User login
FDA adds safety-related information to its dermal filler webpage
On July 6, the .
Along with a list of common reactions such as bruising, redness, swelling, and pain, the webpage now includes language to inform the public and health care providers about reports of delayed-onset inflammation that have been reported to occur near the dermal filler treatment site following viral or bacterial illnesses or infections, vaccinations, or dental procedures. According to an FDA spokesperson, the update is based on several sources of information, including postmarketing data from adverse event–reporting databases, such as the Manufacturer and User Facility Device Experience (MAUDE) for devices and the Vaccine Adverse Event Reporting System (VAERS) for vaccines, published literature, and recommendations from federal agencies and professional societies.
“More specifically, the site was updated to include certain risks of using dermal fillers such as swelling and bruising as well as some less common risks such as inflammation – swelling or redness near the dermal filler injection site – following viral or bacterial illnesses or infections, vaccinations, or dental procedures,” the spokesperson said.
The announcement about the update also states that “typically, the reported inflammation is responsive to treatment or resolves on its own.”
Other less common risks from dermal filler use listed on the website include bumps in or under the skin (nodules or granulomas) that may need to be treated with injections, oral antibiotics, or surgical removal; infection; open or draining wounds; a sore at the injection site; allergic reactions; or necrosis.
Meanwhile, rare risks from dermal filler use that have been reported to the FDA include severe allergic reactions (anaphylactic shock) that require immediate emergency medical assistance; migration (movement of filler material from the site of injection); leakage or rupture of the filler material at the injection site or through the skin (which may result from a tissue reaction or an infection); the formation of permanent hard nodules; and injury to the blood supply after an unintentional injection into a blood vessel, resulting in necrosis, vision abnormalities (including blindness), or stroke.
Lawrence J. Green, MD, of the department of dermatology at George Washington University, Washington, who was asked to comment about the FDA update on dermal fillers, said that the agency “is doing its job by making consumers aware of all possible complications [common and uncommon], as it does when it creates a package insert for a medication. Fortunately, however, comprehensive reviews published in the peer-reviewed dermatology literature show delayed inflammation to be a very rare event. So, while it is important for dermatologists during informed consent – prior to filler – to discuss that redness and/or nodules after infection/vaccinations, etc. are possible, it is important to add that based on the data, they are also highly unlikely.”
Sue Ellen Cox, MD, a dermatologist who practices in Chapel Hill, N.C., said that she was glad to see separate sections of recommendations geared to patients and health care providers. For example, the website recommends that patients seek a physician in the field of dermatology or plastic surgery to perform procedures that use dermal fillers. “These are not procedures to be done in an unsupervised spa setting,” said Dr. Cox, a past president of the American Society for Dermatologic Surgery and one of the task force authors of recommendations on preventing and treating adverse events of injectable fillers.
“It also makes the point of using products that are acquired from FDA-approved manufacturers, not products sold online or bootlegged from other countries. Finally, it goes into detail about the importance of in-depth knowledge of anatomy, which is crucial for safe injections and reviews potential complications such as intravascular events and hypersensitivity reactions. The administering physician should have extensive knowledge regarding how to treat any potential problems that arise.”
Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies. Dr. Cox disclosed that she has been a clinical investigator for many injectable companies including AbbVie, Galderma, Revance, and Chroma.
Health care professionals, patients, and others can report adverse events related to dermal fillers and other medical devices to the FDA at 800-FDA-1088 or on the MAUDE website.
On July 6, the .
Along with a list of common reactions such as bruising, redness, swelling, and pain, the webpage now includes language to inform the public and health care providers about reports of delayed-onset inflammation that have been reported to occur near the dermal filler treatment site following viral or bacterial illnesses or infections, vaccinations, or dental procedures. According to an FDA spokesperson, the update is based on several sources of information, including postmarketing data from adverse event–reporting databases, such as the Manufacturer and User Facility Device Experience (MAUDE) for devices and the Vaccine Adverse Event Reporting System (VAERS) for vaccines, published literature, and recommendations from federal agencies and professional societies.
“More specifically, the site was updated to include certain risks of using dermal fillers such as swelling and bruising as well as some less common risks such as inflammation – swelling or redness near the dermal filler injection site – following viral or bacterial illnesses or infections, vaccinations, or dental procedures,” the spokesperson said.
The announcement about the update also states that “typically, the reported inflammation is responsive to treatment or resolves on its own.”
Other less common risks from dermal filler use listed on the website include bumps in or under the skin (nodules or granulomas) that may need to be treated with injections, oral antibiotics, or surgical removal; infection; open or draining wounds; a sore at the injection site; allergic reactions; or necrosis.
Meanwhile, rare risks from dermal filler use that have been reported to the FDA include severe allergic reactions (anaphylactic shock) that require immediate emergency medical assistance; migration (movement of filler material from the site of injection); leakage or rupture of the filler material at the injection site or through the skin (which may result from a tissue reaction or an infection); the formation of permanent hard nodules; and injury to the blood supply after an unintentional injection into a blood vessel, resulting in necrosis, vision abnormalities (including blindness), or stroke.
Lawrence J. Green, MD, of the department of dermatology at George Washington University, Washington, who was asked to comment about the FDA update on dermal fillers, said that the agency “is doing its job by making consumers aware of all possible complications [common and uncommon], as it does when it creates a package insert for a medication. Fortunately, however, comprehensive reviews published in the peer-reviewed dermatology literature show delayed inflammation to be a very rare event. So, while it is important for dermatologists during informed consent – prior to filler – to discuss that redness and/or nodules after infection/vaccinations, etc. are possible, it is important to add that based on the data, they are also highly unlikely.”
Sue Ellen Cox, MD, a dermatologist who practices in Chapel Hill, N.C., said that she was glad to see separate sections of recommendations geared to patients and health care providers. For example, the website recommends that patients seek a physician in the field of dermatology or plastic surgery to perform procedures that use dermal fillers. “These are not procedures to be done in an unsupervised spa setting,” said Dr. Cox, a past president of the American Society for Dermatologic Surgery and one of the task force authors of recommendations on preventing and treating adverse events of injectable fillers.
“It also makes the point of using products that are acquired from FDA-approved manufacturers, not products sold online or bootlegged from other countries. Finally, it goes into detail about the importance of in-depth knowledge of anatomy, which is crucial for safe injections and reviews potential complications such as intravascular events and hypersensitivity reactions. The administering physician should have extensive knowledge regarding how to treat any potential problems that arise.”
Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies. Dr. Cox disclosed that she has been a clinical investigator for many injectable companies including AbbVie, Galderma, Revance, and Chroma.
Health care professionals, patients, and others can report adverse events related to dermal fillers and other medical devices to the FDA at 800-FDA-1088 or on the MAUDE website.
On July 6, the .
Along with a list of common reactions such as bruising, redness, swelling, and pain, the webpage now includes language to inform the public and health care providers about reports of delayed-onset inflammation that have been reported to occur near the dermal filler treatment site following viral or bacterial illnesses or infections, vaccinations, or dental procedures. According to an FDA spokesperson, the update is based on several sources of information, including postmarketing data from adverse event–reporting databases, such as the Manufacturer and User Facility Device Experience (MAUDE) for devices and the Vaccine Adverse Event Reporting System (VAERS) for vaccines, published literature, and recommendations from federal agencies and professional societies.
“More specifically, the site was updated to include certain risks of using dermal fillers such as swelling and bruising as well as some less common risks such as inflammation – swelling or redness near the dermal filler injection site – following viral or bacterial illnesses or infections, vaccinations, or dental procedures,” the spokesperson said.
The announcement about the update also states that “typically, the reported inflammation is responsive to treatment or resolves on its own.”
Other less common risks from dermal filler use listed on the website include bumps in or under the skin (nodules or granulomas) that may need to be treated with injections, oral antibiotics, or surgical removal; infection; open or draining wounds; a sore at the injection site; allergic reactions; or necrosis.
Meanwhile, rare risks from dermal filler use that have been reported to the FDA include severe allergic reactions (anaphylactic shock) that require immediate emergency medical assistance; migration (movement of filler material from the site of injection); leakage or rupture of the filler material at the injection site or through the skin (which may result from a tissue reaction or an infection); the formation of permanent hard nodules; and injury to the blood supply after an unintentional injection into a blood vessel, resulting in necrosis, vision abnormalities (including blindness), or stroke.
Lawrence J. Green, MD, of the department of dermatology at George Washington University, Washington, who was asked to comment about the FDA update on dermal fillers, said that the agency “is doing its job by making consumers aware of all possible complications [common and uncommon], as it does when it creates a package insert for a medication. Fortunately, however, comprehensive reviews published in the peer-reviewed dermatology literature show delayed inflammation to be a very rare event. So, while it is important for dermatologists during informed consent – prior to filler – to discuss that redness and/or nodules after infection/vaccinations, etc. are possible, it is important to add that based on the data, they are also highly unlikely.”
Sue Ellen Cox, MD, a dermatologist who practices in Chapel Hill, N.C., said that she was glad to see separate sections of recommendations geared to patients and health care providers. For example, the website recommends that patients seek a physician in the field of dermatology or plastic surgery to perform procedures that use dermal fillers. “These are not procedures to be done in an unsupervised spa setting,” said Dr. Cox, a past president of the American Society for Dermatologic Surgery and one of the task force authors of recommendations on preventing and treating adverse events of injectable fillers.
“It also makes the point of using products that are acquired from FDA-approved manufacturers, not products sold online or bootlegged from other countries. Finally, it goes into detail about the importance of in-depth knowledge of anatomy, which is crucial for safe injections and reviews potential complications such as intravascular events and hypersensitivity reactions. The administering physician should have extensive knowledge regarding how to treat any potential problems that arise.”
Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies. Dr. Cox disclosed that she has been a clinical investigator for many injectable companies including AbbVie, Galderma, Revance, and Chroma.
Health care professionals, patients, and others can report adverse events related to dermal fillers and other medical devices to the FDA at 800-FDA-1088 or on the MAUDE website.
Necessary Updates to Skin Cancer Risk Stratification
1. Powers JG, Patel NA, Powers EA, Mayer JE, Stricklin GP, Geller AC. Skin cancer
risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
2. Balci S, Ayaz L, Gorur A, Yildirim Yaroglu H, Akbayir S, Dogruer Unal N, Bulut B,
Tursen U, Tamer L. microRNA profiling for early detection of nonmelanoma skin cancer. Clin Exp Dermatol. 2016;41(4):346-51. doi:10.1111/ced.12736
3. Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7-33. doi:10.3322/caac.21708
4. Agbai ON, Buster K, Sanchez M, Hernandez C, Kundu RV, Chiu M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70(4):748-62.
5. Chou SE, Gaysynsky A, Trivedi N, Vanderpool R. Using social media for health: national data from HINTS 2019. Journ of Health Comm. 2019;26(3):184-193. doi:10.1080/10810730.2021.
6. Stern RS. Prevalence of a history of skin cancer in 2007: results of an incidence-based model. Arch Dermatol. 2010;146(3):279-82.
7. Dennis LK, et al. Sunburns and risk of cutaneous melanoma: does age matter? A comprehensive meta-analysis. Annals of Epidem. 2008;18(8):614-627. doi:10.1016/j.annepidem.2008.
8. Wu S, Han J, Laden F, Qureshi AA. Long-term ultraviolet flux, other potential risk factors, and skin cancer risk: a cohort study. Cancer Epidemiol Biomar Prev. 2014;23(6):1080-1089.
9. 2020 Demographics Profile of the military community. US Department of Defense. 2020:iv. Accessed November 15, 2022. 2020 Demographics Profile of the Military Community (militaryonesource.mil)
10. Apalla Z, Lallas A, Sotiriou E, Lazaridou E, Ioannides D. Epidemiological trends in skin cancer. Dermatol Pract Concept. 2017;7:1-6.
11. Basch CH, Hillyer GC. Skin cancer on Instagram: implications for adolescents and young adults. Int J Adolesc Med Health. 2022;34(3). doi:10.1515/ijamh-2019-0218
1. Powers JG, Patel NA, Powers EA, Mayer JE, Stricklin GP, Geller AC. Skin cancer
risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
2. Balci S, Ayaz L, Gorur A, Yildirim Yaroglu H, Akbayir S, Dogruer Unal N, Bulut B,
Tursen U, Tamer L. microRNA profiling for early detection of nonmelanoma skin cancer. Clin Exp Dermatol. 2016;41(4):346-51. doi:10.1111/ced.12736
3. Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7-33. doi:10.3322/caac.21708
4. Agbai ON, Buster K, Sanchez M, Hernandez C, Kundu RV, Chiu M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70(4):748-62.
5. Chou SE, Gaysynsky A, Trivedi N, Vanderpool R. Using social media for health: national data from HINTS 2019. Journ of Health Comm. 2019;26(3):184-193. doi:10.1080/10810730.2021.
6. Stern RS. Prevalence of a history of skin cancer in 2007: results of an incidence-based model. Arch Dermatol. 2010;146(3):279-82.
7. Dennis LK, et al. Sunburns and risk of cutaneous melanoma: does age matter? A comprehensive meta-analysis. Annals of Epidem. 2008;18(8):614-627. doi:10.1016/j.annepidem.2008.
8. Wu S, Han J, Laden F, Qureshi AA. Long-term ultraviolet flux, other potential risk factors, and skin cancer risk: a cohort study. Cancer Epidemiol Biomar Prev. 2014;23(6):1080-1089.
9. 2020 Demographics Profile of the military community. US Department of Defense. 2020:iv. Accessed November 15, 2022. 2020 Demographics Profile of the Military Community (militaryonesource.mil)
10. Apalla Z, Lallas A, Sotiriou E, Lazaridou E, Ioannides D. Epidemiological trends in skin cancer. Dermatol Pract Concept. 2017;7:1-6.
