User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Keep depression, anxiety screening top of mind in patients with psoriatic disease
DUBLIN – , warranting routine screening and having community contacts for mental health professional referrals, Elizabeth Wallace, MD, said at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
Dr. Wallace, of Cherry Hills Dermatology, Englewood, Colo., discussed the complex interactions between mental illness and psoriatic disease and the potential pitfalls of this comorbidity for these patients.
The topic of mental health is “consistently at the top of our patients’ minds, and certainly our minds too,” said session comoderator and GRAPPA president-elect Joseph F. Merola, MD, MMSc.
“In the U.S., around 17% of people with psoriasis have depression vs. 9% in those without psoriasis,” Dr. Wallace explained. “Psoriasis patients are twice as likely to have depression, compared to those without psoriasis, and psoriasis patients are 33% more likely to attempt suicide and 20% more likely to complete suicide, compared to those without psoriasis.” More severe psoriasis and younger age of onset are also associated with a greater likelihood of suicidality, she added.
Mediators of depression
“The inflammatory mechanisms driving PsD can drive depression and anxiety, and vice-versa,” she said. “There are often also genetic links, for example genetic variations in serotonin receptors, and psychological issues in psoriatic disease are predictably worsened by feelings of stigmatization, embarrassment, and social isolation.”
There are also efforts underway in clinics to “normalize” screening for anxiety and depression among this patient cohort, Dr. Wallace said. “We know that our psoriasis patients face social stigma from the visibility of their disease, and that stress can lead to flares of their condition,” she told the attendees. “We also know that patients who experience stigma also have an increased risk of depressive symptoms. We all know now that psoriasis has well-established pathways with upregulated proinflammatory cytokines.
“Increased cytokines stimulate indoleamine 2,3-dioxygenase, which converts tryptophan to kynurenine. Kynurenine is metabolized to quinolinic acid, which is neurotoxic.” She explained that because serotonin derives from tryptophan, decreases in tryptophan lead to reduced serotonin, and therefore increased risk of depression.
Interleukin-6 is known to be upregulated in depression and downregulated with the use of antidepressant medications, Dr. Wallace said. Mouse models in research have shown that deletion of the IL-6 gene produces antidepressant effects, and studies in humans have shown that IL-6, more than any other serum cytokine, is found at higher levels in humans with depression and psoriatic disease.
IL-17 is also implicated in psoriatic disease and mental health problems, Dr. Wallace said. “With stress, you get upregulation of the Tc17 cells, which produce IL-17,” she explained. “IL-17, along with other inflammatory markers, can actually make the blood-brain barrier more permeable, and when you get more permeability to the blood-brain barrier, you get these cytokines that can cross from the periphery and into the brain.
“With this crossing into the brain, you get further activation of more Th17 [cells] and that, on neurons, leads to increased potassium production, which is directly neurotoxic, so you get neuron destruction.”
Talking about depression
“So, what can we share with our patients?” Dr. Wallace asked. “We can discuss with them that psoriatic patients in general are more likely to be depressed or to have higher rates of suicide. The literature consistently shows that patients whose psoriasis is successfully treated experience reduced depression, and we can provide an understandable review of systemic medications, with warnings on depression and/or suicidality.”
Dr. Wallace advised to screen for depression with the Patient Health Questionnaire-2 (PHQ-2), a validated, two-item tool that asks, “Over the past 2 weeks, how often have you been bothered by having little interest or pleasure in doing things?” and “Over the past 2 weeks, how often have you been bothered by feeling down, depressed, or hopeless?”
She presented a case study illustrative of the type of presentation she sees in her clinic. It involved a 32-year-old man with plaque psoriasis and a high degree of body surface affected. “It’s now July in Colorado, it’s getting warm, people want to wear their shorts and T-shirts, but he said he could no longer hide his psoriasis,” said Dr. Wallace. “Further, it’s in areas that he cannot hide, such as his scalp, his beard, and he also has nail disease. Often, these patients don’t want to shake hands with their bosses or their colleagues and that’s very embarrassing for them.”
Dr. Wallace explained that this patient had seen advertisements for biologic drugs and requested to commence a treatment course. “During the exam, and now that you are developing some rapport with him, you discover that he is feeling down, is embarrassed at work, and has started to avoid social situations.” This is illustrative of a patient who should be screened for mental health conditions, specifically using PHQ-2, she said.
“You can be the person at the front line to screen these patients for mental health conditions, and, specifically for depression, with PHQ-2,” she said. PHQ-2 scores range from 0 to 6, and a score of 3 or higher is considered a positive screen.
“This is where your relationship with another health provider who is most qualified to care for these patients and validate them for their mental health condition can be absolutely critical,” Dr. Wallace said.
Successful PsD treatment lessens the risk for mental health comorbidities, and this is also seen in psoriatic arthritis, Dr. Wallace pointed out. Patient education is critical regarding their increased risk for depression and potential suicidal ideation, she added.
“It’s our job as clinicians to provide patients with an understandable, easy-to-digest review of systemic medications and warnings on depression and suicidality so that they can be aware of these factors.”
Perspective from Dr. Merola
In an interview, Dr. Merola, a double board-certified dermatologist and rheumatologist at Brigham and Women’s Hospital, Boston, discussed the interactions between mental and physical illness.
“One of the things we are learning is that it’s very much a multifactorial issue, in that skin and joints contribute, in some obvious ways, to anxiety and depression, like the fact that somebody doesn’t feel good about their appearance, or they can’t complete daily activities,” he said. “Those are the more obvious ones. But there is data and evidence that there is a biology behind that as well – inflammatory cytokines that drive skin disease probably also have a direct impact on the CNS and probably also drive anxiety and depression.
“We know that disordered sleep contributes to anxiety – think about how we feel if we get a horrible night’s sleep ... it’s hard to pick apart: ‘Am I depressed, am I anxious because I am having too much coffee? Because I am fatigued?’ So, we get into these circles, but the point is, we have to break these cycles, and we have to do it in multiple places. Yes, we have to fix the skin and the joints, but we also have to have interventions and think about how to screen for anxiety and depression. We also have to think about identifying disordered sleep, and how we intervene there as well.”
These challenges require a collaborative approach among physicians. “We can help patients to build their team that gets them help for their skin, for their joints, for their anxiety or depression, their disordered sleep, for their nutritional disorders, their obesity, and so on. So, we are trying to pick apart and unpack those complexities,” he said.
In regard to the potential impacts of this holistic strategy on physician workloads, Dr. Merola acknowledged it is important to consider physician wellness. “There’s no question that we want to be doing the best we can for our colleagues, but we don’t want to overload our colleagues by saying, ‘By the way, not only should we be treating their skin and joints,’ which of course we should be doing, but ‘could you also manage their diabetes, their obesity, their disordered sleep, their anxiety, their depression, difficulties with insurance, getting access to treatments, etc.’
“This is where effective collaboration between physicians becomes important,” he stressed. “We can’t manage every single piece, but we can make sure our patients are informed, are aware, and assist them to get the help that they need.”
In the United States, there “is a real issue” with access to mental health care and greater awareness needs to be created around this issue, he added.
Dr. Wallace and Dr. Merola report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
DUBLIN – , warranting routine screening and having community contacts for mental health professional referrals, Elizabeth Wallace, MD, said at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
Dr. Wallace, of Cherry Hills Dermatology, Englewood, Colo., discussed the complex interactions between mental illness and psoriatic disease and the potential pitfalls of this comorbidity for these patients.
The topic of mental health is “consistently at the top of our patients’ minds, and certainly our minds too,” said session comoderator and GRAPPA president-elect Joseph F. Merola, MD, MMSc.
“In the U.S., around 17% of people with psoriasis have depression vs. 9% in those without psoriasis,” Dr. Wallace explained. “Psoriasis patients are twice as likely to have depression, compared to those without psoriasis, and psoriasis patients are 33% more likely to attempt suicide and 20% more likely to complete suicide, compared to those without psoriasis.” More severe psoriasis and younger age of onset are also associated with a greater likelihood of suicidality, she added.
Mediators of depression
“The inflammatory mechanisms driving PsD can drive depression and anxiety, and vice-versa,” she said. “There are often also genetic links, for example genetic variations in serotonin receptors, and psychological issues in psoriatic disease are predictably worsened by feelings of stigmatization, embarrassment, and social isolation.”
There are also efforts underway in clinics to “normalize” screening for anxiety and depression among this patient cohort, Dr. Wallace said. “We know that our psoriasis patients face social stigma from the visibility of their disease, and that stress can lead to flares of their condition,” she told the attendees. “We also know that patients who experience stigma also have an increased risk of depressive symptoms. We all know now that psoriasis has well-established pathways with upregulated proinflammatory cytokines.
“Increased cytokines stimulate indoleamine 2,3-dioxygenase, which converts tryptophan to kynurenine. Kynurenine is metabolized to quinolinic acid, which is neurotoxic.” She explained that because serotonin derives from tryptophan, decreases in tryptophan lead to reduced serotonin, and therefore increased risk of depression.
Interleukin-6 is known to be upregulated in depression and downregulated with the use of antidepressant medications, Dr. Wallace said. Mouse models in research have shown that deletion of the IL-6 gene produces antidepressant effects, and studies in humans have shown that IL-6, more than any other serum cytokine, is found at higher levels in humans with depression and psoriatic disease.
IL-17 is also implicated in psoriatic disease and mental health problems, Dr. Wallace said. “With stress, you get upregulation of the Tc17 cells, which produce IL-17,” she explained. “IL-17, along with other inflammatory markers, can actually make the blood-brain barrier more permeable, and when you get more permeability to the blood-brain barrier, you get these cytokines that can cross from the periphery and into the brain.
“With this crossing into the brain, you get further activation of more Th17 [cells] and that, on neurons, leads to increased potassium production, which is directly neurotoxic, so you get neuron destruction.”
Talking about depression
“So, what can we share with our patients?” Dr. Wallace asked. “We can discuss with them that psoriatic patients in general are more likely to be depressed or to have higher rates of suicide. The literature consistently shows that patients whose psoriasis is successfully treated experience reduced depression, and we can provide an understandable review of systemic medications, with warnings on depression and/or suicidality.”
Dr. Wallace advised to screen for depression with the Patient Health Questionnaire-2 (PHQ-2), a validated, two-item tool that asks, “Over the past 2 weeks, how often have you been bothered by having little interest or pleasure in doing things?” and “Over the past 2 weeks, how often have you been bothered by feeling down, depressed, or hopeless?”
She presented a case study illustrative of the type of presentation she sees in her clinic. It involved a 32-year-old man with plaque psoriasis and a high degree of body surface affected. “It’s now July in Colorado, it’s getting warm, people want to wear their shorts and T-shirts, but he said he could no longer hide his psoriasis,” said Dr. Wallace. “Further, it’s in areas that he cannot hide, such as his scalp, his beard, and he also has nail disease. Often, these patients don’t want to shake hands with their bosses or their colleagues and that’s very embarrassing for them.”
Dr. Wallace explained that this patient had seen advertisements for biologic drugs and requested to commence a treatment course. “During the exam, and now that you are developing some rapport with him, you discover that he is feeling down, is embarrassed at work, and has started to avoid social situations.” This is illustrative of a patient who should be screened for mental health conditions, specifically using PHQ-2, she said.
“You can be the person at the front line to screen these patients for mental health conditions, and, specifically for depression, with PHQ-2,” she said. PHQ-2 scores range from 0 to 6, and a score of 3 or higher is considered a positive screen.
“This is where your relationship with another health provider who is most qualified to care for these patients and validate them for their mental health condition can be absolutely critical,” Dr. Wallace said.
Successful PsD treatment lessens the risk for mental health comorbidities, and this is also seen in psoriatic arthritis, Dr. Wallace pointed out. Patient education is critical regarding their increased risk for depression and potential suicidal ideation, she added.
“It’s our job as clinicians to provide patients with an understandable, easy-to-digest review of systemic medications and warnings on depression and suicidality so that they can be aware of these factors.”
Perspective from Dr. Merola
In an interview, Dr. Merola, a double board-certified dermatologist and rheumatologist at Brigham and Women’s Hospital, Boston, discussed the interactions between mental and physical illness.
“One of the things we are learning is that it’s very much a multifactorial issue, in that skin and joints contribute, in some obvious ways, to anxiety and depression, like the fact that somebody doesn’t feel good about their appearance, or they can’t complete daily activities,” he said. “Those are the more obvious ones. But there is data and evidence that there is a biology behind that as well – inflammatory cytokines that drive skin disease probably also have a direct impact on the CNS and probably also drive anxiety and depression.
“We know that disordered sleep contributes to anxiety – think about how we feel if we get a horrible night’s sleep ... it’s hard to pick apart: ‘Am I depressed, am I anxious because I am having too much coffee? Because I am fatigued?’ So, we get into these circles, but the point is, we have to break these cycles, and we have to do it in multiple places. Yes, we have to fix the skin and the joints, but we also have to have interventions and think about how to screen for anxiety and depression. We also have to think about identifying disordered sleep, and how we intervene there as well.”
These challenges require a collaborative approach among physicians. “We can help patients to build their team that gets them help for their skin, for their joints, for their anxiety or depression, their disordered sleep, for their nutritional disorders, their obesity, and so on. So, we are trying to pick apart and unpack those complexities,” he said.
In regard to the potential impacts of this holistic strategy on physician workloads, Dr. Merola acknowledged it is important to consider physician wellness. “There’s no question that we want to be doing the best we can for our colleagues, but we don’t want to overload our colleagues by saying, ‘By the way, not only should we be treating their skin and joints,’ which of course we should be doing, but ‘could you also manage their diabetes, their obesity, their disordered sleep, their anxiety, their depression, difficulties with insurance, getting access to treatments, etc.’
“This is where effective collaboration between physicians becomes important,” he stressed. “We can’t manage every single piece, but we can make sure our patients are informed, are aware, and assist them to get the help that they need.”
In the United States, there “is a real issue” with access to mental health care and greater awareness needs to be created around this issue, he added.
Dr. Wallace and Dr. Merola report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
DUBLIN – , warranting routine screening and having community contacts for mental health professional referrals, Elizabeth Wallace, MD, said at the annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
Dr. Wallace, of Cherry Hills Dermatology, Englewood, Colo., discussed the complex interactions between mental illness and psoriatic disease and the potential pitfalls of this comorbidity for these patients.
The topic of mental health is “consistently at the top of our patients’ minds, and certainly our minds too,” said session comoderator and GRAPPA president-elect Joseph F. Merola, MD, MMSc.
“In the U.S., around 17% of people with psoriasis have depression vs. 9% in those without psoriasis,” Dr. Wallace explained. “Psoriasis patients are twice as likely to have depression, compared to those without psoriasis, and psoriasis patients are 33% more likely to attempt suicide and 20% more likely to complete suicide, compared to those without psoriasis.” More severe psoriasis and younger age of onset are also associated with a greater likelihood of suicidality, she added.
Mediators of depression
“The inflammatory mechanisms driving PsD can drive depression and anxiety, and vice-versa,” she said. “There are often also genetic links, for example genetic variations in serotonin receptors, and psychological issues in psoriatic disease are predictably worsened by feelings of stigmatization, embarrassment, and social isolation.”
There are also efforts underway in clinics to “normalize” screening for anxiety and depression among this patient cohort, Dr. Wallace said. “We know that our psoriasis patients face social stigma from the visibility of their disease, and that stress can lead to flares of their condition,” she told the attendees. “We also know that patients who experience stigma also have an increased risk of depressive symptoms. We all know now that psoriasis has well-established pathways with upregulated proinflammatory cytokines.
“Increased cytokines stimulate indoleamine 2,3-dioxygenase, which converts tryptophan to kynurenine. Kynurenine is metabolized to quinolinic acid, which is neurotoxic.” She explained that because serotonin derives from tryptophan, decreases in tryptophan lead to reduced serotonin, and therefore increased risk of depression.
Interleukin-6 is known to be upregulated in depression and downregulated with the use of antidepressant medications, Dr. Wallace said. Mouse models in research have shown that deletion of the IL-6 gene produces antidepressant effects, and studies in humans have shown that IL-6, more than any other serum cytokine, is found at higher levels in humans with depression and psoriatic disease.
IL-17 is also implicated in psoriatic disease and mental health problems, Dr. Wallace said. “With stress, you get upregulation of the Tc17 cells, which produce IL-17,” she explained. “IL-17, along with other inflammatory markers, can actually make the blood-brain barrier more permeable, and when you get more permeability to the blood-brain barrier, you get these cytokines that can cross from the periphery and into the brain.
“With this crossing into the brain, you get further activation of more Th17 [cells] and that, on neurons, leads to increased potassium production, which is directly neurotoxic, so you get neuron destruction.”
Talking about depression
“So, what can we share with our patients?” Dr. Wallace asked. “We can discuss with them that psoriatic patients in general are more likely to be depressed or to have higher rates of suicide. The literature consistently shows that patients whose psoriasis is successfully treated experience reduced depression, and we can provide an understandable review of systemic medications, with warnings on depression and/or suicidality.”
Dr. Wallace advised to screen for depression with the Patient Health Questionnaire-2 (PHQ-2), a validated, two-item tool that asks, “Over the past 2 weeks, how often have you been bothered by having little interest or pleasure in doing things?” and “Over the past 2 weeks, how often have you been bothered by feeling down, depressed, or hopeless?”
She presented a case study illustrative of the type of presentation she sees in her clinic. It involved a 32-year-old man with plaque psoriasis and a high degree of body surface affected. “It’s now July in Colorado, it’s getting warm, people want to wear their shorts and T-shirts, but he said he could no longer hide his psoriasis,” said Dr. Wallace. “Further, it’s in areas that he cannot hide, such as his scalp, his beard, and he also has nail disease. Often, these patients don’t want to shake hands with their bosses or their colleagues and that’s very embarrassing for them.”
Dr. Wallace explained that this patient had seen advertisements for biologic drugs and requested to commence a treatment course. “During the exam, and now that you are developing some rapport with him, you discover that he is feeling down, is embarrassed at work, and has started to avoid social situations.” This is illustrative of a patient who should be screened for mental health conditions, specifically using PHQ-2, she said.
“You can be the person at the front line to screen these patients for mental health conditions, and, specifically for depression, with PHQ-2,” she said. PHQ-2 scores range from 0 to 6, and a score of 3 or higher is considered a positive screen.
“This is where your relationship with another health provider who is most qualified to care for these patients and validate them for their mental health condition can be absolutely critical,” Dr. Wallace said.
Successful PsD treatment lessens the risk for mental health comorbidities, and this is also seen in psoriatic arthritis, Dr. Wallace pointed out. Patient education is critical regarding their increased risk for depression and potential suicidal ideation, she added.
“It’s our job as clinicians to provide patients with an understandable, easy-to-digest review of systemic medications and warnings on depression and suicidality so that they can be aware of these factors.”
Perspective from Dr. Merola
In an interview, Dr. Merola, a double board-certified dermatologist and rheumatologist at Brigham and Women’s Hospital, Boston, discussed the interactions between mental and physical illness.
“One of the things we are learning is that it’s very much a multifactorial issue, in that skin and joints contribute, in some obvious ways, to anxiety and depression, like the fact that somebody doesn’t feel good about their appearance, or they can’t complete daily activities,” he said. “Those are the more obvious ones. But there is data and evidence that there is a biology behind that as well – inflammatory cytokines that drive skin disease probably also have a direct impact on the CNS and probably also drive anxiety and depression.
“We know that disordered sleep contributes to anxiety – think about how we feel if we get a horrible night’s sleep ... it’s hard to pick apart: ‘Am I depressed, am I anxious because I am having too much coffee? Because I am fatigued?’ So, we get into these circles, but the point is, we have to break these cycles, and we have to do it in multiple places. Yes, we have to fix the skin and the joints, but we also have to have interventions and think about how to screen for anxiety and depression. We also have to think about identifying disordered sleep, and how we intervene there as well.”
These challenges require a collaborative approach among physicians. “We can help patients to build their team that gets them help for their skin, for their joints, for their anxiety or depression, their disordered sleep, for their nutritional disorders, their obesity, and so on. So, we are trying to pick apart and unpack those complexities,” he said.
In regard to the potential impacts of this holistic strategy on physician workloads, Dr. Merola acknowledged it is important to consider physician wellness. “There’s no question that we want to be doing the best we can for our colleagues, but we don’t want to overload our colleagues by saying, ‘By the way, not only should we be treating their skin and joints,’ which of course we should be doing, but ‘could you also manage their diabetes, their obesity, their disordered sleep, their anxiety, their depression, difficulties with insurance, getting access to treatments, etc.’
“This is where effective collaboration between physicians becomes important,” he stressed. “We can’t manage every single piece, but we can make sure our patients are informed, are aware, and assist them to get the help that they need.”
In the United States, there “is a real issue” with access to mental health care and greater awareness needs to be created around this issue, he added.
Dr. Wallace and Dr. Merola report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT GRAPPA 2023
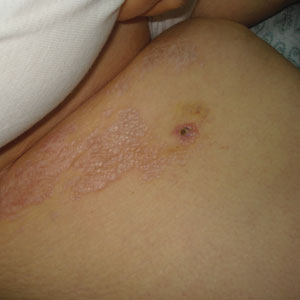
Porcelain White, Crinkled, Violaceous Patches on the Inner Thighs
The Diagnosis: Extragenital Lichen Sclerosus et Atrophicus
A punch biopsy of the lesion revealed epidermal hyperkeratosis, atrophy, follicular plugs with basal vacuolar degeneration, and homogenous dense fibrosis in the papillary dermis with a dense lymphocytic infiltrate beneath the fibrosis (Figure 1). Dermoscopic examination was remarkable for a distinctive rainbow pattern. Clinical, histopathologic, and dermoscopic findings led to the diagnosis of extragenital lichen sclerosus et atrophicus (LSEA). A potent corticosteroid cream was prescribed twice daily for 2 months, after which the lesions completely resolved. At 2-year follow-up, a relapse was not observed (Figure 2).
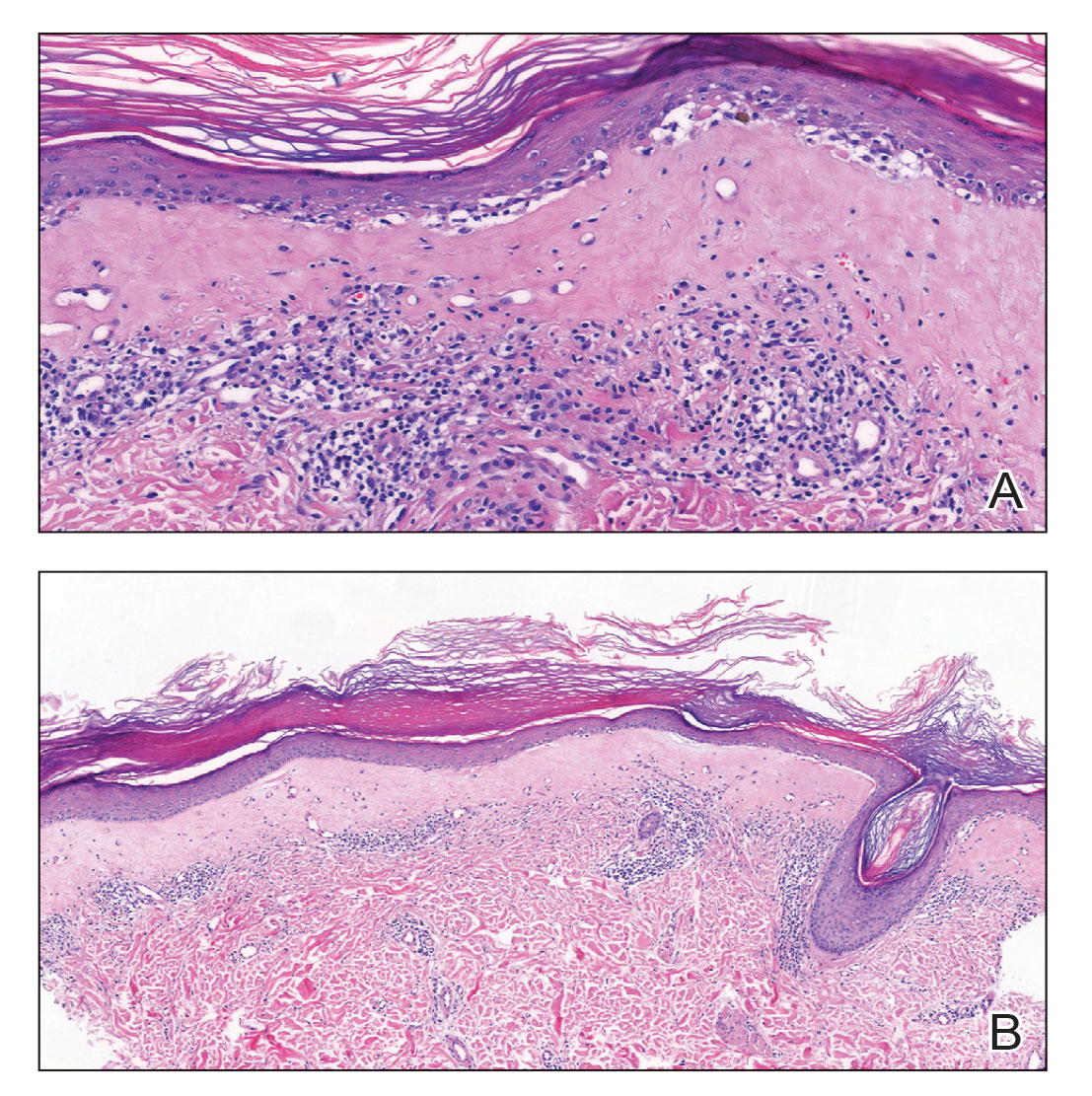
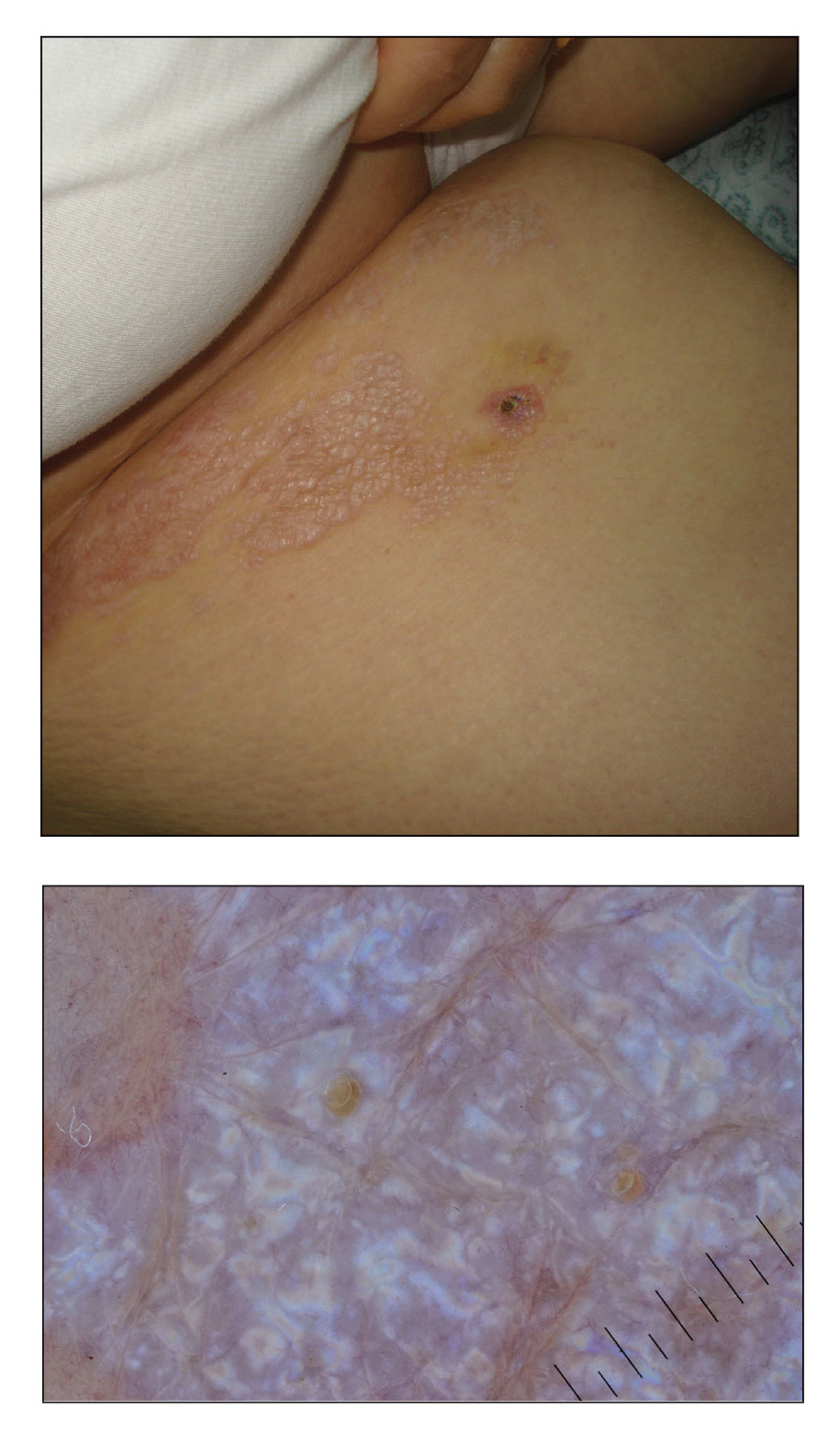
Lichen sclerosus et atrophicus is an inflammatory dermatosis that clinically presents as atrophic or hypertrophic plaques that may show pigmentation changes with anogenital and extragenital involvement. It is common among females and predominantly occurs in prepubescent girls and postmenopausal women. The exact etiology is unclear; however, it is hypothesized to occur secondary to autoimmunity with an underlying genetic predisposition. Local trauma, hormonal influences, and infections are other suspected etiologic factors. Genital lesions often lead to itching, pain, and dyspareunia, whereas extragenital lesions predominantly are asymptomatic. When symptomatic, itching usually is the main concern. Unlike genital LSEA, extragenital lesions are not associated with squamous cell carcinoma development. Reported dermoscopic features of LSEA are white structureless areas with scaling, comedolike openings, follicular plugs, white shiny streaks, blue-gray peppering, pigment network, and red-purple globules.1 In our case, the dermoscopic finding of a rainbow pattern in LSEA is rare.2 Although the mechanism behind this appearance unclear, it can be the result of the birefringence effect—local variations in refractive index—influenced by the direction of structures within the dermis such as collagen. In this case, there was diffuse and dense homogenous fibrosis in the superficial dermis that corresponded to dermoscopic white polygonal clods.

