User login
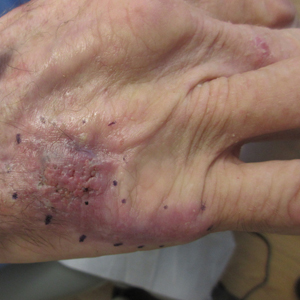
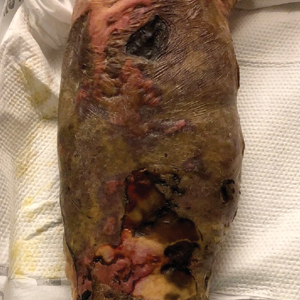
Nonhealing Violaceous Plaque of the Hand Following a Splinter Injury
The Diagnosis: Chromoblastomycosis
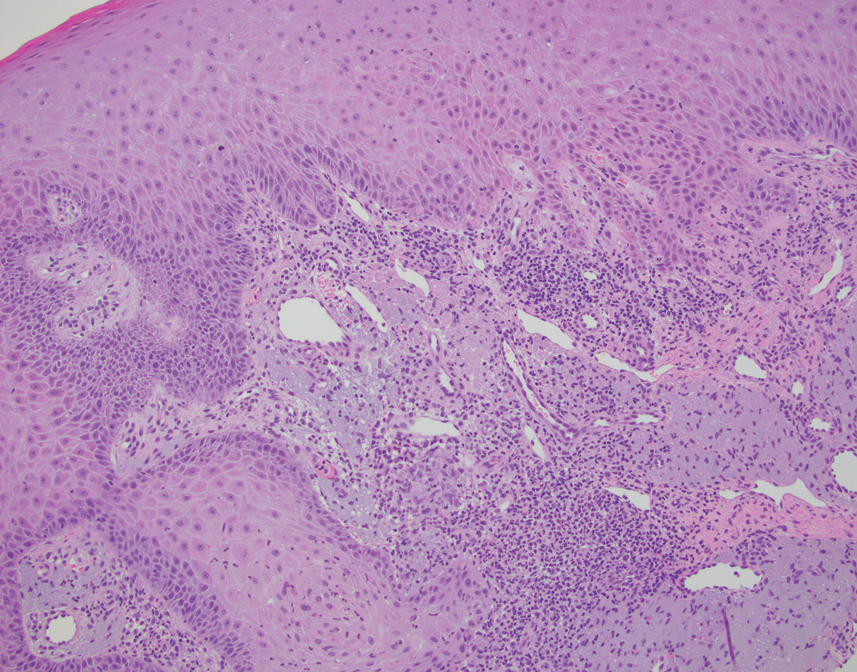
This case highlights the importance of routine skin biopsy and tissue culture when clinical suspicion for mycotic infection is high. Despite nonspecific biopsy results (Figure), a diagnosis of chromoblastomycosis (CBM) was reached based on tissue culture. Surgical excision was not possible in our patient due to the size and location of the lesion. The patient was referred to infectious disease, with the plan to start long-term itraconazole for at least 6 to 12 months.
Cases of CBM were first documented in 1914 and distinguished by the appearance of spherical, brown, muriform cells on skin biopsy—features that now serve as the hallmark of CBM diagnoses.1,2 The implantation mycosis commonly is caused by agents such as Fonsecaea pedrosoi and Fonsecaea monophora of the bantiana-clade, as classified according to molecular phylogeny2; these agents have been isolated from soil, plants, and wood sources in tropical and subtropical regions and are strongly associated with agricultural activities.3
Chromoblastomycosis lesions tend to be asymptomatic with a variable amount of time between inoculation and lesion presentation, delaying medical care by months to years.3 The fungus causes a granulomatous reaction after skin damage, with noticeable pseudoepitheliomatous hyperplasia of the epidermis and granulomas formed by epithelioid and Langerhans cells in the dermis.4 Typically, CBM initially presents as an erythematous macular skin lesion, which then progresses to become more pink, papular, and sometimes pruritic.2 Muriform (sclerotic) bodies, which reflect fungal components, extrude transepidermally and appear as black dots on the lesion’s surface.4 Chromoblastomycosis is limited to the subcutaneous tissue and has been classified into 5 types of lesions: nodular, tumoral, verrucous, scarring, and plaque.2 Diagnosis is established using fungal tests such as potassium hydroxide direct microscopy, which exposes muriform bodies often in combination with dematiaceous hyphae, while fungal culture of F pedrosoi in Sabouraud agar produces velvety dark colonies.3 Although an immune response to CBM infection remains unclear, it has been demonstrated that the response differs based on the severity of the infection. The severe form of CBM produces high levels of IL-10, low levels of IFN-γ, and inefficient T-cell proliferation, while milder forms of CBM display low levels of IL-10, high levels of IFN-γ, and efficient T-cell proliferation.5 Complications of CBM include chronic lymphedema, ankylosis, and secondary bacterial infections, which largely are observed in advanced cases; malignant transformation to squamous cell carcinoma, though rare, also has been observed.6
Several therapeutic methods have been implemented in the treatment of CBM, but lesions often remain refractory, especially in advanced cases.6 Approaches to treatment can be divided into antifungal and physical methods. Commonly employed antifungal agents include itraconazole and terbinafine, which must be taken daily for a period ranging from 6 months to 1 year or longer; flucytosine with or without amphotericin also has been employed.4 Among the physical methods, surgical excision is not suggested due to possible dissemination of disease; other options include cryotherapy, thermotherapy, and laser vaporization.6 The prognosis has improved since the use of extended-spectrum triazoles, but high rates of refractory disease remain unchanged.2
The differential diagnosis includes other infections. Nocardiosis is a bacterial infection in which cutaneous disease can result in actinomycetoma, which presents with grains that are small, round, and stain blue on hematoxylin and eosin with eosinophilic rays at the periphery.7 Although the clinical features and pseudoepitheliomatous hyperplasia seen in CBM can mimic squamous cell carcinoma, the latter would show variable degrees of differentiation, keratinization, nuclear atypia, and architectural atypia with a negative tissue culture.8 Eumycetoma is a fungal infection that typically is not caused by F pedrosoi but rather most commonly Madurella mycetomatis.9 Leishmaniasis is a parasitic infection in which a biopsy of cutaneous lesions often displays parasite-filled histiocytes.10
- Rudolph M. Über die brasilianische “figueira” (vorläufige mitteilung). Arch Schiffs Trop Hyg. 1914;18:498-499.
- Queiroz-Telles F, de Hoog S, Santos DW, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276. doi:10.1128/CMR.00032-16
- Brito AC, Bittencourt MJS. Chromoblastomycosis: an etiological, epidemiological, clinical, diagnostic, and treatment update. An Bras Dermatol. 2018;93:495-506. doi:10.1590/abd1806-4841.20187321
- Kurien G, Sugumar K, Chandran V. Chromoblastomycosis. StatPearls. StatPearls Publishing; 2021. Accessed June 4, 2022. https://www.ncbi.nlm.nih.gov/books/NBK470253/
- Mazo Fávero Gimenes V, Da Glória de Souza M, Ferreira KS, et al. Cytokines and lymphocyte proliferation in patients with different clinical forms of chromoblastomycosis. Microbes Infect. 2005;7:708-713. doi:10.1016/j.micinf.2005.01.006
- Krzys´ciak PM, Pindycka-Piaszczyn´ska M, Piaszczyn´ski M. Chromoblastomycosis. Postepy Dermatol Alergol. 2014;31:310-321. doi:10.5114/pdia.2014.40949
- Siddig EE, van de Sande WWJ, Fahal AH. Actinomycetoma laboratory-based diagnosis: a mini-review. Trans R Soc Trop Med Hyg. 2021;115:355-363.
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll .2017.06.003
- Nenoff P, van de Sande WWJ, Fahal AH, et al. Eumycetoma and actinomycetoma—an update on causative agents, epidemiology, pathogenesis, diagnostics and therapy. J Eur Acad Dermatol Venereol. 2015;29:1873-1883. doi:10.1111/jdv.13008
- Saliba M, Shalhoub A, Taraif S, et al. Cutaneous leishmaniasis: an evolving disease with ancient roots. Int J Dermatol. 2019;58:834-843. doi:10.1111/ijd.14451
The Diagnosis: Chromoblastomycosis
This case highlights the importance of routine skin biopsy and tissue culture when clinical suspicion for mycotic infection is high. Despite nonspecific biopsy results (Figure), a diagnosis of chromoblastomycosis (CBM) was reached based on tissue culture. Surgical excision was not possible in our patient due to the size and location of the lesion. The patient was referred to infectious disease, with the plan to start long-term itraconazole for at least 6 to 12 months.

Cases of CBM were first documented in 1914 and distinguished by the appearance of spherical, brown, muriform cells on skin biopsy—features that now serve as the hallmark of CBM diagnoses.1,2 The implantation mycosis commonly is caused by agents such as Fonsecaea pedrosoi and Fonsecaea monophora of the bantiana-clade, as classified according to molecular phylogeny2; these agents have been isolated from soil, plants, and wood sources in tropical and subtropical regions and are strongly associated with agricultural activities.3
Chromoblastomycosis lesions tend to be asymptomatic with a variable amount of time between inoculation and lesion presentation, delaying medical care by months to years.3 The fungus causes a granulomatous reaction after skin damage, with noticeable pseudoepitheliomatous hyperplasia of the epidermis and granulomas formed by epithelioid and Langerhans cells in the dermis.4 Typically, CBM initially presents as an erythematous macular skin lesion, which then progresses to become more pink, papular, and sometimes pruritic.2 Muriform (sclerotic) bodies, which reflect fungal components, extrude transepidermally and appear as black dots on the lesion’s surface.4 Chromoblastomycosis is limited to the subcutaneous tissue and has been classified into 5 types of lesions: nodular, tumoral, verrucous, scarring, and plaque.2 Diagnosis is established using fungal tests such as potassium hydroxide direct microscopy, which exposes muriform bodies often in combination with dematiaceous hyphae, while fungal culture of F pedrosoi in Sabouraud agar produces velvety dark colonies.3 Although an immune response to CBM infection remains unclear, it has been demonstrated that the response differs based on the severity of the infection. The severe form of CBM produces high levels of IL-10, low levels of IFN-γ, and inefficient T-cell proliferation, while milder forms of CBM display low levels of IL-10, high levels of IFN-γ, and efficient T-cell proliferation.5 Complications of CBM include chronic lymphedema, ankylosis, and secondary bacterial infections, which largely are observed in advanced cases; malignant transformation to squamous cell carcinoma, though rare, also has been observed.6
Several therapeutic methods have been implemented in the treatment of CBM, but lesions often remain refractory, especially in advanced cases.6 Approaches to treatment can be divided into antifungal and physical methods. Commonly employed antifungal agents include itraconazole and terbinafine, which must be taken daily for a period ranging from 6 months to 1 year or longer; flucytosine with or without amphotericin also has been employed.4 Among the physical methods, surgical excision is not suggested due to possible dissemination of disease; other options include cryotherapy, thermotherapy, and laser vaporization.6 The prognosis has improved since the use of extended-spectrum triazoles, but high rates of refractory disease remain unchanged.2
The differential diagnosis includes other infections. Nocardiosis is a bacterial infection in which cutaneous disease can result in actinomycetoma, which presents with grains that are small, round, and stain blue on hematoxylin and eosin with eosinophilic rays at the periphery.7 Although the clinical features and pseudoepitheliomatous hyperplasia seen in CBM can mimic squamous cell carcinoma, the latter would show variable degrees of differentiation, keratinization, nuclear atypia, and architectural atypia with a negative tissue culture.8 Eumycetoma is a fungal infection that typically is not caused by F pedrosoi but rather most commonly Madurella mycetomatis.9 Leishmaniasis is a parasitic infection in which a biopsy of cutaneous lesions often displays parasite-filled histiocytes.10
The Diagnosis: Chromoblastomycosis
This case highlights the importance of routine skin biopsy and tissue culture when clinical suspicion for mycotic infection is high. Despite nonspecific biopsy results (Figure), a diagnosis of chromoblastomycosis (CBM) was reached based on tissue culture. Surgical excision was not possible in our patient due to the size and location of the lesion. The patient was referred to infectious disease, with the plan to start long-term itraconazole for at least 6 to 12 months.

Cases of CBM were first documented in 1914 and distinguished by the appearance of spherical, brown, muriform cells on skin biopsy—features that now serve as the hallmark of CBM diagnoses.1,2 The implantation mycosis commonly is caused by agents such as Fonsecaea pedrosoi and Fonsecaea monophora of the bantiana-clade, as classified according to molecular phylogeny2; these agents have been isolated from soil, plants, and wood sources in tropical and subtropical regions and are strongly associated with agricultural activities.3
Chromoblastomycosis lesions tend to be asymptomatic with a variable amount of time between inoculation and lesion presentation, delaying medical care by months to years.3 The fungus causes a granulomatous reaction after skin damage, with noticeable pseudoepitheliomatous hyperplasia of the epidermis and granulomas formed by epithelioid and Langerhans cells in the dermis.4 Typically, CBM initially presents as an erythematous macular skin lesion, which then progresses to become more pink, papular, and sometimes pruritic.2 Muriform (sclerotic) bodies, which reflect fungal components, extrude transepidermally and appear as black dots on the lesion’s surface.4 Chromoblastomycosis is limited to the subcutaneous tissue and has been classified into 5 types of lesions: nodular, tumoral, verrucous, scarring, and plaque.2 Diagnosis is established using fungal tests such as potassium hydroxide direct microscopy, which exposes muriform bodies often in combination with dematiaceous hyphae, while fungal culture of F pedrosoi in Sabouraud agar produces velvety dark colonies.3 Although an immune response to CBM infection remains unclear, it has been demonstrated that the response differs based on the severity of the infection. The severe form of CBM produces high levels of IL-10, low levels of IFN-γ, and inefficient T-cell proliferation, while milder forms of CBM display low levels of IL-10, high levels of IFN-γ, and efficient T-cell proliferation.5 Complications of CBM include chronic lymphedema, ankylosis, and secondary bacterial infections, which largely are observed in advanced cases; malignant transformation to squamous cell carcinoma, though rare, also has been observed.6
Several therapeutic methods have been implemented in the treatment of CBM, but lesions often remain refractory, especially in advanced cases.6 Approaches to treatment can be divided into antifungal and physical methods. Commonly employed antifungal agents include itraconazole and terbinafine, which must be taken daily for a period ranging from 6 months to 1 year or longer; flucytosine with or without amphotericin also has been employed.4 Among the physical methods, surgical excision is not suggested due to possible dissemination of disease; other options include cryotherapy, thermotherapy, and laser vaporization.6 The prognosis has improved since the use of extended-spectrum triazoles, but high rates of refractory disease remain unchanged.2
The differential diagnosis includes other infections. Nocardiosis is a bacterial infection in which cutaneous disease can result in actinomycetoma, which presents with grains that are small, round, and stain blue on hematoxylin and eosin with eosinophilic rays at the periphery.7 Although the clinical features and pseudoepitheliomatous hyperplasia seen in CBM can mimic squamous cell carcinoma, the latter would show variable degrees of differentiation, keratinization, nuclear atypia, and architectural atypia with a negative tissue culture.8 Eumycetoma is a fungal infection that typically is not caused by F pedrosoi but rather most commonly Madurella mycetomatis.9 Leishmaniasis is a parasitic infection in which a biopsy of cutaneous lesions often displays parasite-filled histiocytes.10
- Rudolph M. Über die brasilianische “figueira” (vorläufige mitteilung). Arch Schiffs Trop Hyg. 1914;18:498-499.
- Queiroz-Telles F, de Hoog S, Santos DW, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276. doi:10.1128/CMR.00032-16
- Brito AC, Bittencourt MJS. Chromoblastomycosis: an etiological, epidemiological, clinical, diagnostic, and treatment update. An Bras Dermatol. 2018;93:495-506. doi:10.1590/abd1806-4841.20187321
- Kurien G, Sugumar K, Chandran V. Chromoblastomycosis. StatPearls. StatPearls Publishing; 2021. Accessed June 4, 2022. https://www.ncbi.nlm.nih.gov/books/NBK470253/
- Mazo Fávero Gimenes V, Da Glória de Souza M, Ferreira KS, et al. Cytokines and lymphocyte proliferation in patients with different clinical forms of chromoblastomycosis. Microbes Infect. 2005;7:708-713. doi:10.1016/j.micinf.2005.01.006
- Krzys´ciak PM, Pindycka-Piaszczyn´ska M, Piaszczyn´ski M. Chromoblastomycosis. Postepy Dermatol Alergol. 2014;31:310-321. doi:10.5114/pdia.2014.40949
- Siddig EE, van de Sande WWJ, Fahal AH. Actinomycetoma laboratory-based diagnosis: a mini-review. Trans R Soc Trop Med Hyg. 2021;115:355-363.
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll .2017.06.003
- Nenoff P, van de Sande WWJ, Fahal AH, et al. Eumycetoma and actinomycetoma—an update on causative agents, epidemiology, pathogenesis, diagnostics and therapy. J Eur Acad Dermatol Venereol. 2015;29:1873-1883. doi:10.1111/jdv.13008
- Saliba M, Shalhoub A, Taraif S, et al. Cutaneous leishmaniasis: an evolving disease with ancient roots. Int J Dermatol. 2019;58:834-843. doi:10.1111/ijd.14451
- Rudolph M. Über die brasilianische “figueira” (vorläufige mitteilung). Arch Schiffs Trop Hyg. 1914;18:498-499.
- Queiroz-Telles F, de Hoog S, Santos DW, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30:233-276. doi:10.1128/CMR.00032-16
- Brito AC, Bittencourt MJS. Chromoblastomycosis: an etiological, epidemiological, clinical, diagnostic, and treatment update. An Bras Dermatol. 2018;93:495-506. doi:10.1590/abd1806-4841.20187321
- Kurien G, Sugumar K, Chandran V. Chromoblastomycosis. StatPearls. StatPearls Publishing; 2021. Accessed June 4, 2022. https://www.ncbi.nlm.nih.gov/books/NBK470253/
- Mazo Fávero Gimenes V, Da Glória de Souza M, Ferreira KS, et al. Cytokines and lymphocyte proliferation in patients with different clinical forms of chromoblastomycosis. Microbes Infect. 2005;7:708-713. doi:10.1016/j.micinf.2005.01.006
- Krzys´ciak PM, Pindycka-Piaszczyn´ska M, Piaszczyn´ski M. Chromoblastomycosis. Postepy Dermatol Alergol. 2014;31:310-321. doi:10.5114/pdia.2014.40949
- Siddig EE, van de Sande WWJ, Fahal AH. Actinomycetoma laboratory-based diagnosis: a mini-review. Trans R Soc Trop Med Hyg. 2021;115:355-363.
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525. doi:10.1016/j.cll .2017.06.003
- Nenoff P, van de Sande WWJ, Fahal AH, et al. Eumycetoma and actinomycetoma—an update on causative agents, epidemiology, pathogenesis, diagnostics and therapy. J Eur Acad Dermatol Venereol. 2015;29:1873-1883. doi:10.1111/jdv.13008
- Saliba M, Shalhoub A, Taraif S, et al. Cutaneous leishmaniasis: an evolving disease with ancient roots. Int J Dermatol. 2019;58:834-843. doi:10.1111/ijd.14451
A 70-year-old immunocompetent man presented to the dermatology department with a progressive asymptomatic hand wound of 2 years’ duration following a splinter injury in Belize. Prior treatment included oral antibiotics without improvement. Physical examination revealed a 5.1×3.0 cm, pink to violaceous, nonpurulent plaque with a cobblestonelike appearance on the dorsal aspect of the right hand. Both the initial and a repeat skin biopsy revealed nonspecific changes, including hyperkeratosis, hypergranulosis, acute and chronic inflammation, and vascular ectasia. Grocott-Gomori methenamine-silver staining was negative for fungal organisms. One month after the repeat biopsy, a tissue culture returned positive for the rare Fonsecaea pedrosoi.
Postirradiation Pseudosclerodermatous Panniculitis: A Rare Complication of Megavoltage External Beam Radiotherapy
To the Editor:
Postirradiation pseudosclerodermatous panniculitis (PIPP) is a rarely reported complication of megavoltage external beam radiotherapy that was first identified in 1993 by Winkelmann et al.1 The condition presents as an erythematous or hyperpigmented indurated plaque at a site of prior radiotherapy. Lesions caused by PIPP most commonly arise several months after treatment, although they may emerge up to 17 years following exposure.2 Herein, we report a rare case of a patient with PIPP occurring on the leg who previously had been treated for Kaposi sarcoma.
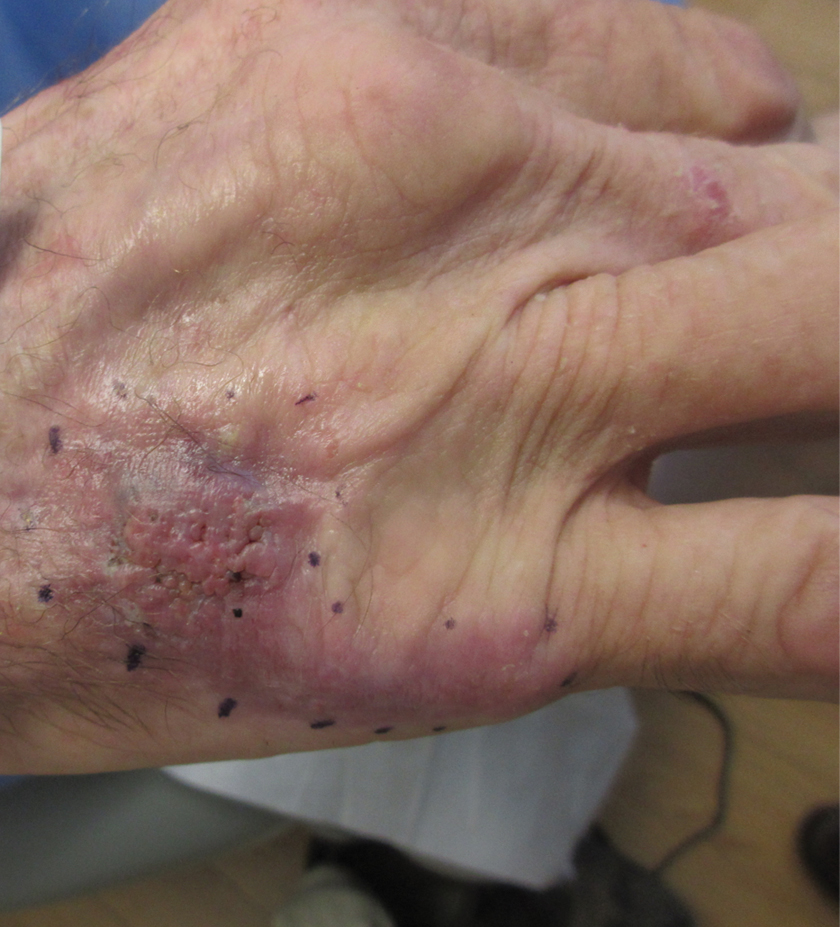
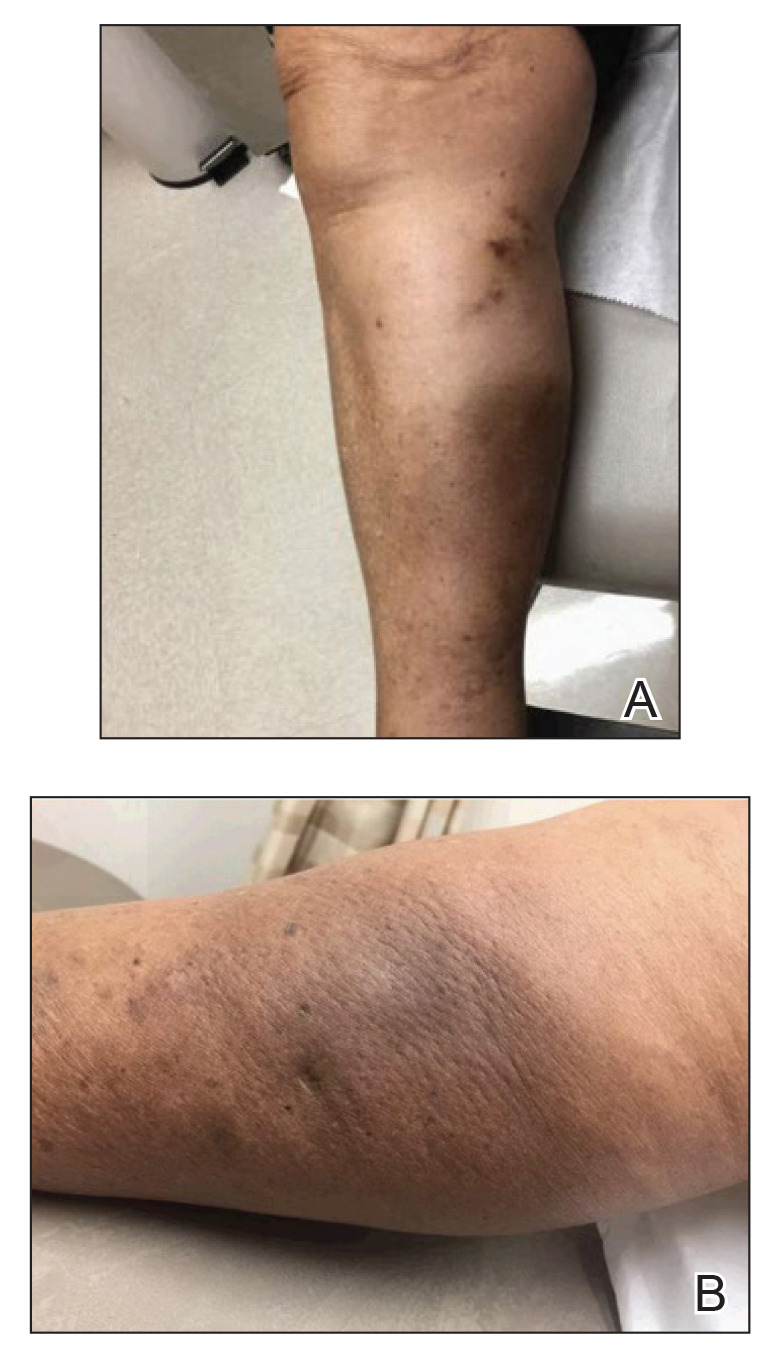
An 84-year-old woman presented with a tender plaque on the right lower leg of 2 months’ duration. Her medical history was remarkable for Kaposi sarcoma, with multiple sites on the body treated with megavoltage external beam radiotherapy during the prior 4 years. The most recent treatment occurred 8 months prior to presentation, at which time she had undergone radiotherapy for lesions on the posterior lower right leg. Physical examination demonstrated a hyperpigmented and indurated plaque at the treatment site (Figure 1). Skin biopsy results showed a mildly sclerotic dermis with atypical radiation fibroblasts scattered interstitially between collagen bundles, and a lobular panniculitis with degenerated adipocytes and foamy histiocytes (Figure 2). Hyalinized dermal vessels also were present. Based on the constellation of these biopsy findings, a diagnosis of PIPP was made.
