User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Delineating factors behind frequent biologic switching in psoriasis
Key clinical point: Switching biologics in patients with psoriasis was mostly impelled by secondary lack of efficacy for skin symptoms with young age and the presence of psoriatic arthritis (PsA) linked to a higher frequency of switching in the long-term.
Major finding: Switching of first- and second-line biologics was likely attributed to a secondary lack of efficacy for skin disease. Each unit increase in age decreased the likelihood of switching twice or more by 4% (odds ratio [OR], 0.964; P = .038), whereas the existence of PsA increased the likelihood by 2.69-fold (OR, 2.69; P = .026).
Study details: This was a retrospective study including 115 adult patients with psoriasis who had been receiving biologics for 12 consecutive months or more and underwent at least a single biologic switch.
Disclosures: No specific funding for the study was disclosed. The authors declared no potential conflict of interests.
Source: Akdogan N et al. Expert Rev Clin Pharmacol. 2021 Sep 23. doi: 10.1080/17512433.2021.1979394.
Key clinical point: Switching biologics in patients with psoriasis was mostly impelled by secondary lack of efficacy for skin symptoms with young age and the presence of psoriatic arthritis (PsA) linked to a higher frequency of switching in the long-term.
Major finding: Switching of first- and second-line biologics was likely attributed to a secondary lack of efficacy for skin disease. Each unit increase in age decreased the likelihood of switching twice or more by 4% (odds ratio [OR], 0.964; P = .038), whereas the existence of PsA increased the likelihood by 2.69-fold (OR, 2.69; P = .026).
Study details: This was a retrospective study including 115 adult patients with psoriasis who had been receiving biologics for 12 consecutive months or more and underwent at least a single biologic switch.
Disclosures: No specific funding for the study was disclosed. The authors declared no potential conflict of interests.
Source: Akdogan N et al. Expert Rev Clin Pharmacol. 2021 Sep 23. doi: 10.1080/17512433.2021.1979394.
Key clinical point: Switching biologics in patients with psoriasis was mostly impelled by secondary lack of efficacy for skin symptoms with young age and the presence of psoriatic arthritis (PsA) linked to a higher frequency of switching in the long-term.
Major finding: Switching of first- and second-line biologics was likely attributed to a secondary lack of efficacy for skin disease. Each unit increase in age decreased the likelihood of switching twice or more by 4% (odds ratio [OR], 0.964; P = .038), whereas the existence of PsA increased the likelihood by 2.69-fold (OR, 2.69; P = .026).
Study details: This was a retrospective study including 115 adult patients with psoriasis who had been receiving biologics for 12 consecutive months or more and underwent at least a single biologic switch.
Disclosures: No specific funding for the study was disclosed. The authors declared no potential conflict of interests.
Source: Akdogan N et al. Expert Rev Clin Pharmacol. 2021 Sep 23. doi: 10.1080/17512433.2021.1979394.
High body weight and previous biologic use counter real-life guselkumab efficacy against plaque psoriasis
Key clinical point: This real-life study showed reduced efficacy of guselkumab for over 36 weeks in patients with moderate-to-severe chronic plaque psoriasis than previously reported, with body weight and exposure to biologics being the major predictors of response.
Major finding: At week 36, 67% of patients achieved Psoriasis Area Severity Index (PASI) 75 with heavier vs. low‐weight patients showing a decreased likelihood of achieving PASI 75 until week 4 (odds ratio [OR], 0.94; 95% confidence interval [CI], 0.88-0.99). Even at week 36, PASI 75 response rates were lower for patients exposed to 1 (OR, 0.07; 95% CI, 0.00-0.68) or more than 1 (OR, 0.00; 95% CI, 0.00-0.044) biologics than for biologic-naïve patients.
Study details: Findings are from a multicenter retrospective cohort study including 135 adult patients with moderate-to-severe chronic plaque psoriasis.
Disclosures: The study was supported by the Chang Gung Memorial Hospital and National Taiwan University Hospital, Hsin-Chu branch. Some of the authors declared serving as clinical trial participant or receiving speaker/consultancy honoraria from various sources.
Source: Hung YT et al. Ther Adv Chronic Dis. 2021 Sep 29. doi: 10.1177/20406223211046685.
Key clinical point: This real-life study showed reduced efficacy of guselkumab for over 36 weeks in patients with moderate-to-severe chronic plaque psoriasis than previously reported, with body weight and exposure to biologics being the major predictors of response.
Major finding: At week 36, 67% of patients achieved Psoriasis Area Severity Index (PASI) 75 with heavier vs. low‐weight patients showing a decreased likelihood of achieving PASI 75 until week 4 (odds ratio [OR], 0.94; 95% confidence interval [CI], 0.88-0.99). Even at week 36, PASI 75 response rates were lower for patients exposed to 1 (OR, 0.07; 95% CI, 0.00-0.68) or more than 1 (OR, 0.00; 95% CI, 0.00-0.044) biologics than for biologic-naïve patients.
Study details: Findings are from a multicenter retrospective cohort study including 135 adult patients with moderate-to-severe chronic plaque psoriasis.
