User login
Employed physicians: A survival guide
The strike by health care workers at Kaiser Permanente may not involve physicians (yet). But as more doctors in the United States are finding themselves working as salaried employees, physicians can – and probably will – become a powerful force for change in a health care system that has shown itself to be increasingly hostile to employee concerns over issues involving patient care, wages and benefits, safety, and well-being.
Salaried employment has its challenges. Physician-employees may have less autonomy and voice in decision-making that affects patients. They may splinter into fragmented work groups; feel isolated; and have different imperatives based on who they are, what they want, and where they work. They may feel more removed from their patients and struggle to build strong relationships, with their employers in the way.
Yet important opportunities exist for doctors when embracing their employee side. Examples of these interests include adequate compensation, wellness, job security, patient and worker safety, health care quality, reasonable workloads and schedules, and fair treatment by employers, including the need to exhibit a strong collective voice in organizational decision-making.
Some believe that physician-employees must be unionized to maximize their rights and power as employees. Many expect physician unionization to take hold more fully over time. Medical residents, the doctors of tomorrow, are already considering unionization in greater numbers. Some are also doing it in the same employment setting alongside other health professionals, such as nurses.
Having studied doctors and their employment situations for years, I am convinced that whether through unionization or another approach, physicians must also change how they think about control; train and learn alongside other health care workers who share similar interests; and elevate at an early career stage their knowledge of the business side of health care.
Adopt a more pragmatic definition of autonomy
Doctors must embrace an updated definition of autonomy – one that matches their status as highly paid labor.
When I have spoken to physicians in my research about what autonomy means to them, many seem unable to reconceptualize it from a vague and absolute form of their profession’s strategic control over their economic fates and technical skills toward an individualized control that is situation-specific, one centered on winning the daily fights about workplace bread-and-butter issues such as those mentioned above.
But a more pragmatic definition of autonomy could get doctors focused on influencing important issues of the patient-care day and enhance their negotiating power with employers. It would allow physicians to break out of what often seems a paralysis of inaction – waiting for employers, insurers, or the government to reinstate the profession’s idealized version of control by handing it back the keys to the health care system through major regulatory, structural, and reimbursement-related changes. This fantasy is unlikely to become reality.
Physician-employees I’ve talked to over the years understand their everyday challenges. But when it comes to engaging in localized and sustained action to overcome them, they often perform less well, leading to feelings of helplessness and burnout. Valuing tactical control over their jobs and work setting will yield smaller but more impactful wins as employees intent on making their everyday work lives better.
Train alongside other health care professionals
Physicians must accept that how they are trained no longer prepares them for the employee world into which most are dropped. For instance, unless doctors are trained collaboratively alongside other health care professionals – such as nurses – they are less likely to identify closely with these colleagues once in practice. There is strength in numbers, so this mutual identification empowers both groups of employees. Yet, medical education remains largely the same: training young medical students in isolation for the first couple of years, then placing them into clerkships and residencies where true interprofessional care opportunities remain stunted and secondary to the “physician as captain of the team” mantra.
Unfortunately, the “hidden curriculum” of medicine helps convince medical students and residents early in their careers that they are the unquestioned leaders in patient care settings. This hierarchy encourages some doctors to keep their psychological distance from other members of the health care team and to resist sharing power, concerns, or insights with less skilled health care workers. This socialization harms the ability of physicians to act in a unified fashion alongside these other workers. Having physicians learn and train alongside other health professionals yields positive benefits for collective advocacy, including a shared sense of purpose, positive views on collaboration with others in the health setting, and greater development of bonds with nonphysician coworkers.
Integrate business with medical training in real time
Medical students and residents generally lack exposure to the everyday business realities of the U.S. health care system. This gap hinders their ability to understand the employee world and push for the types of changes and work conditions that benefit all health care workers. Formal business and management training should be a required part of every U.S. medical school and residency curriculum from day one. If you see it at all in medical schools now, it is mostly by accident, or given separate treatment in the form of standalone MBA or MPH degrees that rarely integrate organically and in real time with actual medical training. Not every doctor needs an MBA or MPH degree. However, all of them require a stronger contextual understanding of how the medicine they wish to practice is shaped by the economic and fiscal circumstances surrounding it – circumstances they do not control.
This is another reason why young doctors are unhappy and burned out. They cannot push for specific changes or properly critique the pros and cons of how their work is structured because they have not been made aware, in real time as they learn clinical practice, how their jobs are shaped by realities such as insurance coverage and reimbursement, the fragmentation of the care delivery system, their employer’s financial health , and the socioeconomic circumstances of their patients. They aren’t given the methods and tools related to process and quality improvement, budgeting, negotiation, risk management, leadership, and talent management that might help them navigate these undermining forces. They also get little advance exposure in their training to important workplace “soft” skills in such areas as how to work in teams, networking, communication and listening, empathy, and problem-solving – all necessary foci for bringing them closer to other health care workers and advocating alongside them effectively with health care employers.
Now is the time for physicians to embrace their identity as employees. Doing so is in their own best interest as professionals. It will help others in the health care workforce as well as patients. Moreover, it provides a needed counterbalance to the powerful corporate ethos now ascendant in U.S. health care.
Timothy Hoff, PhD, is a professor of management and healthcare systems at Northeastern University, Boston, and an associate fellow at the University of Oxford, England. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The strike by health care workers at Kaiser Permanente may not involve physicians (yet). But as more doctors in the United States are finding themselves working as salaried employees, physicians can – and probably will – become a powerful force for change in a health care system that has shown itself to be increasingly hostile to employee concerns over issues involving patient care, wages and benefits, safety, and well-being.
Salaried employment has its challenges. Physician-employees may have less autonomy and voice in decision-making that affects patients. They may splinter into fragmented work groups; feel isolated; and have different imperatives based on who they are, what they want, and where they work. They may feel more removed from their patients and struggle to build strong relationships, with their employers in the way.
Yet important opportunities exist for doctors when embracing their employee side. Examples of these interests include adequate compensation, wellness, job security, patient and worker safety, health care quality, reasonable workloads and schedules, and fair treatment by employers, including the need to exhibit a strong collective voice in organizational decision-making.
Some believe that physician-employees must be unionized to maximize their rights and power as employees. Many expect physician unionization to take hold more fully over time. Medical residents, the doctors of tomorrow, are already considering unionization in greater numbers. Some are also doing it in the same employment setting alongside other health professionals, such as nurses.
Having studied doctors and their employment situations for years, I am convinced that whether through unionization or another approach, physicians must also change how they think about control; train and learn alongside other health care workers who share similar interests; and elevate at an early career stage their knowledge of the business side of health care.
Adopt a more pragmatic definition of autonomy
Doctors must embrace an updated definition of autonomy – one that matches their status as highly paid labor.
When I have spoken to physicians in my research about what autonomy means to them, many seem unable to reconceptualize it from a vague and absolute form of their profession’s strategic control over their economic fates and technical skills toward an individualized control that is situation-specific, one centered on winning the daily fights about workplace bread-and-butter issues such as those mentioned above.
But a more pragmatic definition of autonomy could get doctors focused on influencing important issues of the patient-care day and enhance their negotiating power with employers. It would allow physicians to break out of what often seems a paralysis of inaction – waiting for employers, insurers, or the government to reinstate the profession’s idealized version of control by handing it back the keys to the health care system through major regulatory, structural, and reimbursement-related changes. This fantasy is unlikely to become reality.
Physician-employees I’ve talked to over the years understand their everyday challenges. But when it comes to engaging in localized and sustained action to overcome them, they often perform less well, leading to feelings of helplessness and burnout. Valuing tactical control over their jobs and work setting will yield smaller but more impactful wins as employees intent on making their everyday work lives better.
Train alongside other health care professionals
Physicians must accept that how they are trained no longer prepares them for the employee world into which most are dropped. For instance, unless doctors are trained collaboratively alongside other health care professionals – such as nurses – they are less likely to identify closely with these colleagues once in practice. There is strength in numbers, so this mutual identification empowers both groups of employees. Yet, medical education remains largely the same: training young medical students in isolation for the first couple of years, then placing them into clerkships and residencies where true interprofessional care opportunities remain stunted and secondary to the “physician as captain of the team” mantra.
Unfortunately, the “hidden curriculum” of medicine helps convince medical students and residents early in their careers that they are the unquestioned leaders in patient care settings. This hierarchy encourages some doctors to keep their psychological distance from other members of the health care team and to resist sharing power, concerns, or insights with less skilled health care workers. This socialization harms the ability of physicians to act in a unified fashion alongside these other workers. Having physicians learn and train alongside other health professionals yields positive benefits for collective advocacy, including a shared sense of purpose, positive views on collaboration with others in the health setting, and greater development of bonds with nonphysician coworkers.
Integrate business with medical training in real time
Medical students and residents generally lack exposure to the everyday business realities of the U.S. health care system. This gap hinders their ability to understand the employee world and push for the types of changes and work conditions that benefit all health care workers. Formal business and management training should be a required part of every U.S. medical school and residency curriculum from day one. If you see it at all in medical schools now, it is mostly by accident, or given separate treatment in the form of standalone MBA or MPH degrees that rarely integrate organically and in real time with actual medical training. Not every doctor needs an MBA or MPH degree. However, all of them require a stronger contextual understanding of how the medicine they wish to practice is shaped by the economic and fiscal circumstances surrounding it – circumstances they do not control.
This is another reason why young doctors are unhappy and burned out. They cannot push for specific changes or properly critique the pros and cons of how their work is structured because they have not been made aware, in real time as they learn clinical practice, how their jobs are shaped by realities such as insurance coverage and reimbursement, the fragmentation of the care delivery system, their employer’s financial health , and the socioeconomic circumstances of their patients. They aren’t given the methods and tools related to process and quality improvement, budgeting, negotiation, risk management, leadership, and talent management that might help them navigate these undermining forces. They also get little advance exposure in their training to important workplace “soft” skills in such areas as how to work in teams, networking, communication and listening, empathy, and problem-solving – all necessary foci for bringing them closer to other health care workers and advocating alongside them effectively with health care employers.
Now is the time for physicians to embrace their identity as employees. Doing so is in their own best interest as professionals. It will help others in the health care workforce as well as patients. Moreover, it provides a needed counterbalance to the powerful corporate ethos now ascendant in U.S. health care.
Timothy Hoff, PhD, is a professor of management and healthcare systems at Northeastern University, Boston, and an associate fellow at the University of Oxford, England. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The strike by health care workers at Kaiser Permanente may not involve physicians (yet). But as more doctors in the United States are finding themselves working as salaried employees, physicians can – and probably will – become a powerful force for change in a health care system that has shown itself to be increasingly hostile to employee concerns over issues involving patient care, wages and benefits, safety, and well-being.
Salaried employment has its challenges. Physician-employees may have less autonomy and voice in decision-making that affects patients. They may splinter into fragmented work groups; feel isolated; and have different imperatives based on who they are, what they want, and where they work. They may feel more removed from their patients and struggle to build strong relationships, with their employers in the way.
Yet important opportunities exist for doctors when embracing their employee side. Examples of these interests include adequate compensation, wellness, job security, patient and worker safety, health care quality, reasonable workloads and schedules, and fair treatment by employers, including the need to exhibit a strong collective voice in organizational decision-making.
Some believe that physician-employees must be unionized to maximize their rights and power as employees. Many expect physician unionization to take hold more fully over time. Medical residents, the doctors of tomorrow, are already considering unionization in greater numbers. Some are also doing it in the same employment setting alongside other health professionals, such as nurses.
Having studied doctors and their employment situations for years, I am convinced that whether through unionization or another approach, physicians must also change how they think about control; train and learn alongside other health care workers who share similar interests; and elevate at an early career stage their knowledge of the business side of health care.
Adopt a more pragmatic definition of autonomy
Doctors must embrace an updated definition of autonomy – one that matches their status as highly paid labor.
When I have spoken to physicians in my research about what autonomy means to them, many seem unable to reconceptualize it from a vague and absolute form of their profession’s strategic control over their economic fates and technical skills toward an individualized control that is situation-specific, one centered on winning the daily fights about workplace bread-and-butter issues such as those mentioned above.
But a more pragmatic definition of autonomy could get doctors focused on influencing important issues of the patient-care day and enhance their negotiating power with employers. It would allow physicians to break out of what often seems a paralysis of inaction – waiting for employers, insurers, or the government to reinstate the profession’s idealized version of control by handing it back the keys to the health care system through major regulatory, structural, and reimbursement-related changes. This fantasy is unlikely to become reality.
Physician-employees I’ve talked to over the years understand their everyday challenges. But when it comes to engaging in localized and sustained action to overcome them, they often perform less well, leading to feelings of helplessness and burnout. Valuing tactical control over their jobs and work setting will yield smaller but more impactful wins as employees intent on making their everyday work lives better.
Train alongside other health care professionals
Physicians must accept that how they are trained no longer prepares them for the employee world into which most are dropped. For instance, unless doctors are trained collaboratively alongside other health care professionals – such as nurses – they are less likely to identify closely with these colleagues once in practice. There is strength in numbers, so this mutual identification empowers both groups of employees. Yet, medical education remains largely the same: training young medical students in isolation for the first couple of years, then placing them into clerkships and residencies where true interprofessional care opportunities remain stunted and secondary to the “physician as captain of the team” mantra.
Unfortunately, the “hidden curriculum” of medicine helps convince medical students and residents early in their careers that they are the unquestioned leaders in patient care settings. This hierarchy encourages some doctors to keep their psychological distance from other members of the health care team and to resist sharing power, concerns, or insights with less skilled health care workers. This socialization harms the ability of physicians to act in a unified fashion alongside these other workers. Having physicians learn and train alongside other health professionals yields positive benefits for collective advocacy, including a shared sense of purpose, positive views on collaboration with others in the health setting, and greater development of bonds with nonphysician coworkers.
Integrate business with medical training in real time
Medical students and residents generally lack exposure to the everyday business realities of the U.S. health care system. This gap hinders their ability to understand the employee world and push for the types of changes and work conditions that benefit all health care workers. Formal business and management training should be a required part of every U.S. medical school and residency curriculum from day one. If you see it at all in medical schools now, it is mostly by accident, or given separate treatment in the form of standalone MBA or MPH degrees that rarely integrate organically and in real time with actual medical training. Not every doctor needs an MBA or MPH degree. However, all of them require a stronger contextual understanding of how the medicine they wish to practice is shaped by the economic and fiscal circumstances surrounding it – circumstances they do not control.
This is another reason why young doctors are unhappy and burned out. They cannot push for specific changes or properly critique the pros and cons of how their work is structured because they have not been made aware, in real time as they learn clinical practice, how their jobs are shaped by realities such as insurance coverage and reimbursement, the fragmentation of the care delivery system, their employer’s financial health , and the socioeconomic circumstances of their patients. They aren’t given the methods and tools related to process and quality improvement, budgeting, negotiation, risk management, leadership, and talent management that might help them navigate these undermining forces. They also get little advance exposure in their training to important workplace “soft” skills in such areas as how to work in teams, networking, communication and listening, empathy, and problem-solving – all necessary foci for bringing them closer to other health care workers and advocating alongside them effectively with health care employers.
Now is the time for physicians to embrace their identity as employees. Doing so is in their own best interest as professionals. It will help others in the health care workforce as well as patients. Moreover, it provides a needed counterbalance to the powerful corporate ethos now ascendant in U.S. health care.
