User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Study findings support consideration of second biopsy for transected melanomas
in a review of cases at the university.
Had their true Breslow depths been known before definitive surgery, sentinel lymph node (SLN) biopsies and wider surgical margins would likely have been recommended.
The findings led the investigators to conclude that a second biopsy should be considered when the first one is transected to ensure surgical and other management decisions are based on an accurate Breslow depth.
A second biopsy is especially warranted for broadly transected biopsies and transected T1a tumors with gross residual tumor or pigment on preoperative exam; both scenarios significantly increased the risk of up-staging in the study, according to lead investigator James Duncan, MD, a Mohs surgery and dermatologic oncology fellow at the University of Alabama at Birmingham, who presented the findings at the annual meeting of the American College of Mohs Surgery.
“Accurate staging of malignancies, especially melanoma, is critical to determine prognosis and the best treatment approach,” said Vishal Patel, MD, director of cutaneous oncology at George Washington University, Washington, when asked for comment.
“This study identifies how transected biopsies can underestimate a melanoma’s true depth and thus impact treatment and outcomes. The authors highlight that when a biopsy is transected, or there is notable pigment at the base, attempts should be taken to sample the remaining tumor prior to surgery so the accurate tumor depth can be determined and treatment options be fully discussed with the patient,” Dr. Patel said.
The Birmingham team reviewed invasive melanoma cases at their university from 2017 to 2019.
Almost half (49.6%) of the 726 melanomas they identified were transected on biopsy, which is in line with prior reports. About 60% of the patients were men and 98% were White; the average age was 63 years.
Of the 360 transected tumors, 49 (13.6%) had up-staging at final excision that “would have prompted discussion of alternate surgical treatment such as SLN biopsy or wider surgical margins,” the team said.
Of the 89 transected pT1a melanomas identified, 47.1% with gross residual tumor or pigment on preoperative physical examination were up-staged following excision versus 6.9% with no remaining pigment or tumor prior to surgery (P < .01).
Broadly transected tumors were up-staged in 21.7% of cases versus 4.9% of focally transected tumors (P = .038). The average increase in Breslow depth for broadly transected tumors was 1.03 mm versus 0.03 mm for focally transected lesions (P = .04).
Shave biopsies, ulceration, and lack of concern for melanoma at the initial biopsy were among the factors associated with a higher risk of transection.
Superficial spreading melanoma was the most common subtype. Tumors were evenly distributed between the head, neck, and extremities. The average Breslow depth was 1.51 mm, and the majority of tumors were pT1a or pT2a.
The review excluded melanoma in situ, recurrences, metastases, noncutaneous melanomas, and biopsies where deep margin status was unknown.
There was no funding for the study, and Dr. Duncan and Dr. Patel had no relevant disclosures.
in a review of cases at the university.
Had their true Breslow depths been known before definitive surgery, sentinel lymph node (SLN) biopsies and wider surgical margins would likely have been recommended.
The findings led the investigators to conclude that a second biopsy should be considered when the first one is transected to ensure surgical and other management decisions are based on an accurate Breslow depth.
A second biopsy is especially warranted for broadly transected biopsies and transected T1a tumors with gross residual tumor or pigment on preoperative exam; both scenarios significantly increased the risk of up-staging in the study, according to lead investigator James Duncan, MD, a Mohs surgery and dermatologic oncology fellow at the University of Alabama at Birmingham, who presented the findings at the annual meeting of the American College of Mohs Surgery.
“Accurate staging of malignancies, especially melanoma, is critical to determine prognosis and the best treatment approach,” said Vishal Patel, MD, director of cutaneous oncology at George Washington University, Washington, when asked for comment.
“This study identifies how transected biopsies can underestimate a melanoma’s true depth and thus impact treatment and outcomes. The authors highlight that when a biopsy is transected, or there is notable pigment at the base, attempts should be taken to sample the remaining tumor prior to surgery so the accurate tumor depth can be determined and treatment options be fully discussed with the patient,” Dr. Patel said.
The Birmingham team reviewed invasive melanoma cases at their university from 2017 to 2019.
Almost half (49.6%) of the 726 melanomas they identified were transected on biopsy, which is in line with prior reports. About 60% of the patients were men and 98% were White; the average age was 63 years.
Of the 360 transected tumors, 49 (13.6%) had up-staging at final excision that “would have prompted discussion of alternate surgical treatment such as SLN biopsy or wider surgical margins,” the team said.
Of the 89 transected pT1a melanomas identified, 47.1% with gross residual tumor or pigment on preoperative physical examination were up-staged following excision versus 6.9% with no remaining pigment or tumor prior to surgery (P < .01).
Broadly transected tumors were up-staged in 21.7% of cases versus 4.9% of focally transected tumors (P = .038). The average increase in Breslow depth for broadly transected tumors was 1.03 mm versus 0.03 mm for focally transected lesions (P = .04).
Shave biopsies, ulceration, and lack of concern for melanoma at the initial biopsy were among the factors associated with a higher risk of transection.
Superficial spreading melanoma was the most common subtype. Tumors were evenly distributed between the head, neck, and extremities. The average Breslow depth was 1.51 mm, and the majority of tumors were pT1a or pT2a.
The review excluded melanoma in situ, recurrences, metastases, noncutaneous melanomas, and biopsies where deep margin status was unknown.
There was no funding for the study, and Dr. Duncan and Dr. Patel had no relevant disclosures.
in a review of cases at the university.
Had their true Breslow depths been known before definitive surgery, sentinel lymph node (SLN) biopsies and wider surgical margins would likely have been recommended.
The findings led the investigators to conclude that a second biopsy should be considered when the first one is transected to ensure surgical and other management decisions are based on an accurate Breslow depth.
A second biopsy is especially warranted for broadly transected biopsies and transected T1a tumors with gross residual tumor or pigment on preoperative exam; both scenarios significantly increased the risk of up-staging in the study, according to lead investigator James Duncan, MD, a Mohs surgery and dermatologic oncology fellow at the University of Alabama at Birmingham, who presented the findings at the annual meeting of the American College of Mohs Surgery.
“Accurate staging of malignancies, especially melanoma, is critical to determine prognosis and the best treatment approach,” said Vishal Patel, MD, director of cutaneous oncology at George Washington University, Washington, when asked for comment.
“This study identifies how transected biopsies can underestimate a melanoma’s true depth and thus impact treatment and outcomes. The authors highlight that when a biopsy is transected, or there is notable pigment at the base, attempts should be taken to sample the remaining tumor prior to surgery so the accurate tumor depth can be determined and treatment options be fully discussed with the patient,” Dr. Patel said.
The Birmingham team reviewed invasive melanoma cases at their university from 2017 to 2019.
Almost half (49.6%) of the 726 melanomas they identified were transected on biopsy, which is in line with prior reports. About 60% of the patients were men and 98% were White; the average age was 63 years.
Of the 360 transected tumors, 49 (13.6%) had up-staging at final excision that “would have prompted discussion of alternate surgical treatment such as SLN biopsy or wider surgical margins,” the team said.
Of the 89 transected pT1a melanomas identified, 47.1% with gross residual tumor or pigment on preoperative physical examination were up-staged following excision versus 6.9% with no remaining pigment or tumor prior to surgery (P < .01).
Broadly transected tumors were up-staged in 21.7% of cases versus 4.9% of focally transected tumors (P = .038). The average increase in Breslow depth for broadly transected tumors was 1.03 mm versus 0.03 mm for focally transected lesions (P = .04).
Shave biopsies, ulceration, and lack of concern for melanoma at the initial biopsy were among the factors associated with a higher risk of transection.
Superficial spreading melanoma was the most common subtype. Tumors were evenly distributed between the head, neck, and extremities. The average Breslow depth was 1.51 mm, and the majority of tumors were pT1a or pT2a.
The review excluded melanoma in situ, recurrences, metastases, noncutaneous melanomas, and biopsies where deep margin status was unknown.
There was no funding for the study, and Dr. Duncan and Dr. Patel had no relevant disclosures.
FROM ACMS 2021
Intravenous immunoglobulin controls dermatomyositis in phase 3 trial
Nearly 50% achieve moderate improvement or better
The first multinational, phase 3, placebo-controlled trial conducted with intravenous immunoglobulin therapy (IVIg) for dermatomyositis has confirmed significant efficacy and acceptable safety, according to data presented at the opening plenary abstract session of the annual European Congress of Rheumatology.
At the week 16 evaluation of the trial, called ProDERM, the response rates were 78.7% and 43.8% (P = .0008) for active therapy and placebo, respectively, reported Rohit Aggarwal, MD, medical director of the Arthritis and Autoimmunity Center at the University of Pittsburgh.
ProDERM is a “much-awaited study,” according to session moderator Hendrik Schulze-Koops, MD, PhD, of the division of rheumatology and clinical immunology at Ludwig Maximilian University of Munich (Germany). He was not involved in the study.
“We all have been doing what we have been doing,” Dr. Schulze-Koops said, referring to the use of IVIg for the control of dermatomyositis, “but we had no evidence for support.”
This statement could apply not only to IVIg, which has long been listed among treatment options by the Myositis Association despite the absence of controlled studies, but also to most immunosuppressive therapies and other options used for this challenging disease.
The proprietary IVIg employed in this study, Octagam 10%, has been approved in the United States for the treatment of chronic immune thrombocytopenic purpura. Its manufacturer, Octagam, plans to file a supplemental new drug application with the Food and Drug Administration for the treatment of dermatomyositis. The agent is already approved for dermatomyositis by the European Medicines Agency, according to Dr. Aggarwal.
Multiple response criteria favor IVIg
In the trial, 95 patients with dermatomyositis were randomized to 2 g/kg of IVIg (Octagam 10%) or placebo administered every 4 weeks. In a subsequent open-label extension study in which patients on placebo were switched to active therapy, the same every-4-week treatment schedule was used. The patients’ mean age was 53; 75% were women, and 92% were White.
The primary endpoint was at least minimal improvement on 2016 ACR/EULAR (American College of Rheumatology/European Alliance of Associations for Rheumatology) myositis response criteria, defined as a 20-point or greater gain in the Total Improvement Score (TIS) and no clinical worsening at two consecutive visits. But IVIg also provided a large relative benefit over placebo using more rigorous definitions of improvement. For moderate improvement, defined as at least a 40-point TIS improvement, there was a 45.2% relative advantage for IVIg over placebo (68.1% vs. 22.9%; P < .0001). For major improvement, defined as at least a 60-point TIS improvement, the relative advantage was 23.6% (31.9% vs. 8.3%; P < .0062).
At 16 weeks, the mean TIS score was more than twice as high in those receiving IVIg than in those randomized to placebo (48.4 vs. 21.6). At that point, an open-label extension was initiated. Those in the IVIg group were permitted to remain on therapy for an additional 24 weeks if they had not worsened in the blinded phase.
The mean TIS score in the IVIg group continued to rise during the extension phase. By 12 weeks in this phase, it reached 54.0. Over the same period, mean TIS scores climbed steeply among the placebo-treated patients who had switched to active therapy, reaching 44.4.
At the end of 24 weeks of the extension trial, when patients initiated on IVIg had been on active therapy for 40 weeks, the mean TIS score advantage of starting on IVIg rather than placebo was relatively modest (55.4 vs. 51.1).
Benefit is significant for skin and muscle
Changes in the two major components of dermatomyositis were tracked individually. For skin symptoms, patients were evaluated with the Cutaneous Dermatomyositis Disease Areas and Severity Index (CDASI). For muscle involvement, symptoms were evaluated with the 8-item Manual Muscle Testing (MMT-8) tool.
“The effects of IVIg on the muscle and the skin were both highly statistically significant,” Dr. Aggarwal reported. He said the CDASI score was reduced by almost half at the end of 16 weeks among those treated with IVIg relative to those treated with placebo. Improvement in MMT-8 scores were also clinically as well as statistically significant.
The IVIg therapy was well tolerated. The most common adverse effects in this study, like those reported with IVIg when used to treat other diseases, were headache, pyrexia, and nausea, but Dr. Aggarwal reported that these were generally mild.
Serious adverse events, particularly thromboembolism, did occur over the course of the study, but the rate of events was only slightly higher in the group receiving active therapy (5.8% vs. 4.2%).
Patients who entered the study were permitted to remain on most immunosuppressive therapies, such as methotrexate, mycophenolate, tacrolimus, and glucocorticoids. Dr. Aggarwal said that the majority of patients were taking a glucocorticoid and at least one nonglucocorticoid immunosuppressant.
Effect on associated conditions is planned
The data from this trial have not yet been analyzed for the impact of IVIg on conditions that occur frequently in association with dermatomyositis, such as interstitial lung disease (ILD) and dysphagia, but Dr. Aggarwal reported that there are plans to do so. Although severe ILD was a trial exclusion, the presence of mild to moderate ILD and dysphagia were evaluated at baseline, so the impact of treatment can be assessed.
There are also plans to evaluate how the presence or absence of myositis-specific antibodies, which were also evaluated at baseline, affected response to IVIg.
Dr. Aggarwal has financial relationships with more than 15 pharmaceutical companies, including Octapharma, which provided financial support for this trial. Dr. Schulze-Koops reported no relevant potential conflicts of interest.
Nearly 50% achieve moderate improvement or better
Nearly 50% achieve moderate improvement or better
The first multinational, phase 3, placebo-controlled trial conducted with intravenous immunoglobulin therapy (IVIg) for dermatomyositis has confirmed significant efficacy and acceptable safety, according to data presented at the opening plenary abstract session of the annual European Congress of Rheumatology.
At the week 16 evaluation of the trial, called ProDERM, the response rates were 78.7% and 43.8% (P = .0008) for active therapy and placebo, respectively, reported Rohit Aggarwal, MD, medical director of the Arthritis and Autoimmunity Center at the University of Pittsburgh.
ProDERM is a “much-awaited study,” according to session moderator Hendrik Schulze-Koops, MD, PhD, of the division of rheumatology and clinical immunology at Ludwig Maximilian University of Munich (Germany). He was not involved in the study.
“We all have been doing what we have been doing,” Dr. Schulze-Koops said, referring to the use of IVIg for the control of dermatomyositis, “but we had no evidence for support.”
This statement could apply not only to IVIg, which has long been listed among treatment options by the Myositis Association despite the absence of controlled studies, but also to most immunosuppressive therapies and other options used for this challenging disease.
The proprietary IVIg employed in this study, Octagam 10%, has been approved in the United States for the treatment of chronic immune thrombocytopenic purpura. Its manufacturer, Octagam, plans to file a supplemental new drug application with the Food and Drug Administration for the treatment of dermatomyositis. The agent is already approved for dermatomyositis by the European Medicines Agency, according to Dr. Aggarwal.
Multiple response criteria favor IVIg
In the trial, 95 patients with dermatomyositis were randomized to 2 g/kg of IVIg (Octagam 10%) or placebo administered every 4 weeks. In a subsequent open-label extension study in which patients on placebo were switched to active therapy, the same every-4-week treatment schedule was used. The patients’ mean age was 53; 75% were women, and 92% were White.
The primary endpoint was at least minimal improvement on 2016 ACR/EULAR (American College of Rheumatology/European Alliance of Associations for Rheumatology) myositis response criteria, defined as a 20-point or greater gain in the Total Improvement Score (TIS) and no clinical worsening at two consecutive visits. But IVIg also provided a large relative benefit over placebo using more rigorous definitions of improvement. For moderate improvement, defined as at least a 40-point TIS improvement, there was a 45.2% relative advantage for IVIg over placebo (68.1% vs. 22.9%; P < .0001). For major improvement, defined as at least a 60-point TIS improvement, the relative advantage was 23.6% (31.9% vs. 8.3%; P < .0062).
At 16 weeks, the mean TIS score was more than twice as high in those receiving IVIg than in those randomized to placebo (48.4 vs. 21.6). At that point, an open-label extension was initiated. Those in the IVIg group were permitted to remain on therapy for an additional 24 weeks if they had not worsened in the blinded phase.
The mean TIS score in the IVIg group continued to rise during the extension phase. By 12 weeks in this phase, it reached 54.0. Over the same period, mean TIS scores climbed steeply among the placebo-treated patients who had switched to active therapy, reaching 44.4.
At the end of 24 weeks of the extension trial, when patients initiated on IVIg had been on active therapy for 40 weeks, the mean TIS score advantage of starting on IVIg rather than placebo was relatively modest (55.4 vs. 51.1).
Benefit is significant for skin and muscle
Changes in the two major components of dermatomyositis were tracked individually. For skin symptoms, patients were evaluated with the Cutaneous Dermatomyositis Disease Areas and Severity Index (CDASI). For muscle involvement, symptoms were evaluated with the 8-item Manual Muscle Testing (MMT-8) tool.
“The effects of IVIg on the muscle and the skin were both highly statistically significant,” Dr. Aggarwal reported. He said the CDASI score was reduced by almost half at the end of 16 weeks among those treated with IVIg relative to those treated with placebo. Improvement in MMT-8 scores were also clinically as well as statistically significant.
