User login
ACR, AAD, AAO, RDS issue joint statement on safe use of hydroxychloroquine
Hydroxychloroquine can be used safely and effectively with attention to dosing, risk factors, and screening, but communication among physicians, patients, and eye care specialists is key to optimizing outcomes and preventing complications, according to a joint statement from four medical societies.
The American College of Rheumatology, American Academy of Dermatology, Rheumatologic Dermatology Society, and the American Academy of Ophthalmology have produced a statement, published in Arthritis & Rheumatology, “to emphasize points of agreement that should be recognized by practitioners in all specialties,” lead author James T. Rosenbaum, MD, of Oregon Health & Science University, Portland, and colleagues wrote.
The statement was developed by a working group that included rheumatologists, ophthalmologists, and dermatologists with records of published studies on the use of hydroxychloroquine (HCQ) and its toxicity. The statement updated elements of the 2016 American Academy of Ophthalmology guidelines for monitoring patients for retinal toxicity when using HCQ.
“The need for collaborative management has triggered this joint statement, which applies only to managing the risk of HCQ retinopathy and does not include consideration of cardiac, muscle, dermatologic, or other toxicities,” the authors noted.
The authors emphasized that HCQ plays a valuable role in controlling many rheumatic diseases, and should not be abandoned out of fear of retinopathy. However, proper dosing, recognition of risk factors, and screening strategies are essential.
Dosing data
Data on HCQ dosing and retinopathy are limited, but the authors cited a study of 2,361 rheumatic disease patients with an average HCQ dosing regimen of 5.0 mg/kg per day or less in which the toxicity risk was less than 2% for up to 10 years of use. Although data show some increase in risk with duration of use, “for a patient with a normal screening exam in a given year, the risk of developing retinopathy in the ensuing year is low (e.g., less than 5%), even after 20 years of use,” the authors said.
Risk factor recognition
“High daily [HCQ] dosage relative to body weight and cumulative dose are the primary risk factors for retinopathy,” the authors noted. Reduced renal function is an additional risk factor, and patients with renal insufficiency should be monitored and may need lower doses.
In addition, patients with a phenotype of initial parafoveal toxicity may be at increased risk for advanced disease evidenced by damage to the foveal center. “The phenotype of initial parafoveal toxicity is not universal, and in many patients (East Asians particularly) the retinal changes may appear initially along the pericentral vascular arcades,” so these patients should be screened with additional tests beyond the central macula, they emphasized.
Screening strategies
Patients should receive a baseline retinal exam within a few months of starting HCQ to rule out underlying retinal disease, according to the statement. The goal of screening is “to detect early retinopathy before a bullseye becomes visible on ophthalmoscopy, since at that severe stage the damage tends to progress even after discontinuing the medication and may eventually threaten central vision,” the authors said.
In the absence of risk factors, patients can defer screening for 5 years, but should be screened annually from 5 years and forward, they said. Examples of underlying retinal disease include “significant macular degeneration, severe diabetic retinopathy, or hereditary disorders of retinal function, but these are judgments best made by the ophthalmologist since mild and stable abnormalities that do not interfere with interpretation of critical diagnostic tests may not be a contraindication” to use of HCQ.
The consensus opinion statement has limitations, notably the shortage of data on optimum HCQ dosage and the lack of prospective studies of toxicity, including the need for studies of the impact of blood levels on toxicity and studies of pharmacogenomics to stratify risk, the authors noted.
“It is important that the drug is not stopped prematurely, but also that it is not continued in the face of definitive evidence of retinal toxicity except in some situations with unusual medical need,” they said.
“Suggestive or uncertain findings should be discussed with the patient and prescribing physician to justify further examinations, but the drug need not be stopped until evidence for retinopathy is definitive, in particular for patients with active rheumatic or cutaneous disease,” and the overall risk of retinopathy remains low if the principles described in the statement are followed, they concluded.
First author Dr. Rosenbaum disclosed financial relationships with AbbVie, UCB, Gilead, Novartis, Horizon, Roche, Eyevensys, Santen, Corvus, Affibody, Kyverna, Pfizer, Horizon, and UpToDate. Another 5 of the study’s 11 authors also disclosed relationships with multiple companies.
Hydroxychloroquine can be used safely and effectively with attention to dosing, risk factors, and screening, but communication among physicians, patients, and eye care specialists is key to optimizing outcomes and preventing complications, according to a joint statement from four medical societies.
The American College of Rheumatology, American Academy of Dermatology, Rheumatologic Dermatology Society, and the American Academy of Ophthalmology have produced a statement, published in Arthritis & Rheumatology, “to emphasize points of agreement that should be recognized by practitioners in all specialties,” lead author James T. Rosenbaum, MD, of Oregon Health & Science University, Portland, and colleagues wrote.
The statement was developed by a working group that included rheumatologists, ophthalmologists, and dermatologists with records of published studies on the use of hydroxychloroquine (HCQ) and its toxicity. The statement updated elements of the 2016 American Academy of Ophthalmology guidelines for monitoring patients for retinal toxicity when using HCQ.
“The need for collaborative management has triggered this joint statement, which applies only to managing the risk of HCQ retinopathy and does not include consideration of cardiac, muscle, dermatologic, or other toxicities,” the authors noted.
The authors emphasized that HCQ plays a valuable role in controlling many rheumatic diseases, and should not be abandoned out of fear of retinopathy. However, proper dosing, recognition of risk factors, and screening strategies are essential.
Dosing data
Data on HCQ dosing and retinopathy are limited, but the authors cited a study of 2,361 rheumatic disease patients with an average HCQ dosing regimen of 5.0 mg/kg per day or less in which the toxicity risk was less than 2% for up to 10 years of use. Although data show some increase in risk with duration of use, “for a patient with a normal screening exam in a given year, the risk of developing retinopathy in the ensuing year is low (e.g., less than 5%), even after 20 years of use,” the authors said.
Risk factor recognition
“High daily [HCQ] dosage relative to body weight and cumulative dose are the primary risk factors for retinopathy,” the authors noted. Reduced renal function is an additional risk factor, and patients with renal insufficiency should be monitored and may need lower doses.
In addition, patients with a phenotype of initial parafoveal toxicity may be at increased risk for advanced disease evidenced by damage to the foveal center. “The phenotype of initial parafoveal toxicity is not universal, and in many patients (East Asians particularly) the retinal changes may appear initially along the pericentral vascular arcades,” so these patients should be screened with additional tests beyond the central macula, they emphasized.
Screening strategies
Patients should receive a baseline retinal exam within a few months of starting HCQ to rule out underlying retinal disease, according to the statement. The goal of screening is “to detect early retinopathy before a bullseye becomes visible on ophthalmoscopy, since at that severe stage the damage tends to progress even after discontinuing the medication and may eventually threaten central vision,” the authors said.
In the absence of risk factors, patients can defer screening for 5 years, but should be screened annually from 5 years and forward, they said. Examples of underlying retinal disease include “significant macular degeneration, severe diabetic retinopathy, or hereditary disorders of retinal function, but these are judgments best made by the ophthalmologist since mild and stable abnormalities that do not interfere with interpretation of critical diagnostic tests may not be a contraindication” to use of HCQ.
The consensus opinion statement has limitations, notably the shortage of data on optimum HCQ dosage and the lack of prospective studies of toxicity, including the need for studies of the impact of blood levels on toxicity and studies of pharmacogenomics to stratify risk, the authors noted.
“It is important that the drug is not stopped prematurely, but also that it is not continued in the face of definitive evidence of retinal toxicity except in some situations with unusual medical need,” they said.
“Suggestive or uncertain findings should be discussed with the patient and prescribing physician to justify further examinations, but the drug need not be stopped until evidence for retinopathy is definitive, in particular for patients with active rheumatic or cutaneous disease,” and the overall risk of retinopathy remains low if the principles described in the statement are followed, they concluded.
First author Dr. Rosenbaum disclosed financial relationships with AbbVie, UCB, Gilead, Novartis, Horizon, Roche, Eyevensys, Santen, Corvus, Affibody, Kyverna, Pfizer, Horizon, and UpToDate. Another 5 of the study’s 11 authors also disclosed relationships with multiple companies.
Hydroxychloroquine can be used safely and effectively with attention to dosing, risk factors, and screening, but communication among physicians, patients, and eye care specialists is key to optimizing outcomes and preventing complications, according to a joint statement from four medical societies.
The American College of Rheumatology, American Academy of Dermatology, Rheumatologic Dermatology Society, and the American Academy of Ophthalmology have produced a statement, published in Arthritis & Rheumatology, “to emphasize points of agreement that should be recognized by practitioners in all specialties,” lead author James T. Rosenbaum, MD, of Oregon Health & Science University, Portland, and colleagues wrote.
The statement was developed by a working group that included rheumatologists, ophthalmologists, and dermatologists with records of published studies on the use of hydroxychloroquine (HCQ) and its toxicity. The statement updated elements of the 2016 American Academy of Ophthalmology guidelines for monitoring patients for retinal toxicity when using HCQ.
“The need for collaborative management has triggered this joint statement, which applies only to managing the risk of HCQ retinopathy and does not include consideration of cardiac, muscle, dermatologic, or other toxicities,” the authors noted.
The authors emphasized that HCQ plays a valuable role in controlling many rheumatic diseases, and should not be abandoned out of fear of retinopathy. However, proper dosing, recognition of risk factors, and screening strategies are essential.
Dosing data
Data on HCQ dosing and retinopathy are limited, but the authors cited a study of 2,361 rheumatic disease patients with an average HCQ dosing regimen of 5.0 mg/kg per day or less in which the toxicity risk was less than 2% for up to 10 years of use. Although data show some increase in risk with duration of use, “for a patient with a normal screening exam in a given year, the risk of developing retinopathy in the ensuing year is low (e.g., less than 5%), even after 20 years of use,” the authors said.
Risk factor recognition
“High daily [HCQ] dosage relative to body weight and cumulative dose are the primary risk factors for retinopathy,” the authors noted. Reduced renal function is an additional risk factor, and patients with renal insufficiency should be monitored and may need lower doses.
In addition, patients with a phenotype of initial parafoveal toxicity may be at increased risk for advanced disease evidenced by damage to the foveal center. “The phenotype of initial parafoveal toxicity is not universal, and in many patients (East Asians particularly) the retinal changes may appear initially along the pericentral vascular arcades,” so these patients should be screened with additional tests beyond the central macula, they emphasized.
Screening strategies
Patients should receive a baseline retinal exam within a few months of starting HCQ to rule out underlying retinal disease, according to the statement. The goal of screening is “to detect early retinopathy before a bullseye becomes visible on ophthalmoscopy, since at that severe stage the damage tends to progress even after discontinuing the medication and may eventually threaten central vision,” the authors said.
In the absence of risk factors, patients can defer screening for 5 years, but should be screened annually from 5 years and forward, they said. Examples of underlying retinal disease include “significant macular degeneration, severe diabetic retinopathy, or hereditary disorders of retinal function, but these are judgments best made by the ophthalmologist since mild and stable abnormalities that do not interfere with interpretation of critical diagnostic tests may not be a contraindication” to use of HCQ.
The consensus opinion statement has limitations, notably the shortage of data on optimum HCQ dosage and the lack of prospective studies of toxicity, including the need for studies of the impact of blood levels on toxicity and studies of pharmacogenomics to stratify risk, the authors noted.
“It is important that the drug is not stopped prematurely, but also that it is not continued in the face of definitive evidence of retinal toxicity except in some situations with unusual medical need,” they said.
“Suggestive or uncertain findings should be discussed with the patient and prescribing physician to justify further examinations, but the drug need not be stopped until evidence for retinopathy is definitive, in particular for patients with active rheumatic or cutaneous disease,” and the overall risk of retinopathy remains low if the principles described in the statement are followed, they concluded.
First author Dr. Rosenbaum disclosed financial relationships with AbbVie, UCB, Gilead, Novartis, Horizon, Roche, Eyevensys, Santen, Corvus, Affibody, Kyverna, Pfizer, Horizon, and UpToDate. Another 5 of the study’s 11 authors also disclosed relationships with multiple companies.
FROM ARTHRITIS & rHEUMATOLOGY
Heavier girls hit hormonal puberty earlier, but develop breasts later
Girls with more body fat experienced earlier menarche and hormone changes, but later full breast development, compared with those with normal weight, according to longitudinal data from 90 girls aged 8-15 years.
A link between obesity and early puberty has been observed among U.S. girls for decades, but more recent studies suggest that “girls with greater childhood adiposity have earlier thelarche and progress through puberty at a faster rate than normal weight girls,” wrote Madison T. Ortega, MD, of the National Institute of Environmental Health Sciences, Durham, N.C., and colleagues. However, studies involving hormone levels have yielded mixed results, they said.
In a study published in the Journal of Clinical Endocrinology & Metabolism , the researchers followed 36 girls with overweight or obesity and 54 girls with normal weight for 4 years; normal weight was defined as body mass index in the 5th to 85th percentile, overweight was defined as BMI in the 85th to 95th percentile, and obese was defined as greater than 95th percentile. Overweight and obese were combined into one category for comparison with normal weight girls.
Participants had an average of 2.8 study visits during this period and provided additional information by phone and online. Visits included measurement of total body fat using dual-energy x-ray absorptiometry (DXA), Tanner staging, breast ultrasound for morphological staging (BMORPH; A-E), pelvic ultrasound, hormone tests, and menarchal status assessment.
Overall, girls with overweight/obesity (OW/OB) had significantly more advanced breast development at baseline than did those with normal weight (NW), but these girls progressed through BMORPH stage D later than did NW girls. Early-stage breast development was not affected by total body fat. However, “an increase of 5 percentage points in mean total body fat, for example, was associated with a 26% decrease in the transition rate out of stage D,” the researchers noted.
Hormone levels were similar at baseline for follicle-stimulating hormone, inhibin B, estrone (E1), total and free testosterone, and androstenedione. However, these levels increased more quickly after 1 year for girls with OW/OB, while they plateaued in girls with NW and dropped among girls with lower total body fat. Total body fat had no apparent effect on other reproductive hormones including luteinizing hormone, modified vaginal maturation index, and estradiol 2.
The average age of menarche was 12.4 years across all participants, but girls with higher total body fat at baseline were more likely to reach menarche at a younger age. “For every 1-unit increase in visit one total body fat, the chance of achieving menarche at any given time point was 3% higher,” the researchers said. No interaction appeared between race and total body fat with regard to menarche.
Several surprising findings
The study is important because “there have been no longitudinal studies in U.S. girls to examine how total body fat affects serum reproductive hormones or the development of the breast and ovaries using ultrasound imaging,” corresponding author Natalie Shaw, MD, of the National Institute of Environmental Health Sciences, said in an interview.
Dr. Shaw said she was surprised by several of the study findings. “Others have reported increased male-like hormones (androgens) in overweight/obese girls in cross-sectional studies; however, we were surprised to find that FSH and inhibin B were also elevated in girls with excess body fat,” she said. “We also found, unexpectedly, that even though the breast bud appears earlier in overweight/obese girls (thelarche), which signals the onset of puberty, the breast matured more slowly during the course of puberty in overweight/obese girls compared with normal weight girls,” she noted.
“The main take-home message is that puberty looks different in girls with excess body fat; they develop breast tissue earlier, yet take longer to achieve a fully mature breast, and they undergo menarche earlier,” Dr. Shaw said. Clinicians should be aware of the hormonal differences based on body fat, Dr. Shaw emphasized. “Girls with greater body fat had higher levels of FSH (a pituitary hormone), inhibin B (an ovarian hormone), and male-like reproductive hormones (e.g., testosterone) that are made by the adrenal glands and the ovaries in the late stages of puberty,” she said.
Potential implications for adulthood
“The findings in this study contribute to better understanding how total body fat impacts hormonal findings of puberty,” M. Susan Jay, MD, of the Medical College of Wisconsin and Children’s Hospital of Wisconsin, Milwaukee, said in an interview. “Prior studies have linked weight gain as a factor that contributes to pubertal development, but this study is attempting to longitudinally investigate how body weight may affect clinical and biochemical pubertal markers in girls,” she noted.
“The take-home message is that this study and other earlier studies have illustrated that puberty is not a fixed pattern in all individual girls,” Dr. Jay emphasized. “Rather, there are environmental factors which can impact pubertal course,” she said. “In effect, there are pathways through puberty in individual adolescents that require greater ongoing studies to further identify the arc of puberty and the impact of how the length in various stages may affect exposure to estrogen and other neurohormonal factors,” she explained. These factors impact not only adolescence but also future health in adulthood, she said.
“Ongoing prospective studies are needed to identify how factors such as body weight can affect adolescent pubertal development and the possible impact long after adolescence for health issues such as breast cancer,” Dr. Jay added.
The study findings were limited by several factors including the available data from only two completed study visits for most participants, as well as the racial differences among body weight groups and lack of standardized timing for blood draws, the researchers noted.
The study was supported in part by the National Institute of Environmental Health Sciences, and corresponding author Dr. Shaw disclosed support as a Lasker Clinical Research Scholar. The other researchers, as well as Dr. Jay, had no disclosures.
Girls with more body fat experienced earlier menarche and hormone changes, but later full breast development, compared with those with normal weight, according to longitudinal data from 90 girls aged 8-15 years.
A link between obesity and early puberty has been observed among U.S. girls for decades, but more recent studies suggest that “girls with greater childhood adiposity have earlier thelarche and progress through puberty at a faster rate than normal weight girls,” wrote Madison T. Ortega, MD, of the National Institute of Environmental Health Sciences, Durham, N.C., and colleagues. However, studies involving hormone levels have yielded mixed results, they said.
In a study published in the Journal of Clinical Endocrinology & Metabolism , the researchers followed 36 girls with overweight or obesity and 54 girls with normal weight for 4 years; normal weight was defined as body mass index in the 5th to 85th percentile, overweight was defined as BMI in the 85th to 95th percentile, and obese was defined as greater than 95th percentile. Overweight and obese were combined into one category for comparison with normal weight girls.
Participants had an average of 2.8 study visits during this period and provided additional information by phone and online. Visits included measurement of total body fat using dual-energy x-ray absorptiometry (DXA), Tanner staging, breast ultrasound for morphological staging (BMORPH; A-E), pelvic ultrasound, hormone tests, and menarchal status assessment.
Overall, girls with overweight/obesity (OW/OB) had significantly more advanced breast development at baseline than did those with normal weight (NW), but these girls progressed through BMORPH stage D later than did NW girls. Early-stage breast development was not affected by total body fat. However, “an increase of 5 percentage points in mean total body fat, for example, was associated with a 26% decrease in the transition rate out of stage D,” the researchers noted.
