User login
Online reviews
The last time I gave the talk, "Help! I’ve Been Yelped!" to physicians, there was a full house, a sometimes defiant, sometimes incredulous but always engaged full house. Most physicians don’t like Yelp and other online doctor rating sites because of the potential for negative reviews.
In past columns, I’ve written about these sites and how to respond to negative reviews and comments. Now, I’m going to share data on the use of online reviews and why they are important.
We live in a digital world that values reviews. We compare hotels on TripAdvisor.com before booking them and read reviews on Amazon.com before ordering products. We "like" or "dislike" Facebook pages and give thumbs up or thumbs down to videos on YouTube. We even rate physicians’ comments on medical question-and-answer sites such as HealthTap.com.
But how much do all of these online ratings really matter? A 2012 Nielsen report that surveyed more than 28,000 Internet users in 56 countries found that online consumer reviews are the second-most-trusted source of brand information, following only recommendations from family and friends. In other words, we trust online reviews and use them to make our own decisions.
The same is true when it comes to shopping for a doctor. According to an Internet-based survey of 2,137 adults published in the February issue of JAMA, 59% of respondents said that online doctor ratings were either "somewhat important" or "very important" when choosing a physician (2014;11:734-5).
Similarly, the "2013 Industry View Report" by Software Advice found that 62% of respondents said they read online reviews when seeking a new doctor. Although HealthGrades.com was the most commonly used site, Yelp.com was the most trusted. Forty-four percent of those respondents considered Yelp the most trustworthy review site followed by Health Grades (31%), Vitals.com (17%), and ZocDoc.com (7%).
Whether or not we trust Yelp and other online review sites, our patients do. In the JAMA survey, 35% of respondents said that they selected a physician based on good ratings, while 37% said that they avoided a physician with negative reviews. The 2013 Industry View Report also found that 45% of respondents ranked "quality of care" as the most important type of information sought about a doctor. And since many patients equate service with quality, reviews that focus on service matter.
This isn’t an entirely bad thing. If we really listen to what patients are saying, their comments can help us to improve service and communication. And, in some instances, it can lead to stronger doctor-patient relationships. Like many other industries, health care is moving toward transparency, and doctor rating sites are a key component of that.
Dr. Jeffrey Benabio is a partner physician in the department of dermatology of the Southern California Permanente Group in San Diego and a volunteer clinical assistant professor at the University of California, San Diego. He has published numerous scientific articles and is a member and fellow of the American Academy of Dermatology, and a member of the Telemedicine Association and the American Medical Association, among others. He is board certified in dermatology as well as medicine and surgery in the state of California. Dr. Benabio has a special interest in the uses of social media for education and building dermatology practice. He is the founder of The Derm Blog, an educational website that has had more than 2 million unique visitors. Dr. Benabio is also a founding member and the skin care expert for Livestrong.com, a health and wellness website of Lance Armstrong’s the Livestrong Foundation. Dr. Benabio is @Dermdoc on Twitter.
The last time I gave the talk, "Help! I’ve Been Yelped!" to physicians, there was a full house, a sometimes defiant, sometimes incredulous but always engaged full house. Most physicians don’t like Yelp and other online doctor rating sites because of the potential for negative reviews.
In past columns, I’ve written about these sites and how to respond to negative reviews and comments. Now, I’m going to share data on the use of online reviews and why they are important.
We live in a digital world that values reviews. We compare hotels on TripAdvisor.com before booking them and read reviews on Amazon.com before ordering products. We "like" or "dislike" Facebook pages and give thumbs up or thumbs down to videos on YouTube. We even rate physicians’ comments on medical question-and-answer sites such as HealthTap.com.
But how much do all of these online ratings really matter? A 2012 Nielsen report that surveyed more than 28,000 Internet users in 56 countries found that online consumer reviews are the second-most-trusted source of brand information, following only recommendations from family and friends. In other words, we trust online reviews and use them to make our own decisions.
The same is true when it comes to shopping for a doctor. According to an Internet-based survey of 2,137 adults published in the February issue of JAMA, 59% of respondents said that online doctor ratings were either "somewhat important" or "very important" when choosing a physician (2014;11:734-5).
Similarly, the "2013 Industry View Report" by Software Advice found that 62% of respondents said they read online reviews when seeking a new doctor. Although HealthGrades.com was the most commonly used site, Yelp.com was the most trusted. Forty-four percent of those respondents considered Yelp the most trustworthy review site followed by Health Grades (31%), Vitals.com (17%), and ZocDoc.com (7%).
Whether or not we trust Yelp and other online review sites, our patients do. In the JAMA survey, 35% of respondents said that they selected a physician based on good ratings, while 37% said that they avoided a physician with negative reviews. The 2013 Industry View Report also found that 45% of respondents ranked "quality of care" as the most important type of information sought about a doctor. And since many patients equate service with quality, reviews that focus on service matter.
This isn’t an entirely bad thing. If we really listen to what patients are saying, their comments can help us to improve service and communication. And, in some instances, it can lead to stronger doctor-patient relationships. Like many other industries, health care is moving toward transparency, and doctor rating sites are a key component of that.
Dr. Jeffrey Benabio is a partner physician in the department of dermatology of the Southern California Permanente Group in San Diego and a volunteer clinical assistant professor at the University of California, San Diego. He has published numerous scientific articles and is a member and fellow of the American Academy of Dermatology, and a member of the Telemedicine Association and the American Medical Association, among others. He is board certified in dermatology as well as medicine and surgery in the state of California. Dr. Benabio has a special interest in the uses of social media for education and building dermatology practice. He is the founder of The Derm Blog, an educational website that has had more than 2 million unique visitors. Dr. Benabio is also a founding member and the skin care expert for Livestrong.com, a health and wellness website of Lance Armstrong’s the Livestrong Foundation. Dr. Benabio is @Dermdoc on Twitter.
The last time I gave the talk, "Help! I’ve Been Yelped!" to physicians, there was a full house, a sometimes defiant, sometimes incredulous but always engaged full house. Most physicians don’t like Yelp and other online doctor rating sites because of the potential for negative reviews.
In past columns, I’ve written about these sites and how to respond to negative reviews and comments. Now, I’m going to share data on the use of online reviews and why they are important.
We live in a digital world that values reviews. We compare hotels on TripAdvisor.com before booking them and read reviews on Amazon.com before ordering products. We "like" or "dislike" Facebook pages and give thumbs up or thumbs down to videos on YouTube. We even rate physicians’ comments on medical question-and-answer sites such as HealthTap.com.
But how much do all of these online ratings really matter? A 2012 Nielsen report that surveyed more than 28,000 Internet users in 56 countries found that online consumer reviews are the second-most-trusted source of brand information, following only recommendations from family and friends. In other words, we trust online reviews and use them to make our own decisions.
The same is true when it comes to shopping for a doctor. According to an Internet-based survey of 2,137 adults published in the February issue of JAMA, 59% of respondents said that online doctor ratings were either "somewhat important" or "very important" when choosing a physician (2014;11:734-5).
Similarly, the "2013 Industry View Report" by Software Advice found that 62% of respondents said they read online reviews when seeking a new doctor. Although HealthGrades.com was the most commonly used site, Yelp.com was the most trusted. Forty-four percent of those respondents considered Yelp the most trustworthy review site followed by Health Grades (31%), Vitals.com (17%), and ZocDoc.com (7%).
Whether or not we trust Yelp and other online review sites, our patients do. In the JAMA survey, 35% of respondents said that they selected a physician based on good ratings, while 37% said that they avoided a physician with negative reviews. The 2013 Industry View Report also found that 45% of respondents ranked "quality of care" as the most important type of information sought about a doctor. And since many patients equate service with quality, reviews that focus on service matter.
This isn’t an entirely bad thing. If we really listen to what patients are saying, their comments can help us to improve service and communication. And, in some instances, it can lead to stronger doctor-patient relationships. Like many other industries, health care is moving toward transparency, and doctor rating sites are a key component of that.
Dr. Jeffrey Benabio is a partner physician in the department of dermatology of the Southern California Permanente Group in San Diego and a volunteer clinical assistant professor at the University of California, San Diego. He has published numerous scientific articles and is a member and fellow of the American Academy of Dermatology, and a member of the Telemedicine Association and the American Medical Association, among others. He is board certified in dermatology as well as medicine and surgery in the state of California. Dr. Benabio has a special interest in the uses of social media for education and building dermatology practice. He is the founder of The Derm Blog, an educational website that has had more than 2 million unique visitors. Dr. Benabio is also a founding member and the skin care expert for Livestrong.com, a health and wellness website of Lance Armstrong’s the Livestrong Foundation. Dr. Benabio is @Dermdoc on Twitter.
Cervical cancer screening
Numerous screening methods for cervical cancer have been proposed internationally by various professional societies, including Pap cytology alone, cytology with human papillomavirus testing as triage (HPV testing for atypical squamous cells of unknown significance [ASCUS] on cytology), cytology with HPV cotesting (cytology and HPV testing obtained together), HPV testing alone, or HPV testing followed by Pap cytology triage (cytology in patients who are positive for high-risk oncogenic subtypes of HPV). Recommendations for use of cervical cytology and HPV testing continue to vary among professional societies, with variable adoption of these guidelines by providers as well. (Am. J. Prev. Med. 2013;45:175-81).
In 2012, updated cervical cancer screening recommendations were published by ASCCP (the American Society for Colposcopy and Cervical Pathology) (Am. J. Clin. Pathol. 2012;137:516-42); the USPSTF (U.S. Preventive Services Task Force ); and ACOG (the American College of Obstetricians and Gynecologists) (Obstet. Gynecol. 2009;114:1409-20).
These most recent guidelines show a greater degree of harmony across these governing bodies than did prior guidelines. All three professional societies recommend initiating screening at age 21 years and ceasing screening at age 65 years with an adequate screening history. All groups recommend against HPV cotesting in women under 30 years of age; however, after age 30 years, ASCCP and ACOG recommend HPV cotesting every 5 years as the preferred method of cervical cancer screening, while USPSTF suggests this only as an "option." Primary HPV testing without concurrent cytology for cervical cancer screening is not currently recommended by ASCCP and USPSTF and is not addressed by ACOG.
Efficacy of screening modalities
The rationale behind these screening recommendations depends on the efficacy of both cervical cytology and HPV testing to identify preinvasive cases or invasive cervical cancer. Multiple studies have addressed the sensitivity and specificity of cytology in cervical cancer screening. Overall, the sensitivity of Pap cytology is low at approximately 51%, while specificity is high at 96%-98% (Ann. Intern. Med. 2000;132:810-9; Vaccine 2008;26 Suppl. 10:K29-41). Since the initiation of cervical cytology for cancer screening, serial annual screening has compensated for the overall poor sensitivity of the test. Two consecutive annual Pap tests can increase overall sensitivity for detection of cervical cancer to 76%, and three consecutive annual Pap tests can increase overall sensitivity to 88%.
Unlike Pap cytology, HPV testing has a high sensitivity, ranging from 81%-97% in detection of cervical cancer (N. Engl. J. Med. 2007;357:1579-88). As a result, HPV testing does not rely on serial testing for accuracy and has a high negative predictive value, making negative results very reassuring. However, HPV testing has a slightly lower specificity of 94%, which results in a higher number of false positives. Furthermore, many patients who screen positive for high-risk HPV subtypes may have transient HPV infections, which are not clinically significant, and may not cause invasive cervical cancer.
Several randomized studies have compared Pap cytology to HPV testing for use in cervical cancer screening. A Canadian study randomized more than 10,000 women to either Pap cytology or HPV testing to detect cervical intraepithelial neoplasia (CIN) 2 or higher grade cervical lesions (Int. J. Cancer. 2006;119:615-23). Findings showed a sensitivity of 55.4% for Pap cytology vs. 94.6% for HPV testing. Pap cytology had a specificity of 96.8% while HPV testing had a specificity of 94.1%. The negative predictive value of HPV testing was 100%.
Swedescreen, a Swedish study of more 12,000 women (J. Med. Virol. 2007;79:1169-75), and POBASCAM, a large Dutch study of more than 18,000 women (Lancet 2007;370:1764-72), both compared HPV testing combined with Pap cytology (cotesting) to cytology alone. Both studies found that patients screened with Pap cytology alone had more CIN2 or greater lesions in follow-up than did patients screened with cytology in combination with HPV testing (relative risk, 0.53-0.58 for CIN 2+ and RR 0.45-0.53 for CIN 3+) (J. Natl. Cancer Inst. 2009;101:88-99).
Because of the higher sensitivity of HPV testing compared with Pap cytology, some have advocated the use of HPV testing as primary screening with cytology triage rather than the reverse (cytology with HPV triage), which is more commonly used today. A Finnish study showed that primary HPV testing with cytology performed only in patients who screened positive for high risk oncogenic subtypes of HPV was more sensitive than was conventional cytology in identifying cervical dysplasia and cancer. Additionally, in women over age 35 years, HPV testing combined with Pap cytology triage was more specific than cytology alone, and decreased colposcopy referrals and follow-up tests, making this screening option cost effective (J. Natl. Cancer Inst. 2009;101:1612-23). Nowhere else in medicine is a more specific test used prior to a more sensitive test when screening for disease; the screening test is typically the more sensitive, while the confirmatory test is the more specific.
HPV vaccination and effects on screening
Currently, given that the HPV vaccines available do not protect women from all oncogenic HPV types, the ASCCP, USPSTF, and ACOG all recommend screening vaccinated women in an identical fashion to unvaccinated women. Increasing vaccination rates will likely have an impact on the efficacy of the various cervical cancer screening modalities. Vaccination will result in a reduction in the prevalence of cytologic abnormalities. As disease prevalence decreases and screening intervals increase based on current guidelines, the positive predictive value of Pap cytology also will decline, resulting in more false-positive diagnoses and possibly unnecessary procedures and patient stress (Vaccine 2013;31:5495-9). As prevalence of disease decreases, Pap cytology has the potential to become less reliable. While the positive predictive value of HPV testing also declines with decreasing disease prevalence, HPV testing is more reproducible than interpretation of Pap cytology, so the extent of increasing false-positive results may be less (Vaccine 2006;24 Suppl 3:S3/171-7).
Future directions
HPV testing as primary screening for cervical cancer is not currently recommended. However, in the post-HPV vaccination era, this may become an increasingly reasonable approach, particularly in conjunction with Pap cytology used to triage patients who test positive for high-risk HPV subtypes. HPV testing has much greater sensitivity than Pap cytology does and can better identify patients who are likely to have a cytologic abnormality. In this group of patients with greater disease prevalence, the slightly higher specificity of Pap cytology can then be used to identify precancerous lesions and guide treatment. Once this group of patients with higher lesion prevalence than the general population has been identified through HPV testing, Pap cytology can then be used and will perform better than in a lower prevalence population.
The importance of Pap cytology and HPV testing in cervical cancer screening continues to evolve, particularly in the current era of HPV vaccination. The combination of HPV testing followed by Pap cytology has potential for becoming a highly effective screening strategy; however, the optimal administration of these tests is yet to be determined. As current screening modalities improve and new technologies emerge, ongoing work is needed to identify the most effective screening method for cervical cancer.
Dr. Wysham is currently a fellow in the department of gynecologic oncology at the University of North Carolina at Chapel Hill. Dr. Kim is the department of gynecologic oncology at UNC-Chapel Hill. Dr. Gehrig is professor and director of gynecologic oncology at UNC-Chapel Hill.
Numerous screening methods for cervical cancer have been proposed internationally by various professional societies, including Pap cytology alone, cytology with human papillomavirus testing as triage (HPV testing for atypical squamous cells of unknown significance [ASCUS] on cytology), cytology with HPV cotesting (cytology and HPV testing obtained together), HPV testing alone, or HPV testing followed by Pap cytology triage (cytology in patients who are positive for high-risk oncogenic subtypes of HPV). Recommendations for use of cervical cytology and HPV testing continue to vary among professional societies, with variable adoption of these guidelines by providers as well. (Am. J. Prev. Med. 2013;45:175-81).
In 2012, updated cervical cancer screening recommendations were published by ASCCP (the American Society for Colposcopy and Cervical Pathology) (Am. J. Clin. Pathol. 2012;137:516-42); the USPSTF (U.S. Preventive Services Task Force ); and ACOG (the American College of Obstetricians and Gynecologists) (Obstet. Gynecol. 2009;114:1409-20).
These most recent guidelines show a greater degree of harmony across these governing bodies than did prior guidelines. All three professional societies recommend initiating screening at age 21 years and ceasing screening at age 65 years with an adequate screening history. All groups recommend against HPV cotesting in women under 30 years of age; however, after age 30 years, ASCCP and ACOG recommend HPV cotesting every 5 years as the preferred method of cervical cancer screening, while USPSTF suggests this only as an "option." Primary HPV testing without concurrent cytology for cervical cancer screening is not currently recommended by ASCCP and USPSTF and is not addressed by ACOG.
Efficacy of screening modalities
The rationale behind these screening recommendations depends on the efficacy of both cervical cytology and HPV testing to identify preinvasive cases or invasive cervical cancer. Multiple studies have addressed the sensitivity and specificity of cytology in cervical cancer screening. Overall, the sensitivity of Pap cytology is low at approximately 51%, while specificity is high at 96%-98% (Ann. Intern. Med. 2000;132:810-9; Vaccine 2008;26 Suppl. 10:K29-41). Since the initiation of cervical cytology for cancer screening, serial annual screening has compensated for the overall poor sensitivity of the test. Two consecutive annual Pap tests can increase overall sensitivity for detection of cervical cancer to 76%, and three consecutive annual Pap tests can increase overall sensitivity to 88%.
