User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Misinterpretation is a science, not an art
It isn’t autocorrect’s fault this time, we swear
We’ve come a long way with communication technology. Back in the day, when Gondor needed to call for aid, they had to pull off the greatest signal fire montage of all time. Now we can send each other texts back and forth in an instant. (“Hey Theoden, send army, need help pls” doesn’t quite have the same gravitas though.) The question is, how do our brains keep up with such rapidly advancing technology?
Er, they don’t. Not really. Instead, our brains create shortcuts called “good-enough language processing,” which is exactly what it sounds like.
Psychologists and psycholinguists have been studying misinterpretations such as good-enough language processing since the 1970s. Recently, however, psycholinguists from the Centre for Language and Brain at Higher School of Economics in Moscow have found that, when it comes to reading comprehension over text, older adults are using their knowledge of the world over how it’s grammatically formed in the sentence.
In the study, 349 people were asked to read and interpret four sentences, the third of which (translated from Russian) was: “Misha met the firefighter’s dentist, who had put out a fire in the warehouse.” When asked who put the fire out, 79% of older adults (aged 55 years and older), utilizing good-enough language processing, said the firefighter put out the fire. You probably glossed over that sentence and assumed the same thing. But this time, the dentist was the real hero.
That said, adolescents (aged 13-17) and young adults (aged 20-30) weren’t much better, and got that particular sentence wrong 63%-68% of the time. According to the researchers, good-enough language processing forms in adolescence and intensifies throughout adulthood.
Moral of the story? We should utilize signal fires more often. Less room for misinterpretation. When the beacons of Minas Tirith were lit, Rohan answered.
Singing … your … lungs … out
There’s nothing quite like a karaoke bar to unleash your inner rock star. Hey, why not just go for it, everyone is just as bad at singing as you. That’s part of the fun.
A 25-year-old man named Wang Zhe may have taken the karaoke concept a bit too far, however. While out with friends at a birthday party, Mr. Zhe let loose on a song with a particularly large number of high notes. He tried his best, gamely attacking the song until he felt a pain in his chest. He didn’t think much of it, although he did cut his performance short, but then he awoke the next morning unable to breathe properly.
After a trip to the hospital, he explained the sequence of events to the doctors, and an x-ray found that the culprit of the pain and difficulty breathing was a life-threatening condition in which air bubbles are created between the chest and lung. All the force Mr. Zhe had used trying to sing made air sacks in his lung burst, causing the air bubbles and his lung to be compressed to 15% of what it should be. Mr. Zhe needed surgery to remove the air bubbles, but fortunately turned out just fine.
So, if you’re ever at a karaoke bar, looking for a song to sing, maybe avoid the ones with super high notes and stick with something a little lower. We’re picturing something like Paul Robeson singing Ol’ Man River. That oughta do the trick.
And the word of the year is …
Flibbertigibbet. Bamboozle. Gobbledygook. If the LOTME staff had any say, those would be the words of the year every year, but sadly, we’re not in charge of such things. Instead, we’ll just have to defer to Oxford and Merriam-Webster, both of whom have recently chosen their words of the year. No word yet on whether or not they made their announcement at a red carpet gala dinner attended by all the most fashionable and powerful words out there, but we’re hoping that’s what happened.
We’ll start with Oxford, since they did choose first. We all know Oxford is the bad boy of the dictionary world, so they’ve chosen a casual colloquialism related to the big COVID-sized elephant in the room (or should it be elephant-sized COVID in the room?): Vax. According to them, while vax has been hanging around since the 1980s, it’s only been in the past year that it’s exploded in popularity in a wide range of contexts (we can’t imagine what those would be). According to Oxford, “as a short pithy word, it appeals, perhaps especially to media commentators, when more formal alternatives are much more long-winded.”
Speaking of long-winded, that brings us to Merriam-Webster, the sheltered nerd of the dictionary world. Clearly they’re too good for vax, so they’ve gone with vaccine as their 2021 word of the year. Vaccine, according to Merriam-Webster, carries two big stories: The impressive and herculean feat of bringing a COVID-19 vaccine so quickly to so many people, and the complex political and social upheaval between vaccine supporters and deniers.
Vaccine also serves as a great bookend for Merriam-Webster’s 2020 word of the year: Pandemic. In 2020, the pandemic started, and in 2021, thanks to the vaccine, the pandemic ends. That’s how it works, right? We have a vaccine, it’s all over now. What’s that? Omicron? No! Bad COVID! You do that outside, not on the carpet!
It isn’t autocorrect’s fault this time, we swear
We’ve come a long way with communication technology. Back in the day, when Gondor needed to call for aid, they had to pull off the greatest signal fire montage of all time. Now we can send each other texts back and forth in an instant. (“Hey Theoden, send army, need help pls” doesn’t quite have the same gravitas though.) The question is, how do our brains keep up with such rapidly advancing technology?
Er, they don’t. Not really. Instead, our brains create shortcuts called “good-enough language processing,” which is exactly what it sounds like.
Psychologists and psycholinguists have been studying misinterpretations such as good-enough language processing since the 1970s. Recently, however, psycholinguists from the Centre for Language and Brain at Higher School of Economics in Moscow have found that, when it comes to reading comprehension over text, older adults are using their knowledge of the world over how it’s grammatically formed in the sentence.
In the study, 349 people were asked to read and interpret four sentences, the third of which (translated from Russian) was: “Misha met the firefighter’s dentist, who had put out a fire in the warehouse.” When asked who put the fire out, 79% of older adults (aged 55 years and older), utilizing good-enough language processing, said the firefighter put out the fire. You probably glossed over that sentence and assumed the same thing. But this time, the dentist was the real hero.
That said, adolescents (aged 13-17) and young adults (aged 20-30) weren’t much better, and got that particular sentence wrong 63%-68% of the time. According to the researchers, good-enough language processing forms in adolescence and intensifies throughout adulthood.
Moral of the story? We should utilize signal fires more often. Less room for misinterpretation. When the beacons of Minas Tirith were lit, Rohan answered.
Singing … your … lungs … out
There’s nothing quite like a karaoke bar to unleash your inner rock star. Hey, why not just go for it, everyone is just as bad at singing as you. That’s part of the fun.
A 25-year-old man named Wang Zhe may have taken the karaoke concept a bit too far, however. While out with friends at a birthday party, Mr. Zhe let loose on a song with a particularly large number of high notes. He tried his best, gamely attacking the song until he felt a pain in his chest. He didn’t think much of it, although he did cut his performance short, but then he awoke the next morning unable to breathe properly.
After a trip to the hospital, he explained the sequence of events to the doctors, and an x-ray found that the culprit of the pain and difficulty breathing was a life-threatening condition in which air bubbles are created between the chest and lung. All the force Mr. Zhe had used trying to sing made air sacks in his lung burst, causing the air bubbles and his lung to be compressed to 15% of what it should be. Mr. Zhe needed surgery to remove the air bubbles, but fortunately turned out just fine.
So, if you’re ever at a karaoke bar, looking for a song to sing, maybe avoid the ones with super high notes and stick with something a little lower. We’re picturing something like Paul Robeson singing Ol’ Man River. That oughta do the trick.
And the word of the year is …
Flibbertigibbet. Bamboozle. Gobbledygook. If the LOTME staff had any say, those would be the words of the year every year, but sadly, we’re not in charge of such things. Instead, we’ll just have to defer to Oxford and Merriam-Webster, both of whom have recently chosen their words of the year. No word yet on whether or not they made their announcement at a red carpet gala dinner attended by all the most fashionable and powerful words out there, but we’re hoping that’s what happened.
We’ll start with Oxford, since they did choose first. We all know Oxford is the bad boy of the dictionary world, so they’ve chosen a casual colloquialism related to the big COVID-sized elephant in the room (or should it be elephant-sized COVID in the room?): Vax. According to them, while vax has been hanging around since the 1980s, it’s only been in the past year that it’s exploded in popularity in a wide range of contexts (we can’t imagine what those would be). According to Oxford, “as a short pithy word, it appeals, perhaps especially to media commentators, when more formal alternatives are much more long-winded.”
Speaking of long-winded, that brings us to Merriam-Webster, the sheltered nerd of the dictionary world. Clearly they’re too good for vax, so they’ve gone with vaccine as their 2021 word of the year. Vaccine, according to Merriam-Webster, carries two big stories: The impressive and herculean feat of bringing a COVID-19 vaccine so quickly to so many people, and the complex political and social upheaval between vaccine supporters and deniers.
Vaccine also serves as a great bookend for Merriam-Webster’s 2020 word of the year: Pandemic. In 2020, the pandemic started, and in 2021, thanks to the vaccine, the pandemic ends. That’s how it works, right? We have a vaccine, it’s all over now. What’s that? Omicron? No! Bad COVID! You do that outside, not on the carpet!
It isn’t autocorrect’s fault this time, we swear
We’ve come a long way with communication technology. Back in the day, when Gondor needed to call for aid, they had to pull off the greatest signal fire montage of all time. Now we can send each other texts back and forth in an instant. (“Hey Theoden, send army, need help pls” doesn’t quite have the same gravitas though.) The question is, how do our brains keep up with such rapidly advancing technology?
Er, they don’t. Not really. Instead, our brains create shortcuts called “good-enough language processing,” which is exactly what it sounds like.
Psychologists and psycholinguists have been studying misinterpretations such as good-enough language processing since the 1970s. Recently, however, psycholinguists from the Centre for Language and Brain at Higher School of Economics in Moscow have found that, when it comes to reading comprehension over text, older adults are using their knowledge of the world over how it’s grammatically formed in the sentence.
In the study, 349 people were asked to read and interpret four sentences, the third of which (translated from Russian) was: “Misha met the firefighter’s dentist, who had put out a fire in the warehouse.” When asked who put the fire out, 79% of older adults (aged 55 years and older), utilizing good-enough language processing, said the firefighter put out the fire. You probably glossed over that sentence and assumed the same thing. But this time, the dentist was the real hero.
That said, adolescents (aged 13-17) and young adults (aged 20-30) weren’t much better, and got that particular sentence wrong 63%-68% of the time. According to the researchers, good-enough language processing forms in adolescence and intensifies throughout adulthood.
Moral of the story? We should utilize signal fires more often. Less room for misinterpretation. When the beacons of Minas Tirith were lit, Rohan answered.
Singing … your … lungs … out
There’s nothing quite like a karaoke bar to unleash your inner rock star. Hey, why not just go for it, everyone is just as bad at singing as you. That’s part of the fun.
A 25-year-old man named Wang Zhe may have taken the karaoke concept a bit too far, however. While out with friends at a birthday party, Mr. Zhe let loose on a song with a particularly large number of high notes. He tried his best, gamely attacking the song until he felt a pain in his chest. He didn’t think much of it, although he did cut his performance short, but then he awoke the next morning unable to breathe properly.
After a trip to the hospital, he explained the sequence of events to the doctors, and an x-ray found that the culprit of the pain and difficulty breathing was a life-threatening condition in which air bubbles are created between the chest and lung. All the force Mr. Zhe had used trying to sing made air sacks in his lung burst, causing the air bubbles and his lung to be compressed to 15% of what it should be. Mr. Zhe needed surgery to remove the air bubbles, but fortunately turned out just fine.
So, if you’re ever at a karaoke bar, looking for a song to sing, maybe avoid the ones with super high notes and stick with something a little lower. We’re picturing something like Paul Robeson singing Ol’ Man River. That oughta do the trick.
And the word of the year is …
Flibbertigibbet. Bamboozle. Gobbledygook. If the LOTME staff had any say, those would be the words of the year every year, but sadly, we’re not in charge of such things. Instead, we’ll just have to defer to Oxford and Merriam-Webster, both of whom have recently chosen their words of the year. No word yet on whether or not they made their announcement at a red carpet gala dinner attended by all the most fashionable and powerful words out there, but we’re hoping that’s what happened.
We’ll start with Oxford, since they did choose first. We all know Oxford is the bad boy of the dictionary world, so they’ve chosen a casual colloquialism related to the big COVID-sized elephant in the room (or should it be elephant-sized COVID in the room?): Vax. According to them, while vax has been hanging around since the 1980s, it’s only been in the past year that it’s exploded in popularity in a wide range of contexts (we can’t imagine what those would be). According to Oxford, “as a short pithy word, it appeals, perhaps especially to media commentators, when more formal alternatives are much more long-winded.”
Speaking of long-winded, that brings us to Merriam-Webster, the sheltered nerd of the dictionary world. Clearly they’re too good for vax, so they’ve gone with vaccine as their 2021 word of the year. Vaccine, according to Merriam-Webster, carries two big stories: The impressive and herculean feat of bringing a COVID-19 vaccine so quickly to so many people, and the complex political and social upheaval between vaccine supporters and deniers.
