User login
Oral propranolol shown safe in PHACE
LAHAINA, HAWAII – Reassuring evidence of the safety of oral propranolol for treatment of complicated infantile hemangiomas in patients with PHACE syndrome comes from a recent multicenter study.
Oral propranolol is now well-ensconced as first-line therapy for complicated infantile hemangiomas in otherwise healthy children. However, the beta-blocker’s use in PHACE (Posterior fossa malformations, Hemangiomas, Arterial anomalies, Cardiac defects, and Eye abnormalities) syndrome has been controversial, with concerns raised by some that it might raise the risk for arterial ischemic stroke. Not so, Moise L. Levy, MD, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I’m not suggesting you use propranolol with reckless abandon in this population, but this stroke concern is something that should be put to bed based on this study,” advised Dr. Levy, professor of dermatology and pediatrics at Dell Medical School in Austin, Tex., and physician-in-chief at Dell Children’s Medical Center.
PHACE syndrome is characterized by large, thick, plaque-like hemangiomas greater than 5 cm in size, most commonly on the face, although they can be located elsewhere.
“There was concern that if you found severely altered cerebrovascular arterial flow and you put a kid on a beta-blocker you might be causing some harm. But what I will tell you is that in this recently published paper this was not in fact an issue,” he said.
Dr. Levy was not an investigator in the multicenter retrospective study, which included 76 patients with PHACE syndrome treated for infantile hemangioma with oral propranolol at 0.3 mg/kg per dose or more at 11 academic tertiary care pediatric dermatology clinics. Treatment started at a median age of 56 days.
There were no strokes, TIAs, cardiovascular events, or other significant problems associated with treatment. Twenty-nine children experienced mild adverse events: minor gastrointestinal or respiratory symptoms, and sleep disturbances were threefold more frequent than reported with placebo in another study. The investigators noted that the safety experience in their PHACE syndrome population compared favorably with that in 726 infants without PHACE syndrome who received oral propranolol for hemangiomas, where the incidence of serious adverse events on treatment was 0.4% (JAMA Dermatol. 2019 Dec 11. doi: 10.1001/jamadermatol.2019.3839).
‘Hemangiomas – but we were taught that they go away’
Dr. Levy gave a shout-out to the American Academy of Pediatrics for publishing interdisciplinary expert consensus-based practice guidelines for the management of infantile hemangiomas, which he praised as “quite well done” (Pediatrics. 2019 Jan;143[1]. pii: e20183475. doi: 10.1542/peds.2018-3475).
Following release of the guidelines last year, he and other pediatric vascular anomalies experts saw an uptick in referrals from general pediatricians, which has since tapered off.
“It’s probably like for all of us: We read an article, it’s fresh on the mind, then you forget about the article and what you’ve read. So we need a little reinforcement from a learning perspective. This is a great article,” he said.
The guidelines debunk as myth the classic teaching that infantile hemangiomas go away. Explicit information is provided about the high-risk anatomic sites warranting consideration for early referral, including the periocular, lumbosacral, and perineal areas, the lip, and lower face.
“The major point is early identification of those lesions requiring evaluation and intervention. Hemangiomas generally speaking are at their ultimate size by 3-5 months of age. The bottom line is if you think something needs to be done, please send that patient, or act upon that patient, sooner rather than later. I can’t tell you how many cases of hemangiomas I’ve seen when the kid is 18 months of age, 3 years of age, 5 years, with a large area of redundant skin, scarring, or something of that sort, and it would have been really nice to have seen them earlier and acted upon them then,” the pediatric dermatologist said.
The guidelines recommend intervention or referral by 1 month of age, ideally. Guidance is provided about the use of oral propranolol as first-line therapy.
“Propranolol is something that has been a real game changer for us,” he noted. “Many people continue to be worried about side effects in using this, particularly in the young childhood population, but this paper shows pretty clearly that hypotension or bradycardia is not a real concern. I never hospitalize these patients for propranolol therapy except in high-risk populations: very preemie, any history of breathing problems. We check the blood pressure and heart rate at baseline, again at 7-10 days, and at every visit. We’ve never found any significant drop in blood pressure.”
Dr. Levy reported financial relationships with half a dozen pharmaceutical companies, none relevant to his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the safety of oral propranolol for treatment of complicated infantile hemangiomas in patients with PHACE syndrome comes from a recent multicenter study.
Oral propranolol is now well-ensconced as first-line therapy for complicated infantile hemangiomas in otherwise healthy children. However, the beta-blocker’s use in PHACE (Posterior fossa malformations, Hemangiomas, Arterial anomalies, Cardiac defects, and Eye abnormalities) syndrome has been controversial, with concerns raised by some that it might raise the risk for arterial ischemic stroke. Not so, Moise L. Levy, MD, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I’m not suggesting you use propranolol with reckless abandon in this population, but this stroke concern is something that should be put to bed based on this study,” advised Dr. Levy, professor of dermatology and pediatrics at Dell Medical School in Austin, Tex., and physician-in-chief at Dell Children’s Medical Center.
PHACE syndrome is characterized by large, thick, plaque-like hemangiomas greater than 5 cm in size, most commonly on the face, although they can be located elsewhere.
“There was concern that if you found severely altered cerebrovascular arterial flow and you put a kid on a beta-blocker you might be causing some harm. But what I will tell you is that in this recently published paper this was not in fact an issue,” he said.
Dr. Levy was not an investigator in the multicenter retrospective study, which included 76 patients with PHACE syndrome treated for infantile hemangioma with oral propranolol at 0.3 mg/kg per dose or more at 11 academic tertiary care pediatric dermatology clinics. Treatment started at a median age of 56 days.
There were no strokes, TIAs, cardiovascular events, or other significant problems associated with treatment. Twenty-nine children experienced mild adverse events: minor gastrointestinal or respiratory symptoms, and sleep disturbances were threefold more frequent than reported with placebo in another study. The investigators noted that the safety experience in their PHACE syndrome population compared favorably with that in 726 infants without PHACE syndrome who received oral propranolol for hemangiomas, where the incidence of serious adverse events on treatment was 0.4% (JAMA Dermatol. 2019 Dec 11. doi: 10.1001/jamadermatol.2019.3839).
‘Hemangiomas – but we were taught that they go away’
Dr. Levy gave a shout-out to the American Academy of Pediatrics for publishing interdisciplinary expert consensus-based practice guidelines for the management of infantile hemangiomas, which he praised as “quite well done” (Pediatrics. 2019 Jan;143[1]. pii: e20183475. doi: 10.1542/peds.2018-3475).
Following release of the guidelines last year, he and other pediatric vascular anomalies experts saw an uptick in referrals from general pediatricians, which has since tapered off.
“It’s probably like for all of us: We read an article, it’s fresh on the mind, then you forget about the article and what you’ve read. So we need a little reinforcement from a learning perspective. This is a great article,” he said.
The guidelines debunk as myth the classic teaching that infantile hemangiomas go away. Explicit information is provided about the high-risk anatomic sites warranting consideration for early referral, including the periocular, lumbosacral, and perineal areas, the lip, and lower face.
“The major point is early identification of those lesions requiring evaluation and intervention. Hemangiomas generally speaking are at their ultimate size by 3-5 months of age. The bottom line is if you think something needs to be done, please send that patient, or act upon that patient, sooner rather than later. I can’t tell you how many cases of hemangiomas I’ve seen when the kid is 18 months of age, 3 years of age, 5 years, with a large area of redundant skin, scarring, or something of that sort, and it would have been really nice to have seen them earlier and acted upon them then,” the pediatric dermatologist said.
The guidelines recommend intervention or referral by 1 month of age, ideally. Guidance is provided about the use of oral propranolol as first-line therapy.
“Propranolol is something that has been a real game changer for us,” he noted. “Many people continue to be worried about side effects in using this, particularly in the young childhood population, but this paper shows pretty clearly that hypotension or bradycardia is not a real concern. I never hospitalize these patients for propranolol therapy except in high-risk populations: very preemie, any history of breathing problems. We check the blood pressure and heart rate at baseline, again at 7-10 days, and at every visit. We’ve never found any significant drop in blood pressure.”
