User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
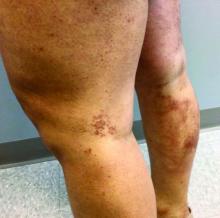
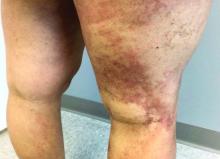
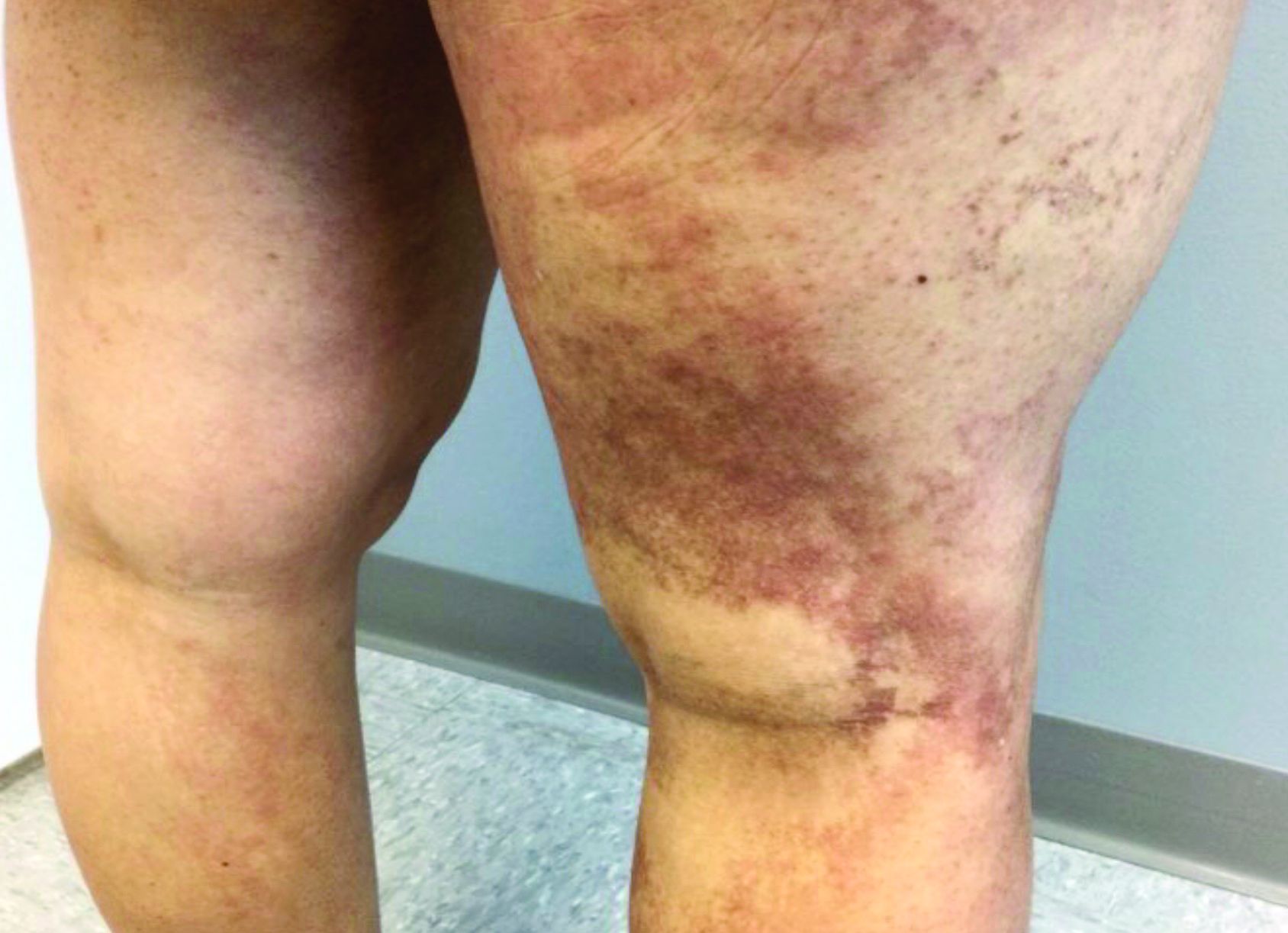
Latest data show increase in breakthrough COVID-19 cases
Breakthrough cases accounted for about one in five newly diagnosed cases in six of the states, according to the New York Times. Hospitalizations and deaths among vaccinated people may be higher than previously thought as well.
“Remember when the early vaccine studies came out, it was like nobody gets hospitalized, nobody dies,” Robert Wachter, MD, chairman of the department of medicine at the University of California, San Francisco, said in an interview. “That clearly is not true.”
The New York Times analyzed data in seven states – California, Colorado, Massachusetts, Oregon, Utah, Vermont, and Virginia – that are tracking the most detailed information. The trends in these states may not reflect the numbers throughout the country, the newspaper reported.
Even still, the numbers back up the idea that vaccinated people may need booster shots this fall to support their earlier vaccine doses. Federal health officials are scheduled to approve the extra shots in coming weeks, potentially in September. The first people to receive booster shots will likely be health care workers and nursing home residents who took the first vaccines in December and January.
“If the chances of a breakthrough infection have gone up considerably, and I think the evidence is clear that they have, and the level of protection against severe illness is no longer as robust as it was, I think the case for boosters goes up pretty quickly,” Dr. Wachter said.
Previous analyses of breakthrough cases included data from June and earlier, the newspaper reported. But since July, COVID-19 cases have soared again because of the Delta variant, and the most recent numbers show an uptick among vaccinated people. In Los Angeles County, for instance, fully vaccinated people account for 20% of new COVID-19 cases, which is up from 11% in May, 5% in April, and 2% in March, according to a late July report from the Los Angeles County Department of Public Health.
What’s more, breakthrough infections in the seven states accounted for 12%-24% of COVID-19 hospitalizations in those states. About 8,000 breakthrough hospitalizations have been reported to the CDC. Still, the overall numbers remain low – in California, for instance, about 1,615 people have been hospitalized with breakthrough infections, which accounts for 0.007% of the state’s 22 million vaccinated people, the Times reported.
The breakthrough infections appear to be more severe among vaccinated people who are older or have weakened immune systems. About 74% of breakthrough cases are among adults 65 or older, the CDC reported.
The increase may shift how vaccinated people see their risks for infection and interact with loved ones. Public health officials have suggested that people follow some COVID-19 safety protocols again, such as wearing masks in public indoor spaces regardless of vaccination status.
As the Delta variant continues to circulate this fall, public health researchers will be researching more about breakthrough cases among vaccinated people, including whether they have prolonged symptoms and how easily they may pass the virus to others.
“I think some of us have been challenged by the numbers of clusters that we’ve seen,” Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, told this news organization.
“I think that really needs to be examined more,” he said.
A version of this article first appeared on WebMD.com.
Breakthrough cases accounted for about one in five newly diagnosed cases in six of the states, according to the New York Times. Hospitalizations and deaths among vaccinated people may be higher than previously thought as well.
“Remember when the early vaccine studies came out, it was like nobody gets hospitalized, nobody dies,” Robert Wachter, MD, chairman of the department of medicine at the University of California, San Francisco, said in an interview. “That clearly is not true.”
The New York Times analyzed data in seven states – California, Colorado, Massachusetts, Oregon, Utah, Vermont, and Virginia – that are tracking the most detailed information. The trends in these states may not reflect the numbers throughout the country, the newspaper reported.
Even still, the numbers back up the idea that vaccinated people may need booster shots this fall to support their earlier vaccine doses. Federal health officials are scheduled to approve the extra shots in coming weeks, potentially in September. The first people to receive booster shots will likely be health care workers and nursing home residents who took the first vaccines in December and January.
“If the chances of a breakthrough infection have gone up considerably, and I think the evidence is clear that they have, and the level of protection against severe illness is no longer as robust as it was, I think the case for boosters goes up pretty quickly,” Dr. Wachter said.
Previous analyses of breakthrough cases included data from June and earlier, the newspaper reported. But since July, COVID-19 cases have soared again because of the Delta variant, and the most recent numbers show an uptick among vaccinated people. In Los Angeles County, for instance, fully vaccinated people account for 20% of new COVID-19 cases, which is up from 11% in May, 5% in April, and 2% in March, according to a late July report from the Los Angeles County Department of Public Health.
What’s more, breakthrough infections in the seven states accounted for 12%-24% of COVID-19 hospitalizations in those states. About 8,000 breakthrough hospitalizations have been reported to the CDC. Still, the overall numbers remain low – in California, for instance, about 1,615 people have been hospitalized with breakthrough infections, which accounts for 0.007% of the state’s 22 million vaccinated people, the Times reported.
The breakthrough infections appear to be more severe among vaccinated people who are older or have weakened immune systems. About 74% of breakthrough cases are among adults 65 or older, the CDC reported.
The increase may shift how vaccinated people see their risks for infection and interact with loved ones. Public health officials have suggested that people follow some COVID-19 safety protocols again, such as wearing masks in public indoor spaces regardless of vaccination status.
As the Delta variant continues to circulate this fall, public health researchers will be researching more about breakthrough cases among vaccinated people, including whether they have prolonged symptoms and how easily they may pass the virus to others.
“I think some of us have been challenged by the numbers of clusters that we’ve seen,” Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, told this news organization.
“I think that really needs to be examined more,” he said.
A version of this article first appeared on WebMD.com.
Breakthrough cases accounted for about one in five newly diagnosed cases in six of the states, according to the New York Times. Hospitalizations and deaths among vaccinated people may be higher than previously thought as well.
“Remember when the early vaccine studies came out, it was like nobody gets hospitalized, nobody dies,” Robert Wachter, MD, chairman of the department of medicine at the University of California, San Francisco, said in an interview. “That clearly is not true.”
The New York Times analyzed data in seven states – California, Colorado, Massachusetts, Oregon, Utah, Vermont, and Virginia – that are tracking the most detailed information. The trends in these states may not reflect the numbers throughout the country, the newspaper reported.
Even still, the numbers back up the idea that vaccinated people may need booster shots this fall to support their earlier vaccine doses. Federal health officials are scheduled to approve the extra shots in coming weeks, potentially in September. The first people to receive booster shots will likely be health care workers and nursing home residents who took the first vaccines in December and January.
“If the chances of a breakthrough infection have gone up considerably, and I think the evidence is clear that they have, and the level of protection against severe illness is no longer as robust as it was, I think the case for boosters goes up pretty quickly,” Dr. Wachter said.
Previous analyses of breakthrough cases included data from June and earlier, the newspaper reported. But since July, COVID-19 cases have soared again because of the Delta variant, and the most recent numbers show an uptick among vaccinated people. In Los Angeles County, for instance, fully vaccinated people account for 20% of new COVID-19 cases, which is up from 11% in May, 5% in April, and 2% in March, according to a late July report from the Los Angeles County Department of Public Health.
What’s more, breakthrough infections in the seven states accounted for 12%-24% of COVID-19 hospitalizations in those states. About 8,000 breakthrough hospitalizations have been reported to the CDC. Still, the overall numbers remain low – in California, for instance, about 1,615 people have been hospitalized with breakthrough infections, which accounts for 0.007% of the state’s 22 million vaccinated people, the Times reported.
The breakthrough infections appear to be more severe among vaccinated people who are older or have weakened immune systems. About 74% of breakthrough cases are among adults 65 or older, the CDC reported.
The increase may shift how vaccinated people see their risks for infection and interact with loved ones. Public health officials have suggested that people follow some COVID-19 safety protocols again, such as wearing masks in public indoor spaces regardless of vaccination status.
As the Delta variant continues to circulate this fall, public health researchers will be researching more about breakthrough cases among vaccinated people, including whether they have prolonged symptoms and how easily they may pass the virus to others.
“I think some of us have been challenged by the numbers of clusters that we’ve seen,” Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, told this news organization.
“I think that really needs to be examined more,” he said.
A version of this article first appeared on WebMD.com.
Exsanguinating the truth about dragon’s blood in cosmeceuticals
The use of dragon’s blood is renowned among various medical traditions around the world.1,2 It is known to confer anti-inflammatory, antioxidant, antitumor, antimicrobial, and wound healing benefits, among others. Dragon’s blood and its characteristic red sap has also been used in folk magic and as a coloring substance and varnish.1 In addition, dragon’s blood resin is one of the many botanical agents with roots in traditional medicine that are among the bioactive ingredients used in the booming contemporary Korean cosmeceutical agent market.3.
Many plants, only some have dermatologic properties
Essentially, the moniker “dragon’s blood” describes the deep red resin or sap that has been derived from multiple plant sources – primarily from the genera Daemonorops, Dracaena, Croton, and Pterocarpus – over multiple centuries.2,4 In traditional Chinese medicine (TCM), various plants have been used as dragon’s blood, including Butea monosperma, Liquidambar formosana, Daemonorops draco, and, more commonly now, Dracaena cochinchinensis.5
Chemical constituents and activity
Dragon’s blood represents the red exudate culled from 27 species of plants from four families. Among the six Dracaena plants (D. cochinchinensis, D. cambodiana, D. cinnabari, D. draco, D. loureiroi, and D. schizantha) from which dragon’s blood is derived, flavonoids and their oligomers are considered the main active constituents. Analgesic, anti-inflammatory, antibacterial, hypolipidemic, hypoglycemic, and cytotoxic activities have been associated with these botanicals.6
D. cochinchinensis is one source of the ethnomedicine “dragon’s blood” that has long been used in TCM. Contemporary studies have shown that the resin of D. cochinchinensis – key constituents of which include loureirin A, loureirin B, loureirin C, cochinchinenin, socotrin-4’-ol, 4’,7-dihydroxyflavan, 4-methylcholest-7-ene-3-ol, ethylparaben, resveratrol, and hydroxyphenol – exhibits antibacterial, anti-inflammatory, analgesic, antidiabetic, and antitumor activities. It has also been shown to support skin repair.4
In 2017, Wang et al. reported that flavonoids from artificially induced dragon’s blood of D. cambodiana showed antibacterial properties.7 The next year, Al Fatimi reported that the dragon’s blood derived from D. cinnabari is a key plant on Yemen’s Socotra Island, where it is used for its antifungal and antioxidant properties to treat various dermal, dental, eye, and gastrointestinal diseases in humans.8Croton lechleri (also one of the plants known as dragon’s blood), a medicinal plant found in the Amazon rainforest and characterized by its red sap, has been shown in preclinical studies to display anti-inflammatory, antioxidant, antimicrobial, antifungal, and antineoplastic activity. Pona et al. note that, while clinical studies of C. lechleri suggest wound healing and antiviral effects, the current use of this plant has limited cutaneous applications.9
Wound healing activity
In 1995, Pieters et al. performed an in vivo study on rats to assess the wound healing activity of dragon’s blood (Croton spp.) from South America. In comparing the effects with those of synthetic proanthocyanidins, the researchers verified the beneficial impact of dragon’s blood in stimulating wound contraction, crust formation, new collagen development, and epithelial layer regeneration. The dragon’s blood component 3’,4-O-dimethylcedrusin was also found to enhance healing by promoting fibroblast and collagen formation, though it was not as effective as crude dragon’s blood. The authors ascribed this effect to the proanthocyanidins in the plant.10
Late in 2003, Jones published a literature review on the evidence related to Croton lechleri (known in South America as “sangre de drago” or dragon’s blood) in support of various biological effects, particularly anti-inflammatory and wound healing capability. The results from multiple in vitro and in vivo investigations buttressed previous ethnomedical justifications for the use of dragon’s blood to treat herpes, insect bites, stomach ulcers, tumors, wounds, and diarrhea, as well as other conditions. Jones added that the sap of the plant has exhibited low toxicity and has been well tolerated in clinical studies.11
In 2012, Hu et al. investigated the impact of dragon’s blood powder with varying grain size on the transdermal absorption and adhesion of ZJHX paste, finding that, with decreasing grain size, penetration of dracorhodin increased, thus promoting transdermal permeability and adhesion.12
Lieu et al. assessed the wound healing potential of Resina Draconis, derived from D. cochinchinensis, which has long been used in traditional medicines by various cultures. In this 2013 evaluation, the investigators substantiated the traditional uses of this herb for wound healing, using excision and incision models in rats. Animals treated with D. cochinchinensis resin displayed significantly superior wound contraction and tensile strength as compared with controls, with histopathological results revealing better microvessel density and growth factor expression levels.13
In 2017, Jiang et al. showed that dracorhodin percolate, derived from dragon’s blood and used extensively to treat wound healing in TCM, accelerated wound healing in Wistar rats.14 A year later, they found that the use of dracorhodin perchlorate was effective in regulating fibroblast proliferation in vitro and in vivo to promote wound healing in rats. In addition, they noted that phosphorylated–extracellular signal-regulated kinase (ERK) in the wound tissue significantly increased with treatment of dracorhodin perchlorate ointment. The researchers called for clinical trials testing this compound in humans as the next step.15
In 2015, Namjoyan et al. conducted a randomized, double-blind, placebo-controlled clinical trial in 60 patients (between 14 and 65 years old) to assess the wound healing effect of a dragon’s blood cream on skin tag removal. Patients were visited every third day during this 3-week study, after which a significant difference in mean wound healing duration was identified. The investigators attributed the accelerated wound healing action to the phenolic constituents and alkaloid taspine in the resin. They also concluded that dragon’s blood warrants inclusion in the wound healing arsenal, while calling for studies in larger populations.16
Conclusion
The red resin extracts of multiple species of plants have and continue to be identified as “dragon’s blood.” This exudate has been used for various medical indications in traditional medicine for several centuries. Despite this lengthy history, modern research is hardly robust. Nevertheless, there are many credible reports of significant salutary activities associated with these resins and some evidence of cutaneous benefits. Much more research is necessary to determine how useful these ingredients are, despite their present use in a number of marketed cosmeceutical agents.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Gupta D et al. J Ethnopharmacol. 2008 Feb 12;115(3):361-80.
2. Jura-Morawiec J & Tulik. Chemoecology. 2016;26:101-5.
3. Nguyen JK et al. J Cosmet Dermatol. 2020 Jul;19(7):155-69.
4. Fan JY et al. Molecules. 2014 Jul 22;19(7):10650-69.
5. Zhang W et al. Zhongguo Zhong Yao Za Zhi. 2016 Apr;41(7):1354-7.
6. Sun J et al. J Ethnopharmacol. 2019 Nov 15;244:112138.
7. Wang H et al. Fitoterapia. 2017 Sep;121:1-5.
8. Al-Fatimi M. Plants (Basel). 2018 Oct 26;7(4):91.
9. Pona A et al. Dermatol Ther. 2019 Mar;32(2):e12786.10. Pieters L et al. Phytomedicine. 1995 Jul;2(1):17-22.
11. Jones K. J Altern Complement Med. 2003 Dec;9(6):877-96.
12. Hu Q et al. Zhongguo Zhong Yao Za Zhi. 2012 Dec;37(23):3549-53.
13. Liu H et al. Evid Based Complement Alternat Med. 2013;2013:709865.
14. Jiang XW et al. Evid Based Complement Alternat Med. 2017:8950516.
15. Jiang X et al. J Pharmacol Sci. 2018 Feb;136(2):66-72.
16. Namjoyan F et al. J Tradit Complement Med. 2015 Jan 22;6(1):37-40.
The use of dragon’s blood is renowned among various medical traditions around the world.1,2 It is known to confer anti-inflammatory, antioxidant, antitumor, antimicrobial, and wound healing benefits, among others. Dragon’s blood and its characteristic red sap has also been used in folk magic and as a coloring substance and varnish.1 In addition, dragon’s blood resin is one of the many botanical agents with roots in traditional medicine that are among the bioactive ingredients used in the booming contemporary Korean cosmeceutical agent market.3.
