User login
In epilepsy, brain-responsive stimulation passes long-term tests
Two new long-term studies, one an extension trial and the other an analysis of real-world experience, show that Both studies showed that the benefit from the devices increased over time.
That accruing benefit may be because of improved protocols as clinicians gain experience with the device or because of network remodeling that occurs over time as seizures are controlled. “I think it’s both,” said Martha Morrell, MD, a clinical professor of neurology at Stanford (Calif.) University and chief medical officer at NeuroPace, the company that has marketed the device since it gained FDA approval in 2013.
In both studies, the slope of improvement over time was similar, but the real-world study showed greater improvement at the beginning of treatment. “I think the slopes represent physiological changes, but the fact that [the real-world study] starts with better outcomes is, I think, directly attributable to learning. When the long-term study was started in 2004, this had never been done before, and we had to make a highly educated guess about what we should do, and the initial stimulatory parameters were programmed in a way that’s very similar to what was used for movement disorders,” Dr. Morrell said in an interview.
The long-term treatment study appeared online July 20 in the journal Neurology, while the real-world analysis was published July 13 in Epilepsia.
An alternative option
Medications can effectively treat some seizures, but 30%-40% of patients must turn to other options for control. Surgery can sometimes be curative, but is not suitable for some patients. Other stimulation devices include vagus nerve stimulation (VNS), which sends pulses from a chest implant to the vagus nerve, reducing epileptic attacks through an unknown mechanism. Deep brain stimulation (DBS) places electrodes that deliver stimulation to the anterior nucleus of the thalamus, which can spread initially localized seizures.
The RNS device consists of a neurostimulator implanted cranially and connected to leads that are placed based on the individual patient’s seizure focus or foci. It also continuously monitors brain activity and delivers stimulation only when its signal suggests the beginning of a seizure.
That capacity for recording is a key benefit because the information can be stored and analyzed, according to Vikram Rao, MD, PhD, a coinvestigator in the real-world trial and an associate professor and the epilepsy division chief at the University of California, San Francisco, which was one of the trial centers. “You know more precisely than we previously did how many seizures a patient is having. Many of our patients are not able to quantify their seizures with perfect accuracy, so we’re better quantifying their seizure burden,” Dr. Rao said in an interview.
The ability to monitor patients can also improve clinical management. Dr. Morrell recounted an elderly patient who for many years has driven 5 hours for appointments. Recently she was able to review his data from the RNS System remotely. She determined that he was doing fine and, after a telephone consultation, told him he didn’t need to come in for a scheduled visit.
Real-world analysis
In the real-world analysis, researchers led by Babak Razavi, PhD, and Casey Halpern, MD, at Stanford University conducted a chart review of 150 patients at eight centers who underwent treatment with the RNS system between 2013 and 2018. All patients were followed at least 1 year, with a mean of 2.3 years. Patients had a median of 7.7 disabling seizures per month. The mean value was 52 and the numbers ranged from 0.1 to 3,000. A total of 60% had abnormal brain MRI findings.
At 1 year, subjects achieved a mean 67% decrease in seizure frequency (interquartile range, 50%-94%). At 2 years, that grew to 77%; at 3 or more years, 84%. There was no significant difference in seizure reduction at 1 year according to age, age at epilepsy onset, duration of epilepsy, location of seizure foci, presence of brain MRI abnormalities, prior intracranial monitoring, prior epilepsy surgery, or prior VNS treatment. When patients who underwent a resection at the time of RNS placement were excluded, the results were similar. There were no significant differences in outcome by center.
A total of 11.3% of patients experienced a device-related serious adverse event, and 4% developed infections. The rate of infection was not significantly different between patients who had the neurostimulator and leads implanted alone (3.0%) and patients who had intracranial EEG diagnostic monitoring (ICM) electrodes removed at the same time (6.1%; P = .38).
Although about one-third of the patients who started the long-term study dropped out before completion, most were because the participants moved away from treatment centers, according to Dr. Morrell, and other evidence points squarely to patient satisfaction. “At the end of the battery’s longevity, the neurostimulator needs to be replaced. It’s an outpatient, 45-minute procedure. Over 90% of patients chose to have it replaced. It’s not the answer for everybody, but the substantial majority of patients choose to continue,” she said.
Extension trial
The open-label extension trial, led by Dileep Nair, MD, of the Cleveland Clinic Foundation and Dr. Morrell, followed 230 of the 256 patients who participated in 2-year phase 3 study or feasibility studies, extending device usage to 9 years. A total of 162 completed follow-up (mean, 7.5 years). The median reduction of seizure frequency was 58% at the end of year 3, and 75% by year 9 (P < .0001; Wilcoxon signed rank). Although patient population enrichment could have explained this observation, other analyses confirmed that the improvement was real.
Nearly 75% had at least a 50% reduction in seizure frequency; 35% had a 90% or greater reduction in seizure frequency. Some patients (18.4%) had at least a full year with no seizures, and 62% who had a 1-year seizure-free period experienced no seizures at the latest follow-up. Overall, 21% had no seizures in the last 6 months of follow-up.
For those with a seizure-free period of more than 1 year, the average duration was 3.2 years (range, 1.04-9.6 years). There was no difference in response among patients based on previous antiseizure medication use or previous epilepsy surgery, VNS treatment, or intracranial monitoring, and there were no differences by patient age at enrollment, age of seizure onset, brain imaging abnormality, seizure onset locality, or number of foci.
The researchers noted improvement in overall Quality of Life in Epilepsy Inventory–89 scores at 1 year (mean, +3.2; P < .0001), which continued through year 9 (mean, +1.9; P < .05). Improvements were also seen in epilepsy targeted (mean, +4.5; P < .001) and cognitive domains (mean, +2.5; P = .005). Risk of infection was 4.1% per procedure, and 12.1% of subjects overall experienced a serious device-related implant infection. Of 35 infections, 16 led to device removal.
The extension study was funded by NeuroPace. NeuroPace supported data entry and institutional review board submission for the real-world trial. Dr. Morrell owns stock and is an employee of NeuroPace. Dr Rao has received support from and/or consulted for NeuroPace.
SOURCE: Nair DR et al. Neurology. 2020 Jul 20. doi: 10.1212/WNL.0000000000010154. Razavi B et al. Epilepsia. 2020 Jul 13. doi: 10.1111/epi.16593.
Two new long-term studies, one an extension trial and the other an analysis of real-world experience, show that Both studies showed that the benefit from the devices increased over time.
That accruing benefit may be because of improved protocols as clinicians gain experience with the device or because of network remodeling that occurs over time as seizures are controlled. “I think it’s both,” said Martha Morrell, MD, a clinical professor of neurology at Stanford (Calif.) University and chief medical officer at NeuroPace, the company that has marketed the device since it gained FDA approval in 2013.
In both studies, the slope of improvement over time was similar, but the real-world study showed greater improvement at the beginning of treatment. “I think the slopes represent physiological changes, but the fact that [the real-world study] starts with better outcomes is, I think, directly attributable to learning. When the long-term study was started in 2004, this had never been done before, and we had to make a highly educated guess about what we should do, and the initial stimulatory parameters were programmed in a way that’s very similar to what was used for movement disorders,” Dr. Morrell said in an interview.
The long-term treatment study appeared online July 20 in the journal Neurology, while the real-world analysis was published July 13 in Epilepsia.
An alternative option
Medications can effectively treat some seizures, but 30%-40% of patients must turn to other options for control. Surgery can sometimes be curative, but is not suitable for some patients. Other stimulation devices include vagus nerve stimulation (VNS), which sends pulses from a chest implant to the vagus nerve, reducing epileptic attacks through an unknown mechanism. Deep brain stimulation (DBS) places electrodes that deliver stimulation to the anterior nucleus of the thalamus, which can spread initially localized seizures.
The RNS device consists of a neurostimulator implanted cranially and connected to leads that are placed based on the individual patient’s seizure focus or foci. It also continuously monitors brain activity and delivers stimulation only when its signal suggests the beginning of a seizure.
That capacity for recording is a key benefit because the information can be stored and analyzed, according to Vikram Rao, MD, PhD, a coinvestigator in the real-world trial and an associate professor and the epilepsy division chief at the University of California, San Francisco, which was one of the trial centers. “You know more precisely than we previously did how many seizures a patient is having. Many of our patients are not able to quantify their seizures with perfect accuracy, so we’re better quantifying their seizure burden,” Dr. Rao said in an interview.
The ability to monitor patients can also improve clinical management. Dr. Morrell recounted an elderly patient who for many years has driven 5 hours for appointments. Recently she was able to review his data from the RNS System remotely. She determined that he was doing fine and, after a telephone consultation, told him he didn’t need to come in for a scheduled visit.
Real-world analysis
In the real-world analysis, researchers led by Babak Razavi, PhD, and Casey Halpern, MD, at Stanford University conducted a chart review of 150 patients at eight centers who underwent treatment with the RNS system between 2013 and 2018. All patients were followed at least 1 year, with a mean of 2.3 years. Patients had a median of 7.7 disabling seizures per month. The mean value was 52 and the numbers ranged from 0.1 to 3,000. A total of 60% had abnormal brain MRI findings.
At 1 year, subjects achieved a mean 67% decrease in seizure frequency (interquartile range, 50%-94%). At 2 years, that grew to 77%; at 3 or more years, 84%. There was no significant difference in seizure reduction at 1 year according to age, age at epilepsy onset, duration of epilepsy, location of seizure foci, presence of brain MRI abnormalities, prior intracranial monitoring, prior epilepsy surgery, or prior VNS treatment. When patients who underwent a resection at the time of RNS placement were excluded, the results were similar. There were no significant differences in outcome by center.
A total of 11.3% of patients experienced a device-related serious adverse event, and 4% developed infections. The rate of infection was not significantly different between patients who had the neurostimulator and leads implanted alone (3.0%) and patients who had intracranial EEG diagnostic monitoring (ICM) electrodes removed at the same time (6.1%; P = .38).
Although about one-third of the patients who started the long-term study dropped out before completion, most were because the participants moved away from treatment centers, according to Dr. Morrell, and other evidence points squarely to patient satisfaction. “At the end of the battery’s longevity, the neurostimulator needs to be replaced. It’s an outpatient, 45-minute procedure. Over 90% of patients chose to have it replaced. It’s not the answer for everybody, but the substantial majority of patients choose to continue,” she said.
Extension trial
The open-label extension trial, led by Dileep Nair, MD, of the Cleveland Clinic Foundation and Dr. Morrell, followed 230 of the 256 patients who participated in 2-year phase 3 study or feasibility studies, extending device usage to 9 years. A total of 162 completed follow-up (mean, 7.5 years). The median reduction of seizure frequency was 58% at the end of year 3, and 75% by year 9 (P < .0001; Wilcoxon signed rank). Although patient population enrichment could have explained this observation, other analyses confirmed that the improvement was real.
Nearly 75% had at least a 50% reduction in seizure frequency; 35% had a 90% or greater reduction in seizure frequency. Some patients (18.4%) had at least a full year with no seizures, and 62% who had a 1-year seizure-free period experienced no seizures at the latest follow-up. Overall, 21% had no seizures in the last 6 months of follow-up.
For those with a seizure-free period of more than 1 year, the average duration was 3.2 years (range, 1.04-9.6 years). There was no difference in response among patients based on previous antiseizure medication use or previous epilepsy surgery, VNS treatment, or intracranial monitoring, and there were no differences by patient age at enrollment, age of seizure onset, brain imaging abnormality, seizure onset locality, or number of foci.
The researchers noted improvement in overall Quality of Life in Epilepsy Inventory–89 scores at 1 year (mean, +3.2; P < .0001), which continued through year 9 (mean, +1.9; P < .05). Improvements were also seen in epilepsy targeted (mean, +4.5; P < .001) and cognitive domains (mean, +2.5; P = .005). Risk of infection was 4.1% per procedure, and 12.1% of subjects overall experienced a serious device-related implant infection. Of 35 infections, 16 led to device removal.
The extension study was funded by NeuroPace. NeuroPace supported data entry and institutional review board submission for the real-world trial. Dr. Morrell owns stock and is an employee of NeuroPace. Dr Rao has received support from and/or consulted for NeuroPace.
SOURCE: Nair DR et al. Neurology. 2020 Jul 20. doi: 10.1212/WNL.0000000000010154. Razavi B et al. Epilepsia. 2020 Jul 13. doi: 10.1111/epi.16593.
Two new long-term studies, one an extension trial and the other an analysis of real-world experience, show that Both studies showed that the benefit from the devices increased over time.
That accruing benefit may be because of improved protocols as clinicians gain experience with the device or because of network remodeling that occurs over time as seizures are controlled. “I think it’s both,” said Martha Morrell, MD, a clinical professor of neurology at Stanford (Calif.) University and chief medical officer at NeuroPace, the company that has marketed the device since it gained FDA approval in 2013.
In both studies, the slope of improvement over time was similar, but the real-world study showed greater improvement at the beginning of treatment. “I think the slopes represent physiological changes, but the fact that [the real-world study] starts with better outcomes is, I think, directly attributable to learning. When the long-term study was started in 2004, this had never been done before, and we had to make a highly educated guess about what we should do, and the initial stimulatory parameters were programmed in a way that’s very similar to what was used for movement disorders,” Dr. Morrell said in an interview.
The long-term treatment study appeared online July 20 in the journal Neurology, while the real-world analysis was published July 13 in Epilepsia.
An alternative option
Medications can effectively treat some seizures, but 30%-40% of patients must turn to other options for control. Surgery can sometimes be curative, but is not suitable for some patients. Other stimulation devices include vagus nerve stimulation (VNS), which sends pulses from a chest implant to the vagus nerve, reducing epileptic attacks through an unknown mechanism. Deep brain stimulation (DBS) places electrodes that deliver stimulation to the anterior nucleus of the thalamus, which can spread initially localized seizures.
The RNS device consists of a neurostimulator implanted cranially and connected to leads that are placed based on the individual patient’s seizure focus or foci. It also continuously monitors brain activity and delivers stimulation only when its signal suggests the beginning of a seizure.
That capacity for recording is a key benefit because the information can be stored and analyzed, according to Vikram Rao, MD, PhD, a coinvestigator in the real-world trial and an associate professor and the epilepsy division chief at the University of California, San Francisco, which was one of the trial centers. “You know more precisely than we previously did how many seizures a patient is having. Many of our patients are not able to quantify their seizures with perfect accuracy, so we’re better quantifying their seizure burden,” Dr. Rao said in an interview.
The ability to monitor patients can also improve clinical management. Dr. Morrell recounted an elderly patient who for many years has driven 5 hours for appointments. Recently she was able to review his data from the RNS System remotely. She determined that he was doing fine and, after a telephone consultation, told him he didn’t need to come in for a scheduled visit.
Real-world analysis
In the real-world analysis, researchers led by Babak Razavi, PhD, and Casey Halpern, MD, at Stanford University conducted a chart review of 150 patients at eight centers who underwent treatment with the RNS system between 2013 and 2018. All patients were followed at least 1 year, with a mean of 2.3 years. Patients had a median of 7.7 disabling seizures per month. The mean value was 52 and the numbers ranged from 0.1 to 3,000. A total of 60% had abnormal brain MRI findings.
At 1 year, subjects achieved a mean 67% decrease in seizure frequency (interquartile range, 50%-94%). At 2 years, that grew to 77%; at 3 or more years, 84%. There was no significant difference in seizure reduction at 1 year according to age, age at epilepsy onset, duration of epilepsy, location of seizure foci, presence of brain MRI abnormalities, prior intracranial monitoring, prior epilepsy surgery, or prior VNS treatment. When patients who underwent a resection at the time of RNS placement were excluded, the results were similar. There were no significant differences in outcome by center.
A total of 11.3% of patients experienced a device-related serious adverse event, and 4% developed infections. The rate of infection was not significantly different between patients who had the neurostimulator and leads implanted alone (3.0%) and patients who had intracranial EEG diagnostic monitoring (ICM) electrodes removed at the same time (6.1%; P = .38).
Although about one-third of the patients who started the long-term study dropped out before completion, most were because the participants moved away from treatment centers, according to Dr. Morrell, and other evidence points squarely to patient satisfaction. “At the end of the battery’s longevity, the neurostimulator needs to be replaced. It’s an outpatient, 45-minute procedure. Over 90% of patients chose to have it replaced. It’s not the answer for everybody, but the substantial majority of patients choose to continue,” she said.
Extension trial
The open-label extension trial, led by Dileep Nair, MD, of the Cleveland Clinic Foundation and Dr. Morrell, followed 230 of the 256 patients who participated in 2-year phase 3 study or feasibility studies, extending device usage to 9 years. A total of 162 completed follow-up (mean, 7.5 years). The median reduction of seizure frequency was 58% at the end of year 3, and 75% by year 9 (P < .0001; Wilcoxon signed rank). Although patient population enrichment could have explained this observation, other analyses confirmed that the improvement was real.
Nearly 75% had at least a 50% reduction in seizure frequency; 35% had a 90% or greater reduction in seizure frequency. Some patients (18.4%) had at least a full year with no seizures, and 62% who had a 1-year seizure-free period experienced no seizures at the latest follow-up. Overall, 21% had no seizures in the last 6 months of follow-up.
For those with a seizure-free period of more than 1 year, the average duration was 3.2 years (range, 1.04-9.6 years). There was no difference in response among patients based on previous antiseizure medication use or previous epilepsy surgery, VNS treatment, or intracranial monitoring, and there were no differences by patient age at enrollment, age of seizure onset, brain imaging abnormality, seizure onset locality, or number of foci.
The researchers noted improvement in overall Quality of Life in Epilepsy Inventory–89 scores at 1 year (mean, +3.2; P < .0001), which continued through year 9 (mean, +1.9; P < .05). Improvements were also seen in epilepsy targeted (mean, +4.5; P < .001) and cognitive domains (mean, +2.5; P = .005). Risk of infection was 4.1% per procedure, and 12.1% of subjects overall experienced a serious device-related implant infection. Of 35 infections, 16 led to device removal.
The extension study was funded by NeuroPace. NeuroPace supported data entry and institutional review board submission for the real-world trial. Dr. Morrell owns stock and is an employee of NeuroPace. Dr Rao has received support from and/or consulted for NeuroPace.
SOURCE: Nair DR et al. Neurology. 2020 Jul 20. doi: 10.1212/WNL.0000000000010154. Razavi B et al. Epilepsia. 2020 Jul 13. doi: 10.1111/epi.16593.
FROM EPILEPSIA AND FROM NEUROLOGY
Psychiatrists report rare case of woman who thinks she’s a chicken
LEUVEN, Belgium — A 54-year-old woman has suffered the delusion of thinking she is a chicken for 24 hours. This very rare condition, known as zoanthropy, in which people think they are an animal is often not recognised, say researchers from the University of Leuven.
Zoanthropy can include people believing they are, or behaving like, any kind of animal: from a dog, to a lion or tiger, crocodile, snake, or bee.
It’s important to recognise this as a potential symptom of something serious, say the researchers in the July issue of the Belgian Journal of Psychiatry, Tijdschrift voor Psychiatrie.
“Additional investigations with brain imaging and electroencephalogram are therefore advised,” say the authors.
Psychiatrists Need to Be Aware That Clinical Zoanthropy Exists
In their paper, they describe the case of the woman who briefly thought she was a chicken, which was followed by her having a generalized epileptic seizure.
“Clinically, we saw a lady who perspired profusely, trembled, blew up her cheeks, and ... seemed to imitate a chicken, [making noises] like clucking, cackling, and crowing like a rooster,” they say.
“After about 10 minutes she seemed to tighten her muscles for a few seconds, her face turned red and for a short time she didn’t react. These symptoms repeated themselves at intervals of a few minutes [and her] consciousness was fluctuating,” with the patient “disoriented in time and space.”
Lead author Dr Athena Beckers of University Psychiatric Centre, KU Leuven, Belgium, said in an interview with MediQuality: “With only 56 case descriptions in the medical literature from 1850 to the present day, the condition is rare. It amounts to about one description every 3 years.
“We suspect, however, that the delusion is not always noticed: the patient shows bizarre behaviour or makes animal sounds, it is probably often catalogued under the general term ‘psychosis’.”
Dr Beckers adds that it is important that the symptoms are recognised, because of the possible underlying causes which can include epilepsy. So this might require a different or complementary treatment “with, for example, antiepileptic drugs”.
“I myself have only seen this type of delusion once, but I ... heard anecdotal stories from other patients whose family member, for example with schizophrenia, sometimes thought he was a cow [during] ... a psychosis.
“After the publication of my article I was also contacted by someone who told me they had experienced the same thing 30 years ago – he thought he was a chicken.
“I think it’s a good thing that we psychiatrists are aware of the fact that clinical zoanthropy exists and may require additional research,” she observed.
Fortunately, this woman’s experience ended well. After about one year of disability, the patient was able to return to work progressively. Her mood remained stable and there were no more psychotic symptoms or any indication of epileptic episodes.
Such Delusions Are Rare
Dr Georges Otte, a recently retired neuropsychiatrist who formerly worked at Ghent University, Belgium, gave his thoughts to Mediquality: “The interface between neurology and psychiatry ... is a fertile meadow on which many crops thrive. But it is in the darkest corners of psychosis that one finds the most bizarre and also rarest excesses.”
There are a number of delusions of identity, said Dr Otte.
These include Cotard’s syndrome, a rare condition marked by the false belief that the person or their body parts are dead, dying, or don’t exist, or Capgras delusion, where the affected person believes that a spouse or close family member has been replaced with an imposter. Delusions can also occur as a result of substance abuse, for example after using psilocybin (magic mushrooms), he added.
“Delusions in which patients are convinced of ‘shape shifting’ (man to animal) are quite rare,” Dr Otte observed.
“In the literature we know that lycanthropy [a person thinks he or she is turning into a werewolf],” has been reported, and has “apparently inspired many authors of horror stories,” he added.
“But it’s not every day that as a psychiatrist, you will encounter such an extreme psychotic depersonalization as someone turning into a chicken.”
This article first appeared on Medscape.com.
LEUVEN, Belgium — A 54-year-old woman has suffered the delusion of thinking she is a chicken for 24 hours. This very rare condition, known as zoanthropy, in which people think they are an animal is often not recognised, say researchers from the University of Leuven.
Zoanthropy can include people believing they are, or behaving like, any kind of animal: from a dog, to a lion or tiger, crocodile, snake, or bee.
It’s important to recognise this as a potential symptom of something serious, say the researchers in the July issue of the Belgian Journal of Psychiatry, Tijdschrift voor Psychiatrie.
“Additional investigations with brain imaging and electroencephalogram are therefore advised,” say the authors.
Psychiatrists Need to Be Aware That Clinical Zoanthropy Exists
In their paper, they describe the case of the woman who briefly thought she was a chicken, which was followed by her having a generalized epileptic seizure.
“Clinically, we saw a lady who perspired profusely, trembled, blew up her cheeks, and ... seemed to imitate a chicken, [making noises] like clucking, cackling, and crowing like a rooster,” they say.
“After about 10 minutes she seemed to tighten her muscles for a few seconds, her face turned red and for a short time she didn’t react. These symptoms repeated themselves at intervals of a few minutes [and her] consciousness was fluctuating,” with the patient “disoriented in time and space.”
Lead author Dr Athena Beckers of University Psychiatric Centre, KU Leuven, Belgium, said in an interview with MediQuality: “With only 56 case descriptions in the medical literature from 1850 to the present day, the condition is rare. It amounts to about one description every 3 years.
