User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Safest, most effective medications for spine-related pain in older adults?
, a new comprehensive literature review suggests.
Investigators assessed the evidence for medications used for this indication in older adults by reviewing 138 double-blind, placebo-controlled trials.
Among their key findings and recommendations: Acetaminophen has a favorable safety profile for spine-related pain but nonsteroidal anti-inflammatory drugs (NSAIDs) have greater efficacy.
However, NSAIDs should be used in lower doses in the short term, with gastrointestinal precaution, the researchers note.
Corticosteroids have the least evidence for treating nonspecific back pain, they add.
“Most older people experience neck or low back pain at some point, bothersome enough to see their doctor,” coinvestigator Michael Perloff, MD, PhD, department of neurology, Boston University, said in a news release.
“Our findings provide a helpful medication guide for physicians to use for spine pain in an older population that can have a complex medical history,” Dr. Perloff added.
The results were published online in Drugs and Aging.
Recommendations, warnings
With the graying of the U.S. population, spine-related pain is increasingly common, the investigators note.
Medications play an important role in pain management, but their use has limitations in the elderly, owing to reduced liver and renal function, comorbid medical problems, and polypharmacy.
Other key findings from the literature review include that, although the nerve pain medications gabapentin and pregabalin may cause dizziness or difficulty walking, they also have some demonstrated benefit for neck and back nerve pain, such as sciatica, in older adults.
These agents should be used in lower doses with smaller dose adjustments, the researchers note.
They caution that the muscle relaxants carisoprodol, chlorzoxazone, cyclobenzaprine, metaxalone, methocarbamol, and orphenadrine should be avoided in older adults because of their association with risk for sedation and falls.
‘Rational therapeutic choices’
Three other muscle relaxants – tizanidine, baclofen, and dantrolene – may be helpful for neck and back pain. The most evidence favors tizanidine and baclofen. These should be used in reduced doses. Tizanidine should be avoided in patients with liver disease, and for patients with kidney disease, the dosing of baclofen should be reduced, the investigators write.
Other findings include the following:
- Older tricyclic antidepressants should typically be avoided in this population because of their side effects, but nortriptyline and desipramine may be better tolerated for neck and back nerve pain at lower doses.
- Newer antidepressants, particularly the selective serotonin-norepinephrine reuptake inhibitor duloxetine, have a better safety profile and good efficacy for spine-related nerve pain.
- Traditional opioids are typically avoided in the treatment of spine-related pain in older adults, owing to their associated risks.
However, low-dose opioid therapy may be helpful for severe refractory pain, with close monitoring of patients, the researchers note.
Weaker opioids, such as tramadol, may be better tolerated by older patients. They work well when combined with acetaminophen, but they carry the risk for sedation, upset stomach, and constipation.
“Medications used at the correct dose, for the correct diagnosis, adjusting for preexisting medical problems can result in better use of treatments for spine pain,” coinvestigator Jonathan Fu, MD, also with the department of neurology, Boston University, said in the release.
“Rational therapeutic choices should be targeted to spine pain diagnosis, such as NSAIDs and acetaminophen for arthritic and myofascial-based complaints, gabapentinoids or duloxetine for neuropathic and radicular symptoms, antispastic agents for myofascial-based pain, and combination therapy for mixed etiologies,” the investigators write.
They also emphasize that medications should be coupled with physical therapy and exercise programs, as well as treatment of the underlying degenerative disease process and medical illness, while keeping in mind the need for possible interventions and/or corrective surgery.
The research had no specific funding. The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a new comprehensive literature review suggests.
Investigators assessed the evidence for medications used for this indication in older adults by reviewing 138 double-blind, placebo-controlled trials.
Among their key findings and recommendations: Acetaminophen has a favorable safety profile for spine-related pain but nonsteroidal anti-inflammatory drugs (NSAIDs) have greater efficacy.
However, NSAIDs should be used in lower doses in the short term, with gastrointestinal precaution, the researchers note.
Corticosteroids have the least evidence for treating nonspecific back pain, they add.
“Most older people experience neck or low back pain at some point, bothersome enough to see their doctor,” coinvestigator Michael Perloff, MD, PhD, department of neurology, Boston University, said in a news release.
“Our findings provide a helpful medication guide for physicians to use for spine pain in an older population that can have a complex medical history,” Dr. Perloff added.
The results were published online in Drugs and Aging.
Recommendations, warnings
With the graying of the U.S. population, spine-related pain is increasingly common, the investigators note.
Medications play an important role in pain management, but their use has limitations in the elderly, owing to reduced liver and renal function, comorbid medical problems, and polypharmacy.
Other key findings from the literature review include that, although the nerve pain medications gabapentin and pregabalin may cause dizziness or difficulty walking, they also have some demonstrated benefit for neck and back nerve pain, such as sciatica, in older adults.
These agents should be used in lower doses with smaller dose adjustments, the researchers note.
They caution that the muscle relaxants carisoprodol, chlorzoxazone, cyclobenzaprine, metaxalone, methocarbamol, and orphenadrine should be avoided in older adults because of their association with risk for sedation and falls.
‘Rational therapeutic choices’
Three other muscle relaxants – tizanidine, baclofen, and dantrolene – may be helpful for neck and back pain. The most evidence favors tizanidine and baclofen. These should be used in reduced doses. Tizanidine should be avoided in patients with liver disease, and for patients with kidney disease, the dosing of baclofen should be reduced, the investigators write.
Other findings include the following:
- Older tricyclic antidepressants should typically be avoided in this population because of their side effects, but nortriptyline and desipramine may be better tolerated for neck and back nerve pain at lower doses.
- Newer antidepressants, particularly the selective serotonin-norepinephrine reuptake inhibitor duloxetine, have a better safety profile and good efficacy for spine-related nerve pain.
- Traditional opioids are typically avoided in the treatment of spine-related pain in older adults, owing to their associated risks.
However, low-dose opioid therapy may be helpful for severe refractory pain, with close monitoring of patients, the researchers note.
Weaker opioids, such as tramadol, may be better tolerated by older patients. They work well when combined with acetaminophen, but they carry the risk for sedation, upset stomach, and constipation.
“Medications used at the correct dose, for the correct diagnosis, adjusting for preexisting medical problems can result in better use of treatments for spine pain,” coinvestigator Jonathan Fu, MD, also with the department of neurology, Boston University, said in the release.
“Rational therapeutic choices should be targeted to spine pain diagnosis, such as NSAIDs and acetaminophen for arthritic and myofascial-based complaints, gabapentinoids or duloxetine for neuropathic and radicular symptoms, antispastic agents for myofascial-based pain, and combination therapy for mixed etiologies,” the investigators write.
They also emphasize that medications should be coupled with physical therapy and exercise programs, as well as treatment of the underlying degenerative disease process and medical illness, while keeping in mind the need for possible interventions and/or corrective surgery.
The research had no specific funding. The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a new comprehensive literature review suggests.
Investigators assessed the evidence for medications used for this indication in older adults by reviewing 138 double-blind, placebo-controlled trials.
Among their key findings and recommendations: Acetaminophen has a favorable safety profile for spine-related pain but nonsteroidal anti-inflammatory drugs (NSAIDs) have greater efficacy.
However, NSAIDs should be used in lower doses in the short term, with gastrointestinal precaution, the researchers note.
Corticosteroids have the least evidence for treating nonspecific back pain, they add.
“Most older people experience neck or low back pain at some point, bothersome enough to see their doctor,” coinvestigator Michael Perloff, MD, PhD, department of neurology, Boston University, said in a news release.
“Our findings provide a helpful medication guide for physicians to use for spine pain in an older population that can have a complex medical history,” Dr. Perloff added.
The results were published online in Drugs and Aging.
Recommendations, warnings
With the graying of the U.S. population, spine-related pain is increasingly common, the investigators note.
Medications play an important role in pain management, but their use has limitations in the elderly, owing to reduced liver and renal function, comorbid medical problems, and polypharmacy.
Other key findings from the literature review include that, although the nerve pain medications gabapentin and pregabalin may cause dizziness or difficulty walking, they also have some demonstrated benefit for neck and back nerve pain, such as sciatica, in older adults.
These agents should be used in lower doses with smaller dose adjustments, the researchers note.
They caution that the muscle relaxants carisoprodol, chlorzoxazone, cyclobenzaprine, metaxalone, methocarbamol, and orphenadrine should be avoided in older adults because of their association with risk for sedation and falls.
‘Rational therapeutic choices’
Three other muscle relaxants – tizanidine, baclofen, and dantrolene – may be helpful for neck and back pain. The most evidence favors tizanidine and baclofen. These should be used in reduced doses. Tizanidine should be avoided in patients with liver disease, and for patients with kidney disease, the dosing of baclofen should be reduced, the investigators write.
Other findings include the following:
- Older tricyclic antidepressants should typically be avoided in this population because of their side effects, but nortriptyline and desipramine may be better tolerated for neck and back nerve pain at lower doses.
- Newer antidepressants, particularly the selective serotonin-norepinephrine reuptake inhibitor duloxetine, have a better safety profile and good efficacy for spine-related nerve pain.
- Traditional opioids are typically avoided in the treatment of spine-related pain in older adults, owing to their associated risks.
However, low-dose opioid therapy may be helpful for severe refractory pain, with close monitoring of patients, the researchers note.
Weaker opioids, such as tramadol, may be better tolerated by older patients. They work well when combined with acetaminophen, but they carry the risk for sedation, upset stomach, and constipation.
“Medications used at the correct dose, for the correct diagnosis, adjusting for preexisting medical problems can result in better use of treatments for spine pain,” coinvestigator Jonathan Fu, MD, also with the department of neurology, Boston University, said in the release.
“Rational therapeutic choices should be targeted to spine pain diagnosis, such as NSAIDs and acetaminophen for arthritic and myofascial-based complaints, gabapentinoids or duloxetine for neuropathic and radicular symptoms, antispastic agents for myofascial-based pain, and combination therapy for mixed etiologies,” the investigators write.
They also emphasize that medications should be coupled with physical therapy and exercise programs, as well as treatment of the underlying degenerative disease process and medical illness, while keeping in mind the need for possible interventions and/or corrective surgery.
The research had no specific funding. The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DRUGS AND AGING
An avocado a day doesn’t shrink belly fat, but helps with cholesterol
according to the findings of a new study.
But it did improve diet quality and led to modest lowering of total cholesterol.
More than 1,000 adults with overweight or obesity and a large waist – at least 35 inches in women and 40 inches in men – took part in this U.S. study, called the Habitual Diet and Avocado Trial (HAT).
The people in the study were divided into two groups: usual diet plus one large avocado every day and usual diet with two avocados at most per month (control group).
Those in the avocado-a-day group were given a regular supply of fresh avocados along with written instructions for how to ripen and prepare them.
They had MRI scans to measure belly fat and fat around other organs at the beginning of the study and after 6 months.
After 6 months, the people who ate an avocado a day did not have less fat around their middles – the main trial outcome – compared with people in the control group.
But at 6 months, those in the avocado-a-day group had:
- No weight gain. People’s weight remained stable in both groups.
- Improved diet quality by 8 points on a 100-point scale
- A 2.9-mg/dL decrease in total cholesterol
- A 2.5-mg/dL decrease in LDL cholesterol
The study was done by researchers at Penn State University; Tufts University; Loma Linda University; and the University of California, Los Angeles, with coordinating support from Wake Forest University.
It was published in the Journal of the American Heart Association.
“While the avocados did not affect belly fat or weight gain, the study still provides evidence that avocados can be a beneficial addition to a well-balanced diet,” Penny M. Kris-Etherton, PhD, one of the researchers and a professor of nutritional sciences at Penn State University, University Park, said in a news release.
“Incorporating an avocado per day in this study did not cause weight gain and also caused a slight decrease in LDL cholesterol, which are all important findings for better health,” she said.
Similarly, study researcher Joan Sabaté, MD, a professor at Loma Linda (Calif.) University, said: “While one avocado a day did not lead to clinically significant improvements in abdominal fat and other cardiometabolic risk factors, consuming one avocado a day did not result in body weight gain.”
“This is positive,” he said, “because eating extra calories from avocados doesn’t impact body weight or abdominal fat, and it slightly decreases total and LDL cholesterol.”
Kristina S. Petersen, PhD, another of the researchers and an assistant professor of nutritional sciences at Texas Tech University, Lubbock, pointed out that people are generally poor at adhering to the Dietary Guidelines for Americans.
This study suggests that an avocado a day can improve diet quality, she noted, which “ is important because we know a higher diet quality is associated with lower risk of several diseases, including heart disease, type 2 diabetes, and some cancers.”
But the researchers also stressed that it is important to consider the diet as a whole.
“Consistent with prior observations, a change in dietary patterns rather than a single food or nutrient may be necessary to achieve clinically significant improvements” in belly fat and other risk factors for heart attack, stroke, and diabetes, they wrote.
HAT was funded by the Hass Avocado Board, which also supplied the avocados.
A version of this article first appeared on WebMD.com.
according to the findings of a new study.
But it did improve diet quality and led to modest lowering of total cholesterol.
More than 1,000 adults with overweight or obesity and a large waist – at least 35 inches in women and 40 inches in men – took part in this U.S. study, called the Habitual Diet and Avocado Trial (HAT).
The people in the study were divided into two groups: usual diet plus one large avocado every day and usual diet with two avocados at most per month (control group).
Those in the avocado-a-day group were given a regular supply of fresh avocados along with written instructions for how to ripen and prepare them.
They had MRI scans to measure belly fat and fat around other organs at the beginning of the study and after 6 months.
After 6 months, the people who ate an avocado a day did not have less fat around their middles – the main trial outcome – compared with people in the control group.
But at 6 months, those in the avocado-a-day group had:
- No weight gain. People’s weight remained stable in both groups.
- Improved diet quality by 8 points on a 100-point scale
- A 2.9-mg/dL decrease in total cholesterol
- A 2.5-mg/dL decrease in LDL cholesterol
The study was done by researchers at Penn State University; Tufts University; Loma Linda University; and the University of California, Los Angeles, with coordinating support from Wake Forest University.
It was published in the Journal of the American Heart Association.
“While the avocados did not affect belly fat or weight gain, the study still provides evidence that avocados can be a beneficial addition to a well-balanced diet,” Penny M. Kris-Etherton, PhD, one of the researchers and a professor of nutritional sciences at Penn State University, University Park, said in a news release.
“Incorporating an avocado per day in this study did not cause weight gain and also caused a slight decrease in LDL cholesterol, which are all important findings for better health,” she said.
Similarly, study researcher Joan Sabaté, MD, a professor at Loma Linda (Calif.) University, said: “While one avocado a day did not lead to clinically significant improvements in abdominal fat and other cardiometabolic risk factors, consuming one avocado a day did not result in body weight gain.”
“This is positive,” he said, “because eating extra calories from avocados doesn’t impact body weight or abdominal fat, and it slightly decreases total and LDL cholesterol.”
Kristina S. Petersen, PhD, another of the researchers and an assistant professor of nutritional sciences at Texas Tech University, Lubbock, pointed out that people are generally poor at adhering to the Dietary Guidelines for Americans.
This study suggests that an avocado a day can improve diet quality, she noted, which “ is important because we know a higher diet quality is associated with lower risk of several diseases, including heart disease, type 2 diabetes, and some cancers.”
But the researchers also stressed that it is important to consider the diet as a whole.
“Consistent with prior observations, a change in dietary patterns rather than a single food or nutrient may be necessary to achieve clinically significant improvements” in belly fat and other risk factors for heart attack, stroke, and diabetes, they wrote.
HAT was funded by the Hass Avocado Board, which also supplied the avocados.
A version of this article first appeared on WebMD.com.
according to the findings of a new study.
But it did improve diet quality and led to modest lowering of total cholesterol.
More than 1,000 adults with overweight or obesity and a large waist – at least 35 inches in women and 40 inches in men – took part in this U.S. study, called the Habitual Diet and Avocado Trial (HAT).
The people in the study were divided into two groups: usual diet plus one large avocado every day and usual diet with two avocados at most per month (control group).
Those in the avocado-a-day group were given a regular supply of fresh avocados along with written instructions for how to ripen and prepare them.
They had MRI scans to measure belly fat and fat around other organs at the beginning of the study and after 6 months.
After 6 months, the people who ate an avocado a day did not have less fat around their middles – the main trial outcome – compared with people in the control group.
But at 6 months, those in the avocado-a-day group had:
- No weight gain. People’s weight remained stable in both groups.
- Improved diet quality by 8 points on a 100-point scale
- A 2.9-mg/dL decrease in total cholesterol
- A 2.5-mg/dL decrease in LDL cholesterol
The study was done by researchers at Penn State University; Tufts University; Loma Linda University; and the University of California, Los Angeles, with coordinating support from Wake Forest University.
It was published in the Journal of the American Heart Association.
“While the avocados did not affect belly fat or weight gain, the study still provides evidence that avocados can be a beneficial addition to a well-balanced diet,” Penny M. Kris-Etherton, PhD, one of the researchers and a professor of nutritional sciences at Penn State University, University Park, said in a news release.
“Incorporating an avocado per day in this study did not cause weight gain and also caused a slight decrease in LDL cholesterol, which are all important findings for better health,” she said.
Similarly, study researcher Joan Sabaté, MD, a professor at Loma Linda (Calif.) University, said: “While one avocado a day did not lead to clinically significant improvements in abdominal fat and other cardiometabolic risk factors, consuming one avocado a day did not result in body weight gain.”
“This is positive,” he said, “because eating extra calories from avocados doesn’t impact body weight or abdominal fat, and it slightly decreases total and LDL cholesterol.”
Kristina S. Petersen, PhD, another of the researchers and an assistant professor of nutritional sciences at Texas Tech University, Lubbock, pointed out that people are generally poor at adhering to the Dietary Guidelines for Americans.
This study suggests that an avocado a day can improve diet quality, she noted, which “ is important because we know a higher diet quality is associated with lower risk of several diseases, including heart disease, type 2 diabetes, and some cancers.”
But the researchers also stressed that it is important to consider the diet as a whole.
“Consistent with prior observations, a change in dietary patterns rather than a single food or nutrient may be necessary to achieve clinically significant improvements” in belly fat and other risk factors for heart attack, stroke, and diabetes, they wrote.
HAT was funded by the Hass Avocado Board, which also supplied the avocados.
A version of this article first appeared on WebMD.com.
Pediatric obesity treatment options: Beyond lifestyle modification
Pediatric obesity is a serious problem, not only in the United States but worldwide. Unfortunately, the ongoing COVID-19 pandemic has worsened the epidemic of childhood obesity. Solutions for treating the millions of children and adolescents with obesity are desperately needed because prevention efforts over the past several decades have not been sufficient in slowing the steady rise in obesity prevalence.
Lifestyle modification, including dietary changes, increases in activity, and behavioral modification, are the cornerstone of any obesity treatment, but they alone are not powerful enough to treat obesity by itself in the vast majority of children and adolescents. This is because obesity is not a lifestyle choice; rather, it is a disease, and a disease that has a tremendous amount of biology driving individuals toward weight gain and the propensity toward weight regain if weight is lost.
Fortunately, the tools to treat the underlying biology driving obesity are becoming safer, more effective, and more widely used every year. The two most effective biology-based treatments for pediatric obesity are antiobesity medications and bariatric surgery. These two treatments, when accompanied by lifestyle modification, have the potential to reduce not only body weight but also treat many other risk factors, such as prediabetes, diabetes, high blood pressure, poor cholesterol, liver disease, and sleep apnea, as well as others.
Rise in antiobesity medications
Antiobesity medications are developing at a rapid pace. Seven medications have been approved by the Food and Drug Administration for adults, and three medications (phentermine, orlistat, and liraglutide) are now approved for children and adolescents.
The number of antiobesity medications for use in children and adolescents is expected to expand to five, with semaglutide and phentermine-topiramate (Qsymia) both completing trials in adolescents in 2022. Each of these medications works by treating the biology that drives weight gain, whether it is decreasing impulsivity, reducing hunger and appetite hormone pathways, or improving energy regulation pathways. Weight loss at 1 year for currently FDA-approved medications in adolescents ranges from 3% to 6% on average, depending on the medications. The newer medications already FDA approved in adults that will soon, hopefully, be available in pediatrics result in 10%-16% weight loss on average.
A common parent and patient question regarding antiobesity medications is: “If I start an antiobesity medication, how long will I need to be on it?” The simple answer is: “Probably for the rest of your life.”
This can be a shock to hear, but obesity treatment is very similar to that of hypertension or diabetes. Using high blood pressure as an example: If a patient has high blood pressure (for example, 160/90 mm Hg), they will be prescribed a medication to treat it. Once blood pressure comes down to near-normal levels (for example, 120/80 mm Hg), a dose will be maintained, not removed, because that is the biological mediator keeping the blood pressure low. Removal of the medication would result in blood pressure going back to homeostasis (160/90 mm Hg in our example) in a short period of time).
The same can be said for obesity. For example, if a 16-year-old girl is prescribed liraglutide, a glucagonlike peptide–1 receptor agonist, and loses 10% of her body weight at 1 year, that is great success. Why would we remove the medication that is treating the underlying biology causing successful weight loss?
In short, we would not want to do that. Even if our example patient only maintained that 10% initial weight loss, that would be very successful, just like someone maintaining their low blood pressure. As medications begin to develop at a rapid pace and become more available to pediatric patients, the messaging and conversation around anti-obesity medications must continue to focus on obesity being a biological disease and not a behavior for treatment to be effective and not stigmatized.
Bariatric surgery most effective treatment for pediatric obesity
Currently, the most effective treatment for pediatric obesity is bariatric surgery. The two most commonly used surgical procedures today are the sleeve gastrectomy and gastric bypass. Sleeve gastrectomy works by removing 75%-85% of the stomach and creating a new stomach, called a “sleeve.” Gastric bypass works by separating the stomach into two parts and connecting one part of the new stomach into the intestine.
Both surgeries are very effective at treating obesity in adolescents, with an average weight loss of 30%-35%. Surgery is not just a restrictive means of controlling body weight; it also changes key hormones for appetite and satiety that signal the brain. In fact, many of the same biological signals that are changed by surgery are the same signals being targeted by antiobesity medications. Long-term outcome of bariatric surgery in adolescents, provided by Teen-LABS, show it to be safe and maybe even more effective than in adults for treating diabetes and hypertension, with similar weight loss.
Does treatment outweigh the potential risks?
Although obesity surgery and antiobesity medications are more successful at treating obesity in children and adolescents than lifestyle medications, they do have some risks. Surgery, depending on the type of surgery, can cause nutritional deficiencies, reduce body mineral density, and is a life-changing medical procedure. Antiobesity medications, depending on the type, can cause nausea and vomiting and increase heart rate – and because they are relatively new, we do not fully understand the long-term impact of continued use past 1 year.
However, an important question to ask is: “Do the risks of obesity surgery and antiobesity medications outweigh the risk of having lifelong obesity?” The answer to me and many of my colleagues is: “Yes!” Although there are risks associated with the two best treatments for pediatric obesity, those risks under proper supervision of a medical professional far outweigh the risks of not properly treating obesity and allowing it to persist and get worse over many years to come. Obesity is a disease deeply rooted in biology, and we must use biology-based treatments to tackle this problem in children and adolescents, who deserve the best care and treatments possible.
Dr. Ryder is assistant professor of pediatrics and associate director of research, Center for Pediatric Obesity Medicine, at the University of Minnesota, Minneapolis. She reported receiving donations for clinical trials from Boehringer Ingelheim. A version of this article first appeared on Medscape.com.
Pediatric obesity is a serious problem, not only in the United States but worldwide. Unfortunately, the ongoing COVID-19 pandemic has worsened the epidemic of childhood obesity. Solutions for treating the millions of children and adolescents with obesity are desperately needed because prevention efforts over the past several decades have not been sufficient in slowing the steady rise in obesity prevalence.
Lifestyle modification, including dietary changes, increases in activity, and behavioral modification, are the cornerstone of any obesity treatment, but they alone are not powerful enough to treat obesity by itself in the vast majority of children and adolescents. This is because obesity is not a lifestyle choice; rather, it is a disease, and a disease that has a tremendous amount of biology driving individuals toward weight gain and the propensity toward weight regain if weight is lost.
Fortunately, the tools to treat the underlying biology driving obesity are becoming safer, more effective, and more widely used every year. The two most effective biology-based treatments for pediatric obesity are antiobesity medications and bariatric surgery. These two treatments, when accompanied by lifestyle modification, have the potential to reduce not only body weight but also treat many other risk factors, such as prediabetes, diabetes, high blood pressure, poor cholesterol, liver disease, and sleep apnea, as well as others.
Rise in antiobesity medications
Antiobesity medications are developing at a rapid pace. Seven medications have been approved by the Food and Drug Administration for adults, and three medications (phentermine, orlistat, and liraglutide) are now approved for children and adolescents.
The number of antiobesity medications for use in children and adolescents is expected to expand to five, with semaglutide and phentermine-topiramate (Qsymia) both completing trials in adolescents in 2022. Each of these medications works by treating the biology that drives weight gain, whether it is decreasing impulsivity, reducing hunger and appetite hormone pathways, or improving energy regulation pathways. Weight loss at 1 year for currently FDA-approved medications in adolescents ranges from 3% to 6% on average, depending on the medications. The newer medications already FDA approved in adults that will soon, hopefully, be available in pediatrics result in 10%-16% weight loss on average.
A common parent and patient question regarding antiobesity medications is: “If I start an antiobesity medication, how long will I need to be on it?” The simple answer is: “Probably for the rest of your life.”
This can be a shock to hear, but obesity treatment is very similar to that of hypertension or diabetes. Using high blood pressure as an example: If a patient has high blood pressure (for example, 160/90 mm Hg), they will be prescribed a medication to treat it. Once blood pressure comes down to near-normal levels (for example, 120/80 mm Hg), a dose will be maintained, not removed, because that is the biological mediator keeping the blood pressure low. Removal of the medication would result in blood pressure going back to homeostasis (160/90 mm Hg in our example) in a short period of time).
The same can be said for obesity. For example, if a 16-year-old girl is prescribed liraglutide, a glucagonlike peptide–1 receptor agonist, and loses 10% of her body weight at 1 year, that is great success. Why would we remove the medication that is treating the underlying biology causing successful weight loss?
In short, we would not want to do that. Even if our example patient only maintained that 10% initial weight loss, that would be very successful, just like someone maintaining their low blood pressure. As medications begin to develop at a rapid pace and become more available to pediatric patients, the messaging and conversation around anti-obesity medications must continue to focus on obesity being a biological disease and not a behavior for treatment to be effective and not stigmatized.
Bariatric surgery most effective treatment for pediatric obesity
Currently, the most effective treatment for pediatric obesity is bariatric surgery. The two most commonly used surgical procedures today are the sleeve gastrectomy and gastric bypass. Sleeve gastrectomy works by removing 75%-85% of the stomach and creating a new stomach, called a “sleeve.” Gastric bypass works by separating the stomach into two parts and connecting one part of the new stomach into the intestine.
Both surgeries are very effective at treating obesity in adolescents, with an average weight loss of 30%-35%. Surgery is not just a restrictive means of controlling body weight; it also changes key hormones for appetite and satiety that signal the brain. In fact, many of the same biological signals that are changed by surgery are the same signals being targeted by antiobesity medications. Long-term outcome of bariatric surgery in adolescents, provided by Teen-LABS, show it to be safe and maybe even more effective than in adults for treating diabetes and hypertension, with similar weight loss.
Does treatment outweigh the potential risks?
Although obesity surgery and antiobesity medications are more successful at treating obesity in children and adolescents than lifestyle medications, they do have some risks. Surgery, depending on the type of surgery, can cause nutritional deficiencies, reduce body mineral density, and is a life-changing medical procedure. Antiobesity medications, depending on the type, can cause nausea and vomiting and increase heart rate – and because they are relatively new, we do not fully understand the long-term impact of continued use past 1 year.
However, an important question to ask is: “Do the risks of obesity surgery and antiobesity medications outweigh the risk of having lifelong obesity?” The answer to me and many of my colleagues is: “Yes!” Although there are risks associated with the two best treatments for pediatric obesity, those risks under proper supervision of a medical professional far outweigh the risks of not properly treating obesity and allowing it to persist and get worse over many years to come. Obesity is a disease deeply rooted in biology, and we must use biology-based treatments to tackle this problem in children and adolescents, who deserve the best care and treatments possible.
Dr. Ryder is assistant professor of pediatrics and associate director of research, Center for Pediatric Obesity Medicine, at the University of Minnesota, Minneapolis. She reported receiving donations for clinical trials from Boehringer Ingelheim. A version of this article first appeared on Medscape.com.
Pediatric obesity is a serious problem, not only in the United States but worldwide. Unfortunately, the ongoing COVID-19 pandemic has worsened the epidemic of childhood obesity. Solutions for treating the millions of children and adolescents with obesity are desperately needed because prevention efforts over the past several decades have not been sufficient in slowing the steady rise in obesity prevalence.