Timothy Hoff, PhD, is a professor of management and healthcare systems at Northeastern University, Boston, and an associate fellow at the University of Oxford, England. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Why legal pot makes this physician sick
Last year, my husband and I took a 16-day road trip from Kentucky through Massachusetts to Maine. On our first morning in Boston, we exited the Park Street Station en route to Boston Common, but instead of being greeted by the aroma of molasses, we were hit full-on with a pungent, repulsive odor. “That’s skunk weed,” my husband chuckled as we stepped right into the middle of the Boston Freedom Rally, a celebration of all things cannabis.
As we boarded a hop-on-hop-off bus, we learned that this was the one week of the year that the city skips testing tour bus drivers for tetrahydrocannabinol (THC), “because we all test positive,” the driver quipped. As our open-air bus circled the Common, a crowd of pot enthusiasts displayed signs in support of relaxed regulation for public consumption.
The 34-year-old Boston Freedom Rally is a sign that U.S. culture has transformed forever. Mary Jane is no friend of emergency physicians nor of staff on hospital wards and offices.
Toking boomers and millennials
Researchers at the University of California, San Diego, looked at cannabis-related emergency department visits from all acute-care hospitals in the state from 2005 to 2019 and found an 1,808% increase in patients aged 65 or older (that is not a typo) who were there for complications from cannabis use.
The lead author said in an interview that, “older patients taking marijuana or related products may have dizziness and falls, heart palpitations, panic attacks, confusion, anxiety or worsening of underlying lung diseases, such as asthma or [chronic obstructive pulmonary disease].”
A recent study from Canada suggests that commercialization has been associated with an increase in related hospitalizations, including cannabis-induced psychosis.
According to a National Study of Drug Use and Health, marijuana use in young adults reached an all-time high (pun intended) in 2021. Nearly 10% of eighth graders and 20% of 10th graders reported using marijuana this past year.
The full downside of any drug, legal or illegal, is largely unknown until it infiltrates the mainstream market, but these are the typical cases we see:
Let’s start with the demotivated high school honors student who dropped out of college to work at the local cinema. He stumbled and broke his clavicle outside a bar at 2 AM, but he wasn’t sure if he passed out, so a cardiology consult was requested to “rule out” arrhythmia associated with syncope. He related that his plan to become a railway conductor had been upended because he knew he would be drug tested and just couldn’t give up pot. After a normal cardiac exam, ECG, labs, a Holter, and an echocardiogram were also requested and normal at a significant cost.
Cannabinoid hyperemesis syndrome
One of my Midwest colleagues related her encounter with two middle-aged pot users with ventricular tachycardia (VT). These episodes coincided with potassium levels less than 3.0 mEq/L in the setting of repetitive vomiting. The QTc interval didn’t normalize despite a corrected potassium level in one patient. They were both informed that they should never smoke pot because vomiting would predictably drop their K+ levels again and prolong their QTc intervals. Then began “the circular argument,” as my friend described it. The patient claims, “I smoke pot to relieve my nausea,” to which she explains that “in many folks, pot use induces nausea.” Of course, the classic reply is, “Not me.” Predictably one of these stoners soon returned with more VT, more puking, and more hypokalemia. “Consider yourself ‘allergic’ to pot smoke,” my friend advised, but “was met with no meaningful hint of understanding or hope for transformative change,” she told me.
I’ve seen cannabinoid hyperemesis syndrome several times in the past few years. It occurs in daily to weekly pot users. Very rarely, it can cause cerebral edema, but it is also associated with seizures and dehydration that can lead to hypovolemic shock and kidney failure.
Heart and brain harm
Then there are the young patients who for various reasons have developed heart failure. Unfortunately, some are repetitively tox screen positive with varying trifectas of methamphetamine (meth), cocaine, and THC; opiates, meth, and THC; alcohol, meth, and THC; or heroin, meth, and THC. THC, the ever present and essential third leg of the stool of stupor. These unfortunate patients often need heart failure medications that they can’t afford or won’t take because illicit drug use is expensive and dulls their ability to prioritize their health. Some desperately need a heart transplant, but the necessary negative drug screen is a pipe dream.
And it’s not just the heart that is affected. There are data linking cannabis use to a higher risk for both ischemic and hemorrhagic stroke. A retrospective study published in Stroke, of more than 1,000 people diagnosed with an aneurysmal subarachnoid hemorrhage, found that more than half of the 46 who tested positive for THC at admission developed delayed cerebral ischemia (DCI), which increases the risk for disability or early death. This was after adjusting for several patient characteristics as well as recent exposure to other illicit substances; cocaine, meth, and tobacco use were not associated with DCI.
Natural my ...
I’m certain my anti-cannabis stance will strike a nerve with those who love their recreational THC and push for its legal sale; after all, “It’s perfectly natural.” But I counter with the fact that tornadoes, earthquakes, cyanide, and appendicitis are all natural but certainly not optimal. And what we are seeing in the vascular specialties is completely unnatural. We are treating a different mix of complications than before pot was readily accessible across several states.
Our most effective action is to educate our patients. We should encourage those who don’t currently smoke cannabis to never start and those who do to quit. People who require marijuana for improved quality of life for terminal care or true (not supposed) disorders that mainstream medicine fails should be approached with empathy and caution.
A good rule of thumb is to never breathe anything you can see. Never put anything in your body that comes off the street: Drug dealers who sell cannabis cut with fentanyl will be ecstatic to take someone’s money then merely keep scrolling when their obituary comes up.
Let’s try to reverse the rise of vascular complications, orthopedic injuries, and vomiting across America. We can start by encouraging our patients to avoid “skunk weed” and get back to the sweet smells of nature in our cities and parks.
Some details have been changed to protect the patients’ identities, but the essence of their diagnoses has been preserved.
Dr. Walton-Shirley is a retired clinical cardiologist from Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Last year, my husband and I took a 16-day road trip from Kentucky through Massachusetts to Maine. On our first morning in Boston, we exited the Park Street Station en route to Boston Common, but instead of being greeted by the aroma of molasses, we were hit full-on with a pungent, repulsive odor. “That’s skunk weed,” my husband chuckled as we stepped right into the middle of the Boston Freedom Rally, a celebration of all things cannabis.
As we boarded a hop-on-hop-off bus, we learned that this was the one week of the year that the city skips testing tour bus drivers for tetrahydrocannabinol (THC), “because we all test positive,” the driver quipped. As our open-air bus circled the Common, a crowd of pot enthusiasts displayed signs in support of relaxed regulation for public consumption.
The 34-year-old Boston Freedom Rally is a sign that U.S. culture has transformed forever. Mary Jane is no friend of emergency physicians nor of staff on hospital wards and offices.
Toking boomers and millennials
Researchers at the University of California, San Diego, looked at cannabis-related emergency department visits from all acute-care hospitals in the state from 2005 to 2019 and found an 1,808% increase in patients aged 65 or older (that is not a typo) who were there for complications from cannabis use.
The lead author said in an interview that, “older patients taking marijuana or related products may have dizziness and falls, heart palpitations, panic attacks, confusion, anxiety or worsening of underlying lung diseases, such as asthma or [chronic obstructive pulmonary disease].”
A recent study from Canada suggests that commercialization has been associated with an increase in related hospitalizations, including cannabis-induced psychosis.
According to a National Study of Drug Use and Health, marijuana use in young adults reached an all-time high (pun intended) in 2021. Nearly 10% of eighth graders and 20% of 10th graders reported using marijuana this past year.
The full downside of any drug, legal or illegal, is largely unknown until it infiltrates the mainstream market, but these are the typical cases we see:
Let’s start with the demotivated high school honors student who dropped out of college to work at the local cinema. He stumbled and broke his clavicle outside a bar at 2 AM, but he wasn’t sure if he passed out, so a cardiology consult was requested to “rule out” arrhythmia associated with syncope. He related that his plan to become a railway conductor had been upended because he knew he would be drug tested and just couldn’t give up pot. After a normal cardiac exam, ECG, labs, a Holter, and an echocardiogram were also requested and normal at a significant cost.
Cannabinoid hyperemesis syndrome
One of my Midwest colleagues related her encounter with two middle-aged pot users with ventricular tachycardia (VT). These episodes coincided with potassium levels less than 3.0 mEq/L in the setting of repetitive vomiting. The QTc interval didn’t normalize despite a corrected potassium level in one patient. They were both informed that they should never smoke pot because vomiting would predictably drop their K+ levels again and prolong their QTc intervals. Then began “the circular argument,” as my friend described it. The patient claims, “I smoke pot to relieve my nausea,” to which she explains that “in many folks, pot use induces nausea.” Of course, the classic reply is, “Not me.” Predictably one of these stoners soon returned with more VT, more puking, and more hypokalemia. “Consider yourself ‘allergic’ to pot smoke,” my friend advised, but “was met with no meaningful hint of understanding or hope for transformative change,” she told me.
I’ve seen cannabinoid hyperemesis syndrome several times in the past few years. It occurs in daily to weekly pot users. Very rarely, it can cause cerebral edema, but it is also associated with seizures and dehydration that can lead to hypovolemic shock and kidney failure.
Heart and brain harm
Then there are the young patients who for various reasons have developed heart failure. Unfortunately, some are repetitively tox screen positive with varying trifectas of methamphetamine (meth), cocaine, and THC; opiates, meth, and THC; alcohol, meth, and THC; or heroin, meth, and THC. THC, the ever present and essential third leg of the stool of stupor. These unfortunate patients often need heart failure medications that they can’t afford or won’t take because illicit drug use is expensive and dulls their ability to prioritize their health. Some desperately need a heart transplant, but the necessary negative drug screen is a pipe dream.
And it’s not just the heart that is affected. There are data linking cannabis use to a higher risk for both ischemic and hemorrhagic stroke. A retrospective study published in Stroke, of more than 1,000 people diagnosed with an aneurysmal subarachnoid hemorrhage, found that more than half of the 46 who tested positive for THC at admission developed delayed cerebral ischemia (DCI), which increases the risk for disability or early death. This was after adjusting for several patient characteristics as well as recent exposure to other illicit substances; cocaine, meth, and tobacco use were not associated with DCI.
Natural my ...
I’m certain my anti-cannabis stance will strike a nerve with those who love their recreational THC and push for its legal sale; after all, “It’s perfectly natural.” But I counter with the fact that tornadoes, earthquakes, cyanide, and appendicitis are all natural but certainly not optimal. And what we are seeing in the vascular specialties is completely unnatural. We are treating a different mix of complications than before pot was readily accessible across several states.
Our most effective action is to educate our patients. We should encourage those who don’t currently smoke cannabis to never start and those who do to quit. People who require marijuana for improved quality of life for terminal care or true (not supposed) disorders that mainstream medicine fails should be approached with empathy and caution.
A good rule of thumb is to never breathe anything you can see. Never put anything in your body that comes off the street: Drug dealers who sell cannabis cut with fentanyl will be ecstatic to take someone’s money then merely keep scrolling when their obituary comes up.
Let’s try to reverse the rise of vascular complications, orthopedic injuries, and vomiting across America. We can start by encouraging our patients to avoid “skunk weed” and get back to the sweet smells of nature in our cities and parks.
Some details have been changed to protect the patients’ identities, but the essence of their diagnoses has been preserved.
Dr. Walton-Shirley is a retired clinical cardiologist from Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Last year, my husband and I took a 16-day road trip from Kentucky through Massachusetts to Maine. On our first morning in Boston, we exited the Park Street Station en route to Boston Common, but instead of being greeted by the aroma of molasses, we were hit full-on with a pungent, repulsive odor. “That’s skunk weed,” my husband chuckled as we stepped right into the middle of the Boston Freedom Rally, a celebration of all things cannabis.
As we boarded a hop-on-hop-off bus, we learned that this was the one week of the year that the city skips testing tour bus drivers for tetrahydrocannabinol (THC), “because we all test positive,” the driver quipped. As our open-air bus circled the Common, a crowd of pot enthusiasts displayed signs in support of relaxed regulation for public consumption.
The 34-year-old Boston Freedom Rally is a sign that U.S. culture has transformed forever. Mary Jane is no friend of emergency physicians nor of staff on hospital wards and offices.
Toking boomers and millennials
Researchers at the University of California, San Diego, looked at cannabis-related emergency department visits from all acute-care hospitals in the state from 2005 to 2019 and found an 1,808% increase in patients aged 65 or older (that is not a typo) who were there for complications from cannabis use.
The lead author said in an interview that, “older patients taking marijuana or related products may have dizziness and falls, heart palpitations, panic attacks, confusion, anxiety or worsening of underlying lung diseases, such as asthma or [chronic obstructive pulmonary disease].”
A recent study from Canada suggests that commercialization has been associated with an increase in related hospitalizations, including cannabis-induced psychosis.
According to a National Study of Drug Use and Health, marijuana use in young adults reached an all-time high (pun intended) in 2021. Nearly 10% of eighth graders and 20% of 10th graders reported using marijuana this past year.
The full downside of any drug, legal or illegal, is largely unknown until it infiltrates the mainstream market, but these are the typical cases we see:
Let’s start with the demotivated high school honors student who dropped out of college to work at the local cinema. He stumbled and broke his clavicle outside a bar at 2 AM, but he wasn’t sure if he passed out, so a cardiology consult was requested to “rule out” arrhythmia associated with syncope. He related that his plan to become a railway conductor had been upended because he knew he would be drug tested and just couldn’t give up pot. After a normal cardiac exam, ECG, labs, a Holter, and an echocardiogram were also requested and normal at a significant cost.
Cannabinoid hyperemesis syndrome
One of my Midwest colleagues related her encounter with two middle-aged pot users with ventricular tachycardia (VT). These episodes coincided with potassium levels less than 3.0 mEq/L in the setting of repetitive vomiting. The QTc interval didn’t normalize despite a corrected potassium level in one patient. They were both informed that they should never smoke pot because vomiting would predictably drop their K+ levels again and prolong their QTc intervals. Then began “the circular argument,” as my friend described it. The patient claims, “I smoke pot to relieve my nausea,” to which she explains that “in many folks, pot use induces nausea.” Of course, the classic reply is, “Not me.” Predictably one of these stoners soon returned with more VT, more puking, and more hypokalemia. “Consider yourself ‘allergic’ to pot smoke,” my friend advised, but “was met with no meaningful hint of understanding or hope for transformative change,” she told me.
I’ve seen cannabinoid hyperemesis syndrome several times in the past few years. It occurs in daily to weekly pot users. Very rarely, it can cause cerebral edema, but it is also associated with seizures and dehydration that can lead to hypovolemic shock and kidney failure.
Heart and brain harm
Then there are the young patients who for various reasons have developed heart failure. Unfortunately, some are repetitively tox screen positive with varying trifectas of methamphetamine (meth), cocaine, and THC; opiates, meth, and THC; alcohol, meth, and THC; or heroin, meth, and THC. THC, the ever present and essential third leg of the stool of stupor. These unfortunate patients often need heart failure medications that they can’t afford or won’t take because illicit drug use is expensive and dulls their ability to prioritize their health. Some desperately need a heart transplant, but the necessary negative drug screen is a pipe dream.
And it’s not just the heart that is affected. There are data linking cannabis use to a higher risk for both ischemic and hemorrhagic stroke. A retrospective study published in Stroke, of more than 1,000 people diagnosed with an aneurysmal subarachnoid hemorrhage, found that more than half of the 46 who tested positive for THC at admission developed delayed cerebral ischemia (DCI), which increases the risk for disability or early death. This was after adjusting for several patient characteristics as well as recent exposure to other illicit substances; cocaine, meth, and tobacco use were not associated with DCI.
Natural my ...