The IVIg therapy was well tolerated. The most common adverse effects in this study, like those reported with IVIg when used to treat other diseases, were headache, pyrexia, and nausea, but Dr. Aggarwal reported that these were generally mild.
Serious adverse events, particularly thromboembolism, did occur over the course of the study, but the rate of events was only slightly higher in the group receiving active therapy (5.8% vs. 4.2%).
Patients who entered the study were permitted to remain on most immunosuppressive therapies, such as methotrexate, mycophenolate, tacrolimus, and glucocorticoids. Dr. Aggarwal said that the majority of patients were taking a glucocorticoid and at least one nonglucocorticoid immunosuppressant.
Effect on associated conditions is planned
The data from this trial have not yet been analyzed for the impact of IVIg on conditions that occur frequently in association with dermatomyositis, such as interstitial lung disease (ILD) and dysphagia, but Dr. Aggarwal reported that there are plans to do so. Although severe ILD was a trial exclusion, the presence of mild to moderate ILD and dysphagia were evaluated at baseline, so the impact of treatment can be assessed.
There are also plans to evaluate how the presence or absence of myositis-specific antibodies, which were also evaluated at baseline, affected response to IVIg.
Dr. Aggarwal has financial relationships with more than 15 pharmaceutical companies, including Octapharma, which provided financial support for this trial. Dr. Schulze-Koops reported no relevant potential conflicts of interest.
The first multinational, phase 3, placebo-controlled trial conducted with intravenous immunoglobulin therapy (IVIg) for dermatomyositis has confirmed significant efficacy and acceptable safety, according to data presented at the opening plenary abstract session of the annual European Congress of Rheumatology.
At the week 16 evaluation of the trial, called ProDERM, the response rates were 78.7% and 43.8% (P = .0008) for active therapy and placebo, respectively, reported Rohit Aggarwal, MD, medical director of the Arthritis and Autoimmunity Center at the University of Pittsburgh.
ProDERM is a “much-awaited study,” according to session moderator Hendrik Schulze-Koops, MD, PhD, of the division of rheumatology and clinical immunology at Ludwig Maximilian University of Munich (Germany). He was not involved in the study.
“We all have been doing what we have been doing,” Dr. Schulze-Koops said, referring to the use of IVIg for the control of dermatomyositis, “but we had no evidence for support.”
This statement could apply not only to IVIg, which has long been listed among treatment options by the Myositis Association despite the absence of controlled studies, but also to most immunosuppressive therapies and other options used for this challenging disease.
The proprietary IVIg employed in this study, Octagam 10%, has been approved in the United States for the treatment of chronic immune thrombocytopenic purpura. Its manufacturer, Octagam, plans to file a supplemental new drug application with the Food and Drug Administration for the treatment of dermatomyositis. The agent is already approved for dermatomyositis by the European Medicines Agency, according to Dr. Aggarwal.
Multiple response criteria favor IVIg
In the trial, 95 patients with dermatomyositis were randomized to 2 g/kg of IVIg (Octagam 10%) or placebo administered every 4 weeks. In a subsequent open-label extension study in which patients on placebo were switched to active therapy, the same every-4-week treatment schedule was used. The patients’ mean age was 53; 75% were women, and 92% were White.
The primary endpoint was at least minimal improvement on 2016 ACR/EULAR (American College of Rheumatology/European Alliance of Associations for Rheumatology) myositis response criteria, defined as a 20-point or greater gain in the Total Improvement Score (TIS) and no clinical worsening at two consecutive visits. But IVIg also provided a large relative benefit over placebo using more rigorous definitions of improvement. For moderate improvement, defined as at least a 40-point TIS improvement, there was a 45.2% relative advantage for IVIg over placebo (68.1% vs. 22.9%; P < .0001). For major improvement, defined as at least a 60-point TIS improvement, the relative advantage was 23.6% (31.9% vs. 8.3%; P < .0062).
At 16 weeks, the mean TIS score was more than twice as high in those receiving IVIg than in those randomized to placebo (48.4 vs. 21.6). At that point, an open-label extension was initiated. Those in the IVIg group were permitted to remain on therapy for an additional 24 weeks if they had not worsened in the blinded phase.
The mean TIS score in the IVIg group continued to rise during the extension phase. By 12 weeks in this phase, it reached 54.0. Over the same period, mean TIS scores climbed steeply among the placebo-treated patients who had switched to active therapy, reaching 44.4.
At the end of 24 weeks of the extension trial, when patients initiated on IVIg had been on active therapy for 40 weeks, the mean TIS score advantage of starting on IVIg rather than placebo was relatively modest (55.4 vs. 51.1).
Benefit is significant for skin and muscle
Changes in the two major components of dermatomyositis were tracked individually. For skin symptoms, patients were evaluated with the Cutaneous Dermatomyositis Disease Areas and Severity Index (CDASI). For muscle involvement, symptoms were evaluated with the 8-item Manual Muscle Testing (MMT-8) tool.
“The effects of IVIg on the muscle and the skin were both highly statistically significant,” Dr. Aggarwal reported. He said the CDASI score was reduced by almost half at the end of 16 weeks among those treated with IVIg relative to those treated with placebo. Improvement in MMT-8 scores were also clinically as well as statistically significant.
The IVIg therapy was well tolerated. The most common adverse effects in this study, like those reported with IVIg when used to treat other diseases, were headache, pyrexia, and nausea, but Dr. Aggarwal reported that these were generally mild.
Serious adverse events, particularly thromboembolism, did occur over the course of the study, but the rate of events was only slightly higher in the group receiving active therapy (5.8% vs. 4.2%).
Patients who entered the study were permitted to remain on most immunosuppressive therapies, such as methotrexate, mycophenolate, tacrolimus, and glucocorticoids. Dr. Aggarwal said that the majority of patients were taking a glucocorticoid and at least one nonglucocorticoid immunosuppressant.
Effect on associated conditions is planned
The data from this trial have not yet been analyzed for the impact of IVIg on conditions that occur frequently in association with dermatomyositis, such as interstitial lung disease (ILD) and dysphagia, but Dr. Aggarwal reported that there are plans to do so. Although severe ILD was a trial exclusion, the presence of mild to moderate ILD and dysphagia were evaluated at baseline, so the impact of treatment can be assessed.
There are also plans to evaluate how the presence or absence of myositis-specific antibodies, which were also evaluated at baseline, affected response to IVIg.
Dr. Aggarwal has financial relationships with more than 15 pharmaceutical companies, including Octapharma, which provided financial support for this trial. Dr. Schulze-Koops reported no relevant potential conflicts of interest.
FROM THE EULAR 2021 CONGRESS
FDA approves secukinumab in psoriasis patients age six and older
The who are candidates for systemic therapy or phototherapy. The expanded indication marks the first time the drug has been available for a pediatric population in the United States.
Children with plaque psoriasis are often undertreated because of fear of side effects of therapies, according to Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco. “Now, more and more medicines are being tested for safety and efficacy in children, and we no longer have to rely on adult studies to inform treatment choices for children,” Dr. Cordoro told this news organization.
The FDA approval of secukinumab for children aged 6 and older with moderate to severe psoriasis “is a welcome addition to the therapeutic toolbox for pediatric psoriasis,” she said. “We’ve entered an era where severe pediatric psoriasis has become a condition that can be adequately controlled with minimal risk and with the convenience of intermittent injections. This has changed the playing field for these children and their families completely. Given the potential short- and long-term negative impact of chronic inflammation on the body of a growing child, we now have approved treatments that can safely offset the risks of undertreated severe psoriasis on the functional and psychological health of the child.”
The approved pediatric dosing for secukinumab is 75 mg or 150 mg depending on the child’s weight at the time of dosing, and it is administered by subcutaneous injection every 4 weeks after an initial loading regimen. According to a press release from Novartis, the FDA approval came on the heels of two phase 3 studies that evaluated the use of secukinumab in children aged 6 to younger than 18 years with plaque psoriasis. The first was a 52-week, randomized, double-blind, placebo- and active-controlled study which included 162 children 6 years of age and older with severe plaque psoriasis. The doses evaluated were 75 mg for children who weighed less than 50 kg and 150 mg for those 50 kg or greater.
At week 12, the Psoriasis Area Severity Index (PASI)-75 response was 55% among children in the 75-mg dosing group vs. 10% in the placebo group and 86% in the 150-mg dosing group vs. 19% in the placebo group.
Meanwhile, the Investigator’s Global Assessment modified 2011 (IGA) “clear” response was achieved in 32% of children in the 75-mg dosing group vs. 5% in the placebo group and in 81% of children in the 150-mg dosing group vs. 5% in the placebo group. An IGA “almost clear” skin response was achieved in 81% of children in the 75-mg dosing group vs. 5% in the placebo group.
The second phase 3 study was a randomized open-label, 208-week trial of 84 subjects 6 years of age and older with moderate to severe plaque psoriasis. According to the Novartis press release, the safety profile reported in both trials was consistent with the safety profile reported in adult plaque psoriasis trials and no new safety signals were observed. The updated prescribing information for secukinumab can be found here.
“When considering treatment with a systemic agent such as a biologic, it is important to consider objective measures of severity, such as extent of disease and involvement of joints but also subjective indicators of severity such as impact beyond the skin on psychological well-being,” Dr. Cordoro said in the interview. “Kids with psoriasis in visible locations may socially isolate themselves due to embarrassment or bullying. Therefore, the impact of moderate to severe psoriasis not only on overall health but on self-esteem and identity formation can be significant, and therefore adequately treating children of all ages to prevent the downstream negative consequences of childhood psoriasis is critical.”
Dr. Cordoro reported having no financial disclosures.
The who are candidates for systemic therapy or phototherapy. The expanded indication marks the first time the drug has been available for a pediatric population in the United States.
Children with plaque psoriasis are often undertreated because of fear of side effects of therapies, according to Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco. “Now, more and more medicines are being tested for safety and efficacy in children, and we no longer have to rely on adult studies to inform treatment choices for children,” Dr. Cordoro told this news organization.
The FDA approval of secukinumab for children aged 6 and older with moderate to severe psoriasis “is a welcome addition to the therapeutic toolbox for pediatric psoriasis,” she said. “We’ve entered an era where severe pediatric psoriasis has become a condition that can be adequately controlled with minimal risk and with the convenience of intermittent injections. This has changed the playing field for these children and their families completely. Given the potential short- and long-term negative impact of chronic inflammation on the body of a growing child, we now have approved treatments that can safely offset the risks of undertreated severe psoriasis on the functional and psychological health of the child.”
The approved pediatric dosing for secukinumab is 75 mg or 150 mg depending on the child’s weight at the time of dosing, and it is administered by subcutaneous injection every 4 weeks after an initial loading regimen. According to a press release from Novartis, the FDA approval came on the heels of two phase 3 studies that evaluated the use of secukinumab in children aged 6 to younger than 18 years with plaque psoriasis. The first was a 52-week, randomized, double-blind, placebo- and active-controlled study which included 162 children 6 years of age and older with severe plaque psoriasis. The doses evaluated were 75 mg for children who weighed less than 50 kg and 150 mg for those 50 kg or greater.
At week 12, the Psoriasis Area Severity Index (PASI)-75 response was 55% among children in the 75-mg dosing group vs. 10% in the placebo group and 86% in the 150-mg dosing group vs. 19% in the placebo group.
Meanwhile, the Investigator’s Global Assessment modified 2011 (IGA) “clear” response was achieved in 32% of children in the 75-mg dosing group vs. 5% in the placebo group and in 81% of children in the 150-mg dosing group vs. 5% in the placebo group. An IGA “almost clear” skin response was achieved in 81% of children in the 75-mg dosing group vs. 5% in the placebo group.
The second phase 3 study was a randomized open-label, 208-week trial of 84 subjects 6 years of age and older with moderate to severe plaque psoriasis. According to the Novartis press release, the safety profile reported in both trials was consistent with the safety profile reported in adult plaque psoriasis trials and no new safety signals were observed. The updated prescribing information for secukinumab can be found here.
“When considering treatment with a systemic agent such as a biologic, it is important to consider objective measures of severity, such as extent of disease and involvement of joints but also subjective indicators of severity such as impact beyond the skin on psychological well-being,” Dr. Cordoro said in the interview. “Kids with psoriasis in visible locations may socially isolate themselves due to embarrassment or bullying. Therefore, the impact of moderate to severe psoriasis not only on overall health but on self-esteem and identity formation can be significant, and therefore adequately treating children of all ages to prevent the downstream negative consequences of childhood psoriasis is critical.”
Dr. Cordoro reported having no financial disclosures.
The who are candidates for systemic therapy or phototherapy. The expanded indication marks the first time the drug has been available for a pediatric population in the United States.
Children with plaque psoriasis are often undertreated because of fear of side effects of therapies, according to Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco. “Now, more and more medicines are being tested for safety and efficacy in children, and we no longer have to rely on adult studies to inform treatment choices for children,” Dr. Cordoro told this news organization.
The FDA approval of secukinumab for children aged 6 and older with moderate to severe psoriasis “is a welcome addition to the therapeutic toolbox for pediatric psoriasis,” she said. “We’ve entered an era where severe pediatric psoriasis has become a condition that can be adequately controlled with minimal risk and with the convenience of intermittent injections. This has changed the playing field for these children and their families completely. Given the potential short- and long-term negative impact of chronic inflammation on the body of a growing child, we now have approved treatments that can safely offset the risks of undertreated severe psoriasis on the functional and psychological health of the child.”
The approved pediatric dosing for secukinumab is 75 mg or 150 mg depending on the child’s weight at the time of dosing, and it is administered by subcutaneous injection every 4 weeks after an initial loading regimen. According to a press release from Novartis, the FDA approval came on the heels of two phase 3 studies that evaluated the use of secukinumab in children aged 6 to younger than 18 years with plaque psoriasis. The first was a 52-week, randomized, double-blind, placebo- and active-controlled study which included 162 children 6 years of age and older with severe plaque psoriasis. The doses evaluated were 75 mg for children who weighed less than 50 kg and 150 mg for those 50 kg or greater.
At week 12, the Psoriasis Area Severity Index (PASI)-75 response was 55% among children in the 75-mg dosing group vs. 10% in the placebo group and 86% in the 150-mg dosing group vs. 19% in the placebo group.
Meanwhile, the Investigator’s Global Assessment modified 2011 (IGA) “clear” response was achieved in 32% of children in the 75-mg dosing group vs. 5% in the placebo group and in 81% of children in the 150-mg dosing group vs. 5% in the placebo group. An IGA “almost clear” skin response was achieved in 81% of children in the 75-mg dosing group vs. 5% in the placebo group.
The second phase 3 study was a randomized open-label, 208-week trial of 84 subjects 6 years of age and older with moderate to severe plaque psoriasis. According to the Novartis press release, the safety profile reported in both trials was consistent with the safety profile reported in adult plaque psoriasis trials and no new safety signals were observed. The updated prescribing information for secukinumab can be found here.
“When considering treatment with a systemic agent such as a biologic, it is important to consider objective measures of severity, such as extent of disease and involvement of joints but also subjective indicators of severity such as impact beyond the skin on psychological well-being,” Dr. Cordoro said in the interview. “Kids with psoriasis in visible locations may socially isolate themselves due to embarrassment or bullying. Therefore, the impact of moderate to severe psoriasis not only on overall health but on self-esteem and identity formation can be significant, and therefore adequately treating children of all ages to prevent the downstream negative consequences of childhood psoriasis is critical.”
Dr. Cordoro reported having no financial disclosures.
Phacomatosis Pigmentokeratotica Associated With Raynaud Phenomenon, Segmental Nevi, Hyperhidrosis, and Scoliosis
To the Editor:
Phacomatosis pigmentokeratotica (PPK) is a rare epidermal nevus syndrome complicated by multiple extracutaneous anomalies, including skeletal defects and neurologic anomalies. Less common associations include lateral curvature of the spine and hyperhidrosis. We present a patient with PPK and unilateral Raynaud phenomenon in addition to a segmental distribution of melanocytic nevi, hyperhidrosis, and scoliosis.
