User login
Cannabis crimps teen cognitive development
BARCELONA – What would you predict has a greater detrimental effect on adolescent cognitive development: alcohol or cannabis use?
The evidence-based answer may come as a surprise. It certainly did for Patricia Conrod, PhD, who led the large population-based study that addressed the question.
“Generally, we found no effect of alcohol on cognitive development, which was a huge surprise to us. It might be related to the fact that the quantity of alcohol consumption in this young sample just wasn’t high enough to produce significant effects on cognitive development. But, to our surprise, we found rather significant effects of cannabis use on cognitive development,” she said at the annual congress of the European College of Neuropsychopharmacology.
Indeed, cannabis use proved to have detrimental effects on all four cognitive domains assessed in the study: working memory, perceptual reasoning, delayed recall, and inhibitory control, reported Dr. Conrod, professor of psychiatry at the University of Montreal.
Her recently study, published in the American Journal of Psychiatry, included 3,826 seventh-grade students at 31 Montreal-area schools. They constituted 5% of all students entering that grade in the greater Montreal area. Participants were prospectively assessed annually for 4 years regarding their use or nonuse of alcohol or cannabis and also underwent neurocognitive testing on the four domains of interest. The assessments were done on school computers with preservation of student confidentiality. Investigators used a Big Data approach to model the relationship between the extent of substance use and neurocognitive function variables over time.
Abstinent students were the best performers on the neurocognitive testing. Cannabis use, but not alcohol, in a given year was associated with concurrent adverse effects on all four cognitive domains. In addition, cannabis use showed evidence of having a neurotoxic lag effect on inhibitory control and working memory. This took the form of a lasting effect: A student who reported using cannabis 1 year but not the next showed impairment of inhibitory control and working memory during both years. And a student who used cannabis both years was even more impaired in those domains.
Dr. Conrod found the evidence of a neurotoxic effect of cannabis use on inhibitory control to be of particular concern because in earlier studies she established that impaired inhibitory control is a strong independent risk factor for subsequent substance use disorders.
”So what we’re seeing is indeed that early onset substance use is interfering with cognitive development, which now sets us up to be able to answer the question of whether evidence-based prevention protects cognitive development by delaying early onset of substance use. And over the longer term, does that protect young people against addiction?”
Dr. Conrod and her coworkers are now in the process of obtaining answers to those questions in the large ongoing Canadian Institutes of Health Research-funded Co-Venture Trial. This randomized trial involving thousands of adolescent students used the investigators’ Preventure Program, a school-based, personality-targeted intervention for prevention of substance use and abuse.
The Preventure Program involves two 90-minute group sessions of manual-based cognitive-behavioral therapy. Students are invited to participate if they score at least one standard deviation above the school mean on one of four personality traits that have been shown to increase the risk of substance misuse and psychiatric disorders. The four personality traits are sensation seeking, impulsivity, anxiety sensitivity, and hopelessness. Typically, about 45% of students met that threshold, and 85% of those invited to participate in the program volunteered to do so. Students of similar personality type are grouped together for the targeted therapy sessions.
This brief coping skills intervention has been shown in multiple randomized trials around the world to reduce the likelihood of substance use in at-risk adolescents. For example, in an early trial involving 732 high school students in London, participation in the Preventure Program was associated with a 30% reduction in the likelihood of taking up the use of cannabis within the next 2 years, an 80% reduction in the likelihood of taking up cocaine, and a 50% reduction in the use of other drugs (Arch Gen Psychiatry. 2010 Jan;67[1]:85-93).
[email protected]
SOURCE: Conrod P. Am J Psychiatry. 2018 Oct 3. doi: 10.1176/appi.ajp.2018.18020202.
BARCELONA – What would you predict has a greater detrimental effect on adolescent cognitive development: alcohol or cannabis use?
The evidence-based answer may come as a surprise. It certainly did for Patricia Conrod, PhD, who led the large population-based study that addressed the question.
“Generally, we found no effect of alcohol on cognitive development, which was a huge surprise to us. It might be related to the fact that the quantity of alcohol consumption in this young sample just wasn’t high enough to produce significant effects on cognitive development. But, to our surprise, we found rather significant effects of cannabis use on cognitive development,” she said at the annual congress of the European College of Neuropsychopharmacology.
Indeed, cannabis use proved to have detrimental effects on all four cognitive domains assessed in the study: working memory, perceptual reasoning, delayed recall, and inhibitory control, reported Dr. Conrod, professor of psychiatry at the University of Montreal.
Her recently study, published in the American Journal of Psychiatry, included 3,826 seventh-grade students at 31 Montreal-area schools. They constituted 5% of all students entering that grade in the greater Montreal area. Participants were prospectively assessed annually for 4 years regarding their use or nonuse of alcohol or cannabis and also underwent neurocognitive testing on the four domains of interest. The assessments were done on school computers with preservation of student confidentiality. Investigators used a Big Data approach to model the relationship between the extent of substance use and neurocognitive function variables over time.
Abstinent students were the best performers on the neurocognitive testing. Cannabis use, but not alcohol, in a given year was associated with concurrent adverse effects on all four cognitive domains. In addition, cannabis use showed evidence of having a neurotoxic lag effect on inhibitory control and working memory. This took the form of a lasting effect: A student who reported using cannabis 1 year but not the next showed impairment of inhibitory control and working memory during both years. And a student who used cannabis both years was even more impaired in those domains.
Dr. Conrod found the evidence of a neurotoxic effect of cannabis use on inhibitory control to be of particular concern because in earlier studies she established that impaired inhibitory control is a strong independent risk factor for subsequent substance use disorders.
”So what we’re seeing is indeed that early onset substance use is interfering with cognitive development, which now sets us up to be able to answer the question of whether evidence-based prevention protects cognitive development by delaying early onset of substance use. And over the longer term, does that protect young people against addiction?”
Dr. Conrod and her coworkers are now in the process of obtaining answers to those questions in the large ongoing Canadian Institutes of Health Research-funded Co-Venture Trial. This randomized trial involving thousands of adolescent students used the investigators’ Preventure Program, a school-based, personality-targeted intervention for prevention of substance use and abuse.
The Preventure Program involves two 90-minute group sessions of manual-based cognitive-behavioral therapy. Students are invited to participate if they score at least one standard deviation above the school mean on one of four personality traits that have been shown to increase the risk of substance misuse and psychiatric disorders. The four personality traits are sensation seeking, impulsivity, anxiety sensitivity, and hopelessness. Typically, about 45% of students met that threshold, and 85% of those invited to participate in the program volunteered to do so. Students of similar personality type are grouped together for the targeted therapy sessions.
This brief coping skills intervention has been shown in multiple randomized trials around the world to reduce the likelihood of substance use in at-risk adolescents. For example, in an early trial involving 732 high school students in London, participation in the Preventure Program was associated with a 30% reduction in the likelihood of taking up the use of cannabis within the next 2 years, an 80% reduction in the likelihood of taking up cocaine, and a 50% reduction in the use of other drugs (Arch Gen Psychiatry. 2010 Jan;67[1]:85-93).
[email protected]
SOURCE: Conrod P. Am J Psychiatry. 2018 Oct 3. doi: 10.1176/appi.ajp.2018.18020202.
BARCELONA – What would you predict has a greater detrimental effect on adolescent cognitive development: alcohol or cannabis use?
The evidence-based answer may come as a surprise. It certainly did for Patricia Conrod, PhD, who led the large population-based study that addressed the question.
“Generally, we found no effect of alcohol on cognitive development, which was a huge surprise to us. It might be related to the fact that the quantity of alcohol consumption in this young sample just wasn’t high enough to produce significant effects on cognitive development. But, to our surprise, we found rather significant effects of cannabis use on cognitive development,” she said at the annual congress of the European College of Neuropsychopharmacology.
Indeed, cannabis use proved to have detrimental effects on all four cognitive domains assessed in the study: working memory, perceptual reasoning, delayed recall, and inhibitory control, reported Dr. Conrod, professor of psychiatry at the University of Montreal.
Her recently study, published in the American Journal of Psychiatry, included 3,826 seventh-grade students at 31 Montreal-area schools. They constituted 5% of all students entering that grade in the greater Montreal area. Participants were prospectively assessed annually for 4 years regarding their use or nonuse of alcohol or cannabis and also underwent neurocognitive testing on the four domains of interest. The assessments were done on school computers with preservation of student confidentiality. Investigators used a Big Data approach to model the relationship between the extent of substance use and neurocognitive function variables over time.
Abstinent students were the best performers on the neurocognitive testing. Cannabis use, but not alcohol, in a given year was associated with concurrent adverse effects on all four cognitive domains. In addition, cannabis use showed evidence of having a neurotoxic lag effect on inhibitory control and working memory. This took the form of a lasting effect: A student who reported using cannabis 1 year but not the next showed impairment of inhibitory control and working memory during both years. And a student who used cannabis both years was even more impaired in those domains.
Dr. Conrod found the evidence of a neurotoxic effect of cannabis use on inhibitory control to be of particular concern because in earlier studies she established that impaired inhibitory control is a strong independent risk factor for subsequent substance use disorders.
”So what we’re seeing is indeed that early onset substance use is interfering with cognitive development, which now sets us up to be able to answer the question of whether evidence-based prevention protects cognitive development by delaying early onset of substance use. And over the longer term, does that protect young people against addiction?”
Dr. Conrod and her coworkers are now in the process of obtaining answers to those questions in the large ongoing Canadian Institutes of Health Research-funded Co-Venture Trial. This randomized trial involving thousands of adolescent students used the investigators’ Preventure Program, a school-based, personality-targeted intervention for prevention of substance use and abuse.
The Preventure Program involves two 90-minute group sessions of manual-based cognitive-behavioral therapy. Students are invited to participate if they score at least one standard deviation above the school mean on one of four personality traits that have been shown to increase the risk of substance misuse and psychiatric disorders. The four personality traits are sensation seeking, impulsivity, anxiety sensitivity, and hopelessness. Typically, about 45% of students met that threshold, and 85% of those invited to participate in the program volunteered to do so. Students of similar personality type are grouped together for the targeted therapy sessions.
This brief coping skills intervention has been shown in multiple randomized trials around the world to reduce the likelihood of substance use in at-risk adolescents. For example, in an early trial involving 732 high school students in London, participation in the Preventure Program was associated with a 30% reduction in the likelihood of taking up the use of cannabis within the next 2 years, an 80% reduction in the likelihood of taking up cocaine, and a 50% reduction in the use of other drugs (Arch Gen Psychiatry. 2010 Jan;67[1]:85-93).
[email protected]
SOURCE: Conrod P. Am J Psychiatry. 2018 Oct 3. doi: 10.1176/appi.ajp.2018.18020202.
REPORTING FROM THE ECNP CONGRESS
Key clinical point:
Major finding: The observed neurotoxic effect on impulse control may spell future trouble.
Study details: This population-based study included 3,826 Montreal-area seventh graders who were prospectively assessed annually for 4 years regarding their cannabis and alcohol use and also underwent neurocognitive testing.
Disclosures: The study was funded by the Canadian Institutes of Health Research.