The diagnosis of PIPP is challenging and invariably requires histologic examination. Clinically, the differential diagnosis includes cutaneous metastasis of the primary neoplasm, cellulitis, lipodermatosclerosis, morphea, and chronic radiation dermatitis.
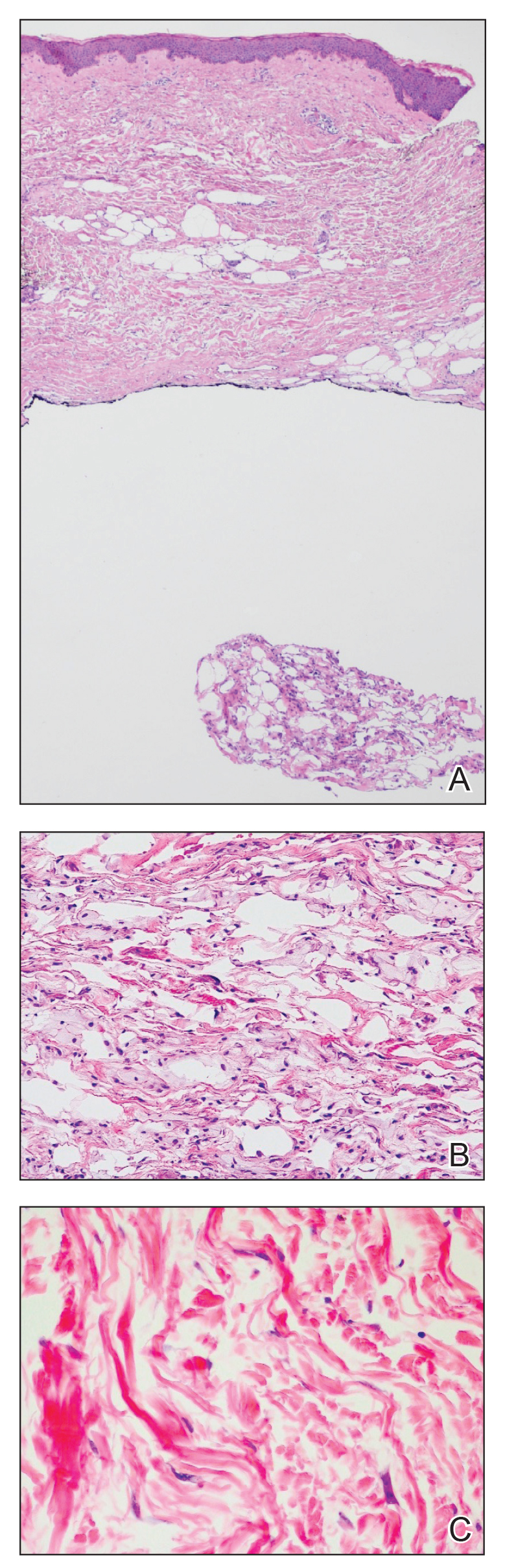
Histologically, PIPP is characterized by a lobular panniculitis without vasculitis. Typical findings include the presence of centrilobular necrotic adipocytes along with a foamy histiocytic infiltrate containing lipophagic granulomas at the periphery of the fat lobules. Septal thickening and sclerosis around fat lobules also have been described, and dermal changes associated with chronic radiation dermatitis, such as papillary dermal sclerosis, endothelial swelling, vascular hyaline arteriosclerosis, and atypical star-shaped radiation fibroblasts, may be present.2 Features of radiation-induced vasculopathy commonly are seen, although the appearance of these features varies over time. Intimal injury and mural thrombosis can develop within 5 years of radiation therapy, fibrosis of the vessel wall can occur within 10 years of radiation therapy, and atherosclerosis and periarterial fibrosis can appear within 20 years of radiation therapy.2,3 The histologic findings in our patient showed characteristic dermal findings seen in radiation dermatitis in addition to a lobular panniculitis with foamy histiocytes and mild vessel damage.
In contrast, lipodermatosclerosis is a septal and lobular panniculitis with septal fibrosis. Membranocystic fat necrosis is present, characterized by fat microcysts lined by feathery eosinophilic material. Stasis changes in the dermis and epidermis are accompanied by a mild perivascular lymphocytic infiltrate.
Patients with traumatic panniculitis, which also may enter the clinical differential diagnosis of PIPP, often demonstrate nonspecific histologic changes. Early lesions show a perivascular infiltrate of lymphocytes and macrophages. Evolving lesions show variably sized fat microcysts surrounded by histiocytes, in addition to possible calcifications and a foreign-body giant cell reaction. A fibrous capsule may develop, surrounding the fat necrosis to form a mobile encapsulated lipoma. Late lesions frequently demonstrate lipomembranous changes and calcium deposits.4
To date, nearly all cases of PIPP in the literature have been described in breast cancer patients.1,2,5,6 However, Sandoval et al7 reported a case of PIPP occurring in the leg of a patient after radiotherapy for a soft tissue sarcoma. Similar to our patient, this patient presented with a painful, dully erythematous, indurated plaque, although her symptoms arose 5 years after radiotherapy.
Megavoltage external beam radiotherapy has become a widely used modality in the treatment of various cancers. As such, PIPP may represent an underdiagnosed condition with potential cases remaining unidentified when the clinical differential diagnosis does not lead to biopsy. Effective therapies have yet to be widely reported, and our patient failed to experience notable improvement with either topical or intralesional corticosteroids. Further studies are needed in order to address this knowledge gap.
- Winkelmann RK, Grado GL, Quimby SR, et al. Pseudosclerodermatous panniculitis after irradiation: an unusual complication of megavoltage treatment of breast carcinoma. Mayo Clin Proc. 1993;68:122-127.
- Pielasinski U, Machan S, Camacho D, et al. Postirradiation pseudosclerodermatous panniculitis: three new cases with additional histopathologic features supporting the radiotherapy etiology. Am J Dermatopathol. 2013;35:129-134.
- Butler MJ, Lane RH, Webster JH. Irradiation injury to large arteries. Br J Surg. 1980;67:341-343. Moreno A, Marcoval J, Peyri J. Traumatic panniculitis. Dermatol Clin. 2008;26:481-483.
- Shirsat HS, Walsh NM, McDonald LJ, et al. Postirradiation pseudosclerodermatous panniculitis with involvement of breast parenchyma: a dramatic example of a rare entity and a pitfall in diagnosis. J Cutan Pathol. 2016;43:444-450.
- Carrasco L, Moreno C, Pastor MA, et al. Postirradiation pseudosclerodermatous panniculitis. Am J Dermatopathol. 2001;23:283-287.
- Sandoval M, Giesen L, Cataldo K, et al. Postirradiation pseudosclerodermatous panniculitis of the leg: report of a case and review of the literature. Am J Dermatopathol. 2015;37:587-589.
To the Editor:
Postirradiation pseudosclerodermatous panniculitis (PIPP) is a rarely reported complication of megavoltage external beam radiotherapy that was first identified in 1993 by Winkelmann et al.1 The condition presents as an erythematous or hyperpigmented indurated plaque at a site of prior radiotherapy. Lesions caused by PIPP most commonly arise several months after treatment, although they may emerge up to 17 years following exposure.2 Herein, we report a rare case of a patient with PIPP occurring on the leg who previously had been treated for Kaposi sarcoma.
An 84-year-old woman presented with a tender plaque on the right lower leg of 2 months’ duration. Her medical history was remarkable for Kaposi sarcoma, with multiple sites on the body treated with megavoltage external beam radiotherapy during the prior 4 years. The most recent treatment occurred 8 months prior to presentation, at which time she had undergone radiotherapy for lesions on the posterior lower right leg. Physical examination demonstrated a hyperpigmented and indurated plaque at the treatment site (Figure 1). Skin biopsy results showed a mildly sclerotic dermis with atypical radiation fibroblasts scattered interstitially between collagen bundles, and a lobular panniculitis with degenerated adipocytes and foamy histiocytes (Figure 2). Hyalinized dermal vessels also were present. Based on the constellation of these biopsy findings, a diagnosis of PIPP was made.

The diagnosis of PIPP is challenging and invariably requires histologic examination. Clinically, the differential diagnosis includes cutaneous metastasis of the primary neoplasm, cellulitis, lipodermatosclerosis, morphea, and chronic radiation dermatitis.

Histologically, PIPP is characterized by a lobular panniculitis without vasculitis. Typical findings include the presence of centrilobular necrotic adipocytes along with a foamy histiocytic infiltrate containing lipophagic granulomas at the periphery of the fat lobules. Septal thickening and sclerosis around fat lobules also have been described, and dermal changes associated with chronic radiation dermatitis, such as papillary dermal sclerosis, endothelial swelling, vascular hyaline arteriosclerosis, and atypical star-shaped radiation fibroblasts, may be present.2 Features of radiation-induced vasculopathy commonly are seen, although the appearance of these features varies over time. Intimal injury and mural thrombosis can develop within 5 years of radiation therapy, fibrosis of the vessel wall can occur within 10 years of radiation therapy, and atherosclerosis and periarterial fibrosis can appear within 20 years of radiation therapy.2,3 The histologic findings in our patient showed characteristic dermal findings seen in radiation dermatitis in addition to a lobular panniculitis with foamy histiocytes and mild vessel damage.
In contrast, lipodermatosclerosis is a septal and lobular panniculitis with septal fibrosis. Membranocystic fat necrosis is present, characterized by fat microcysts lined by feathery eosinophilic material. Stasis changes in the dermis and epidermis are accompanied by a mild perivascular lymphocytic infiltrate.
Patients with traumatic panniculitis, which also may enter the clinical differential diagnosis of PIPP, often demonstrate nonspecific histologic changes. Early lesions show a perivascular infiltrate of lymphocytes and macrophages. Evolving lesions show variably sized fat microcysts surrounded by histiocytes, in addition to possible calcifications and a foreign-body giant cell reaction. A fibrous capsule may develop, surrounding the fat necrosis to form a mobile encapsulated lipoma. Late lesions frequently demonstrate lipomembranous changes and calcium deposits.4
To date, nearly all cases of PIPP in the literature have been described in breast cancer patients.1,2,5,6 However, Sandoval et al7 reported a case of PIPP occurring in the leg of a patient after radiotherapy for a soft tissue sarcoma. Similar to our patient, this patient presented with a painful, dully erythematous, indurated plaque, although her symptoms arose 5 years after radiotherapy.
Megavoltage external beam radiotherapy has become a widely used modality in the treatment of various cancers. As such, PIPP may represent an underdiagnosed condition with potential cases remaining unidentified when the clinical differential diagnosis does not lead to biopsy. Effective therapies have yet to be widely reported, and our patient failed to experience notable improvement with either topical or intralesional corticosteroids. Further studies are needed in order to address this knowledge gap.
To the Editor:
Postirradiation pseudosclerodermatous panniculitis (PIPP) is a rarely reported complication of megavoltage external beam radiotherapy that was first identified in 1993 by Winkelmann et al.1 The condition presents as an erythematous or hyperpigmented indurated plaque at a site of prior radiotherapy. Lesions caused by PIPP most commonly arise several months after treatment, although they may emerge up to 17 years following exposure.2 Herein, we report a rare case of a patient with PIPP occurring on the leg who previously had been treated for Kaposi sarcoma.
An 84-year-old woman presented with a tender plaque on the right lower leg of 2 months’ duration. Her medical history was remarkable for Kaposi sarcoma, with multiple sites on the body treated with megavoltage external beam radiotherapy during the prior 4 years. The most recent treatment occurred 8 months prior to presentation, at which time she had undergone radiotherapy for lesions on the posterior lower right leg. Physical examination demonstrated a hyperpigmented and indurated plaque at the treatment site (Figure 1). Skin biopsy results showed a mildly sclerotic dermis with atypical radiation fibroblasts scattered interstitially between collagen bundles, and a lobular panniculitis with degenerated adipocytes and foamy histiocytes (Figure 2). Hyalinized dermal vessels also were present. Based on the constellation of these biopsy findings, a diagnosis of PIPP was made.

The diagnosis of PIPP is challenging and invariably requires histologic examination. Clinically, the differential diagnosis includes cutaneous metastasis of the primary neoplasm, cellulitis, lipodermatosclerosis, morphea, and chronic radiation dermatitis.

Histologically, PIPP is characterized by a lobular panniculitis without vasculitis. Typical findings include the presence of centrilobular necrotic adipocytes along with a foamy histiocytic infiltrate containing lipophagic granulomas at the periphery of the fat lobules. Septal thickening and sclerosis around fat lobules also have been described, and dermal changes associated with chronic radiation dermatitis, such as papillary dermal sclerosis, endothelial swelling, vascular hyaline arteriosclerosis, and atypical star-shaped radiation fibroblasts, may be present.2 Features of radiation-induced vasculopathy commonly are seen, although the appearance of these features varies over time. Intimal injury and mural thrombosis can develop within 5 years of radiation therapy, fibrosis of the vessel wall can occur within 10 years of radiation therapy, and atherosclerosis and periarterial fibrosis can appear within 20 years of radiation therapy.2,3 The histologic findings in our patient showed characteristic dermal findings seen in radiation dermatitis in addition to a lobular panniculitis with foamy histiocytes and mild vessel damage.
In contrast, lipodermatosclerosis is a septal and lobular panniculitis with septal fibrosis. Membranocystic fat necrosis is present, characterized by fat microcysts lined by feathery eosinophilic material. Stasis changes in the dermis and epidermis are accompanied by a mild perivascular lymphocytic infiltrate.
Patients with traumatic panniculitis, which also may enter the clinical differential diagnosis of PIPP, often demonstrate nonspecific histologic changes. Early lesions show a perivascular infiltrate of lymphocytes and macrophages. Evolving lesions show variably sized fat microcysts surrounded by histiocytes, in addition to possible calcifications and a foreign-body giant cell reaction. A fibrous capsule may develop, surrounding the fat necrosis to form a mobile encapsulated lipoma. Late lesions frequently demonstrate lipomembranous changes and calcium deposits.4
To date, nearly all cases of PIPP in the literature have been described in breast cancer patients.1,2,5,6 However, Sandoval et al7 reported a case of PIPP occurring in the leg of a patient after radiotherapy for a soft tissue sarcoma. Similar to our patient, this patient presented with a painful, dully erythematous, indurated plaque, although her symptoms arose 5 years after radiotherapy.
Megavoltage external beam radiotherapy has become a widely used modality in the treatment of various cancers. As such, PIPP may represent an underdiagnosed condition with potential cases remaining unidentified when the clinical differential diagnosis does not lead to biopsy. Effective therapies have yet to be widely reported, and our patient failed to experience notable improvement with either topical or intralesional corticosteroids. Further studies are needed in order to address this knowledge gap.
- Winkelmann RK, Grado GL, Quimby SR, et al. Pseudosclerodermatous panniculitis after irradiation: an unusual complication of megavoltage treatment of breast carcinoma. Mayo Clin Proc. 1993;68:122-127.
- Pielasinski U, Machan S, Camacho D, et al. Postirradiation pseudosclerodermatous panniculitis: three new cases with additional histopathologic features supporting the radiotherapy etiology. Am J Dermatopathol. 2013;35:129-134.
- Butler MJ, Lane RH, Webster JH. Irradiation injury to large arteries. Br J Surg. 1980;67:341-343. Moreno A, Marcoval J, Peyri J. Traumatic panniculitis. Dermatol Clin. 2008;26:481-483.
- Shirsat HS, Walsh NM, McDonald LJ, et al. Postirradiation pseudosclerodermatous panniculitis with involvement of breast parenchyma: a dramatic example of a rare entity and a pitfall in diagnosis. J Cutan Pathol. 2016;43:444-450.
- Carrasco L, Moreno C, Pastor MA, et al. Postirradiation pseudosclerodermatous panniculitis. Am J Dermatopathol. 2001;23:283-287.
- Sandoval M, Giesen L, Cataldo K, et al. Postirradiation pseudosclerodermatous panniculitis of the leg: report of a case and review of the literature. Am J Dermatopathol. 2015;37:587-589.
- Winkelmann RK, Grado GL, Quimby SR, et al. Pseudosclerodermatous panniculitis after irradiation: an unusual complication of megavoltage treatment of breast carcinoma. Mayo Clin Proc. 1993;68:122-127.
- Pielasinski U, Machan S, Camacho D, et al. Postirradiation pseudosclerodermatous panniculitis: three new cases with additional histopathologic features supporting the radiotherapy etiology. Am J Dermatopathol. 2013;35:129-134.
- Butler MJ, Lane RH, Webster JH. Irradiation injury to large arteries. Br J Surg. 1980;67:341-343. Moreno A, Marcoval J, Peyri J. Traumatic panniculitis. Dermatol Clin. 2008;26:481-483.
- Shirsat HS, Walsh NM, McDonald LJ, et al. Postirradiation pseudosclerodermatous panniculitis with involvement of breast parenchyma: a dramatic example of a rare entity and a pitfall in diagnosis. J Cutan Pathol. 2016;43:444-450.
- Carrasco L, Moreno C, Pastor MA, et al. Postirradiation pseudosclerodermatous panniculitis. Am J Dermatopathol. 2001;23:283-287.
- Sandoval M, Giesen L, Cataldo K, et al. Postirradiation pseudosclerodermatous panniculitis of the leg: report of a case and review of the literature. Am J Dermatopathol. 2015;37:587-589.
Practice Points
- Postirradiation pseudosclerodermatous panniculitis presents as an erythematous or indurated plaque at a site of prior radiotherapy.
- This rare entity may be underreported and requires biopsy for accurate diagnosis.
Topical treatment for EB recommended for approval in the EU
A topical (EMA’s) Committee for Medicinal Products for Human Use.
“The benefit of Filsuvez is its ability to promote healing of EB partial thickness wounds,” the EMA said in an announcement on April 22. “It is thought to work by modulating inflammatory mediators and stimulating keratinocyte differentiation and migration, thereby promoting wound health and closure,” the statement adds.
The recommended indication for the product – developed by Amryt Pharmaceuticals DAC and currently designated as an orphan drug – is for the treatment of partial-thickness wounds associated with dystrophic and junctional EB in patients aged 6 months and older. The recommendation for approval came after the EMA sought and received external advice from independent physicians treating EB and from patients with the rare disease.
The most common side effects, according to the EMA announcement, are wound complications, application site reactions, wound infections, pruritus, and hypersensitivity reactions.
In February 2022, the Food and Drug Administration declined to approve the company’s new drug application as it was presented and asked the company to submit additional evidence of effectiveness for Oleogel-S10 in EB, the company announced at that time. The statement noted that the company was committed to working with the FDA to identify "the most expeditious pathway towards a potential approval.”
The company’s pivotal phase 3 trial enrolled 223 patients with EB, including 156 pediatric patients. The patients variously had three types of EB. The trial has two components: A 3-month, double-blind, randomized controlled phase, which has been completed, and an ongoing 24-month open-label, single-arm phase. The trial is being performed at 58 sites in 28 countries.
Results from the randomized controlled phase, reported in 2020, include a statistically significant increase in the proportion of patients achieving complete closure of an EB target wound within 45 days: 41.3% in the Oleogel-S10 group and 28.9% in the control group (P = .013). (Target wounds measured 10 cm² to 50 cm² and were present for at least 21 days but less than 9 months.) The safety profile of the treatment gel was acceptable and was well tolerated, compared with the control gel, according to Amryt’s press release. The results were presented at the European Academy of Dermatology and Venereology Congress in October 2020.
Data from a 12-month interim analysis of the follow-up phase were presented at the annual meeting of the American Academy of Dermatology in March 2022. Results showed further reductions in total body surface area percentage wounding to 5.4% among (from 7.4% at the end of the double-blind period and 12.1% at the beginning of the study) among the patients who continued treatment and who underwent assessment, according to a company press release. Treatment was well tolerated, and no new safety signals were identified, the release said.
A decision by the European Commission is expected within the next 2 months.
A version of this article first appeared on Medscape.com.
A topical (EMA’s) Committee for Medicinal Products for Human Use.
“The benefit of Filsuvez is its ability to promote healing of EB partial thickness wounds,” the EMA said in an announcement on April 22. “It is thought to work by modulating inflammatory mediators and stimulating keratinocyte differentiation and migration, thereby promoting wound health and closure,” the statement adds.
The recommended indication for the product – developed by Amryt Pharmaceuticals DAC and currently designated as an orphan drug – is for the treatment of partial-thickness wounds associated with dystrophic and junctional EB in patients aged 6 months and older. The recommendation for approval came after the EMA sought and received external advice from independent physicians treating EB and from patients with the rare disease.
The most common side effects, according to the EMA announcement, are wound complications, application site reactions, wound infections, pruritus, and hypersensitivity reactions.
In February 2022, the Food and Drug Administration declined to approve the company’s new drug application as it was presented and asked the company to submit additional evidence of effectiveness for Oleogel-S10 in EB, the company announced at that time. The statement noted that the company was committed to working with the FDA to identify "the most expeditious pathway towards a potential approval.”
The company’s pivotal phase 3 trial enrolled 223 patients with EB, including 156 pediatric patients. The patients variously had three types of EB. The trial has two components: A 3-month, double-blind, randomized controlled phase, which has been completed, and an ongoing 24-month open-label, single-arm phase. The trial is being performed at 58 sites in 28 countries.
Results from the randomized controlled phase, reported in 2020, include a statistically significant increase in the proportion of patients achieving complete closure of an EB target wound within 45 days: 41.3% in the Oleogel-S10 group and 28.9% in the control group (P = .013). (Target wounds measured 10 cm² to 50 cm² and were present for at least 21 days but less than 9 months.) The safety profile of the treatment gel was acceptable and was well tolerated, compared with the control gel, according to Amryt’s press release. The results were presented at the European Academy of Dermatology and Venereology Congress in October 2020.
Data from a 12-month interim analysis of the follow-up phase were presented at the annual meeting of the American Academy of Dermatology in March 2022. Results showed further reductions in total body surface area percentage wounding to 5.4% among (from 7.4% at the end of the double-blind period and 12.1% at the beginning of the study) among the patients who continued treatment and who underwent assessment, according to a company press release. Treatment was well tolerated, and no new safety signals were identified, the release said.