Vaccine also serves as a great bookend for Merriam-Webster’s 2020 word of the year: Pandemic. In 2020, the pandemic started, and in 2021, thanks to the vaccine, the pandemic ends. That’s how it works, right? We have a vaccine, it’s all over now. What’s that? Omicron? No! Bad COVID! You do that outside, not on the carpet!
Clinical Edge Journal Scan Commentary: Psoriasis December 2021
A retrospective observational study of nearly 1200 prior biologic-experienced adult patients with psoriasis now receiving either secukinumab (n=780) or ixekizumab (n=411), both of which inhibit IL-17A, examined drug persistence after 18 months. Ixekizumab was associated with significantly higher rates of high treatment adherence (42% vs 35%; P = .019) and persistence (44.9% vs 36.9%; P = .007) and lower discontinuation (48.4% vs 56.0%; P = .018) and switching (26.6% vs 34.0%; P = .009) rates than secukinumab. Both drugs are dosed monthly after an initial loading dose (Blauvelt A et al. Dermatol Ther (Heidelb).
With several biologic options available, patients may be switched from one biologic to another if they are not having an adequate response. Separate studies have shown that patients with an inadequate response to ustekinumab are likely to have a better response when switched to brodalumab or guselkumab, although this does not tell us if one is more likely to be effective than the other. In a recent matching-adjusted indirect comparison study using data for patients with psoriasis who responded inadequately to ustekinumab and switched to receive brodalumab (n=121) or guselkumab (n=135) the authors found that brodalumab was associated with greater improvements than guselkumab in inadequate responders to ustekinumab with a PASI 100 rate at week 36 of 40.3% for brodalumab vs 20.0% for guselkumab; P < 0.001. (Hampton P et al. Psoriasis (Auckl).
While biologics have revolutionized the treatment of psoriasis, not all patients with extensive disease desire or are appropriate for a systemic therapy. Topical steroids, the most commonly used topical psoriasis therapy, still carry some risk of systemic absorption and are associate with cutaneous side effects such as atrophy with prolonged use. Tarpinarof 1% cream is a novel aryl hydrocarbon receptor modulating agent that has been shown in phase 3 studies to be an effective treatment for psoriasis when applied once a day. A recent open label study of 21 adult patients with extensive plaque psoriasis (20% or more body surface area (BSA) involvement, mean baseline BSA 27.2%) who applied tapinarof cream 1% QD daily showed that 94.7% of patients had a decrease in their PASI score (mean PASI decrease of 59.6%). Despite the large BSA being treated, tapinarof plasma concentration were low and remained below the quantification level in the majority (67.9%) of samples tested. There were also no concerning EKG changes such as QT prolongation. Folliculitis and headache were the most common adverse events (each reported by 4 patients) (Jett JE et al. Am J Clin Dermatol).
These studies all provide valuable data in helping us to make the best treatment decisions for patients with moderate to severe plaque psoriasis.
A retrospective observational study of nearly 1200 prior biologic-experienced adult patients with psoriasis now receiving either secukinumab (n=780) or ixekizumab (n=411), both of which inhibit IL-17A, examined drug persistence after 18 months. Ixekizumab was associated with significantly higher rates of high treatment adherence (42% vs 35%; P = .019) and persistence (44.9% vs 36.9%; P = .007) and lower discontinuation (48.4% vs 56.0%; P = .018) and switching (26.6% vs 34.0%; P = .009) rates than secukinumab. Both drugs are dosed monthly after an initial loading dose (Blauvelt A et al. Dermatol Ther (Heidelb).
With several biologic options available, patients may be switched from one biologic to another if they are not having an adequate response. Separate studies have shown that patients with an inadequate response to ustekinumab are likely to have a better response when switched to brodalumab or guselkumab, although this does not tell us if one is more likely to be effective than the other. In a recent matching-adjusted indirect comparison study using data for patients with psoriasis who responded inadequately to ustekinumab and switched to receive brodalumab (n=121) or guselkumab (n=135) the authors found that brodalumab was associated with greater improvements than guselkumab in inadequate responders to ustekinumab with a PASI 100 rate at week 36 of 40.3% for brodalumab vs 20.0% for guselkumab; P < 0.001. (Hampton P et al. Psoriasis (Auckl).
While biologics have revolutionized the treatment of psoriasis, not all patients with extensive disease desire or are appropriate for a systemic therapy. Topical steroids, the most commonly used topical psoriasis therapy, still carry some risk of systemic absorption and are associate with cutaneous side effects such as atrophy with prolonged use. Tarpinarof 1% cream is a novel aryl hydrocarbon receptor modulating agent that has been shown in phase 3 studies to be an effective treatment for psoriasis when applied once a day. A recent open label study of 21 adult patients with extensive plaque psoriasis (20% or more body surface area (BSA) involvement, mean baseline BSA 27.2%) who applied tapinarof cream 1% QD daily showed that 94.7% of patients had a decrease in their PASI score (mean PASI decrease of 59.6%). Despite the large BSA being treated, tapinarof plasma concentration were low and remained below the quantification level in the majority (67.9%) of samples tested. There were also no concerning EKG changes such as QT prolongation. Folliculitis and headache were the most common adverse events (each reported by 4 patients) (Jett JE et al. Am J Clin Dermatol).
These studies all provide valuable data in helping us to make the best treatment decisions for patients with moderate to severe plaque psoriasis.
A retrospective observational study of nearly 1200 prior biologic-experienced adult patients with psoriasis now receiving either secukinumab (n=780) or ixekizumab (n=411), both of which inhibit IL-17A, examined drug persistence after 18 months. Ixekizumab was associated with significantly higher rates of high treatment adherence (42% vs 35%; P = .019) and persistence (44.9% vs 36.9%; P = .007) and lower discontinuation (48.4% vs 56.0%; P = .018) and switching (26.6% vs 34.0%; P = .009) rates than secukinumab. Both drugs are dosed monthly after an initial loading dose (Blauvelt A et al. Dermatol Ther (Heidelb).
With several biologic options available, patients may be switched from one biologic to another if they are not having an adequate response. Separate studies have shown that patients with an inadequate response to ustekinumab are likely to have a better response when switched to brodalumab or guselkumab, although this does not tell us if one is more likely to be effective than the other. In a recent matching-adjusted indirect comparison study using data for patients with psoriasis who responded inadequately to ustekinumab and switched to receive brodalumab (n=121) or guselkumab (n=135) the authors found that brodalumab was associated with greater improvements than guselkumab in inadequate responders to ustekinumab with a PASI 100 rate at week 36 of 40.3% for brodalumab vs 20.0% for guselkumab; P < 0.001. (Hampton P et al. Psoriasis (Auckl).
While biologics have revolutionized the treatment of psoriasis, not all patients with extensive disease desire or are appropriate for a systemic therapy. Topical steroids, the most commonly used topical psoriasis therapy, still carry some risk of systemic absorption and are associate with cutaneous side effects such as atrophy with prolonged use. Tarpinarof 1% cream is a novel aryl hydrocarbon receptor modulating agent that has been shown in phase 3 studies to be an effective treatment for psoriasis when applied once a day. A recent open label study of 21 adult patients with extensive plaque psoriasis (20% or more body surface area (BSA) involvement, mean baseline BSA 27.2%) who applied tapinarof cream 1% QD daily showed that 94.7% of patients had a decrease in their PASI score (mean PASI decrease of 59.6%). Despite the large BSA being treated, tapinarof plasma concentration were low and remained below the quantification level in the majority (67.9%) of samples tested. There were also no concerning EKG changes such as QT prolongation. Folliculitis and headache were the most common adverse events (each reported by 4 patients) (Jett JE et al. Am J Clin Dermatol).
These studies all provide valuable data in helping us to make the best treatment decisions for patients with moderate to severe plaque psoriasis.
Acute Severe Urticaria From Minocycline
To the Editor:
Minocycline is a commonly prescribed semisynthetic tetracycline derivative used for long-term treatment of acne vulgaris.1 Given the continued popularity of minocycline and other tetracyclines in treating acne, more adverse side effects are being reported. We report a patient who experienced acute severe urticaria with angioedema from minocycline.
A 35-year-old woman with a history of acne vulgaris presented to the emergency department with urticaria and associated angioedema. Fifteen days after starting minocycline, she awoke with diffuse hives sparing only the abdomen that resolved with diphenhydramine. Later that day, she developed generalized pruritus, hives, and lip swelling. She received intravenous methylprednisolone, diphenhydramine, and famotidine in the emergency department. She returned to the emergency department the next day due to facial and lip swelling, diffuse urticaria that was most pronounced on the arms, and throat irritation. Intramuscular epinephrine was administered first followed by methylprednisolone, famotidine, and cetirizine. She was discharged and advised to start daily prednisone 50 mg and cetirizine 20 mg every evening.
She returned to the emergency department the following morning due to worsening generalized urticaria and angioedema of the lips. She denied any associated respiratory, joint, or gastrointestinal tract symptoms. She had several urticarial plaques on the scalp, face, and body (Figure), only sparing the abdomen. Her hives were erythematous, raised, pruritic, and blanching. There was no residual purpura, ecchymosis, or hyperpigmentation associated with the urticaria, and each lesion was present for less than 24 hours. There was no swelling on examination. Additionally, she was afebrile. The C4 level was 18 mg/dL (reference range, 15–45 mg/dL). She did not develop eosinophilia (absolute eosinophil count, 0/µL [reference range, 50–500/µL]), lymphocytosis (absolute lymphocyte count, 1300/µL [reference range, 1000–4800/µL]), or abnormal liver or renal function. She was hospitalized for 3 days with severe urticaria and required 7 days of prednisone 40 to 50 mg, fexofenadine 360 mg, and cetirizine 20 mg. A viral infection was considered as a possible etiology; however, she had no supporting signs or symptoms of an upper respiratory illness or other viral illness.
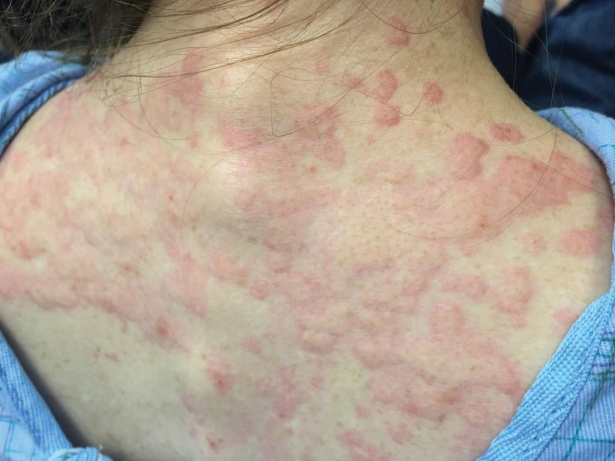
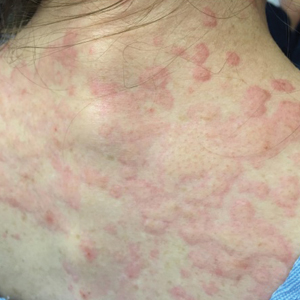
The patient’s minocycline use was considered the most likely etiology, as an oral contraceptive was the only other medication. She was labelled allergic to minocycline and discharged with intramuscular epinephrine. She was evaluated in the outpatient allergy immunology clinic 9 days later, and all her symptoms had resolved. Due to the severity of our patient’s reaction and the possibility of further severe reactions, an oral challenge was not carried out. Our patient was not interested in pursuing any further minocycline or other tetracycline-based therapy for her acne. She also was not interested in pursuing any minocycline skin-prick testing or oral challenge. One limitation to this case is our patient declining a confirmatory drug challenge; however, given the severity of the symptoms, the physicians involved agreed the patient's safety outweighed the benefits of confirmatory testing.
A PubMed search of articles indexed for MEDLINE and a Google Scholar search using the terms minocycline, drug hypersensitivity, urticaria, anaphylaxis, minocycline allergy, and angioedema yielded only 16 articles and correspondences. Reported adverse effects of minocycline included drug-induced lupus erythematosus, vasculitis, nausea, photosensitivity, and DRESS-like (drug reaction with eosinophilia and systemic symptoms syndrome) conditions. Three case reports of anaphylaxis/anaphylactoid reactions have been published,2-4 but cases of urticaria attributable to minocycline appear to be exceedingly rare.2,3 Reports of serum sickness in patients aged 15 to 62 years were rare. Women were noted to experience a higher frequency of adverse effects compared to men.5 Symptoms typically presented 3 to 28 days after initiation of minocycline. Data currently suggest that the pathogenesis of hypersensitivity reactions to minocycline remains unknown6; however, one hypothesis is that minocycline or its metabolites act as a superantigen, resulting in lymphocyte overactivation and massive cytokine release.7
Minocycline generally is well tolerated by patients. Physicians should be aware that minocycline is a possible causative agent of allergic drug reactions. Our patient’s presentation of severe acute urticaria with angioedema of the face and lips is a rarity.