Source: Conrod P. Am J Psychiatry. 2018 Oct 3. doi: 10.1176/appi.ajp.2018.18020202.
High systemic inflammation at midlife portends cognitive changes in later life
ATLANTA – results from a long-term analysis showed.
“There is considerable evidence suggesting that abnormal immune functioning and inflammation may influence cognitive functioning and promote dementia,” lead study author Keenan Walker, PhD, said in an interview in advance of the annual meeting of the American Neurological Association. “For example, several studies have found higher levels of inflammatory markers in the blood and cerebral spinal fluid of patients with dementia, compared to nondemented individuals of a comparable age. What is less clear, however, is whether inflammation actually promotes cognitive decline or occurs simply as a result of the brain changes underlying dementia.”
To help answer this question, Dr. Walker, of Johns Hopkins University, Baltimore, and his colleagues evaluated blood biomarkers of inflammation in 12,727 middle-aged participants during visits one and two of the Atherosclerosis Risk in Communities (ARIC) study, a prospective epidemiologic analysis conducted in four U.S. communities and funded by the National Heart, Lung, and Blood Institute. Visit one occurred between 1987 and 1989, while visit two took place between 1990 and 1992. The researchers related these markers to cognitive change over the subsequent decades. Specifically, they used four biomarkers (fibrinogen, white blood cell count, von Willebrand factor, and Factor VIII) to create an inflammation composite score at visit one, and measured C-reactive protein (CRP) at visit two. Next, they used measures of memory, executive function, and language assess cognition over three visits spanning 20 years.
The average age of study participants at first cognitive assessment was 57 years, 56% were women, and 21% were black. After controlling for differences in demographic variables, vascular risk factors and comorbidities, the researchers observed that each standard deviation (SD) increase in the midlife inflammation composite score was associated with an additional 20-year global cognitive decline of –0.035 SD (P less than .01). They found a similar association between each SD higher midlife CRP level and additional 20-year decline in global cognition (–0.038 SD; P less than .01). In addition, study participants with a midlife inflammation composite score in the top quartile had a 7.6% steeper decline in global cognition, compared with participants in the lowest quartile; a similar association was observed for CRP.
“We were surprised at what we found when we looked at inflammation’s effect on individual cognitive domains,” Dr. Walker said. “Specifically, we found that inflammation was associated with declines in memory, but not declines in other domains, such as executive function or language. One possible interpretation for these findings is that inflammation may selectively influence brain regions such as the hippocampus, which are necessary for memory consolidation and are also most vulnerable to Alzheimer’s disease.” He added that the current findings “provide support for the idea that systemic inflammation may have an early pathogenic role in the cognitive decline that occurs in the decades leading up to older adulthood.”
Dr. Walker acknowledged certain limitations of the study, including the attrition of participants because of deaths and dropouts over the 20-year follow-up period. “Because high levels of inflammation and comorbid disease are related to death and risk of study dropout, the sample of participants who completed the entirety of the study may represent a group that is healthier overall than the general population,” he said. “However, we took several steps to reduce any potential attrition bias using advanced statistical techniques.”
Dr. Walker disclosed that he receives funding support from the National Institutes of Health.
Source: Walker et al. ANA 2018, Abstract 305.
ATLANTA – results from a long-term analysis showed.
“There is considerable evidence suggesting that abnormal immune functioning and inflammation may influence cognitive functioning and promote dementia,” lead study author Keenan Walker, PhD, said in an interview in advance of the annual meeting of the American Neurological Association. “For example, several studies have found higher levels of inflammatory markers in the blood and cerebral spinal fluid of patients with dementia, compared to nondemented individuals of a comparable age. What is less clear, however, is whether inflammation actually promotes cognitive decline or occurs simply as a result of the brain changes underlying dementia.”
To help answer this question, Dr. Walker, of Johns Hopkins University, Baltimore, and his colleagues evaluated blood biomarkers of inflammation in 12,727 middle-aged participants during visits one and two of the Atherosclerosis Risk in Communities (ARIC) study, a prospective epidemiologic analysis conducted in four U.S. communities and funded by the National Heart, Lung, and Blood Institute. Visit one occurred between 1987 and 1989, while visit two took place between 1990 and 1992. The researchers related these markers to cognitive change over the subsequent decades. Specifically, they used four biomarkers (fibrinogen, white blood cell count, von Willebrand factor, and Factor VIII) to create an inflammation composite score at visit one, and measured C-reactive protein (CRP) at visit two. Next, they used measures of memory, executive function, and language assess cognition over three visits spanning 20 years.
The average age of study participants at first cognitive assessment was 57 years, 56% were women, and 21% were black. After controlling for differences in demographic variables, vascular risk factors and comorbidities, the researchers observed that each standard deviation (SD) increase in the midlife inflammation composite score was associated with an additional 20-year global cognitive decline of –0.035 SD (P less than .01). They found a similar association between each SD higher midlife CRP level and additional 20-year decline in global cognition (–0.038 SD; P less than .01). In addition, study participants with a midlife inflammation composite score in the top quartile had a 7.6% steeper decline in global cognition, compared with participants in the lowest quartile; a similar association was observed for CRP.
“We were surprised at what we found when we looked at inflammation’s effect on individual cognitive domains,” Dr. Walker said. “Specifically, we found that inflammation was associated with declines in memory, but not declines in other domains, such as executive function or language. One possible interpretation for these findings is that inflammation may selectively influence brain regions such as the hippocampus, which are necessary for memory consolidation and are also most vulnerable to Alzheimer’s disease.” He added that the current findings “provide support for the idea that systemic inflammation may have an early pathogenic role in the cognitive decline that occurs in the decades leading up to older adulthood.”
Dr. Walker acknowledged certain limitations of the study, including the attrition of participants because of deaths and dropouts over the 20-year follow-up period. “Because high levels of inflammation and comorbid disease are related to death and risk of study dropout, the sample of participants who completed the entirety of the study may represent a group that is healthier overall than the general population,” he said. “However, we took several steps to reduce any potential attrition bias using advanced statistical techniques.”
Dr. Walker disclosed that he receives funding support from the National Institutes of Health.
Source: Walker et al. ANA 2018, Abstract 305.
ATLANTA – results from a long-term analysis showed.
“There is considerable evidence suggesting that abnormal immune functioning and inflammation may influence cognitive functioning and promote dementia,” lead study author Keenan Walker, PhD, said in an interview in advance of the annual meeting of the American Neurological Association. “For example, several studies have found higher levels of inflammatory markers in the blood and cerebral spinal fluid of patients with dementia, compared to nondemented individuals of a comparable age. What is less clear, however, is whether inflammation actually promotes cognitive decline or occurs simply as a result of the brain changes underlying dementia.”
To help answer this question, Dr. Walker, of Johns Hopkins University, Baltimore, and his colleagues evaluated blood biomarkers of inflammation in 12,727 middle-aged participants during visits one and two of the Atherosclerosis Risk in Communities (ARIC) study, a prospective epidemiologic analysis conducted in four U.S. communities and funded by the National Heart, Lung, and Blood Institute. Visit one occurred between 1987 and 1989, while visit two took place between 1990 and 1992. The researchers related these markers to cognitive change over the subsequent decades. Specifically, they used four biomarkers (fibrinogen, white blood cell count, von Willebrand factor, and Factor VIII) to create an inflammation composite score at visit one, and measured C-reactive protein (CRP) at visit two. Next, they used measures of memory, executive function, and language assess cognition over three visits spanning 20 years.
The average age of study participants at first cognitive assessment was 57 years, 56% were women, and 21% were black. After controlling for differences in demographic variables, vascular risk factors and comorbidities, the researchers observed that each standard deviation (SD) increase in the midlife inflammation composite score was associated with an additional 20-year global cognitive decline of –0.035 SD (P less than .01). They found a similar association between each SD higher midlife CRP level and additional 20-year decline in global cognition (–0.038 SD; P less than .01). In addition, study participants with a midlife inflammation composite score in the top quartile had a 7.6% steeper decline in global cognition, compared with participants in the lowest quartile; a similar association was observed for CRP.
“We were surprised at what we found when we looked at inflammation’s effect on individual cognitive domains,” Dr. Walker said. “Specifically, we found that inflammation was associated with declines in memory, but not declines in other domains, such as executive function or language. One possible interpretation for these findings is that inflammation may selectively influence brain regions such as the hippocampus, which are necessary for memory consolidation and are also most vulnerable to Alzheimer’s disease.” He added that the current findings “provide support for the idea that systemic inflammation may have an early pathogenic role in the cognitive decline that occurs in the decades leading up to older adulthood.”
Dr. Walker acknowledged certain limitations of the study, including the attrition of participants because of deaths and dropouts over the 20-year follow-up period. “Because high levels of inflammation and comorbid disease are related to death and risk of study dropout, the sample of participants who completed the entirety of the study may represent a group that is healthier overall than the general population,” he said. “However, we took several steps to reduce any potential attrition bias using advanced statistical techniques.”
Dr. Walker disclosed that he receives funding support from the National Institutes of Health.
Source: Walker et al. ANA 2018, Abstract 305.
REPORTING FROM ANA 2018
Key clinical point: Inflammation was associated with declines in memory, but not declines in other domains, such as executive function or language.
Major finding: Each standard deviation (SD) increase in the midlife inflammation composite score was associated with an additional 20-year global cognitive decline of –0.035 SD (P less than .01).
Study details: An evaluation of blood biomarkers of inflammation in 12,727 middle-aged participants in the Atherosclerosis Risk in Communities Study.
Disclosures: Dr. Walker disclosed that he receives funding support from the National Institutes of Health.
Source: Walker et al. ANA 2018, Abstract 305.
Cross-contamination of Pathology Specimens: A Cautionary Tale
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
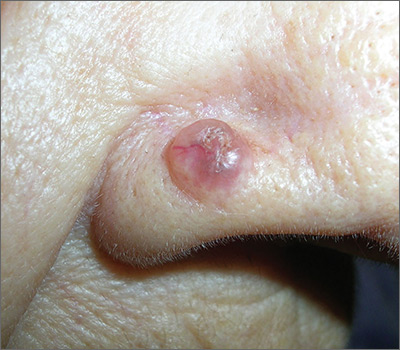
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.