Unlike Pap cytology, HPV testing has a high sensitivity, ranging from 81%-97% in detection of cervical cancer (N. Engl. J. Med. 2007;357:1579-88). As a result, HPV testing does not rely on serial testing for accuracy and has a high negative predictive value, making negative results very reassuring. However, HPV testing has a slightly lower specificity of 94%, which results in a higher number of false positives. Furthermore, many patients who screen positive for high-risk HPV subtypes may have transient HPV infections, which are not clinically significant, and may not cause invasive cervical cancer.
Several randomized studies have compared Pap cytology to HPV testing for use in cervical cancer screening. A Canadian study randomized more than 10,000 women to either Pap cytology or HPV testing to detect cervical intraepithelial neoplasia (CIN) 2 or higher grade cervical lesions (Int. J. Cancer. 2006;119:615-23). Findings showed a sensitivity of 55.4% for Pap cytology vs. 94.6% for HPV testing. Pap cytology had a specificity of 96.8% while HPV testing had a specificity of 94.1%. The negative predictive value of HPV testing was 100%.
Swedescreen, a Swedish study of more 12,000 women (J. Med. Virol. 2007;79:1169-75), and POBASCAM, a large Dutch study of more than 18,000 women (Lancet 2007;370:1764-72), both compared HPV testing combined with Pap cytology (cotesting) to cytology alone. Both studies found that patients screened with Pap cytology alone had more CIN2 or greater lesions in follow-up than did patients screened with cytology in combination with HPV testing (relative risk, 0.53-0.58 for CIN 2+ and RR 0.45-0.53 for CIN 3+) (J. Natl. Cancer Inst. 2009;101:88-99).
Because of the higher sensitivity of HPV testing compared with Pap cytology, some have advocated the use of HPV testing as primary screening with cytology triage rather than the reverse (cytology with HPV triage), which is more commonly used today. A Finnish study showed that primary HPV testing with cytology performed only in patients who screened positive for high risk oncogenic subtypes of HPV was more sensitive than was conventional cytology in identifying cervical dysplasia and cancer. Additionally, in women over age 35 years, HPV testing combined with Pap cytology triage was more specific than cytology alone, and decreased colposcopy referrals and follow-up tests, making this screening option cost effective (J. Natl. Cancer Inst. 2009;101:1612-23). Nowhere else in medicine is a more specific test used prior to a more sensitive test when screening for disease; the screening test is typically the more sensitive, while the confirmatory test is the more specific.
HPV vaccination and effects on screening
Currently, given that the HPV vaccines available do not protect women from all oncogenic HPV types, the ASCCP, USPSTF, and ACOG all recommend screening vaccinated women in an identical fashion to unvaccinated women. Increasing vaccination rates will likely have an impact on the efficacy of the various cervical cancer screening modalities. Vaccination will result in a reduction in the prevalence of cytologic abnormalities. As disease prevalence decreases and screening intervals increase based on current guidelines, the positive predictive value of Pap cytology also will decline, resulting in more false-positive diagnoses and possibly unnecessary procedures and patient stress (Vaccine 2013;31:5495-9). As prevalence of disease decreases, Pap cytology has the potential to become less reliable. While the positive predictive value of HPV testing also declines with decreasing disease prevalence, HPV testing is more reproducible than interpretation of Pap cytology, so the extent of increasing false-positive results may be less (Vaccine 2006;24 Suppl 3:S3/171-7).
Future directions
HPV testing as primary screening for cervical cancer is not currently recommended. However, in the post-HPV vaccination era, this may become an increasingly reasonable approach, particularly in conjunction with Pap cytology used to triage patients who test positive for high-risk HPV subtypes. HPV testing has much greater sensitivity than Pap cytology does and can better identify patients who are likely to have a cytologic abnormality. In this group of patients with greater disease prevalence, the slightly higher specificity of Pap cytology can then be used to identify precancerous lesions and guide treatment. Once this group of patients with higher lesion prevalence than the general population has been identified through HPV testing, Pap cytology can then be used and will perform better than in a lower prevalence population.
The importance of Pap cytology and HPV testing in cervical cancer screening continues to evolve, particularly in the current era of HPV vaccination. The combination of HPV testing followed by Pap cytology has potential for becoming a highly effective screening strategy; however, the optimal administration of these tests is yet to be determined. As current screening modalities improve and new technologies emerge, ongoing work is needed to identify the most effective screening method for cervical cancer.
Dr. Wysham is currently a fellow in the department of gynecologic oncology at the University of North Carolina at Chapel Hill. Dr. Kim is the department of gynecologic oncology at UNC-Chapel Hill. Dr. Gehrig is professor and director of gynecologic oncology at UNC-Chapel Hill.
Numerous screening methods for cervical cancer have been proposed internationally by various professional societies, including Pap cytology alone, cytology with human papillomavirus testing as triage (HPV testing for atypical squamous cells of unknown significance [ASCUS] on cytology), cytology with HPV cotesting (cytology and HPV testing obtained together), HPV testing alone, or HPV testing followed by Pap cytology triage (cytology in patients who are positive for high-risk oncogenic subtypes of HPV). Recommendations for use of cervical cytology and HPV testing continue to vary among professional societies, with variable adoption of these guidelines by providers as well. (Am. J. Prev. Med. 2013;45:175-81).
In 2012, updated cervical cancer screening recommendations were published by ASCCP (the American Society for Colposcopy and Cervical Pathology) (Am. J. Clin. Pathol. 2012;137:516-42); the USPSTF (U.S. Preventive Services Task Force ); and ACOG (the American College of Obstetricians and Gynecologists) (Obstet. Gynecol. 2009;114:1409-20).
These most recent guidelines show a greater degree of harmony across these governing bodies than did prior guidelines. All three professional societies recommend initiating screening at age 21 years and ceasing screening at age 65 years with an adequate screening history. All groups recommend against HPV cotesting in women under 30 years of age; however, after age 30 years, ASCCP and ACOG recommend HPV cotesting every 5 years as the preferred method of cervical cancer screening, while USPSTF suggests this only as an "option." Primary HPV testing without concurrent cytology for cervical cancer screening is not currently recommended by ASCCP and USPSTF and is not addressed by ACOG.
Efficacy of screening modalities
The rationale behind these screening recommendations depends on the efficacy of both cervical cytology and HPV testing to identify preinvasive cases or invasive cervical cancer. Multiple studies have addressed the sensitivity and specificity of cytology in cervical cancer screening. Overall, the sensitivity of Pap cytology is low at approximately 51%, while specificity is high at 96%-98% (Ann. Intern. Med. 2000;132:810-9; Vaccine 2008;26 Suppl. 10:K29-41). Since the initiation of cervical cytology for cancer screening, serial annual screening has compensated for the overall poor sensitivity of the test. Two consecutive annual Pap tests can increase overall sensitivity for detection of cervical cancer to 76%, and three consecutive annual Pap tests can increase overall sensitivity to 88%.
Unlike Pap cytology, HPV testing has a high sensitivity, ranging from 81%-97% in detection of cervical cancer (N. Engl. J. Med. 2007;357:1579-88). As a result, HPV testing does not rely on serial testing for accuracy and has a high negative predictive value, making negative results very reassuring. However, HPV testing has a slightly lower specificity of 94%, which results in a higher number of false positives. Furthermore, many patients who screen positive for high-risk HPV subtypes may have transient HPV infections, which are not clinically significant, and may not cause invasive cervical cancer.
Several randomized studies have compared Pap cytology to HPV testing for use in cervical cancer screening. A Canadian study randomized more than 10,000 women to either Pap cytology or HPV testing to detect cervical intraepithelial neoplasia (CIN) 2 or higher grade cervical lesions (Int. J. Cancer. 2006;119:615-23). Findings showed a sensitivity of 55.4% for Pap cytology vs. 94.6% for HPV testing. Pap cytology had a specificity of 96.8% while HPV testing had a specificity of 94.1%. The negative predictive value of HPV testing was 100%.
Swedescreen, a Swedish study of more 12,000 women (J. Med. Virol. 2007;79:1169-75), and POBASCAM, a large Dutch study of more than 18,000 women (Lancet 2007;370:1764-72), both compared HPV testing combined with Pap cytology (cotesting) to cytology alone. Both studies found that patients screened with Pap cytology alone had more CIN2 or greater lesions in follow-up than did patients screened with cytology in combination with HPV testing (relative risk, 0.53-0.58 for CIN 2+ and RR 0.45-0.53 for CIN 3+) (J. Natl. Cancer Inst. 2009;101:88-99).
Because of the higher sensitivity of HPV testing compared with Pap cytology, some have advocated the use of HPV testing as primary screening with cytology triage rather than the reverse (cytology with HPV triage), which is more commonly used today. A Finnish study showed that primary HPV testing with cytology performed only in patients who screened positive for high risk oncogenic subtypes of HPV was more sensitive than was conventional cytology in identifying cervical dysplasia and cancer. Additionally, in women over age 35 years, HPV testing combined with Pap cytology triage was more specific than cytology alone, and decreased colposcopy referrals and follow-up tests, making this screening option cost effective (J. Natl. Cancer Inst. 2009;101:1612-23). Nowhere else in medicine is a more specific test used prior to a more sensitive test when screening for disease; the screening test is typically the more sensitive, while the confirmatory test is the more specific.
HPV vaccination and effects on screening
Currently, given that the HPV vaccines available do not protect women from all oncogenic HPV types, the ASCCP, USPSTF, and ACOG all recommend screening vaccinated women in an identical fashion to unvaccinated women. Increasing vaccination rates will likely have an impact on the efficacy of the various cervical cancer screening modalities. Vaccination will result in a reduction in the prevalence of cytologic abnormalities. As disease prevalence decreases and screening intervals increase based on current guidelines, the positive predictive value of Pap cytology also will decline, resulting in more false-positive diagnoses and possibly unnecessary procedures and patient stress (Vaccine 2013;31:5495-9). As prevalence of disease decreases, Pap cytology has the potential to become less reliable. While the positive predictive value of HPV testing also declines with decreasing disease prevalence, HPV testing is more reproducible than interpretation of Pap cytology, so the extent of increasing false-positive results may be less (Vaccine 2006;24 Suppl 3:S3/171-7).
Future directions
HPV testing as primary screening for cervical cancer is not currently recommended. However, in the post-HPV vaccination era, this may become an increasingly reasonable approach, particularly in conjunction with Pap cytology used to triage patients who test positive for high-risk HPV subtypes. HPV testing has much greater sensitivity than Pap cytology does and can better identify patients who are likely to have a cytologic abnormality. In this group of patients with greater disease prevalence, the slightly higher specificity of Pap cytology can then be used to identify precancerous lesions and guide treatment. Once this group of patients with higher lesion prevalence than the general population has been identified through HPV testing, Pap cytology can then be used and will perform better than in a lower prevalence population.
The importance of Pap cytology and HPV testing in cervical cancer screening continues to evolve, particularly in the current era of HPV vaccination. The combination of HPV testing followed by Pap cytology has potential for becoming a highly effective screening strategy; however, the optimal administration of these tests is yet to be determined. As current screening modalities improve and new technologies emerge, ongoing work is needed to identify the most effective screening method for cervical cancer.
Dr. Wysham is currently a fellow in the department of gynecologic oncology at the University of North Carolina at Chapel Hill. Dr. Kim is the department of gynecologic oncology at UNC-Chapel Hill. Dr. Gehrig is professor and director of gynecologic oncology at UNC-Chapel Hill.
Appearances matter as time goes by
Story
M.S. was an 89-year-old woman whose medical history was consistent with chronic mesenteric ischemia (weight loss, postprandial abdominal pain, diarrhea), and a 6-cm infrarenal aortic aneurysm for which she had previously declined surgery. She was admitted to the hospital with increased abdominal pain (constant, and unrelated to meals), bloody stools, nausea with emesis, a profound leukocytosis (WBC, 56,000 cells/mcL) and renal insufficiency (creatinine ratio, 1.63 mg/dL). One week prior to this admission, she had been discharged from the same hospital following treatment for urosepsis. She had just finished 10 days of antibiotics (ceftriaxone and cephalexin) the day before this admission.
M.S. was admitted to a regular nursing floor by Dr. Hospitalist 1 and started on intravenous fluids (2L, then hep-lock) plus oral metronidazole (for suspected Clostridium difficile infection [CDI]) and oral ciprofloxacin (for possible infectious colitis/diverticulitis). No abdominal imaging was performed by the emergency room or Dr. Hospitalist 1.
For the next 2 days, M.S. was seen daily by Dr. Hospitalist 2 in conjunction with a GI consultant. The GI consultant discontinued the ciprofloxacin and both were awaiting the results of the C. difficile toxin assay. M.S. continued to have leukocytosis (WBC 36,000 cells/mcL) with a heart rate consistently greater than 90 beats/min. However, M.S. was able to achieve adequate abdominal pain control with oral acetaminophen, and she was taking a liquid diet. Dr. Hospitalist 2 commented in his progress notes that M.S. was improving clinically.
At 11 p.m. of hospital day 2, M.S. suffered a fall from her bedside commode and hit her head, causing a laceration. Nurses noted she was lethargic and hypotensive (BP 80/50 mm Hg) with labored breathing. Dr. Hospitalist 3 was contacted, but deferred a physical assessment in favor of restarting intravenous fluids (the IV fluids ordered on admission had been discontinued earlier that afternoon) and obtaining an arterial blood gas. One hour later, M.S. had a BP 102/65 mm Hg with saline running at 150 cc/hr. The arterial blood gas demonstrated a mixed respiratory and metabolic acidosis (pH, 7.06; PaCO2 21 mm Hg). Dr. Hospitalist 3 continued to defer a physical assessment and ordered a repeat arterial blood gas that showed a worsening respiratory and metabolic acidosis (pH, 7.00; PaCO2 37 mm Hg).
At 3 a.m., the nurses called a rapid response team for lethargy and tachypnea, and M.S. quickly lost her pulse. M.S. was a "Do not resuscitate/Do not intubate" patient, and she was therefore pronounced dead. No autopsy was performed.
Complaint
Although M.S. was 89 years old, the family was surprised at her passing. Prior to her last two admissions (urosepsis and this index event), M.S. was independent, lived in her own home, hosted a weekly bridge club, and routinely interacted with family and friends.
The family was particularly troubled by the laceration on her forehead, which at the time of her death, was a deep enough wound to have required stitches for closure (which had not been performed antemortem). The laceration led to discovery of the fall, which the led to the realization that no physician went to see M.S. prior to her death. The family was suspicious and contacted an attorney who had the case reviewed and subsequently filed suit.
The complaint alleged that all of the hospitalists failed to appreciate, appropriately monitor, and appropriately treat severe CDI with severe sepsis. The complaint further alleged that Dr. Hospitalist 3 breached the standard of care by failing to attend to M.S. after her fall and initiating therapies for her acidosis.
Scientific principles
C. difficile infection is one of the most common hospital-acquired (nosocomial) infections and is an increasingly frequent cause of morbidity and mortality among elderly hospitalized patients. Patients with acute CDI may develop signs of systemic toxicity with or without profuse diarrhea warranting admission to an ICU or for emergency surgery.
Guideline parameters for severe CDI include WBC count of more than 15,000 cells/mcL or a serum creatinine level equal to 1.5 times the premorbid level. Patients with severe disease should be treated with oral vancomycin (125 mg to 500 mg q.i.d.). In the setting of ileus, addition of IV metronidazole (500 mg every 8 hours) is appropriate. Intracolonic vancomycin may be considered in patients with profound ileus. Some severely ill patients with CDI require surgical intervention as a result of toxic megacolon, perforation, or impending perforation, necrotizing colitis or rapidly progressive and/or refractory disease with systemic inflammatory response syndrome leading to multiorgan system failure
Complaint rebuttal and discussion
Without an autopsy (or abdominal imaging of any kind), defense experts first argued that M.S. died from a progression of her mesenteric ischemia (gangrene, with or without perforation), not CDI. The defense further argued that if M.S. did have severe CDI, then she ultimately would have needed surgery (regardless of appropriate fluid resuscitation, ICU care, and appropriate antibiotics), and she was not an operative candidate. If M.S. had gone for surgery, she would have in all probability died intraoperatively.
In sum, the defense focused on causation, not the failures of the hospitalists to treat severe CDI. The plaintiff maintained that severe CDI was far more likely than acute mesenteric thrombosis (given her clinical presentation and pain control with acetaminophen), and that had appropriate and timely treatment been given, M.S.’s severe CDI was a reversible condition without surgery.
Conclusion
This case further highlights the importance of family perception in the circumstances surrounding death. If M.S. had not fallen and suffered a visible injury, or if Dr. Hospitalist 3 had responded immediately to the fall with care and treatment, it is very likely that this case would not have been filed.
Nonetheless, the hospitalist care appeared deficient in several important aspects – particularly the approach to sepsis and the possibility of severe CDI. Also, it is not easy to defend a hospitalist who failed to respond to an 89-year-old inpatient who is status post a fall with a deep forehead laceration and a pH of less than 7.1. That being said, this case went to trial, and the hospitalists received a full defense verdict from the jury.
Dr. Michota is director of academic affairs in the hospital medicine department at the Cleveland Clinic and medical editor of Hospitalist News. He has been involved in peer review both within and outside the legal system.
Story
M.S. was an 89-year-old woman whose medical history was consistent with chronic mesenteric ischemia (weight loss, postprandial abdominal pain, diarrhea), and a 6-cm infrarenal aortic aneurysm for which she had previously declined surgery. She was admitted to the hospital with increased abdominal pain (constant, and unrelated to meals), bloody stools, nausea with emesis, a profound leukocytosis (WBC, 56,000 cells/mcL) and renal insufficiency (creatinine ratio, 1.63 mg/dL). One week prior to this admission, she had been discharged from the same hospital following treatment for urosepsis. She had just finished 10 days of antibiotics (ceftriaxone and cephalexin) the day before this admission.
