elude when in season.11 Keeping windows closed, wiping down surfaces where pollen and dust collects, and avoiding flowering plants can help reduce contact with allergens.
Lowering the incidence of air pollution and the symptoms of allergies and asthma can improve quality of life. By implementing these approaches to contribute to cleaner air and reduce triggers, we can help our patients and ourselves.
The month of May marks peak season for asthma and allergies. Coincidentally, May is also Asthma and Allergy Awareness and Clean Air month. Seems a perfect match, don’t you agree? Air quality plays a menacing role in the manifestation of asthma and allergies. Let’s take a closer look at that synergy of poor air quality and respiratory sequelae and discuss approaches to mitigate these problems.
Clean air has a natural balance of gases (ie, oxygen, nitrogen, carbon dioxide) and does not contain pollutants or allergens.1 The Environmental Protection Agency (EPA), whose mission is to protect human health and the environment, identifies six common air pollutants: particulate matter (eg, dust, dirt, soot, smoke), photochemical oxidants (including ozone), carbon monoxide, sulfur dioxide, nitrogen dioxide, and lead.2 The EPA has five goals in their strategic plan, the first of which is “Addressing Climate Change and Improving Air Quality.”3 The agency is working with a multitude of stakeholders to achieve its mission of reducing air pollution. One major way we can contribute to this goal is to use public transportation, walk, or ride a bike instead of driving a car.
As we know, pollutants have detrimental consequences not only on air quality, but the environment as a whole. Pollutants infiltrate our lakes and rivers and can also damage plants and trees. Recall from your early science classes the role that plants and trees have as natural air cleaners—scrubbers, if you will. They rid the air of carbon dioxide and can also remove formaldehyde, benzene, and a host of other toxins. Trees can eradicate gaseous pollutants and airborne particles.4 But these natural air scrubbers are slowly being destroyed by the overwhelming amount of pollutants—and our short- and long-term health will ultimately pay the price. These pollutants contribute to the risk for cardiovascular and respiratory diseases, primarily asthma.5
Surveillance data show the deadly, disruptive, and costly impact asthma has on the nation.6 Asthma is a serious, potentially life-threatening chronic respiratory disease that affects quality of life in more than 24 million Americans, including an estimated six million children.7 Moreover, it is one of the most common diseases associated with poor air quality. Environmental asthma triggers include pollen, chemicals, extreme weather changes, and smoke. Educating patients with asthma on how to avoid these triggers will help them manage their disease and lessen the disruption it can cause. Additionally, a concerted effort to minimize our contribution to air pollution will assist in reducing the incidence of asthma caused by external factors.
Creating an “asthma-friendly environment” is recommended by the American Lung Association. Some might view this as a major undertaking, but simple steps such as avoiding use of aerosol products, changing the air filter in HVAC units, and dusting and vacuuming the house frequently are low-cost, effective approaches to clean the air. Avoiding perfumes and perfumed products is another way to reduce indoor triggers.
In addition to trigger avoidance, proper management of asthma is essential. Vigilance in screening for early indicators of asthma and prompt initiation of treatment can decrease or eliminate the development of long-term consequences, such as COPD and asthma-COPD overlap syndrome.8
Allergies, which affect more than 50 million people in the US, can also result from poor air quality.9 In some cases, allergies are food-based and therefore fairly easy to evade. But others are induced by pollen, pet dander, and other air-based allergens. How do we manage these pesky allergens so we can breathe easy? While the field of allergy research continues to grow, we rely heavily on allergen-specific immunotherapy as a potentially curative treat
In order for a house to be allergen-free, it should also be plant-free, right? Contrary to popular belief, this is not the case! Not all plants are problematic. A houseplant can remove formaldehyde, benzene, and a multitude of other toxins that typically reside in indoor air. Plants such as areca palm, pothos (known as Mother-in-Law’s Tongue), and the Money Plant have been shown to improve air quality.10 Additionally, decreasing or eliminating the use of room deodorizers and other allergen-containing products can reduce flare-ups. More and more frequently, we see notices in offices and at conferences to avoid the use of perfumes; this is an effective measure for the safety and comfort of those with allergies.
Alas, outdoor allergens are a bit more difficult to manage. Airborne allergens exist in such high quantities that they are nearly impossible to elude when in season.11 Keeping windows closed, wiping down surfaces where pollen and dust collects, and avoiding flowering plants can help reduce contact with allergens.
Lowering the incidence of air pollution and the symptoms of allergies and asthma can improve quality of life. By implementing these approaches to contribute to cleaner air and reduce triggers, we can help our patients and ourselves.
1. Clean air month 2017. www.whathealth.com/awareness/event/cleanairmonth.html. Accessed April 4, 2017.
2. United States Environmental Protection Agency (EPA). Particulate matter (PM) pollution. www.epa.gov/pm-pollution. Accessed April 19, 2017.
3. United States Environmental Protection Agency (EPA). EPA strategic plan. www.epa.gov/planandbudget/strategicplan. Accessed April 4, 2017.
4. Downing A. Air: what’s a tree got to do with it? www.ecology.com/2011/09/02/air-and-trees. Accessed April 4, 2017.
5. World Health Organization. Ambient (outdoor) air quality and health. www.who.int/mediacentre/factsheets/fs313/en. Ac-cessed April 4, 2017.
6. CDC. Asthma’s impact on the nation. www.cdc.gov/asthma/impacts_nation/default.htm. Accessed April 4, 2017.
7. CDC. Asthma. www.cdc.gov/asthma/default.htm. Accessed April 4, 2017.
8. Bui DS, Burgess JA, Lowe AJ, et al. Childhood lung function predicts adult COPD and asthma-COPD overlap syndrome (ACOS). Am J Respir Crit Care Med. 2017 Feb 1. [Epub ahead of print]
9. Doheny K. Living with severe allergies. www.webmd.com/allergies/features/severe-allergies#1. Accessed April 4, 2017.
10. Gromicko N, Tarasenko K. Plants and indoor air quality. www.nachi.org/plants-indoor-air-quality.htm. Accessed April 4, 2017.
11. Asthma and Allergy Foundation of America. Fight the cause of allergy. http://fightthecauseofallergy.org/page/fight. Accessed April 4, 2017.
1. Clean air month 2017. www.whathealth.com/awareness/event/cleanairmonth.html. Accessed April 4, 2017.
2. United States Environmental Protection Agency (EPA). Particulate matter (PM) pollution. www.epa.gov/pm-pollution. Accessed April 19, 2017.
3. United States Environmental Protection Agency (EPA). EPA strategic plan. www.epa.gov/planandbudget/strategicplan. Accessed April 4, 2017.
4. Downing A. Air: what’s a tree got to do with it? www.ecology.com/2011/09/02/air-and-trees. Accessed April 4, 2017.
5. World Health Organization. Ambient (outdoor) air quality and health. www.who.int/mediacentre/factsheets/fs313/en. Ac-cessed April 4, 2017.
6. CDC. Asthma’s impact on the nation. www.cdc.gov/asthma/impacts_nation/default.htm. Accessed April 4, 2017.
7. CDC. Asthma. www.cdc.gov/asthma/default.htm. Accessed April 4, 2017.
8. Bui DS, Burgess JA, Lowe AJ, et al. Childhood lung function predicts adult COPD and asthma-COPD overlap syndrome (ACOS). Am J Respir Crit Care Med. 2017 Feb 1. [Epub ahead of print]
9. Doheny K. Living with severe allergies. www.webmd.com/allergies/features/severe-allergies#1. Accessed April 4, 2017.
10. Gromicko N, Tarasenko K. Plants and indoor air quality. www.nachi.org/plants-indoor-air-quality.htm. Accessed April 4, 2017.