The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
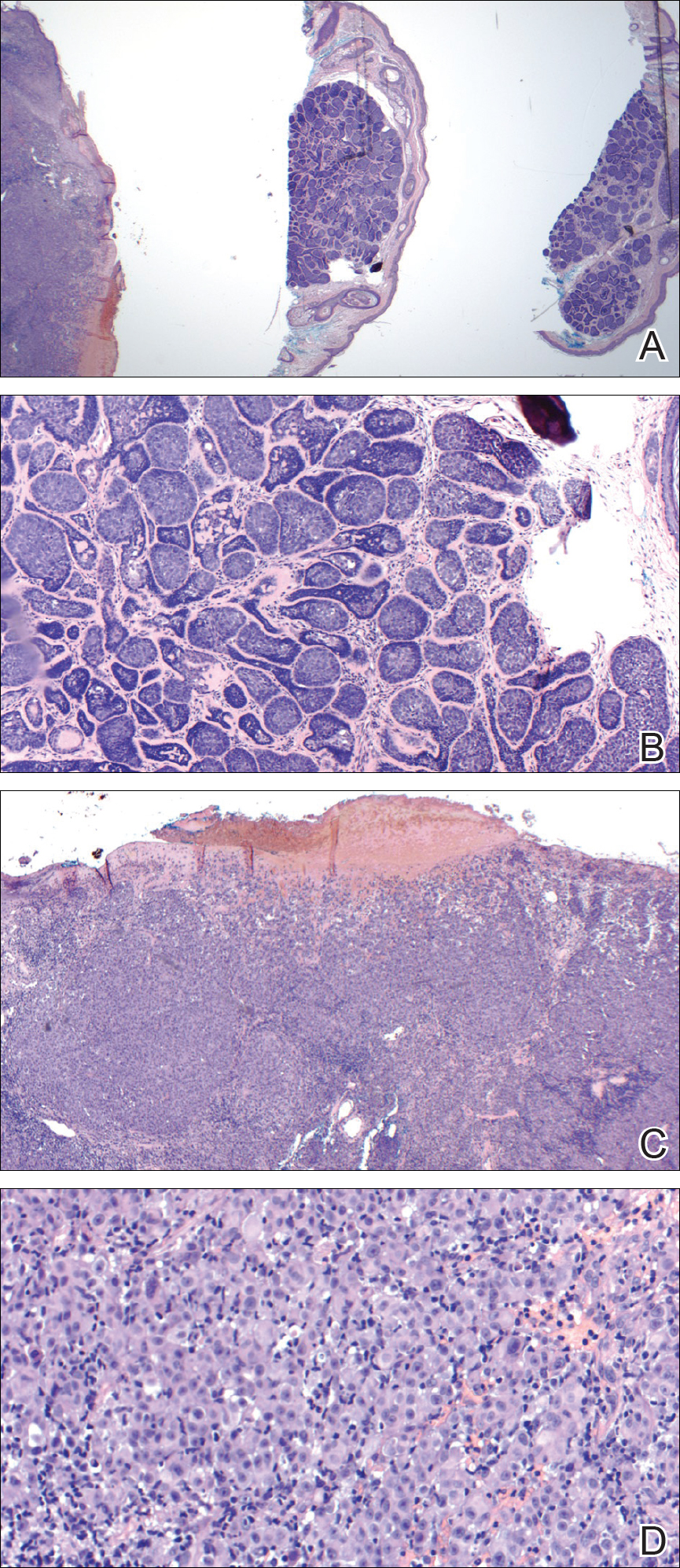
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.
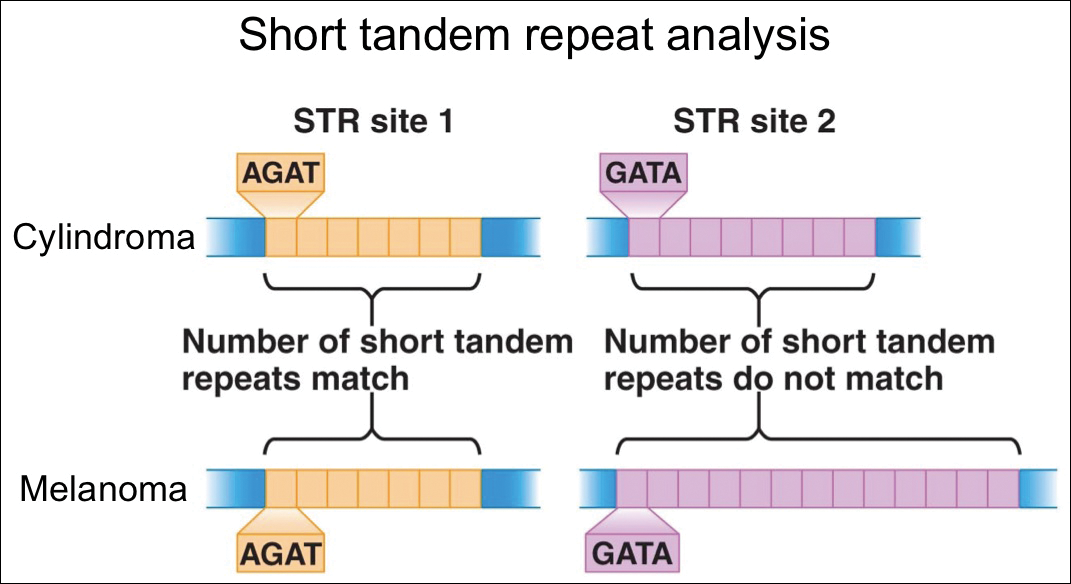
For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.
Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.

The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.

For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.

Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.

The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.

For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.

Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
Resident Pearl
- Although cross-contamination of pathology specimens is rare, it does occur and can impact diagnosis and management if detected early.
Growth on forehead
The FP was concerned about a possible melanoma due to the dark pigmentation and the positive “ABDCE criteria” of melanoma. The FP used his dermatoscope to determine whether this was a melanoma or a pigmented basal cell carcinoma (BCC).
The multiple leaf-like structures and blue-gray ovoid nests seen with dermoscopy suggested that this was a pigmented BCC. (The ulceration could be seen in either melanoma or BCC.) The FP told the patient that this was most certainly a skin cancer and she needed a biopsy that day. The patient consented and anesthesia was obtained with 1% lidocaine and epinephrine. The physician used a DermaBlade to perform a deep shave (saucerization) under the pigmentation. (See the Watch & Learn video on “Shave biopsy.”)
The pathology confirmed pigmented BCC. The physician recommended an elliptical excision and scheduled it for the following week.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP was concerned about a possible melanoma due to the dark pigmentation and the positive “ABDCE criteria” of melanoma. The FP used his dermatoscope to determine whether this was a melanoma or a pigmented basal cell carcinoma (BCC).
The multiple leaf-like structures and blue-gray ovoid nests seen with dermoscopy suggested that this was a pigmented BCC. (The ulceration could be seen in either melanoma or BCC.) The FP told the patient that this was most certainly a skin cancer and she needed a biopsy that day. The patient consented and anesthesia was obtained with 1% lidocaine and epinephrine. The physician used a DermaBlade to perform a deep shave (saucerization) under the pigmentation. (See the Watch & Learn video on “Shave biopsy.”)
The pathology confirmed pigmented BCC. The physician recommended an elliptical excision and scheduled it for the following week.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP was concerned about a possible melanoma due to the dark pigmentation and the positive “ABDCE criteria” of melanoma. The FP used his dermatoscope to determine whether this was a melanoma or a pigmented basal cell carcinoma (BCC).
The multiple leaf-like structures and blue-gray ovoid nests seen with dermoscopy suggested that this was a pigmented BCC. (The ulceration could be seen in either melanoma or BCC.) The FP told the patient that this was most certainly a skin cancer and she needed a biopsy that day. The patient consented and anesthesia was obtained with 1% lidocaine and epinephrine. The physician used a DermaBlade to perform a deep shave (saucerization) under the pigmentation. (See the Watch & Learn video on “Shave biopsy.”)
The pathology confirmed pigmented BCC. The physician recommended an elliptical excision and scheduled it for the following week.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
What Would I Tell My Intern-Year Self?
The training path to dermatology can seem interminable. From getting good grades in college to seeking out the “right” extracurricular activities and cramming for the MCAT, just getting into medical school was a huge challenge. In medical school, you may recognize the same chaos as you begin to prepare for US Medical Licensing Examination Step 1, try to volunteer, and publish original research. Dermatology is undeniably a competitive specialty. The 2018 data released by the National Resident Match Program (also called The Match) showed that only 83% of 412 US seniors who applied were matched to dermatology.1 The average Step 1 score for those who matched was 249 versus 241 for those who did not match. In addition, they had an average of 5.2 research experiences, 9.1 volunteer experiences, and 49.1 were members of Alpha Omega Alpha.1
After studying and working to meet these targets, it is not surprising that the transition to residency is a big change. As a dermatology preliminary intern, or“prelim,” our experience differs compared to other specialties, as other interns are jumping into their area of practice right away.
During my intern year, I had a tremendous amount of anxiety about 2 things: (1) being a subpar medical intern and (2) being unprepared for the beginning of my dermatology residency. This anxiety drove me to read a tremendous amount of medical and dermatological literature in an effort to do everything. Although hindsight is always 20/20, I will share some thoughts of my own as well as some from friends and colleagues.
First, enjoy intern year. I know that may sound ridiculous, but there were many aspects of intern year that I loved! When your pager beeps, it’s for YOU! You are no longer a subintern, running every decision past your intern or explaining your student status to the patients! Proudly introduce yourself as Dr. So-and-So. You earned it! I loved the camaraderie of working with my co-interns and senior residents. Going through the challenges of intern year together is a deep bonding experience, and I absolutely made lifelong friendships. It also does not hurt that I met my boyfriend (now husband), which has changed my life in a big way.
When it comes to learning internal medicine, pediatrics, or surgery (depending on your intern year), prepare for rounds, read about your patients, and pay attention in Grand Rounds. You can even consider taking the dermatologic cases that may be on your team, just for fun. I am always grateful for my internal medicine knowledge when managing complex medical dermatology patients and rounding on our consultation service on the wards. However, do not burden yourself with excessive studying. Enjoy your time off: spend it with family and friends or rediscover a hobby that has been neglected while you have been working toward your achievements.
When it comes to learning dermatology, do not rush it! You have 3 years and a ton of studying ahead of you! You will learn all of it. When July 1 of your first year of dermatology finally starts, immerse yourself in this new world:
- Attend conferences. Even if they are on topics you might not be interested in—from cosmetics to psoriasis—they provide a real-world perspective and often have great lecturers sharing their knowledge.
- Get involved. There are many dermatologic societies to take part in, and dues are waived or reduced when you sign up as a resident. Many of them provide great resources from study materials to journals, and they are always a great way to network when there are events.
- Volunteer. Many of the dermatologic societies sponsor volunteer events such as skin cancer screenings. It can be a fun way to network while also giving back to the community.
- Spend time figuring out what you really enjoy. This step may seem self-evident, but after many years of fulfilling the necessary criteria to get into medical school and residency, it can be habitual to start fulfilling the same criteria all over again. Explore all aspects of dermatology and see what truly interests you. Consider how you expect your life after residency to be and think what learning opportunities might be helpful down the road. Reach out to attendings you would like to work with, both in dermatology and in other specialties. I personally enjoyed working in wound and oncology clinics, learning how other specialties approach clinical dilemmas that we see in dermatology.
As I embark on my final year of dermatology residency, I am truly grateful for the wisdom that has been shared with me on this journey. Many people have provided key pieces of information that have helped shape my training and my plans for the future, and I hope that sharing it will help others!
- National Resident Matching Program, Charting Outcomes in the Match: U.S. Allopathic Seniors, 2018. Washington, DC: National Resident Matching Program; 2018. http://www.nrmp.org/wp-content/uploads/2018/06/Charting-Outcomes-in-the-Match-2018-Seniors.pdf. Accessed September 20, 2018.
The training path to dermatology can seem interminable. From getting good grades in college to seeking out the “right” extracurricular activities and cramming for the MCAT, just getting into medical school was a huge challenge. In medical school, you may recognize the same chaos as you begin to prepare for US Medical Licensing Examination Step 1, try to volunteer, and publish original research. Dermatology is undeniably a competitive specialty. The 2018 data released by the National Resident Match Program (also called The Match) showed that only 83% of 412 US seniors who applied were matched to dermatology.1 The average Step 1 score for those who matched was 249 versus 241 for those who did not match. In addition, they had an average of 5.2 research experiences, 9.1 volunteer experiences, and 49.1 were members of Alpha Omega Alpha.1
After studying and working to meet these targets, it is not surprising that the transition to residency is a big change. As a dermatology preliminary intern, or“prelim,” our experience differs compared to other specialties, as other interns are jumping into their area of practice right away.
During my intern year, I had a tremendous amount of anxiety about 2 things: (1) being a subpar medical intern and (2) being unprepared for the beginning of my dermatology residency. This anxiety drove me to read a tremendous amount of medical and dermatological literature in an effort to do everything. Although hindsight is always 20/20, I will share some thoughts of my own as well as some from friends and colleagues.
First, enjoy intern year. I know that may sound ridiculous, but there were many aspects of intern year that I loved! When your pager beeps, it’s for YOU! You are no longer a subintern, running every decision past your intern or explaining your student status to the patients! Proudly introduce yourself as Dr. So-and-So. You earned it! I loved the camaraderie of working with my co-interns and senior residents. Going through the challenges of intern year together is a deep bonding experience, and I absolutely made lifelong friendships. It also does not hurt that I met my boyfriend (now husband), which has changed my life in a big way.
When it comes to learning internal medicine, pediatrics, or surgery (depending on your intern year), prepare for rounds, read about your patients, and pay attention in Grand Rounds. You can even consider taking the dermatologic cases that may be on your team, just for fun. I am always grateful for my internal medicine knowledge when managing complex medical dermatology patients and rounding on our consultation service on the wards. However, do not burden yourself with excessive studying. Enjoy your time off: spend it with family and friends or rediscover a hobby that has been neglected while you have been working toward your achievements.
When it comes to learning dermatology, do not rush it! You have 3 years and a ton of studying ahead of you! You will learn all of it. When July 1 of your first year of dermatology finally starts, immerse yourself in this new world:
- Attend conferences. Even if they are on topics you might not be interested in—from cosmetics to psoriasis—they provide a real-world perspective and often have great lecturers sharing their knowledge.
- Get involved. There are many dermatologic societies to take part in, and dues are waived or reduced when you sign up as a resident. Many of them provide great resources from study materials to journals, and they are always a great way to network when there are events.
- Volunteer. Many of the dermatologic societies sponsor volunteer events such as skin cancer screenings. It can be a fun way to network while also giving back to the community.
- Spend time figuring out what you really enjoy. This step may seem self-evident, but after many years of fulfilling the necessary criteria to get into medical school and residency, it can be habitual to start fulfilling the same criteria all over again. Explore all aspects of dermatology and see what truly interests you. Consider how you expect your life after residency to be and think what learning opportunities might be helpful down the road. Reach out to attendings you would like to work with, both in dermatology and in other specialties. I personally enjoyed working in wound and oncology clinics, learning how other specialties approach clinical dilemmas that we see in dermatology.
As I embark on my final year of dermatology residency, I am truly grateful for the wisdom that has been shared with me on this journey. Many people have provided key pieces of information that have helped shape my training and my plans for the future, and I hope that sharing it will help others!
The training path to dermatology can seem interminable. From getting good grades in college to seeking out the “right” extracurricular activities and cramming for the MCAT, just getting into medical school was a huge challenge. In medical school, you may recognize the same chaos as you begin to prepare for US Medical Licensing Examination Step 1, try to volunteer, and publish original research. Dermatology is undeniably a competitive specialty. The 2018 data released by the National Resident Match Program (also called The Match) showed that only 83% of 412 US seniors who applied were matched to dermatology.1 The average Step 1 score for those who matched was 249 versus 241 for those who did not match. In addition, they had an average of 5.2 research experiences, 9.1 volunteer experiences, and 49.1 were members of Alpha Omega Alpha.1
After studying and working to meet these targets, it is not surprising that the transition to residency is a big change. As a dermatology preliminary intern, or“prelim,” our experience differs compared to other specialties, as other interns are jumping into their area of practice right away.
During my intern year, I had a tremendous amount of anxiety about 2 things: (1) being a subpar medical intern and (2) being unprepared for the beginning of my dermatology residency. This anxiety drove me to read a tremendous amount of medical and dermatological literature in an effort to do everything. Although hindsight is always 20/20, I will share some thoughts of my own as well as some from friends and colleagues.
First, enjoy intern year. I know that may sound ridiculous, but there were many aspects of intern year that I loved! When your pager beeps, it’s for YOU! You are no longer a subintern, running every decision past your intern or explaining your student status to the patients! Proudly introduce yourself as Dr. So-and-So. You earned it! I loved the camaraderie of working with my co-interns and senior residents. Going through the challenges of intern year together is a deep bonding experience, and I absolutely made lifelong friendships. It also does not hurt that I met my boyfriend (now husband), which has changed my life in a big way.
When it comes to learning internal medicine, pediatrics, or surgery (depending on your intern year), prepare for rounds, read about your patients, and pay attention in Grand Rounds. You can even consider taking the dermatologic cases that may be on your team, just for fun. I am always grateful for my internal medicine knowledge when managing complex medical dermatology patients and rounding on our consultation service on the wards. However, do not burden yourself with excessive studying. Enjoy your time off: spend it with family and friends or rediscover a hobby that has been neglected while you have been working toward your achievements.
When it comes to learning dermatology, do not rush it! You have 3 years and a ton of studying ahead of you! You will learn all of it. When July 1 of your first year of dermatology finally starts, immerse yourself in this new world:
- Attend conferences. Even if they are on topics you might not be interested in—from cosmetics to psoriasis—they provide a real-world perspective and often have great lecturers sharing their knowledge.
- Get involved. There are many dermatologic societies to take part in, and dues are waived or reduced when you sign up as a resident. Many of them provide great resources from study materials to journals, and they are always a great way to network when there are events.
- Volunteer. Many of the dermatologic societies sponsor volunteer events such as skin cancer screenings. It can be a fun way to network while also giving back to the community.
- Spend time figuring out what you really enjoy. This step may seem self-evident, but after many years of fulfilling the necessary criteria to get into medical school and residency, it can be habitual to start fulfilling the same criteria all over again. Explore all aspects of dermatology and see what truly interests you. Consider how you expect your life after residency to be and think what learning opportunities might be helpful down the road. Reach out to attendings you would like to work with, both in dermatology and in other specialties. I personally enjoyed working in wound and oncology clinics, learning how other specialties approach clinical dilemmas that we see in dermatology.
As I embark on my final year of dermatology residency, I am truly grateful for the wisdom that has been shared with me on this journey. Many people have provided key pieces of information that have helped shape my training and my plans for the future, and I hope that sharing it will help others!
- National Resident Matching Program, Charting Outcomes in the Match: U.S. Allopathic Seniors, 2018. Washington, DC: National Resident Matching Program; 2018. http://www.nrmp.org/wp-content/uploads/2018/06/Charting-Outcomes-in-the-Match-2018-Seniors.pdf. Accessed September 20, 2018.
- National Resident Matching Program, Charting Outcomes in the Match: U.S. Allopathic Seniors, 2018. Washington, DC: National Resident Matching Program; 2018. http://www.nrmp.org/wp-content/uploads/2018/06/Charting-Outcomes-in-the-Match-2018-Seniors.pdf. Accessed September 20, 2018.
Real-world data, machine learning, and the reemergence of humanism
As we relentlessly enter information into our EHRs, we typically perceive that we are just recording information about our patients to provide continuity of care and have an accurate representation of what was done. While that is true, the information we record is now increasingly being examined for many additional purposes. A whole new area of study has emerged over the last few years known as “real-world data,” and innovators are beginning to explore how machine learning (currently employed in other areas by such companies as Amazon and Google) may be used to improve the care of patients. The information we are putting into our EHRs is being translated into discrete data and is then combined with data from labs, pharmacies, and claims databases to examine how medications actually work when used in the wide and wild world of practice.
Let’s first talk about why real-world data are important. Traditionally, the evidence we rely upon in medicine has come from randomized trials to give us an unbiased assessment about the safety and the efficacy of the medications that we use. The Achilles’ heel of randomized trials is that, by their nature, they employ a carefully defined group of patients – with specific inclusion and exclusion criteria – who may not be like the patients in our practices. Randomized trials are also conducted in sites that are different than most of our offices. The clinics where randomized trials are conducted have dedicated personnel to follow up on patients, to make sure that patients take their medications, and ensure that patients remember their follow up visits. What this means is that the results in of those studies might not reflect the results seen in the real world.
A nice example of this was reported recently in the area of diabetes management. Randomized trials have shown that the glucagonlike peptide–1 (GLP-1) class of medications have about twice the effectiveness in lowering hemoglobin A1c as do the dipeptidyl peptidase–4 (DPP-4) inhibitor class of medications, but that difference in efficacy is not seen in practice. When looked at in real-world studies, the two classes of medications have about the same glucose-lowering efficacy. Why might that be? In reality, it might be that compliance with GLP-1s is less than that of DPP-4s because of the side effects of nausea and GI intolerance. When patients miss more doses of their GLP-1, they do not achieve the HbA1c lowering seen in trials in which compliance is far better.1
This exploration of real-world outcomes is just a first step in using the information documented in our charts. The exciting next step will be machine learning, also called deep learning.2 In this process, computers look at an enormous number of data points and find relationships that would otherwise not be detected. Imagine a supercomputer analyzing every blood pressure after any medication is changed across thousands, or even millions, of patients, and linking the outcome of that medication choice with the next blood pressure.3 Then imagine the computer meshing millions of data points that include all patients’ weights, ages, sexes, family histories of cardiovascular disease, renal function, etc. and matching those parameters with the specific medication and follow-up blood pressures. While much has been discussed about using genetics to advance personalized medicine, one can imagine these machine-based algorithms discovering connections about which medications work best for individuals with specific characteristics – without the need for additional testing. When the final loop of this cascade is connected, the computer could present recommendations to the clinician about which medication is optimal for the patient and then refine these recommendations, based on outcomes, to optimize safety and efficacy.
Some have argued that there is no way a computer will be able to perform as well as an experienced clinician who utilizes a combination of data and intuition to choose the best medication for his or her patient. This argument is similar to the controversy over autonomous driving cars. Many have asked how you can be assured that the cars will never have an accident. That is, of course, the wrong question. The correct question, as articulated very nicely by one of the innovators in that field, George Holtz, is how we can make a car that is safer than the way that cars are currently being driven (which means fewer deaths than the 15,000 that occur annually with humans behind the wheel).4
Our current method of providing care often leaves patients without appropriate guideline-recommended medications, and many don’t reach their HbA1c, blood pressure, cholesterol, and asthma-control goals. The era of machine learning with machine-generated algorithms may be much closer than we think, which will allow us to spend more time talking with patients, educating them about their disease, and supporting them in their efforts to remain healthy – an attractive future for both us and our patients.