- Levenson T, Masood D, Patterson R. Minocycline-induced serum sickness. Allergy Asthma Proc. 1996;17:79-81.
- Okano M, Imai S. Anaphylactoid symptoms due to oral minocycline. Acta Derm Venereol. 1996;76:164.
- Jang JW, Bae Y-J, Kim YG, et al. A case of anaphylaxis to oral minocycline. J Korean Med Sci. 2010;25:1233.
- Nakamura R, Tanaka A, Kinoshita H, et al. Minocycline-induced anaphylaxis mediated by antigen-specific immunoglobulin E [published online November 9, 2021]. J Dermatol. doi:10.1111/1346-8138.16228
- MacNeil M, Haase DA, Tremaine R, et al. Fever, lymphadenopathy, eosinophilia, lymphocytosis, hepatitis, and dermatitis: a severe adverse reaction to minocycline. J Am Acad Dermatol. 1997;36:347-350.
- DePaz S, Perez A, Gomez M, et al. Severe hypersensitivity reaction to minocycline. J Invest Allergol Clin Immunol. 1999;9:403-404.
- Somech R, Arav-Boger R, Assia A, et al. Complications of minocycline therapy for acne vulgaris: case reports and review of the literature. Pediatr Dermatol. 1999;16:469-472.
To the Editor:
Minocycline is a commonly prescribed semisynthetic tetracycline derivative used for long-term treatment of acne vulgaris.1 Given the continued popularity of minocycline and other tetracyclines in treating acne, more adverse side effects are being reported. We report a patient who experienced acute severe urticaria with angioedema from minocycline.
A 35-year-old woman with a history of acne vulgaris presented to the emergency department with urticaria and associated angioedema. Fifteen days after starting minocycline, she awoke with diffuse hives sparing only the abdomen that resolved with diphenhydramine. Later that day, she developed generalized pruritus, hives, and lip swelling. She received intravenous methylprednisolone, diphenhydramine, and famotidine in the emergency department. She returned to the emergency department the next day due to facial and lip swelling, diffuse urticaria that was most pronounced on the arms, and throat irritation. Intramuscular epinephrine was administered first followed by methylprednisolone, famotidine, and cetirizine. She was discharged and advised to start daily prednisone 50 mg and cetirizine 20 mg every evening.
She returned to the emergency department the following morning due to worsening generalized urticaria and angioedema of the lips. She denied any associated respiratory, joint, or gastrointestinal tract symptoms. She had several urticarial plaques on the scalp, face, and body (Figure), only sparing the abdomen. Her hives were erythematous, raised, pruritic, and blanching. There was no residual purpura, ecchymosis, or hyperpigmentation associated with the urticaria, and each lesion was present for less than 24 hours. There was no swelling on examination. Additionally, she was afebrile. The C4 level was 18 mg/dL (reference range, 15–45 mg/dL). She did not develop eosinophilia (absolute eosinophil count, 0/µL [reference range, 50–500/µL]), lymphocytosis (absolute lymphocyte count, 1300/µL [reference range, 1000–4800/µL]), or abnormal liver or renal function. She was hospitalized for 3 days with severe urticaria and required 7 days of prednisone 40 to 50 mg, fexofenadine 360 mg, and cetirizine 20 mg. A viral infection was considered as a possible etiology; however, she had no supporting signs or symptoms of an upper respiratory illness or other viral illness.

The patient’s minocycline use was considered the most likely etiology, as an oral contraceptive was the only other medication. She was labelled allergic to minocycline and discharged with intramuscular epinephrine. She was evaluated in the outpatient allergy immunology clinic 9 days later, and all her symptoms had resolved. Due to the severity of our patient’s reaction and the possibility of further severe reactions, an oral challenge was not carried out. Our patient was not interested in pursuing any further minocycline or other tetracycline-based therapy for her acne. She also was not interested in pursuing any minocycline skin-prick testing or oral challenge. One limitation to this case is our patient declining a confirmatory drug challenge; however, given the severity of the symptoms, the physicians involved agreed the patient's safety outweighed the benefits of confirmatory testing.
A PubMed search of articles indexed for MEDLINE and a Google Scholar search using the terms minocycline, drug hypersensitivity, urticaria, anaphylaxis, minocycline allergy, and angioedema yielded only 16 articles and correspondences. Reported adverse effects of minocycline included drug-induced lupus erythematosus, vasculitis, nausea, photosensitivity, and DRESS-like (drug reaction with eosinophilia and systemic symptoms syndrome) conditions. Three case reports of anaphylaxis/anaphylactoid reactions have been published,2-4 but cases of urticaria attributable to minocycline appear to be exceedingly rare.2,3 Reports of serum sickness in patients aged 15 to 62 years were rare. Women were noted to experience a higher frequency of adverse effects compared to men.5 Symptoms typically presented 3 to 28 days after initiation of minocycline. Data currently suggest that the pathogenesis of hypersensitivity reactions to minocycline remains unknown6; however, one hypothesis is that minocycline or its metabolites act as a superantigen, resulting in lymphocyte overactivation and massive cytokine release.7
Minocycline generally is well tolerated by patients. Physicians should be aware that minocycline is a possible causative agent of allergic drug reactions. Our patient’s presentation of severe acute urticaria with angioedema of the face and lips is a rarity.
To the Editor:
Minocycline is a commonly prescribed semisynthetic tetracycline derivative used for long-term treatment of acne vulgaris.1 Given the continued popularity of minocycline and other tetracyclines in treating acne, more adverse side effects are being reported. We report a patient who experienced acute severe urticaria with angioedema from minocycline.
A 35-year-old woman with a history of acne vulgaris presented to the emergency department with urticaria and associated angioedema. Fifteen days after starting minocycline, she awoke with diffuse hives sparing only the abdomen that resolved with diphenhydramine. Later that day, she developed generalized pruritus, hives, and lip swelling. She received intravenous methylprednisolone, diphenhydramine, and famotidine in the emergency department. She returned to the emergency department the next day due to facial and lip swelling, diffuse urticaria that was most pronounced on the arms, and throat irritation. Intramuscular epinephrine was administered first followed by methylprednisolone, famotidine, and cetirizine. She was discharged and advised to start daily prednisone 50 mg and cetirizine 20 mg every evening.
She returned to the emergency department the following morning due to worsening generalized urticaria and angioedema of the lips. She denied any associated respiratory, joint, or gastrointestinal tract symptoms. She had several urticarial plaques on the scalp, face, and body (Figure), only sparing the abdomen. Her hives were erythematous, raised, pruritic, and blanching. There was no residual purpura, ecchymosis, or hyperpigmentation associated with the urticaria, and each lesion was present for less than 24 hours. There was no swelling on examination. Additionally, she was afebrile. The C4 level was 18 mg/dL (reference range, 15–45 mg/dL). She did not develop eosinophilia (absolute eosinophil count, 0/µL [reference range, 50–500/µL]), lymphocytosis (absolute lymphocyte count, 1300/µL [reference range, 1000–4800/µL]), or abnormal liver or renal function. She was hospitalized for 3 days with severe urticaria and required 7 days of prednisone 40 to 50 mg, fexofenadine 360 mg, and cetirizine 20 mg. A viral infection was considered as a possible etiology; however, she had no supporting signs or symptoms of an upper respiratory illness or other viral illness.

The patient’s minocycline use was considered the most likely etiology, as an oral contraceptive was the only other medication. She was labelled allergic to minocycline and discharged with intramuscular epinephrine. She was evaluated in the outpatient allergy immunology clinic 9 days later, and all her symptoms had resolved. Due to the severity of our patient’s reaction and the possibility of further severe reactions, an oral challenge was not carried out. Our patient was not interested in pursuing any further minocycline or other tetracycline-based therapy for her acne. She also was not interested in pursuing any minocycline skin-prick testing or oral challenge. One limitation to this case is our patient declining a confirmatory drug challenge; however, given the severity of the symptoms, the physicians involved agreed the patient's safety outweighed the benefits of confirmatory testing.
A PubMed search of articles indexed for MEDLINE and a Google Scholar search using the terms minocycline, drug hypersensitivity, urticaria, anaphylaxis, minocycline allergy, and angioedema yielded only 16 articles and correspondences. Reported adverse effects of minocycline included drug-induced lupus erythematosus, vasculitis, nausea, photosensitivity, and DRESS-like (drug reaction with eosinophilia and systemic symptoms syndrome) conditions. Three case reports of anaphylaxis/anaphylactoid reactions have been published,2-4 but cases of urticaria attributable to minocycline appear to be exceedingly rare.2,3 Reports of serum sickness in patients aged 15 to 62 years were rare. Women were noted to experience a higher frequency of adverse effects compared to men.5 Symptoms typically presented 3 to 28 days after initiation of minocycline. Data currently suggest that the pathogenesis of hypersensitivity reactions to minocycline remains unknown6; however, one hypothesis is that minocycline or its metabolites act as a superantigen, resulting in lymphocyte overactivation and massive cytokine release.7
Minocycline generally is well tolerated by patients. Physicians should be aware that minocycline is a possible causative agent of allergic drug reactions. Our patient’s presentation of severe acute urticaria with angioedema of the face and lips is a rarity.
- Levenson T, Masood D, Patterson R. Minocycline-induced serum sickness. Allergy Asthma Proc. 1996;17:79-81.
- Okano M, Imai S. Anaphylactoid symptoms due to oral minocycline. Acta Derm Venereol. 1996;76:164.
- Jang JW, Bae Y-J, Kim YG, et al. A case of anaphylaxis to oral minocycline. J Korean Med Sci. 2010;25:1233.
- Nakamura R, Tanaka A, Kinoshita H, et al. Minocycline-induced anaphylaxis mediated by antigen-specific immunoglobulin E [published online November 9, 2021]. J Dermatol. doi:10.1111/1346-8138.16228
- MacNeil M, Haase DA, Tremaine R, et al. Fever, lymphadenopathy, eosinophilia, lymphocytosis, hepatitis, and dermatitis: a severe adverse reaction to minocycline. J Am Acad Dermatol. 1997;36:347-350.
- DePaz S, Perez A, Gomez M, et al. Severe hypersensitivity reaction to minocycline. J Invest Allergol Clin Immunol. 1999;9:403-404.
- Somech R, Arav-Boger R, Assia A, et al. Complications of minocycline therapy for acne vulgaris: case reports and review of the literature. Pediatr Dermatol. 1999;16:469-472.
- Levenson T, Masood D, Patterson R. Minocycline-induced serum sickness. Allergy Asthma Proc. 1996;17:79-81.
- Okano M, Imai S. Anaphylactoid symptoms due to oral minocycline. Acta Derm Venereol. 1996;76:164.
- Jang JW, Bae Y-J, Kim YG, et al. A case of anaphylaxis to oral minocycline. J Korean Med Sci. 2010;25:1233.
- Nakamura R, Tanaka A, Kinoshita H, et al. Minocycline-induced anaphylaxis mediated by antigen-specific immunoglobulin E [published online November 9, 2021]. J Dermatol. doi:10.1111/1346-8138.16228
- MacNeil M, Haase DA, Tremaine R, et al. Fever, lymphadenopathy, eosinophilia, lymphocytosis, hepatitis, and dermatitis: a severe adverse reaction to minocycline. J Am Acad Dermatol. 1997;36:347-350.
- DePaz S, Perez A, Gomez M, et al. Severe hypersensitivity reaction to minocycline. J Invest Allergol Clin Immunol. 1999;9:403-404.
- Somech R, Arav-Boger R, Assia A, et al. Complications of minocycline therapy for acne vulgaris: case reports and review of the literature. Pediatr Dermatol. 1999;16:469-472.
Practice Points
- Minocycline is a commonly prescribed long-term treatment for acne vulgaris.
- Minocycline-induced acute urticaria and anaphylaxis are rare adverse events.
First Omicron variant case identified in U.S.
He or she was fully vaccinated against COVID-19 and experienced only “mild symptoms that are improving,” officials with the Centers for Disease Control and Prevention said.
The patient, who was not named in the CDC’s announcement of the first U.S. case of the Omicron variant Dec. 1, is self-quarantining.
“All close contacts have been contacted and have tested negative,” officials said.
The announcement comes as no surprise to many as the Omicron variant, first identified in South Africa, has been reported in countries around the world in recent days. Hong Kong, the United Kingdom, and Germany each reported this variant, as have Italy and the Netherlands. Over the weekend, the first North American cases were identified in Canada.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, announced over the weekend that this newest variant was likely already in the United States, telling ABC’s This Week its appearance here was “inevitable.”
Similar to previous variants, this new strain likely started circulating in the United States before scientists could do genetic tests to confirm its presence.
The World Health Organization named Omicron a “variant of concern” on Nov. 26, even though much remains unknown about how well it spreads, how severe it can be, and how it may resist vaccines. In the meantime, the United States enacted travel bans from multiple South African countries.
