User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
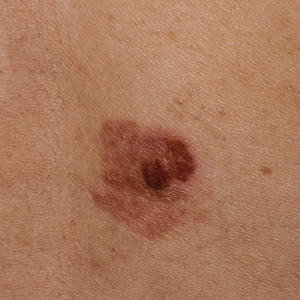
AAD unveils new guidelines for actinic keratosis management
. They also conditionally recommend the use of photodynamic therapy (PDT) and diclofenac for the treatment of AK, both individually and as part of combination therapy regimens.
Those are two of 18 recommendations made by 14 members of the multidisciplinary work group that convened to assemble the AAD’s first-ever guidelines on the management of AKs, which were published online April 2 in the Journal of the American Academy of Dermatology. The group, cochaired by Daniel B. Eisen, MD, professor of clinical dermatology at the University of California, Davis, and Todd E. Schlesinger, MD, medical director of the Dermatology and Laser Center of Charleston, S.C., conducted a systematic review to address five clinical questions on the management of AKs in adults. The questions were: What are the efficacy, effectiveness, and adverse effects of surgical and chemical peel treatments for AK; of topically applied agents for AK; of energy devices and other miscellaneous treatments for AK; and of combination therapy for the treatment of AK? And what are the special considerations to be taken into account when treating AK in immunocompromised individuals?
Next, the work group applied the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for assessing the certainty of the evidence and formulating and grading clinical recommendations based on relevant randomized trials in the medical literature.
“As a participant in the work group, I was impressed by the level of care and detail and the involvement of relevant stakeholders, including a patient advocate, as well as having the draft guidelines go out to the AAD membership at large, and evaluating every comment that came in,” Maryam Asgari, MD, MPH, professor of dermatology at Harvard University, Boston, said in an interview. “The academy sought stakeholder and leadership input in revising and revamping the guidelines. The AAD also made sure the work group had minimal conflicts of interest by requiring that the majority of experts convened did not have relevant financial conflicts of interest. That might not be the case in a publication such as a systematic review, where no threshold for financial conflict of interest for coauthorship is set.”
Of the 18 recommendations the work group made for patients with AKs, only four were ranked as “strong” based on the evidence reviewed, while the rest were ranked as “conditional.”
The strong recommendations include the use of UV protection, field treatment with 5-FU, field treatment with imiquimod, and the use of cryosurgery.
The first four conditional recommendations for patients with AKs include the use of diclofenac, treatment with cryosurgery over CO2 laser ablation, aminolevulinic acid (ALA)–red-light PDT, and 1- to 4-hour 5-ALA incubation time to enhance complete clearance with red-light PDT. The work group also conditionally recommends ALA-daylight PDT as less painful than but equally effective as ALA–red-light PDT.
In the clinical experience of Catherine M. DiGiorgio, MD, who was not involved in the guidelines, daylight PDT with ALA is a viable, cost-effective option. “Patients can come into the office, apply the ALA and then they go outside for 2 hours – not in direct sunlight but in a shady area,” Dr. DiGiorgio, a dermatologist who practices at the Boston Center for Facial Rejuvenation, said in an interview. “That’s a cost-effective treatment for patients who perhaps can’t afford some of the chemotherapy creams. I don’t think we’ve adopted ALA-daylight PDT here in the U.S. very much.”
The work group noted that topical 1% tirbanibulin ointment, a novel microtubule inhibitor, was approved for treatment of AKs on the face and scalp by the Food and Drug Administration after the guidelines had been put together.
Several trials of combination therapy were included in the review of evidence, prompting several recommendations. For example, the work group conditionally recommends combined 5-FU cream and cryosurgery over cryosurgery alone, based on moderate-quality evidence and conditionally recommends combined imiquimod and cryosurgery over cryosurgery alone based on low-quality evidence. In addition, the work group conditionally recommends against the use of 3% diclofenac in addition to cryosurgery, favoring cryosurgery alone based on low-quality evidence, and conditionally recommends against the use of imiquimod typically after ALA–blue-light PDT, based on moderate-quality data.
“The additional treatment with imiquimod was thought to add both expense and burden to the patient, which negates much of the perceived convenience of using PDT as a stand-alone treatment modality and which is not mitigated by the modest increase in lesion reduction,” the authors wrote.
The guidelines emphasize the importance of shared decision-making between patients and clinicians on the choice of therapy, a point that resonates with Dr. DiGiorgio. Success of a treatment can depend on whether a patient is willing to go through with it, she said. “Some patients don’t want to do a therapeutic topical like 5-FU. They prefer to come in and have cryotherapy done. Others prefer to not come in and have the cream at home and treat themselves.”
Assembling the guidelines exposed certain gaps in research, according to the work group. Of the 18 recommendations, seven were based on low-quality evidence, and there were not enough data to make guidelines for the treatment of AKs in immunocompromised individuals.
“I can’t tell you the number of times we in the committee sat back and said, ‘we need to have a randomized trial that looks at this, or compares this to that head on,’” Dr. Asgari said. Such limitations “give researchers direction for where the areas of study need to go to help us answer some of these management conundrums.”
She added that the new guidelines “give clinicians a leg to stand on” when an insurer pushes back on a recommended treatment for AK. “It gives you a way to have dialogue with insurers if you’re prescribing some of these treatments.”
The guidelines authors write that there is “strong theoretic rationale for the treatment of AK to prevent skin cancers” but acknowledge that only a few studies in the review “report the incidence of skin cancer as an outcome measure or have sufficient follow-up to viably measure carcinoma development.” In addition, “more long-term research is needed to validate our current understanding of skin cancer progression from AKs to keratinocyte carcinoma.”
Dr. DiGiorgio thinks about this differently. “I think treatment of AKs does prevent skin cancers,” she said. “We call them precancers as we’re treating our patients because we know a certain percentage of them can develop into skin cancers over time.”
The study was funded by internal funds from the AAD. Dr. Asgari disclosed that she serves as an investigator for Pfizer. Several of the other authors reported having financial disclosures.
Dr. DiGiorgio reported having no financial disclosures.
. They also conditionally recommend the use of photodynamic therapy (PDT) and diclofenac for the treatment of AK, both individually and as part of combination therapy regimens.
Those are two of 18 recommendations made by 14 members of the multidisciplinary work group that convened to assemble the AAD’s first-ever guidelines on the management of AKs, which were published online April 2 in the Journal of the American Academy of Dermatology. The group, cochaired by Daniel B. Eisen, MD, professor of clinical dermatology at the University of California, Davis, and Todd E. Schlesinger, MD, medical director of the Dermatology and Laser Center of Charleston, S.C., conducted a systematic review to address five clinical questions on the management of AKs in adults. The questions were: What are the efficacy, effectiveness, and adverse effects of surgical and chemical peel treatments for AK; of topically applied agents for AK; of energy devices and other miscellaneous treatments for AK; and of combination therapy for the treatment of AK? And what are the special considerations to be taken into account when treating AK in immunocompromised individuals?
Next, the work group applied the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for assessing the certainty of the evidence and formulating and grading clinical recommendations based on relevant randomized trials in the medical literature.
“As a participant in the work group, I was impressed by the level of care and detail and the involvement of relevant stakeholders, including a patient advocate, as well as having the draft guidelines go out to the AAD membership at large, and evaluating every comment that came in,” Maryam Asgari, MD, MPH, professor of dermatology at Harvard University, Boston, said in an interview. “The academy sought stakeholder and leadership input in revising and revamping the guidelines. The AAD also made sure the work group had minimal conflicts of interest by requiring that the majority of experts convened did not have relevant financial conflicts of interest. That might not be the case in a publication such as a systematic review, where no threshold for financial conflict of interest for coauthorship is set.”
Of the 18 recommendations the work group made for patients with AKs, only four were ranked as “strong” based on the evidence reviewed, while the rest were ranked as “conditional.”
The strong recommendations include the use of UV protection, field treatment with 5-FU, field treatment with imiquimod, and the use of cryosurgery.
The first four conditional recommendations for patients with AKs include the use of diclofenac, treatment with cryosurgery over CO2 laser ablation, aminolevulinic acid (ALA)–red-light PDT, and 1- to 4-hour 5-ALA incubation time to enhance complete clearance with red-light PDT. The work group also conditionally recommends ALA-daylight PDT as less painful than but equally effective as ALA–red-light PDT.
In the clinical experience of Catherine M. DiGiorgio, MD, who was not involved in the guidelines, daylight PDT with ALA is a viable, cost-effective option. “Patients can come into the office, apply the ALA and then they go outside for 2 hours – not in direct sunlight but in a shady area,” Dr. DiGiorgio, a dermatologist who practices at the Boston Center for Facial Rejuvenation, said in an interview. “That’s a cost-effective treatment for patients who perhaps can’t afford some of the chemotherapy creams. I don’t think we’ve adopted ALA-daylight PDT here in the U.S. very much.”
The work group noted that topical 1% tirbanibulin ointment, a novel microtubule inhibitor, was approved for treatment of AKs on the face and scalp by the Food and Drug Administration after the guidelines had been put together.
Several trials of combination therapy were included in the review of evidence, prompting several recommendations. For example, the work group conditionally recommends combined 5-FU cream and cryosurgery over cryosurgery alone, based on moderate-quality evidence and conditionally recommends combined imiquimod and cryosurgery over cryosurgery alone based on low-quality evidence. In addition, the work group conditionally recommends against the use of 3% diclofenac in addition to cryosurgery, favoring cryosurgery alone based on low-quality evidence, and conditionally recommends against the use of imiquimod typically after ALA–blue-light PDT, based on moderate-quality data.
“The additional treatment with imiquimod was thought to add both expense and burden to the patient, which negates much of the perceived convenience of using PDT as a stand-alone treatment modality and which is not mitigated by the modest increase in lesion reduction,” the authors wrote.
The guidelines emphasize the importance of shared decision-making between patients and clinicians on the choice of therapy, a point that resonates with Dr. DiGiorgio. Success of a treatment can depend on whether a patient is willing to go through with it, she said. “Some patients don’t want to do a therapeutic topical like 5-FU. They prefer to come in and have cryotherapy done. Others prefer to not come in and have the cream at home and treat themselves.”
Assembling the guidelines exposed certain gaps in research, according to the work group. Of the 18 recommendations, seven were based on low-quality evidence, and there were not enough data to make guidelines for the treatment of AKs in immunocompromised individuals.
“I can’t tell you the number of times we in the committee sat back and said, ‘we need to have a randomized trial that looks at this, or compares this to that head on,’” Dr. Asgari said. Such limitations “give researchers direction for where the areas of study need to go to help us answer some of these management conundrums.”
She added that the new guidelines “give clinicians a leg to stand on” when an insurer pushes back on a recommended treatment for AK. “It gives you a way to have dialogue with insurers if you’re prescribing some of these treatments.”
The guidelines authors write that there is “strong theoretic rationale for the treatment of AK to prevent skin cancers” but acknowledge that only a few studies in the review “report the incidence of skin cancer as an outcome measure or have sufficient follow-up to viably measure carcinoma development.” In addition, “more long-term research is needed to validate our current understanding of skin cancer progression from AKs to keratinocyte carcinoma.”
Dr. DiGiorgio thinks about this differently. “I think treatment of AKs does prevent skin cancers,” she said. “We call them precancers as we’re treating our patients because we know a certain percentage of them can develop into skin cancers over time.”
The study was funded by internal funds from the AAD. Dr. Asgari disclosed that she serves as an investigator for Pfizer. Several of the other authors reported having financial disclosures.
Dr. DiGiorgio reported having no financial disclosures.
. They also conditionally recommend the use of photodynamic therapy (PDT) and diclofenac for the treatment of AK, both individually and as part of combination therapy regimens.
Those are two of 18 recommendations made by 14 members of the multidisciplinary work group that convened to assemble the AAD’s first-ever guidelines on the management of AKs, which were published online April 2 in the Journal of the American Academy of Dermatology. The group, cochaired by Daniel B. Eisen, MD, professor of clinical dermatology at the University of California, Davis, and Todd E. Schlesinger, MD, medical director of the Dermatology and Laser Center of Charleston, S.C., conducted a systematic review to address five clinical questions on the management of AKs in adults. The questions were: What are the efficacy, effectiveness, and adverse effects of surgical and chemical peel treatments for AK; of topically applied agents for AK; of energy devices and other miscellaneous treatments for AK; and of combination therapy for the treatment of AK? And what are the special considerations to be taken into account when treating AK in immunocompromised individuals?
Next, the work group applied the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach for assessing the certainty of the evidence and formulating and grading clinical recommendations based on relevant randomized trials in the medical literature.
“As a participant in the work group, I was impressed by the level of care and detail and the involvement of relevant stakeholders, including a patient advocate, as well as having the draft guidelines go out to the AAD membership at large, and evaluating every comment that came in,” Maryam Asgari, MD, MPH, professor of dermatology at Harvard University, Boston, said in an interview. “The academy sought stakeholder and leadership input in revising and revamping the guidelines. The AAD also made sure the work group had minimal conflicts of interest by requiring that the majority of experts convened did not have relevant financial conflicts of interest. That might not be the case in a publication such as a systematic review, where no threshold for financial conflict of interest for coauthorship is set.”
Of the 18 recommendations the work group made for patients with AKs, only four were ranked as “strong” based on the evidence reviewed, while the rest were ranked as “conditional.”
The strong recommendations include the use of UV protection, field treatment with 5-FU, field treatment with imiquimod, and the use of cryosurgery.
The first four conditional recommendations for patients with AKs include the use of diclofenac, treatment with cryosurgery over CO2 laser ablation, aminolevulinic acid (ALA)–red-light PDT, and 1- to 4-hour 5-ALA incubation time to enhance complete clearance with red-light PDT. The work group also conditionally recommends ALA-daylight PDT as less painful than but equally effective as ALA–red-light PDT.
In the clinical experience of Catherine M. DiGiorgio, MD, who was not involved in the guidelines, daylight PDT with ALA is a viable, cost-effective option. “Patients can come into the office, apply the ALA and then they go outside for 2 hours – not in direct sunlight but in a shady area,” Dr. DiGiorgio, a dermatologist who practices at the Boston Center for Facial Rejuvenation, said in an interview. “That’s a cost-effective treatment for patients who perhaps can’t afford some of the chemotherapy creams. I don’t think we’ve adopted ALA-daylight PDT here in the U.S. very much.”
The work group noted that topical 1% tirbanibulin ointment, a novel microtubule inhibitor, was approved for treatment of AKs on the face and scalp by the Food and Drug Administration after the guidelines had been put together.
Several trials of combination therapy were included in the review of evidence, prompting several recommendations. For example, the work group conditionally recommends combined 5-FU cream and cryosurgery over cryosurgery alone, based on moderate-quality evidence and conditionally recommends combined imiquimod and cryosurgery over cryosurgery alone based on low-quality evidence. In addition, the work group conditionally recommends against the use of 3% diclofenac in addition to cryosurgery, favoring cryosurgery alone based on low-quality evidence, and conditionally recommends against the use of imiquimod typically after ALA–blue-light PDT, based on moderate-quality data.
“The additional treatment with imiquimod was thought to add both expense and burden to the patient, which negates much of the perceived convenience of using PDT as a stand-alone treatment modality and which is not mitigated by the modest increase in lesion reduction,” the authors wrote.
The guidelines emphasize the importance of shared decision-making between patients and clinicians on the choice of therapy, a point that resonates with Dr. DiGiorgio. Success of a treatment can depend on whether a patient is willing to go through with it, she said. “Some patients don’t want to do a therapeutic topical like 5-FU. They prefer to come in and have cryotherapy done. Others prefer to not come in and have the cream at home and treat themselves.”