I’m certain my anti-cannabis stance will strike a nerve with those who love their recreational THC and push for its legal sale; after all, “It’s perfectly natural.” But I counter with the fact that tornadoes, earthquakes, cyanide, and appendicitis are all natural but certainly not optimal. And what we are seeing in the vascular specialties is completely unnatural. We are treating a different mix of complications than before pot was readily accessible across several states.
Our most effective action is to educate our patients. We should encourage those who don’t currently smoke cannabis to never start and those who do to quit. People who require marijuana for improved quality of life for terminal care or true (not supposed) disorders that mainstream medicine fails should be approached with empathy and caution.
A good rule of thumb is to never breathe anything you can see. Never put anything in your body that comes off the street: Drug dealers who sell cannabis cut with fentanyl will be ecstatic to take someone’s money then merely keep scrolling when their obituary comes up.
Let’s try to reverse the rise of vascular complications, orthopedic injuries, and vomiting across America. We can start by encouraging our patients to avoid “skunk weed” and get back to the sweet smells of nature in our cities and parks.
Some details have been changed to protect the patients’ identities, but the essence of their diagnoses has been preserved.
Dr. Walton-Shirley is a retired clinical cardiologist from Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Addressing obesity bias in health care
Obesity is a major factor affecting the health of many Americans. It is estimated by the Centers for Disease Control and Prevention that 41% of adults and 19.7% of children in our country now meet the criteria for being obese. Obesity costs the United States approximately $147 billion annually in health care costs. While these numbers are staggering, they continue to rise.
The recent craze over medications such as Ozempic, Wegovy, and Mounjaro shows how eager people are to lose weight. Yet, many of them face bias, not just in their daily lives, but from health care professionals who should do better. No one should feel stigmatized when they come for medical help. This just drives away patients who need us and who may then suffer more severe consequences of obesity-related illnesses.
Earlier this year, the American Association of Clinical Endocrinology issued a consensus statement on the role stigma and weight bias play in the management of obesity. They proposed a staging system to address the severity of obesity and suggested stigma and bias should be assessed in all patients.
While we are good at diagnosing obesity, many of us fail at addressing it empathetically with patients. I’ve seen many patients cry about past encounters they’ve had in the health care system. We need to address the emotional effect that obesity has as well as the physical complications.
Obesity is a major contributor to many diseases such as diabetes and heart disease, but we are finding it also plays a role in other diseases such as certain cancers. Treating obesity is imperative to prevent these diseases as well as to promote better treatment outcomes. We’ve all seen the diabetic patient lose weight and have their blood glucose levels come under control.
Many patients have tried hard to lose weight yet health care providers talk to them as if they haven’t made any efforts. This is very frustrating for patients. Simply telling a patient to diet and lose weight is a setup for failure. We need to address their past efforts and see what has worked and what hasn’t. Redoing the same thing over and over again is not a recipe for success.
Additionally, the focus on “diet and exercise” fails to account for emotional factors that may be contributing to a person’s obesity. Some people eat when they are stressed or depressed. It can become a habit or even an addiction. If this contributor to obesity isn’t fixed, nothing will work.
However, no medication will work well without the basic building blocks of diet and exercise. Routinely prescribing weight-loss medications without discussing diet and exercise will not result in much weight loss. Some patients simply don’t know how to eat healthfully or what they should do for exercise. A little education can go a long way. Ancillary staff, such as nutritionists or diabetic counselors, can help and free up the doctor’s time. In small practices, we can’t afford to provide those services in house but we should learn where patients can go for these services.
The AACE guidelines do a great job staging obesity. The guidelines make it easier to measure progress and decide on treatment plans. With this system, it is no longer necessary to use terms such as “excess weight” or “morbid obesity.” Patients already know they are overweight. What they need to know are clear steps so that they can reach goals. These guidelines greatly assist with providing those steps.
Most of us can do better when treating patients with obesity, We are probably not even aware of the times we have been guilty of stigmatization or weight bias. When we start treating obesity as a serious medical problem rather than something that’s the fault of the patient, it becomes much easier. When we remind ourselves what can happen to our patients when we fail to treat their obesity, we can become more serious about trying to help them reverse this critical medical problem. Bring an end to throwing out a “lose weight” or “eat healthier” suggestion to our already stressed patients. In order to address the obesity crisis that is here, we need to look inside ourselves and ask how we are going to contribute to the solution.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant of medicine at Robert Wood Johnson Medical School, New Brunswick, N.J.
Obesity is a major factor affecting the health of many Americans. It is estimated by the Centers for Disease Control and Prevention that 41% of adults and 19.7% of children in our country now meet the criteria for being obese. Obesity costs the United States approximately $147 billion annually in health care costs. While these numbers are staggering, they continue to rise.
The recent craze over medications such as Ozempic, Wegovy, and Mounjaro shows how eager people are to lose weight. Yet, many of them face bias, not just in their daily lives, but from health care professionals who should do better. No one should feel stigmatized when they come for medical help. This just drives away patients who need us and who may then suffer more severe consequences of obesity-related illnesses.
Earlier this year, the American Association of Clinical Endocrinology issued a consensus statement on the role stigma and weight bias play in the management of obesity. They proposed a staging system to address the severity of obesity and suggested stigma and bias should be assessed in all patients.
While we are good at diagnosing obesity, many of us fail at addressing it empathetically with patients. I’ve seen many patients cry about past encounters they’ve had in the health care system. We need to address the emotional effect that obesity has as well as the physical complications.
Obesity is a major contributor to many diseases such as diabetes and heart disease, but we are finding it also plays a role in other diseases such as certain cancers. Treating obesity is imperative to prevent these diseases as well as to promote better treatment outcomes. We’ve all seen the diabetic patient lose weight and have their blood glucose levels come under control.
Many patients have tried hard to lose weight yet health care providers talk to them as if they haven’t made any efforts. This is very frustrating for patients. Simply telling a patient to diet and lose weight is a setup for failure. We need to address their past efforts and see what has worked and what hasn’t. Redoing the same thing over and over again is not a recipe for success.
Additionally, the focus on “diet and exercise” fails to account for emotional factors that may be contributing to a person’s obesity. Some people eat when they are stressed or depressed. It can become a habit or even an addiction. If this contributor to obesity isn’t fixed, nothing will work.
However, no medication will work well without the basic building blocks of diet and exercise. Routinely prescribing weight-loss medications without discussing diet and exercise will not result in much weight loss. Some patients simply don’t know how to eat healthfully or what they should do for exercise. A little education can go a long way. Ancillary staff, such as nutritionists or diabetic counselors, can help and free up the doctor’s time. In small practices, we can’t afford to provide those services in house but we should learn where patients can go for these services.
The AACE guidelines do a great job staging obesity. The guidelines make it easier to measure progress and decide on treatment plans. With this system, it is no longer necessary to use terms such as “excess weight” or “morbid obesity.” Patients already know they are overweight. What they need to know are clear steps so that they can reach goals. These guidelines greatly assist with providing those steps.
Most of us can do better when treating patients with obesity, We are probably not even aware of the times we have been guilty of stigmatization or weight bias. When we start treating obesity as a serious medical problem rather than something that’s the fault of the patient, it becomes much easier. When we remind ourselves what can happen to our patients when we fail to treat their obesity, we can become more serious about trying to help them reverse this critical medical problem. Bring an end to throwing out a “lose weight” or “eat healthier” suggestion to our already stressed patients. In order to address the obesity crisis that is here, we need to look inside ourselves and ask how we are going to contribute to the solution.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant of medicine at Robert Wood Johnson Medical School, New Brunswick, N.J.
Obesity is a major factor affecting the health of many Americans. It is estimated by the Centers for Disease Control and Prevention that 41% of adults and 19.7% of children in our country now meet the criteria for being obese. Obesity costs the United States approximately $147 billion annually in health care costs. While these numbers are staggering, they continue to rise.
The recent craze over medications such as Ozempic, Wegovy, and Mounjaro shows how eager people are to lose weight. Yet, many of them face bias, not just in their daily lives, but from health care professionals who should do better. No one should feel stigmatized when they come for medical help. This just drives away patients who need us and who may then suffer more severe consequences of obesity-related illnesses.
Earlier this year, the American Association of Clinical Endocrinology issued a consensus statement on the role stigma and weight bias play in the management of obesity. They proposed a staging system to address the severity of obesity and suggested stigma and bias should be assessed in all patients.
While we are good at diagnosing obesity, many of us fail at addressing it empathetically with patients. I’ve seen many patients cry about past encounters they’ve had in the health care system. We need to address the emotional effect that obesity has as well as the physical complications.
Obesity is a major contributor to many diseases such as diabetes and heart disease, but we are finding it also plays a role in other diseases such as certain cancers. Treating obesity is imperative to prevent these diseases as well as to promote better treatment outcomes. We’ve all seen the diabetic patient lose weight and have their blood glucose levels come under control.
Many patients have tried hard to lose weight yet health care providers talk to them as if they haven’t made any efforts. This is very frustrating for patients. Simply telling a patient to diet and lose weight is a setup for failure. We need to address their past efforts and see what has worked and what hasn’t. Redoing the same thing over and over again is not a recipe for success.
Additionally, the focus on “diet and exercise” fails to account for emotional factors that may be contributing to a person’s obesity. Some people eat when they are stressed or depressed. It can become a habit or even an addiction. If this contributor to obesity isn’t fixed, nothing will work.
However, no medication will work well without the basic building blocks of diet and exercise. Routinely prescribing weight-loss medications without discussing diet and exercise will not result in much weight loss. Some patients simply don’t know how to eat healthfully or what they should do for exercise. A little education can go a long way. Ancillary staff, such as nutritionists or diabetic counselors, can help and free up the doctor’s time. In small practices, we can’t afford to provide those services in house but we should learn where patients can go for these services.
The AACE guidelines do a great job staging obesity. The guidelines make it easier to measure progress and decide on treatment plans. With this system, it is no longer necessary to use terms such as “excess weight” or “morbid obesity.” Patients already know they are overweight. What they need to know are clear steps so that they can reach goals. These guidelines greatly assist with providing those steps.
Most of us can do better when treating patients with obesity, We are probably not even aware of the times we have been guilty of stigmatization or weight bias. When we start treating obesity as a serious medical problem rather than something that’s the fault of the patient, it becomes much easier. When we remind ourselves what can happen to our patients when we fail to treat their obesity, we can become more serious about trying to help them reverse this critical medical problem. Bring an end to throwing out a “lose weight” or “eat healthier” suggestion to our already stressed patients. In order to address the obesity crisis that is here, we need to look inside ourselves and ask how we are going to contribute to the solution.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant of medicine at Robert Wood Johnson Medical School, New Brunswick, N.J.
Artificial intelligence in the office: Part 2
In the year since generative artificial intelligence (AI) software first began to emerge for use, the staggering pace and breadth of development has condensed years of growth and change into months and weeks. Among the settings where these tools may find the greatest straight-line relevance is private medical practice.
. A multitude of generative AI products with potential medical applications are now available, with new ones appearing almost weekly. (As always, I have no financial interest in any product or service mentioned in this column.)
Last month, I discussed ChatGPT, the best-known AI algorithm, and some of its applications in clinical practice, such as generating website, video, and blog content. ChatGPT can also provide rapid and concise answers to general medical questions, like a search engine – but with more natural language processing and contextual understanding. Additionally, the algorithm can draft generic medical documents, including templates for after-visit summaries, postprocedure instructions, referrals, prior authorization appeal letters, and educational handouts.
Another useful feature of ChatGPT is its ability to provide accurate and conversational language translations, thus serving as an interpreter during clinic visits in situations where a human translator is not available. It also has potential uses in clinical research by finding resources, formulating hypotheses, drafting study protocols, and collecting large amounts of data in short periods of time. Other possibilities include survey administration, clinical trial recruitment, and automatic medication monitoring.
GPT-4, the latest version of ChatGPT, is reported to have greater problem-solving abilities and an even broader knowledge base. Among its claimed skills are the ability to find the latest literature in a given area, write a discharge summary for a patient following an uncomplicated surgery, and an image analysis feature to identify objects in photos. GPT-4 has been praised as having “the potential to help drive medical innovation, from aiding with patient discharge notes, summarizing recent clinical trials, providing information on ethical guidelines, and much more.”
Bard, an AI “chat bot” introduced by Google earlier this year, intends to leverage Google’s enormous database to compete with ChatGPT in providing answers to medical questions. Bard also hopes to play a pivotal role in expanding telemedicine and remote care via Google’s secure connections and access to patient records and medical history, and “facilitate seamless communication through appointment scheduling, messaging, and sharing medical images,” according to PackT, a website for IT professionals. The company claims that Bard’s integration of AI and machine learning capabilities will serve to elevate health care efficiency and patient outcomes, PackT says, and “the platform’s AI system quickly and accurately analyzes patient records, identifies patterns and trends, and aids medical professionals in developing effective treatment plans.”
Doximity has introduced an AI engine called DocsGPT, an encrypted, HIPAA-compliant writing assistant that, the company says, can draft any form of professional correspondence, including prior authorization letters, insurance appeals, patient support letters, and patient education materials. The service is available at no charge to all U.S. physicians and medical students through their Doximity accounts.
Microsoft has introduced several AI products. BioGPT is a language model specifically designed for health care. Compared with GPT models that are trained on more general text data, BioGPT is purported to have a deeper understanding of the language used in biomedical research and can generate more accurate and relevant outputs for biomedical tasks, such as drug discovery, disease classification, and clinical decision support. Fabric is another health care–specific data and analytics platform the company described in an announcement in May. It can combine data from sources such as electronic health records, images, lab systems, medical devices, and claims systems so hospitals and offices can standardize it and access it in the same place. Microsoft said the new tools will help eliminate the “time-consuming” process of searching through these sources one by one. Microsoft will also offer a new generative AI chatbot called the Azure Health Bot, which can pull information from a health organization’s own internal data as well as reputable external sources such as the Food and Drug Administration and the National Institutes of Health.
Several other AI products are available for clinicians. Tana served as an administrative aid and a clinical helper during the height of the COVID-19 pandemic, answering frequently asked questions, facilitating appointment management, and gathering preliminary medical information prior to teleconsultations. Dougall GPT is another AI chatbot tailored for health care professionals. It provides clinicians with AI-tuned answers to their queries, augmented by links to relevant, up-to-date, authoritative resources. It also assists in drafting patient instructions, consultation summaries, speeches, and professional correspondence. Wang has created Clinical Camel, an open-source health care–focused chatbot that assembles medical data with a combination of user-shared conversations and synthetic conversations derived from curated clinical articles. The Chinese company Baidu has rolled out Ernie as a potential rival to ChatGPT. You get the idea.
Of course, the inherent drawbacks of AI, such as producing false or biased information, perpetuating harmful stereotypes, and presenting information that has since been proven inaccurate or out-of-date, must always be kept in mind. All AI algorithms have been criticized for giving wrong answers, as their datasets are generally culled from information published in 2021 or earlier. Several of them have been shown to fabricate information – a phenomenon labeled “artificial hallucinations” in one article. “The scientific community must be vigilant in verifying the accuracy and reliability of the information provided by AI tools,” wrote the authors of that paper. “Researchers should use AI as an aid rather than a replacement for critical thinking and fact-checking.”
In the year since generative artificial intelligence (AI) software first began to emerge for use, the staggering pace and breadth of development has condensed years of growth and change into months and weeks. Among the settings where these tools may find the greatest straight-line relevance is private medical practice.
. A multitude of generative AI products with potential medical applications are now available, with new ones appearing almost weekly. (As always, I have no financial interest in any product or service mentioned in this column.)
Last month, I discussed ChatGPT, the best-known AI algorithm, and some of its applications in clinical practice, such as generating website, video, and blog content. ChatGPT can also provide rapid and concise answers to general medical questions, like a search engine – but with more natural language processing and contextual understanding. Additionally, the algorithm can draft generic medical documents, including templates for after-visit summaries, postprocedure instructions, referrals, prior authorization appeal letters, and educational handouts.
Another useful feature of ChatGPT is its ability to provide accurate and conversational language translations, thus serving as an interpreter during clinic visits in situations where a human translator is not available. It also has potential uses in clinical research by finding resources, formulating hypotheses, drafting study protocols, and collecting large amounts of data in short periods of time. Other possibilities include survey administration, clinical trial recruitment, and automatic medication monitoring.
GPT-4, the latest version of ChatGPT, is reported to have greater problem-solving abilities and an even broader knowledge base. Among its claimed skills are the ability to find the latest literature in a given area, write a discharge summary for a patient following an uncomplicated surgery, and an image analysis feature to identify objects in photos. GPT-4 has been praised as having “the potential to help drive medical innovation, from aiding with patient discharge notes, summarizing recent clinical trials, providing information on ethical guidelines, and much more.”
Bard, an AI “chat bot” introduced by Google earlier this year, intends to leverage Google’s enormous database to compete with ChatGPT in providing answers to medical questions. Bard also hopes to play a pivotal role in expanding telemedicine and remote care via Google’s secure connections and access to patient records and medical history, and “facilitate seamless communication through appointment scheduling, messaging, and sharing medical images,” according to PackT, a website for IT professionals. The company claims that Bard’s integration of AI and machine learning capabilities will serve to elevate health care efficiency and patient outcomes, PackT says, and “the platform’s AI system quickly and accurately analyzes patient records, identifies patterns and trends, and aids medical professionals in developing effective treatment plans.”
Doximity has introduced an AI engine called DocsGPT, an encrypted, HIPAA-compliant writing assistant that, the company says, can draft any form of professional correspondence, including prior authorization letters, insurance appeals, patient support letters, and patient education materials. The service is available at no charge to all U.S. physicians and medical students through their Doximity accounts.
Microsoft has introduced several AI products. BioGPT is a language model specifically designed for health care. Compared with GPT models that are trained on more general text data, BioGPT is purported to have a deeper understanding of the language used in biomedical research and can generate more accurate and relevant outputs for biomedical tasks, such as drug discovery, disease classification, and clinical decision support. Fabric is another health care–specific data and analytics platform the company described in an announcement in May. It can combine data from sources such as electronic health records, images, lab systems, medical devices, and claims systems so hospitals and offices can standardize it and access it in the same place. Microsoft said the new tools will help eliminate the “time-consuming” process of searching through these sources one by one. Microsoft will also offer a new generative AI chatbot called the Azure Health Bot, which can pull information from a health organization’s own internal data as well as reputable external sources such as the Food and Drug Administration and the National Institutes of Health.
Several other AI products are available for clinicians. Tana served as an administrative aid and a clinical helper during the height of the COVID-19 pandemic, answering frequently asked questions, facilitating appointment management, and gathering preliminary medical information prior to teleconsultations. Dougall GPT is another AI chatbot tailored for health care professionals. It provides clinicians with AI-tuned answers to their queries, augmented by links to relevant, up-to-date, authoritative resources. It also assists in drafting patient instructions, consultation summaries, speeches, and professional correspondence. Wang has created Clinical Camel, an open-source health care–focused chatbot that assembles medical data with a combination of user-shared conversations and synthetic conversations derived from curated clinical articles. The Chinese company Baidu has rolled out Ernie as a potential rival to ChatGPT. You get the idea.
Of course, the inherent drawbacks of AI, such as producing false or biased information, perpetuating harmful stereotypes, and presenting information that has since been proven inaccurate or out-of-date, must always be kept in mind. All AI algorithms have been criticized for giving wrong answers, as their datasets are generally culled from information published in 2021 or earlier. Several of them have been shown to fabricate information – a phenomenon labeled “artificial hallucinations” in one article. “The scientific community must be vigilant in verifying the accuracy and reliability of the information provided by AI tools,” wrote the authors of that paper. “Researchers should use AI as an aid rather than a replacement for critical thinking and fact-checking.”
In the year since generative artificial intelligence (AI) software first began to emerge for use, the staggering pace and breadth of development has condensed years of growth and change into months and weeks. Among the settings where these tools may find the greatest straight-line relevance is private medical practice.
. A multitude of generative AI products with potential medical applications are now available, with new ones appearing almost weekly. (As always, I have no financial interest in any product or service mentioned in this column.)
Last month, I discussed ChatGPT, the best-known AI algorithm, and some of its applications in clinical practice, such as generating website, video, and blog content. ChatGPT can also provide rapid and concise answers to general medical questions, like a search engine – but with more natural language processing and contextual understanding. Additionally, the algorithm can draft generic medical documents, including templates for after-visit summaries, postprocedure instructions, referrals, prior authorization appeal letters, and educational handouts.
Another useful feature of ChatGPT is its ability to provide accurate and conversational language translations, thus serving as an interpreter during clinic visits in situations where a human translator is not available. It also has potential uses in clinical research by finding resources, formulating hypotheses, drafting study protocols, and collecting large amounts of data in short periods of time. Other possibilities include survey administration, clinical trial recruitment, and automatic medication monitoring.
GPT-4, the latest version of ChatGPT, is reported to have greater problem-solving abilities and an even broader knowledge base. Among its claimed skills are the ability to find the latest literature in a given area, write a discharge summary for a patient following an uncomplicated surgery, and an image analysis feature to identify objects in photos. GPT-4 has been praised as having “the potential to help drive medical innovation, from aiding with patient discharge notes, summarizing recent clinical trials, providing information on ethical guidelines, and much more.”
Bard, an AI “chat bot” introduced by Google earlier this year, intends to leverage Google’s enormous database to compete with ChatGPT in providing answers to medical questions. Bard also hopes to play a pivotal role in expanding telemedicine and remote care via Google’s secure connections and access to patient records and medical history, and “facilitate seamless communication through appointment scheduling, messaging, and sharing medical images,” according to PackT, a website for IT professionals. The company claims that Bard’s integration of AI and machine learning capabilities will serve to elevate health care efficiency and patient outcomes, PackT says, and “the platform’s AI system quickly and accurately analyzes patient records, identifies patterns and trends, and aids medical professionals in developing effective treatment plans.”
Doximity has introduced an AI engine called DocsGPT, an encrypted, HIPAA-compliant writing assistant that, the company says, can draft any form of professional correspondence, including prior authorization letters, insurance appeals, patient support letters, and patient education materials. The service is available at no charge to all U.S. physicians and medical students through their Doximity accounts.
Microsoft has introduced several AI products. BioGPT is a language model specifically designed for health care. Compared with GPT models that are trained on more general text data, BioGPT is purported to have a deeper understanding of the language used in biomedical research and can generate more accurate and relevant outputs for biomedical tasks, such as drug discovery, disease classification, and clinical decision support. Fabric is another health care–specific data and analytics platform the company described in an announcement in May. It can combine data from sources such as electronic health records, images, lab systems, medical devices, and claims systems so hospitals and offices can standardize it and access it in the same place. Microsoft said the new tools will help eliminate the “time-consuming” process of searching through these sources one by one. Microsoft will also offer a new generative AI chatbot called the Azure Health Bot, which can pull information from a health organization’s own internal data as well as reputable external sources such as the Food and Drug Administration and the National Institutes of Health.
Several other AI products are available for clinicians. Tana served as an administrative aid and a clinical helper during the height of the COVID-19 pandemic, answering frequently asked questions, facilitating appointment management, and gathering preliminary medical information prior to teleconsultations. Dougall GPT is another AI chatbot tailored for health care professionals. It provides clinicians with AI-tuned answers to their queries, augmented by links to relevant, up-to-date, authoritative resources. It also assists in drafting patient instructions, consultation summaries, speeches, and professional correspondence. Wang has created Clinical Camel, an open-source health care–focused chatbot that assembles medical data with a combination of user-shared conversations and synthetic conversations derived from curated clinical articles. The Chinese company Baidu has rolled out Ernie as a potential rival to ChatGPT. You get the idea.
Of course, the inherent drawbacks of AI, such as producing false or biased information, perpetuating harmful stereotypes, and presenting information that has since been proven inaccurate or out-of-date, must always be kept in mind. All AI algorithms have been criticized for giving wrong answers, as their datasets are generally culled from information published in 2021 or earlier. Several of them have been shown to fabricate information – a phenomenon labeled “artificial hallucinations” in one article. “The scientific community must be vigilant in verifying the accuracy and reliability of the information provided by AI tools,” wrote the authors of that paper. “Researchers should use AI as an aid rather than a replacement for critical thinking and fact-checking.”
Neoadjuvant advantages: Treating locally advanced lung cancer
Many of you saw the press release from Merck announcing that their randomized trial comparing chemo with chemo plus pembrolizumab in the neoadjuvant setting led to improved event-free survival and also improved pathologic complete response rate.
This comes in addition to the data from the AstraZeneca trial with durvalumab saying they’ve already achieved their endpoint of higher pathologic complete response rate vs. chemotherapy alone and also the data with nivolumab from Bristol-Myers Squibb saying that nivolumab plus chemotherapy leads to a better event-free survival and a better pathologic complete response rate. That information has led to Food and Drug Administration approval for their regimen.
We’re running the table with these very positive data, and I think it’s just a sign that the approach is safe and effective.
A huge question has come up. I just came from a meeting of lung cancer experts asking what to do if you have a patient with a small tumor, for example, a 3-cm tumor. Do you recommend immediate surgery followed by adjuvant therapy, chemotherapy, and then a checkpoint inhibitor if appropriate? Or do you proceed with neoadjuvant therapy if appropriate? The truth is that it’s a very difficult decision.
We have overwhelming data that the neoadjuvant approach works for that patient. Please remember that this is a clinically staged patient. This is not the patient after their surgery, where I think we have a very clear path. We have adjuvant data and adjuvant trials for those patients.
For the patient who’s in your office with a small tumor or a small tumor and only hilar lymphadenopathy, the decision there isn’t data driven, but rather it is experience driven. The data that are out there right now suggest that neoadjuvant therapy is a better way to go. Why is that?
Well, I think that the first reason is that it is probably a better regimen. I think many of you saw the recent clinical trial by Patel and colleagues in the New England Journal of Medicine with melanoma. It was an interesting trial. They gave a checkpoint inhibitor for 18 doses after surgery for melanoma versus three doses of checkpoint inhibitor, surgery, and then 15 doses of the checkpoint inhibitor.
It was 18 doses versus 18 doses, with the only difference being the three doses before surgery. Lo and behold, the three doses before surgery led to a better event-free survival.
There are preclinical data in lung cancer demonstrating that the same thing is true. Tina Cascone published on that years ago. We could talk about why, but it appears that neoadjuvant is just better.
There are other advantages to it as well. I think a big one is that all the information shows that it’s better tolerated, so you’re more likely to give all the drug. You can see if the drug isn’t working, and you can stop the drug. Also, if the drug is causing a side effect, you can see whether it’s working or not and use that decision to stop. It’s different than when you’re giving a drug in the adjuvant setting where you don’t really know whether it is working or not.
I think that it’s time to change some of our standards. When patients appear with lung cancers other than tiny ones that might be detected through screening, you need to convene your multidisciplinary group. You need to weigh the pros and cons I think that it’s time to change some of our standards. When patients appear with lung cancers other than tiny ones that might be detected through screening, you need to convene your multidisciplinary group coming in. It’s already an FDA-approved regimen with nivolumab and chemotherapy, and I think we’re moving to making that our standard of care now.
The way to handle it today, though, is to convene your multidisciplinary panel about every patient other than those with the tiniest of lung cancers and put your heads together to see what the best treatment is for that patient.
Dr. Kris is professor of medicine, Weill Cornell Medicine, and the William and Joy Ruane Chair in Thoracic Oncology, Memorial Sloan Kettering Cancer Center, both in New York. He disclosed ties with Ariad Pharmaceuticals, AstraZeneca, Pfizer, PUMA, and Roche/Genentech.
A version of this article appeared on Medscape.com.
Many of you saw the press release from Merck announcing that their randomized trial comparing chemo with chemo plus pembrolizumab in the neoadjuvant setting led to improved event-free survival and also improved pathologic complete response rate.
This comes in addition to the data from the AstraZeneca trial with durvalumab saying they’ve already achieved their endpoint of higher pathologic complete response rate vs. chemotherapy alone and also the data with nivolumab from Bristol-Myers Squibb saying that nivolumab plus chemotherapy leads to a better event-free survival and a better pathologic complete response rate. That information has led to Food and Drug Administration approval for their regimen.
We’re running the table with these very positive data, and I think it’s just a sign that the approach is safe and effective.
A huge question has come up. I just came from a meeting of lung cancer experts asking what to do if you have a patient with a small tumor, for example, a 3-cm tumor. Do you recommend immediate surgery followed by adjuvant therapy, chemotherapy, and then a checkpoint inhibitor if appropriate? Or do you proceed with neoadjuvant therapy if appropriate? The truth is that it’s a very difficult decision.
We have overwhelming data that the neoadjuvant approach works for that patient. Please remember that this is a clinically staged patient. This is not the patient after their surgery, where I think we have a very clear path. We have adjuvant data and adjuvant trials for those patients.
For the patient who’s in your office with a small tumor or a small tumor and only hilar lymphadenopathy, the decision there isn’t data driven, but rather it is experience driven. The data that are out there right now suggest that neoadjuvant therapy is a better way to go. Why is that?
Well, I think that the first reason is that it is probably a better regimen. I think many of you saw the recent clinical trial by Patel and colleagues in the New England Journal of Medicine with melanoma. It was an interesting trial. They gave a checkpoint inhibitor for 18 doses after surgery for melanoma versus three doses of checkpoint inhibitor, surgery, and then 15 doses of the checkpoint inhibitor.
It was 18 doses versus 18 doses, with the only difference being the three doses before surgery. Lo and behold, the three doses before surgery led to a better event-free survival.
There are preclinical data in lung cancer demonstrating that the same thing is true. Tina Cascone published on that years ago. We could talk about why, but it appears that neoadjuvant is just better.
There are other advantages to it as well. I think a big one is that all the information shows that it’s better tolerated, so you’re more likely to give all the drug. You can see if the drug isn’t working, and you can stop the drug. Also, if the drug is causing a side effect, you can see whether it’s working or not and use that decision to stop. It’s different than when you’re giving a drug in the adjuvant setting where you don’t really know whether it is working or not.
I think that it’s time to change some of our standards. When patients appear with lung cancers other than tiny ones that might be detected through screening, you need to convene your multidisciplinary group. You need to weigh the pros and cons I think that it’s time to change some of our standards. When patients appear with lung cancers other than tiny ones that might be detected through screening, you need to convene your multidisciplinary group coming in. It’s already an FDA-approved regimen with nivolumab and chemotherapy, and I think we’re moving to making that our standard of care now.
The way to handle it today, though, is to convene your multidisciplinary panel about every patient other than those with the tiniest of lung cancers and put your heads together to see what the best treatment is for that patient.
Dr. Kris is professor of medicine, Weill Cornell Medicine, and the William and Joy Ruane Chair in Thoracic Oncology, Memorial Sloan Kettering Cancer Center, both in New York. He disclosed ties with Ariad Pharmaceuticals, AstraZeneca, Pfizer, PUMA, and Roche/Genentech.
A version of this article appeared on Medscape.com.
Many of you saw the press release from Merck announcing that their randomized trial comparing chemo with chemo plus pembrolizumab in the neoadjuvant setting led to improved event-free survival and also improved pathologic complete response rate.
This comes in addition to the data from the AstraZeneca trial with durvalumab saying they’ve already achieved their endpoint of higher pathologic complete response rate vs. chemotherapy alone and also the data with nivolumab from Bristol-Myers Squibb saying that nivolumab plus chemotherapy leads to a better event-free survival and a better pathologic complete response rate. That information has led to Food and Drug Administration approval for their regimen.
We’re running the table with these very positive data, and I think it’s just a sign that the approach is safe and effective.
A huge question has come up. I just came from a meeting of lung cancer experts asking what to do if you have a patient with a small tumor, for example, a 3-cm tumor. Do you recommend immediate surgery followed by adjuvant therapy, chemotherapy, and then a checkpoint inhibitor if appropriate? Or do you proceed with neoadjuvant therapy if appropriate? The truth is that it’s a very difficult decision.
We have overwhelming data that the neoadjuvant approach works for that patient. Please remember that this is a clinically staged patient. This is not the patient after their surgery, where I think we have a very clear path. We have adjuvant data and adjuvant trials for those patients.
For the patient who’s in your office with a small tumor or a small tumor and only hilar lymphadenopathy, the decision there isn’t data driven, but rather it is experience driven. The data that are out there right now suggest that neoadjuvant therapy is a better way to go. Why is that?
Well, I think that the first reason is that it is probably a better regimen. I think many of you saw the recent clinical trial by Patel and colleagues in the New England Journal of Medicine with melanoma. It was an interesting trial. They gave a checkpoint inhibitor for 18 doses after surgery for melanoma versus three doses of checkpoint inhibitor, surgery, and then 15 doses of the checkpoint inhibitor.
It was 18 doses versus 18 doses, with the only difference being the three doses before surgery. Lo and behold, the three doses before surgery led to a better event-free survival.
There are preclinical data in lung cancer demonstrating that the same thing is true. Tina Cascone published on that years ago. We could talk about why, but it appears that neoadjuvant is just better.
There are other advantages to it as well. I think a big one is that all the information shows that it’s better tolerated, so you’re more likely to give all the drug. You can see if the drug isn’t working, and you can stop the drug. Also, if the drug is causing a side effect, you can see whether it’s working or not and use that decision to stop. It’s different than when you’re giving a drug in the adjuvant setting where you don’t really know whether it is working or not.
I think that it’s time to change some of our standards. When patients appear with lung cancers other than tiny ones that might be detected through screening, you need to convene your multidisciplinary group. You need to weigh the pros and cons I think that it’s time to change some of our standards. When patients appear with lung cancers other than tiny ones that might be detected through screening, you need to convene your multidisciplinary group coming in. It’s already an FDA-approved regimen with nivolumab and chemotherapy, and I think we’re moving to making that our standard of care now.
The way to handle it today, though, is to convene your multidisciplinary panel about every patient other than those with the tiniest of lung cancers and put your heads together to see what the best treatment is for that patient.
Dr. Kris is professor of medicine, Weill Cornell Medicine, and the William and Joy Ruane Chair in Thoracic Oncology, Memorial Sloan Kettering Cancer Center, both in New York. He disclosed ties with Ariad Pharmaceuticals, AstraZeneca, Pfizer, PUMA, and Roche/Genentech.
A version of this article appeared on Medscape.com.
New kids on the block for migraine treatment and prophylaxis
This transcript has been edited for clarity.
Dear colleagues, I’m Hans-Christoph Diener from the Faculty of Medicine at the University of Duisburg-Essen in Germany.
CGRP receptor agonists
Let me start with the treatment of acute migraine attacks. Until recently, we had analgesics, nonsteroidal anti-inflammatory drugs like ibuprofen, ergot alkaloids, and triptans. There are new developments, which are small molecules that are antagonists at the calcitonin gene-related peptide (CGRP) receptor. At the moment, we have three of them: rimegepant 75 mg, ubrogepant 50 mg or 100 mg, and zavegepant (a nasal spray) 10 mg.
These are all effective and superior to placebo. The 2-hour pain-free rate is somewhere between 25% and 30%. They have very few side effects; these include a little bit of nausea, somnolence, nasopharyngitis, and for zavegepant, the nasal spray, taste disturbance. In indirect comparisons, the so-called gepants are about as effective as ibuprofen and aspirin, and they seem to be less effective than sumatriptan 100 mg.
Unfortunately, until now, we have no direct comparison with triptans and we have no data demonstrating whether they are effective in people where triptans do not work. The major shortcoming is the cost in the United States. The cost per tablet or nasal spray is somewhere between $80 and $200. This means we definitely need more studies for these gepants.
Migraine prophylaxis
Let me move to the prophylaxis of migraine with drugs. Previously and still, we have all medications like beta-blockers, flunarizine, topiramate, valproic acid, amitriptyline, and candesartan, and for chronic migraine, onabotulinumtoxinA. We have now 5 years’ experience with the monoclonal antibodies against CGRP or the CGRP receptor like eptinezumab, erenumab, fremanezumab, and galcanezumab.
These are all equally effective. They reduce migraine-days between 3 and 7 per month. They are effective both in episodic and chronic migraine, and most importantly, they are effective in people with medication overuse headaches. The 50% responder rates are somewhere between 40% and 60%, and there are no significant differences between the four monoclonal antibodies.
The major advantage is a very good tolerability profile; very few patients terminate treatment because of adverse events. There has been, with one exception, no direct comparison of the monoclonal antibodies with traditional migraine preventive drugs or onabotulinumtoxinA. The only exception is a trial that compared topiramate and erenumab, showing that erenumab was definitely more effective and better tolerated.
At the moment, the recommendation is to use these monoclonal antibodies for 12 months in episodic migraine and 24 months in chronic migraine and then pause. It usually turns out that between 50% and 70% of these patients need to continue the treatment. If they are not working, there is a possibility to switch between the monoclonal antibodies, and the success rate after this is somewhere between 15% and 30%.
Gepants were also developed for the prevention of migraine. Here, we have rimegepant 75 mg every other day or atogepant 60 mg daily. They are effective, but in indirect comparisons, they are less effective than the monoclonal antibodies. At present, we have no comparative trials with monoclonal antibodies or the traditional migraine preventive drugs.
Potential patients are those who have needle phobia or patients who do not respond to monoclonal antibodies. Again, the biggest shortcoming is cost in the United States. The cost per year for migraine prevention or prophylaxis is between $12,000 and $20,000.
Finally, we also had very exciting news. There is a new therapeutic approach via PACAP. PACAP is pituitary adenylate cyclase-activating polypeptide, which has similar biological actions as CGRP but with additional actions. It could very well be that people who do not respond to a monoclonal antibody would respond to a monoclonal antibody against PACAP.
At the congress, the first randomized, placebo-controlled trial with a monoclonal antibody against PACAP was presented. This monoclonal antibody was effective in a population of people in whom prior preventive therapy had failed. A phase 3 study is planned, and most probably the PACAP monoclonal could work in people who do not respond to monoclonal antibodies against CGRP.
Dear colleagues, we have now many choices for the acute treatment of migraine and migraine prophylaxis. We have new kids on the block, and we have to learn more about how to use these drugs, their benefits, and their shortcomings.
He has disclosed the following relevant financial relationships:Received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbott; Addex Pharma; Alder; Allergan; Almirall; Amgen; Autonomic Technology; AstraZeneca; Bayer Vital; Berlin Chemie; Bristol-Myers Squibb; Boehringer Ingelheim; Chordate; CoAxia; Corimmun; Covidien; Coherex; CoLucid; Daiichi-Sankyo; D-Pharm; Electrocore; Fresenius; GlaxoSmithKline; Grunenthal; Janssen-Cilag; Labrys Biologics Lilly; La Roche; 3M Medica; MSD; Medtronic; Menarini; MindFrame; Minster; Neuroscore; Neurobiological Technologies; Novartis; Novo Nordisk; Johnson & Johnson; Knoll; Paion; Parke-Davis; Pierre Fabre; Pfizer; Schaper and Brummer; Sanofi-Aventis; Schering-Plough; Servier; Solvay; Syngis; St. Jude; Talecris; Thrombogenics; WebMD Global; Weber and Weber; Wyeth; and Yamanouchi.
Dr. Diener is professor, department of neurology, Stroke Center-Headache Center, University Duisburg-Essen (Germany).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Dear colleagues, I’m Hans-Christoph Diener from the Faculty of Medicine at the University of Duisburg-Essen in Germany.
CGRP receptor agonists
Let me start with the treatment of acute migraine attacks. Until recently, we had analgesics, nonsteroidal anti-inflammatory drugs like ibuprofen, ergot alkaloids, and triptans. There are new developments, which are small molecules that are antagonists at the calcitonin gene-related peptide (CGRP) receptor. At the moment, we have three of them: rimegepant 75 mg, ubrogepant 50 mg or 100 mg, and zavegepant (a nasal spray) 10 mg.
These are all effective and superior to placebo. The 2-hour pain-free rate is somewhere between 25% and 30%. They have very few side effects; these include a little bit of nausea, somnolence, nasopharyngitis, and for zavegepant, the nasal spray, taste disturbance. In indirect comparisons, the so-called gepants are about as effective as ibuprofen and aspirin, and they seem to be less effective than sumatriptan 100 mg.
Unfortunately, until now, we have no direct comparison with triptans and we have no data demonstrating whether they are effective in people where triptans do not work. The major shortcoming is the cost in the United States. The cost per tablet or nasal spray is somewhere between $80 and $200. This means we definitely need more studies for these gepants.
Migraine prophylaxis
Let me move to the prophylaxis of migraine with drugs. Previously and still, we have all medications like beta-blockers, flunarizine, topiramate, valproic acid, amitriptyline, and candesartan, and for chronic migraine, onabotulinumtoxinA. We have now 5 years’ experience with the monoclonal antibodies against CGRP or the CGRP receptor like eptinezumab, erenumab, fremanezumab, and galcanezumab.
These are all equally effective. They reduce migraine-days between 3 and 7 per month. They are effective both in episodic and chronic migraine, and most importantly, they are effective in people with medication overuse headaches. The 50% responder rates are somewhere between 40% and 60%, and there are no significant differences between the four monoclonal antibodies.
The major advantage is a very good tolerability profile; very few patients terminate treatment because of adverse events. There has been, with one exception, no direct comparison of the monoclonal antibodies with traditional migraine preventive drugs or onabotulinumtoxinA. The only exception is a trial that compared topiramate and erenumab, showing that erenumab was definitely more effective and better tolerated.
At the moment, the recommendation is to use these monoclonal antibodies for 12 months in episodic migraine and 24 months in chronic migraine and then pause. It usually turns out that between 50% and 70% of these patients need to continue the treatment. If they are not working, there is a possibility to switch between the monoclonal antibodies, and the success rate after this is somewhere between 15% and 30%.
Gepants were also developed for the prevention of migraine. Here, we have rimegepant 75 mg every other day or atogepant 60 mg daily. They are effective, but in indirect comparisons, they are less effective than the monoclonal antibodies. At present, we have no comparative trials with monoclonal antibodies or the traditional migraine preventive drugs.
Potential patients are those who have needle phobia or patients who do not respond to monoclonal antibodies. Again, the biggest shortcoming is cost in the United States. The cost per year for migraine prevention or prophylaxis is between $12,000 and $20,000.
Finally, we also had very exciting news. There is a new therapeutic approach via PACAP. PACAP is pituitary adenylate cyclase-activating polypeptide, which has similar biological actions as CGRP but with additional actions. It could very well be that people who do not respond to a monoclonal antibody would respond to a monoclonal antibody against PACAP.
At the congress, the first randomized, placebo-controlled trial with a monoclonal antibody against PACAP was presented. This monoclonal antibody was effective in a population of people in whom prior preventive therapy had failed. A phase 3 study is planned, and most probably the PACAP monoclonal could work in people who do not respond to monoclonal antibodies against CGRP.
Dear colleagues, we have now many choices for the acute treatment of migraine and migraine prophylaxis. We have new kids on the block, and we have to learn more about how to use these drugs, their benefits, and their shortcomings.
He has disclosed the following relevant financial relationships:Received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbott; Addex Pharma; Alder; Allergan; Almirall; Amgen; Autonomic Technology; AstraZeneca; Bayer Vital; Berlin Chemie; Bristol-Myers Squibb; Boehringer Ingelheim; Chordate; CoAxia; Corimmun; Covidien; Coherex; CoLucid; Daiichi-Sankyo; D-Pharm; Electrocore; Fresenius; GlaxoSmithKline; Grunenthal; Janssen-Cilag; Labrys Biologics Lilly; La Roche; 3M Medica; MSD; Medtronic; Menarini; MindFrame; Minster; Neuroscore; Neurobiological Technologies; Novartis; Novo Nordisk; Johnson & Johnson; Knoll; Paion; Parke-Davis; Pierre Fabre; Pfizer; Schaper and Brummer; Sanofi-Aventis; Schering-Plough; Servier; Solvay; Syngis; St. Jude; Talecris; Thrombogenics; WebMD Global; Weber and Weber; Wyeth; and Yamanouchi.
Dr. Diener is professor, department of neurology, Stroke Center-Headache Center, University Duisburg-Essen (Germany).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Dear colleagues, I’m Hans-Christoph Diener from the Faculty of Medicine at the University of Duisburg-Essen in Germany.
CGRP receptor agonists
Let me start with the treatment of acute migraine attacks. Until recently, we had analgesics, nonsteroidal anti-inflammatory drugs like ibuprofen, ergot alkaloids, and triptans. There are new developments, which are small molecules that are antagonists at the calcitonin gene-related peptide (CGRP) receptor. At the moment, we have three of them: rimegepant 75 mg, ubrogepant 50 mg or 100 mg, and zavegepant (a nasal spray) 10 mg.
These are all effective and superior to placebo. The 2-hour pain-free rate is somewhere between 25% and 30%. They have very few side effects; these include a little bit of nausea, somnolence, nasopharyngitis, and for zavegepant, the nasal spray, taste disturbance. In indirect comparisons, the so-called gepants are about as effective as ibuprofen and aspirin, and they seem to be less effective than sumatriptan 100 mg.
Unfortunately, until now, we have no direct comparison with triptans and we have no data demonstrating whether they are effective in people where triptans do not work. The major shortcoming is the cost in the United States. The cost per tablet or nasal spray is somewhere between $80 and $200. This means we definitely need more studies for these gepants.
Migraine prophylaxis
Let me move to the prophylaxis of migraine with drugs. Previously and still, we have all medications like beta-blockers, flunarizine, topiramate, valproic acid, amitriptyline, and candesartan, and for chronic migraine, onabotulinumtoxinA. We have now 5 years’ experience with the monoclonal antibodies against CGRP or the CGRP receptor like eptinezumab, erenumab, fremanezumab, and galcanezumab.
These are all equally effective. They reduce migraine-days between 3 and 7 per month. They are effective both in episodic and chronic migraine, and most importantly, they are effective in people with medication overuse headaches. The 50% responder rates are somewhere between 40% and 60%, and there are no significant differences between the four monoclonal antibodies.
The major advantage is a very good tolerability profile; very few patients terminate treatment because of adverse events. There has been, with one exception, no direct comparison of the monoclonal antibodies with traditional migraine preventive drugs or onabotulinumtoxinA. The only exception is a trial that compared topiramate and erenumab, showing that erenumab was definitely more effective and better tolerated.
At the moment, the recommendation is to use these monoclonal antibodies for 12 months in episodic migraine and 24 months in chronic migraine and then pause. It usually turns out that between 50% and 70% of these patients need to continue the treatment. If they are not working, there is a possibility to switch between the monoclonal antibodies, and the success rate after this is somewhere between 15% and 30%.
Gepants were also developed for the prevention of migraine. Here, we have rimegepant 75 mg every other day or atogepant 60 mg daily. They are effective, but in indirect comparisons, they are less effective than the monoclonal antibodies. At present, we have no comparative trials with monoclonal antibodies or the traditional migraine preventive drugs.
Potential patients are those who have needle phobia or patients who do not respond to monoclonal antibodies. Again, the biggest shortcoming is cost in the United States. The cost per year for migraine prevention or prophylaxis is between $12,000 and $20,000.
Finally, we also had very exciting news. There is a new therapeutic approach via PACAP. PACAP is pituitary adenylate cyclase-activating polypeptide, which has similar biological actions as CGRP but with additional actions. It could very well be that people who do not respond to a monoclonal antibody would respond to a monoclonal antibody against PACAP.
At the congress, the first randomized, placebo-controlled trial with a monoclonal antibody against PACAP was presented. This monoclonal antibody was effective in a population of people in whom prior preventive therapy had failed. A phase 3 study is planned, and most probably the PACAP monoclonal could work in people who do not respond to monoclonal antibodies against CGRP.
Dear colleagues, we have now many choices for the acute treatment of migraine and migraine prophylaxis. We have new kids on the block, and we have to learn more about how to use these drugs, their benefits, and their shortcomings.
He has disclosed the following relevant financial relationships:Received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbott; Addex Pharma; Alder; Allergan; Almirall; Amgen; Autonomic Technology; AstraZeneca; Bayer Vital; Berlin Chemie; Bristol-Myers Squibb; Boehringer Ingelheim; Chordate; CoAxia; Corimmun; Covidien; Coherex; CoLucid; Daiichi-Sankyo; D-Pharm; Electrocore; Fresenius; GlaxoSmithKline; Grunenthal; Janssen-Cilag; Labrys Biologics Lilly; La Roche; 3M Medica; MSD; Medtronic; Menarini; MindFrame; Minster; Neuroscore; Neurobiological Technologies; Novartis; Novo Nordisk; Johnson & Johnson; Knoll; Paion; Parke-Davis; Pierre Fabre; Pfizer; Schaper and Brummer; Sanofi-Aventis; Schering-Plough; Servier; Solvay; Syngis; St. Jude; Talecris; Thrombogenics; WebMD Global; Weber and Weber; Wyeth; and Yamanouchi.
Dr. Diener is professor, department of neurology, Stroke Center-Headache Center, University Duisburg-Essen (Germany).
A version of this article appeared on Medscape.com.
Where do you stand on the Middle East conflict?
“What do you think about the whole Israel thing?”
That question came at the end of an otherwise routine appointment.
Maybe she was just chatting. Maybe she wanted something deeper. I have no idea. I just said, “I don’t discuss those things with patients.”
My answer surprised her, but she didn’t push it. She paid her copay, scheduled a follow-up for 3 months, and left.
As I’ve written before, I try to avoid all news except the local weather. The sad reality is that most of it is bad and there’s nothing I can really do about it. It only upsets me, which isn’t good for my mental health and blood pressure, and if I can’t change it, what’s the point of knowing? It falls under the serenity prayer.
Of course, some news stories are too big not to hear something. I pass TVs in the doctors lounge or coffee house, hear others talking as I stand in line for the elevator, or see blurbs go by when checking the weather. It’s not entirely unavoidable.
I’m not trivializing the Middle East. But, to me, it’s not part of the doctor-patient relationship any more than my political views are. You run the risk of driving a wedge between you and the person you’re caring for. If you don’t like their opinion, you may find yourself less interested in them and their care. If they don’t like your opinion on news, they may start to question your ability as a doctor.
That’s not what we strive for, but it can be human nature. For better or worse we often reduce things to “us against them,” and learning someone is on the opposite side may, even subconsciously, alter how you treat them.
That’s not good, so to me it’s best not to know.
Some may think I’m being petty, or aloof, to be unwilling to discuss nonmedical issues of significance, but I don’t see it that way. Time is limited at the appointment and is best spent on medical care. Something unrelated to the visit that may alter my objective opinion of a patient – or theirs of me as a doctor – is best left out of it.
I’m here to be your doctor, and to do the best I can for you. I’m not here to be a debate partner. Whenever a patient asks me a question on politics or news I always think of the Monty Python skit “Argument Clinic.” That’s not why you’re here. There are plenty places to discuss such things. My office isn’t one of them.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“What do you think about the whole Israel thing?”
That question came at the end of an otherwise routine appointment.
Maybe she was just chatting. Maybe she wanted something deeper. I have no idea. I just said, “I don’t discuss those things with patients.”
My answer surprised her, but she didn’t push it. She paid her copay, scheduled a follow-up for 3 months, and left.
As I’ve written before, I try to avoid all news except the local weather. The sad reality is that most of it is bad and there’s nothing I can really do about it. It only upsets me, which isn’t good for my mental health and blood pressure, and if I can’t change it, what’s the point of knowing? It falls under the serenity prayer.
Of course, some news stories are too big not to hear something. I pass TVs in the doctors lounge or coffee house, hear others talking as I stand in line for the elevator, or see blurbs go by when checking the weather. It’s not entirely unavoidable.
I’m not trivializing the Middle East. But, to me, it’s not part of the doctor-patient relationship any more than my political views are. You run the risk of driving a wedge between you and the person you’re caring for. If you don’t like their opinion, you may find yourself less interested in them and their care. If they don’t like your opinion on news, they may start to question your ability as a doctor.
That’s not what we strive for, but it can be human nature. For better or worse we often reduce things to “us against them,” and learning someone is on the opposite side may, even subconsciously, alter how you treat them.
That’s not good, so to me it’s best not to know.
Some may think I’m being petty, or aloof, to be unwilling to discuss nonmedical issues of significance, but I don’t see it that way. Time is limited at the appointment and is best spent on medical care. Something unrelated to the visit that may alter my objective opinion of a patient – or theirs of me as a doctor – is best left out of it.
I’m here to be your doctor, and to do the best I can for you. I’m not here to be a debate partner. Whenever a patient asks me a question on politics or news I always think of the Monty Python skit “Argument Clinic.” That’s not why you’re here. There are plenty places to discuss such things. My office isn’t one of them.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“What do you think about the whole Israel thing?”
That question came at the end of an otherwise routine appointment.
Maybe she was just chatting. Maybe she wanted something deeper. I have no idea. I just said, “I don’t discuss those things with patients.”
My answer surprised her, but she didn’t push it. She paid her copay, scheduled a follow-up for 3 months, and left.
As I’ve written before, I try to avoid all news except the local weather. The sad reality is that most of it is bad and there’s nothing I can really do about it. It only upsets me, which isn’t good for my mental health and blood pressure, and if I can’t change it, what’s the point of knowing? It falls under the serenity prayer.
Of course, some news stories are too big not to hear something. I pass TVs in the doctors lounge or coffee house, hear others talking as I stand in line for the elevator, or see blurbs go by when checking the weather. It’s not entirely unavoidable.
I’m not trivializing the Middle East. But, to me, it’s not part of the doctor-patient relationship any more than my political views are. You run the risk of driving a wedge between you and the person you’re caring for. If you don’t like their opinion, you may find yourself less interested in them and their care. If they don’t like your opinion on news, they may start to question your ability as a doctor.
That’s not what we strive for, but it can be human nature. For better or worse we often reduce things to “us against them,” and learning someone is on the opposite side may, even subconsciously, alter how you treat them.
That’s not good, so to me it’s best not to know.
Some may think I’m being petty, or aloof, to be unwilling to discuss nonmedical issues of significance, but I don’t see it that way. Time is limited at the appointment and is best spent on medical care. Something unrelated to the visit that may alter my objective opinion of a patient – or theirs of me as a doctor – is best left out of it.
I’m here to be your doctor, and to do the best I can for you. I’m not here to be a debate partner. Whenever a patient asks me a question on politics or news I always think of the Monty Python skit “Argument Clinic.” That’s not why you’re here. There are plenty places to discuss such things. My office isn’t one of them.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
AI in medicine has a major Cassandra problem
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.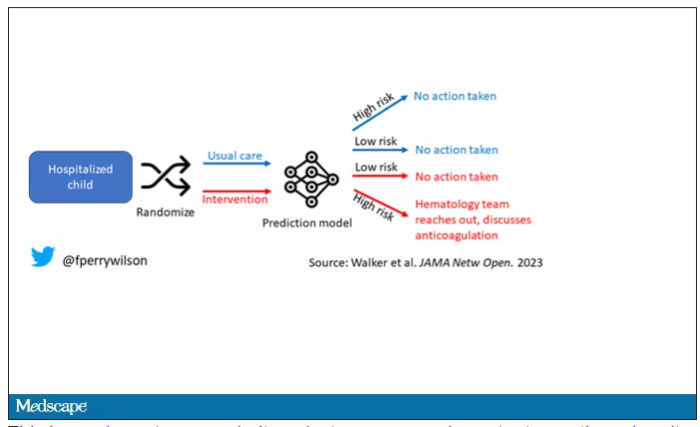
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.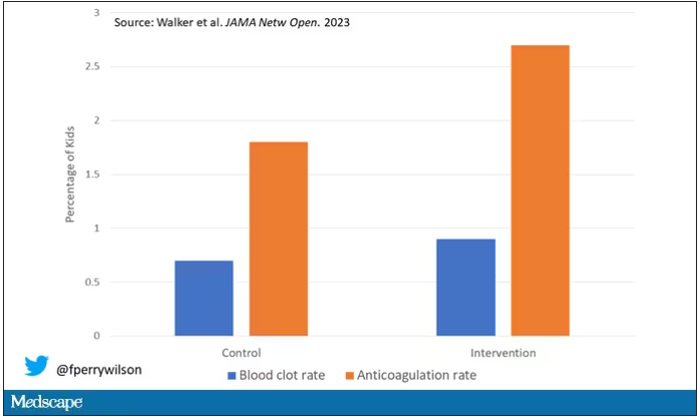
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.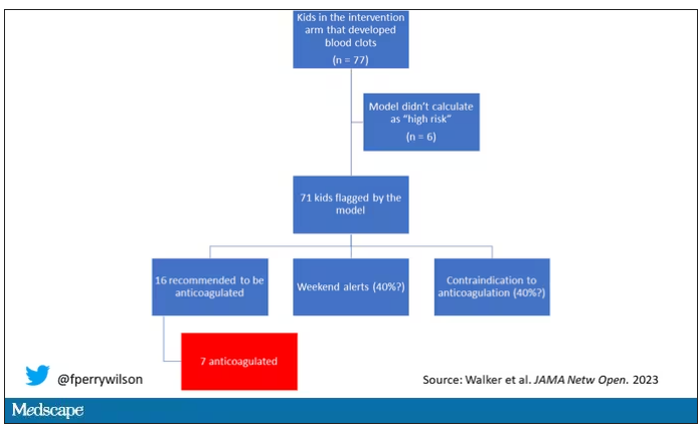
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Debate: Is lasting remission of type 2 diabetes feasible in the real-world setting?
The prospect of remission of type 2 diabetes (T2D) has captured the hearts and minds of many patients with T2D and health care professionals, including myself.
I have changed my narrative when supporting my patients with T2D. I used to say that T2D is a progressive condition, but considering seminal recent evidence like the DiRECT trial, I now say that T2D can be a progressive condition. Through significant weight loss, patients can reverse it and achieve remission of T2D. This has given my patients hope that their T2D is no longer an inexorable condition. And hope, of course, is a powerful enabler of change.
However,
I therefore relished the opportunity to attend a debate on this topic at the annual meeting of the European Association for the Study of Diabetes in Hamburg, Germany, between Roy Taylor, MD, principal investigator for the DiRECT study and professor of medicine and metabolism at the University of Newcastle, England, and Kamlesh Khunti, MD, PhD, professor of primary care diabetes at the University of Leicester, England.
Remarkable weight loss
Dr. Taylor powerfully recapitulated the initial results of the DiRECT study. T2D remission was achieved in 46% of participants who underwent a low-energy formula diet (around 850 calories daily) for 3-5 months. After 2 years’ follow-up, an impressive 36% of participants were still in remission. Dr. Taylor then discussed unpublished 5-year extension follow-up data of the DiRECT study. Average weight loss in the remaining intervention group was 6.1 kg. I echo Taylor’s sentiment that this finding is remarkable in the context of a dietary study.
Overall, 13% of participants were still in remission, and this cohort maintained an average weight loss of 8.9 kg. Dr. Taylor concluded that lasting remission of T2D is indeed feasible in a primary care setting.
Yet he acknowledged that although remission appears feasible in the longer term, it was not necessarily easy, or indeed possible, for everyone. He used a wonderful analogy about climbing Mount Everest: It is feasible, but not everyone can or wants to climb it. And even if you try, you might not reach the top.
This analogy perfectly encapsulates the challenges I have observed when my patients have striven for T2D remission. In my opinion, intensive weight management with a low-energy formula diet is not a panacea for T2D but another tool in our toolbox to offer patients.
He also described some “jaw-dropping” results regarding incidence of cancer: There were no cases of cancer in the intervention group during the 5-year period, but there were eight cases of cancer in the control group. The latter figure is consistent with published data for cancer incidence in patients with T2D and the body mass index (BMI) inclusion criteria for the DiRECT study (a BMI of 27-45 kg/m2). Obesity is an established risk factor for 13 types of cancer, and excess body fat entails an approximately 17% increased risk for cancer-specific mortality. This indeed is a powerful motivator to facilitate meaningful lifestyle change.
In primary care, we also need to be aware that most weight regain usually occurs secondary to a life event (for example, financial, family, or illness). We should reiterate to our patients that weight regain is not a failure; it is just part of life. Once the life event has passed, rapid weight loss can be attempted again. In the “rescue plans” that were integral to the DiRECT study, participants were offered further periods of total diet replacement, depending on quantity of weight gain. In fact, 50% of participants in DiRECT required rescue therapy, and their outcomes, reassuringly, were the same as the other 50%.
Dr. Taylor also quoted data from the ReTUNE study suggesting that weight regain was less of an issue for those with initial BMI of 21-27, and there is “more bang for your buck” in approaching remission of T2D in patients with lower BMI. The fact that people with normal or near-normal BMI can also reverse their T2D was also a game changer for my clinical practice; the concept of an individual or personal fat threshold that results in T2D offers a pragmatic explanation to patients with T2D who are frustrated by the lack of improvements in cardiometabolic parameters despite significant weight loss.
Finally, Dr. Taylor acknowledged the breadth of the definition of T2D remission: A1c < 48 mmol/mol at least 2 months off all antidiabetic medication. This definition includes A1c values within the “prediabetes” range: 42-47 mmol/mol.
He cited 10-year cardiovascular risk data driven by hypertension and dyslipidemia before significant weight loss and compared it with 10-year cardiovascular risk data after significant weight loss. Cardiovascular risk profile was more favorable after weight loss, compared with controls with prediabetes without weight loss, even though some of the intervention group who lost significant weight still had an A1c of 42-47 mmol/mol. Dr. Taylor suggested that we not label these individuals who have lost significant weight as having prediabetes. Instead “postdiabetes” should be preferred, because these patients had more favorable cardiovascular profiles.
This is a very important take-home message for primary care: prediabetes is more than just dysglycemia.
New terminology proposed
Dr. Khunti outlined a recent large, systematic review that concluded that the definition of T2D remission encompassed substantial heterogeneity. This heterogeneity complicates the interpretation of previous research on T2D remission and complicates the implementation of remission pathways into routine clinical practice. Furthermore, Dr. Khunti highlighted a recent consensus report on the definition and interpretation of remission in T2D that explicitly stated that the underlying pathophysiology of T2D is rarely normalized completely by interventions, thus reducing the possibility of lasting remission.
Dr. Khunti also challenged the cardiovascular benefits seen after T2D remission. Recent Danish registry data were presented, demonstrating a twofold increased risk for major adverse cardiovascular events over 5 years in individuals who achieved remission of T2D, but not on glucose-lowering drug therapy.
Adherence to strict dietary interventions in the longer term was also addressed. Diet-induced weight loss causes changes in circulating hormones such as ghrelin, glucose-dependent insulinotropic polypeptide (GIP), and leptin, which mediate appetite and drive hunger and an increased preference for energy-dense foods (that is, high-fat or sugary foods), all of which encourage weight regain. Dr. Khunti suggested that other interventions, such as glucagon-like peptide 1 (GLP-1) receptor agonists or bariatric surgery, specifically target some of these hormonal responses.
The challenges in recruitment and retention for lifestyle studies were also discussed; they reflect the challenges of behavioral programs in primary care. The DiRECT study had 20% participation of screened candidates and an attrition rate approaching 30%. The seminal Diabetes Prevention Program study and Finnish Diabetes Prevention Study had similar results. At a population level, individuals do not appear to want to participate in behavioral programs.
Dr. Khunti also warned that the review of annual care processes for diabetes is declining for patients who had achieved remission, possibly because of a false sense of reassurance among health care professionals. It is essential that all those in remission remain under at least annual follow-up, because there is still a risk for future microvascular and macrovascular complications, especially in the event of weight regain.
Dr. Khunti concluded by proposing new terminology for remission: remission of hyperglycemia or euglycemia, aiming for A1c < 48 mmol/mol with or without glucose-lowering therapy. I do agree with this; it reflects the zeitgeist of cardiorenal protective diabetes therapies and is analogous to rheumatoid arthritis, where remission is defined as no disease activity while on therapy. But one size does not fit all.
Sir William Osler’s words provide a fitting conclusion: “If it were not for the great variability among individuals, medicine might as well be a science and not an art.”
Dr. Fernando has disclosed that he has received speakers’ fees from Eli Lilly and Novo Nordisk.
Dr. Fernando is a general practitioner near Edinburgh, with a specialist interest in diabetes; cardiovascular, renal, and metabolic diseases; and medical education.
A version of this article first appeared on Medscape.com.
The prospect of remission of type 2 diabetes (T2D) has captured the hearts and minds of many patients with T2D and health care professionals, including myself.
I have changed my narrative when supporting my patients with T2D. I used to say that T2D is a progressive condition, but considering seminal recent evidence like the DiRECT trial, I now say that T2D can be a progressive condition. Through significant weight loss, patients can reverse it and achieve remission of T2D. This has given my patients hope that their T2D is no longer an inexorable condition. And hope, of course, is a powerful enabler of change.
However,
I therefore relished the opportunity to attend a debate on this topic at the annual meeting of the European Association for the Study of Diabetes in Hamburg, Germany, between Roy Taylor, MD, principal investigator for the DiRECT study and professor of medicine and metabolism at the University of Newcastle, England, and Kamlesh Khunti, MD, PhD, professor of primary care diabetes at the University of Leicester, England.
Remarkable weight loss
Dr. Taylor powerfully recapitulated the initial results of the DiRECT study. T2D remission was achieved in 46% of participants who underwent a low-energy formula diet (around 850 calories daily) for 3-5 months. After 2 years’ follow-up, an impressive 36% of participants were still in remission. Dr. Taylor then discussed unpublished 5-year extension follow-up data of the DiRECT study. Average weight loss in the remaining intervention group was 6.1 kg. I echo Taylor’s sentiment that this finding is remarkable in the context of a dietary study.
Overall, 13% of participants were still in remission, and this cohort maintained an average weight loss of 8.9 kg. Dr. Taylor concluded that lasting remission of T2D is indeed feasible in a primary care setting.
Yet he acknowledged that although remission appears feasible in the longer term, it was not necessarily easy, or indeed possible, for everyone. He used a wonderful analogy about climbing Mount Everest: It is feasible, but not everyone can or wants to climb it. And even if you try, you might not reach the top.
This analogy perfectly encapsulates the challenges I have observed when my patients have striven for T2D remission. In my opinion, intensive weight management with a low-energy formula diet is not a panacea for T2D but another tool in our toolbox to offer patients.
He also described some “jaw-dropping” results regarding incidence of cancer: There were no cases of cancer in the intervention group during the 5-year period, but there were eight cases of cancer in the control group. The latter figure is consistent with published data for cancer incidence in patients with T2D and the body mass index (BMI) inclusion criteria for the DiRECT study (a BMI of 27-45 kg/m2). Obesity is an established risk factor for 13 types of cancer, and excess body fat entails an approximately 17% increased risk for cancer-specific mortality. This indeed is a powerful motivator to facilitate meaningful lifestyle change.
In primary care, we also need to be aware that most weight regain usually occurs secondary to a life event (for example, financial, family, or illness). We should reiterate to our patients that weight regain is not a failure; it is just part of life. Once the life event has passed, rapid weight loss can be attempted again. In the “rescue plans” that were integral to the DiRECT study, participants were offered further periods of total diet replacement, depending on quantity of weight gain. In fact, 50% of participants in DiRECT required rescue therapy, and their outcomes, reassuringly, were the same as the other 50%.
Dr. Taylor also quoted data from the ReTUNE study suggesting that weight regain was less of an issue for those with initial BMI of 21-27, and there is “more bang for your buck” in approaching remission of T2D in patients with lower BMI. The fact that people with normal or near-normal BMI can also reverse their T2D was also a game changer for my clinical practice; the concept of an individual or personal fat threshold that results in T2D offers a pragmatic explanation to patients with T2D who are frustrated by the lack of improvements in cardiometabolic parameters despite significant weight loss.
Finally, Dr. Taylor acknowledged the breadth of the definition of T2D remission: A1c < 48 mmol/mol at least 2 months off all antidiabetic medication. This definition includes A1c values within the “prediabetes” range: 42-47 mmol/mol.
He cited 10-year cardiovascular risk data driven by hypertension and dyslipidemia before significant weight loss and compared it with 10-year cardiovascular risk data after significant weight loss. Cardiovascular risk profile was more favorable after weight loss, compared with controls with prediabetes without weight loss, even though some of the intervention group who lost significant weight still had an A1c of 42-47 mmol/mol. Dr. Taylor suggested that we not label these individuals who have lost significant weight as having prediabetes. Instead “postdiabetes” should be preferred, because these patients had more favorable cardiovascular profiles.
This is a very important take-home message for primary care: prediabetes is more than just dysglycemia.
New terminology proposed
Dr. Khunti outlined a recent large, systematic review that concluded that the definition of T2D remission encompassed substantial heterogeneity. This heterogeneity complicates the interpretation of previous research on T2D remission and complicates the implementation of remission pathways into routine clinical practice. Furthermore, Dr. Khunti highlighted a recent consensus report on the definition and interpretation of remission in T2D that explicitly stated that the underlying pathophysiology of T2D is rarely normalized completely by interventions, thus reducing the possibility of lasting remission.
Dr. Khunti also challenged the cardiovascular benefits seen after T2D remission. Recent Danish registry data were presented, demonstrating a twofold increased risk for major adverse cardiovascular events over 5 years in individuals who achieved remission of T2D, but not on glucose-lowering drug therapy.
Adherence to strict dietary interventions in the longer term was also addressed. Diet-induced weight loss causes changes in circulating hormones such as ghrelin, glucose-dependent insulinotropic polypeptide (GIP), and leptin, which mediate appetite and drive hunger and an increased preference for energy-dense foods (that is, high-fat or sugary foods), all of which encourage weight regain. Dr. Khunti suggested that other interventions, such as glucagon-like peptide 1 (GLP-1) receptor agonists or bariatric surgery, specifically target some of these hormonal responses.
The challenges in recruitment and retention for lifestyle studies were also discussed; they reflect the challenges of behavioral programs in primary care. The DiRECT study had 20% participation of screened candidates and an attrition rate approaching 30%. The seminal Diabetes Prevention Program study and Finnish Diabetes Prevention Study had similar results. At a population level, individuals do not appear to want to participate in behavioral programs.
Dr. Khunti also warned that the review of annual care processes for diabetes is declining for patients who had achieved remission, possibly because of a false sense of reassurance among health care professionals. It is essential that all those in remission remain under at least annual follow-up, because there is still a risk for future microvascular and macrovascular complications, especially in the event of weight regain.
Dr. Khunti concluded by proposing new terminology for remission: remission of hyperglycemia or euglycemia, aiming for A1c < 48 mmol/mol with or without glucose-lowering therapy. I do agree with this; it reflects the zeitgeist of cardiorenal protective diabetes therapies and is analogous to rheumatoid arthritis, where remission is defined as no disease activity while on therapy. But one size does not fit all.
Sir William Osler’s words provide a fitting conclusion: “If it were not for the great variability among individuals, medicine might as well be a science and not an art.”
Dr. Fernando has disclosed that he has received speakers’ fees from Eli Lilly and Novo Nordisk.
Dr. Fernando is a general practitioner near Edinburgh, with a specialist interest in diabetes; cardiovascular, renal, and metabolic diseases; and medical education.
A version of this article first appeared on Medscape.com.
The prospect of remission of type 2 diabetes (T2D) has captured the hearts and minds of many patients with T2D and health care professionals, including myself.
I have changed my narrative when supporting my patients with T2D. I used to say that T2D is a progressive condition, but considering seminal recent evidence like the DiRECT trial, I now say that T2D can be a progressive condition. Through significant weight loss, patients can reverse it and achieve remission of T2D. This has given my patients hope that their T2D is no longer an inexorable condition. And hope, of course, is a powerful enabler of change.
However,
I therefore relished the opportunity to attend a debate on this topic at the annual meeting of the European Association for the Study of Diabetes in Hamburg, Germany, between Roy Taylor, MD, principal investigator for the DiRECT study and professor of medicine and metabolism at the University of Newcastle, England, and Kamlesh Khunti, MD, PhD, professor of primary care diabetes at the University of Leicester, England.
Remarkable weight loss
Dr. Taylor powerfully recapitulated the initial results of the DiRECT study. T2D remission was achieved in 46% of participants who underwent a low-energy formula diet (around 850 calories daily) for 3-5 months. After 2 years’ follow-up, an impressive 36% of participants were still in remission. Dr. Taylor then discussed unpublished 5-year extension follow-up data of the DiRECT study. Average weight loss in the remaining intervention group was 6.1 kg. I echo Taylor’s sentiment that this finding is remarkable in the context of a dietary study.
Overall, 13% of participants were still in remission, and this cohort maintained an average weight loss of 8.9 kg. Dr. Taylor concluded that lasting remission of T2D is indeed feasible in a primary care setting.
Yet he acknowledged that although remission appears feasible in the longer term, it was not necessarily easy, or indeed possible, for everyone. He used a wonderful analogy about climbing Mount Everest: It is feasible, but not everyone can or wants to climb it. And even if you try, you might not reach the top.
This analogy perfectly encapsulates the challenges I have observed when my patients have striven for T2D remission. In my opinion, intensive weight management with a low-energy formula diet is not a panacea for T2D but another tool in our toolbox to offer patients.
He also described some “jaw-dropping” results regarding incidence of cancer: There were no cases of cancer in the intervention group during the 5-year period, but there were eight cases of cancer in the control group. The latter figure is consistent with published data for cancer incidence in patients with T2D and the body mass index (BMI) inclusion criteria for the DiRECT study (a BMI of 27-45 kg/m2). Obesity is an established risk factor for 13 types of cancer, and excess body fat entails an approximately 17% increased risk for cancer-specific mortality. This indeed is a powerful motivator to facilitate meaningful lifestyle change.
In primary care, we also need to be aware that most weight regain usually occurs secondary to a life event (for example, financial, family, or illness). We should reiterate to our patients that weight regain is not a failure; it is just part of life. Once the life event has passed, rapid weight loss can be attempted again. In the “rescue plans” that were integral to the DiRECT study, participants were offered further periods of total diet replacement, depending on quantity of weight gain. In fact, 50% of participants in DiRECT required rescue therapy, and their outcomes, reassuringly, were the same as the other 50%.
Dr. Taylor also quoted data from the ReTUNE study suggesting that weight regain was less of an issue for those with initial BMI of 21-27, and there is “more bang for your buck” in approaching remission of T2D in patients with lower BMI. The fact that people with normal or near-normal BMI can also reverse their T2D was also a game changer for my clinical practice; the concept of an individual or personal fat threshold that results in T2D offers a pragmatic explanation to patients with T2D who are frustrated by the lack of improvements in cardiometabolic parameters despite significant weight loss.
Finally, Dr. Taylor acknowledged the breadth of the definition of T2D remission: A1c < 48 mmol/mol at least 2 months off all antidiabetic medication. This definition includes A1c values within the “prediabetes” range: 42-47 mmol/mol.
He cited 10-year cardiovascular risk data driven by hypertension and dyslipidemia before significant weight loss and compared it with 10-year cardiovascular risk data after significant weight loss. Cardiovascular risk profile was more favorable after weight loss, compared with controls with prediabetes without weight loss, even though some of the intervention group who lost significant weight still had an A1c of 42-47 mmol/mol. Dr. Taylor suggested that we not label these individuals who have lost significant weight as having prediabetes. Instead “postdiabetes” should be preferred, because these patients had more favorable cardiovascular profiles.
This is a very important take-home message for primary care: prediabetes is more than just dysglycemia.
New terminology proposed
Dr. Khunti outlined a recent large, systematic review that concluded that the definition of T2D remission encompassed substantial heterogeneity. This heterogeneity complicates the interpretation of previous research on T2D remission and complicates the implementation of remission pathways into routine clinical practice. Furthermore, Dr. Khunti highlighted a recent consensus report on the definition and interpretation of remission in T2D that explicitly stated that the underlying pathophysiology of T2D is rarely normalized completely by interventions, thus reducing the possibility of lasting remission.
Dr. Khunti also challenged the cardiovascular benefits seen after T2D remission. Recent Danish registry data were presented, demonstrating a twofold increased risk for major adverse cardiovascular events over 5 years in individuals who achieved remission of T2D, but not on glucose-lowering drug therapy.
Adherence to strict dietary interventions in the longer term was also addressed. Diet-induced weight loss causes changes in circulating hormones such as ghrelin, glucose-dependent insulinotropic polypeptide (GIP), and leptin, which mediate appetite and drive hunger and an increased preference for energy-dense foods (that is, high-fat or sugary foods), all of which encourage weight regain. Dr. Khunti suggested that other interventions, such as glucagon-like peptide 1 (GLP-1) receptor agonists or bariatric surgery, specifically target some of these hormonal responses.
The challenges in recruitment and retention for lifestyle studies were also discussed; they reflect the challenges of behavioral programs in primary care. The DiRECT study had 20% participation of screened candidates and an attrition rate approaching 30%. The seminal Diabetes Prevention Program study and Finnish Diabetes Prevention Study had similar results. At a population level, individuals do not appear to want to participate in behavioral programs.