Extragenital LSEA commonly is located on the neck, shoulders, wrists, and upper trunk and manifests clinically as whitish papules coalescing into scarlike plaques. Of all patients who have LSEA, 20% have extragenital lesions, and most of these lesions are seen in patients who also have genital LSEA. Approximately 6% of all LSEA patients have extragenital LSEA without genital involvement.3
For experienced dermatologists, clinical symptoms and lesion characteristics usually are sufficient for diagnosis; however, a differential diagnosis of atypical lesions and isolated extragenital presentations such as morphea, lichen simplex chronicus, lichen planus, and vitiligo requires the correlation of clinical findings with histopathology and dermoscopy. Morphea, known as localized scleroderma, is an idiopathic inflammatory skin disease with sclerotic changes. It manifests as inflammatory plaques that vary in color from red to purple. If there is moderate sclerosis in the center of this plaque, the color progressively fades to white, leaving a purplish ring around the edges. Dermoscopic features of morphea are reported as areas of erythema; red-focused vessels of linear, irregular, or dotted morphology; white fibrotic beams; and pigmentary structures.2 Lichen simplex chronicus is characterized by single or multiple dry and patchy skin lesions that are intensely pruritic. It commonly occurs on the neck, scalp, extremities, genital areas, and buttocks. Scratching the lesions leads to scarring, thickening of the skin, and increased frequency of itching. Histopathology of lichen simplex chronicus most frequently demonstrates a thickening of the epidermis and papillary dermis, irregularly elongated rete ridges, and fibroplasia with stellate or multinucleated fibroblasts completed by perivascular lymphocytic inflammation.4 Lichen planus presents with pruritic, polygonal, purple papules and/or plaques that can present in a variety of clinical forms, including atrophic and hypertrophic lichen planus.5 Lichen planus was an unlikely diagnosis for our patient due to the presence of patchy scarlike lesions and dermoscopic features that are well described in patients with LSEA. Lichen sclerosus et atrophicus presents with hypopigmented and/or hyperpigmented patches and plaques, distinguishing itself from vitiligo, which has flat lesions.
Topical steroids are the first-line therapeutic agents in the treatment of LSEA.6 Despite frequent use in this setting, common side effects such as localized scarring and atrophic degenerations have led to debate about their use. In our patient, the lesions resolved almost completely in 2 months, and no relapse was observed in the following 2 years. In the setting of topical steroid resistance, topical calcineurin inhibitors, UVA/UVB phototherapy, and topical tacrolimus can be used for treatment.6
The diagnosis of isolated extragenital LSEA may be a clinical challenge and generally requires further workup. When evaluating extragenital lesions, dermatologists should keep in mind extragenital LSEA as a differential diagnosis in the presence of a dermoscopic rainbow pattern arranged over white polygonal clods.
- Wang Y-K, Hao J-C, Liu J, et al. Dermoscopic features of morphea and extragenital lichen sclerosus in Chinese patients. Chin Med J (Engl). 2020;133:2109-2111.
- Errichetti E, Lallas A, Apalla Z, et al. Dermoscopy of morphea and cutaneous lichen sclerosus: clinicopathological correlation study and comparative analysis. Dermatology. 2017;233:462-470.
- Wallace HJ. Lichen sclerosus et atrophicus. Trans St Johns Hosp Dermatol Soc. 1971;57:9-30.
- Balan R, Grigoras¸ A, Popovici D, et al. The histopathological landscape of the major psoriasiform dermatoses. Arch Clin Cases. 2021;6:59-68.
- Weston G, Payette M. Update on lichen planus and its clinical variants. Int J Womens Dermatol. 2015;1:140-149.
- Kirtschig G, Becker K, Günthert A, et al. Evidence-based (S3) guideline on (anogenital) lichen sclerosus. J Eur Acad Dermatol Venereol. 2015;29:E1-E43.
The Diagnosis: Extragenital Lichen Sclerosus et Atrophicus
A punch biopsy of the lesion revealed epidermal hyperkeratosis, atrophy, follicular plugs with basal vacuolar degeneration, and homogenous dense fibrosis in the papillary dermis with a dense lymphocytic infiltrate beneath the fibrosis (Figure 1). Dermoscopic examination was remarkable for a distinctive rainbow pattern. Clinical, histopathologic, and dermoscopic findings led to the diagnosis of extragenital lichen sclerosus et atrophicus (LSEA). A potent corticosteroid cream was prescribed twice daily for 2 months, after which the lesions completely resolved. At 2-year follow-up, a relapse was not observed (Figure 2).

Lichen sclerosus et atrophicus is an inflammatory dermatosis that clinically presents as atrophic or hypertrophic plaques that may show pigmentation changes with anogenital and extragenital involvement. It is common among females and predominantly occurs in prepubescent girls and postmenopausal women. The exact etiology is unclear; however, it is hypothesized to occur secondary to autoimmunity with an underlying genetic predisposition. Local trauma, hormonal influences, and infections are other suspected etiologic factors. Genital lesions often lead to itching, pain, and dyspareunia, whereas extragenital lesions predominantly are asymptomatic. When symptomatic, itching usually is the main concern. Unlike genital LSEA, extragenital lesions are not associated with squamous cell carcinoma development. Reported dermoscopic features of LSEA are white structureless areas with scaling, comedolike openings, follicular plugs, white shiny streaks, blue-gray peppering, pigment network, and red-purple globules.1 In our case, the dermoscopic finding of a rainbow pattern in LSEA is rare.2 Although the mechanism behind this appearance unclear, it can be the result of the birefringence effect—local variations in refractive index—influenced by the direction of structures within the dermis such as collagen. In this case, there was diffuse and dense homogenous fibrosis in the superficial dermis that corresponded to dermoscopic white polygonal clods.

Extragenital LSEA commonly is located on the neck, shoulders, wrists, and upper trunk and manifests clinically as whitish papules coalescing into scarlike plaques. Of all patients who have LSEA, 20% have extragenital lesions, and most of these lesions are seen in patients who also have genital LSEA. Approximately 6% of all LSEA patients have extragenital LSEA without genital involvement.3
For experienced dermatologists, clinical symptoms and lesion characteristics usually are sufficient for diagnosis; however, a differential diagnosis of atypical lesions and isolated extragenital presentations such as morphea, lichen simplex chronicus, lichen planus, and vitiligo requires the correlation of clinical findings with histopathology and dermoscopy. Morphea, known as localized scleroderma, is an idiopathic inflammatory skin disease with sclerotic changes. It manifests as inflammatory plaques that vary in color from red to purple. If there is moderate sclerosis in the center of this plaque, the color progressively fades to white, leaving a purplish ring around the edges. Dermoscopic features of morphea are reported as areas of erythema; red-focused vessels of linear, irregular, or dotted morphology; white fibrotic beams; and pigmentary structures.2 Lichen simplex chronicus is characterized by single or multiple dry and patchy skin lesions that are intensely pruritic. It commonly occurs on the neck, scalp, extremities, genital areas, and buttocks. Scratching the lesions leads to scarring, thickening of the skin, and increased frequency of itching. Histopathology of lichen simplex chronicus most frequently demonstrates a thickening of the epidermis and papillary dermis, irregularly elongated rete ridges, and fibroplasia with stellate or multinucleated fibroblasts completed by perivascular lymphocytic inflammation.4 Lichen planus presents with pruritic, polygonal, purple papules and/or plaques that can present in a variety of clinical forms, including atrophic and hypertrophic lichen planus.5 Lichen planus was an unlikely diagnosis for our patient due to the presence of patchy scarlike lesions and dermoscopic features that are well described in patients with LSEA. Lichen sclerosus et atrophicus presents with hypopigmented and/or hyperpigmented patches and plaques, distinguishing itself from vitiligo, which has flat lesions.
Topical steroids are the first-line therapeutic agents in the treatment of LSEA.6 Despite frequent use in this setting, common side effects such as localized scarring and atrophic degenerations have led to debate about their use. In our patient, the lesions resolved almost completely in 2 months, and no relapse was observed in the following 2 years. In the setting of topical steroid resistance, topical calcineurin inhibitors, UVA/UVB phototherapy, and topical tacrolimus can be used for treatment.6
The diagnosis of isolated extragenital LSEA may be a clinical challenge and generally requires further workup. When evaluating extragenital lesions, dermatologists should keep in mind extragenital LSEA as a differential diagnosis in the presence of a dermoscopic rainbow pattern arranged over white polygonal clods.
The Diagnosis: Extragenital Lichen Sclerosus et Atrophicus
A punch biopsy of the lesion revealed epidermal hyperkeratosis, atrophy, follicular plugs with basal vacuolar degeneration, and homogenous dense fibrosis in the papillary dermis with a dense lymphocytic infiltrate beneath the fibrosis (Figure 1). Dermoscopic examination was remarkable for a distinctive rainbow pattern. Clinical, histopathologic, and dermoscopic findings led to the diagnosis of extragenital lichen sclerosus et atrophicus (LSEA). A potent corticosteroid cream was prescribed twice daily for 2 months, after which the lesions completely resolved. At 2-year follow-up, a relapse was not observed (Figure 2).

Lichen sclerosus et atrophicus is an inflammatory dermatosis that clinically presents as atrophic or hypertrophic plaques that may show pigmentation changes with anogenital and extragenital involvement. It is common among females and predominantly occurs in prepubescent girls and postmenopausal women. The exact etiology is unclear; however, it is hypothesized to occur secondary to autoimmunity with an underlying genetic predisposition. Local trauma, hormonal influences, and infections are other suspected etiologic factors. Genital lesions often lead to itching, pain, and dyspareunia, whereas extragenital lesions predominantly are asymptomatic. When symptomatic, itching usually is the main concern. Unlike genital LSEA, extragenital lesions are not associated with squamous cell carcinoma development. Reported dermoscopic features of LSEA are white structureless areas with scaling, comedolike openings, follicular plugs, white shiny streaks, blue-gray peppering, pigment network, and red-purple globules.1 In our case, the dermoscopic finding of a rainbow pattern in LSEA is rare.2 Although the mechanism behind this appearance unclear, it can be the result of the birefringence effect—local variations in refractive index—influenced by the direction of structures within the dermis such as collagen. In this case, there was diffuse and dense homogenous fibrosis in the superficial dermis that corresponded to dermoscopic white polygonal clods.

Extragenital LSEA commonly is located on the neck, shoulders, wrists, and upper trunk and manifests clinically as whitish papules coalescing into scarlike plaques. Of all patients who have LSEA, 20% have extragenital lesions, and most of these lesions are seen in patients who also have genital LSEA. Approximately 6% of all LSEA patients have extragenital LSEA without genital involvement.3
For experienced dermatologists, clinical symptoms and lesion characteristics usually are sufficient for diagnosis; however, a differential diagnosis of atypical lesions and isolated extragenital presentations such as morphea, lichen simplex chronicus, lichen planus, and vitiligo requires the correlation of clinical findings with histopathology and dermoscopy. Morphea, known as localized scleroderma, is an idiopathic inflammatory skin disease with sclerotic changes. It manifests as inflammatory plaques that vary in color from red to purple. If there is moderate sclerosis in the center of this plaque, the color progressively fades to white, leaving a purplish ring around the edges. Dermoscopic features of morphea are reported as areas of erythema; red-focused vessels of linear, irregular, or dotted morphology; white fibrotic beams; and pigmentary structures.2 Lichen simplex chronicus is characterized by single or multiple dry and patchy skin lesions that are intensely pruritic. It commonly occurs on the neck, scalp, extremities, genital areas, and buttocks. Scratching the lesions leads to scarring, thickening of the skin, and increased frequency of itching. Histopathology of lichen simplex chronicus most frequently demonstrates a thickening of the epidermis and papillary dermis, irregularly elongated rete ridges, and fibroplasia with stellate or multinucleated fibroblasts completed by perivascular lymphocytic inflammation.4 Lichen planus presents with pruritic, polygonal, purple papules and/or plaques that can present in a variety of clinical forms, including atrophic and hypertrophic lichen planus.5 Lichen planus was an unlikely diagnosis for our patient due to the presence of patchy scarlike lesions and dermoscopic features that are well described in patients with LSEA. Lichen sclerosus et atrophicus presents with hypopigmented and/or hyperpigmented patches and plaques, distinguishing itself from vitiligo, which has flat lesions.
Topical steroids are the first-line therapeutic agents in the treatment of LSEA.6 Despite frequent use in this setting, common side effects such as localized scarring and atrophic degenerations have led to debate about their use. In our patient, the lesions resolved almost completely in 2 months, and no relapse was observed in the following 2 years. In the setting of topical steroid resistance, topical calcineurin inhibitors, UVA/UVB phototherapy, and topical tacrolimus can be used for treatment.6
The diagnosis of isolated extragenital LSEA may be a clinical challenge and generally requires further workup. When evaluating extragenital lesions, dermatologists should keep in mind extragenital LSEA as a differential diagnosis in the presence of a dermoscopic rainbow pattern arranged over white polygonal clods.
- Wang Y-K, Hao J-C, Liu J, et al. Dermoscopic features of morphea and extragenital lichen sclerosus in Chinese patients. Chin Med J (Engl). 2020;133:2109-2111.
- Errichetti E, Lallas A, Apalla Z, et al. Dermoscopy of morphea and cutaneous lichen sclerosus: clinicopathological correlation study and comparative analysis. Dermatology. 2017;233:462-470.
- Wallace HJ. Lichen sclerosus et atrophicus. Trans St Johns Hosp Dermatol Soc. 1971;57:9-30.
- Balan R, Grigoras¸ A, Popovici D, et al. The histopathological landscape of the major psoriasiform dermatoses. Arch Clin Cases. 2021;6:59-68.
- Weston G, Payette M. Update on lichen planus and its clinical variants. Int J Womens Dermatol. 2015;1:140-149.
- Kirtschig G, Becker K, Günthert A, et al. Evidence-based (S3) guideline on (anogenital) lichen sclerosus. J Eur Acad Dermatol Venereol. 2015;29:E1-E43.
- Wang Y-K, Hao J-C, Liu J, et al. Dermoscopic features of morphea and extragenital lichen sclerosus in Chinese patients. Chin Med J (Engl). 2020;133:2109-2111.
- Errichetti E, Lallas A, Apalla Z, et al. Dermoscopy of morphea and cutaneous lichen sclerosus: clinicopathological correlation study and comparative analysis. Dermatology. 2017;233:462-470.
- Wallace HJ. Lichen sclerosus et atrophicus. Trans St Johns Hosp Dermatol Soc. 1971;57:9-30.
- Balan R, Grigoras¸ A, Popovici D, et al. The histopathological landscape of the major psoriasiform dermatoses. Arch Clin Cases. 2021;6:59-68.
- Weston G, Payette M. Update on lichen planus and its clinical variants. Int J Womens Dermatol. 2015;1:140-149.
- Kirtschig G, Becker K, Günthert A, et al. Evidence-based (S3) guideline on (anogenital) lichen sclerosus. J Eur Acad Dermatol Venereol. 2015;29:E1-E43.
A 50-year-old woman presented with multiple pruritic lesions on the right inner thigh of 2 years’ duration. Physical examination revealed porcelain white, crinkled, violaceous patches extending from the right inner thigh to the inguinal fold (top). Dermoscopic examination revealed follicular plugs, white structureless areas, white lines, and a rainbow pattern arranged over white polygonal clods on polarized mode (bottom).
For love or money: How do doctors choose their specialty?
Medical student loans top hundreds of thousands of dollars, so it’s understandable that physicians may want to select a specialty that pays well.
Moreover, most advised young doctors to follow their hearts rather than their wallets.
“There is no question that many young kids immediately think about money when deciding to pursue medicine, but the thought of a big paycheck will never sustain someone long enough to get them here,” says Sergio Alvarez, MD, a board-certified plastic surgeon based in Miami, Fla., and the CEO and medical director of Mia Aesthetics, which has several national locations.
“Getting into medicine is a long game, and there are many hurdles along the way that only the dedicated overcome,” says Dr. Alvarez.
Unfortunately, he says it may be late in that long game before some realize that the pay rate for certain specialties isn’t commensurate with the immense workload and responsibility they require.
“The short of it is that to become a happy doctor, medicine really needs to be a calling: a passion! There are far easier things to do to make money.”
Here is what physicians said about choosing between love or money.
The lowest-paying subspecialty in a low-paying specialty
Sophia Yen, MD, MPH, cofounder and CEO of Pandia Health, a women-founded, doctor-led birth control delivery service in Sunnyvale, Calif., and clinical associate professor at Stanford (Calif.) University, says you should pursue a specialty because you love the work.
“I chose the lowest-paying subspecialty (adolescent medicine) of a low-paying specialty (pediatrics), but I’d do it all again because I love the patient population – I love what I do.”
Dr. Yen says she chose adolescent medicine because she loves doing “outpatient gynecology” without going through the surgical training of a full ob.gyn. “I love the target population of young adults because you can talk to the patient versus in pediatrics, where you often talk to the parent. With young adults you can catch things – for example, teach a young person about consent, alcohol, marijuana’s effects on the growing brain, prevent unplanned pregnancies and sexually transmitted infections, instill healthy eating, and more.
“Do I wish that I got paid as much as a surgeon?” Dr. Yen says yes. “I hope that someday society will realize the time spent preventing future disease is worth it and pay us accordingly.”
Unfortunately, she says, since the health care system makes more money if you get pregnant, need a cardiac bypass, or need gastric surgery, those who deliver babies or do surgery get paid more than someone who prevents the need for those services.
Money doesn’t buy happiness
Stella Bard, MD, a rheumatologist in McKinney, Tex., says she eats, lives, and breathes rheumatology. “I never regret the decision of choosing this specialty for a single second,” says Dr. Bard. “I feel like it’s a rewarding experience with every single patient encounter.” Dr. Bard notes that money is no guarantee of happiness and that she feels blessed to wake up every morning doing what she loves.
Career or calling?
For Dr. Alvarez, inspiration came when watching his father help change people’s lives. “I saw how impactful a doctor is during a person’s most desperate moments, and that was enough to make medicine my life’s passion at the age of 10.”
He says once you’re in medical school, choosing a specialty is far easier than you think. “Each specialty requires a certain personality or specific characteristics, and some will call to you while others simply won’t.”
“For me, plastics was about finesse, art, and life-changing surgeries that affected people from kids to adults and involved every aspect of the human body. Changing someone’s outward appearance has a profoundly positive impact on their confidence and self-esteem, making plastic surgery a genuinely transformative experience.”
Patricia Celan, MD, a postgraduate psychiatry resident in Canada, also chose psychiatry for the love of the field. “I enjoy helping vulnerable people and exploring what makes a person tick, the source of their difficulties, and how to help people counteract and overcome the difficult cards they’ve been dealt in life.”
She says it’s incredibly rewarding to watch someone turn their life around from severe mental illness, especially those who have been victimized and traumatized, and learn to trust people again.
“I could have made more money in a higher-paying specialty, yes, but I’m not sure I would have felt as fulfilled as psychiatry can make me feel.”
Dr. Celan says everyone has their calling, and some lucky people find their deepest passion in higher-paying specialties. “My calling is psychiatry, and I am at peace with this no matter the money.”
For the love of surgery
“In my experience, most people don’t choose their specialty based on money,” says Nicole Aaronson, MD, MBA, an otolaryngologist and board-certified in the subspecialty of pediatric otolaryngology, an attending surgeon at Nemours Children’s Health of Delaware and clinical associate professor of otolaryngology and pediatrics at Sidney Kimmel Medical College, Philadelphia.
“The first decision point in medical school is usually figuring out if you are a surgery person or a medicine person. I knew very early that I wanted to be a surgeon and wanted to spend time in the OR fixing problems with my hands.”
Part of what attracted Dr. Aaronson to otolaryngology was the variety of conditions managed within the specialty, from head and neck cancer to voice problems to sleep disorders to sinus disease. “I chose my subspecialty because I enjoy working with children and making an impact that will help them live their best possible lives.”
She says a relatively simple surgery like placing ear tubes may help a child’s hearing and allow them to be more successful in school, opening up a new world of opportunities for the child’s future.
“While I don’t think most people choose their specialty based on prospective compensation, I do think all physicians want to be compensated fairly for their time, effort, and level of training,” says Dr. Aaronson.
Choosing a specialty for the money can lead to burnout and dissatisfaction
“For me, the decision to pursue gastroenterology went beyond financial considerations,” says Saurabh Sethi, MD, MPH, a gastroenterologist specializing in hepatology and interventional endoscopy. “While financial stability is undoubtedly important, no doctor enters this field solely for the love of money. The primary driving force for most medical professionals, myself included, is the passion to help people and make a positive difference in their lives.”
Dr. Sethi says the gratification that comes from providing quality care and witnessing patients’ improved well-being is priceless. Moreover, he believes that selecting a specialty based solely on financial gain is likely to lead to burnout and greater dissatisfaction over time.
“By following my love for gut health and prioritizing patient care, I have found a sense of fulfillment and purpose in my career. It has been a rewarding journey, and I’m grateful for the opportunity to contribute to the well-being of my patients through my expertise in gastroenterology.”
Key takeaways: Love or money?
Multiple factors influence doctors’ specialty choices, including genuine love for the work and the future of the specialty. Others include job prospects, hands-on experience they receive, mentors, childhood dreams, parental expectations, complexity of cases, the lifestyle of each specialty, including office hours worked, on-call requirements, and autonomy.
Physicians also mentioned other factors they considered when choosing their specialty:
- Personal interest.
- Intellectual stimulation.
- Work-life balance.
- Patient populations.
- Future opportunities.
- Desire to make a difference.
- Passion.
- Financial stability.
- Being personally fulfilled.
Overwhelmingly, doctors say to pick a specialty you can envision yourself loving 40 years from now and you won’t go wrong.
A version of this article first appeared on Medscape.com.
Medical student loans top hundreds of thousands of dollars, so it’s understandable that physicians may want to select a specialty that pays well.
Moreover, most advised young doctors to follow their hearts rather than their wallets.
“There is no question that many young kids immediately think about money when deciding to pursue medicine, but the thought of a big paycheck will never sustain someone long enough to get them here,” says Sergio Alvarez, MD, a board-certified plastic surgeon based in Miami, Fla., and the CEO and medical director of Mia Aesthetics, which has several national locations.
“Getting into medicine is a long game, and there are many hurdles along the way that only the dedicated overcome,” says Dr. Alvarez.
Unfortunately, he says it may be late in that long game before some realize that the pay rate for certain specialties isn’t commensurate with the immense workload and responsibility they require.
“The short of it is that to become a happy doctor, medicine really needs to be a calling: a passion! There are far easier things to do to make money.”
Here is what physicians said about choosing between love or money.
The lowest-paying subspecialty in a low-paying specialty
Sophia Yen, MD, MPH, cofounder and CEO of Pandia Health, a women-founded, doctor-led birth control delivery service in Sunnyvale, Calif., and clinical associate professor at Stanford (Calif.) University, says you should pursue a specialty because you love the work.
“I chose the lowest-paying subspecialty (adolescent medicine) of a low-paying specialty (pediatrics), but I’d do it all again because I love the patient population – I love what I do.”
Dr. Yen says she chose adolescent medicine because she loves doing “outpatient gynecology” without going through the surgical training of a full ob.gyn. “I love the target population of young adults because you can talk to the patient versus in pediatrics, where you often talk to the parent. With young adults you can catch things – for example, teach a young person about consent, alcohol, marijuana’s effects on the growing brain, prevent unplanned pregnancies and sexually transmitted infections, instill healthy eating, and more.
“Do I wish that I got paid as much as a surgeon?” Dr. Yen says yes. “I hope that someday society will realize the time spent preventing future disease is worth it and pay us accordingly.”
Unfortunately, she says, since the health care system makes more money if you get pregnant, need a cardiac bypass, or need gastric surgery, those who deliver babies or do surgery get paid more than someone who prevents the need for those services.
Money doesn’t buy happiness
Stella Bard, MD, a rheumatologist in McKinney, Tex., says she eats, lives, and breathes rheumatology. “I never regret the decision of choosing this specialty for a single second,” says Dr. Bard. “I feel like it’s a rewarding experience with every single patient encounter.” Dr. Bard notes that money is no guarantee of happiness and that she feels blessed to wake up every morning doing what she loves.
Career or calling?
For Dr. Alvarez, inspiration came when watching his father help change people’s lives. “I saw how impactful a doctor is during a person’s most desperate moments, and that was enough to make medicine my life’s passion at the age of 10.”
He says once you’re in medical school, choosing a specialty is far easier than you think. “Each specialty requires a certain personality or specific characteristics, and some will call to you while others simply won’t.”
“For me, plastics was about finesse, art, and life-changing surgeries that affected people from kids to adults and involved every aspect of the human body. Changing someone’s outward appearance has a profoundly positive impact on their confidence and self-esteem, making plastic surgery a genuinely transformative experience.”
Patricia Celan, MD, a postgraduate psychiatry resident in Canada, also chose psychiatry for the love of the field. “I enjoy helping vulnerable people and exploring what makes a person tick, the source of their difficulties, and how to help people counteract and overcome the difficult cards they’ve been dealt in life.”
She says it’s incredibly rewarding to watch someone turn their life around from severe mental illness, especially those who have been victimized and traumatized, and learn to trust people again.
“I could have made more money in a higher-paying specialty, yes, but I’m not sure I would have felt as fulfilled as psychiatry can make me feel.”
Dr. Celan says everyone has their calling, and some lucky people find their deepest passion in higher-paying specialties. “My calling is psychiatry, and I am at peace with this no matter the money.”
For the love of surgery
“In my experience, most people don’t choose their specialty based on money,” says Nicole Aaronson, MD, MBA, an otolaryngologist and board-certified in the subspecialty of pediatric otolaryngology, an attending surgeon at Nemours Children’s Health of Delaware and clinical associate professor of otolaryngology and pediatrics at Sidney Kimmel Medical College, Philadelphia.
“The first decision point in medical school is usually figuring out if you are a surgery person or a medicine person. I knew very early that I wanted to be a surgeon and wanted to spend time in the OR fixing problems with my hands.”
Part of what attracted Dr. Aaronson to otolaryngology was the variety of conditions managed within the specialty, from head and neck cancer to voice problems to sleep disorders to sinus disease. “I chose my subspecialty because I enjoy working with children and making an impact that will help them live their best possible lives.”
She says a relatively simple surgery like placing ear tubes may help a child’s hearing and allow them to be more successful in school, opening up a new world of opportunities for the child’s future.
“While I don’t think most people choose their specialty based on prospective compensation, I do think all physicians want to be compensated fairly for their time, effort, and level of training,” says Dr. Aaronson.
Choosing a specialty for the money can lead to burnout and dissatisfaction
“For me, the decision to pursue gastroenterology went beyond financial considerations,” says Saurabh Sethi, MD, MPH, a gastroenterologist specializing in hepatology and interventional endoscopy. “While financial stability is undoubtedly important, no doctor enters this field solely for the love of money. The primary driving force for most medical professionals, myself included, is the passion to help people and make a positive difference in their lives.”
Dr. Sethi says the gratification that comes from providing quality care and witnessing patients’ improved well-being is priceless. Moreover, he believes that selecting a specialty based solely on financial gain is likely to lead to burnout and greater dissatisfaction over time.
“By following my love for gut health and prioritizing patient care, I have found a sense of fulfillment and purpose in my career. It has been a rewarding journey, and I’m grateful for the opportunity to contribute to the well-being of my patients through my expertise in gastroenterology.”
Key takeaways: Love or money?
Multiple factors influence doctors’ specialty choices, including genuine love for the work and the future of the specialty. Others include job prospects, hands-on experience they receive, mentors, childhood dreams, parental expectations, complexity of cases, the lifestyle of each specialty, including office hours worked, on-call requirements, and autonomy.
Physicians also mentioned other factors they considered when choosing their specialty:
- Personal interest.
- Intellectual stimulation.
- Work-life balance.
- Patient populations.
- Future opportunities.
- Desire to make a difference.
- Passion.
- Financial stability.
- Being personally fulfilled.
Overwhelmingly, doctors say to pick a specialty you can envision yourself loving 40 years from now and you won’t go wrong.
A version of this article first appeared on Medscape.com.
Medical student loans top hundreds of thousands of dollars, so it’s understandable that physicians may want to select a specialty that pays well.
Moreover, most advised young doctors to follow their hearts rather than their wallets.
“There is no question that many young kids immediately think about money when deciding to pursue medicine, but the thought of a big paycheck will never sustain someone long enough to get them here,” says Sergio Alvarez, MD, a board-certified plastic surgeon based in Miami, Fla., and the CEO and medical director of Mia Aesthetics, which has several national locations.
“Getting into medicine is a long game, and there are many hurdles along the way that only the dedicated overcome,” says Dr. Alvarez.
Unfortunately, he says it may be late in that long game before some realize that the pay rate for certain specialties isn’t commensurate with the immense workload and responsibility they require.
“The short of it is that to become a happy doctor, medicine really needs to be a calling: a passion! There are far easier things to do to make money.”
Here is what physicians said about choosing between love or money.
The lowest-paying subspecialty in a low-paying specialty
Sophia Yen, MD, MPH, cofounder and CEO of Pandia Health, a women-founded, doctor-led birth control delivery service in Sunnyvale, Calif., and clinical associate professor at Stanford (Calif.) University, says you should pursue a specialty because you love the work.
“I chose the lowest-paying subspecialty (adolescent medicine) of a low-paying specialty (pediatrics), but I’d do it all again because I love the patient population – I love what I do.”
Dr. Yen says she chose adolescent medicine because she loves doing “outpatient gynecology” without going through the surgical training of a full ob.gyn. “I love the target population of young adults because you can talk to the patient versus in pediatrics, where you often talk to the parent. With young adults you can catch things – for example, teach a young person about consent, alcohol, marijuana’s effects on the growing brain, prevent unplanned pregnancies and sexually transmitted infections, instill healthy eating, and more.
“Do I wish that I got paid as much as a surgeon?” Dr. Yen says yes. “I hope that someday society will realize the time spent preventing future disease is worth it and pay us accordingly.”
Unfortunately, she says, since the health care system makes more money if you get pregnant, need a cardiac bypass, or need gastric surgery, those who deliver babies or do surgery get paid more than someone who prevents the need for those services.
Money doesn’t buy happiness
Stella Bard, MD, a rheumatologist in McKinney, Tex., says she eats, lives, and breathes rheumatology. “I never regret the decision of choosing this specialty for a single second,” says Dr. Bard. “I feel like it’s a rewarding experience with every single patient encounter.” Dr. Bard notes that money is no guarantee of happiness and that she feels blessed to wake up every morning doing what she loves.
Career or calling?
For Dr. Alvarez, inspiration came when watching his father help change people’s lives. “I saw how impactful a doctor is during a person’s most desperate moments, and that was enough to make medicine my life’s passion at the age of 10.”
He says once you’re in medical school, choosing a specialty is far easier than you think. “Each specialty requires a certain personality or specific characteristics, and some will call to you while others simply won’t.”
“For me, plastics was about finesse, art, and life-changing surgeries that affected people from kids to adults and involved every aspect of the human body. Changing someone’s outward appearance has a profoundly positive impact on their confidence and self-esteem, making plastic surgery a genuinely transformative experience.”
Patricia Celan, MD, a postgraduate psychiatry resident in Canada, also chose psychiatry for the love of the field. “I enjoy helping vulnerable people and exploring what makes a person tick, the source of their difficulties, and how to help people counteract and overcome the difficult cards they’ve been dealt in life.”
She says it’s incredibly rewarding to watch someone turn their life around from severe mental illness, especially those who have been victimized and traumatized, and learn to trust people again.
“I could have made more money in a higher-paying specialty, yes, but I’m not sure I would have felt as fulfilled as psychiatry can make me feel.”
Dr. Celan says everyone has their calling, and some lucky people find their deepest passion in higher-paying specialties. “My calling is psychiatry, and I am at peace with this no matter the money.”
For the love of surgery
“In my experience, most people don’t choose their specialty based on money,” says Nicole Aaronson, MD, MBA, an otolaryngologist and board-certified in the subspecialty of pediatric otolaryngology, an attending surgeon at Nemours Children’s Health of Delaware and clinical associate professor of otolaryngology and pediatrics at Sidney Kimmel Medical College, Philadelphia.
“The first decision point in medical school is usually figuring out if you are a surgery person or a medicine person. I knew very early that I wanted to be a surgeon and wanted to spend time in the OR fixing problems with my hands.”
Part of what attracted Dr. Aaronson to otolaryngology was the variety of conditions managed within the specialty, from head and neck cancer to voice problems to sleep disorders to sinus disease. “I chose my subspecialty because I enjoy working with children and making an impact that will help them live their best possible lives.”
She says a relatively simple surgery like placing ear tubes may help a child’s hearing and allow them to be more successful in school, opening up a new world of opportunities for the child’s future.
“While I don’t think most people choose their specialty based on prospective compensation, I do think all physicians want to be compensated fairly for their time, effort, and level of training,” says Dr. Aaronson.
Choosing a specialty for the money can lead to burnout and dissatisfaction
“For me, the decision to pursue gastroenterology went beyond financial considerations,” says Saurabh Sethi, MD, MPH, a gastroenterologist specializing in hepatology and interventional endoscopy. “While financial stability is undoubtedly important, no doctor enters this field solely for the love of money. The primary driving force for most medical professionals, myself included, is the passion to help people and make a positive difference in their lives.”
Dr. Sethi says the gratification that comes from providing quality care and witnessing patients’ improved well-being is priceless. Moreover, he believes that selecting a specialty based solely on financial gain is likely to lead to burnout and greater dissatisfaction over time.
“By following my love for gut health and prioritizing patient care, I have found a sense of fulfillment and purpose in my career. It has been a rewarding journey, and I’m grateful for the opportunity to contribute to the well-being of my patients through my expertise in gastroenterology.”
Key takeaways: Love or money?
Multiple factors influence doctors’ specialty choices, including genuine love for the work and the future of the specialty. Others include job prospects, hands-on experience they receive, mentors, childhood dreams, parental expectations, complexity of cases, the lifestyle of each specialty, including office hours worked, on-call requirements, and autonomy.
Physicians also mentioned other factors they considered when choosing their specialty:
- Personal interest.
- Intellectual stimulation.
- Work-life balance.
- Patient populations.
- Future opportunities.
- Desire to make a difference.
- Passion.
- Financial stability.
- Being personally fulfilled.
Overwhelmingly, doctors say to pick a specialty you can envision yourself loving 40 years from now and you won’t go wrong.
A version of this article first appeared on Medscape.com.
Camp Discovery: A place for children to be comfortable in their own skin
The talent show, the grand finale of the 1-week camp, was nearly 7 years ago, but Emily Haygood of Houston, now 17 and about to start her senior year, remembers it in detail. She sang “Death of a Bachelor,” an R&B pop song and Billboard No. 1 hit at the time about a former bachelor who had happily married. These days, she said, if she watched the video of her 10-year-old singing self, “I would probably throw up.” But she still treasures the audience response, “having all those people I’d gotten close to cheer for me.”
Emily was at , but share one feature: they are the kind of dermatologic issues that can make doing everyday kid or teen activities like swimming difficult and can elicit mean comments from classmates and other would-be friends.

Emily was first diagnosed with atopic dermatitis at age 4, her mother, Amber Haygood, says. By age 9, it had become severe. Emily remembers being teased some in elementary school. “I did feel bad a lot of the time, when asked insensitive questions.” Her mother still bristles that adults often could be cruel, too.
But at Camp Discovery, those issues were nonexistent. “Camp was so cool,” Emily said. Besides the usual camp activities, it had things that “normal” camp didn’t, like other kids who didn’t stare at your skin condition or make fun of it.
30th anniversary season begins
This year is the 30th anniversary of Camp Discovery. Sessions began July 23 and continue through Aug. 18, with locations in Crosslake, Minn.; Hebron, Conn.; and Millville, Pa., in addition to Burton, Tex. About 300 campers will attend this year, according to the AAD, and 6,151 campers have attended from 1993 to 2022.
The 1-week camp accepts youth with conditions ranging from eczema and psoriasis to vitiligo, alopecia, epidermolysis bullosa, and ichthyosis, according to the academy. A dermatologist first refers a child, downloading and completing the referral form and sending it to the academy.
The 1-week session, including travel, is free for the campers, thanks to donors. As a nonprofit and membership-based organization, the AAD does not release the detailed financial information about the operating budget for the camp. Dermatologists, nurses, and counselors volunteer their time.
In his presidential address at the AAD’s annual meeting in March, outgoing president Mark D. Kaufmann, MD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, referred to camp volunteering as an antidote to professional burnout. Remembering why as a dermatologist one entered the profession can be one solution, he said, and described his own recent 3-day volunteer stint at the camp.
“Those 3 magical days, being with kids as they discovered they weren’t alone in the world, sharing their experiences and ideas, reminded me why I became a physician in the first place,” he told the audience of meeting attendees. He vowed to expand the program, with a goal of having every dermatology resident attend Camp Discovery.
Mental health effects of skin conditions
Much research has focused on the mental health fallout from living with chronic skin conditions, and even young children can be adversely affected. In one review of the literature, researchers concluded that pediatric skin disease, including acne, atopic dermatitis, and psoriasis, can affect quality of life, carry stigma, and lead to bullying and eventually even suicidal behavior. Another study, published earlier this year, found that atopic dermatitis affected children’s quality of life, impacting sleep and leading to feelings of being ashamed.
“It’s not necessarily about what their skin condition is and more about the psychosocial impact,’’ said Samantha Hill, MD, a pediatric and general dermatologist in Lynchburg, Va., who is the medical director of Camp Discovery in Minnesota this year.
Camp activities, reactions
The overriding theme of camp is allowing all the youth to be “just one of the kids at camp,” Dr. Hill said in an interview. “They come to do all kinds of things they don’t do in normal life because people don’t give them the credit to [be able to] do it.”
Every year, she said, “I tell my staff we are in the business of making things happen, so if there is a kid bandaged head to toe [because of a skin condition] and they want to go tubing and get in the lake, we figure out how to make it happen. We have done that multiple times.”
Newcomers are initially nervous, Dr. Hill acknowledged, but in time let their guard down. Returnees are a different story. “When kids who have been at camp before arrive, you can see them start breathing again, looking for their friends. You can see them relax right before your eyes.”
“The single most empowering thing is the realization you are not alone,” said Meena Julapalli, MD, a Houston dermatologist who is a medical team member and long-time volunteer at Camp Discovery. That, she said, and “You get to be a kid, and you don’t have to have people staring at you.”
Dr. Julapalli remembers one of her patients with keratitis-ichthyosis-deafness (KID) syndrome. “She needed more than what I could offer,” she said. “She needed camp.” At camp, the organizers found a counselor who knew sign language to accompany her. At first, she was quiet and didn’t smile much. By the end of the week, as she was about to observe her birthday, things changed. After breakfast, she was led to the stage, where fellow campers began singing – and signing the song they had just learned.
Camp staff gets it
Allyson Garin, who was diagnosed with vitiligo at age 6 months, is a camp program director at Camp Discovery in Crosslake, Minn. She first went to camp in 1990 at age 11, returning until she “aged out” at 16, then worked as a counselor. She gets it when campers tell her they hear rude comments about their skin conditions.
“I remember being in swimming pools, in lines at fairgrounds or amusement parks,” she said in an interview, “and hearing people say, ‘Don’t touch her,’ ’’ fearing contagion, perhaps. “People would make jokes about cows, since they are spotted,” she said, or people would simply step back.
All those years ago, her mother found out about the camp and decided to figure out how to get her there. She got there, and she met a fellow camper with vitiligo, and they became pen pals. “We still talk,” she said.
Meeting someone with the same skin condition, she said, isn’t just about commiserating. “There is a lot of information sharing,” on topics such as best treatments, strategies, and other conversations.
Other lessons
While campers can feel comfortable around others who also have skin conditions, and understand, the lesson extends beyond that, Ms. Garin said. “It gave me a perspective,” she said of her camp experience. “I always felt, ‘Woe is me.’ ” But when she met others with, as she said, conditions “way worse than vitiligo, it really grounds you.”
Dr. Hill agreed. Campers get the benefit of others accepting and including them, but also practicing that same attitude toward fellow campers, she said. “It insures that we are providing this environment of inclusion, but that they are practicing it as well. They need to practice it like everyone else.”
Getting parents on board
The idea of camp, especially for those at the younger end of the 8- to 16-years age range accepted for Camp Discovery, can take some getting used to for some parents. Ms. Haygood, Emily’s mother, relates to that. Her daughter’s dermatologist at the time, who is now retired, had first suggested the camp. Her first reaction? “I am not sending my chronically ill child to camp with strangers.” She also acknowledged that she, like other parents of children with a chronic illness, can be a helicopter parent.
Then, she noticed that Emily seemed interested, so she got more information, finding out that it was staffed by doctors. It all sounded good, she said, and the social interaction, she knew, would be beneficial. “Then my husband was a no,” she said, concerned about their daughter being with strangers. “Eventually he came around,” Ms. Haygood said. All along, Emily said, “it seemed fun. I was probably trying to talk them into it.” She admits she was very nervous at first, but calmed down when she realized her own dermatologist was going to be there.
Vanessa Hadley of Spring, Tex., was on board the moment she heard about Camp Discovery. “I just thought it was amazing,” she said. Her daughter Isabelle, 13, has been to the camp. “She has alopecia areata and severe eczema,” Ms. Hadley said. Now, Isabelle is returning to camp and coaching her sister Penelope, 8, who has eczema and mild alopecia and is a first-timer this summer.
One tip the 8-year-old has learned so far: Turn to your counselor for support if you’re nervous. That worked, Isabelle said, the first year when she was wary of the zipline – then surprised herself and conquered it.
Dr. Hill and Dr. Julapalli have no disclosures.
The talent show, the grand finale of the 1-week camp, was nearly 7 years ago, but Emily Haygood of Houston, now 17 and about to start her senior year, remembers it in detail. She sang “Death of a Bachelor,” an R&B pop song and Billboard No. 1 hit at the time about a former bachelor who had happily married. These days, she said, if she watched the video of her 10-year-old singing self, “I would probably throw up.” But she still treasures the audience response, “having all those people I’d gotten close to cheer for me.”
Emily was at , but share one feature: they are the kind of dermatologic issues that can make doing everyday kid or teen activities like swimming difficult and can elicit mean comments from classmates and other would-be friends.

Emily was first diagnosed with atopic dermatitis at age 4, her mother, Amber Haygood, says. By age 9, it had become severe. Emily remembers being teased some in elementary school. “I did feel bad a lot of the time, when asked insensitive questions.” Her mother still bristles that adults often could be cruel, too.
But at Camp Discovery, those issues were nonexistent. “Camp was so cool,” Emily said. Besides the usual camp activities, it had things that “normal” camp didn’t, like other kids who didn’t stare at your skin condition or make fun of it.
30th anniversary season begins
This year is the 30th anniversary of Camp Discovery. Sessions began July 23 and continue through Aug. 18, with locations in Crosslake, Minn.; Hebron, Conn.; and Millville, Pa., in addition to Burton, Tex. About 300 campers will attend this year, according to the AAD, and 6,151 campers have attended from 1993 to 2022.
The 1-week camp accepts youth with conditions ranging from eczema and psoriasis to vitiligo, alopecia, epidermolysis bullosa, and ichthyosis, according to the academy. A dermatologist first refers a child, downloading and completing the referral form and sending it to the academy.
The 1-week session, including travel, is free for the campers, thanks to donors. As a nonprofit and membership-based organization, the AAD does not release the detailed financial information about the operating budget for the camp. Dermatologists, nurses, and counselors volunteer their time.
In his presidential address at the AAD’s annual meeting in March, outgoing president Mark D. Kaufmann, MD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, referred to camp volunteering as an antidote to professional burnout. Remembering why as a dermatologist one entered the profession can be one solution, he said, and described his own recent 3-day volunteer stint at the camp.
“Those 3 magical days, being with kids as they discovered they weren’t alone in the world, sharing their experiences and ideas, reminded me why I became a physician in the first place,” he told the audience of meeting attendees. He vowed to expand the program, with a goal of having every dermatology resident attend Camp Discovery.
Mental health effects of skin conditions
Much research has focused on the mental health fallout from living with chronic skin conditions, and even young children can be adversely affected. In one review of the literature, researchers concluded that pediatric skin disease, including acne, atopic dermatitis, and psoriasis, can affect quality of life, carry stigma, and lead to bullying and eventually even suicidal behavior. Another study, published earlier this year, found that atopic dermatitis affected children’s quality of life, impacting sleep and leading to feelings of being ashamed.
“It’s not necessarily about what their skin condition is and more about the psychosocial impact,’’ said Samantha Hill, MD, a pediatric and general dermatologist in Lynchburg, Va., who is the medical director of Camp Discovery in Minnesota this year.
Camp activities, reactions
The overriding theme of camp is allowing all the youth to be “just one of the kids at camp,” Dr. Hill said in an interview. “They come to do all kinds of things they don’t do in normal life because people don’t give them the credit to [be able to] do it.”
Every year, she said, “I tell my staff we are in the business of making things happen, so if there is a kid bandaged head to toe [because of a skin condition] and they want to go tubing and get in the lake, we figure out how to make it happen. We have done that multiple times.”
Newcomers are initially nervous, Dr. Hill acknowledged, but in time let their guard down. Returnees are a different story. “When kids who have been at camp before arrive, you can see them start breathing again, looking for their friends. You can see them relax right before your eyes.”
“The single most empowering thing is the realization you are not alone,” said Meena Julapalli, MD, a Houston dermatologist who is a medical team member and long-time volunteer at Camp Discovery. That, she said, and “You get to be a kid, and you don’t have to have people staring at you.”
Dr. Julapalli remembers one of her patients with keratitis-ichthyosis-deafness (KID) syndrome. “She needed more than what I could offer,” she said. “She needed camp.” At camp, the organizers found a counselor who knew sign language to accompany her. At first, she was quiet and didn’t smile much. By the end of the week, as she was about to observe her birthday, things changed. After breakfast, she was led to the stage, where fellow campers began singing – and signing the song they had just learned.
Camp staff gets it
Allyson Garin, who was diagnosed with vitiligo at age 6 months, is a camp program director at Camp Discovery in Crosslake, Minn. She first went to camp in 1990 at age 11, returning until she “aged out” at 16, then worked as a counselor. She gets it when campers tell her they hear rude comments about their skin conditions.
“I remember being in swimming pools, in lines at fairgrounds or amusement parks,” she said in an interview, “and hearing people say, ‘Don’t touch her,’ ’’ fearing contagion, perhaps. “People would make jokes about cows, since they are spotted,” she said, or people would simply step back.
All those years ago, her mother found out about the camp and decided to figure out how to get her there. She got there, and she met a fellow camper with vitiligo, and they became pen pals. “We still talk,” she said.
Meeting someone with the same skin condition, she said, isn’t just about commiserating. “There is a lot of information sharing,” on topics such as best treatments, strategies, and other conversations.
Other lessons
While campers can feel comfortable around others who also have skin conditions, and understand, the lesson extends beyond that, Ms. Garin said. “It gave me a perspective,” she said of her camp experience. “I always felt, ‘Woe is me.’ ” But when she met others with, as she said, conditions “way worse than vitiligo, it really grounds you.”
Dr. Hill agreed. Campers get the benefit of others accepting and including them, but also practicing that same attitude toward fellow campers, she said. “It insures that we are providing this environment of inclusion, but that they are practicing it as well. They need to practice it like everyone else.”
Getting parents on board
The idea of camp, especially for those at the younger end of the 8- to 16-years age range accepted for Camp Discovery, can take some getting used to for some parents. Ms. Haygood, Emily’s mother, relates to that. Her daughter’s dermatologist at the time, who is now retired, had first suggested the camp. Her first reaction? “I am not sending my chronically ill child to camp with strangers.” She also acknowledged that she, like other parents of children with a chronic illness, can be a helicopter parent.
Then, she noticed that Emily seemed interested, so she got more information, finding out that it was staffed by doctors. It all sounded good, she said, and the social interaction, she knew, would be beneficial. “Then my husband was a no,” she said, concerned about their daughter being with strangers. “Eventually he came around,” Ms. Haygood said. All along, Emily said, “it seemed fun. I was probably trying to talk them into it.” She admits she was very nervous at first, but calmed down when she realized her own dermatologist was going to be there.
Vanessa Hadley of Spring, Tex., was on board the moment she heard about Camp Discovery. “I just thought it was amazing,” she said. Her daughter Isabelle, 13, has been to the camp. “She has alopecia areata and severe eczema,” Ms. Hadley said. Now, Isabelle is returning to camp and coaching her sister Penelope, 8, who has eczema and mild alopecia and is a first-timer this summer.
One tip the 8-year-old has learned so far: Turn to your counselor for support if you’re nervous. That worked, Isabelle said, the first year when she was wary of the zipline – then surprised herself and conquered it.
Dr. Hill and Dr. Julapalli have no disclosures.
The talent show, the grand finale of the 1-week camp, was nearly 7 years ago, but Emily Haygood of Houston, now 17 and about to start her senior year, remembers it in detail. She sang “Death of a Bachelor,” an R&B pop song and Billboard No. 1 hit at the time about a former bachelor who had happily married. These days, she said, if she watched the video of her 10-year-old singing self, “I would probably throw up.” But she still treasures the audience response, “having all those people I’d gotten close to cheer for me.”
Emily was at , but share one feature: they are the kind of dermatologic issues that can make doing everyday kid or teen activities like swimming difficult and can elicit mean comments from classmates and other would-be friends.

Emily was first diagnosed with atopic dermatitis at age 4, her mother, Amber Haygood, says. By age 9, it had become severe. Emily remembers being teased some in elementary school. “I did feel bad a lot of the time, when asked insensitive questions.” Her mother still bristles that adults often could be cruel, too.
But at Camp Discovery, those issues were nonexistent. “Camp was so cool,” Emily said. Besides the usual camp activities, it had things that “normal” camp didn’t, like other kids who didn’t stare at your skin condition or make fun of it.
30th anniversary season begins
This year is the 30th anniversary of Camp Discovery. Sessions began July 23 and continue through Aug. 18, with locations in Crosslake, Minn.; Hebron, Conn.; and Millville, Pa., in addition to Burton, Tex. About 300 campers will attend this year, according to the AAD, and 6,151 campers have attended from 1993 to 2022.
The 1-week camp accepts youth with conditions ranging from eczema and psoriasis to vitiligo, alopecia, epidermolysis bullosa, and ichthyosis, according to the academy. A dermatologist first refers a child, downloading and completing the referral form and sending it to the academy.
The 1-week session, including travel, is free for the campers, thanks to donors. As a nonprofit and membership-based organization, the AAD does not release the detailed financial information about the operating budget for the camp. Dermatologists, nurses, and counselors volunteer their time.
In his presidential address at the AAD’s annual meeting in March, outgoing president Mark D. Kaufmann, MD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, referred to camp volunteering as an antidote to professional burnout. Remembering why as a dermatologist one entered the profession can be one solution, he said, and described his own recent 3-day volunteer stint at the camp.
“Those 3 magical days, being with kids as they discovered they weren’t alone in the world, sharing their experiences and ideas, reminded me why I became a physician in the first place,” he told the audience of meeting attendees. He vowed to expand the program, with a goal of having every dermatology resident attend Camp Discovery.
Mental health effects of skin conditions
Much research has focused on the mental health fallout from living with chronic skin conditions, and even young children can be adversely affected. In one review of the literature, researchers concluded that pediatric skin disease, including acne, atopic dermatitis, and psoriasis, can affect quality of life, carry stigma, and lead to bullying and eventually even suicidal behavior. Another study, published earlier this year, found that atopic dermatitis affected children’s quality of life, impacting sleep and leading to feelings of being ashamed.
“It’s not necessarily about what their skin condition is and more about the psychosocial impact,’’ said Samantha Hill, MD, a pediatric and general dermatologist in Lynchburg, Va., who is the medical director of Camp Discovery in Minnesota this year.
Camp activities, reactions
The overriding theme of camp is allowing all the youth to be “just one of the kids at camp,” Dr. Hill said in an interview. “They come to do all kinds of things they don’t do in normal life because people don’t give them the credit to [be able to] do it.”
Every year, she said, “I tell my staff we are in the business of making things happen, so if there is a kid bandaged head to toe [because of a skin condition] and they want to go tubing and get in the lake, we figure out how to make it happen. We have done that multiple times.”
Newcomers are initially nervous, Dr. Hill acknowledged, but in time let their guard down. Returnees are a different story. “When kids who have been at camp before arrive, you can see them start breathing again, looking for their friends. You can see them relax right before your eyes.”
“The single most empowering thing is the realization you are not alone,” said Meena Julapalli, MD, a Houston dermatologist who is a medical team member and long-time volunteer at Camp Discovery. That, she said, and “You get to be a kid, and you don’t have to have people staring at you.”
Dr. Julapalli remembers one of her patients with keratitis-ichthyosis-deafness (KID) syndrome. “She needed more than what I could offer,” she said. “She needed camp.” At camp, the organizers found a counselor who knew sign language to accompany her. At first, she was quiet and didn’t smile much. By the end of the week, as she was about to observe her birthday, things changed. After breakfast, she was led to the stage, where fellow campers began singing – and signing the song they had just learned.
Camp staff gets it
Allyson Garin, who was diagnosed with vitiligo at age 6 months, is a camp program director at Camp Discovery in Crosslake, Minn. She first went to camp in 1990 at age 11, returning until she “aged out” at 16, then worked as a counselor. She gets it when campers tell her they hear rude comments about their skin conditions.
“I remember being in swimming pools, in lines at fairgrounds or amusement parks,” she said in an interview, “and hearing people say, ‘Don’t touch her,’ ’’ fearing contagion, perhaps. “People would make jokes about cows, since they are spotted,” she said, or people would simply step back.
All those years ago, her mother found out about the camp and decided to figure out how to get her there. She got there, and she met a fellow camper with vitiligo, and they became pen pals. “We still talk,” she said.
Meeting someone with the same skin condition, she said, isn’t just about commiserating. “There is a lot of information sharing,” on topics such as best treatments, strategies, and other conversations.
Other lessons
While campers can feel comfortable around others who also have skin conditions, and understand, the lesson extends beyond that, Ms. Garin said. “It gave me a perspective,” she said of her camp experience. “I always felt, ‘Woe is me.’ ” But when she met others with, as she said, conditions “way worse than vitiligo, it really grounds you.”
Dr. Hill agreed. Campers get the benefit of others accepting and including them, but also practicing that same attitude toward fellow campers, she said. “It insures that we are providing this environment of inclusion, but that they are practicing it as well. They need to practice it like everyone else.”
Getting parents on board
The idea of camp, especially for those at the younger end of the 8- to 16-years age range accepted for Camp Discovery, can take some getting used to for some parents. Ms. Haygood, Emily’s mother, relates to that. Her daughter’s dermatologist at the time, who is now retired, had first suggested the camp. Her first reaction? “I am not sending my chronically ill child to camp with strangers.” She also acknowledged that she, like other parents of children with a chronic illness, can be a helicopter parent.
Then, she noticed that Emily seemed interested, so she got more information, finding out that it was staffed by doctors. It all sounded good, she said, and the social interaction, she knew, would be beneficial. “Then my husband was a no,” she said, concerned about their daughter being with strangers. “Eventually he came around,” Ms. Haygood said. All along, Emily said, “it seemed fun. I was probably trying to talk them into it.” She admits she was very nervous at first, but calmed down when she realized her own dermatologist was going to be there.
Vanessa Hadley of Spring, Tex., was on board the moment she heard about Camp Discovery. “I just thought it was amazing,” she said. Her daughter Isabelle, 13, has been to the camp. “She has alopecia areata and severe eczema,” Ms. Hadley said. Now, Isabelle is returning to camp and coaching her sister Penelope, 8, who has eczema and mild alopecia and is a first-timer this summer.
One tip the 8-year-old has learned so far: Turn to your counselor for support if you’re nervous. That worked, Isabelle said, the first year when she was wary of the zipline – then surprised herself and conquered it.
Dr. Hill and Dr. Julapalli have no disclosures.
Impostor syndrome is a risk for doctors of all ages
COVID-19 imposed challenges on health care professionals and systems by forcing changes in how doctors organize themselves professionally as well as in their relationships with patients and in their expectations (realistic or not) of their roles. The situation was bound to generate high rates of frustration and discomfort among younger and older physicians. It was compounded by a generational transition of the profession, which was accelerated by the virus. It was not managed by the decision-makers and was painful for doctors and patients.
Impostor syndrome (IS) is a psychological construct characterized by the persistent belief that one’s success is undeserved, rather than stemming from personal effort, skill, and ability. The phenomenon is common among medics for various reasons, including professional burnout. Recent studies have helped to better define the extent and characteristic features of the syndrome, as well as efforts to combat it.
Doctors and burnout
Although occupational burnout among physicians is a systemic issue primarily attributable to problems in the practice environment, professional norms and aspects of medical culture often contribute to the distress that individual physicians experience.
These dimensions have been well characterized and include suggestions that physicians should be impervious to normal human limitations (that is, superhuman), that work should always come first, and that seeking help is a sign of weakness. In aggregate, these attitudes lead many physicians to engage in unhealthy levels of self-sacrifice, manifested by excessive work hours, anxiety about missing something that would benefit their patients, and prioritizing work over personal health. These factors are familiar to many hospital-based and family physicians.
The impostor phenomenon
The impostor phenomenon (IP) is a psychological experience of intellectual and professional fraud. Individuals who suffer from it believe that others have inflated perceptions of the individual’s abilities and fear being judged. This fear persists despite continual proof of the individual’s successes. These people ignore praise, are highly self-critical, and attribute their successes to external factors, such as luck, hard work, or receiving help from others, rather than to qualities such as skill, intelligence, or ability.
IP is common among men and women. Some studies suggest it may be more prevalent among women. Studies across industries suggest that the phenomenon is associated with personal consequences (for example, low emotional well-being, problems with work-life integration, anxiety, depression, suicide) and professional consequences (for example, impaired job performance, occupational burnout). Studies involving U.S. medical students have revealed that more than one in four medical students experience IP and that those who experience it are at higher risk for burnout.
Surveying IS
IS, which is not a formal psychiatric diagnosis, is defined as having feelings of uncertainty, inadequacy, and being undeserving of one’s achievements despite evidence to the contrary. There are five subtypes of IS:
- Perfectionist: insecurity related to self-imposed, unachievable goals
- Expert: feeling inadequate from lacking sufficient knowledge
- Superperson: assuming excessive workloads just to feel okay among peers
- Natural genius: experiencing shame when it takes effort to develop a skill
- Soloist: believing that requesting help is a sign of weakness
Risk factors
Studies suggest that IS is a problem early in the physician training process. There is limited information on IS among physicians in practice.
Because transitions represent a risk factor for IP, the frequent rotation between clerkships and being a “perpetual novice” during medical school training may contribute to the high prevalence. Qualitative studies suggest that, once in practice, other professional experiences (for example, unfavorable patient outcomes, patient complaints, rejection of grants or manuscripts, and poor teaching evaluations or patient satisfaction scores) may contribute to IP.
Impact on doctors
Several methods have been used to classify how much the phenomenon interferes with a person’s life. The Clance Impostor Phenomenon Scale is a 20-item scale that asks respondents to indicate how well each item characterizes their experience on a 5-point scale. Options range from “not at all” to “very true.” The sum of responses to the individual items is used to create an aggregate score (IP score). The higher the score, the more frequently and seriously IP interferes with a person’s life.
A simplified version of the IP score was used in a study of 3,237 U.S. doctors that investigated the association between IS and burnout among doctors and to compare their rates of IS with those of other professionals.
Mean IP scores were higher for female physicians than for male physicians (mean, 10.91 vs. 9.12; P < .001). Scores decreased with age and were lower among those who were married or widowed.
With respect to professional characteristics, IP scores were greater among those in academic practice or who worked in the Veterans Affairs medical system and decreased with years in practice.
The highest IP scores were among pediatric subspecialists, general pediatricians, and emergency medicine physicians. Scores were lowest among ophthalmologists, radiologists, and orthopedic surgeons. IP has been independently associated with the risk of burnout and low professional fulfillment.
Lessening the impact
An article commenting on the study highlighted the following expert practice strategies that doctors can use to reduce the impact of IS in their professional life.
- Review and celebrate feats that have led to your professional role.
- Share concerns with trusted colleagues who can validate your accomplishments and normalize your feelings by reporting their own struggles with IS.
- Combat perfectionism by accepting that it is okay to be good enough when meeting the challenges of a demanding profession.
- Exercise self-compassion as an alternative to relying on an external locus of self-worth.
- Understand that IS may be common, especially during transitions, such as when entering medical school, graduate medical training, or starting a new career.
This article was translated from Univadis Italy. A version appeared on Medscape.com.
COVID-19 imposed challenges on health care professionals and systems by forcing changes in how doctors organize themselves professionally as well as in their relationships with patients and in their expectations (realistic or not) of their roles. The situation was bound to generate high rates of frustration and discomfort among younger and older physicians. It was compounded by a generational transition of the profession, which was accelerated by the virus. It was not managed by the decision-makers and was painful for doctors and patients.
Impostor syndrome (IS) is a psychological construct characterized by the persistent belief that one’s success is undeserved, rather than stemming from personal effort, skill, and ability. The phenomenon is common among medics for various reasons, including professional burnout. Recent studies have helped to better define the extent and characteristic features of the syndrome, as well as efforts to combat it.
Doctors and burnout
Although occupational burnout among physicians is a systemic issue primarily attributable to problems in the practice environment, professional norms and aspects of medical culture often contribute to the distress that individual physicians experience.
These dimensions have been well characterized and include suggestions that physicians should be impervious to normal human limitations (that is, superhuman), that work should always come first, and that seeking help is a sign of weakness. In aggregate, these attitudes lead many physicians to engage in unhealthy levels of self-sacrifice, manifested by excessive work hours, anxiety about missing something that would benefit their patients, and prioritizing work over personal health. These factors are familiar to many hospital-based and family physicians.
The impostor phenomenon
The impostor phenomenon (IP) is a psychological experience of intellectual and professional fraud. Individuals who suffer from it believe that others have inflated perceptions of the individual’s abilities and fear being judged. This fear persists despite continual proof of the individual’s successes. These people ignore praise, are highly self-critical, and attribute their successes to external factors, such as luck, hard work, or receiving help from others, rather than to qualities such as skill, intelligence, or ability.
IP is common among men and women. Some studies suggest it may be more prevalent among women. Studies across industries suggest that the phenomenon is associated with personal consequences (for example, low emotional well-being, problems with work-life integration, anxiety, depression, suicide) and professional consequences (for example, impaired job performance, occupational burnout). Studies involving U.S. medical students have revealed that more than one in four medical students experience IP and that those who experience it are at higher risk for burnout.
Surveying IS
IS, which is not a formal psychiatric diagnosis, is defined as having feelings of uncertainty, inadequacy, and being undeserving of one’s achievements despite evidence to the contrary. There are five subtypes of IS:
- Perfectionist: insecurity related to self-imposed, unachievable goals
- Expert: feeling inadequate from lacking sufficient knowledge
- Superperson: assuming excessive workloads just to feel okay among peers
- Natural genius: experiencing shame when it takes effort to develop a skill
- Soloist: believing that requesting help is a sign of weakness
Risk factors
Studies suggest that IS is a problem early in the physician training process. There is limited information on IS among physicians in practice.
Because transitions represent a risk factor for IP, the frequent rotation between clerkships and being a “perpetual novice” during medical school training may contribute to the high prevalence. Qualitative studies suggest that, once in practice, other professional experiences (for example, unfavorable patient outcomes, patient complaints, rejection of grants or manuscripts, and poor teaching evaluations or patient satisfaction scores) may contribute to IP.
Impact on doctors
Several methods have been used to classify how much the phenomenon interferes with a person’s life. The Clance Impostor Phenomenon Scale is a 20-item scale that asks respondents to indicate how well each item characterizes their experience on a 5-point scale. Options range from “not at all” to “very true.” The sum of responses to the individual items is used to create an aggregate score (IP score). The higher the score, the more frequently and seriously IP interferes with a person’s life.
A simplified version of the IP score was used in a study of 3,237 U.S. doctors that investigated the association between IS and burnout among doctors and to compare their rates of IS with those of other professionals.
Mean IP scores were higher for female physicians than for male physicians (mean, 10.91 vs. 9.12; P < .001). Scores decreased with age and were lower among those who were married or widowed.
With respect to professional characteristics, IP scores were greater among those in academic practice or who worked in the Veterans Affairs medical system and decreased with years in practice.
The highest IP scores were among pediatric subspecialists, general pediatricians, and emergency medicine physicians. Scores were lowest among ophthalmologists, radiologists, and orthopedic surgeons. IP has been independently associated with the risk of burnout and low professional fulfillment.
Lessening the impact
An article commenting on the study highlighted the following expert practice strategies that doctors can use to reduce the impact of IS in their professional life.
- Review and celebrate feats that have led to your professional role.
- Share concerns with trusted colleagues who can validate your accomplishments and normalize your feelings by reporting their own struggles with IS.
- Combat perfectionism by accepting that it is okay to be good enough when meeting the challenges of a demanding profession.
- Exercise self-compassion as an alternative to relying on an external locus of self-worth.
- Understand that IS may be common, especially during transitions, such as when entering medical school, graduate medical training, or starting a new career.
This article was translated from Univadis Italy. A version appeared on Medscape.com.
COVID-19 imposed challenges on health care professionals and systems by forcing changes in how doctors organize themselves professionally as well as in their relationships with patients and in their expectations (realistic or not) of their roles. The situation was bound to generate high rates of frustration and discomfort among younger and older physicians. It was compounded by a generational transition of the profession, which was accelerated by the virus. It was not managed by the decision-makers and was painful for doctors and patients.
Impostor syndrome (IS) is a psychological construct characterized by the persistent belief that one’s success is undeserved, rather than stemming from personal effort, skill, and ability. The phenomenon is common among medics for various reasons, including professional burnout. Recent studies have helped to better define the extent and characteristic features of the syndrome, as well as efforts to combat it.
Doctors and burnout
Although occupational burnout among physicians is a systemic issue primarily attributable to problems in the practice environment, professional norms and aspects of medical culture often contribute to the distress that individual physicians experience.
These dimensions have been well characterized and include suggestions that physicians should be impervious to normal human limitations (that is, superhuman), that work should always come first, and that seeking help is a sign of weakness. In aggregate, these attitudes lead many physicians to engage in unhealthy levels of self-sacrifice, manifested by excessive work hours, anxiety about missing something that would benefit their patients, and prioritizing work over personal health. These factors are familiar to many hospital-based and family physicians.
The impostor phenomenon
The impostor phenomenon (IP) is a psychological experience of intellectual and professional fraud. Individuals who suffer from it believe that others have inflated perceptions of the individual’s abilities and fear being judged. This fear persists despite continual proof of the individual’s successes. These people ignore praise, are highly self-critical, and attribute their successes to external factors, such as luck, hard work, or receiving help from others, rather than to qualities such as skill, intelligence, or ability.
IP is common among men and women. Some studies suggest it may be more prevalent among women. Studies across industries suggest that the phenomenon is associated with personal consequences (for example, low emotional well-being, problems with work-life integration, anxiety, depression, suicide) and professional consequences (for example, impaired job performance, occupational burnout). Studies involving U.S. medical students have revealed that more than one in four medical students experience IP and that those who experience it are at higher risk for burnout.
Surveying IS
IS, which is not a formal psychiatric diagnosis, is defined as having feelings of uncertainty, inadequacy, and being undeserving of one’s achievements despite evidence to the contrary. There are five subtypes of IS:
- Perfectionist: insecurity related to self-imposed, unachievable goals
- Expert: feeling inadequate from lacking sufficient knowledge
- Superperson: assuming excessive workloads just to feel okay among peers
- Natural genius: experiencing shame when it takes effort to develop a skill
- Soloist: believing that requesting help is a sign of weakness
Risk factors
Studies suggest that IS is a problem early in the physician training process. There is limited information on IS among physicians in practice.
Because transitions represent a risk factor for IP, the frequent rotation between clerkships and being a “perpetual novice” during medical school training may contribute to the high prevalence. Qualitative studies suggest that, once in practice, other professional experiences (for example, unfavorable patient outcomes, patient complaints, rejection of grants or manuscripts, and poor teaching evaluations or patient satisfaction scores) may contribute to IP.
Impact on doctors
Several methods have been used to classify how much the phenomenon interferes with a person’s life. The Clance Impostor Phenomenon Scale is a 20-item scale that asks respondents to indicate how well each item characterizes their experience on a 5-point scale. Options range from “not at all” to “very true.” The sum of responses to the individual items is used to create an aggregate score (IP score). The higher the score, the more frequently and seriously IP interferes with a person’s life.
A simplified version of the IP score was used in a study of 3,237 U.S. doctors that investigated the association between IS and burnout among doctors and to compare their rates of IS with those of other professionals.
Mean IP scores were higher for female physicians than for male physicians (mean, 10.91 vs. 9.12; P < .001). Scores decreased with age and were lower among those who were married or widowed.
With respect to professional characteristics, IP scores were greater among those in academic practice or who worked in the Veterans Affairs medical system and decreased with years in practice.
The highest IP scores were among pediatric subspecialists, general pediatricians, and emergency medicine physicians. Scores were lowest among ophthalmologists, radiologists, and orthopedic surgeons. IP has been independently associated with the risk of burnout and low professional fulfillment.
Lessening the impact
An article commenting on the study highlighted the following expert practice strategies that doctors can use to reduce the impact of IS in their professional life.
- Review and celebrate feats that have led to your professional role.
- Share concerns with trusted colleagues who can validate your accomplishments and normalize your feelings by reporting their own struggles with IS.
- Combat perfectionism by accepting that it is okay to be good enough when meeting the challenges of a demanding profession.
- Exercise self-compassion as an alternative to relying on an external locus of self-worth.
- Understand that IS may be common, especially during transitions, such as when entering medical school, graduate medical training, or starting a new career.
This article was translated from Univadis Italy. A version appeared on Medscape.com.
Study finds big growth in advanced-practice clinicians in Medicare dermatology
A .
Researchers from the University of Texas MD Anderson Cancer Center, Houston, and the University of Pennsylvania, Philadelphia, reported in JAMA Dermatology that in 2013 APCs made up 28% of the dermatology clinician workforce. By 2020, they made up 37% of the dermatology clinicians giving care to Medicare beneficiaries.
Retrospective cohort study
APCs provided care in 15.5% of dermatology office visits in 2013 and 27.4% in 2020 (P =.02), the authors reported. “By 2020, more than one in four dermatology visits for patients with Medicare were delivered by APCs,” wrote the authors, led by Mackenzie R. Wehner, MD, MPhil, assistant professor of dermatology and health services research at MD Anderson.
“Everyone in dermatology is aware of the increasing adoption of advanced practice clinicians in the field,” Justin D. Arnold, MD, MMSc, a 3rd-year dermatology resident at the University of California, Irvine, said in an interview. “However, seeing how quickly this happening and the absolute number of these clinicians is still startling,” said Dr. Arnold, who in 2022 published a research letter in JAMA Dermatology on the impact of physician assistants in dermatology.
In that study, he and his coauthors reported that the PA workforce in dermatology was growing faster than in other specialties.
In the current study, Dr. Wehner and her colleagues identified 8,444 dermatology APCs and 14,402 physician dermatologists who provided 109.3 million Medicare office visits from 2013 to 2020. More than 80% of the procedures were performed by physicians, but APCs appeared to increasingly be taking on more of the procedural load.
Over the study period, APCs had an average annual increase of 12.6% in the number of premalignant lesion destructions performed; physicians saw an average 1.4% decline. For skin biopsies, APCs performed 11.7% more per year on average, compared with a 1.4% drop for physicians.
“This data is not surprising given most agree that skin biopsies and destruction of premalignant lesions are well within the scope of practice of APCs,” Dr. Arnold told this news organization.
The authors also reported that, while most APCs – similar to physician dermatologists – practice in metropolitan areas, they are working in other locations also. Slightly more than half of dermatology clinicians in micropolitan areas are APCs, and in rural areas, 88% of clinicians are APCs, Dr. Wehner and colleagues found.
APCs may be filling a gap in rural areas for Medicare patients, said Dr. Arnold, but, he added, “it is unclear if dermatology APCs are growing as quickly in practices that predominantly accept Medicaid and if dermatology APCs are expanding access to these populations.”
Dr. Arnold said he expected the number of APCs in dermatology to continue growing, serving commercially insured patients, as well. “There are a multitude of potential reasons for more APCs in dermatology, including difficulty recruiting dermatologists in rural communities, financial motivators, and the expansion of private equity, and the increasing acceptance of these clinicians within medicine and by patients.”
APCs can provide good-quality care if they are properly trained and supervised, said Dr. Arnold, adding that he is concerned, however, that the training and supervision is not being provided. “This study provides further evidence that dermatologists, and national dermatology organizations such as the AAD [American Academy of Dermatology], need to take a more active role in the leadership of APC training,” he said.
Dermatology, he noted, “would benefit from consensus guidelines on clinical competencies for dermatology APCs,” similar to an effort by the American College of Cardiology.
A review* published online in July noted that, compared with dermatologists, some data suggest that non-physician operators (NPOs) may have a higher rate of adverse events when performing aesthetic procedures, according to the authors of the review, led by Shelby L. Kubicki, MD, of the department of dermatology at the UTHealth Science Center in Houston. There is no mandatory reporting of complications for nonphysician providers, so the authors relied on data from cosmetic-focused practices, medical spas, and a survey by the American Society of Dermatologic Surgery of consumers and its members. More than half of the responding physicians “reported treating complications of a cosmetic procedure performed by an NPO,” the authors wrote.
They also found higher rates of burns and discoloration among patients who were treated by NPOs. The injuries occurred primarily at medical spas.
“Although NPOs may help to meet the rising demand for dermatologic procedures, care should still be taken to prioritize patient safety and outcomes above all else, including financial profits and revenues,” the authors wrote.
Dr. Wehner and her colleagues report no relevant financial relationships. Their study research was supported, in part, by a Cancer Center Support Grant and by the Cancer Prevention and Research Institute of Texas. Dr. Arnold also reports no relevant financial relationships. No author disclosures or funding information were available for the Clinics in Dermatology paper.
*Correction, 12/8/23: An earlier version of this story misstated the study design.
A .
Researchers from the University of Texas MD Anderson Cancer Center, Houston, and the University of Pennsylvania, Philadelphia, reported in JAMA Dermatology that in 2013 APCs made up 28% of the dermatology clinician workforce. By 2020, they made up 37% of the dermatology clinicians giving care to Medicare beneficiaries.
Retrospective cohort study
APCs provided care in 15.5% of dermatology office visits in 2013 and 27.4% in 2020 (P =.02), the authors reported. “By 2020, more than one in four dermatology visits for patients with Medicare were delivered by APCs,” wrote the authors, led by Mackenzie R. Wehner, MD, MPhil, assistant professor of dermatology and health services research at MD Anderson.
“Everyone in dermatology is aware of the increasing adoption of advanced practice clinicians in the field,” Justin D. Arnold, MD, MMSc, a 3rd-year dermatology resident at the University of California, Irvine, said in an interview. “However, seeing how quickly this happening and the absolute number of these clinicians is still startling,” said Dr. Arnold, who in 2022 published a research letter in JAMA Dermatology on the impact of physician assistants in dermatology.
In that study, he and his coauthors reported that the PA workforce in dermatology was growing faster than in other specialties.
In the current study, Dr. Wehner and her colleagues identified 8,444 dermatology APCs and 14,402 physician dermatologists who provided 109.3 million Medicare office visits from 2013 to 2020. More than 80% of the procedures were performed by physicians, but APCs appeared to increasingly be taking on more of the procedural load.
Over the study period, APCs had an average annual increase of 12.6% in the number of premalignant lesion destructions performed; physicians saw an average 1.4% decline. For skin biopsies, APCs performed 11.7% more per year on average, compared with a 1.4% drop for physicians.
“This data is not surprising given most agree that skin biopsies and destruction of premalignant lesions are well within the scope of practice of APCs,” Dr. Arnold told this news organization.
The authors also reported that, while most APCs – similar to physician dermatologists – practice in metropolitan areas, they are working in other locations also. Slightly more than half of dermatology clinicians in micropolitan areas are APCs, and in rural areas, 88% of clinicians are APCs, Dr. Wehner and colleagues found.
APCs may be filling a gap in rural areas for Medicare patients, said Dr. Arnold, but, he added, “it is unclear if dermatology APCs are growing as quickly in practices that predominantly accept Medicaid and if dermatology APCs are expanding access to these populations.”
Dr. Arnold said he expected the number of APCs in dermatology to continue growing, serving commercially insured patients, as well. “There are a multitude of potential reasons for more APCs in dermatology, including difficulty recruiting dermatologists in rural communities, financial motivators, and the expansion of private equity, and the increasing acceptance of these clinicians within medicine and by patients.”
APCs can provide good-quality care if they are properly trained and supervised, said Dr. Arnold, adding that he is concerned, however, that the training and supervision is not being provided. “This study provides further evidence that dermatologists, and national dermatology organizations such as the AAD [American Academy of Dermatology], need to take a more active role in the leadership of APC training,” he said.
Dermatology, he noted, “would benefit from consensus guidelines on clinical competencies for dermatology APCs,” similar to an effort by the American College of Cardiology.
A review* published online in July noted that, compared with dermatologists, some data suggest that non-physician operators (NPOs) may have a higher rate of adverse events when performing aesthetic procedures, according to the authors of the review, led by Shelby L. Kubicki, MD, of the department of dermatology at the UTHealth Science Center in Houston. There is no mandatory reporting of complications for nonphysician providers, so the authors relied on data from cosmetic-focused practices, medical spas, and a survey by the American Society of Dermatologic Surgery of consumers and its members. More than half of the responding physicians “reported treating complications of a cosmetic procedure performed by an NPO,” the authors wrote.
They also found higher rates of burns and discoloration among patients who were treated by NPOs. The injuries occurred primarily at medical spas.
“Although NPOs may help to meet the rising demand for dermatologic procedures, care should still be taken to prioritize patient safety and outcomes above all else, including financial profits and revenues,” the authors wrote.
Dr. Wehner and her colleagues report no relevant financial relationships. Their study research was supported, in part, by a Cancer Center Support Grant and by the Cancer Prevention and Research Institute of Texas. Dr. Arnold also reports no relevant financial relationships. No author disclosures or funding information were available for the Clinics in Dermatology paper.
*Correction, 12/8/23: An earlier version of this story misstated the study design.
A .
Researchers from the University of Texas MD Anderson Cancer Center, Houston, and the University of Pennsylvania, Philadelphia, reported in JAMA Dermatology that in 2013 APCs made up 28% of the dermatology clinician workforce. By 2020, they made up 37% of the dermatology clinicians giving care to Medicare beneficiaries.
Retrospective cohort study
APCs provided care in 15.5% of dermatology office visits in 2013 and 27.4% in 2020 (P =.02), the authors reported. “By 2020, more than one in four dermatology visits for patients with Medicare were delivered by APCs,” wrote the authors, led by Mackenzie R. Wehner, MD, MPhil, assistant professor of dermatology and health services research at MD Anderson.
“Everyone in dermatology is aware of the increasing adoption of advanced practice clinicians in the field,” Justin D. Arnold, MD, MMSc, a 3rd-year dermatology resident at the University of California, Irvine, said in an interview. “However, seeing how quickly this happening and the absolute number of these clinicians is still startling,” said Dr. Arnold, who in 2022 published a research letter in JAMA Dermatology on the impact of physician assistants in dermatology.
In that study, he and his coauthors reported that the PA workforce in dermatology was growing faster than in other specialties.
In the current study, Dr. Wehner and her colleagues identified 8,444 dermatology APCs and 14,402 physician dermatologists who provided 109.3 million Medicare office visits from 2013 to 2020. More than 80% of the procedures were performed by physicians, but APCs appeared to increasingly be taking on more of the procedural load.
Over the study period, APCs had an average annual increase of 12.6% in the number of premalignant lesion destructions performed; physicians saw an average 1.4% decline. For skin biopsies, APCs performed 11.7% more per year on average, compared with a 1.4% drop for physicians.
“This data is not surprising given most agree that skin biopsies and destruction of premalignant lesions are well within the scope of practice of APCs,” Dr. Arnold told this news organization.
The authors also reported that, while most APCs – similar to physician dermatologists – practice in metropolitan areas, they are working in other locations also. Slightly more than half of dermatology clinicians in micropolitan areas are APCs, and in rural areas, 88% of clinicians are APCs, Dr. Wehner and colleagues found.
APCs may be filling a gap in rural areas for Medicare patients, said Dr. Arnold, but, he added, “it is unclear if dermatology APCs are growing as quickly in practices that predominantly accept Medicaid and if dermatology APCs are expanding access to these populations.”
Dr. Arnold said he expected the number of APCs in dermatology to continue growing, serving commercially insured patients, as well. “There are a multitude of potential reasons for more APCs in dermatology, including difficulty recruiting dermatologists in rural communities, financial motivators, and the expansion of private equity, and the increasing acceptance of these clinicians within medicine and by patients.”
APCs can provide good-quality care if they are properly trained and supervised, said Dr. Arnold, adding that he is concerned, however, that the training and supervision is not being provided. “This study provides further evidence that dermatologists, and national dermatology organizations such as the AAD [American Academy of Dermatology], need to take a more active role in the leadership of APC training,” he said.
Dermatology, he noted, “would benefit from consensus guidelines on clinical competencies for dermatology APCs,” similar to an effort by the American College of Cardiology.
A review* published online in July noted that, compared with dermatologists, some data suggest that non-physician operators (NPOs) may have a higher rate of adverse events when performing aesthetic procedures, according to the authors of the review, led by Shelby L. Kubicki, MD, of the department of dermatology at the UTHealth Science Center in Houston. There is no mandatory reporting of complications for nonphysician providers, so the authors relied on data from cosmetic-focused practices, medical spas, and a survey by the American Society of Dermatologic Surgery of consumers and its members. More than half of the responding physicians “reported treating complications of a cosmetic procedure performed by an NPO,” the authors wrote.
They also found higher rates of burns and discoloration among patients who were treated by NPOs. The injuries occurred primarily at medical spas.
“Although NPOs may help to meet the rising demand for dermatologic procedures, care should still be taken to prioritize patient safety and outcomes above all else, including financial profits and revenues,” the authors wrote.
Dr. Wehner and her colleagues report no relevant financial relationships. Their study research was supported, in part, by a Cancer Center Support Grant and by the Cancer Prevention and Research Institute of Texas. Dr. Arnold also reports no relevant financial relationships. No author disclosures or funding information were available for the Clinics in Dermatology paper.
*Correction, 12/8/23: An earlier version of this story misstated the study design.
FROM JAMA DERMATOLOGY
FDA approves first over-the-counter birth control pill
The Food and Drug Administration’s approval today of the first birth control pill for women to be available without a prescription is being hailed by many as a long-needed development, but there remain questions to be resolved, including how much the drug will cost and how it will be used.
The drug, Opill, is expected to be available early next year, and its maker has yet to reveal a retail price. It is the same birth control pill that has been available by prescription for 50 years. But for the first time, women will be able to buy the contraception at a local pharmacy, other retail locations, or online without having to see a doctor first.
Likely to drive debate
Contraception in the United States is not without controversy. The FDA’s approval spurred reactions both for and against making hormonal birth control for women available without a prescription.
“It’s an exciting time, especially right now when reproductive rights are being curtailed in a lot of states. Giving people an additional option for contraception will change people’s lives,” said Beverly Gray, MD, division director of Women’s Community and Population Health at Duke University Medical Center in Durham, N.C.
“It’s a huge win for patients who need better access to contraception,” said Dr. Gray, who is also a spokesperson for the American College of Obstetricians and Gynecologists.
Women who want hormonal birth control but live in areas without convenient access to a doctor, women who cannot easily take time off of work to see a doctor and get a prescription filled, and women without insurance are examples of people who will benefit, she said.
The Catholic Medical Association, in contrast, expressed “deep concern and disappointment” after an FDA advisory committee’s unanimous vote on May 11 recommending the drug be available over the counter. In a statement after the vote, the group cited “extensive medical studies demonstrating the risks and adverse effects of hormonal contraceptives,” adding that “the social impact of [full approval] would be dramatic.”
But doctors largely disagreed.
“It is definitely a huge win for reproductive autonomy. I’m glad that the FDA is prioritizing patient safety and well-being over politics,” said Catherine Cansino, MD, MPH, an ob.gyn. and clinical professor in the University of California Davis department of obstetrics and gynecology. She said the FDA approved the over-the-counter version because the medication is safe.
While opponents like the Catholic Medical Association cite safety concerns and believe doctors should screen all women before prescribing hormonal contraception, Dr. Gray disagreed. “There’s a lot of evidence that patients can figure out if a progestin-only pill is right for them and safe for them. Medical professionals don’t have to be the gatekeepers for contraception,” she said.
Pricing unknown
Whether insurance companies will pay for Opill now that it will be available without a prescription remains unknown. For some medications, paying a copay through insurance can be less expensive than buying at a retail price.
“Although pricing issues will be relevant, the FDA’s decision will enhance women’s access to hormonal birth control,” said Andrew M. Kaunitz, MD, a professor and associate chairman in the department of obstetrics and gynecology at the University of Florida College of Medicine in Jacksonville.
The drugmaker, Perrigo, based in Ireland, has not yet announced how much the pill will cost. The price tag could affect how widely available this form of birth control is. The drug has been shown to be as much as 93% effective for pregnancy prevention. Perrigo says it plans to make the pill available at low or no cost to some women.
Caveats to consider
There are some women for whom hormonal contraceptives have always carried greater risks. For example, women who have breast cancer or a history of breast cancer should not use hormonal contraceptives, the FDA said in a news release announcing the approval. Women with other types of cancer should check with their doctors first, the agency noted.
Women who smoke, who take some medications to lower blood pressure, or who have migraines should also take caution, Dr. Cansino said. “People with migraines may not be suitable for over-the-counter oral contraceptives. But a simple screening through a provider can identify whether you are truly eligible or not.”
Irregular bleeding, headaches, dizziness, nausea, increased appetite, belly pain, cramps, or bloating are the most common side effects of Opill, the FDA said.
The Opill is a progestin-only birth control pill. Similar pills have been available in the United Kingdiom for about 2 years, often referred to as “mini pills” because they contain a single hormone. In contrast, prescription birth control pills in the United States and elsewhere contain more than one hormone, estrogen and progestin, to prevent pregnancy.
Prescription pill packs for combination contraception often feature a week of placebo pills without an active ingredient. While skipping a placebo pill might not make a difference in pregnancy prevention, Opill is different. Every pill in the packet will contain medication, Gray said. “So it’s important to take the pill the same time every day for it to be most effective.”
Even though this may mean one less visit to your doctor, Dr. Kaunitz hopes women will stay up to date on their other medical checkups. “One of our challenges as providers of care to women will be to encourage them to continue to receive important services, including cancer screening and vaccinations, even while they can initiate and continue hormonal contraception without contact with a provider.”
Just the beginning?
The American Medical Association hopes this approval signals more to come.
“While we applaud this move, the AMA continues to urge the FDA and HHS to consider a variety of oral contraceptive options for over-the-counter use,” the association, which has more than 250,000 doctor members, said in a statement. “It is important patients have options when choosing which type of birth control works best for them,”
The American College of Obstetricians and Gynecologists said the FDA’s decision will help many women. “We are glad that more patients will now be empowered to choose when and where they obtain a safe method of contraception without having to wait for a medical appointment or for a prescription to be filled,” Verda J. Hicks, MD, the group’s president, and Christopher M. Zahn, MD, interim chief executive officer, said in a statement.
“Allowing individuals to access birth control at their local pharmacy or drug store will eliminate some barriers,” they said.
A version of this article first appeared on WebMD.com.
This article was updated 7/13/23.
The Food and Drug Administration’s approval today of the first birth control pill for women to be available without a prescription is being hailed by many as a long-needed development, but there remain questions to be resolved, including how much the drug will cost and how it will be used.
The drug, Opill, is expected to be available early next year, and its maker has yet to reveal a retail price. It is the same birth control pill that has been available by prescription for 50 years. But for the first time, women will be able to buy the contraception at a local pharmacy, other retail locations, or online without having to see a doctor first.
Likely to drive debate
Contraception in the United States is not without controversy. The FDA’s approval spurred reactions both for and against making hormonal birth control for women available without a prescription.
“It’s an exciting time, especially right now when reproductive rights are being curtailed in a lot of states. Giving people an additional option for contraception will change people’s lives,” said Beverly Gray, MD, division director of Women’s Community and Population Health at Duke University Medical Center in Durham, N.C.
“It’s a huge win for patients who need better access to contraception,” said Dr. Gray, who is also a spokesperson for the American College of Obstetricians and Gynecologists.
Women who want hormonal birth control but live in areas without convenient access to a doctor, women who cannot easily take time off of work to see a doctor and get a prescription filled, and women without insurance are examples of people who will benefit, she said.
The Catholic Medical Association, in contrast, expressed “deep concern and disappointment” after an FDA advisory committee’s unanimous vote on May 11 recommending the drug be available over the counter. In a statement after the vote, the group cited “extensive medical studies demonstrating the risks and adverse effects of hormonal contraceptives,” adding that “the social impact of [full approval] would be dramatic.”
But doctors largely disagreed.
“It is definitely a huge win for reproductive autonomy. I’m glad that the FDA is prioritizing patient safety and well-being over politics,” said Catherine Cansino, MD, MPH, an ob.gyn. and clinical professor in the University of California Davis department of obstetrics and gynecology. She said the FDA approved the over-the-counter version because the medication is safe.
While opponents like the Catholic Medical Association cite safety concerns and believe doctors should screen all women before prescribing hormonal contraception, Dr. Gray disagreed. “There’s a lot of evidence that patients can figure out if a progestin-only pill is right for them and safe for them. Medical professionals don’t have to be the gatekeepers for contraception,” she said.
Pricing unknown
Whether insurance companies will pay for Opill now that it will be available without a prescription remains unknown. For some medications, paying a copay through insurance can be less expensive than buying at a retail price.
“Although pricing issues will be relevant, the FDA’s decision will enhance women’s access to hormonal birth control,” said Andrew M. Kaunitz, MD, a professor and associate chairman in the department of obstetrics and gynecology at the University of Florida College of Medicine in Jacksonville.
The drugmaker, Perrigo, based in Ireland, has not yet announced how much the pill will cost. The price tag could affect how widely available this form of birth control is. The drug has been shown to be as much as 93% effective for pregnancy prevention. Perrigo says it plans to make the pill available at low or no cost to some women.
Caveats to consider
There are some women for whom hormonal contraceptives have always carried greater risks. For example, women who have breast cancer or a history of breast cancer should not use hormonal contraceptives, the FDA said in a news release announcing the approval. Women with other types of cancer should check with their doctors first, the agency noted.
Women who smoke, who take some medications to lower blood pressure, or who have migraines should also take caution, Dr. Cansino said. “People with migraines may not be suitable for over-the-counter oral contraceptives. But a simple screening through a provider can identify whether you are truly eligible or not.”
Irregular bleeding, headaches, dizziness, nausea, increased appetite, belly pain, cramps, or bloating are the most common side effects of Opill, the FDA said.
The Opill is a progestin-only birth control pill. Similar pills have been available in the United Kingdiom for about 2 years, often referred to as “mini pills” because they contain a single hormone. In contrast, prescription birth control pills in the United States and elsewhere contain more than one hormone, estrogen and progestin, to prevent pregnancy.
Prescription pill packs for combination contraception often feature a week of placebo pills without an active ingredient. While skipping a placebo pill might not make a difference in pregnancy prevention, Opill is different. Every pill in the packet will contain medication, Gray said. “So it’s important to take the pill the same time every day for it to be most effective.”
Even though this may mean one less visit to your doctor, Dr. Kaunitz hopes women will stay up to date on their other medical checkups. “One of our challenges as providers of care to women will be to encourage them to continue to receive important services, including cancer screening and vaccinations, even while they can initiate and continue hormonal contraception without contact with a provider.”
Just the beginning?
The American Medical Association hopes this approval signals more to come.
“While we applaud this move, the AMA continues to urge the FDA and HHS to consider a variety of oral contraceptive options for over-the-counter use,” the association, which has more than 250,000 doctor members, said in a statement. “It is important patients have options when choosing which type of birth control works best for them,”
The American College of Obstetricians and Gynecologists said the FDA’s decision will help many women. “We are glad that more patients will now be empowered to choose when and where they obtain a safe method of contraception without having to wait for a medical appointment or for a prescription to be filled,” Verda J. Hicks, MD, the group’s president, and Christopher M. Zahn, MD, interim chief executive officer, said in a statement.
“Allowing individuals to access birth control at their local pharmacy or drug store will eliminate some barriers,” they said.
A version of this article first appeared on WebMD.com.
This article was updated 7/13/23.
The Food and Drug Administration’s approval today of the first birth control pill for women to be available without a prescription is being hailed by many as a long-needed development, but there remain questions to be resolved, including how much the drug will cost and how it will be used.
The drug, Opill, is expected to be available early next year, and its maker has yet to reveal a retail price. It is the same birth control pill that has been available by prescription for 50 years. But for the first time, women will be able to buy the contraception at a local pharmacy, other retail locations, or online without having to see a doctor first.
Likely to drive debate
Contraception in the United States is not without controversy. The FDA’s approval spurred reactions both for and against making hormonal birth control for women available without a prescription.
“It’s an exciting time, especially right now when reproductive rights are being curtailed in a lot of states. Giving people an additional option for contraception will change people’s lives,” said Beverly Gray, MD, division director of Women’s Community and Population Health at Duke University Medical Center in Durham, N.C.
“It’s a huge win for patients who need better access to contraception,” said Dr. Gray, who is also a spokesperson for the American College of Obstetricians and Gynecologists.
Women who want hormonal birth control but live in areas without convenient access to a doctor, women who cannot easily take time off of work to see a doctor and get a prescription filled, and women without insurance are examples of people who will benefit, she said.
The Catholic Medical Association, in contrast, expressed “deep concern and disappointment” after an FDA advisory committee’s unanimous vote on May 11 recommending the drug be available over the counter. In a statement after the vote, the group cited “extensive medical studies demonstrating the risks and adverse effects of hormonal contraceptives,” adding that “the social impact of [full approval] would be dramatic.”
But doctors largely disagreed.
“It is definitely a huge win for reproductive autonomy. I’m glad that the FDA is prioritizing patient safety and well-being over politics,” said Catherine Cansino, MD, MPH, an ob.gyn. and clinical professor in the University of California Davis department of obstetrics and gynecology. She said the FDA approved the over-the-counter version because the medication is safe.
While opponents like the Catholic Medical Association cite safety concerns and believe doctors should screen all women before prescribing hormonal contraception, Dr. Gray disagreed. “There’s a lot of evidence that patients can figure out if a progestin-only pill is right for them and safe for them. Medical professionals don’t have to be the gatekeepers for contraception,” she said.
Pricing unknown
Whether insurance companies will pay for Opill now that it will be available without a prescription remains unknown. For some medications, paying a copay through insurance can be less expensive than buying at a retail price.
“Although pricing issues will be relevant, the FDA’s decision will enhance women’s access to hormonal birth control,” said Andrew M. Kaunitz, MD, a professor and associate chairman in the department of obstetrics and gynecology at the University of Florida College of Medicine in Jacksonville.
The drugmaker, Perrigo, based in Ireland, has not yet announced how much the pill will cost. The price tag could affect how widely available this form of birth control is. The drug has been shown to be as much as 93% effective for pregnancy prevention. Perrigo says it plans to make the pill available at low or no cost to some women.
Caveats to consider
There are some women for whom hormonal contraceptives have always carried greater risks. For example, women who have breast cancer or a history of breast cancer should not use hormonal contraceptives, the FDA said in a news release announcing the approval. Women with other types of cancer should check with their doctors first, the agency noted.
Women who smoke, who take some medications to lower blood pressure, or who have migraines should also take caution, Dr. Cansino said. “People with migraines may not be suitable for over-the-counter oral contraceptives. But a simple screening through a provider can identify whether you are truly eligible or not.”
Irregular bleeding, headaches, dizziness, nausea, increased appetite, belly pain, cramps, or bloating are the most common side effects of Opill, the FDA said.
The Opill is a progestin-only birth control pill. Similar pills have been available in the United Kingdiom for about 2 years, often referred to as “mini pills” because they contain a single hormone. In contrast, prescription birth control pills in the United States and elsewhere contain more than one hormone, estrogen and progestin, to prevent pregnancy.
Prescription pill packs for combination contraception often feature a week of placebo pills without an active ingredient. While skipping a placebo pill might not make a difference in pregnancy prevention, Opill is different. Every pill in the packet will contain medication, Gray said. “So it’s important to take the pill the same time every day for it to be most effective.”
Even though this may mean one less visit to your doctor, Dr. Kaunitz hopes women will stay up to date on their other medical checkups. “One of our challenges as providers of care to women will be to encourage them to continue to receive important services, including cancer screening and vaccinations, even while they can initiate and continue hormonal contraception without contact with a provider.”
Just the beginning?
The American Medical Association hopes this approval signals more to come.
“While we applaud this move, the AMA continues to urge the FDA and HHS to consider a variety of oral contraceptive options for over-the-counter use,” the association, which has more than 250,000 doctor members, said in a statement. “It is important patients have options when choosing which type of birth control works best for them,”
The American College of Obstetricians and Gynecologists said the FDA’s decision will help many women. “We are glad that more patients will now be empowered to choose when and where they obtain a safe method of contraception without having to wait for a medical appointment or for a prescription to be filled,” Verda J. Hicks, MD, the group’s president, and Christopher M. Zahn, MD, interim chief executive officer, said in a statement.
“Allowing individuals to access birth control at their local pharmacy or drug store will eliminate some barriers,” they said.
A version of this article first appeared on WebMD.com.
This article was updated 7/13/23.
Porocarcinoma Development in a Prior Trauma Site
To the Editor:
Porocarcinoma, or malignant poroma, is a rare adnexal malignancy of a predominantly glandular origin that comprises less than 0.01% of all cutaneous neoplasms.1,2 Although exposure to UV radiation and immunosuppression have been implicated in the malignant degeneration of benign poromas into porocarcinomas, at least half of all malignant variants will arise de novo.3,4 Patients present with an evolving nodule or plaque and often are in their seventh or eighth decade of life at the time of diagnosis.2 Localized trauma from burns or radiation exposure has been causatively linked to de novo porocarcinoma formation.2,5 These suppressive and traumatic stimuli drive increased genetic heterogeneity along with characteristic gene mutations in known tumor suppressor genes.6
A 62-year-old man presented with a nonhealing wound on the right hand of 5 years’ duration that had previously been attributed to a penetrating injury with a piece of copper from a refrigerant coolant system. The wound initially blistered and then eventually callused and developed areas of ulceration. The patient consulted multiple physicians for treatment of the intensely pruritic and ulcerated lesion. He received prescriptions for cephalexin, trimethoprim-sulfamethoxazole, doxycycline, clindamycin, and clobetasol cream, all of which offered minimal improvement. Home therapies including vitamin E and tea tree oil yielded no benefit. The lesion roughly quadrupled in size over the last 5 years.
Physical examination revealed a 7.5×4.2-cm ulcerated plaque with ragged borders and abundant central neoepithelialization on the right palmar surface (Figure 1). No gross motor or sensory defects were identified. There was no epitrochlear, axillary, cervical, or supraclavicular lymphadenopathy. A shave biopsy of the plaque’s edge was performed, which demonstrated a hyperplastic epidermis comprising atypical poroid cells with frequent mitoses, scant necrosis, and regular ductal structures confined to the epidermis (Figure 2). Immunohistochemical profiling results were positive for anticytokeratin (CAM 5.2) and Ber-EP4 (Figure 3). When evaluated in aggregate, these findings were consistent with porocarcinoma in situ.
The patient was referred to a surgical oncologist for evaluation. At that time, an exophytic mass had developed in the central lesion. Although no lymphadenopathy was identified upon examination, the patient had developed tremoring and a contracture deformity of the right hand. Extensive imaging and urgent surgical resection were recommended, but the patient did not wish to pursue these options, opting instead to continue home remedies. At a 15-month follow-up via telephone, the patient reported that the home therapy had failed and he had moved back to Vietnam. Partial limb amputation had been recommended by a local provider. Unfortunately, the patient was subsequently lost to follow-up, and his current status is unknown.
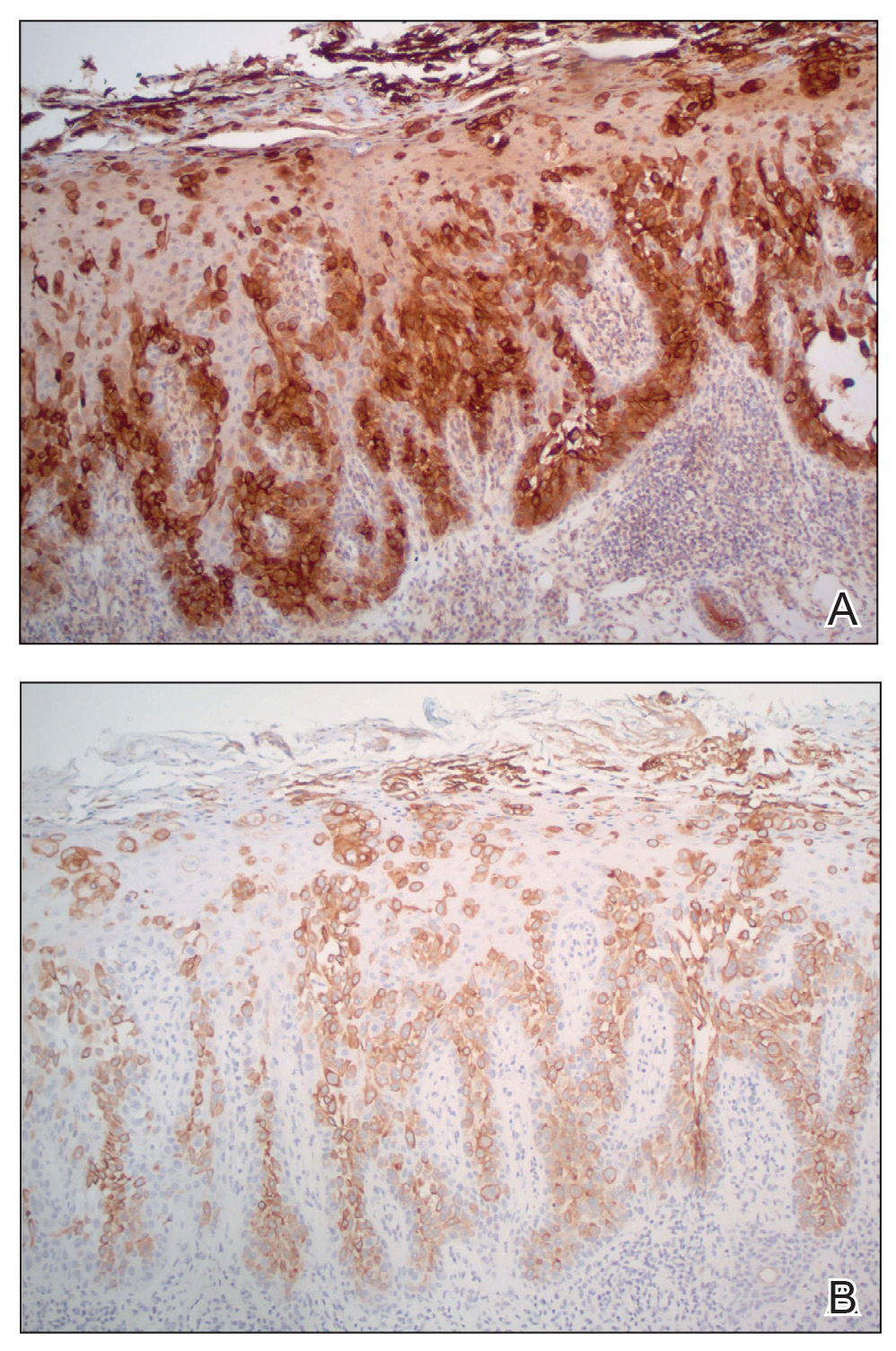
Porocarcinomas are rare tumors, comprising just 0.005% to 0.01% of all cutaneous epithelial tumors.1,2,5 They affect men and women equally, with an average age at diagnosis of 60 to 70 years.1,2 At least half of all porocarcinomas develop de novo, while 18% to 50% arise from the degeneration of an existing poroma.2,3 Exposure to UV light and immunosuppression, particularly following organ transplantation, represent 2 commonly suspected catalysts for this malignant transformation.4 De novo porocarcinomas are most causatively linked to localized trauma from burns or radiation exposure.5 Gene mutations in classic tumor suppressor genes—tumor protein p53 (TP53), phosphatase and tensin homolog (PTEN), rearranged during transfection (RET), adenomatous polyposis coli (APC)—and increased genetic heterogeneity follow these stimuli.6
The morphologic presentation of porocarcinoma is highly variable and may manifest as papules, nodules, or plaques in various states of erosion, ulceration, or excoriation. Diagnoses of basal and squamous cell carcinoma, primary adnexal tumors, seborrheic keratosis, pyogenic granuloma, and melanoma must all be considered and methodically ruled out.7 Porocarcinomas may arise nearly anywhere on the body, with a particular predilection for the lower extremities (35%), head/neck (24%), and upper extremities (14%).3,4 Primary lesions arising from the extremities, genitalia, or buttocks herald a higher risk for lymphatic invasion and distant metastasis, while head and neck tumors more commonly remain localized.8 Bleeding, ulceration, or rapid expansion of a preexisting poroma is suggestive of malignant transformation and may portend a more aggressive disease pattern.2,9
Unequivocal diagnosis relies on histological and immunohistochemical studies due to the marked clinical variance of this neoplasm.7 An irregular histologic pattern of poromatous basaloid cells with ductal differentiation and cytologic atypia commonly are seen with porocarcinomas.2,8 Nuclear pleomorphism with cellular necrosis, increased mitotic figures, and abortive ductal formation with a distinct lack of retraction around cellular aggregates often are found. Immunohistochemical staining is needed to confirm the primary tumor diagnosis. Histochemical stains commonly employed include carcinoembryonic antigen (CEA), cytokeratin AE1/AE3, epithelial membrane antigen, p53, p63, Ki67, and periodic acid-Schiff.10 The use of BerEP4 has been reported as efficacious in highlighting sweat structures, which can be particularly useful in cases when basal cell carcinoma is not in the histologic differential.11 These staining profiles afford confirmation of ductal differentiation with CEA, epithelial membrane antigen, and BerEP4, while p63 and Ki67 are used as surrogates for primary cutaneous neoplasia and cell proliferation, respectively.5,11 Porocarcinoma lesions may be most sensitive to CEA and most specific to CK19 (a component of cytokeratin AE1/AE3), though these findings have not been widely reproduced.7
The treatment and prognosis of porocarcinoma vary widely. Surgically excised lesions recur in roughly 20% of cases, though these rates likely include tumors that were incompletely resected in the primary attempt. Although wide local excision with an average 1-cm margin remains the most employed removal technique, Mohs micrographic surgery may more effectively limit recurrence and metastasis of localized disease.7,8,12 Metastatic disease foretells a mortality rate of at least 65%, which is problematic in that 10% to 20% of patients have metastatic disease at the time of diagnosis and another 20% will show metastasis following primary tumor excision.8,10 Neoplasms with high mitotic rates and depths greater than 7 mm should prompt thorough diagnostic imaging, such as positron emission tomography or magnetic resonance imaging. A sentinel lymph node biopsy should be strongly considered and discussed with the patient.10 Treatment options for nodal and distant metastases include a combination of localized surgery, lymphadenectomy, radiotherapy, and chemotherapeutic agents.2,4,5 The response to systemic treatment and radiotherapy often is quite poor, though the use of combinations of docetaxel, paclitaxel, cetuximab, and immunotherapy have been efficacious in smaller studies.8,10 The highest rates of morbidity and mortality are seen in patients with metastases on presentation or with localized tumors in the groin and buttocks.8
The diagnosis of porocarcinoma may be elusive due to its relatively rare occurrence. Therefore, it is critical to consider this neoplasm in high-risk sites in older patients who present with an evolving nodule or tumor on an extremity. Routine histology and astute histochemical profiling are necessary to exclude diseases that mimic porocarcinoma. Once diagnosis is confirmed, management with prompt excision and diagnostic imaging is recommended, including a lymph node biopsy if appropriate. Due to its high metastatic potential and associated morbidity and mortality, patients with porocarcinoma should be followed closely by a multidisciplinary care team.
- Belin E, Ezzedine K, Stanislas S, et al. Factors in the surgical management of primary eccrine porocarcinoma: prognostic histological factors can guide the surgical procedure. Br J Dermatol. 2011;165:985-989.
- Robson A, Greene J, Ansari N, et al. Eccrine porocarcinoma (malignant eccrine poroma): a clinicopathologic study of 69 cases. Am J Surg Pathol. 2001;25:710-720.
- Spencer DM, Bigler LR, Hearne DW, et al. Pedal papule. eccrine porocarcinoma (EPC) in association with poroma. Arch Dermatol. 1995;131:211, 214.
- Salih AM, Kakamad FH, Essa RA, et al. Porocarcinoma: a systematic review of literature with a single case report. Int J Surg Case Rep. 2017;30:13-16.
- Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Mosby Elsevier; 2018.
- Bosic M, Kirchner M, Brasanac D, et al. Targeted molecular profiling reveals genetic heterogeneity of poromas and porocarcinomas. Pathology. 2018;50:327-332.
- Mahalingam M, Richards JE, Selim MA, et al. An immunohistochemical comparison of cytokeratin 7, cytokeratin 15, cytokeratin 19, CAM 5.2, carcinoembryonic antigen, and nestin in differentiating porocarcinoma from squamous cell carcinoma. Hum Pathol. 2012;43:1265-1272.
- Nazemi A, Higgins S, Swift R, et al. Eccrine porocarcinoma: new insights and a systematic review of the literature. Dermatol Surg. 2018;44:1247-1261.
- Wen SY. Case report of eccrine porocarcinoma in situ associated with eccrine poroma on the forehead. J Dermatol. 2012;39:649-651.
- Gerber PA, Schulte KW, Ruzicka T, et al. Eccrine porocarcinoma of the head: an important differential diagnosis in the elderly patient. Dermatology. 2008;216:229-233.
- Afshar M, Deroide F, Robson A. BerEP4 is widely expressed in tumors of the sweat apparatus: a source of potential diagnostic error. J Cutan Pathol. 2013;40:259-264.
- Tolkachjov SN, Hocker TL, Camilleri MJ, et al. Treatment of porocarcinoma with Mohs micrographic surgery: the Mayo clinic experience. Dermatol Surg. 2016;42:745-750.
To the Editor:
Porocarcinoma, or malignant poroma, is a rare adnexal malignancy of a predominantly glandular origin that comprises less than 0.01% of all cutaneous neoplasms.1,2 Although exposure to UV radiation and immunosuppression have been implicated in the malignant degeneration of benign poromas into porocarcinomas, at least half of all malignant variants will arise de novo.3,4 Patients present with an evolving nodule or plaque and often are in their seventh or eighth decade of life at the time of diagnosis.2 Localized trauma from burns or radiation exposure has been causatively linked to de novo porocarcinoma formation.2,5 These suppressive and traumatic stimuli drive increased genetic heterogeneity along with characteristic gene mutations in known tumor suppressor genes.6
A 62-year-old man presented with a nonhealing wound on the right hand of 5 years’ duration that had previously been attributed to a penetrating injury with a piece of copper from a refrigerant coolant system. The wound initially blistered and then eventually callused and developed areas of ulceration. The patient consulted multiple physicians for treatment of the intensely pruritic and ulcerated lesion. He received prescriptions for cephalexin, trimethoprim-sulfamethoxazole, doxycycline, clindamycin, and clobetasol cream, all of which offered minimal improvement. Home therapies including vitamin E and tea tree oil yielded no benefit. The lesion roughly quadrupled in size over the last 5 years.

Physical examination revealed a 7.5×4.2-cm ulcerated plaque with ragged borders and abundant central neoepithelialization on the right palmar surface (Figure 1). No gross motor or sensory defects were identified. There was no epitrochlear, axillary, cervical, or supraclavicular lymphadenopathy. A shave biopsy of the plaque’s edge was performed, which demonstrated a hyperplastic epidermis comprising atypical poroid cells with frequent mitoses, scant necrosis, and regular ductal structures confined to the epidermis (Figure 2). Immunohistochemical profiling results were positive for anticytokeratin (CAM 5.2) and Ber-EP4 (Figure 3). When evaluated in aggregate, these findings were consistent with porocarcinoma in situ.

The patient was referred to a surgical oncologist for evaluation. At that time, an exophytic mass had developed in the central lesion. Although no lymphadenopathy was identified upon examination, the patient had developed tremoring and a contracture deformity of the right hand. Extensive imaging and urgent surgical resection were recommended, but the patient did not wish to pursue these options, opting instead to continue home remedies. At a 15-month follow-up via telephone, the patient reported that the home therapy had failed and he had moved back to Vietnam. Partial limb amputation had been recommended by a local provider. Unfortunately, the patient was subsequently lost to follow-up, and his current status is unknown.

Porocarcinomas are rare tumors, comprising just 0.005% to 0.01% of all cutaneous epithelial tumors.1,2,5 They affect men and women equally, with an average age at diagnosis of 60 to 70 years.1,2 At least half of all porocarcinomas develop de novo, while 18% to 50% arise from the degeneration of an existing poroma.2,3 Exposure to UV light and immunosuppression, particularly following organ transplantation, represent 2 commonly suspected catalysts for this malignant transformation.4 De novo porocarcinomas are most causatively linked to localized trauma from burns or radiation exposure.5 Gene mutations in classic tumor suppressor genes—tumor protein p53 (TP53), phosphatase and tensin homolog (PTEN), rearranged during transfection (RET), adenomatous polyposis coli (APC)—and increased genetic heterogeneity follow these stimuli.6
The morphologic presentation of porocarcinoma is highly variable and may manifest as papules, nodules, or plaques in various states of erosion, ulceration, or excoriation. Diagnoses of basal and squamous cell carcinoma, primary adnexal tumors, seborrheic keratosis, pyogenic granuloma, and melanoma must all be considered and methodically ruled out.7 Porocarcinomas may arise nearly anywhere on the body, with a particular predilection for the lower extremities (35%), head/neck (24%), and upper extremities (14%).3,4 Primary lesions arising from the extremities, genitalia, or buttocks herald a higher risk for lymphatic invasion and distant metastasis, while head and neck tumors more commonly remain localized.8 Bleeding, ulceration, or rapid expansion of a preexisting poroma is suggestive of malignant transformation and may portend a more aggressive disease pattern.2,9
Unequivocal diagnosis relies on histological and immunohistochemical studies due to the marked clinical variance of this neoplasm.7 An irregular histologic pattern of poromatous basaloid cells with ductal differentiation and cytologic atypia commonly are seen with porocarcinomas.2,8 Nuclear pleomorphism with cellular necrosis, increased mitotic figures, and abortive ductal formation with a distinct lack of retraction around cellular aggregates often are found. Immunohistochemical staining is needed to confirm the primary tumor diagnosis. Histochemical stains commonly employed include carcinoembryonic antigen (CEA), cytokeratin AE1/AE3, epithelial membrane antigen, p53, p63, Ki67, and periodic acid-Schiff.10 The use of BerEP4 has been reported as efficacious in highlighting sweat structures, which can be particularly useful in cases when basal cell carcinoma is not in the histologic differential.11 These staining profiles afford confirmation of ductal differentiation with CEA, epithelial membrane antigen, and BerEP4, while p63 and Ki67 are used as surrogates for primary cutaneous neoplasia and cell proliferation, respectively.5,11 Porocarcinoma lesions may be most sensitive to CEA and most specific to CK19 (a component of cytokeratin AE1/AE3), though these findings have not been widely reproduced.7
The treatment and prognosis of porocarcinoma vary widely. Surgically excised lesions recur in roughly 20% of cases, though these rates likely include tumors that were incompletely resected in the primary attempt. Although wide local excision with an average 1-cm margin remains the most employed removal technique, Mohs micrographic surgery may more effectively limit recurrence and metastasis of localized disease.7,8,12 Metastatic disease foretells a mortality rate of at least 65%, which is problematic in that 10% to 20% of patients have metastatic disease at the time of diagnosis and another 20% will show metastasis following primary tumor excision.8,10 Neoplasms with high mitotic rates and depths greater than 7 mm should prompt thorough diagnostic imaging, such as positron emission tomography or magnetic resonance imaging. A sentinel lymph node biopsy should be strongly considered and discussed with the patient.10 Treatment options for nodal and distant metastases include a combination of localized surgery, lymphadenectomy, radiotherapy, and chemotherapeutic agents.2,4,5 The response to systemic treatment and radiotherapy often is quite poor, though the use of combinations of docetaxel, paclitaxel, cetuximab, and immunotherapy have been efficacious in smaller studies.8,10 The highest rates of morbidity and mortality are seen in patients with metastases on presentation or with localized tumors in the groin and buttocks.8
The diagnosis of porocarcinoma may be elusive due to its relatively rare occurrence. Therefore, it is critical to consider this neoplasm in high-risk sites in older patients who present with an evolving nodule or tumor on an extremity. Routine histology and astute histochemical profiling are necessary to exclude diseases that mimic porocarcinoma. Once diagnosis is confirmed, management with prompt excision and diagnostic imaging is recommended, including a lymph node biopsy if appropriate. Due to its high metastatic potential and associated morbidity and mortality, patients with porocarcinoma should be followed closely by a multidisciplinary care team.
To the Editor:
Porocarcinoma, or malignant poroma, is a rare adnexal malignancy of a predominantly glandular origin that comprises less than 0.01% of all cutaneous neoplasms.1,2 Although exposure to UV radiation and immunosuppression have been implicated in the malignant degeneration of benign poromas into porocarcinomas, at least half of all malignant variants will arise de novo.3,4 Patients present with an evolving nodule or plaque and often are in their seventh or eighth decade of life at the time of diagnosis.2 Localized trauma from burns or radiation exposure has been causatively linked to de novo porocarcinoma formation.2,5 These suppressive and traumatic stimuli drive increased genetic heterogeneity along with characteristic gene mutations in known tumor suppressor genes.6
A 62-year-old man presented with a nonhealing wound on the right hand of 5 years’ duration that had previously been attributed to a penetrating injury with a piece of copper from a refrigerant coolant system. The wound initially blistered and then eventually callused and developed areas of ulceration. The patient consulted multiple physicians for treatment of the intensely pruritic and ulcerated lesion. He received prescriptions for cephalexin, trimethoprim-sulfamethoxazole, doxycycline, clindamycin, and clobetasol cream, all of which offered minimal improvement. Home therapies including vitamin E and tea tree oil yielded no benefit. The lesion roughly quadrupled in size over the last 5 years.

Physical examination revealed a 7.5×4.2-cm ulcerated plaque with ragged borders and abundant central neoepithelialization on the right palmar surface (Figure 1). No gross motor or sensory defects were identified. There was no epitrochlear, axillary, cervical, or supraclavicular lymphadenopathy. A shave biopsy of the plaque’s edge was performed, which demonstrated a hyperplastic epidermis comprising atypical poroid cells with frequent mitoses, scant necrosis, and regular ductal structures confined to the epidermis (Figure 2). Immunohistochemical profiling results were positive for anticytokeratin (CAM 5.2) and Ber-EP4 (Figure 3). When evaluated in aggregate, these findings were consistent with porocarcinoma in situ.

The patient was referred to a surgical oncologist for evaluation. At that time, an exophytic mass had developed in the central lesion. Although no lymphadenopathy was identified upon examination, the patient had developed tremoring and a contracture deformity of the right hand. Extensive imaging and urgent surgical resection were recommended, but the patient did not wish to pursue these options, opting instead to continue home remedies. At a 15-month follow-up via telephone, the patient reported that the home therapy had failed and he had moved back to Vietnam. Partial limb amputation had been recommended by a local provider. Unfortunately, the patient was subsequently lost to follow-up, and his current status is unknown.

Porocarcinomas are rare tumors, comprising just 0.005% to 0.01% of all cutaneous epithelial tumors.1,2,5 They affect men and women equally, with an average age at diagnosis of 60 to 70 years.1,2 At least half of all porocarcinomas develop de novo, while 18% to 50% arise from the degeneration of an existing poroma.2,3 Exposure to UV light and immunosuppression, particularly following organ transplantation, represent 2 commonly suspected catalysts for this malignant transformation.4 De novo porocarcinomas are most causatively linked to localized trauma from burns or radiation exposure.5 Gene mutations in classic tumor suppressor genes—tumor protein p53 (TP53), phosphatase and tensin homolog (PTEN), rearranged during transfection (RET), adenomatous polyposis coli (APC)—and increased genetic heterogeneity follow these stimuli.6
The morphologic presentation of porocarcinoma is highly variable and may manifest as papules, nodules, or plaques in various states of erosion, ulceration, or excoriation. Diagnoses of basal and squamous cell carcinoma, primary adnexal tumors, seborrheic keratosis, pyogenic granuloma, and melanoma must all be considered and methodically ruled out.7 Porocarcinomas may arise nearly anywhere on the body, with a particular predilection for the lower extremities (35%), head/neck (24%), and upper extremities (14%).3,4 Primary lesions arising from the extremities, genitalia, or buttocks herald a higher risk for lymphatic invasion and distant metastasis, while head and neck tumors more commonly remain localized.8 Bleeding, ulceration, or rapid expansion of a preexisting poroma is suggestive of malignant transformation and may portend a more aggressive disease pattern.2,9
Unequivocal diagnosis relies on histological and immunohistochemical studies due to the marked clinical variance of this neoplasm.7 An irregular histologic pattern of poromatous basaloid cells with ductal differentiation and cytologic atypia commonly are seen with porocarcinomas.2,8 Nuclear pleomorphism with cellular necrosis, increased mitotic figures, and abortive ductal formation with a distinct lack of retraction around cellular aggregates often are found. Immunohistochemical staining is needed to confirm the primary tumor diagnosis. Histochemical stains commonly employed include carcinoembryonic antigen (CEA), cytokeratin AE1/AE3, epithelial membrane antigen, p53, p63, Ki67, and periodic acid-Schiff.10 The use of BerEP4 has been reported as efficacious in highlighting sweat structures, which can be particularly useful in cases when basal cell carcinoma is not in the histologic differential.11 These staining profiles afford confirmation of ductal differentiation with CEA, epithelial membrane antigen, and BerEP4, while p63 and Ki67 are used as surrogates for primary cutaneous neoplasia and cell proliferation, respectively.5,11 Porocarcinoma lesions may be most sensitive to CEA and most specific to CK19 (a component of cytokeratin AE1/AE3), though these findings have not been widely reproduced.7
The treatment and prognosis of porocarcinoma vary widely. Surgically excised lesions recur in roughly 20% of cases, though these rates likely include tumors that were incompletely resected in the primary attempt. Although wide local excision with an average 1-cm margin remains the most employed removal technique, Mohs micrographic surgery may more effectively limit recurrence and metastasis of localized disease.7,8,12 Metastatic disease foretells a mortality rate of at least 65%, which is problematic in that 10% to 20% of patients have metastatic disease at the time of diagnosis and another 20% will show metastasis following primary tumor excision.8,10 Neoplasms with high mitotic rates and depths greater than 7 mm should prompt thorough diagnostic imaging, such as positron emission tomography or magnetic resonance imaging. A sentinel lymph node biopsy should be strongly considered and discussed with the patient.10 Treatment options for nodal and distant metastases include a combination of localized surgery, lymphadenectomy, radiotherapy, and chemotherapeutic agents.2,4,5 The response to systemic treatment and radiotherapy often is quite poor, though the use of combinations of docetaxel, paclitaxel, cetuximab, and immunotherapy have been efficacious in smaller studies.8,10 The highest rates of morbidity and mortality are seen in patients with metastases on presentation or with localized tumors in the groin and buttocks.8
The diagnosis of porocarcinoma may be elusive due to its relatively rare occurrence. Therefore, it is critical to consider this neoplasm in high-risk sites in older patients who present with an evolving nodule or tumor on an extremity. Routine histology and astute histochemical profiling are necessary to exclude diseases that mimic porocarcinoma. Once diagnosis is confirmed, management with prompt excision and diagnostic imaging is recommended, including a lymph node biopsy if appropriate. Due to its high metastatic potential and associated morbidity and mortality, patients with porocarcinoma should be followed closely by a multidisciplinary care team.
- Belin E, Ezzedine K, Stanislas S, et al. Factors in the surgical management of primary eccrine porocarcinoma: prognostic histological factors can guide the surgical procedure. Br J Dermatol. 2011;165:985-989.
- Robson A, Greene J, Ansari N, et al. Eccrine porocarcinoma (malignant eccrine poroma): a clinicopathologic study of 69 cases. Am J Surg Pathol. 2001;25:710-720.
- Spencer DM, Bigler LR, Hearne DW, et al. Pedal papule. eccrine porocarcinoma (EPC) in association with poroma. Arch Dermatol. 1995;131:211, 214.
- Salih AM, Kakamad FH, Essa RA, et al. Porocarcinoma: a systematic review of literature with a single case report. Int J Surg Case Rep. 2017;30:13-16.
- Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Mosby Elsevier; 2018.
- Bosic M, Kirchner M, Brasanac D, et al. Targeted molecular profiling reveals genetic heterogeneity of poromas and porocarcinomas. Pathology. 2018;50:327-332.
- Mahalingam M, Richards JE, Selim MA, et al. An immunohistochemical comparison of cytokeratin 7, cytokeratin 15, cytokeratin 19, CAM 5.2, carcinoembryonic antigen, and nestin in differentiating porocarcinoma from squamous cell carcinoma. Hum Pathol. 2012;43:1265-1272.
- Nazemi A, Higgins S, Swift R, et al. Eccrine porocarcinoma: new insights and a systematic review of the literature. Dermatol Surg. 2018;44:1247-1261.
- Wen SY. Case report of eccrine porocarcinoma in situ associated with eccrine poroma on the forehead. J Dermatol. 2012;39:649-651.
- Gerber PA, Schulte KW, Ruzicka T, et al. Eccrine porocarcinoma of the head: an important differential diagnosis in the elderly patient. Dermatology. 2008;216:229-233.
- Afshar M, Deroide F, Robson A. BerEP4 is widely expressed in tumors of the sweat apparatus: a source of potential diagnostic error. J Cutan Pathol. 2013;40:259-264.
- Tolkachjov SN, Hocker TL, Camilleri MJ, et al. Treatment of porocarcinoma with Mohs micrographic surgery: the Mayo clinic experience. Dermatol Surg. 2016;42:745-750.
- Belin E, Ezzedine K, Stanislas S, et al. Factors in the surgical management of primary eccrine porocarcinoma: prognostic histological factors can guide the surgical procedure. Br J Dermatol. 2011;165:985-989.
- Robson A, Greene J, Ansari N, et al. Eccrine porocarcinoma (malignant eccrine poroma): a clinicopathologic study of 69 cases. Am J Surg Pathol. 2001;25:710-720.
- Spencer DM, Bigler LR, Hearne DW, et al. Pedal papule. eccrine porocarcinoma (EPC) in association with poroma. Arch Dermatol. 1995;131:211, 214.
- Salih AM, Kakamad FH, Essa RA, et al. Porocarcinoma: a systematic review of literature with a single case report. Int J Surg Case Rep. 2017;30:13-16.
- Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Mosby Elsevier; 2018.
- Bosic M, Kirchner M, Brasanac D, et al. Targeted molecular profiling reveals genetic heterogeneity of poromas and porocarcinomas. Pathology. 2018;50:327-332.
- Mahalingam M, Richards JE, Selim MA, et al. An immunohistochemical comparison of cytokeratin 7, cytokeratin 15, cytokeratin 19, CAM 5.2, carcinoembryonic antigen, and nestin in differentiating porocarcinoma from squamous cell carcinoma. Hum Pathol. 2012;43:1265-1272.
- Nazemi A, Higgins S, Swift R, et al. Eccrine porocarcinoma: new insights and a systematic review of the literature. Dermatol Surg. 2018;44:1247-1261.
- Wen SY. Case report of eccrine porocarcinoma in situ associated with eccrine poroma on the forehead. J Dermatol. 2012;39:649-651.
- Gerber PA, Schulte KW, Ruzicka T, et al. Eccrine porocarcinoma of the head: an important differential diagnosis in the elderly patient. Dermatology. 2008;216:229-233.
- Afshar M, Deroide F, Robson A. BerEP4 is widely expressed in tumors of the sweat apparatus: a source of potential diagnostic error. J Cutan Pathol. 2013;40:259-264.
- Tolkachjov SN, Hocker TL, Camilleri MJ, et al. Treatment of porocarcinoma with Mohs micrographic surgery: the Mayo clinic experience. Dermatol Surg. 2016;42:745-750.
Practice Points
- Porocarcinoma is a rare, potentially aggressive, glandular malignancy that should be a clinical consideration in patients presenting with a cutaneous neoplasm.
- Although wide local excision historically has been the treatment of choice for porocarcinoma, Mohs micrographic surgery has demonstrated excellent cure rates.
- Patients with unresectable or metastatic porocarcinomas have a poor prognosis but may respond to combination chemotherapy regimens.
Genital Ulcerations With Swelling
The Diagnosis: Mpox (Monkeypox)
Tests for active herpes simplex virus (HHV), gonorrhea, chlamydia, HIV, and syphilis were negative. Swabs from the penile lesion demonstrated positivity for the West African clade of mpox (monkeypox) virus (MPXV) by polymerase chain reaction. The patient was treated supportively without the addition of antiviral therapy, and he experienced a complete recovery.
Mpox virus was first isolated in 1958 in a research facility and was named after the laboratory animals that were housed there. The first human documentation of the disease occurred in 1970, and it was first documented in the United States in 2003 in an infection that was traced to a shipment of small mammals from Ghana to Texas.1 The disease has always been endemic to Africa; however, the incidence has been increasing.2 A new MPXV outbreak was reported in many countries in early 2022, including the United States.1
The MPXV is a double-stranded DNA virus of the genus Orthopoxvirus, and 2 genetic clades have been identified: clade I (formerly the Central African clade) and clade II (formerly the West African clade). The virus has the capability to infect many mammals; however, its host remains unidentified.1 The exact mechanism of transmission from infected animals to humans largely is unknown; however, direct or indirect contact with infected animals likely is responsible. Human-to-human transmission can occur by many mechanisms including contact with large respiratory droplets, bodily fluids, and contaminated surfaces. The incubation period is 5 to 21 days, and the symptoms last 2 to 5 weeks.1
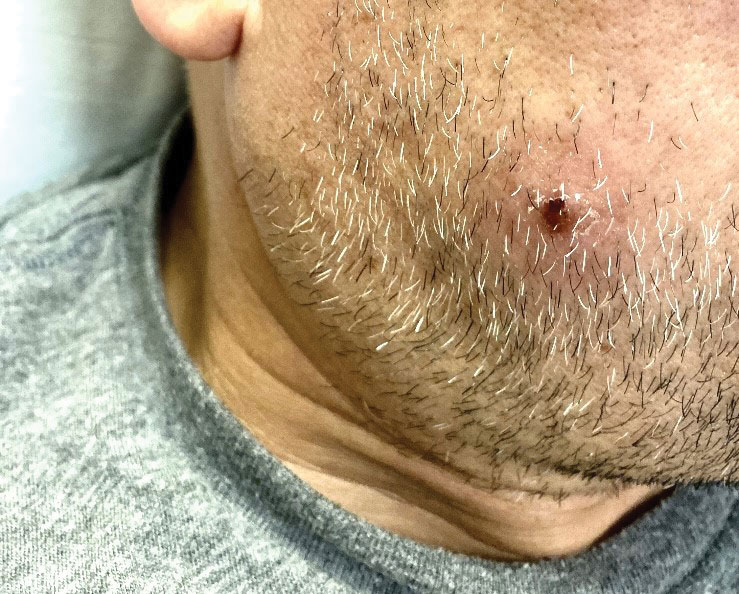
The clinical manifestations of MPXV during the most recent outbreak differ from prior outbreaks. Patients are more likely to experience minimal to no systemic symptoms, and cutaneous lesions can be few and localized to a focal area, especially on the face and in the anogenital region,3 similar to the presentation in our patient (Figure 1). Cutaneous lesions of the most recent MPXV outbreak also include painless ulcerations similar to syphilitic chancres and lesions that are in various stages of healing.3 Lesions often begin as pseudopustules, which are firm white papules with or without a necrotic center that resemble pustules; unlike true pustules, there is no identifiable purulent material within it. Bacterial superinfection of the lesions is not uncommon.4 Over time, a secondary pustular eruption resembling folliculitis also may occur,4 as noted in our patient (Figure 2).
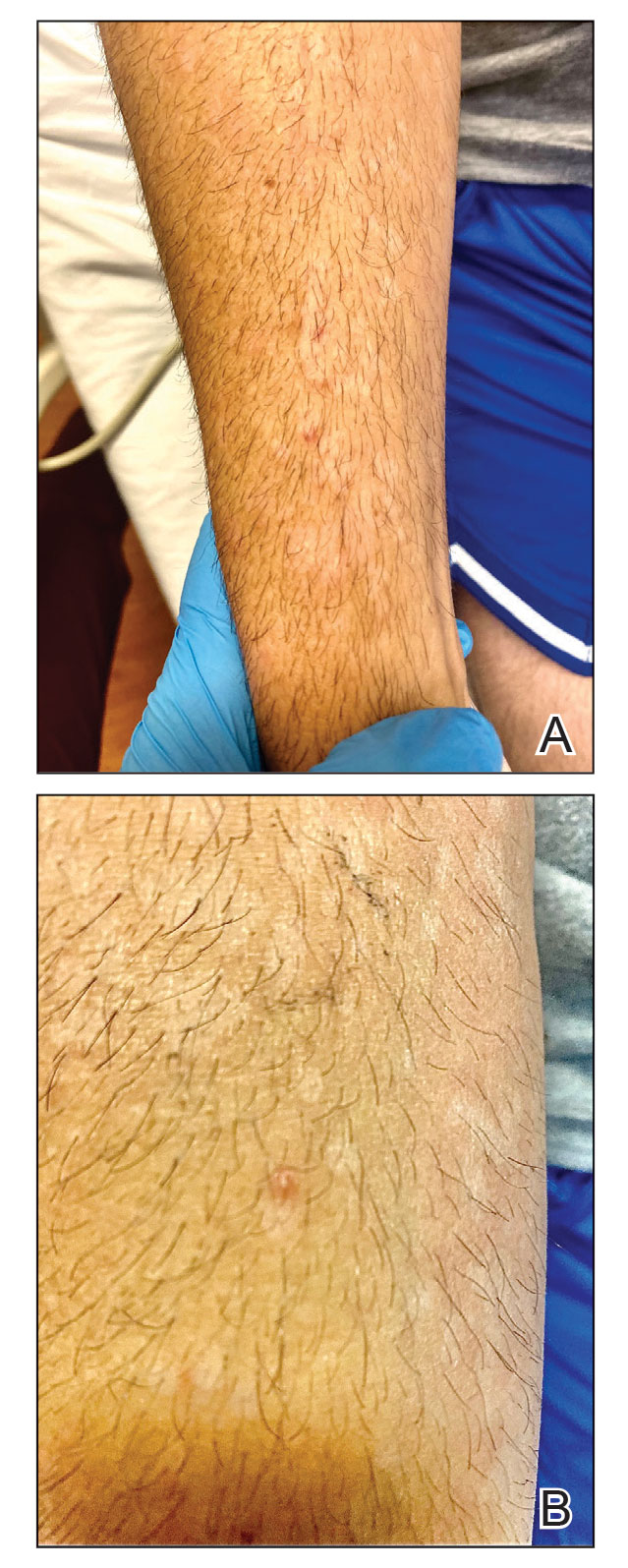
Although we did not have a biopsy to support the diagnosis of associated erythema multiforme (EM) in our patient, features supportive of this diagnosis included the classic clinical appearance of typical, well-defined, targetoid plaques with 3 distinct zones (Figure 3); the association with a known infection; the distribution on the arms with truncal sparing; and self-limited lesions. More than 90% of EM cases are associated with infection, with HHV representing the most common culprit5; therefore, the relationship with a different virus is not an unreasonable suggestion. Additionally, there have been rare reports of EM in association with MPXV.4
Histopathology of MPXV may have distinctive features. Lesions often demonstrate keratinocytic necrosis and basal layer vacuolization with an associated superficial and deep perivascular lymphohistiocytic infiltrate. When the morphology of the lesion is vesicular, histopathology reveals spongiosis and ballooning degeneration with epidermal necrosis. Viral inclusion bodies within keratinocytes may be identified.1 Death rates from MPXV has been reported from 1% to 11%, with increased mortality among high-risk populations including children and immunocompromised individuals. Treatment of the disease largely consists of supportive care and management of any associated complications including bacterial infection, pneumonia, and encephalitis.1
The differential diagnosis of MPXV includes other ulcerative lesions that can occur on the genital skin. Fixed drug eruptions often present on the penis,6 but there was no identifiable inciting drug in our patient. Herpes simplex virus infection was very high on the differential given our patient’s history of recurrent infections and association with a targetoid rash, but HHV type 1 and HHV type 2 testing of the lesion was negative. A syphilitic chancre also may present with the nontender genital ulceration7 that was seen in our patient, but serology did not support this diagnosis. Cutaneous Crohn disease also may manifest with genital ulceration even before a diagnosis of Crohn disease is made, but these lesions often present as linear knife-cut ulcerations of the anogenital region.8
Our case further supports a clinical presentation that diverges from the more traditional cases of MPXV. Additionally, associated EM may be a clue to infection, especially in cases of negative HHV and other sexually transmitted infection testing.
- Bunge EM, Hoet B, Chen L, et al. The changing epidemiology of human monkeypox—a potential threat? a systematic review. PLoS Negl Trop Dis. 2022;16:E0010141.
- Kumar N, Acharya A, Gendelman HE, et al. The 2022 outbreak and the pathobiology of the monkeypox virus. J Autoimmun. 2022;131:102855.
- Eisenstadt R, Liszewski WJ, Nguyen CV. Recognizing minimal cutaneous involvement or systemic symptoms in monkeypox. JAMA Dermatol. 2022;158:1457-1458.
- Català A, Clavo-Escribano P, Riera-Monroig J, et al. Monkeypox outbreak in Spain: clinical and epidemiological findings in a prospective cross-sectional study of 185 cases [published online August 2, 2022]. Br J Dermatol. 2022;187:765-772.
- Sokumbi O, Wetter DA. Clinical features, diagnosis, and treatment of erythema multiforme: a review for the practicing dermatologist. Int J Dermatol. 2012;51:889-902.
- Waleryie-Allanore L, Obeid G, Revuz J. Drug reactions. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:348-375.
- Stary G, Stary A. Sexually transmitted infections. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:1447-1469.
- Rosenbach MA, Wanat KA, Reisenauer A, et al. Non-infectious granulomas. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:1644-1663.
The Diagnosis: Mpox (Monkeypox)
Tests for active herpes simplex virus (HHV), gonorrhea, chlamydia, HIV, and syphilis were negative. Swabs from the penile lesion demonstrated positivity for the West African clade of mpox (monkeypox) virus (MPXV) by polymerase chain reaction. The patient was treated supportively without the addition of antiviral therapy, and he experienced a complete recovery.
Mpox virus was first isolated in 1958 in a research facility and was named after the laboratory animals that were housed there. The first human documentation of the disease occurred in 1970, and it was first documented in the United States in 2003 in an infection that was traced to a shipment of small mammals from Ghana to Texas.1 The disease has always been endemic to Africa; however, the incidence has been increasing.2 A new MPXV outbreak was reported in many countries in early 2022, including the United States.1
The MPXV is a double-stranded DNA virus of the genus Orthopoxvirus, and 2 genetic clades have been identified: clade I (formerly the Central African clade) and clade II (formerly the West African clade). The virus has the capability to infect many mammals; however, its host remains unidentified.1 The exact mechanism of transmission from infected animals to humans largely is unknown; however, direct or indirect contact with infected animals likely is responsible. Human-to-human transmission can occur by many mechanisms including contact with large respiratory droplets, bodily fluids, and contaminated surfaces. The incubation period is 5 to 21 days, and the symptoms last 2 to 5 weeks.1

The clinical manifestations of MPXV during the most recent outbreak differ from prior outbreaks. Patients are more likely to experience minimal to no systemic symptoms, and cutaneous lesions can be few and localized to a focal area, especially on the face and in the anogenital region,3 similar to the presentation in our patient (Figure 1). Cutaneous lesions of the most recent MPXV outbreak also include painless ulcerations similar to syphilitic chancres and lesions that are in various stages of healing.3 Lesions often begin as pseudopustules, which are firm white papules with or without a necrotic center that resemble pustules; unlike true pustules, there is no identifiable purulent material within it. Bacterial superinfection of the lesions is not uncommon.4 Over time, a secondary pustular eruption resembling folliculitis also may occur,4 as noted in our patient (Figure 2).