Hormone levels were similar at baseline for follicle-stimulating hormone, inhibin B, estrone (E1), total and free testosterone, and androstenedione. However, these levels increased more quickly after 1 year for girls with OW/OB, while they plateaued in girls with NW and dropped among girls with lower total body fat. Total body fat had no apparent effect on other reproductive hormones including luteinizing hormone, modified vaginal maturation index, and estradiol 2.
The average age of menarche was 12.4 years across all participants, but girls with higher total body fat at baseline were more likely to reach menarche at a younger age. “For every 1-unit increase in visit one total body fat, the chance of achieving menarche at any given time point was 3% higher,” the researchers said. No interaction appeared between race and total body fat with regard to menarche.
Several surprising findings
The study is important because “there have been no longitudinal studies in U.S. girls to examine how total body fat affects serum reproductive hormones or the development of the breast and ovaries using ultrasound imaging,” corresponding author Natalie Shaw, MD, of the National Institute of Environmental Health Sciences, said in an interview.
Dr. Shaw said she was surprised by several of the study findings. “Others have reported increased male-like hormones (androgens) in overweight/obese girls in cross-sectional studies; however, we were surprised to find that FSH and inhibin B were also elevated in girls with excess body fat,” she said. “We also found, unexpectedly, that even though the breast bud appears earlier in overweight/obese girls (thelarche), which signals the onset of puberty, the breast matured more slowly during the course of puberty in overweight/obese girls compared with normal weight girls,” she noted.
“The main take-home message is that puberty looks different in girls with excess body fat; they develop breast tissue earlier, yet take longer to achieve a fully mature breast, and they undergo menarche earlier,” Dr. Shaw said. Clinicians should be aware of the hormonal differences based on body fat, Dr. Shaw emphasized. “Girls with greater body fat had higher levels of FSH (a pituitary hormone), inhibin B (an ovarian hormone), and male-like reproductive hormones (e.g., testosterone) that are made by the adrenal glands and the ovaries in the late stages of puberty,” she said.
Potential implications for adulthood
“The findings in this study contribute to better understanding how total body fat impacts hormonal findings of puberty,” M. Susan Jay, MD, of the Medical College of Wisconsin and Children’s Hospital of Wisconsin, Milwaukee, said in an interview. “Prior studies have linked weight gain as a factor that contributes to pubertal development, but this study is attempting to longitudinally investigate how body weight may affect clinical and biochemical pubertal markers in girls,” she noted.
“The take-home message is that this study and other earlier studies have illustrated that puberty is not a fixed pattern in all individual girls,” Dr. Jay emphasized. “Rather, there are environmental factors which can impact pubertal course,” she said. “In effect, there are pathways through puberty in individual adolescents that require greater ongoing studies to further identify the arc of puberty and the impact of how the length in various stages may affect exposure to estrogen and other neurohormonal factors,” she explained. These factors impact not only adolescence but also future health in adulthood, she said.
“Ongoing prospective studies are needed to identify how factors such as body weight can affect adolescent pubertal development and the possible impact long after adolescence for health issues such as breast cancer,” Dr. Jay added.
The study findings were limited by several factors including the available data from only two completed study visits for most participants, as well as the racial differences among body weight groups and lack of standardized timing for blood draws, the researchers noted.
The study was supported in part by the National Institute of Environmental Health Sciences, and corresponding author Dr. Shaw disclosed support as a Lasker Clinical Research Scholar. The other researchers, as well as Dr. Jay, had no disclosures.
Girls with more body fat experienced earlier menarche and hormone changes, but later full breast development, compared with those with normal weight, according to longitudinal data from 90 girls aged 8-15 years.
A link between obesity and early puberty has been observed among U.S. girls for decades, but more recent studies suggest that “girls with greater childhood adiposity have earlier thelarche and progress through puberty at a faster rate than normal weight girls,” wrote Madison T. Ortega, MD, of the National Institute of Environmental Health Sciences, Durham, N.C., and colleagues. However, studies involving hormone levels have yielded mixed results, they said.
In a study published in the Journal of Clinical Endocrinology & Metabolism , the researchers followed 36 girls with overweight or obesity and 54 girls with normal weight for 4 years; normal weight was defined as body mass index in the 5th to 85th percentile, overweight was defined as BMI in the 85th to 95th percentile, and obese was defined as greater than 95th percentile. Overweight and obese were combined into one category for comparison with normal weight girls.
Participants had an average of 2.8 study visits during this period and provided additional information by phone and online. Visits included measurement of total body fat using dual-energy x-ray absorptiometry (DXA), Tanner staging, breast ultrasound for morphological staging (BMORPH; A-E), pelvic ultrasound, hormone tests, and menarchal status assessment.
Overall, girls with overweight/obesity (OW/OB) had significantly more advanced breast development at baseline than did those with normal weight (NW), but these girls progressed through BMORPH stage D later than did NW girls. Early-stage breast development was not affected by total body fat. However, “an increase of 5 percentage points in mean total body fat, for example, was associated with a 26% decrease in the transition rate out of stage D,” the researchers noted.
Hormone levels were similar at baseline for follicle-stimulating hormone, inhibin B, estrone (E1), total and free testosterone, and androstenedione. However, these levels increased more quickly after 1 year for girls with OW/OB, while they plateaued in girls with NW and dropped among girls with lower total body fat. Total body fat had no apparent effect on other reproductive hormones including luteinizing hormone, modified vaginal maturation index, and estradiol 2.
The average age of menarche was 12.4 years across all participants, but girls with higher total body fat at baseline were more likely to reach menarche at a younger age. “For every 1-unit increase in visit one total body fat, the chance of achieving menarche at any given time point was 3% higher,” the researchers said. No interaction appeared between race and total body fat with regard to menarche.
Several surprising findings
The study is important because “there have been no longitudinal studies in U.S. girls to examine how total body fat affects serum reproductive hormones or the development of the breast and ovaries using ultrasound imaging,” corresponding author Natalie Shaw, MD, of the National Institute of Environmental Health Sciences, said in an interview.
Dr. Shaw said she was surprised by several of the study findings. “Others have reported increased male-like hormones (androgens) in overweight/obese girls in cross-sectional studies; however, we were surprised to find that FSH and inhibin B were also elevated in girls with excess body fat,” she said. “We also found, unexpectedly, that even though the breast bud appears earlier in overweight/obese girls (thelarche), which signals the onset of puberty, the breast matured more slowly during the course of puberty in overweight/obese girls compared with normal weight girls,” she noted.
“The main take-home message is that puberty looks different in girls with excess body fat; they develop breast tissue earlier, yet take longer to achieve a fully mature breast, and they undergo menarche earlier,” Dr. Shaw said. Clinicians should be aware of the hormonal differences based on body fat, Dr. Shaw emphasized. “Girls with greater body fat had higher levels of FSH (a pituitary hormone), inhibin B (an ovarian hormone), and male-like reproductive hormones (e.g., testosterone) that are made by the adrenal glands and the ovaries in the late stages of puberty,” she said.
Potential implications for adulthood
“The findings in this study contribute to better understanding how total body fat impacts hormonal findings of puberty,” M. Susan Jay, MD, of the Medical College of Wisconsin and Children’s Hospital of Wisconsin, Milwaukee, said in an interview. “Prior studies have linked weight gain as a factor that contributes to pubertal development, but this study is attempting to longitudinally investigate how body weight may affect clinical and biochemical pubertal markers in girls,” she noted.
“The take-home message is that this study and other earlier studies have illustrated that puberty is not a fixed pattern in all individual girls,” Dr. Jay emphasized. “Rather, there are environmental factors which can impact pubertal course,” she said. “In effect, there are pathways through puberty in individual adolescents that require greater ongoing studies to further identify the arc of puberty and the impact of how the length in various stages may affect exposure to estrogen and other neurohormonal factors,” she explained. These factors impact not only adolescence but also future health in adulthood, she said.
“Ongoing prospective studies are needed to identify how factors such as body weight can affect adolescent pubertal development and the possible impact long after adolescence for health issues such as breast cancer,” Dr. Jay added.
The study findings were limited by several factors including the available data from only two completed study visits for most participants, as well as the racial differences among body weight groups and lack of standardized timing for blood draws, the researchers noted.
The study was supported in part by the National Institute of Environmental Health Sciences, and corresponding author Dr. Shaw disclosed support as a Lasker Clinical Research Scholar. The other researchers, as well as Dr. Jay, had no disclosures.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
The lasting effects of childhood trauma
Childhood trauma, which is also called adverse childhood experiences (ACEs), can have lasting detrimental effects on individuals as they grow and mature into adulthood. ACEs may occur in children age ≤18 years if they experience abuse or neglect, violence, or other traumatic losses. More than 60% of people experience at least 1 ACE, and 1 in 6 individuals reported that they had experienced ≥4 ACEs.1 Subsequent additional ACEs have a cumulative deteriorating impact on the brain. This predisposes individuals to mental health disorders, substance use disorders, and other psychosocial problems. The efficacy of current therapeutic approaches provides only partial symptom resolution. For such individuals, the illness load and health care costs typically remain high across the lifespan.1,2
In this article, we discuss types of ACEs, protective factors and risk factors that influence the development of posttraumatic stress disorder (PTSD) in individuals who experience ACEs, how ACEs can negatively impact mental health in adulthood, and approaches to prevent or treat PTSD and other symptoms.
Types of trauma and correlation with PTSD
ACEs can be indexed as neglect or emotional, physical, or sexual abuse. Physical and sexual abuse strongly correlate with an increased risk of PTSD.3 Although neglect and emotional abuse do not directly predict the development of PTSD, these experiences foretell high rates of lifelong trauma exposure and are indirectly related to late PTSD symptoms.4,5 ACEs can impede an individual’s cognitive, social, and emotional development, diminish quality of life, and lead to an early death.6 The lifetime prevalence of PTSD is 6.1% to 9.2%.7 Compared with men, women are 4 times more likely to develop PTSD following a traumatic event.7
The development of PTSD is influenced by the nature, duration, and degree of trauma, and age at the time of exposure to trauma. Children who survive complex trauma (≥2 types of trauma) have a higher likelihood of developing PTSD.8 Prolonged trauma exposure has a more substantial negative impact than a one-time occurrence. However, it is an erroneous oversimplification to assume that each type of ACE has an equally traumatic effect.6
Factors that protect against PTSD
Factors that can protect against developing PTSD are listed in Table 1.7 Two of these are resilience and hope.
Resilience is defined as an individual’s strength to cope with difficulties in life.9 Resilience has internal psychological characteristics and external factors that aid in protecting against childhood adversities.10,11 The Brief Resilience Scale is a self-assessment that measures innate abilities to cope, including optimism, self-efficacy, patience, faith, and humor.12,13 External factors associated with resilience are family, friends, and community support.11,13
Hope can help in surmounting ACEs. The Adult Hope Scale has been used in many studies to assess this construct in individuals who have survived trauma.13 Some studies have found decreased hope in individuals who sustained early trauma and were diagnosed with PTSD in adulthood.14 A study examining children exposed to domestic violence found that children who showed high hope, endurance, and curiosity were better able to cope with adversities.15
Continue to: PTSD risk factors
PTSD risk factors
Many individual and societal risk factors can influence the likelihood of developing PTSD. Some of these factors are outlined in Table 1.7
Pathophysiology of PTSD
Multiple brain regions, pathways, and neurotransmitters are involved in the development of PTSD. Neuroimaging has identified volume and activity changes of the hippocampus, prefrontal cortex, and amygdala in patients with early trauma and PTSD. Some researchers have suggested a gross reduction in locus coeruleus neuronal volume in war veterans with a likely diagnosis of PTSD compared with controls.16,17 In other studies, chronic stress exposure has been found to cause neuronal cell death and affect neuronal plasticity in the limbic area of the brain.18
Diagnosing PTSD
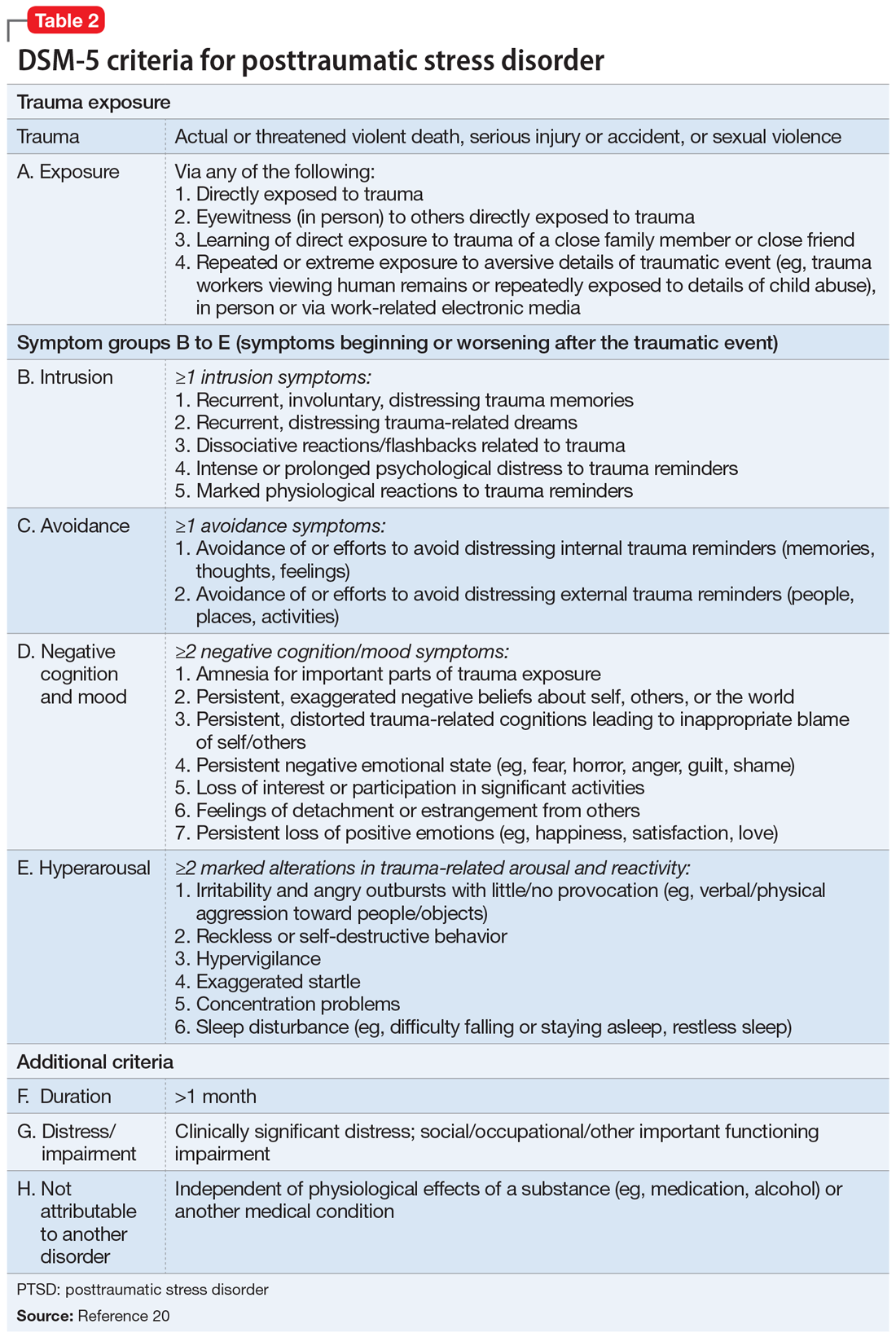
More than 30% of individuals who experience ACEs develop PTSD.19 The DSM-5 diagnostic criteria for PTSD are outlined in Table 2.20 Several instruments are used to determine the diagnosis and assess the severity of PTSD. These include the Clinician-Administered PTSD Scale for DSM-5,21 which is a 30-item structured interview that can be administered in 45 to 60 minutes; the PTSD Symptom Scale Self-Report Version, which is a 17-item, Likert scale, self-report questionnaire; and the Structured Clinical Interview: PTSD Module, which is a semi-structured interview that can take up to several hours to administer.21
Other disorders. In addition to PTSD, individuals with ACEs are at high risk for other mental health issues throughout their lifetime. Individuals with ACE often experience depressive symptoms (approximately 40%); anxiety (approximately 30%); anger; guilt or shame; negative self-cognition; interpersonal difficulties; rumination; and thoughts of self-harm and suicide.22 Epidemiological studies suggest that patients who experience childhood sexual abuse are more likely to develop mood, anxiety, and substance use disorders in adulthood.23,24
Psychotherapeutic treatments for PTSD
Cognitive-behavioral therapy (CBT) addresses the relationship between an individual’s thoughts, emotions, and behaviors. CBT can be used to treat adults and children with PTSD. Before starting CBT, assess the patient’s current safety to ensure that they have the coping skills to manage distress related to their ACEs, and address any coexisting substance use.25
Continue to: According to the American Psychological Association...
According to the American Psychological Association, several CBT-based psychotherapies are recommended for treating PTSD26:
Trauma-focused–CBT includes psychoeducation, trauma narrative, processing, exposure, and relaxation skills training. It consists of approximately 12 to 16 sessions and incorporates elements of family therapy.
Cognitive processing therapy (CPT) focuses on helping patients develop adaptive cognitive domains about the self, the people around them, and the world. CPT therapists assist in information processing by accessing the traumatic memory and trying to eliminate emotions tied to it.25,27 CPT consists of 12 to 16 structured individual, group, or combined sessions.
Prolonged exposure (PE) targets fear-related emotions and works on the principles of habituation to extinguish trauma and fear response to the trigger. This increases self-reliance and competence and decreases the generalization of anxiety to innocuous triggers. PE typically consists of 9 to 12 sessions. PE alone or in combination with cognitive restructuring is successful in treating patients with PTSD, but cognitive restructuring has limited utility in young children.25,27
Cognitive exposure can be individual or group therapy delivered over 3 months, where negative self-evaluation and traumatic memories are challenged with the goal of interrupting maladaptive behaviors and thoughts.27
Continue to: Stress inoculation training
Stress inoculation training (SIT) provides psychoeducation, skills training, role-playing, deep muscle relaxation, paced breathing, and thought stopping. Emphasis is on coaching skills to alleviate anxiety, fear, and symptoms of depression associated with trauma. In SIT, exposures to traumatic memories are indirect (eg, role play), compared with PE, where the exposures are direct.25
The American Psychological Association conditionally recommended several other forms for psychotherapy for treating patients with PTSD26:
Brief eclectic psychotherapy uses CBT and psychodynamic approaches to target feelings of guilt and shame in 16 sessions.27
Narrative exposure therapy consists of 4 to 10 group sessions in which individuals provide detailed narration of the events; the focus is on self-respect and personal rights.27
Eye movement desensitization and reprocessing (EMDR) is a 6- to 12-session, 8-phase treatment that uses principles of accelerated information processing to target nonverbal expression of trauma and dissociative experiences. Patients with PTSD are suggested to have disrupted rapid eye movements. In EMDR, patients follow rhythmic movements of the therapist’s hands or flashed light. This is designed to decrease stress associated with accessing trauma memories, the emotional/physiologic response from the memories, and negative cognitive distortions about self, and to replace negative cognition distortions with positive thoughts about self.25,27
Continue to: Accelerated resolution therapy
Accelerated resolution therapy is a derivative of EMDR. It helps to reconsolidate the emotional and physical experiences associated with distressing memories by replacing them with positive ones or decreasing physiological arousal and anxiety related to the recall of traumatic memories.28
Pharmacologic treatments
Selective serotonin reuptake inhibitors (SSRIs). Multiple studies using different scales have found that paroxetine, sertraline, and fluoxetine can decrease PTSD symptoms. Approximately 60% of patients treated with SSRIs experience partial remission of symptoms, and 20% to 30% experience complete symptom resolution.29 Davidson et al30 found that 22% of patients with PTSD who received fluoxetine had a relapse of symptoms, compared with 50% of patients who received placebo.