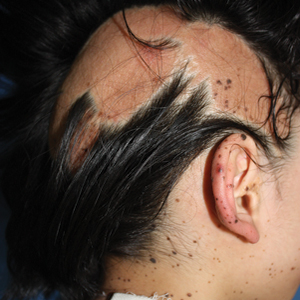
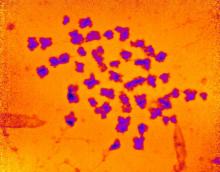
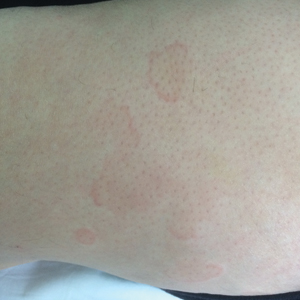
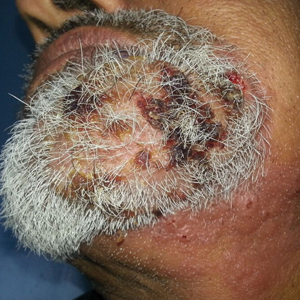
A 9-year-old girl was born with a yellow-orange alopecic plaque on the right side of the scalp (Figure 1). There also were 2 large, irregularly pigmented patches localized on the right side of the upper back and buttock. Over 3 years, numerous papular nevi developed within these pigmented patches and were diagnosed as speckled lentiginous nevi (Figure 2). In addition, numerous nevi of various sizes affected the right face, right shoulder, right arm (Figure 3), and right neck and were clearly demarcated along the midline. Several nevi also were noted within the nevus sebaceous on the right scalp. These skin lesions expanded progressively with age. At 6 years of age, she was diagnosed with hyperhidrosis of the right half of the body, which was most pronounced on the face. Raynaud phenomenon restricted to the right hand also was noted (Figure 4). Upon cold exposure, the digits become pale white, cold, and numb; then blue; and finally red. She lacked other features of connective tissue disease, and autoantibody testing was negative. She also was noted to have an abnormal lateral curvature of the spine (scoliosis). Auditory, ocular, and neurologic examinations were normal. Cranial and cerebral magnetic resonance imaging showed no central nervous system abnormalities. Her family history was negative for nevus spilus, nevus sebaceous, and neurofibromatosis. The clinical findings in our patient led to the diagnosis of PPK.
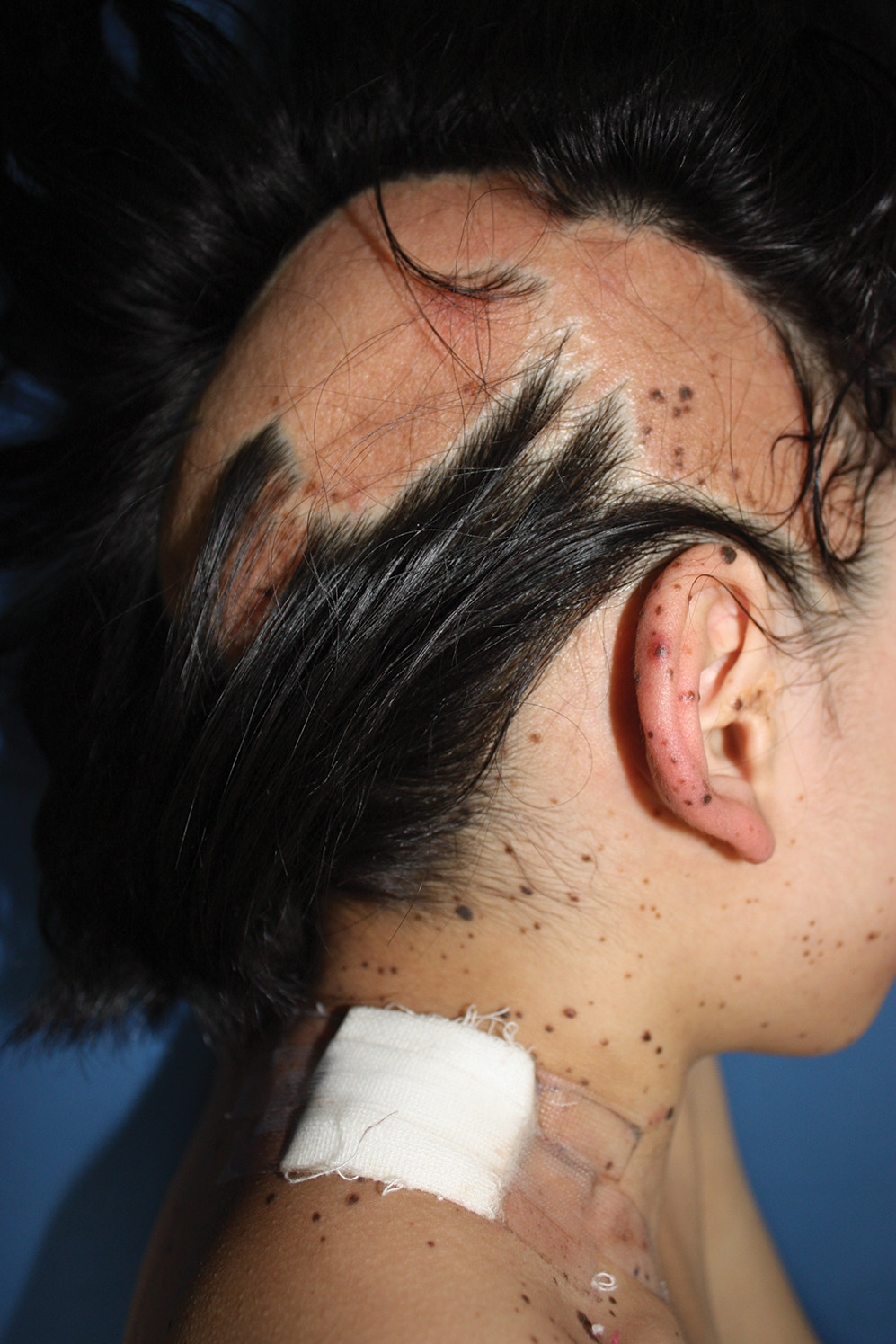
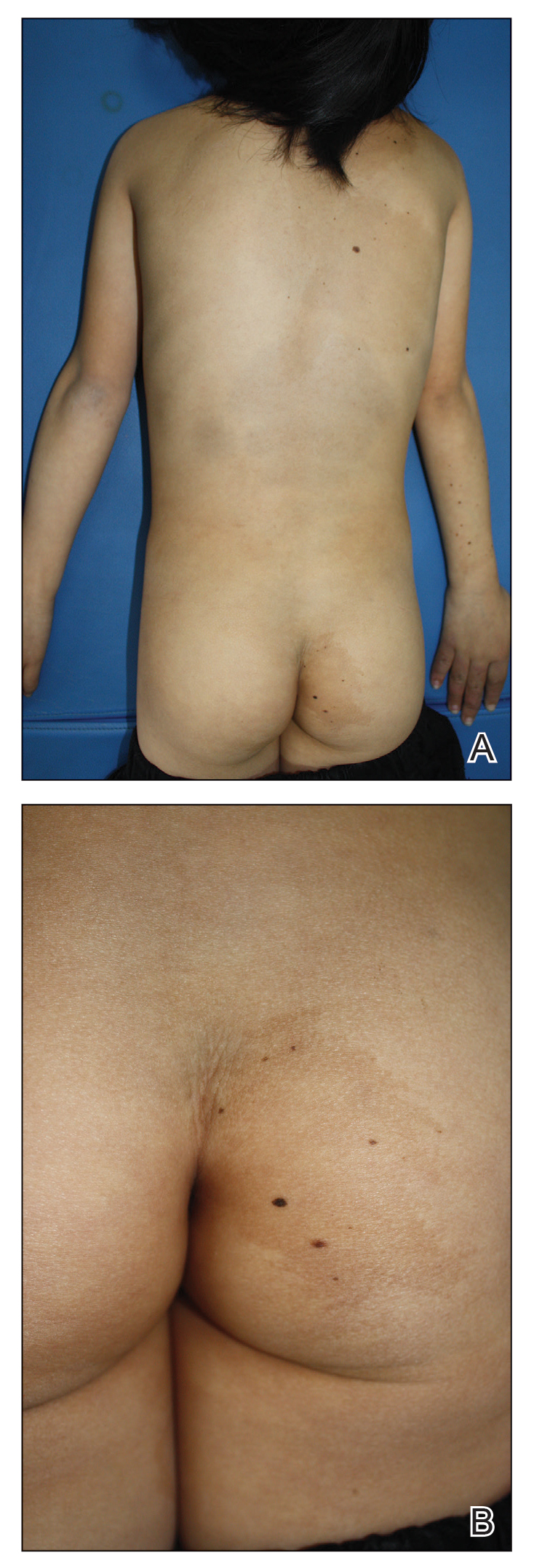
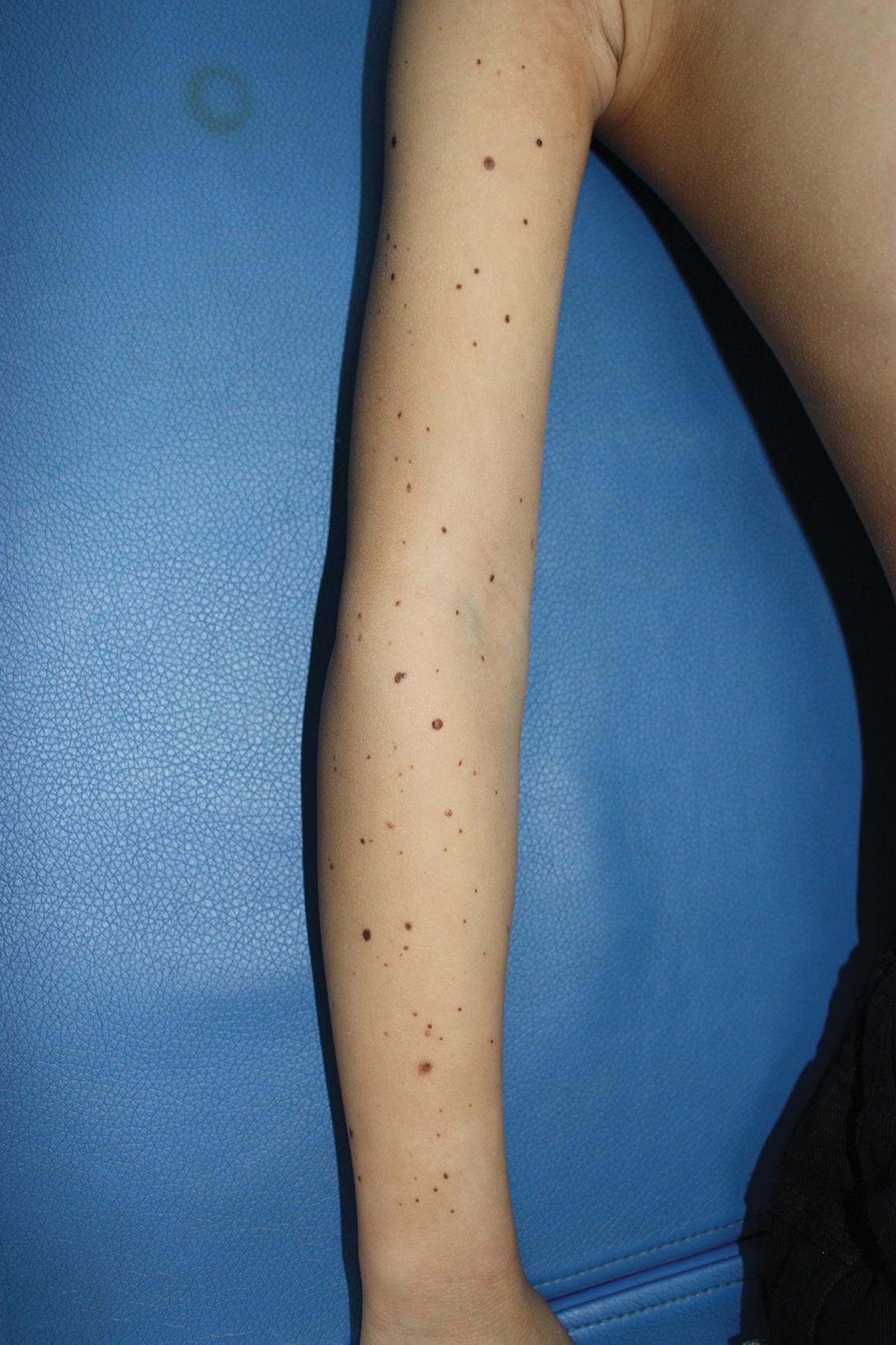
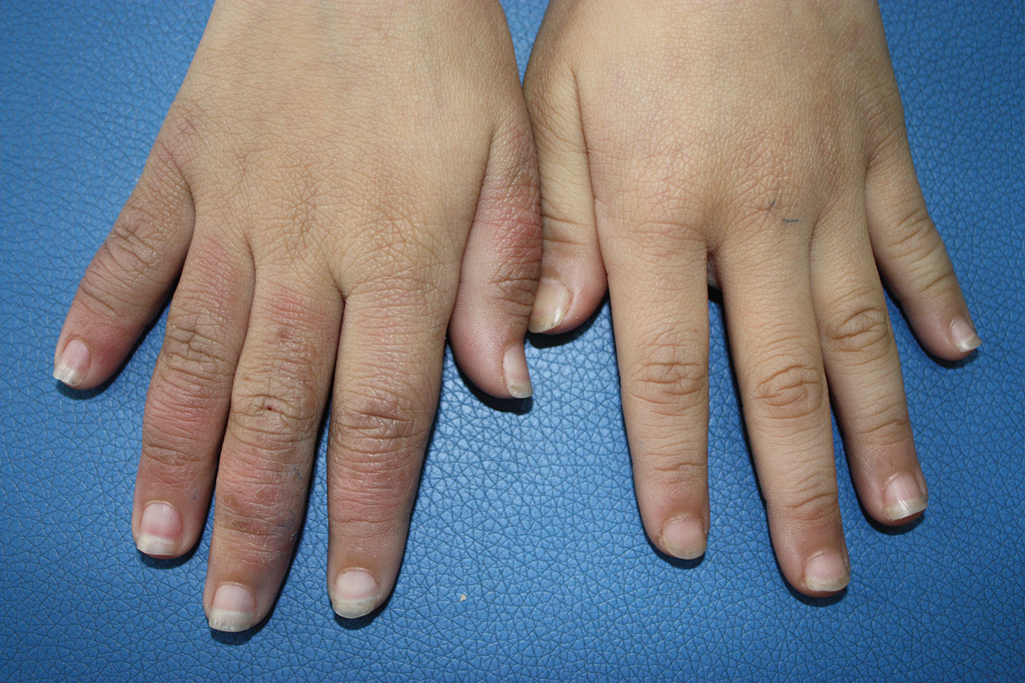
Phacomatosis pigmentokeratotica is a distinctive epidermal nevus syndrome characterized by the coexistence of a speckled lentiginous nevus, also known as a nevus spilus, and a nevus sebaceous1; PPK frequently is complicated by skeletal, ophthalmic, or neurologic abnormalities.2 Most cases reported are sporadic, and a postzygotic mosaic HRas proto-oncogene, GTPase, HRAS, mutation has been demonstrated in some patients and may contribute to the phenotype of PPK.3,4
Other anomalies have included ichthyosislike diffuse hyperkeratosis, laxity of the hands, pelvic hypoplasia, glaucoma, psychomotor retardation, and hypophosphatemic rickets. These patients also should be monitored for the development of malignant neoplasms within the nevus sebaceous.5 Segmental hyperhidrosis may be seen in association with the nevus spilus component.2
Raynaud phenomenon involving only the right hand was a unique finding in our patient. In 3 years of follow-up, our patient developed no evidence of connective tissue disease or other systemic illness. We speculate that Raynaud phenomenon of the right hand along with hyperhidrosis of the right side of the body could be a result of dysfunction of the autonomic nervous system. We propose that Raynaud phenomenon represents an unusual manifestation of PPK and may broaden the spectrum of extracutaneous anomalies associated with the disease. The finding of segmental nevi outside of the confines of the nevus spilus was another unusual manifestation of mosaicism.
- Happle R, Hoffmann R, Restano L, et al. Phacomatosis pigmentokeratotica: a melanocytic-epidermal twin nevus syndrome. Am J Med Genet. 1996;65:363-365.
- Happle R. The group of epidermal nevus syndromes part I. well defined phenotypes. J Am Acad Dermatol. 2010;63:1-22, 23-24.
- Groesser L, Herschberger E, Sagrera A, et al. Phacomatosis pigmentokeratotica is caused by a postzygotic HRAS mutation in a multipotent progenitor cell. J Invest Dermatol. 2013;133:1998-2003.
- Martin RJ, Arefi M, Splitt M, et al. Phacomatosis pigmentokeratotica and precocious puberty associated with HRAS mutation. Br J Dermatol. 2018;178:289-291.
- Chu GY, Wu CY. Phacomatosis pigmentokeratotica: a follow-up report with fatal outcome. Acta Derm Venereol. 2014;94:467-468.
To the Editor:
Phacomatosis pigmentokeratotica (PPK) is a rare epidermal nevus syndrome complicated by multiple extracutaneous anomalies, including skeletal defects and neurologic anomalies. Less common associations include lateral curvature of the spine and hyperhidrosis. We present a patient with PPK and unilateral Raynaud phenomenon in addition to a segmental distribution of melanocytic nevi, hyperhidrosis, and scoliosis.