Although we did not have a biopsy to support the diagnosis of associated erythema multiforme (EM) in our patient, features supportive of this diagnosis included the classic clinical appearance of typical, well-defined, targetoid plaques with 3 distinct zones (Figure 3); the association with a known infection; the distribution on the arms with truncal sparing; and self-limited lesions. More than 90% of EM cases are associated with infection, with HHV representing the most common culprit5; therefore, the relationship with a different virus is not an unreasonable suggestion. Additionally, there have been rare reports of EM in association with MPXV.4

Histopathology of MPXV may have distinctive features. Lesions often demonstrate keratinocytic necrosis and basal layer vacuolization with an associated superficial and deep perivascular lymphohistiocytic infiltrate. When the morphology of the lesion is vesicular, histopathology reveals spongiosis and ballooning degeneration with epidermal necrosis. Viral inclusion bodies within keratinocytes may be identified.1 Death rates from MPXV has been reported from 1% to 11%, with increased mortality among high-risk populations including children and immunocompromised individuals. Treatment of the disease largely consists of supportive care and management of any associated complications including bacterial infection, pneumonia, and encephalitis.1
The differential diagnosis of MPXV includes other ulcerative lesions that can occur on the genital skin. Fixed drug eruptions often present on the penis,6 but there was no identifiable inciting drug in our patient. Herpes simplex virus infection was very high on the differential given our patient’s history of recurrent infections and association with a targetoid rash, but HHV type 1 and HHV type 2 testing of the lesion was negative. A syphilitic chancre also may present with the nontender genital ulceration7 that was seen in our patient, but serology did not support this diagnosis. Cutaneous Crohn disease also may manifest with genital ulceration even before a diagnosis of Crohn disease is made, but these lesions often present as linear knife-cut ulcerations of the anogenital region.8
Our case further supports a clinical presentation that diverges from the more traditional cases of MPXV. Additionally, associated EM may be a clue to infection, especially in cases of negative HHV and other sexually transmitted infection testing.
The Diagnosis: Mpox (Monkeypox)
Tests for active herpes simplex virus (HHV), gonorrhea, chlamydia, HIV, and syphilis were negative. Swabs from the penile lesion demonstrated positivity for the West African clade of mpox (monkeypox) virus (MPXV) by polymerase chain reaction. The patient was treated supportively without the addition of antiviral therapy, and he experienced a complete recovery.
Mpox virus was first isolated in 1958 in a research facility and was named after the laboratory animals that were housed there. The first human documentation of the disease occurred in 1970, and it was first documented in the United States in 2003 in an infection that was traced to a shipment of small mammals from Ghana to Texas.1 The disease has always been endemic to Africa; however, the incidence has been increasing.2 A new MPXV outbreak was reported in many countries in early 2022, including the United States.1
The MPXV is a double-stranded DNA virus of the genus Orthopoxvirus, and 2 genetic clades have been identified: clade I (formerly the Central African clade) and clade II (formerly the West African clade). The virus has the capability to infect many mammals; however, its host remains unidentified.1 The exact mechanism of transmission from infected animals to humans largely is unknown; however, direct or indirect contact with infected animals likely is responsible. Human-to-human transmission can occur by many mechanisms including contact with large respiratory droplets, bodily fluids, and contaminated surfaces. The incubation period is 5 to 21 days, and the symptoms last 2 to 5 weeks.1

The clinical manifestations of MPXV during the most recent outbreak differ from prior outbreaks. Patients are more likely to experience minimal to no systemic symptoms, and cutaneous lesions can be few and localized to a focal area, especially on the face and in the anogenital region,3 similar to the presentation in our patient (Figure 1). Cutaneous lesions of the most recent MPXV outbreak also include painless ulcerations similar to syphilitic chancres and lesions that are in various stages of healing.3 Lesions often begin as pseudopustules, which are firm white papules with or without a necrotic center that resemble pustules; unlike true pustules, there is no identifiable purulent material within it. Bacterial superinfection of the lesions is not uncommon.4 Over time, a secondary pustular eruption resembling folliculitis also may occur,4 as noted in our patient (Figure 2).

Although we did not have a biopsy to support the diagnosis of associated erythema multiforme (EM) in our patient, features supportive of this diagnosis included the classic clinical appearance of typical, well-defined, targetoid plaques with 3 distinct zones (Figure 3); the association with a known infection; the distribution on the arms with truncal sparing; and self-limited lesions. More than 90% of EM cases are associated with infection, with HHV representing the most common culprit5; therefore, the relationship with a different virus is not an unreasonable suggestion. Additionally, there have been rare reports of EM in association with MPXV.4

Histopathology of MPXV may have distinctive features. Lesions often demonstrate keratinocytic necrosis and basal layer vacuolization with an associated superficial and deep perivascular lymphohistiocytic infiltrate. When the morphology of the lesion is vesicular, histopathology reveals spongiosis and ballooning degeneration with epidermal necrosis. Viral inclusion bodies within keratinocytes may be identified.1 Death rates from MPXV has been reported from 1% to 11%, with increased mortality among high-risk populations including children and immunocompromised individuals. Treatment of the disease largely consists of supportive care and management of any associated complications including bacterial infection, pneumonia, and encephalitis.1
The differential diagnosis of MPXV includes other ulcerative lesions that can occur on the genital skin. Fixed drug eruptions often present on the penis,6 but there was no identifiable inciting drug in our patient. Herpes simplex virus infection was very high on the differential given our patient’s history of recurrent infections and association with a targetoid rash, but HHV type 1 and HHV type 2 testing of the lesion was negative. A syphilitic chancre also may present with the nontender genital ulceration7 that was seen in our patient, but serology did not support this diagnosis. Cutaneous Crohn disease also may manifest with genital ulceration even before a diagnosis of Crohn disease is made, but these lesions often present as linear knife-cut ulcerations of the anogenital region.8
Our case further supports a clinical presentation that diverges from the more traditional cases of MPXV. Additionally, associated EM may be a clue to infection, especially in cases of negative HHV and other sexually transmitted infection testing.
- Bunge EM, Hoet B, Chen L, et al. The changing epidemiology of human monkeypox—a potential threat? a systematic review. PLoS Negl Trop Dis. 2022;16:E0010141.
- Kumar N, Acharya A, Gendelman HE, et al. The 2022 outbreak and the pathobiology of the monkeypox virus. J Autoimmun. 2022;131:102855.
- Eisenstadt R, Liszewski WJ, Nguyen CV. Recognizing minimal cutaneous involvement or systemic symptoms in monkeypox. JAMA Dermatol. 2022;158:1457-1458.
- Català A, Clavo-Escribano P, Riera-Monroig J, et al. Monkeypox outbreak in Spain: clinical and epidemiological findings in a prospective cross-sectional study of 185 cases [published online August 2, 2022]. Br J Dermatol. 2022;187:765-772.
- Sokumbi O, Wetter DA. Clinical features, diagnosis, and treatment of erythema multiforme: a review for the practicing dermatologist. Int J Dermatol. 2012;51:889-902.
- Waleryie-Allanore L, Obeid G, Revuz J. Drug reactions. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:348-375.
- Stary G, Stary A. Sexually transmitted infections. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:1447-1469.
- Rosenbach MA, Wanat KA, Reisenauer A, et al. Non-infectious granulomas. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:1644-1663.
- Bunge EM, Hoet B, Chen L, et al. The changing epidemiology of human monkeypox—a potential threat? a systematic review. PLoS Negl Trop Dis. 2022;16:E0010141.
- Kumar N, Acharya A, Gendelman HE, et al. The 2022 outbreak and the pathobiology of the monkeypox virus. J Autoimmun. 2022;131:102855.
- Eisenstadt R, Liszewski WJ, Nguyen CV. Recognizing minimal cutaneous involvement or systemic symptoms in monkeypox. JAMA Dermatol. 2022;158:1457-1458.
- Català A, Clavo-Escribano P, Riera-Monroig J, et al. Monkeypox outbreak in Spain: clinical and epidemiological findings in a prospective cross-sectional study of 185 cases [published online August 2, 2022]. Br J Dermatol. 2022;187:765-772.
- Sokumbi O, Wetter DA. Clinical features, diagnosis, and treatment of erythema multiforme: a review for the practicing dermatologist. Int J Dermatol. 2012;51:889-902.
- Waleryie-Allanore L, Obeid G, Revuz J. Drug reactions. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:348-375.
- Stary G, Stary A. Sexually transmitted infections. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:1447-1469.
- Rosenbach MA, Wanat KA, Reisenauer A, et al. Non-infectious granulomas. In: Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. Elsevier; 2018:1644-1663.
A 50-year-old man with a history of recurrent genital herpes simplex virus infections presented to the hospital with genital lesions and swelling of 5 days’ duration. Prior to admission, the patient was treated with a course of valacyclovir by an urgent care physician without improvement. Physical examination revealed a 3-cm, nontender, shallow, ulcerative plaque with irregular borders and a purulent yellow base distributed on the distal shaft of the penis with extension into the coronal sulcus. A few other scattered erosions were noted on the distal penile shaft. He had associated diffuse nonpitting edema of the penis and scrotum as well as tender bilateral inguinal lymphadenopathy. Three days after the genital ulcerations began, the patient developed a nontender erythematous papule with a necrotic center on the right jaw followed by an eruption of erythematous papulopustules on the arms and trunk. The patient denied dysuria, purulent penile discharge, fevers, chills, headaches, myalgia, arthralgia, nausea, vomiting, or diarrhea. The patient was sexually active exclusively with females and had more than 10 partners in the prior year. Shortly after hospital admission, the patient developed red targetoid plaques on the groin, trunk, and arms. No oral mucosal lesions were identified.
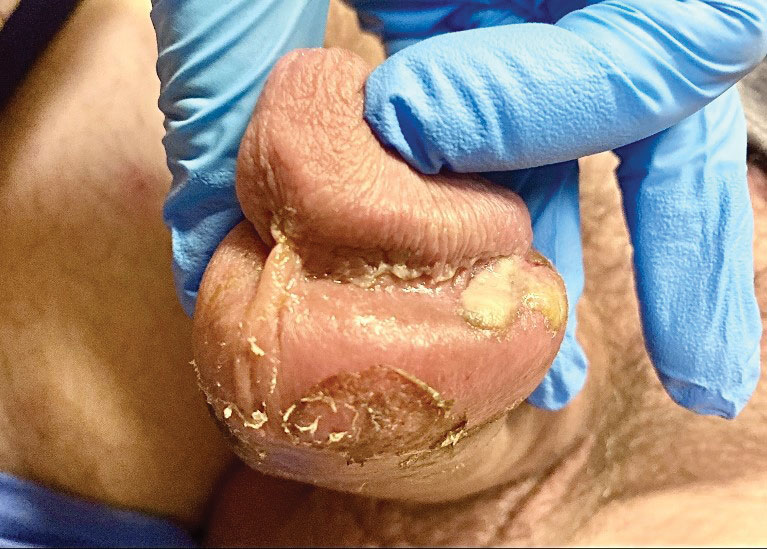
Oral IL-23 receptor antagonist for psoriasis promising: Phase 2b study
SINGAPORE – across all doses, compared with placebo, according to results of the FRONTIER 1 trial.
In the 16-week phase 2b study, 255 adults with moderate to severe plaque psoriasis were randomly assigned into six treatment groups: placebo (n = 43), JNJ-2113 25 mg daily (n = 43), 25 mg twice daily (n = 41), 50 mg daily (n = 43), 100 mg daily (n = 43), or 100 mg twice daily (n = 42).
Of those who took the placebo, only 9.3% achieved the study’s primary endpoint of a 75% or greater improvement in the Psoriasis Area and Severity Index (PASI-75) by week 16. This was compared with 78.6% in the group that took the highest dose.
“Additionally, the onset of action was fairly fast: at week 4, more than 20% of patients had achieved PASI 75,” said Robert Bissonnette, MD, CEO of Innovaderm Research in Montreal, who presented the findings during a late-breaker session at the World Congress of Dermatology.
Patients in the remaining groups demonstrated a response that corresponded to dosing level: with 37.2%, 51.2%, 58.1%, and 65.1% achieving PASI-75 in the 25 mg daily, 25 mg twice-daily, 50 mg daily, and 100 mg daily groups, respectively.
“These results are very interesting because in terms of psoriasis treatment, if this is confirmed in phase 3, it would give us an oral alternative that would be selective for IL-23,” said Dr. Bissonnette, referring to the signaling pathway that plays a critical role in the pathogenesis of several immune-mediated inflammatory diseases, including plaque psoriasis.
Although rarely life-threatening, the skin disorder is often intractable to treatment. In recent years, therapies that block IL-23 signaling and downstream inflammatory cytokine production have proven useful. “We have on the market a number of biological agents targeting IL-23 that we use on a regular basis,” said Dr. Bissonnette. “However, there are currently no orally delivered therapies.”
If successful, JNJ-2113 – a first-in-class oral IL-23 antagonist peptide developed by Janssen – could change the treatment paradigm for patients with moderate to severe plaque psoriasis. “When I was first introduced to the concept, I thought it wouldn’t work as it’s a peptide, that it would be digested by the stomach,” he told the audience. “But because of its GI stability and its potency, when you administer it orally, you can detect pharmacological activity.”
A well-tolerated alternative
Participants in the FRONTIER 1 trial were on average about 44 years old and weighed 88.9 kg (195 lb). Most had been living with psoriasis for about 18 years, with a total PASI score of 19.05. In addition, 43.1% had been treated with phototherapy in the past, 22% with biologics, and 78.4% with systemics.
PASI 90 and 100 were among some of the secondary outcomes measured. Similar to the primary outcome of PASI 75, all treatment groups demonstrated a statistically significant dose-response in PASI 90, compared with placebo. For those on the highest dose of JNJ-2113, 59.5% and 40.5% achieved PASI 90 and PASI 100, respectively, by week 16. The corresponding figures for those receiving placebo were 2.3% and 0%.
The safety profile for JNJ-2113 across all doses was similar to that of placebo, with no evidence of a dose-dependent increase in the occurrence of adverse events (AEs). The most frequently reported AEs were COVID-19 and nasopharyngitis. There were three serious AEs (COVID-19, infected cyst, suicide attempt) among those on the active drug, but the investigators assessed that they were not related to the study intervention. No deaths, major adverse cardiac events, or malignancies were reported during the study.
Approached for an independent comment, Marius-Anton Ionescu, MD, PhD, from the University Hospital Saint Louis, Paris, who specializes in psoriasis, told this news organization that the new development with JNJ-2113 “is really promising.”
Treatment for plaque psoriasis has improved to the point where some biologics, such as risankizumab (Skyrizi), only require patients to have “four shots a year,” he says. “This is the future of psoriasis treatment; it might go down to two shots a year” – a regimen that will be easier than taking an oral medication once or twice a day.
“But it’s good to have an oral option because you will always have some patients who say: ‘Shots are not for me, I’m afraid,’ ” he says.
However, Dr. Ionescu noted that if JNJ-2113 were to pass phase 3 trials, it might face stiff competition from the selective tyrosine kinase 2 (TYK2) inhibitor deucravacitinib (Sotyktu), which the U.S. Food and Drug Administration approved for use in adults with moderate to severe plaque psoriasis last September. “It has very good results and is the first oral therapy that is comparable with biologics for plaque psoriasis,” he says.
But Dr. Bissonnette remains hopeful for the future. “I think JNJ-2113 goes way beyond psoriasis because this type of strategy using oral peptide–blocking receptors could be used in other immune-mediated diseases, including atopic dermatitis and other diseases outside of dermatology.” In addition to running a phase 3 study for moderate to severe plaque psoriasis, Janssen is planning to initiate a phase 2b clinical trial of JNJ-2113 in adults with ulcerative colitis.
The study was funded by Janssen. Dr. Bissonnette reports consulting and investigating for Janssen, and being on advisory panels and receiving research funding from multiple other pharmaceutical companies. Dr. Ionescu is an investigator for Psoriasis National Register France Psobioteq (no honoraria), and an investigator and speaker for Uriage cosmetics (honoraria).
A version of this article first appeared on Medscape.com.
SINGAPORE – across all doses, compared with placebo, according to results of the FRONTIER 1 trial.
In the 16-week phase 2b study, 255 adults with moderate to severe plaque psoriasis were randomly assigned into six treatment groups: placebo (n = 43), JNJ-2113 25 mg daily (n = 43), 25 mg twice daily (n = 41), 50 mg daily (n = 43), 100 mg daily (n = 43), or 100 mg twice daily (n = 42).
Of those who took the placebo, only 9.3% achieved the study’s primary endpoint of a 75% or greater improvement in the Psoriasis Area and Severity Index (PASI-75) by week 16. This was compared with 78.6% in the group that took the highest dose.
“Additionally, the onset of action was fairly fast: at week 4, more than 20% of patients had achieved PASI 75,” said Robert Bissonnette, MD, CEO of Innovaderm Research in Montreal, who presented the findings during a late-breaker session at the World Congress of Dermatology.
Patients in the remaining groups demonstrated a response that corresponded to dosing level: with 37.2%, 51.2%, 58.1%, and 65.1% achieving PASI-75 in the 25 mg daily, 25 mg twice-daily, 50 mg daily, and 100 mg daily groups, respectively.
“These results are very interesting because in terms of psoriasis treatment, if this is confirmed in phase 3, it would give us an oral alternative that would be selective for IL-23,” said Dr. Bissonnette, referring to the signaling pathway that plays a critical role in the pathogenesis of several immune-mediated inflammatory diseases, including plaque psoriasis.
Although rarely life-threatening, the skin disorder is often intractable to treatment. In recent years, therapies that block IL-23 signaling and downstream inflammatory cytokine production have proven useful. “We have on the market a number of biological agents targeting IL-23 that we use on a regular basis,” said Dr. Bissonnette. “However, there are currently no orally delivered therapies.”
If successful, JNJ-2113 – a first-in-class oral IL-23 antagonist peptide developed by Janssen – could change the treatment paradigm for patients with moderate to severe plaque psoriasis. “When I was first introduced to the concept, I thought it wouldn’t work as it’s a peptide, that it would be digested by the stomach,” he told the audience. “But because of its GI stability and its potency, when you administer it orally, you can detect pharmacological activity.”
A well-tolerated alternative
Participants in the FRONTIER 1 trial were on average about 44 years old and weighed 88.9 kg (195 lb). Most had been living with psoriasis for about 18 years, with a total PASI score of 19.05. In addition, 43.1% had been treated with phototherapy in the past, 22% with biologics, and 78.4% with systemics.
PASI 90 and 100 were among some of the secondary outcomes measured. Similar to the primary outcome of PASI 75, all treatment groups demonstrated a statistically significant dose-response in PASI 90, compared with placebo. For those on the highest dose of JNJ-2113, 59.5% and 40.5% achieved PASI 90 and PASI 100, respectively, by week 16. The corresponding figures for those receiving placebo were 2.3% and 0%.
The safety profile for JNJ-2113 across all doses was similar to that of placebo, with no evidence of a dose-dependent increase in the occurrence of adverse events (AEs). The most frequently reported AEs were COVID-19 and nasopharyngitis. There were three serious AEs (COVID-19, infected cyst, suicide attempt) among those on the active drug, but the investigators assessed that they were not related to the study intervention. No deaths, major adverse cardiac events, or malignancies were reported during the study.
Approached for an independent comment, Marius-Anton Ionescu, MD, PhD, from the University Hospital Saint Louis, Paris, who specializes in psoriasis, told this news organization that the new development with JNJ-2113 “is really promising.”
Treatment for plaque psoriasis has improved to the point where some biologics, such as risankizumab (Skyrizi), only require patients to have “four shots a year,” he says. “This is the future of psoriasis treatment; it might go down to two shots a year” – a regimen that will be easier than taking an oral medication once or twice a day.
“But it’s good to have an oral option because you will always have some patients who say: ‘Shots are not for me, I’m afraid,’ ” he says.
However, Dr. Ionescu noted that if JNJ-2113 were to pass phase 3 trials, it might face stiff competition from the selective tyrosine kinase 2 (TYK2) inhibitor deucravacitinib (Sotyktu), which the U.S. Food and Drug Administration approved for use in adults with moderate to severe plaque psoriasis last September. “It has very good results and is the first oral therapy that is comparable with biologics for plaque psoriasis,” he says.
But Dr. Bissonnette remains hopeful for the future. “I think JNJ-2113 goes way beyond psoriasis because this type of strategy using oral peptide–blocking receptors could be used in other immune-mediated diseases, including atopic dermatitis and other diseases outside of dermatology.” In addition to running a phase 3 study for moderate to severe plaque psoriasis, Janssen is planning to initiate a phase 2b clinical trial of JNJ-2113 in adults with ulcerative colitis.
The study was funded by Janssen. Dr. Bissonnette reports consulting and investigating for Janssen, and being on advisory panels and receiving research funding from multiple other pharmaceutical companies. Dr. Ionescu is an investigator for Psoriasis National Register France Psobioteq (no honoraria), and an investigator and speaker for Uriage cosmetics (honoraria).
A version of this article first appeared on Medscape.com.
SINGAPORE – across all doses, compared with placebo, according to results of the FRONTIER 1 trial.
In the 16-week phase 2b study, 255 adults with moderate to severe plaque psoriasis were randomly assigned into six treatment groups: placebo (n = 43), JNJ-2113 25 mg daily (n = 43), 25 mg twice daily (n = 41), 50 mg daily (n = 43), 100 mg daily (n = 43), or 100 mg twice daily (n = 42).
Of those who took the placebo, only 9.3% achieved the study’s primary endpoint of a 75% or greater improvement in the Psoriasis Area and Severity Index (PASI-75) by week 16. This was compared with 78.6% in the group that took the highest dose.
“Additionally, the onset of action was fairly fast: at week 4, more than 20% of patients had achieved PASI 75,” said Robert Bissonnette, MD, CEO of Innovaderm Research in Montreal, who presented the findings during a late-breaker session at the World Congress of Dermatology.
Patients in the remaining groups demonstrated a response that corresponded to dosing level: with 37.2%, 51.2%, 58.1%, and 65.1% achieving PASI-75 in the 25 mg daily, 25 mg twice-daily, 50 mg daily, and 100 mg daily groups, respectively.
“These results are very interesting because in terms of psoriasis treatment, if this is confirmed in phase 3, it would give us an oral alternative that would be selective for IL-23,” said Dr. Bissonnette, referring to the signaling pathway that plays a critical role in the pathogenesis of several immune-mediated inflammatory diseases, including plaque psoriasis.
Although rarely life-threatening, the skin disorder is often intractable to treatment. In recent years, therapies that block IL-23 signaling and downstream inflammatory cytokine production have proven useful. “We have on the market a number of biological agents targeting IL-23 that we use on a regular basis,” said Dr. Bissonnette. “However, there are currently no orally delivered therapies.”
If successful, JNJ-2113 – a first-in-class oral IL-23 antagonist peptide developed by Janssen – could change the treatment paradigm for patients with moderate to severe plaque psoriasis. “When I was first introduced to the concept, I thought it wouldn’t work as it’s a peptide, that it would be digested by the stomach,” he told the audience. “But because of its GI stability and its potency, when you administer it orally, you can detect pharmacological activity.”
A well-tolerated alternative
Participants in the FRONTIER 1 trial were on average about 44 years old and weighed 88.9 kg (195 lb). Most had been living with psoriasis for about 18 years, with a total PASI score of 19.05. In addition, 43.1% had been treated with phototherapy in the past, 22% with biologics, and 78.4% with systemics.
PASI 90 and 100 were among some of the secondary outcomes measured. Similar to the primary outcome of PASI 75, all treatment groups demonstrated a statistically significant dose-response in PASI 90, compared with placebo. For those on the highest dose of JNJ-2113, 59.5% and 40.5% achieved PASI 90 and PASI 100, respectively, by week 16. The corresponding figures for those receiving placebo were 2.3% and 0%.
The safety profile for JNJ-2113 across all doses was similar to that of placebo, with no evidence of a dose-dependent increase in the occurrence of adverse events (AEs). The most frequently reported AEs were COVID-19 and nasopharyngitis. There were three serious AEs (COVID-19, infected cyst, suicide attempt) among those on the active drug, but the investigators assessed that they were not related to the study intervention. No deaths, major adverse cardiac events, or malignancies were reported during the study.
Approached for an independent comment, Marius-Anton Ionescu, MD, PhD, from the University Hospital Saint Louis, Paris, who specializes in psoriasis, told this news organization that the new development with JNJ-2113 “is really promising.”
Treatment for plaque psoriasis has improved to the point where some biologics, such as risankizumab (Skyrizi), only require patients to have “four shots a year,” he says. “This is the future of psoriasis treatment; it might go down to two shots a year” – a regimen that will be easier than taking an oral medication once or twice a day.
“But it’s good to have an oral option because you will always have some patients who say: ‘Shots are not for me, I’m afraid,’ ” he says.
However, Dr. Ionescu noted that if JNJ-2113 were to pass phase 3 trials, it might face stiff competition from the selective tyrosine kinase 2 (TYK2) inhibitor deucravacitinib (Sotyktu), which the U.S. Food and Drug Administration approved for use in adults with moderate to severe plaque psoriasis last September. “It has very good results and is the first oral therapy that is comparable with biologics for plaque psoriasis,” he says.
But Dr. Bissonnette remains hopeful for the future. “I think JNJ-2113 goes way beyond psoriasis because this type of strategy using oral peptide–blocking receptors could be used in other immune-mediated diseases, including atopic dermatitis and other diseases outside of dermatology.” In addition to running a phase 3 study for moderate to severe plaque psoriasis, Janssen is planning to initiate a phase 2b clinical trial of JNJ-2113 in adults with ulcerative colitis.
The study was funded by Janssen. Dr. Bissonnette reports consulting and investigating for Janssen, and being on advisory panels and receiving research funding from multiple other pharmaceutical companies. Dr. Ionescu is an investigator for Psoriasis National Register France Psobioteq (no honoraria), and an investigator and speaker for Uriage cosmetics (honoraria).
A version of this article first appeared on Medscape.com.
AT WCD 2023