11. Asthma and Allergy Foundation of America. Fight the cause of allergy. http://fightthecauseofallergy.org/page/fight. Accessed April 4, 2017.
Safety of corticosteroids in pregnancy: Is it the drug or the disease?
Corticosteroids such as prednisone are relatively frequently administered in pregnancy for their immunosuppressive and anti-inflammatory effects. Treatment may be initiated on a short-term basis for acute conditions. Alternatively, treatment may be more or less ongoing for severe chronic diseases such as asthma or a variety of other autoimmune conditions when disease symptoms do not remit in pregnancy. However, the safety of corticosteroid use with respect to risk of specific birth defects, preterm delivery, and low birth weight has been the subject of debate over some time.
Concerns about the teratogenicity of corticosteroids were raised as early as the 1950s, based on animal studies suggesting an increased risk for oral clefts. The association between corticosteroids and oral clefts has also been observed in some human epidemiologic studies. However, results of these studies have been inconsistent.
Similar to the NBDPS findings, in a large Danish cohort study covering 832,636 live births from 1996 to 2008, exposure to any corticosteroids during the first trimester was not associated with an increased risk for cleft lip or cleft palate. Only those exposed to topical corticosteroids had a higher risk of cleft lip with or without cleft palate (odds ratio, 1.45; 95% CI, 1.03-2.05).3 Another, smaller Danish study covered primiparous births from 1999 to 2009 (n = 83,043). The unadjusted odds of oral clefts following exposure to any corticosteroids (inhaled or oral) in the first trimester was null (OR, 0.4; 95% CI, 0.1-2.8).4
Inconsistencies across these studies, as speculated by authors of the NBDPS analysis, may result from a lack of information on the dose of drug used by the mother, the indication for its use, or any measure of the severity of the underlying maternal disease for which the corticosteroids were prescribed. It is possible that maternal disease or disease activity in and of itself is a direct cause of oral clefts or that corticosteroids are linked to increased risk for clefts through co-occurring other exposures such as smoking, alcohol, or obesity. However, these questions have yet to be answered.
With respect to other birth outcomes, a few disease-specific studies have examined birth weight or intrauterine growth restriction following corticosteroid use. In general, study findings have been reassuring. Among Danish women with Crohn’s disease, corticosteroids were not associated with reduced birth weight after adjusting for gestational age and disease activity (adjusted risk ratio, 1.1; 95% CI, 0.2-5.7).5 In another study of pregnant women with rheumatoid arthritis, birth weight was not associated with prednisone use after adjustment for gestational age at delivery and sex of the newborn.6 In a third cohort study of pregnant women with systemic lupus erythematosus, there was no a significant elevation in odds of intrauterine growth restriction following prednisone use.7
Several disease-specific studies have also examined corticosteroid use and risk of preterm birth. From the Danish cohort of pregnant women with Crohn’s disease, the researchers reported no association between prednisolone and preterm birth after adjustment for covariates. In contrast, in a separate Danish cohort of pregnant women with irritable bowel disease, there was an increased risk of preterm delivery following systemic corticosteroid use, compared with women without disease (adjusted hazard ratio, 6.3; 95% CI, 3.1-12.7).8 However, data were not available to address underlying disease severity as a possible contributing factor. Of note, among women with irritable bowel disease who did not use medication in pregnancy, there was a 50% increase in the risk of preterm birth, compared with women without disease (aHR, 1.5; 95% CI, 1.0-2.4). This suggests that the disease itself contributed to the increased risk of preterm birth.
Currently available data regarding corticosteroid use and adverse birth outcomes are generally reassuring. Recent estimates for oral clefts suggest a low elevation in risk, if any at all. This translates to a very low absolute risk for clefts, which occur in the general population in approximately 1 in 1,000 births. The clinical benefit of adequate treatment in the first trimester for inflammatory or immune-mediated diseases may far outweigh any small and tenuous risks for oral clefts.
With respect to reduced birth weight and preterm delivery, available evidence suggests either no association or that maternal disease and disease severity are driving any increased risks noted for these outcomes. Future studies of pregnancy safety for medications used to treat maternal diseases that themselves are potentially linked to adverse outcomes must incorporate appropriate measures of disease type and disease severity in the study designs.
Dr. Chambers is a professor of pediatrics and director of clinical research at Rady Children’s Hospital and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is also director of MotherToBaby California, a past president of the Organization of Teratology Information Specialists, and past president of the Teratology Society. She has no relevant financial disclosures. To comment, e-mail her at [email protected].
References
1. Teratology. 2000 Dec;62(6):385-92.
2. Birth Defects Res A Clin Mol Teratol. 2014 Jun;100(6):499-506.
3. CMAJ. 2011 Apr 19;183(7):796-804.
4. Am J Ther. 2014 Mar-Apr;21(2):73-80.
5. Am J Gastroenterol. 2007 Jul;102(7):1406-13.
6. Arthritis Rheum. 2009 Nov;60(11):3196-206.
7. Lupus. 2010 Dec;19(14):1665-73.
8. PLoS One. 2015 Jun 17;10(6):e0129567.
Corticosteroids such as prednisone are relatively frequently administered in pregnancy for their immunosuppressive and anti-inflammatory effects. Treatment may be initiated on a short-term basis for acute conditions. Alternatively, treatment may be more or less ongoing for severe chronic diseases such as asthma or a variety of other autoimmune conditions when disease symptoms do not remit in pregnancy. However, the safety of corticosteroid use with respect to risk of specific birth defects, preterm delivery, and low birth weight has been the subject of debate over some time.
Concerns about the teratogenicity of corticosteroids were raised as early as the 1950s, based on animal studies suggesting an increased risk for oral clefts. The association between corticosteroids and oral clefts has also been observed in some human epidemiologic studies. However, results of these studies have been inconsistent.
Similar to the NBDPS findings, in a large Danish cohort study covering 832,636 live births from 1996 to 2008, exposure to any corticosteroids during the first trimester was not associated with an increased risk for cleft lip or cleft palate. Only those exposed to topical corticosteroids had a higher risk of cleft lip with or without cleft palate (odds ratio, 1.45; 95% CI, 1.03-2.05).3 Another, smaller Danish study covered primiparous births from 1999 to 2009 (n = 83,043). The unadjusted odds of oral clefts following exposure to any corticosteroids (inhaled or oral) in the first trimester was null (OR, 0.4; 95% CI, 0.1-2.8).4
Inconsistencies across these studies, as speculated by authors of the NBDPS analysis, may result from a lack of information on the dose of drug used by the mother, the indication for its use, or any measure of the severity of the underlying maternal disease for which the corticosteroids were prescribed. It is possible that maternal disease or disease activity in and of itself is a direct cause of oral clefts or that corticosteroids are linked to increased risk for clefts through co-occurring other exposures such as smoking, alcohol, or obesity. However, these questions have yet to be answered.