Lifestyle modification, including dietary changes, increases in activity, and behavioral modification, are the cornerstone of any obesity treatment, but they alone are not powerful enough to treat obesity by itself in the vast majority of children and adolescents. This is because obesity is not a lifestyle choice; rather, it is a disease, and a disease that has a tremendous amount of biology driving individuals toward weight gain and the propensity toward weight regain if weight is lost.
Fortunately, the tools to treat the underlying biology driving obesity are becoming safer, more effective, and more widely used every year. The two most effective biology-based treatments for pediatric obesity are antiobesity medications and bariatric surgery. These two treatments, when accompanied by lifestyle modification, have the potential to reduce not only body weight but also treat many other risk factors, such as prediabetes, diabetes, high blood pressure, poor cholesterol, liver disease, and sleep apnea, as well as others.
Rise in antiobesity medications
Antiobesity medications are developing at a rapid pace. Seven medications have been approved by the Food and Drug Administration for adults, and three medications (phentermine, orlistat, and liraglutide) are now approved for children and adolescents.
The number of antiobesity medications for use in children and adolescents is expected to expand to five, with semaglutide and phentermine-topiramate (Qsymia) both completing trials in adolescents in 2022. Each of these medications works by treating the biology that drives weight gain, whether it is decreasing impulsivity, reducing hunger and appetite hormone pathways, or improving energy regulation pathways. Weight loss at 1 year for currently FDA-approved medications in adolescents ranges from 3% to 6% on average, depending on the medications. The newer medications already FDA approved in adults that will soon, hopefully, be available in pediatrics result in 10%-16% weight loss on average.
A common parent and patient question regarding antiobesity medications is: “If I start an antiobesity medication, how long will I need to be on it?” The simple answer is: “Probably for the rest of your life.”
This can be a shock to hear, but obesity treatment is very similar to that of hypertension or diabetes. Using high blood pressure as an example: If a patient has high blood pressure (for example, 160/90 mm Hg), they will be prescribed a medication to treat it. Once blood pressure comes down to near-normal levels (for example, 120/80 mm Hg), a dose will be maintained, not removed, because that is the biological mediator keeping the blood pressure low. Removal of the medication would result in blood pressure going back to homeostasis (160/90 mm Hg in our example) in a short period of time).
The same can be said for obesity. For example, if a 16-year-old girl is prescribed liraglutide, a glucagonlike peptide–1 receptor agonist, and loses 10% of her body weight at 1 year, that is great success. Why would we remove the medication that is treating the underlying biology causing successful weight loss?
In short, we would not want to do that. Even if our example patient only maintained that 10% initial weight loss, that would be very successful, just like someone maintaining their low blood pressure. As medications begin to develop at a rapid pace and become more available to pediatric patients, the messaging and conversation around anti-obesity medications must continue to focus on obesity being a biological disease and not a behavior for treatment to be effective and not stigmatized.
Bariatric surgery most effective treatment for pediatric obesity
Currently, the most effective treatment for pediatric obesity is bariatric surgery. The two most commonly used surgical procedures today are the sleeve gastrectomy and gastric bypass. Sleeve gastrectomy works by removing 75%-85% of the stomach and creating a new stomach, called a “sleeve.” Gastric bypass works by separating the stomach into two parts and connecting one part of the new stomach into the intestine.
Both surgeries are very effective at treating obesity in adolescents, with an average weight loss of 30%-35%. Surgery is not just a restrictive means of controlling body weight; it also changes key hormones for appetite and satiety that signal the brain. In fact, many of the same biological signals that are changed by surgery are the same signals being targeted by antiobesity medications. Long-term outcome of bariatric surgery in adolescents, provided by Teen-LABS, show it to be safe and maybe even more effective than in adults for treating diabetes and hypertension, with similar weight loss.
Does treatment outweigh the potential risks?
Although obesity surgery and antiobesity medications are more successful at treating obesity in children and adolescents than lifestyle medications, they do have some risks. Surgery, depending on the type of surgery, can cause nutritional deficiencies, reduce body mineral density, and is a life-changing medical procedure. Antiobesity medications, depending on the type, can cause nausea and vomiting and increase heart rate – and because they are relatively new, we do not fully understand the long-term impact of continued use past 1 year.
However, an important question to ask is: “Do the risks of obesity surgery and antiobesity medications outweigh the risk of having lifelong obesity?” The answer to me and many of my colleagues is: “Yes!” Although there are risks associated with the two best treatments for pediatric obesity, those risks under proper supervision of a medical professional far outweigh the risks of not properly treating obesity and allowing it to persist and get worse over many years to come. Obesity is a disease deeply rooted in biology, and we must use biology-based treatments to tackle this problem in children and adolescents, who deserve the best care and treatments possible.
Dr. Ryder is assistant professor of pediatrics and associate director of research, Center for Pediatric Obesity Medicine, at the University of Minnesota, Minneapolis. She reported receiving donations for clinical trials from Boehringer Ingelheim. A version of this article first appeared on Medscape.com.
PCOS in mothers tied to health problems in children
Children whose mothers have polycystic ovary syndrome (PCOS) have increased rates of hospitalization for various conditions, including asthma, pneumonia, and ear infection, a study of more than 1 million children shows.
The associations were not particularly strong, according to the researchers. But they raise questions about the reasons for the increased risk and whether interventions such as diet, exercise, or medications could lead to healthier outcomes for children whose mothers have PCOS.
“The findings suggest that maternal PCOS may have a negative impact on offspring development, enough to lead to a measurable increase in the risk of childhood hospitalization,” study coauthor Nathalie Auger, MD, associate professor of epidemiology at University of Montreal, and colleagues reported in Human Reproduction.
“They are minor differences, just enough that we can statistically identify them. They’re not something where everyone should be worrying at this point,” Dr. Auger told this news organization.
Still, some of the hospitalizations, such as those related to infection or allergy, could be prevented with earlier ambulatory care, so some degree of greater awareness among parents and clinicians may be warranted, she said.
Thirteen years of follow-up
PCOS – a reproductive disorder characterized by irregular periods, increased male hormones, and metabolic complications – affects some 10% of women. People with the condition are at increased risk for obesity, type 2 diabetes, and cardiovascular disease.
Although prior research has shown that maternal PCOS may be associated with higher body mass index and attention deficit disorder in children, data on long-term childhood health outcomes have been limited, Dr. Auger’s group noted.
To examine illness in children exposed to maternal PCOS, the investigators analyzed hospitalization rates for nearly 1.04 million children in Quebec between 2006 and 2020; 7,160 of the children had mothers with PCOS.
In all, 275,354 children were hospitalized during 13 years of follow-up, including 2,314 whose mothers had PCOS.
Children exposed to PCOS were hospitalized at a rate of 68.9 per 1,000 person-years – roughly 50% more often than the rate of 45.3 per 1,000 person-years for children not exposed to maternal PCOS.
In an analysis that adjusted for maternal characteristics, childhood hospitalization for any reason was 1.32 times more likely for children exposed to maternal PCOS.
Hospitalizations linked to infectious diseases – such as for bronchitis, bronchiolitis, pneumonia, nephritis, otitis media, or meningitis – were 1.31 times more likely among children exposed to PCOS. Allergy-related hospitalizations, such as for allergic asthma and anaphylaxis, were 1.47 times more likely, according to the researchers.
Metabolic hospitalizations were 1.59 times more likely. For gastrointestinal hospitalizations, the hazard ratio was 1.72. For central nervous system hospitalizations, it was 1.74.
The associations were stronger in earlier childhood, and results were similar for boys and girls, the investigators reported.
Hospitalizations for cardiovascular disease, musculoskeletal conditions, or malignancy were not increased.
‘Surprising’ links
“The findings are surprising in that some of the conditions that they showed increased risk for, like asthma and some infections, are not conditions that we think of as being typically associated with PCOS,” said Andrea E. Dunaif, MD, chief of the Hilda and J. Lester Gabrilove Division of Endocrinology, Diabetes, and Bone Disease at Mount Sinai Health System, New York, who was not part of the study team.
Earlier studies of offspring of women with PCOS have suggested that children may be at increased risk for insulin resistance and obesity.
Differences in genetics, intrauterine environments, patterns of health care use by women with PCOS, and behavioral factors, such as diet and how children are raised, are variables that could have contributed to the different hospitalization rates among children exposed to maternal PCOS, Dr. Auger said.
“Everything is interconnected,” she said.
The study was supported by a grant from the Canadian Institutes of Health Research. Dr. Auger has received a career award from Fonds de Recherche du Québec-Santé. Dr. Dunaif has consulted for Novo Nordisk and Fractyl Laboratories (now Fractyl Health).
A version of this article first appeared on Medscape.com.
Children whose mothers have polycystic ovary syndrome (PCOS) have increased rates of hospitalization for various conditions, including asthma, pneumonia, and ear infection, a study of more than 1 million children shows.
The associations were not particularly strong, according to the researchers. But they raise questions about the reasons for the increased risk and whether interventions such as diet, exercise, or medications could lead to healthier outcomes for children whose mothers have PCOS.
“The findings suggest that maternal PCOS may have a negative impact on offspring development, enough to lead to a measurable increase in the risk of childhood hospitalization,” study coauthor Nathalie Auger, MD, associate professor of epidemiology at University of Montreal, and colleagues reported in Human Reproduction.
“They are minor differences, just enough that we can statistically identify them. They’re not something where everyone should be worrying at this point,” Dr. Auger told this news organization.
Still, some of the hospitalizations, such as those related to infection or allergy, could be prevented with earlier ambulatory care, so some degree of greater awareness among parents and clinicians may be warranted, she said.
Thirteen years of follow-up
PCOS – a reproductive disorder characterized by irregular periods, increased male hormones, and metabolic complications – affects some 10% of women. People with the condition are at increased risk for obesity, type 2 diabetes, and cardiovascular disease.
Although prior research has shown that maternal PCOS may be associated with higher body mass index and attention deficit disorder in children, data on long-term childhood health outcomes have been limited, Dr. Auger’s group noted.
To examine illness in children exposed to maternal PCOS, the investigators analyzed hospitalization rates for nearly 1.04 million children in Quebec between 2006 and 2020; 7,160 of the children had mothers with PCOS.
In all, 275,354 children were hospitalized during 13 years of follow-up, including 2,314 whose mothers had PCOS.
Children exposed to PCOS were hospitalized at a rate of 68.9 per 1,000 person-years – roughly 50% more often than the rate of 45.3 per 1,000 person-years for children not exposed to maternal PCOS.
In an analysis that adjusted for maternal characteristics, childhood hospitalization for any reason was 1.32 times more likely for children exposed to maternal PCOS.
Hospitalizations linked to infectious diseases – such as for bronchitis, bronchiolitis, pneumonia, nephritis, otitis media, or meningitis – were 1.31 times more likely among children exposed to PCOS. Allergy-related hospitalizations, such as for allergic asthma and anaphylaxis, were 1.47 times more likely, according to the researchers.
Metabolic hospitalizations were 1.59 times more likely. For gastrointestinal hospitalizations, the hazard ratio was 1.72. For central nervous system hospitalizations, it was 1.74.
The associations were stronger in earlier childhood, and results were similar for boys and girls, the investigators reported.
Hospitalizations for cardiovascular disease, musculoskeletal conditions, or malignancy were not increased.
‘Surprising’ links
“The findings are surprising in that some of the conditions that they showed increased risk for, like asthma and some infections, are not conditions that we think of as being typically associated with PCOS,” said Andrea E. Dunaif, MD, chief of the Hilda and J. Lester Gabrilove Division of Endocrinology, Diabetes, and Bone Disease at Mount Sinai Health System, New York, who was not part of the study team.
Earlier studies of offspring of women with PCOS have suggested that children may be at increased risk for insulin resistance and obesity.
Differences in genetics, intrauterine environments, patterns of health care use by women with PCOS, and behavioral factors, such as diet and how children are raised, are variables that could have contributed to the different hospitalization rates among children exposed to maternal PCOS, Dr. Auger said.
“Everything is interconnected,” she said.
The study was supported by a grant from the Canadian Institutes of Health Research. Dr. Auger has received a career award from Fonds de Recherche du Québec-Santé. Dr. Dunaif has consulted for Novo Nordisk and Fractyl Laboratories (now Fractyl Health).
A version of this article first appeared on Medscape.com.
Children whose mothers have polycystic ovary syndrome (PCOS) have increased rates of hospitalization for various conditions, including asthma, pneumonia, and ear infection, a study of more than 1 million children shows.
The associations were not particularly strong, according to the researchers. But they raise questions about the reasons for the increased risk and whether interventions such as diet, exercise, or medications could lead to healthier outcomes for children whose mothers have PCOS.
“The findings suggest that maternal PCOS may have a negative impact on offspring development, enough to lead to a measurable increase in the risk of childhood hospitalization,” study coauthor Nathalie Auger, MD, associate professor of epidemiology at University of Montreal, and colleagues reported in Human Reproduction.
“They are minor differences, just enough that we can statistically identify them. They’re not something where everyone should be worrying at this point,” Dr. Auger told this news organization.
Still, some of the hospitalizations, such as those related to infection or allergy, could be prevented with earlier ambulatory care, so some degree of greater awareness among parents and clinicians may be warranted, she said.
Thirteen years of follow-up
PCOS – a reproductive disorder characterized by irregular periods, increased male hormones, and metabolic complications – affects some 10% of women. People with the condition are at increased risk for obesity, type 2 diabetes, and cardiovascular disease.
Although prior research has shown that maternal PCOS may be associated with higher body mass index and attention deficit disorder in children, data on long-term childhood health outcomes have been limited, Dr. Auger’s group noted.
To examine illness in children exposed to maternal PCOS, the investigators analyzed hospitalization rates for nearly 1.04 million children in Quebec between 2006 and 2020; 7,160 of the children had mothers with PCOS.
In all, 275,354 children were hospitalized during 13 years of follow-up, including 2,314 whose mothers had PCOS.
Children exposed to PCOS were hospitalized at a rate of 68.9 per 1,000 person-years – roughly 50% more often than the rate of 45.3 per 1,000 person-years for children not exposed to maternal PCOS.
In an analysis that adjusted for maternal characteristics, childhood hospitalization for any reason was 1.32 times more likely for children exposed to maternal PCOS.
Hospitalizations linked to infectious diseases – such as for bronchitis, bronchiolitis, pneumonia, nephritis, otitis media, or meningitis – were 1.31 times more likely among children exposed to PCOS. Allergy-related hospitalizations, such as for allergic asthma and anaphylaxis, were 1.47 times more likely, according to the researchers.
Metabolic hospitalizations were 1.59 times more likely. For gastrointestinal hospitalizations, the hazard ratio was 1.72. For central nervous system hospitalizations, it was 1.74.
The associations were stronger in earlier childhood, and results were similar for boys and girls, the investigators reported.