Disclosures: The study was supported by the Chang Gung Memorial Hospital and National Taiwan University Hospital, Hsin-Chu branch. Some of the authors declared serving as clinical trial participant or receiving speaker/consultancy honoraria from various sources.
Source: Hung YT et al. Ther Adv Chronic Dis. 2021 Sep 29. doi: 10.1177/20406223211046685.
Key clinical point: This real-life study showed reduced efficacy of guselkumab for over 36 weeks in patients with moderate-to-severe chronic plaque psoriasis than previously reported, with body weight and exposure to biologics being the major predictors of response.
Major finding: At week 36, 67% of patients achieved Psoriasis Area Severity Index (PASI) 75 with heavier vs. low‐weight patients showing a decreased likelihood of achieving PASI 75 until week 4 (odds ratio [OR], 0.94; 95% confidence interval [CI], 0.88-0.99). Even at week 36, PASI 75 response rates were lower for patients exposed to 1 (OR, 0.07; 95% CI, 0.00-0.68) or more than 1 (OR, 0.00; 95% CI, 0.00-0.044) biologics than for biologic-naïve patients.
Study details: Findings are from a multicenter retrospective cohort study including 135 adult patients with moderate-to-severe chronic plaque psoriasis.
Disclosures: The study was supported by the Chang Gung Memorial Hospital and National Taiwan University Hospital, Hsin-Chu branch. Some of the authors declared serving as clinical trial participant or receiving speaker/consultancy honoraria from various sources.
Source: Hung YT et al. Ther Adv Chronic Dis. 2021 Sep 29. doi: 10.1177/20406223211046685.
Occult blood in feces tied to increased risk for psoriasis
Key clinical point: Patients with positive fecal immunochemistry test (FIT) results showed a significantly higher risk for psoriasis than those who were FIT-negative.
Major finding: During a 6.68-year median follow-up, the incidence rate of psoriasis per 1000 person-years was higher for the FIT-positive vs the FIT-negative group (4.14 vs 3.76). After multivariable adjustment, the adjusted hazard ratios for psoriasis were 1.029 (95% confidence interval [CI], 0.997-1.061), 1.118 (95% CI, 1.04-1.201), and 1.342 (95% CI, 1.157-1.557) for 1, 2, and 3 positive FIT results, respectively, vs negative FIT results.
Study details: Findings are from a retrospective nationwide population-based study of 1,395,147 participants aged 50 years or above who underwent screening for colorectal cancer.
Disclosures: The study was supported by the New Faculty Startup Fund from Seoul National University. The authors declared no potential conflict of interests.
Source: Lee HJ et al. Dermatology. 2021 Sep 16. doi: 10.1159/000518625.
Key clinical point: Patients with positive fecal immunochemistry test (FIT) results showed a significantly higher risk for psoriasis than those who were FIT-negative.
Major finding: During a 6.68-year median follow-up, the incidence rate of psoriasis per 1000 person-years was higher for the FIT-positive vs the FIT-negative group (4.14 vs 3.76). After multivariable adjustment, the adjusted hazard ratios for psoriasis were 1.029 (95% confidence interval [CI], 0.997-1.061), 1.118 (95% CI, 1.04-1.201), and 1.342 (95% CI, 1.157-1.557) for 1, 2, and 3 positive FIT results, respectively, vs negative FIT results.
Study details: Findings are from a retrospective nationwide population-based study of 1,395,147 participants aged 50 years or above who underwent screening for colorectal cancer.
Disclosures: The study was supported by the New Faculty Startup Fund from Seoul National University. The authors declared no potential conflict of interests.
Source: Lee HJ et al. Dermatology. 2021 Sep 16. doi: 10.1159/000518625.
Key clinical point: Patients with positive fecal immunochemistry test (FIT) results showed a significantly higher risk for psoriasis than those who were FIT-negative.
Major finding: During a 6.68-year median follow-up, the incidence rate of psoriasis per 1000 person-years was higher for the FIT-positive vs the FIT-negative group (4.14 vs 3.76). After multivariable adjustment, the adjusted hazard ratios for psoriasis were 1.029 (95% confidence interval [CI], 0.997-1.061), 1.118 (95% CI, 1.04-1.201), and 1.342 (95% CI, 1.157-1.557) for 1, 2, and 3 positive FIT results, respectively, vs negative FIT results.
Study details: Findings are from a retrospective nationwide population-based study of 1,395,147 participants aged 50 years or above who underwent screening for colorectal cancer.
Disclosures: The study was supported by the New Faculty Startup Fund from Seoul National University. The authors declared no potential conflict of interests.
Source: Lee HJ et al. Dermatology. 2021 Sep 16. doi: 10.1159/000518625.
Guselkumab: A promising treatment option for moderate-to-severe plaque psoriasis
Key clinical point: Guselkumab positively affected both clinician- and patient-reported outcomes in patients with moderate-to-severe plaque psoriasis, irrespective of their history of psoriasis therapies.
Major finding: After 28 weeks, 56.8% of patients attained a Dermatology Life Quality Index (DLQI) score of 1 or less, with the mean DLQI score decreasing from 13.7 at baseline to 2.8 (95% confidence interval [CI], 2.3-3.2), and mean Psoriasis Area Severity Index (PASI) decreasing from 16.4 at baseline to 3.0 (95% CI, 2.3-3.6), with a 55.3% PASI 90 response rate. Most adverse events were mild or moderate.
Study details: The data come from PERSIST, an ongoing, prospective, real-life study that enrolled 303 patients aged 18 years or above with moderate-to-severe plaque psoriasis for 2 years or more who were prescribed guselkumab.
Disclosures: This study was supported by Janssen-Cilag GmbH (Germany). S Wegner, Y Personke, and M Gomez reported being employees of Janssen-Cilag, whereas the other authors declared serving as an advisor, speaker, or clinical trial participant or receiving grants from various companies including Janssen-Cilag.
Source: Gerdes S et al. J Dermatol. 2021 Sep 12. doi: 10.1111/1346-8138.16128.
Key clinical point: Guselkumab positively affected both clinician- and patient-reported outcomes in patients with moderate-to-severe plaque psoriasis, irrespective of their history of psoriasis therapies.
Major finding: After 28 weeks, 56.8% of patients attained a Dermatology Life Quality Index (DLQI) score of 1 or less, with the mean DLQI score decreasing from 13.7 at baseline to 2.8 (95% confidence interval [CI], 2.3-3.2), and mean Psoriasis Area Severity Index (PASI) decreasing from 16.4 at baseline to 3.0 (95% CI, 2.3-3.6), with a 55.3% PASI 90 response rate. Most adverse events were mild or moderate.
Study details: The data come from PERSIST, an ongoing, prospective, real-life study that enrolled 303 patients aged 18 years or above with moderate-to-severe plaque psoriasis for 2 years or more who were prescribed guselkumab.
Disclosures: This study was supported by Janssen-Cilag GmbH (Germany). S Wegner, Y Personke, and M Gomez reported being employees of Janssen-Cilag, whereas the other authors declared serving as an advisor, speaker, or clinical trial participant or receiving grants from various companies including Janssen-Cilag.
Source: Gerdes S et al. J Dermatol. 2021 Sep 12. doi: 10.1111/1346-8138.16128.
Key clinical point: Guselkumab positively affected both clinician- and patient-reported outcomes in patients with moderate-to-severe plaque psoriasis, irrespective of their history of psoriasis therapies.
Major finding: After 28 weeks, 56.8% of patients attained a Dermatology Life Quality Index (DLQI) score of 1 or less, with the mean DLQI score decreasing from 13.7 at baseline to 2.8 (95% confidence interval [CI], 2.3-3.2), and mean Psoriasis Area Severity Index (PASI) decreasing from 16.4 at baseline to 3.0 (95% CI, 2.3-3.6), with a 55.3% PASI 90 response rate. Most adverse events were mild or moderate.
Study details: The data come from PERSIST, an ongoing, prospective, real-life study that enrolled 303 patients aged 18 years or above with moderate-to-severe plaque psoriasis for 2 years or more who were prescribed guselkumab.
Disclosures: This study was supported by Janssen-Cilag GmbH (Germany). S Wegner, Y Personke, and M Gomez reported being employees of Janssen-Cilag, whereas the other authors declared serving as an advisor, speaker, or clinical trial participant or receiving grants from various companies including Janssen-Cilag.
Source: Gerdes S et al. J Dermatol. 2021 Sep 12. doi: 10.1111/1346-8138.16128.
Smokers with periodontitis are at soaring risk for psoriasis
Key clinical point: Periodontitis serves as an independent risk factor for psoriasis and in combination with smoking synergistically contributes to psoriasis development.
Major finding: The risk for psoriasis was higher in patients with vs. without periodontitis (adjusted hazard ratio [aHR], 1.116; 95% confidence interval [CI], 1.101-1.13). Compared with nonsmokers without periodontitis, the risk for psoriasis in nonsmokers with periodontitis and smokers with periodontitis increased by 11% (aHR, 1.11; 95% CI, 1.094-1.127) and 26.5% (aHR, 1.265; 95% CI, 1.234-1.296), respectively.
Study details: The data come from a 9-year follow-up, nationwide, population-based cohort study that included 1,063,004 and 8,655,587 patients with and without periodontitis, respectively, and not pre-diagnosed with psoriasis.
Disclosures: The study was supported by a National Research Foundation of Korea grant funded by the Korean government. The authors declared no potential conflict of interests.
Source: Han JH et al. Dermatology. 2021 Sep 15. doi: 10.1159/000518296.
Key clinical point: Periodontitis serves as an independent risk factor for psoriasis and in combination with smoking synergistically contributes to psoriasis development.
Major finding: The risk for psoriasis was higher in patients with vs. without periodontitis (adjusted hazard ratio [aHR], 1.116; 95% confidence interval [CI], 1.101-1.13). Compared with nonsmokers without periodontitis, the risk for psoriasis in nonsmokers with periodontitis and smokers with periodontitis increased by 11% (aHR, 1.11; 95% CI, 1.094-1.127) and 26.5% (aHR, 1.265; 95% CI, 1.234-1.296), respectively.
Study details: The data come from a 9-year follow-up, nationwide, population-based cohort study that included 1,063,004 and 8,655,587 patients with and without periodontitis, respectively, and not pre-diagnosed with psoriasis.
Disclosures: The study was supported by a National Research Foundation of Korea grant funded by the Korean government. The authors declared no potential conflict of interests.
Source: Han JH et al. Dermatology. 2021 Sep 15. doi: 10.1159/000518296.
Key clinical point: Periodontitis serves as an independent risk factor for psoriasis and in combination with smoking synergistically contributes to psoriasis development.
Major finding: The risk for psoriasis was higher in patients with vs. without periodontitis (adjusted hazard ratio [aHR], 1.116; 95% confidence interval [CI], 1.101-1.13). Compared with nonsmokers without periodontitis, the risk for psoriasis in nonsmokers with periodontitis and smokers with periodontitis increased by 11% (aHR, 1.11; 95% CI, 1.094-1.127) and 26.5% (aHR, 1.265; 95% CI, 1.234-1.296), respectively.
Study details: The data come from a 9-year follow-up, nationwide, population-based cohort study that included 1,063,004 and 8,655,587 patients with and without periodontitis, respectively, and not pre-diagnosed with psoriasis.
Disclosures: The study was supported by a National Research Foundation of Korea grant funded by the Korean government. The authors declared no potential conflict of interests.
Source: Han JH et al. Dermatology. 2021 Sep 15. doi: 10.1159/000518296.
Cal/BD foam gains ground among patients with plaque psoriasis
Key clinical point: Proactive management with calcipotriene 50 μg/g and betamethasone dipropionate 0.5 mg/g (Cal/BD) foam vs. reactive management with vehicle foam decreased the severity of patient-reported symptoms in patients with plaque psoriasis.
Major finding: Proactive vs. reactive management during 52-week maintenance showed greater improvement in Psoriasis Symptom Inventory (difference, −0.75; P = .0128) and Dermatology Life Quality Index (difference −0.45; P = .007) and nonsignificantly higher EuroQol-5D for psoriasis (0.89 vs 0.88; P = .0842) scores.
Study details: Findings are from a post hoc analysis of phase 3 PSO-LONG trial including 521 patients with plaque psoriasis randomly assigned to proactive management (Cal/BD foam twice weekly) or reactive management (vehicle foam twice weekly) arms.
Disclosures: The study was sponsored by Leo Pharma. The authors declared serving as consultants, advisory board members, and clinical trial investigators or receiving grants/speaker honoraria from various sources, including LEO Pharma.
Source: Jalili A et al. J Eur Acad Dermatol Venereol. 2021 Sep 20. doi: 10.1111/jdv.17673.
Key clinical point: Proactive management with calcipotriene 50 μg/g and betamethasone dipropionate 0.5 mg/g (Cal/BD) foam vs. reactive management with vehicle foam decreased the severity of patient-reported symptoms in patients with plaque psoriasis.
Major finding: Proactive vs. reactive management during 52-week maintenance showed greater improvement in Psoriasis Symptom Inventory (difference, −0.75; P = .0128) and Dermatology Life Quality Index (difference −0.45; P = .007) and nonsignificantly higher EuroQol-5D for psoriasis (0.89 vs 0.88; P = .0842) scores.
Study details: Findings are from a post hoc analysis of phase 3 PSO-LONG trial including 521 patients with plaque psoriasis randomly assigned to proactive management (Cal/BD foam twice weekly) or reactive management (vehicle foam twice weekly) arms.
Disclosures: The study was sponsored by Leo Pharma. The authors declared serving as consultants, advisory board members, and clinical trial investigators or receiving grants/speaker honoraria from various sources, including LEO Pharma.
Source: Jalili A et al. J Eur Acad Dermatol Venereol. 2021 Sep 20. doi: 10.1111/jdv.17673.
Key clinical point: Proactive management with calcipotriene 50 μg/g and betamethasone dipropionate 0.5 mg/g (Cal/BD) foam vs. reactive management with vehicle foam decreased the severity of patient-reported symptoms in patients with plaque psoriasis.
Major finding: Proactive vs. reactive management during 52-week maintenance showed greater improvement in Psoriasis Symptom Inventory (difference, −0.75; P = .0128) and Dermatology Life Quality Index (difference −0.45; P = .007) and nonsignificantly higher EuroQol-5D for psoriasis (0.89 vs 0.88; P = .0842) scores.
Study details: Findings are from a post hoc analysis of phase 3 PSO-LONG trial including 521 patients with plaque psoriasis randomly assigned to proactive management (Cal/BD foam twice weekly) or reactive management (vehicle foam twice weekly) arms.
Disclosures: The study was sponsored by Leo Pharma. The authors declared serving as consultants, advisory board members, and clinical trial investigators or receiving grants/speaker honoraria from various sources, including LEO Pharma.
Source: Jalili A et al. J Eur Acad Dermatol Venereol. 2021 Sep 20. doi: 10.1111/jdv.17673.
Patient-reported outcome measures complement clinician-reported outcomes in psoriasis
Key clinical point: The disease severity assessment, guiding initiation of systemic therapy, differed when either patient- or clinician-reported outcomes were considered alone, highlighting the complementary value of both for appropriate treatment.
Major finding: Overall, 72.4% (95% confidence interval [CI], 67.3%-77.0%) of patients who qualified for systemic therapy initiation on account of Psoriasis Area Severity Index (PASI) scores or body surface area (BSA) of 10 or higher had a Dermatology Life Quality Index (DLQI) score of less than 10. Conversely, 10.4% (95% CI, 8.8%-12.1%) of patients with a DLQI score higher than 10 did not qualify for systemic therapy based on PASI scores or BSA.
Study details: Findings are from a cross-sectional, observational study including 1,733 patients with mild, moderate, or severe psoriasis who met the criteria for initiation of systemic therapy based on DLQI score, PASI score, or BSA.
Disclosures: No specific funding for the study was disclosed. Dr. Gelfand declared receiving consultation fees and research grants from various sources.
Source: Barbieri JS et al. JAMA Dermatol. 2021 Sep 8. doi: 10.1001/jamadermatol.2021.3341.
Key clinical point: The disease severity assessment, guiding initiation of systemic therapy, differed when either patient- or clinician-reported outcomes were considered alone, highlighting the complementary value of both for appropriate treatment.
Major finding: Overall, 72.4% (95% confidence interval [CI], 67.3%-77.0%) of patients who qualified for systemic therapy initiation on account of Psoriasis Area Severity Index (PASI) scores or body surface area (BSA) of 10 or higher had a Dermatology Life Quality Index (DLQI) score of less than 10. Conversely, 10.4% (95% CI, 8.8%-12.1%) of patients with a DLQI score higher than 10 did not qualify for systemic therapy based on PASI scores or BSA.
Study details: Findings are from a cross-sectional, observational study including 1,733 patients with mild, moderate, or severe psoriasis who met the criteria for initiation of systemic therapy based on DLQI score, PASI score, or BSA.
Disclosures: No specific funding for the study was disclosed. Dr. Gelfand declared receiving consultation fees and research grants from various sources.
Source: Barbieri JS et al. JAMA Dermatol. 2021 Sep 8. doi: 10.1001/jamadermatol.2021.3341.
Key clinical point: The disease severity assessment, guiding initiation of systemic therapy, differed when either patient- or clinician-reported outcomes were considered alone, highlighting the complementary value of both for appropriate treatment.
Major finding: Overall, 72.4% (95% confidence interval [CI], 67.3%-77.0%) of patients who qualified for systemic therapy initiation on account of Psoriasis Area Severity Index (PASI) scores or body surface area (BSA) of 10 or higher had a Dermatology Life Quality Index (DLQI) score of less than 10. Conversely, 10.4% (95% CI, 8.8%-12.1%) of patients with a DLQI score higher than 10 did not qualify for systemic therapy based on PASI scores or BSA.
Study details: Findings are from a cross-sectional, observational study including 1,733 patients with mild, moderate, or severe psoriasis who met the criteria for initiation of systemic therapy based on DLQI score, PASI score, or BSA.
Disclosures: No specific funding for the study was disclosed. Dr. Gelfand declared receiving consultation fees and research grants from various sources.
Source: Barbieri JS et al. JAMA Dermatol. 2021 Sep 8. doi: 10.1001/jamadermatol.2021.3341.
Risankizumab shows excellent long-term PASI response in psoriasis under real-world conditions
Key clinical point: Risankizumab displayed superior long-term clinical efficacy against moderate-to-severe psoriasis, with no new safety signals in the 52-week update of a multicenter real-life study.
Major finding: Psoriasis Area and Severity Index (PASI) 75, PASI 90, and PASI 100 response rates at week 36 were 92.7%, 83.6%, and 65.5%, which were maintained through week 52 at 96.4%, 85.5%, and 60.0%, respectively. Unlike in phase 3 trials, more biologic-experienced patients reached PASI 100 at weeks 36 and 52 (71.8% and 69.2%, respectively) compared with biologic-naïve patients (50.0% and 37.5%, respectively).
Study details: Findings are from a real-life multicenter study including 57 adult patients with moderate-to-severe psoriasis who were receiving risankizumab therapy.
Disclosures: The study did not receive any funding. Some of the authors including the lead author declared receiving consultation/personal fees or research grants from various sources.
Source: Hansel K et al. J Eur Acad Dermatol Venereol. 2021 Sep 12. doi: 10.1111/jdv.17656.
Key clinical point: Risankizumab displayed superior long-term clinical efficacy against moderate-to-severe psoriasis, with no new safety signals in the 52-week update of a multicenter real-life study.
Major finding: Psoriasis Area and Severity Index (PASI) 75, PASI 90, and PASI 100 response rates at week 36 were 92.7%, 83.6%, and 65.5%, which were maintained through week 52 at 96.4%, 85.5%, and 60.0%, respectively. Unlike in phase 3 trials, more biologic-experienced patients reached PASI 100 at weeks 36 and 52 (71.8% and 69.2%, respectively) compared with biologic-naïve patients (50.0% and 37.5%, respectively).
Study details: Findings are from a real-life multicenter study including 57 adult patients with moderate-to-severe psoriasis who were receiving risankizumab therapy.
Disclosures: The study did not receive any funding. Some of the authors including the lead author declared receiving consultation/personal fees or research grants from various sources.
Source: Hansel K et al. J Eur Acad Dermatol Venereol. 2021 Sep 12. doi: 10.1111/jdv.17656.
Key clinical point: Risankizumab displayed superior long-term clinical efficacy against moderate-to-severe psoriasis, with no new safety signals in the 52-week update of a multicenter real-life study.
Major finding: Psoriasis Area and Severity Index (PASI) 75, PASI 90, and PASI 100 response rates at week 36 were 92.7%, 83.6%, and 65.5%, which were maintained through week 52 at 96.4%, 85.5%, and 60.0%, respectively. Unlike in phase 3 trials, more biologic-experienced patients reached PASI 100 at weeks 36 and 52 (71.8% and 69.2%, respectively) compared with biologic-naïve patients (50.0% and 37.5%, respectively).
Study details: Findings are from a real-life multicenter study including 57 adult patients with moderate-to-severe psoriasis who were receiving risankizumab therapy.
Disclosures: The study did not receive any funding. Some of the authors including the lead author declared receiving consultation/personal fees or research grants from various sources.
Source: Hansel K et al. J Eur Acad Dermatol Venereol. 2021 Sep 12. doi: 10.1111/jdv.17656.
Secukinumab hits mark against pediatric plaque psoriasis
Key clinical point: Secukinumab demonstrated a high skin-clearing effect and favorable safety in pediatric patients with moderate-to-severe plaque psoriasis through week 24.
Major finding: At week 12, low dose (LD) and high dose (HD) groups showed a Psoriasis Area Severity Index (PASI) 75 response rate (RR) of 92.9%, whereas their Investigator's Global Assessment (IGA) 0/1 RRs were 78.5% and 83.4%, respectively. At week 24, the PASI 75 RR for LD and HD groups increased to 95.2% and IGA 0/1 RRs increased to 88.1% and 92.9%, respectively. Both groups displayed similar treatment-emergent adverse events.
Study details: These are 24-week findings of a 224-week, open-label, phase 3 study including 84 pediatric patients with moderate-to-severe plaque psoriasis randomly assigned to LD or HD secukinumab (75/75/150 or 75/150/300 mg for body weight (kg) less than 25/25 to less than 50/50 or more, respectively).
Disclosures: The study was sponsored by Novartis Pharma AG, Switzerland. R Mazur, P Forrer, and M Patekar declared being employees of Novartis Pharma AG. Some of the authors declared serving on advisory board, speaker, consultant, or principal/subinvestigator for various organizations including Novartis.
Source: Magnolo N et al. J Am Acad Dermatol. 2021 Sep 20. doi: 10.1016/j.jaad.2021.08.066.
Key clinical point: Secukinumab demonstrated a high skin-clearing effect and favorable safety in pediatric patients with moderate-to-severe plaque psoriasis through week 24.
Major finding: At week 12, low dose (LD) and high dose (HD) groups showed a Psoriasis Area Severity Index (PASI) 75 response rate (RR) of 92.9%, whereas their Investigator's Global Assessment (IGA) 0/1 RRs were 78.5% and 83.4%, respectively. At week 24, the PASI 75 RR for LD and HD groups increased to 95.2% and IGA 0/1 RRs increased to 88.1% and 92.9%, respectively. Both groups displayed similar treatment-emergent adverse events.
Study details: These are 24-week findings of a 224-week, open-label, phase 3 study including 84 pediatric patients with moderate-to-severe plaque psoriasis randomly assigned to LD or HD secukinumab (75/75/150 or 75/150/300 mg for body weight (kg) less than 25/25 to less than 50/50 or more, respectively).
Disclosures: The study was sponsored by Novartis Pharma AG, Switzerland. R Mazur, P Forrer, and M Patekar declared being employees of Novartis Pharma AG. Some of the authors declared serving on advisory board, speaker, consultant, or principal/subinvestigator for various organizations including Novartis.
Source: Magnolo N et al. J Am Acad Dermatol. 2021 Sep 20. doi: 10.1016/j.jaad.2021.08.066.
Key clinical point: Secukinumab demonstrated a high skin-clearing effect and favorable safety in pediatric patients with moderate-to-severe plaque psoriasis through week 24.
Major finding: At week 12, low dose (LD) and high dose (HD) groups showed a Psoriasis Area Severity Index (PASI) 75 response rate (RR) of 92.9%, whereas their Investigator's Global Assessment (IGA) 0/1 RRs were 78.5% and 83.4%, respectively. At week 24, the PASI 75 RR for LD and HD groups increased to 95.2% and IGA 0/1 RRs increased to 88.1% and 92.9%, respectively. Both groups displayed similar treatment-emergent adverse events.
Study details: These are 24-week findings of a 224-week, open-label, phase 3 study including 84 pediatric patients with moderate-to-severe plaque psoriasis randomly assigned to LD or HD secukinumab (75/75/150 or 75/150/300 mg for body weight (kg) less than 25/25 to less than 50/50 or more, respectively).
Disclosures: The study was sponsored by Novartis Pharma AG, Switzerland. R Mazur, P Forrer, and M Patekar declared being employees of Novartis Pharma AG. Some of the authors declared serving on advisory board, speaker, consultant, or principal/subinvestigator for various organizations including Novartis.
Source: Magnolo N et al. J Am Acad Dermatol. 2021 Sep 20. doi: 10.1016/j.jaad.2021.08.066.
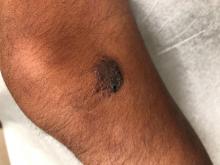
Teen boy’s knee lesion has changed
A biopsy of the lesion was performed which showed an increased number of eccrine glands and blood vessels within the dermis. Some areas showed an increase in adipocytes and smooth muscle bundles. The changes were consistent with eccrine angiomatous hamartoma (EAH).
The boy was referred to vascular laser therapy for treatment of the lesion.
EAH is a rare benign vascular growth characterized by an increased number of mature eccrine glands and blood vessels in the dermis and subcutis. The lesions are mostly present on the extremities, but cases of diffuse congenital lesions and lesions on the face and trunk have also been described. The lesions can be seen at birth or during the first years of life in about half of the cases, and the others tend to occur later in puberty and rarely in adulthood.1
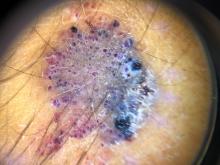
Clinically, EAH lesions present as red, yellow to brown papules and plaques. Different dermoscopic patterns have been described which include the popcorn pattern that presents as yellow, confluent nodules with popcornlike shapes over a background of erythema, and linear arborizing vessels. The spitzoid pattern are brown globules on a background of erythema and pseudoreticular pigmentation around the globules. The verrucous hemangiomalike pattern has a bluish-white hue, reddish-blue or bluish lacunae, as seen in our patient.2-4
Most of the lesions are asymptomatic, but in some patients, they can be associated with pain, hyperhidrosis, and sometimes bleeding. Hyperhidrosis has been reported early in the presentation or during puberty or pregnancy. Our patient had started on amphetamines when hyperhidrosis occurred. Hyperhidrosis is a knowns side effect of this type of medication and may have had a role in the increased sweating noted on the hamartoma.
EAH can clinically look like verrucous hemangiomas, angiokeratomas, and vascular malformations, and histopathology may be needed to differentiate between them. Eccrine nevi and EAH can be similar. Hyperhidrosis is an early and predominant component of eccrine nevi, compared with one-third of EAH.
The exact etiology of this lesion is not known. It is thought to be caused by an abnormal differentiation of the epithelium, adnexal structure, and the mesenchyme during organogenesis.3 No other associated conditions have been described with EAH.
EAH are benign lesions that rarely require treatment. If the lesions are symptomatic or because of cosmetic reasons, they can be removed surgically. There are some reports of successful treatment with pulse dual-wavelength sequential 595- and 1064-nm lasers.5 Botulinum toxin has also been used in cases of symptomatic hyperhidrosis.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She has no conflicts. Email her at [email protected].
References
1. Smith SD et al. Pediatr Dermatol. 2019 Nov;36(6):909-12.
2. Patterson AT et al. Am J Dermatopathol. 2016;38:413-7.
3. Garcıa-Garcıa SC et al. JAAD Case Rep. 2018;4(2):165-7.
4. Awatef Kelati et al. JAAD Case Rep. 2018;4(8)835-6.
5. Felgueiras J et al. Dermatol Surg. 2015 Mar;41(3):428-30.
A biopsy of the lesion was performed which showed an increased number of eccrine glands and blood vessels within the dermis. Some areas showed an increase in adipocytes and smooth muscle bundles. The changes were consistent with eccrine angiomatous hamartoma (EAH).
The boy was referred to vascular laser therapy for treatment of the lesion.
EAH is a rare benign vascular growth characterized by an increased number of mature eccrine glands and blood vessels in the dermis and subcutis. The lesions are mostly present on the extremities, but cases of diffuse congenital lesions and lesions on the face and trunk have also been described. The lesions can be seen at birth or during the first years of life in about half of the cases, and the others tend to occur later in puberty and rarely in adulthood.1
Clinically, EAH lesions present as red, yellow to brown papules and plaques. Different dermoscopic patterns have been described which include the popcorn pattern that presents as yellow, confluent nodules with popcornlike shapes over a background of erythema, and linear arborizing vessels. The spitzoid pattern are brown globules on a background of erythema and pseudoreticular pigmentation around the globules. The verrucous hemangiomalike pattern has a bluish-white hue, reddish-blue or bluish lacunae, as seen in our patient.2-4
Most of the lesions are asymptomatic, but in some patients, they can be associated with pain, hyperhidrosis, and sometimes bleeding. Hyperhidrosis has been reported early in the presentation or during puberty or pregnancy. Our patient had started on amphetamines when hyperhidrosis occurred. Hyperhidrosis is a knowns side effect of this type of medication and may have had a role in the increased sweating noted on the hamartoma.
EAH can clinically look like verrucous hemangiomas, angiokeratomas, and vascular malformations, and histopathology may be needed to differentiate between them. Eccrine nevi and EAH can be similar. Hyperhidrosis is an early and predominant component of eccrine nevi, compared with one-third of EAH.
The exact etiology of this lesion is not known. It is thought to be caused by an abnormal differentiation of the epithelium, adnexal structure, and the mesenchyme during organogenesis.3 No other associated conditions have been described with EAH.
EAH are benign lesions that rarely require treatment. If the lesions are symptomatic or because of cosmetic reasons, they can be removed surgically. There are some reports of successful treatment with pulse dual-wavelength sequential 595- and 1064-nm lasers.5 Botulinum toxin has also been used in cases of symptomatic hyperhidrosis.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She has no conflicts. Email her at [email protected].
References
1. Smith SD et al. Pediatr Dermatol. 2019 Nov;36(6):909-12.
2. Patterson AT et al. Am J Dermatopathol. 2016;38:413-7.
3. Garcıa-Garcıa SC et al. JAAD Case Rep. 2018;4(2):165-7.
4. Awatef Kelati et al. JAAD Case Rep. 2018;4(8)835-6.
5. Felgueiras J et al. Dermatol Surg. 2015 Mar;41(3):428-30.
A biopsy of the lesion was performed which showed an increased number of eccrine glands and blood vessels within the dermis. Some areas showed an increase in adipocytes and smooth muscle bundles. The changes were consistent with eccrine angiomatous hamartoma (EAH).
The boy was referred to vascular laser therapy for treatment of the lesion.
EAH is a rare benign vascular growth characterized by an increased number of mature eccrine glands and blood vessels in the dermis and subcutis. The lesions are mostly present on the extremities, but cases of diffuse congenital lesions and lesions on the face and trunk have also been described. The lesions can be seen at birth or during the first years of life in about half of the cases, and the others tend to occur later in puberty and rarely in adulthood.1
Clinically, EAH lesions present as red, yellow to brown papules and plaques. Different dermoscopic patterns have been described which include the popcorn pattern that presents as yellow, confluent nodules with popcornlike shapes over a background of erythema, and linear arborizing vessels. The spitzoid pattern are brown globules on a background of erythema and pseudoreticular pigmentation around the globules. The verrucous hemangiomalike pattern has a bluish-white hue, reddish-blue or bluish lacunae, as seen in our patient.2-4
Most of the lesions are asymptomatic, but in some patients, they can be associated with pain, hyperhidrosis, and sometimes bleeding. Hyperhidrosis has been reported early in the presentation or during puberty or pregnancy. Our patient had started on amphetamines when hyperhidrosis occurred. Hyperhidrosis is a knowns side effect of this type of medication and may have had a role in the increased sweating noted on the hamartoma.
EAH can clinically look like verrucous hemangiomas, angiokeratomas, and vascular malformations, and histopathology may be needed to differentiate between them. Eccrine nevi and EAH can be similar. Hyperhidrosis is an early and predominant component of eccrine nevi, compared with one-third of EAH.
The exact etiology of this lesion is not known. It is thought to be caused by an abnormal differentiation of the epithelium, adnexal structure, and the mesenchyme during organogenesis.3 No other associated conditions have been described with EAH.
EAH are benign lesions that rarely require treatment. If the lesions are symptomatic or because of cosmetic reasons, they can be removed surgically. There are some reports of successful treatment with pulse dual-wavelength sequential 595- and 1064-nm lasers.5 Botulinum toxin has also been used in cases of symptomatic hyperhidrosis.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She has no conflicts. Email her at [email protected].
References
1. Smith SD et al. Pediatr Dermatol. 2019 Nov;36(6):909-12.
2. Patterson AT et al. Am J Dermatopathol. 2016;38:413-7.
3. Garcıa-Garcıa SC et al. JAAD Case Rep. 2018;4(2):165-7.
4. Awatef Kelati et al. JAAD Case Rep. 2018;4(8)835-6.
5. Felgueiras J et al. Dermatol Surg. 2015 Mar;41(3):428-30.
A 14-year-old male was referred to our pediatric dermatology clinic for evaluation of a lesion on the left knee that appeared at 1 year of age. The lesion has been growing with him and was not symptomatic until 6 months prior to the consultation, when it started bleeding and feeling wet.
He has a history of attention-deficit/hyperactivity disorder managed with dextroamphetamine-amphetamine. The changes noted on the knee lesion seem to occur at the same time that his ADHD medication was started.
On physical exam he had a violaceous circular plaque on the left knee.
On dermoscopy the lesion showed multiple dilated red and violaceous lacunae and whitish blue hue.