User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Skinny-label biosimilars provide substantial savings to Medicare
Recent court rulings could put such saving under threat
Competition between five biologic drugs and their skinny-label biosimilars saved Medicare an estimated $1.5 billion during 2015-2020. But these savings accruing to Medicare and the availability of those and other biosimilars through skinny labeling is under threat from recent court rulings, according to a research letter published online in JAMA Internal Medicine.
The authors highlighted the need for such savings by noting that, while biologics comprise less than 5% of prescription drug use, their price tag amounts to about 40% of U.S. drug spending, Biologic manufacturers often delay the availability of biosimilars for additional years beyond the original patent expiration through further patents for supplemental indications. To provide a counterbalance, federal law allows the Food and Drug Administration to approve “skinny-label” generics and biosimilars that carve out patent-protected indications or regulatory exclusivities. But once a generic drug reaches the market through this process with a skinny label, it may often be substituted for indications that go beyond the ones listed on the skinny label. In fact, some state laws mandate that pharmacists substitute interchangeable generics for brand-name drugs, helping to decrease drug prices. In response to legal threats to the skinny-label pathway, Alexander C. Egilman and colleagues at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, assessed the frequency of approval and marketing of skinny-label biosimilars from 2015 to 2021 and the resultant savings to Medicare.
The authors estimated annual Part B (clinician-administered) savings from skinny-label biosimilars through 2020 by comparing actual biologic and skinny-label biosimilar spending with estimated biologic spending without competition using the Medicare Dashboard. They assumed that the unit price of the biologic would increase at its 5-year compound annual growth rate prior to competition.
In that period, the FDA approved 33 biosimilars linked to 11 biologics. Among them, 22 (66.7%) had a skinny label. Of 21 biosimilars marketed before 2022, 13 (61.9%) were launched with a skinny label. Of the 8 biologics linked to these 21 biosimilars, 5 of the first-to-market biosimilars had skinny labels (bevacizumab, filgrastim, infliximab, pegfilgrastim, and rituximab), leading to earlier competition through 2021.
The estimated $1.5 billion in savings to Medicare from these skinny-label biosimilars over the 2015-2020 span represents 4.9% of the $30.2 billion that Medicare spent on the five biologics during this period. The researchers pointed out that once adalimumab (Humira) faces skinny-label biosimilar competition in 2023, savings will likely grow substantially.
In response to the research letter, an editor’s note by JAMA Internal Medicine Editorial Fellow Eric Ward, MD, and JAMA Internal Medicine Editor at Large and Online Editor Robert Steinbrook, MD, stated that, between 2015 and 2019, 24 (43%) of 56 brand-name drugs had competition from skinny-labeled generic formulations after first becoming available as generics.
The editors also referenced a JAMA Viewpoints article from 2021 that reviewed the most recent case challenging the skinny-label pathway in which GlaxoSmithKline sued Teva for its marketing of a skinny-label generic of the brand-name beta-blocker carvedilol (Coreg) that the plaintive claimed “induced physicians to prescribe carvedilol for indications that had been carved out by Teva’s skinny label, thus infringing GlaxoSmithKline’s patents.” A $235 million judgment against Teva was overturned by a district court and then reversed again by a Federal Circuit court that, after receiving criticism, reconsidered the case, and a panel affirmed the judgment against Teva.
“The Federal Circuit panel’s decision has the potential to put generic drugs that fail to adequately carve out indications from the brand name labeling at risk for damages related to infringement,” the authors wrote. Similar claims of infringement are being heard in other courts, they wrote, and they urged careful targeting of skinny-label carveouts, and suggest also that challenges to the arguments used against Teva focus on preservation of First Amendment rights as protection for lawful and accurate speech in drug labels.
“The legal uncertainties are likely to continue, as manufacturers pursue novel and complex strategies to protect the patents and regulatory exclusivities of brand-name drugs and biologics,” Dr. Ward and Dr. Steinbrook wrote, adding that “the path forward is for Congress to enact additional legislation that reaffirms and strengthens the permissibility of skinny labeling.”
The research letter’s corresponding author, Ameet Sarpatwari, PhD, JD, assistant professor at Harvard Medical School, and assistant director for the Harvard Program On Regulation, Therapeutics, And Law, echoed concerns over the Teva case in an interview. “There has certainly been concern that should the appellate decision stand, there will be a chilling effect. As the lone dissenter in that case noted, ‘no skinny-label generic is safe.’ I think many generic and biosimilar manufacturers are awaiting to see whether the Supreme Court will take the case.”
He added: “I do not believe the likelihood of skinny-label-supportive legislation making it through Congress will be greatly diminished in a divided Congress. Democrats and Republicans alike should seek to promote competition in the marketplace, which is what the skinny-labeling pathway accomplishes.”
The authors reported no relevant conflicts of interest. The research was funded by a grant from Arnold Ventures.
Recent court rulings could put such saving under threat
Recent court rulings could put such saving under threat
Competition between five biologic drugs and their skinny-label biosimilars saved Medicare an estimated $1.5 billion during 2015-2020. But these savings accruing to Medicare and the availability of those and other biosimilars through skinny labeling is under threat from recent court rulings, according to a research letter published online in JAMA Internal Medicine.
The authors highlighted the need for such savings by noting that, while biologics comprise less than 5% of prescription drug use, their price tag amounts to about 40% of U.S. drug spending, Biologic manufacturers often delay the availability of biosimilars for additional years beyond the original patent expiration through further patents for supplemental indications. To provide a counterbalance, federal law allows the Food and Drug Administration to approve “skinny-label” generics and biosimilars that carve out patent-protected indications or regulatory exclusivities. But once a generic drug reaches the market through this process with a skinny label, it may often be substituted for indications that go beyond the ones listed on the skinny label. In fact, some state laws mandate that pharmacists substitute interchangeable generics for brand-name drugs, helping to decrease drug prices. In response to legal threats to the skinny-label pathway, Alexander C. Egilman and colleagues at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, assessed the frequency of approval and marketing of skinny-label biosimilars from 2015 to 2021 and the resultant savings to Medicare.
The authors estimated annual Part B (clinician-administered) savings from skinny-label biosimilars through 2020 by comparing actual biologic and skinny-label biosimilar spending with estimated biologic spending without competition using the Medicare Dashboard. They assumed that the unit price of the biologic would increase at its 5-year compound annual growth rate prior to competition.
In that period, the FDA approved 33 biosimilars linked to 11 biologics. Among them, 22 (66.7%) had a skinny label. Of 21 biosimilars marketed before 2022, 13 (61.9%) were launched with a skinny label. Of the 8 biologics linked to these 21 biosimilars, 5 of the first-to-market biosimilars had skinny labels (bevacizumab, filgrastim, infliximab, pegfilgrastim, and rituximab), leading to earlier competition through 2021.
The estimated $1.5 billion in savings to Medicare from these skinny-label biosimilars over the 2015-2020 span represents 4.9% of the $30.2 billion that Medicare spent on the five biologics during this period. The researchers pointed out that once adalimumab (Humira) faces skinny-label biosimilar competition in 2023, savings will likely grow substantially.
In response to the research letter, an editor’s note by JAMA Internal Medicine Editorial Fellow Eric Ward, MD, and JAMA Internal Medicine Editor at Large and Online Editor Robert Steinbrook, MD, stated that, between 2015 and 2019, 24 (43%) of 56 brand-name drugs had competition from skinny-labeled generic formulations after first becoming available as generics.
The editors also referenced a JAMA Viewpoints article from 2021 that reviewed the most recent case challenging the skinny-label pathway in which GlaxoSmithKline sued Teva for its marketing of a skinny-label generic of the brand-name beta-blocker carvedilol (Coreg) that the plaintive claimed “induced physicians to prescribe carvedilol for indications that had been carved out by Teva’s skinny label, thus infringing GlaxoSmithKline’s patents.” A $235 million judgment against Teva was overturned by a district court and then reversed again by a Federal Circuit court that, after receiving criticism, reconsidered the case, and a panel affirmed the judgment against Teva.
“The Federal Circuit panel’s decision has the potential to put generic drugs that fail to adequately carve out indications from the brand name labeling at risk for damages related to infringement,” the authors wrote. Similar claims of infringement are being heard in other courts, they wrote, and they urged careful targeting of skinny-label carveouts, and suggest also that challenges to the arguments used against Teva focus on preservation of First Amendment rights as protection for lawful and accurate speech in drug labels.
“The legal uncertainties are likely to continue, as manufacturers pursue novel and complex strategies to protect the patents and regulatory exclusivities of brand-name drugs and biologics,” Dr. Ward and Dr. Steinbrook wrote, adding that “the path forward is for Congress to enact additional legislation that reaffirms and strengthens the permissibility of skinny labeling.”
The research letter’s corresponding author, Ameet Sarpatwari, PhD, JD, assistant professor at Harvard Medical School, and assistant director for the Harvard Program On Regulation, Therapeutics, And Law, echoed concerns over the Teva case in an interview. “There has certainly been concern that should the appellate decision stand, there will be a chilling effect. As the lone dissenter in that case noted, ‘no skinny-label generic is safe.’ I think many generic and biosimilar manufacturers are awaiting to see whether the Supreme Court will take the case.”
He added: “I do not believe the likelihood of skinny-label-supportive legislation making it through Congress will be greatly diminished in a divided Congress. Democrats and Republicans alike should seek to promote competition in the marketplace, which is what the skinny-labeling pathway accomplishes.”
The authors reported no relevant conflicts of interest. The research was funded by a grant from Arnold Ventures.
Competition between five biologic drugs and their skinny-label biosimilars saved Medicare an estimated $1.5 billion during 2015-2020. But these savings accruing to Medicare and the availability of those and other biosimilars through skinny labeling is under threat from recent court rulings, according to a research letter published online in JAMA Internal Medicine.
The authors highlighted the need for such savings by noting that, while biologics comprise less than 5% of prescription drug use, their price tag amounts to about 40% of U.S. drug spending, Biologic manufacturers often delay the availability of biosimilars for additional years beyond the original patent expiration through further patents for supplemental indications. To provide a counterbalance, federal law allows the Food and Drug Administration to approve “skinny-label” generics and biosimilars that carve out patent-protected indications or regulatory exclusivities. But once a generic drug reaches the market through this process with a skinny label, it may often be substituted for indications that go beyond the ones listed on the skinny label. In fact, some state laws mandate that pharmacists substitute interchangeable generics for brand-name drugs, helping to decrease drug prices. In response to legal threats to the skinny-label pathway, Alexander C. Egilman and colleagues at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, assessed the frequency of approval and marketing of skinny-label biosimilars from 2015 to 2021 and the resultant savings to Medicare.
The authors estimated annual Part B (clinician-administered) savings from skinny-label biosimilars through 2020 by comparing actual biologic and skinny-label biosimilar spending with estimated biologic spending without competition using the Medicare Dashboard. They assumed that the unit price of the biologic would increase at its 5-year compound annual growth rate prior to competition.
In that period, the FDA approved 33 biosimilars linked to 11 biologics. Among them, 22 (66.7%) had a skinny label. Of 21 biosimilars marketed before 2022, 13 (61.9%) were launched with a skinny label. Of the 8 biologics linked to these 21 biosimilars, 5 of the first-to-market biosimilars had skinny labels (bevacizumab, filgrastim, infliximab, pegfilgrastim, and rituximab), leading to earlier competition through 2021.
The estimated $1.5 billion in savings to Medicare from these skinny-label biosimilars over the 2015-2020 span represents 4.9% of the $30.2 billion that Medicare spent on the five biologics during this period. The researchers pointed out that once adalimumab (Humira) faces skinny-label biosimilar competition in 2023, savings will likely grow substantially.
In response to the research letter, an editor’s note by JAMA Internal Medicine Editorial Fellow Eric Ward, MD, and JAMA Internal Medicine Editor at Large and Online Editor Robert Steinbrook, MD, stated that, between 2015 and 2019, 24 (43%) of 56 brand-name drugs had competition from skinny-labeled generic formulations after first becoming available as generics.
The editors also referenced a JAMA Viewpoints article from 2021 that reviewed the most recent case challenging the skinny-label pathway in which GlaxoSmithKline sued Teva for its marketing of a skinny-label generic of the brand-name beta-blocker carvedilol (Coreg) that the plaintive claimed “induced physicians to prescribe carvedilol for indications that had been carved out by Teva’s skinny label, thus infringing GlaxoSmithKline’s patents.” A $235 million judgment against Teva was overturned by a district court and then reversed again by a Federal Circuit court that, after receiving criticism, reconsidered the case, and a panel affirmed the judgment against Teva.
“The Federal Circuit panel’s decision has the potential to put generic drugs that fail to adequately carve out indications from the brand name labeling at risk for damages related to infringement,” the authors wrote. Similar claims of infringement are being heard in other courts, they wrote, and they urged careful targeting of skinny-label carveouts, and suggest also that challenges to the arguments used against Teva focus on preservation of First Amendment rights as protection for lawful and accurate speech in drug labels.
“The legal uncertainties are likely to continue, as manufacturers pursue novel and complex strategies to protect the patents and regulatory exclusivities of brand-name drugs and biologics,” Dr. Ward and Dr. Steinbrook wrote, adding that “the path forward is for Congress to enact additional legislation that reaffirms and strengthens the permissibility of skinny labeling.”
The research letter’s corresponding author, Ameet Sarpatwari, PhD, JD, assistant professor at Harvard Medical School, and assistant director for the Harvard Program On Regulation, Therapeutics, And Law, echoed concerns over the Teva case in an interview. “There has certainly been concern that should the appellate decision stand, there will be a chilling effect. As the lone dissenter in that case noted, ‘no skinny-label generic is safe.’ I think many generic and biosimilar manufacturers are awaiting to see whether the Supreme Court will take the case.”
He added: “I do not believe the likelihood of skinny-label-supportive legislation making it through Congress will be greatly diminished in a divided Congress. Democrats and Republicans alike should seek to promote competition in the marketplace, which is what the skinny-labeling pathway accomplishes.”
The authors reported no relevant conflicts of interest. The research was funded by a grant from Arnold Ventures.
FROM JAMA INTERNAL MEDICINE
Therapeutic Considerations in Adults With Hidradenitis Suppurativa
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition characterized by recurrent boils, abscesses, and nodules that can progress to narrow channels that form under the skin. An estimated 1%-4% of the US population has the condition, and women are affected more commonly than men.
Treatment of HS is challenging and the pathogenesis is still under investigation. Many believe that the disease involves follicular occlusion that leads to perifollicular cyst development followed by ruptures of the cyst contents. Many drug classes, including antibiotics and topical therapies, as well as lifestyle modifications, have been used to successfully treat mild to moderate HS. Management of moderate to severe HS has been less successful, however.
Dr Jennifer Hsiao, from the University of Southern California, highlights the various approaches to HS treatment, including medical, procedural, and emerging options.
--
Jennifer Hsiao, MD, Associate Professor, Physician, Department of Dermatology, University of Southern California, Los Angeles, California
Jennifer Hsiao, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Novartis; UCB
Serve(d) as a speaker or a member of a speakers bureau for: AbbVie
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition characterized by recurrent boils, abscesses, and nodules that can progress to narrow channels that form under the skin. An estimated 1%-4% of the US population has the condition, and women are affected more commonly than men.
Treatment of HS is challenging and the pathogenesis is still under investigation. Many believe that the disease involves follicular occlusion that leads to perifollicular cyst development followed by ruptures of the cyst contents. Many drug classes, including antibiotics and topical therapies, as well as lifestyle modifications, have been used to successfully treat mild to moderate HS. Management of moderate to severe HS has been less successful, however.
Dr Jennifer Hsiao, from the University of Southern California, highlights the various approaches to HS treatment, including medical, procedural, and emerging options.
--
Jennifer Hsiao, MD, Associate Professor, Physician, Department of Dermatology, University of Southern California, Los Angeles, California
Jennifer Hsiao, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Novartis; UCB
Serve(d) as a speaker or a member of a speakers bureau for: AbbVie
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition characterized by recurrent boils, abscesses, and nodules that can progress to narrow channels that form under the skin. An estimated 1%-4% of the US population has the condition, and women are affected more commonly than men.
Treatment of HS is challenging and the pathogenesis is still under investigation. Many believe that the disease involves follicular occlusion that leads to perifollicular cyst development followed by ruptures of the cyst contents. Many drug classes, including antibiotics and topical therapies, as well as lifestyle modifications, have been used to successfully treat mild to moderate HS. Management of moderate to severe HS has been less successful, however.
Dr Jennifer Hsiao, from the University of Southern California, highlights the various approaches to HS treatment, including medical, procedural, and emerging options.
--
Jennifer Hsiao, MD, Associate Professor, Physician, Department of Dermatology, University of Southern California, Los Angeles, California
Jennifer Hsiao, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AbbVie; Novartis; UCB
Serve(d) as a speaker or a member of a speakers bureau for: AbbVie
The right indoor relative humidity could ward off COVID
The “sweet spot” associated with reduced COVID-19 cases and deaths is 40%-60% indoor relative humidity, an MIT news release said. People who maintained indoor relative humidity outside those parameters had higher rates of catching COVID-19.
Most people are comfortable with 30%-50% relative humidity, researchers said. An airplane cabin has about 20% relative humidity.
Relative humidity is the amount of moisture in the air, compared with the total moisture the air can hold at a given temperature before saturating and forming condensation.
The study was published in The Journal of the Royal Society Interface. Researchers examined COVID-19 data and meteorological measurements from 121 countries from January 2020 through August 2020, before vaccines became available to the public.
“When outdoor temperatures were below the typical human comfort range, they assumed indoor spaces were heated to reach that comfort range. Based on the added heating, they calculated the associated drop in indoor relative humidity,” the MIT news release said.
The research teams found that when a region reported a rise in COVID-19 cases and deaths, the region’s estimated indoor relative humidity was either lower than 40% or higher than 60%, the release said.
“There’s potentially a protective effect of this intermediate indoor relative humidity,” said Connor Verheyen, the lead author and a PhD student in medical engineering and medical physics in the Harvard-MIT Program in Health Sciences and Technology.
Widespread use of the 40%-60% indoor humidity range could reduce the need for lockdowns and other widespread restrictions, the study concluded.
“Unlike measures that depend on individual compliance (for example, masking or hand-washing), indoor RH optimization would achieve high compliance because all occupants of a common indoor space would be exposed to similar ambient conditions,” the study said. “Compared to the long timelines and high costs of vaccine production and distribution, humidity control systems could potentially be implemented more quickly and cheaply in certain indoor settings.”
A version of this article first appeared on WebMD.com.
The “sweet spot” associated with reduced COVID-19 cases and deaths is 40%-60% indoor relative humidity, an MIT news release said. People who maintained indoor relative humidity outside those parameters had higher rates of catching COVID-19.
Most people are comfortable with 30%-50% relative humidity, researchers said. An airplane cabin has about 20% relative humidity.
Relative humidity is the amount of moisture in the air, compared with the total moisture the air can hold at a given temperature before saturating and forming condensation.
The study was published in The Journal of the Royal Society Interface. Researchers examined COVID-19 data and meteorological measurements from 121 countries from January 2020 through August 2020, before vaccines became available to the public.
“When outdoor temperatures were below the typical human comfort range, they assumed indoor spaces were heated to reach that comfort range. Based on the added heating, they calculated the associated drop in indoor relative humidity,” the MIT news release said.
The research teams found that when a region reported a rise in COVID-19 cases and deaths, the region’s estimated indoor relative humidity was either lower than 40% or higher than 60%, the release said.
“There’s potentially a protective effect of this intermediate indoor relative humidity,” said Connor Verheyen, the lead author and a PhD student in medical engineering and medical physics in the Harvard-MIT Program in Health Sciences and Technology.
Widespread use of the 40%-60% indoor humidity range could reduce the need for lockdowns and other widespread restrictions, the study concluded.
“Unlike measures that depend on individual compliance (for example, masking or hand-washing), indoor RH optimization would achieve high compliance because all occupants of a common indoor space would be exposed to similar ambient conditions,” the study said. “Compared to the long timelines and high costs of vaccine production and distribution, humidity control systems could potentially be implemented more quickly and cheaply in certain indoor settings.”
A version of this article first appeared on WebMD.com.
The “sweet spot” associated with reduced COVID-19 cases and deaths is 40%-60% indoor relative humidity, an MIT news release said. People who maintained indoor relative humidity outside those parameters had higher rates of catching COVID-19.
Most people are comfortable with 30%-50% relative humidity, researchers said. An airplane cabin has about 20% relative humidity.
Relative humidity is the amount of moisture in the air, compared with the total moisture the air can hold at a given temperature before saturating and forming condensation.
The study was published in The Journal of the Royal Society Interface. Researchers examined COVID-19 data and meteorological measurements from 121 countries from January 2020 through August 2020, before vaccines became available to the public.
“When outdoor temperatures were below the typical human comfort range, they assumed indoor spaces were heated to reach that comfort range. Based on the added heating, they calculated the associated drop in indoor relative humidity,” the MIT news release said.
The research teams found that when a region reported a rise in COVID-19 cases and deaths, the region’s estimated indoor relative humidity was either lower than 40% or higher than 60%, the release said.
“There’s potentially a protective effect of this intermediate indoor relative humidity,” said Connor Verheyen, the lead author and a PhD student in medical engineering and medical physics in the Harvard-MIT Program in Health Sciences and Technology.
Widespread use of the 40%-60% indoor humidity range could reduce the need for lockdowns and other widespread restrictions, the study concluded.
“Unlike measures that depend on individual compliance (for example, masking or hand-washing), indoor RH optimization would achieve high compliance because all occupants of a common indoor space would be exposed to similar ambient conditions,” the study said. “Compared to the long timelines and high costs of vaccine production and distribution, humidity control systems could potentially be implemented more quickly and cheaply in certain indoor settings.”
A version of this article first appeared on WebMD.com.
FROM THE JOURNAL OF THE ROYAL SOCIETY INTERFACE
Laser and light devices for acne treatment continue to advance
The calendar year
This was preceded by the FDA clearance of AviClear, marketed by Cutera, in March, and the commercial launch of TheraClearX, marketed by StrataSkin, in July.
“It’s an exciting time to be working with acne,” Fernanda H. Sakamoto, MD, PhD, a dermatologist at the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston. “We’ll see a lot of people using new devices. I’m looking forward to seeing results in the long term.”
AviClear and the Accure Laser System, marketed by Accure, are both powered by a 1,726-nm laser, but they work differently. AviClear, which was cleared for the treatment of mild, moderate, and severe acne, has a maximum fluence of 30 J/cm2 in single-pulse mode and a maximum fluence of 20 J/cm2 in double-pulse mode. The treatment handpiece has an integrated scanner for delivering treatment spot(s) in an operator-selected pattern. “It’s a little bit lower powered than the Accure and has a maximum pulse energy of 5 joules and a pulse duration of up to 50 milliseconds,” Dr. Sakamoto said. In the treatment of acne, laser and light treatments target the sebaceous gland.
In pivotal data submitted to the FDA, 104 patients with acne who were enrolled at 7 U.S. sites received 304 treatments with AviClear spaced 2-5 weeks apart. Each treatment took about 30 minutes. Treatment success was defined as having at least 50% fewer inflammatory acne lesions 12 weeks after the final treatment visit, compared with baseline. At the week 4 follow-up visit, there were median and mean reductions of 42% and 37%, respectively, in the inflammatory lesion counts from baseline (P < .001). The researchers found that, at the week 4 follow-up visit, 36% of patients had achieved treatment success, which increased to 78% at the 12-week follow-up visit. Treatment was considered safe and tolerable, according to the manufacturer.
The other newcomer device with a 1,726-nm wavelength is the Accure Laser System, which features a smart laser handpiece for real-time thermal monitoring and precise delivery of laser emissions. The device received CE Mark approval in 2020 for the treatment of moderate acne, and on Nov. 22, 2022, the manufacturer announced that it had been cleared by the FDA for the treatment of mild to severe inflammatory acne vulgaris.
Dr. Sakamoto and her Wellman colleagues have been working with five dermatologists to conduct clinical trials of the device: Emil Tanghetti, MD, and Mitchel Goldman, MD, in California; Roy Geronemus, MD, in New York; Joel Cohen, MD, in Colorado; and Daniel Friedmann, MD, in Texas. As of Oct. 2, 2022, more than 50 patients with mild to severe acne were enrolled in four studies and an additional 30 were enrolled in a pilot facial acne trial, Dr. Sakamoto said. In the trials, patients are followed at 4, 8, 12, and 24 weeks post treatment.
Among patients enrolled in the facial acne trial, researchers have observed a 100% responder rate for patients with more than five acne lesions at 4, 8, 12, and 24 weeks post treatment after four monthly treatment sessions. The average lesion reduction at week 12 was 82% and the mean visual analog scale score immediately after treatment was 2.09 out of 10. Each patient received more than 12,000 trigger pulls of energy from the device overall with no adverse events reported. At 12 months, they observed a 90% inflammatory lesion count reduction from baseline and a rapid response to treatment: a 73% reduction achieved after the first two treatment sessions. Histologic studies revealed selective sebaceous gland destruction with no damage to the epidermis, surrounding dermis, or other skin structures.
Dr. Sakamoto emphasized that to date no direct clinical comparisons have been made between the AviClear and Accure devices. “Are all 1,726-nm lasers made equal? That is a question that we have to keep in our mind,” she said during the meeting, which was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. “They are using the same wavelength, but they are different types of lasers.”
For example, the Accure Laser treats to temperature, relies on air cooling, and is targeted to dermatologists and plastic surgeons, while the AviClear treats to fluence, relies on contact cooling, and includes med spas and other nonphysician providers as the target users. “Mathematically, the difference between the two devices is that the Accure can achieve deeper penetration in a single pulse, while the AviClear is a little more superficial,” she said. “Whether that is translated clinically is unknown at this point.”
Dr. Sakamoto also discussed the TheraClearX, which is FDA cleared for the treatment of mild, moderate, and severe acne, including comedonal, pustular, and inflammatory acne vulgaris. The device, which is a new version of the Palomar Acleara, uses a vacuum technique with up to 3 psi pressure in conjunction with broadband light with a wavelength spectrum of 500 nm–1,200 nm delivered through a liquid-cooled, handheld delivery system. The predicate device was the Aesthera Isolaz System. The vacuum extracts buildup of sebaceous material. “At the same time, it takes the blood out of the competing chromophore,” she said. “By doing so, it potentially damages the sebaceous glands and reduces the inflammatory lesions.”
Dr. Sakamoto disclosed that she is the founder of and science advisor for Lightwater Bioscience. She is also a science advisor for Accure Acne and has received portions of patent royalties from Massachusetts General Hospital.
The calendar year
This was preceded by the FDA clearance of AviClear, marketed by Cutera, in March, and the commercial launch of TheraClearX, marketed by StrataSkin, in July.
“It’s an exciting time to be working with acne,” Fernanda H. Sakamoto, MD, PhD, a dermatologist at the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston. “We’ll see a lot of people using new devices. I’m looking forward to seeing results in the long term.”
AviClear and the Accure Laser System, marketed by Accure, are both powered by a 1,726-nm laser, but they work differently. AviClear, which was cleared for the treatment of mild, moderate, and severe acne, has a maximum fluence of 30 J/cm2 in single-pulse mode and a maximum fluence of 20 J/cm2 in double-pulse mode. The treatment handpiece has an integrated scanner for delivering treatment spot(s) in an operator-selected pattern. “It’s a little bit lower powered than the Accure and has a maximum pulse energy of 5 joules and a pulse duration of up to 50 milliseconds,” Dr. Sakamoto said. In the treatment of acne, laser and light treatments target the sebaceous gland.
In pivotal data submitted to the FDA, 104 patients with acne who were enrolled at 7 U.S. sites received 304 treatments with AviClear spaced 2-5 weeks apart. Each treatment took about 30 minutes. Treatment success was defined as having at least 50% fewer inflammatory acne lesions 12 weeks after the final treatment visit, compared with baseline. At the week 4 follow-up visit, there were median and mean reductions of 42% and 37%, respectively, in the inflammatory lesion counts from baseline (P < .001). The researchers found that, at the week 4 follow-up visit, 36% of patients had achieved treatment success, which increased to 78% at the 12-week follow-up visit. Treatment was considered safe and tolerable, according to the manufacturer.
The other newcomer device with a 1,726-nm wavelength is the Accure Laser System, which features a smart laser handpiece for real-time thermal monitoring and precise delivery of laser emissions. The device received CE Mark approval in 2020 for the treatment of moderate acne, and on Nov. 22, 2022, the manufacturer announced that it had been cleared by the FDA for the treatment of mild to severe inflammatory acne vulgaris.
Dr. Sakamoto and her Wellman colleagues have been working with five dermatologists to conduct clinical trials of the device: Emil Tanghetti, MD, and Mitchel Goldman, MD, in California; Roy Geronemus, MD, in New York; Joel Cohen, MD, in Colorado; and Daniel Friedmann, MD, in Texas. As of Oct. 2, 2022, more than 50 patients with mild to severe acne were enrolled in four studies and an additional 30 were enrolled in a pilot facial acne trial, Dr. Sakamoto said. In the trials, patients are followed at 4, 8, 12, and 24 weeks post treatment.
Among patients enrolled in the facial acne trial, researchers have observed a 100% responder rate for patients with more than five acne lesions at 4, 8, 12, and 24 weeks post treatment after four monthly treatment sessions. The average lesion reduction at week 12 was 82% and the mean visual analog scale score immediately after treatment was 2.09 out of 10. Each patient received more than 12,000 trigger pulls of energy from the device overall with no adverse events reported. At 12 months, they observed a 90% inflammatory lesion count reduction from baseline and a rapid response to treatment: a 73% reduction achieved after the first two treatment sessions. Histologic studies revealed selective sebaceous gland destruction with no damage to the epidermis, surrounding dermis, or other skin structures.
Dr. Sakamoto emphasized that to date no direct clinical comparisons have been made between the AviClear and Accure devices. “Are all 1,726-nm lasers made equal? That is a question that we have to keep in our mind,” she said during the meeting, which was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. “They are using the same wavelength, but they are different types of lasers.”
For example, the Accure Laser treats to temperature, relies on air cooling, and is targeted to dermatologists and plastic surgeons, while the AviClear treats to fluence, relies on contact cooling, and includes med spas and other nonphysician providers as the target users. “Mathematically, the difference between the two devices is that the Accure can achieve deeper penetration in a single pulse, while the AviClear is a little more superficial,” she said. “Whether that is translated clinically is unknown at this point.”
Dr. Sakamoto also discussed the TheraClearX, which is FDA cleared for the treatment of mild, moderate, and severe acne, including comedonal, pustular, and inflammatory acne vulgaris. The device, which is a new version of the Palomar Acleara, uses a vacuum technique with up to 3 psi pressure in conjunction with broadband light with a wavelength spectrum of 500 nm–1,200 nm delivered through a liquid-cooled, handheld delivery system. The predicate device was the Aesthera Isolaz System. The vacuum extracts buildup of sebaceous material. “At the same time, it takes the blood out of the competing chromophore,” she said. “By doing so, it potentially damages the sebaceous glands and reduces the inflammatory lesions.”
Dr. Sakamoto disclosed that she is the founder of and science advisor for Lightwater Bioscience. She is also a science advisor for Accure Acne and has received portions of patent royalties from Massachusetts General Hospital.
The calendar year
This was preceded by the FDA clearance of AviClear, marketed by Cutera, in March, and the commercial launch of TheraClearX, marketed by StrataSkin, in July.
“It’s an exciting time to be working with acne,” Fernanda H. Sakamoto, MD, PhD, a dermatologist at the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston. “We’ll see a lot of people using new devices. I’m looking forward to seeing results in the long term.”
AviClear and the Accure Laser System, marketed by Accure, are both powered by a 1,726-nm laser, but they work differently. AviClear, which was cleared for the treatment of mild, moderate, and severe acne, has a maximum fluence of 30 J/cm2 in single-pulse mode and a maximum fluence of 20 J/cm2 in double-pulse mode. The treatment handpiece has an integrated scanner for delivering treatment spot(s) in an operator-selected pattern. “It’s a little bit lower powered than the Accure and has a maximum pulse energy of 5 joules and a pulse duration of up to 50 milliseconds,” Dr. Sakamoto said. In the treatment of acne, laser and light treatments target the sebaceous gland.
In pivotal data submitted to the FDA, 104 patients with acne who were enrolled at 7 U.S. sites received 304 treatments with AviClear spaced 2-5 weeks apart. Each treatment took about 30 minutes. Treatment success was defined as having at least 50% fewer inflammatory acne lesions 12 weeks after the final treatment visit, compared with baseline. At the week 4 follow-up visit, there were median and mean reductions of 42% and 37%, respectively, in the inflammatory lesion counts from baseline (P < .001). The researchers found that, at the week 4 follow-up visit, 36% of patients had achieved treatment success, which increased to 78% at the 12-week follow-up visit. Treatment was considered safe and tolerable, according to the manufacturer.
The other newcomer device with a 1,726-nm wavelength is the Accure Laser System, which features a smart laser handpiece for real-time thermal monitoring and precise delivery of laser emissions. The device received CE Mark approval in 2020 for the treatment of moderate acne, and on Nov. 22, 2022, the manufacturer announced that it had been cleared by the FDA for the treatment of mild to severe inflammatory acne vulgaris.
Dr. Sakamoto and her Wellman colleagues have been working with five dermatologists to conduct clinical trials of the device: Emil Tanghetti, MD, and Mitchel Goldman, MD, in California; Roy Geronemus, MD, in New York; Joel Cohen, MD, in Colorado; and Daniel Friedmann, MD, in Texas. As of Oct. 2, 2022, more than 50 patients with mild to severe acne were enrolled in four studies and an additional 30 were enrolled in a pilot facial acne trial, Dr. Sakamoto said. In the trials, patients are followed at 4, 8, 12, and 24 weeks post treatment.
Among patients enrolled in the facial acne trial, researchers have observed a 100% responder rate for patients with more than five acne lesions at 4, 8, 12, and 24 weeks post treatment after four monthly treatment sessions. The average lesion reduction at week 12 was 82% and the mean visual analog scale score immediately after treatment was 2.09 out of 10. Each patient received more than 12,000 trigger pulls of energy from the device overall with no adverse events reported. At 12 months, they observed a 90% inflammatory lesion count reduction from baseline and a rapid response to treatment: a 73% reduction achieved after the first two treatment sessions. Histologic studies revealed selective sebaceous gland destruction with no damage to the epidermis, surrounding dermis, or other skin structures.
Dr. Sakamoto emphasized that to date no direct clinical comparisons have been made between the AviClear and Accure devices. “Are all 1,726-nm lasers made equal? That is a question that we have to keep in our mind,” she said during the meeting, which was sponsored by Harvard Medical School, Massachusetts General Hospital, and the Wellman Center for Photomedicine. “They are using the same wavelength, but they are different types of lasers.”
For example, the Accure Laser treats to temperature, relies on air cooling, and is targeted to dermatologists and plastic surgeons, while the AviClear treats to fluence, relies on contact cooling, and includes med spas and other nonphysician providers as the target users. “Mathematically, the difference between the two devices is that the Accure can achieve deeper penetration in a single pulse, while the AviClear is a little more superficial,” she said. “Whether that is translated clinically is unknown at this point.”
Dr. Sakamoto also discussed the TheraClearX, which is FDA cleared for the treatment of mild, moderate, and severe acne, including comedonal, pustular, and inflammatory acne vulgaris. The device, which is a new version of the Palomar Acleara, uses a vacuum technique with up to 3 psi pressure in conjunction with broadband light with a wavelength spectrum of 500 nm–1,200 nm delivered through a liquid-cooled, handheld delivery system. The predicate device was the Aesthera Isolaz System. The vacuum extracts buildup of sebaceous material. “At the same time, it takes the blood out of the competing chromophore,” she said. “By doing so, it potentially damages the sebaceous glands and reduces the inflammatory lesions.”
Dr. Sakamoto disclosed that she is the founder of and science advisor for Lightwater Bioscience. She is also a science advisor for Accure Acne and has received portions of patent royalties from Massachusetts General Hospital.
FROM A LASER & AESTHETIC SKIN THERAPY COURSE
Primary Malignant Melanoma of the Middle Ear
To the Editor:
An 82-year-old man presented to our dermatology clinic for a total-body skin examination due to a recently diagnosed primary melanoma of the left middle ear. He reported pain of the left ear and water behind the left eardrum of 1 year’s duration. An otorhinolaryngologist performed surgery due to the severe mastoiditis. A biopsy of the contents of the left middle ear revealed malignant melanoma. Positron emission tomography–computed tomography revealed the mass was mainly located in the anterior aspect of the left middle ear with suspicion of tumor extension into the bony portion of the eustachian tube. No other disease was present. Prior to presentation to dermatology, gross excision of the left middle ear with removal of additional melanoma was confirmed by biopsy, and further analysis revealed v-Raf murine sarcoma viral oncogene (BRAF) was not detected while cellular proto-oncogene receptor kinase (KIT) mutation was detected on exon 13p (K642E).
The patient had no family history of melanoma. He never smoked and did not have contact with hazardous material. Initial examination at our clinic revealed no other suspicious pigmented lesions. After additional negative workup by the oncologist, the patient was presented to the tumor board, and postoperative radiotherapy was recommended to improve local control. Eight months after the patient’s initial diagnosis of the primary middle ear melanoma, a computed tomography–guided right lung biopsy showed metastatic melanoma. After various treatment modalities were discussed with the patient and his family, he was started on pembrolizumab. After 6 months on pembrolizumab, the patient developed autoimmune pneumonitis and pembrolizumab was discontinued. The patient elected to discontinue treatment and died 6 months later.
Malignant melanoma with primary involvement of the middle ear and mastoid mucosa rarely has been reported.1-3 Primary malignant melanoma of the middle ear mucosa is difficult to diagnose clinically. Difficulty and delay in diagnosis occur because of the location and frequent lack of pathognomonic symptoms of the disease.2 A comprehensive literature review by Maxwell et al3 in 2018 of the 10 reported primary middle ear mucosal melanomas found that patients most commonly presented with otorrhea, aural fullness, and hearing loss. Less common symptoms included otalgia, tinnitus, and facial weakness. Clinical examination revealed patients presented with serous otitis and/or a visible mass within the middle ear or external auditory canal. These melanomas demonstrated particularly poor outcomes, with 70% mortality, 20% local recurrence, and 50% distant metastasis. Distant metastases that occurred with primary middle ear mucosal melanoma include lung, liver, intraparotid, abdomen, and cutaneous metastasis.3
The specific pathophysiologic factors underlying the development of primary malignant melanoma of the middle ear mucosa are not known.2 The middle ear and its components develop from the first and second pharyngeal arches.4 Melanocyte precursors from the neural crest migrate during the seventh or eighth week of embryogenesis. These precursors migrate to the epidermis, various mucosal epithelial, hair follicles, dermis, retina, uveal tract, leptomeninges, inner ear, and other tissues.5 The ossicles of the middle ear develop from the neural crest6 and remain in the mesenchyme until the eighth month, when the surrounding tissue dissolves.4 Cutaneous melanomas arise from the malignant transformation of melanocytes in the skin of neural crest lineage. Noncutaneous melanomas are hypothesized to arise from melanoblasts migrating to noncutaneous organs after neural crest cells undergo an epithelial-mesenchymal translation.7
Melanoma 5-year survival rates vary based on the melanoma disease stage: 98% for stage 1, 90% for stage 2, 70% for stage 3, and 10% for stage 4. Although early-stage disease mainly is treated with surgery, advanced and unresectable disease is managed with different therapeutic options, including BRAF inhibitors such as vemurafenib, dabrafenib mesylate, and encorafenib; immune checkpoint inhibitors such as ipilimumab, nivolumab, and pembrolizumab; and oncolytic virus such as talimogene laherparepvec.8,9
Ninety percent of melanomas are of cutaneous origin. Extracutaneous melanomas may be derived from the uvea, leptomeninges, mucous membranes, and gastrointestinal tract.10 Mucosal melanomas are rare and represent only approximately 1% of all melanomas.11 In order of frequency, primary mucosal melanomas include the head and neck, anorectal region, vulvovaginal region, and urinary tract. UV radiation exposure is an important risk factor for cutaneous melanoma but has not been associated with the development of mucosal melanoma.7 In 2019, Altieri et al11 analyzed 1824 cases of mucosal melanoma and found that anatomic site influences survival because mucosal melanomas in the most occult anatomic sites—spinal/central nervous system, lung and pleura, liver, and pancreas—have the worst prognosis, likely because they have already metastasized by the time they are diagnosed. Due to their occult anatomic location and lack of early presenting signs and symptoms, mucosal melanomas are difficult to diagnose at an early stage, resulting in a poorer prognosis compared with cutaneous melanomas. The most important prognostic indicator for cutaneous melanomas of tumor thickness (ie, Breslow depth) provides less prognostic value for patients with mucosal melanoma. Limitations also include the lack of a standardized staging system for mucosal melanoma, but Altieri et al11 found that poorer survival in patients with mucosal melanoma was observed in relation to stage based on the clinical and pathologic tumor-node-metastasis staging system of the Surveillance, Epidemiology, and End Results program. An aggregate 5-year survival estimate of patients diagnosed with mucosal melanoma is 28%, underscoring that mucosal melanoma is an aggressive melanoma that carries a poor prognosis and warrants a more aggressive treatment approach at the time of diagnosis.11
Common treatment of primary middle ear mucosal melanoma involves a multimodality therapy including surgical oncological resection for most patients. Currently, radiation is in use for adjuvant treatment and definitive therapy in unresectable tumors or patients who are poor surgical candidates. Malignant melanoma traditionally was considered radioresistant, yet considerable variability in responsiveness has been observed both within and between tumors. Although there are no defined indications for adjuvant therapy, it is often administered in advanced or recurrent cases and those with positive or close margins. Chemotherapy generally is reserved for patients with systemic disease. The chemotherapeutic agents that have been used in the treatment of patients with melanoma of the middle ear include the alkylating agents dacarbazine, cisplatin, nimustine, paclitaxel, and temozolomide. Also, chemotherapeutic agents that have been reported in the treatment of melanoma of the middle ear include tamoxifen, the selective estrogen receptor inhibitor, and interferon. Most recently, programed cell death protein 1 inhibitors pembrolizumab and nivolumab have been used in the treatment of middle ear melanoma. Outcomes remain poor with a high rate of mortality. Novel immunotherapeutic agents combined with adjuvant radiotherapy have been proposed to improve disease control and survival rates.3
Data on systemic therapies for mucosal melanomas are limited due to the rarity of the disease. Even with the development of novel therapies, outcomes remain poor for mucosal melanomas, and additional treatment strategies are needed. Although proto-oncogene BRAF mutations occur in 50% to 70% of cutaneous melanomas, these mutations are rare in mucosal melanomas.3 In mucosal melanomas, activating mutations of the cell receptor KIT are identified more frequently.7 Alterations in proto-oncogene KIT have been found in acral, mucosal, and cutaneous melanoma. KIT mutations were found on exons 11 and 13.12 Variability in the biology of KIT is suggested. Treatment of melanomas with the KIT mutations with tyrosine inhibitors imatinib and nilotinib have shown variable benefits.10 In a 2019 study of 44 patients with mucosal melanoma, Moya-Plana et al13 found that in cases of unresectable and/or metastatic disease, immunotherapy with pembrolizumab had a better benefit-risk ratio than immune treatment with ipilimumab, a cytotoxic T-cell lymphocyte-associated protein 4 inhibitor.
Primary malignant melanoma of the middle ear is unusual and difficult to diagnose clinically. These melanomas have a poor prognosis and can have distant metastasis including cutaneous metastasis. We present this case to emphasize the need to be aware that melanoma can arise in the middle ear.
- Ozturk O, Baglam T, Uneri C, et al. Primary malignant melanoma of the middle ear mucosa: a case report. Kulak Burun Bogaz Ihtis Derg. 2006;16:83-86.
- Idris IA, Daud KM, Yusof Z, et al. Primary malignant melanoma of the middle ear mucosa: a case report. Egypt J ENT Allied Sci. 2017;18:307-309.
- Maxwell AK, Takeda H, Gubbels SP. Primary middle ear mucosal melanoma: case report and comprehensive literature review of 21 cases of primary middle ear and eustachian tube melanoma. Ann Otol Rhinol Laryngol. 2018;127:856-863.
- Sadler TW. Ear. In: Sadler TW, ed. Langman’s Medical Embryology. Lippincott Williams & Wilkins; 2012:324-325.
- Jakubovic HR, Akerman AB. Structure and function of skin: development, morphology and physiology. In: Moschella SL, Hurley HJ, eds. Dermatology. Vol 1. WB Saunders Co; 1985:22-23.
- Sadler TW. The axial skeleton. In: Sadler TW, ed. Langman’s Medical Embryology. Lippincott Williams & Wilkins; 2012:133-137.
- Tacastacas JD, Bray J, Cohen YK, et al. Update on primary mucosal melanoma. J Am Acad Dermatol. 2014;71:366-375.
- Abdutaali R, Alkhattib NS, Oh M, et al. Economic evaluation of talimogene laherparepvec plus ipilimumab combination therapy vs ipilimumab monotherapy in patients with advanced unresectable melanoma. JAMA Dermatol. 2019;155:22-28.
- Skudalski L, Waldeman R, Kerr PE, et al. Melanoma: an update on systemic therapies. J Am Acad Dermatol. 2022;86:515-524.
- Heymann WR. A step toward demystifying melanomas of unknown primary sites. J Am Acad Dermatol. 2018;79:208-209.
- Altieri L, Eguchi M, Peng DH, et al. Predictors of mucosal melanoma survival in a population-based setting. J Am Acad Dermatol. 2019;81:136-142.
- Volpe VO, Klufas DM, Hegde U, et al. The new paradigm of systemic therapies for metastatic melanoma. J Am Acad Dermatol. 2017;77:356-368.
- Moya-Plana A, Herrera Gomez RG, Rossoni C, et al. Evaluation of the efficacy of immunotherapy for non-resectable mucosal melanoma. Cancer Immunol Immunother. 2019;68:1171-1178.
To the Editor:
An 82-year-old man presented to our dermatology clinic for a total-body skin examination due to a recently diagnosed primary melanoma of the left middle ear. He reported pain of the left ear and water behind the left eardrum of 1 year’s duration. An otorhinolaryngologist performed surgery due to the severe mastoiditis. A biopsy of the contents of the left middle ear revealed malignant melanoma. Positron emission tomography–computed tomography revealed the mass was mainly located in the anterior aspect of the left middle ear with suspicion of tumor extension into the bony portion of the eustachian tube. No other disease was present. Prior to presentation to dermatology, gross excision of the left middle ear with removal of additional melanoma was confirmed by biopsy, and further analysis revealed v-Raf murine sarcoma viral oncogene (BRAF) was not detected while cellular proto-oncogene receptor kinase (KIT) mutation was detected on exon 13p (K642E).
The patient had no family history of melanoma. He never smoked and did not have contact with hazardous material. Initial examination at our clinic revealed no other suspicious pigmented lesions. After additional negative workup by the oncologist, the patient was presented to the tumor board, and postoperative radiotherapy was recommended to improve local control. Eight months after the patient’s initial diagnosis of the primary middle ear melanoma, a computed tomography–guided right lung biopsy showed metastatic melanoma. After various treatment modalities were discussed with the patient and his family, he was started on pembrolizumab. After 6 months on pembrolizumab, the patient developed autoimmune pneumonitis and pembrolizumab was discontinued. The patient elected to discontinue treatment and died 6 months later.
Malignant melanoma with primary involvement of the middle ear and mastoid mucosa rarely has been reported.1-3 Primary malignant melanoma of the middle ear mucosa is difficult to diagnose clinically. Difficulty and delay in diagnosis occur because of the location and frequent lack of pathognomonic symptoms of the disease.2 A comprehensive literature review by Maxwell et al3 in 2018 of the 10 reported primary middle ear mucosal melanomas found that patients most commonly presented with otorrhea, aural fullness, and hearing loss. Less common symptoms included otalgia, tinnitus, and facial weakness. Clinical examination revealed patients presented with serous otitis and/or a visible mass within the middle ear or external auditory canal. These melanomas demonstrated particularly poor outcomes, with 70% mortality, 20% local recurrence, and 50% distant metastasis. Distant metastases that occurred with primary middle ear mucosal melanoma include lung, liver, intraparotid, abdomen, and cutaneous metastasis.3
The specific pathophysiologic factors underlying the development of primary malignant melanoma of the middle ear mucosa are not known.2 The middle ear and its components develop from the first and second pharyngeal arches.4 Melanocyte precursors from the neural crest migrate during the seventh or eighth week of embryogenesis. These precursors migrate to the epidermis, various mucosal epithelial, hair follicles, dermis, retina, uveal tract, leptomeninges, inner ear, and other tissues.5 The ossicles of the middle ear develop from the neural crest6 and remain in the mesenchyme until the eighth month, when the surrounding tissue dissolves.4 Cutaneous melanomas arise from the malignant transformation of melanocytes in the skin of neural crest lineage. Noncutaneous melanomas are hypothesized to arise from melanoblasts migrating to noncutaneous organs after neural crest cells undergo an epithelial-mesenchymal translation.7
Melanoma 5-year survival rates vary based on the melanoma disease stage: 98% for stage 1, 90% for stage 2, 70% for stage 3, and 10% for stage 4. Although early-stage disease mainly is treated with surgery, advanced and unresectable disease is managed with different therapeutic options, including BRAF inhibitors such as vemurafenib, dabrafenib mesylate, and encorafenib; immune checkpoint inhibitors such as ipilimumab, nivolumab, and pembrolizumab; and oncolytic virus such as talimogene laherparepvec.8,9
Ninety percent of melanomas are of cutaneous origin. Extracutaneous melanomas may be derived from the uvea, leptomeninges, mucous membranes, and gastrointestinal tract.10 Mucosal melanomas are rare and represent only approximately 1% of all melanomas.11 In order of frequency, primary mucosal melanomas include the head and neck, anorectal region, vulvovaginal region, and urinary tract. UV radiation exposure is an important risk factor for cutaneous melanoma but has not been associated with the development of mucosal melanoma.7 In 2019, Altieri et al11 analyzed 1824 cases of mucosal melanoma and found that anatomic site influences survival because mucosal melanomas in the most occult anatomic sites—spinal/central nervous system, lung and pleura, liver, and pancreas—have the worst prognosis, likely because they have already metastasized by the time they are diagnosed. Due to their occult anatomic location and lack of early presenting signs and symptoms, mucosal melanomas are difficult to diagnose at an early stage, resulting in a poorer prognosis compared with cutaneous melanomas. The most important prognostic indicator for cutaneous melanomas of tumor thickness (ie, Breslow depth) provides less prognostic value for patients with mucosal melanoma. Limitations also include the lack of a standardized staging system for mucosal melanoma, but Altieri et al11 found that poorer survival in patients with mucosal melanoma was observed in relation to stage based on the clinical and pathologic tumor-node-metastasis staging system of the Surveillance, Epidemiology, and End Results program. An aggregate 5-year survival estimate of patients diagnosed with mucosal melanoma is 28%, underscoring that mucosal melanoma is an aggressive melanoma that carries a poor prognosis and warrants a more aggressive treatment approach at the time of diagnosis.11
Common treatment of primary middle ear mucosal melanoma involves a multimodality therapy including surgical oncological resection for most patients. Currently, radiation is in use for adjuvant treatment and definitive therapy in unresectable tumors or patients who are poor surgical candidates. Malignant melanoma traditionally was considered radioresistant, yet considerable variability in responsiveness has been observed both within and between tumors. Although there are no defined indications for adjuvant therapy, it is often administered in advanced or recurrent cases and those with positive or close margins. Chemotherapy generally is reserved for patients with systemic disease. The chemotherapeutic agents that have been used in the treatment of patients with melanoma of the middle ear include the alkylating agents dacarbazine, cisplatin, nimustine, paclitaxel, and temozolomide. Also, chemotherapeutic agents that have been reported in the treatment of melanoma of the middle ear include tamoxifen, the selective estrogen receptor inhibitor, and interferon. Most recently, programed cell death protein 1 inhibitors pembrolizumab and nivolumab have been used in the treatment of middle ear melanoma. Outcomes remain poor with a high rate of mortality. Novel immunotherapeutic agents combined with adjuvant radiotherapy have been proposed to improve disease control and survival rates.3
Data on systemic therapies for mucosal melanomas are limited due to the rarity of the disease. Even with the development of novel therapies, outcomes remain poor for mucosal melanomas, and additional treatment strategies are needed. Although proto-oncogene BRAF mutations occur in 50% to 70% of cutaneous melanomas, these mutations are rare in mucosal melanomas.3 In mucosal melanomas, activating mutations of the cell receptor KIT are identified more frequently.7 Alterations in proto-oncogene KIT have been found in acral, mucosal, and cutaneous melanoma. KIT mutations were found on exons 11 and 13.12 Variability in the biology of KIT is suggested. Treatment of melanomas with the KIT mutations with tyrosine inhibitors imatinib and nilotinib have shown variable benefits.10 In a 2019 study of 44 patients with mucosal melanoma, Moya-Plana et al13 found that in cases of unresectable and/or metastatic disease, immunotherapy with pembrolizumab had a better benefit-risk ratio than immune treatment with ipilimumab, a cytotoxic T-cell lymphocyte-associated protein 4 inhibitor.
Primary malignant melanoma of the middle ear is unusual and difficult to diagnose clinically. These melanomas have a poor prognosis and can have distant metastasis including cutaneous metastasis. We present this case to emphasize the need to be aware that melanoma can arise in the middle ear.
To the Editor:
An 82-year-old man presented to our dermatology clinic for a total-body skin examination due to a recently diagnosed primary melanoma of the left middle ear. He reported pain of the left ear and water behind the left eardrum of 1 year’s duration. An otorhinolaryngologist performed surgery due to the severe mastoiditis. A biopsy of the contents of the left middle ear revealed malignant melanoma. Positron emission tomography–computed tomography revealed the mass was mainly located in the anterior aspect of the left middle ear with suspicion of tumor extension into the bony portion of the eustachian tube. No other disease was present. Prior to presentation to dermatology, gross excision of the left middle ear with removal of additional melanoma was confirmed by biopsy, and further analysis revealed v-Raf murine sarcoma viral oncogene (BRAF) was not detected while cellular proto-oncogene receptor kinase (KIT) mutation was detected on exon 13p (K642E).
The patient had no family history of melanoma. He never smoked and did not have contact with hazardous material. Initial examination at our clinic revealed no other suspicious pigmented lesions. After additional negative workup by the oncologist, the patient was presented to the tumor board, and postoperative radiotherapy was recommended to improve local control. Eight months after the patient’s initial diagnosis of the primary middle ear melanoma, a computed tomography–guided right lung biopsy showed metastatic melanoma. After various treatment modalities were discussed with the patient and his family, he was started on pembrolizumab. After 6 months on pembrolizumab, the patient developed autoimmune pneumonitis and pembrolizumab was discontinued. The patient elected to discontinue treatment and died 6 months later.
Malignant melanoma with primary involvement of the middle ear and mastoid mucosa rarely has been reported.1-3 Primary malignant melanoma of the middle ear mucosa is difficult to diagnose clinically. Difficulty and delay in diagnosis occur because of the location and frequent lack of pathognomonic symptoms of the disease.2 A comprehensive literature review by Maxwell et al3 in 2018 of the 10 reported primary middle ear mucosal melanomas found that patients most commonly presented with otorrhea, aural fullness, and hearing loss. Less common symptoms included otalgia, tinnitus, and facial weakness. Clinical examination revealed patients presented with serous otitis and/or a visible mass within the middle ear or external auditory canal. These melanomas demonstrated particularly poor outcomes, with 70% mortality, 20% local recurrence, and 50% distant metastasis. Distant metastases that occurred with primary middle ear mucosal melanoma include lung, liver, intraparotid, abdomen, and cutaneous metastasis.3
The specific pathophysiologic factors underlying the development of primary malignant melanoma of the middle ear mucosa are not known.2 The middle ear and its components develop from the first and second pharyngeal arches.4 Melanocyte precursors from the neural crest migrate during the seventh or eighth week of embryogenesis. These precursors migrate to the epidermis, various mucosal epithelial, hair follicles, dermis, retina, uveal tract, leptomeninges, inner ear, and other tissues.5 The ossicles of the middle ear develop from the neural crest6 and remain in the mesenchyme until the eighth month, when the surrounding tissue dissolves.4 Cutaneous melanomas arise from the malignant transformation of melanocytes in the skin of neural crest lineage. Noncutaneous melanomas are hypothesized to arise from melanoblasts migrating to noncutaneous organs after neural crest cells undergo an epithelial-mesenchymal translation.7
Melanoma 5-year survival rates vary based on the melanoma disease stage: 98% for stage 1, 90% for stage 2, 70% for stage 3, and 10% for stage 4. Although early-stage disease mainly is treated with surgery, advanced and unresectable disease is managed with different therapeutic options, including BRAF inhibitors such as vemurafenib, dabrafenib mesylate, and encorafenib; immune checkpoint inhibitors such as ipilimumab, nivolumab, and pembrolizumab; and oncolytic virus such as talimogene laherparepvec.8,9
Ninety percent of melanomas are of cutaneous origin. Extracutaneous melanomas may be derived from the uvea, leptomeninges, mucous membranes, and gastrointestinal tract.10 Mucosal melanomas are rare and represent only approximately 1% of all melanomas.11 In order of frequency, primary mucosal melanomas include the head and neck, anorectal region, vulvovaginal region, and urinary tract. UV radiation exposure is an important risk factor for cutaneous melanoma but has not been associated with the development of mucosal melanoma.7 In 2019, Altieri et al11 analyzed 1824 cases of mucosal melanoma and found that anatomic site influences survival because mucosal melanomas in the most occult anatomic sites—spinal/central nervous system, lung and pleura, liver, and pancreas—have the worst prognosis, likely because they have already metastasized by the time they are diagnosed. Due to their occult anatomic location and lack of early presenting signs and symptoms, mucosal melanomas are difficult to diagnose at an early stage, resulting in a poorer prognosis compared with cutaneous melanomas. The most important prognostic indicator for cutaneous melanomas of tumor thickness (ie, Breslow depth) provides less prognostic value for patients with mucosal melanoma. Limitations also include the lack of a standardized staging system for mucosal melanoma, but Altieri et al11 found that poorer survival in patients with mucosal melanoma was observed in relation to stage based on the clinical and pathologic tumor-node-metastasis staging system of the Surveillance, Epidemiology, and End Results program. An aggregate 5-year survival estimate of patients diagnosed with mucosal melanoma is 28%, underscoring that mucosal melanoma is an aggressive melanoma that carries a poor prognosis and warrants a more aggressive treatment approach at the time of diagnosis.11
Common treatment of primary middle ear mucosal melanoma involves a multimodality therapy including surgical oncological resection for most patients. Currently, radiation is in use for adjuvant treatment and definitive therapy in unresectable tumors or patients who are poor surgical candidates. Malignant melanoma traditionally was considered radioresistant, yet considerable variability in responsiveness has been observed both within and between tumors. Although there are no defined indications for adjuvant therapy, it is often administered in advanced or recurrent cases and those with positive or close margins. Chemotherapy generally is reserved for patients with systemic disease. The chemotherapeutic agents that have been used in the treatment of patients with melanoma of the middle ear include the alkylating agents dacarbazine, cisplatin, nimustine, paclitaxel, and temozolomide. Also, chemotherapeutic agents that have been reported in the treatment of melanoma of the middle ear include tamoxifen, the selective estrogen receptor inhibitor, and interferon. Most recently, programed cell death protein 1 inhibitors pembrolizumab and nivolumab have been used in the treatment of middle ear melanoma. Outcomes remain poor with a high rate of mortality. Novel immunotherapeutic agents combined with adjuvant radiotherapy have been proposed to improve disease control and survival rates.3
Data on systemic therapies for mucosal melanomas are limited due to the rarity of the disease. Even with the development of novel therapies, outcomes remain poor for mucosal melanomas, and additional treatment strategies are needed. Although proto-oncogene BRAF mutations occur in 50% to 70% of cutaneous melanomas, these mutations are rare in mucosal melanomas.3 In mucosal melanomas, activating mutations of the cell receptor KIT are identified more frequently.7 Alterations in proto-oncogene KIT have been found in acral, mucosal, and cutaneous melanoma. KIT mutations were found on exons 11 and 13.12 Variability in the biology of KIT is suggested. Treatment of melanomas with the KIT mutations with tyrosine inhibitors imatinib and nilotinib have shown variable benefits.10 In a 2019 study of 44 patients with mucosal melanoma, Moya-Plana et al13 found that in cases of unresectable and/or metastatic disease, immunotherapy with pembrolizumab had a better benefit-risk ratio than immune treatment with ipilimumab, a cytotoxic T-cell lymphocyte-associated protein 4 inhibitor.
Primary malignant melanoma of the middle ear is unusual and difficult to diagnose clinically. These melanomas have a poor prognosis and can have distant metastasis including cutaneous metastasis. We present this case to emphasize the need to be aware that melanoma can arise in the middle ear.
- Ozturk O, Baglam T, Uneri C, et al. Primary malignant melanoma of the middle ear mucosa: a case report. Kulak Burun Bogaz Ihtis Derg. 2006;16:83-86.
- Idris IA, Daud KM, Yusof Z, et al. Primary malignant melanoma of the middle ear mucosa: a case report. Egypt J ENT Allied Sci. 2017;18:307-309.
- Maxwell AK, Takeda H, Gubbels SP. Primary middle ear mucosal melanoma: case report and comprehensive literature review of 21 cases of primary middle ear and eustachian tube melanoma. Ann Otol Rhinol Laryngol. 2018;127:856-863.
- Sadler TW. Ear. In: Sadler TW, ed. Langman’s Medical Embryology. Lippincott Williams & Wilkins; 2012:324-325.
- Jakubovic HR, Akerman AB. Structure and function of skin: development, morphology and physiology. In: Moschella SL, Hurley HJ, eds. Dermatology. Vol 1. WB Saunders Co; 1985:22-23.
- Sadler TW. The axial skeleton. In: Sadler TW, ed. Langman’s Medical Embryology. Lippincott Williams & Wilkins; 2012:133-137.
- Tacastacas JD, Bray J, Cohen YK, et al. Update on primary mucosal melanoma. J Am Acad Dermatol. 2014;71:366-375.
- Abdutaali R, Alkhattib NS, Oh M, et al. Economic evaluation of talimogene laherparepvec plus ipilimumab combination therapy vs ipilimumab monotherapy in patients with advanced unresectable melanoma. JAMA Dermatol. 2019;155:22-28.
- Skudalski L, Waldeman R, Kerr PE, et al. Melanoma: an update on systemic therapies. J Am Acad Dermatol. 2022;86:515-524.
- Heymann WR. A step toward demystifying melanomas of unknown primary sites. J Am Acad Dermatol. 2018;79:208-209.
- Altieri L, Eguchi M, Peng DH, et al. Predictors of mucosal melanoma survival in a population-based setting. J Am Acad Dermatol. 2019;81:136-142.
- Volpe VO, Klufas DM, Hegde U, et al. The new paradigm of systemic therapies for metastatic melanoma. J Am Acad Dermatol. 2017;77:356-368.
- Moya-Plana A, Herrera Gomez RG, Rossoni C, et al. Evaluation of the efficacy of immunotherapy for non-resectable mucosal melanoma. Cancer Immunol Immunother. 2019;68:1171-1178.
- Ozturk O, Baglam T, Uneri C, et al. Primary malignant melanoma of the middle ear mucosa: a case report. Kulak Burun Bogaz Ihtis Derg. 2006;16:83-86.
- Idris IA, Daud KM, Yusof Z, et al. Primary malignant melanoma of the middle ear mucosa: a case report. Egypt J ENT Allied Sci. 2017;18:307-309.
- Maxwell AK, Takeda H, Gubbels SP. Primary middle ear mucosal melanoma: case report and comprehensive literature review of 21 cases of primary middle ear and eustachian tube melanoma. Ann Otol Rhinol Laryngol. 2018;127:856-863.
- Sadler TW. Ear. In: Sadler TW, ed. Langman’s Medical Embryology. Lippincott Williams & Wilkins; 2012:324-325.
- Jakubovic HR, Akerman AB. Structure and function of skin: development, morphology and physiology. In: Moschella SL, Hurley HJ, eds. Dermatology. Vol 1. WB Saunders Co; 1985:22-23.
- Sadler TW. The axial skeleton. In: Sadler TW, ed. Langman’s Medical Embryology. Lippincott Williams & Wilkins; 2012:133-137.
- Tacastacas JD, Bray J, Cohen YK, et al. Update on primary mucosal melanoma. J Am Acad Dermatol. 2014;71:366-375.
- Abdutaali R, Alkhattib NS, Oh M, et al. Economic evaluation of talimogene laherparepvec plus ipilimumab combination therapy vs ipilimumab monotherapy in patients with advanced unresectable melanoma. JAMA Dermatol. 2019;155:22-28.
- Skudalski L, Waldeman R, Kerr PE, et al. Melanoma: an update on systemic therapies. J Am Acad Dermatol. 2022;86:515-524.
- Heymann WR. A step toward demystifying melanomas of unknown primary sites. J Am Acad Dermatol. 2018;79:208-209.
- Altieri L, Eguchi M, Peng DH, et al. Predictors of mucosal melanoma survival in a population-based setting. J Am Acad Dermatol. 2019;81:136-142.
- Volpe VO, Klufas DM, Hegde U, et al. The new paradigm of systemic therapies for metastatic melanoma. J Am Acad Dermatol. 2017;77:356-368.
- Moya-Plana A, Herrera Gomez RG, Rossoni C, et al. Evaluation of the efficacy of immunotherapy for non-resectable mucosal melanoma. Cancer Immunol Immunother. 2019;68:1171-1178.
PRACTICE POINTS
- Primary malignant melanoma of the middle ear is rare and has poor prognosis.
- Distant metastasis, including cutaneous metastasis, results from primary middle ear melanoma.
A Trauma-Induced Fatty Mass: The Facts About Posttraumatic Pseudolipomas
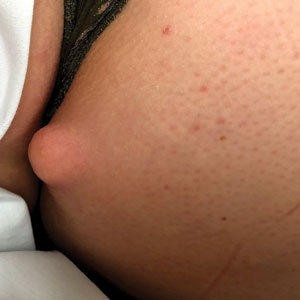
To the Editor:
The posttraumatic pseudolipoma (PTL) is a painless localized mass comprised of unencapsulated adipose tissue that develops at the site of acute or prolonged blunt soft tissue trauma. It may be round or fusiform in shape and has areas of saponification leading to fat necrosis.1 Posttraumatic pseudolipomas are 12 times more likely to occur in females, which may be attributed to sex-determined adipose tissue distribution or cosmetic concerns.2 Most PTLs are found in areas of the body with high adiposity, including the hip, thigh, and gluteal regions.3 A patient history of a traumatic event resulting in a hematoma and a subsequent latent period of several months to years before the pseudolipoma formation occurs is common.1,2,4-6
A 27-year-old woman presented to the family medicine clinic for examination of a deformity on the right buttock. She noticed a soft protruding mass months after landing on the buttocks and on top of a stick during routine physical training. Prior ultrasonography of the deformity proved unhelpful in determining the etiology. Physical examination revealed a protruding, 2-cm, flesh-colored mass on the right buttock intergluteal fold that was soft, compressible, and nontender (Figure 1). There was no capsule, nodule, loculation, or sinus tract. The patient underwent excisional resection with findings of benign-appearing unencapsulated adipose tissue (Figure 2). The wound was closed without difficulty. After several weeks, she had a well-healing scar without contour deficits of the buttocks. Two to 3 months after the initial repair, the patient presented to the family medicine clinic with recurrence of the fatty protrusion. She was referred for consultation and definitive management to a plastic surgeon but was lost to follow up.
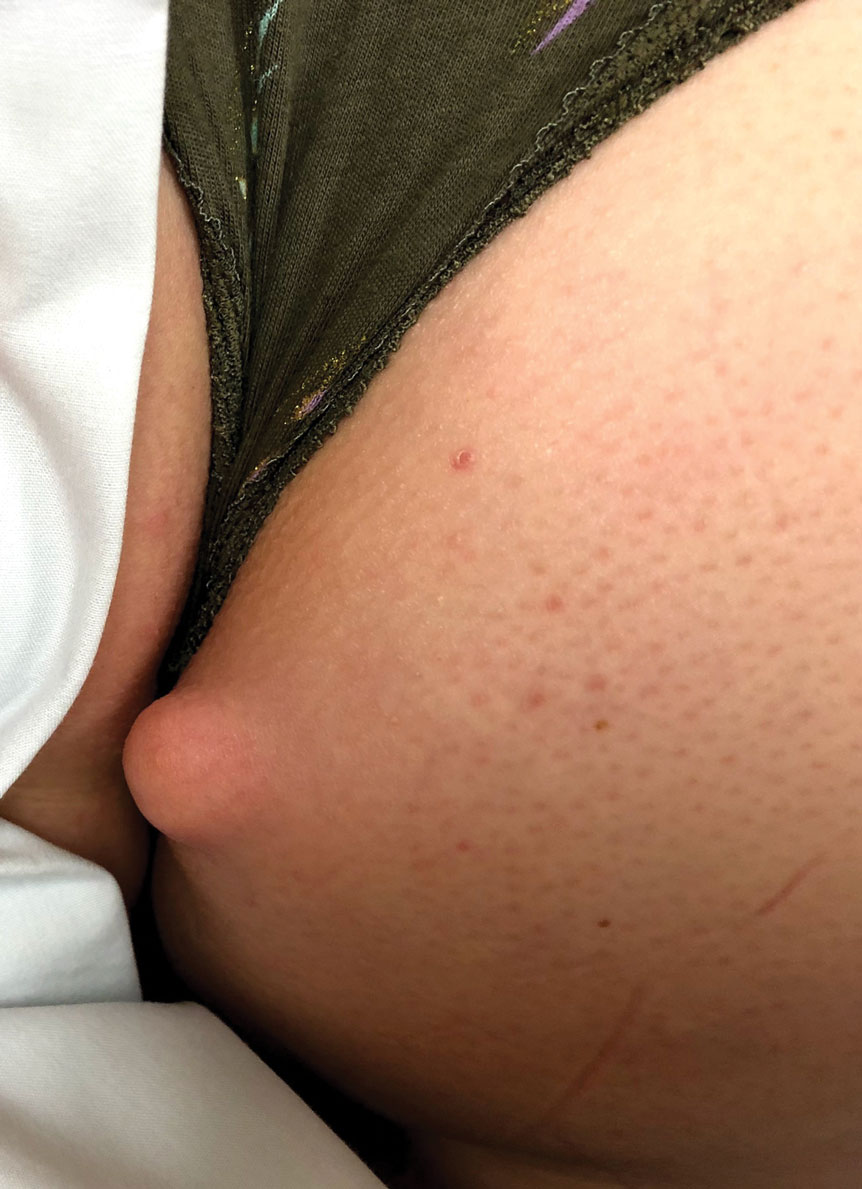
In a systematic review of the literature to research pathogenesis theories, a PubMed search of articles indexed for MEDLINE using the terms trauma and pseudolipoma, lipoma, fat, or adipose yielded 45 citations, with only 10 publications addressing the pathology specific to pseudolipomas. Two leading theories of the pathogenesis of PTLs include the adipose herniation pathway and the inflammatory proliferation pathway.4,5
Adipose tissue comprises fat lobules that are organized underneath the supportive elastic fascial layers. Injury from forces exceeding the fascial strength is the basis for the oldest pathogenesis theory. The adipose herniation theory suggests that fat lobules are displaced through the damaged septae, allowing for the development of an epidermal pseudolipoma at the site of blunt trauma.7 This theory has been supported by many case reports; however, more recent reports have identified a larger number of PTL cases that showed no identifiable disruptions in the fascia.1,4,8
In 1997, the inflammatory proliferation theory began to gain attention. The theory describes how local tissue trauma leads to the release of inflammatory cytokines, which successively signals the development of preadipocytes or adipose tissue–derived stem cells (ASCs) into mature adipocytes.4 Most patients report a history of a hematoma in the area of pseudolipoma development, which strongly supports this newer theory. Studies exploring hematomas have found elevated levels of growth factors and inflammatory markers.2,9 In particular, tumor necrosis factor α, peroxisome proliferator–activated receptor γ, vascular endothelial growth factor, and IL-6 and IL-8 may foster an environment in which adipogenic cells are both chemotaxed to the area of trauma and differentiated to white adipose tissue.2,10
Despite addressing the role of the preadipocyte, the available research fails to address the general development of mesenchymal cells into the preadipocyte. White adipose tissue develops at sites of neovascularization and frequently has been observed spreading into the nearby tissue toward other blood vessels. Furthermore, these white adipose tissue expansions remain reliant on multiple growth factors and cell-signaling molecules.10 Numerous investigations into stem cell grafting have found that implantation of ASCs in vivo within animal models does not result in the proliferation and differentiation of ASCs unless specific conditions have been met such as prior tissue injury or immunodeficiency.10-12 These investigations support and expand on the inflammatory proliferation pathway. Thus, most of the true PTLs in the available research appear as de novo tumors and are more congruent with the inflammatory proliferation model.1,2,4-6,8
Typical treatment of a PTL is surgical excision or liposuction depending on the pathology and size of the pseudolipoma. Biopsy examination prior to liposuction is critical for evaluation of liposarcoma and may help identify damage to Scarpa fascia. Recurrence of a PTL is rare regardless of treatment method; however, in a study of 31 PTL cases, only 6 were pathologically identified as PTLs without fibrous material.1
Our patient experienced a blunt trauma to the buttocks and subsequently developed a PTL that was surgically excised and recurred within 3 months. Research surrounding the pathogenesis of the PTL has evolved from the theory of physical herniation of adipose tissue to an inflammatory differentiation of preadipocytes, but there is still much to learn about how and why it occurs and the mesenchymal differentiation following tissue injury.
- Aust MC, Spies M, Kall S, et al. Lipomas after blunt soft tissue trauma: are they real? analysis of 31 cases. Br J Dermatol. 2007;157:92-99. doi:10.1111/j.1365-2133.2007.07970.x
- Galea LA, Penington AJ, Morrison WA. Post-traumatic pseudolipomas—a review and postulated mechanisms of their development. J Plast Reconstr Aesthet Surg. 2009;62:737-741. doi:10.1016/j.bjps.2008.12.021
- Zajac JC, Mandelbaum M, Economides JM, et al. Immediate massive posttraumatic pseudolipoma of the buttocks: a case of a heterotopic “love handle.” Plast Reconstr Surg Glob Open. 2018;6:E1887. doi:10.1097/GOX.0000000000001887
- Signorini M, Campiglio GL. Posttraumatic lipomas: where do they really come from? Plast Reconstr Surg. 1998;101:699-705. doi:10.1097/00006534-199803000-00017
- Khadilkar AS, Goyal A, Gauba K. The enigma of “traumatic pseudolipoma” and “traumatic herniation of buccal fat pad”: a systematic review and new classification system of post-traumatic craniofacial fatty masses. J Oral Maxillofac Surg. 2018;76:1267-1278. doi:10.1016/j.joms.2017.01.024
- Copcu E, Sivrioglu NS. Posttraumatic lipoma: analysis of 10 cases and explanation of possible mechanisms. Dermatol Surg. 2003;29:215-220. doi:10.1046/j.1524-4725.2003.29052.x
- Penoff JH. Traumatic lipomas/pseudolipomas. J Trauma. 1982;22:63-65. doi:10.1097/00005373-198201000-00013
- Theumann N, Abdelmoumene A, Wintermark M, et al. Posttraumatic pseudolipoma: MRI appearances. Eur Radiol. 2005;15:1876-1880. doi:10.1007/s00330-005-2757-2
- David LR, DeFranzo A, Marks M, et al. Posttraumatic pseudolipoma. J Trauma. 1996;40:396-400. doi:10.1097/00005373-199603000-00012
- Cawthorn WP, Scheller EL, MacDougald OA. Adipose tissue stem cells meet preadipocyte commitment: going back to the future. J Lipid Res. 2012;53:227-246. doi:10.1194/jlr.R021089
- Joe AW, Yi L, Natarajan A, et al. Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat Cell Biol. 2010;12:153-163. doi:10.1038/ncb2015
- Miranville A, Heeschen C, Sengenès C, et al. Improvement of postnatal neovascularization by human adipose tissue-derived stem cells. Circulation. 2004;110:349-355. doi:10.1161/01.Cir.0000135466.16823.D0
To the Editor:
The posttraumatic pseudolipoma (PTL) is a painless localized mass comprised of unencapsulated adipose tissue that develops at the site of acute or prolonged blunt soft tissue trauma. It may be round or fusiform in shape and has areas of saponification leading to fat necrosis.1 Posttraumatic pseudolipomas are 12 times more likely to occur in females, which may be attributed to sex-determined adipose tissue distribution or cosmetic concerns.2 Most PTLs are found in areas of the body with high adiposity, including the hip, thigh, and gluteal regions.3 A patient history of a traumatic event resulting in a hematoma and a subsequent latent period of several months to years before the pseudolipoma formation occurs is common.1,2,4-6
A 27-year-old woman presented to the family medicine clinic for examination of a deformity on the right buttock. She noticed a soft protruding mass months after landing on the buttocks and on top of a stick during routine physical training. Prior ultrasonography of the deformity proved unhelpful in determining the etiology. Physical examination revealed a protruding, 2-cm, flesh-colored mass on the right buttock intergluteal fold that was soft, compressible, and nontender (Figure 1). There was no capsule, nodule, loculation, or sinus tract. The patient underwent excisional resection with findings of benign-appearing unencapsulated adipose tissue (Figure 2). The wound was closed without difficulty. After several weeks, she had a well-healing scar without contour deficits of the buttocks. Two to 3 months after the initial repair, the patient presented to the family medicine clinic with recurrence of the fatty protrusion. She was referred for consultation and definitive management to a plastic surgeon but was lost to follow up.

In a systematic review of the literature to research pathogenesis theories, a PubMed search of articles indexed for MEDLINE using the terms trauma and pseudolipoma, lipoma, fat, or adipose yielded 45 citations, with only 10 publications addressing the pathology specific to pseudolipomas. Two leading theories of the pathogenesis of PTLs include the adipose herniation pathway and the inflammatory proliferation pathway.4,5

Adipose tissue comprises fat lobules that are organized underneath the supportive elastic fascial layers. Injury from forces exceeding the fascial strength is the basis for the oldest pathogenesis theory. The adipose herniation theory suggests that fat lobules are displaced through the damaged septae, allowing for the development of an epidermal pseudolipoma at the site of blunt trauma.7 This theory has been supported by many case reports; however, more recent reports have identified a larger number of PTL cases that showed no identifiable disruptions in the fascia.1,4,8
In 1997, the inflammatory proliferation theory began to gain attention. The theory describes how local tissue trauma leads to the release of inflammatory cytokines, which successively signals the development of preadipocytes or adipose tissue–derived stem cells (ASCs) into mature adipocytes.4 Most patients report a history of a hematoma in the area of pseudolipoma development, which strongly supports this newer theory. Studies exploring hematomas have found elevated levels of growth factors and inflammatory markers.2,9 In particular, tumor necrosis factor α, peroxisome proliferator–activated receptor γ, vascular endothelial growth factor, and IL-6 and IL-8 may foster an environment in which adipogenic cells are both chemotaxed to the area of trauma and differentiated to white adipose tissue.2,10
Despite addressing the role of the preadipocyte, the available research fails to address the general development of mesenchymal cells into the preadipocyte. White adipose tissue develops at sites of neovascularization and frequently has been observed spreading into the nearby tissue toward other blood vessels. Furthermore, these white adipose tissue expansions remain reliant on multiple growth factors and cell-signaling molecules.10 Numerous investigations into stem cell grafting have found that implantation of ASCs in vivo within animal models does not result in the proliferation and differentiation of ASCs unless specific conditions have been met such as prior tissue injury or immunodeficiency.10-12 These investigations support and expand on the inflammatory proliferation pathway. Thus, most of the true PTLs in the available research appear as de novo tumors and are more congruent with the inflammatory proliferation model.1,2,4-6,8
Typical treatment of a PTL is surgical excision or liposuction depending on the pathology and size of the pseudolipoma. Biopsy examination prior to liposuction is critical for evaluation of liposarcoma and may help identify damage to Scarpa fascia. Recurrence of a PTL is rare regardless of treatment method; however, in a study of 31 PTL cases, only 6 were pathologically identified as PTLs without fibrous material.1
Our patient experienced a blunt trauma to the buttocks and subsequently developed a PTL that was surgically excised and recurred within 3 months. Research surrounding the pathogenesis of the PTL has evolved from the theory of physical herniation of adipose tissue to an inflammatory differentiation of preadipocytes, but there is still much to learn about how and why it occurs and the mesenchymal differentiation following tissue injury.
To the Editor:
The posttraumatic pseudolipoma (PTL) is a painless localized mass comprised of unencapsulated adipose tissue that develops at the site of acute or prolonged blunt soft tissue trauma. It may be round or fusiform in shape and has areas of saponification leading to fat necrosis.1 Posttraumatic pseudolipomas are 12 times more likely to occur in females, which may be attributed to sex-determined adipose tissue distribution or cosmetic concerns.2 Most PTLs are found in areas of the body with high adiposity, including the hip, thigh, and gluteal regions.3 A patient history of a traumatic event resulting in a hematoma and a subsequent latent period of several months to years before the pseudolipoma formation occurs is common.1,2,4-6
A 27-year-old woman presented to the family medicine clinic for examination of a deformity on the right buttock. She noticed a soft protruding mass months after landing on the buttocks and on top of a stick during routine physical training. Prior ultrasonography of the deformity proved unhelpful in determining the etiology. Physical examination revealed a protruding, 2-cm, flesh-colored mass on the right buttock intergluteal fold that was soft, compressible, and nontender (Figure 1). There was no capsule, nodule, loculation, or sinus tract. The patient underwent excisional resection with findings of benign-appearing unencapsulated adipose tissue (Figure 2). The wound was closed without difficulty. After several weeks, she had a well-healing scar without contour deficits of the buttocks. Two to 3 months after the initial repair, the patient presented to the family medicine clinic with recurrence of the fatty protrusion. She was referred for consultation and definitive management to a plastic surgeon but was lost to follow up.

In a systematic review of the literature to research pathogenesis theories, a PubMed search of articles indexed for MEDLINE using the terms trauma and pseudolipoma, lipoma, fat, or adipose yielded 45 citations, with only 10 publications addressing the pathology specific to pseudolipomas. Two leading theories of the pathogenesis of PTLs include the adipose herniation pathway and the inflammatory proliferation pathway.4,5

Adipose tissue comprises fat lobules that are organized underneath the supportive elastic fascial layers. Injury from forces exceeding the fascial strength is the basis for the oldest pathogenesis theory. The adipose herniation theory suggests that fat lobules are displaced through the damaged septae, allowing for the development of an epidermal pseudolipoma at the site of blunt trauma.7 This theory has been supported by many case reports; however, more recent reports have identified a larger number of PTL cases that showed no identifiable disruptions in the fascia.1,4,8
In 1997, the inflammatory proliferation theory began to gain attention. The theory describes how local tissue trauma leads to the release of inflammatory cytokines, which successively signals the development of preadipocytes or adipose tissue–derived stem cells (ASCs) into mature adipocytes.4 Most patients report a history of a hematoma in the area of pseudolipoma development, which strongly supports this newer theory. Studies exploring hematomas have found elevated levels of growth factors and inflammatory markers.2,9 In particular, tumor necrosis factor α, peroxisome proliferator–activated receptor γ, vascular endothelial growth factor, and IL-6 and IL-8 may foster an environment in which adipogenic cells are both chemotaxed to the area of trauma and differentiated to white adipose tissue.2,10
Despite addressing the role of the preadipocyte, the available research fails to address the general development of mesenchymal cells into the preadipocyte. White adipose tissue develops at sites of neovascularization and frequently has been observed spreading into the nearby tissue toward other blood vessels. Furthermore, these white adipose tissue expansions remain reliant on multiple growth factors and cell-signaling molecules.10 Numerous investigations into stem cell grafting have found that implantation of ASCs in vivo within animal models does not result in the proliferation and differentiation of ASCs unless specific conditions have been met such as prior tissue injury or immunodeficiency.10-12 These investigations support and expand on the inflammatory proliferation pathway. Thus, most of the true PTLs in the available research appear as de novo tumors and are more congruent with the inflammatory proliferation model.1,2,4-6,8
Typical treatment of a PTL is surgical excision or liposuction depending on the pathology and size of the pseudolipoma. Biopsy examination prior to liposuction is critical for evaluation of liposarcoma and may help identify damage to Scarpa fascia. Recurrence of a PTL is rare regardless of treatment method; however, in a study of 31 PTL cases, only 6 were pathologically identified as PTLs without fibrous material.1
Our patient experienced a blunt trauma to the buttocks and subsequently developed a PTL that was surgically excised and recurred within 3 months. Research surrounding the pathogenesis of the PTL has evolved from the theory of physical herniation of adipose tissue to an inflammatory differentiation of preadipocytes, but there is still much to learn about how and why it occurs and the mesenchymal differentiation following tissue injury.
- Aust MC, Spies M, Kall S, et al. Lipomas after blunt soft tissue trauma: are they real? analysis of 31 cases. Br J Dermatol. 2007;157:92-99. doi:10.1111/j.1365-2133.2007.07970.x
- Galea LA, Penington AJ, Morrison WA. Post-traumatic pseudolipomas—a review and postulated mechanisms of their development. J Plast Reconstr Aesthet Surg. 2009;62:737-741. doi:10.1016/j.bjps.2008.12.021
- Zajac JC, Mandelbaum M, Economides JM, et al. Immediate massive posttraumatic pseudolipoma of the buttocks: a case of a heterotopic “love handle.” Plast Reconstr Surg Glob Open. 2018;6:E1887. doi:10.1097/GOX.0000000000001887
- Signorini M, Campiglio GL. Posttraumatic lipomas: where do they really come from? Plast Reconstr Surg. 1998;101:699-705. doi:10.1097/00006534-199803000-00017
- Khadilkar AS, Goyal A, Gauba K. The enigma of “traumatic pseudolipoma” and “traumatic herniation of buccal fat pad”: a systematic review and new classification system of post-traumatic craniofacial fatty masses. J Oral Maxillofac Surg. 2018;76:1267-1278. doi:10.1016/j.joms.2017.01.024
- Copcu E, Sivrioglu NS. Posttraumatic lipoma: analysis of 10 cases and explanation of possible mechanisms. Dermatol Surg. 2003;29:215-220. doi:10.1046/j.1524-4725.2003.29052.x
- Penoff JH. Traumatic lipomas/pseudolipomas. J Trauma. 1982;22:63-65. doi:10.1097/00005373-198201000-00013
- Theumann N, Abdelmoumene A, Wintermark M, et al. Posttraumatic pseudolipoma: MRI appearances. Eur Radiol. 2005;15:1876-1880. doi:10.1007/s00330-005-2757-2
- David LR, DeFranzo A, Marks M, et al. Posttraumatic pseudolipoma. J Trauma. 1996;40:396-400. doi:10.1097/00005373-199603000-00012
- Cawthorn WP, Scheller EL, MacDougald OA. Adipose tissue stem cells meet preadipocyte commitment: going back to the future. J Lipid Res. 2012;53:227-246. doi:10.1194/jlr.R021089
- Joe AW, Yi L, Natarajan A, et al. Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat Cell Biol. 2010;12:153-163. doi:10.1038/ncb2015
- Miranville A, Heeschen C, Sengenès C, et al. Improvement of postnatal neovascularization by human adipose tissue-derived stem cells. Circulation. 2004;110:349-355. doi:10.1161/01.Cir.0000135466.16823.D0
- Aust MC, Spies M, Kall S, et al. Lipomas after blunt soft tissue trauma: are they real? analysis of 31 cases. Br J Dermatol. 2007;157:92-99. doi:10.1111/j.1365-2133.2007.07970.x
- Galea LA, Penington AJ, Morrison WA. Post-traumatic pseudolipomas—a review and postulated mechanisms of their development. J Plast Reconstr Aesthet Surg. 2009;62:737-741. doi:10.1016/j.bjps.2008.12.021
- Zajac JC, Mandelbaum M, Economides JM, et al. Immediate massive posttraumatic pseudolipoma of the buttocks: a case of a heterotopic “love handle.” Plast Reconstr Surg Glob Open. 2018;6:E1887. doi:10.1097/GOX.0000000000001887
- Signorini M, Campiglio GL. Posttraumatic lipomas: where do they really come from? Plast Reconstr Surg. 1998;101:699-705. doi:10.1097/00006534-199803000-00017
- Khadilkar AS, Goyal A, Gauba K. The enigma of “traumatic pseudolipoma” and “traumatic herniation of buccal fat pad”: a systematic review and new classification system of post-traumatic craniofacial fatty masses. J Oral Maxillofac Surg. 2018;76:1267-1278. doi:10.1016/j.joms.2017.01.024
- Copcu E, Sivrioglu NS. Posttraumatic lipoma: analysis of 10 cases and explanation of possible mechanisms. Dermatol Surg. 2003;29:215-220. doi:10.1046/j.1524-4725.2003.29052.x
- Penoff JH. Traumatic lipomas/pseudolipomas. J Trauma. 1982;22:63-65. doi:10.1097/00005373-198201000-00013
- Theumann N, Abdelmoumene A, Wintermark M, et al. Posttraumatic pseudolipoma: MRI appearances. Eur Radiol. 2005;15:1876-1880. doi:10.1007/s00330-005-2757-2
- David LR, DeFranzo A, Marks M, et al. Posttraumatic pseudolipoma. J Trauma. 1996;40:396-400. doi:10.1097/00005373-199603000-00012
- Cawthorn WP, Scheller EL, MacDougald OA. Adipose tissue stem cells meet preadipocyte commitment: going back to the future. J Lipid Res. 2012;53:227-246. doi:10.1194/jlr.R021089
- Joe AW, Yi L, Natarajan A, et al. Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat Cell Biol. 2010;12:153-163. doi:10.1038/ncb2015
- Miranville A, Heeschen C, Sengenès C, et al. Improvement of postnatal neovascularization by human adipose tissue-derived stem cells. Circulation. 2004;110:349-355. doi:10.1161/01.Cir.0000135466.16823.D0
Practice Points
- Physicians should include pseudolipoma in the differential diagnosis when evaluating masses that develop in patients at sites of blunt or prolonged trauma.
- A pseudolipoma is an unencapsulated, round, or fusiform fatty mass that differs from a traditional lipoma by the absence of a capsule.
- Further research may elucidate the pathogenesis of these adiposities.
Nurse practitioner fined $20k for advertising herself as ‘Doctor Sarah’
Last month, the San Luis Obispo County, California, District Attorney Dan Dow filed a complaint against Sarah Erny, RN, NP, citing unfair business practices and unprofessional conduct.
According to court documents, California’s Medical Practice Act does not permit individuals to refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
In addition to the fine, Ms. Erny agreed to refrain from referring to herself as a doctor in her practice and on social media. She has already deleted her Twitter account.
The case underscores tensions between physicians fighting to preserve their scope of practice and the allied professionals that U.S. lawmakers increasingly see as a less expensive way to improve access to health care.
The American Medical Association and specialty groups strongly oppose a new bill, the Improving Care and Access to Nurses Act, that would expand the scope of practice for nurse practitioners and physician assistants.
Court records show that Ms. Erny earned a doctor of nursing practice (DNP) degree from Vanderbilt University, Nashville, Tenn., and that she met the state requirements to obtain licensure as a registered nurse and nurse practitioner. In 2018, she opened a practice in Arroyo Grande, California, called Holistic Women’s Healing, where she provided medical services and drug supplements to patients.
She also entered a collaborative agreement with ob.gyn. Anika Moore, MD, for approximately 3 years. Dr. Moore’s medical practice was in another county and state, and the physician returned every 2 to 3 months to review a portion of Ms. Erny’s patient files.
Ms. Erny and Dr. Moore terminated the collaborative agreement in March, according to court documents.
However, Mr. Dow alleged that Ms. Erny regularly referred to herself as “Dr. Sarah” or “Dr. Sarah Erny” in her online advertising and social media accounts. Her patients “were so proud of her” that they called her doctor, and her supervising physician instructed staff to do the same.
Mr. Dow said Ms. Erny did not clearly advise the public that she was not a medical doctor and failed to identify her supervising physician. “Simply put, there is a great need for health care providers to state their level of training and licensing clearly and honestly in all of their advertising and marketing materials,” he said in a press release.
In California, nurse practitioners who have been certified by the Board of Registered Nursing may use the following titles: Advanced Practice Registered Nurse; Certified Nurse Practitioner; APRN-CNP; RN and NP; or a combination of other letters or words to identify specialization, such as adult nurse practitioner, pediatric nurse practitioner, obstetrical-gynecological nurse practitioner, and family nurse practitioner.
As educational requirements shift for advanced practice clinicians, similar cases will likely emerge, said Grant Martsolf, PhD, MPH, RN, FAAN, professor at the University of Pittsburgh School of Nursing.
“Scope of practice is governed by states, [so they] will have to figure [it] out as more professional disciplines move to clinical doctorates as the entry to practice. Pharma, [physical therapy], and [occupational therapy] have already done this, and advanced practice nursing is on its way. [Certified registered nurse anesthetists] are already required to get a DNP to sit for certification,” he said.
More guidance is needed, especially when considering other professions like dentists, clinical psychologists, and individuals with clinical or research doctorates who often call themselves doctors, Dr. Martsolf said.
“It seems that the honorific of ‘Dr.’ emerges from the degree, not from being a physician or surgeon,” he said.
Beyond the false advertising, Mr. Dow alleged that Ms. Erny did not file a fictitious business name statement for 2020 and 2021 – a requirement under the California Business and Professions Code to identify who is operating the business.
A version of this article first appeared on Medscape.com.
Last month, the San Luis Obispo County, California, District Attorney Dan Dow filed a complaint against Sarah Erny, RN, NP, citing unfair business practices and unprofessional conduct.
According to court documents, California’s Medical Practice Act does not permit individuals to refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
In addition to the fine, Ms. Erny agreed to refrain from referring to herself as a doctor in her practice and on social media. She has already deleted her Twitter account.
The case underscores tensions between physicians fighting to preserve their scope of practice and the allied professionals that U.S. lawmakers increasingly see as a less expensive way to improve access to health care.
The American Medical Association and specialty groups strongly oppose a new bill, the Improving Care and Access to Nurses Act, that would expand the scope of practice for nurse practitioners and physician assistants.
Court records show that Ms. Erny earned a doctor of nursing practice (DNP) degree from Vanderbilt University, Nashville, Tenn., and that she met the state requirements to obtain licensure as a registered nurse and nurse practitioner. In 2018, she opened a practice in Arroyo Grande, California, called Holistic Women’s Healing, where she provided medical services and drug supplements to patients.
She also entered a collaborative agreement with ob.gyn. Anika Moore, MD, for approximately 3 years. Dr. Moore’s medical practice was in another county and state, and the physician returned every 2 to 3 months to review a portion of Ms. Erny’s patient files.
Ms. Erny and Dr. Moore terminated the collaborative agreement in March, according to court documents.
However, Mr. Dow alleged that Ms. Erny regularly referred to herself as “Dr. Sarah” or “Dr. Sarah Erny” in her online advertising and social media accounts. Her patients “were so proud of her” that they called her doctor, and her supervising physician instructed staff to do the same.
Mr. Dow said Ms. Erny did not clearly advise the public that she was not a medical doctor and failed to identify her supervising physician. “Simply put, there is a great need for health care providers to state their level of training and licensing clearly and honestly in all of their advertising and marketing materials,” he said in a press release.
In California, nurse practitioners who have been certified by the Board of Registered Nursing may use the following titles: Advanced Practice Registered Nurse; Certified Nurse Practitioner; APRN-CNP; RN and NP; or a combination of other letters or words to identify specialization, such as adult nurse practitioner, pediatric nurse practitioner, obstetrical-gynecological nurse practitioner, and family nurse practitioner.
As educational requirements shift for advanced practice clinicians, similar cases will likely emerge, said Grant Martsolf, PhD, MPH, RN, FAAN, professor at the University of Pittsburgh School of Nursing.
“Scope of practice is governed by states, [so they] will have to figure [it] out as more professional disciplines move to clinical doctorates as the entry to practice. Pharma, [physical therapy], and [occupational therapy] have already done this, and advanced practice nursing is on its way. [Certified registered nurse anesthetists] are already required to get a DNP to sit for certification,” he said.
More guidance is needed, especially when considering other professions like dentists, clinical psychologists, and individuals with clinical or research doctorates who often call themselves doctors, Dr. Martsolf said.
“It seems that the honorific of ‘Dr.’ emerges from the degree, not from being a physician or surgeon,” he said.
Beyond the false advertising, Mr. Dow alleged that Ms. Erny did not file a fictitious business name statement for 2020 and 2021 – a requirement under the California Business and Professions Code to identify who is operating the business.
A version of this article first appeared on Medscape.com.
Last month, the San Luis Obispo County, California, District Attorney Dan Dow filed a complaint against Sarah Erny, RN, NP, citing unfair business practices and unprofessional conduct.
According to court documents, California’s Medical Practice Act does not permit individuals to refer to themselves as “doctor, physician, or any other terms or letters indicating or implying that he or she is a physician and surgeon ... without having ... a certificate as a physician and surgeon.”
Individuals who misrepresent themselves are subject to misdemeanor charges and civil penalties.
In addition to the fine, Ms. Erny agreed to refrain from referring to herself as a doctor in her practice and on social media. She has already deleted her Twitter account.
The case underscores tensions between physicians fighting to preserve their scope of practice and the allied professionals that U.S. lawmakers increasingly see as a less expensive way to improve access to health care.
The American Medical Association and specialty groups strongly oppose a new bill, the Improving Care and Access to Nurses Act, that would expand the scope of practice for nurse practitioners and physician assistants.
Court records show that Ms. Erny earned a doctor of nursing practice (DNP) degree from Vanderbilt University, Nashville, Tenn., and that she met the state requirements to obtain licensure as a registered nurse and nurse practitioner. In 2018, she opened a practice in Arroyo Grande, California, called Holistic Women’s Healing, where she provided medical services and drug supplements to patients.
She also entered a collaborative agreement with ob.gyn. Anika Moore, MD, for approximately 3 years. Dr. Moore’s medical practice was in another county and state, and the physician returned every 2 to 3 months to review a portion of Ms. Erny’s patient files.
Ms. Erny and Dr. Moore terminated the collaborative agreement in March, according to court documents.
However, Mr. Dow alleged that Ms. Erny regularly referred to herself as “Dr. Sarah” or “Dr. Sarah Erny” in her online advertising and social media accounts. Her patients “were so proud of her” that they called her doctor, and her supervising physician instructed staff to do the same.
Mr. Dow said Ms. Erny did not clearly advise the public that she was not a medical doctor and failed to identify her supervising physician. “Simply put, there is a great need for health care providers to state their level of training and licensing clearly and honestly in all of their advertising and marketing materials,” he said in a press release.
In California, nurse practitioners who have been certified by the Board of Registered Nursing may use the following titles: Advanced Practice Registered Nurse; Certified Nurse Practitioner; APRN-CNP; RN and NP; or a combination of other letters or words to identify specialization, such as adult nurse practitioner, pediatric nurse practitioner, obstetrical-gynecological nurse practitioner, and family nurse practitioner.
As educational requirements shift for advanced practice clinicians, similar cases will likely emerge, said Grant Martsolf, PhD, MPH, RN, FAAN, professor at the University of Pittsburgh School of Nursing.
“Scope of practice is governed by states, [so they] will have to figure [it] out as more professional disciplines move to clinical doctorates as the entry to practice. Pharma, [physical therapy], and [occupational therapy] have already done this, and advanced practice nursing is on its way. [Certified registered nurse anesthetists] are already required to get a DNP to sit for certification,” he said.
More guidance is needed, especially when considering other professions like dentists, clinical psychologists, and individuals with clinical or research doctorates who often call themselves doctors, Dr. Martsolf said.
“It seems that the honorific of ‘Dr.’ emerges from the degree, not from being a physician or surgeon,” he said.
Beyond the false advertising, Mr. Dow alleged that Ms. Erny did not file a fictitious business name statement for 2020 and 2021 – a requirement under the California Business and Professions Code to identify who is operating the business.
A version of this article first appeared on Medscape.com.
Diffuse Papular Eruption With Erosions and Ulcerations
The Diagnosis: Immunotherapy-Related Lichenoid Drug Eruption
Direct immunofluorescence was negative, and histopathology revealed a lichenoid interface dermatitis, minimal parakeratosis, and saw-toothed rete ridges (Figure 1). He was diagnosed with an immunotherapyrelated lichenoid drug eruption based on the morphology of the skin lesions and clinicopathologic correlation. Bullous pemphigoid and lichen planus pemphigoides were ruled out given the negative direct immunofluorescence findings. Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) was not consistent with the clinical presentation, especially given the lack of mucosal findings. The histology also was not consistent, as the biopsy specimen lacked apoptotic and necrotic keratinocytes to the degree seen in SJS/TEN and also had a greater degree of inflammatory infiltrate. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome was ruled out given the lack of systemic findings, including facial swelling and lymphadenopathy and the clinical appearance of the rash. No morbilliform features were present, which is the most common presentation of DRESS syndrome.
Checkpoint inhibitor (CPI) therapy has become the cornerstone in management of certain advanced malignancies.1 Checkpoint inhibitors block cytotoxic T lymphocyte–associated protein 4, programmed cell death-1, and/or programmed cell death ligand-1, allowing activated T cells to infiltrate the tumor microenvironment and destroy malignant cells. Checkpoint inhibitors are approved for the treatment of melanoma, cutaneous squamous cell carcinoma, and Merkel cell carcinoma and are being investigated in various other cutaneous and soft tissue malignancies.1-3
Although CPIs have shown substantial efficacy in the management of advanced malignancies, immune-related adverse events (AEs) are common due to nonspecific immune activation.2 Immune-related cutaneous AEs are the most common immune-related AEs, occurring in 30% to 50% of patients who undergo treatment.2-5 Common immune-related cutaneous AEs include maculopapular, psoriasiform, and lichenoid dermatitis, as well as pruritus without dermatitis.2,3,6 Other reactions include but are not limited to bullous pemphigoid, vitiligolike depigmentation, and alopecia.2,3 Immune-related cutaneous AEs usually are self-limited; however, severe life-threatening reactions such as the spectrum of SJS/TEN and DRESS syndrome also can occur.2-4 Immune-related cutaneous AEs are graded based on the Common Terminology Criteria for Adverse Events: grade 1 reactions are asymptomatic and cover less than 10% of the patient’s body surface area (BSA), grade 2 reactions have mild symptoms and cover 10% to 30% of the patient’s BSA, grade 3 reactions have moderate to severe symptoms and cover greater than 30% of the patient’s BSA, and grade 4 reactions are life-threatening.2,3 With prompt recognition and adequate treatment, mild to moderate immune-related cutaneous AEs—grades 1 and 2—largely are reversible, and less than 5% require discontinuation of therapy.2,3,6 It has been suggested that immune-related cutaneous AEs may be a positive prognostic factor in the treatment of underlying malignancy, indicating adequate immune activation targeting the malignant cells.6
Although our patient had some typical violaceous, flat-topped papules and plaques with Wickham striae, he also had atypical findings for a lichenoid reaction. Given the endorsement of blisters, it is possible that some of these lesions initially were bullous and subsequently ruptured, leaving behind erosions. However, in other areas, there also were eroded papules and ulcerations without a reported history of excoriation, scratching, picking, or prior bullae, including difficult-to-reach areas such as the back. It is favored that these lesions represented a robust lichenoid dermatitis leading to erosive and ulcerated lesions, similar to the formation of bullous lichen planus. Lichenoid eruptions secondary to immunotherapy are well-known phenomena, but a PubMed search of articles indexed for MEDLINE using the terms ulcer, lichenoid, and immunotherapy revealed only 2 cases of ulcerative lichenoid eruptions: a localized digital erosive lichenoid dermatitis and a widespread ulcerative lichenoid drug eruption without true erosions.7,8 However, widespread erosive and ulcerated lichenoid reactions are rare.
Lichenoid eruptions most strongly are associated with anti–programmed cell death-1/ programmed cell death ligand-1 therapy, occurring in 20% of patients undergoing treatment.3 Lichenoid eruptions present as discrete, pruritic, erythematous, violaceous papules and plaques on the chest and back and rarely may involve the limbs, palmoplantar surfaces, and oral mucosa.2,3,6 Histopathologic features include a dense bandlike lymphocytic infiltrate in the dermis with scattered apoptotic keratinocytes in the basal layer of the epidermis.2,4,6 Grades 1 to 2 lesions can be managed with high-potency topical corticosteroids without CPI dose interruption, with more extensive grade 2 lesions requiring systemic corticosteroids.2,6,9 Lichenoid eruptions grade 3 or higher also require systemic corticosteroid therapy CPI therapy cessation until the eruption has receded to grade 0 to 1.2 Alternative treatment options for high-grade toxicity include phototherapy and acitretin.2,4,9
Our patient was treated with cessation of immunotherapy and initiation of a systemic corticosteroid taper, acitretin, and narrowband UVB therapy. After 6 weeks of treatment, the pain and pruritus improved and the rash had resolved in some areas while it had taken on a more classic lichenoid appearance with violaceous scaly papules and plaques (Figure 2) in areas of prior ulcers and erosions. He no longer had any bullae, erosions, or ulcers.
- Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83:1239-1253. doi:10.1016/j.jaad.2020.03.131
- Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83:1255-1268. doi:10.1016/j.jaad.2020.03.132
- Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93:123-132.
- Si X, He C, Zhang L, et al. Management of immune checkpoint inhibitor-related dermatologic adverse events. Thorac Cancer. 2020;11:488-492. doi:10.1111/1759-7714.13275
- Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020;6:519-527. doi:10.1001 /jamaoncol.2019.5570
- Sibaud V, Meyer N, Lamant L, et al. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28:254-263. doi:10.1097/CCO.0000000000000290
- Martínez-Doménech Á, García-Legaz Martínez M, Magdaleno-Tapial J, et al. Digital ulcerative lichenoid dermatitis in a patient receiving anti-PD-1 therapy. Dermatol Online J. 2019;25:13030/qt8sm0j7t7.
- Davis MJ, Wilken R, Fung MA, et al. Debilitating erosive lichenoid interface dermatitis from checkpoint inhibitor therapy. Dermatol Online J. 2018;24:13030/qt3vq6b04v.
- Apalla Z, Papageorgiou C, Lallas A, et al. Cutaneous adverse events of immune checkpoint inhibitors: a literature review [published online January 29, 2021]. Dermatol Pract Concept. 2021;11:E2021155. doi:10.5826/dpc.1101a155
The Diagnosis: Immunotherapy-Related Lichenoid Drug Eruption
Direct immunofluorescence was negative, and histopathology revealed a lichenoid interface dermatitis, minimal parakeratosis, and saw-toothed rete ridges (Figure 1). He was diagnosed with an immunotherapyrelated lichenoid drug eruption based on the morphology of the skin lesions and clinicopathologic correlation. Bullous pemphigoid and lichen planus pemphigoides were ruled out given the negative direct immunofluorescence findings. Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) was not consistent with the clinical presentation, especially given the lack of mucosal findings. The histology also was not consistent, as the biopsy specimen lacked apoptotic and necrotic keratinocytes to the degree seen in SJS/TEN and also had a greater degree of inflammatory infiltrate. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome was ruled out given the lack of systemic findings, including facial swelling and lymphadenopathy and the clinical appearance of the rash. No morbilliform features were present, which is the most common presentation of DRESS syndrome.

Checkpoint inhibitor (CPI) therapy has become the cornerstone in management of certain advanced malignancies.1 Checkpoint inhibitors block cytotoxic T lymphocyte–associated protein 4, programmed cell death-1, and/or programmed cell death ligand-1, allowing activated T cells to infiltrate the tumor microenvironment and destroy malignant cells. Checkpoint inhibitors are approved for the treatment of melanoma, cutaneous squamous cell carcinoma, and Merkel cell carcinoma and are being investigated in various other cutaneous and soft tissue malignancies.1-3
Although CPIs have shown substantial efficacy in the management of advanced malignancies, immune-related adverse events (AEs) are common due to nonspecific immune activation.2 Immune-related cutaneous AEs are the most common immune-related AEs, occurring in 30% to 50% of patients who undergo treatment.2-5 Common immune-related cutaneous AEs include maculopapular, psoriasiform, and lichenoid dermatitis, as well as pruritus without dermatitis.2,3,6 Other reactions include but are not limited to bullous pemphigoid, vitiligolike depigmentation, and alopecia.2,3 Immune-related cutaneous AEs usually are self-limited; however, severe life-threatening reactions such as the spectrum of SJS/TEN and DRESS syndrome also can occur.2-4 Immune-related cutaneous AEs are graded based on the Common Terminology Criteria for Adverse Events: grade 1 reactions are asymptomatic and cover less than 10% of the patient’s body surface area (BSA), grade 2 reactions have mild symptoms and cover 10% to 30% of the patient’s BSA, grade 3 reactions have moderate to severe symptoms and cover greater than 30% of the patient’s BSA, and grade 4 reactions are life-threatening.2,3 With prompt recognition and adequate treatment, mild to moderate immune-related cutaneous AEs—grades 1 and 2—largely are reversible, and less than 5% require discontinuation of therapy.2,3,6 It has been suggested that immune-related cutaneous AEs may be a positive prognostic factor in the treatment of underlying malignancy, indicating adequate immune activation targeting the malignant cells.6
Although our patient had some typical violaceous, flat-topped papules and plaques with Wickham striae, he also had atypical findings for a lichenoid reaction. Given the endorsement of blisters, it is possible that some of these lesions initially were bullous and subsequently ruptured, leaving behind erosions. However, in other areas, there also were eroded papules and ulcerations without a reported history of excoriation, scratching, picking, or prior bullae, including difficult-to-reach areas such as the back. It is favored that these lesions represented a robust lichenoid dermatitis leading to erosive and ulcerated lesions, similar to the formation of bullous lichen planus. Lichenoid eruptions secondary to immunotherapy are well-known phenomena, but a PubMed search of articles indexed for MEDLINE using the terms ulcer, lichenoid, and immunotherapy revealed only 2 cases of ulcerative lichenoid eruptions: a localized digital erosive lichenoid dermatitis and a widespread ulcerative lichenoid drug eruption without true erosions.7,8 However, widespread erosive and ulcerated lichenoid reactions are rare.
Lichenoid eruptions most strongly are associated with anti–programmed cell death-1/ programmed cell death ligand-1 therapy, occurring in 20% of patients undergoing treatment.3 Lichenoid eruptions present as discrete, pruritic, erythematous, violaceous papules and plaques on the chest and back and rarely may involve the limbs, palmoplantar surfaces, and oral mucosa.2,3,6 Histopathologic features include a dense bandlike lymphocytic infiltrate in the dermis with scattered apoptotic keratinocytes in the basal layer of the epidermis.2,4,6 Grades 1 to 2 lesions can be managed with high-potency topical corticosteroids without CPI dose interruption, with more extensive grade 2 lesions requiring systemic corticosteroids.2,6,9 Lichenoid eruptions grade 3 or higher also require systemic corticosteroid therapy CPI therapy cessation until the eruption has receded to grade 0 to 1.2 Alternative treatment options for high-grade toxicity include phototherapy and acitretin.2,4,9
Our patient was treated with cessation of immunotherapy and initiation of a systemic corticosteroid taper, acitretin, and narrowband UVB therapy. After 6 weeks of treatment, the pain and pruritus improved and the rash had resolved in some areas while it had taken on a more classic lichenoid appearance with violaceous scaly papules and plaques (Figure 2) in areas of prior ulcers and erosions. He no longer had any bullae, erosions, or ulcers.

The Diagnosis: Immunotherapy-Related Lichenoid Drug Eruption
Direct immunofluorescence was negative, and histopathology revealed a lichenoid interface dermatitis, minimal parakeratosis, and saw-toothed rete ridges (Figure 1). He was diagnosed with an immunotherapyrelated lichenoid drug eruption based on the morphology of the skin lesions and clinicopathologic correlation. Bullous pemphigoid and lichen planus pemphigoides were ruled out given the negative direct immunofluorescence findings. Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) was not consistent with the clinical presentation, especially given the lack of mucosal findings. The histology also was not consistent, as the biopsy specimen lacked apoptotic and necrotic keratinocytes to the degree seen in SJS/TEN and also had a greater degree of inflammatory infiltrate. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome was ruled out given the lack of systemic findings, including facial swelling and lymphadenopathy and the clinical appearance of the rash. No morbilliform features were present, which is the most common presentation of DRESS syndrome.

Checkpoint inhibitor (CPI) therapy has become the cornerstone in management of certain advanced malignancies.1 Checkpoint inhibitors block cytotoxic T lymphocyte–associated protein 4, programmed cell death-1, and/or programmed cell death ligand-1, allowing activated T cells to infiltrate the tumor microenvironment and destroy malignant cells. Checkpoint inhibitors are approved for the treatment of melanoma, cutaneous squamous cell carcinoma, and Merkel cell carcinoma and are being investigated in various other cutaneous and soft tissue malignancies.1-3
Although CPIs have shown substantial efficacy in the management of advanced malignancies, immune-related adverse events (AEs) are common due to nonspecific immune activation.2 Immune-related cutaneous AEs are the most common immune-related AEs, occurring in 30% to 50% of patients who undergo treatment.2-5 Common immune-related cutaneous AEs include maculopapular, psoriasiform, and lichenoid dermatitis, as well as pruritus without dermatitis.2,3,6 Other reactions include but are not limited to bullous pemphigoid, vitiligolike depigmentation, and alopecia.2,3 Immune-related cutaneous AEs usually are self-limited; however, severe life-threatening reactions such as the spectrum of SJS/TEN and DRESS syndrome also can occur.2-4 Immune-related cutaneous AEs are graded based on the Common Terminology Criteria for Adverse Events: grade 1 reactions are asymptomatic and cover less than 10% of the patient’s body surface area (BSA), grade 2 reactions have mild symptoms and cover 10% to 30% of the patient’s BSA, grade 3 reactions have moderate to severe symptoms and cover greater than 30% of the patient’s BSA, and grade 4 reactions are life-threatening.2,3 With prompt recognition and adequate treatment, mild to moderate immune-related cutaneous AEs—grades 1 and 2—largely are reversible, and less than 5% require discontinuation of therapy.2,3,6 It has been suggested that immune-related cutaneous AEs may be a positive prognostic factor in the treatment of underlying malignancy, indicating adequate immune activation targeting the malignant cells.6
Although our patient had some typical violaceous, flat-topped papules and plaques with Wickham striae, he also had atypical findings for a lichenoid reaction. Given the endorsement of blisters, it is possible that some of these lesions initially were bullous and subsequently ruptured, leaving behind erosions. However, in other areas, there also were eroded papules and ulcerations without a reported history of excoriation, scratching, picking, or prior bullae, including difficult-to-reach areas such as the back. It is favored that these lesions represented a robust lichenoid dermatitis leading to erosive and ulcerated lesions, similar to the formation of bullous lichen planus. Lichenoid eruptions secondary to immunotherapy are well-known phenomena, but a PubMed search of articles indexed for MEDLINE using the terms ulcer, lichenoid, and immunotherapy revealed only 2 cases of ulcerative lichenoid eruptions: a localized digital erosive lichenoid dermatitis and a widespread ulcerative lichenoid drug eruption without true erosions.7,8 However, widespread erosive and ulcerated lichenoid reactions are rare.
Lichenoid eruptions most strongly are associated with anti–programmed cell death-1/ programmed cell death ligand-1 therapy, occurring in 20% of patients undergoing treatment.3 Lichenoid eruptions present as discrete, pruritic, erythematous, violaceous papules and plaques on the chest and back and rarely may involve the limbs, palmoplantar surfaces, and oral mucosa.2,3,6 Histopathologic features include a dense bandlike lymphocytic infiltrate in the dermis with scattered apoptotic keratinocytes in the basal layer of the epidermis.2,4,6 Grades 1 to 2 lesions can be managed with high-potency topical corticosteroids without CPI dose interruption, with more extensive grade 2 lesions requiring systemic corticosteroids.2,6,9 Lichenoid eruptions grade 3 or higher also require systemic corticosteroid therapy CPI therapy cessation until the eruption has receded to grade 0 to 1.2 Alternative treatment options for high-grade toxicity include phototherapy and acitretin.2,4,9
Our patient was treated with cessation of immunotherapy and initiation of a systemic corticosteroid taper, acitretin, and narrowband UVB therapy. After 6 weeks of treatment, the pain and pruritus improved and the rash had resolved in some areas while it had taken on a more classic lichenoid appearance with violaceous scaly papules and plaques (Figure 2) in areas of prior ulcers and erosions. He no longer had any bullae, erosions, or ulcers.

- Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83:1239-1253. doi:10.1016/j.jaad.2020.03.131
- Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83:1255-1268. doi:10.1016/j.jaad.2020.03.132
- Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93:123-132.
- Si X, He C, Zhang L, et al. Management of immune checkpoint inhibitor-related dermatologic adverse events. Thorac Cancer. 2020;11:488-492. doi:10.1111/1759-7714.13275
- Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020;6:519-527. doi:10.1001 /jamaoncol.2019.5570
- Sibaud V, Meyer N, Lamant L, et al. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28:254-263. doi:10.1097/CCO.0000000000000290
- Martínez-Doménech Á, García-Legaz Martínez M, Magdaleno-Tapial J, et al. Digital ulcerative lichenoid dermatitis in a patient receiving anti-PD-1 therapy. Dermatol Online J. 2019;25:13030/qt8sm0j7t7.
- Davis MJ, Wilken R, Fung MA, et al. Debilitating erosive lichenoid interface dermatitis from checkpoint inhibitor therapy. Dermatol Online J. 2018;24:13030/qt3vq6b04v.
- Apalla Z, Papageorgiou C, Lallas A, et al. Cutaneous adverse events of immune checkpoint inhibitors: a literature review [published online January 29, 2021]. Dermatol Pract Concept. 2021;11:E2021155. doi:10.5826/dpc.1101a155
- Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83:1239-1253. doi:10.1016/j.jaad.2020.03.131
- Geisler AN, Phillips GS, Barrios DM, et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83:1255-1268. doi:10.1016/j.jaad.2020.03.132
- Tattersall IW, Leventhal JS. Cutaneous toxicities of immune checkpoint inhibitors: the role of the dermatologist. Yale J Biol Med. 2020;93:123-132.
- Si X, He C, Zhang L, et al. Management of immune checkpoint inhibitor-related dermatologic adverse events. Thorac Cancer. 2020;11:488-492. doi:10.1111/1759-7714.13275
- Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2020;6:519-527. doi:10.1001 /jamaoncol.2019.5570
- Sibaud V, Meyer N, Lamant L, et al. Dermatologic complications of anti-PD-1/PD-L1 immune checkpoint antibodies. Curr Opin Oncol. 2016;28:254-263. doi:10.1097/CCO.0000000000000290
- Martínez-Doménech Á, García-Legaz Martínez M, Magdaleno-Tapial J, et al. Digital ulcerative lichenoid dermatitis in a patient receiving anti-PD-1 therapy. Dermatol Online J. 2019;25:13030/qt8sm0j7t7.
- Davis MJ, Wilken R, Fung MA, et al. Debilitating erosive lichenoid interface dermatitis from checkpoint inhibitor therapy. Dermatol Online J. 2018;24:13030/qt3vq6b04v.
- Apalla Z, Papageorgiou C, Lallas A, et al. Cutaneous adverse events of immune checkpoint inhibitors: a literature review [published online January 29, 2021]. Dermatol Pract Concept. 2021;11:E2021155. doi:10.5826/dpc.1101a155
A 70-year-old man presented with a painful, pruritic, diffuse eruption on the trunk, legs, and arms of 2 months’ duration. He had a history of stage IV pleomorphic cell sarcoma of the retroperitoneum and was started on pembrolizumab therapy 6 weeks prior to the eruption. Physical examination revealed violaceous papules and plaques with shiny reticulated scaling as well as multiple scattered eroded papules and shallow ulcerations. The oral mucosa and genitals were spared. The patient endorsed blisters followed by open sores that were both itchy and painful. He denied self-infliction. Both the patient and his wife denied scratching. Two biopsies for direct immunofluorescence and histopathology were performed.
Add tezepelumab to SCIT to improve cat allergy symptoms?
according to results of a phase 1/2 clinical trial.
“One year of allergen immunotherapy [AIT] combined with tezepelumab was significantly more effective than SCIT alone in reducing the nasal response to allergen challenge both at the end of treatment and one year after stopping treatment,” lead study author Jonathan Corren, MD, of the University of California, Los Angeles, and his colleagues wrote in The Journal of Allergy and Clinical Immunology.
“This persistent improvement in clinical response was paralleled by reductions in nasal transcripts for multiple immunologic pathways, including mast cell activation.”
The study was cited in a news release from the National Institutes of Health that said that the approach may work in a similar way with other allergens.
The Food and Drug Administration recently approved tezepelumab for the treatment of severe asthma in people aged 12 years and older. Tezelumab, a monoclonal antibody, works by blocking the cytokine thymic stromal lymphopoietin (TSLP).
“Cells that cover the surface of organs like the skin and intestines or that line the inside of the nose and lungs rapidly secrete TSLP in response to signals of potential danger,” according to the NIH news release. “In allergic disease, TSLP helps initiate an overreactive immune response to otherwise harmless substances like cat dander, provoking airway inflammation that leads to the symptoms of allergic rhinitis.”
Testing an enhanced strategy
The double-blind CATNIP trial was conducted by Dr. Corren and colleagues at nine sites in the United States. The trial included patients aged 18-65 years who’d had moderate to severe cat-induced allergic rhinitis for at least 2 years from 2015 to 2019.
The researchers excluded patients with recurrent acute or chronic sinusitis. They excluded patients who had undergone SCIT with cat allergen within the past 10 years or seasonal or perennial allergen sensitivity during nasal challenges. They also excluded persons with a history of persistent asthma.
In the parallel-design study, 121 participants were randomly allocated into four groups: 32 patients were treated with intravenous tezepelumab plus cat SCIT, 31 received the allergy shots alone, 30 received tezepelumab alone, and 28 received placebo alone for 52 weeks, followed by 52 weeks of observation.
Participants received SCIT (10,000 bioequivalent allergy units per milliliter) or matched placebo via subcutaneous injections weekly in increasing doses for around 12 weeks, followed by monthly maintenance injections (4,000 BAU or maximum tolerated dose) until week 48.
They received tezepelumab (700 mg IV) or matched placebo 1-3 days prior to the SCIT or placebo SCIT injections once every 4 weeks through week 24, then before or on the same day as the SCIT or placebo injections through week 48.
Measures of effectiveness
Participants were also given nasal allergy challenges – one spritz of a nasal spray containing cat allergen extract in each nostril at screening, baseline, and weeks 26, 52, 78, and 104. The researchers recorded participants’ total nasal symptom score (TNSS) and peak nasal inspiratory flow at 5, 15, 30, and 60 minutes after being sprayed and hourly for up to 6 hours post challenge. Blood and nasal cell samples were also collected.
The research team performed skin prick tests using serial dilutions of cat extract and an intradermal skin test (IDST) using the concentration of allergen that produced an early response of at least 15 mm at baseline. They measured early-phase responses for the both tests at 15 minutes and late-phase response to the IDST at 6 hours.
They measured serum levels of cat dander–specific IgE, IgG4, and total IgE using fluoroenzyme immunoassay. They measured serum interleukin-5 and IL-13 using high-sensitivity single-molecule digital immunoassay and performed nasal brushing using a 3-mm cytology brush 6 hours after a nasal allergy challenge. They performed whole-genome transcriptional profiling on the extracted RNA.
Combination therapy worked better and longer
The combined therapy worked better while being administered. Although the allergy shots alone stopped working after they were discontinued, the combination continued to benefit participants 1 year after that therapy ended.
At week 52, statistically significant reductions in TNSS induced by nasal allergy challenges occurred in patients receiving tezepelumab plus SCIT compared with patients receiving SCIT alone.
At week 104, 1 year after treatment ended, the primary endpoint TNSS was not significantly different in the tezepelumab-plus-SCIT group than in the SCIT-alone group, but TNSS peak 0–1 hour was significantly lower in the combination treatment group than in the SCIT-alone group.
In analysis of gene expression from nasal epithelial samples, participants who had been treated with the combination but not with either therapy by itself showed persistent modulation of the nasal immunologic environment, including diminished mast cell function. This was explained in large part by decreased transcription of the gene TPSAB1 (tryptase). Tryptase protein in nasal fluid was also decreased in the combination group, compared with the SCIT-alone group.
Adverse and serious adverse events, including infections and infestations as well as respiratory, thoracic, mediastinal, gastrointestinal, immune system, and nervous system disorders, did not differ significantly between treatment groups.
Four independent experts welcome the results
Patricia Lynne Lugar, MD, associate professor of medicine in the division of pulmonology, allergy, and critical care medicine at Duke University, Durham, N.C., found the results, especially the 1-year posttreatment response durability, surprising.
“AIT is a very effective treatment that often provides prolonged symptom improvement and is ‘curative’ in many cases,” she said in an interview. “If further studies show that tezepelumab offers long-term results, more patients might opt for combination therapy.
“A significant strength of the study is its evaluation of responses of the combination therapy on cellular output and gene expression,” Dr. Lugar added. “The mechanism by which AIT modulates the allergic response is largely understood. Tezepelumab may augment this modulation to alter the Th2 response upon exposure to the allergens.”
Will payors cover the prohibitively costly biologic?
Scott Frank, MD, associate professor in the department of family medicine and community health at Case Western Reserve University, Cleveland, called the study well designed and rigorous.
“The practicality of the approach may be limited by the need for intravenous administration of tezepelumab in addition to the traditional allergy shot,” he noted by email, “and the cost of this therapeutic approach is not addressed.”
Christopher Brooks, MD, clinical assistant professor of allergy and immunology in the department of otolaryngology at Ohio State University Wexner Medical Center, Columbus, also pointed out the drug’s cost.
“Tezepelumab is currently an expensive biologic, so it remains to be seen whether patients and payors will be willing to pay for this add-on medication when AIT by itself still remains very effective,” he said by email.
“AIT is most effective when given for 5 years, so it also remains to be seen whether the results and conclusions of this study would still hold true if done for the typical 5-year treatment period,” he added.
Stokes Peebles, MD, professor of medicine in the division of allergy, pulmonary, and critical care medicine at Vanderbilt University Medical Center, Nashville, Tenn., called the study “very well designed by a highly respected group of investigators using well-matched study populations.
“Tezepelumab has been shown to work in asthma, and there is no reason to think it would not work in allergic rhinitis,” he said in an interview.
“However, while the results of the combined therapy were statistically significant, their clinical significance was not clear. Patients do not care about statistical significance. They want to know whether a drug will be clinically significant,” he added.
Many people avoid cat allergy symptoms by avoiding cats and, in some cases, by avoiding people who live with cats, he said. Medical therapy, usually involving nasal corticosteroids and antihistamines, helps most people avoid cat allergy symptoms.
“Patients with bad allergies who have not done well with SCIT may consider adding tezepelumab, but it incurs a major cost. If medical therapy doesn’t work, allergy shots are available at roughly $3,000 per year. Adding tezepelumab costs around $40,000 more per year,” he explained. “Does the slight clinical benefit justify the greatly increased cost?”
The authors and uninvolved experts recommend further related research.
The research was supported by the National Institute of Allergy and Infectious Diseases. AstraZeneca and Amgen donated the drug used in the study. Dr. Corren reported financial relationships with AstraZeneca, and one coauthor reported relevant financial relationships with Amgen and other pharmaceutical companies. The remaining coauthors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to results of a phase 1/2 clinical trial.
“One year of allergen immunotherapy [AIT] combined with tezepelumab was significantly more effective than SCIT alone in reducing the nasal response to allergen challenge both at the end of treatment and one year after stopping treatment,” lead study author Jonathan Corren, MD, of the University of California, Los Angeles, and his colleagues wrote in The Journal of Allergy and Clinical Immunology.
“This persistent improvement in clinical response was paralleled by reductions in nasal transcripts for multiple immunologic pathways, including mast cell activation.”
The study was cited in a news release from the National Institutes of Health that said that the approach may work in a similar way with other allergens.
The Food and Drug Administration recently approved tezepelumab for the treatment of severe asthma in people aged 12 years and older. Tezelumab, a monoclonal antibody, works by blocking the cytokine thymic stromal lymphopoietin (TSLP).
“Cells that cover the surface of organs like the skin and intestines or that line the inside of the nose and lungs rapidly secrete TSLP in response to signals of potential danger,” according to the NIH news release. “In allergic disease, TSLP helps initiate an overreactive immune response to otherwise harmless substances like cat dander, provoking airway inflammation that leads to the symptoms of allergic rhinitis.”
Testing an enhanced strategy
The double-blind CATNIP trial was conducted by Dr. Corren and colleagues at nine sites in the United States. The trial included patients aged 18-65 years who’d had moderate to severe cat-induced allergic rhinitis for at least 2 years from 2015 to 2019.
The researchers excluded patients with recurrent acute or chronic sinusitis. They excluded patients who had undergone SCIT with cat allergen within the past 10 years or seasonal or perennial allergen sensitivity during nasal challenges. They also excluded persons with a history of persistent asthma.
In the parallel-design study, 121 participants were randomly allocated into four groups: 32 patients were treated with intravenous tezepelumab plus cat SCIT, 31 received the allergy shots alone, 30 received tezepelumab alone, and 28 received placebo alone for 52 weeks, followed by 52 weeks of observation.
Participants received SCIT (10,000 bioequivalent allergy units per milliliter) or matched placebo via subcutaneous injections weekly in increasing doses for around 12 weeks, followed by monthly maintenance injections (4,000 BAU or maximum tolerated dose) until week 48.
They received tezepelumab (700 mg IV) or matched placebo 1-3 days prior to the SCIT or placebo SCIT injections once every 4 weeks through week 24, then before or on the same day as the SCIT or placebo injections through week 48.
Measures of effectiveness
Participants were also given nasal allergy challenges – one spritz of a nasal spray containing cat allergen extract in each nostril at screening, baseline, and weeks 26, 52, 78, and 104. The researchers recorded participants’ total nasal symptom score (TNSS) and peak nasal inspiratory flow at 5, 15, 30, and 60 minutes after being sprayed and hourly for up to 6 hours post challenge. Blood and nasal cell samples were also collected.
The research team performed skin prick tests using serial dilutions of cat extract and an intradermal skin test (IDST) using the concentration of allergen that produced an early response of at least 15 mm at baseline. They measured early-phase responses for the both tests at 15 minutes and late-phase response to the IDST at 6 hours.
They measured serum levels of cat dander–specific IgE, IgG4, and total IgE using fluoroenzyme immunoassay. They measured serum interleukin-5 and IL-13 using high-sensitivity single-molecule digital immunoassay and performed nasal brushing using a 3-mm cytology brush 6 hours after a nasal allergy challenge. They performed whole-genome transcriptional profiling on the extracted RNA.
Combination therapy worked better and longer
The combined therapy worked better while being administered. Although the allergy shots alone stopped working after they were discontinued, the combination continued to benefit participants 1 year after that therapy ended.
At week 52, statistically significant reductions in TNSS induced by nasal allergy challenges occurred in patients receiving tezepelumab plus SCIT compared with patients receiving SCIT alone.
At week 104, 1 year after treatment ended, the primary endpoint TNSS was not significantly different in the tezepelumab-plus-SCIT group than in the SCIT-alone group, but TNSS peak 0–1 hour was significantly lower in the combination treatment group than in the SCIT-alone group.
In analysis of gene expression from nasal epithelial samples, participants who had been treated with the combination but not with either therapy by itself showed persistent modulation of the nasal immunologic environment, including diminished mast cell function. This was explained in large part by decreased transcription of the gene TPSAB1 (tryptase). Tryptase protein in nasal fluid was also decreased in the combination group, compared with the SCIT-alone group.
Adverse and serious adverse events, including infections and infestations as well as respiratory, thoracic, mediastinal, gastrointestinal, immune system, and nervous system disorders, did not differ significantly between treatment groups.
Four independent experts welcome the results
Patricia Lynne Lugar, MD, associate professor of medicine in the division of pulmonology, allergy, and critical care medicine at Duke University, Durham, N.C., found the results, especially the 1-year posttreatment response durability, surprising.
“AIT is a very effective treatment that often provides prolonged symptom improvement and is ‘curative’ in many cases,” she said in an interview. “If further studies show that tezepelumab offers long-term results, more patients might opt for combination therapy.
“A significant strength of the study is its evaluation of responses of the combination therapy on cellular output and gene expression,” Dr. Lugar added. “The mechanism by which AIT modulates the allergic response is largely understood. Tezepelumab may augment this modulation to alter the Th2 response upon exposure to the allergens.”
Will payors cover the prohibitively costly biologic?
Scott Frank, MD, associate professor in the department of family medicine and community health at Case Western Reserve University, Cleveland, called the study well designed and rigorous.
“The practicality of the approach may be limited by the need for intravenous administration of tezepelumab in addition to the traditional allergy shot,” he noted by email, “and the cost of this therapeutic approach is not addressed.”
Christopher Brooks, MD, clinical assistant professor of allergy and immunology in the department of otolaryngology at Ohio State University Wexner Medical Center, Columbus, also pointed out the drug’s cost.
“Tezepelumab is currently an expensive biologic, so it remains to be seen whether patients and payors will be willing to pay for this add-on medication when AIT by itself still remains very effective,” he said by email.
“AIT is most effective when given for 5 years, so it also remains to be seen whether the results and conclusions of this study would still hold true if done for the typical 5-year treatment period,” he added.
Stokes Peebles, MD, professor of medicine in the division of allergy, pulmonary, and critical care medicine at Vanderbilt University Medical Center, Nashville, Tenn., called the study “very well designed by a highly respected group of investigators using well-matched study populations.
“Tezepelumab has been shown to work in asthma, and there is no reason to think it would not work in allergic rhinitis,” he said in an interview.
“However, while the results of the combined therapy were statistically significant, their clinical significance was not clear. Patients do not care about statistical significance. They want to know whether a drug will be clinically significant,” he added.
Many people avoid cat allergy symptoms by avoiding cats and, in some cases, by avoiding people who live with cats, he said. Medical therapy, usually involving nasal corticosteroids and antihistamines, helps most people avoid cat allergy symptoms.
“Patients with bad allergies who have not done well with SCIT may consider adding tezepelumab, but it incurs a major cost. If medical therapy doesn’t work, allergy shots are available at roughly $3,000 per year. Adding tezepelumab costs around $40,000 more per year,” he explained. “Does the slight clinical benefit justify the greatly increased cost?”
The authors and uninvolved experts recommend further related research.
The research was supported by the National Institute of Allergy and Infectious Diseases. AstraZeneca and Amgen donated the drug used in the study. Dr. Corren reported financial relationships with AstraZeneca, and one coauthor reported relevant financial relationships with Amgen and other pharmaceutical companies. The remaining coauthors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to results of a phase 1/2 clinical trial.
“One year of allergen immunotherapy [AIT] combined with tezepelumab was significantly more effective than SCIT alone in reducing the nasal response to allergen challenge both at the end of treatment and one year after stopping treatment,” lead study author Jonathan Corren, MD, of the University of California, Los Angeles, and his colleagues wrote in The Journal of Allergy and Clinical Immunology.
“This persistent improvement in clinical response was paralleled by reductions in nasal transcripts for multiple immunologic pathways, including mast cell activation.”
The study was cited in a news release from the National Institutes of Health that said that the approach may work in a similar way with other allergens.
The Food and Drug Administration recently approved tezepelumab for the treatment of severe asthma in people aged 12 years and older. Tezelumab, a monoclonal antibody, works by blocking the cytokine thymic stromal lymphopoietin (TSLP).
“Cells that cover the surface of organs like the skin and intestines or that line the inside of the nose and lungs rapidly secrete TSLP in response to signals of potential danger,” according to the NIH news release. “In allergic disease, TSLP helps initiate an overreactive immune response to otherwise harmless substances like cat dander, provoking airway inflammation that leads to the symptoms of allergic rhinitis.”
Testing an enhanced strategy
The double-blind CATNIP trial was conducted by Dr. Corren and colleagues at nine sites in the United States. The trial included patients aged 18-65 years who’d had moderate to severe cat-induced allergic rhinitis for at least 2 years from 2015 to 2019.
The researchers excluded patients with recurrent acute or chronic sinusitis. They excluded patients who had undergone SCIT with cat allergen within the past 10 years or seasonal or perennial allergen sensitivity during nasal challenges. They also excluded persons with a history of persistent asthma.
In the parallel-design study, 121 participants were randomly allocated into four groups: 32 patients were treated with intravenous tezepelumab plus cat SCIT, 31 received the allergy shots alone, 30 received tezepelumab alone, and 28 received placebo alone for 52 weeks, followed by 52 weeks of observation.
Participants received SCIT (10,000 bioequivalent allergy units per milliliter) or matched placebo via subcutaneous injections weekly in increasing doses for around 12 weeks, followed by monthly maintenance injections (4,000 BAU or maximum tolerated dose) until week 48.
They received tezepelumab (700 mg IV) or matched placebo 1-3 days prior to the SCIT or placebo SCIT injections once every 4 weeks through week 24, then before or on the same day as the SCIT or placebo injections through week 48.
Measures of effectiveness
Participants were also given nasal allergy challenges – one spritz of a nasal spray containing cat allergen extract in each nostril at screening, baseline, and weeks 26, 52, 78, and 104. The researchers recorded participants’ total nasal symptom score (TNSS) and peak nasal inspiratory flow at 5, 15, 30, and 60 minutes after being sprayed and hourly for up to 6 hours post challenge. Blood and nasal cell samples were also collected.
The research team performed skin prick tests using serial dilutions of cat extract and an intradermal skin test (IDST) using the concentration of allergen that produced an early response of at least 15 mm at baseline. They measured early-phase responses for the both tests at 15 minutes and late-phase response to the IDST at 6 hours.
They measured serum levels of cat dander–specific IgE, IgG4, and total IgE using fluoroenzyme immunoassay. They measured serum interleukin-5 and IL-13 using high-sensitivity single-molecule digital immunoassay and performed nasal brushing using a 3-mm cytology brush 6 hours after a nasal allergy challenge. They performed whole-genome transcriptional profiling on the extracted RNA.
Combination therapy worked better and longer
The combined therapy worked better while being administered. Although the allergy shots alone stopped working after they were discontinued, the combination continued to benefit participants 1 year after that therapy ended.
At week 52, statistically significant reductions in TNSS induced by nasal allergy challenges occurred in patients receiving tezepelumab plus SCIT compared with patients receiving SCIT alone.
At week 104, 1 year after treatment ended, the primary endpoint TNSS was not significantly different in the tezepelumab-plus-SCIT group than in the SCIT-alone group, but TNSS peak 0–1 hour was significantly lower in the combination treatment group than in the SCIT-alone group.
In analysis of gene expression from nasal epithelial samples, participants who had been treated with the combination but not with either therapy by itself showed persistent modulation of the nasal immunologic environment, including diminished mast cell function. This was explained in large part by decreased transcription of the gene TPSAB1 (tryptase). Tryptase protein in nasal fluid was also decreased in the combination group, compared with the SCIT-alone group.
Adverse and serious adverse events, including infections and infestations as well as respiratory, thoracic, mediastinal, gastrointestinal, immune system, and nervous system disorders, did not differ significantly between treatment groups.
Four independent experts welcome the results
Patricia Lynne Lugar, MD, associate professor of medicine in the division of pulmonology, allergy, and critical care medicine at Duke University, Durham, N.C., found the results, especially the 1-year posttreatment response durability, surprising.
“AIT is a very effective treatment that often provides prolonged symptom improvement and is ‘curative’ in many cases,” she said in an interview. “If further studies show that tezepelumab offers long-term results, more patients might opt for combination therapy.
“A significant strength of the study is its evaluation of responses of the combination therapy on cellular output and gene expression,” Dr. Lugar added. “The mechanism by which AIT modulates the allergic response is largely understood. Tezepelumab may augment this modulation to alter the Th2 response upon exposure to the allergens.”
Will payors cover the prohibitively costly biologic?
Scott Frank, MD, associate professor in the department of family medicine and community health at Case Western Reserve University, Cleveland, called the study well designed and rigorous.
“The practicality of the approach may be limited by the need for intravenous administration of tezepelumab in addition to the traditional allergy shot,” he noted by email, “and the cost of this therapeutic approach is not addressed.”
Christopher Brooks, MD, clinical assistant professor of allergy and immunology in the department of otolaryngology at Ohio State University Wexner Medical Center, Columbus, also pointed out the drug’s cost.
“Tezepelumab is currently an expensive biologic, so it remains to be seen whether patients and payors will be willing to pay for this add-on medication when AIT by itself still remains very effective,” he said by email.
“AIT is most effective when given for 5 years, so it also remains to be seen whether the results and conclusions of this study would still hold true if done for the typical 5-year treatment period,” he added.
Stokes Peebles, MD, professor of medicine in the division of allergy, pulmonary, and critical care medicine at Vanderbilt University Medical Center, Nashville, Tenn., called the study “very well designed by a highly respected group of investigators using well-matched study populations.
“Tezepelumab has been shown to work in asthma, and there is no reason to think it would not work in allergic rhinitis,” he said in an interview.
“However, while the results of the combined therapy were statistically significant, their clinical significance was not clear. Patients do not care about statistical significance. They want to know whether a drug will be clinically significant,” he added.
Many people avoid cat allergy symptoms by avoiding cats and, in some cases, by avoiding people who live with cats, he said. Medical therapy, usually involving nasal corticosteroids and antihistamines, helps most people avoid cat allergy symptoms.
“Patients with bad allergies who have not done well with SCIT may consider adding tezepelumab, but it incurs a major cost. If medical therapy doesn’t work, allergy shots are available at roughly $3,000 per year. Adding tezepelumab costs around $40,000 more per year,” he explained. “Does the slight clinical benefit justify the greatly increased cost?”
The authors and uninvolved experts recommend further related research.
The research was supported by the National Institute of Allergy and Infectious Diseases. AstraZeneca and Amgen donated the drug used in the study. Dr. Corren reported financial relationships with AstraZeneca, and one coauthor reported relevant financial relationships with Amgen and other pharmaceutical companies. The remaining coauthors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Why your professional persona may be considered unprofessional
On one of the first days of medical school, Adaira Landry, MD, applied her favorite dark shade of lipstick and headed to her orientation. She was eager to learn about program expectations and connect with fellow aspiring physicians. But when Dr. Landry got there, one of her brand-new peers turned to her and asked, “Why do you wear your lipstick like an angry Black woman?”
“Imagine hearing that,” Dr. Landry, now an emergency medical physician in Boston, says. “It was so hurtful.”
So, what is a “standard-issue doctor” expected to look like? Physicians manage their appearances in myriad ways: through clothes, accessories, hair style, makeup; through a social media presence or lack thereof; in the rhythms and nuances of their interactions with patients and colleagues. These things add up to a professional “persona” – the Latin word for “mask,” or the face on display for the world to see.
While the health care field itself is diversifying, its guidelines for professionalism appear slower to change, often excluding or frowning upon expressions of individual personality or identity.
“Medicine is run primarily by men. It’s an objective truth,” Dr. Landry says. “Currently and historically, the standard of professionalism, especially in the physical sense, was set by them. As we increase diversity and welcome people bringing their authentic self to work, the prior definitions of professionalism are obviously in need of change.”
Split social media personalities
In August 2020, the Journal of Vascular Surgery published a study on the “prevalence of unprofessional social media content among young vascular surgeons.” The content that was deemed “unprofessional” included opinions on political issues like abortion and gun control. Photos of physicians holding alcoholic drinks or wearing “inappropriate/offensive attire,” including underwear, “provocative Halloween costumes,” and “bikinis/swimwear” were also censured. Six men and one woman worked on the study, and three of the male researchers took on the task of seeking out the “unprofessional” photos on social media. The resulting paper was reviewed by an all-male editorial board.
The study sparked immediate backlash and prompted hundreds of health care professionals to post photos of themselves in bathing suits with the hashtag “#medbikini.” The journal then retracted the study and issued an apology on Twitter, recognizing “errors in the design of the study with regards to conscious and unconscious bias.”
The researchers’ original definition of professionalism suggests that physicians should manage their personae even outside of work hours. “I think medicine in general is a very conservative and hierarchical field of study and of work, to say the least,” says Sarah Fraser, MD, a family medicine physician in Nova Scotia, Canada. “There’s this view that we have to have completely separate personal and professional lives, like church and state.”
The #medbikini controversy inspired Dr. Fraser to write an op-ed for the British Medical Journal blog about the flaws of requiring physicians to keep their personal and professional selves separate. The piece referenced Robert Louis Stevenson’s 1886 Gothic novella “The Strange Case of Dr. Jekyll and Mr. Hyde,” in which the respected scientist Dr. Jekyll creates an alter ego so he can express his evil urges without experiencing guilt, punishment, or loss of livelihood. Dr. Fraser likened this story to the pressure physicians feel to shrink or split themselves to squeeze into a narrow definition of professionalism.
But Dr. Landry points out that some elements of expression seen as unprofessional cannot be entirely separated from a physician’s fundamental identity. “For Black women, our daily behaviors and forms of expression that are deemed ‘unprofessional’ are much more subtle than being able to wear a bikini on social media,” she says. “The way we wear our hair, the tone of our voice, the color of our lipstick, the way we wear scrub caps are parts of us that are called into question.”
Keeping up appearances
The stereotype of what a doctor should look like starts to shape physicians’ professional personae in medical school. When Jennifer Caputo-Seidler, MD, started medical school in 2008, the dress code requirements for male students were simple: pants, a button-down shirt, a tie. But then there were the rules for women: Hair should be tied back. Minimal makeup. No flashy jewelry. Nothing without sleeves. Neutral colors. High necklines. Low hemlines. “The message I got was that we need to dress like the men in order to be taken seriously and to be seen as professional,” says Dr. Caputo-Seidler, now an assistant professor of medicine at the University of South Florida, Tampa, “and so that’s what I did.”
A 2018 analysis of 78 “draw-a-scientist” studies found that children have overwhelmingly associated scientific fields with men for the last 50 years. Overall, children drew 73% of scientists as men. The drawings grew more gender diverse over time, but even as more women entered scientific fields, both boys and girls continued to draw significantly more male than female scientists.
Not everyone at Dr. Caputo-Seidler’s medical school adhered to the environment’s gendered expectations. One resident she worked with often wore voluminous hairstyles, lipstick, and high heels. Dr. Caputo-Seidler overheard her peers as they gossiped behind the resident’s back, ridiculing the way she looked.
“She was good at her job,” Dr. Caputo-Seidler says. “She knew her patients. She had things down. She was, by all measures, very competent. But when people saw her dressing outside the norm and being forward with her femininity, there was definitely a lot of chatter about it.”
While expectations for a conservative appearance may disproportionately affect women, and particularly women of color, they also affect men who deviate from the norm. “As an LGBTQ+ person working as a ‘professional,’ I have countless stories and moments where I had my professionalism questioned,” Blair Peters, MD, a plastic surgeon and assistant professor at Oregon Health & Science University, Portland, wrote on Twitter. “Why is it ‘unprofessional’ to have colored hair? Why is it ‘unprofessional’ to have a visible tattoo? Why is it ‘unprofessional’ to wear bright colors and patterns?”
Dr. Fraser remembers a fellow medical student who had full-sleeve tattoos on both of his arms. A preceptor made a comment about it to Dr. Fraser, and then instructed the student to cover up his tattoos. “I think that there are scenarios when having tattoos or having different-colored hair or expressing your individual personality could help you even better bond with your patients,” Dr. Fraser says, “especially if you’re, for example, working with youth.”
Unmasking health care
Beyond the facets of dress codes and social media posts, the issue of professional personae speaks to the deeper issue of inclusion in medicine. As the field grows increasingly diverse, health care institutions and those they serve may need to expand their definitions of professionalism to include more truthful expressions of who contemporary health care professionals are as people.
Dr. Fraser suggests that the benefits of physicians embracing self-expression – rather than assimilating to an outdated model of professionalism – extend beyond the individual.
“Whether it comes to what you choose to wear to the clinic on a day-to-day basis, or what you choose to share on a social media account, as long as it’s not harming others, then I think that it’s a positive thing to be able to be yourself and express yourself,” she says. “I feel like doctors are expected to have a different personality when we’re at the clinic, and usually it’s more conservative or objective or aloof. But I think that by being open about who we are, we’ll actually help build a trusting relationship with both patients and society.”
A version of this article first appeared on Medscape.com.
On one of the first days of medical school, Adaira Landry, MD, applied her favorite dark shade of lipstick and headed to her orientation. She was eager to learn about program expectations and connect with fellow aspiring physicians. But when Dr. Landry got there, one of her brand-new peers turned to her and asked, “Why do you wear your lipstick like an angry Black woman?”
“Imagine hearing that,” Dr. Landry, now an emergency medical physician in Boston, says. “It was so hurtful.”
So, what is a “standard-issue doctor” expected to look like? Physicians manage their appearances in myriad ways: through clothes, accessories, hair style, makeup; through a social media presence or lack thereof; in the rhythms and nuances of their interactions with patients and colleagues. These things add up to a professional “persona” – the Latin word for “mask,” or the face on display for the world to see.
While the health care field itself is diversifying, its guidelines for professionalism appear slower to change, often excluding or frowning upon expressions of individual personality or identity.
“Medicine is run primarily by men. It’s an objective truth,” Dr. Landry says. “Currently and historically, the standard of professionalism, especially in the physical sense, was set by them. As we increase diversity and welcome people bringing their authentic self to work, the prior definitions of professionalism are obviously in need of change.”
Split social media personalities
In August 2020, the Journal of Vascular Surgery published a study on the “prevalence of unprofessional social media content among young vascular surgeons.” The content that was deemed “unprofessional” included opinions on political issues like abortion and gun control. Photos of physicians holding alcoholic drinks or wearing “inappropriate/offensive attire,” including underwear, “provocative Halloween costumes,” and “bikinis/swimwear” were also censured. Six men and one woman worked on the study, and three of the male researchers took on the task of seeking out the “unprofessional” photos on social media. The resulting paper was reviewed by an all-male editorial board.
The study sparked immediate backlash and prompted hundreds of health care professionals to post photos of themselves in bathing suits with the hashtag “#medbikini.” The journal then retracted the study and issued an apology on Twitter, recognizing “errors in the design of the study with regards to conscious and unconscious bias.”
The researchers’ original definition of professionalism suggests that physicians should manage their personae even outside of work hours. “I think medicine in general is a very conservative and hierarchical field of study and of work, to say the least,” says Sarah Fraser, MD, a family medicine physician in Nova Scotia, Canada. “There’s this view that we have to have completely separate personal and professional lives, like church and state.”
The #medbikini controversy inspired Dr. Fraser to write an op-ed for the British Medical Journal blog about the flaws of requiring physicians to keep their personal and professional selves separate. The piece referenced Robert Louis Stevenson’s 1886 Gothic novella “The Strange Case of Dr. Jekyll and Mr. Hyde,” in which the respected scientist Dr. Jekyll creates an alter ego so he can express his evil urges without experiencing guilt, punishment, or loss of livelihood. Dr. Fraser likened this story to the pressure physicians feel to shrink or split themselves to squeeze into a narrow definition of professionalism.
But Dr. Landry points out that some elements of expression seen as unprofessional cannot be entirely separated from a physician’s fundamental identity. “For Black women, our daily behaviors and forms of expression that are deemed ‘unprofessional’ are much more subtle than being able to wear a bikini on social media,” she says. “The way we wear our hair, the tone of our voice, the color of our lipstick, the way we wear scrub caps are parts of us that are called into question.”
Keeping up appearances
The stereotype of what a doctor should look like starts to shape physicians’ professional personae in medical school. When Jennifer Caputo-Seidler, MD, started medical school in 2008, the dress code requirements for male students were simple: pants, a button-down shirt, a tie. But then there were the rules for women: Hair should be tied back. Minimal makeup. No flashy jewelry. Nothing without sleeves. Neutral colors. High necklines. Low hemlines. “The message I got was that we need to dress like the men in order to be taken seriously and to be seen as professional,” says Dr. Caputo-Seidler, now an assistant professor of medicine at the University of South Florida, Tampa, “and so that’s what I did.”
A 2018 analysis of 78 “draw-a-scientist” studies found that children have overwhelmingly associated scientific fields with men for the last 50 years. Overall, children drew 73% of scientists as men. The drawings grew more gender diverse over time, but even as more women entered scientific fields, both boys and girls continued to draw significantly more male than female scientists.
Not everyone at Dr. Caputo-Seidler’s medical school adhered to the environment’s gendered expectations. One resident she worked with often wore voluminous hairstyles, lipstick, and high heels. Dr. Caputo-Seidler overheard her peers as they gossiped behind the resident’s back, ridiculing the way she looked.
“She was good at her job,” Dr. Caputo-Seidler says. “She knew her patients. She had things down. She was, by all measures, very competent. But when people saw her dressing outside the norm and being forward with her femininity, there was definitely a lot of chatter about it.”
While expectations for a conservative appearance may disproportionately affect women, and particularly women of color, they also affect men who deviate from the norm. “As an LGBTQ+ person working as a ‘professional,’ I have countless stories and moments where I had my professionalism questioned,” Blair Peters, MD, a plastic surgeon and assistant professor at Oregon Health & Science University, Portland, wrote on Twitter. “Why is it ‘unprofessional’ to have colored hair? Why is it ‘unprofessional’ to have a visible tattoo? Why is it ‘unprofessional’ to wear bright colors and patterns?”
Dr. Fraser remembers a fellow medical student who had full-sleeve tattoos on both of his arms. A preceptor made a comment about it to Dr. Fraser, and then instructed the student to cover up his tattoos. “I think that there are scenarios when having tattoos or having different-colored hair or expressing your individual personality could help you even better bond with your patients,” Dr. Fraser says, “especially if you’re, for example, working with youth.”
Unmasking health care
Beyond the facets of dress codes and social media posts, the issue of professional personae speaks to the deeper issue of inclusion in medicine. As the field grows increasingly diverse, health care institutions and those they serve may need to expand their definitions of professionalism to include more truthful expressions of who contemporary health care professionals are as people.
Dr. Fraser suggests that the benefits of physicians embracing self-expression – rather than assimilating to an outdated model of professionalism – extend beyond the individual.
“Whether it comes to what you choose to wear to the clinic on a day-to-day basis, or what you choose to share on a social media account, as long as it’s not harming others, then I think that it’s a positive thing to be able to be yourself and express yourself,” she says. “I feel like doctors are expected to have a different personality when we’re at the clinic, and usually it’s more conservative or objective or aloof. But I think that by being open about who we are, we’ll actually help build a trusting relationship with both patients and society.”
A version of this article first appeared on Medscape.com.
On one of the first days of medical school, Adaira Landry, MD, applied her favorite dark shade of lipstick and headed to her orientation. She was eager to learn about program expectations and connect with fellow aspiring physicians. But when Dr. Landry got there, one of her brand-new peers turned to her and asked, “Why do you wear your lipstick like an angry Black woman?”
“Imagine hearing that,” Dr. Landry, now an emergency medical physician in Boston, says. “It was so hurtful.”
So, what is a “standard-issue doctor” expected to look like? Physicians manage their appearances in myriad ways: through clothes, accessories, hair style, makeup; through a social media presence or lack thereof; in the rhythms and nuances of their interactions with patients and colleagues. These things add up to a professional “persona” – the Latin word for “mask,” or the face on display for the world to see.
While the health care field itself is diversifying, its guidelines for professionalism appear slower to change, often excluding or frowning upon expressions of individual personality or identity.
“Medicine is run primarily by men. It’s an objective truth,” Dr. Landry says. “Currently and historically, the standard of professionalism, especially in the physical sense, was set by them. As we increase diversity and welcome people bringing their authentic self to work, the prior definitions of professionalism are obviously in need of change.”
Split social media personalities
In August 2020, the Journal of Vascular Surgery published a study on the “prevalence of unprofessional social media content among young vascular surgeons.” The content that was deemed “unprofessional” included opinions on political issues like abortion and gun control. Photos of physicians holding alcoholic drinks or wearing “inappropriate/offensive attire,” including underwear, “provocative Halloween costumes,” and “bikinis/swimwear” were also censured. Six men and one woman worked on the study, and three of the male researchers took on the task of seeking out the “unprofessional” photos on social media. The resulting paper was reviewed by an all-male editorial board.
The study sparked immediate backlash and prompted hundreds of health care professionals to post photos of themselves in bathing suits with the hashtag “#medbikini.” The journal then retracted the study and issued an apology on Twitter, recognizing “errors in the design of the study with regards to conscious and unconscious bias.”
The researchers’ original definition of professionalism suggests that physicians should manage their personae even outside of work hours. “I think medicine in general is a very conservative and hierarchical field of study and of work, to say the least,” says Sarah Fraser, MD, a family medicine physician in Nova Scotia, Canada. “There’s this view that we have to have completely separate personal and professional lives, like church and state.”
The #medbikini controversy inspired Dr. Fraser to write an op-ed for the British Medical Journal blog about the flaws of requiring physicians to keep their personal and professional selves separate. The piece referenced Robert Louis Stevenson’s 1886 Gothic novella “The Strange Case of Dr. Jekyll and Mr. Hyde,” in which the respected scientist Dr. Jekyll creates an alter ego so he can express his evil urges without experiencing guilt, punishment, or loss of livelihood. Dr. Fraser likened this story to the pressure physicians feel to shrink or split themselves to squeeze into a narrow definition of professionalism.
But Dr. Landry points out that some elements of expression seen as unprofessional cannot be entirely separated from a physician’s fundamental identity. “For Black women, our daily behaviors and forms of expression that are deemed ‘unprofessional’ are much more subtle than being able to wear a bikini on social media,” she says. “The way we wear our hair, the tone of our voice, the color of our lipstick, the way we wear scrub caps are parts of us that are called into question.”
Keeping up appearances
The stereotype of what a doctor should look like starts to shape physicians’ professional personae in medical school. When Jennifer Caputo-Seidler, MD, started medical school in 2008, the dress code requirements for male students were simple: pants, a button-down shirt, a tie. But then there were the rules for women: Hair should be tied back. Minimal makeup. No flashy jewelry. Nothing without sleeves. Neutral colors. High necklines. Low hemlines. “The message I got was that we need to dress like the men in order to be taken seriously and to be seen as professional,” says Dr. Caputo-Seidler, now an assistant professor of medicine at the University of South Florida, Tampa, “and so that’s what I did.”
A 2018 analysis of 78 “draw-a-scientist” studies found that children have overwhelmingly associated scientific fields with men for the last 50 years. Overall, children drew 73% of scientists as men. The drawings grew more gender diverse over time, but even as more women entered scientific fields, both boys and girls continued to draw significantly more male than female scientists.
Not everyone at Dr. Caputo-Seidler’s medical school adhered to the environment’s gendered expectations. One resident she worked with often wore voluminous hairstyles, lipstick, and high heels. Dr. Caputo-Seidler overheard her peers as they gossiped behind the resident’s back, ridiculing the way she looked.
“She was good at her job,” Dr. Caputo-Seidler says. “She knew her patients. She had things down. She was, by all measures, very competent. But when people saw her dressing outside the norm and being forward with her femininity, there was definitely a lot of chatter about it.”
While expectations for a conservative appearance may disproportionately affect women, and particularly women of color, they also affect men who deviate from the norm. “As an LGBTQ+ person working as a ‘professional,’ I have countless stories and moments where I had my professionalism questioned,” Blair Peters, MD, a plastic surgeon and assistant professor at Oregon Health & Science University, Portland, wrote on Twitter. “Why is it ‘unprofessional’ to have colored hair? Why is it ‘unprofessional’ to have a visible tattoo? Why is it ‘unprofessional’ to wear bright colors and patterns?”
Dr. Fraser remembers a fellow medical student who had full-sleeve tattoos on both of his arms. A preceptor made a comment about it to Dr. Fraser, and then instructed the student to cover up his tattoos. “I think that there are scenarios when having tattoos or having different-colored hair or expressing your individual personality could help you even better bond with your patients,” Dr. Fraser says, “especially if you’re, for example, working with youth.”
Unmasking health care
Beyond the facets of dress codes and social media posts, the issue of professional personae speaks to the deeper issue of inclusion in medicine. As the field grows increasingly diverse, health care institutions and those they serve may need to expand their definitions of professionalism to include more truthful expressions of who contemporary health care professionals are as people.
Dr. Fraser suggests that the benefits of physicians embracing self-expression – rather than assimilating to an outdated model of professionalism – extend beyond the individual.
“Whether it comes to what you choose to wear to the clinic on a day-to-day basis, or what you choose to share on a social media account, as long as it’s not harming others, then I think that it’s a positive thing to be able to be yourself and express yourself,” she says. “I feel like doctors are expected to have a different personality when we’re at the clinic, and usually it’s more conservative or objective or aloof. But I think that by being open about who we are, we’ll actually help build a trusting relationship with both patients and society.”
A version of this article first appeared on Medscape.com.