User login
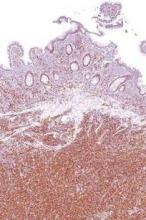
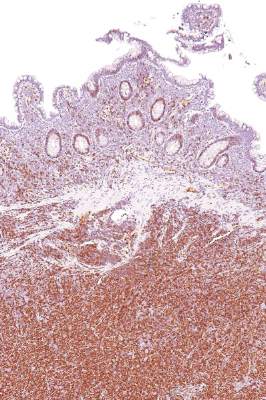
Intralesional interferon excels for challenging basal cell carcinomas
WAIKOLOA, HAWAII – Intralesional injection of interferon alfa-2b is an excellent option for the treatment of large problematic basal cell carcinomas in patients who aren’t interested in the higher-morbidity options, Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“This is a really effective way to treat basal cell carcinomas. These things just melt away before your eyes. It’s really quite amazing,” observed Dr. Swanson of Mayo Clinic Scottsdale (Ariz.).
He finds this therapy particularly useful in frail elderly patients who have a large BCC on the head or neck. A good example would be an 89-year-old with multiple comorbid conditions who has a 2-cm BCC on the tip of the nose and doesn’t want anything done about it. The patient declines the options of Mohs micrographic surgery or radiotherapy.
“This is the patient who just wants to be left alone. That’s fine if they’re going to be dead within a year, but if they’re going to be around for several years, that basal cell carcinoma could become a major issue for them,” the dermatologist continued.
He and his colleagues at the Mayo Clinic follow a treatment regimen similar to one laid out by Turkish investigators more than a decade ago in one of the few long-term outcome studies of intralesional interferon for treatment of BCCs.
Although interferon alfa-2b is approved for the intralesional treatment of genital warts and subcutaneously for Kaposi’s sarcoma and malignant melanoma, among other conditions, it’s off-label therapy for BCCs. The treatment entails thrice-weekly intralesional injections for 3 weeks. The dosing is 1.5 million units per injection for BCCs smaller than 2 cm and 3 million units per injection for BCCs that are 2 cm or larger. The injections are given without anesthesia, but premedication with 500-1,000 mg of acetaminophen is advisable to minimize aches and fever.
Interferon alfa-2b (Intron A) comes in a vial containing 10 million units with 1 mL of diluent. It’s important to reconstitute it carefully, similar to onabotulinumtoxin. Don’t shake it, Dr. Swanson advised.
The Turkish report included 20 patients with histopathologically proven BCCs on the head or neck. At clinical and dermatopathologic follow-up 8 weeks after the last interferon injection, 11 BCCs showed complete clinical and histopathologic cure, six showed partial remission, two showed no response, and one actually increased in size during treatment.
The 11 patients with an initial complete cure were followed for 7 years. During that period, only one of the 11 skin cancers recurred, at the fifth year (Clin Drug Investig. 2005;25[10]:661-7).
Dr. Swanson reported having no financial conflicts regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Intralesional injection of interferon alfa-2b is an excellent option for the treatment of large problematic basal cell carcinomas in patients who aren’t interested in the higher-morbidity options, Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“This is a really effective way to treat basal cell carcinomas. These things just melt away before your eyes. It’s really quite amazing,” observed Dr. Swanson of Mayo Clinic Scottsdale (Ariz.).
He finds this therapy particularly useful in frail elderly patients who have a large BCC on the head or neck. A good example would be an 89-year-old with multiple comorbid conditions who has a 2-cm BCC on the tip of the nose and doesn’t want anything done about it. The patient declines the options of Mohs micrographic surgery or radiotherapy.
“This is the patient who just wants to be left alone. That’s fine if they’re going to be dead within a year, but if they’re going to be around for several years, that basal cell carcinoma could become a major issue for them,” the dermatologist continued.
He and his colleagues at the Mayo Clinic follow a treatment regimen similar to one laid out by Turkish investigators more than a decade ago in one of the few long-term outcome studies of intralesional interferon for treatment of BCCs.
Although interferon alfa-2b is approved for the intralesional treatment of genital warts and subcutaneously for Kaposi’s sarcoma and malignant melanoma, among other conditions, it’s off-label therapy for BCCs. The treatment entails thrice-weekly intralesional injections for 3 weeks. The dosing is 1.5 million units per injection for BCCs smaller than 2 cm and 3 million units per injection for BCCs that are 2 cm or larger. The injections are given without anesthesia, but premedication with 500-1,000 mg of acetaminophen is advisable to minimize aches and fever.
Interferon alfa-2b (Intron A) comes in a vial containing 10 million units with 1 mL of diluent. It’s important to reconstitute it carefully, similar to onabotulinumtoxin. Don’t shake it, Dr. Swanson advised.
The Turkish report included 20 patients with histopathologically proven BCCs on the head or neck. At clinical and dermatopathologic follow-up 8 weeks after the last interferon injection, 11 BCCs showed complete clinical and histopathologic cure, six showed partial remission, two showed no response, and one actually increased in size during treatment.
The 11 patients with an initial complete cure were followed for 7 years. During that period, only one of the 11 skin cancers recurred, at the fifth year (Clin Drug Investig. 2005;25[10]:661-7).
Dr. Swanson reported having no financial conflicts regarding his presentation. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – Intralesional injection of interferon alfa-2b is an excellent option for the treatment of large problematic basal cell carcinomas in patients who aren’t interested in the higher-morbidity options, Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
“This is a really effective way to treat basal cell carcinomas. These things just melt away before your eyes. It’s really quite amazing,” observed Dr. Swanson of Mayo Clinic Scottsdale (Ariz.).
He finds this therapy particularly useful in frail elderly patients who have a large BCC on the head or neck. A good example would be an 89-year-old with multiple comorbid conditions who has a 2-cm BCC on the tip of the nose and doesn’t want anything done about it. The patient declines the options of Mohs micrographic surgery or radiotherapy.
“This is the patient who just wants to be left alone. That’s fine if they’re going to be dead within a year, but if they’re going to be around for several years, that basal cell carcinoma could become a major issue for them,” the dermatologist continued.
He and his colleagues at the Mayo Clinic follow a treatment regimen similar to one laid out by Turkish investigators more than a decade ago in one of the few long-term outcome studies of intralesional interferon for treatment of BCCs.
Although interferon alfa-2b is approved for the intralesional treatment of genital warts and subcutaneously for Kaposi’s sarcoma and malignant melanoma, among other conditions, it’s off-label therapy for BCCs. The treatment entails thrice-weekly intralesional injections for 3 weeks. The dosing is 1.5 million units per injection for BCCs smaller than 2 cm and 3 million units per injection for BCCs that are 2 cm or larger. The injections are given without anesthesia, but premedication with 500-1,000 mg of acetaminophen is advisable to minimize aches and fever.
Interferon alfa-2b (Intron A) comes in a vial containing 10 million units with 1 mL of diluent. It’s important to reconstitute it carefully, similar to onabotulinumtoxin. Don’t shake it, Dr. Swanson advised.
The Turkish report included 20 patients with histopathologically proven BCCs on the head or neck. At clinical and dermatopathologic follow-up 8 weeks after the last interferon injection, 11 BCCs showed complete clinical and histopathologic cure, six showed partial remission, two showed no response, and one actually increased in size during treatment.
The 11 patients with an initial complete cure were followed for 7 years. During that period, only one of the 11 skin cancers recurred, at the fifth year (Clin Drug Investig. 2005;25[10]:661-7).
Dr. Swanson reported having no financial conflicts regarding his presentation. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Tools for Diagnosing Skin Cancer Earlier: Report From the AAD Meeting
At the 74th Annual Meeting of the American Academy of Dermatology, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She provides highlights from this session, including the use of dermoscopy and optical coherence technology to detect features of early melanoma and nonmelanoma skin cancers as well as monitor skin cancer management. A lesion that is pink clinically but shows pigment dermoscopically should be biopsied, Dr. Markowtiz advises, as it may be an early amelanotic melanoma. She also notes that noninvasive imaging tools can be used to detect residual tumor cells in treated skin that otherwise looks clinically normal.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At the 74th Annual Meeting of the American Academy of Dermatology, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She provides highlights from this session, including the use of dermoscopy and optical coherence technology to detect features of early melanoma and nonmelanoma skin cancers as well as monitor skin cancer management. A lesion that is pink clinically but shows pigment dermoscopically should be biopsied, Dr. Markowtiz advises, as it may be an early amelanotic melanoma. She also notes that noninvasive imaging tools can be used to detect residual tumor cells in treated skin that otherwise looks clinically normal.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At the 74th Annual Meeting of the American Academy of Dermatology, Dr. Orit Markowitz discussed noninvasive imaging tools that can help dermatologists diagnose skin cancers earlier. She provides highlights from this session, including the use of dermoscopy and optical coherence technology to detect features of early melanoma and nonmelanoma skin cancers as well as monitor skin cancer management. A lesion that is pink clinically but shows pigment dermoscopically should be biopsied, Dr. Markowtiz advises, as it may be an early amelanotic melanoma. She also notes that noninvasive imaging tools can be used to detect residual tumor cells in treated skin that otherwise looks clinically normal.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Does sharing genetic risk change behavior?
In the era of individualized (or precision) medicine, we are presented with a unique opportunity to peer into the genetic “maps” of our patients. Through this window, we can envision the self-evident present or predict a possible future.
For the front-line provider, knowing that we could someday have a large amount of these data to deal with can be overwhelming. We may be loath to think that, amongst all the other daily battles we wage with current disease states, we may now need to understand and explain risk for future disease states.
But would we be more likely to use these data if we thought that they would change patient behavior? Maybe.
So does it?
Gareth Hollands, Ph.D., of the University of Cambridge, England, and his colleagues conducted a brilliantly timed and welcome systematic review of the literature assessing the impact of communicating DNA-based disease risk estimates on risk-reducing health behaviors and motivation to engage in such behaviors (BMJ. 2016 Mar 15;352:i1102).
Eighteen studies were found reporting on seven behavioral outcomes, including smoking cessation (six studies, n = 2,663), diet (seven studies, n = 1,784), and physical activity (six studies, n = 1,704). The smoking studies related genetic risk for lung or esophageal cancer; the diet studies related risk for diabetes, obesity, cardiovascular disease, hypertension, hyperlipidemia, and Alzheimer’s disease; and the physical activity studies related risks similar to the diet studies.
No evidence was found that communicating DNA-based risk increased smoking cessation or led to positive changes in diet or physical activity. Nor did the investigators find any effects on motivation to change behavior. Although this information is not motivating to patients, no evidence was found suggesting that it is demotivating, either.
If neither behavior nor motivation is modified by DNA-based risk assessment, what is it good for? As the authors pointed out, this information can be used for clinical risk stratification and for refining screening and treatment procedures.
It’s important to note that this puts the responsibility for the required action in response to DNA data in the hands of medical providers – sadly reminding us that the list of ways to motivate patients to change behavior remains frustratingly short.
Dr. Ebbert is professor of medicine, a general internist at the Mayo Clinic in Rochester, Minn., and a diplomate of the American Board of Addiction Medicine. The opinions expressed are those of the author and do not necessarily represent the views and opinions of the Mayo Clinic. The opinions expressed in this article should not be used to diagnose or treat any medical condition nor should they be used as a substitute for medical advice from a qualified, board-certified practicing clinician. Dr. Ebbert has no relevant financial disclosures about this article.
In the era of individualized (or precision) medicine, we are presented with a unique opportunity to peer into the genetic “maps” of our patients. Through this window, we can envision the self-evident present or predict a possible future.
For the front-line provider, knowing that we could someday have a large amount of these data to deal with can be overwhelming. We may be loath to think that, amongst all the other daily battles we wage with current disease states, we may now need to understand and explain risk for future disease states.
But would we be more likely to use these data if we thought that they would change patient behavior? Maybe.
So does it?
Gareth Hollands, Ph.D., of the University of Cambridge, England, and his colleagues conducted a brilliantly timed and welcome systematic review of the literature assessing the impact of communicating DNA-based disease risk estimates on risk-reducing health behaviors and motivation to engage in such behaviors (BMJ. 2016 Mar 15;352:i1102).
Eighteen studies were found reporting on seven behavioral outcomes, including smoking cessation (six studies, n = 2,663), diet (seven studies, n = 1,784), and physical activity (six studies, n = 1,704). The smoking studies related genetic risk for lung or esophageal cancer; the diet studies related risk for diabetes, obesity, cardiovascular disease, hypertension, hyperlipidemia, and Alzheimer’s disease; and the physical activity studies related risks similar to the diet studies.
No evidence was found that communicating DNA-based risk increased smoking cessation or led to positive changes in diet or physical activity. Nor did the investigators find any effects on motivation to change behavior. Although this information is not motivating to patients, no evidence was found suggesting that it is demotivating, either.
If neither behavior nor motivation is modified by DNA-based risk assessment, what is it good for? As the authors pointed out, this information can be used for clinical risk stratification and for refining screening and treatment procedures.
It’s important to note that this puts the responsibility for the required action in response to DNA data in the hands of medical providers – sadly reminding us that the list of ways to motivate patients to change behavior remains frustratingly short.
Dr. Ebbert is professor of medicine, a general internist at the Mayo Clinic in Rochester, Minn., and a diplomate of the American Board of Addiction Medicine. The opinions expressed are those of the author and do not necessarily represent the views and opinions of the Mayo Clinic. The opinions expressed in this article should not be used to diagnose or treat any medical condition nor should they be used as a substitute for medical advice from a qualified, board-certified practicing clinician. Dr. Ebbert has no relevant financial disclosures about this article.
In the era of individualized (or precision) medicine, we are presented with a unique opportunity to peer into the genetic “maps” of our patients. Through this window, we can envision the self-evident present or predict a possible future.
For the front-line provider, knowing that we could someday have a large amount of these data to deal with can be overwhelming. We may be loath to think that, amongst all the other daily battles we wage with current disease states, we may now need to understand and explain risk for future disease states.
But would we be more likely to use these data if we thought that they would change patient behavior? Maybe.
So does it?
Gareth Hollands, Ph.D., of the University of Cambridge, England, and his colleagues conducted a brilliantly timed and welcome systematic review of the literature assessing the impact of communicating DNA-based disease risk estimates on risk-reducing health behaviors and motivation to engage in such behaviors (BMJ. 2016 Mar 15;352:i1102).
Eighteen studies were found reporting on seven behavioral outcomes, including smoking cessation (six studies, n = 2,663), diet (seven studies, n = 1,784), and physical activity (six studies, n = 1,704). The smoking studies related genetic risk for lung or esophageal cancer; the diet studies related risk for diabetes, obesity, cardiovascular disease, hypertension, hyperlipidemia, and Alzheimer’s disease; and the physical activity studies related risks similar to the diet studies.
No evidence was found that communicating DNA-based risk increased smoking cessation or led to positive changes in diet or physical activity. Nor did the investigators find any effects on motivation to change behavior. Although this information is not motivating to patients, no evidence was found suggesting that it is demotivating, either.
If neither behavior nor motivation is modified by DNA-based risk assessment, what is it good for? As the authors pointed out, this information can be used for clinical risk stratification and for refining screening and treatment procedures.
It’s important to note that this puts the responsibility for the required action in response to DNA data in the hands of medical providers – sadly reminding us that the list of ways to motivate patients to change behavior remains frustratingly short.
Dr. Ebbert is professor of medicine, a general internist at the Mayo Clinic in Rochester, Minn., and a diplomate of the American Board of Addiction Medicine. The opinions expressed are those of the author and do not necessarily represent the views and opinions of the Mayo Clinic. The opinions expressed in this article should not be used to diagnose or treat any medical condition nor should they be used as a substitute for medical advice from a qualified, board-certified practicing clinician. Dr. Ebbert has no relevant financial disclosures about this article.
Low vitamin D linked with worse outcome in melanoma
Lower levels of vitamin D in patients with melanoma were associated with poorer survival, independent of effects from systemic inflammation indicated by simultaneous C-reactive protein (CRP) measures, according to researchers.
Multivariate analysis showed that vitamin D level was associated with overall survival (OS) (hazard ratio, 1.02 per unit decrease of vitamin D; 95% confidence interval, 1.01-1.04; P = .005), melanoma-specific survival (MSS) (HR, 1.02; 95% CI, 1.00-1.04; P = .048), and disease-free survival (DFS) (HR, 1.02; 95% CI, 1.00-1.04; P = .043). Patients with a vitamin D level less than 16 ng/mL* were 2.0 times more likely to die of all-cause disease than were patients with a higher level (95% CI, 1.50-2.66; P less than .001), investigators reported (J Clin Oncol. 2016 Mar 21. doi: 10.1200/JCO.2015.64.1357).
“Importantly, after adjustment for CRP, vitamin D remained an independent predictor of OS, MSS, and DFS. This suggests that, although blood levels of vitamin D and CRP are highly correlated with each other, each independently predicts clinical outcome in patients with melanoma,” wrote Dr. Shenying Fang of the University of Texas MD Anderson Cancer Center, Houston, and colleagues.
Although the role of the biomarkers in disease remains unclear and warrants further research, the authors added, “these data suggest that interventions to increase vitamin D or to reduce [systematic inflammatory response] and CRP could ultimately benefit patients with melanoma.”
Previous research has demonstrated an association between vitamin D deficiency and advanced melanoma stage, but investigations of vitamin D blood levels and melanoma risk have yielded inconsistent results. The researchers examined confounders, such as systemic inflammatory response (assessed by simultaneous CRP measurement), age, disease stage, and blood draw season to assess the relationship between vitamin D level and melanoma outcomes.
Decreased vitamin D was significantly associated with higher patient age, increased primary tumor thickness, ulcerated tumors, advanced-stage disease at blood draw, and increased CRP. Blood drawn in the fall/winter had lower vitamin D levels than that of spring/summer, reflecting average differences in sun exposure. These factors were included in the multivariable model.
A vitamin D level less than 20 ng/mL* is considered deficient, and deficient levels were significantly associated with poorer OS and DFS but not with poorer MSS. At levels below the optimal cutoff, determined by recursive partitioning, of 16 ng/mL*, patients had poorer OS (HR, 2.0; 95% CI, 1.50-2.66; P less than .001), MSS (HR, 1.76; 95% CI, 1.22-2.53; P = .003), and DFS (HR, 1.62; 95% CI, 1.04-2.53; P = .036) on univariate analysis, and associations remained significant on multivariable analysis.
The hospital-based investigation evaluated peripheral blood samples collected from 1,042 non-Hispanic white patients with melanoma from 1997 to 2009.
Dr. Fang reported having no disclosures. Several of his coauthors reported financial ties to industry sources.
*Correction: An earlier version of this article used an incorrect unit of measurement for vitamin D deficiency.
Lower levels of vitamin D in patients with melanoma were associated with poorer survival, independent of effects from systemic inflammation indicated by simultaneous C-reactive protein (CRP) measures, according to researchers.
Multivariate analysis showed that vitamin D level was associated with overall survival (OS) (hazard ratio, 1.02 per unit decrease of vitamin D; 95% confidence interval, 1.01-1.04; P = .005), melanoma-specific survival (MSS) (HR, 1.02; 95% CI, 1.00-1.04; P = .048), and disease-free survival (DFS) (HR, 1.02; 95% CI, 1.00-1.04; P = .043). Patients with a vitamin D level less than 16 ng/mL* were 2.0 times more likely to die of all-cause disease than were patients with a higher level (95% CI, 1.50-2.66; P less than .001), investigators reported (J Clin Oncol. 2016 Mar 21. doi: 10.1200/JCO.2015.64.1357).
“Importantly, after adjustment for CRP, vitamin D remained an independent predictor of OS, MSS, and DFS. This suggests that, although blood levels of vitamin D and CRP are highly correlated with each other, each independently predicts clinical outcome in patients with melanoma,” wrote Dr. Shenying Fang of the University of Texas MD Anderson Cancer Center, Houston, and colleagues.
Although the role of the biomarkers in disease remains unclear and warrants further research, the authors added, “these data suggest that interventions to increase vitamin D or to reduce [systematic inflammatory response] and CRP could ultimately benefit patients with melanoma.”
Previous research has demonstrated an association between vitamin D deficiency and advanced melanoma stage, but investigations of vitamin D blood levels and melanoma risk have yielded inconsistent results. The researchers examined confounders, such as systemic inflammatory response (assessed by simultaneous CRP measurement), age, disease stage, and blood draw season to assess the relationship between vitamin D level and melanoma outcomes.
Decreased vitamin D was significantly associated with higher patient age, increased primary tumor thickness, ulcerated tumors, advanced-stage disease at blood draw, and increased CRP. Blood drawn in the fall/winter had lower vitamin D levels than that of spring/summer, reflecting average differences in sun exposure. These factors were included in the multivariable model.
A vitamin D level less than 20 ng/mL* is considered deficient, and deficient levels were significantly associated with poorer OS and DFS but not with poorer MSS. At levels below the optimal cutoff, determined by recursive partitioning, of 16 ng/mL*, patients had poorer OS (HR, 2.0; 95% CI, 1.50-2.66; P less than .001), MSS (HR, 1.76; 95% CI, 1.22-2.53; P = .003), and DFS (HR, 1.62; 95% CI, 1.04-2.53; P = .036) on univariate analysis, and associations remained significant on multivariable analysis.
The hospital-based investigation evaluated peripheral blood samples collected from 1,042 non-Hispanic white patients with melanoma from 1997 to 2009.
Dr. Fang reported having no disclosures. Several of his coauthors reported financial ties to industry sources.
*Correction: An earlier version of this article used an incorrect unit of measurement for vitamin D deficiency.
Lower levels of vitamin D in patients with melanoma were associated with poorer survival, independent of effects from systemic inflammation indicated by simultaneous C-reactive protein (CRP) measures, according to researchers.
Multivariate analysis showed that vitamin D level was associated with overall survival (OS) (hazard ratio, 1.02 per unit decrease of vitamin D; 95% confidence interval, 1.01-1.04; P = .005), melanoma-specific survival (MSS) (HR, 1.02; 95% CI, 1.00-1.04; P = .048), and disease-free survival (DFS) (HR, 1.02; 95% CI, 1.00-1.04; P = .043). Patients with a vitamin D level less than 16 ng/mL* were 2.0 times more likely to die of all-cause disease than were patients with a higher level (95% CI, 1.50-2.66; P less than .001), investigators reported (J Clin Oncol. 2016 Mar 21. doi: 10.1200/JCO.2015.64.1357).
“Importantly, after adjustment for CRP, vitamin D remained an independent predictor of OS, MSS, and DFS. This suggests that, although blood levels of vitamin D and CRP are highly correlated with each other, each independently predicts clinical outcome in patients with melanoma,” wrote Dr. Shenying Fang of the University of Texas MD Anderson Cancer Center, Houston, and colleagues.
Although the role of the biomarkers in disease remains unclear and warrants further research, the authors added, “these data suggest that interventions to increase vitamin D or to reduce [systematic inflammatory response] and CRP could ultimately benefit patients with melanoma.”
Previous research has demonstrated an association between vitamin D deficiency and advanced melanoma stage, but investigations of vitamin D blood levels and melanoma risk have yielded inconsistent results. The researchers examined confounders, such as systemic inflammatory response (assessed by simultaneous CRP measurement), age, disease stage, and blood draw season to assess the relationship between vitamin D level and melanoma outcomes.
Decreased vitamin D was significantly associated with higher patient age, increased primary tumor thickness, ulcerated tumors, advanced-stage disease at blood draw, and increased CRP. Blood drawn in the fall/winter had lower vitamin D levels than that of spring/summer, reflecting average differences in sun exposure. These factors were included in the multivariable model.
A vitamin D level less than 20 ng/mL* is considered deficient, and deficient levels were significantly associated with poorer OS and DFS but not with poorer MSS. At levels below the optimal cutoff, determined by recursive partitioning, of 16 ng/mL*, patients had poorer OS (HR, 2.0; 95% CI, 1.50-2.66; P less than .001), MSS (HR, 1.76; 95% CI, 1.22-2.53; P = .003), and DFS (HR, 1.62; 95% CI, 1.04-2.53; P = .036) on univariate analysis, and associations remained significant on multivariable analysis.
The hospital-based investigation evaluated peripheral blood samples collected from 1,042 non-Hispanic white patients with melanoma from 1997 to 2009.
Dr. Fang reported having no disclosures. Several of his coauthors reported financial ties to industry sources.
*Correction: An earlier version of this article used an incorrect unit of measurement for vitamin D deficiency.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: After adjusting for C-reactive protein (CRP), lower vitamin D levels were independently associated with worse outcomes in melanoma patients.
Major finding: After adjusting for CRP and other confounders, vitamin D level was associated with overall survival (hazard ratio, 1.02 per unit decrease of vitamin D; 95% confidence interval, 1.01-1.04; P = .005), melanoma-specific survival (HR, 1.02; 95% CI, 1.00-1.04; P = .048), and disease-free survival (HR, 1.02; 95% CI, 1.00-1.04; P = .043).
Data sources: Peripheral blood samples from 1,042 non-Hispanic white patients with melanoma were collected from 1997 to 2009.
Disclosures: Dr. Fang reported having no disclosures. Several of his coauthors reported financial ties to industry sources.
Nicotinamide Prevents Actinic Keratoses, Basal Cell Carcinomas, and Squamous Cell Carcinomas

Chen et al (N Engl J Med. 2015;373:1618-1626) performed a multicenter, phase 3, double-blind, randomized, placebo-controlled trial. Results demonstrated that nicotinamide effectively decreased the rates of new nonmelanoma skin cancers (NMSCs) and actinic keratoses (AKs) in high-risk patients who had at least 2 histologically confirmed NMSCs in the last 5 years. In comparison to participants who received placebo, individuals who received nicotinamide 500 mg twice daily during the 12-month study (branded with a predictive acronym ONTRAC [oral nicotinamide to reduce actinic cancer]) had reduced rates of AKs of up to 20%, basal cell carcinomas of 20%, squamous cell carcinomas of 30%, and NMSCs of 23%. However, the effect of nicotinamide on NMSCs was not maintained at evaluation 6 months after discontinuation; the number of basal cell carcinomas was similar, and the number of squamous cell carcinomas was greater in participants who received nicotinamide in comparison to individuals who received placebo.
What’s the issue?
The risk for skin cancer is increased by UV radiation that damages DNA, suppresses cutaneous immunity, and inhibits DNA repair by depleting cellular adenosine triphosphate. Nicotinamide, an amide form of vitamin B3, has been demonstrated to not only reduce UV radiation–induced immunosuppression but also to prevent UV radiation–induced adenosine triphosphate depletion and glycolic blockade. Nicotinamide, which is classified as a food additive, also has neuroprotective and antioxidant functions and reduces pigmentation, wrinkles, and sebum production. Although oral nicotinamide has been demonstrated to reduce NMSCs and AKs, topical application has been shown to improve many skin conditions such as acne, atopic dermatitis, isoniazid-induced pellagra, and rosacea.
In contrast to nicotinic acid (niacin), nicotinamide is not associated with headaches, hypotension, flushing, itching, or vasodilatation. At high oral doses, side effects of nicotinamide that have been hypothesized or observed in animals, humans, or both have included the development of Parkinson disease, insulin sensitivity and diabetes mellitus, and liver toxicity. Although there are no reports in humans of growth retardation, teratogenicity, or oncogenicity, Rolfe (J Cosmet Dermatol. 2014;13:324-328) discussed that fetal blood levels of nicotinamide are greater than corresponding maternal blood levels because it is able to cross the placenta. However, according to Chen et al, no clinically significant between-group differences were found with respect to the number or types of adverse events that occurred in the placebo participants and the individuals who received 1000 mg daily of nicotinamide. Chen et al implied that there were additional benefits in the recipients of nicotinamide with regards to cognitive function and transepidermal water loss.
Perhaps all patients with a history of AKs, basal cell carcinomas, or squamous cell carcinomas should receive lifelong nicotinamide. Also, it might be reasonable to consider that all individuals older than 18 years who are not pregnant or breastfeeding with increased sun exposure but no history of AKs or NMSC add nicotinamide to their daily diets as a proactive measure for chemoprevention. Would you suggest nicotinamide to your patients?

Chen et al (N Engl J Med. 2015;373:1618-1626) performed a multicenter, phase 3, double-blind, randomized, placebo-controlled trial. Results demonstrated that nicotinamide effectively decreased the rates of new nonmelanoma skin cancers (NMSCs) and actinic keratoses (AKs) in high-risk patients who had at least 2 histologically confirmed NMSCs in the last 5 years. In comparison to participants who received placebo, individuals who received nicotinamide 500 mg twice daily during the 12-month study (branded with a predictive acronym ONTRAC [oral nicotinamide to reduce actinic cancer]) had reduced rates of AKs of up to 20%, basal cell carcinomas of 20%, squamous cell carcinomas of 30%, and NMSCs of 23%. However, the effect of nicotinamide on NMSCs was not maintained at evaluation 6 months after discontinuation; the number of basal cell carcinomas was similar, and the number of squamous cell carcinomas was greater in participants who received nicotinamide in comparison to individuals who received placebo.
What’s the issue?
The risk for skin cancer is increased by UV radiation that damages DNA, suppresses cutaneous immunity, and inhibits DNA repair by depleting cellular adenosine triphosphate. Nicotinamide, an amide form of vitamin B3, has been demonstrated to not only reduce UV radiation–induced immunosuppression but also to prevent UV radiation–induced adenosine triphosphate depletion and glycolic blockade. Nicotinamide, which is classified as a food additive, also has neuroprotective and antioxidant functions and reduces pigmentation, wrinkles, and sebum production. Although oral nicotinamide has been demonstrated to reduce NMSCs and AKs, topical application has been shown to improve many skin conditions such as acne, atopic dermatitis, isoniazid-induced pellagra, and rosacea.
In contrast to nicotinic acid (niacin), nicotinamide is not associated with headaches, hypotension, flushing, itching, or vasodilatation. At high oral doses, side effects of nicotinamide that have been hypothesized or observed in animals, humans, or both have included the development of Parkinson disease, insulin sensitivity and diabetes mellitus, and liver toxicity. Although there are no reports in humans of growth retardation, teratogenicity, or oncogenicity, Rolfe (J Cosmet Dermatol. 2014;13:324-328) discussed that fetal blood levels of nicotinamide are greater than corresponding maternal blood levels because it is able to cross the placenta. However, according to Chen et al, no clinically significant between-group differences were found with respect to the number or types of adverse events that occurred in the placebo participants and the individuals who received 1000 mg daily of nicotinamide. Chen et al implied that there were additional benefits in the recipients of nicotinamide with regards to cognitive function and transepidermal water loss.
Perhaps all patients with a history of AKs, basal cell carcinomas, or squamous cell carcinomas should receive lifelong nicotinamide. Also, it might be reasonable to consider that all individuals older than 18 years who are not pregnant or breastfeeding with increased sun exposure but no history of AKs or NMSC add nicotinamide to their daily diets as a proactive measure for chemoprevention. Would you suggest nicotinamide to your patients?

Chen et al (N Engl J Med. 2015;373:1618-1626) performed a multicenter, phase 3, double-blind, randomized, placebo-controlled trial. Results demonstrated that nicotinamide effectively decreased the rates of new nonmelanoma skin cancers (NMSCs) and actinic keratoses (AKs) in high-risk patients who had at least 2 histologically confirmed NMSCs in the last 5 years. In comparison to participants who received placebo, individuals who received nicotinamide 500 mg twice daily during the 12-month study (branded with a predictive acronym ONTRAC [oral nicotinamide to reduce actinic cancer]) had reduced rates of AKs of up to 20%, basal cell carcinomas of 20%, squamous cell carcinomas of 30%, and NMSCs of 23%. However, the effect of nicotinamide on NMSCs was not maintained at evaluation 6 months after discontinuation; the number of basal cell carcinomas was similar, and the number of squamous cell carcinomas was greater in participants who received nicotinamide in comparison to individuals who received placebo.
What’s the issue?
The risk for skin cancer is increased by UV radiation that damages DNA, suppresses cutaneous immunity, and inhibits DNA repair by depleting cellular adenosine triphosphate. Nicotinamide, an amide form of vitamin B3, has been demonstrated to not only reduce UV radiation–induced immunosuppression but also to prevent UV radiation–induced adenosine triphosphate depletion and glycolic blockade. Nicotinamide, which is classified as a food additive, also has neuroprotective and antioxidant functions and reduces pigmentation, wrinkles, and sebum production. Although oral nicotinamide has been demonstrated to reduce NMSCs and AKs, topical application has been shown to improve many skin conditions such as acne, atopic dermatitis, isoniazid-induced pellagra, and rosacea.
In contrast to nicotinic acid (niacin), nicotinamide is not associated with headaches, hypotension, flushing, itching, or vasodilatation. At high oral doses, side effects of nicotinamide that have been hypothesized or observed in animals, humans, or both have included the development of Parkinson disease, insulin sensitivity and diabetes mellitus, and liver toxicity. Although there are no reports in humans of growth retardation, teratogenicity, or oncogenicity, Rolfe (J Cosmet Dermatol. 2014;13:324-328) discussed that fetal blood levels of nicotinamide are greater than corresponding maternal blood levels because it is able to cross the placenta. However, according to Chen et al, no clinically significant between-group differences were found with respect to the number or types of adverse events that occurred in the placebo participants and the individuals who received 1000 mg daily of nicotinamide. Chen et al implied that there were additional benefits in the recipients of nicotinamide with regards to cognitive function and transepidermal water loss.
Perhaps all patients with a history of AKs, basal cell carcinomas, or squamous cell carcinomas should receive lifelong nicotinamide. Also, it might be reasonable to consider that all individuals older than 18 years who are not pregnant or breastfeeding with increased sun exposure but no history of AKs or NMSC add nicotinamide to their daily diets as a proactive measure for chemoprevention. Would you suggest nicotinamide to your patients?
Poor Sun Protection Practices in Hispanic Patients Increases Risk for Skin Cancer
The most common type of skin cancer in Hispanic patients is basal cell carcinoma, according to Maritza Perez, MD, Associate Clinical Professor of Dermatology at the Icahn School of Medicine at Mount Sinai, New York, New York. Dr. Perez reviewed skin cancer in Hispanics as part of the “Skin Issues in Latino Patients” forum at the 74th Annual Meeting of the American Academy of Dermatology (AAD)(March 4-8, 2016) in Washington, DC. The head and neck region is the most common location of BCC in Hispanics, and it is more common among Hispanic males. Squamous cell carcinomas are the second most common, and melanomas are third most common.
The mortality rate from squamous cell carcinoma is higher in Hispanic patients compared to white patients. Dr. Perez indicated that UV light exposure is a predisposing factor for these types of skin cancer, and poor sun protection practices in this population are to blame.
“Patients [in this population] do not perceive that they are prone to skin cancer,” said Dr. Perez. This erroneous perception has led to delayed diagnosis and treatment of patients in this group, which has caused increased mortality and decreased survival.
Marta Rendon, MD, director of the session at the AAD and Medical Director of The Rendon Center for Dermatology & Aesthetic Medicine, Boca Raton, Florida, noted that Latinos are the fastest growing minority population in the United States and it is estimated that they will be approximately 30% of the population by the year 2050. As a result, dermatologists must emphasize sun protection, avoidance of midday sun, and avoidance of UV radiation.
In his Cutis practical pearls, “Patient Compliance With Photoprotection,” Vincent A. DeLeo, MD, provided tips on sunscreen use. “Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history . . . and lifestyle history, which should include location of residence as well as occupation and recreational pursuits,” he said. “These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer.”
Encourage routine use of a sunscreen with a sun protection factor of 30 or higher that carries a “broad spectrum” label. “There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma,” Dr. DeLeo reported.
The most common type of skin cancer in Hispanic patients is basal cell carcinoma, according to Maritza Perez, MD, Associate Clinical Professor of Dermatology at the Icahn School of Medicine at Mount Sinai, New York, New York. Dr. Perez reviewed skin cancer in Hispanics as part of the “Skin Issues in Latino Patients” forum at the 74th Annual Meeting of the American Academy of Dermatology (AAD)(March 4-8, 2016) in Washington, DC. The head and neck region is the most common location of BCC in Hispanics, and it is more common among Hispanic males. Squamous cell carcinomas are the second most common, and melanomas are third most common.
The mortality rate from squamous cell carcinoma is higher in Hispanic patients compared to white patients. Dr. Perez indicated that UV light exposure is a predisposing factor for these types of skin cancer, and poor sun protection practices in this population are to blame.
“Patients [in this population] do not perceive that they are prone to skin cancer,” said Dr. Perez. This erroneous perception has led to delayed diagnosis and treatment of patients in this group, which has caused increased mortality and decreased survival.
Marta Rendon, MD, director of the session at the AAD and Medical Director of The Rendon Center for Dermatology & Aesthetic Medicine, Boca Raton, Florida, noted that Latinos are the fastest growing minority population in the United States and it is estimated that they will be approximately 30% of the population by the year 2050. As a result, dermatologists must emphasize sun protection, avoidance of midday sun, and avoidance of UV radiation.
In his Cutis practical pearls, “Patient Compliance With Photoprotection,” Vincent A. DeLeo, MD, provided tips on sunscreen use. “Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history . . . and lifestyle history, which should include location of residence as well as occupation and recreational pursuits,” he said. “These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer.”
Encourage routine use of a sunscreen with a sun protection factor of 30 or higher that carries a “broad spectrum” label. “There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma,” Dr. DeLeo reported.
The most common type of skin cancer in Hispanic patients is basal cell carcinoma, according to Maritza Perez, MD, Associate Clinical Professor of Dermatology at the Icahn School of Medicine at Mount Sinai, New York, New York. Dr. Perez reviewed skin cancer in Hispanics as part of the “Skin Issues in Latino Patients” forum at the 74th Annual Meeting of the American Academy of Dermatology (AAD)(March 4-8, 2016) in Washington, DC. The head and neck region is the most common location of BCC in Hispanics, and it is more common among Hispanic males. Squamous cell carcinomas are the second most common, and melanomas are third most common.
The mortality rate from squamous cell carcinoma is higher in Hispanic patients compared to white patients. Dr. Perez indicated that UV light exposure is a predisposing factor for these types of skin cancer, and poor sun protection practices in this population are to blame.
“Patients [in this population] do not perceive that they are prone to skin cancer,” said Dr. Perez. This erroneous perception has led to delayed diagnosis and treatment of patients in this group, which has caused increased mortality and decreased survival.
Marta Rendon, MD, director of the session at the AAD and Medical Director of The Rendon Center for Dermatology & Aesthetic Medicine, Boca Raton, Florida, noted that Latinos are the fastest growing minority population in the United States and it is estimated that they will be approximately 30% of the population by the year 2050. As a result, dermatologists must emphasize sun protection, avoidance of midday sun, and avoidance of UV radiation.
In his Cutis practical pearls, “Patient Compliance With Photoprotection,” Vincent A. DeLeo, MD, provided tips on sunscreen use. “Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history . . . and lifestyle history, which should include location of residence as well as occupation and recreational pursuits,” he said. “These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer.”
Encourage routine use of a sunscreen with a sun protection factor of 30 or higher that carries a “broad spectrum” label. “There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma,” Dr. DeLeo reported.
Itraconazole targets basal cell carcinoma
WAIKOLOA, HAWAII – The familiar oral triazole antifungal agent itraconazole (Sporanox) is under active investigation for an unexpected use: as adjunctive therapy in patients with locally advanced or metastatic basal cell carcinoma.
“The promise of this drug is that the use of itraconazole with vismodegib or sonidegib may actually enhance the effectiveness of those drugs and also reduce the frequency of grade 2 toxicities by perhaps allowing a lower dose of vismodegib or sonidegib,” Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Be looking for this drug that we use to treat toenail fungus as a potential drug for locally advanced or metastatic basal cell carcinoma,” advised Dr. Swanson, a dermatologist at the Mayo Clinic in Scottsdale, Ariz.
Vismodegib (Erivedge) has been a game changer for patients with inoperable locally advanced or metastatic breast cancer. “The response to this drug was amazing,” Dr. Swanson said of the landmark study which led to its approval in 2012 (N Engl J Med. 2012; 366[23]:2171-9).
Sonidegib (Odomzo), which like vismodegib inhibits the essential Hedgehog signaling pathway component known as Smoothened, was approved by the Food and Drug Administration in 2015 as the second oral drug in this novel class.
While the clinical benefits of these two drugs in patients with the most horrific basal cell carcinomas are extremely impressive, vismodegib and sonidegib have two major drawbacks: The tumors eventually develop resistance and commence growing again, and onerous grade 2 side effects requiring dose reduction are extremely common. The most frequent of these limiting side effects are a disturbed sense of taste, muscle spasms, alopecia, and weight loss.
The hope is that itraconazole may be of help with both issues, according to Dr. Swanson. It turns out that the antifungal agent is also an inhibitor of the Hedgehog pathway, and via a different mechanism than that of vismodegib and sonidegib.
He pointed to an international open-label exploratory phase II study led by Dr. Jean Y. Tang of Stanford (Calif.) University. The investigators treated 19 patients with a total of 90 basal cell carcinomas with oral itraconazole at 200 mg twice a day for 1 month or 100 mg twice a day for an average of 2.3 months.
The treatment reduced Hedgehog signaling pathway activity by 65%, Ki67 tumor cell proliferation by 45%, and tumor area by 24% (J Clin Oncol. 2014 Mar 10;32[8]:745-51).
These results aren’t as dramatic as what’s achieved using vismodegib or sonidegib. As stand-alone therapy, itraconazole doesn’t compare with those agents. However, the hope is that when itraconazole is prescribed in conjunction with vismodegib or sonidegib it will permit the latter drugs to be used at lower doses with no drop-off in efficacy, which would mean less grade 2 toxicity. Moreover, since itraconazole inhibits Hedgehog signaling through a mechanism that is different from that of the more potent agents, combination therapy might delay onset of tumor resistance, Dr. Swanson explained.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The familiar oral triazole antifungal agent itraconazole (Sporanox) is under active investigation for an unexpected use: as adjunctive therapy in patients with locally advanced or metastatic basal cell carcinoma.
“The promise of this drug is that the use of itraconazole with vismodegib or sonidegib may actually enhance the effectiveness of those drugs and also reduce the frequency of grade 2 toxicities by perhaps allowing a lower dose of vismodegib or sonidegib,” Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Be looking for this drug that we use to treat toenail fungus as a potential drug for locally advanced or metastatic basal cell carcinoma,” advised Dr. Swanson, a dermatologist at the Mayo Clinic in Scottsdale, Ariz.
Vismodegib (Erivedge) has been a game changer for patients with inoperable locally advanced or metastatic breast cancer. “The response to this drug was amazing,” Dr. Swanson said of the landmark study which led to its approval in 2012 (N Engl J Med. 2012; 366[23]:2171-9).
Sonidegib (Odomzo), which like vismodegib inhibits the essential Hedgehog signaling pathway component known as Smoothened, was approved by the Food and Drug Administration in 2015 as the second oral drug in this novel class.
While the clinical benefits of these two drugs in patients with the most horrific basal cell carcinomas are extremely impressive, vismodegib and sonidegib have two major drawbacks: The tumors eventually develop resistance and commence growing again, and onerous grade 2 side effects requiring dose reduction are extremely common. The most frequent of these limiting side effects are a disturbed sense of taste, muscle spasms, alopecia, and weight loss.
The hope is that itraconazole may be of help with both issues, according to Dr. Swanson. It turns out that the antifungal agent is also an inhibitor of the Hedgehog pathway, and via a different mechanism than that of vismodegib and sonidegib.
He pointed to an international open-label exploratory phase II study led by Dr. Jean Y. Tang of Stanford (Calif.) University. The investigators treated 19 patients with a total of 90 basal cell carcinomas with oral itraconazole at 200 mg twice a day for 1 month or 100 mg twice a day for an average of 2.3 months.
The treatment reduced Hedgehog signaling pathway activity by 65%, Ki67 tumor cell proliferation by 45%, and tumor area by 24% (J Clin Oncol. 2014 Mar 10;32[8]:745-51).
These results aren’t as dramatic as what’s achieved using vismodegib or sonidegib. As stand-alone therapy, itraconazole doesn’t compare with those agents. However, the hope is that when itraconazole is prescribed in conjunction with vismodegib or sonidegib it will permit the latter drugs to be used at lower doses with no drop-off in efficacy, which would mean less grade 2 toxicity. Moreover, since itraconazole inhibits Hedgehog signaling through a mechanism that is different from that of the more potent agents, combination therapy might delay onset of tumor resistance, Dr. Swanson explained.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The familiar oral triazole antifungal agent itraconazole (Sporanox) is under active investigation for an unexpected use: as adjunctive therapy in patients with locally advanced or metastatic basal cell carcinoma.
“The promise of this drug is that the use of itraconazole with vismodegib or sonidegib may actually enhance the effectiveness of those drugs and also reduce the frequency of grade 2 toxicities by perhaps allowing a lower dose of vismodegib or sonidegib,” Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Be looking for this drug that we use to treat toenail fungus as a potential drug for locally advanced or metastatic basal cell carcinoma,” advised Dr. Swanson, a dermatologist at the Mayo Clinic in Scottsdale, Ariz.
Vismodegib (Erivedge) has been a game changer for patients with inoperable locally advanced or metastatic breast cancer. “The response to this drug was amazing,” Dr. Swanson said of the landmark study which led to its approval in 2012 (N Engl J Med. 2012; 366[23]:2171-9).
Sonidegib (Odomzo), which like vismodegib inhibits the essential Hedgehog signaling pathway component known as Smoothened, was approved by the Food and Drug Administration in 2015 as the second oral drug in this novel class.
While the clinical benefits of these two drugs in patients with the most horrific basal cell carcinomas are extremely impressive, vismodegib and sonidegib have two major drawbacks: The tumors eventually develop resistance and commence growing again, and onerous grade 2 side effects requiring dose reduction are extremely common. The most frequent of these limiting side effects are a disturbed sense of taste, muscle spasms, alopecia, and weight loss.
The hope is that itraconazole may be of help with both issues, according to Dr. Swanson. It turns out that the antifungal agent is also an inhibitor of the Hedgehog pathway, and via a different mechanism than that of vismodegib and sonidegib.
He pointed to an international open-label exploratory phase II study led by Dr. Jean Y. Tang of Stanford (Calif.) University. The investigators treated 19 patients with a total of 90 basal cell carcinomas with oral itraconazole at 200 mg twice a day for 1 month or 100 mg twice a day for an average of 2.3 months.
The treatment reduced Hedgehog signaling pathway activity by 65%, Ki67 tumor cell proliferation by 45%, and tumor area by 24% (J Clin Oncol. 2014 Mar 10;32[8]:745-51).
These results aren’t as dramatic as what’s achieved using vismodegib or sonidegib. As stand-alone therapy, itraconazole doesn’t compare with those agents. However, the hope is that when itraconazole is prescribed in conjunction with vismodegib or sonidegib it will permit the latter drugs to be used at lower doses with no drop-off in efficacy, which would mean less grade 2 toxicity. Moreover, since itraconazole inhibits Hedgehog signaling through a mechanism that is different from that of the more potent agents, combination therapy might delay onset of tumor resistance, Dr. Swanson explained.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
RECIST underestimates benefit of pembrolizumab in subset of melanoma patients
Certain patients with advanced melanoma treated with pembrolizumab had unique response patterns, such as early or delayed tumor pseudoprogression, and Response Evaluation Criteria in Solid Tumors (RECIST) criteria might underestimate the immunotherapy benefit in this subset.
Among 592 patients who survived 12 weeks or more, 84 (14%) had progressive disease per RECIST v1.1 but nonprogressive disease per immune-related response criteria (irRC). Overall survival (OS) was longer in this group compared with the 177 patients who had progressive disease per both RECIST v1.1 and irRC: median OS was 22.5 months (95% CI, 16.5 to not yet reached) vs. 8.4 months (95% CI, 6.6 to 9.9), investigators reported (J Clin Oncol. 2016 March 7. doi: 10.1200/JCO.2015.64.0391).
“RECIST v1.1 might underestimate the benefit of pembrolizumab in approximately 15% of patients. These data suggest that patients may benefit from receiving treatment beyond initial evidence of radiographic progression and thus support the use of modified response criteria on the basis of immune-related response patterns. Furthermore, clinicians aware to these criteria might be able to avoid otherwise premature termination of potentially effective treatment,” wrote Dr. F. Stephen Hodi Jr., director of immuno-oncology at Dana Farber Cancer Institute, Boston, and colleagues.
Two-year OS rates for patients with nonprogressive disease per both criteria, progressive disease per RECIST v1.1 and nonprogressive disease per irRC, and progressive disease per both criteria were 77.6%, 37.5%, and 17.3%, respectively.
Conventional response criteria such as RECIST might underestimate benefit of immune checkpoint blockade, such as ipilimumab and pembrolizumab, because objective response and prolonged disease stabilization can occur after an initial increase in tumor burden or new lesions. To better characterize atypical response patterns observed in trials of ipilimumab in melanoma, irRC were developed, where the initial evidence of disease progression is handled differently. The current study retrospectively examined responses from KEYNOTE-001 melanoma expansion cohorts, which enrolled 655 patients with advanced melanoma; 327 had 28 weeks or more of imaging follow up and were eligible for atypical response analysis.
In total, 24 of 327 patients (7.3%) had atypical responses, including 15 (4.5%) with early pseudoprogression and nine (2.8%) with delayed pseudoprogression. Patterns of atypical response included regression of tumor burden and stable disease despite the development of new lesions, and initial increases in the size of target lesions followed by decreases without evidence of new lesions.
The irRC uses bidimensional tumor measurements, which introduce greater variability than unidimensional measurements. In addition, the investigators note that irRC may not fully capture all patterns of clinical response to immunotherapies.
“Given the rapid development of effective immuno-oncology agents in multiple cancers, there is a growing effort to develop new standard response criteria for patients treated with immunotherapy to provide for robust clinical end points in evaluating these new treatments,” the investigators wrote.
Certain patients with advanced melanoma treated with pembrolizumab had unique response patterns, such as early or delayed tumor pseudoprogression, and Response Evaluation Criteria in Solid Tumors (RECIST) criteria might underestimate the immunotherapy benefit in this subset.
Among 592 patients who survived 12 weeks or more, 84 (14%) had progressive disease per RECIST v1.1 but nonprogressive disease per immune-related response criteria (irRC). Overall survival (OS) was longer in this group compared with the 177 patients who had progressive disease per both RECIST v1.1 and irRC: median OS was 22.5 months (95% CI, 16.5 to not yet reached) vs. 8.4 months (95% CI, 6.6 to 9.9), investigators reported (J Clin Oncol. 2016 March 7. doi: 10.1200/JCO.2015.64.0391).
“RECIST v1.1 might underestimate the benefit of pembrolizumab in approximately 15% of patients. These data suggest that patients may benefit from receiving treatment beyond initial evidence of radiographic progression and thus support the use of modified response criteria on the basis of immune-related response patterns. Furthermore, clinicians aware to these criteria might be able to avoid otherwise premature termination of potentially effective treatment,” wrote Dr. F. Stephen Hodi Jr., director of immuno-oncology at Dana Farber Cancer Institute, Boston, and colleagues.
Two-year OS rates for patients with nonprogressive disease per both criteria, progressive disease per RECIST v1.1 and nonprogressive disease per irRC, and progressive disease per both criteria were 77.6%, 37.5%, and 17.3%, respectively.
Conventional response criteria such as RECIST might underestimate benefit of immune checkpoint blockade, such as ipilimumab and pembrolizumab, because objective response and prolonged disease stabilization can occur after an initial increase in tumor burden or new lesions. To better characterize atypical response patterns observed in trials of ipilimumab in melanoma, irRC were developed, where the initial evidence of disease progression is handled differently. The current study retrospectively examined responses from KEYNOTE-001 melanoma expansion cohorts, which enrolled 655 patients with advanced melanoma; 327 had 28 weeks or more of imaging follow up and were eligible for atypical response analysis.
In total, 24 of 327 patients (7.3%) had atypical responses, including 15 (4.5%) with early pseudoprogression and nine (2.8%) with delayed pseudoprogression. Patterns of atypical response included regression of tumor burden and stable disease despite the development of new lesions, and initial increases in the size of target lesions followed by decreases without evidence of new lesions.
The irRC uses bidimensional tumor measurements, which introduce greater variability than unidimensional measurements. In addition, the investigators note that irRC may not fully capture all patterns of clinical response to immunotherapies.
“Given the rapid development of effective immuno-oncology agents in multiple cancers, there is a growing effort to develop new standard response criteria for patients treated with immunotherapy to provide for robust clinical end points in evaluating these new treatments,” the investigators wrote.
Certain patients with advanced melanoma treated with pembrolizumab had unique response patterns, such as early or delayed tumor pseudoprogression, and Response Evaluation Criteria in Solid Tumors (RECIST) criteria might underestimate the immunotherapy benefit in this subset.
Among 592 patients who survived 12 weeks or more, 84 (14%) had progressive disease per RECIST v1.1 but nonprogressive disease per immune-related response criteria (irRC). Overall survival (OS) was longer in this group compared with the 177 patients who had progressive disease per both RECIST v1.1 and irRC: median OS was 22.5 months (95% CI, 16.5 to not yet reached) vs. 8.4 months (95% CI, 6.6 to 9.9), investigators reported (J Clin Oncol. 2016 March 7. doi: 10.1200/JCO.2015.64.0391).
“RECIST v1.1 might underestimate the benefit of pembrolizumab in approximately 15% of patients. These data suggest that patients may benefit from receiving treatment beyond initial evidence of radiographic progression and thus support the use of modified response criteria on the basis of immune-related response patterns. Furthermore, clinicians aware to these criteria might be able to avoid otherwise premature termination of potentially effective treatment,” wrote Dr. F. Stephen Hodi Jr., director of immuno-oncology at Dana Farber Cancer Institute, Boston, and colleagues.
Two-year OS rates for patients with nonprogressive disease per both criteria, progressive disease per RECIST v1.1 and nonprogressive disease per irRC, and progressive disease per both criteria were 77.6%, 37.5%, and 17.3%, respectively.
Conventional response criteria such as RECIST might underestimate benefit of immune checkpoint blockade, such as ipilimumab and pembrolizumab, because objective response and prolonged disease stabilization can occur after an initial increase in tumor burden or new lesions. To better characterize atypical response patterns observed in trials of ipilimumab in melanoma, irRC were developed, where the initial evidence of disease progression is handled differently. The current study retrospectively examined responses from KEYNOTE-001 melanoma expansion cohorts, which enrolled 655 patients with advanced melanoma; 327 had 28 weeks or more of imaging follow up and were eligible for atypical response analysis.
In total, 24 of 327 patients (7.3%) had atypical responses, including 15 (4.5%) with early pseudoprogression and nine (2.8%) with delayed pseudoprogression. Patterns of atypical response included regression of tumor burden and stable disease despite the development of new lesions, and initial increases in the size of target lesions followed by decreases without evidence of new lesions.
The irRC uses bidimensional tumor measurements, which introduce greater variability than unidimensional measurements. In addition, the investigators note that irRC may not fully capture all patterns of clinical response to immunotherapies.
“Given the rapid development of effective immuno-oncology agents in multiple cancers, there is a growing effort to develop new standard response criteria for patients treated with immunotherapy to provide for robust clinical end points in evaluating these new treatments,” the investigators wrote.
Key clinical point: Approximately 15% of patients with advanced melanoma treated with pembrolizumab had unique response patterns, and RECIST criteria may underestimate the immunotherapy benefit in this subset.
Major finding: Among 592 patients, 84 (14%) had progressive disease per RECIST v1.1 but nonprogressive disease per immune-related response criteria (irRC); overall survival was longer in this group compared with patients who had progressive disease per both RECIST v1.1 and irRC (median OS 22.5 vs. 8.4 months, respectively).
Data sources: The KEYNOTE-001 expansion cohorts included 655 patients with advanced melanoma; 327 had 28 weeks or more of imaging follow up for atypical response analysis.
Disclosures: Dr. Hodi reported financial ties to Merck, Novartis, Bristol-Myers Squibb, Genentech, and Dana-Farber Cancer Institute (intellectual property). Several of his coauthors reported financial ties to industry sources.
Children who have stem cell transplants need skin exams, sun protection
WASHINGTON – Children who have had a hematopoietic stem cell transplant (HSCT) have an increased risk of benign and atypical nevi, Dr. Johanna Sheu reported at the annual meeting of the American Academy of Dermatology.
These patients need to have routine skin exams and be educated about sun protection needs, she said. Based on her study, these needs are not routinely met.
At least 1 year after undergoing HSCT at Boston Children’s Hospital, 85 posttransplant patients had significantly more nevi and more atypical nevi than did 85 healthy controls who were matched by age, gender, and Fitzpatrick skin type. In addition, 41% of the transplant recipients had at least one actinic keratosis, a basal or squamous cell carcinoma, or a solar lentigo; 11% had at least one nevus spilus.
Moreover, “sun protection … and dermatology follow-up was poor” among the transplant recipients, said Dr. Sheu, of MassGeneral Hospital for Children, Boston. About 40% of the transplant recipients reported having a sunburn since their transplant, only 15% reported daily use of sunscreen, and 53% said they did not recall being told that sunburn could trigger graft-versus-host disease (GVHD).
About one-third of the patients had never seen a dermatologist; of those who had, two-thirds had only seen the dermatologist once, Dr. Sheu reported.
Late skin effects of HSCT are not as well described in children as they are in adults, she said. In adults, late skin effects include vitiligo, psoriasis, nonmelanoma skin cancers, and an increased nevi count.
The children in the study had undergone an HSCT between 1998 and 2013, at a median age of about 7 years (range was 1 month to 19 years). At the time of their skin exams, their mean age was 14 years, and they had been followed for a median of almost 4 years. Nevi were counted on the forearms, backs, legs, palms, and soles.
The median nevi count was 44 nevi, significantly more than the level seen in control subjects. Transplant recipients also had significantly more nevi in sun-exposed areas of the body, as well as on the palms and soles. Transplant recipients were more likely to have atypical nevi and to have nevi greater than 5 mm in diameter.
In addition to fair skin, factors associated with an increase in the overall nevi count included being older than age 10 at the time of the transplant and having total body irradiation, pretransplant chemotherapy, and myeloablative conditioning. Having had a sunburn since the transplant, reported by 40%, was also a risk factor.
Chronic GVHD and chronic GVHD of the skin were associated with the presence of atypical nevi; acute GVHD, the duration of immune suppression, and the use of topical steroids or calcineurin inhibitors were not associated with increased risk of atypical nevi.
She and her coinvestigators are currently analyzing the pathogenesis of these late effects in this population, and autoimmune skin conditions – vitiligo and alopecia – in 25% of the transplant recipients in the study.
In 2013, 1,100 children under aged 16 years in the United States underwent a bone marrow transplant, she noted.
Dr. Sheu had no disclosures.
WASHINGTON – Children who have had a hematopoietic stem cell transplant (HSCT) have an increased risk of benign and atypical nevi, Dr. Johanna Sheu reported at the annual meeting of the American Academy of Dermatology.
These patients need to have routine skin exams and be educated about sun protection needs, she said. Based on her study, these needs are not routinely met.
At least 1 year after undergoing HSCT at Boston Children’s Hospital, 85 posttransplant patients had significantly more nevi and more atypical nevi than did 85 healthy controls who were matched by age, gender, and Fitzpatrick skin type. In addition, 41% of the transplant recipients had at least one actinic keratosis, a basal or squamous cell carcinoma, or a solar lentigo; 11% had at least one nevus spilus.
Moreover, “sun protection … and dermatology follow-up was poor” among the transplant recipients, said Dr. Sheu, of MassGeneral Hospital for Children, Boston. About 40% of the transplant recipients reported having a sunburn since their transplant, only 15% reported daily use of sunscreen, and 53% said they did not recall being told that sunburn could trigger graft-versus-host disease (GVHD).
About one-third of the patients had never seen a dermatologist; of those who had, two-thirds had only seen the dermatologist once, Dr. Sheu reported.
Late skin effects of HSCT are not as well described in children as they are in adults, she said. In adults, late skin effects include vitiligo, psoriasis, nonmelanoma skin cancers, and an increased nevi count.
The children in the study had undergone an HSCT between 1998 and 2013, at a median age of about 7 years (range was 1 month to 19 years). At the time of their skin exams, their mean age was 14 years, and they had been followed for a median of almost 4 years. Nevi were counted on the forearms, backs, legs, palms, and soles.
The median nevi count was 44 nevi, significantly more than the level seen in control subjects. Transplant recipients also had significantly more nevi in sun-exposed areas of the body, as well as on the palms and soles. Transplant recipients were more likely to have atypical nevi and to have nevi greater than 5 mm in diameter.
In addition to fair skin, factors associated with an increase in the overall nevi count included being older than age 10 at the time of the transplant and having total body irradiation, pretransplant chemotherapy, and myeloablative conditioning. Having had a sunburn since the transplant, reported by 40%, was also a risk factor.
Chronic GVHD and chronic GVHD of the skin were associated with the presence of atypical nevi; acute GVHD, the duration of immune suppression, and the use of topical steroids or calcineurin inhibitors were not associated with increased risk of atypical nevi.
She and her coinvestigators are currently analyzing the pathogenesis of these late effects in this population, and autoimmune skin conditions – vitiligo and alopecia – in 25% of the transplant recipients in the study.
In 2013, 1,100 children under aged 16 years in the United States underwent a bone marrow transplant, she noted.
Dr. Sheu had no disclosures.
WASHINGTON – Children who have had a hematopoietic stem cell transplant (HSCT) have an increased risk of benign and atypical nevi, Dr. Johanna Sheu reported at the annual meeting of the American Academy of Dermatology.
These patients need to have routine skin exams and be educated about sun protection needs, she said. Based on her study, these needs are not routinely met.
At least 1 year after undergoing HSCT at Boston Children’s Hospital, 85 posttransplant patients had significantly more nevi and more atypical nevi than did 85 healthy controls who were matched by age, gender, and Fitzpatrick skin type. In addition, 41% of the transplant recipients had at least one actinic keratosis, a basal or squamous cell carcinoma, or a solar lentigo; 11% had at least one nevus spilus.
Moreover, “sun protection … and dermatology follow-up was poor” among the transplant recipients, said Dr. Sheu, of MassGeneral Hospital for Children, Boston. About 40% of the transplant recipients reported having a sunburn since their transplant, only 15% reported daily use of sunscreen, and 53% said they did not recall being told that sunburn could trigger graft-versus-host disease (GVHD).
About one-third of the patients had never seen a dermatologist; of those who had, two-thirds had only seen the dermatologist once, Dr. Sheu reported.
Late skin effects of HSCT are not as well described in children as they are in adults, she said. In adults, late skin effects include vitiligo, psoriasis, nonmelanoma skin cancers, and an increased nevi count.
The children in the study had undergone an HSCT between 1998 and 2013, at a median age of about 7 years (range was 1 month to 19 years). At the time of their skin exams, their mean age was 14 years, and they had been followed for a median of almost 4 years. Nevi were counted on the forearms, backs, legs, palms, and soles.
The median nevi count was 44 nevi, significantly more than the level seen in control subjects. Transplant recipients also had significantly more nevi in sun-exposed areas of the body, as well as on the palms and soles. Transplant recipients were more likely to have atypical nevi and to have nevi greater than 5 mm in diameter.
In addition to fair skin, factors associated with an increase in the overall nevi count included being older than age 10 at the time of the transplant and having total body irradiation, pretransplant chemotherapy, and myeloablative conditioning. Having had a sunburn since the transplant, reported by 40%, was also a risk factor.
Chronic GVHD and chronic GVHD of the skin were associated with the presence of atypical nevi; acute GVHD, the duration of immune suppression, and the use of topical steroids or calcineurin inhibitors were not associated with increased risk of atypical nevi.
She and her coinvestigators are currently analyzing the pathogenesis of these late effects in this population, and autoimmune skin conditions – vitiligo and alopecia – in 25% of the transplant recipients in the study.
In 2013, 1,100 children under aged 16 years in the United States underwent a bone marrow transplant, she noted.
Dr. Sheu had no disclosures.
AT AAD 16
Key clinical point: Children who have had a hematopoietic stem cell transplant need to have routine skin exams and be educated about sun protection needs.
Major finding: 41% of the transplant recipients had at least one actinic keratosis, a basal or squamous cell carcinoma, or a solar lentigo; 11% had at least one nevus spilus.
Data source: A single-center study of 85 posttransplant patients and 85 healthy controls who were matched by age, gender, and Fitzpatrick skin type.
Disclosures: The study was not sponsored and Dr. Sheu had no disclosures.
Ibrutinib bodes well for relapsed mantle-cell lymphoma
Progression-free survival was significantly better when patients with relapsed or refractory mantle-cell lymphoma were treated with oral ibrutinib than with intravenous temsirolimus, based on results from 280 patients in an international, randomized, open-label phase III trial.
Study subjects had undergone one or more previous rituximab-containing chemotherapy regimens to receive intravenous temsirolimus or oral ibrutinib at a daily dose of 560 mg.
Compared with temsirolimus, ibrutinib resulted in a 57% reduction in the risk of disease progression or death at a median follow-up of 20 months. Median progression-free survival – the trial’s primary endpoint – was 14.6 months for the ibrutinib group and 6.2 months for the temsirolimus group.
Ibrutinib was also better tolerated, with 68% of patients having grade 3 or higher treatment-emergent adverse events as compared to 87% of patients in the temsirolimus group, despite a median 4-fold longer treatment duration for the ibrutinib group than the temsirolimus group. Additionally, 6% of patients discontinued ibrutinib because of adverse events versus 26% in the temsirolimus group, reported Dr. Martin Dreyling of Klinikum der Universität in Munich, Germany, and his associates.
Based on results of the Functional Assessment of Cancer Therapy-Lymphoma (FACT-Lym) questionnaire, ibrutinib was associated with greater and more rapid improvements, and also with less worsening in lymphoma symptoms, as measured by the lymphoma subscale of the FACT-Lym (Lancet. 2016;387:770-78).
Ibrutinib, a first-in-class oral inhibitor of Bruton’s tyrosine kinase, is approved in the United States and the European Union at a dose of 560 mg per day for patients with mantle cell lymphoma who have received at least one previous line of therapy.
The mammalian target of rapamycin (mTOR) inhibitor temsirolimus is approved in the European Union for relapsed or refractory mantle-cell lymphoma, but does not have FDA approval for this indication.
The study, funded by Janssen, is ongoing. Future research, the investigators say, should examine ibrutinib-based combination approaches for patients with relapsed or refractory mantle-cell lymphoma and in front-line therapy.
Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study. Several other authors reported grants from Janssen during the study and financial ties to the company.
The findings from this phase III trial clearly establish ibrutinib as a new standard for treatment of relapsed mantle-cell lymphoma. Within the next 2 years, many expect the agent will find its way into the frontline setting for treatment of mantle cell lymphoma in combination with standard chemotherapy, based on results of another already completed phase III trial (the SHINE trial).
Despite this remarkable progress, however, mantle-cell lymphoma remains incurable. Roughly 30%-40% of people with the disease will not respond to ibrutinib, and even among responders relapse seems inevitable.
Mantle-cell lymphoma has been a model for accelerated development of novel drugs. Ibrutinib was developed with tremendous speed, and the FDA’s approval of the agent in 2013 based on findings from a non-pivotal phase II trial was surprising to everyone other than the participating patients and physicians. Hopefully the resources mobilized to bring ibrutinib so far, so fast, will continue to be available to help us learn how best to use the drug.
Dr. Peter Martin is with the department of medicine at Weill Cornell Medical College in New York. His comments are excerpted from an editorial that accompanied the study in The Lancet. Dr. Martin reported that he is a consultant for Janssen and has received honoraria from the company for speaking.
The findings from this phase III trial clearly establish ibrutinib as a new standard for treatment of relapsed mantle-cell lymphoma. Within the next 2 years, many expect the agent will find its way into the frontline setting for treatment of mantle cell lymphoma in combination with standard chemotherapy, based on results of another already completed phase III trial (the SHINE trial).
Despite this remarkable progress, however, mantle-cell lymphoma remains incurable. Roughly 30%-40% of people with the disease will not respond to ibrutinib, and even among responders relapse seems inevitable.
Mantle-cell lymphoma has been a model for accelerated development of novel drugs. Ibrutinib was developed with tremendous speed, and the FDA’s approval of the agent in 2013 based on findings from a non-pivotal phase II trial was surprising to everyone other than the participating patients and physicians. Hopefully the resources mobilized to bring ibrutinib so far, so fast, will continue to be available to help us learn how best to use the drug.
Dr. Peter Martin is with the department of medicine at Weill Cornell Medical College in New York. His comments are excerpted from an editorial that accompanied the study in The Lancet. Dr. Martin reported that he is a consultant for Janssen and has received honoraria from the company for speaking.
The findings from this phase III trial clearly establish ibrutinib as a new standard for treatment of relapsed mantle-cell lymphoma. Within the next 2 years, many expect the agent will find its way into the frontline setting for treatment of mantle cell lymphoma in combination with standard chemotherapy, based on results of another already completed phase III trial (the SHINE trial).
Despite this remarkable progress, however, mantle-cell lymphoma remains incurable. Roughly 30%-40% of people with the disease will not respond to ibrutinib, and even among responders relapse seems inevitable.
Mantle-cell lymphoma has been a model for accelerated development of novel drugs. Ibrutinib was developed with tremendous speed, and the FDA’s approval of the agent in 2013 based on findings from a non-pivotal phase II trial was surprising to everyone other than the participating patients and physicians. Hopefully the resources mobilized to bring ibrutinib so far, so fast, will continue to be available to help us learn how best to use the drug.
Dr. Peter Martin is with the department of medicine at Weill Cornell Medical College in New York. His comments are excerpted from an editorial that accompanied the study in The Lancet. Dr. Martin reported that he is a consultant for Janssen and has received honoraria from the company for speaking.
Progression-free survival was significantly better when patients with relapsed or refractory mantle-cell lymphoma were treated with oral ibrutinib than with intravenous temsirolimus, based on results from 280 patients in an international, randomized, open-label phase III trial.
Study subjects had undergone one or more previous rituximab-containing chemotherapy regimens to receive intravenous temsirolimus or oral ibrutinib at a daily dose of 560 mg.
Compared with temsirolimus, ibrutinib resulted in a 57% reduction in the risk of disease progression or death at a median follow-up of 20 months. Median progression-free survival – the trial’s primary endpoint – was 14.6 months for the ibrutinib group and 6.2 months for the temsirolimus group.
Ibrutinib was also better tolerated, with 68% of patients having grade 3 or higher treatment-emergent adverse events as compared to 87% of patients in the temsirolimus group, despite a median 4-fold longer treatment duration for the ibrutinib group than the temsirolimus group. Additionally, 6% of patients discontinued ibrutinib because of adverse events versus 26% in the temsirolimus group, reported Dr. Martin Dreyling of Klinikum der Universität in Munich, Germany, and his associates.
Based on results of the Functional Assessment of Cancer Therapy-Lymphoma (FACT-Lym) questionnaire, ibrutinib was associated with greater and more rapid improvements, and also with less worsening in lymphoma symptoms, as measured by the lymphoma subscale of the FACT-Lym (Lancet. 2016;387:770-78).
Ibrutinib, a first-in-class oral inhibitor of Bruton’s tyrosine kinase, is approved in the United States and the European Union at a dose of 560 mg per day for patients with mantle cell lymphoma who have received at least one previous line of therapy.
The mammalian target of rapamycin (mTOR) inhibitor temsirolimus is approved in the European Union for relapsed or refractory mantle-cell lymphoma, but does not have FDA approval for this indication.
The study, funded by Janssen, is ongoing. Future research, the investigators say, should examine ibrutinib-based combination approaches for patients with relapsed or refractory mantle-cell lymphoma and in front-line therapy.
Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study. Several other authors reported grants from Janssen during the study and financial ties to the company.
Progression-free survival was significantly better when patients with relapsed or refractory mantle-cell lymphoma were treated with oral ibrutinib than with intravenous temsirolimus, based on results from 280 patients in an international, randomized, open-label phase III trial.
Study subjects had undergone one or more previous rituximab-containing chemotherapy regimens to receive intravenous temsirolimus or oral ibrutinib at a daily dose of 560 mg.
Compared with temsirolimus, ibrutinib resulted in a 57% reduction in the risk of disease progression or death at a median follow-up of 20 months. Median progression-free survival – the trial’s primary endpoint – was 14.6 months for the ibrutinib group and 6.2 months for the temsirolimus group.
Ibrutinib was also better tolerated, with 68% of patients having grade 3 or higher treatment-emergent adverse events as compared to 87% of patients in the temsirolimus group, despite a median 4-fold longer treatment duration for the ibrutinib group than the temsirolimus group. Additionally, 6% of patients discontinued ibrutinib because of adverse events versus 26% in the temsirolimus group, reported Dr. Martin Dreyling of Klinikum der Universität in Munich, Germany, and his associates.
Based on results of the Functional Assessment of Cancer Therapy-Lymphoma (FACT-Lym) questionnaire, ibrutinib was associated with greater and more rapid improvements, and also with less worsening in lymphoma symptoms, as measured by the lymphoma subscale of the FACT-Lym (Lancet. 2016;387:770-78).
Ibrutinib, a first-in-class oral inhibitor of Bruton’s tyrosine kinase, is approved in the United States and the European Union at a dose of 560 mg per day for patients with mantle cell lymphoma who have received at least one previous line of therapy.
The mammalian target of rapamycin (mTOR) inhibitor temsirolimus is approved in the European Union for relapsed or refractory mantle-cell lymphoma, but does not have FDA approval for this indication.
The study, funded by Janssen, is ongoing. Future research, the investigators say, should examine ibrutinib-based combination approaches for patients with relapsed or refractory mantle-cell lymphoma and in front-line therapy.
Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study. Several other authors reported grants from Janssen during the study and financial ties to the company.
FROM THE LANCET
Key clinical point: Ibrutinib significantly improved progression-free survival, compared with temsirolimus in patients with relapsed or refractory mantle-cell lymphoma.
Major finding: Median progression-free survival was 14.6 months with ibrutinib and 6.2 months with temsirolimus.
Data source: A randomized open-label phase III trial (ongoing) that randomized 280 patients to each treatment group.
Disclosures: The study was funded by Janssen. Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study, and other authors reported grants from Janssen during the study and financial ties to the company.