Assembling the guidelines exposed certain gaps in research, according to the work group. Of the 18 recommendations, seven were based on low-quality evidence, and there were not enough data to make guidelines for the treatment of AKs in immunocompromised individuals.
“I can’t tell you the number of times we in the committee sat back and said, ‘we need to have a randomized trial that looks at this, or compares this to that head on,’” Dr. Asgari said. Such limitations “give researchers direction for where the areas of study need to go to help us answer some of these management conundrums.”
She added that the new guidelines “give clinicians a leg to stand on” when an insurer pushes back on a recommended treatment for AK. “It gives you a way to have dialogue with insurers if you’re prescribing some of these treatments.”
The guidelines authors write that there is “strong theoretic rationale for the treatment of AK to prevent skin cancers” but acknowledge that only a few studies in the review “report the incidence of skin cancer as an outcome measure or have sufficient follow-up to viably measure carcinoma development.” In addition, “more long-term research is needed to validate our current understanding of skin cancer progression from AKs to keratinocyte carcinoma.”
Dr. DiGiorgio thinks about this differently. “I think treatment of AKs does prevent skin cancers,” she said. “We call them precancers as we’re treating our patients because we know a certain percentage of them can develop into skin cancers over time.”
The study was funded by internal funds from the AAD. Dr. Asgari disclosed that she serves as an investigator for Pfizer. Several of the other authors reported having financial disclosures.
Dr. DiGiorgio reported having no financial disclosures.
FROM JAAD
Pandemic took a cut of cosmetic procedures in 2020
pandemic, according to the American Society of Plastic Surgeons.
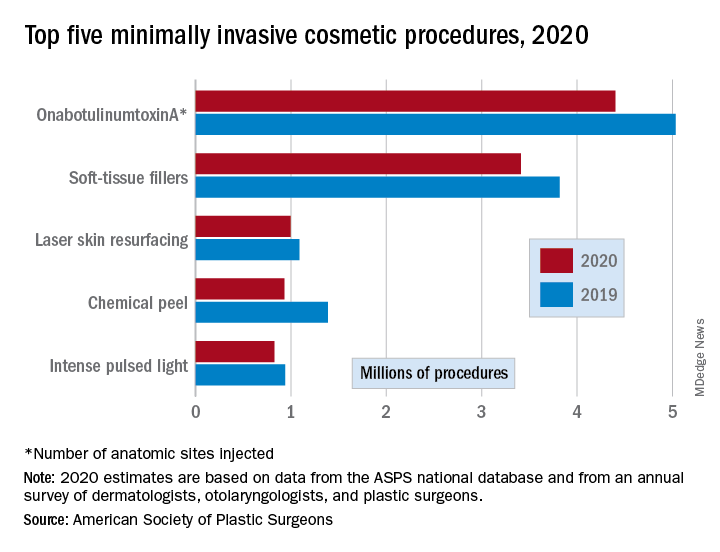
There were an estimated 15.6 million cosmetic procedures performed in 2020, compared with 18.4 million in 2019, a drop of 15.2%. Meanwhile, society members reported that they stopped performing elective surgery for an average of 8.1 weeks, which works out to 15.6% of a 52-week year, the ASPS said in its annual statistics report.
“The pandemic isn’t over, but thanks to vaccines, a new normal is starting to define itself – and some surgeons’ offices that were closed or offered only limited services within the last year are seeing higher demand,” Lynn Jeffers, MD, MBA, immediate past president of the ASPS, said in a written statement.
Minimally invasive procedures, which made up the majority of cosmetic procedures in 2020, dropped by a slightly higher 16%, compared with 14% on the surgical side. “Injectables continued to be the most sought-after treatments in 2020,” the ASPS said, with survey respondents citing “a significant uptick in demand during the coronavirus pandemic.”
OnabotuliumtoxinA injection, the most popular form of minimally invasive procedure, was down by 13% from 2019, while use of soft-tissue fillers fell by 11%. Laser skin resurfacing was third in popularity and had the smallest drop, just 8%, among the top five from 2019 to 2020, the ASPS data show.
The drop in volume for chemical peels was large enough (33%), to move it from third place in 2019 to fourth in 2020, and a slightly less than average drop of 12% moved intense pulsed-light treatments from sixth place in 2019 to fifth in 2020, switching places with laser hair removal (down 28%), the ASPS reported.
Among the surgical procedures, rhinoplasty was the most popular in 2020, as it was in 2019, after dropping by just 3%. Blepharoplasty was down by 8% from 2019, but two other common procedures, liposuction and breast augmentation, fell by 20% and 33%, respectively, the ASPS said.
pandemic, according to the American Society of Plastic Surgeons.

There were an estimated 15.6 million cosmetic procedures performed in 2020, compared with 18.4 million in 2019, a drop of 15.2%. Meanwhile, society members reported that they stopped performing elective surgery for an average of 8.1 weeks, which works out to 15.6% of a 52-week year, the ASPS said in its annual statistics report.
“The pandemic isn’t over, but thanks to vaccines, a new normal is starting to define itself – and some surgeons’ offices that were closed or offered only limited services within the last year are seeing higher demand,” Lynn Jeffers, MD, MBA, immediate past president of the ASPS, said in a written statement.
Minimally invasive procedures, which made up the majority of cosmetic procedures in 2020, dropped by a slightly higher 16%, compared with 14% on the surgical side. “Injectables continued to be the most sought-after treatments in 2020,” the ASPS said, with survey respondents citing “a significant uptick in demand during the coronavirus pandemic.”
OnabotuliumtoxinA injection, the most popular form of minimally invasive procedure, was down by 13% from 2019, while use of soft-tissue fillers fell by 11%. Laser skin resurfacing was third in popularity and had the smallest drop, just 8%, among the top five from 2019 to 2020, the ASPS data show.
The drop in volume for chemical peels was large enough (33%), to move it from third place in 2019 to fourth in 2020, and a slightly less than average drop of 12% moved intense pulsed-light treatments from sixth place in 2019 to fifth in 2020, switching places with laser hair removal (down 28%), the ASPS reported.
Among the surgical procedures, rhinoplasty was the most popular in 2020, as it was in 2019, after dropping by just 3%. Blepharoplasty was down by 8% from 2019, but two other common procedures, liposuction and breast augmentation, fell by 20% and 33%, respectively, the ASPS said.
pandemic, according to the American Society of Plastic Surgeons.

There were an estimated 15.6 million cosmetic procedures performed in 2020, compared with 18.4 million in 2019, a drop of 15.2%. Meanwhile, society members reported that they stopped performing elective surgery for an average of 8.1 weeks, which works out to 15.6% of a 52-week year, the ASPS said in its annual statistics report.
“The pandemic isn’t over, but thanks to vaccines, a new normal is starting to define itself – and some surgeons’ offices that were closed or offered only limited services within the last year are seeing higher demand,” Lynn Jeffers, MD, MBA, immediate past president of the ASPS, said in a written statement.
Minimally invasive procedures, which made up the majority of cosmetic procedures in 2020, dropped by a slightly higher 16%, compared with 14% on the surgical side. “Injectables continued to be the most sought-after treatments in 2020,” the ASPS said, with survey respondents citing “a significant uptick in demand during the coronavirus pandemic.”
OnabotuliumtoxinA injection, the most popular form of minimally invasive procedure, was down by 13% from 2019, while use of soft-tissue fillers fell by 11%. Laser skin resurfacing was third in popularity and had the smallest drop, just 8%, among the top five from 2019 to 2020, the ASPS data show.
The drop in volume for chemical peels was large enough (33%), to move it from third place in 2019 to fourth in 2020, and a slightly less than average drop of 12% moved intense pulsed-light treatments from sixth place in 2019 to fifth in 2020, switching places with laser hair removal (down 28%), the ASPS reported.
Among the surgical procedures, rhinoplasty was the most popular in 2020, as it was in 2019, after dropping by just 3%. Blepharoplasty was down by 8% from 2019, but two other common procedures, liposuction and breast augmentation, fell by 20% and 33%, respectively, the ASPS said.
FDA panel narrowly backs avacopan approval
A panel of federal advisers on May 6 lent support to the ChemoCentryx bid for approval of avacopan for a rare and serious autoimmune condition. But they also flagged concerns about both the evidence supporting claims of a benefit for this experimental drug and its safety.
At a meeting of the Food and Drug Administration’s Arthritis Advisory Committee, panelists voted 10-8 on a question of whether the risk-benefit profile of avacopan is adequate to support approval.
ChemoCentryx is seeking approval of avacopan for antineutrophil cytoplasmic autoantibody (ANCA)–associated vasculitis in the subtypes of granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA).
Regardless of their vote on this approval question, the panelists shared an interest in avacopan’s potential to reduce glucocorticoid use among some patients with ANCA-associated vasculitis, also called AAV. Mara L. Becker, MD, MSCE, the chair of the FDA’s panel, was among the panelists who said they reluctantly voted no.
“It pains me because I really want more steroid-sparing” medicines, said Dr. Becker of Duke University, Durham, N.C., who cited a need to gather more data on avacopan.
Margrit Wiesendanger, MD, PhD, of the Icahn School of Medicine at Mount Sinai, New York, who was among the panelists voting yes, spoke of a need for caution if the FDA approves avacopan.
“Judicious use of this new medication will be warranted and perhaps additional guidance could be given to rheumatologists to help them decide for whom this medication is best,” she said.
Panelists had spoken earlier of avacopan as a possible alternative medicine for people with AAV who have conditions that make glucocorticoids riskier for them, such as those who have diabetes.
Close votes on safety profile, efficacy
The panel also voted 10-8 on a question about whether the safety profile of avacopan is adequate to support approval of avacopan for the treatment of adult patients with AAV.
In addition, the panel voted 9-9 on a question about whether efficacy data support approval of avacopan for the treatment of adult patients with AAV.
The FDA considers the recommendations of its advisory panels, but is not bound by them.
The FDA staff clearly expressed the view that ChemoCentryx fell short with the evidence presented for avacopan approval. Shares of San Carlos, Calif.–based ChemoCentryx dropped sharply from a May 3 closing price of $48.82 to a May 4 closing price of $26.63 after the FDA released the staff’s review of avacopan.
In a briefing prepared for the meeting, FDA staff detailed concerns about the evidence ChemoCentryx is using to seek approval. While acknowledging a need for new treatments for AAV as a rare condition, FDA staff honed in on what they described flaws in the testing of this experimental medicine, which is a small-molecule antagonist of the receptor of C5a, an end product of the complement cascade that acts as a potent neutrophil chemoattractant and agonist.
The FDA usually requires two phase 3 studies for approval of a new medicine but will do so with a single trial in cases of exceptional need, the agency staff said. But in these cases, the bar rises for the evidence provided from that single trial.
Difficulties in interpretation of complex study design
In the case of avacopan, though, the data from the key avacopan trial, Study CL010_168, known as ADVOCATE, there were substantial uncertainties around the phase 3 study design and results, raising questions about the adequacy of this single trial to inform the benefit-risk assessment.
In the briefing document, the FDA staff noted that it had “communicated many of the concerns” about ChemoCentryx’s research earlier to the company.
“Complexities of the study design, as detailed in the briefing document, raise questions about the interpretability of the data to define a clinically meaningful benefit of avacopan and its role in the management of AAV,” the FDA staff wrote.
“We acknowledge that AAV is a rare and serious disease associated with high morbidity and increased mortality. It is also a disease with high unmet need for new therapies. However, FDA wants to ensure that new products have a defined context of use, i.e., how a product would be used, and a favorable benefit-risk assessment for patients,” the staff added.
In addition, there were differences in the assessments performed by investigators and the adjudication committee, most frequently related to the attribution of persistent vasculitis, the FDA staff noted.
Statistical analyses of the primary endpoint using investigators’ estimates “resulted in more conservative estimates of treatment effect, e.g., statistical significance for superiority would no longer be demonstrated,” the FDA staff noted. “While the prespecified analysis used the Adjudicator assessments, the assessment based on the Investigators, experienced in management of vasculitis, may better reflect real-world use.”
Imbalances in use of glucocorticoids and maintenance therapy
Also among the complications in assessing the ADVOCATE trial data were the glucocorticoids taken by patients in the study, the FDA staff said.
In the avacopan arm of the trial, 86% of patients received non–study-supplied glucocorticoids. In addition, more avacopan‐treated patients experienced adverse events and serious adverse events within the hepatobiliary system leading to discontinuation.
Subgroups given different treatments represented another challenge in interpreting ADVOCATE results for the FDA staff.
At week 26, the proportion of patients in disease remission in the avacopan group (72.3%) was noninferior to the prednisone group (70.1%), the FDA staff said in the briefing document.
But at week 52, a disparity was observed between subgroups that had received rituximab and cyclophosphamide (intravenous and oral) induction treatment. The estimated risk difference for disease remission at week 52 was 15.0% (95% CI, 2.2%-27.7%) in the subgroup receiving induction with rituximab and 3.3% (95% CI, –14.8% to 21.4%) in the cyclophosphamide plus maintenance azathioprine subgroup, the agency’s staff said.
“Based on the data, there is no evidence of clinically meaningful treatment effect in the cyclophosphamide induction subgroup,” the FDA staff wrote. “Further, the treatment comparison in the complementary rituximab induction subgroup may not be considered meaningful because these patients did not receive maintenance therapy, i.e., due to undertreating of patients, the effect observed in the rituximab subgroup may not represent a clinically meaningful treatment effect, compared to standard of care.”
Rachel L. Glaser, MD, clinical team leader in FDA’s division of rheumatology and transplant medicine, reiterated these concerns to the advisory committee at the May 6 meeting.
“Throughout the development program, FDA advised the applicant that a noninferiority comparison would not be sufficient to show that avacopan can replaced glucocorticoids as it would be difficult to establish whether avacopan is effective or whether an effect was due to the rituximab or cyclophosphamide administered to both treatment arms,” she said.
In its briefing for the meeting, ChemoCentryx noted the limits of treatments now available for AAV. It also emphasized the toll of the condition, ranging from skin manifestations to glomerulonephritis to life-threatening pulmonary hemorrhage. If untreated, 80% of patients with GPA or MPA die within 2 years of disease onset, ChemoCentryx said in its briefing materials for the meeting.
The side effects of glucocorticoids were well known to the FDA panelists and the ChemoCentryx presenters. Witnesses at an open public hearing told their own stories of depression, anxiety, and irritability caused by these medicines.
During the ChemoCentryx presentation, a presenter for the company, Peter Merkel, MD, MPH, of the University of Pennsylvania, Philadelphia, said avacopan would provide patients with AAV with an alternative allowing them “to go on a much lower glucocorticoids regimen.”
A similar view was presented in a February 2021 editorial in the New England Journal of Medicine, titled “Avacopan – Time to Replace Glucocorticoids?” Written by Kenneth J. Warrington, MD, of the Mayo Clinic, Rochester, Minn., the opinion article called the ADVOCATE trial “a milestone in the treatment of ANCA-associated vasculitis; complement inhibition with avacopan has glucocorticoid-sparing effects and results in superior disease control.”
Dr. Warrington reported no conflicts in connection with his editorial nor payments from ChemoCentryx. He did report grants from other firms such as Eli Lilly.
