User login
Higher industriousness reduces risk of predementia syndrome in older adults
Higher industriousness was associated with a 25% reduced risk of concurrent motoric cognitive risk syndrome (MCR), based on data from approximately 6,000 individuals.
Previous research supports an association between conscientiousness and a lower risk of MCR, a form of predementia that involves slow gait speed and cognitive complaints, wrote Yannick Stephan, PhD, of the University of Montpellier (France), and colleagues. However, the specific facets of conscientiousness that impact MCR have not been examined.
In a study published in the Journal of Psychiatric Research, the authors reviewed data from 6,001 dementia-free adults aged 65-99 years who were enrolled in the Health and Retirement Study, a nationally representative longitudinal study of adults aged 50 years and older in the United States.
Baseline data were collected between 2008 and 2010, and participants were assessed for MCR at follow-up points during 2012-2014 and 2016-2018. Six facets of conscientiousness were assessed using a 24-item scale that has been used in previous studies. The six facets were industriousness, self-control, order, traditionalism, virtue, and responsibility. The researchers controlled for variables including demographic factors, cognition, physical activity, disease burden, depressive symptoms, and body mass index.
Overall, increased industriousness was significantly associated with a lower likelihood of concurrent MCR (odds ratio, 0.75) and a reduced risk of incident MCR (hazard ratio, 0.63,; P < .001 for both).
The conscientiousness facets of order, self-control, and responsibility also were associated with a lower likelihood of both concurrent and incident MCR, with ORs ranging from 0.82-0.88 for concurrent and HRs ranging from 0.72-0.82 for incident.
Traditionalism and virtue were significantly associated with a lower risk of incident MCR, but not concurrent MCR (HR, 0.84; P < .01 for both).
The mechanism of action for the association may be explained by several cognitive, health-related, behavioral, and psychological pathways, the researchers wrote. With regard to industriousness, the relationship could be partly explained by cognition, physical activity, disease burden, BMI, and depressive symptoms. However, industriousness also has been associated with a reduced risk of systemic inflammation, which may in turn reduce MCR risk. Also, data suggest that industriousness and MCR share a common genetic cause.
The study findings were limited by several factors including the observational design and the positive selection effect from patients with complete follow-up data, as these patients likely have higher levels of order, industriousness, and responsibility, the researchers noted. However, the results support those from previous studies and were strengthened by the large sample and examination of six facets of conscientiousness.
“This study thus provides a more detailed understanding of the specific components of conscientiousness that are associated with risk of MCR among older adults,” and the facets could be targeted in interventions to reduce both MCR and dementia, they concluded.
The Health and Retirement Study is supported by the National Institute on Aging and conducted by the University of Michigan. The current study was supported in part by the National Institutes of Health. The researchers had no financial conflicts to disclose.
Higher industriousness was associated with a 25% reduced risk of concurrent motoric cognitive risk syndrome (MCR), based on data from approximately 6,000 individuals.
Previous research supports an association between conscientiousness and a lower risk of MCR, a form of predementia that involves slow gait speed and cognitive complaints, wrote Yannick Stephan, PhD, of the University of Montpellier (France), and colleagues. However, the specific facets of conscientiousness that impact MCR have not been examined.
In a study published in the Journal of Psychiatric Research, the authors reviewed data from 6,001 dementia-free adults aged 65-99 years who were enrolled in the Health and Retirement Study, a nationally representative longitudinal study of adults aged 50 years and older in the United States.
Baseline data were collected between 2008 and 2010, and participants were assessed for MCR at follow-up points during 2012-2014 and 2016-2018. Six facets of conscientiousness were assessed using a 24-item scale that has been used in previous studies. The six facets were industriousness, self-control, order, traditionalism, virtue, and responsibility. The researchers controlled for variables including demographic factors, cognition, physical activity, disease burden, depressive symptoms, and body mass index.
Overall, increased industriousness was significantly associated with a lower likelihood of concurrent MCR (odds ratio, 0.75) and a reduced risk of incident MCR (hazard ratio, 0.63,; P < .001 for both).
The conscientiousness facets of order, self-control, and responsibility also were associated with a lower likelihood of both concurrent and incident MCR, with ORs ranging from 0.82-0.88 for concurrent and HRs ranging from 0.72-0.82 for incident.
Traditionalism and virtue were significantly associated with a lower risk of incident MCR, but not concurrent MCR (HR, 0.84; P < .01 for both).
The mechanism of action for the association may be explained by several cognitive, health-related, behavioral, and psychological pathways, the researchers wrote. With regard to industriousness, the relationship could be partly explained by cognition, physical activity, disease burden, BMI, and depressive symptoms. However, industriousness also has been associated with a reduced risk of systemic inflammation, which may in turn reduce MCR risk. Also, data suggest that industriousness and MCR share a common genetic cause.
The study findings were limited by several factors including the observational design and the positive selection effect from patients with complete follow-up data, as these patients likely have higher levels of order, industriousness, and responsibility, the researchers noted. However, the results support those from previous studies and were strengthened by the large sample and examination of six facets of conscientiousness.
“This study thus provides a more detailed understanding of the specific components of conscientiousness that are associated with risk of MCR among older adults,” and the facets could be targeted in interventions to reduce both MCR and dementia, they concluded.
The Health and Retirement Study is supported by the National Institute on Aging and conducted by the University of Michigan. The current study was supported in part by the National Institutes of Health. The researchers had no financial conflicts to disclose.
Higher industriousness was associated with a 25% reduced risk of concurrent motoric cognitive risk syndrome (MCR), based on data from approximately 6,000 individuals.
Previous research supports an association between conscientiousness and a lower risk of MCR, a form of predementia that involves slow gait speed and cognitive complaints, wrote Yannick Stephan, PhD, of the University of Montpellier (France), and colleagues. However, the specific facets of conscientiousness that impact MCR have not been examined.
In a study published in the Journal of Psychiatric Research, the authors reviewed data from 6,001 dementia-free adults aged 65-99 years who were enrolled in the Health and Retirement Study, a nationally representative longitudinal study of adults aged 50 years and older in the United States.
Baseline data were collected between 2008 and 2010, and participants were assessed for MCR at follow-up points during 2012-2014 and 2016-2018. Six facets of conscientiousness were assessed using a 24-item scale that has been used in previous studies. The six facets were industriousness, self-control, order, traditionalism, virtue, and responsibility. The researchers controlled for variables including demographic factors, cognition, physical activity, disease burden, depressive symptoms, and body mass index.
Overall, increased industriousness was significantly associated with a lower likelihood of concurrent MCR (odds ratio, 0.75) and a reduced risk of incident MCR (hazard ratio, 0.63,; P < .001 for both).
The conscientiousness facets of order, self-control, and responsibility also were associated with a lower likelihood of both concurrent and incident MCR, with ORs ranging from 0.82-0.88 for concurrent and HRs ranging from 0.72-0.82 for incident.
Traditionalism and virtue were significantly associated with a lower risk of incident MCR, but not concurrent MCR (HR, 0.84; P < .01 for both).
The mechanism of action for the association may be explained by several cognitive, health-related, behavioral, and psychological pathways, the researchers wrote. With regard to industriousness, the relationship could be partly explained by cognition, physical activity, disease burden, BMI, and depressive symptoms. However, industriousness also has been associated with a reduced risk of systemic inflammation, which may in turn reduce MCR risk. Also, data suggest that industriousness and MCR share a common genetic cause.
The study findings were limited by several factors including the observational design and the positive selection effect from patients with complete follow-up data, as these patients likely have higher levels of order, industriousness, and responsibility, the researchers noted. However, the results support those from previous studies and were strengthened by the large sample and examination of six facets of conscientiousness.
“This study thus provides a more detailed understanding of the specific components of conscientiousness that are associated with risk of MCR among older adults,” and the facets could be targeted in interventions to reduce both MCR and dementia, they concluded.
The Health and Retirement Study is supported by the National Institute on Aging and conducted by the University of Michigan. The current study was supported in part by the National Institutes of Health. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRIC RESEARCH
Managing TIA: Early action and essential risk-reduction steps
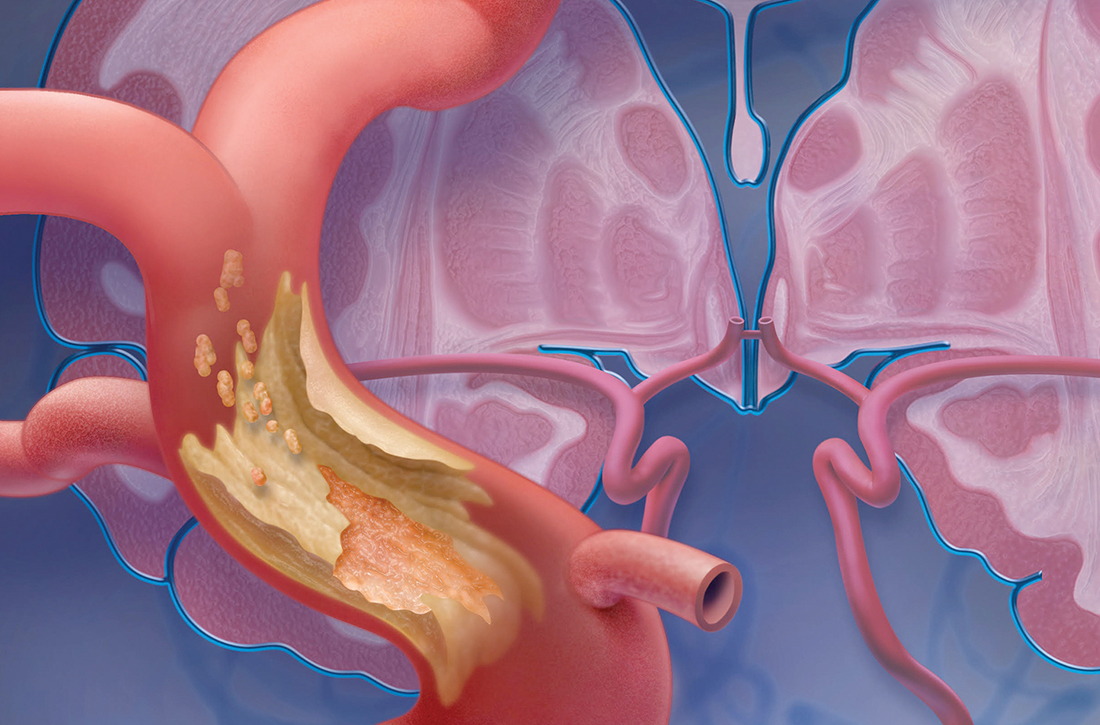
As many as 240,000 people per year in the United States experience a transient ischemic attack (TIA),1,2 which is now defined by the American Heart Association and American Stroke Association as a “transient episode of neurological dysfunction caused by focal brain, spinal cord, or retinal ischemia, without acute infarction.”3 An older definition of TIA was based on the duration of the event (ie, resolution of symptoms at 24 hours); in the updated (2009) definition, the diagnostic criterion is the extent of focal tissue damage.3 Using the 2009 definition might mean a decrease in the number of patients who have a diagnosis of a TIA and an increase in the number who are determined to have had a stroke because an infarction is found on initial imaging.
Guided by the 2009 revised definition of a TIA, we review here the work-up and treatment of TIA, emphasizing immediacy of management to (1) prevent further tissue damage and (2) decrease the risk of a second event.
CASE
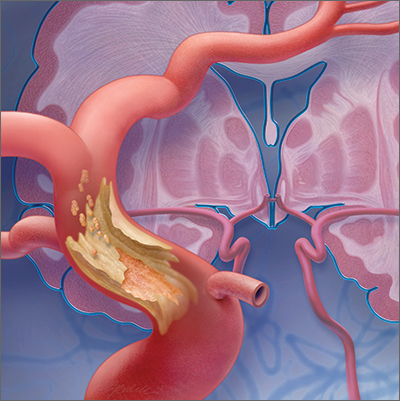
Martin L, 69 years old, retired, a nonsmoker, and with a history of peripheral arterial disease and hypercholesterolemia, presents to the emergency department (ED) of a rural hospital complaining of slurred speech and left-side facial numbness. He had an episode of facial numbness that lasted 30 minutes, then resolved, each of the 2 previous evenings; he did not seek care at those times. Now, in the ED, Mr. L is normotensive.
The patient’s medication history includes a selective serotonin reuptake inhibitor and melatonin to improve sleep. He reports having discontinued a statin because he could not tolerate its adverse effects.
What immediate steps are recommended for Mr. L’s care?
Common event callsfor quick action
A TIA is the strongest predictor of subsequent stroke and stroke-related death; the highest period of risk of these devastating outcomes is immediately following a TIA.1,2,4,5 It is essential, therefore, for the physician who sees a patient with a current complaint or recent history of suspected focal neurologic deficits to direct that patient to an ED for an accurate diagnosis and, as appropriate, early treatment for the best possible outcome.
Imaging—preferably, diffusion-weighted magnetic resonance imaging (DW-MRI), the gold standard for diagnosing stroke (see “Diagnosis includes ruling out mimics”)2,3—should be performed as soon as the patient with a suspected TIA arrives in the ED. Imaging should not be held while waiting for a stroke to declare itself—ie, by allowing symptoms to persist for longer than 24 hours. 6
Continue to: Late presentation
Late presentation. Some patients present ≥ 48 hours after onset of early symptoms of a TIA; for them, the work-up is the same as for prompt presentation but can be completed in the outpatient clinic—as long as the patient is stable clinically and imaging is accessible there. DW-MRI should be completed within 48 hours after late presentation. In such cases, the patient should be cautioned regarding risks and any recurrence of symptoms.7,8
Diagnosis includes ruling out mimics
All patients in whom a stroke is suspected should be evaluated on an emergency basis with brain imaging upon arrival at the hospital, before any therapy is initiated. As noted, DW-MRI is the preferred modality; noncontrast computed tomography (CT) or CT angiography can be used if MRI is unavailable.2,3
Mimics. Stroke has many mimics; quickly eliminating them from the differential diagnosis is important so that appropriate therapy can be initiated. Mimics usually have a prolonged presentation of symptoms, whereas the presentation of a TIA is usually abrupt. The 3 more common diagnoses that mimic a TIA are migraine with aura, seizure, and syncope.9,10 Symptoms that generally are not associated with a TIA are chest pain, generalized weakness, and confusion.11 A complete history and physical exam provide the path to the imaging, laboratory, and cardiac testing that is needed to differentiate these diagnoses from a TIA.
A thorough history is best obtained from the patient and a witness, if available, and should include identification of any focal neurologic deficits and the duration and time to resolution of symptoms. Obtain a history of risk factors for ischemia—tobacco use, diabetes, obesity, dyslipidemia, hypertension, previous TIA or stroke, atrial fibrillation, and any coagulopathy. Ask questions about a family history of TIA, stroke, and coagulopathy.11
A comprehensive physical exam, including vital signs, cardiac exam, a check for carotid bruits, and complete neurologic exam, should be performed. Most patients present with concerns for unilateral weakness and changes in speech, which are usually associated with infarction on DW-MRI.12 The most common findings on physical exam include cranial nerve abnormalities, such as diplopia, hemianopia, monocular blindness, disconjugate gaze, facial drooping, lateral tongue movement, dysphagia, and vestibular dysfunction. Cerebellar abnormalities are also often noted, including past pointing, dystaxia, ataxia, nystagmus, and motor abnormalities (eg, spasticity, clonus, or unilateral weakness in the face or extremities).11
Electrocardiography at the bedside can confirm atrial fibrillation or another arrhythmia quickly.
Essential laboratory testing includes measurement of blood glucose and serum electrolytes to determine if these particular imbalances are the cause of symptoms. The presence of a hypercoaguable state is determined by a complete blood count and coagulation studies.3,13 Urine toxicology should also be obtained to rule out other causes of symptoms. A lipid profile is beneficial for making long-term treatment decisions.
Continue to: ABCD2 score
ABCD2 score. Patients who have had a TIA and present within 72 hours after symptoms have resolved should be hospitalized if they have an ABCD2 (Age, Blood pressure [BP], Clinical presentation, Diabetes mellitus [type 1 or 2], Duration of symptoms) prediction system score > 3.14 ABCD2 criteria can be used to help identify patients who are at higher risk of stroke or need further therapy (TABLE 1).14,15
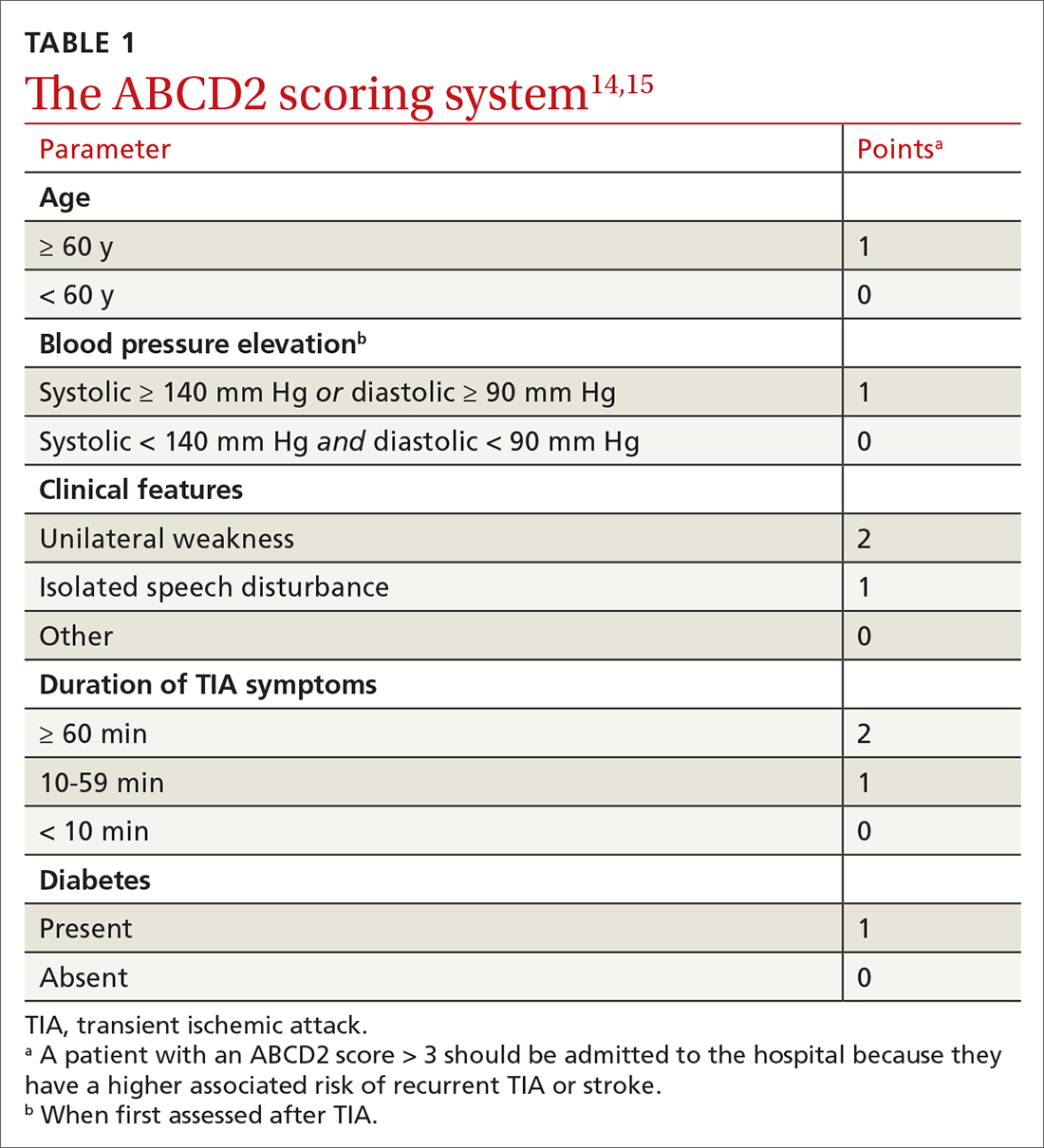
The ABCD2 score is also used to determine whether a patient needs dual antiplatelet therapy. Patients who score at the higher end of the ABCD2 system usually have an increased risk of stroke, longer hospitalization, and greater disability.
CASE
In the ED, Mr. L is immediately assessed and airlifted to a larger regional medical center, where MRI confirms a stroke.
Management
Initial management of a TIA is aimed at reducing the risk of recurrent TIA or stroke. Early medical and possibly surgical treatment are key for preventing stroke and improving outcomes. The first 48 hours after a TIA are the most critical because the incidence of recurrent TIA or stroke is highest during this period.16-18
What is the accepted strategy for early treatment?
Initial treatment must include antiplatelet therapy, BP management, anticoagulation, statin therapy, and carotid endarterectomy as indicated.2,19,20 Control of hypertension and anticoagulation decrease the risk of recurrent stroke by the largest margin20; both are “A”-level Strength of Recommendation Taxonomy interventions.2,3
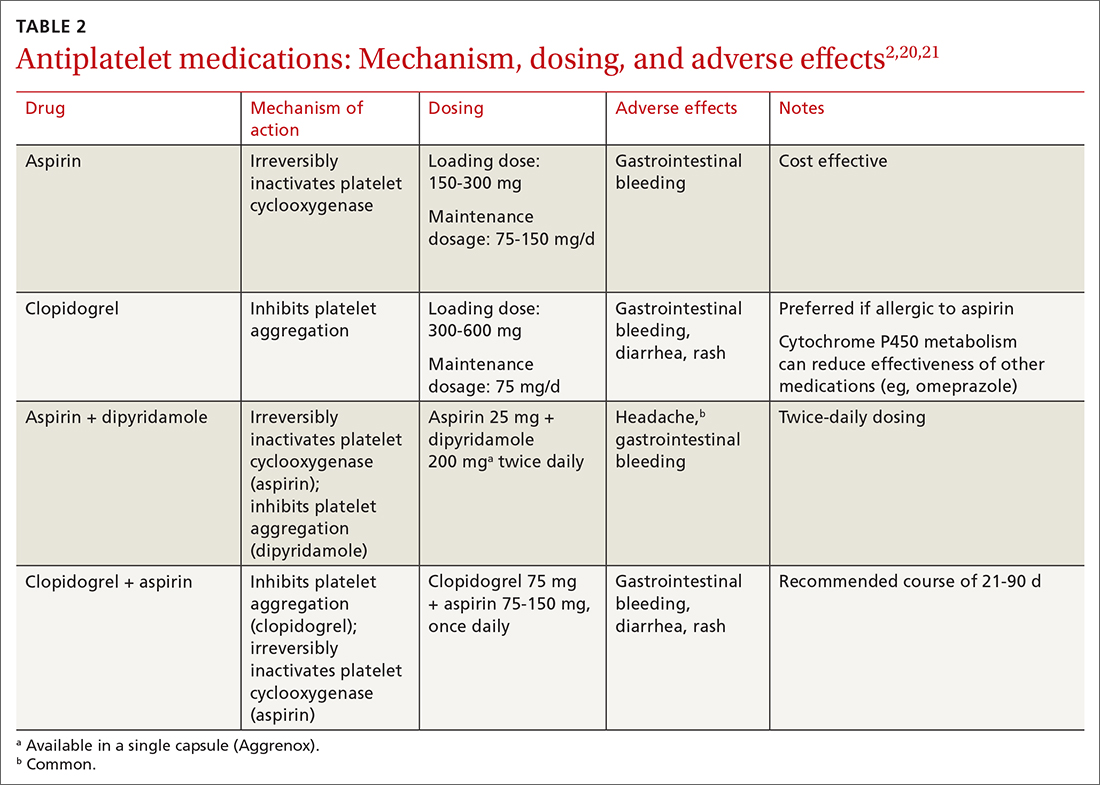
Step 1: Antiplatelet therapy. After initial imaging is complete and if there are no contraindications, antiplatelet agents are recommended for patients who have had a noncardioembolic TIA. The American Heart Association and American Stroke Association recommend either aspirin, clopidogrel, dipyridamole + aspirin (available in a single capsule [Aggrenox]), or clopidogrel + aspirin as first-line therapy.2,20 The choice of agent needs to be individualized, based on tolerability and adverse effects (TABLE 22,20,21).
A meta-analysis of antiplatelet therapy reviewed the optimum dosing of each medication.21,22 Reduction of the risk of ischemic stroke with aspirin is 21% to 22% at the optimal dosing of 75 to 150 mg/d, which also reduces the risk of gastrointestinal bleeding.
Continue to: For a patient who has...
For a patient who has an ABCD2 score ≥ 4, has had a prior TIA, or has large-vessel disease, dual antiplatelet therapy is recommended for the first 21 days, with a subsequent return to monotherapy. Dual antiplatelet therapy of clopidogrel + aspirin increases the risk of adverse reactions and has not been shown to have greater long-term benefit23-25 (TABLE 22,20,21).
Step 2: BP management. This is the next immediate step. As many as 80% of patients who present with a TIA have elevated BP upon admission. BP needs to be treated and carefully monitored during this early treatment phase. The recommendation is for a systolic BP < 185 mm Hg and a diastolic BP < 110 mm Hg.24
Step 3: Anticoagulation. Treatment with warfarin or a direct oral anticoagulant (DOAC) is recommended for patients who have the potential for forming emboli—eg, in the setting of atrial fibrillation, ventricular thrombus, mechanical heart valve, or venous thromboembolism.
Step 4. High-intensity statin. A statin agent is recommended as part of immediate and long-term medical management, regardless of the low-density lipoprotein cholesterol (LDL-C) level, to reduce the risk of stroke.2,24
Carotid artery management. Surgical intervention is not always considered a component of immediate medical management. However, guidelines recommend that carotid endarterectomy or stenting be considered in patients who have stenosis > 70%.2
CASE
Mr. L is admitted to the hospital and undergoes neurosurgical intervention. Medical management is instituted.
Long-term management and secondary prevention
The main risk factors for stroke can be divided into modifiable, vascular, and unmodifiable. Addressing both modifiable and vascular risks is important for secondary prevention.
Continue to: Modifiable and vascular risk factors
Modifiable and vascular risk factors
Modifiable risk factors for stroke include hypertension, diabetes, dyslipidemia, smoking, and physical activity; the most important of these, for preventing subsequent stroke after an initial TIA, is hypertension.26
The 2 more significant vascular risk factors for stroke are carotid artery stenosis and atrial fibrillation.
Hypertension. Improving control of hypertension can improve secondary risk reduction for recurrent stroke. Control of both systolic and diastolic BP is important in this regard, with larger systolic BP reductions having a greater impact on decreasing the risk of recurrent stroke.24 Evidence supports lowering BP to improve secondary risk reduction in people with and without diagnosed hypertension: The goal is to lower systolic BP by ≥ 10 mm Hg and diastolic BP by 5 mm Hg.24 No particular class of antihypertensive is recommended in the first line, although preliminary evidence shows that a diuretic, with or without an angiotensin-converting enzyme inhibitor, might be more beneficial than other options.24
Diabetes. The risk of cardiovascular disease, including stroke, is higher in people with diabetes. Evidence shows that various (but not all) agents in 2 pharmaceutical classes—glucagon-like peptide-1 (GLP-1) receptor agonists and the sodium glucose-2 cotransporter (SGLT2) inhibitors—reduce the risk of major cardiovascular events and improve secondary prevention of recurrent stroke:
- EMPA-REG OUTCOME (ClinicalTrials.gov Identifier: NCT01131676) was the first trial to show cardiovascular benefit from an SGLT2 inhibitor (empagliflozin); subsequent studies confirmed the cardiovascular benefits found in EMPA-REG OUTCOME.27,28
- The ELIXA trial (ClinicalTrials.gov Identifier: NCT01147250) was the first to show cardiovascular benefit from a GLP-1 receptor agonist (lixisenatide); subsequent studies supported this finding.29,30
Appropriate agents in these 2 classes should be considered as first-line or adjunctive in patients with both diabetes and known cardiovascular disease, as long as there are no contraindications.27,28
Pioglitazone, a thiazolidinedione-class antidiabetic agent, was once considered a potential option to improve secondary prevention of stroke. However, the thiazolidinediones are generally no longer considered; instead, the SGLT2 inhibitors and GLP-1 receptor agonists are favored.31
Evidence demonstrates the effect of hyperglycemia on cardiovascular events; however, it is important to note that hypoglycemia can result in symptoms and focal changes that mimic a stroke. In addition, some evidence suggests that hypoglycemia can increase cardiovascular risk—thereby supporting the importance of strict control of diabetes and maintenance of euglycemia in reducing overall cardiovascular risk.32
Continue to: Lipids
Lipids. The SPARCL trial (ClinicalTrials.gov Identifier: NCT00147602) was the first study to demonstrate the benefit of high-intensity statin therapy—specifically, atorvastatin 80 mg/d—for secondary prevention for recurrent stroke.33 The recommendation is to use high-intensity statin therapy to decrease the risk of recurrent stroke by reducing the level of LDL-C—by ≥ 50% or to < 70 mg/dL, for maximum risk reduction.24,34
The IMPROVE-IT trial (ClinicalTrials.gov Identifier: NCT00202878) demonstrated the benefit of adding ezetimibe, 10 mg/d, to a moderate-to-high-intensity statin (simvastatin, 40-80 mg/d) to reduce the risk of recurrent stroke.35
Results of recent studies support the use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors for regulating levels of LDL-C, as an additional option to consider—if needed to further reduce the LDL-C level or if statins are contraindicated in a particular patient.34
Smoking cessation. Cigarette smoking is known to increase the risk of ischemic stroke; newer evidence shows that second-hand exposure to smoke also increases the risk of ischemic stroke.36,37 Although these studies focused on primary prevention of ischemic stroke, the data can reasonably be applied to secondary prevention.38 The recommendation for secondary prevention is to quit smoking and avoid secondhand smoke.24
Alcohol. Evidence demonstrates that heavy alcohol consumption and alcoholism increase the risk of stroke; similar to what is known about smoking, most available data relate to primary prevention.38 The recommendation for providing secondary stroke prevention is to stop or decrease alcohol intake.24
Weight reduction. Obesity (body mass index > 30) increases the risk of ischemic stroke. However, there is, as yet, no evidence that weight loss diminishes the risk of subsequent stroke for secondary prevention.24
Physical activity. Aerobic exercise and strength-training programs after a stroke improve cardiovascular health and mobility. There is no evidence that exercise leads to a reduction in the risk of subsequent stroke.24
Continue to: Nutrition
Nutrition. No current randomized controlled trials are focused on the relationship between diet and recurrent stroke for purposes of prevention; however, evidence for both BP and lipid control incorporate dietary guidance. Recommendations include reducing intake of saturated fats and of sodium (the latter, to < 2.3 g/d) and increasing intake of fruits and vegetables, both of which are beneficial for controlling BP and lipid levels and promoting overall cardiovascular health.38
Carotid artery stenosis. Several randomized controlled trials have demonstrated benefit from treating carotid stenosis (> 70% stenosis but not < 50%) with carotid endarterectomy to reduce the risk of recurrent stroke after TIA.2 The ideal timing of carotid endarterectomy is still being studied; however, available evidence supports intervention within 2 to 6 weeks after TIA or stroke.25 Studies are ongoing that compare carotid angioplasty and stenting against carotid endarterectomy. Medical therapy, with antiplatelet agents and statins, is recommended after carotid endarterectomy.25
Atrial fibrillation increases the risk of recurrent stroke after a TIA, and is the most important indication for secondary stroke prevention with anticoagulation therapy:
- Warfarin. Several studies have shown that warfarin provides a 68% relative risk reduction and a 1.4% absolute risk reduction in the annual stroke rate.24 To achieve this reduction in risk, the optimal international normalized ratio is 2.5 (range, 2-3).24
- Aspirin provides a 13% relative risk reduction for recurrent stroke, although there is evidence that long-term anticoagulation provides more benefit than aspirin after a TIA.39-41 Optimal dosing of aspirin ranges from 75-100 mg/d; greatest benefit is likely in the 12 weeks after stroke, when the risk of recurrent stroke is highest.31,41,42
- DOACs have similar efficacy to warfarin but more rapid onset, lower risk of bleeding, fewer drug interactions, and no requirement for monitoring—often making them a more tolerable long-term choice. Options are rivaroxaban 20 mg/d, dabigatran 150 mg twice daily, apixaban 5 mg twice daily, and edoxaban 60 mg/d.39
When to start anticoagulation and the choice of agent should be weighed against a risk of bleeding, which is highest after the initial stroke. Cost is also a consideration: DOACs are more expensive than warfarin.
CASE
Mr. L is discharged 3 days after carotid endarterectomy and free of residual deficits. He is started on dual antiplatelet therapy (aspirin + clopidogrel) for 21 days, to be followed by a return to monotherapy. He is restarted on a high-intensity statin. He is instructed to resume taking the selective serotonin reuptake inhibitor and melatonin for sleep, as needed. Last, he is told to schedule follow-up with his primary care physician in 7 to 10 days to begin post-stroke care.
Final thoughts
Primary care physicians are often the first point of contact for patients with current or remote TIA symptoms. Based on that provider–patient relationship, evidence supports several recommendations for diagnosing and treating a TIA and for reducing the risk of recurrent stroke after TIA. Addressing each of these areas, in this order, is imperative to reduce the risk of recurrent stroke and improve overall cardiovascular outcomes:
- Obtain an accurate diagnosis of a TIA, using DW-MRI or comparable brain imaging, to allow for prompt intervention.
- Initiate BP management promptly in the acute setting and establish optimal BP control over the long term.
- Begin appropriate antiplatelet therapy.
- When indicated (eg, atrial fibrillation), begin anticoagulation therapy with a DOAC or warfarin.
- Begin high-intensity statin therapy.
- Consider treating patients with diabetes using an SGLT2 inhibitor or GLP-1 receptor agonist.
- Encourage smoking cessation, prescribe quit-smoking medications, or refer a smoker for behavioral support.
Education. Last, it is important to educate patients—especially those who have risk factors for a TIA or stroke—about the presentation of events, so that they know to seek immediate medical attention.
CORRESPONDENCE
Kristen Rundell, MD, Department of Family and Community Medicine, University of Arizona College of Medicine, 655 North Alvernon Way, Suite 228, Tucson, AZ 85711; [email protected]
1. Kleindorfer D, Panagos P, Pancioli A, et al. Incidence and short-term prognosis of transient ischemic attack in a population-based study. Stroke. 2005;36:720-723. doi: 10.1161/01.STR.0000158917.59233.b7
2. Kleindorfer DO, Towfighi A, Chaturvedi S, et al. 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke. 2021;52:e364-e467. doi: 10.1161/STR.0000000000000375
3. Easton JD, Saver JL, Albers GW, et al. Definition and evaluation of transient ischemic attack: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. The American Academy of Neurology affirms the value of this statement as an educational tool for neurologists. Stroke. 2009;40:2276-2293. doi: 10.1161/STROKEAHA.108.192218
4. Thacker EL, Wiggins KL, Rice KM, et al. Short-term and long-term risk of incident ischemic stroke after transient ischemic attack. Stroke. 2010;41:239-243. doi: 10.1161/STROKEAHA.109.569707
5. Hill MD, Yiannakoulias N, Jeerakathil T, et al. The high risk of stroke immediately after transient ischemic attack: a population-based study. Neurology. 2004;62:2015-2020. doi: 10.1212/01.wnl.0000129482.70315.2f
6. Giles MF, Albers GW, Amarenco P, et al. Early stroke risk and ABCD2 score performance in tissue- vs time-defined TIA: a multicenter study. Neurology. 2011;77:1222-1228. doi: 10.1212/WNL.0b013e3182309f91
7. Cucchiara BL, Kasner SE. All patients should be admitted to the hospital after a transient ischemic attack. Stroke. 2012;43:1446-1447. doi: 10.1161/STROKEAHA.111.636746
8. Amarenco P. Not all patients should be admitted to the hospital for observation after a transient ischemic attack. Stroke. 2012;43:1448-1449. doi: 10.1161/STROKEAHA.111.636753
9. Amort M, Fluri F, Schäfer J, et al. Transient ischemic attack versus transient ischemic attack mimics: frequency, clinical characteristics and outcome. Cerebrovasc Dis. 2011;32:57-64. doi: 10.1159/000327034
10. Hand PJ, Kwan J, Lindley RI, et al. Distinguishing between stroke and mimic at the bedside: The Brain Attack Study. Stroke. 2006;37:769-775. doi: 10.1161/01.STR.0000204041.13466.4c
11. Shah KH, Edlow JA. Transient ischemic attack: review for the emergency physician. Ann Emerg Med. 2004;43:592-604. doi: 10.1016/S0196064404000058
12. Crisostomo RA, Garcia MM, Tong DC. Detection of diffusion-weighted MRI abnormalities in patients with transient ischemic attack: correlation with clinical characteristics. Stroke. 2003;34:932-937. doi: 10.1161/01.STR.0000061496.00669.5E
13. Adams HP Jr, del Zoppo G, Alberts MJ, et al; ; ; ; ; . Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Stroke. 2007;38:1655-1711. doi: 10.1161/STROKEAHA.107.181486
14. Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369:283-292. doi: 10.1016/S0140-6736(07)60150-0
15. Cucchiara BL, Messe SR, Taylor RA, et al. Is the ABCD score useful for risk stratification of patients with acute transient ischemic attack? Stroke. 2006;37:1710-1714. doi: 10.1161/01.STR.0000227195.46336.93
16. Amarenco P, Lavallée PC, Labreuche J, et al; . One-year risk of stroke after transient ischemic attack or minor stroke. N Engl J Med. 2016;374:1533-1542. doi: 10.1056/NEJMoa1412981
17. Wu CM, McLaughlin K, Lorenzetti DL, et al. Early risk of stroke after transient ischemic attack: a systematic review and meta-analysis. Arch Intern Med. 2007;167:2417-2422. doi: 10.1001/archinte.167.22.2417
18. Rothwell PM, Warlow CP. Timing of TIAs preceding stroke: time window for prevention is very short. Neurology. 2005;64:817-820. doi: 10.1212/01.WNL.0000152985.32732.EE
19. Kernan WN, Ovbiagele B, Black HR, et al; American Heart Association Stroke Council, Council on Cardiovascular and Stroke Nursing, Council on Clinical Cardiology, Council on Peripheral Vascular Disease. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45:2160-2236. doi: 10.1161/STR.0000000000000024
20. Rothwell PM, Giles MF, Chandratheva A, et al. Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population-based sequential comparison. Lancet. 2007;370:1432-1442. doi: 10.1016/S0140-6736(07)61448-2
21. Hackam DG, Spence JD. Antiplatelet therapy in ischemic stroke and transient ischemic attack: an overview of major trials and meta-analyses. Stroke. 2019;50:773-778. doi: c10.1161/STROKEAHA.118.023954
22. Bhatia K, Jain V, Aggarwal D, et al. Dual antiplatelet therapy versus aspirin in patients with stroke or transient ischemic attack: meta-analysis of randomized controlled trials. Stroke. 2021;52:e217-e223. doi: 10.1161/STROKEAHA.120.033033
23. Wang Y, Pan Y, Zhao X, et al; CHANCE Investigators. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack (CHANCE) trial: one-year outcomes. Circulation. 2015;132:40-46. doi: 10.1161/CIRCULATIONAHA.114.014791
24. Furie KL, Kasner SE, Adams RJ, et al; . Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:227-276. doi: 10.1161/STR.0b013e3181f7d043
25. Powers WJ, Rabinstein AA, Ackerson T, et al; American Heart Association Stroke Council. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2018;49:e46-e110. doi: 10.1161/STR.0000000000000158
26. O’Donnell MJ, Chin SL, Rangarajan S, et al; INTERSTROKE Investigators. Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case-control study. Lancet. 2016;388:761-775. doi: 10.1016/S0140-6736(16)30506-2
27. Kristensen SL, Rørth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776-785. doi:10.1016/S2213-8587(19)30249-9
28. Bertoccini L, Baroni MG. GLP-1 receptor agonists and SGLT2 inhibitors for the treatment of type 2 diabetes: new insights and opportunities for cardiovascular protection. Adv Exp Med Biol. 2021;1307:193-212. doi:10.1007/5584_2020_494
29. Pfeffer MA, Claggett B, Diaz R, et al; ELIXA Investigators. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome N Engl J Med. 2015;373:2247-2257. doi: 10.1056/NEJMoa1509225
30. Sheahan KH, Wahlberg EA, Gilbert MP. An overview of GLP-1 agonists and recent cardiovascular outcomes trials. Postgrad Med J. 2020;96:156-161. doi:10.1136/postgradmedj-2019-137186
31. Kim AS. Medical management for secondary stroke prevention. Continuum (Minneap Minn). 2020;26:435-456. doi:10.1212/CON.0000000000000849
32. Smith L, Chakraborty D, Bhattacharya P, et al. Exposure to hypoglycemia and risk of stroke. Ann N Y Acad Sci. 2018;1431:25-34. doi:10.1111/nyas.13872
33. Amarenco P, Bogousslavsky J, Callahan A 3rd, et al; . High-dose atorvastatin after stroke or transient ischemic attack. N Engl J Med. 2006;355:549-559. doi:10.1056/NEJMoa061894
34. Castilla-Guerra, L, Fernandez-Moreno M, Leon-Jimenez D, et al. Statins in ischemic stroke prevention: what have we learned in the post-SPARCL (The Stroke Prevention by Aggressive Reduction in Cholesterol Levels) decade? Curr Treat Options Neurol. 2019;21:22. doi: 10.1007/s11940-019-0563-4
35. Bohula EA, Wiviott SD, Giugliano RP, et al. Prevention of stroke with the addition of ezetimibe to statin therapy in patients with acute coronary syndrome in IMPROVE-IT (Improved Reduction of Outcomes: Vytorin Efficacy International Trial). Circulation. 2017;136:2440-2450. doi:10.1161/CIRCULATIONAHA.117.029095
36. Moritsugu KP. The 2006 report of the Surgeon General: the health consequences of involuntary exposure to tobacco smoke. Am J Prev Med. 20067;32:542-543. doi: 10.1016/j.amepre.2007.02.026
37. Wolf PA, D’Agostino RB, Kannel WB, et al. Cigarette smoking as a risk factor for stroke: the Framingham Study. JAMA. 1988;259:1025-1029.
38. Goldstein LB, Adams R, Alberts MJ, et al. Primary prevention of ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council: cosponsored by the Atherosclerotic Peripheral Vascular Disease Interdisciplinary Working Group; Cardiovascular Nursing Council; Clinical Cardiology Council; Nutrition, Physical Activity, and Metabolism Council; and the Quality of Care and Outcomes Research Interdisciplinary Working Group: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37:1583-1633. doi: 10.1161/01.STR.0000223048.70103.F1
39. Klijn CJ, Paciaroni M, Berge E, et al. Antithrombotic treatment for secondary prevention of stroke and other thromboembolic events in patients with stroke or transient ischemic attack and non-valvular atrial fibrillation: A European Stroke Organisation guideline. Eur Stroke J. 2019;4:198-223. doi:10.1177/2396987319841187
40. Antithrombotic Trialists’ (ATT) Collaboration; Baigent C, Blackwell L, Collins R, et al. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373:1849-1860. doi:10.1016/S0140-6736(09)60503-1
41. Singer DE, Albers GW, Dalen JE, et al. Antithrombotic therapy in atrial fibrillation: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 suppl):546S–592S. doi: 10.1378/chest.08-0678
42. Rothwell PM, Algra A, Chen Z, et al. Effects of aspirin on risk and severity of early recurrent stroke after transient ischaemic attack and ischaemic stroke: time-course analysis of randomised trials. Lancet. 2016;388:365-375. doi:10.1016/S0140-6736(16)30468-8
As many as 240,000 people per year in the United States experience a transient ischemic attack (TIA),1,2 which is now defined by the American Heart Association and American Stroke Association as a “transient episode of neurological dysfunction caused by focal brain, spinal cord, or retinal ischemia, without acute infarction.”3 An older definition of TIA was based on the duration of the event (ie, resolution of symptoms at 24 hours); in the updated (2009) definition, the diagnostic criterion is the extent of focal tissue damage.3 Using the 2009 definition might mean a decrease in the number of patients who have a diagnosis of a TIA and an increase in the number who are determined to have had a stroke because an infarction is found on initial imaging.
Guided by the 2009 revised definition of a TIA, we review here the work-up and treatment of TIA, emphasizing immediacy of management to (1) prevent further tissue damage and (2) decrease the risk of a second event.

CASE
Martin L, 69 years old, retired, a nonsmoker, and with a history of peripheral arterial disease and hypercholesterolemia, presents to the emergency department (ED) of a rural hospital complaining of slurred speech and left-side facial numbness. He had an episode of facial numbness that lasted 30 minutes, then resolved, each of the 2 previous evenings; he did not seek care at those times. Now, in the ED, Mr. L is normotensive.
The patient’s medication history includes a selective serotonin reuptake inhibitor and melatonin to improve sleep. He reports having discontinued a statin because he could not tolerate its adverse effects.
What immediate steps are recommended for Mr. L’s care?
Common event callsfor quick action
A TIA is the strongest predictor of subsequent stroke and stroke-related death; the highest period of risk of these devastating outcomes is immediately following a TIA.1,2,4,5 It is essential, therefore, for the physician who sees a patient with a current complaint or recent history of suspected focal neurologic deficits to direct that patient to an ED for an accurate diagnosis and, as appropriate, early treatment for the best possible outcome.
Imaging—preferably, diffusion-weighted magnetic resonance imaging (DW-MRI), the gold standard for diagnosing stroke (see “Diagnosis includes ruling out mimics”)2,3—should be performed as soon as the patient with a suspected TIA arrives in the ED. Imaging should not be held while waiting for a stroke to declare itself—ie, by allowing symptoms to persist for longer than 24 hours. 6
Continue to: Late presentation
Late presentation. Some patients present ≥ 48 hours after onset of early symptoms of a TIA; for them, the work-up is the same as for prompt presentation but can be completed in the outpatient clinic—as long as the patient is stable clinically and imaging is accessible there. DW-MRI should be completed within 48 hours after late presentation. In such cases, the patient should be cautioned regarding risks and any recurrence of symptoms.7,8
Diagnosis includes ruling out mimics
All patients in whom a stroke is suspected should be evaluated on an emergency basis with brain imaging upon arrival at the hospital, before any therapy is initiated. As noted, DW-MRI is the preferred modality; noncontrast computed tomography (CT) or CT angiography can be used if MRI is unavailable.2,3
Mimics. Stroke has many mimics; quickly eliminating them from the differential diagnosis is important so that appropriate therapy can be initiated. Mimics usually have a prolonged presentation of symptoms, whereas the presentation of a TIA is usually abrupt. The 3 more common diagnoses that mimic a TIA are migraine with aura, seizure, and syncope.9,10 Symptoms that generally are not associated with a TIA are chest pain, generalized weakness, and confusion.11 A complete history and physical exam provide the path to the imaging, laboratory, and cardiac testing that is needed to differentiate these diagnoses from a TIA.
A thorough history is best obtained from the patient and a witness, if available, and should include identification of any focal neurologic deficits and the duration and time to resolution of symptoms. Obtain a history of risk factors for ischemia—tobacco use, diabetes, obesity, dyslipidemia, hypertension, previous TIA or stroke, atrial fibrillation, and any coagulopathy. Ask questions about a family history of TIA, stroke, and coagulopathy.11
A comprehensive physical exam, including vital signs, cardiac exam, a check for carotid bruits, and complete neurologic exam, should be performed. Most patients present with concerns for unilateral weakness and changes in speech, which are usually associated with infarction on DW-MRI.12 The most common findings on physical exam include cranial nerve abnormalities, such as diplopia, hemianopia, monocular blindness, disconjugate gaze, facial drooping, lateral tongue movement, dysphagia, and vestibular dysfunction. Cerebellar abnormalities are also often noted, including past pointing, dystaxia, ataxia, nystagmus, and motor abnormalities (eg, spasticity, clonus, or unilateral weakness in the face or extremities).11
Electrocardiography at the bedside can confirm atrial fibrillation or another arrhythmia quickly.
Essential laboratory testing includes measurement of blood glucose and serum electrolytes to determine if these particular imbalances are the cause of symptoms. The presence of a hypercoaguable state is determined by a complete blood count and coagulation studies.3,13 Urine toxicology should also be obtained to rule out other causes of symptoms. A lipid profile is beneficial for making long-term treatment decisions.
Continue to: ABCD2 score
ABCD2 score. Patients who have had a TIA and present within 72 hours after symptoms have resolved should be hospitalized if they have an ABCD2 (Age, Blood pressure [BP], Clinical presentation, Diabetes mellitus [type 1 or 2], Duration of symptoms) prediction system score > 3.14 ABCD2 criteria can be used to help identify patients who are at higher risk of stroke or need further therapy (TABLE 1).14,15

The ABCD2 score is also used to determine whether a patient needs dual antiplatelet therapy. Patients who score at the higher end of the ABCD2 system usually have an increased risk of stroke, longer hospitalization, and greater disability.
CASE
In the ED, Mr. L is immediately assessed and airlifted to a larger regional medical center, where MRI confirms a stroke.
Management
Initial management of a TIA is aimed at reducing the risk of recurrent TIA or stroke. Early medical and possibly surgical treatment are key for preventing stroke and improving outcomes. The first 48 hours after a TIA are the most critical because the incidence of recurrent TIA or stroke is highest during this period.16-18
What is the accepted strategy for early treatment?
Initial treatment must include antiplatelet therapy, BP management, anticoagulation, statin therapy, and carotid endarterectomy as indicated.2,19,20 Control of hypertension and anticoagulation decrease the risk of recurrent stroke by the largest margin20; both are “A”-level Strength of Recommendation Taxonomy interventions.2,3
Step 1: Antiplatelet therapy. After initial imaging is complete and if there are no contraindications, antiplatelet agents are recommended for patients who have had a noncardioembolic TIA. The American Heart Association and American Stroke Association recommend either aspirin, clopidogrel, dipyridamole + aspirin (available in a single capsule [Aggrenox]), or clopidogrel + aspirin as first-line therapy.2,20 The choice of agent needs to be individualized, based on tolerability and adverse effects (TABLE 22,20,21).

A meta-analysis of antiplatelet therapy reviewed the optimum dosing of each medication.21,22 Reduction of the risk of ischemic stroke with aspirin is 21% to 22% at the optimal dosing of 75 to 150 mg/d, which also reduces the risk of gastrointestinal bleeding.
Continue to: For a patient who has...
For a patient who has an ABCD2 score ≥ 4, has had a prior TIA, or has large-vessel disease, dual antiplatelet therapy is recommended for the first 21 days, with a subsequent return to monotherapy. Dual antiplatelet therapy of clopidogrel + aspirin increases the risk of adverse reactions and has not been shown to have greater long-term benefit23-25 (TABLE 22,20,21).
Step 2: BP management. This is the next immediate step. As many as 80% of patients who present with a TIA have elevated BP upon admission. BP needs to be treated and carefully monitored during this early treatment phase. The recommendation is for a systolic BP < 185 mm Hg and a diastolic BP < 110 mm Hg.24
Step 3: Anticoagulation. Treatment with warfarin or a direct oral anticoagulant (DOAC) is recommended for patients who have the potential for forming emboli—eg, in the setting of atrial fibrillation, ventricular thrombus, mechanical heart valve, or venous thromboembolism.
Step 4. High-intensity statin. A statin agent is recommended as part of immediate and long-term medical management, regardless of the low-density lipoprotein cholesterol (LDL-C) level, to reduce the risk of stroke.2,24
Carotid artery management. Surgical intervention is not always considered a component of immediate medical management. However, guidelines recommend that carotid endarterectomy or stenting be considered in patients who have stenosis > 70%.2
CASE
Mr. L is admitted to the hospital and undergoes neurosurgical intervention. Medical management is instituted.
Long-term management and secondary prevention
The main risk factors for stroke can be divided into modifiable, vascular, and unmodifiable. Addressing both modifiable and vascular risks is important for secondary prevention.
Continue to: Modifiable and vascular risk factors
Modifiable and vascular risk factors
Modifiable risk factors for stroke include hypertension, diabetes, dyslipidemia, smoking, and physical activity; the most important of these, for preventing subsequent stroke after an initial TIA, is hypertension.26
The 2 more significant vascular risk factors for stroke are carotid artery stenosis and atrial fibrillation.
Hypertension. Improving control of hypertension can improve secondary risk reduction for recurrent stroke. Control of both systolic and diastolic BP is important in this regard, with larger systolic BP reductions having a greater impact on decreasing the risk of recurrent stroke.24 Evidence supports lowering BP to improve secondary risk reduction in people with and without diagnosed hypertension: The goal is to lower systolic BP by ≥ 10 mm Hg and diastolic BP by 5 mm Hg.24 No particular class of antihypertensive is recommended in the first line, although preliminary evidence shows that a diuretic, with or without an angiotensin-converting enzyme inhibitor, might be more beneficial than other options.24
Diabetes. The risk of cardiovascular disease, including stroke, is higher in people with diabetes. Evidence shows that various (but not all) agents in 2 pharmaceutical classes—glucagon-like peptide-1 (GLP-1) receptor agonists and the sodium glucose-2 cotransporter (SGLT2) inhibitors—reduce the risk of major cardiovascular events and improve secondary prevention of recurrent stroke:
- EMPA-REG OUTCOME (ClinicalTrials.gov Identifier: NCT01131676) was the first trial to show cardiovascular benefit from an SGLT2 inhibitor (empagliflozin); subsequent studies confirmed the cardiovascular benefits found in EMPA-REG OUTCOME.27,28
- The ELIXA trial (ClinicalTrials.gov Identifier: NCT01147250) was the first to show cardiovascular benefit from a GLP-1 receptor agonist (lixisenatide); subsequent studies supported this finding.29,30
Appropriate agents in these 2 classes should be considered as first-line or adjunctive in patients with both diabetes and known cardiovascular disease, as long as there are no contraindications.27,28
Pioglitazone, a thiazolidinedione-class antidiabetic agent, was once considered a potential option to improve secondary prevention of stroke. However, the thiazolidinediones are generally no longer considered; instead, the SGLT2 inhibitors and GLP-1 receptor agonists are favored.31
Evidence demonstrates the effect of hyperglycemia on cardiovascular events; however, it is important to note that hypoglycemia can result in symptoms and focal changes that mimic a stroke. In addition, some evidence suggests that hypoglycemia can increase cardiovascular risk—thereby supporting the importance of strict control of diabetes and maintenance of euglycemia in reducing overall cardiovascular risk.32
Continue to: Lipids
Lipids. The SPARCL trial (ClinicalTrials.gov Identifier: NCT00147602) was the first study to demonstrate the benefit of high-intensity statin therapy—specifically, atorvastatin 80 mg/d—for secondary prevention for recurrent stroke.33 The recommendation is to use high-intensity statin therapy to decrease the risk of recurrent stroke by reducing the level of LDL-C—by ≥ 50% or to < 70 mg/dL, for maximum risk reduction.24,34
The IMPROVE-IT trial (ClinicalTrials.gov Identifier: NCT00202878) demonstrated the benefit of adding ezetimibe, 10 mg/d, to a moderate-to-high-intensity statin (simvastatin, 40-80 mg/d) to reduce the risk of recurrent stroke.35
Results of recent studies support the use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors for regulating levels of LDL-C, as an additional option to consider—if needed to further reduce the LDL-C level or if statins are contraindicated in a particular patient.34
Smoking cessation. Cigarette smoking is known to increase the risk of ischemic stroke; newer evidence shows that second-hand exposure to smoke also increases the risk of ischemic stroke.36,37 Although these studies focused on primary prevention of ischemic stroke, the data can reasonably be applied to secondary prevention.38 The recommendation for secondary prevention is to quit smoking and avoid secondhand smoke.24
Alcohol. Evidence demonstrates that heavy alcohol consumption and alcoholism increase the risk of stroke; similar to what is known about smoking, most available data relate to primary prevention.38 The recommendation for providing secondary stroke prevention is to stop or decrease alcohol intake.24
Weight reduction. Obesity (body mass index > 30) increases the risk of ischemic stroke. However, there is, as yet, no evidence that weight loss diminishes the risk of subsequent stroke for secondary prevention.24
Physical activity. Aerobic exercise and strength-training programs after a stroke improve cardiovascular health and mobility. There is no evidence that exercise leads to a reduction in the risk of subsequent stroke.24
Continue to: Nutrition
Nutrition. No current randomized controlled trials are focused on the relationship between diet and recurrent stroke for purposes of prevention; however, evidence for both BP and lipid control incorporate dietary guidance. Recommendations include reducing intake of saturated fats and of sodium (the latter, to < 2.3 g/d) and increasing intake of fruits and vegetables, both of which are beneficial for controlling BP and lipid levels and promoting overall cardiovascular health.38
Carotid artery stenosis. Several randomized controlled trials have demonstrated benefit from treating carotid stenosis (> 70% stenosis but not < 50%) with carotid endarterectomy to reduce the risk of recurrent stroke after TIA.2 The ideal timing of carotid endarterectomy is still being studied; however, available evidence supports intervention within 2 to 6 weeks after TIA or stroke.25 Studies are ongoing that compare carotid angioplasty and stenting against carotid endarterectomy. Medical therapy, with antiplatelet agents and statins, is recommended after carotid endarterectomy.25
Atrial fibrillation increases the risk of recurrent stroke after a TIA, and is the most important indication for secondary stroke prevention with anticoagulation therapy:
- Warfarin. Several studies have shown that warfarin provides a 68% relative risk reduction and a 1.4% absolute risk reduction in the annual stroke rate.24 To achieve this reduction in risk, the optimal international normalized ratio is 2.5 (range, 2-3).24
- Aspirin provides a 13% relative risk reduction for recurrent stroke, although there is evidence that long-term anticoagulation provides more benefit than aspirin after a TIA.39-41 Optimal dosing of aspirin ranges from 75-100 mg/d; greatest benefit is likely in the 12 weeks after stroke, when the risk of recurrent stroke is highest.31,41,42
- DOACs have similar efficacy to warfarin but more rapid onset, lower risk of bleeding, fewer drug interactions, and no requirement for monitoring—often making them a more tolerable long-term choice. Options are rivaroxaban 20 mg/d, dabigatran 150 mg twice daily, apixaban 5 mg twice daily, and edoxaban 60 mg/d.39
When to start anticoagulation and the choice of agent should be weighed against a risk of bleeding, which is highest after the initial stroke. Cost is also a consideration: DOACs are more expensive than warfarin.
CASE
Mr. L is discharged 3 days after carotid endarterectomy and free of residual deficits. He is started on dual antiplatelet therapy (aspirin + clopidogrel) for 21 days, to be followed by a return to monotherapy. He is restarted on a high-intensity statin. He is instructed to resume taking the selective serotonin reuptake inhibitor and melatonin for sleep, as needed. Last, he is told to schedule follow-up with his primary care physician in 7 to 10 days to begin post-stroke care.
Final thoughts
Primary care physicians are often the first point of contact for patients with current or remote TIA symptoms. Based on that provider–patient relationship, evidence supports several recommendations for diagnosing and treating a TIA and for reducing the risk of recurrent stroke after TIA. Addressing each of these areas, in this order, is imperative to reduce the risk of recurrent stroke and improve overall cardiovascular outcomes:
- Obtain an accurate diagnosis of a TIA, using DW-MRI or comparable brain imaging, to allow for prompt intervention.
- Initiate BP management promptly in the acute setting and establish optimal BP control over the long term.
- Begin appropriate antiplatelet therapy.
- When indicated (eg, atrial fibrillation), begin anticoagulation therapy with a DOAC or warfarin.
- Begin high-intensity statin therapy.
- Consider treating patients with diabetes using an SGLT2 inhibitor or GLP-1 receptor agonist.
- Encourage smoking cessation, prescribe quit-smoking medications, or refer a smoker for behavioral support.
Education. Last, it is important to educate patients—especially those who have risk factors for a TIA or stroke—about the presentation of events, so that they know to seek immediate medical attention.
CORRESPONDENCE
Kristen Rundell, MD, Department of Family and Community Medicine, University of Arizona College of Medicine, 655 North Alvernon Way, Suite 228, Tucson, AZ 85711; [email protected]
As many as 240,000 people per year in the United States experience a transient ischemic attack (TIA),1,2 which is now defined by the American Heart Association and American Stroke Association as a “transient episode of neurological dysfunction caused by focal brain, spinal cord, or retinal ischemia, without acute infarction.”3 An older definition of TIA was based on the duration of the event (ie, resolution of symptoms at 24 hours); in the updated (2009) definition, the diagnostic criterion is the extent of focal tissue damage.3 Using the 2009 definition might mean a decrease in the number of patients who have a diagnosis of a TIA and an increase in the number who are determined to have had a stroke because an infarction is found on initial imaging.
Guided by the 2009 revised definition of a TIA, we review here the work-up and treatment of TIA, emphasizing immediacy of management to (1) prevent further tissue damage and (2) decrease the risk of a second event.

CASE
Martin L, 69 years old, retired, a nonsmoker, and with a history of peripheral arterial disease and hypercholesterolemia, presents to the emergency department (ED) of a rural hospital complaining of slurred speech and left-side facial numbness. He had an episode of facial numbness that lasted 30 minutes, then resolved, each of the 2 previous evenings; he did not seek care at those times. Now, in the ED, Mr. L is normotensive.
The patient’s medication history includes a selective serotonin reuptake inhibitor and melatonin to improve sleep. He reports having discontinued a statin because he could not tolerate its adverse effects.
What immediate steps are recommended for Mr. L’s care?
Common event callsfor quick action
A TIA is the strongest predictor of subsequent stroke and stroke-related death; the highest period of risk of these devastating outcomes is immediately following a TIA.1,2,4,5 It is essential, therefore, for the physician who sees a patient with a current complaint or recent history of suspected focal neurologic deficits to direct that patient to an ED for an accurate diagnosis and, as appropriate, early treatment for the best possible outcome.
Imaging—preferably, diffusion-weighted magnetic resonance imaging (DW-MRI), the gold standard for diagnosing stroke (see “Diagnosis includes ruling out mimics”)2,3—should be performed as soon as the patient with a suspected TIA arrives in the ED. Imaging should not be held while waiting for a stroke to declare itself—ie, by allowing symptoms to persist for longer than 24 hours. 6
Continue to: Late presentation
Late presentation. Some patients present ≥ 48 hours after onset of early symptoms of a TIA; for them, the work-up is the same as for prompt presentation but can be completed in the outpatient clinic—as long as the patient is stable clinically and imaging is accessible there. DW-MRI should be completed within 48 hours after late presentation. In such cases, the patient should be cautioned regarding risks and any recurrence of symptoms.7,8
Diagnosis includes ruling out mimics
All patients in whom a stroke is suspected should be evaluated on an emergency basis with brain imaging upon arrival at the hospital, before any therapy is initiated. As noted, DW-MRI is the preferred modality; noncontrast computed tomography (CT) or CT angiography can be used if MRI is unavailable.2,3
Mimics. Stroke has many mimics; quickly eliminating them from the differential diagnosis is important so that appropriate therapy can be initiated. Mimics usually have a prolonged presentation of symptoms, whereas the presentation of a TIA is usually abrupt. The 3 more common diagnoses that mimic a TIA are migraine with aura, seizure, and syncope.9,10 Symptoms that generally are not associated with a TIA are chest pain, generalized weakness, and confusion.11 A complete history and physical exam provide the path to the imaging, laboratory, and cardiac testing that is needed to differentiate these diagnoses from a TIA.
A thorough history is best obtained from the patient and a witness, if available, and should include identification of any focal neurologic deficits and the duration and time to resolution of symptoms. Obtain a history of risk factors for ischemia—tobacco use, diabetes, obesity, dyslipidemia, hypertension, previous TIA or stroke, atrial fibrillation, and any coagulopathy. Ask questions about a family history of TIA, stroke, and coagulopathy.11
A comprehensive physical exam, including vital signs, cardiac exam, a check for carotid bruits, and complete neurologic exam, should be performed. Most patients present with concerns for unilateral weakness and changes in speech, which are usually associated with infarction on DW-MRI.12 The most common findings on physical exam include cranial nerve abnormalities, such as diplopia, hemianopia, monocular blindness, disconjugate gaze, facial drooping, lateral tongue movement, dysphagia, and vestibular dysfunction. Cerebellar abnormalities are also often noted, including past pointing, dystaxia, ataxia, nystagmus, and motor abnormalities (eg, spasticity, clonus, or unilateral weakness in the face or extremities).11
Electrocardiography at the bedside can confirm atrial fibrillation or another arrhythmia quickly.
Essential laboratory testing includes measurement of blood glucose and serum electrolytes to determine if these particular imbalances are the cause of symptoms. The presence of a hypercoaguable state is determined by a complete blood count and coagulation studies.3,13 Urine toxicology should also be obtained to rule out other causes of symptoms. A lipid profile is beneficial for making long-term treatment decisions.
Continue to: ABCD2 score
ABCD2 score. Patients who have had a TIA and present within 72 hours after symptoms have resolved should be hospitalized if they have an ABCD2 (Age, Blood pressure [BP], Clinical presentation, Diabetes mellitus [type 1 or 2], Duration of symptoms) prediction system score > 3.14 ABCD2 criteria can be used to help identify patients who are at higher risk of stroke or need further therapy (TABLE 1).14,15

The ABCD2 score is also used to determine whether a patient needs dual antiplatelet therapy. Patients who score at the higher end of the ABCD2 system usually have an increased risk of stroke, longer hospitalization, and greater disability.
CASE
In the ED, Mr. L is immediately assessed and airlifted to a larger regional medical center, where MRI confirms a stroke.
Management
Initial management of a TIA is aimed at reducing the risk of recurrent TIA or stroke. Early medical and possibly surgical treatment are key for preventing stroke and improving outcomes. The first 48 hours after a TIA are the most critical because the incidence of recurrent TIA or stroke is highest during this period.16-18
What is the accepted strategy for early treatment?
Initial treatment must include antiplatelet therapy, BP management, anticoagulation, statin therapy, and carotid endarterectomy as indicated.2,19,20 Control of hypertension and anticoagulation decrease the risk of recurrent stroke by the largest margin20; both are “A”-level Strength of Recommendation Taxonomy interventions.2,3
Step 1: Antiplatelet therapy. After initial imaging is complete and if there are no contraindications, antiplatelet agents are recommended for patients who have had a noncardioembolic TIA. The American Heart Association and American Stroke Association recommend either aspirin, clopidogrel, dipyridamole + aspirin (available in a single capsule [Aggrenox]), or clopidogrel + aspirin as first-line therapy.2,20 The choice of agent needs to be individualized, based on tolerability and adverse effects (TABLE 22,20,21).

A meta-analysis of antiplatelet therapy reviewed the optimum dosing of each medication.21,22 Reduction of the risk of ischemic stroke with aspirin is 21% to 22% at the optimal dosing of 75 to 150 mg/d, which also reduces the risk of gastrointestinal bleeding.
Continue to: For a patient who has...
For a patient who has an ABCD2 score ≥ 4, has had a prior TIA, or has large-vessel disease, dual antiplatelet therapy is recommended for the first 21 days, with a subsequent return to monotherapy. Dual antiplatelet therapy of clopidogrel + aspirin increases the risk of adverse reactions and has not been shown to have greater long-term benefit23-25 (TABLE 22,20,21).
Step 2: BP management. This is the next immediate step. As many as 80% of patients who present with a TIA have elevated BP upon admission. BP needs to be treated and carefully monitored during this early treatment phase. The recommendation is for a systolic BP < 185 mm Hg and a diastolic BP < 110 mm Hg.24
Step 3: Anticoagulation. Treatment with warfarin or a direct oral anticoagulant (DOAC) is recommended for patients who have the potential for forming emboli—eg, in the setting of atrial fibrillation, ventricular thrombus, mechanical heart valve, or venous thromboembolism.
Step 4. High-intensity statin. A statin agent is recommended as part of immediate and long-term medical management, regardless of the low-density lipoprotein cholesterol (LDL-C) level, to reduce the risk of stroke.2,24
Carotid artery management. Surgical intervention is not always considered a component of immediate medical management. However, guidelines recommend that carotid endarterectomy or stenting be considered in patients who have stenosis > 70%.2
CASE
Mr. L is admitted to the hospital and undergoes neurosurgical intervention. Medical management is instituted.
Long-term management and secondary prevention
The main risk factors for stroke can be divided into modifiable, vascular, and unmodifiable. Addressing both modifiable and vascular risks is important for secondary prevention.
Continue to: Modifiable and vascular risk factors
Modifiable and vascular risk factors
Modifiable risk factors for stroke include hypertension, diabetes, dyslipidemia, smoking, and physical activity; the most important of these, for preventing subsequent stroke after an initial TIA, is hypertension.26
The 2 more significant vascular risk factors for stroke are carotid artery stenosis and atrial fibrillation.
Hypertension. Improving control of hypertension can improve secondary risk reduction for recurrent stroke. Control of both systolic and diastolic BP is important in this regard, with larger systolic BP reductions having a greater impact on decreasing the risk of recurrent stroke.24 Evidence supports lowering BP to improve secondary risk reduction in people with and without diagnosed hypertension: The goal is to lower systolic BP by ≥ 10 mm Hg and diastolic BP by 5 mm Hg.24 No particular class of antihypertensive is recommended in the first line, although preliminary evidence shows that a diuretic, with or without an angiotensin-converting enzyme inhibitor, might be more beneficial than other options.24
Diabetes. The risk of cardiovascular disease, including stroke, is higher in people with diabetes. Evidence shows that various (but not all) agents in 2 pharmaceutical classes—glucagon-like peptide-1 (GLP-1) receptor agonists and the sodium glucose-2 cotransporter (SGLT2) inhibitors—reduce the risk of major cardiovascular events and improve secondary prevention of recurrent stroke:
- EMPA-REG OUTCOME (ClinicalTrials.gov Identifier: NCT01131676) was the first trial to show cardiovascular benefit from an SGLT2 inhibitor (empagliflozin); subsequent studies confirmed the cardiovascular benefits found in EMPA-REG OUTCOME.27,28
- The ELIXA trial (ClinicalTrials.gov Identifier: NCT01147250) was the first to show cardiovascular benefit from a GLP-1 receptor agonist (lixisenatide); subsequent studies supported this finding.29,30
Appropriate agents in these 2 classes should be considered as first-line or adjunctive in patients with both diabetes and known cardiovascular disease, as long as there are no contraindications.27,28
Pioglitazone, a thiazolidinedione-class antidiabetic agent, was once considered a potential option to improve secondary prevention of stroke. However, the thiazolidinediones are generally no longer considered; instead, the SGLT2 inhibitors and GLP-1 receptor agonists are favored.31
Evidence demonstrates the effect of hyperglycemia on cardiovascular events; however, it is important to note that hypoglycemia can result in symptoms and focal changes that mimic a stroke. In addition, some evidence suggests that hypoglycemia can increase cardiovascular risk—thereby supporting the importance of strict control of diabetes and maintenance of euglycemia in reducing overall cardiovascular risk.32
Continue to: Lipids
Lipids. The SPARCL trial (ClinicalTrials.gov Identifier: NCT00147602) was the first study to demonstrate the benefit of high-intensity statin therapy—specifically, atorvastatin 80 mg/d—for secondary prevention for recurrent stroke.33 The recommendation is to use high-intensity statin therapy to decrease the risk of recurrent stroke by reducing the level of LDL-C—by ≥ 50% or to < 70 mg/dL, for maximum risk reduction.24,34
The IMPROVE-IT trial (ClinicalTrials.gov Identifier: NCT00202878) demonstrated the benefit of adding ezetimibe, 10 mg/d, to a moderate-to-high-intensity statin (simvastatin, 40-80 mg/d) to reduce the risk of recurrent stroke.35
Results of recent studies support the use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors for regulating levels of LDL-C, as an additional option to consider—if needed to further reduce the LDL-C level or if statins are contraindicated in a particular patient.34
Smoking cessation. Cigarette smoking is known to increase the risk of ischemic stroke; newer evidence shows that second-hand exposure to smoke also increases the risk of ischemic stroke.36,37 Although these studies focused on primary prevention of ischemic stroke, the data can reasonably be applied to secondary prevention.38 The recommendation for secondary prevention is to quit smoking and avoid secondhand smoke.24
Alcohol. Evidence demonstrates that heavy alcohol consumption and alcoholism increase the risk of stroke; similar to what is known about smoking, most available data relate to primary prevention.38 The recommendation for providing secondary stroke prevention is to stop or decrease alcohol intake.24
Weight reduction. Obesity (body mass index > 30) increases the risk of ischemic stroke. However, there is, as yet, no evidence that weight loss diminishes the risk of subsequent stroke for secondary prevention.24
Physical activity. Aerobic exercise and strength-training programs after a stroke improve cardiovascular health and mobility. There is no evidence that exercise leads to a reduction in the risk of subsequent stroke.24
Continue to: Nutrition
Nutrition. No current randomized controlled trials are focused on the relationship between diet and recurrent stroke for purposes of prevention; however, evidence for both BP and lipid control incorporate dietary guidance. Recommendations include reducing intake of saturated fats and of sodium (the latter, to < 2.3 g/d) and increasing intake of fruits and vegetables, both of which are beneficial for controlling BP and lipid levels and promoting overall cardiovascular health.38
Carotid artery stenosis. Several randomized controlled trials have demonstrated benefit from treating carotid stenosis (> 70% stenosis but not < 50%) with carotid endarterectomy to reduce the risk of recurrent stroke after TIA.2 The ideal timing of carotid endarterectomy is still being studied; however, available evidence supports intervention within 2 to 6 weeks after TIA or stroke.25 Studies are ongoing that compare carotid angioplasty and stenting against carotid endarterectomy. Medical therapy, with antiplatelet agents and statins, is recommended after carotid endarterectomy.25
Atrial fibrillation increases the risk of recurrent stroke after a TIA, and is the most important indication for secondary stroke prevention with anticoagulation therapy:
- Warfarin. Several studies have shown that warfarin provides a 68% relative risk reduction and a 1.4% absolute risk reduction in the annual stroke rate.24 To achieve this reduction in risk, the optimal international normalized ratio is 2.5 (range, 2-3).24
- Aspirin provides a 13% relative risk reduction for recurrent stroke, although there is evidence that long-term anticoagulation provides more benefit than aspirin after a TIA.39-41 Optimal dosing of aspirin ranges from 75-100 mg/d; greatest benefit is likely in the 12 weeks after stroke, when the risk of recurrent stroke is highest.31,41,42
- DOACs have similar efficacy to warfarin but more rapid onset, lower risk of bleeding, fewer drug interactions, and no requirement for monitoring—often making them a more tolerable long-term choice. Options are rivaroxaban 20 mg/d, dabigatran 150 mg twice daily, apixaban 5 mg twice daily, and edoxaban 60 mg/d.39
When to start anticoagulation and the choice of agent should be weighed against a risk of bleeding, which is highest after the initial stroke. Cost is also a consideration: DOACs are more expensive than warfarin.
CASE
Mr. L is discharged 3 days after carotid endarterectomy and free of residual deficits. He is started on dual antiplatelet therapy (aspirin + clopidogrel) for 21 days, to be followed by a return to monotherapy. He is restarted on a high-intensity statin. He is instructed to resume taking the selective serotonin reuptake inhibitor and melatonin for sleep, as needed. Last, he is told to schedule follow-up with his primary care physician in 7 to 10 days to begin post-stroke care.
Final thoughts
Primary care physicians are often the first point of contact for patients with current or remote TIA symptoms. Based on that provider–patient relationship, evidence supports several recommendations for diagnosing and treating a TIA and for reducing the risk of recurrent stroke after TIA. Addressing each of these areas, in this order, is imperative to reduce the risk of recurrent stroke and improve overall cardiovascular outcomes:
- Obtain an accurate diagnosis of a TIA, using DW-MRI or comparable brain imaging, to allow for prompt intervention.
- Initiate BP management promptly in the acute setting and establish optimal BP control over the long term.
- Begin appropriate antiplatelet therapy.
- When indicated (eg, atrial fibrillation), begin anticoagulation therapy with a DOAC or warfarin.
- Begin high-intensity statin therapy.
- Consider treating patients with diabetes using an SGLT2 inhibitor or GLP-1 receptor agonist.
- Encourage smoking cessation, prescribe quit-smoking medications, or refer a smoker for behavioral support.
Education. Last, it is important to educate patients—especially those who have risk factors for a TIA or stroke—about the presentation of events, so that they know to seek immediate medical attention.
CORRESPONDENCE
Kristen Rundell, MD, Department of Family and Community Medicine, University of Arizona College of Medicine, 655 North Alvernon Way, Suite 228, Tucson, AZ 85711; [email protected]
1. Kleindorfer D, Panagos P, Pancioli A, et al. Incidence and short-term prognosis of transient ischemic attack in a population-based study. Stroke. 2005;36:720-723. doi: 10.1161/01.STR.0000158917.59233.b7
2. Kleindorfer DO, Towfighi A, Chaturvedi S, et al. 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke. 2021;52:e364-e467. doi: 10.1161/STR.0000000000000375
3. Easton JD, Saver JL, Albers GW, et al. Definition and evaluation of transient ischemic attack: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. The American Academy of Neurology affirms the value of this statement as an educational tool for neurologists. Stroke. 2009;40:2276-2293. doi: 10.1161/STROKEAHA.108.192218
4. Thacker EL, Wiggins KL, Rice KM, et al. Short-term and long-term risk of incident ischemic stroke after transient ischemic attack. Stroke. 2010;41:239-243. doi: 10.1161/STROKEAHA.109.569707
5. Hill MD, Yiannakoulias N, Jeerakathil T, et al. The high risk of stroke immediately after transient ischemic attack: a population-based study. Neurology. 2004;62:2015-2020. doi: 10.1212/01.wnl.0000129482.70315.2f
6. Giles MF, Albers GW, Amarenco P, et al. Early stroke risk and ABCD2 score performance in tissue- vs time-defined TIA: a multicenter study. Neurology. 2011;77:1222-1228. doi: 10.1212/WNL.0b013e3182309f91
7. Cucchiara BL, Kasner SE. All patients should be admitted to the hospital after a transient ischemic attack. Stroke. 2012;43:1446-1447. doi: 10.1161/STROKEAHA.111.636746
8. Amarenco P. Not all patients should be admitted to the hospital for observation after a transient ischemic attack. Stroke. 2012;43:1448-1449. doi: 10.1161/STROKEAHA.111.636753
9. Amort M, Fluri F, Schäfer J, et al. Transient ischemic attack versus transient ischemic attack mimics: frequency, clinical characteristics and outcome. Cerebrovasc Dis. 2011;32:57-64. doi: 10.1159/000327034
10. Hand PJ, Kwan J, Lindley RI, et al. Distinguishing between stroke and mimic at the bedside: The Brain Attack Study. Stroke. 2006;37:769-775. doi: 10.1161/01.STR.0000204041.13466.4c
11. Shah KH, Edlow JA. Transient ischemic attack: review for the emergency physician. Ann Emerg Med. 2004;43:592-604. doi: 10.1016/S0196064404000058
12. Crisostomo RA, Garcia MM, Tong DC. Detection of diffusion-weighted MRI abnormalities in patients with transient ischemic attack: correlation with clinical characteristics. Stroke. 2003;34:932-937. doi: 10.1161/01.STR.0000061496.00669.5E
13. Adams HP Jr, del Zoppo G, Alberts MJ, et al; ; ; ; ; . Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Stroke. 2007;38:1655-1711. doi: 10.1161/STROKEAHA.107.181486
14. Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369:283-292. doi: 10.1016/S0140-6736(07)60150-0
15. Cucchiara BL, Messe SR, Taylor RA, et al. Is the ABCD score useful for risk stratification of patients with acute transient ischemic attack? Stroke. 2006;37:1710-1714. doi: 10.1161/01.STR.0000227195.46336.93
16. Amarenco P, Lavallée PC, Labreuche J, et al; . One-year risk of stroke after transient ischemic attack or minor stroke. N Engl J Med. 2016;374:1533-1542. doi: 10.1056/NEJMoa1412981
17. Wu CM, McLaughlin K, Lorenzetti DL, et al. Early risk of stroke after transient ischemic attack: a systematic review and meta-analysis. Arch Intern Med. 2007;167:2417-2422. doi: 10.1001/archinte.167.22.2417
18. Rothwell PM, Warlow CP. Timing of TIAs preceding stroke: time window for prevention is very short. Neurology. 2005;64:817-820. doi: 10.1212/01.WNL.0000152985.32732.EE
19. Kernan WN, Ovbiagele B, Black HR, et al; American Heart Association Stroke Council, Council on Cardiovascular and Stroke Nursing, Council on Clinical Cardiology, Council on Peripheral Vascular Disease. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45:2160-2236. doi: 10.1161/STR.0000000000000024
20. Rothwell PM, Giles MF, Chandratheva A, et al. Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population-based sequential comparison. Lancet. 2007;370:1432-1442. doi: 10.1016/S0140-6736(07)61448-2
21. Hackam DG, Spence JD. Antiplatelet therapy in ischemic stroke and transient ischemic attack: an overview of major trials and meta-analyses. Stroke. 2019;50:773-778. doi: c10.1161/STROKEAHA.118.023954
22. Bhatia K, Jain V, Aggarwal D, et al. Dual antiplatelet therapy versus aspirin in patients with stroke or transient ischemic attack: meta-analysis of randomized controlled trials. Stroke. 2021;52:e217-e223. doi: 10.1161/STROKEAHA.120.033033
23. Wang Y, Pan Y, Zhao X, et al; CHANCE Investigators. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack (CHANCE) trial: one-year outcomes. Circulation. 2015;132:40-46. doi: 10.1161/CIRCULATIONAHA.114.014791
24. Furie KL, Kasner SE, Adams RJ, et al; . Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:227-276. doi: 10.1161/STR.0b013e3181f7d043
25. Powers WJ, Rabinstein AA, Ackerson T, et al; American Heart Association Stroke Council. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2018;49:e46-e110. doi: 10.1161/STR.0000000000000158
26. O’Donnell MJ, Chin SL, Rangarajan S, et al; INTERSTROKE Investigators. Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case-control study. Lancet. 2016;388:761-775. doi: 10.1016/S0140-6736(16)30506-2
27. Kristensen SL, Rørth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776-785. doi:10.1016/S2213-8587(19)30249-9
28. Bertoccini L, Baroni MG. GLP-1 receptor agonists and SGLT2 inhibitors for the treatment of type 2 diabetes: new insights and opportunities for cardiovascular protection. Adv Exp Med Biol. 2021;1307:193-212. doi:10.1007/5584_2020_494
29. Pfeffer MA, Claggett B, Diaz R, et al; ELIXA Investigators. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome N Engl J Med. 2015;373:2247-2257. doi: 10.1056/NEJMoa1509225
30. Sheahan KH, Wahlberg EA, Gilbert MP. An overview of GLP-1 agonists and recent cardiovascular outcomes trials. Postgrad Med J. 2020;96:156-161. doi:10.1136/postgradmedj-2019-137186
31. Kim AS. Medical management for secondary stroke prevention. Continuum (Minneap Minn). 2020;26:435-456. doi:10.1212/CON.0000000000000849
32. Smith L, Chakraborty D, Bhattacharya P, et al. Exposure to hypoglycemia and risk of stroke. Ann N Y Acad Sci. 2018;1431:25-34. doi:10.1111/nyas.13872
33. Amarenco P, Bogousslavsky J, Callahan A 3rd, et al; . High-dose atorvastatin after stroke or transient ischemic attack. N Engl J Med. 2006;355:549-559. doi:10.1056/NEJMoa061894
34. Castilla-Guerra, L, Fernandez-Moreno M, Leon-Jimenez D, et al. Statins in ischemic stroke prevention: what have we learned in the post-SPARCL (The Stroke Prevention by Aggressive Reduction in Cholesterol Levels) decade? Curr Treat Options Neurol. 2019;21:22. doi: 10.1007/s11940-019-0563-4
35. Bohula EA, Wiviott SD, Giugliano RP, et al. Prevention of stroke with the addition of ezetimibe to statin therapy in patients with acute coronary syndrome in IMPROVE-IT (Improved Reduction of Outcomes: Vytorin Efficacy International Trial). Circulation. 2017;136:2440-2450. doi:10.1161/CIRCULATIONAHA.117.029095
36. Moritsugu KP. The 2006 report of the Surgeon General: the health consequences of involuntary exposure to tobacco smoke. Am J Prev Med. 20067;32:542-543. doi: 10.1016/j.amepre.2007.02.026
37. Wolf PA, D’Agostino RB, Kannel WB, et al. Cigarette smoking as a risk factor for stroke: the Framingham Study. JAMA. 1988;259:1025-1029.
38. Goldstein LB, Adams R, Alberts MJ, et al. Primary prevention of ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council: cosponsored by the Atherosclerotic Peripheral Vascular Disease Interdisciplinary Working Group; Cardiovascular Nursing Council; Clinical Cardiology Council; Nutrition, Physical Activity, and Metabolism Council; and the Quality of Care and Outcomes Research Interdisciplinary Working Group: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37:1583-1633. doi: 10.1161/01.STR.0000223048.70103.F1
39. Klijn CJ, Paciaroni M, Berge E, et al. Antithrombotic treatment for secondary prevention of stroke and other thromboembolic events in patients with stroke or transient ischemic attack and non-valvular atrial fibrillation: A European Stroke Organisation guideline. Eur Stroke J. 2019;4:198-223. doi:10.1177/2396987319841187
40. Antithrombotic Trialists’ (ATT) Collaboration; Baigent C, Blackwell L, Collins R, et al. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373:1849-1860. doi:10.1016/S0140-6736(09)60503-1
41. Singer DE, Albers GW, Dalen JE, et al. Antithrombotic therapy in atrial fibrillation: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 suppl):546S–592S. doi: 10.1378/chest.08-0678
42. Rothwell PM, Algra A, Chen Z, et al. Effects of aspirin on risk and severity of early recurrent stroke after transient ischaemic attack and ischaemic stroke: time-course analysis of randomised trials. Lancet. 2016;388:365-375. doi:10.1016/S0140-6736(16)30468-8
1. Kleindorfer D, Panagos P, Pancioli A, et al. Incidence and short-term prognosis of transient ischemic attack in a population-based study. Stroke. 2005;36:720-723. doi: 10.1161/01.STR.0000158917.59233.b7
2. Kleindorfer DO, Towfighi A, Chaturvedi S, et al. 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke. 2021;52:e364-e467. doi: 10.1161/STR.0000000000000375
3. Easton JD, Saver JL, Albers GW, et al. Definition and evaluation of transient ischemic attack: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. The American Academy of Neurology affirms the value of this statement as an educational tool for neurologists. Stroke. 2009;40:2276-2293. doi: 10.1161/STROKEAHA.108.192218
4. Thacker EL, Wiggins KL, Rice KM, et al. Short-term and long-term risk of incident ischemic stroke after transient ischemic attack. Stroke. 2010;41:239-243. doi: 10.1161/STROKEAHA.109.569707
5. Hill MD, Yiannakoulias N, Jeerakathil T, et al. The high risk of stroke immediately after transient ischemic attack: a population-based study. Neurology. 2004;62:2015-2020. doi: 10.1212/01.wnl.0000129482.70315.2f
6. Giles MF, Albers GW, Amarenco P, et al. Early stroke risk and ABCD2 score performance in tissue- vs time-defined TIA: a multicenter study. Neurology. 2011;77:1222-1228. doi: 10.1212/WNL.0b013e3182309f91
7. Cucchiara BL, Kasner SE. All patients should be admitted to the hospital after a transient ischemic attack. Stroke. 2012;43:1446-1447. doi: 10.1161/STROKEAHA.111.636746
8. Amarenco P. Not all patients should be admitted to the hospital for observation after a transient ischemic attack. Stroke. 2012;43:1448-1449. doi: 10.1161/STROKEAHA.111.636753
9. Amort M, Fluri F, Schäfer J, et al. Transient ischemic attack versus transient ischemic attack mimics: frequency, clinical characteristics and outcome. Cerebrovasc Dis. 2011;32:57-64. doi: 10.1159/000327034
10. Hand PJ, Kwan J, Lindley RI, et al. Distinguishing between stroke and mimic at the bedside: The Brain Attack Study. Stroke. 2006;37:769-775. doi: 10.1161/01.STR.0000204041.13466.4c
11. Shah KH, Edlow JA. Transient ischemic attack: review for the emergency physician. Ann Emerg Med. 2004;43:592-604. doi: 10.1016/S0196064404000058
12. Crisostomo RA, Garcia MM, Tong DC. Detection of diffusion-weighted MRI abnormalities in patients with transient ischemic attack: correlation with clinical characteristics. Stroke. 2003;34:932-937. doi: 10.1161/01.STR.0000061496.00669.5E
13. Adams HP Jr, del Zoppo G, Alberts MJ, et al; ; ; ; ; . Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: the American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Stroke. 2007;38:1655-1711. doi: 10.1161/STROKEAHA.107.181486
14. Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369:283-292. doi: 10.1016/S0140-6736(07)60150-0
15. Cucchiara BL, Messe SR, Taylor RA, et al. Is the ABCD score useful for risk stratification of patients with acute transient ischemic attack? Stroke. 2006;37:1710-1714. doi: 10.1161/01.STR.0000227195.46336.93
16. Amarenco P, Lavallée PC, Labreuche J, et al; . One-year risk of stroke after transient ischemic attack or minor stroke. N Engl J Med. 2016;374:1533-1542. doi: 10.1056/NEJMoa1412981
17. Wu CM, McLaughlin K, Lorenzetti DL, et al. Early risk of stroke after transient ischemic attack: a systematic review and meta-analysis. Arch Intern Med. 2007;167:2417-2422. doi: 10.1001/archinte.167.22.2417
18. Rothwell PM, Warlow CP. Timing of TIAs preceding stroke: time window for prevention is very short. Neurology. 2005;64:817-820. doi: 10.1212/01.WNL.0000152985.32732.EE
19. Kernan WN, Ovbiagele B, Black HR, et al; American Heart Association Stroke Council, Council on Cardiovascular and Stroke Nursing, Council on Clinical Cardiology, Council on Peripheral Vascular Disease. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45:2160-2236. doi: 10.1161/STR.0000000000000024
20. Rothwell PM, Giles MF, Chandratheva A, et al. Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population-based sequential comparison. Lancet. 2007;370:1432-1442. doi: 10.1016/S0140-6736(07)61448-2
21. Hackam DG, Spence JD. Antiplatelet therapy in ischemic stroke and transient ischemic attack: an overview of major trials and meta-analyses. Stroke. 2019;50:773-778. doi: c10.1161/STROKEAHA.118.023954
22. Bhatia K, Jain V, Aggarwal D, et al. Dual antiplatelet therapy versus aspirin in patients with stroke or transient ischemic attack: meta-analysis of randomized controlled trials. Stroke. 2021;52:e217-e223. doi: 10.1161/STROKEAHA.120.033033
23. Wang Y, Pan Y, Zhao X, et al; CHANCE Investigators. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack (CHANCE) trial: one-year outcomes. Circulation. 2015;132:40-46. doi: 10.1161/CIRCULATIONAHA.114.014791
24. Furie KL, Kasner SE, Adams RJ, et al; . Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:227-276. doi: 10.1161/STR.0b013e3181f7d043
25. Powers WJ, Rabinstein AA, Ackerson T, et al; American Heart Association Stroke Council. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2018;49:e46-e110. doi: 10.1161/STR.0000000000000158
26. O’Donnell MJ, Chin SL, Rangarajan S, et al; INTERSTROKE Investigators. Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case-control study. Lancet. 2016;388:761-775. doi: 10.1016/S0140-6736(16)30506-2
27. Kristensen SL, Rørth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776-785. doi:10.1016/S2213-8587(19)30249-9
28. Bertoccini L, Baroni MG. GLP-1 receptor agonists and SGLT2 inhibitors for the treatment of type 2 diabetes: new insights and opportunities for cardiovascular protection. Adv Exp Med Biol. 2021;1307:193-212. doi:10.1007/5584_2020_494
29. Pfeffer MA, Claggett B, Diaz R, et al; ELIXA Investigators. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome N Engl J Med. 2015;373:2247-2257. doi: 10.1056/NEJMoa1509225
30. Sheahan KH, Wahlberg EA, Gilbert MP. An overview of GLP-1 agonists and recent cardiovascular outcomes trials. Postgrad Med J. 2020;96:156-161. doi:10.1136/postgradmedj-2019-137186
31. Kim AS. Medical management for secondary stroke prevention. Continuum (Minneap Minn). 2020;26:435-456. doi:10.1212/CON.0000000000000849
32. Smith L, Chakraborty D, Bhattacharya P, et al. Exposure to hypoglycemia and risk of stroke. Ann N Y Acad Sci. 2018;1431:25-34. doi:10.1111/nyas.13872
33. Amarenco P, Bogousslavsky J, Callahan A 3rd, et al; . High-dose atorvastatin after stroke or transient ischemic attack. N Engl J Med. 2006;355:549-559. doi:10.1056/NEJMoa061894
34. Castilla-Guerra, L, Fernandez-Moreno M, Leon-Jimenez D, et al. Statins in ischemic stroke prevention: what have we learned in the post-SPARCL (The Stroke Prevention by Aggressive Reduction in Cholesterol Levels) decade? Curr Treat Options Neurol. 2019;21:22. doi: 10.1007/s11940-019-0563-4
35. Bohula EA, Wiviott SD, Giugliano RP, et al. Prevention of stroke with the addition of ezetimibe to statin therapy in patients with acute coronary syndrome in IMPROVE-IT (Improved Reduction of Outcomes: Vytorin Efficacy International Trial). Circulation. 2017;136:2440-2450. doi:10.1161/CIRCULATIONAHA.117.029095
36. Moritsugu KP. The 2006 report of the Surgeon General: the health consequences of involuntary exposure to tobacco smoke. Am J Prev Med. 20067;32:542-543. doi: 10.1016/j.amepre.2007.02.026
37. Wolf PA, D’Agostino RB, Kannel WB, et al. Cigarette smoking as a risk factor for stroke: the Framingham Study. JAMA. 1988;259:1025-1029.
38. Goldstein LB, Adams R, Alberts MJ, et al. Primary prevention of ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council: cosponsored by the Atherosclerotic Peripheral Vascular Disease Interdisciplinary Working Group; Cardiovascular Nursing Council; Clinical Cardiology Council; Nutrition, Physical Activity, and Metabolism Council; and the Quality of Care and Outcomes Research Interdisciplinary Working Group: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37:1583-1633. doi: 10.1161/01.STR.0000223048.70103.F1
39. Klijn CJ, Paciaroni M, Berge E, et al. Antithrombotic treatment for secondary prevention of stroke and other thromboembolic events in patients with stroke or transient ischemic attack and non-valvular atrial fibrillation: A European Stroke Organisation guideline. Eur Stroke J. 2019;4:198-223. doi:10.1177/2396987319841187
40. Antithrombotic Trialists’ (ATT) Collaboration; Baigent C, Blackwell L, Collins R, et al. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373:1849-1860. doi:10.1016/S0140-6736(09)60503-1
41. Singer DE, Albers GW, Dalen JE, et al. Antithrombotic therapy in atrial fibrillation: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 suppl):546S–592S. doi: 10.1378/chest.08-0678
42. Rothwell PM, Algra A, Chen Z, et al. Effects of aspirin on risk and severity of early recurrent stroke after transient ischaemic attack and ischaemic stroke: time-course analysis of randomised trials. Lancet. 2016;388:365-375. doi:10.1016/S0140-6736(16)30468-8
PRACTICE RECOMMENDATIONS
In the hospital, the treating physician should:
› Immediately initiate brain imaging with diffusion-weighted magnetic resonance imaging when TIA is suspected, upon the patient’s arrival at the hospital. A
› Control blood pressure when a TIA is confirmed, to decrease the risk of recurrent stroke. A
› Initiate antiplatelet therapy, to decrease the risk of recurrent stroke. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Mechanical touch therapy device promising for anxiety
An at-home investigational device is a promising noninvasive therapeutic approach for anxiety disorders, results from an open-label pilot trial suggest.
The small study showed
“MATT is part of a large movement toward developing therapeutic devices that patients can self-administer at home,” study author Linda L. Carpenter, MD, professor of psychiatry at Brown University and director of the Neuromodulation & Neuroimaging Core at Butler Hospital, both in Providence, R.I., told this news organization, adding that the new study is a step in the right direction of improving the technology used to treat anxiety disorders.
The study was published online in Frontiers in Psychiatry.
Robust safety profile
Therapeutic noninvasive peripheral nerve stimulation is under investigation for anxiety as well as pain and depression. Nerve activation is achieved by delivering electrical or mechanical energy, although most devices to date have used electrical stimulation.
Although electrical stimulation is considered low risk, mechanical stimulation that activates somatosensory pathways has an even more robust safety profile, the investigators note.
The MATT device targets C-tactile fibers (CT) specialized unmyelinated Group C peripheral nerve fibers that fire when stroked at velocities perceived as pleasurable or comforting.
To use the device, participants wear a headset with a small vibrating piece that sits on the mastoid bone behind each ear. These pieces deliver gentle vibrations that can be adjusted by patients.
During development of the MATT stimulation, researchers noted that an isochronic 10 Hz wave, cycling 2 seconds on and 2 seconds off, induced a state of relaxation and increased occipital alpha oscillations in pilot study participants.
The current study was designed to confirm preliminary efficacy and feasibility signals. The sample included 22 patients (mean age 37.3 years, 72.7% female, 77.3% White). All study participants were diagnosed with an anxiety disorder and had at least moderately severe anxiety symptoms. Some also had symptoms of panic or depression.
Many participants were on medications that weren’t effective, and they wanted to find a nondrug method of relieving their symptoms, said Dr. Carpenter.
What’s the mechanism?
Participants learned how to administer the stimulation and adjust the intensity of vibrations to a level where it was consistently detectable but not uncomfortable. Then they received a MATT device to use at home at least twice daily for 20 minutes.
Patients kept daily diaries documenting device use, adverse effects, and technological problems. In-person assessments were held at 2 and 4 weeks.
Researchers collected resting EEG immediately before, and after, the second stimulation session and again following 4 weeks of MATT use.
At baseline and after 2 and 4 weeks, patients self-reported anxiety using the 7-item Generalized Anxiety Disorder (GAD-7) scale, depression with the Beck Depression Inventory (BDI), and stress using the Perceived Stress Scale (PSS). They also reported symptoms with the Depression, Anxiety, Stress Scale (DASS).
Researchers also investigated “interoceptive awareness” or being mindful of your body and internal feelings. For this, they had participants complete the 32-item Multidimensional Assessment of Interoceptive Awareness pre- and post treatment.
Interoceptive awareness “is a whole new area of interest in neuroscience and brain health,” said Dr. Carpenter. “The hypothesis was that one way this device might work is that vibrations would travel to the insular cortex, the part of the brain that involves mindfulness and self-awareness.”
Symptom reduction
In the completer sample of 17 participants, mean scores on anxiety and depression symptoms fell significantly from baseline to 4 weeks (all P < .01). For example, the GAD-7 mean score fell from 14.3 to 7.1 and the BDI mean score from 30.6 to 14.8.
The study also showed that mindfulness was enhanced. The MAIA total score increased from 83.1 to 93.5 (P = .014).
Device users had increased alpha and theta brainwave activity, findings that “go along with the concept of decreased anxiety,” said Dr. Carpenter. She noted a recent study of the same patient population showed the device enhanced functional brain connectivity.
This current study was too small to pick up signals showing the device was effective in any particular subpopulation, said Dr. Carpenter.
Unlike other stimulation interventions that require clinic visits, patients use the MATT in the comfort of their own home and at their own convenience.
However, there are still questions surrounding the use of the noninvasive device. For example, said Dr. Carpenter, it’s unclear if it would be more effective if combined with psychotherapy or whether patients can use it while sleeping and driving. A next step could be a sham-controlled trial, she said.
The study was supported by Affect Neuro, developer of MATT therapy, and the National Institute of General Medical Sciences. Dr. Carpenter reports receiving a consultancy fee from Affect Neuro.
A version of this article first appeared on Medscape.com.
An at-home investigational device is a promising noninvasive therapeutic approach for anxiety disorders, results from an open-label pilot trial suggest.
The small study showed
“MATT is part of a large movement toward developing therapeutic devices that patients can self-administer at home,” study author Linda L. Carpenter, MD, professor of psychiatry at Brown University and director of the Neuromodulation & Neuroimaging Core at Butler Hospital, both in Providence, R.I., told this news organization, adding that the new study is a step in the right direction of improving the technology used to treat anxiety disorders.
The study was published online in Frontiers in Psychiatry.
Robust safety profile
Therapeutic noninvasive peripheral nerve stimulation is under investigation for anxiety as well as pain and depression. Nerve activation is achieved by delivering electrical or mechanical energy, although most devices to date have used electrical stimulation.
Although electrical stimulation is considered low risk, mechanical stimulation that activates somatosensory pathways has an even more robust safety profile, the investigators note.
The MATT device targets C-tactile fibers (CT) specialized unmyelinated Group C peripheral nerve fibers that fire when stroked at velocities perceived as pleasurable or comforting.
To use the device, participants wear a headset with a small vibrating piece that sits on the mastoid bone behind each ear. These pieces deliver gentle vibrations that can be adjusted by patients.
During development of the MATT stimulation, researchers noted that an isochronic 10 Hz wave, cycling 2 seconds on and 2 seconds off, induced a state of relaxation and increased occipital alpha oscillations in pilot study participants.
The current study was designed to confirm preliminary efficacy and feasibility signals. The sample included 22 patients (mean age 37.3 years, 72.7% female, 77.3% White). All study participants were diagnosed with an anxiety disorder and had at least moderately severe anxiety symptoms. Some also had symptoms of panic or depression.
Many participants were on medications that weren’t effective, and they wanted to find a nondrug method of relieving their symptoms, said Dr. Carpenter.
What’s the mechanism?
Participants learned how to administer the stimulation and adjust the intensity of vibrations to a level where it was consistently detectable but not uncomfortable. Then they received a MATT device to use at home at least twice daily for 20 minutes.
Patients kept daily diaries documenting device use, adverse effects, and technological problems. In-person assessments were held at 2 and 4 weeks.
Researchers collected resting EEG immediately before, and after, the second stimulation session and again following 4 weeks of MATT use.
At baseline and after 2 and 4 weeks, patients self-reported anxiety using the 7-item Generalized Anxiety Disorder (GAD-7) scale, depression with the Beck Depression Inventory (BDI), and stress using the Perceived Stress Scale (PSS). They also reported symptoms with the Depression, Anxiety, Stress Scale (DASS).
Researchers also investigated “interoceptive awareness” or being mindful of your body and internal feelings. For this, they had participants complete the 32-item Multidimensional Assessment of Interoceptive Awareness pre- and post treatment.
Interoceptive awareness “is a whole new area of interest in neuroscience and brain health,” said Dr. Carpenter. “The hypothesis was that one way this device might work is that vibrations would travel to the insular cortex, the part of the brain that involves mindfulness and self-awareness.”
Symptom reduction
In the completer sample of 17 participants, mean scores on anxiety and depression symptoms fell significantly from baseline to 4 weeks (all P < .01). For example, the GAD-7 mean score fell from 14.3 to 7.1 and the BDI mean score from 30.6 to 14.8.
The study also showed that mindfulness was enhanced. The MAIA total score increased from 83.1 to 93.5 (P = .014).
Device users had increased alpha and theta brainwave activity, findings that “go along with the concept of decreased anxiety,” said Dr. Carpenter. She noted a recent study of the same patient population showed the device enhanced functional brain connectivity.
This current study was too small to pick up signals showing the device was effective in any particular subpopulation, said Dr. Carpenter.
Unlike other stimulation interventions that require clinic visits, patients use the MATT in the comfort of their own home and at their own convenience.
However, there are still questions surrounding the use of the noninvasive device. For example, said Dr. Carpenter, it’s unclear if it would be more effective if combined with psychotherapy or whether patients can use it while sleeping and driving. A next step could be a sham-controlled trial, she said.
The study was supported by Affect Neuro, developer of MATT therapy, and the National Institute of General Medical Sciences. Dr. Carpenter reports receiving a consultancy fee from Affect Neuro.
A version of this article first appeared on Medscape.com.
An at-home investigational device is a promising noninvasive therapeutic approach for anxiety disorders, results from an open-label pilot trial suggest.
The small study showed
“MATT is part of a large movement toward developing therapeutic devices that patients can self-administer at home,” study author Linda L. Carpenter, MD, professor of psychiatry at Brown University and director of the Neuromodulation & Neuroimaging Core at Butler Hospital, both in Providence, R.I., told this news organization, adding that the new study is a step in the right direction of improving the technology used to treat anxiety disorders.
The study was published online in Frontiers in Psychiatry.
Robust safety profile
Therapeutic noninvasive peripheral nerve stimulation is under investigation for anxiety as well as pain and depression. Nerve activation is achieved by delivering electrical or mechanical energy, although most devices to date have used electrical stimulation.
Although electrical stimulation is considered low risk, mechanical stimulation that activates somatosensory pathways has an even more robust safety profile, the investigators note.
The MATT device targets C-tactile fibers (CT) specialized unmyelinated Group C peripheral nerve fibers that fire when stroked at velocities perceived as pleasurable or comforting.
To use the device, participants wear a headset with a small vibrating piece that sits on the mastoid bone behind each ear. These pieces deliver gentle vibrations that can be adjusted by patients.
During development of the MATT stimulation, researchers noted that an isochronic 10 Hz wave, cycling 2 seconds on and 2 seconds off, induced a state of relaxation and increased occipital alpha oscillations in pilot study participants.
The current study was designed to confirm preliminary efficacy and feasibility signals. The sample included 22 patients (mean age 37.3 years, 72.7% female, 77.3% White). All study participants were diagnosed with an anxiety disorder and had at least moderately severe anxiety symptoms. Some also had symptoms of panic or depression.
Many participants were on medications that weren’t effective, and they wanted to find a nondrug method of relieving their symptoms, said Dr. Carpenter.
What’s the mechanism?
Participants learned how to administer the stimulation and adjust the intensity of vibrations to a level where it was consistently detectable but not uncomfortable. Then they received a MATT device to use at home at least twice daily for 20 minutes.
Patients kept daily diaries documenting device use, adverse effects, and technological problems. In-person assessments were held at 2 and 4 weeks.
Researchers collected resting EEG immediately before, and after, the second stimulation session and again following 4 weeks of MATT use.
At baseline and after 2 and 4 weeks, patients self-reported anxiety using the 7-item Generalized Anxiety Disorder (GAD-7) scale, depression with the Beck Depression Inventory (BDI), and stress using the Perceived Stress Scale (PSS). They also reported symptoms with the Depression, Anxiety, Stress Scale (DASS).
Researchers also investigated “interoceptive awareness” or being mindful of your body and internal feelings. For this, they had participants complete the 32-item Multidimensional Assessment of Interoceptive Awareness pre- and post treatment.
Interoceptive awareness “is a whole new area of interest in neuroscience and brain health,” said Dr. Carpenter. “The hypothesis was that one way this device might work is that vibrations would travel to the insular cortex, the part of the brain that involves mindfulness and self-awareness.”
Symptom reduction
In the completer sample of 17 participants, mean scores on anxiety and depression symptoms fell significantly from baseline to 4 weeks (all P < .01). For example, the GAD-7 mean score fell from 14.3 to 7.1 and the BDI mean score from 30.6 to 14.8.
The study also showed that mindfulness was enhanced. The MAIA total score increased from 83.1 to 93.5 (P = .014).
Device users had increased alpha and theta brainwave activity, findings that “go along with the concept of decreased anxiety,” said Dr. Carpenter. She noted a recent study of the same patient population showed the device enhanced functional brain connectivity.
This current study was too small to pick up signals showing the device was effective in any particular subpopulation, said Dr. Carpenter.
Unlike other stimulation interventions that require clinic visits, patients use the MATT in the comfort of their own home and at their own convenience.
However, there are still questions surrounding the use of the noninvasive device. For example, said Dr. Carpenter, it’s unclear if it would be more effective if combined with psychotherapy or whether patients can use it while sleeping and driving. A next step could be a sham-controlled trial, she said.
The study was supported by Affect Neuro, developer of MATT therapy, and the National Institute of General Medical Sciences. Dr. Carpenter reports receiving a consultancy fee from Affect Neuro.
A version of this article first appeared on Medscape.com.
Best antioxidants to prevent age-related dementia identified?
Investigators found that individuals with the highest serum levels of lutein + zeaxanthin and beta-cryptoxanthin at baseline were less likely to have dementia decades later than were their peers with lower levels of these antioxidants.
Lutein and zeaxanthin are found in green leafy vegetables such as kale, spinach, broccoli, and peas. Beta-cryptoxanthin is found in fruits such as oranges, papaya, tangerines, and persimmons.
“Antioxidants may help protect the brain from oxidative stress, which can cause cell damage,” first author May A. Beydoun, PhD, with the National Institute on Aging (NIA), said in a news release.
“This is the first nationally representative study to analyze blood levels of antioxidants in relation to dementia risk,” NIA scientific director Luigi Ferrucci, MD, said in an interview.
“Blood test results may be more representative of the actual antioxidant level than a person’s report of what kind of foods they regularly consume,” Dr. Ferrucci added.
The study was published online in Neurology.
Reduced dementia risk
The researchers tested associations and interactions of serum vitamins A, C and E, and total and individual serum carotenoids and interactions with incident Alzheimer’s disease (AD) and all-cause dementia.
They analyzed data from 7,283 participants in the Third National Health and Nutrition Examination Survey (NHANES III) who were at least 45 years old at baseline and followed for an average of 16-17 years.
They found serum levels of lutein + zeaxanthin were associated with reduced risk of all-cause dementia among people aged 65 and older in models adjusted for lifestyle.
For lutein + zeaxanthin, every standard deviation (SD) increase (roughly 15.4 µmol/liter) was associated with a 7% decrease in risk for dementia (hazard ratio [HR] 0.93; 95% confidence interval [CI], 0.87-0.99, P = .037). This association was attenuated somewhat after adjustment for socioeconomic status.
Serum levels of beta-cryptoxanthin showed a “strong” inverse relationship with all-cause dementia in age- and sex-adjusted models.
For beta-cryptoxanthin, every SD increase (roughly 8.6 µmol/liter) was associated with a 14% reduced risk for dementia in people aged 45 and older (HR, 0.86; 95% CI, 0.80-0.93, P < .001) and 65 and older (HR, 0.86; 95% CI, 0.80-0.93, P = .001).
This relationship remained strong in models adjusted for sociodemographic and socioeconomic factors but attenuated in subsequent models.
No associations were found for lycopene, alpha-carotene, beta-carotene, or vitamins A, C, or E in the fully adjusted models.
Antagonistic interactions were observed for vitamin A and alpha-carotene, vitamin A and beta-carotene, vitamin E and lycopene, and lycopene and beta-carotene, suggesting putative protective effects of one antioxidant at lower levels of the other, the researchers noted.
“This analysis of an observational study found that the most important carotenoids in potentially protecting the brain may be lutein + zeaxanthin and beta-cryptoxanthin. However, randomized controlled trials are needed to prove causality,” said Dr. Ferrucci.
“Experts do not yet know the daily level of antioxidant intake to promote healthy aging of the brain. More research is needed to establish the necessary level of antioxidant intake – through the diet and/or supplements – to promote brain health and healthy aging,” he added.
An important step forward
In an accompanying editorial, Babak Hooshmand, MD, PhD, and Miia Kivipelto, MD, PhD, with Karolinska Institute, Stockholm, noted that while nutrition and dietary components are “potential targets” for dementia risk reduction, observational studies to date have reported “inconsistent findings.”
This study is “an important step towards exploring the complex relationship between antioxidants and dementia because it accounts for factors that could possibly influence the associations and considers interactions between different components,” they wrote.
The findings are “challenging,” they added, because they may lead to the hypothesis that inhibition of oxidative damage by antioxidants might have beneficial effects on preventing dementia.
However, clinical trials of antioxidant supplementation have been mainly “disappointing” and a recent Cochrane review found a lack of evidence for supplement use to preserve cognitive function or prevent dementia, Dr. Hooshmand and Dr. Kivipelto noted.
They added that the study contributes to the belief that antioxidants don’t act independently of each other or other factors, including socioeconomic status and lifestyle, in the mediation of dementia risk.
“A careful examination of the evidence is required to learn how antioxidants influence the complex pathology of dementia, because it appears to be more to it than meets the eye,”they concluded.
The research was supported in part by the Intramural Research Program of the National Institutes of Health and the National Institute on Aging. Dr. Beydoun, Dr. Ferrucci, and Dr. Hooshmand report no relevant disclosures. Dr. Kivipelto has supported advisory boards for Combinostics, Roche, and Biogen.
A version of this article first appeared on Medscape.com.
Investigators found that individuals with the highest serum levels of lutein + zeaxanthin and beta-cryptoxanthin at baseline were less likely to have dementia decades later than were their peers with lower levels of these antioxidants.
Lutein and zeaxanthin are found in green leafy vegetables such as kale, spinach, broccoli, and peas. Beta-cryptoxanthin is found in fruits such as oranges, papaya, tangerines, and persimmons.
“Antioxidants may help protect the brain from oxidative stress, which can cause cell damage,” first author May A. Beydoun, PhD, with the National Institute on Aging (NIA), said in a news release.
“This is the first nationally representative study to analyze blood levels of antioxidants in relation to dementia risk,” NIA scientific director Luigi Ferrucci, MD, said in an interview.
“Blood test results may be more representative of the actual antioxidant level than a person’s report of what kind of foods they regularly consume,” Dr. Ferrucci added.
The study was published online in Neurology.
Reduced dementia risk
The researchers tested associations and interactions of serum vitamins A, C and E, and total and individual serum carotenoids and interactions with incident Alzheimer’s disease (AD) and all-cause dementia.
They analyzed data from 7,283 participants in the Third National Health and Nutrition Examination Survey (NHANES III) who were at least 45 years old at baseline and followed for an average of 16-17 years.
They found serum levels of lutein + zeaxanthin were associated with reduced risk of all-cause dementia among people aged 65 and older in models adjusted for lifestyle.
For lutein + zeaxanthin, every standard deviation (SD) increase (roughly 15.4 µmol/liter) was associated with a 7% decrease in risk for dementia (hazard ratio [HR] 0.93; 95% confidence interval [CI], 0.87-0.99, P = .037). This association was attenuated somewhat after adjustment for socioeconomic status.
Serum levels of beta-cryptoxanthin showed a “strong” inverse relationship with all-cause dementia in age- and sex-adjusted models.
For beta-cryptoxanthin, every SD increase (roughly 8.6 µmol/liter) was associated with a 14% reduced risk for dementia in people aged 45 and older (HR, 0.86; 95% CI, 0.80-0.93, P < .001) and 65 and older (HR, 0.86; 95% CI, 0.80-0.93, P = .001).
This relationship remained strong in models adjusted for sociodemographic and socioeconomic factors but attenuated in subsequent models.
No associations were found for lycopene, alpha-carotene, beta-carotene, or vitamins A, C, or E in the fully adjusted models.
Antagonistic interactions were observed for vitamin A and alpha-carotene, vitamin A and beta-carotene, vitamin E and lycopene, and lycopene and beta-carotene, suggesting putative protective effects of one antioxidant at lower levels of the other, the researchers noted.
“This analysis of an observational study found that the most important carotenoids in potentially protecting the brain may be lutein + zeaxanthin and beta-cryptoxanthin. However, randomized controlled trials are needed to prove causality,” said Dr. Ferrucci.
“Experts do not yet know the daily level of antioxidant intake to promote healthy aging of the brain. More research is needed to establish the necessary level of antioxidant intake – through the diet and/or supplements – to promote brain health and healthy aging,” he added.
An important step forward
In an accompanying editorial, Babak Hooshmand, MD, PhD, and Miia Kivipelto, MD, PhD, with Karolinska Institute, Stockholm, noted that while nutrition and dietary components are “potential targets” for dementia risk reduction, observational studies to date have reported “inconsistent findings.”
This study is “an important step towards exploring the complex relationship between antioxidants and dementia because it accounts for factors that could possibly influence the associations and considers interactions between different components,” they wrote.
The findings are “challenging,” they added, because they may lead to the hypothesis that inhibition of oxidative damage by antioxidants might have beneficial effects on preventing dementia.
However, clinical trials of antioxidant supplementation have been mainly “disappointing” and a recent Cochrane review found a lack of evidence for supplement use to preserve cognitive function or prevent dementia, Dr. Hooshmand and Dr. Kivipelto noted.
They added that the study contributes to the belief that antioxidants don’t act independently of each other or other factors, including socioeconomic status and lifestyle, in the mediation of dementia risk.
“A careful examination of the evidence is required to learn how antioxidants influence the complex pathology of dementia, because it appears to be more to it than meets the eye,”they concluded.
The research was supported in part by the Intramural Research Program of the National Institutes of Health and the National Institute on Aging. Dr. Beydoun, Dr. Ferrucci, and Dr. Hooshmand report no relevant disclosures. Dr. Kivipelto has supported advisory boards for Combinostics, Roche, and Biogen.
A version of this article first appeared on Medscape.com.
Investigators found that individuals with the highest serum levels of lutein + zeaxanthin and beta-cryptoxanthin at baseline were less likely to have dementia decades later than were their peers with lower levels of these antioxidants.
Lutein and zeaxanthin are found in green leafy vegetables such as kale, spinach, broccoli, and peas. Beta-cryptoxanthin is found in fruits such as oranges, papaya, tangerines, and persimmons.
“Antioxidants may help protect the brain from oxidative stress, which can cause cell damage,” first author May A. Beydoun, PhD, with the National Institute on Aging (NIA), said in a news release.
“This is the first nationally representative study to analyze blood levels of antioxidants in relation to dementia risk,” NIA scientific director Luigi Ferrucci, MD, said in an interview.
“Blood test results may be more representative of the actual antioxidant level than a person’s report of what kind of foods they regularly consume,” Dr. Ferrucci added.
The study was published online in Neurology.
Reduced dementia risk
The researchers tested associations and interactions of serum vitamins A, C and E, and total and individual serum carotenoids and interactions with incident Alzheimer’s disease (AD) and all-cause dementia.
They analyzed data from 7,283 participants in the Third National Health and Nutrition Examination Survey (NHANES III) who were at least 45 years old at baseline and followed for an average of 16-17 years.
They found serum levels of lutein + zeaxanthin were associated with reduced risk of all-cause dementia among people aged 65 and older in models adjusted for lifestyle.
For lutein + zeaxanthin, every standard deviation (SD) increase (roughly 15.4 µmol/liter) was associated with a 7% decrease in risk for dementia (hazard ratio [HR] 0.93; 95% confidence interval [CI], 0.87-0.99, P = .037). This association was attenuated somewhat after adjustment for socioeconomic status.
Serum levels of beta-cryptoxanthin showed a “strong” inverse relationship with all-cause dementia in age- and sex-adjusted models.
For beta-cryptoxanthin, every SD increase (roughly 8.6 µmol/liter) was associated with a 14% reduced risk for dementia in people aged 45 and older (HR, 0.86; 95% CI, 0.80-0.93, P < .001) and 65 and older (HR, 0.86; 95% CI, 0.80-0.93, P = .001).
This relationship remained strong in models adjusted for sociodemographic and socioeconomic factors but attenuated in subsequent models.
No associations were found for lycopene, alpha-carotene, beta-carotene, or vitamins A, C, or E in the fully adjusted models.
Antagonistic interactions were observed for vitamin A and alpha-carotene, vitamin A and beta-carotene, vitamin E and lycopene, and lycopene and beta-carotene, suggesting putative protective effects of one antioxidant at lower levels of the other, the researchers noted.
“This analysis of an observational study found that the most important carotenoids in potentially protecting the brain may be lutein + zeaxanthin and beta-cryptoxanthin. However, randomized controlled trials are needed to prove causality,” said Dr. Ferrucci.
“Experts do not yet know the daily level of antioxidant intake to promote healthy aging of the brain. More research is needed to establish the necessary level of antioxidant intake – through the diet and/or supplements – to promote brain health and healthy aging,” he added.
An important step forward
In an accompanying editorial, Babak Hooshmand, MD, PhD, and Miia Kivipelto, MD, PhD, with Karolinska Institute, Stockholm, noted that while nutrition and dietary components are “potential targets” for dementia risk reduction, observational studies to date have reported “inconsistent findings.”
This study is “an important step towards exploring the complex relationship between antioxidants and dementia because it accounts for factors that could possibly influence the associations and considers interactions between different components,” they wrote.
The findings are “challenging,” they added, because they may lead to the hypothesis that inhibition of oxidative damage by antioxidants might have beneficial effects on preventing dementia.
However, clinical trials of antioxidant supplementation have been mainly “disappointing” and a recent Cochrane review found a lack of evidence for supplement use to preserve cognitive function or prevent dementia, Dr. Hooshmand and Dr. Kivipelto noted.
They added that the study contributes to the belief that antioxidants don’t act independently of each other or other factors, including socioeconomic status and lifestyle, in the mediation of dementia risk.
“A careful examination of the evidence is required to learn how antioxidants influence the complex pathology of dementia, because it appears to be more to it than meets the eye,”they concluded.
The research was supported in part by the Intramural Research Program of the National Institutes of Health and the National Institute on Aging. Dr. Beydoun, Dr. Ferrucci, and Dr. Hooshmand report no relevant disclosures. Dr. Kivipelto has supported advisory boards for Combinostics, Roche, and Biogen.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Study provides new analysis of isotretinoin and risk for adverse neuropsychiatric outcomes
The use of , in a large retrospective cohort study published in the British Journal of Dermatology.
Although severe neuropsychiatric effects associated with isotretinoin therapy in patients with acne have been reported, “the evidence base ... is mixed and inconclusive,” and many studies are small, Seena Fazel, MBChB, MD, of the department of psychiatry, Oxford University, England, and co-authors write in the study.
The study results suggest that isotretinoin is conferring protection against adverse neuropsychiatric outcomes, particularly when compared with using oral antibiotics to treat acne, Dr. Fazel, professor of forensic psychiatry at Oxford University and the study’s senior author, said in an interview.
In the study, the investigators reviewed electronic health records (2013-2019) from a primarily United States–based dataset (TriNetX) of patients with acne aged 12-27 who had been followed for up to 1 year after their prescriptions had been dispensed.
There were four arms: those prescribed isotretinoin (30,866), oral antibiotics (44,748), topical anti-acne treatments (108,367), and those who had not been prescribed any acne treatment (78,666). The primary outcomes were diagnoses of a neuropsychiatric disorder (psychotic, mood, anxiety, personality, behavioral, and sleep disorders; and non-fatal self-harm) within one year of being prescribed treatment.
After using propensity score matching to adjust for confounders at baseline, the investigators determined that the odds ratio for any incident neuropsychiatric outcomes among patients with acne treated with isotretinoin was 0.80 (95% confidence interval, 0.74-0.87), compared with patients on oral antibiotics; 0.94 (95% CI, 0.87-1.02), compared with patients on topical anti-acne medications; and 1.06 (95% CI, 0.97-1.16), compared with those without a prescription for anti-acne medicines.
Side effects of isotretinoin – such as headache, dry mouth, and fatigue – were higher among those on isotretinoin than in the other three groups.
The authors concluded that isotretinoin was not independently linked to excess adverse neuropsychiatric outcomes at a population level. “We observed a consistent association between increasing acne severity as indicated by anti-acne treatment options and incidence of adverse neuropsychiatric outcomes, but the findings showed that isotretinoin exposure did not add to the risk of neuropsychiatric adverse outcomes over and above what was associated with oral antibiotics,” they write.
Isotretinoin treatment “appeared to mitigate the excess neuropsychiatric risk associated with recalcitrant moderate-to-severe acne,” they add.
The dermatology community has been interested in the impact isotretinoin has on mental health, and “I think clinically, they see that people get better on isotretinoin and their mental health improves,” Dr. Fazel told this news organization.
Asked to comment on the study results, John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, commended the investigators for the design of the trial.
“One of the strengths of this study is that they use a technique called propensity-score matching, where you try to make the groups of patients similar with respect to their other characteristics to minimize the risks of confounding and bias in the study, which I think is a real strength,” he told this news organization. “The other thing that they do, which I think is a strength, is to think about the impact of acne severity on these outcomes, because we know acne itself is associated with depression and risk for suicide and other neuropsychiatric outcomes.”
Including a cohort of patients who had acne and received oral antibiotics for comparison “is a nice way to address the potential for confounding by severity and confounding by indication,” Dr. Barbieri said. “Those who get antibiotics usually have more severe acne. They may not have it as severely as those who get isotretinoin, but it is a nice approach to account for background levels of depression and neuropsychiatric outcomes in patients with acne. I think that is a real strength of the study. This is one of the best studies to have looked at this question.”
However, although the study found that isotretinoin decreased the excess psychiatric risk associated with refractory moderate-to-severe acne, it does not rule out the possibility that individuals may experience an adverse psychiatric outcome while on isotretinoin, Dr. Barbieri said.
“While I think on a population level, we absolutely can feel reassured by these data, I do think there are individual patients who have idiosyncratic, unpredictable reactions to isotretinoin where they have mood changes, whether it be irritability, depression, or other mood changes,” he cautioned. “Given the association of acne itself with mental health comorbidities, it is important to screen for comorbidities such as depression in all patients with acne.”
The study was funded by the Wellcome Trust, which provided Dr. Fazel and the first author with financial support for the study. One author is an employee of TriNetX; the other authors had no relevant disclosures. Dr. Barbieri reported no financial disclosures. He is cochair of the AAD’s Acne Guidelines Workgroup and associate editor at JAMA Dermatology.
A version of this article first appeared on Medscape.com.
The use of , in a large retrospective cohort study published in the British Journal of Dermatology.
Although severe neuropsychiatric effects associated with isotretinoin therapy in patients with acne have been reported, “the evidence base ... is mixed and inconclusive,” and many studies are small, Seena Fazel, MBChB, MD, of the department of psychiatry, Oxford University, England, and co-authors write in the study.
The study results suggest that isotretinoin is conferring protection against adverse neuropsychiatric outcomes, particularly when compared with using oral antibiotics to treat acne, Dr. Fazel, professor of forensic psychiatry at Oxford University and the study’s senior author, said in an interview.
In the study, the investigators reviewed electronic health records (2013-2019) from a primarily United States–based dataset (TriNetX) of patients with acne aged 12-27 who had been followed for up to 1 year after their prescriptions had been dispensed.
There were four arms: those prescribed isotretinoin (30,866), oral antibiotics (44,748), topical anti-acne treatments (108,367), and those who had not been prescribed any acne treatment (78,666). The primary outcomes were diagnoses of a neuropsychiatric disorder (psychotic, mood, anxiety, personality, behavioral, and sleep disorders; and non-fatal self-harm) within one year of being prescribed treatment.
After using propensity score matching to adjust for confounders at baseline, the investigators determined that the odds ratio for any incident neuropsychiatric outcomes among patients with acne treated with isotretinoin was 0.80 (95% confidence interval, 0.74-0.87), compared with patients on oral antibiotics; 0.94 (95% CI, 0.87-1.02), compared with patients on topical anti-acne medications; and 1.06 (95% CI, 0.97-1.16), compared with those without a prescription for anti-acne medicines.
Side effects of isotretinoin – such as headache, dry mouth, and fatigue – were higher among those on isotretinoin than in the other three groups.
The authors concluded that isotretinoin was not independently linked to excess adverse neuropsychiatric outcomes at a population level. “We observed a consistent association between increasing acne severity as indicated by anti-acne treatment options and incidence of adverse neuropsychiatric outcomes, but the findings showed that isotretinoin exposure did not add to the risk of neuropsychiatric adverse outcomes over and above what was associated with oral antibiotics,” they write.
Isotretinoin treatment “appeared to mitigate the excess neuropsychiatric risk associated with recalcitrant moderate-to-severe acne,” they add.
The dermatology community has been interested in the impact isotretinoin has on mental health, and “I think clinically, they see that people get better on isotretinoin and their mental health improves,” Dr. Fazel told this news organization.
Asked to comment on the study results, John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, commended the investigators for the design of the trial.
“One of the strengths of this study is that they use a technique called propensity-score matching, where you try to make the groups of patients similar with respect to their other characteristics to minimize the risks of confounding and bias in the study, which I think is a real strength,” he told this news organization. “The other thing that they do, which I think is a strength, is to think about the impact of acne severity on these outcomes, because we know acne itself is associated with depression and risk for suicide and other neuropsychiatric outcomes.”
Including a cohort of patients who had acne and received oral antibiotics for comparison “is a nice way to address the potential for confounding by severity and confounding by indication,” Dr. Barbieri said. “Those who get antibiotics usually have more severe acne. They may not have it as severely as those who get isotretinoin, but it is a nice approach to account for background levels of depression and neuropsychiatric outcomes in patients with acne. I think that is a real strength of the study. This is one of the best studies to have looked at this question.”
However, although the study found that isotretinoin decreased the excess psychiatric risk associated with refractory moderate-to-severe acne, it does not rule out the possibility that individuals may experience an adverse psychiatric outcome while on isotretinoin, Dr. Barbieri said.
“While I think on a population level, we absolutely can feel reassured by these data, I do think there are individual patients who have idiosyncratic, unpredictable reactions to isotretinoin where they have mood changes, whether it be irritability, depression, or other mood changes,” he cautioned. “Given the association of acne itself with mental health comorbidities, it is important to screen for comorbidities such as depression in all patients with acne.”
The study was funded by the Wellcome Trust, which provided Dr. Fazel and the first author with financial support for the study. One author is an employee of TriNetX; the other authors had no relevant disclosures. Dr. Barbieri reported no financial disclosures. He is cochair of the AAD’s Acne Guidelines Workgroup and associate editor at JAMA Dermatology.
A version of this article first appeared on Medscape.com.
The use of , in a large retrospective cohort study published in the British Journal of Dermatology.
Although severe neuropsychiatric effects associated with isotretinoin therapy in patients with acne have been reported, “the evidence base ... is mixed and inconclusive,” and many studies are small, Seena Fazel, MBChB, MD, of the department of psychiatry, Oxford University, England, and co-authors write in the study.
The study results suggest that isotretinoin is conferring protection against adverse neuropsychiatric outcomes, particularly when compared with using oral antibiotics to treat acne, Dr. Fazel, professor of forensic psychiatry at Oxford University and the study’s senior author, said in an interview.
In the study, the investigators reviewed electronic health records (2013-2019) from a primarily United States–based dataset (TriNetX) of patients with acne aged 12-27 who had been followed for up to 1 year after their prescriptions had been dispensed.
There were four arms: those prescribed isotretinoin (30,866), oral antibiotics (44,748), topical anti-acne treatments (108,367), and those who had not been prescribed any acne treatment (78,666). The primary outcomes were diagnoses of a neuropsychiatric disorder (psychotic, mood, anxiety, personality, behavioral, and sleep disorders; and non-fatal self-harm) within one year of being prescribed treatment.
After using propensity score matching to adjust for confounders at baseline, the investigators determined that the odds ratio for any incident neuropsychiatric outcomes among patients with acne treated with isotretinoin was 0.80 (95% confidence interval, 0.74-0.87), compared with patients on oral antibiotics; 0.94 (95% CI, 0.87-1.02), compared with patients on topical anti-acne medications; and 1.06 (95% CI, 0.97-1.16), compared with those without a prescription for anti-acne medicines.
Side effects of isotretinoin – such as headache, dry mouth, and fatigue – were higher among those on isotretinoin than in the other three groups.
The authors concluded that isotretinoin was not independently linked to excess adverse neuropsychiatric outcomes at a population level. “We observed a consistent association between increasing acne severity as indicated by anti-acne treatment options and incidence of adverse neuropsychiatric outcomes, but the findings showed that isotretinoin exposure did not add to the risk of neuropsychiatric adverse outcomes over and above what was associated with oral antibiotics,” they write.
Isotretinoin treatment “appeared to mitigate the excess neuropsychiatric risk associated with recalcitrant moderate-to-severe acne,” they add.
The dermatology community has been interested in the impact isotretinoin has on mental health, and “I think clinically, they see that people get better on isotretinoin and their mental health improves,” Dr. Fazel told this news organization.
Asked to comment on the study results, John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, commended the investigators for the design of the trial.
“One of the strengths of this study is that they use a technique called propensity-score matching, where you try to make the groups of patients similar with respect to their other characteristics to minimize the risks of confounding and bias in the study, which I think is a real strength,” he told this news organization. “The other thing that they do, which I think is a strength, is to think about the impact of acne severity on these outcomes, because we know acne itself is associated with depression and risk for suicide and other neuropsychiatric outcomes.”
Including a cohort of patients who had acne and received oral antibiotics for comparison “is a nice way to address the potential for confounding by severity and confounding by indication,” Dr. Barbieri said. “Those who get antibiotics usually have more severe acne. They may not have it as severely as those who get isotretinoin, but it is a nice approach to account for background levels of depression and neuropsychiatric outcomes in patients with acne. I think that is a real strength of the study. This is one of the best studies to have looked at this question.”
However, although the study found that isotretinoin decreased the excess psychiatric risk associated with refractory moderate-to-severe acne, it does not rule out the possibility that individuals may experience an adverse psychiatric outcome while on isotretinoin, Dr. Barbieri said.
“While I think on a population level, we absolutely can feel reassured by these data, I do think there are individual patients who have idiosyncratic, unpredictable reactions to isotretinoin where they have mood changes, whether it be irritability, depression, or other mood changes,” he cautioned. “Given the association of acne itself with mental health comorbidities, it is important to screen for comorbidities such as depression in all patients with acne.”
The study was funded by the Wellcome Trust, which provided Dr. Fazel and the first author with financial support for the study. One author is an employee of TriNetX; the other authors had no relevant disclosures. Dr. Barbieri reported no financial disclosures. He is cochair of the AAD’s Acne Guidelines Workgroup and associate editor at JAMA Dermatology.
A version of this article first appeared on Medscape.com.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
FDA clears diagnostic test for early Alzheimer’s
The Lumipulse G β-Amyloid Ratio 1-42/1-40 (Fujirebio Diagnostics) test detects amyloid plaques associated with AD in adults age 55 or older who are under investigation for AD and other causes of cognitive decline.
“The availability of an in vitro diagnostic test that can potentially eliminate the need for time-consuming and expensive [positron emission tomography (PET)] scans is great news for individuals and families concerned with the possibility of an Alzheimer’s disease diagnosis,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“With the Lumipulse test, there is a new option that can typically be completed the same day and can give doctors the same information regarding brain amyloid status, without the radiation risk, to help determine if a patient’s cognitive impairment is due to Alzheimer’s disease,” he added.
In its statement, the FDA notes that there is an “unmet need for a reliable and safe test that can accurately identify patients with amyloid plaques consistent with Alzheimer’s disease.”
The agency goes on to state that this new test may eliminate the need to use PET brain scans, a “potentially costly and cumbersome option” to visualize amyloid plaques for the diagnosis of AD.
The Lumipulse test measures the ratio of β-amyloid 1-42 and β-amyloid 1-40 concentrations in human cerebral spinal fluid (CSF). A positive Lumipulse G β-amyloid Ratio (1-42/1-40) test result is consistent with the presence of amyloid plaques, similar to that revealed in a PET scan. A negative result is consistent with a negative amyloid PET scan result.
However, the FDA notes that the test is not a stand-alone assay and should be used in conjunction with other clinical evaluations and additional tests to determine treatment options.
The FDA reports that it evaluated the safety and efficacy of the test in a clinical study of 292 CSF samples from the Alzheimer’s Disease Neuroimaging Initiative sample bank.
The samples were tested by the Lumipulse G β-amyloid Ratio (1-42/1-40) and compared with amyloid PET scan results. In this clinical study, 97% of individuals with Lumipulse G β-amyloid Ratio (1-42/1-40) positive results had the presence of amyloid plaques by PET scan and 84% of individuals with negative results had a negative amyloid PET scan.
The risks associated with the Lumipulse G β-amyloid Ratio (1-42/1-40) test are mainly the possibility of false-positive and false-negative test results.
False-positive results, in conjunction with other clinical information, could lead to an inappropriate diagnosis of, and unnecessary treatment for AD.
False-negative test results could result in additional unnecessary diagnostic tests and potential delay in effective treatment for AD.
The FDA reviewed the device through the De Novo premarket review pathway, a regulatory pathway for low- to moderate-risk devices of a new type.
The agency says this action “creates a new regulatory classification, which means that subsequent devices of the same type with the same intended use may go through FDA’s 510(k) premarket process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a predicate device.”
The Lumipulse G β-amyloid Ratio (1-42/1-40) was granted Breakthrough Device designation, a process designed to expedite the development and review of devices that may provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
A version of this article first appeared on Medscape.com.
The Lumipulse G β-Amyloid Ratio 1-42/1-40 (Fujirebio Diagnostics) test detects amyloid plaques associated with AD in adults age 55 or older who are under investigation for AD and other causes of cognitive decline.
“The availability of an in vitro diagnostic test that can potentially eliminate the need for time-consuming and expensive [positron emission tomography (PET)] scans is great news for individuals and families concerned with the possibility of an Alzheimer’s disease diagnosis,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“With the Lumipulse test, there is a new option that can typically be completed the same day and can give doctors the same information regarding brain amyloid status, without the radiation risk, to help determine if a patient’s cognitive impairment is due to Alzheimer’s disease,” he added.
In its statement, the FDA notes that there is an “unmet need for a reliable and safe test that can accurately identify patients with amyloid plaques consistent with Alzheimer’s disease.”
The agency goes on to state that this new test may eliminate the need to use PET brain scans, a “potentially costly and cumbersome option” to visualize amyloid plaques for the diagnosis of AD.
The Lumipulse test measures the ratio of β-amyloid 1-42 and β-amyloid 1-40 concentrations in human cerebral spinal fluid (CSF). A positive Lumipulse G β-amyloid Ratio (1-42/1-40) test result is consistent with the presence of amyloid plaques, similar to that revealed in a PET scan. A negative result is consistent with a negative amyloid PET scan result.
However, the FDA notes that the test is not a stand-alone assay and should be used in conjunction with other clinical evaluations and additional tests to determine treatment options.
The FDA reports that it evaluated the safety and efficacy of the test in a clinical study of 292 CSF samples from the Alzheimer’s Disease Neuroimaging Initiative sample bank.
The samples were tested by the Lumipulse G β-amyloid Ratio (1-42/1-40) and compared with amyloid PET scan results. In this clinical study, 97% of individuals with Lumipulse G β-amyloid Ratio (1-42/1-40) positive results had the presence of amyloid plaques by PET scan and 84% of individuals with negative results had a negative amyloid PET scan.
The risks associated with the Lumipulse G β-amyloid Ratio (1-42/1-40) test are mainly the possibility of false-positive and false-negative test results.
False-positive results, in conjunction with other clinical information, could lead to an inappropriate diagnosis of, and unnecessary treatment for AD.
False-negative test results could result in additional unnecessary diagnostic tests and potential delay in effective treatment for AD.
The FDA reviewed the device through the De Novo premarket review pathway, a regulatory pathway for low- to moderate-risk devices of a new type.
The agency says this action “creates a new regulatory classification, which means that subsequent devices of the same type with the same intended use may go through FDA’s 510(k) premarket process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a predicate device.”
The Lumipulse G β-amyloid Ratio (1-42/1-40) was granted Breakthrough Device designation, a process designed to expedite the development and review of devices that may provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
A version of this article first appeared on Medscape.com.
The Lumipulse G β-Amyloid Ratio 1-42/1-40 (Fujirebio Diagnostics) test detects amyloid plaques associated with AD in adults age 55 or older who are under investigation for AD and other causes of cognitive decline.
“The availability of an in vitro diagnostic test that can potentially eliminate the need for time-consuming and expensive [positron emission tomography (PET)] scans is great news for individuals and families concerned with the possibility of an Alzheimer’s disease diagnosis,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“With the Lumipulse test, there is a new option that can typically be completed the same day and can give doctors the same information regarding brain amyloid status, without the radiation risk, to help determine if a patient’s cognitive impairment is due to Alzheimer’s disease,” he added.
In its statement, the FDA notes that there is an “unmet need for a reliable and safe test that can accurately identify patients with amyloid plaques consistent with Alzheimer’s disease.”
The agency goes on to state that this new test may eliminate the need to use PET brain scans, a “potentially costly and cumbersome option” to visualize amyloid plaques for the diagnosis of AD.
The Lumipulse test measures the ratio of β-amyloid 1-42 and β-amyloid 1-40 concentrations in human cerebral spinal fluid (CSF). A positive Lumipulse G β-amyloid Ratio (1-42/1-40) test result is consistent with the presence of amyloid plaques, similar to that revealed in a PET scan. A negative result is consistent with a negative amyloid PET scan result.
However, the FDA notes that the test is not a stand-alone assay and should be used in conjunction with other clinical evaluations and additional tests to determine treatment options.
The FDA reports that it evaluated the safety and efficacy of the test in a clinical study of 292 CSF samples from the Alzheimer’s Disease Neuroimaging Initiative sample bank.
The samples were tested by the Lumipulse G β-amyloid Ratio (1-42/1-40) and compared with amyloid PET scan results. In this clinical study, 97% of individuals with Lumipulse G β-amyloid Ratio (1-42/1-40) positive results had the presence of amyloid plaques by PET scan and 84% of individuals with negative results had a negative amyloid PET scan.
The risks associated with the Lumipulse G β-amyloid Ratio (1-42/1-40) test are mainly the possibility of false-positive and false-negative test results.
False-positive results, in conjunction with other clinical information, could lead to an inappropriate diagnosis of, and unnecessary treatment for AD.
False-negative test results could result in additional unnecessary diagnostic tests and potential delay in effective treatment for AD.
The FDA reviewed the device through the De Novo premarket review pathway, a regulatory pathway for low- to moderate-risk devices of a new type.
The agency says this action “creates a new regulatory classification, which means that subsequent devices of the same type with the same intended use may go through FDA’s 510(k) premarket process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a predicate device.”
The Lumipulse G β-amyloid Ratio (1-42/1-40) was granted Breakthrough Device designation, a process designed to expedite the development and review of devices that may provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
A version of this article first appeared on Medscape.com.
Depression biomarkers: Which ones matter most?
Multiple biomarkers of depression involved in several brain circuits are altered in patients with unipolar depression.
because they suggest neuroimmunological alterations, disturbances in the blood-brain-barrier, hyperactivity in the hypothalamic-pituitary-adrenal (HPA) axis, and impaired neuroplasticity as factors in depression pathophysiology.
However, said study investigator Michael E. Benros, MD, PhD, professor and head of research at Mental Health Centre Copenhagen and University of Copenhagen, this is on a group level. “So in order to be relevant in a clinical context, the results need to be validated by further high-quality studies identifying subgroups with different biological underpinnings,” he told this news organization.
Identification of potential subgroups of depression with different biomarkers might help explain the diverse symptomatology and variability in treatment response observed in patients with depression, he noted.
The study was published online in JAMA Psychiatry.
Multiple pathways to depression
The systematic review and meta-analysis included 97 studies investigating 165 CSF biomarkers.
Of the 42 biomarkers investigated in at least two studies, patients with unipolar depression had higher CSF levels of interleukin 6, a marker of chronic inflammation; total protein, which signals blood-brain barrier dysfunction and increased permeability; and cortisol, which is linked to psychological stress, compared with healthy controls.
Depression was also associated with:
- Lower CSF levels of homovanillic acid, the major terminal metabolite of dopamine.
- Gamma-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the CNS thought to play a vital role in the control of stress and depression.
- Somatostatin, a neuropeptide often coexpressed with GABA.
- Brain-derived neurotrophic factor (BDNF), a protein involved in neurogenesis, synaptic plasticity, and neurotransmission.
- Amyloid-β 40, implicated in Alzheimer’s disease.
- Transthyretin, involved in transport of thyroxine across the blood-brain barrier.
Collectively, the findings point toward a “dysregulated dopaminergic system, a compromised inhibitory system, HPA axis hyperactivity, increased neuroinflammation and blood-brain barrier permeability, and impaired neuroplasticity as important factors in depression pathophysiology,” the investigators wrote.
“It is notable that we did not find significant difference in the metabolite levels of serotonin and noradrenalin, which are the most targeted neurotransmitters in modern antidepressant treatment,” said Dr. Benros.
However, this could be explained by substantial heterogeneity between studies and the fact that quantification of total CSF biomarker concentrations does not reflect local alteration within the brain, he explained.
Many of the studies had small cohorts and most quantified only a few biomarkers, making it hard to examine potential interactions between biomarkers or identify specific phenotypes of depression.
“Novel high-quality studies including larger cohorts with an integrative approach and extensive numbers of biomarkers are needed to validate these potential biomarkers of depression and set the stage for the development of more effective and precise treatments,” the researchers noted.
Which ones hold water?
Reached for comment, Dean MacKinnon, MD, associate professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, noted that this analysis “extracts the vast amount of knowledge” gained from different studies on biomarkers in the CSF for depression.
“They were able to identify 97 papers that have enough information in them that they could sort of lump them together and see which ones still hold water. It’s always useful to be able to look at patterns in the research and see if you can find some consistent trends,” he told this news organization.
Dr. MacKinnon, who was not part of the research team, also noted that “nonreplicability” is a problem in psychiatry and psychology research, “so being able to show that at least some studies were sufficiently well done, to get a good result, and that they could be replicated in at least one other good study is useful information.”
When it comes to depression, Dr. MacKinnon said, “We just don’t know enough to really pin down a physiologic pathway to explain it. The fact that some people seem to have high cortisol and some people seem to have high permeability of blood-brain barrier, and others have abnormalities in dopamine, is interesting and suggests that depression is likely not a unitary disease with a single cause.”
He cautioned, however, that the findings don’t have immediate clinical implications for individual patients with depression.
“Theoretically, down the road, if you extrapolate from what they found, and if it’s truly the case that this research maps to something that could suggest a different clinical approach, you might be able to determine whether one patient might respond better to an SSRI or an SNRI or something like that,” Dr. MacKinnon said.
Dr. Benros reported grants from Lundbeck Foundation during the conduct of the study. Dr. MacKinnon has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Multiple biomarkers of depression involved in several brain circuits are altered in patients with unipolar depression.
because they suggest neuroimmunological alterations, disturbances in the blood-brain-barrier, hyperactivity in the hypothalamic-pituitary-adrenal (HPA) axis, and impaired neuroplasticity as factors in depression pathophysiology.
However, said study investigator Michael E. Benros, MD, PhD, professor and head of research at Mental Health Centre Copenhagen and University of Copenhagen, this is on a group level. “So in order to be relevant in a clinical context, the results need to be validated by further high-quality studies identifying subgroups with different biological underpinnings,” he told this news organization.
Identification of potential subgroups of depression with different biomarkers might help explain the diverse symptomatology and variability in treatment response observed in patients with depression, he noted.
The study was published online in JAMA Psychiatry.
Multiple pathways to depression
The systematic review and meta-analysis included 97 studies investigating 165 CSF biomarkers.
Of the 42 biomarkers investigated in at least two studies, patients with unipolar depression had higher CSF levels of interleukin 6, a marker of chronic inflammation; total protein, which signals blood-brain barrier dysfunction and increased permeability; and cortisol, which is linked to psychological stress, compared with healthy controls.
Depression was also associated with:
- Lower CSF levels of homovanillic acid, the major terminal metabolite of dopamine.
- Gamma-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the CNS thought to play a vital role in the control of stress and depression.
- Somatostatin, a neuropeptide often coexpressed with GABA.
- Brain-derived neurotrophic factor (BDNF), a protein involved in neurogenesis, synaptic plasticity, and neurotransmission.
- Amyloid-β 40, implicated in Alzheimer’s disease.
- Transthyretin, involved in transport of thyroxine across the blood-brain barrier.
Collectively, the findings point toward a “dysregulated dopaminergic system, a compromised inhibitory system, HPA axis hyperactivity, increased neuroinflammation and blood-brain barrier permeability, and impaired neuroplasticity as important factors in depression pathophysiology,” the investigators wrote.
“It is notable that we did not find significant difference in the metabolite levels of serotonin and noradrenalin, which are the most targeted neurotransmitters in modern antidepressant treatment,” said Dr. Benros.
However, this could be explained by substantial heterogeneity between studies and the fact that quantification of total CSF biomarker concentrations does not reflect local alteration within the brain, he explained.
Many of the studies had small cohorts and most quantified only a few biomarkers, making it hard to examine potential interactions between biomarkers or identify specific phenotypes of depression.
“Novel high-quality studies including larger cohorts with an integrative approach and extensive numbers of biomarkers are needed to validate these potential biomarkers of depression and set the stage for the development of more effective and precise treatments,” the researchers noted.
Which ones hold water?
Reached for comment, Dean MacKinnon, MD, associate professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, noted that this analysis “extracts the vast amount of knowledge” gained from different studies on biomarkers in the CSF for depression.
“They were able to identify 97 papers that have enough information in them that they could sort of lump them together and see which ones still hold water. It’s always useful to be able to look at patterns in the research and see if you can find some consistent trends,” he told this news organization.
Dr. MacKinnon, who was not part of the research team, also noted that “nonreplicability” is a problem in psychiatry and psychology research, “so being able to show that at least some studies were sufficiently well done, to get a good result, and that they could be replicated in at least one other good study is useful information.”
When it comes to depression, Dr. MacKinnon said, “We just don’t know enough to really pin down a physiologic pathway to explain it. The fact that some people seem to have high cortisol and some people seem to have high permeability of blood-brain barrier, and others have abnormalities in dopamine, is interesting and suggests that depression is likely not a unitary disease with a single cause.”
He cautioned, however, that the findings don’t have immediate clinical implications for individual patients with depression.
“Theoretically, down the road, if you extrapolate from what they found, and if it’s truly the case that this research maps to something that could suggest a different clinical approach, you might be able to determine whether one patient might respond better to an SSRI or an SNRI or something like that,” Dr. MacKinnon said.
Dr. Benros reported grants from Lundbeck Foundation during the conduct of the study. Dr. MacKinnon has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Multiple biomarkers of depression involved in several brain circuits are altered in patients with unipolar depression.
because they suggest neuroimmunological alterations, disturbances in the blood-brain-barrier, hyperactivity in the hypothalamic-pituitary-adrenal (HPA) axis, and impaired neuroplasticity as factors in depression pathophysiology.
However, said study investigator Michael E. Benros, MD, PhD, professor and head of research at Mental Health Centre Copenhagen and University of Copenhagen, this is on a group level. “So in order to be relevant in a clinical context, the results need to be validated by further high-quality studies identifying subgroups with different biological underpinnings,” he told this news organization.
Identification of potential subgroups of depression with different biomarkers might help explain the diverse symptomatology and variability in treatment response observed in patients with depression, he noted.
The study was published online in JAMA Psychiatry.
Multiple pathways to depression
The systematic review and meta-analysis included 97 studies investigating 165 CSF biomarkers.
Of the 42 biomarkers investigated in at least two studies, patients with unipolar depression had higher CSF levels of interleukin 6, a marker of chronic inflammation; total protein, which signals blood-brain barrier dysfunction and increased permeability; and cortisol, which is linked to psychological stress, compared with healthy controls.
Depression was also associated with:
- Lower CSF levels of homovanillic acid, the major terminal metabolite of dopamine.
- Gamma-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the CNS thought to play a vital role in the control of stress and depression.
- Somatostatin, a neuropeptide often coexpressed with GABA.
- Brain-derived neurotrophic factor (BDNF), a protein involved in neurogenesis, synaptic plasticity, and neurotransmission.
- Amyloid-β 40, implicated in Alzheimer’s disease.
- Transthyretin, involved in transport of thyroxine across the blood-brain barrier.
Collectively, the findings point toward a “dysregulated dopaminergic system, a compromised inhibitory system, HPA axis hyperactivity, increased neuroinflammation and blood-brain barrier permeability, and impaired neuroplasticity as important factors in depression pathophysiology,” the investigators wrote.
“It is notable that we did not find significant difference in the metabolite levels of serotonin and noradrenalin, which are the most targeted neurotransmitters in modern antidepressant treatment,” said Dr. Benros.
However, this could be explained by substantial heterogeneity between studies and the fact that quantification of total CSF biomarker concentrations does not reflect local alteration within the brain, he explained.
Many of the studies had small cohorts and most quantified only a few biomarkers, making it hard to examine potential interactions between biomarkers or identify specific phenotypes of depression.
“Novel high-quality studies including larger cohorts with an integrative approach and extensive numbers of biomarkers are needed to validate these potential biomarkers of depression and set the stage for the development of more effective and precise treatments,” the researchers noted.
Which ones hold water?
Reached for comment, Dean MacKinnon, MD, associate professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, noted that this analysis “extracts the vast amount of knowledge” gained from different studies on biomarkers in the CSF for depression.
“They were able to identify 97 papers that have enough information in them that they could sort of lump them together and see which ones still hold water. It’s always useful to be able to look at patterns in the research and see if you can find some consistent trends,” he told this news organization.
Dr. MacKinnon, who was not part of the research team, also noted that “nonreplicability” is a problem in psychiatry and psychology research, “so being able to show that at least some studies were sufficiently well done, to get a good result, and that they could be replicated in at least one other good study is useful information.”
When it comes to depression, Dr. MacKinnon said, “We just don’t know enough to really pin down a physiologic pathway to explain it. The fact that some people seem to have high cortisol and some people seem to have high permeability of blood-brain barrier, and others have abnormalities in dopamine, is interesting and suggests that depression is likely not a unitary disease with a single cause.”
He cautioned, however, that the findings don’t have immediate clinical implications for individual patients with depression.
“Theoretically, down the road, if you extrapolate from what they found, and if it’s truly the case that this research maps to something that could suggest a different clinical approach, you might be able to determine whether one patient might respond better to an SSRI or an SNRI or something like that,” Dr. MacKinnon said.
Dr. Benros reported grants from Lundbeck Foundation during the conduct of the study. Dr. MacKinnon has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Seven hours of sleep is ideal for middle aged and older
Sleep disturbances are common in older age, and previous studies have shown associations between too much or too little sleep and increased risk of cognitive decline, but the ideal amount of sleep for preserving mental health has not been well described, according to the authors of the new paper.
In the study published in Nature Aging, the team of researchers from China and the United Kingdom reviewed data from the UK Biobank, a national database of individuals in the United Kingdom that includes cognitive assessments, mental health questionnaires, and brain imaging data, as well as genetic information.
Sleep is important for physical and psychological health, and also serves a neuroprotective function by clearing waste products from the brain, lead author Yuzhu Li of Fudan University, Shanghai, China, and colleagues wrote.
The study population included 498,277 participants, aged 38-73 years, who completed touchscreen questionnaires about sleep duration between 2006 and 2010. The average age at baseline was 56.5 years, 54% were female, and the mean sleep duration was 7.15 hours.
The researchers also reviewed brain imaging data and genetic data from 39,692 participants in 2014 to examine the relationships between sleep duration and brain structure and between sleep duration and genetic risk. In addition, 156,884 participants completed an online follow-up mental health questionnaire in 2016-2017 to assess the longitudinal impact of sleep on mental health.
Both excessive and insufficient sleep was associated with impaired cognitive performance, evidenced by the U-shaped curve found by the researchers in their data analysis, which used quadratic associations.
Specific cognitive functions including pair matching, trail making, prospective memory, and reaction time were significantly impaired with too much or too little sleep, the researchers said. “This demonstrated the positive association of both insufficient and excessive sleep duration with inferior performance on cognitive tasks.”
When the researchers analyzed the association between sleep duration and mental health, sleep duration also showed a U-shaped association with symptoms of anxiety, depression, mental distress, mania, and self-harm, while well-being showed an inverted U-shape. All associations between sleep duration and mental health were statistically significant after controlling for confounding variables (P < .001).
On further analysis (using two-line tests), the researchers determined that consistent sleep duration of approximately 7 hours per night was optimal for cognitive performance and for good mental health.
The researchers also used neuroimaging data to examine the relationship between sleep duration and brain structure. Overall, greater changes were seen in the regions of the brain involved in cognitive processing and memory.
“The most significant cortical volumes nonlinearly associated with sleep duration included the precentral cortex, the superior frontal gyrus, the lateral orbitofrontal cortex, the pars orbitalis, the frontal pole, and the middle temporal cortex,” the researchers wrote (P < .05 for all).
The association between sleep duration and cognitive function diminished among individuals older than 65 years, compared with those aged approximately 40 years, which suggests that optimal sleep duration may be more beneficial in middle age, the researchers noted. However, no similar impact of age was seen for mental health. For brain structure, the nonlinear relationship between sleep duration and cortical volumes was greatest in those aged 44-59 years, and gradually flattened with older age.
Research supports sleep discussions with patients
“Primary care physicians can use this study in their discussions with middle-aged and older patients to recommend optimal sleep duration and measures to achieve this sleep target,” Noel Deep, MD, a general internist in group practice in Antigo, Wisc., who was not involved in the study, said in an interview.
“This study is important because it demonstrated that both inadequate and excessive sleep patterns were associated with cognitive and mental health changes,” said Dr. Deep. “It supported previous observations of cognitive decline and mental health disorders being linked to disturbed sleep. But this study was unique because it provides data supporting an optimal sleep duration of 7 hours and the ill effects of both insufficient and excessive sleep duration.
“The usual thought process has been to assume that older individuals may not require as much sleep as the younger individuals, but this study supports an optimal time duration of sleep of 7 hours that benefits the older individuals. It was also interesting to note the mental health effects caused by the inadequate and excessive sleep durations,” he added.
As for additional research, “I would like to look into the quality of the sleep, in addition to the duration of sleep,” said Dr. Deep. For example, whether the excessive sleep was caused by poor quality sleep or fragmented sleep leading to the structural and subsequent cognitive decline.
Study limitations
“The current study relied on self-reporting of the sleep duration and was not observed and recorded data,” Dr. Deep noted. “It would also be beneficial to not only rely on healthy volunteers reporting the sleep duration, but also obtain sleep data from individuals with known brain disorders.”
The study findings were limited by several other factors, including the use of total sleep duration only, without other measures of sleep hygiene, the researchers noted. More research is needed to investigate the mechanisms driving the association between too much and not enough sleep and poor mental health and cognitive function.
The study was supported by the National Key R&D Program of China, the Shanghai Municipal Science and Technology Major Project, the Shanghai Center for Brain Science and Brain-Inspired Technology, the 111 Project, the National Natural Sciences Foundation of China and the Shanghai Rising Star Program.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
Sleep disturbances are common in older age, and previous studies have shown associations between too much or too little sleep and increased risk of cognitive decline, but the ideal amount of sleep for preserving mental health has not been well described, according to the authors of the new paper.
In the study published in Nature Aging, the team of researchers from China and the United Kingdom reviewed data from the UK Biobank, a national database of individuals in the United Kingdom that includes cognitive assessments, mental health questionnaires, and brain imaging data, as well as genetic information.
Sleep is important for physical and psychological health, and also serves a neuroprotective function by clearing waste products from the brain, lead author Yuzhu Li of Fudan University, Shanghai, China, and colleagues wrote.
The study population included 498,277 participants, aged 38-73 years, who completed touchscreen questionnaires about sleep duration between 2006 and 2010. The average age at baseline was 56.5 years, 54% were female, and the mean sleep duration was 7.15 hours.
The researchers also reviewed brain imaging data and genetic data from 39,692 participants in 2014 to examine the relationships between sleep duration and brain structure and between sleep duration and genetic risk. In addition, 156,884 participants completed an online follow-up mental health questionnaire in 2016-2017 to assess the longitudinal impact of sleep on mental health.
Both excessive and insufficient sleep was associated with impaired cognitive performance, evidenced by the U-shaped curve found by the researchers in their data analysis, which used quadratic associations.
Specific cognitive functions including pair matching, trail making, prospective memory, and reaction time were significantly impaired with too much or too little sleep, the researchers said. “This demonstrated the positive association of both insufficient and excessive sleep duration with inferior performance on cognitive tasks.”
When the researchers analyzed the association between sleep duration and mental health, sleep duration also showed a U-shaped association with symptoms of anxiety, depression, mental distress, mania, and self-harm, while well-being showed an inverted U-shape. All associations between sleep duration and mental health were statistically significant after controlling for confounding variables (P < .001).
On further analysis (using two-line tests), the researchers determined that consistent sleep duration of approximately 7 hours per night was optimal for cognitive performance and for good mental health.
The researchers also used neuroimaging data to examine the relationship between sleep duration and brain structure. Overall, greater changes were seen in the regions of the brain involved in cognitive processing and memory.
“The most significant cortical volumes nonlinearly associated with sleep duration included the precentral cortex, the superior frontal gyrus, the lateral orbitofrontal cortex, the pars orbitalis, the frontal pole, and the middle temporal cortex,” the researchers wrote (P < .05 for all).
The association between sleep duration and cognitive function diminished among individuals older than 65 years, compared with those aged approximately 40 years, which suggests that optimal sleep duration may be more beneficial in middle age, the researchers noted. However, no similar impact of age was seen for mental health. For brain structure, the nonlinear relationship between sleep duration and cortical volumes was greatest in those aged 44-59 years, and gradually flattened with older age.
Research supports sleep discussions with patients
“Primary care physicians can use this study in their discussions with middle-aged and older patients to recommend optimal sleep duration and measures to achieve this sleep target,” Noel Deep, MD, a general internist in group practice in Antigo, Wisc., who was not involved in the study, said in an interview.
“This study is important because it demonstrated that both inadequate and excessive sleep patterns were associated with cognitive and mental health changes,” said Dr. Deep. “It supported previous observations of cognitive decline and mental health disorders being linked to disturbed sleep. But this study was unique because it provides data supporting an optimal sleep duration of 7 hours and the ill effects of both insufficient and excessive sleep duration.
“The usual thought process has been to assume that older individuals may not require as much sleep as the younger individuals, but this study supports an optimal time duration of sleep of 7 hours that benefits the older individuals. It was also interesting to note the mental health effects caused by the inadequate and excessive sleep durations,” he added.
As for additional research, “I would like to look into the quality of the sleep, in addition to the duration of sleep,” said Dr. Deep. For example, whether the excessive sleep was caused by poor quality sleep or fragmented sleep leading to the structural and subsequent cognitive decline.
Study limitations
“The current study relied on self-reporting of the sleep duration and was not observed and recorded data,” Dr. Deep noted. “It would also be beneficial to not only rely on healthy volunteers reporting the sleep duration, but also obtain sleep data from individuals with known brain disorders.”
The study findings were limited by several other factors, including the use of total sleep duration only, without other measures of sleep hygiene, the researchers noted. More research is needed to investigate the mechanisms driving the association between too much and not enough sleep and poor mental health and cognitive function.
The study was supported by the National Key R&D Program of China, the Shanghai Municipal Science and Technology Major Project, the Shanghai Center for Brain Science and Brain-Inspired Technology, the 111 Project, the National Natural Sciences Foundation of China and the Shanghai Rising Star Program.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
Sleep disturbances are common in older age, and previous studies have shown associations between too much or too little sleep and increased risk of cognitive decline, but the ideal amount of sleep for preserving mental health has not been well described, according to the authors of the new paper.
In the study published in Nature Aging, the team of researchers from China and the United Kingdom reviewed data from the UK Biobank, a national database of individuals in the United Kingdom that includes cognitive assessments, mental health questionnaires, and brain imaging data, as well as genetic information.
Sleep is important for physical and psychological health, and also serves a neuroprotective function by clearing waste products from the brain, lead author Yuzhu Li of Fudan University, Shanghai, China, and colleagues wrote.
The study population included 498,277 participants, aged 38-73 years, who completed touchscreen questionnaires about sleep duration between 2006 and 2010. The average age at baseline was 56.5 years, 54% were female, and the mean sleep duration was 7.15 hours.
The researchers also reviewed brain imaging data and genetic data from 39,692 participants in 2014 to examine the relationships between sleep duration and brain structure and between sleep duration and genetic risk. In addition, 156,884 participants completed an online follow-up mental health questionnaire in 2016-2017 to assess the longitudinal impact of sleep on mental health.
Both excessive and insufficient sleep was associated with impaired cognitive performance, evidenced by the U-shaped curve found by the researchers in their data analysis, which used quadratic associations.
Specific cognitive functions including pair matching, trail making, prospective memory, and reaction time were significantly impaired with too much or too little sleep, the researchers said. “This demonstrated the positive association of both insufficient and excessive sleep duration with inferior performance on cognitive tasks.”
When the researchers analyzed the association between sleep duration and mental health, sleep duration also showed a U-shaped association with symptoms of anxiety, depression, mental distress, mania, and self-harm, while well-being showed an inverted U-shape. All associations between sleep duration and mental health were statistically significant after controlling for confounding variables (P < .001).
On further analysis (using two-line tests), the researchers determined that consistent sleep duration of approximately 7 hours per night was optimal for cognitive performance and for good mental health.
The researchers also used neuroimaging data to examine the relationship between sleep duration and brain structure. Overall, greater changes were seen in the regions of the brain involved in cognitive processing and memory.
“The most significant cortical volumes nonlinearly associated with sleep duration included the precentral cortex, the superior frontal gyrus, the lateral orbitofrontal cortex, the pars orbitalis, the frontal pole, and the middle temporal cortex,” the researchers wrote (P < .05 for all).
The association between sleep duration and cognitive function diminished among individuals older than 65 years, compared with those aged approximately 40 years, which suggests that optimal sleep duration may be more beneficial in middle age, the researchers noted. However, no similar impact of age was seen for mental health. For brain structure, the nonlinear relationship between sleep duration and cortical volumes was greatest in those aged 44-59 years, and gradually flattened with older age.
Research supports sleep discussions with patients
“Primary care physicians can use this study in their discussions with middle-aged and older patients to recommend optimal sleep duration and measures to achieve this sleep target,” Noel Deep, MD, a general internist in group practice in Antigo, Wisc., who was not involved in the study, said in an interview.
“This study is important because it demonstrated that both inadequate and excessive sleep patterns were associated with cognitive and mental health changes,” said Dr. Deep. “It supported previous observations of cognitive decline and mental health disorders being linked to disturbed sleep. But this study was unique because it provides data supporting an optimal sleep duration of 7 hours and the ill effects of both insufficient and excessive sleep duration.
“The usual thought process has been to assume that older individuals may not require as much sleep as the younger individuals, but this study supports an optimal time duration of sleep of 7 hours that benefits the older individuals. It was also interesting to note the mental health effects caused by the inadequate and excessive sleep durations,” he added.
As for additional research, “I would like to look into the quality of the sleep, in addition to the duration of sleep,” said Dr. Deep. For example, whether the excessive sleep was caused by poor quality sleep or fragmented sleep leading to the structural and subsequent cognitive decline.
Study limitations
“The current study relied on self-reporting of the sleep duration and was not observed and recorded data,” Dr. Deep noted. “It would also be beneficial to not only rely on healthy volunteers reporting the sleep duration, but also obtain sleep data from individuals with known brain disorders.”
The study findings were limited by several other factors, including the use of total sleep duration only, without other measures of sleep hygiene, the researchers noted. More research is needed to investigate the mechanisms driving the association between too much and not enough sleep and poor mental health and cognitive function.
The study was supported by the National Key R&D Program of China, the Shanghai Municipal Science and Technology Major Project, the Shanghai Center for Brain Science and Brain-Inspired Technology, the 111 Project, the National Natural Sciences Foundation of China and the Shanghai Rising Star Program.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
FROM NATURE AGING
Severe COVID-19 adds 20 years of cognitive aging: Study
adding that the impairment is “equivalent to losing 10 IQ points.”
In their study, published in eClinicalMedicine, a team of scientists from the University of Cambridge and Imperial College London said there is growing evidence that COVID-19 can cause lasting cognitive and mental health problems. Patients report fatigue, “brain fog,” problems recalling words, sleep disturbances, anxiety, and even posttraumatic stress disorder months after infection.
The researchers analyzed data from 46 individuals who received critical care for COVID-19 at Addenbrooke’s Hospital between March and July 2020 (27 females, 19 males, mean age 51 years, 16 of whom had mechanical ventilation) and were recruited to the NIHR COVID-19 BioResource project.
At an average of 6 months after acute COVID-19 illness, the study participants underwent detailed computerized cognitive tests via the Cognitron platform, comprising eight tasks deployed on an iPad measuring mental function such as memory, attention, and reasoning. Also assessed were anxiety, depression, and posttraumatic stress disorder via standard mood, anxiety, and posttraumatic stress scales – specifically the Generalized Anxiety Disorder 7 (GAD-7), the Patient Health Questionnaire 9 (PHQ-9), and the PTSD Checklist for Diagnostic and Statistical Manual of Mental Disorders 5 (PCL-5). Their data were compared against 460 controls – matched for age, sex, education, and first language – and the pattern of deficits across tasks was qualitatively compared with normal age-related decline and early-stage dementia.
Less accurate and slower response times
The authors highlighted how this was the first time a “rigorous assessment and comparison” had been carried out in relation to the after-effects of severe COVID-19.
“Cognitive impairment is common to a wide range of neurological disorders, including dementia, and even routine aging, but the patterns we saw – the cognitive ‘fingerprint’ of COVID-19 – was distinct from all of these,” said David Menon, MD, division of anesthesia at the University of Cambridge, England, and the study’s senior author.
The scientists found that COVID-19 survivors were less accurate and had slower response times than the control population, and added that survivors scored particularly poorly on verbal analogical reasoning and showed slower processing speeds.
Critically, the scale of the cognitive deficits correlated with acute illness severity, but not fatigue or mental health status at the time of cognitive assessment, said the authors.
Recovery ‘at best gradual’
The effects were strongest for those with more severe acute illness, and who required mechanical ventilation, said the authors, who found that acute illness severity was “better at predicting the cognitive deficits.”
The authors pointed out how these deficits were still detectable when patients were followed up 6 months later, and that, although patients’ scores and reaction times began to improve over time, any recovery was “at best gradual” and likely to be influenced by factors such as illness severity and its neurological or psychological impacts.
“We followed some patients up as late as 10 months after their acute infection, so were able to see a very slow improvement,” Dr. Menon said. He explained how, while this improvement was not statistically significant, it was “at least heading in the right direction.”
However, he warned it is very possible that some of these individuals “will never fully recover.”
The cognitive deficits observed may be due to several factors in combination, said the authors, including inadequate oxygen or blood supply to the brain, blockage of large or small blood vessels due to clotting, and microscopic bleeds. They highlighted how the most important mechanism, however, may be “damage caused by the body’s own inflammatory response and immune system.”
Adam Hampshire, PhD, of the department of brain sciences at Imperial College London, one of the study’s authors, described how around 40,000 people have been through intensive care with COVID-19 in England alone, with many more despite having been very sick not admitted to hospital. This means there is a “large number of people out there still experiencing problems with cognition many months later,” he said. “We urgently need to look at what can be done to help these people.”
A version of this article first appeared on Univadis.
adding that the impairment is “equivalent to losing 10 IQ points.”
In their study, published in eClinicalMedicine, a team of scientists from the University of Cambridge and Imperial College London said there is growing evidence that COVID-19 can cause lasting cognitive and mental health problems. Patients report fatigue, “brain fog,” problems recalling words, sleep disturbances, anxiety, and even posttraumatic stress disorder months after infection.
The researchers analyzed data from 46 individuals who received critical care for COVID-19 at Addenbrooke’s Hospital between March and July 2020 (27 females, 19 males, mean age 51 years, 16 of whom had mechanical ventilation) and were recruited to the NIHR COVID-19 BioResource project.
At an average of 6 months after acute COVID-19 illness, the study participants underwent detailed computerized cognitive tests via the Cognitron platform, comprising eight tasks deployed on an iPad measuring mental function such as memory, attention, and reasoning. Also assessed were anxiety, depression, and posttraumatic stress disorder via standard mood, anxiety, and posttraumatic stress scales – specifically the Generalized Anxiety Disorder 7 (GAD-7), the Patient Health Questionnaire 9 (PHQ-9), and the PTSD Checklist for Diagnostic and Statistical Manual of Mental Disorders 5 (PCL-5). Their data were compared against 460 controls – matched for age, sex, education, and first language – and the pattern of deficits across tasks was qualitatively compared with normal age-related decline and early-stage dementia.
Less accurate and slower response times
The authors highlighted how this was the first time a “rigorous assessment and comparison” had been carried out in relation to the after-effects of severe COVID-19.
“Cognitive impairment is common to a wide range of neurological disorders, including dementia, and even routine aging, but the patterns we saw – the cognitive ‘fingerprint’ of COVID-19 – was distinct from all of these,” said David Menon, MD, division of anesthesia at the University of Cambridge, England, and the study’s senior author.
The scientists found that COVID-19 survivors were less accurate and had slower response times than the control population, and added that survivors scored particularly poorly on verbal analogical reasoning and showed slower processing speeds.
Critically, the scale of the cognitive deficits correlated with acute illness severity, but not fatigue or mental health status at the time of cognitive assessment, said the authors.
Recovery ‘at best gradual’
The effects were strongest for those with more severe acute illness, and who required mechanical ventilation, said the authors, who found that acute illness severity was “better at predicting the cognitive deficits.”
The authors pointed out how these deficits were still detectable when patients were followed up 6 months later, and that, although patients’ scores and reaction times began to improve over time, any recovery was “at best gradual” and likely to be influenced by factors such as illness severity and its neurological or psychological impacts.
“We followed some patients up as late as 10 months after their acute infection, so were able to see a very slow improvement,” Dr. Menon said. He explained how, while this improvement was not statistically significant, it was “at least heading in the right direction.”
However, he warned it is very possible that some of these individuals “will never fully recover.”
The cognitive deficits observed may be due to several factors in combination, said the authors, including inadequate oxygen or blood supply to the brain, blockage of large or small blood vessels due to clotting, and microscopic bleeds. They highlighted how the most important mechanism, however, may be “damage caused by the body’s own inflammatory response and immune system.”
Adam Hampshire, PhD, of the department of brain sciences at Imperial College London, one of the study’s authors, described how around 40,000 people have been through intensive care with COVID-19 in England alone, with many more despite having been very sick not admitted to hospital. This means there is a “large number of people out there still experiencing problems with cognition many months later,” he said. “We urgently need to look at what can be done to help these people.”
A version of this article first appeared on Univadis.
adding that the impairment is “equivalent to losing 10 IQ points.”
In their study, published in eClinicalMedicine, a team of scientists from the University of Cambridge and Imperial College London said there is growing evidence that COVID-19 can cause lasting cognitive and mental health problems. Patients report fatigue, “brain fog,” problems recalling words, sleep disturbances, anxiety, and even posttraumatic stress disorder months after infection.
The researchers analyzed data from 46 individuals who received critical care for COVID-19 at Addenbrooke’s Hospital between March and July 2020 (27 females, 19 males, mean age 51 years, 16 of whom had mechanical ventilation) and were recruited to the NIHR COVID-19 BioResource project.
At an average of 6 months after acute COVID-19 illness, the study participants underwent detailed computerized cognitive tests via the Cognitron platform, comprising eight tasks deployed on an iPad measuring mental function such as memory, attention, and reasoning. Also assessed were anxiety, depression, and posttraumatic stress disorder via standard mood, anxiety, and posttraumatic stress scales – specifically the Generalized Anxiety Disorder 7 (GAD-7), the Patient Health Questionnaire 9 (PHQ-9), and the PTSD Checklist for Diagnostic and Statistical Manual of Mental Disorders 5 (PCL-5). Their data were compared against 460 controls – matched for age, sex, education, and first language – and the pattern of deficits across tasks was qualitatively compared with normal age-related decline and early-stage dementia.
Less accurate and slower response times
The authors highlighted how this was the first time a “rigorous assessment and comparison” had been carried out in relation to the after-effects of severe COVID-19.
“Cognitive impairment is common to a wide range of neurological disorders, including dementia, and even routine aging, but the patterns we saw – the cognitive ‘fingerprint’ of COVID-19 – was distinct from all of these,” said David Menon, MD, division of anesthesia at the University of Cambridge, England, and the study’s senior author.
The scientists found that COVID-19 survivors were less accurate and had slower response times than the control population, and added that survivors scored particularly poorly on verbal analogical reasoning and showed slower processing speeds.
Critically, the scale of the cognitive deficits correlated with acute illness severity, but not fatigue or mental health status at the time of cognitive assessment, said the authors.
Recovery ‘at best gradual’
The effects were strongest for those with more severe acute illness, and who required mechanical ventilation, said the authors, who found that acute illness severity was “better at predicting the cognitive deficits.”
The authors pointed out how these deficits were still detectable when patients were followed up 6 months later, and that, although patients’ scores and reaction times began to improve over time, any recovery was “at best gradual” and likely to be influenced by factors such as illness severity and its neurological or psychological impacts.
“We followed some patients up as late as 10 months after their acute infection, so were able to see a very slow improvement,” Dr. Menon said. He explained how, while this improvement was not statistically significant, it was “at least heading in the right direction.”
However, he warned it is very possible that some of these individuals “will never fully recover.”
The cognitive deficits observed may be due to several factors in combination, said the authors, including inadequate oxygen or blood supply to the brain, blockage of large or small blood vessels due to clotting, and microscopic bleeds. They highlighted how the most important mechanism, however, may be “damage caused by the body’s own inflammatory response and immune system.”
Adam Hampshire, PhD, of the department of brain sciences at Imperial College London, one of the study’s authors, described how around 40,000 people have been through intensive care with COVID-19 in England alone, with many more despite having been very sick not admitted to hospital. This means there is a “large number of people out there still experiencing problems with cognition many months later,” he said. “We urgently need to look at what can be done to help these people.”
A version of this article first appeared on Univadis.
FROM ECLINICAL MEDICINE
Cutting dementia risk in AFib: Does rhythm control strategy matter?
The risk for dementia goes up in patients with atrial fibrillation (AFib), but some evidence suggests that risk can be blunted with therapies that restore sinus rhythm. However, a new cohort study suggests that the treatment effect’s magnitude might depend on the rhythm control strategy. It hinted that AFib catheter ablation might be more effective than pharmacologic rhythm control alone at cutting the risk for dementia.
The case-matched study of more than 38,000 adults with AFib saw a 41% reduction (P < .0001) in risk for dementia among those who underwent catheter ablation after attempted rhythm control with antiarrhythmic drugs (AAD), compared with those managed with pharmacologic rhythm control therapy alone.
The observational study comprising 20 years of data comes with big limitations and can’t say for sure whether catheter ablation is better than AAD-only at cutting the dementia risk in AFib. But it and other evidence support the idea, which has yet to be explored in a randomized fashion.
In a secondary finding, the analysis showed a similar reduction in dementia risk from catheter ablation, compared with AAD, in women and in men by 40% and 45%, respectively (P < .0001 for both). The findings are particularly relevant “given the higher life-long risk of dementia among women and the lower likelihood that women will be offered ablation, which has been demonstrated repeatedly,” Emily P. Zeitler, MD, MHS, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire, told this news organization. “I think this is another reason to try to be more generous in offering ablation to women.”
Management of AFib certainly evolved in important ways from 2000 to 2021, the period covered by the study. But a sensitivity analysis based on data from 2010 to 2021 showed “no meaningful differences” in the results, said Dr. Zeitler, who is slated to present the findings April 30 at the Heart Rhythm Society 2022 Scientific Sessions, conducted virtually and live in San Francisco.
Dr. Zeitler acknowledged that the observational study, even with its propensity-matched ablation and AAD cohorts, can only hint at a preference for ablation over AAD for lowering risk for AFib-associated dementia. “We know there’s unmeasured and unfixable confounding between those two groups, so we see this really as hypothesis-generating.”
It was “a well-done analysis,” and the conclusion that the dementia risk was lower with catheter ablation is “absolutely correct,” but only as far as the study and its limitations allow, agreed David Conen, MD, MPH, McMaster University, Hamilton, Ontario, who is not a coauthor.
“Even with propensity matching, you can get rid of some sorts of confounding, but you can never get rid of all selection bias issues.” That, he said when interviewed, takes randomized trials.
Dr. Conen, who is studying cognitive decline in AFib as a SWISS-AF trial principal investigator, pointed to a secondary finding of the analysis as evidence for such confounding. He said the ablation group’s nearly 50% drop (P < .0001) in competing risk for death, compared with patients managed with AAD, isn’t plausible.
The finding “strongly suggests these people were healthier and that there’s some sort of selection bias. They were at lower risk of death, they were at lower risk of dementia, and they were probably also at lower risk of stroke, myocardial infarction, thrombosis, and cancer because they were just probably a little healthier than the others,” Dr. Conen said. The ablation and AAD groups “were two very different populations from the get-go.”
The analysis was based on U.S. insurance and Medicare claims data from AFib patients who either underwent catheter ablation after at least one AAD trial or filled prescriptions for at least two different antiarrhythmic agents in the year after AFib diagnosis. Patients with history of dementia, catheter or surgical AFib ablation, or a valve procedure were excluded.
The ablation and AAD-only groups each consisted of 19,066 patients after propensity matching, and the groups were balanced with respect to age, sex, type of insurance, CHA2DS2-VASc scores, and use of renin-angiotensin-system inhibitors, oral anticoagulants, and antiplatelets.
The overall risk for dementia was 1.9% for the ablation group and 3.3% for AAD-only patients (hazard ratio, 0.59; 95% confidence interval, 0.52-0.67). Corresponding HRs by sex were 0.55 (95% CI, 0.46-0.66) for men and 0.60 (95% CI, 0.50-0.72) for women.
The competing risk for death was also significantly decreased in the ablation group (HR, 0.51; 95% CI, 0.46-0.55).
Dr. Zeitler pointed to a randomized trial now in the early stages called Neurocognition and Greater Maintenance of Sinus Rhythm in Atrial Fibrillation, or NOGGIN-AF, which will explore relationships between rhythm control therapy and dementia in patients with AFib, whether catheter ablation or AAD can mitigate that risk, and whether either strategy works better than the other, among other goals.
“I’m optimistic,” she said, “and I think it’s going to add to the growing motivations to get patients ablated more quickly and more broadly.”
The analysis was funded by Biosense-Webster. Dr. Zeitler discloses consulting for Biosense-Webster and Arena Pharmaceuticals (now Pfizer); fees for speaking from Medtronic; and receiving research support from Boston Scientific, Sanofi, and Biosense-Webster. Dr. Conen has previously reported receiving speaker fees from Servier Canada.
A version of this article first appeared on Medscape.com.
The risk for dementia goes up in patients with atrial fibrillation (AFib), but some evidence suggests that risk can be blunted with therapies that restore sinus rhythm. However, a new cohort study suggests that the treatment effect’s magnitude might depend on the rhythm control strategy. It hinted that AFib catheter ablation might be more effective than pharmacologic rhythm control alone at cutting the risk for dementia.
The case-matched study of more than 38,000 adults with AFib saw a 41% reduction (P < .0001) in risk for dementia among those who underwent catheter ablation after attempted rhythm control with antiarrhythmic drugs (AAD), compared with those managed with pharmacologic rhythm control therapy alone.
The observational study comprising 20 years of data comes with big limitations and can’t say for sure whether catheter ablation is better than AAD-only at cutting the dementia risk in AFib. But it and other evidence support the idea, which has yet to be explored in a randomized fashion.
In a secondary finding, the analysis showed a similar reduction in dementia risk from catheter ablation, compared with AAD, in women and in men by 40% and 45%, respectively (P < .0001 for both). The findings are particularly relevant “given the higher life-long risk of dementia among women and the lower likelihood that women will be offered ablation, which has been demonstrated repeatedly,” Emily P. Zeitler, MD, MHS, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire, told this news organization. “I think this is another reason to try to be more generous in offering ablation to women.”
Management of AFib certainly evolved in important ways from 2000 to 2021, the period covered by the study. But a sensitivity analysis based on data from 2010 to 2021 showed “no meaningful differences” in the results, said Dr. Zeitler, who is slated to present the findings April 30 at the Heart Rhythm Society 2022 Scientific Sessions, conducted virtually and live in San Francisco.
Dr. Zeitler acknowledged that the observational study, even with its propensity-matched ablation and AAD cohorts, can only hint at a preference for ablation over AAD for lowering risk for AFib-associated dementia. “We know there’s unmeasured and unfixable confounding between those two groups, so we see this really as hypothesis-generating.”
It was “a well-done analysis,” and the conclusion that the dementia risk was lower with catheter ablation is “absolutely correct,” but only as far as the study and its limitations allow, agreed David Conen, MD, MPH, McMaster University, Hamilton, Ontario, who is not a coauthor.
“Even with propensity matching, you can get rid of some sorts of confounding, but you can never get rid of all selection bias issues.” That, he said when interviewed, takes randomized trials.
Dr. Conen, who is studying cognitive decline in AFib as a SWISS-AF trial principal investigator, pointed to a secondary finding of the analysis as evidence for such confounding. He said the ablation group’s nearly 50% drop (P < .0001) in competing risk for death, compared with patients managed with AAD, isn’t plausible.
The finding “strongly suggests these people were healthier and that there’s some sort of selection bias. They were at lower risk of death, they were at lower risk of dementia, and they were probably also at lower risk of stroke, myocardial infarction, thrombosis, and cancer because they were just probably a little healthier than the others,” Dr. Conen said. The ablation and AAD groups “were two very different populations from the get-go.”
The analysis was based on U.S. insurance and Medicare claims data from AFib patients who either underwent catheter ablation after at least one AAD trial or filled prescriptions for at least two different antiarrhythmic agents in the year after AFib diagnosis. Patients with history of dementia, catheter or surgical AFib ablation, or a valve procedure were excluded.
The ablation and AAD-only groups each consisted of 19,066 patients after propensity matching, and the groups were balanced with respect to age, sex, type of insurance, CHA2DS2-VASc scores, and use of renin-angiotensin-system inhibitors, oral anticoagulants, and antiplatelets.
The overall risk for dementia was 1.9% for the ablation group and 3.3% for AAD-only patients (hazard ratio, 0.59; 95% confidence interval, 0.52-0.67). Corresponding HRs by sex were 0.55 (95% CI, 0.46-0.66) for men and 0.60 (95% CI, 0.50-0.72) for women.
The competing risk for death was also significantly decreased in the ablation group (HR, 0.51; 95% CI, 0.46-0.55).
Dr. Zeitler pointed to a randomized trial now in the early stages called Neurocognition and Greater Maintenance of Sinus Rhythm in Atrial Fibrillation, or NOGGIN-AF, which will explore relationships between rhythm control therapy and dementia in patients with AFib, whether catheter ablation or AAD can mitigate that risk, and whether either strategy works better than the other, among other goals.
“I’m optimistic,” she said, “and I think it’s going to add to the growing motivations to get patients ablated more quickly and more broadly.”
The analysis was funded by Biosense-Webster. Dr. Zeitler discloses consulting for Biosense-Webster and Arena Pharmaceuticals (now Pfizer); fees for speaking from Medtronic; and receiving research support from Boston Scientific, Sanofi, and Biosense-Webster. Dr. Conen has previously reported receiving speaker fees from Servier Canada.
A version of this article first appeared on Medscape.com.
The risk for dementia goes up in patients with atrial fibrillation (AFib), but some evidence suggests that risk can be blunted with therapies that restore sinus rhythm. However, a new cohort study suggests that the treatment effect’s magnitude might depend on the rhythm control strategy. It hinted that AFib catheter ablation might be more effective than pharmacologic rhythm control alone at cutting the risk for dementia.
The case-matched study of more than 38,000 adults with AFib saw a 41% reduction (P < .0001) in risk for dementia among those who underwent catheter ablation after attempted rhythm control with antiarrhythmic drugs (AAD), compared with those managed with pharmacologic rhythm control therapy alone.
The observational study comprising 20 years of data comes with big limitations and can’t say for sure whether catheter ablation is better than AAD-only at cutting the dementia risk in AFib. But it and other evidence support the idea, which has yet to be explored in a randomized fashion.
In a secondary finding, the analysis showed a similar reduction in dementia risk from catheter ablation, compared with AAD, in women and in men by 40% and 45%, respectively (P < .0001 for both). The findings are particularly relevant “given the higher life-long risk of dementia among women and the lower likelihood that women will be offered ablation, which has been demonstrated repeatedly,” Emily P. Zeitler, MD, MHS, Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire, told this news organization. “I think this is another reason to try to be more generous in offering ablation to women.”
Management of AFib certainly evolved in important ways from 2000 to 2021, the period covered by the study. But a sensitivity analysis based on data from 2010 to 2021 showed “no meaningful differences” in the results, said Dr. Zeitler, who is slated to present the findings April 30 at the Heart Rhythm Society 2022 Scientific Sessions, conducted virtually and live in San Francisco.
Dr. Zeitler acknowledged that the observational study, even with its propensity-matched ablation and AAD cohorts, can only hint at a preference for ablation over AAD for lowering risk for AFib-associated dementia. “We know there’s unmeasured and unfixable confounding between those two groups, so we see this really as hypothesis-generating.”
It was “a well-done analysis,” and the conclusion that the dementia risk was lower with catheter ablation is “absolutely correct,” but only as far as the study and its limitations allow, agreed David Conen, MD, MPH, McMaster University, Hamilton, Ontario, who is not a coauthor.
“Even with propensity matching, you can get rid of some sorts of confounding, but you can never get rid of all selection bias issues.” That, he said when interviewed, takes randomized trials.
Dr. Conen, who is studying cognitive decline in AFib as a SWISS-AF trial principal investigator, pointed to a secondary finding of the analysis as evidence for such confounding. He said the ablation group’s nearly 50% drop (P < .0001) in competing risk for death, compared with patients managed with AAD, isn’t plausible.
The finding “strongly suggests these people were healthier and that there’s some sort of selection bias. They were at lower risk of death, they were at lower risk of dementia, and they were probably also at lower risk of stroke, myocardial infarction, thrombosis, and cancer because they were just probably a little healthier than the others,” Dr. Conen said. The ablation and AAD groups “were two very different populations from the get-go.”
The analysis was based on U.S. insurance and Medicare claims data from AFib patients who either underwent catheter ablation after at least one AAD trial or filled prescriptions for at least two different antiarrhythmic agents in the year after AFib diagnosis. Patients with history of dementia, catheter or surgical AFib ablation, or a valve procedure were excluded.
The ablation and AAD-only groups each consisted of 19,066 patients after propensity matching, and the groups were balanced with respect to age, sex, type of insurance, CHA2DS2-VASc scores, and use of renin-angiotensin-system inhibitors, oral anticoagulants, and antiplatelets.
The overall risk for dementia was 1.9% for the ablation group and 3.3% for AAD-only patients (hazard ratio, 0.59; 95% confidence interval, 0.52-0.67). Corresponding HRs by sex were 0.55 (95% CI, 0.46-0.66) for men and 0.60 (95% CI, 0.50-0.72) for women.
The competing risk for death was also significantly decreased in the ablation group (HR, 0.51; 95% CI, 0.46-0.55).
Dr. Zeitler pointed to a randomized trial now in the early stages called Neurocognition and Greater Maintenance of Sinus Rhythm in Atrial Fibrillation, or NOGGIN-AF, which will explore relationships between rhythm control therapy and dementia in patients with AFib, whether catheter ablation or AAD can mitigate that risk, and whether either strategy works better than the other, among other goals.
“I’m optimistic,” she said, “and I think it’s going to add to the growing motivations to get patients ablated more quickly and more broadly.”
The analysis was funded by Biosense-Webster. Dr. Zeitler discloses consulting for Biosense-Webster and Arena Pharmaceuticals (now Pfizer); fees for speaking from Medtronic; and receiving research support from Boston Scientific, Sanofi, and Biosense-Webster. Dr. Conen has previously reported receiving speaker fees from Servier Canada.
A version of this article first appeared on Medscape.com.