Dr. Levy reported financial relationships with half a dozen pharmaceutical companies, none relevant to his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the safety of oral propranolol for treatment of complicated infantile hemangiomas in patients with PHACE syndrome comes from a recent multicenter study.
Oral propranolol is now well-ensconced as first-line therapy for complicated infantile hemangiomas in otherwise healthy children. However, the beta-blocker’s use in PHACE (Posterior fossa malformations, Hemangiomas, Arterial anomalies, Cardiac defects, and Eye abnormalities) syndrome has been controversial, with concerns raised by some that it might raise the risk for arterial ischemic stroke. Not so, Moise L. Levy, MD, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“I’m not suggesting you use propranolol with reckless abandon in this population, but this stroke concern is something that should be put to bed based on this study,” advised Dr. Levy, professor of dermatology and pediatrics at Dell Medical School in Austin, Tex., and physician-in-chief at Dell Children’s Medical Center.
PHACE syndrome is characterized by large, thick, plaque-like hemangiomas greater than 5 cm in size, most commonly on the face, although they can be located elsewhere.
“There was concern that if you found severely altered cerebrovascular arterial flow and you put a kid on a beta-blocker you might be causing some harm. But what I will tell you is that in this recently published paper this was not in fact an issue,” he said.
Dr. Levy was not an investigator in the multicenter retrospective study, which included 76 patients with PHACE syndrome treated for infantile hemangioma with oral propranolol at 0.3 mg/kg per dose or more at 11 academic tertiary care pediatric dermatology clinics. Treatment started at a median age of 56 days.
There were no strokes, TIAs, cardiovascular events, or other significant problems associated with treatment. Twenty-nine children experienced mild adverse events: minor gastrointestinal or respiratory symptoms, and sleep disturbances were threefold more frequent than reported with placebo in another study. The investigators noted that the safety experience in their PHACE syndrome population compared favorably with that in 726 infants without PHACE syndrome who received oral propranolol for hemangiomas, where the incidence of serious adverse events on treatment was 0.4% (JAMA Dermatol. 2019 Dec 11. doi: 10.1001/jamadermatol.2019.3839).
‘Hemangiomas – but we were taught that they go away’
Dr. Levy gave a shout-out to the American Academy of Pediatrics for publishing interdisciplinary expert consensus-based practice guidelines for the management of infantile hemangiomas, which he praised as “quite well done” (Pediatrics. 2019 Jan;143[1]. pii: e20183475. doi: 10.1542/peds.2018-3475).
Following release of the guidelines last year, he and other pediatric vascular anomalies experts saw an uptick in referrals from general pediatricians, which has since tapered off.
“It’s probably like for all of us: We read an article, it’s fresh on the mind, then you forget about the article and what you’ve read. So we need a little reinforcement from a learning perspective. This is a great article,” he said.
The guidelines debunk as myth the classic teaching that infantile hemangiomas go away. Explicit information is provided about the high-risk anatomic sites warranting consideration for early referral, including the periocular, lumbosacral, and perineal areas, the lip, and lower face.
“The major point is early identification of those lesions requiring evaluation and intervention. Hemangiomas generally speaking are at their ultimate size by 3-5 months of age. The bottom line is if you think something needs to be done, please send that patient, or act upon that patient, sooner rather than later. I can’t tell you how many cases of hemangiomas I’ve seen when the kid is 18 months of age, 3 years of age, 5 years, with a large area of redundant skin, scarring, or something of that sort, and it would have been really nice to have seen them earlier and acted upon them then,” the pediatric dermatologist said.
The guidelines recommend intervention or referral by 1 month of age, ideally. Guidance is provided about the use of oral propranolol as first-line therapy.
“Propranolol is something that has been a real game changer for us,” he noted. “Many people continue to be worried about side effects in using this, particularly in the young childhood population, but this paper shows pretty clearly that hypotension or bradycardia is not a real concern. I never hospitalize these patients for propranolol therapy except in high-risk populations: very preemie, any history of breathing problems. We check the blood pressure and heart rate at baseline, again at 7-10 days, and at every visit. We’ve never found any significant drop in blood pressure.”
Dr. Levy reported financial relationships with half a dozen pharmaceutical companies, none relevant to his presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
Don’t call it perioral dermatitis
LAHAINA, HAWAII – , according to Jessica Sprague, MD, a pediatric dermatologist at the University of California, San Diego, and Rady Children’s Hospital.
Years ago, some of her senior colleagues at the children’s hospital carried out a retrospective study of 79 patients, aged 6 months to 18 years, who were treated for what’s typically called perioral dermatitis. Of note, only 40% of patients had isolated perioral involvement, while 30% of the patients had no perioral lesions at all. Perinasal lesions were present in 43%, and 25% had periocular involvement, she noted at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
The peak incidence of periorificial dermatitis in this series was under age 5 years. At presentation, the rash had been present for an average of 8 months. Seventy-two percent of patients had a history of exposure to corticosteroids, most often in the form of topical steroids, but in some cases inhaled or systemic steroids.
“Obviously you want to discontinue the topical steroid. Sometimes you need to taper them off, or you can switch to a topical calcineurin inhibitor [TCI] because they tend to flare a lot when you stop their topical steroid, although there are cases of TCIs precipitating periorificial dermatitis, so keep that in mind,” Dr. Sprague said.
If a patient is on inhaled steroids by mask for asthma, switching to a tube can sometimes limit the exposure, she continued.
Her first-line therapy for mild to moderate periorificial dermatitis, and the one supported by the strongest evidence base, is metronidazole cream. Other topical agents shown to be effective include azelaic acid, sulfacetamide, clindamycin, and topical calcineurin inhibitors.
Oral therapy is a good option for more extensive or recalcitrant cases.
“If parents are very anxious, like before school photos or holiday photos, sometimes I’ll use oral therapy as well. In younger kids, I prefer erythromycin at 30 mg/kg per day t.i.d. for 3-6 weeks. In kids 8 years old and up you can use doxycycline at 50-100 mg b.i.d., again for 3-6 weeks. And you have to tell them it’s going to take a while for this to go away,” Dr. Sprague said.
She reported having no financial conflicts regarding her presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – , according to Jessica Sprague, MD, a pediatric dermatologist at the University of California, San Diego, and Rady Children’s Hospital.
Years ago, some of her senior colleagues at the children’s hospital carried out a retrospective study of 79 patients, aged 6 months to 18 years, who were treated for what’s typically called perioral dermatitis. Of note, only 40% of patients had isolated perioral involvement, while 30% of the patients had no perioral lesions at all. Perinasal lesions were present in 43%, and 25% had periocular involvement, she noted at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
The peak incidence of periorificial dermatitis in this series was under age 5 years. At presentation, the rash had been present for an average of 8 months. Seventy-two percent of patients had a history of exposure to corticosteroids, most often in the form of topical steroids, but in some cases inhaled or systemic steroids.
“Obviously you want to discontinue the topical steroid. Sometimes you need to taper them off, or you can switch to a topical calcineurin inhibitor [TCI] because they tend to flare a lot when you stop their topical steroid, although there are cases of TCIs precipitating periorificial dermatitis, so keep that in mind,” Dr. Sprague said.
If a patient is on inhaled steroids by mask for asthma, switching to a tube can sometimes limit the exposure, she continued.
Her first-line therapy for mild to moderate periorificial dermatitis, and the one supported by the strongest evidence base, is metronidazole cream. Other topical agents shown to be effective include azelaic acid, sulfacetamide, clindamycin, and topical calcineurin inhibitors.
Oral therapy is a good option for more extensive or recalcitrant cases.
“If parents are very anxious, like before school photos or holiday photos, sometimes I’ll use oral therapy as well. In younger kids, I prefer erythromycin at 30 mg/kg per day t.i.d. for 3-6 weeks. In kids 8 years old and up you can use doxycycline at 50-100 mg b.i.d., again for 3-6 weeks. And you have to tell them it’s going to take a while for this to go away,” Dr. Sprague said.
She reported having no financial conflicts regarding her presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – , according to Jessica Sprague, MD, a pediatric dermatologist at the University of California, San Diego, and Rady Children’s Hospital.