References
1. Carls GS et al. Understanding the gap between efficacy in randomized controlled trials and effectiveness in real-world use of GLP-1RA and DPP-4 therapies in patients with type 2 diabetes. Diabetes Care. 2017 Nov;40(11):1469-78.
2. Naylor CD. On the prospects for a (deep) learning health care system. JAMA. 2018 Sep 18;320(11):1099-100.
3. Wang YR et al. Outpatient hypertension treatment, treatment intensification, and control in Western Europe and the United States. Arch Intern Med. 2007 Jan 22;167(2):141-7.
4. Super Hacker George Hotz: “I can make your car drive itself for under $1,000.”
As we relentlessly enter information into our EHRs, we typically perceive that we are just recording information about our patients to provide continuity of care and have an accurate representation of what was done. While that is true, the information we record is now increasingly being examined for many additional purposes. A whole new area of study has emerged over the last few years known as “real-world data,” and innovators are beginning to explore how machine learning (currently employed in other areas by such companies as Amazon and Google) may be used to improve the care of patients. The information we are putting into our EHRs is being translated into discrete data and is then combined with data from labs, pharmacies, and claims databases to examine how medications actually work when used in the wide and wild world of practice.
Let’s first talk about why real-world data are important. Traditionally, the evidence we rely upon in medicine has come from randomized trials to give us an unbiased assessment about the safety and the efficacy of the medications that we use. The Achilles’ heel of randomized trials is that, by their nature, they employ a carefully defined group of patients – with specific inclusion and exclusion criteria – who may not be like the patients in our practices. Randomized trials are also conducted in sites that are different than most of our offices. The clinics where randomized trials are conducted have dedicated personnel to follow up on patients, to make sure that patients take their medications, and ensure that patients remember their follow up visits. What this means is that the results in of those studies might not reflect the results seen in the real world.
A nice example of this was reported recently in the area of diabetes management. Randomized trials have shown that the glucagonlike peptide–1 (GLP-1) class of medications have about twice the effectiveness in lowering hemoglobin A1c as do the dipeptidyl peptidase–4 (DPP-4) inhibitor class of medications, but that difference in efficacy is not seen in practice. When looked at in real-world studies, the two classes of medications have about the same glucose-lowering efficacy. Why might that be? In reality, it might be that compliance with GLP-1s is less than that of DPP-4s because of the side effects of nausea and GI intolerance. When patients miss more doses of their GLP-1, they do not achieve the HbA1c lowering seen in trials in which compliance is far better.1
This exploration of real-world outcomes is just a first step in using the information documented in our charts. The exciting next step will be machine learning, also called deep learning.2 In this process, computers look at an enormous number of data points and find relationships that would otherwise not be detected. Imagine a supercomputer analyzing every blood pressure after any medication is changed across thousands, or even millions, of patients, and linking the outcome of that medication choice with the next blood pressure.3 Then imagine the computer meshing millions of data points that include all patients’ weights, ages, sexes, family histories of cardiovascular disease, renal function, etc. and matching those parameters with the specific medication and follow-up blood pressures. While much has been discussed about using genetics to advance personalized medicine, one can imagine these machine-based algorithms discovering connections about which medications work best for individuals with specific characteristics – without the need for additional testing. When the final loop of this cascade is connected, the computer could present recommendations to the clinician about which medication is optimal for the patient and then refine these recommendations, based on outcomes, to optimize safety and efficacy.
Some have argued that there is no way a computer will be able to perform as well as an experienced clinician who utilizes a combination of data and intuition to choose the best medication for his or her patient. This argument is similar to the controversy over autonomous driving cars. Many have asked how you can be assured that the cars will never have an accident. That is, of course, the wrong question. The correct question, as articulated very nicely by one of the innovators in that field, George Holtz, is how we can make a car that is safer than the way that cars are currently being driven (which means fewer deaths than the 15,000 that occur annually with humans behind the wheel).4
Our current method of providing care often leaves patients without appropriate guideline-recommended medications, and many don’t reach their HbA1c, blood pressure, cholesterol, and asthma-control goals. The era of machine learning with machine-generated algorithms may be much closer than we think, which will allow us to spend more time talking with patients, educating them about their disease, and supporting them in their efforts to remain healthy – an attractive future for both us and our patients.
References
1. Carls GS et al. Understanding the gap between efficacy in randomized controlled trials and effectiveness in real-world use of GLP-1RA and DPP-4 therapies in patients with type 2 diabetes. Diabetes Care. 2017 Nov;40(11):1469-78.
2. Naylor CD. On the prospects for a (deep) learning health care system. JAMA. 2018 Sep 18;320(11):1099-100.
3. Wang YR et al. Outpatient hypertension treatment, treatment intensification, and control in Western Europe and the United States. Arch Intern Med. 2007 Jan 22;167(2):141-7.
4. Super Hacker George Hotz: “I can make your car drive itself for under $1,000.”
As we relentlessly enter information into our EHRs, we typically perceive that we are just recording information about our patients to provide continuity of care and have an accurate representation of what was done. While that is true, the information we record is now increasingly being examined for many additional purposes. A whole new area of study has emerged over the last few years known as “real-world data,” and innovators are beginning to explore how machine learning (currently employed in other areas by such companies as Amazon and Google) may be used to improve the care of patients. The information we are putting into our EHRs is being translated into discrete data and is then combined with data from labs, pharmacies, and claims databases to examine how medications actually work when used in the wide and wild world of practice.
Let’s first talk about why real-world data are important. Traditionally, the evidence we rely upon in medicine has come from randomized trials to give us an unbiased assessment about the safety and the efficacy of the medications that we use. The Achilles’ heel of randomized trials is that, by their nature, they employ a carefully defined group of patients – with specific inclusion and exclusion criteria – who may not be like the patients in our practices. Randomized trials are also conducted in sites that are different than most of our offices. The clinics where randomized trials are conducted have dedicated personnel to follow up on patients, to make sure that patients take their medications, and ensure that patients remember their follow up visits. What this means is that the results in of those studies might not reflect the results seen in the real world.
A nice example of this was reported recently in the area of diabetes management. Randomized trials have shown that the glucagonlike peptide–1 (GLP-1) class of medications have about twice the effectiveness in lowering hemoglobin A1c as do the dipeptidyl peptidase–4 (DPP-4) inhibitor class of medications, but that difference in efficacy is not seen in practice. When looked at in real-world studies, the two classes of medications have about the same glucose-lowering efficacy. Why might that be? In reality, it might be that compliance with GLP-1s is less than that of DPP-4s because of the side effects of nausea and GI intolerance. When patients miss more doses of their GLP-1, they do not achieve the HbA1c lowering seen in trials in which compliance is far better.1
This exploration of real-world outcomes is just a first step in using the information documented in our charts. The exciting next step will be machine learning, also called deep learning.2 In this process, computers look at an enormous number of data points and find relationships that would otherwise not be detected. Imagine a supercomputer analyzing every blood pressure after any medication is changed across thousands, or even millions, of patients, and linking the outcome of that medication choice with the next blood pressure.3 Then imagine the computer meshing millions of data points that include all patients’ weights, ages, sexes, family histories of cardiovascular disease, renal function, etc. and matching those parameters with the specific medication and follow-up blood pressures. While much has been discussed about using genetics to advance personalized medicine, one can imagine these machine-based algorithms discovering connections about which medications work best for individuals with specific characteristics – without the need for additional testing. When the final loop of this cascade is connected, the computer could present recommendations to the clinician about which medication is optimal for the patient and then refine these recommendations, based on outcomes, to optimize safety and efficacy.
Some have argued that there is no way a computer will be able to perform as well as an experienced clinician who utilizes a combination of data and intuition to choose the best medication for his or her patient. This argument is similar to the controversy over autonomous driving cars. Many have asked how you can be assured that the cars will never have an accident. That is, of course, the wrong question. The correct question, as articulated very nicely by one of the innovators in that field, George Holtz, is how we can make a car that is safer than the way that cars are currently being driven (which means fewer deaths than the 15,000 that occur annually with humans behind the wheel).4
Our current method of providing care often leaves patients without appropriate guideline-recommended medications, and many don’t reach their HbA1c, blood pressure, cholesterol, and asthma-control goals. The era of machine learning with machine-generated algorithms may be much closer than we think, which will allow us to spend more time talking with patients, educating them about their disease, and supporting them in their efforts to remain healthy – an attractive future for both us and our patients.
References
1. Carls GS et al. Understanding the gap between efficacy in randomized controlled trials and effectiveness in real-world use of GLP-1RA and DPP-4 therapies in patients with type 2 diabetes. Diabetes Care. 2017 Nov;40(11):1469-78.
2. Naylor CD. On the prospects for a (deep) learning health care system. JAMA. 2018 Sep 18;320(11):1099-100.
3. Wang YR et al. Outpatient hypertension treatment, treatment intensification, and control in Western Europe and the United States. Arch Intern Med. 2007 Jan 22;167(2):141-7.
4. Super Hacker George Hotz: “I can make your car drive itself for under $1,000.”
Dr. Bawa-Garba and trainee liability
Question: Which of the following regarding medical trainee liability is best?
A. Trainees are commonly named as codefendants with their attending physician in a medical malpractice lawsuit.
B. “From a culture of blame to a culture of safety” is a rallying cry against poor work conditions.
C. House officers are always judged by a lower standard, because they are not fully qualified.
D. A, B, and C are correct.
E. A and C are correct.
Answer: A. A recent case of trainee liability in the United Kingdom resulted in criminal prosecution followed by the trainee being struck off the medical register.1 Dr. Hadiza Bawa-Garba, a pediatric trainee in the U.K. National Health Service, was prosecuted in a court of law and found guilty of manslaughter by gross negligence for the septic death of a 6-year-old boy with Down syndrome. The General Medical Council (GMC), the U.K. medical regulatory agency, voted to take away her license. The decision aroused the ire of physicians worldwide, who noted the poor supervision and undue pressures she was under.
In August 2018, the U.K. Court of Appeal noted that the general clinical competency of Dr. Bawa-Garba was never at issue, and that “the risk of her clinical practice suddenly and without explanation falling below the standards expected on any given day is no higher than for any other reasonably competent doctor.” It reversed the expulsion order and reinstated the 1-year suspension recommended by the Medical Practitioners Tribunal.
Even as the GMC accepted this appellate decision and had convened a commission to look into criminal negligence, it nonetheless received heavy criticism for having overreacted – and for its failure to speak out more forcefully to support those practicing under oppressive conditions.
For example, the Doctors’ Association UK said the GMC had shown it could not be trusted to be objective and nonpunitive. The case, it noted, had “united the medical profession in fear and outrage,” whereby “a pediatrician in training ... a highly regarded doctor, with a previously unblemished record, [was] convicted of [the criminal offence of] gross negligence manslaughter for judgments made whilst doing the jobs of several doctors at once, covering six wards across four floors, responding to numerous pediatric emergencies, without a functioning IT system, and in the absence of a consultant [senior physician], all when just returning from 14 months of maternity leave.”
