User login
Flu vaccines cut seasonal death in heart failure patients
WASHINGTON – Patients with heart failure who received an annual influenza vaccine for 3 years running had significantly fewer all-cause hospitalizations and significantly fewer cases of pneumonia during that time, compared with placebo-treated patients with heart failure, in a prospective, randomized, global trial with 5,129 participants.
Although the results failed to show a significant reduction in all-cause deaths linked to influenza vaccination, compared with controls during the entire 3 years of the study, the results did show a significant 21% relative mortality-risk reduction by vaccination during periods of peak influenza circulation, and a significant 23% reduction in cardiovascular deaths, compared with controls during peak seasons.
“This is the first randomized, controlled trial of influenza vaccine in patients with heart failure, and we showed that vaccination reduces deaths” during peak influenza seasons, Mark Loeb, MD, said during a press briefing at the annual scientific sessions of the American College of Cardiology. The results send “an important global message that patients with heart failure should receive the influenza vaccine,” said Dr. Loeb, a professor at McMaster University, Hamilton, Ont., who specializes in clinical epidemiology and infectious diseases.
Dr. Loeb admitted that he and his associates erred when they picked the time window to assess the two primary endpoints for the trial: the combined rate of cardiovascular death, nonfatal MI, and nonfatal stroke, and this combined endpoint plus hospitalizations for heart failure.
The time window they selected was the entirety of all 3 years following three annual immunizations. That was a mistake.
No flu vaccine benefit outside flu season
“We know that the influenza vaccine will not have any effect outside of when influenza is circulating. In retrospect, we should have done that,” Dr. Loeb bemoaned during his talk. He chalked up the bad choice to concern over collecting enough endpoints to see a significant between-group difference when the researchers designed the study.
For the entire 3 years of follow-up, influenza vaccination was tied to a nonsignificant 7% relative risk reduction for the first primary endpoint, and a nonsignificant 9% relative risk reduction for the second primary endpoint, he reported.
But Dr. Loeb lobbied for the relevance of several significant secondary endpoints that collectively showed a compelling pattern of benefit during his talk. These included, for the full 3-years of follow-up, important, significant reductions relative to placebo of 16% for first all-cause hospitalizations (P = .01), and a 42% relative risk reduction in first cases of pneumonia (P = .0006).
Then there were the benefits that appeared during influenza season. In that analysis, first events for the first primary endpoint fell after vaccination by a significant 18% relative to placebo. The in-season analysis also showed the significant cuts in both all-cause and cardiovascular deaths.
Despite the neutral primary endpoints, “if you look at these data as a whole I think they speak to the importance of vaccinating patients with heart failure against influenza,” Dr. Loeb maintained.
‘Totality of evidence supports vaccination’
“I agree that the totality of evidence supports influenza vaccination,” commented Mark H. Drazner, MD, professor and clinical chief of cardiology at the University of Texas Southwestern Medical Center, Dallas, who was designated discussant for the report.
“The message should be to offer influenza vaccine to patients with heart failure,” Dr. Drazner said in an interview. “Previous data on influenza vaccine in patients with heart failure were largely observational. This was a randomized, prospective, placebo-controlled trial. That’s a step forward. Proving efficacy in a randomized trial is important.”
Dr Drazner added that his institution already promotes a “strong mandate” to vaccinate patients with heart failure against influenza.
“The influenza vaccine is a very effective and cost-efficient public health measure. Preventing hospitalizations of patients with heart failure has so many benefits,” commented Craig Beavers, PharmD, vice president of professional services at Baptist Health in Paducah, Ky., and a discussant during the press briefing.
The Influenza Vaccine To Prevent Adverse Vascular Events (IVVE) trial enrolled people with heart failure in New York Heart Association functional class II, III, or IV from any of 10 low- and middle-income countries including China, India, the Philippines, and multiple countries from Africa and the Middle East. They averaged 57 years of age, and slightly more than half were women.
IVVE was sponsored by McMaster University; the only commercial support that IVVE received was a free supply of influenza vaccine from Sanofi Pasteur. Dr. Loeb, Dr. Drazner, and Dr. Beavers had no disclosures.
WASHINGTON – Patients with heart failure who received an annual influenza vaccine for 3 years running had significantly fewer all-cause hospitalizations and significantly fewer cases of pneumonia during that time, compared with placebo-treated patients with heart failure, in a prospective, randomized, global trial with 5,129 participants.
Although the results failed to show a significant reduction in all-cause deaths linked to influenza vaccination, compared with controls during the entire 3 years of the study, the results did show a significant 21% relative mortality-risk reduction by vaccination during periods of peak influenza circulation, and a significant 23% reduction in cardiovascular deaths, compared with controls during peak seasons.
“This is the first randomized, controlled trial of influenza vaccine in patients with heart failure, and we showed that vaccination reduces deaths” during peak influenza seasons, Mark Loeb, MD, said during a press briefing at the annual scientific sessions of the American College of Cardiology. The results send “an important global message that patients with heart failure should receive the influenza vaccine,” said Dr. Loeb, a professor at McMaster University, Hamilton, Ont., who specializes in clinical epidemiology and infectious diseases.
Dr. Loeb admitted that he and his associates erred when they picked the time window to assess the two primary endpoints for the trial: the combined rate of cardiovascular death, nonfatal MI, and nonfatal stroke, and this combined endpoint plus hospitalizations for heart failure.
The time window they selected was the entirety of all 3 years following three annual immunizations. That was a mistake.
No flu vaccine benefit outside flu season
“We know that the influenza vaccine will not have any effect outside of when influenza is circulating. In retrospect, we should have done that,” Dr. Loeb bemoaned during his talk. He chalked up the bad choice to concern over collecting enough endpoints to see a significant between-group difference when the researchers designed the study.
For the entire 3 years of follow-up, influenza vaccination was tied to a nonsignificant 7% relative risk reduction for the first primary endpoint, and a nonsignificant 9% relative risk reduction for the second primary endpoint, he reported.
But Dr. Loeb lobbied for the relevance of several significant secondary endpoints that collectively showed a compelling pattern of benefit during his talk. These included, for the full 3-years of follow-up, important, significant reductions relative to placebo of 16% for first all-cause hospitalizations (P = .01), and a 42% relative risk reduction in first cases of pneumonia (P = .0006).
Then there were the benefits that appeared during influenza season. In that analysis, first events for the first primary endpoint fell after vaccination by a significant 18% relative to placebo. The in-season analysis also showed the significant cuts in both all-cause and cardiovascular deaths.
Despite the neutral primary endpoints, “if you look at these data as a whole I think they speak to the importance of vaccinating patients with heart failure against influenza,” Dr. Loeb maintained.
‘Totality of evidence supports vaccination’
“I agree that the totality of evidence supports influenza vaccination,” commented Mark H. Drazner, MD, professor and clinical chief of cardiology at the University of Texas Southwestern Medical Center, Dallas, who was designated discussant for the report.
“The message should be to offer influenza vaccine to patients with heart failure,” Dr. Drazner said in an interview. “Previous data on influenza vaccine in patients with heart failure were largely observational. This was a randomized, prospective, placebo-controlled trial. That’s a step forward. Proving efficacy in a randomized trial is important.”
Dr Drazner added that his institution already promotes a “strong mandate” to vaccinate patients with heart failure against influenza.
“The influenza vaccine is a very effective and cost-efficient public health measure. Preventing hospitalizations of patients with heart failure has so many benefits,” commented Craig Beavers, PharmD, vice president of professional services at Baptist Health in Paducah, Ky., and a discussant during the press briefing.
The Influenza Vaccine To Prevent Adverse Vascular Events (IVVE) trial enrolled people with heart failure in New York Heart Association functional class II, III, or IV from any of 10 low- and middle-income countries including China, India, the Philippines, and multiple countries from Africa and the Middle East. They averaged 57 years of age, and slightly more than half were women.
IVVE was sponsored by McMaster University; the only commercial support that IVVE received was a free supply of influenza vaccine from Sanofi Pasteur. Dr. Loeb, Dr. Drazner, and Dr. Beavers had no disclosures.
WASHINGTON – Patients with heart failure who received an annual influenza vaccine for 3 years running had significantly fewer all-cause hospitalizations and significantly fewer cases of pneumonia during that time, compared with placebo-treated patients with heart failure, in a prospective, randomized, global trial with 5,129 participants.
Although the results failed to show a significant reduction in all-cause deaths linked to influenza vaccination, compared with controls during the entire 3 years of the study, the results did show a significant 21% relative mortality-risk reduction by vaccination during periods of peak influenza circulation, and a significant 23% reduction in cardiovascular deaths, compared with controls during peak seasons.
“This is the first randomized, controlled trial of influenza vaccine in patients with heart failure, and we showed that vaccination reduces deaths” during peak influenza seasons, Mark Loeb, MD, said during a press briefing at the annual scientific sessions of the American College of Cardiology. The results send “an important global message that patients with heart failure should receive the influenza vaccine,” said Dr. Loeb, a professor at McMaster University, Hamilton, Ont., who specializes in clinical epidemiology and infectious diseases.
Dr. Loeb admitted that he and his associates erred when they picked the time window to assess the two primary endpoints for the trial: the combined rate of cardiovascular death, nonfatal MI, and nonfatal stroke, and this combined endpoint plus hospitalizations for heart failure.
The time window they selected was the entirety of all 3 years following three annual immunizations. That was a mistake.
No flu vaccine benefit outside flu season
“We know that the influenza vaccine will not have any effect outside of when influenza is circulating. In retrospect, we should have done that,” Dr. Loeb bemoaned during his talk. He chalked up the bad choice to concern over collecting enough endpoints to see a significant between-group difference when the researchers designed the study.
For the entire 3 years of follow-up, influenza vaccination was tied to a nonsignificant 7% relative risk reduction for the first primary endpoint, and a nonsignificant 9% relative risk reduction for the second primary endpoint, he reported.
But Dr. Loeb lobbied for the relevance of several significant secondary endpoints that collectively showed a compelling pattern of benefit during his talk. These included, for the full 3-years of follow-up, important, significant reductions relative to placebo of 16% for first all-cause hospitalizations (P = .01), and a 42% relative risk reduction in first cases of pneumonia (P = .0006).
Then there were the benefits that appeared during influenza season. In that analysis, first events for the first primary endpoint fell after vaccination by a significant 18% relative to placebo. The in-season analysis also showed the significant cuts in both all-cause and cardiovascular deaths.
Despite the neutral primary endpoints, “if you look at these data as a whole I think they speak to the importance of vaccinating patients with heart failure against influenza,” Dr. Loeb maintained.
‘Totality of evidence supports vaccination’
“I agree that the totality of evidence supports influenza vaccination,” commented Mark H. Drazner, MD, professor and clinical chief of cardiology at the University of Texas Southwestern Medical Center, Dallas, who was designated discussant for the report.
“The message should be to offer influenza vaccine to patients with heart failure,” Dr. Drazner said in an interview. “Previous data on influenza vaccine in patients with heart failure were largely observational. This was a randomized, prospective, placebo-controlled trial. That’s a step forward. Proving efficacy in a randomized trial is important.”
Dr Drazner added that his institution already promotes a “strong mandate” to vaccinate patients with heart failure against influenza.
“The influenza vaccine is a very effective and cost-efficient public health measure. Preventing hospitalizations of patients with heart failure has so many benefits,” commented Craig Beavers, PharmD, vice president of professional services at Baptist Health in Paducah, Ky., and a discussant during the press briefing.
The Influenza Vaccine To Prevent Adverse Vascular Events (IVVE) trial enrolled people with heart failure in New York Heart Association functional class II, III, or IV from any of 10 low- and middle-income countries including China, India, the Philippines, and multiple countries from Africa and the Middle East. They averaged 57 years of age, and slightly more than half were women.
IVVE was sponsored by McMaster University; the only commercial support that IVVE received was a free supply of influenza vaccine from Sanofi Pasteur. Dr. Loeb, Dr. Drazner, and Dr. Beavers had no disclosures.
AT ACC 2022
Pneumococcal pneumonia outcomes worse than those of Legionnaires disease
Outcomes for patients with bacteremic Streptococcus pneumoniae were significantly worse than those for patients with Legionnaires disease (LD), based on data from 106 individuals.
Reported cases of LD in the United States have increased in recent decades, but they are likely under-reported, wrote Sima Salahie, MD, of Wayne State University School of Medicine, Detroit, and Central Michigan University College of Medicine, Grosse Pointe Woods, and colleagues.
Clinical presentations may be similar for both conditions, but different antimicrobial therapies are needed; therefore, identifying distinguishing factors can promote better management of hospitalized patients, they reported.
In a retrospective case companion study published in the American Journal of the Medical Sciences, the researchers reviewed data from 51 adults with LD and 55 with bacteremic S. pneumoniae pneumonia (SP) who were hospitalized at a single center between 2013 and 2018. Diagnoses were confirmed by laboratory and radiology results. In addition, data were collected on clinical features including body mass index, systolic and diastolic blood pressure, pulse, respiratory rate, and temperature.
Overall, patients with SP were significantly more likely than those with LD to require mechanical ventilation (P = .04), intensive care unit stay (P = .004), and to die (P = .002). Patients with SP also had higher rates of septic shock compared to LD patients, although this difference fell short of statistical significance (49.1% vs. 30.4%; P = .06).
In a multivariate analysis, male sex, diarrhea, higher body mass index, hyponatremia, and lower Charleston Weighted Index of Comorbidity (CWIC) score were significant independent predictors of LD, with odds ratios of 21.6, 4.5, 1.13, 5.6, and 0.61, respectively.
The incidence of LD peaked in summer, while the incidence of SP peaked in the winter, the researchers noted. “Seasonality is a variable that has not always been included in previous scoring systems but should be considered in future modeling,” they said.
“Noteworthy is that LD represented almost as many cases as documented bacteremic pneumococcal pneumonia,” the researchers wrote in their discussion. “This occurred at a time when there was no outbreak of L. pneumophila in our community, and as these were all community acquired, there was no evidence of a nosocomial outbreak in our institution,” they said.
The study findings were limited by several factors, including the possible underestimation of SP because of the requirement for positive blood cultures and the lack of other methods of diagnosing SP, the researchers noted.
“However, the data suggest variables to distinguish LD from SP,” they said. “Establishing reliable clinical and laboratory parameters embedded in a simple diagnostic score that can accurately identify patients with LD may be helpful in aiding physicians’ early diagnosis in distinguishing LD from SP but will need to be defined.”
The study received no outside funding. The researchers disclosed no financial conflicts.
A version of this article first appeared on Medscape.com.
Outcomes for patients with bacteremic Streptococcus pneumoniae were significantly worse than those for patients with Legionnaires disease (LD), based on data from 106 individuals.
Reported cases of LD in the United States have increased in recent decades, but they are likely under-reported, wrote Sima Salahie, MD, of Wayne State University School of Medicine, Detroit, and Central Michigan University College of Medicine, Grosse Pointe Woods, and colleagues.
Clinical presentations may be similar for both conditions, but different antimicrobial therapies are needed; therefore, identifying distinguishing factors can promote better management of hospitalized patients, they reported.
In a retrospective case companion study published in the American Journal of the Medical Sciences, the researchers reviewed data from 51 adults with LD and 55 with bacteremic S. pneumoniae pneumonia (SP) who were hospitalized at a single center between 2013 and 2018. Diagnoses were confirmed by laboratory and radiology results. In addition, data were collected on clinical features including body mass index, systolic and diastolic blood pressure, pulse, respiratory rate, and temperature.
Overall, patients with SP were significantly more likely than those with LD to require mechanical ventilation (P = .04), intensive care unit stay (P = .004), and to die (P = .002). Patients with SP also had higher rates of septic shock compared to LD patients, although this difference fell short of statistical significance (49.1% vs. 30.4%; P = .06).
In a multivariate analysis, male sex, diarrhea, higher body mass index, hyponatremia, and lower Charleston Weighted Index of Comorbidity (CWIC) score were significant independent predictors of LD, with odds ratios of 21.6, 4.5, 1.13, 5.6, and 0.61, respectively.
The incidence of LD peaked in summer, while the incidence of SP peaked in the winter, the researchers noted. “Seasonality is a variable that has not always been included in previous scoring systems but should be considered in future modeling,” they said.
“Noteworthy is that LD represented almost as many cases as documented bacteremic pneumococcal pneumonia,” the researchers wrote in their discussion. “This occurred at a time when there was no outbreak of L. pneumophila in our community, and as these were all community acquired, there was no evidence of a nosocomial outbreak in our institution,” they said.
The study findings were limited by several factors, including the possible underestimation of SP because of the requirement for positive blood cultures and the lack of other methods of diagnosing SP, the researchers noted.
“However, the data suggest variables to distinguish LD from SP,” they said. “Establishing reliable clinical and laboratory parameters embedded in a simple diagnostic score that can accurately identify patients with LD may be helpful in aiding physicians’ early diagnosis in distinguishing LD from SP but will need to be defined.”
The study received no outside funding. The researchers disclosed no financial conflicts.
A version of this article first appeared on Medscape.com.
Outcomes for patients with bacteremic Streptococcus pneumoniae were significantly worse than those for patients with Legionnaires disease (LD), based on data from 106 individuals.
Reported cases of LD in the United States have increased in recent decades, but they are likely under-reported, wrote Sima Salahie, MD, of Wayne State University School of Medicine, Detroit, and Central Michigan University College of Medicine, Grosse Pointe Woods, and colleagues.
Clinical presentations may be similar for both conditions, but different antimicrobial therapies are needed; therefore, identifying distinguishing factors can promote better management of hospitalized patients, they reported.
In a retrospective case companion study published in the American Journal of the Medical Sciences, the researchers reviewed data from 51 adults with LD and 55 with bacteremic S. pneumoniae pneumonia (SP) who were hospitalized at a single center between 2013 and 2018. Diagnoses were confirmed by laboratory and radiology results. In addition, data were collected on clinical features including body mass index, systolic and diastolic blood pressure, pulse, respiratory rate, and temperature.
Overall, patients with SP were significantly more likely than those with LD to require mechanical ventilation (P = .04), intensive care unit stay (P = .004), and to die (P = .002). Patients with SP also had higher rates of septic shock compared to LD patients, although this difference fell short of statistical significance (49.1% vs. 30.4%; P = .06).
In a multivariate analysis, male sex, diarrhea, higher body mass index, hyponatremia, and lower Charleston Weighted Index of Comorbidity (CWIC) score were significant independent predictors of LD, with odds ratios of 21.6, 4.5, 1.13, 5.6, and 0.61, respectively.
The incidence of LD peaked in summer, while the incidence of SP peaked in the winter, the researchers noted. “Seasonality is a variable that has not always been included in previous scoring systems but should be considered in future modeling,” they said.
“Noteworthy is that LD represented almost as many cases as documented bacteremic pneumococcal pneumonia,” the researchers wrote in their discussion. “This occurred at a time when there was no outbreak of L. pneumophila in our community, and as these were all community acquired, there was no evidence of a nosocomial outbreak in our institution,” they said.
The study findings were limited by several factors, including the possible underestimation of SP because of the requirement for positive blood cultures and the lack of other methods of diagnosing SP, the researchers noted.
“However, the data suggest variables to distinguish LD from SP,” they said. “Establishing reliable clinical and laboratory parameters embedded in a simple diagnostic score that can accurately identify patients with LD may be helpful in aiding physicians’ early diagnosis in distinguishing LD from SP but will need to be defined.”
The study received no outside funding. The researchers disclosed no financial conflicts.
A version of this article first appeared on Medscape.com.
Pneumonia decision tool reduces death in ED patients
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
a 3-year, pragmatic, cluster-controlled study shows.
“We designed the ePNa specifically to require minimal input from the clinician so everything it does is already in the electronic medical record,” Nathan Dean, MD, University of Utah, Salt Lake City, told this news organization.
“So it’s actually putting the guideline recommendations into effect for physicians so that they can make better decisions by having all this information – it’s a comprehensive best practice kind of tool where best practices are likely to make the biggest difference for patients with a high severity of illness,” he added.
The study was published online in the American Journal of Respiratory and Critical Care Medicine.
Guideline-based tool
The ePNa makes use of pneumonia guidelines of 2007 and 2019 from the American Thoracic Society/Infectious Disease Society of America. The system was deployed into six geographic clusters of 16 Intermountain hospital EDs at 2-month intervals between December 2017 and November 2018. Simultaneous deployment was impractical, as implementation of the tool takes education, monitoring, and feedback that can be facilitated by focusing on only a few hospitals at a time.
The decision support tool gathers key patient indicators including age, fever, oxygen saturation, vital signs, and laboratory and chest imaging results to offer recommendations on care, including appropriate antibiotic therapy, microbiology studies, and whether a given patient should be sent to the intensive care unit, admitted to hospital, or may safely be discharged home.
Investigators analyzed a total of 6,848 patients, of whom 4,536 were managed for pneumonia before the ePNa was deployed and 2,312 after deployment.
The median age of patients was 67 years (interquartile range, 50-79 years). Roughly half were female and almost all were White. “Observed 30-day all-cause mortality including both outpatients and inpatients was 8.6% before deployment versus 4.8% after deployment of ePNa,” Dr. Dean and colleagues reported.
Adjusted for severity of illness, the odds ratio for lower mortality post-ePNa launch was 0.62 (95% confidence interval, 0.49-0.79; P < .0010) “and lower morality was consistent across hospital clusters.”
Compared with patients who were discharged home, reductions in mortality were greatest in patients who were directly admitted to ICUs from the ED (OR, 0.32; 95% CI, 0.14-0.77; P = .01). The OR for patients admitted to the medical floor was 0.53 (95% CI, 0.25-1.1; P = .09), which did not reach statistical significance.
Dr. Dean explained that the reductions in mortality were seen among those with the most severe illness, in whom best practices would benefit the most. In contrast, patients who are sent home on an antibiotic are at low risk for mortality while patients admitted to the medical floor may well have another, more lethal illness from which they end up dying, rather than simple pneumonia.
“For me, this was a clear demonstration that these best practices made the biggest difference in patients who were sick and who did not have any underlying disease that was going to kill them anyway,” he emphasized. On the other hand, both 30-day mortality and 7-day secondary hospital admission were higher among patients the tool recommended for hospital ward admission but who were discharged home from the ED.
“This was an unexpected finding,” Dr. Dean observed. However, as he explained, the authors reviewed 25% of randomly selected patients who fell into this subgroup and discovered that the ePNa tool was used in only about 20% of patients – “so doctors did not use the tool in the majority of this group.”
In addition, some of these patients declined hospital admission, so the doctors may have recommended that they be admitted but the patients said no. “The hypothesis here is that if they had been admitted to the hospital, they may have had a lower mortality risk,” Dr. Dean said.
Noticeable changes
Another noticeable change following the introduction of the ePNa tool was that guideline-concordant antibiotic prescribing increased in the 8 hours after patients presented to the ED, from 79.5% prior to the tool’s launch to 87.9%, again after adjusting for pneumonia severity (P < .001). Use of broad-spectrum antibiotics was not significantly different between the two treatment intervals, but administration of antibiotics active against methicillin-resistant Staphylococcus aureus dropped significantly between the two treatment intervals (P < .001). And the mean time from admission to the ED to the first antibiotic taken was slightly faster, improving from 159.4 minutes (95% CI, 156.9-161.9 minutes) prior to the ePNa launch to 150.9 minutes (95% CI, 144.1-157.8) post deployment (P < .001).
“Overall outpatient disposition for treatment of pneumonia from the emergency department increased from 29.2% before ePNa to 46.9% [post ePNA],” the authors noted, while a similar increase was observed in patients for whom ePNA recommended outpatient care – from 49.2% pre-ePNA to 66.6% after ePNA.
Both hospital ward admission and admission to the ICU decreased after ePNa had been introduced. Despite a significant increase in the percentage of patients being discharged home, neither 7-day secondary hospital admission nor severity-adjusted, 30-day mortality were significantly different before versus after the introduction of ePNa, the authors stressed.
A limitation of the study was that the trial was confined to a single health care system in one region of the United States with a patient population that may differ from that in other regions.
Reason for its success
Asked to comment on the findings, Adam Balls, MD, emergency department chair, Intermountain Medical Center, Murray, Utah, suggested that the reason the ePNa tool has been so successful at improving care for pneumonia patients is that it puts the guidelines directly into the hands of individual providers and tells them what’s going on. (Dr. Balls was not involved in the study.) “The tool allows us to take into consideration various clinical features – a patient’s oxygen requirements and whether or not they had prior complicated pneumonias that required additional antibiotics, for example – and then it makes the best determination for not only the disposition for that patient but antibiotic treatment as well,” he said in an interview.
This then allows physicians to either appropriately discharge less severely ill patients and admit those who are more ill – “and in general, just do a better job of treating pneumonia with this tool,” Dr. Balls said. He himself uses the decision support tool when attending to his own patients with pneumonia, as he feels that the tool really does make his care of these patients better. “There is a disparity around how we treat pneumonia in the U.S.
“Clinicians sometimes have a bias or a preference for certain antibiotics and we may not be appropriately treating these patients with broad-spectrum antibiotics or are perhaps using antibiotics that are not as effective based on an individual patient scenario so this is definitely a user-friendly tool that hopefully can be deployed throughout other health care systems to improve the treatment of pneumonia overall,” Dr. Balls emphasized.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Mild COVID-19 infection linked to later type 2 diabetes
People who recover from a mild case of COVID-19 appear to have an increased risk for subsequent new-onset type 2 diabetes but not other types of diabetes, new data suggest.
“If confirmed, the results of the present study indicate that diabetes screening in individuals who have recovered from even mild COVID-19 should be recommended,” say Wolfgang Rathmann, MD, of the Leibniz Center for Diabetes Research at Heinrich Heine University, Düsseldorf, Germany, and colleagues.
The findings, from a nationwide primary care database in Germany, were recently published in Diabetologia.
These primary care data align with those from other studies of more seriously ill patients with COVID-19 that found increased rates of type 2 diabetes diagnoses in the subsequent months following illness, they point out.
“COVID-19 infection may lead to diabetes by upregulation of the immune system after remission, which may induce pancreatic beta-cell dysfunction and insulin resistance, or patients may have been at risk for developing diabetes due to having obesity or prediabetes, and the stress COVID-19 put on their bodies sped it up,” said Dr. Rathmann in a press release.
However, because the patients with COVID-19 in the study were only followed for about 3 months, “further follow-up is needed to understand whether type 2 diabetes after mild COVID-19 is just temporary and can be reversed after they have fully recovered or whether it leads to a chronic condition,” he noted.
Increase in type 2 diabetes 3 months after mild COVID-19
The retrospective cohort analysis was performed using data from the Disease Analyzer, a representative panel of 1,171 physician practices in Germany, from March 2020 to January 2021, with follow-up through July 2021.
Individuals with a history of COVID-19 or diabetes and those taking corticosteroids within 30 days after the index dates were excluded.
A total of 35,865 patients with confirmed SARS-CoV-2 infection were propensity score-matched on a one-to-one basis for sex, age, health insurance, and comorbidities with those who had acute respiratory tract infections (controls) but were COVID-19 negative. Median follow-up was 119 days for the COVID-19 group and 161 days for controls.
There was a 28% increased risk of type 2 diabetes for those who had COVID-19 versus controls (15.8 per 1,000 person-years vs. 12.3 per 1,000 person-years, respectively, which was significantly different, and an incidence rate ratio of 1.28).
The incidence of other types of diabetes or unspecified diabetes for the COVID-19 and control groups did not differ significantly (4.3 per 1,000 person-years vs. 3.7 per 1,000 person-years; IRR, 1.17).
Similar findings were seen in sensitivity analyses by glucose-lowering medication prescriptions and by ICD-10 codes.
Although type 2 diabetes is not likely to be a problem for the vast majority of people who have mild COVID-19, the authors recommend that anyone who has recovered from COVID-19 be aware of the warning signs and symptoms such as fatigue, frequent urination, and increased thirst, and seek treatment right away.
CoviDiab registry tracking type 1 and type 2 diabetes
Over the course of the pandemic, there have been conflicting data on whether COVID-19 induces or reveals a propensity for type 1 and type 2 diabetes.
The CoviDiab global registry is tracking this and will include diabetes type for adults and children.
The aim is to have “as many as possible cases of new-onset diabetes for which we can have also a minimum set of clinical data including type of diabetes and A1c,” coprincipal investigator Francesco Rubino, MD, of King’s College London, previously told this news organization.
“By looking at this information we can infer whether a role of COVID-19 in triggering diabetes is clinically plausible – or not – and what type of diabetes is most frequently associated with COVID-19.”
Rubino said that the CoviDiab team is approaching the data with the assumption that, at least in adults diagnosed with type 2 diabetes, the explanation might be that the person already had undiagnosed diabetes or the hyperglycemia may be stress-induced and temporary.
The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Culture and Science of the State of North Rhine-Westphalia. Dr. Rathmann has reported receiving consulting fees for attending educational sessions or advisory boards for AstraZeneca, Boehringer Ingelheim, and Novo Nordisk and institutional research grants from Novo Nordisk outside of the topic of the current work.
A version of this article first appeared on Medscape.com.
People who recover from a mild case of COVID-19 appear to have an increased risk for subsequent new-onset type 2 diabetes but not other types of diabetes, new data suggest.
“If confirmed, the results of the present study indicate that diabetes screening in individuals who have recovered from even mild COVID-19 should be recommended,” say Wolfgang Rathmann, MD, of the Leibniz Center for Diabetes Research at Heinrich Heine University, Düsseldorf, Germany, and colleagues.
The findings, from a nationwide primary care database in Germany, were recently published in Diabetologia.
These primary care data align with those from other studies of more seriously ill patients with COVID-19 that found increased rates of type 2 diabetes diagnoses in the subsequent months following illness, they point out.
“COVID-19 infection may lead to diabetes by upregulation of the immune system after remission, which may induce pancreatic beta-cell dysfunction and insulin resistance, or patients may have been at risk for developing diabetes due to having obesity or prediabetes, and the stress COVID-19 put on their bodies sped it up,” said Dr. Rathmann in a press release.
However, because the patients with COVID-19 in the study were only followed for about 3 months, “further follow-up is needed to understand whether type 2 diabetes after mild COVID-19 is just temporary and can be reversed after they have fully recovered or whether it leads to a chronic condition,” he noted.
Increase in type 2 diabetes 3 months after mild COVID-19
The retrospective cohort analysis was performed using data from the Disease Analyzer, a representative panel of 1,171 physician practices in Germany, from March 2020 to January 2021, with follow-up through July 2021.
Individuals with a history of COVID-19 or diabetes and those taking corticosteroids within 30 days after the index dates were excluded.
A total of 35,865 patients with confirmed SARS-CoV-2 infection were propensity score-matched on a one-to-one basis for sex, age, health insurance, and comorbidities with those who had acute respiratory tract infections (controls) but were COVID-19 negative. Median follow-up was 119 days for the COVID-19 group and 161 days for controls.
There was a 28% increased risk of type 2 diabetes for those who had COVID-19 versus controls (15.8 per 1,000 person-years vs. 12.3 per 1,000 person-years, respectively, which was significantly different, and an incidence rate ratio of 1.28).
The incidence of other types of diabetes or unspecified diabetes for the COVID-19 and control groups did not differ significantly (4.3 per 1,000 person-years vs. 3.7 per 1,000 person-years; IRR, 1.17).
Similar findings were seen in sensitivity analyses by glucose-lowering medication prescriptions and by ICD-10 codes.
Although type 2 diabetes is not likely to be a problem for the vast majority of people who have mild COVID-19, the authors recommend that anyone who has recovered from COVID-19 be aware of the warning signs and symptoms such as fatigue, frequent urination, and increased thirst, and seek treatment right away.
CoviDiab registry tracking type 1 and type 2 diabetes
Over the course of the pandemic, there have been conflicting data on whether COVID-19 induces or reveals a propensity for type 1 and type 2 diabetes.
The CoviDiab global registry is tracking this and will include diabetes type for adults and children.
The aim is to have “as many as possible cases of new-onset diabetes for which we can have also a minimum set of clinical data including type of diabetes and A1c,” coprincipal investigator Francesco Rubino, MD, of King’s College London, previously told this news organization.
“By looking at this information we can infer whether a role of COVID-19 in triggering diabetes is clinically plausible – or not – and what type of diabetes is most frequently associated with COVID-19.”
Rubino said that the CoviDiab team is approaching the data with the assumption that, at least in adults diagnosed with type 2 diabetes, the explanation might be that the person already had undiagnosed diabetes or the hyperglycemia may be stress-induced and temporary.
The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Culture and Science of the State of North Rhine-Westphalia. Dr. Rathmann has reported receiving consulting fees for attending educational sessions or advisory boards for AstraZeneca, Boehringer Ingelheim, and Novo Nordisk and institutional research grants from Novo Nordisk outside of the topic of the current work.
A version of this article first appeared on Medscape.com.
People who recover from a mild case of COVID-19 appear to have an increased risk for subsequent new-onset type 2 diabetes but not other types of diabetes, new data suggest.
“If confirmed, the results of the present study indicate that diabetes screening in individuals who have recovered from even mild COVID-19 should be recommended,” say Wolfgang Rathmann, MD, of the Leibniz Center for Diabetes Research at Heinrich Heine University, Düsseldorf, Germany, and colleagues.
The findings, from a nationwide primary care database in Germany, were recently published in Diabetologia.
These primary care data align with those from other studies of more seriously ill patients with COVID-19 that found increased rates of type 2 diabetes diagnoses in the subsequent months following illness, they point out.
“COVID-19 infection may lead to diabetes by upregulation of the immune system after remission, which may induce pancreatic beta-cell dysfunction and insulin resistance, or patients may have been at risk for developing diabetes due to having obesity or prediabetes, and the stress COVID-19 put on their bodies sped it up,” said Dr. Rathmann in a press release.
However, because the patients with COVID-19 in the study were only followed for about 3 months, “further follow-up is needed to understand whether type 2 diabetes after mild COVID-19 is just temporary and can be reversed after they have fully recovered or whether it leads to a chronic condition,” he noted.
Increase in type 2 diabetes 3 months after mild COVID-19
The retrospective cohort analysis was performed using data from the Disease Analyzer, a representative panel of 1,171 physician practices in Germany, from March 2020 to January 2021, with follow-up through July 2021.
Individuals with a history of COVID-19 or diabetes and those taking corticosteroids within 30 days after the index dates were excluded.
A total of 35,865 patients with confirmed SARS-CoV-2 infection were propensity score-matched on a one-to-one basis for sex, age, health insurance, and comorbidities with those who had acute respiratory tract infections (controls) but were COVID-19 negative. Median follow-up was 119 days for the COVID-19 group and 161 days for controls.
There was a 28% increased risk of type 2 diabetes for those who had COVID-19 versus controls (15.8 per 1,000 person-years vs. 12.3 per 1,000 person-years, respectively, which was significantly different, and an incidence rate ratio of 1.28).
The incidence of other types of diabetes or unspecified diabetes for the COVID-19 and control groups did not differ significantly (4.3 per 1,000 person-years vs. 3.7 per 1,000 person-years; IRR, 1.17).
Similar findings were seen in sensitivity analyses by glucose-lowering medication prescriptions and by ICD-10 codes.
Although type 2 diabetes is not likely to be a problem for the vast majority of people who have mild COVID-19, the authors recommend that anyone who has recovered from COVID-19 be aware of the warning signs and symptoms such as fatigue, frequent urination, and increased thirst, and seek treatment right away.
CoviDiab registry tracking type 1 and type 2 diabetes
Over the course of the pandemic, there have been conflicting data on whether COVID-19 induces or reveals a propensity for type 1 and type 2 diabetes.
The CoviDiab global registry is tracking this and will include diabetes type for adults and children.
The aim is to have “as many as possible cases of new-onset diabetes for which we can have also a minimum set of clinical data including type of diabetes and A1c,” coprincipal investigator Francesco Rubino, MD, of King’s College London, previously told this news organization.
“By looking at this information we can infer whether a role of COVID-19 in triggering diabetes is clinically plausible – or not – and what type of diabetes is most frequently associated with COVID-19.”
Rubino said that the CoviDiab team is approaching the data with the assumption that, at least in adults diagnosed with type 2 diabetes, the explanation might be that the person already had undiagnosed diabetes or the hyperglycemia may be stress-induced and temporary.
The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Culture and Science of the State of North Rhine-Westphalia. Dr. Rathmann has reported receiving consulting fees for attending educational sessions or advisory boards for AstraZeneca, Boehringer Ingelheim, and Novo Nordisk and institutional research grants from Novo Nordisk outside of the topic of the current work.
A version of this article first appeared on Medscape.com.
FROM DIABETOLOGIA
WHO issues new TB guidelines for children and adolescents
The World Health Organization now recommends shortened treatment for children with mild tuberculosis, as well as two oral TB treatments (bedaquiline and delamanid) for use in children of all ages. The updated guidelines for TB management in children and adolescents were announced March 21 ahead of World Tuberculosis Day on March 24.
The agency also called for increased investment in global TB programs, noting that in 2020, TB deaths increased for the first time in over a decade. “We cannot falter in our commitment to reach and save every man, woman, child, family, and community impacted by this deadly disease,” said Tereza Kasaeva, MD, PhD, director of the WHO Global Tuberculosis Programme during a press conference.
TB is the 13th-leading cause of death and the second top infectious killer after COVID-19, with more than 4,100 people dying from TB every day. WHO estimates that 1.1 million children fall ill with TB each year.
Calls for investment
The increase in TB deaths from 1.4 million in 2019 to 1.5 million in 2020 was coupled with a decrease in funding. From 2019-2020, global spending for TB diagnostic, treatment, and prevention services fell from $5.8 billion to $5.3 billion. This is less than half of the $13 billion target funding amount for 2022, Dr. Kasaeva said.
Efforts to expand access to TB care have fallen short mainly because of this lack of funding, especially for children. In 2020, about 63% of children under 15 years of age with TB either did not receive or were not reported to have access to TB diagnosis and treatment services, which rose to 72% in children under age 5. Almost two-thirds of children under age 5 also did not receive TB preventive treatment in 2022, according to WHO statistics.
The socioeconomic ramifications of the COVID-19 pandemic as well as ongoing conflict in Eastern Europe, Africa, and the Middle East have “further exacerbated the situation,” Dr. Kasaeva said. “This conveys the urgent need to dramatically increase investments to ramp up the fight against TB and achieve commitments to end TB made by global leaders.”
Dr. Kasaeva laid out WHO’s main points for global investment in TB care:
- Increase domestic and international funding to close gaps in TB research and program implementation. For countries with smaller economies, increased international investment will be necessary in the short or medium term to help regain progress.
- Double funding for TB research, including vaccines.
- Invest in sustaining TB programs and services during the COVID-19 pandemic and ongoing crises so care is not disrupted.
New guidelines
Dr. Kasaeva also noted that adoption of WHO’s new guidelines for children and adolescents should be fast-tracked to improve access to and quality of care. The updates include:
- Rapid molecular tests called Xpert Ultra should be used as the initial test for TB in children and adolescents.
- Diagnostic testing can now include noninvasive specimens, like stool samples.
- Children with mild TB can be treated with a , rather than 6 months. This shortened regimen will allow children to return to school faster and save money for families and the health care system, said Kerri Viney, MD, PhD, a team lead for the WHO Tuberculosis Programme, with a focus on vulnerable populations, including children. She presented the new guidelines during the WHO press conference.
- The recommended treatment regimen for TB meningitis has also been shortened from 12 to 6 months.
Two oral medications for drug-resistant TB (bedaquiline and delamanid) are now recommended for use in children of all ages. “There is no longer a need for painful injections that can have serious side effects, including deafness,” Dr. Viney said.
Health systems should develop new models of decentralized and integrated TB care to bring TB care closer to where children live.
The guidelines are available on the WHO website.
“The WHO guidelines issued today are a game changer for children and adolescents with TB,” Dr. Kasaeva said. The next step is assisting countries in implementing these updates so that children and adolescents globally have access to high quality TB care,” Dr. Viney added. “We have the policy recommendations. We have the implementation guidance, we have child-friendly formulations of TB medicines,” she said. “Let us not wait any longer. Let us invest to end TB in children and adolescents.”
A version of this article first appeared on Medscape.com.
The World Health Organization now recommends shortened treatment for children with mild tuberculosis, as well as two oral TB treatments (bedaquiline and delamanid) for use in children of all ages. The updated guidelines for TB management in children and adolescents were announced March 21 ahead of World Tuberculosis Day on March 24.
The agency also called for increased investment in global TB programs, noting that in 2020, TB deaths increased for the first time in over a decade. “We cannot falter in our commitment to reach and save every man, woman, child, family, and community impacted by this deadly disease,” said Tereza Kasaeva, MD, PhD, director of the WHO Global Tuberculosis Programme during a press conference.
TB is the 13th-leading cause of death and the second top infectious killer after COVID-19, with more than 4,100 people dying from TB every day. WHO estimates that 1.1 million children fall ill with TB each year.
Calls for investment
The increase in TB deaths from 1.4 million in 2019 to 1.5 million in 2020 was coupled with a decrease in funding. From 2019-2020, global spending for TB diagnostic, treatment, and prevention services fell from $5.8 billion to $5.3 billion. This is less than half of the $13 billion target funding amount for 2022, Dr. Kasaeva said.
Efforts to expand access to TB care have fallen short mainly because of this lack of funding, especially for children. In 2020, about 63% of children under 15 years of age with TB either did not receive or were not reported to have access to TB diagnosis and treatment services, which rose to 72% in children under age 5. Almost two-thirds of children under age 5 also did not receive TB preventive treatment in 2022, according to WHO statistics.
The socioeconomic ramifications of the COVID-19 pandemic as well as ongoing conflict in Eastern Europe, Africa, and the Middle East have “further exacerbated the situation,” Dr. Kasaeva said. “This conveys the urgent need to dramatically increase investments to ramp up the fight against TB and achieve commitments to end TB made by global leaders.”
Dr. Kasaeva laid out WHO’s main points for global investment in TB care:
- Increase domestic and international funding to close gaps in TB research and program implementation. For countries with smaller economies, increased international investment will be necessary in the short or medium term to help regain progress.
- Double funding for TB research, including vaccines.
- Invest in sustaining TB programs and services during the COVID-19 pandemic and ongoing crises so care is not disrupted.