11. Basch CH, Hillyer GC. Skin cancer on Instagram: implications for adolescents and young adults. Int J Adolesc Med Health. 2022;34(3). doi:10.1515/ijamh-2019-0218
1. Powers JG, Patel NA, Powers EA, Mayer JE, Stricklin GP, Geller AC. Skin cancer
risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
2. Balci S, Ayaz L, Gorur A, Yildirim Yaroglu H, Akbayir S, Dogruer Unal N, Bulut B,
Tursen U, Tamer L. microRNA profiling for early detection of nonmelanoma skin cancer. Clin Exp Dermatol. 2016;41(4):346-51. doi:10.1111/ced.12736
3. Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7-33. doi:10.3322/caac.21708
4. Agbai ON, Buster K, Sanchez M, Hernandez C, Kundu RV, Chiu M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70(4):748-62.
5. Chou SE, Gaysynsky A, Trivedi N, Vanderpool R. Using social media for health: national data from HINTS 2019. Journ of Health Comm. 2019;26(3):184-193. doi:10.1080/10810730.2021.
6. Stern RS. Prevalence of a history of skin cancer in 2007: results of an incidence-based model. Arch Dermatol. 2010;146(3):279-82.
7. Dennis LK, et al. Sunburns and risk of cutaneous melanoma: does age matter? A comprehensive meta-analysis. Annals of Epidem. 2008;18(8):614-627. doi:10.1016/j.annepidem.2008.
8. Wu S, Han J, Laden F, Qureshi AA. Long-term ultraviolet flux, other potential risk factors, and skin cancer risk: a cohort study. Cancer Epidemiol Biomar Prev. 2014;23(6):1080-1089.
9. 2020 Demographics Profile of the military community. US Department of Defense. 2020:iv. Accessed November 15, 2022. 2020 Demographics Profile of the Military Community (militaryonesource.mil)
10. Apalla Z, Lallas A, Sotiriou E, Lazaridou E, Ioannides D. Epidemiological trends in skin cancer. Dermatol Pract Concept. 2017;7:1-6.
11. Basch CH, Hillyer GC. Skin cancer on Instagram: implications for adolescents and young adults. Int J Adolesc Med Health. 2022;34(3). doi:10.1515/ijamh-2019-0218
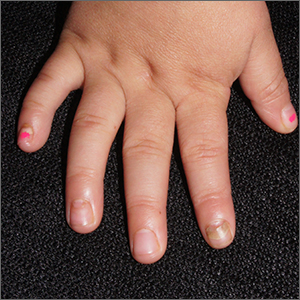
Nails falling off in a 3-year-old
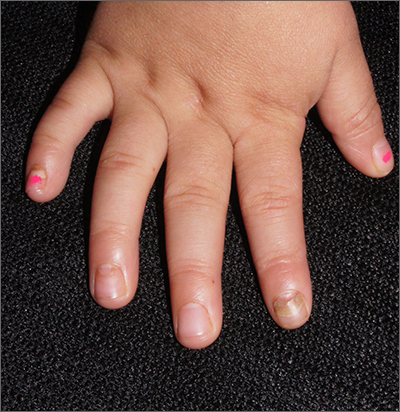
When the nails peel off from the proximal nail folds, the clinical term is onychomadesis and it is important to ask about recent infections or severe metabolic stressors. In children and adults, onychomadesis on multiple fingers may occur after infections and has been associated with hand-foot-mouth disease caused by common viral infections—especially strains of coxsackievirus.1
Because shed nails show evidence of viral infection, one hypothesis for their peeling off is that the tissue of the nail matrix is infected, leading to metabolic changes. As the nail matrix returns to normal function, a new nail is made and ultimately will replace the nail that has come off. In healthy US adults, fingernails grow 3.47 mm per month on average while toenails grow 1.62 mm per month on average.2
Sometimes it’s hard to elicit a history of a very mild viral illness weeks or months after it has resolved. Asking specifically about mouth ulcers may help. If there is a history of a viral illness, no specific work-up or treatment is necessary. Patients may be reassured that nails will improve over several months without lasting effects.
In this case, the patient and her family were given reassurance and the nails returned to normal within a few months.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Kim EJ, Park HS, Yoon HS, et al. Four cases of onychomadesis after hand-foot-mouth disease. Ann Dermatol. 2014;26:777-778. doi: 10.5021/ad.2014.26.6.777
2. Yaemsiri S, Hou N, Slining MM, et al. Growth rate of human fingernails and toenails in healthy American young adults. J Eur Acad Dermatol Venereol. 2010;24:420-423. doi: 10.1111/j.1468-3083.2009.03426.x

When the nails peel off from the proximal nail folds, the clinical term is onychomadesis and it is important to ask about recent infections or severe metabolic stressors. In children and adults, onychomadesis on multiple fingers may occur after infections and has been associated with hand-foot-mouth disease caused by common viral infections—especially strains of coxsackievirus.1
Because shed nails show evidence of viral infection, one hypothesis for their peeling off is that the tissue of the nail matrix is infected, leading to metabolic changes. As the nail matrix returns to normal function, a new nail is made and ultimately will replace the nail that has come off. In healthy US adults, fingernails grow 3.47 mm per month on average while toenails grow 1.62 mm per month on average.2
Sometimes it’s hard to elicit a history of a very mild viral illness weeks or months after it has resolved. Asking specifically about mouth ulcers may help. If there is a history of a viral illness, no specific work-up or treatment is necessary. Patients may be reassured that nails will improve over several months without lasting effects.
In this case, the patient and her family were given reassurance and the nails returned to normal within a few months.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

When the nails peel off from the proximal nail folds, the clinical term is onychomadesis and it is important to ask about recent infections or severe metabolic stressors. In children and adults, onychomadesis on multiple fingers may occur after infections and has been associated with hand-foot-mouth disease caused by common viral infections—especially strains of coxsackievirus.1
Because shed nails show evidence of viral infection, one hypothesis for their peeling off is that the tissue of the nail matrix is infected, leading to metabolic changes. As the nail matrix returns to normal function, a new nail is made and ultimately will replace the nail that has come off. In healthy US adults, fingernails grow 3.47 mm per month on average while toenails grow 1.62 mm per month on average.2
Sometimes it’s hard to elicit a history of a very mild viral illness weeks or months after it has resolved. Asking specifically about mouth ulcers may help. If there is a history of a viral illness, no specific work-up or treatment is necessary. Patients may be reassured that nails will improve over several months without lasting effects.
In this case, the patient and her family were given reassurance and the nails returned to normal within a few months.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Kim EJ, Park HS, Yoon HS, et al. Four cases of onychomadesis after hand-foot-mouth disease. Ann Dermatol. 2014;26:777-778. doi: 10.5021/ad.2014.26.6.777
2. Yaemsiri S, Hou N, Slining MM, et al. Growth rate of human fingernails and toenails in healthy American young adults. J Eur Acad Dermatol Venereol. 2010;24:420-423. doi: 10.1111/j.1468-3083.2009.03426.x
1. Kim EJ, Park HS, Yoon HS, et al. Four cases of onychomadesis after hand-foot-mouth disease. Ann Dermatol. 2014;26:777-778. doi: 10.5021/ad.2014.26.6.777
2. Yaemsiri S, Hou N, Slining MM, et al. Growth rate of human fingernails and toenails in healthy American young adults. J Eur Acad Dermatol Venereol. 2010;24:420-423. doi: 10.1111/j.1468-3083.2009.03426.x
New guidelines for MTX use in pediatric inflammatory skin disease unveiled
While the typical dose of methotrexate (MTX) for inflammatory disease in pediatric patients varies in published studies, the maximum dose is considered to be 1 mg/kg and not to exceed 25 mg/week. In addition, test doses are not necessary for pediatric patients starting low dose (1 mg/kg or less) MTX for inflammatory skin disease, and the onset of efficacy with MTX may take 8-16 weeks.
and published online in Pediatric Dermatology.
“Methotrexate is a cost-effective, readily accessible, well-tolerated, useful, and time-honored option for children with a spectrum of inflammatory skin diseases,” project cochair Elaine C. Siegfried, MD, professor of pediatrics and dermatology at Saint Louis University, told this news organization. “Although considered an ‘immune suppressant’ by some, it is more accurately classified as an immune modulator and has been widely used for more than 50 years, and remains the standard of care when administered at very high doses and intrathecally in children with acute lymphoblastic leukemia – a practice that supports safety. But many details that support optimized treatment are not widely appreciated.”
In their guidelines document, Dr. Siegfried and her 22 coauthors noted that Food and Drug Administration labeling does not include approved indications for the use of MTX for many inflammatory skin diseases in pediatric patients, including morphea, psoriasis, atopic dermatitis, and alopecia areata. “Furthermore, some clinicians may be unfamiliar or uncomfortable prescribing medications off label for pediatric patients, causing delayed initiation, premature drug discontinuation, or use of less advantageous alternatives,” they wrote.
To address this unmet need, Dr. Siegfried and the other committee members used a modified Delphi process to reach agreement on recommendations related to five key topic areas: indications and contraindications, dosing, interactions with immunizations and medications, potential for and management of adverse effects, and monitoring needs. Consensus was predefined as at least 70% of participants rating a statement as 7-9 on the Likert scale. The effort to develop 46 recommendations has been a work in progress for almost 5 years, “somewhat delayed by the pandemic,” Dr. Siegfried, past president and director of the American Board of Dermatology, said in an interview. “But it remains relevant, despite the emergence of biologics and JAK inhibitors for treating inflammatory skin conditions in children. Although the mechanism-of-action of low-dose MTX is not clear, it may overlap with the newer small molecules.”
The guidelines contain several pearls to guide optimal dosing, including the following key points:
- MTX can be discontinued abruptly without adverse effects, other than the risk of disease worsening.
- Folic acid supplementation (starting at 1 mg/day, regardless of weight) is an effective approach to minimizing associated gastrointestinal adverse effects.
- Concomitant use of MTX and antibiotics (including trimethoprim-sulfamethoxazole) and NSAIDS are not contraindicated for most pediatric patients treated for inflammatory skin disease.
- Live virus vaccine boosters such as varicella-zoster virus (VZV) and measles, mumps, and rubella (MMR) are not contraindicated in patients taking MTX; there are insufficient data to make recommendations for or against primary immunization with MMR vaccine in patients taking MTX; inactivated vaccines should be given to patients taking MTX.
- Routine surveillance laboratory monitoring (i.e., CBC with differential, alanine transaminase, aspartate aminotransferase, creatinine) is recommended at baseline, after 1 month of treatment, and every 3-4 months thereafter.
- Transient transaminase elevation (≤ 3 upper limit normal for < 3 months) is not uncommon with low-dose MTX and does not usually require interruption of MTX. The most likely causes are concomitant viral infection, MTX dosing within 24 hours prior to phlebotomy, recent administration of other medications (such as acetaminophen), and/or recent alcohol consumption.
- Liver biopsy is not indicated for routine monitoring of pediatric patients taking low-dose MTX.
According to Dr. Siegfried, consensus of the committee members was lowest on the need for a test dose of MTX.
Overall, she said in the interview, helping to craft the guidelines caused her to reflect on how her approach to using MTX has evolved over the past 35 years, after treating “many hundreds” of patients. “I was gratified to confirm similar practice patterns among my colleagues,” she added.
The project’s other cochair was Heather Brandling-Bennett, MD, a dermatologist at Seattle Children’s Hospital. This work was supported by a grant from the Pediatric Dermatology Research Alliance (PeDRA), with additional funding from the National Eczema Association and the National Psoriasis Foundation. Dr. Siegfried disclosed ties with AbbVie, Boehringer Ingelheim, Incyte, LEO Pharma, Novan, Novartis, Pierre Fabre, Pfizer, Regeneron, Sanofi Genzyme, UCB, and Verrica. She has participated in contracted research for AI Therapeutics, and has served as principal investigator for Janssen. Many of the guideline coauthors disclosed having received grant support and other funding from pharmaceutical companies.
While the typical dose of methotrexate (MTX) for inflammatory disease in pediatric patients varies in published studies, the maximum dose is considered to be 1 mg/kg and not to exceed 25 mg/week. In addition, test doses are not necessary for pediatric patients starting low dose (1 mg/kg or less) MTX for inflammatory skin disease, and the onset of efficacy with MTX may take 8-16 weeks.
and published online in Pediatric Dermatology.
“Methotrexate is a cost-effective, readily accessible, well-tolerated, useful, and time-honored option for children with a spectrum of inflammatory skin diseases,” project cochair Elaine C. Siegfried, MD, professor of pediatrics and dermatology at Saint Louis University, told this news organization. “Although considered an ‘immune suppressant’ by some, it is more accurately classified as an immune modulator and has been widely used for more than 50 years, and remains the standard of care when administered at very high doses and intrathecally in children with acute lymphoblastic leukemia – a practice that supports safety. But many details that support optimized treatment are not widely appreciated.”
In their guidelines document, Dr. Siegfried and her 22 coauthors noted that Food and Drug Administration labeling does not include approved indications for the use of MTX for many inflammatory skin diseases in pediatric patients, including morphea, psoriasis, atopic dermatitis, and alopecia areata. “Furthermore, some clinicians may be unfamiliar or uncomfortable prescribing medications off label for pediatric patients, causing delayed initiation, premature drug discontinuation, or use of less advantageous alternatives,” they wrote.
To address this unmet need, Dr. Siegfried and the other committee members used a modified Delphi process to reach agreement on recommendations related to five key topic areas: indications and contraindications, dosing, interactions with immunizations and medications, potential for and management of adverse effects, and monitoring needs. Consensus was predefined as at least 70% of participants rating a statement as 7-9 on the Likert scale. The effort to develop 46 recommendations has been a work in progress for almost 5 years, “somewhat delayed by the pandemic,” Dr. Siegfried, past president and director of the American Board of Dermatology, said in an interview. “But it remains relevant, despite the emergence of biologics and JAK inhibitors for treating inflammatory skin conditions in children. Although the mechanism-of-action of low-dose MTX is not clear, it may overlap with the newer small molecules.”
The guidelines contain several pearls to guide optimal dosing, including the following key points:
- MTX can be discontinued abruptly without adverse effects, other than the risk of disease worsening.
- Folic acid supplementation (starting at 1 mg/day, regardless of weight) is an effective approach to minimizing associated gastrointestinal adverse effects.
- Concomitant use of MTX and antibiotics (including trimethoprim-sulfamethoxazole) and NSAIDS are not contraindicated for most pediatric patients treated for inflammatory skin disease.
- Live virus vaccine boosters such as varicella-zoster virus (VZV) and measles, mumps, and rubella (MMR) are not contraindicated in patients taking MTX; there are insufficient data to make recommendations for or against primary immunization with MMR vaccine in patients taking MTX; inactivated vaccines should be given to patients taking MTX.
- Routine surveillance laboratory monitoring (i.e., CBC with differential, alanine transaminase, aspartate aminotransferase, creatinine) is recommended at baseline, after 1 month of treatment, and every 3-4 months thereafter.
- Transient transaminase elevation (≤ 3 upper limit normal for < 3 months) is not uncommon with low-dose MTX and does not usually require interruption of MTX. The most likely causes are concomitant viral infection, MTX dosing within 24 hours prior to phlebotomy, recent administration of other medications (such as acetaminophen), and/or recent alcohol consumption.
- Liver biopsy is not indicated for routine monitoring of pediatric patients taking low-dose MTX.
According to Dr. Siegfried, consensus of the committee members was lowest on the need for a test dose of MTX.
Overall, she said in the interview, helping to craft the guidelines caused her to reflect on how her approach to using MTX has evolved over the past 35 years, after treating “many hundreds” of patients. “I was gratified to confirm similar practice patterns among my colleagues,” she added.
The project’s other cochair was Heather Brandling-Bennett, MD, a dermatologist at Seattle Children’s Hospital. This work was supported by a grant from the Pediatric Dermatology Research Alliance (PeDRA), with additional funding from the National Eczema Association and the National Psoriasis Foundation. Dr. Siegfried disclosed ties with AbbVie, Boehringer Ingelheim, Incyte, LEO Pharma, Novan, Novartis, Pierre Fabre, Pfizer, Regeneron, Sanofi Genzyme, UCB, and Verrica. She has participated in contracted research for AI Therapeutics, and has served as principal investigator for Janssen. Many of the guideline coauthors disclosed having received grant support and other funding from pharmaceutical companies.
While the typical dose of methotrexate (MTX) for inflammatory disease in pediatric patients varies in published studies, the maximum dose is considered to be 1 mg/kg and not to exceed 25 mg/week. In addition, test doses are not necessary for pediatric patients starting low dose (1 mg/kg or less) MTX for inflammatory skin disease, and the onset of efficacy with MTX may take 8-16 weeks.
and published online in Pediatric Dermatology.
“Methotrexate is a cost-effective, readily accessible, well-tolerated, useful, and time-honored option for children with a spectrum of inflammatory skin diseases,” project cochair Elaine C. Siegfried, MD, professor of pediatrics and dermatology at Saint Louis University, told this news organization. “Although considered an ‘immune suppressant’ by some, it is more accurately classified as an immune modulator and has been widely used for more than 50 years, and remains the standard of care when administered at very high doses and intrathecally in children with acute lymphoblastic leukemia – a practice that supports safety. But many details that support optimized treatment are not widely appreciated.”
In their guidelines document, Dr. Siegfried and her 22 coauthors noted that Food and Drug Administration labeling does not include approved indications for the use of MTX for many inflammatory skin diseases in pediatric patients, including morphea, psoriasis, atopic dermatitis, and alopecia areata. “Furthermore, some clinicians may be unfamiliar or uncomfortable prescribing medications off label for pediatric patients, causing delayed initiation, premature drug discontinuation, or use of less advantageous alternatives,” they wrote.
To address this unmet need, Dr. Siegfried and the other committee members used a modified Delphi process to reach agreement on recommendations related to five key topic areas: indications and contraindications, dosing, interactions with immunizations and medications, potential for and management of adverse effects, and monitoring needs. Consensus was predefined as at least 70% of participants rating a statement as 7-9 on the Likert scale. The effort to develop 46 recommendations has been a work in progress for almost 5 years, “somewhat delayed by the pandemic,” Dr. Siegfried, past president and director of the American Board of Dermatology, said in an interview. “But it remains relevant, despite the emergence of biologics and JAK inhibitors for treating inflammatory skin conditions in children. Although the mechanism-of-action of low-dose MTX is not clear, it may overlap with the newer small molecules.”
The guidelines contain several pearls to guide optimal dosing, including the following key points:
- MTX can be discontinued abruptly without adverse effects, other than the risk of disease worsening.
- Folic acid supplementation (starting at 1 mg/day, regardless of weight) is an effective approach to minimizing associated gastrointestinal adverse effects.
- Concomitant use of MTX and antibiotics (including trimethoprim-sulfamethoxazole) and NSAIDS are not contraindicated for most pediatric patients treated for inflammatory skin disease.
- Live virus vaccine boosters such as varicella-zoster virus (VZV) and measles, mumps, and rubella (MMR) are not contraindicated in patients taking MTX; there are insufficient data to make recommendations for or against primary immunization with MMR vaccine in patients taking MTX; inactivated vaccines should be given to patients taking MTX.
- Routine surveillance laboratory monitoring (i.e., CBC with differential, alanine transaminase, aspartate aminotransferase, creatinine) is recommended at baseline, after 1 month of treatment, and every 3-4 months thereafter.
- Transient transaminase elevation (≤ 3 upper limit normal for < 3 months) is not uncommon with low-dose MTX and does not usually require interruption of MTX. The most likely causes are concomitant viral infection, MTX dosing within 24 hours prior to phlebotomy, recent administration of other medications (such as acetaminophen), and/or recent alcohol consumption.
- Liver biopsy is not indicated for routine monitoring of pediatric patients taking low-dose MTX.
According to Dr. Siegfried, consensus of the committee members was lowest on the need for a test dose of MTX.
Overall, she said in the interview, helping to craft the guidelines caused her to reflect on how her approach to using MTX has evolved over the past 35 years, after treating “many hundreds” of patients. “I was gratified to confirm similar practice patterns among my colleagues,” she added.
The project’s other cochair was Heather Brandling-Bennett, MD, a dermatologist at Seattle Children’s Hospital. This work was supported by a grant from the Pediatric Dermatology Research Alliance (PeDRA), with additional funding from the National Eczema Association and the National Psoriasis Foundation. Dr. Siegfried disclosed ties with AbbVie, Boehringer Ingelheim, Incyte, LEO Pharma, Novan, Novartis, Pierre Fabre, Pfizer, Regeneron, Sanofi Genzyme, UCB, and Verrica. She has participated in contracted research for AI Therapeutics, and has served as principal investigator for Janssen. Many of the guideline coauthors disclosed having received grant support and other funding from pharmaceutical companies.
FROM PEDIATRIC DERMATOLOGY
Hairy moles may contain the cure for baldness: Study
.
The researchers found that a specific molecule in those hairy moles “causes normally dormant and diminutive hair follicles to activate their stem cells for robust growth of long and thick hairs,” lead researcher Maksim Plikus, PhD, professor of developmental and cell biology at the University of California, Irvine, said in a statement.
The findings could lead to new treatments for the hair loss condition known as androgenetic alopecia, which researchers said occurs in both men and women. It is also known as male-pattern baldness in men.
The global team led by researchers at the university analyzed hair follicle stem cells and discovered that a molecule called osteopontin drives accelerated hair growth. Stem cells can develop into different kinds of cells, whether they are in the body or in a laboratory, and are often involved in regenerative or repair processes, according to the Mayo Clinic.
This latest study, published in the journal Nature, was done on mice. A drug company cofounded by Dr. Plikus said in a news release that it had further tested the hair growth technique on human hair follicles, and “the researchers were able to induce new growth by human hair follicles in a robust preclinical model.” The company, Amplifica, said in the release that it has an exclusive licensing agreement with the university for the new hair growth “inventions” described in the newly published findings.
Hair loss from androgenetic alopecia occurs in two out of every three men, according to the Cleveland Clinic. Amplifica said the condition affects an estimated 50 million men and 30 million women in the United States.
The hair loss and thinning can begin as early as the late teens, the Cleveland Clinic says. The condition is progressive and can follow a specific pattern, such as the hairline creating an “M” or “U” shape midway through the process toward complete baldness on the top of the head, with a remaining thin band of hair around the sides of the head.
A version of this article first appeared on WebMD.com.
.
The researchers found that a specific molecule in those hairy moles “causes normally dormant and diminutive hair follicles to activate their stem cells for robust growth of long and thick hairs,” lead researcher Maksim Plikus, PhD, professor of developmental and cell biology at the University of California, Irvine, said in a statement.
The findings could lead to new treatments for the hair loss condition known as androgenetic alopecia, which researchers said occurs in both men and women. It is also known as male-pattern baldness in men.
The global team led by researchers at the university analyzed hair follicle stem cells and discovered that a molecule called osteopontin drives accelerated hair growth. Stem cells can develop into different kinds of cells, whether they are in the body or in a laboratory, and are often involved in regenerative or repair processes, according to the Mayo Clinic.
This latest study, published in the journal Nature, was done on mice. A drug company cofounded by Dr. Plikus said in a news release that it had further tested the hair growth technique on human hair follicles, and “the researchers were able to induce new growth by human hair follicles in a robust preclinical model.” The company, Amplifica, said in the release that it has an exclusive licensing agreement with the university for the new hair growth “inventions” described in the newly published findings.
Hair loss from androgenetic alopecia occurs in two out of every three men, according to the Cleveland Clinic. Amplifica said the condition affects an estimated 50 million men and 30 million women in the United States.
The hair loss and thinning can begin as early as the late teens, the Cleveland Clinic says. The condition is progressive and can follow a specific pattern, such as the hairline creating an “M” or “U” shape midway through the process toward complete baldness on the top of the head, with a remaining thin band of hair around the sides of the head.
A version of this article first appeared on WebMD.com.
.
The researchers found that a specific molecule in those hairy moles “causes normally dormant and diminutive hair follicles to activate their stem cells for robust growth of long and thick hairs,” lead researcher Maksim Plikus, PhD, professor of developmental and cell biology at the University of California, Irvine, said in a statement.
The findings could lead to new treatments for the hair loss condition known as androgenetic alopecia, which researchers said occurs in both men and women. It is also known as male-pattern baldness in men.
The global team led by researchers at the university analyzed hair follicle stem cells and discovered that a molecule called osteopontin drives accelerated hair growth. Stem cells can develop into different kinds of cells, whether they are in the body or in a laboratory, and are often involved in regenerative or repair processes, according to the Mayo Clinic.
This latest study, published in the journal Nature, was done on mice. A drug company cofounded by Dr. Plikus said in a news release that it had further tested the hair growth technique on human hair follicles, and “the researchers were able to induce new growth by human hair follicles in a robust preclinical model.” The company, Amplifica, said in the release that it has an exclusive licensing agreement with the university for the new hair growth “inventions” described in the newly published findings.
Hair loss from androgenetic alopecia occurs in two out of every three men, according to the Cleveland Clinic. Amplifica said the condition affects an estimated 50 million men and 30 million women in the United States.
The hair loss and thinning can begin as early as the late teens, the Cleveland Clinic says. The condition is progressive and can follow a specific pattern, such as the hairline creating an “M” or “U” shape midway through the process toward complete baldness on the top of the head, with a remaining thin band of hair around the sides of the head.
A version of this article first appeared on WebMD.com.
FROM NATURE
For psoriasis, review finds several biosimilars as safe and effective as biologics
The effectiveness and safety of biosimilars for psoriasis appear to be similar to the originator biologics, reported the authors of a review of studies comparing the two.
“This systematic review found that there was no clinically or statistically significant difference in the efficacy and safety between biosimilars and originators of adalimumab, etanercept, infliximab, and ustekinumab for the treatment of psoriasis,” senior study author and clinical lecturer Zenas Z. N. Yiu, MBChB, PhD, and his colleagues at the University of Manchester, England, wrote in JAMA Dermatology.“The biosimilars evaluated in this study could be considered alongside originators for biologic-naive patients to improve the accessibility of biological treatments,” they added. “Switching patients currently on originators to biosimilars could be considered where clinically appropriate to reduce treatment costs.”
Biologics versus biosimilars
In contrast to most chemically synthesized drugs, biologics are created from living organisms, and they have complex structures that can vary slightly from batch to batch, Luigi Naldi, MD, director of the department of dermatology of Ospedale San Bortolo, Vicenza, Italy, and Antonio Addis, PharmD, researcher in the department of epidemiology, Regione Lazio, in Rome, wrote in an accompanying editorial.
Once the patent on the “originator” biologic expires, U.S. and European regulators allow other manufacturers to develop similar molecules – biosimilars – through an abbreviated approval process. If the results of a limited number of equivalence or noninferiority clinical trials are acceptable, registration for all the indications of the originator is allowed for its biosimilars. Referring to the expense of biologics, Dr. Naldi and Dr. Addis noted that in the United States, “biologics comprise less than 3% of the volume of drugs on the market, but account for more than one-third of all drug spending.”
Systematic review
Dr. Yiu and his colleagues queried standard medical research databases in August 2022, and included 14 randomized clinical trials (10 adalimumab, 2 etanercept, 1 infliximab, and 1 ustekinumab) and 3 cohort studies (1 adalimumab, 1 etanercept, 1 infliximab and etanercept) in their review.
Twelve trials compared biosimilars vs. originators in originator-naive patients, and 11 trials compared switching from originators to biosimilars vs. continuous treatment with the originator.
The researchers found the following:
At week 16, mean PASI75 (Psoriasis Area and Severity Index) response rates ranges from 60.7% to 90.6% for adalimumab biosimilars, vs. 61.5% to 91.7% for the originator. Mean PASI75 responses for the two etanercept biosimilars were 56.1% and 76.7% vs. 55.5% and 73.4% for the originator. In the ustekinumab study, mean PASI75 responses were 86.1% for the biosimilar vs. 84.0% for the originator.
At week 52, mean PASI75 responses were between 86.3% and 92.8% for adalimumab biosimilars vs. 84.9% and 93.9% for the originator. In the one comparison of an etanercept biosimilar, mean PAS175 responses were 80.9% for the biosimilar vs. 82.9% for the originator.
In studies involving patients switching from the originator to a biosimilar vs. continuing treatment with the originator, 32-week response rates ranged from 87.0% to 91.3% for adalimumab biosimilars and from 88.2% to 93.2% for the originator. In the one ustekinumab study, the 32-week mean PASI75 response was 92.6% after switching from the originator to a biosimilar vs. 92.9% with continuous treatment with the originator.
At week 52, mean PASI75 responses to adalimumab were between 84.2% and 94.8% for patients who switched to biosimilars and between 88.1% and 93.9% for those who stayed on the originator.
At week 52, in all the randomized trials, the incidence of adverse events and serious adverse events among those who switched to the biosimilar and those who continued with the originator were similar. Two cohort studies showed similar safety outcomes between originators and biosimilars, but one reported more adverse events in patients who switched to adalimumab biosimilars (P = .04).
Three clinical trials showed low risk for bias, 11 had moderate risk, and all cohort studies had moderate to high risk for bias.
Experts weigh in
Asked to comment on the study, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, Winston-Salem, N.C., told this news organization that he expects that the results will affect patient care.
However, he added, “I believe the decision of whether to use a biosimilar instead of the originator biologic may be more in the hands of the insurers than in the hands of physicians and patients.
“Biologics for psoriasis are so complicated that even the originator products vary from batch to batch. A biosimilar is basically like another batch of the innovative product,” explained Dr. Feldman, who was not involved in the study. “If we’re comfortable with patients being on different batches of the innovator product, we probably should be comfortable with them being on a biosimilar, as we have more evidence for the similarity of the biosimilar than we do for the current batch of the originator product.”
Aída Lugo-Somolinos, MD, professor of dermatology and director of the Contact Dermatitis Clinic at the University of North Carolina, Chapel Hill, said that “biologics have become the treatment of choice for moderate to severe psoriasis, and the use of biosimilars may be an alternative to reduce psoriasis treatment costs.
“Unfortunately, this study included a comparison of the existing biosimilars, which are drugs that are not the first line of treatment for psoriasis any longer,” added Dr. Lugo-Somolinos, who was not involved in the study.
Neil J. Korman, MD, PhD, professor of dermatology and codirector of the Skin Study Center at Case Western Reserve University, Cleveland, said the study was an important systematic review.
“This is a very timely publication because in the United States, several biosimilars are reaching the market in 2023,” he said. “The costs of the originator biologics are extraordinarily high, and the promise of biosimilars is that their costs will be significantly lower.”
Because all the studies were short term, Dr. Korman, who was not involved in the study, joins the study authors in recommending further related research into the long-term safety and efficacy of these agents.
Dr. Feldman, as well as one study author and one editorial author, reported relevant relationships with various pharmaceutical companies, including those that develop biosimilars. The remaining study authors, as well as Dr. Lugo-Somolinos and Dr. Korman, reported no relevant relationships. The study was funded by the Psoriasis Association and supported by the NIHR (National Institute for Health and Care Research) Manchester Biomedical Research Centre. All outside experts commented by email.
The effectiveness and safety of biosimilars for psoriasis appear to be similar to the originator biologics, reported the authors of a review of studies comparing the two.
“This systematic review found that there was no clinically or statistically significant difference in the efficacy and safety between biosimilars and originators of adalimumab, etanercept, infliximab, and ustekinumab for the treatment of psoriasis,” senior study author and clinical lecturer Zenas Z. N. Yiu, MBChB, PhD, and his colleagues at the University of Manchester, England, wrote in JAMA Dermatology.“The biosimilars evaluated in this study could be considered alongside originators for biologic-naive patients to improve the accessibility of biological treatments,” they added. “Switching patients currently on originators to biosimilars could be considered where clinically appropriate to reduce treatment costs.”
Biologics versus biosimilars
In contrast to most chemically synthesized drugs, biologics are created from living organisms, and they have complex structures that can vary slightly from batch to batch, Luigi Naldi, MD, director of the department of dermatology of Ospedale San Bortolo, Vicenza, Italy, and Antonio Addis, PharmD, researcher in the department of epidemiology, Regione Lazio, in Rome, wrote in an accompanying editorial.
Once the patent on the “originator” biologic expires, U.S. and European regulators allow other manufacturers to develop similar molecules – biosimilars – through an abbreviated approval process. If the results of a limited number of equivalence or noninferiority clinical trials are acceptable, registration for all the indications of the originator is allowed for its biosimilars. Referring to the expense of biologics, Dr. Naldi and Dr. Addis noted that in the United States, “biologics comprise less than 3% of the volume of drugs on the market, but account for more than one-third of all drug spending.”
Systematic review
Dr. Yiu and his colleagues queried standard medical research databases in August 2022, and included 14 randomized clinical trials (10 adalimumab, 2 etanercept, 1 infliximab, and 1 ustekinumab) and 3 cohort studies (1 adalimumab, 1 etanercept, 1 infliximab and etanercept) in their review.
Twelve trials compared biosimilars vs. originators in originator-naive patients, and 11 trials compared switching from originators to biosimilars vs. continuous treatment with the originator.
The researchers found the following:
At week 16, mean PASI75 (Psoriasis Area and Severity Index) response rates ranges from 60.7% to 90.6% for adalimumab biosimilars, vs. 61.5% to 91.7% for the originator. Mean PASI75 responses for the two etanercept biosimilars were 56.1% and 76.7% vs. 55.5% and 73.4% for the originator. In the ustekinumab study, mean PASI75 responses were 86.1% for the biosimilar vs. 84.0% for the originator.
At week 52, mean PASI75 responses were between 86.3% and 92.8% for adalimumab biosimilars vs. 84.9% and 93.9% for the originator. In the one comparison of an etanercept biosimilar, mean PAS175 responses were 80.9% for the biosimilar vs. 82.9% for the originator.
In studies involving patients switching from the originator to a biosimilar vs. continuing treatment with the originator, 32-week response rates ranged from 87.0% to 91.3% for adalimumab biosimilars and from 88.2% to 93.2% for the originator. In the one ustekinumab study, the 32-week mean PASI75 response was 92.6% after switching from the originator to a biosimilar vs. 92.9% with continuous treatment with the originator.
At week 52, mean PASI75 responses to adalimumab were between 84.2% and 94.8% for patients who switched to biosimilars and between 88.1% and 93.9% for those who stayed on the originator.
At week 52, in all the randomized trials, the incidence of adverse events and serious adverse events among those who switched to the biosimilar and those who continued with the originator were similar. Two cohort studies showed similar safety outcomes between originators and biosimilars, but one reported more adverse events in patients who switched to adalimumab biosimilars (P = .04).
Three clinical trials showed low risk for bias, 11 had moderate risk, and all cohort studies had moderate to high risk for bias.
Experts weigh in
Asked to comment on the study, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, Winston-Salem, N.C., told this news organization that he expects that the results will affect patient care.
However, he added, “I believe the decision of whether to use a biosimilar instead of the originator biologic may be more in the hands of the insurers than in the hands of physicians and patients.
“Biologics for psoriasis are so complicated that even the originator products vary from batch to batch. A biosimilar is basically like another batch of the innovative product,” explained Dr. Feldman, who was not involved in the study. “If we’re comfortable with patients being on different batches of the innovator product, we probably should be comfortable with them being on a biosimilar, as we have more evidence for the similarity of the biosimilar than we do for the current batch of the originator product.”
Aída Lugo-Somolinos, MD, professor of dermatology and director of the Contact Dermatitis Clinic at the University of North Carolina, Chapel Hill, said that “biologics have become the treatment of choice for moderate to severe psoriasis, and the use of biosimilars may be an alternative to reduce psoriasis treatment costs.
“Unfortunately, this study included a comparison of the existing biosimilars, which are drugs that are not the first line of treatment for psoriasis any longer,” added Dr. Lugo-Somolinos, who was not involved in the study.
Neil J. Korman, MD, PhD, professor of dermatology and codirector of the Skin Study Center at Case Western Reserve University, Cleveland, said the study was an important systematic review.
“This is a very timely publication because in the United States, several biosimilars are reaching the market in 2023,” he said. “The costs of the originator biologics are extraordinarily high, and the promise of biosimilars is that their costs will be significantly lower.”
Because all the studies were short term, Dr. Korman, who was not involved in the study, joins the study authors in recommending further related research into the long-term safety and efficacy of these agents.
Dr. Feldman, as well as one study author and one editorial author, reported relevant relationships with various pharmaceutical companies, including those that develop biosimilars. The remaining study authors, as well as Dr. Lugo-Somolinos and Dr. Korman, reported no relevant relationships. The study was funded by the Psoriasis Association and supported by the NIHR (National Institute for Health and Care Research) Manchester Biomedical Research Centre. All outside experts commented by email.
The effectiveness and safety of biosimilars for psoriasis appear to be similar to the originator biologics, reported the authors of a review of studies comparing the two.
“This systematic review found that there was no clinically or statistically significant difference in the efficacy and safety between biosimilars and originators of adalimumab, etanercept, infliximab, and ustekinumab for the treatment of psoriasis,” senior study author and clinical lecturer Zenas Z. N. Yiu, MBChB, PhD, and his colleagues at the University of Manchester, England, wrote in JAMA Dermatology.“The biosimilars evaluated in this study could be considered alongside originators for biologic-naive patients to improve the accessibility of biological treatments,” they added. “Switching patients currently on originators to biosimilars could be considered where clinically appropriate to reduce treatment costs.”
Biologics versus biosimilars
In contrast to most chemically synthesized drugs, biologics are created from living organisms, and they have complex structures that can vary slightly from batch to batch, Luigi Naldi, MD, director of the department of dermatology of Ospedale San Bortolo, Vicenza, Italy, and Antonio Addis, PharmD, researcher in the department of epidemiology, Regione Lazio, in Rome, wrote in an accompanying editorial.
Once the patent on the “originator” biologic expires, U.S. and European regulators allow other manufacturers to develop similar molecules – biosimilars – through an abbreviated approval process. If the results of a limited number of equivalence or noninferiority clinical trials are acceptable, registration for all the indications of the originator is allowed for its biosimilars. Referring to the expense of biologics, Dr. Naldi and Dr. Addis noted that in the United States, “biologics comprise less than 3% of the volume of drugs on the market, but account for more than one-third of all drug spending.”
Systematic review
Dr. Yiu and his colleagues queried standard medical research databases in August 2022, and included 14 randomized clinical trials (10 adalimumab, 2 etanercept, 1 infliximab, and 1 ustekinumab) and 3 cohort studies (1 adalimumab, 1 etanercept, 1 infliximab and etanercept) in their review.
Twelve trials compared biosimilars vs. originators in originator-naive patients, and 11 trials compared switching from originators to biosimilars vs. continuous treatment with the originator.
The researchers found the following:
At week 16, mean PASI75 (Psoriasis Area and Severity Index) response rates ranges from 60.7% to 90.6% for adalimumab biosimilars, vs. 61.5% to 91.7% for the originator. Mean PASI75 responses for the two etanercept biosimilars were 56.1% and 76.7% vs. 55.5% and 73.4% for the originator. In the ustekinumab study, mean PASI75 responses were 86.1% for the biosimilar vs. 84.0% for the originator.
At week 52, mean PASI75 responses were between 86.3% and 92.8% for adalimumab biosimilars vs. 84.9% and 93.9% for the originator. In the one comparison of an etanercept biosimilar, mean PAS175 responses were 80.9% for the biosimilar vs. 82.9% for the originator.
In studies involving patients switching from the originator to a biosimilar vs. continuing treatment with the originator, 32-week response rates ranged from 87.0% to 91.3% for adalimumab biosimilars and from 88.2% to 93.2% for the originator. In the one ustekinumab study, the 32-week mean PASI75 response was 92.6% after switching from the originator to a biosimilar vs. 92.9% with continuous treatment with the originator.
At week 52, mean PASI75 responses to adalimumab were between 84.2% and 94.8% for patients who switched to biosimilars and between 88.1% and 93.9% for those who stayed on the originator.
At week 52, in all the randomized trials, the incidence of adverse events and serious adverse events among those who switched to the biosimilar and those who continued with the originator were similar. Two cohort studies showed similar safety outcomes between originators and biosimilars, but one reported more adverse events in patients who switched to adalimumab biosimilars (P = .04).
Three clinical trials showed low risk for bias, 11 had moderate risk, and all cohort studies had moderate to high risk for bias.
Experts weigh in
Asked to comment on the study, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, Winston-Salem, N.C., told this news organization that he expects that the results will affect patient care.
However, he added, “I believe the decision of whether to use a biosimilar instead of the originator biologic may be more in the hands of the insurers than in the hands of physicians and patients.
“Biologics for psoriasis are so complicated that even the originator products vary from batch to batch. A biosimilar is basically like another batch of the innovative product,” explained Dr. Feldman, who was not involved in the study. “If we’re comfortable with patients being on different batches of the innovator product, we probably should be comfortable with them being on a biosimilar, as we have more evidence for the similarity of the biosimilar than we do for the current batch of the originator product.”
Aída Lugo-Somolinos, MD, professor of dermatology and director of the Contact Dermatitis Clinic at the University of North Carolina, Chapel Hill, said that “biologics have become the treatment of choice for moderate to severe psoriasis, and the use of biosimilars may be an alternative to reduce psoriasis treatment costs.
“Unfortunately, this study included a comparison of the existing biosimilars, which are drugs that are not the first line of treatment for psoriasis any longer,” added Dr. Lugo-Somolinos, who was not involved in the study.
Neil J. Korman, MD, PhD, professor of dermatology and codirector of the Skin Study Center at Case Western Reserve University, Cleveland, said the study was an important systematic review.
“This is a very timely publication because in the United States, several biosimilars are reaching the market in 2023,” he said. “The costs of the originator biologics are extraordinarily high, and the promise of biosimilars is that their costs will be significantly lower.”
Because all the studies were short term, Dr. Korman, who was not involved in the study, joins the study authors in recommending further related research into the long-term safety and efficacy of these agents.
Dr. Feldman, as well as one study author and one editorial author, reported relevant relationships with various pharmaceutical companies, including those that develop biosimilars. The remaining study authors, as well as Dr. Lugo-Somolinos and Dr. Korman, reported no relevant relationships. The study was funded by the Psoriasis Association and supported by the NIHR (National Institute for Health and Care Research) Manchester Biomedical Research Centre. All outside experts commented by email.
FROM JAMA DERMATOLOGY
Does colchicine have a role in treating excess ASCVD risk in patients with chronic inflammatory conditions?
The recent Food and Drug Administration approval of colchicine 0.5 mg (Lodoco) for use in atherosclerotic cardiovascular disease (ASCVD) prevention will possibly create opportunities to use the drug to treat residual risk for ASCVD in some patients with immune-mediated inflammatory diseases, particularly in rheumatology.
Potential in rheumatology
The 0.5-mg dose is just a shade under the 0.6-mg, twice daily dosing rheumatologists typically prescribe for gout, Christie Bartels, MD, MS, chief of rheumatology at the University of Wisconsin–Madison, said in an interview. Clinicians also use the 0.6-mg dose off-label for pseudogout or calcium pyrophosphate deposition disease (CPPD), Dr. Bartels noted.
The new formulation opens the consideration for using colchicine more in patients with psoriatic arthritis, lupus, and rheumatoid arthritis, she said. “I think we could certainly discuss it, particularly, in secondary prevention patients who already had an event or who are at the highest risk and already on optimal traditional agents,” she said.
She cited previous comments by Paul Ridker, MD, director of the center for cardiovascular disease prevention at Brigham and Women’s Hospital in Boston, and developer of the high-sensitivity C-reactive protein (hsCRP) test for measuring inflammatory markers. “We might not know the answer because Dr. Ridker pointed out he used colchicine 0.5 mg in patients that had a high-sensitivity CRP that was high; we need patients who have had inflammation of unknown origin, so those patients presumably weren’t already on another anti-inflammatory,” she said, noting that hydroxychloroquine, methotrexate, and some biologics provide some protection from cardiovascular risks.
However, a potential role for long-term colchicine 0.5 mg in ASCVD prevention may cause consideration for changing the drug’s role in gout treatment, Dr. Bartels said. “In gout, where we do have an FDA-approved indication for colchicine, we used to use it only for the first 6 months while we were getting patients to goal on allopurinol, which was usually then monotherapy after the first 6 months,” she said. “I think this will likely change how I treat gout patients in that I may also offer to continue both medications [colchicine and allopurinol] if they are tolerating them well.
“And then in patients where I’m using it off-label in CPPD, I might again share with them that in addition to possibly helping their CPPD, there may be this added benefit to reduce inflammation just in discussing the risks and benefits of the medicine.”
However, rheumatologists must be careful in using colchicine beyond the typical 6-month cycle, Dr. Bartels said. “One of the tricky things with colchicine, and part of the reason we did not traditionally continue it specifically past the first 6 months, was that it can cause myopathies or cytopenias, so we still have to counsel patients regarding these risks and monitor that,” she said.
Additionally, colchicine can have drug interactions with statins or calcium channel blockers that can change colchicine levels. “I think the dose here is so low, the 0.5 mg, that it’s probably still safe, but again, it’s something that we have to take a look at in the patient’s whole picture and the rest of their burden of their meds in order to make a decision with them,” Dr. Bartels said.
Possibilities in dermatology
The LoDoCo2 trial one of two major randomized trials that supported approval of colchicine 0.5 mg, reported that treated patients had a 60% lower rate of gout than the placebo group (1.4% vs. 3.4%). Joel Gelfand, MD, MSCE, the James J. Leyden professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, pointed to this in evaluating the dermatologic implications of the drug’s approval. “This may be of particular interest as people with psoriasis have an increased risk of gout,” he said in emailed comments.
Colchicine’s mechanism of action to reduce inflammation parallels that of tumor necrosis factor (TNF) inhibitors used for dermatologic indications, namely by inhibiting leukocyte adhesion to disrupt the downregulation of TNF receptors, Dr. Gelfand said.
“Interestingly, observational data suggests biologics that target TNF such as adalimumab, etanercept, etc., are associated with a reduction in CV events, and in placebo-controlled trials we conducted in psoriasis patients, it reduced key inflammatory mediators of cardiovascular disease, including IL [interleukin]-6,” he said. “Randomized clinical trials to evaluate the ability of TNF inhibitors, which are now available as biosimilars, to prevent cardiovascular events in high-risk patients, should be conducted, and more work is needed to identify which additional immune-targeted treatments may lower CV risk with an acceptable safety profile.”
Colchicine currently has few indications for rare conditions in dermatology, Dr. Gelfand said, including Sweets syndrome, subcorneal pustular dermatosis, and cutaneous vasculitis. “There are some reports to suggest it may help psoriatic disease, but current data are limited and insufficient to recommend its use for psoriasis and/or psoriatic arthritis,” he said.
The approval of colchicine 0.5 mg for ASCVD could be meaningful for people with psoriasis who are also being treated for CV risk factors, Dr. Gelfand said. “Additional considerations such as signs of residual inflammation (elevated hsCRP) and CV imaging findings may be used to further guide shared decision-making for optimal use,” he said.
Another consideration he noted: “This is also a novel 0.5-mg formulation, and thus cost may be an issue.”
Would side effects bar use in gastroenterology?
Colchicine 0.5 mg may not move the needle much for expanding treatment of ASCVD in patients with inflammatory bowel disease (IBD) and potentially other gastrointestinal conditions, Edward Loftus Jr., MD, the Maxine and Jack Zarrow Family professor of gastroenterology specifically for IBD at the Mayo Clinic in Rochester, Minn., told MDEdge in emailed comments. “Given the GI side effect profile [of colchicine], I am not sure I would go there,” he said.
“Hopefully, the prescribers of this low-dose formulation are aware of the gastrointestinal side effects, such as diarrhea and nausea, and educate patients about these side effects so that a proper risk-benefit discussion can ensue,” he said.
Dr. Bartels reporting a previous financial relationship with Pfizer. Dr. Gelfand said he has financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, GlaxoSmithKline, Twill, Lilly, Leo, Moonlake, Janssen Biologics, Novartis, Pfizer, UCB, Neuroderm, and Veolia North America. Dr. Loftus disclosed relationships with AbbVie, Alvotech, Amgen, Arena, Avalo, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene/Receptos, Celltrion Healthcare, Eli Lilly, Fresenius Kabi, Genentech, Gilead, GlaxoSmithKline, Gossamer Bio, Iterative Health, Janssen, KSL Diagnostics, Morphic, Ono, Pfizer, Sun, Surrozen, Takeda, Theravance, and UCB.
The recent Food and Drug Administration approval of colchicine 0.5 mg (Lodoco) for use in atherosclerotic cardiovascular disease (ASCVD) prevention will possibly create opportunities to use the drug to treat residual risk for ASCVD in some patients with immune-mediated inflammatory diseases, particularly in rheumatology.
Potential in rheumatology
The 0.5-mg dose is just a shade under the 0.6-mg, twice daily dosing rheumatologists typically prescribe for gout, Christie Bartels, MD, MS, chief of rheumatology at the University of Wisconsin–Madison, said in an interview. Clinicians also use the 0.6-mg dose off-label for pseudogout or calcium pyrophosphate deposition disease (CPPD), Dr. Bartels noted.
The new formulation opens the consideration for using colchicine more in patients with psoriatic arthritis, lupus, and rheumatoid arthritis, she said. “I think we could certainly discuss it, particularly, in secondary prevention patients who already had an event or who are at the highest risk and already on optimal traditional agents,” she said.
She cited previous comments by Paul Ridker, MD, director of the center for cardiovascular disease prevention at Brigham and Women’s Hospital in Boston, and developer of the high-sensitivity C-reactive protein (hsCRP) test for measuring inflammatory markers. “We might not know the answer because Dr. Ridker pointed out he used colchicine 0.5 mg in patients that had a high-sensitivity CRP that was high; we need patients who have had inflammation of unknown origin, so those patients presumably weren’t already on another anti-inflammatory,” she said, noting that hydroxychloroquine, methotrexate, and some biologics provide some protection from cardiovascular risks.
However, a potential role for long-term colchicine 0.5 mg in ASCVD prevention may cause consideration for changing the drug’s role in gout treatment, Dr. Bartels said. “In gout, where we do have an FDA-approved indication for colchicine, we used to use it only for the first 6 months while we were getting patients to goal on allopurinol, which was usually then monotherapy after the first 6 months,” she said. “I think this will likely change how I treat gout patients in that I may also offer to continue both medications [colchicine and allopurinol] if they are tolerating them well.
“And then in patients where I’m using it off-label in CPPD, I might again share with them that in addition to possibly helping their CPPD, there may be this added benefit to reduce inflammation just in discussing the risks and benefits of the medicine.”
However, rheumatologists must be careful in using colchicine beyond the typical 6-month cycle, Dr. Bartels said. “One of the tricky things with colchicine, and part of the reason we did not traditionally continue it specifically past the first 6 months, was that it can cause myopathies or cytopenias, so we still have to counsel patients regarding these risks and monitor that,” she said.
Additionally, colchicine can have drug interactions with statins or calcium channel blockers that can change colchicine levels. “I think the dose here is so low, the 0.5 mg, that it’s probably still safe, but again, it’s something that we have to take a look at in the patient’s whole picture and the rest of their burden of their meds in order to make a decision with them,” Dr. Bartels said.
Possibilities in dermatology
The LoDoCo2 trial one of two major randomized trials that supported approval of colchicine 0.5 mg, reported that treated patients had a 60% lower rate of gout than the placebo group (1.4% vs. 3.4%). Joel Gelfand, MD, MSCE, the James J. Leyden professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, pointed to this in evaluating the dermatologic implications of the drug’s approval. “This may be of particular interest as people with psoriasis have an increased risk of gout,” he said in emailed comments.
Colchicine’s mechanism of action to reduce inflammation parallels that of tumor necrosis factor (TNF) inhibitors used for dermatologic indications, namely by inhibiting leukocyte adhesion to disrupt the downregulation of TNF receptors, Dr. Gelfand said.
“Interestingly, observational data suggests biologics that target TNF such as adalimumab, etanercept, etc., are associated with a reduction in CV events, and in placebo-controlled trials we conducted in psoriasis patients, it reduced key inflammatory mediators of cardiovascular disease, including IL [interleukin]-6,” he said. “Randomized clinical trials to evaluate the ability of TNF inhibitors, which are now available as biosimilars, to prevent cardiovascular events in high-risk patients, should be conducted, and more work is needed to identify which additional immune-targeted treatments may lower CV risk with an acceptable safety profile.”
Colchicine currently has few indications for rare conditions in dermatology, Dr. Gelfand said, including Sweets syndrome, subcorneal pustular dermatosis, and cutaneous vasculitis. “There are some reports to suggest it may help psoriatic disease, but current data are limited and insufficient to recommend its use for psoriasis and/or psoriatic arthritis,” he said.
The approval of colchicine 0.5 mg for ASCVD could be meaningful for people with psoriasis who are also being treated for CV risk factors, Dr. Gelfand said. “Additional considerations such as signs of residual inflammation (elevated hsCRP) and CV imaging findings may be used to further guide shared decision-making for optimal use,” he said.
Another consideration he noted: “This is also a novel 0.5-mg formulation, and thus cost may be an issue.”
Would side effects bar use in gastroenterology?
Colchicine 0.5 mg may not move the needle much for expanding treatment of ASCVD in patients with inflammatory bowel disease (IBD) and potentially other gastrointestinal conditions, Edward Loftus Jr., MD, the Maxine and Jack Zarrow Family professor of gastroenterology specifically for IBD at the Mayo Clinic in Rochester, Minn., told MDEdge in emailed comments. “Given the GI side effect profile [of colchicine], I am not sure I would go there,” he said.
“Hopefully, the prescribers of this low-dose formulation are aware of the gastrointestinal side effects, such as diarrhea and nausea, and educate patients about these side effects so that a proper risk-benefit discussion can ensue,” he said.
Dr. Bartels reporting a previous financial relationship with Pfizer. Dr. Gelfand said he has financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, GlaxoSmithKline, Twill, Lilly, Leo, Moonlake, Janssen Biologics, Novartis, Pfizer, UCB, Neuroderm, and Veolia North America. Dr. Loftus disclosed relationships with AbbVie, Alvotech, Amgen, Arena, Avalo, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene/Receptos, Celltrion Healthcare, Eli Lilly, Fresenius Kabi, Genentech, Gilead, GlaxoSmithKline, Gossamer Bio, Iterative Health, Janssen, KSL Diagnostics, Morphic, Ono, Pfizer, Sun, Surrozen, Takeda, Theravance, and UCB.
The recent Food and Drug Administration approval of colchicine 0.5 mg (Lodoco) for use in atherosclerotic cardiovascular disease (ASCVD) prevention will possibly create opportunities to use the drug to treat residual risk for ASCVD in some patients with immune-mediated inflammatory diseases, particularly in rheumatology.
Potential in rheumatology
The 0.5-mg dose is just a shade under the 0.6-mg, twice daily dosing rheumatologists typically prescribe for gout, Christie Bartels, MD, MS, chief of rheumatology at the University of Wisconsin–Madison, said in an interview. Clinicians also use the 0.6-mg dose off-label for pseudogout or calcium pyrophosphate deposition disease (CPPD), Dr. Bartels noted.
The new formulation opens the consideration for using colchicine more in patients with psoriatic arthritis, lupus, and rheumatoid arthritis, she said. “I think we could certainly discuss it, particularly, in secondary prevention patients who already had an event or who are at the highest risk and already on optimal traditional agents,” she said.
She cited previous comments by Paul Ridker, MD, director of the center for cardiovascular disease prevention at Brigham and Women’s Hospital in Boston, and developer of the high-sensitivity C-reactive protein (hsCRP) test for measuring inflammatory markers. “We might not know the answer because Dr. Ridker pointed out he used colchicine 0.5 mg in patients that had a high-sensitivity CRP that was high; we need patients who have had inflammation of unknown origin, so those patients presumably weren’t already on another anti-inflammatory,” she said, noting that hydroxychloroquine, methotrexate, and some biologics provide some protection from cardiovascular risks.
However, a potential role for long-term colchicine 0.5 mg in ASCVD prevention may cause consideration for changing the drug’s role in gout treatment, Dr. Bartels said. “In gout, where we do have an FDA-approved indication for colchicine, we used to use it only for the first 6 months while we were getting patients to goal on allopurinol, which was usually then monotherapy after the first 6 months,” she said. “I think this will likely change how I treat gout patients in that I may also offer to continue both medications [colchicine and allopurinol] if they are tolerating them well.
“And then in patients where I’m using it off-label in CPPD, I might again share with them that in addition to possibly helping their CPPD, there may be this added benefit to reduce inflammation just in discussing the risks and benefits of the medicine.”
However, rheumatologists must be careful in using colchicine beyond the typical 6-month cycle, Dr. Bartels said. “One of the tricky things with colchicine, and part of the reason we did not traditionally continue it specifically past the first 6 months, was that it can cause myopathies or cytopenias, so we still have to counsel patients regarding these risks and monitor that,” she said.
Additionally, colchicine can have drug interactions with statins or calcium channel blockers that can change colchicine levels. “I think the dose here is so low, the 0.5 mg, that it’s probably still safe, but again, it’s something that we have to take a look at in the patient’s whole picture and the rest of their burden of their meds in order to make a decision with them,” Dr. Bartels said.
Possibilities in dermatology
The LoDoCo2 trial one of two major randomized trials that supported approval of colchicine 0.5 mg, reported that treated patients had a 60% lower rate of gout than the placebo group (1.4% vs. 3.4%). Joel Gelfand, MD, MSCE, the James J. Leyden professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, pointed to this in evaluating the dermatologic implications of the drug’s approval. “This may be of particular interest as people with psoriasis have an increased risk of gout,” he said in emailed comments.
Colchicine’s mechanism of action to reduce inflammation parallels that of tumor necrosis factor (TNF) inhibitors used for dermatologic indications, namely by inhibiting leukocyte adhesion to disrupt the downregulation of TNF receptors, Dr. Gelfand said.
“Interestingly, observational data suggests biologics that target TNF such as adalimumab, etanercept, etc., are associated with a reduction in CV events, and in placebo-controlled trials we conducted in psoriasis patients, it reduced key inflammatory mediators of cardiovascular disease, including IL [interleukin]-6,” he said. “Randomized clinical trials to evaluate the ability of TNF inhibitors, which are now available as biosimilars, to prevent cardiovascular events in high-risk patients, should be conducted, and more work is needed to identify which additional immune-targeted treatments may lower CV risk with an acceptable safety profile.”
Colchicine currently has few indications for rare conditions in dermatology, Dr. Gelfand said, including Sweets syndrome, subcorneal pustular dermatosis, and cutaneous vasculitis. “There are some reports to suggest it may help psoriatic disease, but current data are limited and insufficient to recommend its use for psoriasis and/or psoriatic arthritis,” he said.
The approval of colchicine 0.5 mg for ASCVD could be meaningful for people with psoriasis who are also being treated for CV risk factors, Dr. Gelfand said. “Additional considerations such as signs of residual inflammation (elevated hsCRP) and CV imaging findings may be used to further guide shared decision-making for optimal use,” he said.
Another consideration he noted: “This is also a novel 0.5-mg formulation, and thus cost may be an issue.”
Would side effects bar use in gastroenterology?
Colchicine 0.5 mg may not move the needle much for expanding treatment of ASCVD in patients with inflammatory bowel disease (IBD) and potentially other gastrointestinal conditions, Edward Loftus Jr., MD, the Maxine and Jack Zarrow Family professor of gastroenterology specifically for IBD at the Mayo Clinic in Rochester, Minn., told MDEdge in emailed comments. “Given the GI side effect profile [of colchicine], I am not sure I would go there,” he said.
“Hopefully, the prescribers of this low-dose formulation are aware of the gastrointestinal side effects, such as diarrhea and nausea, and educate patients about these side effects so that a proper risk-benefit discussion can ensue,” he said.
Dr. Bartels reporting a previous financial relationship with Pfizer. Dr. Gelfand said he has financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, GlaxoSmithKline, Twill, Lilly, Leo, Moonlake, Janssen Biologics, Novartis, Pfizer, UCB, Neuroderm, and Veolia North America. Dr. Loftus disclosed relationships with AbbVie, Alvotech, Amgen, Arena, Avalo, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene/Receptos, Celltrion Healthcare, Eli Lilly, Fresenius Kabi, Genentech, Gilead, GlaxoSmithKline, Gossamer Bio, Iterative Health, Janssen, KSL Diagnostics, Morphic, Ono, Pfizer, Sun, Surrozen, Takeda, Theravance, and UCB.
Study points to need to improve identification of culprit in SJS/TEN cases
demonstrated.
“Prompt identification and discontinuation of a culprit drug is critical to improving patient outcomes and preventing recurrence,” researchers led by Sherrie J. Divito, MD, PhD, of the department of dermatology at Brigham and Women’s Hospital, Boston, wrote in the study, which was published online in JAMA Dermatology. “Identification is difficult because there is no laboratory test or other criterion standard (in the absence of rechallenge) to identify a culprit drug, and patients take on average six medications at the time of their reaction. Consequently, many patients may be labeled as allergic to multiple agents.”
Although failing to identify the culprit drug can have severe consequences, they added, “overlabeling” (labeling a patient as allergic to a drug or drugs that they can tolerate safely) “is not insignificant.” As a result of overlabeling, “the patient may receive a less efficacious, more toxic, and/or more expensive agent than necessary, and in some cases may be left without treatment for their underlying disease.”
To evaluate the outcomes of patients’ allergy lists, current approaches to identify culprit drugs such as the Algorithm for Drug Causality for Epidermal Necrolysis (ALDEN), which was published in 2010, and potential methods of improving culprit drug identification, the researchers performed a retrospective cohort study of 48 patients at Brigham and Women’s Hospital and Massachusetts General Hospital, with clinically and histologically confirmed cases of SJS/TEN between January 2000 and July 2018. Of the 48 patients, 26 had SJS/TEN overlap and 22 had TEN. Their median age at diagnosis was 40 years; 60.4% were female; and 52.1% were white, 12.5% were Black, 10.4% were Hispanic, and 8.3% were Asian. They took a median of 6.5 drugs in the 3 months prior to disease onset.
The researchers observed that all patients had at least one drug labeled as an allergy. A single culprit drug was labeled in 17 cases, but physicians communicated certainty in only 7 of those cases. Among all 48 patients, 104 drugs were labeled as allergies.
To identify a culprit drug, physicians appeared to mainly rely on two factors: drug notoriety and timing of exposure, compared with the onset of SJS/TEN. “Identifying high-risk medications seemed heuristic, with one or more drugs in question noted in the record as a common culprit without reference to published or vetted data regarding risk,” the researchers wrote. “Regarding timing, drug charts when present in medical records were incomplete, as they focused predominantly on high-notoriety drugs.”
In other findings, ALDEN scoring was discordant with physician-labeled lists in 28 cases. It labeled an additional 9 drugs missed by physicians and scored 43 drugs labeled as allergens by physicians as “unlikely.” The researchers also reported that 20 cases could have potentially benefited from human leukocyte antigen testing.
Their results “underscore the need for a laboratory test to identify culprit drugs,” but without such a test, “a systematic unbiased approach, such as ALDEN or the RegiSCAR database, with possibly HLA testing, should be considered to ensure the true culprit drug is not missed and exonerate as many nonculprit drugs as possible,” Dr. Divito and colleagues concluded.
They acknowledged certain limitations of the analysis, including its retrospective design and that many cases predated research advances in the topic area that took place during the 18-year study period.
Karl Saardi, MD, director of the inpatient dermatology service at George Washington University Hospital, Washington, who was asked to comment on the study, said that the findings “are consistent with clinical practice in that drug causality is usually determined by ‘gestalt’ rather than objective tools like the ALDEN algorithm.”
“The main limitation is the small size, which suggests the study sites are low-volume centers for SJS/TEN. The fact that the ALDEN score wasn’t developed until 2010 means that all the cases included prior to 2010 would not have applied the ALDEN algorithm, so I think the metric about how infrequently ALDEN was applied is not very meaningful.”
Still, he said that he was “surprised” by the number of medications that were added as allergies based on clinical impression, “and I’m glad this article does cast some light on the issue. In my experience, beta-lactam antibiotics are often – incorrectly – deemed to be the cause of SJS/TEN when further review of the patient’s medication history clearly shows they have tolerated these drugs multiple times in the past.”
Since 2000, he added, “our understanding of SJS/TEN has grown substantially including the identification of MIRM [mycoplasma-induced rash and mucositis]/RIME [reactive infections mucocutaneous eruptions] and GBFDE [generalized bullous fixed drug eruption] as mimickers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was also asked to comment on the study, agreed that a limitation of the study is that it partially preceded development of the unbiased approaches to determining the cause of a medication reaction, such as the ALDEN system. “A strength of this study is the examination of heuristics in dermatology and how they relate to patient safety,” she added.
The study was funded by grants from the National Institutes of Health, a Dermatology Foundation Diversity Research Supplement Award, and by the German Research Foundation. Dr. Divito reported receiving personal fees from Adaptimmune and MEI Pharma and a provisional patent issued from Brigham and Women’s Hospital outside the submitted work. Neither Dr. Saardi nor Dr. Ko reported having relevant disclosures.
demonstrated.
“Prompt identification and discontinuation of a culprit drug is critical to improving patient outcomes and preventing recurrence,” researchers led by Sherrie J. Divito, MD, PhD, of the department of dermatology at Brigham and Women’s Hospital, Boston, wrote in the study, which was published online in JAMA Dermatology. “Identification is difficult because there is no laboratory test or other criterion standard (in the absence of rechallenge) to identify a culprit drug, and patients take on average six medications at the time of their reaction. Consequently, many patients may be labeled as allergic to multiple agents.”
Although failing to identify the culprit drug can have severe consequences, they added, “overlabeling” (labeling a patient as allergic to a drug or drugs that they can tolerate safely) “is not insignificant.” As a result of overlabeling, “the patient may receive a less efficacious, more toxic, and/or more expensive agent than necessary, and in some cases may be left without treatment for their underlying disease.”
To evaluate the outcomes of patients’ allergy lists, current approaches to identify culprit drugs such as the Algorithm for Drug Causality for Epidermal Necrolysis (ALDEN), which was published in 2010, and potential methods of improving culprit drug identification, the researchers performed a retrospective cohort study of 48 patients at Brigham and Women’s Hospital and Massachusetts General Hospital, with clinically and histologically confirmed cases of SJS/TEN between January 2000 and July 2018. Of the 48 patients, 26 had SJS/TEN overlap and 22 had TEN. Their median age at diagnosis was 40 years; 60.4% were female; and 52.1% were white, 12.5% were Black, 10.4% were Hispanic, and 8.3% were Asian. They took a median of 6.5 drugs in the 3 months prior to disease onset.
The researchers observed that all patients had at least one drug labeled as an allergy. A single culprit drug was labeled in 17 cases, but physicians communicated certainty in only 7 of those cases. Among all 48 patients, 104 drugs were labeled as allergies.
To identify a culprit drug, physicians appeared to mainly rely on two factors: drug notoriety and timing of exposure, compared with the onset of SJS/TEN. “Identifying high-risk medications seemed heuristic, with one or more drugs in question noted in the record as a common culprit without reference to published or vetted data regarding risk,” the researchers wrote. “Regarding timing, drug charts when present in medical records were incomplete, as they focused predominantly on high-notoriety drugs.”
In other findings, ALDEN scoring was discordant with physician-labeled lists in 28 cases. It labeled an additional 9 drugs missed by physicians and scored 43 drugs labeled as allergens by physicians as “unlikely.” The researchers also reported that 20 cases could have potentially benefited from human leukocyte antigen testing.
Their results “underscore the need for a laboratory test to identify culprit drugs,” but without such a test, “a systematic unbiased approach, such as ALDEN or the RegiSCAR database, with possibly HLA testing, should be considered to ensure the true culprit drug is not missed and exonerate as many nonculprit drugs as possible,” Dr. Divito and colleagues concluded.
They acknowledged certain limitations of the analysis, including its retrospective design and that many cases predated research advances in the topic area that took place during the 18-year study period.
Karl Saardi, MD, director of the inpatient dermatology service at George Washington University Hospital, Washington, who was asked to comment on the study, said that the findings “are consistent with clinical practice in that drug causality is usually determined by ‘gestalt’ rather than objective tools like the ALDEN algorithm.”
“The main limitation is the small size, which suggests the study sites are low-volume centers for SJS/TEN. The fact that the ALDEN score wasn’t developed until 2010 means that all the cases included prior to 2010 would not have applied the ALDEN algorithm, so I think the metric about how infrequently ALDEN was applied is not very meaningful.”
Still, he said that he was “surprised” by the number of medications that were added as allergies based on clinical impression, “and I’m glad this article does cast some light on the issue. In my experience, beta-lactam antibiotics are often – incorrectly – deemed to be the cause of SJS/TEN when further review of the patient’s medication history clearly shows they have tolerated these drugs multiple times in the past.”
Since 2000, he added, “our understanding of SJS/TEN has grown substantially including the identification of MIRM [mycoplasma-induced rash and mucositis]/RIME [reactive infections mucocutaneous eruptions] and GBFDE [generalized bullous fixed drug eruption] as mimickers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was also asked to comment on the study, agreed that a limitation of the study is that it partially preceded development of the unbiased approaches to determining the cause of a medication reaction, such as the ALDEN system. “A strength of this study is the examination of heuristics in dermatology and how they relate to patient safety,” she added.
The study was funded by grants from the National Institutes of Health, a Dermatology Foundation Diversity Research Supplement Award, and by the German Research Foundation. Dr. Divito reported receiving personal fees from Adaptimmune and MEI Pharma and a provisional patent issued from Brigham and Women’s Hospital outside the submitted work. Neither Dr. Saardi nor Dr. Ko reported having relevant disclosures.
demonstrated.
“Prompt identification and discontinuation of a culprit drug is critical to improving patient outcomes and preventing recurrence,” researchers led by Sherrie J. Divito, MD, PhD, of the department of dermatology at Brigham and Women’s Hospital, Boston, wrote in the study, which was published online in JAMA Dermatology. “Identification is difficult because there is no laboratory test or other criterion standard (in the absence of rechallenge) to identify a culprit drug, and patients take on average six medications at the time of their reaction. Consequently, many patients may be labeled as allergic to multiple agents.”
Although failing to identify the culprit drug can have severe consequences, they added, “overlabeling” (labeling a patient as allergic to a drug or drugs that they can tolerate safely) “is not insignificant.” As a result of overlabeling, “the patient may receive a less efficacious, more toxic, and/or more expensive agent than necessary, and in some cases may be left without treatment for their underlying disease.”
To evaluate the outcomes of patients’ allergy lists, current approaches to identify culprit drugs such as the Algorithm for Drug Causality for Epidermal Necrolysis (ALDEN), which was published in 2010, and potential methods of improving culprit drug identification, the researchers performed a retrospective cohort study of 48 patients at Brigham and Women’s Hospital and Massachusetts General Hospital, with clinically and histologically confirmed cases of SJS/TEN between January 2000 and July 2018. Of the 48 patients, 26 had SJS/TEN overlap and 22 had TEN. Their median age at diagnosis was 40 years; 60.4% were female; and 52.1% were white, 12.5% were Black, 10.4% were Hispanic, and 8.3% were Asian. They took a median of 6.5 drugs in the 3 months prior to disease onset.
The researchers observed that all patients had at least one drug labeled as an allergy. A single culprit drug was labeled in 17 cases, but physicians communicated certainty in only 7 of those cases. Among all 48 patients, 104 drugs were labeled as allergies.
To identify a culprit drug, physicians appeared to mainly rely on two factors: drug notoriety and timing of exposure, compared with the onset of SJS/TEN. “Identifying high-risk medications seemed heuristic, with one or more drugs in question noted in the record as a common culprit without reference to published or vetted data regarding risk,” the researchers wrote. “Regarding timing, drug charts when present in medical records were incomplete, as they focused predominantly on high-notoriety drugs.”
In other findings, ALDEN scoring was discordant with physician-labeled lists in 28 cases. It labeled an additional 9 drugs missed by physicians and scored 43 drugs labeled as allergens by physicians as “unlikely.” The researchers also reported that 20 cases could have potentially benefited from human leukocyte antigen testing.
Their results “underscore the need for a laboratory test to identify culprit drugs,” but without such a test, “a systematic unbiased approach, such as ALDEN or the RegiSCAR database, with possibly HLA testing, should be considered to ensure the true culprit drug is not missed and exonerate as many nonculprit drugs as possible,” Dr. Divito and colleagues concluded.
They acknowledged certain limitations of the analysis, including its retrospective design and that many cases predated research advances in the topic area that took place during the 18-year study period.
Karl Saardi, MD, director of the inpatient dermatology service at George Washington University Hospital, Washington, who was asked to comment on the study, said that the findings “are consistent with clinical practice in that drug causality is usually determined by ‘gestalt’ rather than objective tools like the ALDEN algorithm.”
“The main limitation is the small size, which suggests the study sites are low-volume centers for SJS/TEN. The fact that the ALDEN score wasn’t developed until 2010 means that all the cases included prior to 2010 would not have applied the ALDEN algorithm, so I think the metric about how infrequently ALDEN was applied is not very meaningful.”
Still, he said that he was “surprised” by the number of medications that were added as allergies based on clinical impression, “and I’m glad this article does cast some light on the issue. In my experience, beta-lactam antibiotics are often – incorrectly – deemed to be the cause of SJS/TEN when further review of the patient’s medication history clearly shows they have tolerated these drugs multiple times in the past.”
Since 2000, he added, “our understanding of SJS/TEN has grown substantially including the identification of MIRM [mycoplasma-induced rash and mucositis]/RIME [reactive infections mucocutaneous eruptions] and GBFDE [generalized bullous fixed drug eruption] as mimickers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was also asked to comment on the study, agreed that a limitation of the study is that it partially preceded development of the unbiased approaches to determining the cause of a medication reaction, such as the ALDEN system. “A strength of this study is the examination of heuristics in dermatology and how they relate to patient safety,” she added.
The study was funded by grants from the National Institutes of Health, a Dermatology Foundation Diversity Research Supplement Award, and by the German Research Foundation. Dr. Divito reported receiving personal fees from Adaptimmune and MEI Pharma and a provisional patent issued from Brigham and Women’s Hospital outside the submitted work. Neither Dr. Saardi nor Dr. Ko reported having relevant disclosures.
FROM JAMA DERMATOLOGY
Fade haircut or something else?
The areas of hair loss and different length hairs in a patient with otherwise normal-density hair was consistent with a diagnosis of trichotillomania. The physician initially thought the patient had a “fade” hairstyle, but then observed him twirling his hair. Further queries confirmed a history of hair pulling (trichotillomania) and ingesting the hair (trichophagia).
In adulthood, trichotillomania affects females about 4 times more than males, although in childhood, the sex distribution appears about equal.1 The typical age of onset is between 10 to 13 years. Several mental health conditions are associated with trichotillomania, such as major depressive disorder, anxiety disorders, and substance use disorder, with trichotillomania generally preceding these comorbid disorders. It is important to rule out obsessive compulsive disorder and consider body dysmorphic disorder when making a diagnosis of trichotillomania.1
It is common to see androgenetic alopecia precipitated by hormone therapy in FTM individuals. This would usually cause thinning of the hair rather than the irregular hair lengths and pattern seen in this patient. Tinea capitis is also part of the differential diagnosis in a case such as this. It can be diagnosed by potassium hydroxide (KOH) preparations in the setting of friable and broken hair shafts (and sometimes erythema and inflammation).
Patients with trichotillomania may have hair with blunt or tapered ends; hair may also look like uneven stubble.2 The scalp is the most commonly involved site (72.8%), followed by eyebrows (50.7%), and the pubic region (50.7%).1 Useful diagnostic clues include an unusual shape of the affected area, accessible location, and a changing pattern from visit to visit.2 If trichotillomania is suspected, it might be useful to ask about hair-playing activities, such as twirling or twisting, or playing with the ends of eyelashes or eyebrows.2
Unwanted medical consequences of trichotillomania include skin damage (if sharp instruments are used for hair pulling), and the formation of gastrointestinal trichobezoars (hairballs) if trichophagia is present. Trichobezoars that cause bowel obstructions may need surgical intervention.1
Behavioral therapy is the first-line treatment for trichotillomania in all age groups.1,3 Treating any coexisting mental health disorders is also essential. There are currently no FDA-approved medications for treatment; however, there is evidence that N-acetylcysteine may be useful in treating adults with trichotillomania and other repetitive skin disorders.3 Antipsychotics and cannabinoid agonists also may be beneficial.1
This patient was continued on his current lithium, naltrexone, and bupropion regimen (rather than adding additional psychiatric medicines) as he was doing better than his previous baseline. He was advised to continue with his cognitive behavioral therapy. His hormone replacement therapy also was continued because it was not thought to be contributory. He was provided with education about symptoms of trichobezoar and red flag symptoms (eg, worsening nausea, vomiting, abdominal pain) that would necessitate emergency follow-up. His GERD symptoms were thought not to be related to his trichophagia. He was scheduled to follow up for routine primary care in 6 months.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Sarasawati Keeni, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Grant JE and Chamberlain SR. Trichotillomania. Am J Psychiatry. 2016;173:868-874. doi: 10.1176/appi.ajp.2016.15111432
2. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21:13-21. doi: 10.1111/j.1529-8019.2008.00165.x
3. Henkel ED, Jaquez SD, Diaz LZ. Pediatric trichotillomania: review of management. Pediatr Dermatol. 2019;36:803-807. doi: 10.1111/pde.13954

The areas of hair loss and different length hairs in a patient with otherwise normal-density hair was consistent with a diagnosis of trichotillomania. The physician initially thought the patient had a “fade” hairstyle, but then observed him twirling his hair. Further queries confirmed a history of hair pulling (trichotillomania) and ingesting the hair (trichophagia).
In adulthood, trichotillomania affects females about 4 times more than males, although in childhood, the sex distribution appears about equal.1 The typical age of onset is between 10 to 13 years. Several mental health conditions are associated with trichotillomania, such as major depressive disorder, anxiety disorders, and substance use disorder, with trichotillomania generally preceding these comorbid disorders. It is important to rule out obsessive compulsive disorder and consider body dysmorphic disorder when making a diagnosis of trichotillomania.1
It is common to see androgenetic alopecia precipitated by hormone therapy in FTM individuals. This would usually cause thinning of the hair rather than the irregular hair lengths and pattern seen in this patient. Tinea capitis is also part of the differential diagnosis in a case such as this. It can be diagnosed by potassium hydroxide (KOH) preparations in the setting of friable and broken hair shafts (and sometimes erythema and inflammation).
Patients with trichotillomania may have hair with blunt or tapered ends; hair may also look like uneven stubble.2 The scalp is the most commonly involved site (72.8%), followed by eyebrows (50.7%), and the pubic region (50.7%).1 Useful diagnostic clues include an unusual shape of the affected area, accessible location, and a changing pattern from visit to visit.2 If trichotillomania is suspected, it might be useful to ask about hair-playing activities, such as twirling or twisting, or playing with the ends of eyelashes or eyebrows.2
Unwanted medical consequences of trichotillomania include skin damage (if sharp instruments are used for hair pulling), and the formation of gastrointestinal trichobezoars (hairballs) if trichophagia is present. Trichobezoars that cause bowel obstructions may need surgical intervention.1
Behavioral therapy is the first-line treatment for trichotillomania in all age groups.1,3 Treating any coexisting mental health disorders is also essential. There are currently no FDA-approved medications for treatment; however, there is evidence that N-acetylcysteine may be useful in treating adults with trichotillomania and other repetitive skin disorders.3 Antipsychotics and cannabinoid agonists also may be beneficial.1
This patient was continued on his current lithium, naltrexone, and bupropion regimen (rather than adding additional psychiatric medicines) as he was doing better than his previous baseline. He was advised to continue with his cognitive behavioral therapy. His hormone replacement therapy also was continued because it was not thought to be contributory. He was provided with education about symptoms of trichobezoar and red flag symptoms (eg, worsening nausea, vomiting, abdominal pain) that would necessitate emergency follow-up. His GERD symptoms were thought not to be related to his trichophagia. He was scheduled to follow up for routine primary care in 6 months.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Sarasawati Keeni, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

The areas of hair loss and different length hairs in a patient with otherwise normal-density hair was consistent with a diagnosis of trichotillomania. The physician initially thought the patient had a “fade” hairstyle, but then observed him twirling his hair. Further queries confirmed a history of hair pulling (trichotillomania) and ingesting the hair (trichophagia).
In adulthood, trichotillomania affects females about 4 times more than males, although in childhood, the sex distribution appears about equal.1 The typical age of onset is between 10 to 13 years. Several mental health conditions are associated with trichotillomania, such as major depressive disorder, anxiety disorders, and substance use disorder, with trichotillomania generally preceding these comorbid disorders. It is important to rule out obsessive compulsive disorder and consider body dysmorphic disorder when making a diagnosis of trichotillomania.1
It is common to see androgenetic alopecia precipitated by hormone therapy in FTM individuals. This would usually cause thinning of the hair rather than the irregular hair lengths and pattern seen in this patient. Tinea capitis is also part of the differential diagnosis in a case such as this. It can be diagnosed by potassium hydroxide (KOH) preparations in the setting of friable and broken hair shafts (and sometimes erythema and inflammation).
Patients with trichotillomania may have hair with blunt or tapered ends; hair may also look like uneven stubble.2 The scalp is the most commonly involved site (72.8%), followed by eyebrows (50.7%), and the pubic region (50.7%).1 Useful diagnostic clues include an unusual shape of the affected area, accessible location, and a changing pattern from visit to visit.2 If trichotillomania is suspected, it might be useful to ask about hair-playing activities, such as twirling or twisting, or playing with the ends of eyelashes or eyebrows.2
Unwanted medical consequences of trichotillomania include skin damage (if sharp instruments are used for hair pulling), and the formation of gastrointestinal trichobezoars (hairballs) if trichophagia is present. Trichobezoars that cause bowel obstructions may need surgical intervention.1
Behavioral therapy is the first-line treatment for trichotillomania in all age groups.1,3 Treating any coexisting mental health disorders is also essential. There are currently no FDA-approved medications for treatment; however, there is evidence that N-acetylcysteine may be useful in treating adults with trichotillomania and other repetitive skin disorders.3 Antipsychotics and cannabinoid agonists also may be beneficial.1
This patient was continued on his current lithium, naltrexone, and bupropion regimen (rather than adding additional psychiatric medicines) as he was doing better than his previous baseline. He was advised to continue with his cognitive behavioral therapy. His hormone replacement therapy also was continued because it was not thought to be contributory. He was provided with education about symptoms of trichobezoar and red flag symptoms (eg, worsening nausea, vomiting, abdominal pain) that would necessitate emergency follow-up. His GERD symptoms were thought not to be related to his trichophagia. He was scheduled to follow up for routine primary care in 6 months.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Sarasawati Keeni, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Grant JE and Chamberlain SR. Trichotillomania. Am J Psychiatry. 2016;173:868-874. doi: 10.1176/appi.ajp.2016.15111432
2. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21:13-21. doi: 10.1111/j.1529-8019.2008.00165.x
3. Henkel ED, Jaquez SD, Diaz LZ. Pediatric trichotillomania: review of management. Pediatr Dermatol. 2019;36:803-807. doi: 10.1111/pde.13954
1. Grant JE and Chamberlain SR. Trichotillomania. Am J Psychiatry. 2016;173:868-874. doi: 10.1176/appi.ajp.2016.15111432
2. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21:13-21. doi: 10.1111/j.1529-8019.2008.00165.x
3. Henkel ED, Jaquez SD, Diaz LZ. Pediatric trichotillomania: review of management. Pediatr Dermatol. 2019;36:803-807. doi: 10.1111/pde.13954
Imaging techniques will revolutionize cancer detection, expert predicts
PHOENIX –
In a lecture during a multispecialty roundup of cutting-edge energy-based device applications at the annual conference of the American Society for Laser Medicine and Surgery, Dr. Barton, a biomedical engineer who directs the BIO5 Institute at the University of Arizona, Tucson, said that while no current modality exists to enable physicians in dermatology and other specialties to view internal structures throughout the entire body with cellular resolution, refining existing technologies is a good way to start.
In 2011, renowned cancer researchers Douglas Hanahan, PhD, and Robert A. Weinberg, PhD, proposed six hallmarks of cancer, which include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Each hallmark poses unique imaging challenges. For example, enabling replicative immortality “means that the cell nuclei change size and shape; they change their position,” said Dr. Barton, who is also professor of biomedical engineering and optical sciences at the university. “If we want to see that, we’re going to need an imaging modality that’s subcellular in resolution.”
Similarly, if clinicians want to view how proliferative signaling is changing, “that means being able to visualize the cell surface receptors; those are even smaller to actually visualize,” she said. “But we have technologies where we can target those receptors with fluorophores. And then we can look at large areas very quickly.” Meanwhile, the ability of cancer cells to resist cell death and evade growth suppressors often results in thickening of epithelium throughout the body. “So, if we can measure the thickness of the epithelium, we can see that there’s something wrong with that tissue,” she said.
As for cancer’s propensity for invasion and metastasis, “here, we’re looking at how the collagen structure [between the cells] has changed and whether there’s layer breakdown or not. Optical imaging can detect cancer. However, high resolution optical techniques can only image about 1 mm deep, so unless you’re looking at the skin or the eye, you’re going to have to develop an endoscope to be able to view these hallmarks.”
OCT images the tissue microstructure, generally in a resolution of 2-20 microns, at a depth of 1-2 mm, and it measures reflected light. When possible, Dr. Barton combines OCT with laser-induced fluorescence for enhanced accuracy of detection of cancer. Induced fluorescence senses molecular information with the natural fluorophores in the body or with targeted exogenous agents. Then there’s multiphoton microscopy, an advanced imaging technique that enables clinicians to view cellular and subcellular events within living tissue. Early models of this technology “took up entire benches” in physics labs, Dr. Barton said, but she and other investigators are designing smaller devices for use in clinics. “This is exciting, because not only do we [view] subcellular structure with this modality, but it can also be highly sensitive to collagen structure,” she said.
Ovarian cancer model
In a model of ovarian cancer, she and colleagues externalized the ovaries of a mouse, imaged the organs, put them back in, and reassessed them at 8 weeks. “This model develops cancer very quickly,” said Dr. Barton, who once worked for McDonnell Douglas on the Space Station program. At 8 weeks, using fluorescence and targeted agents with a tabletop multiphoton microscopy system, they observed that the proliferation signals of cancer had begun. “So, with an agent targeted to the folate receptor or to other receptors that are implicated in cancer development, we can see that ovaries and fallopian tubes are lighting up,” she said.
With proof of concept established with the mouse study, she and other researchers are drawing from technological advances to create tiny laser systems for use in the clinic to image a variety of structures in the human body. Optics advances include bulk optics and all-fiber designs where engineers can create an imaging probe that’s only 125 microns in diameter, “or maybe even as small as 70 microns in diameter,” she said. “We can do fabrications on the tips of endoscopes to redirect the light and focus it. We can also do 3-D printing and spiral scanning to create miniature devices to make new advances. That means that instead of just white light imaging of the colon or the lung like we have had in the past, we can start moving into smaller structures, such as the eustachian tube, the fallopian tube, the bile ducts, or making miniature devices for brain biopsies, lung biopsies, and maybe being able to get into bronchioles and arterioles.”
According to Dr. Barton, prior research has demonstrated that cerebral vasculature can be imaged with a catheter 400 microns in diameter, the spaces in the lungs can be imaged with a needle that is 310 microns in diameter, and the inner structures of the eustachian tube can be viewed with an endoscope 1 mm in diameter.
She and her colleagues are developing an OCT/fluorescence imaging falloposcope that is 0.8 mm in diameter, flexible, and steerable, as a tool for early detection of ovarian cancer in humans. “It’s now known that most ovarian cancer starts in the fallopian tubes,” Dr. Barton said. “It’s metastatic disease when those cells break off from the fallopian tubes and go to the ovaries. We wanted to create an imaging system where we created a fiber bundle that we could navigate with white light and with fluorescence so that we can see these early stages of cancer [and] how they fluoresce differently. We also wanted to have an OCT system so that we could image through the wall of the fallopian tube and look for that layer thickening and other precursors to ovarian cancer.”
To date, in vivo testing in healthy women has demonstrated that the miniature endoscope is able to reach the fallopian tubes through the natural orifice of the vagina and uterus. “That is pretty exciting,” she said. “The images may not be of the highest quality, but we are advancing.”
Dr. Barton reported having no relevant financial disclosures.
PHOENIX –
In a lecture during a multispecialty roundup of cutting-edge energy-based device applications at the annual conference of the American Society for Laser Medicine and Surgery, Dr. Barton, a biomedical engineer who directs the BIO5 Institute at the University of Arizona, Tucson, said that while no current modality exists to enable physicians in dermatology and other specialties to view internal structures throughout the entire body with cellular resolution, refining existing technologies is a good way to start.
In 2011, renowned cancer researchers Douglas Hanahan, PhD, and Robert A. Weinberg, PhD, proposed six hallmarks of cancer, which include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Each hallmark poses unique imaging challenges. For example, enabling replicative immortality “means that the cell nuclei change size and shape; they change their position,” said Dr. Barton, who is also professor of biomedical engineering and optical sciences at the university. “If we want to see that, we’re going to need an imaging modality that’s subcellular in resolution.”
Similarly, if clinicians want to view how proliferative signaling is changing, “that means being able to visualize the cell surface receptors; those are even smaller to actually visualize,” she said. “But we have technologies where we can target those receptors with fluorophores. And then we can look at large areas very quickly.” Meanwhile, the ability of cancer cells to resist cell death and evade growth suppressors often results in thickening of epithelium throughout the body. “So, if we can measure the thickness of the epithelium, we can see that there’s something wrong with that tissue,” she said.
As for cancer’s propensity for invasion and metastasis, “here, we’re looking at how the collagen structure [between the cells] has changed and whether there’s layer breakdown or not. Optical imaging can detect cancer. However, high resolution optical techniques can only image about 1 mm deep, so unless you’re looking at the skin or the eye, you’re going to have to develop an endoscope to be able to view these hallmarks.”
OCT images the tissue microstructure, generally in a resolution of 2-20 microns, at a depth of 1-2 mm, and it measures reflected light. When possible, Dr. Barton combines OCT with laser-induced fluorescence for enhanced accuracy of detection of cancer. Induced fluorescence senses molecular information with the natural fluorophores in the body or with targeted exogenous agents. Then there’s multiphoton microscopy, an advanced imaging technique that enables clinicians to view cellular and subcellular events within living tissue. Early models of this technology “took up entire benches” in physics labs, Dr. Barton said, but she and other investigators are designing smaller devices for use in clinics. “This is exciting, because not only do we [view] subcellular structure with this modality, but it can also be highly sensitive to collagen structure,” she said.
Ovarian cancer model
In a model of ovarian cancer, she and colleagues externalized the ovaries of a mouse, imaged the organs, put them back in, and reassessed them at 8 weeks. “This model develops cancer very quickly,” said Dr. Barton, who once worked for McDonnell Douglas on the Space Station program. At 8 weeks, using fluorescence and targeted agents with a tabletop multiphoton microscopy system, they observed that the proliferation signals of cancer had begun. “So, with an agent targeted to the folate receptor or to other receptors that are implicated in cancer development, we can see that ovaries and fallopian tubes are lighting up,” she said.
With proof of concept established with the mouse study, she and other researchers are drawing from technological advances to create tiny laser systems for use in the clinic to image a variety of structures in the human body. Optics advances include bulk optics and all-fiber designs where engineers can create an imaging probe that’s only 125 microns in diameter, “or maybe even as small as 70 microns in diameter,” she said. “We can do fabrications on the tips of endoscopes to redirect the light and focus it. We can also do 3-D printing and spiral scanning to create miniature devices to make new advances. That means that instead of just white light imaging of the colon or the lung like we have had in the past, we can start moving into smaller structures, such as the eustachian tube, the fallopian tube, the bile ducts, or making miniature devices for brain biopsies, lung biopsies, and maybe being able to get into bronchioles and arterioles.”
According to Dr. Barton, prior research has demonstrated that cerebral vasculature can be imaged with a catheter 400 microns in diameter, the spaces in the lungs can be imaged with a needle that is 310 microns in diameter, and the inner structures of the eustachian tube can be viewed with an endoscope 1 mm in diameter.
She and her colleagues are developing an OCT/fluorescence imaging falloposcope that is 0.8 mm in diameter, flexible, and steerable, as a tool for early detection of ovarian cancer in humans. “It’s now known that most ovarian cancer starts in the fallopian tubes,” Dr. Barton said. “It’s metastatic disease when those cells break off from the fallopian tubes and go to the ovaries. We wanted to create an imaging system where we created a fiber bundle that we could navigate with white light and with fluorescence so that we can see these early stages of cancer [and] how they fluoresce differently. We also wanted to have an OCT system so that we could image through the wall of the fallopian tube and look for that layer thickening and other precursors to ovarian cancer.”
To date, in vivo testing in healthy women has demonstrated that the miniature endoscope is able to reach the fallopian tubes through the natural orifice of the vagina and uterus. “That is pretty exciting,” she said. “The images may not be of the highest quality, but we are advancing.”
Dr. Barton reported having no relevant financial disclosures.
PHOENIX –
In a lecture during a multispecialty roundup of cutting-edge energy-based device applications at the annual conference of the American Society for Laser Medicine and Surgery, Dr. Barton, a biomedical engineer who directs the BIO5 Institute at the University of Arizona, Tucson, said that while no current modality exists to enable physicians in dermatology and other specialties to view internal structures throughout the entire body with cellular resolution, refining existing technologies is a good way to start.
In 2011, renowned cancer researchers Douglas Hanahan, PhD, and Robert A. Weinberg, PhD, proposed six hallmarks of cancer, which include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Each hallmark poses unique imaging challenges. For example, enabling replicative immortality “means that the cell nuclei change size and shape; they change their position,” said Dr. Barton, who is also professor of biomedical engineering and optical sciences at the university. “If we want to see that, we’re going to need an imaging modality that’s subcellular in resolution.”
Similarly, if clinicians want to view how proliferative signaling is changing, “that means being able to visualize the cell surface receptors; those are even smaller to actually visualize,” she said. “But we have technologies where we can target those receptors with fluorophores. And then we can look at large areas very quickly.” Meanwhile, the ability of cancer cells to resist cell death and evade growth suppressors often results in thickening of epithelium throughout the body. “So, if we can measure the thickness of the epithelium, we can see that there’s something wrong with that tissue,” she said.
As for cancer’s propensity for invasion and metastasis, “here, we’re looking at how the collagen structure [between the cells] has changed and whether there’s layer breakdown or not. Optical imaging can detect cancer. However, high resolution optical techniques can only image about 1 mm deep, so unless you’re looking at the skin or the eye, you’re going to have to develop an endoscope to be able to view these hallmarks.”
OCT images the tissue microstructure, generally in a resolution of 2-20 microns, at a depth of 1-2 mm, and it measures reflected light. When possible, Dr. Barton combines OCT with laser-induced fluorescence for enhanced accuracy of detection of cancer. Induced fluorescence senses molecular information with the natural fluorophores in the body or with targeted exogenous agents. Then there’s multiphoton microscopy, an advanced imaging technique that enables clinicians to view cellular and subcellular events within living tissue. Early models of this technology “took up entire benches” in physics labs, Dr. Barton said, but she and other investigators are designing smaller devices for use in clinics. “This is exciting, because not only do we [view] subcellular structure with this modality, but it can also be highly sensitive to collagen structure,” she said.
Ovarian cancer model
In a model of ovarian cancer, she and colleagues externalized the ovaries of a mouse, imaged the organs, put them back in, and reassessed them at 8 weeks. “This model develops cancer very quickly,” said Dr. Barton, who once worked for McDonnell Douglas on the Space Station program. At 8 weeks, using fluorescence and targeted agents with a tabletop multiphoton microscopy system, they observed that the proliferation signals of cancer had begun. “So, with an agent targeted to the folate receptor or to other receptors that are implicated in cancer development, we can see that ovaries and fallopian tubes are lighting up,” she said.
With proof of concept established with the mouse study, she and other researchers are drawing from technological advances to create tiny laser systems for use in the clinic to image a variety of structures in the human body. Optics advances include bulk optics and all-fiber designs where engineers can create an imaging probe that’s only 125 microns in diameter, “or maybe even as small as 70 microns in diameter,” she said. “We can do fabrications on the tips of endoscopes to redirect the light and focus it. We can also do 3-D printing and spiral scanning to create miniature devices to make new advances. That means that instead of just white light imaging of the colon or the lung like we have had in the past, we can start moving into smaller structures, such as the eustachian tube, the fallopian tube, the bile ducts, or making miniature devices for brain biopsies, lung biopsies, and maybe being able to get into bronchioles and arterioles.”
According to Dr. Barton, prior research has demonstrated that cerebral vasculature can be imaged with a catheter 400 microns in diameter, the spaces in the lungs can be imaged with a needle that is 310 microns in diameter, and the inner structures of the eustachian tube can be viewed with an endoscope 1 mm in diameter.
She and her colleagues are developing an OCT/fluorescence imaging falloposcope that is 0.8 mm in diameter, flexible, and steerable, as a tool for early detection of ovarian cancer in humans. “It’s now known that most ovarian cancer starts in the fallopian tubes,” Dr. Barton said. “It’s metastatic disease when those cells break off from the fallopian tubes and go to the ovaries. We wanted to create an imaging system where we created a fiber bundle that we could navigate with white light and with fluorescence so that we can see these early stages of cancer [and] how they fluoresce differently. We also wanted to have an OCT system so that we could image through the wall of the fallopian tube and look for that layer thickening and other precursors to ovarian cancer.”
To date, in vivo testing in healthy women has demonstrated that the miniature endoscope is able to reach the fallopian tubes through the natural orifice of the vagina and uterus. “That is pretty exciting,” she said. “The images may not be of the highest quality, but we are advancing.”
Dr. Barton reported having no relevant financial disclosures.
AT ASLMS 2023