Hospitalizations for cardiovascular disease, musculoskeletal conditions, or malignancy were not increased.
‘Surprising’ links
“The findings are surprising in that some of the conditions that they showed increased risk for, like asthma and some infections, are not conditions that we think of as being typically associated with PCOS,” said Andrea E. Dunaif, MD, chief of the Hilda and J. Lester Gabrilove Division of Endocrinology, Diabetes, and Bone Disease at Mount Sinai Health System, New York, who was not part of the study team.
Earlier studies of offspring of women with PCOS have suggested that children may be at increased risk for insulin resistance and obesity.
Differences in genetics, intrauterine environments, patterns of health care use by women with PCOS, and behavioral factors, such as diet and how children are raised, are variables that could have contributed to the different hospitalization rates among children exposed to maternal PCOS, Dr. Auger said.
“Everything is interconnected,” she said.
The study was supported by a grant from the Canadian Institutes of Health Research. Dr. Auger has received a career award from Fonds de Recherche du Québec-Santé. Dr. Dunaif has consulted for Novo Nordisk and Fractyl Laboratories (now Fractyl Health).
A version of this article first appeared on Medscape.com.
FROM HUMAN REPRODUCTION
Pre-endoscopy COVID-19 testing may not be needed
Pre-endoscopy viral testing may not be necessary to prevent coronavirus transmission from patients to endoscopy staff members, according to a new study published in Gut.
Instead, using personal protective equipment (PPE) and ensuring up-to-date COVID-19 vaccination among the medical team was found to be enough to substantially reduce the risk of spreading SARS-CoV-2, wrote Alexander Hann, Dr.med., gastroenterologist at University Hospital W
“We suggest that pre-selection of patients using respective questionnaires, vaccination, and particularly PPE appears to be sufficient for the prevention of SARS-CoV-2 transmission in GI endoscopy,” they wrote.
Dr. Hann and colleagues analyzed 15,750 endoscopies performed by 29 staff members during the period between May 2020 and December 2021. The researchers looked at three test approaches: No testing (4,543 patients), rapid antigen testing (682 patients), and real-time PCR testing (10,465 patients). In addition, 60 endoscopies were performed in patients with known COVID-19. Overall, no staff members became infected with SARS-CoV-2 during the study period. In all three scenarios, staff used PPE, and the vaccination rate of the team was 97%.
University Hospital W
All patients were interviewed before admission for COVID-19 symptoms, close contact with infected people, and recent travel to high-risk countries. Moreover, some endoscopies were performed even if a patient had positive markers for COVID-19.
The clinical team wore recommended PPE, including a high-filter FFP2 mask, one pair of gloves, protective eyewear, and disposable gowns. For patients with known COVID-19, staff wore two pairs of gloves, a disposable hairnet, and a water-resistant disposable gown. In addition, endoscopies were performed in negative pressure intervention rooms.
Among the 29 staff members involved, 16 physicians and 13 assistants worked in the endoscopy unit for at least 2 days per week for at least 6 months. The hospital’s internal policy required medical staff to undergo PCR testing if a rapid antigen test was positive or symptoms developed. Staff were vaccinated with two doses of the Pfizer-BioNTech vaccine in January and February 2021. A single booster dose of the Pfizer or Moderna vaccine was administered in November and December 2021.
The clinical team was not tested routinely, so asymptomatic infections may have existed. Moreover, the relatively low COVID-19 incidence in the local area might have influenced the risk of transmission. “However, even at the end of 2021, when the incidence was increasing, we did not see any higher risk of transmission,” the researchers explained.
“An important limitation of our study relates to the new variant Omicron that was dominant in our local area after the analyzed time frame.” Additional studies may be needed to understand the risk of transmission with the latest Omicron variants, and given the additional costs and implications on routine activity, current testing guidelines may need to be reconsidered.
“Although our data were not part of a randomized prospective study, we were able to demonstrate on a fairly high number of patients that PPE measures in addition to a short interview for assessment of a patient’s individual risks appear to be highly effective to control transmission of SARS-CoV-2 during an endoscopy. ... Pre-procedural RT-PCR testing or RA testing did not show any additional benefit,” Dr. Hann and colleagues concluded.
The authors reported no conflicts of interest.
Pre-endoscopy viral testing may not be necessary to prevent coronavirus transmission from patients to endoscopy staff members, according to a new study published in Gut.
Instead, using personal protective equipment (PPE) and ensuring up-to-date COVID-19 vaccination among the medical team was found to be enough to substantially reduce the risk of spreading SARS-CoV-2, wrote Alexander Hann, Dr.med., gastroenterologist at University Hospital W
“We suggest that pre-selection of patients using respective questionnaires, vaccination, and particularly PPE appears to be sufficient for the prevention of SARS-CoV-2 transmission in GI endoscopy,” they wrote.
Dr. Hann and colleagues analyzed 15,750 endoscopies performed by 29 staff members during the period between May 2020 and December 2021. The researchers looked at three test approaches: No testing (4,543 patients), rapid antigen testing (682 patients), and real-time PCR testing (10,465 patients). In addition, 60 endoscopies were performed in patients with known COVID-19. Overall, no staff members became infected with SARS-CoV-2 during the study period. In all three scenarios, staff used PPE, and the vaccination rate of the team was 97%.
University Hospital W
All patients were interviewed before admission for COVID-19 symptoms, close contact with infected people, and recent travel to high-risk countries. Moreover, some endoscopies were performed even if a patient had positive markers for COVID-19.
The clinical team wore recommended PPE, including a high-filter FFP2 mask, one pair of gloves, protective eyewear, and disposable gowns. For patients with known COVID-19, staff wore two pairs of gloves, a disposable hairnet, and a water-resistant disposable gown. In addition, endoscopies were performed in negative pressure intervention rooms.
Among the 29 staff members involved, 16 physicians and 13 assistants worked in the endoscopy unit for at least 2 days per week for at least 6 months. The hospital’s internal policy required medical staff to undergo PCR testing if a rapid antigen test was positive or symptoms developed. Staff were vaccinated with two doses of the Pfizer-BioNTech vaccine in January and February 2021. A single booster dose of the Pfizer or Moderna vaccine was administered in November and December 2021.
The clinical team was not tested routinely, so asymptomatic infections may have existed. Moreover, the relatively low COVID-19 incidence in the local area might have influenced the risk of transmission. “However, even at the end of 2021, when the incidence was increasing, we did not see any higher risk of transmission,” the researchers explained.
“An important limitation of our study relates to the new variant Omicron that was dominant in our local area after the analyzed time frame.” Additional studies may be needed to understand the risk of transmission with the latest Omicron variants, and given the additional costs and implications on routine activity, current testing guidelines may need to be reconsidered.
“Although our data were not part of a randomized prospective study, we were able to demonstrate on a fairly high number of patients that PPE measures in addition to a short interview for assessment of a patient’s individual risks appear to be highly effective to control transmission of SARS-CoV-2 during an endoscopy. ... Pre-procedural RT-PCR testing or RA testing did not show any additional benefit,” Dr. Hann and colleagues concluded.
The authors reported no conflicts of interest.
Pre-endoscopy viral testing may not be necessary to prevent coronavirus transmission from patients to endoscopy staff members, according to a new study published in Gut.
Instead, using personal protective equipment (PPE) and ensuring up-to-date COVID-19 vaccination among the medical team was found to be enough to substantially reduce the risk of spreading SARS-CoV-2, wrote Alexander Hann, Dr.med., gastroenterologist at University Hospital W
“We suggest that pre-selection of patients using respective questionnaires, vaccination, and particularly PPE appears to be sufficient for the prevention of SARS-CoV-2 transmission in GI endoscopy,” they wrote.
Dr. Hann and colleagues analyzed 15,750 endoscopies performed by 29 staff members during the period between May 2020 and December 2021. The researchers looked at three test approaches: No testing (4,543 patients), rapid antigen testing (682 patients), and real-time PCR testing (10,465 patients). In addition, 60 endoscopies were performed in patients with known COVID-19. Overall, no staff members became infected with SARS-CoV-2 during the study period. In all three scenarios, staff used PPE, and the vaccination rate of the team was 97%.
University Hospital W
All patients were interviewed before admission for COVID-19 symptoms, close contact with infected people, and recent travel to high-risk countries. Moreover, some endoscopies were performed even if a patient had positive markers for COVID-19.
The clinical team wore recommended PPE, including a high-filter FFP2 mask, one pair of gloves, protective eyewear, and disposable gowns. For patients with known COVID-19, staff wore two pairs of gloves, a disposable hairnet, and a water-resistant disposable gown. In addition, endoscopies were performed in negative pressure intervention rooms.
Among the 29 staff members involved, 16 physicians and 13 assistants worked in the endoscopy unit for at least 2 days per week for at least 6 months. The hospital’s internal policy required medical staff to undergo PCR testing if a rapid antigen test was positive or symptoms developed. Staff were vaccinated with two doses of the Pfizer-BioNTech vaccine in January and February 2021. A single booster dose of the Pfizer or Moderna vaccine was administered in November and December 2021.
The clinical team was not tested routinely, so asymptomatic infections may have existed. Moreover, the relatively low COVID-19 incidence in the local area might have influenced the risk of transmission. “However, even at the end of 2021, when the incidence was increasing, we did not see any higher risk of transmission,” the researchers explained.
“An important limitation of our study relates to the new variant Omicron that was dominant in our local area after the analyzed time frame.” Additional studies may be needed to understand the risk of transmission with the latest Omicron variants, and given the additional costs and implications on routine activity, current testing guidelines may need to be reconsidered.
“Although our data were not part of a randomized prospective study, we were able to demonstrate on a fairly high number of patients that PPE measures in addition to a short interview for assessment of a patient’s individual risks appear to be highly effective to control transmission of SARS-CoV-2 during an endoscopy. ... Pre-procedural RT-PCR testing or RA testing did not show any additional benefit,” Dr. Hann and colleagues concluded.
The authors reported no conflicts of interest.
FROM GUT
Children and COVID: Does latest rise in new cases point toward stabilization?
New COVID-19 cases rose for the second time in 3 weeks, as the effort to vaccinate the youngest children continued to slow after just 3 full weeks.
Nationally, over 75,000 children under age 5 years received their first dose of COVID-19 vaccine during the week of July 7-13. That number is down from the previous week – 118,000 from June 30 to July 6 – which, in turn, was lower than the 206,000 doses administered through the first 10 days after approval, based on data from the Centers for Disease Control and Prevention. That all adds up to just under 400,000 vaccinated children, or 2% of the eligible population under age 5, as of July 13.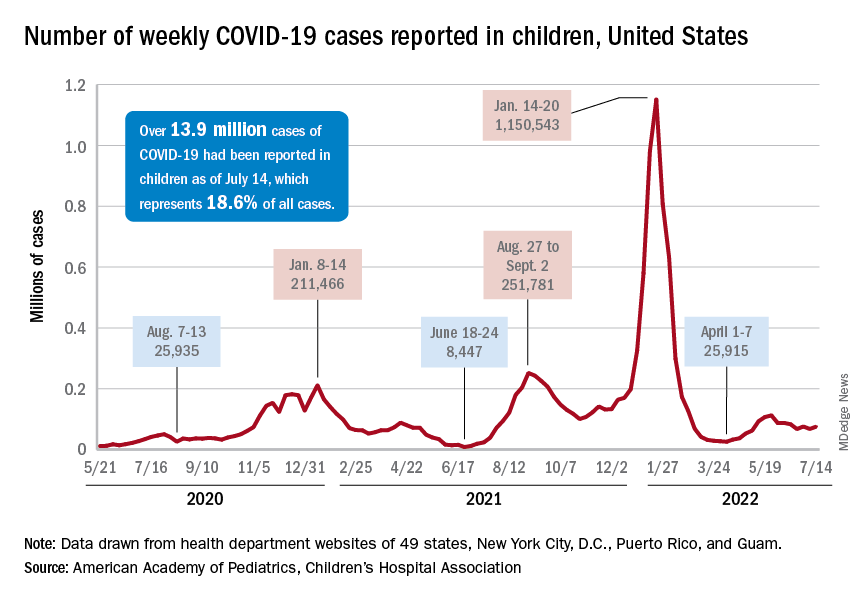
State-level data, meanwhile, show considerable variation, the American Academy of Pediatrics noted in its weekly analysis of the CDC vaccine data. Vermont has already vaccinated 10.0% of children under age 5 years, and Massachusetts is at 9.5%, while Mississippi (0.3%), Alabama (0.5%), and Louisiana (0.8%) are still below 1%, the AAP said.
New cases show signs of steadying
The national count was up by 11.1% for the week of July 8-14, rising to 75,000 new cases, compared with 68,000 the previous week, but the recent trend seems to be leaning toward steadiness. The overall number has been between 67,000 and 76,000 over the past 4 weeks, alternating between rising and falling in that time span, according to data gathered by the AAP and the Children’s Hospital Association from state and territorial health departments.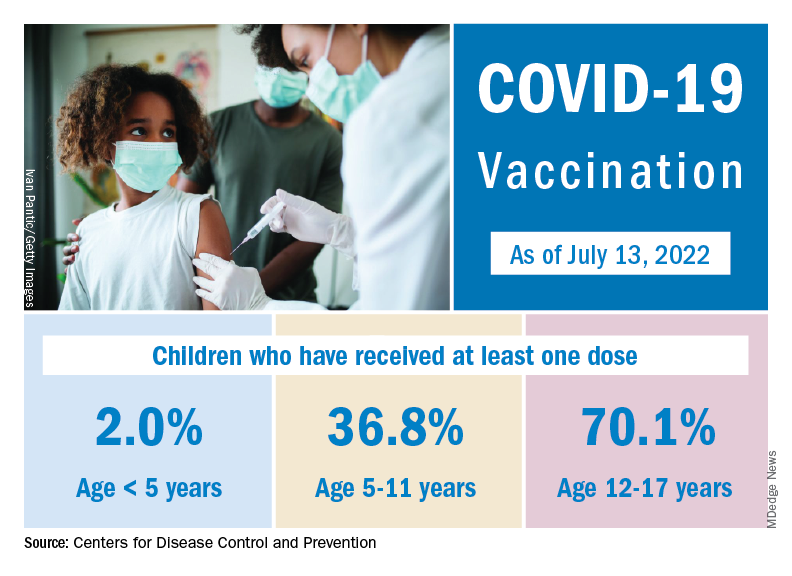
the two groups said, also noting that several states have stopped updating their online dashboards over the past year, making the current total artificially low in comparison.
Taken with that grain of salt, the cumulative number of child cases since the start of the pandemic is just over 13.9 million, which represents 18.6% of all cases in the United States. That proportion has been declining in recent weeks and was as high as 19.0% as late as mid-May. “While COVID-19 cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases,” the AAP and CHA said.