M.S. was admitted to a regular nursing floor by Dr. Hospitalist 1 and started on intravenous fluids (2L, then hep-lock) plus oral metronidazole (for suspected Clostridium difficile infection [CDI]) and oral ciprofloxacin (for possible infectious colitis/diverticulitis). No abdominal imaging was performed by the emergency room or Dr. Hospitalist 1.
For the next 2 days, M.S. was seen daily by Dr. Hospitalist 2 in conjunction with a GI consultant. The GI consultant discontinued the ciprofloxacin and both were awaiting the results of the C. difficile toxin assay. M.S. continued to have leukocytosis (WBC 36,000 cells/mcL) with a heart rate consistently greater than 90 beats/min. However, M.S. was able to achieve adequate abdominal pain control with oral acetaminophen, and she was taking a liquid diet. Dr. Hospitalist 2 commented in his progress notes that M.S. was improving clinically.
At 11 p.m. of hospital day 2, M.S. suffered a fall from her bedside commode and hit her head, causing a laceration. Nurses noted she was lethargic and hypotensive (BP 80/50 mm Hg) with labored breathing. Dr. Hospitalist 3 was contacted, but deferred a physical assessment in favor of restarting intravenous fluids (the IV fluids ordered on admission had been discontinued earlier that afternoon) and obtaining an arterial blood gas. One hour later, M.S. had a BP 102/65 mm Hg with saline running at 150 cc/hr. The arterial blood gas demonstrated a mixed respiratory and metabolic acidosis (pH, 7.06; PaCO2 21 mm Hg). Dr. Hospitalist 3 continued to defer a physical assessment and ordered a repeat arterial blood gas that showed a worsening respiratory and metabolic acidosis (pH, 7.00; PaCO2 37 mm Hg).
At 3 a.m., the nurses called a rapid response team for lethargy and tachypnea, and M.S. quickly lost her pulse. M.S. was a "Do not resuscitate/Do not intubate" patient, and she was therefore pronounced dead. No autopsy was performed.
Complaint
Although M.S. was 89 years old, the family was surprised at her passing. Prior to her last two admissions (urosepsis and this index event), M.S. was independent, lived in her own home, hosted a weekly bridge club, and routinely interacted with family and friends.
The family was particularly troubled by the laceration on her forehead, which at the time of her death, was a deep enough wound to have required stitches for closure (which had not been performed antemortem). The laceration led to discovery of the fall, which the led to the realization that no physician went to see M.S. prior to her death. The family was suspicious and contacted an attorney who had the case reviewed and subsequently filed suit.
The complaint alleged that all of the hospitalists failed to appreciate, appropriately monitor, and appropriately treat severe CDI with severe sepsis. The complaint further alleged that Dr. Hospitalist 3 breached the standard of care by failing to attend to M.S. after her fall and initiating therapies for her acidosis.
Scientific principles
C. difficile infection is one of the most common hospital-acquired (nosocomial) infections and is an increasingly frequent cause of morbidity and mortality among elderly hospitalized patients. Patients with acute CDI may develop signs of systemic toxicity with or without profuse diarrhea warranting admission to an ICU or for emergency surgery.
Guideline parameters for severe CDI include WBC count of more than 15,000 cells/mcL or a serum creatinine level equal to 1.5 times the premorbid level. Patients with severe disease should be treated with oral vancomycin (125 mg to 500 mg q.i.d.). In the setting of ileus, addition of IV metronidazole (500 mg every 8 hours) is appropriate. Intracolonic vancomycin may be considered in patients with profound ileus. Some severely ill patients with CDI require surgical intervention as a result of toxic megacolon, perforation, or impending perforation, necrotizing colitis or rapidly progressive and/or refractory disease with systemic inflammatory response syndrome leading to multiorgan system failure
Complaint rebuttal and discussion
Without an autopsy (or abdominal imaging of any kind), defense experts first argued that M.S. died from a progression of her mesenteric ischemia (gangrene, with or without perforation), not CDI. The defense further argued that if M.S. did have severe CDI, then she ultimately would have needed surgery (regardless of appropriate fluid resuscitation, ICU care, and appropriate antibiotics), and she was not an operative candidate. If M.S. had gone for surgery, she would have in all probability died intraoperatively.
In sum, the defense focused on causation, not the failures of the hospitalists to treat severe CDI. The plaintiff maintained that severe CDI was far more likely than acute mesenteric thrombosis (given her clinical presentation and pain control with acetaminophen), and that had appropriate and timely treatment been given, M.S.’s severe CDI was a reversible condition without surgery.
Conclusion
This case further highlights the importance of family perception in the circumstances surrounding death. If M.S. had not fallen and suffered a visible injury, or if Dr. Hospitalist 3 had responded immediately to the fall with care and treatment, it is very likely that this case would not have been filed.
Nonetheless, the hospitalist care appeared deficient in several important aspects – particularly the approach to sepsis and the possibility of severe CDI. Also, it is not easy to defend a hospitalist who failed to respond to an 89-year-old inpatient who is status post a fall with a deep forehead laceration and a pH of less than 7.1. That being said, this case went to trial, and the hospitalists received a full defense verdict from the jury.
Dr. Michota is director of academic affairs in the hospital medicine department at the Cleveland Clinic and medical editor of Hospitalist News. He has been involved in peer review both within and outside the legal system.
Story
M.S. was an 89-year-old woman whose medical history was consistent with chronic mesenteric ischemia (weight loss, postprandial abdominal pain, diarrhea), and a 6-cm infrarenal aortic aneurysm for which she had previously declined surgery. She was admitted to the hospital with increased abdominal pain (constant, and unrelated to meals), bloody stools, nausea with emesis, a profound leukocytosis (WBC, 56,000 cells/mcL) and renal insufficiency (creatinine ratio, 1.63 mg/dL). One week prior to this admission, she had been discharged from the same hospital following treatment for urosepsis. She had just finished 10 days of antibiotics (ceftriaxone and cephalexin) the day before this admission.
M.S. was admitted to a regular nursing floor by Dr. Hospitalist 1 and started on intravenous fluids (2L, then hep-lock) plus oral metronidazole (for suspected Clostridium difficile infection [CDI]) and oral ciprofloxacin (for possible infectious colitis/diverticulitis). No abdominal imaging was performed by the emergency room or Dr. Hospitalist 1.
For the next 2 days, M.S. was seen daily by Dr. Hospitalist 2 in conjunction with a GI consultant. The GI consultant discontinued the ciprofloxacin and both were awaiting the results of the C. difficile toxin assay. M.S. continued to have leukocytosis (WBC 36,000 cells/mcL) with a heart rate consistently greater than 90 beats/min. However, M.S. was able to achieve adequate abdominal pain control with oral acetaminophen, and she was taking a liquid diet. Dr. Hospitalist 2 commented in his progress notes that M.S. was improving clinically.
At 11 p.m. of hospital day 2, M.S. suffered a fall from her bedside commode and hit her head, causing a laceration. Nurses noted she was lethargic and hypotensive (BP 80/50 mm Hg) with labored breathing. Dr. Hospitalist 3 was contacted, but deferred a physical assessment in favor of restarting intravenous fluids (the IV fluids ordered on admission had been discontinued earlier that afternoon) and obtaining an arterial blood gas. One hour later, M.S. had a BP 102/65 mm Hg with saline running at 150 cc/hr. The arterial blood gas demonstrated a mixed respiratory and metabolic acidosis (pH, 7.06; PaCO2 21 mm Hg). Dr. Hospitalist 3 continued to defer a physical assessment and ordered a repeat arterial blood gas that showed a worsening respiratory and metabolic acidosis (pH, 7.00; PaCO2 37 mm Hg).
At 3 a.m., the nurses called a rapid response team for lethargy and tachypnea, and M.S. quickly lost her pulse. M.S. was a "Do not resuscitate/Do not intubate" patient, and she was therefore pronounced dead. No autopsy was performed.
Complaint
Although M.S. was 89 years old, the family was surprised at her passing. Prior to her last two admissions (urosepsis and this index event), M.S. was independent, lived in her own home, hosted a weekly bridge club, and routinely interacted with family and friends.
The family was particularly troubled by the laceration on her forehead, which at the time of her death, was a deep enough wound to have required stitches for closure (which had not been performed antemortem). The laceration led to discovery of the fall, which the led to the realization that no physician went to see M.S. prior to her death. The family was suspicious and contacted an attorney who had the case reviewed and subsequently filed suit.
The complaint alleged that all of the hospitalists failed to appreciate, appropriately monitor, and appropriately treat severe CDI with severe sepsis. The complaint further alleged that Dr. Hospitalist 3 breached the standard of care by failing to attend to M.S. after her fall and initiating therapies for her acidosis.
Scientific principles
C. difficile infection is one of the most common hospital-acquired (nosocomial) infections and is an increasingly frequent cause of morbidity and mortality among elderly hospitalized patients. Patients with acute CDI may develop signs of systemic toxicity with or without profuse diarrhea warranting admission to an ICU or for emergency surgery.
Guideline parameters for severe CDI include WBC count of more than 15,000 cells/mcL or a serum creatinine level equal to 1.5 times the premorbid level. Patients with severe disease should be treated with oral vancomycin (125 mg to 500 mg q.i.d.). In the setting of ileus, addition of IV metronidazole (500 mg every 8 hours) is appropriate. Intracolonic vancomycin may be considered in patients with profound ileus. Some severely ill patients with CDI require surgical intervention as a result of toxic megacolon, perforation, or impending perforation, necrotizing colitis or rapidly progressive and/or refractory disease with systemic inflammatory response syndrome leading to multiorgan system failure
Complaint rebuttal and discussion
Without an autopsy (or abdominal imaging of any kind), defense experts first argued that M.S. died from a progression of her mesenteric ischemia (gangrene, with or without perforation), not CDI. The defense further argued that if M.S. did have severe CDI, then she ultimately would have needed surgery (regardless of appropriate fluid resuscitation, ICU care, and appropriate antibiotics), and she was not an operative candidate. If M.S. had gone for surgery, she would have in all probability died intraoperatively.
In sum, the defense focused on causation, not the failures of the hospitalists to treat severe CDI. The plaintiff maintained that severe CDI was far more likely than acute mesenteric thrombosis (given her clinical presentation and pain control with acetaminophen), and that had appropriate and timely treatment been given, M.S.’s severe CDI was a reversible condition without surgery.
Conclusion
This case further highlights the importance of family perception in the circumstances surrounding death. If M.S. had not fallen and suffered a visible injury, or if Dr. Hospitalist 3 had responded immediately to the fall with care and treatment, it is very likely that this case would not have been filed.
Nonetheless, the hospitalist care appeared deficient in several important aspects – particularly the approach to sepsis and the possibility of severe CDI. Also, it is not easy to defend a hospitalist who failed to respond to an 89-year-old inpatient who is status post a fall with a deep forehead laceration and a pH of less than 7.1. That being said, this case went to trial, and the hospitalists received a full defense verdict from the jury.
Dr. Michota is director of academic affairs in the hospital medicine department at the Cleveland Clinic and medical editor of Hospitalist News. He has been involved in peer review both within and outside the legal system.
Putting morcellation into perspective – ‘Just the facts, Ma’am, nothing but the facts’
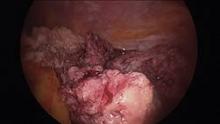
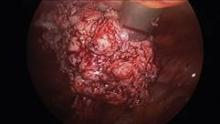
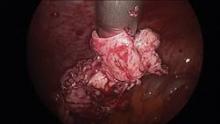
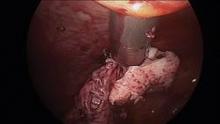
Intra-abdominal (intracorporeal) morcellation, especially electronically powered morcellation, has recently come under scrutiny. Generally performed at the time of conventional laparoscopic or robotic supracervical hysterectomy, total hysterectomy for the large uterus, or myomectomy, both power and cold-knife morcellation may splatter tissue fragments in the pelvis and abdomen, leading to potential parasitizing of the tissue and ectopic growth. Recent evidence indicates inadvertent morcellation of a leiomyosarcoma may negatively affect the patient’s subsequent disease-free survival and overall survival.
Concerns about morcellation heightened after Dr. Amy J. Reed, an anesthesiologist at Beth Israel Deaconess Medical Center, Boston, and a mother of 6, underwent presumed fibroid surgery and was diagnosed, post morcellation, with leiomyosarcoma. Dr. Reed’s husband, Dr. Hooman Noorchashm, a cardiothoracic surgeon at Brigham and Women’s Hospital, Boston, where his wife’s surgery was performed, is calling for a moratorium on intra-abdominal morcellation, whether it involves the use of a power morcellator, or for that matter, the cold knife.
It is imperative and incumbent upon our specialty to have a detailed evaluation of the risks and benefits of morcellation. While morcellation of the rare leiomyosarcoma is a risk, banning intraabdominal/intrapelvic morcellation will certainly have a profound negative impact on patients who are able to undergo a minimally invasive gynecologic procedure. Banning morcellation would increase intraoperative risk and subsequent concern of postoperative pelvic adhesions and thus, potential impact on fertility (post myomectomy), dyspareunia, and pelvic pain. Further, a ban would incur higher costs and more loss of patient productivity (Hum. Reprod. 1998 13:2102-6). These concerns were the basis for the AAGL position statement touting a minimally invasive approach to hysterectomy (J. Minim. Invasive Gynecol. 2011;18:1-3).
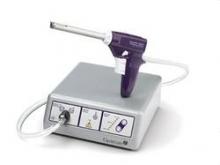
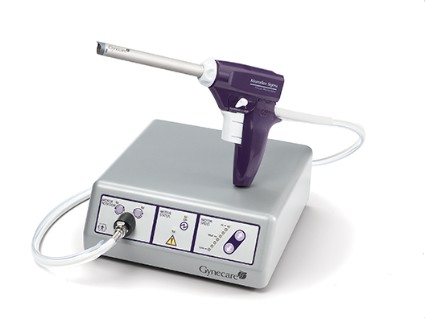
Since their introduction in the mid-1990s, electronically powered morcellators have been used to remove the uterus, fibroid(s), spleen, or kidney. Varying in size from 12-20 mm, electronic morcellators generally consist of a rotating circular blade at the end of a hollow tube. A tenaculum or multitoothed grasper is placed through the tube and blade to grasp the tissue to the revolving blade. The specimen is then removed in strips. Tissue splatter is inevitable, at least until the technique evolves to allow morcellation to be performed within the confines of a bag.
Benign uterine fibroids are the most common pelvic tumor in women. Literature reviews indicate the lifetime risk is 70% for white women and 80% in women of African ancestry. Uterine sarcomas occur in 3-7 women per 100,000 (Am. J. Obstet. Gynecol. 2011;205:492.e1-5). Further, Dr. Kimberly A. Kho of the University of Texas Southwestern Medical Center, Dallas, and Dr. Ceana H. Dr. Nezhat of Atlanta Center for Minimally Invasive Surgery and Reproductive Medicine, conducted a meta-analysis of 5,666 uterine procedures, and found 13 unsuspected uterine sarcomas, for a prevalence of 0.23% (JAMA 2014 [doi:10.1001/jama.2014.1093]).
This finding is consistent with that of a previous study by Dr. W.H. Parker who also noted a 0.23% risk, based on data from 1,332 women undergoing surgery secondary to uterine fibroids. Interestingly, in Dr. Parker’s study, the risk was 0.27% among women with rapidly growing leiomyoma, often thought to be a risk factor for sarcoma development (Obstet. Gynecol. 1994;83:414-8).
Because of the difficulty of making a preoperative diagnosis of leiomyosarcoma, it is doubtful that this risk will be decreased in the near future. Risk factors have not been well established, although a twofold higher incidence of leiomyosarcomas has been observed in black women (Gynecol. Oncol. 2004;93:204-8). Increasing age would appear to increase uterine sarcoma risk, as the majority of cases are diagnosed in postmenopausal women. Tamoxifen, when used for 5 or more years, appears to be associated with higher sarcoma rates (J. Clin. Oncol. 2002;20:2758-60) as is a history of pelvic irradiation or childhood retinoblastoma.
Unless metastatic disease is present, symptoms are similar for leiomyomas and leiomyosarcomas. A rapidly growing mass, a finding associated with an increased risk of uterine sarcoma, was not seen in Parker’s study of 1,332 women undergoing hysterectomy or myomectomy for uterine leiomyoma. Similarly, size does not count; a large uterine mass or increased uterine size did not appear to be associated with a greater risk of sarcoma (Gynecol. Oncol. 2003;89:460-9).
Some contend that failed response with such therapies as gonadotropin-releasing hormone agonists and uterine artery embolization are associated with increased incidence of leiomyosarcoma, but the data are not convincing (Eur. J. Obstet. Gynecol. Reprod. Biol. 1998;76:237-40).
Physical examination and imaging may be helpful in finding enlarged lymph nodes, but imaging methods have not been reliably shown to enable a preoperative diagnosis of uterine leiomyosarcoma (Lancet Oncol. 2009;10:1188-98; AJR Am. J. Roentgenol. 2003;181:1369-74). Further, while some physicians point out that an ill-defined margin may increase leiomyosarcoma risk, this finding is certainly noted as well with benign adenomyomas.