With respect to other birth outcomes, a few disease-specific studies have examined birth weight or intrauterine growth restriction following corticosteroid use. In general, study findings have been reassuring. Among Danish women with Crohn’s disease, corticosteroids were not associated with reduced birth weight after adjusting for gestational age and disease activity (adjusted risk ratio, 1.1; 95% CI, 0.2-5.7).5 In another study of pregnant women with rheumatoid arthritis, birth weight was not associated with prednisone use after adjustment for gestational age at delivery and sex of the newborn.6 In a third cohort study of pregnant women with systemic lupus erythematosus, there was no a significant elevation in odds of intrauterine growth restriction following prednisone use.7
Several disease-specific studies have also examined corticosteroid use and risk of preterm birth. From the Danish cohort of pregnant women with Crohn’s disease, the researchers reported no association between prednisolone and preterm birth after adjustment for covariates. In contrast, in a separate Danish cohort of pregnant women with irritable bowel disease, there was an increased risk of preterm delivery following systemic corticosteroid use, compared with women without disease (adjusted hazard ratio, 6.3; 95% CI, 3.1-12.7).8 However, data were not available to address underlying disease severity as a possible contributing factor. Of note, among women with irritable bowel disease who did not use medication in pregnancy, there was a 50% increase in the risk of preterm birth, compared with women without disease (aHR, 1.5; 95% CI, 1.0-2.4). This suggests that the disease itself contributed to the increased risk of preterm birth.
Currently available data regarding corticosteroid use and adverse birth outcomes are generally reassuring. Recent estimates for oral clefts suggest a low elevation in risk, if any at all. This translates to a very low absolute risk for clefts, which occur in the general population in approximately 1 in 1,000 births. The clinical benefit of adequate treatment in the first trimester for inflammatory or immune-mediated diseases may far outweigh any small and tenuous risks for oral clefts.
With respect to reduced birth weight and preterm delivery, available evidence suggests either no association or that maternal disease and disease severity are driving any increased risks noted for these outcomes. Future studies of pregnancy safety for medications used to treat maternal diseases that themselves are potentially linked to adverse outcomes must incorporate appropriate measures of disease type and disease severity in the study designs.
Dr. Chambers is a professor of pediatrics and director of clinical research at Rady Children’s Hospital and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is also director of MotherToBaby California, a past president of the Organization of Teratology Information Specialists, and past president of the Teratology Society. She has no relevant financial disclosures. To comment, e-mail her at [email protected].
References
1. Teratology. 2000 Dec;62(6):385-92.
2. Birth Defects Res A Clin Mol Teratol. 2014 Jun;100(6):499-506.
3. CMAJ. 2011 Apr 19;183(7):796-804.
4. Am J Ther. 2014 Mar-Apr;21(2):73-80.
5. Am J Gastroenterol. 2007 Jul;102(7):1406-13.
6. Arthritis Rheum. 2009 Nov;60(11):3196-206.
7. Lupus. 2010 Dec;19(14):1665-73.
8. PLoS One. 2015 Jun 17;10(6):e0129567.
Corticosteroids such as prednisone are relatively frequently administered in pregnancy for their immunosuppressive and anti-inflammatory effects. Treatment may be initiated on a short-term basis for acute conditions. Alternatively, treatment may be more or less ongoing for severe chronic diseases such as asthma or a variety of other autoimmune conditions when disease symptoms do not remit in pregnancy. However, the safety of corticosteroid use with respect to risk of specific birth defects, preterm delivery, and low birth weight has been the subject of debate over some time.
Concerns about the teratogenicity of corticosteroids were raised as early as the 1950s, based on animal studies suggesting an increased risk for oral clefts. The association between corticosteroids and oral clefts has also been observed in some human epidemiologic studies. However, results of these studies have been inconsistent.
Similar to the NBDPS findings, in a large Danish cohort study covering 832,636 live births from 1996 to 2008, exposure to any corticosteroids during the first trimester was not associated with an increased risk for cleft lip or cleft palate. Only those exposed to topical corticosteroids had a higher risk of cleft lip with or without cleft palate (odds ratio, 1.45; 95% CI, 1.03-2.05).3 Another, smaller Danish study covered primiparous births from 1999 to 2009 (n = 83,043). The unadjusted odds of oral clefts following exposure to any corticosteroids (inhaled or oral) in the first trimester was null (OR, 0.4; 95% CI, 0.1-2.8).4
Inconsistencies across these studies, as speculated by authors of the NBDPS analysis, may result from a lack of information on the dose of drug used by the mother, the indication for its use, or any measure of the severity of the underlying maternal disease for which the corticosteroids were prescribed. It is possible that maternal disease or disease activity in and of itself is a direct cause of oral clefts or that corticosteroids are linked to increased risk for clefts through co-occurring other exposures such as smoking, alcohol, or obesity. However, these questions have yet to be answered.
With respect to other birth outcomes, a few disease-specific studies have examined birth weight or intrauterine growth restriction following corticosteroid use. In general, study findings have been reassuring. Among Danish women with Crohn’s disease, corticosteroids were not associated with reduced birth weight after adjusting for gestational age and disease activity (adjusted risk ratio, 1.1; 95% CI, 0.2-5.7).5 In another study of pregnant women with rheumatoid arthritis, birth weight was not associated with prednisone use after adjustment for gestational age at delivery and sex of the newborn.6 In a third cohort study of pregnant women with systemic lupus erythematosus, there was no a significant elevation in odds of intrauterine growth restriction following prednisone use.7
Several disease-specific studies have also examined corticosteroid use and risk of preterm birth. From the Danish cohort of pregnant women with Crohn’s disease, the researchers reported no association between prednisolone and preterm birth after adjustment for covariates. In contrast, in a separate Danish cohort of pregnant women with irritable bowel disease, there was an increased risk of preterm delivery following systemic corticosteroid use, compared with women without disease (adjusted hazard ratio, 6.3; 95% CI, 3.1-12.7).8 However, data were not available to address underlying disease severity as a possible contributing factor. Of note, among women with irritable bowel disease who did not use medication in pregnancy, there was a 50% increase in the risk of preterm birth, compared with women without disease (aHR, 1.5; 95% CI, 1.0-2.4). This suggests that the disease itself contributed to the increased risk of preterm birth.
Currently available data regarding corticosteroid use and adverse birth outcomes are generally reassuring. Recent estimates for oral clefts suggest a low elevation in risk, if any at all. This translates to a very low absolute risk for clefts, which occur in the general population in approximately 1 in 1,000 births. The clinical benefit of adequate treatment in the first trimester for inflammatory or immune-mediated diseases may far outweigh any small and tenuous risks for oral clefts.
With respect to reduced birth weight and preterm delivery, available evidence suggests either no association or that maternal disease and disease severity are driving any increased risks noted for these outcomes. Future studies of pregnancy safety for medications used to treat maternal diseases that themselves are potentially linked to adverse outcomes must incorporate appropriate measures of disease type and disease severity in the study designs.
Dr. Chambers is a professor of pediatrics and director of clinical research at Rady Children’s Hospital and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is also director of MotherToBaby California, a past president of the Organization of Teratology Information Specialists, and past president of the Teratology Society. She has no relevant financial disclosures. To comment, e-mail her at [email protected].
References
1. Teratology. 2000 Dec;62(6):385-92.
2. Birth Defects Res A Clin Mol Teratol. 2014 Jun;100(6):499-506.
3. CMAJ. 2011 Apr 19;183(7):796-804.
4. Am J Ther. 2014 Mar-Apr;21(2):73-80.
5. Am J Gastroenterol. 2007 Jul;102(7):1406-13.
6. Arthritis Rheum. 2009 Nov;60(11):3196-206.
7. Lupus. 2010 Dec;19(14):1665-73.
8. PLoS One. 2015 Jun 17;10(6):e0129567.
Optimizing surveillance for gynecologic cancers
Gynecologic cancers contribute to approximately 15% of cancer survivorship care for women. Many patients share surveillance visits between their gynecologic oncologist and their ob.gyn. or primary care physician to capitalize on preexisting relationships and ensure the provision of comprehensive wellness care. Providing high quality surveillance care is challenging because it requires vigilance in the detection of recurrence but also avoidance of unnecessary, costly, and inaccurate testing.