New COVID-19 cases rose for the second time in 3 weeks, as the effort to vaccinate the youngest children continued to slow after just 3 full weeks.
Nationally, over 75,000 children under age 5 years received their first dose of COVID-19 vaccine during the week of July 7-13. That number is down from the previous week – 118,000 from June 30 to July 6 – which, in turn, was lower than the 206,000 doses administered through the first 10 days after approval, based on data from the Centers for Disease Control and Prevention. That all adds up to just under 400,000 vaccinated children, or 2% of the eligible population under age 5, as of July 13.
State-level data, meanwhile, show considerable variation, the American Academy of Pediatrics noted in its weekly analysis of the CDC vaccine data. Vermont has already vaccinated 10.0% of children under age 5 years, and Massachusetts is at 9.5%, while Mississippi (0.3%), Alabama (0.5%), and Louisiana (0.8%) are still below 1%, the AAP said.
New cases show signs of steadying
The national count was up by 11.1% for the week of July 8-14, rising to 75,000 new cases, compared with 68,000 the previous week, but the recent trend seems to be leaning toward steadiness. The overall number has been between 67,000 and 76,000 over the past 4 weeks, alternating between rising and falling in that time span, according to data gathered by the AAP and the Children’s Hospital Association from state and territorial health departments.
the two groups said, also noting that several states have stopped updating their online dashboards over the past year, making the current total artificially low in comparison.
Taken with that grain of salt, the cumulative number of child cases since the start of the pandemic is just over 13.9 million, which represents 18.6% of all cases in the United States. That proportion has been declining in recent weeks and was as high as 19.0% as late as mid-May. “While COVID-19 cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases,” the AAP and CHA said.
New COVID-19 cases rose for the second time in 3 weeks, as the effort to vaccinate the youngest children continued to slow after just 3 full weeks.
Nationally, over 75,000 children under age 5 years received their first dose of COVID-19 vaccine during the week of July 7-13. That number is down from the previous week – 118,000 from June 30 to July 6 – which, in turn, was lower than the 206,000 doses administered through the first 10 days after approval, based on data from the Centers for Disease Control and Prevention. That all adds up to just under 400,000 vaccinated children, or 2% of the eligible population under age 5, as of July 13.
State-level data, meanwhile, show considerable variation, the American Academy of Pediatrics noted in its weekly analysis of the CDC vaccine data. Vermont has already vaccinated 10.0% of children under age 5 years, and Massachusetts is at 9.5%, while Mississippi (0.3%), Alabama (0.5%), and Louisiana (0.8%) are still below 1%, the AAP said.
New cases show signs of steadying
The national count was up by 11.1% for the week of July 8-14, rising to 75,000 new cases, compared with 68,000 the previous week, but the recent trend seems to be leaning toward steadiness. The overall number has been between 67,000 and 76,000 over the past 4 weeks, alternating between rising and falling in that time span, according to data gathered by the AAP and the Children’s Hospital Association from state and territorial health departments.
the two groups said, also noting that several states have stopped updating their online dashboards over the past year, making the current total artificially low in comparison.
Taken with that grain of salt, the cumulative number of child cases since the start of the pandemic is just over 13.9 million, which represents 18.6% of all cases in the United States. That proportion has been declining in recent weeks and was as high as 19.0% as late as mid-May. “While COVID-19 cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases,” the AAP and CHA said.
RV dysfunction slams survival in acute COVID, flu, pneumonia
The study covered in this summary was published in medRxiv.org as a preprint and has not yet been peer reviewed.
Key takeaways
- Right ventricular (RV) dilation or dysfunction in patients hospitalized with acute COVID-19 is associated with an elevated risk for in-hospital death.
- The impact of RV dilation or dysfunction on in-hospital mortality is similar for patients with acute COVID-19 and those with influenza, pneumonia, or acute respiratory distress syndrome (ARDS), but COVID-19 patients have greater absolute in-hospital mortality.
- RV dilatation or dysfunction in patients with acute COVID-19 is associated with a diagnosis of venous thromboembolism and subsequent intubation and mechanical ventilation.
Why this matters
- Right ventricular dysfunction increases mortality risk in acute COVID-19, and this study shows that
- The findings suggest that abnormal RV findings should be considered a mortality risk marker in patients with acute respiratory illness, especially COVID-19.
Study design
- The retrospective study involved 225 consecutive patients admitted for acute COVID-19 from March 2020 to February 2021 at four major hospitals in the same metropolitan region and a control group of 6,150 adults admitted to the hospital for influenza, pneumonia, or ARDS; mean age in the study cohort was 63 years.
- All participants underwent echocardiography during their hospitalization, including evaluation of any RV dilation or dysfunction.
- Associations between RV measurements and in-hospital mortality, the primary outcome, were adjusted for potential confounders.
Key results
- Patients in the COVID-19 group were more likely than were those in the control group to be male (66% vs. 54%; P < .001), to identify as Hispanic (38% vs. 15%; P < .001), and to have a higher mean body mass index (29.4 vs. 27.9 kg/m2; P = .008).
- Compared with the control group, patients in the COVID-19 group more often required admission to the intensive care unit (75% vs. 54%; P < .001), mechanical ventilation (P < .001), and initiation of renal replacement therapy (P = .002), and more often were diagnosed with deep-vein thrombosis or pulmonary embolism (25% vs. 14%; P < .001). The median length of hospital stay was 20 days in the COVID-19 group, compared with 10 days in the control group (P < .001).
- In-hospital mortality was 21.3% in the COVID-19 group and 11.8% in the control group (P = .001). Those hospitalized with COVID-19 had an adjusted relative risk (RR) of 1.54 (95% confidence interval [CI], 1.06-2.24; P = .02) for in-hospital mortality, compared with those hospitalized for other respiratory illnesses.
- Mild RV dilation was associated with an adjusted RR of 1.4 (95% CI, 1.17-1.69; P = .0003) for in-hospital death, and moderate to severe RV dilation was associated with an adjusted RR of 2.0 (95% CI, 1.62-2.47; P < .0001).
- The corresponding adjusted risks for mild RV dysfunction and greater-than-mild RV dysfunction were, respectively, 1.39 (95% CI, 1.10-1.77; P = .007) and 1.68 (95% CI, 1.17-2.42; P = .005).
- The RR for in-hospital mortality associated with RV dilation and dysfunction was similar in those with COVID-19 and those with other respiratory illness, but the former had a higher baseline risk that yielded a greater absolute risk in the COVID-19 group.
Limitations
- The study was based primarily on a retrospective review of electronic health records, which poses a risk for misclassification.
- Echocardiography was performed without blinding operators to patient clinical status, and echocardiograms were interpreted in a single university hospital system, so were not externally validated.
- Because echocardiograms obtained during hospitalization could not be compared with previous echocardiograms, it could not be determined whether any of the patients had preexisting RV dilation or dysfunction.
- Strain imaging was not feasible in many cases.
Disclosures
- The study received no commercial funding.
- The authors disclosed no financial relationships.
This is a summary of a preprint research study, Association of Right Ventricular Dilation and Dysfunction on Echocardiogram With In-Hospital Mortality Among Patients Hospitalized with COVID-19 Compared With Other Acute Respiratory Illness, written by researchers at the University of California, San Francisco, department of medicine, and Zuckerberg San Francisco General Hospital, division of cardiology. A version of this article first appeared on Medscape.com.
The study covered in this summary was published in medRxiv.org as a preprint and has not yet been peer reviewed.
Key takeaways
- Right ventricular (RV) dilation or dysfunction in patients hospitalized with acute COVID-19 is associated with an elevated risk for in-hospital death.
- The impact of RV dilation or dysfunction on in-hospital mortality is similar for patients with acute COVID-19 and those with influenza, pneumonia, or acute respiratory distress syndrome (ARDS), but COVID-19 patients have greater absolute in-hospital mortality.
- RV dilatation or dysfunction in patients with acute COVID-19 is associated with a diagnosis of venous thromboembolism and subsequent intubation and mechanical ventilation.
Why this matters
- Right ventricular dysfunction increases mortality risk in acute COVID-19, and this study shows that
- The findings suggest that abnormal RV findings should be considered a mortality risk marker in patients with acute respiratory illness, especially COVID-19.
Study design
- The retrospective study involved 225 consecutive patients admitted for acute COVID-19 from March 2020 to February 2021 at four major hospitals in the same metropolitan region and a control group of 6,150 adults admitted to the hospital for influenza, pneumonia, or ARDS; mean age in the study cohort was 63 years.
- All participants underwent echocardiography during their hospitalization, including evaluation of any RV dilation or dysfunction.
- Associations between RV measurements and in-hospital mortality, the primary outcome, were adjusted for potential confounders.
Key results
- Patients in the COVID-19 group were more likely than were those in the control group to be male (66% vs. 54%; P < .001), to identify as Hispanic (38% vs. 15%; P < .001), and to have a higher mean body mass index (29.4 vs. 27.9 kg/m2; P = .008).
- Compared with the control group, patients in the COVID-19 group more often required admission to the intensive care unit (75% vs. 54%; P < .001), mechanical ventilation (P < .001), and initiation of renal replacement therapy (P = .002), and more often were diagnosed with deep-vein thrombosis or pulmonary embolism (25% vs. 14%; P < .001). The median length of hospital stay was 20 days in the COVID-19 group, compared with 10 days in the control group (P < .001).
- In-hospital mortality was 21.3% in the COVID-19 group and 11.8% in the control group (P = .001). Those hospitalized with COVID-19 had an adjusted relative risk (RR) of 1.54 (95% confidence interval [CI], 1.06-2.24; P = .02) for in-hospital mortality, compared with those hospitalized for other respiratory illnesses.
- Mild RV dilation was associated with an adjusted RR of 1.4 (95% CI, 1.17-1.69; P = .0003) for in-hospital death, and moderate to severe RV dilation was associated with an adjusted RR of 2.0 (95% CI, 1.62-2.47; P < .0001).
- The corresponding adjusted risks for mild RV dysfunction and greater-than-mild RV dysfunction were, respectively, 1.39 (95% CI, 1.10-1.77; P = .007) and 1.68 (95% CI, 1.17-2.42; P = .005).
- The RR for in-hospital mortality associated with RV dilation and dysfunction was similar in those with COVID-19 and those with other respiratory illness, but the former had a higher baseline risk that yielded a greater absolute risk in the COVID-19 group.
Limitations
- The study was based primarily on a retrospective review of electronic health records, which poses a risk for misclassification.
- Echocardiography was performed without blinding operators to patient clinical status, and echocardiograms were interpreted in a single university hospital system, so were not externally validated.
- Because echocardiograms obtained during hospitalization could not be compared with previous echocardiograms, it could not be determined whether any of the patients had preexisting RV dilation or dysfunction.
- Strain imaging was not feasible in many cases.
Disclosures
- The study received no commercial funding.
- The authors disclosed no financial relationships.
This is a summary of a preprint research study, Association of Right Ventricular Dilation and Dysfunction on Echocardiogram With In-Hospital Mortality Among Patients Hospitalized with COVID-19 Compared With Other Acute Respiratory Illness, written by researchers at the University of California, San Francisco, department of medicine, and Zuckerberg San Francisco General Hospital, division of cardiology. A version of this article first appeared on Medscape.com.
The study covered in this summary was published in medRxiv.org as a preprint and has not yet been peer reviewed.
Key takeaways
- Right ventricular (RV) dilation or dysfunction in patients hospitalized with acute COVID-19 is associated with an elevated risk for in-hospital death.
- The impact of RV dilation or dysfunction on in-hospital mortality is similar for patients with acute COVID-19 and those with influenza, pneumonia, or acute respiratory distress syndrome (ARDS), but COVID-19 patients have greater absolute in-hospital mortality.
- RV dilatation or dysfunction in patients with acute COVID-19 is associated with a diagnosis of venous thromboembolism and subsequent intubation and mechanical ventilation.
Why this matters
- Right ventricular dysfunction increases mortality risk in acute COVID-19, and this study shows that
- The findings suggest that abnormal RV findings should be considered a mortality risk marker in patients with acute respiratory illness, especially COVID-19.
Study design
- The retrospective study involved 225 consecutive patients admitted for acute COVID-19 from March 2020 to February 2021 at four major hospitals in the same metropolitan region and a control group of 6,150 adults admitted to the hospital for influenza, pneumonia, or ARDS; mean age in the study cohort was 63 years.
- All participants underwent echocardiography during their hospitalization, including evaluation of any RV dilation or dysfunction.
- Associations between RV measurements and in-hospital mortality, the primary outcome, were adjusted for potential confounders.
Key results
- Patients in the COVID-19 group were more likely than were those in the control group to be male (66% vs. 54%; P < .001), to identify as Hispanic (38% vs. 15%; P < .001), and to have a higher mean body mass index (29.4 vs. 27.9 kg/m2; P = .008).
- Compared with the control group, patients in the COVID-19 group more often required admission to the intensive care unit (75% vs. 54%; P < .001), mechanical ventilation (P < .001), and initiation of renal replacement therapy (P = .002), and more often were diagnosed with deep-vein thrombosis or pulmonary embolism (25% vs. 14%; P < .001). The median length of hospital stay was 20 days in the COVID-19 group, compared with 10 days in the control group (P < .001).
- In-hospital mortality was 21.3% in the COVID-19 group and 11.8% in the control group (P = .001). Those hospitalized with COVID-19 had an adjusted relative risk (RR) of 1.54 (95% confidence interval [CI], 1.06-2.24; P = .02) for in-hospital mortality, compared with those hospitalized for other respiratory illnesses.
- Mild RV dilation was associated with an adjusted RR of 1.4 (95% CI, 1.17-1.69; P = .0003) for in-hospital death, and moderate to severe RV dilation was associated with an adjusted RR of 2.0 (95% CI, 1.62-2.47; P < .0001).
- The corresponding adjusted risks for mild RV dysfunction and greater-than-mild RV dysfunction were, respectively, 1.39 (95% CI, 1.10-1.77; P = .007) and 1.68 (95% CI, 1.17-2.42; P = .005).
- The RR for in-hospital mortality associated with RV dilation and dysfunction was similar in those with COVID-19 and those with other respiratory illness, but the former had a higher baseline risk that yielded a greater absolute risk in the COVID-19 group.
Limitations
- The study was based primarily on a retrospective review of electronic health records, which poses a risk for misclassification.
- Echocardiography was performed without blinding operators to patient clinical status, and echocardiograms were interpreted in a single university hospital system, so were not externally validated.
- Because echocardiograms obtained during hospitalization could not be compared with previous echocardiograms, it could not be determined whether any of the patients had preexisting RV dilation or dysfunction.