Finally, data are scant in support of preoperative endometrial sampling to establish a diagnosis of leiomyosarcoma. In two studies comparing a total of 14 patients, 7 were correctly diagnosed with leiomyosarcoma prior to surgery (Am. J. Obstet. Gynecol. 1990;162:968-74; Gynecol. Oncol. 2008;110:43-8).
With little differentiation in clinical presentation and the inability to distinguish leiomyoma from leiomyosarcoma based on imaging or sampling, it is not surprising that patients undergoing morcellation for an expected benign condition would subsequently be diagnosed with uterine leiomyosarcoma. With this in mind, it is important to review the current body of literature to further evaluate the risks and benefits of morcellation, and what place minimally invasive gynecologic surgery will have for the treatment of uterine masses.
Tumor morcellation of unrecognized leiomyosarcomas was significantly associated with poorer disease free survival (odds ratio, 2.59, P = 1.43), higher stage (I vs. II; [OR, 19.12, P = .037]) and poorer overall survival (OR, 3.07, P =.040) in a 2011 study. Park et al. assessed 56 consecutive patients, 25 with morcellation and 31 without tumor morcellation, who had stage I and stage II uterine leiomyosarcomas and were treated between 1989 and 2010. The percentage of patients with dissemination also was noted to be greater in patients with tumor morcellation (44% vs. 12.9%, P =.032). Interestingly, ovarian tissue was more frequently preserved in the morcellation group (38.7% vs. 72%, P =.013) (Gynecol. Oncol. 2011;122:255-9)
In response to a subsequent Letter to the Editor about these risks, the study’s author put the findings in perspective. "The frequency of incidental uterine leiomyosarcoma in patients who undergo surgery for presumed uterine leiomyoma is extremely rare. At our medical center, only 49 of 22,825 patients (0.21%) who underwent surgery for presumed uterine leiomyoma had incidental uterine leiomyosarcoma. Therefore, we believe that surgeons need not avoid non-laparotomic* surgical routes because of the rare possibility of an incidental diagnosis of leiomyosarcoma, even when tumor morcellation is required" (Gynecol. Oncol. 2012;124:172-3).
Additionally, a retrospective study from Brigham & Women’s Hospital found that disease was often already disseminated before morcellation procedures. In 21 patients with a median age of 46 years and no documented evidence of extrauterine disease, 15 had uterine leiomyosarcomas and 6 had smooth muscle tumors of uncertain malignant potential that were inadvertently morcellated; data was incorporated from January 2005 to January 2012. While most patients underwent power morcellation with laparoscopy, two underwent laparoscopically assisted vaginal hysterectomy with hand morcellation, and one patient had a vaginal hysterectomy with hand morcellation.
Immediate surgical reexploration was performed for staging in 12 patients. Significant findings of disseminated intraperitoneal disease were detected in two of seven patients with presumed stage I uterine leiomyosarcoma and in one of four patients with presumed stage I smooth muscle tumors of uncertain malignant potential. Moreover, of the eight patients who did not have disseminated disease at the time of the staging procedure, one subsequently had a recurrence. The remaining patients had no recurrences and remain disease free.
One patient was already FIGO stage IV at the original surgery, two more patients were upstaged at the original surgery and underwent re-exploration at 18 and 20 months respectively (certainly, a long period prior to second look). Moreover, the authors note various reasons why a significant number of patients were upstaged; including incorrect staging after initial surgery, progression of disease during the time interval, or secondary to direct seeding of morcellated tumor fragments. Five of the 15 leiomyosarcoma patients were deceased at the time of the publication. The authors also point out that their study is limited by the fact that it is retrospective, and access to information regarding care received from non-affiliated institutions is limited (Gynecol. Oncol. 2014;132:360-5).
In summary, morcellation of an unsuspected uterine sarcoma, whether using an electrically powered morcellator at the time of laparoscopy or cold knife at time of vaginal surgery, appears to have a negative impact; however, the studies to date are merely retrospective case studies. By no means do they provide the evidence required to place a moratorium on morcellation.
Further, if such a ban is imposed, would it then not be equally justifiable to pose similar regulations on use of oral contraceptives for symptom relief, endometrial ablation when fibroids are involved, or for that matter, uterine artery embolization? All these potential treatment regimens delay diagnosis and treatment and leave the potential uterine sarcoma in situ.
In the end, while the disease-free survival as well as overall survival appears to be hindered by dissemination of leiomyosarcoma at time of both electronic and cold-knife morcellation, the diagnosis is fortunately rare. A moratorium on the technique, however, would increase the number of concomitant laparotomies that would be required, and along with it, the increased inherent risk as well as prolonged recovery. At the present time, without better diagnostic tools or safer morcellation techniques, it is imperative to have an open dialogue of the risks and benefits of morcellation and minimally invasive surgery with patients presenting with anticipated fibroids. Additionally, our industry partners must be empowered to create safer morcellation techniques. This would appear to be morcellation within a bag.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, immediate past president of the International Society for Gynecologic Endoscopy, and a past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville, Ill., and Schaumburg, Ill.; the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column. Dr. Miller said he is a consultant for Ethicon, which manufactures a morcellator.
*Correction, 3/19/2014: An earlier version of this story misstated the type of surgical route.
Intra-abdominal (intracorporeal) morcellation, especially electronically powered morcellation, has recently come under scrutiny. Generally performed at the time of conventional laparoscopic or robotic supracervical hysterectomy, total hysterectomy for the large uterus, or myomectomy, both power and cold-knife morcellation may splatter tissue fragments in the pelvis and abdomen, leading to potential parasitizing of the tissue and ectopic growth. Recent evidence indicates inadvertent morcellation of a leiomyosarcoma may negatively affect the patient’s subsequent disease-free survival and overall survival.
Concerns about morcellation heightened after Dr. Amy J. Reed, an anesthesiologist at Beth Israel Deaconess Medical Center, Boston, and a mother of 6, underwent presumed fibroid surgery and was diagnosed, post morcellation, with leiomyosarcoma. Dr. Reed’s husband, Dr. Hooman Noorchashm, a cardiothoracic surgeon at Brigham and Women’s Hospital, Boston, where his wife’s surgery was performed, is calling for a moratorium on intra-abdominal morcellation, whether it involves the use of a power morcellator, or for that matter, the cold knife.
It is imperative and incumbent upon our specialty to have a detailed evaluation of the risks and benefits of morcellation. While morcellation of the rare leiomyosarcoma is a risk, banning intraabdominal/intrapelvic morcellation will certainly have a profound negative impact on patients who are able to undergo a minimally invasive gynecologic procedure. Banning morcellation would increase intraoperative risk and subsequent concern of postoperative pelvic adhesions and thus, potential impact on fertility (post myomectomy), dyspareunia, and pelvic pain. Further, a ban would incur higher costs and more loss of patient productivity (Hum. Reprod. 1998 13:2102-6). These concerns were the basis for the AAGL position statement touting a minimally invasive approach to hysterectomy (J. Minim. Invasive Gynecol. 2011;18:1-3).
Since their introduction in the mid-1990s, electronically powered morcellators have been used to remove the uterus, fibroid(s), spleen, or kidney. Varying in size from 12-20 mm, electronic morcellators generally consist of a rotating circular blade at the end of a hollow tube. A tenaculum or multitoothed grasper is placed through the tube and blade to grasp the tissue to the revolving blade. The specimen is then removed in strips. Tissue splatter is inevitable, at least until the technique evolves to allow morcellation to be performed within the confines of a bag.
Benign uterine fibroids are the most common pelvic tumor in women. Literature reviews indicate the lifetime risk is 70% for white women and 80% in women of African ancestry. Uterine sarcomas occur in 3-7 women per 100,000 (Am. J. Obstet. Gynecol. 2011;205:492.e1-5). Further, Dr. Kimberly A. Kho of the University of Texas Southwestern Medical Center, Dallas, and Dr. Ceana H. Dr. Nezhat of Atlanta Center for Minimally Invasive Surgery and Reproductive Medicine, conducted a meta-analysis of 5,666 uterine procedures, and found 13 unsuspected uterine sarcomas, for a prevalence of 0.23% (JAMA 2014 [doi:10.1001/jama.2014.1093]).
This finding is consistent with that of a previous study by Dr. W.H. Parker who also noted a 0.23% risk, based on data from 1,332 women undergoing surgery secondary to uterine fibroids. Interestingly, in Dr. Parker’s study, the risk was 0.27% among women with rapidly growing leiomyoma, often thought to be a risk factor for sarcoma development (Obstet. Gynecol. 1994;83:414-8).
Because of the difficulty of making a preoperative diagnosis of leiomyosarcoma, it is doubtful that this risk will be decreased in the near future. Risk factors have not been well established, although a twofold higher incidence of leiomyosarcomas has been observed in black women (Gynecol. Oncol. 2004;93:204-8). Increasing age would appear to increase uterine sarcoma risk, as the majority of cases are diagnosed in postmenopausal women. Tamoxifen, when used for 5 or more years, appears to be associated with higher sarcoma rates (J. Clin. Oncol. 2002;20:2758-60) as is a history of pelvic irradiation or childhood retinoblastoma.
Unless metastatic disease is present, symptoms are similar for leiomyomas and leiomyosarcomas. A rapidly growing mass, a finding associated with an increased risk of uterine sarcoma, was not seen in Parker’s study of 1,332 women undergoing hysterectomy or myomectomy for uterine leiomyoma. Similarly, size does not count; a large uterine mass or increased uterine size did not appear to be associated with a greater risk of sarcoma (Gynecol. Oncol. 2003;89:460-9).
Some contend that failed response with such therapies as gonadotropin-releasing hormone agonists and uterine artery embolization are associated with increased incidence of leiomyosarcoma, but the data are not convincing (Eur. J. Obstet. Gynecol. Reprod. Biol. 1998;76:237-40).
Physical examination and imaging may be helpful in finding enlarged lymph nodes, but imaging methods have not been reliably shown to enable a preoperative diagnosis of uterine leiomyosarcoma (Lancet Oncol. 2009;10:1188-98; AJR Am. J. Roentgenol. 2003;181:1369-74). Further, while some physicians point out that an ill-defined margin may increase leiomyosarcoma risk, this finding is certainly noted as well with benign adenomyomas.
Finally, data are scant in support of preoperative endometrial sampling to establish a diagnosis of leiomyosarcoma. In two studies comparing a total of 14 patients, 7 were correctly diagnosed with leiomyosarcoma prior to surgery (Am. J. Obstet. Gynecol. 1990;162:968-74; Gynecol. Oncol. 2008;110:43-8).
With little differentiation in clinical presentation and the inability to distinguish leiomyoma from leiomyosarcoma based on imaging or sampling, it is not surprising that patients undergoing morcellation for an expected benign condition would subsequently be diagnosed with uterine leiomyosarcoma. With this in mind, it is important to review the current body of literature to further evaluate the risks and benefits of morcellation, and what place minimally invasive gynecologic surgery will have for the treatment of uterine masses.
Tumor morcellation of unrecognized leiomyosarcomas was significantly associated with poorer disease free survival (odds ratio, 2.59, P = 1.43), higher stage (I vs. II; [OR, 19.12, P = .037]) and poorer overall survival (OR, 3.07, P =.040) in a 2011 study. Park et al. assessed 56 consecutive patients, 25 with morcellation and 31 without tumor morcellation, who had stage I and stage II uterine leiomyosarcomas and were treated between 1989 and 2010. The percentage of patients with dissemination also was noted to be greater in patients with tumor morcellation (44% vs. 12.9%, P =.032). Interestingly, ovarian tissue was more frequently preserved in the morcellation group (38.7% vs. 72%, P =.013) (Gynecol. Oncol. 2011;122:255-9)
In response to a subsequent Letter to the Editor about these risks, the study’s author put the findings in perspective. "The frequency of incidental uterine leiomyosarcoma in patients who undergo surgery for presumed uterine leiomyoma is extremely rare. At our medical center, only 49 of 22,825 patients (0.21%) who underwent surgery for presumed uterine leiomyoma had incidental uterine leiomyosarcoma. Therefore, we believe that surgeons need not avoid non-laparotomic* surgical routes because of the rare possibility of an incidental diagnosis of leiomyosarcoma, even when tumor morcellation is required" (Gynecol. Oncol. 2012;124:172-3).
Additionally, a retrospective study from Brigham & Women’s Hospital found that disease was often already disseminated before morcellation procedures. In 21 patients with a median age of 46 years and no documented evidence of extrauterine disease, 15 had uterine leiomyosarcomas and 6 had smooth muscle tumors of uncertain malignant potential that were inadvertently morcellated; data was incorporated from January 2005 to January 2012. While most patients underwent power morcellation with laparoscopy, two underwent laparoscopically assisted vaginal hysterectomy with hand morcellation, and one patient had a vaginal hysterectomy with hand morcellation.
Immediate surgical reexploration was performed for staging in 12 patients. Significant findings of disseminated intraperitoneal disease were detected in two of seven patients with presumed stage I uterine leiomyosarcoma and in one of four patients with presumed stage I smooth muscle tumors of uncertain malignant potential. Moreover, of the eight patients who did not have disseminated disease at the time of the staging procedure, one subsequently had a recurrence. The remaining patients had no recurrences and remain disease free.
One patient was already FIGO stage IV at the original surgery, two more patients were upstaged at the original surgery and underwent re-exploration at 18 and 20 months respectively (certainly, a long period prior to second look). Moreover, the authors note various reasons why a significant number of patients were upstaged; including incorrect staging after initial surgery, progression of disease during the time interval, or secondary to direct seeding of morcellated tumor fragments. Five of the 15 leiomyosarcoma patients were deceased at the time of the publication. The authors also point out that their study is limited by the fact that it is retrospective, and access to information regarding care received from non-affiliated institutions is limited (Gynecol. Oncol. 2014;132:360-5).
In summary, morcellation of an unsuspected uterine sarcoma, whether using an electrically powered morcellator at the time of laparoscopy or cold knife at time of vaginal surgery, appears to have a negative impact; however, the studies to date are merely retrospective case studies. By no means do they provide the evidence required to place a moratorium on morcellation.
Further, if such a ban is imposed, would it then not be equally justifiable to pose similar regulations on use of oral contraceptives for symptom relief, endometrial ablation when fibroids are involved, or for that matter, uterine artery embolization? All these potential treatment regimens delay diagnosis and treatment and leave the potential uterine sarcoma in situ.
In the end, while the disease-free survival as well as overall survival appears to be hindered by dissemination of leiomyosarcoma at time of both electronic and cold-knife morcellation, the diagnosis is fortunately rare. A moratorium on the technique, however, would increase the number of concomitant laparotomies that would be required, and along with it, the increased inherent risk as well as prolonged recovery. At the present time, without better diagnostic tools or safer morcellation techniques, it is imperative to have an open dialogue of the risks and benefits of morcellation and minimally invasive surgery with patients presenting with anticipated fibroids. Additionally, our industry partners must be empowered to create safer morcellation techniques. This would appear to be morcellation within a bag.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, immediate past president of the International Society for Gynecologic Endoscopy, and a past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville, Ill., and Schaumburg, Ill.; the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column. Dr. Miller said he is a consultant for Ethicon, which manufactures a morcellator.
*Correction, 3/19/2014: An earlier version of this story misstated the type of surgical route.
Intra-abdominal (intracorporeal) morcellation, especially electronically powered morcellation, has recently come under scrutiny. Generally performed at the time of conventional laparoscopic or robotic supracervical hysterectomy, total hysterectomy for the large uterus, or myomectomy, both power and cold-knife morcellation may splatter tissue fragments in the pelvis and abdomen, leading to potential parasitizing of the tissue and ectopic growth. Recent evidence indicates inadvertent morcellation of a leiomyosarcoma may negatively affect the patient’s subsequent disease-free survival and overall survival.
Concerns about morcellation heightened after Dr. Amy J. Reed, an anesthesiologist at Beth Israel Deaconess Medical Center, Boston, and a mother of 6, underwent presumed fibroid surgery and was diagnosed, post morcellation, with leiomyosarcoma. Dr. Reed’s husband, Dr. Hooman Noorchashm, a cardiothoracic surgeon at Brigham and Women’s Hospital, Boston, where his wife’s surgery was performed, is calling for a moratorium on intra-abdominal morcellation, whether it involves the use of a power morcellator, or for that matter, the cold knife.
It is imperative and incumbent upon our specialty to have a detailed evaluation of the risks and benefits of morcellation. While morcellation of the rare leiomyosarcoma is a risk, banning intraabdominal/intrapelvic morcellation will certainly have a profound negative impact on patients who are able to undergo a minimally invasive gynecologic procedure. Banning morcellation would increase intraoperative risk and subsequent concern of postoperative pelvic adhesions and thus, potential impact on fertility (post myomectomy), dyspareunia, and pelvic pain. Further, a ban would incur higher costs and more loss of patient productivity (Hum. Reprod. 1998 13:2102-6). These concerns were the basis for the AAGL position statement touting a minimally invasive approach to hysterectomy (J. Minim. Invasive Gynecol. 2011;18:1-3).
Since their introduction in the mid-1990s, electronically powered morcellators have been used to remove the uterus, fibroid(s), spleen, or kidney. Varying in size from 12-20 mm, electronic morcellators generally consist of a rotating circular blade at the end of a hollow tube. A tenaculum or multitoothed grasper is placed through the tube and blade to grasp the tissue to the revolving blade. The specimen is then removed in strips. Tissue splatter is inevitable, at least until the technique evolves to allow morcellation to be performed within the confines of a bag.