A 9-year-old girl was born with a yellow-orange alopecic plaque on the right side of the scalp (Figure 1). There also were 2 large, irregularly pigmented patches localized on the right side of the upper back and buttock. Over 3 years, numerous papular nevi developed within these pigmented patches and were diagnosed as speckled lentiginous nevi (Figure 2). In addition, numerous nevi of various sizes affected the right face, right shoulder, right arm (Figure 3), and right neck and were clearly demarcated along the midline. Several nevi also were noted within the nevus sebaceous on the right scalp. These skin lesions expanded progressively with age. At 6 years of age, she was diagnosed with hyperhidrosis of the right half of the body, which was most pronounced on the face. Raynaud phenomenon restricted to the right hand also was noted (Figure 4). Upon cold exposure, the digits become pale white, cold, and numb; then blue; and finally red. She lacked other features of connective tissue disease, and autoantibody testing was negative. She also was noted to have an abnormal lateral curvature of the spine (scoliosis). Auditory, ocular, and neurologic examinations were normal. Cranial and cerebral magnetic resonance imaging showed no central nervous system abnormalities. Her family history was negative for nevus spilus, nevus sebaceous, and neurofibromatosis. The clinical findings in our patient led to the diagnosis of PPK.




Phacomatosis pigmentokeratotica is a distinctive epidermal nevus syndrome characterized by the coexistence of a speckled lentiginous nevus, also known as a nevus spilus, and a nevus sebaceous1; PPK frequently is complicated by skeletal, ophthalmic, or neurologic abnormalities.2 Most cases reported are sporadic, and a postzygotic mosaic HRas proto-oncogene, GTPase, HRAS, mutation has been demonstrated in some patients and may contribute to the phenotype of PPK.3,4
Other anomalies have included ichthyosislike diffuse hyperkeratosis, laxity of the hands, pelvic hypoplasia, glaucoma, psychomotor retardation, and hypophosphatemic rickets. These patients also should be monitored for the development of malignant neoplasms within the nevus sebaceous.5 Segmental hyperhidrosis may be seen in association with the nevus spilus component.2
Raynaud phenomenon involving only the right hand was a unique finding in our patient. In 3 years of follow-up, our patient developed no evidence of connective tissue disease or other systemic illness. We speculate that Raynaud phenomenon of the right hand along with hyperhidrosis of the right side of the body could be a result of dysfunction of the autonomic nervous system. We propose that Raynaud phenomenon represents an unusual manifestation of PPK and may broaden the spectrum of extracutaneous anomalies associated with the disease. The finding of segmental nevi outside of the confines of the nevus spilus was another unusual manifestation of mosaicism.
To the Editor:
Phacomatosis pigmentokeratotica (PPK) is a rare epidermal nevus syndrome complicated by multiple extracutaneous anomalies, including skeletal defects and neurologic anomalies. Less common associations include lateral curvature of the spine and hyperhidrosis. We present a patient with PPK and unilateral Raynaud phenomenon in addition to a segmental distribution of melanocytic nevi, hyperhidrosis, and scoliosis.
A 9-year-old girl was born with a yellow-orange alopecic plaque on the right side of the scalp (Figure 1). There also were 2 large, irregularly pigmented patches localized on the right side of the upper back and buttock. Over 3 years, numerous papular nevi developed within these pigmented patches and were diagnosed as speckled lentiginous nevi (Figure 2). In addition, numerous nevi of various sizes affected the right face, right shoulder, right arm (Figure 3), and right neck and were clearly demarcated along the midline. Several nevi also were noted within the nevus sebaceous on the right scalp. These skin lesions expanded progressively with age. At 6 years of age, she was diagnosed with hyperhidrosis of the right half of the body, which was most pronounced on the face. Raynaud phenomenon restricted to the right hand also was noted (Figure 4). Upon cold exposure, the digits become pale white, cold, and numb; then blue; and finally red. She lacked other features of connective tissue disease, and autoantibody testing was negative. She also was noted to have an abnormal lateral curvature of the spine (scoliosis). Auditory, ocular, and neurologic examinations were normal. Cranial and cerebral magnetic resonance imaging showed no central nervous system abnormalities. Her family history was negative for nevus spilus, nevus sebaceous, and neurofibromatosis. The clinical findings in our patient led to the diagnosis of PPK.




Phacomatosis pigmentokeratotica is a distinctive epidermal nevus syndrome characterized by the coexistence of a speckled lentiginous nevus, also known as a nevus spilus, and a nevus sebaceous1; PPK frequently is complicated by skeletal, ophthalmic, or neurologic abnormalities.2 Most cases reported are sporadic, and a postzygotic mosaic HRas proto-oncogene, GTPase, HRAS, mutation has been demonstrated in some patients and may contribute to the phenotype of PPK.3,4
Other anomalies have included ichthyosislike diffuse hyperkeratosis, laxity of the hands, pelvic hypoplasia, glaucoma, psychomotor retardation, and hypophosphatemic rickets. These patients also should be monitored for the development of malignant neoplasms within the nevus sebaceous.5 Segmental hyperhidrosis may be seen in association with the nevus spilus component.2
Raynaud phenomenon involving only the right hand was a unique finding in our patient. In 3 years of follow-up, our patient developed no evidence of connective tissue disease or other systemic illness. We speculate that Raynaud phenomenon of the right hand along with hyperhidrosis of the right side of the body could be a result of dysfunction of the autonomic nervous system. We propose that Raynaud phenomenon represents an unusual manifestation of PPK and may broaden the spectrum of extracutaneous anomalies associated with the disease. The finding of segmental nevi outside of the confines of the nevus spilus was another unusual manifestation of mosaicism.
- Happle R, Hoffmann R, Restano L, et al. Phacomatosis pigmentokeratotica: a melanocytic-epidermal twin nevus syndrome. Am J Med Genet. 1996;65:363-365.
- Happle R. The group of epidermal nevus syndromes part I. well defined phenotypes. J Am Acad Dermatol. 2010;63:1-22, 23-24.
- Groesser L, Herschberger E, Sagrera A, et al. Phacomatosis pigmentokeratotica is caused by a postzygotic HRAS mutation in a multipotent progenitor cell. J Invest Dermatol. 2013;133:1998-2003.
- Martin RJ, Arefi M, Splitt M, et al. Phacomatosis pigmentokeratotica and precocious puberty associated with HRAS mutation. Br J Dermatol. 2018;178:289-291.
- Chu GY, Wu CY. Phacomatosis pigmentokeratotica: a follow-up report with fatal outcome. Acta Derm Venereol. 2014;94:467-468.
- Happle R, Hoffmann R, Restano L, et al. Phacomatosis pigmentokeratotica: a melanocytic-epidermal twin nevus syndrome. Am J Med Genet. 1996;65:363-365.
- Happle R. The group of epidermal nevus syndromes part I. well defined phenotypes. J Am Acad Dermatol. 2010;63:1-22, 23-24.
- Groesser L, Herschberger E, Sagrera A, et al. Phacomatosis pigmentokeratotica is caused by a postzygotic HRAS mutation in a multipotent progenitor cell. J Invest Dermatol. 2013;133:1998-2003.
- Martin RJ, Arefi M, Splitt M, et al. Phacomatosis pigmentokeratotica and precocious puberty associated with HRAS mutation. Br J Dermatol. 2018;178:289-291.
- Chu GY, Wu CY. Phacomatosis pigmentokeratotica: a follow-up report with fatal outcome. Acta Derm Venereol. 2014;94:467-468.
Practice Points
- Phacomatosis pigmentokeratotica (PPK) is characterized by the coexistence of speckled lentiginous nevus and nevus sebaceous.
- Raynaud phenomenon may be an unreported association with PPK.
The Power of a Multidisciplinary Tumor Board: Managing Unresectable and/or High-Risk Skin Cancers
Multidisciplinary tumor boards are composed of providers from many fields who deliver coordinated care for patients with unresectable and high-risk skin cancers. Providers who comprise the tumor board often are radiation oncologists, hematologists/oncologists, general surgeons, dermatologists, dermatologic surgeons, and pathologists. The benefit of having a tumor board is that each patient is evaluated simultaneously by a group of physicians from various specialties who bring diverse perspectives that will contribute to the overall treatment plan. The cases often encompass high-risk tumors including unresectable basal cell carcinomas or invasive melanomas. By combining knowledge from each specialty in a team approach, the tumor board can effectively and holistically develop a care plan for each patient.
For the tumor board at the Warren Alpert Medical School of Brown University (Providence, Rhode Island), we often prepare a presentation with comprehensive details about the patient and tumor. During the presentation, we also propose a treatment plan prior to describing each patient at the weekly conference and amend the plans during the discussion. Tumor boards also provide a consulting role to the community and hospital providers in which patients are being referred by their primary provider and are seeking a second opinion or guidance.
In many ways, the tumor board is a multidisciplinary approach for patient advocacy in the form of treatment. These physicians meet on a regular basis to check on the patient’s progress and continually reevaluate how to have discussions about the patient’s care. There are many reasons why it is important to refer patients to a multidisciplinary tumor board.
Improved Workup and Diagnosis
One of the values of a tumor board is that it allows for patient data to be collected and assembled in a way that tells a story. The specialist from each field can then discuss and weigh the benefits and risks for each diagnostic test that should be performed for the workup in each patient. Physicians who refer their patients to the tumor board use their recommendations to both confirm the diagnosis and shift their treatment plans, depending on the information presented during the meeting.1 There may be a change in the tumor type, decision to refer for surgery, cancer staging, and list of viable options, especially after reviewing pathology and imaging.2 The discussion of the treatment plan may consider not only surgical considerations but also the patient’s quality of life. At times, noninvasive interventions are more appropriate and align with the patient’s goals of care. In addition, during the tumor board clinic there may be new tumors that are identified and biopsied, providing increased diagnosis and surveillance for patients who may have a higher risk for developing skin cancer.
Education for Residents and Providers
The multidisciplinary tumor board not only helps patients but also educates both residents and providers on the evidence-based therapeutic management of high-risk tumors.2 Research literature on cutaneous oncology is dynamic, and the weekly tumor board meetings help providers stay informed about the best and most effective treatments for their patients.3 In addition to the attending specialists, participants of the tumor board also may include residents, medical students, medical assistance staff, nurses, physician assistants, and fellows. Furthermore, the recommendations given by the tumor board serve to educate both the patient and the provider who referred them to the tumor board. Although we have access to excellent dermatology textbooks as residents, the most impactful educational experience is seeing the patients in tumor board clinic and participating in the immensely educational discussions at the weekly conferences. Through this experience, I have learned that treatment plans should be personalized to the patient. There are many factors to take into consideration when deciphering what the best course of treatment will be for a patient. Sometimes the best option is Mohs micrographic surgery, while other times it may be scheduling several sessions of palliative radiation oncology. Treatment depends on the individual patient and their condition.
Coordination of Care
During a week that I was on call, I was consulted to biopsy a patient with a giant hemorrhagic basal cell carcinoma that caused substantial cheek and nose distortion as well as anemia secondary to acute blood loss. The patient not only did not have a dermatologist but also did not have a primary care physician given he had not had contact with the health care system in more than 30 years. The reason for him not seeking care was multifactorial, but the approach to his care became multidisciplinary. We sought to connect him with the right providers to help him in any way that we could. We presented him at our multidisciplinary tumor board and started him on sonedigib, a medication that binds to and inhibits the smoothened protein.4 Through the tumor board, we were able to establish sustained contact with the patient. The tumor board created effective communication between providers to get him the referrals that he needed for dermatology, pathology, radiation oncology, hematology/oncology, and otolaryngology. The discussions centered around being cognizant of the patient’s apprehension with the health care system as well as providing medical and surgical treatment that would help his quality of life. We built a consensus on what the best plan was for the patient and his family. This consensus would have been more difficult had it not been for the combined specialties of the tumor board. In general, studies have shown that weekly tumor boards have resulted in decreased mortality rates for patients with advanced cancers.5
Final Thoughts
The multidisciplinary tumor board is a powerful resource for hospitals and the greater medical community. At these weekly conferences you realize there may still be hope that begins at the line where your expertise ends. It represents a team of providers who compassionately refuse to give up on patients when they are the last refuge.
- Foster TJ, Bouchard-Fortier A, Olivotto IA, et al. Effect of multidisciplinary case conferences on physician decision making: breast diagnostic rounds. Cureus. 2016;8:E895.
- El Saghir NS, Charara RN, Kreidieh FY, et al. Global practice and efficiency of multidisciplinary tumor boards: results of an American Society of Clinical Oncology international survey. J Glob Oncol. 2015;1:57-64.
- Mori S, Navarrete-Dechent C, Petukhova TA, et al. Tumor board conferences for multidisciplinary skin cancer management: a survey of US cancer centers. J Natl Compr Canc Netw. 2018;16:1209-1215.
- Dummer R, Ascierto PA, Basset-Seguin N, et al. Sonidegib and vismodegib in the treatment of patients with locally advanced basal cell carcinoma: a joint expert opinion. J Eur Acad Dermatol Venereol. 2020;34:1944-1956.
- Kehl KL, Landrum MB, Kahn KL, et al. Tumor board participation among physicians caring for patients with lung or colorectal cancer. J Oncol Pract. 2015;11:E267-E278.
Multidisciplinary tumor boards are composed of providers from many fields who deliver coordinated care for patients with unresectable and high-risk skin cancers. Providers who comprise the tumor board often are radiation oncologists, hematologists/oncologists, general surgeons, dermatologists, dermatologic surgeons, and pathologists. The benefit of having a tumor board is that each patient is evaluated simultaneously by a group of physicians from various specialties who bring diverse perspectives that will contribute to the overall treatment plan. The cases often encompass high-risk tumors including unresectable basal cell carcinomas or invasive melanomas. By combining knowledge from each specialty in a team approach, the tumor board can effectively and holistically develop a care plan for each patient.
For the tumor board at the Warren Alpert Medical School of Brown University (Providence, Rhode Island), we often prepare a presentation with comprehensive details about the patient and tumor. During the presentation, we also propose a treatment plan prior to describing each patient at the weekly conference and amend the plans during the discussion. Tumor boards also provide a consulting role to the community and hospital providers in which patients are being referred by their primary provider and are seeking a second opinion or guidance.
In many ways, the tumor board is a multidisciplinary approach for patient advocacy in the form of treatment. These physicians meet on a regular basis to check on the patient’s progress and continually reevaluate how to have discussions about the patient’s care. There are many reasons why it is important to refer patients to a multidisciplinary tumor board.
Improved Workup and Diagnosis
One of the values of a tumor board is that it allows for patient data to be collected and assembled in a way that tells a story. The specialist from each field can then discuss and weigh the benefits and risks for each diagnostic test that should be performed for the workup in each patient. Physicians who refer their patients to the tumor board use their recommendations to both confirm the diagnosis and shift their treatment plans, depending on the information presented during the meeting.1 There may be a change in the tumor type, decision to refer for surgery, cancer staging, and list of viable options, especially after reviewing pathology and imaging.2 The discussion of the treatment plan may consider not only surgical considerations but also the patient’s quality of life. At times, noninvasive interventions are more appropriate and align with the patient’s goals of care. In addition, during the tumor board clinic there may be new tumors that are identified and biopsied, providing increased diagnosis and surveillance for patients who may have a higher risk for developing skin cancer.
Education for Residents and Providers
The multidisciplinary tumor board not only helps patients but also educates both residents and providers on the evidence-based therapeutic management of high-risk tumors.2 Research literature on cutaneous oncology is dynamic, and the weekly tumor board meetings help providers stay informed about the best and most effective treatments for their patients.3 In addition to the attending specialists, participants of the tumor board also may include residents, medical students, medical assistance staff, nurses, physician assistants, and fellows. Furthermore, the recommendations given by the tumor board serve to educate both the patient and the provider who referred them to the tumor board. Although we have access to excellent dermatology textbooks as residents, the most impactful educational experience is seeing the patients in tumor board clinic and participating in the immensely educational discussions at the weekly conferences. Through this experience, I have learned that treatment plans should be personalized to the patient. There are many factors to take into consideration when deciphering what the best course of treatment will be for a patient. Sometimes the best option is Mohs micrographic surgery, while other times it may be scheduling several sessions of palliative radiation oncology. Treatment depends on the individual patient and their condition.
Coordination of Care
During a week that I was on call, I was consulted to biopsy a patient with a giant hemorrhagic basal cell carcinoma that caused substantial cheek and nose distortion as well as anemia secondary to acute blood loss. The patient not only did not have a dermatologist but also did not have a primary care physician given he had not had contact with the health care system in more than 30 years. The reason for him not seeking care was multifactorial, but the approach to his care became multidisciplinary. We sought to connect him with the right providers to help him in any way that we could. We presented him at our multidisciplinary tumor board and started him on sonedigib, a medication that binds to and inhibits the smoothened protein.4 Through the tumor board, we were able to establish sustained contact with the patient. The tumor board created effective communication between providers to get him the referrals that he needed for dermatology, pathology, radiation oncology, hematology/oncology, and otolaryngology. The discussions centered around being cognizant of the patient’s apprehension with the health care system as well as providing medical and surgical treatment that would help his quality of life. We built a consensus on what the best plan was for the patient and his family. This consensus would have been more difficult had it not been for the combined specialties of the tumor board. In general, studies have shown that weekly tumor boards have resulted in decreased mortality rates for patients with advanced cancers.5
Final Thoughts
The multidisciplinary tumor board is a powerful resource for hospitals and the greater medical community. At these weekly conferences you realize there may still be hope that begins at the line where your expertise ends. It represents a team of providers who compassionately refuse to give up on patients when they are the last refuge.