A decision by the European Commission is expected within the next 2 months.
A version of this article first appeared on Medscape.com.
A topical (EMA’s) Committee for Medicinal Products for Human Use.
“The benefit of Filsuvez is its ability to promote healing of EB partial thickness wounds,” the EMA said in an announcement on April 22. “It is thought to work by modulating inflammatory mediators and stimulating keratinocyte differentiation and migration, thereby promoting wound health and closure,” the statement adds.
The recommended indication for the product – developed by Amryt Pharmaceuticals DAC and currently designated as an orphan drug – is for the treatment of partial-thickness wounds associated with dystrophic and junctional EB in patients aged 6 months and older. The recommendation for approval came after the EMA sought and received external advice from independent physicians treating EB and from patients with the rare disease.
The most common side effects, according to the EMA announcement, are wound complications, application site reactions, wound infections, pruritus, and hypersensitivity reactions.
In February 2022, the Food and Drug Administration declined to approve the company’s new drug application as it was presented and asked the company to submit additional evidence of effectiveness for Oleogel-S10 in EB, the company announced at that time. The statement noted that the company was committed to working with the FDA to identify "the most expeditious pathway towards a potential approval.”
The company’s pivotal phase 3 trial enrolled 223 patients with EB, including 156 pediatric patients. The patients variously had three types of EB. The trial has two components: A 3-month, double-blind, randomized controlled phase, which has been completed, and an ongoing 24-month open-label, single-arm phase. The trial is being performed at 58 sites in 28 countries.
Results from the randomized controlled phase, reported in 2020, include a statistically significant increase in the proportion of patients achieving complete closure of an EB target wound within 45 days: 41.3% in the Oleogel-S10 group and 28.9% in the control group (P = .013). (Target wounds measured 10 cm² to 50 cm² and were present for at least 21 days but less than 9 months.) The safety profile of the treatment gel was acceptable and was well tolerated, compared with the control gel, according to Amryt’s press release. The results were presented at the European Academy of Dermatology and Venereology Congress in October 2020.
Data from a 12-month interim analysis of the follow-up phase were presented at the annual meeting of the American Academy of Dermatology in March 2022. Results showed further reductions in total body surface area percentage wounding to 5.4% among (from 7.4% at the end of the double-blind period and 12.1% at the beginning of the study) among the patients who continued treatment and who underwent assessment, according to a company press release. Treatment was well tolerated, and no new safety signals were identified, the release said.
A decision by the European Commission is expected within the next 2 months.
A version of this article first appeared on Medscape.com.
IV gentamicin improves junctional epidermolysis bullosa in children
Intravenous (JEB) caused by nonsense variants.
The newly generated structural protein persisted during the 3-month randomized clinical trial and was associated with significant wound closure – with no signs of ototoxic effects, nephrotoxic effects, or anti–laminin 332 autoantibody induction, investigators recently reported in JAMA Dermatology.
JEB is a rare, autosomal recessive disorder caused mainly by nonsense variants (i.e., mutations) in the LAMA3, LAMB3, or LAMC2 genes that encode laminin, resulting in widespread blisters and erosions of the skin. Current treatment is limited to supportive management and palliative care, and children with its severe subtype are likely to die within the first year of life.
“With data indicating a robust response to short-term gentamicin treatment and the marked stability of laminin 332, we envision that gentamicin could be delivered as a short-term pulse therapy every 2-3 months for patients with JEB caused by nonsense variants,” the researchers wrote.
Of the five patients, ages 3 months to 10 years, three received 7.5 mg/kg IV gentamicin daily for 14 days, and two received 10 mg/kg daily for 24 days at the University of Southern California, Los Angeles.
All had confirmed nonsense variants in LAMA3 or LAMB3 in one or two alleles, and all had minimal laminin 332 expression at baseline as determined by immunofluorescence. After treatment, each of the children had increased, sustained expression of laminin 332.
The researchers monitored three open wounds in each patient. By 1 month, seven of nine wounds in those receiving the lower-dose therapy and all of the wounds in those receiving the higher-dose therapy showed at least 50% closure. By 3 months, eight of nine wounds in the lower-dose group, and all wounds in the higher-dose group showed greater than 85% closure.
In an interview, senior investigators Mei Chen, PhD, professor of dermatology, and David T. Woodley, MD, professor and chair of dermatology, both at USC, emphasized laminin’s long half-life.“Once these skin structural proteins are generated at the dermal-epidermal junction, they are long-lasting structures, which means the therapy can be pulsed rather than continuously delivered, which can obviate some of the known side effects of the medication,” Dr. Woodley said.
Gentamicin, an aminoglycoside, works as a “read-through therapy,” inducing ribosomal read-through of premature termination codons (PTCs) caused by nonsense mutations. The read-through allows translation to proceed and full-length proteins to be generated.
Gentamicin read-through therapy is also being investigated for recessive dystrophic epidermolysis bullosa (RDEB) attributable to nonsense mutations. The culprit mutations in this form of EB occur in a gene that encodes collagen type VII alpha 1, which, like laminin, is responsible for dermal-epidermal adherence. A clinical trial of intravenous gentamicin for RDEB is ongoing at USC, Dr. Chen said.
EBS-MD case report
It may also have a role in treating epidermolysis bullosa simplex with muscular dystrophy (EBS-MD), according to investigators in Madrid. Their case report, published in JAMA Dermatology, details how two 14-day courses of infused gentamicin therapy were followed by re-expression of plectin in the skin for 4-5 months and mild improvement in symptoms in one patient, a woman in her 30s, with a homozygous nonsense variant in PLEC1.
In an editorial accompanying the two reports, Anna L. Bruckner, MD, MSCS, professor of dermatology, University of Colorado at Denver, Aurora, and colleagues expressed cautious optimism and said that additional research on the feasibility, possible cumulative toxic effects, risk of microbial resistance, and overall clinical relevance is needed.
Still, the “investigators should be applauded for taking advantage of a readily available systemic treatment to target cutaneous and extracutaneous symptoms of patients who have very limited treatment options at this time,” they wrote. While all forms of EB are considered orphan disorders, JEB and EBS-MD have received less research attention than RDEB.
The JEB study evaluated patients with clinical assessments/quality of life surveys and with a validated clinical score that considers skin and mucosae – the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI). There were small positive changes in EBDASI scores, but data were incomplete and therefore difficult to interpret.
A “noteworthy” finding, the authors wrote, were improvements in emotions and functioning in two of the children who were eligible given their older ages for assessment with the Skindex-16 quality-of-life survey. The improvements suggest “potential psychosocial benefits” of the gentamicin therapy.
The JEB study was supported in part by grants from the EB Research Partnership and EB Medical Research Foundation and an award from the Congressionally Directed Medical Research Program. In addition to the grants, Dr. Woodley and Dr. Chen reported receiving personal fees from Phoenix Tissue Repair outside of the submitted work. For the EBS-MD case report, the authors reported no disclosures. Dr. Bruckner, corresponding author of the editorial, reported grants from several companies outside the submitted work.
Intravenous (JEB) caused by nonsense variants.
The newly generated structural protein persisted during the 3-month randomized clinical trial and was associated with significant wound closure – with no signs of ototoxic effects, nephrotoxic effects, or anti–laminin 332 autoantibody induction, investigators recently reported in JAMA Dermatology.
JEB is a rare, autosomal recessive disorder caused mainly by nonsense variants (i.e., mutations) in the LAMA3, LAMB3, or LAMC2 genes that encode laminin, resulting in widespread blisters and erosions of the skin. Current treatment is limited to supportive management and palliative care, and children with its severe subtype are likely to die within the first year of life.
“With data indicating a robust response to short-term gentamicin treatment and the marked stability of laminin 332, we envision that gentamicin could be delivered as a short-term pulse therapy every 2-3 months for patients with JEB caused by nonsense variants,” the researchers wrote.
Of the five patients, ages 3 months to 10 years, three received 7.5 mg/kg IV gentamicin daily for 14 days, and two received 10 mg/kg daily for 24 days at the University of Southern California, Los Angeles.
All had confirmed nonsense variants in LAMA3 or LAMB3 in one or two alleles, and all had minimal laminin 332 expression at baseline as determined by immunofluorescence. After treatment, each of the children had increased, sustained expression of laminin 332.
The researchers monitored three open wounds in each patient. By 1 month, seven of nine wounds in those receiving the lower-dose therapy and all of the wounds in those receiving the higher-dose therapy showed at least 50% closure. By 3 months, eight of nine wounds in the lower-dose group, and all wounds in the higher-dose group showed greater than 85% closure.
In an interview, senior investigators Mei Chen, PhD, professor of dermatology, and David T. Woodley, MD, professor and chair of dermatology, both at USC, emphasized laminin’s long half-life.“Once these skin structural proteins are generated at the dermal-epidermal junction, they are long-lasting structures, which means the therapy can be pulsed rather than continuously delivered, which can obviate some of the known side effects of the medication,” Dr. Woodley said.
Gentamicin, an aminoglycoside, works as a “read-through therapy,” inducing ribosomal read-through of premature termination codons (PTCs) caused by nonsense mutations. The read-through allows translation to proceed and full-length proteins to be generated.
Gentamicin read-through therapy is also being investigated for recessive dystrophic epidermolysis bullosa (RDEB) attributable to nonsense mutations. The culprit mutations in this form of EB occur in a gene that encodes collagen type VII alpha 1, which, like laminin, is responsible for dermal-epidermal adherence. A clinical trial of intravenous gentamicin for RDEB is ongoing at USC, Dr. Chen said.
EBS-MD case report
It may also have a role in treating epidermolysis bullosa simplex with muscular dystrophy (EBS-MD), according to investigators in Madrid. Their case report, published in JAMA Dermatology, details how two 14-day courses of infused gentamicin therapy were followed by re-expression of plectin in the skin for 4-5 months and mild improvement in symptoms in one patient, a woman in her 30s, with a homozygous nonsense variant in PLEC1.
In an editorial accompanying the two reports, Anna L. Bruckner, MD, MSCS, professor of dermatology, University of Colorado at Denver, Aurora, and colleagues expressed cautious optimism and said that additional research on the feasibility, possible cumulative toxic effects, risk of microbial resistance, and overall clinical relevance is needed.
Still, the “investigators should be applauded for taking advantage of a readily available systemic treatment to target cutaneous and extracutaneous symptoms of patients who have very limited treatment options at this time,” they wrote. While all forms of EB are considered orphan disorders, JEB and EBS-MD have received less research attention than RDEB.
The JEB study evaluated patients with clinical assessments/quality of life surveys and with a validated clinical score that considers skin and mucosae – the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI). There were small positive changes in EBDASI scores, but data were incomplete and therefore difficult to interpret.
A “noteworthy” finding, the authors wrote, were improvements in emotions and functioning in two of the children who were eligible given their older ages for assessment with the Skindex-16 quality-of-life survey. The improvements suggest “potential psychosocial benefits” of the gentamicin therapy.
The JEB study was supported in part by grants from the EB Research Partnership and EB Medical Research Foundation and an award from the Congressionally Directed Medical Research Program. In addition to the grants, Dr. Woodley and Dr. Chen reported receiving personal fees from Phoenix Tissue Repair outside of the submitted work. For the EBS-MD case report, the authors reported no disclosures. Dr. Bruckner, corresponding author of the editorial, reported grants from several companies outside the submitted work.
Intravenous (JEB) caused by nonsense variants.
The newly generated structural protein persisted during the 3-month randomized clinical trial and was associated with significant wound closure – with no signs of ototoxic effects, nephrotoxic effects, or anti–laminin 332 autoantibody induction, investigators recently reported in JAMA Dermatology.
JEB is a rare, autosomal recessive disorder caused mainly by nonsense variants (i.e., mutations) in the LAMA3, LAMB3, or LAMC2 genes that encode laminin, resulting in widespread blisters and erosions of the skin. Current treatment is limited to supportive management and palliative care, and children with its severe subtype are likely to die within the first year of life.
“With data indicating a robust response to short-term gentamicin treatment and the marked stability of laminin 332, we envision that gentamicin could be delivered as a short-term pulse therapy every 2-3 months for patients with JEB caused by nonsense variants,” the researchers wrote.
Of the five patients, ages 3 months to 10 years, three received 7.5 mg/kg IV gentamicin daily for 14 days, and two received 10 mg/kg daily for 24 days at the University of Southern California, Los Angeles.
All had confirmed nonsense variants in LAMA3 or LAMB3 in one or two alleles, and all had minimal laminin 332 expression at baseline as determined by immunofluorescence. After treatment, each of the children had increased, sustained expression of laminin 332.
The researchers monitored three open wounds in each patient. By 1 month, seven of nine wounds in those receiving the lower-dose therapy and all of the wounds in those receiving the higher-dose therapy showed at least 50% closure. By 3 months, eight of nine wounds in the lower-dose group, and all wounds in the higher-dose group showed greater than 85% closure.
In an interview, senior investigators Mei Chen, PhD, professor of dermatology, and David T. Woodley, MD, professor and chair of dermatology, both at USC, emphasized laminin’s long half-life.“Once these skin structural proteins are generated at the dermal-epidermal junction, they are long-lasting structures, which means the therapy can be pulsed rather than continuously delivered, which can obviate some of the known side effects of the medication,” Dr. Woodley said.
Gentamicin, an aminoglycoside, works as a “read-through therapy,” inducing ribosomal read-through of premature termination codons (PTCs) caused by nonsense mutations. The read-through allows translation to proceed and full-length proteins to be generated.
Gentamicin read-through therapy is also being investigated for recessive dystrophic epidermolysis bullosa (RDEB) attributable to nonsense mutations. The culprit mutations in this form of EB occur in a gene that encodes collagen type VII alpha 1, which, like laminin, is responsible for dermal-epidermal adherence. A clinical trial of intravenous gentamicin for RDEB is ongoing at USC, Dr. Chen said.
EBS-MD case report
It may also have a role in treating epidermolysis bullosa simplex with muscular dystrophy (EBS-MD), according to investigators in Madrid. Their case report, published in JAMA Dermatology, details how two 14-day courses of infused gentamicin therapy were followed by re-expression of plectin in the skin for 4-5 months and mild improvement in symptoms in one patient, a woman in her 30s, with a homozygous nonsense variant in PLEC1.
In an editorial accompanying the two reports, Anna L. Bruckner, MD, MSCS, professor of dermatology, University of Colorado at Denver, Aurora, and colleagues expressed cautious optimism and said that additional research on the feasibility, possible cumulative toxic effects, risk of microbial resistance, and overall clinical relevance is needed.
Still, the “investigators should be applauded for taking advantage of a readily available systemic treatment to target cutaneous and extracutaneous symptoms of patients who have very limited treatment options at this time,” they wrote. While all forms of EB are considered orphan disorders, JEB and EBS-MD have received less research attention than RDEB.
The JEB study evaluated patients with clinical assessments/quality of life surveys and with a validated clinical score that considers skin and mucosae – the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI). There were small positive changes in EBDASI scores, but data were incomplete and therefore difficult to interpret.
A “noteworthy” finding, the authors wrote, were improvements in emotions and functioning in two of the children who were eligible given their older ages for assessment with the Skindex-16 quality-of-life survey. The improvements suggest “potential psychosocial benefits” of the gentamicin therapy.
The JEB study was supported in part by grants from the EB Research Partnership and EB Medical Research Foundation and an award from the Congressionally Directed Medical Research Program. In addition to the grants, Dr. Woodley and Dr. Chen reported receiving personal fees from Phoenix Tissue Repair outside of the submitted work. For the EBS-MD case report, the authors reported no disclosures. Dr. Bruckner, corresponding author of the editorial, reported grants from several companies outside the submitted work.
FROM JAMA DERMATOLOGY
Cellulitis care costly from misdiagnosis, needless hospitalizations
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Necrotic Ulcerations After the Use of an Over-the-counter Mole and Skin Tag Removal Product
To the Editor:
Several mole and skin tag removal products are available online and over the counter (OTC).1 Patients concerned with the cosmetic appearance of nevi may use these products as a do-it-yourself alternative to surgical removal. However, these products have the potential to cause harm.2 Beyond the cosmetic adverse effects of skin necrosis and scar formation, these products can mask premalignant and malignant skin lesions.2 Herein, we describe a patient with a family history of melanoma who developed facial and chest ulcerations with necrosis after applying an OTC mole and skin tag removal product.
A 45-year-old woman with fair skin presented to a clinic with multiple superficial ulcerations measuring approximately 1 cm in diameter with necrotic black bases and erythematous rims on the face, right side of the upper chest, and left earlobe after using the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set, an OTC mole and skin tag removal product. The patient reported using the product 24 hours prior for the cosmetic removal of multiple nevi. After applying the product, she observed that it “immediately melted [her] skin” and the areas where the product was applied “turned black.” She reported that the product was applied to the skin for no longer than 30 seconds, after which she developed the necrotic lesions (Figure). After removing the product, she applied an OTC ointment containing bacitracin, neomycin, and polymyxin B to the lesions.
The patient had no history of nonmelanoma skin cancers or atypical nevi. She had a family history of melanoma in her mother and maternal uncle. The treatment plan was aimed primarily at reducing scar formation. We advised frequent application of petroleum-based ointments for moisture and overlying silicone scar tape to protect the area from photodamage and promote wound healing. We further advocated for sun protection and the use of a physical sunscreen on the lesions as they healed. We discussed potential laser-based scar revision options in the future.
With more than 180 reviews on Amazon and almost 70% of these reviews made within the month prior to compiling this manuscript, the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set appeared to be popular; however, the product currently is unavailable on Amazon. Testimonials and before-and-after pictures advertising the product show an all-natural, safe, and effective method as an alternative to surgical removal of skin tags and nevi. The product website claims that skin tags and moles will “fall off naturally within 7 to 10 days” and the product can be used for “almost all skin types.” Users are instructed to apply the removal product and wipe it off when the skin surrounding the mole becomes swollen. The product kit also includes a repair lotion, which claims to help heal the skin after scab formation and scar development.
The ingredients listed on the product packaging are salicylic acid 25%, Melaleuca alternifolia (tea tree) leaf oil, propylene glycol, hydroxyethylcellulose, and alcohol. Salicylic acid 25% is a superficial peeling agent that penetrates the epidermis to the dermoepidermal junction. The potential side effects are mild and include superficial desquamation and epidermolysis.3 The Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set is not regulated by the US Food and Drug Administration and may contain variable concentrations of salicylic acid and other unknown compounds. Higher concentrations of salicylic acid can penetrate the full thickness of the epidermis into the papillary dermis, which can result in postinflammatory pigmentation, superficial infection, scarring, and deeper desquamation and epidermolysis.3 The product website advertises the use of only natural ingredients and an “advanced blend of concentrated natural ingredients contributing a broad spectrum of healing properties” in the formula. Although these claims are attractive to patients seeking alternatives to surgical approaches to nevi removal, the unfounded claims and unregulated ingredients may pose a threat to unsuspecting consumers.
Other OTC and “all-natural” mole removal products previously have been reported to cause harm.2Sanguinaria canadensis, also known as bloodroot, contains an alkaloid compound (sanguinarine) that has been shown to induce mitochondrial apoptosis and activation of Bcl-2 proteins in keratinocytes.4 Some products, such as Wart & Mole Vanish cream, may claim not to contain bloodroot specifically. However, sanguinarine can be extracted from other plants and may be listed as Argemone mexicana, Chelidonium majus, or Macleaya cordata in the ingredients list.5 The use of alternative medicine products such as black or yellow salve for the removal of suspected skin cancers also is not recommended because these escharotic treatments have not been proven safe or effective, and the manufacturing process for these compounds is unregulated.6,7 Self-treatment with alternative remedies for nevi or suspected skin cancers has been associated with progression of disease and even death due to metastatic spread.2
Self-removal of moles is concerning because the nevi are masked by necrotic lesions and can no longer be assessed by dermoscopy or histopathology. Furthermore, the compounds in the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set may have unknown effects on the transformation of premalignant cells. They also may mask an underlying process for which clinically proven and effective treatments such as cryotherapy, prescription topical agents, and surgical excision are warranted. Awareness of this product and similar products is important to educate patients on the harmful effects they may cause.
- Clayton R, Turner R. Cosmetic surgery: who needs surgeons when you’ve got creams? Br J Dermatol. 2007;156:1383-1384.
- McAllister JC, Petzold CR, Lio PA. Adverse effects of a mole removal cream. Pediatr Dermatol. 2009;26:628-629.
- Soleymani T, Lanoue J, Rahman Z. A practical approach to chemical peels: a review of fundamentals and step-by-step algorithmic protocol for treatment. J Clin Aesthet Dermatol. 2018;11:21-28.
- Adhami VM, Aziz MH, Mukhatar M, et al. Activation of prodeath Bcl-2 family proteins and mitochondrial apoptosis pathway by sanguinarine in immortalized human HaCaT keratinocytes. Clin Cancer Res. 2003;9:3176-3182.
- Santos AC, Adkilen P. The alkaloids of Argemone mexicana. J Am Chem Soc. 1932;54:2923-2924.
- Osswald SS, Elston DM, Farley MF, et al. Self-treatment of a basal cell carcinoma with “black and yellow salve.” J Am Acad Dermatol. 2005;53:509-511.
- McDaniel S, Goldman GD. Consequences of using escharotic agents as primary treatment for nonmelanoma skin cancer. Arch Dermatol. 2002;138:1593-1596.
To the Editor:
Several mole and skin tag removal products are available online and over the counter (OTC).1 Patients concerned with the cosmetic appearance of nevi may use these products as a do-it-yourself alternative to surgical removal. However, these products have the potential to cause harm.2 Beyond the cosmetic adverse effects of skin necrosis and scar formation, these products can mask premalignant and malignant skin lesions.2 Herein, we describe a patient with a family history of melanoma who developed facial and chest ulcerations with necrosis after applying an OTC mole and skin tag removal product.
A 45-year-old woman with fair skin presented to a clinic with multiple superficial ulcerations measuring approximately 1 cm in diameter with necrotic black bases and erythematous rims on the face, right side of the upper chest, and left earlobe after using the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set, an OTC mole and skin tag removal product. The patient reported using the product 24 hours prior for the cosmetic removal of multiple nevi. After applying the product, she observed that it “immediately melted [her] skin” and the areas where the product was applied “turned black.” She reported that the product was applied to the skin for no longer than 30 seconds, after which she developed the necrotic lesions (Figure). After removing the product, she applied an OTC ointment containing bacitracin, neomycin, and polymyxin B to the lesions.

The patient had no history of nonmelanoma skin cancers or atypical nevi. She had a family history of melanoma in her mother and maternal uncle. The treatment plan was aimed primarily at reducing scar formation. We advised frequent application of petroleum-based ointments for moisture and overlying silicone scar tape to protect the area from photodamage and promote wound healing. We further advocated for sun protection and the use of a physical sunscreen on the lesions as they healed. We discussed potential laser-based scar revision options in the future.
With more than 180 reviews on Amazon and almost 70% of these reviews made within the month prior to compiling this manuscript, the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set appeared to be popular; however, the product currently is unavailable on Amazon. Testimonials and before-and-after pictures advertising the product show an all-natural, safe, and effective method as an alternative to surgical removal of skin tags and nevi. The product website claims that skin tags and moles will “fall off naturally within 7 to 10 days” and the product can be used for “almost all skin types.” Users are instructed to apply the removal product and wipe it off when the skin surrounding the mole becomes swollen. The product kit also includes a repair lotion, which claims to help heal the skin after scab formation and scar development.
The ingredients listed on the product packaging are salicylic acid 25%, Melaleuca alternifolia (tea tree) leaf oil, propylene glycol, hydroxyethylcellulose, and alcohol. Salicylic acid 25% is a superficial peeling agent that penetrates the epidermis to the dermoepidermal junction. The potential side effects are mild and include superficial desquamation and epidermolysis.3 The Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set is not regulated by the US Food and Drug Administration and may contain variable concentrations of salicylic acid and other unknown compounds. Higher concentrations of salicylic acid can penetrate the full thickness of the epidermis into the papillary dermis, which can result in postinflammatory pigmentation, superficial infection, scarring, and deeper desquamation and epidermolysis.3 The product website advertises the use of only natural ingredients and an “advanced blend of concentrated natural ingredients contributing a broad spectrum of healing properties” in the formula. Although these claims are attractive to patients seeking alternatives to surgical approaches to nevi removal, the unfounded claims and unregulated ingredients may pose a threat to unsuspecting consumers.