Julia Lewis, MD, of Vanderbilt University, Nashville, Tenn., was among the more skeptical members of the FDA panel. She was among the “nays” in all three voting questions put to the panel. Still, she said there were signs of “clinically meaningful benefit” in the data presented, but noted that the nonstudy use of glucocorticoids made it difficult to interpret the ADVOCATE results.
Dr. Lewis noted that the FDA usually requires two studies for a drug approval, particularly with a compound not yet cleared for any use. While ANCA-associated vasculitis is rare, it would be possible to recruit patients for another trial of avacopan, adding to the results reported already for avacopan from ADVOCATE, she said.
“Were there to be another study, this would certainly be a supportive study and maybe qualify as two studies,” she said.
A panel of federal advisers on May 6 lent support to the ChemoCentryx bid for approval of avacopan for a rare and serious autoimmune condition. But they also flagged concerns about both the evidence supporting claims of a benefit for this experimental drug and its safety.
At a meeting of the Food and Drug Administration’s Arthritis Advisory Committee, panelists voted 10-8 on a question of whether the risk-benefit profile of avacopan is adequate to support approval.
ChemoCentryx is seeking approval of avacopan for antineutrophil cytoplasmic autoantibody (ANCA)–associated vasculitis in the subtypes of granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA).
Regardless of their vote on this approval question, the panelists shared an interest in avacopan’s potential to reduce glucocorticoid use among some patients with ANCA-associated vasculitis, also called AAV. Mara L. Becker, MD, MSCE, the chair of the FDA’s panel, was among the panelists who said they reluctantly voted no.
“It pains me because I really want more steroid-sparing” medicines, said Dr. Becker of Duke University, Durham, N.C., who cited a need to gather more data on avacopan.
Margrit Wiesendanger, MD, PhD, of the Icahn School of Medicine at Mount Sinai, New York, who was among the panelists voting yes, spoke of a need for caution if the FDA approves avacopan.
“Judicious use of this new medication will be warranted and perhaps additional guidance could be given to rheumatologists to help them decide for whom this medication is best,” she said.
Panelists had spoken earlier of avacopan as a possible alternative medicine for people with AAV who have conditions that make glucocorticoids riskier for them, such as those who have diabetes.
Close votes on safety profile, efficacy
The panel also voted 10-8 on a question about whether the safety profile of avacopan is adequate to support approval of avacopan for the treatment of adult patients with AAV.
In addition, the panel voted 9-9 on a question about whether efficacy data support approval of avacopan for the treatment of adult patients with AAV.
The FDA considers the recommendations of its advisory panels, but is not bound by them.
The FDA staff clearly expressed the view that ChemoCentryx fell short with the evidence presented for avacopan approval. Shares of San Carlos, Calif.–based ChemoCentryx dropped sharply from a May 3 closing price of $48.82 to a May 4 closing price of $26.63 after the FDA released the staff’s review of avacopan.
In a briefing prepared for the meeting, FDA staff detailed concerns about the evidence ChemoCentryx is using to seek approval. While acknowledging a need for new treatments for AAV as a rare condition, FDA staff honed in on what they described flaws in the testing of this experimental medicine, which is a small-molecule antagonist of the receptor of C5a, an end product of the complement cascade that acts as a potent neutrophil chemoattractant and agonist.
The FDA usually requires two phase 3 studies for approval of a new medicine but will do so with a single trial in cases of exceptional need, the agency staff said. But in these cases, the bar rises for the evidence provided from that single trial.
Difficulties in interpretation of complex study design
In the case of avacopan, though, the data from the key avacopan trial, Study CL010_168, known as ADVOCATE, there were substantial uncertainties around the phase 3 study design and results, raising questions about the adequacy of this single trial to inform the benefit-risk assessment.
In the briefing document, the FDA staff noted that it had “communicated many of the concerns” about ChemoCentryx’s research earlier to the company.
“Complexities of the study design, as detailed in the briefing document, raise questions about the interpretability of the data to define a clinically meaningful benefit of avacopan and its role in the management of AAV,” the FDA staff wrote.
“We acknowledge that AAV is a rare and serious disease associated with high morbidity and increased mortality. It is also a disease with high unmet need for new therapies. However, FDA wants to ensure that new products have a defined context of use, i.e., how a product would be used, and a favorable benefit-risk assessment for patients,” the staff added.
In addition, there were differences in the assessments performed by investigators and the adjudication committee, most frequently related to the attribution of persistent vasculitis, the FDA staff noted.
Statistical analyses of the primary endpoint using investigators’ estimates “resulted in more conservative estimates of treatment effect, e.g., statistical significance for superiority would no longer be demonstrated,” the FDA staff noted. “While the prespecified analysis used the Adjudicator assessments, the assessment based on the Investigators, experienced in management of vasculitis, may better reflect real-world use.”
Imbalances in use of glucocorticoids and maintenance therapy
Also among the complications in assessing the ADVOCATE trial data were the glucocorticoids taken by patients in the study, the FDA staff said.
In the avacopan arm of the trial, 86% of patients received non–study-supplied glucocorticoids. In addition, more avacopan‐treated patients experienced adverse events and serious adverse events within the hepatobiliary system leading to discontinuation.
Subgroups given different treatments represented another challenge in interpreting ADVOCATE results for the FDA staff.
At week 26, the proportion of patients in disease remission in the avacopan group (72.3%) was noninferior to the prednisone group (70.1%), the FDA staff said in the briefing document.
But at week 52, a disparity was observed between subgroups that had received rituximab and cyclophosphamide (intravenous and oral) induction treatment. The estimated risk difference for disease remission at week 52 was 15.0% (95% CI, 2.2%-27.7%) in the subgroup receiving induction with rituximab and 3.3% (95% CI, –14.8% to 21.4%) in the cyclophosphamide plus maintenance azathioprine subgroup, the agency’s staff said.
“Based on the data, there is no evidence of clinically meaningful treatment effect in the cyclophosphamide induction subgroup,” the FDA staff wrote. “Further, the treatment comparison in the complementary rituximab induction subgroup may not be considered meaningful because these patients did not receive maintenance therapy, i.e., due to undertreating of patients, the effect observed in the rituximab subgroup may not represent a clinically meaningful treatment effect, compared to standard of care.”
Rachel L. Glaser, MD, clinical team leader in FDA’s division of rheumatology and transplant medicine, reiterated these concerns to the advisory committee at the May 6 meeting.
“Throughout the development program, FDA advised the applicant that a noninferiority comparison would not be sufficient to show that avacopan can replaced glucocorticoids as it would be difficult to establish whether avacopan is effective or whether an effect was due to the rituximab or cyclophosphamide administered to both treatment arms,” she said.
In its briefing for the meeting, ChemoCentryx noted the limits of treatments now available for AAV. It also emphasized the toll of the condition, ranging from skin manifestations to glomerulonephritis to life-threatening pulmonary hemorrhage. If untreated, 80% of patients with GPA or MPA die within 2 years of disease onset, ChemoCentryx said in its briefing materials for the meeting.
The side effects of glucocorticoids were well known to the FDA panelists and the ChemoCentryx presenters. Witnesses at an open public hearing told their own stories of depression, anxiety, and irritability caused by these medicines.
During the ChemoCentryx presentation, a presenter for the company, Peter Merkel, MD, MPH, of the University of Pennsylvania, Philadelphia, said avacopan would provide patients with AAV with an alternative allowing them “to go on a much lower glucocorticoids regimen.”
A similar view was presented in a February 2021 editorial in the New England Journal of Medicine, titled “Avacopan – Time to Replace Glucocorticoids?” Written by Kenneth J. Warrington, MD, of the Mayo Clinic, Rochester, Minn., the opinion article called the ADVOCATE trial “a milestone in the treatment of ANCA-associated vasculitis; complement inhibition with avacopan has glucocorticoid-sparing effects and results in superior disease control.”
Dr. Warrington reported no conflicts in connection with his editorial nor payments from ChemoCentryx. He did report grants from other firms such as Eli Lilly.
Julia Lewis, MD, of Vanderbilt University, Nashville, Tenn., was among the more skeptical members of the FDA panel. She was among the “nays” in all three voting questions put to the panel. Still, she said there were signs of “clinically meaningful benefit” in the data presented, but noted that the nonstudy use of glucocorticoids made it difficult to interpret the ADVOCATE results.
Dr. Lewis noted that the FDA usually requires two studies for a drug approval, particularly with a compound not yet cleared for any use. While ANCA-associated vasculitis is rare, it would be possible to recruit patients for another trial of avacopan, adding to the results reported already for avacopan from ADVOCATE, she said.
“Were there to be another study, this would certainly be a supportive study and maybe qualify as two studies,” she said.
A panel of federal advisers on May 6 lent support to the ChemoCentryx bid for approval of avacopan for a rare and serious autoimmune condition. But they also flagged concerns about both the evidence supporting claims of a benefit for this experimental drug and its safety.
At a meeting of the Food and Drug Administration’s Arthritis Advisory Committee, panelists voted 10-8 on a question of whether the risk-benefit profile of avacopan is adequate to support approval.
ChemoCentryx is seeking approval of avacopan for antineutrophil cytoplasmic autoantibody (ANCA)–associated vasculitis in the subtypes of granulomatosis with polyangiitis (GPA) and microscopic polyangiitis (MPA).
Regardless of their vote on this approval question, the panelists shared an interest in avacopan’s potential to reduce glucocorticoid use among some patients with ANCA-associated vasculitis, also called AAV. Mara L. Becker, MD, MSCE, the chair of the FDA’s panel, was among the panelists who said they reluctantly voted no.
“It pains me because I really want more steroid-sparing” medicines, said Dr. Becker of Duke University, Durham, N.C., who cited a need to gather more data on avacopan.
Margrit Wiesendanger, MD, PhD, of the Icahn School of Medicine at Mount Sinai, New York, who was among the panelists voting yes, spoke of a need for caution if the FDA approves avacopan.
“Judicious use of this new medication will be warranted and perhaps additional guidance could be given to rheumatologists to help them decide for whom this medication is best,” she said.
Panelists had spoken earlier of avacopan as a possible alternative medicine for people with AAV who have conditions that make glucocorticoids riskier for them, such as those who have diabetes.
Close votes on safety profile, efficacy
The panel also voted 10-8 on a question about whether the safety profile of avacopan is adequate to support approval of avacopan for the treatment of adult patients with AAV.
In addition, the panel voted 9-9 on a question about whether efficacy data support approval of avacopan for the treatment of adult patients with AAV.
The FDA considers the recommendations of its advisory panels, but is not bound by them.
The FDA staff clearly expressed the view that ChemoCentryx fell short with the evidence presented for avacopan approval. Shares of San Carlos, Calif.–based ChemoCentryx dropped sharply from a May 3 closing price of $48.82 to a May 4 closing price of $26.63 after the FDA released the staff’s review of avacopan.
In a briefing prepared for the meeting, FDA staff detailed concerns about the evidence ChemoCentryx is using to seek approval. While acknowledging a need for new treatments for AAV as a rare condition, FDA staff honed in on what they described flaws in the testing of this experimental medicine, which is a small-molecule antagonist of the receptor of C5a, an end product of the complement cascade that acts as a potent neutrophil chemoattractant and agonist.
The FDA usually requires two phase 3 studies for approval of a new medicine but will do so with a single trial in cases of exceptional need, the agency staff said. But in these cases, the bar rises for the evidence provided from that single trial.
Difficulties in interpretation of complex study design
In the case of avacopan, though, the data from the key avacopan trial, Study CL010_168, known as ADVOCATE, there were substantial uncertainties around the phase 3 study design and results, raising questions about the adequacy of this single trial to inform the benefit-risk assessment.
In the briefing document, the FDA staff noted that it had “communicated many of the concerns” about ChemoCentryx’s research earlier to the company.
“Complexities of the study design, as detailed in the briefing document, raise questions about the interpretability of the data to define a clinically meaningful benefit of avacopan and its role in the management of AAV,” the FDA staff wrote.
“We acknowledge that AAV is a rare and serious disease associated with high morbidity and increased mortality. It is also a disease with high unmet need for new therapies. However, FDA wants to ensure that new products have a defined context of use, i.e., how a product would be used, and a favorable benefit-risk assessment for patients,” the staff added.
In addition, there were differences in the assessments performed by investigators and the adjudication committee, most frequently related to the attribution of persistent vasculitis, the FDA staff noted.
Statistical analyses of the primary endpoint using investigators’ estimates “resulted in more conservative estimates of treatment effect, e.g., statistical significance for superiority would no longer be demonstrated,” the FDA staff noted. “While the prespecified analysis used the Adjudicator assessments, the assessment based on the Investigators, experienced in management of vasculitis, may better reflect real-world use.”
Imbalances in use of glucocorticoids and maintenance therapy
Also among the complications in assessing the ADVOCATE trial data were the glucocorticoids taken by patients in the study, the FDA staff said.
In the avacopan arm of the trial, 86% of patients received non–study-supplied glucocorticoids. In addition, more avacopan‐treated patients experienced adverse events and serious adverse events within the hepatobiliary system leading to discontinuation.
Subgroups given different treatments represented another challenge in interpreting ADVOCATE results for the FDA staff.
At week 26, the proportion of patients in disease remission in the avacopan group (72.3%) was noninferior to the prednisone group (70.1%), the FDA staff said in the briefing document.
But at week 52, a disparity was observed between subgroups that had received rituximab and cyclophosphamide (intravenous and oral) induction treatment. The estimated risk difference for disease remission at week 52 was 15.0% (95% CI, 2.2%-27.7%) in the subgroup receiving induction with rituximab and 3.3% (95% CI, –14.8% to 21.4%) in the cyclophosphamide plus maintenance azathioprine subgroup, the agency’s staff said.
“Based on the data, there is no evidence of clinically meaningful treatment effect in the cyclophosphamide induction subgroup,” the FDA staff wrote. “Further, the treatment comparison in the complementary rituximab induction subgroup may not be considered meaningful because these patients did not receive maintenance therapy, i.e., due to undertreating of patients, the effect observed in the rituximab subgroup may not represent a clinically meaningful treatment effect, compared to standard of care.”
Rachel L. Glaser, MD, clinical team leader in FDA’s division of rheumatology and transplant medicine, reiterated these concerns to the advisory committee at the May 6 meeting.
“Throughout the development program, FDA advised the applicant that a noninferiority comparison would not be sufficient to show that avacopan can replaced glucocorticoids as it would be difficult to establish whether avacopan is effective or whether an effect was due to the rituximab or cyclophosphamide administered to both treatment arms,” she said.
In its briefing for the meeting, ChemoCentryx noted the limits of treatments now available for AAV. It also emphasized the toll of the condition, ranging from skin manifestations to glomerulonephritis to life-threatening pulmonary hemorrhage. If untreated, 80% of patients with GPA or MPA die within 2 years of disease onset, ChemoCentryx said in its briefing materials for the meeting.
The side effects of glucocorticoids were well known to the FDA panelists and the ChemoCentryx presenters. Witnesses at an open public hearing told their own stories of depression, anxiety, and irritability caused by these medicines.
During the ChemoCentryx presentation, a presenter for the company, Peter Merkel, MD, MPH, of the University of Pennsylvania, Philadelphia, said avacopan would provide patients with AAV with an alternative allowing them “to go on a much lower glucocorticoids regimen.”
A similar view was presented in a February 2021 editorial in the New England Journal of Medicine, titled “Avacopan – Time to Replace Glucocorticoids?” Written by Kenneth J. Warrington, MD, of the Mayo Clinic, Rochester, Minn., the opinion article called the ADVOCATE trial “a milestone in the treatment of ANCA-associated vasculitis; complement inhibition with avacopan has glucocorticoid-sparing effects and results in superior disease control.”