Many plants, only some have dermatologic properties
Essentially, the moniker “dragon’s blood” describes the deep red resin or sap that has been derived from multiple plant sources – primarily from the genera Daemonorops, Dracaena, Croton, and Pterocarpus – over multiple centuries.2,4 In traditional Chinese medicine (TCM), various plants have been used as dragon’s blood, including Butea monosperma, Liquidambar formosana, Daemonorops draco, and, more commonly now, Dracaena cochinchinensis.5
Chemical constituents and activity
Dragon’s blood represents the red exudate culled from 27 species of plants from four families. Among the six Dracaena plants (D. cochinchinensis, D. cambodiana, D. cinnabari, D. draco, D. loureiroi, and D. schizantha) from which dragon’s blood is derived, flavonoids and their oligomers are considered the main active constituents. Analgesic, anti-inflammatory, antibacterial, hypolipidemic, hypoglycemic, and cytotoxic activities have been associated with these botanicals.6
D. cochinchinensis is one source of the ethnomedicine “dragon’s blood” that has long been used in TCM. Contemporary studies have shown that the resin of D. cochinchinensis – key constituents of which include loureirin A, loureirin B, loureirin C, cochinchinenin, socotrin-4’-ol, 4’,7-dihydroxyflavan, 4-methylcholest-7-ene-3-ol, ethylparaben, resveratrol, and hydroxyphenol – exhibits antibacterial, anti-inflammatory, analgesic, antidiabetic, and antitumor activities. It has also been shown to support skin repair.4
In 2017, Wang et al. reported that flavonoids from artificially induced dragon’s blood of D. cambodiana showed antibacterial properties.7 The next year, Al Fatimi reported that the dragon’s blood derived from D. cinnabari is a key plant on Yemen’s Socotra Island, where it is used for its antifungal and antioxidant properties to treat various dermal, dental, eye, and gastrointestinal diseases in humans.8Croton lechleri (also one of the plants known as dragon’s blood), a medicinal plant found in the Amazon rainforest and characterized by its red sap, has been shown in preclinical studies to display anti-inflammatory, antioxidant, antimicrobial, antifungal, and antineoplastic activity. Pona et al. note that, while clinical studies of C. lechleri suggest wound healing and antiviral effects, the current use of this plant has limited cutaneous applications.9
Wound healing activity
In 1995, Pieters et al. performed an in vivo study on rats to assess the wound healing activity of dragon’s blood (Croton spp.) from South America. In comparing the effects with those of synthetic proanthocyanidins, the researchers verified the beneficial impact of dragon’s blood in stimulating wound contraction, crust formation, new collagen development, and epithelial layer regeneration. The dragon’s blood component 3’,4-O-dimethylcedrusin was also found to enhance healing by promoting fibroblast and collagen formation, though it was not as effective as crude dragon’s blood. The authors ascribed this effect to the proanthocyanidins in the plant.10
Late in 2003, Jones published a literature review on the evidence related to Croton lechleri (known in South America as “sangre de drago” or dragon’s blood) in support of various biological effects, particularly anti-inflammatory and wound healing capability. The results from multiple in vitro and in vivo investigations buttressed previous ethnomedical justifications for the use of dragon’s blood to treat herpes, insect bites, stomach ulcers, tumors, wounds, and diarrhea, as well as other conditions. Jones added that the sap of the plant has exhibited low toxicity and has been well tolerated in clinical studies.11
In 2012, Hu et al. investigated the impact of dragon’s blood powder with varying grain size on the transdermal absorption and adhesion of ZJHX paste, finding that, with decreasing grain size, penetration of dracorhodin increased, thus promoting transdermal permeability and adhesion.12
Lieu et al. assessed the wound healing potential of Resina Draconis, derived from D. cochinchinensis, which has long been used in traditional medicines by various cultures. In this 2013 evaluation, the investigators substantiated the traditional uses of this herb for wound healing, using excision and incision models in rats. Animals treated with D. cochinchinensis resin displayed significantly superior wound contraction and tensile strength as compared with controls, with histopathological results revealing better microvessel density and growth factor expression levels.13
In 2017, Jiang et al. showed that dracorhodin percolate, derived from dragon’s blood and used extensively to treat wound healing in TCM, accelerated wound healing in Wistar rats.14 A year later, they found that the use of dracorhodin perchlorate was effective in regulating fibroblast proliferation in vitro and in vivo to promote wound healing in rats. In addition, they noted that phosphorylated–extracellular signal-regulated kinase (ERK) in the wound tissue significantly increased with treatment of dracorhodin perchlorate ointment. The researchers called for clinical trials testing this compound in humans as the next step.15
In 2015, Namjoyan et al. conducted a randomized, double-blind, placebo-controlled clinical trial in 60 patients (between 14 and 65 years old) to assess the wound healing effect of a dragon’s blood cream on skin tag removal. Patients were visited every third day during this 3-week study, after which a significant difference in mean wound healing duration was identified. The investigators attributed the accelerated wound healing action to the phenolic constituents and alkaloid taspine in the resin. They also concluded that dragon’s blood warrants inclusion in the wound healing arsenal, while calling for studies in larger populations.16
Conclusion
The red resin extracts of multiple species of plants have and continue to be identified as “dragon’s blood.” This exudate has been used for various medical indications in traditional medicine for several centuries. Despite this lengthy history, modern research is hardly robust. Nevertheless, there are many credible reports of significant salutary activities associated with these resins and some evidence of cutaneous benefits. Much more research is necessary to determine how useful these ingredients are, despite their present use in a number of marketed cosmeceutical agents.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Gupta D et al. J Ethnopharmacol. 2008 Feb 12;115(3):361-80.
2. Jura-Morawiec J & Tulik. Chemoecology. 2016;26:101-5.
3. Nguyen JK et al. J Cosmet Dermatol. 2020 Jul;19(7):155-69.
4. Fan JY et al. Molecules. 2014 Jul 22;19(7):10650-69.
5. Zhang W et al. Zhongguo Zhong Yao Za Zhi. 2016 Apr;41(7):1354-7.
6. Sun J et al. J Ethnopharmacol. 2019 Nov 15;244:112138.
7. Wang H et al. Fitoterapia. 2017 Sep;121:1-5.
8. Al-Fatimi M. Plants (Basel). 2018 Oct 26;7(4):91.
9. Pona A et al. Dermatol Ther. 2019 Mar;32(2):e12786.10. Pieters L et al. Phytomedicine. 1995 Jul;2(1):17-22.
11. Jones K. J Altern Complement Med. 2003 Dec;9(6):877-96.
12. Hu Q et al. Zhongguo Zhong Yao Za Zhi. 2012 Dec;37(23):3549-53.
13. Liu H et al. Evid Based Complement Alternat Med. 2013;2013:709865.
14. Jiang XW et al. Evid Based Complement Alternat Med. 2017:8950516.
15. Jiang X et al. J Pharmacol Sci. 2018 Feb;136(2):66-72.
16. Namjoyan F et al. J Tradit Complement Med. 2015 Jan 22;6(1):37-40.
The use of dragon’s blood is renowned among various medical traditions around the world.1,2 It is known to confer anti-inflammatory, antioxidant, antitumor, antimicrobial, and wound healing benefits, among others. Dragon’s blood and its characteristic red sap has also been used in folk magic and as a coloring substance and varnish.1 In addition, dragon’s blood resin is one of the many botanical agents with roots in traditional medicine that are among the bioactive ingredients used in the booming contemporary Korean cosmeceutical agent market.3.
Many plants, only some have dermatologic properties
Essentially, the moniker “dragon’s blood” describes the deep red resin or sap that has been derived from multiple plant sources – primarily from the genera Daemonorops, Dracaena, Croton, and Pterocarpus – over multiple centuries.2,4 In traditional Chinese medicine (TCM), various plants have been used as dragon’s blood, including Butea monosperma, Liquidambar formosana, Daemonorops draco, and, more commonly now, Dracaena cochinchinensis.5
Chemical constituents and activity
Dragon’s blood represents the red exudate culled from 27 species of plants from four families. Among the six Dracaena plants (D. cochinchinensis, D. cambodiana, D. cinnabari, D. draco, D. loureiroi, and D. schizantha) from which dragon’s blood is derived, flavonoids and their oligomers are considered the main active constituents. Analgesic, anti-inflammatory, antibacterial, hypolipidemic, hypoglycemic, and cytotoxic activities have been associated with these botanicals.6
D. cochinchinensis is one source of the ethnomedicine “dragon’s blood” that has long been used in TCM. Contemporary studies have shown that the resin of D. cochinchinensis – key constituents of which include loureirin A, loureirin B, loureirin C, cochinchinenin, socotrin-4’-ol, 4’,7-dihydroxyflavan, 4-methylcholest-7-ene-3-ol, ethylparaben, resveratrol, and hydroxyphenol – exhibits antibacterial, anti-inflammatory, analgesic, antidiabetic, and antitumor activities. It has also been shown to support skin repair.4
In 2017, Wang et al. reported that flavonoids from artificially induced dragon’s blood of D. cambodiana showed antibacterial properties.7 The next year, Al Fatimi reported that the dragon’s blood derived from D. cinnabari is a key plant on Yemen’s Socotra Island, where it is used for its antifungal and antioxidant properties to treat various dermal, dental, eye, and gastrointestinal diseases in humans.8Croton lechleri (also one of the plants known as dragon’s blood), a medicinal plant found in the Amazon rainforest and characterized by its red sap, has been shown in preclinical studies to display anti-inflammatory, antioxidant, antimicrobial, antifungal, and antineoplastic activity. Pona et al. note that, while clinical studies of C. lechleri suggest wound healing and antiviral effects, the current use of this plant has limited cutaneous applications.9
Wound healing activity
In 1995, Pieters et al. performed an in vivo study on rats to assess the wound healing activity of dragon’s blood (Croton spp.) from South America. In comparing the effects with those of synthetic proanthocyanidins, the researchers verified the beneficial impact of dragon’s blood in stimulating wound contraction, crust formation, new collagen development, and epithelial layer regeneration. The dragon’s blood component 3’,4-O-dimethylcedrusin was also found to enhance healing by promoting fibroblast and collagen formation, though it was not as effective as crude dragon’s blood. The authors ascribed this effect to the proanthocyanidins in the plant.10
Late in 2003, Jones published a literature review on the evidence related to Croton lechleri (known in South America as “sangre de drago” or dragon’s blood) in support of various biological effects, particularly anti-inflammatory and wound healing capability. The results from multiple in vitro and in vivo investigations buttressed previous ethnomedical justifications for the use of dragon’s blood to treat herpes, insect bites, stomach ulcers, tumors, wounds, and diarrhea, as well as other conditions. Jones added that the sap of the plant has exhibited low toxicity and has been well tolerated in clinical studies.11
In 2012, Hu et al. investigated the impact of dragon’s blood powder with varying grain size on the transdermal absorption and adhesion of ZJHX paste, finding that, with decreasing grain size, penetration of dracorhodin increased, thus promoting transdermal permeability and adhesion.12
Lieu et al. assessed the wound healing potential of Resina Draconis, derived from D. cochinchinensis, which has long been used in traditional medicines by various cultures. In this 2013 evaluation, the investigators substantiated the traditional uses of this herb for wound healing, using excision and incision models in rats. Animals treated with D. cochinchinensis resin displayed significantly superior wound contraction and tensile strength as compared with controls, with histopathological results revealing better microvessel density and growth factor expression levels.13
In 2017, Jiang et al. showed that dracorhodin percolate, derived from dragon’s blood and used extensively to treat wound healing in TCM, accelerated wound healing in Wistar rats.14 A year later, they found that the use of dracorhodin perchlorate was effective in regulating fibroblast proliferation in vitro and in vivo to promote wound healing in rats. In addition, they noted that phosphorylated–extracellular signal-regulated kinase (ERK) in the wound tissue significantly increased with treatment of dracorhodin perchlorate ointment. The researchers called for clinical trials testing this compound in humans as the next step.15
In 2015, Namjoyan et al. conducted a randomized, double-blind, placebo-controlled clinical trial in 60 patients (between 14 and 65 years old) to assess the wound healing effect of a dragon’s blood cream on skin tag removal. Patients were visited every third day during this 3-week study, after which a significant difference in mean wound healing duration was identified. The investigators attributed the accelerated wound healing action to the phenolic constituents and alkaloid taspine in the resin. They also concluded that dragon’s blood warrants inclusion in the wound healing arsenal, while calling for studies in larger populations.16
Conclusion
The red resin extracts of multiple species of plants have and continue to be identified as “dragon’s blood.” This exudate has been used for various medical indications in traditional medicine for several centuries. Despite this lengthy history, modern research is hardly robust. Nevertheless, there are many credible reports of significant salutary activities associated with these resins and some evidence of cutaneous benefits. Much more research is necessary to determine how useful these ingredients are, despite their present use in a number of marketed cosmeceutical agents.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Gupta D et al. J Ethnopharmacol. 2008 Feb 12;115(3):361-80.
2. Jura-Morawiec J & Tulik. Chemoecology. 2016;26:101-5.
3. Nguyen JK et al. J Cosmet Dermatol. 2020 Jul;19(7):155-69.
4. Fan JY et al. Molecules. 2014 Jul 22;19(7):10650-69.
5. Zhang W et al. Zhongguo Zhong Yao Za Zhi. 2016 Apr;41(7):1354-7.
6. Sun J et al. J Ethnopharmacol. 2019 Nov 15;244:112138.
7. Wang H et al. Fitoterapia. 2017 Sep;121:1-5.
8. Al-Fatimi M. Plants (Basel). 2018 Oct 26;7(4):91.
9. Pona A et al. Dermatol Ther. 2019 Mar;32(2):e12786.10. Pieters L et al. Phytomedicine. 1995 Jul;2(1):17-22.
11. Jones K. J Altern Complement Med. 2003 Dec;9(6):877-96.
12. Hu Q et al. Zhongguo Zhong Yao Za Zhi. 2012 Dec;37(23):3549-53.
13. Liu H et al. Evid Based Complement Alternat Med. 2013;2013:709865.
14. Jiang XW et al. Evid Based Complement Alternat Med. 2017:8950516.
15. Jiang X et al. J Pharmacol Sci. 2018 Feb;136(2):66-72.
16. Namjoyan F et al. J Tradit Complement Med. 2015 Jan 22;6(1):37-40.
What’s under my toenail?
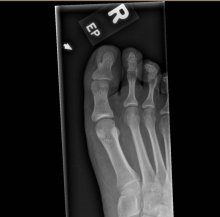
After the teledermatology consultation, an x-ray was recommended. The x-ray showed an elongated irregular radiopaque mass projecting from the anterior medial aspect of the midshaft of the distal phalanx of the great toe (Picture 3). With these findings, subungual exostosis was suspected, and she was referred to orthopedic surgery for excision of the lesion. Histopathology showed a stack of trabecular bone with a fibrocartilaginous cap, confirming the diagnosis of subungual exostosis.
Subungual exostosis is a benign osteocartilaginous tumor, first described by Dupuytren in 1874. These lesions are rare and are seen mainly in children and young adults. Females appear to be affected more often than males.1 In a systematic review by DaCambra and colleagues, 55% of the cases occur in patients aged younger than 18 years, and the hallux was the most commonly affected digit, though any finger or toe can be affected.2 There are reported case of congenital multiple exostosis delineated to translocation t(X;6)(q22;q13-14).3
The exact cause of these lesions is unknown, but there are multiple theories, which include a reactive process secondary to trauma, infection, or genetic causes. Pathologic examination of the lesions shows an osseous center covered by a fibrocartilaginous cap. There is proliferation of spindle cells that generate cartilage, which later forms trabecular bone.4
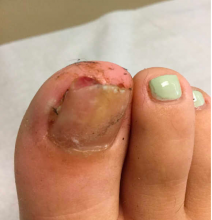
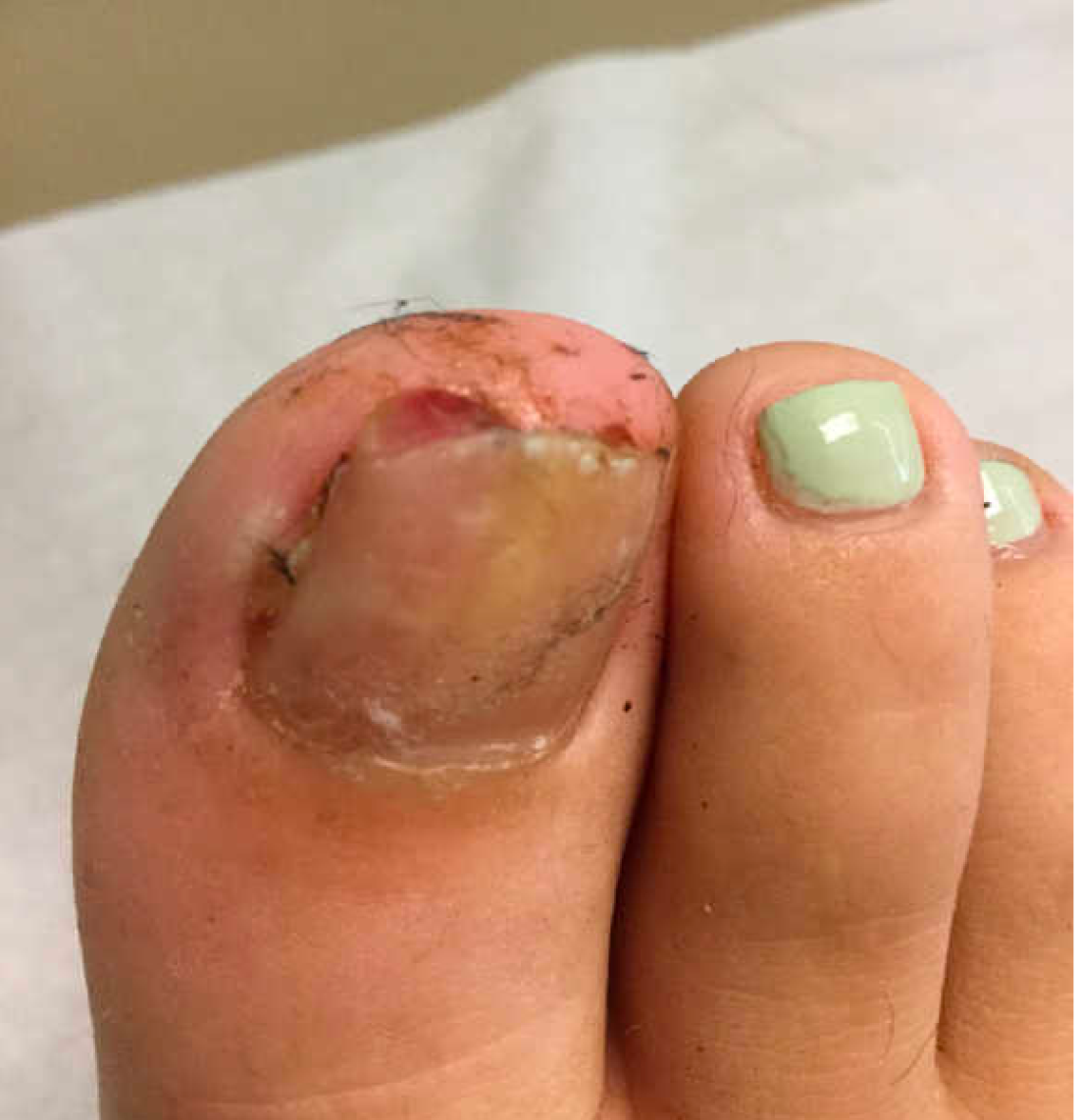
On physical examination, subungual exostosis appear like a firm, fixed nodule with a hyperkeratotic smooth surface at the distal end of the nail bed, that slowly grows and can distort and lift up the nail. Dermoscopy features of these lesions include vascular ectasia, hyperkeratosis, onycholysis, and ulceration.
The differential diagnosis of subungual growths includes osteochondromas, which can present in a similar way but are rarer. Pathologic examination is usually required to differentiate between both lesions.5 In exostoses, bone is formed directly from fibrous tissue, whereas in osteochondromas they derive from enchondral ossification.6 The cartilaginous cap of this lesion is what helps to differentiate it in histopathology. In subungual exostosis, the cap is composed of fibrocartilage, while in osteochondromas it is made of hyaline cartilage similar to what is seen in normal growing epiphysis.5 Subungual exostosis can be confused with pyogenic granulomas and verruca, and often are treated as such, which delays appropriate surgical management.
Firm, slow-growing tumors in the fingers or toes of children should raise suspicion for underlying bony lesions like subungual exostosis and osteochondromas. X-rays of the lesion should be performed in order to clarify the diagnosis. Referral to orthopedic surgery is needed for definitive surgical management.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Zhang W et al. JAAD Case Rep. 2020 Jun 1;6(8):725-6.
2. DaCambra MP et al. Clin Orthop Relat Res. 2014 Apr;472(4):1251-9.
3. Torlazzi C et al. Int J Cancer. 2006;118:1972-6.
4. Calonje E et al. McKee’s pathology of the skin: With clinical correlations. (4th ed.) Philadelphia: Elsevier/Saunders, 2012.
5. Lee SK et al. Foot Ankle Int. 2007 May;28(5):595-601.
6. Mavrogenis A et al. Orthopedics. 2008 Oct;31(10).
After the teledermatology consultation, an x-ray was recommended. The x-ray showed an elongated irregular radiopaque mass projecting from the anterior medial aspect of the midshaft of the distal phalanx of the great toe (Picture 3). With these findings, subungual exostosis was suspected, and she was referred to orthopedic surgery for excision of the lesion. Histopathology showed a stack of trabecular bone with a fibrocartilaginous cap, confirming the diagnosis of subungual exostosis.
Subungual exostosis is a benign osteocartilaginous tumor, first described by Dupuytren in 1874. These lesions are rare and are seen mainly in children and young adults. Females appear to be affected more often than males.1 In a systematic review by DaCambra and colleagues, 55% of the cases occur in patients aged younger than 18 years, and the hallux was the most commonly affected digit, though any finger or toe can be affected.2 There are reported case of congenital multiple exostosis delineated to translocation t(X;6)(q22;q13-14).3
The exact cause of these lesions is unknown, but there are multiple theories, which include a reactive process secondary to trauma, infection, or genetic causes. Pathologic examination of the lesions shows an osseous center covered by a fibrocartilaginous cap. There is proliferation of spindle cells that generate cartilage, which later forms trabecular bone.4
On physical examination, subungual exostosis appear like a firm, fixed nodule with a hyperkeratotic smooth surface at the distal end of the nail bed, that slowly grows and can distort and lift up the nail. Dermoscopy features of these lesions include vascular ectasia, hyperkeratosis, onycholysis, and ulceration.
The differential diagnosis of subungual growths includes osteochondromas, which can present in a similar way but are rarer. Pathologic examination is usually required to differentiate between both lesions.5 In exostoses, bone is formed directly from fibrous tissue, whereas in osteochondromas they derive from enchondral ossification.6 The cartilaginous cap of this lesion is what helps to differentiate it in histopathology. In subungual exostosis, the cap is composed of fibrocartilage, while in osteochondromas it is made of hyaline cartilage similar to what is seen in normal growing epiphysis.5 Subungual exostosis can be confused with pyogenic granulomas and verruca, and often are treated as such, which delays appropriate surgical management.
Firm, slow-growing tumors in the fingers or toes of children should raise suspicion for underlying bony lesions like subungual exostosis and osteochondromas. X-rays of the lesion should be performed in order to clarify the diagnosis. Referral to orthopedic surgery is needed for definitive surgical management.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Zhang W et al. JAAD Case Rep. 2020 Jun 1;6(8):725-6.
2. DaCambra MP et al. Clin Orthop Relat Res. 2014 Apr;472(4):1251-9.
3. Torlazzi C et al. Int J Cancer. 2006;118:1972-6.
4. Calonje E et al. McKee’s pathology of the skin: With clinical correlations. (4th ed.) Philadelphia: Elsevier/Saunders, 2012.
5. Lee SK et al. Foot Ankle Int. 2007 May;28(5):595-601.
6. Mavrogenis A et al. Orthopedics. 2008 Oct;31(10).
After the teledermatology consultation, an x-ray was recommended. The x-ray showed an elongated irregular radiopaque mass projecting from the anterior medial aspect of the midshaft of the distal phalanx of the great toe (Picture 3). With these findings, subungual exostosis was suspected, and she was referred to orthopedic surgery for excision of the lesion. Histopathology showed a stack of trabecular bone with a fibrocartilaginous cap, confirming the diagnosis of subungual exostosis.
Subungual exostosis is a benign osteocartilaginous tumor, first described by Dupuytren in 1874. These lesions are rare and are seen mainly in children and young adults. Females appear to be affected more often than males.1 In a systematic review by DaCambra and colleagues, 55% of the cases occur in patients aged younger than 18 years, and the hallux was the most commonly affected digit, though any finger or toe can be affected.2 There are reported case of congenital multiple exostosis delineated to translocation t(X;6)(q22;q13-14).3
The exact cause of these lesions is unknown, but there are multiple theories, which include a reactive process secondary to trauma, infection, or genetic causes. Pathologic examination of the lesions shows an osseous center covered by a fibrocartilaginous cap. There is proliferation of spindle cells that generate cartilage, which later forms trabecular bone.4
On physical examination, subungual exostosis appear like a firm, fixed nodule with a hyperkeratotic smooth surface at the distal end of the nail bed, that slowly grows and can distort and lift up the nail. Dermoscopy features of these lesions include vascular ectasia, hyperkeratosis, onycholysis, and ulceration.
The differential diagnosis of subungual growths includes osteochondromas, which can present in a similar way but are rarer. Pathologic examination is usually required to differentiate between both lesions.5 In exostoses, bone is formed directly from fibrous tissue, whereas in osteochondromas they derive from enchondral ossification.6 The cartilaginous cap of this lesion is what helps to differentiate it in histopathology. In subungual exostosis, the cap is composed of fibrocartilage, while in osteochondromas it is made of hyaline cartilage similar to what is seen in normal growing epiphysis.5 Subungual exostosis can be confused with pyogenic granulomas and verruca, and often are treated as such, which delays appropriate surgical management.
Firm, slow-growing tumors in the fingers or toes of children should raise suspicion for underlying bony lesions like subungual exostosis and osteochondromas. X-rays of the lesion should be performed in order to clarify the diagnosis. Referral to orthopedic surgery is needed for definitive surgical management.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Zhang W et al. JAAD Case Rep. 2020 Jun 1;6(8):725-6.
2. DaCambra MP et al. Clin Orthop Relat Res. 2014 Apr;472(4):1251-9.
3. Torlazzi C et al. Int J Cancer. 2006;118:1972-6.
4. Calonje E et al. McKee’s pathology of the skin: With clinical correlations. (4th ed.) Philadelphia: Elsevier/Saunders, 2012.
5. Lee SK et al. Foot Ankle Int. 2007 May;28(5):595-601.
6. Mavrogenis A et al. Orthopedics. 2008 Oct;31(10).
A 13-year-old female was seen by her pediatrician for a lesion that had been on her right toe for about 6 months. She is unaware of any trauma to the area. The lesion has been growing slowly and recently it started lifting up the nail, became tender, and was bleeding, which is the reason why she sought care.
At the pediatrician's office, he noted a pink crusted papule under the nail. The nail was lifting up and was tender to the touch. She is a healthy girl who is not taking any medications and has no allergies. There is no family history of similar lesions.
The pediatrician took a picture of the lesion and he send it to our pediatric teledermatology service for consultation.
Ulcerated and Verrucous Plaque on the Chest
The Diagnosis: Disseminated Coccidioidomycosis
A6-mm punch biopsy was performed at the periphery of the ulcerated cutaneous lesion on the chest revealing extensive spherules. Serum antibody immunodiffusion for histoplasmosis and blastomycoses both were negative; however, B-D-glucan assay was positive at 364 pg/mL (reference range: <60 pg/mL, negative). Initial HIV-1 and HIV-2 antibody and antigen testing was negative as well as repeat testing at 3 weeks. Immunodiffusion for Coccidioides IgM and IgG was positive, and cocci antibody IgG complement fixation assays were positive at titers of 1:64 (reference range: <1:2, negative). A computed tomography needle-guided biopsy of the paravertebral soft tissue was performed. Gram stains and bacterial cultures of the biopsies were negative; however, fungal cultures were notable for growth of Coccidioides. Given the pertinent testing, a diagnosis of disseminated coccidioidomycosis was made.
Cutaneous coccidioidomycosis can occur in 3 situations: direct inoculation (primary cutaneous coccidioidomycosis), disseminated infection (disseminated cutaneous coccidioidomycosis), or as a reactive component of pulmonary infection.1,2 Of them, primary and disseminated cutaneous coccidioidomycosis are organism specific and display characteristic spherules and fungus on histopathology and cultures, respectively. Reactive coccidioidomycosis differs from organism-specific disease, as it does not contain spherules in histopathologic sections of tissue biopsies.1 Reactive skin manifestations occur in 12% to 50% of patients with primary pulmonary infection and include erythema nodosum, erythema multiforme, acute generalized exanthema, reactive interstitial granulomatous dermatitis, and Sweet syndrome.3
Organism-specific cutaneous coccidioidomycosis most often is correlated with hematogenous dissemination of primary pulmonary disease rather than direct inoculation of skin.1 The skin is the most common site of extrapulmonary involvement in disseminated coccidioidomycosis, and cutaneous lesions have been reported in 15% to 67% of cases of disseminated disease.1,4 In cutaneous disseminated disease, nodules, papules, macules, and verrucous plaques have been described. In a case series of disseminated cutaneous coccidioidomycosis, nodules were the most common cutaneous presentation and occurred in 39% (7/18) of patients, while verrucous plaques were the rarest and occurred in only 6% (1/18) of patients.5
The rate of coccidioidomycosis dissemination varies based on exposure and patient characteristics. Increased rates of dissemination have been reported in patients of African and Filipino descent, along with individuals that are immunosuppressed due to disease or medical therapy. Dissemination is clinically significant, as patients with multifocal dissemination have a greater than 50% risk for mortality.6
Disseminated coccidioidomycosis is a relatively rare manifestation of Coccidioides infection; approximately 1.6% of patients exposed to and infected with Coccidioides ultimately will develop systemic or disseminated disease.7,8 Although the rates of primary pulmonary infection are similar between patients of varying ethnicities, the rates of dissemination are higher in patients of African and Filipino ethnicity.8 In population studies of coccidioidomycosis (N=332), Black patients represented 33.3% (4/12) of disseminated cases but only 8.7% of Coccidioides cases overall.7
Population studies of Black patients with coccidioidomycosis have shown a 4-fold higher predisposition for severe disease compared to mild disease.9 Spondylitis and meningitis also are disproportionately more common in Black patients.8 Black patients comprised 75% of all spondylitis cases in a cohort where only 25% of patients were Black. Additionally, 33% of all meningitis cases occurred in Black patients in a cohort where 8% of total cases were Black patients.8 Within the United States, the highest rates of coccidioidomycosis meningitis are seen in Black patients.10
The pathophysiology underlying the increased susceptibility of individuals of African or Filipino descent to disseminated and severe coccidioidomycosis remains unknown.8 The level of vulnerability within this patient population has no association with increased environmental exposure or poor immunologic response to Coccidioides, as demonstrated by the ability of these populations to respond to experimental vaccination and skin testing (spherulin, coccidioidin) to a similar extent as other ethnicities.8 Class II HLA-DRB1*1301 alleles have been associated with an increased risk for severe disseminated Coccidioides infection regardless of ethnicity; however, these alleles are not overrepresented in these patient populations.8
In patients with primary pulmonary coccidioidomycosis with no evidence of dissemination, guidelines generally recommend offering treatment to groups at high risk of dissemination, such as pregnant women and patients with diabetes mellitus. Given the high incidence of disseminated and severe disease in Black and Filipino patients, some guidelines endorse treatment of all cases of coccidioidomycosis in this patient population.8 No current data are available to help determine whether this broad treatment approach reduces the development of disseminated infection in these populations. Frequent monitoring for disease progression and/or dissemination involving clinical and laboratory reevaluation every 3 months for 2 years is highly recommended.8
Treatment generally is based on location and severity of infection, with disseminated nonmeningeal infection being treated with oral azole therapy (ketoconazole, itraconazole, or fluconazole).11 If there is involvement of the central nervous system structures or rapidly worsening disease despite azole therapy, amphotericin B is recommended at 0.5 to 0.7 mg/kg daily. In patients with disseminated meningeal infection, oral fluconazole (800–1000 mg/d) or a combination of an azole with intrathecal amphotericin B (0.01–1.5 mg/dose, interval ranging from daily to 1 week) is recommended to improve response.11
The differential diagnosis of cutaneous disseminated coccidioidomycosis is broad and includes other systemic endemic mycoses (histoplasmosis, blastomycosis) and infections (mycobacteria, leishmania). Lupus vulgaris, a form of cutaneous tuberculosis, presents as a palpable tubercular lesion that may coalesce into erythematous plaques, which may mimic endemic mycoses, especially in patients with risk factors for both infectious etiologies such as our patient.12 Disseminated histoplasmosis may present as polymorphic plaques, pustules, nodules, and ulcerated skin lesions, whereas disseminated blastomycosis characteristically presents as a crusted verrucous lesion with raised borders and painful ulcers, both of which may mimic coccidioidomycosis.13 Biopsy would reveal the characteristic intracellular yeast in Histoplasma capsulatum and broad-based budding yeast form of Blastomyces dermatitidis in histoplasmosis and blastomycosis, respectively, in contrast to the spherules seen in our patient’s biopsy.13 Localized cutaneous leishmaniasis initially develops as a nodular or papular lesion and can progress to open ulcerations with raised borders. Biopsy and histopathology would reveal round protozoal amastigotes.14 Other diagnoses that should be considered include mycetoma, nocardiosis, and sporotrichosis.15 As the cutaneous manifestations of Coccidioides infections are varied, a broad differential diagnosis should be maintained, and probable environmental and infectious exposures should be considered prior to ordering diagnostic studies.
- Garcia Garcia SC, Salas Alanis JC, Flores MG, et al. Coccidioidomycosis and the skin: a comprehensive review. An Bras Dermatol. 2015; 90:610-619.
- DiCaudo DJ. Coccidioidomycosis: a review and update. J Am Acad Dermatol. 2006;55:929-942; quiz 943-925.
- DiCaudo DJ, Yiannias JA, Laman SD, et al. The exanthem of acute pulmonary coccidioidomycosis: clinical and histopathologic features of 3 cases and review of the literature. Arch Dermatol. 2006;142:744-746.
- Blair JE. State-of-the-art treatment of coccidioidomycosis: skin and soft-tissue infections. Ann N Y Acad Sci. 2007;1111:411-421.
- Crum NF, Lederman ER, Stafford CM, et al. Coccidioidomycosis: a descriptive survey of a reemerging disease. clinical characteristics and current controversies. Medicine (Baltimore). 2004;83:149-175.
- Borchers AT, Gershwin ME. The immune response in coccidioidomycosis. Autoimmun Rev. 2010;10:94-102.
- Smith CE, Beard RR. Varieties of coccidioidal infection in relation to the epidemiology and control of the diseases. Am J Public Health Nations Health. 1946;36:1394-1402.
- Ruddy BE, Mayer AP, Ko MG, et al. Coccidioidomycosis in African Americans. Mayo Clin Proc. 2011;86:63-69.
- Louie L, Ng S, Hajjeh R, et al. Influence of host genetics on the severity of coccidioidomycosis. Emerg Infect Dis. 1999;5:672-680.
- McCotter OZ, Benedict K, Engelthaler DM, et al. Update on the epidemiology of coccidioidomycosis in the United States. Med Mycol. 2019;57(suppl 1):S30-S40.
- Galgiani JN, Ampel NM, Catanzaro A, et al. Practice guideline for the treatment of coccidioidomycosis. Infectious Diseases Society of America. Clin Infect Dis. 2000;30:658-661.
- Khadka P, Koirala S, Thapaliya J. Cutaneous tuberculosis: clinicopathologic arrays and diagnostic challenges. Dermatol Res Pract. 2018;2018:7201973.
- Smith JA, Riddell JT, Kauffman CA. Cutaneous manifestations of endemic mycoses. Curr Infect Dis Rep. 2013;15:440-449.
- Scorza BM, Carvalho EM, Wilson ME. Cutaneous manifestations of human and murine leishmaniasis. Int J Mol Sci. 2017;18:1296.
The Diagnosis: Disseminated Coccidioidomycosis
A6-mm punch biopsy was performed at the periphery of the ulcerated cutaneous lesion on the chest revealing extensive spherules. Serum antibody immunodiffusion for histoplasmosis and blastomycoses both were negative; however, B-D-glucan assay was positive at 364 pg/mL (reference range: <60 pg/mL, negative). Initial HIV-1 and HIV-2 antibody and antigen testing was negative as well as repeat testing at 3 weeks. Immunodiffusion for Coccidioides IgM and IgG was positive, and cocci antibody IgG complement fixation assays were positive at titers of 1:64 (reference range: <1:2, negative). A computed tomography needle-guided biopsy of the paravertebral soft tissue was performed. Gram stains and bacterial cultures of the biopsies were negative; however, fungal cultures were notable for growth of Coccidioides. Given the pertinent testing, a diagnosis of disseminated coccidioidomycosis was made.
Cutaneous coccidioidomycosis can occur in 3 situations: direct inoculation (primary cutaneous coccidioidomycosis), disseminated infection (disseminated cutaneous coccidioidomycosis), or as a reactive component of pulmonary infection.1,2 Of them, primary and disseminated cutaneous coccidioidomycosis are organism specific and display characteristic spherules and fungus on histopathology and cultures, respectively. Reactive coccidioidomycosis differs from organism-specific disease, as it does not contain spherules in histopathologic sections of tissue biopsies.1 Reactive skin manifestations occur in 12% to 50% of patients with primary pulmonary infection and include erythema nodosum, erythema multiforme, acute generalized exanthema, reactive interstitial granulomatous dermatitis, and Sweet syndrome.3
Organism-specific cutaneous coccidioidomycosis most often is correlated with hematogenous dissemination of primary pulmonary disease rather than direct inoculation of skin.1 The skin is the most common site of extrapulmonary involvement in disseminated coccidioidomycosis, and cutaneous lesions have been reported in 15% to 67% of cases of disseminated disease.1,4 In cutaneous disseminated disease, nodules, papules, macules, and verrucous plaques have been described. In a case series of disseminated cutaneous coccidioidomycosis, nodules were the most common cutaneous presentation and occurred in 39% (7/18) of patients, while verrucous plaques were the rarest and occurred in only 6% (1/18) of patients.5
The rate of coccidioidomycosis dissemination varies based on exposure and patient characteristics. Increased rates of dissemination have been reported in patients of African and Filipino descent, along with individuals that are immunosuppressed due to disease or medical therapy. Dissemination is clinically significant, as patients with multifocal dissemination have a greater than 50% risk for mortality.6
Disseminated coccidioidomycosis is a relatively rare manifestation of Coccidioides infection; approximately 1.6% of patients exposed to and infected with Coccidioides ultimately will develop systemic or disseminated disease.7,8 Although the rates of primary pulmonary infection are similar between patients of varying ethnicities, the rates of dissemination are higher in patients of African and Filipino ethnicity.8 In population studies of coccidioidomycosis (N=332), Black patients represented 33.3% (4/12) of disseminated cases but only 8.7% of Coccidioides cases overall.7
Population studies of Black patients with coccidioidomycosis have shown a 4-fold higher predisposition for severe disease compared to mild disease.9 Spondylitis and meningitis also are disproportionately more common in Black patients.8 Black patients comprised 75% of all spondylitis cases in a cohort where only 25% of patients were Black. Additionally, 33% of all meningitis cases occurred in Black patients in a cohort where 8% of total cases were Black patients.8 Within the United States, the highest rates of coccidioidomycosis meningitis are seen in Black patients.10
The pathophysiology underlying the increased susceptibility of individuals of African or Filipino descent to disseminated and severe coccidioidomycosis remains unknown.8 The level of vulnerability within this patient population has no association with increased environmental exposure or poor immunologic response to Coccidioides, as demonstrated by the ability of these populations to respond to experimental vaccination and skin testing (spherulin, coccidioidin) to a similar extent as other ethnicities.8 Class II HLA-DRB1*1301 alleles have been associated with an increased risk for severe disseminated Coccidioides infection regardless of ethnicity; however, these alleles are not overrepresented in these patient populations.8
In patients with primary pulmonary coccidioidomycosis with no evidence of dissemination, guidelines generally recommend offering treatment to groups at high risk of dissemination, such as pregnant women and patients with diabetes mellitus. Given the high incidence of disseminated and severe disease in Black and Filipino patients, some guidelines endorse treatment of all cases of coccidioidomycosis in this patient population.8 No current data are available to help determine whether this broad treatment approach reduces the development of disseminated infection in these populations. Frequent monitoring for disease progression and/or dissemination involving clinical and laboratory reevaluation every 3 months for 2 years is highly recommended.8
Treatment generally is based on location and severity of infection, with disseminated nonmeningeal infection being treated with oral azole therapy (ketoconazole, itraconazole, or fluconazole).11 If there is involvement of the central nervous system structures or rapidly worsening disease despite azole therapy, amphotericin B is recommended at 0.5 to 0.7 mg/kg daily. In patients with disseminated meningeal infection, oral fluconazole (800–1000 mg/d) or a combination of an azole with intrathecal amphotericin B (0.01–1.5 mg/dose, interval ranging from daily to 1 week) is recommended to improve response.11
The differential diagnosis of cutaneous disseminated coccidioidomycosis is broad and includes other systemic endemic mycoses (histoplasmosis, blastomycosis) and infections (mycobacteria, leishmania). Lupus vulgaris, a form of cutaneous tuberculosis, presents as a palpable tubercular lesion that may coalesce into erythematous plaques, which may mimic endemic mycoses, especially in patients with risk factors for both infectious etiologies such as our patient.12 Disseminated histoplasmosis may present as polymorphic plaques, pustules, nodules, and ulcerated skin lesions, whereas disseminated blastomycosis characteristically presents as a crusted verrucous lesion with raised borders and painful ulcers, both of which may mimic coccidioidomycosis.13 Biopsy would reveal the characteristic intracellular yeast in Histoplasma capsulatum and broad-based budding yeast form of Blastomyces dermatitidis in histoplasmosis and blastomycosis, respectively, in contrast to the spherules seen in our patient’s biopsy.13 Localized cutaneous leishmaniasis initially develops as a nodular or papular lesion and can progress to open ulcerations with raised borders. Biopsy and histopathology would reveal round protozoal amastigotes.14 Other diagnoses that should be considered include mycetoma, nocardiosis, and sporotrichosis.15 As the cutaneous manifestations of Coccidioides infections are varied, a broad differential diagnosis should be maintained, and probable environmental and infectious exposures should be considered prior to ordering diagnostic studies.
The Diagnosis: Disseminated Coccidioidomycosis
A6-mm punch biopsy was performed at the periphery of the ulcerated cutaneous lesion on the chest revealing extensive spherules. Serum antibody immunodiffusion for histoplasmosis and blastomycoses both were negative; however, B-D-glucan assay was positive at 364 pg/mL (reference range: <60 pg/mL, negative). Initial HIV-1 and HIV-2 antibody and antigen testing was negative as well as repeat testing at 3 weeks. Immunodiffusion for Coccidioides IgM and IgG was positive, and cocci antibody IgG complement fixation assays were positive at titers of 1:64 (reference range: <1:2, negative). A computed tomography needle-guided biopsy of the paravertebral soft tissue was performed. Gram stains and bacterial cultures of the biopsies were negative; however, fungal cultures were notable for growth of Coccidioides. Given the pertinent testing, a diagnosis of disseminated coccidioidomycosis was made.
Cutaneous coccidioidomycosis can occur in 3 situations: direct inoculation (primary cutaneous coccidioidomycosis), disseminated infection (disseminated cutaneous coccidioidomycosis), or as a reactive component of pulmonary infection.1,2 Of them, primary and disseminated cutaneous coccidioidomycosis are organism specific and display characteristic spherules and fungus on histopathology and cultures, respectively. Reactive coccidioidomycosis differs from organism-specific disease, as it does not contain spherules in histopathologic sections of tissue biopsies.1 Reactive skin manifestations occur in 12% to 50% of patients with primary pulmonary infection and include erythema nodosum, erythema multiforme, acute generalized exanthema, reactive interstitial granulomatous dermatitis, and Sweet syndrome.3
Organism-specific cutaneous coccidioidomycosis most often is correlated with hematogenous dissemination of primary pulmonary disease rather than direct inoculation of skin.1 The skin is the most common site of extrapulmonary involvement in disseminated coccidioidomycosis, and cutaneous lesions have been reported in 15% to 67% of cases of disseminated disease.1,4 In cutaneous disseminated disease, nodules, papules, macules, and verrucous plaques have been described. In a case series of disseminated cutaneous coccidioidomycosis, nodules were the most common cutaneous presentation and occurred in 39% (7/18) of patients, while verrucous plaques were the rarest and occurred in only 6% (1/18) of patients.5
The rate of coccidioidomycosis dissemination varies based on exposure and patient characteristics. Increased rates of dissemination have been reported in patients of African and Filipino descent, along with individuals that are immunosuppressed due to disease or medical therapy. Dissemination is clinically significant, as patients with multifocal dissemination have a greater than 50% risk for mortality.6
Disseminated coccidioidomycosis is a relatively rare manifestation of Coccidioides infection; approximately 1.6% of patients exposed to and infected with Coccidioides ultimately will develop systemic or disseminated disease.7,8 Although the rates of primary pulmonary infection are similar between patients of varying ethnicities, the rates of dissemination are higher in patients of African and Filipino ethnicity.8 In population studies of coccidioidomycosis (N=332), Black patients represented 33.3% (4/12) of disseminated cases but only 8.7% of Coccidioides cases overall.7
Population studies of Black patients with coccidioidomycosis have shown a 4-fold higher predisposition for severe disease compared to mild disease.9 Spondylitis and meningitis also are disproportionately more common in Black patients.8 Black patients comprised 75% of all spondylitis cases in a cohort where only 25% of patients were Black. Additionally, 33% of all meningitis cases occurred in Black patients in a cohort where 8% of total cases were Black patients.8 Within the United States, the highest rates of coccidioidomycosis meningitis are seen in Black patients.10
The pathophysiology underlying the increased susceptibility of individuals of African or Filipino descent to disseminated and severe coccidioidomycosis remains unknown.8 The level of vulnerability within this patient population has no association with increased environmental exposure or poor immunologic response to Coccidioides, as demonstrated by the ability of these populations to respond to experimental vaccination and skin testing (spherulin, coccidioidin) to a similar extent as other ethnicities.8 Class II HLA-DRB1*1301 alleles have been associated with an increased risk for severe disseminated Coccidioides infection regardless of ethnicity; however, these alleles are not overrepresented in these patient populations.8
In patients with primary pulmonary coccidioidomycosis with no evidence of dissemination, guidelines generally recommend offering treatment to groups at high risk of dissemination, such as pregnant women and patients with diabetes mellitus. Given the high incidence of disseminated and severe disease in Black and Filipino patients, some guidelines endorse treatment of all cases of coccidioidomycosis in this patient population.8 No current data are available to help determine whether this broad treatment approach reduces the development of disseminated infection in these populations. Frequent monitoring for disease progression and/or dissemination involving clinical and laboratory reevaluation every 3 months for 2 years is highly recommended.8
Treatment generally is based on location and severity of infection, with disseminated nonmeningeal infection being treated with oral azole therapy (ketoconazole, itraconazole, or fluconazole).11 If there is involvement of the central nervous system structures or rapidly worsening disease despite azole therapy, amphotericin B is recommended at 0.5 to 0.7 mg/kg daily. In patients with disseminated meningeal infection, oral fluconazole (800–1000 mg/d) or a combination of an azole with intrathecal amphotericin B (0.01–1.5 mg/dose, interval ranging from daily to 1 week) is recommended to improve response.11
The differential diagnosis of cutaneous disseminated coccidioidomycosis is broad and includes other systemic endemic mycoses (histoplasmosis, blastomycosis) and infections (mycobacteria, leishmania). Lupus vulgaris, a form of cutaneous tuberculosis, presents as a palpable tubercular lesion that may coalesce into erythematous plaques, which may mimic endemic mycoses, especially in patients with risk factors for both infectious etiologies such as our patient.12 Disseminated histoplasmosis may present as polymorphic plaques, pustules, nodules, and ulcerated skin lesions, whereas disseminated blastomycosis characteristically presents as a crusted verrucous lesion with raised borders and painful ulcers, both of which may mimic coccidioidomycosis.13 Biopsy would reveal the characteristic intracellular yeast in Histoplasma capsulatum and broad-based budding yeast form of Blastomyces dermatitidis in histoplasmosis and blastomycosis, respectively, in contrast to the spherules seen in our patient’s biopsy.13 Localized cutaneous leishmaniasis initially develops as a nodular or papular lesion and can progress to open ulcerations with raised borders. Biopsy and histopathology would reveal round protozoal amastigotes.14 Other diagnoses that should be considered include mycetoma, nocardiosis, and sporotrichosis.15 As the cutaneous manifestations of Coccidioides infections are varied, a broad differential diagnosis should be maintained, and probable environmental and infectious exposures should be considered prior to ordering diagnostic studies.
- Garcia Garcia SC, Salas Alanis JC, Flores MG, et al. Coccidioidomycosis and the skin: a comprehensive review. An Bras Dermatol. 2015; 90:610-619.
- DiCaudo DJ. Coccidioidomycosis: a review and update. J Am Acad Dermatol. 2006;55:929-942; quiz 943-925.
- DiCaudo DJ, Yiannias JA, Laman SD, et al. The exanthem of acute pulmonary coccidioidomycosis: clinical and histopathologic features of 3 cases and review of the literature. Arch Dermatol. 2006;142:744-746.
- Blair JE. State-of-the-art treatment of coccidioidomycosis: skin and soft-tissue infections. Ann N Y Acad Sci. 2007;1111:411-421.
- Crum NF, Lederman ER, Stafford CM, et al. Coccidioidomycosis: a descriptive survey of a reemerging disease. clinical characteristics and current controversies. Medicine (Baltimore). 2004;83:149-175.
- Borchers AT, Gershwin ME. The immune response in coccidioidomycosis. Autoimmun Rev. 2010;10:94-102.
- Smith CE, Beard RR. Varieties of coccidioidal infection in relation to the epidemiology and control of the diseases. Am J Public Health Nations Health. 1946;36:1394-1402.
- Ruddy BE, Mayer AP, Ko MG, et al. Coccidioidomycosis in African Americans. Mayo Clin Proc. 2011;86:63-69.
- Louie L, Ng S, Hajjeh R, et al. Influence of host genetics on the severity of coccidioidomycosis. Emerg Infect Dis. 1999;5:672-680.
- McCotter OZ, Benedict K, Engelthaler DM, et al. Update on the epidemiology of coccidioidomycosis in the United States. Med Mycol. 2019;57(suppl 1):S30-S40.
- Galgiani JN, Ampel NM, Catanzaro A, et al. Practice guideline for the treatment of coccidioidomycosis. Infectious Diseases Society of America. Clin Infect Dis. 2000;30:658-661.
- Khadka P, Koirala S, Thapaliya J. Cutaneous tuberculosis: clinicopathologic arrays and diagnostic challenges. Dermatol Res Pract. 2018;2018:7201973.
- Smith JA, Riddell JT, Kauffman CA. Cutaneous manifestations of endemic mycoses. Curr Infect Dis Rep. 2013;15:440-449.
- Scorza BM, Carvalho EM, Wilson ME. Cutaneous manifestations of human and murine leishmaniasis. Int J Mol Sci. 2017;18:1296.
- Garcia Garcia SC, Salas Alanis JC, Flores MG, et al. Coccidioidomycosis and the skin: a comprehensive review. An Bras Dermatol. 2015; 90:610-619.
- DiCaudo DJ. Coccidioidomycosis: a review and update. J Am Acad Dermatol. 2006;55:929-942; quiz 943-925.
- DiCaudo DJ, Yiannias JA, Laman SD, et al. The exanthem of acute pulmonary coccidioidomycosis: clinical and histopathologic features of 3 cases and review of the literature. Arch Dermatol. 2006;142:744-746.
- Blair JE. State-of-the-art treatment of coccidioidomycosis: skin and soft-tissue infections. Ann N Y Acad Sci. 2007;1111:411-421.
- Crum NF, Lederman ER, Stafford CM, et al. Coccidioidomycosis: a descriptive survey of a reemerging disease. clinical characteristics and current controversies. Medicine (Baltimore). 2004;83:149-175.
- Borchers AT, Gershwin ME. The immune response in coccidioidomycosis. Autoimmun Rev. 2010;10:94-102.
- Smith CE, Beard RR. Varieties of coccidioidal infection in relation to the epidemiology and control of the diseases. Am J Public Health Nations Health. 1946;36:1394-1402.
- Ruddy BE, Mayer AP, Ko MG, et al. Coccidioidomycosis in African Americans. Mayo Clin Proc. 2011;86:63-69.
- Louie L, Ng S, Hajjeh R, et al. Influence of host genetics on the severity of coccidioidomycosis. Emerg Infect Dis. 1999;5:672-680.
- McCotter OZ, Benedict K, Engelthaler DM, et al. Update on the epidemiology of coccidioidomycosis in the United States. Med Mycol. 2019;57(suppl 1):S30-S40.
- Galgiani JN, Ampel NM, Catanzaro A, et al. Practice guideline for the treatment of coccidioidomycosis. Infectious Diseases Society of America. Clin Infect Dis. 2000;30:658-661.
- Khadka P, Koirala S, Thapaliya J. Cutaneous tuberculosis: clinicopathologic arrays and diagnostic challenges. Dermatol Res Pract. 2018;2018:7201973.
- Smith JA, Riddell JT, Kauffman CA. Cutaneous manifestations of endemic mycoses. Curr Infect Dis Rep. 2013;15:440-449.
- Scorza BM, Carvalho EM, Wilson ME. Cutaneous manifestations of human and murine leishmaniasis. Int J Mol Sci. 2017;18:1296.
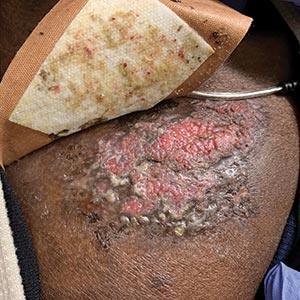
A 36-year-old man presented to an emergency department in the southwestern United States with a cough, fatigue, and worsening back pain associated with night sweats of 1 month’s duration. He experienced a 9.07-kg weight loss, as well as development of a rough, nontender, nonpruritic rash along the left upper chest over the prior month. The patient was born in West Africa and reported that he had moved to the southwestern United States from the eastern United States approximately 6 years prior to presentation. Physical examination on admission revealed a 5×3-cm, purple-brown, verrucous plaque with a central pink cobblestone appearance and ulceration. Chest radiography was notable for perihilar adenopathy with no focal infiltrates or cavitary lesions. Computed tomography and magnetic resonance imaging of the chest were notable for miliary nodules throughout the lungs; extensive lytic spine lesions of cervical, thoracic, and lumbar vertebral bodies and left twelfth rib; and a left paraspinal thoracic epidural soft tissue phlegmon. Initial laboratory investigations revealed peripheral eosinophilia without absolute leukocytosis and a microcytic anemia.
Shedding the super-doctor myth requires an honest look at systemic racism
An overwhelmingly loud and high-pitched screech rattles against your hip. You startle and groan into the pillow as your thoughts settle into conscious awareness. It is 3 a.m. You are a 2nd-year resident trudging through the night shift, alerted to the presence of a new patient awaiting an emergency assessment. You are the only in-house physician. Walking steadfastly toward the emergency unit, you enter and greet the patient. Immediately, you observe a look of surprise followed immediately by a scowl.
You extend a hand, but your greeting is abruptly cut short with: “I want to see a doctor!” You pace your breaths to quell annoyance and resume your introduction, asserting that you are a doctor and indeed the only doctor on duty. After moments of deep sighs and questions regarding your credentials, you persuade the patient to start the interview.
It is now 8 a.m. The frustration of the night starts to ease as you prepare to leave. While gathering your things, a visitor is overheard inquiring the whereabouts of a hospital unit. Volunteering as a guide, you walk the person toward the opposite end of the hospital. Bleary eyed, muscle laxed, and bone weary, you point out the entrance, then turn to leave. The steady rhythm of your steps suddenly halts as you hear from behind: “Thank you! You speak English really well!” Blankly, you stare. Your voice remains mute while your brain screams: “What is that supposed to mean?” But you do not utter a sound, because intuitively, you know the answer.
While reading this scenario, what did you feel? Pride in knowing that the physician was able to successfully navigate a busy night? Relief in the physician’s ability to maintain a professional demeanor despite belittling microaggressions? Are you angry? Would you replay those moments like reruns of a bad TV show? Can you imagine entering your home and collapsing onto the bed as your tears of fury pool over your rumpled sheets?
The emotional release of that morning is seared into my memory. Over the years, I questioned my reactions. Was I too passive? Should I have schooled them on their ignorance? Had I done so, would I have incurred reprimands? Would standing up for myself cause years of hard work to fall away? Moreover, had I defended myself, would I forever have been viewed as “The Angry Black Woman?”
This story is more than a vignette. For me, it is another reminder that, despite how far we have come, we have much further to go. As a Black woman in a professional sphere, I stand upon the shoulders of those who sacrificed for a dream, a greater purpose. My foremothers and forefathers fought bravely and tirelessly so that we could attain levels of success that were only once but a dream. Despite this progress, a grimace, carelessly spoken words, or a mindless gesture remind me that, no matter how much I toil and what levels of success I achieve, when I meet someone for the first time or encounter someone from my past, I find myself wondering whether I am remembered for me or because I am “The Black One.”
Honest look at medicine is imperative
It is important to consider multiple facets of the super-doctor myth. We are dedicated, fearless, authoritative, ambitious individuals. We do not yield to sickness, family obligations, or fatigue. Medicine is a calling, and the patient deserves the utmost respect and professional behavior. Impervious to ethnicity, race, nationality, or creed, we are unbiased and always in service of the greater good. Often, however, I wonder how the expectations of patient-focused, patient-centered care can prevail without an honest look at the vicissitudes facing medicine.
We find ourselves amid a tumultuous year overshadowed by a devastating pandemic that skews heavily toward Black and Brown communities, in addition to political turmoil and racial reckoning that sprang forth from fear, anger, and determination ignited by the murders of Breonna Taylor and George Floyd – communities united in outrage lamenting the cries of Black Lives Matter.
I remember the tears briskly falling upon my blouse as I watched Mr. Floyd’s life violently ripped from this Earth. Shortly thereafter, I remember the phone calls, emails, and texts from close friends, acquaintances, and colleagues offering support, listening ears, pledging to learn and endeavoring to understand the struggle for recognition and the fight for human rights. Even so, the deafening support was clouded by the preternatural silence of some medical organizations. Within the Black physician community, outrage was palpable. We reflected upon years of sacrifice and perseverance despite the challenge of bigotry, ignorance, and racism – not only from patients and their families – but also colleagues and administrators. Yet, in our time of horror and need, in those moments of vulnerability ... silence. Eventually, lengthy proclamations of support were expressed through various media. However, it felt too safe, too corporate, and too generic and inauthentic. As a result, an exodus of Black physicians from leadership positions and academic medicine took hold as the blatant continuation of rhetoric – coupled with ineffective outreach and support – finally took its toll.
Frequently, I question how the obstacles of medical school, residency, and beyond are expected to be traversed while living in a world that consistently affords additional challenges to those who look, act, or speak in a manner that varies from the perceived standard. In a culture where the myth of the super doctor reigns, how do we reconcile attainment of a false and detrimental narrative while the overarching pressure acutely felt by Black physicians magnifies in the setting of stereotypes, sociopolitical turbulence, bigotry, and racism? How can one sacrifice for an entity that is unwilling to acknowledge the psychological implications of that sacrifice?
For instance, while in medical school, I transitioned my hair to its natural state but was counseled against doing so because of the risk of losing residency opportunities as a direct result of my “unprofessional” appearance. Throughout residency, multiple incidents come to mind, including frequent demands to see my hospital badge despite the same not being of asked of my White cohorts; denial of entry into physician entrance within the residency building because, despite my professional attire, I was presumed to be a member of the custodial staff; and patients being confused and asking for a doctor despite my long white coat and clear introductions.
Furthermore, the fluency of my speech and the absence of regional dialect or vernacular are quite often lauded by patients. Inquiries to touch my hair as well as hypotheses regarding my nationality or degree of “blackness” with respect to the shape of my nose, eyes, and lips are openly questioned. Unfortunately, those uncomfortable incidents have not been limited to patient encounters.
In one instance, while presenting a patient in the presence of my attending and a 3rd-year medical student, I was sternly admonished for disclosing the race of the patient. I sat still and resolute as this doctor spoke on increased risk of bias in diagnosis and treatment when race is identified. Outwardly, I projected patience but inside, I seethed. In that moment, I realized that I would never have the luxury of ignorance or denial. Although I desire to be valued for my prowess in medicine, the mythical status was not created with my skin color in mind. For is avoidance not but a reflection of denial?
In these chaotic and uncertain times, how can we continue to promote a pathological ideal when the roads traveled are so fundamentally skewed? If a White physician faces a belligerent and argumentative patient, there is opportunity for debriefing both individually and among a larger cohort via classes, conferences, and supervisions. Conversely, when a Black physician is derided with racist sentiment, will they have the same opportunity for reflection and support? Despite identical expectations of professionalism and growth, how can one be successful in a system that either directly or indirectly encourages the opposite?
As we try to shed the super-doctor myth, we must recognize that this unattainable and detrimental persona hinders progress. This myth undermines our ability to understand our fragility, the limitations of our capabilities, and the strength of our vulnerability. We must take an honest look at the manner in which our individual biases and the deeply ingrained (and potentially unconscious) systemic biases are counterintuitive to the success and support of physicians of color.
Dr. Thomas is a board-certified adult psychiatrist with an interest in chronic illness, women’s behavioral health, and minority mental health. She currently practices in North Kingstown and East Providence, R.I. She has no conflicts of interest.
An overwhelmingly loud and high-pitched screech rattles against your hip. You startle and groan into the pillow as your thoughts settle into conscious awareness. It is 3 a.m. You are a 2nd-year resident trudging through the night shift, alerted to the presence of a new patient awaiting an emergency assessment. You are the only in-house physician. Walking steadfastly toward the emergency unit, you enter and greet the patient. Immediately, you observe a look of surprise followed immediately by a scowl.
You extend a hand, but your greeting is abruptly cut short with: “I want to see a doctor!” You pace your breaths to quell annoyance and resume your introduction, asserting that you are a doctor and indeed the only doctor on duty. After moments of deep sighs and questions regarding your credentials, you persuade the patient to start the interview.
It is now 8 a.m. The frustration of the night starts to ease as you prepare to leave. While gathering your things, a visitor is overheard inquiring the whereabouts of a hospital unit. Volunteering as a guide, you walk the person toward the opposite end of the hospital. Bleary eyed, muscle laxed, and bone weary, you point out the entrance, then turn to leave. The steady rhythm of your steps suddenly halts as you hear from behind: “Thank you! You speak English really well!” Blankly, you stare. Your voice remains mute while your brain screams: “What is that supposed to mean?” But you do not utter a sound, because intuitively, you know the answer.
While reading this scenario, what did you feel? Pride in knowing that the physician was able to successfully navigate a busy night? Relief in the physician’s ability to maintain a professional demeanor despite belittling microaggressions? Are you angry? Would you replay those moments like reruns of a bad TV show? Can you imagine entering your home and collapsing onto the bed as your tears of fury pool over your rumpled sheets?
The emotional release of that morning is seared into my memory. Over the years, I questioned my reactions. Was I too passive? Should I have schooled them on their ignorance? Had I done so, would I have incurred reprimands? Would standing up for myself cause years of hard work to fall away? Moreover, had I defended myself, would I forever have been viewed as “The Angry Black Woman?”
This story is more than a vignette. For me, it is another reminder that, despite how far we have come, we have much further to go. As a Black woman in a professional sphere, I stand upon the shoulders of those who sacrificed for a dream, a greater purpose. My foremothers and forefathers fought bravely and tirelessly so that we could attain levels of success that were only once but a dream. Despite this progress, a grimace, carelessly spoken words, or a mindless gesture remind me that, no matter how much I toil and what levels of success I achieve, when I meet someone for the first time or encounter someone from my past, I find myself wondering whether I am remembered for me or because I am “The Black One.”
Honest look at medicine is imperative
It is important to consider multiple facets of the super-doctor myth. We are dedicated, fearless, authoritative, ambitious individuals. We do not yield to sickness, family obligations, or fatigue. Medicine is a calling, and the patient deserves the utmost respect and professional behavior. Impervious to ethnicity, race, nationality, or creed, we are unbiased and always in service of the greater good. Often, however, I wonder how the expectations of patient-focused, patient-centered care can prevail without an honest look at the vicissitudes facing medicine.
We find ourselves amid a tumultuous year overshadowed by a devastating pandemic that skews heavily toward Black and Brown communities, in addition to political turmoil and racial reckoning that sprang forth from fear, anger, and determination ignited by the murders of Breonna Taylor and George Floyd – communities united in outrage lamenting the cries of Black Lives Matter.
I remember the tears briskly falling upon my blouse as I watched Mr. Floyd’s life violently ripped from this Earth. Shortly thereafter, I remember the phone calls, emails, and texts from close friends, acquaintances, and colleagues offering support, listening ears, pledging to learn and endeavoring to understand the struggle for recognition and the fight for human rights. Even so, the deafening support was clouded by the preternatural silence of some medical organizations. Within the Black physician community, outrage was palpable. We reflected upon years of sacrifice and perseverance despite the challenge of bigotry, ignorance, and racism – not only from patients and their families – but also colleagues and administrators. Yet, in our time of horror and need, in those moments of vulnerability ... silence. Eventually, lengthy proclamations of support were expressed through various media. However, it felt too safe, too corporate, and too generic and inauthentic. As a result, an exodus of Black physicians from leadership positions and academic medicine took hold as the blatant continuation of rhetoric – coupled with ineffective outreach and support – finally took its toll.
Frequently, I question how the obstacles of medical school, residency, and beyond are expected to be traversed while living in a world that consistently affords additional challenges to those who look, act, or speak in a manner that varies from the perceived standard. In a culture where the myth of the super doctor reigns, how do we reconcile attainment of a false and detrimental narrative while the overarching pressure acutely felt by Black physicians magnifies in the setting of stereotypes, sociopolitical turbulence, bigotry, and racism? How can one sacrifice for an entity that is unwilling to acknowledge the psychological implications of that sacrifice?
For instance, while in medical school, I transitioned my hair to its natural state but was counseled against doing so because of the risk of losing residency opportunities as a direct result of my “unprofessional” appearance. Throughout residency, multiple incidents come to mind, including frequent demands to see my hospital badge despite the same not being of asked of my White cohorts; denial of entry into physician entrance within the residency building because, despite my professional attire, I was presumed to be a member of the custodial staff; and patients being confused and asking for a doctor despite my long white coat and clear introductions.
Furthermore, the fluency of my speech and the absence of regional dialect or vernacular are quite often lauded by patients. Inquiries to touch my hair as well as hypotheses regarding my nationality or degree of “blackness” with respect to the shape of my nose, eyes, and lips are openly questioned. Unfortunately, those uncomfortable incidents have not been limited to patient encounters.
In one instance, while presenting a patient in the presence of my attending and a 3rd-year medical student, I was sternly admonished for disclosing the race of the patient. I sat still and resolute as this doctor spoke on increased risk of bias in diagnosis and treatment when race is identified. Outwardly, I projected patience but inside, I seethed. In that moment, I realized that I would never have the luxury of ignorance or denial. Although I desire to be valued for my prowess in medicine, the mythical status was not created with my skin color in mind. For is avoidance not but a reflection of denial?
In these chaotic and uncertain times, how can we continue to promote a pathological ideal when the roads traveled are so fundamentally skewed? If a White physician faces a belligerent and argumentative patient, there is opportunity for debriefing both individually and among a larger cohort via classes, conferences, and supervisions. Conversely, when a Black physician is derided with racist sentiment, will they have the same opportunity for reflection and support? Despite identical expectations of professionalism and growth, how can one be successful in a system that either directly or indirectly encourages the opposite?
As we try to shed the super-doctor myth, we must recognize that this unattainable and detrimental persona hinders progress. This myth undermines our ability to understand our fragility, the limitations of our capabilities, and the strength of our vulnerability. We must take an honest look at the manner in which our individual biases and the deeply ingrained (and potentially unconscious) systemic biases are counterintuitive to the success and support of physicians of color.
Dr. Thomas is a board-certified adult psychiatrist with an interest in chronic illness, women’s behavioral health, and minority mental health. She currently practices in North Kingstown and East Providence, R.I. She has no conflicts of interest.
An overwhelmingly loud and high-pitched screech rattles against your hip. You startle and groan into the pillow as your thoughts settle into conscious awareness. It is 3 a.m. You are a 2nd-year resident trudging through the night shift, alerted to the presence of a new patient awaiting an emergency assessment. You are the only in-house physician. Walking steadfastly toward the emergency unit, you enter and greet the patient. Immediately, you observe a look of surprise followed immediately by a scowl.
You extend a hand, but your greeting is abruptly cut short with: “I want to see a doctor!” You pace your breaths to quell annoyance and resume your introduction, asserting that you are a doctor and indeed the only doctor on duty. After moments of deep sighs and questions regarding your credentials, you persuade the patient to start the interview.
It is now 8 a.m. The frustration of the night starts to ease as you prepare to leave. While gathering your things, a visitor is overheard inquiring the whereabouts of a hospital unit. Volunteering as a guide, you walk the person toward the opposite end of the hospital. Bleary eyed, muscle laxed, and bone weary, you point out the entrance, then turn to leave. The steady rhythm of your steps suddenly halts as you hear from behind: “Thank you! You speak English really well!” Blankly, you stare. Your voice remains mute while your brain screams: “What is that supposed to mean?” But you do not utter a sound, because intuitively, you know the answer.
While reading this scenario, what did you feel? Pride in knowing that the physician was able to successfully navigate a busy night? Relief in the physician’s ability to maintain a professional demeanor despite belittling microaggressions? Are you angry? Would you replay those moments like reruns of a bad TV show? Can you imagine entering your home and collapsing onto the bed as your tears of fury pool over your rumpled sheets?
The emotional release of that morning is seared into my memory. Over the years, I questioned my reactions. Was I too passive? Should I have schooled them on their ignorance? Had I done so, would I have incurred reprimands? Would standing up for myself cause years of hard work to fall away? Moreover, had I defended myself, would I forever have been viewed as “The Angry Black Woman?”
This story is more than a vignette. For me, it is another reminder that, despite how far we have come, we have much further to go. As a Black woman in a professional sphere, I stand upon the shoulders of those who sacrificed for a dream, a greater purpose. My foremothers and forefathers fought bravely and tirelessly so that we could attain levels of success that were only once but a dream. Despite this progress, a grimace, carelessly spoken words, or a mindless gesture remind me that, no matter how much I toil and what levels of success I achieve, when I meet someone for the first time or encounter someone from my past, I find myself wondering whether I am remembered for me or because I am “The Black One.”
Honest look at medicine is imperative
It is important to consider multiple facets of the super-doctor myth. We are dedicated, fearless, authoritative, ambitious individuals. We do not yield to sickness, family obligations, or fatigue. Medicine is a calling, and the patient deserves the utmost respect and professional behavior. Impervious to ethnicity, race, nationality, or creed, we are unbiased and always in service of the greater good. Often, however, I wonder how the expectations of patient-focused, patient-centered care can prevail without an honest look at the vicissitudes facing medicine.
We find ourselves amid a tumultuous year overshadowed by a devastating pandemic that skews heavily toward Black and Brown communities, in addition to political turmoil and racial reckoning that sprang forth from fear, anger, and determination ignited by the murders of Breonna Taylor and George Floyd – communities united in outrage lamenting the cries of Black Lives Matter.
I remember the tears briskly falling upon my blouse as I watched Mr. Floyd’s life violently ripped from this Earth. Shortly thereafter, I remember the phone calls, emails, and texts from close friends, acquaintances, and colleagues offering support, listening ears, pledging to learn and endeavoring to understand the struggle for recognition and the fight for human rights. Even so, the deafening support was clouded by the preternatural silence of some medical organizations. Within the Black physician community, outrage was palpable. We reflected upon years of sacrifice and perseverance despite the challenge of bigotry, ignorance, and racism – not only from patients and their families – but also colleagues and administrators. Yet, in our time of horror and need, in those moments of vulnerability ... silence. Eventually, lengthy proclamations of support were expressed through various media. However, it felt too safe, too corporate, and too generic and inauthentic. As a result, an exodus of Black physicians from leadership positions and academic medicine took hold as the blatant continuation of rhetoric – coupled with ineffective outreach and support – finally took its toll.
Frequently, I question how the obstacles of medical school, residency, and beyond are expected to be traversed while living in a world that consistently affords additional challenges to those who look, act, or speak in a manner that varies from the perceived standard. In a culture where the myth of the super doctor reigns, how do we reconcile attainment of a false and detrimental narrative while the overarching pressure acutely felt by Black physicians magnifies in the setting of stereotypes, sociopolitical turbulence, bigotry, and racism? How can one sacrifice for an entity that is unwilling to acknowledge the psychological implications of that sacrifice?
For instance, while in medical school, I transitioned my hair to its natural state but was counseled against doing so because of the risk of losing residency opportunities as a direct result of my “unprofessional” appearance. Throughout residency, multiple incidents come to mind, including frequent demands to see my hospital badge despite the same not being of asked of my White cohorts; denial of entry into physician entrance within the residency building because, despite my professional attire, I was presumed to be a member of the custodial staff; and patients being confused and asking for a doctor despite my long white coat and clear introductions.
Furthermore, the fluency of my speech and the absence of regional dialect or vernacular are quite often lauded by patients. Inquiries to touch my hair as well as hypotheses regarding my nationality or degree of “blackness” with respect to the shape of my nose, eyes, and lips are openly questioned. Unfortunately, those uncomfortable incidents have not been limited to patient encounters.
In one instance, while presenting a patient in the presence of my attending and a 3rd-year medical student, I was sternly admonished for disclosing the race of the patient. I sat still and resolute as this doctor spoke on increased risk of bias in diagnosis and treatment when race is identified. Outwardly, I projected patience but inside, I seethed. In that moment, I realized that I would never have the luxury of ignorance or denial. Although I desire to be valued for my prowess in medicine, the mythical status was not created with my skin color in mind. For is avoidance not but a reflection of denial?
In these chaotic and uncertain times, how can we continue to promote a pathological ideal when the roads traveled are so fundamentally skewed? If a White physician faces a belligerent and argumentative patient, there is opportunity for debriefing both individually and among a larger cohort via classes, conferences, and supervisions. Conversely, when a Black physician is derided with racist sentiment, will they have the same opportunity for reflection and support? Despite identical expectations of professionalism and growth, how can one be successful in a system that either directly or indirectly encourages the opposite?
As we try to shed the super-doctor myth, we must recognize that this unattainable and detrimental persona hinders progress. This myth undermines our ability to understand our fragility, the limitations of our capabilities, and the strength of our vulnerability. We must take an honest look at the manner in which our individual biases and the deeply ingrained (and potentially unconscious) systemic biases are counterintuitive to the success and support of physicians of color.
Dr. Thomas is a board-certified adult psychiatrist with an interest in chronic illness, women’s behavioral health, and minority mental health. She currently practices in North Kingstown and East Providence, R.I. She has no conflicts of interest.
Outstanding medical bills: Dealing with deadbeats
Since the COVID-19 pandemic began, I have received a growing number of inquiries about collection issues. For a variety of reasons, many patients seem increasingly reluctant to pay their medical bills. I’ve written many columns on keeping credit card numbers on file, and other techniques for keeping your accounts receivable in check; but despite your best efforts, there will always be a few deadbeats that you will need to pursue.
For the record, I am not speaking about patients who lost income due to the pandemic and are now struggling with debts, or otherwise have fallen on hard times and are unable to pay.
The worst kinds of deadbeats are the ones who rob you twice; they accept payments from insurance companies and keep them. Such crooks must be pursued aggressively, with all the means at your disposal; but to reiterate the point I’ve tried to drive home repeatedly, the best cure is prevention.
You already know that you should collect as many fees as possible at the time of service. For cosmetic procedures you should require a substantial deposit in advance, with the balance due at the time of service. When that is impossible, maximize the chances you will be paid by making sure all available payment mechanisms are in place.
With my credit-card-on-file system that I’ve described many times, patients who fail to pay their credit card bill are the credit card company’s problem, not yours. In cases where you suspect fees might exceed credit card limits, you can arrange a realistic payment schedule in advance and have the patient fill out a credit application. You can find forms for this online at formswift.com, templates.office.com, and many other websites.
In some cases, it may be worth the trouble to run a background check. There are easy and affordable ways to do this. Dunn & Bradstreet, for example, will furnish a report containing payment records and details of any lawsuits, liens, and other legal actions for a nominal fee. The more financial information you have on file, the more leverage you have if a patient later balks at paying his or her balance.
For cosmetic work, always take before and after photos, and have all patients sign a written consent giving permission for the procedure, assuming full financial responsibility, and acknowledging that no guarantees have been given or implied. This defuses the common deadbeat tactics of claiming ignorance of personal financial obligations and professing dissatisfaction with the results.
Despite all your precautions, a deadbeat will inevitably slip through on occasion; but even then, you have options for extracting payment. Collection agencies are the traditional first line of attack for most medical practices. Ideally, your agency should specialize in handling medical accounts, so it will know exactly how much pressure to exert to avoid charges of harassment. Delinquent accounts should be submitted earlier rather than later to maximize the chances of success; my manager never allows an account to age more than 90 days, and if circumstances dictate, she refers them sooner than that.
When collection agencies fail, think about small claims court. You will need to learn the rules in your state, but in most states there is a small filing fee and a limit of $5,000 or so on claims. No attorneys are involved. If your paperwork is in order, the court will nearly always rule in your favor, but it will not provide the means for actual collection. In other words, you will still have to persuade the deadbeat to pay up. However, in many states a court order will give you the authority to attach a lien to property, or garnish wages, which often provides enough leverage to force payment.
What about those double-deadbeats who keep the insurance checks for themselves? First, check your third-party contract; sometimes the insurance company or HMO will be compelled to pay you directly and then go after the patient to get back its money. (They won’t volunteer this service, however – you’ll have to ask for it.)
If that’s not an option, consider reporting the misdirected payment to the Internal Revenue Service as income to the patient, by submitting a 1099 Miscellaneous Income form. Be sure to notify the deadbeat that you will be doing this. Sometimes the threat of such action will convince the individual to pay up; if not, at least you’ll have the satisfaction of knowing he or she will have to pay taxes on the money.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Since the COVID-19 pandemic began, I have received a growing number of inquiries about collection issues. For a variety of reasons, many patients seem increasingly reluctant to pay their medical bills. I’ve written many columns on keeping credit card numbers on file, and other techniques for keeping your accounts receivable in check; but despite your best efforts, there will always be a few deadbeats that you will need to pursue.
For the record, I am not speaking about patients who lost income due to the pandemic and are now struggling with debts, or otherwise have fallen on hard times and are unable to pay.
The worst kinds of deadbeats are the ones who rob you twice; they accept payments from insurance companies and keep them. Such crooks must be pursued aggressively, with all the means at your disposal; but to reiterate the point I’ve tried to drive home repeatedly, the best cure is prevention.
You already know that you should collect as many fees as possible at the time of service. For cosmetic procedures you should require a substantial deposit in advance, with the balance due at the time of service. When that is impossible, maximize the chances you will be paid by making sure all available payment mechanisms are in place.
With my credit-card-on-file system that I’ve described many times, patients who fail to pay their credit card bill are the credit card company’s problem, not yours. In cases where you suspect fees might exceed credit card limits, you can arrange a realistic payment schedule in advance and have the patient fill out a credit application. You can find forms for this online at formswift.com, templates.office.com, and many other websites.
In some cases, it may be worth the trouble to run a background check. There are easy and affordable ways to do this. Dunn & Bradstreet, for example, will furnish a report containing payment records and details of any lawsuits, liens, and other legal actions for a nominal fee. The more financial information you have on file, the more leverage you have if a patient later balks at paying his or her balance.
For cosmetic work, always take before and after photos, and have all patients sign a written consent giving permission for the procedure, assuming full financial responsibility, and acknowledging that no guarantees have been given or implied. This defuses the common deadbeat tactics of claiming ignorance of personal financial obligations and professing dissatisfaction with the results.
Despite all your precautions, a deadbeat will inevitably slip through on occasion; but even then, you have options for extracting payment. Collection agencies are the traditional first line of attack for most medical practices. Ideally, your agency should specialize in handling medical accounts, so it will know exactly how much pressure to exert to avoid charges of harassment. Delinquent accounts should be submitted earlier rather than later to maximize the chances of success; my manager never allows an account to age more than 90 days, and if circumstances dictate, she refers them sooner than that.
When collection agencies fail, think about small claims court. You will need to learn the rules in your state, but in most states there is a small filing fee and a limit of $5,000 or so on claims. No attorneys are involved. If your paperwork is in order, the court will nearly always rule in your favor, but it will not provide the means for actual collection. In other words, you will still have to persuade the deadbeat to pay up. However, in many states a court order will give you the authority to attach a lien to property, or garnish wages, which often provides enough leverage to force payment.
What about those double-deadbeats who keep the insurance checks for themselves? First, check your third-party contract; sometimes the insurance company or HMO will be compelled to pay you directly and then go after the patient to get back its money. (They won’t volunteer this service, however – you’ll have to ask for it.)
If that’s not an option, consider reporting the misdirected payment to the Internal Revenue Service as income to the patient, by submitting a 1099 Miscellaneous Income form. Be sure to notify the deadbeat that you will be doing this. Sometimes the threat of such action will convince the individual to pay up; if not, at least you’ll have the satisfaction of knowing he or she will have to pay taxes on the money.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Since the COVID-19 pandemic began, I have received a growing number of inquiries about collection issues. For a variety of reasons, many patients seem increasingly reluctant to pay their medical bills. I’ve written many columns on keeping credit card numbers on file, and other techniques for keeping your accounts receivable in check; but despite your best efforts, there will always be a few deadbeats that you will need to pursue.
For the record, I am not speaking about patients who lost income due to the pandemic and are now struggling with debts, or otherwise have fallen on hard times and are unable to pay.
The worst kinds of deadbeats are the ones who rob you twice; they accept payments from insurance companies and keep them. Such crooks must be pursued aggressively, with all the means at your disposal; but to reiterate the point I’ve tried to drive home repeatedly, the best cure is prevention.
You already know that you should collect as many fees as possible at the time of service. For cosmetic procedures you should require a substantial deposit in advance, with the balance due at the time of service. When that is impossible, maximize the chances you will be paid by making sure all available payment mechanisms are in place.
With my credit-card-on-file system that I’ve described many times, patients who fail to pay their credit card bill are the credit card company’s problem, not yours. In cases where you suspect fees might exceed credit card limits, you can arrange a realistic payment schedule in advance and have the patient fill out a credit application. You can find forms for this online at formswift.com, templates.office.com, and many other websites.
In some cases, it may be worth the trouble to run a background check. There are easy and affordable ways to do this. Dunn & Bradstreet, for example, will furnish a report containing payment records and details of any lawsuits, liens, and other legal actions for a nominal fee. The more financial information you have on file, the more leverage you have if a patient later balks at paying his or her balance.
For cosmetic work, always take before and after photos, and have all patients sign a written consent giving permission for the procedure, assuming full financial responsibility, and acknowledging that no guarantees have been given or implied. This defuses the common deadbeat tactics of claiming ignorance of personal financial obligations and professing dissatisfaction with the results.
Despite all your precautions, a deadbeat will inevitably slip through on occasion; but even then, you have options for extracting payment. Collection agencies are the traditional first line of attack for most medical practices. Ideally, your agency should specialize in handling medical accounts, so it will know exactly how much pressure to exert to avoid charges of harassment. Delinquent accounts should be submitted earlier rather than later to maximize the chances of success; my manager never allows an account to age more than 90 days, and if circumstances dictate, she refers them sooner than that.
When collection agencies fail, think about small claims court. You will need to learn the rules in your state, but in most states there is a small filing fee and a limit of $5,000 or so on claims. No attorneys are involved. If your paperwork is in order, the court will nearly always rule in your favor, but it will not provide the means for actual collection. In other words, you will still have to persuade the deadbeat to pay up. However, in many states a court order will give you the authority to attach a lien to property, or garnish wages, which often provides enough leverage to force payment.
What about those double-deadbeats who keep the insurance checks for themselves? First, check your third-party contract; sometimes the insurance company or HMO will be compelled to pay you directly and then go after the patient to get back its money. (They won’t volunteer this service, however – you’ll have to ask for it.)
If that’s not an option, consider reporting the misdirected payment to the Internal Revenue Service as income to the patient, by submitting a 1099 Miscellaneous Income form. Be sure to notify the deadbeat that you will be doing this. Sometimes the threat of such action will convince the individual to pay up; if not, at least you’ll have the satisfaction of knowing he or she will have to pay taxes on the money.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
A 35-year-old with erythematous, dusky patches on both lower extremities
Zinc deficiency may be inherited or acquired. Acrodermatitis enteropathica is an autosomal recessive genetic disorder caused by a mutation in the gene that encodes a zinc transporter. It presents in infancy with the classic triad of diarrhea, dermatitis, and alopecia. Acquired zinc deficiency is due to causes such as alcoholism, malabsorption disorders like cystic fibrosis, inflammatory disease, gastrointestinal surgery, metabolic stress following general surgery, eating disorders, infections, malignancy, or occasionally in pregnancy. Classically, the face, groin, and extremities are affected (often acral), with erythematous, scaly patches. Pustules and bullae may be present. Angular cheilitis is often seen.
Necrolytic migratory erythema, or glucagonoma syndrome, is a very rare syndrome that presents as annular, erythematous patches with blisters that erode on the lower extremities and groin. The condition results from a cancerous tumor in the alpha cells of the pancreas called a glucagonoma, which secretes the hormone glucagon. It is often associated with diabetes and hyperglycemia.
Necrolytic acral erythema resembles acrodermatitis enteropathica and necrolytic migratory erythema clinically, however, it is associated with hepatitis C infection. Lesions are plaques with well defined borders distributed acrally. Treatment of the hepatitis C often improves the dermatitis.
Our patient’s blood work was consistent with nutritional deficiency and revealed low levels of zinc, vitamin A, ceruloplasmin, albumin and prealbumin, total protein, calcium, selenium, vitamin E, vitamin K, and vitamin C. Her hemoglobin A1C was under 4. Her hepatitis serologies were negative. The patient received total parenteral nutrition with subsequent complete resolution of her rash. Follow up for gastric bypass patients should be performed long term as they are at risk for nutritional deficiencies.
Dr. Bilu Martin, and Andrew Harris, DO, Mount Sinai Medical Center, Aventura, Fla., provided the case and photos.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Dermatol Online J. 2016 Nov 15; 22(11):13030.
Andrews’ Disease of the Skin: Clinical Dermatology. Philadelphia: Saunders Elsevier, 2006.
Bolognia et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
Zinc deficiency may be inherited or acquired. Acrodermatitis enteropathica is an autosomal recessive genetic disorder caused by a mutation in the gene that encodes a zinc transporter. It presents in infancy with the classic triad of diarrhea, dermatitis, and alopecia. Acquired zinc deficiency is due to causes such as alcoholism, malabsorption disorders like cystic fibrosis, inflammatory disease, gastrointestinal surgery, metabolic stress following general surgery, eating disorders, infections, malignancy, or occasionally in pregnancy. Classically, the face, groin, and extremities are affected (often acral), with erythematous, scaly patches. Pustules and bullae may be present. Angular cheilitis is often seen.
Necrolytic migratory erythema, or glucagonoma syndrome, is a very rare syndrome that presents as annular, erythematous patches with blisters that erode on the lower extremities and groin. The condition results from a cancerous tumor in the alpha cells of the pancreas called a glucagonoma, which secretes the hormone glucagon. It is often associated with diabetes and hyperglycemia.
Necrolytic acral erythema resembles acrodermatitis enteropathica and necrolytic migratory erythema clinically, however, it is associated with hepatitis C infection. Lesions are plaques with well defined borders distributed acrally. Treatment of the hepatitis C often improves the dermatitis.
Our patient’s blood work was consistent with nutritional deficiency and revealed low levels of zinc, vitamin A, ceruloplasmin, albumin and prealbumin, total protein, calcium, selenium, vitamin E, vitamin K, and vitamin C. Her hemoglobin A1C was under 4. Her hepatitis serologies were negative. The patient received total parenteral nutrition with subsequent complete resolution of her rash. Follow up for gastric bypass patients should be performed long term as they are at risk for nutritional deficiencies.
Dr. Bilu Martin, and Andrew Harris, DO, Mount Sinai Medical Center, Aventura, Fla., provided the case and photos.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Dermatol Online J. 2016 Nov 15; 22(11):13030.
Andrews’ Disease of the Skin: Clinical Dermatology. Philadelphia: Saunders Elsevier, 2006.
Bolognia et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
Zinc deficiency may be inherited or acquired. Acrodermatitis enteropathica is an autosomal recessive genetic disorder caused by a mutation in the gene that encodes a zinc transporter. It presents in infancy with the classic triad of diarrhea, dermatitis, and alopecia. Acquired zinc deficiency is due to causes such as alcoholism, malabsorption disorders like cystic fibrosis, inflammatory disease, gastrointestinal surgery, metabolic stress following general surgery, eating disorders, infections, malignancy, or occasionally in pregnancy. Classically, the face, groin, and extremities are affected (often acral), with erythematous, scaly patches. Pustules and bullae may be present. Angular cheilitis is often seen.
Necrolytic migratory erythema, or glucagonoma syndrome, is a very rare syndrome that presents as annular, erythematous patches with blisters that erode on the lower extremities and groin. The condition results from a cancerous tumor in the alpha cells of the pancreas called a glucagonoma, which secretes the hormone glucagon. It is often associated with diabetes and hyperglycemia.
Necrolytic acral erythema resembles acrodermatitis enteropathica and necrolytic migratory erythema clinically, however, it is associated with hepatitis C infection. Lesions are plaques with well defined borders distributed acrally. Treatment of the hepatitis C often improves the dermatitis.
Our patient’s blood work was consistent with nutritional deficiency and revealed low levels of zinc, vitamin A, ceruloplasmin, albumin and prealbumin, total protein, calcium, selenium, vitamin E, vitamin K, and vitamin C. Her hemoglobin A1C was under 4. Her hepatitis serologies were negative. The patient received total parenteral nutrition with subsequent complete resolution of her rash. Follow up for gastric bypass patients should be performed long term as they are at risk for nutritional deficiencies.
Dr. Bilu Martin, and Andrew Harris, DO, Mount Sinai Medical Center, Aventura, Fla., provided the case and photos.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Dermatol Online J. 2016 Nov 15; 22(11):13030.
Andrews’ Disease of the Skin: Clinical Dermatology. Philadelphia: Saunders Elsevier, 2006.
Bolognia et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
Western diet promoted skin, joint inflammation in preclinical study
A short-term Western diet facilitated the development of interleukin (IL)-23-mediated psoriasis-like skin and joint inflammation and caused shifts in the intestinal microbiota in a murine model – , say the investigators and other experts who reviewed the findings.
The mice did not become obese during the short duration of the multilayered study, which suggests that a Western diet (high sugar, moderate fat) can be impactful independent of obesity, Samuel T. Hwang, MD, PhD, professor and chair of dermatology at the University of California, Davis, and senior author of the study, said in an interview. The study was published in the Journal of Investigative Dermatology.
In an accompanying commentary, Renuka R. Nayak, MD, PhD, of the department of rheumatology at the University of California, San Francisco, wrote that the findings “add to the mounting evidence suggesting that diet has a prominent role in the treatment of psoriasis and [psoriatic arthritis] and raise the possibility that the microbiome may contribute to disease severity”.
Mice were fed a Western diet (WD) or conventional chow diet for 6 weeks and then injected with IL-23 minicircle (MC) DNA to induce systemic IL-23 overexpression – or a control minicircle DNA injection – and continued on these diets for another 4 weeks.
The mice in the WD/IL-23 MC DNA group developed erythema and scaling and increased epidermal thickness in the ears; such changes were “remarkably milder” or nonexistent in the other groups. Skin and joint immune cell populations, such as gamma delta T cells, neutrophils, and T helper type 17 cytokines were elevated in WD-fed mice, as were other markers of IL-23-mediated joint inflammation.
Recent research has suggested that the gut microbiota is dysbiotic in patients with psoriasis, and this new study found that WD-fed mice had less microbial diversity than that of mice fed a conventional diet. After IL-23 MC delivery, WD-fed reduced microbial diversity and pronounced dysbiosis.
“When we combined the Western diet and IL-23, we saw some very different microbes in abundance. The whole landscape changed,” Dr. Hwang said in the interview.
The data “suggest that WD and overexpression of IL-23 may contribute to gut microbiota dysbiosis in a synergistic and complex manner,” he and his coinvestigators wrote.
Treatment with broad-spectrum antibiotics suppressed IL-23-mediated skin and joint inflammation in the WD-fed mice – and moderately affected skin inflammation in conventionally-fed mice as well – which affirmed the role of dysbiosis.
And “notably,” in another layer of the study, mice that switched diets from a WD to a conventional diet had reduced skin and joint inflammation and increased diversity of gut microbiota. (Mice that were fed a WD for 6 weeks and given the IL-23 MC DNA were randomized to continue this diet for another 4 weeks or switch to a conventional diet.)
Commenting on the new research, Wilson Liao, MD, professor and vice chair of research in the department of dermatology at the University of California, San Francisco, said it “provides evidence” that diet can affect not only psoriasis, but psoriatic arthritis (PsA) as well, “through altering the ratio of good to bad bacteria in the gut.”
Going forward, better understanding “which specific gut bacteria and bacterial products lead to increased psoriatic inflammation, and the immunologic mechanism by which this occurs” will be important and could lead to novel treatments for psoriasis and PsA, said Dr. Liao, director of the UCSF Psoriasis and Skin Treatment Center.
Next on his research agenda, Dr. Hwang said, is the question of “how microbiota in the gut are actually able to influence inflammation at very distant sites in the joints and the skin.
“We want to understand the metabolic mechanisms,” he said, noting that “we invariably talk about cytokines, but there are other substances, like certain bile acids that are metabolized through the gut microbiome,” which may play a role.
The findings also offer a basis for treatment experiments in humans – of diet, probiotic therapy, or selective antibiotic modulation, for instance, Dr. Hwang said.
And in the meantime, the findings should encourage patients who are interested in making dietary changes, such as reducing sugar intake. “There’s wide interest – patients will ask, is there something I can change to make this better?” Dr. Hwang said. “Before, we could say it might be logical, but now we have some evidence. The message now is [high-sugar, moderate-fat] diets, apart from their ability to stimulate obesity, probably have some effects.”
Dietary change may not replace the need for other psoriasis treatments, he said, “but I think there’s good reason to believe that if you do change your diet, your treatment will be better than it would be without that dietary change,” he said.
In their discussion, Dr. Hwang and coauthors note that WD with IL-23 overexpression also decreased the mRNA expression of barrier-forming tight junction proteins, thus increasing intestinal permeability. This finding may be relevant, they wrote, because “leaky gut has been proposed as a pathogenic link between unhealthy diet, gut dysbiosis, and enhanced immune response,” and has been observed in a number of autoimmune diseases, including psoriasis.
Dr. Hwang, lead author Zhenrui Shi, MD, PhD, and coauthors reported no conflicts of interest. Their study was supported by the National Psoriasis Foundation, as well as the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the National Cancer Institute.
A short-term Western diet facilitated the development of interleukin (IL)-23-mediated psoriasis-like skin and joint inflammation and caused shifts in the intestinal microbiota in a murine model – , say the investigators and other experts who reviewed the findings.
The mice did not become obese during the short duration of the multilayered study, which suggests that a Western diet (high sugar, moderate fat) can be impactful independent of obesity, Samuel T. Hwang, MD, PhD, professor and chair of dermatology at the University of California, Davis, and senior author of the study, said in an interview. The study was published in the Journal of Investigative Dermatology.
In an accompanying commentary, Renuka R. Nayak, MD, PhD, of the department of rheumatology at the University of California, San Francisco, wrote that the findings “add to the mounting evidence suggesting that diet has a prominent role in the treatment of psoriasis and [psoriatic arthritis] and raise the possibility that the microbiome may contribute to disease severity”.
Mice were fed a Western diet (WD) or conventional chow diet for 6 weeks and then injected with IL-23 minicircle (MC) DNA to induce systemic IL-23 overexpression – or a control minicircle DNA injection – and continued on these diets for another 4 weeks.
The mice in the WD/IL-23 MC DNA group developed erythema and scaling and increased epidermal thickness in the ears; such changes were “remarkably milder” or nonexistent in the other groups. Skin and joint immune cell populations, such as gamma delta T cells, neutrophils, and T helper type 17 cytokines were elevated in WD-fed mice, as were other markers of IL-23-mediated joint inflammation.
Recent research has suggested that the gut microbiota is dysbiotic in patients with psoriasis, and this new study found that WD-fed mice had less microbial diversity than that of mice fed a conventional diet. After IL-23 MC delivery, WD-fed reduced microbial diversity and pronounced dysbiosis.
“When we combined the Western diet and IL-23, we saw some very different microbes in abundance. The whole landscape changed,” Dr. Hwang said in the interview.
The data “suggest that WD and overexpression of IL-23 may contribute to gut microbiota dysbiosis in a synergistic and complex manner,” he and his coinvestigators wrote.
Treatment with broad-spectrum antibiotics suppressed IL-23-mediated skin and joint inflammation in the WD-fed mice – and moderately affected skin inflammation in conventionally-fed mice as well – which affirmed the role of dysbiosis.
And “notably,” in another layer of the study, mice that switched diets from a WD to a conventional diet had reduced skin and joint inflammation and increased diversity of gut microbiota. (Mice that were fed a WD for 6 weeks and given the IL-23 MC DNA were randomized to continue this diet for another 4 weeks or switch to a conventional diet.)
Commenting on the new research, Wilson Liao, MD, professor and vice chair of research in the department of dermatology at the University of California, San Francisco, said it “provides evidence” that diet can affect not only psoriasis, but psoriatic arthritis (PsA) as well, “through altering the ratio of good to bad bacteria in the gut.”
Going forward, better understanding “which specific gut bacteria and bacterial products lead to increased psoriatic inflammation, and the immunologic mechanism by which this occurs” will be important and could lead to novel treatments for psoriasis and PsA, said Dr. Liao, director of the UCSF Psoriasis and Skin Treatment Center.
Next on his research agenda, Dr. Hwang said, is the question of “how microbiota in the gut are actually able to influence inflammation at very distant sites in the joints and the skin.
“We want to understand the metabolic mechanisms,” he said, noting that “we invariably talk about cytokines, but there are other substances, like certain bile acids that are metabolized through the gut microbiome,” which may play a role.
The findings also offer a basis for treatment experiments in humans – of diet, probiotic therapy, or selective antibiotic modulation, for instance, Dr. Hwang said.
And in the meantime, the findings should encourage patients who are interested in making dietary changes, such as reducing sugar intake. “There’s wide interest – patients will ask, is there something I can change to make this better?” Dr. Hwang said. “Before, we could say it might be logical, but now we have some evidence. The message now is [high-sugar, moderate-fat] diets, apart from their ability to stimulate obesity, probably have some effects.”
Dietary change may not replace the need for other psoriasis treatments, he said, “but I think there’s good reason to believe that if you do change your diet, your treatment will be better than it would be without that dietary change,” he said.
In their discussion, Dr. Hwang and coauthors note that WD with IL-23 overexpression also decreased the mRNA expression of barrier-forming tight junction proteins, thus increasing intestinal permeability. This finding may be relevant, they wrote, because “leaky gut has been proposed as a pathogenic link between unhealthy diet, gut dysbiosis, and enhanced immune response,” and has been observed in a number of autoimmune diseases, including psoriasis.
Dr. Hwang, lead author Zhenrui Shi, MD, PhD, and coauthors reported no conflicts of interest. Their study was supported by the National Psoriasis Foundation, as well as the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the National Cancer Institute.
A short-term Western diet facilitated the development of interleukin (IL)-23-mediated psoriasis-like skin and joint inflammation and caused shifts in the intestinal microbiota in a murine model – , say the investigators and other experts who reviewed the findings.
The mice did not become obese during the short duration of the multilayered study, which suggests that a Western diet (high sugar, moderate fat) can be impactful independent of obesity, Samuel T. Hwang, MD, PhD, professor and chair of dermatology at the University of California, Davis, and senior author of the study, said in an interview. The study was published in the Journal of Investigative Dermatology.
In an accompanying commentary, Renuka R. Nayak, MD, PhD, of the department of rheumatology at the University of California, San Francisco, wrote that the findings “add to the mounting evidence suggesting that diet has a prominent role in the treatment of psoriasis and [psoriatic arthritis] and raise the possibility that the microbiome may contribute to disease severity”.
Mice were fed a Western diet (WD) or conventional chow diet for 6 weeks and then injected with IL-23 minicircle (MC) DNA to induce systemic IL-23 overexpression – or a control minicircle DNA injection – and continued on these diets for another 4 weeks.
The mice in the WD/IL-23 MC DNA group developed erythema and scaling and increased epidermal thickness in the ears; such changes were “remarkably milder” or nonexistent in the other groups. Skin and joint immune cell populations, such as gamma delta T cells, neutrophils, and T helper type 17 cytokines were elevated in WD-fed mice, as were other markers of IL-23-mediated joint inflammation.
Recent research has suggested that the gut microbiota is dysbiotic in patients with psoriasis, and this new study found that WD-fed mice had less microbial diversity than that of mice fed a conventional diet. After IL-23 MC delivery, WD-fed reduced microbial diversity and pronounced dysbiosis.
“When we combined the Western diet and IL-23, we saw some very different microbes in abundance. The whole landscape changed,” Dr. Hwang said in the interview.
The data “suggest that WD and overexpression of IL-23 may contribute to gut microbiota dysbiosis in a synergistic and complex manner,” he and his coinvestigators wrote.
Treatment with broad-spectrum antibiotics suppressed IL-23-mediated skin and joint inflammation in the WD-fed mice – and moderately affected skin inflammation in conventionally-fed mice as well – which affirmed the role of dysbiosis.
And “notably,” in another layer of the study, mice that switched diets from a WD to a conventional diet had reduced skin and joint inflammation and increased diversity of gut microbiota. (Mice that were fed a WD for 6 weeks and given the IL-23 MC DNA were randomized to continue this diet for another 4 weeks or switch to a conventional diet.)
Commenting on the new research, Wilson Liao, MD, professor and vice chair of research in the department of dermatology at the University of California, San Francisco, said it “provides evidence” that diet can affect not only psoriasis, but psoriatic arthritis (PsA) as well, “through altering the ratio of good to bad bacteria in the gut.”
Going forward, better understanding “which specific gut bacteria and bacterial products lead to increased psoriatic inflammation, and the immunologic mechanism by which this occurs” will be important and could lead to novel treatments for psoriasis and PsA, said Dr. Liao, director of the UCSF Psoriasis and Skin Treatment Center.
Next on his research agenda, Dr. Hwang said, is the question of “how microbiota in the gut are actually able to influence inflammation at very distant sites in the joints and the skin.
“We want to understand the metabolic mechanisms,” he said, noting that “we invariably talk about cytokines, but there are other substances, like certain bile acids that are metabolized through the gut microbiome,” which may play a role.
The findings also offer a basis for treatment experiments in humans – of diet, probiotic therapy, or selective antibiotic modulation, for instance, Dr. Hwang said.
And in the meantime, the findings should encourage patients who are interested in making dietary changes, such as reducing sugar intake. “There’s wide interest – patients will ask, is there something I can change to make this better?” Dr. Hwang said. “Before, we could say it might be logical, but now we have some evidence. The message now is [high-sugar, moderate-fat] diets, apart from their ability to stimulate obesity, probably have some effects.”
Dietary change may not replace the need for other psoriasis treatments, he said, “but I think there’s good reason to believe that if you do change your diet, your treatment will be better than it would be without that dietary change,” he said.
In their discussion, Dr. Hwang and coauthors note that WD with IL-23 overexpression also decreased the mRNA expression of barrier-forming tight junction proteins, thus increasing intestinal permeability. This finding may be relevant, they wrote, because “leaky gut has been proposed as a pathogenic link between unhealthy diet, gut dysbiosis, and enhanced immune response,” and has been observed in a number of autoimmune diseases, including psoriasis.
Dr. Hwang, lead author Zhenrui Shi, MD, PhD, and coauthors reported no conflicts of interest. Their study was supported by the National Psoriasis Foundation, as well as the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the National Cancer Institute.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
CDC officially endorses third dose of mRNA vaccines for immunocompromised
Centers for Disease Control and Prevention Director Rochelle Walensky, MD, has officially signed off on a recommendation by an independent panel of 11 experts to allow people with weakened immune function to get a third dose of certain COVID-19 vaccines.
The decision follows a unanimous vote by the CDC’s Advisory Committee on Immunization Practices (ACIP), which in turn came hours after the U.S. Food and Drug Administration updated its Emergency Use Authorization (EUA) for the Pfizer and Moderna mRNA vaccines.
About 7 million adults in the United States have moderately to severely impaired immune function because of a medical condition they live with or a medication they take to manage a health condition.
People who fall into this category are at higher risk of being hospitalized or dying if they get COVID-19. They are also more likely to transmit the infection. About 40% of vaccinated patients who are hospitalized with breakthrough cases are immunocompromised.
Recent studies have shown that between one-third and one-half of immunocompromised people who didn’t develop antibodies after two doses of a vaccine do get some level of protection after a third dose.
Even then, however, the protection immunocompromised people get from vaccines is not as robust as someone who has healthy immune function, and some panel members were concerned that a third dose might come with a false sense of security.
“My only concern with adding a third dose for the immunocompromised is the impression that our immunocompromised population [will] then be safe,” said ACIP member Helen Talbot, MD, MPH, an associate professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn.
“I think the reality is they’ll be safer but still at incredibly high risk for severe disease and death,” she said.
In updating its EUA, the FDA stressed that, even after a third dose, people who are immunocompromised will still need to wear a mask indoors, socially distance, and avoid large crowds. In addition, family members and other close contacts should be fully vaccinated to protect these vulnerable individuals.
Johnson & Johnson not in the mix
The boosters will be available to children as young as 12 years of age who’ve had a Pfizer vaccine or those ages 18 and older who’ve gotten the Moderna vaccine.
For now, people who’ve had the one-dose Johnson & Johnson vaccine have not been cleared to get a second dose of any vaccine.
FDA experts acknowledged the gap but said that people who had received the Johnson & Johnson vaccine represented a small slice of vaccinated Americans, and said they couldn’t act before the FDA had updated its authorization for that vaccine, which the agency is actively exploring.
“We had to do what we’re doing based on the data we have in hand,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA, the division of the agency that regulates vaccines.
“We think at least there is a solution here for the very large majority of immunocompromised individuals, and we believe we will probably have a solution for the remainder in the not-too-distant future,” Dr. Marks said.
In its updated EUA, the FDA said that the third shots were intended for people who had undergone solid organ transplants or have an “equivalent level of immunocompromise.”
The details
Clinical experts on the CDC panel spent a good deal of time trying to suss out exactly what conditions might fall under the FDA’s umbrella for a third dose.
In a presentation to the committee, Neela Goswami, MD, PhD, an assistant professor of infectious diseases at Emory University School of Medicine and of epidemiology at the Emory Rollins School of Public Health, Atlanta, stressed that the shots are intended for patients who are moderately or severely immunocompromised, in close consultation with their doctors, but that people who should qualify would include those:
- Receiving treatment for solid tumors or blood cancers
- Taking immunosuppressing medications after a solid organ transplant
- Within 2 years of receiving CAR-T therapy or a stem cell transplant
- Who have primary immunodeficiencies – rare genetic disorders that prevent the immune system from working properly
- With advanced or untreated
- Taking high-dose corticosteroids (more than 20 milligrams of or its equivalent daily), alkylating agents, antimetabolites, chemotherapy, TNF blockers, or other immunomodulating or immunosuppressing biologics
- With certain chronic medical conditions, such as or asplenia – living without a spleen
- Receiving dialysis
In discussion, CDC experts clarified that these third doses were not intended for people whose immune function had waned with age, such as elderly residents of long-term care facilities or people with chronic diseases like diabetes.
The idea is to try to get a third dose of the vaccine they’ve already had – Moderna or Pfizer – but if that’s not feasible, it’s fine for the third dose to be different from what someone has had before. The third dose should be given at least 28 days after a second dose, and, ideally, before the initiation of immunosuppressive therapy.
Participants in the meeting said that the CDC would post updated materials on its website to help guide physicians on exactly who should receive third doses.
Ultimately, however, the extra doses will be given on an honor system; no prescriptions or other kinds of clinical documentation will be required for people to get a third dose of these shots.
Tests to measure neutralizing antibodies are also not recommended before the shots are given because of differences in the types of tests used to measure these antibodies and the difficulty in interpreting them. It’s unclear right now what level of neutralizing antibodies is needed for protection.
‘Peace of mind’
In public testimony, Heather Braaten, a 44-year-old being treated for ovarian cancer, said she was grateful to have gotten two shots of the Pfizer vaccine last winter, in between rounds of chemotherapy, but she knew she was probably not well protected. She said she’d become obsessive over the past few months reading medical studies and trying to understand her risk.
“I have felt distraught over the situation. My prognosis is poor. I most likely have about two to three years left to live, so everything counts,” Ms. Braaten said.
She said her life ambitions were humble. She wants to visit with friends and family and not have to worry that she’ll be a breakthrough case. She wants to go grocery shopping again and “not panic and leave the store after five minutes.” She’d love to feel free to travel, she said.
“While I understand I still need to be cautious, I am hopeful for the peace of mind and greater freedom a third shot can provide,” Ms. Braaten said.
More boosters on the way?
In the second half of the meeting, the CDC also signaled that it was considering the use of boosters for people whose immunity might have waned in the months since they had completed their vaccine series, particularly seniors. About 75% of people hospitalized with vaccine breakthrough cases are over age 65, according to CDC data.
Those considerations are becoming more urgent as the Delta variant continues to pummel less vaccinated states and counties.
In its presentation to the ACIP, Heather Scobie, PhD, MPH, a member of the CDC’s COVID Response Team, highlighted data from Canada, Israel, Qatar, and the United Kingdom showing that, while the Pfizer vaccine was still highly effective at preventing hospitalizations and death, it’s far less likely when faced with Delta to prevent an infection that causes symptoms.
In Israel, Pfizer’s vaccine prevented symptoms an average of 41% of the time. In Qatar, which is also using the Moderna vaccine, Pfizer’s prevented symptomatic infections with Delta about 54% of the time compared with 85% with Moderna’s.
Dr. Scobie noted that Pfizer’s waning efficacy may have something to do with the fact that it uses a lower dosage than Moderna’s. Pfizer’s recommended dosing interval is also shorter – 3 weeks compared with 4 weeks for Moderna’s. Stretching the time between shots has been shown to boost vaccine effectiveness, she said.
New data from the Mayo clinic, published ahead of peer review, also suggest that Pfizer’s protection may be fading more quickly than Moderna’s.
In February, both shots were nearly 100% effective at preventing the SARS-CoV-2 infection, but by July, against Delta, Pfizer’s efficacy had dropped to somewhere between 13% and 62%, while Moderna’s was still effective at preventing infection between 58% and 87% of the time.
In July, Pfizer’s was between 24% and 94% effective at preventing hospitalization with a COVID-19 infection and Moderna’s was between 33% and 96% effective at preventing hospitalization.
While that may sound like cause for concern, Dr. Scobie noted that, as of August 2, severe COVD-19 outcomes after vaccination are still very rare. Among 164 million fully vaccinated people in the United States there have been about 7,000 hospitalizations and 1,500 deaths; nearly three out of four of these have been in people over the age of 65.
The ACIP will next meet on August 24 to focus solely on the COVID-19 vaccines.
A version of this article first appeared on Medscape.com.
Centers for Disease Control and Prevention Director Rochelle Walensky, MD, has officially signed off on a recommendation by an independent panel of 11 experts to allow people with weakened immune function to get a third dose of certain COVID-19 vaccines.
The decision follows a unanimous vote by the CDC’s Advisory Committee on Immunization Practices (ACIP), which in turn came hours after the U.S. Food and Drug Administration updated its Emergency Use Authorization (EUA) for the Pfizer and Moderna mRNA vaccines.
About 7 million adults in the United States have moderately to severely impaired immune function because of a medical condition they live with or a medication they take to manage a health condition.
People who fall into this category are at higher risk of being hospitalized or dying if they get COVID-19. They are also more likely to transmit the infection. About 40% of vaccinated patients who are hospitalized with breakthrough cases are immunocompromised.
Recent studies have shown that between one-third and one-half of immunocompromised people who didn’t develop antibodies after two doses of a vaccine do get some level of protection after a third dose.
Even then, however, the protection immunocompromised people get from vaccines is not as robust as someone who has healthy immune function, and some panel members were concerned that a third dose might come with a false sense of security.
“My only concern with adding a third dose for the immunocompromised is the impression that our immunocompromised population [will] then be safe,” said ACIP member Helen Talbot, MD, MPH, an associate professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn.
“I think the reality is they’ll be safer but still at incredibly high risk for severe disease and death,” she said.
In updating its EUA, the FDA stressed that, even after a third dose, people who are immunocompromised will still need to wear a mask indoors, socially distance, and avoid large crowds. In addition, family members and other close contacts should be fully vaccinated to protect these vulnerable individuals.
Johnson & Johnson not in the mix
The boosters will be available to children as young as 12 years of age who’ve had a Pfizer vaccine or those ages 18 and older who’ve gotten the Moderna vaccine.
For now, people who’ve had the one-dose Johnson & Johnson vaccine have not been cleared to get a second dose of any vaccine.
FDA experts acknowledged the gap but said that people who had received the Johnson & Johnson vaccine represented a small slice of vaccinated Americans, and said they couldn’t act before the FDA had updated its authorization for that vaccine, which the agency is actively exploring.
“We had to do what we’re doing based on the data we have in hand,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA, the division of the agency that regulates vaccines.
“We think at least there is a solution here for the very large majority of immunocompromised individuals, and we believe we will probably have a solution for the remainder in the not-too-distant future,” Dr. Marks said.
In its updated EUA, the FDA said that the third shots were intended for people who had undergone solid organ transplants or have an “equivalent level of immunocompromise.”
The details
Clinical experts on the CDC panel spent a good deal of time trying to suss out exactly what conditions might fall under the FDA’s umbrella for a third dose.
In a presentation to the committee, Neela Goswami, MD, PhD, an assistant professor of infectious diseases at Emory University School of Medicine and of epidemiology at the Emory Rollins School of Public Health, Atlanta, stressed that the shots are intended for patients who are moderately or severely immunocompromised, in close consultation with their doctors, but that people who should qualify would include those:
- Receiving treatment for solid tumors or blood cancers
- Taking immunosuppressing medications after a solid organ transplant
- Within 2 years of receiving CAR-T therapy or a stem cell transplant
- Who have primary immunodeficiencies – rare genetic disorders that prevent the immune system from working properly
- With advanced or untreated
- Taking high-dose corticosteroids (more than 20 milligrams of or its equivalent daily), alkylating agents, antimetabolites, chemotherapy, TNF blockers, or other immunomodulating or immunosuppressing biologics
- With certain chronic medical conditions, such as or asplenia – living without a spleen
- Receiving dialysis
In discussion, CDC experts clarified that these third doses were not intended for people whose immune function had waned with age, such as elderly residents of long-term care facilities or people with chronic diseases like diabetes.
The idea is to try to get a third dose of the vaccine they’ve already had – Moderna or Pfizer – but if that’s not feasible, it’s fine for the third dose to be different from what someone has had before. The third dose should be given at least 28 days after a second dose, and, ideally, before the initiation of immunosuppressive therapy.
Participants in the meeting said that the CDC would post updated materials on its website to help guide physicians on exactly who should receive third doses.
Ultimately, however, the extra doses will be given on an honor system; no prescriptions or other kinds of clinical documentation will be required for people to get a third dose of these shots.
Tests to measure neutralizing antibodies are also not recommended before the shots are given because of differences in the types of tests used to measure these antibodies and the difficulty in interpreting them. It’s unclear right now what level of neutralizing antibodies is needed for protection.
‘Peace of mind’
In public testimony, Heather Braaten, a 44-year-old being treated for ovarian cancer, said she was grateful to have gotten two shots of the Pfizer vaccine last winter, in between rounds of chemotherapy, but she knew she was probably not well protected. She said she’d become obsessive over the past few months reading medical studies and trying to understand her risk.
“I have felt distraught over the situation. My prognosis is poor. I most likely have about two to three years left to live, so everything counts,” Ms. Braaten said.
She said her life ambitions were humble. She wants to visit with friends and family and not have to worry that she’ll be a breakthrough case. She wants to go grocery shopping again and “not panic and leave the store after five minutes.” She’d love to feel free to travel, she said.
“While I understand I still need to be cautious, I am hopeful for the peace of mind and greater freedom a third shot can provide,” Ms. Braaten said.
More boosters on the way?
In the second half of the meeting, the CDC also signaled that it was considering the use of boosters for people whose immunity might have waned in the months since they had completed their vaccine series, particularly seniors. About 75% of people hospitalized with vaccine breakthrough cases are over age 65, according to CDC data.
Those considerations are becoming more urgent as the Delta variant continues to pummel less vaccinated states and counties.
In its presentation to the ACIP, Heather Scobie, PhD, MPH, a member of the CDC’s COVID Response Team, highlighted data from Canada, Israel, Qatar, and the United Kingdom showing that, while the Pfizer vaccine was still highly effective at preventing hospitalizations and death, it’s far less likely when faced with Delta to prevent an infection that causes symptoms.
In Israel, Pfizer’s vaccine prevented symptoms an average of 41% of the time. In Qatar, which is also using the Moderna vaccine, Pfizer’s prevented symptomatic infections with Delta about 54% of the time compared with 85% with Moderna’s.
Dr. Scobie noted that Pfizer’s waning efficacy may have something to do with the fact that it uses a lower dosage than Moderna’s. Pfizer’s recommended dosing interval is also shorter – 3 weeks compared with 4 weeks for Moderna’s. Stretching the time between shots has been shown to boost vaccine effectiveness, she said.
New data from the Mayo clinic, published ahead of peer review, also suggest that Pfizer’s protection may be fading more quickly than Moderna’s.
In February, both shots were nearly 100% effective at preventing the SARS-CoV-2 infection, but by July, against Delta, Pfizer’s efficacy had dropped to somewhere between 13% and 62%, while Moderna’s was still effective at preventing infection between 58% and 87% of the time.
In July, Pfizer’s was between 24% and 94% effective at preventing hospitalization with a COVID-19 infection and Moderna’s was between 33% and 96% effective at preventing hospitalization.
While that may sound like cause for concern, Dr. Scobie noted that, as of August 2, severe COVD-19 outcomes after vaccination are still very rare. Among 164 million fully vaccinated people in the United States there have been about 7,000 hospitalizations and 1,500 deaths; nearly three out of four of these have been in people over the age of 65.
The ACIP will next meet on August 24 to focus solely on the COVID-19 vaccines.
A version of this article first appeared on Medscape.com.
Centers for Disease Control and Prevention Director Rochelle Walensky, MD, has officially signed off on a recommendation by an independent panel of 11 experts to allow people with weakened immune function to get a third dose of certain COVID-19 vaccines.
The decision follows a unanimous vote by the CDC’s Advisory Committee on Immunization Practices (ACIP), which in turn came hours after the U.S. Food and Drug Administration updated its Emergency Use Authorization (EUA) for the Pfizer and Moderna mRNA vaccines.
About 7 million adults in the United States have moderately to severely impaired immune function because of a medical condition they live with or a medication they take to manage a health condition.
People who fall into this category are at higher risk of being hospitalized or dying if they get COVID-19. They are also more likely to transmit the infection. About 40% of vaccinated patients who are hospitalized with breakthrough cases are immunocompromised.
Recent studies have shown that between one-third and one-half of immunocompromised people who didn’t develop antibodies after two doses of a vaccine do get some level of protection after a third dose.
Even then, however, the protection immunocompromised people get from vaccines is not as robust as someone who has healthy immune function, and some panel members were concerned that a third dose might come with a false sense of security.
“My only concern with adding a third dose for the immunocompromised is the impression that our immunocompromised population [will] then be safe,” said ACIP member Helen Talbot, MD, MPH, an associate professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn.
“I think the reality is they’ll be safer but still at incredibly high risk for severe disease and death,” she said.
In updating its EUA, the FDA stressed that, even after a third dose, people who are immunocompromised will still need to wear a mask indoors, socially distance, and avoid large crowds. In addition, family members and other close contacts should be fully vaccinated to protect these vulnerable individuals.
Johnson & Johnson not in the mix
The boosters will be available to children as young as 12 years of age who’ve had a Pfizer vaccine or those ages 18 and older who’ve gotten the Moderna vaccine.
For now, people who’ve had the one-dose Johnson & Johnson vaccine have not been cleared to get a second dose of any vaccine.
FDA experts acknowledged the gap but said that people who had received the Johnson & Johnson vaccine represented a small slice of vaccinated Americans, and said they couldn’t act before the FDA had updated its authorization for that vaccine, which the agency is actively exploring.
“We had to do what we’re doing based on the data we have in hand,” said Peter Marks, MD, director of the Center for Biologics Evaluation and Research at the FDA, the division of the agency that regulates vaccines.
“We think at least there is a solution here for the very large majority of immunocompromised individuals, and we believe we will probably have a solution for the remainder in the not-too-distant future,” Dr. Marks said.
In its updated EUA, the FDA said that the third shots were intended for people who had undergone solid organ transplants or have an “equivalent level of immunocompromise.”
The details
Clinical experts on the CDC panel spent a good deal of time trying to suss out exactly what conditions might fall under the FDA’s umbrella for a third dose.
In a presentation to the committee, Neela Goswami, MD, PhD, an assistant professor of infectious diseases at Emory University School of Medicine and of epidemiology at the Emory Rollins School of Public Health, Atlanta, stressed that the shots are intended for patients who are moderately or severely immunocompromised, in close consultation with their doctors, but that people who should qualify would include those:
- Receiving treatment for solid tumors or blood cancers
- Taking immunosuppressing medications after a solid organ transplant
- Within 2 years of receiving CAR-T therapy or a stem cell transplant
- Who have primary immunodeficiencies – rare genetic disorders that prevent the immune system from working properly
- With advanced or untreated
- Taking high-dose corticosteroids (more than 20 milligrams of or its equivalent daily), alkylating agents, antimetabolites, chemotherapy, TNF blockers, or other immunomodulating or immunosuppressing biologics
- With certain chronic medical conditions, such as or asplenia – living without a spleen
- Receiving dialysis
In discussion, CDC experts clarified that these third doses were not intended for people whose immune function had waned with age, such as elderly residents of long-term care facilities or people with chronic diseases like diabetes.
The idea is to try to get a third dose of the vaccine they’ve already had – Moderna or Pfizer – but if that’s not feasible, it’s fine for the third dose to be different from what someone has had before. The third dose should be given at least 28 days after a second dose, and, ideally, before the initiation of immunosuppressive therapy.
Participants in the meeting said that the CDC would post updated materials on its website to help guide physicians on exactly who should receive third doses.
Ultimately, however, the extra doses will be given on an honor system; no prescriptions or other kinds of clinical documentation will be required for people to get a third dose of these shots.
Tests to measure neutralizing antibodies are also not recommended before the shots are given because of differences in the types of tests used to measure these antibodies and the difficulty in interpreting them. It’s unclear right now what level of neutralizing antibodies is needed for protection.
‘Peace of mind’
In public testimony, Heather Braaten, a 44-year-old being treated for ovarian cancer, said she was grateful to have gotten two shots of the Pfizer vaccine last winter, in between rounds of chemotherapy, but she knew she was probably not well protected. She said she’d become obsessive over the past few months reading medical studies and trying to understand her risk.
“I have felt distraught over the situation. My prognosis is poor. I most likely have about two to three years left to live, so everything counts,” Ms. Braaten said.
She said her life ambitions were humble. She wants to visit with friends and family and not have to worry that she’ll be a breakthrough case. She wants to go grocery shopping again and “not panic and leave the store after five minutes.” She’d love to feel free to travel, she said.
“While I understand I still need to be cautious, I am hopeful for the peace of mind and greater freedom a third shot can provide,” Ms. Braaten said.
More boosters on the way?
In the second half of the meeting, the CDC also signaled that it was considering the use of boosters for people whose immunity might have waned in the months since they had completed their vaccine series, particularly seniors. About 75% of people hospitalized with vaccine breakthrough cases are over age 65, according to CDC data.
Those considerations are becoming more urgent as the Delta variant continues to pummel less vaccinated states and counties.
In its presentation to the ACIP, Heather Scobie, PhD, MPH, a member of the CDC’s COVID Response Team, highlighted data from Canada, Israel, Qatar, and the United Kingdom showing that, while the Pfizer vaccine was still highly effective at preventing hospitalizations and death, it’s far less likely when faced with Delta to prevent an infection that causes symptoms.
In Israel, Pfizer’s vaccine prevented symptoms an average of 41% of the time. In Qatar, which is also using the Moderna vaccine, Pfizer’s prevented symptomatic infections with Delta about 54% of the time compared with 85% with Moderna’s.
Dr. Scobie noted that Pfizer’s waning efficacy may have something to do with the fact that it uses a lower dosage than Moderna’s. Pfizer’s recommended dosing interval is also shorter – 3 weeks compared with 4 weeks for Moderna’s. Stretching the time between shots has been shown to boost vaccine effectiveness, she said.
New data from the Mayo clinic, published ahead of peer review, also suggest that Pfizer’s protection may be fading more quickly than Moderna’s.
In February, both shots were nearly 100% effective at preventing the SARS-CoV-2 infection, but by July, against Delta, Pfizer’s efficacy had dropped to somewhere between 13% and 62%, while Moderna’s was still effective at preventing infection between 58% and 87% of the time.
In July, Pfizer’s was between 24% and 94% effective at preventing hospitalization with a COVID-19 infection and Moderna’s was between 33% and 96% effective at preventing hospitalization.
While that may sound like cause for concern, Dr. Scobie noted that, as of August 2, severe COVD-19 outcomes after vaccination are still very rare. Among 164 million fully vaccinated people in the United States there have been about 7,000 hospitalizations and 1,500 deaths; nearly three out of four of these have been in people over the age of 65.
The ACIP will next meet on August 24 to focus solely on the COVID-19 vaccines.
A version of this article first appeared on Medscape.com.
Heparin’s COVID-19 benefit greatest in moderately ill patients
Critically ill derive no benefit
Therapeutic levels of heparin can have widely varying effects on COVID-19 patients depending on the severity of their disease, according to a multiplatform clinical trial that analyzed patient data from three international trials.
COVID-19 patients in the ICU, or at least receiving ICU-level care, derived no benefit from anticoagulation with heparin, while non–critically ill COVID-19 patients – those who were hospitalized but not receiving ICU-level care – on the same anticoagulation were less likely to progress to need respiratory or cardiovascular organ support despite a slightly heightened risk of bleeding events.
Reporting in two articles published online in the New England Journal of Medicine, authors of three international trials combined their data into one multiplatform trial that makes a strong case for prescribing therapeutic levels of heparin in hospitalized patients not receiving ICU-level care were non–critically ill and critically ill.
“I think this is going to be a game changer,” said Jeffrey S. Berger, MD, ACTIV-4a co–principal investigator and co–first author of the study of non–critically ill patients. “I think that using therapeutic-dose anticoagulation should improve outcomes in the tens of thousands of patients worldwide. I hope our data can have a global impact.”
Outcomes based on disease severity
The multiplatform trial analyzed data from the Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC); A Multicenter, Adaptive, Randomized Controlled Platform Trial of the Safety and Efficacy of Antithrombotic Strategies in Hospitalized Adults with COVID-19 (ACTIV-4a); and Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP).
The trial evaluated 2,219 non–critically ill hospitalized patients, 1,181 of whom were randomized to therapeutic-dose anticoagulation; and 1,098 critically ill patients, 534 of whom were prescribed therapeutic levels of heparin.
In the critically ill patients, those on heparin were no more likely to get discharged or spend fewer days on respiratory or CV organ support – oxygen, mechanical ventilation, life support, vasopressors or inotropes – than were those on usual-care thromboprophylaxis. The investigators stopped the trial in both patient populations: in critically ill patients when it became obvious therapeutic-dose anticoagulation was having no impact; and in moderately ill patients when the trial met the prespecified criteria for the superiority of therapeutic-dose anticoagulation.
ICU patients on therapeutic-level heparin spent an average of 1 day free of organ support vs. 4 for patients on usual-care prophylactic antithrombotic drugs. The percentage of patients who survived to hospital discharge was similar in the therapeutic-level and usual-care critically ill patients: 62.7% and 64.5%, respectively. Major bleeding occurred in 3.8% and 2.8%, respectively. Demographic and clinical characteristics were similar between both patient groups.
However, in non–critically ill patients, therapeutic levels of heparin resulted in a marked improvement in outcomes. The researchers estimated that, for every 1,000 hospitalized patients with what they labeled moderate disease, an initial treatment with therapeutic-dose heparin resulted in 40 additional patients surviving compared to usual-care thromboprophylaxis.
The percentages of patients not needing organ support before hospital discharge was 80.2% on therapeutic-dose heparin and 76.4% on usual-care therapy. In terms of adjusted odds ratio, the anticoagulation group had a 27% improved chance of not needing daily organ support.
Those improvements came with an additional seven major bleeding events per 1,000 patients. That broke down to a rate of 1.9% in the therapeutic-dose and 0.9% in the usual-care patients.
As the Delta variant of COVID-19 spreads, Patrick R. Lawler, MD, MPH, principal investigator of the ATTACC trial, said there’s no reason these findings shouldn’t apply for all variants of the disease.
Dr. Lawler, a physician-scientist at Peter Munk Cardiac Centre at Toronto General Hospital, noted that the multiplatform study did not account for disease variant. “Ongoing clinical trials are tracking the variant patients have or the variants that are most prevalent in an area at that time,” he said. “It may be easier in future trials to look at that question.”
Explaining heparin’s varying effects
The study did not specifically sort out why moderately ill patients fared better on heparin than their critically ill counterparts, but Dr. Lawler speculated on possible reasons. “One might be that the extent of illness severity is too extreme in the ICU-level population for heparin to have a beneficial extent,” he said.
He acknowledged that higher rates of macrovascular thrombosis, such as venous thromboembolism, in ICU patients would suggest that heparin would have a greater beneficial effect, but, he added, “it may also suggest how advanced that process is, and perhaps heparin is not adequate to reverse the course at that point given relatively extensive thrombosis and associate organ failure.”
As clinicians have gained experience dealing with COVID-19, they’ve learned that infected patients carry a high burden of macro- and microthrombosis, Dr. Berger said, which may explain why critically ill patients didn’t respond as well to therapeutic levels of heparin. “I think the cat is out of the bag; patients who are severe are too ill to benefit,” he said. “I would think there’s too much microthrombosis that is already in their bodies.”
However, this doesn’t completely rule out therapeutic levels of heparin in critically ill COVID-19 patients. There are some scenarios where it’s needed, said Dr. Berger, associate professor of medicine and surgery and director of the Center for the Prevention of Cardiovascular Disease at New York University Langone Health. “Anyone who has a known clot already, like a known macrothrombosis in their leg or lung, needs to be on full-dose heparin,” he said.
That rationale can help reconcile the different outcomes in the critically and non–critically ill COVID-19 patients, wrote Hugo ten Cate, MD, PhD, of Maastricht University in the Netherlands, wrote in an accompanying editorial. But differences in the study populations may also explain the divergent outcomes, Dr. ten Cate noted.
The studies suggest that critically ill patients may need hon-heparin antithrombotic approaches “or even profibrinolytic strategies,” Dr. Cate wrote, and that the safety and effectiveness of thromboprophylaxis “remains an important question.” Nonetheless, he added, treating physicians must deal with the bleeding risk when using heparin or low-molecular-weight heparin in moderately ill COVID-19 patients.
Deepak L. Bhatt MD, MPH, of Brigham and Women’s Hospital Heart & Vascular Center, Boston, said in an interview that reconciling the two studies was “a bit challenging,” because effective therapies tend to have a greater impact in sicker patients.
“Of course, with antithrombotic therapies, bleeding side effects can sometimes overwhelm benefits in patients who are at high risk of both bleeding and ischemic complications, though that does not seem to be the explanation here,” Dr. Bhatt said. “I do think we need more data to clarify exactly which COVID patients benefit from various antithrombotic regimens, and fortunately, there are other ongoing studies, some of which will report relatively soon.”
He concurred with Dr. Berger that patients who need anticoagulation should receive it “apart from their COVID status,” Dr. Bhatt said. “Sick, hospitalized patients with or without COVID should receive appropriate prophylactic doses of anticoagulation.” However, he added, “Whether we should routinely go beyond that in COVID-positive inpatients, I think we need more data.”
The ATTACC platform received grants from the Canadian Institutes of Health Research and several other research foundations. The ACTIV-4a platform received funding from the National Heart, Lung, and Blood Institute. REMAP-CAP received funding from the European Union and several international research foundations, as well as Amgen and Eisai.
Dr. Lawler had no relationships to disclose. Dr. Berger disclosed receiving grants from the NHLBI, and financial relationships with AstraZeneca, Janssen, and Amgen outside the submitted work. Dr. ten Cate reported relationships with Alveron, Coagulation Profile, Portola/Alexion, Bayer, Pfizer, Stago, Leo Pharma, Daiichi, and Gilead/Galapagos. Dr. Bhatt is chair of the data safety and monitoring board of the FREEDOM COVID anticoagulation clinical trial.
Critically ill derive no benefit
Critically ill derive no benefit
Therapeutic levels of heparin can have widely varying effects on COVID-19 patients depending on the severity of their disease, according to a multiplatform clinical trial that analyzed patient data from three international trials.
COVID-19 patients in the ICU, or at least receiving ICU-level care, derived no benefit from anticoagulation with heparin, while non–critically ill COVID-19 patients – those who were hospitalized but not receiving ICU-level care – on the same anticoagulation were less likely to progress to need respiratory or cardiovascular organ support despite a slightly heightened risk of bleeding events.
Reporting in two articles published online in the New England Journal of Medicine, authors of three international trials combined their data into one multiplatform trial that makes a strong case for prescribing therapeutic levels of heparin in hospitalized patients not receiving ICU-level care were non–critically ill and critically ill.
“I think this is going to be a game changer,” said Jeffrey S. Berger, MD, ACTIV-4a co–principal investigator and co–first author of the study of non–critically ill patients. “I think that using therapeutic-dose anticoagulation should improve outcomes in the tens of thousands of patients worldwide. I hope our data can have a global impact.”
Outcomes based on disease severity
The multiplatform trial analyzed data from the Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC); A Multicenter, Adaptive, Randomized Controlled Platform Trial of the Safety and Efficacy of Antithrombotic Strategies in Hospitalized Adults with COVID-19 (ACTIV-4a); and Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP).
The trial evaluated 2,219 non–critically ill hospitalized patients, 1,181 of whom were randomized to therapeutic-dose anticoagulation; and 1,098 critically ill patients, 534 of whom were prescribed therapeutic levels of heparin.
In the critically ill patients, those on heparin were no more likely to get discharged or spend fewer days on respiratory or CV organ support – oxygen, mechanical ventilation, life support, vasopressors or inotropes – than were those on usual-care thromboprophylaxis. The investigators stopped the trial in both patient populations: in critically ill patients when it became obvious therapeutic-dose anticoagulation was having no impact; and in moderately ill patients when the trial met the prespecified criteria for the superiority of therapeutic-dose anticoagulation.
ICU patients on therapeutic-level heparin spent an average of 1 day free of organ support vs. 4 for patients on usual-care prophylactic antithrombotic drugs. The percentage of patients who survived to hospital discharge was similar in the therapeutic-level and usual-care critically ill patients: 62.7% and 64.5%, respectively. Major bleeding occurred in 3.8% and 2.8%, respectively. Demographic and clinical characteristics were similar between both patient groups.
However, in non–critically ill patients, therapeutic levels of heparin resulted in a marked improvement in outcomes. The researchers estimated that, for every 1,000 hospitalized patients with what they labeled moderate disease, an initial treatment with therapeutic-dose heparin resulted in 40 additional patients surviving compared to usual-care thromboprophylaxis.
The percentages of patients not needing organ support before hospital discharge was 80.2% on therapeutic-dose heparin and 76.4% on usual-care therapy. In terms of adjusted odds ratio, the anticoagulation group had a 27% improved chance of not needing daily organ support.
Those improvements came with an additional seven major bleeding events per 1,000 patients. That broke down to a rate of 1.9% in the therapeutic-dose and 0.9% in the usual-care patients.
As the Delta variant of COVID-19 spreads, Patrick R. Lawler, MD, MPH, principal investigator of the ATTACC trial, said there’s no reason these findings shouldn’t apply for all variants of the disease.
Dr. Lawler, a physician-scientist at Peter Munk Cardiac Centre at Toronto General Hospital, noted that the multiplatform study did not account for disease variant. “Ongoing clinical trials are tracking the variant patients have or the variants that are most prevalent in an area at that time,” he said. “It may be easier in future trials to look at that question.”
Explaining heparin’s varying effects
The study did not specifically sort out why moderately ill patients fared better on heparin than their critically ill counterparts, but Dr. Lawler speculated on possible reasons. “One might be that the extent of illness severity is too extreme in the ICU-level population for heparin to have a beneficial extent,” he said.
He acknowledged that higher rates of macrovascular thrombosis, such as venous thromboembolism, in ICU patients would suggest that heparin would have a greater beneficial effect, but, he added, “it may also suggest how advanced that process is, and perhaps heparin is not adequate to reverse the course at that point given relatively extensive thrombosis and associate organ failure.”
As clinicians have gained experience dealing with COVID-19, they’ve learned that infected patients carry a high burden of macro- and microthrombosis, Dr. Berger said, which may explain why critically ill patients didn’t respond as well to therapeutic levels of heparin. “I think the cat is out of the bag; patients who are severe are too ill to benefit,” he said. “I would think there’s too much microthrombosis that is already in their bodies.”
However, this doesn’t completely rule out therapeutic levels of heparin in critically ill COVID-19 patients. There are some scenarios where it’s needed, said Dr. Berger, associate professor of medicine and surgery and director of the Center for the Prevention of Cardiovascular Disease at New York University Langone Health. “Anyone who has a known clot already, like a known macrothrombosis in their leg or lung, needs to be on full-dose heparin,” he said.
That rationale can help reconcile the different outcomes in the critically and non–critically ill COVID-19 patients, wrote Hugo ten Cate, MD, PhD, of Maastricht University in the Netherlands, wrote in an accompanying editorial. But differences in the study populations may also explain the divergent outcomes, Dr. ten Cate noted.
The studies suggest that critically ill patients may need hon-heparin antithrombotic approaches “or even profibrinolytic strategies,” Dr. Cate wrote, and that the safety and effectiveness of thromboprophylaxis “remains an important question.” Nonetheless, he added, treating physicians must deal with the bleeding risk when using heparin or low-molecular-weight heparin in moderately ill COVID-19 patients.
Deepak L. Bhatt MD, MPH, of Brigham and Women’s Hospital Heart & Vascular Center, Boston, said in an interview that reconciling the two studies was “a bit challenging,” because effective therapies tend to have a greater impact in sicker patients.
“Of course, with antithrombotic therapies, bleeding side effects can sometimes overwhelm benefits in patients who are at high risk of both bleeding and ischemic complications, though that does not seem to be the explanation here,” Dr. Bhatt said. “I do think we need more data to clarify exactly which COVID patients benefit from various antithrombotic regimens, and fortunately, there are other ongoing studies, some of which will report relatively soon.”
He concurred with Dr. Berger that patients who need anticoagulation should receive it “apart from their COVID status,” Dr. Bhatt said. “Sick, hospitalized patients with or without COVID should receive appropriate prophylactic doses of anticoagulation.” However, he added, “Whether we should routinely go beyond that in COVID-positive inpatients, I think we need more data.”
The ATTACC platform received grants from the Canadian Institutes of Health Research and several other research foundations. The ACTIV-4a platform received funding from the National Heart, Lung, and Blood Institute. REMAP-CAP received funding from the European Union and several international research foundations, as well as Amgen and Eisai.
Dr. Lawler had no relationships to disclose. Dr. Berger disclosed receiving grants from the NHLBI, and financial relationships with AstraZeneca, Janssen, and Amgen outside the submitted work. Dr. ten Cate reported relationships with Alveron, Coagulation Profile, Portola/Alexion, Bayer, Pfizer, Stago, Leo Pharma, Daiichi, and Gilead/Galapagos. Dr. Bhatt is chair of the data safety and monitoring board of the FREEDOM COVID anticoagulation clinical trial.
Therapeutic levels of heparin can have widely varying effects on COVID-19 patients depending on the severity of their disease, according to a multiplatform clinical trial that analyzed patient data from three international trials.
COVID-19 patients in the ICU, or at least receiving ICU-level care, derived no benefit from anticoagulation with heparin, while non–critically ill COVID-19 patients – those who were hospitalized but not receiving ICU-level care – on the same anticoagulation were less likely to progress to need respiratory or cardiovascular organ support despite a slightly heightened risk of bleeding events.
Reporting in two articles published online in the New England Journal of Medicine, authors of three international trials combined their data into one multiplatform trial that makes a strong case for prescribing therapeutic levels of heparin in hospitalized patients not receiving ICU-level care were non–critically ill and critically ill.
“I think this is going to be a game changer,” said Jeffrey S. Berger, MD, ACTIV-4a co–principal investigator and co–first author of the study of non–critically ill patients. “I think that using therapeutic-dose anticoagulation should improve outcomes in the tens of thousands of patients worldwide. I hope our data can have a global impact.”
Outcomes based on disease severity
The multiplatform trial analyzed data from the Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC); A Multicenter, Adaptive, Randomized Controlled Platform Trial of the Safety and Efficacy of Antithrombotic Strategies in Hospitalized Adults with COVID-19 (ACTIV-4a); and Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP).
The trial evaluated 2,219 non–critically ill hospitalized patients, 1,181 of whom were randomized to therapeutic-dose anticoagulation; and 1,098 critically ill patients, 534 of whom were prescribed therapeutic levels of heparin.
In the critically ill patients, those on heparin were no more likely to get discharged or spend fewer days on respiratory or CV organ support – oxygen, mechanical ventilation, life support, vasopressors or inotropes – than were those on usual-care thromboprophylaxis. The investigators stopped the trial in both patient populations: in critically ill patients when it became obvious therapeutic-dose anticoagulation was having no impact; and in moderately ill patients when the trial met the prespecified criteria for the superiority of therapeutic-dose anticoagulation.
ICU patients on therapeutic-level heparin spent an average of 1 day free of organ support vs. 4 for patients on usual-care prophylactic antithrombotic drugs. The percentage of patients who survived to hospital discharge was similar in the therapeutic-level and usual-care critically ill patients: 62.7% and 64.5%, respectively. Major bleeding occurred in 3.8% and 2.8%, respectively. Demographic and clinical characteristics were similar between both patient groups.
However, in non–critically ill patients, therapeutic levels of heparin resulted in a marked improvement in outcomes. The researchers estimated that, for every 1,000 hospitalized patients with what they labeled moderate disease, an initial treatment with therapeutic-dose heparin resulted in 40 additional patients surviving compared to usual-care thromboprophylaxis.
The percentages of patients not needing organ support before hospital discharge was 80.2% on therapeutic-dose heparin and 76.4% on usual-care therapy. In terms of adjusted odds ratio, the anticoagulation group had a 27% improved chance of not needing daily organ support.
Those improvements came with an additional seven major bleeding events per 1,000 patients. That broke down to a rate of 1.9% in the therapeutic-dose and 0.9% in the usual-care patients.
As the Delta variant of COVID-19 spreads, Patrick R. Lawler, MD, MPH, principal investigator of the ATTACC trial, said there’s no reason these findings shouldn’t apply for all variants of the disease.
Dr. Lawler, a physician-scientist at Peter Munk Cardiac Centre at Toronto General Hospital, noted that the multiplatform study did not account for disease variant. “Ongoing clinical trials are tracking the variant patients have or the variants that are most prevalent in an area at that time,” he said. “It may be easier in future trials to look at that question.”
Explaining heparin’s varying effects
The study did not specifically sort out why moderately ill patients fared better on heparin than their critically ill counterparts, but Dr. Lawler speculated on possible reasons. “One might be that the extent of illness severity is too extreme in the ICU-level population for heparin to have a beneficial extent,” he said.
He acknowledged that higher rates of macrovascular thrombosis, such as venous thromboembolism, in ICU patients would suggest that heparin would have a greater beneficial effect, but, he added, “it may also suggest how advanced that process is, and perhaps heparin is not adequate to reverse the course at that point given relatively extensive thrombosis and associate organ failure.”
As clinicians have gained experience dealing with COVID-19, they’ve learned that infected patients carry a high burden of macro- and microthrombosis, Dr. Berger said, which may explain why critically ill patients didn’t respond as well to therapeutic levels of heparin. “I think the cat is out of the bag; patients who are severe are too ill to benefit,” he said. “I would think there’s too much microthrombosis that is already in their bodies.”
However, this doesn’t completely rule out therapeutic levels of heparin in critically ill COVID-19 patients. There are some scenarios where it’s needed, said Dr. Berger, associate professor of medicine and surgery and director of the Center for the Prevention of Cardiovascular Disease at New York University Langone Health. “Anyone who has a known clot already, like a known macrothrombosis in their leg or lung, needs to be on full-dose heparin,” he said.
That rationale can help reconcile the different outcomes in the critically and non–critically ill COVID-19 patients, wrote Hugo ten Cate, MD, PhD, of Maastricht University in the Netherlands, wrote in an accompanying editorial. But differences in the study populations may also explain the divergent outcomes, Dr. ten Cate noted.
The studies suggest that critically ill patients may need hon-heparin antithrombotic approaches “or even profibrinolytic strategies,” Dr. Cate wrote, and that the safety and effectiveness of thromboprophylaxis “remains an important question.” Nonetheless, he added, treating physicians must deal with the bleeding risk when using heparin or low-molecular-weight heparin in moderately ill COVID-19 patients.
Deepak L. Bhatt MD, MPH, of Brigham and Women’s Hospital Heart & Vascular Center, Boston, said in an interview that reconciling the two studies was “a bit challenging,” because effective therapies tend to have a greater impact in sicker patients.
“Of course, with antithrombotic therapies, bleeding side effects can sometimes overwhelm benefits in patients who are at high risk of both bleeding and ischemic complications, though that does not seem to be the explanation here,” Dr. Bhatt said. “I do think we need more data to clarify exactly which COVID patients benefit from various antithrombotic regimens, and fortunately, there are other ongoing studies, some of which will report relatively soon.”
He concurred with Dr. Berger that patients who need anticoagulation should receive it “apart from their COVID status,” Dr. Bhatt said. “Sick, hospitalized patients with or without COVID should receive appropriate prophylactic doses of anticoagulation.” However, he added, “Whether we should routinely go beyond that in COVID-positive inpatients, I think we need more data.”
The ATTACC platform received grants from the Canadian Institutes of Health Research and several other research foundations. The ACTIV-4a platform received funding from the National Heart, Lung, and Blood Institute. REMAP-CAP received funding from the European Union and several international research foundations, as well as Amgen and Eisai.
Dr. Lawler had no relationships to disclose. Dr. Berger disclosed receiving grants from the NHLBI, and financial relationships with AstraZeneca, Janssen, and Amgen outside the submitted work. Dr. ten Cate reported relationships with Alveron, Coagulation Profile, Portola/Alexion, Bayer, Pfizer, Stago, Leo Pharma, Daiichi, and Gilead/Galapagos. Dr. Bhatt is chair of the data safety and monitoring board of the FREEDOM COVID anticoagulation clinical trial.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE