User login
Lowering portal pressure boosts cirrhosis outcomes
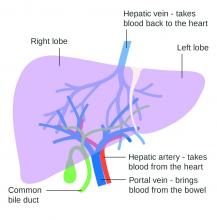
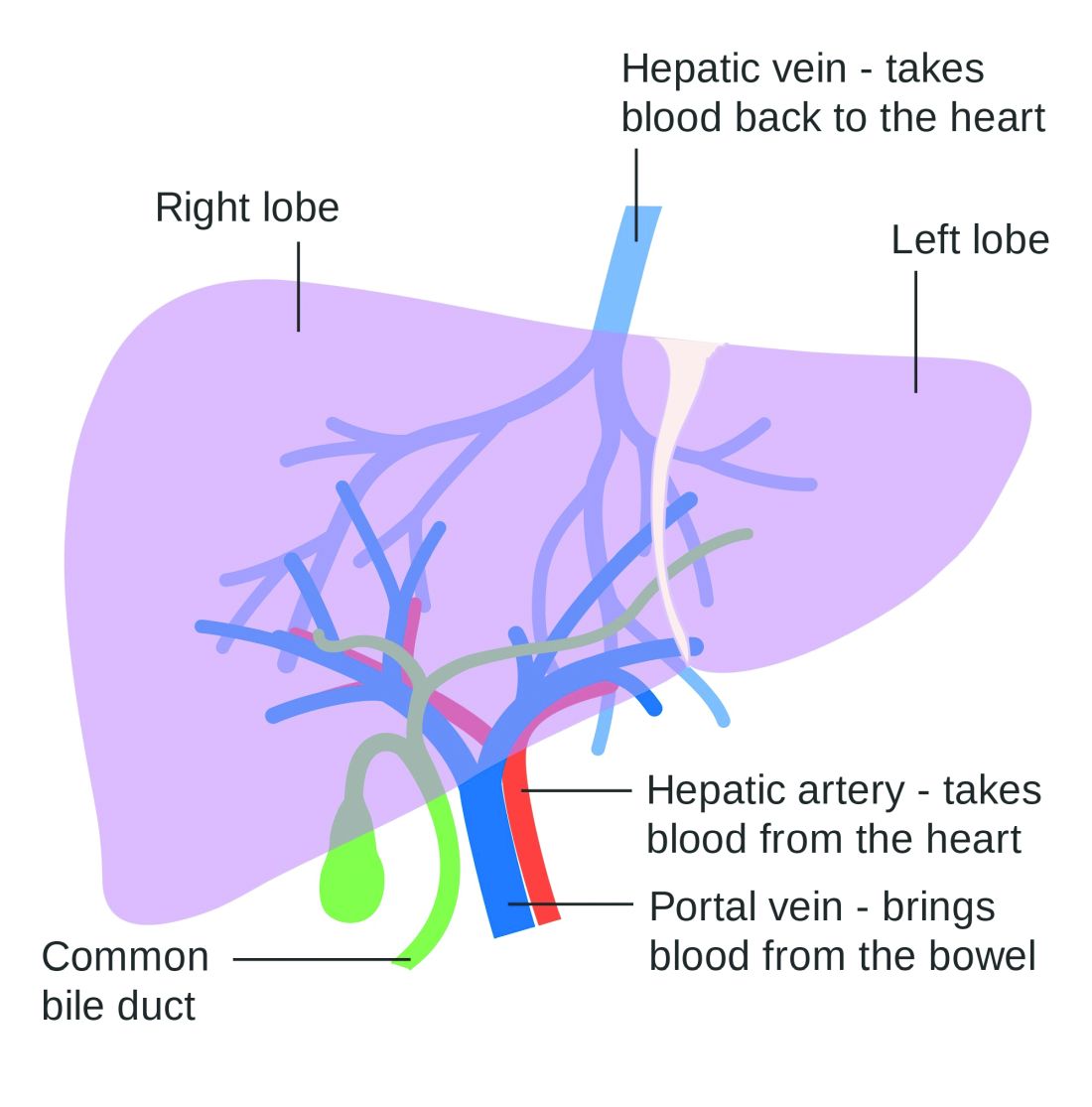
Use of nonselective beta-blockers to reduce portal pressure in cirrhosis improved outcomes in adults with or without ascites, based on data from a meta-analysis of more than 1,000 patients.
Previous research has suggested that nonselective beta-blockers (NSBBs) might have a negative effect on patients with refractory ascites, but the effect on patients with and without ascites has not been assessed, wrote Laura Turco, MD, of the University of Modena and Reggio Emilia, Emilia-Romagna, Italy, and colleagues.
In a study published in Clinical Gastroenterology and Hepatology, the researchers analyzed 1,113 cirrhosis patients including 452 with ascites. Overall, 968 patients had received treatment with NSBBs. Response to pressure reduction was defined as a decrease of more than 20% from baseline or a decrease to less than 12 mm Hg.
A total of 329 of the 661 patients without ascites (50%) met the definition as responders. These responders had significantly lower odds than did nonresponders of a combination of clinical events including ascites, variceal hemorrhage, or encephalopathy (odds ratio, 0.35) and also had significantly lower odds than nonresponders of liver transplantation or death (OR, 0.50).
A total of 188 of the 452 patients with ascites were responders (42%). These responders had significantly lower odds than those of nonresponders (OR, 0.27) of variceal hemorrhage, refractory ascites, spontaneous bacterial peritonitis, or hepatorenal syndrome. The responders also had significantly lower odds of liver transplantation or death (OR, 0.47).
The results are important in light of concerns about the impact of NSBBs on renal function and mortality in cirrhosis patients with ascites, the researchers said.
The study findings were limited by several factors, including the use of retrospective data from prospective studies, and the incomplete collection of data on the variables of comorbidities, hepatocellular carcinoma, and other predictive scores; alcohol use or abstinence was a potential confounder as well, the researchers noted.
However, “By showing that reductions in portal pressure induced by NSBB-based pharmacologic therapy improve outcomes and decrease mortality, our study supports the use of NSBB in all clinical settings (primary or secondary prophylaxis) and in both patients with or without ascites,” they concluded. They found no heterogeneity among the studies included in the analysis.
The study was supported by multiple sources including the University of Modena and Reggio Emilia, Yale Liver Center, National Institutes of Health, and Instituto de Salud Carlos III, and was cofunded by the European Union. The researchers had no financial conflicts to disclose.
SOURCE: Turco L et al. Clin Gastroenterol Hepatol. 2019 June 5. doi: 10.1016/j.cgh.2019.05.050.
Use of nonselective beta-blockers to reduce portal pressure in cirrhosis improved outcomes in adults with or without ascites, based on data from a meta-analysis of more than 1,000 patients.
Previous research has suggested that nonselective beta-blockers (NSBBs) might have a negative effect on patients with refractory ascites, but the effect on patients with and without ascites has not been assessed, wrote Laura Turco, MD, of the University of Modena and Reggio Emilia, Emilia-Romagna, Italy, and colleagues.
In a study published in Clinical Gastroenterology and Hepatology, the researchers analyzed 1,113 cirrhosis patients including 452 with ascites. Overall, 968 patients had received treatment with NSBBs. Response to pressure reduction was defined as a decrease of more than 20% from baseline or a decrease to less than 12 mm Hg.
A total of 329 of the 661 patients without ascites (50%) met the definition as responders. These responders had significantly lower odds than did nonresponders of a combination of clinical events including ascites, variceal hemorrhage, or encephalopathy (odds ratio, 0.35) and also had significantly lower odds than nonresponders of liver transplantation or death (OR, 0.50).
A total of 188 of the 452 patients with ascites were responders (42%). These responders had significantly lower odds than those of nonresponders (OR, 0.27) of variceal hemorrhage, refractory ascites, spontaneous bacterial peritonitis, or hepatorenal syndrome. The responders also had significantly lower odds of liver transplantation or death (OR, 0.47).
The results are important in light of concerns about the impact of NSBBs on renal function and mortality in cirrhosis patients with ascites, the researchers said.
The study findings were limited by several factors, including the use of retrospective data from prospective studies, and the incomplete collection of data on the variables of comorbidities, hepatocellular carcinoma, and other predictive scores; alcohol use or abstinence was a potential confounder as well, the researchers noted.
However, “By showing that reductions in portal pressure induced by NSBB-based pharmacologic therapy improve outcomes and decrease mortality, our study supports the use of NSBB in all clinical settings (primary or secondary prophylaxis) and in both patients with or without ascites,” they concluded. They found no heterogeneity among the studies included in the analysis.
The study was supported by multiple sources including the University of Modena and Reggio Emilia, Yale Liver Center, National Institutes of Health, and Instituto de Salud Carlos III, and was cofunded by the European Union. The researchers had no financial conflicts to disclose.
SOURCE: Turco L et al. Clin Gastroenterol Hepatol. 2019 June 5. doi: 10.1016/j.cgh.2019.05.050.
Use of nonselective beta-blockers to reduce portal pressure in cirrhosis improved outcomes in adults with or without ascites, based on data from a meta-analysis of more than 1,000 patients.
Previous research has suggested that nonselective beta-blockers (NSBBs) might have a negative effect on patients with refractory ascites, but the effect on patients with and without ascites has not been assessed, wrote Laura Turco, MD, of the University of Modena and Reggio Emilia, Emilia-Romagna, Italy, and colleagues.
In a study published in Clinical Gastroenterology and Hepatology, the researchers analyzed 1,113 cirrhosis patients including 452 with ascites. Overall, 968 patients had received treatment with NSBBs. Response to pressure reduction was defined as a decrease of more than 20% from baseline or a decrease to less than 12 mm Hg.
A total of 329 of the 661 patients without ascites (50%) met the definition as responders. These responders had significantly lower odds than did nonresponders of a combination of clinical events including ascites, variceal hemorrhage, or encephalopathy (odds ratio, 0.35) and also had significantly lower odds than nonresponders of liver transplantation or death (OR, 0.50).
A total of 188 of the 452 patients with ascites were responders (42%). These responders had significantly lower odds than those of nonresponders (OR, 0.27) of variceal hemorrhage, refractory ascites, spontaneous bacterial peritonitis, or hepatorenal syndrome. The responders also had significantly lower odds of liver transplantation or death (OR, 0.47).
The results are important in light of concerns about the impact of NSBBs on renal function and mortality in cirrhosis patients with ascites, the researchers said.
The study findings were limited by several factors, including the use of retrospective data from prospective studies, and the incomplete collection of data on the variables of comorbidities, hepatocellular carcinoma, and other predictive scores; alcohol use or abstinence was a potential confounder as well, the researchers noted.
However, “By showing that reductions in portal pressure induced by NSBB-based pharmacologic therapy improve outcomes and decrease mortality, our study supports the use of NSBB in all clinical settings (primary or secondary prophylaxis) and in both patients with or without ascites,” they concluded. They found no heterogeneity among the studies included in the analysis.
The study was supported by multiple sources including the University of Modena and Reggio Emilia, Yale Liver Center, National Institutes of Health, and Instituto de Salud Carlos III, and was cofunded by the European Union. The researchers had no financial conflicts to disclose.
SOURCE: Turco L et al. Clin Gastroenterol Hepatol. 2019 June 5. doi: 10.1016/j.cgh.2019.05.050.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
ACP unveils clinical guideline disclosure strategy
The American College of Physicians recently described its methods for developing clinical guidelines and guidance statements, in a paper published in the Annals of Internal Medicine.
Any person involved in the development of an ACP clinical guideline or guidance statement must disclose all financial and intellectual interests related to health care from the previous 3 years,” Amir Qaseem, MD, and Timothy J. Wilt, MD, wrote.
“The goals of our process are to mitigate any actual bias during the development of ACP’s clinical recommendations and to ensure creditability and public trust in our clinical policies by reducing the potential for perceived bias,” noted Robert M. McLean, MD, president of the ACP, in a statement.
This paper’s publication comes on the heels of authors of a Cancer paper having reported that nearly 25% of the American Society of Clinical Oncology’s guideline authors who were not exempt from reporting conflicts of interest failed to disclose receiving industry payments.
The ACP committee’s guiding principle for collection of disclosures of interest and management of conflicts of interests “is to prioritize the interests of the patient over any competing or professional interests via an evidence-based assessment of the benefits, harms, and costs of an intervention,” wrote the authors on behalf of the CGC.
The CGC created a tiered system to classify potential conflicts as low level, moderate level, or high level based on three tenets: transparency (all disclosures are freely accessible so readers can assess them for themselves), proportionality (not all conflicts of interest have equal risk), and consistency (policies should be impartially applied across all variables).
Examples of low-level conflicts of interest (COIs) include high-level COIs that have become inactive and intellectual interests tangentially related to the topic under discussion. Moderate-level COIs are usually intellectual interests clinically relevant to the guideline topic; these interests might prompt an individual to seek professional or financial advantages through association with guideline development.
High-level COIs are active relationships with high-risk entities, defined by the CGC as “an entity that has a direct financial stake in the clinical conclusions of a guideline or guidance statement.”
While the time frame for reporting health-related interests is 3 years, disclosure is an ongoing process when clinical guidelines are in development because interests change over time, the authors said. Prospective guidelines committee members complete disclosure of interest forms before working on CGC projects, and they update these forms before each in-person CGC meeting.
“The CGC’s policy does not mandate disclosure of interests related primarily to personal matters or relationships outside the household,” such as political, religious, or ideological views, they noted.
The CGC maintains a DOI-COI Review and Management Panel to reviews conflicts, and all ACP guidelines include a list of relevant conflicts for committee members.
The authors of this paper disclosed no conflicts of interest.
SOURCE: Qaseem A and TJ Wilt. Ann Intern Med. 2019 Aug 20. doi: 10.7326/M18-3279 .
This article was updated 8/22/19.
The American College of Physicians recently described its methods for developing clinical guidelines and guidance statements, in a paper published in the Annals of Internal Medicine.
Any person involved in the development of an ACP clinical guideline or guidance statement must disclose all financial and intellectual interests related to health care from the previous 3 years,” Amir Qaseem, MD, and Timothy J. Wilt, MD, wrote.
“The goals of our process are to mitigate any actual bias during the development of ACP’s clinical recommendations and to ensure creditability and public trust in our clinical policies by reducing the potential for perceived bias,” noted Robert M. McLean, MD, president of the ACP, in a statement.
This paper’s publication comes on the heels of authors of a Cancer paper having reported that nearly 25% of the American Society of Clinical Oncology’s guideline authors who were not exempt from reporting conflicts of interest failed to disclose receiving industry payments.
The ACP committee’s guiding principle for collection of disclosures of interest and management of conflicts of interests “is to prioritize the interests of the patient over any competing or professional interests via an evidence-based assessment of the benefits, harms, and costs of an intervention,” wrote the authors on behalf of the CGC.
The CGC created a tiered system to classify potential conflicts as low level, moderate level, or high level based on three tenets: transparency (all disclosures are freely accessible so readers can assess them for themselves), proportionality (not all conflicts of interest have equal risk), and consistency (policies should be impartially applied across all variables).
Examples of low-level conflicts of interest (COIs) include high-level COIs that have become inactive and intellectual interests tangentially related to the topic under discussion. Moderate-level COIs are usually intellectual interests clinically relevant to the guideline topic; these interests might prompt an individual to seek professional or financial advantages through association with guideline development.
High-level COIs are active relationships with high-risk entities, defined by the CGC as “an entity that has a direct financial stake in the clinical conclusions of a guideline or guidance statement.”
While the time frame for reporting health-related interests is 3 years, disclosure is an ongoing process when clinical guidelines are in development because interests change over time, the authors said. Prospective guidelines committee members complete disclosure of interest forms before working on CGC projects, and they update these forms before each in-person CGC meeting.
“The CGC’s policy does not mandate disclosure of interests related primarily to personal matters or relationships outside the household,” such as political, religious, or ideological views, they noted.
The CGC maintains a DOI-COI Review and Management Panel to reviews conflicts, and all ACP guidelines include a list of relevant conflicts for committee members.
The authors of this paper disclosed no conflicts of interest.
SOURCE: Qaseem A and TJ Wilt. Ann Intern Med. 2019 Aug 20. doi: 10.7326/M18-3279 .
This article was updated 8/22/19.
The American College of Physicians recently described its methods for developing clinical guidelines and guidance statements, in a paper published in the Annals of Internal Medicine.
Any person involved in the development of an ACP clinical guideline or guidance statement must disclose all financial and intellectual interests related to health care from the previous 3 years,” Amir Qaseem, MD, and Timothy J. Wilt, MD, wrote.
“The goals of our process are to mitigate any actual bias during the development of ACP’s clinical recommendations and to ensure creditability and public trust in our clinical policies by reducing the potential for perceived bias,” noted Robert M. McLean, MD, president of the ACP, in a statement.
This paper’s publication comes on the heels of authors of a Cancer paper having reported that nearly 25% of the American Society of Clinical Oncology’s guideline authors who were not exempt from reporting conflicts of interest failed to disclose receiving industry payments.
The ACP committee’s guiding principle for collection of disclosures of interest and management of conflicts of interests “is to prioritize the interests of the patient over any competing or professional interests via an evidence-based assessment of the benefits, harms, and costs of an intervention,” wrote the authors on behalf of the CGC.
The CGC created a tiered system to classify potential conflicts as low level, moderate level, or high level based on three tenets: transparency (all disclosures are freely accessible so readers can assess them for themselves), proportionality (not all conflicts of interest have equal risk), and consistency (policies should be impartially applied across all variables).
Examples of low-level conflicts of interest (COIs) include high-level COIs that have become inactive and intellectual interests tangentially related to the topic under discussion. Moderate-level COIs are usually intellectual interests clinically relevant to the guideline topic; these interests might prompt an individual to seek professional or financial advantages through association with guideline development.
High-level COIs are active relationships with high-risk entities, defined by the CGC as “an entity that has a direct financial stake in the clinical conclusions of a guideline or guidance statement.”
While the time frame for reporting health-related interests is 3 years, disclosure is an ongoing process when clinical guidelines are in development because interests change over time, the authors said. Prospective guidelines committee members complete disclosure of interest forms before working on CGC projects, and they update these forms before each in-person CGC meeting.
“The CGC’s policy does not mandate disclosure of interests related primarily to personal matters or relationships outside the household,” such as political, religious, or ideological views, they noted.
The CGC maintains a DOI-COI Review and Management Panel to reviews conflicts, and all ACP guidelines include a list of relevant conflicts for committee members.
The authors of this paper disclosed no conflicts of interest.
SOURCE: Qaseem A and TJ Wilt. Ann Intern Med. 2019 Aug 20. doi: 10.7326/M18-3279 .
This article was updated 8/22/19.
FROM THE ANNALS OF INTERNAL MEDICINE
Statins hamper hepatocellular carcinoma in viral hepatitis patients
Lipophilic statin therapy significantly reduced the incidence and mortality of hepatocellular carcinoma in adults with viral hepatitis, based on data from 16,668 patients.
The mortality rates for hepatocellular carcinoma in the United States and Europe have been on the rise for decades, and the risk may persist in severe cases despite the use of hepatitis B virus suppression or hepatitis C virus eradication, wrote Tracey G. Simon, MD, of Harvard Medical School, Boston, and colleagues. Previous studies suggest that statins might reduce HCC risk in viral hepatitis patients, but evidence supporting one type of statin over another for HCC prevention is limited, they said.
In a study published in the Annals of Internal Medicine, the researchers reviewed data from a national registry of hepatitis patients in Sweden to assess the effect of lipophilic or hydrophilic statin use on HCC incidence and mortality.
They found a significant reduction in 10-year HCC risk for lipophilic statin users, compared with nonusers (8.1% vs. 3.3%. However, the difference was not significant for hydrophilic statin users vs. nonusers (8.0% vs. 6.8%). The effect of lipophilic statin use was dose dependent; the largest effect on reduction in HCC risk occurred with 600 or more lipophilic statin cumulative daily doses in users, compared with nonusers (8.4% vs. 2.5%).
The study population included 6,554 lipophilic statin users and 1,780 hydrophilic statin users, matched with 8,334 nonusers. Patient demographics were similar between both types of statin user and nonuser groups.
In addition, 10-year mortality was significantly lower for lipophilic statin users compared with nonusers (15.2% vs. 7.3%) and also for hydrophilic statin users, compared with nonusers (16.0% vs. 11.5%).
In a small number of patients with liver disease (462), liver-specific mortality was significantly reduced in lipophilic statin users, compared with nonusers (adjusted hazard ratio, 0.76 vs. 0.98).
“Of note, our findings were robust across several sensitivity analyses and were similar in all predefined subgroups, including among men and women and persons with and without cirrhosis or antiviral therapy use,” the researchers noted.
The study findings were limited by several factors including the potential confounding from variables such as smoking, hepatitis B viral DNA, hepatitis C virus eradication, stage of fibrosis, and HCC screening, as well as a lack of laboratory data to assess cholesterol levels’ impact on statin use, the researchers said. In addition, the study did not compare lipophilic and hydrophilic statins.
However, the results suggest potential distinct benefits of lipophilic statins to reduce HCC risk and support the need for further research, the researchers concluded.
Dr. Simon had no financial conflicts to disclose, but disclosed support from a North American Training Grant from the American College of Gastroenterology. Several coauthors disclosed relationships with multiple companies including AbbVie, Bristol-Myers Squibb, Gilead, Janssen, and Merck Sharp & Dohme. The study was supported in part by the American College of Gastroenterology, the American Association for the Study of Liver Diseases, the Boston Nutrition Obesity Research Center, the National Institutes of Health, Nyckelfonden, Region Orebro (Sweden) County, and the Karolinska Institutet.
SOURCE: Simon TG et al. Ann Intern Med. 2019 Aug 19. doi: 10.7326/M18-2753.
Lipophilic statin therapy significantly reduced the incidence and mortality of hepatocellular carcinoma in adults with viral hepatitis, based on data from 16,668 patients.
The mortality rates for hepatocellular carcinoma in the United States and Europe have been on the rise for decades, and the risk may persist in severe cases despite the use of hepatitis B virus suppression or hepatitis C virus eradication, wrote Tracey G. Simon, MD, of Harvard Medical School, Boston, and colleagues. Previous studies suggest that statins might reduce HCC risk in viral hepatitis patients, but evidence supporting one type of statin over another for HCC prevention is limited, they said.
In a study published in the Annals of Internal Medicine, the researchers reviewed data from a national registry of hepatitis patients in Sweden to assess the effect of lipophilic or hydrophilic statin use on HCC incidence and mortality.
They found a significant reduction in 10-year HCC risk for lipophilic statin users, compared with nonusers (8.1% vs. 3.3%. However, the difference was not significant for hydrophilic statin users vs. nonusers (8.0% vs. 6.8%). The effect of lipophilic statin use was dose dependent; the largest effect on reduction in HCC risk occurred with 600 or more lipophilic statin cumulative daily doses in users, compared with nonusers (8.4% vs. 2.5%).