Dr. Warrington reported no conflicts in connection with his editorial nor payments from ChemoCentryx. He did report grants from other firms such as Eli Lilly.
Julia Lewis, MD, of Vanderbilt University, Nashville, Tenn., was among the more skeptical members of the FDA panel. She was among the “nays” in all three voting questions put to the panel. Still, she said there were signs of “clinically meaningful benefit” in the data presented, but noted that the nonstudy use of glucocorticoids made it difficult to interpret the ADVOCATE results.
Dr. Lewis noted that the FDA usually requires two studies for a drug approval, particularly with a compound not yet cleared for any use. While ANCA-associated vasculitis is rare, it would be possible to recruit patients for another trial of avacopan, adding to the results reported already for avacopan from ADVOCATE, she said.
“Were there to be another study, this would certainly be a supportive study and maybe qualify as two studies,” she said.
Atopic dermatitis genes vary with ethnicity
patients, researchers say.
The finding moves researchers another step forward in the effort to figure out which patients are most at risk for the disease and who will respond best to which treatments.
“Because atopic dermatitis is considered a complex trait, we think if there is any method to detect AD gene variations simultaneously, it could be possible to prevent the development of AD and then the atopic march,” said Eung Ho Choi, MD, PhD, a dermatology professor at Yonsei University, Wonju, South Korea.
He presented the finding at the International Society of Atopic Dermatitis (ISAD) 2021 Annual Meeting.
Atopic dermatitis is not caused by a single genetic mutation. But genetic factors play an important role, with about 75% concordance between monozygotic twins versus only 23% for dizygotic twins.
“Genetic biomarkers are needed in predicting the occurrence, severity, and treatment response,” as well as determining the prognosis of atopic dermatitis “and applying it to precision medicine,” Dr. Choi said.
Researchers have identified multiple genetic variations related to atopic dermatitis. One of the most significant genetic contributions found so far is the filaggrin gene variation, which can produce a defective skin barrier, Dr. Choi said. Others are involved in the immune response.
Although variations in the filaggrin gene (FLG ) are the most reliable genetic predictor of atopic dermatitis in Korean patients, they are less common in Korean patients than in Northwestern Europeans, Chinese, and Japanese patients. In Korean patients, the most common reported mutations of this gene are 3321delA and K4022X, Dr. Choi said.
To find out what other gene variants are important in Korean patients with atopic dermatitis, Dr. Choi and his colleagues developed the reverse blot hybridization assay (REBA) to detect skin barrier variations in the FLG, SPINK5 and KLK7 genes, and genes involved in immune response variations, KDR, IL-5RA, IL-9, DEFB1 (Defensin Beta 1), IL-12RB1 (interleukin-12 receptor subunit beta 1), and IL-12RB2.
They compared the prevalence of these variations in 279 Koreans with atopic dermatitis to the prevalence in 224 healthy people without atopic dermatitis and found that the odds ratio for atopic dermatitis increased with the number of these variants: People with three or four variants had a 3.75 times greater risk of AD, and those with 5 or more variants had a 10.3 times greater risk. The number of variants did not correlate to the severity of the disease, however.
The filaggrin variation was present in 13.9% of those with atopic dermatitis. About a quarter (28%) of the patients with AD who had this variation had impetigo, 15% had eczema herpeticum, and 5% had prurigo nodularis. By comparison, 14% of the patients with AD who did not have this variation had impetigo, and 5% had eczema herpeticum, but 19% had prurigo nodularis.
In a separate study, Dr. Choi and his colleagues identified a mutation in IL-17RA, present in 8.1% of 332 patients with AD compared with 3.3% of 245 controls. The patients with IL-17RA mutations all had extrinsic AD.
The variation was associated with longer disease duration, more frequent keratosis pilaris, higher blood eosinophil counts, higher serum total immunoglobulin E (IgE) levels, higher house dust mite allergen-specific IgE levels, and a greater need for systemic treatment than patients without the IL-17RA mutation.
Such findings are important for progress in treating atopic dermatitis because the mechanism differs among patients, said Emma Guttman-Yassky, MD, PhD, director of the Center for Excellence in Eczema and professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
“It’s not one size fits all in atopic dermatitis, and we need better biomarkers that will be able to tell us which treatment will work best for each patient,” she said in an interview.
In addition to genetic biomarkers, she and her colleagues are analyzing proteins involved in inflammation. They are using adhesive tape strips to harvest these markers, a less invasive approach than skin biopsies.
A version of this article first appeared on Medscape.com.
patients, researchers say.
The finding moves researchers another step forward in the effort to figure out which patients are most at risk for the disease and who will respond best to which treatments.
“Because atopic dermatitis is considered a complex trait, we think if there is any method to detect AD gene variations simultaneously, it could be possible to prevent the development of AD and then the atopic march,” said Eung Ho Choi, MD, PhD, a dermatology professor at Yonsei University, Wonju, South Korea.
He presented the finding at the International Society of Atopic Dermatitis (ISAD) 2021 Annual Meeting.
Atopic dermatitis is not caused by a single genetic mutation. But genetic factors play an important role, with about 75% concordance between monozygotic twins versus only 23% for dizygotic twins.
“Genetic biomarkers are needed in predicting the occurrence, severity, and treatment response,” as well as determining the prognosis of atopic dermatitis “and applying it to precision medicine,” Dr. Choi said.
Researchers have identified multiple genetic variations related to atopic dermatitis. One of the most significant genetic contributions found so far is the filaggrin gene variation, which can produce a defective skin barrier, Dr. Choi said. Others are involved in the immune response.
Although variations in the filaggrin gene (FLG ) are the most reliable genetic predictor of atopic dermatitis in Korean patients, they are less common in Korean patients than in Northwestern Europeans, Chinese, and Japanese patients. In Korean patients, the most common reported mutations of this gene are 3321delA and K4022X, Dr. Choi said.
To find out what other gene variants are important in Korean patients with atopic dermatitis, Dr. Choi and his colleagues developed the reverse blot hybridization assay (REBA) to detect skin barrier variations in the FLG, SPINK5 and KLK7 genes, and genes involved in immune response variations, KDR, IL-5RA, IL-9, DEFB1 (Defensin Beta 1), IL-12RB1 (interleukin-12 receptor subunit beta 1), and IL-12RB2.
They compared the prevalence of these variations in 279 Koreans with atopic dermatitis to the prevalence in 224 healthy people without atopic dermatitis and found that the odds ratio for atopic dermatitis increased with the number of these variants: People with three or four variants had a 3.75 times greater risk of AD, and those with 5 or more variants had a 10.3 times greater risk. The number of variants did not correlate to the severity of the disease, however.
The filaggrin variation was present in 13.9% of those with atopic dermatitis. About a quarter (28%) of the patients with AD who had this variation had impetigo, 15% had eczema herpeticum, and 5% had prurigo nodularis. By comparison, 14% of the patients with AD who did not have this variation had impetigo, and 5% had eczema herpeticum, but 19% had prurigo nodularis.
In a separate study, Dr. Choi and his colleagues identified a mutation in IL-17RA, present in 8.1% of 332 patients with AD compared with 3.3% of 245 controls. The patients with IL-17RA mutations all had extrinsic AD.
The variation was associated with longer disease duration, more frequent keratosis pilaris, higher blood eosinophil counts, higher serum total immunoglobulin E (IgE) levels, higher house dust mite allergen-specific IgE levels, and a greater need for systemic treatment than patients without the IL-17RA mutation.
Such findings are important for progress in treating atopic dermatitis because the mechanism differs among patients, said Emma Guttman-Yassky, MD, PhD, director of the Center for Excellence in Eczema and professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
“It’s not one size fits all in atopic dermatitis, and we need better biomarkers that will be able to tell us which treatment will work best for each patient,” she said in an interview.
In addition to genetic biomarkers, she and her colleagues are analyzing proteins involved in inflammation. They are using adhesive tape strips to harvest these markers, a less invasive approach than skin biopsies.
A version of this article first appeared on Medscape.com.
patients, researchers say.
The finding moves researchers another step forward in the effort to figure out which patients are most at risk for the disease and who will respond best to which treatments.
“Because atopic dermatitis is considered a complex trait, we think if there is any method to detect AD gene variations simultaneously, it could be possible to prevent the development of AD and then the atopic march,” said Eung Ho Choi, MD, PhD, a dermatology professor at Yonsei University, Wonju, South Korea.
He presented the finding at the International Society of Atopic Dermatitis (ISAD) 2021 Annual Meeting.
Atopic dermatitis is not caused by a single genetic mutation. But genetic factors play an important role, with about 75% concordance between monozygotic twins versus only 23% for dizygotic twins.
“Genetic biomarkers are needed in predicting the occurrence, severity, and treatment response,” as well as determining the prognosis of atopic dermatitis “and applying it to precision medicine,” Dr. Choi said.
Researchers have identified multiple genetic variations related to atopic dermatitis. One of the most significant genetic contributions found so far is the filaggrin gene variation, which can produce a defective skin barrier, Dr. Choi said. Others are involved in the immune response.
Although variations in the filaggrin gene (FLG ) are the most reliable genetic predictor of atopic dermatitis in Korean patients, they are less common in Korean patients than in Northwestern Europeans, Chinese, and Japanese patients. In Korean patients, the most common reported mutations of this gene are 3321delA and K4022X, Dr. Choi said.
To find out what other gene variants are important in Korean patients with atopic dermatitis, Dr. Choi and his colleagues developed the reverse blot hybridization assay (REBA) to detect skin barrier variations in the FLG, SPINK5 and KLK7 genes, and genes involved in immune response variations, KDR, IL-5RA, IL-9, DEFB1 (Defensin Beta 1), IL-12RB1 (interleukin-12 receptor subunit beta 1), and IL-12RB2.
They compared the prevalence of these variations in 279 Koreans with atopic dermatitis to the prevalence in 224 healthy people without atopic dermatitis and found that the odds ratio for atopic dermatitis increased with the number of these variants: People with three or four variants had a 3.75 times greater risk of AD, and those with 5 or more variants had a 10.3 times greater risk. The number of variants did not correlate to the severity of the disease, however.
The filaggrin variation was present in 13.9% of those with atopic dermatitis. About a quarter (28%) of the patients with AD who had this variation had impetigo, 15% had eczema herpeticum, and 5% had prurigo nodularis. By comparison, 14% of the patients with AD who did not have this variation had impetigo, and 5% had eczema herpeticum, but 19% had prurigo nodularis.
In a separate study, Dr. Choi and his colleagues identified a mutation in IL-17RA, present in 8.1% of 332 patients with AD compared with 3.3% of 245 controls. The patients with IL-17RA mutations all had extrinsic AD.
The variation was associated with longer disease duration, more frequent keratosis pilaris, higher blood eosinophil counts, higher serum total immunoglobulin E (IgE) levels, higher house dust mite allergen-specific IgE levels, and a greater need for systemic treatment than patients without the IL-17RA mutation.
Such findings are important for progress in treating atopic dermatitis because the mechanism differs among patients, said Emma Guttman-Yassky, MD, PhD, director of the Center for Excellence in Eczema and professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
“It’s not one size fits all in atopic dermatitis, and we need better biomarkers that will be able to tell us which treatment will work best for each patient,” she said in an interview.
In addition to genetic biomarkers, she and her colleagues are analyzing proteins involved in inflammation. They are using adhesive tape strips to harvest these markers, a less invasive approach than skin biopsies.
A version of this article first appeared on Medscape.com.
Who can call themselves ‘doctor’? The debate heats up
Who Should Get to Be Called ‘Doctor’? shows. The topic has clearly struck a nerve, since a record number of respondents – over 12,000 – voted in the poll.
Most physicians think it’s appropriate for people with other doctorate degrees such as a PhD or EdD to call themselves ‘doctor,’ although slightly more than half said it depends on the context.
The controversy over who gets to be called a doctor was reignited when a Wall Street Journal opinion piece criticized First Lady Jill Biden, EdD, for wanting to be called “Dr Biden.” The piece also challenged the idea that having a PhD is worth the honorific of ‘doctor.’
Medical ethicist Arthur Caplan, PhD, disagreed with that viewpoint, saying the context matters. For example, he prefers to be called “professor” when he’s introduced to the public rather than “doctor” to avoid any confusion about his professional status.
More than 12,000 clinicians including physicians, medical students, nurses, pharmacists, and other health care professionals responded to the poll. The non-MD clinicians were the most likely to say it was always appropriate to be called “doctor” while physicians were the least likely.
Context matters
Large percentages of clinicians – 54% of doctors, 62% of medical students, and 41% of nurses – said that the context matters for being called “doctor.’’
“I earned my PhD in 1995 and my MD in 2000. I think it is contextual. In a research or University setting, “Dr.” seems appropriate for a PhD. That same person in public should probably not hold themselves out as “Dr.” So, maybe MDs and DOs can choose, while others maintain the title in their specific setting.”
Some readers proposed that people with MDs call themselves physicians rather than doctors. Said one: “Anyone with a terminal doctorate degree has the right to use the word doctor. As a physician when someone asks what I do, I say: ‘I am a physician.’ Problem solved. There can only be one physician but there are many types of doctors.”
Physicians and nurses differed most in their views. Just 24% of physicians said it was always appropriate for people with other doctorate degrees to call themselves doctor whereas about an equal number (22%) thought it was never appropriate.
In contrast, 43% of nurses (including advance practice nurses) said it was always appropriate for people with non-MD doctorates to be called doctor. Only 16% said it’s never appropriate.
This difference may reflect the growing number of nurses with doctorate degrees, either a DNP or PhD, who want to be called doctor in clinical settings.
Age made a difference too. Only 16% of physicians younger than age 45 said it was always appropriate for people with non-MD doctorate degrees to be called doctor, compared with 27% of physicians aged 45 and up.
Medical students (31%) were also more likely than physicians to say it was always appropriate for non-MD doctorates to use the title “doctor” and 64% said it depends on the context. This was noteworthy because twice as many medical students as physicians (16% vs. 8%) said they work in academia, research, or military government settings.
Too many ‘doctors’ confuse the public
Physicians (70%) were also more likely to say it was always or often confusing for the public to hear someone without a medical degree addressed as “doctor.” Only 6% of physicians thought it was never or rarely confusing.
Nurses disagreed. Just 45% said that it was always or often confusing while 16% said it was never or rarely confusing.
Medical students were more aligned with physicians on this issue – 60% said it was always or often confusing to the public and just 10% said it was never or rarely confusing.
One reader commented, “The problem is the confusion the ‘doctor’ title causes for patients, especially in a hospital setting. Is the ‘doctor’ a physician, a pharmacist, a psychologist, a nurse, etc., etc.? We need to think not of our own egos but if and how the confusion about this plethora of titles may be hindering good patient care.”
These concerns are not unfounded. The American Medical Association reported in its Truth in Advertising campaign that “patients mistake physicians with nonphysician providers” based on an online survey of 802 adults in 2018. The participants thought these specialists were MDs: dentists (61%), podiatrists (67%), optometrists (47%), psychologists (43%), doctors of nursing (39%), and chiropractors (27%).
The AMA has advocated that states pass the “Health Care Professional Transparency Act,” which New Jersey has enacted. The law requires all health care professionals dealing with patients to wear a name tag that clearly identifies their licensure. Health care professionals must also display their education, training, and licensure in their office.
A version of this article first appeared on Medscape.com.