The Royal College of Pediatrics and Child Health said it had “previously flagged the importance of fostering a culture of supporting doctors to learn from their mistakes, rather than one which seeks to blame.” And the British Medical Association said, “lessons must be learned from this case, which raises wider issues about the multiple factors that affect patient safety in an NHS under extreme pressure, rather than narrowly focusing only on individuals.”2
The fiasco surrounding the Dr. Bawa-Garba case will hopefully result in action similar to that following the seminal report that medical errors account for nearly 100,000 annual hospital deaths in the United States. That study was not restricted to house staff mistakes, but involved multiple hospitals and hospital staff members. It spawned a nationwide reappraisal of how to approach medical errors, and it spurred the Institute of Medicine to recommend that the profession shift “from a culture of blame to a culture of safety.”3
Criminal prosecution in the United States is decidedly rare in death or injury occurring during the course of patient care – for either trainees or attending physicians. A malpractice lawsuit would have been a far more likely outcome had the Dr. Bawa-Garba case taken place in the United States.
Lawsuits against U.S. house staff are not rare, and resident physicians are regularly joined as codefendants with their supervisors, who may be medical school faculty or community practitioners admitting to “team care.” Regulatory actions are typically directed against fully licensed physicians, rather than the trainees. Instead, the director of the training program itself would take corrective action against an errant resident, if warranted, which can range from a warning to outright dismissal from the program.
How is negligence law applied to a trainee? Should it demand the same standard of care as it would a fully qualified attending physician?4 Surprisingly, the courts are split on this question. Some have favored using a dual standard of conduct, with trainees being held to a lower standard.
This was articulated in Rush v. Akron General Hospital, which involved a patient who had fallen through a glass door. The patient suffered several lacerations to his shoulder, which the intern treated. However, when two remaining pieces of glass were later discovered in the area of injury, the patient sued the intern for negligence.
The court dismissed the claim, finding that the intern had practiced with the skill and care of his peers of similar training. “It would be unreasonable to exact from an intern, doing emergency work in a hospital, that high degree of skill which is impliedly possessed by a physician and surgeon in the general practice of his profession, with an extensive and constant practice in hospitals and the community,” the court noted.5
However, not all courts have embraced this dual standard of review. The New Jersey Superior Court held that licensed residents should be judged by a standard applicable to a general practitioner, because any reduction in the standard of care would set a problematic precedent.6 In that case, the residents allegedly failed to reinsert a nasogastric tube, which caused the patient to aspirate.
And in Pratt v. Stein, a second-year resident was judged by an even higher standard – that of a specialist – after he had allegedly administered a toxic dose of neomycin to a postoperative patient, which resulted in deafness. Although the lower court had ruled that the resident should be held to the standard of an ordinary physician, the Pennsylvania appellate court disagreed, reasoning that “a resident should be held to the standard of a specialist when the resident is acting within his field of specialty. In our estimation, this is a sound conclusion. A resident is already a physician who has chosen to specialize, and thus possesses a higher degree of knowledge and skill in the chosen specialty than does the nonspecialist.”7
However, a subsequent decision from the same jurisdiction suggests a retreat from this unrealistic standard.
An orthopedic resident allegedly applied a cast with insufficient padding to the broken wrist of a patient. The plaintiff claimed this led to soft-tissue infection with Staphylococcus aureus, with complicating septicemia, staphylococcal endocarditis, and eventual death.
The court held that the resident’s standard of care should be “higher than that for general practitioners but less than that for fully trained orthopedic specialists. ... To require a resident to meet the same standard of care as fully trained specialists would be unrealistic. A resident may have had only days or weeks of training in the specialized residency program; a specialist, on the other hand, will have completed the residency program and may also have had years of experience in the specialized field. If we were to require the resident to exercise the same degree of skill and training as the specialist, we would, in effect, be requiring the resident to do the impossible.”8
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Saurabh Jha, “To Err Is Homicide in Britain: The Case of Hadiza Bawa-Garba.” The Health Care Blog, Jan. 30, 2018.
2. “‘Lessons Must Be Learned’: UK Societies on Bawa-Garba Ruling.” Medscape, Aug. 14, 2018.
3. “To Err is Human: Building a Safer Health System.” Institute of Medicine, National Academies Press, Washington D.C., 1999.
4. JAMA. 2004 Sep 1;292(9):1051-6.
5. Rush v. Akron General Hospital, 171 N.E.2d 378 (Ohio Ct. App. 1987).
6. Clark v. University Hospital, 914 A.2d 838 (N.J. Super. 2006).
7. Pratt v. Stein, 444 A.2d 674 (Pa. Super. 1980).
8. Jistarri v. Nappi, 549 A.2d 210 (Pa. Super. 1988).
Question: Which of the following regarding medical trainee liability is best?
A. Trainees are commonly named as codefendants with their attending physician in a medical malpractice lawsuit.
B. “From a culture of blame to a culture of safety” is a rallying cry against poor work conditions.
C. House officers are always judged by a lower standard, because they are not fully qualified.
D. A, B, and C are correct.
E. A and C are correct.
Answer: A. A recent case of trainee liability in the United Kingdom resulted in criminal prosecution followed by the trainee being struck off the medical register.1 Dr. Hadiza Bawa-Garba, a pediatric trainee in the U.K. National Health Service, was prosecuted in a court of law and found guilty of manslaughter by gross negligence for the septic death of a 6-year-old boy with Down syndrome. The General Medical Council (GMC), the U.K. medical regulatory agency, voted to take away her license. The decision aroused the ire of physicians worldwide, who noted the poor supervision and undue pressures she was under.
In August 2018, the U.K. Court of Appeal noted that the general clinical competency of Dr. Bawa-Garba was never at issue, and that “the risk of her clinical practice suddenly and without explanation falling below the standards expected on any given day is no higher than for any other reasonably competent doctor.” It reversed the expulsion order and reinstated the 1-year suspension recommended by the Medical Practitioners Tribunal.
Even as the GMC accepted this appellate decision and had convened a commission to look into criminal negligence, it nonetheless received heavy criticism for having overreacted – and for its failure to speak out more forcefully to support those practicing under oppressive conditions.
For example, the Doctors’ Association UK said the GMC had shown it could not be trusted to be objective and nonpunitive. The case, it noted, had “united the medical profession in fear and outrage,” whereby “a pediatrician in training ... a highly regarded doctor, with a previously unblemished record, [was] convicted of [the criminal offence of] gross negligence manslaughter for judgments made whilst doing the jobs of several doctors at once, covering six wards across four floors, responding to numerous pediatric emergencies, without a functioning IT system, and in the absence of a consultant [senior physician], all when just returning from 14 months of maternity leave.”
The Royal College of Pediatrics and Child Health said it had “previously flagged the importance of fostering a culture of supporting doctors to learn from their mistakes, rather than one which seeks to blame.” And the British Medical Association said, “lessons must be learned from this case, which raises wider issues about the multiple factors that affect patient safety in an NHS under extreme pressure, rather than narrowly focusing only on individuals.”2
The fiasco surrounding the Dr. Bawa-Garba case will hopefully result in action similar to that following the seminal report that medical errors account for nearly 100,000 annual hospital deaths in the United States. That study was not restricted to house staff mistakes, but involved multiple hospitals and hospital staff members. It spawned a nationwide reappraisal of how to approach medical errors, and it spurred the Institute of Medicine to recommend that the profession shift “from a culture of blame to a culture of safety.”3
Criminal prosecution in the United States is decidedly rare in death or injury occurring during the course of patient care – for either trainees or attending physicians. A malpractice lawsuit would have been a far more likely outcome had the Dr. Bawa-Garba case taken place in the United States.
Lawsuits against U.S. house staff are not rare, and resident physicians are regularly joined as codefendants with their supervisors, who may be medical school faculty or community practitioners admitting to “team care.” Regulatory actions are typically directed against fully licensed physicians, rather than the trainees. Instead, the director of the training program itself would take corrective action against an errant resident, if warranted, which can range from a warning to outright dismissal from the program.
How is negligence law applied to a trainee? Should it demand the same standard of care as it would a fully qualified attending physician?4 Surprisingly, the courts are split on this question. Some have favored using a dual standard of conduct, with trainees being held to a lower standard.
This was articulated in Rush v. Akron General Hospital, which involved a patient who had fallen through a glass door. The patient suffered several lacerations to his shoulder, which the intern treated. However, when two remaining pieces of glass were later discovered in the area of injury, the patient sued the intern for negligence.
The court dismissed the claim, finding that the intern had practiced with the skill and care of his peers of similar training. “It would be unreasonable to exact from an intern, doing emergency work in a hospital, that high degree of skill which is impliedly possessed by a physician and surgeon in the general practice of his profession, with an extensive and constant practice in hospitals and the community,” the court noted.5
However, not all courts have embraced this dual standard of review. The New Jersey Superior Court held that licensed residents should be judged by a standard applicable to a general practitioner, because any reduction in the standard of care would set a problematic precedent.6 In that case, the residents allegedly failed to reinsert a nasogastric tube, which caused the patient to aspirate.
And in Pratt v. Stein, a second-year resident was judged by an even higher standard – that of a specialist – after he had allegedly administered a toxic dose of neomycin to a postoperative patient, which resulted in deafness. Although the lower court had ruled that the resident should be held to the standard of an ordinary physician, the Pennsylvania appellate court disagreed, reasoning that “a resident should be held to the standard of a specialist when the resident is acting within his field of specialty. In our estimation, this is a sound conclusion. A resident is already a physician who has chosen to specialize, and thus possesses a higher degree of knowledge and skill in the chosen specialty than does the nonspecialist.”7
However, a subsequent decision from the same jurisdiction suggests a retreat from this unrealistic standard.
An orthopedic resident allegedly applied a cast with insufficient padding to the broken wrist of a patient. The plaintiff claimed this led to soft-tissue infection with Staphylococcus aureus, with complicating septicemia, staphylococcal endocarditis, and eventual death.
The court held that the resident’s standard of care should be “higher than that for general practitioners but less than that for fully trained orthopedic specialists. ... To require a resident to meet the same standard of care as fully trained specialists would be unrealistic. A resident may have had only days or weeks of training in the specialized residency program; a specialist, on the other hand, will have completed the residency program and may also have had years of experience in the specialized field. If we were to require the resident to exercise the same degree of skill and training as the specialist, we would, in effect, be requiring the resident to do the impossible.”8
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Saurabh Jha, “To Err Is Homicide in Britain: The Case of Hadiza Bawa-Garba.” The Health Care Blog, Jan. 30, 2018.
2. “‘Lessons Must Be Learned’: UK Societies on Bawa-Garba Ruling.” Medscape, Aug. 14, 2018.
3. “To Err is Human: Building a Safer Health System.” Institute of Medicine, National Academies Press, Washington D.C., 1999.
4. JAMA. 2004 Sep 1;292(9):1051-6.
5. Rush v. Akron General Hospital, 171 N.E.2d 378 (Ohio Ct. App. 1987).
6. Clark v. University Hospital, 914 A.2d 838 (N.J. Super. 2006).
7. Pratt v. Stein, 444 A.2d 674 (Pa. Super. 1980).
8. Jistarri v. Nappi, 549 A.2d 210 (Pa. Super. 1988).
Question: Which of the following regarding medical trainee liability is best?
A. Trainees are commonly named as codefendants with their attending physician in a medical malpractice lawsuit.
B. “From a culture of blame to a culture of safety” is a rallying cry against poor work conditions.
C. House officers are always judged by a lower standard, because they are not fully qualified.
D. A, B, and C are correct.
E. A and C are correct.
Answer: A. A recent case of trainee liability in the United Kingdom resulted in criminal prosecution followed by the trainee being struck off the medical register.1 Dr. Hadiza Bawa-Garba, a pediatric trainee in the U.K. National Health Service, was prosecuted in a court of law and found guilty of manslaughter by gross negligence for the septic death of a 6-year-old boy with Down syndrome. The General Medical Council (GMC), the U.K. medical regulatory agency, voted to take away her license. The decision aroused the ire of physicians worldwide, who noted the poor supervision and undue pressures she was under.
In August 2018, the U.K. Court of Appeal noted that the general clinical competency of Dr. Bawa-Garba was never at issue, and that “the risk of her clinical practice suddenly and without explanation falling below the standards expected on any given day is no higher than for any other reasonably competent doctor.” It reversed the expulsion order and reinstated the 1-year suspension recommended by the Medical Practitioners Tribunal.
Even as the GMC accepted this appellate decision and had convened a commission to look into criminal negligence, it nonetheless received heavy criticism for having overreacted – and for its failure to speak out more forcefully to support those practicing under oppressive conditions.
For example, the Doctors’ Association UK said the GMC had shown it could not be trusted to be objective and nonpunitive. The case, it noted, had “united the medical profession in fear and outrage,” whereby “a pediatrician in training ... a highly regarded doctor, with a previously unblemished record, [was] convicted of [the criminal offence of] gross negligence manslaughter for judgments made whilst doing the jobs of several doctors at once, covering six wards across four floors, responding to numerous pediatric emergencies, without a functioning IT system, and in the absence of a consultant [senior physician], all when just returning from 14 months of maternity leave.”
The Royal College of Pediatrics and Child Health said it had “previously flagged the importance of fostering a culture of supporting doctors to learn from their mistakes, rather than one which seeks to blame.” And the British Medical Association said, “lessons must be learned from this case, which raises wider issues about the multiple factors that affect patient safety in an NHS under extreme pressure, rather than narrowly focusing only on individuals.”2
The fiasco surrounding the Dr. Bawa-Garba case will hopefully result in action similar to that following the seminal report that medical errors account for nearly 100,000 annual hospital deaths in the United States. That study was not restricted to house staff mistakes, but involved multiple hospitals and hospital staff members. It spawned a nationwide reappraisal of how to approach medical errors, and it spurred the Institute of Medicine to recommend that the profession shift “from a culture of blame to a culture of safety.”3
Criminal prosecution in the United States is decidedly rare in death or injury occurring during the course of patient care – for either trainees or attending physicians. A malpractice lawsuit would have been a far more likely outcome had the Dr. Bawa-Garba case taken place in the United States.
Lawsuits against U.S. house staff are not rare, and resident physicians are regularly joined as codefendants with their supervisors, who may be medical school faculty or community practitioners admitting to “team care.” Regulatory actions are typically directed against fully licensed physicians, rather than the trainees. Instead, the director of the training program itself would take corrective action against an errant resident, if warranted, which can range from a warning to outright dismissal from the program.
How is negligence law applied to a trainee? Should it demand the same standard of care as it would a fully qualified attending physician?4 Surprisingly, the courts are split on this question. Some have favored using a dual standard of conduct, with trainees being held to a lower standard.
This was articulated in Rush v. Akron General Hospital, which involved a patient who had fallen through a glass door. The patient suffered several lacerations to his shoulder, which the intern treated. However, when two remaining pieces of glass were later discovered in the area of injury, the patient sued the intern for negligence.
The court dismissed the claim, finding that the intern had practiced with the skill and care of his peers of similar training. “It would be unreasonable to exact from an intern, doing emergency work in a hospital, that high degree of skill which is impliedly possessed by a physician and surgeon in the general practice of his profession, with an extensive and constant practice in hospitals and the community,” the court noted.5
However, not all courts have embraced this dual standard of review. The New Jersey Superior Court held that licensed residents should be judged by a standard applicable to a general practitioner, because any reduction in the standard of care would set a problematic precedent.6 In that case, the residents allegedly failed to reinsert a nasogastric tube, which caused the patient to aspirate.
And in Pratt v. Stein, a second-year resident was judged by an even higher standard – that of a specialist – after he had allegedly administered a toxic dose of neomycin to a postoperative patient, which resulted in deafness. Although the lower court had ruled that the resident should be held to the standard of an ordinary physician, the Pennsylvania appellate court disagreed, reasoning that “a resident should be held to the standard of a specialist when the resident is acting within his field of specialty. In our estimation, this is a sound conclusion. A resident is already a physician who has chosen to specialize, and thus possesses a higher degree of knowledge and skill in the chosen specialty than does the nonspecialist.”7
However, a subsequent decision from the same jurisdiction suggests a retreat from this unrealistic standard.
An orthopedic resident allegedly applied a cast with insufficient padding to the broken wrist of a patient. The plaintiff claimed this led to soft-tissue infection with Staphylococcus aureus, with complicating septicemia, staphylococcal endocarditis, and eventual death.
The court held that the resident’s standard of care should be “higher than that for general practitioners but less than that for fully trained orthopedic specialists. ... To require a resident to meet the same standard of care as fully trained specialists would be unrealistic. A resident may have had only days or weeks of training in the specialized residency program; a specialist, on the other hand, will have completed the residency program and may also have had years of experience in the specialized field. If we were to require the resident to exercise the same degree of skill and training as the specialist, we would, in effect, be requiring the resident to do the impossible.”8
Dr. Tan is emeritus professor of medicine and former adjunct professor of law at the University of Hawaii, Honolulu. This article is meant to be educational and does not constitute medical, ethical, or legal advice. For additional information, readers may contact the author at [email protected].
References
1. Saurabh Jha, “To Err Is Homicide in Britain: The Case of Hadiza Bawa-Garba.” The Health Care Blog, Jan. 30, 2018.
2. “‘Lessons Must Be Learned’: UK Societies on Bawa-Garba Ruling.” Medscape, Aug. 14, 2018.
3. “To Err is Human: Building a Safer Health System.” Institute of Medicine, National Academies Press, Washington D.C., 1999.
4. JAMA. 2004 Sep 1;292(9):1051-6.
5. Rush v. Akron General Hospital, 171 N.E.2d 378 (Ohio Ct. App. 1987).
6. Clark v. University Hospital, 914 A.2d 838 (N.J. Super. 2006).
7. Pratt v. Stein, 444 A.2d 674 (Pa. Super. 1980).
8. Jistarri v. Nappi, 549 A.2d 210 (Pa. Super. 1988).
Rash on arm
The FP looked closely at the so-called rash and realized that while it could be nummular eczema it could also be a superficial basal cell carcinoma (BCC).
He explained the differential diagnosis to the patient and suggested that he perform a shave biopsy that day. The patient consented to the biopsy, and the physician numbed the area with 1% lidocaine and epinephrine. He used a DermaBlade and obtained hemostasis with aluminum chloride in water. (See the Watch & Learn video on “Shave biopsy.”) The biopsy result confirmed the FP’s suspicion: The lesion was a superficial BCC.
On the follow-up visit the FP explained the options for treatment, including electrodesiccation and curettage, cryosurgery, or an elliptical excision. He told the patient that the cure rates are about the same, regardless of which of these treatments were chosen. He also explained that either of the 2 destructive methods could be performed immediately, whereas the elliptical excision would require scheduling a longer appointment.
The patient chose the cryosurgery. (See the Watch & Learn video on cryosurgery.) After numbing the area with 1% lidocaine and epinephrine, the physician froze the lesion with a 3 mm halo for 30 seconds using liquid nitrogen spray. At follow-up 3 months later, there was some hypopigmentation, but no evidence of the BCC.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP looked closely at the so-called rash and realized that while it could be nummular eczema it could also be a superficial basal cell carcinoma (BCC).
He explained the differential diagnosis to the patient and suggested that he perform a shave biopsy that day. The patient consented to the biopsy, and the physician numbed the area with 1% lidocaine and epinephrine. He used a DermaBlade and obtained hemostasis with aluminum chloride in water. (See the Watch & Learn video on “Shave biopsy.”) The biopsy result confirmed the FP’s suspicion: The lesion was a superficial BCC.
On the follow-up visit the FP explained the options for treatment, including electrodesiccation and curettage, cryosurgery, or an elliptical excision. He told the patient that the cure rates are about the same, regardless of which of these treatments were chosen. He also explained that either of the 2 destructive methods could be performed immediately, whereas the elliptical excision would require scheduling a longer appointment.
The patient chose the cryosurgery. (See the Watch & Learn video on cryosurgery.) After numbing the area with 1% lidocaine and epinephrine, the physician froze the lesion with a 3 mm halo for 30 seconds using liquid nitrogen spray. At follow-up 3 months later, there was some hypopigmentation, but no evidence of the BCC.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP looked closely at the so-called rash and realized that while it could be nummular eczema it could also be a superficial basal cell carcinoma (BCC).
He explained the differential diagnosis to the patient and suggested that he perform a shave biopsy that day. The patient consented to the biopsy, and the physician numbed the area with 1% lidocaine and epinephrine. He used a DermaBlade and obtained hemostasis with aluminum chloride in water. (See the Watch & Learn video on “Shave biopsy.”) The biopsy result confirmed the FP’s suspicion: The lesion was a superficial BCC.
On the follow-up visit the FP explained the options for treatment, including electrodesiccation and curettage, cryosurgery, or an elliptical excision. He told the patient that the cure rates are about the same, regardless of which of these treatments were chosen. He also explained that either of the 2 destructive methods could be performed immediately, whereas the elliptical excision would require scheduling a longer appointment.
The patient chose the cryosurgery. (See the Watch & Learn video on cryosurgery.) After numbing the area with 1% lidocaine and epinephrine, the physician froze the lesion with a 3 mm halo for 30 seconds using liquid nitrogen spray. At follow-up 3 months later, there was some hypopigmentation, but no evidence of the BCC.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Review finds some therapies show promise in managing prurigo nodularis
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Recent success in the use of (PN) suggests that safer and more effective systemic therapies that provide relief and management of this debilitating chronic skin condition are possible, reported Azam A. Qureshi, BA, of the department of dermatology, George Washington University, Washington, and his coauthors.
Their report was published in the Journal of the American Academy of Dermatology.
They conducted a systematic review of 35 clinical studies of different therapies for PN, published between Jan. 1, 1990, and March 22, 2018, using PubMed and Scopus databases. Their goal was twofold: to provide a summary of current evidence-based therapies and their corresponding level of evidence ratings, and to help researchers “identify gaps in PN treatment development and study.”
From 706 studies, the authors selected 35 clinical studies of treatment strategies for PN, for which the pathogenesis is virtually unknown, they noted. The studies included 15 prospective cohort studies, 11 retrospective reviews, 8 randomized controlled trials (with 10-127 patients), and 1 case series. Studies that failed to report treatment outcomes and those with fewer than five patients were excluded from the review.
Treatment modalities included topical agents, phototherapy and photochemotherapy, thalidomide, systemic immunomodulatory drugs, antiepileptics and antidepressants, and emerging treatment approaches. Many of the treatments evaluated in the review were found to offer limited promise for clinical application as a result of low efficacy or frequent side effects. The authors attributed the overall lack of success with treatments to the “heterogeneous nature of the etiology of chronic pruritus.”
Thalidomide was found to have limited use given its poor safety profile; lenalidomide achieved better outcomes, but treatment in one case was stopped because of possible drug-induced neuropathy or myopathy, they wrote. The systemic immunomodulatory agents methotrexate and cyclosporine exhibited successful treatment but also were found to have poor safety profiles. Greater promise was seen with antiepileptics and antidepressants, which were associated with fewer side effects. “Antidepressants, such as mirtazapine and amitriptyline, and antiepileptics, such as gabapentin and pregabalin, can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” senior author Adam Friedman, MD, professor of dermatology at George Washington University, said in an interview.
Among the most positive outcomes achieved were those in which promising newer treatments, including targeting IL-31 signaling and opioid receptor modulation, were used. In particular, the authors cited nemolizumab, an IL-31 receptor A antagonist, which is currently in a phase 2 trial of PN. Beneficial effects of extended release nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, were found in an unpublished multicenter, double-blind randomized controlled trial, with no serious adverse effects attributed to treatment.
Among the emerging treatments are the neurokinin-1 receptor antagonists, which include serlopitant, which showed beneficial effects compared with placebo in an 8-week randomized controlled study, they wrote. The neurokinin-1 receptor “is a target of substance P, a mediator of itch and a probable pathogenic agent in PN,” they wrote. “Binding by these agents likely disrupts substance P signaling, thereby halting PN pathogenesis,” they added.
“Antidepressants such as mirtazapine and amitriptyline and antiepileptics such as gabapentin and pregabalin can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” Dr. Friedman said in the interview.
Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
SOURCE: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Recent success in the use of (PN) suggests that safer and more effective systemic therapies that provide relief and management of this debilitating chronic skin condition are possible, reported Azam A. Qureshi, BA, of the department of dermatology, George Washington University, Washington, and his coauthors.
Their report was published in the Journal of the American Academy of Dermatology.
They conducted a systematic review of 35 clinical studies of different therapies for PN, published between Jan. 1, 1990, and March 22, 2018, using PubMed and Scopus databases. Their goal was twofold: to provide a summary of current evidence-based therapies and their corresponding level of evidence ratings, and to help researchers “identify gaps in PN treatment development and study.”
From 706 studies, the authors selected 35 clinical studies of treatment strategies for PN, for which the pathogenesis is virtually unknown, they noted. The studies included 15 prospective cohort studies, 11 retrospective reviews, 8 randomized controlled trials (with 10-127 patients), and 1 case series. Studies that failed to report treatment outcomes and those with fewer than five patients were excluded from the review.
Treatment modalities included topical agents, phototherapy and photochemotherapy, thalidomide, systemic immunomodulatory drugs, antiepileptics and antidepressants, and emerging treatment approaches. Many of the treatments evaluated in the review were found to offer limited promise for clinical application as a result of low efficacy or frequent side effects. The authors attributed the overall lack of success with treatments to the “heterogeneous nature of the etiology of chronic pruritus.”
Thalidomide was found to have limited use given its poor safety profile; lenalidomide achieved better outcomes, but treatment in one case was stopped because of possible drug-induced neuropathy or myopathy, they wrote. The systemic immunomodulatory agents methotrexate and cyclosporine exhibited successful treatment but also were found to have poor safety profiles. Greater promise was seen with antiepileptics and antidepressants, which were associated with fewer side effects. “Antidepressants, such as mirtazapine and amitriptyline, and antiepileptics, such as gabapentin and pregabalin, can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” senior author Adam Friedman, MD, professor of dermatology at George Washington University, said in an interview.
Among the most positive outcomes achieved were those in which promising newer treatments, including targeting IL-31 signaling and opioid receptor modulation, were used. In particular, the authors cited nemolizumab, an IL-31 receptor A antagonist, which is currently in a phase 2 trial of PN. Beneficial effects of extended release nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, were found in an unpublished multicenter, double-blind randomized controlled trial, with no serious adverse effects attributed to treatment.
Among the emerging treatments are the neurokinin-1 receptor antagonists, which include serlopitant, which showed beneficial effects compared with placebo in an 8-week randomized controlled study, they wrote. The neurokinin-1 receptor “is a target of substance P, a mediator of itch and a probable pathogenic agent in PN,” they wrote. “Binding by these agents likely disrupts substance P signaling, thereby halting PN pathogenesis,” they added.
“Antidepressants such as mirtazapine and amitriptyline and antiepileptics such as gabapentin and pregabalin can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” Dr. Friedman said in the interview.
Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
SOURCE: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Recent success in the use of (PN) suggests that safer and more effective systemic therapies that provide relief and management of this debilitating chronic skin condition are possible, reported Azam A. Qureshi, BA, of the department of dermatology, George Washington University, Washington, and his coauthors.
Their report was published in the Journal of the American Academy of Dermatology.
They conducted a systematic review of 35 clinical studies of different therapies for PN, published between Jan. 1, 1990, and March 22, 2018, using PubMed and Scopus databases. Their goal was twofold: to provide a summary of current evidence-based therapies and their corresponding level of evidence ratings, and to help researchers “identify gaps in PN treatment development and study.”
From 706 studies, the authors selected 35 clinical studies of treatment strategies for PN, for which the pathogenesis is virtually unknown, they noted. The studies included 15 prospective cohort studies, 11 retrospective reviews, 8 randomized controlled trials (with 10-127 patients), and 1 case series. Studies that failed to report treatment outcomes and those with fewer than five patients were excluded from the review.
Treatment modalities included topical agents, phototherapy and photochemotherapy, thalidomide, systemic immunomodulatory drugs, antiepileptics and antidepressants, and emerging treatment approaches. Many of the treatments evaluated in the review were found to offer limited promise for clinical application as a result of low efficacy or frequent side effects. The authors attributed the overall lack of success with treatments to the “heterogeneous nature of the etiology of chronic pruritus.”
Thalidomide was found to have limited use given its poor safety profile; lenalidomide achieved better outcomes, but treatment in one case was stopped because of possible drug-induced neuropathy or myopathy, they wrote. The systemic immunomodulatory agents methotrexate and cyclosporine exhibited successful treatment but also were found to have poor safety profiles. Greater promise was seen with antiepileptics and antidepressants, which were associated with fewer side effects. “Antidepressants, such as mirtazapine and amitriptyline, and antiepileptics, such as gabapentin and pregabalin, can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” senior author Adam Friedman, MD, professor of dermatology at George Washington University, said in an interview.
Among the most positive outcomes achieved were those in which promising newer treatments, including targeting IL-31 signaling and opioid receptor modulation, were used. In particular, the authors cited nemolizumab, an IL-31 receptor A antagonist, which is currently in a phase 2 trial of PN. Beneficial effects of extended release nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, were found in an unpublished multicenter, double-blind randomized controlled trial, with no serious adverse effects attributed to treatment.
Among the emerging treatments are the neurokinin-1 receptor antagonists, which include serlopitant, which showed beneficial effects compared with placebo in an 8-week randomized controlled study, they wrote. The neurokinin-1 receptor “is a target of substance P, a mediator of itch and a probable pathogenic agent in PN,” they wrote. “Binding by these agents likely disrupts substance P signaling, thereby halting PN pathogenesis,” they added.
“Antidepressants such as mirtazapine and amitriptyline and antiepileptics such as gabapentin and pregabalin can offer significant relief to a good number of patients, though success is variable and as we found in our study, the evidence available is limited,” Dr. Friedman said in the interview.
Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
SOURCE: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
Key clinical point: Additional research is needed to develop more individualized treatment strategies, but data for some treatments are promising.
Major finding: Treatments identified as having beneficial effects in the review included nemolizumab, an IL-31 receptor A antagonist; nalbuphine, an opioid k-receptor agonist and mu-receptor antagonist, and serlopitant, a neurokinin-1 receptor antagonist.
Study details: A systematic review of 35 studies evaluating treatments for PN, published between Jan. 1, 1990, and March 22, 2018.
Disclosures: Mr. Qureshi had no relevant financial disclosures. Dr. Friedman has received honoraria for participation in advisory boards for Menlo Therapeutics, and serves as an investigator for Kiniksa Pharmaceuticals. A third author received honoraria for participation in advisory boards for Menlo, and several other companies, and received grant/research support as an investigator from Menlo, Kiniksa, and several other companies.
Source: Qureshi AA et al. J Am Acad Dermatol. 2018. doi: 10.1016/j.jaad.2018.09.020.
Growth on nose
The FP made the presumptive diagnosis of a nodular basal cell carcinoma.
He explained the importance of performing a biopsy and obtained informed consent. On the same day of the patient’s visit, he injected 1% lidocaine with epinephrine under the lesion with a single stick of a 30 gauge needle. He knew that it was safe to use epinephrine on the nose, and that it would prevent excessive bleeding during the biopsy. Contrary to the myth frequently taught in medical school, the nose is a very vascular area. The physician performed a shave biopsy that removed the top of the lesion flush with the skin around it. (See the Watch & Learn video on “Shave biopsy.”)
The pathology report came back as a nodular basal cell carcinoma. On the following visit, the physician recommended Mohs surgery as a way to preserve the vital anatomy of the nasal ala and achieve the highest cure rate.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP made the presumptive diagnosis of a nodular basal cell carcinoma.
He explained the importance of performing a biopsy and obtained informed consent. On the same day of the patient’s visit, he injected 1% lidocaine with epinephrine under the lesion with a single stick of a 30 gauge needle. He knew that it was safe to use epinephrine on the nose, and that it would prevent excessive bleeding during the biopsy. Contrary to the myth frequently taught in medical school, the nose is a very vascular area. The physician performed a shave biopsy that removed the top of the lesion flush with the skin around it. (See the Watch & Learn video on “Shave biopsy.”)
The pathology report came back as a nodular basal cell carcinoma. On the following visit, the physician recommended Mohs surgery as a way to preserve the vital anatomy of the nasal ala and achieve the highest cure rate.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP made the presumptive diagnosis of a nodular basal cell carcinoma.
He explained the importance of performing a biopsy and obtained informed consent. On the same day of the patient’s visit, he injected 1% lidocaine with epinephrine under the lesion with a single stick of a 30 gauge needle. He knew that it was safe to use epinephrine on the nose, and that it would prevent excessive bleeding during the biopsy. Contrary to the myth frequently taught in medical school, the nose is a very vascular area. The physician performed a shave biopsy that removed the top of the lesion flush with the skin around it. (See the Watch & Learn video on “Shave biopsy.”)
The pathology report came back as a nodular basal cell carcinoma. On the following visit, the physician recommended Mohs surgery as a way to preserve the vital anatomy of the nasal ala and achieve the highest cure rate.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.