New guidelines
Dr. Kasaeva also noted that adoption of WHO’s new guidelines for children and adolescents should be fast-tracked to improve access to and quality of care. The updates include:
- Rapid molecular tests called Xpert Ultra should be used as the initial test for TB in children and adolescents.
- Diagnostic testing can now include noninvasive specimens, like stool samples.
- Children with mild TB can be treated with a , rather than 6 months. This shortened regimen will allow children to return to school faster and save money for families and the health care system, said Kerri Viney, MD, PhD, a team lead for the WHO Tuberculosis Programme, with a focus on vulnerable populations, including children. She presented the new guidelines during the WHO press conference.
- The recommended treatment regimen for TB meningitis has also been shortened from 12 to 6 months.
Two oral medications for drug-resistant TB (bedaquiline and delamanid) are now recommended for use in children of all ages. “There is no longer a need for painful injections that can have serious side effects, including deafness,” Dr. Viney said.
Health systems should develop new models of decentralized and integrated TB care to bring TB care closer to where children live.
The guidelines are available on the WHO website.
“The WHO guidelines issued today are a game changer for children and adolescents with TB,” Dr. Kasaeva said. The next step is assisting countries in implementing these updates so that children and adolescents globally have access to high quality TB care,” Dr. Viney added. “We have the policy recommendations. We have the implementation guidance, we have child-friendly formulations of TB medicines,” she said. “Let us not wait any longer. Let us invest to end TB in children and adolescents.”
A version of this article first appeared on Medscape.com.
The World Health Organization now recommends shortened treatment for children with mild tuberculosis, as well as two oral TB treatments (bedaquiline and delamanid) for use in children of all ages. The updated guidelines for TB management in children and adolescents were announced March 21 ahead of World Tuberculosis Day on March 24.
The agency also called for increased investment in global TB programs, noting that in 2020, TB deaths increased for the first time in over a decade. “We cannot falter in our commitment to reach and save every man, woman, child, family, and community impacted by this deadly disease,” said Tereza Kasaeva, MD, PhD, director of the WHO Global Tuberculosis Programme during a press conference.
TB is the 13th-leading cause of death and the second top infectious killer after COVID-19, with more than 4,100 people dying from TB every day. WHO estimates that 1.1 million children fall ill with TB each year.
Calls for investment
The increase in TB deaths from 1.4 million in 2019 to 1.5 million in 2020 was coupled with a decrease in funding. From 2019-2020, global spending for TB diagnostic, treatment, and prevention services fell from $5.8 billion to $5.3 billion. This is less than half of the $13 billion target funding amount for 2022, Dr. Kasaeva said.
Efforts to expand access to TB care have fallen short mainly because of this lack of funding, especially for children. In 2020, about 63% of children under 15 years of age with TB either did not receive or were not reported to have access to TB diagnosis and treatment services, which rose to 72% in children under age 5. Almost two-thirds of children under age 5 also did not receive TB preventive treatment in 2022, according to WHO statistics.
The socioeconomic ramifications of the COVID-19 pandemic as well as ongoing conflict in Eastern Europe, Africa, and the Middle East have “further exacerbated the situation,” Dr. Kasaeva said. “This conveys the urgent need to dramatically increase investments to ramp up the fight against TB and achieve commitments to end TB made by global leaders.”
Dr. Kasaeva laid out WHO’s main points for global investment in TB care:
- Increase domestic and international funding to close gaps in TB research and program implementation. For countries with smaller economies, increased international investment will be necessary in the short or medium term to help regain progress.
- Double funding for TB research, including vaccines.
- Invest in sustaining TB programs and services during the COVID-19 pandemic and ongoing crises so care is not disrupted.
New guidelines
Dr. Kasaeva also noted that adoption of WHO’s new guidelines for children and adolescents should be fast-tracked to improve access to and quality of care. The updates include:
- Rapid molecular tests called Xpert Ultra should be used as the initial test for TB in children and adolescents.
- Diagnostic testing can now include noninvasive specimens, like stool samples.
- Children with mild TB can be treated with a , rather than 6 months. This shortened regimen will allow children to return to school faster and save money for families and the health care system, said Kerri Viney, MD, PhD, a team lead for the WHO Tuberculosis Programme, with a focus on vulnerable populations, including children. She presented the new guidelines during the WHO press conference.
- The recommended treatment regimen for TB meningitis has also been shortened from 12 to 6 months.
Two oral medications for drug-resistant TB (bedaquiline and delamanid) are now recommended for use in children of all ages. “There is no longer a need for painful injections that can have serious side effects, including deafness,” Dr. Viney said.
Health systems should develop new models of decentralized and integrated TB care to bring TB care closer to where children live.
The guidelines are available on the WHO website.
“The WHO guidelines issued today are a game changer for children and adolescents with TB,” Dr. Kasaeva said. The next step is assisting countries in implementing these updates so that children and adolescents globally have access to high quality TB care,” Dr. Viney added. “We have the policy recommendations. We have the implementation guidance, we have child-friendly formulations of TB medicines,” she said. “Let us not wait any longer. Let us invest to end TB in children and adolescents.”
A version of this article first appeared on Medscape.com.
Oxygen Therapies and Clinical Outcomes for Patients Hospitalized With COVID-19: First Surge vs Second Surge
From Lahey Hospital and Medical Center, Burlington, MA (Drs. Liesching and Lei), and Tufts University School of Medicine, Boston, MA (Dr. Liesching)
ABSTRACT
Objective: To compare the utilization of oxygen therapies and clinical outcomes of patients admitted for COVID-19 during the second surge of the pandemic to that of patients admitted during the first surge.
Design: Observational study using a registry database.
Setting: Three hospitals (791 inpatient beds and 76 intensive care unit [ICU] beds) within the Beth Israel Lahey Health system in Massachusetts.
Participants: We included 3183 patients with COVID-19 admitted to hospitals.
Measurements: Baseline data included demographics and comorbidities. Treatments included low-flow supplemental oxygen (2-6 L/min), high-flow oxygen via nasal cannula, and invasive mechanical ventilation. Outcomes included ICU admission, length of stay, ventilator days, and mortality.
Results: A total of 3183 patients were included: 1586 during the first surge and 1597 during the second surge. Compared to the first surge, patients admitted during the second surge had a similar rate of receiving low-flow supplemental oxygen (65.8% vs 64.1%, P = .3), a higher rate of receiving high-flow nasal cannula (15.4% vs 10.8%, P = .0001), and a lower ventilation rate (5.6% vs 9.7%, P < .0001). The outcomes during the second surge were better than those during the first surge: lower ICU admission rate (8.1% vs 12.7%, P < .0001), shorter length of hospital stay (5 vs 6 days, P < .0001), fewer ventilator days (10 vs 16, P = .01), and lower mortality (8.3% vs 19.2%, P < .0001). Among ventilated patients, those who received high-flow nasal cannula had lower mortality.
Conclusion: Compared to the first surge of the COVID-19 pandemic, patients admitted during the second surge had similar likelihood of receiving low-flow supplemental oxygen, were more likely to receive high-flow nasal cannula, were less likely to be ventilated, and had better outcomes.
Keywords: supplemental oxygen, high-flow nasal cannula, ventilator.
The respiratory system receives the major impact of SARS-CoV-2 virus, and hypoxemia has been the predominant diagnosis for patients hospitalized with COVID-19.1,2 During the initial stage of the pandemic, oxygen therapies and mechanical ventilation were the only choices for these patients.3-6 Standard-of-care treatment for patients with COVID-19 during the initial surge included oxygen therapies and mechanical ventilation for hypoxemia and medications for comorbidities and COVID-19–associated sequelae, such as multi-organ dysfunction and failure. A report from New York during the first surge (May 2020) showed that among 5700 hospitalized patients with COVID-19, 27.8% received supplemental oxygen and 12.2% received invasive mechanical ventilation.7 High-flow nasal cannula (HFNC) oxygen delivery has been utilized widely throughout the pandemic due to its superiority over other noninvasive respiratory support techniques.8-12 Mechanical ventilation is always necessary for critically ill patients with acute respiratory distress syndrome. However, ventilator scarcity has become a bottleneck in caring for severely ill patients with COVID-19 during the pandemic.13
The clinical outcomes of hospitalized COVID-19 patients include a high intubation rate, long length of hospital and intensive care unit (ICU) stay, and high mortality.14,15 As the pandemic evolved, new medications, including remdesivir, hydroxychloroquine, lopinavir, or interferon β-1a, were used in addition to the standard of care, but these did not result in significantly different mortality from standard of care.16 Steroids are becoming foundational to the treatment of severe COVID-19 pneumonia, but evidence from high-quality randomized controlled clinical trials is lacking.17
During the first surge from March to May 2020, Massachusetts had the third highest number of COVID-19 cases among states in the United States.18 In early 2021, COVID-19 cases were climbing close to the peak of the second surge in Massachusetts. In this study, we compared utilization of low-flow supplemental oxygen, HFNC, and mechanical ventilation and clinical outcomes of patients admitted to 3 hospitals in Massachusetts during the second surge of the pandemic to that of patients admitted during the first surge.
Methods
Setting
Beth Israel Lahey Health is a system of academic and teaching hospitals with primary care and specialty care providers. We included 3 centers within the Beth Israel Lahey Health system in Massachusetts: Lahey Hospital and Medical Center, with 335 inpatient hospital beds and 52 critical care beds; Beverly Hospital, with 227 beds and 14 critical care beds; and Winchester Hospital, with 229 beds and 10 ICU beds.
Participants
We included patients admitted to the 3 hospitals with COVID-19 as a primary or secondary diagnosis during the first surge of the pandemic (March 1, 2020 to June 15, 2020) and the second surge (November 15, 2020 to January 27, 2021). The timeframe of the first surge was defined as the window between the start date and the end date of data collection. During the time window of the first surge, 1586 patients were included. The start time of the second surge was defined as the date when the data collection was restarted; the end date was set when the number of patients (1597) accumulated was close to the number of patients in the first surge (1586), so that the two groups had similar sample size.
Study Design
A data registry of COVID-19 patients was created by our institution, and the data were prospectively collected starting in March 2020. We retrospectively extracted data on the following from the registry database for this observational study: demographics and baseline comorbidities; the use of low-flow supplemental oxygen, HFNC, and invasive mechanical ventilator; and ICU admission, length of hospital stay, length of ICU stay, and hospital discharge disposition. Start and end times for each oxygen therapy were not entered in the registry. Data about other oxygen therapies, such as noninvasive positive-pressure ventilation, were not collected in the registry database, and therefore were not included in the analysis.
Statistical Analysis
Continuous variables (eg, age) were tested for data distribution normality using the Shapiro-Wilk test. Normally distributed data were tested using unpaired t-tests and displayed as mean (SD). The skewed data were tested using the Wilcoxon rank sum test and displayed as median (interquartile range [IQR]). The categorical variables were compared using chi-square test. Comparisons with P ≤ .05 were considered significantly different. Statistical analysis for this study was generated using Statistical Analysis Software (SAS), version 9.4 for Windows (SAS Institute Inc.).
Results
Baseline Characteristics
We included 3183 patients: 1586 admitted during the first surge and 1597 admitted during the second surge. Baseline characteristics of patients with COVID-19 admitted during the first and second surges are shown in Table 1. Patients admitted during the second surge were older (73 years vs 71 years, P = .01) and had higher rates of hypertension (64.8% vs 59.6%, P = .003) and asthma (12.9% vs 10.7%, P = .049) but a lower rate of interstitial lung disease (3.3% vs 7.7%, P < .001). Sequential organ failure assessment scores at admission and the rates of other comorbidities were not significantly different between the 2 surges.

Oxygen Therapies
The number of patients who were hospitalized and received low-flow supplemental oxygen, and/or HFNC, and/or ventilator in the first surge and the second surge is shown in the Figure. Of all patients included, 2067 (64.9%) received low-flow supplemental oxygen; of these, 374 (18.1%) subsequently received HFNC, and 85 (22.7%) of these subsequently received mechanical ventilation. Of all 3183 patients, 417 (13.1%) received HFNC; 43 of these patients received HFNC without receiving low-flow supplemental oxygen, and 98 (23.5%) subsequently received mechanical ventilation. Out of all 3183 patients, 244 (7.7%) received mechanical ventilation; 98 (40.2%) of these received HFNC while the remaining 146 (59.8%) did not. At the beginning of the first surge, the ratio of patients who received invasive mechanical ventilation to patients who received HFNC was close to 1:1 (10/10); the ratio decreased to 6:10 in May and June 2020. At the beginning of the second surge, the ratio was 8:10 and then decreased to 3:10 in December 2020 and January 2021.
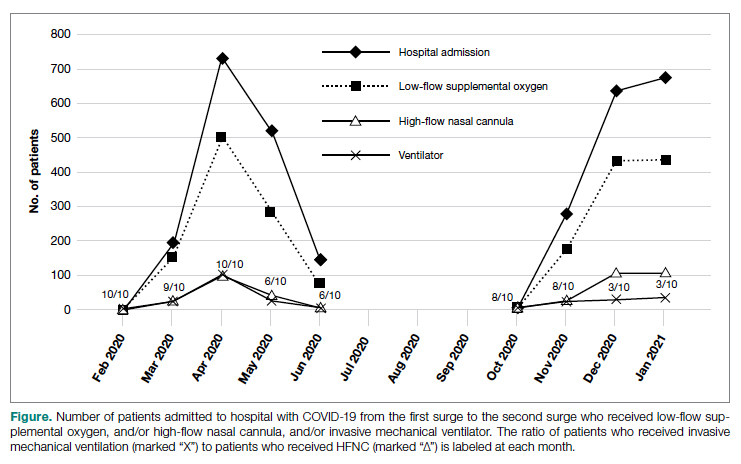
As shown in Table 2, the proportion of patients who received low-flow supplemental oxygen during the second surge was similar to that during the first surge (65.8% vs 64.1%, P = .3). Patients admitted during the second surge were more likely to receive HFNC than patients admitted during the first surge (15.4% vs 10.8%, P = .0001). Patients admitted during the second surge were less likely to be ventilated than the patients admitted during the first surge (5.6% vs 9.7%, P < .0001).
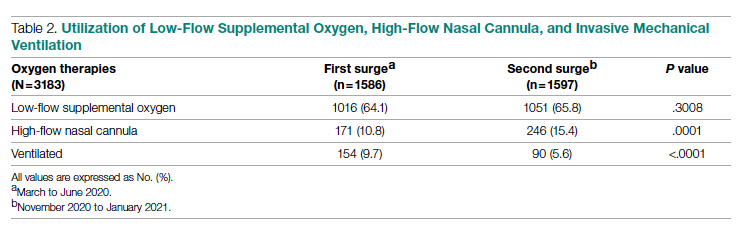
Clinical Outcomes
As shown in Table 3, second surge outcomes were much better than first surge outcomes: the ICU admission rate was lower (8.1% vs 12.7%, P < .0001); patients were more likely to be discharged to home (60.2% vs 47.4%, P < .0001), had a shorter length of hospital stay (5 vs 6 days, P < .0001), and had fewer ventilator days (10 vs 16, P = .01); and mortality was lower (8.3% vs 19.2%, P < .0001). There was a trend that length of ICU stay was shorter during the second surge than during the first surge (7 days vs 9 days, P = .09).
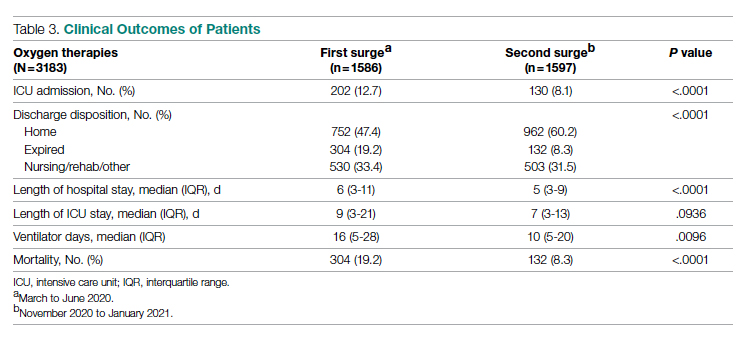
As noted (Figure), the ratio of patients who received invasive mechanical ventilation to patients who received HFNC was decreasing during both the first surge and the second surge. To further analyze the relation between ventilator and HFNC, we performed a subgroup analysis for 244 ventilated patients during both surges to compare outcomes between patients who received HFNC and those who did not receive HFNC (Table 4). Ninety-eight (40%) patients received HFNC. Ventilated patients who received HFNC had lower mortality than those patients who did not receive HFNC (31.6% vs 48%, P = .01), but had a longer length of hospital stay (29 days vs 14 days, P < .0001), longer length of ICU stay (17 days vs 9 days, P < .0001), and a higher number of ventilator days (16 vs 11, P = .001).
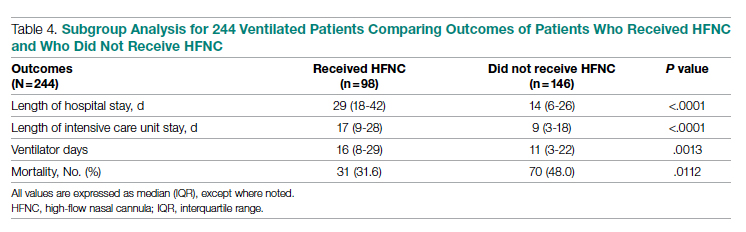
Discussion
Our study compared the baseline patient characteristics; utilization of low-flow supplemental oxygen therapy, HFNC, and mechanical ventilation; and clinical outcomes between the first surge (n = 1586) and the second surge (n = 1597) of the COVID-19 pandemic. During both surges, about two-thirds of admitted patients received low-flow supplemental oxygen. A higher proportion of the admitted patients received HFNC during the second surge than during the first surge, while the intubation rate was lower during the second surge than during the first surge.
Reported low-flow supplemental oxygen use ranged from 28% to 63% depending on the cohort characteristics and location during the first surge.6,7,19 A report from New York during the first surge (March 1 to April 4, 2020) showed that among 5700 hospitalized patients with COVID-19, 27.8% received low-flow supplemental oxygen.7 HFNC is recommended in guidelines on management of patients with acute respiratory failure due to COVID-19.20 In our study, HFNC was utilized in a higher proportion of patients admitted for COVID-19 during the second surge (15.5% vs 10.8%, P = .0001). During the early pandemic period in Wuhan, China, 11% to 21% of admitted COVID-19 patients received HFNC.21,22 Utilization of HFNC in New York during the first surge (March to May 2020) varied from 5% to 14.3% of patients admitted with COVID-19.23,24 Our subgroup analysis of the ventilated patients showed that patients who received HFNC had lower mortality than those who did not (31.6% vs 48.0%, P = .011). Comparably, a report from Paris, France, showed that among patients admitted to ICUs for acute hypoxemic respiratory failure, those who received HFNC had lower mortality at day 60 than those who did not (21% vs 31%, P = .052).25 Our recent analysis showed that patients treated with HFNC prior to mechanical ventilation had lower mortality than those treated with only conventional oxygen (30% vs 52%, P = .05).26 In this subgroup analysis, we could not determine if HFNC treatment was administered before or after ventilation because HFNC was entered as dichotomous data (“Yes” or “No”) in the registry database. We merely showed the beneficial effect of HFNC on reducing mortality for ventilated COVID-19 patients, but did not mean to focus on how and when to apply HFNC.
We observed that the patients admitted during the second surge were less likely to be ventilated than the patients admitted during the first surge (5.6% vs 9.7%, P < .0001). During the first surge in New York, among 5700 patients admitted with COVID-19, 12.2% received invasive mechanical ventilation.7 In another report, also from New York during the first surge, 26.1% of 2015 hospitalized COVID-19 patients received mechanical ventilation.27 In our study, the ventilation rate of 9.7% during the first surge was lower.
Outcomes during the second surge were better than during the first surge, including ICU admission rate, hospital and ICU length of stay, ventilator days, and mortality. The mortality was 19.2% during the first surge vs 8.3% during the second surge (P < .0001). The mortality of 19.2% was lower than the 30.6% mortality reported for 2015 hospitalized COVID-19 patients in New York during the first surge.27 A retrospective study showed that early administration of remdesivir was associated with reduced ICU admission, ventilation use, and mortality.28 The RECOVERY clinical trial showed that dexamethasone improved mortality for COVID-19 patients who received respiratory support, but not for patients who did not receive any respiratory support.29 Perhaps some, if not all, of the improvement in ICU admission and mortality during the second surge was attributed to the new medications, such as antivirals and steroids.
The length of hospital stay for patients with moderate to severe COVID-19 varied from 4 to 53 days at different locations of the world, as shown in a meta-analysis by Rees and colleagues.30 Our results showing a length of stay of 6 days during the first surge and 5 days during the second surge fell into the shorter end of this range. In a retrospective analysis of 1643 adults with severe COVID-19 admitted to hospitals in New York City between March 9, 2020 and April 23, 2020, median hospital length of stay was 7 (IQR, 3-14) days.31 For the ventilated patients in our study, the length of stay of 14 days (did not receive HFNC) and 29 days (received HFNC) was much longer. This longer length of stay might be attributed to the patients in our study being older and having more severe comorbidities.
The main purpose of this study was to compare the oxygen therapies and outcomes between 2 surges. It is difficult to associate the clinical outcomes with the oxygen therapies because new therapies and medications were available after the first surge. It was not possible to adjust the outcomes with confounders (other therapies and medications) because the registry data did not include the new therapies and medications.
A strength of this study was that we included a large, balanced number of patients in the first surge and the second surge. We did not plan the sample size in both groups as we could not predict the number of admissions. We set the end date of data collection for analysis as the time when the number of patients admitted during the second surge was similar to the number of patients admitted during the first surge. A limitation was that the registry database was created by the institution and was not designed solely for this study. The data for oxygen therapies were limited to low-flow supplemental oxygen, HFNC, and invasive mechanical ventilation; data for noninvasive ventilation were not included.
Conclusion
At our centers, during the second surge of COVID-19 pandemic, patients hospitalized with COVID-19 infection were more likely to receive HFNC but less likely to be ventilated. Compared to the first surge, the hospitalized patients with COVID-19 infection had a lower ICU admission rate, shorter length of hospital stay, fewer ventilator days, and lower mortality. For ventilated patients, those who received HFNC had lower mortality than those who did not.
Corresponding author: Timothy N. Liesching, MD, 41 Mall Road, Burlington, MA 01805; [email protected]
Disclosures: None reported.
doi:10.12788/jcom.0086
1. Xie J, Covassin N, Fan Z, et al. Association between hypoxemia and mortality in patients with COVID-19. Mayo Clin Proc. 2020;95(6):1138-1147. doi:10.1016/j.mayocp.2020.04.006
2. Asleh R, Asher E, Yagel O, et al. Predictors of hypoxemia and related adverse outcomes in patients hospitalized with COVID-19: a double-center retrospective study. J Clin Med. 2021;10(16):3581. doi:10.3390/jcm10163581
3. Choi KJ, Hong HL, Kim EJ. Association between oxygen saturation/fraction of inhaled oxygen and mortality in patients with COVID-19 associated pneumonia requiring oxygen therapy. Tuberc Respir Dis (Seoul). 2021;84(2):125-133. doi:10.4046/trd.2020.0126
4. Dixit SB. Role of noninvasive oxygen therapy strategies in COVID-19 patients: Where are we going? Indian J Crit Care Med. 2020;24(10):897-898. doi:10.5005/jp-journals-10071-23625
5. Gonzalez-Castro A, Fajardo Campoverde A, Medina A, et al. Non-invasive mechanical ventilation and high-flow oxygen therapy in the COVID-19 pandemic: the value of a draw. Med Intensiva (Engl Ed). 2021;45(5):320-321. doi:10.1016/j.medine.2021.04.001
6. Pan W, Li J, Ou Y, et al. Clinical outcome of standardized oxygen therapy nursing strategy in COVID-19. Ann Palliat Med. 2020;9(4):2171-2177. doi:10.21037/apm-20-1272
7. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775
8. He G, Han Y, Fang Q, et al. Clinical experience of high-flow nasal cannula oxygen therapy in severe COVID-19 patients. Article in Chinese. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020;49(2):232-239. doi:10.3785/j.issn.1008-9292.2020.03.13
9. Lalla U, Allwood BW, Louw EH, et al. The utility of high-flow nasal cannula oxygen therapy in the management of respiratory failure secondary to COVID-19 pneumonia. S Afr Med J. 2020;110(6):12941.
10. Zhang TT, Dai B, Wang W. Should the high-flow nasal oxygen therapy be used or avoided in COVID-19? J Transl Int Med. 2020;8(2):57-58. doi:10.2478/jtim-2020-0018
11. Agarwal A, Basmaji J, Muttalib F, et al. High-flow nasal cannula for acute hypoxemic respiratory failure in patients with COVID-19: systematic reviews of effectiveness and its risks of aerosolization, dispersion, and infection transmission. Can J Anaesth. 2020;67(9):1217-1248. doi:10.1007/s12630-020-01740-2
12. Geng S, Mei Q, Zhu C, et al. High flow nasal cannula is a good treatment option for COVID-19. Heart Lung. 2020;49(5):444-445. doi:10.1016/j.hrtlng.2020.03.018
13. Feinstein MM, Niforatos JD, Hyun I, et al. Considerations for ventilator triage during the COVID-19 pandemic. Lancet Respir Med. 2020;8(6):e53. doi:10.1016/S2213-2600(20)30192-2
14. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239-1242. doi:10.1001/jama.2020.2648
15. Rojas-Marte G, Hashmi AT, Khalid M, et al. Outcomes in patients with COVID-19 disease and high oxygen requirements. J Clin Med Res. 2021;13(1):26-37. doi:10.14740/jocmr4405
16. Zhang R, Mylonakis E. In inpatients with COVID-19, none of remdesivir, hydroxychloroquine, lopinavir, or interferon β-1a differed from standard care for in-hospital mortality. Ann Intern Med. 2021;174(2):JC17. doi:10.7326/ACPJ202102160-017
17. Rello J, Waterer GW, Bourdiol A, Roquilly A. COVID-19, steroids and other immunomodulators: The jigsaw is not complete. Anaesth Crit Care Pain Med. 2020;39(6):699-701. doi:10.1016/j.accpm.2020.10.011
18. Dargin J, Stempek S, Lei Y, Gray Jr. A, Liesching T. The effect of a tiered provider staffing model on patient outcomes during the coronavirus disease 2019 pandemic: A single-center observational study. Int J Crit Illn Inj Sci. 2021;11(3). doi:10.4103/ijciis.ijciis_37_21
19. Ni YN, Wang T, Liang BM, Liang ZA. The independent factors associated with oxygen therapy in COVID-19 patients under 65 years old. PLoS One. 2021;16(1):e0245690. doi:10.1371/journal.pone.0245690
20. Alhazzani W, Moller MH, Arabi YM, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Crit Care Med. 2020;48(6):e440-e469. doi:10.1097/CCM.0000000000004363
21. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061-1069. doi:10.1001/jama.2020.1585
22. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. doi:10.1016/S0140-6736(20)30566-3
23. Argenziano MG, Bruce SL, Slater CL, et al. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. doi:10.1136/bmj.m1996
24. Cummings MJ, Baldwin MR, Abrams D, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020;395(10239):1763-1770. doi:10.1016/S0140-6736(20)31189-2
25. Demoule A, Vieillard Baron A, Darmon M, et al. High-flow nasal cannula in critically ill patients with severe COVID-19. Am J Respir Crit Care Med. 2020;202(7):1039-1042. doi:10.1164/rccm.202005-2007LE
26. Hansen CK, Stempek S, Liesching T, Lei Y, Dargin J. Characteristics and outcomes of patients receiving high flow nasal cannula therapy prior to mechanical ventilation in COVID-19 respiratory failure: a prospective observational study. Int J Crit Illn Inj Sci. 2021;11(2):56-60. doi:10.4103/IJCIIS.IJCIIS_181_20
27. van Gerwen M, Alsen M, Little C, et al. Risk factors and outcomes of COVID-19 in New York City; a retrospective cohort study. J Med Virol. 2021;93(2):907-915. doi:10.1002/jmv.26337
28. Hussain Alsayed HA, Saheb Sharif-Askari F, Saheb Sharif-Askari N, Hussain AAS, Hamid Q, Halwani R. Early administration of remdesivir to COVID-19 patients associates with higher recovery rate and lower need for ICU admission: A retrospective cohort study. PLoS One. 2021;16(10):e0258643. doi:10.1371/journal.pone.0258643
29. RECOVERY Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704. doi:10.1056/NEJMoa2021436
30. Rees EM, Nightingale ES, Jafari Y, et al. COVID-19 length of hospital stay: a systematic review and data synthesis. BMC Med. 2020;18(1):270. doi:10.1186/s12916-020-01726-3
31. Anderson M, Bach P, Baldwin MR. Hospital length of stay for severe COVID-19: implications for Remdesivir’s value. medRxiv. 2020;2020.08.10.20171637. doi:10.1101/2020.08.10.20171637
From Lahey Hospital and Medical Center, Burlington, MA (Drs. Liesching and Lei), and Tufts University School of Medicine, Boston, MA (Dr. Liesching)
ABSTRACT
Objective: To compare the utilization of oxygen therapies and clinical outcomes of patients admitted for COVID-19 during the second surge of the pandemic to that of patients admitted during the first surge.
Design: Observational study using a registry database.
Setting: Three hospitals (791 inpatient beds and 76 intensive care unit [ICU] beds) within the Beth Israel Lahey Health system in Massachusetts.
Participants: We included 3183 patients with COVID-19 admitted to hospitals.
Measurements: Baseline data included demographics and comorbidities. Treatments included low-flow supplemental oxygen (2-6 L/min), high-flow oxygen via nasal cannula, and invasive mechanical ventilation. Outcomes included ICU admission, length of stay, ventilator days, and mortality.
Results: A total of 3183 patients were included: 1586 during the first surge and 1597 during the second surge. Compared to the first surge, patients admitted during the second surge had a similar rate of receiving low-flow supplemental oxygen (65.8% vs 64.1%, P = .3), a higher rate of receiving high-flow nasal cannula (15.4% vs 10.8%, P = .0001), and a lower ventilation rate (5.6% vs 9.7%, P < .0001). The outcomes during the second surge were better than those during the first surge: lower ICU admission rate (8.1% vs 12.7%, P < .0001), shorter length of hospital stay (5 vs 6 days, P < .0001), fewer ventilator days (10 vs 16, P = .01), and lower mortality (8.3% vs 19.2%, P < .0001). Among ventilated patients, those who received high-flow nasal cannula had lower mortality.
Conclusion: Compared to the first surge of the COVID-19 pandemic, patients admitted during the second surge had similar likelihood of receiving low-flow supplemental oxygen, were more likely to receive high-flow nasal cannula, were less likely to be ventilated, and had better outcomes.
Keywords: supplemental oxygen, high-flow nasal cannula, ventilator.
The respiratory system receives the major impact of SARS-CoV-2 virus, and hypoxemia has been the predominant diagnosis for patients hospitalized with COVID-19.1,2 During the initial stage of the pandemic, oxygen therapies and mechanical ventilation were the only choices for these patients.3-6 Standard-of-care treatment for patients with COVID-19 during the initial surge included oxygen therapies and mechanical ventilation for hypoxemia and medications for comorbidities and COVID-19–associated sequelae, such as multi-organ dysfunction and failure. A report from New York during the first surge (May 2020) showed that among 5700 hospitalized patients with COVID-19, 27.8% received supplemental oxygen and 12.2% received invasive mechanical ventilation.7 High-flow nasal cannula (HFNC) oxygen delivery has been utilized widely throughout the pandemic due to its superiority over other noninvasive respiratory support techniques.8-12 Mechanical ventilation is always necessary for critically ill patients with acute respiratory distress syndrome. However, ventilator scarcity has become a bottleneck in caring for severely ill patients with COVID-19 during the pandemic.13
The clinical outcomes of hospitalized COVID-19 patients include a high intubation rate, long length of hospital and intensive care unit (ICU) stay, and high mortality.14,15 As the pandemic evolved, new medications, including remdesivir, hydroxychloroquine, lopinavir, or interferon β-1a, were used in addition to the standard of care, but these did not result in significantly different mortality from standard of care.16 Steroids are becoming foundational to the treatment of severe COVID-19 pneumonia, but evidence from high-quality randomized controlled clinical trials is lacking.17
During the first surge from March to May 2020, Massachusetts had the third highest number of COVID-19 cases among states in the United States.18 In early 2021, COVID-19 cases were climbing close to the peak of the second surge in Massachusetts. In this study, we compared utilization of low-flow supplemental oxygen, HFNC, and mechanical ventilation and clinical outcomes of patients admitted to 3 hospitals in Massachusetts during the second surge of the pandemic to that of patients admitted during the first surge.
Methods
Setting
Beth Israel Lahey Health is a system of academic and teaching hospitals with primary care and specialty care providers. We included 3 centers within the Beth Israel Lahey Health system in Massachusetts: Lahey Hospital and Medical Center, with 335 inpatient hospital beds and 52 critical care beds; Beverly Hospital, with 227 beds and 14 critical care beds; and Winchester Hospital, with 229 beds and 10 ICU beds.
Participants
We included patients admitted to the 3 hospitals with COVID-19 as a primary or secondary diagnosis during the first surge of the pandemic (March 1, 2020 to June 15, 2020) and the second surge (November 15, 2020 to January 27, 2021). The timeframe of the first surge was defined as the window between the start date and the end date of data collection. During the time window of the first surge, 1586 patients were included. The start time of the second surge was defined as the date when the data collection was restarted; the end date was set when the number of patients (1597) accumulated was close to the number of patients in the first surge (1586), so that the two groups had similar sample size.
Study Design
A data registry of COVID-19 patients was created by our institution, and the data were prospectively collected starting in March 2020. We retrospectively extracted data on the following from the registry database for this observational study: demographics and baseline comorbidities; the use of low-flow supplemental oxygen, HFNC, and invasive mechanical ventilator; and ICU admission, length of hospital stay, length of ICU stay, and hospital discharge disposition. Start and end times for each oxygen therapy were not entered in the registry. Data about other oxygen therapies, such as noninvasive positive-pressure ventilation, were not collected in the registry database, and therefore were not included in the analysis.
Statistical Analysis
Continuous variables (eg, age) were tested for data distribution normality using the Shapiro-Wilk test. Normally distributed data were tested using unpaired t-tests and displayed as mean (SD). The skewed data were tested using the Wilcoxon rank sum test and displayed as median (interquartile range [IQR]). The categorical variables were compared using chi-square test. Comparisons with P ≤ .05 were considered significantly different. Statistical analysis for this study was generated using Statistical Analysis Software (SAS), version 9.4 for Windows (SAS Institute Inc.).
Results
Baseline Characteristics
We included 3183 patients: 1586 admitted during the first surge and 1597 admitted during the second surge. Baseline characteristics of patients with COVID-19 admitted during the first and second surges are shown in Table 1. Patients admitted during the second surge were older (73 years vs 71 years, P = .01) and had higher rates of hypertension (64.8% vs 59.6%, P = .003) and asthma (12.9% vs 10.7%, P = .049) but a lower rate of interstitial lung disease (3.3% vs 7.7%, P < .001). Sequential organ failure assessment scores at admission and the rates of other comorbidities were not significantly different between the 2 surges.

Oxygen Therapies
The number of patients who were hospitalized and received low-flow supplemental oxygen, and/or HFNC, and/or ventilator in the first surge and the second surge is shown in the Figure. Of all patients included, 2067 (64.9%) received low-flow supplemental oxygen; of these, 374 (18.1%) subsequently received HFNC, and 85 (22.7%) of these subsequently received mechanical ventilation. Of all 3183 patients, 417 (13.1%) received HFNC; 43 of these patients received HFNC without receiving low-flow supplemental oxygen, and 98 (23.5%) subsequently received mechanical ventilation. Out of all 3183 patients, 244 (7.7%) received mechanical ventilation; 98 (40.2%) of these received HFNC while the remaining 146 (59.8%) did not. At the beginning of the first surge, the ratio of patients who received invasive mechanical ventilation to patients who received HFNC was close to 1:1 (10/10); the ratio decreased to 6:10 in May and June 2020. At the beginning of the second surge, the ratio was 8:10 and then decreased to 3:10 in December 2020 and January 2021.

As shown in Table 2, the proportion of patients who received low-flow supplemental oxygen during the second surge was similar to that during the first surge (65.8% vs 64.1%, P = .3). Patients admitted during the second surge were more likely to receive HFNC than patients admitted during the first surge (15.4% vs 10.8%, P = .0001). Patients admitted during the second surge were less likely to be ventilated than the patients admitted during the first surge (5.6% vs 9.7%, P < .0001).

Clinical Outcomes
As shown in Table 3, second surge outcomes were much better than first surge outcomes: the ICU admission rate was lower (8.1% vs 12.7%, P < .0001); patients were more likely to be discharged to home (60.2% vs 47.4%, P < .0001), had a shorter length of hospital stay (5 vs 6 days, P < .0001), and had fewer ventilator days (10 vs 16, P = .01); and mortality was lower (8.3% vs 19.2%, P < .0001). There was a trend that length of ICU stay was shorter during the second surge than during the first surge (7 days vs 9 days, P = .09).

As noted (Figure), the ratio of patients who received invasive mechanical ventilation to patients who received HFNC was decreasing during both the first surge and the second surge. To further analyze the relation between ventilator and HFNC, we performed a subgroup analysis for 244 ventilated patients during both surges to compare outcomes between patients who received HFNC and those who did not receive HFNC (Table 4). Ninety-eight (40%) patients received HFNC. Ventilated patients who received HFNC had lower mortality than those patients who did not receive HFNC (31.6% vs 48%, P = .01), but had a longer length of hospital stay (29 days vs 14 days, P < .0001), longer length of ICU stay (17 days vs 9 days, P < .0001), and a higher number of ventilator days (16 vs 11, P = .001).

Discussion
Our study compared the baseline patient characteristics; utilization of low-flow supplemental oxygen therapy, HFNC, and mechanical ventilation; and clinical outcomes between the first surge (n = 1586) and the second surge (n = 1597) of the COVID-19 pandemic. During both surges, about two-thirds of admitted patients received low-flow supplemental oxygen. A higher proportion of the admitted patients received HFNC during the second surge than during the first surge, while the intubation rate was lower during the second surge than during the first surge.
Reported low-flow supplemental oxygen use ranged from 28% to 63% depending on the cohort characteristics and location during the first surge.6,7,19 A report from New York during the first surge (March 1 to April 4, 2020) showed that among 5700 hospitalized patients with COVID-19, 27.8% received low-flow supplemental oxygen.7 HFNC is recommended in guidelines on management of patients with acute respiratory failure due to COVID-19.20 In our study, HFNC was utilized in a higher proportion of patients admitted for COVID-19 during the second surge (15.5% vs 10.8%, P = .0001). During the early pandemic period in Wuhan, China, 11% to 21% of admitted COVID-19 patients received HFNC.21,22 Utilization of HFNC in New York during the first surge (March to May 2020) varied from 5% to 14.3% of patients admitted with COVID-19.23,24 Our subgroup analysis of the ventilated patients showed that patients who received HFNC had lower mortality than those who did not (31.6% vs 48.0%, P = .011). Comparably, a report from Paris, France, showed that among patients admitted to ICUs for acute hypoxemic respiratory failure, those who received HFNC had lower mortality at day 60 than those who did not (21% vs 31%, P = .052).25 Our recent analysis showed that patients treated with HFNC prior to mechanical ventilation had lower mortality than those treated with only conventional oxygen (30% vs 52%, P = .05).26 In this subgroup analysis, we could not determine if HFNC treatment was administered before or after ventilation because HFNC was entered as dichotomous data (“Yes” or “No”) in the registry database. We merely showed the beneficial effect of HFNC on reducing mortality for ventilated COVID-19 patients, but did not mean to focus on how and when to apply HFNC.
We observed that the patients admitted during the second surge were less likely to be ventilated than the patients admitted during the first surge (5.6% vs 9.7%, P < .0001). During the first surge in New York, among 5700 patients admitted with COVID-19, 12.2% received invasive mechanical ventilation.7 In another report, also from New York during the first surge, 26.1% of 2015 hospitalized COVID-19 patients received mechanical ventilation.27 In our study, the ventilation rate of 9.7% during the first surge was lower.
Outcomes during the second surge were better than during the first surge, including ICU admission rate, hospital and ICU length of stay, ventilator days, and mortality. The mortality was 19.2% during the first surge vs 8.3% during the second surge (P < .0001). The mortality of 19.2% was lower than the 30.6% mortality reported for 2015 hospitalized COVID-19 patients in New York during the first surge.27 A retrospective study showed that early administration of remdesivir was associated with reduced ICU admission, ventilation use, and mortality.28 The RECOVERY clinical trial showed that dexamethasone improved mortality for COVID-19 patients who received respiratory support, but not for patients who did not receive any respiratory support.29 Perhaps some, if not all, of the improvement in ICU admission and mortality during the second surge was attributed to the new medications, such as antivirals and steroids.
The length of hospital stay for patients with moderate to severe COVID-19 varied from 4 to 53 days at different locations of the world, as shown in a meta-analysis by Rees and colleagues.30 Our results showing a length of stay of 6 days during the first surge and 5 days during the second surge fell into the shorter end of this range. In a retrospective analysis of 1643 adults with severe COVID-19 admitted to hospitals in New York City between March 9, 2020 and April 23, 2020, median hospital length of stay was 7 (IQR, 3-14) days.31 For the ventilated patients in our study, the length of stay of 14 days (did not receive HFNC) and 29 days (received HFNC) was much longer. This longer length of stay might be attributed to the patients in our study being older and having more severe comorbidities.
The main purpose of this study was to compare the oxygen therapies and outcomes between 2 surges. It is difficult to associate the clinical outcomes with the oxygen therapies because new therapies and medications were available after the first surge. It was not possible to adjust the outcomes with confounders (other therapies and medications) because the registry data did not include the new therapies and medications.
A strength of this study was that we included a large, balanced number of patients in the first surge and the second surge. We did not plan the sample size in both groups as we could not predict the number of admissions. We set the end date of data collection for analysis as the time when the number of patients admitted during the second surge was similar to the number of patients admitted during the first surge. A limitation was that the registry database was created by the institution and was not designed solely for this study. The data for oxygen therapies were limited to low-flow supplemental oxygen, HFNC, and invasive mechanical ventilation; data for noninvasive ventilation were not included.
Conclusion
At our centers, during the second surge of COVID-19 pandemic, patients hospitalized with COVID-19 infection were more likely to receive HFNC but less likely to be ventilated. Compared to the first surge, the hospitalized patients with COVID-19 infection had a lower ICU admission rate, shorter length of hospital stay, fewer ventilator days, and lower mortality. For ventilated patients, those who received HFNC had lower mortality than those who did not.
Corresponding author: Timothy N. Liesching, MD, 41 Mall Road, Burlington, MA 01805; [email protected]
Disclosures: None reported.
doi:10.12788/jcom.0086
From Lahey Hospital and Medical Center, Burlington, MA (Drs. Liesching and Lei), and Tufts University School of Medicine, Boston, MA (Dr. Liesching)
ABSTRACT
Objective: To compare the utilization of oxygen therapies and clinical outcomes of patients admitted for COVID-19 during the second surge of the pandemic to that of patients admitted during the first surge.
Design: Observational study using a registry database.
Setting: Three hospitals (791 inpatient beds and 76 intensive care unit [ICU] beds) within the Beth Israel Lahey Health system in Massachusetts.
Participants: We included 3183 patients with COVID-19 admitted to hospitals.
Measurements: Baseline data included demographics and comorbidities. Treatments included low-flow supplemental oxygen (2-6 L/min), high-flow oxygen via nasal cannula, and invasive mechanical ventilation. Outcomes included ICU admission, length of stay, ventilator days, and mortality.
Results: A total of 3183 patients were included: 1586 during the first surge and 1597 during the second surge. Compared to the first surge, patients admitted during the second surge had a similar rate of receiving low-flow supplemental oxygen (65.8% vs 64.1%, P = .3), a higher rate of receiving high-flow nasal cannula (15.4% vs 10.8%, P = .0001), and a lower ventilation rate (5.6% vs 9.7%, P < .0001). The outcomes during the second surge were better than those during the first surge: lower ICU admission rate (8.1% vs 12.7%, P < .0001), shorter length of hospital stay (5 vs 6 days, P < .0001), fewer ventilator days (10 vs 16, P = .01), and lower mortality (8.3% vs 19.2%, P < .0001). Among ventilated patients, those who received high-flow nasal cannula had lower mortality.
Conclusion: Compared to the first surge of the COVID-19 pandemic, patients admitted during the second surge had similar likelihood of receiving low-flow supplemental oxygen, were more likely to receive high-flow nasal cannula, were less likely to be ventilated, and had better outcomes.
Keywords: supplemental oxygen, high-flow nasal cannula, ventilator.
The respiratory system receives the major impact of SARS-CoV-2 virus, and hypoxemia has been the predominant diagnosis for patients hospitalized with COVID-19.1,2 During the initial stage of the pandemic, oxygen therapies and mechanical ventilation were the only choices for these patients.3-6 Standard-of-care treatment for patients with COVID-19 during the initial surge included oxygen therapies and mechanical ventilation for hypoxemia and medications for comorbidities and COVID-19–associated sequelae, such as multi-organ dysfunction and failure. A report from New York during the first surge (May 2020) showed that among 5700 hospitalized patients with COVID-19, 27.8% received supplemental oxygen and 12.2% received invasive mechanical ventilation.7 High-flow nasal cannula (HFNC) oxygen delivery has been utilized widely throughout the pandemic due to its superiority over other noninvasive respiratory support techniques.8-12 Mechanical ventilation is always necessary for critically ill patients with acute respiratory distress syndrome. However, ventilator scarcity has become a bottleneck in caring for severely ill patients with COVID-19 during the pandemic.13
The clinical outcomes of hospitalized COVID-19 patients include a high intubation rate, long length of hospital and intensive care unit (ICU) stay, and high mortality.14,15 As the pandemic evolved, new medications, including remdesivir, hydroxychloroquine, lopinavir, or interferon β-1a, were used in addition to the standard of care, but these did not result in significantly different mortality from standard of care.16 Steroids are becoming foundational to the treatment of severe COVID-19 pneumonia, but evidence from high-quality randomized controlled clinical trials is lacking.17
During the first surge from March to May 2020, Massachusetts had the third highest number of COVID-19 cases among states in the United States.18 In early 2021, COVID-19 cases were climbing close to the peak of the second surge in Massachusetts. In this study, we compared utilization of low-flow supplemental oxygen, HFNC, and mechanical ventilation and clinical outcomes of patients admitted to 3 hospitals in Massachusetts during the second surge of the pandemic to that of patients admitted during the first surge.
Methods
Setting
Beth Israel Lahey Health is a system of academic and teaching hospitals with primary care and specialty care providers. We included 3 centers within the Beth Israel Lahey Health system in Massachusetts: Lahey Hospital and Medical Center, with 335 inpatient hospital beds and 52 critical care beds; Beverly Hospital, with 227 beds and 14 critical care beds; and Winchester Hospital, with 229 beds and 10 ICU beds.
Participants
We included patients admitted to the 3 hospitals with COVID-19 as a primary or secondary diagnosis during the first surge of the pandemic (March 1, 2020 to June 15, 2020) and the second surge (November 15, 2020 to January 27, 2021). The timeframe of the first surge was defined as the window between the start date and the end date of data collection. During the time window of the first surge, 1586 patients were included. The start time of the second surge was defined as the date when the data collection was restarted; the end date was set when the number of patients (1597) accumulated was close to the number of patients in the first surge (1586), so that the two groups had similar sample size.
Study Design
A data registry of COVID-19 patients was created by our institution, and the data were prospectively collected starting in March 2020. We retrospectively extracted data on the following from the registry database for this observational study: demographics and baseline comorbidities; the use of low-flow supplemental oxygen, HFNC, and invasive mechanical ventilator; and ICU admission, length of hospital stay, length of ICU stay, and hospital discharge disposition. Start and end times for each oxygen therapy were not entered in the registry. Data about other oxygen therapies, such as noninvasive positive-pressure ventilation, were not collected in the registry database, and therefore were not included in the analysis.
Statistical Analysis
Continuous variables (eg, age) were tested for data distribution normality using the Shapiro-Wilk test. Normally distributed data were tested using unpaired t-tests and displayed as mean (SD). The skewed data were tested using the Wilcoxon rank sum test and displayed as median (interquartile range [IQR]). The categorical variables were compared using chi-square test. Comparisons with P ≤ .05 were considered significantly different. Statistical analysis for this study was generated using Statistical Analysis Software (SAS), version 9.4 for Windows (SAS Institute Inc.).
Results
Baseline Characteristics
We included 3183 patients: 1586 admitted during the first surge and 1597 admitted during the second surge. Baseline characteristics of patients with COVID-19 admitted during the first and second surges are shown in Table 1. Patients admitted during the second surge were older (73 years vs 71 years, P = .01) and had higher rates of hypertension (64.8% vs 59.6%, P = .003) and asthma (12.9% vs 10.7%, P = .049) but a lower rate of interstitial lung disease (3.3% vs 7.7%, P < .001). Sequential organ failure assessment scores at admission and the rates of other comorbidities were not significantly different between the 2 surges.

Oxygen Therapies
The number of patients who were hospitalized and received low-flow supplemental oxygen, and/or HFNC, and/or ventilator in the first surge and the second surge is shown in the Figure. Of all patients included, 2067 (64.9%) received low-flow supplemental oxygen; of these, 374 (18.1%) subsequently received HFNC, and 85 (22.7%) of these subsequently received mechanical ventilation. Of all 3183 patients, 417 (13.1%) received HFNC; 43 of these patients received HFNC without receiving low-flow supplemental oxygen, and 98 (23.5%) subsequently received mechanical ventilation. Out of all 3183 patients, 244 (7.7%) received mechanical ventilation; 98 (40.2%) of these received HFNC while the remaining 146 (59.8%) did not. At the beginning of the first surge, the ratio of patients who received invasive mechanical ventilation to patients who received HFNC was close to 1:1 (10/10); the ratio decreased to 6:10 in May and June 2020. At the beginning of the second surge, the ratio was 8:10 and then decreased to 3:10 in December 2020 and January 2021.

As shown in Table 2, the proportion of patients who received low-flow supplemental oxygen during the second surge was similar to that during the first surge (65.8% vs 64.1%, P = .3). Patients admitted during the second surge were more likely to receive HFNC than patients admitted during the first surge (15.4% vs 10.8%, P = .0001). Patients admitted during the second surge were less likely to be ventilated than the patients admitted during the first surge (5.6% vs 9.7%, P < .0001).

Clinical Outcomes
As shown in Table 3, second surge outcomes were much better than first surge outcomes: the ICU admission rate was lower (8.1% vs 12.7%, P < .0001); patients were more likely to be discharged to home (60.2% vs 47.4%, P < .0001), had a shorter length of hospital stay (5 vs 6 days, P < .0001), and had fewer ventilator days (10 vs 16, P = .01); and mortality was lower (8.3% vs 19.2%, P < .0001). There was a trend that length of ICU stay was shorter during the second surge than during the first surge (7 days vs 9 days, P = .09).

As noted (Figure), the ratio of patients who received invasive mechanical ventilation to patients who received HFNC was decreasing during both the first surge and the second surge. To further analyze the relation between ventilator and HFNC, we performed a subgroup analysis for 244 ventilated patients during both surges to compare outcomes between patients who received HFNC and those who did not receive HFNC (Table 4). Ninety-eight (40%) patients received HFNC. Ventilated patients who received HFNC had lower mortality than those patients who did not receive HFNC (31.6% vs 48%, P = .01), but had a longer length of hospital stay (29 days vs 14 days, P < .0001), longer length of ICU stay (17 days vs 9 days, P < .0001), and a higher number of ventilator days (16 vs 11, P = .001).

Discussion
Our study compared the baseline patient characteristics; utilization of low-flow supplemental oxygen therapy, HFNC, and mechanical ventilation; and clinical outcomes between the first surge (n = 1586) and the second surge (n = 1597) of the COVID-19 pandemic. During both surges, about two-thirds of admitted patients received low-flow supplemental oxygen. A higher proportion of the admitted patients received HFNC during the second surge than during the first surge, while the intubation rate was lower during the second surge than during the first surge.
Reported low-flow supplemental oxygen use ranged from 28% to 63% depending on the cohort characteristics and location during the first surge.6,7,19 A report from New York during the first surge (March 1 to April 4, 2020) showed that among 5700 hospitalized patients with COVID-19, 27.8% received low-flow supplemental oxygen.7 HFNC is recommended in guidelines on management of patients with acute respiratory failure due to COVID-19.20 In our study, HFNC was utilized in a higher proportion of patients admitted for COVID-19 during the second surge (15.5% vs 10.8%, P = .0001). During the early pandemic period in Wuhan, China, 11% to 21% of admitted COVID-19 patients received HFNC.21,22 Utilization of HFNC in New York during the first surge (March to May 2020) varied from 5% to 14.3% of patients admitted with COVID-19.23,24 Our subgroup analysis of the ventilated patients showed that patients who received HFNC had lower mortality than those who did not (31.6% vs 48.0%, P = .011). Comparably, a report from Paris, France, showed that among patients admitted to ICUs for acute hypoxemic respiratory failure, those who received HFNC had lower mortality at day 60 than those who did not (21% vs 31%, P = .052).25 Our recent analysis showed that patients treated with HFNC prior to mechanical ventilation had lower mortality than those treated with only conventional oxygen (30% vs 52%, P = .05).26 In this subgroup analysis, we could not determine if HFNC treatment was administered before or after ventilation because HFNC was entered as dichotomous data (“Yes” or “No”) in the registry database. We merely showed the beneficial effect of HFNC on reducing mortality for ventilated COVID-19 patients, but did not mean to focus on how and when to apply HFNC.
We observed that the patients admitted during the second surge were less likely to be ventilated than the patients admitted during the first surge (5.6% vs 9.7%, P < .0001). During the first surge in New York, among 5700 patients admitted with COVID-19, 12.2% received invasive mechanical ventilation.7 In another report, also from New York during the first surge, 26.1% of 2015 hospitalized COVID-19 patients received mechanical ventilation.27 In our study, the ventilation rate of 9.7% during the first surge was lower.
Outcomes during the second surge were better than during the first surge, including ICU admission rate, hospital and ICU length of stay, ventilator days, and mortality. The mortality was 19.2% during the first surge vs 8.3% during the second surge (P < .0001). The mortality of 19.2% was lower than the 30.6% mortality reported for 2015 hospitalized COVID-19 patients in New York during the first surge.27 A retrospective study showed that early administration of remdesivir was associated with reduced ICU admission, ventilation use, and mortality.28 The RECOVERY clinical trial showed that dexamethasone improved mortality for COVID-19 patients who received respiratory support, but not for patients who did not receive any respiratory support.29 Perhaps some, if not all, of the improvement in ICU admission and mortality during the second surge was attributed to the new medications, such as antivirals and steroids.
The length of hospital stay for patients with moderate to severe COVID-19 varied from 4 to 53 days at different locations of the world, as shown in a meta-analysis by Rees and colleagues.30 Our results showing a length of stay of 6 days during the first surge and 5 days during the second surge fell into the shorter end of this range. In a retrospective analysis of 1643 adults with severe COVID-19 admitted to hospitals in New York City between March 9, 2020 and April 23, 2020, median hospital length of stay was 7 (IQR, 3-14) days.31 For the ventilated patients in our study, the length of stay of 14 days (did not receive HFNC) and 29 days (received HFNC) was much longer. This longer length of stay might be attributed to the patients in our study being older and having more severe comorbidities.
The main purpose of this study was to compare the oxygen therapies and outcomes between 2 surges. It is difficult to associate the clinical outcomes with the oxygen therapies because new therapies and medications were available after the first surge. It was not possible to adjust the outcomes with confounders (other therapies and medications) because the registry data did not include the new therapies and medications.
A strength of this study was that we included a large, balanced number of patients in the first surge and the second surge. We did not plan the sample size in both groups as we could not predict the number of admissions. We set the end date of data collection for analysis as the time when the number of patients admitted during the second surge was similar to the number of patients admitted during the first surge. A limitation was that the registry database was created by the institution and was not designed solely for this study. The data for oxygen therapies were limited to low-flow supplemental oxygen, HFNC, and invasive mechanical ventilation; data for noninvasive ventilation were not included.
Conclusion
At our centers, during the second surge of COVID-19 pandemic, patients hospitalized with COVID-19 infection were more likely to receive HFNC but less likely to be ventilated. Compared to the first surge, the hospitalized patients with COVID-19 infection had a lower ICU admission rate, shorter length of hospital stay, fewer ventilator days, and lower mortality. For ventilated patients, those who received HFNC had lower mortality than those who did not.
Corresponding author: Timothy N. Liesching, MD, 41 Mall Road, Burlington, MA 01805; [email protected]
Disclosures: None reported.
doi:10.12788/jcom.0086
1. Xie J, Covassin N, Fan Z, et al. Association between hypoxemia and mortality in patients with COVID-19. Mayo Clin Proc. 2020;95(6):1138-1147. doi:10.1016/j.mayocp.2020.04.006
2. Asleh R, Asher E, Yagel O, et al. Predictors of hypoxemia and related adverse outcomes in patients hospitalized with COVID-19: a double-center retrospective study. J Clin Med. 2021;10(16):3581. doi:10.3390/jcm10163581
3. Choi KJ, Hong HL, Kim EJ. Association between oxygen saturation/fraction of inhaled oxygen and mortality in patients with COVID-19 associated pneumonia requiring oxygen therapy. Tuberc Respir Dis (Seoul). 2021;84(2):125-133. doi:10.4046/trd.2020.0126
4. Dixit SB. Role of noninvasive oxygen therapy strategies in COVID-19 patients: Where are we going? Indian J Crit Care Med. 2020;24(10):897-898. doi:10.5005/jp-journals-10071-23625
5. Gonzalez-Castro A, Fajardo Campoverde A, Medina A, et al. Non-invasive mechanical ventilation and high-flow oxygen therapy in the COVID-19 pandemic: the value of a draw. Med Intensiva (Engl Ed). 2021;45(5):320-321. doi:10.1016/j.medine.2021.04.001
6. Pan W, Li J, Ou Y, et al. Clinical outcome of standardized oxygen therapy nursing strategy in COVID-19. Ann Palliat Med. 2020;9(4):2171-2177. doi:10.21037/apm-20-1272
7. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775
8. He G, Han Y, Fang Q, et al. Clinical experience of high-flow nasal cannula oxygen therapy in severe COVID-19 patients. Article in Chinese. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020;49(2):232-239. doi:10.3785/j.issn.1008-9292.2020.03.13
9. Lalla U, Allwood BW, Louw EH, et al. The utility of high-flow nasal cannula oxygen therapy in the management of respiratory failure secondary to COVID-19 pneumonia. S Afr Med J. 2020;110(6):12941.
10. Zhang TT, Dai B, Wang W. Should the high-flow nasal oxygen therapy be used or avoided in COVID-19? J Transl Int Med. 2020;8(2):57-58. doi:10.2478/jtim-2020-0018
11. Agarwal A, Basmaji J, Muttalib F, et al. High-flow nasal cannula for acute hypoxemic respiratory failure in patients with COVID-19: systematic reviews of effectiveness and its risks of aerosolization, dispersion, and infection transmission. Can J Anaesth. 2020;67(9):1217-1248. doi:10.1007/s12630-020-01740-2
12. Geng S, Mei Q, Zhu C, et al. High flow nasal cannula is a good treatment option for COVID-19. Heart Lung. 2020;49(5):444-445. doi:10.1016/j.hrtlng.2020.03.018
13. Feinstein MM, Niforatos JD, Hyun I, et al. Considerations for ventilator triage during the COVID-19 pandemic. Lancet Respir Med. 2020;8(6):e53. doi:10.1016/S2213-2600(20)30192-2
14. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239-1242. doi:10.1001/jama.2020.2648
15. Rojas-Marte G, Hashmi AT, Khalid M, et al. Outcomes in patients with COVID-19 disease and high oxygen requirements. J Clin Med Res. 2021;13(1):26-37. doi:10.14740/jocmr4405
16. Zhang R, Mylonakis E. In inpatients with COVID-19, none of remdesivir, hydroxychloroquine, lopinavir, or interferon β-1a differed from standard care for in-hospital mortality. Ann Intern Med. 2021;174(2):JC17. doi:10.7326/ACPJ202102160-017
17. Rello J, Waterer GW, Bourdiol A, Roquilly A. COVID-19, steroids and other immunomodulators: The jigsaw is not complete. Anaesth Crit Care Pain Med. 2020;39(6):699-701. doi:10.1016/j.accpm.2020.10.011
18. Dargin J, Stempek S, Lei Y, Gray Jr. A, Liesching T. The effect of a tiered provider staffing model on patient outcomes during the coronavirus disease 2019 pandemic: A single-center observational study. Int J Crit Illn Inj Sci. 2021;11(3). doi:10.4103/ijciis.ijciis_37_21
19. Ni YN, Wang T, Liang BM, Liang ZA. The independent factors associated with oxygen therapy in COVID-19 patients under 65 years old. PLoS One. 2021;16(1):e0245690. doi:10.1371/journal.pone.0245690
20. Alhazzani W, Moller MH, Arabi YM, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Crit Care Med. 2020;48(6):e440-e469. doi:10.1097/CCM.0000000000004363
21. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061-1069. doi:10.1001/jama.2020.1585
22. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. doi:10.1016/S0140-6736(20)30566-3
23. Argenziano MG, Bruce SL, Slater CL, et al. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. doi:10.1136/bmj.m1996
24. Cummings MJ, Baldwin MR, Abrams D, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020;395(10239):1763-1770. doi:10.1016/S0140-6736(20)31189-2
25. Demoule A, Vieillard Baron A, Darmon M, et al. High-flow nasal cannula in critically ill patients with severe COVID-19. Am J Respir Crit Care Med. 2020;202(7):1039-1042. doi:10.1164/rccm.202005-2007LE
26. Hansen CK, Stempek S, Liesching T, Lei Y, Dargin J. Characteristics and outcomes of patients receiving high flow nasal cannula therapy prior to mechanical ventilation in COVID-19 respiratory failure: a prospective observational study. Int J Crit Illn Inj Sci. 2021;11(2):56-60. doi:10.4103/IJCIIS.IJCIIS_181_20
27. van Gerwen M, Alsen M, Little C, et al. Risk factors and outcomes of COVID-19 in New York City; a retrospective cohort study. J Med Virol. 2021;93(2):907-915. doi:10.1002/jmv.26337
28. Hussain Alsayed HA, Saheb Sharif-Askari F, Saheb Sharif-Askari N, Hussain AAS, Hamid Q, Halwani R. Early administration of remdesivir to COVID-19 patients associates with higher recovery rate and lower need for ICU admission: A retrospective cohort study. PLoS One. 2021;16(10):e0258643. doi:10.1371/journal.pone.0258643
29. RECOVERY Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704. doi:10.1056/NEJMoa2021436
30. Rees EM, Nightingale ES, Jafari Y, et al. COVID-19 length of hospital stay: a systematic review and data synthesis. BMC Med. 2020;18(1):270. doi:10.1186/s12916-020-01726-3
31. Anderson M, Bach P, Baldwin MR. Hospital length of stay for severe COVID-19: implications for Remdesivir’s value. medRxiv. 2020;2020.08.10.20171637. doi:10.1101/2020.08.10.20171637
1. Xie J, Covassin N, Fan Z, et al. Association between hypoxemia and mortality in patients with COVID-19. Mayo Clin Proc. 2020;95(6):1138-1147. doi:10.1016/j.mayocp.2020.04.006
2. Asleh R, Asher E, Yagel O, et al. Predictors of hypoxemia and related adverse outcomes in patients hospitalized with COVID-19: a double-center retrospective study. J Clin Med. 2021;10(16):3581. doi:10.3390/jcm10163581
3. Choi KJ, Hong HL, Kim EJ. Association between oxygen saturation/fraction of inhaled oxygen and mortality in patients with COVID-19 associated pneumonia requiring oxygen therapy. Tuberc Respir Dis (Seoul). 2021;84(2):125-133. doi:10.4046/trd.2020.0126
4. Dixit SB. Role of noninvasive oxygen therapy strategies in COVID-19 patients: Where are we going? Indian J Crit Care Med. 2020;24(10):897-898. doi:10.5005/jp-journals-10071-23625
5. Gonzalez-Castro A, Fajardo Campoverde A, Medina A, et al. Non-invasive mechanical ventilation and high-flow oxygen therapy in the COVID-19 pandemic: the value of a draw. Med Intensiva (Engl Ed). 2021;45(5):320-321. doi:10.1016/j.medine.2021.04.001
6. Pan W, Li J, Ou Y, et al. Clinical outcome of standardized oxygen therapy nursing strategy in COVID-19. Ann Palliat Med. 2020;9(4):2171-2177. doi:10.21037/apm-20-1272
7. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775
8. He G, Han Y, Fang Q, et al. Clinical experience of high-flow nasal cannula oxygen therapy in severe COVID-19 patients. Article in Chinese. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020;49(2):232-239. doi:10.3785/j.issn.1008-9292.2020.03.13
9. Lalla U, Allwood BW, Louw EH, et al. The utility of high-flow nasal cannula oxygen therapy in the management of respiratory failure secondary to COVID-19 pneumonia. S Afr Med J. 2020;110(6):12941.
10. Zhang TT, Dai B, Wang W. Should the high-flow nasal oxygen therapy be used or avoided in COVID-19? J Transl Int Med. 2020;8(2):57-58. doi:10.2478/jtim-2020-0018
11. Agarwal A, Basmaji J, Muttalib F, et al. High-flow nasal cannula for acute hypoxemic respiratory failure in patients with COVID-19: systematic reviews of effectiveness and its risks of aerosolization, dispersion, and infection transmission. Can J Anaesth. 2020;67(9):1217-1248. doi:10.1007/s12630-020-01740-2
12. Geng S, Mei Q, Zhu C, et al. High flow nasal cannula is a good treatment option for COVID-19. Heart Lung. 2020;49(5):444-445. doi:10.1016/j.hrtlng.2020.03.018
13. Feinstein MM, Niforatos JD, Hyun I, et al. Considerations for ventilator triage during the COVID-19 pandemic. Lancet Respir Med. 2020;8(6):e53. doi:10.1016/S2213-2600(20)30192-2
14. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239-1242. doi:10.1001/jama.2020.2648
15. Rojas-Marte G, Hashmi AT, Khalid M, et al. Outcomes in patients with COVID-19 disease and high oxygen requirements. J Clin Med Res. 2021;13(1):26-37. doi:10.14740/jocmr4405
16. Zhang R, Mylonakis E. In inpatients with COVID-19, none of remdesivir, hydroxychloroquine, lopinavir, or interferon β-1a differed from standard care for in-hospital mortality. Ann Intern Med. 2021;174(2):JC17. doi:10.7326/ACPJ202102160-017
17. Rello J, Waterer GW, Bourdiol A, Roquilly A. COVID-19, steroids and other immunomodulators: The jigsaw is not complete. Anaesth Crit Care Pain Med. 2020;39(6):699-701. doi:10.1016/j.accpm.2020.10.011
18. Dargin J, Stempek S, Lei Y, Gray Jr. A, Liesching T. The effect of a tiered provider staffing model on patient outcomes during the coronavirus disease 2019 pandemic: A single-center observational study. Int J Crit Illn Inj Sci. 2021;11(3). doi:10.4103/ijciis.ijciis_37_21
19. Ni YN, Wang T, Liang BM, Liang ZA. The independent factors associated with oxygen therapy in COVID-19 patients under 65 years old. PLoS One. 2021;16(1):e0245690. doi:10.1371/journal.pone.0245690
20. Alhazzani W, Moller MH, Arabi YM, et al. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Crit Care Med. 2020;48(6):e440-e469. doi:10.1097/CCM.0000000000004363
21. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061-1069. doi:10.1001/jama.2020.1585
22. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. doi:10.1016/S0140-6736(20)30566-3
23. Argenziano MG, Bruce SL, Slater CL, et al. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. doi:10.1136/bmj.m1996
24. Cummings MJ, Baldwin MR, Abrams D, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020;395(10239):1763-1770. doi:10.1016/S0140-6736(20)31189-2
25. Demoule A, Vieillard Baron A, Darmon M, et al. High-flow nasal cannula in critically ill patients with severe COVID-19. Am J Respir Crit Care Med. 2020;202(7):1039-1042. doi:10.1164/rccm.202005-2007LE
26. Hansen CK, Stempek S, Liesching T, Lei Y, Dargin J. Characteristics and outcomes of patients receiving high flow nasal cannula therapy prior to mechanical ventilation in COVID-19 respiratory failure: a prospective observational study. Int J Crit Illn Inj Sci. 2021;11(2):56-60. doi:10.4103/IJCIIS.IJCIIS_181_20
27. van Gerwen M, Alsen M, Little C, et al. Risk factors and outcomes of COVID-19 in New York City; a retrospective cohort study. J Med Virol. 2021;93(2):907-915. doi:10.1002/jmv.26337
28. Hussain Alsayed HA, Saheb Sharif-Askari F, Saheb Sharif-Askari N, Hussain AAS, Hamid Q, Halwani R. Early administration of remdesivir to COVID-19 patients associates with higher recovery rate and lower need for ICU admission: A retrospective cohort study. PLoS One. 2021;16(10):e0258643. doi:10.1371/journal.pone.0258643
29. RECOVERY Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704. doi:10.1056/NEJMoa2021436
30. Rees EM, Nightingale ES, Jafari Y, et al. COVID-19 length of hospital stay: a systematic review and data synthesis. BMC Med. 2020;18(1):270. doi:10.1186/s12916-020-01726-3
31. Anderson M, Bach P, Baldwin MR. Hospital length of stay for severe COVID-19: implications for Remdesivir’s value. medRxiv. 2020;2020.08.10.20171637. doi:10.1101/2020.08.10.20171637
Obesity linked to combined OSA syndrome and severe asthma
Almost all patients with both obstructive sleep apnea syndrome and severe asthma fell into the obesity phenotype, not the allergy phenotype, based on data from nearly 1,500 adults.
Both asthma and sleep-disordered breathing are common conditions worldwide, and previous research suggests that obstructive sleep apnea syndrome (OSAS) and severe asthma in particular could be associated, wrote Laurent Portel, MD, of Centre Hospitalier de Libourne, France, and colleagues.
“Even if the underlying mechanisms are not well established, it is clear that both OSAS and obesity act to aggravate existing asthma, making it more difficult to control,” they said. However, the pathology of this relationship is not well-understood, and data on severe asthma phenotypes and OSAS are limited, they said.
In a study published in Respiratory Medicine and Research, the investigators reviewed data from 1,465 patients older than 18 years with severe asthma who were part of a larger, prospective multicenter study of the management of asthma patients. The larger study, developed by the Collège des Pneumologues des Hôpitaux Généraux (CPHG) is known as the FASE-CPHG (France Asthme SEvère-CPHG) and includes 104 nonacademic hospitals in France.
Diagnosis of OSAS was reported by physicians; diagnosis of severe asthma was based on the Global Initiative for Asthma criteria. The average age of the patients was 54.4 years, 63% were women, and 60% were nonsmokers.
A total of 161 patients were diagnosed with OSAS. The researchers conducted a cluster analysis on 1,424 patients, including 156 of the OSAS patients. They identified five clusters: early-onset atopic asthma (690 patients), obese asthma (153 patients), late-onset asthma (299 patients), eosinophilic asthma (143 patients), and aspirin sensitivity asthma (139 patients).
All 153 patients in the obese asthma cluster had OSAS, by contrast, none of the patients in the early atopic asthma cluster had OSAS.
Overall, obesity, male sex, high blood pressure, depression, late-onset asthma, and early-onset atopic asthma were independently associated with OSAS, with odds ratios of 5.782, 3.047, 2.875, 2.552, 1.789, and 0.622, respectively.
Notably, OSAS patients were more frequently treated with long-term oral corticosteroids than those without OSAS (30% vs. 15%, P < .0001), the researchers said. “It is possible that this treatment may be responsible for obesity, and it represents a well-known risk factor for developing OSAS,” they wrote.
Uncontrolled asthma was significantly more common in OSAS patients than in those without OSAS (77.7% vs. 69%, P = .03), and significantly more OSAS patients reported no or occasional physical activity (79.8% vs. 68.2%, P ≤ .001).
The study findings were limited by several factors including the lack of patients from primary care or university hospitals, which may limit the generalizability of the results, the reliance on physician statements for diagnosis of OSAS, and the lack of data on OSAS severity or treatment, the researchers noted.
However, the results fill a needed gap in the literature because of the limited data on severe asthma patients in real life, and identifying severe asthma patients by phenotype may help identify those at greatest risk for OSAS, they said.
“Identified patients could more easily benefit from specific examinations such as poly(somno)graphy and, consequently, could benefit from a better management of both asthma and OSAS,” they emphasized.
The larger FASE-CPHG study was supported in part by ALK, AstraZeneca, Boehringer Ingelheim, GSK, and Le Nouveau Souffle. The researchers had no financial conflicts to disclose.
Almost all patients with both obstructive sleep apnea syndrome and severe asthma fell into the obesity phenotype, not the allergy phenotype, based on data from nearly 1,500 adults.
Both asthma and sleep-disordered breathing are common conditions worldwide, and previous research suggests that obstructive sleep apnea syndrome (OSAS) and severe asthma in particular could be associated, wrote Laurent Portel, MD, of Centre Hospitalier de Libourne, France, and colleagues.
“Even if the underlying mechanisms are not well established, it is clear that both OSAS and obesity act to aggravate existing asthma, making it more difficult to control,” they said. However, the pathology of this relationship is not well-understood, and data on severe asthma phenotypes and OSAS are limited, they said.
In a study published in Respiratory Medicine and Research, the investigators reviewed data from 1,465 patients older than 18 years with severe asthma who were part of a larger, prospective multicenter study of the management of asthma patients. The larger study, developed by the Collège des Pneumologues des Hôpitaux Généraux (CPHG) is known as the FASE-CPHG (France Asthme SEvère-CPHG) and includes 104 nonacademic hospitals in France.
Diagnosis of OSAS was reported by physicians; diagnosis of severe asthma was based on the Global Initiative for Asthma criteria. The average age of the patients was 54.4 years, 63% were women, and 60% were nonsmokers.
A total of 161 patients were diagnosed with OSAS. The researchers conducted a cluster analysis on 1,424 patients, including 156 of the OSAS patients. They identified five clusters: early-onset atopic asthma (690 patients), obese asthma (153 patients), late-onset asthma (299 patients), eosinophilic asthma (143 patients), and aspirin sensitivity asthma (139 patients).
All 153 patients in the obese asthma cluster had OSAS, by contrast, none of the patients in the early atopic asthma cluster had OSAS.
Overall, obesity, male sex, high blood pressure, depression, late-onset asthma, and early-onset atopic asthma were independently associated with OSAS, with odds ratios of 5.782, 3.047, 2.875, 2.552, 1.789, and 0.622, respectively.
Notably, OSAS patients were more frequently treated with long-term oral corticosteroids than those without OSAS (30% vs. 15%, P < .0001), the researchers said. “It is possible that this treatment may be responsible for obesity, and it represents a well-known risk factor for developing OSAS,” they wrote.
Uncontrolled asthma was significantly more common in OSAS patients than in those without OSAS (77.7% vs. 69%, P = .03), and significantly more OSAS patients reported no or occasional physical activity (79.8% vs. 68.2%, P ≤ .001).
The study findings were limited by several factors including the lack of patients from primary care or university hospitals, which may limit the generalizability of the results, the reliance on physician statements for diagnosis of OSAS, and the lack of data on OSAS severity or treatment, the researchers noted.
However, the results fill a needed gap in the literature because of the limited data on severe asthma patients in real life, and identifying severe asthma patients by phenotype may help identify those at greatest risk for OSAS, they said.
“Identified patients could more easily benefit from specific examinations such as poly(somno)graphy and, consequently, could benefit from a better management of both asthma and OSAS,” they emphasized.
The larger FASE-CPHG study was supported in part by ALK, AstraZeneca, Boehringer Ingelheim, GSK, and Le Nouveau Souffle. The researchers had no financial conflicts to disclose.
Almost all patients with both obstructive sleep apnea syndrome and severe asthma fell into the obesity phenotype, not the allergy phenotype, based on data from nearly 1,500 adults.
Both asthma and sleep-disordered breathing are common conditions worldwide, and previous research suggests that obstructive sleep apnea syndrome (OSAS) and severe asthma in particular could be associated, wrote Laurent Portel, MD, of Centre Hospitalier de Libourne, France, and colleagues.
“Even if the underlying mechanisms are not well established, it is clear that both OSAS and obesity act to aggravate existing asthma, making it more difficult to control,” they said. However, the pathology of this relationship is not well-understood, and data on severe asthma phenotypes and OSAS are limited, they said.
In a study published in Respiratory Medicine and Research, the investigators reviewed data from 1,465 patients older than 18 years with severe asthma who were part of a larger, prospective multicenter study of the management of asthma patients. The larger study, developed by the Collège des Pneumologues des Hôpitaux Généraux (CPHG) is known as the FASE-CPHG (France Asthme SEvère-CPHG) and includes 104 nonacademic hospitals in France.
Diagnosis of OSAS was reported by physicians; diagnosis of severe asthma was based on the Global Initiative for Asthma criteria. The average age of the patients was 54.4 years, 63% were women, and 60% were nonsmokers.
A total of 161 patients were diagnosed with OSAS. The researchers conducted a cluster analysis on 1,424 patients, including 156 of the OSAS patients. They identified five clusters: early-onset atopic asthma (690 patients), obese asthma (153 patients), late-onset asthma (299 patients), eosinophilic asthma (143 patients), and aspirin sensitivity asthma (139 patients).
All 153 patients in the obese asthma cluster had OSAS, by contrast, none of the patients in the early atopic asthma cluster had OSAS.
Overall, obesity, male sex, high blood pressure, depression, late-onset asthma, and early-onset atopic asthma were independently associated with OSAS, with odds ratios of 5.782, 3.047, 2.875, 2.552, 1.789, and 0.622, respectively.
Notably, OSAS patients were more frequently treated with long-term oral corticosteroids than those without OSAS (30% vs. 15%, P < .0001), the researchers said. “It is possible that this treatment may be responsible for obesity, and it represents a well-known risk factor for developing OSAS,” they wrote.
Uncontrolled asthma was significantly more common in OSAS patients than in those without OSAS (77.7% vs. 69%, P = .03), and significantly more OSAS patients reported no or occasional physical activity (79.8% vs. 68.2%, P ≤ .001).
The study findings were limited by several factors including the lack of patients from primary care or university hospitals, which may limit the generalizability of the results, the reliance on physician statements for diagnosis of OSAS, and the lack of data on OSAS severity or treatment, the researchers noted.
However, the results fill a needed gap in the literature because of the limited data on severe asthma patients in real life, and identifying severe asthma patients by phenotype may help identify those at greatest risk for OSAS, they said.
“Identified patients could more easily benefit from specific examinations such as poly(somno)graphy and, consequently, could benefit from a better management of both asthma and OSAS,” they emphasized.
The larger FASE-CPHG study was supported in part by ALK, AstraZeneca, Boehringer Ingelheim, GSK, and Le Nouveau Souffle. The researchers had no financial conflicts to disclose.
FROM RESPIRATORY MEDICINE AND RESEARCH
Dealing with life-threatening asthma attacks
In assessing and managing patients presenting with acute, life-threatening asthma, if the exacerbation does not resolve relatively quickly, clinicians need to start looking for other causes of the patient’s respiratory distress, a review of the literature suggests.
“I think one of the most important points of this review is that asthma is a self-limiting disease, and it’s important to understand that with appropriate treatment and immediate response to it, exacerbations will get better with time,” Orlando Garner, MD, Baylor College of Medicine, Houston, said in an interview.
“So I think one of the key points is, if these exacerbations do not resolve within 24-48 hours, clinicians need to start thinking: ‘This could be something else,’ and not get stuck in the diagnosis that this is an asthmatic patient who is having an exacerbation. If the distress doesn’t resolve within 48 hours, it’s time to look for other clues,” he stressed.
The study was published online in the journal CHEST®.
Appropriate triage
Appropriate triage is key in the management of acute asthma, Dr. Garner and colleagues pointed out. A simplified severity score for the evaluation of asthma in the ED can help in this regard. Depending on the presence or absence of a number of key signs and symptoms, patients can be readily categorized as having mild, moderate, or severe asthma. “Static assessments and dynamic assessments of acute asthma exacerbation in the ED can also help triage patients,” the authors added.
Static assessment involves assessing the severity at presentation, which in turn determines the aggressiveness of initial treatment. Objective static assessments include the measurement of peak expiratory flow (PEF) or forced expiratory volume in 1 second (FEV1). A severe exacerbation is usually defined as a PEF or an FEV1 of less than 50%-60% of predicted normal values, the authors noted.
Dynamic assessment is more helpful than static assessment because it gauges response to treatment. “A lack of improvement in expiratory flow rates after initial bronchodilator therapy with continuous or worsening symptoms suggests need for hospitalization,” Dr. Garner and colleagues observed. The main treatment goals for patients with acute asthma are reversal of bronchospasm and correction of hypoxemia.
These are achieved at least initially with conventional agents, such as repeated doses of inhaled short-acting beta2-agonists, inhaled short-acting anticholinergics, systemic corticosteroids, and occasionally intravenous magnesium sulfate. If there is concomitant hypoxemia, oxygen therapy should be initiated as well. Patients who have evidence of hypercapnic respiratory failure or diaphragmatic fatigue need to be admitted to the intensive care unit, the authors indicated.
For these patients, clinicians need to remember that there are therapies other than inhalers, such as epinephrine and systemic terbutaline. During a life-threatening asthma episode, airflow in the medium and small airways often becomes turbulent, increasing the work of breathing, the researchers pointed out.
Heliox, a combination of helium and oxygen, reduces turbulent flow, they noted, although FiO2 requirements need to be less than 30% in order for it to work. “Heliox can be used in patients with severe bronchospasm who do not respond to the conventional therapies,” the authors noted, “[but] therapy should be abandoned if there is no clinical improvement after 15 minutes of use.”
Although none of the biologics such as dupilumab (Dupixent) has yet been approved for the treatment of acute exacerbations, Dr. Garner predicts they will become the “future of medicine” for patients with severe asthma as well.
Ventilation in life-threatening asthma
Rapid sequence intubation is generally recommended for patients who require mechanical ventilation, but as an alternative, “we are advocating a slower approach, where we get patients to slow down their breathing and relax them with something like ketamine infusions and wait before we given them a paralytic to see if the work of breathing improves,” Dr. Garner said. Bag-mask ventilation should be avoided because it can worsen dynamic hyperinflation or cause barotrauma.
Salvage therapies such as the use of bronchoscopy with N-acetylcysteine instilled directly into the airway is another option in cases in which mucus plugging is considered to be the main driver of airflow limitation.
Asked to comment, Brit Long, MD, an emergency medicine physician at the Brooke Army Medical Center in San Antonio, Tex., felt the review was extremely useful and well done.
“We see these patients very frequently, and being able to assess them right away and get an accurate picture of what’s going on is very important,” he said in an interview. The one thing that is often more difficult, at least in the ED, is obtaining a PEF or the FEV1 – “both very helpful if the patient can do them, but if the patient is critically ill, it’s more likely you will not be able to get those assessments, and if patients are speaking in one-word sentences and are working really hard to breathe, that’s a severe exacerbation, and they need immediate intervention.” Dr. Long also liked all the essential treatments the authors recommended that patients be given immediately, although he noted that Heliox is not going to be available in most EDs.
On the other hand, he agreed with the authors’ recommendation to take a slower approach to mechanical ventilation, if it is needed at all. “I try my best to absolutely avoid intubating these patients – you are not fixing the issue with mechanical ventilation, you are just creating further problems.
“And while I see the entire spectrum of asthma patients from very mild to severe patients, these authors did a good job in explaining what the goals of treatment are and what to do with the severe ones,” he said.
Dr. Garner and Dr. Long disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In assessing and managing patients presenting with acute, life-threatening asthma, if the exacerbation does not resolve relatively quickly, clinicians need to start looking for other causes of the patient’s respiratory distress, a review of the literature suggests.
“I think one of the most important points of this review is that asthma is a self-limiting disease, and it’s important to understand that with appropriate treatment and immediate response to it, exacerbations will get better with time,” Orlando Garner, MD, Baylor College of Medicine, Houston, said in an interview.
“So I think one of the key points is, if these exacerbations do not resolve within 24-48 hours, clinicians need to start thinking: ‘This could be something else,’ and not get stuck in the diagnosis that this is an asthmatic patient who is having an exacerbation. If the distress doesn’t resolve within 48 hours, it’s time to look for other clues,” he stressed.
The study was published online in the journal CHEST®.
Appropriate triage
Appropriate triage is key in the management of acute asthma, Dr. Garner and colleagues pointed out. A simplified severity score for the evaluation of asthma in the ED can help in this regard. Depending on the presence or absence of a number of key signs and symptoms, patients can be readily categorized as having mild, moderate, or severe asthma. “Static assessments and dynamic assessments of acute asthma exacerbation in the ED can also help triage patients,” the authors added.
Static assessment involves assessing the severity at presentation, which in turn determines the aggressiveness of initial treatment. Objective static assessments include the measurement of peak expiratory flow (PEF) or forced expiratory volume in 1 second (FEV1). A severe exacerbation is usually defined as a PEF or an FEV1 of less than 50%-60% of predicted normal values, the authors noted.
Dynamic assessment is more helpful than static assessment because it gauges response to treatment. “A lack of improvement in expiratory flow rates after initial bronchodilator therapy with continuous or worsening symptoms suggests need for hospitalization,” Dr. Garner and colleagues observed. The main treatment goals for patients with acute asthma are reversal of bronchospasm and correction of hypoxemia.
These are achieved at least initially with conventional agents, such as repeated doses of inhaled short-acting beta2-agonists, inhaled short-acting anticholinergics, systemic corticosteroids, and occasionally intravenous magnesium sulfate. If there is concomitant hypoxemia, oxygen therapy should be initiated as well. Patients who have evidence of hypercapnic respiratory failure or diaphragmatic fatigue need to be admitted to the intensive care unit, the authors indicated.
For these patients, clinicians need to remember that there are therapies other than inhalers, such as epinephrine and systemic terbutaline. During a life-threatening asthma episode, airflow in the medium and small airways often becomes turbulent, increasing the work of breathing, the researchers pointed out.
Heliox, a combination of helium and oxygen, reduces turbulent flow, they noted, although FiO2 requirements need to be less than 30% in order for it to work. “Heliox can be used in patients with severe bronchospasm who do not respond to the conventional therapies,” the authors noted, “[but] therapy should be abandoned if there is no clinical improvement after 15 minutes of use.”
Although none of the biologics such as dupilumab (Dupixent) has yet been approved for the treatment of acute exacerbations, Dr. Garner predicts they will become the “future of medicine” for patients with severe asthma as well.
Ventilation in life-threatening asthma
Rapid sequence intubation is generally recommended for patients who require mechanical ventilation, but as an alternative, “we are advocating a slower approach, where we get patients to slow down their breathing and relax them with something like ketamine infusions and wait before we given them a paralytic to see if the work of breathing improves,” Dr. Garner said. Bag-mask ventilation should be avoided because it can worsen dynamic hyperinflation or cause barotrauma.
Salvage therapies such as the use of bronchoscopy with N-acetylcysteine instilled directly into the airway is another option in cases in which mucus plugging is considered to be the main driver of airflow limitation.
Asked to comment, Brit Long, MD, an emergency medicine physician at the Brooke Army Medical Center in San Antonio, Tex., felt the review was extremely useful and well done.
“We see these patients very frequently, and being able to assess them right away and get an accurate picture of what’s going on is very important,” he said in an interview. The one thing that is often more difficult, at least in the ED, is obtaining a PEF or the FEV1 – “both very helpful if the patient can do them, but if the patient is critically ill, it’s more likely you will not be able to get those assessments, and if patients are speaking in one-word sentences and are working really hard to breathe, that’s a severe exacerbation, and they need immediate intervention.” Dr. Long also liked all the essential treatments the authors recommended that patients be given immediately, although he noted that Heliox is not going to be available in most EDs.
On the other hand, he agreed with the authors’ recommendation to take a slower approach to mechanical ventilation, if it is needed at all. “I try my best to absolutely avoid intubating these patients – you are not fixing the issue with mechanical ventilation, you are just creating further problems.
“And while I see the entire spectrum of asthma patients from very mild to severe patients, these authors did a good job in explaining what the goals of treatment are and what to do with the severe ones,” he said.
Dr. Garner and Dr. Long disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In assessing and managing patients presenting with acute, life-threatening asthma, if the exacerbation does not resolve relatively quickly, clinicians need to start looking for other causes of the patient’s respiratory distress, a review of the literature suggests.
“I think one of the most important points of this review is that asthma is a self-limiting disease, and it’s important to understand that with appropriate treatment and immediate response to it, exacerbations will get better with time,” Orlando Garner, MD, Baylor College of Medicine, Houston, said in an interview.
“So I think one of the key points is, if these exacerbations do not resolve within 24-48 hours, clinicians need to start thinking: ‘This could be something else,’ and not get stuck in the diagnosis that this is an asthmatic patient who is having an exacerbation. If the distress doesn’t resolve within 48 hours, it’s time to look for other clues,” he stressed.
The study was published online in the journal CHEST®.
Appropriate triage
Appropriate triage is key in the management of acute asthma, Dr. Garner and colleagues pointed out. A simplified severity score for the evaluation of asthma in the ED can help in this regard. Depending on the presence or absence of a number of key signs and symptoms, patients can be readily categorized as having mild, moderate, or severe asthma. “Static assessments and dynamic assessments of acute asthma exacerbation in the ED can also help triage patients,” the authors added.
Static assessment involves assessing the severity at presentation, which in turn determines the aggressiveness of initial treatment. Objective static assessments include the measurement of peak expiratory flow (PEF) or forced expiratory volume in 1 second (FEV1). A severe exacerbation is usually defined as a PEF or an FEV1 of less than 50%-60% of predicted normal values, the authors noted.
Dynamic assessment is more helpful than static assessment because it gauges response to treatment. “A lack of improvement in expiratory flow rates after initial bronchodilator therapy with continuous or worsening symptoms suggests need for hospitalization,” Dr. Garner and colleagues observed. The main treatment goals for patients with acute asthma are reversal of bronchospasm and correction of hypoxemia.
These are achieved at least initially with conventional agents, such as repeated doses of inhaled short-acting beta2-agonists, inhaled short-acting anticholinergics, systemic corticosteroids, and occasionally intravenous magnesium sulfate. If there is concomitant hypoxemia, oxygen therapy should be initiated as well. Patients who have evidence of hypercapnic respiratory failure or diaphragmatic fatigue need to be admitted to the intensive care unit, the authors indicated.
For these patients, clinicians need to remember that there are therapies other than inhalers, such as epinephrine and systemic terbutaline. During a life-threatening asthma episode, airflow in the medium and small airways often becomes turbulent, increasing the work of breathing, the researchers pointed out.
Heliox, a combination of helium and oxygen, reduces turbulent flow, they noted, although FiO2 requirements need to be less than 30% in order for it to work. “Heliox can be used in patients with severe bronchospasm who do not respond to the conventional therapies,” the authors noted, “[but] therapy should be abandoned if there is no clinical improvement after 15 minutes of use.”
Although none of the biologics such as dupilumab (Dupixent) has yet been approved for the treatment of acute exacerbations, Dr. Garner predicts they will become the “future of medicine” for patients with severe asthma as well.
Ventilation in life-threatening asthma
Rapid sequence intubation is generally recommended for patients who require mechanical ventilation, but as an alternative, “we are advocating a slower approach, where we get patients to slow down their breathing and relax them with something like ketamine infusions and wait before we given them a paralytic to see if the work of breathing improves,” Dr. Garner said. Bag-mask ventilation should be avoided because it can worsen dynamic hyperinflation or cause barotrauma.
Salvage therapies such as the use of bronchoscopy with N-acetylcysteine instilled directly into the airway is another option in cases in which mucus plugging is considered to be the main driver of airflow limitation.
Asked to comment, Brit Long, MD, an emergency medicine physician at the Brooke Army Medical Center in San Antonio, Tex., felt the review was extremely useful and well done.
“We see these patients very frequently, and being able to assess them right away and get an accurate picture of what’s going on is very important,” he said in an interview. The one thing that is often more difficult, at least in the ED, is obtaining a PEF or the FEV1 – “both very helpful if the patient can do them, but if the patient is critically ill, it’s more likely you will not be able to get those assessments, and if patients are speaking in one-word sentences and are working really hard to breathe, that’s a severe exacerbation, and they need immediate intervention.” Dr. Long also liked all the essential treatments the authors recommended that patients be given immediately, although he noted that Heliox is not going to be available in most EDs.
On the other hand, he agreed with the authors’ recommendation to take a slower approach to mechanical ventilation, if it is needed at all. “I try my best to absolutely avoid intubating these patients – you are not fixing the issue with mechanical ventilation, you are just creating further problems.
“And while I see the entire spectrum of asthma patients from very mild to severe patients, these authors did a good job in explaining what the goals of treatment are and what to do with the severe ones,” he said.
Dr. Garner and Dr. Long disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CHEST
Pollution levels linked to physical and mental health problems
Other analyses of data have found environmental air pollution from sources such as car exhaust and factory output can trigger an inflammatory response in the body. What’s new about a study published in RMD Open is that it explored an association between long-term exposure to pollution and risk of autoimmune diseases, wrote Giovanni Adami, MD, of the University of Verona (Italy) and colleagues.
“Environmental air pollution, according to the World Health Organization, is a major risk to health and 99% of the population worldwide is living in places where recommendations for air quality are not met,” said Dr. Adami in an interview. The limited data on the precise role of air pollution on rheumatic diseases in particular prompted the study, he said.
To explore the potential link between air pollution exposure and autoimmune disease, the researchers reviewed medical information from 81,363 adults via a national medical database in Italy; the data were submitted between June 2016 and November 2020.
The average age of the study population was 65 years, and 92% were women; 22% had at least one coexisting health condition. Each study participant was linked to local environmental monitoring via their residential postcode.
The researchers obtained details about concentrations of particulate matter in the environment from the Italian Institute of Environmental Protection that included 617 monitoring stations in 110 Italian provinces. They focused on concentrations of 10 and 2.5 (PM10 and PM2.5).
Exposure thresholds of 30 mcg/m3 for PM10 and 20 mcg/m3 for PM2.5 are generally considered harmful to health, they noted. On average, the long-term exposure was 16 mcg/m3 for PM2.5 and 25 mcg/m3 for PM10 between 2013 and 2019.
Overall, 9,723 individuals (12%) were diagnosed with an autoimmune disease between 2016 and 2020.
Exposure to PM10 was associated with a 7% higher risk of diagnosis with any autoimmune disease for every 10 mcg/m3 increase in concentration, but no association appeared between PM2.5 exposure and increased risk of autoimmune diseases.
However, in an adjusted model, chronic exposure to PM10 above 30 mcg/m3 and to PM2.5 above 20 mcg/m3 were associated with a 12% and 13% higher risk, respectively, of any autoimmune disease.
Chronic exposure to high levels of PM10 was specifically associated with a higher risk of rheumatoid arthritis, but no other autoimmune diseases. Chronic exposure to high levels of PM2.5 was associated with a higher risk of rheumatoid arthritis, connective tissue diseases, and inflammatory bowel diseases.
In their discussion, the researchers noted that the smaller diameter of PM2.5 molecules fluctuate less in response to rain and other weather, compared with PM10 molecules, which might make them a more accurate predictor of exposure to chronic air pollution.
The study findings were limited by several factors including the observational design, which prohibits the establishment of cause, and a lack of data on the start of symptoms and dates of diagnoses for autoimmune diseases, the researchers noted. Other limitations include the high percentage of older women in the study, which may limit generalizability, and the inability to account for additional personal exposure to pollutants outside of the environmental exposure, they said.
However, the results were strengthened by the large sample size and wide geographic distribution with variable pollution exposure, they said.
“Unfortunately, we were not surprised at all,” by the findings, Dr. Adami said in an interview.
“The biological rationale underpinning our findings is strong. Nevertheless, the magnitude of the effect was overwhelming. In addition, we saw an effect even at threshold of exposure that is widely considered as safe,” Dr. Adami noted.
Clinicians have been taught to consider cigarette smoking or other lifestyle behaviors as major risk factors for the development of several autoimmune diseases, said Dr. Adami. “In the future, we probably should include air pollution exposure as a risk factor as well. Interestingly, there is also accumulating evidence linking acute exposure to environmental air pollution with flares of chronic arthritis,” he said.
“Our study could have direct societal and political consequences,” and might help direct policy makers’ decisions on addressing strategies aimed to reduce fossil emissions, he said. As for additional research, “we certainly need multination studies to confirm our results on a larger scale,” Dr. Adami emphasized. “In addition, it is time to take action and start designing interventions aimed to reduce acute and chronic exposure to air pollution in patients suffering from RMDs.”
Consider the big picture of air quality
The Italian study is especially timely “given our evolving and emerging understanding of environmental risk factors for acute and chronic diseases, which we must first understand before we can address,” said Eileen Barrett, MD, of the University of New Mexico, Albuquerque, in an interview.
“I am largely surprised about the findings, as most physicians aren’t studying ambient air quality and risk for autoimmune disease,” said Dr. Barrett. “More often we think of air quality when we think of risk for respiratory diseases than autoimmune diseases, per se,” she said.
“There are several take-home messages from this study,” said Dr. Barrett. “The first is that we need more research to understand the consequences of air pollutants on health. Second, this study reminds us to think broadly about how air quality and our environment can affect health. And third, all clinicians should be committed to promoting science that can improve public health and reduce death and disability,” she emphasized.
The findings do not specifically reflect associations between pollution and other conditions such as chronic obstructive pulmonary disease and asthma although previous studies have shown an association between asthma and COPD exacerbations and air pollution, Dr. Barrett said.
“Further research will be needed to confirm the associations reported in this study,” Dr. Barrett said.
More research in other countries, including research related to other autoimmune diseases, and with other datasets on population and community level risks from poor air quality, would be helpful, and that information could be used to advise smart public policy, Dr. Barrett added.
Air pollution’s mental health impact
Air pollution’s effects extend beyond physical to the psychological, a new study of depression in teenagers showed. This study was published in Developmental Psychology.
Previous research on the environmental factors associated with depressive symptoms in teens has focused mainly on individual and family level contributors; the impact of the physical environment has not been well studied, the investigators, Erika M. Manczak, PhD, of the University of Denver and colleagues, wrote.
In their paper, the authors found a significant impact of neighborhood ozone exposure on the trajectory of depressive symptoms in teens over a 4-year period.
“Given that inhaling pollution activates biological pathways implicated in the development of depression, including immune, cardiovascular, and neurodevelopmental processes, exposure to ambient air pollution may influence the development and/or trajectory of depressive symptoms in youth,” they said.
The researchers recruited 213 adolescents in the San Francisco Bay area through local advertisements. The participants were aged 9-13 years at baseline, with an average age of 11 years. A total of 121 were female, 47% were white, 8.5% were African American, 12.3% were Asian, 10.4% were nonwhite Latin, and 21.7% were biracial or another ethnicity. The participants self-reported depressive symptoms and other psychopathology symptoms up to three times during the study period. Ozone exposure was calculated based on home addresses.
After controlling for other personal, family, and neighborhood variables, the researchers found that higher levels of ozone exposure were significantly associated with increased depressive symptoms over time, and the slope of trajectory of depressive symptoms became steeper as the ozone levels increased (P less than .001). Ozone did not significantly predict the trajectory of any other psychopathology symptoms.
“The results of this study provide preliminary support for the possibility that ozone is an overlooked contributor to the development or course of youth depressive symptoms,” the researchers wrote in their discussion.
“Interestingly, the association between ozone and symptom trajectories as measured by Anxious/Depressed subscale of the [Youth Self-Report] was not as strong as it was for the [Children’s Depression Inventory-Short Version] or Withdrawn/Depressed scales, suggesting that associations are more robust for behavioral withdrawal symptoms of depression than for other types of symptoms,” they noted.
The study findings were limited by the use of self-reports and by the inability of the study design to show causality, the researchers said. Other limitations include the use of average assessments of ozone that are less precise, lack of assessment of biological pathways for risk, lack of formal psychiatric diagnoses, and the small geographic region included in the study, they said.
However, the results provide preliminary evidence that ozone exposure is a potential contributing factor to depressive symptoms in youth, and serve as a jumping-off point for future research, they noted. Future studies should address changes in systemic inflammation, neurodevelopment, or stress reactivity, as well as concurrent psychosocial or biological factors, and temporal associations between air pollution and mental health symptoms, they concluded.
Environmental factors drive inflammatory responses
Peter L. Loper Jr., MD, considers the findings of the Developmental Psychology study to be unsurprising but important – because air pollution is simply getting worse.
“As the study authors cite, there is sufficient data correlating ozone to negative physical health outcomes in youth, but a paucity of data exploring the impact of poor air quality on mental health outcomes in this demographic,” noted Dr. Loper, of the University of South Carolina, Columbia, in an interview.
“As discussed by the study researchers, any environmental exposure that increases immune-mediated inflammation can result in negative health outcomes. In fact, there is already data to suggest that similar cytokines, or immune cell signalers, that get released by our immune system due to environmental exposures and that contribute to asthma, may also be implicated in depression and other mental health problems,” he noted.
“Just like downstream symptom indicators of physical illnesses such as asthma are secondary to immune-mediated pulmonary inflammation, downstream symptom indicators of mental illness, such as depression, are secondary to immune-mediated neuroinflammation,” Dr. Loper emphasized. “The most well-characterized upstream phenomenon perpetuating the downstream symptom indicators of depression involve neuroinflammatory states due to psychosocial and relational factors such as chronic stress, poor relationships, or substance use. However, any environmental factor that triggers an immune response and inflammation can promote neuroinflammation that manifests as symptoms of mental illness.”
The message for teens with depression and their families is that “we are a product of our environment,” Dr. Loper said. “When our environments are proinflammatory, or cause our immune system to become overactive, then we will develop illness; however, the most potent mediator of inflammation in the brain, and the downstream symptoms of depression, is our relationships with those we love most,” he said.
Dr. Loper suggested research aimed at identifying other sources of immune-mediated inflammation caused by physical environments and better understanding how environmental phenomenon like ozone may compound previously established risk factors for mental illness could be useful.
The RMD Open study received no outside funding, and its authors had no financial conflicts.
The Developmental Psychology study was supported by the National Institute of Mental Health and the Stanford University Precision Health and Integrated Diagnostics Center. The researchers for that report, and Dr. Loper and Dr. Barrett had no conflicts to disclose.
Other analyses of data have found environmental air pollution from sources such as car exhaust and factory output can trigger an inflammatory response in the body. What’s new about a study published in RMD Open is that it explored an association between long-term exposure to pollution and risk of autoimmune diseases, wrote Giovanni Adami, MD, of the University of Verona (Italy) and colleagues.
“Environmental air pollution, according to the World Health Organization, is a major risk to health and 99% of the population worldwide is living in places where recommendations for air quality are not met,” said Dr. Adami in an interview. The limited data on the precise role of air pollution on rheumatic diseases in particular prompted the study, he said.
To explore the potential link between air pollution exposure and autoimmune disease, the researchers reviewed medical information from 81,363 adults via a national medical database in Italy; the data were submitted between June 2016 and November 2020.
The average age of the study population was 65 years, and 92% were women; 22% had at least one coexisting health condition. Each study participant was linked to local environmental monitoring via their residential postcode.
The researchers obtained details about concentrations of particulate matter in the environment from the Italian Institute of Environmental Protection that included 617 monitoring stations in 110 Italian provinces. They focused on concentrations of 10 and 2.5 (PM10 and PM2.5).
Exposure thresholds of 30 mcg/m3 for PM10 and 20 mcg/m3 for PM2.5 are generally considered harmful to health, they noted. On average, the long-term exposure was 16 mcg/m3 for PM2.5 and 25 mcg/m3 for PM10 between 2013 and 2019.
Overall, 9,723 individuals (12%) were diagnosed with an autoimmune disease between 2016 and 2020.
Exposure to PM10 was associated with a 7% higher risk of diagnosis with any autoimmune disease for every 10 mcg/m3 increase in concentration, but no association appeared between PM2.5 exposure and increased risk of autoimmune diseases.
However, in an adjusted model, chronic exposure to PM10 above 30 mcg/m3 and to PM2.5 above 20 mcg/m3 were associated with a 12% and 13% higher risk, respectively, of any autoimmune disease.
Chronic exposure to high levels of PM10 was specifically associated with a higher risk of rheumatoid arthritis, but no other autoimmune diseases. Chronic exposure to high levels of PM2.5 was associated with a higher risk of rheumatoid arthritis, connective tissue diseases, and inflammatory bowel diseases.
In their discussion, the researchers noted that the smaller diameter of PM2.5 molecules fluctuate less in response to rain and other weather, compared with PM10 molecules, which might make them a more accurate predictor of exposure to chronic air pollution.
The study findings were limited by several factors including the observational design, which prohibits the establishment of cause, and a lack of data on the start of symptoms and dates of diagnoses for autoimmune diseases, the researchers noted. Other limitations include the high percentage of older women in the study, which may limit generalizability, and the inability to account for additional personal exposure to pollutants outside of the environmental exposure, they said.
