User login
Hypermucoviscous K pneumoniae Shows Reduced Drug Resistance
TOPLINE:
Hypermucoviscous Klebsiella pneumoniae (hmKp) strains demonstrate a significantly lower prevalence of extended-spectrum beta-lactamase (ESBL) production and slightly lower carbapenem resistance than non-hmKp strains, according to a recent meta-analysis of 2049 clinical isolates.
METHODOLOGY:
- Researchers conducted a meta-analysis to assess the prevalence of ESBL-producing strains and carbapenem-resistant strains among the hmKp and non-hmKp clinical isolates.
- They included 15 studies published between 2014 and 2023, with 2049 clinical isolates of K pneumoniae identified using a string test to distinguish hypermucoviscous from non-hypermucoviscous strains.
- These studies spanned across four continents: Asia, Africa, Europe, and North America.
- The primary outcome was the prevalence of ESBL-producing and carbapenem-resistant strains, determined through antimicrobial susceptibility testing.
TAKEAWAY:
- The hmKp strains were associated with a significantly lower prevalence of ESBL-producing strains than non-hmKp strains (pooled odds ratio [OR], 0.26; P = .003).
- Similarly, hmKp strains were associated with a slightly lower prevalence of carbapenem-resistant strains than non-hmKp strains (pooled OR, 0.63; P = .038).
IN PRACTICE:
“Therapeutic options for CRKP [carbapenem-resistant K pneumoniae] infections are extremely limited due to the scarcity of effective antibacterial drugs. Therefore, it is crucial to consider the risks posed by CRKP strains when administering treatment to patients with hmKp infections and a history of the aforementioned risk factors,” the authors wrote.
SOURCE:
The study was led by Hiroki Namikawa, Department of Medical Education and General Practice, Graduate School of Medicine, Osaka Metropolitan University, Japan. It was published online on December 16, 2024, in Emerging Microbes & Infections.
LIMITATIONS:
Only three databases (PubMed, Scopus, and Cochrane Library) were searched for identifying studies, potentially missing relevant studies from other sources. Furthermore, only articles published in English were included, which may have restricted the scope of analysis. Additionally, geographical distribution was predominantly limited to Asia, limiting the global applicability of the results.
DISCLOSURES:
No funding sources were mentioned, and no conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Hypermucoviscous Klebsiella pneumoniae (hmKp) strains demonstrate a significantly lower prevalence of extended-spectrum beta-lactamase (ESBL) production and slightly lower carbapenem resistance than non-hmKp strains, according to a recent meta-analysis of 2049 clinical isolates.
METHODOLOGY:
- Researchers conducted a meta-analysis to assess the prevalence of ESBL-producing strains and carbapenem-resistant strains among the hmKp and non-hmKp clinical isolates.
- They included 15 studies published between 2014 and 2023, with 2049 clinical isolates of K pneumoniae identified using a string test to distinguish hypermucoviscous from non-hypermucoviscous strains.
- These studies spanned across four continents: Asia, Africa, Europe, and North America.
- The primary outcome was the prevalence of ESBL-producing and carbapenem-resistant strains, determined through antimicrobial susceptibility testing.
TAKEAWAY:
- The hmKp strains were associated with a significantly lower prevalence of ESBL-producing strains than non-hmKp strains (pooled odds ratio [OR], 0.26; P = .003).
- Similarly, hmKp strains were associated with a slightly lower prevalence of carbapenem-resistant strains than non-hmKp strains (pooled OR, 0.63; P = .038).
IN PRACTICE:
“Therapeutic options for CRKP [carbapenem-resistant K pneumoniae] infections are extremely limited due to the scarcity of effective antibacterial drugs. Therefore, it is crucial to consider the risks posed by CRKP strains when administering treatment to patients with hmKp infections and a history of the aforementioned risk factors,” the authors wrote.
SOURCE:
The study was led by Hiroki Namikawa, Department of Medical Education and General Practice, Graduate School of Medicine, Osaka Metropolitan University, Japan. It was published online on December 16, 2024, in Emerging Microbes & Infections.
LIMITATIONS:
Only three databases (PubMed, Scopus, and Cochrane Library) were searched for identifying studies, potentially missing relevant studies from other sources. Furthermore, only articles published in English were included, which may have restricted the scope of analysis. Additionally, geographical distribution was predominantly limited to Asia, limiting the global applicability of the results.
DISCLOSURES:
No funding sources were mentioned, and no conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Hypermucoviscous Klebsiella pneumoniae (hmKp) strains demonstrate a significantly lower prevalence of extended-spectrum beta-lactamase (ESBL) production and slightly lower carbapenem resistance than non-hmKp strains, according to a recent meta-analysis of 2049 clinical isolates.
METHODOLOGY:
- Researchers conducted a meta-analysis to assess the prevalence of ESBL-producing strains and carbapenem-resistant strains among the hmKp and non-hmKp clinical isolates.
- They included 15 studies published between 2014 and 2023, with 2049 clinical isolates of K pneumoniae identified using a string test to distinguish hypermucoviscous from non-hypermucoviscous strains.
- These studies spanned across four continents: Asia, Africa, Europe, and North America.
- The primary outcome was the prevalence of ESBL-producing and carbapenem-resistant strains, determined through antimicrobial susceptibility testing.
TAKEAWAY:
- The hmKp strains were associated with a significantly lower prevalence of ESBL-producing strains than non-hmKp strains (pooled odds ratio [OR], 0.26; P = .003).
- Similarly, hmKp strains were associated with a slightly lower prevalence of carbapenem-resistant strains than non-hmKp strains (pooled OR, 0.63; P = .038).
IN PRACTICE:
“Therapeutic options for CRKP [carbapenem-resistant K pneumoniae] infections are extremely limited due to the scarcity of effective antibacterial drugs. Therefore, it is crucial to consider the risks posed by CRKP strains when administering treatment to patients with hmKp infections and a history of the aforementioned risk factors,” the authors wrote.
SOURCE:
The study was led by Hiroki Namikawa, Department of Medical Education and General Practice, Graduate School of Medicine, Osaka Metropolitan University, Japan. It was published online on December 16, 2024, in Emerging Microbes & Infections.
LIMITATIONS:
Only three databases (PubMed, Scopus, and Cochrane Library) were searched for identifying studies, potentially missing relevant studies from other sources. Furthermore, only articles published in English were included, which may have restricted the scope of analysis. Additionally, geographical distribution was predominantly limited to Asia, limiting the global applicability of the results.
DISCLOSURES:
No funding sources were mentioned, and no conflicts of interest were reported.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
First Human Bird Flu Death Confirmed in US; Overall Risk Remains Low
The first human patient in the United States with a confirmed case of avian influenza has died, according to a press release from the Louisiana Department of Health. The individual was older than 65 years and had underlying medical conditions and remains the only known human case in the state.
The patient contracted highly pathogenic avian influenza, also known as H5N1, through exposure to wild birds and a noncommercial backyard flock, according to the release.
The Centers for Disease Control and Prevention (CDC) conducted genetic sequencing of specimens of the virus collected from the Louisiana patient. The agency compared the sequences with sequences from dairy cows, wild birds, and poultry in various areas of the United States that were infected with the H5N1 virus.
The Louisiana patient was infected with the D1.1 genotype of the H5N1 virus. Although D1.1 is related to other D1.1 viruses found in recent human cases in Washington State and British Columbia, Canada, it is distinct from the widely spreading B3.13 genotype that has caused H5N1 outbreaks in dairy cows, poultry, and other animals and has been linked to sporadic human cases in the United States, according to the CDC.
Despite evidence of some changes in the virus between the Louisiana patient and samples from poultry on the patient’s property, “these changes would be more concerning if found in animal hosts or in early stages of infection,” according to the CDC. The CDC and the Louisiana Department of Health are conducting additional sequencing to facilitate further analysis.
In the meantime, the risk to the general public for H5N1 remains low, but individuals who work with or have recreational exposure to birds, poultry, or cows remain at increased risk.
The CDC and the Louisiana Department of Health advise individuals to reduce the risk for H5N1 exposure by avoiding direct contact with wild birds or other animals infected or possibly infected with the virus, avoiding any contact with dead animals, and keeping pets away from sick or dead animals and their feces. Additional safety measures include avoiding uncooked food products such as unpasteurized raw milk or cheese from animals with suspected or confirmed infections and reporting sick or dead birds or animals to the US Department of Agriculture by calling 1-866-536-7593 or the Louisiana Department of Agriculture and Forestry Diagnostic Lab by calling 318-927-3441.
The CDC advises clinicians to consider H5N1 in patients presenting with conjunctivitis or signs of acute respiratory illness and a history of high-risk exposure, including handling sick or dead animals, notably birds and livestock, within 10 days before the onset of symptoms. Other risk factors include consuming uncooked or undercooked food, direct contact with areas contaminated with feces, direct contact with unpasteurized milk or other dairy products or with parts of potentially infected animals, and prolonged exposure to infected animals in a confined space.
Clinical symptoms also may include gastrointestinal complaints such as diarrhea, as well as fatigue, arthralgia, and headache. Patients with more severe H5N1 may experience shortness of breath, altered mental state, and seizures, and serious complications of the virus include pneumonia, acute respiratory distress syndrome, multiorgan failure, and sepsis, according to the CDC.
Clinicians who suspect H5N1 cases should contact their local public health departments. The CDC offers additional advice on evaluating and managing patients with novel influenza A viruses.
A Clinician’s Take
“Some symptoms of avian flu include fever, cough, sore throat, runny nose, fatigue, body aches or eye redness or irritation,” Shirin A. Mazumder, MD, associate professor and infectious disease specialist at The University of Tennessee Health Science Center, Memphis, said in an interview. “The timing to the development of symptoms after exposure is typically within 10 days. Avian influenza should be considered when individuals develop symptoms with a relevant exposure history,” she said.
Whenever possible, avoidance of sick or dead birds and other animals is ideal, but for those who must have contact with sick or dead birds, poultry, or other animals, personal protective equipment (PPE) including a respirator, goggles, and disposable gloves is recommended, said Mazumder.
“For those working in high-exposure settings, additional PPE including boots or boot covers, hair cover, and fluid-resistant coveralls are recommended,” she said. “Other protective measures include avoiding touching surfaces or materials contaminated with feces, mucus, and saliva from infected animals and avoid[ing] the consumption of raw milk, raw milk products, and undercooked meat from infected animals,” she added.
Hunters handling wild game should dress birds in the field, practice good hand hygiene, and use a respirator or well-fitting mask and gloves when handling the animals to help prevent disease, said Mazumder.
In addition, those working with confirmed or suspected H5N1 cases should monitor themselves for symptoms, said Mazumder. “Those who become ill within 10 days of exposure to an infected animal or source should isolate from household members and avoid going to work or school until infection is excluded. It is important to reach out to a healthcare professional if you think you may have been exposed or if you think you are infected,” she said.
There is no currently available vaccine for H5N1 infection, but oseltamivir can be used for chemoprophylaxis and treatment, said Mazumder. “The seasonal flu vaccine does not protect against avian influenza; however, it is still important to ensure that you are up to date on the latest flu vaccine to prevent the possibility of a coinfection with seasonal flu and avian flu,” she emphasized.
More research is needed to better understand how the influenza virus is transmitted, said Mazumder. “The potential for the virus to evolve and mutate, and how it affects different hosts, are all factors that can impact public health decisions,” she said. “In addition, further research into finding a vaccine and improving surveillance methods are necessary for disease prevention,” she said.
Mazumder had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
The first human patient in the United States with a confirmed case of avian influenza has died, according to a press release from the Louisiana Department of Health. The individual was older than 65 years and had underlying medical conditions and remains the only known human case in the state.
The patient contracted highly pathogenic avian influenza, also known as H5N1, through exposure to wild birds and a noncommercial backyard flock, according to the release.
The Centers for Disease Control and Prevention (CDC) conducted genetic sequencing of specimens of the virus collected from the Louisiana patient. The agency compared the sequences with sequences from dairy cows, wild birds, and poultry in various areas of the United States that were infected with the H5N1 virus.
The Louisiana patient was infected with the D1.1 genotype of the H5N1 virus. Although D1.1 is related to other D1.1 viruses found in recent human cases in Washington State and British Columbia, Canada, it is distinct from the widely spreading B3.13 genotype that has caused H5N1 outbreaks in dairy cows, poultry, and other animals and has been linked to sporadic human cases in the United States, according to the CDC.
Despite evidence of some changes in the virus between the Louisiana patient and samples from poultry on the patient’s property, “these changes would be more concerning if found in animal hosts or in early stages of infection,” according to the CDC. The CDC and the Louisiana Department of Health are conducting additional sequencing to facilitate further analysis.
In the meantime, the risk to the general public for H5N1 remains low, but individuals who work with or have recreational exposure to birds, poultry, or cows remain at increased risk.
The CDC and the Louisiana Department of Health advise individuals to reduce the risk for H5N1 exposure by avoiding direct contact with wild birds or other animals infected or possibly infected with the virus, avoiding any contact with dead animals, and keeping pets away from sick or dead animals and their feces. Additional safety measures include avoiding uncooked food products such as unpasteurized raw milk or cheese from animals with suspected or confirmed infections and reporting sick or dead birds or animals to the US Department of Agriculture by calling 1-866-536-7593 or the Louisiana Department of Agriculture and Forestry Diagnostic Lab by calling 318-927-3441.
The CDC advises clinicians to consider H5N1 in patients presenting with conjunctivitis or signs of acute respiratory illness and a history of high-risk exposure, including handling sick or dead animals, notably birds and livestock, within 10 days before the onset of symptoms. Other risk factors include consuming uncooked or undercooked food, direct contact with areas contaminated with feces, direct contact with unpasteurized milk or other dairy products or with parts of potentially infected animals, and prolonged exposure to infected animals in a confined space.
Clinical symptoms also may include gastrointestinal complaints such as diarrhea, as well as fatigue, arthralgia, and headache. Patients with more severe H5N1 may experience shortness of breath, altered mental state, and seizures, and serious complications of the virus include pneumonia, acute respiratory distress syndrome, multiorgan failure, and sepsis, according to the CDC.
Clinicians who suspect H5N1 cases should contact their local public health departments. The CDC offers additional advice on evaluating and managing patients with novel influenza A viruses.
A Clinician’s Take
“Some symptoms of avian flu include fever, cough, sore throat, runny nose, fatigue, body aches or eye redness or irritation,” Shirin A. Mazumder, MD, associate professor and infectious disease specialist at The University of Tennessee Health Science Center, Memphis, said in an interview. “The timing to the development of symptoms after exposure is typically within 10 days. Avian influenza should be considered when individuals develop symptoms with a relevant exposure history,” she said.
Whenever possible, avoidance of sick or dead birds and other animals is ideal, but for those who must have contact with sick or dead birds, poultry, or other animals, personal protective equipment (PPE) including a respirator, goggles, and disposable gloves is recommended, said Mazumder.
“For those working in high-exposure settings, additional PPE including boots or boot covers, hair cover, and fluid-resistant coveralls are recommended,” she said. “Other protective measures include avoiding touching surfaces or materials contaminated with feces, mucus, and saliva from infected animals and avoid[ing] the consumption of raw milk, raw milk products, and undercooked meat from infected animals,” she added.
Hunters handling wild game should dress birds in the field, practice good hand hygiene, and use a respirator or well-fitting mask and gloves when handling the animals to help prevent disease, said Mazumder.
In addition, those working with confirmed or suspected H5N1 cases should monitor themselves for symptoms, said Mazumder. “Those who become ill within 10 days of exposure to an infected animal or source should isolate from household members and avoid going to work or school until infection is excluded. It is important to reach out to a healthcare professional if you think you may have been exposed or if you think you are infected,” she said.
There is no currently available vaccine for H5N1 infection, but oseltamivir can be used for chemoprophylaxis and treatment, said Mazumder. “The seasonal flu vaccine does not protect against avian influenza; however, it is still important to ensure that you are up to date on the latest flu vaccine to prevent the possibility of a coinfection with seasonal flu and avian flu,” she emphasized.
More research is needed to better understand how the influenza virus is transmitted, said Mazumder. “The potential for the virus to evolve and mutate, and how it affects different hosts, are all factors that can impact public health decisions,” she said. “In addition, further research into finding a vaccine and improving surveillance methods are necessary for disease prevention,” she said.
Mazumder had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
The first human patient in the United States with a confirmed case of avian influenza has died, according to a press release from the Louisiana Department of Health. The individual was older than 65 years and had underlying medical conditions and remains the only known human case in the state.
The patient contracted highly pathogenic avian influenza, also known as H5N1, through exposure to wild birds and a noncommercial backyard flock, according to the release.
The Centers for Disease Control and Prevention (CDC) conducted genetic sequencing of specimens of the virus collected from the Louisiana patient. The agency compared the sequences with sequences from dairy cows, wild birds, and poultry in various areas of the United States that were infected with the H5N1 virus.
The Louisiana patient was infected with the D1.1 genotype of the H5N1 virus. Although D1.1 is related to other D1.1 viruses found in recent human cases in Washington State and British Columbia, Canada, it is distinct from the widely spreading B3.13 genotype that has caused H5N1 outbreaks in dairy cows, poultry, and other animals and has been linked to sporadic human cases in the United States, according to the CDC.
Despite evidence of some changes in the virus between the Louisiana patient and samples from poultry on the patient’s property, “these changes would be more concerning if found in animal hosts or in early stages of infection,” according to the CDC. The CDC and the Louisiana Department of Health are conducting additional sequencing to facilitate further analysis.
In the meantime, the risk to the general public for H5N1 remains low, but individuals who work with or have recreational exposure to birds, poultry, or cows remain at increased risk.
The CDC and the Louisiana Department of Health advise individuals to reduce the risk for H5N1 exposure by avoiding direct contact with wild birds or other animals infected or possibly infected with the virus, avoiding any contact with dead animals, and keeping pets away from sick or dead animals and their feces. Additional safety measures include avoiding uncooked food products such as unpasteurized raw milk or cheese from animals with suspected or confirmed infections and reporting sick or dead birds or animals to the US Department of Agriculture by calling 1-866-536-7593 or the Louisiana Department of Agriculture and Forestry Diagnostic Lab by calling 318-927-3441.
The CDC advises clinicians to consider H5N1 in patients presenting with conjunctivitis or signs of acute respiratory illness and a history of high-risk exposure, including handling sick or dead animals, notably birds and livestock, within 10 days before the onset of symptoms. Other risk factors include consuming uncooked or undercooked food, direct contact with areas contaminated with feces, direct contact with unpasteurized milk or other dairy products or with parts of potentially infected animals, and prolonged exposure to infected animals in a confined space.
Clinical symptoms also may include gastrointestinal complaints such as diarrhea, as well as fatigue, arthralgia, and headache. Patients with more severe H5N1 may experience shortness of breath, altered mental state, and seizures, and serious complications of the virus include pneumonia, acute respiratory distress syndrome, multiorgan failure, and sepsis, according to the CDC.
Clinicians who suspect H5N1 cases should contact their local public health departments. The CDC offers additional advice on evaluating and managing patients with novel influenza A viruses.
A Clinician’s Take
“Some symptoms of avian flu include fever, cough, sore throat, runny nose, fatigue, body aches or eye redness or irritation,” Shirin A. Mazumder, MD, associate professor and infectious disease specialist at The University of Tennessee Health Science Center, Memphis, said in an interview. “The timing to the development of symptoms after exposure is typically within 10 days. Avian influenza should be considered when individuals develop symptoms with a relevant exposure history,” she said.
Whenever possible, avoidance of sick or dead birds and other animals is ideal, but for those who must have contact with sick or dead birds, poultry, or other animals, personal protective equipment (PPE) including a respirator, goggles, and disposable gloves is recommended, said Mazumder.
“For those working in high-exposure settings, additional PPE including boots or boot covers, hair cover, and fluid-resistant coveralls are recommended,” she said. “Other protective measures include avoiding touching surfaces or materials contaminated with feces, mucus, and saliva from infected animals and avoid[ing] the consumption of raw milk, raw milk products, and undercooked meat from infected animals,” she added.
Hunters handling wild game should dress birds in the field, practice good hand hygiene, and use a respirator or well-fitting mask and gloves when handling the animals to help prevent disease, said Mazumder.
In addition, those working with confirmed or suspected H5N1 cases should monitor themselves for symptoms, said Mazumder. “Those who become ill within 10 days of exposure to an infected animal or source should isolate from household members and avoid going to work or school until infection is excluded. It is important to reach out to a healthcare professional if you think you may have been exposed or if you think you are infected,” she said.
There is no currently available vaccine for H5N1 infection, but oseltamivir can be used for chemoprophylaxis and treatment, said Mazumder. “The seasonal flu vaccine does not protect against avian influenza; however, it is still important to ensure that you are up to date on the latest flu vaccine to prevent the possibility of a coinfection with seasonal flu and avian flu,” she emphasized.
More research is needed to better understand how the influenza virus is transmitted, said Mazumder. “The potential for the virus to evolve and mutate, and how it affects different hosts, are all factors that can impact public health decisions,” she said. “In addition, further research into finding a vaccine and improving surveillance methods are necessary for disease prevention,” she said.
Mazumder had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Group A Streptococcal Pharyngitis Diagnosis
It’s wintertime, peak season for GAS pharyngitis, and you’d think that this far into the 21st century we would have a foolproof process for diagnosing which among the many patients with pharyngitis have true GAS pharyngitis. Thinking back to the 1980s, we have come a long way from simple throat cultures for detecting GAS, e.g., numerous point of care (POC) Clinical Laboratory Improvement Amendments (CLIA), waved rapid antigen detection tests (RADT), and numerous highly sensitive molecular assays, e.g. nucleic acid amplification tests (NAAT). But if you think the issues surrounding management of GAS pharyngitis have been solved by these newer tests, think again.
Several good reviews1-3 are excellent resources for those wishing a refresher on GAS diagnosis/management issues. They present nitty gritty details on comparative advantages/disadvantages of the many testing options while reminding us of the nuts and bolts of GAS pharyngitis. The following are a few nuggets from these articles.
Properly collected throat specimen. A quality throat specimen involves swabbing both tonsillar pillars plus posterior pharynx without touching tongue or inner cheeks. Two swab collections increase sensitivity by almost 10% compared with a single swab. Transport media is preferred if samples will not be cultured within 24 hours. Caveat: RADT testing of a transport media-diluted sample lowers sensitivity compared with direct swab use.
Reliable GAS detection. Commercially available tests in 2025 are well studied. Culture is considered a gold standard for detecting clinically relevant GAS by CDC.4 Culture has good sensitivity (estimated 80%-90% varying among studies and by quality of specimens) and 99% specificity but requires 16-24 hours for results. RADT solves the time-delay issues and has near 100% specificity but sensitivity used to be as low as 65%, hence the 2012 Infectious Diseases Society of America guideline recommendation for backup throat culture for negative tests.5 However, current RADT have sensitivities in the 85%-90% range.3,4 So a positive RADT reliably and quickly indicates GAS antigens are present. NAAT have the highest combined sensitivity and specificity, near 100% for each, and a positive reliably indicates GAS nucleic acids are present.
So why not simply always use NAAT? First, it’s a “be careful what you wish for” scenario. NAAT can, and do, detect dead remnants and colonizing GAS way more than culture.2,3 So NAAT are overly sensitive, adding an extra layer of interpretation difficulty, ie, as many as 20% of positive NAAT detections may be carriers or dead GAS. Second, NAAT often requires special instrumentation and kits are more expensive. That said, reimbursement is often higher for NAAT.
Choice based on accuracy in detecting GAS. If time delays were not a problem, culture would still seem the answer. If more rapid detection is needed, either RADT with culture back up or NAAT could be the answer. That said, consider that in the real world, throat cultures are less sensitive and RADT are less specific than indicated by some published data.6 So, the ideal answer, it seems, would be NAAT GAS detection coupled with a confirmatory biomarker of GAS infection. Such innate immune biomarkers may be on the horizon.3
But first, pretest screening. In 2025 what do we do with a positive result? Do we prescribe antibiotics? Do we think the detected GAS bacteria/antigens/nucleic acids represent the cause of the pharyngitis? Or did we just detect dead GAS or even a carrier, while a virus is the true cause? Challenges for this decision include most pharyngitis (up to 70%) being due to viruses, not GAS, plus up to 20% of GAS detections even by less sensitive culture or RADT can be carriers, plus an added 10%-20% of RADT and NAAT detections are dead GAS. Thus, with indiscriminate testing of all pharyngitis patients, the number of truly positive GAS detections that are actually “false positives” (GAS in some form is present but not causing pharyngitis) may be almost as high as for those representing true GAS pharyngitis.
Some tool is needed to minimize testing patients who are likely to have viral pharyngitis to reduce test-positive/GAS-pharyngitis-negative scenarios. Pretest patient screening therefore is critical to increase the positive predictive value of positive GAS testing results. The history and physical can be helpful. In the simplest form of pretest screening, eliminate those younger than 3 years old* or those with viral type sign/symptoms, eg conjunctivitis, cough, coryza.7 This could cut “false” positives by as much as a half. More complete validated scoring systems are also available but remain imperfect. The most published is the McIsaac score (modified Centor score).3-5,8 (See Table and Figure.)
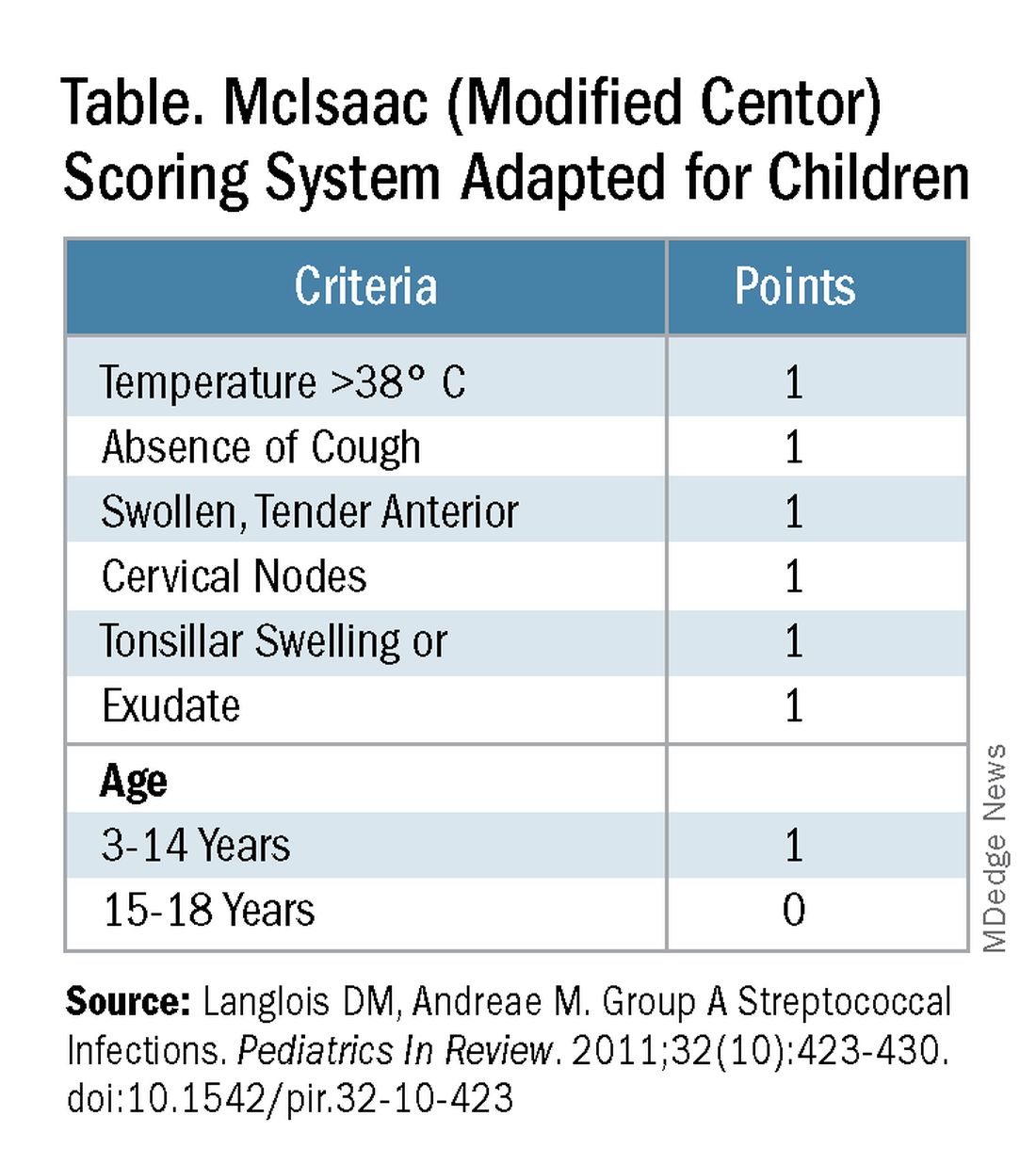
However, even with this validated scoring system, misdiagnoses and some antibiotic misuse will likely occur, particularly if the controversial option to treat a patient with a score above 4 without testing is used. For example, a 2004 study in patients older than 3 years old revealed that 45% with a score above 4 points did not have GAS pharyngitis. (McIsaac et al.) A 2012 study showed similar potential overdiagnosis from using the score without testing (45% with > 4 points did not have GAS pharyngitis). Of note, clinical scores of below 2 comprised up to 10% and would be neither tested nor treated. (Figure.)
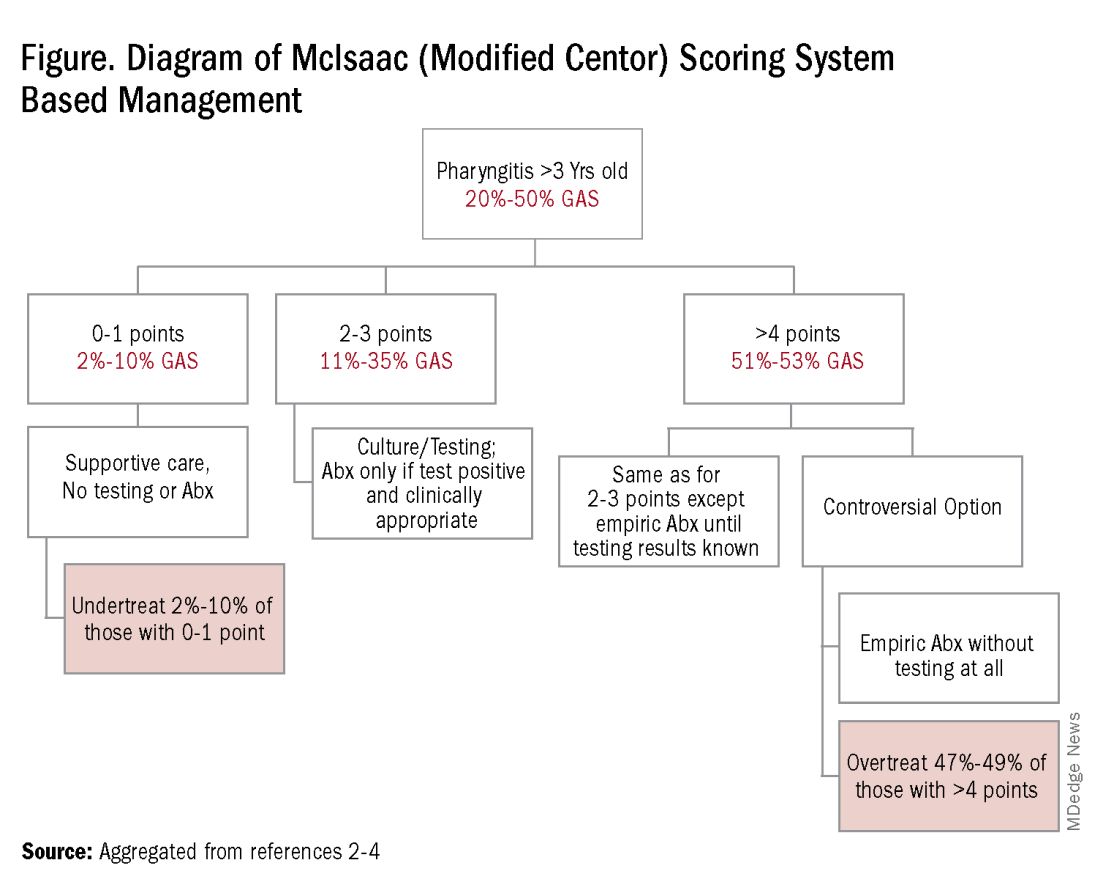
Best clinical judgment. Regardless of the chosen test, we still need to interpret positive results, ie, use best clinical judgment. We know that even with pretest screening some positives tests will represent carriers or nonviable GAS. Yet true GAS pharyngitis needs antibiotic treatment to minimize nonpyogenic and pyogenic complications, plus reduce contagion/transmission risk and days of illness. Thus, we are forced to use best clinical judgment when considering if what could be GAS pharyngitis, particularly exudative pharyngitis, could actually be due to EBV, adenovirus, or gonococcus, each of which can mimic GAS findings. Differentiating these requires discussion beyond the scope of this article, but clues are often found in the history, the patient’s age, associated symptoms and distribution of tonsillopharyngeal exudate. Likewise Group C and G streptococcal pharyngitis can mimic GAS. Note: A comprehensive throat culture can identify these streptococci but requires a special order and likely a call to the laboratory.
Summary: The age-old problem persists, ie, differentiating the minority (~30%) of pharyngitis cases needing antibiotics from the majority that do not. We all wish to promptly treat true GAS pharyngitis; however our current tools remain imperfect. That said, we should strive to correctly diagnose/manage as many patients with pharyngitis as possible. I, for one, can’t wait until we get a validated biomarker that confirms GAS as the culprit in pharyngitis episodes. In the meantime, most providers likely have clinic or hospital approved pathways for managing GAS pharyngitis, many of which are at least in part based on data from sources for this discussion. If not, a firm foundation for creating one can be found in sources among the reference list below. Finally, if you think such pathways somehow interfere with patient flow, consider that a busy multi-provider private practice successfully integrated pretest screening and a pathway while maintaining patient flow and improving antibiotic stewardship.7
*Focal pharyngotonsillar GAS infection is rare in children younger than 3 years old, when GAS nasal passage infection may manifest as streptococcosis.9
Dr Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He has no relevant financial disclosures. Email him at [email protected].
References
1. Bannerjee D, Selvarangan RS. The Evolution of Group A Streptococcus Pharyngitis Testing. Association for Diagnostics and Laboratory Medicine, 2018, Sep 1.
2. Cohen JF et al. Group A Streptococcus Pharyngitis in Children: New Perspectives on Rapid Diagnostic Testing and Antimicrobial Stewardship. J Pediatric Infect Dis Soc. 2024 Apr 24;13(4):250-256. doi: 10.1093/jpids/piae0223.
3. Boyanton Jr BL et al. Current Laboratory and Point-of-Care Pharyngitis Diagnostic Testing and Knowledge Gaps. J Infect Dis. 2024 Oct 23;230(Suppl 3):S182–S189. doi: 10.1093/infdis/jiae415.
4. Group A Strep Infection. Centers for Disease Control and Prevention, 2024, Mar 1.
5. Shulman ST et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2012 Nov 15;55(10):e86-102. doi: 10.1093/cid/cis629.
6. Rao A et al. Diagnosis and Antibiotic Treatment of Group A Streptococcal Pharyngitis in Children in a Primary Care Setting: Impact of Point-of-Care Polymerase Chain Reaction. BMC Pediatr. 2019 Jan 16;19(1):24. doi: 10.1186/s12887-019-1393-y.
7. Norton LE et al. Improving Guideline-Based Streptococcal Pharyngitis Testing: A Quality Improvement Initiative. Pediatrics. 2018 Jul;142(1):e20172033. doi: 10.1542/peds.2017-2033.
8. MD+ Calc website. Centor Score (Modified/McIsaac) for Strep Pharyngitis.
9. Langlois DM, Andreae M. Group A Streptococcal Infections. Pediatr Rev. 2011 Oct;32(10):423-9; quiz 430. doi: 10.1542/pir.32-10-423.
It’s wintertime, peak season for GAS pharyngitis, and you’d think that this far into the 21st century we would have a foolproof process for diagnosing which among the many patients with pharyngitis have true GAS pharyngitis. Thinking back to the 1980s, we have come a long way from simple throat cultures for detecting GAS, e.g., numerous point of care (POC) Clinical Laboratory Improvement Amendments (CLIA), waved rapid antigen detection tests (RADT), and numerous highly sensitive molecular assays, e.g. nucleic acid amplification tests (NAAT). But if you think the issues surrounding management of GAS pharyngitis have been solved by these newer tests, think again.
Several good reviews1-3 are excellent resources for those wishing a refresher on GAS diagnosis/management issues. They present nitty gritty details on comparative advantages/disadvantages of the many testing options while reminding us of the nuts and bolts of GAS pharyngitis. The following are a few nuggets from these articles.
Properly collected throat specimen. A quality throat specimen involves swabbing both tonsillar pillars plus posterior pharynx without touching tongue or inner cheeks. Two swab collections increase sensitivity by almost 10% compared with a single swab. Transport media is preferred if samples will not be cultured within 24 hours. Caveat: RADT testing of a transport media-diluted sample lowers sensitivity compared with direct swab use.
Reliable GAS detection. Commercially available tests in 2025 are well studied. Culture is considered a gold standard for detecting clinically relevant GAS by CDC.4 Culture has good sensitivity (estimated 80%-90% varying among studies and by quality of specimens) and 99% specificity but requires 16-24 hours for results. RADT solves the time-delay issues and has near 100% specificity but sensitivity used to be as low as 65%, hence the 2012 Infectious Diseases Society of America guideline recommendation for backup throat culture for negative tests.5 However, current RADT have sensitivities in the 85%-90% range.3,4 So a positive RADT reliably and quickly indicates GAS antigens are present. NAAT have the highest combined sensitivity and specificity, near 100% for each, and a positive reliably indicates GAS nucleic acids are present.
So why not simply always use NAAT? First, it’s a “be careful what you wish for” scenario. NAAT can, and do, detect dead remnants and colonizing GAS way more than culture.2,3 So NAAT are overly sensitive, adding an extra layer of interpretation difficulty, ie, as many as 20% of positive NAAT detections may be carriers or dead GAS. Second, NAAT often requires special instrumentation and kits are more expensive. That said, reimbursement is often higher for NAAT.
Choice based on accuracy in detecting GAS. If time delays were not a problem, culture would still seem the answer. If more rapid detection is needed, either RADT with culture back up or NAAT could be the answer. That said, consider that in the real world, throat cultures are less sensitive and RADT are less specific than indicated by some published data.6 So, the ideal answer, it seems, would be NAAT GAS detection coupled with a confirmatory biomarker of GAS infection. Such innate immune biomarkers may be on the horizon.3
But first, pretest screening. In 2025 what do we do with a positive result? Do we prescribe antibiotics? Do we think the detected GAS bacteria/antigens/nucleic acids represent the cause of the pharyngitis? Or did we just detect dead GAS or even a carrier, while a virus is the true cause? Challenges for this decision include most pharyngitis (up to 70%) being due to viruses, not GAS, plus up to 20% of GAS detections even by less sensitive culture or RADT can be carriers, plus an added 10%-20% of RADT and NAAT detections are dead GAS. Thus, with indiscriminate testing of all pharyngitis patients, the number of truly positive GAS detections that are actually “false positives” (GAS in some form is present but not causing pharyngitis) may be almost as high as for those representing true GAS pharyngitis.
Some tool is needed to minimize testing patients who are likely to have viral pharyngitis to reduce test-positive/GAS-pharyngitis-negative scenarios. Pretest patient screening therefore is critical to increase the positive predictive value of positive GAS testing results. The history and physical can be helpful. In the simplest form of pretest screening, eliminate those younger than 3 years old* or those with viral type sign/symptoms, eg conjunctivitis, cough, coryza.7 This could cut “false” positives by as much as a half. More complete validated scoring systems are also available but remain imperfect. The most published is the McIsaac score (modified Centor score).3-5,8 (See Table and Figure.)

However, even with this validated scoring system, misdiagnoses and some antibiotic misuse will likely occur, particularly if the controversial option to treat a patient with a score above 4 without testing is used. For example, a 2004 study in patients older than 3 years old revealed that 45% with a score above 4 points did not have GAS pharyngitis. (McIsaac et al.) A 2012 study showed similar potential overdiagnosis from using the score without testing (45% with > 4 points did not have GAS pharyngitis). Of note, clinical scores of below 2 comprised up to 10% and would be neither tested nor treated. (Figure.)

Best clinical judgment. Regardless of the chosen test, we still need to interpret positive results, ie, use best clinical judgment. We know that even with pretest screening some positives tests will represent carriers or nonviable GAS. Yet true GAS pharyngitis needs antibiotic treatment to minimize nonpyogenic and pyogenic complications, plus reduce contagion/transmission risk and days of illness. Thus, we are forced to use best clinical judgment when considering if what could be GAS pharyngitis, particularly exudative pharyngitis, could actually be due to EBV, adenovirus, or gonococcus, each of which can mimic GAS findings. Differentiating these requires discussion beyond the scope of this article, but clues are often found in the history, the patient’s age, associated symptoms and distribution of tonsillopharyngeal exudate. Likewise Group C and G streptococcal pharyngitis can mimic GAS. Note: A comprehensive throat culture can identify these streptococci but requires a special order and likely a call to the laboratory.
Summary: The age-old problem persists, ie, differentiating the minority (~30%) of pharyngitis cases needing antibiotics from the majority that do not. We all wish to promptly treat true GAS pharyngitis; however our current tools remain imperfect. That said, we should strive to correctly diagnose/manage as many patients with pharyngitis as possible. I, for one, can’t wait until we get a validated biomarker that confirms GAS as the culprit in pharyngitis episodes. In the meantime, most providers likely have clinic or hospital approved pathways for managing GAS pharyngitis, many of which are at least in part based on data from sources for this discussion. If not, a firm foundation for creating one can be found in sources among the reference list below. Finally, if you think such pathways somehow interfere with patient flow, consider that a busy multi-provider private practice successfully integrated pretest screening and a pathway while maintaining patient flow and improving antibiotic stewardship.7
*Focal pharyngotonsillar GAS infection is rare in children younger than 3 years old, when GAS nasal passage infection may manifest as streptococcosis.9
Dr Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He has no relevant financial disclosures. Email him at [email protected].
References
1. Bannerjee D, Selvarangan RS. The Evolution of Group A Streptococcus Pharyngitis Testing. Association for Diagnostics and Laboratory Medicine, 2018, Sep 1.
2. Cohen JF et al. Group A Streptococcus Pharyngitis in Children: New Perspectives on Rapid Diagnostic Testing and Antimicrobial Stewardship. J Pediatric Infect Dis Soc. 2024 Apr 24;13(4):250-256. doi: 10.1093/jpids/piae0223.
3. Boyanton Jr BL et al. Current Laboratory and Point-of-Care Pharyngitis Diagnostic Testing and Knowledge Gaps. J Infect Dis. 2024 Oct 23;230(Suppl 3):S182–S189. doi: 10.1093/infdis/jiae415.
4. Group A Strep Infection. Centers for Disease Control and Prevention, 2024, Mar 1.
5. Shulman ST et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2012 Nov 15;55(10):e86-102. doi: 10.1093/cid/cis629.
6. Rao A et al. Diagnosis and Antibiotic Treatment of Group A Streptococcal Pharyngitis in Children in a Primary Care Setting: Impact of Point-of-Care Polymerase Chain Reaction. BMC Pediatr. 2019 Jan 16;19(1):24. doi: 10.1186/s12887-019-1393-y.
7. Norton LE et al. Improving Guideline-Based Streptococcal Pharyngitis Testing: A Quality Improvement Initiative. Pediatrics. 2018 Jul;142(1):e20172033. doi: 10.1542/peds.2017-2033.
8. MD+ Calc website. Centor Score (Modified/McIsaac) for Strep Pharyngitis.
9. Langlois DM, Andreae M. Group A Streptococcal Infections. Pediatr Rev. 2011 Oct;32(10):423-9; quiz 430. doi: 10.1542/pir.32-10-423.
It’s wintertime, peak season for GAS pharyngitis, and you’d think that this far into the 21st century we would have a foolproof process for diagnosing which among the many patients with pharyngitis have true GAS pharyngitis. Thinking back to the 1980s, we have come a long way from simple throat cultures for detecting GAS, e.g., numerous point of care (POC) Clinical Laboratory Improvement Amendments (CLIA), waved rapid antigen detection tests (RADT), and numerous highly sensitive molecular assays, e.g. nucleic acid amplification tests (NAAT). But if you think the issues surrounding management of GAS pharyngitis have been solved by these newer tests, think again.
Several good reviews1-3 are excellent resources for those wishing a refresher on GAS diagnosis/management issues. They present nitty gritty details on comparative advantages/disadvantages of the many testing options while reminding us of the nuts and bolts of GAS pharyngitis. The following are a few nuggets from these articles.
Properly collected throat specimen. A quality throat specimen involves swabbing both tonsillar pillars plus posterior pharynx without touching tongue or inner cheeks. Two swab collections increase sensitivity by almost 10% compared with a single swab. Transport media is preferred if samples will not be cultured within 24 hours. Caveat: RADT testing of a transport media-diluted sample lowers sensitivity compared with direct swab use.
Reliable GAS detection. Commercially available tests in 2025 are well studied. Culture is considered a gold standard for detecting clinically relevant GAS by CDC.4 Culture has good sensitivity (estimated 80%-90% varying among studies and by quality of specimens) and 99% specificity but requires 16-24 hours for results. RADT solves the time-delay issues and has near 100% specificity but sensitivity used to be as low as 65%, hence the 2012 Infectious Diseases Society of America guideline recommendation for backup throat culture for negative tests.5 However, current RADT have sensitivities in the 85%-90% range.3,4 So a positive RADT reliably and quickly indicates GAS antigens are present. NAAT have the highest combined sensitivity and specificity, near 100% for each, and a positive reliably indicates GAS nucleic acids are present.
So why not simply always use NAAT? First, it’s a “be careful what you wish for” scenario. NAAT can, and do, detect dead remnants and colonizing GAS way more than culture.2,3 So NAAT are overly sensitive, adding an extra layer of interpretation difficulty, ie, as many as 20% of positive NAAT detections may be carriers or dead GAS. Second, NAAT often requires special instrumentation and kits are more expensive. That said, reimbursement is often higher for NAAT.
Choice based on accuracy in detecting GAS. If time delays were not a problem, culture would still seem the answer. If more rapid detection is needed, either RADT with culture back up or NAAT could be the answer. That said, consider that in the real world, throat cultures are less sensitive and RADT are less specific than indicated by some published data.6 So, the ideal answer, it seems, would be NAAT GAS detection coupled with a confirmatory biomarker of GAS infection. Such innate immune biomarkers may be on the horizon.3
But first, pretest screening. In 2025 what do we do with a positive result? Do we prescribe antibiotics? Do we think the detected GAS bacteria/antigens/nucleic acids represent the cause of the pharyngitis? Or did we just detect dead GAS or even a carrier, while a virus is the true cause? Challenges for this decision include most pharyngitis (up to 70%) being due to viruses, not GAS, plus up to 20% of GAS detections even by less sensitive culture or RADT can be carriers, plus an added 10%-20% of RADT and NAAT detections are dead GAS. Thus, with indiscriminate testing of all pharyngitis patients, the number of truly positive GAS detections that are actually “false positives” (GAS in some form is present but not causing pharyngitis) may be almost as high as for those representing true GAS pharyngitis.
Some tool is needed to minimize testing patients who are likely to have viral pharyngitis to reduce test-positive/GAS-pharyngitis-negative scenarios. Pretest patient screening therefore is critical to increase the positive predictive value of positive GAS testing results. The history and physical can be helpful. In the simplest form of pretest screening, eliminate those younger than 3 years old* or those with viral type sign/symptoms, eg conjunctivitis, cough, coryza.7 This could cut “false” positives by as much as a half. More complete validated scoring systems are also available but remain imperfect. The most published is the McIsaac score (modified Centor score).3-5,8 (See Table and Figure.)

However, even with this validated scoring system, misdiagnoses and some antibiotic misuse will likely occur, particularly if the controversial option to treat a patient with a score above 4 without testing is used. For example, a 2004 study in patients older than 3 years old revealed that 45% with a score above 4 points did not have GAS pharyngitis. (McIsaac et al.) A 2012 study showed similar potential overdiagnosis from using the score without testing (45% with > 4 points did not have GAS pharyngitis). Of note, clinical scores of below 2 comprised up to 10% and would be neither tested nor treated. (Figure.)

Best clinical judgment. Regardless of the chosen test, we still need to interpret positive results, ie, use best clinical judgment. We know that even with pretest screening some positives tests will represent carriers or nonviable GAS. Yet true GAS pharyngitis needs antibiotic treatment to minimize nonpyogenic and pyogenic complications, plus reduce contagion/transmission risk and days of illness. Thus, we are forced to use best clinical judgment when considering if what could be GAS pharyngitis, particularly exudative pharyngitis, could actually be due to EBV, adenovirus, or gonococcus, each of which can mimic GAS findings. Differentiating these requires discussion beyond the scope of this article, but clues are often found in the history, the patient’s age, associated symptoms and distribution of tonsillopharyngeal exudate. Likewise Group C and G streptococcal pharyngitis can mimic GAS. Note: A comprehensive throat culture can identify these streptococci but requires a special order and likely a call to the laboratory.
Summary: The age-old problem persists, ie, differentiating the minority (~30%) of pharyngitis cases needing antibiotics from the majority that do not. We all wish to promptly treat true GAS pharyngitis; however our current tools remain imperfect. That said, we should strive to correctly diagnose/manage as many patients with pharyngitis as possible. I, for one, can’t wait until we get a validated biomarker that confirms GAS as the culprit in pharyngitis episodes. In the meantime, most providers likely have clinic or hospital approved pathways for managing GAS pharyngitis, many of which are at least in part based on data from sources for this discussion. If not, a firm foundation for creating one can be found in sources among the reference list below. Finally, if you think such pathways somehow interfere with patient flow, consider that a busy multi-provider private practice successfully integrated pretest screening and a pathway while maintaining patient flow and improving antibiotic stewardship.7
*Focal pharyngotonsillar GAS infection is rare in children younger than 3 years old, when GAS nasal passage infection may manifest as streptococcosis.9
Dr Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics, Kansas City, Missouri. He has no relevant financial disclosures. Email him at [email protected].
References
1. Bannerjee D, Selvarangan RS. The Evolution of Group A Streptococcus Pharyngitis Testing. Association for Diagnostics and Laboratory Medicine, 2018, Sep 1.
2. Cohen JF et al. Group A Streptococcus Pharyngitis in Children: New Perspectives on Rapid Diagnostic Testing and Antimicrobial Stewardship. J Pediatric Infect Dis Soc. 2024 Apr 24;13(4):250-256. doi: 10.1093/jpids/piae0223.
3. Boyanton Jr BL et al. Current Laboratory and Point-of-Care Pharyngitis Diagnostic Testing and Knowledge Gaps. J Infect Dis. 2024 Oct 23;230(Suppl 3):S182–S189. doi: 10.1093/infdis/jiae415.
4. Group A Strep Infection. Centers for Disease Control and Prevention, 2024, Mar 1.
5. Shulman ST et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2012 Nov 15;55(10):e86-102. doi: 10.1093/cid/cis629.
6. Rao A et al. Diagnosis and Antibiotic Treatment of Group A Streptococcal Pharyngitis in Children in a Primary Care Setting: Impact of Point-of-Care Polymerase Chain Reaction. BMC Pediatr. 2019 Jan 16;19(1):24. doi: 10.1186/s12887-019-1393-y.
7. Norton LE et al. Improving Guideline-Based Streptococcal Pharyngitis Testing: A Quality Improvement Initiative. Pediatrics. 2018 Jul;142(1):e20172033. doi: 10.1542/peds.2017-2033.
8. MD+ Calc website. Centor Score (Modified/McIsaac) for Strep Pharyngitis.
9. Langlois DM, Andreae M. Group A Streptococcal Infections. Pediatr Rev. 2011 Oct;32(10):423-9; quiz 430. doi: 10.1542/pir.32-10-423.
Central Line Skin Reactions in Children: Survey Addresses Treatment Protocols in Use
TOPLINE:
A and reported varying management approaches.
METHODOLOGY:
- Researchers developed and administered a 14-item Qualtrics survey to 107 dermatologists providing pediatric inpatient care through the Society for Pediatric Dermatology’s Inpatient Dermatology Section and Section Chief email lists.
- A total of 35 dermatologists (33%) from multiple institutions responded to the survey; most respondents (94%) specialized in pediatric dermatology.
- Researchers assessed management of CLD-associated adverse skin reactions.
TAKEAWAY:
- All respondents reported receiving CLD-related consults, but 66% indicated there was no personal or institutional standardized approach for managing CLD-associated skin reactions.
- Respondents said most reactions were in children aged 1-12 years (19 or 76% of 25 respondents) compared with those aged < 1 year (3 or 12% of 25 respondents).
- Management strategies included switching to alternative products, applying topical corticosteroids, and performing patch testing for allergies.
IN PRACTICE:
“Insights derived from this study, including variation in clinician familiarity with reaction patterns, underscore the necessity of a standardized protocol for classifying and managing cutaneous CLD reactions in pediatric patients,” the authors wrote. “Further investigation is needed to better characterize CLD-associated allergic CD [contact dermatitis], irritant CD, and skin infections, as well as at-risk populations, to better inform clinical approaches,” they added.
SOURCE:
The study was led by Carly Mulinda, Columbia University College of Physicians and Surgeons, New York, and was published online on December 16 in Pediatric Dermatology.
LIMITATIONS:
The authors noted variable respondent awareness of institutional CLD and potential recency bias as key limitations of the study.
DISCLOSURES:
Study funding source was not declared. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
A and reported varying management approaches.
METHODOLOGY:
- Researchers developed and administered a 14-item Qualtrics survey to 107 dermatologists providing pediatric inpatient care through the Society for Pediatric Dermatology’s Inpatient Dermatology Section and Section Chief email lists.
- A total of 35 dermatologists (33%) from multiple institutions responded to the survey; most respondents (94%) specialized in pediatric dermatology.
- Researchers assessed management of CLD-associated adverse skin reactions.
TAKEAWAY:
- All respondents reported receiving CLD-related consults, but 66% indicated there was no personal or institutional standardized approach for managing CLD-associated skin reactions.
- Respondents said most reactions were in children aged 1-12 years (19 or 76% of 25 respondents) compared with those aged < 1 year (3 or 12% of 25 respondents).
- Management strategies included switching to alternative products, applying topical corticosteroids, and performing patch testing for allergies.
IN PRACTICE:
“Insights derived from this study, including variation in clinician familiarity with reaction patterns, underscore the necessity of a standardized protocol for classifying and managing cutaneous CLD reactions in pediatric patients,” the authors wrote. “Further investigation is needed to better characterize CLD-associated allergic CD [contact dermatitis], irritant CD, and skin infections, as well as at-risk populations, to better inform clinical approaches,” they added.
SOURCE:
The study was led by Carly Mulinda, Columbia University College of Physicians and Surgeons, New York, and was published online on December 16 in Pediatric Dermatology.
LIMITATIONS:
The authors noted variable respondent awareness of institutional CLD and potential recency bias as key limitations of the study.
DISCLOSURES:
Study funding source was not declared. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
A and reported varying management approaches.
METHODOLOGY:
- Researchers developed and administered a 14-item Qualtrics survey to 107 dermatologists providing pediatric inpatient care through the Society for Pediatric Dermatology’s Inpatient Dermatology Section and Section Chief email lists.
- A total of 35 dermatologists (33%) from multiple institutions responded to the survey; most respondents (94%) specialized in pediatric dermatology.
- Researchers assessed management of CLD-associated adverse skin reactions.
TAKEAWAY:
- All respondents reported receiving CLD-related consults, but 66% indicated there was no personal or institutional standardized approach for managing CLD-associated skin reactions.
- Respondents said most reactions were in children aged 1-12 years (19 or 76% of 25 respondents) compared with those aged < 1 year (3 or 12% of 25 respondents).
- Management strategies included switching to alternative products, applying topical corticosteroids, and performing patch testing for allergies.
IN PRACTICE:
“Insights derived from this study, including variation in clinician familiarity with reaction patterns, underscore the necessity of a standardized protocol for classifying and managing cutaneous CLD reactions in pediatric patients,” the authors wrote. “Further investigation is needed to better characterize CLD-associated allergic CD [contact dermatitis], irritant CD, and skin infections, as well as at-risk populations, to better inform clinical approaches,” they added.
SOURCE:
The study was led by Carly Mulinda, Columbia University College of Physicians and Surgeons, New York, and was published online on December 16 in Pediatric Dermatology.
LIMITATIONS:
The authors noted variable respondent awareness of institutional CLD and potential recency bias as key limitations of the study.
DISCLOSURES:
Study funding source was not declared. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Imipenem-Cilastatin-Relebactam, the New Go-To for Pneumonia?
TOPLINE:
In a multinational phase 3 trial, imipenem-cilastatin-relebactam demonstrated noninferiority to piperacillin-tazobactam in treating critically ill patients with hospital-acquired bacterial pneumonia (HABP) or ventilator-associated bacterial pneumonia (VABP), with a comparable safety profile.
METHODOLOGY:
- This multinational phase 3 trial, conducted between September 2018 and July 2022, compared imipenem-cilastatin-relebactam with piperacillin-tazobactam for HABP and VABP to support its use across multiple countries.
- Overall, 270 patients with HABP or VABP (mean age, 57.6 years; 73.3% men) were randomly assigned to receive either intravenous imipenem-cilastatin-relebactam (500 mg/250 mg) or piperacillin-tazobactam (4000 mg/500 mg) every 6 hours over 30 minutes for 7-14 days.
- Both treatment groups included critically ill patients, with 54.5% and 55.1% of patients in the imipenem-cilastatin-relebactam and piperacillin-tazobactam groups, respectively, having an Acute Physiology and Chronic Health Evaluation II score ≥ 15.
- The primary outcome was the 28-day all-cause mortality; secondary outcomes included the rates of clinical and microbiological responses, as well as the incidence of adverse events.
TAKEAWAY:
- Imipenem-cilastatin-relebactam was noninferior to piperacillin-tazobactam in terms of 28-day all-cause mortality (adjusted difference, 5.2%; 95% CI, −1.5-12.4; P = .024 for noninferiority).
- After treatment, microbiological response rates were 48.8% in the imipenem-cilastatin-relebactam group, whereas the rates were 47.9% in the piperacillin-tazobactam group.
- The incidence of drug-related adverse events was similar across the treatment groups, with diarrhea, increased levels of alanine aminotransferase and aspartate aminotransferase, and abnormal hepatic function being the most common events.
IN PRACTICE:
“These results support the use of IMI/REL [imipenem-cilastatin-relebactam] in MDR [multidrug-resistant] infections globally, including to expand the range of available treatments for critically ill patients with HABP/VABP in China, and provide additional data to inform the World Health Organization’s MDR pathogen strategy,” the authors wrote.
SOURCE:
This study was led by Junjie Li, Department of Pulmonary and Critical Care Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online on December 12, 2024, in the International Journal of Infectious Diseases.
LIMITATIONS:
This study excluded patients with immunosuppression and those on intermittent hemodialysis, limiting the generalizability of the results to these populations.
DISCLOSURES:
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co. Inc., Rahway, New Jersey. Some authors served as employees of Merck Sharp & Dohme LLC, New Jersey, and MSD, China.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In a multinational phase 3 trial, imipenem-cilastatin-relebactam demonstrated noninferiority to piperacillin-tazobactam in treating critically ill patients with hospital-acquired bacterial pneumonia (HABP) or ventilator-associated bacterial pneumonia (VABP), with a comparable safety profile.
METHODOLOGY:
- This multinational phase 3 trial, conducted between September 2018 and July 2022, compared imipenem-cilastatin-relebactam with piperacillin-tazobactam for HABP and VABP to support its use across multiple countries.
- Overall, 270 patients with HABP or VABP (mean age, 57.6 years; 73.3% men) were randomly assigned to receive either intravenous imipenem-cilastatin-relebactam (500 mg/250 mg) or piperacillin-tazobactam (4000 mg/500 mg) every 6 hours over 30 minutes for 7-14 days.
- Both treatment groups included critically ill patients, with 54.5% and 55.1% of patients in the imipenem-cilastatin-relebactam and piperacillin-tazobactam groups, respectively, having an Acute Physiology and Chronic Health Evaluation II score ≥ 15.
- The primary outcome was the 28-day all-cause mortality; secondary outcomes included the rates of clinical and microbiological responses, as well as the incidence of adverse events.
TAKEAWAY:
- Imipenem-cilastatin-relebactam was noninferior to piperacillin-tazobactam in terms of 28-day all-cause mortality (adjusted difference, 5.2%; 95% CI, −1.5-12.4; P = .024 for noninferiority).
- After treatment, microbiological response rates were 48.8% in the imipenem-cilastatin-relebactam group, whereas the rates were 47.9% in the piperacillin-tazobactam group.
- The incidence of drug-related adverse events was similar across the treatment groups, with diarrhea, increased levels of alanine aminotransferase and aspartate aminotransferase, and abnormal hepatic function being the most common events.
IN PRACTICE:
“These results support the use of IMI/REL [imipenem-cilastatin-relebactam] in MDR [multidrug-resistant] infections globally, including to expand the range of available treatments for critically ill patients with HABP/VABP in China, and provide additional data to inform the World Health Organization’s MDR pathogen strategy,” the authors wrote.
SOURCE:
This study was led by Junjie Li, Department of Pulmonary and Critical Care Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online on December 12, 2024, in the International Journal of Infectious Diseases.
LIMITATIONS:
This study excluded patients with immunosuppression and those on intermittent hemodialysis, limiting the generalizability of the results to these populations.
DISCLOSURES:
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co. Inc., Rahway, New Jersey. Some authors served as employees of Merck Sharp & Dohme LLC, New Jersey, and MSD, China.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
In a multinational phase 3 trial, imipenem-cilastatin-relebactam demonstrated noninferiority to piperacillin-tazobactam in treating critically ill patients with hospital-acquired bacterial pneumonia (HABP) or ventilator-associated bacterial pneumonia (VABP), with a comparable safety profile.
METHODOLOGY:
- This multinational phase 3 trial, conducted between September 2018 and July 2022, compared imipenem-cilastatin-relebactam with piperacillin-tazobactam for HABP and VABP to support its use across multiple countries.
- Overall, 270 patients with HABP or VABP (mean age, 57.6 years; 73.3% men) were randomly assigned to receive either intravenous imipenem-cilastatin-relebactam (500 mg/250 mg) or piperacillin-tazobactam (4000 mg/500 mg) every 6 hours over 30 minutes for 7-14 days.
- Both treatment groups included critically ill patients, with 54.5% and 55.1% of patients in the imipenem-cilastatin-relebactam and piperacillin-tazobactam groups, respectively, having an Acute Physiology and Chronic Health Evaluation II score ≥ 15.
- The primary outcome was the 28-day all-cause mortality; secondary outcomes included the rates of clinical and microbiological responses, as well as the incidence of adverse events.
TAKEAWAY:
- Imipenem-cilastatin-relebactam was noninferior to piperacillin-tazobactam in terms of 28-day all-cause mortality (adjusted difference, 5.2%; 95% CI, −1.5-12.4; P = .024 for noninferiority).
- After treatment, microbiological response rates were 48.8% in the imipenem-cilastatin-relebactam group, whereas the rates were 47.9% in the piperacillin-tazobactam group.
- The incidence of drug-related adverse events was similar across the treatment groups, with diarrhea, increased levels of alanine aminotransferase and aspartate aminotransferase, and abnormal hepatic function being the most common events.
IN PRACTICE:
“These results support the use of IMI/REL [imipenem-cilastatin-relebactam] in MDR [multidrug-resistant] infections globally, including to expand the range of available treatments for critically ill patients with HABP/VABP in China, and provide additional data to inform the World Health Organization’s MDR pathogen strategy,” the authors wrote.
SOURCE:
This study was led by Junjie Li, Department of Pulmonary and Critical Care Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. It was published online on December 12, 2024, in the International Journal of Infectious Diseases.
LIMITATIONS:
This study excluded patients with immunosuppression and those on intermittent hemodialysis, limiting the generalizability of the results to these populations.
DISCLOSURES:
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co. Inc., Rahway, New Jersey. Some authors served as employees of Merck Sharp & Dohme LLC, New Jersey, and MSD, China.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Doxycycline Kits Boost Chlamydia Treatment in ED
TOPLINE:
METHODOLOGY:
- A single-center retrospective chart review included adults with positive chlamydia tests in the ED between 2021 and 2023.
- In total, 98 received doxycycline discharge kits; 72 patients were enrolled before the implementation of discharge kits for comparison.
- There were no differences in symptoms of infection between patients who received and those who did not receive the kit.
- Main outcome was the number of patients who received treatment.
- Secondary outcomes included 90-day return visits for complaints of sexually transmitted infections and time to treatment initiation.
TAKEAWAY:
- Appropriate treatment rates rose significantly post-implementation of the discharge kit (69.1% vs 45.8%; odds ratio, 2.63; P = .002).
- Implementation of the discharge kit also reduced the time to definitive treatment from 22.7 hours to 1.3 hours (P < .001).
- No significant differences in 90-day ED return visits, time to initial treatment in the ED, and doxycycline prescription via culture callback programs between the two groups.
IN PRACTICE:
“Pharmacy-driven doxycycline discharge kits significantly increased guideline-directed treatment and decreased time to treatment for chlamydia infections in the ED population at an urban academic medical center,” the authors wrote. “Overall, this initiative overcame barriers to treatment for a significant public health issue, supporting the need for expansion to other emergency departments across the country.”
SOURCE:
The study was led by Carly Loudermilk, Department of Pharmacy, Louisville, Kentucky, and was published online on November 14, 2024, in The American Journal of Emergency Medicine.
LIMITATIONS:
Retrospective design is the main limitation and lack of insurance fill history in some patients.
DISCLOSURES:
The study received no external funding. The authors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A single-center retrospective chart review included adults with positive chlamydia tests in the ED between 2021 and 2023.
- In total, 98 received doxycycline discharge kits; 72 patients were enrolled before the implementation of discharge kits for comparison.
- There were no differences in symptoms of infection between patients who received and those who did not receive the kit.
- Main outcome was the number of patients who received treatment.
- Secondary outcomes included 90-day return visits for complaints of sexually transmitted infections and time to treatment initiation.
TAKEAWAY:
- Appropriate treatment rates rose significantly post-implementation of the discharge kit (69.1% vs 45.8%; odds ratio, 2.63; P = .002).
- Implementation of the discharge kit also reduced the time to definitive treatment from 22.7 hours to 1.3 hours (P < .001).
- No significant differences in 90-day ED return visits, time to initial treatment in the ED, and doxycycline prescription via culture callback programs between the two groups.
IN PRACTICE:
“Pharmacy-driven doxycycline discharge kits significantly increased guideline-directed treatment and decreased time to treatment for chlamydia infections in the ED population at an urban academic medical center,” the authors wrote. “Overall, this initiative overcame barriers to treatment for a significant public health issue, supporting the need for expansion to other emergency departments across the country.”
SOURCE:
The study was led by Carly Loudermilk, Department of Pharmacy, Louisville, Kentucky, and was published online on November 14, 2024, in The American Journal of Emergency Medicine.
LIMITATIONS:
Retrospective design is the main limitation and lack of insurance fill history in some patients.
DISCLOSURES:
The study received no external funding. The authors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A single-center retrospective chart review included adults with positive chlamydia tests in the ED between 2021 and 2023.
- In total, 98 received doxycycline discharge kits; 72 patients were enrolled before the implementation of discharge kits for comparison.
- There were no differences in symptoms of infection between patients who received and those who did not receive the kit.
- Main outcome was the number of patients who received treatment.
- Secondary outcomes included 90-day return visits for complaints of sexually transmitted infections and time to treatment initiation.
TAKEAWAY:
- Appropriate treatment rates rose significantly post-implementation of the discharge kit (69.1% vs 45.8%; odds ratio, 2.63; P = .002).
- Implementation of the discharge kit also reduced the time to definitive treatment from 22.7 hours to 1.3 hours (P < .001).
- No significant differences in 90-day ED return visits, time to initial treatment in the ED, and doxycycline prescription via culture callback programs between the two groups.
IN PRACTICE:
“Pharmacy-driven doxycycline discharge kits significantly increased guideline-directed treatment and decreased time to treatment for chlamydia infections in the ED population at an urban academic medical center,” the authors wrote. “Overall, this initiative overcame barriers to treatment for a significant public health issue, supporting the need for expansion to other emergency departments across the country.”
SOURCE:
The study was led by Carly Loudermilk, Department of Pharmacy, Louisville, Kentucky, and was published online on November 14, 2024, in The American Journal of Emergency Medicine.
LIMITATIONS:
Retrospective design is the main limitation and lack of insurance fill history in some patients.
DISCLOSURES:
The study received no external funding. The authors disclosed no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Common Gut Infection Tied to Alzheimer’s Disease
Researchers are gaining new insight into the relationship between the human cytomegalovirus (HCMV), a common herpes virus found in the gut, and the immune response associated with CD83 antibody in some individuals with Alzheimer’s disease (AD).
Using tissue samples from deceased donors with AD, the study showed CD83-positive (CD83+) microglia in the superior frontal gyrus (SFG) are significantly associated with elevated immunoglobulin gamma 4 (IgG4) and HCMV in the transverse colon (TC), increased anti-HCMV IgG4 in the cerebrospinal fluid (CSF), and both HCMV and IgG4 in the SFG and vagus nerve.
“Our results indicate a complex, cross-tissue interaction between HCMV and the host adaptive immune response associated with CD83+ microglia in persons with AD,” noted the investigators, including Benjamin P. Readhead, MBBS, research associate professor, ASU-Banner Neurodegenerative Disease Research Center, Arizona State University, Tempe.
The results suggest antiviral therapy in patients with biomarker evidence of HCMV, IgG4, or CD83+ microglia might ward off dementia.
“We’re preparing to conduct a clinical trial to evaluate whether careful use of existing antivirals might be clinically helpful in preventing or slowing progression of CD83+ associated Alzheimer’s disease,” Readhead said in an interview.
The study was published on December 19, 2024, in Alzheimer’s & Dementia.
Vagus Nerve a Potential Pathway?
CMV is a common virus. In the United States, nearly one in three children are already infected with CMV by age 5 years. Over half the adults have been infected with CMV by age 40 years, the Centers for Disease Control and Prevention reported.
It is typically passed through bodily fluids and spread only when the virus is active. It’s not considered a sexually transmitted disease.
Compared with other IgG subclasses, IgG4 is believed to be a less inflammatory, and therefore less damaging, immune response. But this response may be less effective at clearing infections and allow invasion of HCMV into the brain.
The researchers previously found a CD83+ microglial subtype in the SFG of 47% of brain donors with AD vs 25% of unaffected control individuals. They reported this subtype is associated with increased IgG4 in the TC.
The current analysis extends investigations of the potential etiology and clinicopathologic relevance of CD83+ microglia in the context of AD.
Researchers conducted experiments using donated tissue samples from deceased patients with AD and control individuals. Sources for these samples included the Banner cohort, for whom classifications for the presence of CD83+ microglia were available, as were tissue samples from the SFG, TC, and vagus nerve, and the Religious Orders Study and Rush Memory and Aging Project (ROSMAP), in which participants without known dementia are evaluated annually.
From the Banner cohort, researchers completed immunohistochemistry (IHC) studies on 34 SFG samples (21 AD and 13 control individuals) and included 25 TC samples (13 AD and 12 control individuals) and 8 vagal nerve samples (6 AD and 2 control individuals) in the study. From the ROSMAP cohort, they completed IHC studies on 27 prefrontal cortex samples from individuals with AD.
They carefully selected these samples to ensure matching for critical factors such as postmortem interval, age, and sex, as well as other relevant covariates, said the authors.
The study verified that CD83+ microglia are associated with IgG4 and HCMV in the TC and showed a significant association between CD83+ microglia and IgG4 immunoreactivity in the TC.
Investigators confirmed HCMV positivity in all nine CD83+ TC samples evaluated and in one CD83– TC sample, indicating a strong positive association between HCMV within the TC and CD83+ microglia within the SFG.
HCMV IgG seroprevalence is common, varies by age and comorbidity, and is present in 79% of 85-year-olds, the investigators noted. “Despite this, we note that HCMV presence in the TC was not ubiquitous and was significantly associated with CD83+ microglia and HCMV in the SFG,” they wrote.
This observation, they added, “may help reconcile how a common pathogen might contribute to a disease that most individuals do not develop.”
The experiments also uncovered increased anti-HCMV IgG4 in the CSF and evidence of HCMV and IgG4 in the vagus nerve.
“Overall, the histochemical staining patterns observed in TC, SFG, and vagus nerve of CD83+ subjects are consistent with active HCMV infection,” the investigators wrote. “Taken together, these results indicate a multiorgan presence of IgG4 and HCMV in subjects with CD83+ microglia within the SFG,” they added.
Accelerated AD Pathology
The team showed HCMV infection accelerates production of two pathologic features of AD — amyloid beta (Abeta) and tau — and causes neuronal death. “We observed high, positive correlations between the abundance of HCMV, and both Abeta42 and pTau-212,” they wrote.
As HCMV histochemistry is consistent with an active HCMV infection, the findings “may indicate an opportunity for the administration of antiviral therapy in subjects with AD and biomarker evidence of HCMV, IgG4, or CD83+ microglia,” they added.
In addition to planning a clinical trial of existing antivirals, the research team is developing a blood test that can help identify patients with an active HCMV infection who might benefit from such an intervention, said Readhead.
But he emphasized that the research is still in its infancy. “Our study is best understood as a series of interesting scientific findings that warrant further exploration, replication, and validation in additional study populations.”
Although it’s too early for the study to impact practice, “we’re motivated to understand whether these findings have implications for clinical care,” he added.
Tipping the Balance
A number of experts have weighed in on the research via the Science Media Center, an independent forum featuring the voices and views on science news from experts in the field.
Andrew Doig, PhD, professor, Division of Neuroscience, University of Manchester in England, said the new work supports the hypothesis that HCMV might be a trigger that tips the balance from a healthy brain to one with dementia. “If so, antiviral drugs against HCMV might be beneficial in reducing the risk of AD.”
Doig noted newly approved drugs for AD are expensive, provide only a small benefit, and have significant risks, such as causing brain hemorrhages. “Antiviral drugs are an attractive alternative that are well worth exploring.”
Richard Oakley, PhD, associate director of research and innovation, Alzheimer’s Society, cautioned the study only established a connection and didn’t directly show the virus leads to AD. “Also, the virus is not found in the brain of everyone with Alzheimer’s disease, the most common form of dementia.”
The significance of the new findings is “far from clear,” commented William McEwan, PhD, group leader at the UK Dementia Research Institute at Cambridge, England. “The study does not address how common this infection is in people without Alzheimer’s and therefore cannot by itself suggest that HCMV infection, or the associated immune response, is a driver of disease.”
The experts agreed follow-up research is needed to confirm these new findings and understand what they mean.
The study received support from the National Institute on Aging, National Institutes of Health, Global Lyme Alliance, National Institute of Neurological Disorders and Stroke, Arizona Alzheimer’s Consortium, The Benter Foundation, and NOMIS Stiftung. Readhead is a coinventor on a patent application for an IgG4-based peripheral biomarker for the detection of CD83+ microglia. Doig is a founder, director, and consultant for PharmaKure, which works on AD drugs and diagnostics, although not on viruses. He has cowritten a review on Viral Involvement in Alzheimer’s Disease. McEwan reported receiving research funding from Takeda Pharmaceuticals and is a founder and consultant to Trimtech Therapeutics.
A version of this article first appeared on Medscape.com.
Researchers are gaining new insight into the relationship between the human cytomegalovirus (HCMV), a common herpes virus found in the gut, and the immune response associated with CD83 antibody in some individuals with Alzheimer’s disease (AD).
Using tissue samples from deceased donors with AD, the study showed CD83-positive (CD83+) microglia in the superior frontal gyrus (SFG) are significantly associated with elevated immunoglobulin gamma 4 (IgG4) and HCMV in the transverse colon (TC), increased anti-HCMV IgG4 in the cerebrospinal fluid (CSF), and both HCMV and IgG4 in the SFG and vagus nerve.
“Our results indicate a complex, cross-tissue interaction between HCMV and the host adaptive immune response associated with CD83+ microglia in persons with AD,” noted the investigators, including Benjamin P. Readhead, MBBS, research associate professor, ASU-Banner Neurodegenerative Disease Research Center, Arizona State University, Tempe.
The results suggest antiviral therapy in patients with biomarker evidence of HCMV, IgG4, or CD83+ microglia might ward off dementia.
“We’re preparing to conduct a clinical trial to evaluate whether careful use of existing antivirals might be clinically helpful in preventing or slowing progression of CD83+ associated Alzheimer’s disease,” Readhead said in an interview.
The study was published on December 19, 2024, in Alzheimer’s & Dementia.
Vagus Nerve a Potential Pathway?
CMV is a common virus. In the United States, nearly one in three children are already infected with CMV by age 5 years. Over half the adults have been infected with CMV by age 40 years, the Centers for Disease Control and Prevention reported.
It is typically passed through bodily fluids and spread only when the virus is active. It’s not considered a sexually transmitted disease.
Compared with other IgG subclasses, IgG4 is believed to be a less inflammatory, and therefore less damaging, immune response. But this response may be less effective at clearing infections and allow invasion of HCMV into the brain.
The researchers previously found a CD83+ microglial subtype in the SFG of 47% of brain donors with AD vs 25% of unaffected control individuals. They reported this subtype is associated with increased IgG4 in the TC.
The current analysis extends investigations of the potential etiology and clinicopathologic relevance of CD83+ microglia in the context of AD.
Researchers conducted experiments using donated tissue samples from deceased patients with AD and control individuals. Sources for these samples included the Banner cohort, for whom classifications for the presence of CD83+ microglia were available, as were tissue samples from the SFG, TC, and vagus nerve, and the Religious Orders Study and Rush Memory and Aging Project (ROSMAP), in which participants without known dementia are evaluated annually.
From the Banner cohort, researchers completed immunohistochemistry (IHC) studies on 34 SFG samples (21 AD and 13 control individuals) and included 25 TC samples (13 AD and 12 control individuals) and 8 vagal nerve samples (6 AD and 2 control individuals) in the study. From the ROSMAP cohort, they completed IHC studies on 27 prefrontal cortex samples from individuals with AD.
They carefully selected these samples to ensure matching for critical factors such as postmortem interval, age, and sex, as well as other relevant covariates, said the authors.
The study verified that CD83+ microglia are associated with IgG4 and HCMV in the TC and showed a significant association between CD83+ microglia and IgG4 immunoreactivity in the TC.
Investigators confirmed HCMV positivity in all nine CD83+ TC samples evaluated and in one CD83– TC sample, indicating a strong positive association between HCMV within the TC and CD83+ microglia within the SFG.
HCMV IgG seroprevalence is common, varies by age and comorbidity, and is present in 79% of 85-year-olds, the investigators noted. “Despite this, we note that HCMV presence in the TC was not ubiquitous and was significantly associated with CD83+ microglia and HCMV in the SFG,” they wrote.
This observation, they added, “may help reconcile how a common pathogen might contribute to a disease that most individuals do not develop.”
The experiments also uncovered increased anti-HCMV IgG4 in the CSF and evidence of HCMV and IgG4 in the vagus nerve.
“Overall, the histochemical staining patterns observed in TC, SFG, and vagus nerve of CD83+ subjects are consistent with active HCMV infection,” the investigators wrote. “Taken together, these results indicate a multiorgan presence of IgG4 and HCMV in subjects with CD83+ microglia within the SFG,” they added.
Accelerated AD Pathology
The team showed HCMV infection accelerates production of two pathologic features of AD — amyloid beta (Abeta) and tau — and causes neuronal death. “We observed high, positive correlations between the abundance of HCMV, and both Abeta42 and pTau-212,” they wrote.
As HCMV histochemistry is consistent with an active HCMV infection, the findings “may indicate an opportunity for the administration of antiviral therapy in subjects with AD and biomarker evidence of HCMV, IgG4, or CD83+ microglia,” they added.
In addition to planning a clinical trial of existing antivirals, the research team is developing a blood test that can help identify patients with an active HCMV infection who might benefit from such an intervention, said Readhead.
But he emphasized that the research is still in its infancy. “Our study is best understood as a series of interesting scientific findings that warrant further exploration, replication, and validation in additional study populations.”
Although it’s too early for the study to impact practice, “we’re motivated to understand whether these findings have implications for clinical care,” he added.
Tipping the Balance
A number of experts have weighed in on the research via the Science Media Center, an independent forum featuring the voices and views on science news from experts in the field.
Andrew Doig, PhD, professor, Division of Neuroscience, University of Manchester in England, said the new work supports the hypothesis that HCMV might be a trigger that tips the balance from a healthy brain to one with dementia. “If so, antiviral drugs against HCMV might be beneficial in reducing the risk of AD.”
Doig noted newly approved drugs for AD are expensive, provide only a small benefit, and have significant risks, such as causing brain hemorrhages. “Antiviral drugs are an attractive alternative that are well worth exploring.”
Richard Oakley, PhD, associate director of research and innovation, Alzheimer’s Society, cautioned the study only established a connection and didn’t directly show the virus leads to AD. “Also, the virus is not found in the brain of everyone with Alzheimer’s disease, the most common form of dementia.”
The significance of the new findings is “far from clear,” commented William McEwan, PhD, group leader at the UK Dementia Research Institute at Cambridge, England. “The study does not address how common this infection is in people without Alzheimer’s and therefore cannot by itself suggest that HCMV infection, or the associated immune response, is a driver of disease.”
The experts agreed follow-up research is needed to confirm these new findings and understand what they mean.
The study received support from the National Institute on Aging, National Institutes of Health, Global Lyme Alliance, National Institute of Neurological Disorders and Stroke, Arizona Alzheimer’s Consortium, The Benter Foundation, and NOMIS Stiftung. Readhead is a coinventor on a patent application for an IgG4-based peripheral biomarker for the detection of CD83+ microglia. Doig is a founder, director, and consultant for PharmaKure, which works on AD drugs and diagnostics, although not on viruses. He has cowritten a review on Viral Involvement in Alzheimer’s Disease. McEwan reported receiving research funding from Takeda Pharmaceuticals and is a founder and consultant to Trimtech Therapeutics.
A version of this article first appeared on Medscape.com.
Researchers are gaining new insight into the relationship between the human cytomegalovirus (HCMV), a common herpes virus found in the gut, and the immune response associated with CD83 antibody in some individuals with Alzheimer’s disease (AD).
Using tissue samples from deceased donors with AD, the study showed CD83-positive (CD83+) microglia in the superior frontal gyrus (SFG) are significantly associated with elevated immunoglobulin gamma 4 (IgG4) and HCMV in the transverse colon (TC), increased anti-HCMV IgG4 in the cerebrospinal fluid (CSF), and both HCMV and IgG4 in the SFG and vagus nerve.
“Our results indicate a complex, cross-tissue interaction between HCMV and the host adaptive immune response associated with CD83+ microglia in persons with AD,” noted the investigators, including Benjamin P. Readhead, MBBS, research associate professor, ASU-Banner Neurodegenerative Disease Research Center, Arizona State University, Tempe.
The results suggest antiviral therapy in patients with biomarker evidence of HCMV, IgG4, or CD83+ microglia might ward off dementia.
“We’re preparing to conduct a clinical trial to evaluate whether careful use of existing antivirals might be clinically helpful in preventing or slowing progression of CD83+ associated Alzheimer’s disease,” Readhead said in an interview.
The study was published on December 19, 2024, in Alzheimer’s & Dementia.
Vagus Nerve a Potential Pathway?
CMV is a common virus. In the United States, nearly one in three children are already infected with CMV by age 5 years. Over half the adults have been infected with CMV by age 40 years, the Centers for Disease Control and Prevention reported.
It is typically passed through bodily fluids and spread only when the virus is active. It’s not considered a sexually transmitted disease.
Compared with other IgG subclasses, IgG4 is believed to be a less inflammatory, and therefore less damaging, immune response. But this response may be less effective at clearing infections and allow invasion of HCMV into the brain.
The researchers previously found a CD83+ microglial subtype in the SFG of 47% of brain donors with AD vs 25% of unaffected control individuals. They reported this subtype is associated with increased IgG4 in the TC.
The current analysis extends investigations of the potential etiology and clinicopathologic relevance of CD83+ microglia in the context of AD.
Researchers conducted experiments using donated tissue samples from deceased patients with AD and control individuals. Sources for these samples included the Banner cohort, for whom classifications for the presence of CD83+ microglia were available, as were tissue samples from the SFG, TC, and vagus nerve, and the Religious Orders Study and Rush Memory and Aging Project (ROSMAP), in which participants without known dementia are evaluated annually.
From the Banner cohort, researchers completed immunohistochemistry (IHC) studies on 34 SFG samples (21 AD and 13 control individuals) and included 25 TC samples (13 AD and 12 control individuals) and 8 vagal nerve samples (6 AD and 2 control individuals) in the study. From the ROSMAP cohort, they completed IHC studies on 27 prefrontal cortex samples from individuals with AD.
They carefully selected these samples to ensure matching for critical factors such as postmortem interval, age, and sex, as well as other relevant covariates, said the authors.
The study verified that CD83+ microglia are associated with IgG4 and HCMV in the TC and showed a significant association between CD83+ microglia and IgG4 immunoreactivity in the TC.
Investigators confirmed HCMV positivity in all nine CD83+ TC samples evaluated and in one CD83– TC sample, indicating a strong positive association between HCMV within the TC and CD83+ microglia within the SFG.
HCMV IgG seroprevalence is common, varies by age and comorbidity, and is present in 79% of 85-year-olds, the investigators noted. “Despite this, we note that HCMV presence in the TC was not ubiquitous and was significantly associated with CD83+ microglia and HCMV in the SFG,” they wrote.
This observation, they added, “may help reconcile how a common pathogen might contribute to a disease that most individuals do not develop.”
The experiments also uncovered increased anti-HCMV IgG4 in the CSF and evidence of HCMV and IgG4 in the vagus nerve.
“Overall, the histochemical staining patterns observed in TC, SFG, and vagus nerve of CD83+ subjects are consistent with active HCMV infection,” the investigators wrote. “Taken together, these results indicate a multiorgan presence of IgG4 and HCMV in subjects with CD83+ microglia within the SFG,” they added.
Accelerated AD Pathology
The team showed HCMV infection accelerates production of two pathologic features of AD — amyloid beta (Abeta) and tau — and causes neuronal death. “We observed high, positive correlations between the abundance of HCMV, and both Abeta42 and pTau-212,” they wrote.
As HCMV histochemistry is consistent with an active HCMV infection, the findings “may indicate an opportunity for the administration of antiviral therapy in subjects with AD and biomarker evidence of HCMV, IgG4, or CD83+ microglia,” they added.
In addition to planning a clinical trial of existing antivirals, the research team is developing a blood test that can help identify patients with an active HCMV infection who might benefit from such an intervention, said Readhead.
But he emphasized that the research is still in its infancy. “Our study is best understood as a series of interesting scientific findings that warrant further exploration, replication, and validation in additional study populations.”
Although it’s too early for the study to impact practice, “we’re motivated to understand whether these findings have implications for clinical care,” he added.
Tipping the Balance
A number of experts have weighed in on the research via the Science Media Center, an independent forum featuring the voices and views on science news from experts in the field.
Andrew Doig, PhD, professor, Division of Neuroscience, University of Manchester in England, said the new work supports the hypothesis that HCMV might be a trigger that tips the balance from a healthy brain to one with dementia. “If so, antiviral drugs against HCMV might be beneficial in reducing the risk of AD.”
Doig noted newly approved drugs for AD are expensive, provide only a small benefit, and have significant risks, such as causing brain hemorrhages. “Antiviral drugs are an attractive alternative that are well worth exploring.”
Richard Oakley, PhD, associate director of research and innovation, Alzheimer’s Society, cautioned the study only established a connection and didn’t directly show the virus leads to AD. “Also, the virus is not found in the brain of everyone with Alzheimer’s disease, the most common form of dementia.”
The significance of the new findings is “far from clear,” commented William McEwan, PhD, group leader at the UK Dementia Research Institute at Cambridge, England. “The study does not address how common this infection is in people without Alzheimer’s and therefore cannot by itself suggest that HCMV infection, or the associated immune response, is a driver of disease.”
The experts agreed follow-up research is needed to confirm these new findings and understand what they mean.
The study received support from the National Institute on Aging, National Institutes of Health, Global Lyme Alliance, National Institute of Neurological Disorders and Stroke, Arizona Alzheimer’s Consortium, The Benter Foundation, and NOMIS Stiftung. Readhead is a coinventor on a patent application for an IgG4-based peripheral biomarker for the detection of CD83+ microglia. Doig is a founder, director, and consultant for PharmaKure, which works on AD drugs and diagnostics, although not on viruses. He has cowritten a review on Viral Involvement in Alzheimer’s Disease. McEwan reported receiving research funding from Takeda Pharmaceuticals and is a founder and consultant to Trimtech Therapeutics.
A version of this article first appeared on Medscape.com.
FROM ALZHEIMER’S & DEMENTIA
How Bird Flu Spread Unchecked, Posing a Pandemic Risk
Keith Poulsen’s jaw dropped when farmers showed him images on their cellphones at the World Dairy Expo in Wisconsin in October. A livestock veterinarian at the University of Wisconsin-Madison, Poulsen had seen sick cows before, with their noses dripping and udders slack.
In 14-hour shifts, dairy workers pumped gallons of electrolyte-rich fluids into ailing cows through metal tubes inserted into the esophagus.
“It was like watching a field hospital on an active battlefront treating hundreds of wounded soldiers,” he said.
Nearly a year into the first outbreak of the bird flu among cattle, the virus shows no sign of slowing. The US government failed to eliminate the virus on dairy farms when it was confined to a handful of states, by quickly identifying infected cows and taking measures to keep their infections from spreading. Now at least 875 herds across 16 states have tested positive.
Experts say they have lost faith in the government’s ability to contain the outbreak.
“We are in a terrible situation and going into a worse situation,” said Angela Rasmussen, a virologist at the University of Saskatchewan in Canada. “I don’t know if the bird flu will become a pandemic, but if it does, we are screwed.”
To understand how the bird flu got out of hand, KFF Health News interviewed nearly 70 government officials, farmers and farmworkers, and researchers with expertise in virology, pandemics, veterinary medicine, and more.
Together with emails obtained from local health departments through public records requests, this investigation revealed key problems, including deference to the farm industry, eroded public health budgets, neglect for the safety of agriculture workers, and the sluggish pace of federal interventions.
Case in point: The US Department of Agriculture this month announced a federal order to test milk nationwide. Researchers welcomed the news but said it should have happened months ago — before the virus was so entrenched.
“It’s disheartening to see so many of the same failures that emerged during the COVID-19 crisis reemerge,” said Tom Bollyky, director of the Global Health Program at the Council on Foreign Relations.
Far more bird flu damage is inevitable, but the extent of it will be left to the Trump administration and Mother Nature. Already, the USDA has funneled more than $1.7 billion into tamping down the bird flu on poultry farms since 2022, which includes reimbursing farmers who’ve had to cull their flocks, and more than $430 million into combating the bird flu on dairy farms. In coming years, the bird flu may cost billions of dollars more in expenses and losses. Dairy industry experts say the virus kills roughly 2%-5% of infected dairy cows and reduces a herd’s milk production by about 20%.
Worse, the outbreak poses the threat of a pandemic. More than 60 people in the US have been infected, mainly by cows or poultry, but cases could skyrocket if the virus evolves to spread efficiently from person to person. And the recent news of a person critically ill in Louisiana with the bird flu shows that the virus can be dangerous.
Just a few mutations could allow the bird flu to spread between people. Because viruses mutate within human and animal bodies, each infection is like a pull of a slot machine lever.
“Even if there’s only a 5% chance of a bird flu pandemic happening, we’re talking about a pandemic that probably looks like 2020 or worse,” said Tom Peacock, a bird flu researcher at the Pirbright Institute in the United Kingdom, referring to COVID. “The US knows the risk but hasn’t done anything to slow this down,” he added.
Beyond the bird flu, the federal government’s handling of the outbreak reveals cracks in the US health security system that would allow other risky new pathogens to take root. “This virus may not be the one that takes off,” said Maria Van Kerkhove, director of the emerging diseases group at the World Health Organization. “But this is a real-fire exercise right now, and it demonstrates what needs to be improved.”
A Slow Start
It may have been a grackle, a goose, or some other wild bird that infected a cow in northern Texas. In February, the state’s dairy farmers took note when cows stopped making milk. They worked alongside veterinarians to figure out why. In less than two months, veterinary researchers identified the highly pathogenic H5N1 bird flu virus as the culprit.
Long listed among pathogens with pandemic potential, the bird flu’s unprecedented spread among cows marked a worrying shift. It had evolved to thrive in animals that are more like people biologically than birds.
After the USDA announced the dairy outbreak on March 25, control shifted from farmers, veterinarians, and local officials to state and federal agencies. Collaboration disintegrated almost immediately.
Farmers worried the government might block their milk sales or even demand sick cows be killed, as poultry are, said Kay Russo, a livestock veterinarian in Fort Collins, Colorado.
Instead, Russo and other veterinarians said, they were dismayed by inaction. The USDA didn’t respond to their urgent requests to support studies on dairy farms — and for money and confidentiality policies to protect farmers from financial loss if they agreed to test animals.
The USDA announced that it would conduct studies itself. But researchers grew anxious as weeks passed without results. “Probably the biggest mistake from the USDA was not involving the boots-on-the-ground veterinarians,” Russo said.
Will Clement, a USDA senior adviser for communications, said in an email: “Since first learning of H5N1 in dairy cattle in late March 2024, USDA has worked swiftly and diligently to assess the prevalence of the virus in US dairy herds.” The agency provided research funds to state and national animal health labs beginning in April, he added.
The USDA didn’t require lactating cows to be tested before interstate travel until April 29. By then, the outbreak had spread to eight other states. Farmers often move cattle across great distances, for calving in one place, raising in warm, dry climates, and milking in cooler ones. Analyses of the virus’s genes implied that it spread between cows rather than repeatedly jumping from birds into herds.
Milking equipment was a likely source of infection, and there were hints of other possibilities, such as through the air as cows coughed or in droplets on objects, like work boots. But not enough data had been collected to know how exactly it was happening. Many farmers declined to test their herds, despite an announcement of funds to compensate them for lost milk production in May.
“There is a fear within the dairy farmer community that if they become officially listed as an affected farm, they may lose their milk market,” said Jamie Jonker, chief science officer at the National Milk Producers Federation, an organization that represents dairy farmers. To his knowledge, he added, this hasn’t happened.
Speculation filled knowledge gaps. Zach Riley, head of the Colorado Livestock Association, said he suspected that wild birds may be spreading the virus to herds across the country, despite scientific data suggesting otherwise. Riley said farmers were considering whether to install “floppy inflatable men you see outside of car dealerships” to ward off the birds.
Advisories from agriculture departments to farmers were somewhat speculative, too. Officials recommended biosecurity measures such as disinfecting equipment and limiting visitors. As the virus kept spreading throughout the summer, USDA senior official Eric Deeble said at a press briefing, “The response is adequate.”
The USDA, the Centers for Disease Control and Prevention, and the Food and Drug Administration presented a united front at these briefings, calling it a “One Health” approach. In reality, agriculture agencies took the lead.
This was explicit in an email from a local health department in Colorado to the county’s commissioners. “The State is treating this primarily as an agriculture issue (rightly so) and the public health part is secondary,” wrote Jason Chessher, public health director in Weld County, Colorado. The state’s leading agriculture county, Weld’s livestock and poultry industry produces about $1.9 billion in sales each year.
Patchy Surveillance
In July, the bird flu spread from dairies in Colorado to poultry farms. To contain it, two poultry operations employed about 650 temporary workers — Spanish-speaking immigrants as young as 15 — to cull flocks. Inside hot barns, they caught infected birds, gassed them with carbon dioxide, and disposed of the carcasses. Many did the hazardous job without goggles, face masks, and gloves.
By the time Colorado’s health department asked if workers felt sick, five women and four men had been infected. They all had red, swollen eyes — conjunctivitis — and several had such symptoms as fevers, body aches, and nausea.
State health departments posted online notices offering farms protective gear, but dairy workers in several states told KFF Health News that they had none. They also hadn’t heard about the bird flu, never mind tests for it.
Studies in Colorado, Michigan, and Texas would later show that bird flu cases had gone under the radar. In one analysis, eight dairy workers who hadn’t been tested — 7% of those studied — had antibodies against the virus, a sign that they had been infected.
Missed cases made it impossible to determine how the virus jumped into people and whether it was growing more infectious or dangerous. “I have been distressed and depressed by the lack of epidemiologic data and the lack of surveillance,” said Nicole Lurie, an executive director at the international organization the Coalition for Epidemic Preparedness Innovations, who served as assistant secretary for preparedness and response in the Obama administration.
Citing “insufficient data,” the British government raised its assessment of the risk posed by the US dairy outbreak in July from three to four on a six-tier scale.
Virologists around the world said they were flabbergasted by how poorly the United States was tracking the situation. “You are surrounded by highly pathogenic viruses in the wild and in farm animals,” said Marion Koopmans, head of virology at Erasmus Medical Center in the Netherlands. “If 3 months from now we are at the start of the pandemic, it is nobody’s surprise.”
Although the bird flu is not yet spreading swiftly between people, a shift in that direction could cause immense suffering. The CDC has repeatedly described the cases among farmworkers this year as mild — they weren’t hospitalized. But that doesn’t mean symptoms are a breeze, or that the virus can’t cause worse.
“It does not look pleasant,” wrote Sean Roberts, an emergency services specialist at the Tulare County, California, health department in an email to colleagues in May. He described photographs of an infected dairy worker in another state: “Apparently, the conjunctivitis that this is causing is not a mild one, but rather ruptured blood vessels and bleeding conjunctiva.”
Over the past 30 years, half of around 900 people diagnosed with bird flu around the world have died. Even if the case fatality rate is much lower for this strain of the bird flu, COVID showed how devastating a 1% death rate can be when a virus spreads easily.
Like other cases around the world, the person now hospitalized with the bird flu in Louisiana appears to have gotten the virus directly from birds. After the case was announced, the CDC released a statement saying, “A sporadic case of severe H5N1 bird flu illness in a person is not unexpected.”
‘The Cows Are More Valuable Than Us’
Local health officials were trying hard to track infections, according to hundreds of emails from county health departments in five states. But their efforts were stymied. Even if farmers reported infected herds to the USDA and agriculture agencies told health departments where the infected cows were, health officials had to rely on farm owners for access.
“The agriculture community has dictated the rules of engagement from the start,” said Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota. “That was a big mistake.”
Some farmers told health officials not to visit and declined to monitor their employees for signs of sickness. Sending workers to clinics for testing could leave them shorthanded when cattle needed care. “Producer refuses to send workers to Sunrise [clinic] to get tested since they’re too busy. He has pink eye, too,” said an email from the Weld, Colorado, health department.
“We know of 386 persons exposed — but we know this is far from the total,” said an email from a public health specialist to officials at Tulare’s health department recounting a call with state health officials. “Employers do not want to run this through worker’s compensation. Workers are hesitant to get tested due to cost,” she wrote.
Jennifer Morse, medical director of the Mid-Michigan District Health Department, said local health officials have been hesitant to apply pressure after the backlash many faced at the peak of COVID. Describing the 19 rural counties she serves as “very minimal government–minded,” she said, “if you try to work against them, it will not go well.”
Rural health departments are also stretched thin. Organizations that specialize in outreach to farmworkers offered to assist health officials early in the outbreak, but months passed without contracts or funding. During the first years of COVID, lagging government funds for outreach to farmworkers and other historically marginalized groups led to a disproportionate toll of the disease among people of color.
Kevin Griffis, director of communications at the CDC, said the agency worked with the National Center for Farmworker Health throughout the summer “to reach every farmworker impacted by H5N1.” But Bethany Boggess Alcauter, the center’s director of public health programs, said it didn’t receive a CDC grant for bird flu outreach until October, to the tune of $4 million. Before then, she said, the group had very limited funds for the task. “We are certainly not reaching ‘every farmworker,’” she added.
Farmworker advocates also pressed the CDC for money to offset workers’ financial concerns about testing, including paying for medical care, sick leave, and the risk of being fired. This amounted to an offer of $75 each. “Outreach is clearly not a huge priority,” Boggess said. “I hear over and over from workers, ‘The cows are more valuable than us.’ ”
The USDA has so far put more than $2.1 billion into reimbursing poultry and dairy farmers for losses due to the bird flu and other measures to control the spread on farms. Federal agencies have also put $292 million into developing and stockpiling bird flu vaccines for animals and people. In a controversial decision, the CDC has advised against offering the ones on hand to farmworkers.
“If you want to keep this from becoming a human pandemic, you focus on protecting farmworkers, since that’s the most likely way that this will enter the human population,” said Peg Seminario, an occupational health researcher in Bethesda, Maryland. “The fact that this isn’t happening drives me crazy.”
Nirav Shah, principal deputy director of the CDC, said the agency aims to keep workers safe. “Widespread awareness does take time,” he said. “And that’s the work we’re committed to doing.”
As President-elect Donald Trump comes into office in January, farmworkers may be even less protected. Trump’s pledge of mass deportations will have repercussions whether they happen or not, said Tania Pacheco-Werner, director of the Central Valley Health Policy Institute in California.
Many dairy and poultry workers are living in the United States without authorization or on temporary visas linked to their employers. Such precarity made people less willing to see doctors about COVID symptoms or complain about unsafe working conditions in 2020. Pacheco-Werner said, “Mass deportation is an astronomical challenge for public health.”
Not ‘Immaculate Conception’
A switch flipped in September among experts who study pandemics as national security threats. A patient in Missouri had the bird flu, and no one knew why. “Evidence points to this being a one-off case,” Shah said at a briefing with journalists. About a month later, the agency revealed it was not.
Antibody tests found that a person who lived with the patient had been infected, too. The CDC didn’t know how the two had gotten the virus, and the possibility of human transmission couldn’t be ruled out.
Nonetheless, at an October briefing, Shah said the public risk remained low and Deeble said he was optimistic that the dairy outbreak could be eliminated.
Experts were perturbed by such confident statements in the face of uncertainty, especially as California’s outbreak spiked and a child was mysteriously infected by the same strain of virus found on dairy farms.
“This wasn’t just immaculate conception,” said Stephen Morrison, director of the Global Health Policy Center at the Center for Strategic and International Studies. “It came from somewhere and we don’t know where, but that hasn’t triggered any kind of reset in approach — just the same kind of complacency and low energy.”
Sam Scarpino, a disease surveillance specialist in the Boston area, wondered how many other mysterious infections had gone undetected. Surveillance outside of farms was even patchier than on them, and bird flu tests have been hard to get.
Although pandemic experts had identified the CDC’s singular hold on testing for new viruses as a key explanation for why America was hit so hard by COVID in 2020, the system remained the same. Bird flu tests could be run only by the CDC and public health labs until this month, even though commercial and academic diagnostic laboratories had inquired about running tests since April. The CDC and FDA should have tried to help them along months ago, said Ali Khan, a former top CDC official who now leads the University of Nebraska Medical Center College of Public Health.
As winter sets in, the bird flu becomes harder to spot because patient symptoms may be mistaken for the seasonal flu. Flu season also raises a risk that the two flu viruses could swap genes if they infect a person simultaneously. That could form a hybrid bird flu that spreads swiftly through coughs and sneezes.
A sluggish response to emerging outbreaks may simply be a new, unfortunate norm for America, said Bollyky, at the Council on Foreign Relations. If so, the nation has gotten lucky that the bird flu still can’t spread easily between people. Controlling the virus will be much harder and costlier than it would have been when the outbreak was small. But it’s possible.
Agriculture officials could start testing every silo of bulk milk, in every state, monthly, said Poulsen, the livestock veterinarian. “Not one and done,” he added. If they detect the virus, they’d need to determine the affected farm in time to stop sick cows from spreading infections to the rest of the herd — or at least to other farms. Cows can spread the bird flu before they’re sick, he said, so speed is crucial.
Curtailing the virus on farms is the best way to prevent human infections, said Jennifer Nuzzo, director of the Pandemic Center at Brown University, but human surveillance must be stepped up, too. Every clinic serving communities where farmworkers live should have easy access to bird flu tests — and be encouraged to use them. Funds for farmworker outreach must be boosted. And, she added, the CDC should change its position and offer farmworkers bird flu vaccines to protect them and ward off the chance of a hybrid bird flu that spreads quickly.
The rising number of cases not linked to farms signals a need for more testing in general. When patients are positive on a general flu test — a common diagnostic that indicates human, swine, or bird flu — clinics should probe more deeply, Nuzzo said.
The alternative is a wait-and-see approach in which the nation responds only after enormous damage to lives or businesses. This tack tends to rely on mass vaccination. But an effort analogous to Trump’s Operation Warp Speed is not assured, and neither is rollout like that for the first COVID shots, given a rise in vaccine skepticism among Republican lawmakers.
Change may instead need to start from the bottom up — on dairy farms, still the most common source of human infections, said Poulsen. He noticed a shift in attitudes among farmers at the Dairy Expo: “They’re starting to say, ‘How do I save my dairy for the next generation?’ They recognize how severe this is, and that it’s not just going away.”
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF — the independent source for health policy research, polling, and journalism.
Keith Poulsen’s jaw dropped when farmers showed him images on their cellphones at the World Dairy Expo in Wisconsin in October. A livestock veterinarian at the University of Wisconsin-Madison, Poulsen had seen sick cows before, with their noses dripping and udders slack.
In 14-hour shifts, dairy workers pumped gallons of electrolyte-rich fluids into ailing cows through metal tubes inserted into the esophagus.
“It was like watching a field hospital on an active battlefront treating hundreds of wounded soldiers,” he said.
Nearly a year into the first outbreak of the bird flu among cattle, the virus shows no sign of slowing. The US government failed to eliminate the virus on dairy farms when it was confined to a handful of states, by quickly identifying infected cows and taking measures to keep their infections from spreading. Now at least 875 herds across 16 states have tested positive.
Experts say they have lost faith in the government’s ability to contain the outbreak.
“We are in a terrible situation and going into a worse situation,” said Angela Rasmussen, a virologist at the University of Saskatchewan in Canada. “I don’t know if the bird flu will become a pandemic, but if it does, we are screwed.”
To understand how the bird flu got out of hand, KFF Health News interviewed nearly 70 government officials, farmers and farmworkers, and researchers with expertise in virology, pandemics, veterinary medicine, and more.
Together with emails obtained from local health departments through public records requests, this investigation revealed key problems, including deference to the farm industry, eroded public health budgets, neglect for the safety of agriculture workers, and the sluggish pace of federal interventions.
Case in point: The US Department of Agriculture this month announced a federal order to test milk nationwide. Researchers welcomed the news but said it should have happened months ago — before the virus was so entrenched.
“It’s disheartening to see so many of the same failures that emerged during the COVID-19 crisis reemerge,” said Tom Bollyky, director of the Global Health Program at the Council on Foreign Relations.
Far more bird flu damage is inevitable, but the extent of it will be left to the Trump administration and Mother Nature. Already, the USDA has funneled more than $1.7 billion into tamping down the bird flu on poultry farms since 2022, which includes reimbursing farmers who’ve had to cull their flocks, and more than $430 million into combating the bird flu on dairy farms. In coming years, the bird flu may cost billions of dollars more in expenses and losses. Dairy industry experts say the virus kills roughly 2%-5% of infected dairy cows and reduces a herd’s milk production by about 20%.
Worse, the outbreak poses the threat of a pandemic. More than 60 people in the US have been infected, mainly by cows or poultry, but cases could skyrocket if the virus evolves to spread efficiently from person to person. And the recent news of a person critically ill in Louisiana with the bird flu shows that the virus can be dangerous.
Just a few mutations could allow the bird flu to spread between people. Because viruses mutate within human and animal bodies, each infection is like a pull of a slot machine lever.
“Even if there’s only a 5% chance of a bird flu pandemic happening, we’re talking about a pandemic that probably looks like 2020 or worse,” said Tom Peacock, a bird flu researcher at the Pirbright Institute in the United Kingdom, referring to COVID. “The US knows the risk but hasn’t done anything to slow this down,” he added.
Beyond the bird flu, the federal government’s handling of the outbreak reveals cracks in the US health security system that would allow other risky new pathogens to take root. “This virus may not be the one that takes off,” said Maria Van Kerkhove, director of the emerging diseases group at the World Health Organization. “But this is a real-fire exercise right now, and it demonstrates what needs to be improved.”
A Slow Start
It may have been a grackle, a goose, or some other wild bird that infected a cow in northern Texas. In February, the state’s dairy farmers took note when cows stopped making milk. They worked alongside veterinarians to figure out why. In less than two months, veterinary researchers identified the highly pathogenic H5N1 bird flu virus as the culprit.
Long listed among pathogens with pandemic potential, the bird flu’s unprecedented spread among cows marked a worrying shift. It had evolved to thrive in animals that are more like people biologically than birds.
After the USDA announced the dairy outbreak on March 25, control shifted from farmers, veterinarians, and local officials to state and federal agencies. Collaboration disintegrated almost immediately.
Farmers worried the government might block their milk sales or even demand sick cows be killed, as poultry are, said Kay Russo, a livestock veterinarian in Fort Collins, Colorado.
Instead, Russo and other veterinarians said, they were dismayed by inaction. The USDA didn’t respond to their urgent requests to support studies on dairy farms — and for money and confidentiality policies to protect farmers from financial loss if they agreed to test animals.
The USDA announced that it would conduct studies itself. But researchers grew anxious as weeks passed without results. “Probably the biggest mistake from the USDA was not involving the boots-on-the-ground veterinarians,” Russo said.
Will Clement, a USDA senior adviser for communications, said in an email: “Since first learning of H5N1 in dairy cattle in late March 2024, USDA has worked swiftly and diligently to assess the prevalence of the virus in US dairy herds.” The agency provided research funds to state and national animal health labs beginning in April, he added.
The USDA didn’t require lactating cows to be tested before interstate travel until April 29. By then, the outbreak had spread to eight other states. Farmers often move cattle across great distances, for calving in one place, raising in warm, dry climates, and milking in cooler ones. Analyses of the virus’s genes implied that it spread between cows rather than repeatedly jumping from birds into herds.
Milking equipment was a likely source of infection, and there were hints of other possibilities, such as through the air as cows coughed or in droplets on objects, like work boots. But not enough data had been collected to know how exactly it was happening. Many farmers declined to test their herds, despite an announcement of funds to compensate them for lost milk production in May.
“There is a fear within the dairy farmer community that if they become officially listed as an affected farm, they may lose their milk market,” said Jamie Jonker, chief science officer at the National Milk Producers Federation, an organization that represents dairy farmers. To his knowledge, he added, this hasn’t happened.
Speculation filled knowledge gaps. Zach Riley, head of the Colorado Livestock Association, said he suspected that wild birds may be spreading the virus to herds across the country, despite scientific data suggesting otherwise. Riley said farmers were considering whether to install “floppy inflatable men you see outside of car dealerships” to ward off the birds.
Advisories from agriculture departments to farmers were somewhat speculative, too. Officials recommended biosecurity measures such as disinfecting equipment and limiting visitors. As the virus kept spreading throughout the summer, USDA senior official Eric Deeble said at a press briefing, “The response is adequate.”
The USDA, the Centers for Disease Control and Prevention, and the Food and Drug Administration presented a united front at these briefings, calling it a “One Health” approach. In reality, agriculture agencies took the lead.
This was explicit in an email from a local health department in Colorado to the county’s commissioners. “The State is treating this primarily as an agriculture issue (rightly so) and the public health part is secondary,” wrote Jason Chessher, public health director in Weld County, Colorado. The state’s leading agriculture county, Weld’s livestock and poultry industry produces about $1.9 billion in sales each year.
Patchy Surveillance
In July, the bird flu spread from dairies in Colorado to poultry farms. To contain it, two poultry operations employed about 650 temporary workers — Spanish-speaking immigrants as young as 15 — to cull flocks. Inside hot barns, they caught infected birds, gassed them with carbon dioxide, and disposed of the carcasses. Many did the hazardous job without goggles, face masks, and gloves.
By the time Colorado’s health department asked if workers felt sick, five women and four men had been infected. They all had red, swollen eyes — conjunctivitis — and several had such symptoms as fevers, body aches, and nausea.
State health departments posted online notices offering farms protective gear, but dairy workers in several states told KFF Health News that they had none. They also hadn’t heard about the bird flu, never mind tests for it.
Studies in Colorado, Michigan, and Texas would later show that bird flu cases had gone under the radar. In one analysis, eight dairy workers who hadn’t been tested — 7% of those studied — had antibodies against the virus, a sign that they had been infected.
Missed cases made it impossible to determine how the virus jumped into people and whether it was growing more infectious or dangerous. “I have been distressed and depressed by the lack of epidemiologic data and the lack of surveillance,” said Nicole Lurie, an executive director at the international organization the Coalition for Epidemic Preparedness Innovations, who served as assistant secretary for preparedness and response in the Obama administration.
Citing “insufficient data,” the British government raised its assessment of the risk posed by the US dairy outbreak in July from three to four on a six-tier scale.
Virologists around the world said they were flabbergasted by how poorly the United States was tracking the situation. “You are surrounded by highly pathogenic viruses in the wild and in farm animals,” said Marion Koopmans, head of virology at Erasmus Medical Center in the Netherlands. “If 3 months from now we are at the start of the pandemic, it is nobody’s surprise.”
Although the bird flu is not yet spreading swiftly between people, a shift in that direction could cause immense suffering. The CDC has repeatedly described the cases among farmworkers this year as mild — they weren’t hospitalized. But that doesn’t mean symptoms are a breeze, or that the virus can’t cause worse.
“It does not look pleasant,” wrote Sean Roberts, an emergency services specialist at the Tulare County, California, health department in an email to colleagues in May. He described photographs of an infected dairy worker in another state: “Apparently, the conjunctivitis that this is causing is not a mild one, but rather ruptured blood vessels and bleeding conjunctiva.”
Over the past 30 years, half of around 900 people diagnosed with bird flu around the world have died. Even if the case fatality rate is much lower for this strain of the bird flu, COVID showed how devastating a 1% death rate can be when a virus spreads easily.
Like other cases around the world, the person now hospitalized with the bird flu in Louisiana appears to have gotten the virus directly from birds. After the case was announced, the CDC released a statement saying, “A sporadic case of severe H5N1 bird flu illness in a person is not unexpected.”
‘The Cows Are More Valuable Than Us’
Local health officials were trying hard to track infections, according to hundreds of emails from county health departments in five states. But their efforts were stymied. Even if farmers reported infected herds to the USDA and agriculture agencies told health departments where the infected cows were, health officials had to rely on farm owners for access.
“The agriculture community has dictated the rules of engagement from the start,” said Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota. “That was a big mistake.”
Some farmers told health officials not to visit and declined to monitor their employees for signs of sickness. Sending workers to clinics for testing could leave them shorthanded when cattle needed care. “Producer refuses to send workers to Sunrise [clinic] to get tested since they’re too busy. He has pink eye, too,” said an email from the Weld, Colorado, health department.
“We know of 386 persons exposed — but we know this is far from the total,” said an email from a public health specialist to officials at Tulare’s health department recounting a call with state health officials. “Employers do not want to run this through worker’s compensation. Workers are hesitant to get tested due to cost,” she wrote.
Jennifer Morse, medical director of the Mid-Michigan District Health Department, said local health officials have been hesitant to apply pressure after the backlash many faced at the peak of COVID. Describing the 19 rural counties she serves as “very minimal government–minded,” she said, “if you try to work against them, it will not go well.”
Rural health departments are also stretched thin. Organizations that specialize in outreach to farmworkers offered to assist health officials early in the outbreak, but months passed without contracts or funding. During the first years of COVID, lagging government funds for outreach to farmworkers and other historically marginalized groups led to a disproportionate toll of the disease among people of color.
Kevin Griffis, director of communications at the CDC, said the agency worked with the National Center for Farmworker Health throughout the summer “to reach every farmworker impacted by H5N1.” But Bethany Boggess Alcauter, the center’s director of public health programs, said it didn’t receive a CDC grant for bird flu outreach until October, to the tune of $4 million. Before then, she said, the group had very limited funds for the task. “We are certainly not reaching ‘every farmworker,’” she added.
Farmworker advocates also pressed the CDC for money to offset workers’ financial concerns about testing, including paying for medical care, sick leave, and the risk of being fired. This amounted to an offer of $75 each. “Outreach is clearly not a huge priority,” Boggess said. “I hear over and over from workers, ‘The cows are more valuable than us.’ ”
The USDA has so far put more than $2.1 billion into reimbursing poultry and dairy farmers for losses due to the bird flu and other measures to control the spread on farms. Federal agencies have also put $292 million into developing and stockpiling bird flu vaccines for animals and people. In a controversial decision, the CDC has advised against offering the ones on hand to farmworkers.
“If you want to keep this from becoming a human pandemic, you focus on protecting farmworkers, since that’s the most likely way that this will enter the human population,” said Peg Seminario, an occupational health researcher in Bethesda, Maryland. “The fact that this isn’t happening drives me crazy.”
Nirav Shah, principal deputy director of the CDC, said the agency aims to keep workers safe. “Widespread awareness does take time,” he said. “And that’s the work we’re committed to doing.”
As President-elect Donald Trump comes into office in January, farmworkers may be even less protected. Trump’s pledge of mass deportations will have repercussions whether they happen or not, said Tania Pacheco-Werner, director of the Central Valley Health Policy Institute in California.
Many dairy and poultry workers are living in the United States without authorization or on temporary visas linked to their employers. Such precarity made people less willing to see doctors about COVID symptoms or complain about unsafe working conditions in 2020. Pacheco-Werner said, “Mass deportation is an astronomical challenge for public health.”
Not ‘Immaculate Conception’
A switch flipped in September among experts who study pandemics as national security threats. A patient in Missouri had the bird flu, and no one knew why. “Evidence points to this being a one-off case,” Shah said at a briefing with journalists. About a month later, the agency revealed it was not.
Antibody tests found that a person who lived with the patient had been infected, too. The CDC didn’t know how the two had gotten the virus, and the possibility of human transmission couldn’t be ruled out.
Nonetheless, at an October briefing, Shah said the public risk remained low and Deeble said he was optimistic that the dairy outbreak could be eliminated.
Experts were perturbed by such confident statements in the face of uncertainty, especially as California’s outbreak spiked and a child was mysteriously infected by the same strain of virus found on dairy farms.
“This wasn’t just immaculate conception,” said Stephen Morrison, director of the Global Health Policy Center at the Center for Strategic and International Studies. “It came from somewhere and we don’t know where, but that hasn’t triggered any kind of reset in approach — just the same kind of complacency and low energy.”
Sam Scarpino, a disease surveillance specialist in the Boston area, wondered how many other mysterious infections had gone undetected. Surveillance outside of farms was even patchier than on them, and bird flu tests have been hard to get.
Although pandemic experts had identified the CDC’s singular hold on testing for new viruses as a key explanation for why America was hit so hard by COVID in 2020, the system remained the same. Bird flu tests could be run only by the CDC and public health labs until this month, even though commercial and academic diagnostic laboratories had inquired about running tests since April. The CDC and FDA should have tried to help them along months ago, said Ali Khan, a former top CDC official who now leads the University of Nebraska Medical Center College of Public Health.
As winter sets in, the bird flu becomes harder to spot because patient symptoms may be mistaken for the seasonal flu. Flu season also raises a risk that the two flu viruses could swap genes if they infect a person simultaneously. That could form a hybrid bird flu that spreads swiftly through coughs and sneezes.
A sluggish response to emerging outbreaks may simply be a new, unfortunate norm for America, said Bollyky, at the Council on Foreign Relations. If so, the nation has gotten lucky that the bird flu still can’t spread easily between people. Controlling the virus will be much harder and costlier than it would have been when the outbreak was small. But it’s possible.
Agriculture officials could start testing every silo of bulk milk, in every state, monthly, said Poulsen, the livestock veterinarian. “Not one and done,” he added. If they detect the virus, they’d need to determine the affected farm in time to stop sick cows from spreading infections to the rest of the herd — or at least to other farms. Cows can spread the bird flu before they’re sick, he said, so speed is crucial.
Curtailing the virus on farms is the best way to prevent human infections, said Jennifer Nuzzo, director of the Pandemic Center at Brown University, but human surveillance must be stepped up, too. Every clinic serving communities where farmworkers live should have easy access to bird flu tests — and be encouraged to use them. Funds for farmworker outreach must be boosted. And, she added, the CDC should change its position and offer farmworkers bird flu vaccines to protect them and ward off the chance of a hybrid bird flu that spreads quickly.
The rising number of cases not linked to farms signals a need for more testing in general. When patients are positive on a general flu test — a common diagnostic that indicates human, swine, or bird flu — clinics should probe more deeply, Nuzzo said.
The alternative is a wait-and-see approach in which the nation responds only after enormous damage to lives or businesses. This tack tends to rely on mass vaccination. But an effort analogous to Trump’s Operation Warp Speed is not assured, and neither is rollout like that for the first COVID shots, given a rise in vaccine skepticism among Republican lawmakers.
Change may instead need to start from the bottom up — on dairy farms, still the most common source of human infections, said Poulsen. He noticed a shift in attitudes among farmers at the Dairy Expo: “They’re starting to say, ‘How do I save my dairy for the next generation?’ They recognize how severe this is, and that it’s not just going away.”
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF — the independent source for health policy research, polling, and journalism.
Keith Poulsen’s jaw dropped when farmers showed him images on their cellphones at the World Dairy Expo in Wisconsin in October. A livestock veterinarian at the University of Wisconsin-Madison, Poulsen had seen sick cows before, with their noses dripping and udders slack.
In 14-hour shifts, dairy workers pumped gallons of electrolyte-rich fluids into ailing cows through metal tubes inserted into the esophagus.
“It was like watching a field hospital on an active battlefront treating hundreds of wounded soldiers,” he said.
Nearly a year into the first outbreak of the bird flu among cattle, the virus shows no sign of slowing. The US government failed to eliminate the virus on dairy farms when it was confined to a handful of states, by quickly identifying infected cows and taking measures to keep their infections from spreading. Now at least 875 herds across 16 states have tested positive.
Experts say they have lost faith in the government’s ability to contain the outbreak.
“We are in a terrible situation and going into a worse situation,” said Angela Rasmussen, a virologist at the University of Saskatchewan in Canada. “I don’t know if the bird flu will become a pandemic, but if it does, we are screwed.”
To understand how the bird flu got out of hand, KFF Health News interviewed nearly 70 government officials, farmers and farmworkers, and researchers with expertise in virology, pandemics, veterinary medicine, and more.
Together with emails obtained from local health departments through public records requests, this investigation revealed key problems, including deference to the farm industry, eroded public health budgets, neglect for the safety of agriculture workers, and the sluggish pace of federal interventions.
Case in point: The US Department of Agriculture this month announced a federal order to test milk nationwide. Researchers welcomed the news but said it should have happened months ago — before the virus was so entrenched.
“It’s disheartening to see so many of the same failures that emerged during the COVID-19 crisis reemerge,” said Tom Bollyky, director of the Global Health Program at the Council on Foreign Relations.
Far more bird flu damage is inevitable, but the extent of it will be left to the Trump administration and Mother Nature. Already, the USDA has funneled more than $1.7 billion into tamping down the bird flu on poultry farms since 2022, which includes reimbursing farmers who’ve had to cull their flocks, and more than $430 million into combating the bird flu on dairy farms. In coming years, the bird flu may cost billions of dollars more in expenses and losses. Dairy industry experts say the virus kills roughly 2%-5% of infected dairy cows and reduces a herd’s milk production by about 20%.
Worse, the outbreak poses the threat of a pandemic. More than 60 people in the US have been infected, mainly by cows or poultry, but cases could skyrocket if the virus evolves to spread efficiently from person to person. And the recent news of a person critically ill in Louisiana with the bird flu shows that the virus can be dangerous.
Just a few mutations could allow the bird flu to spread between people. Because viruses mutate within human and animal bodies, each infection is like a pull of a slot machine lever.
“Even if there’s only a 5% chance of a bird flu pandemic happening, we’re talking about a pandemic that probably looks like 2020 or worse,” said Tom Peacock, a bird flu researcher at the Pirbright Institute in the United Kingdom, referring to COVID. “The US knows the risk but hasn’t done anything to slow this down,” he added.
Beyond the bird flu, the federal government’s handling of the outbreak reveals cracks in the US health security system that would allow other risky new pathogens to take root. “This virus may not be the one that takes off,” said Maria Van Kerkhove, director of the emerging diseases group at the World Health Organization. “But this is a real-fire exercise right now, and it demonstrates what needs to be improved.”
A Slow Start
It may have been a grackle, a goose, or some other wild bird that infected a cow in northern Texas. In February, the state’s dairy farmers took note when cows stopped making milk. They worked alongside veterinarians to figure out why. In less than two months, veterinary researchers identified the highly pathogenic H5N1 bird flu virus as the culprit.
Long listed among pathogens with pandemic potential, the bird flu’s unprecedented spread among cows marked a worrying shift. It had evolved to thrive in animals that are more like people biologically than birds.
After the USDA announced the dairy outbreak on March 25, control shifted from farmers, veterinarians, and local officials to state and federal agencies. Collaboration disintegrated almost immediately.
Farmers worried the government might block their milk sales or even demand sick cows be killed, as poultry are, said Kay Russo, a livestock veterinarian in Fort Collins, Colorado.
Instead, Russo and other veterinarians said, they were dismayed by inaction. The USDA didn’t respond to their urgent requests to support studies on dairy farms — and for money and confidentiality policies to protect farmers from financial loss if they agreed to test animals.
The USDA announced that it would conduct studies itself. But researchers grew anxious as weeks passed without results. “Probably the biggest mistake from the USDA was not involving the boots-on-the-ground veterinarians,” Russo said.
Will Clement, a USDA senior adviser for communications, said in an email: “Since first learning of H5N1 in dairy cattle in late March 2024, USDA has worked swiftly and diligently to assess the prevalence of the virus in US dairy herds.” The agency provided research funds to state and national animal health labs beginning in April, he added.
The USDA didn’t require lactating cows to be tested before interstate travel until April 29. By then, the outbreak had spread to eight other states. Farmers often move cattle across great distances, for calving in one place, raising in warm, dry climates, and milking in cooler ones. Analyses of the virus’s genes implied that it spread between cows rather than repeatedly jumping from birds into herds.
Milking equipment was a likely source of infection, and there were hints of other possibilities, such as through the air as cows coughed or in droplets on objects, like work boots. But not enough data had been collected to know how exactly it was happening. Many farmers declined to test their herds, despite an announcement of funds to compensate them for lost milk production in May.
“There is a fear within the dairy farmer community that if they become officially listed as an affected farm, they may lose their milk market,” said Jamie Jonker, chief science officer at the National Milk Producers Federation, an organization that represents dairy farmers. To his knowledge, he added, this hasn’t happened.
Speculation filled knowledge gaps. Zach Riley, head of the Colorado Livestock Association, said he suspected that wild birds may be spreading the virus to herds across the country, despite scientific data suggesting otherwise. Riley said farmers were considering whether to install “floppy inflatable men you see outside of car dealerships” to ward off the birds.
Advisories from agriculture departments to farmers were somewhat speculative, too. Officials recommended biosecurity measures such as disinfecting equipment and limiting visitors. As the virus kept spreading throughout the summer, USDA senior official Eric Deeble said at a press briefing, “The response is adequate.”
The USDA, the Centers for Disease Control and Prevention, and the Food and Drug Administration presented a united front at these briefings, calling it a “One Health” approach. In reality, agriculture agencies took the lead.
This was explicit in an email from a local health department in Colorado to the county’s commissioners. “The State is treating this primarily as an agriculture issue (rightly so) and the public health part is secondary,” wrote Jason Chessher, public health director in Weld County, Colorado. The state’s leading agriculture county, Weld’s livestock and poultry industry produces about $1.9 billion in sales each year.
Patchy Surveillance
In July, the bird flu spread from dairies in Colorado to poultry farms. To contain it, two poultry operations employed about 650 temporary workers — Spanish-speaking immigrants as young as 15 — to cull flocks. Inside hot barns, they caught infected birds, gassed them with carbon dioxide, and disposed of the carcasses. Many did the hazardous job without goggles, face masks, and gloves.
By the time Colorado’s health department asked if workers felt sick, five women and four men had been infected. They all had red, swollen eyes — conjunctivitis — and several had such symptoms as fevers, body aches, and nausea.
State health departments posted online notices offering farms protective gear, but dairy workers in several states told KFF Health News that they had none. They also hadn’t heard about the bird flu, never mind tests for it.
Studies in Colorado, Michigan, and Texas would later show that bird flu cases had gone under the radar. In one analysis, eight dairy workers who hadn’t been tested — 7% of those studied — had antibodies against the virus, a sign that they had been infected.
Missed cases made it impossible to determine how the virus jumped into people and whether it was growing more infectious or dangerous. “I have been distressed and depressed by the lack of epidemiologic data and the lack of surveillance,” said Nicole Lurie, an executive director at the international organization the Coalition for Epidemic Preparedness Innovations, who served as assistant secretary for preparedness and response in the Obama administration.
Citing “insufficient data,” the British government raised its assessment of the risk posed by the US dairy outbreak in July from three to four on a six-tier scale.
Virologists around the world said they were flabbergasted by how poorly the United States was tracking the situation. “You are surrounded by highly pathogenic viruses in the wild and in farm animals,” said Marion Koopmans, head of virology at Erasmus Medical Center in the Netherlands. “If 3 months from now we are at the start of the pandemic, it is nobody’s surprise.”
Although the bird flu is not yet spreading swiftly between people, a shift in that direction could cause immense suffering. The CDC has repeatedly described the cases among farmworkers this year as mild — they weren’t hospitalized. But that doesn’t mean symptoms are a breeze, or that the virus can’t cause worse.
“It does not look pleasant,” wrote Sean Roberts, an emergency services specialist at the Tulare County, California, health department in an email to colleagues in May. He described photographs of an infected dairy worker in another state: “Apparently, the conjunctivitis that this is causing is not a mild one, but rather ruptured blood vessels and bleeding conjunctiva.”
Over the past 30 years, half of around 900 people diagnosed with bird flu around the world have died. Even if the case fatality rate is much lower for this strain of the bird flu, COVID showed how devastating a 1% death rate can be when a virus spreads easily.
Like other cases around the world, the person now hospitalized with the bird flu in Louisiana appears to have gotten the virus directly from birds. After the case was announced, the CDC released a statement saying, “A sporadic case of severe H5N1 bird flu illness in a person is not unexpected.”
‘The Cows Are More Valuable Than Us’
Local health officials were trying hard to track infections, according to hundreds of emails from county health departments in five states. But their efforts were stymied. Even if farmers reported infected herds to the USDA and agriculture agencies told health departments where the infected cows were, health officials had to rely on farm owners for access.
“The agriculture community has dictated the rules of engagement from the start,” said Michael Osterholm, director of the Center for Infectious Disease Research and Policy at the University of Minnesota. “That was a big mistake.”
Some farmers told health officials not to visit and declined to monitor their employees for signs of sickness. Sending workers to clinics for testing could leave them shorthanded when cattle needed care. “Producer refuses to send workers to Sunrise [clinic] to get tested since they’re too busy. He has pink eye, too,” said an email from the Weld, Colorado, health department.
“We know of 386 persons exposed — but we know this is far from the total,” said an email from a public health specialist to officials at Tulare’s health department recounting a call with state health officials. “Employers do not want to run this through worker’s compensation. Workers are hesitant to get tested due to cost,” she wrote.
Jennifer Morse, medical director of the Mid-Michigan District Health Department, said local health officials have been hesitant to apply pressure after the backlash many faced at the peak of COVID. Describing the 19 rural counties she serves as “very minimal government–minded,” she said, “if you try to work against them, it will not go well.”
Rural health departments are also stretched thin. Organizations that specialize in outreach to farmworkers offered to assist health officials early in the outbreak, but months passed without contracts or funding. During the first years of COVID, lagging government funds for outreach to farmworkers and other historically marginalized groups led to a disproportionate toll of the disease among people of color.
Kevin Griffis, director of communications at the CDC, said the agency worked with the National Center for Farmworker Health throughout the summer “to reach every farmworker impacted by H5N1.” But Bethany Boggess Alcauter, the center’s director of public health programs, said it didn’t receive a CDC grant for bird flu outreach until October, to the tune of $4 million. Before then, she said, the group had very limited funds for the task. “We are certainly not reaching ‘every farmworker,’” she added.
Farmworker advocates also pressed the CDC for money to offset workers’ financial concerns about testing, including paying for medical care, sick leave, and the risk of being fired. This amounted to an offer of $75 each. “Outreach is clearly not a huge priority,” Boggess said. “I hear over and over from workers, ‘The cows are more valuable than us.’ ”
The USDA has so far put more than $2.1 billion into reimbursing poultry and dairy farmers for losses due to the bird flu and other measures to control the spread on farms. Federal agencies have also put $292 million into developing and stockpiling bird flu vaccines for animals and people. In a controversial decision, the CDC has advised against offering the ones on hand to farmworkers.
“If you want to keep this from becoming a human pandemic, you focus on protecting farmworkers, since that’s the most likely way that this will enter the human population,” said Peg Seminario, an occupational health researcher in Bethesda, Maryland. “The fact that this isn’t happening drives me crazy.”
Nirav Shah, principal deputy director of the CDC, said the agency aims to keep workers safe. “Widespread awareness does take time,” he said. “And that’s the work we’re committed to doing.”
As President-elect Donald Trump comes into office in January, farmworkers may be even less protected. Trump’s pledge of mass deportations will have repercussions whether they happen or not, said Tania Pacheco-Werner, director of the Central Valley Health Policy Institute in California.
Many dairy and poultry workers are living in the United States without authorization or on temporary visas linked to their employers. Such precarity made people less willing to see doctors about COVID symptoms or complain about unsafe working conditions in 2020. Pacheco-Werner said, “Mass deportation is an astronomical challenge for public health.”
Not ‘Immaculate Conception’
A switch flipped in September among experts who study pandemics as national security threats. A patient in Missouri had the bird flu, and no one knew why. “Evidence points to this being a one-off case,” Shah said at a briefing with journalists. About a month later, the agency revealed it was not.
Antibody tests found that a person who lived with the patient had been infected, too. The CDC didn’t know how the two had gotten the virus, and the possibility of human transmission couldn’t be ruled out.
Nonetheless, at an October briefing, Shah said the public risk remained low and Deeble said he was optimistic that the dairy outbreak could be eliminated.
Experts were perturbed by such confident statements in the face of uncertainty, especially as California’s outbreak spiked and a child was mysteriously infected by the same strain of virus found on dairy farms.
“This wasn’t just immaculate conception,” said Stephen Morrison, director of the Global Health Policy Center at the Center for Strategic and International Studies. “It came from somewhere and we don’t know where, but that hasn’t triggered any kind of reset in approach — just the same kind of complacency and low energy.”
Sam Scarpino, a disease surveillance specialist in the Boston area, wondered how many other mysterious infections had gone undetected. Surveillance outside of farms was even patchier than on them, and bird flu tests have been hard to get.
Although pandemic experts had identified the CDC’s singular hold on testing for new viruses as a key explanation for why America was hit so hard by COVID in 2020, the system remained the same. Bird flu tests could be run only by the CDC and public health labs until this month, even though commercial and academic diagnostic laboratories had inquired about running tests since April. The CDC and FDA should have tried to help them along months ago, said Ali Khan, a former top CDC official who now leads the University of Nebraska Medical Center College of Public Health.
As winter sets in, the bird flu becomes harder to spot because patient symptoms may be mistaken for the seasonal flu. Flu season also raises a risk that the two flu viruses could swap genes if they infect a person simultaneously. That could form a hybrid bird flu that spreads swiftly through coughs and sneezes.
A sluggish response to emerging outbreaks may simply be a new, unfortunate norm for America, said Bollyky, at the Council on Foreign Relations. If so, the nation has gotten lucky that the bird flu still can’t spread easily between people. Controlling the virus will be much harder and costlier than it would have been when the outbreak was small. But it’s possible.
Agriculture officials could start testing every silo of bulk milk, in every state, monthly, said Poulsen, the livestock veterinarian. “Not one and done,” he added. If they detect the virus, they’d need to determine the affected farm in time to stop sick cows from spreading infections to the rest of the herd — or at least to other farms. Cows can spread the bird flu before they’re sick, he said, so speed is crucial.
Curtailing the virus on farms is the best way to prevent human infections, said Jennifer Nuzzo, director of the Pandemic Center at Brown University, but human surveillance must be stepped up, too. Every clinic serving communities where farmworkers live should have easy access to bird flu tests — and be encouraged to use them. Funds for farmworker outreach must be boosted. And, she added, the CDC should change its position and offer farmworkers bird flu vaccines to protect them and ward off the chance of a hybrid bird flu that spreads quickly.
The rising number of cases not linked to farms signals a need for more testing in general. When patients are positive on a general flu test — a common diagnostic that indicates human, swine, or bird flu — clinics should probe more deeply, Nuzzo said.
The alternative is a wait-and-see approach in which the nation responds only after enormous damage to lives or businesses. This tack tends to rely on mass vaccination. But an effort analogous to Trump’s Operation Warp Speed is not assured, and neither is rollout like that for the first COVID shots, given a rise in vaccine skepticism among Republican lawmakers.
Change may instead need to start from the bottom up — on dairy farms, still the most common source of human infections, said Poulsen. He noticed a shift in attitudes among farmers at the Dairy Expo: “They’re starting to say, ‘How do I save my dairy for the next generation?’ They recognize how severe this is, and that it’s not just going away.”
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF — the independent source for health policy research, polling, and journalism.
Does Virtual Care for UTIs Lead to Increased Antibiotic Use Without Better Outcomes?
TOPLINE:
Virtual visits for urinary tract infections (UTIs) increased by more than 600% from 2015 to 2022, with overall UTI encounters growing by 325.9%. The rate of antibiotic dispensation climbed by 227.3% per 1000 patients, outpacing the 159.8% increase in positive urine cultures.
METHODOLOGY:
- Researchers conducted a retrospective cohort study analyzing 1,220,698 UTI encounters among 428,855 nonpregnant women aged ≥ 18 years at Kaiser Permanente Southern California from 2015 to 2022.
- Analysis included outpatient UTI encounters in ambulatory and urgent care settings, excluding emergency and inpatient visits.
- Data collection encompassed demographic information, urine tests, antibiotic dispensation, and UTI diagnoses using International Classification of Diseases, 9th and 10th Revision codes.
- Encounters conducted by physicians, physician assistants, nurse practitioners, and registered nurses through in-person, phone, video, and health portal platforms were evaluated.
TAKEAWAY:
- Virtual encounters grew by 603.2% compared with a 122.8% increase for in-person visits, with virtual visits accounting for 60% (733,263) of all UTI encounters.
- The rate of UTI encounters per 1000 adult female patients increased by 241.6%, while membership in the health system grew by only 24.4%.
- Antibiotics were prescribed without urine testing in 42.5% (519,135) of encounters, and among encounters with both antibiotic dispensation and urine testing, 57.1% (278,903) had a positive culture.
- According to the authors, the increasing rate of antibiotic dispensation surpassed the growth in positive urine culture rates, suggesting increased use of empiric antibiotics.
IN PRACTICE:
“Our findings underscore the importance of balancing telemedicine’s accessibility with maintaining antibiotic stewardship and highlight the need for updated guidelines,” wrote the authors of the study. An accompanying editorial said, “Unfortunately, our misguided conceptual model has led to several decades of UTI research focusing on bad bugs rather than investigating the natural host defenses, how we might boost these, what perturbs the ecosystem, and how microbial defense occurs within the bladder.”
SOURCE:
The study was led by Ghanshyam Yadav, MD, Kaiser Permanente Southern California in San Diego. It was published online in Obstetrics & Gynecology. The editorial, written by Nazema Y. Siddiqui, MD, MHSc, from the Department of Obstetrics and Gynecology, Duke University Medical Center, Durham, North Carolina, was also published in Obstetrics & Gynecology.
LIMITATIONS:
The retrospective design and analysis at the encounter level did not allow for control of patient and clinician clustering. The study was limited to a single health maintenance organization, which may affect the generalizability of the findings.
DISCLOSURES:
This research received support through a grant from the Regional Research Committee of Kaiser Permanente Southern California (RRC grant number: KP-RRC-20221002). Heidi Brown and Jasmine Tan-Kim disclosed receiving royalties from UpToDate. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Virtual visits for urinary tract infections (UTIs) increased by more than 600% from 2015 to 2022, with overall UTI encounters growing by 325.9%. The rate of antibiotic dispensation climbed by 227.3% per 1000 patients, outpacing the 159.8% increase in positive urine cultures.
METHODOLOGY:
- Researchers conducted a retrospective cohort study analyzing 1,220,698 UTI encounters among 428,855 nonpregnant women aged ≥ 18 years at Kaiser Permanente Southern California from 2015 to 2022.
- Analysis included outpatient UTI encounters in ambulatory and urgent care settings, excluding emergency and inpatient visits.
- Data collection encompassed demographic information, urine tests, antibiotic dispensation, and UTI diagnoses using International Classification of Diseases, 9th and 10th Revision codes.
- Encounters conducted by physicians, physician assistants, nurse practitioners, and registered nurses through in-person, phone, video, and health portal platforms were evaluated.
TAKEAWAY:
- Virtual encounters grew by 603.2% compared with a 122.8% increase for in-person visits, with virtual visits accounting for 60% (733,263) of all UTI encounters.
- The rate of UTI encounters per 1000 adult female patients increased by 241.6%, while membership in the health system grew by only 24.4%.
- Antibiotics were prescribed without urine testing in 42.5% (519,135) of encounters, and among encounters with both antibiotic dispensation and urine testing, 57.1% (278,903) had a positive culture.
- According to the authors, the increasing rate of antibiotic dispensation surpassed the growth in positive urine culture rates, suggesting increased use of empiric antibiotics.
IN PRACTICE:
“Our findings underscore the importance of balancing telemedicine’s accessibility with maintaining antibiotic stewardship and highlight the need for updated guidelines,” wrote the authors of the study. An accompanying editorial said, “Unfortunately, our misguided conceptual model has led to several decades of UTI research focusing on bad bugs rather than investigating the natural host defenses, how we might boost these, what perturbs the ecosystem, and how microbial defense occurs within the bladder.”
SOURCE:
The study was led by Ghanshyam Yadav, MD, Kaiser Permanente Southern California in San Diego. It was published online in Obstetrics & Gynecology. The editorial, written by Nazema Y. Siddiqui, MD, MHSc, from the Department of Obstetrics and Gynecology, Duke University Medical Center, Durham, North Carolina, was also published in Obstetrics & Gynecology.
LIMITATIONS:
The retrospective design and analysis at the encounter level did not allow for control of patient and clinician clustering. The study was limited to a single health maintenance organization, which may affect the generalizability of the findings.
DISCLOSURES:
This research received support through a grant from the Regional Research Committee of Kaiser Permanente Southern California (RRC grant number: KP-RRC-20221002). Heidi Brown and Jasmine Tan-Kim disclosed receiving royalties from UpToDate. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Virtual visits for urinary tract infections (UTIs) increased by more than 600% from 2015 to 2022, with overall UTI encounters growing by 325.9%. The rate of antibiotic dispensation climbed by 227.3% per 1000 patients, outpacing the 159.8% increase in positive urine cultures.
METHODOLOGY:
- Researchers conducted a retrospective cohort study analyzing 1,220,698 UTI encounters among 428,855 nonpregnant women aged ≥ 18 years at Kaiser Permanente Southern California from 2015 to 2022.
- Analysis included outpatient UTI encounters in ambulatory and urgent care settings, excluding emergency and inpatient visits.
- Data collection encompassed demographic information, urine tests, antibiotic dispensation, and UTI diagnoses using International Classification of Diseases, 9th and 10th Revision codes.
- Encounters conducted by physicians, physician assistants, nurse practitioners, and registered nurses through in-person, phone, video, and health portal platforms were evaluated.
TAKEAWAY:
- Virtual encounters grew by 603.2% compared with a 122.8% increase for in-person visits, with virtual visits accounting for 60% (733,263) of all UTI encounters.
- The rate of UTI encounters per 1000 adult female patients increased by 241.6%, while membership in the health system grew by only 24.4%.
- Antibiotics were prescribed without urine testing in 42.5% (519,135) of encounters, and among encounters with both antibiotic dispensation and urine testing, 57.1% (278,903) had a positive culture.
- According to the authors, the increasing rate of antibiotic dispensation surpassed the growth in positive urine culture rates, suggesting increased use of empiric antibiotics.
IN PRACTICE:
“Our findings underscore the importance of balancing telemedicine’s accessibility with maintaining antibiotic stewardship and highlight the need for updated guidelines,” wrote the authors of the study. An accompanying editorial said, “Unfortunately, our misguided conceptual model has led to several decades of UTI research focusing on bad bugs rather than investigating the natural host defenses, how we might boost these, what perturbs the ecosystem, and how microbial defense occurs within the bladder.”
SOURCE:
The study was led by Ghanshyam Yadav, MD, Kaiser Permanente Southern California in San Diego. It was published online in Obstetrics & Gynecology. The editorial, written by Nazema Y. Siddiqui, MD, MHSc, from the Department of Obstetrics and Gynecology, Duke University Medical Center, Durham, North Carolina, was also published in Obstetrics & Gynecology.
LIMITATIONS:
The retrospective design and analysis at the encounter level did not allow for control of patient and clinician clustering. The study was limited to a single health maintenance organization, which may affect the generalizability of the findings.
DISCLOSURES:
This research received support through a grant from the Regional Research Committee of Kaiser Permanente Southern California (RRC grant number: KP-RRC-20221002). Heidi Brown and Jasmine Tan-Kim disclosed receiving royalties from UpToDate. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
UTI in Primary Care: New Guidelines
This transcript has been edited for clarity.
We often see urinary tract infections in primary care, so these guidelines for the prevention, diagnosis and management of urinary tract infection (UTI) are very helpful to reaffirm our knowledge in the areas where know what we’re doing and update our knowledge in areas of uncertainty. These guidelines are from a new group called the WikiGuidelines group. Ordinarily, I wouldn’t have considered reviewing one of these guidelines, but this one was published in JAMA Network Open. It is evidence based and covers the topic really well.
Diagnosis. Order a urinalysis or a urine culture only if the patient is having symptoms of a UTI. This may seem obvious, but particularly among older individuals, in whom asymptomatic bacteriuria is very common and should not be treated, nonspecific symptoms such as just not feeling well for a day do not warrant obtaining a urinalysis and culture. With no clear way to distinguish between asymptomatic bacteriuria and a true UTI, the first step in making the diagnosis of a UTI accurately is ordering urine studies only in people who have a reasonable chance of having an infection.
The guideline suggests that the diagnosis of UTI should be primarily based on clinical symptoms. A urinalysis can provide further information, but the authors caution us against relying solely on the urinalysis. This is an incredibly important evidence-based recommendation. If you think about it, this supports the common practice of treating UTIs over the phone without having to see the patient or check a urinalysis.
The rationale for this recommendation is that urinalysis is neither a sensitive nor specific test for UTI. The sensitivity of leukocyte esterase is only about 80%, and the specificity is even lower. For positive nitrite on urinalysis, the sensitivity is below 50%, meaning the test would be negative more than half the time when someone actually has a UTI. The specificity of urine nitrate is very high (more than 90%), so if the patient is nitrite positive, they clearly have a UTI. This means that a patient’s report of classic UTI symptoms — urinary burning, frequency, and urgency — is about as good if not a better indicator of a UTI than a urinalysis.
The guidelines also say that in simple uncomplicated cystitis in healthy nonpregnant patients, routine urine cultures are not necessary. A fascinating meta-analysis in JAMA showed that, for women presenting to outpatient clinics with at least two symptoms of UTI and absence of vaginal discharge, there was a greater than 90% likelihood of having acute cystitis. A reminder here, however: If a woman is sexually active and at risk for sexually transmitted infections, then consider testing for STIs as well, because the symptoms of an STI can mimic those of a UTI.
Treatment. Treatment for UTI is usually empiric, with treatment initiated before the culture results are known and with cultures being done only for people with complicated infections, such as pyelonephritis, or with recurrent infections. Decisions about what to use for treatment can be influenced by local patterns of resistance and an individual’s risk factors for antimicrobial resistance. As a general rule, for uncomplicated cystitis, nitrofurantoin for 5 days is a reasonable first-line agent. Evidence of efficacy is good, and the risk for antimicrobial resistance is lower vs using antibiotics for other systemic infections.
Other reasonable first-line agents for uncomplicated cystitis include trimethoprim-sulfamethoxazole (TMP-SMX) for 3 days; fosfomycin (oral) single dose; or a beta-lactam (most commonly a first generation cephalosporin), although evidence for duration is unclear. Also mentioned are two unfamiliar antibiotics: pivmecillinam (a beta-lactam agent recently approved by the Food and Drug Administration [FDA], given for 3 days) and gepotidacin (from a new class of antibiotic that is currently under FDA review). Fluoroquinolones should not usually be first-line agents unless other treatment options are not appropriate.
It’s important to distinguish between uncomplicated cystitis and pyelonephritis. For pyelonephritis (infection of the upper urinary tract), the first decision has to do with setting for care, depending on how sick someone is, and the likelihood of gram-negative bacteremia — all of which help whether the patient needs to be hospitalized for intravenous antibiotics, or can be treated as an outpatient. Determine if they need to be admitted for intravenous antibiotics or whether they can be treated as an outpatient. For outpatient treatment of pyelonephritis, the guideline suggests that TMP-SMX or a first-generation cephalosporin are both reasonable first-line agents, with fluoroquinolones being a reasonable choice as well. Ceftriaxone is recommended for first-line therapy for patients who require intravenous treatment.
People often forget that we can do a lot to prevent UTIs, particularly among women with recurrent UTIs. The prevention of UTIs has both nonpharmacologic and pharmacologic approaches.
Nonpharmacologic prevention. One nonpharmacologic strategy is increasing water intake. A randomized controlled trial in women with recurrent cystitis who drank less than 1.5 L of fluid a day showed that the women randomized to consume an additional 1.5 L of water daily had significantly reduced cystitis frequency — approximately 50%. Because this was the only randomized trial to show this effect, this is not a strong recommendation, but there is very little downside in healthy women, so increasing water intake is a reasonable recommendation.
Another commonly discussed intervention is the use of cranberry products. As it turns out, most prospective studies have shown that cranberry products can reduce the risk for symptomatic UTIs in women with recurrent UTI.
Pharmacologic prevention. For postmenopausal women with recurrent UTI, topical vaginal estrogen has a strong base of evidence — more than 30 randomized trials — supporting its effectiveness in UTI: a 50%-90% reduction in the incidence of recurrent UTIs. Topical estrogen has minimal systemic absorption, and there are no concerning safety signals with respect to either thromboembolic disease or cancer (endometrial or breast).
Methenamine hippurate is also recommended and is FDA-approved for prevention of UTIs. It works by releasing formaldehyde in the urine, leading to bacteriostasis, which is how it leads to a decrease in UTIs. Finally, postcoital or daily administration of TMP-SMX, nitrofurantoin, norfloxacin, and ciprofloxacin all have comparable efficacy for prophylaxis, with a meta-analysis showing a decrease in recurrence rate of approximately 85%. The guideline states that there is insufficient evidence to support the use of either probiotics or D-mannose to prevent UTIs.
This is a wonderful update on a common problem. We all have a lot of clinical experience here.
Dr Skolnik, Department of Family Medicine, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia; Associate Director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
We often see urinary tract infections in primary care, so these guidelines for the prevention, diagnosis and management of urinary tract infection (UTI) are very helpful to reaffirm our knowledge in the areas where know what we’re doing and update our knowledge in areas of uncertainty. These guidelines are from a new group called the WikiGuidelines group. Ordinarily, I wouldn’t have considered reviewing one of these guidelines, but this one was published in JAMA Network Open. It is evidence based and covers the topic really well.
Diagnosis. Order a urinalysis or a urine culture only if the patient is having symptoms of a UTI. This may seem obvious, but particularly among older individuals, in whom asymptomatic bacteriuria is very common and should not be treated, nonspecific symptoms such as just not feeling well for a day do not warrant obtaining a urinalysis and culture. With no clear way to distinguish between asymptomatic bacteriuria and a true UTI, the first step in making the diagnosis of a UTI accurately is ordering urine studies only in people who have a reasonable chance of having an infection.
The guideline suggests that the diagnosis of UTI should be primarily based on clinical symptoms. A urinalysis can provide further information, but the authors caution us against relying solely on the urinalysis. This is an incredibly important evidence-based recommendation. If you think about it, this supports the common practice of treating UTIs over the phone without having to see the patient or check a urinalysis.
The rationale for this recommendation is that urinalysis is neither a sensitive nor specific test for UTI. The sensitivity of leukocyte esterase is only about 80%, and the specificity is even lower. For positive nitrite on urinalysis, the sensitivity is below 50%, meaning the test would be negative more than half the time when someone actually has a UTI. The specificity of urine nitrate is very high (more than 90%), so if the patient is nitrite positive, they clearly have a UTI. This means that a patient’s report of classic UTI symptoms — urinary burning, frequency, and urgency — is about as good if not a better indicator of a UTI than a urinalysis.
The guidelines also say that in simple uncomplicated cystitis in healthy nonpregnant patients, routine urine cultures are not necessary. A fascinating meta-analysis in JAMA showed that, for women presenting to outpatient clinics with at least two symptoms of UTI and absence of vaginal discharge, there was a greater than 90% likelihood of having acute cystitis. A reminder here, however: If a woman is sexually active and at risk for sexually transmitted infections, then consider testing for STIs as well, because the symptoms of an STI can mimic those of a UTI.
Treatment. Treatment for UTI is usually empiric, with treatment initiated before the culture results are known and with cultures being done only for people with complicated infections, such as pyelonephritis, or with recurrent infections. Decisions about what to use for treatment can be influenced by local patterns of resistance and an individual’s risk factors for antimicrobial resistance. As a general rule, for uncomplicated cystitis, nitrofurantoin for 5 days is a reasonable first-line agent. Evidence of efficacy is good, and the risk for antimicrobial resistance is lower vs using antibiotics for other systemic infections.
Other reasonable first-line agents for uncomplicated cystitis include trimethoprim-sulfamethoxazole (TMP-SMX) for 3 days; fosfomycin (oral) single dose; or a beta-lactam (most commonly a first generation cephalosporin), although evidence for duration is unclear. Also mentioned are two unfamiliar antibiotics: pivmecillinam (a beta-lactam agent recently approved by the Food and Drug Administration [FDA], given for 3 days) and gepotidacin (from a new class of antibiotic that is currently under FDA review). Fluoroquinolones should not usually be first-line agents unless other treatment options are not appropriate.
It’s important to distinguish between uncomplicated cystitis and pyelonephritis. For pyelonephritis (infection of the upper urinary tract), the first decision has to do with setting for care, depending on how sick someone is, and the likelihood of gram-negative bacteremia — all of which help whether the patient needs to be hospitalized for intravenous antibiotics, or can be treated as an outpatient. Determine if they need to be admitted for intravenous antibiotics or whether they can be treated as an outpatient. For outpatient treatment of pyelonephritis, the guideline suggests that TMP-SMX or a first-generation cephalosporin are both reasonable first-line agents, with fluoroquinolones being a reasonable choice as well. Ceftriaxone is recommended for first-line therapy for patients who require intravenous treatment.
People often forget that we can do a lot to prevent UTIs, particularly among women with recurrent UTIs. The prevention of UTIs has both nonpharmacologic and pharmacologic approaches.
Nonpharmacologic prevention. One nonpharmacologic strategy is increasing water intake. A randomized controlled trial in women with recurrent cystitis who drank less than 1.5 L of fluid a day showed that the women randomized to consume an additional 1.5 L of water daily had significantly reduced cystitis frequency — approximately 50%. Because this was the only randomized trial to show this effect, this is not a strong recommendation, but there is very little downside in healthy women, so increasing water intake is a reasonable recommendation.
Another commonly discussed intervention is the use of cranberry products. As it turns out, most prospective studies have shown that cranberry products can reduce the risk for symptomatic UTIs in women with recurrent UTI.
Pharmacologic prevention. For postmenopausal women with recurrent UTI, topical vaginal estrogen has a strong base of evidence — more than 30 randomized trials — supporting its effectiveness in UTI: a 50%-90% reduction in the incidence of recurrent UTIs. Topical estrogen has minimal systemic absorption, and there are no concerning safety signals with respect to either thromboembolic disease or cancer (endometrial or breast).
Methenamine hippurate is also recommended and is FDA-approved for prevention of UTIs. It works by releasing formaldehyde in the urine, leading to bacteriostasis, which is how it leads to a decrease in UTIs. Finally, postcoital or daily administration of TMP-SMX, nitrofurantoin, norfloxacin, and ciprofloxacin all have comparable efficacy for prophylaxis, with a meta-analysis showing a decrease in recurrence rate of approximately 85%. The guideline states that there is insufficient evidence to support the use of either probiotics or D-mannose to prevent UTIs.
This is a wonderful update on a common problem. We all have a lot of clinical experience here.
Dr Skolnik, Department of Family Medicine, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia; Associate Director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
We often see urinary tract infections in primary care, so these guidelines for the prevention, diagnosis and management of urinary tract infection (UTI) are very helpful to reaffirm our knowledge in the areas where know what we’re doing and update our knowledge in areas of uncertainty. These guidelines are from a new group called the WikiGuidelines group. Ordinarily, I wouldn’t have considered reviewing one of these guidelines, but this one was published in JAMA Network Open. It is evidence based and covers the topic really well.
Diagnosis. Order a urinalysis or a urine culture only if the patient is having symptoms of a UTI. This may seem obvious, but particularly among older individuals, in whom asymptomatic bacteriuria is very common and should not be treated, nonspecific symptoms such as just not feeling well for a day do not warrant obtaining a urinalysis and culture. With no clear way to distinguish between asymptomatic bacteriuria and a true UTI, the first step in making the diagnosis of a UTI accurately is ordering urine studies only in people who have a reasonable chance of having an infection.
The guideline suggests that the diagnosis of UTI should be primarily based on clinical symptoms. A urinalysis can provide further information, but the authors caution us against relying solely on the urinalysis. This is an incredibly important evidence-based recommendation. If you think about it, this supports the common practice of treating UTIs over the phone without having to see the patient or check a urinalysis.
The rationale for this recommendation is that urinalysis is neither a sensitive nor specific test for UTI. The sensitivity of leukocyte esterase is only about 80%, and the specificity is even lower. For positive nitrite on urinalysis, the sensitivity is below 50%, meaning the test would be negative more than half the time when someone actually has a UTI. The specificity of urine nitrate is very high (more than 90%), so if the patient is nitrite positive, they clearly have a UTI. This means that a patient’s report of classic UTI symptoms — urinary burning, frequency, and urgency — is about as good if not a better indicator of a UTI than a urinalysis.
The guidelines also say that in simple uncomplicated cystitis in healthy nonpregnant patients, routine urine cultures are not necessary. A fascinating meta-analysis in JAMA showed that, for women presenting to outpatient clinics with at least two symptoms of UTI and absence of vaginal discharge, there was a greater than 90% likelihood of having acute cystitis. A reminder here, however: If a woman is sexually active and at risk for sexually transmitted infections, then consider testing for STIs as well, because the symptoms of an STI can mimic those of a UTI.
Treatment. Treatment for UTI is usually empiric, with treatment initiated before the culture results are known and with cultures being done only for people with complicated infections, such as pyelonephritis, or with recurrent infections. Decisions about what to use for treatment can be influenced by local patterns of resistance and an individual’s risk factors for antimicrobial resistance. As a general rule, for uncomplicated cystitis, nitrofurantoin for 5 days is a reasonable first-line agent. Evidence of efficacy is good, and the risk for antimicrobial resistance is lower vs using antibiotics for other systemic infections.
Other reasonable first-line agents for uncomplicated cystitis include trimethoprim-sulfamethoxazole (TMP-SMX) for 3 days; fosfomycin (oral) single dose; or a beta-lactam (most commonly a first generation cephalosporin), although evidence for duration is unclear. Also mentioned are two unfamiliar antibiotics: pivmecillinam (a beta-lactam agent recently approved by the Food and Drug Administration [FDA], given for 3 days) and gepotidacin (from a new class of antibiotic that is currently under FDA review). Fluoroquinolones should not usually be first-line agents unless other treatment options are not appropriate.
It’s important to distinguish between uncomplicated cystitis and pyelonephritis. For pyelonephritis (infection of the upper urinary tract), the first decision has to do with setting for care, depending on how sick someone is, and the likelihood of gram-negative bacteremia — all of which help whether the patient needs to be hospitalized for intravenous antibiotics, or can be treated as an outpatient. Determine if they need to be admitted for intravenous antibiotics or whether they can be treated as an outpatient. For outpatient treatment of pyelonephritis, the guideline suggests that TMP-SMX or a first-generation cephalosporin are both reasonable first-line agents, with fluoroquinolones being a reasonable choice as well. Ceftriaxone is recommended for first-line therapy for patients who require intravenous treatment.
People often forget that we can do a lot to prevent UTIs, particularly among women with recurrent UTIs. The prevention of UTIs has both nonpharmacologic and pharmacologic approaches.
Nonpharmacologic prevention. One nonpharmacologic strategy is increasing water intake. A randomized controlled trial in women with recurrent cystitis who drank less than 1.5 L of fluid a day showed that the women randomized to consume an additional 1.5 L of water daily had significantly reduced cystitis frequency — approximately 50%. Because this was the only randomized trial to show this effect, this is not a strong recommendation, but there is very little downside in healthy women, so increasing water intake is a reasonable recommendation.
Another commonly discussed intervention is the use of cranberry products. As it turns out, most prospective studies have shown that cranberry products can reduce the risk for symptomatic UTIs in women with recurrent UTI.
Pharmacologic prevention. For postmenopausal women with recurrent UTI, topical vaginal estrogen has a strong base of evidence — more than 30 randomized trials — supporting its effectiveness in UTI: a 50%-90% reduction in the incidence of recurrent UTIs. Topical estrogen has minimal systemic absorption, and there are no concerning safety signals with respect to either thromboembolic disease or cancer (endometrial or breast).
Methenamine hippurate is also recommended and is FDA-approved for prevention of UTIs. It works by releasing formaldehyde in the urine, leading to bacteriostasis, which is how it leads to a decrease in UTIs. Finally, postcoital or daily administration of TMP-SMX, nitrofurantoin, norfloxacin, and ciprofloxacin all have comparable efficacy for prophylaxis, with a meta-analysis showing a decrease in recurrence rate of approximately 85%. The guideline states that there is insufficient evidence to support the use of either probiotics or D-mannose to prevent UTIs.
This is a wonderful update on a common problem. We all have a lot of clinical experience here.
Dr Skolnik, Department of Family Medicine, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia; Associate Director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article appeared on Medscape.com.