The oncologic benefits for various surveillance guidelines are not well established by prospective studies. However, in updated surveillance recommendations, the Society of Gynecologic Oncology (SGO) takes available data, costs, and benefits into consideration.1 The guidelines, authored by Ritu Salani, MD, provide an excellent resource for understanding appropriate testing and evaluation during surveillance care.
The cornerstone of a surveillance visit is a thorough symptom assessment. Positive reporting of symptoms remains the most sensitive method for detecting recurrences; therefore, patients should be educated and quizzed on common recurrence symptoms. For example, endometrial cancer most commonly recurs in the vagina with symptoms of vaginal bleeding or discharge. Lower extremity swelling can signify pelvic sidewall recurrences and abdominal bloating or pain can signify peritoneal recurrence of ovarian or endometrial cancer.
All women who are undergoing surveillance for gynecologic cancers should receive physical examinations that include a pelvic exam with a speculum and bimanual exam with rectovaginal exam. Many locoregional recurrences are salvageable for most gynecologic cancers, which is not true for most distant recurrences, emphasizing the importance of pelvic examinations.
In addition to surveillance of recurrence, these visits should focus on risk modification – tobacco, obesity, bone demineralization – as well as preventive health strategies, such as vaccinations, nongynecologic cancer screenings, and cardiovascular disease intervention. Clinicians should also ask about sequelae to cancer therapy, such as neuropathy, lymphedema, sexual dysfunction, depression, and fatigue.
Endometrial cancer
Endometrial cancer recurs most commonly among women with a history of advanced stage cancer or early stage disease with high/intermediate risk factors, but all survivors should be evaluated regularly for recurrence. The vagina is the most common site for recurrence. Fortunately, many vaginal recurrences can be cured with salvage therapies.
Women with the lowest risk for recurrence (stage IA, grades 1 and 2 disease) who did not originally qualify for adjuvant therapy can be followed every 6 months for 2 years and then annually.
Vaginal cytology is no longer recommended for the routine surveillance of endometrial cancer because of its poor sensitivity in detecting recurrence and low positive predictive value (particularly after vaginal radiation).2 Any suspicious lesions identified on speculum examination should be biopsied, rather than sampled with cytologic smear. Routine imaging (with CT or PET/CT) and cancer antigen (CA) 125 tumor marker assessment is not supported unless the initial stage was advanced. These tests should be reserved for confirmation of concerning symptoms or examination findings.
This group of patients has particular survivorship needs with respect to obesity interventions. Obesity is associated with poor prognosis from endometrial cancer, and patients should be counseled about this and offered strategies for weight loss and lifestyle modification. Lynch syndrome testing and colon cancer screening are also an important consideration in this population.
Ovarian cancer
Ovarian cancer recurrence rates are high, and, while salvage therapies are rarely curative, enduring responses may be achieved in some patients, making surveillance visits critical. The SGO recommends surveillance visits every 3 months in the first 2 years, every 4 months in year 3, and then every 6 months for an additional 2 years. At these visits, patients should be queried about symptoms with particular emphasis on peritoneal signs (bloating, distension, gastrointestinal disturbance, and abdominal pain) as most recurrences will be within the peritoneal cavity.
CA 125 tumor marker elevation during the surveillance phase may signal recurrence prior to the development of symptoms but initiating chemotherapy early because of elevations in CA 125 does not improve survival.3 However, in the platinum-sensitive population with a longer disease-free interval, earlier detection of recurrence by CA 125 may identify patients in whom complete secondary cytoreduction is more attainable and is associated with improved survival.4 Therefore, the SGO suggests that CA 125 assessment is optional. The benefits and limitations of earlier detection of recurrence should be discussed with each patient. This recommendation differs for survivors of nonepithelial ovarian cancers (such as germ-cell or sex-cord stromal), in which case the measurement of the appropriate tumor markers (such as alpha-fetoprotein, human chorionic gonadotropin, and inhibin) should be performed routinely as part of surveillance evaluation.
Evidence does not support routine imaging (such as CT or PET). It should be reserved as a confirmatory measure for patients with concerning symptoms, examination findings, or elevations in tumor markers. When ovarian cancer has been treated with fertility-preserving surgery in women of younger reproductive ages, pelvic ultrasounds may be used as part of their surveillance care to monitor retained ovaries and pelvic structures.
BRCA-gene testing should be offered to all women with epithelial ovarian, fallopian tube, and primary peritoneal cancer as it impacts future cancer risk, as well as chemotherapy selection.5
Cervical cancer
In the first 2 years after completing primary treatment for cervical cancer, those at high risk for recurrence (including those who were recommended to adjuvant therapy) should be evaluated every 3 months for 2 years, followed by visits at 6-month intervals for an additional 3 years. Low-risk patients can be followed every 6 months for 2 years, followed by annual visits thereafter.
Pap testing should be performed annually, rather than at each surveillance visit. It should not to detect recurrence – for which it has poor sensitivity and specificity – but rather to detect new HPV-related dysplasia.6
Many patients with cervical cancer have a tobacco use history, placing them at risk for other cancers. Educate patients about the risk and provide cessation assistance.
Vulvar cancer
Prognosis for early stage vulvar cancer is very good; however, local recurrences are common (as much as 40%) in the 10 years following diagnosis.7 It is important to thoroughly inspect the vulva, vagina, and cervix at each surveillance visit. In high-risk patients, examinations should take place every 3 months for the first 2 years after completing primary treatment and every 6 months until 5 years post treatment. Low-risk patients can be followed every 6 months for 2 years and annually thereafter.
Identification and early treatment of dysplasia is important. Careful attention should also be made to palpation of the inguinal nodal regions. One in 10 women will have a late recurrence (greater than 5 years), so vulvar inspections should continue at least annually for the remainder of a woman’s life.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She reported having no relevant financial disclosures.
References
1. Gynecol Oncol. 2017 Mar 31. doi: 10.1016/j.ygno.2017.03.022.
2. Obstet Gynecol. 2013 Jan;121(1):129-35.
3. Lancet. 2010 Oct 2;376(9747):1155-63.
4. Gynecol Oncol. 2009 Jan;112(1):265-74.
5. Gynecol Oncol. 2015 Jan;136(1):3-7.
6. Obstet Gynecol. 2011 Sep;118(3):548-53.
7. Gynecol Oncol. 2016 Jan;140(1):8-14.
Gynecologic cancers contribute to approximately 15% of cancer survivorship care for women. Many patients share surveillance visits between their gynecologic oncologist and their ob.gyn. or primary care physician to capitalize on preexisting relationships and ensure the provision of comprehensive wellness care. Providing high quality surveillance care is challenging because it requires vigilance in the detection of recurrence but also avoidance of unnecessary, costly, and inaccurate testing.
The oncologic benefits for various surveillance guidelines are not well established by prospective studies. However, in updated surveillance recommendations, the Society of Gynecologic Oncology (SGO) takes available data, costs, and benefits into consideration.1 The guidelines, authored by Ritu Salani, MD, provide an excellent resource for understanding appropriate testing and evaluation during surveillance care.
The cornerstone of a surveillance visit is a thorough symptom assessment. Positive reporting of symptoms remains the most sensitive method for detecting recurrences; therefore, patients should be educated and quizzed on common recurrence symptoms. For example, endometrial cancer most commonly recurs in the vagina with symptoms of vaginal bleeding or discharge. Lower extremity swelling can signify pelvic sidewall recurrences and abdominal bloating or pain can signify peritoneal recurrence of ovarian or endometrial cancer.