It remains to be seen if Omicron will follow the pattern of the Delta variant, which was first identified in the United States in May and became the dominant strain by July. It’s also possible it will follow the path taken by the Mu variant. Mu emerged in March and April to much concern, only to fizzle out by September because it was unable to compete with the Delta variant.
A version of this article first appeared on WebMD.com.
He or she was fully vaccinated against COVID-19 and experienced only “mild symptoms that are improving,” officials with the Centers for Disease Control and Prevention said.
The patient, who was not named in the CDC’s announcement of the first U.S. case of the Omicron variant Dec. 1, is self-quarantining.
“All close contacts have been contacted and have tested negative,” officials said.
The announcement comes as no surprise to many as the Omicron variant, first identified in South Africa, has been reported in countries around the world in recent days. Hong Kong, the United Kingdom, and Germany each reported this variant, as have Italy and the Netherlands. Over the weekend, the first North American cases were identified in Canada.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, announced over the weekend that this newest variant was likely already in the United States, telling ABC’s This Week its appearance here was “inevitable.”
Similar to previous variants, this new strain likely started circulating in the United States before scientists could do genetic tests to confirm its presence.
The World Health Organization named Omicron a “variant of concern” on Nov. 26, even though much remains unknown about how well it spreads, how severe it can be, and how it may resist vaccines. In the meantime, the United States enacted travel bans from multiple South African countries.
It remains to be seen if Omicron will follow the pattern of the Delta variant, which was first identified in the United States in May and became the dominant strain by July. It’s also possible it will follow the path taken by the Mu variant. Mu emerged in March and April to much concern, only to fizzle out by September because it was unable to compete with the Delta variant.
A version of this article first appeared on WebMD.com.
He or she was fully vaccinated against COVID-19 and experienced only “mild symptoms that are improving,” officials with the Centers for Disease Control and Prevention said.
The patient, who was not named in the CDC’s announcement of the first U.S. case of the Omicron variant Dec. 1, is self-quarantining.
“All close contacts have been contacted and have tested negative,” officials said.
The announcement comes as no surprise to many as the Omicron variant, first identified in South Africa, has been reported in countries around the world in recent days. Hong Kong, the United Kingdom, and Germany each reported this variant, as have Italy and the Netherlands. Over the weekend, the first North American cases were identified in Canada.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, announced over the weekend that this newest variant was likely already in the United States, telling ABC’s This Week its appearance here was “inevitable.”
Similar to previous variants, this new strain likely started circulating in the United States before scientists could do genetic tests to confirm its presence.
The World Health Organization named Omicron a “variant of concern” on Nov. 26, even though much remains unknown about how well it spreads, how severe it can be, and how it may resist vaccines. In the meantime, the United States enacted travel bans from multiple South African countries.
It remains to be seen if Omicron will follow the pattern of the Delta variant, which was first identified in the United States in May and became the dominant strain by July. It’s also possible it will follow the path taken by the Mu variant. Mu emerged in March and April to much concern, only to fizzle out by September because it was unable to compete with the Delta variant.
A version of this article first appeared on WebMD.com.
Moderna warns of material drop in vaccine efficacy against Omicron
“There is no world, I think, where [the effectiveness] is the same level … we had with Delta,” Stephane Bancel told the Financial Times .
“I think it’s going to be a material drop,” he said. “I just don’t know how much, because we need to wait for the data. But all the scientists I’ve talked to … are like, ‘This is not going to be good.’”
Vaccine companies are now studying whether the new Omicron variant could evade the current shots. Some data is expected in about 2 weeks.
Mr. Bancel said that if a new vaccine is needed, it could take several months to produce at scale. He estimated that Moderna could make billions of vaccine doses in 2022.
“[Moderna] and Pfizer cannot get a billion doses next week. The math doesn’t work,” he said. “But could we get the billion doses out by the summer? Sure.”
The news caused some panic on Nov. 30, prompting financial markets to fall sharply, according to Reuters. But the markets recovered after European officials gave a more reassuring outlook.
“Even if the new variant becomes more widespread, the vaccines we have will continue to provide protection,” Emer Cooke, executive director of the European Medicines Agency, told the European Parliament.
Mr. Cooke said the agency could approve new vaccines that target the Omicron variant within 3 to 4 months, if needed. Moderna and Pfizer have announced they are beginning to tailor a shot to address the Omicron variant in case the data shows they are necessary.
Also on Nov. 30, the European Centre for Disease Prevention and Control announced that 42 Omicron cases had been identified in 10 European Union countries, according to Reuters.
The cases were mild or had no symptoms, although they were found in younger people who may have mild or no symptoms anyway.
“For the assessment of whether [Omicron] escapes immunity, we still have to wait until investigations in the laboratories with [blood samples] from people who have recovered have been carried out,” Andrea Ammon, MD, chair of the agency, said during an online conference.
The University of Oxford, which developed a COVID-19 vaccine with AstraZeneca, said Nov. 30 that there’s no evidence that vaccines won’t prevent severe disease from the Omicron variant, according to Reuters.
“Despite the appearance of new variants over the past year, vaccines have continued to provide very high levels of protection against severe disease and there is no evidence so far that Omicron is any different,” the university said in a statement. “However, we have the necessary tools and processes in place for rapid development of an updated COVID-19 vaccine if it should be necessary.”
A version of this article first appeared on WebMD.com.
“There is no world, I think, where [the effectiveness] is the same level … we had with Delta,” Stephane Bancel told the Financial Times .
“I think it’s going to be a material drop,” he said. “I just don’t know how much, because we need to wait for the data. But all the scientists I’ve talked to … are like, ‘This is not going to be good.’”
Vaccine companies are now studying whether the new Omicron variant could evade the current shots. Some data is expected in about 2 weeks.
Mr. Bancel said that if a new vaccine is needed, it could take several months to produce at scale. He estimated that Moderna could make billions of vaccine doses in 2022.
“[Moderna] and Pfizer cannot get a billion doses next week. The math doesn’t work,” he said. “But could we get the billion doses out by the summer? Sure.”
The news caused some panic on Nov. 30, prompting financial markets to fall sharply, according to Reuters. But the markets recovered after European officials gave a more reassuring outlook.
“Even if the new variant becomes more widespread, the vaccines we have will continue to provide protection,” Emer Cooke, executive director of the European Medicines Agency, told the European Parliament.
Mr. Cooke said the agency could approve new vaccines that target the Omicron variant within 3 to 4 months, if needed. Moderna and Pfizer have announced they are beginning to tailor a shot to address the Omicron variant in case the data shows they are necessary.
Also on Nov. 30, the European Centre for Disease Prevention and Control announced that 42 Omicron cases had been identified in 10 European Union countries, according to Reuters.
The cases were mild or had no symptoms, although they were found in younger people who may have mild or no symptoms anyway.
“For the assessment of whether [Omicron] escapes immunity, we still have to wait until investigations in the laboratories with [blood samples] from people who have recovered have been carried out,” Andrea Ammon, MD, chair of the agency, said during an online conference.
The University of Oxford, which developed a COVID-19 vaccine with AstraZeneca, said Nov. 30 that there’s no evidence that vaccines won’t prevent severe disease from the Omicron variant, according to Reuters.
“Despite the appearance of new variants over the past year, vaccines have continued to provide very high levels of protection against severe disease and there is no evidence so far that Omicron is any different,” the university said in a statement. “However, we have the necessary tools and processes in place for rapid development of an updated COVID-19 vaccine if it should be necessary.”
A version of this article first appeared on WebMD.com.
“There is no world, I think, where [the effectiveness] is the same level … we had with Delta,” Stephane Bancel told the Financial Times .
“I think it’s going to be a material drop,” he said. “I just don’t know how much, because we need to wait for the data. But all the scientists I’ve talked to … are like, ‘This is not going to be good.’”
Vaccine companies are now studying whether the new Omicron variant could evade the current shots. Some data is expected in about 2 weeks.
Mr. Bancel said that if a new vaccine is needed, it could take several months to produce at scale. He estimated that Moderna could make billions of vaccine doses in 2022.
“[Moderna] and Pfizer cannot get a billion doses next week. The math doesn’t work,” he said. “But could we get the billion doses out by the summer? Sure.”
The news caused some panic on Nov. 30, prompting financial markets to fall sharply, according to Reuters. But the markets recovered after European officials gave a more reassuring outlook.
“Even if the new variant becomes more widespread, the vaccines we have will continue to provide protection,” Emer Cooke, executive director of the European Medicines Agency, told the European Parliament.
Mr. Cooke said the agency could approve new vaccines that target the Omicron variant within 3 to 4 months, if needed. Moderna and Pfizer have announced they are beginning to tailor a shot to address the Omicron variant in case the data shows they are necessary.
Also on Nov. 30, the European Centre for Disease Prevention and Control announced that 42 Omicron cases had been identified in 10 European Union countries, according to Reuters.
The cases were mild or had no symptoms, although they were found in younger people who may have mild or no symptoms anyway.
“For the assessment of whether [Omicron] escapes immunity, we still have to wait until investigations in the laboratories with [blood samples] from people who have recovered have been carried out,” Andrea Ammon, MD, chair of the agency, said during an online conference.
The University of Oxford, which developed a COVID-19 vaccine with AstraZeneca, said Nov. 30 that there’s no evidence that vaccines won’t prevent severe disease from the Omicron variant, according to Reuters.
“Despite the appearance of new variants over the past year, vaccines have continued to provide very high levels of protection against severe disease and there is no evidence so far that Omicron is any different,” the university said in a statement. “However, we have the necessary tools and processes in place for rapid development of an updated COVID-19 vaccine if it should be necessary.”
A version of this article first appeared on WebMD.com.
Association of height, BMI, and AD in young children may be transient
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM JAMA DERMATOLOGY
A Starter Guide to Immunofluorescence Testing in Dermatology
Direct immunofluorescence (DIF) is the go-to diagnostic test when evaluating vesiculobullous eruptions, connective tissue disease, and vasculitis. This specialized test allows visualization of autoantibodies and their reaction products in the epidermis and dermis (skin) and epithelium and subepithelium (mucosa). Indirect immunofluorescence (IIF) and enzyme-linked immunosorbent assay (ELISA) are additional tests that can help in the diagnosis of autoimmune blistering disease. In the blistering autoimmune diseases, the autoantibodies target components in skin and mucous membranes that are essential for cell-cell and cell-matrix adhesion causing separation within or beneath the epidermis, depending on where the target components are located. This article is intended to serve as a helpful primer for immunofluorescence testing in dermatology, with an overview of the tests available as well as pragmatic tips for optimal biopsy sites and specimen transport.
Direct Immunofluorescence
Immunofluorescence techniques date back to 1941 when Albert Coons, an American physician, pathologist, and immunologist, fluorescently labelled antibodies to visualize pneumococcal antigens in infected tissues.1-3 In dermatology, similar methodology was used to visualize the deposition of immunoglobulins and complement in the skin of patients with systemic lupus erythematosus in 1963.4 Basement membrane zone antibodies were first visualized via DIF in bullous pemphigoid in 1967.5 This elegant test utilizes specific antibodies labeled with fluorophores that are then incubated with the patient’s tissue, ultimately forming antibody-antigen conjugates that can be visualized with a fluorescent microscope. Antibodies usually include IgG, IgM, IgA, fibrinogen, and C3. Some institutions also evaluate for IgG4.
Transport medium is critical for proper evaluation of tissues using DIF. Inappropriate storage of tissue can degrade the antigen and confuse the interpretation of specimens. An acceptable medium for DIF includes Michel transport medium, which allows tissue to be stored for days while being transported at ambient temperature without loss of signal.6,7 Zeus medium also can be used and is more readily available. Alternatively, biopsy tissue can be snap frozen using liquid nitrogen. Specimens also may be stored on saline gauze but should be analyzed within 24 to 48 hours.8 Most importantly, do not place the specimen in formalin; even a brief soak in formalin can greatly alter results, especially when trying to diagnose pemphigus.9 Proper transport conditions are critical to prevent autolysis, mitigate putrefaction, and preserve morphology while maintaining antigenicity.10
Indirect Immunofluorescence
Indirect immunofluorescence can be helpful for detecting antibodies circulating in patient serum. Indirect immunofluorescence can be used to help diagnose pemphigoid, pemphigus, epidermolysis bullosa acquisita, bullous lupus erythematosus, and dermatitis herpetiformis. Serum testing also can be a helpful alternative when obtaining tissue is difficult, such as in children.
Indirect immunofluorescence is a 2-part technique that takes a bit longer to assay than DIF.11 The first step involves incubating prepared tissue substrates with patient serum. Unlabeled antibodies in the patient serum are allowed to bind to antigens in the substrate tissue for about 30 minutes. Doubling dilutions of patient serum can be performed to titer antibody levels. The second step uses fluorescein-labeled antihuman antibodies to recognize the antigen-antibody conjugates. Normal whole tissues (eg, monkey esophagus for pemphigus vulgaris, rat bladder for paraneoplastic pemphigus, salt-split normal human skin substrate for pemphigoid and epidermolysis bullosa) are the usual substrates for testing.11,12 Again, this test requires serum and should be collected in a red-top tube or serum-separator tube. Usually, a minimum of 0.5 mL is required for testing, but check with your preferred immunodermatology send-out laboratory before collecting.13
Indirect immunofluorescence usually involves an initial screening panel using 1 or 2 tissue substrates followed by individual antigen-specific assays that correspond to the clinical suspicion and IIF screening results.11 Salt-split skin is used to localize basement membrane zone autoantibodies to either the epidermal (roof) or dermal (floor) side. Although many dermatopathology laboratories offer DIF testing, IIF is more specialized and may be a send-out test at your institution.
Enzyme-linked Immunosorbent Assays
Another tool in the immunodermatology armamentarium is ELISA. Commercial ELISA systems are available for the detection of autoantibodies against bullous pemphigoid (BP) antigen 180, BP230, type VII collagen, desmoglein (Dsg) 1, Dsg3, and envoplakin.11 This test allows semiquantitative measurement of antibody levels and thus can be used to monitor response to treatment or identify relapse and treatment failure.11 For example, in BP, significantly increased baseline anti-BP180 IgG levels correlate with 1-year mortality rates (P=.001) and relapse rates (P=.041).14,15 Numerous additional studies support the observation that monitoring anti-BP180 as a potential marker of disease relapse can be helpful.16,17 In pemphigus, the presence or increase of autoantibodies at remission, either anti-Dsg3 or anti-Dsg1, may be a useful tool in predicting disease relapse.18 It is important for physicians to be aware of this to be able to offer guidance on prognosis.
Where Should I Biopsy?
Knowing where to biopsy can be confusing when beginning residency. But the short answer is, it depends. Let your clinical suspicion guide your specimen site. The Figure provides a quick reference for which location will give you the highest yield for a specific diagnosis.

A few cardinal rules should guide which site is biopsied. Avoid obtaining specimens from the lower extremities as much as possible, as this site has been linked with false-negative results, especially in bullous pemphigoid.19,20 As a dependent area prone to stasis, this site gets a lot of abuse and inflammatory changes secondary to everyday insults that can theoretically alter DIF findings, especially fibrinogen deposition.
Although tissue sent for hematoxylin and eosin staining should be lesional, biopsy for DIF ideally should not contain a new or active blister, ulcer, erosion, or bulla. Immunoreactants are more likely to be degraded in these areas, and DIF may be falsely negative.21
It is worthwhile to briefly discuss the definitions of the terms perilesional and nonlesional. Perilesional skin most frequently refers to skin adjacent to a bulla or vesicle. This skin can be erythematous/inflamed or appear normal. When obtaining tissue for a diagnosis of blistering disease, the general recommendation is to obtain the biopsy from lesional nonbullous skin or perilesional uninvolved skin within 1 cm of the bulla.22-24 The only exception to this is dermatitis herpetiformis, which is best diagnosed on tissue obtained from normal-appearing perilesional skin within 1 cm of an active lesion.25 Additionally, if your patient has oral disease, the recommendation is to obtain the biopsy from nonlesional buccal mucosa, especially if there is desquamative gingivitis.26,27
The ideal biopsy size is 4 or 5 mm. If considering both DIF and histopathology, it is best to procure 2 separate specimens. One larger biopsy can be carefully bisected in 2 but often is subject to more handling artifacts, which can affect findings. In the case of 1 biopsy bisected into 2 specimens, the punch should be at least 6 mm. Shave biopsies also can be performed as long as they extend into the reticular dermis.23
For vasculitis, biopsies for DIF should be taken from lesions that are less than 24 hours old for highest yield, as the level of tissue immunoreactants tends to decline over time.28 This guideline does differ from hematoxylin and eosin specimens sent for evaluation of vasculitis, which ideally should be lesional tissue over 72 hours old. When evaluating for lupus (including subacute cutaneous lupus, discoid lupus, and systemic lupus), DIF is more likely to be positive in well-established, active lesions.
Which Test Should I Order?
The answer to this question depends, but the use of all 3 tests has a specificity close to 100% when evaluating for autoantibody-associated diseases.23 For autoimmune blistering disease, DIF is considered the diagnostic standard. The sensitivity of DIF for diagnosing BP is in the range of 82% to 90.5%, while specificity is 98%.29-31 Other autoimmune blistering diseases, such as pemphigus or dermatitis herpetiformis, have even higher sensitivities and specificities. Direct immunofluorescence often is used as a screening test, but false negatives do occur.32,33 Although rare, false positives also can occur, especially in cases of infection, and should be suspected when there is a lack of clinicopathologic correlation.34 If DIF is negative but clinical suspicion remains high, IIF should be ordered to directly evaluate a patient’s serum for autoantibodies.
In acute cutaneous lupus, subacute cutaneous lupus, and discoid lupus, DIF of active lesions may be helpful if histopathologic examination of a cutaneous lupus erythematosus lesion is nondiagnostic. However, histopathologic examination of formalin-fixed tissue remains the standard for these diagnoses. In vasculitis, while DIF is not used for diagnosis, it is useful to evaluate for IgA deposition. This is important in adults, as IgA deposition has been associated with a greater risk for developing end-stage renal disease.35
Final Thoughts
This is an overview of the tests available for diagnosing autoimmune blistering diseases. Residents should keep in mind that these tests are just one part of the puzzle when it comes to diagnosing these diseases. Results of DIF, IIF, and ELISA testing should be considered in conjunction with patient history and physical examination as well as histopathologic examination of lesional tissue when evaluating for dermatologic diseases with autoantibodies.
- Arthur G. Albert Coons: harnessing the power of the antibody. Lancet Respir Med. 2016;4:181-182.
- Coons AH, Creech HJ, Jones RN. Immunological properties of an antibody containing a fluorescent group. Proc Soc Exp Biol Med. 1941;47:200-202.
- Coons AH, Creech HJ, Jones RN, et al. The demonstration of pneumococcal antigen in tissues by the use of fluorescent antibody. J Immunol. 1942;45:159-170.
- Burnham TK, Neblett TR, Fine G. The application of the fluorescent antibody technic to the investigation of lupus erythematosus and various dermatoses. J Invest Dermatol. 1963;41:451-456.
- Jordon RE, Beutner EH, Witebsky E, et al. Basement zone antibodies in bullous pemphigoid. JAMA. 1967;200:751-756.
- Vaughan Jones SA, Salas J, McGrath JA, et al. A retrospective analysis of tissue-fixed immunoreactants from skin biopsies maintained in Michel’s medium. Dermatology. 1994;189(suppl 1):131-132.
- Kim RH, Brinster NK. Practical direct immunofluorescence. Am J Dermatopathol. 2020;42:75-85.
- Vodegel RM, de Jong MC, Meijer HJ, et al. Enhanced diagnostic immunofluorescence using biopsies transported in saline. BMC Dermatol. 2004;4:10.
- Arbesman J, Grover R, Helm TN, et al. Can direct immunofluorescence testing still be accurate if performed on biopsy specimens after brief inadvertent immersion in formalin? J Am Acad Dermatol. 2011;65:106-111.
- Im K, Mareninov S, Diaz MFP, et al. An introduction to performing immunofluorescence staining. Methods Mol Biol. 2019;1897:299-311.
- Saschenbrecker S, Karl I, Komorowski L, et al. Serological diagnosis of autoimmune bullous skin diseases. Front Immunol. 2019;10:1974.
- Baum S, Sakka N, Artsi O, et al. Diagnosis and classification of autoimmune blistering diseases. Autoimmun Rev. 2014;13:482-489.
- Immunobullous disease panel, epithelial. ARUP Laboratories website. Accessed November 22, 2021. https://ltd.aruplab.com/Tests/Pub/3001409
- Monshi B, Gulz L, Piringer B, et al. Anti-BP180 autoantibody levels at diagnosis correlate with 1-year mortality rates in patients with bullous pemphigoid. J Eur Acad Dermatol Venereol. 2020;34:1583-1589.
- Koga H, Teye K, Ishii N, et al. High index values of enzyme-linked immunosorbent assay for BP180 at baseline predict relapse in patients with bullous pemphigoid. Front Med (Lausanne). 2018;5:139.
- Fichel F, Barbe C, Joly P, et al. Clinical and immunologic factors associated with bullous pemphigoid relapse during the first year of treatment: a multicenter, prospective study. JAMA Dermatol. 2014;150:25-33.
- Cai SC, Lim YL, Li W, et al. Anti-BP180 NC16A IgG titres as an indicator of disease activity and outcome in Asian patients with bullous pemphigoid. Ann Acad Med Singap. 2015;44:119-126.
- Genovese G, Maronese CA, Casazza G, et al. Clinical and serological predictors of relapse in pemphigus: a study of 143 patients [published online July 20, 2021]. Clin Exp Dermatol. doi:10.1111/ced.14854
- Weigand DA. Effect of anatomic region on immunofluorescence diagnosis of bullous pemphigoid. J Am Acad Dermatol. 1985;12(2, pt 1):274-278.
- Weigand DA, Clements MK. Direct immunofluorescence in bullous pemphigoid: effects of extent and location of lesions. J Am Acad Dermatol. 1989;20:437-440.
- Mutasim DF, Adams BB. Immunofluorescence in dermatology. J Am Acad Dermatol. 2001;45:803-822; quiz 822-824.
- Sladden C, Kirchhof MG, Crawford RI. Biopsy location for direct immunofluorescence in patients with suspected bullous pemphigoid impacts probability of a positive test result. J Cutan Med Surg. 2014;18:392-396.
- Elston DM, Stratman EJ, Miller SJ. Skin biopsy: biopsy issues in specific diseases. J Am Acad Dermatol. 2016;74:1-16; quiz 17-18.
- Seishima M, Izumi T, Kitajima Y. Antibody to bullous pemphigoid antigen 1 binds to the antigen at perilesional but not uninvolved skin, in localized bullous pemphigoid. Eur J Dermatol. 1999;9:39-42.
- Zone JJ, Meyer LJ, Petersen MJ. Deposition of granular IgA relative to clinical lesions in dermatitis herpetiformis. Arch Dermatol. 1996;132:912-918.
- Kamaguchi M, Iwata H, Ujiie I, et al. Direct immunofluorescence using non-lesional buccal mucosa in mucous membrane pemphigoid. Front Med (Lausanne). 2018;5:20.
- Carey B, Joshi S, Abdelghani A, et al. The optimal oral biopsy site for diagnosis of mucous membrane pemphigoid and pemphigus vulgaris. Br J Dermatol. 2020;182:747-753.
- Kulthanan K, Pinkaew S, Jiamton S, et al. Cutaneous leukocytoclastic vasculitis: the yield of direct immunofluorescence study. J Med Assoc Thai. 2004;87:531-535.
- Chaidemenos GC, Maltezos E, Chrysomallis F, et al. Value of routine diagnostic criteria of bullous pemphigoid. Int J Dermatol. 1998;37:206-210.
- Mysorekar VV, Sumathy TK, Shyam Prasad AL. Role of direct immunofluorescence in dermatological disorders. Indian Dermatol Online J. 2015;6:172-180.
- Fudge JG, Crawford RI. Bullous pemphigoid: a 10-year study of discordant results on direct immunofluorescence. J Cutan Med Surg. 2018;22:472-475.
- Sárdy M, Kostaki D, Varga R, et al. Comparative study of direct and indirect immunofluorescence and of bullous pemphigoid 180 and 230 enzyme-linked immunosorbent assays for diagnosis of bullous pemphigoid. J Am Acad Dermatol. 2013;69:748-753.
- Buch AC, Kumar H, Panicker N, et al. A cross-sectional study of direct immunofluorescence in the diagnosis of immunobullous dermatoses. Indian J Dermatol. 2014;59:364-368.
- Miller DD, Bhawan J. Bullous tinea pedis with direct immunofluorescence positivity: when is a positive result not autoimmune bullous disease? Am J Dermatopathol. 2013;35:587-594.
- Cao R, Lau S, Tan V, et al. Adult Henoch-Schönlein purpura: clinical and histopathological predictors of systemic disease and profound renal disease. Indian J Dermatol Venereol Leprol. 2017;83:577-582.
Direct immunofluorescence (DIF) is the go-to diagnostic test when evaluating vesiculobullous eruptions, connective tissue disease, and vasculitis. This specialized test allows visualization of autoantibodies and their reaction products in the epidermis and dermis (skin) and epithelium and subepithelium (mucosa). Indirect immunofluorescence (IIF) and enzyme-linked immunosorbent assay (ELISA) are additional tests that can help in the diagnosis of autoimmune blistering disease. In the blistering autoimmune diseases, the autoantibodies target components in skin and mucous membranes that are essential for cell-cell and cell-matrix adhesion causing separation within or beneath the epidermis, depending on where the target components are located. This article is intended to serve as a helpful primer for immunofluorescence testing in dermatology, with an overview of the tests available as well as pragmatic tips for optimal biopsy sites and specimen transport.