The study population included 6,554 lipophilic statin users and 1,780 hydrophilic statin users, matched with 8,334 nonusers. Patient demographics were similar between both types of statin user and nonuser groups.
In addition, 10-year mortality was significantly lower for lipophilic statin users compared with nonusers (15.2% vs. 7.3%) and also for hydrophilic statin users, compared with nonusers (16.0% vs. 11.5%).
In a small number of patients with liver disease (462), liver-specific mortality was significantly reduced in lipophilic statin users, compared with nonusers (adjusted hazard ratio, 0.76 vs. 0.98).
“Of note, our findings were robust across several sensitivity analyses and were similar in all predefined subgroups, including among men and women and persons with and without cirrhosis or antiviral therapy use,” the researchers noted.
The study findings were limited by several factors including the potential confounding from variables such as smoking, hepatitis B viral DNA, hepatitis C virus eradication, stage of fibrosis, and HCC screening, as well as a lack of laboratory data to assess cholesterol levels’ impact on statin use, the researchers said. In addition, the study did not compare lipophilic and hydrophilic statins.
However, the results suggest potential distinct benefits of lipophilic statins to reduce HCC risk and support the need for further research, the researchers concluded.
Dr. Simon had no financial conflicts to disclose, but disclosed support from a North American Training Grant from the American College of Gastroenterology. Several coauthors disclosed relationships with multiple companies including AbbVie, Bristol-Myers Squibb, Gilead, Janssen, and Merck Sharp & Dohme. The study was supported in part by the American College of Gastroenterology, the American Association for the Study of Liver Diseases, the Boston Nutrition Obesity Research Center, the National Institutes of Health, Nyckelfonden, Region Orebro (Sweden) County, and the Karolinska Institutet.
SOURCE: Simon TG et al. Ann Intern Med. 2019 Aug 19. doi: 10.7326/M18-2753.
Lipophilic statin therapy significantly reduced the incidence and mortality of hepatocellular carcinoma in adults with viral hepatitis, based on data from 16,668 patients.
The mortality rates for hepatocellular carcinoma in the United States and Europe have been on the rise for decades, and the risk may persist in severe cases despite the use of hepatitis B virus suppression or hepatitis C virus eradication, wrote Tracey G. Simon, MD, of Harvard Medical School, Boston, and colleagues. Previous studies suggest that statins might reduce HCC risk in viral hepatitis patients, but evidence supporting one type of statin over another for HCC prevention is limited, they said.
In a study published in the Annals of Internal Medicine, the researchers reviewed data from a national registry of hepatitis patients in Sweden to assess the effect of lipophilic or hydrophilic statin use on HCC incidence and mortality.
They found a significant reduction in 10-year HCC risk for lipophilic statin users, compared with nonusers (8.1% vs. 3.3%. However, the difference was not significant for hydrophilic statin users vs. nonusers (8.0% vs. 6.8%). The effect of lipophilic statin use was dose dependent; the largest effect on reduction in HCC risk occurred with 600 or more lipophilic statin cumulative daily doses in users, compared with nonusers (8.4% vs. 2.5%).
The study population included 6,554 lipophilic statin users and 1,780 hydrophilic statin users, matched with 8,334 nonusers. Patient demographics were similar between both types of statin user and nonuser groups.
In addition, 10-year mortality was significantly lower for lipophilic statin users compared with nonusers (15.2% vs. 7.3%) and also for hydrophilic statin users, compared with nonusers (16.0% vs. 11.5%).
In a small number of patients with liver disease (462), liver-specific mortality was significantly reduced in lipophilic statin users, compared with nonusers (adjusted hazard ratio, 0.76 vs. 0.98).
“Of note, our findings were robust across several sensitivity analyses and were similar in all predefined subgroups, including among men and women and persons with and without cirrhosis or antiviral therapy use,” the researchers noted.
The study findings were limited by several factors including the potential confounding from variables such as smoking, hepatitis B viral DNA, hepatitis C virus eradication, stage of fibrosis, and HCC screening, as well as a lack of laboratory data to assess cholesterol levels’ impact on statin use, the researchers said. In addition, the study did not compare lipophilic and hydrophilic statins.
However, the results suggest potential distinct benefits of lipophilic statins to reduce HCC risk and support the need for further research, the researchers concluded.
Dr. Simon had no financial conflicts to disclose, but disclosed support from a North American Training Grant from the American College of Gastroenterology. Several coauthors disclosed relationships with multiple companies including AbbVie, Bristol-Myers Squibb, Gilead, Janssen, and Merck Sharp & Dohme. The study was supported in part by the American College of Gastroenterology, the American Association for the Study of Liver Diseases, the Boston Nutrition Obesity Research Center, the National Institutes of Health, Nyckelfonden, Region Orebro (Sweden) County, and the Karolinska Institutet.
SOURCE: Simon TG et al. Ann Intern Med. 2019 Aug 19. doi: 10.7326/M18-2753.
FROM THE ANNALS OF INTERNAL MEDICINE
Key clinical point: Use of lipophilic statins significantly reduced incidence and mortality of hepatocellular cancer in adults with viral hepatitis.
Major finding: The 10-year risk of HCC was 8.1% among patients taking lipophilic statins, compared with 3.3% among those not on statins.
Study details: The data come from a population-based cohort study of 16,668 adult with viral hepatitis from a national registry in Sweden.
Disclosures: Dr. Simon had no financial conflicts to disclose, but disclosed support from a North American Training Grant from the American College of Gastroenterology. Several coauthors disclosed relationships with multiple companies including AbbVie, Bristol-Myers Squibb, Gilead, Janssen, and MSD.
Source: Simon TG et al. Ann Intern Med. 2019 Aug 19. doi: 10.7326/M18-2753.
USPSTF draft guidance calls for drug use screening
according to a draft recommendation statement now available for public comment.
The statement defines illicit drug use as “use of illegal drugs and the nonmedical use of prescription psychoactive medications (i.e., use for reasons, for duration, in amounts, or with frequency other than prescribed or use by persons other than the prescribed individual).”
The guidelines do not apply to individuals younger than 18 years, for whom the USPSTF found insufficient evidence to recommend routine screening, or to adults currently diagnosed or in treatment for a drug use disorder.
In the draft recommendation statement, available online, the USPSTF noted that several screening tools are available for use in primary care practices, including the BSTAD (Brief Screener for Tobacco, Alcohol, and Other Drugs) that consists of six questions. The task force noted that they have found “adequate evidence” that these screening tools can detect illicit drug use. In addition, they wrote that no studies offer evidence of benefits versus harms of these screening tools, and evidence of harms associated with screening are limited.
Screening intervals can be simplified by screening young adults whenever they seek medical services and when clinicians suspect illicit drug use, the USPSTF said.
When the draft recommendation is finalized, it will replace the 2008 recommendation, which found insufficient evidence for screening in adults, as well as in adolescents. New evidence since 2008 supports the value of screening for adults aged 18 years and older, including pregnant and postpartum women.
The draft recommendations are based on the results of two systematic evidence reviews that assessed the accuracy and harms of routine illicit drug use screening. The USPSTF’s review included 12 studies on the accuracy of 15 screening tools. Overall, the sensitivity of direct screening tools to identify “unhealthy use of ‘any drug’ (including illegal drugs and nonmedical use of prescription drugs) in the past month or year” ranged from 0.71 to 0.94, and the specificity ranged from 0.87 to 0.97.
Based on the current evidence, the USPSTF assigned drug screening for adults a grade B recommendation, defined as “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial.”
For treatment, the Task Force found evidence to support strategies including pharmacotherapy with naltrexone, buprenorphine, and methadone, as well as for psychosocial interventions.
The USPSTF acknowledged that many factors may affect a clinicians’ decision of whether to implement the drug screening recommendation. “In many communities, affordable, accessible, and timely services for diagnostic assessment and treatment for patients with positive screening results are in limited supply or unaffordable. Providers should be aware of any state requirements for mandatory screening or reporting of screening results to medicolegal authorities and understand the positive and negative implications of reporting,” they wrote.
The draft recommendations also identified several research gaps including the effectiveness of screening for illicit drug use in adolescents, the optimal screening interval for all patients, the accuracy of screening tools for detecting opioids, the accuracy of screening within the same population, the benefits of naloxone as rescue therapy, and nonmedical use of other prescription drugs, as well as ways to improve access to care for those diagnosed with drug use disorders.
The draft recommendation is available for public comment until Sept. 9, 2019, at 8 p.m. EST.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers had no financial conflicts to disclose.
according to a draft recommendation statement now available for public comment.
The statement defines illicit drug use as “use of illegal drugs and the nonmedical use of prescription psychoactive medications (i.e., use for reasons, for duration, in amounts, or with frequency other than prescribed or use by persons other than the prescribed individual).”
The guidelines do not apply to individuals younger than 18 years, for whom the USPSTF found insufficient evidence to recommend routine screening, or to adults currently diagnosed or in treatment for a drug use disorder.
In the draft recommendation statement, available online, the USPSTF noted that several screening tools are available for use in primary care practices, including the BSTAD (Brief Screener for Tobacco, Alcohol, and Other Drugs) that consists of six questions. The task force noted that they have found “adequate evidence” that these screening tools can detect illicit drug use. In addition, they wrote that no studies offer evidence of benefits versus harms of these screening tools, and evidence of harms associated with screening are limited.
Screening intervals can be simplified by screening young adults whenever they seek medical services and when clinicians suspect illicit drug use, the USPSTF said.
When the draft recommendation is finalized, it will replace the 2008 recommendation, which found insufficient evidence for screening in adults, as well as in adolescents. New evidence since 2008 supports the value of screening for adults aged 18 years and older, including pregnant and postpartum women.
The draft recommendations are based on the results of two systematic evidence reviews that assessed the accuracy and harms of routine illicit drug use screening. The USPSTF’s review included 12 studies on the accuracy of 15 screening tools. Overall, the sensitivity of direct screening tools to identify “unhealthy use of ‘any drug’ (including illegal drugs and nonmedical use of prescription drugs) in the past month or year” ranged from 0.71 to 0.94, and the specificity ranged from 0.87 to 0.97.
Based on the current evidence, the USPSTF assigned drug screening for adults a grade B recommendation, defined as “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial.”
For treatment, the Task Force found evidence to support strategies including pharmacotherapy with naltrexone, buprenorphine, and methadone, as well as for psychosocial interventions.
The USPSTF acknowledged that many factors may affect a clinicians’ decision of whether to implement the drug screening recommendation. “In many communities, affordable, accessible, and timely services for diagnostic assessment and treatment for patients with positive screening results are in limited supply or unaffordable. Providers should be aware of any state requirements for mandatory screening or reporting of screening results to medicolegal authorities and understand the positive and negative implications of reporting,” they wrote.
The draft recommendations also identified several research gaps including the effectiveness of screening for illicit drug use in adolescents, the optimal screening interval for all patients, the accuracy of screening tools for detecting opioids, the accuracy of screening within the same population, the benefits of naloxone as rescue therapy, and nonmedical use of other prescription drugs, as well as ways to improve access to care for those diagnosed with drug use disorders.
The draft recommendation is available for public comment until Sept. 9, 2019, at 8 p.m. EST.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers had no financial conflicts to disclose.
according to a draft recommendation statement now available for public comment.
The statement defines illicit drug use as “use of illegal drugs and the nonmedical use of prescription psychoactive medications (i.e., use for reasons, for duration, in amounts, or with frequency other than prescribed or use by persons other than the prescribed individual).”
The guidelines do not apply to individuals younger than 18 years, for whom the USPSTF found insufficient evidence to recommend routine screening, or to adults currently diagnosed or in treatment for a drug use disorder.
In the draft recommendation statement, available online, the USPSTF noted that several screening tools are available for use in primary care practices, including the BSTAD (Brief Screener for Tobacco, Alcohol, and Other Drugs) that consists of six questions. The task force noted that they have found “adequate evidence” that these screening tools can detect illicit drug use. In addition, they wrote that no studies offer evidence of benefits versus harms of these screening tools, and evidence of harms associated with screening are limited.
Screening intervals can be simplified by screening young adults whenever they seek medical services and when clinicians suspect illicit drug use, the USPSTF said.
When the draft recommendation is finalized, it will replace the 2008 recommendation, which found insufficient evidence for screening in adults, as well as in adolescents. New evidence since 2008 supports the value of screening for adults aged 18 years and older, including pregnant and postpartum women.
The draft recommendations are based on the results of two systematic evidence reviews that assessed the accuracy and harms of routine illicit drug use screening. The USPSTF’s review included 12 studies on the accuracy of 15 screening tools. Overall, the sensitivity of direct screening tools to identify “unhealthy use of ‘any drug’ (including illegal drugs and nonmedical use of prescription drugs) in the past month or year” ranged from 0.71 to 0.94, and the specificity ranged from 0.87 to 0.97.
Based on the current evidence, the USPSTF assigned drug screening for adults a grade B recommendation, defined as “high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial.”
For treatment, the Task Force found evidence to support strategies including pharmacotherapy with naltrexone, buprenorphine, and methadone, as well as for psychosocial interventions.
The USPSTF acknowledged that many factors may affect a clinicians’ decision of whether to implement the drug screening recommendation. “In many communities, affordable, accessible, and timely services for diagnostic assessment and treatment for patients with positive screening results are in limited supply or unaffordable. Providers should be aware of any state requirements for mandatory screening or reporting of screening results to medicolegal authorities and understand the positive and negative implications of reporting,” they wrote.
The draft recommendations also identified several research gaps including the effectiveness of screening for illicit drug use in adolescents, the optimal screening interval for all patients, the accuracy of screening tools for detecting opioids, the accuracy of screening within the same population, the benefits of naloxone as rescue therapy, and nonmedical use of other prescription drugs, as well as ways to improve access to care for those diagnosed with drug use disorders.
The draft recommendation is available for public comment until Sept. 9, 2019, at 8 p.m. EST.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers had no financial conflicts to disclose.
FROM THE USPSTF
Maternal factors impact childhood skin microbiota
Bacteria on children’s skin was similar to their mothers’ and affected by factors that included method of delivery and breastfeeding in a study of 154 children aged 10 years and younger.
Understanding the wrote Ting Zhu of Fudan University, Shanghai, China, and colleagues.
In a study published in the Journal of Investigative Dermatology, the researchers compared the skin microbiota of the 158 children aged 1-10 years and 50 mothers using 16S rRNA gene amplicon sequencing after collecting study samples from three skin areas: face, calf, and ventral forearm. The samples were pooled into 36 groups based on age, gender, and skin site.
“We observed significant differences in alpha diversity and the most prevalent taxa and identified factors that contributed to variation at each site,” the authors reported.
Overall, the “alpha diversity” – a measure of microbial diversity used in microbiome studies – of the skin microbiome increased with age, with the highest alpha diversity seen in the 10-year-olds (n = 28), notably on the face, but differences in alpha diversity between skin sites were seen only in the 1-year-olds (n = 26). Overall, the most commonly identified bacterial phyla at all skin sites in children were Proteobacteria (42%), Firmicutes (25%), Actinobacteria (13%), and Bacteroidetes (11%). In the three sites, the genera with high relative abundance (over 3%) included Streptococcus (13%), Enhydrobacter (6%), and Propionibacterium (5%). Of these, Streptococcus and Granulicatella showed negative linear correlations with age.
The researchers found significant differences between the bacterial communities of 10-year-olds delivered by Cesarean section and those delivered vaginally, particularly in the facial samples; however the difference wasn’t observed among face samples taken from 1-year-olds, according to the authors. They found significant variation in bacteria in calf samples based on whether the children were fed breast milk, formula, or a combination.
When the researchers examined the correlations between mother/child pairs, they found that the relative abundance of most bacteria in the children were more similar to their mothers than to unrelated adults, and they found the strongest correlations for the genera Deinococcus, Microbacterium, Chryseobacterium, Klebsiella, and Enhydrobacter. The relationships between the bacterial communities of mothers and children may be influenced by the shared living environment, topical products, and daily diet, they noted.
The study findings were limited by not controlling for certain variables, including daily diet, choice of topical products, bathing habits, and daily variation in environmental factors, the researchers wrote. However, the results show “that the skin microbiome is strongly affected by the surrounding microenvironment and that the alpha diversity of the skin microbiome increases during childhood,” they concluded.
The study was fully funded by Johnson & Johnson International, and several coauthors are employees of that company. Lead author Ms. Zhu had no financial conflicts to disclose.
SOURCE: Zhu T et al. J Invest Dermatol. 2019 August 13. doi: 10.1016/j.jid.2019.05.018.
Bacteria on children’s skin was similar to their mothers’ and affected by factors that included method of delivery and breastfeeding in a study of 154 children aged 10 years and younger.
Understanding the wrote Ting Zhu of Fudan University, Shanghai, China, and colleagues.
In a study published in the Journal of Investigative Dermatology, the researchers compared the skin microbiota of the 158 children aged 1-10 years and 50 mothers using 16S rRNA gene amplicon sequencing after collecting study samples from three skin areas: face, calf, and ventral forearm. The samples were pooled into 36 groups based on age, gender, and skin site.
“We observed significant differences in alpha diversity and the most prevalent taxa and identified factors that contributed to variation at each site,” the authors reported.
Overall, the “alpha diversity” – a measure of microbial diversity used in microbiome studies – of the skin microbiome increased with age, with the highest alpha diversity seen in the 10-year-olds (n = 28), notably on the face, but differences in alpha diversity between skin sites were seen only in the 1-year-olds (n = 26). Overall, the most commonly identified bacterial phyla at all skin sites in children were Proteobacteria (42%), Firmicutes (25%), Actinobacteria (13%), and Bacteroidetes (11%). In the three sites, the genera with high relative abundance (over 3%) included Streptococcus (13%), Enhydrobacter (6%), and Propionibacterium (5%). Of these, Streptococcus and Granulicatella showed negative linear correlations with age.
The researchers found significant differences between the bacterial communities of 10-year-olds delivered by Cesarean section and those delivered vaginally, particularly in the facial samples; however the difference wasn’t observed among face samples taken from 1-year-olds, according to the authors. They found significant variation in bacteria in calf samples based on whether the children were fed breast milk, formula, or a combination.
When the researchers examined the correlations between mother/child pairs, they found that the relative abundance of most bacteria in the children were more similar to their mothers than to unrelated adults, and they found the strongest correlations for the genera Deinococcus, Microbacterium, Chryseobacterium, Klebsiella, and Enhydrobacter. The relationships between the bacterial communities of mothers and children may be influenced by the shared living environment, topical products, and daily diet, they noted.
The study findings were limited by not controlling for certain variables, including daily diet, choice of topical products, bathing habits, and daily variation in environmental factors, the researchers wrote. However, the results show “that the skin microbiome is strongly affected by the surrounding microenvironment and that the alpha diversity of the skin microbiome increases during childhood,” they concluded.
The study was fully funded by Johnson & Johnson International, and several coauthors are employees of that company. Lead author Ms. Zhu had no financial conflicts to disclose.
SOURCE: Zhu T et al. J Invest Dermatol. 2019 August 13. doi: 10.1016/j.jid.2019.05.018.
Bacteria on children’s skin was similar to their mothers’ and affected by factors that included method of delivery and breastfeeding in a study of 154 children aged 10 years and younger.
Understanding the wrote Ting Zhu of Fudan University, Shanghai, China, and colleagues.
In a study published in the Journal of Investigative Dermatology, the researchers compared the skin microbiota of the 158 children aged 1-10 years and 50 mothers using 16S rRNA gene amplicon sequencing after collecting study samples from three skin areas: face, calf, and ventral forearm. The samples were pooled into 36 groups based on age, gender, and skin site.
“We observed significant differences in alpha diversity and the most prevalent taxa and identified factors that contributed to variation at each site,” the authors reported.
Overall, the “alpha diversity” – a measure of microbial diversity used in microbiome studies – of the skin microbiome increased with age, with the highest alpha diversity seen in the 10-year-olds (n = 28), notably on the face, but differences in alpha diversity between skin sites were seen only in the 1-year-olds (n = 26). Overall, the most commonly identified bacterial phyla at all skin sites in children were Proteobacteria (42%), Firmicutes (25%), Actinobacteria (13%), and Bacteroidetes (11%). In the three sites, the genera with high relative abundance (over 3%) included Streptococcus (13%), Enhydrobacter (6%), and Propionibacterium (5%). Of these, Streptococcus and Granulicatella showed negative linear correlations with age.
The researchers found significant differences between the bacterial communities of 10-year-olds delivered by Cesarean section and those delivered vaginally, particularly in the facial samples; however the difference wasn’t observed among face samples taken from 1-year-olds, according to the authors. They found significant variation in bacteria in calf samples based on whether the children were fed breast milk, formula, or a combination.
When the researchers examined the correlations between mother/child pairs, they found that the relative abundance of most bacteria in the children were more similar to their mothers than to unrelated adults, and they found the strongest correlations for the genera Deinococcus, Microbacterium, Chryseobacterium, Klebsiella, and Enhydrobacter. The relationships between the bacterial communities of mothers and children may be influenced by the shared living environment, topical products, and daily diet, they noted.
The study findings were limited by not controlling for certain variables, including daily diet, choice of topical products, bathing habits, and daily variation in environmental factors, the researchers wrote. However, the results show “that the skin microbiome is strongly affected by the surrounding microenvironment and that the alpha diversity of the skin microbiome increases during childhood,” they concluded.
The study was fully funded by Johnson & Johnson International, and several coauthors are employees of that company. Lead author Ms. Zhu had no financial conflicts to disclose.
SOURCE: Zhu T et al. J Invest Dermatol. 2019 August 13. doi: 10.1016/j.jid.2019.05.018.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Key clinical point: Age, skin site, and maternal factors including delivery method and breastfeeding impact the bacterial makeup of children’s skin.
Major finding: The most common bacteria at all skin sites in children were Proteobacteria (42%), Firmicutes (25%), Actinobacteria (13%), and Bacteroidetes (11%).
Study details: The data come from 158 children aged 10 years and younger and included 474 skin samples.
Disclosures: The study was fully funded by Johnson & Johnson International, and several coauthors are employees of that company. Lead author Ms. Zhu had no financial conflicts to disclose.
Source: Zhu T et al. J Invest Dermatol. 2019 August 13. doi: 10.1016/j.jid.2019.05.018.
Oral semaglutide monotherapy delivers HbA1c improvements in type 2 diabetes
Oral semaglutide monotherapy was superior to placebo for improving glycated hemoglobin (HbA1c) levels at all doses tested in adults with type 2 diabetes who had been previously insufficiently managed with diet and exercise, according to findings from a global, randomized trial.
The drug also showed dose-dependent weight loss, with a statistically significant effect on body weight, compared with placebo, at higher doses.