All women who are undergoing surveillance for gynecologic cancers should receive physical examinations that include a pelvic exam with a speculum and bimanual exam with rectovaginal exam. Many locoregional recurrences are salvageable for most gynecologic cancers, which is not true for most distant recurrences, emphasizing the importance of pelvic examinations.
In addition to surveillance of recurrence, these visits should focus on risk modification – tobacco, obesity, bone demineralization – as well as preventive health strategies, such as vaccinations, nongynecologic cancer screenings, and cardiovascular disease intervention. Clinicians should also ask about sequelae to cancer therapy, such as neuropathy, lymphedema, sexual dysfunction, depression, and fatigue.

Endometrial cancer
Endometrial cancer recurs most commonly among women with a history of advanced stage cancer or early stage disease with high/intermediate risk factors, but all survivors should be evaluated regularly for recurrence. The vagina is the most common site for recurrence. Fortunately, many vaginal recurrences can be cured with salvage therapies.
Women with the lowest risk for recurrence (stage IA, grades 1 and 2 disease) who did not originally qualify for adjuvant therapy can be followed every 6 months for 2 years and then annually.
Vaginal cytology is no longer recommended for the routine surveillance of endometrial cancer because of its poor sensitivity in detecting recurrence and low positive predictive value (particularly after vaginal radiation).2 Any suspicious lesions identified on speculum examination should be biopsied, rather than sampled with cytologic smear. Routine imaging (with CT or PET/CT) and cancer antigen (CA) 125 tumor marker assessment is not supported unless the initial stage was advanced. These tests should be reserved for confirmation of concerning symptoms or examination findings.
This group of patients has particular survivorship needs with respect to obesity interventions. Obesity is associated with poor prognosis from endometrial cancer, and patients should be counseled about this and offered strategies for weight loss and lifestyle modification. Lynch syndrome testing and colon cancer screening are also an important consideration in this population.
Ovarian cancer
Ovarian cancer recurrence rates are high, and, while salvage therapies are rarely curative, enduring responses may be achieved in some patients, making surveillance visits critical. The SGO recommends surveillance visits every 3 months in the first 2 years, every 4 months in year 3, and then every 6 months for an additional 2 years. At these visits, patients should be queried about symptoms with particular emphasis on peritoneal signs (bloating, distension, gastrointestinal disturbance, and abdominal pain) as most recurrences will be within the peritoneal cavity.
CA 125 tumor marker elevation during the surveillance phase may signal recurrence prior to the development of symptoms but initiating chemotherapy early because of elevations in CA 125 does not improve survival.3 However, in the platinum-sensitive population with a longer disease-free interval, earlier detection of recurrence by CA 125 may identify patients in whom complete secondary cytoreduction is more attainable and is associated with improved survival.4 Therefore, the SGO suggests that CA 125 assessment is optional. The benefits and limitations of earlier detection of recurrence should be discussed with each patient. This recommendation differs for survivors of nonepithelial ovarian cancers (such as germ-cell or sex-cord stromal), in which case the measurement of the appropriate tumor markers (such as alpha-fetoprotein, human chorionic gonadotropin, and inhibin) should be performed routinely as part of surveillance evaluation.
Evidence does not support routine imaging (such as CT or PET). It should be reserved as a confirmatory measure for patients with concerning symptoms, examination findings, or elevations in tumor markers. When ovarian cancer has been treated with fertility-preserving surgery in women of younger reproductive ages, pelvic ultrasounds may be used as part of their surveillance care to monitor retained ovaries and pelvic structures.
BRCA-gene testing should be offered to all women with epithelial ovarian, fallopian tube, and primary peritoneal cancer as it impacts future cancer risk, as well as chemotherapy selection.5
Cervical cancer
In the first 2 years after completing primary treatment for cervical cancer, those at high risk for recurrence (including those who were recommended to adjuvant therapy) should be evaluated every 3 months for 2 years, followed by visits at 6-month intervals for an additional 3 years. Low-risk patients can be followed every 6 months for 2 years, followed by annual visits thereafter.
Pap testing should be performed annually, rather than at each surveillance visit. It should not to detect recurrence – for which it has poor sensitivity and specificity – but rather to detect new HPV-related dysplasia.6
Many patients with cervical cancer have a tobacco use history, placing them at risk for other cancers. Educate patients about the risk and provide cessation assistance.
Vulvar cancer
Prognosis for early stage vulvar cancer is very good; however, local recurrences are common (as much as 40%) in the 10 years following diagnosis.7 It is important to thoroughly inspect the vulva, vagina, and cervix at each surveillance visit. In high-risk patients, examinations should take place every 3 months for the first 2 years after completing primary treatment and every 6 months until 5 years post treatment. Low-risk patients can be followed every 6 months for 2 years and annually thereafter.
Identification and early treatment of dysplasia is important. Careful attention should also be made to palpation of the inguinal nodal regions. One in 10 women will have a late recurrence (greater than 5 years), so vulvar inspections should continue at least annually for the remainder of a woman’s life.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She reported having no relevant financial disclosures.
References
1. Gynecol Oncol. 2017 Mar 31. doi: 10.1016/j.ygno.2017.03.022.
2. Obstet Gynecol. 2013 Jan;121(1):129-35.
3. Lancet. 2010 Oct 2;376(9747):1155-63.
4. Gynecol Oncol. 2009 Jan;112(1):265-74.
5. Gynecol Oncol. 2015 Jan;136(1):3-7.
6. Obstet Gynecol. 2011 Sep;118(3):548-53.
7. Gynecol Oncol. 2016 Jan;140(1):8-14.
Gynecologic cancers contribute to approximately 15% of cancer survivorship care for women. Many patients share surveillance visits between their gynecologic oncologist and their ob.gyn. or primary care physician to capitalize on preexisting relationships and ensure the provision of comprehensive wellness care. Providing high quality surveillance care is challenging because it requires vigilance in the detection of recurrence but also avoidance of unnecessary, costly, and inaccurate testing.
The oncologic benefits for various surveillance guidelines are not well established by prospective studies. However, in updated surveillance recommendations, the Society of Gynecologic Oncology (SGO) takes available data, costs, and benefits into consideration.1 The guidelines, authored by Ritu Salani, MD, provide an excellent resource for understanding appropriate testing and evaluation during surveillance care.
The cornerstone of a surveillance visit is a thorough symptom assessment. Positive reporting of symptoms remains the most sensitive method for detecting recurrences; therefore, patients should be educated and quizzed on common recurrence symptoms. For example, endometrial cancer most commonly recurs in the vagina with symptoms of vaginal bleeding or discharge. Lower extremity swelling can signify pelvic sidewall recurrences and abdominal bloating or pain can signify peritoneal recurrence of ovarian or endometrial cancer.
All women who are undergoing surveillance for gynecologic cancers should receive physical examinations that include a pelvic exam with a speculum and bimanual exam with rectovaginal exam. Many locoregional recurrences are salvageable for most gynecologic cancers, which is not true for most distant recurrences, emphasizing the importance of pelvic examinations.
In addition to surveillance of recurrence, these visits should focus on risk modification – tobacco, obesity, bone demineralization – as well as preventive health strategies, such as vaccinations, nongynecologic cancer screenings, and cardiovascular disease intervention. Clinicians should also ask about sequelae to cancer therapy, such as neuropathy, lymphedema, sexual dysfunction, depression, and fatigue.

Endometrial cancer
Endometrial cancer recurs most commonly among women with a history of advanced stage cancer or early stage disease with high/intermediate risk factors, but all survivors should be evaluated regularly for recurrence. The vagina is the most common site for recurrence. Fortunately, many vaginal recurrences can be cured with salvage therapies.
Women with the lowest risk for recurrence (stage IA, grades 1 and 2 disease) who did not originally qualify for adjuvant therapy can be followed every 6 months for 2 years and then annually.
Vaginal cytology is no longer recommended for the routine surveillance of endometrial cancer because of its poor sensitivity in detecting recurrence and low positive predictive value (particularly after vaginal radiation).2 Any suspicious lesions identified on speculum examination should be biopsied, rather than sampled with cytologic smear. Routine imaging (with CT or PET/CT) and cancer antigen (CA) 125 tumor marker assessment is not supported unless the initial stage was advanced. These tests should be reserved for confirmation of concerning symptoms or examination findings.
This group of patients has particular survivorship needs with respect to obesity interventions. Obesity is associated with poor prognosis from endometrial cancer, and patients should be counseled about this and offered strategies for weight loss and lifestyle modification. Lynch syndrome testing and colon cancer screening are also an important consideration in this population.
Ovarian cancer
Ovarian cancer recurrence rates are high, and, while salvage therapies are rarely curative, enduring responses may be achieved in some patients, making surveillance visits critical. The SGO recommends surveillance visits every 3 months in the first 2 years, every 4 months in year 3, and then every 6 months for an additional 2 years. At these visits, patients should be queried about symptoms with particular emphasis on peritoneal signs (bloating, distension, gastrointestinal disturbance, and abdominal pain) as most recurrences will be within the peritoneal cavity.
CA 125 tumor marker elevation during the surveillance phase may signal recurrence prior to the development of symptoms but initiating chemotherapy early because of elevations in CA 125 does not improve survival.3 However, in the platinum-sensitive population with a longer disease-free interval, earlier detection of recurrence by CA 125 may identify patients in whom complete secondary cytoreduction is more attainable and is associated with improved survival.4 Therefore, the SGO suggests that CA 125 assessment is optional. The benefits and limitations of earlier detection of recurrence should be discussed with each patient. This recommendation differs for survivors of nonepithelial ovarian cancers (such as germ-cell or sex-cord stromal), in which case the measurement of the appropriate tumor markers (such as alpha-fetoprotein, human chorionic gonadotropin, and inhibin) should be performed routinely as part of surveillance evaluation.
Evidence does not support routine imaging (such as CT or PET). It should be reserved as a confirmatory measure for patients with concerning symptoms, examination findings, or elevations in tumor markers. When ovarian cancer has been treated with fertility-preserving surgery in women of younger reproductive ages, pelvic ultrasounds may be used as part of their surveillance care to monitor retained ovaries and pelvic structures.
BRCA-gene testing should be offered to all women with epithelial ovarian, fallopian tube, and primary peritoneal cancer as it impacts future cancer risk, as well as chemotherapy selection.5
Cervical cancer
In the first 2 years after completing primary treatment for cervical cancer, those at high risk for recurrence (including those who were recommended to adjuvant therapy) should be evaluated every 3 months for 2 years, followed by visits at 6-month intervals for an additional 3 years. Low-risk patients can be followed every 6 months for 2 years, followed by annual visits thereafter.
Pap testing should be performed annually, rather than at each surveillance visit. It should not to detect recurrence – for which it has poor sensitivity and specificity – but rather to detect new HPV-related dysplasia.6
Many patients with cervical cancer have a tobacco use history, placing them at risk for other cancers. Educate patients about the risk and provide cessation assistance.
Vulvar cancer
Prognosis for early stage vulvar cancer is very good; however, local recurrences are common (as much as 40%) in the 10 years following diagnosis.7 It is important to thoroughly inspect the vulva, vagina, and cervix at each surveillance visit. In high-risk patients, examinations should take place every 3 months for the first 2 years after completing primary treatment and every 6 months until 5 years post treatment. Low-risk patients can be followed every 6 months for 2 years and annually thereafter.
Identification and early treatment of dysplasia is important. Careful attention should also be made to palpation of the inguinal nodal regions. One in 10 women will have a late recurrence (greater than 5 years), so vulvar inspections should continue at least annually for the remainder of a woman’s life.
Dr. Rossi is an assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She reported having no relevant financial disclosures.
References
1. Gynecol Oncol. 2017 Mar 31. doi: 10.1016/j.ygno.2017.03.022.
2. Obstet Gynecol. 2013 Jan;121(1):129-35.
3. Lancet. 2010 Oct 2;376(9747):1155-63.
4. Gynecol Oncol. 2009 Jan;112(1):265-74.
5. Gynecol Oncol. 2015 Jan;136(1):3-7.
6. Obstet Gynecol. 2011 Sep;118(3):548-53.
7. Gynecol Oncol. 2016 Jan;140(1):8-14.
When to consider external cephalic version
Editor’s Note: This is the fourth installment of a six-part series that will review key concepts and articles that ob.gyns. can use to prepare for the American Board of Obstetrics and Gynecology Maintenance of Certification examination. The series is adapted from Ob/Gyn Board Master (obgynboardmaster.com), an online board review course created by Erudyte Inc. The series will cover issues in reproductive endocrinology and infertility, maternal-fetal medicine, gynecologic oncology, and female pelvic medicine, as well as general test-taking and study tips.
The American College of Obstetricians and Gynecologists’ “Practice Bulletins” are important practice management guidelines for ob.gyn. clinicians. The Practice Bulletins are rich sources of material often tested on board exams. In February 2016, ACOG issued a revised Practice Bulletin (#161) on external cephalic version outlining clinical considerations and recommendations and providing an algorithm for patient management.1 We recommend you read this bulletin and review this topic carefully.
Let’s begin with a possible medical board question: According to the Practice Bulletin, which of the following is TRUE about external cephalic version (ECV)?
A. Success rate is lower in women with a previous cesarean delivery.
B. Placental location affects the success rate.
C. External cephalic version should be stopped after 15 minutes.
D. Women at 37 weeks’ gestation are preferred candidates.
E. Tocolysis decreases success rate.
The correct answer is D.
Women at 37 weeks’ gestation are the preferred candidates for an ECV because spontaneous version would likely have already occurred by this time and the risk of spontaneous reversion is lower. Answers A-C and E are incorrect statements.
Key points
Women at 37 weeks’ gestation are the preferred candidates for an ECV.
The overall pooled success rate for ECV is 58% with a 6% pooled complication rate.
The use of parenteral tocolysis has been associated with increased success rates of ECV, though there are not enough data to make a recommendation regarding use of regional anesthesia with the procedure.
Literature summary
ECV is a procedure designed to turn a fetus into vertex presentation by applying external pressure to a woman’s abdomen. Women at 37 weeks’ gestation are the preferred candidates. At this gestational age, spontaneous version is most likely to have occurred, and there is decreased risk of spontaneous reversion after the ECV. All patients who are near term and found to have a fetus in a nonvertex presentation should be offered an ECV as long as there are no contraindications to the procedure. ECV is not appropriate for women who have a contraindication to a vaginal delivery.
There are limited studies of ECV in women who undergo the procedure in early labor and in those who have had a previous uterine surgery. ECV success rates are not affected by a previous cesarean delivery, though the risks of uterine rupture are not clear. The procedure should be attempted only in settings where cesarean delivery services are immediately available.
The success rates of ECV have been reported to be anywhere from 16% to 100%, with an overall pooled success of 58% with a 6% pooled complication rate. Some studies have documented higher success rates with higher parity and a transverse or oblique fetal lie. However, placental location, maternal weight, and amniotic fluid volume have not been consistently found to be predictive of ECV success. The use of parenteral tocolysis has been associated with increased success rates of ECV, though there are not enough data to make a recommendation regarding use of regional anesthesia with the procedure.
ECV should be stopped in the face of a prolonged or significant fetal bradycardia or if the patient is experiencing intolerable levels of discomfort. However, there are no guidelines to recommend the total time limit of the procedure. After the ECV, there should be fetal heart rate monitoring for at least 30 minutes and anti-D immune globulin should be administered to those women who are Rh-negative if delivery is not anticipated in the next 72 hours.
Dr. Siddighi is editor-in-chief of the Ob/Gyn Board Master and director of female pelvic medicine and reconstructive surgery and director of grand rounds at Loma Linda University Health in California. Ob.Gyn. News and Ob/Gyn Board Master are owned by the same parent company, Frontline Medical Communications.
Reference
Editor’s Note: This is the fourth installment of a six-part series that will review key concepts and articles that ob.gyns. can use to prepare for the American Board of Obstetrics and Gynecology Maintenance of Certification examination. The series is adapted from Ob/Gyn Board Master (obgynboardmaster.com), an online board review course created by Erudyte Inc. The series will cover issues in reproductive endocrinology and infertility, maternal-fetal medicine, gynecologic oncology, and female pelvic medicine, as well as general test-taking and study tips.
The American College of Obstetricians and Gynecologists’ “Practice Bulletins” are important practice management guidelines for ob.gyn. clinicians. The Practice Bulletins are rich sources of material often tested on board exams. In February 2016, ACOG issued a revised Practice Bulletin (#161) on external cephalic version outlining clinical considerations and recommendations and providing an algorithm for patient management.1 We recommend you read this bulletin and review this topic carefully.
Let’s begin with a possible medical board question: According to the Practice Bulletin, which of the following is TRUE about external cephalic version (ECV)?
A. Success rate is lower in women with a previous cesarean delivery.
B. Placental location affects the success rate.
C. External cephalic version should be stopped after 15 minutes.
D. Women at 37 weeks’ gestation are preferred candidates.
E. Tocolysis decreases success rate.
The correct answer is D.
Women at 37 weeks’ gestation are the preferred candidates for an ECV because spontaneous version would likely have already occurred by this time and the risk of spontaneous reversion is lower. Answers A-C and E are incorrect statements.
Key points
Women at 37 weeks’ gestation are the preferred candidates for an ECV.
The overall pooled success rate for ECV is 58% with a 6% pooled complication rate.
The use of parenteral tocolysis has been associated with increased success rates of ECV, though there are not enough data to make a recommendation regarding use of regional anesthesia with the procedure.
Literature summary
ECV is a procedure designed to turn a fetus into vertex presentation by applying external pressure to a woman’s abdomen. Women at 37 weeks’ gestation are the preferred candidates. At this gestational age, spontaneous version is most likely to have occurred, and there is decreased risk of spontaneous reversion after the ECV. All patients who are near term and found to have a fetus in a nonvertex presentation should be offered an ECV as long as there are no contraindications to the procedure. ECV is not appropriate for women who have a contraindication to a vaginal delivery.
There are limited studies of ECV in women who undergo the procedure in early labor and in those who have had a previous uterine surgery. ECV success rates are not affected by a previous cesarean delivery, though the risks of uterine rupture are not clear. The procedure should be attempted only in settings where cesarean delivery services are immediately available.
The success rates of ECV have been reported to be anywhere from 16% to 100%, with an overall pooled success of 58% with a 6% pooled complication rate. Some studies have documented higher success rates with higher parity and a transverse or oblique fetal lie. However, placental location, maternal weight, and amniotic fluid volume have not been consistently found to be predictive of ECV success. The use of parenteral tocolysis has been associated with increased success rates of ECV, though there are not enough data to make a recommendation regarding use of regional anesthesia with the procedure.
ECV should be stopped in the face of a prolonged or significant fetal bradycardia or if the patient is experiencing intolerable levels of discomfort. However, there are no guidelines to recommend the total time limit of the procedure. After the ECV, there should be fetal heart rate monitoring for at least 30 minutes and anti-D immune globulin should be administered to those women who are Rh-negative if delivery is not anticipated in the next 72 hours.
Dr. Siddighi is editor-in-chief of the Ob/Gyn Board Master and director of female pelvic medicine and reconstructive surgery and director of grand rounds at Loma Linda University Health in California. Ob.Gyn. News and Ob/Gyn Board Master are owned by the same parent company, Frontline Medical Communications.
Reference
Editor’s Note: This is the fourth installment of a six-part series that will review key concepts and articles that ob.gyns. can use to prepare for the American Board of Obstetrics and Gynecology Maintenance of Certification examination. The series is adapted from Ob/Gyn Board Master (obgynboardmaster.com), an online board review course created by Erudyte Inc. The series will cover issues in reproductive endocrinology and infertility, maternal-fetal medicine, gynecologic oncology, and female pelvic medicine, as well as general test-taking and study tips.
The American College of Obstetricians and Gynecologists’ “Practice Bulletins” are important practice management guidelines for ob.gyn. clinicians. The Practice Bulletins are rich sources of material often tested on board exams. In February 2016, ACOG issued a revised Practice Bulletin (#161) on external cephalic version outlining clinical considerations and recommendations and providing an algorithm for patient management.1 We recommend you read this bulletin and review this topic carefully.
Let’s begin with a possible medical board question: According to the Practice Bulletin, which of the following is TRUE about external cephalic version (ECV)?
A. Success rate is lower in women with a previous cesarean delivery.
B. Placental location affects the success rate.
C. External cephalic version should be stopped after 15 minutes.
D. Women at 37 weeks’ gestation are preferred candidates.
E. Tocolysis decreases success rate.
The correct answer is D.
Women at 37 weeks’ gestation are the preferred candidates for an ECV because spontaneous version would likely have already occurred by this time and the risk of spontaneous reversion is lower. Answers A-C and E are incorrect statements.
Key points
Women at 37 weeks’ gestation are the preferred candidates for an ECV.
The overall pooled success rate for ECV is 58% with a 6% pooled complication rate.
The use of parenteral tocolysis has been associated with increased success rates of ECV, though there are not enough data to make a recommendation regarding use of regional anesthesia with the procedure.
Literature summary
ECV is a procedure designed to turn a fetus into vertex presentation by applying external pressure to a woman’s abdomen. Women at 37 weeks’ gestation are the preferred candidates. At this gestational age, spontaneous version is most likely to have occurred, and there is decreased risk of spontaneous reversion after the ECV. All patients who are near term and found to have a fetus in a nonvertex presentation should be offered an ECV as long as there are no contraindications to the procedure. ECV is not appropriate for women who have a contraindication to a vaginal delivery.
There are limited studies of ECV in women who undergo the procedure in early labor and in those who have had a previous uterine surgery. ECV success rates are not affected by a previous cesarean delivery, though the risks of uterine rupture are not clear. The procedure should be attempted only in settings where cesarean delivery services are immediately available.
The success rates of ECV have been reported to be anywhere from 16% to 100%, with an overall pooled success of 58% with a 6% pooled complication rate. Some studies have documented higher success rates with higher parity and a transverse or oblique fetal lie. However, placental location, maternal weight, and amniotic fluid volume have not been consistently found to be predictive of ECV success. The use of parenteral tocolysis has been associated with increased success rates of ECV, though there are not enough data to make a recommendation regarding use of regional anesthesia with the procedure.
ECV should be stopped in the face of a prolonged or significant fetal bradycardia or if the patient is experiencing intolerable levels of discomfort. However, there are no guidelines to recommend the total time limit of the procedure. After the ECV, there should be fetal heart rate monitoring for at least 30 minutes and anti-D immune globulin should be administered to those women who are Rh-negative if delivery is not anticipated in the next 72 hours.