Who Should Get to Be Called ‘Doctor’? shows. The topic has clearly struck a nerve, since a record number of respondents – over 12,000 – voted in the poll.
Most physicians think it’s appropriate for people with other doctorate degrees such as a PhD or EdD to call themselves ‘doctor,’ although slightly more than half said it depends on the context.
The controversy over who gets to be called a doctor was reignited when a Wall Street Journal opinion piece criticized First Lady Jill Biden, EdD, for wanting to be called “Dr Biden.” The piece also challenged the idea that having a PhD is worth the honorific of ‘doctor.’
Medical ethicist Arthur Caplan, PhD, disagreed with that viewpoint, saying the context matters. For example, he prefers to be called “professor” when he’s introduced to the public rather than “doctor” to avoid any confusion about his professional status.
More than 12,000 clinicians including physicians, medical students, nurses, pharmacists, and other health care professionals responded to the poll. The non-MD clinicians were the most likely to say it was always appropriate to be called “doctor” while physicians were the least likely.
Context matters
Large percentages of clinicians – 54% of doctors, 62% of medical students, and 41% of nurses – said that the context matters for being called “doctor.’’
“I earned my PhD in 1995 and my MD in 2000. I think it is contextual. In a research or University setting, “Dr.” seems appropriate for a PhD. That same person in public should probably not hold themselves out as “Dr.” So, maybe MDs and DOs can choose, while others maintain the title in their specific setting.”
Some readers proposed that people with MDs call themselves physicians rather than doctors. Said one: “Anyone with a terminal doctorate degree has the right to use the word doctor. As a physician when someone asks what I do, I say: ‘I am a physician.’ Problem solved. There can only be one physician but there are many types of doctors.”
Physicians and nurses differed most in their views. Just 24% of physicians said it was always appropriate for people with other doctorate degrees to call themselves doctor whereas about an equal number (22%) thought it was never appropriate.
In contrast, 43% of nurses (including advance practice nurses) said it was always appropriate for people with non-MD doctorates to be called doctor. Only 16% said it’s never appropriate.
This difference may reflect the growing number of nurses with doctorate degrees, either a DNP or PhD, who want to be called doctor in clinical settings.
Age made a difference too. Only 16% of physicians younger than age 45 said it was always appropriate for people with non-MD doctorate degrees to be called doctor, compared with 27% of physicians aged 45 and up.
Medical students (31%) were also more likely than physicians to say it was always appropriate for non-MD doctorates to use the title “doctor” and 64% said it depends on the context. This was noteworthy because twice as many medical students as physicians (16% vs. 8%) said they work in academia, research, or military government settings.
Too many ‘doctors’ confuse the public
Physicians (70%) were also more likely to say it was always or often confusing for the public to hear someone without a medical degree addressed as “doctor.” Only 6% of physicians thought it was never or rarely confusing.
Nurses disagreed. Just 45% said that it was always or often confusing while 16% said it was never or rarely confusing.
Medical students were more aligned with physicians on this issue – 60% said it was always or often confusing to the public and just 10% said it was never or rarely confusing.
One reader commented, “The problem is the confusion the ‘doctor’ title causes for patients, especially in a hospital setting. Is the ‘doctor’ a physician, a pharmacist, a psychologist, a nurse, etc., etc.? We need to think not of our own egos but if and how the confusion about this plethora of titles may be hindering good patient care.”
These concerns are not unfounded. The American Medical Association reported in its Truth in Advertising campaign that “patients mistake physicians with nonphysician providers” based on an online survey of 802 adults in 2018. The participants thought these specialists were MDs: dentists (61%), podiatrists (67%), optometrists (47%), psychologists (43%), doctors of nursing (39%), and chiropractors (27%).
The AMA has advocated that states pass the “Health Care Professional Transparency Act,” which New Jersey has enacted. The law requires all health care professionals dealing with patients to wear a name tag that clearly identifies their licensure. Health care professionals must also display their education, training, and licensure in their office.
A version of this article first appeared on Medscape.com.
Who Should Get to Be Called ‘Doctor’? shows. The topic has clearly struck a nerve, since a record number of respondents – over 12,000 – voted in the poll.
Most physicians think it’s appropriate for people with other doctorate degrees such as a PhD or EdD to call themselves ‘doctor,’ although slightly more than half said it depends on the context.
The controversy over who gets to be called a doctor was reignited when a Wall Street Journal opinion piece criticized First Lady Jill Biden, EdD, for wanting to be called “Dr Biden.” The piece also challenged the idea that having a PhD is worth the honorific of ‘doctor.’
Medical ethicist Arthur Caplan, PhD, disagreed with that viewpoint, saying the context matters. For example, he prefers to be called “professor” when he’s introduced to the public rather than “doctor” to avoid any confusion about his professional status.
More than 12,000 clinicians including physicians, medical students, nurses, pharmacists, and other health care professionals responded to the poll. The non-MD clinicians were the most likely to say it was always appropriate to be called “doctor” while physicians were the least likely.
Context matters
Large percentages of clinicians – 54% of doctors, 62% of medical students, and 41% of nurses – said that the context matters for being called “doctor.’’
“I earned my PhD in 1995 and my MD in 2000. I think it is contextual. In a research or University setting, “Dr.” seems appropriate for a PhD. That same person in public should probably not hold themselves out as “Dr.” So, maybe MDs and DOs can choose, while others maintain the title in their specific setting.”
Some readers proposed that people with MDs call themselves physicians rather than doctors. Said one: “Anyone with a terminal doctorate degree has the right to use the word doctor. As a physician when someone asks what I do, I say: ‘I am a physician.’ Problem solved. There can only be one physician but there are many types of doctors.”
Physicians and nurses differed most in their views. Just 24% of physicians said it was always appropriate for people with other doctorate degrees to call themselves doctor whereas about an equal number (22%) thought it was never appropriate.
In contrast, 43% of nurses (including advance practice nurses) said it was always appropriate for people with non-MD doctorates to be called doctor. Only 16% said it’s never appropriate.
This difference may reflect the growing number of nurses with doctorate degrees, either a DNP or PhD, who want to be called doctor in clinical settings.
Age made a difference too. Only 16% of physicians younger than age 45 said it was always appropriate for people with non-MD doctorate degrees to be called doctor, compared with 27% of physicians aged 45 and up.
Medical students (31%) were also more likely than physicians to say it was always appropriate for non-MD doctorates to use the title “doctor” and 64% said it depends on the context. This was noteworthy because twice as many medical students as physicians (16% vs. 8%) said they work in academia, research, or military government settings.
Too many ‘doctors’ confuse the public
Physicians (70%) were also more likely to say it was always or often confusing for the public to hear someone without a medical degree addressed as “doctor.” Only 6% of physicians thought it was never or rarely confusing.
Nurses disagreed. Just 45% said that it was always or often confusing while 16% said it was never or rarely confusing.
Medical students were more aligned with physicians on this issue – 60% said it was always or often confusing to the public and just 10% said it was never or rarely confusing.
One reader commented, “The problem is the confusion the ‘doctor’ title causes for patients, especially in a hospital setting. Is the ‘doctor’ a physician, a pharmacist, a psychologist, a nurse, etc., etc.? We need to think not of our own egos but if and how the confusion about this plethora of titles may be hindering good patient care.”
These concerns are not unfounded. The American Medical Association reported in its Truth in Advertising campaign that “patients mistake physicians with nonphysician providers” based on an online survey of 802 adults in 2018. The participants thought these specialists were MDs: dentists (61%), podiatrists (67%), optometrists (47%), psychologists (43%), doctors of nursing (39%), and chiropractors (27%).
The AMA has advocated that states pass the “Health Care Professional Transparency Act,” which New Jersey has enacted. The law requires all health care professionals dealing with patients to wear a name tag that clearly identifies their licensure. Health care professionals must also display their education, training, and licensure in their office.
A version of this article first appeared on Medscape.com.
Checkpoint inhibitor skin side effects more common in women
of 235 patients at Dana Farber Cancer Center, Boston.
Overall, 62.4% of the 93 women in the review and 48.6% of the 142 men experienced confirmed skin reactions, for an odds ratio (OR) of 2.11 for women compared with men (P = .01).
“Clinicians should consider these results in counseling female patients regarding an elevated risk of dermatologic adverse events” when taking checkpoint inhibitors, said investigators led by Harvard University medical student Jordan Said, who presented the results at the American Academy of Dermatology Virtual Meeting Experience.
Autoimmune-like adverse events are common with checkpoint inhibitors. Dermatologic side effects occur in about half of people receiving monotherapy and more than that among patients receiving combination therapy.
Skin reactions can include psoriasiform dermatitis, lichenoid reactions, vitiligo, and bullous pemphigoid and may require hospitalization and prolonged steroid treatment.
Not much is known about risk factors for these reactions. A higher incidence among women has been previously reported. A 2019 study found a higher risk for pneumonitis and endocrinopathy, including hypophysitis, among women who underwent treatment for non–small cell lung cancer or metastatic melanoma.
The 2019 study found that the risk was higher among premenopausal women than postmenopausal women, which led some to suggest that estrogen may play a role.
The results of the Dana Farber review argue against that notion. In their review, the investigators found that the risk was similarly elevated among the 27 premenopausal women (OR, 1.97; P = .40) and the 66 postmenopausal women (OR, 2.17, P = .05). In the study, women who were aged 52 years or older at the start of treatment were considered to be postmenopausal.
“This suggests that factors beyond sex hormones are likely contributory” to the difference in risk between men and women. It’s known that women are at higher risk for autoimmune disease overall, which might be related to the increased odds of autoimmune-like reactions, and it may be that sex-related differences in innate and adoptive immunity are at work, Mr. Said noted.
When asked for comment, Douglas Johnson, MD, assistant professor of hematology/oncology at Vanderbilt University, Nashville, Tenn., said that although some studies have reported a greater risk for side effects among women, others have not. “Additional research is needed to determine the interactions between sex and effects of immune checkpoint inhibitors, as well as many other possible triggers of immune-related adverse events,” he said.
“Continued work in this area will be so important to help determine how to best counsel women and to ensure early recognition and intervention for dermatologic side effects,” said Bernice Kwong, MD, director of the Supportive Dermato-Oncology Program at Stanford (Calif.) University.
The patients in the review were treated from 2011 to 2016 and underwent at least monthly evaluations by their medical teams. They were taking either nivolumab, pembrolizumab, or ipilimumab or a nivolumab/ipilimumab combination.
The median age of the men in the study was 65 years; the median age of women was 60 years. Almost 98% of the participants were White. The majority received one to three infusions, most commonly with pembrolizumab monotherapy.
No funding for the study was reported. Mr. Said has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
of 235 patients at Dana Farber Cancer Center, Boston.
Overall, 62.4% of the 93 women in the review and 48.6% of the 142 men experienced confirmed skin reactions, for an odds ratio (OR) of 2.11 for women compared with men (P = .01).
“Clinicians should consider these results in counseling female patients regarding an elevated risk of dermatologic adverse events” when taking checkpoint inhibitors, said investigators led by Harvard University medical student Jordan Said, who presented the results at the American Academy of Dermatology Virtual Meeting Experience.
Autoimmune-like adverse events are common with checkpoint inhibitors. Dermatologic side effects occur in about half of people receiving monotherapy and more than that among patients receiving combination therapy.
Skin reactions can include psoriasiform dermatitis, lichenoid reactions, vitiligo, and bullous pemphigoid and may require hospitalization and prolonged steroid treatment.
Not much is known about risk factors for these reactions. A higher incidence among women has been previously reported. A 2019 study found a higher risk for pneumonitis and endocrinopathy, including hypophysitis, among women who underwent treatment for non–small cell lung cancer or metastatic melanoma.
The 2019 study found that the risk was higher among premenopausal women than postmenopausal women, which led some to suggest that estrogen may play a role.
The results of the Dana Farber review argue against that notion. In their review, the investigators found that the risk was similarly elevated among the 27 premenopausal women (OR, 1.97; P = .40) and the 66 postmenopausal women (OR, 2.17, P = .05). In the study, women who were aged 52 years or older at the start of treatment were considered to be postmenopausal.
“This suggests that factors beyond sex hormones are likely contributory” to the difference in risk between men and women. It’s known that women are at higher risk for autoimmune disease overall, which might be related to the increased odds of autoimmune-like reactions, and it may be that sex-related differences in innate and adoptive immunity are at work, Mr. Said noted.
When asked for comment, Douglas Johnson, MD, assistant professor of hematology/oncology at Vanderbilt University, Nashville, Tenn., said that although some studies have reported a greater risk for side effects among women, others have not. “Additional research is needed to determine the interactions between sex and effects of immune checkpoint inhibitors, as well as many other possible triggers of immune-related adverse events,” he said.
“Continued work in this area will be so important to help determine how to best counsel women and to ensure early recognition and intervention for dermatologic side effects,” said Bernice Kwong, MD, director of the Supportive Dermato-Oncology Program at Stanford (Calif.) University.
The patients in the review were treated from 2011 to 2016 and underwent at least monthly evaluations by their medical teams. They were taking either nivolumab, pembrolizumab, or ipilimumab or a nivolumab/ipilimumab combination.
The median age of the men in the study was 65 years; the median age of women was 60 years. Almost 98% of the participants were White. The majority received one to three infusions, most commonly with pembrolizumab monotherapy.
No funding for the study was reported. Mr. Said has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
of 235 patients at Dana Farber Cancer Center, Boston.
Overall, 62.4% of the 93 women in the review and 48.6% of the 142 men experienced confirmed skin reactions, for an odds ratio (OR) of 2.11 for women compared with men (P = .01).
“Clinicians should consider these results in counseling female patients regarding an elevated risk of dermatologic adverse events” when taking checkpoint inhibitors, said investigators led by Harvard University medical student Jordan Said, who presented the results at the American Academy of Dermatology Virtual Meeting Experience.
Autoimmune-like adverse events are common with checkpoint inhibitors. Dermatologic side effects occur in about half of people receiving monotherapy and more than that among patients receiving combination therapy.
Skin reactions can include psoriasiform dermatitis, lichenoid reactions, vitiligo, and bullous pemphigoid and may require hospitalization and prolonged steroid treatment.
Not much is known about risk factors for these reactions. A higher incidence among women has been previously reported. A 2019 study found a higher risk for pneumonitis and endocrinopathy, including hypophysitis, among women who underwent treatment for non–small cell lung cancer or metastatic melanoma.
The 2019 study found that the risk was higher among premenopausal women than postmenopausal women, which led some to suggest that estrogen may play a role.
The results of the Dana Farber review argue against that notion. In their review, the investigators found that the risk was similarly elevated among the 27 premenopausal women (OR, 1.97; P = .40) and the 66 postmenopausal women (OR, 2.17, P = .05). In the study, women who were aged 52 years or older at the start of treatment were considered to be postmenopausal.
“This suggests that factors beyond sex hormones are likely contributory” to the difference in risk between men and women. It’s known that women are at higher risk for autoimmune disease overall, which might be related to the increased odds of autoimmune-like reactions, and it may be that sex-related differences in innate and adoptive immunity are at work, Mr. Said noted.
When asked for comment, Douglas Johnson, MD, assistant professor of hematology/oncology at Vanderbilt University, Nashville, Tenn., said that although some studies have reported a greater risk for side effects among women, others have not. “Additional research is needed to determine the interactions between sex and effects of immune checkpoint inhibitors, as well as many other possible triggers of immune-related adverse events,” he said.
“Continued work in this area will be so important to help determine how to best counsel women and to ensure early recognition and intervention for dermatologic side effects,” said Bernice Kwong, MD, director of the Supportive Dermato-Oncology Program at Stanford (Calif.) University.
The patients in the review were treated from 2011 to 2016 and underwent at least monthly evaluations by their medical teams. They were taking either nivolumab, pembrolizumab, or ipilimumab or a nivolumab/ipilimumab combination.
The median age of the men in the study was 65 years; the median age of women was 60 years. Almost 98% of the participants were White. The majority received one to three infusions, most commonly with pembrolizumab monotherapy.
No funding for the study was reported. Mr. Said has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In pemphigus, phase 2 results with BTK inhibitor raise hopes for phase 3 trial
In patients with newly diagnosed or relapsing pemphigus vulgaris, an update of the phase 2 BELIEVE study with the Bruton tyrosine kinase (BTK) inhibitor rilzabrutinib has raised hopes that the ongoing phase 3 trial will confirm that this drug is a breakthrough therapy, according to an investigator who presented the data at the American Academy of Dermatology Virtual Meeting Experience.
Among the highlights of the phase 2 data presented during a late-breaking research session was that a substantial minority of patients achieved a complete response within 12 weeks of starting treatment with rilzabrutinib. Treatment was associated with mostly mild and transient adverse events, according to Dedee F. Murrell, MD, director of dermatology, St. George Hospital, University of New South Wales, Sydney.
Many of the phase 2 results have been presented previously and the phase 3 trial, called PEGASUS, has now completed enrollment.
Focusing on part A of the BELIEVE study, Dr. Murrell reported that about one-third of the 27 patients enrolled had newly diagnosed pemphigus. The remaining patients had relapsing disease after a mean 8.9 years after diagnosis. The disease was judged moderate to severe in 59%. The daily oral dose of rilzabrutinib ranged from 400 mg to 600 mg twice daily.
For the primary endpoint of control of disease activity (CDA), meaning no formation of new lesions with diminishing activity of existing lesions, 52% had responded by week 4 and 70% had responded by week 12, which was the end of active treatment. Responses at both time points were comparable among patients with newly diagnosed disease (56% at week 4 and 67% at week 12) relapsing disease (50% and 72%, respectively), moderate disease severity at baseline (55% and 64%, respectively) and more severe disease (50% and 75%, respectively), Dr. Murrell noted.
“A complete response was achieved by 22% of patients at week 4 and nearly 30% by the end of the study,” she said.
These response rates were reflected in the Pemphigus Disease Area Index (PDAI) and the Autoimmune Bullous Disease Quality of Life (ABQOL) Score. From a baseline score of 20, the PDAI fell to 10 at 4 weeks and then to 6 at 12 weeks in the newly diagnosed cohort. In the relapsing cohort, the score fell from a baseline of 18 to 13 at week 4 and then to 7 at week 12.
“The improvement corresponded to a reduction in steroid doses,” Dr. Murrell reported. By the end of the study, the mean daily dose of corticosteroids fell to 10 mg from a baseline of 20 mg. In a 12-week follow-up, corticosteroid doses rose slowly and did not reach baseline levels until about eight weeks after rilzabrutinib was discontinued.
The ABQOL scores fell most rapidly in the newly diagnosed cohort. By week 12, there was about a 6.6-point reduction. In the relapsing group, the score fell by 3.7 points from a similar baseline level. Both reductions are considered highly clinically meaningful, according to Dr. Murrell. At the end of the 12 weeks of follow-up after the drug was discontinued, ABQOL scores had increased but remained below the baseline.
Nausea was reported by 15% of patients, making it the most commonly reported adverse event. All cases were grade 1 severity. Three patients had grade 2 abdominal pain. The only grade 3 event in this series was a case of cellulitis in a patient who had developed steroid-induced diabetes mellitus. With treatment, the cellulitis resolved, and the patient completed the study.
Data from part B of the BELIEVE study, which was similarly designed and enrolled 15 patients with newly diagnosed or relapsing pemphigus vulgaris, was not updated by Dr. Murrell at the meeting, but these data have been presented before and showed similar results, including achievement of CDA in the majority of patients accompanied by a reduction in corticosteroid doses.
“In summary, rilzabrutinib produced a rapid clinical effect with an overall favorable benefit-to-risk profile,” said Dr. Murrell, who reiterated that the improvement in quality of life underscored meaningful activity.
Three BTK inhibitors, ibrutinib, acalabrutinib, and zanubrutinib, have been approved for the treatment of hematologic malignancies. These have also been well tolerated. The shared mechanism of action of these drugs is a reduction in B-cell activity achieved by blocking BTK enzyme signaling. The autoimmune activity of pemphigus vulgaris is at least partially mediated by B cells.
“Rilzabrutinib is the first BTK inhibitor tried in pemphigus,” said Dr. Murrell, who cited evidence that pemphigus is at least partially mediated by B-cell activity. The proof-of-concept phase 2 study has increased expectations for the phase 3 PEGASUS trial, which is scheduled for completion in about 1 year, she said.
Dr. Murrell reports financial relationship with multiple pharmaceutical companies, including Principia Biopharma, a Sanofi subsidiary that is developing rilzabrutinib and sponsored the BELIEVE trial.
In patients with newly diagnosed or relapsing pemphigus vulgaris, an update of the phase 2 BELIEVE study with the Bruton tyrosine kinase (BTK) inhibitor rilzabrutinib has raised hopes that the ongoing phase 3 trial will confirm that this drug is a breakthrough therapy, according to an investigator who presented the data at the American Academy of Dermatology Virtual Meeting Experience.
Among the highlights of the phase 2 data presented during a late-breaking research session was that a substantial minority of patients achieved a complete response within 12 weeks of starting treatment with rilzabrutinib. Treatment was associated with mostly mild and transient adverse events, according to Dedee F. Murrell, MD, director of dermatology, St. George Hospital, University of New South Wales, Sydney.
Many of the phase 2 results have been presented previously and the phase 3 trial, called PEGASUS, has now completed enrollment.
Focusing on part A of the BELIEVE study, Dr. Murrell reported that about one-third of the 27 patients enrolled had newly diagnosed pemphigus. The remaining patients had relapsing disease after a mean 8.9 years after diagnosis. The disease was judged moderate to severe in 59%. The daily oral dose of rilzabrutinib ranged from 400 mg to 600 mg twice daily.
For the primary endpoint of control of disease activity (CDA), meaning no formation of new lesions with diminishing activity of existing lesions, 52% had responded by week 4 and 70% had responded by week 12, which was the end of active treatment. Responses at both time points were comparable among patients with newly diagnosed disease (56% at week 4 and 67% at week 12) relapsing disease (50% and 72%, respectively), moderate disease severity at baseline (55% and 64%, respectively) and more severe disease (50% and 75%, respectively), Dr. Murrell noted.
“A complete response was achieved by 22% of patients at week 4 and nearly 30% by the end of the study,” she said.
These response rates were reflected in the Pemphigus Disease Area Index (PDAI) and the Autoimmune Bullous Disease Quality of Life (ABQOL) Score. From a baseline score of 20, the PDAI fell to 10 at 4 weeks and then to 6 at 12 weeks in the newly diagnosed cohort. In the relapsing cohort, the score fell from a baseline of 18 to 13 at week 4 and then to 7 at week 12.
“The improvement corresponded to a reduction in steroid doses,” Dr. Murrell reported. By the end of the study, the mean daily dose of corticosteroids fell to 10 mg from a baseline of 20 mg. In a 12-week follow-up, corticosteroid doses rose slowly and did not reach baseline levels until about eight weeks after rilzabrutinib was discontinued.
The ABQOL scores fell most rapidly in the newly diagnosed cohort. By week 12, there was about a 6.6-point reduction. In the relapsing group, the score fell by 3.7 points from a similar baseline level. Both reductions are considered highly clinically meaningful, according to Dr. Murrell. At the end of the 12 weeks of follow-up after the drug was discontinued, ABQOL scores had increased but remained below the baseline.
Nausea was reported by 15% of patients, making it the most commonly reported adverse event. All cases were grade 1 severity. Three patients had grade 2 abdominal pain. The only grade 3 event in this series was a case of cellulitis in a patient who had developed steroid-induced diabetes mellitus. With treatment, the cellulitis resolved, and the patient completed the study.
Data from part B of the BELIEVE study, which was similarly designed and enrolled 15 patients with newly diagnosed or relapsing pemphigus vulgaris, was not updated by Dr. Murrell at the meeting, but these data have been presented before and showed similar results, including achievement of CDA in the majority of patients accompanied by a reduction in corticosteroid doses.
“In summary, rilzabrutinib produced a rapid clinical effect with an overall favorable benefit-to-risk profile,” said Dr. Murrell, who reiterated that the improvement in quality of life underscored meaningful activity.
Three BTK inhibitors, ibrutinib, acalabrutinib, and zanubrutinib, have been approved for the treatment of hematologic malignancies. These have also been well tolerated. The shared mechanism of action of these drugs is a reduction in B-cell activity achieved by blocking BTK enzyme signaling. The autoimmune activity of pemphigus vulgaris is at least partially mediated by B cells.
“Rilzabrutinib is the first BTK inhibitor tried in pemphigus,” said Dr. Murrell, who cited evidence that pemphigus is at least partially mediated by B-cell activity. The proof-of-concept phase 2 study has increased expectations for the phase 3 PEGASUS trial, which is scheduled for completion in about 1 year, she said.
Dr. Murrell reports financial relationship with multiple pharmaceutical companies, including Principia Biopharma, a Sanofi subsidiary that is developing rilzabrutinib and sponsored the BELIEVE trial.
In patients with newly diagnosed or relapsing pemphigus vulgaris, an update of the phase 2 BELIEVE study with the Bruton tyrosine kinase (BTK) inhibitor rilzabrutinib has raised hopes that the ongoing phase 3 trial will confirm that this drug is a breakthrough therapy, according to an investigator who presented the data at the American Academy of Dermatology Virtual Meeting Experience.
Among the highlights of the phase 2 data presented during a late-breaking research session was that a substantial minority of patients achieved a complete response within 12 weeks of starting treatment with rilzabrutinib. Treatment was associated with mostly mild and transient adverse events, according to Dedee F. Murrell, MD, director of dermatology, St. George Hospital, University of New South Wales, Sydney.
Many of the phase 2 results have been presented previously and the phase 3 trial, called PEGASUS, has now completed enrollment.
Focusing on part A of the BELIEVE study, Dr. Murrell reported that about one-third of the 27 patients enrolled had newly diagnosed pemphigus. The remaining patients had relapsing disease after a mean 8.9 years after diagnosis. The disease was judged moderate to severe in 59%. The daily oral dose of rilzabrutinib ranged from 400 mg to 600 mg twice daily.
For the primary endpoint of control of disease activity (CDA), meaning no formation of new lesions with diminishing activity of existing lesions, 52% had responded by week 4 and 70% had responded by week 12, which was the end of active treatment. Responses at both time points were comparable among patients with newly diagnosed disease (56% at week 4 and 67% at week 12) relapsing disease (50% and 72%, respectively), moderate disease severity at baseline (55% and 64%, respectively) and more severe disease (50% and 75%, respectively), Dr. Murrell noted.
“A complete response was achieved by 22% of patients at week 4 and nearly 30% by the end of the study,” she said.
These response rates were reflected in the Pemphigus Disease Area Index (PDAI) and the Autoimmune Bullous Disease Quality of Life (ABQOL) Score. From a baseline score of 20, the PDAI fell to 10 at 4 weeks and then to 6 at 12 weeks in the newly diagnosed cohort. In the relapsing cohort, the score fell from a baseline of 18 to 13 at week 4 and then to 7 at week 12.
“The improvement corresponded to a reduction in steroid doses,” Dr. Murrell reported. By the end of the study, the mean daily dose of corticosteroids fell to 10 mg from a baseline of 20 mg. In a 12-week follow-up, corticosteroid doses rose slowly and did not reach baseline levels until about eight weeks after rilzabrutinib was discontinued.
The ABQOL scores fell most rapidly in the newly diagnosed cohort. By week 12, there was about a 6.6-point reduction. In the relapsing group, the score fell by 3.7 points from a similar baseline level. Both reductions are considered highly clinically meaningful, according to Dr. Murrell. At the end of the 12 weeks of follow-up after the drug was discontinued, ABQOL scores had increased but remained below the baseline.
Nausea was reported by 15% of patients, making it the most commonly reported adverse event. All cases were grade 1 severity. Three patients had grade 2 abdominal pain. The only grade 3 event in this series was a case of cellulitis in a patient who had developed steroid-induced diabetes mellitus. With treatment, the cellulitis resolved, and the patient completed the study.
Data from part B of the BELIEVE study, which was similarly designed and enrolled 15 patients with newly diagnosed or relapsing pemphigus vulgaris, was not updated by Dr. Murrell at the meeting, but these data have been presented before and showed similar results, including achievement of CDA in the majority of patients accompanied by a reduction in corticosteroid doses.
“In summary, rilzabrutinib produced a rapid clinical effect with an overall favorable benefit-to-risk profile,” said Dr. Murrell, who reiterated that the improvement in quality of life underscored meaningful activity.
Three BTK inhibitors, ibrutinib, acalabrutinib, and zanubrutinib, have been approved for the treatment of hematologic malignancies. These have also been well tolerated. The shared mechanism of action of these drugs is a reduction in B-cell activity achieved by blocking BTK enzyme signaling. The autoimmune activity of pemphigus vulgaris is at least partially mediated by B cells.
“Rilzabrutinib is the first BTK inhibitor tried in pemphigus,” said Dr. Murrell, who cited evidence that pemphigus is at least partially mediated by B-cell activity. The proof-of-concept phase 2 study has increased expectations for the phase 3 PEGASUS trial, which is scheduled for completion in about 1 year, she said.
Dr. Murrell reports financial relationship with multiple pharmaceutical companies, including Principia Biopharma, a Sanofi subsidiary that is developing rilzabrutinib and sponsored the BELIEVE trial.
FROM AAD VMX 2021
Moderna announces first data showing efficacy of COVID-19 vaccine booster in development
among people already vaccinated for COVID-19, according to first results released May 5.
Furthermore, data from the company’s ongoing phase 2 study show the variant-specific booster, known as mRNA-1273.351, achieved higher antibody titers against the B.1.351 variant than did a booster with the original Moderna vaccine.
“We are encouraged by these new data, which reinforce our confidence that our booster strategy should be protective against these newly detected variants. The strong and rapid boost in titers to levels above primary vaccination also clearly demonstrates the ability of mRNA-1273 to induce immune memory,” Stéphane Bancel, chief executive officer of Moderna, said in a statement.
The phase 2 study researchers also are evaluating a multivariant booster that is a 50/50 mix of mRNA-1273.351 and mRNA-1273, the initial vaccine given Food and Drug Administration emergency use authorization, in a single vial.
Unlike the two-dose regimen with the original vaccine, the boosters are administered as a single dose immunization.
The trial participants received a booster 6-8 months after primary vaccination. Titers to the wild-type SARS-CoV-2 virus remained high and detectable in 37 out of 40 participants. However, prior to the booster, titers against the two variants of concern, B.1.351 and P.1, were lower, with about half of participants showing undetectable levels.
In contrast, 2 weeks after a booster with the original vaccine or the B.1.351 strain-specific product, pseudovirus neutralizing titers were boosted in all participants and all variants tested.
“Following [the] boost, geometric mean titers against the wild-type, B.1.351, and P.1 variants increased to levels similar to or higher than the previously reported peak titers against the ancestral (D614G) strain following primary vaccination,” the company stated.
Both mRNA-1273.351 and mRNA-1273 booster doses were generally well tolerated, the company reported. Safety and tolerability were generally comparable to those reported after the second dose of the original vaccine. Most adverse events were mild to moderate, with injection site pain most common in both groups. Participants also reported fatigue, headache, myalgia, and arthralgia.
The company plans to release data shortly on the booster efficacy at additional time points beyond 2 weeks for mRNA-1273.351, a lower-dose booster with mRNA-1272/351, as well as data on the multivariant mRNA vaccine booster.
In addition to the company’s phase 2 study, the National Institute of Allergy and Infectious Diseases is conducting a separate phase 1 study of mRNA-1273.351.
A version of this article first appeared on Medscape.com.
among people already vaccinated for COVID-19, according to first results released May 5.
Furthermore, data from the company’s ongoing phase 2 study show the variant-specific booster, known as mRNA-1273.351, achieved higher antibody titers against the B.1.351 variant than did a booster with the original Moderna vaccine.
“We are encouraged by these new data, which reinforce our confidence that our booster strategy should be protective against these newly detected variants. The strong and rapid boost in titers to levels above primary vaccination also clearly demonstrates the ability of mRNA-1273 to induce immune memory,” Stéphane Bancel, chief executive officer of Moderna, said in a statement.
The phase 2 study researchers also are evaluating a multivariant booster that is a 50/50 mix of mRNA-1273.351 and mRNA-1273, the initial vaccine given Food and Drug Administration emergency use authorization, in a single vial.
Unlike the two-dose regimen with the original vaccine, the boosters are administered as a single dose immunization.
The trial participants received a booster 6-8 months after primary vaccination. Titers to the wild-type SARS-CoV-2 virus remained high and detectable in 37 out of 40 participants. However, prior to the booster, titers against the two variants of concern, B.1.351 and P.1, were lower, with about half of participants showing undetectable levels.
In contrast, 2 weeks after a booster with the original vaccine or the B.1.351 strain-specific product, pseudovirus neutralizing titers were boosted in all participants and all variants tested.
“Following [the] boost, geometric mean titers against the wild-type, B.1.351, and P.1 variants increased to levels similar to or higher than the previously reported peak titers against the ancestral (D614G) strain following primary vaccination,” the company stated.
Both mRNA-1273.351 and mRNA-1273 booster doses were generally well tolerated, the company reported. Safety and tolerability were generally comparable to those reported after the second dose of the original vaccine. Most adverse events were mild to moderate, with injection site pain most common in both groups. Participants also reported fatigue, headache, myalgia, and arthralgia.
The company plans to release data shortly on the booster efficacy at additional time points beyond 2 weeks for mRNA-1273.351, a lower-dose booster with mRNA-1272/351, as well as data on the multivariant mRNA vaccine booster.
In addition to the company’s phase 2 study, the National Institute of Allergy and Infectious Diseases is conducting a separate phase 1 study of mRNA-1273.351.
A version of this article first appeared on Medscape.com.
among people already vaccinated for COVID-19, according to first results released May 5.
Furthermore, data from the company’s ongoing phase 2 study show the variant-specific booster, known as mRNA-1273.351, achieved higher antibody titers against the B.1.351 variant than did a booster with the original Moderna vaccine.
“We are encouraged by these new data, which reinforce our confidence that our booster strategy should be protective against these newly detected variants. The strong and rapid boost in titers to levels above primary vaccination also clearly demonstrates the ability of mRNA-1273 to induce immune memory,” Stéphane Bancel, chief executive officer of Moderna, said in a statement.
The phase 2 study researchers also are evaluating a multivariant booster that is a 50/50 mix of mRNA-1273.351 and mRNA-1273, the initial vaccine given Food and Drug Administration emergency use authorization, in a single vial.
Unlike the two-dose regimen with the original vaccine, the boosters are administered as a single dose immunization.
The trial participants received a booster 6-8 months after primary vaccination. Titers to the wild-type SARS-CoV-2 virus remained high and detectable in 37 out of 40 participants. However, prior to the booster, titers against the two variants of concern, B.1.351 and P.1, were lower, with about half of participants showing undetectable levels.
In contrast, 2 weeks after a booster with the original vaccine or the B.1.351 strain-specific product, pseudovirus neutralizing titers were boosted in all participants and all variants tested.
“Following [the] boost, geometric mean titers against the wild-type, B.1.351, and P.1 variants increased to levels similar to or higher than the previously reported peak titers against the ancestral (D614G) strain following primary vaccination,” the company stated.
Both mRNA-1273.351 and mRNA-1273 booster doses were generally well tolerated, the company reported. Safety and tolerability were generally comparable to those reported after the second dose of the original vaccine. Most adverse events were mild to moderate, with injection site pain most common in both groups. Participants also reported fatigue, headache, myalgia, and arthralgia.
The company plans to release data shortly on the booster efficacy at additional time points beyond 2 weeks for mRNA-1273.351, a lower-dose booster with mRNA-1272/351, as well as data on the multivariant mRNA vaccine booster.
In addition to the company’s phase 2 study, the National Institute of Allergy and Infectious Diseases is conducting a separate phase 1 study of mRNA-1273.351.
A version of this article first appeared on Medscape.com.
Home Treatment of Presumed Melanocytic Nevus With Frankincense
To the Editor:
Melanocytic nevi are ubiquitous, and although they are benign, patients often desire to have them removed. We report a patient who presented to our clinic after attempting home removal of a concerning mole on the back with frankincense, a remedy that she found online.
A 43-year-old woman presented with a worrisome mole on the back. She had no personal history of skin cancer, but her father had a history of melanoma in situ in his 60s. The patient reported that she had the mole for years, but approximately 1 month prior to her visit she noticed that it began to bleed and crust, causing concern for melanoma. She read online that the lesion could be removed with topical application of the essential oil frankincense; she applied it directly to the lesion on the back. Within hours she developed a burn where it was applied with associated blistering.
Clinically, the lesion appeared as a darkly pigmented, well-circumscribed papule with hemorrhagic crust overlying a well-demarcated pink plaque (Figure 1). Dermatoscopically, the lesion lacked a pigment network and demonstrated 2 distinct pink papules with peripheral telangiectasia and a pink background with white streaks (Figure 2). A shave biopsy of the lesion demonstrated a nodular basal cell carcinoma extending to the base and margin.
Frankincense is the common name given to oleo-gum-resins of Boswellia species.1 It has been studied extensively for anti-inflammatory and antitumoral properties. It has been demonstrated that high concentrations of its active component, boswellic acid, can have a cytotoxic or cytostatic effect on certain malignant cell lines, such as melanoma, in vitro.2,3 It also has been shown to be antitumoral in mouse models.4 There are limited in vivo studies in the literature assessing the effects of boswellic acid or frankincense on cutaneous melanocytic lesions or other cutaneous malignancies, such as basal cell carcinoma.
A Google search of home remedy mole removal yielded more than 1,000,000 results. At the time of submission, the top 5 results all listed frankincense as a potential treatment along with garlic, iodine, castor oil, onion juice, pineapple juice, banana peels, honey, and aloe vera. None of the results cited evidence for their treatments. Although all recommended dilution of the frankincense prior to application, none warned of potential risks or side effects of its use.
Natural methods of home mole removal have long been sought after. Escharotics are most commonly utilized, including bloodroot (Sanguinaria canadensis), zinc chloride, Chelidonium majus, and Solanum sodomaeum. Many formulations are commercially available online, despite the fact that they can be mutilating and potentially dangerous when used without appropriate supervision.5 This case and an online search demonstrated that these agents are not only potentially harmful home remedies but also are currently falsely advertised as effective therapeutic management for melanocytic nevi.
Approximately 6 million individuals in the United States search the internet for health information daily, and as many as 41% of those do so to learn about alternative medicine.5,6 Although information gleaned from search engines can be useful, it is unregulated and often can be inaccurate. Clinicians generally are unaware of the erroneous material presented online and, therefore, cannot appropriately combat patient misinformation. Our case demonstrates the need to maintain an awareness of common online fallacies to better answer patient questions and guide them to more accurate sources of dermatologic information and appropriate treatment.
- Du Z, Liu Z, Ning Z, et al. Prospects of boswellic acids as potential pharmaceutics. Planta Med. 2015;81:259-271.
- Eichhorn T, Greten HJ, Efferth T. Molecular determinants of the response of tumor cells to boswellic acids. Pharmaceuticals (Basel). 2011;4:1171-1182.
- Zhao W, Entschladen F, Liu H, et al. Boswellic acid acetate induces differentiation and apoptosis in highly metastatic melanoma and fibrosarcoma cell. Cancer Detect Prev. 2003;27:67-75.
- Huang MT, Badmaev V, Ding Y, et al. Anti-tumor and anti-carcinogenic activities of triterpenoid, beta-boswellic acid. Biofactors. 2000;13:225-230.
- Adler BL, Friedman AJ. Safety & efficacy of agents used for home mole removal and skin cancer treatment in the internet age, and analysis of cases. J Drugs Dermatol. 2013;12:1058-1063.
- Kanthawala S, Vermeesch A, Given B, et al. Answers to health questions: internet search results versus online health community responses. J Med Internet Res. 2016;18:E95.
To the Editor:
Melanocytic nevi are ubiquitous, and although they are benign, patients often desire to have them removed. We report a patient who presented to our clinic after attempting home removal of a concerning mole on the back with frankincense, a remedy that she found online.
A 43-year-old woman presented with a worrisome mole on the back. She had no personal history of skin cancer, but her father had a history of melanoma in situ in his 60s. The patient reported that she had the mole for years, but approximately 1 month prior to her visit she noticed that it began to bleed and crust, causing concern for melanoma. She read online that the lesion could be removed with topical application of the essential oil frankincense; she applied it directly to the lesion on the back. Within hours she developed a burn where it was applied with associated blistering.
Clinically, the lesion appeared as a darkly pigmented, well-circumscribed papule with hemorrhagic crust overlying a well-demarcated pink plaque (Figure 1). Dermatoscopically, the lesion lacked a pigment network and demonstrated 2 distinct pink papules with peripheral telangiectasia and a pink background with white streaks (Figure 2). A shave biopsy of the lesion demonstrated a nodular basal cell carcinoma extending to the base and margin.


Frankincense is the common name given to oleo-gum-resins of Boswellia species.1 It has been studied extensively for anti-inflammatory and antitumoral properties. It has been demonstrated that high concentrations of its active component, boswellic acid, can have a cytotoxic or cytostatic effect on certain malignant cell lines, such as melanoma, in vitro.2,3 It also has been shown to be antitumoral in mouse models.4 There are limited in vivo studies in the literature assessing the effects of boswellic acid or frankincense on cutaneous melanocytic lesions or other cutaneous malignancies, such as basal cell carcinoma.
A Google search of home remedy mole removal yielded more than 1,000,000 results. At the time of submission, the top 5 results all listed frankincense as a potential treatment along with garlic, iodine, castor oil, onion juice, pineapple juice, banana peels, honey, and aloe vera. None of the results cited evidence for their treatments. Although all recommended dilution of the frankincense prior to application, none warned of potential risks or side effects of its use.
Natural methods of home mole removal have long been sought after. Escharotics are most commonly utilized, including bloodroot (Sanguinaria canadensis), zinc chloride, Chelidonium majus, and Solanum sodomaeum. Many formulations are commercially available online, despite the fact that they can be mutilating and potentially dangerous when used without appropriate supervision.5 This case and an online search demonstrated that these agents are not only potentially harmful home remedies but also are currently falsely advertised as effective therapeutic management for melanocytic nevi.
Approximately 6 million individuals in the United States search the internet for health information daily, and as many as 41% of those do so to learn about alternative medicine.5,6 Although information gleaned from search engines can be useful, it is unregulated and often can be inaccurate. Clinicians generally are unaware of the erroneous material presented online and, therefore, cannot appropriately combat patient misinformation. Our case demonstrates the need to maintain an awareness of common online fallacies to better answer patient questions and guide them to more accurate sources of dermatologic information and appropriate treatment.
To the Editor:
Melanocytic nevi are ubiquitous, and although they are benign, patients often desire to have them removed. We report a patient who presented to our clinic after attempting home removal of a concerning mole on the back with frankincense, a remedy that she found online.
A 43-year-old woman presented with a worrisome mole on the back. She had no personal history of skin cancer, but her father had a history of melanoma in situ in his 60s. The patient reported that she had the mole for years, but approximately 1 month prior to her visit she noticed that it began to bleed and crust, causing concern for melanoma. She read online that the lesion could be removed with topical application of the essential oil frankincense; she applied it directly to the lesion on the back. Within hours she developed a burn where it was applied with associated blistering.
Clinically, the lesion appeared as a darkly pigmented, well-circumscribed papule with hemorrhagic crust overlying a well-demarcated pink plaque (Figure 1). Dermatoscopically, the lesion lacked a pigment network and demonstrated 2 distinct pink papules with peripheral telangiectasia and a pink background with white streaks (Figure 2). A shave biopsy of the lesion demonstrated a nodular basal cell carcinoma extending to the base and margin.


Frankincense is the common name given to oleo-gum-resins of Boswellia species.1 It has been studied extensively for anti-inflammatory and antitumoral properties. It has been demonstrated that high concentrations of its active component, boswellic acid, can have a cytotoxic or cytostatic effect on certain malignant cell lines, such as melanoma, in vitro.2,3 It also has been shown to be antitumoral in mouse models.4 There are limited in vivo studies in the literature assessing the effects of boswellic acid or frankincense on cutaneous melanocytic lesions or other cutaneous malignancies, such as basal cell carcinoma.
A Google search of home remedy mole removal yielded more than 1,000,000 results. At the time of submission, the top 5 results all listed frankincense as a potential treatment along with garlic, iodine, castor oil, onion juice, pineapple juice, banana peels, honey, and aloe vera. None of the results cited evidence for their treatments. Although all recommended dilution of the frankincense prior to application, none warned of potential risks or side effects of its use.
Natural methods of home mole removal have long been sought after. Escharotics are most commonly utilized, including bloodroot (Sanguinaria canadensis), zinc chloride, Chelidonium majus, and Solanum sodomaeum. Many formulations are commercially available online, despite the fact that they can be mutilating and potentially dangerous when used without appropriate supervision.5 This case and an online search demonstrated that these agents are not only potentially harmful home remedies but also are currently falsely advertised as effective therapeutic management for melanocytic nevi.
Approximately 6 million individuals in the United States search the internet for health information daily, and as many as 41% of those do so to learn about alternative medicine.5,6 Although information gleaned from search engines can be useful, it is unregulated and often can be inaccurate. Clinicians generally are unaware of the erroneous material presented online and, therefore, cannot appropriately combat patient misinformation. Our case demonstrates the need to maintain an awareness of common online fallacies to better answer patient questions and guide them to more accurate sources of dermatologic information and appropriate treatment.
- Du Z, Liu Z, Ning Z, et al. Prospects of boswellic acids as potential pharmaceutics. Planta Med. 2015;81:259-271.
- Eichhorn T, Greten HJ, Efferth T. Molecular determinants of the response of tumor cells to boswellic acids. Pharmaceuticals (Basel). 2011;4:1171-1182.
- Zhao W, Entschladen F, Liu H, et al. Boswellic acid acetate induces differentiation and apoptosis in highly metastatic melanoma and fibrosarcoma cell. Cancer Detect Prev. 2003;27:67-75.
- Huang MT, Badmaev V, Ding Y, et al. Anti-tumor and anti-carcinogenic activities of triterpenoid, beta-boswellic acid. Biofactors. 2000;13:225-230.
- Adler BL, Friedman AJ. Safety & efficacy of agents used for home mole removal and skin cancer treatment in the internet age, and analysis of cases. J Drugs Dermatol. 2013;12:1058-1063.
- Kanthawala S, Vermeesch A, Given B, et al. Answers to health questions: internet search results versus online health community responses. J Med Internet Res. 2016;18:E95.
- Du Z, Liu Z, Ning Z, et al. Prospects of boswellic acids as potential pharmaceutics. Planta Med. 2015;81:259-271.
- Eichhorn T, Greten HJ, Efferth T. Molecular determinants of the response of tumor cells to boswellic acids. Pharmaceuticals (Basel). 2011;4:1171-1182.
- Zhao W, Entschladen F, Liu H, et al. Boswellic acid acetate induces differentiation and apoptosis in highly metastatic melanoma and fibrosarcoma cell. Cancer Detect Prev. 2003;27:67-75.
- Huang MT, Badmaev V, Ding Y, et al. Anti-tumor and anti-carcinogenic activities of triterpenoid, beta-boswellic acid. Biofactors. 2000;13:225-230.
- Adler BL, Friedman AJ. Safety & efficacy of agents used for home mole removal and skin cancer treatment in the internet age, and analysis of cases. J Drugs Dermatol. 2013;12:1058-1063.
- Kanthawala S, Vermeesch A, Given B, et al. Answers to health questions: internet search results versus online health community responses. J Med Internet Res. 2016;18:E95.
Practice Points
- Many patients seek natural methods of home mole removal online, including topical application of essential oils such as frankincense.
- These agents often are unregulated and can be potentially harmful when used without appropriate supervision.
- Dermatologists should be aware of common online fallacies to better answer patient questions and guide them to more accurate sources of dermatologic information and appropriate treatment.
Porous pill printing and prognostic poop
Printing meds per patient
What if there was a way to get exact doses of a medication, tailored specifically for each and every patient that needed it? Well, apparently it’s as easy as getting them out of a printer.
Researchers from the University of East Anglia in England may have found a new method to do just that.
Currently, medicine is “manufactured in ‘one-size-fits-all’ fashion,” said Dr. Sheng Qi, the research lead. But no patient is exactly the same, so why shouldn’t their medications be just as unique? Research on pharmaceutical 3D printing has been developing over the past 5 years, with the most common method requiring the drug to be put into “spaghetti-like filaments” before printing.
Dr. Qi and his team developed a process that bypasses the filaments, allowing them to 3D-print pills with varied porous structures that can regulate the rate of release of the drug into the body. This could be revolutionary for elderly patients and patients with complicated conditions – who often take many different drugs – to ensure more accurate doses that provide maximum benefits and minimal adverse effects.
Just as a custom-tailored suit perfectly fits the body for which it was made, the ability to tailor medication could have the same effect on a patient’s health. The only difference is what’s coming through the printer would be pills, not fabric.
It’s hip to be Pfizered
COVID-19 vaccination levels are rising, but we’ve heard a rumor that some people are still a bit reticent to participate. So how can physicians get more people to come in for a shot?
Make sure that they’re giving patients the right vaccine, for one thing. And by “right” vaccine, we mean, of course, the cool vaccine. Yes, the Internet has decided that the Pfizer vaccine is cooler than the others, according to the Atlantic.
There is, it seems, such a thing as “Pfizer superiority complex,” the article noted, while adding that, “on TikTok, hundreds of videos use a soundtrack of a woman explaining – slowly, voice full of disdain, like the rudest preschool teacher on Earth – ‘Only hot people get the Pfizer vaccine.’ ” A reporter from Slate was welcomed “to the ruling class” after sharing her upcoming Pfizer vaccination.
For the ultimate test of coolness, we surveyed the LOTME staff about the COVID-19 vaccines they had received. The results? Two Pfizers (coincidentally, the only two who knew what the hell TikTok is), one Moderna, one Johnson & Johnson, and one Godbold’s Vegetable Balsam (coincidentally, the same one who told us to get off his lawn).
And yes, we are checking on that last one.
Allergies stink!
A baby’s first bowel movement might mean more than just being the first of many diaper changes.
That particular bowel movement, called meconium, is a mixture of materials that have gone into a baby’s mouth late in the pregnancy, such as skin cells and amniotic fluid. Sounds lovely, right? The contents also include certain biochemicals and gut bacteria, and a lack of these can show an increased risk of allergies, eczema, and asthma.
Studies show that certain gut bacteria actually teach the immune system to accept compounds that are not harmful. Since allergies and other conditions are caused by a person’s immune system telling them harmless compounds are bad, it makes sense that lacking gut bacteria might show potential for developing such conditions.
Charisse Petersen, a researcher at the University of British Columbia in Vancouver, told NewScientist that parents could help decrease the development of allergies by not giving their children antibiotics that aren’t necessary and by letting kids play outside more.
Tom Marrs of King’s College London even noted that having a dog in the house is linked to a lower risk of allergies, so it might be time to get that puppy that the kids have been begging you for all through the pandemic.
Indiana Jones and the outhouse of parasites
Some archaeological finds are more impressive than others. Sometimes you find evidence of some long-lost civilization, sometimes you find a 200-year-old outhouse. That was the case with an outhouse buried near Dartmouth College that belonged to Mill Olcott, a wealthy businessman and politician who was a graduate of the college, and his family.
Now, that’s not particularly medically interesting, but the contents of the outhouse were very well preserved. That treasure trove included some fecal samples, and that’s where the story gets good, since they were preserved enough to be analyzed for parasites. Now, researchers know that parasites were very common in urban areas back in those days, when medicinal knowledge and sanitation were still deep in the dark ages, but whether or not people who lived in rural areas, wealthy or not, had them as well was a mystery.
Of course, 200-year-old poop is 200-year-old poop, so, in a task we wouldn’t envy anyone, the samples were rehydrated and run through several sieves to isolate the ancient goodies within. When all was said and done, both tapeworm and whipworm eggs were found, a surprise considering parasitic preference for warmer environments – not something northern New England is known for. But don’t forget, parasites can be your friend, too.
We will probably never know just which member of the Olcott household the poop belonged to, but the researchers noted that it was almost certain the entire house was infected. They added that, without proper infrastructure, even wealth was unable to protect people from disease. Hmm, we can’t think of any relevance that has in today’s world. Nope, absolutely none, since our health infrastructure is literally without flaw.
Printing meds per patient
What if there was a way to get exact doses of a medication, tailored specifically for each and every patient that needed it? Well, apparently it’s as easy as getting them out of a printer.
Researchers from the University of East Anglia in England may have found a new method to do just that.
Currently, medicine is “manufactured in ‘one-size-fits-all’ fashion,” said Dr. Sheng Qi, the research lead. But no patient is exactly the same, so why shouldn’t their medications be just as unique? Research on pharmaceutical 3D printing has been developing over the past 5 years, with the most common method requiring the drug to be put into “spaghetti-like filaments” before printing.
Dr. Qi and his team developed a process that bypasses the filaments, allowing them to 3D-print pills with varied porous structures that can regulate the rate of release of the drug into the body. This could be revolutionary for elderly patients and patients with complicated conditions – who often take many different drugs – to ensure more accurate doses that provide maximum benefits and minimal adverse effects.
Just as a custom-tailored suit perfectly fits the body for which it was made, the ability to tailor medication could have the same effect on a patient’s health. The only difference is what’s coming through the printer would be pills, not fabric.
It’s hip to be Pfizered
COVID-19 vaccination levels are rising, but we’ve heard a rumor that some people are still a bit reticent to participate. So how can physicians get more people to come in for a shot?
Make sure that they’re giving patients the right vaccine, for one thing. And by “right” vaccine, we mean, of course, the cool vaccine. Yes, the Internet has decided that the Pfizer vaccine is cooler than the others, according to the Atlantic.
There is, it seems, such a thing as “Pfizer superiority complex,” the article noted, while adding that, “on TikTok, hundreds of videos use a soundtrack of a woman explaining – slowly, voice full of disdain, like the rudest preschool teacher on Earth – ‘Only hot people get the Pfizer vaccine.’ ” A reporter from Slate was welcomed “to the ruling class” after sharing her upcoming Pfizer vaccination.
For the ultimate test of coolness, we surveyed the LOTME staff about the COVID-19 vaccines they had received. The results? Two Pfizers (coincidentally, the only two who knew what the hell TikTok is), one Moderna, one Johnson & Johnson, and one Godbold’s Vegetable Balsam (coincidentally, the same one who told us to get off his lawn).
And yes, we are checking on that last one.
Allergies stink!
A baby’s first bowel movement might mean more than just being the first of many diaper changes.
That particular bowel movement, called meconium, is a mixture of materials that have gone into a baby’s mouth late in the pregnancy, such as skin cells and amniotic fluid. Sounds lovely, right? The contents also include certain biochemicals and gut bacteria, and a lack of these can show an increased risk of allergies, eczema, and asthma.
Studies show that certain gut bacteria actually teach the immune system to accept compounds that are not harmful. Since allergies and other conditions are caused by a person’s immune system telling them harmless compounds are bad, it makes sense that lacking gut bacteria might show potential for developing such conditions.
Charisse Petersen, a researcher at the University of British Columbia in Vancouver, told NewScientist that parents could help decrease the development of allergies by not giving their children antibiotics that aren’t necessary and by letting kids play outside more.
Tom Marrs of King’s College London even noted that having a dog in the house is linked to a lower risk of allergies, so it might be time to get that puppy that the kids have been begging you for all through the pandemic.
Indiana Jones and the outhouse of parasites
Some archaeological finds are more impressive than others. Sometimes you find evidence of some long-lost civilization, sometimes you find a 200-year-old outhouse. That was the case with an outhouse buried near Dartmouth College that belonged to Mill Olcott, a wealthy businessman and politician who was a graduate of the college, and his family.
Now, that’s not particularly medically interesting, but the contents of the outhouse were very well preserved. That treasure trove included some fecal samples, and that’s where the story gets good, since they were preserved enough to be analyzed for parasites. Now, researchers know that parasites were very common in urban areas back in those days, when medicinal knowledge and sanitation were still deep in the dark ages, but whether or not people who lived in rural areas, wealthy or not, had them as well was a mystery.
Of course, 200-year-old poop is 200-year-old poop, so, in a task we wouldn’t envy anyone, the samples were rehydrated and run through several sieves to isolate the ancient goodies within. When all was said and done, both tapeworm and whipworm eggs were found, a surprise considering parasitic preference for warmer environments – not something northern New England is known for. But don’t forget, parasites can be your friend, too.
We will probably never know just which member of the Olcott household the poop belonged to, but the researchers noted that it was almost certain the entire house was infected. They added that, without proper infrastructure, even wealth was unable to protect people from disease. Hmm, we can’t think of any relevance that has in today’s world. Nope, absolutely none, since our health infrastructure is literally without flaw.
Printing meds per patient
What if there was a way to get exact doses of a medication, tailored specifically for each and every patient that needed it? Well, apparently it’s as easy as getting them out of a printer.
Researchers from the University of East Anglia in England may have found a new method to do just that.
Currently, medicine is “manufactured in ‘one-size-fits-all’ fashion,” said Dr. Sheng Qi, the research lead. But no patient is exactly the same, so why shouldn’t their medications be just as unique? Research on pharmaceutical 3D printing has been developing over the past 5 years, with the most common method requiring the drug to be put into “spaghetti-like filaments” before printing.
Dr. Qi and his team developed a process that bypasses the filaments, allowing them to 3D-print pills with varied porous structures that can regulate the rate of release of the drug into the body. This could be revolutionary for elderly patients and patients with complicated conditions – who often take many different drugs – to ensure more accurate doses that provide maximum benefits and minimal adverse effects.
Just as a custom-tailored suit perfectly fits the body for which it was made, the ability to tailor medication could have the same effect on a patient’s health. The only difference is what’s coming through the printer would be pills, not fabric.
It’s hip to be Pfizered
COVID-19 vaccination levels are rising, but we’ve heard a rumor that some people are still a bit reticent to participate. So how can physicians get more people to come in for a shot?
Make sure that they’re giving patients the right vaccine, for one thing. And by “right” vaccine, we mean, of course, the cool vaccine. Yes, the Internet has decided that the Pfizer vaccine is cooler than the others, according to the Atlantic.
There is, it seems, such a thing as “Pfizer superiority complex,” the article noted, while adding that, “on TikTok, hundreds of videos use a soundtrack of a woman explaining – slowly, voice full of disdain, like the rudest preschool teacher on Earth – ‘Only hot people get the Pfizer vaccine.’ ” A reporter from Slate was welcomed “to the ruling class” after sharing her upcoming Pfizer vaccination.
For the ultimate test of coolness, we surveyed the LOTME staff about the COVID-19 vaccines they had received. The results? Two Pfizers (coincidentally, the only two who knew what the hell TikTok is), one Moderna, one Johnson & Johnson, and one Godbold’s Vegetable Balsam (coincidentally, the same one who told us to get off his lawn).
And yes, we are checking on that last one.
Allergies stink!
A baby’s first bowel movement might mean more than just being the first of many diaper changes.
That particular bowel movement, called meconium, is a mixture of materials that have gone into a baby’s mouth late in the pregnancy, such as skin cells and amniotic fluid. Sounds lovely, right? The contents also include certain biochemicals and gut bacteria, and a lack of these can show an increased risk of allergies, eczema, and asthma.
Studies show that certain gut bacteria actually teach the immune system to accept compounds that are not harmful. Since allergies and other conditions are caused by a person’s immune system telling them harmless compounds are bad, it makes sense that lacking gut bacteria might show potential for developing such conditions.
Charisse Petersen, a researcher at the University of British Columbia in Vancouver, told NewScientist that parents could help decrease the development of allergies by not giving their children antibiotics that aren’t necessary and by letting kids play outside more.
Tom Marrs of King’s College London even noted that having a dog in the house is linked to a lower risk of allergies, so it might be time to get that puppy that the kids have been begging you for all through the pandemic.
Indiana Jones and the outhouse of parasites
Some archaeological finds are more impressive than others. Sometimes you find evidence of some long-lost civilization, sometimes you find a 200-year-old outhouse. That was the case with an outhouse buried near Dartmouth College that belonged to Mill Olcott, a wealthy businessman and politician who was a graduate of the college, and his family.
Now, that’s not particularly medically interesting, but the contents of the outhouse were very well preserved. That treasure trove included some fecal samples, and that’s where the story gets good, since they were preserved enough to be analyzed for parasites. Now, researchers know that parasites were very common in urban areas back in those days, when medicinal knowledge and sanitation were still deep in the dark ages, but whether or not people who lived in rural areas, wealthy or not, had them as well was a mystery.
Of course, 200-year-old poop is 200-year-old poop, so, in a task we wouldn’t envy anyone, the samples were rehydrated and run through several sieves to isolate the ancient goodies within. When all was said and done, both tapeworm and whipworm eggs were found, a surprise considering parasitic preference for warmer environments – not something northern New England is known for. But don’t forget, parasites can be your friend, too.
We will probably never know just which member of the Olcott household the poop belonged to, but the researchers noted that it was almost certain the entire house was infected. They added that, without proper infrastructure, even wealth was unable to protect people from disease. Hmm, we can’t think of any relevance that has in today’s world. Nope, absolutely none, since our health infrastructure is literally without flaw.