“We suspect, however, that the delusion is not always noticed: the patient shows bizarre behaviour or makes animal sounds, it is probably often catalogued under the general term ‘psychosis’.”
Dr Beckers adds that it is important that the symptoms are recognised, because of the possible underlying causes which can include epilepsy. So this might require a different or complementary treatment “with, for example, antiepileptic drugs”.
“I myself have only seen this type of delusion once, but I ... heard anecdotal stories from other patients whose family member, for example with schizophrenia, sometimes thought he was a cow [during] ... a psychosis.
“After the publication of my article I was also contacted by someone who told me they had experienced the same thing 30 years ago – he thought he was a chicken.
“I think it’s a good thing that we psychiatrists are aware of the fact that clinical zoanthropy exists and may require additional research,” she observed.
Fortunately, this woman’s experience ended well. After about one year of disability, the patient was able to return to work progressively. Her mood remained stable and there were no more psychotic symptoms or any indication of epileptic episodes.
Such Delusions Are Rare
Dr Georges Otte, a recently retired neuropsychiatrist who formerly worked at Ghent University, Belgium, gave his thoughts to Mediquality: “The interface between neurology and psychiatry ... is a fertile meadow on which many crops thrive. But it is in the darkest corners of psychosis that one finds the most bizarre and also rarest excesses.”
There are a number of delusions of identity, said Dr Otte.
These include Cotard’s syndrome, a rare condition marked by the false belief that the person or their body parts are dead, dying, or don’t exist, or Capgras delusion, where the affected person believes that a spouse or close family member has been replaced with an imposter. Delusions can also occur as a result of substance abuse, for example after using psilocybin (magic mushrooms), he added.
“Delusions in which patients are convinced of ‘shape shifting’ (man to animal) are quite rare,” Dr Otte observed.
“In the literature we know that lycanthropy [a person thinks he or she is turning into a werewolf],” has been reported, and has “apparently inspired many authors of horror stories,” he added.
“But it’s not every day that as a psychiatrist, you will encounter such an extreme psychotic depersonalization as someone turning into a chicken.”
This article first appeared on Medscape.com.
LEUVEN, Belgium — A 54-year-old woman has suffered the delusion of thinking she is a chicken for 24 hours. This very rare condition, known as zoanthropy, in which people think they are an animal is often not recognised, say researchers from the University of Leuven.
Zoanthropy can include people believing they are, or behaving like, any kind of animal: from a dog, to a lion or tiger, crocodile, snake, or bee.
It’s important to recognise this as a potential symptom of something serious, say the researchers in the July issue of the Belgian Journal of Psychiatry, Tijdschrift voor Psychiatrie.
“Additional investigations with brain imaging and electroencephalogram are therefore advised,” say the authors.
Psychiatrists Need to Be Aware That Clinical Zoanthropy Exists
In their paper, they describe the case of the woman who briefly thought she was a chicken, which was followed by her having a generalized epileptic seizure.
“Clinically, we saw a lady who perspired profusely, trembled, blew up her cheeks, and ... seemed to imitate a chicken, [making noises] like clucking, cackling, and crowing like a rooster,” they say.
“After about 10 minutes she seemed to tighten her muscles for a few seconds, her face turned red and for a short time she didn’t react. These symptoms repeated themselves at intervals of a few minutes [and her] consciousness was fluctuating,” with the patient “disoriented in time and space.”
Lead author Dr Athena Beckers of University Psychiatric Centre, KU Leuven, Belgium, said in an interview with MediQuality: “With only 56 case descriptions in the medical literature from 1850 to the present day, the condition is rare. It amounts to about one description every 3 years.
“We suspect, however, that the delusion is not always noticed: the patient shows bizarre behaviour or makes animal sounds, it is probably often catalogued under the general term ‘psychosis’.”
Dr Beckers adds that it is important that the symptoms are recognised, because of the possible underlying causes which can include epilepsy. So this might require a different or complementary treatment “with, for example, antiepileptic drugs”.
“I myself have only seen this type of delusion once, but I ... heard anecdotal stories from other patients whose family member, for example with schizophrenia, sometimes thought he was a cow [during] ... a psychosis.
“After the publication of my article I was also contacted by someone who told me they had experienced the same thing 30 years ago – he thought he was a chicken.
“I think it’s a good thing that we psychiatrists are aware of the fact that clinical zoanthropy exists and may require additional research,” she observed.
Fortunately, this woman’s experience ended well. After about one year of disability, the patient was able to return to work progressively. Her mood remained stable and there were no more psychotic symptoms or any indication of epileptic episodes.
Such Delusions Are Rare
Dr Georges Otte, a recently retired neuropsychiatrist who formerly worked at Ghent University, Belgium, gave his thoughts to Mediquality: “The interface between neurology and psychiatry ... is a fertile meadow on which many crops thrive. But it is in the darkest corners of psychosis that one finds the most bizarre and also rarest excesses.”
There are a number of delusions of identity, said Dr Otte.
These include Cotard’s syndrome, a rare condition marked by the false belief that the person or their body parts are dead, dying, or don’t exist, or Capgras delusion, where the affected person believes that a spouse or close family member has been replaced with an imposter. Delusions can also occur as a result of substance abuse, for example after using psilocybin (magic mushrooms), he added.
“Delusions in which patients are convinced of ‘shape shifting’ (man to animal) are quite rare,” Dr Otte observed.
“In the literature we know that lycanthropy [a person thinks he or she is turning into a werewolf],” has been reported, and has “apparently inspired many authors of horror stories,” he added.
“But it’s not every day that as a psychiatrist, you will encounter such an extreme psychotic depersonalization as someone turning into a chicken.”
This article first appeared on Medscape.com.
Microbiome research ‘opening doors’ to new Alzheimer’s disease treatments
Research into the microbiome is yielding some positive new potential treatment options for Alzheimer’s disease, according to George T. Grossberg, MD.
“I think the growing focus on the gut-brain axis is opening doors to new Alzheimer’s disease and other brain disorders, and I think the first of a possible future generation of compounds for prevention or treatment of Alzheimer’s disease may indeed be emerging,” Dr. Grossberg said at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
Focus on the microbiome and microbiota is “a really hot, really new, really emerging area,” said Dr. Grossberg, professor in the department of psychiatry & behavioral neuroscience at Saint Louis University. But the microbiota, which is the microorganisms within a specific organ such as the colon, is sometimes confused with the microbiome – which is defined as all of the bacteria, viruses, fungi and other microorganisms within a habitat as well as their genomes and the environment around them. “These are often used interchangeably, but they’re not the same,” Dr. Grossberg said at the meeting, presented by Global Academy for Medical Education.
A person’s microbiome is unique to them, and nearly all of the microbiome is contained in the gut. A reduction in diversity of the microbiota in the digestive system has been linked to a wide variety of diseases, Dr. Grossberg explained. Conversely, a microbial imbalance or dysbiosis has been implicated in anxiety and/or depression, dementia, and certain cancers, he noted.
Bacteria that positively affect the microbiome come from two main genera: Lactobacillus and Bifidobacterium. Factors such as diet, medications, geography, stage of life, birthing process, infant feeding method, and stress can all affect a person’s microbiome. “We’re all beginning to understand that trying to manage or trying to diversify, trying to manipulate the microbiota may have a lot of remote effects – even effects on weight or diabetes, or other disorders,” Dr. Grossberg said.
Fecal microbiota transplantation (FMT), or the process of administering a donor’s fecal matter into a recipient’s intestinal tract, has proved beneficial in improving the health of patients suffering from recurrent Clostridioides difficile infection. A recent Harvard Health Letter, written by Jessica Allegretti, MD, MPH, observed that FMT is standard of care for patients with C. diff, and the procedure has a success rate of between 80% and 90%.
“It shows us very directly, in a very practical way, how addressing the dysbiosis – the imbalance of the gut microbiome – by infusing healthy bacteria may make a potential lifesaving difference,” Dr. Grossberg said.
Research is beginning to show that the link between gut microbiota and health extends to Alzheimer’s disease as well. Within the last few years, “we’ve started to understand that the microbial diversity in Alzheimer’s disease versus healthy age-matched controls is decreased,” Dr. Grossberg said.
In a study published by Nicholas M. Vogt and colleagues, there was decreased fecal microbial diversity among individuals with Alzheimer’s, compared with healthy individuals matched for age. Another study by Ping Liu, PhD, and colleagues found that patients with Alzheimer’s disease had decreased fecal microbial diversity, compared with individuals who had pre-onset amnestic mild cognitive impairment and normal cognition.
Dr. Grossberg noted that, while these studies do not prove that less fecal microbial diversity is responsible for mild cognitive impairment or Alzheimer’s disease, “it makes us think that, maybe, there’s a contributing factor.”
“What happens with the dysbiosis of the gut microbiome is increased permeability of the epithelial area of the gut, which can then lead to the gut-brain axis dysregulation and may in fact allow the selective entry of bacteria into the central nervous system because the blood-brain barrier comes to be dysfunctional,” he said.
Early evidence suggests that the gut-brain axis can affect cognition. In an animal model study, transferring the microbiota of a mouse with Alzheimer’s disease to one that had been bred to be germ-free resulted in cognitive decline – but there was no cognitive decline for germ-free mice that received a microbiota transplant from a mouse in a healthy control group. Results from another animal study showed that transferring healthy microbiota from a mouse model into a mouse with Alzheimer’s disease reduces amyloid and tau pathology. “The conclusions of these studies seems to be that microbiota mediated intestinal and systemic immune changes or aberrations seem to contribute to the pathogenesis of Alzheimer’s disease in these mouse models,” Dr. Grossberg said. “Consequently, restoring the gut microbial homeostasis may have beneficial effects on Alzheimer’s disease treatment.”
Periodontal disease also might be linked to Alzheimer’s disease, Dr. Grossberg said. Several studies have shown gingipains secreted from Porphyromonas gingivalis, which contribute to inflammation in the brain, have been found in cadavers of patients with Alzheimer’s disease (Sci Adv. 2019 Jan 23;5[1]:eaau3333). “There’s reason to think that the same changes may be occurring in the human brain with periodontal disease,” he said.
The relationship also might extend to the gut microbiota and the central nervous system. “There seems to be a direct communication, a direct relationship between normal gut physiology and healthy central nervous system functioning, and then, when you have abnormal gut function, it may result in a variety of abnormal central nervous system functions,” Dr. Grossberg said.
Studies that have examined a relationship between Alzheimer’s disease and gut microbiota have highlighted the potential of probiotics and prebiotics as a method of restoring the gut microbiota (Aging [Albany NY]. 2020 Mar 31; 12[6]:5539-50). Probiotics are popularly sold in health food aisles of grocery stores, and prebiotics are available in foods such as yogurts, tempeh, sauerkraut, and kimchi, as well as in drinks such as Kombucha tea. The effectiveness of probiotics and prebiotics also are being examined in randomized, controlled trials in patients with mild cognitive decline and mild Alzheimer’s disease, Dr. Grossberg said. One therapy, Sodium oligomannate, a marine algae–derived oral oligosaccharide, has shown effectiveness in remodeling gut microbiota and has been approved in China to treat patients with mild or moderate Alzheimer’s disease. Currently, no approved gut microbiota therapies are approved in the United States to treat Alzheimer’s disease; however, encouraging use of a prebiotic, a probiotic, or a Mediterranean diet is something clinicians might want to consider for their patients.
“The fact that we’re studying these things has really led to the notion that it may not be a bad idea for people to consume these healthy bacteria in later life, either as a way to prevent or delay, or to treat Alzheimer’s disease,” Dr. Grossberg said. “There’s really no downside.”
Global Academy and this news organization are owned by the same parent company. Dr. Grossberg reported that he is a consultant for Acadia, Alkahest, Avanir, Axsome, Biogen, BioXcel, Karuna, Lundbeck, Otsuka, Roche, and Takeda; receives research support from the National Institute on Aging, Janssen, and Roche; performs safety monitoring for EryDel, Merck, and Newron; and serves on data monitoring committees for Avanex and ITI Therapeutics.
Research into the microbiome is yielding some positive new potential treatment options for Alzheimer’s disease, according to George T. Grossberg, MD.
“I think the growing focus on the gut-brain axis is opening doors to new Alzheimer’s disease and other brain disorders, and I think the first of a possible future generation of compounds for prevention or treatment of Alzheimer’s disease may indeed be emerging,” Dr. Grossberg said at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
Focus on the microbiome and microbiota is “a really hot, really new, really emerging area,” said Dr. Grossberg, professor in the department of psychiatry & behavioral neuroscience at Saint Louis University. But the microbiota, which is the microorganisms within a specific organ such as the colon, is sometimes confused with the microbiome – which is defined as all of the bacteria, viruses, fungi and other microorganisms within a habitat as well as their genomes and the environment around them. “These are often used interchangeably, but they’re not the same,” Dr. Grossberg said at the meeting, presented by Global Academy for Medical Education.
A person’s microbiome is unique to them, and nearly all of the microbiome is contained in the gut. A reduction in diversity of the microbiota in the digestive system has been linked to a wide variety of diseases, Dr. Grossberg explained. Conversely, a microbial imbalance or dysbiosis has been implicated in anxiety and/or depression, dementia, and certain cancers, he noted.
Bacteria that positively affect the microbiome come from two main genera: Lactobacillus and Bifidobacterium. Factors such as diet, medications, geography, stage of life, birthing process, infant feeding method, and stress can all affect a person’s microbiome. “We’re all beginning to understand that trying to manage or trying to diversify, trying to manipulate the microbiota may have a lot of remote effects – even effects on weight or diabetes, or other disorders,” Dr. Grossberg said.
Fecal microbiota transplantation (FMT), or the process of administering a donor’s fecal matter into a recipient’s intestinal tract, has proved beneficial in improving the health of patients suffering from recurrent Clostridioides difficile infection. A recent Harvard Health Letter, written by Jessica Allegretti, MD, MPH, observed that FMT is standard of care for patients with C. diff, and the procedure has a success rate of between 80% and 90%.
“It shows us very directly, in a very practical way, how addressing the dysbiosis – the imbalance of the gut microbiome – by infusing healthy bacteria may make a potential lifesaving difference,” Dr. Grossberg said.
Research is beginning to show that the link between gut microbiota and health extends to Alzheimer’s disease as well. Within the last few years, “we’ve started to understand that the microbial diversity in Alzheimer’s disease versus healthy age-matched controls is decreased,” Dr. Grossberg said.
In a study published by Nicholas M. Vogt and colleagues, there was decreased fecal microbial diversity among individuals with Alzheimer’s, compared with healthy individuals matched for age. Another study by Ping Liu, PhD, and colleagues found that patients with Alzheimer’s disease had decreased fecal microbial diversity, compared with individuals who had pre-onset amnestic mild cognitive impairment and normal cognition.
Dr. Grossberg noted that, while these studies do not prove that less fecal microbial diversity is responsible for mild cognitive impairment or Alzheimer’s disease, “it makes us think that, maybe, there’s a contributing factor.”
“What happens with the dysbiosis of the gut microbiome is increased permeability of the epithelial area of the gut, which can then lead to the gut-brain axis dysregulation and may in fact allow the selective entry of bacteria into the central nervous system because the blood-brain barrier comes to be dysfunctional,” he said.
Early evidence suggests that the gut-brain axis can affect cognition. In an animal model study, transferring the microbiota of a mouse with Alzheimer’s disease to one that had been bred to be germ-free resulted in cognitive decline – but there was no cognitive decline for germ-free mice that received a microbiota transplant from a mouse in a healthy control group. Results from another animal study showed that transferring healthy microbiota from a mouse model into a mouse with Alzheimer’s disease reduces amyloid and tau pathology. “The conclusions of these studies seems to be that microbiota mediated intestinal and systemic immune changes or aberrations seem to contribute to the pathogenesis of Alzheimer’s disease in these mouse models,” Dr. Grossberg said. “Consequently, restoring the gut microbial homeostasis may have beneficial effects on Alzheimer’s disease treatment.”
Periodontal disease also might be linked to Alzheimer’s disease, Dr. Grossberg said. Several studies have shown gingipains secreted from Porphyromonas gingivalis, which contribute to inflammation in the brain, have been found in cadavers of patients with Alzheimer’s disease (Sci Adv. 2019 Jan 23;5[1]:eaau3333). “There’s reason to think that the same changes may be occurring in the human brain with periodontal disease,” he said.
The relationship also might extend to the gut microbiota and the central nervous system. “There seems to be a direct communication, a direct relationship between normal gut physiology and healthy central nervous system functioning, and then, when you have abnormal gut function, it may result in a variety of abnormal central nervous system functions,” Dr. Grossberg said.
Studies that have examined a relationship between Alzheimer’s disease and gut microbiota have highlighted the potential of probiotics and prebiotics as a method of restoring the gut microbiota (Aging [Albany NY]. 2020 Mar 31; 12[6]:5539-50). Probiotics are popularly sold in health food aisles of grocery stores, and prebiotics are available in foods such as yogurts, tempeh, sauerkraut, and kimchi, as well as in drinks such as Kombucha tea. The effectiveness of probiotics and prebiotics also are being examined in randomized, controlled trials in patients with mild cognitive decline and mild Alzheimer’s disease, Dr. Grossberg said. One therapy, Sodium oligomannate, a marine algae–derived oral oligosaccharide, has shown effectiveness in remodeling gut microbiota and has been approved in China to treat patients with mild or moderate Alzheimer’s disease. Currently, no approved gut microbiota therapies are approved in the United States to treat Alzheimer’s disease; however, encouraging use of a prebiotic, a probiotic, or a Mediterranean diet is something clinicians might want to consider for their patients.
“The fact that we’re studying these things has really led to the notion that it may not be a bad idea for people to consume these healthy bacteria in later life, either as a way to prevent or delay, or to treat Alzheimer’s disease,” Dr. Grossberg said. “There’s really no downside.”
Global Academy and this news organization are owned by the same parent company. Dr. Grossberg reported that he is a consultant for Acadia, Alkahest, Avanir, Axsome, Biogen, BioXcel, Karuna, Lundbeck, Otsuka, Roche, and Takeda; receives research support from the National Institute on Aging, Janssen, and Roche; performs safety monitoring for EryDel, Merck, and Newron; and serves on data monitoring committees for Avanex and ITI Therapeutics.
Research into the microbiome is yielding some positive new potential treatment options for Alzheimer’s disease, according to George T. Grossberg, MD.
“I think the growing focus on the gut-brain axis is opening doors to new Alzheimer’s disease and other brain disorders, and I think the first of a possible future generation of compounds for prevention or treatment of Alzheimer’s disease may indeed be emerging,” Dr. Grossberg said at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
Focus on the microbiome and microbiota is “a really hot, really new, really emerging area,” said Dr. Grossberg, professor in the department of psychiatry & behavioral neuroscience at Saint Louis University. But the microbiota, which is the microorganisms within a specific organ such as the colon, is sometimes confused with the microbiome – which is defined as all of the bacteria, viruses, fungi and other microorganisms within a habitat as well as their genomes and the environment around them. “These are often used interchangeably, but they’re not the same,” Dr. Grossberg said at the meeting, presented by Global Academy for Medical Education.
A person’s microbiome is unique to them, and nearly all of the microbiome is contained in the gut. A reduction in diversity of the microbiota in the digestive system has been linked to a wide variety of diseases, Dr. Grossberg explained. Conversely, a microbial imbalance or dysbiosis has been implicated in anxiety and/or depression, dementia, and certain cancers, he noted.
Bacteria that positively affect the microbiome come from two main genera: Lactobacillus and Bifidobacterium. Factors such as diet, medications, geography, stage of life, birthing process, infant feeding method, and stress can all affect a person’s microbiome. “We’re all beginning to understand that trying to manage or trying to diversify, trying to manipulate the microbiota may have a lot of remote effects – even effects on weight or diabetes, or other disorders,” Dr. Grossberg said.
Fecal microbiota transplantation (FMT), or the process of administering a donor’s fecal matter into a recipient’s intestinal tract, has proved beneficial in improving the health of patients suffering from recurrent Clostridioides difficile infection. A recent Harvard Health Letter, written by Jessica Allegretti, MD, MPH, observed that FMT is standard of care for patients with C. diff, and the procedure has a success rate of between 80% and 90%.
“It shows us very directly, in a very practical way, how addressing the dysbiosis – the imbalance of the gut microbiome – by infusing healthy bacteria may make a potential lifesaving difference,” Dr. Grossberg said.
Research is beginning to show that the link between gut microbiota and health extends to Alzheimer’s disease as well. Within the last few years, “we’ve started to understand that the microbial diversity in Alzheimer’s disease versus healthy age-matched controls is decreased,” Dr. Grossberg said.
In a study published by Nicholas M. Vogt and colleagues, there was decreased fecal microbial diversity among individuals with Alzheimer’s, compared with healthy individuals matched for age. Another study by Ping Liu, PhD, and colleagues found that patients with Alzheimer’s disease had decreased fecal microbial diversity, compared with individuals who had pre-onset amnestic mild cognitive impairment and normal cognition.
Dr. Grossberg noted that, while these studies do not prove that less fecal microbial diversity is responsible for mild cognitive impairment or Alzheimer’s disease, “it makes us think that, maybe, there’s a contributing factor.”
“What happens with the dysbiosis of the gut microbiome is increased permeability of the epithelial area of the gut, which can then lead to the gut-brain axis dysregulation and may in fact allow the selective entry of bacteria into the central nervous system because the blood-brain barrier comes to be dysfunctional,” he said.
Early evidence suggests that the gut-brain axis can affect cognition. In an animal model study, transferring the microbiota of a mouse with Alzheimer’s disease to one that had been bred to be germ-free resulted in cognitive decline – but there was no cognitive decline for germ-free mice that received a microbiota transplant from a mouse in a healthy control group. Results from another animal study showed that transferring healthy microbiota from a mouse model into a mouse with Alzheimer’s disease reduces amyloid and tau pathology. “The conclusions of these studies seems to be that microbiota mediated intestinal and systemic immune changes or aberrations seem to contribute to the pathogenesis of Alzheimer’s disease in these mouse models,” Dr. Grossberg said. “Consequently, restoring the gut microbial homeostasis may have beneficial effects on Alzheimer’s disease treatment.”
Periodontal disease also might be linked to Alzheimer’s disease, Dr. Grossberg said. Several studies have shown gingipains secreted from Porphyromonas gingivalis, which contribute to inflammation in the brain, have been found in cadavers of patients with Alzheimer’s disease (Sci Adv. 2019 Jan 23;5[1]:eaau3333). “There’s reason to think that the same changes may be occurring in the human brain with periodontal disease,” he said.
The relationship also might extend to the gut microbiota and the central nervous system. “There seems to be a direct communication, a direct relationship between normal gut physiology and healthy central nervous system functioning, and then, when you have abnormal gut function, it may result in a variety of abnormal central nervous system functions,” Dr. Grossberg said.
Studies that have examined a relationship between Alzheimer’s disease and gut microbiota have highlighted the potential of probiotics and prebiotics as a method of restoring the gut microbiota (Aging [Albany NY]. 2020 Mar 31; 12[6]:5539-50). Probiotics are popularly sold in health food aisles of grocery stores, and prebiotics are available in foods such as yogurts, tempeh, sauerkraut, and kimchi, as well as in drinks such as Kombucha tea. The effectiveness of probiotics and prebiotics also are being examined in randomized, controlled trials in patients with mild cognitive decline and mild Alzheimer’s disease, Dr. Grossberg said. One therapy, Sodium oligomannate, a marine algae–derived oral oligosaccharide, has shown effectiveness in remodeling gut microbiota and has been approved in China to treat patients with mild or moderate Alzheimer’s disease. Currently, no approved gut microbiota therapies are approved in the United States to treat Alzheimer’s disease; however, encouraging use of a prebiotic, a probiotic, or a Mediterranean diet is something clinicians might want to consider for their patients.