Direct Immunofluorescence
Immunofluorescence techniques date back to 1941 when Albert Coons, an American physician, pathologist, and immunologist, fluorescently labelled antibodies to visualize pneumococcal antigens in infected tissues.1-3 In dermatology, similar methodology was used to visualize the deposition of immunoglobulins and complement in the skin of patients with systemic lupus erythematosus in 1963.4 Basement membrane zone antibodies were first visualized via DIF in bullous pemphigoid in 1967.5 This elegant test utilizes specific antibodies labeled with fluorophores that are then incubated with the patient’s tissue, ultimately forming antibody-antigen conjugates that can be visualized with a fluorescent microscope. Antibodies usually include IgG, IgM, IgA, fibrinogen, and C3. Some institutions also evaluate for IgG4.
Transport medium is critical for proper evaluation of tissues using DIF. Inappropriate storage of tissue can degrade the antigen and confuse the interpretation of specimens. An acceptable medium for DIF includes Michel transport medium, which allows tissue to be stored for days while being transported at ambient temperature without loss of signal.6,7 Zeus medium also can be used and is more readily available. Alternatively, biopsy tissue can be snap frozen using liquid nitrogen. Specimens also may be stored on saline gauze but should be analyzed within 24 to 48 hours.8 Most importantly, do not place the specimen in formalin; even a brief soak in formalin can greatly alter results, especially when trying to diagnose pemphigus.9 Proper transport conditions are critical to prevent autolysis, mitigate putrefaction, and preserve morphology while maintaining antigenicity.10
Indirect Immunofluorescence
Indirect immunofluorescence can be helpful for detecting antibodies circulating in patient serum. Indirect immunofluorescence can be used to help diagnose pemphigoid, pemphigus, epidermolysis bullosa acquisita, bullous lupus erythematosus, and dermatitis herpetiformis. Serum testing also can be a helpful alternative when obtaining tissue is difficult, such as in children.
Indirect immunofluorescence is a 2-part technique that takes a bit longer to assay than DIF.11 The first step involves incubating prepared tissue substrates with patient serum. Unlabeled antibodies in the patient serum are allowed to bind to antigens in the substrate tissue for about 30 minutes. Doubling dilutions of patient serum can be performed to titer antibody levels. The second step uses fluorescein-labeled antihuman antibodies to recognize the antigen-antibody conjugates. Normal whole tissues (eg, monkey esophagus for pemphigus vulgaris, rat bladder for paraneoplastic pemphigus, salt-split normal human skin substrate for pemphigoid and epidermolysis bullosa) are the usual substrates for testing.11,12 Again, this test requires serum and should be collected in a red-top tube or serum-separator tube. Usually, a minimum of 0.5 mL is required for testing, but check with your preferred immunodermatology send-out laboratory before collecting.13
Indirect immunofluorescence usually involves an initial screening panel using 1 or 2 tissue substrates followed by individual antigen-specific assays that correspond to the clinical suspicion and IIF screening results.11 Salt-split skin is used to localize basement membrane zone autoantibodies to either the epidermal (roof) or dermal (floor) side. Although many dermatopathology laboratories offer DIF testing, IIF is more specialized and may be a send-out test at your institution.
Enzyme-linked Immunosorbent Assays
Another tool in the immunodermatology armamentarium is ELISA. Commercial ELISA systems are available for the detection of autoantibodies against bullous pemphigoid (BP) antigen 180, BP230, type VII collagen, desmoglein (Dsg) 1, Dsg3, and envoplakin.11 This test allows semiquantitative measurement of antibody levels and thus can be used to monitor response to treatment or identify relapse and treatment failure.11 For example, in BP, significantly increased baseline anti-BP180 IgG levels correlate with 1-year mortality rates (P=.001) and relapse rates (P=.041).14,15 Numerous additional studies support the observation that monitoring anti-BP180 as a potential marker of disease relapse can be helpful.16,17 In pemphigus, the presence or increase of autoantibodies at remission, either anti-Dsg3 or anti-Dsg1, may be a useful tool in predicting disease relapse.18 It is important for physicians to be aware of this to be able to offer guidance on prognosis.
Where Should I Biopsy?
Knowing where to biopsy can be confusing when beginning residency. But the short answer is, it depends. Let your clinical suspicion guide your specimen site. The Figure provides a quick reference for which location will give you the highest yield for a specific diagnosis.

A few cardinal rules should guide which site is biopsied. Avoid obtaining specimens from the lower extremities as much as possible, as this site has been linked with false-negative results, especially in bullous pemphigoid.19,20 As a dependent area prone to stasis, this site gets a lot of abuse and inflammatory changes secondary to everyday insults that can theoretically alter DIF findings, especially fibrinogen deposition.
Although tissue sent for hematoxylin and eosin staining should be lesional, biopsy for DIF ideally should not contain a new or active blister, ulcer, erosion, or bulla. Immunoreactants are more likely to be degraded in these areas, and DIF may be falsely negative.21
It is worthwhile to briefly discuss the definitions of the terms perilesional and nonlesional. Perilesional skin most frequently refers to skin adjacent to a bulla or vesicle. This skin can be erythematous/inflamed or appear normal. When obtaining tissue for a diagnosis of blistering disease, the general recommendation is to obtain the biopsy from lesional nonbullous skin or perilesional uninvolved skin within 1 cm of the bulla.22-24 The only exception to this is dermatitis herpetiformis, which is best diagnosed on tissue obtained from normal-appearing perilesional skin within 1 cm of an active lesion.25 Additionally, if your patient has oral disease, the recommendation is to obtain the biopsy from nonlesional buccal mucosa, especially if there is desquamative gingivitis.26,27
The ideal biopsy size is 4 or 5 mm. If considering both DIF and histopathology, it is best to procure 2 separate specimens. One larger biopsy can be carefully bisected in 2 but often is subject to more handling artifacts, which can affect findings. In the case of 1 biopsy bisected into 2 specimens, the punch should be at least 6 mm. Shave biopsies also can be performed as long as they extend into the reticular dermis.23
For vasculitis, biopsies for DIF should be taken from lesions that are less than 24 hours old for highest yield, as the level of tissue immunoreactants tends to decline over time.28 This guideline does differ from hematoxylin and eosin specimens sent for evaluation of vasculitis, which ideally should be lesional tissue over 72 hours old. When evaluating for lupus (including subacute cutaneous lupus, discoid lupus, and systemic lupus), DIF is more likely to be positive in well-established, active lesions.
Which Test Should I Order?
The answer to this question depends, but the use of all 3 tests has a specificity close to 100% when evaluating for autoantibody-associated diseases.23 For autoimmune blistering disease, DIF is considered the diagnostic standard. The sensitivity of DIF for diagnosing BP is in the range of 82% to 90.5%, while specificity is 98%.29-31 Other autoimmune blistering diseases, such as pemphigus or dermatitis herpetiformis, have even higher sensitivities and specificities. Direct immunofluorescence often is used as a screening test, but false negatives do occur.32,33 Although rare, false positives also can occur, especially in cases of infection, and should be suspected when there is a lack of clinicopathologic correlation.34 If DIF is negative but clinical suspicion remains high, IIF should be ordered to directly evaluate a patient’s serum for autoantibodies.
In acute cutaneous lupus, subacute cutaneous lupus, and discoid lupus, DIF of active lesions may be helpful if histopathologic examination of a cutaneous lupus erythematosus lesion is nondiagnostic. However, histopathologic examination of formalin-fixed tissue remains the standard for these diagnoses. In vasculitis, while DIF is not used for diagnosis, it is useful to evaluate for IgA deposition. This is important in adults, as IgA deposition has been associated with a greater risk for developing end-stage renal disease.35
Final Thoughts
This is an overview of the tests available for diagnosing autoimmune blistering diseases. Residents should keep in mind that these tests are just one part of the puzzle when it comes to diagnosing these diseases. Results of DIF, IIF, and ELISA testing should be considered in conjunction with patient history and physical examination as well as histopathologic examination of lesional tissue when evaluating for dermatologic diseases with autoantibodies.
Direct immunofluorescence (DIF) is the go-to diagnostic test when evaluating vesiculobullous eruptions, connective tissue disease, and vasculitis. This specialized test allows visualization of autoantibodies and their reaction products in the epidermis and dermis (skin) and epithelium and subepithelium (mucosa). Indirect immunofluorescence (IIF) and enzyme-linked immunosorbent assay (ELISA) are additional tests that can help in the diagnosis of autoimmune blistering disease. In the blistering autoimmune diseases, the autoantibodies target components in skin and mucous membranes that are essential for cell-cell and cell-matrix adhesion causing separation within or beneath the epidermis, depending on where the target components are located. This article is intended to serve as a helpful primer for immunofluorescence testing in dermatology, with an overview of the tests available as well as pragmatic tips for optimal biopsy sites and specimen transport.
Direct Immunofluorescence
Immunofluorescence techniques date back to 1941 when Albert Coons, an American physician, pathologist, and immunologist, fluorescently labelled antibodies to visualize pneumococcal antigens in infected tissues.1-3 In dermatology, similar methodology was used to visualize the deposition of immunoglobulins and complement in the skin of patients with systemic lupus erythematosus in 1963.4 Basement membrane zone antibodies were first visualized via DIF in bullous pemphigoid in 1967.5 This elegant test utilizes specific antibodies labeled with fluorophores that are then incubated with the patient’s tissue, ultimately forming antibody-antigen conjugates that can be visualized with a fluorescent microscope. Antibodies usually include IgG, IgM, IgA, fibrinogen, and C3. Some institutions also evaluate for IgG4.
Transport medium is critical for proper evaluation of tissues using DIF. Inappropriate storage of tissue can degrade the antigen and confuse the interpretation of specimens. An acceptable medium for DIF includes Michel transport medium, which allows tissue to be stored for days while being transported at ambient temperature without loss of signal.6,7 Zeus medium also can be used and is more readily available. Alternatively, biopsy tissue can be snap frozen using liquid nitrogen. Specimens also may be stored on saline gauze but should be analyzed within 24 to 48 hours.8 Most importantly, do not place the specimen in formalin; even a brief soak in formalin can greatly alter results, especially when trying to diagnose pemphigus.9 Proper transport conditions are critical to prevent autolysis, mitigate putrefaction, and preserve morphology while maintaining antigenicity.10
Indirect Immunofluorescence
Indirect immunofluorescence can be helpful for detecting antibodies circulating in patient serum. Indirect immunofluorescence can be used to help diagnose pemphigoid, pemphigus, epidermolysis bullosa acquisita, bullous lupus erythematosus, and dermatitis herpetiformis. Serum testing also can be a helpful alternative when obtaining tissue is difficult, such as in children.
Indirect immunofluorescence is a 2-part technique that takes a bit longer to assay than DIF.11 The first step involves incubating prepared tissue substrates with patient serum. Unlabeled antibodies in the patient serum are allowed to bind to antigens in the substrate tissue for about 30 minutes. Doubling dilutions of patient serum can be performed to titer antibody levels. The second step uses fluorescein-labeled antihuman antibodies to recognize the antigen-antibody conjugates. Normal whole tissues (eg, monkey esophagus for pemphigus vulgaris, rat bladder for paraneoplastic pemphigus, salt-split normal human skin substrate for pemphigoid and epidermolysis bullosa) are the usual substrates for testing.11,12 Again, this test requires serum and should be collected in a red-top tube or serum-separator tube. Usually, a minimum of 0.5 mL is required for testing, but check with your preferred immunodermatology send-out laboratory before collecting.13
Indirect immunofluorescence usually involves an initial screening panel using 1 or 2 tissue substrates followed by individual antigen-specific assays that correspond to the clinical suspicion and IIF screening results.11 Salt-split skin is used to localize basement membrane zone autoantibodies to either the epidermal (roof) or dermal (floor) side. Although many dermatopathology laboratories offer DIF testing, IIF is more specialized and may be a send-out test at your institution.
Enzyme-linked Immunosorbent Assays
Another tool in the immunodermatology armamentarium is ELISA. Commercial ELISA systems are available for the detection of autoantibodies against bullous pemphigoid (BP) antigen 180, BP230, type VII collagen, desmoglein (Dsg) 1, Dsg3, and envoplakin.11 This test allows semiquantitative measurement of antibody levels and thus can be used to monitor response to treatment or identify relapse and treatment failure.11 For example, in BP, significantly increased baseline anti-BP180 IgG levels correlate with 1-year mortality rates (P=.001) and relapse rates (P=.041).14,15 Numerous additional studies support the observation that monitoring anti-BP180 as a potential marker of disease relapse can be helpful.16,17 In pemphigus, the presence or increase of autoantibodies at remission, either anti-Dsg3 or anti-Dsg1, may be a useful tool in predicting disease relapse.18 It is important for physicians to be aware of this to be able to offer guidance on prognosis.
Where Should I Biopsy?
Knowing where to biopsy can be confusing when beginning residency. But the short answer is, it depends. Let your clinical suspicion guide your specimen site. The Figure provides a quick reference for which location will give you the highest yield for a specific diagnosis.

A few cardinal rules should guide which site is biopsied. Avoid obtaining specimens from the lower extremities as much as possible, as this site has been linked with false-negative results, especially in bullous pemphigoid.19,20 As a dependent area prone to stasis, this site gets a lot of abuse and inflammatory changes secondary to everyday insults that can theoretically alter DIF findings, especially fibrinogen deposition.
Although tissue sent for hematoxylin and eosin staining should be lesional, biopsy for DIF ideally should not contain a new or active blister, ulcer, erosion, or bulla. Immunoreactants are more likely to be degraded in these areas, and DIF may be falsely negative.21
It is worthwhile to briefly discuss the definitions of the terms perilesional and nonlesional. Perilesional skin most frequently refers to skin adjacent to a bulla or vesicle. This skin can be erythematous/inflamed or appear normal. When obtaining tissue for a diagnosis of blistering disease, the general recommendation is to obtain the biopsy from lesional nonbullous skin or perilesional uninvolved skin within 1 cm of the bulla.22-24 The only exception to this is dermatitis herpetiformis, which is best diagnosed on tissue obtained from normal-appearing perilesional skin within 1 cm of an active lesion.25 Additionally, if your patient has oral disease, the recommendation is to obtain the biopsy from nonlesional buccal mucosa, especially if there is desquamative gingivitis.26,27
The ideal biopsy size is 4 or 5 mm. If considering both DIF and histopathology, it is best to procure 2 separate specimens. One larger biopsy can be carefully bisected in 2 but often is subject to more handling artifacts, which can affect findings. In the case of 1 biopsy bisected into 2 specimens, the punch should be at least 6 mm. Shave biopsies also can be performed as long as they extend into the reticular dermis.23
For vasculitis, biopsies for DIF should be taken from lesions that are less than 24 hours old for highest yield, as the level of tissue immunoreactants tends to decline over time.28 This guideline does differ from hematoxylin and eosin specimens sent for evaluation of vasculitis, which ideally should be lesional tissue over 72 hours old. When evaluating for lupus (including subacute cutaneous lupus, discoid lupus, and systemic lupus), DIF is more likely to be positive in well-established, active lesions.
Which Test Should I Order?
The answer to this question depends, but the use of all 3 tests has a specificity close to 100% when evaluating for autoantibody-associated diseases.23 For autoimmune blistering disease, DIF is considered the diagnostic standard. The sensitivity of DIF for diagnosing BP is in the range of 82% to 90.5%, while specificity is 98%.29-31 Other autoimmune blistering diseases, such as pemphigus or dermatitis herpetiformis, have even higher sensitivities and specificities. Direct immunofluorescence often is used as a screening test, but false negatives do occur.32,33 Although rare, false positives also can occur, especially in cases of infection, and should be suspected when there is a lack of clinicopathologic correlation.34 If DIF is negative but clinical suspicion remains high, IIF should be ordered to directly evaluate a patient’s serum for autoantibodies.
In acute cutaneous lupus, subacute cutaneous lupus, and discoid lupus, DIF of active lesions may be helpful if histopathologic examination of a cutaneous lupus erythematosus lesion is nondiagnostic. However, histopathologic examination of formalin-fixed tissue remains the standard for these diagnoses. In vasculitis, while DIF is not used for diagnosis, it is useful to evaluate for IgA deposition. This is important in adults, as IgA deposition has been associated with a greater risk for developing end-stage renal disease.35
Final Thoughts
This is an overview of the tests available for diagnosing autoimmune blistering diseases. Residents should keep in mind that these tests are just one part of the puzzle when it comes to diagnosing these diseases. Results of DIF, IIF, and ELISA testing should be considered in conjunction with patient history and physical examination as well as histopathologic examination of lesional tissue when evaluating for dermatologic diseases with autoantibodies.
- Arthur G. Albert Coons: harnessing the power of the antibody. Lancet Respir Med. 2016;4:181-182.
- Coons AH, Creech HJ, Jones RN. Immunological properties of an antibody containing a fluorescent group. Proc Soc Exp Biol Med. 1941;47:200-202.
- Coons AH, Creech HJ, Jones RN, et al. The demonstration of pneumococcal antigen in tissues by the use of fluorescent antibody. J Immunol. 1942;45:159-170.
- Burnham TK, Neblett TR, Fine G. The application of the fluorescent antibody technic to the investigation of lupus erythematosus and various dermatoses. J Invest Dermatol. 1963;41:451-456.
- Jordon RE, Beutner EH, Witebsky E, et al. Basement zone antibodies in bullous pemphigoid. JAMA. 1967;200:751-756.
- Vaughan Jones SA, Salas J, McGrath JA, et al. A retrospective analysis of tissue-fixed immunoreactants from skin biopsies maintained in Michel’s medium. Dermatology. 1994;189(suppl 1):131-132.
- Kim RH, Brinster NK. Practical direct immunofluorescence. Am J Dermatopathol. 2020;42:75-85.
- Vodegel RM, de Jong MC, Meijer HJ, et al. Enhanced diagnostic immunofluorescence using biopsies transported in saline. BMC Dermatol. 2004;4:10.
- Arbesman J, Grover R, Helm TN, et al. Can direct immunofluorescence testing still be accurate if performed on biopsy specimens after brief inadvertent immersion in formalin? J Am Acad Dermatol. 2011;65:106-111.
- Im K, Mareninov S, Diaz MFP, et al. An introduction to performing immunofluorescence staining. Methods Mol Biol. 2019;1897:299-311.
- Saschenbrecker S, Karl I, Komorowski L, et al. Serological diagnosis of autoimmune bullous skin diseases. Front Immunol. 2019;10:1974.
- Baum S, Sakka N, Artsi O, et al. Diagnosis and classification of autoimmune blistering diseases. Autoimmun Rev. 2014;13:482-489.
- Immunobullous disease panel, epithelial. ARUP Laboratories website. Accessed November 22, 2021. https://ltd.aruplab.com/Tests/Pub/3001409
- Monshi B, Gulz L, Piringer B, et al. Anti-BP180 autoantibody levels at diagnosis correlate with 1-year mortality rates in patients with bullous pemphigoid. J Eur Acad Dermatol Venereol. 2020;34:1583-1589.
- Koga H, Teye K, Ishii N, et al. High index values of enzyme-linked immunosorbent assay for BP180 at baseline predict relapse in patients with bullous pemphigoid. Front Med (Lausanne). 2018;5:139.
- Fichel F, Barbe C, Joly P, et al. Clinical and immunologic factors associated with bullous pemphigoid relapse during the first year of treatment: a multicenter, prospective study. JAMA Dermatol. 2014;150:25-33.
- Cai SC, Lim YL, Li W, et al. Anti-BP180 NC16A IgG titres as an indicator of disease activity and outcome in Asian patients with bullous pemphigoid. Ann Acad Med Singap. 2015;44:119-126.
- Genovese G, Maronese CA, Casazza G, et al. Clinical and serological predictors of relapse in pemphigus: a study of 143 patients [published online July 20, 2021]. Clin Exp Dermatol. doi:10.1111/ced.14854
- Weigand DA. Effect of anatomic region on immunofluorescence diagnosis of bullous pemphigoid. J Am Acad Dermatol. 1985;12(2, pt 1):274-278.
- Weigand DA, Clements MK. Direct immunofluorescence in bullous pemphigoid: effects of extent and location of lesions. J Am Acad Dermatol. 1989;20:437-440.
- Mutasim DF, Adams BB. Immunofluorescence in dermatology. J Am Acad Dermatol. 2001;45:803-822; quiz 822-824.
- Sladden C, Kirchhof MG, Crawford RI. Biopsy location for direct immunofluorescence in patients with suspected bullous pemphigoid impacts probability of a positive test result. J Cutan Med Surg. 2014;18:392-396.
- Elston DM, Stratman EJ, Miller SJ. Skin biopsy: biopsy issues in specific diseases. J Am Acad Dermatol. 2016;74:1-16; quiz 17-18.
- Seishima M, Izumi T, Kitajima Y. Antibody to bullous pemphigoid antigen 1 binds to the antigen at perilesional but not uninvolved skin, in localized bullous pemphigoid. Eur J Dermatol. 1999;9:39-42.
- Zone JJ, Meyer LJ, Petersen MJ. Deposition of granular IgA relative to clinical lesions in dermatitis herpetiformis. Arch Dermatol. 1996;132:912-918.
- Kamaguchi M, Iwata H, Ujiie I, et al. Direct immunofluorescence using non-lesional buccal mucosa in mucous membrane pemphigoid. Front Med (Lausanne). 2018;5:20.
- Carey B, Joshi S, Abdelghani A, et al. The optimal oral biopsy site for diagnosis of mucous membrane pemphigoid and pemphigus vulgaris. Br J Dermatol. 2020;182:747-753.
- Kulthanan K, Pinkaew S, Jiamton S, et al. Cutaneous leukocytoclastic vasculitis: the yield of direct immunofluorescence study. J Med Assoc Thai. 2004;87:531-535.
- Chaidemenos GC, Maltezos E, Chrysomallis F, et al. Value of routine diagnostic criteria of bullous pemphigoid. Int J Dermatol. 1998;37:206-210.
- Mysorekar VV, Sumathy TK, Shyam Prasad AL. Role of direct immunofluorescence in dermatological disorders. Indian Dermatol Online J. 2015;6:172-180.
- Fudge JG, Crawford RI. Bullous pemphigoid: a 10-year study of discordant results on direct immunofluorescence. J Cutan Med Surg. 2018;22:472-475.
- Sárdy M, Kostaki D, Varga R, et al. Comparative study of direct and indirect immunofluorescence and of bullous pemphigoid 180 and 230 enzyme-linked immunosorbent assays for diagnosis of bullous pemphigoid. J Am Acad Dermatol. 2013;69:748-753.
- Buch AC, Kumar H, Panicker N, et al. A cross-sectional study of direct immunofluorescence in the diagnosis of immunobullous dermatoses. Indian J Dermatol. 2014;59:364-368.
- Miller DD, Bhawan J. Bullous tinea pedis with direct immunofluorescence positivity: when is a positive result not autoimmune bullous disease? Am J Dermatopathol. 2013;35:587-594.
- Cao R, Lau S, Tan V, et al. Adult Henoch-Schönlein purpura: clinical and histopathological predictors of systemic disease and profound renal disease. Indian J Dermatol Venereol Leprol. 2017;83:577-582.
- Arthur G. Albert Coons: harnessing the power of the antibody. Lancet Respir Med. 2016;4:181-182.
- Coons AH, Creech HJ, Jones RN. Immunological properties of an antibody containing a fluorescent group. Proc Soc Exp Biol Med. 1941;47:200-202.
- Coons AH, Creech HJ, Jones RN, et al. The demonstration of pneumococcal antigen in tissues by the use of fluorescent antibody. J Immunol. 1942;45:159-170.
- Burnham TK, Neblett TR, Fine G. The application of the fluorescent antibody technic to the investigation of lupus erythematosus and various dermatoses. J Invest Dermatol. 1963;41:451-456.
- Jordon RE, Beutner EH, Witebsky E, et al. Basement zone antibodies in bullous pemphigoid. JAMA. 1967;200:751-756.
- Vaughan Jones SA, Salas J, McGrath JA, et al. A retrospective analysis of tissue-fixed immunoreactants from skin biopsies maintained in Michel’s medium. Dermatology. 1994;189(suppl 1):131-132.
- Kim RH, Brinster NK. Practical direct immunofluorescence. Am J Dermatopathol. 2020;42:75-85.
- Vodegel RM, de Jong MC, Meijer HJ, et al. Enhanced diagnostic immunofluorescence using biopsies transported in saline. BMC Dermatol. 2004;4:10.
- Arbesman J, Grover R, Helm TN, et al. Can direct immunofluorescence testing still be accurate if performed on biopsy specimens after brief inadvertent immersion in formalin? J Am Acad Dermatol. 2011;65:106-111.
- Im K, Mareninov S, Diaz MFP, et al. An introduction to performing immunofluorescence staining. Methods Mol Biol. 2019;1897:299-311.
- Saschenbrecker S, Karl I, Komorowski L, et al. Serological diagnosis of autoimmune bullous skin diseases. Front Immunol. 2019;10:1974.
- Baum S, Sakka N, Artsi O, et al. Diagnosis and classification of autoimmune blistering diseases. Autoimmun Rev. 2014;13:482-489.
- Immunobullous disease panel, epithelial. ARUP Laboratories website. Accessed November 22, 2021. https://ltd.aruplab.com/Tests/Pub/3001409
- Monshi B, Gulz L, Piringer B, et al. Anti-BP180 autoantibody levels at diagnosis correlate with 1-year mortality rates in patients with bullous pemphigoid. J Eur Acad Dermatol Venereol. 2020;34:1583-1589.
- Koga H, Teye K, Ishii N, et al. High index values of enzyme-linked immunosorbent assay for BP180 at baseline predict relapse in patients with bullous pemphigoid. Front Med (Lausanne). 2018;5:139.
- Fichel F, Barbe C, Joly P, et al. Clinical and immunologic factors associated with bullous pemphigoid relapse during the first year of treatment: a multicenter, prospective study. JAMA Dermatol. 2014;150:25-33.
- Cai SC, Lim YL, Li W, et al. Anti-BP180 NC16A IgG titres as an indicator of disease activity and outcome in Asian patients with bullous pemphigoid. Ann Acad Med Singap. 2015;44:119-126.
- Genovese G, Maronese CA, Casazza G, et al. Clinical and serological predictors of relapse in pemphigus: a study of 143 patients [published online July 20, 2021]. Clin Exp Dermatol. doi:10.1111/ced.14854
- Weigand DA. Effect of anatomic region on immunofluorescence diagnosis of bullous pemphigoid. J Am Acad Dermatol. 1985;12(2, pt 1):274-278.
- Weigand DA, Clements MK. Direct immunofluorescence in bullous pemphigoid: effects of extent and location of lesions. J Am Acad Dermatol. 1989;20:437-440.
- Mutasim DF, Adams BB. Immunofluorescence in dermatology. J Am Acad Dermatol. 2001;45:803-822; quiz 822-824.
- Sladden C, Kirchhof MG, Crawford RI. Biopsy location for direct immunofluorescence in patients with suspected bullous pemphigoid impacts probability of a positive test result. J Cutan Med Surg. 2014;18:392-396.
- Elston DM, Stratman EJ, Miller SJ. Skin biopsy: biopsy issues in specific diseases. J Am Acad Dermatol. 2016;74:1-16; quiz 17-18.
- Seishima M, Izumi T, Kitajima Y. Antibody to bullous pemphigoid antigen 1 binds to the antigen at perilesional but not uninvolved skin, in localized bullous pemphigoid. Eur J Dermatol. 1999;9:39-42.
- Zone JJ, Meyer LJ, Petersen MJ. Deposition of granular IgA relative to clinical lesions in dermatitis herpetiformis. Arch Dermatol. 1996;132:912-918.
- Kamaguchi M, Iwata H, Ujiie I, et al. Direct immunofluorescence using non-lesional buccal mucosa in mucous membrane pemphigoid. Front Med (Lausanne). 2018;5:20.
- Carey B, Joshi S, Abdelghani A, et al. The optimal oral biopsy site for diagnosis of mucous membrane pemphigoid and pemphigus vulgaris. Br J Dermatol. 2020;182:747-753.
- Kulthanan K, Pinkaew S, Jiamton S, et al. Cutaneous leukocytoclastic vasculitis: the yield of direct immunofluorescence study. J Med Assoc Thai. 2004;87:531-535.
- Chaidemenos GC, Maltezos E, Chrysomallis F, et al. Value of routine diagnostic criteria of bullous pemphigoid. Int J Dermatol. 1998;37:206-210.
- Mysorekar VV, Sumathy TK, Shyam Prasad AL. Role of direct immunofluorescence in dermatological disorders. Indian Dermatol Online J. 2015;6:172-180.
- Fudge JG, Crawford RI. Bullous pemphigoid: a 10-year study of discordant results on direct immunofluorescence. J Cutan Med Surg. 2018;22:472-475.
- Sárdy M, Kostaki D, Varga R, et al. Comparative study of direct and indirect immunofluorescence and of bullous pemphigoid 180 and 230 enzyme-linked immunosorbent assays for diagnosis of bullous pemphigoid. J Am Acad Dermatol. 2013;69:748-753.
- Buch AC, Kumar H, Panicker N, et al. A cross-sectional study of direct immunofluorescence in the diagnosis of immunobullous dermatoses. Indian J Dermatol. 2014;59:364-368.
- Miller DD, Bhawan J. Bullous tinea pedis with direct immunofluorescence positivity: when is a positive result not autoimmune bullous disease? Am J Dermatopathol. 2013;35:587-594.
- Cao R, Lau S, Tan V, et al. Adult Henoch-Schönlein purpura: clinical and histopathological predictors of systemic disease and profound renal disease. Indian J Dermatol Venereol Leprol. 2017;83:577-582.
Resident Pearl
- Direct immunofluorescence, indirect immunofluorescence, and enzyme-linked immunosorbent assay are important tests for residents to have in their diagnostic tool box, especially when evaluating patients with blistering diseases.
Test Your Knowledge: Generalized Pustular Psoriasis Quiz
In this quiz to Dermatology News, test your current knowledge of Generalized Pustular Psoriasis. Join Abby S. Van Voorhees, MD, as she shares insights on GPP and understanding the disorder.
PC-US-123740 December 2021
In this quiz to Dermatology News, test your current knowledge of Generalized Pustular Psoriasis. Join Abby S. Van Voorhees, MD, as she shares insights on GPP and understanding the disorder.
PC-US-123740 December 2021
In this quiz to Dermatology News, test your current knowledge of Generalized Pustular Psoriasis. Join Abby S. Van Voorhees, MD, as she shares insights on GPP and understanding the disorder.
PC-US-123740 December 2021
TNF-α inhibitor remains a promising therapy for nail psoriasis and concomitant PsA
Key clinical point: Tumor necrosis factor (TNF)-α inhibitors effectively improved skin, nail, and joint symptoms and quality of life (QoL) in patients with nail psoriasis and concomitant psoriatic arthritis (PsA).
Major finding: During the first 3 months, nail psoriasis improved significantly along with improvements in the mean number of swollen joints (SJ), which reduced from 6.4 to 3.1, and the mean number of tender joints, which reduced from 10.8 to 6.4 (both P < .001). Low disease activity according to SJ status was achieved at month 24. QoL improved by 50% (P < .001) after 3 months with further improvements up to month 24.
Study details: Findings are from a noninterventional, prospective cohort study that evaluated effectiveness of 24 months of continuous therapy with adalimumab, etanercept, or infliximab in 100 adult patients with concomitant psoriasis, nail psoriasis, and PsA.
Disclosures: This study was sponsored by AbbVie Deutschland GmbH & Co. Two authors declared being employees and shareholders of AbbVie, and the other authors reported ties with several sources, including AbbVie Deutschland GmbH & Co.
Source: Kokolakis G et al. J Pers Med. 2021;11(11):1083 (Oct 25). Doi: 10.3390/jpm11111083.
Key clinical point: Tumor necrosis factor (TNF)-α inhibitors effectively improved skin, nail, and joint symptoms and quality of life (QoL) in patients with nail psoriasis and concomitant psoriatic arthritis (PsA).
Major finding: During the first 3 months, nail psoriasis improved significantly along with improvements in the mean number of swollen joints (SJ), which reduced from 6.4 to 3.1, and the mean number of tender joints, which reduced from 10.8 to 6.4 (both P < .001). Low disease activity according to SJ status was achieved at month 24. QoL improved by 50% (P < .001) after 3 months with further improvements up to month 24.
Study details: Findings are from a noninterventional, prospective cohort study that evaluated effectiveness of 24 months of continuous therapy with adalimumab, etanercept, or infliximab in 100 adult patients with concomitant psoriasis, nail psoriasis, and PsA.
Disclosures: This study was sponsored by AbbVie Deutschland GmbH & Co. Two authors declared being employees and shareholders of AbbVie, and the other authors reported ties with several sources, including AbbVie Deutschland GmbH & Co.
Source: Kokolakis G et al. J Pers Med. 2021;11(11):1083 (Oct 25). Doi: 10.3390/jpm11111083.
Key clinical point: Tumor necrosis factor (TNF)-α inhibitors effectively improved skin, nail, and joint symptoms and quality of life (QoL) in patients with nail psoriasis and concomitant psoriatic arthritis (PsA).
Major finding: During the first 3 months, nail psoriasis improved significantly along with improvements in the mean number of swollen joints (SJ), which reduced from 6.4 to 3.1, and the mean number of tender joints, which reduced from 10.8 to 6.4 (both P < .001). Low disease activity according to SJ status was achieved at month 24. QoL improved by 50% (P < .001) after 3 months with further improvements up to month 24.
Study details: Findings are from a noninterventional, prospective cohort study that evaluated effectiveness of 24 months of continuous therapy with adalimumab, etanercept, or infliximab in 100 adult patients with concomitant psoriasis, nail psoriasis, and PsA.
Disclosures: This study was sponsored by AbbVie Deutschland GmbH & Co. Two authors declared being employees and shareholders of AbbVie, and the other authors reported ties with several sources, including AbbVie Deutschland GmbH & Co.
Source: Kokolakis G et al. J Pers Med. 2021;11(11):1083 (Oct 25). Doi: 10.3390/jpm11111083.
Upadacitinib improves PRO in PsA patients with inadequate response to non-bDMARD
Key clinical point: Upadacitinib led to early and sustained clinically meaningful improvements in patient-reported outcomes (PRO) in patients with psoriatic arthritis (PsA) having an inadequate response to nonbiologic disease-modifying antirheumatic drugs (non-bDMARD).
Major finding: A greater proportion of patients treated with 15 and 30 mg upadacitinib vs. placebo showed clinically meaningful improvement in the patient global assessment of disease activity, pain, and health assessment questionnaire disability index as early as week 2 (P < .01) which was maintained or further improved at weeks 12 and 24 (P < .01).
Study details: This was a post hoc analysis of the phase 3 SELECT-PsA 1 trial, including 1,704 patients with PsA who had an inadequate response to at least 1 non-bDMARD and who were randomly assigned to upadacitinib, adalimumab, or placebo.
Disclosures: This work was funded by AbbVie. K Kato, E McDearmon-Blondell, P Zueger, and CD Saffore reported being employees and stockholders of AbbVie. The other authors reported ties with several sources including AbbVie.
Source: Strand V et al. Rheumatol Ther. 2021;8:1789-1808 (Oct 12). Doi: 10.1007/s40744-021-00379-9.
Key clinical point: Upadacitinib led to early and sustained clinically meaningful improvements in patient-reported outcomes (PRO) in patients with psoriatic arthritis (PsA) having an inadequate response to nonbiologic disease-modifying antirheumatic drugs (non-bDMARD).
Major finding: A greater proportion of patients treated with 15 and 30 mg upadacitinib vs. placebo showed clinically meaningful improvement in the patient global assessment of disease activity, pain, and health assessment questionnaire disability index as early as week 2 (P < .01) which was maintained or further improved at weeks 12 and 24 (P < .01).
Study details: This was a post hoc analysis of the phase 3 SELECT-PsA 1 trial, including 1,704 patients with PsA who had an inadequate response to at least 1 non-bDMARD and who were randomly assigned to upadacitinib, adalimumab, or placebo.
Disclosures: This work was funded by AbbVie. K Kato, E McDearmon-Blondell, P Zueger, and CD Saffore reported being employees and stockholders of AbbVie. The other authors reported ties with several sources including AbbVie.
Source: Strand V et al. Rheumatol Ther. 2021;8:1789-1808 (Oct 12). Doi: 10.1007/s40744-021-00379-9.
Key clinical point: Upadacitinib led to early and sustained clinically meaningful improvements in patient-reported outcomes (PRO) in patients with psoriatic arthritis (PsA) having an inadequate response to nonbiologic disease-modifying antirheumatic drugs (non-bDMARD).
Major finding: A greater proportion of patients treated with 15 and 30 mg upadacitinib vs. placebo showed clinically meaningful improvement in the patient global assessment of disease activity, pain, and health assessment questionnaire disability index as early as week 2 (P < .01) which was maintained or further improved at weeks 12 and 24 (P < .01).
Study details: This was a post hoc analysis of the phase 3 SELECT-PsA 1 trial, including 1,704 patients with PsA who had an inadequate response to at least 1 non-bDMARD and who were randomly assigned to upadacitinib, adalimumab, or placebo.
Disclosures: This work was funded by AbbVie. K Kato, E McDearmon-Blondell, P Zueger, and CD Saffore reported being employees and stockholders of AbbVie. The other authors reported ties with several sources including AbbVie.
Source: Strand V et al. Rheumatol Ther. 2021;8:1789-1808 (Oct 12). Doi: 10.1007/s40744-021-00379-9.