Multidisciplinary tumor boards are composed of providers from many fields who deliver coordinated care for patients with unresectable and high-risk skin cancers. Providers who comprise the tumor board often are radiation oncologists, hematologists/oncologists, general surgeons, dermatologists, dermatologic surgeons, and pathologists. The benefit of having a tumor board is that each patient is evaluated simultaneously by a group of physicians from various specialties who bring diverse perspectives that will contribute to the overall treatment plan. The cases often encompass high-risk tumors including unresectable basal cell carcinomas or invasive melanomas. By combining knowledge from each specialty in a team approach, the tumor board can effectively and holistically develop a care plan for each patient.
For the tumor board at the Warren Alpert Medical School of Brown University (Providence, Rhode Island), we often prepare a presentation with comprehensive details about the patient and tumor. During the presentation, we also propose a treatment plan prior to describing each patient at the weekly conference and amend the plans during the discussion. Tumor boards also provide a consulting role to the community and hospital providers in which patients are being referred by their primary provider and are seeking a second opinion or guidance.
In many ways, the tumor board is a multidisciplinary approach for patient advocacy in the form of treatment. These physicians meet on a regular basis to check on the patient’s progress and continually reevaluate how to have discussions about the patient’s care. There are many reasons why it is important to refer patients to a multidisciplinary tumor board.
Improved Workup and Diagnosis
One of the values of a tumor board is that it allows for patient data to be collected and assembled in a way that tells a story. The specialist from each field can then discuss and weigh the benefits and risks for each diagnostic test that should be performed for the workup in each patient. Physicians who refer their patients to the tumor board use their recommendations to both confirm the diagnosis and shift their treatment plans, depending on the information presented during the meeting.1 There may be a change in the tumor type, decision to refer for surgery, cancer staging, and list of viable options, especially after reviewing pathology and imaging.2 The discussion of the treatment plan may consider not only surgical considerations but also the patient’s quality of life. At times, noninvasive interventions are more appropriate and align with the patient’s goals of care. In addition, during the tumor board clinic there may be new tumors that are identified and biopsied, providing increased diagnosis and surveillance for patients who may have a higher risk for developing skin cancer.
Education for Residents and Providers
The multidisciplinary tumor board not only helps patients but also educates both residents and providers on the evidence-based therapeutic management of high-risk tumors.2 Research literature on cutaneous oncology is dynamic, and the weekly tumor board meetings help providers stay informed about the best and most effective treatments for their patients.3 In addition to the attending specialists, participants of the tumor board also may include residents, medical students, medical assistance staff, nurses, physician assistants, and fellows. Furthermore, the recommendations given by the tumor board serve to educate both the patient and the provider who referred them to the tumor board. Although we have access to excellent dermatology textbooks as residents, the most impactful educational experience is seeing the patients in tumor board clinic and participating in the immensely educational discussions at the weekly conferences. Through this experience, I have learned that treatment plans should be personalized to the patient. There are many factors to take into consideration when deciphering what the best course of treatment will be for a patient. Sometimes the best option is Mohs micrographic surgery, while other times it may be scheduling several sessions of palliative radiation oncology. Treatment depends on the individual patient and their condition.
Coordination of Care
During a week that I was on call, I was consulted to biopsy a patient with a giant hemorrhagic basal cell carcinoma that caused substantial cheek and nose distortion as well as anemia secondary to acute blood loss. The patient not only did not have a dermatologist but also did not have a primary care physician given he had not had contact with the health care system in more than 30 years. The reason for him not seeking care was multifactorial, but the approach to his care became multidisciplinary. We sought to connect him with the right providers to help him in any way that we could. We presented him at our multidisciplinary tumor board and started him on sonedigib, a medication that binds to and inhibits the smoothened protein.4 Through the tumor board, we were able to establish sustained contact with the patient. The tumor board created effective communication between providers to get him the referrals that he needed for dermatology, pathology, radiation oncology, hematology/oncology, and otolaryngology. The discussions centered around being cognizant of the patient’s apprehension with the health care system as well as providing medical and surgical treatment that would help his quality of life. We built a consensus on what the best plan was for the patient and his family. This consensus would have been more difficult had it not been for the combined specialties of the tumor board. In general, studies have shown that weekly tumor boards have resulted in decreased mortality rates for patients with advanced cancers.5
Final Thoughts
The multidisciplinary tumor board is a powerful resource for hospitals and the greater medical community. At these weekly conferences you realize there may still be hope that begins at the line where your expertise ends. It represents a team of providers who compassionately refuse to give up on patients when they are the last refuge.
- Foster TJ, Bouchard-Fortier A, Olivotto IA, et al. Effect of multidisciplinary case conferences on physician decision making: breast diagnostic rounds. Cureus. 2016;8:E895.
- El Saghir NS, Charara RN, Kreidieh FY, et al. Global practice and efficiency of multidisciplinary tumor boards: results of an American Society of Clinical Oncology international survey. J Glob Oncol. 2015;1:57-64.
- Mori S, Navarrete-Dechent C, Petukhova TA, et al. Tumor board conferences for multidisciplinary skin cancer management: a survey of US cancer centers. J Natl Compr Canc Netw. 2018;16:1209-1215.
- Dummer R, Ascierto PA, Basset-Seguin N, et al. Sonidegib and vismodegib in the treatment of patients with locally advanced basal cell carcinoma: a joint expert opinion. J Eur Acad Dermatol Venereol. 2020;34:1944-1956.
- Kehl KL, Landrum MB, Kahn KL, et al. Tumor board participation among physicians caring for patients with lung or colorectal cancer. J Oncol Pract. 2015;11:E267-E278.
- Foster TJ, Bouchard-Fortier A, Olivotto IA, et al. Effect of multidisciplinary case conferences on physician decision making: breast diagnostic rounds. Cureus. 2016;8:E895.
- El Saghir NS, Charara RN, Kreidieh FY, et al. Global practice and efficiency of multidisciplinary tumor boards: results of an American Society of Clinical Oncology international survey. J Glob Oncol. 2015;1:57-64.
- Mori S, Navarrete-Dechent C, Petukhova TA, et al. Tumor board conferences for multidisciplinary skin cancer management: a survey of US cancer centers. J Natl Compr Canc Netw. 2018;16:1209-1215.
- Dummer R, Ascierto PA, Basset-Seguin N, et al. Sonidegib and vismodegib in the treatment of patients with locally advanced basal cell carcinoma: a joint expert opinion. J Eur Acad Dermatol Venereol. 2020;34:1944-1956.
- Kehl KL, Landrum MB, Kahn KL, et al. Tumor board participation among physicians caring for patients with lung or colorectal cancer. J Oncol Pract. 2015;11:E267-E278.
Resident Pearl
- Participating in a multidisciplinary tumor board allows residents to learn more about how to manage and treat high-risk skin cancers. The multidisciplinary team approach provides high-quality care for challenging patients.
Mohs surgery favorable as monotherapy for early Merkel cell carcinomas
Pittsburgh.
The results compare favorably with the standard treatment approach, wide local excision with or without radiation, which has a local recurrence rate of 4.2%-31.7% because of incomplete excision or false negative margins, said Vitaly Terushkin, MD, a Mohs surgeon who presented the findings of the study, a retrospective chart review, at the annual meeting of the American College of Mohs Surgery.
Mohs surgery as monotherapy offered “survival at least as good as historical controls treated with wide local excision plus radiation therapy, and because of the superior local control, Mohs surgery may obviate the need for adjuvant radiation and decrease the chance for additional surgery for the treatment of local recurrence,” said Dr. Terushkin, now in practice in the New York City area.
“We hope this data fuel additional studies with larger cohorts to continue to explore the value of Mohs for Merkel cell carcinoma,” he said.
The findings add to a growing body of literature supporting Mohs for many types of rare tumors. “Micrographic surgery or complete circumferential peripheral and deep margin analysis has been shown to be superior to wide local excision in a variety of tumors and clinical scenarios,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington.
“When the entire margin is able to be evaluated over random bread-loafed sections, there is growing evidence that this leads to superior outcomes and disease specific mortality,” he said when asked for comment on the study results.
In all, 56 primary Merkel cell carcinomas were treated in the 53 patients from 2001 to 2019; about two-thirds of the patients had stage 1 tumors and the rest stage 2a.
They were treated with Mohs alone, without radiation. Average follow up was 4.6 years, with about a third of patients followed for 5 or more years.
The average age of the patients was 78 years, and just over half were men. In more than half the cases, tumors were located on the head and neck (62.5%), and the mean tumor size was 1.7 cm. Patients were negative for lymphadenopathy and declined lymph node biopsy.
Although there was no local recurrence, defined as tumor reemerging within or adjacent to the surgery site, 7 patients (12.7%) developed in-transit metastases, 13 (23.6%) developed nodal metastases, and 3 developed distant metastases.
The 5-year disease-specific survival rate was 91.2% for stage 1 and 68.6% for stage 2a patients, which compared favorably with historical controls treated with wide local excision with or without radiation, with reported 5-year disease-specific survival rates of 81%-87% for stage 1 disease and 63%-67% for stage 2. Although radiation wasn’t used in the study, Dr. Patel noted that more investigation is needed about the role of adjuvant radiation therapy after Mohs surgery “given recent publications showing improved outcomes in patients with narrow margin excision and postoperative radiation therapy.”
No external funding of the study was reported. Dr. Terushkin had no disclosures. Dr. Patel is a consultant for Sanofi, Regeneron, and Almirall.
Pittsburgh.
The results compare favorably with the standard treatment approach, wide local excision with or without radiation, which has a local recurrence rate of 4.2%-31.7% because of incomplete excision or false negative margins, said Vitaly Terushkin, MD, a Mohs surgeon who presented the findings of the study, a retrospective chart review, at the annual meeting of the American College of Mohs Surgery.
Mohs surgery as monotherapy offered “survival at least as good as historical controls treated with wide local excision plus radiation therapy, and because of the superior local control, Mohs surgery may obviate the need for adjuvant radiation and decrease the chance for additional surgery for the treatment of local recurrence,” said Dr. Terushkin, now in practice in the New York City area.
“We hope this data fuel additional studies with larger cohorts to continue to explore the value of Mohs for Merkel cell carcinoma,” he said.
The findings add to a growing body of literature supporting Mohs for many types of rare tumors. “Micrographic surgery or complete circumferential peripheral and deep margin analysis has been shown to be superior to wide local excision in a variety of tumors and clinical scenarios,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington.
“When the entire margin is able to be evaluated over random bread-loafed sections, there is growing evidence that this leads to superior outcomes and disease specific mortality,” he said when asked for comment on the study results.
In all, 56 primary Merkel cell carcinomas were treated in the 53 patients from 2001 to 2019; about two-thirds of the patients had stage 1 tumors and the rest stage 2a.
They were treated with Mohs alone, without radiation. Average follow up was 4.6 years, with about a third of patients followed for 5 or more years.
The average age of the patients was 78 years, and just over half were men. In more than half the cases, tumors were located on the head and neck (62.5%), and the mean tumor size was 1.7 cm. Patients were negative for lymphadenopathy and declined lymph node biopsy.
Although there was no local recurrence, defined as tumor reemerging within or adjacent to the surgery site, 7 patients (12.7%) developed in-transit metastases, 13 (23.6%) developed nodal metastases, and 3 developed distant metastases.
The 5-year disease-specific survival rate was 91.2% for stage 1 and 68.6% for stage 2a patients, which compared favorably with historical controls treated with wide local excision with or without radiation, with reported 5-year disease-specific survival rates of 81%-87% for stage 1 disease and 63%-67% for stage 2. Although radiation wasn’t used in the study, Dr. Patel noted that more investigation is needed about the role of adjuvant radiation therapy after Mohs surgery “given recent publications showing improved outcomes in patients with narrow margin excision and postoperative radiation therapy.”
No external funding of the study was reported. Dr. Terushkin had no disclosures. Dr. Patel is a consultant for Sanofi, Regeneron, and Almirall.
Pittsburgh.
The results compare favorably with the standard treatment approach, wide local excision with or without radiation, which has a local recurrence rate of 4.2%-31.7% because of incomplete excision or false negative margins, said Vitaly Terushkin, MD, a Mohs surgeon who presented the findings of the study, a retrospective chart review, at the annual meeting of the American College of Mohs Surgery.
Mohs surgery as monotherapy offered “survival at least as good as historical controls treated with wide local excision plus radiation therapy, and because of the superior local control, Mohs surgery may obviate the need for adjuvant radiation and decrease the chance for additional surgery for the treatment of local recurrence,” said Dr. Terushkin, now in practice in the New York City area.
“We hope this data fuel additional studies with larger cohorts to continue to explore the value of Mohs for Merkel cell carcinoma,” he said.
The findings add to a growing body of literature supporting Mohs for many types of rare tumors. “Micrographic surgery or complete circumferential peripheral and deep margin analysis has been shown to be superior to wide local excision in a variety of tumors and clinical scenarios,” said Vishal Patel, MD, assistant professor of dermatology and director of the cutaneous oncology program at George Washington University, Washington.
“When the entire margin is able to be evaluated over random bread-loafed sections, there is growing evidence that this leads to superior outcomes and disease specific mortality,” he said when asked for comment on the study results.
In all, 56 primary Merkel cell carcinomas were treated in the 53 patients from 2001 to 2019; about two-thirds of the patients had stage 1 tumors and the rest stage 2a.
They were treated with Mohs alone, without radiation. Average follow up was 4.6 years, with about a third of patients followed for 5 or more years.
The average age of the patients was 78 years, and just over half were men. In more than half the cases, tumors were located on the head and neck (62.5%), and the mean tumor size was 1.7 cm. Patients were negative for lymphadenopathy and declined lymph node biopsy.
Although there was no local recurrence, defined as tumor reemerging within or adjacent to the surgery site, 7 patients (12.7%) developed in-transit metastases, 13 (23.6%) developed nodal metastases, and 3 developed distant metastases.
The 5-year disease-specific survival rate was 91.2% for stage 1 and 68.6% for stage 2a patients, which compared favorably with historical controls treated with wide local excision with or without radiation, with reported 5-year disease-specific survival rates of 81%-87% for stage 1 disease and 63%-67% for stage 2. Although radiation wasn’t used in the study, Dr. Patel noted that more investigation is needed about the role of adjuvant radiation therapy after Mohs surgery “given recent publications showing improved outcomes in patients with narrow margin excision and postoperative radiation therapy.”
No external funding of the study was reported. Dr. Terushkin had no disclosures. Dr. Patel is a consultant for Sanofi, Regeneron, and Almirall.
FROM ACMS 2021
Noses can be electronic, and toilets can be smart
Cancer loses … by a nose
Since the human nose is unpredictable at best, we’ve learned to rely on animals for our detailed nozzle needs. But researchers have found the next best thing to man’s best friend to accurately identify cancers.
A team at the University of Pennsylvania has developed an electronic olfaction, or “e-nose,” that has a 95% accuracy rate in distinguishing benign and malignant pancreatic and ovarian cancer cells from a single blood sample. How?
The e-nose system is equipped with nanosensors that are able to detect the volatile organic compounds (VOCs) emitted by cells in a blood sample. Not only does this create an opportunity for an easier, noninvasive screening practice, but it’s fast. The e-nose can distinguish VOCs from healthy to cancerous blood cells in 20 minutes or less and is just as effective in picking up on early- and late-stage cancers.
The investigators hope that this innovative technology can pave the way for similar devices with other uses. Thanks to the e-nose, a handheld device is in development that may be able to sniff out the signature odor of people with COVID-19.
That’s one smart schnoz.
Do you think this is a (food) game?
Dieting and eating healthy is tough, even during the best of times, and it has not been the best of times. With all respect to Charles Dickens, it’s been the worst of times, full stop. Millions of people have spent the past year sitting around their homes doing nothing, and it’s only natural that many would let their discipline slide.
Naturally, the solution to unhealthy eating habits is to sit down and play with your phone. No, that’s not the joke, the Food Trainer app, available on all cellular devices near you, is designed to encourage healthy eating by turning it into a game of sorts. When users open the app, they’re presented with images of food, and they’re trained to tap on images of healthy food and pass on images of unhealthy ones. The process takes less than 5 minutes.
It sounds really simple, but in a study of more than 1,000 people, consumption of junk food fell by 1 point on an 8-point scale (ranging from four times per day to zero to one time per month), participants lost about half a kilogram (a little over one pound), and more healthy food was eaten. Those who used the app more regularly, along the lines of 10 times per month or more, saw greater benefits.
The authors did acknowledge that those who used the app more may have been more motivated to lose weight anyway, which perhaps limits the overall benefit, but reviews on Google Play were overall quite positive, and if there’s one great truth in this world, it’s that Internet reviewers are almost impossible to please. So perhaps this app is worth looking into if you’re like the LOTME staff and you’re up at the top end of that 8-point scale. What, pizza is delicious, who wouldn’t eat it four times a day? And you can also get it from your phone!
It’s time for a little mass kickin’
The universe, scientists tell us, is a big place. Really big. Chromosomes, scientists tell us, are small. Really small. But despite this very fundamental difference, the universe and chromosomes share a deep, dark secret: unexplained mass.
This being a medical publication, we’ll start with chromosomes. A group of researchers measured their mass with x-rays for the first time and found that “the 46 chromosomes in each of our cells weigh 242 picograms (trillionths of a gram). This is heavier than we would expect, and, if replicated, points to unexplained excess mass in chromosomes,” Ian K. Robinson, PhD, said in a written statement.
We’re not just talking about a bit of a beer belly here. “The chromosomes were about 20 times heavier than the DNA they contained,” according to the investigators.
Now to the universe. Here’s what CERN, the European Council for Nuclear Research, has to say about the mass of the universe: “Galaxies in our universe … are rotating with such speed that the gravity generated by their observable matter could not possibly hold them together. … which leads scientists to believe that something we cannot see is at work. They think something we have yet to detect directly is giving these galaxies extra mass.”
But wait, there’s more! “The matter we know and that makes up all stars and galaxies only accounts for 5% of the content of the universe!”
So chromosomes are about 20 times heavier than the DNA they contain, and the universe is about 20 times heavier than the matter that can be seen. Interesting.
We are, of course, happy to share this news with our readers, but there is one catch: Don’t tell Neil deGrasse Tyson. He’ll want to reclassify our genetic solar system into 45 chromosomes and one dwarf chromosome.
A photo finish for the Smart Toilet
We know that poop can tell us a lot about our health, but new research by scientists at Duke University is really on a roll. Their Smart Toilet has been created to help people keep an eye on their bowel health. The device takes pictures of poop after it is flushed and can tell whether the consistency is loose, bloody, or normal.
The Smart Toilet can really help people with issues such as irritable bowel syndrome and inflammatory bowel disease by helping them, and their doctors, keep tabs on their poop. “Typically, gastroenterologists have to rely on patient self-reported information about their stool to help determine the cause of their gastrointestinal health issues, which can be very unreliable,” study lead author Deborah Fisher said.
Not many people look too closely at their poop before it’s flushed, so the fecal photos can make a big difference. The Smart Toilet is installed into the pipes of a toilet and does its thing when the toilet is flushed, so there doesn’t seem to be much work on the patient’s end. Other than the, um, you know, usual work from the patient’s end.
Cancer loses … by a nose
Since the human nose is unpredictable at best, we’ve learned to rely on animals for our detailed nozzle needs. But researchers have found the next best thing to man’s best friend to accurately identify cancers.
A team at the University of Pennsylvania has developed an electronic olfaction, or “e-nose,” that has a 95% accuracy rate in distinguishing benign and malignant pancreatic and ovarian cancer cells from a single blood sample. How?
The e-nose system is equipped with nanosensors that are able to detect the volatile organic compounds (VOCs) emitted by cells in a blood sample. Not only does this create an opportunity for an easier, noninvasive screening practice, but it’s fast. The e-nose can distinguish VOCs from healthy to cancerous blood cells in 20 minutes or less and is just as effective in picking up on early- and late-stage cancers.
The investigators hope that this innovative technology can pave the way for similar devices with other uses. Thanks to the e-nose, a handheld device is in development that may be able to sniff out the signature odor of people with COVID-19.
That’s one smart schnoz.
Do you think this is a (food) game?
Dieting and eating healthy is tough, even during the best of times, and it has not been the best of times. With all respect to Charles Dickens, it’s been the worst of times, full stop. Millions of people have spent the past year sitting around their homes doing nothing, and it’s only natural that many would let their discipline slide.
Naturally, the solution to unhealthy eating habits is to sit down and play with your phone. No, that’s not the joke, the Food Trainer app, available on all cellular devices near you, is designed to encourage healthy eating by turning it into a game of sorts. When users open the app, they’re presented with images of food, and they’re trained to tap on images of healthy food and pass on images of unhealthy ones. The process takes less than 5 minutes.
It sounds really simple, but in a study of more than 1,000 people, consumption of junk food fell by 1 point on an 8-point scale (ranging from four times per day to zero to one time per month), participants lost about half a kilogram (a little over one pound), and more healthy food was eaten. Those who used the app more regularly, along the lines of 10 times per month or more, saw greater benefits.
The authors did acknowledge that those who used the app more may have been more motivated to lose weight anyway, which perhaps limits the overall benefit, but reviews on Google Play were overall quite positive, and if there’s one great truth in this world, it’s that Internet reviewers are almost impossible to please. So perhaps this app is worth looking into if you’re like the LOTME staff and you’re up at the top end of that 8-point scale. What, pizza is delicious, who wouldn’t eat it four times a day? And you can also get it from your phone!
It’s time for a little mass kickin’
The universe, scientists tell us, is a big place. Really big. Chromosomes, scientists tell us, are small. Really small. But despite this very fundamental difference, the universe and chromosomes share a deep, dark secret: unexplained mass.
This being a medical publication, we’ll start with chromosomes. A group of researchers measured their mass with x-rays for the first time and found that “the 46 chromosomes in each of our cells weigh 242 picograms (trillionths of a gram). This is heavier than we would expect, and, if replicated, points to unexplained excess mass in chromosomes,” Ian K. Robinson, PhD, said in a written statement.
We’re not just talking about a bit of a beer belly here. “The chromosomes were about 20 times heavier than the DNA they contained,” according to the investigators.
Now to the universe. Here’s what CERN, the European Council for Nuclear Research, has to say about the mass of the universe: “Galaxies in our universe … are rotating with such speed that the gravity generated by their observable matter could not possibly hold them together. … which leads scientists to believe that something we cannot see is at work. They think something we have yet to detect directly is giving these galaxies extra mass.”
But wait, there’s more! “The matter we know and that makes up all stars and galaxies only accounts for 5% of the content of the universe!”
So chromosomes are about 20 times heavier than the DNA they contain, and the universe is about 20 times heavier than the matter that can be seen. Interesting.
We are, of course, happy to share this news with our readers, but there is one catch: Don’t tell Neil deGrasse Tyson. He’ll want to reclassify our genetic solar system into 45 chromosomes and one dwarf chromosome.
A photo finish for the Smart Toilet
We know that poop can tell us a lot about our health, but new research by scientists at Duke University is really on a roll. Their Smart Toilet has been created to help people keep an eye on their bowel health. The device takes pictures of poop after it is flushed and can tell whether the consistency is loose, bloody, or normal.
The Smart Toilet can really help people with issues such as irritable bowel syndrome and inflammatory bowel disease by helping them, and their doctors, keep tabs on their poop. “Typically, gastroenterologists have to rely on patient self-reported information about their stool to help determine the cause of their gastrointestinal health issues, which can be very unreliable,” study lead author Deborah Fisher said.
Not many people look too closely at their poop before it’s flushed, so the fecal photos can make a big difference. The Smart Toilet is installed into the pipes of a toilet and does its thing when the toilet is flushed, so there doesn’t seem to be much work on the patient’s end. Other than the, um, you know, usual work from the patient’s end.
Cancer loses … by a nose
Since the human nose is unpredictable at best, we’ve learned to rely on animals for our detailed nozzle needs. But researchers have found the next best thing to man’s best friend to accurately identify cancers.
A team at the University of Pennsylvania has developed an electronic olfaction, or “e-nose,” that has a 95% accuracy rate in distinguishing benign and malignant pancreatic and ovarian cancer cells from a single blood sample. How?
The e-nose system is equipped with nanosensors that are able to detect the volatile organic compounds (VOCs) emitted by cells in a blood sample. Not only does this create an opportunity for an easier, noninvasive screening practice, but it’s fast. The e-nose can distinguish VOCs from healthy to cancerous blood cells in 20 minutes or less and is just as effective in picking up on early- and late-stage cancers.
The investigators hope that this innovative technology can pave the way for similar devices with other uses. Thanks to the e-nose, a handheld device is in development that may be able to sniff out the signature odor of people with COVID-19.
That’s one smart schnoz.
Do you think this is a (food) game?
Dieting and eating healthy is tough, even during the best of times, and it has not been the best of times. With all respect to Charles Dickens, it’s been the worst of times, full stop. Millions of people have spent the past year sitting around their homes doing nothing, and it’s only natural that many would let their discipline slide.
Naturally, the solution to unhealthy eating habits is to sit down and play with your phone. No, that’s not the joke, the Food Trainer app, available on all cellular devices near you, is designed to encourage healthy eating by turning it into a game of sorts. When users open the app, they’re presented with images of food, and they’re trained to tap on images of healthy food and pass on images of unhealthy ones. The process takes less than 5 minutes.
It sounds really simple, but in a study of more than 1,000 people, consumption of junk food fell by 1 point on an 8-point scale (ranging from four times per day to zero to one time per month), participants lost about half a kilogram (a little over one pound), and more healthy food was eaten. Those who used the app more regularly, along the lines of 10 times per month or more, saw greater benefits.
The authors did acknowledge that those who used the app more may have been more motivated to lose weight anyway, which perhaps limits the overall benefit, but reviews on Google Play were overall quite positive, and if there’s one great truth in this world, it’s that Internet reviewers are almost impossible to please. So perhaps this app is worth looking into if you’re like the LOTME staff and you’re up at the top end of that 8-point scale. What, pizza is delicious, who wouldn’t eat it four times a day? And you can also get it from your phone!
It’s time for a little mass kickin’
The universe, scientists tell us, is a big place. Really big. Chromosomes, scientists tell us, are small. Really small. But despite this very fundamental difference, the universe and chromosomes share a deep, dark secret: unexplained mass.
This being a medical publication, we’ll start with chromosomes. A group of researchers measured their mass with x-rays for the first time and found that “the 46 chromosomes in each of our cells weigh 242 picograms (trillionths of a gram). This is heavier than we would expect, and, if replicated, points to unexplained excess mass in chromosomes,” Ian K. Robinson, PhD, said in a written statement.
We’re not just talking about a bit of a beer belly here. “The chromosomes were about 20 times heavier than the DNA they contained,” according to the investigators.
Now to the universe. Here’s what CERN, the European Council for Nuclear Research, has to say about the mass of the universe: “Galaxies in our universe … are rotating with such speed that the gravity generated by their observable matter could not possibly hold them together. … which leads scientists to believe that something we cannot see is at work. They think something we have yet to detect directly is giving these galaxies extra mass.”
But wait, there’s more! “The matter we know and that makes up all stars and galaxies only accounts for 5% of the content of the universe!”
So chromosomes are about 20 times heavier than the DNA they contain, and the universe is about 20 times heavier than the matter that can be seen. Interesting.
We are, of course, happy to share this news with our readers, but there is one catch: Don’t tell Neil deGrasse Tyson. He’ll want to reclassify our genetic solar system into 45 chromosomes and one dwarf chromosome.
A photo finish for the Smart Toilet
We know that poop can tell us a lot about our health, but new research by scientists at Duke University is really on a roll. Their Smart Toilet has been created to help people keep an eye on their bowel health. The device takes pictures of poop after it is flushed and can tell whether the consistency is loose, bloody, or normal.
The Smart Toilet can really help people with issues such as irritable bowel syndrome and inflammatory bowel disease by helping them, and their doctors, keep tabs on their poop. “Typically, gastroenterologists have to rely on patient self-reported information about their stool to help determine the cause of their gastrointestinal health issues, which can be very unreliable,” study lead author Deborah Fisher said.
Not many people look too closely at their poop before it’s flushed, so the fecal photos can make a big difference. The Smart Toilet is installed into the pipes of a toilet and does its thing when the toilet is flushed, so there doesn’t seem to be much work on the patient’s end. Other than the, um, you know, usual work from the patient’s end.
Urticarial Vasculitis Successfully Treated With Omalizumab
To the Editor:
Urticarial vasculitis (UV) is a clinicopathologic entity. It manifests as an eruption of erythematous wheals that clinically resemble urticaria, but the lesions of UV last longer, may leave residual hyperpigmentation, and may or may not be pruritic.1 Therapies most often employed include oral antihistamines and systemic immunosuppressant drugs such as corticosteroids, dapsone, colchicine, or hydroxychloroquine.2 We present a woman with UV who successfully was treated with omalizumab.
A 49-year-old woman presented to our outpatient clinic with generalized pruritic skin rashes of 2 years’ duration. She also described swelling on the upper eyelids 2 times monthly. She used several antihistamines (up to 4 times daily) and was taking systemic corticosteroids and antidepressants. Physical examination revealed generalized erythematous and edematous papules and plaques on the trunk and extremities (Figure 1). At follow-up a few days later, we observed that the lesions were lasting for more than 24 hours, but there was no residual pigmentation. According to clinical concerns and the association with angioedema, we initially thought the diagnosis was chronic urticaria and angioedema. The patient had no extracutaneous manifestations such as fever, arthralgia, or lymphadenopathy. Routine laboratory examinations including antinuclear antibodies were within reference range. She had normal C3 and C4 levels and an elevated total IgE level (344 IU/mL [reference range, 0–170 IU/mL]). Because the IgE level was elevated and she had no response to the highest dosages of antihistamines, we decided to start omalizumab therapy. Prior to starting omalizumab, we performed a skin biopsy for histopathologic and direct immunofluorescence examinations for UV, as the duration of the lesions was more than 24 hours. Histopathologic examination revealed lymphocytes within the vessel wall and perivascular lymphocytic infiltration with eosinophils (Figure 2). On direct immunofluorescence, perivascular IgA deposition was observed (Figure 3). Histopathologic findings were associated with lymphocytic vasculitis. Systemic involvement was not detected on detailed laboratory and radiologic examinations.
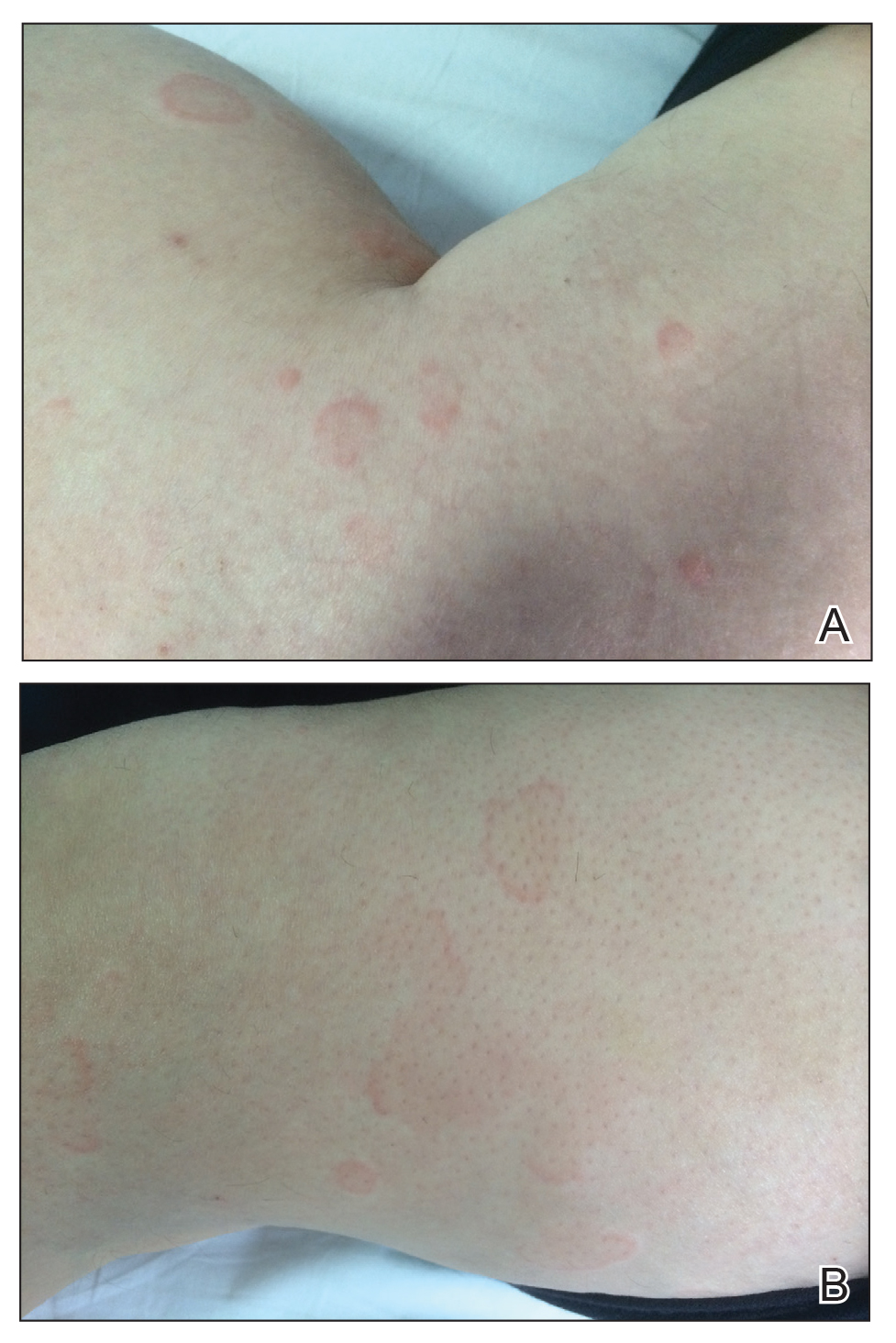


After the first application of omalizumab, the lesions disappeared within a few days. She was treated with subcutaneous omalizumab 300 mg every 4 weeks for 6 months, and we did not observe any adverse effects related to the drug. There was no relapse after therapy cessation.
Omalizumab is a recombinant humanized anti-IgE monoclonal antibody that is approved by the US Food and Drug Administration for treatment of chronic idiopathic urticaria.3-5 Studies have suggested that omalizumab might play an important role in the treatment of other potentially IgE-mediated disease processes including allergic asthma, atopic dermatitis, allergic rhinitis, nasal polyposis, and severe ocular allergies.6 The proposed mechanism of action of omalizumab includes reduction of free IgE through the reversible formation of tiny, biologically inert complexes; targeting IgE-expressing B cells; and inhibiting production of IgE. Because it reduces free IgE, omalizumab has been used in normal IgE or hyper-IgE situations. Omalizumab also induces eosinophil apoptosis; increases IL-2, IL-3, tumor necrosis factor α, and IFN-γ; and reduces IL-4.7 A number of off-label uses have been described such as atopic dermatitis, bullous pemphigoid, hyper-IgE syndrome, cutaneous mastocytosis, toxic epidermal necrolysis, and eosinophilic granulomatosis with polyangitis.8 There are no clinical studies of omalizumab for UV, and only a few case reports have shown that omalizumab also might be beneficial for this condition.2-4 Diez et al4 reported 3 cases of women aged 28, 51, and 54 years with spontaneous chronic urticaria with autoimmune and pressure components as well as vasculitis whose symptoms completely improved after starting omalizumab. Kai et al3 successfully treated a patient with normocomplementemic UV with omalizumab and suggested that omalizumab markedly improved the patient’s quality of life with chronic urticaria and UV. Ghazanfar and Thomsen2 reported the case of a 68-year-old man diagnosed with histopathologically confirmed leukocytoclastic vasculitis. He had used systemic corticosteroid therapy and dapsone without notable improvement. The patient was switched to subcutaneous omalizumab 300 mg once every 4 weeks; after 1 month, he observed complete remission of the UV and symptoms.2
Our case suggests that omalizumab has a beneficial effect on patients with UV. Omalizumab may be effective in UV through its reduction of IgE, as in chronic urticaria, and through downstream effects on cellular activation mechanisms (possibly a reduction in chemotaxis or immune complex formation). However, the mechanism of action of omalizumab for UV remains, in part, unresolved. It is not known whether omalizumab is efficacious against both normocomplementemic and hypocomplementemic UV. Further studies with a greater number of patients are needed to confirm the effects of omalizumab for vasculitic patients.
- Chang S, Carr W. Urticarial vasculitis. Allergy Asthma Proc. 2007;28:97-100.
- Ghazanfar MN, Thomsen SF. Omalizumab for urticarial vasculitis: case report and review of the literature. Case Rep Dermatol Med. 2015:576893.
- Kai AC, Flohr C, Grattan CE. Improvement in quality of life impairment followed by relapse with 6-monthly periodic administration of omalizumab for severe treatment-refractory chronic urticaria and urticarial vasculitis. Clin Exp Dermatol. 2014;39:651-652.
- Diez LS, Tamayo LM, Cardona R. Omalizumab: therapeutic option in chronic spontaneous urticaria difficult to control with associated vasculitis, report of three cases. Biomedica. 2013;33:503-512.
- Maurer M, Rosen K, Hsieh HJ. Omalizumab for chronic urticaria. N Engl J Med. 2013;368:2530.
- Ben Shoshan M. Omalizumab: not only for asthma. Recent Pat Inflamm Allergy Drug Discov. 2008;2:191-201.
- Fueyo-Casado A, Campos-Munoz L, Gonzalez-Guerra E, et al. Effectiveness of omalizumab in a case of urticarial vasculitis. Clin Exp Dermatol. Published March 1, 2017. doi:10.1111/ced.13076
- Chia JC, Mydlarski PR. Dermatologic uses of omalizumab. J Dermatol Treat. Published November 7, 2016. doi:10.1080/09546634.2016.1249819
To the Editor:
Urticarial vasculitis (UV) is a clinicopathologic entity. It manifests as an eruption of erythematous wheals that clinically resemble urticaria, but the lesions of UV last longer, may leave residual hyperpigmentation, and may or may not be pruritic.1 Therapies most often employed include oral antihistamines and systemic immunosuppressant drugs such as corticosteroids, dapsone, colchicine, or hydroxychloroquine.2 We present a woman with UV who successfully was treated with omalizumab.
A 49-year-old woman presented to our outpatient clinic with generalized pruritic skin rashes of 2 years’ duration. She also described swelling on the upper eyelids 2 times monthly. She used several antihistamines (up to 4 times daily) and was taking systemic corticosteroids and antidepressants. Physical examination revealed generalized erythematous and edematous papules and plaques on the trunk and extremities (Figure 1). At follow-up a few days later, we observed that the lesions were lasting for more than 24 hours, but there was no residual pigmentation. According to clinical concerns and the association with angioedema, we initially thought the diagnosis was chronic urticaria and angioedema. The patient had no extracutaneous manifestations such as fever, arthralgia, or lymphadenopathy. Routine laboratory examinations including antinuclear antibodies were within reference range. She had normal C3 and C4 levels and an elevated total IgE level (344 IU/mL [reference range, 0–170 IU/mL]). Because the IgE level was elevated and she had no response to the highest dosages of antihistamines, we decided to start omalizumab therapy. Prior to starting omalizumab, we performed a skin biopsy for histopathologic and direct immunofluorescence examinations for UV, as the duration of the lesions was more than 24 hours. Histopathologic examination revealed lymphocytes within the vessel wall and perivascular lymphocytic infiltration with eosinophils (Figure 2). On direct immunofluorescence, perivascular IgA deposition was observed (Figure 3). Histopathologic findings were associated with lymphocytic vasculitis. Systemic involvement was not detected on detailed laboratory and radiologic examinations.



After the first application of omalizumab, the lesions disappeared within a few days. She was treated with subcutaneous omalizumab 300 mg every 4 weeks for 6 months, and we did not observe any adverse effects related to the drug. There was no relapse after therapy cessation.
Omalizumab is a recombinant humanized anti-IgE monoclonal antibody that is approved by the US Food and Drug Administration for treatment of chronic idiopathic urticaria.3-5 Studies have suggested that omalizumab might play an important role in the treatment of other potentially IgE-mediated disease processes including allergic asthma, atopic dermatitis, allergic rhinitis, nasal polyposis, and severe ocular allergies.6 The proposed mechanism of action of omalizumab includes reduction of free IgE through the reversible formation of tiny, biologically inert complexes; targeting IgE-expressing B cells; and inhibiting production of IgE. Because it reduces free IgE, omalizumab has been used in normal IgE or hyper-IgE situations. Omalizumab also induces eosinophil apoptosis; increases IL-2, IL-3, tumor necrosis factor α, and IFN-γ; and reduces IL-4.7 A number of off-label uses have been described such as atopic dermatitis, bullous pemphigoid, hyper-IgE syndrome, cutaneous mastocytosis, toxic epidermal necrolysis, and eosinophilic granulomatosis with polyangitis.8 There are no clinical studies of omalizumab for UV, and only a few case reports have shown that omalizumab also might be beneficial for this condition.2-4 Diez et al4 reported 3 cases of women aged 28, 51, and 54 years with spontaneous chronic urticaria with autoimmune and pressure components as well as vasculitis whose symptoms completely improved after starting omalizumab. Kai et al3 successfully treated a patient with normocomplementemic UV with omalizumab and suggested that omalizumab markedly improved the patient’s quality of life with chronic urticaria and UV. Ghazanfar and Thomsen2 reported the case of a 68-year-old man diagnosed with histopathologically confirmed leukocytoclastic vasculitis. He had used systemic corticosteroid therapy and dapsone without notable improvement. The patient was switched to subcutaneous omalizumab 300 mg once every 4 weeks; after 1 month, he observed complete remission of the UV and symptoms.2
Our case suggests that omalizumab has a beneficial effect on patients with UV. Omalizumab may be effective in UV through its reduction of IgE, as in chronic urticaria, and through downstream effects on cellular activation mechanisms (possibly a reduction in chemotaxis or immune complex formation). However, the mechanism of action of omalizumab for UV remains, in part, unresolved. It is not known whether omalizumab is efficacious against both normocomplementemic and hypocomplementemic UV. Further studies with a greater number of patients are needed to confirm the effects of omalizumab for vasculitic patients.
To the Editor:
Urticarial vasculitis (UV) is a clinicopathologic entity. It manifests as an eruption of erythematous wheals that clinically resemble urticaria, but the lesions of UV last longer, may leave residual hyperpigmentation, and may or may not be pruritic.1 Therapies most often employed include oral antihistamines and systemic immunosuppressant drugs such as corticosteroids, dapsone, colchicine, or hydroxychloroquine.2 We present a woman with UV who successfully was treated with omalizumab.
A 49-year-old woman presented to our outpatient clinic with generalized pruritic skin rashes of 2 years’ duration. She also described swelling on the upper eyelids 2 times monthly. She used several antihistamines (up to 4 times daily) and was taking systemic corticosteroids and antidepressants. Physical examination revealed generalized erythematous and edematous papules and plaques on the trunk and extremities (Figure 1). At follow-up a few days later, we observed that the lesions were lasting for more than 24 hours, but there was no residual pigmentation. According to clinical concerns and the association with angioedema, we initially thought the diagnosis was chronic urticaria and angioedema. The patient had no extracutaneous manifestations such as fever, arthralgia, or lymphadenopathy. Routine laboratory examinations including antinuclear antibodies were within reference range. She had normal C3 and C4 levels and an elevated total IgE level (344 IU/mL [reference range, 0–170 IU/mL]). Because the IgE level was elevated and she had no response to the highest dosages of antihistamines, we decided to start omalizumab therapy. Prior to starting omalizumab, we performed a skin biopsy for histopathologic and direct immunofluorescence examinations for UV, as the duration of the lesions was more than 24 hours. Histopathologic examination revealed lymphocytes within the vessel wall and perivascular lymphocytic infiltration with eosinophils (Figure 2). On direct immunofluorescence, perivascular IgA deposition was observed (Figure 3). Histopathologic findings were associated with lymphocytic vasculitis. Systemic involvement was not detected on detailed laboratory and radiologic examinations.



After the first application of omalizumab, the lesions disappeared within a few days. She was treated with subcutaneous omalizumab 300 mg every 4 weeks for 6 months, and we did not observe any adverse effects related to the drug. There was no relapse after therapy cessation.
Omalizumab is a recombinant humanized anti-IgE monoclonal antibody that is approved by the US Food and Drug Administration for treatment of chronic idiopathic urticaria.3-5 Studies have suggested that omalizumab might play an important role in the treatment of other potentially IgE-mediated disease processes including allergic asthma, atopic dermatitis, allergic rhinitis, nasal polyposis, and severe ocular allergies.6 The proposed mechanism of action of omalizumab includes reduction of free IgE through the reversible formation of tiny, biologically inert complexes; targeting IgE-expressing B cells; and inhibiting production of IgE. Because it reduces free IgE, omalizumab has been used in normal IgE or hyper-IgE situations. Omalizumab also induces eosinophil apoptosis; increases IL-2, IL-3, tumor necrosis factor α, and IFN-γ; and reduces IL-4.7 A number of off-label uses have been described such as atopic dermatitis, bullous pemphigoid, hyper-IgE syndrome, cutaneous mastocytosis, toxic epidermal necrolysis, and eosinophilic granulomatosis with polyangitis.8 There are no clinical studies of omalizumab for UV, and only a few case reports have shown that omalizumab also might be beneficial for this condition.2-4 Diez et al4 reported 3 cases of women aged 28, 51, and 54 years with spontaneous chronic urticaria with autoimmune and pressure components as well as vasculitis whose symptoms completely improved after starting omalizumab. Kai et al3 successfully treated a patient with normocomplementemic UV with omalizumab and suggested that omalizumab markedly improved the patient’s quality of life with chronic urticaria and UV. Ghazanfar and Thomsen2 reported the case of a 68-year-old man diagnosed with histopathologically confirmed leukocytoclastic vasculitis. He had used systemic corticosteroid therapy and dapsone without notable improvement. The patient was switched to subcutaneous omalizumab 300 mg once every 4 weeks; after 1 month, he observed complete remission of the UV and symptoms.2
Our case suggests that omalizumab has a beneficial effect on patients with UV. Omalizumab may be effective in UV through its reduction of IgE, as in chronic urticaria, and through downstream effects on cellular activation mechanisms (possibly a reduction in chemotaxis or immune complex formation). However, the mechanism of action of omalizumab for UV remains, in part, unresolved. It is not known whether omalizumab is efficacious against both normocomplementemic and hypocomplementemic UV. Further studies with a greater number of patients are needed to confirm the effects of omalizumab for vasculitic patients.
- Chang S, Carr W. Urticarial vasculitis. Allergy Asthma Proc. 2007;28:97-100.
- Ghazanfar MN, Thomsen SF. Omalizumab for urticarial vasculitis: case report and review of the literature. Case Rep Dermatol Med. 2015:576893.
- Kai AC, Flohr C, Grattan CE. Improvement in quality of life impairment followed by relapse with 6-monthly periodic administration of omalizumab for severe treatment-refractory chronic urticaria and urticarial vasculitis. Clin Exp Dermatol. 2014;39:651-652.
- Diez LS, Tamayo LM, Cardona R. Omalizumab: therapeutic option in chronic spontaneous urticaria difficult to control with associated vasculitis, report of three cases. Biomedica. 2013;33:503-512.
- Maurer M, Rosen K, Hsieh HJ. Omalizumab for chronic urticaria. N Engl J Med. 2013;368:2530.
- Ben Shoshan M. Omalizumab: not only for asthma. Recent Pat Inflamm Allergy Drug Discov. 2008;2:191-201.
- Fueyo-Casado A, Campos-Munoz L, Gonzalez-Guerra E, et al. Effectiveness of omalizumab in a case of urticarial vasculitis. Clin Exp Dermatol. Published March 1, 2017. doi:10.1111/ced.13076
- Chia JC, Mydlarski PR. Dermatologic uses of omalizumab. J Dermatol Treat. Published November 7, 2016. doi:10.1080/09546634.2016.1249819
- Chang S, Carr W. Urticarial vasculitis. Allergy Asthma Proc. 2007;28:97-100.
- Ghazanfar MN, Thomsen SF. Omalizumab for urticarial vasculitis: case report and review of the literature. Case Rep Dermatol Med. 2015:576893.
- Kai AC, Flohr C, Grattan CE. Improvement in quality of life impairment followed by relapse with 6-monthly periodic administration of omalizumab for severe treatment-refractory chronic urticaria and urticarial vasculitis. Clin Exp Dermatol. 2014;39:651-652.
- Diez LS, Tamayo LM, Cardona R. Omalizumab: therapeutic option in chronic spontaneous urticaria difficult to control with associated vasculitis, report of three cases. Biomedica. 2013;33:503-512.
- Maurer M, Rosen K, Hsieh HJ. Omalizumab for chronic urticaria. N Engl J Med. 2013;368:2530.
- Ben Shoshan M. Omalizumab: not only for asthma. Recent Pat Inflamm Allergy Drug Discov. 2008;2:191-201.
- Fueyo-Casado A, Campos-Munoz L, Gonzalez-Guerra E, et al. Effectiveness of omalizumab in a case of urticarial vasculitis. Clin Exp Dermatol. Published March 1, 2017. doi:10.1111/ced.13076
- Chia JC, Mydlarski PR. Dermatologic uses of omalizumab. J Dermatol Treat. Published November 7, 2016. doi:10.1080/09546634.2016.1249819
Practice Points
- The differential diagnosis of urticaria and urticarial vasculitis may be complicated.
- Omalizumab is an effective urticaria treatment and also can be an alternative treatment choice in resistant urticarial vasculitis.
Multiple Crusted Swellings on the Chin
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).
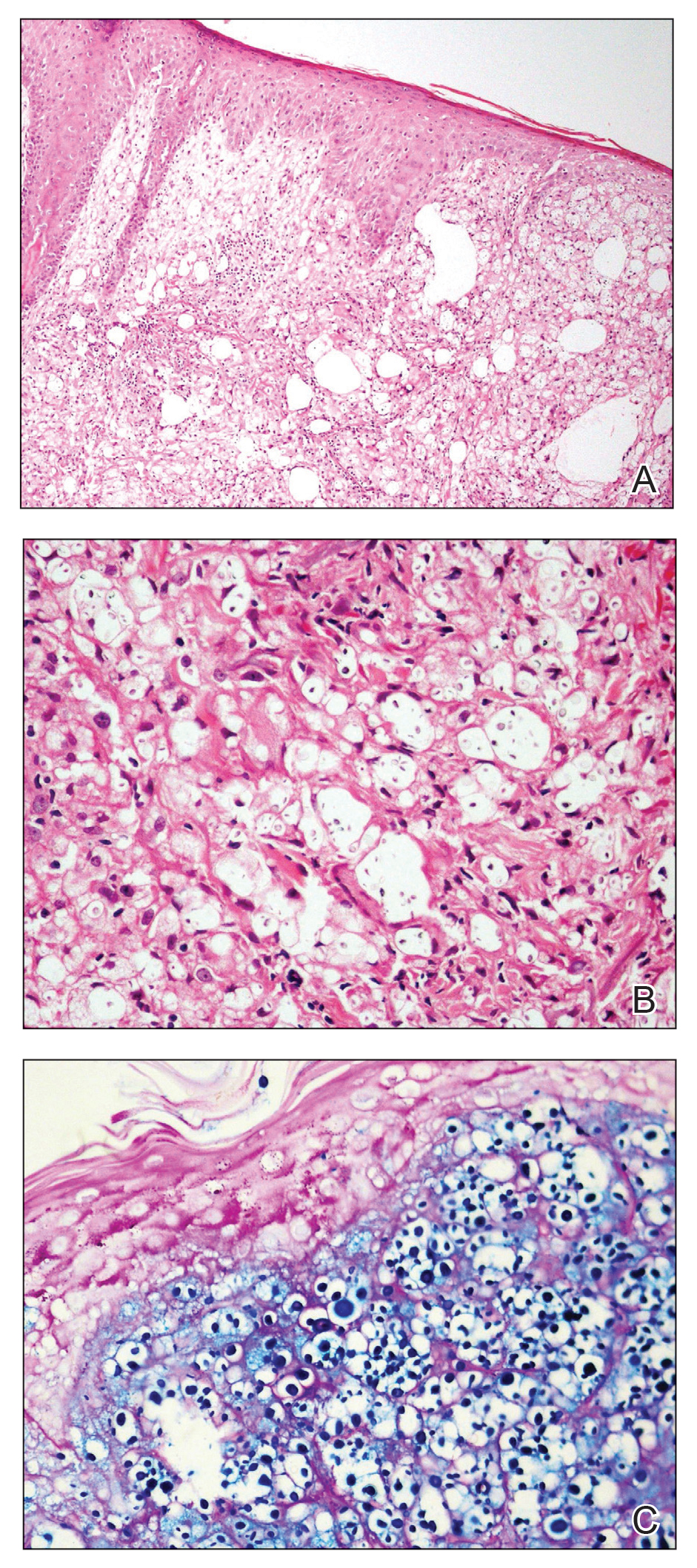
Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).


Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).


Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
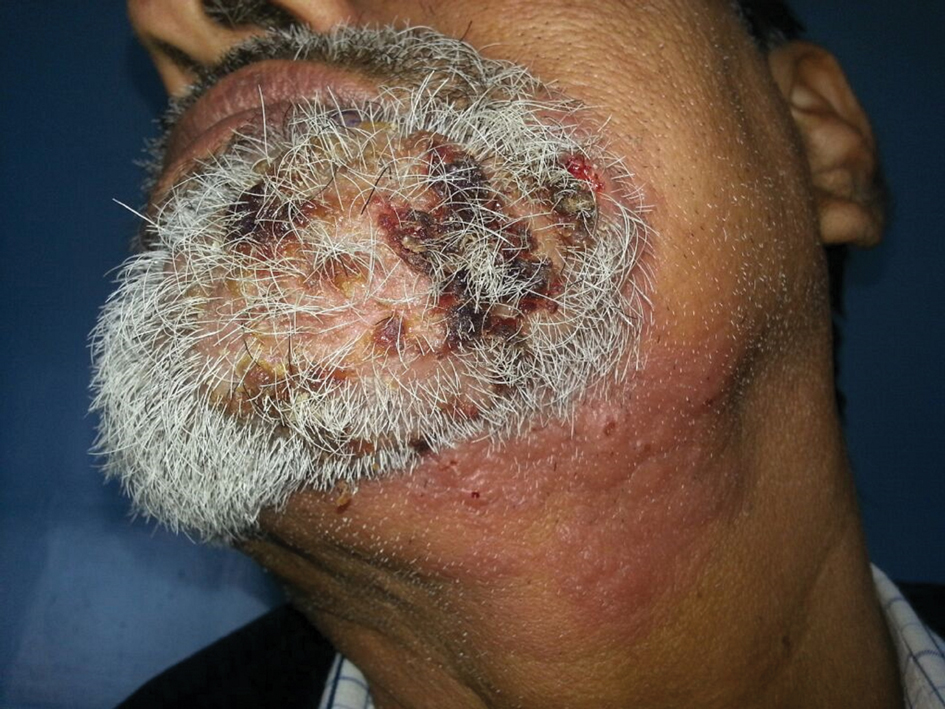
A 54-year-old man with no comorbidities presented with multiple painless swellings on the left side of the chin of 1 month’s duration that progressively were increasing, both in size and number. He denied any discharge of pus or grains from the lesion, facial trauma, insect bites, or dental procedures. The patient was treated with oral antibiotics for 15 days with no relief at an outside hospital. All routine blood and serologic investigations including viral markers and chest radiography were normal. Bacterial and fungal cultures as well as an acid-fast bacilli culture were negative. Systemic examination was normal, and vitals were within reference range. Mucocutaneous examination revealed multiple nontender small nodules and plaques with yellow-brown to dark brown hemorrhagic crusts with mild perilesional erythema on the left side of the chin extending to the adjacent neck. All mucosal sites were normal, and a biopsy was performed.
DOJ charges 14 with COVID-19–related fraud nearing $150M
The U.S. Department of Justice (DOJ) on May 26 announced charges against 14 defendants across the country who allegedly engaged in health care fraud schemes that exploited the COVID-19 pandemic and resulted in over $143 million in false billings to Medicare.
Among the defendants, a DOJ news release said, were a telemedicine company executive, a physician, marketers, and medical business owners.
In addition, the Centers for Medicare and Medicaid Services separately announced that it had taken “adverse administrative actions” against more than 50 providers for their involvement in fraud schemes related to COVID-19 or the abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Several of the defendants allegedly offered COVID-19 tests to Medicare beneficiaries in senior living facilities, drive-through COVID-19 testing sites, and medical offices to induce the beneficiaries to provide their personal identifying information and a saliva or a blood sample.
The DOJ charges claim the defendants then misused the information and the samples to submit claims to Medicare for unrelated, medically unnecessary, and far more expensive lab tests, including cancer genetic testing, allergy testing, and respiratory pathogen panel tests.
In some cases, it’s alleged, the lab results were not provided to the individuals in a timely fashion or were not reliable.
Other defendants are charged with exploiting temporary changes in CMS telehealth regulations that were designed to increase access to health care during the pandemic. In these cases, which the DOJ said were the first charges related to the expansion of telehealth under the COVID-19 emergency declaration, the defendants allegedly submitted false and fraudulent claims to Medicare for sham telemedicine encounters that did not occur.
“As part of these cases, medical professionals are alleged to have [been] offered and paid bribes in exchange for the medical professionals’ referral of unnecessary testing,” the DOJ news release said. However, no physicians were identified by the department.
Commenting on this aspect of the law enforcement action, FBI Director Christopher Wray said in the release: “Medical providers have been the unsung heroes for the American public throughout the pandemic. It’s disheartening that some have abused their authorities and committed COVID-19–related fraud against trusting citizens. The FBI, along with our federal law enforcement and private sector partners, are committed to continuing to combat health care fraud and protect the American people.”
The law enforcement action includes the third set of criminal charges related to the misuse of Provider Relief Fund monies, according to the release.
More than 340 individuals were charged in September 2020 with submitting $6 billion in fraudulent claims to federal health care programs and private insurers for telehealth consultations and substance abuse treatment. About $4.5 billion of that was related to telehealth, as reported by this news organization.
The new criminal charges were brought in federal district courts in Arkansas, California, Louisiana, Florida, New Jersey, and New York.
Case summaries
The DOJ provided several case summaries. One defendant, lab owner Billy Joe Taylor of Lavaca, Ark., was charged with participating in a scheme to defraud the government of over $42 million by filing false claims that were billed in combination with COVID-19 testing claims. He also allegedly billed for tests that were not performed.
Petros Hannesyan of Burbank, Calif., the owner of a home health agency, was charged with obtaining over $229,000 from COVID-19 relief programs under false pretenses. His firm allegedly misappropriated funds from the CARES Act Provider Relief Fund and submitted false loan applications and a false loan agreement to the Economic Injury Disaster Loan Program.
Michael Stein and Leonel Palatnik of Palm Beach County, Fla., were charged in a connection with an alleged $73 million conspiracy to defraud the government and to pay and receive health care kickbacks during the pandemic.
Mr. Stein, who owned a “purported” consulting company, and Mr. Palatnik, who owned testing labs in Texas, allegedly exploited Medicare’s waiver of telehealth restrictions “by offering telehealth providers access to Medicare beneficiaries for whom they could bill consultations. In exchange, these providers agreed to refer beneficiaries to [Mr. Palatnik’s] laboratories for expensive and medically unnecessary cancer and cardiovascular genetic testing.”
A version of this article first appeared on Medscape.com.
The U.S. Department of Justice (DOJ) on May 26 announced charges against 14 defendants across the country who allegedly engaged in health care fraud schemes that exploited the COVID-19 pandemic and resulted in over $143 million in false billings to Medicare.
Among the defendants, a DOJ news release said, were a telemedicine company executive, a physician, marketers, and medical business owners.
In addition, the Centers for Medicare and Medicaid Services separately announced that it had taken “adverse administrative actions” against more than 50 providers for their involvement in fraud schemes related to COVID-19 or the abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Several of the defendants allegedly offered COVID-19 tests to Medicare beneficiaries in senior living facilities, drive-through COVID-19 testing sites, and medical offices to induce the beneficiaries to provide their personal identifying information and a saliva or a blood sample.
The DOJ charges claim the defendants then misused the information and the samples to submit claims to Medicare for unrelated, medically unnecessary, and far more expensive lab tests, including cancer genetic testing, allergy testing, and respiratory pathogen panel tests.
In some cases, it’s alleged, the lab results were not provided to the individuals in a timely fashion or were not reliable.
Other defendants are charged with exploiting temporary changes in CMS telehealth regulations that were designed to increase access to health care during the pandemic. In these cases, which the DOJ said were the first charges related to the expansion of telehealth under the COVID-19 emergency declaration, the defendants allegedly submitted false and fraudulent claims to Medicare for sham telemedicine encounters that did not occur.
“As part of these cases, medical professionals are alleged to have [been] offered and paid bribes in exchange for the medical professionals’ referral of unnecessary testing,” the DOJ news release said. However, no physicians were identified by the department.
Commenting on this aspect of the law enforcement action, FBI Director Christopher Wray said in the release: “Medical providers have been the unsung heroes for the American public throughout the pandemic. It’s disheartening that some have abused their authorities and committed COVID-19–related fraud against trusting citizens. The FBI, along with our federal law enforcement and private sector partners, are committed to continuing to combat health care fraud and protect the American people.”
The law enforcement action includes the third set of criminal charges related to the misuse of Provider Relief Fund monies, according to the release.
More than 340 individuals were charged in September 2020 with submitting $6 billion in fraudulent claims to federal health care programs and private insurers for telehealth consultations and substance abuse treatment. About $4.5 billion of that was related to telehealth, as reported by this news organization.
The new criminal charges were brought in federal district courts in Arkansas, California, Louisiana, Florida, New Jersey, and New York.
Case summaries
The DOJ provided several case summaries. One defendant, lab owner Billy Joe Taylor of Lavaca, Ark., was charged with participating in a scheme to defraud the government of over $42 million by filing false claims that were billed in combination with COVID-19 testing claims. He also allegedly billed for tests that were not performed.
Petros Hannesyan of Burbank, Calif., the owner of a home health agency, was charged with obtaining over $229,000 from COVID-19 relief programs under false pretenses. His firm allegedly misappropriated funds from the CARES Act Provider Relief Fund and submitted false loan applications and a false loan agreement to the Economic Injury Disaster Loan Program.
Michael Stein and Leonel Palatnik of Palm Beach County, Fla., were charged in a connection with an alleged $73 million conspiracy to defraud the government and to pay and receive health care kickbacks during the pandemic.
Mr. Stein, who owned a “purported” consulting company, and Mr. Palatnik, who owned testing labs in Texas, allegedly exploited Medicare’s waiver of telehealth restrictions “by offering telehealth providers access to Medicare beneficiaries for whom they could bill consultations. In exchange, these providers agreed to refer beneficiaries to [Mr. Palatnik’s] laboratories for expensive and medically unnecessary cancer and cardiovascular genetic testing.”
A version of this article first appeared on Medscape.com.
The U.S. Department of Justice (DOJ) on May 26 announced charges against 14 defendants across the country who allegedly engaged in health care fraud schemes that exploited the COVID-19 pandemic and resulted in over $143 million in false billings to Medicare.
Among the defendants, a DOJ news release said, were a telemedicine company executive, a physician, marketers, and medical business owners.
In addition, the Centers for Medicare and Medicaid Services separately announced that it had taken “adverse administrative actions” against more than 50 providers for their involvement in fraud schemes related to COVID-19 or the abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Several of the defendants allegedly offered COVID-19 tests to Medicare beneficiaries in senior living facilities, drive-through COVID-19 testing sites, and medical offices to induce the beneficiaries to provide their personal identifying information and a saliva or a blood sample.
The DOJ charges claim the defendants then misused the information and the samples to submit claims to Medicare for unrelated, medically unnecessary, and far more expensive lab tests, including cancer genetic testing, allergy testing, and respiratory pathogen panel tests.
In some cases, it’s alleged, the lab results were not provided to the individuals in a timely fashion or were not reliable.
Other defendants are charged with exploiting temporary changes in CMS telehealth regulations that were designed to increase access to health care during the pandemic. In these cases, which the DOJ said were the first charges related to the expansion of telehealth under the COVID-19 emergency declaration, the defendants allegedly submitted false and fraudulent claims to Medicare for sham telemedicine encounters that did not occur.
“As part of these cases, medical professionals are alleged to have [been] offered and paid bribes in exchange for the medical professionals’ referral of unnecessary testing,” the DOJ news release said. However, no physicians were identified by the department.
Commenting on this aspect of the law enforcement action, FBI Director Christopher Wray said in the release: “Medical providers have been the unsung heroes for the American public throughout the pandemic. It’s disheartening that some have abused their authorities and committed COVID-19–related fraud against trusting citizens. The FBI, along with our federal law enforcement and private sector partners, are committed to continuing to combat health care fraud and protect the American people.”
The law enforcement action includes the third set of criminal charges related to the misuse of Provider Relief Fund monies, according to the release.
More than 340 individuals were charged in September 2020 with submitting $6 billion in fraudulent claims to federal health care programs and private insurers for telehealth consultations and substance abuse treatment. About $4.5 billion of that was related to telehealth, as reported by this news organization.
The new criminal charges were brought in federal district courts in Arkansas, California, Louisiana, Florida, New Jersey, and New York.
Case summaries
The DOJ provided several case summaries. One defendant, lab owner Billy Joe Taylor of Lavaca, Ark., was charged with participating in a scheme to defraud the government of over $42 million by filing false claims that were billed in combination with COVID-19 testing claims. He also allegedly billed for tests that were not performed.
Petros Hannesyan of Burbank, Calif., the owner of a home health agency, was charged with obtaining over $229,000 from COVID-19 relief programs under false pretenses. His firm allegedly misappropriated funds from the CARES Act Provider Relief Fund and submitted false loan applications and a false loan agreement to the Economic Injury Disaster Loan Program.
Michael Stein and Leonel Palatnik of Palm Beach County, Fla., were charged in a connection with an alleged $73 million conspiracy to defraud the government and to pay and receive health care kickbacks during the pandemic.
Mr. Stein, who owned a “purported” consulting company, and Mr. Palatnik, who owned testing labs in Texas, allegedly exploited Medicare’s waiver of telehealth restrictions “by offering telehealth providers access to Medicare beneficiaries for whom they could bill consultations. In exchange, these providers agreed to refer beneficiaries to [Mr. Palatnik’s] laboratories for expensive and medically unnecessary cancer and cardiovascular genetic testing.”
A version of this article first appeared on Medscape.com.