Years ago, some of her senior colleagues at the children’s hospital carried out a retrospective study of 79 patients, aged 6 months to 18 years, who were treated for what’s typically called perioral dermatitis. Of note, only 40% of patients had isolated perioral involvement, while 30% of the patients had no perioral lesions at all. Perinasal lesions were present in 43%, and 25% had periocular involvement, she noted at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
The peak incidence of periorificial dermatitis in this series was under age 5 years. At presentation, the rash had been present for an average of 8 months. Seventy-two percent of patients had a history of exposure to corticosteroids, most often in the form of topical steroids, but in some cases inhaled or systemic steroids.
“Obviously you want to discontinue the topical steroid. Sometimes you need to taper them off, or you can switch to a topical calcineurin inhibitor [TCI] because they tend to flare a lot when you stop their topical steroid, although there are cases of TCIs precipitating periorificial dermatitis, so keep that in mind,” Dr. Sprague said.
If a patient is on inhaled steroids by mask for asthma, switching to a tube can sometimes limit the exposure, she continued.
Her first-line therapy for mild to moderate periorificial dermatitis, and the one supported by the strongest evidence base, is metronidazole cream. Other topical agents shown to be effective include azelaic acid, sulfacetamide, clindamycin, and topical calcineurin inhibitors.
Oral therapy is a good option for more extensive or recalcitrant cases.
“If parents are very anxious, like before school photos or holiday photos, sometimes I’ll use oral therapy as well. In younger kids, I prefer erythromycin at 30 mg/kg per day t.i.d. for 3-6 weeks. In kids 8 years old and up you can use doxycycline at 50-100 mg b.i.d., again for 3-6 weeks. And you have to tell them it’s going to take a while for this to go away,” Dr. Sprague said.
She reported having no financial conflicts regarding her presentation.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
New topicals coming for pediatric atopic dermatitis
LAHAINA, HAWAII – Novel topical medications are in the works that will address the longstanding unmet need for a Food and Drug Administration–approved noncorticosteroid topical for use in pediatric atopic dermatitis, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
These new agents will be embraced by clinicians for use in delicate skin areas, as well as in the common clinical scenario involving steroid-averse parents, predicted Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
First up is crisaborole (Eucrisa), which is approved for atopic dermatitis (AD) in children aged two years and older and has been under review at the Food and Drug Administration for use in infantile AD. (On March 24, several weeks after the meeting, the FDA approved crisaborole down to aged three months for treatment of mild to moderate AD). Agents earlier in the developmental pipeline include two topical Janus kinase (JAK) inhibitors, ruxolitinib and delgocitinib, as well as tapinarof.
Crisaborole: This phosphodiesterase 4 inhibitor is FDA approved down to 2 years of age. In the phase 4, open-label CrisADe CARE 1 study, crisaborole was studied in 137 children ages 3 months to under 24 months. CrisADe CARE 1, presented at the 2019 annual conference of the Pediatric Dermatology Research Alliance (PeDRA), showed close to a 60% reduction from baseline in Eczema Area and Severity Index (EASI) scores after 28 days of twice-daily therapy in the youngsters, 61% of who had moderate AD, the rest mild disease.
Tolerability and safety were reassuring in the phase 4 study. Although about 3% of subjects each experienced application site pain, discomfort, or erythema, the rate of study discontinuation was impressively low at 2.9%, Dr. Eichenfield observed.
Delgocitinib: Japanese investigators have reported positive results in a phase 2 study of delgocitinib ointment in 98 children and adolescents aged 2-15 years, with AD. After 4 weeks of twice-daily treatment, modified EASI scores improved by a mean of 54% with delgocitinib 0.25% and by 62% with 0.5%, compared with less than a 5% improvement with the vehicle control (J Allergy Clin Immunol. 2019 Dec;144[6]:1575-83). The ointment formulation is being developed specifically for the Japanese market.
Studies of an alternative formulation of the JAK inhibitor as a cream rather than ointment, intended for the U.S. and European markets, are in the early stages, conducted by Leo Pharma. Delgocitinib cream, under study in adults and children down to age 2 years with AD, is also under study for chronic hand dermatitis, a program Dr. Eichenfield is enthusiastic about.
“Hand eczema is something you’re going to hear a lot about in the next 2 years. In the U.S., we have no drug approved specifically for hand eczema. And we actually see a lot of hand eczema in pediatric and adolescent patients. I’d say 75%-80% of the ones I see also have atopic dermatitis,” he said.
Ruxolitinib: Incyte, which is developing the topical JAK inhibitor, recently announced positive results in the first of four phase 3 randomized trials, this one conducted in AD patients aged 12 years and older. The efficacy appears to be comparable to that of topical steroids. Studies in younger children are also planned. Ruxolitinib cream is in advanced clinical trials for treatment of vitiligo.
Tapinarof: This topical aryl hydrocarbon receptor agonist downregulates Th17 cytokines, an attribute desirable for treatment of psoriasis. But it also downregulates Th2 cytokines and improves the damaged skin barrier characteristic of AD via upregulation of the filaggrin and involucrin genes in keratinocytes. In a phase 2b, double-blind clinical trial conducted in 247 adults and adolescents with moderate to severe AD, 12 weeks of once-daily tapinarof 1% enabled 51% of patients to achieve a 75% or greater improvement in EASI scores, compared with 18% in controls on vehicle (J Am Acad Dermatol. 2019 Jan;80[1]:89-98.e3).
Dermavant, which is developing the drug, plans to seek an initial indication for treatment of psoriasis, where a phase 3 study is underway, before pursuing regulatory approval in AD.
Dr. Eichenfield disclosed serving as a consultant or investigator for various pharmaceutical companies, including Pfizer, and Dermavant.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
This article was updated 3/27/20.
LAHAINA, HAWAII – Novel topical medications are in the works that will address the longstanding unmet need for a Food and Drug Administration–approved noncorticosteroid topical for use in pediatric atopic dermatitis, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
These new agents will be embraced by clinicians for use in delicate skin areas, as well as in the common clinical scenario involving steroid-averse parents, predicted Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
First up is crisaborole (Eucrisa), which is approved for atopic dermatitis (AD) in children aged two years and older and has been under review at the Food and Drug Administration for use in infantile AD. (On March 24, several weeks after the meeting, the FDA approved crisaborole down to aged three months for treatment of mild to moderate AD). Agents earlier in the developmental pipeline include two topical Janus kinase (JAK) inhibitors, ruxolitinib and delgocitinib, as well as tapinarof.
Crisaborole: This phosphodiesterase 4 inhibitor is FDA approved down to 2 years of age. In the phase 4, open-label CrisADe CARE 1 study, crisaborole was studied in 137 children ages 3 months to under 24 months. CrisADe CARE 1, presented at the 2019 annual conference of the Pediatric Dermatology Research Alliance (PeDRA), showed close to a 60% reduction from baseline in Eczema Area and Severity Index (EASI) scores after 28 days of twice-daily therapy in the youngsters, 61% of who had moderate AD, the rest mild disease.
Tolerability and safety were reassuring in the phase 4 study. Although about 3% of subjects each experienced application site pain, discomfort, or erythema, the rate of study discontinuation was impressively low at 2.9%, Dr. Eichenfield observed.
Delgocitinib: Japanese investigators have reported positive results in a phase 2 study of delgocitinib ointment in 98 children and adolescents aged 2-15 years, with AD. After 4 weeks of twice-daily treatment, modified EASI scores improved by a mean of 54% with delgocitinib 0.25% and by 62% with 0.5%, compared with less than a 5% improvement with the vehicle control (J Allergy Clin Immunol. 2019 Dec;144[6]:1575-83). The ointment formulation is being developed specifically for the Japanese market.
Studies of an alternative formulation of the JAK inhibitor as a cream rather than ointment, intended for the U.S. and European markets, are in the early stages, conducted by Leo Pharma. Delgocitinib cream, under study in adults and children down to age 2 years with AD, is also under study for chronic hand dermatitis, a program Dr. Eichenfield is enthusiastic about.