- Strain imaging was not feasible in many cases.
Disclosures
- The study received no commercial funding.
- The authors disclosed no financial relationships.
This is a summary of a preprint research study, Association of Right Ventricular Dilation and Dysfunction on Echocardiogram With In-Hospital Mortality Among Patients Hospitalized with COVID-19 Compared With Other Acute Respiratory Illness, written by researchers at the University of California, San Francisco, department of medicine, and Zuckerberg San Francisco General Hospital, division of cardiology. A version of this article first appeared on Medscape.com.
CDC warns about potentially deadly virus in infants
The potentially fatal parechovirus is now circulating in multiple states, causing fevers, seizures, and sepsis-like symptoms, including confusion and extreme pain, according to the CDC.
Human parechoviruses are common in children and most have been infected before they start kindergarten, the CDC said. Between 6 months and 5 years of age, symptoms include an upper respiratory tract infection, fever, and rash.
But infants younger than 3 months may have more serious, and possibly fatal, infections. They may get “sepsis-like illness, seizures, and meningitis or meningoencephalitis, particularly in infants younger than 1 month,” the CDC said. At least one newborn has reportedly died from the infection.
Parechovirus can spread like other common germs, from feces that are later ingested – likely due to poor handwashing – and through droplets sent airborne by coughing or sneezing. It can be transmitted by people both with and without symptoms of the infection.
The microbe can reproduce for 1-3 weeks in the upper respiratory tract and up to 6 months in the gastrointestinal tract, the CDC said.
Kristina Angel Bryant, MD, a pediatric infectious disease specialist at the University of Louisville Hospital, says parechoviruses often cause rashes on the hands and feet, which some experts refer to as “mittens and booties.”
The CDC is urging doctors to test for parechovirus if they recognize these symptoms in infants if there is no other explanation for what might be distressing them.
There is no specific treatment for parechovirus. And with no standard testing system in place, experts are unsure if the number of parechovirus cases is higher in 2022 than in previous years.
The message for parents, Dr. Bryant says, is: Don’t panic. “This is not a new virus.”
“One of the most common symptoms is fever, and in some kids, that is the only symptom,” she says. “Older infants and toddlers may have only cold symptoms, and some kids have no symptoms at all.”
Parents can take the usual steps to protect their child from the viral illness, including diligent handwashing and having less contact with people who are sick, Dr. Bryant says.
A version of this article first appeared on Medscape.com.
The potentially fatal parechovirus is now circulating in multiple states, causing fevers, seizures, and sepsis-like symptoms, including confusion and extreme pain, according to the CDC.
Human parechoviruses are common in children and most have been infected before they start kindergarten, the CDC said. Between 6 months and 5 years of age, symptoms include an upper respiratory tract infection, fever, and rash.
But infants younger than 3 months may have more serious, and possibly fatal, infections. They may get “sepsis-like illness, seizures, and meningitis or meningoencephalitis, particularly in infants younger than 1 month,” the CDC said. At least one newborn has reportedly died from the infection.
Parechovirus can spread like other common germs, from feces that are later ingested – likely due to poor handwashing – and through droplets sent airborne by coughing or sneezing. It can be transmitted by people both with and without symptoms of the infection.
The microbe can reproduce for 1-3 weeks in the upper respiratory tract and up to 6 months in the gastrointestinal tract, the CDC said.
Kristina Angel Bryant, MD, a pediatric infectious disease specialist at the University of Louisville Hospital, says parechoviruses often cause rashes on the hands and feet, which some experts refer to as “mittens and booties.”
The CDC is urging doctors to test for parechovirus if they recognize these symptoms in infants if there is no other explanation for what might be distressing them.
There is no specific treatment for parechovirus. And with no standard testing system in place, experts are unsure if the number of parechovirus cases is higher in 2022 than in previous years.
The message for parents, Dr. Bryant says, is: Don’t panic. “This is not a new virus.”
“One of the most common symptoms is fever, and in some kids, that is the only symptom,” she says. “Older infants and toddlers may have only cold symptoms, and some kids have no symptoms at all.”
Parents can take the usual steps to protect their child from the viral illness, including diligent handwashing and having less contact with people who are sick, Dr. Bryant says.
A version of this article first appeared on Medscape.com.
The potentially fatal parechovirus is now circulating in multiple states, causing fevers, seizures, and sepsis-like symptoms, including confusion and extreme pain, according to the CDC.
Human parechoviruses are common in children and most have been infected before they start kindergarten, the CDC said. Between 6 months and 5 years of age, symptoms include an upper respiratory tract infection, fever, and rash.
But infants younger than 3 months may have more serious, and possibly fatal, infections. They may get “sepsis-like illness, seizures, and meningitis or meningoencephalitis, particularly in infants younger than 1 month,” the CDC said. At least one newborn has reportedly died from the infection.
Parechovirus can spread like other common germs, from feces that are later ingested – likely due to poor handwashing – and through droplets sent airborne by coughing or sneezing. It can be transmitted by people both with and without symptoms of the infection.
The microbe can reproduce for 1-3 weeks in the upper respiratory tract and up to 6 months in the gastrointestinal tract, the CDC said.
Kristina Angel Bryant, MD, a pediatric infectious disease specialist at the University of Louisville Hospital, says parechoviruses often cause rashes on the hands and feet, which some experts refer to as “mittens and booties.”
The CDC is urging doctors to test for parechovirus if they recognize these symptoms in infants if there is no other explanation for what might be distressing them.
There is no specific treatment for parechovirus. And with no standard testing system in place, experts are unsure if the number of parechovirus cases is higher in 2022 than in previous years.
The message for parents, Dr. Bryant says, is: Don’t panic. “This is not a new virus.”
“One of the most common symptoms is fever, and in some kids, that is the only symptom,” she says. “Older infants and toddlers may have only cold symptoms, and some kids have no symptoms at all.”
Parents can take the usual steps to protect their child from the viral illness, including diligent handwashing and having less contact with people who are sick, Dr. Bryant says.
A version of this article first appeared on Medscape.com.
FDA approves topical ruxolitinib for nonsegmental vitiligo
The on July 18. The treatment, which was approved for treating mild to moderate atopic dermatitis in September 2021, is a cream formulation of ruxolitinib, a Janus kinase 1 (JAK1)/JAK2 inhibitor.
Previously, no treatment was approved to repigment patients with vitiligo, says David Rosmarin, MD, vice chair for research and education in the department of dermatology at Tufts Medical Center, Boston. “It’s important to have options that we can give to patients that are both safe and effective to get them the desired results,” Dr. Rosmarin, the lead investigator of the phase 3 clinical trials of topical ruxolitinib, said in an interview. Vitiligo is “a disease that can really affect quality of life. Some people [with vitiligo] feel as if they’re being stared at or they’re being bullied; they don’t feel confident. It can affect relationships and intimacy.”
Approval was based on the results of two phase 3 trials (TruE-V1 and TruE-V2) in 674 patients with nonsegmental vitiligo aged 12 years or older. At 24 weeks, about 30% of the patients on treatment, applied twice a day, achieved at least a 75% improvement in the facial Vitiligo Area Scoring Index (F-VASI75), compared with about 8% and 13% among those in the vehicle groups in the two trials.
At 52 weeks, about 50% of the patients treated with topical ruxolitinib achieved F-VASI75.
Also, using self-reporting as measured by the Vitiligo Noticeability Scale, about 30%-40% of patients described their vitiligo as being “a lot less noticeable” or “no longer noticeable” at week 52. Dr. Rosmarin reported the 52-week results at the 2022 annual meeting of the American Academy of Dermatology.
The trial group used 1.5% ruxolitinib cream twice daily for the full year. The vehicle group began using ruxolitinib halfway through the trial. In this group, 26.8% and 29.6% achieved F-VASI 75 at 52 weeks in the two trials.
For treating vitiligo, patients are advised to apply a thin layer of topical ruxolitinib to affected areas twice a day, “up to 10% body surface area,” according to the prescribing information, which adds: “Satisfactory patient response may require treatment … for more than 24 weeks. If the patient does not find the repigmentation meaningful by 24 weeks, the patient should be reevaluated by the health care provider.”
The most common side effects during the vehicle-controlled part of the trials were development of acne and pruritus at the application site, headache, urinary tract infections, erythema at the application site, and pyrexia, according to the company.
The approved label for topical ruxolitinib includes a boxed warning about serious infections, mortality, cancer, major adverse cardiovascular events, and thrombosis – which, the warning notes, is based on reports in patients treated with oral JAK inhibitors for inflammatory conditions.
Dr. Rosmarin believes that using this drug with other therapies, like light treatment, might yield even better responses. The available data are in patients treated with ruxolitinib as monotherapy, without complementary therapies.
William Damsky, MD, PhD, professor of dermatology and dermatopathology at Yale University, New Haven, who was not involved in the trials, said what is most exciting about this drug is its novelty. Although some topical steroids are used off-label to treat vitiligo, their efficacy is far from what’s been observed in these trials of topical ruxolitinib, he told this news organization. “It’s huge for a number of reasons. … One very big reason is it just provides some hope” for the many patients with vitiligo who, over the years, have been told “that there’s nothing that could be done for their disease, and this really changes that.”
Dr. Rosmarin reports financial relationships with over 20 pharmaceutical companies. Dr. Damsky disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The on July 18. The treatment, which was approved for treating mild to moderate atopic dermatitis in September 2021, is a cream formulation of ruxolitinib, a Janus kinase 1 (JAK1)/JAK2 inhibitor.
Previously, no treatment was approved to repigment patients with vitiligo, says David Rosmarin, MD, vice chair for research and education in the department of dermatology at Tufts Medical Center, Boston. “It’s important to have options that we can give to patients that are both safe and effective to get them the desired results,” Dr. Rosmarin, the lead investigator of the phase 3 clinical trials of topical ruxolitinib, said in an interview. Vitiligo is “a disease that can really affect quality of life. Some people [with vitiligo] feel as if they’re being stared at or they’re being bullied; they don’t feel confident. It can affect relationships and intimacy.”
Approval was based on the results of two phase 3 trials (TruE-V1 and TruE-V2) in 674 patients with nonsegmental vitiligo aged 12 years or older. At 24 weeks, about 30% of the patients on treatment, applied twice a day, achieved at least a 75% improvement in the facial Vitiligo Area Scoring Index (F-VASI75), compared with about 8% and 13% among those in the vehicle groups in the two trials.
At 52 weeks, about 50% of the patients treated with topical ruxolitinib achieved F-VASI75.
Also, using self-reporting as measured by the Vitiligo Noticeability Scale, about 30%-40% of patients described their vitiligo as being “a lot less noticeable” or “no longer noticeable” at week 52. Dr. Rosmarin reported the 52-week results at the 2022 annual meeting of the American Academy of Dermatology.
The trial group used 1.5% ruxolitinib cream twice daily for the full year. The vehicle group began using ruxolitinib halfway through the trial. In this group, 26.8% and 29.6% achieved F-VASI 75 at 52 weeks in the two trials.
For treating vitiligo, patients are advised to apply a thin layer of topical ruxolitinib to affected areas twice a day, “up to 10% body surface area,” according to the prescribing information, which adds: “Satisfactory patient response may require treatment … for more than 24 weeks. If the patient does not find the repigmentation meaningful by 24 weeks, the patient should be reevaluated by the health care provider.”
The most common side effects during the vehicle-controlled part of the trials were development of acne and pruritus at the application site, headache, urinary tract infections, erythema at the application site, and pyrexia, according to the company.
The approved label for topical ruxolitinib includes a boxed warning about serious infections, mortality, cancer, major adverse cardiovascular events, and thrombosis – which, the warning notes, is based on reports in patients treated with oral JAK inhibitors for inflammatory conditions.
Dr. Rosmarin believes that using this drug with other therapies, like light treatment, might yield even better responses. The available data are in patients treated with ruxolitinib as monotherapy, without complementary therapies.
William Damsky, MD, PhD, professor of dermatology and dermatopathology at Yale University, New Haven, who was not involved in the trials, said what is most exciting about this drug is its novelty. Although some topical steroids are used off-label to treat vitiligo, their efficacy is far from what’s been observed in these trials of topical ruxolitinib, he told this news organization. “It’s huge for a number of reasons. … One very big reason is it just provides some hope” for the many patients with vitiligo who, over the years, have been told “that there’s nothing that could be done for their disease, and this really changes that.”
Dr. Rosmarin reports financial relationships with over 20 pharmaceutical companies. Dr. Damsky disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The on July 18. The treatment, which was approved for treating mild to moderate atopic dermatitis in September 2021, is a cream formulation of ruxolitinib, a Janus kinase 1 (JAK1)/JAK2 inhibitor.
Previously, no treatment was approved to repigment patients with vitiligo, says David Rosmarin, MD, vice chair for research and education in the department of dermatology at Tufts Medical Center, Boston. “It’s important to have options that we can give to patients that are both safe and effective to get them the desired results,” Dr. Rosmarin, the lead investigator of the phase 3 clinical trials of topical ruxolitinib, said in an interview. Vitiligo is “a disease that can really affect quality of life. Some people [with vitiligo] feel as if they’re being stared at or they’re being bullied; they don’t feel confident. It can affect relationships and intimacy.”
Approval was based on the results of two phase 3 trials (TruE-V1 and TruE-V2) in 674 patients with nonsegmental vitiligo aged 12 years or older. At 24 weeks, about 30% of the patients on treatment, applied twice a day, achieved at least a 75% improvement in the facial Vitiligo Area Scoring Index (F-VASI75), compared with about 8% and 13% among those in the vehicle groups in the two trials.
At 52 weeks, about 50% of the patients treated with topical ruxolitinib achieved F-VASI75.
Also, using self-reporting as measured by the Vitiligo Noticeability Scale, about 30%-40% of patients described their vitiligo as being “a lot less noticeable” or “no longer noticeable” at week 52. Dr. Rosmarin reported the 52-week results at the 2022 annual meeting of the American Academy of Dermatology.
The trial group used 1.5% ruxolitinib cream twice daily for the full year. The vehicle group began using ruxolitinib halfway through the trial. In this group, 26.8% and 29.6% achieved F-VASI 75 at 52 weeks in the two trials.
For treating vitiligo, patients are advised to apply a thin layer of topical ruxolitinib to affected areas twice a day, “up to 10% body surface area,” according to the prescribing information, which adds: “Satisfactory patient response may require treatment … for more than 24 weeks. If the patient does not find the repigmentation meaningful by 24 weeks, the patient should be reevaluated by the health care provider.”
The most common side effects during the vehicle-controlled part of the trials were development of acne and pruritus at the application site, headache, urinary tract infections, erythema at the application site, and pyrexia, according to the company.