“The fact that we’re studying these things has really led to the notion that it may not be a bad idea for people to consume these healthy bacteria in later life, either as a way to prevent or delay, or to treat Alzheimer’s disease,” Dr. Grossberg said. “There’s really no downside.”
Global Academy and this news organization are owned by the same parent company. Dr. Grossberg reported that he is a consultant for Acadia, Alkahest, Avanir, Axsome, Biogen, BioXcel, Karuna, Lundbeck, Otsuka, Roche, and Takeda; receives research support from the National Institute on Aging, Janssen, and Roche; performs safety monitoring for EryDel, Merck, and Newron; and serves on data monitoring committees for Avanex and ITI Therapeutics.
FROM CP/AACP PSYCHIATRY UPDATE
A better tau blood test for diagnosing Alzheimer’s disease?
.
In one new development, experts at the University of California, San Francisco (UCSF) compared phosphorylated-tau181 (P-tau181) to a related form of tau called P-tau217 to determine which can best identify individuals with Alzheimer’s disease.
Results showed that the two biomarkers were similar overall, but P-tau 217 had a slight edge in terms of accuracy. Importantly, both tau isoforms distinguished frontotemporal lobar degeneration (FTLD).
“These new blood tests for P-tau are going to be really exciting because they will improve our ability to simply and inexpensively assess whether someone is at high risk for having Alzheimer’s disease,” said study author Adam L. Boxer, MD, PhD, professor in UCSF’s department of neurology.
With the approval of the first disease-modifying therapy for Alzheimer’s disease possibly around the corner, developing an accurate diagnostic blood test for this condition is even more urgent, added Dr. Boxer, who is also director of UCSF’s Neurosciences Clinical Research Unit and AD and FTD Clinical Trials Program.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Important implications
Currently, the only approved Alzheimer’s disease biomarkers are expensive positron emission tomography (PET) scans using agents that detect tau or amyloid, another hallmark Alzheimer’s disease protein, and cerebrospinal fluid levels of amyloid and tau, the measurement of which entails invasive lumbar puncture procedures. This limits the ability to easily confirm the underlying cause of dementia or cognitive impairment, which “obviously has important prognostic and therapeutic implications,” said Dr. Boxer.
Having a plasma biomarker, especially for tau, would be extremely useful. Patients with increased tau in the brain tend to exhibit Alzheimer’s disease symptoms while those with amyloid plaques do not always have clear signs, at least not immediately. “We think that P-tau is probably a better measure because it is much more closely related to symptoms of disease,” said Dr. Boxer.
Earlier this year, he and colleagues published a study in Nature Medicine showing that P-tau181 is more than three times as high in individuals with Alzheimer’s disease compared with healthy elderly people. It also differentiated Alzheimer’s disease from frontotemporal dementia (FTD). “We found that P-tau 181 was almost as good as a PET scan or lumbar puncture at identifying individuals with Alzheimer’s disease pathology in the brain,” said Dr. Boxer.
They next wanted to assess how well P-tau 217 held up as a possible biomarker.
The new retrospective study was composed of 210 participants: 37 who acted as healthy controls, 99 who had FTLD, 39 who had Alzheimer’s disease, and 35 who had mild cognitive impairment.
More accurate test
Results showed that plasma P-tau217 was increased 5.7-fold in the participants with Alzheimer’s disease compared with the healthy controls group, and increased fivefold compared with those who had FTLD (both comparisons, P < .001).
The increase in plasma P-tau181 was lower. It was increased only 4.5-times in participants with Alzheimer’s disease compared with the healthy controls and 3.8-times relative to those with FTLD (both, P < .001). In addition, P-tau217 was potentially superior in predicting whether a person had a tau positive FTP-PET brain scan.
“This newer P-tau 217 test produces very similar results to the previous test we published [on P-tau181], but might be incrementally better or slightly more accurate, and even more closely related to the signal you get with a tau PET scan,” Dr. Boxer said.
The researchers are now examining these issues in a larger group of participants (N = 617). Results for those analyses are expected to be published soon. In addition to tau and amyloid markers, the researchers are examining another potential biomarker of neurodegeneration: the triple protein neurofilament light chain.
It’s too early to say which biomarker or biomarkers will prove to be the most useful in diagnosing Alzheimer’s disease, Dr. Boxer noted. “It’s an open question whether it will be necessary to measure multiple P-taus plus beta amyloid plus neurofilament, or maybe just measuring one P-tau level will be sufficient,” he said.
Upcoming therapy?
Having a test that verifies Alzheimer’s disease is becoming all the more important now that a therapy might soon be available. Massachusetts-based biotech company Biogen has submitted aducanumab, a monoclonal antibody that targets amyloid-beta (Abeta), to the Food and Drug Administration for approval. Should that move forward, aducanumab would be the first disease-modifying therapy for Alzheimer’s disease.
“If that’s the case, it will be even more important to have simple ways to screen people, to see if they might eventually be eligible for treatment,” said Dr. Boxer. Even if the drug isn’t approved, many patients simply want to know what is causing their cognitive problems, he added. Knowing they have Alzheimer’s disease might impact their life planning. If they have mild symptoms, interventions such as exercise and reducing cardiovascular risk could improve their overall health and quality of life, he said.
If individuals have another type of dementia, such as FTLD, that, too, might determine a different approach. Some forms of FTLD are caused by “completely different biological processes,” which are now being studied, Dr. Boxer said. So knowing that patients have this condition would allow them to participate in relevant clinical trials.
Exciting aspect
Having a tau blood test will also help those in underserviced and minority communities who can’t easily access memory specialists, Dr. Boxer noted. “It might allow them to access care, and get help much more easily, and that is a really exciting aspect of this new technology,” he said. It’s not clear when such blood tests will be on the market, although many companies are “scrambling” to make them available, said Dr. Boxer.
P-tau217 also holds promise as a marker for early Alzheimer’s disease pathology, according to another study presented at AAIC 2020. A Swedish research team measured P-tau217 in more than 1,000 participants, including those who were unimpaired and those with mild cognitive impairment, Alzheimer’s disease dementia, or non-Alzheimer’s disease neurodegenerative diseases.
Results showed that plasma P-tau217 levels increase in early stages of Alzheimer’s disease when insoluble tau aggregates are not yet detectable with PET. They also predict subsequent increases in tau-PET, as well as conversion to Alzheimer’s disease dementia.
‘Incredible breakthrough’
Commenting on the research, Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation, called the study amazing and “an incredible breakthrough.
“Researchers are able to detect disease up to 20 years before symptoms. The blood test has very good characteristics in terms of sensitivity and specificity. It correlates with the spinal fluid, it’s better than the PET imaging, it correlates with the amyloid test, and the results are being confirmed in many different cohorts,” said Dr. Fillit, who was not involved with the research.
A tau blood test, especially for P-tau 217, has the potential to be as important to determining dementia risk as cholesterol is to gauging heart disease risk, he added.
Having a tau blood test will “make our clinical trials much more precise and more efficient and reduce costs tremendously,” Dr. Fillit said, adding that he thinks tau blood tests might come to market as early as within a year.
Also commenting on the research, Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, said the new studies illustrate the rapid progress being made “in the blood biomarker space.”
Even 5 years ago, researchers would “never have thought” that blood biomarkers could be used as a tool to detect brain changes related to Alzheimer’s disease, said Dr. Edelmayer.
These new studies are “filling a gap in our understanding around tau” in Alzheimer’s disease and other neurodegenerative diseases, she said. “Being able to distinguish between diseases is going to be very, very crucial for clinicians in the future,” she added.
Dr. Edelmayer foresees that in the future there will be a panel of blood biomarkers in addition to imaging tests to help clinicians make an accurate diagnosis.
The study was supported by the National Institutes of Health and the Tau Research Consortium. Dr. Boxer disclosed that the blood p-tau test was done as part of a research collaboration between UCSF and Eli Lilly. Dr. Fillit and Dr. Edelmayer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
.
In one new development, experts at the University of California, San Francisco (UCSF) compared phosphorylated-tau181 (P-tau181) to a related form of tau called P-tau217 to determine which can best identify individuals with Alzheimer’s disease.
Results showed that the two biomarkers were similar overall, but P-tau 217 had a slight edge in terms of accuracy. Importantly, both tau isoforms distinguished frontotemporal lobar degeneration (FTLD).
“These new blood tests for P-tau are going to be really exciting because they will improve our ability to simply and inexpensively assess whether someone is at high risk for having Alzheimer’s disease,” said study author Adam L. Boxer, MD, PhD, professor in UCSF’s department of neurology.
With the approval of the first disease-modifying therapy for Alzheimer’s disease possibly around the corner, developing an accurate diagnostic blood test for this condition is even more urgent, added Dr. Boxer, who is also director of UCSF’s Neurosciences Clinical Research Unit and AD and FTD Clinical Trials Program.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Important implications
Currently, the only approved Alzheimer’s disease biomarkers are expensive positron emission tomography (PET) scans using agents that detect tau or amyloid, another hallmark Alzheimer’s disease protein, and cerebrospinal fluid levels of amyloid and tau, the measurement of which entails invasive lumbar puncture procedures. This limits the ability to easily confirm the underlying cause of dementia or cognitive impairment, which “obviously has important prognostic and therapeutic implications,” said Dr. Boxer.
Having a plasma biomarker, especially for tau, would be extremely useful. Patients with increased tau in the brain tend to exhibit Alzheimer’s disease symptoms while those with amyloid plaques do not always have clear signs, at least not immediately. “We think that P-tau is probably a better measure because it is much more closely related to symptoms of disease,” said Dr. Boxer.
Earlier this year, he and colleagues published a study in Nature Medicine showing that P-tau181 is more than three times as high in individuals with Alzheimer’s disease compared with healthy elderly people. It also differentiated Alzheimer’s disease from frontotemporal dementia (FTD). “We found that P-tau 181 was almost as good as a PET scan or lumbar puncture at identifying individuals with Alzheimer’s disease pathology in the brain,” said Dr. Boxer.
They next wanted to assess how well P-tau 217 held up as a possible biomarker.
The new retrospective study was composed of 210 participants: 37 who acted as healthy controls, 99 who had FTLD, 39 who had Alzheimer’s disease, and 35 who had mild cognitive impairment.
More accurate test
Results showed that plasma P-tau217 was increased 5.7-fold in the participants with Alzheimer’s disease compared with the healthy controls group, and increased fivefold compared with those who had FTLD (both comparisons, P < .001).
The increase in plasma P-tau181 was lower. It was increased only 4.5-times in participants with Alzheimer’s disease compared with the healthy controls and 3.8-times relative to those with FTLD (both, P < .001). In addition, P-tau217 was potentially superior in predicting whether a person had a tau positive FTP-PET brain scan.
“This newer P-tau 217 test produces very similar results to the previous test we published [on P-tau181], but might be incrementally better or slightly more accurate, and even more closely related to the signal you get with a tau PET scan,” Dr. Boxer said.
The researchers are now examining these issues in a larger group of participants (N = 617). Results for those analyses are expected to be published soon. In addition to tau and amyloid markers, the researchers are examining another potential biomarker of neurodegeneration: the triple protein neurofilament light chain.
It’s too early to say which biomarker or biomarkers will prove to be the most useful in diagnosing Alzheimer’s disease, Dr. Boxer noted. “It’s an open question whether it will be necessary to measure multiple P-taus plus beta amyloid plus neurofilament, or maybe just measuring one P-tau level will be sufficient,” he said.
Upcoming therapy?
Having a test that verifies Alzheimer’s disease is becoming all the more important now that a therapy might soon be available. Massachusetts-based biotech company Biogen has submitted aducanumab, a monoclonal antibody that targets amyloid-beta (Abeta), to the Food and Drug Administration for approval. Should that move forward, aducanumab would be the first disease-modifying therapy for Alzheimer’s disease.
“If that’s the case, it will be even more important to have simple ways to screen people, to see if they might eventually be eligible for treatment,” said Dr. Boxer. Even if the drug isn’t approved, many patients simply want to know what is causing their cognitive problems, he added. Knowing they have Alzheimer’s disease might impact their life planning. If they have mild symptoms, interventions such as exercise and reducing cardiovascular risk could improve their overall health and quality of life, he said.
If individuals have another type of dementia, such as FTLD, that, too, might determine a different approach. Some forms of FTLD are caused by “completely different biological processes,” which are now being studied, Dr. Boxer said. So knowing that patients have this condition would allow them to participate in relevant clinical trials.
Exciting aspect
Having a tau blood test will also help those in underserviced and minority communities who can’t easily access memory specialists, Dr. Boxer noted. “It might allow them to access care, and get help much more easily, and that is a really exciting aspect of this new technology,” he said. It’s not clear when such blood tests will be on the market, although many companies are “scrambling” to make them available, said Dr. Boxer.
P-tau217 also holds promise as a marker for early Alzheimer’s disease pathology, according to another study presented at AAIC 2020. A Swedish research team measured P-tau217 in more than 1,000 participants, including those who were unimpaired and those with mild cognitive impairment, Alzheimer’s disease dementia, or non-Alzheimer’s disease neurodegenerative diseases.
Results showed that plasma P-tau217 levels increase in early stages of Alzheimer’s disease when insoluble tau aggregates are not yet detectable with PET. They also predict subsequent increases in tau-PET, as well as conversion to Alzheimer’s disease dementia.
‘Incredible breakthrough’
Commenting on the research, Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation, called the study amazing and “an incredible breakthrough.
“Researchers are able to detect disease up to 20 years before symptoms. The blood test has very good characteristics in terms of sensitivity and specificity. It correlates with the spinal fluid, it’s better than the PET imaging, it correlates with the amyloid test, and the results are being confirmed in many different cohorts,” said Dr. Fillit, who was not involved with the research.
A tau blood test, especially for P-tau 217, has the potential to be as important to determining dementia risk as cholesterol is to gauging heart disease risk, he added.
Having a tau blood test will “make our clinical trials much more precise and more efficient and reduce costs tremendously,” Dr. Fillit said, adding that he thinks tau blood tests might come to market as early as within a year.
Also commenting on the research, Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, said the new studies illustrate the rapid progress being made “in the blood biomarker space.”
Even 5 years ago, researchers would “never have thought” that blood biomarkers could be used as a tool to detect brain changes related to Alzheimer’s disease, said Dr. Edelmayer.
These new studies are “filling a gap in our understanding around tau” in Alzheimer’s disease and other neurodegenerative diseases, she said. “Being able to distinguish between diseases is going to be very, very crucial for clinicians in the future,” she added.
Dr. Edelmayer foresees that in the future there will be a panel of blood biomarkers in addition to imaging tests to help clinicians make an accurate diagnosis.
The study was supported by the National Institutes of Health and the Tau Research Consortium. Dr. Boxer disclosed that the blood p-tau test was done as part of a research collaboration between UCSF and Eli Lilly. Dr. Fillit and Dr. Edelmayer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
.
In one new development, experts at the University of California, San Francisco (UCSF) compared phosphorylated-tau181 (P-tau181) to a related form of tau called P-tau217 to determine which can best identify individuals with Alzheimer’s disease.
Results showed that the two biomarkers were similar overall, but P-tau 217 had a slight edge in terms of accuracy. Importantly, both tau isoforms distinguished frontotemporal lobar degeneration (FTLD).
“These new blood tests for P-tau are going to be really exciting because they will improve our ability to simply and inexpensively assess whether someone is at high risk for having Alzheimer’s disease,” said study author Adam L. Boxer, MD, PhD, professor in UCSF’s department of neurology.
With the approval of the first disease-modifying therapy for Alzheimer’s disease possibly around the corner, developing an accurate diagnostic blood test for this condition is even more urgent, added Dr. Boxer, who is also director of UCSF’s Neurosciences Clinical Research Unit and AD and FTD Clinical Trials Program.
The findings were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Important implications
Currently, the only approved Alzheimer’s disease biomarkers are expensive positron emission tomography (PET) scans using agents that detect tau or amyloid, another hallmark Alzheimer’s disease protein, and cerebrospinal fluid levels of amyloid and tau, the measurement of which entails invasive lumbar puncture procedures. This limits the ability to easily confirm the underlying cause of dementia or cognitive impairment, which “obviously has important prognostic and therapeutic implications,” said Dr. Boxer.
Having a plasma biomarker, especially for tau, would be extremely useful. Patients with increased tau in the brain tend to exhibit Alzheimer’s disease symptoms while those with amyloid plaques do not always have clear signs, at least not immediately. “We think that P-tau is probably a better measure because it is much more closely related to symptoms of disease,” said Dr. Boxer.
Earlier this year, he and colleagues published a study in Nature Medicine showing that P-tau181 is more than three times as high in individuals with Alzheimer’s disease compared with healthy elderly people. It also differentiated Alzheimer’s disease from frontotemporal dementia (FTD). “We found that P-tau 181 was almost as good as a PET scan or lumbar puncture at identifying individuals with Alzheimer’s disease pathology in the brain,” said Dr. Boxer.
They next wanted to assess how well P-tau 217 held up as a possible biomarker.
The new retrospective study was composed of 210 participants: 37 who acted as healthy controls, 99 who had FTLD, 39 who had Alzheimer’s disease, and 35 who had mild cognitive impairment.
More accurate test
Results showed that plasma P-tau217 was increased 5.7-fold in the participants with Alzheimer’s disease compared with the healthy controls group, and increased fivefold compared with those who had FTLD (both comparisons, P < .001).
The increase in plasma P-tau181 was lower. It was increased only 4.5-times in participants with Alzheimer’s disease compared with the healthy controls and 3.8-times relative to those with FTLD (both, P < .001). In addition, P-tau217 was potentially superior in predicting whether a person had a tau positive FTP-PET brain scan.
“This newer P-tau 217 test produces very similar results to the previous test we published [on P-tau181], but might be incrementally better or slightly more accurate, and even more closely related to the signal you get with a tau PET scan,” Dr. Boxer said.
The researchers are now examining these issues in a larger group of participants (N = 617). Results for those analyses are expected to be published soon. In addition to tau and amyloid markers, the researchers are examining another potential biomarker of neurodegeneration: the triple protein neurofilament light chain.
It’s too early to say which biomarker or biomarkers will prove to be the most useful in diagnosing Alzheimer’s disease, Dr. Boxer noted. “It’s an open question whether it will be necessary to measure multiple P-taus plus beta amyloid plus neurofilament, or maybe just measuring one P-tau level will be sufficient,” he said.
Upcoming therapy?
Having a test that verifies Alzheimer’s disease is becoming all the more important now that a therapy might soon be available. Massachusetts-based biotech company Biogen has submitted aducanumab, a monoclonal antibody that targets amyloid-beta (Abeta), to the Food and Drug Administration for approval. Should that move forward, aducanumab would be the first disease-modifying therapy for Alzheimer’s disease.
“If that’s the case, it will be even more important to have simple ways to screen people, to see if they might eventually be eligible for treatment,” said Dr. Boxer. Even if the drug isn’t approved, many patients simply want to know what is causing their cognitive problems, he added. Knowing they have Alzheimer’s disease might impact their life planning. If they have mild symptoms, interventions such as exercise and reducing cardiovascular risk could improve their overall health and quality of life, he said.
If individuals have another type of dementia, such as FTLD, that, too, might determine a different approach. Some forms of FTLD are caused by “completely different biological processes,” which are now being studied, Dr. Boxer said. So knowing that patients have this condition would allow them to participate in relevant clinical trials.
Exciting aspect
Having a tau blood test will also help those in underserviced and minority communities who can’t easily access memory specialists, Dr. Boxer noted. “It might allow them to access care, and get help much more easily, and that is a really exciting aspect of this new technology,” he said. It’s not clear when such blood tests will be on the market, although many companies are “scrambling” to make them available, said Dr. Boxer.
P-tau217 also holds promise as a marker for early Alzheimer’s disease pathology, according to another study presented at AAIC 2020. A Swedish research team measured P-tau217 in more than 1,000 participants, including those who were unimpaired and those with mild cognitive impairment, Alzheimer’s disease dementia, or non-Alzheimer’s disease neurodegenerative diseases.
Results showed that plasma P-tau217 levels increase in early stages of Alzheimer’s disease when insoluble tau aggregates are not yet detectable with PET. They also predict subsequent increases in tau-PET, as well as conversion to Alzheimer’s disease dementia.
‘Incredible breakthrough’
Commenting on the research, Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation, called the study amazing and “an incredible breakthrough.
“Researchers are able to detect disease up to 20 years before symptoms. The blood test has very good characteristics in terms of sensitivity and specificity. It correlates with the spinal fluid, it’s better than the PET imaging, it correlates with the amyloid test, and the results are being confirmed in many different cohorts,” said Dr. Fillit, who was not involved with the research.
A tau blood test, especially for P-tau 217, has the potential to be as important to determining dementia risk as cholesterol is to gauging heart disease risk, he added.
Having a tau blood test will “make our clinical trials much more precise and more efficient and reduce costs tremendously,” Dr. Fillit said, adding that he thinks tau blood tests might come to market as early as within a year.
Also commenting on the research, Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, said the new studies illustrate the rapid progress being made “in the blood biomarker space.”
Even 5 years ago, researchers would “never have thought” that blood biomarkers could be used as a tool to detect brain changes related to Alzheimer’s disease, said Dr. Edelmayer.
These new studies are “filling a gap in our understanding around tau” in Alzheimer’s disease and other neurodegenerative diseases, she said. “Being able to distinguish between diseases is going to be very, very crucial for clinicians in the future,” she added.
Dr. Edelmayer foresees that in the future there will be a panel of blood biomarkers in addition to imaging tests to help clinicians make an accurate diagnosis.
The study was supported by the National Institutes of Health and the Tau Research Consortium. Dr. Boxer disclosed that the blood p-tau test was done as part of a research collaboration between UCSF and Eli Lilly. Dr. Fillit and Dr. Edelmayer have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
Flu and pneumonia vaccination tied to lower dementia risk
In a cohort study of more than 9,000 older adults, receiving a single influenza vaccination was associated with a 17% lower prevalence of Alzheimer’s disease compared with not receiving the vaccine. In addition, for those who were vaccinated more than once over the years, there was an additional 13% reduction in Alzheimer’s disease incidence.
In another study, which included more than 5,000 older participants, being vaccinated against pneumonia between the ages of 65 and 75 reduced the risk of developing Alzheimer’s disease by 30%.
The subject of vaccines “is obviously very topical with the COVID-19 pandemic,” said Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association. “While these are very preliminary data, these studies do suggest that with vaccination against both respiratory illnesses, there is the potential to lower risk for developing cognitive decline and dementia,” said Dr. Edelmayer, who was not involved in the research.
The findings of both studies were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Lower Alzheimer’s disease prevalence
The influenza vaccine study was presented by Albert Amran, a fourth-year medical student at McGovern Medical School at the University of Texas Health Science Center at Houston. The researchers used electronic health record data to create a propensity-matched cohort of 9,066 vaccinated and unvaccinated adults ages 60 and older.
Influenza vaccination, increased frequency of administration, and younger age at time of vaccination were all associated with reduced incidence of Alzheimer’s disease, Mr. Amran reported.
Being vaccinated for influenza was significantly linked to a lower prevalence of Alzheimer’s disease (odds ratio [OR], 0.83; P < .0001) in comparison with not being vaccinated. Receiving more than one vaccination over the years was associated with an additional reduction in AD incidence (OR, 0.87; P = .0342). The protection appeared to be strongest for those who received their first vaccination at a younger age, for example, at age 60 versus 70.
Mr. Amran and research colleagues have two theories as to why influenza vaccination may protect the brain.
One is that vaccination may aid the immune system as people age. “As people get older, their immune systems become less able to control infection. We’ve seen this with the ongoing pandemic, with older people at much higher risk for dying. Giving people the vaccine once a year may help keep the immune system in shape,” Mr. Amran said.
Another theory is that the prevention of influenza itself may be relevant. “Flu infections can be extremely deadly in older patients. Maybe the results of our study will give another reason for people to get vaccinated,” Mr. Amran said.
Pneumonia vaccine
The other study was presented by Svetlana Ukraintseva, PhD, of Duke University, Durham, N.C.
Dr. Ukraintseva and colleagues investigated associations between pneumococcal vaccine, with and without an accompanying influenza vaccine, and the risk for Alzheimer’s disease among 5,146 participants in the Cardiovascular Health Study. Covariates included sex, race, birth cohort, education, smoking, and a known genetic risk factor for Alzheimer’s disease: the rs2075650 G allele in the TOMM40 gene.
In a logistic model with all covariates, vaccination against pneumonia between ages 65 and 75 was significantly associated with reduced risk of developing AD (OR, 0.70; P < .04). The largest reduction in Alzheimer’s disease risk (OR, 0.62; P < .04) was among those vaccinated against pneumonia who were noncarriers of the rs2075650 G allele.
Total number of vaccinations against pneumonia and influenza between ages 65 and 75 was also associated with a lower risk for Alzheimer’s disease (OR, 0.88; P < .01). However, the effect was not evident for the influenza vaccination alone.
“The fact that very different pathogens – viral, bacterial, fungal – have been linked to Alzheimer’s disease indicates a possibility that compromised host immunity may play a role in Alzheimer’s disease through increasing overall brain’s vulnerability to various microbes,” said Dr. Ukraintseva.
The current findings support further investigation of pneumococcal vaccine as a “reasonable candidate for repurposing in personalized AD prevention,” she noted. “These results also support the important role of boosting overall immune robustness/resilience in preventing Alzheimer’s disease,” Dr. Ukraintseva added.
Her group is currently working on confirming the findings in another population.
Brain protective?
“Neither study can prove that the benefit is directly related to the vaccine itself, but what they can indicate is that potentially, vaccines are a way to protect your health and brain,” Dr. Edelmayer said.
In a statement, Maria Carrillo, PhD, chief science officer for the Alzheimer’s Association, noted that more research is needed.
The new data call “for further studies in large, diverse clinical trials to inform whether vaccinations as a public health strategy decrease our risk for developing dementia as we age,” Dr. Carillo said.
Funding for the influenza vaccine study was provided by the Christopher Sarofim Family Professorship in Biomedical Informatics and Bioengineering, a UT STARs Award, the Cancer Prevention and Research Institute of Texas, and the National Institutes of Health. Funding for the pneumonia study was provided by the National Institute on Aging. Dr. Amran, Dr. Ukraintseva, Dr. Edelmayer, and Dr. Carrillo have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In a cohort study of more than 9,000 older adults, receiving a single influenza vaccination was associated with a 17% lower prevalence of Alzheimer’s disease compared with not receiving the vaccine. In addition, for those who were vaccinated more than once over the years, there was an additional 13% reduction in Alzheimer’s disease incidence.
In another study, which included more than 5,000 older participants, being vaccinated against pneumonia between the ages of 65 and 75 reduced the risk of developing Alzheimer’s disease by 30%.
The subject of vaccines “is obviously very topical with the COVID-19 pandemic,” said Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association. “While these are very preliminary data, these studies do suggest that with vaccination against both respiratory illnesses, there is the potential to lower risk for developing cognitive decline and dementia,” said Dr. Edelmayer, who was not involved in the research.
The findings of both studies were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Lower Alzheimer’s disease prevalence
The influenza vaccine study was presented by Albert Amran, a fourth-year medical student at McGovern Medical School at the University of Texas Health Science Center at Houston. The researchers used electronic health record data to create a propensity-matched cohort of 9,066 vaccinated and unvaccinated adults ages 60 and older.
Influenza vaccination, increased frequency of administration, and younger age at time of vaccination were all associated with reduced incidence of Alzheimer’s disease, Mr. Amran reported.
Being vaccinated for influenza was significantly linked to a lower prevalence of Alzheimer’s disease (odds ratio [OR], 0.83; P < .0001) in comparison with not being vaccinated. Receiving more than one vaccination over the years was associated with an additional reduction in AD incidence (OR, 0.87; P = .0342). The protection appeared to be strongest for those who received their first vaccination at a younger age, for example, at age 60 versus 70.
Mr. Amran and research colleagues have two theories as to why influenza vaccination may protect the brain.
One is that vaccination may aid the immune system as people age. “As people get older, their immune systems become less able to control infection. We’ve seen this with the ongoing pandemic, with older people at much higher risk for dying. Giving people the vaccine once a year may help keep the immune system in shape,” Mr. Amran said.
Another theory is that the prevention of influenza itself may be relevant. “Flu infections can be extremely deadly in older patients. Maybe the results of our study will give another reason for people to get vaccinated,” Mr. Amran said.
Pneumonia vaccine
The other study was presented by Svetlana Ukraintseva, PhD, of Duke University, Durham, N.C.
Dr. Ukraintseva and colleagues investigated associations between pneumococcal vaccine, with and without an accompanying influenza vaccine, and the risk for Alzheimer’s disease among 5,146 participants in the Cardiovascular Health Study. Covariates included sex, race, birth cohort, education, smoking, and a known genetic risk factor for Alzheimer’s disease: the rs2075650 G allele in the TOMM40 gene.
In a logistic model with all covariates, vaccination against pneumonia between ages 65 and 75 was significantly associated with reduced risk of developing AD (OR, 0.70; P < .04). The largest reduction in Alzheimer’s disease risk (OR, 0.62; P < .04) was among those vaccinated against pneumonia who were noncarriers of the rs2075650 G allele.
Total number of vaccinations against pneumonia and influenza between ages 65 and 75 was also associated with a lower risk for Alzheimer’s disease (OR, 0.88; P < .01). However, the effect was not evident for the influenza vaccination alone.
“The fact that very different pathogens – viral, bacterial, fungal – have been linked to Alzheimer’s disease indicates a possibility that compromised host immunity may play a role in Alzheimer’s disease through increasing overall brain’s vulnerability to various microbes,” said Dr. Ukraintseva.
The current findings support further investigation of pneumococcal vaccine as a “reasonable candidate for repurposing in personalized AD prevention,” she noted. “These results also support the important role of boosting overall immune robustness/resilience in preventing Alzheimer’s disease,” Dr. Ukraintseva added.
Her group is currently working on confirming the findings in another population.
Brain protective?
“Neither study can prove that the benefit is directly related to the vaccine itself, but what they can indicate is that potentially, vaccines are a way to protect your health and brain,” Dr. Edelmayer said.
In a statement, Maria Carrillo, PhD, chief science officer for the Alzheimer’s Association, noted that more research is needed.
The new data call “for further studies in large, diverse clinical trials to inform whether vaccinations as a public health strategy decrease our risk for developing dementia as we age,” Dr. Carillo said.
Funding for the influenza vaccine study was provided by the Christopher Sarofim Family Professorship in Biomedical Informatics and Bioengineering, a UT STARs Award, the Cancer Prevention and Research Institute of Texas, and the National Institutes of Health. Funding for the pneumonia study was provided by the National Institute on Aging. Dr. Amran, Dr. Ukraintseva, Dr. Edelmayer, and Dr. Carrillo have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In a cohort study of more than 9,000 older adults, receiving a single influenza vaccination was associated with a 17% lower prevalence of Alzheimer’s disease compared with not receiving the vaccine. In addition, for those who were vaccinated more than once over the years, there was an additional 13% reduction in Alzheimer’s disease incidence.
In another study, which included more than 5,000 older participants, being vaccinated against pneumonia between the ages of 65 and 75 reduced the risk of developing Alzheimer’s disease by 30%.
The subject of vaccines “is obviously very topical with the COVID-19 pandemic,” said Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association. “While these are very preliminary data, these studies do suggest that with vaccination against both respiratory illnesses, there is the potential to lower risk for developing cognitive decline and dementia,” said Dr. Edelmayer, who was not involved in the research.
The findings of both studies were presented at the virtual annual meeting of the Alzheimer’s Association International Conference.
Lower Alzheimer’s disease prevalence
The influenza vaccine study was presented by Albert Amran, a fourth-year medical student at McGovern Medical School at the University of Texas Health Science Center at Houston. The researchers used electronic health record data to create a propensity-matched cohort of 9,066 vaccinated and unvaccinated adults ages 60 and older.
Influenza vaccination, increased frequency of administration, and younger age at time of vaccination were all associated with reduced incidence of Alzheimer’s disease, Mr. Amran reported.
Being vaccinated for influenza was significantly linked to a lower prevalence of Alzheimer’s disease (odds ratio [OR], 0.83; P < .0001) in comparison with not being vaccinated. Receiving more than one vaccination over the years was associated with an additional reduction in AD incidence (OR, 0.87; P = .0342). The protection appeared to be strongest for those who received their first vaccination at a younger age, for example, at age 60 versus 70.
Mr. Amran and research colleagues have two theories as to why influenza vaccination may protect the brain.
One is that vaccination may aid the immune system as people age. “As people get older, their immune systems become less able to control infection. We’ve seen this with the ongoing pandemic, with older people at much higher risk for dying. Giving people the vaccine once a year may help keep the immune system in shape,” Mr. Amran said.
Another theory is that the prevention of influenza itself may be relevant. “Flu infections can be extremely deadly in older patients. Maybe the results of our study will give another reason for people to get vaccinated,” Mr. Amran said.
Pneumonia vaccine
The other study was presented by Svetlana Ukraintseva, PhD, of Duke University, Durham, N.C.
Dr. Ukraintseva and colleagues investigated associations between pneumococcal vaccine, with and without an accompanying influenza vaccine, and the risk for Alzheimer’s disease among 5,146 participants in the Cardiovascular Health Study. Covariates included sex, race, birth cohort, education, smoking, and a known genetic risk factor for Alzheimer’s disease: the rs2075650 G allele in the TOMM40 gene.
In a logistic model with all covariates, vaccination against pneumonia between ages 65 and 75 was significantly associated with reduced risk of developing AD (OR, 0.70; P < .04). The largest reduction in Alzheimer’s disease risk (OR, 0.62; P < .04) was among those vaccinated against pneumonia who were noncarriers of the rs2075650 G allele.
Total number of vaccinations against pneumonia and influenza between ages 65 and 75 was also associated with a lower risk for Alzheimer’s disease (OR, 0.88; P < .01). However, the effect was not evident for the influenza vaccination alone.
“The fact that very different pathogens – viral, bacterial, fungal – have been linked to Alzheimer’s disease indicates a possibility that compromised host immunity may play a role in Alzheimer’s disease through increasing overall brain’s vulnerability to various microbes,” said Dr. Ukraintseva.
The current findings support further investigation of pneumococcal vaccine as a “reasonable candidate for repurposing in personalized AD prevention,” she noted. “These results also support the important role of boosting overall immune robustness/resilience in preventing Alzheimer’s disease,” Dr. Ukraintseva added.
Her group is currently working on confirming the findings in another population.
Brain protective?
“Neither study can prove that the benefit is directly related to the vaccine itself, but what they can indicate is that potentially, vaccines are a way to protect your health and brain,” Dr. Edelmayer said.
In a statement, Maria Carrillo, PhD, chief science officer for the Alzheimer’s Association, noted that more research is needed.
The new data call “for further studies in large, diverse clinical trials to inform whether vaccinations as a public health strategy decrease our risk for developing dementia as we age,” Dr. Carillo said.
Funding for the influenza vaccine study was provided by the Christopher Sarofim Family Professorship in Biomedical Informatics and Bioengineering, a UT STARs Award, the Cancer Prevention and Research Institute of Texas, and the National Institutes of Health. Funding for the pneumonia study was provided by the National Institute on Aging. Dr. Amran, Dr. Ukraintseva, Dr. Edelmayer, and Dr. Carrillo have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
A Multidisciplinary Ambulation Protocol to Reduce Postoperative Venous Thromboembolism After Colorectal Surgery
From the Department of Surgery, Washington University School of Medicine, St. Louis, MO.
Abstract
Background: Patients undergoing colorectal surgery are at high risk for postoperative venous thromboembolism (VTE). Early ambulation has been encouraged to lower rates of VTE, but evidence demonstrating its effectiveness outside of a bundle is limited.
Objective: To create a multidisciplinary ambulation protocol in an effort to reduce postoperative VTE.
Methods: A single-center, retrospective, comparative study of patients who underwent colectomy or proctectomy was conducted. Outcomes of patients operated on prior to protocol implementation were compared with a cohort after implementation. The intervention studied was the implementation of a multidisciplinary ambulation protocol. The primary endpoint was postoperative VTE.
Results: There was no difference between the pre-intervention group (n = 1762) and the postintervention group (n = 253) in terms of sex, race, origin, emergency status, operative time, and the majority of medical comorbidities (with the exception of smoking status and congestive heart failure). After the protocol was implemented, ambulation rates on postoperative days 0, 1, and 2 improved from 36.4%, 47.3%, and 50.2% to 36.8%, 74.7%, and 82.6%, respectively The VTE rate in the pre-intervention group was 2.7% versus a rate of 0.4% in the postintervention group (P = 0.02).
Conclusion: Creation of an ambulation protocol is associated with a significant reduction in VTE. Commitment from patients, families, nurses, physician extenders, and physicians is critical to the success of the program.
Keywords: VTE; pulmonary embolism; deep vein thrombosis; postoperative; quality improvement.
Postoperative venous thromboembolism (VTE) is a significant source of morbidity, mortality, and cost.1,2 Colorectal surgery patients are at particularly high risk for VTE due to positioning during surgery, pelvic dissection, and other conditions often found in these patients, such as cancer and inflammatory bowel disease.3 A National Surgical Quality Improvement Program (NSQIP) analysis demonstrated an overall rate of VTE in colorectal surgery patients of 2.4%, although other studies have demonstrated rates up to 9%, even in those receiving appropriate chemoprophylaxis.4-6 Many of these VTEs occur in the postdischarge setting. In a NSQIP study of colorectal surgery patients, the rate of VTE between discharge and 30 days was 0.47%.7 The cost burdenfor a postoperative VTE has been estimated to be more than $18,000.8
Studies from NSQIP have identified multiple factors associated with VTE in colorectal surgery patients, but NSQIP does not record ambulation as a standard variable.9 Multiple strategies have been implemented to reduce postoperative VTE. Often, these studies focus on increasing compliance with appropriate chemoprophylaxis, risk stratification, or bundling multiple strategies.10,11 However, despite the fact that postsurgical ambulation is widely encouraged and recommended by the American Society of Colon and Rectal Surgeons clinical practice guidelines, there is little evidence demonstrating the role of ambulation alone in the reduction of VTE.4,12 The purpose of this study was to create a multidisciplinary protocol to increase postoperative ambulation and evaluate its effect on VTE.
Methods
Setting
This study was conducted at a single academic tertiary care center.
Patients and Outcome Measures
All patients undergoing colectomy or proctectomy by surgeons in the section of colon and rectal surgery at a single institution between January 2011 and March 2017 were included. Colectomy and proctectomy were defined by CPT codes 44140, 44141, 44143, 44144, 44145, 44146, 44147, 44150, 44151, 44155, 44156, 44157, 44158, 44160, 44204, 44205, 44206, 44207, 44208, 44210, 44211, 44212, 44213, 45110, 45111, 45112, 45113, 45114, 45116, 45119, 45120, 45121, 45123, 45126, 45160, 45395, and 45397. The primary outcome of VTE within 30 days, including deep venous thrombosis (DVT) and pulmonary embolism (PE), was measured using institution-specific data from NSQIP in both the pre-intervention and postintervention setting. The occurrence of both DVT and PE in 1 patient was counted as a single event of VTE. Ambulation rate on postoperative day (POD) 0, 1, and 2 was calculated by NSQIP in the pre-intervention setting (our institution-specific NSQIP recorded ambulation data for an unrelated project) and by review of the electronic health record in the postintervention setting, as this institution-specific variable was no longer being collected. Ambulation was defined as getting out of bed and taking at least 1 step. The threshold for ambulating each day was once on POD 0 and twice on PODs 1 and 2. Patients with missing ambulation data were excluded from the analysis. Both prior to and throughout the intervention, all patients were given VTE chemoprophylaxis with either low-dose unfractionated heparin or low-molecular-weight heparin prior to induction of anesthesia, with chemoprophylaxis extending an additional 21 days after discharge (unless specifically contraindicated); sequential compression devices; and standard orders to ambulate 3 times daily from POD 0 as part of the standard Enhanced Recovery After Surgery protocol.
Analysis
Statistical analysis was performed using univariate analysis. Chi-square test and univariate logistic regression were used to determine the association between ambulation rates and VTE in the pre-intervention group. Chi-square test was also used to compare ambulation and VTE rates between the pre-intervention and postintervention groups. Plan-Do-Study-Act (PDSA) cycle fidelity (the degree to which a PDSA cycle is carried out in accordance with the guiding principles of its use) was measured by recording the ambulation rates both before and after the intervention.13 Statistical analysis was performed using SAS Version 9.4 (SAS Institute, Cary, NC). This study was reviewed by the Washington University School of Medicine Institutional Review Board and deemed to be quality improvement, not human subjects research, and therefore did not require formal approval.
Baseline Outcome Rates
A total of 1762 patients were identified during the pre-intervention period. The overall VTE rate in the pre-intervention group was 2.7% (n = 48), with 39 DVTs (2.2%) and 13 PEs (0.7%). Pre-intervention ambulation data were available on 590 patients. Baseline ambulation rates on PODs 0, 1, and 2 were 36.4% (213/590), 47.3% (279/590), and 50.2% (296/590), respectively. Patients who did not ambulate on POD 0 had a VTE rate of 4.3%, as compared to 0.9% in those who did ambulate (Table 1). Patients who did not ambulate twice on POD 1 had a VTE rate of 4.8%, compared to 1.1% in those who did ambulate (odds ratio [OR], 4.66; 95% confidence interval [CI], 1.34 to 16.28). Patients who did not ambulate twice on POD 2 had a VTE rate of 5.4%, compared to 0.7% in those who did. Finally, those who ambulated twice on both PODs 1 and 2 had a 0% rate of VTE, compared to 4.9% in those who did not ambulate on both PODs.
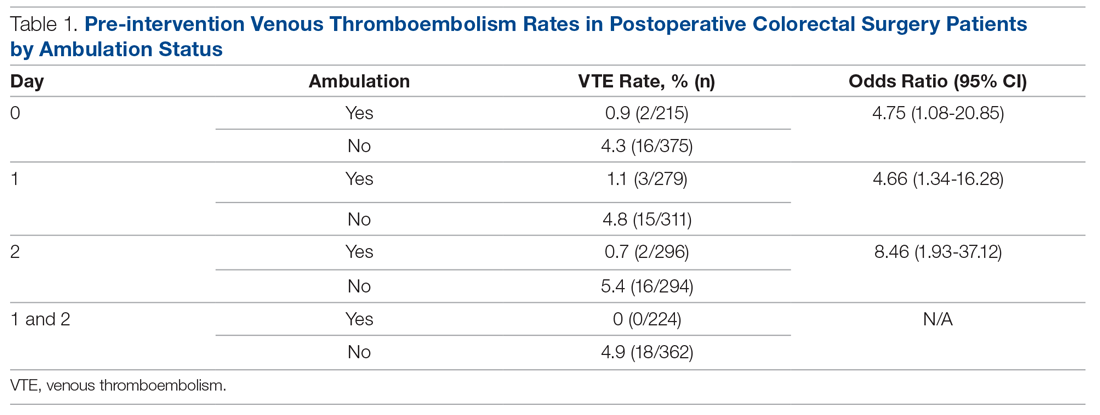
Ambulation Protocol
After baseline outcome rates had been established, a multidisciplinary team of medical assistants, nurses, nurse practitioners, and physicians worked together to identify all processes that involved postoperative ambulation. Given the significant differences in VTE rates between patients who ambulated and those that did not, we created a multidisciplinary ambulation protocol using the PDSA method.14 Multiple points of patient contact were chosen for intervention, and the ambulation protocol was implemented in June 2018 and continued for 7 months.
Patients were observed from their initial office visit with a surgeon, during the preoperative education encounter, and in the operating room and on the surgical ward until discharge. Representatives from multiple disciplines who encountered patients at various times in the process, including medical assistants, patient care technicians, nurses, nurse practitioners, physical therapists, and physicians, participated in a kick-off meeting to identify difficulties they encounter when encouraging patient ambulation. The following 4 areas were identified.
Barriers to Patient Ambulation
Patient Expectations. Patients did not appear to have a clear expectation of what their ambulation goals were postoperatively, despite the fact that each patient is given an operative pathway booklet that includes their goals for each day, including ambulation. The consensus was that patients were overwhelmed with the amount of information and, oftentimes, the severity of their diagnosis, so the information regarding ambulation was not retained. Nurses commented that patients frequently stated that they did not think their surgeon wanted them to get out of bed postoperatively.
Electronic Orders. There was confusion within the nursing staff regarding orders in the electronic health record compared to physician expectations. Orders stated patients should ambulate 3 times daily, but did not specify on which postoperative day this should start. Often, nursing verbal sign-out from the post-anesthesia care unit (PACU) would be an order for bedrest, despite no clear origin of this order. This created confusion among the nursing staff as to what the appropriate ambulation orders should be.
Nursing Workflow. The initial state of the nursing workflow was not conducive to evaluating for, or assisting with, ambulation. With no set time to assist and evaluate patients for ambulation, it turned into a task nurses needed to accomplish when they had extra time. With increasing demands of charting in the electronic health record, nurses often had to skip ambulation in order to accomplish other tasks.
Family Expectations. In addition to patient expectations, family members often had expectations that were not congruent with the planned postoperative course. Nurses stated family members would often tell them that they did not feel that their family member should be ambulating so soon after surgery. Often these family members had not attended preoperative education sessions with the patient. This was compounded by the uncertainty among the nursing staff regarding what exactly the ambulation orders were.
Interventions
Targeted interventions were created to address these 4 barriers to ambulation identified by staff.
Preoperative Education. Although all elective patients received a printed operative pathway booklet describing daily goals, including ambulation, patients still did not have a sufficient understanding of what was expected of them. The education session was modified to increase the time spent on both the expectation for and the rationale behind ambulation. That section of the education session ended with a verbal commitment and read-back of the expectations for ambulation by the patient.
Clarification of Electronic Orders. Postoperative orders within the colorectal standard pathway were changed, including specific time frames and frequency, to match the information provided in the patient education booklet. These orders were for ambulation within 4 hours of arrival to the floor, and the orders also noted that no patient should be on bedrest unless explicitly stated. From POD 1, all patients were to ambulate at least twice daily for the remainder of the hospital stay (patients were encouraged to walk 4 times daily, but we set a minimum expectation of twice daily for the order set). These orders were clarified with in-person meetings with the nursing staff and leadership from the PACU and the colorectal surgical ward.
Adjusted Nursing Workflow. Nurses were interviewed and asked to create a plan regarding how they could better incorporate ambulation into their daily workflow. Ambulation assessment was incorporated into the twice-per-shift recording of vital signs and patient safety assessment. This was recorded into the electronic health record at the same time as the patients’ vital signs. This allowed nurses to keep track of which patients would need extra assistance in ambulation and which patients were doing well on their own with the assistance of family. It also helped focus the resources of physical therapy and the single ambulation technician on the floor and to assist patients who needed more assistance.
Creation of Ambulation Encouragement Signs. The authors discovered that despite patients being told preoperatively about ambulation expectations, friends and family are not always included in these conversations. As nurses frequently cited both patients and family as reasons patients thought they should not walk, multiple signs inviting patients to take an active role in their recovery by ambulating were created and placed around the unit. The signs outlined the expectations of being out of bed and taking at least 1 step on the day of surgery and walking at least 4 times per day thereafter. In addition, we addressed frequently asked questions around issues such as walking with intravenous poles and urinary catheters. The posters were signed by all staff colorectal surgeons.
Results
Over the course of 7 months (June 2018 to December 2018), 253 postintervention patients were identified (Table 2). There was no difference between the pre-intervention group (n = 1762) and the postintervention group in terms of sex, race, origin, emergency status, operative time, and the majority of medical comorbidities (with the exception of smoking status and congestive heart failure). The postintervention group was slightly older (60 versus 57 years) and had a higher percentage of patients with an American Society of Anesthesiologists physical status score greater than 2 (66.8% versus 51.2%). The postintervention group also had higher rates of both malignancy (53.4% versus 33.3%) and inflammatory bowel disease (18.2% versus 14.4%).
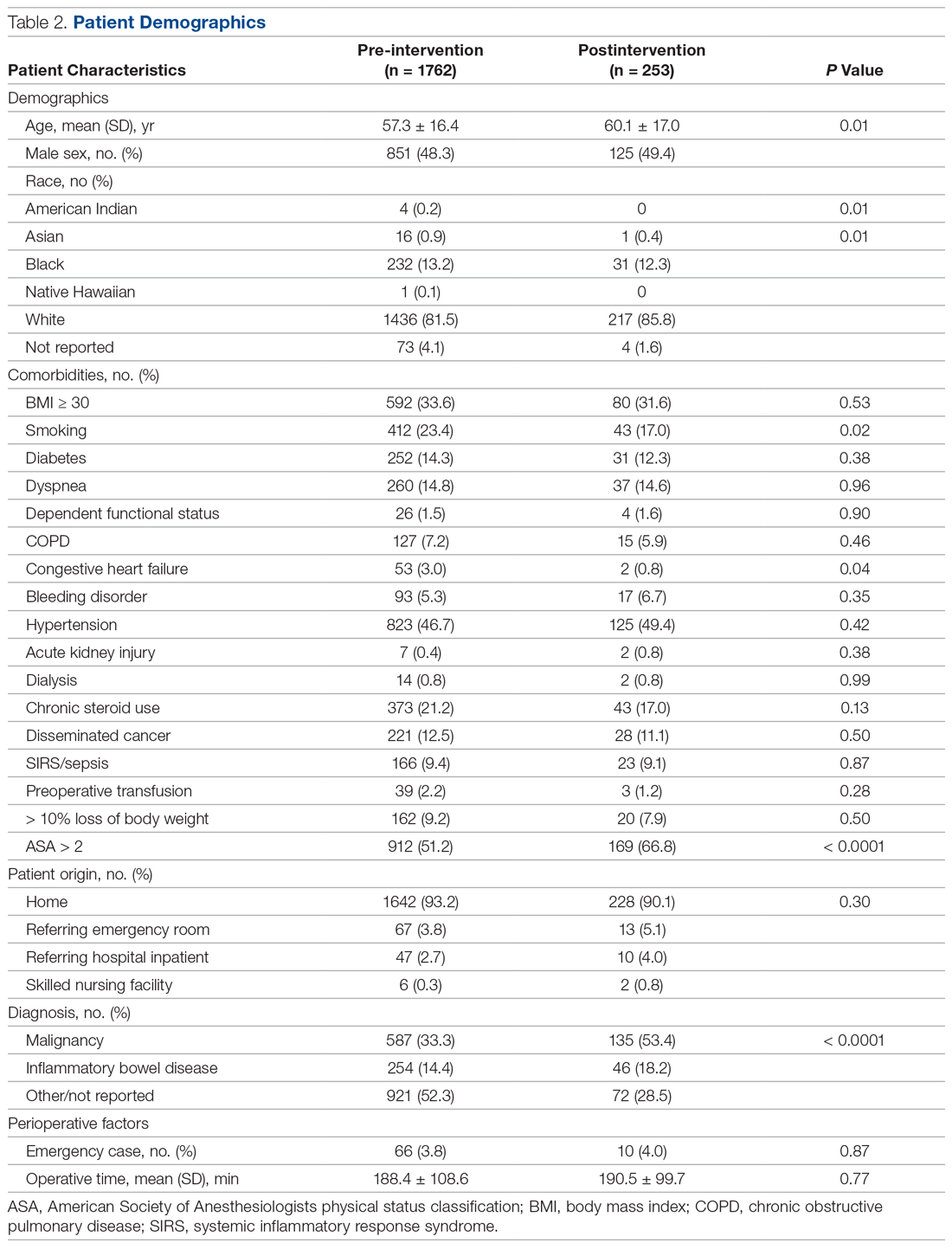
The fidelity of the PDSA cycle was measured by pre-intervention and postintervention ambulation rates. Ambulation rates on POD 0, 1, and 2 improved from 36.4%, 47.3%, and 50.2% to 36.8%, 74.7%, and 82.6%, respectively (Table 3). The VTE rate decreased from 2.7% to 0.4% (P = 0.02), with 1 DVT and 0 PEs. It should be noted that the only patient who developed a VTE postintervention did not ambulate on PODs 0, 1, or 2.
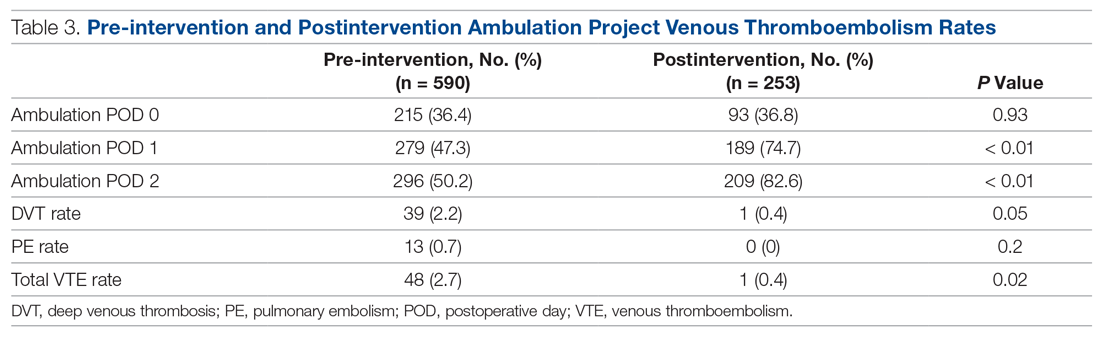
Discussion
Postoperative VTE is a severe complication for postoperative colorectal surgery patients. Previous studies have demonstrated that increasing ambulation is associated with a lower rate of overall complications, and, when incorporated into a bundle, is associated with decreased rates of VTE.11,15 However, this is the first study to our knowledge demonstrating that creation of an ambulation protocol alone is associated with a decrease in VTE.
Analysis of pre-intervention data demonstrated a strong association between ambulation and an absence of VTE. No patient who ambulated on PODs 0, 1, and 2 developed a VTE. Based on those results, we moved forward with creating the ambulation protocol. While ambulation stayed stable on POD 0, there were 60% and 65% increases on PODs 1 and 2, respectively. Nurses cited late arrival to the floor for second and third start cases as the primary difficulty in getting patients to ambulate more on POD 0.
We believe the key to the success of the ambulation protocol was its multidisciplinary nature. Certainly, the easiest way to create an ambulation protocol is to change the postoperative orders to state patients must walk 4 times per day. However, if the nursing staff is unable or unwilling to carry out these orders, the orders serve little purpose. In order to make lasting changes, all stakeholders in the process must be identified. In our case, stakeholders included surgery and nursing leadership, surgeons, nurse practitioners, nurses, medical assistants, physical therapists, patient care technicians, and patients. This is where we utilized kaizen, a core principle of Lean methodology that empowers employees at the level of the work being carried out to propose ideas for improvement.16 From the beginning of the patient experience, the health care practitioners who were carrying out each step of the process were best able to identify the problems and create solutions. In addition, stakeholders were given regular updates regarding how their efforts were increasing ambulation rates and the results at the end of the study period.
This study also demonstrates that, in a health care system increasingly focused on both quality and cost, significant improvements in quality can be made without increasing cost or resource utilization. Early in the process, it was proposed that the only way to increase the ambulation rate would be to increase the number of physical therapists, nurses, and nursing assistants. However, after identifying the root causes of the problem, the solutions had more to do with improving workflow and fixing problem areas identified by the staff.
In addition to having a positive effect on the outcome studied, collaborative projects such as this between physicians and nurses may lead to increased nursing job satisfaction. A meta-analysis of 31 studies identified nurse-physician collaboration and autonomy as 2 factors that correlate most strongly with nursing satisfaction.17 A Cochrane review also suggests that practice-based interprofessional collaboration may lead to improved health care processes and outcomes.18
This study has several limitations. Pre-intervention ambulation rates were abstracted from institution-specific NSQIP data, and missing data were excluded from analysis. Also, due to the retrospective collection of the pre-intervention data, the distance of ambulation could not be quantified. The bar for ambulation is low, as patients were only required to get out of bed and walk 1 step. However, we feel that getting out of bed and taking even 1 step is substantially better than complete bedrest. It is likely that once patients cross the threshold of taking 1 step, they are more likely to ambulate. An area of future study may be to more precisely define the relationship between the quantity of ambulation in steps and its effect on VTE. Finally, we acknowledge that while there is no direct increase in costs, implementing an ambulation protocol does take time from all who participate in the project.
Conclusion
Creation of an ambulation protocol is associated with a decrease in postoperative VTE rates in colorectal surgery patients. A multidisciplinary approach is critical to identify the underlying problems and propose effective solutions. Further studies are required to better correlate the distance of ambulation and its effect on VTE. However, this study shows that even a minimum of 1 step is associated with decreased VTE rates.
Corresponding author: Aneel Damle, MD, MBA, Colon & Rectal Surgery Associates, 3433 Broadway St. NE, Suite 115, Minneapolis, MN 55413; [email protected].
Financial disclosures: None.
1. Gangireddy C, Rectenwald JR, Upchurch GR, et al. Risk factors and clinical impact of postoperative symptomatic venous thromboembolism. J Vasc Surg. 2007;45:341-342.
2. Newhook TE, LaPar DJ, Walters DM, et al. Impact of postoperative venous thromboembolism on postoperative morbidity, mortality, and resource utilization after hepatectomy. Am Surg. 2015;81:1216-1223.
3. Bergqvist D. Venous thromboembolism: a review of risk and prevention in colorectal surgery patients. Dis Colon Rectum. 2006;49:1620-1628.
4. Fleming F, Gaertner W, Ternent CA, et al. The American society of colon and rectal surgeons clinical practice guideline for the prevention of venous thromboembolic disease in colorectal surgery. Dis Colon Rectum. 2018;61:14-20.
5. McLeod RS, Geerts WH, Sniderman KW, et al. Canadian Colorectal Surgery DVT Prophylaxis Trial investigators. Subcutaneous heparin versus low-molecular-weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian colorectal DV prophylaxis trial: a randomized, double-blind trial. Ann Surg. 2001;233:438-444.
6. Shapiro R, Vogel JD, Kiran RP. Risk of postoperative venous thromboembolism after laparoscopic and open colorectal surgery: an additional benefit of the minimally invasive approach? Dis Colon Rectum. 2011;54:1496-1502.
7. Dimick JB, Chen SL, Taheri PA, et al. Hospital costs associated with surgical complications: a report from the private-sector National Surgical Quality Improvement Program. J Am Coll Surg. 2004;199:531-537.
8. Fleming FJ, Kim MJ, Salloum RM, et al. How much do we need to worry about venous thromboembolism after hospital discharge? A study of colorectal surgery patients using the National Surgical Quality Improvement Program database. Dis Colon Rectum. 2010;53:1355-1360.
9. ACS NSQIP. User guide for the 2016 ACS NSQIP participant use data file (PUF). 2017. www.facs.org/~/media/files/quality%20programs/nsqip/nsqip_puf_userguide_2016.ashx Accessed July 10, 2020.
10. Caprini JA. Risk assessment as a guide for the prevention of the many faces of venous thromboembolism. Am J Surg. 2010;199(1 Suppl):S3-S10.
11. Cassidy MR, Rosenkranz P, McAney D. Reducing postoperative venous thromboembolism complications with a standardized risk-stratified prophylaxis protocol and mobilization protocol. J Am Coll Surg. 2014;218:1095-1104.
12. Lau BD, Streiff MB, Kraus PS, et al. No evidence to support ambulation for reducing postoperative venous thromboembolism. J Am Coll Surg. 2014;219:1101-1103.
13. McNicholas C, Lennox L, Woodcock T, et al. Evolving quality improvement support strategies to improve Plan–Do–Study–Act cycle fidelity: a retrospective mixed-methods study. BMJ Qual Saf. 2019;28:356-365.
14. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan–do–study–act method to improve quality in healthcare. BMC Qual Saf. 2014;23:290-298.
15. Nevo Y, Shaltiel T, Constantini N, et al. Effect of ambulation and physical activity on postoperative complications. J Am Coll Surg. 2016;223(Suppl 1):S61.
16. Mazzocato P, Stenfors-Hayes T, von Thiele Schwarz U, et al. Kaizen practice in healthcare: a qualitative analysis of hospital employees’ suggestions for improvement. BMJ Open. 2016;6:e012256.
17. Zangaro GA, Soeken KL. A meta-analysis of studies of nurses’ job satisfaction. Res Nursing Health. 2007;30:445-458.
18. Reeves S, Pelone F, Harrison R, et al. Interprofessional collaboration to improve professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2017;6(6):CD000072.
From the Department of Surgery, Washington University School of Medicine, St. Louis, MO.
Abstract
Background: Patients undergoing colorectal surgery are at high risk for postoperative venous thromboembolism (VTE). Early ambulation has been encouraged to lower rates of VTE, but evidence demonstrating its effectiveness outside of a bundle is limited.
Objective: To create a multidisciplinary ambulation protocol in an effort to reduce postoperative VTE.
Methods: A single-center, retrospective, comparative study of patients who underwent colectomy or proctectomy was conducted. Outcomes of patients operated on prior to protocol implementation were compared with a cohort after implementation. The intervention studied was the implementation of a multidisciplinary ambulation protocol. The primary endpoint was postoperative VTE.
Results: There was no difference between the pre-intervention group (n = 1762) and the postintervention group (n = 253) in terms of sex, race, origin, emergency status, operative time, and the majority of medical comorbidities (with the exception of smoking status and congestive heart failure). After the protocol was implemented, ambulation rates on postoperative days 0, 1, and 2 improved from 36.4%, 47.3%, and 50.2% to 36.8%, 74.7%, and 82.6%, respectively The VTE rate in the pre-intervention group was 2.7% versus a rate of 0.4% in the postintervention group (P = 0.02).
Conclusion: Creation of an ambulation protocol is associated with a significant reduction in VTE. Commitment from patients, families, nurses, physician extenders, and physicians is critical to the success of the program.
Keywords: VTE; pulmonary embolism; deep vein thrombosis; postoperative; quality improvement.
Postoperative venous thromboembolism (VTE) is a significant source of morbidity, mortality, and cost.1,2 Colorectal surgery patients are at particularly high risk for VTE due to positioning during surgery, pelvic dissection, and other conditions often found in these patients, such as cancer and inflammatory bowel disease.3 A National Surgical Quality Improvement Program (NSQIP) analysis demonstrated an overall rate of VTE in colorectal surgery patients of 2.4%, although other studies have demonstrated rates up to 9%, even in those receiving appropriate chemoprophylaxis.4-6 Many of these VTEs occur in the postdischarge setting. In a NSQIP study of colorectal surgery patients, the rate of VTE between discharge and 30 days was 0.47%.7 The cost burdenfor a postoperative VTE has been estimated to be more than $18,000.8
Studies from NSQIP have identified multiple factors associated with VTE in colorectal surgery patients, but NSQIP does not record ambulation as a standard variable.9 Multiple strategies have been implemented to reduce postoperative VTE. Often, these studies focus on increasing compliance with appropriate chemoprophylaxis, risk stratification, or bundling multiple strategies.10,11 However, despite the fact that postsurgical ambulation is widely encouraged and recommended by the American Society of Colon and Rectal Surgeons clinical practice guidelines, there is little evidence demonstrating the role of ambulation alone in the reduction of VTE.4,12 The purpose of this study was to create a multidisciplinary protocol to increase postoperative ambulation and evaluate its effect on VTE.
Methods
Setting
This study was conducted at a single academic tertiary care center.
Patients and Outcome Measures
All patients undergoing colectomy or proctectomy by surgeons in the section of colon and rectal surgery at a single institution between January 2011 and March 2017 were included. Colectomy and proctectomy were defined by CPT codes 44140, 44141, 44143, 44144, 44145, 44146, 44147, 44150, 44151, 44155, 44156, 44157, 44158, 44160, 44204, 44205, 44206, 44207, 44208, 44210, 44211, 44212, 44213, 45110, 45111, 45112, 45113, 45114, 45116, 45119, 45120, 45121, 45123, 45126, 45160, 45395, and 45397. The primary outcome of VTE within 30 days, including deep venous thrombosis (DVT) and pulmonary embolism (PE), was measured using institution-specific data from NSQIP in both the pre-intervention and postintervention setting. The occurrence of both DVT and PE in 1 patient was counted as a single event of VTE. Ambulation rate on postoperative day (POD) 0, 1, and 2 was calculated by NSQIP in the pre-intervention setting (our institution-specific NSQIP recorded ambulation data for an unrelated project) and by review of the electronic health record in the postintervention setting, as this institution-specific variable was no longer being collected. Ambulation was defined as getting out of bed and taking at least 1 step. The threshold for ambulating each day was once on POD 0 and twice on PODs 1 and 2. Patients with missing ambulation data were excluded from the analysis. Both prior to and throughout the intervention, all patients were given VTE chemoprophylaxis with either low-dose unfractionated heparin or low-molecular-weight heparin prior to induction of anesthesia, with chemoprophylaxis extending an additional 21 days after discharge (unless specifically contraindicated); sequential compression devices; and standard orders to ambulate 3 times daily from POD 0 as part of the standard Enhanced Recovery After Surgery protocol.
Analysis
Statistical analysis was performed using univariate analysis. Chi-square test and univariate logistic regression were used to determine the association between ambulation rates and VTE in the pre-intervention group. Chi-square test was also used to compare ambulation and VTE rates between the pre-intervention and postintervention groups. Plan-Do-Study-Act (PDSA) cycle fidelity (the degree to which a PDSA cycle is carried out in accordance with the guiding principles of its use) was measured by recording the ambulation rates both before and after the intervention.13 Statistical analysis was performed using SAS Version 9.4 (SAS Institute, Cary, NC). This study was reviewed by the Washington University School of Medicine Institutional Review Board and deemed to be quality improvement, not human subjects research, and therefore did not require formal approval.
Baseline Outcome Rates
A total of 1762 patients were identified during the pre-intervention period. The overall VTE rate in the pre-intervention group was 2.7% (n = 48), with 39 DVTs (2.2%) and 13 PEs (0.7%). Pre-intervention ambulation data were available on 590 patients. Baseline ambulation rates on PODs 0, 1, and 2 were 36.4% (213/590), 47.3% (279/590), and 50.2% (296/590), respectively. Patients who did not ambulate on POD 0 had a VTE rate of 4.3%, as compared to 0.9% in those who did ambulate (Table 1). Patients who did not ambulate twice on POD 1 had a VTE rate of 4.8%, compared to 1.1% in those who did ambulate (odds ratio [OR], 4.66; 95% confidence interval [CI], 1.34 to 16.28). Patients who did not ambulate twice on POD 2 had a VTE rate of 5.4%, compared to 0.7% in those who did. Finally, those who ambulated twice on both PODs 1 and 2 had a 0% rate of VTE, compared to 4.9% in those who did not ambulate on both PODs.

Ambulation Protocol
After baseline outcome rates had been established, a multidisciplinary team of medical assistants, nurses, nurse practitioners, and physicians worked together to identify all processes that involved postoperative ambulation. Given the significant differences in VTE rates between patients who ambulated and those that did not, we created a multidisciplinary ambulation protocol using the PDSA method.14 Multiple points of patient contact were chosen for intervention, and the ambulation protocol was implemented in June 2018 and continued for 7 months.
Patients were observed from their initial office visit with a surgeon, during the preoperative education encounter, and in the operating room and on the surgical ward until discharge. Representatives from multiple disciplines who encountered patients at various times in the process, including medical assistants, patient care technicians, nurses, nurse practitioners, physical therapists, and physicians, participated in a kick-off meeting to identify difficulties they encounter when encouraging patient ambulation. The following 4 areas were identified.
Barriers to Patient Ambulation
Patient Expectations. Patients did not appear to have a clear expectation of what their ambulation goals were postoperatively, despite the fact that each patient is given an operative pathway booklet that includes their goals for each day, including ambulation. The consensus was that patients were overwhelmed with the amount of information and, oftentimes, the severity of their diagnosis, so the information regarding ambulation was not retained. Nurses commented that patients frequently stated that they did not think their surgeon wanted them to get out of bed postoperatively.
Electronic Orders. There was confusion within the nursing staff regarding orders in the electronic health record compared to physician expectations. Orders stated patients should ambulate 3 times daily, but did not specify on which postoperative day this should start. Often, nursing verbal sign-out from the post-anesthesia care unit (PACU) would be an order for bedrest, despite no clear origin of this order. This created confusion among the nursing staff as to what the appropriate ambulation orders should be.
Nursing Workflow. The initial state of the nursing workflow was not conducive to evaluating for, or assisting with, ambulation. With no set time to assist and evaluate patients for ambulation, it turned into a task nurses needed to accomplish when they had extra time. With increasing demands of charting in the electronic health record, nurses often had to skip ambulation in order to accomplish other tasks.
Family Expectations. In addition to patient expectations, family members often had expectations that were not congruent with the planned postoperative course. Nurses stated family members would often tell them that they did not feel that their family member should be ambulating so soon after surgery. Often these family members had not attended preoperative education sessions with the patient. This was compounded by the uncertainty among the nursing staff regarding what exactly the ambulation orders were.
Interventions
Targeted interventions were created to address these 4 barriers to ambulation identified by staff.
Preoperative Education. Although all elective patients received a printed operative pathway booklet describing daily goals, including ambulation, patients still did not have a sufficient understanding of what was expected of them. The education session was modified to increase the time spent on both the expectation for and the rationale behind ambulation. That section of the education session ended with a verbal commitment and read-back of the expectations for ambulation by the patient.
Clarification of Electronic Orders. Postoperative orders within the colorectal standard pathway were changed, including specific time frames and frequency, to match the information provided in the patient education booklet. These orders were for ambulation within 4 hours of arrival to the floor, and the orders also noted that no patient should be on bedrest unless explicitly stated. From POD 1, all patients were to ambulate at least twice daily for the remainder of the hospital stay (patients were encouraged to walk 4 times daily, but we set a minimum expectation of twice daily for the order set). These orders were clarified with in-person meetings with the nursing staff and leadership from the PACU and the colorectal surgical ward.
Adjusted Nursing Workflow. Nurses were interviewed and asked to create a plan regarding how they could better incorporate ambulation into their daily workflow. Ambulation assessment was incorporated into the twice-per-shift recording of vital signs and patient safety assessment. This was recorded into the electronic health record at the same time as the patients’ vital signs. This allowed nurses to keep track of which patients would need extra assistance in ambulation and which patients were doing well on their own with the assistance of family. It also helped focus the resources of physical therapy and the single ambulation technician on the floor and to assist patients who needed more assistance.
Creation of Ambulation Encouragement Signs. The authors discovered that despite patients being told preoperatively about ambulation expectations, friends and family are not always included in these conversations. As nurses frequently cited both patients and family as reasons patients thought they should not walk, multiple signs inviting patients to take an active role in their recovery by ambulating were created and placed around the unit. The signs outlined the expectations of being out of bed and taking at least 1 step on the day of surgery and walking at least 4 times per day thereafter. In addition, we addressed frequently asked questions around issues such as walking with intravenous poles and urinary catheters. The posters were signed by all staff colorectal surgeons.
Results
Over the course of 7 months (June 2018 to December 2018), 253 postintervention patients were identified (Table 2). There was no difference between the pre-intervention group (n = 1762) and the postintervention group in terms of sex, race, origin, emergency status, operative time, and the majority of medical comorbidities (with the exception of smoking status and congestive heart failure). The postintervention group was slightly older (60 versus 57 years) and had a higher percentage of patients with an American Society of Anesthesiologists physical status score greater than 2 (66.8% versus 51.2%). The postintervention group also had higher rates of both malignancy (53.4% versus 33.3%) and inflammatory bowel disease (18.2% versus 14.4%).

The fidelity of the PDSA cycle was measured by pre-intervention and postintervention ambulation rates. Ambulation rates on POD 0, 1, and 2 improved from 36.4%, 47.3%, and 50.2% to 36.8%, 74.7%, and 82.6%, respectively (Table 3). The VTE rate decreased from 2.7% to 0.4% (P = 0.02), with 1 DVT and 0 PEs. It should be noted that the only patient who developed a VTE postintervention did not ambulate on PODs 0, 1, or 2.

Discussion
Postoperative VTE is a severe complication for postoperative colorectal surgery patients. Previous studies have demonstrated that increasing ambulation is associated with a lower rate of overall complications, and, when incorporated into a bundle, is associated with decreased rates of VTE.11,15 However, this is the first study to our knowledge demonstrating that creation of an ambulation protocol alone is associated with a decrease in VTE.
Analysis of pre-intervention data demonstrated a strong association between ambulation and an absence of VTE. No patient who ambulated on PODs 0, 1, and 2 developed a VTE. Based on those results, we moved forward with creating the ambulation protocol. While ambulation stayed stable on POD 0, there were 60% and 65% increases on PODs 1 and 2, respectively. Nurses cited late arrival to the floor for second and third start cases as the primary difficulty in getting patients to ambulate more on POD 0.
We believe the key to the success of the ambulation protocol was its multidisciplinary nature. Certainly, the easiest way to create an ambulation protocol is to change the postoperative orders to state patients must walk 4 times per day. However, if the nursing staff is unable or unwilling to carry out these orders, the orders serve little purpose. In order to make lasting changes, all stakeholders in the process must be identified. In our case, stakeholders included surgery and nursing leadership, surgeons, nurse practitioners, nurses, medical assistants, physical therapists, patient care technicians, and patients. This is where we utilized kaizen, a core principle of Lean methodology that empowers employees at the level of the work being carried out to propose ideas for improvement.16 From the beginning of the patient experience, the health care practitioners who were carrying out each step of the process were best able to identify the problems and create solutions. In addition, stakeholders were given regular updates regarding how their efforts were increasing ambulation rates and the results at the end of the study period.
This study also demonstrates that, in a health care system increasingly focused on both quality and cost, significant improvements in quality can be made without increasing cost or resource utilization. Early in the process, it was proposed that the only way to increase the ambulation rate would be to increase the number of physical therapists, nurses, and nursing assistants. However, after identifying the root causes of the problem, the solutions had more to do with improving workflow and fixing problem areas identified by the staff.
In addition to having a positive effect on the outcome studied, collaborative projects such as this between physicians and nurses may lead to increased nursing job satisfaction. A meta-analysis of 31 studies identified nurse-physician collaboration and autonomy as 2 factors that correlate most strongly with nursing satisfaction.17 A Cochrane review also suggests that practice-based interprofessional collaboration may lead to improved health care processes and outcomes.18
This study has several limitations. Pre-intervention ambulation rates were abstracted from institution-specific NSQIP data, and missing data were excluded from analysis. Also, due to the retrospective collection of the pre-intervention data, the distance of ambulation could not be quantified. The bar for ambulation is low, as patients were only required to get out of bed and walk 1 step. However, we feel that getting out of bed and taking even 1 step is substantially better than complete bedrest. It is likely that once patients cross the threshold of taking 1 step, they are more likely to ambulate. An area of future study may be to more precisely define the relationship between the quantity of ambulation in steps and its effect on VTE. Finally, we acknowledge that while there is no direct increase in costs, implementing an ambulation protocol does take time from all who participate in the project.
Conclusion
Creation of an ambulation protocol is associated with a decrease in postoperative VTE rates in colorectal surgery patients. A multidisciplinary approach is critical to identify the underlying problems and propose effective solutions. Further studies are required to better correlate the distance of ambulation and its effect on VTE. However, this study shows that even a minimum of 1 step is associated with decreased VTE rates.
Corresponding author: Aneel Damle, MD, MBA, Colon & Rectal Surgery Associates, 3433 Broadway St. NE, Suite 115, Minneapolis, MN 55413; [email protected].
Financial disclosures: None.
From the Department of Surgery, Washington University School of Medicine, St. Louis, MO.
Abstract
Background: Patients undergoing colorectal surgery are at high risk for postoperative venous thromboembolism (VTE). Early ambulation has been encouraged to lower rates of VTE, but evidence demonstrating its effectiveness outside of a bundle is limited.
Objective: To create a multidisciplinary ambulation protocol in an effort to reduce postoperative VTE.
Methods: A single-center, retrospective, comparative study of patients who underwent colectomy or proctectomy was conducted. Outcomes of patients operated on prior to protocol implementation were compared with a cohort after implementation. The intervention studied was the implementation of a multidisciplinary ambulation protocol. The primary endpoint was postoperative VTE.
Results: There was no difference between the pre-intervention group (n = 1762) and the postintervention group (n = 253) in terms of sex, race, origin, emergency status, operative time, and the majority of medical comorbidities (with the exception of smoking status and congestive heart failure). After the protocol was implemented, ambulation rates on postoperative days 0, 1, and 2 improved from 36.4%, 47.3%, and 50.2% to 36.8%, 74.7%, and 82.6%, respectively The VTE rate in the pre-intervention group was 2.7% versus a rate of 0.4% in the postintervention group (P = 0.02).
Conclusion: Creation of an ambulation protocol is associated with a significant reduction in VTE. Commitment from patients, families, nurses, physician extenders, and physicians is critical to the success of the program.
Keywords: VTE; pulmonary embolism; deep vein thrombosis; postoperative; quality improvement.
Postoperative venous thromboembolism (VTE) is a significant source of morbidity, mortality, and cost.1,2 Colorectal surgery patients are at particularly high risk for VTE due to positioning during surgery, pelvic dissection, and other conditions often found in these patients, such as cancer and inflammatory bowel disease.3 A National Surgical Quality Improvement Program (NSQIP) analysis demonstrated an overall rate of VTE in colorectal surgery patients of 2.4%, although other studies have demonstrated rates up to 9%, even in those receiving appropriate chemoprophylaxis.4-6 Many of these VTEs occur in the postdischarge setting. In a NSQIP study of colorectal surgery patients, the rate of VTE between discharge and 30 days was 0.47%.7 The cost burdenfor a postoperative VTE has been estimated to be more than $18,000.8
Studies from NSQIP have identified multiple factors associated with VTE in colorectal surgery patients, but NSQIP does not record ambulation as a standard variable.9 Multiple strategies have been implemented to reduce postoperative VTE. Often, these studies focus on increasing compliance with appropriate chemoprophylaxis, risk stratification, or bundling multiple strategies.10,11 However, despite the fact that postsurgical ambulation is widely encouraged and recommended by the American Society of Colon and Rectal Surgeons clinical practice guidelines, there is little evidence demonstrating the role of ambulation alone in the reduction of VTE.4,12 The purpose of this study was to create a multidisciplinary protocol to increase postoperative ambulation and evaluate its effect on VTE.
Methods
Setting
This study was conducted at a single academic tertiary care center.
Patients and Outcome Measures
All patients undergoing colectomy or proctectomy by surgeons in the section of colon and rectal surgery at a single institution between January 2011 and March 2017 were included. Colectomy and proctectomy were defined by CPT codes 44140, 44141, 44143, 44144, 44145, 44146, 44147, 44150, 44151, 44155, 44156, 44157, 44158, 44160, 44204, 44205, 44206, 44207, 44208, 44210, 44211, 44212, 44213, 45110, 45111, 45112, 45113, 45114, 45116, 45119, 45120, 45121, 45123, 45126, 45160, 45395, and 45397. The primary outcome of VTE within 30 days, including deep venous thrombosis (DVT) and pulmonary embolism (PE), was measured using institution-specific data from NSQIP in both the pre-intervention and postintervention setting. The occurrence of both DVT and PE in 1 patient was counted as a single event of VTE. Ambulation rate on postoperative day (POD) 0, 1, and 2 was calculated by NSQIP in the pre-intervention setting (our institution-specific NSQIP recorded ambulation data for an unrelated project) and by review of the electronic health record in the postintervention setting, as this institution-specific variable was no longer being collected. Ambulation was defined as getting out of bed and taking at least 1 step. The threshold for ambulating each day was once on POD 0 and twice on PODs 1 and 2. Patients with missing ambulation data were excluded from the analysis. Both prior to and throughout the intervention, all patients were given VTE chemoprophylaxis with either low-dose unfractionated heparin or low-molecular-weight heparin prior to induction of anesthesia, with chemoprophylaxis extending an additional 21 days after discharge (unless specifically contraindicated); sequential compression devices; and standard orders to ambulate 3 times daily from POD 0 as part of the standard Enhanced Recovery After Surgery protocol.
Analysis
Statistical analysis was performed using univariate analysis. Chi-square test and univariate logistic regression were used to determine the association between ambulation rates and VTE in the pre-intervention group. Chi-square test was also used to compare ambulation and VTE rates between the pre-intervention and postintervention groups. Plan-Do-Study-Act (PDSA) cycle fidelity (the degree to which a PDSA cycle is carried out in accordance with the guiding principles of its use) was measured by recording the ambulation rates both before and after the intervention.13 Statistical analysis was performed using SAS Version 9.4 (SAS Institute, Cary, NC). This study was reviewed by the Washington University School of Medicine Institutional Review Board and deemed to be quality improvement, not human subjects research, and therefore did not require formal approval.
Baseline Outcome Rates
A total of 1762 patients were identified during the pre-intervention period. The overall VTE rate in the pre-intervention group was 2.7% (n = 48), with 39 DVTs (2.2%) and 13 PEs (0.7%). Pre-intervention ambulation data were available on 590 patients. Baseline ambulation rates on PODs 0, 1, and 2 were 36.4% (213/590), 47.3% (279/590), and 50.2% (296/590), respectively. Patients who did not ambulate on POD 0 had a VTE rate of 4.3%, as compared to 0.9% in those who did ambulate (Table 1). Patients who did not ambulate twice on POD 1 had a VTE rate of 4.8%, compared to 1.1% in those who did ambulate (odds ratio [OR], 4.66; 95% confidence interval [CI], 1.34 to 16.28). Patients who did not ambulate twice on POD 2 had a VTE rate of 5.4%, compared to 0.7% in those who did. Finally, those who ambulated twice on both PODs 1 and 2 had a 0% rate of VTE, compared to 4.9% in those who did not ambulate on both PODs.

Ambulation Protocol
After baseline outcome rates had been established, a multidisciplinary team of medical assistants, nurses, nurse practitioners, and physicians worked together to identify all processes that involved postoperative ambulation. Given the significant differences in VTE rates between patients who ambulated and those that did not, we created a multidisciplinary ambulation protocol using the PDSA method.14 Multiple points of patient contact were chosen for intervention, and the ambulation protocol was implemented in June 2018 and continued for 7 months.
Patients were observed from their initial office visit with a surgeon, during the preoperative education encounter, and in the operating room and on the surgical ward until discharge. Representatives from multiple disciplines who encountered patients at various times in the process, including medical assistants, patient care technicians, nurses, nurse practitioners, physical therapists, and physicians, participated in a kick-off meeting to identify difficulties they encounter when encouraging patient ambulation. The following 4 areas were identified.
Barriers to Patient Ambulation
Patient Expectations. Patients did not appear to have a clear expectation of what their ambulation goals were postoperatively, despite the fact that each patient is given an operative pathway booklet that includes their goals for each day, including ambulation. The consensus was that patients were overwhelmed with the amount of information and, oftentimes, the severity of their diagnosis, so the information regarding ambulation was not retained. Nurses commented that patients frequently stated that they did not think their surgeon wanted them to get out of bed postoperatively.
Electronic Orders. There was confusion within the nursing staff regarding orders in the electronic health record compared to physician expectations. Orders stated patients should ambulate 3 times daily, but did not specify on which postoperative day this should start. Often, nursing verbal sign-out from the post-anesthesia care unit (PACU) would be an order for bedrest, despite no clear origin of this order. This created confusion among the nursing staff as to what the appropriate ambulation orders should be.
Nursing Workflow. The initial state of the nursing workflow was not conducive to evaluating for, or assisting with, ambulation. With no set time to assist and evaluate patients for ambulation, it turned into a task nurses needed to accomplish when they had extra time. With increasing demands of charting in the electronic health record, nurses often had to skip ambulation in order to accomplish other tasks.
Family Expectations. In addition to patient expectations, family members often had expectations that were not congruent with the planned postoperative course. Nurses stated family members would often tell them that they did not feel that their family member should be ambulating so soon after surgery. Often these family members had not attended preoperative education sessions with the patient. This was compounded by the uncertainty among the nursing staff regarding what exactly the ambulation orders were.
Interventions
Targeted interventions were created to address these 4 barriers to ambulation identified by staff.
Preoperative Education. Although all elective patients received a printed operative pathway booklet describing daily goals, including ambulation, patients still did not have a sufficient understanding of what was expected of them. The education session was modified to increase the time spent on both the expectation for and the rationale behind ambulation. That section of the education session ended with a verbal commitment and read-back of the expectations for ambulation by the patient.
Clarification of Electronic Orders. Postoperative orders within the colorectal standard pathway were changed, including specific time frames and frequency, to match the information provided in the patient education booklet. These orders were for ambulation within 4 hours of arrival to the floor, and the orders also noted that no patient should be on bedrest unless explicitly stated. From POD 1, all patients were to ambulate at least twice daily for the remainder of the hospital stay (patients were encouraged to walk 4 times daily, but we set a minimum expectation of twice daily for the order set). These orders were clarified with in-person meetings with the nursing staff and leadership from the PACU and the colorectal surgical ward.
Adjusted Nursing Workflow. Nurses were interviewed and asked to create a plan regarding how they could better incorporate ambulation into their daily workflow. Ambulation assessment was incorporated into the twice-per-shift recording of vital signs and patient safety assessment. This was recorded into the electronic health record at the same time as the patients’ vital signs. This allowed nurses to keep track of which patients would need extra assistance in ambulation and which patients were doing well on their own with the assistance of family. It also helped focus the resources of physical therapy and the single ambulation technician on the floor and to assist patients who needed more assistance.
Creation of Ambulation Encouragement Signs. The authors discovered that despite patients being told preoperatively about ambulation expectations, friends and family are not always included in these conversations. As nurses frequently cited both patients and family as reasons patients thought they should not walk, multiple signs inviting patients to take an active role in their recovery by ambulating were created and placed around the unit. The signs outlined the expectations of being out of bed and taking at least 1 step on the day of surgery and walking at least 4 times per day thereafter. In addition, we addressed frequently asked questions around issues such as walking with intravenous poles and urinary catheters. The posters were signed by all staff colorectal surgeons.
Results
Over the course of 7 months (June 2018 to December 2018), 253 postintervention patients were identified (Table 2). There was no difference between the pre-intervention group (n = 1762) and the postintervention group in terms of sex, race, origin, emergency status, operative time, and the majority of medical comorbidities (with the exception of smoking status and congestive heart failure). The postintervention group was slightly older (60 versus 57 years) and had a higher percentage of patients with an American Society of Anesthesiologists physical status score greater than 2 (66.8% versus 51.2%). The postintervention group also had higher rates of both malignancy (53.4% versus 33.3%) and inflammatory bowel disease (18.2% versus 14.4%).

The fidelity of the PDSA cycle was measured by pre-intervention and postintervention ambulation rates. Ambulation rates on POD 0, 1, and 2 improved from 36.4%, 47.3%, and 50.2% to 36.8%, 74.7%, and 82.6%, respectively (Table 3). The VTE rate decreased from 2.7% to 0.4% (P = 0.02), with 1 DVT and 0 PEs. It should be noted that the only patient who developed a VTE postintervention did not ambulate on PODs 0, 1, or 2.

Discussion
Postoperative VTE is a severe complication for postoperative colorectal surgery patients. Previous studies have demonstrated that increasing ambulation is associated with a lower rate of overall complications, and, when incorporated into a bundle, is associated with decreased rates of VTE.11,15 However, this is the first study to our knowledge demonstrating that creation of an ambulation protocol alone is associated with a decrease in VTE.
Analysis of pre-intervention data demonstrated a strong association between ambulation and an absence of VTE. No patient who ambulated on PODs 0, 1, and 2 developed a VTE. Based on those results, we moved forward with creating the ambulation protocol. While ambulation stayed stable on POD 0, there were 60% and 65% increases on PODs 1 and 2, respectively. Nurses cited late arrival to the floor for second and third start cases as the primary difficulty in getting patients to ambulate more on POD 0.
We believe the key to the success of the ambulation protocol was its multidisciplinary nature. Certainly, the easiest way to create an ambulation protocol is to change the postoperative orders to state patients must walk 4 times per day. However, if the nursing staff is unable or unwilling to carry out these orders, the orders serve little purpose. In order to make lasting changes, all stakeholders in the process must be identified. In our case, stakeholders included surgery and nursing leadership, surgeons, nurse practitioners, nurses, medical assistants, physical therapists, patient care technicians, and patients. This is where we utilized kaizen, a core principle of Lean methodology that empowers employees at the level of the work being carried out to propose ideas for improvement.16 From the beginning of the patient experience, the health care practitioners who were carrying out each step of the process were best able to identify the problems and create solutions. In addition, stakeholders were given regular updates regarding how their efforts were increasing ambulation rates and the results at the end of the study period.
This study also demonstrates that, in a health care system increasingly focused on both quality and cost, significant improvements in quality can be made without increasing cost or resource utilization. Early in the process, it was proposed that the only way to increase the ambulation rate would be to increase the number of physical therapists, nurses, and nursing assistants. However, after identifying the root causes of the problem, the solutions had more to do with improving workflow and fixing problem areas identified by the staff.
In addition to having a positive effect on the outcome studied, collaborative projects such as this between physicians and nurses may lead to increased nursing job satisfaction. A meta-analysis of 31 studies identified nurse-physician collaboration and autonomy as 2 factors that correlate most strongly with nursing satisfaction.17 A Cochrane review also suggests that practice-based interprofessional collaboration may lead to improved health care processes and outcomes.18
This study has several limitations. Pre-intervention ambulation rates were abstracted from institution-specific NSQIP data, and missing data were excluded from analysis. Also, due to the retrospective collection of the pre-intervention data, the distance of ambulation could not be quantified. The bar for ambulation is low, as patients were only required to get out of bed and walk 1 step. However, we feel that getting out of bed and taking even 1 step is substantially better than complete bedrest. It is likely that once patients cross the threshold of taking 1 step, they are more likely to ambulate. An area of future study may be to more precisely define the relationship between the quantity of ambulation in steps and its effect on VTE. Finally, we acknowledge that while there is no direct increase in costs, implementing an ambulation protocol does take time from all who participate in the project.
Conclusion
Creation of an ambulation protocol is associated with a decrease in postoperative VTE rates in colorectal surgery patients. A multidisciplinary approach is critical to identify the underlying problems and propose effective solutions. Further studies are required to better correlate the distance of ambulation and its effect on VTE. However, this study shows that even a minimum of 1 step is associated with decreased VTE rates.
Corresponding author: Aneel Damle, MD, MBA, Colon & Rectal Surgery Associates, 3433 Broadway St. NE, Suite 115, Minneapolis, MN 55413; [email protected].
Financial disclosures: None.
1. Gangireddy C, Rectenwald JR, Upchurch GR, et al. Risk factors and clinical impact of postoperative symptomatic venous thromboembolism. J Vasc Surg. 2007;45:341-342.
2. Newhook TE, LaPar DJ, Walters DM, et al. Impact of postoperative venous thromboembolism on postoperative morbidity, mortality, and resource utilization after hepatectomy. Am Surg. 2015;81:1216-1223.
3. Bergqvist D. Venous thromboembolism: a review of risk and prevention in colorectal surgery patients. Dis Colon Rectum. 2006;49:1620-1628.
4. Fleming F, Gaertner W, Ternent CA, et al. The American society of colon and rectal surgeons clinical practice guideline for the prevention of venous thromboembolic disease in colorectal surgery. Dis Colon Rectum. 2018;61:14-20.
5. McLeod RS, Geerts WH, Sniderman KW, et al. Canadian Colorectal Surgery DVT Prophylaxis Trial investigators. Subcutaneous heparin versus low-molecular-weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian colorectal DV prophylaxis trial: a randomized, double-blind trial. Ann Surg. 2001;233:438-444.
6. Shapiro R, Vogel JD, Kiran RP. Risk of postoperative venous thromboembolism after laparoscopic and open colorectal surgery: an additional benefit of the minimally invasive approach? Dis Colon Rectum. 2011;54:1496-1502.
7. Dimick JB, Chen SL, Taheri PA, et al. Hospital costs associated with surgical complications: a report from the private-sector National Surgical Quality Improvement Program. J Am Coll Surg. 2004;199:531-537.
8. Fleming FJ, Kim MJ, Salloum RM, et al. How much do we need to worry about venous thromboembolism after hospital discharge? A study of colorectal surgery patients using the National Surgical Quality Improvement Program database. Dis Colon Rectum. 2010;53:1355-1360.
9. ACS NSQIP. User guide for the 2016 ACS NSQIP participant use data file (PUF). 2017. www.facs.org/~/media/files/quality%20programs/nsqip/nsqip_puf_userguide_2016.ashx Accessed July 10, 2020.
10. Caprini JA. Risk assessment as a guide for the prevention of the many faces of venous thromboembolism. Am J Surg. 2010;199(1 Suppl):S3-S10.
11. Cassidy MR, Rosenkranz P, McAney D. Reducing postoperative venous thromboembolism complications with a standardized risk-stratified prophylaxis protocol and mobilization protocol. J Am Coll Surg. 2014;218:1095-1104.
12. Lau BD, Streiff MB, Kraus PS, et al. No evidence to support ambulation for reducing postoperative venous thromboembolism. J Am Coll Surg. 2014;219:1101-1103.
13. McNicholas C, Lennox L, Woodcock T, et al. Evolving quality improvement support strategies to improve Plan–Do–Study–Act cycle fidelity: a retrospective mixed-methods study. BMJ Qual Saf. 2019;28:356-365.
14. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan–do–study–act method to improve quality in healthcare. BMC Qual Saf. 2014;23:290-298.
15. Nevo Y, Shaltiel T, Constantini N, et al. Effect of ambulation and physical activity on postoperative complications. J Am Coll Surg. 2016;223(Suppl 1):S61.
16. Mazzocato P, Stenfors-Hayes T, von Thiele Schwarz U, et al. Kaizen practice in healthcare: a qualitative analysis of hospital employees’ suggestions for improvement. BMJ Open. 2016;6:e012256.
17. Zangaro GA, Soeken KL. A meta-analysis of studies of nurses’ job satisfaction. Res Nursing Health. 2007;30:445-458.
18. Reeves S, Pelone F, Harrison R, et al. Interprofessional collaboration to improve professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2017;6(6):CD000072.
1. Gangireddy C, Rectenwald JR, Upchurch GR, et al. Risk factors and clinical impact of postoperative symptomatic venous thromboembolism. J Vasc Surg. 2007;45:341-342.
2. Newhook TE, LaPar DJ, Walters DM, et al. Impact of postoperative venous thromboembolism on postoperative morbidity, mortality, and resource utilization after hepatectomy. Am Surg. 2015;81:1216-1223.
3. Bergqvist D. Venous thromboembolism: a review of risk and prevention in colorectal surgery patients. Dis Colon Rectum. 2006;49:1620-1628.
4. Fleming F, Gaertner W, Ternent CA, et al. The American society of colon and rectal surgeons clinical practice guideline for the prevention of venous thromboembolic disease in colorectal surgery. Dis Colon Rectum. 2018;61:14-20.
5. McLeod RS, Geerts WH, Sniderman KW, et al. Canadian Colorectal Surgery DVT Prophylaxis Trial investigators. Subcutaneous heparin versus low-molecular-weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian colorectal DV prophylaxis trial: a randomized, double-blind trial. Ann Surg. 2001;233:438-444.
6. Shapiro R, Vogel JD, Kiran RP. Risk of postoperative venous thromboembolism after laparoscopic and open colorectal surgery: an additional benefit of the minimally invasive approach? Dis Colon Rectum. 2011;54:1496-1502.
7. Dimick JB, Chen SL, Taheri PA, et al. Hospital costs associated with surgical complications: a report from the private-sector National Surgical Quality Improvement Program. J Am Coll Surg. 2004;199:531-537.
8. Fleming FJ, Kim MJ, Salloum RM, et al. How much do we need to worry about venous thromboembolism after hospital discharge? A study of colorectal surgery patients using the National Surgical Quality Improvement Program database. Dis Colon Rectum. 2010;53:1355-1360.
9. ACS NSQIP. User guide for the 2016 ACS NSQIP participant use data file (PUF). 2017. www.facs.org/~/media/files/quality%20programs/nsqip/nsqip_puf_userguide_2016.ashx Accessed July 10, 2020.
10. Caprini JA. Risk assessment as a guide for the prevention of the many faces of venous thromboembolism. Am J Surg. 2010;199(1 Suppl):S3-S10.
11. Cassidy MR, Rosenkranz P, McAney D. Reducing postoperative venous thromboembolism complications with a standardized risk-stratified prophylaxis protocol and mobilization protocol. J Am Coll Surg. 2014;218:1095-1104.
12. Lau BD, Streiff MB, Kraus PS, et al. No evidence to support ambulation for reducing postoperative venous thromboembolism. J Am Coll Surg. 2014;219:1101-1103.
13. McNicholas C, Lennox L, Woodcock T, et al. Evolving quality improvement support strategies to improve Plan–Do–Study–Act cycle fidelity: a retrospective mixed-methods study. BMJ Qual Saf. 2019;28:356-365.
14. Taylor MJ, McNicholas C, Nicolay C, et al. Systematic review of the application of the plan–do–study–act method to improve quality in healthcare. BMC Qual Saf. 2014;23:290-298.
15. Nevo Y, Shaltiel T, Constantini N, et al. Effect of ambulation and physical activity on postoperative complications. J Am Coll Surg. 2016;223(Suppl 1):S61.
16. Mazzocato P, Stenfors-Hayes T, von Thiele Schwarz U, et al. Kaizen practice in healthcare: a qualitative analysis of hospital employees’ suggestions for improvement. BMJ Open. 2016;6:e012256.
17. Zangaro GA, Soeken KL. A meta-analysis of studies of nurses’ job satisfaction. Res Nursing Health. 2007;30:445-458.
18. Reeves S, Pelone F, Harrison R, et al. Interprofessional collaboration to improve professional practice and healthcare outcomes. Cochrane Database Syst Rev. 2017;6(6):CD000072.
COVID-19 fears would keep most Hispanics with stroke, MI symptoms home
More than half of Hispanic adults would be afraid to go to a hospital for a possible heart attack or stroke because they might get infected with SARS-CoV-2, according to a new survey from the American Heart Association.
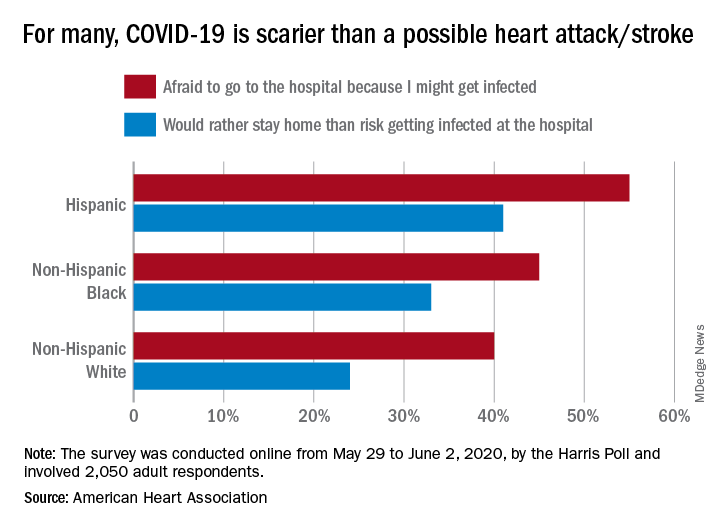
Compared with Hispanic respondents, 55% of whom said they feared COVID-19, significantly fewer Blacks (45%) and Whites (40%) would be scared to go to the hospital if they thought they were having a heart attack or stroke, the AHA said based on the survey of 2,050 adults, which was conducted May 29 to June 2, 2020, by the Harris Poll.
Hispanics also were significantly more likely to stay home if they thought they were experiencing a heart attack or stroke (41%), rather than risk getting infected at the hospital, than were Blacks (33%), who were significantly more likely than Whites (24%) to stay home, the AHA reported.
White respondents, on the other hand, were the most likely to believe (89%) that a hospital would give them the same quality of care provided to everyone else. Hispanics and Blacks had significantly lower rates, at 78% and 74%, respectively, the AHA noted.
These findings are “yet another challenge for Black and Hispanic communities, who are more likely to have underlying health conditions such as cardiovascular disease and diabetes and dying of COVID-19 at disproportionately high rates,” Rafael Ortiz, MD, American Heart Association volunteer medical expert and chief of neuro-endovascular surgery at Lenox Hill Hospital, New York, said in the AHA statement.
The survey was performed in conjunction with the AHA’s “Don’t Die of Doubt” campaign, which “reminds Americans, especially in Hispanic and Black communities, that the hospital remains the safest place to be if experiencing symptoms of a heart attack or a stroke.”
Among all the survey respondents, 57% said they would feel better if hospitals treated COVID-19 patients in a separate area. A number of other possible precautions ranked lower in helping them feel better:
- Screen all visitors, patients, and staff for COVID-19 symptoms when they enter the hospital: 39%.
- Require all patients, visitors, and staff to wear masks: 30%.
- Put increased cleaning protocols in place to disinfect multiple times per day: 23%.
- “Nothing would make me feel comfortable”: 6%.
Despite all the concerns about the risk of coronavirus infection, however, most Americans (77%) still believe that hospitals are the safest place to be in the event of a medical emergency, and 84% said that hospitals are prepared to safely treat emergencies that are not related to the pandemic, the AHA reported.
“Health care professionals know what to do even when things seem chaotic, and emergency departments have made plans behind the scenes to keep patients and healthcare workers safe even during a pandemic,” Dr. Ortiz pointed out.
More than half of Hispanic adults would be afraid to go to a hospital for a possible heart attack or stroke because they might get infected with SARS-CoV-2, according to a new survey from the American Heart Association.

Compared with Hispanic respondents, 55% of whom said they feared COVID-19, significantly fewer Blacks (45%) and Whites (40%) would be scared to go to the hospital if they thought they were having a heart attack or stroke, the AHA said based on the survey of 2,050 adults, which was conducted May 29 to June 2, 2020, by the Harris Poll.
Hispanics also were significantly more likely to stay home if they thought they were experiencing a heart attack or stroke (41%), rather than risk getting infected at the hospital, than were Blacks (33%), who were significantly more likely than Whites (24%) to stay home, the AHA reported.
White respondents, on the other hand, were the most likely to believe (89%) that a hospital would give them the same quality of care provided to everyone else. Hispanics and Blacks had significantly lower rates, at 78% and 74%, respectively, the AHA noted.
These findings are “yet another challenge for Black and Hispanic communities, who are more likely to have underlying health conditions such as cardiovascular disease and diabetes and dying of COVID-19 at disproportionately high rates,” Rafael Ortiz, MD, American Heart Association volunteer medical expert and chief of neuro-endovascular surgery at Lenox Hill Hospital, New York, said in the AHA statement.
The survey was performed in conjunction with the AHA’s “Don’t Die of Doubt” campaign, which “reminds Americans, especially in Hispanic and Black communities, that the hospital remains the safest place to be if experiencing symptoms of a heart attack or a stroke.”
Among all the survey respondents, 57% said they would feel better if hospitals treated COVID-19 patients in a separate area. A number of other possible precautions ranked lower in helping them feel better:
- Screen all visitors, patients, and staff for COVID-19 symptoms when they enter the hospital: 39%.
- Require all patients, visitors, and staff to wear masks: 30%.
- Put increased cleaning protocols in place to disinfect multiple times per day: 23%.
- “Nothing would make me feel comfortable”: 6%.
Despite all the concerns about the risk of coronavirus infection, however, most Americans (77%) still believe that hospitals are the safest place to be in the event of a medical emergency, and 84% said that hospitals are prepared to safely treat emergencies that are not related to the pandemic, the AHA reported.
“Health care professionals know what to do even when things seem chaotic, and emergency departments have made plans behind the scenes to keep patients and healthcare workers safe even during a pandemic,” Dr. Ortiz pointed out.
More than half of Hispanic adults would be afraid to go to a hospital for a possible heart attack or stroke because they might get infected with SARS-CoV-2, according to a new survey from the American Heart Association.

Compared with Hispanic respondents, 55% of whom said they feared COVID-19, significantly fewer Blacks (45%) and Whites (40%) would be scared to go to the hospital if they thought they were having a heart attack or stroke, the AHA said based on the survey of 2,050 adults, which was conducted May 29 to June 2, 2020, by the Harris Poll.
Hispanics also were significantly more likely to stay home if they thought they were experiencing a heart attack or stroke (41%), rather than risk getting infected at the hospital, than were Blacks (33%), who were significantly more likely than Whites (24%) to stay home, the AHA reported.
White respondents, on the other hand, were the most likely to believe (89%) that a hospital would give them the same quality of care provided to everyone else. Hispanics and Blacks had significantly lower rates, at 78% and 74%, respectively, the AHA noted.
These findings are “yet another challenge for Black and Hispanic communities, who are more likely to have underlying health conditions such as cardiovascular disease and diabetes and dying of COVID-19 at disproportionately high rates,” Rafael Ortiz, MD, American Heart Association volunteer medical expert and chief of neuro-endovascular surgery at Lenox Hill Hospital, New York, said in the AHA statement.
The survey was performed in conjunction with the AHA’s “Don’t Die of Doubt” campaign, which “reminds Americans, especially in Hispanic and Black communities, that the hospital remains the safest place to be if experiencing symptoms of a heart attack or a stroke.”
Among all the survey respondents, 57% said they would feel better if hospitals treated COVID-19 patients in a separate area. A number of other possible precautions ranked lower in helping them feel better:
- Screen all visitors, patients, and staff for COVID-19 symptoms when they enter the hospital: 39%.
- Require all patients, visitors, and staff to wear masks: 30%.
- Put increased cleaning protocols in place to disinfect multiple times per day: 23%.
- “Nothing would make me feel comfortable”: 6%.
Despite all the concerns about the risk of coronavirus infection, however, most Americans (77%) still believe that hospitals are the safest place to be in the event of a medical emergency, and 84% said that hospitals are prepared to safely treat emergencies that are not related to the pandemic, the AHA reported.
“Health care professionals know what to do even when things seem chaotic, and emergency departments have made plans behind the scenes to keep patients and healthcare workers safe even during a pandemic,” Dr. Ortiz pointed out.
Migraine headache pearls
A 25-year-old woman presents to discuss treatment of her headaches. They occur two or three times a month and last for 4-6 hours. The headaches are disabling, have a pounding quality behind the patient’s right eye, and worsen with exercise. The patient’s neurologic exam is normal.
She has tried oral sumatriptan and naproxen, but neither drug provided her with any relief from the headaches. What treatment would you recommend?
A. Topiramate
B. Beta-blocker
C. Lasmiditan
D. Metoclopramide plus sumatriptan
E. Ubrogepant
It is common to see patients with migraine headaches and to see patients with migraines who have not responded to previous migraine therapies.
For this patient, I would try choice D first, giving metoclopramide with oral sumatriptan to see if it can improve response to sumatriptan. The two new classes of drugs for acute migraine therapy, the gepants and ditans, certainly have a role in patients unresponsive or intolerant of triptans/NSAIDS, but I would try several tricks with these less expensive medications first before entering into prior authorization hell trying to get a gepant or ditan.
When a patient has already used a triptan but experienced no benefit from it, often the next medication a patient tries is a different triptan. Dahlof reviewed four trials that looked at the efficacy of switching sumatriptan nonresponders to a different triptan and found that lack of response to sumatriptan did not predict lack of response to an alternative triptan.1 Unfortunately, acquiring insurance coverage for an alternate triptan can be difficult.
Other treatment options are nasal or injectable formulations of sumatriptan. Both of these are more costly than oral sumatriptan, and injectable sumatriptan has more side effects than oral triptans.
Combining treatment with metoclopramide can be helpful. In a study by Schulman and Dermott looking at patients who had previously been triptan nonresponders, 63% of those who took metoclopramide with sumatriptan had meaningful pain relief, compared with 31% of those who received sumatriptan and placebo.2
In a different study, Tfelt-Hansen et al. compared treatment with the combination of lysine acetylsalicylate plus metoclopramide versus treatment with 100 mg of sumatriptan.3 There was no difference in outcomes between the two treatment groups, with the lysine acetylsalicylate plus metoclopramide patients having a 57% success rate for first treated migraine compared with 53% of the sumatriptan-treated patients.
Treating with the combination of naproxen plus sumatriptan is superior to treating with either medication alone. Brandes et al. reported on two studies involving the use of the sumatriptan/naproxen combination, compared with using sumatriptan, naproxen, or placebo.4 In both, taking the sumatriptan/naproxen combination was superior to taking sumatriptan, naproxen, or placebo (P < .001).
In a study of patients with poor prior response to triptans, Mathew et al. found that the sumatriptan/naproxen combination was superior to placebo for both 2- and 24-hour headache relief (P < .001).5
Pearl
Try several options before abandoning triptans in previous triptan nonresponders, including trying a different triptan, adding metoclopramide, orcombining with an NSAID.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Dahlöf CG. Infrequent or nonresponse to oral sumatriptan does not predict response to other triptans – review of four trials. Cephalalgia. 2006 Feb;26(2):98-106.
2. Schulman EA, Dermott KF. Sumatriptan plus metoclopramide in triptan-nonresponsive migraineurs. Headache. 2003 Jul-Aug;43(7):729-33.
3. Tfelt-Hansen P et al. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet. 1995 Oct 7;346(8980):923-6.
4. Brandes JL et al. Sumatriptan‐naproxen for acute treatment of migraine: A randomized trial. JAMA. 2007;297:1443‐54.
5. Mathew NT, Landy S, Stark S, et al. Fixed‐dose sumatriptan and naproxen in poor responders to triptans with a short half‐life. Headache. 2009;49:971‐82.
A 25-year-old woman presents to discuss treatment of her headaches. They occur two or three times a month and last for 4-6 hours. The headaches are disabling, have a pounding quality behind the patient’s right eye, and worsen with exercise. The patient’s neurologic exam is normal.
She has tried oral sumatriptan and naproxen, but neither drug provided her with any relief from the headaches. What treatment would you recommend?
A. Topiramate
B. Beta-blocker
C. Lasmiditan
D. Metoclopramide plus sumatriptan
E. Ubrogepant
It is common to see patients with migraine headaches and to see patients with migraines who have not responded to previous migraine therapies.
For this patient, I would try choice D first, giving metoclopramide with oral sumatriptan to see if it can improve response to sumatriptan. The two new classes of drugs for acute migraine therapy, the gepants and ditans, certainly have a role in patients unresponsive or intolerant of triptans/NSAIDS, but I would try several tricks with these less expensive medications first before entering into prior authorization hell trying to get a gepant or ditan.
When a patient has already used a triptan but experienced no benefit from it, often the next medication a patient tries is a different triptan. Dahlof reviewed four trials that looked at the efficacy of switching sumatriptan nonresponders to a different triptan and found that lack of response to sumatriptan did not predict lack of response to an alternative triptan.1 Unfortunately, acquiring insurance coverage for an alternate triptan can be difficult.
Other treatment options are nasal or injectable formulations of sumatriptan. Both of these are more costly than oral sumatriptan, and injectable sumatriptan has more side effects than oral triptans.
Combining treatment with metoclopramide can be helpful. In a study by Schulman and Dermott looking at patients who had previously been triptan nonresponders, 63% of those who took metoclopramide with sumatriptan had meaningful pain relief, compared with 31% of those who received sumatriptan and placebo.2
In a different study, Tfelt-Hansen et al. compared treatment with the combination of lysine acetylsalicylate plus metoclopramide versus treatment with 100 mg of sumatriptan.3 There was no difference in outcomes between the two treatment groups, with the lysine acetylsalicylate plus metoclopramide patients having a 57% success rate for first treated migraine compared with 53% of the sumatriptan-treated patients.
Treating with the combination of naproxen plus sumatriptan is superior to treating with either medication alone. Brandes et al. reported on two studies involving the use of the sumatriptan/naproxen combination, compared with using sumatriptan, naproxen, or placebo.4 In both, taking the sumatriptan/naproxen combination was superior to taking sumatriptan, naproxen, or placebo (P < .001).
In a study of patients with poor prior response to triptans, Mathew et al. found that the sumatriptan/naproxen combination was superior to placebo for both 2- and 24-hour headache relief (P < .001).5
Pearl
Try several options before abandoning triptans in previous triptan nonresponders, including trying a different triptan, adding metoclopramide, orcombining with an NSAID.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Dahlöf CG. Infrequent or nonresponse to oral sumatriptan does not predict response to other triptans – review of four trials. Cephalalgia. 2006 Feb;26(2):98-106.
2. Schulman EA, Dermott KF. Sumatriptan plus metoclopramide in triptan-nonresponsive migraineurs. Headache. 2003 Jul-Aug;43(7):729-33.
3. Tfelt-Hansen P et al. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet. 1995 Oct 7;346(8980):923-6.
4. Brandes JL et al. Sumatriptan‐naproxen for acute treatment of migraine: A randomized trial. JAMA. 2007;297:1443‐54.
5. Mathew NT, Landy S, Stark S, et al. Fixed‐dose sumatriptan and naproxen in poor responders to triptans with a short half‐life. Headache. 2009;49:971‐82.
A 25-year-old woman presents to discuss treatment of her headaches. They occur two or three times a month and last for 4-6 hours. The headaches are disabling, have a pounding quality behind the patient’s right eye, and worsen with exercise. The patient’s neurologic exam is normal.
She has tried oral sumatriptan and naproxen, but neither drug provided her with any relief from the headaches. What treatment would you recommend?
A. Topiramate
B. Beta-blocker
C. Lasmiditan
D. Metoclopramide plus sumatriptan
E. Ubrogepant
It is common to see patients with migraine headaches and to see patients with migraines who have not responded to previous migraine therapies.
For this patient, I would try choice D first, giving metoclopramide with oral sumatriptan to see if it can improve response to sumatriptan. The two new classes of drugs for acute migraine therapy, the gepants and ditans, certainly have a role in patients unresponsive or intolerant of triptans/NSAIDS, but I would try several tricks with these less expensive medications first before entering into prior authorization hell trying to get a gepant or ditan.
When a patient has already used a triptan but experienced no benefit from it, often the next medication a patient tries is a different triptan. Dahlof reviewed four trials that looked at the efficacy of switching sumatriptan nonresponders to a different triptan and found that lack of response to sumatriptan did not predict lack of response to an alternative triptan.1 Unfortunately, acquiring insurance coverage for an alternate triptan can be difficult.
Other treatment options are nasal or injectable formulations of sumatriptan. Both of these are more costly than oral sumatriptan, and injectable sumatriptan has more side effects than oral triptans.
Combining treatment with metoclopramide can be helpful. In a study by Schulman and Dermott looking at patients who had previously been triptan nonresponders, 63% of those who took metoclopramide with sumatriptan had meaningful pain relief, compared with 31% of those who received sumatriptan and placebo.2
In a different study, Tfelt-Hansen et al. compared treatment with the combination of lysine acetylsalicylate plus metoclopramide versus treatment with 100 mg of sumatriptan.3 There was no difference in outcomes between the two treatment groups, with the lysine acetylsalicylate plus metoclopramide patients having a 57% success rate for first treated migraine compared with 53% of the sumatriptan-treated patients.
Treating with the combination of naproxen plus sumatriptan is superior to treating with either medication alone. Brandes et al. reported on two studies involving the use of the sumatriptan/naproxen combination, compared with using sumatriptan, naproxen, or placebo.4 In both, taking the sumatriptan/naproxen combination was superior to taking sumatriptan, naproxen, or placebo (P < .001).
In a study of patients with poor prior response to triptans, Mathew et al. found that the sumatriptan/naproxen combination was superior to placebo for both 2- and 24-hour headache relief (P < .001).5
Pearl
Try several options before abandoning triptans in previous triptan nonresponders, including trying a different triptan, adding metoclopramide, orcombining with an NSAID.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Dahlöf CG. Infrequent or nonresponse to oral sumatriptan does not predict response to other triptans – review of four trials. Cephalalgia. 2006 Feb;26(2):98-106.