To date, the glucagon-like peptide–1 receptor agonist has been available as weekly subcutaneous shots for patients with type 2 diabetes, and in that form they have been shown to be effective in reducing HbA1c, inducing weight loss, and lowering the risk of cardiovascular events in patients with cardiovascular disease or those who are at high risk for it, wrote Vanita R. Aroda, MD, of Brigham and Women’s Hospital, Boston, and colleagues. The report is in Diabetes Care.
The novel oral semaglutide tablet is designed to enhance medication absorption, and the pharmacokinetics and dosage were established in phase 2 studies, they noted.
In the phase 3 Peptide Innovation for Early Diabetes Treatment 1 (PIONEER 1) study, Dr. Aroda and colleagues randomized 703 adults with type 2 diabetes to receive either 3 mg, 7 mg, or 14 mg of oral semaglutide daily, or placebo. The average age of the patients was 55 years, about half were women, and the average baseline HbA1c was 8.0% (64 mmol/mol). The primary endpoint was change in HbA1c level from baseline to week 26, and the secondary endpoint was change in body weight over the same period.
After 26 weeks of once-daily treatment, patients in semaglutide group showed significant reductions in HbA1c from baseline with all three doses: –0.6% (3 mg), –0.9% (7 mg), and –1.1% (14 mg), with P less than .001 for all, based on an intention-to-treat analysis. Similar results occurred using an on-treatment analysis, with differences of –0.7%, –1.2%, and –1.4%, respectively, for the three doses.
In addition, patients in all dose groups achieved the secondary endpoint of reduction in body weight, compared with placebo, from baseline to 26 weeks based on both types of analyses. “Significantly more patients achieved body weight loss of at least 5% with oral semaglutide at 7 mg and 14 mg, compared with placebo,” Dr. Aroda and colleagues wrote (intention-to-treat: –0.1 for 3 mg daily [P = .87], –0.9 for 7 mg [P = .09], –2.3 for 14 mg [P less than .001]; and on-treatment: –0.2 for 3 mg [P = .71], –1.0 for 7 mg [P = .01], –2.6 for 14 mg [P less than .001]).
The overall incidence of adverse events and serious adverse events was similar in the treatment and placebo groups, with the most frequent being nausea and diarrhea. No deaths occurred among patients on the medication.
The findings were limited by several factors, including a patient population that had a relatively short duration of diabetes (mean, 3.5 years) and that the oral semaglutide was used as first-line monotherapy, without first using metformin, the researchers noted. However, oral semaglutide “achieved clinically meaningful and superior glucose lowering,” compared with placebo, at all three doses, they wrote.
“Ongoing additional studies in the PIONEER program will further define the effect when used in combination with other glucose-lowering therapies and in other populations of interest, such as those with high cardiovascular risk or renal impairment,” they emphasized
Novo Nordisk funded the study. The lead author disclosed relationships with Novo Nordisk, and several coauthors disclosed relationships with or employment by the company.
SOURCE: Aroda VR et al. Diabetes Care. 2019 Jul. doi: 10.2337/dc19-0749.
Oral semaglutide monotherapy was superior to placebo for improving glycated hemoglobin (HbA1c) levels at all doses tested in adults with type 2 diabetes who had been previously insufficiently managed with diet and exercise, according to findings from a global, randomized trial.
The drug also showed dose-dependent weight loss, with a statistically significant effect on body weight, compared with placebo, at higher doses.
To date, the glucagon-like peptide–1 receptor agonist has been available as weekly subcutaneous shots for patients with type 2 diabetes, and in that form they have been shown to be effective in reducing HbA1c, inducing weight loss, and lowering the risk of cardiovascular events in patients with cardiovascular disease or those who are at high risk for it, wrote Vanita R. Aroda, MD, of Brigham and Women’s Hospital, Boston, and colleagues. The report is in Diabetes Care.
The novel oral semaglutide tablet is designed to enhance medication absorption, and the pharmacokinetics and dosage were established in phase 2 studies, they noted.
In the phase 3 Peptide Innovation for Early Diabetes Treatment 1 (PIONEER 1) study, Dr. Aroda and colleagues randomized 703 adults with type 2 diabetes to receive either 3 mg, 7 mg, or 14 mg of oral semaglutide daily, or placebo. The average age of the patients was 55 years, about half were women, and the average baseline HbA1c was 8.0% (64 mmol/mol). The primary endpoint was change in HbA1c level from baseline to week 26, and the secondary endpoint was change in body weight over the same period.
After 26 weeks of once-daily treatment, patients in semaglutide group showed significant reductions in HbA1c from baseline with all three doses: –0.6% (3 mg), –0.9% (7 mg), and –1.1% (14 mg), with P less than .001 for all, based on an intention-to-treat analysis. Similar results occurred using an on-treatment analysis, with differences of –0.7%, –1.2%, and –1.4%, respectively, for the three doses.
In addition, patients in all dose groups achieved the secondary endpoint of reduction in body weight, compared with placebo, from baseline to 26 weeks based on both types of analyses. “Significantly more patients achieved body weight loss of at least 5% with oral semaglutide at 7 mg and 14 mg, compared with placebo,” Dr. Aroda and colleagues wrote (intention-to-treat: –0.1 for 3 mg daily [P = .87], –0.9 for 7 mg [P = .09], –2.3 for 14 mg [P less than .001]; and on-treatment: –0.2 for 3 mg [P = .71], –1.0 for 7 mg [P = .01], –2.6 for 14 mg [P less than .001]).
The overall incidence of adverse events and serious adverse events was similar in the treatment and placebo groups, with the most frequent being nausea and diarrhea. No deaths occurred among patients on the medication.
The findings were limited by several factors, including a patient population that had a relatively short duration of diabetes (mean, 3.5 years) and that the oral semaglutide was used as first-line monotherapy, without first using metformin, the researchers noted. However, oral semaglutide “achieved clinically meaningful and superior glucose lowering,” compared with placebo, at all three doses, they wrote.
“Ongoing additional studies in the PIONEER program will further define the effect when used in combination with other glucose-lowering therapies and in other populations of interest, such as those with high cardiovascular risk or renal impairment,” they emphasized
Novo Nordisk funded the study. The lead author disclosed relationships with Novo Nordisk, and several coauthors disclosed relationships with or employment by the company.
SOURCE: Aroda VR et al. Diabetes Care. 2019 Jul. doi: 10.2337/dc19-0749.
Oral semaglutide monotherapy was superior to placebo for improving glycated hemoglobin (HbA1c) levels at all doses tested in adults with type 2 diabetes who had been previously insufficiently managed with diet and exercise, according to findings from a global, randomized trial.
The drug also showed dose-dependent weight loss, with a statistically significant effect on body weight, compared with placebo, at higher doses.
To date, the glucagon-like peptide–1 receptor agonist has been available as weekly subcutaneous shots for patients with type 2 diabetes, and in that form they have been shown to be effective in reducing HbA1c, inducing weight loss, and lowering the risk of cardiovascular events in patients with cardiovascular disease or those who are at high risk for it, wrote Vanita R. Aroda, MD, of Brigham and Women’s Hospital, Boston, and colleagues. The report is in Diabetes Care.
The novel oral semaglutide tablet is designed to enhance medication absorption, and the pharmacokinetics and dosage were established in phase 2 studies, they noted.
In the phase 3 Peptide Innovation for Early Diabetes Treatment 1 (PIONEER 1) study, Dr. Aroda and colleagues randomized 703 adults with type 2 diabetes to receive either 3 mg, 7 mg, or 14 mg of oral semaglutide daily, or placebo. The average age of the patients was 55 years, about half were women, and the average baseline HbA1c was 8.0% (64 mmol/mol). The primary endpoint was change in HbA1c level from baseline to week 26, and the secondary endpoint was change in body weight over the same period.
After 26 weeks of once-daily treatment, patients in semaglutide group showed significant reductions in HbA1c from baseline with all three doses: –0.6% (3 mg), –0.9% (7 mg), and –1.1% (14 mg), with P less than .001 for all, based on an intention-to-treat analysis. Similar results occurred using an on-treatment analysis, with differences of –0.7%, –1.2%, and –1.4%, respectively, for the three doses.
In addition, patients in all dose groups achieved the secondary endpoint of reduction in body weight, compared with placebo, from baseline to 26 weeks based on both types of analyses. “Significantly more patients achieved body weight loss of at least 5% with oral semaglutide at 7 mg and 14 mg, compared with placebo,” Dr. Aroda and colleagues wrote (intention-to-treat: –0.1 for 3 mg daily [P = .87], –0.9 for 7 mg [P = .09], –2.3 for 14 mg [P less than .001]; and on-treatment: –0.2 for 3 mg [P = .71], –1.0 for 7 mg [P = .01], –2.6 for 14 mg [P less than .001]).
The overall incidence of adverse events and serious adverse events was similar in the treatment and placebo groups, with the most frequent being nausea and diarrhea. No deaths occurred among patients on the medication.
The findings were limited by several factors, including a patient population that had a relatively short duration of diabetes (mean, 3.5 years) and that the oral semaglutide was used as first-line monotherapy, without first using metformin, the researchers noted. However, oral semaglutide “achieved clinically meaningful and superior glucose lowering,” compared with placebo, at all three doses, they wrote.
“Ongoing additional studies in the PIONEER program will further define the effect when used in combination with other glucose-lowering therapies and in other populations of interest, such as those with high cardiovascular risk or renal impairment,” they emphasized
Novo Nordisk funded the study. The lead author disclosed relationships with Novo Nordisk, and several coauthors disclosed relationships with or employment by the company.
SOURCE: Aroda VR et al. Diabetes Care. 2019 Jul. doi: 10.2337/dc19-0749.
FROM DIABETES CARE
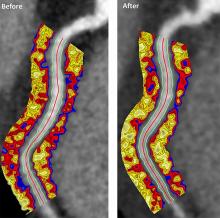
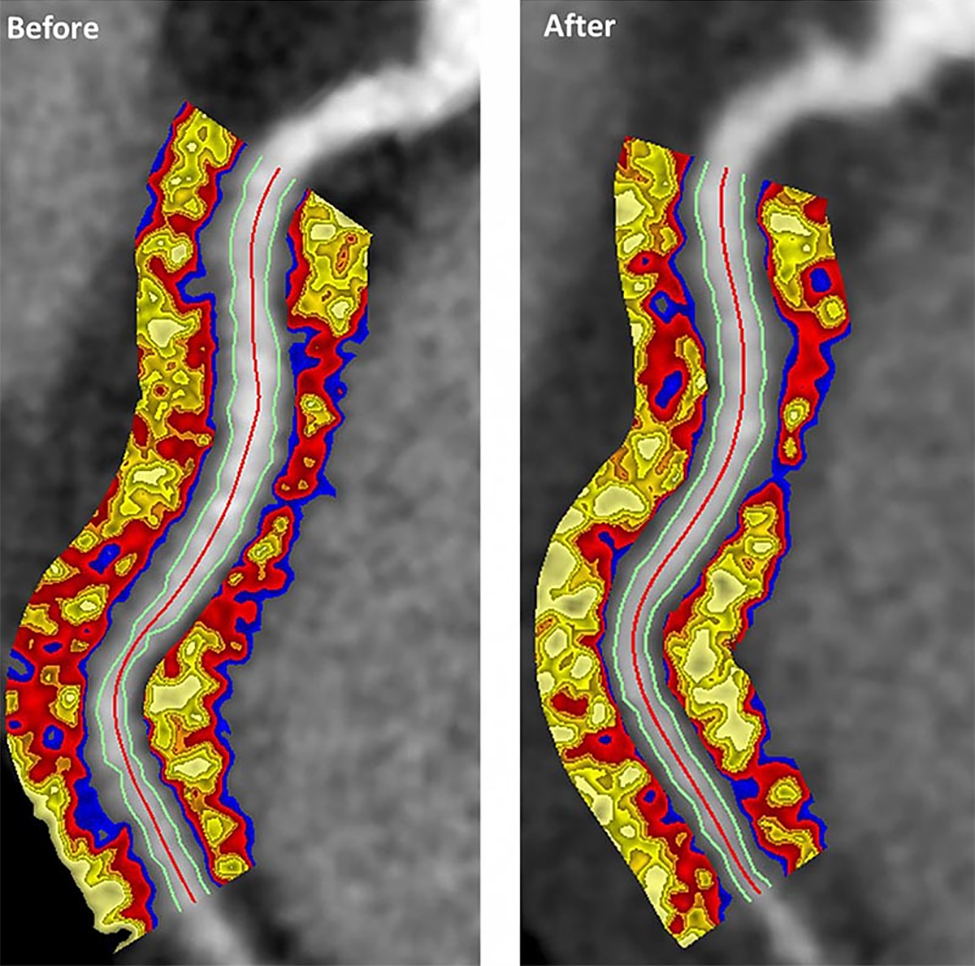
Psoriasis patients on biologics show improved heart health
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
FROM JAMA CARDIOLOGY
Preoperative tramadol fails to improve function after knee surgery
according to findings of a study based on pre- and postsurgery data.
Tramadol has become a popular choice for nonoperative knee pain relief because of its low potential for abuse and favorable safety profile, but its impact on postoperative outcomes when given before knee surgery has not been well studied, wrote Adam Driesman, MD, of the New York University Langone Orthopedic Hospital and colleagues.
In a study published in the Journal of Arthroplasty, the researchers compared patient-reported outcomes (PRO) after total knee arthroplasty among 136 patients who received no opiates, 21 who received tramadol, and 42 who received other opiates. All patients who did not have preoperative and postoperative PRO scores were excluded
All patients received the same multimodal perioperative pain protocol, and all were placed on oxycodone postoperatively for maintenance and breakthrough pain as needed, with discharge prescriptions for acetaminophen/oxycodone combination (Percocet) for breakthrough pain.
Patients preoperative assessment using the Knee Disability and Osteoarthritis Outcome Score Jr. (KOOS, JR.) were similar among the groups prior to surgery; baseline scores for the groups receiving either tramadol, no opiates, or other opiates were 49.95, 50.4, and 48.0, respectively. Demographics also were not significantly different among the groups.
At 3 months, the average KOOS, JR., score for the tramadol group (62.4) was significantly lower, compared with the other-opiate group (67.1) and treatment-naive group (70.1). In addition, patients in the tramadol group had the least change in scores on KOOS, JR., with an average of 12.5 points, compared with 19.1-point and 20.1-point improvements, respectively, in the alternate-opiate group and opiate-naive group.
The data expand on previous findings that patients given preoperative opioids had proportionally less postoperative pain relief than those not on opioids, the researchers said, but noted that they were surprised by the worse outcomes in the tramadol group given its demonstrated side-effect profile.
The study findings were limited by several factors including the retrospective design and relatively short follow-up period, as well as the inability to accurately determine outpatient medication use, not only of opioids, but of nonopioid postoperative pain medications that could have affected the results, the researchers said.
“However, given the conflicting evidence presented in this study and despite the 2013 American Academy of Orthopedic Surgeons Clinical Practice Guidelines, it is recommended providers remain very conservative in their administration of outpatient narcotics including tramadol prior to surgery,” they concluded.
SOURCE: Driesman A et al. J Arthroplasty. 2019;34(8):1662-66.
according to findings of a study based on pre- and postsurgery data.
Tramadol has become a popular choice for nonoperative knee pain relief because of its low potential for abuse and favorable safety profile, but its impact on postoperative outcomes when given before knee surgery has not been well studied, wrote Adam Driesman, MD, of the New York University Langone Orthopedic Hospital and colleagues.
In a study published in the Journal of Arthroplasty, the researchers compared patient-reported outcomes (PRO) after total knee arthroplasty among 136 patients who received no opiates, 21 who received tramadol, and 42 who received other opiates. All patients who did not have preoperative and postoperative PRO scores were excluded
All patients received the same multimodal perioperative pain protocol, and all were placed on oxycodone postoperatively for maintenance and breakthrough pain as needed, with discharge prescriptions for acetaminophen/oxycodone combination (Percocet) for breakthrough pain.
Patients preoperative assessment using the Knee Disability and Osteoarthritis Outcome Score Jr. (KOOS, JR.) were similar among the groups prior to surgery; baseline scores for the groups receiving either tramadol, no opiates, or other opiates were 49.95, 50.4, and 48.0, respectively. Demographics also were not significantly different among the groups.
At 3 months, the average KOOS, JR., score for the tramadol group (62.4) was significantly lower, compared with the other-opiate group (67.1) and treatment-naive group (70.1). In addition, patients in the tramadol group had the least change in scores on KOOS, JR., with an average of 12.5 points, compared with 19.1-point and 20.1-point improvements, respectively, in the alternate-opiate group and opiate-naive group.
The data expand on previous findings that patients given preoperative opioids had proportionally less postoperative pain relief than those not on opioids, the researchers said, but noted that they were surprised by the worse outcomes in the tramadol group given its demonstrated side-effect profile.
The study findings were limited by several factors including the retrospective design and relatively short follow-up period, as well as the inability to accurately determine outpatient medication use, not only of opioids, but of nonopioid postoperative pain medications that could have affected the results, the researchers said.
“However, given the conflicting evidence presented in this study and despite the 2013 American Academy of Orthopedic Surgeons Clinical Practice Guidelines, it is recommended providers remain very conservative in their administration of outpatient narcotics including tramadol prior to surgery,” they concluded.
SOURCE: Driesman A et al. J Arthroplasty. 2019;34(8):1662-66.
according to findings of a study based on pre- and postsurgery data.
Tramadol has become a popular choice for nonoperative knee pain relief because of its low potential for abuse and favorable safety profile, but its impact on postoperative outcomes when given before knee surgery has not been well studied, wrote Adam Driesman, MD, of the New York University Langone Orthopedic Hospital and colleagues.
In a study published in the Journal of Arthroplasty, the researchers compared patient-reported outcomes (PRO) after total knee arthroplasty among 136 patients who received no opiates, 21 who received tramadol, and 42 who received other opiates. All patients who did not have preoperative and postoperative PRO scores were excluded
All patients received the same multimodal perioperative pain protocol, and all were placed on oxycodone postoperatively for maintenance and breakthrough pain as needed, with discharge prescriptions for acetaminophen/oxycodone combination (Percocet) for breakthrough pain.
Patients preoperative assessment using the Knee Disability and Osteoarthritis Outcome Score Jr. (KOOS, JR.) were similar among the groups prior to surgery; baseline scores for the groups receiving either tramadol, no opiates, or other opiates were 49.95, 50.4, and 48.0, respectively. Demographics also were not significantly different among the groups.
At 3 months, the average KOOS, JR., score for the tramadol group (62.4) was significantly lower, compared with the other-opiate group (67.1) and treatment-naive group (70.1). In addition, patients in the tramadol group had the least change in scores on KOOS, JR., with an average of 12.5 points, compared with 19.1-point and 20.1-point improvements, respectively, in the alternate-opiate group and opiate-naive group.
The data expand on previous findings that patients given preoperative opioids had proportionally less postoperative pain relief than those not on opioids, the researchers said, but noted that they were surprised by the worse outcomes in the tramadol group given its demonstrated side-effect profile.
The study findings were limited by several factors including the retrospective design and relatively short follow-up period, as well as the inability to accurately determine outpatient medication use, not only of opioids, but of nonopioid postoperative pain medications that could have affected the results, the researchers said.
“However, given the conflicting evidence presented in this study and despite the 2013 American Academy of Orthopedic Surgeons Clinical Practice Guidelines, it is recommended providers remain very conservative in their administration of outpatient narcotics including tramadol prior to surgery,” they concluded.
SOURCE: Driesman A et al. J Arthroplasty. 2019;34(8):1662-66.
FROM THE JOURNAL OF ARTHROPLASTY
AI technology meets AFib detection
An artificial intelligence-enabled ECG model identified patients with intermittent atrial fibrillation in a 10-second test with 83% accuracy, based on data from more than 180,000 individuals.
“We have previously shown convolution neural networks can evaluate the resting ECG for detection of antiarrhythmic drug levels, abnormal electrolytes levels, and detection of asymptomatic left ventricular dysfunction, providing proof of concept that clinically important phenomena can be detected with artificial intelligence (AI) applications to the ECG,” wrote Zachi I. Attia, an electrical engineer and a primary author of the study, is with the Mayo Clinic, Rochester, Minn., and colleagues.
In a study published in the Lancet, the researchers reviewed data from 649,931 normal sinus rhythm ECGs collected from 180,922 adults between December 1993 and July 2017.
The ECGs were divided into three groups: training (454,789 ECGs from 126,526 patients) internal validation (64,340 ECGs from 18,116 patients) and testing (130,802 ECGs from 36,280 patients). The primary outcome was whether the AI-programmed ECG could identify AFib in a total of 3,051 patients in the testing data set who had verified AFib before being tested with the AI device. The AI-enabled ECG was designed to detect subtle changes using neural network technology previously used by the researchers to identify ventricular dysfunction.
Overall, a single ECG scan identified AFib with an accuracy of 79.4%, an area under the curve (AUC) of 0.87, sensitivity of 79.0%, and specificity of 79.5%. When researchers reviewed multiple ECGs from a 1-month window of either the study start date or 31 days before the first AFib, the accuracy increased to 83.3%, with an AUC of 0.90, sensitivity of 82.3%, and specificity of 83.4%.
The results support the use of subtle changes on normal sinus rhythm ECG to identify patient with potentially undetected AFib, and suggest that AI-enabled ECGs could be used at the point of care to identify patients at risk after unexplained strokes, also known as embolic stroke of undetermined source (ESUS), or heart failure, the researchers noted.
“Although it would require further study, it is possible that this algorithm could identify a high-risk subset of patients with ESUS who could benefit from empirical anticoagulation,” the researchers said.
The study findings were limited by several factors, including possible mislabeling of patients with unidentified atrial fibrillation who were classified negative. In addition, the prevalence of AFib in the study population may be higher than in the general population, they said.
However, the results suggest that use a noninvasive, widely available, point of care test to identify AFib “could have important implications for atrial fibrillation screening and for the management of patients with unexplained stroke,” they concluded.
This study was funded by internal resources of the Mayo Clinic. The researchers had no financial conflicts to disclose.
SOURCE: Attia ZI et al. Lancet. 2019 Aug 1. doi. org/10.1016/S0140-6736(19)31721-0.
This artificial intelligence-enabled ECG interpretation is groundbreaking in creating an algorithm to reveal the likelihood of atrial fibrillation in ECGs showing sinus rhythm.
AFib is now considered a global pandemic and needs to be detected not only to manage the arrhythmia but also to prevent comorbidities and death.
A 10-second, 12-lead ECG in current clinical practice is unlikely to reveal possible AFib if not present in this short monitoring time. However, the findings have clinical importance, particularly in identifying silent AFib and may have important implications for secondary prevention of patients with embolic stroke of undetermined source in terms of providing appropriate oral anticoagulation to prevent recurrences of stroke. The AI-enabled algorithm would require further validation in a different patient cohort, testing a healthier out-of-hospital population, as well as a rigorous prospective clinical trial assessment.