The approved label for topical ruxolitinib includes a boxed warning about serious infections, mortality, cancer, major adverse cardiovascular events, and thrombosis – which, the warning notes, is based on reports in patients treated with oral JAK inhibitors for inflammatory conditions.
Dr. Rosmarin believes that using this drug with other therapies, like light treatment, might yield even better responses. The available data are in patients treated with ruxolitinib as monotherapy, without complementary therapies.
William Damsky, MD, PhD, professor of dermatology and dermatopathology at Yale University, New Haven, who was not involved in the trials, said what is most exciting about this drug is its novelty. Although some topical steroids are used off-label to treat vitiligo, their efficacy is far from what’s been observed in these trials of topical ruxolitinib, he told this news organization. “It’s huge for a number of reasons. … One very big reason is it just provides some hope” for the many patients with vitiligo who, over the years, have been told “that there’s nothing that could be done for their disease, and this really changes that.”
Dr. Rosmarin reports financial relationships with over 20 pharmaceutical companies. Dr. Damsky disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Methotrexate’s impact on COVID-19 vaccination: New insights made
Patients who take methotrexate for a variety of immune-mediated inflammatory diseases and pause taking the drug following receipt of a COVID-19 vaccine dose did not have a higher risk of disease flare and had higher antireceptor binding domain (anti-RBD) antibody titers and increased immunogenicity when compared with continuing the drug, three recent studies suggest.
In one study, British researchers examined the effects of a 2-week break in methotrexate therapy on anti-RBD titers following receipt of a third COVID-19 vaccine dose. In their paper published in The Lancet: Respiratory Medicine, they reported results from a randomized, open-label, superiority trial that suggested pausing the drug improved immunogenicity, compared with no break.
In two trials presented at the European Alliance of Associations for Rheumatology (EULAR) 2022 Congress, a team from India set out to determine whether holding methotrexate after receiving both doses of a COVID-19 vaccine, or holding it only after the second dose, was safe and effective. They found that pausing methotrexate only following the second dose contributed to a lower flare risk, and that patients had higher anti-RBD titers when holding methotrexate for 2 weeks following each dose.
Pausing methotrexate after booster
The 2-week methotrexate break and booster vaccine dose data in the Vaccine Response On Off Methotrexate (VROOM) trial showed that after a month, the geometric mean antispike 1 (S1)-RBD antibody titer was 10,798 U/mL (95% confidence interval [CI], 8,970-12,997) in the group that continued methotrexate and 22,750 U/mL (95% CI, 19,314-26,796) in the group that suspended methotrexate; the geometric mean ratio was 2.19 (P < .0001; mixed-effects model), reported Abhishek Abhishek, MD, PhD, professor of rheumatology at the University of Nottingham in Nottingham, England, and colleagues.
Prior research showed that stopping methotrexate therapy for 2 weeks following the seasonal influenza vaccine contributed to better vaccine immunity among patients with rheumatoid arthritis, but there was no impact of stopping the drug for up to 4 weeks before vaccination on vaccine-related immunity, the researchers noted.
It is crucial in maximizing long-lasting vaccine protection in people who are possibly susceptible through immune suppression at this point in the COVID-19 vaccination regimen, the study team noted.
“Evidence from this study will be useful for policymakers, national immunization advisory committees, and specialist societies formulating recommendations on the use of methotrexate around the time of COVID-19 vaccination. This evidence will help patients and clinicians make informed choices about the risks and benefits of interrupting methotrexate treatment around the time of COVID-19 vaccination, with implications for the potential to extend such approaches to other therapeutics,” they wrote.
In American College of Rheumatology (ACR) guidance for COVID-19 vaccination, the organization advised against using standard synthetic disease-modifying antirheumatic medicines such as methotrexate “for 1-2 weeks (as disease activity allows) after each COVID-19 vaccine dose,” given the at-risk population and public health concerns, Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine and associate physician at Brigham and Women’s Hospital and Harvard Medical School, Boston, and Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, noted in an accompanying editorial in The Lancet: Respiratory Medicine.
However, when the ACR developed this statement, there was only one trial involving patients with rheumatoid arthritis who paused methotrexate following seasonal influenza vaccination, the editorialists said.
“Although this finding adds to the evidence base to support interruption of methotrexate after vaccination, a shared decision process is needed to weigh the possible benefit of optimizing protection from COVID-19 and the possible risk of underlying disease flare,” they added.
Dr. Abhishek and colleagues assessed 254 patients with immune-mediated inflammatory disease from dermatology and rheumatology clinics across 26 hospitals in the United Kingdom. Participants had been diagnosed with systemic lupus erythematosus, rheumatoid arthritis, atopic dermatitis, polymyalgia rheumatica, axial spondyloarthritis, and psoriasis without or with arthritis. They had also been taking up to 25 mg of methotrexate per week for 3 months or longer and had received two doses of either the Pfizer/BioNTech BNT162b2 vaccine or AstraZeneca/Oxford viral vector vaccine. The booster dose was most often the Pfizer BNT162b2 vaccine (82%). The patients’ mean age was 59 years, with females comprising 61% of the cohort. Participants were randomly assigned 1:1 to either group.
Investigators performing laboratory analysis were masked to cohort assignment, and clinical research staff, data analysts, participants, and researchers were unmasked.
The elevated antibody response of patients who suspended methotrexate was the same across different kinds of immune-mediated inflammatory disease, primary vaccination platform, SARS-CoV-2 infection history, and age.
Notably, no intervention-associated adverse events were reported, the study team noted.
The conclusions that could be drawn from the booster-dose study were limited by the trial’s modest cohort size, the small number of patients in exploratory subgroup analyses, a lack of information about differences in prescription drug behavior, and early termination’s effect on the researchers’ ability to identify differences between subgroups and in secondary outcomes, the authors noted.
Other limitations included a lack of generalizability to patients with active disease who couldn’t stop therapy and were not included in the investigation, and participants were not blinded to what group they were in, the researchers said.
Expert commentary
This current study is consistent with other studies over the last several months showing that methotrexate harms both humoral and cell-mediated COVID-19 responses, noted Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study. “And so now the new wave of studies are like this one, where they are holding methotrexate experimentally and seeing if it makes a difference,” he said.
“The one shortcoming of this study – and so far, the studies to date – is that no one has looked at whether the experimental hold has resulted in a change in T-cell responses, which ... we are [now] recognizing [the importance of] more and more in long-term protection, particularly in severe disease. Theoretically, holding [methotrexate] might help enhance T-cell responses, but that hasn’t been shown experimentally.”
Dr. Winthrop pointed out that one might get the same benefit from holding methotrexate for 1 week instead of 2 and that there likely is a reduced risk of flare-up from underlying autoimmune disease.
It is still not certain that this benefit extends to other vaccines, Dr. Winthrop noted. “It is probably true for most vaccines that if you hold methotrexate for 1 or 2 weeks, you might see some short-term benefit in responsiveness, but you don’t know that there is any clinical meaningfulness of this. That’s going to take other long-term studies. You don’t know how long this benefit lasts.”
Pausing methotrexate during initial COVID vaccine doses
Patients with either rheumatoid arthritis or psoriatic arthritis had higher anti-RBD antibody titers when methotrexate was stopped after both doses of the AstraZeneca vaccine, or simply after the second dose, than when methotrexate was continued, according to results from two single-center, randomized controlled trials called MIVAC I and II, Anu Sreekanth, MD, of Sree Sudheendra Medical Mission in Kochi, Kerala, India, and colleagues reported at EULAR 2022.
Results from MIVAC I indicated that there was a higher flare rate when methotrexate was stopped after both vaccine doses, but there was no difference in flare rate in MIVAC II when methotrexate was stopped only after the second dose as opposed to stopping it after both doses.
In the MIVAC I trial, 158 unvaccinated patients were randomized 1:1 to a cohort in which methotrexate was held for 2 weeks after both doses and a cohort in which methotrexate was continued despite the vaccine. In MIVAC II, 157 patients continued methotrexate while receiving the first vaccine dose. These patients were subsequently randomized either to continue or to stop methotrexate for 2 weeks following the second dose.
The findings from MIVAC I demonstrated the flare rate was lower in the methotrexate-continue group than in the methotrexate-pause group (8% vs. 25%; P = .005) and that the median anti-RBD titer was significantly higher for the methotrexate-pause group than the methotrexate-continue group (2,484 vs. 1,147; P = .001).
The results from MIVAC II trial indicated that there was no difference in flare rates between the two study groups (7.9% vs. 11.8%; P = .15). Yet, the median anti-RBD titer was significantly higher in the methotrexate-pause cohort than in the methotrexate-continue cohort (2,553 vs. 990; P = .001).
The report suggests there is a flare risk when methotrexate is stopped, Dr. Sreekanth noted. “It appears more logical to hold only after the second dose, as comparable anti-RBD titers are generated” with either approach, Dr. Sreekanth said.
Expert commentary: MIVAC I and II
Inés Colmegna, MD, associate professor at McGill University in Montreal, noted that it was intriguing that the risk of flares in MIVAC II is half of that reported after each of the doses of MIVAC I. “It is also worth emphasizing that despite the reported frequency of flares, the actual disease activity [as measured by the Disease Activity Score in 28 joints] in patients who did or did not withhold methotrexate was similar.
“MIVAC I and II have practical implications as they help to adequately inform patients about the risk and benefit trade of withholding methotrexate post–COVID-19 vaccination,” Dr. Colmegna told this news organization.
“Additional information would help to [further] interpret the findings of these studies, including whether any of the participants were taking any other DMARDs; data on the severity of the flares and functional impact; analysis of factors that predict the risk of flares, such as higher doses of methotrexate; [and change in] disease activity scores pre- and postvaccination,” Dr. Colmegna concluded.
Dr. Abhishek disclosed relationships with Springer, UpTodate, Oxford, Immunotec, AstraZeneca, Inflazome, NGM Biopharmaceuticals, Menarini Pharmaceuticals, and Cadila Pharmaceuticals. Dr. Abhishek is cochair of the ACR/EULAR CPPD Classification Criteria Working Group and the OMERACT CPPD Working Group. Dr. Sparks disclosed relationships with Gilead, Boehringer Ingelheim, Amgen, Bristol-Myers Squibb, and AbbVie, unrelated to this study. Dr. Tedeschi disclosed relationships with ModernaTx and NGM Biopharmaceuticals. Dr. Winthrop disclosed a research grant and serving as a scientific consultant for Pfizer. Dr. Sreekanth and Dr. Colmegna have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients who take methotrexate for a variety of immune-mediated inflammatory diseases and pause taking the drug following receipt of a COVID-19 vaccine dose did not have a higher risk of disease flare and had higher antireceptor binding domain (anti-RBD) antibody titers and increased immunogenicity when compared with continuing the drug, three recent studies suggest.
In one study, British researchers examined the effects of a 2-week break in methotrexate therapy on anti-RBD titers following receipt of a third COVID-19 vaccine dose. In their paper published in The Lancet: Respiratory Medicine, they reported results from a randomized, open-label, superiority trial that suggested pausing the drug improved immunogenicity, compared with no break.
In two trials presented at the European Alliance of Associations for Rheumatology (EULAR) 2022 Congress, a team from India set out to determine whether holding methotrexate after receiving both doses of a COVID-19 vaccine, or holding it only after the second dose, was safe and effective. They found that pausing methotrexate only following the second dose contributed to a lower flare risk, and that patients had higher anti-RBD titers when holding methotrexate for 2 weeks following each dose.
Pausing methotrexate after booster
The 2-week methotrexate break and booster vaccine dose data in the Vaccine Response On Off Methotrexate (VROOM) trial showed that after a month, the geometric mean antispike 1 (S1)-RBD antibody titer was 10,798 U/mL (95% confidence interval [CI], 8,970-12,997) in the group that continued methotrexate and 22,750 U/mL (95% CI, 19,314-26,796) in the group that suspended methotrexate; the geometric mean ratio was 2.19 (P < .0001; mixed-effects model), reported Abhishek Abhishek, MD, PhD, professor of rheumatology at the University of Nottingham in Nottingham, England, and colleagues.
Prior research showed that stopping methotrexate therapy for 2 weeks following the seasonal influenza vaccine contributed to better vaccine immunity among patients with rheumatoid arthritis, but there was no impact of stopping the drug for up to 4 weeks before vaccination on vaccine-related immunity, the researchers noted.
It is crucial in maximizing long-lasting vaccine protection in people who are possibly susceptible through immune suppression at this point in the COVID-19 vaccination regimen, the study team noted.
“Evidence from this study will be useful for policymakers, national immunization advisory committees, and specialist societies formulating recommendations on the use of methotrexate around the time of COVID-19 vaccination. This evidence will help patients and clinicians make informed choices about the risks and benefits of interrupting methotrexate treatment around the time of COVID-19 vaccination, with implications for the potential to extend such approaches to other therapeutics,” they wrote.
In American College of Rheumatology (ACR) guidance for COVID-19 vaccination, the organization advised against using standard synthetic disease-modifying antirheumatic medicines such as methotrexate “for 1-2 weeks (as disease activity allows) after each COVID-19 vaccine dose,” given the at-risk population and public health concerns, Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine and associate physician at Brigham and Women’s Hospital and Harvard Medical School, Boston, and Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, noted in an accompanying editorial in The Lancet: Respiratory Medicine.
However, when the ACR developed this statement, there was only one trial involving patients with rheumatoid arthritis who paused methotrexate following seasonal influenza vaccination, the editorialists said.
“Although this finding adds to the evidence base to support interruption of methotrexate after vaccination, a shared decision process is needed to weigh the possible benefit of optimizing protection from COVID-19 and the possible risk of underlying disease flare,” they added.
Dr. Abhishek and colleagues assessed 254 patients with immune-mediated inflammatory disease from dermatology and rheumatology clinics across 26 hospitals in the United Kingdom. Participants had been diagnosed with systemic lupus erythematosus, rheumatoid arthritis, atopic dermatitis, polymyalgia rheumatica, axial spondyloarthritis, and psoriasis without or with arthritis. They had also been taking up to 25 mg of methotrexate per week for 3 months or longer and had received two doses of either the Pfizer/BioNTech BNT162b2 vaccine or AstraZeneca/Oxford viral vector vaccine. The booster dose was most often the Pfizer BNT162b2 vaccine (82%). The patients’ mean age was 59 years, with females comprising 61% of the cohort. Participants were randomly assigned 1:1 to either group.
Investigators performing laboratory analysis were masked to cohort assignment, and clinical research staff, data analysts, participants, and researchers were unmasked.
The elevated antibody response of patients who suspended methotrexate was the same across different kinds of immune-mediated inflammatory disease, primary vaccination platform, SARS-CoV-2 infection history, and age.
Notably, no intervention-associated adverse events were reported, the study team noted.
The conclusions that could be drawn from the booster-dose study were limited by the trial’s modest cohort size, the small number of patients in exploratory subgroup analyses, a lack of information about differences in prescription drug behavior, and early termination’s effect on the researchers’ ability to identify differences between subgroups and in secondary outcomes, the authors noted.
Other limitations included a lack of generalizability to patients with active disease who couldn’t stop therapy and were not included in the investigation, and participants were not blinded to what group they were in, the researchers said.
Expert commentary
This current study is consistent with other studies over the last several months showing that methotrexate harms both humoral and cell-mediated COVID-19 responses, noted Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study. “And so now the new wave of studies are like this one, where they are holding methotrexate experimentally and seeing if it makes a difference,” he said.
“The one shortcoming of this study – and so far, the studies to date – is that no one has looked at whether the experimental hold has resulted in a change in T-cell responses, which ... we are [now] recognizing [the importance of] more and more in long-term protection, particularly in severe disease. Theoretically, holding [methotrexate] might help enhance T-cell responses, but that hasn’t been shown experimentally.”
Dr. Winthrop pointed out that one might get the same benefit from holding methotrexate for 1 week instead of 2 and that there likely is a reduced risk of flare-up from underlying autoimmune disease.
It is still not certain that this benefit extends to other vaccines, Dr. Winthrop noted. “It is probably true for most vaccines that if you hold methotrexate for 1 or 2 weeks, you might see some short-term benefit in responsiveness, but you don’t know that there is any clinical meaningfulness of this. That’s going to take other long-term studies. You don’t know how long this benefit lasts.”
Pausing methotrexate during initial COVID vaccine doses
Patients with either rheumatoid arthritis or psoriatic arthritis had higher anti-RBD antibody titers when methotrexate was stopped after both doses of the AstraZeneca vaccine, or simply after the second dose, than when methotrexate was continued, according to results from two single-center, randomized controlled trials called MIVAC I and II, Anu Sreekanth, MD, of Sree Sudheendra Medical Mission in Kochi, Kerala, India, and colleagues reported at EULAR 2022.
Results from MIVAC I indicated that there was a higher flare rate when methotrexate was stopped after both vaccine doses, but there was no difference in flare rate in MIVAC II when methotrexate was stopped only after the second dose as opposed to stopping it after both doses.
In the MIVAC I trial, 158 unvaccinated patients were randomized 1:1 to a cohort in which methotrexate was held for 2 weeks after both doses and a cohort in which methotrexate was continued despite the vaccine. In MIVAC II, 157 patients continued methotrexate while receiving the first vaccine dose. These patients were subsequently randomized either to continue or to stop methotrexate for 2 weeks following the second dose.
The findings from MIVAC I demonstrated the flare rate was lower in the methotrexate-continue group than in the methotrexate-pause group (8% vs. 25%; P = .005) and that the median anti-RBD titer was significantly higher for the methotrexate-pause group than the methotrexate-continue group (2,484 vs. 1,147; P = .001).
The results from MIVAC II trial indicated that there was no difference in flare rates between the two study groups (7.9% vs. 11.8%; P = .15). Yet, the median anti-RBD titer was significantly higher in the methotrexate-pause cohort than in the methotrexate-continue cohort (2,553 vs. 990; P = .001).
The report suggests there is a flare risk when methotrexate is stopped, Dr. Sreekanth noted. “It appears more logical to hold only after the second dose, as comparable anti-RBD titers are generated” with either approach, Dr. Sreekanth said.
Expert commentary: MIVAC I and II
Inés Colmegna, MD, associate professor at McGill University in Montreal, noted that it was intriguing that the risk of flares in MIVAC II is half of that reported after each of the doses of MIVAC I. “It is also worth emphasizing that despite the reported frequency of flares, the actual disease activity [as measured by the Disease Activity Score in 28 joints] in patients who did or did not withhold methotrexate was similar.
“MIVAC I and II have practical implications as they help to adequately inform patients about the risk and benefit trade of withholding methotrexate post–COVID-19 vaccination,” Dr. Colmegna told this news organization.
“Additional information would help to [further] interpret the findings of these studies, including whether any of the participants were taking any other DMARDs; data on the severity of the flares and functional impact; analysis of factors that predict the risk of flares, such as higher doses of methotrexate; [and change in] disease activity scores pre- and postvaccination,” Dr. Colmegna concluded.
Dr. Abhishek disclosed relationships with Springer, UpTodate, Oxford, Immunotec, AstraZeneca, Inflazome, NGM Biopharmaceuticals, Menarini Pharmaceuticals, and Cadila Pharmaceuticals. Dr. Abhishek is cochair of the ACR/EULAR CPPD Classification Criteria Working Group and the OMERACT CPPD Working Group. Dr. Sparks disclosed relationships with Gilead, Boehringer Ingelheim, Amgen, Bristol-Myers Squibb, and AbbVie, unrelated to this study. Dr. Tedeschi disclosed relationships with ModernaTx and NGM Biopharmaceuticals. Dr. Winthrop disclosed a research grant and serving as a scientific consultant for Pfizer. Dr. Sreekanth and Dr. Colmegna have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients who take methotrexate for a variety of immune-mediated inflammatory diseases and pause taking the drug following receipt of a COVID-19 vaccine dose did not have a higher risk of disease flare and had higher antireceptor binding domain (anti-RBD) antibody titers and increased immunogenicity when compared with continuing the drug, three recent studies suggest.
In one study, British researchers examined the effects of a 2-week break in methotrexate therapy on anti-RBD titers following receipt of a third COVID-19 vaccine dose. In their paper published in The Lancet: Respiratory Medicine, they reported results from a randomized, open-label, superiority trial that suggested pausing the drug improved immunogenicity, compared with no break.
In two trials presented at the European Alliance of Associations for Rheumatology (EULAR) 2022 Congress, a team from India set out to determine whether holding methotrexate after receiving both doses of a COVID-19 vaccine, or holding it only after the second dose, was safe and effective. They found that pausing methotrexate only following the second dose contributed to a lower flare risk, and that patients had higher anti-RBD titers when holding methotrexate for 2 weeks following each dose.
Pausing methotrexate after booster
The 2-week methotrexate break and booster vaccine dose data in the Vaccine Response On Off Methotrexate (VROOM) trial showed that after a month, the geometric mean antispike 1 (S1)-RBD antibody titer was 10,798 U/mL (95% confidence interval [CI], 8,970-12,997) in the group that continued methotrexate and 22,750 U/mL (95% CI, 19,314-26,796) in the group that suspended methotrexate; the geometric mean ratio was 2.19 (P < .0001; mixed-effects model), reported Abhishek Abhishek, MD, PhD, professor of rheumatology at the University of Nottingham in Nottingham, England, and colleagues.
Prior research showed that stopping methotrexate therapy for 2 weeks following the seasonal influenza vaccine contributed to better vaccine immunity among patients with rheumatoid arthritis, but there was no impact of stopping the drug for up to 4 weeks before vaccination on vaccine-related immunity, the researchers noted.
It is crucial in maximizing long-lasting vaccine protection in people who are possibly susceptible through immune suppression at this point in the COVID-19 vaccination regimen, the study team noted.
“Evidence from this study will be useful for policymakers, national immunization advisory committees, and specialist societies formulating recommendations on the use of methotrexate around the time of COVID-19 vaccination. This evidence will help patients and clinicians make informed choices about the risks and benefits of interrupting methotrexate treatment around the time of COVID-19 vaccination, with implications for the potential to extend such approaches to other therapeutics,” they wrote.
In American College of Rheumatology (ACR) guidance for COVID-19 vaccination, the organization advised against using standard synthetic disease-modifying antirheumatic medicines such as methotrexate “for 1-2 weeks (as disease activity allows) after each COVID-19 vaccine dose,” given the at-risk population and public health concerns, Jeffrey A. Sparks, MD, MMSc, assistant professor of medicine and associate physician at Brigham and Women’s Hospital and Harvard Medical School, Boston, and Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, noted in an accompanying editorial in The Lancet: Respiratory Medicine.
However, when the ACR developed this statement, there was only one trial involving patients with rheumatoid arthritis who paused methotrexate following seasonal influenza vaccination, the editorialists said.
“Although this finding adds to the evidence base to support interruption of methotrexate after vaccination, a shared decision process is needed to weigh the possible benefit of optimizing protection from COVID-19 and the possible risk of underlying disease flare,” they added.
Dr. Abhishek and colleagues assessed 254 patients with immune-mediated inflammatory disease from dermatology and rheumatology clinics across 26 hospitals in the United Kingdom. Participants had been diagnosed with systemic lupus erythematosus, rheumatoid arthritis, atopic dermatitis, polymyalgia rheumatica, axial spondyloarthritis, and psoriasis without or with arthritis. They had also been taking up to 25 mg of methotrexate per week for 3 months or longer and had received two doses of either the Pfizer/BioNTech BNT162b2 vaccine or AstraZeneca/Oxford viral vector vaccine. The booster dose was most often the Pfizer BNT162b2 vaccine (82%). The patients’ mean age was 59 years, with females comprising 61% of the cohort. Participants were randomly assigned 1:1 to either group.
Investigators performing laboratory analysis were masked to cohort assignment, and clinical research staff, data analysts, participants, and researchers were unmasked.
The elevated antibody response of patients who suspended methotrexate was the same across different kinds of immune-mediated inflammatory disease, primary vaccination platform, SARS-CoV-2 infection history, and age.
Notably, no intervention-associated adverse events were reported, the study team noted.
The conclusions that could be drawn from the booster-dose study were limited by the trial’s modest cohort size, the small number of patients in exploratory subgroup analyses, a lack of information about differences in prescription drug behavior, and early termination’s effect on the researchers’ ability to identify differences between subgroups and in secondary outcomes, the authors noted.
Other limitations included a lack of generalizability to patients with active disease who couldn’t stop therapy and were not included in the investigation, and participants were not blinded to what group they were in, the researchers said.
Expert commentary
This current study is consistent with other studies over the last several months showing that methotrexate harms both humoral and cell-mediated COVID-19 responses, noted Kevin Winthrop, MD, MPH, professor of infectious disease and public health at Oregon Health & Science University, Portland, who was not involved in the study. “And so now the new wave of studies are like this one, where they are holding methotrexate experimentally and seeing if it makes a difference,” he said.
“The one shortcoming of this study – and so far, the studies to date – is that no one has looked at whether the experimental hold has resulted in a change in T-cell responses, which ... we are [now] recognizing [the importance of] more and more in long-term protection, particularly in severe disease. Theoretically, holding [methotrexate] might help enhance T-cell responses, but that hasn’t been shown experimentally.”
Dr. Winthrop pointed out that one might get the same benefit from holding methotrexate for 1 week instead of 2 and that there likely is a reduced risk of flare-up from underlying autoimmune disease.
It is still not certain that this benefit extends to other vaccines, Dr. Winthrop noted. “It is probably true for most vaccines that if you hold methotrexate for 1 or 2 weeks, you might see some short-term benefit in responsiveness, but you don’t know that there is any clinical meaningfulness of this. That’s going to take other long-term studies. You don’t know how long this benefit lasts.”
Pausing methotrexate during initial COVID vaccine doses
Patients with either rheumatoid arthritis or psoriatic arthritis had higher anti-RBD antibody titers when methotrexate was stopped after both doses of the AstraZeneca vaccine, or simply after the second dose, than when methotrexate was continued, according to results from two single-center, randomized controlled trials called MIVAC I and II, Anu Sreekanth, MD, of Sree Sudheendra Medical Mission in Kochi, Kerala, India, and colleagues reported at EULAR 2022.
Results from MIVAC I indicated that there was a higher flare rate when methotrexate was stopped after both vaccine doses, but there was no difference in flare rate in MIVAC II when methotrexate was stopped only after the second dose as opposed to stopping it after both doses.
In the MIVAC I trial, 158 unvaccinated patients were randomized 1:1 to a cohort in which methotrexate was held for 2 weeks after both doses and a cohort in which methotrexate was continued despite the vaccine. In MIVAC II, 157 patients continued methotrexate while receiving the first vaccine dose. These patients were subsequently randomized either to continue or to stop methotrexate for 2 weeks following the second dose.
The findings from MIVAC I demonstrated the flare rate was lower in the methotrexate-continue group than in the methotrexate-pause group (8% vs. 25%; P = .005) and that the median anti-RBD titer was significantly higher for the methotrexate-pause group than the methotrexate-continue group (2,484 vs. 1,147; P = .001).
The results from MIVAC II trial indicated that there was no difference in flare rates between the two study groups (7.9% vs. 11.8%; P = .15). Yet, the median anti-RBD titer was significantly higher in the methotrexate-pause cohort than in the methotrexate-continue cohort (2,553 vs. 990; P = .001).
The report suggests there is a flare risk when methotrexate is stopped, Dr. Sreekanth noted. “It appears more logical to hold only after the second dose, as comparable anti-RBD titers are generated” with either approach, Dr. Sreekanth said.
Expert commentary: MIVAC I and II
Inés Colmegna, MD, associate professor at McGill University in Montreal, noted that it was intriguing that the risk of flares in MIVAC II is half of that reported after each of the doses of MIVAC I. “It is also worth emphasizing that despite the reported frequency of flares, the actual disease activity [as measured by the Disease Activity Score in 28 joints] in patients who did or did not withhold methotrexate was similar.
“MIVAC I and II have practical implications as they help to adequately inform patients about the risk and benefit trade of withholding methotrexate post–COVID-19 vaccination,” Dr. Colmegna told this news organization.
“Additional information would help to [further] interpret the findings of these studies, including whether any of the participants were taking any other DMARDs; data on the severity of the flares and functional impact; analysis of factors that predict the risk of flares, such as higher doses of methotrexate; [and change in] disease activity scores pre- and postvaccination,” Dr. Colmegna concluded.
Dr. Abhishek disclosed relationships with Springer, UpTodate, Oxford, Immunotec, AstraZeneca, Inflazome, NGM Biopharmaceuticals, Menarini Pharmaceuticals, and Cadila Pharmaceuticals. Dr. Abhishek is cochair of the ACR/EULAR CPPD Classification Criteria Working Group and the OMERACT CPPD Working Group. Dr. Sparks disclosed relationships with Gilead, Boehringer Ingelheim, Amgen, Bristol-Myers Squibb, and AbbVie, unrelated to this study. Dr. Tedeschi disclosed relationships with ModernaTx and NGM Biopharmaceuticals. Dr. Winthrop disclosed a research grant and serving as a scientific consultant for Pfizer. Dr. Sreekanth and Dr. Colmegna have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.







