User login
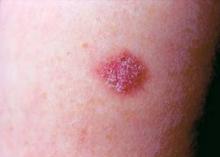
Aggressive Nonmelanoma Skin Cancers Often Misdiagnosed
LISBON – Two of 10 nonmelanoma skin cancers are misdiagnosed as being of a nonaggressive tumor subtype at initial biopsy, according to Dr. Nathalie Zeitouni.
This raises concern that a substantial number of biopsied squamous and basal cell carcinomas are being treated suboptimally, Dr. Zeitouni said at the congress.
She presented a consecutive series of 513 patients referred for Mohs micrographic surgery for biopsy-proven BCC or SCC. Based upon routine Mohs intraoperative evaluation of all histologic tumor layers, 21.1% of the cancers were of aggressive subtypes that went undiagnosed on initial biopsy.
Aggressive subtypes of nonmelanoma skin cancer include basosquamous carcinoma, invasive SCC, and morpheaform, infiltrating, keratinizing, and micronodular BCC. Nonaggressive subtypes include follicular, nodular, adenoid cystic, and superficial BCC, as well as SCC in situ, according to Dr. Zeitouni, chief of dermatologic surgery at the Roswell Park Cancer Institute, Buffalo, N.Y.
In only 51% of cases was there concordance between the preoperative and the definitive intraoperative diagnosis of a nonmelanoma skin cancer as being of an aggressive or nonaggressive subtype.
In 21% of cases the intraoperative evaluation showed no residual tumor present, only scar. In 5.5% of cases, intraoperative histologic tumor layer evaluation resulted in downgrading of the nonmelanoma skin cancer from an aggressive to a nonaggressive subtype.
Dr. Zeitouni stressed that dermatologists need to have a low threshold for suspecting that a nonmelanoma skin cancer is of an undiagnosed aggressive subtype. If the lesion is clinically atypical or it responds poorly to standard excision or simple destructive measures, that possibility becomes distinctly more likely. Aggressive subtypes, she added, are best managed by Mohs surgery.
She said she had no relevant financial disclosures.
LISBON – Two of 10 nonmelanoma skin cancers are misdiagnosed as being of a nonaggressive tumor subtype at initial biopsy, according to Dr. Nathalie Zeitouni.
This raises concern that a substantial number of biopsied squamous and basal cell carcinomas are being treated suboptimally, Dr. Zeitouni said at the congress.
She presented a consecutive series of 513 patients referred for Mohs micrographic surgery for biopsy-proven BCC or SCC. Based upon routine Mohs intraoperative evaluation of all histologic tumor layers, 21.1% of the cancers were of aggressive subtypes that went undiagnosed on initial biopsy.
Aggressive subtypes of nonmelanoma skin cancer include basosquamous carcinoma, invasive SCC, and morpheaform, infiltrating, keratinizing, and micronodular BCC. Nonaggressive subtypes include follicular, nodular, adenoid cystic, and superficial BCC, as well as SCC in situ, according to Dr. Zeitouni, chief of dermatologic surgery at the Roswell Park Cancer Institute, Buffalo, N.Y.
In only 51% of cases was there concordance between the preoperative and the definitive intraoperative diagnosis of a nonmelanoma skin cancer as being of an aggressive or nonaggressive subtype.
In 21% of cases the intraoperative evaluation showed no residual tumor present, only scar. In 5.5% of cases, intraoperative histologic tumor layer evaluation resulted in downgrading of the nonmelanoma skin cancer from an aggressive to a nonaggressive subtype.
Dr. Zeitouni stressed that dermatologists need to have a low threshold for suspecting that a nonmelanoma skin cancer is of an undiagnosed aggressive subtype. If the lesion is clinically atypical or it responds poorly to standard excision or simple destructive measures, that possibility becomes distinctly more likely. Aggressive subtypes, she added, are best managed by Mohs surgery.
She said she had no relevant financial disclosures.
LISBON – Two of 10 nonmelanoma skin cancers are misdiagnosed as being of a nonaggressive tumor subtype at initial biopsy, according to Dr. Nathalie Zeitouni.
This raises concern that a substantial number of biopsied squamous and basal cell carcinomas are being treated suboptimally, Dr. Zeitouni said at the congress.
She presented a consecutive series of 513 patients referred for Mohs micrographic surgery for biopsy-proven BCC or SCC. Based upon routine Mohs intraoperative evaluation of all histologic tumor layers, 21.1% of the cancers were of aggressive subtypes that went undiagnosed on initial biopsy.
Aggressive subtypes of nonmelanoma skin cancer include basosquamous carcinoma, invasive SCC, and morpheaform, infiltrating, keratinizing, and micronodular BCC. Nonaggressive subtypes include follicular, nodular, adenoid cystic, and superficial BCC, as well as SCC in situ, according to Dr. Zeitouni, chief of dermatologic surgery at the Roswell Park Cancer Institute, Buffalo, N.Y.
In only 51% of cases was there concordance between the preoperative and the definitive intraoperative diagnosis of a nonmelanoma skin cancer as being of an aggressive or nonaggressive subtype.
In 21% of cases the intraoperative evaluation showed no residual tumor present, only scar. In 5.5% of cases, intraoperative histologic tumor layer evaluation resulted in downgrading of the nonmelanoma skin cancer from an aggressive to a nonaggressive subtype.
Dr. Zeitouni stressed that dermatologists need to have a low threshold for suspecting that a nonmelanoma skin cancer is of an undiagnosed aggressive subtype. If the lesion is clinically atypical or it responds poorly to standard excision or simple destructive measures, that possibility becomes distinctly more likely. Aggressive subtypes, she added, are best managed by Mohs surgery.
She said she had no relevant financial disclosures.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: In only 51% of cases was there concordance between the preoperative and the definitive intraoperative diagnosis of a nonmelanoma skin cancer as being of an aggressive or nonaggressive subtype.
Data Source: A retrospective analysis of 513 consecutive patients with biopsy-proven basal or squamous cell carcinoma treated with Mohs micrographic surgery.
Disclosures: Dr. Zeitouni reported having no relevant financial disclosures.
Translating 'Crazy Fungus:' The Skinny Podcast
'In this month's podcast, Reporter Sherry Boschert talks to Dr. Libby Edwards about how best to manage genital itching in female patients, while Reporter Jeff Evans catches up with Dr. Darrell Rigel on what dermatologists can expect now that MelaFind has received FDA approval.
Vincent DeLeo reminds dermatologists that they need to tell patients how much sunscreen to use during application because most aren't using enough.
And last but not least, Dr. Alan Rockoff shows off his ability to say "fungus" in multiple languages.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
'In this month's podcast, Reporter Sherry Boschert talks to Dr. Libby Edwards about how best to manage genital itching in female patients, while Reporter Jeff Evans catches up with Dr. Darrell Rigel on what dermatologists can expect now that MelaFind has received FDA approval.
Vincent DeLeo reminds dermatologists that they need to tell patients how much sunscreen to use during application because most aren't using enough.
And last but not least, Dr. Alan Rockoff shows off his ability to say "fungus" in multiple languages.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
'In this month's podcast, Reporter Sherry Boschert talks to Dr. Libby Edwards about how best to manage genital itching in female patients, while Reporter Jeff Evans catches up with Dr. Darrell Rigel on what dermatologists can expect now that MelaFind has received FDA approval.
Vincent DeLeo reminds dermatologists that they need to tell patients how much sunscreen to use during application because most aren't using enough.
And last but not least, Dr. Alan Rockoff shows off his ability to say "fungus" in multiple languages.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
Fewer Visits Don't Jeopardize Melanoma Patient Care
A novel, less-intensive monitoring schedule for patients with localized primary cutaneous melanoma appears to reduce follow-up visits without affecting the rate of detection of new primary or recurrent melanomas, according to an analysis of data from an inception cohort of 3,081 patients.
Given that follow-up care for melanoma patients in the United States costs up to $500 million per year, the findings could have important implications for cost savings, according to Robin M. Turner, Ph.D., and colleagues.
Using data from the Melanoma Institute Australia on the inception cohort, which included consecutive patients with stage I or II melanoma first diagnosed between January 1985 and December 2009, the investigators modeled the delay in diagnosis of a new primary melanoma or recurrence with two different monitoring schedules.
Schedule one was based on 2008 Australian and New Zealand guidelines, which recommend follow-up every 6 months for 5 years, then annually for 5 years for patients with stage 1A and 1B disease, or every 3 months for 5 years, then annually for 5 years in patients with stages IIA, IIB, or IIC.
Schedule two, the novel monitoring arm, called for annual follow-up for 10 years for patients with stage I disease; every 6 months for 2 years, and then annually for 8 years, for patients with stage IIA disease; and every 4 months for 2 years, every 6 months in year 3, and then annually for 5 years, for patients with stages IIB and IIC disease.
Within 10 years, the total number of recurrences was 229 for every 1,000 patients, and the number of new primary melanomas was 61 for every 1,000 patients.
Compared with the 2008 Australia and New Zealand guidelines for monitoring, the novel schedule, which required almost 3,000 fewer visits per 1,000 patients (8,044 vs. 5,221 visits), resulted in a delay in the detection – by at least 2 months – of an additional 44.9 recurrences per 1,000 patients, and an additional 9.6 new primary melanomas, Robin M. Turner, Ph.D., of the School of Public Health at the University of Sydney, Australia, and colleagues reported online in the Journal of Clinical Oncology (2011 Nov. 7 [doi:10.1200/JCO.2010.34.2956]).
"If we assume self-detection rates based on published estimates [75% for recurrence and 50% for new primary], use of schedule two rather than schedule one results in only a small number of additional patients experiencing a delay in recurrence diagnosis (up to [a] maximum of 3 months; extra 11.3 patients per 1,000 with delay [greater than] 2 months) and even fewer experiencing a delay in new primary diagnosis (up to [a] maximum of 6 months; extra 4.9 per 1,000)," the investigators wrote.
Moreover, if there were no patients lost to follow-up and they attended all visits until diagnosis or 10 years, the total number of visits for schedule one would be 19,546, compared with 12,303 for schedule two.
"Not only would these fewer monitoring visits reduce the burden on patients in terms of time and expense in attendance and possible unnecessary anxiety, but they would also represent substantial savings for the health care system," the investigators wrote.
They also found that the risk of recurrence was greatest in the first year of follow-up; that the most important predictor of recurrence was American Joint Committee on Cancer substage (hazard ratios of 1, 1.12, 0.56, 0.70, and 0.95 for stages IA, IB, IIA, IIB, and IIC, respectively); and that the most important predictors of new primary melanomas were age and date of primary diagnosis after 1992 (hazard ratios of 1.29 and 2.21, respectively).
This study was supported by grants from the Australian National Health and Medical Research Council. The investigators said they had no relevant financial disclosures.
In an accompanying editorial, Dr. Vernon K. Sondak and Dr. Sancy A.
Leachman wrote that the findings of Dr. Turner and colleagues "have
begun to move us down the path toward data-driven, personalized
follow-up guidelines."
This path is an important one, because in
the current era of personalized medicine, too little attention has been
paid to personalizing follow-up for patients after curative treatment of
cancer, including localized cutaneous melanoma, they noted (J. Clin.
Oncol. 2011 Nov. 7 [doi:10.1200/JCO.2011.37.6681]).
Success,
however, will require new partnerships and collaborations. Specifically,
the ultimate solution should involve shared follow-up with physicians
in the community, as well as with the patient and the patient’s family,
through education designed to maximize their ability to identify
recurrent and new melanomas.
Shared follow-up with local
dermatologists of patients with melanoma would address the goal of
detecting recurrences and new primary melanomas while simultaneously
decreasing the frequency of visits to surgical or medical oncologists.
Focusing
the lens of personalized medicine onto follow-up strategies, and
pursuing new partnerships and collaborations, will provide "a golden
opportunity to deliver the right care to the right patients at the right
time and in the right place," they concluded.
Dr. Sondak is with the Moffitt Cancer Center and the University of South Florida, Tampa. Dr. Leachman is with the Huntsman Cancer Institute and the University of Utah, Salt Lake City. Dr. Sondak reported serving as a consultant or adviser to Merck/Schering Plough, and receiving honoraria from Merck. Dr. Leachman reported no relevant financial disclosures.
In an accompanying editorial, Dr. Vernon K. Sondak and Dr. Sancy A.
Leachman wrote that the findings of Dr. Turner and colleagues "have
begun to move us down the path toward data-driven, personalized
follow-up guidelines."
This path is an important one, because in
the current era of personalized medicine, too little attention has been
paid to personalizing follow-up for patients after curative treatment of
cancer, including localized cutaneous melanoma, they noted (J. Clin.
Oncol. 2011 Nov. 7 [doi:10.1200/JCO.2011.37.6681]).
Success,
however, will require new partnerships and collaborations. Specifically,
the ultimate solution should involve shared follow-up with physicians
in the community, as well as with the patient and the patient’s family,
through education designed to maximize their ability to identify
recurrent and new melanomas.
Shared follow-up with local
dermatologists of patients with melanoma would address the goal of
detecting recurrences and new primary melanomas while simultaneously
decreasing the frequency of visits to surgical or medical oncologists.
Focusing
the lens of personalized medicine onto follow-up strategies, and
pursuing new partnerships and collaborations, will provide "a golden
opportunity to deliver the right care to the right patients at the right
time and in the right place," they concluded.
Dr. Sondak is with the Moffitt Cancer Center and the University of South Florida, Tampa. Dr. Leachman is with the Huntsman Cancer Institute and the University of Utah, Salt Lake City. Dr. Sondak reported serving as a consultant or adviser to Merck/Schering Plough, and receiving honoraria from Merck. Dr. Leachman reported no relevant financial disclosures.
In an accompanying editorial, Dr. Vernon K. Sondak and Dr. Sancy A.
Leachman wrote that the findings of Dr. Turner and colleagues "have
begun to move us down the path toward data-driven, personalized
follow-up guidelines."
This path is an important one, because in
the current era of personalized medicine, too little attention has been
paid to personalizing follow-up for patients after curative treatment of
cancer, including localized cutaneous melanoma, they noted (J. Clin.
Oncol. 2011 Nov. 7 [doi:10.1200/JCO.2011.37.6681]).
Success,
however, will require new partnerships and collaborations. Specifically,
the ultimate solution should involve shared follow-up with physicians
in the community, as well as with the patient and the patient’s family,
through education designed to maximize their ability to identify
recurrent and new melanomas.
Shared follow-up with local
dermatologists of patients with melanoma would address the goal of
detecting recurrences and new primary melanomas while simultaneously
decreasing the frequency of visits to surgical or medical oncologists.
Focusing
the lens of personalized medicine onto follow-up strategies, and
pursuing new partnerships and collaborations, will provide "a golden
opportunity to deliver the right care to the right patients at the right
time and in the right place," they concluded.
Dr. Sondak is with the Moffitt Cancer Center and the University of South Florida, Tampa. Dr. Leachman is with the Huntsman Cancer Institute and the University of Utah, Salt Lake City. Dr. Sondak reported serving as a consultant or adviser to Merck/Schering Plough, and receiving honoraria from Merck. Dr. Leachman reported no relevant financial disclosures.
A novel, less-intensive monitoring schedule for patients with localized primary cutaneous melanoma appears to reduce follow-up visits without affecting the rate of detection of new primary or recurrent melanomas, according to an analysis of data from an inception cohort of 3,081 patients.
Given that follow-up care for melanoma patients in the United States costs up to $500 million per year, the findings could have important implications for cost savings, according to Robin M. Turner, Ph.D., and colleagues.
Using data from the Melanoma Institute Australia on the inception cohort, which included consecutive patients with stage I or II melanoma first diagnosed between January 1985 and December 2009, the investigators modeled the delay in diagnosis of a new primary melanoma or recurrence with two different monitoring schedules.
Schedule one was based on 2008 Australian and New Zealand guidelines, which recommend follow-up every 6 months for 5 years, then annually for 5 years for patients with stage 1A and 1B disease, or every 3 months for 5 years, then annually for 5 years in patients with stages IIA, IIB, or IIC.
Schedule two, the novel monitoring arm, called for annual follow-up for 10 years for patients with stage I disease; every 6 months for 2 years, and then annually for 8 years, for patients with stage IIA disease; and every 4 months for 2 years, every 6 months in year 3, and then annually for 5 years, for patients with stages IIB and IIC disease.
Within 10 years, the total number of recurrences was 229 for every 1,000 patients, and the number of new primary melanomas was 61 for every 1,000 patients.
Compared with the 2008 Australia and New Zealand guidelines for monitoring, the novel schedule, which required almost 3,000 fewer visits per 1,000 patients (8,044 vs. 5,221 visits), resulted in a delay in the detection – by at least 2 months – of an additional 44.9 recurrences per 1,000 patients, and an additional 9.6 new primary melanomas, Robin M. Turner, Ph.D., of the School of Public Health at the University of Sydney, Australia, and colleagues reported online in the Journal of Clinical Oncology (2011 Nov. 7 [doi:10.1200/JCO.2010.34.2956]).
"If we assume self-detection rates based on published estimates [75% for recurrence and 50% for new primary], use of schedule two rather than schedule one results in only a small number of additional patients experiencing a delay in recurrence diagnosis (up to [a] maximum of 3 months; extra 11.3 patients per 1,000 with delay [greater than] 2 months) and even fewer experiencing a delay in new primary diagnosis (up to [a] maximum of 6 months; extra 4.9 per 1,000)," the investigators wrote.
Moreover, if there were no patients lost to follow-up and they attended all visits until diagnosis or 10 years, the total number of visits for schedule one would be 19,546, compared with 12,303 for schedule two.
"Not only would these fewer monitoring visits reduce the burden on patients in terms of time and expense in attendance and possible unnecessary anxiety, but they would also represent substantial savings for the health care system," the investigators wrote.
They also found that the risk of recurrence was greatest in the first year of follow-up; that the most important predictor of recurrence was American Joint Committee on Cancer substage (hazard ratios of 1, 1.12, 0.56, 0.70, and 0.95 for stages IA, IB, IIA, IIB, and IIC, respectively); and that the most important predictors of new primary melanomas were age and date of primary diagnosis after 1992 (hazard ratios of 1.29 and 2.21, respectively).
This study was supported by grants from the Australian National Health and Medical Research Council. The investigators said they had no relevant financial disclosures.
A novel, less-intensive monitoring schedule for patients with localized primary cutaneous melanoma appears to reduce follow-up visits without affecting the rate of detection of new primary or recurrent melanomas, according to an analysis of data from an inception cohort of 3,081 patients.
Given that follow-up care for melanoma patients in the United States costs up to $500 million per year, the findings could have important implications for cost savings, according to Robin M. Turner, Ph.D., and colleagues.
Using data from the Melanoma Institute Australia on the inception cohort, which included consecutive patients with stage I or II melanoma first diagnosed between January 1985 and December 2009, the investigators modeled the delay in diagnosis of a new primary melanoma or recurrence with two different monitoring schedules.
Schedule one was based on 2008 Australian and New Zealand guidelines, which recommend follow-up every 6 months for 5 years, then annually for 5 years for patients with stage 1A and 1B disease, or every 3 months for 5 years, then annually for 5 years in patients with stages IIA, IIB, or IIC.
Schedule two, the novel monitoring arm, called for annual follow-up for 10 years for patients with stage I disease; every 6 months for 2 years, and then annually for 8 years, for patients with stage IIA disease; and every 4 months for 2 years, every 6 months in year 3, and then annually for 5 years, for patients with stages IIB and IIC disease.
Within 10 years, the total number of recurrences was 229 for every 1,000 patients, and the number of new primary melanomas was 61 for every 1,000 patients.
Compared with the 2008 Australia and New Zealand guidelines for monitoring, the novel schedule, which required almost 3,000 fewer visits per 1,000 patients (8,044 vs. 5,221 visits), resulted in a delay in the detection – by at least 2 months – of an additional 44.9 recurrences per 1,000 patients, and an additional 9.6 new primary melanomas, Robin M. Turner, Ph.D., of the School of Public Health at the University of Sydney, Australia, and colleagues reported online in the Journal of Clinical Oncology (2011 Nov. 7 [doi:10.1200/JCO.2010.34.2956]).
"If we assume self-detection rates based on published estimates [75% for recurrence and 50% for new primary], use of schedule two rather than schedule one results in only a small number of additional patients experiencing a delay in recurrence diagnosis (up to [a] maximum of 3 months; extra 11.3 patients per 1,000 with delay [greater than] 2 months) and even fewer experiencing a delay in new primary diagnosis (up to [a] maximum of 6 months; extra 4.9 per 1,000)," the investigators wrote.
Moreover, if there were no patients lost to follow-up and they attended all visits until diagnosis or 10 years, the total number of visits for schedule one would be 19,546, compared with 12,303 for schedule two.
"Not only would these fewer monitoring visits reduce the burden on patients in terms of time and expense in attendance and possible unnecessary anxiety, but they would also represent substantial savings for the health care system," the investigators wrote.
They also found that the risk of recurrence was greatest in the first year of follow-up; that the most important predictor of recurrence was American Joint Committee on Cancer substage (hazard ratios of 1, 1.12, 0.56, 0.70, and 0.95 for stages IA, IB, IIA, IIB, and IIC, respectively); and that the most important predictors of new primary melanomas were age and date of primary diagnosis after 1992 (hazard ratios of 1.29 and 2.21, respectively).
This study was supported by grants from the Australian National Health and Medical Research Council. The investigators said they had no relevant financial disclosures.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Major Finding: Compared with the 2008 Australia and New Zealand guidelines for monitoring, the novel schedule, which required almost 3,000 fewer visits per 1,000 patients (8,044 vs. 5,221), resulted in a delay in the detection – by at least 2 months – of an additional 44.9 recurrences per 1,000 patients, and an additional 9.6 new primary melanomas.
Data Source: An analysis of data from an inception cohort of more than 3,081 patients with stage I or II melanoma.
Disclosures: This study was supported by grants from the Australian National Health and Medical Research Council. The authors said they had no relevant financial disclosures.
Atopy May Protect Against Skin Cancer
LISBON – In the latest installment in an ongoing debate, dermatologist Thomas Kornek reported that atopic dermatitis was found to provide protection against skin cancer in patients in a large German study.
Standardized, onsite, dermatologist-conducted skin screenings of 90,880 employees in more than 300 German businesses demonstrated that the prevalence of nonmelanoma skin cancer among the 1.3% of workers with atopic dermatitis was less than half of that in coworkers without atopic dermatitis, Dr. Kornek reported at the congress.
Moreover, even though the subgroup with atopic dermatitis had significantly higher rates of well-established risk factors for melanoma, their actual prevalence of the malignancy was identical to that in workers without atopic dermatitis, implying a protective effect against melanoma as well, added Dr. Kornek of the University Medical Center Hamburg, Germany.
Participants in the workplace skin screening program averaged 43 years of age, and 53% were men. The prevalence of clinically diagnosed nonmelanoma skin cancer among patients with atopic dermatitis was 0.4%, compared with 0.9% in participants without atopy (P less than .05).
Premalignant skin lesions were identified in 1.7% of workers with atopic dermatitis and 2.1% of controls, a nonsignificant difference.
Melanomas were detected in 0.2% of participants with or without atopic dermatitis. Yet the prevalences of Fitzpatrick skin type I, more than 40 nevi, and a history of severe sunburns in childhood – all of which are risk factors for melanoma – were significantly greater in the atopic dermatitis subgroup, which in theory should have translated into more melanomas, said Dr. Kornek.
Two warring schools of thought have clashed with regard to the relationship between atopic dermatitis and cancer. One holds that the hyperreactive immune system that defines atopy in the form of asthma, hay fever, or atopic dermatitis ought to protect against carcinoma. The other school holds that the chronic immune stimulation present in individuals with atopic dermatitis ought to result in elevated risk. An additional consideration in patients with atopic dermatitis is that local and systemic immunotherapy could potentially boost the risk of developing skin cancer, noted Dr. Kornek.
Each side can point to supporting epidemiologic studies. For example, researchers in Sweden found that the risk of developing melanoma in 6,280 atopic dermatitis patients followed for more than 230,000 person-years was half that in the general Swedish population (J. Eur. Acad. Dermatol. Venereol. 2008;22:1423-8).
Dermatologists in Stockholm used the Swedish Cancer Registry to determine that a large cohort of atopic patients had no increased risk of nonmelanoma skin cancer, lymphoma, or cancer of the lung, pancreas, or cervix (Allergy 2005;60:1116-20).
Another group of investigators in Sweden reported modest but significantly increased risks of cancer of the pancreas, esophagus, lung, and brain, as well as lymphoma, in 15,666 patients earlier hospitalized for atopic dermatitis. There was a nonsignificant trend for more nonmelanoma skin cancers in the group, but no increase in melanoma (Arch. Dermatol. 2005;141:1123-7).
A literature review by dermatologists in Germany found mixed results regarding a possible association between atopy and cancer, although the investigators summed up the findings by noting that "the emerging picture from most of the currently available epidemiological data indicates that atopic disease is associated with a reduced risk of cancer" (Allergy 2005;60:1098-11).
Dr. Kornek speculated that one possible explanation for the reduced risk of skin cancer noted in the German workplace study is because individuals with atopic dermatitis are more aware of their skin and more alert to the rise of abnormal lesions than nonatopic persons.
He declared having no financial conflicts of interest.
LISBON – In the latest installment in an ongoing debate, dermatologist Thomas Kornek reported that atopic dermatitis was found to provide protection against skin cancer in patients in a large German study.
Standardized, onsite, dermatologist-conducted skin screenings of 90,880 employees in more than 300 German businesses demonstrated that the prevalence of nonmelanoma skin cancer among the 1.3% of workers with atopic dermatitis was less than half of that in coworkers without atopic dermatitis, Dr. Kornek reported at the congress.
Moreover, even though the subgroup with atopic dermatitis had significantly higher rates of well-established risk factors for melanoma, their actual prevalence of the malignancy was identical to that in workers without atopic dermatitis, implying a protective effect against melanoma as well, added Dr. Kornek of the University Medical Center Hamburg, Germany.
Participants in the workplace skin screening program averaged 43 years of age, and 53% were men. The prevalence of clinically diagnosed nonmelanoma skin cancer among patients with atopic dermatitis was 0.4%, compared with 0.9% in participants without atopy (P less than .05).
Premalignant skin lesions were identified in 1.7% of workers with atopic dermatitis and 2.1% of controls, a nonsignificant difference.
Melanomas were detected in 0.2% of participants with or without atopic dermatitis. Yet the prevalences of Fitzpatrick skin type I, more than 40 nevi, and a history of severe sunburns in childhood – all of which are risk factors for melanoma – were significantly greater in the atopic dermatitis subgroup, which in theory should have translated into more melanomas, said Dr. Kornek.
Two warring schools of thought have clashed with regard to the relationship between atopic dermatitis and cancer. One holds that the hyperreactive immune system that defines atopy in the form of asthma, hay fever, or atopic dermatitis ought to protect against carcinoma. The other school holds that the chronic immune stimulation present in individuals with atopic dermatitis ought to result in elevated risk. An additional consideration in patients with atopic dermatitis is that local and systemic immunotherapy could potentially boost the risk of developing skin cancer, noted Dr. Kornek.
Each side can point to supporting epidemiologic studies. For example, researchers in Sweden found that the risk of developing melanoma in 6,280 atopic dermatitis patients followed for more than 230,000 person-years was half that in the general Swedish population (J. Eur. Acad. Dermatol. Venereol. 2008;22:1423-8).
Dermatologists in Stockholm used the Swedish Cancer Registry to determine that a large cohort of atopic patients had no increased risk of nonmelanoma skin cancer, lymphoma, or cancer of the lung, pancreas, or cervix (Allergy 2005;60:1116-20).
Another group of investigators in Sweden reported modest but significantly increased risks of cancer of the pancreas, esophagus, lung, and brain, as well as lymphoma, in 15,666 patients earlier hospitalized for atopic dermatitis. There was a nonsignificant trend for more nonmelanoma skin cancers in the group, but no increase in melanoma (Arch. Dermatol. 2005;141:1123-7).
A literature review by dermatologists in Germany found mixed results regarding a possible association between atopy and cancer, although the investigators summed up the findings by noting that "the emerging picture from most of the currently available epidemiological data indicates that atopic disease is associated with a reduced risk of cancer" (Allergy 2005;60:1098-11).
Dr. Kornek speculated that one possible explanation for the reduced risk of skin cancer noted in the German workplace study is because individuals with atopic dermatitis are more aware of their skin and more alert to the rise of abnormal lesions than nonatopic persons.
He declared having no financial conflicts of interest.
LISBON – In the latest installment in an ongoing debate, dermatologist Thomas Kornek reported that atopic dermatitis was found to provide protection against skin cancer in patients in a large German study.
Standardized, onsite, dermatologist-conducted skin screenings of 90,880 employees in more than 300 German businesses demonstrated that the prevalence of nonmelanoma skin cancer among the 1.3% of workers with atopic dermatitis was less than half of that in coworkers without atopic dermatitis, Dr. Kornek reported at the congress.
Moreover, even though the subgroup with atopic dermatitis had significantly higher rates of well-established risk factors for melanoma, their actual prevalence of the malignancy was identical to that in workers without atopic dermatitis, implying a protective effect against melanoma as well, added Dr. Kornek of the University Medical Center Hamburg, Germany.
Participants in the workplace skin screening program averaged 43 years of age, and 53% were men. The prevalence of clinically diagnosed nonmelanoma skin cancer among patients with atopic dermatitis was 0.4%, compared with 0.9% in participants without atopy (P less than .05).
Premalignant skin lesions were identified in 1.7% of workers with atopic dermatitis and 2.1% of controls, a nonsignificant difference.
Melanomas were detected in 0.2% of participants with or without atopic dermatitis. Yet the prevalences of Fitzpatrick skin type I, more than 40 nevi, and a history of severe sunburns in childhood – all of which are risk factors for melanoma – were significantly greater in the atopic dermatitis subgroup, which in theory should have translated into more melanomas, said Dr. Kornek.
Two warring schools of thought have clashed with regard to the relationship between atopic dermatitis and cancer. One holds that the hyperreactive immune system that defines atopy in the form of asthma, hay fever, or atopic dermatitis ought to protect against carcinoma. The other school holds that the chronic immune stimulation present in individuals with atopic dermatitis ought to result in elevated risk. An additional consideration in patients with atopic dermatitis is that local and systemic immunotherapy could potentially boost the risk of developing skin cancer, noted Dr. Kornek.
Each side can point to supporting epidemiologic studies. For example, researchers in Sweden found that the risk of developing melanoma in 6,280 atopic dermatitis patients followed for more than 230,000 person-years was half that in the general Swedish population (J. Eur. Acad. Dermatol. Venereol. 2008;22:1423-8).
Dermatologists in Stockholm used the Swedish Cancer Registry to determine that a large cohort of atopic patients had no increased risk of nonmelanoma skin cancer, lymphoma, or cancer of the lung, pancreas, or cervix (Allergy 2005;60:1116-20).
Another group of investigators in Sweden reported modest but significantly increased risks of cancer of the pancreas, esophagus, lung, and brain, as well as lymphoma, in 15,666 patients earlier hospitalized for atopic dermatitis. There was a nonsignificant trend for more nonmelanoma skin cancers in the group, but no increase in melanoma (Arch. Dermatol. 2005;141:1123-7).
A literature review by dermatologists in Germany found mixed results regarding a possible association between atopy and cancer, although the investigators summed up the findings by noting that "the emerging picture from most of the currently available epidemiological data indicates that atopic disease is associated with a reduced risk of cancer" (Allergy 2005;60:1098-11).
Dr. Kornek speculated that one possible explanation for the reduced risk of skin cancer noted in the German workplace study is because individuals with atopic dermatitis are more aware of their skin and more alert to the rise of abnormal lesions than nonatopic persons.
He declared having no financial conflicts of interest.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: German workers with atopic dermatitis had a 56% lower prevalence of nonmelanoma skin cancer than their coworkers without atopy.
Data Source: A dermatologist-conducted skin screening program involving nearly 91,000 employees at more than 300 German companies.
Disclosures: Dr. Kornek had no conflicts of interest.
MelaFind Receives FDA Approval
MelaFind, a noninvasive melanoma detection device, was approved by the Food and Drug Administration on Nov. 2.
The road to approval was marked by some controversy. It included a closely split advisory panel recommendation in November 2010, followed by the May 2011 submission of a citizen petition to the FDA seeking action by the manufacturer, Mela Sciences. The FDA deemed the device approvable in late September, pending agreement between the agency and Mela Sciences on how the device would be used and by whom.
The approval includes a long list of indications and caveats, according to the company. They include the following:
• "MelaFind is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma.
• It is designed to be used when a dermatologist chooses to obtain additional information for a decision to biopsy. MelaFind should not be used to confirm a clinical diagnosis of melanoma.
• MelaFind is only for use by physicians trained in the clinical diagnosis and management of skin cancer [that is, dermatologists] who have also successfully completed a training program in the appropriate use of MelaFind."
• MelaFind positive lesions should be considered for biopsy. "Non-evaluable" lesions should be carefully reevaluated for biopsy.
• MelaFind is indicated for use only on the following lesions: those with a diameter between 2 mm and 22 mm; lesions accessible by the MelaFind imager; those sufficiently pigmented; lesions that do not contain a scar or fibrosis consistent with previous trauma; those in which the skin is intact; lesions greater than 1 cm away from the eye; and those that do not contain foreign matter. It should not be used on acral, palmar, plantar, mucosal, or subungual areas.
Dr. Darrell S. Rigel, a dermatologist at New York University and a consultant for Mela Sciences, said in a statement that the device "represents one of the most significant advances in early melanoma detection since the advent of the ABCD criteria that our group developed over a quarter century ago."
Dr. Joseph V. Gulfo, president and CEO of Mela Sciences, said that the company aims to launch MelaFind in the United States and Germany in the spring of 2012. "We’re planning a steadfast, deliberate and measured approach to the commercial launch to ensure that dermatologists in practices of all shapes and sizes are trained and set up to use MelaFind effectively on the patients who can benefit most from the objective information that the system provides during skin examinations," he said in the statement.
Initially, the device will be sold only to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with the dermatologists to fine-tune the device before rolling it out to a larger number of practices.
The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; its software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind, a noninvasive melanoma detection device, was approved by the Food and Drug Administration on Nov. 2.
The road to approval was marked by some controversy. It included a closely split advisory panel recommendation in November 2010, followed by the May 2011 submission of a citizen petition to the FDA seeking action by the manufacturer, Mela Sciences. The FDA deemed the device approvable in late September, pending agreement between the agency and Mela Sciences on how the device would be used and by whom.
The approval includes a long list of indications and caveats, according to the company. They include the following:
• "MelaFind is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma.
• It is designed to be used when a dermatologist chooses to obtain additional information for a decision to biopsy. MelaFind should not be used to confirm a clinical diagnosis of melanoma.
• MelaFind is only for use by physicians trained in the clinical diagnosis and management of skin cancer [that is, dermatologists] who have also successfully completed a training program in the appropriate use of MelaFind."
• MelaFind positive lesions should be considered for biopsy. "Non-evaluable" lesions should be carefully reevaluated for biopsy.
• MelaFind is indicated for use only on the following lesions: those with a diameter between 2 mm and 22 mm; lesions accessible by the MelaFind imager; those sufficiently pigmented; lesions that do not contain a scar or fibrosis consistent with previous trauma; those in which the skin is intact; lesions greater than 1 cm away from the eye; and those that do not contain foreign matter. It should not be used on acral, palmar, plantar, mucosal, or subungual areas.
Dr. Darrell S. Rigel, a dermatologist at New York University and a consultant for Mela Sciences, said in a statement that the device "represents one of the most significant advances in early melanoma detection since the advent of the ABCD criteria that our group developed over a quarter century ago."
Dr. Joseph V. Gulfo, president and CEO of Mela Sciences, said that the company aims to launch MelaFind in the United States and Germany in the spring of 2012. "We’re planning a steadfast, deliberate and measured approach to the commercial launch to ensure that dermatologists in practices of all shapes and sizes are trained and set up to use MelaFind effectively on the patients who can benefit most from the objective information that the system provides during skin examinations," he said in the statement.
Initially, the device will be sold only to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with the dermatologists to fine-tune the device before rolling it out to a larger number of practices.
The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; its software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
MelaFind, a noninvasive melanoma detection device, was approved by the Food and Drug Administration on Nov. 2.
The road to approval was marked by some controversy. It included a closely split advisory panel recommendation in November 2010, followed by the May 2011 submission of a citizen petition to the FDA seeking action by the manufacturer, Mela Sciences. The FDA deemed the device approvable in late September, pending agreement between the agency and Mela Sciences on how the device would be used and by whom.
The approval includes a long list of indications and caveats, according to the company. They include the following:
• "MelaFind is intended for use on clinically atypical cutaneous pigmented lesions with one or more clinical or historical characteristics of melanoma, excluding those with a clinical diagnosis of melanoma or likely melanoma.
• It is designed to be used when a dermatologist chooses to obtain additional information for a decision to biopsy. MelaFind should not be used to confirm a clinical diagnosis of melanoma.
• MelaFind is only for use by physicians trained in the clinical diagnosis and management of skin cancer [that is, dermatologists] who have also successfully completed a training program in the appropriate use of MelaFind."
• MelaFind positive lesions should be considered for biopsy. "Non-evaluable" lesions should be carefully reevaluated for biopsy.
• MelaFind is indicated for use only on the following lesions: those with a diameter between 2 mm and 22 mm; lesions accessible by the MelaFind imager; those sufficiently pigmented; lesions that do not contain a scar or fibrosis consistent with previous trauma; those in which the skin is intact; lesions greater than 1 cm away from the eye; and those that do not contain foreign matter. It should not be used on acral, palmar, plantar, mucosal, or subungual areas.
Dr. Darrell S. Rigel, a dermatologist at New York University and a consultant for Mela Sciences, said in a statement that the device "represents one of the most significant advances in early melanoma detection since the advent of the ABCD criteria that our group developed over a quarter century ago."
Dr. Joseph V. Gulfo, president and CEO of Mela Sciences, said that the company aims to launch MelaFind in the United States and Germany in the spring of 2012. "We’re planning a steadfast, deliberate and measured approach to the commercial launch to ensure that dermatologists in practices of all shapes and sizes are trained and set up to use MelaFind effectively on the patients who can benefit most from the objective information that the system provides during skin examinations," he said in the statement.
Initially, the device will be sold only to a handful of dermatologists in Connecticut, New Jersey, and New York. Mela Sciences will be working with the dermatologists to fine-tune the device before rolling it out to a larger number of practices.
The device received European Union approval in September. The company aims to have about 75 systems in Germany by next September, Dr. Gulfo said.
MelaFind is a multispectral computer vision system with a handheld imager that captures the image of a lesion; its software uses algorithms to analyze the image, indicating within 2 minutes whether a biopsy should be done.
The pivotal study of the device included 1,383 patients. According to the company, MelaFind had a sensitivity of 98%. The device’s sensitivity rating for malignant melanoma was significantly better than that of dermatologists, who showed a wide range of variability about which lesions would have been recommended for biopsy and which relegated to observation.
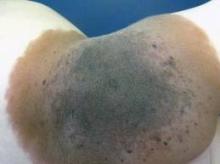
First Basal Cell Carcinoma Brings Risk for More
LISBON – Nearly 30% of patients diagnosed with a first pathologically confirmed basal cell carcinoma will develop one or more additional primary basal cell carcinomas within 5 years.
This finding, from a comprehensive Dutch national cancer registry, has important implications for clinical practice, noted Dr. Sophie C. Flohil at the annual congress of the European Academy of Dermatology and Venereology.
"Based upon our results, we would like to recommend that all BCC [basal cell carcinoma] patients undergo annual follow-up visits with full-body skin examinations for at least 3 years after their first BCC diagnosis."
She presented a cohort study involving 2,483 consecutive unselected Dutch patients diagnosed with a first histological confirmed BCC during 2004.
During 5 years of prospective follow-up in PALGA, the Dutch nationwide network and registry of histo- and cytopathology, 29.2% of the patients were diagnosed with one or more additional primary BCCs, according to Dr. Flohil, a dermatology resident at Erasmus University, Rotterdam, the Netherlands.
More than one-third of patients who developed a second primary BCC within 5 years did so during the first 6 months after diagnosis of the first.
The cumulative risk during the first 6 months of follow-up was extraordinarily high at 11.2%, as compared with 13.9% through 1 year, 18.5% after 2 years, 22.1% at 3 years, 25.5% after 4 years, and 29.2% after 5 years, she said. This translates into an incidence rate of 25,318 cases per 100,000 person-years during the first half-year after diagnosis of a first primary tumor.
"We think that this could indicate that these second BCCs that occur in the first 6 months after a first BCC diagnosis were already present at the date of the first BCC diagnosis, but were somehow missed then by the dermatologist or the doctor they went to," she said.
Still, the incidence rate remained quite high during the first several years of follow-up: 15,737 cases per 100,000 person-years through the first year and 10,779 per 100,000 person-years after 2 years before tailing off to 8,752 per 100,000 person-years at 3 years.
In a multivariate analysis, men had an adjusted 30% increased risk of developing one or more subsequent BCCs, and patients aged 65-79 years had an 81% greater risk than those under age 50.
Dr. Flohil declared having no financial conflicts of interest.
LISBON – Nearly 30% of patients diagnosed with a first pathologically confirmed basal cell carcinoma will develop one or more additional primary basal cell carcinomas within 5 years.
This finding, from a comprehensive Dutch national cancer registry, has important implications for clinical practice, noted Dr. Sophie C. Flohil at the annual congress of the European Academy of Dermatology and Venereology.
"Based upon our results, we would like to recommend that all BCC [basal cell carcinoma] patients undergo annual follow-up visits with full-body skin examinations for at least 3 years after their first BCC diagnosis."
She presented a cohort study involving 2,483 consecutive unselected Dutch patients diagnosed with a first histological confirmed BCC during 2004.
During 5 years of prospective follow-up in PALGA, the Dutch nationwide network and registry of histo- and cytopathology, 29.2% of the patients were diagnosed with one or more additional primary BCCs, according to Dr. Flohil, a dermatology resident at Erasmus University, Rotterdam, the Netherlands.
More than one-third of patients who developed a second primary BCC within 5 years did so during the first 6 months after diagnosis of the first.
The cumulative risk during the first 6 months of follow-up was extraordinarily high at 11.2%, as compared with 13.9% through 1 year, 18.5% after 2 years, 22.1% at 3 years, 25.5% after 4 years, and 29.2% after 5 years, she said. This translates into an incidence rate of 25,318 cases per 100,000 person-years during the first half-year after diagnosis of a first primary tumor.
"We think that this could indicate that these second BCCs that occur in the first 6 months after a first BCC diagnosis were already present at the date of the first BCC diagnosis, but were somehow missed then by the dermatologist or the doctor they went to," she said.
Still, the incidence rate remained quite high during the first several years of follow-up: 15,737 cases per 100,000 person-years through the first year and 10,779 per 100,000 person-years after 2 years before tailing off to 8,752 per 100,000 person-years at 3 years.
In a multivariate analysis, men had an adjusted 30% increased risk of developing one or more subsequent BCCs, and patients aged 65-79 years had an 81% greater risk than those under age 50.
Dr. Flohil declared having no financial conflicts of interest.
LISBON – Nearly 30% of patients diagnosed with a first pathologically confirmed basal cell carcinoma will develop one or more additional primary basal cell carcinomas within 5 years.
This finding, from a comprehensive Dutch national cancer registry, has important implications for clinical practice, noted Dr. Sophie C. Flohil at the annual congress of the European Academy of Dermatology and Venereology.
"Based upon our results, we would like to recommend that all BCC [basal cell carcinoma] patients undergo annual follow-up visits with full-body skin examinations for at least 3 years after their first BCC diagnosis."
She presented a cohort study involving 2,483 consecutive unselected Dutch patients diagnosed with a first histological confirmed BCC during 2004.
During 5 years of prospective follow-up in PALGA, the Dutch nationwide network and registry of histo- and cytopathology, 29.2% of the patients were diagnosed with one or more additional primary BCCs, according to Dr. Flohil, a dermatology resident at Erasmus University, Rotterdam, the Netherlands.
More than one-third of patients who developed a second primary BCC within 5 years did so during the first 6 months after diagnosis of the first.
The cumulative risk during the first 6 months of follow-up was extraordinarily high at 11.2%, as compared with 13.9% through 1 year, 18.5% after 2 years, 22.1% at 3 years, 25.5% after 4 years, and 29.2% after 5 years, she said. This translates into an incidence rate of 25,318 cases per 100,000 person-years during the first half-year after diagnosis of a first primary tumor.
"We think that this could indicate that these second BCCs that occur in the first 6 months after a first BCC diagnosis were already present at the date of the first BCC diagnosis, but were somehow missed then by the dermatologist or the doctor they went to," she said.
Still, the incidence rate remained quite high during the first several years of follow-up: 15,737 cases per 100,000 person-years through the first year and 10,779 per 100,000 person-years after 2 years before tailing off to 8,752 per 100,000 person-years at 3 years.
In a multivariate analysis, men had an adjusted 30% increased risk of developing one or more subsequent BCCs, and patients aged 65-79 years had an 81% greater risk than those under age 50.
Dr. Flohil declared having no financial conflicts of interest.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: More than 29% of 2,483 consecutive patients diagnosed with a first histological confirmed basal cell carcinoma developed a second one within 5 years.
Data Source: A Dutch national cancer registry.
Disclosures: No financial conflicts.
Blog: Morning May Be "Safer" Time to Tan
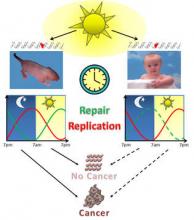
Dr. Aziz Sancar and colleagues at the University of North Carolina School of Medicine in Chapel Hill and the North Carolina State University in Raleigh conducted a study in hairless mice to see if the cyclical nature of a particular type of DNA repair might also influence the onset of skin cancer.
Dr. Sancar had already proven that a protein called XPA, which helps repair UV-related DNA damage, is primarily controlled by the circadian clock.
XPA activity reaches its peak between the hours of 4 p.m. and 6 p.m. and is at its lowest between 4 a.m. and 6 a.m. in mice, according to the study published in the early edition of the Proceedings of the National Academy of Sciences (doi:10.1073/pnas.1115249108).
They analyzed the expression pattern of XPA and its excision repair activity in mouse skin. One group of mice were exposed to UVB when repair activity was at its lowest in the morning, and another group was exposed when repair activity was at its peak, at 4 p.m.
Mice that were exposed when repair activity was low developed skin cancers faster and five times more frequently than mice exposed when repair activity was at its peak.
The authors reported that the human circadian clock is almost identical to the mouse clock, but that humans run on a diurnal pattern, while mice on a nocturnal pattern. Thus, humans are more susceptible to UV radiation in the late afternoon hours.
"Our research would suggest that restricting sunbathing or visits to the tanning booth to morning hours would reduce the risk of skin cancer in humans," said Dr. Sancar, in a statement issued by the UNC. Dr. Sancar is also a member of the UNC Lineberger Comprehensive Cancer Center.
Dermatologists might be a little miffed at Dr. Sancar's suggestion that there is a safe time to tan. But if his findings hold up, it might at least give physicians some leeway with patients who insist on getting some UV exposure. Maybe there will be a small window where getting sun is not as dangerous.
The researchers did note that findings "must be considered provisional until actual DNA repair rates are measured in the skin of human volunteers." That study is being planned.
Dr. Sancar's research received funding from the National Institutes of Health. The investigators reported having no conflicts of interest.
- Alicia Ault (on Twitter @aliciaault)
Dr. Aziz Sancar and colleagues at the University of North Carolina School of Medicine in Chapel Hill and the North Carolina State University in Raleigh conducted a study in hairless mice to see if the cyclical nature of a particular type of DNA repair might also influence the onset of skin cancer.
Dr. Sancar had already proven that a protein called XPA, which helps repair UV-related DNA damage, is primarily controlled by the circadian clock.
XPA activity reaches its peak between the hours of 4 p.m. and 6 p.m. and is at its lowest between 4 a.m. and 6 a.m. in mice, according to the study published in the early edition of the Proceedings of the National Academy of Sciences (doi:10.1073/pnas.1115249108).
They analyzed the expression pattern of XPA and its excision repair activity in mouse skin. One group of mice were exposed to UVB when repair activity was at its lowest in the morning, and another group was exposed when repair activity was at its peak, at 4 p.m.
Mice that were exposed when repair activity was low developed skin cancers faster and five times more frequently than mice exposed when repair activity was at its peak.
The authors reported that the human circadian clock is almost identical to the mouse clock, but that humans run on a diurnal pattern, while mice on a nocturnal pattern. Thus, humans are more susceptible to UV radiation in the late afternoon hours.
"Our research would suggest that restricting sunbathing or visits to the tanning booth to morning hours would reduce the risk of skin cancer in humans," said Dr. Sancar, in a statement issued by the UNC. Dr. Sancar is also a member of the UNC Lineberger Comprehensive Cancer Center.
Dermatologists might be a little miffed at Dr. Sancar's suggestion that there is a safe time to tan. But if his findings hold up, it might at least give physicians some leeway with patients who insist on getting some UV exposure. Maybe there will be a small window where getting sun is not as dangerous.
The researchers did note that findings "must be considered provisional until actual DNA repair rates are measured in the skin of human volunteers." That study is being planned.
Dr. Sancar's research received funding from the National Institutes of Health. The investigators reported having no conflicts of interest.
- Alicia Ault (on Twitter @aliciaault)
Dr. Aziz Sancar and colleagues at the University of North Carolina School of Medicine in Chapel Hill and the North Carolina State University in Raleigh conducted a study in hairless mice to see if the cyclical nature of a particular type of DNA repair might also influence the onset of skin cancer.
Dr. Sancar had already proven that a protein called XPA, which helps repair UV-related DNA damage, is primarily controlled by the circadian clock.
XPA activity reaches its peak between the hours of 4 p.m. and 6 p.m. and is at its lowest between 4 a.m. and 6 a.m. in mice, according to the study published in the early edition of the Proceedings of the National Academy of Sciences (doi:10.1073/pnas.1115249108).
They analyzed the expression pattern of XPA and its excision repair activity in mouse skin. One group of mice were exposed to UVB when repair activity was at its lowest in the morning, and another group was exposed when repair activity was at its peak, at 4 p.m.
Mice that were exposed when repair activity was low developed skin cancers faster and five times more frequently than mice exposed when repair activity was at its peak.
The authors reported that the human circadian clock is almost identical to the mouse clock, but that humans run on a diurnal pattern, while mice on a nocturnal pattern. Thus, humans are more susceptible to UV radiation in the late afternoon hours.
"Our research would suggest that restricting sunbathing or visits to the tanning booth to morning hours would reduce the risk of skin cancer in humans," said Dr. Sancar, in a statement issued by the UNC. Dr. Sancar is also a member of the UNC Lineberger Comprehensive Cancer Center.
Dermatologists might be a little miffed at Dr. Sancar's suggestion that there is a safe time to tan. But if his findings hold up, it might at least give physicians some leeway with patients who insist on getting some UV exposure. Maybe there will be a small window where getting sun is not as dangerous.
The researchers did note that findings "must be considered provisional until actual DNA repair rates are measured in the skin of human volunteers." That study is being planned.
Dr. Sancar's research received funding from the National Institutes of Health. The investigators reported having no conflicts of interest.
- Alicia Ault (on Twitter @aliciaault)
Survival Similar With 2- vs. 4-cm Margins for Thick Melanomas
A 2-cm resection margin resulted in the same 5-year overall survival and recurrence-free survival as did a 4-cm margin in a randomized controlled trial of more than 900 patients with cutaneous melanoma thicker than 2 mm.
The findings suggest that in patients with thicker melanomas, a 2-cm margin is safe and sufficient, according to Dr. Peter Gillgren of Karolinska Institute, Stockholm, and Stockholm Söder Hospital and his colleagues.
At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin. Recurrence-free survival at 5 years was 56% in both groups, and 10-year survival was 50% in both groups, the investigators reported (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61546-8]).
"The failure to clear genetically abnormal melanocytes with an adequately wide excision might be the precursor to locoregional recurrence."
Surgical resection margin size in patients with cutaneous melanoma thicker than 2 mm has been a point of controversy, largely due to a paucity of data comparing outcomes based on margin size, but most international guidelines suggest an excision margin of 2-3 cm for thick melanomas, the investigators noted. "A trade-off exists between a wide excision, with consequent surgical difficulties, and the relapse risk with a narrow excision, which could compromise disease-free survival, or worse, overall survival," they wrote.
However, the findings of this study indicate that not only is survival similar with 2-cm and 4-cm margins, but that the smaller margin size also improves the likelihood of skin closure without skin grafting or skin flaps. Primary closure was possible in 69% vs. 37% of patients in the 2-cm and 4-cm groups, respectively, while split skin grafts were used in 12% and 47% of the patients in the two groups. And surgical flaps were used in 4% and 6% of patients in the two groups, noted the investigators.
Patients in this study, which was launched by the Swedish Melanoma Study Group in cooperation with the Danish Melanoma Group, were adults aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm and with clinically localized disease on the trunk or upper or lower extremities. Patients were enrolled from Jan. 22, 1992, to May 19, 2004. One patient in each group was lost to follow-up but was included in the analysis.
Despite some limitations, such as protocol violations in 15% of cases and the fact that the study was planned as an equivalency trial that was to include 2,000 patients, the investigators noted that the study is the largest randomized controlled trial to date of resection margins for thick melanomas, and that they believe the results provide "the best evidence yet about the size of surgical excision margins."
"We show that with a surgical margin of 2 cm, the skin can be closed without skin grafting or skin flaps in most cases," they wrote, noting that previous data have already shown that hospital stay is longer in patients treated with a 4-cm margin, and that complication rates are higher in patients treated with split skin grafts, compared with primary sutures.
A meta-analysis of all randomized trials of cutaneous melanoma thicker than 2 mm should be conducted, they concluded.
The study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
In an editorial that accompanied the article by Dr. Gillgren and
colleagues, Dr. John F. Thompson and Dr. David W. Ollila reiterated that
the optimal excisional margin size for cutaneous melanoma has not been
clear. These findings, therefore, provide welcome news, but the next
question to be addressed is whether a 2-cm margin is preferable to a
1-cm margin, they wrote, noting that a large-scale multicenter trial to
address this question is in development (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61615-2]).

|
|
Another area of importance is "proper understanding of the inherent
tumor biology necessary for a safe excision margin," they noted,
explaining that assessment of margins using hematoxylin and eosin
staining is a "relatively crude pathological technique."
Investigators in another recent study used "comparative genomic
hybridization and fluorescent in situ hybridization to identify and map
genetically abnormal melanocytes in histopathologically normal epidermis
in acral melanoma wide excision specimens," Dr. Thompson of the
Melanoma Institute Australia at the Poche Centre in North Sydney and Dr.
Ollila of the University of North Carolina at Chapel Hill wrote.
They found abnormal melanocytes – extending a mean of 6.1 mm and 4.5
mm from the histologically assessed margin of in situ melanomas and
invasive melanomas, respectively – in 84% of 19 cases (J. Invest. Dermatol. 2008;128:2024-30).
"The failure to clear genetically abnormal melanocytes with an
adequately wide excision might be the precursor to locoregional
recurrence, which in turn could reduce survival," the investigators
wrote, noting that sophisticated multidisciplinary science may provide
the most rational approach to future recommendations for excisional
margin size in melanoma patients.
Dr. Thompson is with the Melanoma Institute Australia at the Poche Centre in North Sydney. Dr. Ollila is with the division of surgical oncology and endocrine surgery at the University of North Carolina at Chapel Hill. They reported having no relevant financial disclosures.
In an editorial that accompanied the article by Dr. Gillgren and
colleagues, Dr. John F. Thompson and Dr. David W. Ollila reiterated that
the optimal excisional margin size for cutaneous melanoma has not been
clear. These findings, therefore, provide welcome news, but the next
question to be addressed is whether a 2-cm margin is preferable to a
1-cm margin, they wrote, noting that a large-scale multicenter trial to
address this question is in development (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61615-2]).

|
|
Another area of importance is "proper understanding of the inherent
tumor biology necessary for a safe excision margin," they noted,
explaining that assessment of margins using hematoxylin and eosin
staining is a "relatively crude pathological technique."
Investigators in another recent study used "comparative genomic
hybridization and fluorescent in situ hybridization to identify and map
genetically abnormal melanocytes in histopathologically normal epidermis
in acral melanoma wide excision specimens," Dr. Thompson of the
Melanoma Institute Australia at the Poche Centre in North Sydney and Dr.
Ollila of the University of North Carolina at Chapel Hill wrote.
They found abnormal melanocytes – extending a mean of 6.1 mm and 4.5
mm from the histologically assessed margin of in situ melanomas and
invasive melanomas, respectively – in 84% of 19 cases (J. Invest. Dermatol. 2008;128:2024-30).
"The failure to clear genetically abnormal melanocytes with an
adequately wide excision might be the precursor to locoregional
recurrence, which in turn could reduce survival," the investigators
wrote, noting that sophisticated multidisciplinary science may provide
the most rational approach to future recommendations for excisional
margin size in melanoma patients.
Dr. Thompson is with the Melanoma Institute Australia at the Poche Centre in North Sydney. Dr. Ollila is with the division of surgical oncology and endocrine surgery at the University of North Carolina at Chapel Hill. They reported having no relevant financial disclosures.
In an editorial that accompanied the article by Dr. Gillgren and
colleagues, Dr. John F. Thompson and Dr. David W. Ollila reiterated that
the optimal excisional margin size for cutaneous melanoma has not been
clear. These findings, therefore, provide welcome news, but the next
question to be addressed is whether a 2-cm margin is preferable to a
1-cm margin, they wrote, noting that a large-scale multicenter trial to
address this question is in development (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61615-2]).

|
|
Another area of importance is "proper understanding of the inherent
tumor biology necessary for a safe excision margin," they noted,
explaining that assessment of margins using hematoxylin and eosin
staining is a "relatively crude pathological technique."
Investigators in another recent study used "comparative genomic
hybridization and fluorescent in situ hybridization to identify and map
genetically abnormal melanocytes in histopathologically normal epidermis
in acral melanoma wide excision specimens," Dr. Thompson of the
Melanoma Institute Australia at the Poche Centre in North Sydney and Dr.
Ollila of the University of North Carolina at Chapel Hill wrote.
They found abnormal melanocytes – extending a mean of 6.1 mm and 4.5
mm from the histologically assessed margin of in situ melanomas and
invasive melanomas, respectively – in 84% of 19 cases (J. Invest. Dermatol. 2008;128:2024-30).
"The failure to clear genetically abnormal melanocytes with an
adequately wide excision might be the precursor to locoregional
recurrence, which in turn could reduce survival," the investigators
wrote, noting that sophisticated multidisciplinary science may provide
the most rational approach to future recommendations for excisional
margin size in melanoma patients.
Dr. Thompson is with the Melanoma Institute Australia at the Poche Centre in North Sydney. Dr. Ollila is with the division of surgical oncology and endocrine surgery at the University of North Carolina at Chapel Hill. They reported having no relevant financial disclosures.
A 2-cm resection margin resulted in the same 5-year overall survival and recurrence-free survival as did a 4-cm margin in a randomized controlled trial of more than 900 patients with cutaneous melanoma thicker than 2 mm.
The findings suggest that in patients with thicker melanomas, a 2-cm margin is safe and sufficient, according to Dr. Peter Gillgren of Karolinska Institute, Stockholm, and Stockholm Söder Hospital and his colleagues.
At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin. Recurrence-free survival at 5 years was 56% in both groups, and 10-year survival was 50% in both groups, the investigators reported (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61546-8]).
"The failure to clear genetically abnormal melanocytes with an adequately wide excision might be the precursor to locoregional recurrence."
Surgical resection margin size in patients with cutaneous melanoma thicker than 2 mm has been a point of controversy, largely due to a paucity of data comparing outcomes based on margin size, but most international guidelines suggest an excision margin of 2-3 cm for thick melanomas, the investigators noted. "A trade-off exists between a wide excision, with consequent surgical difficulties, and the relapse risk with a narrow excision, which could compromise disease-free survival, or worse, overall survival," they wrote.
However, the findings of this study indicate that not only is survival similar with 2-cm and 4-cm margins, but that the smaller margin size also improves the likelihood of skin closure without skin grafting or skin flaps. Primary closure was possible in 69% vs. 37% of patients in the 2-cm and 4-cm groups, respectively, while split skin grafts were used in 12% and 47% of the patients in the two groups. And surgical flaps were used in 4% and 6% of patients in the two groups, noted the investigators.
Patients in this study, which was launched by the Swedish Melanoma Study Group in cooperation with the Danish Melanoma Group, were adults aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm and with clinically localized disease on the trunk or upper or lower extremities. Patients were enrolled from Jan. 22, 1992, to May 19, 2004. One patient in each group was lost to follow-up but was included in the analysis.
Despite some limitations, such as protocol violations in 15% of cases and the fact that the study was planned as an equivalency trial that was to include 2,000 patients, the investigators noted that the study is the largest randomized controlled trial to date of resection margins for thick melanomas, and that they believe the results provide "the best evidence yet about the size of surgical excision margins."
"We show that with a surgical margin of 2 cm, the skin can be closed without skin grafting or skin flaps in most cases," they wrote, noting that previous data have already shown that hospital stay is longer in patients treated with a 4-cm margin, and that complication rates are higher in patients treated with split skin grafts, compared with primary sutures.
A meta-analysis of all randomized trials of cutaneous melanoma thicker than 2 mm should be conducted, they concluded.
The study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
A 2-cm resection margin resulted in the same 5-year overall survival and recurrence-free survival as did a 4-cm margin in a randomized controlled trial of more than 900 patients with cutaneous melanoma thicker than 2 mm.
The findings suggest that in patients with thicker melanomas, a 2-cm margin is safe and sufficient, according to Dr. Peter Gillgren of Karolinska Institute, Stockholm, and Stockholm Söder Hospital and his colleagues.
At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin. Recurrence-free survival at 5 years was 56% in both groups, and 10-year survival was 50% in both groups, the investigators reported (Lancet 2011 Oct. 24 [doi:10.1016/S0140-6736(11)61546-8]).
"The failure to clear genetically abnormal melanocytes with an adequately wide excision might be the precursor to locoregional recurrence."
Surgical resection margin size in patients with cutaneous melanoma thicker than 2 mm has been a point of controversy, largely due to a paucity of data comparing outcomes based on margin size, but most international guidelines suggest an excision margin of 2-3 cm for thick melanomas, the investigators noted. "A trade-off exists between a wide excision, with consequent surgical difficulties, and the relapse risk with a narrow excision, which could compromise disease-free survival, or worse, overall survival," they wrote.
However, the findings of this study indicate that not only is survival similar with 2-cm and 4-cm margins, but that the smaller margin size also improves the likelihood of skin closure without skin grafting or skin flaps. Primary closure was possible in 69% vs. 37% of patients in the 2-cm and 4-cm groups, respectively, while split skin grafts were used in 12% and 47% of the patients in the two groups. And surgical flaps were used in 4% and 6% of patients in the two groups, noted the investigators.
Patients in this study, which was launched by the Swedish Melanoma Study Group in cooperation with the Danish Melanoma Group, were adults aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm and with clinically localized disease on the trunk or upper or lower extremities. Patients were enrolled from Jan. 22, 1992, to May 19, 2004. One patient in each group was lost to follow-up but was included in the analysis.
Despite some limitations, such as protocol violations in 15% of cases and the fact that the study was planned as an equivalency trial that was to include 2,000 patients, the investigators noted that the study is the largest randomized controlled trial to date of resection margins for thick melanomas, and that they believe the results provide "the best evidence yet about the size of surgical excision margins."
"We show that with a surgical margin of 2 cm, the skin can be closed without skin grafting or skin flaps in most cases," they wrote, noting that previous data have already shown that hospital stay is longer in patients treated with a 4-cm margin, and that complication rates are higher in patients treated with split skin grafts, compared with primary sutures.
A meta-analysis of all randomized trials of cutaneous melanoma thicker than 2 mm should be conducted, they concluded.
The study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
FROM THE LANCET
Major Finding: At a median follow-up of 6.7 years, the 5-year overall survival was 65% in 465 patients randomized to treatment with a 2-cm surgical resection margin, as well as in 471 patients randomized to treatment with a 4-cm resection margin.
Data Source: A multicenter, randomized controlled trial of 936 patients aged 75 years or younger with a primary cutaneous melanoma thicker than 2 mm.
Disclosures: This study was funded by the Swedish Cancer Society and the Stockholm Cancer Society. The authors reported having no relevant financial disclosures.
Series of Articles on Melanoma Aimed at Informing Policy
Melanoma was diagnosed in more than 45,000 people annually in the United States during 2004-2006, with a rate of 19 per 100,000 people.
That statistic, from two population-based cancer registries covering 78% of the U.S. population for those years, was among the melanoma data published in a series of articles in a special supplement to the November issue of the Journal of the American Academy of Dermatology (2011;65:S1-S156).
Among other significant findings were the following:
• Melanoma mortality accounted for $3.5 billion in lost productivity each year, with approximately $2.4 billion for men (average, $441,903 per man) and $1.2 billion for women (average, $401,046 per woman). Those who died from melanoma did so 20 years prematurely, compared with 17 years from other cancers, reported Donatus Ekwueme, Ph.D., of the Centers for Disease Control and Prevention.
• Melanoma rates were higher among white women aged 50 years and younger, Hispanic women aged 50 and younger, and Asian Pacific Islander women aged 40 and younger, compared with their male counterparts. Hispanics, American Indians/Alaska Natives, and Asians were diagnosed with melanoma at younger ages than were whites and blacks, according to analysis from Dr. Xiao-Cheng Wu of the school of public health at Louisiana State University in New Orleans.
• In 2005, 34% of adults reported having been sunburned in the past year, compared with 69% of adolescents, David Buller, Ph.D., of Klein Buendel Inc., a research firm, said in a report that also addressed the prevalence of sunburn, sun protection, and indoor-tanning behaviors.
• Fully 50% of dermatologists were unaware that they are required by state law to report their diagnosed cases of melanoma to their state central cancer registry, based on a survey of more than 400 dermatologists at a medical meeting. A slightly higher percentage (56%) do not actively report their melanoma diagnoses to a registry, according to the findings of the survey conducted by Dr. Todd Cartee of Emory University, Atlanta.
Other topics addressed include recent cutaneous melanoma trends and death rates, disease incidence in specific racial and ethnic populations as well as in adolescents and young adults, and associations between melanoma incidence and solar ultraviolet exposure and socioeconomic indicators. Also included are data on melanoma mortality and survival, sun-related behaviors such as tanning and sunscreen use, and rates of reporting.
The authors mentioned above declared no relevant conflicts of interest; the supplement was supported by the CDC.
Melanoma was diagnosed in more than 45,000 people annually in the United States during 2004-2006, with a rate of 19 per 100,000 people.
That statistic, from two population-based cancer registries covering 78% of the U.S. population for those years, was among the melanoma data published in a series of articles in a special supplement to the November issue of the Journal of the American Academy of Dermatology (2011;65:S1-S156).
Among other significant findings were the following:
• Melanoma mortality accounted for $3.5 billion in lost productivity each year, with approximately $2.4 billion for men (average, $441,903 per man) and $1.2 billion for women (average, $401,046 per woman). Those who died from melanoma did so 20 years prematurely, compared with 17 years from other cancers, reported Donatus Ekwueme, Ph.D., of the Centers for Disease Control and Prevention.
• Melanoma rates were higher among white women aged 50 years and younger, Hispanic women aged 50 and younger, and Asian Pacific Islander women aged 40 and younger, compared with their male counterparts. Hispanics, American Indians/Alaska Natives, and Asians were diagnosed with melanoma at younger ages than were whites and blacks, according to analysis from Dr. Xiao-Cheng Wu of the school of public health at Louisiana State University in New Orleans.
• In 2005, 34% of adults reported having been sunburned in the past year, compared with 69% of adolescents, David Buller, Ph.D., of Klein Buendel Inc., a research firm, said in a report that also addressed the prevalence of sunburn, sun protection, and indoor-tanning behaviors.
• Fully 50% of dermatologists were unaware that they are required by state law to report their diagnosed cases of melanoma to their state central cancer registry, based on a survey of more than 400 dermatologists at a medical meeting. A slightly higher percentage (56%) do not actively report their melanoma diagnoses to a registry, according to the findings of the survey conducted by Dr. Todd Cartee of Emory University, Atlanta.
Other topics addressed include recent cutaneous melanoma trends and death rates, disease incidence in specific racial and ethnic populations as well as in adolescents and young adults, and associations between melanoma incidence and solar ultraviolet exposure and socioeconomic indicators. Also included are data on melanoma mortality and survival, sun-related behaviors such as tanning and sunscreen use, and rates of reporting.
The authors mentioned above declared no relevant conflicts of interest; the supplement was supported by the CDC.
Melanoma was diagnosed in more than 45,000 people annually in the United States during 2004-2006, with a rate of 19 per 100,000 people.
That statistic, from two population-based cancer registries covering 78% of the U.S. population for those years, was among the melanoma data published in a series of articles in a special supplement to the November issue of the Journal of the American Academy of Dermatology (2011;65:S1-S156).
Among other significant findings were the following:
• Melanoma mortality accounted for $3.5 billion in lost productivity each year, with approximately $2.4 billion for men (average, $441,903 per man) and $1.2 billion for women (average, $401,046 per woman). Those who died from melanoma did so 20 years prematurely, compared with 17 years from other cancers, reported Donatus Ekwueme, Ph.D., of the Centers for Disease Control and Prevention.
• Melanoma rates were higher among white women aged 50 years and younger, Hispanic women aged 50 and younger, and Asian Pacific Islander women aged 40 and younger, compared with their male counterparts. Hispanics, American Indians/Alaska Natives, and Asians were diagnosed with melanoma at younger ages than were whites and blacks, according to analysis from Dr. Xiao-Cheng Wu of the school of public health at Louisiana State University in New Orleans.
• In 2005, 34% of adults reported having been sunburned in the past year, compared with 69% of adolescents, David Buller, Ph.D., of Klein Buendel Inc., a research firm, said in a report that also addressed the prevalence of sunburn, sun protection, and indoor-tanning behaviors.
• Fully 50% of dermatologists were unaware that they are required by state law to report their diagnosed cases of melanoma to their state central cancer registry, based on a survey of more than 400 dermatologists at a medical meeting. A slightly higher percentage (56%) do not actively report their melanoma diagnoses to a registry, according to the findings of the survey conducted by Dr. Todd Cartee of Emory University, Atlanta.
Other topics addressed include recent cutaneous melanoma trends and death rates, disease incidence in specific racial and ethnic populations as well as in adolescents and young adults, and associations between melanoma incidence and solar ultraviolet exposure and socioeconomic indicators. Also included are data on melanoma mortality and survival, sun-related behaviors such as tanning and sunscreen use, and rates of reporting.
The authors mentioned above declared no relevant conflicts of interest; the supplement was supported by the CDC.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Definition of Giant Congenital Nevus Refined
SAN FRANCISCO – A new definition of giant congenital nevi should help physicians identify infants with supersized moles that are at risk for melanoma, neurocutaneous melanocytosis, liposarcoma, and other tumors.
For many years pediatric dermatologists sized congenital nevi as small, medium, or large, with "large" comprising nevi that are estimated to be 20 cm or greater in diameter when the patient has grown into adulthood. Then, physicians started referring to some of the large nevi as "giant" congenital nevi, with no universal agreement on what defines a giant nevus, Dr. Sheila Fallon Friedlander said at the Women’s & Pediatric Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
"Here’s our new definition that you’re going to be seeing in the literature," said Dr. Friedlander of the University of California, San Diego. Congenital nevi with diameters less than 1.5 cm after the patient is fully grown are considered small, nevi with diameters of at least 1.5 cm but less than 20 cm are medium size, and large nevi are at least 20 cm but less than 40 cm in diameter. Giant congenital nevi are those with diameters of 40 cm or greater when the patient is fully grown.
"When you are examining a baby in a nursery, how does that help you? That’s an adult-type figure," she noted. To estimate the grown size of a congenital nevus, measure the nevus on the baby and multiply by a scaling factor that’s based on different growth rates for different parts of the body. Lower extremities grow the most, so multiply the size of nevi located there by 3.3. Multiply the size of a nevus on the torso or an upper extremity by 2.8, and a nevus on the head by 1.7.
For example, a congenital nevus 7 cm in diameter on a baby’s leg qualifies as a large congenital nevus (7 x 3.3 = 23.1 cm), "so you need to worry more" about risks for that baby, she said.
Estimates of risk have been evolving with the terminology. Most studies have lumped large and giant categories together and referred to them as large congenital nevi. The true risks in studies of registry data are clouded by the fact that many families choose to excise or partially excise large congenital nevi.
Between 1% and 11% of patients with large congenital nevi will develop malignant melanoma or neurocutaneous melanocytosis, a 52-1,000 times greater risk than for someone without the lesion, according to the results of a New York University study of 160 patients (Pediatrics 2000;106:736-41). Large congenital nevi also are associated with increased risks for liposarcoma, rhabdomyosarcoma, and (in some body locations) tethered spinal cord syndrome.
In the New York University study, 14 of 15 patients who developed melanoma or neurocutaneous melanocytosis had lesions greater than 50 cm in diameter, which now would be considered giant. The risk is highest for lesions on the torso. The melanoma often can occur deep in the dermal-epidermal junction, making them hard to find, Dr. Friedlander said. The melanoma also can occur underneath the lesion in the CNS or mesentery.
Although melanomas in the general population are unlikely to develop until adolescence or adulthood, in children with large congenital nevi who develop melanoma, 70% of the cancers appear by age 10 years, and 50% appear by age 5. "So this is something that you can’t put off. You really need to deal with it," she said.
Studies report that 7% of patients with large congenital nevi will develop neurocutaneous melanocytosis, an aberrant migration of melanocytes in leptomeninges that creates a risk for ventricular obstruction. The presence of more than 20 satellite lesions around a large congenital nevus increases the risk for neurocutaneous melanocytosis five-fold and warrants an MRI evaluation, she said.
"Five years ago I was taught that posterior axial location may have a higher risk for neurocutaneous melanocytosis, but recent data shows that it’s the satellite lesions rather than the location" that matters, Dr. Friedlander said.
A posterior axial location is, however, a risk factor for cutaneous melanoma in patients with large congenital nevi. Nevus size is a risk factor for both melanoma and neurocutaneous melanocytosis, said Dr. Friedlander; "20 cm is bad, and 40 cm is worse."
Dr. Friedlander disclosed that she has been a speaker, consultant, or researcher for Pierre Fabre, Onset Therapeutics, Johnson & Johnson, and Galderma.
SDEF and this news organization are owned by Elsevier.
SAN FRANCISCO – A new definition of giant congenital nevi should help physicians identify infants with supersized moles that are at risk for melanoma, neurocutaneous melanocytosis, liposarcoma, and other tumors.
For many years pediatric dermatologists sized congenital nevi as small, medium, or large, with "large" comprising nevi that are estimated to be 20 cm or greater in diameter when the patient has grown into adulthood. Then, physicians started referring to some of the large nevi as "giant" congenital nevi, with no universal agreement on what defines a giant nevus, Dr. Sheila Fallon Friedlander said at the Women’s & Pediatric Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
"Here’s our new definition that you’re going to be seeing in the literature," said Dr. Friedlander of the University of California, San Diego. Congenital nevi with diameters less than 1.5 cm after the patient is fully grown are considered small, nevi with diameters of at least 1.5 cm but less than 20 cm are medium size, and large nevi are at least 20 cm but less than 40 cm in diameter. Giant congenital nevi are those with diameters of 40 cm or greater when the patient is fully grown.
"When you are examining a baby in a nursery, how does that help you? That’s an adult-type figure," she noted. To estimate the grown size of a congenital nevus, measure the nevus on the baby and multiply by a scaling factor that’s based on different growth rates for different parts of the body. Lower extremities grow the most, so multiply the size of nevi located there by 3.3. Multiply the size of a nevus on the torso or an upper extremity by 2.8, and a nevus on the head by 1.7.
For example, a congenital nevus 7 cm in diameter on a baby’s leg qualifies as a large congenital nevus (7 x 3.3 = 23.1 cm), "so you need to worry more" about risks for that baby, she said.
Estimates of risk have been evolving with the terminology. Most studies have lumped large and giant categories together and referred to them as large congenital nevi. The true risks in studies of registry data are clouded by the fact that many families choose to excise or partially excise large congenital nevi.
Between 1% and 11% of patients with large congenital nevi will develop malignant melanoma or neurocutaneous melanocytosis, a 52-1,000 times greater risk than for someone without the lesion, according to the results of a New York University study of 160 patients (Pediatrics 2000;106:736-41). Large congenital nevi also are associated with increased risks for liposarcoma, rhabdomyosarcoma, and (in some body locations) tethered spinal cord syndrome.
In the New York University study, 14 of 15 patients who developed melanoma or neurocutaneous melanocytosis had lesions greater than 50 cm in diameter, which now would be considered giant. The risk is highest for lesions on the torso. The melanoma often can occur deep in the dermal-epidermal junction, making them hard to find, Dr. Friedlander said. The melanoma also can occur underneath the lesion in the CNS or mesentery.
Although melanomas in the general population are unlikely to develop until adolescence or adulthood, in children with large congenital nevi who develop melanoma, 70% of the cancers appear by age 10 years, and 50% appear by age 5. "So this is something that you can’t put off. You really need to deal with it," she said.
Studies report that 7% of patients with large congenital nevi will develop neurocutaneous melanocytosis, an aberrant migration of melanocytes in leptomeninges that creates a risk for ventricular obstruction. The presence of more than 20 satellite lesions around a large congenital nevus increases the risk for neurocutaneous melanocytosis five-fold and warrants an MRI evaluation, she said.
"Five years ago I was taught that posterior axial location may have a higher risk for neurocutaneous melanocytosis, but recent data shows that it’s the satellite lesions rather than the location" that matters, Dr. Friedlander said.
A posterior axial location is, however, a risk factor for cutaneous melanoma in patients with large congenital nevi. Nevus size is a risk factor for both melanoma and neurocutaneous melanocytosis, said Dr. Friedlander; "20 cm is bad, and 40 cm is worse."
Dr. Friedlander disclosed that she has been a speaker, consultant, or researcher for Pierre Fabre, Onset Therapeutics, Johnson & Johnson, and Galderma.
SDEF and this news organization are owned by Elsevier.
SAN FRANCISCO – A new definition of giant congenital nevi should help physicians identify infants with supersized moles that are at risk for melanoma, neurocutaneous melanocytosis, liposarcoma, and other tumors.
For many years pediatric dermatologists sized congenital nevi as small, medium, or large, with "large" comprising nevi that are estimated to be 20 cm or greater in diameter when the patient has grown into adulthood. Then, physicians started referring to some of the large nevi as "giant" congenital nevi, with no universal agreement on what defines a giant nevus, Dr. Sheila Fallon Friedlander said at the Women’s & Pediatric Dermatology Seminar sponsored by Skin Disease Education Foundation (SDEF).
"Here’s our new definition that you’re going to be seeing in the literature," said Dr. Friedlander of the University of California, San Diego. Congenital nevi with diameters less than 1.5 cm after the patient is fully grown are considered small, nevi with diameters of at least 1.5 cm but less than 20 cm are medium size, and large nevi are at least 20 cm but less than 40 cm in diameter. Giant congenital nevi are those with diameters of 40 cm or greater when the patient is fully grown.
"When you are examining a baby in a nursery, how does that help you? That’s an adult-type figure," she noted. To estimate the grown size of a congenital nevus, measure the nevus on the baby and multiply by a scaling factor that’s based on different growth rates for different parts of the body. Lower extremities grow the most, so multiply the size of nevi located there by 3.3. Multiply the size of a nevus on the torso or an upper extremity by 2.8, and a nevus on the head by 1.7.
For example, a congenital nevus 7 cm in diameter on a baby’s leg qualifies as a large congenital nevus (7 x 3.3 = 23.1 cm), "so you need to worry more" about risks for that baby, she said.
Estimates of risk have been evolving with the terminology. Most studies have lumped large and giant categories together and referred to them as large congenital nevi. The true risks in studies of registry data are clouded by the fact that many families choose to excise or partially excise large congenital nevi.
Between 1% and 11% of patients with large congenital nevi will develop malignant melanoma or neurocutaneous melanocytosis, a 52-1,000 times greater risk than for someone without the lesion, according to the results of a New York University study of 160 patients (Pediatrics 2000;106:736-41). Large congenital nevi also are associated with increased risks for liposarcoma, rhabdomyosarcoma, and (in some body locations) tethered spinal cord syndrome.
In the New York University study, 14 of 15 patients who developed melanoma or neurocutaneous melanocytosis had lesions greater than 50 cm in diameter, which now would be considered giant. The risk is highest for lesions on the torso. The melanoma often can occur deep in the dermal-epidermal junction, making them hard to find, Dr. Friedlander said. The melanoma also can occur underneath the lesion in the CNS or mesentery.
Although melanomas in the general population are unlikely to develop until adolescence or adulthood, in children with large congenital nevi who develop melanoma, 70% of the cancers appear by age 10 years, and 50% appear by age 5. "So this is something that you can’t put off. You really need to deal with it," she said.
Studies report that 7% of patients with large congenital nevi will develop neurocutaneous melanocytosis, an aberrant migration of melanocytes in leptomeninges that creates a risk for ventricular obstruction. The presence of more than 20 satellite lesions around a large congenital nevus increases the risk for neurocutaneous melanocytosis five-fold and warrants an MRI evaluation, she said.
"Five years ago I was taught that posterior axial location may have a higher risk for neurocutaneous melanocytosis, but recent data shows that it’s the satellite lesions rather than the location" that matters, Dr. Friedlander said.
A posterior axial location is, however, a risk factor for cutaneous melanoma in patients with large congenital nevi. Nevus size is a risk factor for both melanoma and neurocutaneous melanocytosis, said Dr. Friedlander; "20 cm is bad, and 40 cm is worse."
Dr. Friedlander disclosed that she has been a speaker, consultant, or researcher for Pierre Fabre, Onset Therapeutics, Johnson & Johnson, and Galderma.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM THE SDEF WOMEN'S & PEDIATRIC DERMATOLOGY SEMINAR