2. Schulman EA, Dermott KF. Sumatriptan plus metoclopramide in triptan-nonresponsive migraineurs. Headache. 2003 Jul-Aug;43(7):729-33.
3. Tfelt-Hansen P et al. The effectiveness of combined oral lysine acetylsalicylate and metoclopramide compared with oral sumatriptan for migraine. Lancet. 1995 Oct 7;346(8980):923-6.
4. Brandes JL et al. Sumatriptan‐naproxen for acute treatment of migraine: A randomized trial. JAMA. 2007;297:1443‐54.
5. Mathew NT, Landy S, Stark S, et al. Fixed‐dose sumatriptan and naproxen in poor responders to triptans with a short half‐life. Headache. 2009;49:971‐82.
Internists’ use of ultrasound can reduce radiology referrals
researchers say.
“It’s a safe and very useful tool,” Marco Barchiesi, MD, an internal medicine resident at Luigi Sacco Hospital in Milan, said in an interview. “We had a great reduction in chest x-rays because of the use of ultrasound.”
The finding addresses concerns that ultrasound used in primary care could consume more health care resources or put patients at risk.
Dr. Barchiesi and colleagues published their findings July 20 in the European Journal of Internal Medicine.
Point-of-care ultrasound has become increasingly common as miniaturization of devices has made them more portable. The approach has caught on particularly in emergency departments where quick decisions are of the essence.
Its use in internal medicine has been more controversial, with concerns raised that improperly trained practitioners may miss diagnoses or refer patients for unnecessary tests as a result of uncertainty about their findings.
To measure the effect of point-of-care ultrasound in an internal medicine hospital ward, Dr. Barchiesi and colleagues alternated months when point-of-care ultrasound was allowed with months when it was not allowed, for a total of 4 months each, on an internal medicine unit. They allowed the ultrasound to be used for invasive procedures and excluded patients whose critical condition made point-of-care ultrasound crucial.
The researchers analyzed data on 263 patients in the “on” months when point-of-care ultrasound was used, and 255 in the “off” months when it wasn’t used. The two groups were well balanced in age, sex, comorbidity, and clinical impairment.
During the on months, the internists ordered 113 diagnostic tests (0.43 per patient). During the off months they ordered 329 tests (1.29 per patient).
The odds of being referred for a chest x-ray were 87% less in the “on” months, compared with the off months, a statistically significant finding (P < .001). The risk for a chest CT scan and abdominal ultrasound were also reduced during the on months, but the risk for an abdominal CT was increased.
Nineteen patients died during the o” months and 10 during the off months, a difference that was not statistically significant (P = .15). The median length of stay in the hospital was almost the same for the two groups: 9 days for the on months and 9 days for the off months. The difference was also not statistically significant (P = .094).
Point-of-care ultrasound is particularly accurate in identifying cardiac abnormalities and pleural fluid and pneumonia, and it can be used effectively for monitoring heart conditions, the researchers wrote. This could explain the reduction in chest x-rays and CT scans.
On the other hand, ultrasound cannot address such questions as staging in an abdominal malignancy, and unexpected findings are more common with abdominal than chest ultrasound. This could explain why the point-of-care ultrasound did not reduce the use of abdominal CT, the researchers speculated.
They acknowledged that the patients in their sample had an average age of 81 years, raising questions about how well their data could be applied to a younger population. And they noted that they used point-of-care ultrasound frequently, so they were particularly adept with it. “We use it almost every day in our clinical practice,” said Dr. Barchiesi.
Those factors may have played a key role in the success of point-of-care ultrasound in this study, said Michael Wagner, MD, an assistant professor of medicine at the University of South Carolina, Greenville, who has helped colleagues incorporate ultrasound into their practices.
Elderly patients often present with multiple comorbidities and atypical signs and symptoms, he said. “Sometimes they can be very confusing as to the underlying clinical picture. Ultrasound is being used frequently to better assess these complicated patients.”
Dr. Wagner said extensive training is required to use point-of-care ultrasound accurately.
Dr. Barchiesi also acknowledged that the devices used in this study were large portable machines, not the simpler and less expensive hand-held versions that are also available for similar purposes.
Point-of-care ultrasound is a promising innovation, said Thomas Melgar, MD, a professor of medicine at Western Michigan University, Kalamazoo. “The advantage is that the exam is being done by someone who knows the patient and specifically what they’re looking for. It’s done at the bedside so you don’t have to move the patient.”
The study could help address opposition to internal medicine residents being trained in the technique, he said, adding that “I think it’s very exciting.”
The study was partially supported by Philips, which provided the ultrasound devices. Dr. Barchiesi, Dr. Melgar, and Dr. Wagner disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
researchers say.
“It’s a safe and very useful tool,” Marco Barchiesi, MD, an internal medicine resident at Luigi Sacco Hospital in Milan, said in an interview. “We had a great reduction in chest x-rays because of the use of ultrasound.”
The finding addresses concerns that ultrasound used in primary care could consume more health care resources or put patients at risk.
Dr. Barchiesi and colleagues published their findings July 20 in the European Journal of Internal Medicine.
Point-of-care ultrasound has become increasingly common as miniaturization of devices has made them more portable. The approach has caught on particularly in emergency departments where quick decisions are of the essence.
Its use in internal medicine has been more controversial, with concerns raised that improperly trained practitioners may miss diagnoses or refer patients for unnecessary tests as a result of uncertainty about their findings.
To measure the effect of point-of-care ultrasound in an internal medicine hospital ward, Dr. Barchiesi and colleagues alternated months when point-of-care ultrasound was allowed with months when it was not allowed, for a total of 4 months each, on an internal medicine unit. They allowed the ultrasound to be used for invasive procedures and excluded patients whose critical condition made point-of-care ultrasound crucial.
The researchers analyzed data on 263 patients in the “on” months when point-of-care ultrasound was used, and 255 in the “off” months when it wasn’t used. The two groups were well balanced in age, sex, comorbidity, and clinical impairment.
During the on months, the internists ordered 113 diagnostic tests (0.43 per patient). During the off months they ordered 329 tests (1.29 per patient).
The odds of being referred for a chest x-ray were 87% less in the “on” months, compared with the off months, a statistically significant finding (P < .001). The risk for a chest CT scan and abdominal ultrasound were also reduced during the on months, but the risk for an abdominal CT was increased.
Nineteen patients died during the o” months and 10 during the off months, a difference that was not statistically significant (P = .15). The median length of stay in the hospital was almost the same for the two groups: 9 days for the on months and 9 days for the off months. The difference was also not statistically significant (P = .094).
Point-of-care ultrasound is particularly accurate in identifying cardiac abnormalities and pleural fluid and pneumonia, and it can be used effectively for monitoring heart conditions, the researchers wrote. This could explain the reduction in chest x-rays and CT scans.
On the other hand, ultrasound cannot address such questions as staging in an abdominal malignancy, and unexpected findings are more common with abdominal than chest ultrasound. This could explain why the point-of-care ultrasound did not reduce the use of abdominal CT, the researchers speculated.
They acknowledged that the patients in their sample had an average age of 81 years, raising questions about how well their data could be applied to a younger population. And they noted that they used point-of-care ultrasound frequently, so they were particularly adept with it. “We use it almost every day in our clinical practice,” said Dr. Barchiesi.
Those factors may have played a key role in the success of point-of-care ultrasound in this study, said Michael Wagner, MD, an assistant professor of medicine at the University of South Carolina, Greenville, who has helped colleagues incorporate ultrasound into their practices.
Elderly patients often present with multiple comorbidities and atypical signs and symptoms, he said. “Sometimes they can be very confusing as to the underlying clinical picture. Ultrasound is being used frequently to better assess these complicated patients.”
Dr. Wagner said extensive training is required to use point-of-care ultrasound accurately.
Dr. Barchiesi also acknowledged that the devices used in this study were large portable machines, not the simpler and less expensive hand-held versions that are also available for similar purposes.
Point-of-care ultrasound is a promising innovation, said Thomas Melgar, MD, a professor of medicine at Western Michigan University, Kalamazoo. “The advantage is that the exam is being done by someone who knows the patient and specifically what they’re looking for. It’s done at the bedside so you don’t have to move the patient.”
The study could help address opposition to internal medicine residents being trained in the technique, he said, adding that “I think it’s very exciting.”
The study was partially supported by Philips, which provided the ultrasound devices. Dr. Barchiesi, Dr. Melgar, and Dr. Wagner disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
researchers say.
“It’s a safe and very useful tool,” Marco Barchiesi, MD, an internal medicine resident at Luigi Sacco Hospital in Milan, said in an interview. “We had a great reduction in chest x-rays because of the use of ultrasound.”
The finding addresses concerns that ultrasound used in primary care could consume more health care resources or put patients at risk.
Dr. Barchiesi and colleagues published their findings July 20 in the European Journal of Internal Medicine.
Point-of-care ultrasound has become increasingly common as miniaturization of devices has made them more portable. The approach has caught on particularly in emergency departments where quick decisions are of the essence.
Its use in internal medicine has been more controversial, with concerns raised that improperly trained practitioners may miss diagnoses or refer patients for unnecessary tests as a result of uncertainty about their findings.
To measure the effect of point-of-care ultrasound in an internal medicine hospital ward, Dr. Barchiesi and colleagues alternated months when point-of-care ultrasound was allowed with months when it was not allowed, for a total of 4 months each, on an internal medicine unit. They allowed the ultrasound to be used for invasive procedures and excluded patients whose critical condition made point-of-care ultrasound crucial.
The researchers analyzed data on 263 patients in the “on” months when point-of-care ultrasound was used, and 255 in the “off” months when it wasn’t used. The two groups were well balanced in age, sex, comorbidity, and clinical impairment.
During the on months, the internists ordered 113 diagnostic tests (0.43 per patient). During the off months they ordered 329 tests (1.29 per patient).
The odds of being referred for a chest x-ray were 87% less in the “on” months, compared with the off months, a statistically significant finding (P < .001). The risk for a chest CT scan and abdominal ultrasound were also reduced during the on months, but the risk for an abdominal CT was increased.
Nineteen patients died during the o” months and 10 during the off months, a difference that was not statistically significant (P = .15). The median length of stay in the hospital was almost the same for the two groups: 9 days for the on months and 9 days for the off months. The difference was also not statistically significant (P = .094).
Point-of-care ultrasound is particularly accurate in identifying cardiac abnormalities and pleural fluid and pneumonia, and it can be used effectively for monitoring heart conditions, the researchers wrote. This could explain the reduction in chest x-rays and CT scans.
On the other hand, ultrasound cannot address such questions as staging in an abdominal malignancy, and unexpected findings are more common with abdominal than chest ultrasound. This could explain why the point-of-care ultrasound did not reduce the use of abdominal CT, the researchers speculated.
They acknowledged that the patients in their sample had an average age of 81 years, raising questions about how well their data could be applied to a younger population. And they noted that they used point-of-care ultrasound frequently, so they were particularly adept with it. “We use it almost every day in our clinical practice,” said Dr. Barchiesi.
Those factors may have played a key role in the success of point-of-care ultrasound in this study, said Michael Wagner, MD, an assistant professor of medicine at the University of South Carolina, Greenville, who has helped colleagues incorporate ultrasound into their practices.
Elderly patients often present with multiple comorbidities and atypical signs and symptoms, he said. “Sometimes they can be very confusing as to the underlying clinical picture. Ultrasound is being used frequently to better assess these complicated patients.”
Dr. Wagner said extensive training is required to use point-of-care ultrasound accurately.
Dr. Barchiesi also acknowledged that the devices used in this study were large portable machines, not the simpler and less expensive hand-held versions that are also available for similar purposes.
Point-of-care ultrasound is a promising innovation, said Thomas Melgar, MD, a professor of medicine at Western Michigan University, Kalamazoo. “The advantage is that the exam is being done by someone who knows the patient and specifically what they’re looking for. It’s done at the bedside so you don’t have to move the patient.”
The study could help address opposition to internal medicine residents being trained in the technique, he said, adding that “I think it’s very exciting.”
The study was partially supported by Philips, which provided the ultrasound devices. Dr. Barchiesi, Dr. Melgar, and Dr. Wagner disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
New oral anticoagulants drive ACC consensus on bleeding
Patients on oral anticoagulants who experience a bleeding event may be able to discontinue therapy if certain circumstances apply, according to updated guidance from the American College of Cardiology.
The emergence of direct-acting oral anticoagulants (DOACs) to prevent venous thromboembolism and the introduction of new reversal strategies for factor Xa inhibitors prompted the creation of an Expert Consensus Decision Pathway to update the version from 2017, according to the ACC. Expert consensus decision pathways (ECDPs) are a component of the solution sets issued by the ACC to “address key questions facing care teams and attempt to provide practical guidance to be applied at the point of care.”
In an ECDP published in the Journal of the American College of Cardiology, the writing committee members developed treatment algorithms for managing bleeding in patients on DOACs and vitamin K antagonists (VKAs).
Bleeding was classified as major or nonmajor, with major defined as “bleeding that is associated with hemodynamic compromise, occurs in an anatomically critical site, requires transfusion of at least 2 units of packed red blood cells [RBCs]), or results in a hemoglobin drop greater than 2 g/dL. All other types of bleeding were classified as nonmajor.
The document includes a graphic algorithm for assessing bleed severity and managing major versus nonmajor bleeding, and a separate graphic describes considerations for reversal and use of hemostatic agents according to whether the patient is taking a VKA (warfarin and other coumarins), a direct thrombin inhibitor (dabigatran), the factor Xa inhibitors apixaban and rivaroxaban, or the factor Xa inhibitors betrixaban and edoxaban.
Another algorithm outlines whether to discontinue, delay, or restart anticoagulation. Considerations for restarting anticoagulation include whether the patient is pregnant, awaiting an invasive procedure, not able to receive medication by mouth, has a high risk of rebleeding, or is being bridged back to a vitamin K antagonist with high thrombotic risk.
In most cases of GI bleeding, for example, current data support restarting oral anticoagulants once hemostasis is achieved, but patients who experience intracranial hemorrhage should delay restarting any anticoagulation for at least 4 weeks if they are without high thrombotic risk, according to the document.
The report also recommends clinician-patient discussion before resuming anticoagulation, ideally with time allowed for patients to develop questions. Discussions should include the signs of bleeding, assessment of risk for a thromboembolic event, and the benefits of anticoagulation.
“The proliferation of oral anticoagulants (warfarin and DOACs) and growing indications for their use prompted the need for guidance on the management of these drugs,” said Gordon F. Tomaselli, MD, chair of the writing committee, in an interview. “This document provides guidance on management at the time of a bleeding complication. This includes acute management, starting and stopping drugs, and use of reversal agents,” he said. “This of course will be a dynamic document as the list of these drugs and their antidotes expand,” he noted.
“The biggest change from the previous guidelines are twofold: an update on laboratory assessment to monitor drug levels and use of reversal agents,” while the acute management strategies have otherwise remained similar to previous documents, said Dr. Tomaselli.
Dr. Tomaselli said that he was not surprised by the biological aspects of recent research while developing the statement. However, “the extent of the use of multiple anticoagulants and antiplatelet agents was a bit surprising and complicates therapy with each of the agents,” he noted.
The way the pathways are presented may make them challenging to follow in clinical practice, said Dr. Tomaselli. “The pathways are described linearly and in practice often many things have to happen at once,” he said. “The other main issue may be limitations in the availability of some of the newer reversal agents,” he added.
“The complication of bleeding is difficult to avoid,” said Dr. Tomaselli, and for future research, “the focus needs to continue to refine the indications for anticoagulation and appropriate use with other drugs that predispose to bleeding. We also need better methods and testing to monitor drugs levels and the effect on coagulation,” he said.
In accordance with the ACC Solution Set Oversight Committee, the writing committee members, including Dr. Tomaselli, had no relevant relationships with industry to disclose.
SOURCE: Tomaselli GF et al. J Am Coll Cardiol. 2020. doi: 10.1016/j.jacc.2020.04.053.
Patients on oral anticoagulants who experience a bleeding event may be able to discontinue therapy if certain circumstances apply, according to updated guidance from the American College of Cardiology.
The emergence of direct-acting oral anticoagulants (DOACs) to prevent venous thromboembolism and the introduction of new reversal strategies for factor Xa inhibitors prompted the creation of an Expert Consensus Decision Pathway to update the version from 2017, according to the ACC. Expert consensus decision pathways (ECDPs) are a component of the solution sets issued by the ACC to “address key questions facing care teams and attempt to provide practical guidance to be applied at the point of care.”
In an ECDP published in the Journal of the American College of Cardiology, the writing committee members developed treatment algorithms for managing bleeding in patients on DOACs and vitamin K antagonists (VKAs).
Bleeding was classified as major or nonmajor, with major defined as “bleeding that is associated with hemodynamic compromise, occurs in an anatomically critical site, requires transfusion of at least 2 units of packed red blood cells [RBCs]), or results in a hemoglobin drop greater than 2 g/dL. All other types of bleeding were classified as nonmajor.
The document includes a graphic algorithm for assessing bleed severity and managing major versus nonmajor bleeding, and a separate graphic describes considerations for reversal and use of hemostatic agents according to whether the patient is taking a VKA (warfarin and other coumarins), a direct thrombin inhibitor (dabigatran), the factor Xa inhibitors apixaban and rivaroxaban, or the factor Xa inhibitors betrixaban and edoxaban.
Another algorithm outlines whether to discontinue, delay, or restart anticoagulation. Considerations for restarting anticoagulation include whether the patient is pregnant, awaiting an invasive procedure, not able to receive medication by mouth, has a high risk of rebleeding, or is being bridged back to a vitamin K antagonist with high thrombotic risk.
In most cases of GI bleeding, for example, current data support restarting oral anticoagulants once hemostasis is achieved, but patients who experience intracranial hemorrhage should delay restarting any anticoagulation for at least 4 weeks if they are without high thrombotic risk, according to the document.
The report also recommends clinician-patient discussion before resuming anticoagulation, ideally with time allowed for patients to develop questions. Discussions should include the signs of bleeding, assessment of risk for a thromboembolic event, and the benefits of anticoagulation.
“The proliferation of oral anticoagulants (warfarin and DOACs) and growing indications for their use prompted the need for guidance on the management of these drugs,” said Gordon F. Tomaselli, MD, chair of the writing committee, in an interview. “This document provides guidance on management at the time of a bleeding complication. This includes acute management, starting and stopping drugs, and use of reversal agents,” he said. “This of course will be a dynamic document as the list of these drugs and their antidotes expand,” he noted.
“The biggest change from the previous guidelines are twofold: an update on laboratory assessment to monitor drug levels and use of reversal agents,” while the acute management strategies have otherwise remained similar to previous documents, said Dr. Tomaselli.
Dr. Tomaselli said that he was not surprised by the biological aspects of recent research while developing the statement. However, “the extent of the use of multiple anticoagulants and antiplatelet agents was a bit surprising and complicates therapy with each of the agents,” he noted.
The way the pathways are presented may make them challenging to follow in clinical practice, said Dr. Tomaselli. “The pathways are described linearly and in practice often many things have to happen at once,” he said. “The other main issue may be limitations in the availability of some of the newer reversal agents,” he added.
“The complication of bleeding is difficult to avoid,” said Dr. Tomaselli, and for future research, “the focus needs to continue to refine the indications for anticoagulation and appropriate use with other drugs that predispose to bleeding. We also need better methods and testing to monitor drugs levels and the effect on coagulation,” he said.
In accordance with the ACC Solution Set Oversight Committee, the writing committee members, including Dr. Tomaselli, had no relevant relationships with industry to disclose.
SOURCE: Tomaselli GF et al. J Am Coll Cardiol. 2020. doi: 10.1016/j.jacc.2020.04.053.
Patients on oral anticoagulants who experience a bleeding event may be able to discontinue therapy if certain circumstances apply, according to updated guidance from the American College of Cardiology.
The emergence of direct-acting oral anticoagulants (DOACs) to prevent venous thromboembolism and the introduction of new reversal strategies for factor Xa inhibitors prompted the creation of an Expert Consensus Decision Pathway to update the version from 2017, according to the ACC. Expert consensus decision pathways (ECDPs) are a component of the solution sets issued by the ACC to “address key questions facing care teams and attempt to provide practical guidance to be applied at the point of care.”
In an ECDP published in the Journal of the American College of Cardiology, the writing committee members developed treatment algorithms for managing bleeding in patients on DOACs and vitamin K antagonists (VKAs).
Bleeding was classified as major or nonmajor, with major defined as “bleeding that is associated with hemodynamic compromise, occurs in an anatomically critical site, requires transfusion of at least 2 units of packed red blood cells [RBCs]), or results in a hemoglobin drop greater than 2 g/dL. All other types of bleeding were classified as nonmajor.
The document includes a graphic algorithm for assessing bleed severity and managing major versus nonmajor bleeding, and a separate graphic describes considerations for reversal and use of hemostatic agents according to whether the patient is taking a VKA (warfarin and other coumarins), a direct thrombin inhibitor (dabigatran), the factor Xa inhibitors apixaban and rivaroxaban, or the factor Xa inhibitors betrixaban and edoxaban.
Another algorithm outlines whether to discontinue, delay, or restart anticoagulation. Considerations for restarting anticoagulation include whether the patient is pregnant, awaiting an invasive procedure, not able to receive medication by mouth, has a high risk of rebleeding, or is being bridged back to a vitamin K antagonist with high thrombotic risk.
In most cases of GI bleeding, for example, current data support restarting oral anticoagulants once hemostasis is achieved, but patients who experience intracranial hemorrhage should delay restarting any anticoagulation for at least 4 weeks if they are without high thrombotic risk, according to the document.
The report also recommends clinician-patient discussion before resuming anticoagulation, ideally with time allowed for patients to develop questions. Discussions should include the signs of bleeding, assessment of risk for a thromboembolic event, and the benefits of anticoagulation.
“The proliferation of oral anticoagulants (warfarin and DOACs) and growing indications for their use prompted the need for guidance on the management of these drugs,” said Gordon F. Tomaselli, MD, chair of the writing committee, in an interview. “This document provides guidance on management at the time of a bleeding complication. This includes acute management, starting and stopping drugs, and use of reversal agents,” he said. “This of course will be a dynamic document as the list of these drugs and their antidotes expand,” he noted.
“The biggest change from the previous guidelines are twofold: an update on laboratory assessment to monitor drug levels and use of reversal agents,” while the acute management strategies have otherwise remained similar to previous documents, said Dr. Tomaselli.
Dr. Tomaselli said that he was not surprised by the biological aspects of recent research while developing the statement. However, “the extent of the use of multiple anticoagulants and antiplatelet agents was a bit surprising and complicates therapy with each of the agents,” he noted.
The way the pathways are presented may make them challenging to follow in clinical practice, said Dr. Tomaselli. “The pathways are described linearly and in practice often many things have to happen at once,” he said. “The other main issue may be limitations in the availability of some of the newer reversal agents,” he added.
“The complication of bleeding is difficult to avoid,” said Dr. Tomaselli, and for future research, “the focus needs to continue to refine the indications for anticoagulation and appropriate use with other drugs that predispose to bleeding. We also need better methods and testing to monitor drugs levels and the effect on coagulation,” he said.
In accordance with the ACC Solution Set Oversight Committee, the writing committee members, including Dr. Tomaselli, had no relevant relationships with industry to disclose.
SOURCE: Tomaselli GF et al. J Am Coll Cardiol. 2020. doi: 10.1016/j.jacc.2020.04.053.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY