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and other antidepressants. The SNRIs venlafaxine and duloxetine can help reduce hyperarousal symptoms and improve mood, anxiety, and sleep.26 Mirtazapine, an alpha 2A/2C adrenoceptor antagonist/5-HT 2A/2C/3 antagonist, can address PTSD symptoms from both serotonergic pathways and increase norepinephrine release by blocking autoreceptors and enhancing alpha-1 receptor activity. This alleviates hyperarousal symptoms and promotes sleep.29 In addition to having monoaminergic effects, antidepressant medications also regulate the hypothalamic–pituitary–adrenal (HPA) axis response to stress and promote neurogenesis in the hippocampal region.29
Adrenergic agents
Adrenergic receptor antagonists. Prazosin, an alpha-1 adrenoceptor antagonist, decreases hyperarousal symptoms, improves sleep, and decreases nightmares related to PTSD by decreasing noradrenergic hyperactivity.29
Beta-blockers such as propranolol can decrease physiological response to trauma but have mixed results in the prevention or improvement of PTSD symptoms.29,31
Continue to: Glucocorticoid receptor agonists
Glucocorticoid receptor agonists. In a very small study, low-dose cortisol decreased the severity of traumatic memory (consolidation phase).32 Glucocorticoid receptor agonists can also diminish memory retrieval (reconsolidation phase) through intrusive thoughts and flashbacks.29
Anticonvulsants, benzodiazepines, and antipsychotics
These medications have had a limited role in the treatment of PTSD.26,29
Future directions: Preventive treatments
Because PTSD has a profound impact on an individual’s quality of life and the development of other illnesses, there is strong interest in finding treatments that can prevent PTSD. Based on limited evidence primarily from animal studies, some researchers have suggested that certain agents may someday be helpful for PTSD prevention29:
Glucocorticoid antagonists such as corticotropin-releasing factor 1 (CRF1) antagonists or cholecystokinin 2 (CCK2) receptor antagonists might promote resilience to stress by inhibiting the HPA axis and influencing the amygdala by decreasing fear conditioning, as observed in animal models. Similarly, in animal models, CRF1 and CCK2 are predicted to decrease memory consolidation in response to exposure to stress.
Adrenoceptor antagonists and agonists also might have a role in preventive treatment, but the evidence is scarce. Prazosin, an alpha-1 adrenoceptor antagonist, was ineffective in animal models.29,31 Propranolol, a beta-adrenoceptor blocker, has had mixed results but can decrease trauma-induced physiological arousal when administered soon after exposure.29
Continue to: N-methyl-
N-methyl-
Bottom Line
Adverse childhood experiences (ACEs) are strong predictors for the development of posttraumatic stress disorder (PTSD) and other mental health or medical issues in late adolescence and adulthood. Experiencing a higher number of ACEs increases the risk of developing PTSD as an adult. Timely psychotherapeutic and pharmacologic interventions can help limit symptoms and reduce the severity of PTSD.
Related Resources
- Smith P, Dalglesih T, Meiser-Stedman R. Practitioner review: posttraumatic stress disorder and its treatment in children and adolescents. J Child Psychol Psychiatry. 2019;60(5):500-515.
- North CS, Hong BA, Downs DL. PTSD: a systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Duloxetine • Cymbalta
Fluoxetine • Prozac
Mirtazapine • Remeron
Paroxetine • Paxil
Prazosin • Minipress
Propranolol • Inderal, Pronol
Sertraline • Zoloft
Venlafaxine • Effexor
1. Centers for Disease Control and Prevention. Preventing adverse childhood experiences. Published April 3, 2020. Accessed January 26, 2021. https://www.cdc.gov/violenceprevention/childabuseandneglect/aces/fastfact.html
2. Kessler RC, McLaughlin KA, Green JG, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197:378-385.
3. Norman RE, Byambaa M, De R, et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Medicine. 2012;9(11):e1001349. doi: 10.1371/journal.pmed.1001349
4. Spertus IL, Yehuda R, Wong CM, et al. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child Abuse Negl. 2003;27(11):1247-1258.
5. Glück TM, Knefel M, Lueger-Schuster B. A network analysis of anger, shame, proposed ICD-11 post-traumatic stress disorder, and different types of childhood trauma in foster care settings in a sample of adult survivors. Eur J Psychotraumatol. 2017;8(suppl 3):1372543. doi: 10.1080/20008198.2017.1372543
6. Edwards VJ, Holden GW, Felitti VJ, et al. Relationship between multiple forms of childhood maltreatment and adult mental health in community respondents: results from the adverse childhood experiences study. Am J Psychiatry. 2003;160:1453-1460.
7. Sareen J. Posttraumatic stress disorder in adults: epidemiology, pathophysiology, clinical manifestations, course, assessment, and diagnosis. UpToDate. Updated December 3, 2020. Accessed January 26, 2021. https://www.uptodate.com/contents/posttraumatic-stress-disorder-in-adults-epidemiology-pathophysiology-clinical-manifestations-course-assessment-and-diagnosis
8. Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999:156;1223-1229.
9. Rutter M. Psychosocial resilience and protective mechanisms. Am J Orthopsychiatry. 1987;57(3):316-331.
10. Ahern NR, Kiehl EM, Sole ML, et al. A review of instruments measuring resilience. Issues Compr Pediatr Nurs. 2006;29(2):103-125.
11. Zimmerman MA. Resiliency theory: a strengths-based approach to research and practice for adolescent health. Health Educ Behav. 2013;40(4):381-383.
12. Connor KM, Davidson JR. Development of a new resilience scale: the Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety. 2003;18(2):76-82.
13. Munoz RT, Hanks H, Hellman CM. Hope and resilience as distinct contributors to psychological flourishing among childhood trauma survivors. Traumatology. 2020;26(2):177-184.
14. Baxter MA, Hemming EJ, McIntosh HC, et al. Exploring the relationship between adverse childhood experiences and hope. J Child Sex Abus. 2017;26(8):948-956.
15. Hellman CM, Gwinn C. Camp HOPE as an intervention for children exposed to domestic violence: a program evaluation of hope, and strength of character. Child Adolesc Soc Work J. 2017;34:269-276.
16. Bracha HS, Garcia-Rill E, Mrak RE, et al. Postmortem locus coeruleus neuron count in three American veterans with probable or possible war-related PTSD. J Neuropsychiatry Clin Neurosci. 2005;17(4):503-9.
17. de Lange GM. Understanding the cellular and molecular alterations in PTSD brains: the necessity of post-mortem brain tissue. Eur J Psychotraumatol. 2017;8(1):1341824. doi: 10.1080/20008198.2017.1341824
18. Zunszain PA, Anacker C, Cattaneo A, et al. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(3):722-729.
19. Greeson JKP, Briggs EC, Kisiel CL, et al. Complex trauma and mental health in children and adolescents placed in foster care: findings from the national child traumatic stress network. Child Welfare. 2011;90(6):91-108.
20. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
21. American Psychological Association. PTSD assessment instruments. Updated September 26, 2018. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/assessment/
22. Bellis MA, Hughes K, Ford K, et al. Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: a systematic review and meta-analysis. Lancet Public Health. 2019;4(10):e517-e528. doi: 10.1016/S2468-2667(19)30145-8
23. Mullen PE, Martin JL, Anderson JC, et al. Childhood sexual abuse and mental health in adult life. Br J Psychiatry. 1993;163:721-732.
24. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women. An epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
25. Chard KM, Gilman R. Counseling trauma victims: 4 brief therapies meet the test. Current Psychiatry. 2005;4(8):50,55-58,61-62.
26. Guideline Development Panel for the Treatment of PTSD in Adults, American Psychological Association. Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. American Psychol. 2019;74(5):596-607.
27. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder. PTSD treatments. Updated June 2020. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/treatments/
28. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART(®)). Behav Sci (Basel). 2012;2(2):115-134.
29. Steckler T, Risbrough V. Pharmacological treatment of PTSD - established and new approaches. Neuropharmacology. 2012;62(2):617-627.
30. Davidson JR, Connor KM, Hertzberg MA, et al. Maintenance therapy with fluoxetine in posttraumatic stress disorder: a placebo-controlled discontinuation study. J Clin Psychopharmacol. 2005;25(2):166-169.
31. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
32. Aerni A, Traber R, Hock C, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiat. 2004;161(8):1488-1490.
33. McGhee LL, Maani CV, Garza TH, et al. The correlation between ketamine and posttraumatic stress disorder in burned service members. J Trauma. 2008;64(2 suppl):S195-S198. doi: 10.1097/TA.0b013e318160ba1d
Childhood trauma, which is also called adverse childhood experiences (ACEs), can have lasting detrimental effects on individuals as they grow and mature into adulthood. ACEs may occur in children age ≤18 years if they experience abuse or neglect, violence, or other traumatic losses. More than 60% of people experience at least 1 ACE, and 1 in 6 individuals reported that they had experienced ≥4 ACEs.1 Subsequent additional ACEs have a cumulative deteriorating impact on the brain. This predisposes individuals to mental health disorders, substance use disorders, and other psychosocial problems. The efficacy of current therapeutic approaches provides only partial symptom resolution. For such individuals, the illness load and health care costs typically remain high across the lifespan.1,2
In this article, we discuss types of ACEs, protective factors and risk factors that influence the development of posttraumatic stress disorder (PTSD) in individuals who experience ACEs, how ACEs can negatively impact mental health in adulthood, and approaches to prevent or treat PTSD and other symptoms.
Types of trauma and correlation with PTSD
ACEs can be indexed as neglect or emotional, physical, or sexual abuse. Physical and sexual abuse strongly correlate with an increased risk of PTSD.3 Although neglect and emotional abuse do not directly predict the development of PTSD, these experiences foretell high rates of lifelong trauma exposure and are indirectly related to late PTSD symptoms.4,5 ACEs can impede an individual’s cognitive, social, and emotional development, diminish quality of life, and lead to an early death.6 The lifetime prevalence of PTSD is 6.1% to 9.2%.7 Compared with men, women are 4 times more likely to develop PTSD following a traumatic event.7
The development of PTSD is influenced by the nature, duration, and degree of trauma, and age at the time of exposure to trauma. Children who survive complex trauma (≥2 types of trauma) have a higher likelihood of developing PTSD.8 Prolonged trauma exposure has a more substantial negative impact than a one-time occurrence. However, it is an erroneous oversimplification to assume that each type of ACE has an equally traumatic effect.6
Factors that protect against PTSD
Factors that can protect against developing PTSD are listed in Table 1.7 Two of these are resilience and hope.
Resilience is defined as an individual’s strength to cope with difficulties in life.9 Resilience has internal psychological characteristics and external factors that aid in protecting against childhood adversities.10,11 The Brief Resilience Scale is a self-assessment that measures innate abilities to cope, including optimism, self-efficacy, patience, faith, and humor.12,13 External factors associated with resilience are family, friends, and community support.11,13
Hope can help in surmounting ACEs. The Adult Hope Scale has been used in many studies to assess this construct in individuals who have survived trauma.13 Some studies have found decreased hope in individuals who sustained early trauma and were diagnosed with PTSD in adulthood.14 A study examining children exposed to domestic violence found that children who showed high hope, endurance, and curiosity were better able to cope with adversities.15
Continue to: PTSD risk factors
PTSD risk factors
Many individual and societal risk factors can influence the likelihood of developing PTSD. Some of these factors are outlined in Table 1.7
Pathophysiology of PTSD
Multiple brain regions, pathways, and neurotransmitters are involved in the development of PTSD. Neuroimaging has identified volume and activity changes of the hippocampus, prefrontal cortex, and amygdala in patients with early trauma and PTSD. Some researchers have suggested a gross reduction in locus coeruleus neuronal volume in war veterans with a likely diagnosis of PTSD compared with controls.16,17 In other studies, chronic stress exposure has been found to cause neuronal cell death and affect neuronal plasticity in the limbic area of the brain.18
Diagnosing PTSD
More than 30% of individuals who experience ACEs develop PTSD.19 The DSM-5 diagnostic criteria for PTSD are outlined in Table 2.20 Several instruments are used to determine the diagnosis and assess the severity of PTSD. These include the Clinician-Administered PTSD Scale for DSM-5,21 which is a 30-item structured interview that can be administered in 45 to 60 minutes; the PTSD Symptom Scale Self-Report Version, which is a 17-item, Likert scale, self-report questionnaire; and the Structured Clinical Interview: PTSD Module, which is a semi-structured interview that can take up to several hours to administer.21

Other disorders. In addition to PTSD, individuals with ACEs are at high risk for other mental health issues throughout their lifetime. Individuals with ACE often experience depressive symptoms (approximately 40%); anxiety (approximately 30%); anger; guilt or shame; negative self-cognition; interpersonal difficulties; rumination; and thoughts of self-harm and suicide.22 Epidemiological studies suggest that patients who experience childhood sexual abuse are more likely to develop mood, anxiety, and substance use disorders in adulthood.23,24
Psychotherapeutic treatments for PTSD
Cognitive-behavioral therapy (CBT) addresses the relationship between an individual’s thoughts, emotions, and behaviors. CBT can be used to treat adults and children with PTSD. Before starting CBT, assess the patient’s current safety to ensure that they have the coping skills to manage distress related to their ACEs, and address any coexisting substance use.25
Continue to: According to the American Psychological Association...
According to the American Psychological Association, several CBT-based psychotherapies are recommended for treating PTSD26:
Trauma-focused–CBT includes psychoeducation, trauma narrative, processing, exposure, and relaxation skills training. It consists of approximately 12 to 16 sessions and incorporates elements of family therapy.
Cognitive processing therapy (CPT) focuses on helping patients develop adaptive cognitive domains about the self, the people around them, and the world. CPT therapists assist in information processing by accessing the traumatic memory and trying to eliminate emotions tied to it.25,27 CPT consists of 12 to 16 structured individual, group, or combined sessions.
Prolonged exposure (PE) targets fear-related emotions and works on the principles of habituation to extinguish trauma and fear response to the trigger. This increases self-reliance and competence and decreases the generalization of anxiety to innocuous triggers. PE typically consists of 9 to 12 sessions. PE alone or in combination with cognitive restructuring is successful in treating patients with PTSD, but cognitive restructuring has limited utility in young children.25,27
Cognitive exposure can be individual or group therapy delivered over 3 months, where negative self-evaluation and traumatic memories are challenged with the goal of interrupting maladaptive behaviors and thoughts.27
Continue to: Stress inoculation training
Stress inoculation training (SIT) provides psychoeducation, skills training, role-playing, deep muscle relaxation, paced breathing, and thought stopping. Emphasis is on coaching skills to alleviate anxiety, fear, and symptoms of depression associated with trauma. In SIT, exposures to traumatic memories are indirect (eg, role play), compared with PE, where the exposures are direct.25
The American Psychological Association conditionally recommended several other forms for psychotherapy for treating patients with PTSD26:
Brief eclectic psychotherapy uses CBT and psychodynamic approaches to target feelings of guilt and shame in 16 sessions.27
Narrative exposure therapy consists of 4 to 10 group sessions in which individuals provide detailed narration of the events; the focus is on self-respect and personal rights.27
Eye movement desensitization and reprocessing (EMDR) is a 6- to 12-session, 8-phase treatment that uses principles of accelerated information processing to target nonverbal expression of trauma and dissociative experiences. Patients with PTSD are suggested to have disrupted rapid eye movements. In EMDR, patients follow rhythmic movements of the therapist’s hands or flashed light. This is designed to decrease stress associated with accessing trauma memories, the emotional/physiologic response from the memories, and negative cognitive distortions about self, and to replace negative cognition distortions with positive thoughts about self.25,27
Continue to: Accelerated resolution therapy
Accelerated resolution therapy is a derivative of EMDR. It helps to reconsolidate the emotional and physical experiences associated with distressing memories by replacing them with positive ones or decreasing physiological arousal and anxiety related to the recall of traumatic memories.28
Pharmacologic treatments
Selective serotonin reuptake inhibitors (SSRIs). Multiple studies using different scales have found that paroxetine, sertraline, and fluoxetine can decrease PTSD symptoms. Approximately 60% of patients treated with SSRIs experience partial remission of symptoms, and 20% to 30% experience complete symptom resolution.29 Davidson et al30 found that 22% of patients with PTSD who received fluoxetine had a relapse of symptoms, compared with 50% of patients who received placebo.
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and other antidepressants. The SNRIs venlafaxine and duloxetine can help reduce hyperarousal symptoms and improve mood, anxiety, and sleep.26 Mirtazapine, an alpha 2A/2C adrenoceptor antagonist/5-HT 2A/2C/3 antagonist, can address PTSD symptoms from both serotonergic pathways and increase norepinephrine release by blocking autoreceptors and enhancing alpha-1 receptor activity. This alleviates hyperarousal symptoms and promotes sleep.29 In addition to having monoaminergic effects, antidepressant medications also regulate the hypothalamic–pituitary–adrenal (HPA) axis response to stress and promote neurogenesis in the hippocampal region.29
Adrenergic agents
Adrenergic receptor antagonists. Prazosin, an alpha-1 adrenoceptor antagonist, decreases hyperarousal symptoms, improves sleep, and decreases nightmares related to PTSD by decreasing noradrenergic hyperactivity.29
Beta-blockers such as propranolol can decrease physiological response to trauma but have mixed results in the prevention or improvement of PTSD symptoms.29,31
Continue to: Glucocorticoid receptor agonists
Glucocorticoid receptor agonists. In a very small study, low-dose cortisol decreased the severity of traumatic memory (consolidation phase).32 Glucocorticoid receptor agonists can also diminish memory retrieval (reconsolidation phase) through intrusive thoughts and flashbacks.29
Anticonvulsants, benzodiazepines, and antipsychotics
These medications have had a limited role in the treatment of PTSD.26,29
Future directions: Preventive treatments
Because PTSD has a profound impact on an individual’s quality of life and the development of other illnesses, there is strong interest in finding treatments that can prevent PTSD. Based on limited evidence primarily from animal studies, some researchers have suggested that certain agents may someday be helpful for PTSD prevention29:
Glucocorticoid antagonists such as corticotropin-releasing factor 1 (CRF1) antagonists or cholecystokinin 2 (CCK2) receptor antagonists might promote resilience to stress by inhibiting the HPA axis and influencing the amygdala by decreasing fear conditioning, as observed in animal models. Similarly, in animal models, CRF1 and CCK2 are predicted to decrease memory consolidation in response to exposure to stress.
Adrenoceptor antagonists and agonists also might have a role in preventive treatment, but the evidence is scarce. Prazosin, an alpha-1 adrenoceptor antagonist, was ineffective in animal models.29,31 Propranolol, a beta-adrenoceptor blocker, has had mixed results but can decrease trauma-induced physiological arousal when administered soon after exposure.29
Continue to: N-methyl-
N-methyl-
Bottom Line
Adverse childhood experiences (ACEs) are strong predictors for the development of posttraumatic stress disorder (PTSD) and other mental health or medical issues in late adolescence and adulthood. Experiencing a higher number of ACEs increases the risk of developing PTSD as an adult. Timely psychotherapeutic and pharmacologic interventions can help limit symptoms and reduce the severity of PTSD.
Related Resources
- Smith P, Dalglesih T, Meiser-Stedman R. Practitioner review: posttraumatic stress disorder and its treatment in children and adolescents. J Child Psychol Psychiatry. 2019;60(5):500-515.
- North CS, Hong BA, Downs DL. PTSD: a systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Duloxetine • Cymbalta
Fluoxetine • Prozac
Mirtazapine • Remeron
Paroxetine • Paxil
Prazosin • Minipress
Propranolol • Inderal, Pronol
Sertraline • Zoloft
Venlafaxine • Effexor
Childhood trauma, which is also called adverse childhood experiences (ACEs), can have lasting detrimental effects on individuals as they grow and mature into adulthood. ACEs may occur in children age ≤18 years if they experience abuse or neglect, violence, or other traumatic losses. More than 60% of people experience at least 1 ACE, and 1 in 6 individuals reported that they had experienced ≥4 ACEs.1 Subsequent additional ACEs have a cumulative deteriorating impact on the brain. This predisposes individuals to mental health disorders, substance use disorders, and other psychosocial problems. The efficacy of current therapeutic approaches provides only partial symptom resolution. For such individuals, the illness load and health care costs typically remain high across the lifespan.1,2
In this article, we discuss types of ACEs, protective factors and risk factors that influence the development of posttraumatic stress disorder (PTSD) in individuals who experience ACEs, how ACEs can negatively impact mental health in adulthood, and approaches to prevent or treat PTSD and other symptoms.
Types of trauma and correlation with PTSD
ACEs can be indexed as neglect or emotional, physical, or sexual abuse. Physical and sexual abuse strongly correlate with an increased risk of PTSD.3 Although neglect and emotional abuse do not directly predict the development of PTSD, these experiences foretell high rates of lifelong trauma exposure and are indirectly related to late PTSD symptoms.4,5 ACEs can impede an individual’s cognitive, social, and emotional development, diminish quality of life, and lead to an early death.6 The lifetime prevalence of PTSD is 6.1% to 9.2%.7 Compared with men, women are 4 times more likely to develop PTSD following a traumatic event.7
The development of PTSD is influenced by the nature, duration, and degree of trauma, and age at the time of exposure to trauma. Children who survive complex trauma (≥2 types of trauma) have a higher likelihood of developing PTSD.8 Prolonged trauma exposure has a more substantial negative impact than a one-time occurrence. However, it is an erroneous oversimplification to assume that each type of ACE has an equally traumatic effect.6
Factors that protect against PTSD
Factors that can protect against developing PTSD are listed in Table 1.7 Two of these are resilience and hope.
Resilience is defined as an individual’s strength to cope with difficulties in life.9 Resilience has internal psychological characteristics and external factors that aid in protecting against childhood adversities.10,11 The Brief Resilience Scale is a self-assessment that measures innate abilities to cope, including optimism, self-efficacy, patience, faith, and humor.12,13 External factors associated with resilience are family, friends, and community support.11,13
Hope can help in surmounting ACEs. The Adult Hope Scale has been used in many studies to assess this construct in individuals who have survived trauma.13 Some studies have found decreased hope in individuals who sustained early trauma and were diagnosed with PTSD in adulthood.14 A study examining children exposed to domestic violence found that children who showed high hope, endurance, and curiosity were better able to cope with adversities.15
Continue to: PTSD risk factors
PTSD risk factors
Many individual and societal risk factors can influence the likelihood of developing PTSD. Some of these factors are outlined in Table 1.7
Pathophysiology of PTSD
Multiple brain regions, pathways, and neurotransmitters are involved in the development of PTSD. Neuroimaging has identified volume and activity changes of the hippocampus, prefrontal cortex, and amygdala in patients with early trauma and PTSD. Some researchers have suggested a gross reduction in locus coeruleus neuronal volume in war veterans with a likely diagnosis of PTSD compared with controls.16,17 In other studies, chronic stress exposure has been found to cause neuronal cell death and affect neuronal plasticity in the limbic area of the brain.18
Diagnosing PTSD
More than 30% of individuals who experience ACEs develop PTSD.19 The DSM-5 diagnostic criteria for PTSD are outlined in Table 2.20 Several instruments are used to determine the diagnosis and assess the severity of PTSD. These include the Clinician-Administered PTSD Scale for DSM-5,21 which is a 30-item structured interview that can be administered in 45 to 60 minutes; the PTSD Symptom Scale Self-Report Version, which is a 17-item, Likert scale, self-report questionnaire; and the Structured Clinical Interview: PTSD Module, which is a semi-structured interview that can take up to several hours to administer.21

Other disorders. In addition to PTSD, individuals with ACEs are at high risk for other mental health issues throughout their lifetime. Individuals with ACE often experience depressive symptoms (approximately 40%); anxiety (approximately 30%); anger; guilt or shame; negative self-cognition; interpersonal difficulties; rumination; and thoughts of self-harm and suicide.22 Epidemiological studies suggest that patients who experience childhood sexual abuse are more likely to develop mood, anxiety, and substance use disorders in adulthood.23,24
Psychotherapeutic treatments for PTSD
Cognitive-behavioral therapy (CBT) addresses the relationship between an individual’s thoughts, emotions, and behaviors. CBT can be used to treat adults and children with PTSD. Before starting CBT, assess the patient’s current safety to ensure that they have the coping skills to manage distress related to their ACEs, and address any coexisting substance use.25
Continue to: According to the American Psychological Association...
According to the American Psychological Association, several CBT-based psychotherapies are recommended for treating PTSD26:
Trauma-focused–CBT includes psychoeducation, trauma narrative, processing, exposure, and relaxation skills training. It consists of approximately 12 to 16 sessions and incorporates elements of family therapy.
Cognitive processing therapy (CPT) focuses on helping patients develop adaptive cognitive domains about the self, the people around them, and the world. CPT therapists assist in information processing by accessing the traumatic memory and trying to eliminate emotions tied to it.25,27 CPT consists of 12 to 16 structured individual, group, or combined sessions.
Prolonged exposure (PE) targets fear-related emotions and works on the principles of habituation to extinguish trauma and fear response to the trigger. This increases self-reliance and competence and decreases the generalization of anxiety to innocuous triggers. PE typically consists of 9 to 12 sessions. PE alone or in combination with cognitive restructuring is successful in treating patients with PTSD, but cognitive restructuring has limited utility in young children.25,27
Cognitive exposure can be individual or group therapy delivered over 3 months, where negative self-evaluation and traumatic memories are challenged with the goal of interrupting maladaptive behaviors and thoughts.27
Continue to: Stress inoculation training
Stress inoculation training (SIT) provides psychoeducation, skills training, role-playing, deep muscle relaxation, paced breathing, and thought stopping. Emphasis is on coaching skills to alleviate anxiety, fear, and symptoms of depression associated with trauma. In SIT, exposures to traumatic memories are indirect (eg, role play), compared with PE, where the exposures are direct.25
The American Psychological Association conditionally recommended several other forms for psychotherapy for treating patients with PTSD26:
Brief eclectic psychotherapy uses CBT and psychodynamic approaches to target feelings of guilt and shame in 16 sessions.27
Narrative exposure therapy consists of 4 to 10 group sessions in which individuals provide detailed narration of the events; the focus is on self-respect and personal rights.27
Eye movement desensitization and reprocessing (EMDR) is a 6- to 12-session, 8-phase treatment that uses principles of accelerated information processing to target nonverbal expression of trauma and dissociative experiences. Patients with PTSD are suggested to have disrupted rapid eye movements. In EMDR, patients follow rhythmic movements of the therapist’s hands or flashed light. This is designed to decrease stress associated with accessing trauma memories, the emotional/physiologic response from the memories, and negative cognitive distortions about self, and to replace negative cognition distortions with positive thoughts about self.25,27
Continue to: Accelerated resolution therapy
Accelerated resolution therapy is a derivative of EMDR. It helps to reconsolidate the emotional and physical experiences associated with distressing memories by replacing them with positive ones or decreasing physiological arousal and anxiety related to the recall of traumatic memories.28
Pharmacologic treatments
Selective serotonin reuptake inhibitors (SSRIs). Multiple studies using different scales have found that paroxetine, sertraline, and fluoxetine can decrease PTSD symptoms. Approximately 60% of patients treated with SSRIs experience partial remission of symptoms, and 20% to 30% experience complete symptom resolution.29 Davidson et al30 found that 22% of patients with PTSD who received fluoxetine had a relapse of symptoms, compared with 50% of patients who received placebo.
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and other antidepressants. The SNRIs venlafaxine and duloxetine can help reduce hyperarousal symptoms and improve mood, anxiety, and sleep.26 Mirtazapine, an alpha 2A/2C adrenoceptor antagonist/5-HT 2A/2C/3 antagonist, can address PTSD symptoms from both serotonergic pathways and increase norepinephrine release by blocking autoreceptors and enhancing alpha-1 receptor activity. This alleviates hyperarousal symptoms and promotes sleep.29 In addition to having monoaminergic effects, antidepressant medications also regulate the hypothalamic–pituitary–adrenal (HPA) axis response to stress and promote neurogenesis in the hippocampal region.29
Adrenergic agents
Adrenergic receptor antagonists. Prazosin, an alpha-1 adrenoceptor antagonist, decreases hyperarousal symptoms, improves sleep, and decreases nightmares related to PTSD by decreasing noradrenergic hyperactivity.29
Beta-blockers such as propranolol can decrease physiological response to trauma but have mixed results in the prevention or improvement of PTSD symptoms.29,31
Continue to: Glucocorticoid receptor agonists
Glucocorticoid receptor agonists. In a very small study, low-dose cortisol decreased the severity of traumatic memory (consolidation phase).32 Glucocorticoid receptor agonists can also diminish memory retrieval (reconsolidation phase) through intrusive thoughts and flashbacks.29
Anticonvulsants, benzodiazepines, and antipsychotics
These medications have had a limited role in the treatment of PTSD.26,29
Future directions: Preventive treatments
Because PTSD has a profound impact on an individual’s quality of life and the development of other illnesses, there is strong interest in finding treatments that can prevent PTSD. Based on limited evidence primarily from animal studies, some researchers have suggested that certain agents may someday be helpful for PTSD prevention29:
Glucocorticoid antagonists such as corticotropin-releasing factor 1 (CRF1) antagonists or cholecystokinin 2 (CCK2) receptor antagonists might promote resilience to stress by inhibiting the HPA axis and influencing the amygdala by decreasing fear conditioning, as observed in animal models. Similarly, in animal models, CRF1 and CCK2 are predicted to decrease memory consolidation in response to exposure to stress.
Adrenoceptor antagonists and agonists also might have a role in preventive treatment, but the evidence is scarce. Prazosin, an alpha-1 adrenoceptor antagonist, was ineffective in animal models.29,31 Propranolol, a beta-adrenoceptor blocker, has had mixed results but can decrease trauma-induced physiological arousal when administered soon after exposure.29
Continue to: N-methyl-
N-methyl-
Bottom Line
Adverse childhood experiences (ACEs) are strong predictors for the development of posttraumatic stress disorder (PTSD) and other mental health or medical issues in late adolescence and adulthood. Experiencing a higher number of ACEs increases the risk of developing PTSD as an adult. Timely psychotherapeutic and pharmacologic interventions can help limit symptoms and reduce the severity of PTSD.
Related Resources
- Smith P, Dalglesih T, Meiser-Stedman R. Practitioner review: posttraumatic stress disorder and its treatment in children and adolescents. J Child Psychol Psychiatry. 2019;60(5):500-515.
- North CS, Hong BA, Downs DL. PTSD: a systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Duloxetine • Cymbalta
Fluoxetine • Prozac
Mirtazapine • Remeron
Paroxetine • Paxil
Prazosin • Minipress
Propranolol • Inderal, Pronol
Sertraline • Zoloft
Venlafaxine • Effexor
1. Centers for Disease Control and Prevention. Preventing adverse childhood experiences. Published April 3, 2020. Accessed January 26, 2021. https://www.cdc.gov/violenceprevention/childabuseandneglect/aces/fastfact.html
2. Kessler RC, McLaughlin KA, Green JG, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197:378-385.
3. Norman RE, Byambaa M, De R, et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Medicine. 2012;9(11):e1001349. doi: 10.1371/journal.pmed.1001349
4. Spertus IL, Yehuda R, Wong CM, et al. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child Abuse Negl. 2003;27(11):1247-1258.
5. Glück TM, Knefel M, Lueger-Schuster B. A network analysis of anger, shame, proposed ICD-11 post-traumatic stress disorder, and different types of childhood trauma in foster care settings in a sample of adult survivors. Eur J Psychotraumatol. 2017;8(suppl 3):1372543. doi: 10.1080/20008198.2017.1372543
6. Edwards VJ, Holden GW, Felitti VJ, et al. Relationship between multiple forms of childhood maltreatment and adult mental health in community respondents: results from the adverse childhood experiences study. Am J Psychiatry. 2003;160:1453-1460.
7. Sareen J. Posttraumatic stress disorder in adults: epidemiology, pathophysiology, clinical manifestations, course, assessment, and diagnosis. UpToDate. Updated December 3, 2020. Accessed January 26, 2021. https://www.uptodate.com/contents/posttraumatic-stress-disorder-in-adults-epidemiology-pathophysiology-clinical-manifestations-course-assessment-and-diagnosis
8. Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999:156;1223-1229.
9. Rutter M. Psychosocial resilience and protective mechanisms. Am J Orthopsychiatry. 1987;57(3):316-331.
10. Ahern NR, Kiehl EM, Sole ML, et al. A review of instruments measuring resilience. Issues Compr Pediatr Nurs. 2006;29(2):103-125.
11. Zimmerman MA. Resiliency theory: a strengths-based approach to research and practice for adolescent health. Health Educ Behav. 2013;40(4):381-383.
12. Connor KM, Davidson JR. Development of a new resilience scale: the Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety. 2003;18(2):76-82.
13. Munoz RT, Hanks H, Hellman CM. Hope and resilience as distinct contributors to psychological flourishing among childhood trauma survivors. Traumatology. 2020;26(2):177-184.
14. Baxter MA, Hemming EJ, McIntosh HC, et al. Exploring the relationship between adverse childhood experiences and hope. J Child Sex Abus. 2017;26(8):948-956.
15. Hellman CM, Gwinn C. Camp HOPE as an intervention for children exposed to domestic violence: a program evaluation of hope, and strength of character. Child Adolesc Soc Work J. 2017;34:269-276.
16. Bracha HS, Garcia-Rill E, Mrak RE, et al. Postmortem locus coeruleus neuron count in three American veterans with probable or possible war-related PTSD. J Neuropsychiatry Clin Neurosci. 2005;17(4):503-9.
17. de Lange GM. Understanding the cellular and molecular alterations in PTSD brains: the necessity of post-mortem brain tissue. Eur J Psychotraumatol. 2017;8(1):1341824. doi: 10.1080/20008198.2017.1341824
18. Zunszain PA, Anacker C, Cattaneo A, et al. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(3):722-729.
19. Greeson JKP, Briggs EC, Kisiel CL, et al. Complex trauma and mental health in children and adolescents placed in foster care: findings from the national child traumatic stress network. Child Welfare. 2011;90(6):91-108.
20. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
21. American Psychological Association. PTSD assessment instruments. Updated September 26, 2018. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/assessment/
22. Bellis MA, Hughes K, Ford K, et al. Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: a systematic review and meta-analysis. Lancet Public Health. 2019;4(10):e517-e528. doi: 10.1016/S2468-2667(19)30145-8
23. Mullen PE, Martin JL, Anderson JC, et al. Childhood sexual abuse and mental health in adult life. Br J Psychiatry. 1993;163:721-732.
24. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women. An epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
25. Chard KM, Gilman R. Counseling trauma victims: 4 brief therapies meet the test. Current Psychiatry. 2005;4(8):50,55-58,61-62.
26. Guideline Development Panel for the Treatment of PTSD in Adults, American Psychological Association. Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. American Psychol. 2019;74(5):596-607.
27. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder. PTSD treatments. Updated June 2020. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/treatments/
28. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART(®)). Behav Sci (Basel). 2012;2(2):115-134.
29. Steckler T, Risbrough V. Pharmacological treatment of PTSD - established and new approaches. Neuropharmacology. 2012;62(2):617-627.
30. Davidson JR, Connor KM, Hertzberg MA, et al. Maintenance therapy with fluoxetine in posttraumatic stress disorder: a placebo-controlled discontinuation study. J Clin Psychopharmacol. 2005;25(2):166-169.
31. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
32. Aerni A, Traber R, Hock C, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiat. 2004;161(8):1488-1490.
33. McGhee LL, Maani CV, Garza TH, et al. The correlation between ketamine and posttraumatic stress disorder in burned service members. J Trauma. 2008;64(2 suppl):S195-S198. doi: 10.1097/TA.0b013e318160ba1d
1. Centers for Disease Control and Prevention. Preventing adverse childhood experiences. Published April 3, 2020. Accessed January 26, 2021. https://www.cdc.gov/violenceprevention/childabuseandneglect/aces/fastfact.html
2. Kessler RC, McLaughlin KA, Green JG, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197:378-385.
3. Norman RE, Byambaa M, De R, et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Medicine. 2012;9(11):e1001349. doi: 10.1371/journal.pmed.1001349
4. Spertus IL, Yehuda R, Wong CM, et al. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child Abuse Negl. 2003;27(11):1247-1258.
5. Glück TM, Knefel M, Lueger-Schuster B. A network analysis of anger, shame, proposed ICD-11 post-traumatic stress disorder, and different types of childhood trauma in foster care settings in a sample of adult survivors. Eur J Psychotraumatol. 2017;8(suppl 3):1372543. doi: 10.1080/20008198.2017.1372543
6. Edwards VJ, Holden GW, Felitti VJ, et al. Relationship between multiple forms of childhood maltreatment and adult mental health in community respondents: results from the adverse childhood experiences study. Am J Psychiatry. 2003;160:1453-1460.
7. Sareen J. Posttraumatic stress disorder in adults: epidemiology, pathophysiology, clinical manifestations, course, assessment, and diagnosis. UpToDate. Updated December 3, 2020. Accessed January 26, 2021. https://www.uptodate.com/contents/posttraumatic-stress-disorder-in-adults-epidemiology-pathophysiology-clinical-manifestations-course-assessment-and-diagnosis
8. Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999:156;1223-1229.
9. Rutter M. Psychosocial resilience and protective mechanisms. Am J Orthopsychiatry. 1987;57(3):316-331.
10. Ahern NR, Kiehl EM, Sole ML, et al. A review of instruments measuring resilience. Issues Compr Pediatr Nurs. 2006;29(2):103-125.
11. Zimmerman MA. Resiliency theory: a strengths-based approach to research and practice for adolescent health. Health Educ Behav. 2013;40(4):381-383.
12. Connor KM, Davidson JR. Development of a new resilience scale: the Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety. 2003;18(2):76-82.
13. Munoz RT, Hanks H, Hellman CM. Hope and resilience as distinct contributors to psychological flourishing among childhood trauma survivors. Traumatology. 2020;26(2):177-184.
14. Baxter MA, Hemming EJ, McIntosh HC, et al. Exploring the relationship between adverse childhood experiences and hope. J Child Sex Abus. 2017;26(8):948-956.
15. Hellman CM, Gwinn C. Camp HOPE as an intervention for children exposed to domestic violence: a program evaluation of hope, and strength of character. Child Adolesc Soc Work J. 2017;34:269-276.
16. Bracha HS, Garcia-Rill E, Mrak RE, et al. Postmortem locus coeruleus neuron count in three American veterans with probable or possible war-related PTSD. J Neuropsychiatry Clin Neurosci. 2005;17(4):503-9.
17. de Lange GM. Understanding the cellular and molecular alterations in PTSD brains: the necessity of post-mortem brain tissue. Eur J Psychotraumatol. 2017;8(1):1341824. doi: 10.1080/20008198.2017.1341824
18. Zunszain PA, Anacker C, Cattaneo A, et al. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(3):722-729.
19. Greeson JKP, Briggs EC, Kisiel CL, et al. Complex trauma and mental health in children and adolescents placed in foster care: findings from the national child traumatic stress network. Child Welfare. 2011;90(6):91-108.
20. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
21. American Psychological Association. PTSD assessment instruments. Updated September 26, 2018. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/assessment/
22. Bellis MA, Hughes K, Ford K, et al. Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: a systematic review and meta-analysis. Lancet Public Health. 2019;4(10):e517-e528. doi: 10.1016/S2468-2667(19)30145-8
23. Mullen PE, Martin JL, Anderson JC, et al. Childhood sexual abuse and mental health in adult life. Br J Psychiatry. 1993;163:721-732.
24. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women. An epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
25. Chard KM, Gilman R. Counseling trauma victims: 4 brief therapies meet the test. Current Psychiatry. 2005;4(8):50,55-58,61-62.
26. Guideline Development Panel for the Treatment of PTSD in Adults, American Psychological Association. Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. American Psychol. 2019;74(5):596-607.
27. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder. PTSD treatments. Updated June 2020. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/treatments/
28. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART(®)). Behav Sci (Basel). 2012;2(2):115-134.
29. Steckler T, Risbrough V. Pharmacological treatment of PTSD - established and new approaches. Neuropharmacology. 2012;62(2):617-627.
30. Davidson JR, Connor KM, Hertzberg MA, et al. Maintenance therapy with fluoxetine in posttraumatic stress disorder: a placebo-controlled discontinuation study. J Clin Psychopharmacol. 2005;25(2):166-169.
31. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
32. Aerni A, Traber R, Hock C, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiat. 2004;161(8):1488-1490.
33. McGhee LL, Maani CV, Garza TH, et al. The correlation between ketamine and posttraumatic stress disorder in burned service members. J Trauma. 2008;64(2 suppl):S195-S198. doi: 10.1097/TA.0b013e318160ba1d
Treatment Options for Atopic Dermatitis in Children
Until recently, atopic dermatitis was considered a childhood disease that was self-limited over a few years. Emerging studies have shown that the burden of atopic dermatitis includes potential cardiac disease in adulthood, comorbidities including allergy and psychological disorders, and possible superinfection complications.
Dr Lawrence F. Eichenfield, chief of the department of pediatric and adolescent dermatology at Rady Children's Hospital, reports on biological, systemic, and topical treatments either currently in use or being studied for children suffering from atopic dermatitis. These studies include both steroid and steroid-sparing topical agents, a novel AhR modulating agent, as well as JAK inhibitors that are under active investigation.
--
Lawrence F. Eichenfield, MD, Distinguished Professor; Vice Chair, Department of Dermatology and Pediatrics, University of California, San Diego; Chief, Department of Pediatric and Adolescent Dermatology, Rady Children's Hospital, San Diego, California.
Lawrence F. Eichenfield, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Dermavant; Dermira; Forte Biosciences; Galderma Laboratories; Incyte; Leo Pharma; Eli Lilly and Company; Otsuka; Novartis; Pfizer. Serve(d) as a speaker or a member of a speakers bureau for: Regeneron; Sanofi-Genzyme; Pfizer. Received research grant from: AbbVie; Regeneron; Sanofi Genzyme; Ortho Dermatology. Serve(d) on the data safety monitoring board for: Asana; Glenmark/Ichnos.
Until recently, atopic dermatitis was considered a childhood disease that was self-limited over a few years. Emerging studies have shown that the burden of atopic dermatitis includes potential cardiac disease in adulthood, comorbidities including allergy and psychological disorders, and possible superinfection complications.
Dr Lawrence F. Eichenfield, chief of the department of pediatric and adolescent dermatology at Rady Children's Hospital, reports on biological, systemic, and topical treatments either currently in use or being studied for children suffering from atopic dermatitis. These studies include both steroid and steroid-sparing topical agents, a novel AhR modulating agent, as well as JAK inhibitors that are under active investigation.
--
Lawrence F. Eichenfield, MD, Distinguished Professor; Vice Chair, Department of Dermatology and Pediatrics, University of California, San Diego; Chief, Department of Pediatric and Adolescent Dermatology, Rady Children's Hospital, San Diego, California.
Lawrence F. Eichenfield, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Dermavant; Dermira; Forte Biosciences; Galderma Laboratories; Incyte; Leo Pharma; Eli Lilly and Company; Otsuka; Novartis; Pfizer. Serve(d) as a speaker or a member of a speakers bureau for: Regeneron; Sanofi-Genzyme; Pfizer. Received research grant from: AbbVie; Regeneron; Sanofi Genzyme; Ortho Dermatology. Serve(d) on the data safety monitoring board for: Asana; Glenmark/Ichnos.
Until recently, atopic dermatitis was considered a childhood disease that was self-limited over a few years. Emerging studies have shown that the burden of atopic dermatitis includes potential cardiac disease in adulthood, comorbidities including allergy and psychological disorders, and possible superinfection complications.
Dr Lawrence F. Eichenfield, chief of the department of pediatric and adolescent dermatology at Rady Children's Hospital, reports on biological, systemic, and topical treatments either currently in use or being studied for children suffering from atopic dermatitis. These studies include both steroid and steroid-sparing topical agents, a novel AhR modulating agent, as well as JAK inhibitors that are under active investigation.
--
Lawrence F. Eichenfield, MD, Distinguished Professor; Vice Chair, Department of Dermatology and Pediatrics, University of California, San Diego; Chief, Department of Pediatric and Adolescent Dermatology, Rady Children's Hospital, San Diego, California.
Lawrence F. Eichenfield, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Dermavant; Dermira; Forte Biosciences; Galderma Laboratories; Incyte; Leo Pharma; Eli Lilly and Company; Otsuka; Novartis; Pfizer. Serve(d) as a speaker or a member of a speakers bureau for: Regeneron; Sanofi-Genzyme; Pfizer. Received research grant from: AbbVie; Regeneron; Sanofi Genzyme; Ortho Dermatology. Serve(d) on the data safety monitoring board for: Asana; Glenmark/Ichnos.

FDA approves first targeted treatment for rare DMD mutation
, the agency has announced.
This particular mutation of the DMD gene “is amenable to exon 45 skipping,” the FDA noted in a press release. The agency added that this is its first approval of a targeted treatment for patients with the mutation.
“Developing drugs designed for patients with specific mutations is a critical part of personalized medicine,” Eric Bastings, MD, deputy director of the Office of Neuroscience at the FDA’s Center for Drug Evaluation and Research, said in a statement.
The approval was based on results from a 43-person randomized controlled trial. Patients who received casimersen had a greater increase in production of the muscle-fiber protein dystrophin compared with their counterparts who received placebo.
Approved – with cautions
The FDA noted that DMD prevalence worldwide is about 1 in 3,600 boys – although it can also affect girls in rare cases. Symptoms of the disorder are commonly first observed around age 3 years but worsen steadily over time. DMD gene mutations lead to a decrease in dystrophin.
As reported by Medscape Medical News in August, the FDA approved viltolarsen (Viltepso, NS Pharma) for the treatment of DMD in patients with a confirmed mutation amenable to exon 53 skipping, following approval of golodirsen injection (Vyondys 53, Sarepta Therapeutics) for the same indication in December 2019.
The DMD gene mutation that is amenable to exon 45 skipping is present in about 8% of patients with DMD.
The trial that carried weight with the FDA included 43 male participants with DMD aged 7-20 years. All were confirmed to have the exon 45-skipping gene mutation and all were randomly assigned 2:1 to received IV casimersen 30 mg/kg or matching placebo.
Results showed that, between baseline and 48 weeks post treatment, the casimersen group showed a significantly higher increase in levels of dystrophin protein than in the placebo group.
Upper respiratory tract infections, fever, joint and throat pain, headache, and cough were the most common adverse events experienced by the active-treatment group.
Although the clinical studies assessing casimersen did not show any reports of kidney toxicity, the adverse event was observed in some nonclinical studies. Therefore, clinicians should monitor kidney function in any patient receiving this treatment, the FDA recommended.
Overall, “the FDA has concluded that the data submitted by the applicant demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit” in this patient population, the agency said in its press release.
However, it noted that definitive clinical benefits such as improved motor function were not “established.”
“In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of [other] available therapy,” the agency said.
It added that the manufacturer is currently conducting a multicenter study focused on the safety and efficacy of the drug in ambulatory patients with DMD.
The FDA approved casimersen using its Accelerated Approval pathway, granted Fast Track and Priority Review designations to its applications, and gave the treatment Orphan Drug designation.
A version of this article first appeared on Medscape.com.
, the agency has announced.
This particular mutation of the DMD gene “is amenable to exon 45 skipping,” the FDA noted in a press release. The agency added that this is its first approval of a targeted treatment for patients with the mutation.
“Developing drugs designed for patients with specific mutations is a critical part of personalized medicine,” Eric Bastings, MD, deputy director of the Office of Neuroscience at the FDA’s Center for Drug Evaluation and Research, said in a statement.
The approval was based on results from a 43-person randomized controlled trial. Patients who received casimersen had a greater increase in production of the muscle-fiber protein dystrophin compared with their counterparts who received placebo.
Approved – with cautions
The FDA noted that DMD prevalence worldwide is about 1 in 3,600 boys – although it can also affect girls in rare cases. Symptoms of the disorder are commonly first observed around age 3 years but worsen steadily over time. DMD gene mutations lead to a decrease in dystrophin.
As reported by Medscape Medical News in August, the FDA approved viltolarsen (Viltepso, NS Pharma) for the treatment of DMD in patients with a confirmed mutation amenable to exon 53 skipping, following approval of golodirsen injection (Vyondys 53, Sarepta Therapeutics) for the same indication in December 2019.
The DMD gene mutation that is amenable to exon 45 skipping is present in about 8% of patients with DMD.
The trial that carried weight with the FDA included 43 male participants with DMD aged 7-20 years. All were confirmed to have the exon 45-skipping gene mutation and all were randomly assigned 2:1 to received IV casimersen 30 mg/kg or matching placebo.
Results showed that, between baseline and 48 weeks post treatment, the casimersen group showed a significantly higher increase in levels of dystrophin protein than in the placebo group.
Upper respiratory tract infections, fever, joint and throat pain, headache, and cough were the most common adverse events experienced by the active-treatment group.
Although the clinical studies assessing casimersen did not show any reports of kidney toxicity, the adverse event was observed in some nonclinical studies. Therefore, clinicians should monitor kidney function in any patient receiving this treatment, the FDA recommended.
Overall, “the FDA has concluded that the data submitted by the applicant demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit” in this patient population, the agency said in its press release.
However, it noted that definitive clinical benefits such as improved motor function were not “established.”
“In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of [other] available therapy,” the agency said.
It added that the manufacturer is currently conducting a multicenter study focused on the safety and efficacy of the drug in ambulatory patients with DMD.
The FDA approved casimersen using its Accelerated Approval pathway, granted Fast Track and Priority Review designations to its applications, and gave the treatment Orphan Drug designation.
A version of this article first appeared on Medscape.com.
, the agency has announced.
This particular mutation of the DMD gene “is amenable to exon 45 skipping,” the FDA noted in a press release. The agency added that this is its first approval of a targeted treatment for patients with the mutation.
“Developing drugs designed for patients with specific mutations is a critical part of personalized medicine,” Eric Bastings, MD, deputy director of the Office of Neuroscience at the FDA’s Center for Drug Evaluation and Research, said in a statement.
The approval was based on results from a 43-person randomized controlled trial. Patients who received casimersen had a greater increase in production of the muscle-fiber protein dystrophin compared with their counterparts who received placebo.
Approved – with cautions
The FDA noted that DMD prevalence worldwide is about 1 in 3,600 boys – although it can also affect girls in rare cases. Symptoms of the disorder are commonly first observed around age 3 years but worsen steadily over time. DMD gene mutations lead to a decrease in dystrophin.
As reported by Medscape Medical News in August, the FDA approved viltolarsen (Viltepso, NS Pharma) for the treatment of DMD in patients with a confirmed mutation amenable to exon 53 skipping, following approval of golodirsen injection (Vyondys 53, Sarepta Therapeutics) for the same indication in December 2019.
The DMD gene mutation that is amenable to exon 45 skipping is present in about 8% of patients with DMD.
The trial that carried weight with the FDA included 43 male participants with DMD aged 7-20 years. All were confirmed to have the exon 45-skipping gene mutation and all were randomly assigned 2:1 to received IV casimersen 30 mg/kg or matching placebo.
Results showed that, between baseline and 48 weeks post treatment, the casimersen group showed a significantly higher increase in levels of dystrophin protein than in the placebo group.
Upper respiratory tract infections, fever, joint and throat pain, headache, and cough were the most common adverse events experienced by the active-treatment group.
Although the clinical studies assessing casimersen did not show any reports of kidney toxicity, the adverse event was observed in some nonclinical studies. Therefore, clinicians should monitor kidney function in any patient receiving this treatment, the FDA recommended.
Overall, “the FDA has concluded that the data submitted by the applicant demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit” in this patient population, the agency said in its press release.
However, it noted that definitive clinical benefits such as improved motor function were not “established.”
“In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of [other] available therapy,” the agency said.
It added that the manufacturer is currently conducting a multicenter study focused on the safety and efficacy of the drug in ambulatory patients with DMD.
The FDA approved casimersen using its Accelerated Approval pathway, granted Fast Track and Priority Review designations to its applications, and gave the treatment Orphan Drug designation.
A version of this article first appeared on Medscape.com.
Data on atopic dermatitis risk factors are accumulating
, according to Zelma Chiesa Fuxench, MD.
This gene codes for profilaggrin, a protein, which is then cleaved to form filaggrin, which helps to organize the cytoskeleton of the skin and is an important structural component of the skin. The understanding is that patients who have filaggrin mutations tend to have earlier onset and more persistent disease, Dr. Chiesa Fuxench, of the department of dermatology at the University of Pennsylvania, Philadelphia, said during the Revolutionizing Atopic Dermatitis virtual symposium.
“Prior studies have shown that mutations in the FLG gene can confer a risk of developed AD that is two- to sevenfold with variants R501X and the 22804del4 frequently described. It is important to note that most of these findings have been described primarily in populations of European descent, with other variants being found in populations of African nation descent, and seem to be more prevalent in populations with early onset disease.”
Environmental factors
Other AD-related risk factors that have been previously described in the literature include environmental factors such as climate, diet, breastfeeding, obesity, pollution, tobacco smoke, pet ownership, and microbiome or gut microflora. “The list of culprits is ever increasing,” she said. “However, it’s important to recognize that data to support some of these associations are lacking, and oftentimes, a lot of the results are contradictory.”
As part of the International Study of Asthma and Allergies in Childhood, researchers evaluated the association between climate factors with the 12-month period prevalence rates of symptoms of atopic eczema in children. They found that patients who lived at higher latitudes and those who lived in areas where there were lower mean outdoor temperatures tended to have a higher prevalence of eczema symptoms. Worldwide, they found that symptoms of eczema were also prevalent in areas where there was lower indoor humidity.
“The authors concluded that they can’t really demonstrate a cause and effect, and that while latitude and temperature changes appear to affect the prevalence of eczema, they may do so indirectly, perhaps to changes in behavior and differences in sun exposure,” said Dr. Chiesa Fuxench, who was not involved with the study. “For example, we know that vitamin D is a protective risk factor for AD. Low vitamin D has been associated with more severe disease in some studies. We also know that UV exposure leads to the conversion of filaggrin degradation products such as trans-urocanic acid into cis-urocanic acid, which has been demonstrated to have immunosuppressive effects.”
A systematic review and meta-analysis of nine articles found small associations, which were significant, between being born in the winter (odds ratio, 1.15) and fall (OR, 1.16) and the risk of developing AD, compared with being born in the spring and summer. However, an analysis of satellite-derived data on air temperature across the United States from 1993 to 2011 found that as ambient air temperature increases, so did the risk for an ambulatory visit for AD to physicians from the National Ambulatory Medical Care Survey.
In all areas but the south, the largest number of AD visits occur in the spring. In the south, more AD visits occur in the summer. “This raises the point that we don’t really know everything when it comes to the influence of temperature and climate change on AD,” Dr. Chiesa Fuxench said.
Several maternal and neonatal risk factors for AD have been described in the literature, including the effect of prenatal exposure to antibiotics. In one large analysis, investigators assessed the association among 18-month-old children in the Danish National Birth Cohort, which included 62,560 mother-child pairs. They found that prenatal antibiotic use was associated with an increased odds of AD among children born to atopic mothers but only when used during all three trimesters (adjusted OR, 1.45). When they further stratified these analyses by type of birth (vaginal versus C-section), the association persisted in both groups, but was stronger among those delivered by C-section.
Probiotics
The role of probiotics to reduce the risk for AD has also been investigated. “We do know that probiotics could potentially be helpful, and it is often a readily available intervention,” Dr. Chiesa Fuxench said. “But the question still is how and when to supplement.”
In a systematic review and meta-analysis, researchers examined supplementation with probiotics given to breastfeeding mothers, pregnant mothers, or directly given to infants, and the risk of developing AD up to 18 months of age. They found that overall, probiotic exposure resulted in decreased risk of developing AD. In stratified analyses, the strongest association was observed for those who received probiotics during their pregnancy, during breastfeeding, and as an infant, which conferred about a 25% reduced risk.
Antibiotic exposure
What about early-life exposure to antibiotics on one’s risk for developing AD? A meta-analysis of 22 studies found that children who had been exposed to antibiotics during the first 2 years of life had an increased risk of eczema (OR, 1.26), compared with children who had not been exposed during the same period of time. “Interesting hypotheses can be generated from this study,” she said. “Perhaps future steps should focus on the impact of antibiotic exposure, the gut microbiome, and maternal risk factors for AD.”
In a separate study that supported these findings, researchers evaluated the association between the use of acid-suppressive medications and antibiotics during infancy and the development of allergic disease in early childhood. They found that exposure to either of these medications during the first six months of infancy resulted in a mild increased risk of developing AD, and concluded that they should be used during infancy only in situations of clear clinical benefit. “We should be good stewards of antibiotic use, in particular due to concern for antibiotic resistance in the population overall,” Dr. Chiesa Fuxench said.
Prevention strategies
Several AD prevention strategies have also been described in the medical literature, including the use of daily emollients during infancy. In a multicenter trial carried out in the United Kingdom, researchers tested whether daily use of emollient in the first year of life could prevent eczema in high-risk children, which was defined as having at least one first-degree relative with parent-reported eczema, allergic rhinitis, or asthma. The primary outcome was eczema at age 2 years. The researchers found no evidence to suggest that daily emollient use during the first year of life prevents eczema.
Another study, the PreventADALL trial of 2,397 infants, consisted of four treatment arms: a control group advised to follow national guidelines on infant nutrition; a skin intervention group that was asked to use skin emollients, a food intervention group with early introduction of peanut, cow’s milk, wheat, and egg, and a combined skin and food intervention. The investigators found no difference in the risk reduction of developing AD among patients who were treated with skin emollients or early complementary feeding, and concluded that these types of interventions should not be considered as interventions to prevent AD in this cohort of patients.
However, Dr. Chiesa Fuxench emphasized that emollients and moisturizers are an important part of the treatment regimen for AD patients. A Cochrane systematic review of nearly 80 randomized, controlled trials evaluating the use of emollients in eczema found that most moisturizers showed some beneficial effects in addition to active treatment, including prolonging the time to flare, reducing the number of flares, and reducing the amount of topical corticosteroids used.
For treatment, Dr. Chiesa Fuxench recommends a proactive approach focused on short-term induction therapy with intensive topical anti-inflammatories until the affected area is almost healed, followed by maintenance therapy that involves use of a long-term, low- to mid-potency steroid or a topical calcineurin inhibitor to previously affected areas. “These interventions have been shown to decrease the risk of recurrence and can shorten the treatment duration in the event of a flare,” she said.
She also favors a time-contingent approach to treating patients with AD. “As physicians, we tend to do our visits more as symptom contingent, which means when a patient is flaring. This reinforces the view that this is a difficult disease to treat, and that there is no hope,” she pointed out. But for chronic diseases, she added, “a time-contingent approach with appointments at set intervals leads to a different perception. It can result in better compliance, because skin care might be performed more regularly. It’s analogous to when you know you’re going to see the dentist so you floss more regularly the week before your appointment. There also seems to be less pressure on physicians and patients because you are seeing each other more frequently; you can talk more openly about what’s working and what’s not.”
Dr. Chiesa Fuxench reported having no disclosures relevant to her presentation.
, according to Zelma Chiesa Fuxench, MD.
This gene codes for profilaggrin, a protein, which is then cleaved to form filaggrin, which helps to organize the cytoskeleton of the skin and is an important structural component of the skin. The understanding is that patients who have filaggrin mutations tend to have earlier onset and more persistent disease, Dr. Chiesa Fuxench, of the department of dermatology at the University of Pennsylvania, Philadelphia, said during the Revolutionizing Atopic Dermatitis virtual symposium.
“Prior studies have shown that mutations in the FLG gene can confer a risk of developed AD that is two- to sevenfold with variants R501X and the 22804del4 frequently described. It is important to note that most of these findings have been described primarily in populations of European descent, with other variants being found in populations of African nation descent, and seem to be more prevalent in populations with early onset disease.”
Environmental factors
Other AD-related risk factors that have been previously described in the literature include environmental factors such as climate, diet, breastfeeding, obesity, pollution, tobacco smoke, pet ownership, and microbiome or gut microflora. “The list of culprits is ever increasing,” she said. “However, it’s important to recognize that data to support some of these associations are lacking, and oftentimes, a lot of the results are contradictory.”
As part of the International Study of Asthma and Allergies in Childhood, researchers evaluated the association between climate factors with the 12-month period prevalence rates of symptoms of atopic eczema in children. They found that patients who lived at higher latitudes and those who lived in areas where there were lower mean outdoor temperatures tended to have a higher prevalence of eczema symptoms. Worldwide, they found that symptoms of eczema were also prevalent in areas where there was lower indoor humidity.
“The authors concluded that they can’t really demonstrate a cause and effect, and that while latitude and temperature changes appear to affect the prevalence of eczema, they may do so indirectly, perhaps to changes in behavior and differences in sun exposure,” said Dr. Chiesa Fuxench, who was not involved with the study. “For example, we know that vitamin D is a protective risk factor for AD. Low vitamin D has been associated with more severe disease in some studies. We also know that UV exposure leads to the conversion of filaggrin degradation products such as trans-urocanic acid into cis-urocanic acid, which has been demonstrated to have immunosuppressive effects.”
A systematic review and meta-analysis of nine articles found small associations, which were significant, between being born in the winter (odds ratio, 1.15) and fall (OR, 1.16) and the risk of developing AD, compared with being born in the spring and summer. However, an analysis of satellite-derived data on air temperature across the United States from 1993 to 2011 found that as ambient air temperature increases, so did the risk for an ambulatory visit for AD to physicians from the National Ambulatory Medical Care Survey.
In all areas but the south, the largest number of AD visits occur in the spring. In the south, more AD visits occur in the summer. “This raises the point that we don’t really know everything when it comes to the influence of temperature and climate change on AD,” Dr. Chiesa Fuxench said.
Several maternal and neonatal risk factors for AD have been described in the literature, including the effect of prenatal exposure to antibiotics. In one large analysis, investigators assessed the association among 18-month-old children in the Danish National Birth Cohort, which included 62,560 mother-child pairs. They found that prenatal antibiotic use was associated with an increased odds of AD among children born to atopic mothers but only when used during all three trimesters (adjusted OR, 1.45). When they further stratified these analyses by type of birth (vaginal versus C-section), the association persisted in both groups, but was stronger among those delivered by C-section.
Probiotics
The role of probiotics to reduce the risk for AD has also been investigated. “We do know that probiotics could potentially be helpful, and it is often a readily available intervention,” Dr. Chiesa Fuxench said. “But the question still is how and when to supplement.”
In a systematic review and meta-analysis, researchers examined supplementation with probiotics given to breastfeeding mothers, pregnant mothers, or directly given to infants, and the risk of developing AD up to 18 months of age. They found that overall, probiotic exposure resulted in decreased risk of developing AD. In stratified analyses, the strongest association was observed for those who received probiotics during their pregnancy, during breastfeeding, and as an infant, which conferred about a 25% reduced risk.
Antibiotic exposure
What about early-life exposure to antibiotics on one’s risk for developing AD? A meta-analysis of 22 studies found that children who had been exposed to antibiotics during the first 2 years of life had an increased risk of eczema (OR, 1.26), compared with children who had not been exposed during the same period of time. “Interesting hypotheses can be generated from this study,” she said. “Perhaps future steps should focus on the impact of antibiotic exposure, the gut microbiome, and maternal risk factors for AD.”
In a separate study that supported these findings, researchers evaluated the association between the use of acid-suppressive medications and antibiotics during infancy and the development of allergic disease in early childhood. They found that exposure to either of these medications during the first six months of infancy resulted in a mild increased risk of developing AD, and concluded that they should be used during infancy only in situations of clear clinical benefit. “We should be good stewards of antibiotic use, in particular due to concern for antibiotic resistance in the population overall,” Dr. Chiesa Fuxench said.
Prevention strategies
Several AD prevention strategies have also been described in the medical literature, including the use of daily emollients during infancy. In a multicenter trial carried out in the United Kingdom, researchers tested whether daily use of emollient in the first year of life could prevent eczema in high-risk children, which was defined as having at least one first-degree relative with parent-reported eczema, allergic rhinitis, or asthma. The primary outcome was eczema at age 2 years. The researchers found no evidence to suggest that daily emollient use during the first year of life prevents eczema.
Another study, the PreventADALL trial of 2,397 infants, consisted of four treatment arms: a control group advised to follow national guidelines on infant nutrition; a skin intervention group that was asked to use skin emollients, a food intervention group with early introduction of peanut, cow’s milk, wheat, and egg, and a combined skin and food intervention. The investigators found no difference in the risk reduction of developing AD among patients who were treated with skin emollients or early complementary feeding, and concluded that these types of interventions should not be considered as interventions to prevent AD in this cohort of patients.
However, Dr. Chiesa Fuxench emphasized that emollients and moisturizers are an important part of the treatment regimen for AD patients. A Cochrane systematic review of nearly 80 randomized, controlled trials evaluating the use of emollients in eczema found that most moisturizers showed some beneficial effects in addition to active treatment, including prolonging the time to flare, reducing the number of flares, and reducing the amount of topical corticosteroids used.
For treatment, Dr. Chiesa Fuxench recommends a proactive approach focused on short-term induction therapy with intensive topical anti-inflammatories until the affected area is almost healed, followed by maintenance therapy that involves use of a long-term, low- to mid-potency steroid or a topical calcineurin inhibitor to previously affected areas. “These interventions have been shown to decrease the risk of recurrence and can shorten the treatment duration in the event of a flare,” she said.
She also favors a time-contingent approach to treating patients with AD. “As physicians, we tend to do our visits more as symptom contingent, which means when a patient is flaring. This reinforces the view that this is a difficult disease to treat, and that there is no hope,” she pointed out. But for chronic diseases, she added, “a time-contingent approach with appointments at set intervals leads to a different perception. It can result in better compliance, because skin care might be performed more regularly. It’s analogous to when you know you’re going to see the dentist so you floss more regularly the week before your appointment. There also seems to be less pressure on physicians and patients because you are seeing each other more frequently; you can talk more openly about what’s working and what’s not.”
Dr. Chiesa Fuxench reported having no disclosures relevant to her presentation.
, according to Zelma Chiesa Fuxench, MD.
This gene codes for profilaggrin, a protein, which is then cleaved to form filaggrin, which helps to organize the cytoskeleton of the skin and is an important structural component of the skin. The understanding is that patients who have filaggrin mutations tend to have earlier onset and more persistent disease, Dr. Chiesa Fuxench, of the department of dermatology at the University of Pennsylvania, Philadelphia, said during the Revolutionizing Atopic Dermatitis virtual symposium.
“Prior studies have shown that mutations in the FLG gene can confer a risk of developed AD that is two- to sevenfold with variants R501X and the 22804del4 frequently described. It is important to note that most of these findings have been described primarily in populations of European descent, with other variants being found in populations of African nation descent, and seem to be more prevalent in populations with early onset disease.”
Environmental factors
Other AD-related risk factors that have been previously described in the literature include environmental factors such as climate, diet, breastfeeding, obesity, pollution, tobacco smoke, pet ownership, and microbiome or gut microflora. “The list of culprits is ever increasing,” she said. “However, it’s important to recognize that data to support some of these associations are lacking, and oftentimes, a lot of the results are contradictory.”
As part of the International Study of Asthma and Allergies in Childhood, researchers evaluated the association between climate factors with the 12-month period prevalence rates of symptoms of atopic eczema in children. They found that patients who lived at higher latitudes and those who lived in areas where there were lower mean outdoor temperatures tended to have a higher prevalence of eczema symptoms. Worldwide, they found that symptoms of eczema were also prevalent in areas where there was lower indoor humidity.
“The authors concluded that they can’t really demonstrate a cause and effect, and that while latitude and temperature changes appear to affect the prevalence of eczema, they may do so indirectly, perhaps to changes in behavior and differences in sun exposure,” said Dr. Chiesa Fuxench, who was not involved with the study. “For example, we know that vitamin D is a protective risk factor for AD. Low vitamin D has been associated with more severe disease in some studies. We also know that UV exposure leads to the conversion of filaggrin degradation products such as trans-urocanic acid into cis-urocanic acid, which has been demonstrated to have immunosuppressive effects.”
A systematic review and meta-analysis of nine articles found small associations, which were significant, between being born in the winter (odds ratio, 1.15) and fall (OR, 1.16) and the risk of developing AD, compared with being born in the spring and summer. However, an analysis of satellite-derived data on air temperature across the United States from 1993 to 2011 found that as ambient air temperature increases, so did the risk for an ambulatory visit for AD to physicians from the National Ambulatory Medical Care Survey.
In all areas but the south, the largest number of AD visits occur in the spring. In the south, more AD visits occur in the summer. “This raises the point that we don’t really know everything when it comes to the influence of temperature and climate change on AD,” Dr. Chiesa Fuxench said.
Several maternal and neonatal risk factors for AD have been described in the literature, including the effect of prenatal exposure to antibiotics. In one large analysis, investigators assessed the association among 18-month-old children in the Danish National Birth Cohort, which included 62,560 mother-child pairs. They found that prenatal antibiotic use was associated with an increased odds of AD among children born to atopic mothers but only when used during all three trimesters (adjusted OR, 1.45). When they further stratified these analyses by type of birth (vaginal versus C-section), the association persisted in both groups, but was stronger among those delivered by C-section.
Probiotics
The role of probiotics to reduce the risk for AD has also been investigated. “We do know that probiotics could potentially be helpful, and it is often a readily available intervention,” Dr. Chiesa Fuxench said. “But the question still is how and when to supplement.”
In a systematic review and meta-analysis, researchers examined supplementation with probiotics given to breastfeeding mothers, pregnant mothers, or directly given to infants, and the risk of developing AD up to 18 months of age. They found that overall, probiotic exposure resulted in decreased risk of developing AD. In stratified analyses, the strongest association was observed for those who received probiotics during their pregnancy, during breastfeeding, and as an infant, which conferred about a 25% reduced risk.
Antibiotic exposure
What about early-life exposure to antibiotics on one’s risk for developing AD? A meta-analysis of 22 studies found that children who had been exposed to antibiotics during the first 2 years of life had an increased risk of eczema (OR, 1.26), compared with children who had not been exposed during the same period of time. “Interesting hypotheses can be generated from this study,” she said. “Perhaps future steps should focus on the impact of antibiotic exposure, the gut microbiome, and maternal risk factors for AD.”
In a separate study that supported these findings, researchers evaluated the association between the use of acid-suppressive medications and antibiotics during infancy and the development of allergic disease in early childhood. They found that exposure to either of these medications during the first six months of infancy resulted in a mild increased risk of developing AD, and concluded that they should be used during infancy only in situations of clear clinical benefit. “We should be good stewards of antibiotic use, in particular due to concern for antibiotic resistance in the population overall,” Dr. Chiesa Fuxench said.
Prevention strategies
Several AD prevention strategies have also been described in the medical literature, including the use of daily emollients during infancy. In a multicenter trial carried out in the United Kingdom, researchers tested whether daily use of emollient in the first year of life could prevent eczema in high-risk children, which was defined as having at least one first-degree relative with parent-reported eczema, allergic rhinitis, or asthma. The primary outcome was eczema at age 2 years. The researchers found no evidence to suggest that daily emollient use during the first year of life prevents eczema.
Another study, the PreventADALL trial of 2,397 infants, consisted of four treatment arms: a control group advised to follow national guidelines on infant nutrition; a skin intervention group that was asked to use skin emollients, a food intervention group with early introduction of peanut, cow’s milk, wheat, and egg, and a combined skin and food intervention. The investigators found no difference in the risk reduction of developing AD among patients who were treated with skin emollients or early complementary feeding, and concluded that these types of interventions should not be considered as interventions to prevent AD in this cohort of patients.
However, Dr. Chiesa Fuxench emphasized that emollients and moisturizers are an important part of the treatment regimen for AD patients. A Cochrane systematic review of nearly 80 randomized, controlled trials evaluating the use of emollients in eczema found that most moisturizers showed some beneficial effects in addition to active treatment, including prolonging the time to flare, reducing the number of flares, and reducing the amount of topical corticosteroids used.
For treatment, Dr. Chiesa Fuxench recommends a proactive approach focused on short-term induction therapy with intensive topical anti-inflammatories until the affected area is almost healed, followed by maintenance therapy that involves use of a long-term, low- to mid-potency steroid or a topical calcineurin inhibitor to previously affected areas. “These interventions have been shown to decrease the risk of recurrence and can shorten the treatment duration in the event of a flare,” she said.
She also favors a time-contingent approach to treating patients with AD. “As physicians, we tend to do our visits more as symptom contingent, which means when a patient is flaring. This reinforces the view that this is a difficult disease to treat, and that there is no hope,” she pointed out. But for chronic diseases, she added, “a time-contingent approach with appointments at set intervals leads to a different perception. It can result in better compliance, because skin care might be performed more regularly. It’s analogous to when you know you’re going to see the dentist so you floss more regularly the week before your appointment. There also seems to be less pressure on physicians and patients because you are seeing each other more frequently; you can talk more openly about what’s working and what’s not.”
Dr. Chiesa Fuxench reported having no disclosures relevant to her presentation.
FROM REVOLUTIONIZING AD 2020
FPs need to remind patients they care for whole families
I think there are multiple factors explaining why the percentage of family physicians treating children declined again. Not the least of these is that pediatricians have a very limited scope of practice and need to market and attract patients, which they do quite a bit. There are even pediatric urgent care centers popping up all over the place now, some likely funded by venture capital just as other urgent care centers have been funded.
The loss of pediatric inpatient volume because of the effectiveness of vaccines that prevent many bacterial and viral illnesses means that fewer pediatric graduates are spending time in the hospital.
Family doctors used to retain their pediatric patients by delivering babies, seeing them in the newborn nursery, and beginning their relationship with the kids there. FPs are delivering fewer babies and the subsequent reduction in new kids in their practices has been a factor in this as well.
Finally, in multispecialty practices, pediatricians are employed there. Families immediately assume that their kids should be going to the pediatricians, not the family doctors. We need to keep talking up the fact that we take care of whole families to retain our pediatric practices.
Neil S. Calman, MD, is president and chief executive officer of the Institute for Family Health and is professor and chair of the Alfred and Gail Engelberg department of family medicine and community health at the Icahn School of Medicine at Mount Sinai and the Mount Sinai Health System, both in New York. Dr. Calman also serves on the editorial advisory board of Family Practice News.
I think there are multiple factors explaining why the percentage of family physicians treating children declined again. Not the least of these is that pediatricians have a very limited scope of practice and need to market and attract patients, which they do quite a bit. There are even pediatric urgent care centers popping up all over the place now, some likely funded by venture capital just as other urgent care centers have been funded.
The loss of pediatric inpatient volume because of the effectiveness of vaccines that prevent many bacterial and viral illnesses means that fewer pediatric graduates are spending time in the hospital.
Family doctors used to retain their pediatric patients by delivering babies, seeing them in the newborn nursery, and beginning their relationship with the kids there. FPs are delivering fewer babies and the subsequent reduction in new kids in their practices has been a factor in this as well.
Finally, in multispecialty practices, pediatricians are employed there. Families immediately assume that their kids should be going to the pediatricians, not the family doctors. We need to keep talking up the fact that we take care of whole families to retain our pediatric practices.
Neil S. Calman, MD, is president and chief executive officer of the Institute for Family Health and is professor and chair of the Alfred and Gail Engelberg department of family medicine and community health at the Icahn School of Medicine at Mount Sinai and the Mount Sinai Health System, both in New York. Dr. Calman also serves on the editorial advisory board of Family Practice News.
I think there are multiple factors explaining why the percentage of family physicians treating children declined again. Not the least of these is that pediatricians have a very limited scope of practice and need to market and attract patients, which they do quite a bit. There are even pediatric urgent care centers popping up all over the place now, some likely funded by venture capital just as other urgent care centers have been funded.
The loss of pediatric inpatient volume because of the effectiveness of vaccines that prevent many bacterial and viral illnesses means that fewer pediatric graduates are spending time in the hospital.
Family doctors used to retain their pediatric patients by delivering babies, seeing them in the newborn nursery, and beginning their relationship with the kids there. FPs are delivering fewer babies and the subsequent reduction in new kids in their practices has been a factor in this as well.
Finally, in multispecialty practices, pediatricians are employed there. Families immediately assume that their kids should be going to the pediatricians, not the family doctors. We need to keep talking up the fact that we take care of whole families to retain our pediatric practices.
Neil S. Calman, MD, is president and chief executive officer of the Institute for Family Health and is professor and chair of the Alfred and Gail Engelberg department of family medicine and community health at the Icahn School of Medicine at Mount Sinai and the Mount Sinai Health System, both in New York. Dr. Calman also serves on the editorial advisory board of Family Practice News.
Emerging treatments for molluscum contagiosum and acne show promise
, but that could soon change, according to Leon H. Kircik, MD.
“The treatment of molluscum is still an unmet need,” Dr. Kircik, clinical professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference. However, a proprietary drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, which has been tested in phase 3 studies, is currently under FDA review. The manufacturer, Verrica Pharmaceuticals resubmitted a new drug application for the product, VP-102, in December 2020.
“VP-102 features a visualization agent so the injector can see which lesions have been treated, as well as a bittering agent to mitigate oral ingestion by children. Complete clearance at 12 weeks ranged from 46% to 54% of patients, while lesion count reduction compared with baseline ranged from 69% to 82%.”
Acne
In August, 2020, clascoterone 1% cream was approved for the treatment of acne in patients 12 years and older, a development that Dr. Kircik said “can be a game changer in acne treatment.” Clascoterone cream 1% exhibits strong, selective anti-androgen activity by targeting androgen receptors in the skin, not systemically. “It limits or blocks transcription of androgen responsive genes, but it also has an anti-inflammatory effect and an anti-sebum effect,” he explained.
According to results from two phase 3 trials of the product, a response of clear or almost clear on the IGA scale at week 12 was achieved in 18.4% of those on treatment vs. 9% of those on vehicle in one study (P less than .001) and 20.3% vs. 6.5%, respectively, in the second study (P less than .001). Clascoterone is also being evaluated for treating androgenetic alopecia.
In Dr. Kircik’s clinical experience, retinoids can be helpful for patients with moderate to severe acne. “We always use them for anticomedogenic effects, but we also know that they have anti-inflammatory effects,” he said. “They actually inhibit toll-like receptor activity. They also inhibit the AP-1 pathway by causing a reduction in inflammatory signaling associated with collagen degradation and scarring.”
The most recent retinoid to be approved for the topical treatment of acne was 0.005% trifarotene cream, in 2019, for patients aged 9 years and older. “But when we got the results, it was not that exciting,” a difference of about 3.6 (mean) inflammatory lesion reduction between the active and the vehicle arm, said Dr. Kircik, medical director of Physicians Skin Care in Louisville, Ky. “According to the package insert, treatment side effects included mild to moderate erythema in 59% of patients, scaling in 65%, dryness in 69%, and stinging/burning in 56%, which makes it difficult to use in our clinical practice.”
The drug was also tested for treating truncal acne. However, one comparative study showed that tazarotene 0.045% lotion spread an average of 36.7 square centimeters farther than the trifarotene cream, which makes the tazarotene lotion easier to use on the chest and back, he said.
Dr. Kircik also discussed 4% minocycline, a hydrophobic, topical foam formulation of minocycline that was approved by the FDA in 2019 for the treatment of moderate to severe acne, for patients aged 9 and older. In a 12-week study that involved 1,488 patients (mean age was about 20 years), investigators observed a 56% reduction in inflammatory lesion count among those treated with minocycline 4%, compared with 43% in the vehicle group.
Dr. Kircik, one of the authors of the study, noted that the hydrophobic composition of minocycline 4% allows for stable and efficient delivery of an inherently unstable active pharmaceutical ingredient such as minocycline. “It’s free of primary irritants such as surfactants and short chain alcohols, which makes it much more tolerable,” he said. “The unique physical foam characteristics facilitate ease of application and absorption at target sites.”
Dr. Kircik reported that he serves as a consultant and/or adviser to numerous pharmaceutical companies, including Galderma, the manufacturer of trifarotene cream.
[email protected]
, but that could soon change, according to Leon H. Kircik, MD.
“The treatment of molluscum is still an unmet need,” Dr. Kircik, clinical professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference. However, a proprietary drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, which has been tested in phase 3 studies, is currently under FDA review. The manufacturer, Verrica Pharmaceuticals resubmitted a new drug application for the product, VP-102, in December 2020.
“VP-102 features a visualization agent so the injector can see which lesions have been treated, as well as a bittering agent to mitigate oral ingestion by children. Complete clearance at 12 weeks ranged from 46% to 54% of patients, while lesion count reduction compared with baseline ranged from 69% to 82%.”
Acne
In August, 2020, clascoterone 1% cream was approved for the treatment of acne in patients 12 years and older, a development that Dr. Kircik said “can be a game changer in acne treatment.” Clascoterone cream 1% exhibits strong, selective anti-androgen activity by targeting androgen receptors in the skin, not systemically. “It limits or blocks transcription of androgen responsive genes, but it also has an anti-inflammatory effect and an anti-sebum effect,” he explained.
According to results from two phase 3 trials of the product, a response of clear or almost clear on the IGA scale at week 12 was achieved in 18.4% of those on treatment vs. 9% of those on vehicle in one study (P less than .001) and 20.3% vs. 6.5%, respectively, in the second study (P less than .001). Clascoterone is also being evaluated for treating androgenetic alopecia.
In Dr. Kircik’s clinical experience, retinoids can be helpful for patients with moderate to severe acne. “We always use them for anticomedogenic effects, but we also know that they have anti-inflammatory effects,” he said. “They actually inhibit toll-like receptor activity. They also inhibit the AP-1 pathway by causing a reduction in inflammatory signaling associated with collagen degradation and scarring.”
The most recent retinoid to be approved for the topical treatment of acne was 0.005% trifarotene cream, in 2019, for patients aged 9 years and older. “But when we got the results, it was not that exciting,” a difference of about 3.6 (mean) inflammatory lesion reduction between the active and the vehicle arm, said Dr. Kircik, medical director of Physicians Skin Care in Louisville, Ky. “According to the package insert, treatment side effects included mild to moderate erythema in 59% of patients, scaling in 65%, dryness in 69%, and stinging/burning in 56%, which makes it difficult to use in our clinical practice.”
The drug was also tested for treating truncal acne. However, one comparative study showed that tazarotene 0.045% lotion spread an average of 36.7 square centimeters farther than the trifarotene cream, which makes the tazarotene lotion easier to use on the chest and back, he said.
Dr. Kircik also discussed 4% minocycline, a hydrophobic, topical foam formulation of minocycline that was approved by the FDA in 2019 for the treatment of moderate to severe acne, for patients aged 9 and older. In a 12-week study that involved 1,488 patients (mean age was about 20 years), investigators observed a 56% reduction in inflammatory lesion count among those treated with minocycline 4%, compared with 43% in the vehicle group.
Dr. Kircik, one of the authors of the study, noted that the hydrophobic composition of minocycline 4% allows for stable and efficient delivery of an inherently unstable active pharmaceutical ingredient such as minocycline. “It’s free of primary irritants such as surfactants and short chain alcohols, which makes it much more tolerable,” he said. “The unique physical foam characteristics facilitate ease of application and absorption at target sites.”
Dr. Kircik reported that he serves as a consultant and/or adviser to numerous pharmaceutical companies, including Galderma, the manufacturer of trifarotene cream.
[email protected]
, but that could soon change, according to Leon H. Kircik, MD.
“The treatment of molluscum is still an unmet need,” Dr. Kircik, clinical professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, said at the Orlando Dermatology Aesthetic and Clinical Conference. However, a proprietary drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, which has been tested in phase 3 studies, is currently under FDA review. The manufacturer, Verrica Pharmaceuticals resubmitted a new drug application for the product, VP-102, in December 2020.
“VP-102 features a visualization agent so the injector can see which lesions have been treated, as well as a bittering agent to mitigate oral ingestion by children. Complete clearance at 12 weeks ranged from 46% to 54% of patients, while lesion count reduction compared with baseline ranged from 69% to 82%.”
Acne
In August, 2020, clascoterone 1% cream was approved for the treatment of acne in patients 12 years and older, a development that Dr. Kircik said “can be a game changer in acne treatment.” Clascoterone cream 1% exhibits strong, selective anti-androgen activity by targeting androgen receptors in the skin, not systemically. “It limits or blocks transcription of androgen responsive genes, but it also has an anti-inflammatory effect and an anti-sebum effect,” he explained.
According to results from two phase 3 trials of the product, a response of clear or almost clear on the IGA scale at week 12 was achieved in 18.4% of those on treatment vs. 9% of those on vehicle in one study (P less than .001) and 20.3% vs. 6.5%, respectively, in the second study (P less than .001). Clascoterone is also being evaluated for treating androgenetic alopecia.
In Dr. Kircik’s clinical experience, retinoids can be helpful for patients with moderate to severe acne. “We always use them for anticomedogenic effects, but we also know that they have anti-inflammatory effects,” he said. “They actually inhibit toll-like receptor activity. They also inhibit the AP-1 pathway by causing a reduction in inflammatory signaling associated with collagen degradation and scarring.”
The most recent retinoid to be approved for the topical treatment of acne was 0.005% trifarotene cream, in 2019, for patients aged 9 years and older. “But when we got the results, it was not that exciting,” a difference of about 3.6 (mean) inflammatory lesion reduction between the active and the vehicle arm, said Dr. Kircik, medical director of Physicians Skin Care in Louisville, Ky. “According to the package insert, treatment side effects included mild to moderate erythema in 59% of patients, scaling in 65%, dryness in 69%, and stinging/burning in 56%, which makes it difficult to use in our clinical practice.”
The drug was also tested for treating truncal acne. However, one comparative study showed that tazarotene 0.045% lotion spread an average of 36.7 square centimeters farther than the trifarotene cream, which makes the tazarotene lotion easier to use on the chest and back, he said.
Dr. Kircik also discussed 4% minocycline, a hydrophobic, topical foam formulation of minocycline that was approved by the FDA in 2019 for the treatment of moderate to severe acne, for patients aged 9 and older. In a 12-week study that involved 1,488 patients (mean age was about 20 years), investigators observed a 56% reduction in inflammatory lesion count among those treated with minocycline 4%, compared with 43% in the vehicle group.
Dr. Kircik, one of the authors of the study, noted that the hydrophobic composition of minocycline 4% allows for stable and efficient delivery of an inherently unstable active pharmaceutical ingredient such as minocycline. “It’s free of primary irritants such as surfactants and short chain alcohols, which makes it much more tolerable,” he said. “The unique physical foam characteristics facilitate ease of application and absorption at target sites.”
Dr. Kircik reported that he serves as a consultant and/or adviser to numerous pharmaceutical companies, including Galderma, the manufacturer of trifarotene cream.
[email protected]
FROM ODAC 2021
Helping parents and children deal with a child’s limb deformity
After 15 years of limping and a gradual downhill slide in mobility, recreational walking had become uncomfortable enough that I’ve decided to shed my proudly worn cloak of denial and seek help. Even I could see that the x-ray made a total knee replacement the only option for some return to near normalcy. Scheduling a total knee replacement became a no-brainer.
My decision to accept the risks to reap the benefits of surgery is small potatoes compared with the decisions that the parents of a child born with a deformed lower extremity must face. In the Family Partnerships section of the February 2021 issue of Pediatrics you will find a heart-wrenching story of a family who embarked on what turned out to be painful and frustrating journey to lengthen their daughter’s congenitally deficient leg. In their own words, the mother and daughter describe how neither of them were prepared for the pain and life-altering complications the daughter has endured. Influenced by the optimism exuded by surgeons, the family gave little thought to the magnitude of the decision they were being asked to make. One has to wonder in retrospect if a well-timed amputation and prosthesis might have been a better decision. However, the thought of removing an extremity, even one that isn’t fully functional, is not one that most of us like to consider.
Over the last several decades I have read stories about people – usually athletes – born with short or deformed lower extremities who have faced the decision of amputation. I recall one college-age young man who despite his deformity and with the help of a prosthesis was a competitive multisport athlete. However, it became clear that his deformed foot was preventing him from accessing the most advanced prosthetic technology. Although he was highly motivated, he described his struggle with the decision to part with a portion of his body that despite its appearance and dysfunction had been with him since birth. On the other hand, I have read stories of young people who had become so frustrated by their deformity that they were more than eager to undergo amputation despite the concerns of their parents.
Early in my career I encountered a 3-year-old with phocomelia whose family was visiting from out of town and had come to our clinic because his older sibling was sick. The youngster, as I recall, had only one complete extremity, an arm. Like most 3-year-olds, he was driven to explore at breakneck speed. I will never forget watching him streak back and forth the length of our linoleum covered hallway like a crab skittering along the beach. His mother described how she and his well-meaning physicians were struggling unsuccessfully to get him to accept prostheses. Later I learned that his resistance is shared by many of the survivors of the thalidomide disaster who felt that the most frustrating period in their lives came when, again well-meaning, caregivers had tried to make them look and function more normally by fitting them with prostheses.
These anecdotal observations make clear a philosophy that we should have already internalized. In most clinic decisions the patient, pretty much regardless of age, should be a full participant in the process. And, to do this the patient and his or her family must be as informed as possible. Managing the aftermath of a traumatic amputation presents it own special set of challenges, but when it comes to elective amputation or prosthetic application for a congenital deficiency it is dangerous for us to insert our personal bias into the decision making.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
After 15 years of limping and a gradual downhill slide in mobility, recreational walking had become uncomfortable enough that I’ve decided to shed my proudly worn cloak of denial and seek help. Even I could see that the x-ray made a total knee replacement the only option for some return to near normalcy. Scheduling a total knee replacement became a no-brainer.
My decision to accept the risks to reap the benefits of surgery is small potatoes compared with the decisions that the parents of a child born with a deformed lower extremity must face. In the Family Partnerships section of the February 2021 issue of Pediatrics you will find a heart-wrenching story of a family who embarked on what turned out to be painful and frustrating journey to lengthen their daughter’s congenitally deficient leg. In their own words, the mother and daughter describe how neither of them were prepared for the pain and life-altering complications the daughter has endured. Influenced by the optimism exuded by surgeons, the family gave little thought to the magnitude of the decision they were being asked to make. One has to wonder in retrospect if a well-timed amputation and prosthesis might have been a better decision. However, the thought of removing an extremity, even one that isn’t fully functional, is not one that most of us like to consider.
Over the last several decades I have read stories about people – usually athletes – born with short or deformed lower extremities who have faced the decision of amputation. I recall one college-age young man who despite his deformity and with the help of a prosthesis was a competitive multisport athlete. However, it became clear that his deformed foot was preventing him from accessing the most advanced prosthetic technology. Although he was highly motivated, he described his struggle with the decision to part with a portion of his body that despite its appearance and dysfunction had been with him since birth. On the other hand, I have read stories of young people who had become so frustrated by their deformity that they were more than eager to undergo amputation despite the concerns of their parents.
Early in my career I encountered a 3-year-old with phocomelia whose family was visiting from out of town and had come to our clinic because his older sibling was sick. The youngster, as I recall, had only one complete extremity, an arm. Like most 3-year-olds, he was driven to explore at breakneck speed. I will never forget watching him streak back and forth the length of our linoleum covered hallway like a crab skittering along the beach. His mother described how she and his well-meaning physicians were struggling unsuccessfully to get him to accept prostheses. Later I learned that his resistance is shared by many of the survivors of the thalidomide disaster who felt that the most frustrating period in their lives came when, again well-meaning, caregivers had tried to make them look and function more normally by fitting them with prostheses.
These anecdotal observations make clear a philosophy that we should have already internalized. In most clinic decisions the patient, pretty much regardless of age, should be a full participant in the process. And, to do this the patient and his or her family must be as informed as possible. Managing the aftermath of a traumatic amputation presents it own special set of challenges, but when it comes to elective amputation or prosthetic application for a congenital deficiency it is dangerous for us to insert our personal bias into the decision making.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
After 15 years of limping and a gradual downhill slide in mobility, recreational walking had become uncomfortable enough that I’ve decided to shed my proudly worn cloak of denial and seek help. Even I could see that the x-ray made a total knee replacement the only option for some return to near normalcy. Scheduling a total knee replacement became a no-brainer.
My decision to accept the risks to reap the benefits of surgery is small potatoes compared with the decisions that the parents of a child born with a deformed lower extremity must face. In the Family Partnerships section of the February 2021 issue of Pediatrics you will find a heart-wrenching story of a family who embarked on what turned out to be painful and frustrating journey to lengthen their daughter’s congenitally deficient leg. In their own words, the mother and daughter describe how neither of them were prepared for the pain and life-altering complications the daughter has endured. Influenced by the optimism exuded by surgeons, the family gave little thought to the magnitude of the decision they were being asked to make. One has to wonder in retrospect if a well-timed amputation and prosthesis might have been a better decision. However, the thought of removing an extremity, even one that isn’t fully functional, is not one that most of us like to consider.
Over the last several decades I have read stories about people – usually athletes – born with short or deformed lower extremities who have faced the decision of amputation. I recall one college-age young man who despite his deformity and with the help of a prosthesis was a competitive multisport athlete. However, it became clear that his deformed foot was preventing him from accessing the most advanced prosthetic technology. Although he was highly motivated, he described his struggle with the decision to part with a portion of his body that despite its appearance and dysfunction had been with him since birth. On the other hand, I have read stories of young people who had become so frustrated by their deformity that they were more than eager to undergo amputation despite the concerns of their parents.
Early in my career I encountered a 3-year-old with phocomelia whose family was visiting from out of town and had come to our clinic because his older sibling was sick. The youngster, as I recall, had only one complete extremity, an arm. Like most 3-year-olds, he was driven to explore at breakneck speed. I will never forget watching him streak back and forth the length of our linoleum covered hallway like a crab skittering along the beach. His mother described how she and his well-meaning physicians were struggling unsuccessfully to get him to accept prostheses. Later I learned that his resistance is shared by many of the survivors of the thalidomide disaster who felt that the most frustrating period in their lives came when, again well-meaning, caregivers had tried to make them look and function more normally by fitting them with prostheses.
These anecdotal observations make clear a philosophy that we should have already internalized. In most clinic decisions the patient, pretty much regardless of age, should be a full participant in the process. And, to do this the patient and his or her family must be as informed as possible. Managing the aftermath of a traumatic amputation presents it own special set of challenges, but when it comes to elective amputation or prosthetic application for a congenital deficiency it is dangerous for us to insert our personal bias into the decision making.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Screening tool may help better predict suicide attempts in adolescents
Researchers have developed a proprietary computer adaptive screening tool that may help emergency departments more accurately predict suicide attempts in adolescents, according to a recent study in JAMA Psychiatry.
The computerized adaptive screen for suicidal youth (CASSY) had an area under the curve (AUC) of 0.87 in an independent validation cohort that predicted an adolescent suicide attempt within 3 months, according to Cheryl A. King, PhD, of the department of psychiatry at the University of Michigan in Ann Arbor, and colleagues. CASSY’s adaptive design, which presents different questions based on a respondent’s answers, means “an individual’s initial item responses are used to determine a provisional estimate of their standing on the measured trait,” the researchers said.
Dr. King and colleagues evaluated the CASSY algorithm in a first study that consisted of 2,845 adolescents who were mean 15.1 years old, mostly girls (63%) enrolled from 13 different emergency departments across the United States within the Pediatric Emergency Care Applied Research Network (PECARN) between June 2015 and July 2016. To develop the CASSY algorithm, the participants received a 92-item self-report survey at baseline with three “anchor” questions from the Ask Suicide-Screening Questions (ASQ) and Columbia–Suicide Severity Rating Scale (C-SSRS). Based on the answers to the baseline survey, the researchers categorized participants as being at low, medium, or high risk for a suicide attempt, and followed participants for 3 months to record suicide attempts reported by a patient or parent.
Retention of participants at 3 months was 72.9%, leaving data available for 2,075 adolescents for review. The researchers found that the AUC was 0.89 (95% confidence interval, 0.85-0.91) in the first study, with a sensitivity of 82.4% and a specificity of 80%. Participants answered a mean number of 11 items during an assessment (range, 5-21 items) administered in a median time of 1 minute, 24 seconds.
In a second study consisting of a validation cohort, 4,050 adolescents from 14 PECARN emergency departments and 1 Indian Health Service hospital were followed, with a retention of 2,754 participants (69.5%) at the end of 3 months. Of the adolescents available at the end of 3 months, 62.1% were girls with a mean age of 15.0 years. The AUC for this validation group was 0.87 (95% CI, 0.85-0.89). Of these participants, 71.5% reported no previous suicide attempts, 9% reported one prior attempt, 18.2% reported multiple attempts, and 1.2% had an unknown number of suicide attempts. During the 3-month window of the second study, 6.0% of participants had at least one suicide attempt.
The researchers said that while the CASSY instrument may be advantageous for some emergency departments, “a standard screen such as the ASQ, which consists of fewer items, may be preferred in some settings, particularly those in which the cost and technical setup of a computerized adaptive screen poses too high a barrier.”
Dr. King and colleagues concluded.
Climbing adolescent suicide rate
In an interview, Igor Galynker, MD, PhD, professor in the department of psychiatry, and director of the suicide lab and the Zirinsky Center for Bipolar Disorder at the Icahn School of Medicine at Mount Sinai, New York, said the study by Dr. King and colleagues is important during a time when the suicide rate for adolescents is substantially increasing.
According to data from the CDC’s Web-based Injury Statistics Query and Reporting System, 1,750 adolescents died of suicide in 2018, and the rate of deaths by suicide has increased by 62% since the year 2000. “The issue really needs to be addressed,” said Dr. Galynker, who was not involved with the study.
Some methods of screening suicidal ideation that open with a direct question can miss suicide attempts in individuals who do not express these suicidal ideations, he explained, and the problem can be magnified in adolescent patients. “This is particularly difficult with adolescents because they’re notoriously poor historians. They cannot describe their feelings as well. It’s even more important to have methods that work for suicide prevention for adolescents and to support those predictors which do not rely on self-report,” he said.
Dr. Galynker said that CASSY is innovative because asking whether the patient is suicidal is not the “gateway question” and does not categorize people into groups determined to be at low, medium, or high risk for a suicide attempt.
“When you categorize people – adolescents in this particular case – you remove clinical judgment from [the] clinician. You deprive [the] clinician of exercising their clinical judgment in terms of somebody is or is not likely to die by suicide. That’s a serious problem,” he said, noting it may be one reason why these screening tools have difficulty identifying patients at risk of suicide.
Regarding limitations, the 3-month follow-up window for patients in the study may be too long to be clinically meaningful.
“If somebody is in treatment, 3 months is a long time. You want to know whether somebody is going to attempt suicide before the next time you see them, which is usually a month or a week,” he said.
But a strength of the CASSY instrument, Dr. Galynker said, is its ability to capture the patient’s mental state in the moment, as opposed to relying only a patient’s electronic medical record. The study also demonstrates “it should be possible to introduce detailed suicide risk assessment in the emergency rooms, and [it] should be done,” he said.
This study was funded with support from the Health Resources and Services Administration, the Maternal and Child Health Bureau, and the Emergency Medical Services for Children Network Development Demonstration Program, and a grant by the National Institute of Mental Health for the Emergency Department Screen for Teens at Risk for Suicide. Twelve authors reported personal and institutional relationships in the form of fees, grants, consultancies, royalties, copyrighted work, founding of technologies, and scientific council memberships for a variety of agencies, societies, foundations, and other organizations inside and outside of the study. Dr. Galynker reported his work unrelated to the study is supported by the National Institute of Mental Health and the American Foundation for Suicide Prevention. But he has no proprietary interests.
Researchers have developed a proprietary computer adaptive screening tool that may help emergency departments more accurately predict suicide attempts in adolescents, according to a recent study in JAMA Psychiatry.
The computerized adaptive screen for suicidal youth (CASSY) had an area under the curve (AUC) of 0.87 in an independent validation cohort that predicted an adolescent suicide attempt within 3 months, according to Cheryl A. King, PhD, of the department of psychiatry at the University of Michigan in Ann Arbor, and colleagues. CASSY’s adaptive design, which presents different questions based on a respondent’s answers, means “an individual’s initial item responses are used to determine a provisional estimate of their standing on the measured trait,” the researchers said.
Dr. King and colleagues evaluated the CASSY algorithm in a first study that consisted of 2,845 adolescents who were mean 15.1 years old, mostly girls (63%) enrolled from 13 different emergency departments across the United States within the Pediatric Emergency Care Applied Research Network (PECARN) between June 2015 and July 2016. To develop the CASSY algorithm, the participants received a 92-item self-report survey at baseline with three “anchor” questions from the Ask Suicide-Screening Questions (ASQ) and Columbia–Suicide Severity Rating Scale (C-SSRS). Based on the answers to the baseline survey, the researchers categorized participants as being at low, medium, or high risk for a suicide attempt, and followed participants for 3 months to record suicide attempts reported by a patient or parent.
Retention of participants at 3 months was 72.9%, leaving data available for 2,075 adolescents for review. The researchers found that the AUC was 0.89 (95% confidence interval, 0.85-0.91) in the first study, with a sensitivity of 82.4% and a specificity of 80%. Participants answered a mean number of 11 items during an assessment (range, 5-21 items) administered in a median time of 1 minute, 24 seconds.
In a second study consisting of a validation cohort, 4,050 adolescents from 14 PECARN emergency departments and 1 Indian Health Service hospital were followed, with a retention of 2,754 participants (69.5%) at the end of 3 months. Of the adolescents available at the end of 3 months, 62.1% were girls with a mean age of 15.0 years. The AUC for this validation group was 0.87 (95% CI, 0.85-0.89). Of these participants, 71.5% reported no previous suicide attempts, 9% reported one prior attempt, 18.2% reported multiple attempts, and 1.2% had an unknown number of suicide attempts. During the 3-month window of the second study, 6.0% of participants had at least one suicide attempt.
The researchers said that while the CASSY instrument may be advantageous for some emergency departments, “a standard screen such as the ASQ, which consists of fewer items, may be preferred in some settings, particularly those in which the cost and technical setup of a computerized adaptive screen poses too high a barrier.”
Dr. King and colleagues concluded.
Climbing adolescent suicide rate
In an interview, Igor Galynker, MD, PhD, professor in the department of psychiatry, and director of the suicide lab and the Zirinsky Center for Bipolar Disorder at the Icahn School of Medicine at Mount Sinai, New York, said the study by Dr. King and colleagues is important during a time when the suicide rate for adolescents is substantially increasing.
According to data from the CDC’s Web-based Injury Statistics Query and Reporting System, 1,750 adolescents died of suicide in 2018, and the rate of deaths by suicide has increased by 62% since the year 2000. “The issue really needs to be addressed,” said Dr. Galynker, who was not involved with the study.
Some methods of screening suicidal ideation that open with a direct question can miss suicide attempts in individuals who do not express these suicidal ideations, he explained, and the problem can be magnified in adolescent patients. “This is particularly difficult with adolescents because they’re notoriously poor historians. They cannot describe their feelings as well. It’s even more important to have methods that work for suicide prevention for adolescents and to support those predictors which do not rely on self-report,” he said.
Dr. Galynker said that CASSY is innovative because asking whether the patient is suicidal is not the “gateway question” and does not categorize people into groups determined to be at low, medium, or high risk for a suicide attempt.
“When you categorize people – adolescents in this particular case – you remove clinical judgment from [the] clinician. You deprive [the] clinician of exercising their clinical judgment in terms of somebody is or is not likely to die by suicide. That’s a serious problem,” he said, noting it may be one reason why these screening tools have difficulty identifying patients at risk of suicide.
Regarding limitations, the 3-month follow-up window for patients in the study may be too long to be clinically meaningful.
“If somebody is in treatment, 3 months is a long time. You want to know whether somebody is going to attempt suicide before the next time you see them, which is usually a month or a week,” he said.
But a strength of the CASSY instrument, Dr. Galynker said, is its ability to capture the patient’s mental state in the moment, as opposed to relying only a patient’s electronic medical record. The study also demonstrates “it should be possible to introduce detailed suicide risk assessment in the emergency rooms, and [it] should be done,” he said.
This study was funded with support from the Health Resources and Services Administration, the Maternal and Child Health Bureau, and the Emergency Medical Services for Children Network Development Demonstration Program, and a grant by the National Institute of Mental Health for the Emergency Department Screen for Teens at Risk for Suicide. Twelve authors reported personal and institutional relationships in the form of fees, grants, consultancies, royalties, copyrighted work, founding of technologies, and scientific council memberships for a variety of agencies, societies, foundations, and other organizations inside and outside of the study. Dr. Galynker reported his work unrelated to the study is supported by the National Institute of Mental Health and the American Foundation for Suicide Prevention. But he has no proprietary interests.
Researchers have developed a proprietary computer adaptive screening tool that may help emergency departments more accurately predict suicide attempts in adolescents, according to a recent study in JAMA Psychiatry.
The computerized adaptive screen for suicidal youth (CASSY) had an area under the curve (AUC) of 0.87 in an independent validation cohort that predicted an adolescent suicide attempt within 3 months, according to Cheryl A. King, PhD, of the department of psychiatry at the University of Michigan in Ann Arbor, and colleagues. CASSY’s adaptive design, which presents different questions based on a respondent’s answers, means “an individual’s initial item responses are used to determine a provisional estimate of their standing on the measured trait,” the researchers said.
Dr. King and colleagues evaluated the CASSY algorithm in a first study that consisted of 2,845 adolescents who were mean 15.1 years old, mostly girls (63%) enrolled from 13 different emergency departments across the United States within the Pediatric Emergency Care Applied Research Network (PECARN) between June 2015 and July 2016. To develop the CASSY algorithm, the participants received a 92-item self-report survey at baseline with three “anchor” questions from the Ask Suicide-Screening Questions (ASQ) and Columbia–Suicide Severity Rating Scale (C-SSRS). Based on the answers to the baseline survey, the researchers categorized participants as being at low, medium, or high risk for a suicide attempt, and followed participants for 3 months to record suicide attempts reported by a patient or parent.
Retention of participants at 3 months was 72.9%, leaving data available for 2,075 adolescents for review. The researchers found that the AUC was 0.89 (95% confidence interval, 0.85-0.91) in the first study, with a sensitivity of 82.4% and a specificity of 80%. Participants answered a mean number of 11 items during an assessment (range, 5-21 items) administered in a median time of 1 minute, 24 seconds.
In a second study consisting of a validation cohort, 4,050 adolescents from 14 PECARN emergency departments and 1 Indian Health Service hospital were followed, with a retention of 2,754 participants (69.5%) at the end of 3 months. Of the adolescents available at the end of 3 months, 62.1% were girls with a mean age of 15.0 years. The AUC for this validation group was 0.87 (95% CI, 0.85-0.89). Of these participants, 71.5% reported no previous suicide attempts, 9% reported one prior attempt, 18.2% reported multiple attempts, and 1.2% had an unknown number of suicide attempts. During the 3-month window of the second study, 6.0% of participants had at least one suicide attempt.
The researchers said that while the CASSY instrument may be advantageous for some emergency departments, “a standard screen such as the ASQ, which consists of fewer items, may be preferred in some settings, particularly those in which the cost and technical setup of a computerized adaptive screen poses too high a barrier.”
Dr. King and colleagues concluded.
Climbing adolescent suicide rate
In an interview, Igor Galynker, MD, PhD, professor in the department of psychiatry, and director of the suicide lab and the Zirinsky Center for Bipolar Disorder at the Icahn School of Medicine at Mount Sinai, New York, said the study by Dr. King and colleagues is important during a time when the suicide rate for adolescents is substantially increasing.
According to data from the CDC’s Web-based Injury Statistics Query and Reporting System, 1,750 adolescents died of suicide in 2018, and the rate of deaths by suicide has increased by 62% since the year 2000. “The issue really needs to be addressed,” said Dr. Galynker, who was not involved with the study.
Some methods of screening suicidal ideation that open with a direct question can miss suicide attempts in individuals who do not express these suicidal ideations, he explained, and the problem can be magnified in adolescent patients. “This is particularly difficult with adolescents because they’re notoriously poor historians. They cannot describe their feelings as well. It’s even more important to have methods that work for suicide prevention for adolescents and to support those predictors which do not rely on self-report,” he said.
Dr. Galynker said that CASSY is innovative because asking whether the patient is suicidal is not the “gateway question” and does not categorize people into groups determined to be at low, medium, or high risk for a suicide attempt.
“When you categorize people – adolescents in this particular case – you remove clinical judgment from [the] clinician. You deprive [the] clinician of exercising their clinical judgment in terms of somebody is or is not likely to die by suicide. That’s a serious problem,” he said, noting it may be one reason why these screening tools have difficulty identifying patients at risk of suicide.
Regarding limitations, the 3-month follow-up window for patients in the study may be too long to be clinically meaningful.
“If somebody is in treatment, 3 months is a long time. You want to know whether somebody is going to attempt suicide before the next time you see them, which is usually a month or a week,” he said.
But a strength of the CASSY instrument, Dr. Galynker said, is its ability to capture the patient’s mental state in the moment, as opposed to relying only a patient’s electronic medical record. The study also demonstrates “it should be possible to introduce detailed suicide risk assessment in the emergency rooms, and [it] should be done,” he said.
This study was funded with support from the Health Resources and Services Administration, the Maternal and Child Health Bureau, and the Emergency Medical Services for Children Network Development Demonstration Program, and a grant by the National Institute of Mental Health for the Emergency Department Screen for Teens at Risk for Suicide. Twelve authors reported personal and institutional relationships in the form of fees, grants, consultancies, royalties, copyrighted work, founding of technologies, and scientific council memberships for a variety of agencies, societies, foundations, and other organizations inside and outside of the study. Dr. Galynker reported his work unrelated to the study is supported by the National Institute of Mental Health and the American Foundation for Suicide Prevention. But he has no proprietary interests.
FROM JAMA PSYCHIATRY