Dr. Siddighi is editor-in-chief of the Ob/Gyn Board Master and director of female pelvic medicine and reconstructive surgery and director of grand rounds at Loma Linda University Health in California. Ob.Gyn. News and Ob/Gyn Board Master are owned by the same parent company, Frontline Medical Communications.
Reference
The End of a Season
Spring, it symbolizes a new beginning. The smell of fresh cut grass hangs in the air, and it’s my favorite time of the sports year. A new season has begun in baseball, and the NHL and NBA playoffs are underway. As a new season begins, two more draw to a close. In this fan’s opinion, there is nothing quite as exciting as playoff hockey, and this month AJO hopes to “Capital-ize” on that excitement by presenting the hockey issue.
In “The Ice Hockey Issue”, Popkin and colleagues present a review of upper extremity injuries in hockey, which will serve as a guide for sports medicine physicians covering hockey games. There’s even a segment covering dental and ocular injuries, in case you don’t have a dentist or ophthalmologist handy. While we typically no longer publish case reports, Degen and colleagues present a unique report detailing an unusual injury to a prominent NHL goaltender. AJO presents it to expand your diagnostic differential for neck injuries.
I had another reason in mind when I mentioned the end of a season in this month’s editorial. The new AJO has seen a lot of changes, and it is our Editorial Team’s goal to continuously improve the journal and to provide timely features that are directly relevant to your practice. We’ve updated our website, and we’ve added some features, such as QR codes and take-home points, to improve your reading experience. But our ability to further enhance the journal is limited in print, and our web statistics show that a large percentage of our readers view the articles on their smartphones.
As I’ve written before, these are challenging times for printed media. The digital age has arrived and technology has made traditional publications less appealing. Our younger readers now demand a portable, electronic, media-rich publication that provides information that directly benefits their practices. To provide this, we envision a digital journal that is immersed in a learning environment, with videos, technique guides, and supplementary materials just a click away.
A few months back, AJO tested the digital waters. Our trial met with a positive response, and so, it is with great excitement that we announce that beginning in 2018, AJO will be the first orthopedic journal to go “All Digital.”
To further our goal of creating material that directly impacts your practice, we will present each feature review article as a learning module. The articles will feature extensive photos and videos, PowerPoint presentations for download, test questions, and patient information sheets. We will publish authors’ preference cards and postoperative protocols.
We’re currently developing applications and tools to improve your interactive experience. In the coming months, look for announcements regarding new strategic partnerships and features that will become mainstays of our electronic environment.
I hope you share the excitement of a new beginning in the digital era. I know the transition will provide a greatly enhanced, valuable resource that will change the way we utilize journals in our practice.
Am J Orthop. 2017;46(3):122. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
Spring, it symbolizes a new beginning. The smell of fresh cut grass hangs in the air, and it’s my favorite time of the sports year. A new season has begun in baseball, and the NHL and NBA playoffs are underway. As a new season begins, two more draw to a close. In this fan’s opinion, there is nothing quite as exciting as playoff hockey, and this month AJO hopes to “Capital-ize” on that excitement by presenting the hockey issue.
In “The Ice Hockey Issue”, Popkin and colleagues present a review of upper extremity injuries in hockey, which will serve as a guide for sports medicine physicians covering hockey games. There’s even a segment covering dental and ocular injuries, in case you don’t have a dentist or ophthalmologist handy. While we typically no longer publish case reports, Degen and colleagues present a unique report detailing an unusual injury to a prominent NHL goaltender. AJO presents it to expand your diagnostic differential for neck injuries.
I had another reason in mind when I mentioned the end of a season in this month’s editorial. The new AJO has seen a lot of changes, and it is our Editorial Team’s goal to continuously improve the journal and to provide timely features that are directly relevant to your practice. We’ve updated our website, and we’ve added some features, such as QR codes and take-home points, to improve your reading experience. But our ability to further enhance the journal is limited in print, and our web statistics show that a large percentage of our readers view the articles on their smartphones.
As I’ve written before, these are challenging times for printed media. The digital age has arrived and technology has made traditional publications less appealing. Our younger readers now demand a portable, electronic, media-rich publication that provides information that directly benefits their practices. To provide this, we envision a digital journal that is immersed in a learning environment, with videos, technique guides, and supplementary materials just a click away.
A few months back, AJO tested the digital waters. Our trial met with a positive response, and so, it is with great excitement that we announce that beginning in 2018, AJO will be the first orthopedic journal to go “All Digital.”
To further our goal of creating material that directly impacts your practice, we will present each feature review article as a learning module. The articles will feature extensive photos and videos, PowerPoint presentations for download, test questions, and patient information sheets. We will publish authors’ preference cards and postoperative protocols.
We’re currently developing applications and tools to improve your interactive experience. In the coming months, look for announcements regarding new strategic partnerships and features that will become mainstays of our electronic environment.
I hope you share the excitement of a new beginning in the digital era. I know the transition will provide a greatly enhanced, valuable resource that will change the way we utilize journals in our practice.
Am J Orthop. 2017;46(3):122. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.
Spring, it symbolizes a new beginning. The smell of fresh cut grass hangs in the air, and it’s my favorite time of the sports year. A new season has begun in baseball, and the NHL and NBA playoffs are underway. As a new season begins, two more draw to a close. In this fan’s opinion, there is nothing quite as exciting as playoff hockey, and this month AJO hopes to “Capital-ize” on that excitement by presenting the hockey issue.
In “The Ice Hockey Issue”, Popkin and colleagues present a review of upper extremity injuries in hockey, which will serve as a guide for sports medicine physicians covering hockey games. There’s even a segment covering dental and ocular injuries, in case you don’t have a dentist or ophthalmologist handy. While we typically no longer publish case reports, Degen and colleagues present a unique report detailing an unusual injury to a prominent NHL goaltender. AJO presents it to expand your diagnostic differential for neck injuries.
I had another reason in mind when I mentioned the end of a season in this month’s editorial. The new AJO has seen a lot of changes, and it is our Editorial Team’s goal to continuously improve the journal and to provide timely features that are directly relevant to your practice. We’ve updated our website, and we’ve added some features, such as QR codes and take-home points, to improve your reading experience. But our ability to further enhance the journal is limited in print, and our web statistics show that a large percentage of our readers view the articles on their smartphones.
As I’ve written before, these are challenging times for printed media. The digital age has arrived and technology has made traditional publications less appealing. Our younger readers now demand a portable, electronic, media-rich publication that provides information that directly benefits their practices. To provide this, we envision a digital journal that is immersed in a learning environment, with videos, technique guides, and supplementary materials just a click away.
A few months back, AJO tested the digital waters. Our trial met with a positive response, and so, it is with great excitement that we announce that beginning in 2018, AJO will be the first orthopedic journal to go “All Digital.”
To further our goal of creating material that directly impacts your practice, we will present each feature review article as a learning module. The articles will feature extensive photos and videos, PowerPoint presentations for download, test questions, and patient information sheets. We will publish authors’ preference cards and postoperative protocols.
We’re currently developing applications and tools to improve your interactive experience. In the coming months, look for announcements regarding new strategic partnerships and features that will become mainstays of our electronic environment.
I hope you share the excitement of a new beginning in the digital era. I know the transition will provide a greatly enhanced, valuable resource that will change the way we utilize journals in our practice.
Am J Orthop. 2017;46(3):122. Copyright Frontline Medical Communications Inc. 2017. All rights reserved.