However, the results were strengthened by the large sample size and wide geographic distribution with variable pollution exposure, they said.
“Unfortunately, we were not surprised at all,” by the findings, Dr. Adami said in an interview.
“The biological rationale underpinning our findings is strong. Nevertheless, the magnitude of the effect was overwhelming. In addition, we saw an effect even at threshold of exposure that is widely considered as safe,” Dr. Adami noted.
Clinicians have been taught to consider cigarette smoking or other lifestyle behaviors as major risk factors for the development of several autoimmune diseases, said Dr. Adami. “In the future, we probably should include air pollution exposure as a risk factor as well. Interestingly, there is also accumulating evidence linking acute exposure to environmental air pollution with flares of chronic arthritis,” he said.
“Our study could have direct societal and political consequences,” and might help direct policy makers’ decisions on addressing strategies aimed to reduce fossil emissions, he said. As for additional research, “we certainly need multination studies to confirm our results on a larger scale,” Dr. Adami emphasized. “In addition, it is time to take action and start designing interventions aimed to reduce acute and chronic exposure to air pollution in patients suffering from RMDs.”
Consider the big picture of air quality
The Italian study is especially timely “given our evolving and emerging understanding of environmental risk factors for acute and chronic diseases, which we must first understand before we can address,” said Eileen Barrett, MD, of the University of New Mexico, Albuquerque, in an interview.
“I am largely surprised about the findings, as most physicians aren’t studying ambient air quality and risk for autoimmune disease,” said Dr. Barrett. “More often we think of air quality when we think of risk for respiratory diseases than autoimmune diseases, per se,” she said.
“There are several take-home messages from this study,” said Dr. Barrett. “The first is that we need more research to understand the consequences of air pollutants on health. Second, this study reminds us to think broadly about how air quality and our environment can affect health. And third, all clinicians should be committed to promoting science that can improve public health and reduce death and disability,” she emphasized.
The findings do not specifically reflect associations between pollution and other conditions such as chronic obstructive pulmonary disease and asthma although previous studies have shown an association between asthma and COPD exacerbations and air pollution, Dr. Barrett said.
“Further research will be needed to confirm the associations reported in this study,” Dr. Barrett said.
More research in other countries, including research related to other autoimmune diseases, and with other datasets on population and community level risks from poor air quality, would be helpful, and that information could be used to advise smart public policy, Dr. Barrett added.
Air pollution’s mental health impact
Air pollution’s effects extend beyond physical to the psychological, a new study of depression in teenagers showed. This study was published in Developmental Psychology.
Previous research on the environmental factors associated with depressive symptoms in teens has focused mainly on individual and family level contributors; the impact of the physical environment has not been well studied, the investigators, Erika M. Manczak, PhD, of the University of Denver and colleagues, wrote.
In their paper, the authors found a significant impact of neighborhood ozone exposure on the trajectory of depressive symptoms in teens over a 4-year period.
“Given that inhaling pollution activates biological pathways implicated in the development of depression, including immune, cardiovascular, and neurodevelopmental processes, exposure to ambient air pollution may influence the development and/or trajectory of depressive symptoms in youth,” they said.
The researchers recruited 213 adolescents in the San Francisco Bay area through local advertisements. The participants were aged 9-13 years at baseline, with an average age of 11 years. A total of 121 were female, 47% were white, 8.5% were African American, 12.3% were Asian, 10.4% were nonwhite Latin, and 21.7% were biracial or another ethnicity. The participants self-reported depressive symptoms and other psychopathology symptoms up to three times during the study period. Ozone exposure was calculated based on home addresses.
After controlling for other personal, family, and neighborhood variables, the researchers found that higher levels of ozone exposure were significantly associated with increased depressive symptoms over time, and the slope of trajectory of depressive symptoms became steeper as the ozone levels increased (P less than .001). Ozone did not significantly predict the trajectory of any other psychopathology symptoms.
“The results of this study provide preliminary support for the possibility that ozone is an overlooked contributor to the development or course of youth depressive symptoms,” the researchers wrote in their discussion.
“Interestingly, the association between ozone and symptom trajectories as measured by Anxious/Depressed subscale of the [Youth Self-Report] was not as strong as it was for the [Children’s Depression Inventory-Short Version] or Withdrawn/Depressed scales, suggesting that associations are more robust for behavioral withdrawal symptoms of depression than for other types of symptoms,” they noted.
The study findings were limited by the use of self-reports and by the inability of the study design to show causality, the researchers said. Other limitations include the use of average assessments of ozone that are less precise, lack of assessment of biological pathways for risk, lack of formal psychiatric diagnoses, and the small geographic region included in the study, they said.
However, the results provide preliminary evidence that ozone exposure is a potential contributing factor to depressive symptoms in youth, and serve as a jumping-off point for future research, they noted. Future studies should address changes in systemic inflammation, neurodevelopment, or stress reactivity, as well as concurrent psychosocial or biological factors, and temporal associations between air pollution and mental health symptoms, they concluded.
Environmental factors drive inflammatory responses
Peter L. Loper Jr., MD, considers the findings of the Developmental Psychology study to be unsurprising but important – because air pollution is simply getting worse.
“As the study authors cite, there is sufficient data correlating ozone to negative physical health outcomes in youth, but a paucity of data exploring the impact of poor air quality on mental health outcomes in this demographic,” noted Dr. Loper, of the University of South Carolina, Columbia, in an interview.
“As discussed by the study researchers, any environmental exposure that increases immune-mediated inflammation can result in negative health outcomes. In fact, there is already data to suggest that similar cytokines, or immune cell signalers, that get released by our immune system due to environmental exposures and that contribute to asthma, may also be implicated in depression and other mental health problems,” he noted.
“Just like downstream symptom indicators of physical illnesses such as asthma are secondary to immune-mediated pulmonary inflammation, downstream symptom indicators of mental illness, such as depression, are secondary to immune-mediated neuroinflammation,” Dr. Loper emphasized. “The most well-characterized upstream phenomenon perpetuating the downstream symptom indicators of depression involve neuroinflammatory states due to psychosocial and relational factors such as chronic stress, poor relationships, or substance use. However, any environmental factor that triggers an immune response and inflammation can promote neuroinflammation that manifests as symptoms of mental illness.”
The message for teens with depression and their families is that “we are a product of our environment,” Dr. Loper said. “When our environments are proinflammatory, or cause our immune system to become overactive, then we will develop illness; however, the most potent mediator of inflammation in the brain, and the downstream symptoms of depression, is our relationships with those we love most,” he said.
Dr. Loper suggested research aimed at identifying other sources of immune-mediated inflammation caused by physical environments and better understanding how environmental phenomenon like ozone may compound previously established risk factors for mental illness could be useful.
The RMD Open study received no outside funding, and its authors had no financial conflicts.
The Developmental Psychology study was supported by the National Institute of Mental Health and the Stanford University Precision Health and Integrated Diagnostics Center. The researchers for that report, and Dr. Loper and Dr. Barrett had no conflicts to disclose.
Other analyses of data have found environmental air pollution from sources such as car exhaust and factory output can trigger an inflammatory response in the body. What’s new about a study published in RMD Open is that it explored an association between long-term exposure to pollution and risk of autoimmune diseases, wrote Giovanni Adami, MD, of the University of Verona (Italy) and colleagues.
“Environmental air pollution, according to the World Health Organization, is a major risk to health and 99% of the population worldwide is living in places where recommendations for air quality are not met,” said Dr. Adami in an interview. The limited data on the precise role of air pollution on rheumatic diseases in particular prompted the study, he said.
To explore the potential link between air pollution exposure and autoimmune disease, the researchers reviewed medical information from 81,363 adults via a national medical database in Italy; the data were submitted between June 2016 and November 2020.
The average age of the study population was 65 years, and 92% were women; 22% had at least one coexisting health condition. Each study participant was linked to local environmental monitoring via their residential postcode.
The researchers obtained details about concentrations of particulate matter in the environment from the Italian Institute of Environmental Protection that included 617 monitoring stations in 110 Italian provinces. They focused on concentrations of 10 and 2.5 (PM10 and PM2.5).
Exposure thresholds of 30 mcg/m3 for PM10 and 20 mcg/m3 for PM2.5 are generally considered harmful to health, they noted. On average, the long-term exposure was 16 mcg/m3 for PM2.5 and 25 mcg/m3 for PM10 between 2013 and 2019.
Overall, 9,723 individuals (12%) were diagnosed with an autoimmune disease between 2016 and 2020.
Exposure to PM10 was associated with a 7% higher risk of diagnosis with any autoimmune disease for every 10 mcg/m3 increase in concentration, but no association appeared between PM2.5 exposure and increased risk of autoimmune diseases.
However, in an adjusted model, chronic exposure to PM10 above 30 mcg/m3 and to PM2.5 above 20 mcg/m3 were associated with a 12% and 13% higher risk, respectively, of any autoimmune disease.
Chronic exposure to high levels of PM10 was specifically associated with a higher risk of rheumatoid arthritis, but no other autoimmune diseases. Chronic exposure to high levels of PM2.5 was associated with a higher risk of rheumatoid arthritis, connective tissue diseases, and inflammatory bowel diseases.
In their discussion, the researchers noted that the smaller diameter of PM2.5 molecules fluctuate less in response to rain and other weather, compared with PM10 molecules, which might make them a more accurate predictor of exposure to chronic air pollution.
The study findings were limited by several factors including the observational design, which prohibits the establishment of cause, and a lack of data on the start of symptoms and dates of diagnoses for autoimmune diseases, the researchers noted. Other limitations include the high percentage of older women in the study, which may limit generalizability, and the inability to account for additional personal exposure to pollutants outside of the environmental exposure, they said.
However, the results were strengthened by the large sample size and wide geographic distribution with variable pollution exposure, they said.
“Unfortunately, we were not surprised at all,” by the findings, Dr. Adami said in an interview.
“The biological rationale underpinning our findings is strong. Nevertheless, the magnitude of the effect was overwhelming. In addition, we saw an effect even at threshold of exposure that is widely considered as safe,” Dr. Adami noted.
Clinicians have been taught to consider cigarette smoking or other lifestyle behaviors as major risk factors for the development of several autoimmune diseases, said Dr. Adami. “In the future, we probably should include air pollution exposure as a risk factor as well. Interestingly, there is also accumulating evidence linking acute exposure to environmental air pollution with flares of chronic arthritis,” he said.
“Our study could have direct societal and political consequences,” and might help direct policy makers’ decisions on addressing strategies aimed to reduce fossil emissions, he said. As for additional research, “we certainly need multination studies to confirm our results on a larger scale,” Dr. Adami emphasized. “In addition, it is time to take action and start designing interventions aimed to reduce acute and chronic exposure to air pollution in patients suffering from RMDs.”
Consider the big picture of air quality
The Italian study is especially timely “given our evolving and emerging understanding of environmental risk factors for acute and chronic diseases, which we must first understand before we can address,” said Eileen Barrett, MD, of the University of New Mexico, Albuquerque, in an interview.
“I am largely surprised about the findings, as most physicians aren’t studying ambient air quality and risk for autoimmune disease,” said Dr. Barrett. “More often we think of air quality when we think of risk for respiratory diseases than autoimmune diseases, per se,” she said.
“There are several take-home messages from this study,” said Dr. Barrett. “The first is that we need more research to understand the consequences of air pollutants on health. Second, this study reminds us to think broadly about how air quality and our environment can affect health. And third, all clinicians should be committed to promoting science that can improve public health and reduce death and disability,” she emphasized.
The findings do not specifically reflect associations between pollution and other conditions such as chronic obstructive pulmonary disease and asthma although previous studies have shown an association between asthma and COPD exacerbations and air pollution, Dr. Barrett said.
“Further research will be needed to confirm the associations reported in this study,” Dr. Barrett said.
More research in other countries, including research related to other autoimmune diseases, and with other datasets on population and community level risks from poor air quality, would be helpful, and that information could be used to advise smart public policy, Dr. Barrett added.
Air pollution’s mental health impact
Air pollution’s effects extend beyond physical to the psychological, a new study of depression in teenagers showed. This study was published in Developmental Psychology.
Previous research on the environmental factors associated with depressive symptoms in teens has focused mainly on individual and family level contributors; the impact of the physical environment has not been well studied, the investigators, Erika M. Manczak, PhD, of the University of Denver and colleagues, wrote.
In their paper, the authors found a significant impact of neighborhood ozone exposure on the trajectory of depressive symptoms in teens over a 4-year period.
“Given that inhaling pollution activates biological pathways implicated in the development of depression, including immune, cardiovascular, and neurodevelopmental processes, exposure to ambient air pollution may influence the development and/or trajectory of depressive symptoms in youth,” they said.
The researchers recruited 213 adolescents in the San Francisco Bay area through local advertisements. The participants were aged 9-13 years at baseline, with an average age of 11 years. A total of 121 were female, 47% were white, 8.5% were African American, 12.3% were Asian, 10.4% were nonwhite Latin, and 21.7% were biracial or another ethnicity. The participants self-reported depressive symptoms and other psychopathology symptoms up to three times during the study period. Ozone exposure was calculated based on home addresses.
After controlling for other personal, family, and neighborhood variables, the researchers found that higher levels of ozone exposure were significantly associated with increased depressive symptoms over time, and the slope of trajectory of depressive symptoms became steeper as the ozone levels increased (P less than .001). Ozone did not significantly predict the trajectory of any other psychopathology symptoms.
“The results of this study provide preliminary support for the possibility that ozone is an overlooked contributor to the development or course of youth depressive symptoms,” the researchers wrote in their discussion.
“Interestingly, the association between ozone and symptom trajectories as measured by Anxious/Depressed subscale of the [Youth Self-Report] was not as strong as it was for the [Children’s Depression Inventory-Short Version] or Withdrawn/Depressed scales, suggesting that associations are more robust for behavioral withdrawal symptoms of depression than for other types of symptoms,” they noted.
The study findings were limited by the use of self-reports and by the inability of the study design to show causality, the researchers said. Other limitations include the use of average assessments of ozone that are less precise, lack of assessment of biological pathways for risk, lack of formal psychiatric diagnoses, and the small geographic region included in the study, they said.
However, the results provide preliminary evidence that ozone exposure is a potential contributing factor to depressive symptoms in youth, and serve as a jumping-off point for future research, they noted. Future studies should address changes in systemic inflammation, neurodevelopment, or stress reactivity, as well as concurrent psychosocial or biological factors, and temporal associations between air pollution and mental health symptoms, they concluded.
Environmental factors drive inflammatory responses
Peter L. Loper Jr., MD, considers the findings of the Developmental Psychology study to be unsurprising but important – because air pollution is simply getting worse.
“As the study authors cite, there is sufficient data correlating ozone to negative physical health outcomes in youth, but a paucity of data exploring the impact of poor air quality on mental health outcomes in this demographic,” noted Dr. Loper, of the University of South Carolina, Columbia, in an interview.
“As discussed by the study researchers, any environmental exposure that increases immune-mediated inflammation can result in negative health outcomes. In fact, there is already data to suggest that similar cytokines, or immune cell signalers, that get released by our immune system due to environmental exposures and that contribute to asthma, may also be implicated in depression and other mental health problems,” he noted.
“Just like downstream symptom indicators of physical illnesses such as asthma are secondary to immune-mediated pulmonary inflammation, downstream symptom indicators of mental illness, such as depression, are secondary to immune-mediated neuroinflammation,” Dr. Loper emphasized. “The most well-characterized upstream phenomenon perpetuating the downstream symptom indicators of depression involve neuroinflammatory states due to psychosocial and relational factors such as chronic stress, poor relationships, or substance use. However, any environmental factor that triggers an immune response and inflammation can promote neuroinflammation that manifests as symptoms of mental illness.”
The message for teens with depression and their families is that “we are a product of our environment,” Dr. Loper said. “When our environments are proinflammatory, or cause our immune system to become overactive, then we will develop illness; however, the most potent mediator of inflammation in the brain, and the downstream symptoms of depression, is our relationships with those we love most,” he said.
Dr. Loper suggested research aimed at identifying other sources of immune-mediated inflammation caused by physical environments and better understanding how environmental phenomenon like ozone may compound previously established risk factors for mental illness could be useful.
The RMD Open study received no outside funding, and its authors had no financial conflicts.
The Developmental Psychology study was supported by the National Institute of Mental Health and the Stanford University Precision Health and Integrated Diagnostics Center. The researchers for that report, and Dr. Loper and Dr. Barrett had no conflicts to disclose.
FROM RMD OPEN
FDA approves generic Symbicort for asthma, COPD
The U.S. Food and Drug Administration approved the first generic of Symbicort (budesonide and formoterol fumarate dihydrate) inhalation aerosol for the treatment of asthma in patients 6 years of age and older and for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
The approval was given for a complex generic drug-device combination product – a metered-dose inhaler that contains both budesonide (a corticosteroid that reduces inflammation) and formoterol (a long-acting bronchodilator that relaxes muscles in the airways to improve breathing). It is intended to be used as two inhalations, two times a day (usually morning and night, about 12 hours apart), to treat both diseases by preventing symptoms, such as wheezing for those with asthma and for improved breathing for patients with COPD.
The inhaler is approved at two strengths (160/4.5 mcg/actuation and 80/4.5 mcg/actuation), according to the March 15 FDA announcement. The device is not intended for the treatment of acute asthma.
“Today’s approval of the first generic for one of the most commonly prescribed complex drug-device combination products to treat asthma and COPD is another step forward in our commitment to bring generic copies of complex drugs to the market, which can improve quality of life and help reduce the cost of treatment,” said Sally Choe, PhD, director of the Office of Generic Drugs in the FDA’s Center for Drug Evaluation and Research.
The most common side effects associated with budesonide and formoterol fumarate dihydrate oral inhalation aerosol for those with asthma are nasopharyngitis pain, sinusitis, influenza, back pain, nasal congestion, stomach discomfort, vomiting, and oral candidiasis (thrush). For those with COPD, the most common side effects are nasopharyngitis, oral candidiasis, bronchitis, sinusitis, and upper respiratory tract infection, the FDA reported.
The approval of this generic drug-device combination was granted to Mylan Pharmaceuticals.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration approved the first generic of Symbicort (budesonide and formoterol fumarate dihydrate) inhalation aerosol for the treatment of asthma in patients 6 years of age and older and for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
The approval was given for a complex generic drug-device combination product – a metered-dose inhaler that contains both budesonide (a corticosteroid that reduces inflammation) and formoterol (a long-acting bronchodilator that relaxes muscles in the airways to improve breathing). It is intended to be used as two inhalations, two times a day (usually morning and night, about 12 hours apart), to treat both diseases by preventing symptoms, such as wheezing for those with asthma and for improved breathing for patients with COPD.
The inhaler is approved at two strengths (160/4.5 mcg/actuation and 80/4.5 mcg/actuation), according to the March 15 FDA announcement. The device is not intended for the treatment of acute asthma.
“Today’s approval of the first generic for one of the most commonly prescribed complex drug-device combination products to treat asthma and COPD is another step forward in our commitment to bring generic copies of complex drugs to the market, which can improve quality of life and help reduce the cost of treatment,” said Sally Choe, PhD, director of the Office of Generic Drugs in the FDA’s Center for Drug Evaluation and Research.
The most common side effects associated with budesonide and formoterol fumarate dihydrate oral inhalation aerosol for those with asthma are nasopharyngitis pain, sinusitis, influenza, back pain, nasal congestion, stomach discomfort, vomiting, and oral candidiasis (thrush). For those with COPD, the most common side effects are nasopharyngitis, oral candidiasis, bronchitis, sinusitis, and upper respiratory tract infection, the FDA reported.
The approval of this generic drug-device combination was granted to Mylan Pharmaceuticals.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration approved the first generic of Symbicort (budesonide and formoterol fumarate dihydrate) inhalation aerosol for the treatment of asthma in patients 6 years of age and older and for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
The approval was given for a complex generic drug-device combination product – a metered-dose inhaler that contains both budesonide (a corticosteroid that reduces inflammation) and formoterol (a long-acting bronchodilator that relaxes muscles in the airways to improve breathing). It is intended to be used as two inhalations, two times a day (usually morning and night, about 12 hours apart), to treat both diseases by preventing symptoms, such as wheezing for those with asthma and for improved breathing for patients with COPD.
The inhaler is approved at two strengths (160/4.5 mcg/actuation and 80/4.5 mcg/actuation), according to the March 15 FDA announcement. The device is not intended for the treatment of acute asthma.
“Today’s approval of the first generic for one of the most commonly prescribed complex drug-device combination products to treat asthma and COPD is another step forward in our commitment to bring generic copies of complex drugs to the market, which can improve quality of life and help reduce the cost of treatment,” said Sally Choe, PhD, director of the Office of Generic Drugs in the FDA’s Center for Drug Evaluation and Research.
The most common side effects associated with budesonide and formoterol fumarate dihydrate oral inhalation aerosol for those with asthma are nasopharyngitis pain, sinusitis, influenza, back pain, nasal congestion, stomach discomfort, vomiting, and oral candidiasis (thrush). For those with COPD, the most common side effects are nasopharyngitis, oral candidiasis, bronchitis, sinusitis, and upper respiratory tract infection, the FDA reported.
The approval of this generic drug-device combination was granted to Mylan Pharmaceuticals.
A version of this article first appeared on Medscape.com.