Other OTC and “all-natural” mole removal products previously have been reported to cause harm.2Sanguinaria canadensis, also known as bloodroot, contains an alkaloid compound (sanguinarine) that has been shown to induce mitochondrial apoptosis and activation of Bcl-2 proteins in keratinocytes.4 Some products, such as Wart & Mole Vanish cream, may claim not to contain bloodroot specifically. However, sanguinarine can be extracted from other plants and may be listed as Argemone mexicana, Chelidonium majus, or Macleaya cordata in the ingredients list.5 The use of alternative medicine products such as black or yellow salve for the removal of suspected skin cancers also is not recommended because these escharotic treatments have not been proven safe or effective, and the manufacturing process for these compounds is unregulated.6,7 Self-treatment with alternative remedies for nevi or suspected skin cancers has been associated with progression of disease and even death due to metastatic spread.2
Self-removal of moles is concerning because the nevi are masked by necrotic lesions and can no longer be assessed by dermoscopy or histopathology. Furthermore, the compounds in the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set may have unknown effects on the transformation of premalignant cells. They also may mask an underlying process for which clinically proven and effective treatments such as cryotherapy, prescription topical agents, and surgical excision are warranted. Awareness of this product and similar products is important to educate patients on the harmful effects they may cause.
To the Editor:
Several mole and skin tag removal products are available online and over the counter (OTC).1 Patients concerned with the cosmetic appearance of nevi may use these products as a do-it-yourself alternative to surgical removal. However, these products have the potential to cause harm.2 Beyond the cosmetic adverse effects of skin necrosis and scar formation, these products can mask premalignant and malignant skin lesions.2 Herein, we describe a patient with a family history of melanoma who developed facial and chest ulcerations with necrosis after applying an OTC mole and skin tag removal product.
A 45-year-old woman with fair skin presented to a clinic with multiple superficial ulcerations measuring approximately 1 cm in diameter with necrotic black bases and erythematous rims on the face, right side of the upper chest, and left earlobe after using the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set, an OTC mole and skin tag removal product. The patient reported using the product 24 hours prior for the cosmetic removal of multiple nevi. After applying the product, she observed that it “immediately melted [her] skin” and the areas where the product was applied “turned black.” She reported that the product was applied to the skin for no longer than 30 seconds, after which she developed the necrotic lesions (Figure). After removing the product, she applied an OTC ointment containing bacitracin, neomycin, and polymyxin B to the lesions.

The patient had no history of nonmelanoma skin cancers or atypical nevi. She had a family history of melanoma in her mother and maternal uncle. The treatment plan was aimed primarily at reducing scar formation. We advised frequent application of petroleum-based ointments for moisture and overlying silicone scar tape to protect the area from photodamage and promote wound healing. We further advocated for sun protection and the use of a physical sunscreen on the lesions as they healed. We discussed potential laser-based scar revision options in the future.
With more than 180 reviews on Amazon and almost 70% of these reviews made within the month prior to compiling this manuscript, the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set appeared to be popular; however, the product currently is unavailable on Amazon. Testimonials and before-and-after pictures advertising the product show an all-natural, safe, and effective method as an alternative to surgical removal of skin tags and nevi. The product website claims that skin tags and moles will “fall off naturally within 7 to 10 days” and the product can be used for “almost all skin types.” Users are instructed to apply the removal product and wipe it off when the skin surrounding the mole becomes swollen. The product kit also includes a repair lotion, which claims to help heal the skin after scab formation and scar development.
The ingredients listed on the product packaging are salicylic acid 25%, Melaleuca alternifolia (tea tree) leaf oil, propylene glycol, hydroxyethylcellulose, and alcohol. Salicylic acid 25% is a superficial peeling agent that penetrates the epidermis to the dermoepidermal junction. The potential side effects are mild and include superficial desquamation and epidermolysis.3 The Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set is not regulated by the US Food and Drug Administration and may contain variable concentrations of salicylic acid and other unknown compounds. Higher concentrations of salicylic acid can penetrate the full thickness of the epidermis into the papillary dermis, which can result in postinflammatory pigmentation, superficial infection, scarring, and deeper desquamation and epidermolysis.3 The product website advertises the use of only natural ingredients and an “advanced blend of concentrated natural ingredients contributing a broad spectrum of healing properties” in the formula. Although these claims are attractive to patients seeking alternatives to surgical approaches to nevi removal, the unfounded claims and unregulated ingredients may pose a threat to unsuspecting consumers.
Other OTC and “all-natural” mole removal products previously have been reported to cause harm.2Sanguinaria canadensis, also known as bloodroot, contains an alkaloid compound (sanguinarine) that has been shown to induce mitochondrial apoptosis and activation of Bcl-2 proteins in keratinocytes.4 Some products, such as Wart & Mole Vanish cream, may claim not to contain bloodroot specifically. However, sanguinarine can be extracted from other plants and may be listed as Argemone mexicana, Chelidonium majus, or Macleaya cordata in the ingredients list.5 The use of alternative medicine products such as black or yellow salve for the removal of suspected skin cancers also is not recommended because these escharotic treatments have not been proven safe or effective, and the manufacturing process for these compounds is unregulated.6,7 Self-treatment with alternative remedies for nevi or suspected skin cancers has been associated with progression of disease and even death due to metastatic spread.2
Self-removal of moles is concerning because the nevi are masked by necrotic lesions and can no longer be assessed by dermoscopy or histopathology. Furthermore, the compounds in the Ariella Mole Corrector and Skin Tag Remover and Repair Lotion Set may have unknown effects on the transformation of premalignant cells. They also may mask an underlying process for which clinically proven and effective treatments such as cryotherapy, prescription topical agents, and surgical excision are warranted. Awareness of this product and similar products is important to educate patients on the harmful effects they may cause.
- Clayton R, Turner R. Cosmetic surgery: who needs surgeons when you’ve got creams? Br J Dermatol. 2007;156:1383-1384.
- McAllister JC, Petzold CR, Lio PA. Adverse effects of a mole removal cream. Pediatr Dermatol. 2009;26:628-629.
- Soleymani T, Lanoue J, Rahman Z. A practical approach to chemical peels: a review of fundamentals and step-by-step algorithmic protocol for treatment. J Clin Aesthet Dermatol. 2018;11:21-28.
- Adhami VM, Aziz MH, Mukhatar M, et al. Activation of prodeath Bcl-2 family proteins and mitochondrial apoptosis pathway by sanguinarine in immortalized human HaCaT keratinocytes. Clin Cancer Res. 2003;9:3176-3182.
- Santos AC, Adkilen P. The alkaloids of Argemone mexicana. J Am Chem Soc. 1932;54:2923-2924.
- Osswald SS, Elston DM, Farley MF, et al. Self-treatment of a basal cell carcinoma with “black and yellow salve.” J Am Acad Dermatol. 2005;53:509-511.
- McDaniel S, Goldman GD. Consequences of using escharotic agents as primary treatment for nonmelanoma skin cancer. Arch Dermatol. 2002;138:1593-1596.
- Clayton R, Turner R. Cosmetic surgery: who needs surgeons when you’ve got creams? Br J Dermatol. 2007;156:1383-1384.
- McAllister JC, Petzold CR, Lio PA. Adverse effects of a mole removal cream. Pediatr Dermatol. 2009;26:628-629.
- Soleymani T, Lanoue J, Rahman Z. A practical approach to chemical peels: a review of fundamentals and step-by-step algorithmic protocol for treatment. J Clin Aesthet Dermatol. 2018;11:21-28.
- Adhami VM, Aziz MH, Mukhatar M, et al. Activation of prodeath Bcl-2 family proteins and mitochondrial apoptosis pathway by sanguinarine in immortalized human HaCaT keratinocytes. Clin Cancer Res. 2003;9:3176-3182.
- Santos AC, Adkilen P. The alkaloids of Argemone mexicana. J Am Chem Soc. 1932;54:2923-2924.
- Osswald SS, Elston DM, Farley MF, et al. Self-treatment of a basal cell carcinoma with “black and yellow salve.” J Am Acad Dermatol. 2005;53:509-511.
- McDaniel S, Goldman GD. Consequences of using escharotic agents as primary treatment for nonmelanoma skin cancer. Arch Dermatol. 2002;138:1593-1596.
Practice Point
- Self-administered mole and skin tag removal products are rising in popularity, but unregulated ingredients in over-the-counter products that are not approved by the US Food and Drug Administration may mask underlying transformation of atypical nevi.
Pencil-core Granuloma Forming 62 Years After Initial Injury
To the Editor:
Trauma from a pencil tip can sometimes result in a fragment of lead being left embedded within the skin. Pencil lead is composed of 66% graphite carbon, 26% aluminum silicate, and 8% paraffin.1,2 While the toxicity of these individual elements is low, paraffin can cause nonallergic foreign-body reactions, aluminum silicate can induce epithelioid granulomatous reactions, and graphite has been reported to cause chronic granulomatous reactions in the lungs of those who work with graphite.2 Penetrating trauma with a pencil can result in the formation of a cutaneous granulomatous reaction that can sometimes occur years to decades after the initial injury.3,4 Several cases of pencil-core granulomas have been published, with lag times between the initial trauma and lesion growth as long as 58 years.1-10 The pencil-core granuloma may simulate malignant melanoma, as it presents clinically as a growing, darkly pigmented lesion, thus prompting biopsy. We present a case of a pencil-core granuloma that began to grow 62 years after the initial trauma.
A 72-year-old woman was referred to our clinic for evaluation of a dark nodule on the forehead. The lesion had been present since the age of 10 years, reportedly from an accidental stabbing with a pencil. The lesion had been flat, stable, and asymptomatic since the trauma occurred; however, the patient reported that approximately 9 months prior to presentation, it had started growing and became painful. Physical examination revealed a 1.0-cm, round, bluish-black nodule on the right superior forehead (Figure 1A). No satellite lesions or local lymphadenopathy were noted on general examination.
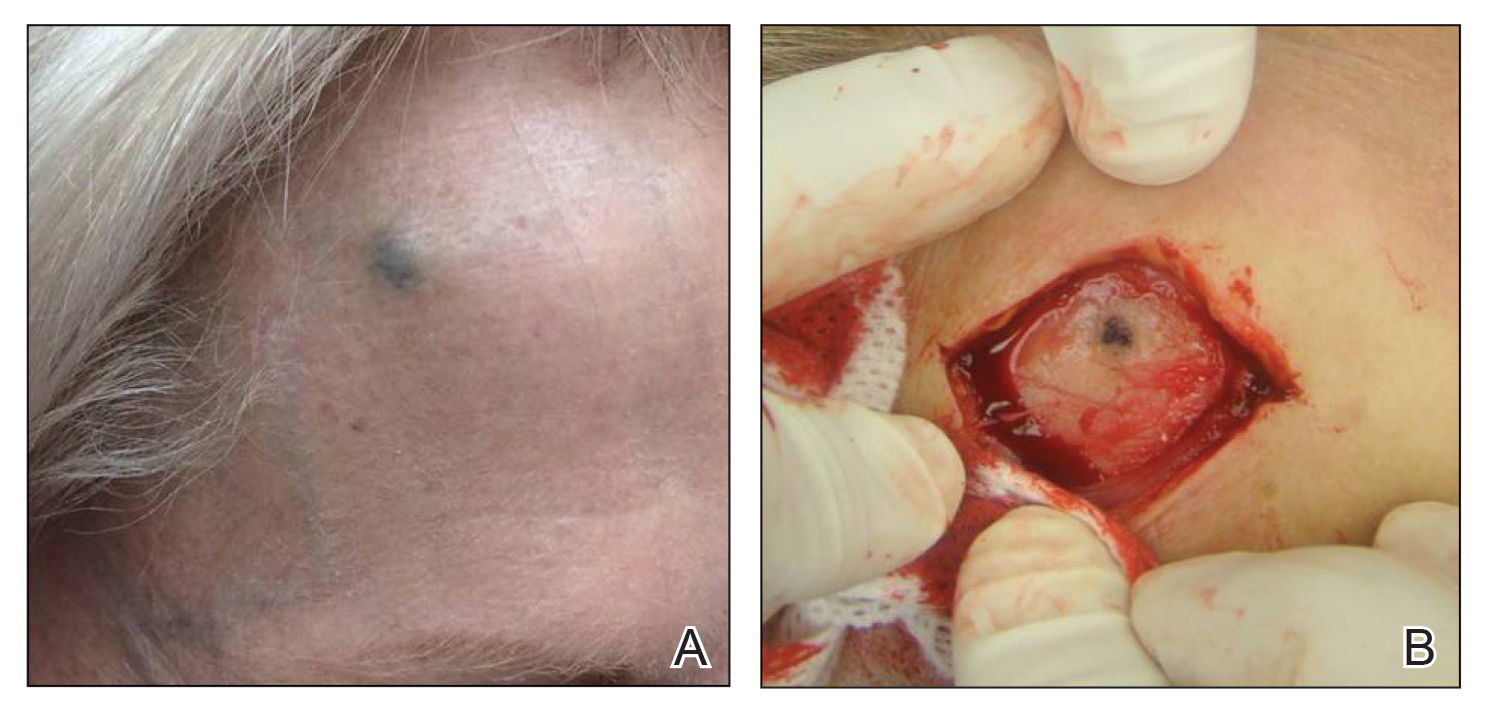
An elliptical excision of the lesion with 1-cm margins revealed a bluish-black mass extending through the dermis, through the frontalis muscle, and into the periosteum and frontal bone (Figure 1B). A No. 15 blade was then used to remove the remaining pigment from the outer table of the frontal bone. Histopathologic findings demonstrated a sarcoidal granulomatous dermatitis associated with abundant, nonpolarizable, black, granular pigment consistent with carbon tattoo. This foreign material was readily identifiable in large extracellular deposits and also within histiocytes, including numerous multinucleated giant cells (Figure 2). Immunostaining for MART-1 and SOX-10 antigens failed to demonstrate a melanocytic proliferation. These findings were consistent with a sarcoidal foreign-body granulomatous reaction to carbon tattoo following traumatic graphite implantation.
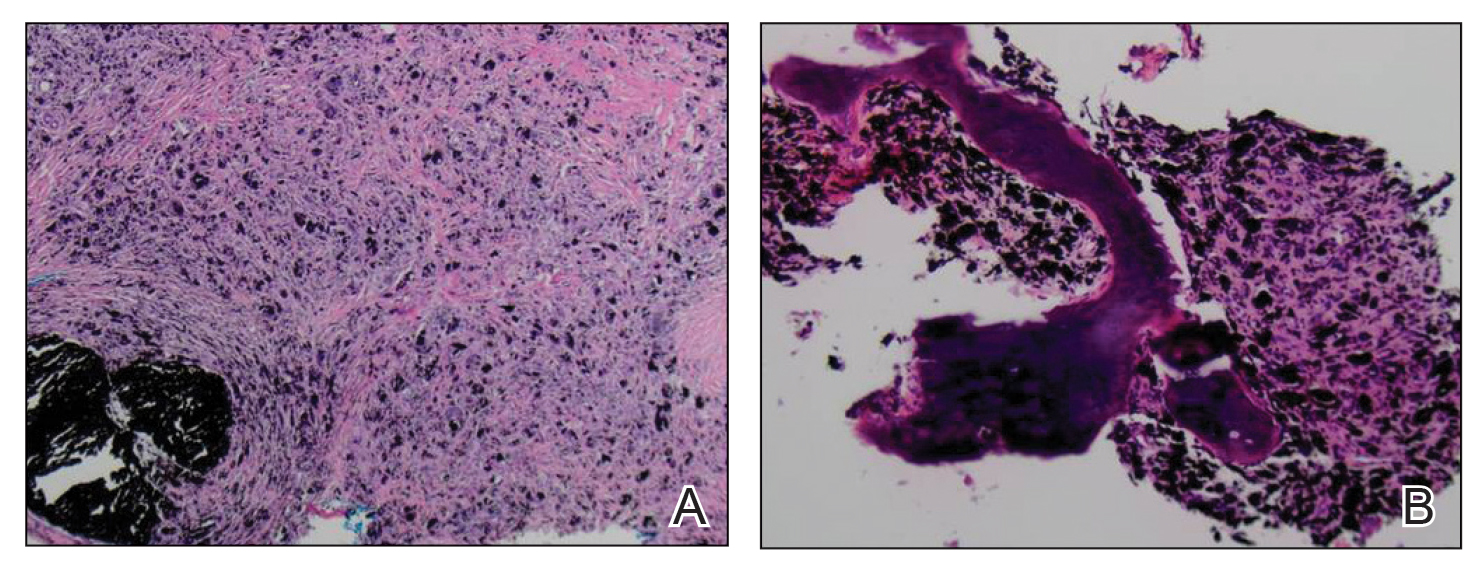
Granulomatous reactions to carbon tattoo may be sarcoidal (foreign-body granulomatous dermatitis), palisading, or rarely tuberculoid (caseating). Sarcoidal granulomatous tattoo reactions may occur in patients with sarcoidosis due to koebnerization, and histology alone is not discriminatory; however, in our patient, the absence of underlying sarcoidosis or clinical or histologic findings of sarcoidosis outside of the site of the pencil-core granuloma excluded that possibility.11 Pencil-core granulomas are characterized by a delayed foreign-body reaction to retained fragments of lead often years following a penetrating trauma with a pencil. Previous reports have described various lag times from injury to lesion growth of up to 58 years.1-10 Our patient claimed to have noticed the lesion growing and becoming painful only after a 62-year lag time following the initial trauma. To our knowledge, this is the longest lag time between the initial pencil injury and induction of the foreign-body reaction reported in the literature. Clinically, the lesion appeared and behaved very similar to a melanoma, prompting further treatment and evaluation.
It has been suggested that the lag period between the initial trauma and the rapid growth of the lesion may correspond to the amount of time required for the breakdown of the pencil lead to a critical size followed by the dispersal of those particles within the interstitium, where they can induce a granulomatous reaction.1,2,9 One case described a patient who reported that the growth and clinical change of the pencil-core granuloma only started when the patient accidentally hit the area where the trauma had occurred 31 years prior.1 This additional trauma may have caused further mechanical breakdown of the lead to set off the tissue reaction. In our case, the patient did not recall any additional trauma to the head prior to the onset of growth of the nodule on the forehead.
Our case indicates that carbon tattoo may be a possible sequela of a penetrating injury from a pencil with retained pencil lead fragments; however, many of these carbon tattoos may remain stable throughout the remainder of the patient’s life.
- Gormley RH, Kovach SJ III, Zhang PJ. Role for trauma in inducing pencil “lead” granuloma in the skin. J Am Acad Dermatol. 2010;62:1074-1075.
- Terasawa N, Kishimoto S, Kibe Y, et al. Graphite foreign body granuloma. Br J Dermatol. 1999;141:774-776.
- Fukunaga Y, Hashimoto I, Nakanishi H, et al. Pencil-core granuloma of the face: report of two rare cases. J Plast Reconstr Aesthet Surg. 2011;64:1235-1237.
- Aswani VH, Kim SL. Fifty-three years after a pencil puncture wound. Case Rep Dermatol. 2015;7:303-305.
- Taylor B, Frumkin A, Pitha JV. Delayed reaction to “lead” pencil simulating melanoma. Cutis. 1988;42:199-201.
- Granick MS, Erickson ER, Solomon MP. Pencil-core granuloma. Plast Reconstr Surg. 1992;89:136-138.
- Andreano J. Stump the experts. foreign body granuloma. J Dermatol Surg Oncol. 1992;18:277, 343.
- Yoshitatsu S, Takagi T. A case of giant pencil-core granuloma. J Dermatol. 2000;27:329-332.
- Hatano Y, Asada Y, Komada S, et al. A case of pencil core granuloma with an unusual temporal profile. Dermatology. 2000;201:151-153.
- Seitz IA, Silva BA, Schechter LS. Unusual sequela from a pencil stab wound reveals a retained graphite foreign body. Pediatr Emerg Care. 2014;30:568-570.
- Motaparthi K. Tattoo ink. In: Cockerell CJ, Hall BJ, eds. Nonneoplastic Dermatopathology. 2nd ed. Amirsys; 2016: 270.
To the Editor:
Trauma from a pencil tip can sometimes result in a fragment of lead being left embedded within the skin. Pencil lead is composed of 66% graphite carbon, 26% aluminum silicate, and 8% paraffin.1,2 While the toxicity of these individual elements is low, paraffin can cause nonallergic foreign-body reactions, aluminum silicate can induce epithelioid granulomatous reactions, and graphite has been reported to cause chronic granulomatous reactions in the lungs of those who work with graphite.2 Penetrating trauma with a pencil can result in the formation of a cutaneous granulomatous reaction that can sometimes occur years to decades after the initial injury.3,4 Several cases of pencil-core granulomas have been published, with lag times between the initial trauma and lesion growth as long as 58 years.1-10 The pencil-core granuloma may simulate malignant melanoma, as it presents clinically as a growing, darkly pigmented lesion, thus prompting biopsy. We present a case of a pencil-core granuloma that began to grow 62 years after the initial trauma.
A 72-year-old woman was referred to our clinic for evaluation of a dark nodule on the forehead. The lesion had been present since the age of 10 years, reportedly from an accidental stabbing with a pencil. The lesion had been flat, stable, and asymptomatic since the trauma occurred; however, the patient reported that approximately 9 months prior to presentation, it had started growing and became painful. Physical examination revealed a 1.0-cm, round, bluish-black nodule on the right superior forehead (Figure 1A). No satellite lesions or local lymphadenopathy were noted on general examination.

An elliptical excision of the lesion with 1-cm margins revealed a bluish-black mass extending through the dermis, through the frontalis muscle, and into the periosteum and frontal bone (Figure 1B). A No. 15 blade was then used to remove the remaining pigment from the outer table of the frontal bone. Histopathologic findings demonstrated a sarcoidal granulomatous dermatitis associated with abundant, nonpolarizable, black, granular pigment consistent with carbon tattoo. This foreign material was readily identifiable in large extracellular deposits and also within histiocytes, including numerous multinucleated giant cells (Figure 2). Immunostaining for MART-1 and SOX-10 antigens failed to demonstrate a melanocytic proliferation. These findings were consistent with a sarcoidal foreign-body granulomatous reaction to carbon tattoo following traumatic graphite implantation.

Granulomatous reactions to carbon tattoo may be sarcoidal (foreign-body granulomatous dermatitis), palisading, or rarely tuberculoid (caseating). Sarcoidal granulomatous tattoo reactions may occur in patients with sarcoidosis due to koebnerization, and histology alone is not discriminatory; however, in our patient, the absence of underlying sarcoidosis or clinical or histologic findings of sarcoidosis outside of the site of the pencil-core granuloma excluded that possibility.11 Pencil-core granulomas are characterized by a delayed foreign-body reaction to retained fragments of lead often years following a penetrating trauma with a pencil. Previous reports have described various lag times from injury to lesion growth of up to 58 years.1-10 Our patient claimed to have noticed the lesion growing and becoming painful only after a 62-year lag time following the initial trauma. To our knowledge, this is the longest lag time between the initial pencil injury and induction of the foreign-body reaction reported in the literature. Clinically, the lesion appeared and behaved very similar to a melanoma, prompting further treatment and evaluation.
It has been suggested that the lag period between the initial trauma and the rapid growth of the lesion may correspond to the amount of time required for the breakdown of the pencil lead to a critical size followed by the dispersal of those particles within the interstitium, where they can induce a granulomatous reaction.1,2,9 One case described a patient who reported that the growth and clinical change of the pencil-core granuloma only started when the patient accidentally hit the area where the trauma had occurred 31 years prior.1 This additional trauma may have caused further mechanical breakdown of the lead to set off the tissue reaction. In our case, the patient did not recall any additional trauma to the head prior to the onset of growth of the nodule on the forehead.
Our case indicates that carbon tattoo may be a possible sequela of a penetrating injury from a pencil with retained pencil lead fragments; however, many of these carbon tattoos may remain stable throughout the remainder of the patient’s life.
To the Editor:
Trauma from a pencil tip can sometimes result in a fragment of lead being left embedded within the skin. Pencil lead is composed of 66% graphite carbon, 26% aluminum silicate, and 8% paraffin.1,2 While the toxicity of these individual elements is low, paraffin can cause nonallergic foreign-body reactions, aluminum silicate can induce epithelioid granulomatous reactions, and graphite has been reported to cause chronic granulomatous reactions in the lungs of those who work with graphite.2 Penetrating trauma with a pencil can result in the formation of a cutaneous granulomatous reaction that can sometimes occur years to decades after the initial injury.3,4 Several cases of pencil-core granulomas have been published, with lag times between the initial trauma and lesion growth as long as 58 years.1-10 The pencil-core granuloma may simulate malignant melanoma, as it presents clinically as a growing, darkly pigmented lesion, thus prompting biopsy. We present a case of a pencil-core granuloma that began to grow 62 years after the initial trauma.