“Hand eczema is something you’re going to hear a lot about in the next 2 years. In the U.S., we have no drug approved specifically for hand eczema. And we actually see a lot of hand eczema in pediatric and adolescent patients. I’d say 75%-80% of the ones I see also have atopic dermatitis,” he said.
Ruxolitinib: Incyte, which is developing the topical JAK inhibitor, recently announced positive results in the first of four phase 3 randomized trials, this one conducted in AD patients aged 12 years and older. The efficacy appears to be comparable to that of topical steroids. Studies in younger children are also planned. Ruxolitinib cream is in advanced clinical trials for treatment of vitiligo.
Tapinarof: This topical aryl hydrocarbon receptor agonist downregulates Th17 cytokines, an attribute desirable for treatment of psoriasis. But it also downregulates Th2 cytokines and improves the damaged skin barrier characteristic of AD via upregulation of the filaggrin and involucrin genes in keratinocytes. In a phase 2b, double-blind clinical trial conducted in 247 adults and adolescents with moderate to severe AD, 12 weeks of once-daily tapinarof 1% enabled 51% of patients to achieve a 75% or greater improvement in EASI scores, compared with 18% in controls on vehicle (J Am Acad Dermatol. 2019 Jan;80[1]:89-98.e3).
Dermavant, which is developing the drug, plans to seek an initial indication for treatment of psoriasis, where a phase 3 study is underway, before pursuing regulatory approval in AD.
Dr. Eichenfield disclosed serving as a consultant or investigator for various pharmaceutical companies, including Pfizer, and Dermavant.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
This article was updated 3/27/20.
LAHAINA, HAWAII – Novel topical medications are in the works that will address the longstanding unmet need for a Food and Drug Administration–approved noncorticosteroid topical for use in pediatric atopic dermatitis, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
These new agents will be embraced by clinicians for use in delicate skin areas, as well as in the common clinical scenario involving steroid-averse parents, predicted Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
First up is crisaborole (Eucrisa), which is approved for atopic dermatitis (AD) in children aged two years and older and has been under review at the Food and Drug Administration for use in infantile AD. (On March 24, several weeks after the meeting, the FDA approved crisaborole down to aged three months for treatment of mild to moderate AD). Agents earlier in the developmental pipeline include two topical Janus kinase (JAK) inhibitors, ruxolitinib and delgocitinib, as well as tapinarof.
Crisaborole: This phosphodiesterase 4 inhibitor is FDA approved down to 2 years of age. In the phase 4, open-label CrisADe CARE 1 study, crisaborole was studied in 137 children ages 3 months to under 24 months. CrisADe CARE 1, presented at the 2019 annual conference of the Pediatric Dermatology Research Alliance (PeDRA), showed close to a 60% reduction from baseline in Eczema Area and Severity Index (EASI) scores after 28 days of twice-daily therapy in the youngsters, 61% of who had moderate AD, the rest mild disease.
Tolerability and safety were reassuring in the phase 4 study. Although about 3% of subjects each experienced application site pain, discomfort, or erythema, the rate of study discontinuation was impressively low at 2.9%, Dr. Eichenfield observed.
Delgocitinib: Japanese investigators have reported positive results in a phase 2 study of delgocitinib ointment in 98 children and adolescents aged 2-15 years, with AD. After 4 weeks of twice-daily treatment, modified EASI scores improved by a mean of 54% with delgocitinib 0.25% and by 62% with 0.5%, compared with less than a 5% improvement with the vehicle control (J Allergy Clin Immunol. 2019 Dec;144[6]:1575-83). The ointment formulation is being developed specifically for the Japanese market.
Studies of an alternative formulation of the JAK inhibitor as a cream rather than ointment, intended for the U.S. and European markets, are in the early stages, conducted by Leo Pharma. Delgocitinib cream, under study in adults and children down to age 2 years with AD, is also under study for chronic hand dermatitis, a program Dr. Eichenfield is enthusiastic about.
“Hand eczema is something you’re going to hear a lot about in the next 2 years. In the U.S., we have no drug approved specifically for hand eczema. And we actually see a lot of hand eczema in pediatric and adolescent patients. I’d say 75%-80% of the ones I see also have atopic dermatitis,” he said.
Ruxolitinib: Incyte, which is developing the topical JAK inhibitor, recently announced positive results in the first of four phase 3 randomized trials, this one conducted in AD patients aged 12 years and older. The efficacy appears to be comparable to that of topical steroids. Studies in younger children are also planned. Ruxolitinib cream is in advanced clinical trials for treatment of vitiligo.
Tapinarof: This topical aryl hydrocarbon receptor agonist downregulates Th17 cytokines, an attribute desirable for treatment of psoriasis. But it also downregulates Th2 cytokines and improves the damaged skin barrier characteristic of AD via upregulation of the filaggrin and involucrin genes in keratinocytes. In a phase 2b, double-blind clinical trial conducted in 247 adults and adolescents with moderate to severe AD, 12 weeks of once-daily tapinarof 1% enabled 51% of patients to achieve a 75% or greater improvement in EASI scores, compared with 18% in controls on vehicle (J Am Acad Dermatol. 2019 Jan;80[1]:89-98.e3).
Dermavant, which is developing the drug, plans to seek an initial indication for treatment of psoriasis, where a phase 3 study is underway, before pursuing regulatory approval in AD.
Dr. Eichenfield disclosed serving as a consultant or investigator for various pharmaceutical companies, including Pfizer, and Dermavant.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
This article was updated 3/27/20.
REPORTING FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
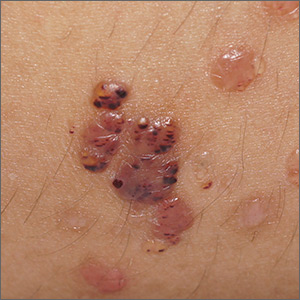
Vesicles on the thigh
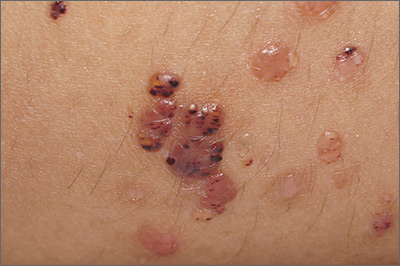
On very close inspection, the physician noted translucent chambers within each vesicle, and within each chamber there was a horizontal line (parallel to the floor) separating serum-colored fluid from dark blood. This unique appearance prompted the physician to diagnose lymphangioma circumscriptum in this patient.
Lymphangioma circumscriptum is a rare type of microcytic lymphatic malformation most commonly found on the shoulders, limbs, axilla, and tongue that may enlarge during puberty. The clustered vesicles are firm. Vesicles can be red, brown, or straw colored in appearance and are focal to widespread; rarely, they may bleed or become infected. Their appearance has been compared to frog spawn.
Lymphangioma circumscriptum is benign and requires no treatment. Any suspected infection could be treated with antibiotics. If removal is desired for cosmesis or functional treatment, areas may be treated with dermabrasion, sclerotherapy, laser ablation, or excision if feasible. Lymphangioma circumscriptum tends to recur in time and appropriate anticipatory guidance is key.
This patient was treated with sclerotherapy using hypertonic saline that was injected monthly for 3 months. The physician injected a 30-g needle into the broadest ectatic chambers after each area was anesthetized with lidocaine. The patient tolerated the injections well, and the treated areas resolved as slightly hypopigmented macules. No recurrence was noted at posttreatment follow-up 1 year later.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Bikowski JB, Dumont AM. Lymphangioma circumscriptum: treatment with hypertonic saline sclerotherapy. J Am Acad Dermatol. 2005;53:442-444.

On very close inspection, the physician noted translucent chambers within each vesicle, and within each chamber there was a horizontal line (parallel to the floor) separating serum-colored fluid from dark blood. This unique appearance prompted the physician to diagnose lymphangioma circumscriptum in this patient.
Lymphangioma circumscriptum is a rare type of microcytic lymphatic malformation most commonly found on the shoulders, limbs, axilla, and tongue that may enlarge during puberty. The clustered vesicles are firm. Vesicles can be red, brown, or straw colored in appearance and are focal to widespread; rarely, they may bleed or become infected. Their appearance has been compared to frog spawn.
Lymphangioma circumscriptum is benign and requires no treatment. Any suspected infection could be treated with antibiotics. If removal is desired for cosmesis or functional treatment, areas may be treated with dermabrasion, sclerotherapy, laser ablation, or excision if feasible. Lymphangioma circumscriptum tends to recur in time and appropriate anticipatory guidance is key.
This patient was treated with sclerotherapy using hypertonic saline that was injected monthly for 3 months. The physician injected a 30-g needle into the broadest ectatic chambers after each area was anesthetized with lidocaine. The patient tolerated the injections well, and the treated areas resolved as slightly hypopigmented macules. No recurrence was noted at posttreatment follow-up 1 year later.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

On very close inspection, the physician noted translucent chambers within each vesicle, and within each chamber there was a horizontal line (parallel to the floor) separating serum-colored fluid from dark blood. This unique appearance prompted the physician to diagnose lymphangioma circumscriptum in this patient.
Lymphangioma circumscriptum is a rare type of microcytic lymphatic malformation most commonly found on the shoulders, limbs, axilla, and tongue that may enlarge during puberty. The clustered vesicles are firm. Vesicles can be red, brown, or straw colored in appearance and are focal to widespread; rarely, they may bleed or become infected. Their appearance has been compared to frog spawn.
Lymphangioma circumscriptum is benign and requires no treatment. Any suspected infection could be treated with antibiotics. If removal is desired for cosmesis or functional treatment, areas may be treated with dermabrasion, sclerotherapy, laser ablation, or excision if feasible. Lymphangioma circumscriptum tends to recur in time and appropriate anticipatory guidance is key.
This patient was treated with sclerotherapy using hypertonic saline that was injected monthly for 3 months. The physician injected a 30-g needle into the broadest ectatic chambers after each area was anesthetized with lidocaine. The patient tolerated the injections well, and the treated areas resolved as slightly hypopigmented macules. No recurrence was noted at posttreatment follow-up 1 year later.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Bikowski JB, Dumont AM. Lymphangioma circumscriptum: treatment with hypertonic saline sclerotherapy. J Am Acad Dermatol. 2005;53:442-444.
Bikowski JB, Dumont AM. Lymphangioma circumscriptum: treatment with hypertonic saline sclerotherapy. J Am Acad Dermatol. 2005;53:442-444.
New melanoma treatments linked to mortality decline
Recent advances in treatment appear to have reversed the course of melanoma mortality since 2013, according to data published in the American Journal of Public Health.

The U.S. death rate for melanoma, which had been rising at a rate of 0.22% a year for more than 2 decades, dropped by 17.9%, or 6.24% per year, during 2013-2016. That decline “coincides with the introduction of multiple new and efficacious treatments for metastatic melanoma,” such as BRAF inhibitors and immune checkpoint inhibitors, study author Juliana Berk-Krauss, MD, of the State University of New York Downstate Medical Center in Brooklyn and colleagues wrote.
The other possible explanation for the decline in deaths, “education and early detection resulting in migration toward earlier stage melanomas with a greater chance of surgical cure,” is unlikely, according to the investigators. That’s because the small decrease in median tumor thickness that occurred during 1989-2009 “is not associated with changes in prognosis.”
The investigators’ analysis encompassed data from the Surveillance, Epidemiology, and End Results registry recorded during 1986-2016. Nine registry areas were included (Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, and Utah), which covered about 9.4% of the U.S. population. The analysis was limited to the white population, which accounts for more than 90% of melanoma cases in the United States.
The data showed a slight decline in annual percent change in melanoma incidence, from 3.24% for 1986-2005 to 1.72% for 2006-2016. However, over the whole period studied (1986-2016), melanoma incidence increased by 108%, or about 2.7% per year.
“Given the increased incidence of melanoma throughout this period and the lack of stage migration, these data strongly suggest that the mortality decline is due to the extended survival associated with these [newer] treatments,” the investigators wrote.
This study was funded by NYU Langone. Two investigators disclosed potential conflicts of interest, including relationships with Bio-Rad Laboratories, Novartis, Merck, and several other companies.
SOURCE: Berk-Krauss J et al. Am J Public Health. 2020 Mar 19. doi: 10.2105/AJPH.2020.305567.
Recent advances in treatment appear to have reversed the course of melanoma mortality since 2013, according to data published in the American Journal of Public Health.

The U.S. death rate for melanoma, which had been rising at a rate of 0.22% a year for more than 2 decades, dropped by 17.9%, or 6.24% per year, during 2013-2016. That decline “coincides with the introduction of multiple new and efficacious treatments for metastatic melanoma,” such as BRAF inhibitors and immune checkpoint inhibitors, study author Juliana Berk-Krauss, MD, of the State University of New York Downstate Medical Center in Brooklyn and colleagues wrote.
The other possible explanation for the decline in deaths, “education and early detection resulting in migration toward earlier stage melanomas with a greater chance of surgical cure,” is unlikely, according to the investigators. That’s because the small decrease in median tumor thickness that occurred during 1989-2009 “is not associated with changes in prognosis.”
The investigators’ analysis encompassed data from the Surveillance, Epidemiology, and End Results registry recorded during 1986-2016. Nine registry areas were included (Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, and Utah), which covered about 9.4% of the U.S. population. The analysis was limited to the white population, which accounts for more than 90% of melanoma cases in the United States.
The data showed a slight decline in annual percent change in melanoma incidence, from 3.24% for 1986-2005 to 1.72% for 2006-2016. However, over the whole period studied (1986-2016), melanoma incidence increased by 108%, or about 2.7% per year.
“Given the increased incidence of melanoma throughout this period and the lack of stage migration, these data strongly suggest that the mortality decline is due to the extended survival associated with these [newer] treatments,” the investigators wrote.
This study was funded by NYU Langone. Two investigators disclosed potential conflicts of interest, including relationships with Bio-Rad Laboratories, Novartis, Merck, and several other companies.
SOURCE: Berk-Krauss J et al. Am J Public Health. 2020 Mar 19. doi: 10.2105/AJPH.2020.305567.
Recent advances in treatment appear to have reversed the course of melanoma mortality since 2013, according to data published in the American Journal of Public Health.

The U.S. death rate for melanoma, which had been rising at a rate of 0.22% a year for more than 2 decades, dropped by 17.9%, or 6.24% per year, during 2013-2016. That decline “coincides with the introduction of multiple new and efficacious treatments for metastatic melanoma,” such as BRAF inhibitors and immune checkpoint inhibitors, study author Juliana Berk-Krauss, MD, of the State University of New York Downstate Medical Center in Brooklyn and colleagues wrote.
The other possible explanation for the decline in deaths, “education and early detection resulting in migration toward earlier stage melanomas with a greater chance of surgical cure,” is unlikely, according to the investigators. That’s because the small decrease in median tumor thickness that occurred during 1989-2009 “is not associated with changes in prognosis.”
The investigators’ analysis encompassed data from the Surveillance, Epidemiology, and End Results registry recorded during 1986-2016. Nine registry areas were included (Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, and Utah), which covered about 9.4% of the U.S. population. The analysis was limited to the white population, which accounts for more than 90% of melanoma cases in the United States.
The data showed a slight decline in annual percent change in melanoma incidence, from 3.24% for 1986-2005 to 1.72% for 2006-2016. However, over the whole period studied (1986-2016), melanoma incidence increased by 108%, or about 2.7% per year.
“Given the increased incidence of melanoma throughout this period and the lack of stage migration, these data strongly suggest that the mortality decline is due to the extended survival associated with these [newer] treatments,” the investigators wrote.
This study was funded by NYU Langone. Two investigators disclosed potential conflicts of interest, including relationships with Bio-Rad Laboratories, Novartis, Merck, and several other companies.
SOURCE: Berk-Krauss J et al. Am J Public Health. 2020 Mar 19. doi: 10.2105/AJPH.2020.305567.
FROM THE AMERICAN JOURNAL OF PUBLIC HEALTH
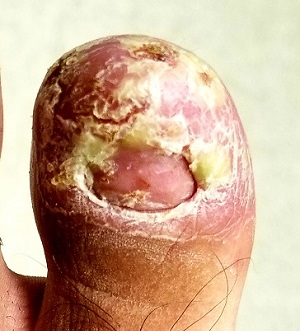
Digits in Distress
ANSWER
The correct answer is acrodermatitis of Hallopeau (choice “c”).
DISCUSSION
Acrodermatitis of Hallopeau (ADH), a rare form of pustular psoriasis, affects the distal digits with changes typified by this patient’s case. It is notoriously difficult to treat, although the advent of the “biologic age” appears to offer a quantum leap in terms of effective treatment alternatives.
Prompt referral to dermatology is needed for ADH to be readily diagnosed and treated. Because ADH is both rare and obscure, it’s not surprising that so many patients suffer for years due to an incorrect diagnosis. And even when correctly diagnosed, treatment is far from satisfactory. For example, methotrexate is commonly used for psoriasis vulgaris but rarely improves ADH—nor do topical steroids or vitamin D–derived topicals.
The 3 other items in the differential would not explain the patient’s condition: (1) Except in cases of immunosuppression, we would not expect Candida (choice “a”) or any other yeast infection to manifest in this manner. (2) Atypical mycobacteria (choice “b”)—such as Mycobacterium marinum—can cause skin infections, but not in a chronically relapsing manner. Moreover, this patient was given minocycline, which would have quickly cleared or at least improved his condition. (3) Pityriasis rubra pilaris (choice “d”) is an unusual papulosquamous disease that can affect nails, but its manifestation would not be as limited as this patient’s was.
TREATMENT
Because the patient’s symptoms are sometimes severe enough to interfere with almost all daily activities, he is clearly in need of prompt treatment. In more severe cases, a short course of cyclosporine followed by a biologic can be an option. For this patient, adalimumab was used. Although it is too early to tell, we expect him to be much improved within a few months.
ANSWER
The correct answer is acrodermatitis of Hallopeau (choice “c”).
DISCUSSION
Acrodermatitis of Hallopeau (ADH), a rare form of pustular psoriasis, affects the distal digits with changes typified by this patient’s case. It is notoriously difficult to treat, although the advent of the “biologic age” appears to offer a quantum leap in terms of effective treatment alternatives.
Prompt referral to dermatology is needed for ADH to be readily diagnosed and treated. Because ADH is both rare and obscure, it’s not surprising that so many patients suffer for years due to an incorrect diagnosis. And even when correctly diagnosed, treatment is far from satisfactory. For example, methotrexate is commonly used for psoriasis vulgaris but rarely improves ADH—nor do topical steroids or vitamin D–derived topicals.
The 3 other items in the differential would not explain the patient’s condition: (1) Except in cases of immunosuppression, we would not expect Candida (choice “a”) or any other yeast infection to manifest in this manner. (2) Atypical mycobacteria (choice “b”)—such as Mycobacterium marinum—can cause skin infections, but not in a chronically relapsing manner. Moreover, this patient was given minocycline, which would have quickly cleared or at least improved his condition. (3) Pityriasis rubra pilaris (choice “d”) is an unusual papulosquamous disease that can affect nails, but its manifestation would not be as limited as this patient’s was.
TREATMENT
Because the patient’s symptoms are sometimes severe enough to interfere with almost all daily activities, he is clearly in need of prompt treatment. In more severe cases, a short course of cyclosporine followed by a biologic can be an option. For this patient, adalimumab was used. Although it is too early to tell, we expect him to be much improved within a few months.
ANSWER
The correct answer is acrodermatitis of Hallopeau (choice “c”).
DISCUSSION
Acrodermatitis of Hallopeau (ADH), a rare form of pustular psoriasis, affects the distal digits with changes typified by this patient’s case. It is notoriously difficult to treat, although the advent of the “biologic age” appears to offer a quantum leap in terms of effective treatment alternatives.
Prompt referral to dermatology is needed for ADH to be readily diagnosed and treated. Because ADH is both rare and obscure, it’s not surprising that so many patients suffer for years due to an incorrect diagnosis. And even when correctly diagnosed, treatment is far from satisfactory. For example, methotrexate is commonly used for psoriasis vulgaris but rarely improves ADH—nor do topical steroids or vitamin D–derived topicals.
The 3 other items in the differential would not explain the patient’s condition: (1) Except in cases of immunosuppression, we would not expect Candida (choice “a”) or any other yeast infection to manifest in this manner. (2) Atypical mycobacteria (choice “b”)—such as Mycobacterium marinum—can cause skin infections, but not in a chronically relapsing manner. Moreover, this patient was given minocycline, which would have quickly cleared or at least improved his condition. (3) Pityriasis rubra pilaris (choice “d”) is an unusual papulosquamous disease that can affect nails, but its manifestation would not be as limited as this patient’s was.
TREATMENT
Because the patient’s symptoms are sometimes severe enough to interfere with almost all daily activities, he is clearly in need of prompt treatment. In more severe cases, a short course of cyclosporine followed by a biologic can be an option. For this patient, adalimumab was used. Although it is too early to tell, we expect him to be much improved within a few months.

For years, a 29-year-old man has been troubled by persistent painful outbreaks on his right thumb and forefinger and left great toe. The condition sometimes prevents him from engaging in daily activities. Several health care providers—none a dermatology specialist—attempted to treat him with topical and oral antifungals (clotrimazole and terbinafine), topical steroids (triamcinolone and clobetasol), and oral antibiotics (including minocycline)—none of which had an impact.
His latest provider, a podiatrist, was sure the problem was fungal in origin and prescribed another course of antifungal treatment. When this failed to produce a benefit, the podiatrist conceded that he was at a loss and referred the patient to dermatology.
On physical exam, the 3 affected digits show similar characteristics: the skin is covered by dense, tenacious scaling on a dark red base. Small pustules are noted on these areas in addition to marked dystrophy of the adjacent nails. Bacterial and fungal cultures of the pustular fluid show no growth.
The rest of the patient’s skin—elbows, oral mucosa, knees, and scalp—show no noteworthy changes. He has no other skin problems. There is no family history of psoriasis or other skin conditions.
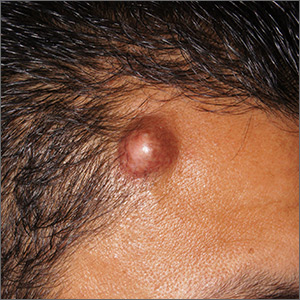
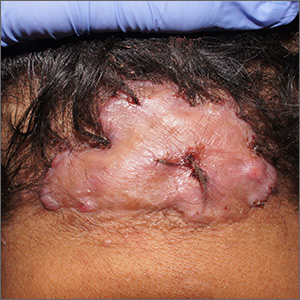
Forehead cyst

Upon palpation, the physician noted a strong pulse consistent with a traumatic arteriovenous fistula (in this case involving the superficial temporal artery). This finding, combined with the cyst’s appearance and the patient’s history, led the physician conclude that this was an epidermoid (sebaceous) cyst. (Prior to palpation, the visual differential diagnosis included dermoid cyst, lipoma, trichilemmal or epidermoid cyst, and foreign body granuloma.)
Cystic nodules on the forehead and midline deserve close scrutiny. This author has seen 3 similar cases in 10 years of daily dermatology consultative practice that have involved the superficial temporal artery and a history of head trauma. Each case had been stable for many months before presentation and had been incorrectly identified as a more common benign cyst.
In this particular case, the planned procedure in the outpatient setting was cancelled and the patient was referred to Vascular Surgery, where surgeons were planning to perform a ligation of the superficial temporal artery.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Matsumoto H, Yamaura I, Yoshida Y. Identity of growing pulsatile mass lesion of the scalp after blunt head injury: case reports and literature review. Trauma Case Rep. 2018;17:43-47.

Upon palpation, the physician noted a strong pulse consistent with a traumatic arteriovenous fistula (in this case involving the superficial temporal artery). This finding, combined with the cyst’s appearance and the patient’s history, led the physician conclude that this was an epidermoid (sebaceous) cyst. (Prior to palpation, the visual differential diagnosis included dermoid cyst, lipoma, trichilemmal or epidermoid cyst, and foreign body granuloma.)
Cystic nodules on the forehead and midline deserve close scrutiny. This author has seen 3 similar cases in 10 years of daily dermatology consultative practice that have involved the superficial temporal artery and a history of head trauma. Each case had been stable for many months before presentation and had been incorrectly identified as a more common benign cyst.
In this particular case, the planned procedure in the outpatient setting was cancelled and the patient was referred to Vascular Surgery, where surgeons were planning to perform a ligation of the superficial temporal artery.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Upon palpation, the physician noted a strong pulse consistent with a traumatic arteriovenous fistula (in this case involving the superficial temporal artery). This finding, combined with the cyst’s appearance and the patient’s history, led the physician conclude that this was an epidermoid (sebaceous) cyst. (Prior to palpation, the visual differential diagnosis included dermoid cyst, lipoma, trichilemmal or epidermoid cyst, and foreign body granuloma.)
Cystic nodules on the forehead and midline deserve close scrutiny. This author has seen 3 similar cases in 10 years of daily dermatology consultative practice that have involved the superficial temporal artery and a history of head trauma. Each case had been stable for many months before presentation and had been incorrectly identified as a more common benign cyst.
In this particular case, the planned procedure in the outpatient setting was cancelled and the patient was referred to Vascular Surgery, where surgeons were planning to perform a ligation of the superficial temporal artery.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Matsumoto H, Yamaura I, Yoshida Y. Identity of growing pulsatile mass lesion of the scalp after blunt head injury: case reports and literature review. Trauma Case Rep. 2018;17:43-47.
Matsumoto H, Yamaura I, Yoshida Y. Identity of growing pulsatile mass lesion of the scalp after blunt head injury: case reports and literature review. Trauma Case Rep. 2018;17:43-47.
Dupilumab approval sought for AD under age 12
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Reassuring evidence of the long-term effectiveness and safety of dupilumab in adolescents with moderate to severe atopic dermatitis comes from a phase 3 open-label extension study of the first teenagers in the world to have received the monoclonal antibody, Lawrence F. Eichenfield, MD, reported at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dupilumab (Dupixent), a monoclonal antibody directed against interleukins-4 and -13 initially approved in adults, received an expanded indication from the Food and Drug Administration in March 2019 for treatment of 12- to 17-year-olds with moderate to severe atopic dermatitis (AD) on the strength of a pivotal 251-patient, phase 3 randomized trial of 16 weeks’ duration (JAMA Dermatol. 2019 Nov 6. doi: 10.1001/jamadermatol.2019.3336). But since AD is a chronic disease, it was important to learn how dupilumab performs well beyond the 16-week mark in adolescents, observed Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital.
In addition to highlighting some of the emerging fine points of dupilumab therapy in adolescents, Dr. Eichenfield discussed the clinical implications of a potential further expanded indication for treatment of 6- to 12-year-olds, an event he considers likely in the coming months. He also described early data from an ongoing dupilumab clinical trials program in the 2- to 5-year-olds.
Long-term dupilumab in teens
Dr. Eichenfield was a coauthor of the recently published phase 3 international long-term extension study. The 40 participants experienced a mean 85% decrease from baseline at 52 weeks in EASI (Eczema Area and Severity Index) scores on 2 mg/kg per week dosing and an 84% reduction on 4 mg/kg per week dosing. This represented a substantial further improvement from week 2, when the EASI reductions were 34% and 51%, respectively.
The mean trough serum dupilumab concentrations over the course of the year were markedly lower in the 2 mg/kg group: 74 mg/L, as compared to 161 mg/L with dosing at 4 mg/kg per week (Br J Dermatol. 2020 Jan;182[1]:85-96).
“It’ll be interesting to see how this works out over time,” the dermatologist commented. “The issue of dose by weight becomes important as we start to treat younger patients because the pharmacokinetics are very different at 4 and 2 mg/kg, and it may have an impact on efficacy.”
The extension study also established the safety and effectiveness of utilizing dupilumab in combination with standard topical corticosteroid therapy, which wasn’t allowed in the pivotal 16-week trial.
Some have commented that dupilumab may be less effective in adolescents than in adults. They point to the 24% rate of an Investigator Global Assessment (IGA) of 0 or 1 – that is, clear or almost clear – at week 16 in the pivotal adolescent trial, a substantially lower rate than in the adult trials. However, Dr. Eichenfield noted that the adolescent study population was heavily skewed to the severe end of the disease spectrum, the placebo response rate was very low, and the absolute placebo-subtracted benefit turned out to be quite similar to what was seen in the adult trials. Moreover, he added, in a post hoc analysis of the pivotal trial data which utilized a different measure of clinically meaningful response – a composite of either a 50% reduction in EASI score, a 3-point or greater improvement on a 10-point pruritus scale, or at least a 6-point improvement from baseline on the Children’s Dermatology Quality Life Index – that outcome was achieved by 74% of adolescents who didn’t achieve clear or almost clear.
What’s next for dupilumab in pediatric AD
Approval of dupilumab in children under aged 12 years is eagerly awaited, Dr. Eichenfield said. The Food and Drug Administration is now analyzing as-yet unreleased data from completed clinical trials of dupilumab in 6- to 12-year-olds with moderate to severe AD with an eye toward a possible further expanded indication. The side effect profile appears to be the same as in 12- to 18-year-olds.
“I assume it will be approved,” Dr. Eichenfield said. “We don’t know what’s going to happen in 6- to 12-year-olds in terms of the ultimate dosing recommendations that will be put out, but be aware that the pharmacokinetics vary by weight over time.”
Early data in children aged 2-5 years with severe AD from the phase 2, open-label, single ascending dose Liberty AD PRESCHOOL study showed that weight-based dosing in that age group made a big difference in terms of pharmacokinetics. In terms of efficacy, the mean reduction in EASI scores 4 weeks after a single dose of dupilumab was 27% with 3 mg/kg and 49% with 6 mg/kg.
Avoidance of live vaccines while on dupilumab becomes more of a consideration in the under-12 population. The second dose of varicella is supposed to be administered at 4 to 6 years of age, as is the second dose of MMR. The nasal influenza vaccine is a live virus vaccine, as is the yellow fever vaccine.
“We don’t know if live vaccines are dangerous for someone on dupilumab, it’s just that it’s listed that you shouldn’t use them and they haven’t been studied,” Dr. Eichenfield observed.
He reported receiving research grants from or serving as a consultant to several dozen pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
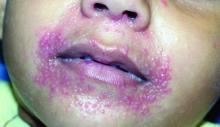
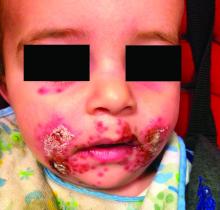
A 7-month-old male presents with perioral rash and fever
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at [email protected].
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at [email protected].
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Patients with atopic dermatitis are at risk for developing the herpes simplex virus (HSV)–related skin complication “eczema herpeticum,” also known as Kaposi’s varicelliform eruption. Eczema herpeticum is characterized by cutaneous pain and vesicular skin lesions, most commonly secondary to infection with HSV-1. The condition may affect individuals with atopic dermatitis or other inflammatory skin disorders. Eczema herpeticum develops when the virus infects large areas of skin, rather than being confined to a small area as in the common cold sore. Eczema herpeticum often appears on the face and neck, although it can appear anywhere on the body. In some cases, the rash may be difficult to distinguish from a patient’s baseline eczema if the latter is poorly controlled. Skin symptoms of eczema herpeticum include clusters of small blisters that are itchy and painful; vesicles that appear red, purple, or black; purulent blisters; or crusting. Classically, the morphology of vesicles or crusted lesions shows a “cluster of grapes” appearance. Eczema herpeticum may present with a high fever, chills, and swollen lymph glands.
While a clinical diagnosis based on the history, physical findings, and morphologic appearance of the rash is reasonable, testing may confirm the diagnosis. The most sensitive and specific tests are polymerase chain reaction sequencing for HSV, direct fluorescent antibody stain, and/or viral culture, while Tzanck smear may show characteristic histologic changes. Treatment is with oral antiviral therapy and treatment of the eczema.
Hand, foot, and mouth disease (HFMD) is a common viral illness usually affecting infants and children. The infection often involves the hands, feet, mouth, and sometimes, the genitals and buttocks. The viral exanthem is most commonly caused by the coxsackievirus, of the enterovirus family. Coxsackievirus A16 and enterovirus A71 are the serotypes that are most commonly implicated as the causative agents. HFMD initially presents with a low-grade fever, reduced appetite, and general malaise. About 1-2 days later, the child may develop painful mouth sores with an exanthem that involves the dorsum of the hands, soles of the feet, buttocks, legs, and arms. The exanthem consists of vesicles surrounded by a thin halo of erythema, eventually rupturing and forming superficial ulcers with a gray-yellow base and erythematous rim. The exanthem is itchy, and can be macular, papular, or vesicular. The lesions are nonpruritic, and typically not painful. The diagnosis of HFMD usually is made clinically, although a physician can swab the mouth or get a stool sample for polymerase chain reaction, which will show the virus; treatment is supportive. In children with atopic dermatitis, lesions also can tend to concentrate in areas previously or currently affected by the dermatitis, similar to eczema herpeticum, and the terms eczema coxsackium or atypical HFMD are applicable. In young adults, the disease may present with erythematous papulovesicular lesions on the face, oral mucosa, extensor surfaces of the upper and lower extremities, and palms and soles; confluent, hemorrhagic, and crusted lesions also can be seen on the extremities. Systemic symptoms usually subside in a few days; the skin lesions resolve without scarring in days to weeks.
Secondary bacterial infection is not uncommon in eczema herpeticum patients, reflecting common Staphylococcus aureus infection in atopic dermatitis patients. Streptococcus also may be seen as a concurrent infection. Treatment of secondary bacterial infection may be considered based on clinic context and culture.
Impetiginized eczema also is in the differential diagnosis of eczema herpeticum. S. aureus and Streptococci are the most important causative organisms. Lesions can manifest as a single red papule or macule that quickly becomes vesicular or eroded. Subsequently, the content dries, forming honey-colored crusts. Impetigo may resolve spontaneously, although in the context of infected eczema both topical anti-inflammatory agents (e.g. topical corticosteroids) along with systemic antibiotics may be a reasonable treatment option. Although our patient had honey-colored crusting, the wound culture showed normal bacterial flora.
Primary varicella infection causes acute fever and rash, with an initial exanthem of disseminated pruritic erythematous macules that progress beyond the papular stage, forming clear, fluid-filled vesicles (like dewdrops on a rose petal). In children, the rash presents on the stomach, back, and face, and then spreads to other parts of the body. Blisters also can arise inside the mouth.
In this patient, perioral HSV PCR 1 was positive, and wound culture showed normal oral flora with no organisms or white blood cells seen. The patient responded well to oral acyclovir, and treatment of his underlying atopic dermatitis with low-potency topical corticosteroids.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither of the physicians had relevant financial disclosures. Email them at [email protected].
Sources
Can Fam Physician. 2012 Dec;58(12):1358-61.
William L Weston, MD., William Howe, MD. UpToDate. Treatment of atopic dermatitis (eczema).
Christine Johnson, MD, Anna Wald, MD, MPH. UpToDate. Epidemiology, clinical manifestations, and diagnosis of herpes simplex virus type 1 infection.
Robert Sidbury, MD, MPH. UpToDate. Atypical exanthems in children.
National Eczema Association. Eczema herpeticum.
Centers for Disease Control and Prevention. Symptoms and diagnosis of hand, foot, and mouth disease (HFMD).
Enlarging scalp plaque
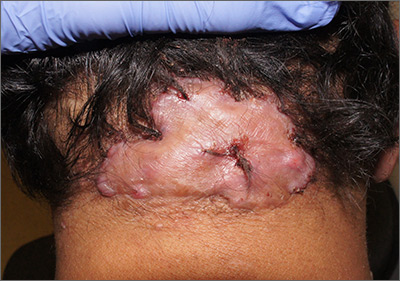
The findings of follicular-based papules, pustules, and scars led to the diagnosis of folliculitis keloidalis nuchae.
Follicular keloidalis, also called acne keloidalis nuchae, is more common in patients with darker skin types (Fitzpatrick skin types IV-VI). The pathogenesis is unclear, but the condition may arise from mechanical occlusion with a retained short hair that leads to follicular destruction. It also may be a primary disorder arising from bacterial infection and subsequent vigorous inflammation.
In the earliest stages, when only small inflammatory papules are present, patients should be instructed not to cut their hair shorter than 0.25-in long to avoid hair retention. Also, topical clindamycin lotion or topical chlorhexidine solution may serve as sufficient treatment. As the disease progresses, oral doxycycline 100 mg bid, combination rifampin 300 mg bid and clindamycin 300 mg bid for 3 months, or isotretinoin 1 mg/kg daily for 6 to 8 months are medical therapeutic options. Procedural options include radiation and laser hair removal.
In this patient, trials of intralesional triamcinolone acetonide 10 mg/mL helped to flatten the plaque, but hair loss persisted. Ultimately, he was referred to Plastic Surgery for excision and was treated for several months with doxycycline 100 mg bid and monitored for recurrence.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Chouk C, Litaiem N, Jones M, et al. Acne keloidalis nuchae: clinical and dermoscopic features. BMJ Case Rep. 2017. pii: bcr-2017-222222. doi: 10.1136/bcr-2017-222222.

The findings of follicular-based papules, pustules, and scars led to the diagnosis of folliculitis keloidalis nuchae.
Follicular keloidalis, also called acne keloidalis nuchae, is more common in patients with darker skin types (Fitzpatrick skin types IV-VI). The pathogenesis is unclear, but the condition may arise from mechanical occlusion with a retained short hair that leads to follicular destruction. It also may be a primary disorder arising from bacterial infection and subsequent vigorous inflammation.
In the earliest stages, when only small inflammatory papules are present, patients should be instructed not to cut their hair shorter than 0.25-in long to avoid hair retention. Also, topical clindamycin lotion or topical chlorhexidine solution may serve as sufficient treatment. As the disease progresses, oral doxycycline 100 mg bid, combination rifampin 300 mg bid and clindamycin 300 mg bid for 3 months, or isotretinoin 1 mg/kg daily for 6 to 8 months are medical therapeutic options. Procedural options include radiation and laser hair removal.
In this patient, trials of intralesional triamcinolone acetonide 10 mg/mL helped to flatten the plaque, but hair loss persisted. Ultimately, he was referred to Plastic Surgery for excision and was treated for several months with doxycycline 100 mg bid and monitored for recurrence.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The findings of follicular-based papules, pustules, and scars led to the diagnosis of folliculitis keloidalis nuchae.
Follicular keloidalis, also called acne keloidalis nuchae, is more common in patients with darker skin types (Fitzpatrick skin types IV-VI). The pathogenesis is unclear, but the condition may arise from mechanical occlusion with a retained short hair that leads to follicular destruction. It also may be a primary disorder arising from bacterial infection and subsequent vigorous inflammation.
In the earliest stages, when only small inflammatory papules are present, patients should be instructed not to cut their hair shorter than 0.25-in long to avoid hair retention. Also, topical clindamycin lotion or topical chlorhexidine solution may serve as sufficient treatment. As the disease progresses, oral doxycycline 100 mg bid, combination rifampin 300 mg bid and clindamycin 300 mg bid for 3 months, or isotretinoin 1 mg/kg daily for 6 to 8 months are medical therapeutic options. Procedural options include radiation and laser hair removal.
In this patient, trials of intralesional triamcinolone acetonide 10 mg/mL helped to flatten the plaque, but hair loss persisted. Ultimately, he was referred to Plastic Surgery for excision and was treated for several months with doxycycline 100 mg bid and monitored for recurrence.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Chouk C, Litaiem N, Jones M, et al. Acne keloidalis nuchae: clinical and dermoscopic features. BMJ Case Rep. 2017. pii: bcr-2017-222222. doi: 10.1136/bcr-2017-222222.
Chouk C, Litaiem N, Jones M, et al. Acne keloidalis nuchae: clinical and dermoscopic features. BMJ Case Rep. 2017. pii: bcr-2017-222222. doi: 10.1136/bcr-2017-222222.