Benign uterine fibroids are the most common pelvic tumor in women. Literature reviews indicate the lifetime risk is 70% for white women and 80% in women of African ancestry. Uterine sarcomas occur in 3-7 women per 100,000 (Am. J. Obstet. Gynecol. 2011;205:492.e1-5). Further, Dr. Kimberly A. Kho of the University of Texas Southwestern Medical Center, Dallas, and Dr. Ceana H. Dr. Nezhat of Atlanta Center for Minimally Invasive Surgery and Reproductive Medicine, conducted a meta-analysis of 5,666 uterine procedures, and found 13 unsuspected uterine sarcomas, for a prevalence of 0.23% (JAMA 2014 [doi:10.1001/jama.2014.1093]).
This finding is consistent with that of a previous study by Dr. W.H. Parker who also noted a 0.23% risk, based on data from 1,332 women undergoing surgery secondary to uterine fibroids. Interestingly, in Dr. Parker’s study, the risk was 0.27% among women with rapidly growing leiomyoma, often thought to be a risk factor for sarcoma development (Obstet. Gynecol. 1994;83:414-8).
Because of the difficulty of making a preoperative diagnosis of leiomyosarcoma, it is doubtful that this risk will be decreased in the near future. Risk factors have not been well established, although a twofold higher incidence of leiomyosarcomas has been observed in black women (Gynecol. Oncol. 2004;93:204-8). Increasing age would appear to increase uterine sarcoma risk, as the majority of cases are diagnosed in postmenopausal women. Tamoxifen, when used for 5 or more years, appears to be associated with higher sarcoma rates (J. Clin. Oncol. 2002;20:2758-60) as is a history of pelvic irradiation or childhood retinoblastoma.
Unless metastatic disease is present, symptoms are similar for leiomyomas and leiomyosarcomas. A rapidly growing mass, a finding associated with an increased risk of uterine sarcoma, was not seen in Parker’s study of 1,332 women undergoing hysterectomy or myomectomy for uterine leiomyoma. Similarly, size does not count; a large uterine mass or increased uterine size did not appear to be associated with a greater risk of sarcoma (Gynecol. Oncol. 2003;89:460-9).
Some contend that failed response with such therapies as gonadotropin-releasing hormone agonists and uterine artery embolization are associated with increased incidence of leiomyosarcoma, but the data are not convincing (Eur. J. Obstet. Gynecol. Reprod. Biol. 1998;76:237-40).
Physical examination and imaging may be helpful in finding enlarged lymph nodes, but imaging methods have not been reliably shown to enable a preoperative diagnosis of uterine leiomyosarcoma (Lancet Oncol. 2009;10:1188-98; AJR Am. J. Roentgenol. 2003;181:1369-74). Further, while some physicians point out that an ill-defined margin may increase leiomyosarcoma risk, this finding is certainly noted as well with benign adenomyomas.
Finally, data are scant in support of preoperative endometrial sampling to establish a diagnosis of leiomyosarcoma. In two studies comparing a total of 14 patients, 7 were correctly diagnosed with leiomyosarcoma prior to surgery (Am. J. Obstet. Gynecol. 1990;162:968-74; Gynecol. Oncol. 2008;110:43-8).
With little differentiation in clinical presentation and the inability to distinguish leiomyoma from leiomyosarcoma based on imaging or sampling, it is not surprising that patients undergoing morcellation for an expected benign condition would subsequently be diagnosed with uterine leiomyosarcoma. With this in mind, it is important to review the current body of literature to further evaluate the risks and benefits of morcellation, and what place minimally invasive gynecologic surgery will have for the treatment of uterine masses.
Tumor morcellation of unrecognized leiomyosarcomas was significantly associated with poorer disease free survival (odds ratio, 2.59, P = 1.43), higher stage (I vs. II; [OR, 19.12, P = .037]) and poorer overall survival (OR, 3.07, P =.040) in a 2011 study. Park et al. assessed 56 consecutive patients, 25 with morcellation and 31 without tumor morcellation, who had stage I and stage II uterine leiomyosarcomas and were treated between 1989 and 2010. The percentage of patients with dissemination also was noted to be greater in patients with tumor morcellation (44% vs. 12.9%, P =.032). Interestingly, ovarian tissue was more frequently preserved in the morcellation group (38.7% vs. 72%, P =.013) (Gynecol. Oncol. 2011;122:255-9)
In response to a subsequent Letter to the Editor about these risks, the study’s author put the findings in perspective. "The frequency of incidental uterine leiomyosarcoma in patients who undergo surgery for presumed uterine leiomyoma is extremely rare. At our medical center, only 49 of 22,825 patients (0.21%) who underwent surgery for presumed uterine leiomyoma had incidental uterine leiomyosarcoma. Therefore, we believe that surgeons need not avoid non-laparotomic* surgical routes because of the rare possibility of an incidental diagnosis of leiomyosarcoma, even when tumor morcellation is required" (Gynecol. Oncol. 2012;124:172-3).
Additionally, a retrospective study from Brigham & Women’s Hospital found that disease was often already disseminated before morcellation procedures. In 21 patients with a median age of 46 years and no documented evidence of extrauterine disease, 15 had uterine leiomyosarcomas and 6 had smooth muscle tumors of uncertain malignant potential that were inadvertently morcellated; data was incorporated from January 2005 to January 2012. While most patients underwent power morcellation with laparoscopy, two underwent laparoscopically assisted vaginal hysterectomy with hand morcellation, and one patient had a vaginal hysterectomy with hand morcellation.
Immediate surgical reexploration was performed for staging in 12 patients. Significant findings of disseminated intraperitoneal disease were detected in two of seven patients with presumed stage I uterine leiomyosarcoma and in one of four patients with presumed stage I smooth muscle tumors of uncertain malignant potential. Moreover, of the eight patients who did not have disseminated disease at the time of the staging procedure, one subsequently had a recurrence. The remaining patients had no recurrences and remain disease free.
One patient was already FIGO stage IV at the original surgery, two more patients were upstaged at the original surgery and underwent re-exploration at 18 and 20 months respectively (certainly, a long period prior to second look). Moreover, the authors note various reasons why a significant number of patients were upstaged; including incorrect staging after initial surgery, progression of disease during the time interval, or secondary to direct seeding of morcellated tumor fragments. Five of the 15 leiomyosarcoma patients were deceased at the time of the publication. The authors also point out that their study is limited by the fact that it is retrospective, and access to information regarding care received from non-affiliated institutions is limited (Gynecol. Oncol. 2014;132:360-5).
In summary, morcellation of an unsuspected uterine sarcoma, whether using an electrically powered morcellator at the time of laparoscopy or cold knife at time of vaginal surgery, appears to have a negative impact; however, the studies to date are merely retrospective case studies. By no means do they provide the evidence required to place a moratorium on morcellation.
Further, if such a ban is imposed, would it then not be equally justifiable to pose similar regulations on use of oral contraceptives for symptom relief, endometrial ablation when fibroids are involved, or for that matter, uterine artery embolization? All these potential treatment regimens delay diagnosis and treatment and leave the potential uterine sarcoma in situ.
In the end, while the disease-free survival as well as overall survival appears to be hindered by dissemination of leiomyosarcoma at time of both electronic and cold-knife morcellation, the diagnosis is fortunately rare. A moratorium on the technique, however, would increase the number of concomitant laparotomies that would be required, and along with it, the increased inherent risk as well as prolonged recovery. At the present time, without better diagnostic tools or safer morcellation techniques, it is imperative to have an open dialogue of the risks and benefits of morcellation and minimally invasive surgery with patients presenting with anticipated fibroids. Additionally, our industry partners must be empowered to create safer morcellation techniques. This would appear to be morcellation within a bag.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, immediate past president of the International Society for Gynecologic Endoscopy, and a past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville, Ill., and Schaumburg, Ill.; the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column. Dr. Miller said he is a consultant for Ethicon, which manufactures a morcellator.
*Correction, 3/19/2014: An earlier version of this story misstated the type of surgical route.
Take care to care for the poor
I’m guilty. I feel a sense of entitlement, as many of us do. I have always had access to the best health care available. Even as a child, whenever I had a runny nose, my parents not only knew what to do but could afford whatever it took to make me feel better. And vaccine-preventable illnesses? They didn’t stand a chance at my house. I remember the day when I was a little girl and my father, a general practitioner, figured out my plan to wait him out as he stood patiently outside the bathroom door, vaccine in hand. He eventually got tired of waiting, barged in, oblivious to the fact that I was sitting on the potty (twiddling my fingers), and shot me right in arm. The nerve!
Many of us have no concept of what it is like to be ill. I mean really ill with no one to help, and no way to pay for that help even if we could find it. In the March 6, 2014, issue of the New England Journal of Medicine, authors of "Global Supply of Health Professionals" note that there’s a worldwide crisis of severe shortages, as well as maldistribution of health care professionals intensified by three global transitions: redistribution of the disability burden, demographic changes, and epidemiologic shifts. An estimated 25% of physicians in America come from other countries. Naturally, in some cases the countries of origin have their own health care challenges, so the trend to immigrate to America has significant potential to exacerbate an already critical shortage.
Of the estimated 9.2 million doctors worldwide, 8% reside in the United States. Of the 18.1 million nurses, 17% are in here. To put it in perspective, only 4% of the world’s population lives in the United States, yet we command a lion’s share of the global health care workforce, leaving the citizens of many other countries vulnerable to excessive suffering, premature death, and preventable diseases.
With all that America has to offer, I don’t see the pendulum shifting back in the other direction any time soon, but many of us are in a position to provide much needed health care to our brothers and sisters in even the most remote areas of the world.
As squeamish as I am about charting unknown territory, even I went on a medical missions trip to Nicaragua a few years ago and I will never forget it. We had to climb up the side of a mountain to get to a clearing that would hold the makeshift medical clinic. Of course, the locals came out in large numbers to receive much-needed medical care.
On the way back to the hotel I noticed some little boys tossing an object back and forth in the street. It seemed like pleasant fun until we got closer and I saw the object they were tossing was a dead rat. Even what we consider an inexpensive child’s toy is a luxury for many. If you consider salary and benefits, we hospitalists make more, on average, in a single shift than many Nicaraguans make in an entire year.
Every physician should spend some time caring for the poorest of the poor, whether it be in a needy foreign country, Appalachia or rural America, or in the inner city, often just 30 minutes from our homes.
Surely it will help or change the patient’s life. But, too, it just might change your perspective, forever, and renew your passion for what we do – help others.
Dr. Hester is a hospitalist with Baltimore-Washington Medical Center who has a passion for empowering patients to partner in their health care. She is the creator of the Patient Whiz, a patient-engagement app for iOS.
I’m guilty. I feel a sense of entitlement, as many of us do. I have always had access to the best health care available. Even as a child, whenever I had a runny nose, my parents not only knew what to do but could afford whatever it took to make me feel better. And vaccine-preventable illnesses? They didn’t stand a chance at my house. I remember the day when I was a little girl and my father, a general practitioner, figured out my plan to wait him out as he stood patiently outside the bathroom door, vaccine in hand. He eventually got tired of waiting, barged in, oblivious to the fact that I was sitting on the potty (twiddling my fingers), and shot me right in arm. The nerve!
Many of us have no concept of what it is like to be ill. I mean really ill with no one to help, and no way to pay for that help even if we could find it. In the March 6, 2014, issue of the New England Journal of Medicine, authors of "Global Supply of Health Professionals" note that there’s a worldwide crisis of severe shortages, as well as maldistribution of health care professionals intensified by three global transitions: redistribution of the disability burden, demographic changes, and epidemiologic shifts. An estimated 25% of physicians in America come from other countries. Naturally, in some cases the countries of origin have their own health care challenges, so the trend to immigrate to America has significant potential to exacerbate an already critical shortage.
Of the estimated 9.2 million doctors worldwide, 8% reside in the United States. Of the 18.1 million nurses, 17% are in here. To put it in perspective, only 4% of the world’s population lives in the United States, yet we command a lion’s share of the global health care workforce, leaving the citizens of many other countries vulnerable to excessive suffering, premature death, and preventable diseases.
With all that America has to offer, I don’t see the pendulum shifting back in the other direction any time soon, but many of us are in a position to provide much needed health care to our brothers and sisters in even the most remote areas of the world.
As squeamish as I am about charting unknown territory, even I went on a medical missions trip to Nicaragua a few years ago and I will never forget it. We had to climb up the side of a mountain to get to a clearing that would hold the makeshift medical clinic. Of course, the locals came out in large numbers to receive much-needed medical care.
On the way back to the hotel I noticed some little boys tossing an object back and forth in the street. It seemed like pleasant fun until we got closer and I saw the object they were tossing was a dead rat. Even what we consider an inexpensive child’s toy is a luxury for many. If you consider salary and benefits, we hospitalists make more, on average, in a single shift than many Nicaraguans make in an entire year.
Every physician should spend some time caring for the poorest of the poor, whether it be in a needy foreign country, Appalachia or rural America, or in the inner city, often just 30 minutes from our homes.
Surely it will help or change the patient’s life. But, too, it just might change your perspective, forever, and renew your passion for what we do – help others.
Dr. Hester is a hospitalist with Baltimore-Washington Medical Center who has a passion for empowering patients to partner in their health care. She is the creator of the Patient Whiz, a patient-engagement app for iOS.
I’m guilty. I feel a sense of entitlement, as many of us do. I have always had access to the best health care available. Even as a child, whenever I had a runny nose, my parents not only knew what to do but could afford whatever it took to make me feel better. And vaccine-preventable illnesses? They didn’t stand a chance at my house. I remember the day when I was a little girl and my father, a general practitioner, figured out my plan to wait him out as he stood patiently outside the bathroom door, vaccine in hand. He eventually got tired of waiting, barged in, oblivious to the fact that I was sitting on the potty (twiddling my fingers), and shot me right in arm. The nerve!
Many of us have no concept of what it is like to be ill. I mean really ill with no one to help, and no way to pay for that help even if we could find it. In the March 6, 2014, issue of the New England Journal of Medicine, authors of "Global Supply of Health Professionals" note that there’s a worldwide crisis of severe shortages, as well as maldistribution of health care professionals intensified by three global transitions: redistribution of the disability burden, demographic changes, and epidemiologic shifts. An estimated 25% of physicians in America come from other countries. Naturally, in some cases the countries of origin have their own health care challenges, so the trend to immigrate to America has significant potential to exacerbate an already critical shortage.
Of the estimated 9.2 million doctors worldwide, 8% reside in the United States. Of the 18.1 million nurses, 17% are in here. To put it in perspective, only 4% of the world’s population lives in the United States, yet we command a lion’s share of the global health care workforce, leaving the citizens of many other countries vulnerable to excessive suffering, premature death, and preventable diseases.
With all that America has to offer, I don’t see the pendulum shifting back in the other direction any time soon, but many of us are in a position to provide much needed health care to our brothers and sisters in even the most remote areas of the world.
As squeamish as I am about charting unknown territory, even I went on a medical missions trip to Nicaragua a few years ago and I will never forget it. We had to climb up the side of a mountain to get to a clearing that would hold the makeshift medical clinic. Of course, the locals came out in large numbers to receive much-needed medical care.
On the way back to the hotel I noticed some little boys tossing an object back and forth in the street. It seemed like pleasant fun until we got closer and I saw the object they were tossing was a dead rat. Even what we consider an inexpensive child’s toy is a luxury for many. If you consider salary and benefits, we hospitalists make more, on average, in a single shift than many Nicaraguans make in an entire year.
Every physician should spend some time caring for the poorest of the poor, whether it be in a needy foreign country, Appalachia or rural America, or in the inner city, often just 30 minutes from our homes.
Surely it will help or change the patient’s life. But, too, it just might change your perspective, forever, and renew your passion for what we do – help others.
Dr. Hester is a hospitalist with Baltimore-Washington Medical Center who has a passion for empowering patients to partner in their health care. She is the creator of the Patient Whiz, a patient-engagement app for iOS.
Are we really publishing too much in cardiovascular medicine?
At the recent International Congress on Peer Review and Biomedical Publication, researchers at Yale University, New Haven, Conn., presented data on 18,411 original papers published in 2006 from 144 journals in the cardiovascular discipline.
They found that the median number of citations was six; 15.0% of papers were never cited; and 33.3% had only one to five citations in the following 5 years. It was no surprise that the impact factor of the journal predicted subsequent citations: That’s probably one of the reasons why those journals have high impact factors. So what do the authors conclude? They state that their data suggest "substantial waste in some combination of the funding, pursuit, publication or dissemination of cardiovascular science."
An interesting thought but flawed. Sure, the number of publications has expanded, but so what? The cardiovascular medicine community is now very large. No one presupposes that all published papers are high quality, useful, or impactful. Sometimes a paper is redundant and poorly constructed, and may not have been subjected to rigorous peer review. But "diamonds in the rough" do exist, and that is why I sometimes read journals with rather paltry impact factors. New ideas or kernels of new ideas can often be found there. The same goes for abstracts at meetings.
Rather than express concern, we should argue that our field is filled with ideas, that the way forward is often incremental and that limiting publications would potentially damage progress. If any "solution" is needed, it will come from quality peer review and economics. After all, a journal with a minuscule impact factor is not likely to sustain itself on subscriptions and advertising dollars.
So, are we really publishing too much in cardiovascular medicine? Dr. Mort Kern, chief of cardiology at the University of California, Irvine, used to say that "every paper can find a home," but the publication process can be time consuming and difficult. Let’s continue to publish and keep the conversation going.
Dr. Paul Hauptman is professor of internal medicine and assistant dean of clinical-translational research at Saint Louis University and director of heart failure at Saint Louis University Hospital. He serves as an associate editor for Circulation: Heart Failure and blogs while staring out his office window at the Arch.
At the recent International Congress on Peer Review and Biomedical Publication, researchers at Yale University, New Haven, Conn., presented data on 18,411 original papers published in 2006 from 144 journals in the cardiovascular discipline.
They found that the median number of citations was six; 15.0% of papers were never cited; and 33.3% had only one to five citations in the following 5 years. It was no surprise that the impact factor of the journal predicted subsequent citations: That’s probably one of the reasons why those journals have high impact factors. So what do the authors conclude? They state that their data suggest "substantial waste in some combination of the funding, pursuit, publication or dissemination of cardiovascular science."
An interesting thought but flawed. Sure, the number of publications has expanded, but so what? The cardiovascular medicine community is now very large. No one presupposes that all published papers are high quality, useful, or impactful. Sometimes a paper is redundant and poorly constructed, and may not have been subjected to rigorous peer review. But "diamonds in the rough" do exist, and that is why I sometimes read journals with rather paltry impact factors. New ideas or kernels of new ideas can often be found there. The same goes for abstracts at meetings.
Rather than express concern, we should argue that our field is filled with ideas, that the way forward is often incremental and that limiting publications would potentially damage progress. If any "solution" is needed, it will come from quality peer review and economics. After all, a journal with a minuscule impact factor is not likely to sustain itself on subscriptions and advertising dollars.
So, are we really publishing too much in cardiovascular medicine? Dr. Mort Kern, chief of cardiology at the University of California, Irvine, used to say that "every paper can find a home," but the publication process can be time consuming and difficult. Let’s continue to publish and keep the conversation going.
Dr. Paul Hauptman is professor of internal medicine and assistant dean of clinical-translational research at Saint Louis University and director of heart failure at Saint Louis University Hospital. He serves as an associate editor for Circulation: Heart Failure and blogs while staring out his office window at the Arch.
At the recent International Congress on Peer Review and Biomedical Publication, researchers at Yale University, New Haven, Conn., presented data on 18,411 original papers published in 2006 from 144 journals in the cardiovascular discipline.
They found that the median number of citations was six; 15.0% of papers were never cited; and 33.3% had only one to five citations in the following 5 years. It was no surprise that the impact factor of the journal predicted subsequent citations: That’s probably one of the reasons why those journals have high impact factors. So what do the authors conclude? They state that their data suggest "substantial waste in some combination of the funding, pursuit, publication or dissemination of cardiovascular science."
An interesting thought but flawed. Sure, the number of publications has expanded, but so what? The cardiovascular medicine community is now very large. No one presupposes that all published papers are high quality, useful, or impactful. Sometimes a paper is redundant and poorly constructed, and may not have been subjected to rigorous peer review. But "diamonds in the rough" do exist, and that is why I sometimes read journals with rather paltry impact factors. New ideas or kernels of new ideas can often be found there. The same goes for abstracts at meetings.
Rather than express concern, we should argue that our field is filled with ideas, that the way forward is often incremental and that limiting publications would potentially damage progress. If any "solution" is needed, it will come from quality peer review and economics. After all, a journal with a minuscule impact factor is not likely to sustain itself on subscriptions and advertising dollars.
So, are we really publishing too much in cardiovascular medicine? Dr. Mort Kern, chief of cardiology at the University of California, Irvine, used to say that "every paper can find a home," but the publication process can be time consuming and difficult. Let’s continue to publish and keep the conversation going.
Dr. Paul Hauptman is professor of internal medicine and assistant dean of clinical-translational research at Saint Louis University and director of heart failure at Saint Louis University Hospital. He serves as an associate editor for Circulation: Heart Failure and blogs while staring out his office window at the Arch.
Office hours: subject to change
My office hours are possibly the most laid back in medicine. In a field like medicine, I try not to worry about such things.
I’ve intentionally never put regular hours on my office door or phone message, because in my small office they’re subject to change. If my secretary is tied up in traffic, the phone may not get rolled on time. But, as a general rule, we open between 8:45 and 9:00.
Once the starting gun goes off, I work straight through. No lunch break (sorry, drug reps). I’ve never been a lunch person, and would rather use the time for seeing people who need me. I see patients nonstop, squeezing in phone calls, refills, and test results in the cracks, and I try very hard to stay on time.
The day ends as quickly as it starts. As soon as the last patient is done, we’re done. Whether it’s at 4:00 (the usual time), 3:15, or (rarely) 2:00, we close. The phones are rolled, the lights out, and my secretary and I walk down to our cars.
I’m sure some feel we should be answering our phones until a specific time, but I don’t. I want to go pick up my kids. My secretary has a long drive home, and the sooner she hits the freeway, the better. If there are emergencies, patients will still be able to reach me. If they’re not having emergencies, they can call back when we reopen the next day. I’ll finish up the dictations when I come in early the next morning.
A medical practice is pretty stressful. There’s nothing wrong with calling it a day as soon as you can. And I do.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
My office hours are possibly the most laid back in medicine. In a field like medicine, I try not to worry about such things.
I’ve intentionally never put regular hours on my office door or phone message, because in my small office they’re subject to change. If my secretary is tied up in traffic, the phone may not get rolled on time. But, as a general rule, we open between 8:45 and 9:00.
Once the starting gun goes off, I work straight through. No lunch break (sorry, drug reps). I’ve never been a lunch person, and would rather use the time for seeing people who need me. I see patients nonstop, squeezing in phone calls, refills, and test results in the cracks, and I try very hard to stay on time.
The day ends as quickly as it starts. As soon as the last patient is done, we’re done. Whether it’s at 4:00 (the usual time), 3:15, or (rarely) 2:00, we close. The phones are rolled, the lights out, and my secretary and I walk down to our cars.
I’m sure some feel we should be answering our phones until a specific time, but I don’t. I want to go pick up my kids. My secretary has a long drive home, and the sooner she hits the freeway, the better. If there are emergencies, patients will still be able to reach me. If they’re not having emergencies, they can call back when we reopen the next day. I’ll finish up the dictations when I come in early the next morning.
A medical practice is pretty stressful. There’s nothing wrong with calling it a day as soon as you can. And I do.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
My office hours are possibly the most laid back in medicine. In a field like medicine, I try not to worry about such things.
I’ve intentionally never put regular hours on my office door or phone message, because in my small office they’re subject to change. If my secretary is tied up in traffic, the phone may not get rolled on time. But, as a general rule, we open between 8:45 and 9:00.
Once the starting gun goes off, I work straight through. No lunch break (sorry, drug reps). I’ve never been a lunch person, and would rather use the time for seeing people who need me. I see patients nonstop, squeezing in phone calls, refills, and test results in the cracks, and I try very hard to stay on time.
The day ends as quickly as it starts. As soon as the last patient is done, we’re done. Whether it’s at 4:00 (the usual time), 3:15, or (rarely) 2:00, we close. The phones are rolled, the lights out, and my secretary and I walk down to our cars.
I’m sure some feel we should be answering our phones until a specific time, but I don’t. I want to go pick up my kids. My secretary has a long drive home, and the sooner she hits the freeway, the better. If there are emergencies, patients will still be able to reach me. If they’re not having emergencies, they can call back when we reopen the next day. I’ll finish up the dictations when I come in early the next morning.
A medical practice is pretty stressful. There’s nothing wrong with calling it a day as soon as you can. And I do.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Natural supplements and the patient’s perspective
America’s use of natural supplements continues to rise, especially in those patients experiencing chronic mental illness. In fact, about 70% of children diagnosed with autism or attention-deficit/hyperactivity disorder use natural supplements or other complementary and alternative medicine (CAM) to augment their care. This is rarely the result of physician advice, though. Many parents simply seek out supplements on their own, rarely discussing them with their child’s psychiatrist. And the psychiatrists seldom think to ask. When it comes to talking about CAM, Dr. David M. Eisenberg and others have documented a "don’t ask, don’t tell" policy between medical professionals and patients (JAMA 1998;280:1569-75).
Outside of mental health, about 38% of Americans use natural supplements on a regular basis. They are typically female, over 30, well educated, and affluent. Children whose parents use CAM treatments are much more likely to use them, both now and in the future. Why do these individuals select CAM options? And why has this utilization continued to grow among families, especially those with chronic illness?
In 1998, John A. Astin, Ph.D., published a landmark article in JAMA asking this very question (1998;279:1548-53). He found that patients were drawn to CAM not by dissatisfaction with conventional medicine or a desire for more control, but rather because CAM better fit their value system. This is perhaps the best explanation for why research continues to document the hesitation of most Americans to take psychiatric drugs. "Americans believe that psychiatric medications are effective at relieving symptoms associated with mental disorders," wrote Dr. Ramin Mojtabai, "yet they are relatively unwilling to use them in most situations" (Psychiatr. Serv. 2009;60:1015-23) As you might expect, that unwillingness is high among parents of psychiatrically ill children, the same group that’s flocking to natural supplements.
It’s values, not empiricism or a physician’s advice, that inform many patients’ and parents’ treatment choices. And that phenomenon sees more and more people turning away from conventional medicine and toward the use of natural supplements. As a practitioner, think of it in terms of cross-cultural psychiatry. By virtue of their choice, CAM users reflect a different worldview, one that shares many of the underlying premises of holistic medicine.
Holistic medicine believes the body has innate healing power and that treatment works best when done with this force. Rather than impose hasty treatments or introduce new, untested agents, the CAM culture prefers to adjust the environmental milieu and lifestyle of the patient in order to gently foster a more positive, innate healing effect. Because of this, and in light of the fact that correctly prescribed medications cause more than 100,000 American deaths a year, safety is often more important than efficacy in the view of these patients.
Both critics and supporters are often quick to assume this safety vs. efficacy dichotomy separates conventional medicine from CAM in the United States. But the characterization is not totally accurate. CAM is not an alternative to conventional care, but a complement to it. This is has long been clear to patients, even if practitioners are slow on the uptick. More than 90% of patients diagnosed with a malignancy, for example, embrace the tools of CAM with their ongoing conventional treatments. Patients and parents appreciate our expertise with diagnosis, experience with significant mental illness, and extensive training. But they often want more.
Sadly, at-home CAM research can often be misguided or lead patients astray through hype and illegitimate claims. Research has consistently demonstrated that patients and parents do not ask their practitioners for advice or information about CAM out of a fear of condemnation, as well as a sense of practitioner bias and ignorance. In the era of Internet self-diagnosis, it’s up to practitioners to understand the mind-set of values-driven patients and create an environment where they feel comfortable bringing all options to the table. If we take the time to do this, we can more effectively reach them and build a strong therapeutic alliance based on common goals, open communication, and shared values.
A few questions can assist the practitioner in better understanding their patient’s worldview. Do you prefer to use natural supplements? What else do you do to support your health (or your child’s health)? Do you believe that the body has the ability to heal itself? Do you believe in holistic or natural medicine? Only after you have a basic grasp of the patient’s perspective can you start a real dialogue about healing. A few dismissive words or a roll of your eyes will shut communication down for good.
Remember, CAM is not only a treatment option but also represents a wide-ranging belief system. And like those who ascribe to any belief system, CAM users range from skeptical to unwavering. The challenge here, just as in cross-cultural psychiatry, is feeling out the individual features of that belief system, and then navigating it with respect and candor.
Most practitioners build trust and rapport with patients through a tried-and-true display of trust and understanding. Discussing natural supplements can provide just that, with a range of options for clinical conditions that are both evidence based and safe. Most of our patients are already using CAM modalities on their own without guidance. If the practitioner is able to incorporate these options into his or her treatment plan for the patient with a holistic or natural perspective, it increases the likelihood of positive rapport, good compliance, and open communication.
Dr. Shannon is an associate clinical professor in the department of psychiatry at the University of Colorado at Denver. He also is author of the textbook titled, Mental Health for the Whole Child (New York: Norton, 2013). He is past president of the American Board of Integrative Holistic Medicine.
America’s use of natural supplements continues to rise, especially in those patients experiencing chronic mental illness. In fact, about 70% of children diagnosed with autism or attention-deficit/hyperactivity disorder use natural supplements or other complementary and alternative medicine (CAM) to augment their care. This is rarely the result of physician advice, though. Many parents simply seek out supplements on their own, rarely discussing them with their child’s psychiatrist. And the psychiatrists seldom think to ask. When it comes to talking about CAM, Dr. David M. Eisenberg and others have documented a "don’t ask, don’t tell" policy between medical professionals and patients (JAMA 1998;280:1569-75).
Outside of mental health, about 38% of Americans use natural supplements on a regular basis. They are typically female, over 30, well educated, and affluent. Children whose parents use CAM treatments are much more likely to use them, both now and in the future. Why do these individuals select CAM options? And why has this utilization continued to grow among families, especially those with chronic illness?
In 1998, John A. Astin, Ph.D., published a landmark article in JAMA asking this very question (1998;279:1548-53). He found that patients were drawn to CAM not by dissatisfaction with conventional medicine or a desire for more control, but rather because CAM better fit their value system. This is perhaps the best explanation for why research continues to document the hesitation of most Americans to take psychiatric drugs. "Americans believe that psychiatric medications are effective at relieving symptoms associated with mental disorders," wrote Dr. Ramin Mojtabai, "yet they are relatively unwilling to use them in most situations" (Psychiatr. Serv. 2009;60:1015-23) As you might expect, that unwillingness is high among parents of psychiatrically ill children, the same group that’s flocking to natural supplements.
It’s values, not empiricism or a physician’s advice, that inform many patients’ and parents’ treatment choices. And that phenomenon sees more and more people turning away from conventional medicine and toward the use of natural supplements. As a practitioner, think of it in terms of cross-cultural psychiatry. By virtue of their choice, CAM users reflect a different worldview, one that shares many of the underlying premises of holistic medicine.
Holistic medicine believes the body has innate healing power and that treatment works best when done with this force. Rather than impose hasty treatments or introduce new, untested agents, the CAM culture prefers to adjust the environmental milieu and lifestyle of the patient in order to gently foster a more positive, innate healing effect. Because of this, and in light of the fact that correctly prescribed medications cause more than 100,000 American deaths a year, safety is often more important than efficacy in the view of these patients.
Both critics and supporters are often quick to assume this safety vs. efficacy dichotomy separates conventional medicine from CAM in the United States. But the characterization is not totally accurate. CAM is not an alternative to conventional care, but a complement to it. This is has long been clear to patients, even if practitioners are slow on the uptick. More than 90% of patients diagnosed with a malignancy, for example, embrace the tools of CAM with their ongoing conventional treatments. Patients and parents appreciate our expertise with diagnosis, experience with significant mental illness, and extensive training. But they often want more.
Sadly, at-home CAM research can often be misguided or lead patients astray through hype and illegitimate claims. Research has consistently demonstrated that patients and parents do not ask their practitioners for advice or information about CAM out of a fear of condemnation, as well as a sense of practitioner bias and ignorance. In the era of Internet self-diagnosis, it’s up to practitioners to understand the mind-set of values-driven patients and create an environment where they feel comfortable bringing all options to the table. If we take the time to do this, we can more effectively reach them and build a strong therapeutic alliance based on common goals, open communication, and shared values.
A few questions can assist the practitioner in better understanding their patient’s worldview. Do you prefer to use natural supplements? What else do you do to support your health (or your child’s health)? Do you believe that the body has the ability to heal itself? Do you believe in holistic or natural medicine? Only after you have a basic grasp of the patient’s perspective can you start a real dialogue about healing. A few dismissive words or a roll of your eyes will shut communication down for good.
Remember, CAM is not only a treatment option but also represents a wide-ranging belief system. And like those who ascribe to any belief system, CAM users range from skeptical to unwavering. The challenge here, just as in cross-cultural psychiatry, is feeling out the individual features of that belief system, and then navigating it with respect and candor.
Most practitioners build trust and rapport with patients through a tried-and-true display of trust and understanding. Discussing natural supplements can provide just that, with a range of options for clinical conditions that are both evidence based and safe. Most of our patients are already using CAM modalities on their own without guidance. If the practitioner is able to incorporate these options into his or her treatment plan for the patient with a holistic or natural perspective, it increases the likelihood of positive rapport, good compliance, and open communication.
Dr. Shannon is an associate clinical professor in the department of psychiatry at the University of Colorado at Denver. He also is author of the textbook titled, Mental Health for the Whole Child (New York: Norton, 2013). He is past president of the American Board of Integrative Holistic Medicine.
America’s use of natural supplements continues to rise, especially in those patients experiencing chronic mental illness. In fact, about 70% of children diagnosed with autism or attention-deficit/hyperactivity disorder use natural supplements or other complementary and alternative medicine (CAM) to augment their care. This is rarely the result of physician advice, though. Many parents simply seek out supplements on their own, rarely discussing them with their child’s psychiatrist. And the psychiatrists seldom think to ask. When it comes to talking about CAM, Dr. David M. Eisenberg and others have documented a "don’t ask, don’t tell" policy between medical professionals and patients (JAMA 1998;280:1569-75).
Outside of mental health, about 38% of Americans use natural supplements on a regular basis. They are typically female, over 30, well educated, and affluent. Children whose parents use CAM treatments are much more likely to use them, both now and in the future. Why do these individuals select CAM options? And why has this utilization continued to grow among families, especially those with chronic illness?
In 1998, John A. Astin, Ph.D., published a landmark article in JAMA asking this very question (1998;279:1548-53). He found that patients were drawn to CAM not by dissatisfaction with conventional medicine or a desire for more control, but rather because CAM better fit their value system. This is perhaps the best explanation for why research continues to document the hesitation of most Americans to take psychiatric drugs. "Americans believe that psychiatric medications are effective at relieving symptoms associated with mental disorders," wrote Dr. Ramin Mojtabai, "yet they are relatively unwilling to use them in most situations" (Psychiatr. Serv. 2009;60:1015-23) As you might expect, that unwillingness is high among parents of psychiatrically ill children, the same group that’s flocking to natural supplements.
It’s values, not empiricism or a physician’s advice, that inform many patients’ and parents’ treatment choices. And that phenomenon sees more and more people turning away from conventional medicine and toward the use of natural supplements. As a practitioner, think of it in terms of cross-cultural psychiatry. By virtue of their choice, CAM users reflect a different worldview, one that shares many of the underlying premises of holistic medicine.
Holistic medicine believes the body has innate healing power and that treatment works best when done with this force. Rather than impose hasty treatments or introduce new, untested agents, the CAM culture prefers to adjust the environmental milieu and lifestyle of the patient in order to gently foster a more positive, innate healing effect. Because of this, and in light of the fact that correctly prescribed medications cause more than 100,000 American deaths a year, safety is often more important than efficacy in the view of these patients.
Both critics and supporters are often quick to assume this safety vs. efficacy dichotomy separates conventional medicine from CAM in the United States. But the characterization is not totally accurate. CAM is not an alternative to conventional care, but a complement to it. This is has long been clear to patients, even if practitioners are slow on the uptick. More than 90% of patients diagnosed with a malignancy, for example, embrace the tools of CAM with their ongoing conventional treatments. Patients and parents appreciate our expertise with diagnosis, experience with significant mental illness, and extensive training. But they often want more.
Sadly, at-home CAM research can often be misguided or lead patients astray through hype and illegitimate claims. Research has consistently demonstrated that patients and parents do not ask their practitioners for advice or information about CAM out of a fear of condemnation, as well as a sense of practitioner bias and ignorance. In the era of Internet self-diagnosis, it’s up to practitioners to understand the mind-set of values-driven patients and create an environment where they feel comfortable bringing all options to the table. If we take the time to do this, we can more effectively reach them and build a strong therapeutic alliance based on common goals, open communication, and shared values.
A few questions can assist the practitioner in better understanding their patient’s worldview. Do you prefer to use natural supplements? What else do you do to support your health (or your child’s health)? Do you believe that the body has the ability to heal itself? Do you believe in holistic or natural medicine? Only after you have a basic grasp of the patient’s perspective can you start a real dialogue about healing. A few dismissive words or a roll of your eyes will shut communication down for good.
Remember, CAM is not only a treatment option but also represents a wide-ranging belief system. And like those who ascribe to any belief system, CAM users range from skeptical to unwavering. The challenge here, just as in cross-cultural psychiatry, is feeling out the individual features of that belief system, and then navigating it with respect and candor.
Most practitioners build trust and rapport with patients through a tried-and-true display of trust and understanding. Discussing natural supplements can provide just that, with a range of options for clinical conditions that are both evidence based and safe. Most of our patients are already using CAM modalities on their own without guidance. If the practitioner is able to incorporate these options into his or her treatment plan for the patient with a holistic or natural perspective, it increases the likelihood of positive rapport, good compliance, and open communication.
Dr. Shannon is an associate clinical professor in the department of psychiatry at the University of Colorado at Denver. He also is author of the textbook titled, Mental Health for the Whole Child (New York: Norton, 2013). He is past president of the American Board of Integrative Holistic Medicine.
Titanium dioxide
Titanium dioxide (TiO2) and zinc oxide (ZnO) in large-particle form have long been used in various sunscreens to protect the skin by reflecting or physically blocking ultraviolet (UV) radiation. In recent years, TiO2 as well as ZnO nanoparticles have been incorporated into sunscreens and cosmetics to act as a UV shield. They have been shown to be effective barriers against UV-induced damage, and yield stronger protection against UV insult, while leaving less white residue, than previous generations of the physical sunblocks.
However, some data suggest that in nanoparticle form, TiO2 and ZnO absorb UV radiation, leading to photocatalysis and the release of reactive oxygen species (Australas. J. Dermatol. 2011;52:1-6). This column will focus primarily on the safety of TiO2 in nanoparticle form.
While numerous studies examine both TiO2 and ZnO, the primary inorganic sunscreens, the sheer number of separate investigations warrants individual articles, and ZnO was addressed in previous columns. Briefly, though, TiO2 is more photoactive and exhibits a higher refractive index in visible light than ZnO (J. Am. Acad. Dermatol. 1999;40:85-90); therefore, TiO2 appears whiter and is more difficult to incorporate into transparent products.
A 2011 study by Kang et al. showed that TiO2 nanoparticles, but not normal-sized TiO2, and UVA synergistically foster rapid production of reactive oxygen species and breakdown of mitochondrial membrane potential, leading to apoptosis, and that TiO2 nanoparticles are more phototoxic than larger ones (Drug Chem. Toxicol. 2011;34:277-84).
However, also in 2011, Tyner et al. investigated the effects of nanoscale TiO2 use on UV attenuation in simple to complex sunscreen products. They found that barrier function was diminished by none of the formulations, and that optimal UV attenuation resulted when TiO2 particles were stabilized with a coating and evenly dispersed. The researchers concluded that nanoscale TiO2 is nontoxic and may impart greater efficacy (Int. J. Cosmet. Sci. 2011;33:234-44).
In vitro and in vivo studies
In 2010, Tiano et al. evaluated five modified TiO2 particles, developed and marketed for sunscreens. They used different in vitro models, including cultured human skin fibroblasts, to determine potential photocatalytic effects after UVA exposure. The investigators found that the kind of modification to and crystal form of the TiO2 nanoparticle influences its ability to augment or reduce DNA damage, increase or decrease intracellular reactive oxygen species, diminish cell viability, and promote other effects of photocatalysis. In particular, they noted that the anatase crystal form of TiO2 retained photocatalytic activity. The authors suggested that while the debate continues over the penetration of nanosized TiO2 into the viable epidermis, their results help elucidate the potential effects of TiO2 particles at the cellular level (Free Radic. Biol. Med. 2010;49:408-15).
A 2010 study by Senzui et al. using in vitro intact, stripped, and hair-removed skin of Yucatan micropigs to test the skin penetration of four different types of rutile (the most natural form of) TiO2 (two coated, two uncoated) revealed no penetration of TiO2 type in intact and stripped skin. The concentration of titanium in skin was significantly higher when one of the coated forms was applied on hair-removed skin, with titanium penetrating into vacant hair follicles (greater than 1 mm below the skin surface), but not into dermis or viable epidermis (J. Toxicol. Sci. 2010;35:107-13).
Animal studies
In 2009, the Food and Drug Administration Center for Drug Evaluation and Research worked with the National Center for Toxicology Research using minipigs and four sunscreen formulations to determine whether nanoscale TiO2 can penetrate intact skin. Their use of scanning electron microscopy and x-ray diffraction revealed that TiO2 particles were the same size as that observed for the raw materials, implying that the formulation process influenced neither the size nor the shape of TiO2 particles (Drug Dev. Ind. Pharm. 2009;35:1180-9).
In 2010, Sadrieh et al. performed a study of the dermal penetration of three TiO2 particles: uncoated submicrometer-sized, uncoated nano-sized, and dimethicone/methicone copolymer-coated nanosized. The investigators applied 5% by weight of each of the types of particles in a sunscreen on minipigs and found no significant penetration into intact normal epidermis (Toxicol. Sci. 2010;115(1):156-66).
In 2011, Furukawa et al. studied the postinitiation carcinogenic potential of coated and uncoated TiO2 nanoparticles in a two-stage skin carcinogenesis model using 7-week-old CD1 (ICR) female mice. They found that application of coated and uncoated nanoparticles after initiation and promotion with 7,12-dimethylbenz[a]anthracene and 12-O-tetradecanoylphorbol 13-acetate at doses of up to 20 mg/mouse failed to augment nodule development. The investigators concluded that TiO2 nanoparticles do not exhibit postinitiation potential for mouse skin carcinogenesis (Food Chem. Toxicol. 2011;49(4):744-9).
Human data
Given the persistent concerns about possible side effects of coated TiO2 and ZnO nanoparticles used in physical sun blockers, Filipe et al., in 2009, assessed the localization and potential skin penetration of TiO2 and ZnO nanoparticles dispersed in three sunscreen formulations, under realistic in vivo conditions in normal and altered skin. The investigators examined a test hydrophobic formulation containing coated 20-nm TiO2 nanoparticles and two commercially available sunscreen formulations containing TiO2 alone or in combination with ZnO, with respect to how consumers actually used sunscreens compared with the recommended standard condition for the sun protection factor test. They found that traces of the physical blockers could be detected only at the skin surface and uppermost area of the stratum corneum in normal human skin after a 2-hour exposure. After 48 hours of exposure, layers deeper than the stratum corneum contained no detectable TiO2 or ZnO nanoparticles. While preferential deposition of the nanoparticles in the openings of pilosebaceous follicles was noted, no penetration into viable skin tissue was observed. The investigators concluded that significant penetration of TiO2 or ZnO nanoparticles into keratinocytes is improbable (Skin Pharmacol. Physiol. 2009;22:266-75).
The weight of evidence
Current evidence suggests minimal risks to human health from the use of TiO2 or ZnO nanoparticles at concentrations up to 25% in cosmetic preparations or sunscreens, according to Schilling et al., regardless of coatings or crystalline structure. In a safety review of these ingredients, they noted that these nanoparticles formulated in topical products occur as aggregates of primary particles 30-150 nm in size, and bond in such a way that renders them impervious to the force of product application. Thus their structure remains unaffected, and no primary particles are released. The authors also noted that nanoparticles exhibit equivalence with larger particles in terms of distribution and duration and, therefore, recognition and elimination from the body (Photochem. Photobiol. Sci. 2010;9:495-509).
But in 2011, Tran and Salmon, in light of findings that nanoparticles may penetrate the stratum corneum under certain conditions, considered the possible photocarcinogenic results of nanoparticle sunscreens. They noted, though, that most such results were obtained through the use of animal skin models, not investigations with human skin (Australas. J. Dermatol. 2011;52:1-6). To this point, the weight of evidence appears to show that such TiO2 nanoparticles are safe when applied to intact human skin (Semin. Cutan. Med. Surg. 2011;30:210-13).
In response to the increased scrutiny and concern exhibited by the general public and government agencies regarding the safety of TiO2 and ZnO nanoparticles, Newman et al. reviewed the literature and position statements from 1980 to 2008 to ascertain and describe the use, safety, and regulatory state of such ingredients in sunscreens. They found no evidence of significant penetration deeper than the stratum corneum of TiO2 and ZnO nanoparticles, but caution that additional studies simulating real-world conditions (i.e., sunburned skin and under UV exposure) are necessary (J. Am. Acad. Dermatol. 2009;61:685-92).
Conclusion
Titanium dioxide is a well-established, safe, and effective physical sunblock. Nanotechnology has introduced some cause for concern regarding its use in physical sunblocks. In particular, evidence suggesting that photoexcitation of TiO2 nanoparticles leads to the generation of reactive oxygen species that damage DNA, potentially launching a cascade of adverse events, has prompted investigations into the safety of TiO2 in nanoparticle form. However, to date, multiple studies suggest that TiO2 nanoparticles do not penetrate or are highly unlikely to penetrate beyond the stratum corneum.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in Miami Beach. She founded the cosmetic dermatology center at the University of Miami in 1997. Dr. Baumann wrote the textbook "Cosmetic Dermatology: Principles and Practice" (McGraw-Hill, 2002), and a book for consumers, "The Skin Type Solution" (Bantam, 2006). Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Galderma, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Stiefel, Topix Pharmaceuticals, and Unilever.
Titanium dioxide (TiO2) and zinc oxide (ZnO) in large-particle form have long been used in various sunscreens to protect the skin by reflecting or physically blocking ultraviolet (UV) radiation. In recent years, TiO2 as well as ZnO nanoparticles have been incorporated into sunscreens and cosmetics to act as a UV shield. They have been shown to be effective barriers against UV-induced damage, and yield stronger protection against UV insult, while leaving less white residue, than previous generations of the physical sunblocks.
However, some data suggest that in nanoparticle form, TiO2 and ZnO absorb UV radiation, leading to photocatalysis and the release of reactive oxygen species (Australas. J. Dermatol. 2011;52:1-6). This column will focus primarily on the safety of TiO2 in nanoparticle form.
While numerous studies examine both TiO2 and ZnO, the primary inorganic sunscreens, the sheer number of separate investigations warrants individual articles, and ZnO was addressed in previous columns. Briefly, though, TiO2 is more photoactive and exhibits a higher refractive index in visible light than ZnO (J. Am. Acad. Dermatol. 1999;40:85-90); therefore, TiO2 appears whiter and is more difficult to incorporate into transparent products.
A 2011 study by Kang et al. showed that TiO2 nanoparticles, but not normal-sized TiO2, and UVA synergistically foster rapid production of reactive oxygen species and breakdown of mitochondrial membrane potential, leading to apoptosis, and that TiO2 nanoparticles are more phototoxic than larger ones (Drug Chem. Toxicol. 2011;34:277-84).
However, also in 2011, Tyner et al. investigated the effects of nanoscale TiO2 use on UV attenuation in simple to complex sunscreen products. They found that barrier function was diminished by none of the formulations, and that optimal UV attenuation resulted when TiO2 particles were stabilized with a coating and evenly dispersed. The researchers concluded that nanoscale TiO2 is nontoxic and may impart greater efficacy (Int. J. Cosmet. Sci. 2011;33:234-44).
In vitro and in vivo studies
In 2010, Tiano et al. evaluated five modified TiO2 particles, developed and marketed for sunscreens. They used different in vitro models, including cultured human skin fibroblasts, to determine potential photocatalytic effects after UVA exposure. The investigators found that the kind of modification to and crystal form of the TiO2 nanoparticle influences its ability to augment or reduce DNA damage, increase or decrease intracellular reactive oxygen species, diminish cell viability, and promote other effects of photocatalysis. In particular, they noted that the anatase crystal form of TiO2 retained photocatalytic activity. The authors suggested that while the debate continues over the penetration of nanosized TiO2 into the viable epidermis, their results help elucidate the potential effects of TiO2 particles at the cellular level (Free Radic. Biol. Med. 2010;49:408-15).
A 2010 study by Senzui et al. using in vitro intact, stripped, and hair-removed skin of Yucatan micropigs to test the skin penetration of four different types of rutile (the most natural form of) TiO2 (two coated, two uncoated) revealed no penetration of TiO2 type in intact and stripped skin. The concentration of titanium in skin was significantly higher when one of the coated forms was applied on hair-removed skin, with titanium penetrating into vacant hair follicles (greater than 1 mm below the skin surface), but not into dermis or viable epidermis (J. Toxicol. Sci. 2010;35:107-13).
Animal studies
In 2009, the Food and Drug Administration Center for Drug Evaluation and Research worked with the National Center for Toxicology Research using minipigs and four sunscreen formulations to determine whether nanoscale TiO2 can penetrate intact skin. Their use of scanning electron microscopy and x-ray diffraction revealed that TiO2 particles were the same size as that observed for the raw materials, implying that the formulation process influenced neither the size nor the shape of TiO2 particles (Drug Dev. Ind. Pharm. 2009;35:1180-9).
In 2010, Sadrieh et al. performed a study of the dermal penetration of three TiO2 particles: uncoated submicrometer-sized, uncoated nano-sized, and dimethicone/methicone copolymer-coated nanosized. The investigators applied 5% by weight of each of the types of particles in a sunscreen on minipigs and found no significant penetration into intact normal epidermis (Toxicol. Sci. 2010;115(1):156-66).
In 2011, Furukawa et al. studied the postinitiation carcinogenic potential of coated and uncoated TiO2 nanoparticles in a two-stage skin carcinogenesis model using 7-week-old CD1 (ICR) female mice. They found that application of coated and uncoated nanoparticles after initiation and promotion with 7,12-dimethylbenz[a]anthracene and 12-O-tetradecanoylphorbol 13-acetate at doses of up to 20 mg/mouse failed to augment nodule development. The investigators concluded that TiO2 nanoparticles do not exhibit postinitiation potential for mouse skin carcinogenesis (Food Chem. Toxicol. 2011;49(4):744-9).
Human data
Given the persistent concerns about possible side effects of coated TiO2 and ZnO nanoparticles used in physical sun blockers, Filipe et al., in 2009, assessed the localization and potential skin penetration of TiO2 and ZnO nanoparticles dispersed in three sunscreen formulations, under realistic in vivo conditions in normal and altered skin. The investigators examined a test hydrophobic formulation containing coated 20-nm TiO2 nanoparticles and two commercially available sunscreen formulations containing TiO2 alone or in combination with ZnO, with respect to how consumers actually used sunscreens compared with the recommended standard condition for the sun protection factor test. They found that traces of the physical blockers could be detected only at the skin surface and uppermost area of the stratum corneum in normal human skin after a 2-hour exposure. After 48 hours of exposure, layers deeper than the stratum corneum contained no detectable TiO2 or ZnO nanoparticles. While preferential deposition of the nanoparticles in the openings of pilosebaceous follicles was noted, no penetration into viable skin tissue was observed. The investigators concluded that significant penetration of TiO2 or ZnO nanoparticles into keratinocytes is improbable (Skin Pharmacol. Physiol. 2009;22:266-75).
The weight of evidence
Current evidence suggests minimal risks to human health from the use of TiO2 or ZnO nanoparticles at concentrations up to 25% in cosmetic preparations or sunscreens, according to Schilling et al., regardless of coatings or crystalline structure. In a safety review of these ingredients, they noted that these nanoparticles formulated in topical products occur as aggregates of primary particles 30-150 nm in size, and bond in such a way that renders them impervious to the force of product application. Thus their structure remains unaffected, and no primary particles are released. The authors also noted that nanoparticles exhibit equivalence with larger particles in terms of distribution and duration and, therefore, recognition and elimination from the body (Photochem. Photobiol. Sci. 2010;9:495-509).
But in 2011, Tran and Salmon, in light of findings that nanoparticles may penetrate the stratum corneum under certain conditions, considered the possible photocarcinogenic results of nanoparticle sunscreens. They noted, though, that most such results were obtained through the use of animal skin models, not investigations with human skin (Australas. J. Dermatol. 2011;52:1-6). To this point, the weight of evidence appears to show that such TiO2 nanoparticles are safe when applied to intact human skin (Semin. Cutan. Med. Surg. 2011;30:210-13).
In response to the increased scrutiny and concern exhibited by the general public and government agencies regarding the safety of TiO2 and ZnO nanoparticles, Newman et al. reviewed the literature and position statements from 1980 to 2008 to ascertain and describe the use, safety, and regulatory state of such ingredients in sunscreens. They found no evidence of significant penetration deeper than the stratum corneum of TiO2 and ZnO nanoparticles, but caution that additional studies simulating real-world conditions (i.e., sunburned skin and under UV exposure) are necessary (J. Am. Acad. Dermatol. 2009;61:685-92).
Conclusion
Titanium dioxide is a well-established, safe, and effective physical sunblock. Nanotechnology has introduced some cause for concern regarding its use in physical sunblocks. In particular, evidence suggesting that photoexcitation of TiO2 nanoparticles leads to the generation of reactive oxygen species that damage DNA, potentially launching a cascade of adverse events, has prompted investigations into the safety of TiO2 in nanoparticle form. However, to date, multiple studies suggest that TiO2 nanoparticles do not penetrate or are highly unlikely to penetrate beyond the stratum corneum.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in Miami Beach. She founded the cosmetic dermatology center at the University of Miami in 1997. Dr. Baumann wrote the textbook "Cosmetic Dermatology: Principles and Practice" (McGraw-Hill, 2002), and a book for consumers, "The Skin Type Solution" (Bantam, 2006). Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Galderma, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Stiefel, Topix Pharmaceuticals, and Unilever.
Titanium dioxide (TiO2) and zinc oxide (ZnO) in large-particle form have long been used in various sunscreens to protect the skin by reflecting or physically blocking ultraviolet (UV) radiation. In recent years, TiO2 as well as ZnO nanoparticles have been incorporated into sunscreens and cosmetics to act as a UV shield. They have been shown to be effective barriers against UV-induced damage, and yield stronger protection against UV insult, while leaving less white residue, than previous generations of the physical sunblocks.
However, some data suggest that in nanoparticle form, TiO2 and ZnO absorb UV radiation, leading to photocatalysis and the release of reactive oxygen species (Australas. J. Dermatol. 2011;52:1-6). This column will focus primarily on the safety of TiO2 in nanoparticle form.
While numerous studies examine both TiO2 and ZnO, the primary inorganic sunscreens, the sheer number of separate investigations warrants individual articles, and ZnO was addressed in previous columns. Briefly, though, TiO2 is more photoactive and exhibits a higher refractive index in visible light than ZnO (J. Am. Acad. Dermatol. 1999;40:85-90); therefore, TiO2 appears whiter and is more difficult to incorporate into transparent products.
A 2011 study by Kang et al. showed that TiO2 nanoparticles, but not normal-sized TiO2, and UVA synergistically foster rapid production of reactive oxygen species and breakdown of mitochondrial membrane potential, leading to apoptosis, and that TiO2 nanoparticles are more phototoxic than larger ones (Drug Chem. Toxicol. 2011;34:277-84).
However, also in 2011, Tyner et al. investigated the effects of nanoscale TiO2 use on UV attenuation in simple to complex sunscreen products. They found that barrier function was diminished by none of the formulations, and that optimal UV attenuation resulted when TiO2 particles were stabilized with a coating and evenly dispersed. The researchers concluded that nanoscale TiO2 is nontoxic and may impart greater efficacy (Int. J. Cosmet. Sci. 2011;33:234-44).
In vitro and in vivo studies
In 2010, Tiano et al. evaluated five modified TiO2 particles, developed and marketed for sunscreens. They used different in vitro models, including cultured human skin fibroblasts, to determine potential photocatalytic effects after UVA exposure. The investigators found that the kind of modification to and crystal form of the TiO2 nanoparticle influences its ability to augment or reduce DNA damage, increase or decrease intracellular reactive oxygen species, diminish cell viability, and promote other effects of photocatalysis. In particular, they noted that the anatase crystal form of TiO2 retained photocatalytic activity. The authors suggested that while the debate continues over the penetration of nanosized TiO2 into the viable epidermis, their results help elucidate the potential effects of TiO2 particles at the cellular level (Free Radic. Biol. Med. 2010;49:408-15).
A 2010 study by Senzui et al. using in vitro intact, stripped, and hair-removed skin of Yucatan micropigs to test the skin penetration of four different types of rutile (the most natural form of) TiO2 (two coated, two uncoated) revealed no penetration of TiO2 type in intact and stripped skin. The concentration of titanium in skin was significantly higher when one of the coated forms was applied on hair-removed skin, with titanium penetrating into vacant hair follicles (greater than 1 mm below the skin surface), but not into dermis or viable epidermis (J. Toxicol. Sci. 2010;35:107-13).
Animal studies
In 2009, the Food and Drug Administration Center for Drug Evaluation and Research worked with the National Center for Toxicology Research using minipigs and four sunscreen formulations to determine whether nanoscale TiO2 can penetrate intact skin. Their use of scanning electron microscopy and x-ray diffraction revealed that TiO2 particles were the same size as that observed for the raw materials, implying that the formulation process influenced neither the size nor the shape of TiO2 particles (Drug Dev. Ind. Pharm. 2009;35:1180-9).
In 2010, Sadrieh et al. performed a study of the dermal penetration of three TiO2 particles: uncoated submicrometer-sized, uncoated nano-sized, and dimethicone/methicone copolymer-coated nanosized. The investigators applied 5% by weight of each of the types of particles in a sunscreen on minipigs and found no significant penetration into intact normal epidermis (Toxicol. Sci. 2010;115(1):156-66).
In 2011, Furukawa et al. studied the postinitiation carcinogenic potential of coated and uncoated TiO2 nanoparticles in a two-stage skin carcinogenesis model using 7-week-old CD1 (ICR) female mice. They found that application of coated and uncoated nanoparticles after initiation and promotion with 7,12-dimethylbenz[a]anthracene and 12-O-tetradecanoylphorbol 13-acetate at doses of up to 20 mg/mouse failed to augment nodule development. The investigators concluded that TiO2 nanoparticles do not exhibit postinitiation potential for mouse skin carcinogenesis (Food Chem. Toxicol. 2011;49(4):744-9).
Human data
Given the persistent concerns about possible side effects of coated TiO2 and ZnO nanoparticles used in physical sun blockers, Filipe et al., in 2009, assessed the localization and potential skin penetration of TiO2 and ZnO nanoparticles dispersed in three sunscreen formulations, under realistic in vivo conditions in normal and altered skin. The investigators examined a test hydrophobic formulation containing coated 20-nm TiO2 nanoparticles and two commercially available sunscreen formulations containing TiO2 alone or in combination with ZnO, with respect to how consumers actually used sunscreens compared with the recommended standard condition for the sun protection factor test. They found that traces of the physical blockers could be detected only at the skin surface and uppermost area of the stratum corneum in normal human skin after a 2-hour exposure. After 48 hours of exposure, layers deeper than the stratum corneum contained no detectable TiO2 or ZnO nanoparticles. While preferential deposition of the nanoparticles in the openings of pilosebaceous follicles was noted, no penetration into viable skin tissue was observed. The investigators concluded that significant penetration of TiO2 or ZnO nanoparticles into keratinocytes is improbable (Skin Pharmacol. Physiol. 2009;22:266-75).
The weight of evidence
Current evidence suggests minimal risks to human health from the use of TiO2 or ZnO nanoparticles at concentrations up to 25% in cosmetic preparations or sunscreens, according to Schilling et al., regardless of coatings or crystalline structure. In a safety review of these ingredients, they noted that these nanoparticles formulated in topical products occur as aggregates of primary particles 30-150 nm in size, and bond in such a way that renders them impervious to the force of product application. Thus their structure remains unaffected, and no primary particles are released. The authors also noted that nanoparticles exhibit equivalence with larger particles in terms of distribution and duration and, therefore, recognition and elimination from the body (Photochem. Photobiol. Sci. 2010;9:495-509).
But in 2011, Tran and Salmon, in light of findings that nanoparticles may penetrate the stratum corneum under certain conditions, considered the possible photocarcinogenic results of nanoparticle sunscreens. They noted, though, that most such results were obtained through the use of animal skin models, not investigations with human skin (Australas. J. Dermatol. 2011;52:1-6). To this point, the weight of evidence appears to show that such TiO2 nanoparticles are safe when applied to intact human skin (Semin. Cutan. Med. Surg. 2011;30:210-13).
In response to the increased scrutiny and concern exhibited by the general public and government agencies regarding the safety of TiO2 and ZnO nanoparticles, Newman et al. reviewed the literature and position statements from 1980 to 2008 to ascertain and describe the use, safety, and regulatory state of such ingredients in sunscreens. They found no evidence of significant penetration deeper than the stratum corneum of TiO2 and ZnO nanoparticles, but caution that additional studies simulating real-world conditions (i.e., sunburned skin and under UV exposure) are necessary (J. Am. Acad. Dermatol. 2009;61:685-92).
Conclusion
Titanium dioxide is a well-established, safe, and effective physical sunblock. Nanotechnology has introduced some cause for concern regarding its use in physical sunblocks. In particular, evidence suggesting that photoexcitation of TiO2 nanoparticles leads to the generation of reactive oxygen species that damage DNA, potentially launching a cascade of adverse events, has prompted investigations into the safety of TiO2 in nanoparticle form. However, to date, multiple studies suggest that TiO2 nanoparticles do not penetrate or are highly unlikely to penetrate beyond the stratum corneum.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in Miami Beach. She founded the cosmetic dermatology center at the University of Miami in 1997. Dr. Baumann wrote the textbook "Cosmetic Dermatology: Principles and Practice" (McGraw-Hill, 2002), and a book for consumers, "The Skin Type Solution" (Bantam, 2006). Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Galderma, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Stiefel, Topix Pharmaceuticals, and Unilever.
Hair washing – Too much or too little?
Many dermatologists continue to battle an overwashing epidemic. From bar soaps to antibacterial washes, dermatologists continue to educate patients that the extensive lather, the alkaline pH, and the antibacterial components of our washing rituals can strip the natural oils from the skin and leave it dry, cracked, and damaged.
This phenomenon is well reported in the literature, and industry has taken notice by developing more "no-soap" soaps than ever before.
But does the same philosophy apply to hair care practices? Hair washing is more complicated, particularly in skin of color patients.
Overwashing the hair often leads to dry hair, split ends, and the need for compensatory conditioners to replace lost moisture. In African American hair, especially that of patients who use chemical or heat treatments, the lost oil and sebum from overwashing can cause even more damage.
Many skin of color patients wash their hair infrequently to protect it from breakage, and they may use topical oils to smooth and protect the fragile hair shaft.
However, can underwashing the scalp and hair cause problems? Yes, in some cases.
You might see African American patients in your practice who are suffering from scalp folliculitis, itchy scalp, seborrheic dermatitis, or alopecia that can be traced to infrequent hair washing. The infrequency of washing and the application of oils to the hair does help the hair shaft, but the buildup of oils and sebum on the scalp itself can lead to scalp inflammation, follicular plugging, extensive seborrhea, acneiform eruptions, and folliculitis.
Depending on its level and degree, the inflammation can cause pruritus and burning of the scalp and can even lead to temporary or permanent hair loss. Although topical and oral antibiotics, topical steroids, and medicated shampoos do help, proper washing also plays an important preventative role.
For skin of color patients with some of the chronic scalp problems mentioned above, decreasing heat and chemical treatments, along with increasing hair washing to two or three times a week can help prevent scalp dermatitides without compromising the hair integrity. In addition, the use of sulfate-free shampoos, use of shampoo on the scalp only (without lathering the ends of the hair), or use of a dry shampoo between washes can help control the oil and product buildup on the scalp itself.
Ultimately, it may take some trial and error to find the right hair washing regimen for skin of color patients. Determining how often to wash the scalp depends on many patient-specific factors including ethnicity, hair type, frequency of chemical and heat treatments, cost, and level of scalp inflammation. Experimenting with new hair care products and possibly a new hairstyle also may be part of a successful treatment plan.
Dr. Talakoub is in private practice at McLean (Va.) Dermatology Center. A graduate of Boston University School of Medicine, Dr. Talakoub did her residency in dermatology at the University of California, San Francisco. She is the author of multiple scholarly articles and a textbook chapter.
Many dermatologists continue to battle an overwashing epidemic. From bar soaps to antibacterial washes, dermatologists continue to educate patients that the extensive lather, the alkaline pH, and the antibacterial components of our washing rituals can strip the natural oils from the skin and leave it dry, cracked, and damaged.
This phenomenon is well reported in the literature, and industry has taken notice by developing more "no-soap" soaps than ever before.
But does the same philosophy apply to hair care practices? Hair washing is more complicated, particularly in skin of color patients.
Overwashing the hair often leads to dry hair, split ends, and the need for compensatory conditioners to replace lost moisture. In African American hair, especially that of patients who use chemical or heat treatments, the lost oil and sebum from overwashing can cause even more damage.
Many skin of color patients wash their hair infrequently to protect it from breakage, and they may use topical oils to smooth and protect the fragile hair shaft.
However, can underwashing the scalp and hair cause problems? Yes, in some cases.
You might see African American patients in your practice who are suffering from scalp folliculitis, itchy scalp, seborrheic dermatitis, or alopecia that can be traced to infrequent hair washing. The infrequency of washing and the application of oils to the hair does help the hair shaft, but the buildup of oils and sebum on the scalp itself can lead to scalp inflammation, follicular plugging, extensive seborrhea, acneiform eruptions, and folliculitis.
Depending on its level and degree, the inflammation can cause pruritus and burning of the scalp and can even lead to temporary or permanent hair loss. Although topical and oral antibiotics, topical steroids, and medicated shampoos do help, proper washing also plays an important preventative role.
For skin of color patients with some of the chronic scalp problems mentioned above, decreasing heat and chemical treatments, along with increasing hair washing to two or three times a week can help prevent scalp dermatitides without compromising the hair integrity. In addition, the use of sulfate-free shampoos, use of shampoo on the scalp only (without lathering the ends of the hair), or use of a dry shampoo between washes can help control the oil and product buildup on the scalp itself.
Ultimately, it may take some trial and error to find the right hair washing regimen for skin of color patients. Determining how often to wash the scalp depends on many patient-specific factors including ethnicity, hair type, frequency of chemical and heat treatments, cost, and level of scalp inflammation. Experimenting with new hair care products and possibly a new hairstyle also may be part of a successful treatment plan.
Dr. Talakoub is in private practice at McLean (Va.) Dermatology Center. A graduate of Boston University School of Medicine, Dr. Talakoub did her residency in dermatology at the University of California, San Francisco. She is the author of multiple scholarly articles and a textbook chapter.
Many dermatologists continue to battle an overwashing epidemic. From bar soaps to antibacterial washes, dermatologists continue to educate patients that the extensive lather, the alkaline pH, and the antibacterial components of our washing rituals can strip the natural oils from the skin and leave it dry, cracked, and damaged.
This phenomenon is well reported in the literature, and industry has taken notice by developing more "no-soap" soaps than ever before.
But does the same philosophy apply to hair care practices? Hair washing is more complicated, particularly in skin of color patients.
Overwashing the hair often leads to dry hair, split ends, and the need for compensatory conditioners to replace lost moisture. In African American hair, especially that of patients who use chemical or heat treatments, the lost oil and sebum from overwashing can cause even more damage.
Many skin of color patients wash their hair infrequently to protect it from breakage, and they may use topical oils to smooth and protect the fragile hair shaft.
However, can underwashing the scalp and hair cause problems? Yes, in some cases.
You might see African American patients in your practice who are suffering from scalp folliculitis, itchy scalp, seborrheic dermatitis, or alopecia that can be traced to infrequent hair washing. The infrequency of washing and the application of oils to the hair does help the hair shaft, but the buildup of oils and sebum on the scalp itself can lead to scalp inflammation, follicular plugging, extensive seborrhea, acneiform eruptions, and folliculitis.
Depending on its level and degree, the inflammation can cause pruritus and burning of the scalp and can even lead to temporary or permanent hair loss. Although topical and oral antibiotics, topical steroids, and medicated shampoos do help, proper washing also plays an important preventative role.
For skin of color patients with some of the chronic scalp problems mentioned above, decreasing heat and chemical treatments, along with increasing hair washing to two or three times a week can help prevent scalp dermatitides without compromising the hair integrity. In addition, the use of sulfate-free shampoos, use of shampoo on the scalp only (without lathering the ends of the hair), or use of a dry shampoo between washes can help control the oil and product buildup on the scalp itself.
Ultimately, it may take some trial and error to find the right hair washing regimen for skin of color patients. Determining how often to wash the scalp depends on many patient-specific factors including ethnicity, hair type, frequency of chemical and heat treatments, cost, and level of scalp inflammation. Experimenting with new hair care products and possibly a new hairstyle also may be part of a successful treatment plan.
Dr. Talakoub is in private practice at McLean (Va.) Dermatology Center. A graduate of Boston University School of Medicine, Dr. Talakoub did her residency in dermatology at the University of California, San Francisco. She is the author of multiple scholarly articles and a textbook chapter.