A 72-year-old woman was referred to our clinic for evaluation of a dark nodule on the forehead. The lesion had been present since the age of 10 years, reportedly from an accidental stabbing with a pencil. The lesion had been flat, stable, and asymptomatic since the trauma occurred; however, the patient reported that approximately 9 months prior to presentation, it had started growing and became painful. Physical examination revealed a 1.0-cm, round, bluish-black nodule on the right superior forehead (Figure 1A). No satellite lesions or local lymphadenopathy were noted on general examination.

An elliptical excision of the lesion with 1-cm margins revealed a bluish-black mass extending through the dermis, through the frontalis muscle, and into the periosteum and frontal bone (Figure 1B). A No. 15 blade was then used to remove the remaining pigment from the outer table of the frontal bone. Histopathologic findings demonstrated a sarcoidal granulomatous dermatitis associated with abundant, nonpolarizable, black, granular pigment consistent with carbon tattoo. This foreign material was readily identifiable in large extracellular deposits and also within histiocytes, including numerous multinucleated giant cells (Figure 2). Immunostaining for MART-1 and SOX-10 antigens failed to demonstrate a melanocytic proliferation. These findings were consistent with a sarcoidal foreign-body granulomatous reaction to carbon tattoo following traumatic graphite implantation.

Granulomatous reactions to carbon tattoo may be sarcoidal (foreign-body granulomatous dermatitis), palisading, or rarely tuberculoid (caseating). Sarcoidal granulomatous tattoo reactions may occur in patients with sarcoidosis due to koebnerization, and histology alone is not discriminatory; however, in our patient, the absence of underlying sarcoidosis or clinical or histologic findings of sarcoidosis outside of the site of the pencil-core granuloma excluded that possibility.11 Pencil-core granulomas are characterized by a delayed foreign-body reaction to retained fragments of lead often years following a penetrating trauma with a pencil. Previous reports have described various lag times from injury to lesion growth of up to 58 years.1-10 Our patient claimed to have noticed the lesion growing and becoming painful only after a 62-year lag time following the initial trauma. To our knowledge, this is the longest lag time between the initial pencil injury and induction of the foreign-body reaction reported in the literature. Clinically, the lesion appeared and behaved very similar to a melanoma, prompting further treatment and evaluation.
It has been suggested that the lag period between the initial trauma and the rapid growth of the lesion may correspond to the amount of time required for the breakdown of the pencil lead to a critical size followed by the dispersal of those particles within the interstitium, where they can induce a granulomatous reaction.1,2,9 One case described a patient who reported that the growth and clinical change of the pencil-core granuloma only started when the patient accidentally hit the area where the trauma had occurred 31 years prior.1 This additional trauma may have caused further mechanical breakdown of the lead to set off the tissue reaction. In our case, the patient did not recall any additional trauma to the head prior to the onset of growth of the nodule on the forehead.
Our case indicates that carbon tattoo may be a possible sequela of a penetrating injury from a pencil with retained pencil lead fragments; however, many of these carbon tattoos may remain stable throughout the remainder of the patient’s life.
- Gormley RH, Kovach SJ III, Zhang PJ. Role for trauma in inducing pencil “lead” granuloma in the skin. J Am Acad Dermatol. 2010;62:1074-1075.
- Terasawa N, Kishimoto S, Kibe Y, et al. Graphite foreign body granuloma. Br J Dermatol. 1999;141:774-776.
- Fukunaga Y, Hashimoto I, Nakanishi H, et al. Pencil-core granuloma of the face: report of two rare cases. J Plast Reconstr Aesthet Surg. 2011;64:1235-1237.
- Aswani VH, Kim SL. Fifty-three years after a pencil puncture wound. Case Rep Dermatol. 2015;7:303-305.
- Taylor B, Frumkin A, Pitha JV. Delayed reaction to “lead” pencil simulating melanoma. Cutis. 1988;42:199-201.
- Granick MS, Erickson ER, Solomon MP. Pencil-core granuloma. Plast Reconstr Surg. 1992;89:136-138.
- Andreano J. Stump the experts. foreign body granuloma. J Dermatol Surg Oncol. 1992;18:277, 343.
- Yoshitatsu S, Takagi T. A case of giant pencil-core granuloma. J Dermatol. 2000;27:329-332.
- Hatano Y, Asada Y, Komada S, et al. A case of pencil core granuloma with an unusual temporal profile. Dermatology. 2000;201:151-153.
- Seitz IA, Silva BA, Schechter LS. Unusual sequela from a pencil stab wound reveals a retained graphite foreign body. Pediatr Emerg Care. 2014;30:568-570.
- Motaparthi K. Tattoo ink. In: Cockerell CJ, Hall BJ, eds. Nonneoplastic Dermatopathology. 2nd ed. Amirsys; 2016: 270.
- Gormley RH, Kovach SJ III, Zhang PJ. Role for trauma in inducing pencil “lead” granuloma in the skin. J Am Acad Dermatol. 2010;62:1074-1075.
- Terasawa N, Kishimoto S, Kibe Y, et al. Graphite foreign body granuloma. Br J Dermatol. 1999;141:774-776.
- Fukunaga Y, Hashimoto I, Nakanishi H, et al. Pencil-core granuloma of the face: report of two rare cases. J Plast Reconstr Aesthet Surg. 2011;64:1235-1237.
- Aswani VH, Kim SL. Fifty-three years after a pencil puncture wound. Case Rep Dermatol. 2015;7:303-305.
- Taylor B, Frumkin A, Pitha JV. Delayed reaction to “lead” pencil simulating melanoma. Cutis. 1988;42:199-201.
- Granick MS, Erickson ER, Solomon MP. Pencil-core granuloma. Plast Reconstr Surg. 1992;89:136-138.
- Andreano J. Stump the experts. foreign body granuloma. J Dermatol Surg Oncol. 1992;18:277, 343.
- Yoshitatsu S, Takagi T. A case of giant pencil-core granuloma. J Dermatol. 2000;27:329-332.
- Hatano Y, Asada Y, Komada S, et al. A case of pencil core granuloma with an unusual temporal profile. Dermatology. 2000;201:151-153.
- Seitz IA, Silva BA, Schechter LS. Unusual sequela from a pencil stab wound reveals a retained graphite foreign body. Pediatr Emerg Care. 2014;30:568-570.
- Motaparthi K. Tattoo ink. In: Cockerell CJ, Hall BJ, eds. Nonneoplastic Dermatopathology. 2nd ed. Amirsys; 2016: 270.
Practice Points
- Pencil-core granulomas can arise even decades after the lead is embedded in the skin.
- It is important to biopsy to confirm the diagnosis, as pencil-core granulomas can very closely mimic melanomas.
Antibiotic choices for inpatients with SSTIs vary by race
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
FROM JAMA NETWORK OPEN
Diet in Wound Care: Can Nutrition Impact Healing?
Dermatologists commonly manage a variety of wounds in the outpatient setting. Wound healing requires a multifaceted approach that often includes topical and oral therapies, adjustment of mechanical factors, and behavioral and lifestyle modifications. Physiologically, wound healing requires an inflammatory phase, a proliferative phase, and a remodeling phase. Chronic wounds undergo a prolonged inflammatory response hindered by decreased growth factors and increased wound bioburden.1 Malnutrition has been routinely associated with wound chronicity and serves as a modifiable risk factor that may improve wound healing outcomes.2
Although the causes of wounds encountered in dermatology vary extensively, the importance of nutrition underlies all wound healing. Caloric needs in wound healing have been estimated at 30 to 40 kcal/kg dependent on baseline body weight, age, medical comorbidities, activity level, stage of wound healing, wound size, and number of wounds.1,3,4 Nutritional supplementation is patient dependent, but this article serves to review the existing literature on macronutrient and micronutrient supplementation to clarify the potentially complementary role for nutritional support in chronic wounds. All patients should be screened with a thorough history, review of systems, and physical examination for existing nutrient deficiencies. Patients with age-related or chronic diseases are at increased risk for nutritional deficiency, and focused laboratory testing may be warranted. Supplementation for specific deficiencies with help from a registered dietician is recommended.
Macronutrients for Wound Healing
Protein—Protein is the most widely known macronutrient required for wound healing. The primary function of dietary protein is to provide amino acids to perform physiologic functions.5 Not only does cutaneous injury increase the metabolic needs of the wounded area, but large amounts of protein can be continually lost through wound exudates. Protein is necessary for the immune response required to transition from inflammatory to proliferative phases of wound healing.6 Protein energy deficiency has been reported to reduce fibroblast activity, delay angiogenesis, and decrease collagen formation.7 Additionally, protein is required for the formation of inflammatory cells and maintenance of oncotic pressure, specifically in venous insufficiency wounds.1
The current recommended dietary allowance for protein in healthy adults is 0.8 g/kg daily of body weight. In patients with pressure ulcerations, a goal recommended dietary allowance of 1.25 to 2.0 g/kg daily of body weight, dependent on ulceration size, has been recommended by the National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel.8 This recommendation was based on a series of studies that reported enhanced healing rates in patients with pressure ulcers receiving higher-protein diets.9 The largest study to date was double-blinded and included 89 residents of long-term care facilities with stage II to stage IV pressure ulcers.10 Participants were randomized to receive commercial protein supplementation vs placebo. At the end of 8 weeks, a statistically significant difference was seen in mean (SD) pressure ulcer scale for healing scores (3.55 [4.66] vs 3.22 [4.11]; P<.05).10 A 2014 Cochrane review failed to identify benefit associated with nutritional interventions for either the prevention and/or treatment of pressure ulcers.11 Specific recommendations on protein intake for other types of chronic wounds have not been proposed. Protein supplementation generally is provided orally, if tolerated. Liquid supplements such as Boost (Nestlé), Carnation Breakfast Essentials (Nestlé), NuBasics (SupremeMed), Resource (Nestlé Health Science), and Ensure (Abbott Laboratories) are frequently used to supplement both protein and caloric intake. Protein oversupplementation has not been associated with improved outcomes and may cause or exacerbate other medical comorbidities.
Fatty Acids for Wound Healing
Wound healing is an anabolic process that requires adequate intake of substrates such as glucose and fat. Carbohydrates serve as the major energy source required for wound healing, while fats are thought to play roles in cell membrane development and modulation of cellular signaling.1 Fats utilize a unique pathway for energy production through beta-oxidation and the production of adenosine triphosphate, allowing available protein to be harnessed for wound healing.1 Omega-3 and omega-6 fatty acids serve as precursors to prostaglandins, leukotrienes, and thromboxane—all key mediators of the inflammatory phase of wound healing.3 Omega-3 fatty acids are thought to downregulate genes involved in proinflammatory pathways,12 as well as to diminish lymphocyte proliferation and levels of IL-1β, tumor necrosis factor α, and IL-6 in vitro.13 In vivo studies assessing the impact of omega-3 fatty acid supplementation on wound healing are minimal, and the role of dietary supplementation for this indication remains unknown. Fish oil contains the omega-3 fatty acid–rich eicosapentaenoic acid and docosahexaenoic acid, which has been compared to mineral oil supplementation for wound healing in healthy adults. When fish oil was supplemented for 4 weeks, no significant differences were identified in time to complete wound healing between groups. Interestingly, significantly higher levels of the proinflammatory cytokine IL-1β were identified in blister fluid at 24 hours after blistering vs the placebo group (t=2.52, df=25, P<.05).14 Prior studies evaluating wound healing in animal models similarly identified longer times to re-epithelialization after omega-3 polyunsaturated fatty acid supplementation orally and topically.15,16 The fatty acid quality and composition consumed also may impact wound healing, as high-fat diets that are not rich in omega-3 fatty acids have been shown to promote inflammation and impair wound healing in rats, but this has not been thoroughly explored in human trials.17 Although adequate intake of these macronutrients is important, excessive intake may be harmful. Larger prospective trials are needed to shed light on the dose and composition of fatty acid supplementation that may optimize wound healing.
Vitamins and Micronutrients Required for Wound Healing
Vitamin A—Many vitamins serve as cofactors for the enzymatic processes required in wound healing. Vitamin A is an essential fat-soluble vitamin that serves a variety of dermatologic functions and promotes wound healing through stimulation of fibroblasts and ground substance, and it facilitates epithelial cell differentiation when applied topically.3,18 Vitamin A works through the activation of retinoid receptors on endothelial cells, fibroblasts, keratinocytes, melanocytes, and sebocytes, and has purported anti-inflammatory effects that aid the healing of open wounds.3 Additionally, vitamin A is thought to enhance cytokine release in the inflammatory phase of wound healing.19 Supplemental vitamin A has been associated with positive effects on acute wound healing, burns, and radiation injuries.3 The utility of vitamin A supplementation in chronic wounds remains unknown; however, it has been shown to be beneficial in patients with inflammatory disease, such as rheumatoid arthritis, on corticosteroid therapy. Vitamin A supplementation in this population has been shown to counteract the negative effects of corticosteroids on wound healing via downregulation of transforming growth factor β and insulinlike growth factor 1.20 Vitamin A deficiency has been associated with impaired progression through inflammatory and remodeling phases of healing due to altered B-cell and T-cell function and antibody production.1 Some experts recommend short courses of oral vitamin A supplementation to enhance wound healing at doses between 10,000 and 25,000 IU daily.2,3 Large, population-based studies are needed, and the safety supporting this recommendation in all patients remains unknown.
Vitamin C—Vitamin C is widely known for its role in collagen formation, immunomodulation, and antioxidant capacity.1 Although vitamin C deficiency is associated with decreased collagen synthesis and impaired wound healing,21 the utility of long-term supplementation in patients who are not deficient remains unexplored. A systematic review evaluating interventional studies utilizing vitamin C supplementation on pressure ulcerations and surgical wound healing concluded that convincing evidence exists only for supplementation with at least 500 mg of vitamin C. The authors noted, “There is little evidence for improved healing of surgical wounds by high-dose single vitamin C supplementation (1–3 g/day).”22 In a prospective, randomized, controlled trial, 20 patients with pressure ulcerations were supplemented with vitamin C vs placebo with a mean reduction in pressure-sore area of 84% after 1 month in the vitamin C–supplemented group compared to 42.7% in the placebo group (P<.005). A limitation of this study is the small population.23 One current recommendation for vitamin C supplementation in chronic wounds is for 500 mg daily in uncomplicated wounds to 2 g daily in severe wounds.3 Additional studies have suggested that the benefits of vitamin C supplementation are maximized when given in combination with zinc and arginine.22 At this time, evaluation for vitamin C deficiency and appropriate supplementation in patients with chronic wounds is needed.
Zinc—Minerals similarly play important roles in enzymatic regulation. Hundreds of zinc-containing enzymes are involved in wound healing and are required in tissue repair, growth, antioxidant capacity, and immune function.1,24 Zinc is specifically critical to collagen, DNA, RNA, and protein synthesis, as well as cellular proliferation.4 Zinc deficiency has been encountered in the setting of chronic wounds with extensive drainage, decreased dietary intake, or excessive gastrointestinal losses.25 Although many studies exist evaluating the utility of zinc supplementation on wound healing, many are confounded by multinutrient supplementation. No studies to date support zinc supplementation when zinc deficiency is absent. Patient assessment for medications or conditions that may impact zinc metabolism should be completed. Importantly, zinc supplementation can interfere with the absorption of other cations, so excessive supplementation should be avoided.1
Amino Acids for Wound Healing
Arginine—Arginine is an essential amino acid that serves as a substrate for cellular proliferation, collagen deposition, and lymphocyte function.8,26,27 Arginine serves as the biologic precursor for nitric oxide (NO), a substrate that has important wound healing properties. Nitric oxide metabolites have been shown to positively regulate wound repair while NO metabolites are reduced in wound environments in diabetic ulcerations.28,29 Arginine also is a proline precursor, an essential building block for collagen synthesis,6,30 and a stimulator of growth hormone and T cells.30,31 Animal studies have suggested L-arginine supplementation may reverse impaired NO synthesis in diabetic wounds.28 A single randomized trial assessing differing doses of arginine supplementation on stage II or stage IV pressure ulcers noted an almost two-fold improvement in healing time.32 However, human studies have not shown increased rates of re-epithelialization of skin graft donor sites when provided oral or parenteral arginine supplementation.33 Inadequate data currently exist to support regular arginine supplementation for all types of wounds, and no safe dose of daily arginine intake has been established.
Glutamine—Similarly, glutamine supplementation has been proposed to accelerate wound healing due to its role as a primary metabolic fuel source for rapidly proliferating cells such as epithelial cells and fibroblasts.8 Glutamine is thought to induce expression of heat-shock proteins and protect against inflammatory and infectious wound complications.34 Additionally, glutamine is thought to increase tissue insulin sensitivity, which may prove beneficial in wounds, as topical insulin previously has been shown in animal and human models to promote healing.35 Glutamine is thought to play a role in the inflammatory phase of wound healing via superoxide production, leukocyte apoptosis, and phagocytosis.6,34,36 Unfortunately, numerous randomized trials on glutamine supplementation have resulted in conflicting evidence confounded by multisupplementation within the same trial.37,38 A double-blind, randomized, controlled trial of 270 participants assessed the effect of oral supplementation with arginine, glutamine, or β-hydroxy-β-methylbutyrate vs control in the healing time of diabetic foot ulcerations. Significant differences in wound closure time at week 16 were only identified in participants with low albumin levels (≤40 g/L) who were supplemented (50.8%) vs the control group (34.9%; P=.0325) and in those with poor limb perfusion (ankle-brachial index of <1.0) who were supplemented (60.3%) vs the control group (39.3%; P=.0079).39 Ongoing clinical trials evaluating the effects of glutamine supplementation on differing wound types will hopefully shed light on the efficacy of supplementation.
Final Thoughts
Wound healing is multifactorial and should consider the health status and medical comorbidities of each patient treated. We propose an individualized approach to wound healing that includes exploration of specific macronutrient and micronutrient deficiencies, as malnutrition has been associated with wound chronicity and serves as a modifiable risk factor to improve healing.2 The evidence backing specific nutrient supplementation in patients with deficiencies is strong and should be considered in patients with chronic wounds. Adequate caloric intake and protein content should be recommended for most wound patients; however, excessive protein intake has not been beneficial in wound healing. The data behind specific amino acid and vitamin supplementation are limited at this time. As with other therapeutics, there is likely an appropriate dose for supplementation that has not yet been elucidated. Consideration of wound type, size, depth, exudate, and underlying cause are important to optimize healing and tailor nutritional supplementation to each patient. We hope future studies will illuminate the complementary role of dietary intake and nutrient supplementation for the treatment of chronic nonhealing wounds.
- Quain AM, Khardori NM. Nutrition in wound care management: a comprehensive overview. Wounds. 2015;27:327-335.
- Stechmiller JK. Understanding the role of nutrition and wound healing. Nutr Clin Pract. 2010;25:61-68. doi:10.1177/0884533609358997
- Molnar JA, Underdown MJ, Clark WA. Nutrition and chronic wounds. Adv Wound Care (New Rochelle). 2014;3:663-681. doi:10.1089/wound.2014.0530
- Dorner B, Posthauer ME, Thomas D; Panel NPUA. The role of nutrition in pressure ulcer prevention and treatment: National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care. 2009;22:212-221. doi:10.1097/01.ASW.0000350838.11854.0a
- Collins N. Protein and wound healing. Adv Skin Wound Care. 2001;14:288-289. doi:10.1097/00129334-200111000-00008
- Barchitta M, Maugeri A, Favara G, et al. Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin [published online March 5, 2019]. Int J Mol Sci. doi:10.3390/ijms20051119
- Harris CL, Fraser C. Malnutrition in the institutionalized elderly: the effects on wound healing. Ostomy Wound Manage. 2004;50:54-63.
- Saghaleini SH, Dehghan K, Shadvar K, et al. Pressure ulcer and nutrition. Indian J Crit Care Med. 2018;22:283-289. doi:10.4103/ijccm.IJCCM_277_17
- Breslow RA, Hallfrisch J, Guy DG, et al. The importance of dietary protein in healing pressure ulcers. J Am Geriatr Soc. 1993;41:357-362. doi:10.1111/j.1532-5415.1993.tb06940.x
- Lee SK, Posthauer ME, Dorner B, et al. Pressure ulcer healing with a concentrated, fortified, collagen protein hydrolysate supplement: a randomized controlled trial. Adv Skin Wound Care. 2006;19:92-96. doi:10.1097/00129334-200603000-00011
- Langer G, Fink A. Nutritional interventions for preventing and treating pressure ulcers. Cochrane Database Syst Rev. 2014;6:CD003216. doi:10.1002/14651858.CD003216.pub2
- Bouwens M, van de Rest O, Dellschaft N, et al. Fish-oil supplementation induces antiinflammatory gene expression profiles in human blood mononuclear cells. Am J Clin Nutr. 2009;90:415-424. doi:10.3945/ajcn.2009.27680
- Meydani SN, Endres S, Woods MM, et al. Oral (n-3) fatty acid supplementation suppresses cytokine production and lymphocyte proliferation: comparison between young and older women. J Nutr. 1991;121:547-555. doi:10.1093/jn/121.4.547
- McDaniel JC, Belury M, Ahijevych K, et al. Omega-3 fatty acids effect on wound healing. Wound Repair Regen. 2008;16:337-345. doi:10.1111/j.1524-475X.2008.00388.x
- Mooney MA, Vaughn DM, Reinhart GA, et al. Evaluation of the effects of omega-3 fatty acid-containing diets on the inflammatory stage of wound healing in dogs. Am J Vet Res. 1998;59:859-863.
- Cardoso CR, Souza MA, Ferro EA, et al. Influence of topical administration of n-3 and n-6 essential and n-9 nonessential fatty acids on the healing of cutaneous wounds. Wound Repair Regen. 2004;12:235-243. doi:10.1111/j.1067-1927.2004.012216.x
- Rosa DF, Sarandy MM, Novaes RD, et al. High-fat diet and alcohol intake promotes inflammation and impairs skin wound healing in Wistar rats. Mediators Inflamm. 2018;2018:4658583. doi:10.1155/2018/4658583
- Levenson SM, Gruber CA, Rettura G, et al. Supplemental vitamin A prevents the acute radiation-induced defect in wound healing. Ann Surg. 1984;200:494-512. doi:10.1097/00000658-198410000-00011
- Palmieri B, Vadalà M, Laurino C. Nutrition in wound healing: investigation of the molecular mechanisms, a narrative review. J Wound Care. 2019;28:683-693. doi:10.12968/jowc.2019.28.10.683
- Ehrlich HP, Hunt TK. Effects of cortisone and vitamin A on wound healing. Ann Surg. 1968;167:324-328. doi:10.1097/00000658-196803000-00004
- Pullar JM, Carr AC, Vissers MCM. The roles of vitamin C in skin health [published online August 12, 2017]. Nutrients. doi:10.3390/nu9080866
- Ellinger S, Stehle P. Efficacy of vitamin supplementation in situations with wound healing disorders: results from clinical intervention studies. Curr Opin Clin Nutr Metab Care. 2009;12:588-595. doi:10.1097/MCO.0b013e328331a5b5
- Taylor TV, Rimmer S, Day B, et al. Ascorbic acid supplementation in the treatment of pressure-sores. Lancet. 1974;2:544-546. doi:10.1016/s0140-6736(74)91874-1
- Ibs KH, Rink L. Zinc-altered immune function. J Nutr. 2003;133(5 suppl 1):1452S-1456S. doi:10.1093/jn/133.5.1452S
- Hoffman M, Micheletti RG, Shields BE. Nutritional dermatoses in the hospitalized patient. Cutis. 2020;105:296-302, 308, E1-E5.
- Chow O, Barbul A. Immunonutrition: role in wound healing and tissue regeneration. Adv Wound Care (New Rochelle). 2014;3:46-53. doi:10.1089/wound.2012.0415
- Singh K, Coburn LA, Barry DP, et al. L-arginine uptake by cationic amino acid transporter 2 is essential for colonic epithelial cell restitution. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1061-G1073. doi:10.1152/ajpgi.00544.2011
- Witte MB, Thornton FJ, Tantry U, et al. L-Arginine supplementation enhances diabetic wound healing: involvement of the nitric oxide synthase and arginase pathways. Metabolism. 2002;51:1269-1273. doi:10.1053/meta.2002.35185
- Witte MB, Barbul A. Role of nitric oxide in wound repair. Am J Surg. 2002;183:406-412. doi:10.1016/s0002-9610(02)00815-2
- Barbul A. Proline precursors to sustain Mammalian collagen synthesis. J Nutr. 2008;138:2021S-2024S. doi:10.1093/jn/138.10.2021S
- Wu G, Bazer FW, Davis TA, et al. Arginine metabolism and nutrition in growth, health and disease. Amino Acids. 2009;37:153-168. doi:10.1007/s00726-008-0210-y
- Leigh B, Desneves K, Rafferty J, et al. The effect of different doses of an arginine-containing supplement on the healing of pressure ulcers. J Wound Care. 2012;21:150-156. doi:10.12968/jowc.2012.21.3.150
- Debats IB, Koeneman MM, Booi DI, et al. Intravenous arginine and human skin graft donor site healing: a randomized controlled trial. Burns. 2011;37:420-426. doi:10.1016/j.burns.2010.06.003
- Wischmeyer PE. Glutamine and heat shock protein expression. Nutrition. 2002;18:225-228. doi:10.1016/s0899-9007(01)00796-1
- Wang J, Xu J. Effects of topical insulin on wound healing: a review of animal and human evidences. Diabetes Metab Syndr Obes. 2020;13:719-727. doi:10.2147/DMSO.S237294
- Newsholme P. Why is L-glutamine metabolism important to cells of the immune system in health, postinjury, surgery or infection?J Nutr. 2001;131(9 suppl):2515S-2522S; discussion 2523S-2524S. doi:10.1093/jn/131.9.2515S
- Aquino VM, Harvey AR, Garvin JH, et al. A double-blind randomized placebo-controlled study of oral glutamine in the prevention of mucositis in children undergoing hematopoietic stem cell transplantation: a pediatric blood and marrow transplant consortium study. Bone Marrow Transplant. 2005;36:611-616. doi:10.1038/sj.bmt.1705084
- Ward E, Smith M, Henderson M, et al. The effect of high-dose enteral glutamine on the incidence and severity of mucositis in paediatric oncology patients. Eur J Clin Nutr. 2009;63:134-140. doi:10.1038/sj.ejcn.1602894
- Armstrong DG, Hanft JR, Driver VR, et al. Effect of oral nutritional supplementation on wound healing in diabetic foot ulcers: a prospective randomized controlled trial. Diabet Med. 2014;31:1069-1077. doi:10.1111/dme.12509
Dermatologists commonly manage a variety of wounds in the outpatient setting. Wound healing requires a multifaceted approach that often includes topical and oral therapies, adjustment of mechanical factors, and behavioral and lifestyle modifications. Physiologically, wound healing requires an inflammatory phase, a proliferative phase, and a remodeling phase. Chronic wounds undergo a prolonged inflammatory response hindered by decreased growth factors and increased wound bioburden.1 Malnutrition has been routinely associated with wound chronicity and serves as a modifiable risk factor that may improve wound healing outcomes.2
Although the causes of wounds encountered in dermatology vary extensively, the importance of nutrition underlies all wound healing. Caloric needs in wound healing have been estimated at 30 to 40 kcal/kg dependent on baseline body weight, age, medical comorbidities, activity level, stage of wound healing, wound size, and number of wounds.1,3,4 Nutritional supplementation is patient dependent, but this article serves to review the existing literature on macronutrient and micronutrient supplementation to clarify the potentially complementary role for nutritional support in chronic wounds. All patients should be screened with a thorough history, review of systems, and physical examination for existing nutrient deficiencies. Patients with age-related or chronic diseases are at increased risk for nutritional deficiency, and focused laboratory testing may be warranted. Supplementation for specific deficiencies with help from a registered dietician is recommended.
Macronutrients for Wound Healing
Protein—Protein is the most widely known macronutrient required for wound healing. The primary function of dietary protein is to provide amino acids to perform physiologic functions.5 Not only does cutaneous injury increase the metabolic needs of the wounded area, but large amounts of protein can be continually lost through wound exudates. Protein is necessary for the immune response required to transition from inflammatory to proliferative phases of wound healing.6 Protein energy deficiency has been reported to reduce fibroblast activity, delay angiogenesis, and decrease collagen formation.7 Additionally, protein is required for the formation of inflammatory cells and maintenance of oncotic pressure, specifically in venous insufficiency wounds.1
The current recommended dietary allowance for protein in healthy adults is 0.8 g/kg daily of body weight. In patients with pressure ulcerations, a goal recommended dietary allowance of 1.25 to 2.0 g/kg daily of body weight, dependent on ulceration size, has been recommended by the National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel.8 This recommendation was based on a series of studies that reported enhanced healing rates in patients with pressure ulcers receiving higher-protein diets.9 The largest study to date was double-blinded and included 89 residents of long-term care facilities with stage II to stage IV pressure ulcers.10 Participants were randomized to receive commercial protein supplementation vs placebo. At the end of 8 weeks, a statistically significant difference was seen in mean (SD) pressure ulcer scale for healing scores (3.55 [4.66] vs 3.22 [4.11]; P<.05).10 A 2014 Cochrane review failed to identify benefit associated with nutritional interventions for either the prevention and/or treatment of pressure ulcers.11 Specific recommendations on protein intake for other types of chronic wounds have not been proposed. Protein supplementation generally is provided orally, if tolerated. Liquid supplements such as Boost (Nestlé), Carnation Breakfast Essentials (Nestlé), NuBasics (SupremeMed), Resource (Nestlé Health Science), and Ensure (Abbott Laboratories) are frequently used to supplement both protein and caloric intake. Protein oversupplementation has not been associated with improved outcomes and may cause or exacerbate other medical comorbidities.
Fatty Acids for Wound Healing
Wound healing is an anabolic process that requires adequate intake of substrates such as glucose and fat. Carbohydrates serve as the major energy source required for wound healing, while fats are thought to play roles in cell membrane development and modulation of cellular signaling.1 Fats utilize a unique pathway for energy production through beta-oxidation and the production of adenosine triphosphate, allowing available protein to be harnessed for wound healing.1 Omega-3 and omega-6 fatty acids serve as precursors to prostaglandins, leukotrienes, and thromboxane—all key mediators of the inflammatory phase of wound healing.3 Omega-3 fatty acids are thought to downregulate genes involved in proinflammatory pathways,12 as well as to diminish lymphocyte proliferation and levels of IL-1β, tumor necrosis factor α, and IL-6 in vitro.13 In vivo studies assessing the impact of omega-3 fatty acid supplementation on wound healing are minimal, and the role of dietary supplementation for this indication remains unknown. Fish oil contains the omega-3 fatty acid–rich eicosapentaenoic acid and docosahexaenoic acid, which has been compared to mineral oil supplementation for wound healing in healthy adults. When fish oil was supplemented for 4 weeks, no significant differences were identified in time to complete wound healing between groups. Interestingly, significantly higher levels of the proinflammatory cytokine IL-1β were identified in blister fluid at 24 hours after blistering vs the placebo group (t=2.52, df=25, P<.05).14 Prior studies evaluating wound healing in animal models similarly identified longer times to re-epithelialization after omega-3 polyunsaturated fatty acid supplementation orally and topically.15,16 The fatty acid quality and composition consumed also may impact wound healing, as high-fat diets that are not rich in omega-3 fatty acids have been shown to promote inflammation and impair wound healing in rats, but this has not been thoroughly explored in human trials.17 Although adequate intake of these macronutrients is important, excessive intake may be harmful. Larger prospective trials are needed to shed light on the dose and composition of fatty acid supplementation that may optimize wound healing.
Vitamins and Micronutrients Required for Wound Healing
Vitamin A—Many vitamins serve as cofactors for the enzymatic processes required in wound healing. Vitamin A is an essential fat-soluble vitamin that serves a variety of dermatologic functions and promotes wound healing through stimulation of fibroblasts and ground substance, and it facilitates epithelial cell differentiation when applied topically.3,18 Vitamin A works through the activation of retinoid receptors on endothelial cells, fibroblasts, keratinocytes, melanocytes, and sebocytes, and has purported anti-inflammatory effects that aid the healing of open wounds.3 Additionally, vitamin A is thought to enhance cytokine release in the inflammatory phase of wound healing.19 Supplemental vitamin A has been associated with positive effects on acute wound healing, burns, and radiation injuries.3 The utility of vitamin A supplementation in chronic wounds remains unknown; however, it has been shown to be beneficial in patients with inflammatory disease, such as rheumatoid arthritis, on corticosteroid therapy. Vitamin A supplementation in this population has been shown to counteract the negative effects of corticosteroids on wound healing via downregulation of transforming growth factor β and insulinlike growth factor 1.20 Vitamin A deficiency has been associated with impaired progression through inflammatory and remodeling phases of healing due to altered B-cell and T-cell function and antibody production.1 Some experts recommend short courses of oral vitamin A supplementation to enhance wound healing at doses between 10,000 and 25,000 IU daily.2,3 Large, population-based studies are needed, and the safety supporting this recommendation in all patients remains unknown.
Vitamin C—Vitamin C is widely known for its role in collagen formation, immunomodulation, and antioxidant capacity.1 Although vitamin C deficiency is associated with decreased collagen synthesis and impaired wound healing,21 the utility of long-term supplementation in patients who are not deficient remains unexplored. A systematic review evaluating interventional studies utilizing vitamin C supplementation on pressure ulcerations and surgical wound healing concluded that convincing evidence exists only for supplementation with at least 500 mg of vitamin C. The authors noted, “There is little evidence for improved healing of surgical wounds by high-dose single vitamin C supplementation (1–3 g/day).”22 In a prospective, randomized, controlled trial, 20 patients with pressure ulcerations were supplemented with vitamin C vs placebo with a mean reduction in pressure-sore area of 84% after 1 month in the vitamin C–supplemented group compared to 42.7% in the placebo group (P<.005). A limitation of this study is the small population.23 One current recommendation for vitamin C supplementation in chronic wounds is for 500 mg daily in uncomplicated wounds to 2 g daily in severe wounds.3 Additional studies have suggested that the benefits of vitamin C supplementation are maximized when given in combination with zinc and arginine.22 At this time, evaluation for vitamin C deficiency and appropriate supplementation in patients with chronic wounds is needed.
Zinc—Minerals similarly play important roles in enzymatic regulation. Hundreds of zinc-containing enzymes are involved in wound healing and are required in tissue repair, growth, antioxidant capacity, and immune function.1,24 Zinc is specifically critical to collagen, DNA, RNA, and protein synthesis, as well as cellular proliferation.4 Zinc deficiency has been encountered in the setting of chronic wounds with extensive drainage, decreased dietary intake, or excessive gastrointestinal losses.25 Although many studies exist evaluating the utility of zinc supplementation on wound healing, many are confounded by multinutrient supplementation. No studies to date support zinc supplementation when zinc deficiency is absent. Patient assessment for medications or conditions that may impact zinc metabolism should be completed. Importantly, zinc supplementation can interfere with the absorption of other cations, so excessive supplementation should be avoided.1
Amino Acids for Wound Healing
Arginine—Arginine is an essential amino acid that serves as a substrate for cellular proliferation, collagen deposition, and lymphocyte function.8,26,27 Arginine serves as the biologic precursor for nitric oxide (NO), a substrate that has important wound healing properties. Nitric oxide metabolites have been shown to positively regulate wound repair while NO metabolites are reduced in wound environments in diabetic ulcerations.28,29 Arginine also is a proline precursor, an essential building block for collagen synthesis,6,30 and a stimulator of growth hormone and T cells.30,31 Animal studies have suggested L-arginine supplementation may reverse impaired NO synthesis in diabetic wounds.28 A single randomized trial assessing differing doses of arginine supplementation on stage II or stage IV pressure ulcers noted an almost two-fold improvement in healing time.32 However, human studies have not shown increased rates of re-epithelialization of skin graft donor sites when provided oral or parenteral arginine supplementation.33 Inadequate data currently exist to support regular arginine supplementation for all types of wounds, and no safe dose of daily arginine intake has been established.
Glutamine—Similarly, glutamine supplementation has been proposed to accelerate wound healing due to its role as a primary metabolic fuel source for rapidly proliferating cells such as epithelial cells and fibroblasts.8 Glutamine is thought to induce expression of heat-shock proteins and protect against inflammatory and infectious wound complications.34 Additionally, glutamine is thought to increase tissue insulin sensitivity, which may prove beneficial in wounds, as topical insulin previously has been shown in animal and human models to promote healing.35 Glutamine is thought to play a role in the inflammatory phase of wound healing via superoxide production, leukocyte apoptosis, and phagocytosis.6,34,36 Unfortunately, numerous randomized trials on glutamine supplementation have resulted in conflicting evidence confounded by multisupplementation within the same trial.37,38 A double-blind, randomized, controlled trial of 270 participants assessed the effect of oral supplementation with arginine, glutamine, or β-hydroxy-β-methylbutyrate vs control in the healing time of diabetic foot ulcerations. Significant differences in wound closure time at week 16 were only identified in participants with low albumin levels (≤40 g/L) who were supplemented (50.8%) vs the control group (34.9%; P=.0325) and in those with poor limb perfusion (ankle-brachial index of <1.0) who were supplemented (60.3%) vs the control group (39.3%; P=.0079).39 Ongoing clinical trials evaluating the effects of glutamine supplementation on differing wound types will hopefully shed light on the efficacy of supplementation.
Final Thoughts
Wound healing is multifactorial and should consider the health status and medical comorbidities of each patient treated. We propose an individualized approach to wound healing that includes exploration of specific macronutrient and micronutrient deficiencies, as malnutrition has been associated with wound chronicity and serves as a modifiable risk factor to improve healing.2 The evidence backing specific nutrient supplementation in patients with deficiencies is strong and should be considered in patients with chronic wounds. Adequate caloric intake and protein content should be recommended for most wound patients; however, excessive protein intake has not been beneficial in wound healing. The data behind specific amino acid and vitamin supplementation are limited at this time. As with other therapeutics, there is likely an appropriate dose for supplementation that has not yet been elucidated. Consideration of wound type, size, depth, exudate, and underlying cause are important to optimize healing and tailor nutritional supplementation to each patient. We hope future studies will illuminate the complementary role of dietary intake and nutrient supplementation for the treatment of chronic nonhealing wounds.
Dermatologists commonly manage a variety of wounds in the outpatient setting. Wound healing requires a multifaceted approach that often includes topical and oral therapies, adjustment of mechanical factors, and behavioral and lifestyle modifications. Physiologically, wound healing requires an inflammatory phase, a proliferative phase, and a remodeling phase. Chronic wounds undergo a prolonged inflammatory response hindered by decreased growth factors and increased wound bioburden.1 Malnutrition has been routinely associated with wound chronicity and serves as a modifiable risk factor that may improve wound healing outcomes.2
Although the causes of wounds encountered in dermatology vary extensively, the importance of nutrition underlies all wound healing. Caloric needs in wound healing have been estimated at 30 to 40 kcal/kg dependent on baseline body weight, age, medical comorbidities, activity level, stage of wound healing, wound size, and number of wounds.1,3,4 Nutritional supplementation is patient dependent, but this article serves to review the existing literature on macronutrient and micronutrient supplementation to clarify the potentially complementary role for nutritional support in chronic wounds. All patients should be screened with a thorough history, review of systems, and physical examination for existing nutrient deficiencies. Patients with age-related or chronic diseases are at increased risk for nutritional deficiency, and focused laboratory testing may be warranted. Supplementation for specific deficiencies with help from a registered dietician is recommended.
Macronutrients for Wound Healing
Protein—Protein is the most widely known macronutrient required for wound healing. The primary function of dietary protein is to provide amino acids to perform physiologic functions.5 Not only does cutaneous injury increase the metabolic needs of the wounded area, but large amounts of protein can be continually lost through wound exudates. Protein is necessary for the immune response required to transition from inflammatory to proliferative phases of wound healing.6 Protein energy deficiency has been reported to reduce fibroblast activity, delay angiogenesis, and decrease collagen formation.7 Additionally, protein is required for the formation of inflammatory cells and maintenance of oncotic pressure, specifically in venous insufficiency wounds.1
The current recommended dietary allowance for protein in healthy adults is 0.8 g/kg daily of body weight. In patients with pressure ulcerations, a goal recommended dietary allowance of 1.25 to 2.0 g/kg daily of body weight, dependent on ulceration size, has been recommended by the National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel.8 This recommendation was based on a series of studies that reported enhanced healing rates in patients with pressure ulcers receiving higher-protein diets.9 The largest study to date was double-blinded and included 89 residents of long-term care facilities with stage II to stage IV pressure ulcers.10 Participants were randomized to receive commercial protein supplementation vs placebo. At the end of 8 weeks, a statistically significant difference was seen in mean (SD) pressure ulcer scale for healing scores (3.55 [4.66] vs 3.22 [4.11]; P<.05).10 A 2014 Cochrane review failed to identify benefit associated with nutritional interventions for either the prevention and/or treatment of pressure ulcers.11 Specific recommendations on protein intake for other types of chronic wounds have not been proposed. Protein supplementation generally is provided orally, if tolerated. Liquid supplements such as Boost (Nestlé), Carnation Breakfast Essentials (Nestlé), NuBasics (SupremeMed), Resource (Nestlé Health Science), and Ensure (Abbott Laboratories) are frequently used to supplement both protein and caloric intake. Protein oversupplementation has not been associated with improved outcomes and may cause or exacerbate other medical comorbidities.
Fatty Acids for Wound Healing
Wound healing is an anabolic process that requires adequate intake of substrates such as glucose and fat. Carbohydrates serve as the major energy source required for wound healing, while fats are thought to play roles in cell membrane development and modulation of cellular signaling.1 Fats utilize a unique pathway for energy production through beta-oxidation and the production of adenosine triphosphate, allowing available protein to be harnessed for wound healing.1 Omega-3 and omega-6 fatty acids serve as precursors to prostaglandins, leukotrienes, and thromboxane—all key mediators of the inflammatory phase of wound healing.3 Omega-3 fatty acids are thought to downregulate genes involved in proinflammatory pathways,12 as well as to diminish lymphocyte proliferation and levels of IL-1β, tumor necrosis factor α, and IL-6 in vitro.13 In vivo studies assessing the impact of omega-3 fatty acid supplementation on wound healing are minimal, and the role of dietary supplementation for this indication remains unknown. Fish oil contains the omega-3 fatty acid–rich eicosapentaenoic acid and docosahexaenoic acid, which has been compared to mineral oil supplementation for wound healing in healthy adults. When fish oil was supplemented for 4 weeks, no significant differences were identified in time to complete wound healing between groups. Interestingly, significantly higher levels of the proinflammatory cytokine IL-1β were identified in blister fluid at 24 hours after blistering vs the placebo group (t=2.52, df=25, P<.05).14 Prior studies evaluating wound healing in animal models similarly identified longer times to re-epithelialization after omega-3 polyunsaturated fatty acid supplementation orally and topically.15,16 The fatty acid quality and composition consumed also may impact wound healing, as high-fat diets that are not rich in omega-3 fatty acids have been shown to promote inflammation and impair wound healing in rats, but this has not been thoroughly explored in human trials.17 Although adequate intake of these macronutrients is important, excessive intake may be harmful. Larger prospective trials are needed to shed light on the dose and composition of fatty acid supplementation that may optimize wound healing.
Vitamins and Micronutrients Required for Wound Healing
Vitamin A—Many vitamins serve as cofactors for the enzymatic processes required in wound healing. Vitamin A is an essential fat-soluble vitamin that serves a variety of dermatologic functions and promotes wound healing through stimulation of fibroblasts and ground substance, and it facilitates epithelial cell differentiation when applied topically.3,18 Vitamin A works through the activation of retinoid receptors on endothelial cells, fibroblasts, keratinocytes, melanocytes, and sebocytes, and has purported anti-inflammatory effects that aid the healing of open wounds.3 Additionally, vitamin A is thought to enhance cytokine release in the inflammatory phase of wound healing.19 Supplemental vitamin A has been associated with positive effects on acute wound healing, burns, and radiation injuries.3 The utility of vitamin A supplementation in chronic wounds remains unknown; however, it has been shown to be beneficial in patients with inflammatory disease, such as rheumatoid arthritis, on corticosteroid therapy. Vitamin A supplementation in this population has been shown to counteract the negative effects of corticosteroids on wound healing via downregulation of transforming growth factor β and insulinlike growth factor 1.20 Vitamin A deficiency has been associated with impaired progression through inflammatory and remodeling phases of healing due to altered B-cell and T-cell function and antibody production.1 Some experts recommend short courses of oral vitamin A supplementation to enhance wound healing at doses between 10,000 and 25,000 IU daily.2,3 Large, population-based studies are needed, and the safety supporting this recommendation in all patients remains unknown.
Vitamin C—Vitamin C is widely known for its role in collagen formation, immunomodulation, and antioxidant capacity.1 Although vitamin C deficiency is associated with decreased collagen synthesis and impaired wound healing,21 the utility of long-term supplementation in patients who are not deficient remains unexplored. A systematic review evaluating interventional studies utilizing vitamin C supplementation on pressure ulcerations and surgical wound healing concluded that convincing evidence exists only for supplementation with at least 500 mg of vitamin C. The authors noted, “There is little evidence for improved healing of surgical wounds by high-dose single vitamin C supplementation (1–3 g/day).”22 In a prospective, randomized, controlled trial, 20 patients with pressure ulcerations were supplemented with vitamin C vs placebo with a mean reduction in pressure-sore area of 84% after 1 month in the vitamin C–supplemented group compared to 42.7% in the placebo group (P<.005). A limitation of this study is the small population.23 One current recommendation for vitamin C supplementation in chronic wounds is for 500 mg daily in uncomplicated wounds to 2 g daily in severe wounds.3 Additional studies have suggested that the benefits of vitamin C supplementation are maximized when given in combination with zinc and arginine.22 At this time, evaluation for vitamin C deficiency and appropriate supplementation in patients with chronic wounds is needed.
Zinc—Minerals similarly play important roles in enzymatic regulation. Hundreds of zinc-containing enzymes are involved in wound healing and are required in tissue repair, growth, antioxidant capacity, and immune function.1,24 Zinc is specifically critical to collagen, DNA, RNA, and protein synthesis, as well as cellular proliferation.4 Zinc deficiency has been encountered in the setting of chronic wounds with extensive drainage, decreased dietary intake, or excessive gastrointestinal losses.25 Although many studies exist evaluating the utility of zinc supplementation on wound healing, many are confounded by multinutrient supplementation. No studies to date support zinc supplementation when zinc deficiency is absent. Patient assessment for medications or conditions that may impact zinc metabolism should be completed. Importantly, zinc supplementation can interfere with the absorption of other cations, so excessive supplementation should be avoided.1
Amino Acids for Wound Healing
Arginine—Arginine is an essential amino acid that serves as a substrate for cellular proliferation, collagen deposition, and lymphocyte function.8,26,27 Arginine serves as the biologic precursor for nitric oxide (NO), a substrate that has important wound healing properties. Nitric oxide metabolites have been shown to positively regulate wound repair while NO metabolites are reduced in wound environments in diabetic ulcerations.28,29 Arginine also is a proline precursor, an essential building block for collagen synthesis,6,30 and a stimulator of growth hormone and T cells.30,31 Animal studies have suggested L-arginine supplementation may reverse impaired NO synthesis in diabetic wounds.28 A single randomized trial assessing differing doses of arginine supplementation on stage II or stage IV pressure ulcers noted an almost two-fold improvement in healing time.32 However, human studies have not shown increased rates of re-epithelialization of skin graft donor sites when provided oral or parenteral arginine supplementation.33 Inadequate data currently exist to support regular arginine supplementation for all types of wounds, and no safe dose of daily arginine intake has been established.
Glutamine—Similarly, glutamine supplementation has been proposed to accelerate wound healing due to its role as a primary metabolic fuel source for rapidly proliferating cells such as epithelial cells and fibroblasts.8 Glutamine is thought to induce expression of heat-shock proteins and protect against inflammatory and infectious wound complications.34 Additionally, glutamine is thought to increase tissue insulin sensitivity, which may prove beneficial in wounds, as topical insulin previously has been shown in animal and human models to promote healing.35 Glutamine is thought to play a role in the inflammatory phase of wound healing via superoxide production, leukocyte apoptosis, and phagocytosis.6,34,36 Unfortunately, numerous randomized trials on glutamine supplementation have resulted in conflicting evidence confounded by multisupplementation within the same trial.37,38 A double-blind, randomized, controlled trial of 270 participants assessed the effect of oral supplementation with arginine, glutamine, or β-hydroxy-β-methylbutyrate vs control in the healing time of diabetic foot ulcerations. Significant differences in wound closure time at week 16 were only identified in participants with low albumin levels (≤40 g/L) who were supplemented (50.8%) vs the control group (34.9%; P=.0325) and in those with poor limb perfusion (ankle-brachial index of <1.0) who were supplemented (60.3%) vs the control group (39.3%; P=.0079).39 Ongoing clinical trials evaluating the effects of glutamine supplementation on differing wound types will hopefully shed light on the efficacy of supplementation.
Final Thoughts
Wound healing is multifactorial and should consider the health status and medical comorbidities of each patient treated. We propose an individualized approach to wound healing that includes exploration of specific macronutrient and micronutrient deficiencies, as malnutrition has been associated with wound chronicity and serves as a modifiable risk factor to improve healing.2 The evidence backing specific nutrient supplementation in patients with deficiencies is strong and should be considered in patients with chronic wounds. Adequate caloric intake and protein content should be recommended for most wound patients; however, excessive protein intake has not been beneficial in wound healing. The data behind specific amino acid and vitamin supplementation are limited at this time. As with other therapeutics, there is likely an appropriate dose for supplementation that has not yet been elucidated. Consideration of wound type, size, depth, exudate, and underlying cause are important to optimize healing and tailor nutritional supplementation to each patient. We hope future studies will illuminate the complementary role of dietary intake and nutrient supplementation for the treatment of chronic nonhealing wounds.
- Quain AM, Khardori NM. Nutrition in wound care management: a comprehensive overview. Wounds. 2015;27:327-335.
- Stechmiller JK. Understanding the role of nutrition and wound healing. Nutr Clin Pract. 2010;25:61-68. doi:10.1177/0884533609358997
- Molnar JA, Underdown MJ, Clark WA. Nutrition and chronic wounds. Adv Wound Care (New Rochelle). 2014;3:663-681. doi:10.1089/wound.2014.0530
- Dorner B, Posthauer ME, Thomas D; Panel NPUA. The role of nutrition in pressure ulcer prevention and treatment: National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care. 2009;22:212-221. doi:10.1097/01.ASW.0000350838.11854.0a
- Collins N. Protein and wound healing. Adv Skin Wound Care. 2001;14:288-289. doi:10.1097/00129334-200111000-00008
- Barchitta M, Maugeri A, Favara G, et al. Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin [published online March 5, 2019]. Int J Mol Sci. doi:10.3390/ijms20051119
- Harris CL, Fraser C. Malnutrition in the institutionalized elderly: the effects on wound healing. Ostomy Wound Manage. 2004;50:54-63.
- Saghaleini SH, Dehghan K, Shadvar K, et al. Pressure ulcer and nutrition. Indian J Crit Care Med. 2018;22:283-289. doi:10.4103/ijccm.IJCCM_277_17
- Breslow RA, Hallfrisch J, Guy DG, et al. The importance of dietary protein in healing pressure ulcers. J Am Geriatr Soc. 1993;41:357-362. doi:10.1111/j.1532-5415.1993.tb06940.x
- Lee SK, Posthauer ME, Dorner B, et al. Pressure ulcer healing with a concentrated, fortified, collagen protein hydrolysate supplement: a randomized controlled trial. Adv Skin Wound Care. 2006;19:92-96. doi:10.1097/00129334-200603000-00011
- Langer G, Fink A. Nutritional interventions for preventing and treating pressure ulcers. Cochrane Database Syst Rev. 2014;6:CD003216. doi:10.1002/14651858.CD003216.pub2
- Bouwens M, van de Rest O, Dellschaft N, et al. Fish-oil supplementation induces antiinflammatory gene expression profiles in human blood mononuclear cells. Am J Clin Nutr. 2009;90:415-424. doi:10.3945/ajcn.2009.27680
- Meydani SN, Endres S, Woods MM, et al. Oral (n-3) fatty acid supplementation suppresses cytokine production and lymphocyte proliferation: comparison between young and older women. J Nutr. 1991;121:547-555. doi:10.1093/jn/121.4.547
- McDaniel JC, Belury M, Ahijevych K, et al. Omega-3 fatty acids effect on wound healing. Wound Repair Regen. 2008;16:337-345. doi:10.1111/j.1524-475X.2008.00388.x
- Mooney MA, Vaughn DM, Reinhart GA, et al. Evaluation of the effects of omega-3 fatty acid-containing diets on the inflammatory stage of wound healing in dogs. Am J Vet Res. 1998;59:859-863.
- Cardoso CR, Souza MA, Ferro EA, et al. Influence of topical administration of n-3 and n-6 essential and n-9 nonessential fatty acids on the healing of cutaneous wounds. Wound Repair Regen. 2004;12:235-243. doi:10.1111/j.1067-1927.2004.012216.x
- Rosa DF, Sarandy MM, Novaes RD, et al. High-fat diet and alcohol intake promotes inflammation and impairs skin wound healing in Wistar rats. Mediators Inflamm. 2018;2018:4658583. doi:10.1155/2018/4658583
- Levenson SM, Gruber CA, Rettura G, et al. Supplemental vitamin A prevents the acute radiation-induced defect in wound healing. Ann Surg. 1984;200:494-512. doi:10.1097/00000658-198410000-00011
- Palmieri B, Vadalà M, Laurino C. Nutrition in wound healing: investigation of the molecular mechanisms, a narrative review. J Wound Care. 2019;28:683-693. doi:10.12968/jowc.2019.28.10.683
- Ehrlich HP, Hunt TK. Effects of cortisone and vitamin A on wound healing. Ann Surg. 1968;167:324-328. doi:10.1097/00000658-196803000-00004
- Pullar JM, Carr AC, Vissers MCM. The roles of vitamin C in skin health [published online August 12, 2017]. Nutrients. doi:10.3390/nu9080866
- Ellinger S, Stehle P. Efficacy of vitamin supplementation in situations with wound healing disorders: results from clinical intervention studies. Curr Opin Clin Nutr Metab Care. 2009;12:588-595. doi:10.1097/MCO.0b013e328331a5b5
- Taylor TV, Rimmer S, Day B, et al. Ascorbic acid supplementation in the treatment of pressure-sores. Lancet. 1974;2:544-546. doi:10.1016/s0140-6736(74)91874-1
- Ibs KH, Rink L. Zinc-altered immune function. J Nutr. 2003;133(5 suppl 1):1452S-1456S. doi:10.1093/jn/133.5.1452S
- Hoffman M, Micheletti RG, Shields BE. Nutritional dermatoses in the hospitalized patient. Cutis. 2020;105:296-302, 308, E1-E5.
- Chow O, Barbul A. Immunonutrition: role in wound healing and tissue regeneration. Adv Wound Care (New Rochelle). 2014;3:46-53. doi:10.1089/wound.2012.0415
- Singh K, Coburn LA, Barry DP, et al. L-arginine uptake by cationic amino acid transporter 2 is essential for colonic epithelial cell restitution. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1061-G1073. doi:10.1152/ajpgi.00544.2011
- Witte MB, Thornton FJ, Tantry U, et al. L-Arginine supplementation enhances diabetic wound healing: involvement of the nitric oxide synthase and arginase pathways. Metabolism. 2002;51:1269-1273. doi:10.1053/meta.2002.35185
- Witte MB, Barbul A. Role of nitric oxide in wound repair. Am J Surg. 2002;183:406-412. doi:10.1016/s0002-9610(02)00815-2
- Barbul A. Proline precursors to sustain Mammalian collagen synthesis. J Nutr. 2008;138:2021S-2024S. doi:10.1093/jn/138.10.2021S
- Wu G, Bazer FW, Davis TA, et al. Arginine metabolism and nutrition in growth, health and disease. Amino Acids. 2009;37:153-168. doi:10.1007/s00726-008-0210-y
- Leigh B, Desneves K, Rafferty J, et al. The effect of different doses of an arginine-containing supplement on the healing of pressure ulcers. J Wound Care. 2012;21:150-156. doi:10.12968/jowc.2012.21.3.150
- Debats IB, Koeneman MM, Booi DI, et al. Intravenous arginine and human skin graft donor site healing: a randomized controlled trial. Burns. 2011;37:420-426. doi:10.1016/j.burns.2010.06.003
- Wischmeyer PE. Glutamine and heat shock protein expression. Nutrition. 2002;18:225-228. doi:10.1016/s0899-9007(01)00796-1
- Wang J, Xu J. Effects of topical insulin on wound healing: a review of animal and human evidences. Diabetes Metab Syndr Obes. 2020;13:719-727. doi:10.2147/DMSO.S237294
- Newsholme P. Why is L-glutamine metabolism important to cells of the immune system in health, postinjury, surgery or infection?J Nutr. 2001;131(9 suppl):2515S-2522S; discussion 2523S-2524S. doi:10.1093/jn/131.9.2515S
- Aquino VM, Harvey AR, Garvin JH, et al. A double-blind randomized placebo-controlled study of oral glutamine in the prevention of mucositis in children undergoing hematopoietic stem cell transplantation: a pediatric blood and marrow transplant consortium study. Bone Marrow Transplant. 2005;36:611-616. doi:10.1038/sj.bmt.1705084
- Ward E, Smith M, Henderson M, et al. The effect of high-dose enteral glutamine on the incidence and severity of mucositis in paediatric oncology patients. Eur J Clin Nutr. 2009;63:134-140. doi:10.1038/sj.ejcn.1602894
- Armstrong DG, Hanft JR, Driver VR, et al. Effect of oral nutritional supplementation on wound healing in diabetic foot ulcers: a prospective randomized controlled trial. Diabet Med. 2014;31:1069-1077. doi:10.1111/dme.12509
- Quain AM, Khardori NM. Nutrition in wound care management: a comprehensive overview. Wounds. 2015;27:327-335.
- Stechmiller JK. Understanding the role of nutrition and wound healing. Nutr Clin Pract. 2010;25:61-68. doi:10.1177/0884533609358997
- Molnar JA, Underdown MJ, Clark WA. Nutrition and chronic wounds. Adv Wound Care (New Rochelle). 2014;3:663-681. doi:10.1089/wound.2014.0530
- Dorner B, Posthauer ME, Thomas D; Panel NPUA. The role of nutrition in pressure ulcer prevention and treatment: National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care. 2009;22:212-221. doi:10.1097/01.ASW.0000350838.11854.0a
- Collins N. Protein and wound healing. Adv Skin Wound Care. 2001;14:288-289. doi:10.1097/00129334-200111000-00008
- Barchitta M, Maugeri A, Favara G, et al. Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin [published online March 5, 2019]. Int J Mol Sci. doi:10.3390/ijms20051119
- Harris CL, Fraser C. Malnutrition in the institutionalized elderly: the effects on wound healing. Ostomy Wound Manage. 2004;50:54-63.
- Saghaleini SH, Dehghan K, Shadvar K, et al. Pressure ulcer and nutrition. Indian J Crit Care Med. 2018;22:283-289. doi:10.4103/ijccm.IJCCM_277_17
- Breslow RA, Hallfrisch J, Guy DG, et al. The importance of dietary protein in healing pressure ulcers. J Am Geriatr Soc. 1993;41:357-362. doi:10.1111/j.1532-5415.1993.tb06940.x
- Lee SK, Posthauer ME, Dorner B, et al. Pressure ulcer healing with a concentrated, fortified, collagen protein hydrolysate supplement: a randomized controlled trial. Adv Skin Wound Care. 2006;19:92-96. doi:10.1097/00129334-200603000-00011
- Langer G, Fink A. Nutritional interventions for preventing and treating pressure ulcers. Cochrane Database Syst Rev. 2014;6:CD003216. doi:10.1002/14651858.CD003216.pub2
- Bouwens M, van de Rest O, Dellschaft N, et al. Fish-oil supplementation induces antiinflammatory gene expression profiles in human blood mononuclear cells. Am J Clin Nutr. 2009;90:415-424. doi:10.3945/ajcn.2009.27680
- Meydani SN, Endres S, Woods MM, et al. Oral (n-3) fatty acid supplementation suppresses cytokine production and lymphocyte proliferation: comparison between young and older women. J Nutr. 1991;121:547-555. doi:10.1093/jn/121.4.547
- McDaniel JC, Belury M, Ahijevych K, et al. Omega-3 fatty acids effect on wound healing. Wound Repair Regen. 2008;16:337-345. doi:10.1111/j.1524-475X.2008.00388.x
- Mooney MA, Vaughn DM, Reinhart GA, et al. Evaluation of the effects of omega-3 fatty acid-containing diets on the inflammatory stage of wound healing in dogs. Am J Vet Res. 1998;59:859-863.
- Cardoso CR, Souza MA, Ferro EA, et al. Influence of topical administration of n-3 and n-6 essential and n-9 nonessential fatty acids on the healing of cutaneous wounds. Wound Repair Regen. 2004;12:235-243. doi:10.1111/j.1067-1927.2004.012216.x
- Rosa DF, Sarandy MM, Novaes RD, et al. High-fat diet and alcohol intake promotes inflammation and impairs skin wound healing in Wistar rats. Mediators Inflamm. 2018;2018:4658583. doi:10.1155/2018/4658583
- Levenson SM, Gruber CA, Rettura G, et al. Supplemental vitamin A prevents the acute radiation-induced defect in wound healing. Ann Surg. 1984;200:494-512. doi:10.1097/00000658-198410000-00011
- Palmieri B, Vadalà M, Laurino C. Nutrition in wound healing: investigation of the molecular mechanisms, a narrative review. J Wound Care. 2019;28:683-693. doi:10.12968/jowc.2019.28.10.683
- Ehrlich HP, Hunt TK. Effects of cortisone and vitamin A on wound healing. Ann Surg. 1968;167:324-328. doi:10.1097/00000658-196803000-00004
- Pullar JM, Carr AC, Vissers MCM. The roles of vitamin C in skin health [published online August 12, 2017]. Nutrients. doi:10.3390/nu9080866
- Ellinger S, Stehle P. Efficacy of vitamin supplementation in situations with wound healing disorders: results from clinical intervention studies. Curr Opin Clin Nutr Metab Care. 2009;12:588-595. doi:10.1097/MCO.0b013e328331a5b5
- Taylor TV, Rimmer S, Day B, et al. Ascorbic acid supplementation in the treatment of pressure-sores. Lancet. 1974;2:544-546. doi:10.1016/s0140-6736(74)91874-1
- Ibs KH, Rink L. Zinc-altered immune function. J Nutr. 2003;133(5 suppl 1):1452S-1456S. doi:10.1093/jn/133.5.1452S
- Hoffman M, Micheletti RG, Shields BE. Nutritional dermatoses in the hospitalized patient. Cutis. 2020;105:296-302, 308, E1-E5.
- Chow O, Barbul A. Immunonutrition: role in wound healing and tissue regeneration. Adv Wound Care (New Rochelle). 2014;3:46-53. doi:10.1089/wound.2012.0415
- Singh K, Coburn LA, Barry DP, et al. L-arginine uptake by cationic amino acid transporter 2 is essential for colonic epithelial cell restitution. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1061-G1073. doi:10.1152/ajpgi.00544.2011
- Witte MB, Thornton FJ, Tantry U, et al. L-Arginine supplementation enhances diabetic wound healing: involvement of the nitric oxide synthase and arginase pathways. Metabolism. 2002;51:1269-1273. doi:10.1053/meta.2002.35185
- Witte MB, Barbul A. Role of nitric oxide in wound repair. Am J Surg. 2002;183:406-412. doi:10.1016/s0002-9610(02)00815-2
- Barbul A. Proline precursors to sustain Mammalian collagen synthesis. J Nutr. 2008;138:2021S-2024S. doi:10.1093/jn/138.10.2021S
- Wu G, Bazer FW, Davis TA, et al. Arginine metabolism and nutrition in growth, health and disease. Amino Acids. 2009;37:153-168. doi:10.1007/s00726-008-0210-y
- Leigh B, Desneves K, Rafferty J, et al. The effect of different doses of an arginine-containing supplement on the healing of pressure ulcers. J Wound Care. 2012;21:150-156. doi:10.12968/jowc.2012.21.3.150
- Debats IB, Koeneman MM, Booi DI, et al. Intravenous arginine and human skin graft donor site healing: a randomized controlled trial. Burns. 2011;37:420-426. doi:10.1016/j.burns.2010.06.003
- Wischmeyer PE. Glutamine and heat shock protein expression. Nutrition. 2002;18:225-228. doi:10.1016/s0899-9007(01)00796-1
- Wang J, Xu J. Effects of topical insulin on wound healing: a review of animal and human evidences. Diabetes Metab Syndr Obes. 2020;13:719-727. doi:10.2147/DMSO.S237294
- Newsholme P. Why is L-glutamine metabolism important to cells of the immune system in health, postinjury, surgery or infection?J Nutr. 2001;131(9 suppl):2515S-2522S; discussion 2523S-2524S. doi:10.1093/jn/131.9.2515S
- Aquino VM, Harvey AR, Garvin JH, et al. A double-blind randomized placebo-controlled study of oral glutamine in the prevention of mucositis in children undergoing hematopoietic stem cell transplantation: a pediatric blood and marrow transplant consortium study. Bone Marrow Transplant. 2005;36:611-616. doi:10.1038/sj.bmt.1705084
- Ward E, Smith M, Henderson M, et al. The effect of high-dose enteral glutamine on the incidence and severity of mucositis in paediatric oncology patients. Eur J Clin Nutr. 2009;63:134-140. doi:10.1038/sj.ejcn.1602894
- Armstrong DG, Hanft JR, Driver VR, et al. Effect of oral nutritional supplementation on wound healing in diabetic foot ulcers: a prospective randomized controlled trial. Diabet Med. 2014;31:1069-1077. doi:10.1111/dme.12509
Practice Points
- Optimizing wound healing requires local and systemic therapies as well as adequate nutritional support.
- Malnutrition is a potentially modifiable risk factor that may contribute to impaired wound healing.
- Patients with chronic wounds and specific nutrient deficiencies should supplement to optimize healing.
Large Leg Ulcers After Swimming in the Ocean
The Diagnosis: Vibrio vulnificus Infection
At the initial presentation, the differential diagnosis included infectious processes such as bacterial or angioinvasive fungal infections or an inflammatory process such as pyoderma gangrenosum. Blood cultures were found to be positive for pansensitive Vibrio vulnificus. He initially was treated with piperacillin-tazobactam and received surgical debridement of the affected tissues. Pathologic interpretation of the wound tissues revealed a diagnosis of necrotizing softtissue infection and positive Candida albicans growth. He received topical bacitracin on discharge as well as a 7-day course of amoxicillin-clavulanate and fluconazole. He continued to receive debridement procedures and skin grafts, followed by topical mupirocin treatment and silver sulfadiazine. He was seen 6 weeks after discharge with healing wounds and healthy-appearing granulation tissue at the base.
Our patient’s presentation of retiform purpura with stellate necrosis was consistent with a wide range of serious pathologies ranging from medium-vessel vasculitis to thromboembolic phenomena and angioinvasive fungal infections.1 Although Vibrio infection rarely is the first explanation that comes to mind when observing necrotic retiform purpura, the chronic nonhealing injury on the leg combined with the recent history of ocean swimming made V vulnificus stand out as a likely culprit. Although V vulnificus infection traditionally presents with cellulitis, edema, and hemorrhagic bulla,2 necrosis also has been observed.3 Vibrio vulnificus produces multiple virulence factors, and it is believed that these severe cutaneous symptoms are attributable to the production of a specific metalloprotease that enhances vascular permeability, thereby inducing hemorrhage within the vascular basement membrane zone.2
Vibrio vulnificus is an opportunistic bacterial pathogen associated with consumption of contaminated seafood or swimming in ocean waters with open wounds. Infections are rare, with only approximately 100 cases reported annually in the United States.4 However, V vulnificus infections have demonstrated increasing incidence in recent years, especially infections of pre-existing wounds.4,5 Risk factors for their development include age over 40 years and underlying conditions including liver disease, diabetes mellitus, and immune dysfunction.4 Vibrio vulnificus infections also demonstrate a strong male predilection, with almost 90% of infections occurring in males.4 Although the precise etiology of this sex discrepancy remains unknown, estrogen has been suggested to be a protective factor.6 Alternatively, behavioral differences also have been proposed as possible explanations for this discrepancy, with women less likely to consume seafood or go swimming. However, epidemiologic data reveal strong correlations between male sex and liver cirrhosis, a primary risk factor for V vulnificus infections, suggesting that male sex may simply be a confounding variable.7
Infections with V vulnificus are notable for their short incubation periods, with onset of symptoms occurring within 24 hours of exposure, making prompt diagnosis and treatment of high importance.8 Although rare, V vulnificus infections are associated with high mortality rates. From 1988 to 2010, nearly 600 deaths were reported secondary to V vulnificus infections.4 Wound infections carry a 17.6% fatality rate,4 while bloodborne V vulnificus infections exceed 50% fatality.8 Although sepsis secondary to V vulnificus usually is caused by ingestion of raw or undercooked shellfish, primarily oysters,4 our case highlights a rarer instance of both sepsis and localized infection stemming from ocean water exposure.
Vibrio vulnificus is an obligate halophile and therefore is found in marine environments rather than freshwater bodies. However, it rarely is isolated from bodies of water with salinities over 25 parts per thousand, such as the Mediterranean Sea; it usually is found in warmer waters, making it more common in the summer months from May to October.4 Given this proclivity for warmer environments, climate change has contributed to both a greater incidence and global distribution of V vulnificus. 9,10
Treatment of V vulnificus infections centers on antibiotic treatment, with Vibrio species generally demonstrating susceptibility to most antibiotics of human significance.11 However, some Vibrio isolates within the United States have demonstrated antibiotic resistance; 45% of a variety of clinical and environmental samples from South Carolina and Georgia demonstrated resistance to at least 3 antibiotic classes, and 17.3% resisted 8 or more classes of antibiotics.12 These included medications such as doxycycline, tetracycline, aminoglycosides, and cephalosporins—agents that normally are prescribed for V vulnificus infections. Although tetracyclines have long been touted as the preferred treatment of V vulnificus infections, the spread of antibiotic resistance may require greater reliance on alternative regimens such as combinations of cephalosporins and doxycycline or a single fluoroquinolone.13 Although rare, Vibrio infections can have rapidly fatal consequences and should be given serious consideration when evaluating patients with relevant risk factors.
The differential diagnosis included angioinvasive mucormycosis, calciphylaxis, pyoderma gangrenosum, and Stevens-Johnson syndrome/toxic epidermal necrolysis. Mucormycosis is a fungal infection caused by Mucorales fungi that most commonly is seen in patients with diabetes mellitus, hematologic malignancies, neutropenia, and immunocompromise.14 Calciphylaxis is a condition involving microvascular occlusion due to diffuse calcium deposition in cutaneous blood vessels. It typically presents as violaceous retiform patches and plaques commonly seen on areas such as the thighs, buttocks, or abdomen and usually is associated with chronic renal failure, hemodialysis, and/or secondary hyperparathyroidism.15 Pyoderma gangrenosum is an inflammatory condition involving neutrophilic ulceration of the skin that typically presents as ulceration with a classically undermined border. It frequently is considered a diagnosis of exclusion and therefore requires that providers rule out other causes of ulceration prior to diagnosis.16 Stevens-Johnson syndrome/toxic epidermal necrolysis is a rare drug reaction involving mucosal erosions and cutaneous detachment.17 This diagnosis is less likely given that our patient lacked mucosal involvement and did not have any notable medication exposures prior to symptom onset.
- Wysong A, Venkatesan P. An approach to the patient with retiform purpura. Dermatol Ther. 2011;24:151-172. doi:10.1111/j .1529-8019.2011.01392.x
- Miyoshi S-I. Vibrio vulnificus infection and metalloprotease. J Dermatol. 2006;33:589-595. doi:10.1111/j.1346-8138.2006.00139.x
- Patel VJ, Gardner E, Burton CS. Vibrio vulnificus septicemia and leg ulcer. J Am Acad Dermatol. 2002;46(5 suppl):S144-S145. doi:10.1067 /mjd.2002.107778
- Baker-Austin C, Oliver JD. Vibrio vulnificus: new insights into a deadly opportunistic pathogen. Environ Microbiol. 2018;20:423-430. doi:10.1111/1462-2920.13955
- Preliminary FoodNet data on the incidence of infection with pathogens transmitted commonly through food —10 states, 2009. CDC website. Published April 16, 2010. Accessed November 3, 2021. https://www.cdc .gov/mmwr/preview/mmwrhtml/mm5914a2.htm
- Merkel SM, Alexander S, Zufall E, et al. Essential role for estrogen in protection against Vibrio vulnificus-induced endotoxic shock. Infect Immun. 2001;69:6119-6122. doi:10.1128/IAI.69.10.6119 -6122.2001
- Scaglione S, Kliethermes S, Cao G, et al. The epidemiology of cirrhosis in the United States: a population-based study. J Clin Gastroenterol. 2015;49:690-696. doi:10.1097/MCG.0000000000000208
- Jones M, Oliver J. Vibrio vulnificus: disease and pathogenesis [published online December 20, 2020]. Infect Immun. https://doi.org/10.1128 /IAI.01046-08
- Paz S, Bisharat N, Paz E, et al. Climate change and the emergence of Vibrio vulnificus disease in Israel. Environ Res. 2007;103:390-396. doi:10.1016/j.envres.2006.07.002
- Martinez-Urtaza J, Bowers JC, Trinanes J, et al. Climate anomalies and the increasing risk of Vibrio parahaemolyticus and Vibrio vulnificus illnesses. Food Res Int. 2010;43:1780-1790. doi:10.1016/j. foodres.2010.04.001
- Oliver JD. Vibrio vulnificus. In: Thompson FL, Austin B, Swings J, eds. The Biology of Vibrios. ASM Press; 2006:349-366.
- Baker-Austin C, McArthur JV, Lindell AH, et al. Multi-site analysis reveals widespread antibiotic resistance in the marine pathogen Vibrio vulnificus. Microb Ecol. 2009;57:151-159. doi:10.1007 /s00248-008-9413-8
- Elmahdi S, DaSilva LV, Parveen S. Antibiotic resistance of Vibrio parahaemolyticus and Vibrio vulnificus in various countries: a review. Food Microbiol. 2016;57:128-134. doi:10.1016/j.fm.2016.02.008
- Prasad P, Wong V, Burgin S, et al. Mucormycosis. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/mucormycosis?diagnosisId=51981 &moduleId=101
- Blum A, Song P, Tan B, et al. Calciphylaxis. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/calciphylaxis?diagnosisId=51241&moduleId=101
- Cohen J, Wong V, Burgin S. Pyoderma gangrenosum. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/pyoderma+gangrenosum?diagnosis Id=52242&moduleId=101
- Walls A, Burgin S. Stevens-Johnson syndrome. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/stevens-johnson+syndrome?diagnosisId=52342&moduleId=101
The Diagnosis: Vibrio vulnificus Infection
At the initial presentation, the differential diagnosis included infectious processes such as bacterial or angioinvasive fungal infections or an inflammatory process such as pyoderma gangrenosum. Blood cultures were found to be positive for pansensitive Vibrio vulnificus. He initially was treated with piperacillin-tazobactam and received surgical debridement of the affected tissues. Pathologic interpretation of the wound tissues revealed a diagnosis of necrotizing softtissue infection and positive Candida albicans growth. He received topical bacitracin on discharge as well as a 7-day course of amoxicillin-clavulanate and fluconazole. He continued to receive debridement procedures and skin grafts, followed by topical mupirocin treatment and silver sulfadiazine. He was seen 6 weeks after discharge with healing wounds and healthy-appearing granulation tissue at the base.
Our patient’s presentation of retiform purpura with stellate necrosis was consistent with a wide range of serious pathologies ranging from medium-vessel vasculitis to thromboembolic phenomena and angioinvasive fungal infections.1 Although Vibrio infection rarely is the first explanation that comes to mind when observing necrotic retiform purpura, the chronic nonhealing injury on the leg combined with the recent history of ocean swimming made V vulnificus stand out as a likely culprit. Although V vulnificus infection traditionally presents with cellulitis, edema, and hemorrhagic bulla,2 necrosis also has been observed.3 Vibrio vulnificus produces multiple virulence factors, and it is believed that these severe cutaneous symptoms are attributable to the production of a specific metalloprotease that enhances vascular permeability, thereby inducing hemorrhage within the vascular basement membrane zone.2
Vibrio vulnificus is an opportunistic bacterial pathogen associated with consumption of contaminated seafood or swimming in ocean waters with open wounds. Infections are rare, with only approximately 100 cases reported annually in the United States.4 However, V vulnificus infections have demonstrated increasing incidence in recent years, especially infections of pre-existing wounds.4,5 Risk factors for their development include age over 40 years and underlying conditions including liver disease, diabetes mellitus, and immune dysfunction.4 Vibrio vulnificus infections also demonstrate a strong male predilection, with almost 90% of infections occurring in males.4 Although the precise etiology of this sex discrepancy remains unknown, estrogen has been suggested to be a protective factor.6 Alternatively, behavioral differences also have been proposed as possible explanations for this discrepancy, with women less likely to consume seafood or go swimming. However, epidemiologic data reveal strong correlations between male sex and liver cirrhosis, a primary risk factor for V vulnificus infections, suggesting that male sex may simply be a confounding variable.7
Infections with V vulnificus are notable for their short incubation periods, with onset of symptoms occurring within 24 hours of exposure, making prompt diagnosis and treatment of high importance.8 Although rare, V vulnificus infections are associated with high mortality rates. From 1988 to 2010, nearly 600 deaths were reported secondary to V vulnificus infections.4 Wound infections carry a 17.6% fatality rate,4 while bloodborne V vulnificus infections exceed 50% fatality.8 Although sepsis secondary to V vulnificus usually is caused by ingestion of raw or undercooked shellfish, primarily oysters,4 our case highlights a rarer instance of both sepsis and localized infection stemming from ocean water exposure.
Vibrio vulnificus is an obligate halophile and therefore is found in marine environments rather than freshwater bodies. However, it rarely is isolated from bodies of water with salinities over 25 parts per thousand, such as the Mediterranean Sea; it usually is found in warmer waters, making it more common in the summer months from May to October.4 Given this proclivity for warmer environments, climate change has contributed to both a greater incidence and global distribution of V vulnificus. 9,10
Treatment of V vulnificus infections centers on antibiotic treatment, with Vibrio species generally demonstrating susceptibility to most antibiotics of human significance.11 However, some Vibrio isolates within the United States have demonstrated antibiotic resistance; 45% of a variety of clinical and environmental samples from South Carolina and Georgia demonstrated resistance to at least 3 antibiotic classes, and 17.3% resisted 8 or more classes of antibiotics.12 These included medications such as doxycycline, tetracycline, aminoglycosides, and cephalosporins—agents that normally are prescribed for V vulnificus infections. Although tetracyclines have long been touted as the preferred treatment of V vulnificus infections, the spread of antibiotic resistance may require greater reliance on alternative regimens such as combinations of cephalosporins and doxycycline or a single fluoroquinolone.13 Although rare, Vibrio infections can have rapidly fatal consequences and should be given serious consideration when evaluating patients with relevant risk factors.
The differential diagnosis included angioinvasive mucormycosis, calciphylaxis, pyoderma gangrenosum, and Stevens-Johnson syndrome/toxic epidermal necrolysis. Mucormycosis is a fungal infection caused by Mucorales fungi that most commonly is seen in patients with diabetes mellitus, hematologic malignancies, neutropenia, and immunocompromise.14 Calciphylaxis is a condition involving microvascular occlusion due to diffuse calcium deposition in cutaneous blood vessels. It typically presents as violaceous retiform patches and plaques commonly seen on areas such as the thighs, buttocks, or abdomen and usually is associated with chronic renal failure, hemodialysis, and/or secondary hyperparathyroidism.15 Pyoderma gangrenosum is an inflammatory condition involving neutrophilic ulceration of the skin that typically presents as ulceration with a classically undermined border. It frequently is considered a diagnosis of exclusion and therefore requires that providers rule out other causes of ulceration prior to diagnosis.16 Stevens-Johnson syndrome/toxic epidermal necrolysis is a rare drug reaction involving mucosal erosions and cutaneous detachment.17 This diagnosis is less likely given that our patient lacked mucosal involvement and did not have any notable medication exposures prior to symptom onset.
The Diagnosis: Vibrio vulnificus Infection
At the initial presentation, the differential diagnosis included infectious processes such as bacterial or angioinvasive fungal infections or an inflammatory process such as pyoderma gangrenosum. Blood cultures were found to be positive for pansensitive Vibrio vulnificus. He initially was treated with piperacillin-tazobactam and received surgical debridement of the affected tissues. Pathologic interpretation of the wound tissues revealed a diagnosis of necrotizing softtissue infection and positive Candida albicans growth. He received topical bacitracin on discharge as well as a 7-day course of amoxicillin-clavulanate and fluconazole. He continued to receive debridement procedures and skin grafts, followed by topical mupirocin treatment and silver sulfadiazine. He was seen 6 weeks after discharge with healing wounds and healthy-appearing granulation tissue at the base.
Our patient’s presentation of retiform purpura with stellate necrosis was consistent with a wide range of serious pathologies ranging from medium-vessel vasculitis to thromboembolic phenomena and angioinvasive fungal infections.1 Although Vibrio infection rarely is the first explanation that comes to mind when observing necrotic retiform purpura, the chronic nonhealing injury on the leg combined with the recent history of ocean swimming made V vulnificus stand out as a likely culprit. Although V vulnificus infection traditionally presents with cellulitis, edema, and hemorrhagic bulla,2 necrosis also has been observed.3 Vibrio vulnificus produces multiple virulence factors, and it is believed that these severe cutaneous symptoms are attributable to the production of a specific metalloprotease that enhances vascular permeability, thereby inducing hemorrhage within the vascular basement membrane zone.2
Vibrio vulnificus is an opportunistic bacterial pathogen associated with consumption of contaminated seafood or swimming in ocean waters with open wounds. Infections are rare, with only approximately 100 cases reported annually in the United States.4 However, V vulnificus infections have demonstrated increasing incidence in recent years, especially infections of pre-existing wounds.4,5 Risk factors for their development include age over 40 years and underlying conditions including liver disease, diabetes mellitus, and immune dysfunction.4 Vibrio vulnificus infections also demonstrate a strong male predilection, with almost 90% of infections occurring in males.4 Although the precise etiology of this sex discrepancy remains unknown, estrogen has been suggested to be a protective factor.6 Alternatively, behavioral differences also have been proposed as possible explanations for this discrepancy, with women less likely to consume seafood or go swimming. However, epidemiologic data reveal strong correlations between male sex and liver cirrhosis, a primary risk factor for V vulnificus infections, suggesting that male sex may simply be a confounding variable.7
Infections with V vulnificus are notable for their short incubation periods, with onset of symptoms occurring within 24 hours of exposure, making prompt diagnosis and treatment of high importance.8 Although rare, V vulnificus infections are associated with high mortality rates. From 1988 to 2010, nearly 600 deaths were reported secondary to V vulnificus infections.4 Wound infections carry a 17.6% fatality rate,4 while bloodborne V vulnificus infections exceed 50% fatality.8 Although sepsis secondary to V vulnificus usually is caused by ingestion of raw or undercooked shellfish, primarily oysters,4 our case highlights a rarer instance of both sepsis and localized infection stemming from ocean water exposure.
Vibrio vulnificus is an obligate halophile and therefore is found in marine environments rather than freshwater bodies. However, it rarely is isolated from bodies of water with salinities over 25 parts per thousand, such as the Mediterranean Sea; it usually is found in warmer waters, making it more common in the summer months from May to October.4 Given this proclivity for warmer environments, climate change has contributed to both a greater incidence and global distribution of V vulnificus. 9,10
Treatment of V vulnificus infections centers on antibiotic treatment, with Vibrio species generally demonstrating susceptibility to most antibiotics of human significance.11 However, some Vibrio isolates within the United States have demonstrated antibiotic resistance; 45% of a variety of clinical and environmental samples from South Carolina and Georgia demonstrated resistance to at least 3 antibiotic classes, and 17.3% resisted 8 or more classes of antibiotics.12 These included medications such as doxycycline, tetracycline, aminoglycosides, and cephalosporins—agents that normally are prescribed for V vulnificus infections. Although tetracyclines have long been touted as the preferred treatment of V vulnificus infections, the spread of antibiotic resistance may require greater reliance on alternative regimens such as combinations of cephalosporins and doxycycline or a single fluoroquinolone.13 Although rare, Vibrio infections can have rapidly fatal consequences and should be given serious consideration when evaluating patients with relevant risk factors.
The differential diagnosis included angioinvasive mucormycosis, calciphylaxis, pyoderma gangrenosum, and Stevens-Johnson syndrome/toxic epidermal necrolysis. Mucormycosis is a fungal infection caused by Mucorales fungi that most commonly is seen in patients with diabetes mellitus, hematologic malignancies, neutropenia, and immunocompromise.14 Calciphylaxis is a condition involving microvascular occlusion due to diffuse calcium deposition in cutaneous blood vessels. It typically presents as violaceous retiform patches and plaques commonly seen on areas such as the thighs, buttocks, or abdomen and usually is associated with chronic renal failure, hemodialysis, and/or secondary hyperparathyroidism.15 Pyoderma gangrenosum is an inflammatory condition involving neutrophilic ulceration of the skin that typically presents as ulceration with a classically undermined border. It frequently is considered a diagnosis of exclusion and therefore requires that providers rule out other causes of ulceration prior to diagnosis.16 Stevens-Johnson syndrome/toxic epidermal necrolysis is a rare drug reaction involving mucosal erosions and cutaneous detachment.17 This diagnosis is less likely given that our patient lacked mucosal involvement and did not have any notable medication exposures prior to symptom onset.
- Wysong A, Venkatesan P. An approach to the patient with retiform purpura. Dermatol Ther. 2011;24:151-172. doi:10.1111/j .1529-8019.2011.01392.x
- Miyoshi S-I. Vibrio vulnificus infection and metalloprotease. J Dermatol. 2006;33:589-595. doi:10.1111/j.1346-8138.2006.00139.x
- Patel VJ, Gardner E, Burton CS. Vibrio vulnificus septicemia and leg ulcer. J Am Acad Dermatol. 2002;46(5 suppl):S144-S145. doi:10.1067 /mjd.2002.107778
- Baker-Austin C, Oliver JD. Vibrio vulnificus: new insights into a deadly opportunistic pathogen. Environ Microbiol. 2018;20:423-430. doi:10.1111/1462-2920.13955
- Preliminary FoodNet data on the incidence of infection with pathogens transmitted commonly through food —10 states, 2009. CDC website. Published April 16, 2010. Accessed November 3, 2021. https://www.cdc .gov/mmwr/preview/mmwrhtml/mm5914a2.htm
- Merkel SM, Alexander S, Zufall E, et al. Essential role for estrogen in protection against Vibrio vulnificus-induced endotoxic shock. Infect Immun. 2001;69:6119-6122. doi:10.1128/IAI.69.10.6119 -6122.2001
- Scaglione S, Kliethermes S, Cao G, et al. The epidemiology of cirrhosis in the United States: a population-based study. J Clin Gastroenterol. 2015;49:690-696. doi:10.1097/MCG.0000000000000208
- Jones M, Oliver J. Vibrio vulnificus: disease and pathogenesis [published online December 20, 2020]. Infect Immun. https://doi.org/10.1128 /IAI.01046-08
- Paz S, Bisharat N, Paz E, et al. Climate change and the emergence of Vibrio vulnificus disease in Israel. Environ Res. 2007;103:390-396. doi:10.1016/j.envres.2006.07.002
- Martinez-Urtaza J, Bowers JC, Trinanes J, et al. Climate anomalies and the increasing risk of Vibrio parahaemolyticus and Vibrio vulnificus illnesses. Food Res Int. 2010;43:1780-1790. doi:10.1016/j. foodres.2010.04.001
- Oliver JD. Vibrio vulnificus. In: Thompson FL, Austin B, Swings J, eds. The Biology of Vibrios. ASM Press; 2006:349-366.
- Baker-Austin C, McArthur JV, Lindell AH, et al. Multi-site analysis reveals widespread antibiotic resistance in the marine pathogen Vibrio vulnificus. Microb Ecol. 2009;57:151-159. doi:10.1007 /s00248-008-9413-8
- Elmahdi S, DaSilva LV, Parveen S. Antibiotic resistance of Vibrio parahaemolyticus and Vibrio vulnificus in various countries: a review. Food Microbiol. 2016;57:128-134. doi:10.1016/j.fm.2016.02.008
- Prasad P, Wong V, Burgin S, et al. Mucormycosis. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/mucormycosis?diagnosisId=51981 &moduleId=101
- Blum A, Song P, Tan B, et al. Calciphylaxis. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/calciphylaxis?diagnosisId=51241&moduleId=101
- Cohen J, Wong V, Burgin S. Pyoderma gangrenosum. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/pyoderma+gangrenosum?diagnosis Id=52242&moduleId=101
- Walls A, Burgin S. Stevens-Johnson syndrome. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/stevens-johnson+syndrome?diagnosisId=52342&moduleId=101
- Wysong A, Venkatesan P. An approach to the patient with retiform purpura. Dermatol Ther. 2011;24:151-172. doi:10.1111/j .1529-8019.2011.01392.x
- Miyoshi S-I. Vibrio vulnificus infection and metalloprotease. J Dermatol. 2006;33:589-595. doi:10.1111/j.1346-8138.2006.00139.x
- Patel VJ, Gardner E, Burton CS. Vibrio vulnificus septicemia and leg ulcer. J Am Acad Dermatol. 2002;46(5 suppl):S144-S145. doi:10.1067 /mjd.2002.107778
- Baker-Austin C, Oliver JD. Vibrio vulnificus: new insights into a deadly opportunistic pathogen. Environ Microbiol. 2018;20:423-430. doi:10.1111/1462-2920.13955
- Preliminary FoodNet data on the incidence of infection with pathogens transmitted commonly through food —10 states, 2009. CDC website. Published April 16, 2010. Accessed November 3, 2021. https://www.cdc .gov/mmwr/preview/mmwrhtml/mm5914a2.htm
- Merkel SM, Alexander S, Zufall E, et al. Essential role for estrogen in protection against Vibrio vulnificus-induced endotoxic shock. Infect Immun. 2001;69:6119-6122. doi:10.1128/IAI.69.10.6119 -6122.2001
- Scaglione S, Kliethermes S, Cao G, et al. The epidemiology of cirrhosis in the United States: a population-based study. J Clin Gastroenterol. 2015;49:690-696. doi:10.1097/MCG.0000000000000208
- Jones M, Oliver J. Vibrio vulnificus: disease and pathogenesis [published online December 20, 2020]. Infect Immun. https://doi.org/10.1128 /IAI.01046-08
- Paz S, Bisharat N, Paz E, et al. Climate change and the emergence of Vibrio vulnificus disease in Israel. Environ Res. 2007;103:390-396. doi:10.1016/j.envres.2006.07.002
- Martinez-Urtaza J, Bowers JC, Trinanes J, et al. Climate anomalies and the increasing risk of Vibrio parahaemolyticus and Vibrio vulnificus illnesses. Food Res Int. 2010;43:1780-1790. doi:10.1016/j. foodres.2010.04.001
- Oliver JD. Vibrio vulnificus. In: Thompson FL, Austin B, Swings J, eds. The Biology of Vibrios. ASM Press; 2006:349-366.
- Baker-Austin C, McArthur JV, Lindell AH, et al. Multi-site analysis reveals widespread antibiotic resistance in the marine pathogen Vibrio vulnificus. Microb Ecol. 2009;57:151-159. doi:10.1007 /s00248-008-9413-8
- Elmahdi S, DaSilva LV, Parveen S. Antibiotic resistance of Vibrio parahaemolyticus and Vibrio vulnificus in various countries: a review. Food Microbiol. 2016;57:128-134. doi:10.1016/j.fm.2016.02.008
- Prasad P, Wong V, Burgin S, et al. Mucormycosis. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/mucormycosis?diagnosisId=51981 &moduleId=101
- Blum A, Song P, Tan B, et al. Calciphylaxis. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/calciphylaxis?diagnosisId=51241&moduleId=101
- Cohen J, Wong V, Burgin S. Pyoderma gangrenosum. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/pyoderma+gangrenosum?diagnosis Id=52242&moduleId=101
- Walls A, Burgin S. Stevens-Johnson syndrome. VisualDx website. Accessed November 13, 2021. https://www-visualdx-com.proxy.lib.ohio-state.edu/visualdx/diagnosis/stevens-johnson+syndrome?diagnosisId=52342&moduleId=101
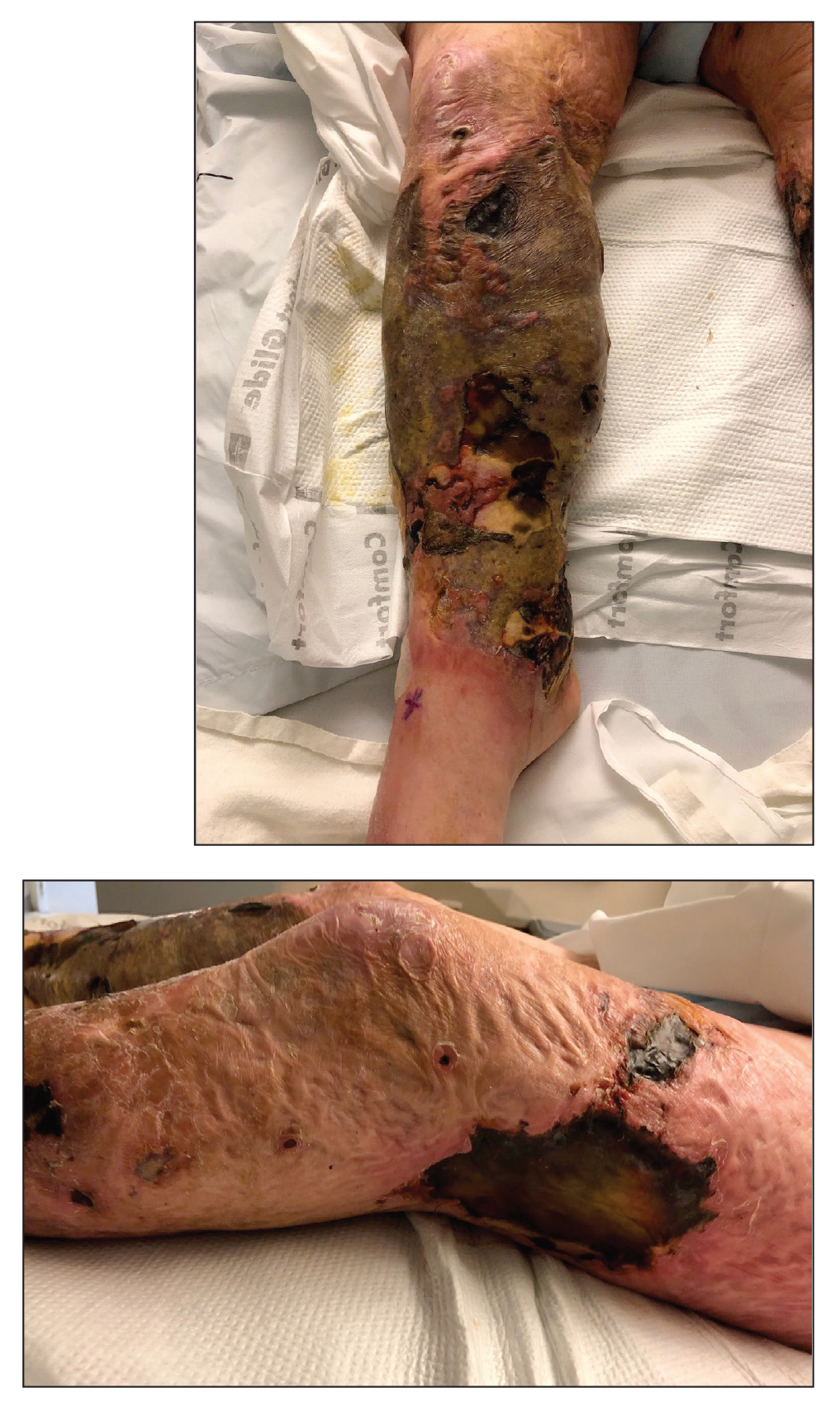
A 48-year-old man presented to the emergency department with pain in both legs after swimming in the ocean surrounding Florida 1 month prior to presentation. His medical history included skin graft treatment of burns during childhood and a chronic lower extremity ulcer that developed after trauma. He received hemodialysis for acute renal failure approximately 1 month prior to the current presentation. At the current presentation he was found to be septic and quickly developed rapidly expanding regions of retiform purpura with stellate necrosis on the legs.