Dr. Khunti also warned that the review of annual care processes for diabetes is declining for patients who had achieved remission, possibly because of a false sense of reassurance among health care professionals. It is essential that all those in remission remain under at least annual follow-up, because there is still a risk for future microvascular and macrovascular complications, especially in the event of weight regain.
Dr. Khunti concluded by proposing new terminology for remission: remission of hyperglycemia or euglycemia, aiming for A1c < 48 mmol/mol with or without glucose-lowering therapy. I do agree with this; it reflects the zeitgeist of cardiorenal protective diabetes therapies and is analogous to rheumatoid arthritis, where remission is defined as no disease activity while on therapy. But one size does not fit all.
Sir William Osler’s words provide a fitting conclusion: “If it were not for the great variability among individuals, medicine might as well be a science and not an art.”
Dr. Fernando has disclosed that he has received speakers’ fees from Eli Lilly and Novo Nordisk.
Dr. Fernando is a general practitioner near Edinburgh, with a specialist interest in diabetes; cardiovascular, renal, and metabolic diseases; and medical education.
A version of this article first appeared on Medscape.com.
Making time to care for patients with diabetes
Can busy primary care offices continue to care for patients with diabetes? No one would argue that it is involved and takes effort, and health care providers are bankrupt when it comes to sparing additional time for this chronic disease. With roughly 37 million people living with diabetes and 96 million with prediabetes or early type 2 diabetes, and just over 8,000 practicing endocrinologists in the United States, we all need to make time especially in primary care to provide insight and holistic care. With limited time and budget, how do we do this?
First, decide to be involved in caring for patients with diabetes. Diabetes is best managed by interprofessional care teams, so you’re not going it alone. These teams may include physicians; pharmacists; physician assistants; advanced practice nurses; registered nurses; certified diabetes care and education specialists (CDCES); dietitians; and other professionals such as social workers, behavioral health professionals, medical assistants, and community health workers. Know which professionals are available to serve on your team, either within your clinic or as a consultant, and reach out to them to share the care and ease the burden. Remember to refer to these professionals to reinforce the diabetes intervention message to the patient.
Second, incorporate “diabetes only” appointments into your schedule, allowing time to focus on current comprehensive diabetes treatment goals, barriers/inertia for care. Remember to have short-interval follow-up as needed to keep that patient engaged to achieve their targets. Instruct your office staff to create diabetes appointment templates and reminders to patients to bring diabetes-related technologies, medication lists, and diabetes questions to the appointment. When I implemented this change, my patients welcomed the focus on their diabetes health, and they knew we were prioritizing this disease that they have for a lifetime. These appointments did not take away from their other conditions; rather, they often reminded me to stay focused on their diabetes and associated coconditions.
Taking the time to establish efficient workflows before implementing diabetes care saves countless hours later and immediately maximizes health care provider–patient interactions. Assign specific staff duties and expectations related to diabetes appointments, such as downloading diabetes technology, medication reconciliation, laboratory data, point-of-care hemoglobin A1c, basic foot exam, and patient goals for diabetes care. This allows the prescriber to focus on the glycemic, cardiologic, renal, and metabolic goals and overcome the therapeutic inertia that plagues us all.
Incorporating diabetes-related technology into clinical practice can be a significant time-saver but requires initial onboarding. Set aside a few hours to create a technology clinic flow, and designate at least one team member to be responsible for obtaining patient data before, during, or after encounters. If possible, obtaining data ahead of the visit will enhance efficiency, allowing for meaningful discussion of blood glucose and lifestyle patterns. Diabetes technology reveals the gaps in care and enhances our ability to identify the areas where glycemic intervention is needed. In addition, it reveals the impact of food choices, activity level, stress, and medication adherence to the person living with diabetes.
Finally, be proactive about therapeutic inertia. This is defined as a prescribers’ failure to intensify or deintensify a patient’s treatment when appropriate to do so. Causes of therapeutic inertia can be placed at the primary care physician level, including time constraints or inexperience in treating diabetes; the patient level, such as concerns about side effects or new treatment regimens; or a systemic level, such as availability of medications or their costs. Be real with yourself: We all have inertia and can identify areas to overcome. Never let inertia be traced back to you.
Not all inertia lives with the health care provider. Patients bring apprehension and concerns, have questions, and just want to share the frustrations associated with living their best life with the disease. Don’t assume that you know what your patients’ treatment barriers are; ask them. If you don’t have an answer, then note it and come up with one by the next follow-up. Remember that this is a chronic disease – a marathon, not a sprint. You don’t have to solve everything at one appointment; rather, keep the momentum going.
Let’s put this into clinical practice. For the next patient with diabetes who comes into your office, discuss with them your intention to prioritize their diabetes by having an appointment set aside to specifically focus on their individual goals and targets for their disease. Have the patient list any barriers and treatment goals they would like to review; flag your schedule to indicate it is a diabetes-only visit; and orient your staff to reconcile diabetes medications and record the patient’s last eye exam, urine albumin-to-creatinine ratio, A1c result, and blood glucose data. During this encounter, identify the patient’s personal targets for control, examine their feet, and review or order necessary laboratory metrics. Explore the patient-reported barriers and make inroads to remove or alleviate these. Advance treatment intervention, and schedule follow-up: every 4-6 weeks if the A1c is > 9%, every 2 months if it’s 7% to < 9%, and every 3-6 months if it’s < 7%. Utilize team diabetes care, such as CDCES referrals, dietitians, online resources, and community members, to help reinforce care and enhance engagement.
We need to take steps in our clinical practice to make the necessary space to accommodate this pervasive disease affecting nearly one-third of our population. Take a moment to look up and determine what needs to be in place so that you can take care of the people in your practice with diabetes. Laying the groundwork for implementing diabetes-only appointments can be time-consuming, but establishing consistent procedures, developing efficient workflows, and clearly defining roles and responsibilities is well worth the effort. This solid foundation equips the office, health care providers, and staff to care for persons with diabetes and will be invaluable to ensure that time for this care is available in the day-to-day clinical practice.
A version of this article first appeared on Medscape.com.
Can busy primary care offices continue to care for patients with diabetes? No one would argue that it is involved and takes effort, and health care providers are bankrupt when it comes to sparing additional time for this chronic disease. With roughly 37 million people living with diabetes and 96 million with prediabetes or early type 2 diabetes, and just over 8,000 practicing endocrinologists in the United States, we all need to make time especially in primary care to provide insight and holistic care. With limited time and budget, how do we do this?
First, decide to be involved in caring for patients with diabetes. Diabetes is best managed by interprofessional care teams, so you’re not going it alone. These teams may include physicians; pharmacists; physician assistants; advanced practice nurses; registered nurses; certified diabetes care and education specialists (CDCES); dietitians; and other professionals such as social workers, behavioral health professionals, medical assistants, and community health workers. Know which professionals are available to serve on your team, either within your clinic or as a consultant, and reach out to them to share the care and ease the burden. Remember to refer to these professionals to reinforce the diabetes intervention message to the patient.
Second, incorporate “diabetes only” appointments into your schedule, allowing time to focus on current comprehensive diabetes treatment goals, barriers/inertia for care. Remember to have short-interval follow-up as needed to keep that patient engaged to achieve their targets. Instruct your office staff to create diabetes appointment templates and reminders to patients to bring diabetes-related technologies, medication lists, and diabetes questions to the appointment. When I implemented this change, my patients welcomed the focus on their diabetes health, and they knew we were prioritizing this disease that they have for a lifetime. These appointments did not take away from their other conditions; rather, they often reminded me to stay focused on their diabetes and associated coconditions.
Taking the time to establish efficient workflows before implementing diabetes care saves countless hours later and immediately maximizes health care provider–patient interactions. Assign specific staff duties and expectations related to diabetes appointments, such as downloading diabetes technology, medication reconciliation, laboratory data, point-of-care hemoglobin A1c, basic foot exam, and patient goals for diabetes care. This allows the prescriber to focus on the glycemic, cardiologic, renal, and metabolic goals and overcome the therapeutic inertia that plagues us all.
Incorporating diabetes-related technology into clinical practice can be a significant time-saver but requires initial onboarding. Set aside a few hours to create a technology clinic flow, and designate at least one team member to be responsible for obtaining patient data before, during, or after encounters. If possible, obtaining data ahead of the visit will enhance efficiency, allowing for meaningful discussion of blood glucose and lifestyle patterns. Diabetes technology reveals the gaps in care and enhances our ability to identify the areas where glycemic intervention is needed. In addition, it reveals the impact of food choices, activity level, stress, and medication adherence to the person living with diabetes.
Finally, be proactive about therapeutic inertia. This is defined as a prescribers’ failure to intensify or deintensify a patient’s treatment when appropriate to do so. Causes of therapeutic inertia can be placed at the primary care physician level, including time constraints or inexperience in treating diabetes; the patient level, such as concerns about side effects or new treatment regimens; or a systemic level, such as availability of medications or their costs. Be real with yourself: We all have inertia and can identify areas to overcome. Never let inertia be traced back to you.
Not all inertia lives with the health care provider. Patients bring apprehension and concerns, have questions, and just want to share the frustrations associated with living their best life with the disease. Don’t assume that you know what your patients’ treatment barriers are; ask them. If you don’t have an answer, then note it and come up with one by the next follow-up. Remember that this is a chronic disease – a marathon, not a sprint. You don’t have to solve everything at one appointment; rather, keep the momentum going.
Let’s put this into clinical practice. For the next patient with diabetes who comes into your office, discuss with them your intention to prioritize their diabetes by having an appointment set aside to specifically focus on their individual goals and targets for their disease. Have the patient list any barriers and treatment goals they would like to review; flag your schedule to indicate it is a diabetes-only visit; and orient your staff to reconcile diabetes medications and record the patient’s last eye exam, urine albumin-to-creatinine ratio, A1c result, and blood glucose data. During this encounter, identify the patient’s personal targets for control, examine their feet, and review or order necessary laboratory metrics. Explore the patient-reported barriers and make inroads to remove or alleviate these. Advance treatment intervention, and schedule follow-up: every 4-6 weeks if the A1c is > 9%, every 2 months if it’s 7% to < 9%, and every 3-6 months if it’s < 7%. Utilize team diabetes care, such as CDCES referrals, dietitians, online resources, and community members, to help reinforce care and enhance engagement.
We need to take steps in our clinical practice to make the necessary space to accommodate this pervasive disease affecting nearly one-third of our population. Take a moment to look up and determine what needs to be in place so that you can take care of the people in your practice with diabetes. Laying the groundwork for implementing diabetes-only appointments can be time-consuming, but establishing consistent procedures, developing efficient workflows, and clearly defining roles and responsibilities is well worth the effort. This solid foundation equips the office, health care providers, and staff to care for persons with diabetes and will be invaluable to ensure that time for this care is available in the day-to-day clinical practice.
A version of this article first appeared on Medscape.com.
Can busy primary care offices continue to care for patients with diabetes? No one would argue that it is involved and takes effort, and health care providers are bankrupt when it comes to sparing additional time for this chronic disease. With roughly 37 million people living with diabetes and 96 million with prediabetes or early type 2 diabetes, and just over 8,000 practicing endocrinologists in the United States, we all need to make time especially in primary care to provide insight and holistic care. With limited time and budget, how do we do this?
First, decide to be involved in caring for patients with diabetes. Diabetes is best managed by interprofessional care teams, so you’re not going it alone. These teams may include physicians; pharmacists; physician assistants; advanced practice nurses; registered nurses; certified diabetes care and education specialists (CDCES); dietitians; and other professionals such as social workers, behavioral health professionals, medical assistants, and community health workers. Know which professionals are available to serve on your team, either within your clinic or as a consultant, and reach out to them to share the care and ease the burden. Remember to refer to these professionals to reinforce the diabetes intervention message to the patient.
Second, incorporate “diabetes only” appointments into your schedule, allowing time to focus on current comprehensive diabetes treatment goals, barriers/inertia for care. Remember to have short-interval follow-up as needed to keep that patient engaged to achieve their targets. Instruct your office staff to create diabetes appointment templates and reminders to patients to bring diabetes-related technologies, medication lists, and diabetes questions to the appointment. When I implemented this change, my patients welcomed the focus on their diabetes health, and they knew we were prioritizing this disease that they have for a lifetime. These appointments did not take away from their other conditions; rather, they often reminded me to stay focused on their diabetes and associated coconditions.
Taking the time to establish efficient workflows before implementing diabetes care saves countless hours later and immediately maximizes health care provider–patient interactions. Assign specific staff duties and expectations related to diabetes appointments, such as downloading diabetes technology, medication reconciliation, laboratory data, point-of-care hemoglobin A1c, basic foot exam, and patient goals for diabetes care. This allows the prescriber to focus on the glycemic, cardiologic, renal, and metabolic goals and overcome the therapeutic inertia that plagues us all.
Incorporating diabetes-related technology into clinical practice can be a significant time-saver but requires initial onboarding. Set aside a few hours to create a technology clinic flow, and designate at least one team member to be responsible for obtaining patient data before, during, or after encounters. If possible, obtaining data ahead of the visit will enhance efficiency, allowing for meaningful discussion of blood glucose and lifestyle patterns. Diabetes technology reveals the gaps in care and enhances our ability to identify the areas where glycemic intervention is needed. In addition, it reveals the impact of food choices, activity level, stress, and medication adherence to the person living with diabetes.
Finally, be proactive about therapeutic inertia. This is defined as a prescribers’ failure to intensify or deintensify a patient’s treatment when appropriate to do so. Causes of therapeutic inertia can be placed at the primary care physician level, including time constraints or inexperience in treating diabetes; the patient level, such as concerns about side effects or new treatment regimens; or a systemic level, such as availability of medications or their costs. Be real with yourself: We all have inertia and can identify areas to overcome. Never let inertia be traced back to you.
Not all inertia lives with the health care provider. Patients bring apprehension and concerns, have questions, and just want to share the frustrations associated with living their best life with the disease. Don’t assume that you know what your patients’ treatment barriers are; ask them. If you don’t have an answer, then note it and come up with one by the next follow-up. Remember that this is a chronic disease – a marathon, not a sprint. You don’t have to solve everything at one appointment; rather, keep the momentum going.
Let’s put this into clinical practice. For the next patient with diabetes who comes into your office, discuss with them your intention to prioritize their diabetes by having an appointment set aside to specifically focus on their individual goals and targets for their disease. Have the patient list any barriers and treatment goals they would like to review; flag your schedule to indicate it is a diabetes-only visit; and orient your staff to reconcile diabetes medications and record the patient’s last eye exam, urine albumin-to-creatinine ratio, A1c result, and blood glucose data. During this encounter, identify the patient’s personal targets for control, examine their feet, and review or order necessary laboratory metrics. Explore the patient-reported barriers and make inroads to remove or alleviate these. Advance treatment intervention, and schedule follow-up: every 4-6 weeks if the A1c is > 9%, every 2 months if it’s 7% to < 9%, and every 3-6 months if it’s < 7%. Utilize team diabetes care, such as CDCES referrals, dietitians, online resources, and community members, to help reinforce care and enhance engagement.
We need to take steps in our clinical practice to make the necessary space to accommodate this pervasive disease affecting nearly one-third of our population. Take a moment to look up and determine what needs to be in place so that you can take care of the people in your practice with diabetes. Laying the groundwork for implementing diabetes-only appointments can be time-consuming, but establishing consistent procedures, developing efficient workflows, and clearly defining roles and responsibilities is well worth the effort. This solid foundation equips the office, health care providers, and staff to care for persons with diabetes and will be invaluable to ensure that time for this care is available in the day-to-day clinical practice.
A version of this article first appeared on Medscape.com.