Future research areas include combining ECG algorithms with demographic variables, clinical features, and biomarkers, as well as exploring the use of wearable devices linking these variables and AI for smart monitoring to diagnose AFib.
Jeroen Hendriks, MD, of the University of Adelaide (Australia), and Larissa Fabritz, MD, of the University of Birmingham (England), made these comments in an accompanying editorial. Dr. Hendriks disclosed lecture or consulting fees from Medtronic and Pfizer/Bristol-Myers Squibb. Dr. Fabritz is the inventor of two patents and disclosed research grants and nonfinancial support from European research institutions and Gilead.
This artificial intelligence-enabled ECG interpretation is groundbreaking in creating an algorithm to reveal the likelihood of atrial fibrillation in ECGs showing sinus rhythm.
AFib is now considered a global pandemic and needs to be detected not only to manage the arrhythmia but also to prevent comorbidities and death.
A 10-second, 12-lead ECG in current clinical practice is unlikely to reveal possible AFib if not present in this short monitoring time. However, the findings have clinical importance, particularly in identifying silent AFib and may have important implications for secondary prevention of patients with embolic stroke of undetermined source in terms of providing appropriate oral anticoagulation to prevent recurrences of stroke. The AI-enabled algorithm would require further validation in a different patient cohort, testing a healthier out-of-hospital population, as well as a rigorous prospective clinical trial assessment.
Future research areas include combining ECG algorithms with demographic variables, clinical features, and biomarkers, as well as exploring the use of wearable devices linking these variables and AI for smart monitoring to diagnose AFib.
Jeroen Hendriks, MD, of the University of Adelaide (Australia), and Larissa Fabritz, MD, of the University of Birmingham (England), made these comments in an accompanying editorial. Dr. Hendriks disclosed lecture or consulting fees from Medtronic and Pfizer/Bristol-Myers Squibb. Dr. Fabritz is the inventor of two patents and disclosed research grants and nonfinancial support from European research institutions and Gilead.
This artificial intelligence-enabled ECG interpretation is groundbreaking in creating an algorithm to reveal the likelihood of atrial fibrillation in ECGs showing sinus rhythm.
AFib is now considered a global pandemic and needs to be detected not only to manage the arrhythmia but also to prevent comorbidities and death.
A 10-second, 12-lead ECG in current clinical practice is unlikely to reveal possible AFib if not present in this short monitoring time. However, the findings have clinical importance, particularly in identifying silent AFib and may have important implications for secondary prevention of patients with embolic stroke of undetermined source in terms of providing appropriate oral anticoagulation to prevent recurrences of stroke. The AI-enabled algorithm would require further validation in a different patient cohort, testing a healthier out-of-hospital population, as well as a rigorous prospective clinical trial assessment.
Future research areas include combining ECG algorithms with demographic variables, clinical features, and biomarkers, as well as exploring the use of wearable devices linking these variables and AI for smart monitoring to diagnose AFib.
Jeroen Hendriks, MD, of the University of Adelaide (Australia), and Larissa Fabritz, MD, of the University of Birmingham (England), made these comments in an accompanying editorial. Dr. Hendriks disclosed lecture or consulting fees from Medtronic and Pfizer/Bristol-Myers Squibb. Dr. Fabritz is the inventor of two patents and disclosed research grants and nonfinancial support from European research institutions and Gilead.
An artificial intelligence-enabled ECG model identified patients with intermittent atrial fibrillation in a 10-second test with 83% accuracy, based on data from more than 180,000 individuals.
“We have previously shown convolution neural networks can evaluate the resting ECG for detection of antiarrhythmic drug levels, abnormal electrolytes levels, and detection of asymptomatic left ventricular dysfunction, providing proof of concept that clinically important phenomena can be detected with artificial intelligence (AI) applications to the ECG,” wrote Zachi I. Attia, an electrical engineer and a primary author of the study, is with the Mayo Clinic, Rochester, Minn., and colleagues.
In a study published in the Lancet, the researchers reviewed data from 649,931 normal sinus rhythm ECGs collected from 180,922 adults between December 1993 and July 2017.
The ECGs were divided into three groups: training (454,789 ECGs from 126,526 patients) internal validation (64,340 ECGs from 18,116 patients) and testing (130,802 ECGs from 36,280 patients). The primary outcome was whether the AI-programmed ECG could identify AFib in a total of 3,051 patients in the testing data set who had verified AFib before being tested with the AI device. The AI-enabled ECG was designed to detect subtle changes using neural network technology previously used by the researchers to identify ventricular dysfunction.
Overall, a single ECG scan identified AFib with an accuracy of 79.4%, an area under the curve (AUC) of 0.87, sensitivity of 79.0%, and specificity of 79.5%. When researchers reviewed multiple ECGs from a 1-month window of either the study start date or 31 days before the first AFib, the accuracy increased to 83.3%, with an AUC of 0.90, sensitivity of 82.3%, and specificity of 83.4%.
The results support the use of subtle changes on normal sinus rhythm ECG to identify patient with potentially undetected AFib, and suggest that AI-enabled ECGs could be used at the point of care to identify patients at risk after unexplained strokes, also known as embolic stroke of undetermined source (ESUS), or heart failure, the researchers noted.
“Although it would require further study, it is possible that this algorithm could identify a high-risk subset of patients with ESUS who could benefit from empirical anticoagulation,” the researchers said.
The study findings were limited by several factors, including possible mislabeling of patients with unidentified atrial fibrillation who were classified negative. In addition, the prevalence of AFib in the study population may be higher than in the general population, they said.
However, the results suggest that use a noninvasive, widely available, point of care test to identify AFib “could have important implications for atrial fibrillation screening and for the management of patients with unexplained stroke,” they concluded.
This study was funded by internal resources of the Mayo Clinic. The researchers had no financial conflicts to disclose.
SOURCE: Attia ZI et al. Lancet. 2019 Aug 1. doi. org/10.1016/S0140-6736(19)31721-0.
An artificial intelligence-enabled ECG model identified patients with intermittent atrial fibrillation in a 10-second test with 83% accuracy, based on data from more than 180,000 individuals.
“We have previously shown convolution neural networks can evaluate the resting ECG for detection of antiarrhythmic drug levels, abnormal electrolytes levels, and detection of asymptomatic left ventricular dysfunction, providing proof of concept that clinically important phenomena can be detected with artificial intelligence (AI) applications to the ECG,” wrote Zachi I. Attia, an electrical engineer and a primary author of the study, is with the Mayo Clinic, Rochester, Minn., and colleagues.
In a study published in the Lancet, the researchers reviewed data from 649,931 normal sinus rhythm ECGs collected from 180,922 adults between December 1993 and July 2017.
The ECGs were divided into three groups: training (454,789 ECGs from 126,526 patients) internal validation (64,340 ECGs from 18,116 patients) and testing (130,802 ECGs from 36,280 patients). The primary outcome was whether the AI-programmed ECG could identify AFib in a total of 3,051 patients in the testing data set who had verified AFib before being tested with the AI device. The AI-enabled ECG was designed to detect subtle changes using neural network technology previously used by the researchers to identify ventricular dysfunction.
Overall, a single ECG scan identified AFib with an accuracy of 79.4%, an area under the curve (AUC) of 0.87, sensitivity of 79.0%, and specificity of 79.5%. When researchers reviewed multiple ECGs from a 1-month window of either the study start date or 31 days before the first AFib, the accuracy increased to 83.3%, with an AUC of 0.90, sensitivity of 82.3%, and specificity of 83.4%.
The results support the use of subtle changes on normal sinus rhythm ECG to identify patient with potentially undetected AFib, and suggest that AI-enabled ECGs could be used at the point of care to identify patients at risk after unexplained strokes, also known as embolic stroke of undetermined source (ESUS), or heart failure, the researchers noted.
“Although it would require further study, it is possible that this algorithm could identify a high-risk subset of patients with ESUS who could benefit from empirical anticoagulation,” the researchers said.
The study findings were limited by several factors, including possible mislabeling of patients with unidentified atrial fibrillation who were classified negative. In addition, the prevalence of AFib in the study population may be higher than in the general population, they said.
However, the results suggest that use a noninvasive, widely available, point of care test to identify AFib “could have important implications for atrial fibrillation screening and for the management of patients with unexplained stroke,” they concluded.
This study was funded by internal resources of the Mayo Clinic. The researchers had no financial conflicts to disclose.
SOURCE: Attia ZI et al. Lancet. 2019 Aug 1. doi. org/10.1016/S0140-6736(19)31721-0.
FROM THE LANCET
Endocrine Society advises on diabetes care for older adults
according to a new guideline on diabetes care for older adults issued by the Endocrine Society.
“The prevalence of diabetes in the United States is projected to increase dramatically during the next 3 decades as the population ages, the numbers of higher-risk minority groups increase, and people with diabetes live longer because of decreasing rates of cardiovascular deaths,” wrote Derek LeRoith, MD, of Icahn School of Medicine at Mount Sinai, New York, and his writing committee colleagues. They said their goal was to provide health care providers with guidance for the management of type 1 or type 2 diabetes in older patients, with a focus on simplifying medication regimens and management strategies to avoid “unnecessary and/or harmful adverse effects.”
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, is based mainly on evidence from controlled trials in two systematic reviews that specifically focused on adults aged 65 years and older. The guideline addresses six areas of consideration for this patient population:
- Role of the endocrinologist and diabetes care specialist.
- Screening for diabetes and prediabetes, and diabetes prevention.
- Assessment of older patients with diabetes.
- Treatment of hyperglycemia.
- Treating complications of diabetes.
- Special settings and populations.
Partnerships and screening
The guideline recommends that primary care providers partner with an endocrinologist or diabetes specialist in the care of patients aged 65 and older with newly diagnosed diabetes, and that the specialist take primary responsibility for diabetes care of patients with type 1 diabetes or those who need more complex intervention to achieve treatment goals.
Screening for diabetes in adults aged 65 years and older using fasting plasma glucose and/or hemoglobin A1c should occur every 2 years, but that schedule should be adjusted based on shared decision making with the patient, the committee said. Providers are advised to assess the patient’s overall health and personal values before settling on treatment goals and strategies. The writing group also recommends periodic cognitive screening and that medication regimens be simplified as much as possible.
Tackling hyperglycemia
For treatment of hyperglycemia, the guideline recommends outpatient strategies to minimize hypoglycemia and periodic or continuous glucose monitoring. The strategies include lifestyle modifications as a first-line intervention for ambulatory patients, as well as nutritional assessment. A high-protein diet is recommended for older patients with frailty, but no restrictions on diet are advised for patients who cannot meet glycemic targets with lifestyle modification and who are at risk for malnutrition.
Metformin is the first-choice recommendation for patients with diabetes aged 65 and older who need medical management in addition to lifestyle modification, but it is not recommended for individuals with impaired kidney function or gastrointestinal intolerance, according to the guideline. Oral and injectable drugs and/or insulin are recommended if metformin and lifestyle changes are insufficient to meet glycemic targets, the writers noted.
Managing complications
Hypertension is among the diabetes-related complications that need to be managed in older adults, and the guideline recommends a target blood pressure of 140/90 mm Hg, but other targets – based on patient-provider shared decision making – may be considered for patients in high-risk groups.
The guideline calls for management of hyperlipidemia with statin therapy and “use of an annual lipid profile to achieve the recommended levels for reducing absolute cardiovascular disease events and all-cause mortality.” The committee does not specify low-density lipoprotein cholesterol targets because of insufficient evidence, but recommends alternative treatments, including ezetimibe or proprotein convertase subtilisin/kexin type 9 inhibitors, if statin therapy is not enough to help the patients meet goals. The writers also advocate fish oil and/or fenofibrate for patients with fasting triglycerides of more than 500 mg/dL.
To manage congestive heart failure in older patients with diabetes, the guideline recommends following standard clinical practice guidelines for the condition, and cautious use of oral hypoglycemic agents, including glinides, rosiglitazone, pioglitazone, and dipeptidyl peptidase–4 inhibitors. The writers noted that low-dose aspirin is recommended for patients with diabetes with a history of atherosclerotic cardiovascular disease.
The committee also recommends an annual comprehensive eye exam for patients with diabetes aged 65 years and older to identify retinal disease and suggests that actions, such as physical therapy and reduced use of sedatives, be taken to minimize the risk of falls in patients with neuropathy or problems with balance and gait.
Older patients with diabetes also should be screened annually for chronic kidney disease, and the dosage of diabetes medications should be adjusted to minimize side effects in patients with kidney problems.
Tailoring care to setting
Finally, the guideline addresses special settings and populations, including managing diabetes in hospitals or nursing homes, or in patients who are transitioning to homes or long-term care facilities. Recommendations in this category include simplifying medications for older adults with terminal illness or severe comorbidities, as well as setting glycemic targets as part of a hospital discharge plan.
“The most important aspect of successful transition is effective, detailed, and thorough bidirectional communication between the discharging and receiving teams of health care providers,” the writers emphasized.
The guideline is cosponsored by the European Society of Endocrinology, the Gerontological Society of America, and the Obesity Society. The chair of the committee had no relevant financial conflicts to disclose, and at least 50% of the committee members were free of relevant conflicts of interest.
SOURCE: LeRoith D et al. J Clin Endocrinol Metab. 2019;104:1520-74.
according to a new guideline on diabetes care for older adults issued by the Endocrine Society.
“The prevalence of diabetes in the United States is projected to increase dramatically during the next 3 decades as the population ages, the numbers of higher-risk minority groups increase, and people with diabetes live longer because of decreasing rates of cardiovascular deaths,” wrote Derek LeRoith, MD, of Icahn School of Medicine at Mount Sinai, New York, and his writing committee colleagues. They said their goal was to provide health care providers with guidance for the management of type 1 or type 2 diabetes in older patients, with a focus on simplifying medication regimens and management strategies to avoid “unnecessary and/or harmful adverse effects.”
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, is based mainly on evidence from controlled trials in two systematic reviews that specifically focused on adults aged 65 years and older. The guideline addresses six areas of consideration for this patient population:
- Role of the endocrinologist and diabetes care specialist.
- Screening for diabetes and prediabetes, and diabetes prevention.
- Assessment of older patients with diabetes.
- Treatment of hyperglycemia.
- Treating complications of diabetes.
- Special settings and populations.
Partnerships and screening
The guideline recommends that primary care providers partner with an endocrinologist or diabetes specialist in the care of patients aged 65 and older with newly diagnosed diabetes, and that the specialist take primary responsibility for diabetes care of patients with type 1 diabetes or those who need more complex intervention to achieve treatment goals.
Screening for diabetes in adults aged 65 years and older using fasting plasma glucose and/or hemoglobin A1c should occur every 2 years, but that schedule should be adjusted based on shared decision making with the patient, the committee said. Providers are advised to assess the patient’s overall health and personal values before settling on treatment goals and strategies. The writing group also recommends periodic cognitive screening and that medication regimens be simplified as much as possible.
Tackling hyperglycemia
For treatment of hyperglycemia, the guideline recommends outpatient strategies to minimize hypoglycemia and periodic or continuous glucose monitoring. The strategies include lifestyle modifications as a first-line intervention for ambulatory patients, as well as nutritional assessment. A high-protein diet is recommended for older patients with frailty, but no restrictions on diet are advised for patients who cannot meet glycemic targets with lifestyle modification and who are at risk for malnutrition.
Metformin is the first-choice recommendation for patients with diabetes aged 65 and older who need medical management in addition to lifestyle modification, but it is not recommended for individuals with impaired kidney function or gastrointestinal intolerance, according to the guideline. Oral and injectable drugs and/or insulin are recommended if metformin and lifestyle changes are insufficient to meet glycemic targets, the writers noted.
Managing complications
Hypertension is among the diabetes-related complications that need to be managed in older adults, and the guideline recommends a target blood pressure of 140/90 mm Hg, but other targets – based on patient-provider shared decision making – may be considered for patients in high-risk groups.
The guideline calls for management of hyperlipidemia with statin therapy and “use of an annual lipid profile to achieve the recommended levels for reducing absolute cardiovascular disease events and all-cause mortality.” The committee does not specify low-density lipoprotein cholesterol targets because of insufficient evidence, but recommends alternative treatments, including ezetimibe or proprotein convertase subtilisin/kexin type 9 inhibitors, if statin therapy is not enough to help the patients meet goals. The writers also advocate fish oil and/or fenofibrate for patients with fasting triglycerides of more than 500 mg/dL.
To manage congestive heart failure in older patients with diabetes, the guideline recommends following standard clinical practice guidelines for the condition, and cautious use of oral hypoglycemic agents, including glinides, rosiglitazone, pioglitazone, and dipeptidyl peptidase–4 inhibitors. The writers noted that low-dose aspirin is recommended for patients with diabetes with a history of atherosclerotic cardiovascular disease.
The committee also recommends an annual comprehensive eye exam for patients with diabetes aged 65 years and older to identify retinal disease and suggests that actions, such as physical therapy and reduced use of sedatives, be taken to minimize the risk of falls in patients with neuropathy or problems with balance and gait.
Older patients with diabetes also should be screened annually for chronic kidney disease, and the dosage of diabetes medications should be adjusted to minimize side effects in patients with kidney problems.
Tailoring care to setting
Finally, the guideline addresses special settings and populations, including managing diabetes in hospitals or nursing homes, or in patients who are transitioning to homes or long-term care facilities. Recommendations in this category include simplifying medications for older adults with terminal illness or severe comorbidities, as well as setting glycemic targets as part of a hospital discharge plan.
“The most important aspect of successful transition is effective, detailed, and thorough bidirectional communication between the discharging and receiving teams of health care providers,” the writers emphasized.
The guideline is cosponsored by the European Society of Endocrinology, the Gerontological Society of America, and the Obesity Society. The chair of the committee had no relevant financial conflicts to disclose, and at least 50% of the committee members were free of relevant conflicts of interest.
SOURCE: LeRoith D et al. J Clin Endocrinol Metab. 2019;104:1520-74.
according to a new guideline on diabetes care for older adults issued by the Endocrine Society.
“The prevalence of diabetes in the United States is projected to increase dramatically during the next 3 decades as the population ages, the numbers of higher-risk minority groups increase, and people with diabetes live longer because of decreasing rates of cardiovascular deaths,” wrote Derek LeRoith, MD, of Icahn School of Medicine at Mount Sinai, New York, and his writing committee colleagues. They said their goal was to provide health care providers with guidance for the management of type 1 or type 2 diabetes in older patients, with a focus on simplifying medication regimens and management strategies to avoid “unnecessary and/or harmful adverse effects.”
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, is based mainly on evidence from controlled trials in two systematic reviews that specifically focused on adults aged 65 years and older. The guideline addresses six areas of consideration for this patient population:
- Role of the endocrinologist and diabetes care specialist.
- Screening for diabetes and prediabetes, and diabetes prevention.
- Assessment of older patients with diabetes.
- Treatment of hyperglycemia.
- Treating complications of diabetes.
- Special settings and populations.
Partnerships and screening
The guideline recommends that primary care providers partner with an endocrinologist or diabetes specialist in the care of patients aged 65 and older with newly diagnosed diabetes, and that the specialist take primary responsibility for diabetes care of patients with type 1 diabetes or those who need more complex intervention to achieve treatment goals.
Screening for diabetes in adults aged 65 years and older using fasting plasma glucose and/or hemoglobin A1c should occur every 2 years, but that schedule should be adjusted based on shared decision making with the patient, the committee said. Providers are advised to assess the patient’s overall health and personal values before settling on treatment goals and strategies. The writing group also recommends periodic cognitive screening and that medication regimens be simplified as much as possible.
Tackling hyperglycemia
For treatment of hyperglycemia, the guideline recommends outpatient strategies to minimize hypoglycemia and periodic or continuous glucose monitoring. The strategies include lifestyle modifications as a first-line intervention for ambulatory patients, as well as nutritional assessment. A high-protein diet is recommended for older patients with frailty, but no restrictions on diet are advised for patients who cannot meet glycemic targets with lifestyle modification and who are at risk for malnutrition.
Metformin is the first-choice recommendation for patients with diabetes aged 65 and older who need medical management in addition to lifestyle modification, but it is not recommended for individuals with impaired kidney function or gastrointestinal intolerance, according to the guideline. Oral and injectable drugs and/or insulin are recommended if metformin and lifestyle changes are insufficient to meet glycemic targets, the writers noted.
Managing complications
Hypertension is among the diabetes-related complications that need to be managed in older adults, and the guideline recommends a target blood pressure of 140/90 mm Hg, but other targets – based on patient-provider shared decision making – may be considered for patients in high-risk groups.
The guideline calls for management of hyperlipidemia with statin therapy and “use of an annual lipid profile to achieve the recommended levels for reducing absolute cardiovascular disease events and all-cause mortality.” The committee does not specify low-density lipoprotein cholesterol targets because of insufficient evidence, but recommends alternative treatments, including ezetimibe or proprotein convertase subtilisin/kexin type 9 inhibitors, if statin therapy is not enough to help the patients meet goals. The writers also advocate fish oil and/or fenofibrate for patients with fasting triglycerides of more than 500 mg/dL.
To manage congestive heart failure in older patients with diabetes, the guideline recommends following standard clinical practice guidelines for the condition, and cautious use of oral hypoglycemic agents, including glinides, rosiglitazone, pioglitazone, and dipeptidyl peptidase–4 inhibitors. The writers noted that low-dose aspirin is recommended for patients with diabetes with a history of atherosclerotic cardiovascular disease.
The committee also recommends an annual comprehensive eye exam for patients with diabetes aged 65 years and older to identify retinal disease and suggests that actions, such as physical therapy and reduced use of sedatives, be taken to minimize the risk of falls in patients with neuropathy or problems with balance and gait.
Older patients with diabetes also should be screened annually for chronic kidney disease, and the dosage of diabetes medications should be adjusted to minimize side effects in patients with kidney problems.
Tailoring care to setting
Finally, the guideline addresses special settings and populations, including managing diabetes in hospitals or nursing homes, or in patients who are transitioning to homes or long-term care facilities. Recommendations in this category include simplifying medications for older adults with terminal illness or severe comorbidities, as well as setting glycemic targets as part of a hospital discharge plan.
“The most important aspect of successful transition is effective, detailed, and thorough bidirectional communication between the discharging and receiving teams of health care providers,” the writers emphasized.
The guideline is cosponsored by the European Society of Endocrinology, the Gerontological Society of America, and the Obesity Society. The chair of the committee had no relevant financial conflicts to disclose, and at least 50% of the committee members were free of relevant conflicts of interest.
SOURCE: LeRoith D et al. J Clin Endocrinol Metab. 2019;104:1520-74.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM