User login
The Journal of Family Practice is a peer-reviewed and indexed journal that provides its 95,000 family physician readers with timely, practical, and evidence-based information that they can immediately put into practice. Research and applied evidence articles, plus patient-oriented departments like Practice Alert, PURLs, and Clinical Inquiries can be found in print and at jfponline.com. The Web site, which logs an average of 125,000 visitors every month, also offers audiocasts by physician specialists and interactive features like Instant Polls and Photo Rounds Friday—a weekly diagnostic puzzle.
gambling
compulsive behaviors
ammunition
assault rifle
black jack
Boko Haram
bondage
child abuse
cocaine
Daech
drug paraphernalia
explosion
gun
human trafficking
ISIL
ISIS
Islamic caliphate
Islamic state
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
pedophile
pedophilia
poker
porn
pornography
psychedelic drug
recreational drug
sex slave rings
slot machine
terrorism
terrorist
Texas hold 'em
UFC
substance abuse
abuseed
abuseer
abusees
abuseing
abusely
abuses
aeolus
aeolused
aeoluser
aeoluses
aeolusing
aeolusly
aeoluss
ahole
aholeed
aholeer
aholees
aholeing
aholely
aholes
alcohol
alcoholed
alcoholer
alcoholes
alcoholing
alcoholly
alcohols
allman
allmaned
allmaner
allmanes
allmaning
allmanly
allmans
alted
altes
alting
altly
alts
analed
analer
anales
analing
anally
analprobe
analprobeed
analprobeer
analprobees
analprobeing
analprobely
analprobes
anals
anilingus
anilingused
anilinguser
anilinguses
anilingusing
anilingusly
anilinguss
anus
anused
anuser
anuses
anusing
anusly
anuss
areola
areolaed
areolaer
areolaes
areolaing
areolaly
areolas
areole
areoleed
areoleer
areolees
areoleing
areolely
areoles
arian
arianed
arianer
arianes
arianing
arianly
arians
aryan
aryaned
aryaner
aryanes
aryaning
aryanly
aryans
asiaed
asiaer
asiaes
asiaing
asialy
asias
ass
ass hole
ass lick
ass licked
ass licker
ass lickes
ass licking
ass lickly
ass licks
assbang
assbanged
assbangeded
assbangeder
assbangedes
assbangeding
assbangedly
assbangeds
assbanger
assbanges
assbanging
assbangly
assbangs
assbangsed
assbangser
assbangses
assbangsing
assbangsly
assbangss
assed
asser
asses
assesed
asseser
asseses
assesing
assesly
assess
assfuck
assfucked
assfucker
assfuckered
assfuckerer
assfuckeres
assfuckering
assfuckerly
assfuckers
assfuckes
assfucking
assfuckly
assfucks
asshat
asshated
asshater
asshates
asshating
asshatly
asshats
assholeed
assholeer
assholees
assholeing
assholely
assholes
assholesed
assholeser
assholeses
assholesing
assholesly
assholess
assing
assly
assmaster
assmastered
assmasterer
assmasteres
assmastering
assmasterly
assmasters
assmunch
assmunched
assmuncher
assmunches
assmunching
assmunchly
assmunchs
asss
asswipe
asswipeed
asswipeer
asswipees
asswipeing
asswipely
asswipes
asswipesed
asswipeser
asswipeses
asswipesing
asswipesly
asswipess
azz
azzed
azzer
azzes
azzing
azzly
azzs
babeed
babeer
babees
babeing
babely
babes
babesed
babeser
babeses
babesing
babesly
babess
ballsac
ballsaced
ballsacer
ballsaces
ballsacing
ballsack
ballsacked
ballsacker
ballsackes
ballsacking
ballsackly
ballsacks
ballsacly
ballsacs
ballsed
ballser
ballses
ballsing
ballsly
ballss
barf
barfed
barfer
barfes
barfing
barfly
barfs
bastard
bastarded
bastarder
bastardes
bastarding
bastardly
bastards
bastardsed
bastardser
bastardses
bastardsing
bastardsly
bastardss
bawdy
bawdyed
bawdyer
bawdyes
bawdying
bawdyly
bawdys
beaner
beanered
beanerer
beaneres
beanering
beanerly
beaners
beardedclam
beardedclamed
beardedclamer
beardedclames
beardedclaming
beardedclamly
beardedclams
beastiality
beastialityed
beastialityer
beastialityes
beastialitying
beastialityly
beastialitys
beatch
beatched
beatcher
beatches
beatching
beatchly
beatchs
beater
beatered
beaterer
beateres
beatering
beaterly
beaters
beered
beerer
beeres
beering
beerly
beeyotch
beeyotched
beeyotcher
beeyotches
beeyotching
beeyotchly
beeyotchs
beotch
beotched
beotcher
beotches
beotching
beotchly
beotchs
biatch
biatched
biatcher
biatches
biatching
biatchly
biatchs
big tits
big titsed
big titser
big titses
big titsing
big titsly
big titss
bigtits
bigtitsed
bigtitser
bigtitses
bigtitsing
bigtitsly
bigtitss
bimbo
bimboed
bimboer
bimboes
bimboing
bimboly
bimbos
bisexualed
bisexualer
bisexuales
bisexualing
bisexually
bisexuals
bitch
bitched
bitcheded
bitcheder
bitchedes
bitcheding
bitchedly
bitcheds
bitcher
bitches
bitchesed
bitcheser
bitcheses
bitchesing
bitchesly
bitchess
bitching
bitchly
bitchs
bitchy
bitchyed
bitchyer
bitchyes
bitchying
bitchyly
bitchys
bleached
bleacher
bleaches
bleaching
bleachly
bleachs
blow job
blow jobed
blow jober
blow jobes
blow jobing
blow jobly
blow jobs
blowed
blower
blowes
blowing
blowjob
blowjobed
blowjober
blowjobes
blowjobing
blowjobly
blowjobs
blowjobsed
blowjobser
blowjobses
blowjobsing
blowjobsly
blowjobss
blowly
blows
boink
boinked
boinker
boinkes
boinking
boinkly
boinks
bollock
bollocked
bollocker
bollockes
bollocking
bollockly
bollocks
bollocksed
bollockser
bollockses
bollocksing
bollocksly
bollockss
bollok
bolloked
bolloker
bollokes
bolloking
bollokly
bolloks
boner
bonered
bonerer
boneres
bonering
bonerly
boners
bonersed
bonerser
bonerses
bonersing
bonersly
bonerss
bong
bonged
bonger
bonges
bonging
bongly
bongs
boob
boobed
boober
boobes
boobies
boobiesed
boobieser
boobieses
boobiesing
boobiesly
boobiess
boobing
boobly
boobs
boobsed
boobser
boobses
boobsing
boobsly
boobss
booby
boobyed
boobyer
boobyes
boobying
boobyly
boobys
booger
boogered
boogerer
boogeres
boogering
boogerly
boogers
bookie
bookieed
bookieer
bookiees
bookieing
bookiely
bookies
bootee
booteeed
booteeer
booteees
booteeing
booteely
bootees
bootie
bootieed
bootieer
bootiees
bootieing
bootiely
booties
booty
bootyed
bootyer
bootyes
bootying
bootyly
bootys
boozeed
boozeer
boozees
boozeing
boozely
boozer
boozered
boozerer
boozeres
boozering
boozerly
boozers
boozes
boozy
boozyed
boozyer
boozyes
boozying
boozyly
boozys
bosomed
bosomer
bosomes
bosoming
bosomly
bosoms
bosomy
bosomyed
bosomyer
bosomyes
bosomying
bosomyly
bosomys
bugger
buggered
buggerer
buggeres
buggering
buggerly
buggers
bukkake
bukkakeed
bukkakeer
bukkakees
bukkakeing
bukkakely
bukkakes
bull shit
bull shited
bull shiter
bull shites
bull shiting
bull shitly
bull shits
bullshit
bullshited
bullshiter
bullshites
bullshiting
bullshitly
bullshits
bullshitsed
bullshitser
bullshitses
bullshitsing
bullshitsly
bullshitss
bullshitted
bullshitteded
bullshitteder
bullshittedes
bullshitteding
bullshittedly
bullshitteds
bullturds
bullturdsed
bullturdser
bullturdses
bullturdsing
bullturdsly
bullturdss
bung
bunged
bunger
bunges
bunging
bungly
bungs
busty
bustyed
bustyer
bustyes
bustying
bustyly
bustys
butt
butt fuck
butt fucked
butt fucker
butt fuckes
butt fucking
butt fuckly
butt fucks
butted
buttes
buttfuck
buttfucked
buttfucker
buttfuckered
buttfuckerer
buttfuckeres
buttfuckering
buttfuckerly
buttfuckers
buttfuckes
buttfucking
buttfuckly
buttfucks
butting
buttly
buttplug
buttpluged
buttpluger
buttpluges
buttpluging
buttplugly
buttplugs
butts
caca
cacaed
cacaer
cacaes
cacaing
cacaly
cacas
cahone
cahoneed
cahoneer
cahonees
cahoneing
cahonely
cahones
cameltoe
cameltoeed
cameltoeer
cameltoees
cameltoeing
cameltoely
cameltoes
carpetmuncher
carpetmunchered
carpetmuncherer
carpetmuncheres
carpetmunchering
carpetmuncherly
carpetmunchers
cawk
cawked
cawker
cawkes
cawking
cawkly
cawks
chinc
chinced
chincer
chinces
chincing
chincly
chincs
chincsed
chincser
chincses
chincsing
chincsly
chincss
chink
chinked
chinker
chinkes
chinking
chinkly
chinks
chode
chodeed
chodeer
chodees
chodeing
chodely
chodes
chodesed
chodeser
chodeses
chodesing
chodesly
chodess
clit
clited
cliter
clites
cliting
clitly
clitoris
clitorised
clitoriser
clitorises
clitorising
clitorisly
clitoriss
clitorus
clitorused
clitoruser
clitoruses
clitorusing
clitorusly
clitoruss
clits
clitsed
clitser
clitses
clitsing
clitsly
clitss
clitty
clittyed
clittyer
clittyes
clittying
clittyly
clittys
cocain
cocaine
cocained
cocaineed
cocaineer
cocainees
cocaineing
cocainely
cocainer
cocaines
cocaining
cocainly
cocains
cock
cock sucker
cock suckered
cock suckerer
cock suckeres
cock suckering
cock suckerly
cock suckers
cockblock
cockblocked
cockblocker
cockblockes
cockblocking
cockblockly
cockblocks
cocked
cocker
cockes
cockholster
cockholstered
cockholsterer
cockholsteres
cockholstering
cockholsterly
cockholsters
cocking
cockknocker
cockknockered
cockknockerer
cockknockeres
cockknockering
cockknockerly
cockknockers
cockly
cocks
cocksed
cockser
cockses
cocksing
cocksly
cocksmoker
cocksmokered
cocksmokerer
cocksmokeres
cocksmokering
cocksmokerly
cocksmokers
cockss
cocksucker
cocksuckered
cocksuckerer
cocksuckeres
cocksuckering
cocksuckerly
cocksuckers
coital
coitaled
coitaler
coitales
coitaling
coitally
coitals
commie
commieed
commieer
commiees
commieing
commiely
commies
condomed
condomer
condomes
condoming
condomly
condoms
coon
cooned
cooner
coones
cooning
coonly
coons
coonsed
coonser
coonses
coonsing
coonsly
coonss
corksucker
corksuckered
corksuckerer
corksuckeres
corksuckering
corksuckerly
corksuckers
cracked
crackwhore
crackwhoreed
crackwhoreer
crackwhorees
crackwhoreing
crackwhorely
crackwhores
crap
craped
craper
crapes
craping
craply
crappy
crappyed
crappyer
crappyes
crappying
crappyly
crappys
cum
cumed
cumer
cumes
cuming
cumly
cummin
cummined
cumminer
cummines
cumming
cumminged
cumminger
cumminges
cumminging
cummingly
cummings
cummining
cumminly
cummins
cums
cumshot
cumshoted
cumshoter
cumshotes
cumshoting
cumshotly
cumshots
cumshotsed
cumshotser
cumshotses
cumshotsing
cumshotsly
cumshotss
cumslut
cumsluted
cumsluter
cumslutes
cumsluting
cumslutly
cumsluts
cumstain
cumstained
cumstainer
cumstaines
cumstaining
cumstainly
cumstains
cunilingus
cunilingused
cunilinguser
cunilinguses
cunilingusing
cunilingusly
cunilinguss
cunnilingus
cunnilingused
cunnilinguser
cunnilinguses
cunnilingusing
cunnilingusly
cunnilinguss
cunny
cunnyed
cunnyer
cunnyes
cunnying
cunnyly
cunnys
cunt
cunted
cunter
cuntes
cuntface
cuntfaceed
cuntfaceer
cuntfacees
cuntfaceing
cuntfacely
cuntfaces
cunthunter
cunthuntered
cunthunterer
cunthunteres
cunthuntering
cunthunterly
cunthunters
cunting
cuntlick
cuntlicked
cuntlicker
cuntlickered
cuntlickerer
cuntlickeres
cuntlickering
cuntlickerly
cuntlickers
cuntlickes
cuntlicking
cuntlickly
cuntlicks
cuntly
cunts
cuntsed
cuntser
cuntses
cuntsing
cuntsly
cuntss
dago
dagoed
dagoer
dagoes
dagoing
dagoly
dagos
dagosed
dagoser
dagoses
dagosing
dagosly
dagoss
dammit
dammited
dammiter
dammites
dammiting
dammitly
dammits
damn
damned
damneded
damneder
damnedes
damneding
damnedly
damneds
damner
damnes
damning
damnit
damnited
damniter
damnites
damniting
damnitly
damnits
damnly
damns
dick
dickbag
dickbaged
dickbager
dickbages
dickbaging
dickbagly
dickbags
dickdipper
dickdippered
dickdipperer
dickdipperes
dickdippering
dickdipperly
dickdippers
dicked
dicker
dickes
dickface
dickfaceed
dickfaceer
dickfacees
dickfaceing
dickfacely
dickfaces
dickflipper
dickflippered
dickflipperer
dickflipperes
dickflippering
dickflipperly
dickflippers
dickhead
dickheaded
dickheader
dickheades
dickheading
dickheadly
dickheads
dickheadsed
dickheadser
dickheadses
dickheadsing
dickheadsly
dickheadss
dicking
dickish
dickished
dickisher
dickishes
dickishing
dickishly
dickishs
dickly
dickripper
dickrippered
dickripperer
dickripperes
dickrippering
dickripperly
dickrippers
dicks
dicksipper
dicksippered
dicksipperer
dicksipperes
dicksippering
dicksipperly
dicksippers
dickweed
dickweeded
dickweeder
dickweedes
dickweeding
dickweedly
dickweeds
dickwhipper
dickwhippered
dickwhipperer
dickwhipperes
dickwhippering
dickwhipperly
dickwhippers
dickzipper
dickzippered
dickzipperer
dickzipperes
dickzippering
dickzipperly
dickzippers
diddle
diddleed
diddleer
diddlees
diddleing
diddlely
diddles
dike
dikeed
dikeer
dikees
dikeing
dikely
dikes
dildo
dildoed
dildoer
dildoes
dildoing
dildoly
dildos
dildosed
dildoser
dildoses
dildosing
dildosly
dildoss
diligaf
diligafed
diligafer
diligafes
diligafing
diligafly
diligafs
dillweed
dillweeded
dillweeder
dillweedes
dillweeding
dillweedly
dillweeds
dimwit
dimwited
dimwiter
dimwites
dimwiting
dimwitly
dimwits
dingle
dingleed
dingleer
dinglees
dingleing
dinglely
dingles
dipship
dipshiped
dipshiper
dipshipes
dipshiping
dipshiply
dipships
dizzyed
dizzyer
dizzyes
dizzying
dizzyly
dizzys
doggiestyleed
doggiestyleer
doggiestylees
doggiestyleing
doggiestylely
doggiestyles
doggystyleed
doggystyleer
doggystylees
doggystyleing
doggystylely
doggystyles
dong
donged
donger
donges
donging
dongly
dongs
doofus
doofused
doofuser
doofuses
doofusing
doofusly
doofuss
doosh
dooshed
doosher
dooshes
dooshing
dooshly
dooshs
dopeyed
dopeyer
dopeyes
dopeying
dopeyly
dopeys
douchebag
douchebaged
douchebager
douchebages
douchebaging
douchebagly
douchebags
douchebagsed
douchebagser
douchebagses
douchebagsing
douchebagsly
douchebagss
doucheed
doucheer
douchees
doucheing
douchely
douches
douchey
doucheyed
doucheyer
doucheyes
doucheying
doucheyly
doucheys
drunk
drunked
drunker
drunkes
drunking
drunkly
drunks
dumass
dumassed
dumasser
dumasses
dumassing
dumassly
dumasss
dumbass
dumbassed
dumbasser
dumbasses
dumbassesed
dumbasseser
dumbasseses
dumbassesing
dumbassesly
dumbassess
dumbassing
dumbassly
dumbasss
dummy
dummyed
dummyer
dummyes
dummying
dummyly
dummys
dyke
dykeed
dykeer
dykees
dykeing
dykely
dykes
dykesed
dykeser
dykeses
dykesing
dykesly
dykess
erotic
eroticed
eroticer
erotices
eroticing
eroticly
erotics
extacy
extacyed
extacyer
extacyes
extacying
extacyly
extacys
extasy
extasyed
extasyer
extasyes
extasying
extasyly
extasys
fack
facked
facker
fackes
facking
fackly
facks
fag
faged
fager
fages
fagg
fagged
faggeded
faggeder
faggedes
faggeding
faggedly
faggeds
fagger
fagges
fagging
faggit
faggited
faggiter
faggites
faggiting
faggitly
faggits
faggly
faggot
faggoted
faggoter
faggotes
faggoting
faggotly
faggots
faggs
faging
fagly
fagot
fagoted
fagoter
fagotes
fagoting
fagotly
fagots
fags
fagsed
fagser
fagses
fagsing
fagsly
fagss
faig
faiged
faiger
faiges
faiging
faigly
faigs
faigt
faigted
faigter
faigtes
faigting
faigtly
faigts
fannybandit
fannybandited
fannybanditer
fannybandites
fannybanditing
fannybanditly
fannybandits
farted
farter
fartes
farting
fartknocker
fartknockered
fartknockerer
fartknockeres
fartknockering
fartknockerly
fartknockers
fartly
farts
felch
felched
felcher
felchered
felcherer
felcheres
felchering
felcherly
felchers
felches
felching
felchinged
felchinger
felchinges
felchinging
felchingly
felchings
felchly
felchs
fellate
fellateed
fellateer
fellatees
fellateing
fellately
fellates
fellatio
fellatioed
fellatioer
fellatioes
fellatioing
fellatioly
fellatios
feltch
feltched
feltcher
feltchered
feltcherer
feltcheres
feltchering
feltcherly
feltchers
feltches
feltching
feltchly
feltchs
feom
feomed
feomer
feomes
feoming
feomly
feoms
fisted
fisteded
fisteder
fistedes
fisteding
fistedly
fisteds
fisting
fistinged
fistinger
fistinges
fistinging
fistingly
fistings
fisty
fistyed
fistyer
fistyes
fistying
fistyly
fistys
floozy
floozyed
floozyer
floozyes
floozying
floozyly
floozys
foad
foaded
foader
foades
foading
foadly
foads
fondleed
fondleer
fondlees
fondleing
fondlely
fondles
foobar
foobared
foobarer
foobares
foobaring
foobarly
foobars
freex
freexed
freexer
freexes
freexing
freexly
freexs
frigg
frigga
friggaed
friggaer
friggaes
friggaing
friggaly
friggas
frigged
frigger
frigges
frigging
friggly
friggs
fubar
fubared
fubarer
fubares
fubaring
fubarly
fubars
fuck
fuckass
fuckassed
fuckasser
fuckasses
fuckassing
fuckassly
fuckasss
fucked
fuckeded
fuckeder
fuckedes
fuckeding
fuckedly
fuckeds
fucker
fuckered
fuckerer
fuckeres
fuckering
fuckerly
fuckers
fuckes
fuckface
fuckfaceed
fuckfaceer
fuckfacees
fuckfaceing
fuckfacely
fuckfaces
fuckin
fuckined
fuckiner
fuckines
fucking
fuckinged
fuckinger
fuckinges
fuckinging
fuckingly
fuckings
fuckining
fuckinly
fuckins
fuckly
fucknugget
fucknuggeted
fucknuggeter
fucknuggetes
fucknuggeting
fucknuggetly
fucknuggets
fucknut
fucknuted
fucknuter
fucknutes
fucknuting
fucknutly
fucknuts
fuckoff
fuckoffed
fuckoffer
fuckoffes
fuckoffing
fuckoffly
fuckoffs
fucks
fucksed
fuckser
fuckses
fucksing
fucksly
fuckss
fucktard
fucktarded
fucktarder
fucktardes
fucktarding
fucktardly
fucktards
fuckup
fuckuped
fuckuper
fuckupes
fuckuping
fuckuply
fuckups
fuckwad
fuckwaded
fuckwader
fuckwades
fuckwading
fuckwadly
fuckwads
fuckwit
fuckwited
fuckwiter
fuckwites
fuckwiting
fuckwitly
fuckwits
fudgepacker
fudgepackered
fudgepackerer
fudgepackeres
fudgepackering
fudgepackerly
fudgepackers
fuk
fuked
fuker
fukes
fuking
fukly
fuks
fvck
fvcked
fvcker
fvckes
fvcking
fvckly
fvcks
fxck
fxcked
fxcker
fxckes
fxcking
fxckly
fxcks
gae
gaeed
gaeer
gaees
gaeing
gaely
gaes
gai
gaied
gaier
gaies
gaiing
gaily
gais
ganja
ganjaed
ganjaer
ganjaes
ganjaing
ganjaly
ganjas
gayed
gayer
gayes
gaying
gayly
gays
gaysed
gayser
gayses
gaysing
gaysly
gayss
gey
geyed
geyer
geyes
geying
geyly
geys
gfc
gfced
gfcer
gfces
gfcing
gfcly
gfcs
gfy
gfyed
gfyer
gfyes
gfying
gfyly
gfys
ghay
ghayed
ghayer
ghayes
ghaying
ghayly
ghays
ghey
gheyed
gheyer
gheyes
gheying
gheyly
gheys
gigolo
gigoloed
gigoloer
gigoloes
gigoloing
gigololy
gigolos
goatse
goatseed
goatseer
goatsees
goatseing
goatsely
goatses
godamn
godamned
godamner
godamnes
godamning
godamnit
godamnited
godamniter
godamnites
godamniting
godamnitly
godamnits
godamnly
godamns
goddam
goddamed
goddamer
goddames
goddaming
goddamly
goddammit
goddammited
goddammiter
goddammites
goddammiting
goddammitly
goddammits
goddamn
goddamned
goddamner
goddamnes
goddamning
goddamnly
goddamns
goddams
goldenshower
goldenshowered
goldenshowerer
goldenshoweres
goldenshowering
goldenshowerly
goldenshowers
gonad
gonaded
gonader
gonades
gonading
gonadly
gonads
gonadsed
gonadser
gonadses
gonadsing
gonadsly
gonadss
gook
gooked
gooker
gookes
gooking
gookly
gooks
gooksed
gookser
gookses
gooksing
gooksly
gookss
gringo
gringoed
gringoer
gringoes
gringoing
gringoly
gringos
gspot
gspoted
gspoter
gspotes
gspoting
gspotly
gspots
gtfo
gtfoed
gtfoer
gtfoes
gtfoing
gtfoly
gtfos
guido
guidoed
guidoer
guidoes
guidoing
guidoly
guidos
handjob
handjobed
handjober
handjobes
handjobing
handjobly
handjobs
hard on
hard oned
hard oner
hard ones
hard oning
hard only
hard ons
hardknight
hardknighted
hardknighter
hardknightes
hardknighting
hardknightly
hardknights
hebe
hebeed
hebeer
hebees
hebeing
hebely
hebes
heeb
heebed
heeber
heebes
heebing
heebly
heebs
hell
helled
heller
helles
helling
hellly
hells
hemp
hemped
hemper
hempes
hemping
hemply
hemps
heroined
heroiner
heroines
heroining
heroinly
heroins
herp
herped
herper
herpes
herpesed
herpeser
herpeses
herpesing
herpesly
herpess
herping
herply
herps
herpy
herpyed
herpyer
herpyes
herpying
herpyly
herpys
hitler
hitlered
hitlerer
hitleres
hitlering
hitlerly
hitlers
hived
hiver
hives
hiving
hivly
hivs
hobag
hobaged
hobager
hobages
hobaging
hobagly
hobags
homey
homeyed
homeyer
homeyes
homeying
homeyly
homeys
homo
homoed
homoer
homoes
homoey
homoeyed
homoeyer
homoeyes
homoeying
homoeyly
homoeys
homoing
homoly
homos
honky
honkyed
honkyer
honkyes
honkying
honkyly
honkys
hooch
hooched
hoocher
hooches
hooching
hoochly
hoochs
hookah
hookahed
hookaher
hookahes
hookahing
hookahly
hookahs
hooker
hookered
hookerer
hookeres
hookering
hookerly
hookers
hoor
hoored
hoorer
hoores
hooring
hoorly
hoors
hootch
hootched
hootcher
hootches
hootching
hootchly
hootchs
hooter
hootered
hooterer
hooteres
hootering
hooterly
hooters
hootersed
hooterser
hooterses
hootersing
hootersly
hooterss
horny
hornyed
hornyer
hornyes
hornying
hornyly
hornys
houstoned
houstoner
houstones
houstoning
houstonly
houstons
hump
humped
humpeded
humpeder
humpedes
humpeding
humpedly
humpeds
humper
humpes
humping
humpinged
humpinger
humpinges
humpinging
humpingly
humpings
humply
humps
husbanded
husbander
husbandes
husbanding
husbandly
husbands
hussy
hussyed
hussyer
hussyes
hussying
hussyly
hussys
hymened
hymener
hymenes
hymening
hymenly
hymens
inbred
inbreded
inbreder
inbredes
inbreding
inbredly
inbreds
incest
incested
incester
incestes
incesting
incestly
incests
injun
injuned
injuner
injunes
injuning
injunly
injuns
jackass
jackassed
jackasser
jackasses
jackassing
jackassly
jackasss
jackhole
jackholeed
jackholeer
jackholees
jackholeing
jackholely
jackholes
jackoff
jackoffed
jackoffer
jackoffes
jackoffing
jackoffly
jackoffs
jap
japed
japer
japes
japing
japly
japs
japsed
japser
japses
japsing
japsly
japss
jerkoff
jerkoffed
jerkoffer
jerkoffes
jerkoffing
jerkoffly
jerkoffs
jerks
jism
jismed
jismer
jismes
jisming
jismly
jisms
jiz
jized
jizer
jizes
jizing
jizly
jizm
jizmed
jizmer
jizmes
jizming
jizmly
jizms
jizs
jizz
jizzed
jizzeded
jizzeder
jizzedes
jizzeding
jizzedly
jizzeds
jizzer
jizzes
jizzing
jizzly
jizzs
junkie
junkieed
junkieer
junkiees
junkieing
junkiely
junkies
junky
junkyed
junkyer
junkyes
junkying
junkyly
junkys
kike
kikeed
kikeer
kikees
kikeing
kikely
kikes
kikesed
kikeser
kikeses
kikesing
kikesly
kikess
killed
killer
killes
killing
killly
kills
kinky
kinkyed
kinkyer
kinkyes
kinkying
kinkyly
kinkys
kkk
kkked
kkker
kkkes
kkking
kkkly
kkks
klan
klaned
klaner
klanes
klaning
klanly
klans
knobend
knobended
knobender
knobendes
knobending
knobendly
knobends
kooch
kooched
koocher
kooches
koochesed
koocheser
koocheses
koochesing
koochesly
koochess
kooching
koochly
koochs
kootch
kootched
kootcher
kootches
kootching
kootchly
kootchs
kraut
krauted
krauter
krautes
krauting
krautly
krauts
kyke
kykeed
kykeer
kykees
kykeing
kykely
kykes
lech
leched
lecher
leches
leching
lechly
lechs
leper
lepered
leperer
leperes
lepering
leperly
lepers
lesbiansed
lesbianser
lesbianses
lesbiansing
lesbiansly
lesbianss
lesbo
lesboed
lesboer
lesboes
lesboing
lesboly
lesbos
lesbosed
lesboser
lesboses
lesbosing
lesbosly
lesboss
lez
lezbianed
lezbianer
lezbianes
lezbianing
lezbianly
lezbians
lezbiansed
lezbianser
lezbianses
lezbiansing
lezbiansly
lezbianss
lezbo
lezboed
lezboer
lezboes
lezboing
lezboly
lezbos
lezbosed
lezboser
lezboses
lezbosing
lezbosly
lezboss
lezed
lezer
lezes
lezing
lezly
lezs
lezzie
lezzieed
lezzieer
lezziees
lezzieing
lezziely
lezzies
lezziesed
lezzieser
lezzieses
lezziesing
lezziesly
lezziess
lezzy
lezzyed
lezzyer
lezzyes
lezzying
lezzyly
lezzys
lmaoed
lmaoer
lmaoes
lmaoing
lmaoly
lmaos
lmfao
lmfaoed
lmfaoer
lmfaoes
lmfaoing
lmfaoly
lmfaos
loined
loiner
loines
loining
loinly
loins
loinsed
loinser
loinses
loinsing
loinsly
loinss
lubeed
lubeer
lubees
lubeing
lubely
lubes
lusty
lustyed
lustyer
lustyes
lustying
lustyly
lustys
massa
massaed
massaer
massaes
massaing
massaly
massas
masterbate
masterbateed
masterbateer
masterbatees
masterbateing
masterbately
masterbates
masterbating
masterbatinged
masterbatinger
masterbatinges
masterbatinging
masterbatingly
masterbatings
masterbation
masterbationed
masterbationer
masterbationes
masterbationing
masterbationly
masterbations
masturbate
masturbateed
masturbateer
masturbatees
masturbateing
masturbately
masturbates
masturbating
masturbatinged
masturbatinger
masturbatinges
masturbatinging
masturbatingly
masturbatings
masturbation
masturbationed
masturbationer
masturbationes
masturbationing
masturbationly
masturbations
methed
mether
methes
mething
methly
meths
militaryed
militaryer
militaryes
militarying
militaryly
militarys
mofo
mofoed
mofoer
mofoes
mofoing
mofoly
mofos
molest
molested
molester
molestes
molesting
molestly
molests
moolie
moolieed
moolieer
mooliees
moolieing
mooliely
moolies
moron
moroned
moroner
morones
moroning
moronly
morons
motherfucka
motherfuckaed
motherfuckaer
motherfuckaes
motherfuckaing
motherfuckaly
motherfuckas
motherfucker
motherfuckered
motherfuckerer
motherfuckeres
motherfuckering
motherfuckerly
motherfuckers
motherfucking
motherfuckinged
motherfuckinger
motherfuckinges
motherfuckinging
motherfuckingly
motherfuckings
mtherfucker
mtherfuckered
mtherfuckerer
mtherfuckeres
mtherfuckering
mtherfuckerly
mtherfuckers
mthrfucker
mthrfuckered
mthrfuckerer
mthrfuckeres
mthrfuckering
mthrfuckerly
mthrfuckers
mthrfucking
mthrfuckinged
mthrfuckinger
mthrfuckinges
mthrfuckinging
mthrfuckingly
mthrfuckings
muff
muffdiver
muffdivered
muffdiverer
muffdiveres
muffdivering
muffdiverly
muffdivers
muffed
muffer
muffes
muffing
muffly
muffs
murdered
murderer
murderes
murdering
murderly
murders
muthafuckaz
muthafuckazed
muthafuckazer
muthafuckazes
muthafuckazing
muthafuckazly
muthafuckazs
muthafucker
muthafuckered
muthafuckerer
muthafuckeres
muthafuckering
muthafuckerly
muthafuckers
mutherfucker
mutherfuckered
mutherfuckerer
mutherfuckeres
mutherfuckering
mutherfuckerly
mutherfuckers
mutherfucking
mutherfuckinged
mutherfuckinger
mutherfuckinges
mutherfuckinging
mutherfuckingly
mutherfuckings
muthrfucking
muthrfuckinged
muthrfuckinger
muthrfuckinges
muthrfuckinging
muthrfuckingly
muthrfuckings
nad
naded
nader
nades
nading
nadly
nads
nadsed
nadser
nadses
nadsing
nadsly
nadss
nakeded
nakeder
nakedes
nakeding
nakedly
nakeds
napalm
napalmed
napalmer
napalmes
napalming
napalmly
napalms
nappy
nappyed
nappyer
nappyes
nappying
nappyly
nappys
nazi
nazied
nazier
nazies
naziing
nazily
nazis
nazism
nazismed
nazismer
nazismes
nazisming
nazismly
nazisms
negro
negroed
negroer
negroes
negroing
negroly
negros
nigga
niggaed
niggaer
niggaes
niggah
niggahed
niggaher
niggahes
niggahing
niggahly
niggahs
niggaing
niggaly
niggas
niggased
niggaser
niggases
niggasing
niggasly
niggass
niggaz
niggazed
niggazer
niggazes
niggazing
niggazly
niggazs
nigger
niggered
niggerer
niggeres
niggering
niggerly
niggers
niggersed
niggerser
niggerses
niggersing
niggersly
niggerss
niggle
niggleed
niggleer
nigglees
niggleing
nigglely
niggles
niglet
nigleted
nigleter
nigletes
nigleting
nigletly
niglets
nimrod
nimroded
nimroder
nimrodes
nimroding
nimrodly
nimrods
ninny
ninnyed
ninnyer
ninnyes
ninnying
ninnyly
ninnys
nooky
nookyed
nookyer
nookyes
nookying
nookyly
nookys
nuccitelli
nuccitellied
nuccitellier
nuccitellies
nuccitelliing
nuccitellily
nuccitellis
nympho
nymphoed
nymphoer
nymphoes
nymphoing
nympholy
nymphos
opium
opiumed
opiumer
opiumes
opiuming
opiumly
opiums
orgies
orgiesed
orgieser
orgieses
orgiesing
orgiesly
orgiess
orgy
orgyed
orgyer
orgyes
orgying
orgyly
orgys
paddy
paddyed
paddyer
paddyes
paddying
paddyly
paddys
paki
pakied
pakier
pakies
pakiing
pakily
pakis
pantie
pantieed
pantieer
pantiees
pantieing
pantiely
panties
pantiesed
pantieser
pantieses
pantiesing
pantiesly
pantiess
panty
pantyed
pantyer
pantyes
pantying
pantyly
pantys
pastie
pastieed
pastieer
pastiees
pastieing
pastiely
pasties
pasty
pastyed
pastyer
pastyes
pastying
pastyly
pastys
pecker
peckered
peckerer
peckeres
peckering
peckerly
peckers
pedo
pedoed
pedoer
pedoes
pedoing
pedoly
pedophile
pedophileed
pedophileer
pedophilees
pedophileing
pedophilely
pedophiles
pedophilia
pedophiliac
pedophiliaced
pedophiliacer
pedophiliaces
pedophiliacing
pedophiliacly
pedophiliacs
pedophiliaed
pedophiliaer
pedophiliaes
pedophiliaing
pedophilialy
pedophilias
pedos
penial
penialed
penialer
peniales
penialing
penially
penials
penile
penileed
penileer
penilees
penileing
penilely
peniles
penis
penised
peniser
penises
penising
penisly
peniss
perversion
perversioned
perversioner
perversiones
perversioning
perversionly
perversions
peyote
peyoteed
peyoteer
peyotees
peyoteing
peyotely
peyotes
phuck
phucked
phucker
phuckes
phucking
phuckly
phucks
pillowbiter
pillowbitered
pillowbiterer
pillowbiteres
pillowbitering
pillowbiterly
pillowbiters
pimp
pimped
pimper
pimpes
pimping
pimply
pimps
pinko
pinkoed
pinkoer
pinkoes
pinkoing
pinkoly
pinkos
pissed
pisseded
pisseder
pissedes
pisseding
pissedly
pisseds
pisser
pisses
pissing
pissly
pissoff
pissoffed
pissoffer
pissoffes
pissoffing
pissoffly
pissoffs
pisss
polack
polacked
polacker
polackes
polacking
polackly
polacks
pollock
pollocked
pollocker
pollockes
pollocking
pollockly
pollocks
poon
pooned
pooner
poones
pooning
poonly
poons
poontang
poontanged
poontanger
poontanges
poontanging
poontangly
poontangs
porn
porned
porner
pornes
porning
pornly
porno
pornoed
pornoer
pornoes
pornography
pornographyed
pornographyer
pornographyes
pornographying
pornographyly
pornographys
pornoing
pornoly
pornos
porns
prick
pricked
pricker
prickes
pricking
prickly
pricks
prig
priged
priger
priges
priging
prigly
prigs
prostitute
prostituteed
prostituteer
prostitutees
prostituteing
prostitutely
prostitutes
prude
prudeed
prudeer
prudees
prudeing
prudely
prudes
punkass
punkassed
punkasser
punkasses
punkassing
punkassly
punkasss
punky
punkyed
punkyer
punkyes
punkying
punkyly
punkys
puss
pussed
pusser
pusses
pussies
pussiesed
pussieser
pussieses
pussiesing
pussiesly
pussiess
pussing
pussly
pusss
pussy
pussyed
pussyer
pussyes
pussying
pussyly
pussypounder
pussypoundered
pussypounderer
pussypounderes
pussypoundering
pussypounderly
pussypounders
pussys
puto
putoed
putoer
putoes
putoing
putoly
putos
queaf
queafed
queafer
queafes
queafing
queafly
queafs
queef
queefed
queefer
queefes
queefing
queefly
queefs
queer
queered
queerer
queeres
queering
queerly
queero
queeroed
queeroer
queeroes
queeroing
queeroly
queeros
queers
queersed
queerser
queerses
queersing
queersly
queerss
quicky
quickyed
quickyer
quickyes
quickying
quickyly
quickys
quim
quimed
quimer
quimes
quiming
quimly
quims
racy
racyed
racyer
racyes
racying
racyly
racys
rape
raped
rapeded
rapeder
rapedes
rapeding
rapedly
rapeds
rapeed
rapeer
rapees
rapeing
rapely
raper
rapered
raperer
raperes
rapering
raperly
rapers
rapes
rapist
rapisted
rapister
rapistes
rapisting
rapistly
rapists
raunch
raunched
rauncher
raunches
raunching
raunchly
raunchs
rectus
rectused
rectuser
rectuses
rectusing
rectusly
rectuss
reefer
reefered
reeferer
reeferes
reefering
reeferly
reefers
reetard
reetarded
reetarder
reetardes
reetarding
reetardly
reetards
reich
reiched
reicher
reiches
reiching
reichly
reichs
retard
retarded
retardeded
retardeder
retardedes
retardeding
retardedly
retardeds
retarder
retardes
retarding
retardly
retards
rimjob
rimjobed
rimjober
rimjobes
rimjobing
rimjobly
rimjobs
ritard
ritarded
ritarder
ritardes
ritarding
ritardly
ritards
rtard
rtarded
rtarder
rtardes
rtarding
rtardly
rtards
rum
rumed
rumer
rumes
ruming
rumly
rump
rumped
rumper
rumpes
rumping
rumply
rumprammer
rumprammered
rumprammerer
rumprammeres
rumprammering
rumprammerly
rumprammers
rumps
rums
ruski
ruskied
ruskier
ruskies
ruskiing
ruskily
ruskis
sadism
sadismed
sadismer
sadismes
sadisming
sadismly
sadisms
sadist
sadisted
sadister
sadistes
sadisting
sadistly
sadists
scag
scaged
scager
scages
scaging
scagly
scags
scantily
scantilyed
scantilyer
scantilyes
scantilying
scantilyly
scantilys
schlong
schlonged
schlonger
schlonges
schlonging
schlongly
schlongs
scrog
scroged
scroger
scroges
scroging
scrogly
scrogs
scrot
scrote
scroted
scroteed
scroteer
scrotees
scroteing
scrotely
scroter
scrotes
scroting
scrotly
scrots
scrotum
scrotumed
scrotumer
scrotumes
scrotuming
scrotumly
scrotums
scrud
scruded
scruder
scrudes
scruding
scrudly
scruds
scum
scumed
scumer
scumes
scuming
scumly
scums
seaman
seamaned
seamaner
seamanes
seamaning
seamanly
seamans
seamen
seamened
seamener
seamenes
seamening
seamenly
seamens
seduceed
seduceer
seducees
seduceing
seducely
seduces
semen
semened
semener
semenes
semening
semenly
semens
shamedame
shamedameed
shamedameer
shamedamees
shamedameing
shamedamely
shamedames
shit
shite
shiteater
shiteatered
shiteaterer
shiteateres
shiteatering
shiteaterly
shiteaters
shited
shiteed
shiteer
shitees
shiteing
shitely
shiter
shites
shitface
shitfaceed
shitfaceer
shitfacees
shitfaceing
shitfacely
shitfaces
shithead
shitheaded
shitheader
shitheades
shitheading
shitheadly
shitheads
shithole
shitholeed
shitholeer
shitholees
shitholeing
shitholely
shitholes
shithouse
shithouseed
shithouseer
shithousees
shithouseing
shithousely
shithouses
shiting
shitly
shits
shitsed
shitser
shitses
shitsing
shitsly
shitss
shitt
shitted
shitteded
shitteder
shittedes
shitteding
shittedly
shitteds
shitter
shittered
shitterer
shitteres
shittering
shitterly
shitters
shittes
shitting
shittly
shitts
shitty
shittyed
shittyer
shittyes
shittying
shittyly
shittys
shiz
shized
shizer
shizes
shizing
shizly
shizs
shooted
shooter
shootes
shooting
shootly
shoots
sissy
sissyed
sissyer
sissyes
sissying
sissyly
sissys
skag
skaged
skager
skages
skaging
skagly
skags
skank
skanked
skanker
skankes
skanking
skankly
skanks
slave
slaveed
slaveer
slavees
slaveing
slavely
slaves
sleaze
sleazeed
sleazeer
sleazees
sleazeing
sleazely
sleazes
sleazy
sleazyed
sleazyer
sleazyes
sleazying
sleazyly
sleazys
slut
slutdumper
slutdumpered
slutdumperer
slutdumperes
slutdumpering
slutdumperly
slutdumpers
sluted
sluter
slutes
sluting
slutkiss
slutkissed
slutkisser
slutkisses
slutkissing
slutkissly
slutkisss
slutly
sluts
slutsed
slutser
slutses
slutsing
slutsly
slutss
smegma
smegmaed
smegmaer
smegmaes
smegmaing
smegmaly
smegmas
smut
smuted
smuter
smutes
smuting
smutly
smuts
smutty
smuttyed
smuttyer
smuttyes
smuttying
smuttyly
smuttys
snatch
snatched
snatcher
snatches
snatching
snatchly
snatchs
sniper
snipered
sniperer
sniperes
snipering
sniperly
snipers
snort
snorted
snorter
snortes
snorting
snortly
snorts
snuff
snuffed
snuffer
snuffes
snuffing
snuffly
snuffs
sodom
sodomed
sodomer
sodomes
sodoming
sodomly
sodoms
spic
spiced
spicer
spices
spicing
spick
spicked
spicker
spickes
spicking
spickly
spicks
spicly
spics
spik
spoof
spoofed
spoofer
spoofes
spoofing
spoofly
spoofs
spooge
spoogeed
spoogeer
spoogees
spoogeing
spoogely
spooges
spunk
spunked
spunker
spunkes
spunking
spunkly
spunks
steamyed
steamyer
steamyes
steamying
steamyly
steamys
stfu
stfued
stfuer
stfues
stfuing
stfuly
stfus
stiffy
stiffyed
stiffyer
stiffyes
stiffying
stiffyly
stiffys
stoneded
stoneder
stonedes
stoneding
stonedly
stoneds
stupided
stupider
stupides
stupiding
stupidly
stupids
suckeded
suckeder
suckedes
suckeding
suckedly
suckeds
sucker
suckes
sucking
suckinged
suckinger
suckinges
suckinging
suckingly
suckings
suckly
sucks
sumofabiatch
sumofabiatched
sumofabiatcher
sumofabiatches
sumofabiatching
sumofabiatchly
sumofabiatchs
tard
tarded
tarder
tardes
tarding
tardly
tards
tawdry
tawdryed
tawdryer
tawdryes
tawdrying
tawdryly
tawdrys
teabagging
teabagginged
teabagginger
teabagginges
teabagginging
teabaggingly
teabaggings
terd
terded
terder
terdes
terding
terdly
terds
teste
testee
testeed
testeeed
testeeer
testeees
testeeing
testeely
testeer
testees
testeing
testely
testes
testesed
testeser
testeses
testesing
testesly
testess
testicle
testicleed
testicleer
testiclees
testicleing
testiclely
testicles
testis
testised
testiser
testises
testising
testisly
testiss
thrusted
thruster
thrustes
thrusting
thrustly
thrusts
thug
thuged
thuger
thuges
thuging
thugly
thugs
tinkle
tinkleed
tinkleer
tinklees
tinkleing
tinklely
tinkles
tit
tited
titer
tites
titfuck
titfucked
titfucker
titfuckes
titfucking
titfuckly
titfucks
titi
titied
titier
tities
titiing
titily
titing
titis
titly
tits
titsed
titser
titses
titsing
titsly
titss
tittiefucker
tittiefuckered
tittiefuckerer
tittiefuckeres
tittiefuckering
tittiefuckerly
tittiefuckers
titties
tittiesed
tittieser
tittieses
tittiesing
tittiesly
tittiess
titty
tittyed
tittyer
tittyes
tittyfuck
tittyfucked
tittyfucker
tittyfuckered
tittyfuckerer
tittyfuckeres
tittyfuckering
tittyfuckerly
tittyfuckers
tittyfuckes
tittyfucking
tittyfuckly
tittyfucks
tittying
tittyly
tittys
toke
tokeed
tokeer
tokees
tokeing
tokely
tokes
toots
tootsed
tootser
tootses
tootsing
tootsly
tootss
tramp
tramped
tramper
trampes
tramping
tramply
tramps
transsexualed
transsexualer
transsexuales
transsexualing
transsexually
transsexuals
trashy
trashyed
trashyer
trashyes
trashying
trashyly
trashys
tubgirl
tubgirled
tubgirler
tubgirles
tubgirling
tubgirlly
tubgirls
turd
turded
turder
turdes
turding
turdly
turds
tush
tushed
tusher
tushes
tushing
tushly
tushs
twat
twated
twater
twates
twating
twatly
twats
twatsed
twatser
twatses
twatsing
twatsly
twatss
undies
undiesed
undieser
undieses
undiesing
undiesly
undiess
unweded
unweder
unwedes
unweding
unwedly
unweds
uzi
uzied
uzier
uzies
uziing
uzily
uzis
vag
vaged
vager
vages
vaging
vagly
vags
valium
valiumed
valiumer
valiumes
valiuming
valiumly
valiums
venous
virgined
virginer
virgines
virgining
virginly
virgins
vixen
vixened
vixener
vixenes
vixening
vixenly
vixens
vodkaed
vodkaer
vodkaes
vodkaing
vodkaly
vodkas
voyeur
voyeured
voyeurer
voyeures
voyeuring
voyeurly
voyeurs
vulgar
vulgared
vulgarer
vulgares
vulgaring
vulgarly
vulgars
wang
wanged
wanger
wanges
wanging
wangly
wangs
wank
wanked
wanker
wankered
wankerer
wankeres
wankering
wankerly
wankers
wankes
wanking
wankly
wanks
wazoo
wazooed
wazooer
wazooes
wazooing
wazooly
wazoos
wedgie
wedgieed
wedgieer
wedgiees
wedgieing
wedgiely
wedgies
weeded
weeder
weedes
weeding
weedly
weeds
weenie
weenieed
weenieer
weeniees
weenieing
weeniely
weenies
weewee
weeweeed
weeweeer
weeweees
weeweeing
weeweely
weewees
weiner
weinered
weinerer
weineres
weinering
weinerly
weiners
weirdo
weirdoed
weirdoer
weirdoes
weirdoing
weirdoly
weirdos
wench
wenched
wencher
wenches
wenching
wenchly
wenchs
wetback
wetbacked
wetbacker
wetbackes
wetbacking
wetbackly
wetbacks
whitey
whiteyed
whiteyer
whiteyes
whiteying
whiteyly
whiteys
whiz
whized
whizer
whizes
whizing
whizly
whizs
whoralicious
whoralicioused
whoraliciouser
whoraliciouses
whoraliciousing
whoraliciously
whoraliciouss
whore
whorealicious
whorealicioused
whorealiciouser
whorealiciouses
whorealiciousing
whorealiciously
whorealiciouss
whored
whoreded
whoreder
whoredes
whoreding
whoredly
whoreds
whoreed
whoreer
whorees
whoreface
whorefaceed
whorefaceer
whorefacees
whorefaceing
whorefacely
whorefaces
whorehopper
whorehoppered
whorehopperer
whorehopperes
whorehoppering
whorehopperly
whorehoppers
whorehouse
whorehouseed
whorehouseer
whorehousees
whorehouseing
whorehousely
whorehouses
whoreing
whorely
whores
whoresed
whoreser
whoreses
whoresing
whoresly
whoress
whoring
whoringed
whoringer
whoringes
whoringing
whoringly
whorings
wigger
wiggered
wiggerer
wiggeres
wiggering
wiggerly
wiggers
woody
woodyed
woodyer
woodyes
woodying
woodyly
woodys
wop
woped
woper
wopes
woping
woply
wops
wtf
wtfed
wtfer
wtfes
wtfing
wtfly
wtfs
xxx
xxxed
xxxer
xxxes
xxxing
xxxly
xxxs
yeasty
yeastyed
yeastyer
yeastyes
yeastying
yeastyly
yeastys
yobbo
yobboed
yobboer
yobboes
yobboing
yobboly
yobbos
zoophile
zoophileed
zoophileer
zoophilees
zoophileing
zoophilely
zoophiles
anal
ass
ass lick
balls
ballsac
bisexual
bleach
causas
cheap
cost of miracles
cunt
display network stats
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gfc
humira AND expensive
illegal
madvocate
masturbation
nuccitelli
overdose
porn
shit
snort
texarkana
abbvie
AbbVie
acid
addicted
addiction
adolescent
adult sites
Advocacy
advocacy
agitated states
AJO, postsurgical analgesic, knee, replacement, surgery
alcohol
amphetamine
androgen
antibody
apple cider vinegar
assistance
Assistance
association
at home
attorney
audit
ayurvedic
baby
ban
baricitinib
bed bugs
best
bible
bisexual
black
bleach
blog
bulimia nervosa
buy
cannabis
certificate
certification
certified
cervical cancer, concurrent chemoradiotherapy, intravoxel incoherent motion magnetic resonance imaging, MRI, IVIM, diffusion-weighted MRI, DWI
charlie sheen
cheap
cheapest
child
childhood
childlike
children
chronic fatigue syndrome
Cladribine Tablets
cocaine
cock
combination therapies, synergistic antitumor efficacy, pertuzumab, trastuzumab, ipilimumab, nivolumab, palbociclib, letrozole, lapatinib, docetaxel, trametinib, dabrafenib, carflzomib, lenalidomide
contagious
Cortical Lesions
cream
creams
crime
criminal
cure
dangerous
dangers
dasabuvir
Dasabuvir
dead
deadly
death
dementia
dependence
dependent
depression
dermatillomania
die
diet
Disability
Discount
discount
dog
drink
drug abuse
drug-induced
dying
eastern medicine
eat
ect
eczema
electroconvulsive therapy
electromagnetic therapy
electrotherapy
epa
epilepsy
erectile dysfunction
explosive disorder
fake
Fake-ovir
fatal
fatalities
fatality
fibromyalgia
financial
Financial
fish oil
food
foods
foundation
free
Gabriel Pardo
gaston
general hospital
genetic
geriatric
Giancarlo Comi
gilead
Gilead
glaucoma
Glenn S. Williams
Glenn Williams
Gloria Dalla Costa
gonorrhea
Greedy
greedy
guns
hallucinations
harvoni
Harvoni
herbal
herbs
heroin
herpes
Hidradenitis Suppurativa,
holistic
home
home remedies
home remedy
homeopathic
homeopathy
hydrocortisone
ice
image
images
job
kid
kids
kill
killer
laser
lawsuit
lawyer
ledipasvir
Ledipasvir
lesbian
lesions
lights
liver
lupus
marijuana
melancholic
memory loss
menopausal
mental retardation
military
milk
moisturizers
monoamine oxidase inhibitor drugs
MRI
MS
murder
national
natural
natural cure
natural cures
natural medications
natural medicine
natural medicines
natural remedies
natural remedy
natural treatment
natural treatments
naturally
Needy
needy
Neurology Reviews
neuropathic
nightclub massacre
nightclub shooting
nude
nudity
nutraceuticals
OASIS
oasis
off label
ombitasvir
Ombitasvir
ombitasvir/paritaprevir/ritonavir with dasabuvir
orlando shooting
overactive thyroid gland
overdose
overdosed
Paolo Preziosa
paritaprevir
Paritaprevir
pediatric
pedophile
photo
photos
picture
post partum
postnatal
pregnancy
pregnant
prenatal
prepartum
prison
program
Program
Protest
protest
psychedelics
pulse nightclub
puppy
purchase
purchasing
rape
recall
recreational drug
Rehabilitation
Retinal Measurements
retrograde ejaculation
risperdal
ritonavir
Ritonavir
ritonavir with dasabuvir
robin williams
sales
sasquatch
schizophrenia
seizure
seizures
sex
sexual
sexy
shock treatment
silver
sleep disorders
smoking
sociopath
sofosbuvir
Sofosbuvir
sovaldi
ssri
store
sue
suicidal
suicide
supplements
support
Support
Support Path
teen
teenage
teenagers
Telerehabilitation
testosterone
Th17
Th17:FoxP3+Treg cell ratio
Th22
toxic
toxin
tragedy
treatment resistant
V Pak
vagina
velpatasvir
Viekira Pa
Viekira Pak
viekira pak
violence
virgin
vitamin
VPak
weight loss
withdrawal
wrinkles
xxx
young adult
young adults
zoloft
financial
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
direct\-acting antivirals
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-jfp')]
div[contains(@class, 'pane-pub-home-jfp')]
div[contains(@class, 'pane-pub-topic-jfp')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Is there benefit to adding ezetimibe to a statin for the secondary prevention of CVD?
Evidence summary
Adding ezetimibe reduces nonfatal events but does not improve mortality
A 2018 Cochrane meta-analysis included 10 RCTs (N = 21,919 patients) that evaluated the efficacy and safety of ezetimibe plus a statin (dual therapy) vs a statin alone or plus placebo (monotherapy) for the secondary prevention of CVD. Mean age of patients ranged from 55 to 84 years. Almost all of the patients (> 99%) included in the analyses had existing ASCVD. The dose of ezetimibe was 10 mg; statins used included atorvastatin 10 to 80 mg, pitavastatin 2 to 4 mg, rosuvastatin 10 mg, and simvastatin 20 to 80 mg.1
The primary outcomes were MACE and all-cause mortality. MACE is defined as a composite of CVD, nonfatal myocardial infarction (MI), nonfatal stroke, hospitalization for unstable angina, or coronary revascularization procedures. The TABLE1 provides a detailed breakdown of each of the outcomes.
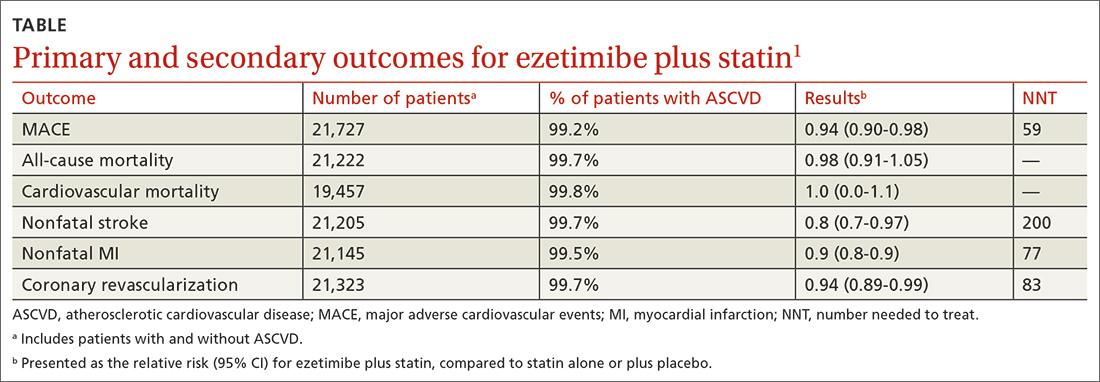
The dual-therapy group compared to the monotherapy group had a lower risk for MACE (26.6% vs 28.3%; 1.7% absolute risk reduction; 6% relative risk reduction; NNT = 59) and little or no difference in the reduction of all-cause mortality. For secondary outcomes, the dual-therapy group had a lower risk for nonfatal MI, nonfatal stroke, and coronary revascularization. There was no difference in cardiovascular mortality or adverse events between the 2 groups. The quality of evidence was high for all-cause mortality and moderate for cardiovascular mortality, MACE, MI, and stroke.1
The 2015 IMPROVE-IT study, the largest included in the Cochrane review, was a double-blind RCT (N = 18,144) conducted at 1147 sites in 39 countries comparing simvastatin 40 mg/d plus ezetimibe 10 mg/d (dual therapy) vs simvastatin 40 mg/d plus placebo (monotherapy). Patients were at least 50 years old (average age, 64 years) and had been hospitalized for acute coronary syndrome (ACS) within the previous 10 days; 76% were male and 84% were White. The average low-density lipoprotein (LDL) concentration at baseline was 94 mg/dL in both groups.2
The primary endpoint was a composite of cardiovascular death, a major coronary event (nonfatal MI, unstable angina requiring hospitalization, coronary revascularization at least 30 days after randomization), or nonfatal stroke, with a median follow-up of 6 years. The simvastatin plus ezetimibe group compared to the simvastatin-only group had a lower risk for the primary end point (HR = 0.94; 95% CI, 0.89-0.99; NNT = 50), but no differences in cardiovascular or all-cause mortality. Since the study only recruited patients with recent ACS, results are only applicable to that specific population.2
The 2022 RACING study was a multicenter, open-label, randomized, noninferiority trial that evaluated the combination of ezetimibe 10 mg and a moderate-intensity statin (rosuvastatin 10 mg) compared to a high-intensity statin alone (rosuvastatin 20 mg) in adults (N = 3780) with ASCVD. Included patients were ages 19 to 80 years (mean, 64 years) and had a baseline LDL concentration of 80 mg/dL (standard deviation, 64-100 mg/dL) with known ASCVD (defined by prior MI, ACS, history of coronary or other arterial revascularization, ischemic stroke, or peripheral artery disease); 75% were male.3
The primary outcome was a composite of cardiovascular death, major cardiovascular events, or nonfatal stroke. At 3 years, an intention-to-treat analysis found no significant difference between the combination and monotherapy groups (9% vs 9.9%; absolute difference, –0.78%; 95% CI, –2.39% to 0.83%). Dose reduction or discontinuation of the study drug(s) due to intolerance was lower in the combination group than in the monotherapy group (4.8% vs 8.2%; P < 0.0001). The study may be limited by the fact that it was nonblinded and all participants were South Korean, which limits generalizability.3
Recommendations from others
A 2022 evidence-based clinical practice guideline published in BMJ recommends adding ezetimibe to a statin to decrease all-cause mortality, cardiovascular mortality, nonfatal stroke, and nonfatal MI in patients with known CVD, regardless of their LDL concentration (weak recommendation based on a systematic review and network meta-analysis).4
In 2019, the American Heart Association and the American College of Cardiology recommended ezetimibe for patients with clinical ASCVD who are on maximally tolerated statin therapy and have an LDL concentration of 70 mg/dL or higher (Class 2b recommendation [meaning it can be considered] based on a meta-analysis of moderate-quality RCTs).5
Editor’s takeaway
The data on this important and well-studied question have inched closer to firm and clear answers. First, adding ezetimibe to a lower-intensity statin when a higher-intensity statin is not tolerated is an effective treatment. Second, adding ezetimibe to a statin improves nonfatal ASCVD outcomes but not fatal ones. What has not yet been made clear, because a noninferiority trial does not answer this question, is whether the highest intensity statin plus ezetimibe is superior to that high-intensity statin alone, regardless of LDL concentration.
1. Zhan S, Tang M, Liu F, et al. Ezetimibe for the prevention of cardiovascular disease and all‐cause mortality events. Cochrane Database System Rev. 2018;11:CD012502. doi: 10.1002/14651858.CD012502.pub2
2. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397. doi: 10.1056/NEJMoa1410489 pmid:26039521
3. Kim BK, Hong SJ, Lee YJ, et al. Long-term efficacy and safety of moderate-intensity statin with ezetimibe combination therapy versus high-intensity statin monotherapy in patients with atherosclerotic cardiovascular disease (RACING): a randomised, open-label, non-inferiority trial. Lancet. 2022;400:380-390. doi: 10.1016/S0140-6736(22)00916-3
4. Hao Q, Aertgeerts B, Guyatt G, et al. PCSK9 inhibitors and ezetimibe for the reduction of cardiovascular events: a clinical practice guideline with risk-stratified recommendations. BMJ. 2022;377:e069066. doi: 10.1136/bmj-2021-069066
5. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;73:e285-e350. doi: 10.1016/j.jacc.2018.11.003
Evidence summary
Adding ezetimibe reduces nonfatal events but does not improve mortality
A 2018 Cochrane meta-analysis included 10 RCTs (N = 21,919 patients) that evaluated the efficacy and safety of ezetimibe plus a statin (dual therapy) vs a statin alone or plus placebo (monotherapy) for the secondary prevention of CVD. Mean age of patients ranged from 55 to 84 years. Almost all of the patients (> 99%) included in the analyses had existing ASCVD. The dose of ezetimibe was 10 mg; statins used included atorvastatin 10 to 80 mg, pitavastatin 2 to 4 mg, rosuvastatin 10 mg, and simvastatin 20 to 80 mg.1
The primary outcomes were MACE and all-cause mortality. MACE is defined as a composite of CVD, nonfatal myocardial infarction (MI), nonfatal stroke, hospitalization for unstable angina, or coronary revascularization procedures. The TABLE1 provides a detailed breakdown of each of the outcomes.

The dual-therapy group compared to the monotherapy group had a lower risk for MACE (26.6% vs 28.3%; 1.7% absolute risk reduction; 6% relative risk reduction; NNT = 59) and little or no difference in the reduction of all-cause mortality. For secondary outcomes, the dual-therapy group had a lower risk for nonfatal MI, nonfatal stroke, and coronary revascularization. There was no difference in cardiovascular mortality or adverse events between the 2 groups. The quality of evidence was high for all-cause mortality and moderate for cardiovascular mortality, MACE, MI, and stroke.1
The 2015 IMPROVE-IT study, the largest included in the Cochrane review, was a double-blind RCT (N = 18,144) conducted at 1147 sites in 39 countries comparing simvastatin 40 mg/d plus ezetimibe 10 mg/d (dual therapy) vs simvastatin 40 mg/d plus placebo (monotherapy). Patients were at least 50 years old (average age, 64 years) and had been hospitalized for acute coronary syndrome (ACS) within the previous 10 days; 76% were male and 84% were White. The average low-density lipoprotein (LDL) concentration at baseline was 94 mg/dL in both groups.2
The primary endpoint was a composite of cardiovascular death, a major coronary event (nonfatal MI, unstable angina requiring hospitalization, coronary revascularization at least 30 days after randomization), or nonfatal stroke, with a median follow-up of 6 years. The simvastatin plus ezetimibe group compared to the simvastatin-only group had a lower risk for the primary end point (HR = 0.94; 95% CI, 0.89-0.99; NNT = 50), but no differences in cardiovascular or all-cause mortality. Since the study only recruited patients with recent ACS, results are only applicable to that specific population.2
The 2022 RACING study was a multicenter, open-label, randomized, noninferiority trial that evaluated the combination of ezetimibe 10 mg and a moderate-intensity statin (rosuvastatin 10 mg) compared to a high-intensity statin alone (rosuvastatin 20 mg) in adults (N = 3780) with ASCVD. Included patients were ages 19 to 80 years (mean, 64 years) and had a baseline LDL concentration of 80 mg/dL (standard deviation, 64-100 mg/dL) with known ASCVD (defined by prior MI, ACS, history of coronary or other arterial revascularization, ischemic stroke, or peripheral artery disease); 75% were male.3
The primary outcome was a composite of cardiovascular death, major cardiovascular events, or nonfatal stroke. At 3 years, an intention-to-treat analysis found no significant difference between the combination and monotherapy groups (9% vs 9.9%; absolute difference, –0.78%; 95% CI, –2.39% to 0.83%). Dose reduction or discontinuation of the study drug(s) due to intolerance was lower in the combination group than in the monotherapy group (4.8% vs 8.2%; P < 0.0001). The study may be limited by the fact that it was nonblinded and all participants were South Korean, which limits generalizability.3
Recommendations from others
A 2022 evidence-based clinical practice guideline published in BMJ recommends adding ezetimibe to a statin to decrease all-cause mortality, cardiovascular mortality, nonfatal stroke, and nonfatal MI in patients with known CVD, regardless of their LDL concentration (weak recommendation based on a systematic review and network meta-analysis).4
In 2019, the American Heart Association and the American College of Cardiology recommended ezetimibe for patients with clinical ASCVD who are on maximally tolerated statin therapy and have an LDL concentration of 70 mg/dL or higher (Class 2b recommendation [meaning it can be considered] based on a meta-analysis of moderate-quality RCTs).5
Editor’s takeaway
The data on this important and well-studied question have inched closer to firm and clear answers. First, adding ezetimibe to a lower-intensity statin when a higher-intensity statin is not tolerated is an effective treatment. Second, adding ezetimibe to a statin improves nonfatal ASCVD outcomes but not fatal ones. What has not yet been made clear, because a noninferiority trial does not answer this question, is whether the highest intensity statin plus ezetimibe is superior to that high-intensity statin alone, regardless of LDL concentration.
Evidence summary
Adding ezetimibe reduces nonfatal events but does not improve mortality
A 2018 Cochrane meta-analysis included 10 RCTs (N = 21,919 patients) that evaluated the efficacy and safety of ezetimibe plus a statin (dual therapy) vs a statin alone or plus placebo (monotherapy) for the secondary prevention of CVD. Mean age of patients ranged from 55 to 84 years. Almost all of the patients (> 99%) included in the analyses had existing ASCVD. The dose of ezetimibe was 10 mg; statins used included atorvastatin 10 to 80 mg, pitavastatin 2 to 4 mg, rosuvastatin 10 mg, and simvastatin 20 to 80 mg.1
The primary outcomes were MACE and all-cause mortality. MACE is defined as a composite of CVD, nonfatal myocardial infarction (MI), nonfatal stroke, hospitalization for unstable angina, or coronary revascularization procedures. The TABLE1 provides a detailed breakdown of each of the outcomes.

The dual-therapy group compared to the monotherapy group had a lower risk for MACE (26.6% vs 28.3%; 1.7% absolute risk reduction; 6% relative risk reduction; NNT = 59) and little or no difference in the reduction of all-cause mortality. For secondary outcomes, the dual-therapy group had a lower risk for nonfatal MI, nonfatal stroke, and coronary revascularization. There was no difference in cardiovascular mortality or adverse events between the 2 groups. The quality of evidence was high for all-cause mortality and moderate for cardiovascular mortality, MACE, MI, and stroke.1
The 2015 IMPROVE-IT study, the largest included in the Cochrane review, was a double-blind RCT (N = 18,144) conducted at 1147 sites in 39 countries comparing simvastatin 40 mg/d plus ezetimibe 10 mg/d (dual therapy) vs simvastatin 40 mg/d plus placebo (monotherapy). Patients were at least 50 years old (average age, 64 years) and had been hospitalized for acute coronary syndrome (ACS) within the previous 10 days; 76% were male and 84% were White. The average low-density lipoprotein (LDL) concentration at baseline was 94 mg/dL in both groups.2
The primary endpoint was a composite of cardiovascular death, a major coronary event (nonfatal MI, unstable angina requiring hospitalization, coronary revascularization at least 30 days after randomization), or nonfatal stroke, with a median follow-up of 6 years. The simvastatin plus ezetimibe group compared to the simvastatin-only group had a lower risk for the primary end point (HR = 0.94; 95% CI, 0.89-0.99; NNT = 50), but no differences in cardiovascular or all-cause mortality. Since the study only recruited patients with recent ACS, results are only applicable to that specific population.2
The 2022 RACING study was a multicenter, open-label, randomized, noninferiority trial that evaluated the combination of ezetimibe 10 mg and a moderate-intensity statin (rosuvastatin 10 mg) compared to a high-intensity statin alone (rosuvastatin 20 mg) in adults (N = 3780) with ASCVD. Included patients were ages 19 to 80 years (mean, 64 years) and had a baseline LDL concentration of 80 mg/dL (standard deviation, 64-100 mg/dL) with known ASCVD (defined by prior MI, ACS, history of coronary or other arterial revascularization, ischemic stroke, or peripheral artery disease); 75% were male.3
The primary outcome was a composite of cardiovascular death, major cardiovascular events, or nonfatal stroke. At 3 years, an intention-to-treat analysis found no significant difference between the combination and monotherapy groups (9% vs 9.9%; absolute difference, –0.78%; 95% CI, –2.39% to 0.83%). Dose reduction or discontinuation of the study drug(s) due to intolerance was lower in the combination group than in the monotherapy group (4.8% vs 8.2%; P < 0.0001). The study may be limited by the fact that it was nonblinded and all participants were South Korean, which limits generalizability.3
Recommendations from others
A 2022 evidence-based clinical practice guideline published in BMJ recommends adding ezetimibe to a statin to decrease all-cause mortality, cardiovascular mortality, nonfatal stroke, and nonfatal MI in patients with known CVD, regardless of their LDL concentration (weak recommendation based on a systematic review and network meta-analysis).4
In 2019, the American Heart Association and the American College of Cardiology recommended ezetimibe for patients with clinical ASCVD who are on maximally tolerated statin therapy and have an LDL concentration of 70 mg/dL or higher (Class 2b recommendation [meaning it can be considered] based on a meta-analysis of moderate-quality RCTs).5
Editor’s takeaway
The data on this important and well-studied question have inched closer to firm and clear answers. First, adding ezetimibe to a lower-intensity statin when a higher-intensity statin is not tolerated is an effective treatment. Second, adding ezetimibe to a statin improves nonfatal ASCVD outcomes but not fatal ones. What has not yet been made clear, because a noninferiority trial does not answer this question, is whether the highest intensity statin plus ezetimibe is superior to that high-intensity statin alone, regardless of LDL concentration.
1. Zhan S, Tang M, Liu F, et al. Ezetimibe for the prevention of cardiovascular disease and all‐cause mortality events. Cochrane Database System Rev. 2018;11:CD012502. doi: 10.1002/14651858.CD012502.pub2
2. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397. doi: 10.1056/NEJMoa1410489 pmid:26039521
3. Kim BK, Hong SJ, Lee YJ, et al. Long-term efficacy and safety of moderate-intensity statin with ezetimibe combination therapy versus high-intensity statin monotherapy in patients with atherosclerotic cardiovascular disease (RACING): a randomised, open-label, non-inferiority trial. Lancet. 2022;400:380-390. doi: 10.1016/S0140-6736(22)00916-3
4. Hao Q, Aertgeerts B, Guyatt G, et al. PCSK9 inhibitors and ezetimibe for the reduction of cardiovascular events: a clinical practice guideline with risk-stratified recommendations. BMJ. 2022;377:e069066. doi: 10.1136/bmj-2021-069066
5. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;73:e285-e350. doi: 10.1016/j.jacc.2018.11.003
1. Zhan S, Tang M, Liu F, et al. Ezetimibe for the prevention of cardiovascular disease and all‐cause mortality events. Cochrane Database System Rev. 2018;11:CD012502. doi: 10.1002/14651858.CD012502.pub2
2. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE-IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372:2387-2397. doi: 10.1056/NEJMoa1410489 pmid:26039521
3. Kim BK, Hong SJ, Lee YJ, et al. Long-term efficacy and safety of moderate-intensity statin with ezetimibe combination therapy versus high-intensity statin monotherapy in patients with atherosclerotic cardiovascular disease (RACING): a randomised, open-label, non-inferiority trial. Lancet. 2022;400:380-390. doi: 10.1016/S0140-6736(22)00916-3
4. Hao Q, Aertgeerts B, Guyatt G, et al. PCSK9 inhibitors and ezetimibe for the reduction of cardiovascular events: a clinical practice guideline with risk-stratified recommendations. BMJ. 2022;377:e069066. doi: 10.1136/bmj-2021-069066
5. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;73:e285-e350. doi: 10.1016/j.jacc.2018.11.003
EVIDENCE-BASED REVIEW:
YES. In patients with known cardio- vascular disease (CVD), ezetimibe with a statin decreases
In adults with atherosclerotic CVD (ASCVD), the combination of ezetimibe and a moderate-intensity statin (rosuvastatin 10 mg) was noninferior at decreasing cardiovascular death, major cardiovascular events, and nonfatal stroke, but was more tolerable, compared to a high-intensity statin (rosuvastatin 20 mg) alone (SOR, B; 1 RCT).
Feeling disconnected? Focus on what you can do
This is the exciting time of year when we graduate new classes of medical students and residents. Med school graduation brings mixed emotions; the new doctors and I both know residency will bring growth and challenges. Residency graduation is a wistful passage as well. It is so rewarding to welcome the newly board-certified family physicians to family medicine, but we miss them even as we orient a new class.
Every year, a few months (or even a few years) after graduation, I hear from a former resident, sometimes several. They ask to talk and, although it can be hard for them to explain exactly the ennui and disillusionment they’re feeling, their concerns boil down to: Is this all there is?
They are not burnt out, exactly, but they were hoping for more from their careers in family medicine.1 They find their hopes and expectations are not fulfilled by seeing patients in the office 8 hours per day, 4.5 days per week. Even those who report rewarding relationships with patients express less overall enthusiasm for jobs they were excited to start just months or years earlier.
Some of the difficulties I hear the graduates report are expected growing pains. It is a transition to go from supervised practice with attending backup to a setting where you are on your own, typically with a 4-fold increase in volume compared with residency. But the monotony is real for family physicians in full-time outpatient practice.
Research suggests an expanded scope of practice—including hospital medicine, obstetrics, and procedures—is associated with physician well-being.2,3 A broad scope of practice can bring stress, but it also brings meaning, and that meaning is protective to our well-being. However, a robust scope of practice is not always supported by medical groups or hospital systems, who prefer a more compartmentalized, widgetized physician.4 It would be easier for their algorithms if family physicians picked a lane and stayed in it. Alas, the broader our scope of practice, the healthier our population, the more equitable our care,5,6 and the happier our physicians.
The disconnect and hopelessness experienced by family physicians is more concerning. Many of my graduates report feeling disconnected from their patients, because they begin to feel disillusioned by the demands and requests that practice and patients place on them. The paperwork, “permission slips,” and requests for tests and studies not only feel overwhelming and exhausting but also create distance between physicians and patients.7 We want to help our patients, so we do the forms and order the tests. As the quantity of forms, slips, and requests adds up, we begin to feel resentful at what the forms take away: time with our patients, perhaps, or time with our families. We get angry at the forms and the “asks,” and then begin to get angry at the patients simply for having needs. Administrative burden is a hassle, but it is also insidiously destructive.8
Family physicians confront hopelessness when, day after day, we diagnose problems that no physician is likely to fix in a single office visit: chronic stress, family dysfunction, violence, unemployment, poverty, racism, loneliness, and the hopelessness of the patients themselves. This is not to say that we ignore these concerns or their impact on health. It is because we see and feel them, and deeply understand their consequences for our patients, that we grow frustrated with the lack of solutions.9,10
Thankfully, we have strong teams working at the policy level to improve the primary care and public health infrastructure so that we can maintain some hope that it will be better in the future. Sometimes when I counsel a former resident, they decide to join those teams so that they can work on the solutions. Others decide to expand their scope of practice. Others seek out virtual scribes to streamline charting and regain time. Some build better boundaries with their EHR inboxes.
The key is figuring out what we can do and making peace with our limits. When disillusionment hits, what we can do includes seeking connection and social contact and remembering that we are not trapped in our situation, even if we are practicing in a less-than-functional health care system. There are many ways to “be” a family physician—if what you’re doing isn’t working for you, look for opportunities (big or small) that make it better. We can all reach out to coaches, therapists, colleagues, and friends for support to remain steadfast in our purpose as family physicians. This support and the power of change means that from residency to the latter parts of our careers, we will continue to bring the tremendous good of family medicine to the communities we serve.
1. Coutinho AJ, Cochrane A, Stelter K, et al. Comparison of intended scope of practice for family medicine residents with reported scope of practice among practicing family physicians. JAMA. 2015;314:2364-2372. doi: 10.1001/jama.2015.13734
2. Weidner AKH, Phillips RL, Fang B, et al. Burnout and scope of practice in new family physicians. Ann Fam Med. 2018;16:200-205. doi: 10.1370/afm.2221
3. Zomahoun HT, Samson I, Sawadogo J, et al. Effects of the scope of practice on family physicians: a systematic review. BMC Family Practice. 2021;22. doi: 10.1186/s12875-020-01328-1
4. Killeen D, Jetty A, Peterson LE, et al. The association of practice type and the scope of care of family physicians. J Am Board Fam Med. 2023;36:79-87. doi: 10.3122/jabfm.2022.220172R1
5. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83:457-502. doi: 10.1111/j.1468-0009.2005.00409.x
6. Ferrer RL. Pursuing equity: contact with primary care and specialist clinicians by demographics, insurance, and health status. Ann Fam Med. 2007;5:492-502. doi: 10.1370/afm.746
7. Rao SK, Kimball AB, Lehrhoff SR, et al. The impact of administrative burden on academic physicians: results of a hospital-wide physician survey. Acad Med. 2017;92:237-243. doi: 10.1097/ACM.0000000000001461
8. McMahon LF, Rize K, Irby-Johnson N, et al. Designed to fail? The future of primary care. J Gen Intern Med. 2021;36:515-517. doi: 10.1007/s11606-020-06077-6
9. Welles CC, Tong A, Brereton E, et al. Sources of clinician burnout in providing care for underserved patients in a safety-net healthcare system. J Gen Intern Med. 2023;38:1468-1475. doi: 10.1007/s11606-022-07896-5
10. Kung A, Cheung T, Knox M, et al. Capacity to address social needs affects primary care clinician burnout. Ann Fam Med. 2019;17:487-494. doi: 10.1370/afm.2470
This is the exciting time of year when we graduate new classes of medical students and residents. Med school graduation brings mixed emotions; the new doctors and I both know residency will bring growth and challenges. Residency graduation is a wistful passage as well. It is so rewarding to welcome the newly board-certified family physicians to family medicine, but we miss them even as we orient a new class.
Every year, a few months (or even a few years) after graduation, I hear from a former resident, sometimes several. They ask to talk and, although it can be hard for them to explain exactly the ennui and disillusionment they’re feeling, their concerns boil down to: Is this all there is?
They are not burnt out, exactly, but they were hoping for more from their careers in family medicine.1 They find their hopes and expectations are not fulfilled by seeing patients in the office 8 hours per day, 4.5 days per week. Even those who report rewarding relationships with patients express less overall enthusiasm for jobs they were excited to start just months or years earlier.
Some of the difficulties I hear the graduates report are expected growing pains. It is a transition to go from supervised practice with attending backup to a setting where you are on your own, typically with a 4-fold increase in volume compared with residency. But the monotony is real for family physicians in full-time outpatient practice.
Research suggests an expanded scope of practice—including hospital medicine, obstetrics, and procedures—is associated with physician well-being.2,3 A broad scope of practice can bring stress, but it also brings meaning, and that meaning is protective to our well-being. However, a robust scope of practice is not always supported by medical groups or hospital systems, who prefer a more compartmentalized, widgetized physician.4 It would be easier for their algorithms if family physicians picked a lane and stayed in it. Alas, the broader our scope of practice, the healthier our population, the more equitable our care,5,6 and the happier our physicians.
The disconnect and hopelessness experienced by family physicians is more concerning. Many of my graduates report feeling disconnected from their patients, because they begin to feel disillusioned by the demands and requests that practice and patients place on them. The paperwork, “permission slips,” and requests for tests and studies not only feel overwhelming and exhausting but also create distance between physicians and patients.7 We want to help our patients, so we do the forms and order the tests. As the quantity of forms, slips, and requests adds up, we begin to feel resentful at what the forms take away: time with our patients, perhaps, or time with our families. We get angry at the forms and the “asks,” and then begin to get angry at the patients simply for having needs. Administrative burden is a hassle, but it is also insidiously destructive.8
Family physicians confront hopelessness when, day after day, we diagnose problems that no physician is likely to fix in a single office visit: chronic stress, family dysfunction, violence, unemployment, poverty, racism, loneliness, and the hopelessness of the patients themselves. This is not to say that we ignore these concerns or their impact on health. It is because we see and feel them, and deeply understand their consequences for our patients, that we grow frustrated with the lack of solutions.9,10
Thankfully, we have strong teams working at the policy level to improve the primary care and public health infrastructure so that we can maintain some hope that it will be better in the future. Sometimes when I counsel a former resident, they decide to join those teams so that they can work on the solutions. Others decide to expand their scope of practice. Others seek out virtual scribes to streamline charting and regain time. Some build better boundaries with their EHR inboxes.
The key is figuring out what we can do and making peace with our limits. When disillusionment hits, what we can do includes seeking connection and social contact and remembering that we are not trapped in our situation, even if we are practicing in a less-than-functional health care system. There are many ways to “be” a family physician—if what you’re doing isn’t working for you, look for opportunities (big or small) that make it better. We can all reach out to coaches, therapists, colleagues, and friends for support to remain steadfast in our purpose as family physicians. This support and the power of change means that from residency to the latter parts of our careers, we will continue to bring the tremendous good of family medicine to the communities we serve.
This is the exciting time of year when we graduate new classes of medical students and residents. Med school graduation brings mixed emotions; the new doctors and I both know residency will bring growth and challenges. Residency graduation is a wistful passage as well. It is so rewarding to welcome the newly board-certified family physicians to family medicine, but we miss them even as we orient a new class.
Every year, a few months (or even a few years) after graduation, I hear from a former resident, sometimes several. They ask to talk and, although it can be hard for them to explain exactly the ennui and disillusionment they’re feeling, their concerns boil down to: Is this all there is?
They are not burnt out, exactly, but they were hoping for more from their careers in family medicine.1 They find their hopes and expectations are not fulfilled by seeing patients in the office 8 hours per day, 4.5 days per week. Even those who report rewarding relationships with patients express less overall enthusiasm for jobs they were excited to start just months or years earlier.
Some of the difficulties I hear the graduates report are expected growing pains. It is a transition to go from supervised practice with attending backup to a setting where you are on your own, typically with a 4-fold increase in volume compared with residency. But the monotony is real for family physicians in full-time outpatient practice.
Research suggests an expanded scope of practice—including hospital medicine, obstetrics, and procedures—is associated with physician well-being.2,3 A broad scope of practice can bring stress, but it also brings meaning, and that meaning is protective to our well-being. However, a robust scope of practice is not always supported by medical groups or hospital systems, who prefer a more compartmentalized, widgetized physician.4 It would be easier for their algorithms if family physicians picked a lane and stayed in it. Alas, the broader our scope of practice, the healthier our population, the more equitable our care,5,6 and the happier our physicians.
The disconnect and hopelessness experienced by family physicians is more concerning. Many of my graduates report feeling disconnected from their patients, because they begin to feel disillusioned by the demands and requests that practice and patients place on them. The paperwork, “permission slips,” and requests for tests and studies not only feel overwhelming and exhausting but also create distance between physicians and patients.7 We want to help our patients, so we do the forms and order the tests. As the quantity of forms, slips, and requests adds up, we begin to feel resentful at what the forms take away: time with our patients, perhaps, or time with our families. We get angry at the forms and the “asks,” and then begin to get angry at the patients simply for having needs. Administrative burden is a hassle, but it is also insidiously destructive.8
Family physicians confront hopelessness when, day after day, we diagnose problems that no physician is likely to fix in a single office visit: chronic stress, family dysfunction, violence, unemployment, poverty, racism, loneliness, and the hopelessness of the patients themselves. This is not to say that we ignore these concerns or their impact on health. It is because we see and feel them, and deeply understand their consequences for our patients, that we grow frustrated with the lack of solutions.9,10
Thankfully, we have strong teams working at the policy level to improve the primary care and public health infrastructure so that we can maintain some hope that it will be better in the future. Sometimes when I counsel a former resident, they decide to join those teams so that they can work on the solutions. Others decide to expand their scope of practice. Others seek out virtual scribes to streamline charting and regain time. Some build better boundaries with their EHR inboxes.
The key is figuring out what we can do and making peace with our limits. When disillusionment hits, what we can do includes seeking connection and social contact and remembering that we are not trapped in our situation, even if we are practicing in a less-than-functional health care system. There are many ways to “be” a family physician—if what you’re doing isn’t working for you, look for opportunities (big or small) that make it better. We can all reach out to coaches, therapists, colleagues, and friends for support to remain steadfast in our purpose as family physicians. This support and the power of change means that from residency to the latter parts of our careers, we will continue to bring the tremendous good of family medicine to the communities we serve.
1. Coutinho AJ, Cochrane A, Stelter K, et al. Comparison of intended scope of practice for family medicine residents with reported scope of practice among practicing family physicians. JAMA. 2015;314:2364-2372. doi: 10.1001/jama.2015.13734
2. Weidner AKH, Phillips RL, Fang B, et al. Burnout and scope of practice in new family physicians. Ann Fam Med. 2018;16:200-205. doi: 10.1370/afm.2221
3. Zomahoun HT, Samson I, Sawadogo J, et al. Effects of the scope of practice on family physicians: a systematic review. BMC Family Practice. 2021;22. doi: 10.1186/s12875-020-01328-1
4. Killeen D, Jetty A, Peterson LE, et al. The association of practice type and the scope of care of family physicians. J Am Board Fam Med. 2023;36:79-87. doi: 10.3122/jabfm.2022.220172R1
5. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83:457-502. doi: 10.1111/j.1468-0009.2005.00409.x
6. Ferrer RL. Pursuing equity: contact with primary care and specialist clinicians by demographics, insurance, and health status. Ann Fam Med. 2007;5:492-502. doi: 10.1370/afm.746
7. Rao SK, Kimball AB, Lehrhoff SR, et al. The impact of administrative burden on academic physicians: results of a hospital-wide physician survey. Acad Med. 2017;92:237-243. doi: 10.1097/ACM.0000000000001461
8. McMahon LF, Rize K, Irby-Johnson N, et al. Designed to fail? The future of primary care. J Gen Intern Med. 2021;36:515-517. doi: 10.1007/s11606-020-06077-6
9. Welles CC, Tong A, Brereton E, et al. Sources of clinician burnout in providing care for underserved patients in a safety-net healthcare system. J Gen Intern Med. 2023;38:1468-1475. doi: 10.1007/s11606-022-07896-5
10. Kung A, Cheung T, Knox M, et al. Capacity to address social needs affects primary care clinician burnout. Ann Fam Med. 2019;17:487-494. doi: 10.1370/afm.2470
1. Coutinho AJ, Cochrane A, Stelter K, et al. Comparison of intended scope of practice for family medicine residents with reported scope of practice among practicing family physicians. JAMA. 2015;314:2364-2372. doi: 10.1001/jama.2015.13734
2. Weidner AKH, Phillips RL, Fang B, et al. Burnout and scope of practice in new family physicians. Ann Fam Med. 2018;16:200-205. doi: 10.1370/afm.2221
3. Zomahoun HT, Samson I, Sawadogo J, et al. Effects of the scope of practice on family physicians: a systematic review. BMC Family Practice. 2021;22. doi: 10.1186/s12875-020-01328-1
4. Killeen D, Jetty A, Peterson LE, et al. The association of practice type and the scope of care of family physicians. J Am Board Fam Med. 2023;36:79-87. doi: 10.3122/jabfm.2022.220172R1
5. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83:457-502. doi: 10.1111/j.1468-0009.2005.00409.x
6. Ferrer RL. Pursuing equity: contact with primary care and specialist clinicians by demographics, insurance, and health status. Ann Fam Med. 2007;5:492-502. doi: 10.1370/afm.746
7. Rao SK, Kimball AB, Lehrhoff SR, et al. The impact of administrative burden on academic physicians: results of a hospital-wide physician survey. Acad Med. 2017;92:237-243. doi: 10.1097/ACM.0000000000001461
8. McMahon LF, Rize K, Irby-Johnson N, et al. Designed to fail? The future of primary care. J Gen Intern Med. 2021;36:515-517. doi: 10.1007/s11606-020-06077-6
9. Welles CC, Tong A, Brereton E, et al. Sources of clinician burnout in providing care for underserved patients in a safety-net healthcare system. J Gen Intern Med. 2023;38:1468-1475. doi: 10.1007/s11606-022-07896-5
10. Kung A, Cheung T, Knox M, et al. Capacity to address social needs affects primary care clinician burnout. Ann Fam Med. 2019;17:487-494. doi: 10.1370/afm.2470
Pedunculated gluteal mass
A 30-YEAR-OLD MAN presented for evaluation of a solitary, flesh-colored, pedunculated mass on his right inner gluteal area (FIGURE) that had gradually enlarged over the previous 18 months. The lesion had manifested 4 years prior as a small papule that was stable for many years. It began to grow steadily after the patient compressed the papule forcefully. Activities of daily living, such as sitting, were now uncomfortable, so he sought treatment. He denied pain, pruritis, and bleeding and reported no history of trauma or surgery in the area of the mass.
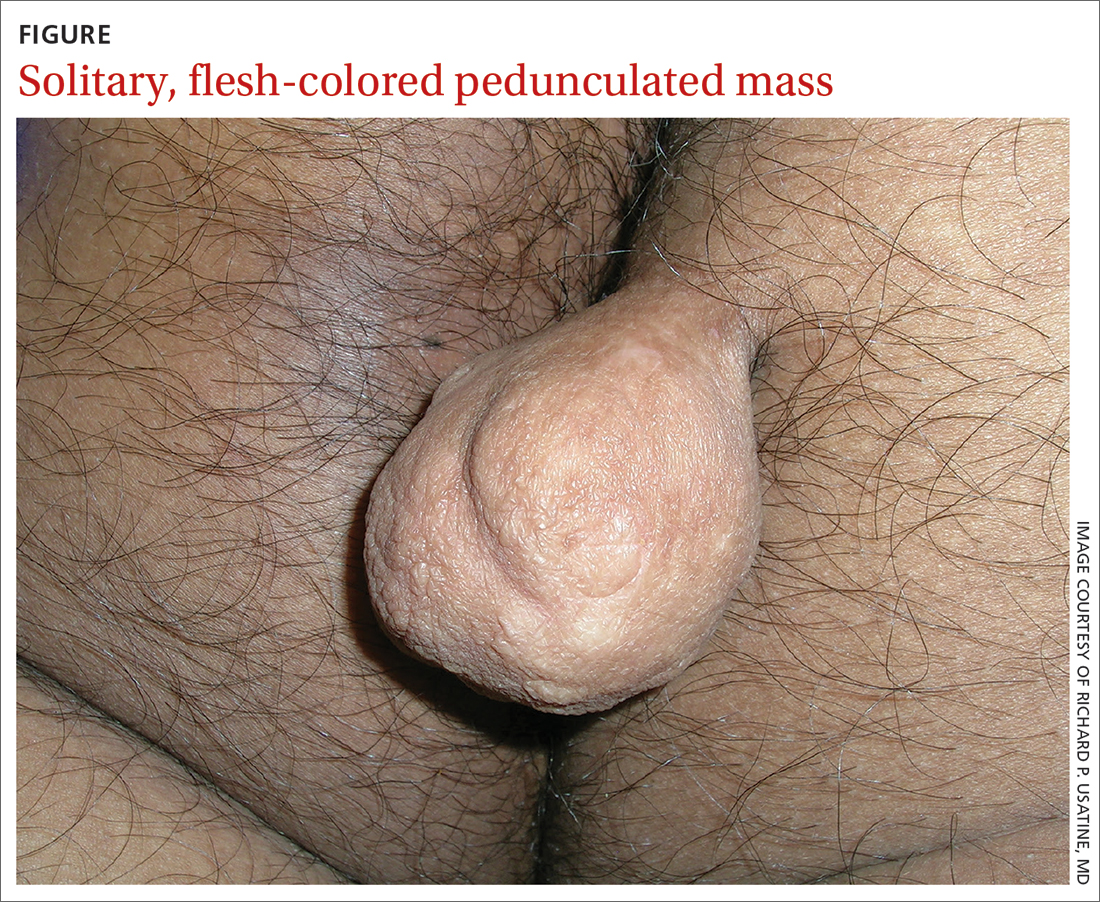
On physical examination, the mass measured 3.5 × 4.5 cm with a 1.2-cm base. It was smooth, soft, nontender, and compressible—but nonfluctuant. There were no signs of ulceration or bleeding. No regional lymphadenopathy was noted. An excisional biopsy was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Fibrolipoma
The biopsy confirmed a diagnosis of fibrolipoma—a rare variant of lipoma composed of a mixture of adipocytes and thick bands of fibrous connective tissues.1 Etiology for fibrolipomas is unknown. Blunt trauma rupture of the fibrous septa that prevent fat migration may result in a proliferation of adipose tissue and thereby enlargement of fibrolipomas and other lipoma variants.2 In this case, the patient’s compression of the original papule likely served as the trauma that led to its enlargement. Malignant change has not been reported with fibrolipomas.
What you’ll see—and on whom. Fibrolipomas typically are flesh-colored, pedunculated, compressible, and relatively asymptomatic.3 They have been reported on the face, neck, back, and pubic areas, among other locations. Size is variable; they can be as small as 1 cm in diameter and as large as 10 cm in diameter.4 However, fibrolipomas can grow to be “giant” if they exceed 10 cm (or 1000 g).2
Men and women are affected equally by fibrolipomas. Prevalence does not differ by race or ethnicity.
The differential include sother lipomas and skin tags
The differential for a mass such as this one includes lipomas, acrochordons (also known as skin tags), and fibrokeratomas.
Lipomas are the most common benign soft-tissue tumors and are composed of adipocytes.5 The fibrolipoma is just one variant of lipoma; others include the myxolipoma, myolipoma, spindle cell lipoma, angiolipoma, osteolipoma, and chondrolipoma.2 Lipomas typically are subcutaneous and located over the scalp, neck, and upper trunk area but can occur anywhere on the body. They are mobile and typically well circumscribed. Lipomas have a broad base with well-demarcated swelling; fibrolipomas are usually pedunculated.
Continue to: Acrochordons ("skin tags")
Acrochordons (“skin tags”) usually contain a peduncle but may be sessile. They range from 1 mm to 1 cm in diameter and typically are located in skin folds, especially in the neck, axillae, and inguinal areas.6 Obesity, older
Fibrokeratomas typically are benign, solitary, fibrous tissue tumors that are found on fingers and seldom are pedunculated. They are flesh-colored and conical or nodular, with a hyperkeratotic collar. Fibrokeratomas are smaller and thicker than fibromas, as well as firm in consistency. They are acquired tumors that have been shown to be related to repetitive trauma.6
Treatment involves surgical excision
The preferred treatment for fibrolipoma is complete surgical excision, although cryotherapy is another option for lesions < 1 cm.4 Without surgical excision, the mass will continue to grow, albeit slowly.
This patient’s mass was excised successfully in its entirety; there were no complications. Follow-up is usually unnecessary.
1. Kim YT, Kim WS, Park YL, et al. A case of fibrolipoma. Korean J Dermatol. 2003;41:939-941.
2. Mazzocchi M, Onesti MG, Pasquini P, et al. Giant fibrolipoma in the leg—a case report. Anticancer Res. 2006;26:3649-3654.
3. Shin SJ. Subcutaneous fibrolipoma on the back. J Craniofac Surg. 2013;24:1051-1053. doi: 10.1097/SCS.0b013e3182802517
4. Suleiman J, Suleman M, Amsi P, et al. Giant pedunculated lipofibroma of the thigh. J Surg Case Rep. 2023;2023(3):rjad153. doi: 10.1093/jscr/rjad153
5. Dai X-M, Li Y-S, Liu H, et al. Giant pedunculated fibrolipoma arising from right facial and cervical region. J Oral and Maxillofac Surg. 2009;67:1323-1326. doi: 10.1016/j.joms.2008.12.037
6. Lee JA, Khodaee M. Enlarging, pedunculated skin lesion. Am Fam Physician. 2012;85:1191-1192.
7. Banik R, Lubach D. Skin tags: localization and frequencies according to sex and age. Dermatologica. 1987;174:180-183. doi: 10.1159/000249169
A 30-YEAR-OLD MAN presented for evaluation of a solitary, flesh-colored, pedunculated mass on his right inner gluteal area (FIGURE) that had gradually enlarged over the previous 18 months. The lesion had manifested 4 years prior as a small papule that was stable for many years. It began to grow steadily after the patient compressed the papule forcefully. Activities of daily living, such as sitting, were now uncomfortable, so he sought treatment. He denied pain, pruritis, and bleeding and reported no history of trauma or surgery in the area of the mass.

On physical examination, the mass measured 3.5 × 4.5 cm with a 1.2-cm base. It was smooth, soft, nontender, and compressible—but nonfluctuant. There were no signs of ulceration or bleeding. No regional lymphadenopathy was noted. An excisional biopsy was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Fibrolipoma
The biopsy confirmed a diagnosis of fibrolipoma—a rare variant of lipoma composed of a mixture of adipocytes and thick bands of fibrous connective tissues.1 Etiology for fibrolipomas is unknown. Blunt trauma rupture of the fibrous septa that prevent fat migration may result in a proliferation of adipose tissue and thereby enlargement of fibrolipomas and other lipoma variants.2 In this case, the patient’s compression of the original papule likely served as the trauma that led to its enlargement. Malignant change has not been reported with fibrolipomas.
What you’ll see—and on whom. Fibrolipomas typically are flesh-colored, pedunculated, compressible, and relatively asymptomatic.3 They have been reported on the face, neck, back, and pubic areas, among other locations. Size is variable; they can be as small as 1 cm in diameter and as large as 10 cm in diameter.4 However, fibrolipomas can grow to be “giant” if they exceed 10 cm (or 1000 g).2
Men and women are affected equally by fibrolipomas. Prevalence does not differ by race or ethnicity.
The differential include sother lipomas and skin tags
The differential for a mass such as this one includes lipomas, acrochordons (also known as skin tags), and fibrokeratomas.
Lipomas are the most common benign soft-tissue tumors and are composed of adipocytes.5 The fibrolipoma is just one variant of lipoma; others include the myxolipoma, myolipoma, spindle cell lipoma, angiolipoma, osteolipoma, and chondrolipoma.2 Lipomas typically are subcutaneous and located over the scalp, neck, and upper trunk area but can occur anywhere on the body. They are mobile and typically well circumscribed. Lipomas have a broad base with well-demarcated swelling; fibrolipomas are usually pedunculated.
Continue to: Acrochordons ("skin tags")
Acrochordons (“skin tags”) usually contain a peduncle but may be sessile. They range from 1 mm to 1 cm in diameter and typically are located in skin folds, especially in the neck, axillae, and inguinal areas.6 Obesity, older
Fibrokeratomas typically are benign, solitary, fibrous tissue tumors that are found on fingers and seldom are pedunculated. They are flesh-colored and conical or nodular, with a hyperkeratotic collar. Fibrokeratomas are smaller and thicker than fibromas, as well as firm in consistency. They are acquired tumors that have been shown to be related to repetitive trauma.6
Treatment involves surgical excision
The preferred treatment for fibrolipoma is complete surgical excision, although cryotherapy is another option for lesions < 1 cm.4 Without surgical excision, the mass will continue to grow, albeit slowly.
This patient’s mass was excised successfully in its entirety; there were no complications. Follow-up is usually unnecessary.
A 30-YEAR-OLD MAN presented for evaluation of a solitary, flesh-colored, pedunculated mass on his right inner gluteal area (FIGURE) that had gradually enlarged over the previous 18 months. The lesion had manifested 4 years prior as a small papule that was stable for many years. It began to grow steadily after the patient compressed the papule forcefully. Activities of daily living, such as sitting, were now uncomfortable, so he sought treatment. He denied pain, pruritis, and bleeding and reported no history of trauma or surgery in the area of the mass.

On physical examination, the mass measured 3.5 × 4.5 cm with a 1.2-cm base. It was smooth, soft, nontender, and compressible—but nonfluctuant. There were no signs of ulceration or bleeding. No regional lymphadenopathy was noted. An excisional biopsy was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Fibrolipoma
The biopsy confirmed a diagnosis of fibrolipoma—a rare variant of lipoma composed of a mixture of adipocytes and thick bands of fibrous connective tissues.1 Etiology for fibrolipomas is unknown. Blunt trauma rupture of the fibrous septa that prevent fat migration may result in a proliferation of adipose tissue and thereby enlargement of fibrolipomas and other lipoma variants.2 In this case, the patient’s compression of the original papule likely served as the trauma that led to its enlargement. Malignant change has not been reported with fibrolipomas.
What you’ll see—and on whom. Fibrolipomas typically are flesh-colored, pedunculated, compressible, and relatively asymptomatic.3 They have been reported on the face, neck, back, and pubic areas, among other locations. Size is variable; they can be as small as 1 cm in diameter and as large as 10 cm in diameter.4 However, fibrolipomas can grow to be “giant” if they exceed 10 cm (or 1000 g).2
Men and women are affected equally by fibrolipomas. Prevalence does not differ by race or ethnicity.
The differential include sother lipomas and skin tags
The differential for a mass such as this one includes lipomas, acrochordons (also known as skin tags), and fibrokeratomas.
Lipomas are the most common benign soft-tissue tumors and are composed of adipocytes.5 The fibrolipoma is just one variant of lipoma; others include the myxolipoma, myolipoma, spindle cell lipoma, angiolipoma, osteolipoma, and chondrolipoma.2 Lipomas typically are subcutaneous and located over the scalp, neck, and upper trunk area but can occur anywhere on the body. They are mobile and typically well circumscribed. Lipomas have a broad base with well-demarcated swelling; fibrolipomas are usually pedunculated.
Continue to: Acrochordons ("skin tags")
Acrochordons (“skin tags”) usually contain a peduncle but may be sessile. They range from 1 mm to 1 cm in diameter and typically are located in skin folds, especially in the neck, axillae, and inguinal areas.6 Obesity, older
Fibrokeratomas typically are benign, solitary, fibrous tissue tumors that are found on fingers and seldom are pedunculated. They are flesh-colored and conical or nodular, with a hyperkeratotic collar. Fibrokeratomas are smaller and thicker than fibromas, as well as firm in consistency. They are acquired tumors that have been shown to be related to repetitive trauma.6
Treatment involves surgical excision
The preferred treatment for fibrolipoma is complete surgical excision, although cryotherapy is another option for lesions < 1 cm.4 Without surgical excision, the mass will continue to grow, albeit slowly.
This patient’s mass was excised successfully in its entirety; there were no complications. Follow-up is usually unnecessary.
1. Kim YT, Kim WS, Park YL, et al. A case of fibrolipoma. Korean J Dermatol. 2003;41:939-941.
2. Mazzocchi M, Onesti MG, Pasquini P, et al. Giant fibrolipoma in the leg—a case report. Anticancer Res. 2006;26:3649-3654.
3. Shin SJ. Subcutaneous fibrolipoma on the back. J Craniofac Surg. 2013;24:1051-1053. doi: 10.1097/SCS.0b013e3182802517
4. Suleiman J, Suleman M, Amsi P, et al. Giant pedunculated lipofibroma of the thigh. J Surg Case Rep. 2023;2023(3):rjad153. doi: 10.1093/jscr/rjad153
5. Dai X-M, Li Y-S, Liu H, et al. Giant pedunculated fibrolipoma arising from right facial and cervical region. J Oral and Maxillofac Surg. 2009;67:1323-1326. doi: 10.1016/j.joms.2008.12.037
6. Lee JA, Khodaee M. Enlarging, pedunculated skin lesion. Am Fam Physician. 2012;85:1191-1192.
7. Banik R, Lubach D. Skin tags: localization and frequencies according to sex and age. Dermatologica. 1987;174:180-183. doi: 10.1159/000249169
1. Kim YT, Kim WS, Park YL, et al. A case of fibrolipoma. Korean J Dermatol. 2003;41:939-941.
2. Mazzocchi M, Onesti MG, Pasquini P, et al. Giant fibrolipoma in the leg—a case report. Anticancer Res. 2006;26:3649-3654.
3. Shin SJ. Subcutaneous fibrolipoma on the back. J Craniofac Surg. 2013;24:1051-1053. doi: 10.1097/SCS.0b013e3182802517
4. Suleiman J, Suleman M, Amsi P, et al. Giant pedunculated lipofibroma of the thigh. J Surg Case Rep. 2023;2023(3):rjad153. doi: 10.1093/jscr/rjad153
5. Dai X-M, Li Y-S, Liu H, et al. Giant pedunculated fibrolipoma arising from right facial and cervical region. J Oral and Maxillofac Surg. 2009;67:1323-1326. doi: 10.1016/j.joms.2008.12.037
6. Lee JA, Khodaee M. Enlarging, pedunculated skin lesion. Am Fam Physician. 2012;85:1191-1192.
7. Banik R, Lubach D. Skin tags: localization and frequencies according to sex and age. Dermatologica. 1987;174:180-183. doi: 10.1159/000249169
64-year-old woman • hot flashes, facial flushing, excessive sweating, and palpitations • daily headaches • history of hypertension • Dx?
THE CASE
A 64-year-old woman sought care after having hot flashes, facial flushing, excessive sweating, palpitations, and daily headaches for 1 month. She had a history of hypertension that was well controlled with hydrochlorothiazide 25 mg/d but over the previous month, it had become more difficult to control. Her blood pressure remained elevated to 150/100 mm Hg despite the addition of lisinopril 40 mg/d and amlodipine 10 mg/d, indicating resistant hypertension. She had no family history of hypertension, diabetes, or obesity or any other pertinent medical or surgical history. Physical examination was negative for weight gain, stretch marks, or muscle weakness.
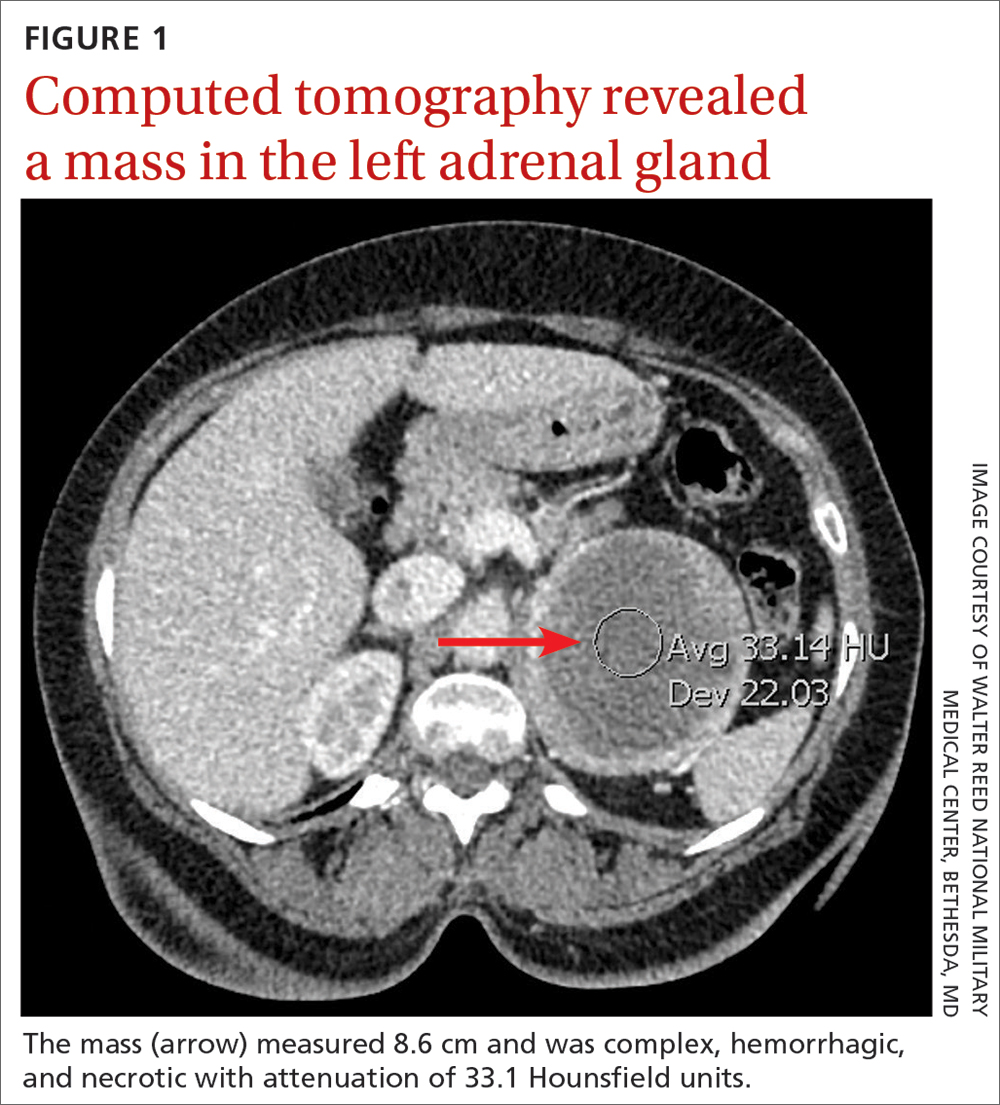
Laboratory tests revealed a normal serum aldosterone-renin ratio, renal function, and thyroid function; however, she had elevated levels of normetanephrine (2429 pg/mL; normal range, 0-145 pg/mL) and metanephrine (143 pg/mL; normal range, 0-62 pg/mL). Computed tomography (CT) revealed an 8.6-cm complex, hemorrhagic, necrotic left adrenal mass with attenuation of 33.1 Hounsfield units (HU) (FIGURE 1). Magnetic resonance imaging (MRI) demonstrated a T2 hyperintense left adrenal mass. An evaluation for Cushing syndrome was negative, and positron emission tomography (PET)/CT with gallium-68 dotatate was ordered. It showed intense radiotracer uptake in the left adrenal gland, with a maximum standardized uptake value of 70.1 (FIGURE 2).
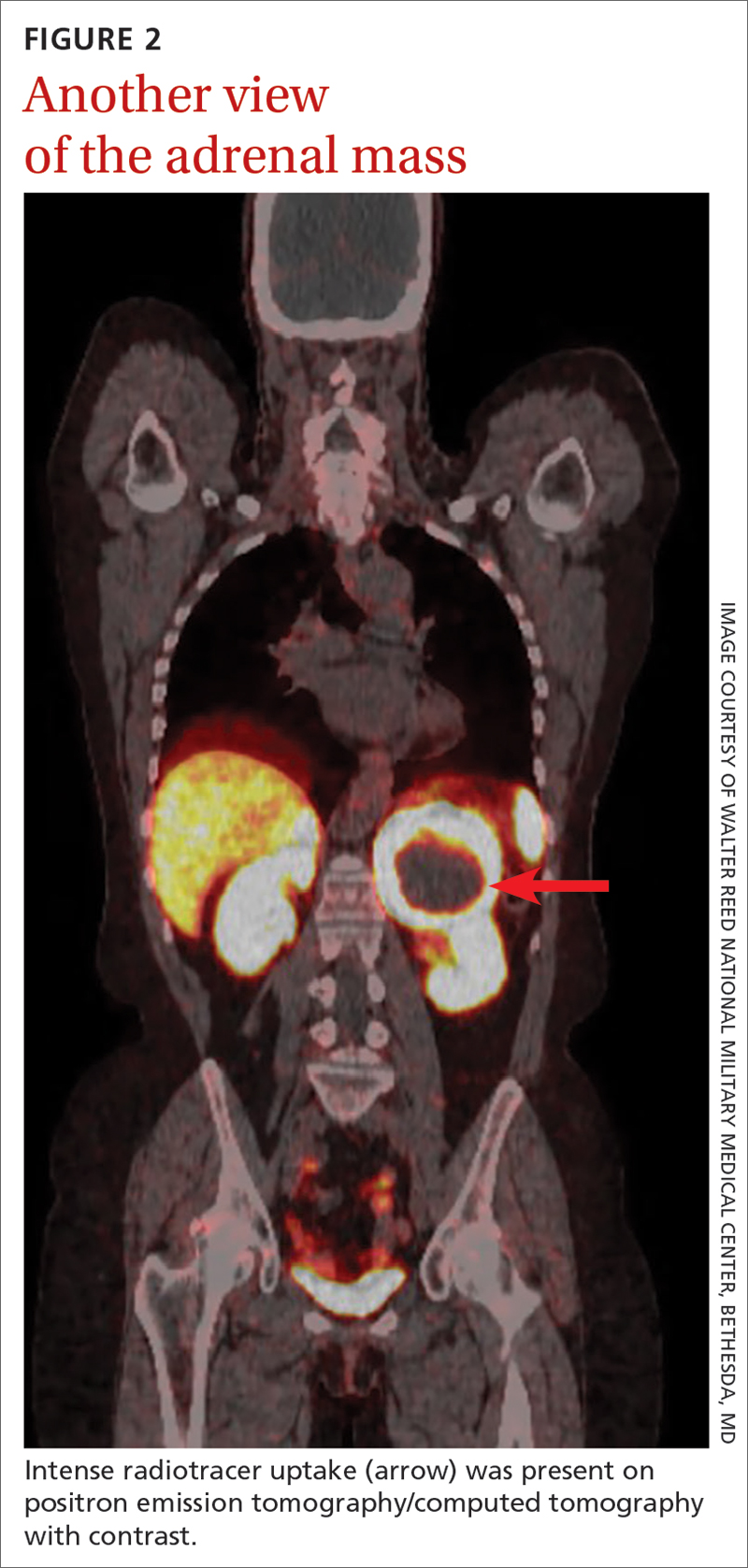
THE DIAGNOSIS
After appropriate preparation with alpha blockade (phenoxybenzamine 20 mg twice daily for 7 days) and fluid resuscitation (normal saline run over 12 hours preoperatively), the patient underwent successful open surgical resection of the adrenal mass, during which her blood pressure was controlled with a nitroprusside infusion and boluses of esmolol and labetalol. Pathology results showed cells in a nested pattern with round to oval nuclei in a vascular background. There was no necrosis, increased mitotic figures, capsular invasion, or increased cellularity. Chromogranin immunohistochemical staining was positive. Given her resistant hypertension, clinical symptoms, and pathology results, the patient was given a diagnosis of pheochromocytoma.
DISCUSSION
Resistant hypertension is defined as blood pressure that is elevated above goal despite the use of 3 maximally titrated antihypertensive agents from different classes or that is well controlled with at least 4 antihypertensive medications.1 The prevalence of resistant hypertension is 12% to 18% in adults being treated for hypertension.1 Patients with resistant hypertension have a higher risk for cardiovascular events and death, are more likely to have a secondary cause of hypertension, and may benefit from special diagnostic testing or treatment approaches to control their blood pressure.1
There are many causes of resistant hypertension; primary aldosteronism is the most common cause (prevalence as high as 20%).2 Given the increased risk for cardiovascular/cerebrovascular disease, all patients with resistant hypertension should be screened for this condition.2 Other causes of resistant hypertension include renal parenchymal disease, renal artery stenosis, coarctation of the aorta, thyroid dysfunction, Cushing syndrome, paraganglioma, and as seen in our case, pheochromocytoma. Although pheochromocytoma is a rare cause of resistant hypertension (0.01%-4%),1 it is associated with high rates of morbidity and mortality if left untreated and may be inherited, making it an essential diagnosis to consider in all patients with resistant hypertension.1,3
Common symptoms of pheochromocytoma are hypertension (paroxysmal or sustained), headaches, palpitations, pallor, and piloerection (or cold sweats).1 Patients with pheochromocytoma typically exhibit metanephrine levels that are more than 4 times the upper limit of normal.4 Therefore, measurement of plasma free metanephrines or urinary fractionated metanephrines is recommended.5 Elevated metanephrine levels also are caused by obesity, obstructive sleep apnea, and certain medications and should be ruled out.5
All pheochromocytomas are potentially malignant. Despite the existence of pathologic scoring systems6,7 and radiographic features that suggest malignancy,8,9 no single risk-stratification tool is recommended in the current literature.10 Ultimately, the only way to confirm malignancy is to see metastases where chromaffin tissue is not normally found on imaging.10
Continue to: Pathologic features to look for...
Pathologic features to look for include capsular/periadrenal adipose invasion, increased cellularity, necrosis, tumor cell spindling, increased/atypical mitotic figures, and nuclear pleomorphism. Radiographic features include larger size (≥ 4-6 cm),11 an irregular shape, necrosis, calcifications, attenuation of 10 HU or higher on noncontrast CT, absolute washout of 60% or lower, and relative washout of 40% or lower.8,12 On MRI, malignant lesions appear hypointense on T1-weighted imaging and hyperintense on T2-weighted imaging.9 Fluorodeoxyglucose avidity on PET scan also is indicative of malignancy.8,9
Treatment for pheochromocytoma is surgical resection. An experienced surgical team and proper preoperative preparation are necessary because the induction of anesthesia, endotracheal intubation, and tumor manipulation can lead to a release of catecholamines, potentially resulting in an intraoperative hypertensive crisis, cardiac arrhythmias, and multiorgan failure.
Proper preoperative preparation includes taking an alpha-adrenergic blocker, such as phenoxybenzamine, prazosin, terazosin, or doxazosin, for at least 7 days to normalize the patient’s blood pressure. Patients should be counseled that they may experience nasal congestion, orthostasis, and fatigue while taking these medications. Volume expansion with intravenous fluids also should be performed and a high-salt diet considered. Beta-adrenergic blockade can be initiated once appropriate alpha-adrenergic blockade is achieved to control the patient’s heart rate; beta-blockers should never be started first because of the risk for severe hypertension. Careful hemodynamic monitoring is vital intraoperatively and postoperatively.5,13 Because metastatic lesions can occur decades after resection, long-term follow-up is critical.5,10
Following tumor resection, our patient’s blood pressure was supported with intravenous fluids and phenylephrine. She was able to discontinue all her antihypertensive medications postoperatively, and her plasma free and urinary fractionated metanephrine levels returned to within normal limits 8 weeks after surgery. Five years after surgery, she continues to have no signs of recurrence, as evidenced by annual negative plasma free metanephrines testing and abdominal/pelvic CT.
THE TAKEAWAY
This case highlights the importance of recognizing resistant hypertension and a potential secondary cause of this disease—pheochromocytoma. Although rare, pheochromocytomas confer increased risk for cardiovascular disease and death. Thus, swift recognition and proper preparation for surgical resection are necessary. Malignant lesions can be diagnosed only upon discovery of metastatic disease and can recur for decades after surgical resection, making diligent long-term follow-up imperative.
CORRESPONDENCE
Nicole O. Vietor, MD, Division of Endocrinology, Walter Reed National Military Medical Center, 8901 Wisconsin Avenue, Bethesda, MD 20889; [email protected]
1. Carey RM, Calhoun DA, Bakris GL, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72:e53-e90. doi: 10.1161/HYP.0000000000000084
2. Young WF Jr. Diagnosis and treatment of primary aldosteronism: practical clinical perspectives. J Intern Med. 2019;285:126-148. doi: 10.1111/joim.12831
3. Young WF Jr, Calhoun DA, Lenders JWM, et al. Screening for endocrine hypertension: an Endocrine Society Scientific Statement. Endocr Rev. 2017;38:103-122. doi: 10.1210/er.2017-00054
4. Lenders JWM, Pacak K, Walther MM, et al. Biochemical diagnosis of pheochromocytoma: which test is best? JAMA. 2002;287:1427-1434. doi: 10.1001/jama.287.11.1427
5. Lenders JW, Duh Q-Y, Eisenhofer G, et al. Pheochromocytoma and paraganglioma: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2014;99:1915-1942. doi: 10.1210/jc.2014-1498
6. Kimura N, Takayanagi R, Takizawa N, et al. Pathological grading for predicting metastasis in phaeochromocytoma and paraganglioma. Endocr Relat Cancer. 2014;21:405-414. doi: 10.1530/ERC-13-0494
7. Thompson LDR. Pheochromocytoma of the Adrenal gland Scaled Score (PASS) to separate benign from malignant neoplasms: a clinicopathologic and immunophenotypic study of 100 cases. Am J Surg Pathol. 2002;26:551-566. doi: 10.1097/00000478-200205000-00002
8. Vaidya A, Hamrahian A, Bancos I, et al. The evaluation of incidentally discovered adrenal masses. Endocr Pract. 2019;25:178-192. doi: 10.4158/DSCR-2018-0565
9. Young WF Jr. Conventional imaging in adrenocortical carcinoma: update and perspectives. Horm Cancer. 2011;2:341-347. doi: 10.1007/s12672-011-0089-z
10. Neumann HPH, Young WF Jr, Eng C. Pheochromocytoma and paraganglioma. N Engl J Med. 2019;381:552-565. doi: 10.1056/NEJMra1806651
11. Iñiguez-Ariza NM, Kohlenberg JD, Delivanis DA, et al. Clinical, biochemical, and radiological characteristics of a single-center retrospective cohort of 705 large adrenal tumors. Mayo Clin Proc Innov Qual Outcomes. 2017;2:30-39. doi: 10.1016/j.mayocpiqo.2017.11.002
12. Marty M, Gaye D, Perez P, et al. Diagnostic accuracy of computed tomography to identify adenomas among adrenal incidentalomas in an endocrinological population. Eur J Endocrinol. 2018;178:439-446. doi: 10.1530/EJE-17-1056
13. Pacak K. Preoperative management of the pheochromocytoma patient. J Clin Endocrinol Metab. 2007;92:4069-4079. doi: 10.1210/jc.2007-1720
THE CASE
A 64-year-old woman sought care after having hot flashes, facial flushing, excessive sweating, palpitations, and daily headaches for 1 month. She had a history of hypertension that was well controlled with hydrochlorothiazide 25 mg/d but over the previous month, it had become more difficult to control. Her blood pressure remained elevated to 150/100 mm Hg despite the addition of lisinopril 40 mg/d and amlodipine 10 mg/d, indicating resistant hypertension. She had no family history of hypertension, diabetes, or obesity or any other pertinent medical or surgical history. Physical examination was negative for weight gain, stretch marks, or muscle weakness.

Laboratory tests revealed a normal serum aldosterone-renin ratio, renal function, and thyroid function; however, she had elevated levels of normetanephrine (2429 pg/mL; normal range, 0-145 pg/mL) and metanephrine (143 pg/mL; normal range, 0-62 pg/mL). Computed tomography (CT) revealed an 8.6-cm complex, hemorrhagic, necrotic left adrenal mass with attenuation of 33.1 Hounsfield units (HU) (FIGURE 1). Magnetic resonance imaging (MRI) demonstrated a T2 hyperintense left adrenal mass. An evaluation for Cushing syndrome was negative, and positron emission tomography (PET)/CT with gallium-68 dotatate was ordered. It showed intense radiotracer uptake in the left adrenal gland, with a maximum standardized uptake value of 70.1 (FIGURE 2).

THE DIAGNOSIS
After appropriate preparation with alpha blockade (phenoxybenzamine 20 mg twice daily for 7 days) and fluid resuscitation (normal saline run over 12 hours preoperatively), the patient underwent successful open surgical resection of the adrenal mass, during which her blood pressure was controlled with a nitroprusside infusion and boluses of esmolol and labetalol. Pathology results showed cells in a nested pattern with round to oval nuclei in a vascular background. There was no necrosis, increased mitotic figures, capsular invasion, or increased cellularity. Chromogranin immunohistochemical staining was positive. Given her resistant hypertension, clinical symptoms, and pathology results, the patient was given a diagnosis of pheochromocytoma.
DISCUSSION
Resistant hypertension is defined as blood pressure that is elevated above goal despite the use of 3 maximally titrated antihypertensive agents from different classes or that is well controlled with at least 4 antihypertensive medications.1 The prevalence of resistant hypertension is 12% to 18% in adults being treated for hypertension.1 Patients with resistant hypertension have a higher risk for cardiovascular events and death, are more likely to have a secondary cause of hypertension, and may benefit from special diagnostic testing or treatment approaches to control their blood pressure.1
There are many causes of resistant hypertension; primary aldosteronism is the most common cause (prevalence as high as 20%).2 Given the increased risk for cardiovascular/cerebrovascular disease, all patients with resistant hypertension should be screened for this condition.2 Other causes of resistant hypertension include renal parenchymal disease, renal artery stenosis, coarctation of the aorta, thyroid dysfunction, Cushing syndrome, paraganglioma, and as seen in our case, pheochromocytoma. Although pheochromocytoma is a rare cause of resistant hypertension (0.01%-4%),1 it is associated with high rates of morbidity and mortality if left untreated and may be inherited, making it an essential diagnosis to consider in all patients with resistant hypertension.1,3
Common symptoms of pheochromocytoma are hypertension (paroxysmal or sustained), headaches, palpitations, pallor, and piloerection (or cold sweats).1 Patients with pheochromocytoma typically exhibit metanephrine levels that are more than 4 times the upper limit of normal.4 Therefore, measurement of plasma free metanephrines or urinary fractionated metanephrines is recommended.5 Elevated metanephrine levels also are caused by obesity, obstructive sleep apnea, and certain medications and should be ruled out.5
All pheochromocytomas are potentially malignant. Despite the existence of pathologic scoring systems6,7 and radiographic features that suggest malignancy,8,9 no single risk-stratification tool is recommended in the current literature.10 Ultimately, the only way to confirm malignancy is to see metastases where chromaffin tissue is not normally found on imaging.10
Continue to: Pathologic features to look for...
Pathologic features to look for include capsular/periadrenal adipose invasion, increased cellularity, necrosis, tumor cell spindling, increased/atypical mitotic figures, and nuclear pleomorphism. Radiographic features include larger size (≥ 4-6 cm),11 an irregular shape, necrosis, calcifications, attenuation of 10 HU or higher on noncontrast CT, absolute washout of 60% or lower, and relative washout of 40% or lower.8,12 On MRI, malignant lesions appear hypointense on T1-weighted imaging and hyperintense on T2-weighted imaging.9 Fluorodeoxyglucose avidity on PET scan also is indicative of malignancy.8,9
Treatment for pheochromocytoma is surgical resection. An experienced surgical team and proper preoperative preparation are necessary because the induction of anesthesia, endotracheal intubation, and tumor manipulation can lead to a release of catecholamines, potentially resulting in an intraoperative hypertensive crisis, cardiac arrhythmias, and multiorgan failure.
Proper preoperative preparation includes taking an alpha-adrenergic blocker, such as phenoxybenzamine, prazosin, terazosin, or doxazosin, for at least 7 days to normalize the patient’s blood pressure. Patients should be counseled that they may experience nasal congestion, orthostasis, and fatigue while taking these medications. Volume expansion with intravenous fluids also should be performed and a high-salt diet considered. Beta-adrenergic blockade can be initiated once appropriate alpha-adrenergic blockade is achieved to control the patient’s heart rate; beta-blockers should never be started first because of the risk for severe hypertension. Careful hemodynamic monitoring is vital intraoperatively and postoperatively.5,13 Because metastatic lesions can occur decades after resection, long-term follow-up is critical.5,10
Following tumor resection, our patient’s blood pressure was supported with intravenous fluids and phenylephrine. She was able to discontinue all her antihypertensive medications postoperatively, and her plasma free and urinary fractionated metanephrine levels returned to within normal limits 8 weeks after surgery. Five years after surgery, she continues to have no signs of recurrence, as evidenced by annual negative plasma free metanephrines testing and abdominal/pelvic CT.
THE TAKEAWAY
This case highlights the importance of recognizing resistant hypertension and a potential secondary cause of this disease—pheochromocytoma. Although rare, pheochromocytomas confer increased risk for cardiovascular disease and death. Thus, swift recognition and proper preparation for surgical resection are necessary. Malignant lesions can be diagnosed only upon discovery of metastatic disease and can recur for decades after surgical resection, making diligent long-term follow-up imperative.
CORRESPONDENCE
Nicole O. Vietor, MD, Division of Endocrinology, Walter Reed National Military Medical Center, 8901 Wisconsin Avenue, Bethesda, MD 20889; [email protected]
THE CASE
A 64-year-old woman sought care after having hot flashes, facial flushing, excessive sweating, palpitations, and daily headaches for 1 month. She had a history of hypertension that was well controlled with hydrochlorothiazide 25 mg/d but over the previous month, it had become more difficult to control. Her blood pressure remained elevated to 150/100 mm Hg despite the addition of lisinopril 40 mg/d and amlodipine 10 mg/d, indicating resistant hypertension. She had no family history of hypertension, diabetes, or obesity or any other pertinent medical or surgical history. Physical examination was negative for weight gain, stretch marks, or muscle weakness.

Laboratory tests revealed a normal serum aldosterone-renin ratio, renal function, and thyroid function; however, she had elevated levels of normetanephrine (2429 pg/mL; normal range, 0-145 pg/mL) and metanephrine (143 pg/mL; normal range, 0-62 pg/mL). Computed tomography (CT) revealed an 8.6-cm complex, hemorrhagic, necrotic left adrenal mass with attenuation of 33.1 Hounsfield units (HU) (FIGURE 1). Magnetic resonance imaging (MRI) demonstrated a T2 hyperintense left adrenal mass. An evaluation for Cushing syndrome was negative, and positron emission tomography (PET)/CT with gallium-68 dotatate was ordered. It showed intense radiotracer uptake in the left adrenal gland, with a maximum standardized uptake value of 70.1 (FIGURE 2).

THE DIAGNOSIS
After appropriate preparation with alpha blockade (phenoxybenzamine 20 mg twice daily for 7 days) and fluid resuscitation (normal saline run over 12 hours preoperatively), the patient underwent successful open surgical resection of the adrenal mass, during which her blood pressure was controlled with a nitroprusside infusion and boluses of esmolol and labetalol. Pathology results showed cells in a nested pattern with round to oval nuclei in a vascular background. There was no necrosis, increased mitotic figures, capsular invasion, or increased cellularity. Chromogranin immunohistochemical staining was positive. Given her resistant hypertension, clinical symptoms, and pathology results, the patient was given a diagnosis of pheochromocytoma.
DISCUSSION
Resistant hypertension is defined as blood pressure that is elevated above goal despite the use of 3 maximally titrated antihypertensive agents from different classes or that is well controlled with at least 4 antihypertensive medications.1 The prevalence of resistant hypertension is 12% to 18% in adults being treated for hypertension.1 Patients with resistant hypertension have a higher risk for cardiovascular events and death, are more likely to have a secondary cause of hypertension, and may benefit from special diagnostic testing or treatment approaches to control their blood pressure.1
There are many causes of resistant hypertension; primary aldosteronism is the most common cause (prevalence as high as 20%).2 Given the increased risk for cardiovascular/cerebrovascular disease, all patients with resistant hypertension should be screened for this condition.2 Other causes of resistant hypertension include renal parenchymal disease, renal artery stenosis, coarctation of the aorta, thyroid dysfunction, Cushing syndrome, paraganglioma, and as seen in our case, pheochromocytoma. Although pheochromocytoma is a rare cause of resistant hypertension (0.01%-4%),1 it is associated with high rates of morbidity and mortality if left untreated and may be inherited, making it an essential diagnosis to consider in all patients with resistant hypertension.1,3
Common symptoms of pheochromocytoma are hypertension (paroxysmal or sustained), headaches, palpitations, pallor, and piloerection (or cold sweats).1 Patients with pheochromocytoma typically exhibit metanephrine levels that are more than 4 times the upper limit of normal.4 Therefore, measurement of plasma free metanephrines or urinary fractionated metanephrines is recommended.5 Elevated metanephrine levels also are caused by obesity, obstructive sleep apnea, and certain medications and should be ruled out.5
All pheochromocytomas are potentially malignant. Despite the existence of pathologic scoring systems6,7 and radiographic features that suggest malignancy,8,9 no single risk-stratification tool is recommended in the current literature.10 Ultimately, the only way to confirm malignancy is to see metastases where chromaffin tissue is not normally found on imaging.10
Continue to: Pathologic features to look for...
Pathologic features to look for include capsular/periadrenal adipose invasion, increased cellularity, necrosis, tumor cell spindling, increased/atypical mitotic figures, and nuclear pleomorphism. Radiographic features include larger size (≥ 4-6 cm),11 an irregular shape, necrosis, calcifications, attenuation of 10 HU or higher on noncontrast CT, absolute washout of 60% or lower, and relative washout of 40% or lower.8,12 On MRI, malignant lesions appear hypointense on T1-weighted imaging and hyperintense on T2-weighted imaging.9 Fluorodeoxyglucose avidity on PET scan also is indicative of malignancy.8,9
Treatment for pheochromocytoma is surgical resection. An experienced surgical team and proper preoperative preparation are necessary because the induction of anesthesia, endotracheal intubation, and tumor manipulation can lead to a release of catecholamines, potentially resulting in an intraoperative hypertensive crisis, cardiac arrhythmias, and multiorgan failure.
Proper preoperative preparation includes taking an alpha-adrenergic blocker, such as phenoxybenzamine, prazosin, terazosin, or doxazosin, for at least 7 days to normalize the patient’s blood pressure. Patients should be counseled that they may experience nasal congestion, orthostasis, and fatigue while taking these medications. Volume expansion with intravenous fluids also should be performed and a high-salt diet considered. Beta-adrenergic blockade can be initiated once appropriate alpha-adrenergic blockade is achieved to control the patient’s heart rate; beta-blockers should never be started first because of the risk for severe hypertension. Careful hemodynamic monitoring is vital intraoperatively and postoperatively.5,13 Because metastatic lesions can occur decades after resection, long-term follow-up is critical.5,10
Following tumor resection, our patient’s blood pressure was supported with intravenous fluids and phenylephrine. She was able to discontinue all her antihypertensive medications postoperatively, and her plasma free and urinary fractionated metanephrine levels returned to within normal limits 8 weeks after surgery. Five years after surgery, she continues to have no signs of recurrence, as evidenced by annual negative plasma free metanephrines testing and abdominal/pelvic CT.
THE TAKEAWAY
This case highlights the importance of recognizing resistant hypertension and a potential secondary cause of this disease—pheochromocytoma. Although rare, pheochromocytomas confer increased risk for cardiovascular disease and death. Thus, swift recognition and proper preparation for surgical resection are necessary. Malignant lesions can be diagnosed only upon discovery of metastatic disease and can recur for decades after surgical resection, making diligent long-term follow-up imperative.
CORRESPONDENCE
Nicole O. Vietor, MD, Division of Endocrinology, Walter Reed National Military Medical Center, 8901 Wisconsin Avenue, Bethesda, MD 20889; [email protected]
1. Carey RM, Calhoun DA, Bakris GL, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72:e53-e90. doi: 10.1161/HYP.0000000000000084
2. Young WF Jr. Diagnosis and treatment of primary aldosteronism: practical clinical perspectives. J Intern Med. 2019;285:126-148. doi: 10.1111/joim.12831
3. Young WF Jr, Calhoun DA, Lenders JWM, et al. Screening for endocrine hypertension: an Endocrine Society Scientific Statement. Endocr Rev. 2017;38:103-122. doi: 10.1210/er.2017-00054
4. Lenders JWM, Pacak K, Walther MM, et al. Biochemical diagnosis of pheochromocytoma: which test is best? JAMA. 2002;287:1427-1434. doi: 10.1001/jama.287.11.1427
5. Lenders JW, Duh Q-Y, Eisenhofer G, et al. Pheochromocytoma and paraganglioma: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2014;99:1915-1942. doi: 10.1210/jc.2014-1498
6. Kimura N, Takayanagi R, Takizawa N, et al. Pathological grading for predicting metastasis in phaeochromocytoma and paraganglioma. Endocr Relat Cancer. 2014;21:405-414. doi: 10.1530/ERC-13-0494
7. Thompson LDR. Pheochromocytoma of the Adrenal gland Scaled Score (PASS) to separate benign from malignant neoplasms: a clinicopathologic and immunophenotypic study of 100 cases. Am J Surg Pathol. 2002;26:551-566. doi: 10.1097/00000478-200205000-00002
8. Vaidya A, Hamrahian A, Bancos I, et al. The evaluation of incidentally discovered adrenal masses. Endocr Pract. 2019;25:178-192. doi: 10.4158/DSCR-2018-0565
9. Young WF Jr. Conventional imaging in adrenocortical carcinoma: update and perspectives. Horm Cancer. 2011;2:341-347. doi: 10.1007/s12672-011-0089-z
10. Neumann HPH, Young WF Jr, Eng C. Pheochromocytoma and paraganglioma. N Engl J Med. 2019;381:552-565. doi: 10.1056/NEJMra1806651
11. Iñiguez-Ariza NM, Kohlenberg JD, Delivanis DA, et al. Clinical, biochemical, and radiological characteristics of a single-center retrospective cohort of 705 large adrenal tumors. Mayo Clin Proc Innov Qual Outcomes. 2017;2:30-39. doi: 10.1016/j.mayocpiqo.2017.11.002
12. Marty M, Gaye D, Perez P, et al. Diagnostic accuracy of computed tomography to identify adenomas among adrenal incidentalomas in an endocrinological population. Eur J Endocrinol. 2018;178:439-446. doi: 10.1530/EJE-17-1056
13. Pacak K. Preoperative management of the pheochromocytoma patient. J Clin Endocrinol Metab. 2007;92:4069-4079. doi: 10.1210/jc.2007-1720
1. Carey RM, Calhoun DA, Bakris GL, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72:e53-e90. doi: 10.1161/HYP.0000000000000084
2. Young WF Jr. Diagnosis and treatment of primary aldosteronism: practical clinical perspectives. J Intern Med. 2019;285:126-148. doi: 10.1111/joim.12831
3. Young WF Jr, Calhoun DA, Lenders JWM, et al. Screening for endocrine hypertension: an Endocrine Society Scientific Statement. Endocr Rev. 2017;38:103-122. doi: 10.1210/er.2017-00054
4. Lenders JWM, Pacak K, Walther MM, et al. Biochemical diagnosis of pheochromocytoma: which test is best? JAMA. 2002;287:1427-1434. doi: 10.1001/jama.287.11.1427
5. Lenders JW, Duh Q-Y, Eisenhofer G, et al. Pheochromocytoma and paraganglioma: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2014;99:1915-1942. doi: 10.1210/jc.2014-1498
6. Kimura N, Takayanagi R, Takizawa N, et al. Pathological grading for predicting metastasis in phaeochromocytoma and paraganglioma. Endocr Relat Cancer. 2014;21:405-414. doi: 10.1530/ERC-13-0494
7. Thompson LDR. Pheochromocytoma of the Adrenal gland Scaled Score (PASS) to separate benign from malignant neoplasms: a clinicopathologic and immunophenotypic study of 100 cases. Am J Surg Pathol. 2002;26:551-566. doi: 10.1097/00000478-200205000-00002
8. Vaidya A, Hamrahian A, Bancos I, et al. The evaluation of incidentally discovered adrenal masses. Endocr Pract. 2019;25:178-192. doi: 10.4158/DSCR-2018-0565
9. Young WF Jr. Conventional imaging in adrenocortical carcinoma: update and perspectives. Horm Cancer. 2011;2:341-347. doi: 10.1007/s12672-011-0089-z
10. Neumann HPH, Young WF Jr, Eng C. Pheochromocytoma and paraganglioma. N Engl J Med. 2019;381:552-565. doi: 10.1056/NEJMra1806651
11. Iñiguez-Ariza NM, Kohlenberg JD, Delivanis DA, et al. Clinical, biochemical, and radiological characteristics of a single-center retrospective cohort of 705 large adrenal tumors. Mayo Clin Proc Innov Qual Outcomes. 2017;2:30-39. doi: 10.1016/j.mayocpiqo.2017.11.002
12. Marty M, Gaye D, Perez P, et al. Diagnostic accuracy of computed tomography to identify adenomas among adrenal incidentalomas in an endocrinological population. Eur J Endocrinol. 2018;178:439-446. doi: 10.1530/EJE-17-1056
13. Pacak K. Preoperative management of the pheochromocytoma patient. J Clin Endocrinol Metab. 2007;92:4069-4079. doi: 10.1210/jc.2007-1720
► Hot flashes, facial flushing, excessive sweating, and palpitations
► Daily headaches
► History of hypertension
Acute Achilles tendon rupture: Skip the surgery?
ILLUSTRATIVE CASE
An otherwise healthy 45-year-old man sustained an acute right-side Achilles tendon rupture while playing tennis. He has not taken quinolones recently, has no history of previous Achilles tendon rupture, and prior to this injury had no difficulty walking. He presents initially to his primary care physician and wants advice: Does he need surgery?
Acute Achilles tendon rupture manifests as acute-onset pain and impaired plantar flexion.2 Older, active, male patients are at increased risk. There is disagreement among treating physicians regarding best practices for managing this common and debilitating injury. Prior clinical trials comparing operative to nonoperative management, as well as those comparing different surgical techniques, were limited by small sample sizes.3-5
A 2019 systematic review and meta-analysis that relied heavily on observational data suggested that nonoperative management carries greater risk for rerupture but lower risk for complications than surgical treatment, without differences in patient-reported functional outcomes.5 This 2022 RCT adds certainty to comparisons of surgical and nonoperative treatment.
STUDY SUMMARY
Equivalent outcomes but higher rates of rerupture for nonoperative patients
Norwegian investigators conducted a prospective, single-blind RCT at 4 treating facilities among patients ages 18 to 60 years with unilateral acute Achilles tendon rupture. A total of 554 patients were randomized in a 1:1:1 ratio to 1 of 3 groups: nonoperative treatment, open-repair surgery, or minimally invasive surgery. Ultimately, 526 patients who completed the intervention and at least 1 follow-up survey were included in the final analysis, which exceeded the number needed according to the pre-study 80% power calculation. Seventy-four percent of the patients were male, and the average age at time of injury was 40 years. Nearly all patients were classified as healthy or having only mild or well-controlled chronic illnesses.
Before randomization, patients completed the 10-item Achilles tendon Total Rupture Score (ATRS) questionnaire to gauge their pre-injury baseline function. ATRS is scored 0 to 100, with lower scores indicating more limitation in function; a clinically important difference is 8 to 10 points. There were no statistically significant differences in pre-injury baseline ATRS (92.7, 93.9, and 94.2 for the nonoperative, open-repair, and minimally invasive groups, respectively) or other patient characteristics among the 3 groups.
For all participants, application of a below-the-knee equinus cast with plantar flexion was performed within 72 hours after the injury. Patients in the surgical arms had surgery within 8 days, followed by application of a new cast. For all study groups, the cast was maintained for a total of 2 weeks, followed by 6 weeks of weight-bearing in an ankle-foot orthosis with heel wedges that were gradually reduced in number. All patients were treated with identical serial immobilization and physical therapy programs for 36 weeks.
The primary study outcome was change from baseline ATRS at 12 months after injury. Secondary outcomes included ATRS at 3 and 6 months and domain-specific quality-of-life scores (from the 36-Item Short Form Health Survey; SF-36) at 6 and 12 months. Patients also underwent physical testing of their Achilles tendon function at 6 and 12 months, during which they wore knee-high socks in order to blind the evaluators. Reruptures were recorded as secondary outcomes as well.
Continue to: There were no significant...
There were no significant differences between groups in the primary outcome. The mean changes in ATRS were −2.6 points (95% CI, −6.5 to 2.0) for nonoperative treatment compared with minimally invasive surgery, and 1.0 point (95% CI, −5.2 to 3.1) for nonoperative treatment compared with open repair.
All groups had similar secondary self-reported ATRS at 3 and 6 months and SF-36 scores at 6 and 12 months. Blinded physical test results also were similar between groups at 6 and 12 months.
Tendon rerupture within 12 months was more common in the nonoperative arm than in the 2 surgical arms (6.2% vs 0.6% in both operative groups; 5.6% difference; 95% CI for difference, 1.9-10.2 for open repair and 1.8-10.2 for minimally invasive surgery). Risk for nerve injury was higher in both the minimally invasive surgery group (5.2%) and the open-repair surgery group (2.8%) compared with the nonoperative group (0.6%; no P value given for comparison).
WHAT’S NEW
Largest RCT to date showed effectiveness of nonoperative Tx
This study is the largest well-powered and rigorously conducted RCT to show that nonoperative management of acute Achilles tendon rupture offers equivalent patient-reported outcomes at 12 months after injury. Nonoperative management was associated with a lower risk for nerve injury but higher risk for tendon rerupture.
These findings support previous studies on the topic. As previously mentioned, a 2019 systematic review and meta-analysis of 10 RCTs (N = 944) and 19 observational studies (N = 14,918) examined operative compared with nonoperative treatment of acute Achilles tendon rupture and found a lower rerupture rate in the operative group but a higher complication rate.5 An underpowered 2010 RCT (N = 97) of operative vs nonoperative treatment of acute Achilles tendon rupture found no statistical difference in ATRS.3 Another underpowered RCT conducted in 2013 (N = 100) compared surgical treatment, accelerated rehabilitation, and nonsurgical treatment in acute Achilles tendon rupture and found no statistical difference in ATRS.4
CAVEATS
Study results may not apply to some patient groups
These findings may not apply to patients older than 60 years, who were excluded from this RCT, or patients with debilitation or significant chronic disease. Patients with prior Achilles rupture also were excluded.
The study population in Norway, which is more physically active than nearby countries, may not be generalizable worldwide.6 Patients wishing to minimize the risk for rerupture may still prefer to have surgery after acute Achilles tendon rupture.
CHALLENGES TO IMPLEMENTATION
Potentially limited options for patients
Most patients with acute Achilles tendon rupture are evaluated by orthopedic surgeons, who may or may not offer nonoperative management. Availability of practitioners to provide serial casting, appropriate heel wedges, and rehabilitation may vary regionally. All patients in this study were evaluated within 72 hours of injury; these findings may not be applicable for patients at a longer time since injury.
1. Myhrvold SB, Brouwer EF, Andresen TKM, et al. Nonoperative or surgical treatment of acute Achilles’ tendon rupture. N Engl J Med. 2022;386:1409-1420. doi: 10.1056/NEJMoa2108447
2. Huttunen TT, Kannus P, Rolf C, et al. Acute achilles tendon ruptures: incidence of injury and surgery in Sweden between 2001 and 2012. Am J Sports Med. 2014;42:2419-2423. doi: 10.1177/0363546514540599
3. Nilsson-Helander K, Silbernagel KG, Thomeé R, et al. Acute achilles tendon rupture: a randomized, controlled study comparing surgical and nonsurgical treatments using validated outcome measures. Am J Sports Med. 2010;38:2186-2193. doi: 10.1177/0363546510376052
4. Olsson N, Silbernagel KG, Eriksson BI, et al. Stable surgical repair with accelerated rehabilitation versus nonsurgical treatment for acute Achilles tendon ruptures: a randomized controlled study. Am J Sports Med. 2013;41:2867-2876. doi: 10.1177/0363546513503282
5. Ochen Y, Beks RB, van Heijl M, et al. Operative treatment versus nonoperative treatment of Achilles tendon ruptures: systematic review and meta-analysis. BMJ. 2019;364:k5120. doi: 10.1136/bmj.k5120
6. Urbaniak-Brekke AM, Pluta B, Krzykała M, et al. Physical activity of Polish and Norwegian local communities in the context of self-government authorities’ projects. Int J Environ Res Public Health. 2019;16:1710. doi: 10.3390/ijerph16101710
ILLUSTRATIVE CASE
An otherwise healthy 45-year-old man sustained an acute right-side Achilles tendon rupture while playing tennis. He has not taken quinolones recently, has no history of previous Achilles tendon rupture, and prior to this injury had no difficulty walking. He presents initially to his primary care physician and wants advice: Does he need surgery?
Acute Achilles tendon rupture manifests as acute-onset pain and impaired plantar flexion.2 Older, active, male patients are at increased risk. There is disagreement among treating physicians regarding best practices for managing this common and debilitating injury. Prior clinical trials comparing operative to nonoperative management, as well as those comparing different surgical techniques, were limited by small sample sizes.3-5
A 2019 systematic review and meta-analysis that relied heavily on observational data suggested that nonoperative management carries greater risk for rerupture but lower risk for complications than surgical treatment, without differences in patient-reported functional outcomes.5 This 2022 RCT adds certainty to comparisons of surgical and nonoperative treatment.
STUDY SUMMARY
Equivalent outcomes but higher rates of rerupture for nonoperative patients
Norwegian investigators conducted a prospective, single-blind RCT at 4 treating facilities among patients ages 18 to 60 years with unilateral acute Achilles tendon rupture. A total of 554 patients were randomized in a 1:1:1 ratio to 1 of 3 groups: nonoperative treatment, open-repair surgery, or minimally invasive surgery. Ultimately, 526 patients who completed the intervention and at least 1 follow-up survey were included in the final analysis, which exceeded the number needed according to the pre-study 80% power calculation. Seventy-four percent of the patients were male, and the average age at time of injury was 40 years. Nearly all patients were classified as healthy or having only mild or well-controlled chronic illnesses.
Before randomization, patients completed the 10-item Achilles tendon Total Rupture Score (ATRS) questionnaire to gauge their pre-injury baseline function. ATRS is scored 0 to 100, with lower scores indicating more limitation in function; a clinically important difference is 8 to 10 points. There were no statistically significant differences in pre-injury baseline ATRS (92.7, 93.9, and 94.2 for the nonoperative, open-repair, and minimally invasive groups, respectively) or other patient characteristics among the 3 groups.
For all participants, application of a below-the-knee equinus cast with plantar flexion was performed within 72 hours after the injury. Patients in the surgical arms had surgery within 8 days, followed by application of a new cast. For all study groups, the cast was maintained for a total of 2 weeks, followed by 6 weeks of weight-bearing in an ankle-foot orthosis with heel wedges that were gradually reduced in number. All patients were treated with identical serial immobilization and physical therapy programs for 36 weeks.
The primary study outcome was change from baseline ATRS at 12 months after injury. Secondary outcomes included ATRS at 3 and 6 months and domain-specific quality-of-life scores (from the 36-Item Short Form Health Survey; SF-36) at 6 and 12 months. Patients also underwent physical testing of their Achilles tendon function at 6 and 12 months, during which they wore knee-high socks in order to blind the evaluators. Reruptures were recorded as secondary outcomes as well.
Continue to: There were no significant...
There were no significant differences between groups in the primary outcome. The mean changes in ATRS were −2.6 points (95% CI, −6.5 to 2.0) for nonoperative treatment compared with minimally invasive surgery, and 1.0 point (95% CI, −5.2 to 3.1) for nonoperative treatment compared with open repair.
All groups had similar secondary self-reported ATRS at 3 and 6 months and SF-36 scores at 6 and 12 months. Blinded physical test results also were similar between groups at 6 and 12 months.
Tendon rerupture within 12 months was more common in the nonoperative arm than in the 2 surgical arms (6.2% vs 0.6% in both operative groups; 5.6% difference; 95% CI for difference, 1.9-10.2 for open repair and 1.8-10.2 for minimally invasive surgery). Risk for nerve injury was higher in both the minimally invasive surgery group (5.2%) and the open-repair surgery group (2.8%) compared with the nonoperative group (0.6%; no P value given for comparison).
WHAT’S NEW
Largest RCT to date showed effectiveness of nonoperative Tx
This study is the largest well-powered and rigorously conducted RCT to show that nonoperative management of acute Achilles tendon rupture offers equivalent patient-reported outcomes at 12 months after injury. Nonoperative management was associated with a lower risk for nerve injury but higher risk for tendon rerupture.
These findings support previous studies on the topic. As previously mentioned, a 2019 systematic review and meta-analysis of 10 RCTs (N = 944) and 19 observational studies (N = 14,918) examined operative compared with nonoperative treatment of acute Achilles tendon rupture and found a lower rerupture rate in the operative group but a higher complication rate.5 An underpowered 2010 RCT (N = 97) of operative vs nonoperative treatment of acute Achilles tendon rupture found no statistical difference in ATRS.3 Another underpowered RCT conducted in 2013 (N = 100) compared surgical treatment, accelerated rehabilitation, and nonsurgical treatment in acute Achilles tendon rupture and found no statistical difference in ATRS.4
CAVEATS
Study results may not apply to some patient groups
These findings may not apply to patients older than 60 years, who were excluded from this RCT, or patients with debilitation or significant chronic disease. Patients with prior Achilles rupture also were excluded.
The study population in Norway, which is more physically active than nearby countries, may not be generalizable worldwide.6 Patients wishing to minimize the risk for rerupture may still prefer to have surgery after acute Achilles tendon rupture.
CHALLENGES TO IMPLEMENTATION
Potentially limited options for patients
Most patients with acute Achilles tendon rupture are evaluated by orthopedic surgeons, who may or may not offer nonoperative management. Availability of practitioners to provide serial casting, appropriate heel wedges, and rehabilitation may vary regionally. All patients in this study were evaluated within 72 hours of injury; these findings may not be applicable for patients at a longer time since injury.
ILLUSTRATIVE CASE
An otherwise healthy 45-year-old man sustained an acute right-side Achilles tendon rupture while playing tennis. He has not taken quinolones recently, has no history of previous Achilles tendon rupture, and prior to this injury had no difficulty walking. He presents initially to his primary care physician and wants advice: Does he need surgery?
Acute Achilles tendon rupture manifests as acute-onset pain and impaired plantar flexion.2 Older, active, male patients are at increased risk. There is disagreement among treating physicians regarding best practices for managing this common and debilitating injury. Prior clinical trials comparing operative to nonoperative management, as well as those comparing different surgical techniques, were limited by small sample sizes.3-5
A 2019 systematic review and meta-analysis that relied heavily on observational data suggested that nonoperative management carries greater risk for rerupture but lower risk for complications than surgical treatment, without differences in patient-reported functional outcomes.5 This 2022 RCT adds certainty to comparisons of surgical and nonoperative treatment.
STUDY SUMMARY
Equivalent outcomes but higher rates of rerupture for nonoperative patients
Norwegian investigators conducted a prospective, single-blind RCT at 4 treating facilities among patients ages 18 to 60 years with unilateral acute Achilles tendon rupture. A total of 554 patients were randomized in a 1:1:1 ratio to 1 of 3 groups: nonoperative treatment, open-repair surgery, or minimally invasive surgery. Ultimately, 526 patients who completed the intervention and at least 1 follow-up survey were included in the final analysis, which exceeded the number needed according to the pre-study 80% power calculation. Seventy-four percent of the patients were male, and the average age at time of injury was 40 years. Nearly all patients were classified as healthy or having only mild or well-controlled chronic illnesses.
Before randomization, patients completed the 10-item Achilles tendon Total Rupture Score (ATRS) questionnaire to gauge their pre-injury baseline function. ATRS is scored 0 to 100, with lower scores indicating more limitation in function; a clinically important difference is 8 to 10 points. There were no statistically significant differences in pre-injury baseline ATRS (92.7, 93.9, and 94.2 for the nonoperative, open-repair, and minimally invasive groups, respectively) or other patient characteristics among the 3 groups.
For all participants, application of a below-the-knee equinus cast with plantar flexion was performed within 72 hours after the injury. Patients in the surgical arms had surgery within 8 days, followed by application of a new cast. For all study groups, the cast was maintained for a total of 2 weeks, followed by 6 weeks of weight-bearing in an ankle-foot orthosis with heel wedges that were gradually reduced in number. All patients were treated with identical serial immobilization and physical therapy programs for 36 weeks.
The primary study outcome was change from baseline ATRS at 12 months after injury. Secondary outcomes included ATRS at 3 and 6 months and domain-specific quality-of-life scores (from the 36-Item Short Form Health Survey; SF-36) at 6 and 12 months. Patients also underwent physical testing of their Achilles tendon function at 6 and 12 months, during which they wore knee-high socks in order to blind the evaluators. Reruptures were recorded as secondary outcomes as well.
Continue to: There were no significant...
There were no significant differences between groups in the primary outcome. The mean changes in ATRS were −2.6 points (95% CI, −6.5 to 2.0) for nonoperative treatment compared with minimally invasive surgery, and 1.0 point (95% CI, −5.2 to 3.1) for nonoperative treatment compared with open repair.
All groups had similar secondary self-reported ATRS at 3 and 6 months and SF-36 scores at 6 and 12 months. Blinded physical test results also were similar between groups at 6 and 12 months.
Tendon rerupture within 12 months was more common in the nonoperative arm than in the 2 surgical arms (6.2% vs 0.6% in both operative groups; 5.6% difference; 95% CI for difference, 1.9-10.2 for open repair and 1.8-10.2 for minimally invasive surgery). Risk for nerve injury was higher in both the minimally invasive surgery group (5.2%) and the open-repair surgery group (2.8%) compared with the nonoperative group (0.6%; no P value given for comparison).
WHAT’S NEW
Largest RCT to date showed effectiveness of nonoperative Tx
This study is the largest well-powered and rigorously conducted RCT to show that nonoperative management of acute Achilles tendon rupture offers equivalent patient-reported outcomes at 12 months after injury. Nonoperative management was associated with a lower risk for nerve injury but higher risk for tendon rerupture.
These findings support previous studies on the topic. As previously mentioned, a 2019 systematic review and meta-analysis of 10 RCTs (N = 944) and 19 observational studies (N = 14,918) examined operative compared with nonoperative treatment of acute Achilles tendon rupture and found a lower rerupture rate in the operative group but a higher complication rate.5 An underpowered 2010 RCT (N = 97) of operative vs nonoperative treatment of acute Achilles tendon rupture found no statistical difference in ATRS.3 Another underpowered RCT conducted in 2013 (N = 100) compared surgical treatment, accelerated rehabilitation, and nonsurgical treatment in acute Achilles tendon rupture and found no statistical difference in ATRS.4
CAVEATS
Study results may not apply to some patient groups
These findings may not apply to patients older than 60 years, who were excluded from this RCT, or patients with debilitation or significant chronic disease. Patients with prior Achilles rupture also were excluded.
The study population in Norway, which is more physically active than nearby countries, may not be generalizable worldwide.6 Patients wishing to minimize the risk for rerupture may still prefer to have surgery after acute Achilles tendon rupture.
CHALLENGES TO IMPLEMENTATION
Potentially limited options for patients
Most patients with acute Achilles tendon rupture are evaluated by orthopedic surgeons, who may or may not offer nonoperative management. Availability of practitioners to provide serial casting, appropriate heel wedges, and rehabilitation may vary regionally. All patients in this study were evaluated within 72 hours of injury; these findings may not be applicable for patients at a longer time since injury.
1. Myhrvold SB, Brouwer EF, Andresen TKM, et al. Nonoperative or surgical treatment of acute Achilles’ tendon rupture. N Engl J Med. 2022;386:1409-1420. doi: 10.1056/NEJMoa2108447
2. Huttunen TT, Kannus P, Rolf C, et al. Acute achilles tendon ruptures: incidence of injury and surgery in Sweden between 2001 and 2012. Am J Sports Med. 2014;42:2419-2423. doi: 10.1177/0363546514540599
3. Nilsson-Helander K, Silbernagel KG, Thomeé R, et al. Acute achilles tendon rupture: a randomized, controlled study comparing surgical and nonsurgical treatments using validated outcome measures. Am J Sports Med. 2010;38:2186-2193. doi: 10.1177/0363546510376052
4. Olsson N, Silbernagel KG, Eriksson BI, et al. Stable surgical repair with accelerated rehabilitation versus nonsurgical treatment for acute Achilles tendon ruptures: a randomized controlled study. Am J Sports Med. 2013;41:2867-2876. doi: 10.1177/0363546513503282
5. Ochen Y, Beks RB, van Heijl M, et al. Operative treatment versus nonoperative treatment of Achilles tendon ruptures: systematic review and meta-analysis. BMJ. 2019;364:k5120. doi: 10.1136/bmj.k5120
6. Urbaniak-Brekke AM, Pluta B, Krzykała M, et al. Physical activity of Polish and Norwegian local communities in the context of self-government authorities’ projects. Int J Environ Res Public Health. 2019;16:1710. doi: 10.3390/ijerph16101710
1. Myhrvold SB, Brouwer EF, Andresen TKM, et al. Nonoperative or surgical treatment of acute Achilles’ tendon rupture. N Engl J Med. 2022;386:1409-1420. doi: 10.1056/NEJMoa2108447
2. Huttunen TT, Kannus P, Rolf C, et al. Acute achilles tendon ruptures: incidence of injury and surgery in Sweden between 2001 and 2012. Am J Sports Med. 2014;42:2419-2423. doi: 10.1177/0363546514540599
3. Nilsson-Helander K, Silbernagel KG, Thomeé R, et al. Acute achilles tendon rupture: a randomized, controlled study comparing surgical and nonsurgical treatments using validated outcome measures. Am J Sports Med. 2010;38:2186-2193. doi: 10.1177/0363546510376052
4. Olsson N, Silbernagel KG, Eriksson BI, et al. Stable surgical repair with accelerated rehabilitation versus nonsurgical treatment for acute Achilles tendon ruptures: a randomized controlled study. Am J Sports Med. 2013;41:2867-2876. doi: 10.1177/0363546513503282
5. Ochen Y, Beks RB, van Heijl M, et al. Operative treatment versus nonoperative treatment of Achilles tendon ruptures: systematic review and meta-analysis. BMJ. 2019;364:k5120. doi: 10.1136/bmj.k5120
6. Urbaniak-Brekke AM, Pluta B, Krzykała M, et al. Physical activity of Polish and Norwegian local communities in the context of self-government authorities’ projects. Int J Environ Res Public Health. 2019;16:1710. doi: 10.3390/ijerph16101710
PRACTICE CHANGER
For healthy patients ages 18 to 60 years with acute Achilles tendon rupture, consider nonoperative immobilization, which offered a benefit in function comparable to open-repair or minimally invasive surgery in this randomized controlled trial (RCT).
STRENGTH OF RECOMMENDATION
B: Based on a single RCT.1
Myhrvold SB, Brouwer EF, Andresen TKM, et al. Nonoperative or surgical treatment of acute Achilles’ tendon rupture. N Engl J Med. 2022;386:1409-1420. doi: 10.1056/NEJMoa2108447
Caring for the caregiver in dementia
THE CASE
Sam C* is a 68-year-old man who presented to his family physician in a rural health clinic due to concerns about weight loss. Since his visit 8 months prior, Mr. C unintentionally had lost 20 pounds. Upon questioning, Mr. C also reported feeling irritable and having difficulty with sleep and concentration.
A review of systems did not indicate the presence of infection or other medical conditions. In the 6 years since becoming a patient to the practice, he had reported no chronic health concerns, was taking no medications, and had only been to the clinic for his annual check-up appointments. He completed a Patient Health Questionnaire (PHQ-9) and scored 18, indicating moderately severe depression.
Mr. C had established care with his physician when he moved to the area from out of state so that he could be closer to his parents, who were in their mid-80s at the time. Mr. C’s physician also had been the family physician for his parents for the previous 20 years. Three years prior to Mr. C’s presentation for weight loss, his mother had received a diagnosis of acute leukemia; she died a year later.
Over the past year, Mr. C had needed to take a more active role in the care of his father, who was now in his early 90s. Mr. C’s father, who was previously in excellent health, had begun to develop significant health problems, including degenerative arthritis and progressive vascular dementia. He also had ataxia, leading to poor mobility, and a neurogenic bladder requiring self-catheterization, which required Mr. C’s assistance. Mr. C lived next door to his father and provided frequent assistance with activities of daily living. However, his father, who always had been the dominant figure in the family, was determined to maintain his independence and not relinquish control to others.
The strain of caregiving activities, along with managing his father’s inflexibility, was causing increasing distress for Mr. C. As he told his family physician, “I just don’t know what to do.”
●
* The patient’s name has been changed to protect his identity.
It is estimated that more than 11 million Americans provided more than 18 billion hours in unpaid support for individuals with dementia in 2022, averaging 30 hours of care per caregiver per week.1 As individuals with dementia progressively decline, they require increased assistance with activities of daily living (ADLs, such as bathing and dressing) and instrumental activities of daily living (IADLs, such as paying bills and using transportation). Most of this assistance comes from informal caregiving provided by family members and friends.
Caregiver burden can be defined as “the strain or load borne by a person who cares for a chronically ill, disabled, or elderly family member.”2 Caregiver stress has been found to be higher for dementia caregiving than other types of caregiving.3 In particular, caring for someone with greater behavioral and psychological symptoms of dementia (BPSDs) has been associated with higher caregiver burden.4-
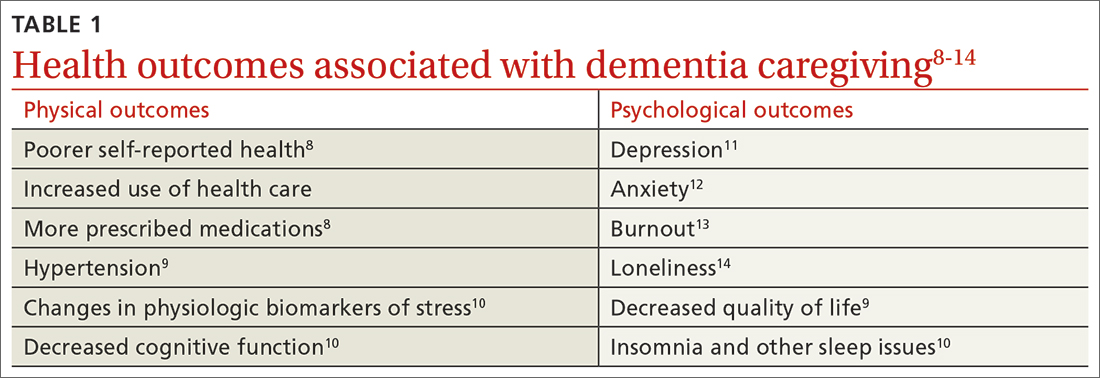
Beyond the subjective burden of caregiving, there are other potential negative consequences for dementia caregivers (see TABLE 18-14 and TABLE 215,16). In addition, caregiver distress is related to a number of care recipient outcomes, including earlier institutionalization, more hospitalizations, more BPSDs, poorer quality of life, and greater likelihood of experiencing elder abuse.17
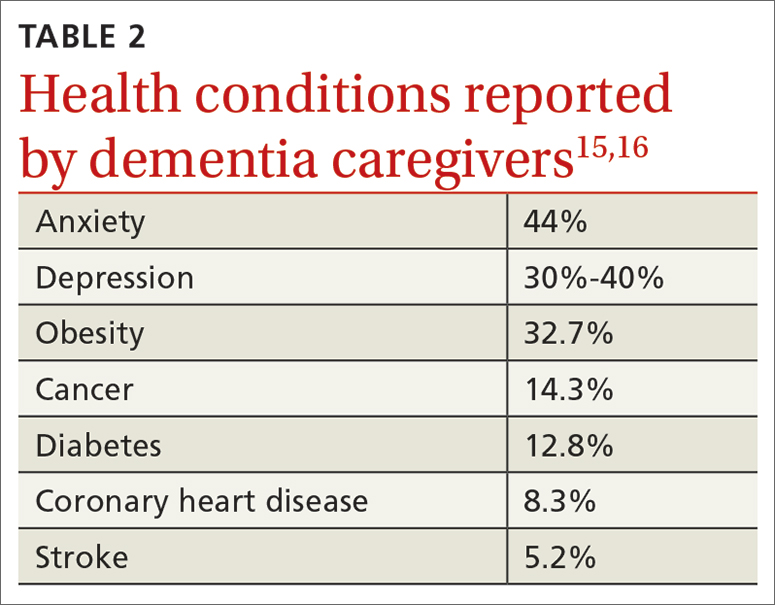
Assessment, reassessment are key to meeting needs
Numerous factors can foster caregiver well-being, including feelings of accomplishment and contribution, a strengthening of the relationship with the care recipient, and feeling supported by friends, family, and formal care systems.18,19 Family physicians can play an important role by assessing and supporting patients with dementia and their caregivers. Ideally, the individual with dementia and the caregiver will be assessed both together and separately.
A thorough assessment includes gathering information about the context and quality of the caregiving relationship; caregiver perception of the care recipient’s health and functional status; caregiver values and preferences; caregiver well-being (including mental health assessment); caregiver skills, abilities, and knowledge about caregiving; and positive and negative consequences of caregiving.20 Caregiver needs—including informational, care support, emotional, physical, and social needs—also should be assessed.
Continue to: Tools are available...
Tools are available to facilitate caregiver assessment. For example, the Zarit Burden Interview is a 22-item self-report measure that can be given to the caregiver21; shorter versions (4 and 12 items) are also available.22 Another resource available for caregiver assessment guidance is a toolkit developed by the Family Caregiver Alliance.20
Continually assess for changing needs
As the condition of the individual with dementia progresses, it will be important to reassess the caregiver, as stressors and needs will change over the course of the caregiving relationship. Support should be adapted accordingly.
In the early stage of dementia, caregivers may need information on disease progression and dementia care planning, ways to navigate the health care system, financial planning, and useful resources. Caregivers also may need emotional support to help them adapt to the role of caregiver, deal with denial, and manage their stress.23,24
With dementia progression, caregivers may need support related to increased decision-making responsibility, managing challenging behaviors, assisting with ADLs and IADLs, and identifying opportunities to meet personal social and well-being needs. They also may need support to accept the changes they are seeing in the individual with dementia and the shifts they are experiencing in their relationship with him or her.23,25
In late-stage dementia, caregiver needs tend to shift to determining the need for long-term care placement vs staying at home, end-of-life planning, loneliness, and anticipatory grief.23,26 Support with managing changing and accumulating stress typically remains a primary need throughout the progression of dementia.27
Continue to: Specific populations have distinct needs
Specific populations have distinct needs. Some caregivers, including members of the LGBTQ+ community and different racial and ethnic groups, as well as caregivers of people with younger-onset dementia, may have additional support needs.28
For example, African American and Latino caregivers tend to have caregiving relationships of longer duration, requiring more time-intensive care, but use fewer formal support services than White caregivers.29 Caregivers from non-White racial and ethnic groups also are more likely to experience discrimination when interacting with health care services on behalf of care recipients.30
Having an awareness of potential specialized needs may help to prevent or address potential care disparities, and cultural humility may help to improve caregiver experiences with primary care physicians.
Resources to support caregivers
Family physicians are well situated to provide informational and emotional support for both patients with dementia and their informal care providers.31 Given the variability of caregiver concerns, multicomponent interventions addressing informational, self-care, social support, and financial needs often are needed.31 Supportive counseling and psychoeducation can help dementia caregivers with stress management, self-care, coping, and skills training—supporting the development of self-efficacy.32,33
Outside resources. Although significant caregiver support can be provided directly by the physician, caregivers should be connected with outside resources, including support groups, counselors, psychotherapists, financial and legal support, and formal care services
Continue to: Psychosocial and complementary interventions
Psychosocial and complementary interventions. Various psychosocial interventions (eg, psychoeducation, cognitive behavioral therapy, support groups) have been found to be beneficial in alleviating caregiver symptoms of depression, anxiety, and stress and improving well-being, perceived burden, and quality of life. However, systematic reviews have found variability in the degree of helpfulness of these interventions.35,36
Some caregivers and care recipients may benefit from complementary and integrative medicine referrals. Mind–body therapies such as mindfulness, yoga, and Tai Chi have shown some beneficial effects.37
Online resources. Caregivers also can be directed to online resources from organizations such as the Alzheimer’s Association (www.alz.org), the National Institutes of Health (www.alzheimers.gov), and the Family Caregiver Alliance (www.caregiver.org).
In rural settings, such as the one in which this case took place, online resources may decrease some barriers to supporting caregivers.38 Internet-based interventions also have been found to have some benefit for dementia caregivers.31,39
However, some rural locations continue to have limited reliable Internet services.40 In affected areas, a strong relationship with a primary care physician may be even more important to the well-being of caregivers, since other support services may be less accessible.41
Continue to: Impacts of the pandemic
Impacts of the pandemic. Although our case took place prior to the COVID-19 pandemic, it is important to acknowledge ways the pandemic has impacted informal dementia caregiving.
Caregiver stress, depression, and anxiety increased during the pandemic, and the need for greater home confinement and social distancing amplified the negative impact of social isolation, including loneliness, on caregivers.42,43 Caregivers often needed to increase their caregiving responsibilities and had more difficulty with care coordination due to limited access to in-person resources.43 The pandemic led to increased reliance on technology and telehealth in the support of dementia caregivers.43
THE CASE
The physician prescribed mirtazapine for Mr. C, titrating the dose as needed to address depressive symptoms and promote weight gain. The physician connected Mr. C’s father with home health services, including physical therapy for fall risk reduction. Mr. C also hired part-time support to provide additional assistance with ADLs and IADLs, allowing Mr. C to have time to attend to his own needs. Though provided with information about a local caregiver support group, Mr. C chose not to attend. The physician also assisted the family with advanced directives.
A particular challenge that occurred during care for the family was addressing Mr. C’s father’s driving capacity, considering his strong need for independence. To address this concern, a family meeting was held with Mr. C, his father, and his siblings from out of town. Although Mr. C’s father was not willing to relinquish his driver’s license during that meeting, he agreed to complete a functional driving assessment.
The physician continued to meet with Mr. C and his father together, as well as with Mr. C individually, to provide supportive counseling as needed. As the father’s dementia progressed and it became more difficult to complete office appointments, the physician transitioned to home visits to provide care until the father’s death.
After the death of Mr. C’s father, the physician continued to serve as Mr. C’s primary care provider.
Keeping the “family”in family medicine
Through longitudinal assessment, needs identification, and provision of relevant information, emotional support, and resources, family physicians can provide care that can improve the quality of life and well-being and help alleviate burden experienced by dementia caregivers. Family physicians also are positioned to provide treatments that can address the negative physical and psychological health outcomes associated with informal dementia caregiving. By building relationships with multiple family members across generations, family physicians can understand the context of caregiving dynamics and work together with individuals with dementia and their caregivers throughout disease progression, providing consistent support to the family unit.
CORRESPONDENCE
Kathleen M. Young, PhD, MPH, Novant Health Family Medicine Wilmington, 2523 Delaney Avenue, Wilmington, NC 28403; [email protected]
1. Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 202319:1598-1695. doi: 10.1002/alz.13016
2. Liu Z, Heffernan C, Tan J. Caregiver burden: a concept analysis. Int J of Nurs Sci. 2020;7:448-435. doi: 10.1016/j.ijnss.2020.07.012
3. Ory MG, Hoffman RR III, Yee JL, et al. Prevalence and impacts of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999;39:177-185. doi: 10.1093/geront/39.2.177
4. Baharudin AD, Din NC, Subramaniam P, et al. The associations between behavioral-psychological symptoms of dementia (BPSD) and coping strategy, burden of care and personality style among low-income caregivers of patients with dementia. BMC Public Health. 2019;19(suppl 4):447. doi: 10.1186/s12889-019-6868-0
5. Cheng S-T. Dementia caregiver burden: a research update and critical analysis. Curr Psychiatry Rep. 2017;19:64. doi: 10.1007/s11920-017-0818-2
6. Reed C, Belger M, Andrews JS, et al. Factors associated with long-term impact on informal caregivers during Alzheimer’s disease dementia progression: 36-month results from GERAS. Int Psychogeriatr. 2020;32:267-277. doi: 10.1017/S1041610219000425
7. Gilhooly KJ, Gilhooly MLM, Sullivan MP, et al. A meta-review of stress, coping and interventions in dementia and dementia caregiving. BMC Geriatr. 2016;16:106. doi: 10.1186/s12877-016-0280-8
8. Haley WE, Levine EG, Brown SL, et al. Psychological, social, and health consequences of caring for a relative with senile dementia. J Am Geriatr Soc. 1987;35:405-411.
9. Bom J, Bakx P, Schut F, et al. The impact of informal caregiving for older adults on the health of various types of caregivers: a systematic review. The Gerontologist. 2019;59:e629-e642. doi: 10.1093/geront/gny137
10. Fonareva I, Oken BS. Physiological and functional consequences of caregiving for relatives with dementia. Int Psychogeriatr. 2014;26:725-747. doi: 10.1017/S1041610214000039
11. Del-Pino-Casado R, Rodriguez Cardosa M, Lopez-Martinez C, et al. The association between subjective caregiver burden and depressive symptoms in carers of older relatives: a systematic review and meta-analysis. PLoS One. 2019;14:e0217648. doi: 10.1371/journal.pone.0217648
12. Del-Pino-Casado R, Priego-Cubero E, Lopez-Martinez C, et al. Subjective caregiver burden and anxiety in informal caregivers: a systematic review and meta-analysis. PLoS One. 2020;16:e0247143. doi: 10.1371/journal.pone.0247143
13. De Souza Alves LC, Quirino Montiero D, Ricarte Bento S, et al. Burnout syndrome in informal caregivers of older adults with dementia: a systematic review. Dement Neuropsychol. 2019;13:415-421. doi: 10.1590/1980-57642018dn13-040008
14. Victor CR, Rippon I, Quinn C, et al. The prevalence and predictors of loneliness in caregivers of people with dementia: findings from the IDEAL programme. Aging Ment Health. 2021;25:1232-1238. doi: 10.1080/13607863.2020.1753014
15. Sallim AB, Sayampanathan AA, Cuttilan A, et al. Prevalence of mental health disorders among caregivers of patients with Alzheimer disease. J Am Med Dir Assoc. 2015;16:1034-1041. doi: 10.1016/j.jamda.2015.09.007
16. Unpublished data from the 2015, 2016 2017, 2020, and 2021 Behavioral Risk Factor Surveillance System survey, analyzed by and provided to the Alzheimer’s Association by the Alzheimer’s Disease and Healthy Aging Program (AD+HP), Centers for Disease Control and Prevention (CDC).
17. Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. J Am Geriatr Soc. 2018;00:1-9. doi: 10.1111/jgs.15690
18. Lindeza P, Rodrigues M, Costa J, et al. Impact of dementia on informal care: a systematic review of family caregivers’ perceptions. BMJ Support Palliat Care. 2020;bmjspcare-2020-002242. doi: 10.1136/bmjspcare-2020-002242
19. Lethin C, Guiteras AR, Zwakhalen S, et al. Psychological well-being over time among informal caregivers caring for persons with dementia living at home. Aging and Ment Health. 2017; 21:1138-1146. doi: 10.1080/13607863.2016.1211621
20. Family Caregiver Alliance. Caregivers Count Too! A Toolkit to Help Practitioners Assess the Needs of Family Caregivers. Family Caregiver Alliance; 2006. Accessed May 16, 2023. www.caregiver.org/uploads/legacy/pdfs/Assessment_Toolkit_20060802.pdf
21. Zarit SH, Zarit JM. Instructions for the Burden Interview. Pennsylvania State University; 1987.
22. University of Wisconsin. Zarit Burden Interview: assessing caregiver burden. Accessed May 19, 2023. https://wai.wisc.edu/wp-content/uploads/sites/1129/2021/11/Zarit-Caregiver-Burden-Assessment-Instruments.pdf
23. Gallagher-Thompson D, Bilbrey AC, Apesoa-Varano EC, et al. Conceptual framework to guide intervention research across the trajectory of dementia caregiving. Gerontologist. 2020;60:S29-S40. doi: 10.1093/geront/gnz157
24. Queluz FNFR, Kervin E, Wozney L, et al. Understanding the needs of caregivers of persons with dementia: a scoping review. Int Psychogeriatr. 2020;32:35-52. doi: 10.1017/S1041610219000243
25. McCabe M, You E, Tatangelo G. Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist. 2016;56:e70-e88. doi: 10.1093/geront/gnw07
26. Zwaanswijk M, Peeters JM, van Beek AP, et al. Informal caregivers of people with dementia: problems, needs and support in the initial stage and in subsequent stages of dementia: a questionnaire survey. Open Nurs J. 2013;7:6-13. doi: 10.2174/1874434601307010006
27. Jennings LA, Palimaru A, Corona MG, et al. Patient and caregiver goals for dementia care. Qual Life Res. 2017;26:685-693. doi: 10.1007/s11136-016-1471-7
28. Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009;11:217-228. doi: 10.31887/DCNS.2009.11.2/hbrodaty
29. Rote SM, Angel JL, Moon H, et al. Caregiving across diverse populations: new evidence from the national study of caregiving and Hispanic EPESE. Innovation in Aging. 2019;3:1-11. doi: 10.1093/geroni/igz033
30. Alzheimer’s Association. 2021 Alzheimer’s Disease facts and figures. Special report—race, ethnicity, and Alzheimer’s in America. Alzheimers Dement. 2021;17:70-104. doi: 10.1002/alz.12328
31. Swartz K, Collins LG. Caregiver care. Am Fam Physician. 2019;99:699-706.
32. Cheng ST, Au A, Losada A, et al. Psychological interventions for dementia caregivers: what we have achieved, what we have learned. Curr Psychiatry Rep. 2019;21:59. doi: 10.1007/s11920-019-1045-9
33. Jennings LA, Reuben DB, Everston LC, et al. Unmet needs of caregivers of patients referred to a dementia care program. J Am Geriatr Soc. 2015;63:282-289. doi: 10.1111/jgs.13251
34. Soong A, Au ST, Kyaw BM, et al. Information needs and information seeking behaviour of people with dementia and their non-professional caregivers: a scoping review. BMC Geriatrics. 2020;20:61. doi: 10.1186/s12877-020-1454-y
35. Cheng S-T, Zhang F. A comprehensive meta-review of systematic reviews and meta-analyses on nonpharmacological interventions for informal dementia caregivers. BMC Geriatrics. 2020;20:137. doi: 10.1186/s12877-020-01547-2
36. Wiegelmann H, Speller S, Verhaert LM, et al. Psychosocial interventions to support the mental health of informal caregivers of persons living with dementia—a systematic literature review. BMC Geriatrics. 2021;21:94. doi: 10.1186/s12877-021-02020-4
37. Nguyen SA, Oughli HA, Lavretsky H. Complementary and integrative medicine for neurocognitive disorders and caregiver health. Current Psychiatry Reports. 2022;24:469-480. doi: 10.1007/s11920-022-01355-y
38. Gibson A, Holmes SD, Fields NL, et al. Providing care for persons with dementia in rural communities: informal caregivers’ perceptions of supports and services. J Gerontol Soc Work. 2019;62:630-648. doi: 10.1080/01634372.2019.1636332
39. Leng M, Zhao Y, Xiau H, et al. Internet-based supportive interventions for family caregivers of people with dementia: systematic review and meta-analysis. J Med Internet Res. 2020;22:e19468. doi: 10.2196/19468
40. Ruggiano N, Brown EL, Li J, et al. Rural dementia caregivers and technology. What is the evidence? Res Gerontol Nurs. 2018;11:216-224. doi: 10.3928/19404921-20180628-04
41. Shuffler J, Lee K, Fields, et al. Challenges experienced by rural informal caregivers of older adults in the United States: a scoping review. J Evid Based Soc Work. Published online 24 February 24, 2023. doi:10.1080/26408066.2023.2183102
42. Hughes MC, Liu Y, Baumbach A. Impact of COVID-19 on the health and well-being of informal caregivers of people with dementia: a rapid systematic review. Gerontol Geriatric Med. 2021;7:1-8. doi: 10.1177/2333721421102164
43. Paplickar A, Rajagopalan J, Alladi S. Care for dementia patients and caregivers amid COVID-19 pandemic. Cereb Circ Cogn Behav. 2022;3:100040. doi: 10.1016/j.cccb.2022.100040
THE CASE
Sam C* is a 68-year-old man who presented to his family physician in a rural health clinic due to concerns about weight loss. Since his visit 8 months prior, Mr. C unintentionally had lost 20 pounds. Upon questioning, Mr. C also reported feeling irritable and having difficulty with sleep and concentration.
A review of systems did not indicate the presence of infection or other medical conditions. In the 6 years since becoming a patient to the practice, he had reported no chronic health concerns, was taking no medications, and had only been to the clinic for his annual check-up appointments. He completed a Patient Health Questionnaire (PHQ-9) and scored 18, indicating moderately severe depression.
Mr. C had established care with his physician when he moved to the area from out of state so that he could be closer to his parents, who were in their mid-80s at the time. Mr. C’s physician also had been the family physician for his parents for the previous 20 years. Three years prior to Mr. C’s presentation for weight loss, his mother had received a diagnosis of acute leukemia; she died a year later.
Over the past year, Mr. C had needed to take a more active role in the care of his father, who was now in his early 90s. Mr. C’s father, who was previously in excellent health, had begun to develop significant health problems, including degenerative arthritis and progressive vascular dementia. He also had ataxia, leading to poor mobility, and a neurogenic bladder requiring self-catheterization, which required Mr. C’s assistance. Mr. C lived next door to his father and provided frequent assistance with activities of daily living. However, his father, who always had been the dominant figure in the family, was determined to maintain his independence and not relinquish control to others.
The strain of caregiving activities, along with managing his father’s inflexibility, was causing increasing distress for Mr. C. As he told his family physician, “I just don’t know what to do.”
●
* The patient’s name has been changed to protect his identity.
It is estimated that more than 11 million Americans provided more than 18 billion hours in unpaid support for individuals with dementia in 2022, averaging 30 hours of care per caregiver per week.1 As individuals with dementia progressively decline, they require increased assistance with activities of daily living (ADLs, such as bathing and dressing) and instrumental activities of daily living (IADLs, such as paying bills and using transportation). Most of this assistance comes from informal caregiving provided by family members and friends.
Caregiver burden can be defined as “the strain or load borne by a person who cares for a chronically ill, disabled, or elderly family member.”2 Caregiver stress has been found to be higher for dementia caregiving than other types of caregiving.3 In particular, caring for someone with greater behavioral and psychological symptoms of dementia (BPSDs) has been associated with higher caregiver burden.4-

Beyond the subjective burden of caregiving, there are other potential negative consequences for dementia caregivers (see TABLE 18-14 and TABLE 215,16). In addition, caregiver distress is related to a number of care recipient outcomes, including earlier institutionalization, more hospitalizations, more BPSDs, poorer quality of life, and greater likelihood of experiencing elder abuse.17

Assessment, reassessment are key to meeting needs
Numerous factors can foster caregiver well-being, including feelings of accomplishment and contribution, a strengthening of the relationship with the care recipient, and feeling supported by friends, family, and formal care systems.18,19 Family physicians can play an important role by assessing and supporting patients with dementia and their caregivers. Ideally, the individual with dementia and the caregiver will be assessed both together and separately.
A thorough assessment includes gathering information about the context and quality of the caregiving relationship; caregiver perception of the care recipient’s health and functional status; caregiver values and preferences; caregiver well-being (including mental health assessment); caregiver skills, abilities, and knowledge about caregiving; and positive and negative consequences of caregiving.20 Caregiver needs—including informational, care support, emotional, physical, and social needs—also should be assessed.
Continue to: Tools are available...
Tools are available to facilitate caregiver assessment. For example, the Zarit Burden Interview is a 22-item self-report measure that can be given to the caregiver21; shorter versions (4 and 12 items) are also available.22 Another resource available for caregiver assessment guidance is a toolkit developed by the Family Caregiver Alliance.20
Continually assess for changing needs
As the condition of the individual with dementia progresses, it will be important to reassess the caregiver, as stressors and needs will change over the course of the caregiving relationship. Support should be adapted accordingly.
In the early stage of dementia, caregivers may need information on disease progression and dementia care planning, ways to navigate the health care system, financial planning, and useful resources. Caregivers also may need emotional support to help them adapt to the role of caregiver, deal with denial, and manage their stress.23,24
With dementia progression, caregivers may need support related to increased decision-making responsibility, managing challenging behaviors, assisting with ADLs and IADLs, and identifying opportunities to meet personal social and well-being needs. They also may need support to accept the changes they are seeing in the individual with dementia and the shifts they are experiencing in their relationship with him or her.23,25
In late-stage dementia, caregiver needs tend to shift to determining the need for long-term care placement vs staying at home, end-of-life planning, loneliness, and anticipatory grief.23,26 Support with managing changing and accumulating stress typically remains a primary need throughout the progression of dementia.27
Continue to: Specific populations have distinct needs
Specific populations have distinct needs. Some caregivers, including members of the LGBTQ+ community and different racial and ethnic groups, as well as caregivers of people with younger-onset dementia, may have additional support needs.28
For example, African American and Latino caregivers tend to have caregiving relationships of longer duration, requiring more time-intensive care, but use fewer formal support services than White caregivers.29 Caregivers from non-White racial and ethnic groups also are more likely to experience discrimination when interacting with health care services on behalf of care recipients.30
Having an awareness of potential specialized needs may help to prevent or address potential care disparities, and cultural humility may help to improve caregiver experiences with primary care physicians.
Resources to support caregivers
Family physicians are well situated to provide informational and emotional support for both patients with dementia and their informal care providers.31 Given the variability of caregiver concerns, multicomponent interventions addressing informational, self-care, social support, and financial needs often are needed.31 Supportive counseling and psychoeducation can help dementia caregivers with stress management, self-care, coping, and skills training—supporting the development of self-efficacy.32,33
Outside resources. Although significant caregiver support can be provided directly by the physician, caregivers should be connected with outside resources, including support groups, counselors, psychotherapists, financial and legal support, and formal care services
Continue to: Psychosocial and complementary interventions
Psychosocial and complementary interventions. Various psychosocial interventions (eg, psychoeducation, cognitive behavioral therapy, support groups) have been found to be beneficial in alleviating caregiver symptoms of depression, anxiety, and stress and improving well-being, perceived burden, and quality of life. However, systematic reviews have found variability in the degree of helpfulness of these interventions.35,36
Some caregivers and care recipients may benefit from complementary and integrative medicine referrals. Mind–body therapies such as mindfulness, yoga, and Tai Chi have shown some beneficial effects.37
Online resources. Caregivers also can be directed to online resources from organizations such as the Alzheimer’s Association (www.alz.org), the National Institutes of Health (www.alzheimers.gov), and the Family Caregiver Alliance (www.caregiver.org).
In rural settings, such as the one in which this case took place, online resources may decrease some barriers to supporting caregivers.38 Internet-based interventions also have been found to have some benefit for dementia caregivers.31,39
However, some rural locations continue to have limited reliable Internet services.40 In affected areas, a strong relationship with a primary care physician may be even more important to the well-being of caregivers, since other support services may be less accessible.41
Continue to: Impacts of the pandemic
Impacts of the pandemic. Although our case took place prior to the COVID-19 pandemic, it is important to acknowledge ways the pandemic has impacted informal dementia caregiving.
Caregiver stress, depression, and anxiety increased during the pandemic, and the need for greater home confinement and social distancing amplified the negative impact of social isolation, including loneliness, on caregivers.42,43 Caregivers often needed to increase their caregiving responsibilities and had more difficulty with care coordination due to limited access to in-person resources.43 The pandemic led to increased reliance on technology and telehealth in the support of dementia caregivers.43
THE CASE
The physician prescribed mirtazapine for Mr. C, titrating the dose as needed to address depressive symptoms and promote weight gain. The physician connected Mr. C’s father with home health services, including physical therapy for fall risk reduction. Mr. C also hired part-time support to provide additional assistance with ADLs and IADLs, allowing Mr. C to have time to attend to his own needs. Though provided with information about a local caregiver support group, Mr. C chose not to attend. The physician also assisted the family with advanced directives.
A particular challenge that occurred during care for the family was addressing Mr. C’s father’s driving capacity, considering his strong need for independence. To address this concern, a family meeting was held with Mr. C, his father, and his siblings from out of town. Although Mr. C’s father was not willing to relinquish his driver’s license during that meeting, he agreed to complete a functional driving assessment.
The physician continued to meet with Mr. C and his father together, as well as with Mr. C individually, to provide supportive counseling as needed. As the father’s dementia progressed and it became more difficult to complete office appointments, the physician transitioned to home visits to provide care until the father’s death.
After the death of Mr. C’s father, the physician continued to serve as Mr. C’s primary care provider.
Keeping the “family”in family medicine
Through longitudinal assessment, needs identification, and provision of relevant information, emotional support, and resources, family physicians can provide care that can improve the quality of life and well-being and help alleviate burden experienced by dementia caregivers. Family physicians also are positioned to provide treatments that can address the negative physical and psychological health outcomes associated with informal dementia caregiving. By building relationships with multiple family members across generations, family physicians can understand the context of caregiving dynamics and work together with individuals with dementia and their caregivers throughout disease progression, providing consistent support to the family unit.
CORRESPONDENCE
Kathleen M. Young, PhD, MPH, Novant Health Family Medicine Wilmington, 2523 Delaney Avenue, Wilmington, NC 28403; [email protected]
THE CASE
Sam C* is a 68-year-old man who presented to his family physician in a rural health clinic due to concerns about weight loss. Since his visit 8 months prior, Mr. C unintentionally had lost 20 pounds. Upon questioning, Mr. C also reported feeling irritable and having difficulty with sleep and concentration.
A review of systems did not indicate the presence of infection or other medical conditions. In the 6 years since becoming a patient to the practice, he had reported no chronic health concerns, was taking no medications, and had only been to the clinic for his annual check-up appointments. He completed a Patient Health Questionnaire (PHQ-9) and scored 18, indicating moderately severe depression.
Mr. C had established care with his physician when he moved to the area from out of state so that he could be closer to his parents, who were in their mid-80s at the time. Mr. C’s physician also had been the family physician for his parents for the previous 20 years. Three years prior to Mr. C’s presentation for weight loss, his mother had received a diagnosis of acute leukemia; she died a year later.
Over the past year, Mr. C had needed to take a more active role in the care of his father, who was now in his early 90s. Mr. C’s father, who was previously in excellent health, had begun to develop significant health problems, including degenerative arthritis and progressive vascular dementia. He also had ataxia, leading to poor mobility, and a neurogenic bladder requiring self-catheterization, which required Mr. C’s assistance. Mr. C lived next door to his father and provided frequent assistance with activities of daily living. However, his father, who always had been the dominant figure in the family, was determined to maintain his independence and not relinquish control to others.
The strain of caregiving activities, along with managing his father’s inflexibility, was causing increasing distress for Mr. C. As he told his family physician, “I just don’t know what to do.”
●
* The patient’s name has been changed to protect his identity.
It is estimated that more than 11 million Americans provided more than 18 billion hours in unpaid support for individuals with dementia in 2022, averaging 30 hours of care per caregiver per week.1 As individuals with dementia progressively decline, they require increased assistance with activities of daily living (ADLs, such as bathing and dressing) and instrumental activities of daily living (IADLs, such as paying bills and using transportation). Most of this assistance comes from informal caregiving provided by family members and friends.
Caregiver burden can be defined as “the strain or load borne by a person who cares for a chronically ill, disabled, or elderly family member.”2 Caregiver stress has been found to be higher for dementia caregiving than other types of caregiving.3 In particular, caring for someone with greater behavioral and psychological symptoms of dementia (BPSDs) has been associated with higher caregiver burden.4-

Beyond the subjective burden of caregiving, there are other potential negative consequences for dementia caregivers (see TABLE 18-14 and TABLE 215,16). In addition, caregiver distress is related to a number of care recipient outcomes, including earlier institutionalization, more hospitalizations, more BPSDs, poorer quality of life, and greater likelihood of experiencing elder abuse.17

Assessment, reassessment are key to meeting needs
Numerous factors can foster caregiver well-being, including feelings of accomplishment and contribution, a strengthening of the relationship with the care recipient, and feeling supported by friends, family, and formal care systems.18,19 Family physicians can play an important role by assessing and supporting patients with dementia and their caregivers. Ideally, the individual with dementia and the caregiver will be assessed both together and separately.
A thorough assessment includes gathering information about the context and quality of the caregiving relationship; caregiver perception of the care recipient’s health and functional status; caregiver values and preferences; caregiver well-being (including mental health assessment); caregiver skills, abilities, and knowledge about caregiving; and positive and negative consequences of caregiving.20 Caregiver needs—including informational, care support, emotional, physical, and social needs—also should be assessed.
Continue to: Tools are available...
Tools are available to facilitate caregiver assessment. For example, the Zarit Burden Interview is a 22-item self-report measure that can be given to the caregiver21; shorter versions (4 and 12 items) are also available.22 Another resource available for caregiver assessment guidance is a toolkit developed by the Family Caregiver Alliance.20
Continually assess for changing needs
As the condition of the individual with dementia progresses, it will be important to reassess the caregiver, as stressors and needs will change over the course of the caregiving relationship. Support should be adapted accordingly.
In the early stage of dementia, caregivers may need information on disease progression and dementia care planning, ways to navigate the health care system, financial planning, and useful resources. Caregivers also may need emotional support to help them adapt to the role of caregiver, deal with denial, and manage their stress.23,24
With dementia progression, caregivers may need support related to increased decision-making responsibility, managing challenging behaviors, assisting with ADLs and IADLs, and identifying opportunities to meet personal social and well-being needs. They also may need support to accept the changes they are seeing in the individual with dementia and the shifts they are experiencing in their relationship with him or her.23,25
In late-stage dementia, caregiver needs tend to shift to determining the need for long-term care placement vs staying at home, end-of-life planning, loneliness, and anticipatory grief.23,26 Support with managing changing and accumulating stress typically remains a primary need throughout the progression of dementia.27
Continue to: Specific populations have distinct needs
Specific populations have distinct needs. Some caregivers, including members of the LGBTQ+ community and different racial and ethnic groups, as well as caregivers of people with younger-onset dementia, may have additional support needs.28
For example, African American and Latino caregivers tend to have caregiving relationships of longer duration, requiring more time-intensive care, but use fewer formal support services than White caregivers.29 Caregivers from non-White racial and ethnic groups also are more likely to experience discrimination when interacting with health care services on behalf of care recipients.30
Having an awareness of potential specialized needs may help to prevent or address potential care disparities, and cultural humility may help to improve caregiver experiences with primary care physicians.
Resources to support caregivers
Family physicians are well situated to provide informational and emotional support for both patients with dementia and their informal care providers.31 Given the variability of caregiver concerns, multicomponent interventions addressing informational, self-care, social support, and financial needs often are needed.31 Supportive counseling and psychoeducation can help dementia caregivers with stress management, self-care, coping, and skills training—supporting the development of self-efficacy.32,33
Outside resources. Although significant caregiver support can be provided directly by the physician, caregivers should be connected with outside resources, including support groups, counselors, psychotherapists, financial and legal support, and formal care services
Continue to: Psychosocial and complementary interventions
Psychosocial and complementary interventions. Various psychosocial interventions (eg, psychoeducation, cognitive behavioral therapy, support groups) have been found to be beneficial in alleviating caregiver symptoms of depression, anxiety, and stress and improving well-being, perceived burden, and quality of life. However, systematic reviews have found variability in the degree of helpfulness of these interventions.35,36
Some caregivers and care recipients may benefit from complementary and integrative medicine referrals. Mind–body therapies such as mindfulness, yoga, and Tai Chi have shown some beneficial effects.37
Online resources. Caregivers also can be directed to online resources from organizations such as the Alzheimer’s Association (www.alz.org), the National Institutes of Health (www.alzheimers.gov), and the Family Caregiver Alliance (www.caregiver.org).
In rural settings, such as the one in which this case took place, online resources may decrease some barriers to supporting caregivers.38 Internet-based interventions also have been found to have some benefit for dementia caregivers.31,39
However, some rural locations continue to have limited reliable Internet services.40 In affected areas, a strong relationship with a primary care physician may be even more important to the well-being of caregivers, since other support services may be less accessible.41
Continue to: Impacts of the pandemic
Impacts of the pandemic. Although our case took place prior to the COVID-19 pandemic, it is important to acknowledge ways the pandemic has impacted informal dementia caregiving.
Caregiver stress, depression, and anxiety increased during the pandemic, and the need for greater home confinement and social distancing amplified the negative impact of social isolation, including loneliness, on caregivers.42,43 Caregivers often needed to increase their caregiving responsibilities and had more difficulty with care coordination due to limited access to in-person resources.43 The pandemic led to increased reliance on technology and telehealth in the support of dementia caregivers.43
THE CASE
The physician prescribed mirtazapine for Mr. C, titrating the dose as needed to address depressive symptoms and promote weight gain. The physician connected Mr. C’s father with home health services, including physical therapy for fall risk reduction. Mr. C also hired part-time support to provide additional assistance with ADLs and IADLs, allowing Mr. C to have time to attend to his own needs. Though provided with information about a local caregiver support group, Mr. C chose not to attend. The physician also assisted the family with advanced directives.
A particular challenge that occurred during care for the family was addressing Mr. C’s father’s driving capacity, considering his strong need for independence. To address this concern, a family meeting was held with Mr. C, his father, and his siblings from out of town. Although Mr. C’s father was not willing to relinquish his driver’s license during that meeting, he agreed to complete a functional driving assessment.
The physician continued to meet with Mr. C and his father together, as well as with Mr. C individually, to provide supportive counseling as needed. As the father’s dementia progressed and it became more difficult to complete office appointments, the physician transitioned to home visits to provide care until the father’s death.
After the death of Mr. C’s father, the physician continued to serve as Mr. C’s primary care provider.
Keeping the “family”in family medicine
Through longitudinal assessment, needs identification, and provision of relevant information, emotional support, and resources, family physicians can provide care that can improve the quality of life and well-being and help alleviate burden experienced by dementia caregivers. Family physicians also are positioned to provide treatments that can address the negative physical and psychological health outcomes associated with informal dementia caregiving. By building relationships with multiple family members across generations, family physicians can understand the context of caregiving dynamics and work together with individuals with dementia and their caregivers throughout disease progression, providing consistent support to the family unit.
CORRESPONDENCE
Kathleen M. Young, PhD, MPH, Novant Health Family Medicine Wilmington, 2523 Delaney Avenue, Wilmington, NC 28403; [email protected]
1. Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 202319:1598-1695. doi: 10.1002/alz.13016
2. Liu Z, Heffernan C, Tan J. Caregiver burden: a concept analysis. Int J of Nurs Sci. 2020;7:448-435. doi: 10.1016/j.ijnss.2020.07.012
3. Ory MG, Hoffman RR III, Yee JL, et al. Prevalence and impacts of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999;39:177-185. doi: 10.1093/geront/39.2.177
4. Baharudin AD, Din NC, Subramaniam P, et al. The associations between behavioral-psychological symptoms of dementia (BPSD) and coping strategy, burden of care and personality style among low-income caregivers of patients with dementia. BMC Public Health. 2019;19(suppl 4):447. doi: 10.1186/s12889-019-6868-0
5. Cheng S-T. Dementia caregiver burden: a research update and critical analysis. Curr Psychiatry Rep. 2017;19:64. doi: 10.1007/s11920-017-0818-2
6. Reed C, Belger M, Andrews JS, et al. Factors associated with long-term impact on informal caregivers during Alzheimer’s disease dementia progression: 36-month results from GERAS. Int Psychogeriatr. 2020;32:267-277. doi: 10.1017/S1041610219000425
7. Gilhooly KJ, Gilhooly MLM, Sullivan MP, et al. A meta-review of stress, coping and interventions in dementia and dementia caregiving. BMC Geriatr. 2016;16:106. doi: 10.1186/s12877-016-0280-8
8. Haley WE, Levine EG, Brown SL, et al. Psychological, social, and health consequences of caring for a relative with senile dementia. J Am Geriatr Soc. 1987;35:405-411.
9. Bom J, Bakx P, Schut F, et al. The impact of informal caregiving for older adults on the health of various types of caregivers: a systematic review. The Gerontologist. 2019;59:e629-e642. doi: 10.1093/geront/gny137
10. Fonareva I, Oken BS. Physiological and functional consequences of caregiving for relatives with dementia. Int Psychogeriatr. 2014;26:725-747. doi: 10.1017/S1041610214000039
11. Del-Pino-Casado R, Rodriguez Cardosa M, Lopez-Martinez C, et al. The association between subjective caregiver burden and depressive symptoms in carers of older relatives: a systematic review and meta-analysis. PLoS One. 2019;14:e0217648. doi: 10.1371/journal.pone.0217648
12. Del-Pino-Casado R, Priego-Cubero E, Lopez-Martinez C, et al. Subjective caregiver burden and anxiety in informal caregivers: a systematic review and meta-analysis. PLoS One. 2020;16:e0247143. doi: 10.1371/journal.pone.0247143
13. De Souza Alves LC, Quirino Montiero D, Ricarte Bento S, et al. Burnout syndrome in informal caregivers of older adults with dementia: a systematic review. Dement Neuropsychol. 2019;13:415-421. doi: 10.1590/1980-57642018dn13-040008
14. Victor CR, Rippon I, Quinn C, et al. The prevalence and predictors of loneliness in caregivers of people with dementia: findings from the IDEAL programme. Aging Ment Health. 2021;25:1232-1238. doi: 10.1080/13607863.2020.1753014
15. Sallim AB, Sayampanathan AA, Cuttilan A, et al. Prevalence of mental health disorders among caregivers of patients with Alzheimer disease. J Am Med Dir Assoc. 2015;16:1034-1041. doi: 10.1016/j.jamda.2015.09.007
16. Unpublished data from the 2015, 2016 2017, 2020, and 2021 Behavioral Risk Factor Surveillance System survey, analyzed by and provided to the Alzheimer’s Association by the Alzheimer’s Disease and Healthy Aging Program (AD+HP), Centers for Disease Control and Prevention (CDC).
17. Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. J Am Geriatr Soc. 2018;00:1-9. doi: 10.1111/jgs.15690
18. Lindeza P, Rodrigues M, Costa J, et al. Impact of dementia on informal care: a systematic review of family caregivers’ perceptions. BMJ Support Palliat Care. 2020;bmjspcare-2020-002242. doi: 10.1136/bmjspcare-2020-002242
19. Lethin C, Guiteras AR, Zwakhalen S, et al. Psychological well-being over time among informal caregivers caring for persons with dementia living at home. Aging and Ment Health. 2017; 21:1138-1146. doi: 10.1080/13607863.2016.1211621
20. Family Caregiver Alliance. Caregivers Count Too! A Toolkit to Help Practitioners Assess the Needs of Family Caregivers. Family Caregiver Alliance; 2006. Accessed May 16, 2023. www.caregiver.org/uploads/legacy/pdfs/Assessment_Toolkit_20060802.pdf
21. Zarit SH, Zarit JM. Instructions for the Burden Interview. Pennsylvania State University; 1987.
22. University of Wisconsin. Zarit Burden Interview: assessing caregiver burden. Accessed May 19, 2023. https://wai.wisc.edu/wp-content/uploads/sites/1129/2021/11/Zarit-Caregiver-Burden-Assessment-Instruments.pdf
23. Gallagher-Thompson D, Bilbrey AC, Apesoa-Varano EC, et al. Conceptual framework to guide intervention research across the trajectory of dementia caregiving. Gerontologist. 2020;60:S29-S40. doi: 10.1093/geront/gnz157
24. Queluz FNFR, Kervin E, Wozney L, et al. Understanding the needs of caregivers of persons with dementia: a scoping review. Int Psychogeriatr. 2020;32:35-52. doi: 10.1017/S1041610219000243
25. McCabe M, You E, Tatangelo G. Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist. 2016;56:e70-e88. doi: 10.1093/geront/gnw07
26. Zwaanswijk M, Peeters JM, van Beek AP, et al. Informal caregivers of people with dementia: problems, needs and support in the initial stage and in subsequent stages of dementia: a questionnaire survey. Open Nurs J. 2013;7:6-13. doi: 10.2174/1874434601307010006
27. Jennings LA, Palimaru A, Corona MG, et al. Patient and caregiver goals for dementia care. Qual Life Res. 2017;26:685-693. doi: 10.1007/s11136-016-1471-7
28. Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009;11:217-228. doi: 10.31887/DCNS.2009.11.2/hbrodaty
29. Rote SM, Angel JL, Moon H, et al. Caregiving across diverse populations: new evidence from the national study of caregiving and Hispanic EPESE. Innovation in Aging. 2019;3:1-11. doi: 10.1093/geroni/igz033
30. Alzheimer’s Association. 2021 Alzheimer’s Disease facts and figures. Special report—race, ethnicity, and Alzheimer’s in America. Alzheimers Dement. 2021;17:70-104. doi: 10.1002/alz.12328
31. Swartz K, Collins LG. Caregiver care. Am Fam Physician. 2019;99:699-706.
32. Cheng ST, Au A, Losada A, et al. Psychological interventions for dementia caregivers: what we have achieved, what we have learned. Curr Psychiatry Rep. 2019;21:59. doi: 10.1007/s11920-019-1045-9
33. Jennings LA, Reuben DB, Everston LC, et al. Unmet needs of caregivers of patients referred to a dementia care program. J Am Geriatr Soc. 2015;63:282-289. doi: 10.1111/jgs.13251
34. Soong A, Au ST, Kyaw BM, et al. Information needs and information seeking behaviour of people with dementia and their non-professional caregivers: a scoping review. BMC Geriatrics. 2020;20:61. doi: 10.1186/s12877-020-1454-y
35. Cheng S-T, Zhang F. A comprehensive meta-review of systematic reviews and meta-analyses on nonpharmacological interventions for informal dementia caregivers. BMC Geriatrics. 2020;20:137. doi: 10.1186/s12877-020-01547-2
36. Wiegelmann H, Speller S, Verhaert LM, et al. Psychosocial interventions to support the mental health of informal caregivers of persons living with dementia—a systematic literature review. BMC Geriatrics. 2021;21:94. doi: 10.1186/s12877-021-02020-4
37. Nguyen SA, Oughli HA, Lavretsky H. Complementary and integrative medicine for neurocognitive disorders and caregiver health. Current Psychiatry Reports. 2022;24:469-480. doi: 10.1007/s11920-022-01355-y
38. Gibson A, Holmes SD, Fields NL, et al. Providing care for persons with dementia in rural communities: informal caregivers’ perceptions of supports and services. J Gerontol Soc Work. 2019;62:630-648. doi: 10.1080/01634372.2019.1636332
39. Leng M, Zhao Y, Xiau H, et al. Internet-based supportive interventions for family caregivers of people with dementia: systematic review and meta-analysis. J Med Internet Res. 2020;22:e19468. doi: 10.2196/19468
40. Ruggiano N, Brown EL, Li J, et al. Rural dementia caregivers and technology. What is the evidence? Res Gerontol Nurs. 2018;11:216-224. doi: 10.3928/19404921-20180628-04
41. Shuffler J, Lee K, Fields, et al. Challenges experienced by rural informal caregivers of older adults in the United States: a scoping review. J Evid Based Soc Work. Published online 24 February 24, 2023. doi:10.1080/26408066.2023.2183102
42. Hughes MC, Liu Y, Baumbach A. Impact of COVID-19 on the health and well-being of informal caregivers of people with dementia: a rapid systematic review. Gerontol Geriatric Med. 2021;7:1-8. doi: 10.1177/2333721421102164
43. Paplickar A, Rajagopalan J, Alladi S. Care for dementia patients and caregivers amid COVID-19 pandemic. Cereb Circ Cogn Behav. 2022;3:100040. doi: 10.1016/j.cccb.2022.100040
1. Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 202319:1598-1695. doi: 10.1002/alz.13016
2. Liu Z, Heffernan C, Tan J. Caregiver burden: a concept analysis. Int J of Nurs Sci. 2020;7:448-435. doi: 10.1016/j.ijnss.2020.07.012
3. Ory MG, Hoffman RR III, Yee JL, et al. Prevalence and impacts of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999;39:177-185. doi: 10.1093/geront/39.2.177
4. Baharudin AD, Din NC, Subramaniam P, et al. The associations between behavioral-psychological symptoms of dementia (BPSD) and coping strategy, burden of care and personality style among low-income caregivers of patients with dementia. BMC Public Health. 2019;19(suppl 4):447. doi: 10.1186/s12889-019-6868-0
5. Cheng S-T. Dementia caregiver burden: a research update and critical analysis. Curr Psychiatry Rep. 2017;19:64. doi: 10.1007/s11920-017-0818-2
6. Reed C, Belger M, Andrews JS, et al. Factors associated with long-term impact on informal caregivers during Alzheimer’s disease dementia progression: 36-month results from GERAS. Int Psychogeriatr. 2020;32:267-277. doi: 10.1017/S1041610219000425
7. Gilhooly KJ, Gilhooly MLM, Sullivan MP, et al. A meta-review of stress, coping and interventions in dementia and dementia caregiving. BMC Geriatr. 2016;16:106. doi: 10.1186/s12877-016-0280-8
8. Haley WE, Levine EG, Brown SL, et al. Psychological, social, and health consequences of caring for a relative with senile dementia. J Am Geriatr Soc. 1987;35:405-411.
9. Bom J, Bakx P, Schut F, et al. The impact of informal caregiving for older adults on the health of various types of caregivers: a systematic review. The Gerontologist. 2019;59:e629-e642. doi: 10.1093/geront/gny137
10. Fonareva I, Oken BS. Physiological and functional consequences of caregiving for relatives with dementia. Int Psychogeriatr. 2014;26:725-747. doi: 10.1017/S1041610214000039
11. Del-Pino-Casado R, Rodriguez Cardosa M, Lopez-Martinez C, et al. The association between subjective caregiver burden and depressive symptoms in carers of older relatives: a systematic review and meta-analysis. PLoS One. 2019;14:e0217648. doi: 10.1371/journal.pone.0217648
12. Del-Pino-Casado R, Priego-Cubero E, Lopez-Martinez C, et al. Subjective caregiver burden and anxiety in informal caregivers: a systematic review and meta-analysis. PLoS One. 2020;16:e0247143. doi: 10.1371/journal.pone.0247143
13. De Souza Alves LC, Quirino Montiero D, Ricarte Bento S, et al. Burnout syndrome in informal caregivers of older adults with dementia: a systematic review. Dement Neuropsychol. 2019;13:415-421. doi: 10.1590/1980-57642018dn13-040008
14. Victor CR, Rippon I, Quinn C, et al. The prevalence and predictors of loneliness in caregivers of people with dementia: findings from the IDEAL programme. Aging Ment Health. 2021;25:1232-1238. doi: 10.1080/13607863.2020.1753014
15. Sallim AB, Sayampanathan AA, Cuttilan A, et al. Prevalence of mental health disorders among caregivers of patients with Alzheimer disease. J Am Med Dir Assoc. 2015;16:1034-1041. doi: 10.1016/j.jamda.2015.09.007
16. Unpublished data from the 2015, 2016 2017, 2020, and 2021 Behavioral Risk Factor Surveillance System survey, analyzed by and provided to the Alzheimer’s Association by the Alzheimer’s Disease and Healthy Aging Program (AD+HP), Centers for Disease Control and Prevention (CDC).
17. Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. J Am Geriatr Soc. 2018;00:1-9. doi: 10.1111/jgs.15690
18. Lindeza P, Rodrigues M, Costa J, et al. Impact of dementia on informal care: a systematic review of family caregivers’ perceptions. BMJ Support Palliat Care. 2020;bmjspcare-2020-002242. doi: 10.1136/bmjspcare-2020-002242
19. Lethin C, Guiteras AR, Zwakhalen S, et al. Psychological well-being over time among informal caregivers caring for persons with dementia living at home. Aging and Ment Health. 2017; 21:1138-1146. doi: 10.1080/13607863.2016.1211621
20. Family Caregiver Alliance. Caregivers Count Too! A Toolkit to Help Practitioners Assess the Needs of Family Caregivers. Family Caregiver Alliance; 2006. Accessed May 16, 2023. www.caregiver.org/uploads/legacy/pdfs/Assessment_Toolkit_20060802.pdf
21. Zarit SH, Zarit JM. Instructions for the Burden Interview. Pennsylvania State University; 1987.
22. University of Wisconsin. Zarit Burden Interview: assessing caregiver burden. Accessed May 19, 2023. https://wai.wisc.edu/wp-content/uploads/sites/1129/2021/11/Zarit-Caregiver-Burden-Assessment-Instruments.pdf
23. Gallagher-Thompson D, Bilbrey AC, Apesoa-Varano EC, et al. Conceptual framework to guide intervention research across the trajectory of dementia caregiving. Gerontologist. 2020;60:S29-S40. doi: 10.1093/geront/gnz157
24. Queluz FNFR, Kervin E, Wozney L, et al. Understanding the needs of caregivers of persons with dementia: a scoping review. Int Psychogeriatr. 2020;32:35-52. doi: 10.1017/S1041610219000243
25. McCabe M, You E, Tatangelo G. Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist. 2016;56:e70-e88. doi: 10.1093/geront/gnw07
26. Zwaanswijk M, Peeters JM, van Beek AP, et al. Informal caregivers of people with dementia: problems, needs and support in the initial stage and in subsequent stages of dementia: a questionnaire survey. Open Nurs J. 2013;7:6-13. doi: 10.2174/1874434601307010006
27. Jennings LA, Palimaru A, Corona MG, et al. Patient and caregiver goals for dementia care. Qual Life Res. 2017;26:685-693. doi: 10.1007/s11136-016-1471-7
28. Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009;11:217-228. doi: 10.31887/DCNS.2009.11.2/hbrodaty
29. Rote SM, Angel JL, Moon H, et al. Caregiving across diverse populations: new evidence from the national study of caregiving and Hispanic EPESE. Innovation in Aging. 2019;3:1-11. doi: 10.1093/geroni/igz033
30. Alzheimer’s Association. 2021 Alzheimer’s Disease facts and figures. Special report—race, ethnicity, and Alzheimer’s in America. Alzheimers Dement. 2021;17:70-104. doi: 10.1002/alz.12328
31. Swartz K, Collins LG. Caregiver care. Am Fam Physician. 2019;99:699-706.
32. Cheng ST, Au A, Losada A, et al. Psychological interventions for dementia caregivers: what we have achieved, what we have learned. Curr Psychiatry Rep. 2019;21:59. doi: 10.1007/s11920-019-1045-9
33. Jennings LA, Reuben DB, Everston LC, et al. Unmet needs of caregivers of patients referred to a dementia care program. J Am Geriatr Soc. 2015;63:282-289. doi: 10.1111/jgs.13251
34. Soong A, Au ST, Kyaw BM, et al. Information needs and information seeking behaviour of people with dementia and their non-professional caregivers: a scoping review. BMC Geriatrics. 2020;20:61. doi: 10.1186/s12877-020-1454-y
35. Cheng S-T, Zhang F. A comprehensive meta-review of systematic reviews and meta-analyses on nonpharmacological interventions for informal dementia caregivers. BMC Geriatrics. 2020;20:137. doi: 10.1186/s12877-020-01547-2
36. Wiegelmann H, Speller S, Verhaert LM, et al. Psychosocial interventions to support the mental health of informal caregivers of persons living with dementia—a systematic literature review. BMC Geriatrics. 2021;21:94. doi: 10.1186/s12877-021-02020-4
37. Nguyen SA, Oughli HA, Lavretsky H. Complementary and integrative medicine for neurocognitive disorders and caregiver health. Current Psychiatry Reports. 2022;24:469-480. doi: 10.1007/s11920-022-01355-y
38. Gibson A, Holmes SD, Fields NL, et al. Providing care for persons with dementia in rural communities: informal caregivers’ perceptions of supports and services. J Gerontol Soc Work. 2019;62:630-648. doi: 10.1080/01634372.2019.1636332
39. Leng M, Zhao Y, Xiau H, et al. Internet-based supportive interventions for family caregivers of people with dementia: systematic review and meta-analysis. J Med Internet Res. 2020;22:e19468. doi: 10.2196/19468
40. Ruggiano N, Brown EL, Li J, et al. Rural dementia caregivers and technology. What is the evidence? Res Gerontol Nurs. 2018;11:216-224. doi: 10.3928/19404921-20180628-04
41. Shuffler J, Lee K, Fields, et al. Challenges experienced by rural informal caregivers of older adults in the United States: a scoping review. J Evid Based Soc Work. Published online 24 February 24, 2023. doi:10.1080/26408066.2023.2183102
42. Hughes MC, Liu Y, Baumbach A. Impact of COVID-19 on the health and well-being of informal caregivers of people with dementia: a rapid systematic review. Gerontol Geriatric Med. 2021;7:1-8. doi: 10.1177/2333721421102164
43. Paplickar A, Rajagopalan J, Alladi S. Care for dementia patients and caregivers amid COVID-19 pandemic. Cereb Circ Cogn Behav. 2022;3:100040. doi: 10.1016/j.cccb.2022.100040
How telehealth can work best for our patients
Social distancing measures instituted during the COVID-19 pandemic challenged the usual way of operating in primary care. To continue delivering medical services, physicians had to transition quickly to forms of remote interaction with patients. Use of technology appeared to be the answer. And it gave clinicians the ability to do what many had long hoped for: offer patients the option of telehealth.
The terms telemedicine and telehealth have similar definitions and are commonly used interchangeably. We think most practices probably would have adopted telehealth earlier were it not for reimbursement barriers. In this article, we adopt the World Health Organization’s definition of telemedicine as: “The delivery of healthcare services, where distance is a critical factor, by all healthcare professionals using information and communication technologies for the exchange of valid information for the diagnosis, treatment, and prevention of disease and injuries, research and evaluation, and for the continuing education of healthcare providers, all in the interests of advancing the health of individuals and their communities.”1
To provide family medicine clinicians with evidence-based recommendations about telehealth, we conducted a critical review of the literature published through April 30, 2021. The scope of this review includes studies found using the PubMed and Google Scholar databases. In addition, we used the keywords “telehealth,” “telemedicine,” “family medicine,” and “primary care.” We divided this review into 6 sections, including focus areas on implementation in primary care, remote diagnostic accuracy, conditions lending themselves to telehealth, physician and patient perceptions, disparities in telehealth, and finally, the conclusions.
Telehealth implementation in primary care
Telehealth in various forms had been around for years before the pandemic, mainly in the form of commercial telehealth businesses. Telehealth was being used in rural and remote areas where it could be difficult to see a primary care provider—let alone a specialist. The family medicine department of the University of Colorado was an early adopter of telehealth and had navigated this transition since 2017, with clinical champions guiding the process. By 2019, 54% of their clinicians were conducting telehealth encounters.2
However, telehealth implementation elsewhere was not accepted so readily. Before the pandemic, a cross-sectional study of more than 1.1 million patients in Northern California showed that 86% preferred in-person care over video.3 Even as the pandemic began and social distancing measures were implemented, a quality improvement project at a family medicine residency clinic in Florida documented that clinicians still preferred telephone interviews despite the capacity for video visits.4 And many primary care systems were simply unprepared to adopt telehealth technologies.
With time, however, family physicians began to improvise using popular videoconferencing technologies (eg, Zoom) that were readily available and familiar to patients, and medical centers began to repurpose their existing videoconferencing systems.5 The Ohio State University Wexner Medical Center launched a virtual health initiative just before the pandemic struck, at which time fewer than 5% of patient visits were conducted through telehealth. Weeks later, nearly 93% of patient visits were offered through telehealth.6
Reimbursement. Another significant impediment to early telehealth uptake was the late reaction by the Centers for Medicare and Medicaid Services (CMS) in changing the payment system. Hectic expansion of telehealth in response to the crisis pointed to the lack of policies that supported primary care with payments based on outcomes rather than fee-for-service models.7 By the end of April 2020, CMS finally announced that video visits would be reimbursed at the same rate as in-person visits. However, telephone-only visits are still very limited in coverage, and appropriate codes should be verified with payers.
Continue to: Remote diagnosis comes with a caveat
Remote diagnosis comes with a caveat
Some primary care practices have found that images of skin lesions submitted by patients (usually by cell phone) suffice for accurate diagnosis in lieu of office visits.8 With chronic conditions, home-based remote monitoring of vital signs may assist in diagnosing and managing acute issues. More efficient triage of patients is increasingly possible with the receipt of still images or video files of concerning lesions (eg, burns, rash, chronic wounds) sent from smartphones alone9,10 or with devices attached to smartphones (eg, parent-managed otoscopes).11,12
Family physicians historically have relied on in-person visits for holistic assessment and diagnosis. Telehealth video visits have the potential to assist with this goal, but there are risks. For example, one patient cut her foot while swimming and the wound became infected.
Specific conditions usually suitable for telehealth evaluation
The pandemic helped us understand that some situations and conditions are better suited than others to coverage by telehealth. The National Ambulatory Medical Care Survey examined 850 million patient–physician encounters and found that 66% of all ambulatory primary care visits required in-office care,15 suggesting that about one-third of patient encounters could be treated via telehealth.
As an example, our southeastern Wisconsin urban clinic has about 20,000 office visits per year. We launched telehealth in March 2020 in direct response to the pandemic. Telehealth usage peaked at the beginning of the pandemic (FIGURE), fell gradually, hit a lower peak in November and December as COVID case counts increased, and then decreased again as our community changed from a “quarantine/lockdown” mentality to “opening up/back to new normal.
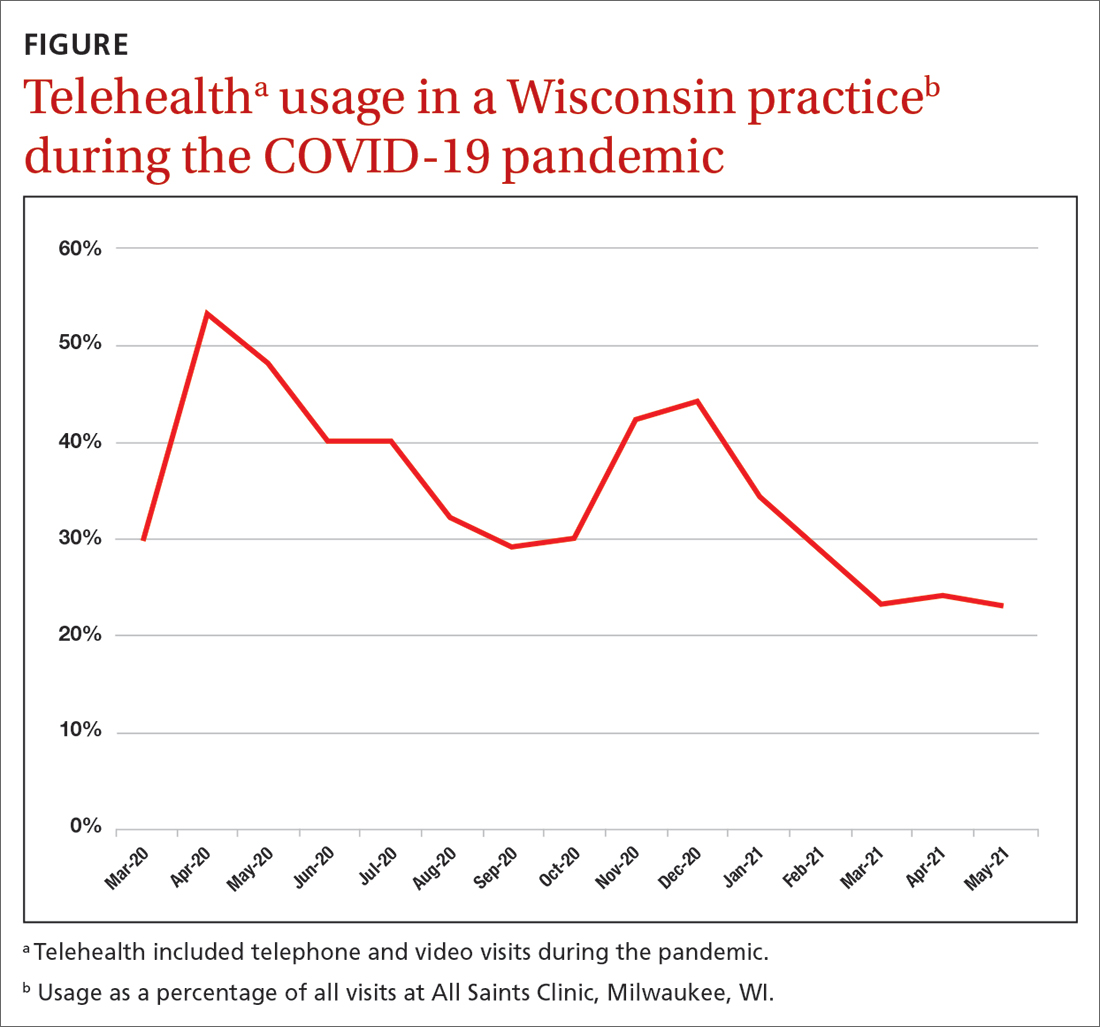
Some conditions can be managed favorably with the telehealth format:
Infectious diseases may be treatable remotely.16,17 Following an initial telehealth visit, the physician can evaluate and recommend further care.
Stable, chronic conditions. Telehealth can be used for stable, chronic conditions such as diabetes, chronic obstructive pulmonary disease, and heart failure when lab or imaging studies are not needed.18
Mental health. Telehealth can be useful in counseling and providing mental health and social support.18 Safeguards can be put in place to protect patient privacy in this setting.19
Behavioral change. Telehealth can be effective in providing support for patients actively trying to quit smoking or lose weight, and for caregivers. A physician who “checks in” can be a positive motivator and can promote a patient’s continued success.20
Continue to: Telehealth is less beneficial...
Telehealth is less beneficial when a physical exam is needed to assess pain, tenderness, strength, or other sensations. Office visits also are required for lab assays and imaging, as in periodic checks of A1C levels in patients with diabetes. As technology advances, home-based laboratory kits and sensors likely will change this picture. New patients may be better served through an initial office visit to develop the patient–physician relationship.
Visual assessment of conditions may be limited by telehealth depending on the quality of the devices used. For example, rashes may be difficult to assess given the clarity of the picture on the device and the ability to see only in 2D. There is still a need for more controlled trials to clarify which conditions can be evaluated and managed by telehealth and which ones need in-person care.21
Physician and patient perceptions of telehealth encounters
Research into family physicians’ perceptions of telehealth is scant. However, 3 studies published in 2021 reveal some advantages and challenges for telehealth adoption.
- A qualitative study found that physicians valued the increased access to care for some patients, changes to reimbursement practices not covered before, and the opportunity to see patients’ home environments.22 Disadvantages included an inability to examine the patient, problems with diagnostic accuracy, hindrances to developing personal connections, and the potential for burnout with on-demand care.22 The researchers suggested that telehealth might better serve to augment in-person care.
- A second study found that clinicians are satisfied with the use of telehealth in general. However, it also noted that the lack of physical examination could hinder accurate diagnosis and treatment.23
- A third study surveyed 109 family physicians, reinforcing the importance of physical exams and highlighting the lack of body language as another barrier.24
In addition, all 3 studies noted that video visits are typically briefer than in-person visits. Previous research predominantly done in specialty and mental health care showed that the benefits of telehealth for physicians include an increase in efficiency, reduced commute time, and improved work-life balance.25
Patient perspectives. Many patients have reported that they prefer telehealth because of lower costs, decreased travel time, and faster health care access.26,27 However, patients also have expressed concerns that the telehealth environment may reduce physician attention, can limit personal interaction (and impart a sense of being rushed), and lacks the physical examination that may be key to an adequate diagnosis.28
Continue to: A survey of 223 patients showed...
A survey of 223 patients showed that sicker patients choose in-person care because they want more in-depth visits with more attention to detail than healthier patients do.29 In a Veterans Affairs health care system qualitative study, patients voiced concerns about communicating with physicians via telehealth, including the potential for errors, less attention paid to their needs, audio difficulties, and challenges to establishing a physician–patient relationship.30 Some patients thought telehealth inhibited their personal expression or that the clinician was not attentive enough. These patient reports underscore the importance of patient–clinician relationships developed in person.31 The perceived level of complexity involved in a visit appears to be an essential factor in a patient opting for telehealth—or not.
In light of these known physician and patient perspectives, it seems wise to develop a hybrid model approach in which visits alternate between telehealth and office.
Patient disparities that may limit the use of telehealth
Race and ethnicity is a major factor in telehealth use. Patients who are Black or Hispanic use telehealth services less often than patients who are White.32,33 A study that looked at patients with chronic conditions—hypertension and diabetes—that disproportionately affect Black and Hispanic patients found that patients in these populations with either of these conditions had a lower prevalence of Internet use when compared with White patients.34 However, subpopulations can vary in their usage. For example, a study in East Harlem, New York, found that Hispanic pregnant women used telehealth frequently for prenatal care and perceived the care as satisfactory.35
Age is also a significant variable in the adoption of telehealth, with pre-COVID-19 studies finding lower use of technology among older adults. However, a study performed at the University of Missouri during the first months of the pandemic found an increase in telehealth use in seniors,32 although the increase was in telephone use and not full video sessions.
Many patients in need of health care services may have older devices and/or low-speed or no Internet access; they also may lack the technical know-how to conduct a telehealth visit.4,36 For example, regardless of race or ethnicity, patients on government insurance (Medicaid and Medicare) have been shown to complete more telephone than video visits,37 underscoring the importance of telehealth practice flexibility and the need for increased technology support to decrease the digital divide. Even with adequate technological support and patient training, telehealth may be more complicated if patients have such comorbidities as hearing, visual, or cognitive impairment.31 Patients from a lower socioeconomic status may feel uncomfortable with providers seeing their home environment on video.38
Overall, incorporating telehealth for the care of older and/or vulnerable patients will present a unique set of challenges that organizations must address. Efforts must be made to understand the available technologies and patients’ comfort in using them. A hybrid model offering telehealth and in-office encounters may be the best solution.
Hernan Barenboim, PhD, KPC Health Group, 301 North San Jacinto Street, Hemet, CA 92543; [email protected]
1. WHO. A health telematics policy: in support of WHO’s Health-for-All strategy for global health development. 1997. Accessed February 8, 2023. https://apps.who.int/iris/bitstream/handle/10665/63857/WHO_DGO_98.1.pdf?sequence=1&isAllowed=y
2. Knierim K, Palmer C, Kramer ES, et al. Lessons learned during COVID-19 that can move telehealth in primary care forward. J Am Board Fam Med. Supplement 2021;34(suppl):S196-S202. doi: 10.3122/jabfm.2021.S1.200419
3. Reed ME, Huang J, Graetz I, et al. Patient characteristics associated with choosing a telemedicine visit vs office visit with the same primary care clinicians. JAMA Netw Open. 2020;3:e205873. doi: 10.1001/jamanetworkopen.2020.5873
4. Silver SL, Lewis MN, Ledford CJ. A stepwise transition to telemedicine in response to COVID-19. J Am Board Fam Med. 2021;34(suppl):S152-S161. doi: 10.3122/jabfm.2021.S1.200358
5. Hron JD, Parsons CR, Williams LA, et al. Rapid implementation of an inpatient telehealth program during the COVID-19 pandemic. Appl Clin Inform. 2020;3:452-459. doi: 10.1055/s-0040-1713635
6. Olayiwola JN, Magaña C, Harmon A, et al. Telehealth as a bright spot of the COVID-19 pandemic: recommendations from the virtual frontlines (“Frontweb”). JMIR Public Health Surveill. 2020;6:e19045. doi: 10.2196/19045
7. Gausvik C, Jabbarpour Y. COVID-19 timeline: Centers for Medicare and Medicaid Services (CMS) changes and primary care support were not enough to prevent practice losses. J Am Board Fam Med. 2021;34(suppl):S7-S9. doi: 10.3122/jabfm.2021.S1.200305
8. Marin-Gomez FX, Vidal-Alaball J, Poch PR, et al. Diagnosis of skin lesions using photographs taken with a mobile phone: an online survey of primary care physicians. J Prim Care Community Health. 2020;11:2150132720937831. doi: 10.1177/2150132720937831
9. Garber RN, Garcia E, Goodwin CW, et al. (2020). Pictures do influence the decision to transfer: outcomes of a telemedicine program serving an eight-state rural population. J Burn Care Res. 2020;41:690-694. doi: 10.1093/jbcr/iraa017
10. Felix F, Greenblatt M, Shin L. Saving limbs in the time of COVID. 2020. Accessed February 8, 2023. https://podiatrym.com/pdf/2020/7/FelixGreenblattShin820web.pdf
11. Erkkola-Anttinen N, Irjala H, Laine MK, et al. Smartphone otoscopy performed by parents. Telemed J E Health. 2019;25:477-484. doi: 10.1089/tmj.2018.0062
12. Verzantvoort NC, Teunis T, Verheij TJ, et al. Self-triage for acute primary care via a smartphone application: practical, safe and efficient? PLoS One. 2018;13:e0199284. doi: 10.1371/journal.pone.0199284
13. Hickner J. When patients don’t get the care they should. J Fam Pract. 2020;69:427.
14. Pappan N, Benkhadra R, Papincak D, et al. Values and limits of telemedicine: a case report. SN Compr Clin Med. 2021;3:317-319. doi: 10.1007/s42399-020-00725-y
15. Jabbarpour Y, Jetty A, Westfall M, et al. Not telehealth: which primary care visits need in-person care? J Am Board Fam Med. 2021;34(suppl):S162-S169. doi: 10.3122/jabfm.2021.S1.200247
16. Parmar P, Mackie D, Varghese S, et al. Use of telemedicine technologies in the management of infectious diseases: a review. Clin Infect Dis. 2015;60:1084-1094. doi: 10.1093/cid/ciu1143
17. Young JD, Abdel-Massih R, Herchline T, et al. Infectious Diseases Society of America position statement on Telehealth and Telemedicine as Applied to the Practice of Infectious Diseases. Clin Infect Dis. 2019;68:1437-1443. doi: 10.1093/cid/ciy907
18. ARHQ. Telehealth: mapping the evidence for patient outcomes from systematic reviews. 2016. Accessed March 27, 2023. https://effectivehealthcare.ahrq.gov/sites/default/files/pdf/telehealth_technical-brief.pdf
19. Lustgarten SD, Garrison YL, Sinnard MT, et al. Digital privacy in mental healthcare: current issues and recommendations for technology use. Curr Opin Psychol. 2020;36:25-31. doi: 10.1016/j.copsyc.2020.03.012
20. Baird A, Xia Y, Cheng Y. Consumer perceptions of telehealth for mental health or substance abuse: a Twitter-based topic modeling analysis. JAMIA Open. 2022;5:ooac028. doi: 10.1093/jamiaopen/ooac028
21. Flumignan CD, da Rocha AP, Pinto AC, et al. What do Cochrane systematic reviews say about telemedicine for healthcare? Sao Paulo Med J. 2019;137:184-192. doi: 10.1590/1516-3180.0177240419
22. Gomez T, Anaya YB, Shih KJ, et al. A qualitative study of primary care physicians’ experiences with telemedicine during COVID-19. J Am Board Fam Med. 2021;34(suppl):S61-S70. doi: 10.3122/jabfm.2021.S1.200517
23. Malliaras P, Merolli M, Williams CM, et al. ‘It’s not hands-on therapy, so it’s very limited’: telehealth use and views among allied health clinicians during the coronavirus pandemic. Musculoskelet Sci Pract. 2021;52:102340. doi: 10.1016/j.msksp.2021.102340
24. Gold KJ, Laurie AR, Kinney DR, et al. Video visits: family physician experiences with uptake during the COVID-19 pandemic. Fam Med. 53:207-210. doi: 10.22454/FamMed.2021.613099
25. Björndell C, Premberg A. Physicians’ experiences of video consultation with patients at a public virtual primary care clinic: a qualitative interview study. Scand J Prim Health Care. 2021;39:67-76. doi: 10.1080/02813432.2021.1882082
26. Powell RE, Henstenburg JM, Cooper G, et al. Patient perceptions of telehealth primary care video visits. Ann Fam Med. 2017;15:225-229. doi: 10.1370/afm.2095
27. Imlach F, McKinlay E, Middleton L, et al. Telehealth consultations in general practice during a pandemic lockdown: survey and interviews on patient experiences and preferences. BMC Fam Pract. 2020;21:1-14. doi: 10.1186/s12875-020-01336-1
28. Gordon HS, Solanki P, Bokhour BG, et al. “I’m not feeling like I’m part of the conversation” patients’ perspectives on communicating in clinical video telehealth visits. J Gen Intern Med. 2020;35:1751-1758. doi: 10.1007/s11606-020-05673-w
29. Volcy J, Smith W, Mills K, et al. Assessment of patient and provider satisfaction with the change to telehealth from in-person visits at an academic safety net institution during the COVID-19 pandemic. J Am Board Fam Med. 2021;34(suppl):S71-S76. doi: 10.3122/jabfm.2021.S1.200393
30. Gopal RK, Solanki P, Bokhour BG, et al. Provider, staff, and patient perspectives on medical visits using clinical video telehealth: a foundation for educational initiatives to improve medical care in telehealth. J Nurse Pract. 2021;17:582-587. doi: 10.1016/j.nurpra.2021.02.020
31. Edgoose JY. Exploring the face-to-face: revisiting patient-doctor relationships in a time of expanding telemedicine. J Am Board Fam Med. 2021;34(suppl):S252-S254. doi: 10.3122/jabfm.2021.S1.200398
32. Pierce RP, Stevermer JJ. Disparities in use of telehealth at the onset of the COVID-19 public health emergency. J Telemed Telecare. 2023;29:3-9. doi: 10.1177/1357633X20963893
33. Lame M, Leyden D, Platt SL. Geocode maps spotlight disparities in telehealth utilization during the COVID-19 pandemic in New York City. Telemed J E Health. 2021;27:251-253. doi: 10.1089/tmj.2020.0297
34. Jain V, Al Rifai M, Lee MT, et al. Racial and geographic disparities in internet use in the US among patients with hypertension or diabetes: implications for telehealth in the era of COVID-19. Diabetes Care. 2021;44:e15-e17. doi: 10.2337/dc20-2016
35. Futterman I, Rosenfeld E, Toaff M, et al. Addressing disparities in prenatal care via telehealth during COVID-19: prenatal satisfaction survey in East Harlem. Am J Perinatol. 2021;38:88-92. doi: 10.1055/s-0040-1718695
36. Wegermann K, Wilder JM, Parish A, et al. Racial and socioeconomic disparities in utilization of telehealth in patients with liver disease during COVID-19. Dig Dis Sci. 2022;67:93-99. doi: 10.1007/s10620-021-06842-5.
37. ASPE. National survey trends in telehealth use in 2021: disparities in utilization and audio vs. video services. Issue brief: February 21, 2022. Accessed March 27, 2023. https://aspe.hhs.gov/sites/default/files/documents/4e1853c0b4885112b2994680a58af9ed/telehealth-hps-ib.pdf
38. Ukoha EP, Davis K, Yinger M, et al. Ensuring equitable implementation of telemedicine in perinatal care. Obstet Gynecol. 2021;137:487-492. doi: 10.1097/AOG.0000000000004276
Social distancing measures instituted during the COVID-19 pandemic challenged the usual way of operating in primary care. To continue delivering medical services, physicians had to transition quickly to forms of remote interaction with patients. Use of technology appeared to be the answer. And it gave clinicians the ability to do what many had long hoped for: offer patients the option of telehealth.
The terms telemedicine and telehealth have similar definitions and are commonly used interchangeably. We think most practices probably would have adopted telehealth earlier were it not for reimbursement barriers. In this article, we adopt the World Health Organization’s definition of telemedicine as: “The delivery of healthcare services, where distance is a critical factor, by all healthcare professionals using information and communication technologies for the exchange of valid information for the diagnosis, treatment, and prevention of disease and injuries, research and evaluation, and for the continuing education of healthcare providers, all in the interests of advancing the health of individuals and their communities.”1
To provide family medicine clinicians with evidence-based recommendations about telehealth, we conducted a critical review of the literature published through April 30, 2021. The scope of this review includes studies found using the PubMed and Google Scholar databases. In addition, we used the keywords “telehealth,” “telemedicine,” “family medicine,” and “primary care.” We divided this review into 6 sections, including focus areas on implementation in primary care, remote diagnostic accuracy, conditions lending themselves to telehealth, physician and patient perceptions, disparities in telehealth, and finally, the conclusions.
Telehealth implementation in primary care
Telehealth in various forms had been around for years before the pandemic, mainly in the form of commercial telehealth businesses. Telehealth was being used in rural and remote areas where it could be difficult to see a primary care provider—let alone a specialist. The family medicine department of the University of Colorado was an early adopter of telehealth and had navigated this transition since 2017, with clinical champions guiding the process. By 2019, 54% of their clinicians were conducting telehealth encounters.2
However, telehealth implementation elsewhere was not accepted so readily. Before the pandemic, a cross-sectional study of more than 1.1 million patients in Northern California showed that 86% preferred in-person care over video.3 Even as the pandemic began and social distancing measures were implemented, a quality improvement project at a family medicine residency clinic in Florida documented that clinicians still preferred telephone interviews despite the capacity for video visits.4 And many primary care systems were simply unprepared to adopt telehealth technologies.
With time, however, family physicians began to improvise using popular videoconferencing technologies (eg, Zoom) that were readily available and familiar to patients, and medical centers began to repurpose their existing videoconferencing systems.5 The Ohio State University Wexner Medical Center launched a virtual health initiative just before the pandemic struck, at which time fewer than 5% of patient visits were conducted through telehealth. Weeks later, nearly 93% of patient visits were offered through telehealth.6
Reimbursement. Another significant impediment to early telehealth uptake was the late reaction by the Centers for Medicare and Medicaid Services (CMS) in changing the payment system. Hectic expansion of telehealth in response to the crisis pointed to the lack of policies that supported primary care with payments based on outcomes rather than fee-for-service models.7 By the end of April 2020, CMS finally announced that video visits would be reimbursed at the same rate as in-person visits. However, telephone-only visits are still very limited in coverage, and appropriate codes should be verified with payers.
Continue to: Remote diagnosis comes with a caveat
Remote diagnosis comes with a caveat
Some primary care practices have found that images of skin lesions submitted by patients (usually by cell phone) suffice for accurate diagnosis in lieu of office visits.8 With chronic conditions, home-based remote monitoring of vital signs may assist in diagnosing and managing acute issues. More efficient triage of patients is increasingly possible with the receipt of still images or video files of concerning lesions (eg, burns, rash, chronic wounds) sent from smartphones alone9,10 or with devices attached to smartphones (eg, parent-managed otoscopes).11,12
Family physicians historically have relied on in-person visits for holistic assessment and diagnosis. Telehealth video visits have the potential to assist with this goal, but there are risks. For example, one patient cut her foot while swimming and the wound became infected.
Specific conditions usually suitable for telehealth evaluation
The pandemic helped us understand that some situations and conditions are better suited than others to coverage by telehealth. The National Ambulatory Medical Care Survey examined 850 million patient–physician encounters and found that 66% of all ambulatory primary care visits required in-office care,15 suggesting that about one-third of patient encounters could be treated via telehealth.
As an example, our southeastern Wisconsin urban clinic has about 20,000 office visits per year. We launched telehealth in March 2020 in direct response to the pandemic. Telehealth usage peaked at the beginning of the pandemic (FIGURE), fell gradually, hit a lower peak in November and December as COVID case counts increased, and then decreased again as our community changed from a “quarantine/lockdown” mentality to “opening up/back to new normal.

Some conditions can be managed favorably with the telehealth format:
Infectious diseases may be treatable remotely.16,17 Following an initial telehealth visit, the physician can evaluate and recommend further care.
Stable, chronic conditions. Telehealth can be used for stable, chronic conditions such as diabetes, chronic obstructive pulmonary disease, and heart failure when lab or imaging studies are not needed.18
Mental health. Telehealth can be useful in counseling and providing mental health and social support.18 Safeguards can be put in place to protect patient privacy in this setting.19
Behavioral change. Telehealth can be effective in providing support for patients actively trying to quit smoking or lose weight, and for caregivers. A physician who “checks in” can be a positive motivator and can promote a patient’s continued success.20
Continue to: Telehealth is less beneficial...
Telehealth is less beneficial when a physical exam is needed to assess pain, tenderness, strength, or other sensations. Office visits also are required for lab assays and imaging, as in periodic checks of A1C levels in patients with diabetes. As technology advances, home-based laboratory kits and sensors likely will change this picture. New patients may be better served through an initial office visit to develop the patient–physician relationship.
Visual assessment of conditions may be limited by telehealth depending on the quality of the devices used. For example, rashes may be difficult to assess given the clarity of the picture on the device and the ability to see only in 2D. There is still a need for more controlled trials to clarify which conditions can be evaluated and managed by telehealth and which ones need in-person care.21
Physician and patient perceptions of telehealth encounters
Research into family physicians’ perceptions of telehealth is scant. However, 3 studies published in 2021 reveal some advantages and challenges for telehealth adoption.
- A qualitative study found that physicians valued the increased access to care for some patients, changes to reimbursement practices not covered before, and the opportunity to see patients’ home environments.22 Disadvantages included an inability to examine the patient, problems with diagnostic accuracy, hindrances to developing personal connections, and the potential for burnout with on-demand care.22 The researchers suggested that telehealth might better serve to augment in-person care.
- A second study found that clinicians are satisfied with the use of telehealth in general. However, it also noted that the lack of physical examination could hinder accurate diagnosis and treatment.23
- A third study surveyed 109 family physicians, reinforcing the importance of physical exams and highlighting the lack of body language as another barrier.24
In addition, all 3 studies noted that video visits are typically briefer than in-person visits. Previous research predominantly done in specialty and mental health care showed that the benefits of telehealth for physicians include an increase in efficiency, reduced commute time, and improved work-life balance.25
Patient perspectives. Many patients have reported that they prefer telehealth because of lower costs, decreased travel time, and faster health care access.26,27 However, patients also have expressed concerns that the telehealth environment may reduce physician attention, can limit personal interaction (and impart a sense of being rushed), and lacks the physical examination that may be key to an adequate diagnosis.28
Continue to: A survey of 223 patients showed...
A survey of 223 patients showed that sicker patients choose in-person care because they want more in-depth visits with more attention to detail than healthier patients do.29 In a Veterans Affairs health care system qualitative study, patients voiced concerns about communicating with physicians via telehealth, including the potential for errors, less attention paid to their needs, audio difficulties, and challenges to establishing a physician–patient relationship.30 Some patients thought telehealth inhibited their personal expression or that the clinician was not attentive enough. These patient reports underscore the importance of patient–clinician relationships developed in person.31 The perceived level of complexity involved in a visit appears to be an essential factor in a patient opting for telehealth—or not.
In light of these known physician and patient perspectives, it seems wise to develop a hybrid model approach in which visits alternate between telehealth and office.
Patient disparities that may limit the use of telehealth
Race and ethnicity is a major factor in telehealth use. Patients who are Black or Hispanic use telehealth services less often than patients who are White.32,33 A study that looked at patients with chronic conditions—hypertension and diabetes—that disproportionately affect Black and Hispanic patients found that patients in these populations with either of these conditions had a lower prevalence of Internet use when compared with White patients.34 However, subpopulations can vary in their usage. For example, a study in East Harlem, New York, found that Hispanic pregnant women used telehealth frequently for prenatal care and perceived the care as satisfactory.35
Age is also a significant variable in the adoption of telehealth, with pre-COVID-19 studies finding lower use of technology among older adults. However, a study performed at the University of Missouri during the first months of the pandemic found an increase in telehealth use in seniors,32 although the increase was in telephone use and not full video sessions.
Many patients in need of health care services may have older devices and/or low-speed or no Internet access; they also may lack the technical know-how to conduct a telehealth visit.4,36 For example, regardless of race or ethnicity, patients on government insurance (Medicaid and Medicare) have been shown to complete more telephone than video visits,37 underscoring the importance of telehealth practice flexibility and the need for increased technology support to decrease the digital divide. Even with adequate technological support and patient training, telehealth may be more complicated if patients have such comorbidities as hearing, visual, or cognitive impairment.31 Patients from a lower socioeconomic status may feel uncomfortable with providers seeing their home environment on video.38
Overall, incorporating telehealth for the care of older and/or vulnerable patients will present a unique set of challenges that organizations must address. Efforts must be made to understand the available technologies and patients’ comfort in using them. A hybrid model offering telehealth and in-office encounters may be the best solution.
Hernan Barenboim, PhD, KPC Health Group, 301 North San Jacinto Street, Hemet, CA 92543; [email protected]
Social distancing measures instituted during the COVID-19 pandemic challenged the usual way of operating in primary care. To continue delivering medical services, physicians had to transition quickly to forms of remote interaction with patients. Use of technology appeared to be the answer. And it gave clinicians the ability to do what many had long hoped for: offer patients the option of telehealth.
The terms telemedicine and telehealth have similar definitions and are commonly used interchangeably. We think most practices probably would have adopted telehealth earlier were it not for reimbursement barriers. In this article, we adopt the World Health Organization’s definition of telemedicine as: “The delivery of healthcare services, where distance is a critical factor, by all healthcare professionals using information and communication technologies for the exchange of valid information for the diagnosis, treatment, and prevention of disease and injuries, research and evaluation, and for the continuing education of healthcare providers, all in the interests of advancing the health of individuals and their communities.”1
To provide family medicine clinicians with evidence-based recommendations about telehealth, we conducted a critical review of the literature published through April 30, 2021. The scope of this review includes studies found using the PubMed and Google Scholar databases. In addition, we used the keywords “telehealth,” “telemedicine,” “family medicine,” and “primary care.” We divided this review into 6 sections, including focus areas on implementation in primary care, remote diagnostic accuracy, conditions lending themselves to telehealth, physician and patient perceptions, disparities in telehealth, and finally, the conclusions.
Telehealth implementation in primary care
Telehealth in various forms had been around for years before the pandemic, mainly in the form of commercial telehealth businesses. Telehealth was being used in rural and remote areas where it could be difficult to see a primary care provider—let alone a specialist. The family medicine department of the University of Colorado was an early adopter of telehealth and had navigated this transition since 2017, with clinical champions guiding the process. By 2019, 54% of their clinicians were conducting telehealth encounters.2
However, telehealth implementation elsewhere was not accepted so readily. Before the pandemic, a cross-sectional study of more than 1.1 million patients in Northern California showed that 86% preferred in-person care over video.3 Even as the pandemic began and social distancing measures were implemented, a quality improvement project at a family medicine residency clinic in Florida documented that clinicians still preferred telephone interviews despite the capacity for video visits.4 And many primary care systems were simply unprepared to adopt telehealth technologies.
With time, however, family physicians began to improvise using popular videoconferencing technologies (eg, Zoom) that were readily available and familiar to patients, and medical centers began to repurpose their existing videoconferencing systems.5 The Ohio State University Wexner Medical Center launched a virtual health initiative just before the pandemic struck, at which time fewer than 5% of patient visits were conducted through telehealth. Weeks later, nearly 93% of patient visits were offered through telehealth.6
Reimbursement. Another significant impediment to early telehealth uptake was the late reaction by the Centers for Medicare and Medicaid Services (CMS) in changing the payment system. Hectic expansion of telehealth in response to the crisis pointed to the lack of policies that supported primary care with payments based on outcomes rather than fee-for-service models.7 By the end of April 2020, CMS finally announced that video visits would be reimbursed at the same rate as in-person visits. However, telephone-only visits are still very limited in coverage, and appropriate codes should be verified with payers.
Continue to: Remote diagnosis comes with a caveat
Remote diagnosis comes with a caveat
Some primary care practices have found that images of skin lesions submitted by patients (usually by cell phone) suffice for accurate diagnosis in lieu of office visits.8 With chronic conditions, home-based remote monitoring of vital signs may assist in diagnosing and managing acute issues. More efficient triage of patients is increasingly possible with the receipt of still images or video files of concerning lesions (eg, burns, rash, chronic wounds) sent from smartphones alone9,10 or with devices attached to smartphones (eg, parent-managed otoscopes).11,12
Family physicians historically have relied on in-person visits for holistic assessment and diagnosis. Telehealth video visits have the potential to assist with this goal, but there are risks. For example, one patient cut her foot while swimming and the wound became infected.
Specific conditions usually suitable for telehealth evaluation
The pandemic helped us understand that some situations and conditions are better suited than others to coverage by telehealth. The National Ambulatory Medical Care Survey examined 850 million patient–physician encounters and found that 66% of all ambulatory primary care visits required in-office care,15 suggesting that about one-third of patient encounters could be treated via telehealth.
As an example, our southeastern Wisconsin urban clinic has about 20,000 office visits per year. We launched telehealth in March 2020 in direct response to the pandemic. Telehealth usage peaked at the beginning of the pandemic (FIGURE), fell gradually, hit a lower peak in November and December as COVID case counts increased, and then decreased again as our community changed from a “quarantine/lockdown” mentality to “opening up/back to new normal.

Some conditions can be managed favorably with the telehealth format:
Infectious diseases may be treatable remotely.16,17 Following an initial telehealth visit, the physician can evaluate and recommend further care.
Stable, chronic conditions. Telehealth can be used for stable, chronic conditions such as diabetes, chronic obstructive pulmonary disease, and heart failure when lab or imaging studies are not needed.18
Mental health. Telehealth can be useful in counseling and providing mental health and social support.18 Safeguards can be put in place to protect patient privacy in this setting.19
Behavioral change. Telehealth can be effective in providing support for patients actively trying to quit smoking or lose weight, and for caregivers. A physician who “checks in” can be a positive motivator and can promote a patient’s continued success.20
Continue to: Telehealth is less beneficial...
Telehealth is less beneficial when a physical exam is needed to assess pain, tenderness, strength, or other sensations. Office visits also are required for lab assays and imaging, as in periodic checks of A1C levels in patients with diabetes. As technology advances, home-based laboratory kits and sensors likely will change this picture. New patients may be better served through an initial office visit to develop the patient–physician relationship.
Visual assessment of conditions may be limited by telehealth depending on the quality of the devices used. For example, rashes may be difficult to assess given the clarity of the picture on the device and the ability to see only in 2D. There is still a need for more controlled trials to clarify which conditions can be evaluated and managed by telehealth and which ones need in-person care.21
Physician and patient perceptions of telehealth encounters
Research into family physicians’ perceptions of telehealth is scant. However, 3 studies published in 2021 reveal some advantages and challenges for telehealth adoption.
- A qualitative study found that physicians valued the increased access to care for some patients, changes to reimbursement practices not covered before, and the opportunity to see patients’ home environments.22 Disadvantages included an inability to examine the patient, problems with diagnostic accuracy, hindrances to developing personal connections, and the potential for burnout with on-demand care.22 The researchers suggested that telehealth might better serve to augment in-person care.
- A second study found that clinicians are satisfied with the use of telehealth in general. However, it also noted that the lack of physical examination could hinder accurate diagnosis and treatment.23
- A third study surveyed 109 family physicians, reinforcing the importance of physical exams and highlighting the lack of body language as another barrier.24
In addition, all 3 studies noted that video visits are typically briefer than in-person visits. Previous research predominantly done in specialty and mental health care showed that the benefits of telehealth for physicians include an increase in efficiency, reduced commute time, and improved work-life balance.25
Patient perspectives. Many patients have reported that they prefer telehealth because of lower costs, decreased travel time, and faster health care access.26,27 However, patients also have expressed concerns that the telehealth environment may reduce physician attention, can limit personal interaction (and impart a sense of being rushed), and lacks the physical examination that may be key to an adequate diagnosis.28
Continue to: A survey of 223 patients showed...
A survey of 223 patients showed that sicker patients choose in-person care because they want more in-depth visits with more attention to detail than healthier patients do.29 In a Veterans Affairs health care system qualitative study, patients voiced concerns about communicating with physicians via telehealth, including the potential for errors, less attention paid to their needs, audio difficulties, and challenges to establishing a physician–patient relationship.30 Some patients thought telehealth inhibited their personal expression or that the clinician was not attentive enough. These patient reports underscore the importance of patient–clinician relationships developed in person.31 The perceived level of complexity involved in a visit appears to be an essential factor in a patient opting for telehealth—or not.
In light of these known physician and patient perspectives, it seems wise to develop a hybrid model approach in which visits alternate between telehealth and office.
Patient disparities that may limit the use of telehealth
Race and ethnicity is a major factor in telehealth use. Patients who are Black or Hispanic use telehealth services less often than patients who are White.32,33 A study that looked at patients with chronic conditions—hypertension and diabetes—that disproportionately affect Black and Hispanic patients found that patients in these populations with either of these conditions had a lower prevalence of Internet use when compared with White patients.34 However, subpopulations can vary in their usage. For example, a study in East Harlem, New York, found that Hispanic pregnant women used telehealth frequently for prenatal care and perceived the care as satisfactory.35
Age is also a significant variable in the adoption of telehealth, with pre-COVID-19 studies finding lower use of technology among older adults. However, a study performed at the University of Missouri during the first months of the pandemic found an increase in telehealth use in seniors,32 although the increase was in telephone use and not full video sessions.
Many patients in need of health care services may have older devices and/or low-speed or no Internet access; they also may lack the technical know-how to conduct a telehealth visit.4,36 For example, regardless of race or ethnicity, patients on government insurance (Medicaid and Medicare) have been shown to complete more telephone than video visits,37 underscoring the importance of telehealth practice flexibility and the need for increased technology support to decrease the digital divide. Even with adequate technological support and patient training, telehealth may be more complicated if patients have such comorbidities as hearing, visual, or cognitive impairment.31 Patients from a lower socioeconomic status may feel uncomfortable with providers seeing their home environment on video.38
Overall, incorporating telehealth for the care of older and/or vulnerable patients will present a unique set of challenges that organizations must address. Efforts must be made to understand the available technologies and patients’ comfort in using them. A hybrid model offering telehealth and in-office encounters may be the best solution.
Hernan Barenboim, PhD, KPC Health Group, 301 North San Jacinto Street, Hemet, CA 92543; [email protected]
1. WHO. A health telematics policy: in support of WHO’s Health-for-All strategy for global health development. 1997. Accessed February 8, 2023. https://apps.who.int/iris/bitstream/handle/10665/63857/WHO_DGO_98.1.pdf?sequence=1&isAllowed=y
2. Knierim K, Palmer C, Kramer ES, et al. Lessons learned during COVID-19 that can move telehealth in primary care forward. J Am Board Fam Med. Supplement 2021;34(suppl):S196-S202. doi: 10.3122/jabfm.2021.S1.200419
3. Reed ME, Huang J, Graetz I, et al. Patient characteristics associated with choosing a telemedicine visit vs office visit with the same primary care clinicians. JAMA Netw Open. 2020;3:e205873. doi: 10.1001/jamanetworkopen.2020.5873
4. Silver SL, Lewis MN, Ledford CJ. A stepwise transition to telemedicine in response to COVID-19. J Am Board Fam Med. 2021;34(suppl):S152-S161. doi: 10.3122/jabfm.2021.S1.200358
5. Hron JD, Parsons CR, Williams LA, et al. Rapid implementation of an inpatient telehealth program during the COVID-19 pandemic. Appl Clin Inform. 2020;3:452-459. doi: 10.1055/s-0040-1713635
6. Olayiwola JN, Magaña C, Harmon A, et al. Telehealth as a bright spot of the COVID-19 pandemic: recommendations from the virtual frontlines (“Frontweb”). JMIR Public Health Surveill. 2020;6:e19045. doi: 10.2196/19045
7. Gausvik C, Jabbarpour Y. COVID-19 timeline: Centers for Medicare and Medicaid Services (CMS) changes and primary care support were not enough to prevent practice losses. J Am Board Fam Med. 2021;34(suppl):S7-S9. doi: 10.3122/jabfm.2021.S1.200305
8. Marin-Gomez FX, Vidal-Alaball J, Poch PR, et al. Diagnosis of skin lesions using photographs taken with a mobile phone: an online survey of primary care physicians. J Prim Care Community Health. 2020;11:2150132720937831. doi: 10.1177/2150132720937831
9. Garber RN, Garcia E, Goodwin CW, et al. (2020). Pictures do influence the decision to transfer: outcomes of a telemedicine program serving an eight-state rural population. J Burn Care Res. 2020;41:690-694. doi: 10.1093/jbcr/iraa017
10. Felix F, Greenblatt M, Shin L. Saving limbs in the time of COVID. 2020. Accessed February 8, 2023. https://podiatrym.com/pdf/2020/7/FelixGreenblattShin820web.pdf
11. Erkkola-Anttinen N, Irjala H, Laine MK, et al. Smartphone otoscopy performed by parents. Telemed J E Health. 2019;25:477-484. doi: 10.1089/tmj.2018.0062
12. Verzantvoort NC, Teunis T, Verheij TJ, et al. Self-triage for acute primary care via a smartphone application: practical, safe and efficient? PLoS One. 2018;13:e0199284. doi: 10.1371/journal.pone.0199284
13. Hickner J. When patients don’t get the care they should. J Fam Pract. 2020;69:427.
14. Pappan N, Benkhadra R, Papincak D, et al. Values and limits of telemedicine: a case report. SN Compr Clin Med. 2021;3:317-319. doi: 10.1007/s42399-020-00725-y
15. Jabbarpour Y, Jetty A, Westfall M, et al. Not telehealth: which primary care visits need in-person care? J Am Board Fam Med. 2021;34(suppl):S162-S169. doi: 10.3122/jabfm.2021.S1.200247
16. Parmar P, Mackie D, Varghese S, et al. Use of telemedicine technologies in the management of infectious diseases: a review. Clin Infect Dis. 2015;60:1084-1094. doi: 10.1093/cid/ciu1143
17. Young JD, Abdel-Massih R, Herchline T, et al. Infectious Diseases Society of America position statement on Telehealth and Telemedicine as Applied to the Practice of Infectious Diseases. Clin Infect Dis. 2019;68:1437-1443. doi: 10.1093/cid/ciy907
18. ARHQ. Telehealth: mapping the evidence for patient outcomes from systematic reviews. 2016. Accessed March 27, 2023. https://effectivehealthcare.ahrq.gov/sites/default/files/pdf/telehealth_technical-brief.pdf
19. Lustgarten SD, Garrison YL, Sinnard MT, et al. Digital privacy in mental healthcare: current issues and recommendations for technology use. Curr Opin Psychol. 2020;36:25-31. doi: 10.1016/j.copsyc.2020.03.012
20. Baird A, Xia Y, Cheng Y. Consumer perceptions of telehealth for mental health or substance abuse: a Twitter-based topic modeling analysis. JAMIA Open. 2022;5:ooac028. doi: 10.1093/jamiaopen/ooac028
21. Flumignan CD, da Rocha AP, Pinto AC, et al. What do Cochrane systematic reviews say about telemedicine for healthcare? Sao Paulo Med J. 2019;137:184-192. doi: 10.1590/1516-3180.0177240419
22. Gomez T, Anaya YB, Shih KJ, et al. A qualitative study of primary care physicians’ experiences with telemedicine during COVID-19. J Am Board Fam Med. 2021;34(suppl):S61-S70. doi: 10.3122/jabfm.2021.S1.200517
23. Malliaras P, Merolli M, Williams CM, et al. ‘It’s not hands-on therapy, so it’s very limited’: telehealth use and views among allied health clinicians during the coronavirus pandemic. Musculoskelet Sci Pract. 2021;52:102340. doi: 10.1016/j.msksp.2021.102340
24. Gold KJ, Laurie AR, Kinney DR, et al. Video visits: family physician experiences with uptake during the COVID-19 pandemic. Fam Med. 53:207-210. doi: 10.22454/FamMed.2021.613099
25. Björndell C, Premberg A. Physicians’ experiences of video consultation with patients at a public virtual primary care clinic: a qualitative interview study. Scand J Prim Health Care. 2021;39:67-76. doi: 10.1080/02813432.2021.1882082
26. Powell RE, Henstenburg JM, Cooper G, et al. Patient perceptions of telehealth primary care video visits. Ann Fam Med. 2017;15:225-229. doi: 10.1370/afm.2095
27. Imlach F, McKinlay E, Middleton L, et al. Telehealth consultations in general practice during a pandemic lockdown: survey and interviews on patient experiences and preferences. BMC Fam Pract. 2020;21:1-14. doi: 10.1186/s12875-020-01336-1
28. Gordon HS, Solanki P, Bokhour BG, et al. “I’m not feeling like I’m part of the conversation” patients’ perspectives on communicating in clinical video telehealth visits. J Gen Intern Med. 2020;35:1751-1758. doi: 10.1007/s11606-020-05673-w
29. Volcy J, Smith W, Mills K, et al. Assessment of patient and provider satisfaction with the change to telehealth from in-person visits at an academic safety net institution during the COVID-19 pandemic. J Am Board Fam Med. 2021;34(suppl):S71-S76. doi: 10.3122/jabfm.2021.S1.200393
30. Gopal RK, Solanki P, Bokhour BG, et al. Provider, staff, and patient perspectives on medical visits using clinical video telehealth: a foundation for educational initiatives to improve medical care in telehealth. J Nurse Pract. 2021;17:582-587. doi: 10.1016/j.nurpra.2021.02.020
31. Edgoose JY. Exploring the face-to-face: revisiting patient-doctor relationships in a time of expanding telemedicine. J Am Board Fam Med. 2021;34(suppl):S252-S254. doi: 10.3122/jabfm.2021.S1.200398
32. Pierce RP, Stevermer JJ. Disparities in use of telehealth at the onset of the COVID-19 public health emergency. J Telemed Telecare. 2023;29:3-9. doi: 10.1177/1357633X20963893
33. Lame M, Leyden D, Platt SL. Geocode maps spotlight disparities in telehealth utilization during the COVID-19 pandemic in New York City. Telemed J E Health. 2021;27:251-253. doi: 10.1089/tmj.2020.0297
34. Jain V, Al Rifai M, Lee MT, et al. Racial and geographic disparities in internet use in the US among patients with hypertension or diabetes: implications for telehealth in the era of COVID-19. Diabetes Care. 2021;44:e15-e17. doi: 10.2337/dc20-2016
35. Futterman I, Rosenfeld E, Toaff M, et al. Addressing disparities in prenatal care via telehealth during COVID-19: prenatal satisfaction survey in East Harlem. Am J Perinatol. 2021;38:88-92. doi: 10.1055/s-0040-1718695
36. Wegermann K, Wilder JM, Parish A, et al. Racial and socioeconomic disparities in utilization of telehealth in patients with liver disease during COVID-19. Dig Dis Sci. 2022;67:93-99. doi: 10.1007/s10620-021-06842-5.
37. ASPE. National survey trends in telehealth use in 2021: disparities in utilization and audio vs. video services. Issue brief: February 21, 2022. Accessed March 27, 2023. https://aspe.hhs.gov/sites/default/files/documents/4e1853c0b4885112b2994680a58af9ed/telehealth-hps-ib.pdf
38. Ukoha EP, Davis K, Yinger M, et al. Ensuring equitable implementation of telemedicine in perinatal care. Obstet Gynecol. 2021;137:487-492. doi: 10.1097/AOG.0000000000004276
1. WHO. A health telematics policy: in support of WHO’s Health-for-All strategy for global health development. 1997. Accessed February 8, 2023. https://apps.who.int/iris/bitstream/handle/10665/63857/WHO_DGO_98.1.pdf?sequence=1&isAllowed=y
2. Knierim K, Palmer C, Kramer ES, et al. Lessons learned during COVID-19 that can move telehealth in primary care forward. J Am Board Fam Med. Supplement 2021;34(suppl):S196-S202. doi: 10.3122/jabfm.2021.S1.200419
3. Reed ME, Huang J, Graetz I, et al. Patient characteristics associated with choosing a telemedicine visit vs office visit with the same primary care clinicians. JAMA Netw Open. 2020;3:e205873. doi: 10.1001/jamanetworkopen.2020.5873
4. Silver SL, Lewis MN, Ledford CJ. A stepwise transition to telemedicine in response to COVID-19. J Am Board Fam Med. 2021;34(suppl):S152-S161. doi: 10.3122/jabfm.2021.S1.200358
5. Hron JD, Parsons CR, Williams LA, et al. Rapid implementation of an inpatient telehealth program during the COVID-19 pandemic. Appl Clin Inform. 2020;3:452-459. doi: 10.1055/s-0040-1713635
6. Olayiwola JN, Magaña C, Harmon A, et al. Telehealth as a bright spot of the COVID-19 pandemic: recommendations from the virtual frontlines (“Frontweb”). JMIR Public Health Surveill. 2020;6:e19045. doi: 10.2196/19045
7. Gausvik C, Jabbarpour Y. COVID-19 timeline: Centers for Medicare and Medicaid Services (CMS) changes and primary care support were not enough to prevent practice losses. J Am Board Fam Med. 2021;34(suppl):S7-S9. doi: 10.3122/jabfm.2021.S1.200305
8. Marin-Gomez FX, Vidal-Alaball J, Poch PR, et al. Diagnosis of skin lesions using photographs taken with a mobile phone: an online survey of primary care physicians. J Prim Care Community Health. 2020;11:2150132720937831. doi: 10.1177/2150132720937831
9. Garber RN, Garcia E, Goodwin CW, et al. (2020). Pictures do influence the decision to transfer: outcomes of a telemedicine program serving an eight-state rural population. J Burn Care Res. 2020;41:690-694. doi: 10.1093/jbcr/iraa017
10. Felix F, Greenblatt M, Shin L. Saving limbs in the time of COVID. 2020. Accessed February 8, 2023. https://podiatrym.com/pdf/2020/7/FelixGreenblattShin820web.pdf
11. Erkkola-Anttinen N, Irjala H, Laine MK, et al. Smartphone otoscopy performed by parents. Telemed J E Health. 2019;25:477-484. doi: 10.1089/tmj.2018.0062
12. Verzantvoort NC, Teunis T, Verheij TJ, et al. Self-triage for acute primary care via a smartphone application: practical, safe and efficient? PLoS One. 2018;13:e0199284. doi: 10.1371/journal.pone.0199284
13. Hickner J. When patients don’t get the care they should. J Fam Pract. 2020;69:427.
14. Pappan N, Benkhadra R, Papincak D, et al. Values and limits of telemedicine: a case report. SN Compr Clin Med. 2021;3:317-319. doi: 10.1007/s42399-020-00725-y
15. Jabbarpour Y, Jetty A, Westfall M, et al. Not telehealth: which primary care visits need in-person care? J Am Board Fam Med. 2021;34(suppl):S162-S169. doi: 10.3122/jabfm.2021.S1.200247
16. Parmar P, Mackie D, Varghese S, et al. Use of telemedicine technologies in the management of infectious diseases: a review. Clin Infect Dis. 2015;60:1084-1094. doi: 10.1093/cid/ciu1143
17. Young JD, Abdel-Massih R, Herchline T, et al. Infectious Diseases Society of America position statement on Telehealth and Telemedicine as Applied to the Practice of Infectious Diseases. Clin Infect Dis. 2019;68:1437-1443. doi: 10.1093/cid/ciy907
18. ARHQ. Telehealth: mapping the evidence for patient outcomes from systematic reviews. 2016. Accessed March 27, 2023. https://effectivehealthcare.ahrq.gov/sites/default/files/pdf/telehealth_technical-brief.pdf
19. Lustgarten SD, Garrison YL, Sinnard MT, et al. Digital privacy in mental healthcare: current issues and recommendations for technology use. Curr Opin Psychol. 2020;36:25-31. doi: 10.1016/j.copsyc.2020.03.012
20. Baird A, Xia Y, Cheng Y. Consumer perceptions of telehealth for mental health or substance abuse: a Twitter-based topic modeling analysis. JAMIA Open. 2022;5:ooac028. doi: 10.1093/jamiaopen/ooac028
21. Flumignan CD, da Rocha AP, Pinto AC, et al. What do Cochrane systematic reviews say about telemedicine for healthcare? Sao Paulo Med J. 2019;137:184-192. doi: 10.1590/1516-3180.0177240419
22. Gomez T, Anaya YB, Shih KJ, et al. A qualitative study of primary care physicians’ experiences with telemedicine during COVID-19. J Am Board Fam Med. 2021;34(suppl):S61-S70. doi: 10.3122/jabfm.2021.S1.200517
23. Malliaras P, Merolli M, Williams CM, et al. ‘It’s not hands-on therapy, so it’s very limited’: telehealth use and views among allied health clinicians during the coronavirus pandemic. Musculoskelet Sci Pract. 2021;52:102340. doi: 10.1016/j.msksp.2021.102340
24. Gold KJ, Laurie AR, Kinney DR, et al. Video visits: family physician experiences with uptake during the COVID-19 pandemic. Fam Med. 53:207-210. doi: 10.22454/FamMed.2021.613099
25. Björndell C, Premberg A. Physicians’ experiences of video consultation with patients at a public virtual primary care clinic: a qualitative interview study. Scand J Prim Health Care. 2021;39:67-76. doi: 10.1080/02813432.2021.1882082
26. Powell RE, Henstenburg JM, Cooper G, et al. Patient perceptions of telehealth primary care video visits. Ann Fam Med. 2017;15:225-229. doi: 10.1370/afm.2095
27. Imlach F, McKinlay E, Middleton L, et al. Telehealth consultations in general practice during a pandemic lockdown: survey and interviews on patient experiences and preferences. BMC Fam Pract. 2020;21:1-14. doi: 10.1186/s12875-020-01336-1
28. Gordon HS, Solanki P, Bokhour BG, et al. “I’m not feeling like I’m part of the conversation” patients’ perspectives on communicating in clinical video telehealth visits. J Gen Intern Med. 2020;35:1751-1758. doi: 10.1007/s11606-020-05673-w
29. Volcy J, Smith W, Mills K, et al. Assessment of patient and provider satisfaction with the change to telehealth from in-person visits at an academic safety net institution during the COVID-19 pandemic. J Am Board Fam Med. 2021;34(suppl):S71-S76. doi: 10.3122/jabfm.2021.S1.200393
30. Gopal RK, Solanki P, Bokhour BG, et al. Provider, staff, and patient perspectives on medical visits using clinical video telehealth: a foundation for educational initiatives to improve medical care in telehealth. J Nurse Pract. 2021;17:582-587. doi: 10.1016/j.nurpra.2021.02.020
31. Edgoose JY. Exploring the face-to-face: revisiting patient-doctor relationships in a time of expanding telemedicine. J Am Board Fam Med. 2021;34(suppl):S252-S254. doi: 10.3122/jabfm.2021.S1.200398
32. Pierce RP, Stevermer JJ. Disparities in use of telehealth at the onset of the COVID-19 public health emergency. J Telemed Telecare. 2023;29:3-9. doi: 10.1177/1357633X20963893
33. Lame M, Leyden D, Platt SL. Geocode maps spotlight disparities in telehealth utilization during the COVID-19 pandemic in New York City. Telemed J E Health. 2021;27:251-253. doi: 10.1089/tmj.2020.0297
34. Jain V, Al Rifai M, Lee MT, et al. Racial and geographic disparities in internet use in the US among patients with hypertension or diabetes: implications for telehealth in the era of COVID-19. Diabetes Care. 2021;44:e15-e17. doi: 10.2337/dc20-2016
35. Futterman I, Rosenfeld E, Toaff M, et al. Addressing disparities in prenatal care via telehealth during COVID-19: prenatal satisfaction survey in East Harlem. Am J Perinatol. 2021;38:88-92. doi: 10.1055/s-0040-1718695
36. Wegermann K, Wilder JM, Parish A, et al. Racial and socioeconomic disparities in utilization of telehealth in patients with liver disease during COVID-19. Dig Dis Sci. 2022;67:93-99. doi: 10.1007/s10620-021-06842-5.
37. ASPE. National survey trends in telehealth use in 2021: disparities in utilization and audio vs. video services. Issue brief: February 21, 2022. Accessed March 27, 2023. https://aspe.hhs.gov/sites/default/files/documents/4e1853c0b4885112b2994680a58af9ed/telehealth-hps-ib.pdf
38. Ukoha EP, Davis K, Yinger M, et al. Ensuring equitable implementation of telemedicine in perinatal care. Obstet Gynecol. 2021;137:487-492. doi: 10.1097/AOG.0000000000004276
PRACTICE RECOMMENDATIONS
› Consider using telehealth encounters for diagnosing and treating infectious diseases and for monitoring stable chronic conditions. C
› Consider telehealth “check-ins” to encourage patients working on behavioral change, such as smoking cessation. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Patient with newly diagnosed type 2 diabetes? Remember these steps
Nearly 40 antihyperglycemic agents have been approved by the US Food and Drug Administration (FDA) since the approval of human insulin in 1982.1 In addition, existing antihyperglycemic medications are constantly gaining FDA approval for new indications for common type 2 diabetes (T2D) comorbidities. For example, in addition to their glycemic benefits, the sodium-glucose cotransporter-2 (SGLT2) inhibitors have been approved for use in patients with T2D and established atherosclerotic cardiovascular disease (ASCVD) to reduce the risk for major adverse cardiovascular events (MACE; canagliflozin), risk for hospitalization for heart failure (dapagliflozin), and cardiovascular death (empagliflozin).2-4
The plethora of new agents and new data for existing agents, coupled with the annual release of guidelines from the American Diabetes Association (ADA) and practice recommendations from several other professional organizations,5-7 make it challenging for family physicians to stay current and provide the most up-to-date, evidence-based care. In this article, we provide advice on how to approach the screening, diagnosis, and evaluation of T2D, and on how to manage newly diagnosed T2D.
Screening, Dx, and evaluation: A quick review
Screening
Screening recommendations vary among professional organizations (TABLE 15,6,8). The US Preventive Services Task Force (USPSTF) recommends screening adults ages 35 to 70 years who are overweight or obese. Clinicians also can consider screening patients with a higher risk for diabetes.5 The ADA suggests screening all adults starting at 35 years, regardless of risk factors.8 Asymptomatic adults of any age with overweight or obesity and 1 or more risk factors should be screened.8
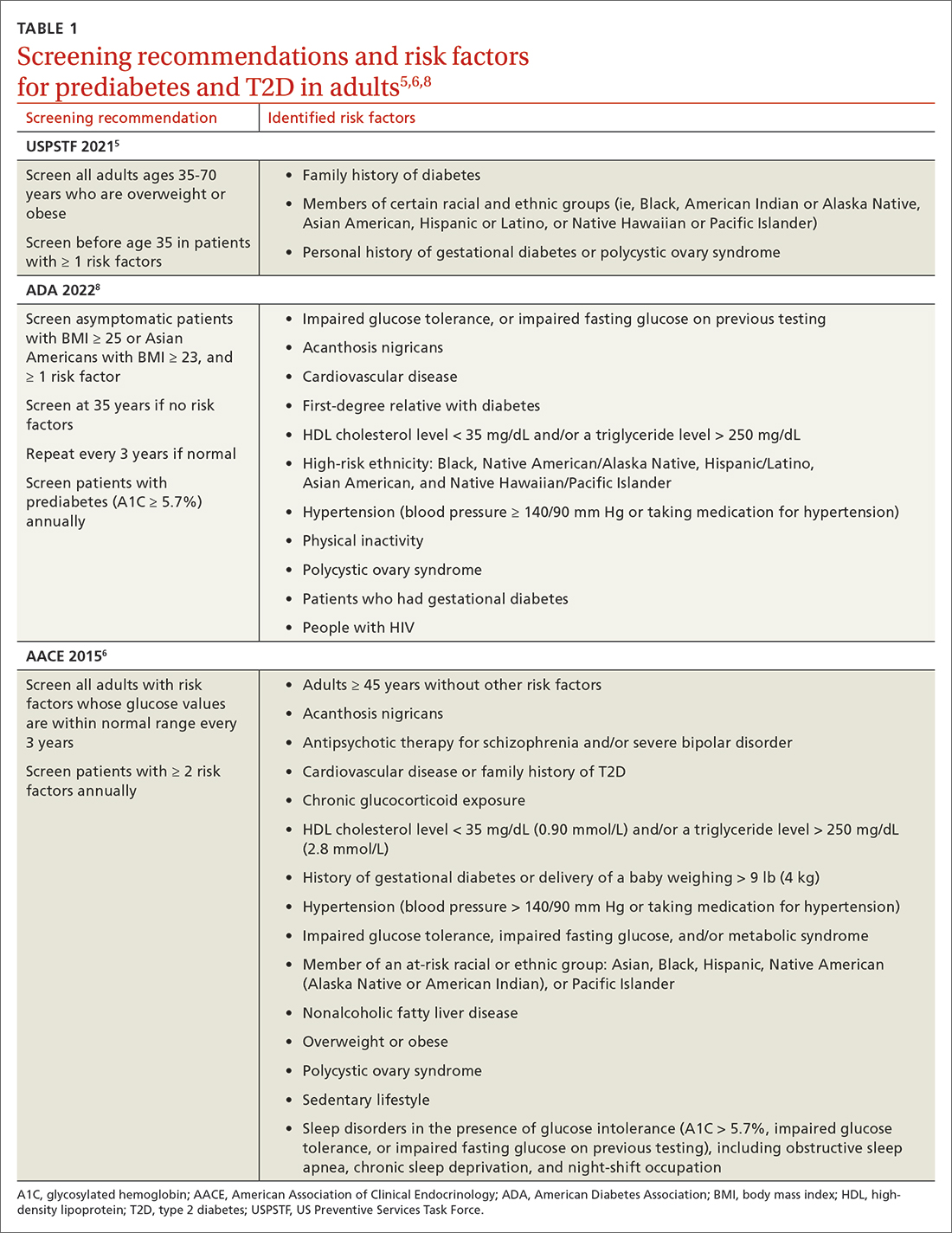
Making the diagnosis
The initial diagnosis of diabetes can be made by a fasting plasma glucose level ≥ 126 mg/dL (7.0 mmol/L); a 2-hour plasma glucose level ≥ 200 mg/dL (11.0 mmol/L) following an oral glucose tolerance test; or an A1C level ≥ 6.5%. Prioritize lab-drawn A1C measurements over point-of-care tests to diagnose T2D. In patients with classic symptoms of hyperglycemia, a random plasma glucose level ≥ 200 mg/dL (11.0 mmol/L) is also diagnostic. Generally, these tests are considered equally appropriate in screening for diabetes and may be used to detect prediabetes. In the absence of clear symptoms of hyperglycemia, the diagnosis of diabetes requires 2 abnormal screening test results, either via 1 blood sample (such as an abnormal A1C and glucose) or 2 separate blood samples of the same test. Further evaluation is advised if there is discordance between the 2 samples.8
Extended evaluations
Patients with newly diagnosed T2D require a thorough evaluation for comorbidities and complications of diabetes. Refer patients to an ophthalmologist for a dilated eye examination, with subsequent exams occurring every 1 to 2 years.6,9 Additional referrals for diabetes education, family planning for women of reproductive age, and dental, social, or mental health services may be clinically appropriate.9
Setting goals for glycemic control
Glycemic control is commonly monitored by the A1C level and by blood glucose monitoring either through traditional point-of-care glucometers or continuous glucose monitors (CGMs).10 Generally, CGMs provide more glycemic data than traditional glucometers and may cue patients to choose healthier dietary options and engage in physical exercise.11 Patients with T2D who use CGMs exhibit lower A1Cs, greater time in glycemic range, and reduced hypoglycemic episodes.11 Generally, CGMs are reserved for patients with type 1 diabetes and patients with T2D who use multiple daily injections, subcutaneous insulin infusions, or basal insulin only.12 Most professional organizations recommend that clinicians consider patient-specific factors to set individualized glycemic goals.6,10,13,14 For example, more stringent glycemic goals could be pursued for patients with longer life expectancy, shorter disease duration, absence of complications (eg, nephropathy, neuropathy, or cardiovascular disease), fewer comorbid conditions, lower hypoglycemia risk, or higher cognitive function.6
More specific A1C goals vary by professional organization. For nonpregnant adults, the ADA recommends an A1C goal of < 7% and a preprandial blood glucose level of 80 to 130 mg/dL (4.4-7.2 mmol/L).10 However, a lower A1C goal may be appropriate if it can be attained safely without causing hypoglycemia or other adverse effects.10 The AACE suggests an A1C goal of ≤ 6.5% and a fasting blood glucose level of < 110 mg/dL when it can be achieved safely.6 More stringent A1C goals may reduce long-term micro- and macrovascular complications—especially in patients with newly diagnosed T2D.10 While older studies such as the ACCORD trial found increased mortality in groups with more stringent glycemic targets, they did not include newer agents (SGLT2 inhibitors or glucagon-like peptide-1 [GLP-1] receptor agonists) that reduce cardiovascular events by mechanisms outside their glycemic-lowering effect. With these newer agents, more aggressive A1C goals can be targeted safely in select patients, particularly those with long life expectancy.10 Both the ADA and AACE recommend a less stringent A1C goal of 7% to 8% for patients with limited life expectancy or risks (eg, a history of hypoglycemia) that outweigh expected benefits.6,10
Continue to: Lifestyle modifications
Lifestyle modifications: As important as medication
Nutrition
The energy-dense Western diet, combined with sedentary behavior, are thought to be a primary cause of T2D.15 Therefore, include lifestyle modifications in the initial management of newly diagnosed T2D. Diets that replace carbohydrates with saturated and trans fats are related to increased mortality in patients with T2D.16 Increased consumption of vegetables, fruits, legumes, nuts, fish, cereal, and oils reduces concentrations of saturated and trans fats and increases dietary intake of monounsaturated fatty acids, fiber, antioxidants, and polyphenols.17
Increasing the intake of fiber, an undigestible carbohydrate, offers numerous benefits in T2D management. High-fiber diets can help regulate blood sugar and lipid levels, increase satiety, reduce inflammation, aid in weight management, and reduce premature mortality.18 Insoluble fiber, found in foods such as whole wheat flour, nuts, and cauliflower, helps food pass more quickly through the stomach and intestines and adds bulk to stool. Soluble fiber, found in foods such as chickpeas, lentils, and Brussels sprouts, absorbs water and forms a gel-like substance that protects nutrients from digestive enzymes and slows down digestion. The result is a more gradual rise in postprandial glucose levels and improved insulin sensitivity.19 Dietary fiber may produce short-chain fatty acids which in turn activate incretin secretion and stimulate a glucose-dependent release of insulin from the pancreas.20
Simple dietary substitutions, such as whole grains and legumes for white rice, can reduce fasting blood glucose and A1C levels.21 In a randomized controlled trial (RCT), increasing whole grain oat intake improved measures of glycemic control, reducing A1C by 1% at 1-year follow-up.19 Encourage patients with T2D to increase consumption of high-fiber foods and replace animal fats and refined grains with vegetable fats (eg, nuts, avocados, olives). Nutritional therapies should be individualized, taking into account personal preferences and cultural customs.22 Nutritional habits may be based on race/ethnicity, religion/spirituality, or even the city in which an individual resides. Nutrition recommendations should account for these differences as well as access to healthy foods. For instance, ethnic groups whose dietary patterns include tortillas could be counseled to choose high-fiber options such as corn instead of flour tortillas and to incorporate vegetables in place of high-fat foods. Additionally, ethnic groups who favor using animal fats in foods such as greens could be advised on ways to add flavor to vegetables without adding saturated fats. Taking this approach may lessen barriers to change and increase ability to make dietary modifications.23
Exercise
Encourage all patients with T2D to exercise regularly. The atherosclerotic plaques found in patients with T2D have increased inflammatory properties and result in worse cardiovascular outcomes compared with plaques in individuals without T2D.24 Regular exercise reduces levels of pro-inflammatory markers—C-reactive protein, interleukin (IL)-6, and tumor necrosis factor alpha—and increases levels of anti-inflammatory markers (IL-4 and IL-10).24 Regular exercise can improve body composition, physical fitness, lipid and glucose metabolism, and insulin sensitivity.25,26
A meta-analysis of RCTs demonstrated that structured exercise > 150 minutes per week resulted in A1C reductions of 0.89%,27 which is comparable to the effect of many oral antihyperglycemic medications.26 The Health Benefits of Aerobic and Resistance Training in individuals with T2D (HART-D) and Diabetes Aerobic and Resistance Exercise (DARE) studies demonstrated that combining endurance and resistance training was superior for improving glycemic control, cardiorespiratory fitness, and body composition, than using either type of training alone.25 Both the American College of Sports Medicine (ACSM) and the ADA recommend that adults engage in at least 150 total minutes of moderate-intensity aerobic activity per week and resistance training 2 to 3 times weekly.26 ACSM defines moderate-intensity exercise as 65% to 75% of maximal heart rate, a rating of perceived exertion of 3 to 4, or a step rate of 100 steps per minute.28
Continue to: Because of their longitudinal relationships...
Because of their longitudinal relationships with patients, family physicians are in an optimal position to assess a patient’s physical capacity level and provide individualized counseling. Several systematic reviews have demonstrated that counseling on exercise increases patients’ participation in physical activity.29 Encourage your patients with T2D to exercise regularly, considering each individual’s ability to engage in physical activity.
Weight loss
Include weight management in the initial treatment of patients with newly diagnosed T2D. Weight loss decreases hepatic glucose production and increases peripheral insulin sensitivity and insulin secretion.30 Moderate decreases in weight (5%-10%) can reduce complications related to diabetes, and sustained significant weight loss (> 10%) can potentially cause T2D remission (A1C < 6.5% after stopping diabetes medications).31,32
Diabetes self-management education supports patients by giving them tools for making and maintaining lifestyle changes. Understanding individual barriers to change and addressing these during motivational interviews is important. Through a qualitative interview study, participants in a diabetes self-management program revealed 4 factors that motivated them to maintain lifestyle changes: support from others, experiencing the impact of the changes they made, fear of T2D complications, and forming new habits.33 Family physicians are key in helping patients acquire knowledge and support to make the lifestyle modifications needed to manage newly diagnosed T2D.
Individualized pharmacotherapy considerations
For decades, the initial pharmacotherapeutic regimen for patients with newly diagnosed T2D considered the patient’s baseline A1C as a major driver for therapy. Metformin has been the mainstay in T2D treatment due to its clinical efficacy, minimal risk for hypoglycemia, and low cost. Regardless of the regimen, pharmacotherapy should be initiated at the time of T2D diagnosis in conjunction with the aforementioned lifestyle modifications.34
When selecting pharmacotherapy, practice guidelines recommend considering the efficacy and adverse effects of medications, patient-specific comorbidities, adherence, cost, and a patient’s lifestyle factors.34 Drug classes with pertinent information are listed in TABLE 2.34-54 After starting medication, monitor the A1C level every 3 months to determine whether therapy should be intensified. Patients should have their labs drawn ahead of the quarterly visit, or point-of-care measurements may be used to facilitate in-person patient–provider discussions.
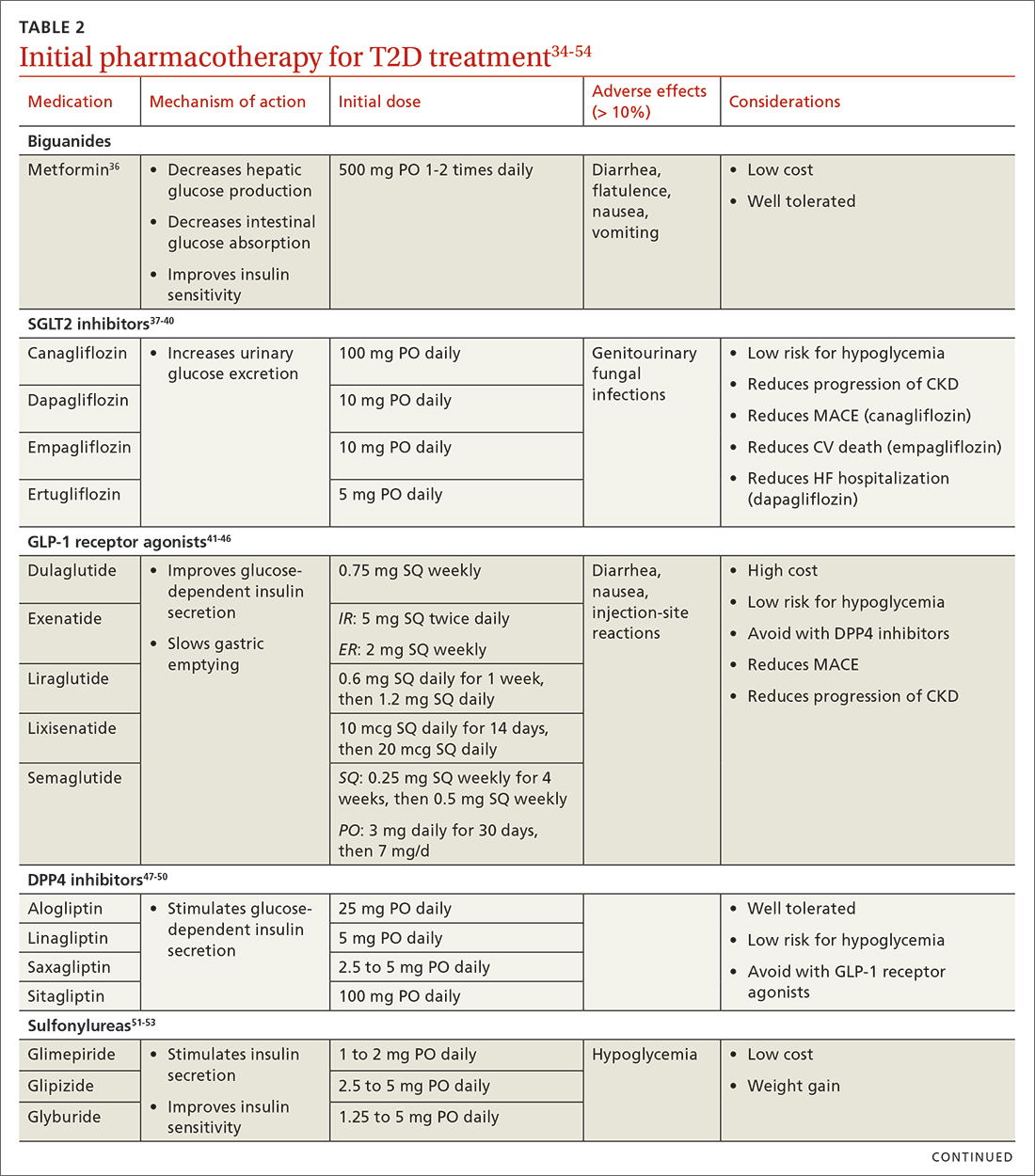
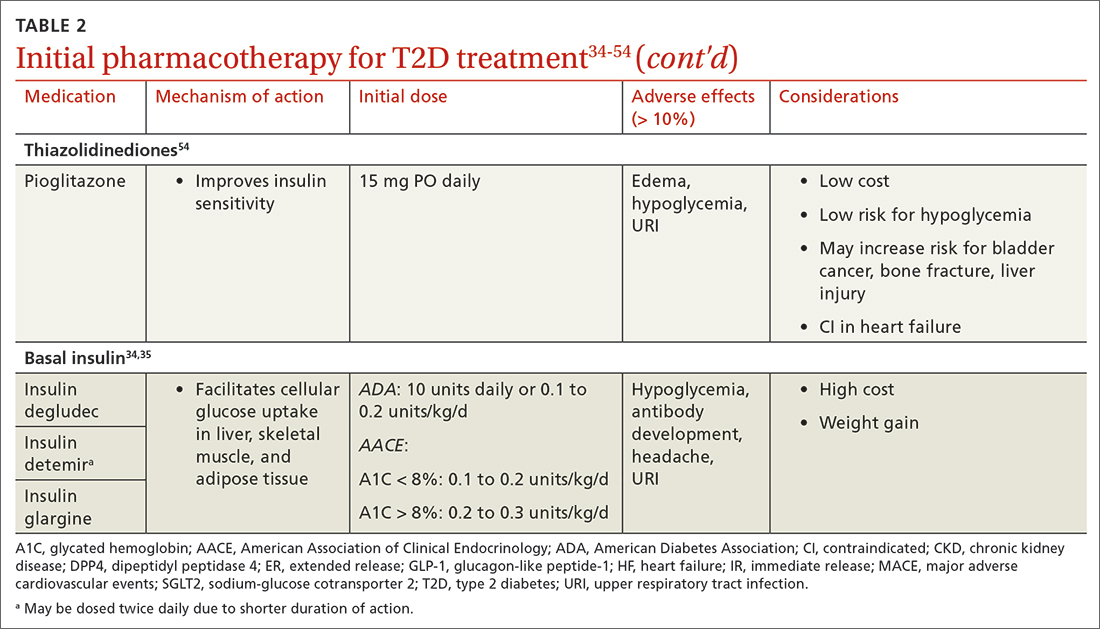
Continue to: Consider patient-specific factors when starting pharmacotherapy
Consider patient-specific factors when starting pharmacotherapy
ASCVD. Regardless of baseline glycemic control, offer patients who have ASCVD, or who are at high risk for it, an SGLT2 inhibitor (canagliflozin, dapagliflozin, or empagliflozin) or a long-acting GLP-1 receptor agonist (dulaglutide, liraglutide, or semaglutide).34,35 SGLT2 inhibitors reduced the risk for MACE by 11% in patients with established ASCVD.55 They also reduced a composite outcome of cardiovascular death or hospitalization for heart failure by 23% in patients with or without ASCVD or heart failure at baseline.55 GLP-1 receptor agonists offer a similar reduction in MACE to SGLT2 inhibitors, but they do not have significant effects in heart failure.56 Thiazolidinediones (TZDs), saxagliptin, and alogliptin should be avoided in patients with heart failure.57 TZDs may reduce the risk for recurrent stroke in patients with T2D.58
Chronic kidney disease (CKD). As with ASCVD, prioritize SGLT2 inhibitors and GLP-1 receptor agonists in patients with CKD. While both classes reduced the risk for progression of kidney disease such as macroalbuminuria, SGLT2 inhibitors offer additional benefits in their reduction of the worsening of estimated glomerular filtration rate, end-stage kidney disease, and renal death.56
Obesity. Consider the effect of each drug class on weight when making initial treatment choices, taking special care to minimize weight gain and potentially promote weight loss.34 The ADA prefers GLP-1 receptor agonists, but also suggests SGLT2 inhibitors in these patients. While all GLP-1 receptor agonists have an impact on weight, weekly subcutaneous semaglutide offers the most pronounced weight loss of 2 to 7 kg over 56 weeks.59 SGLT2 inhibitors promote sustainable weight loss to a lesser degree, contributing to an average loss of 3 kg at 2 years.60 Weight gain is common in patients taking sulfonylureas (2.01-2.3 kg)31 and insulin (3-9 kg weight gain in the first year)61 and should be avoided in patients with T2D and obesity.34
Hypoglycemia risk. In addition to counseling patients on hypoglycemia management and prescribing glucagon rescue kits, offer medications with no or very low risk for hypoglycemia (eg, GLP-1 receptor agonists, SGLT2 inhibitors, dipeptidyl peptidase-4 inhibitors, and TZDs). Generally, avoid insulin and sulfonylureas in patients in whom hypoglycemia is a major concern (eg, older adults, individuals with labile blood glucose levels).34 Patients with reduced renal function are at higher risk for hypoglycemia with insulin or sulfonylureas due to reduced drug clearance. However, insulin is often the only treatment for patients with advanced renal disease. Pay close attention to insulin dosing in patients with advanced renal disease, which may necessitate lower doses and smaller dose adjustments due to this risk.
Social determinants of health. Medication access and cost is a major burden in T2D management and should be considered for every patient. Compared with the period of 2005 to 2007, the annual cost of diabetes medications for an individual in 2015 to 2017 increased by 147%, rising from $1106 to $2727 per year.62 This increase is driven by the cost of insulin and newer medications without generic options.62 Identify local resources in your community, such as patient assistance programs and pharmacies with reduced-price generic prescription programs, which may be useful for patients who are underinsured or uninsured.
Continue to: Even if cost weren't an issue...
Even if cost weren’t an issue, many medications such as insulin and GLP-1 receptor agonists should be kept refrigerated and are only stable at room temperature for a limited time. Medications that are stable at room temperature should be prioritized in patients with limited or inconsistent access to refrigeration or unstable housing who may find it difficult to store their medications appropriately.
Do not delay insulin initiation in patients with high baseline A1C
Whenever possible, a GLP-1 receptor agonist is the preferred injectable medication to insulin. Starting insulin introduces numerous risks, including hypoglycemia, weight gain, and stigma. However, in the patient with newly diagnosed T2D, choose basal insulin when the baseline hyperglycemia is severe,34 as indicated by:
- blood glucose > 300 mg/dL (16.7 mmol/L),
- A1C > 10% (86 mmol/mol),
- symptoms of hyperglycemia (polyuria or polydipsia), or
- evidence of catabolism (weight loss, hypertriglyceridemia, ketosis).
Basal insulin analogs are preferred over NPH given their reduced variability, dosing, and hypoglycemic risk.35 Mixed insulins may be used if a patient is unable to afford an insulin analog, which can be quite costly. However, extensive counseling on dosing and management of hypoglycemia is crucial to patient safety with these agents. The ADA recommends initiating 0.1 to 0.2 units/kg of basal insulin daily or 10 units daily.34 The AACE follows this recommendation for patients with baseline A1C < 8%, but it proposes a more aggressive initiation of 0.2 to 0.3 units/kg/d for patients with baseline A1C > 8%.35 Titrate the dose by 2 units every 3 days to reach the target fasting blood glucose level. As hyperglycemia resolves, simplify the regimen and transition to noninsulin options per the previously discussed considerations.
It’s not just about glycemic control
In addition to the direct effects of hyperglycemia, a T2D diagnosis introduces an increased risk for ASCVD, a reduced ability to fight infection, and heightened risk for depression. Order a lipid panel at the time of T2D diagnosis and initiate lipid management as needed (TABLE 335,63,64). Both the ADA and the American Heart Association recommend starting a moderate-intensity statin as primary prevention for all patients with T2D between 40 and 75 years of age regardless of the 10-year ASCVD risk.63 The AACE uses specific lipid targets and recommends moderate- to high-intensity statin therapy for patients with T2D.35 All recommendations by professional organizations list high-intensity statins for patients with established ASCVD.
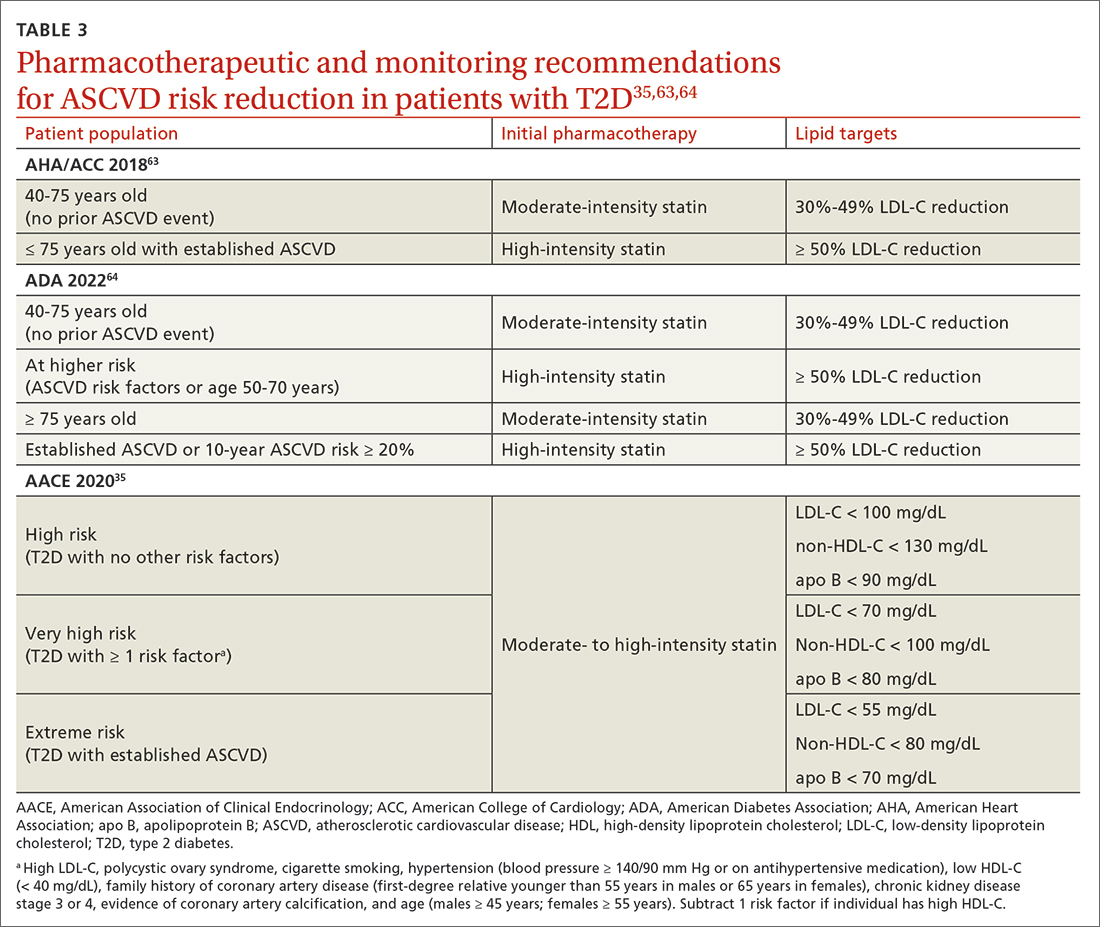
It is also vital to recommend that patients with newly diagnosed T2D remain up to date on all indicated vaccinations. They should promptly receive the hepatitis B and pneumococcal vaccines if they have not already done so for a previous indication. COVID-19 and annual influenza vaccines also should be prioritized for these patients.65
Finally, patients with diabetes are twice as likely to develop depression than patients without diabetes.66 Individuals with T2D and depression exhibit poorer medication adherence, lifestyle choices, and glycemic control.66 Screen for and treat these issues in all patients with T2D across the course of the disease.
Overall, work closely with patients to support them in managing their new diagnosis with evidence-based pharmacologic and nonpharmacologic approaches. The importance of lifestyle changes including high-fiber diets, regular exercise, and weight loss should not be overlooked. Do not delay starting pharmacotherapy after diagnosing T2D and consider medication-specific and patient-specific factors to individualize therapy, improve adherence, and prevent complications.
CORRESPONDENCE
Jennie B. Jarrett, PharmD, MMedEd, 833 South Wood Street (MC 886), Chicago, IL 60612; [email protected]
1. Dahlén AD, Dashi G, Maslov I, et al. Trends in antidiabetic drug discovery: FDA approved drugs, new drugs in clinical trials and global sales. Front Pharmacol. 2022;12. Accessed April 19, 2023. www.frontiersin.org/article/10.3389/fphar.2021.807548
2. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117-2128. doi: 10.1056/NEJMoa1504720
3. Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644-657. doi: 10.1056/NEJMoa1611925
4. Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347-357. doi: 10.1056/NEJMoa1812389
5. Davidson KW, Barry MJ, et al. Screening for prediabetes and type 2 diabetes: US Preventive Services Task Force recommendation statement. JAMA. 2021;326:736-743. doi: 10.1001/jama. 2021.12531
6. Handelsman Y, Bloomgarden ZT, Grunberger G, et al. American Association of Clinical Endocrinologists and American College of Endocrinology - clinical practice guidelines for developing a diabetes mellitus comprehensive care plan - 2015. Endocr Pract. 2015;21(suppl 1):1-87. doi: 10.4158/EP15672.GL
7. ADA. Introduction: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S1-S2. doi: 10.2337/dc22-Sint
8. ADA Professional Practice Committee. Classification and diagnosis of diabetes: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S17-S38. doi: 10.2337/dc22-S002
9. ADA Professional Practice Committee. Comprehensive medical evaluation and assessment of comorbidities: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S46-S59. doi: 10.2337/dc22-S004
10. ADA Professional Practice Committee. Glycemic targets: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S83-S96. doi: 10.2337/dc22-S006
11. Janapala RN, Jayaraj JS, Fathima N, et al. Continuous glucose monitoring versus self-monitoring of blood glucose in type 2 diabetes mellitus: a systematic review with meta-analysis. Cureus. 2019;11:e5634. doi: 10.7759/cureus.5634
12. ADA Professional Practice Committee. Diabetes technology: standards of medical care in diabetes - 2022. Diabetes Care. 2021;45(suppl 1):S97-S112. doi: 10.2337/dc22-S007
13. Qaseem A, Wilt TJ, Kansagara D, et al. Hemoglobin A1c targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: a guidance statement update from the American College of Physicians. Ann Intern Med. 2018;168:569-576. doi: 10.7326/M17-0939
14. Moran GM, Bakhai C, Song SH, et al, Guideline Committee. Type 2 diabetes: summary of updated NICE guidance. BMJ. 2022;377:o775. doi: 10.1136/bmj.o775
15. Kolb H, Martin S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017;15:131. doi: 10.1186/s12916-017-0901-x
16. McMacken M, Shah S. A plant-based diet for the prevention and treatment of type 2 diabetes. J Geriatr Cardiol. 2017;14:342-354. doi: 10.11909/j.issn.1671-5411.2017.05.009
17. Asif M. The prevention and control the type-2 diabetes by changing lifestyle and dietary pattern. J Educ Health Promot. 2014;3:1. doi: 10.4103/2277-9531.127541
18. Reynolds AN, Akerman AP, Mann J. Dietary fibre and whole grains in diabetes management: systematic review and meta-analyses. PLoS Med. 2020;17(3):e1003053. doi: 10.1371/journal.pmed.1003053
19. Li X, Cai X, Ma X, et al. Short- and long-term effects of wholegrain oat intake on weight management and glucolipid metabolism in overweight type-2 diabetics: a randomized control trial. Nutrients. 2016;8:549. doi: 10.3390/nu8090549
20. Fujii H, Iwase M, Ohkuma T, et al. Impact of dietary fiber intake on glycemic control, cardiovascular risk factors and chronic kidney disease in Japanese patients with type 2 diabetes mellitus: the Fukuoka Diabetes Registry. Nutr J. 2013;12:159. doi: 10.1186/1475-2891-12-159
21. Kim M, Jeung SR, Jeong TS, et al. Replacing with whole grains and legumes reduces Lp-PLA2 activities in plasma and PBMCs in patients with prediabetes or T2D. J Lipid Res. 2014;55:1762-1771. doi: 10.1194/jlr.M044834
22. Evert AB, Dennison M, Gardner CD, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731-754. doi: 10.2337/dci19-0014
23. Caballero AE. The “a to z” of managing type 2 diabetes in culturally diverse populations. Front Endocrinol. 2018;9:479. doi: 10.3389/fendo.2018.00479
24. Golbidi S, Badran M, Laher I. Antioxidant and anti-inflammatory effects of exercise in diabetic patients. Exp Diabetes Res. 2012; 2012:941868. doi: 10.1155/2012/941868
25. Karstoft K, Pedersen BK. Exercise and type 2 diabetes: focus on metabolism and inflammation. Immunol Cell Biol. 2016;94:146-150. doi: 10.1038/icb.2015.101
26. Dugan JA. Exercise recommendations for patients with type 2 diabetes. JAAPA. 2016;29:13-18. doi: 10.1097/01.JAA. 0000475460.77476.f6
27. Umpierre D, Ribeiro PA, Kramer CK, et al. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2011;305:1790–1799. doi: 10.1001/jama.2011.576
28. Zuhl M. Tips for monitoring aerobic exercise intensity. 2020. Accessed April 19, 2023. www.acsm.org/docs/default-source/files-for-resource-library/exercise-intensity-infographic.pdf? sfvrsn=f467c793_2
29. Williams A, Radford J, O’Brien J, Davison K. Type 2 diabetes and the medicine of exercise: the role of general practice in ensuring exercise is part of every patient’s plan. Aust J Gen Pract. 2020;49:189-193. doi: 10.31128/AJGP-09-19-5091
30. Grams J, Garvey WT. Weight loss and the prevention and treatment of type 2 diabetes using lifestyle therapy, pharmacotherapy, and bariatric surgery: mechanisms of action. Curr Obes Rep. 2015;4:287-302. doi: 10.1007/s13679-015-0155-x
31. Apovian CM, Okemah J, O’Neil PM. Body weight considerations in the management of type 2 diabetes. Adv Ther. 2019;36:44-58. doi: 10.1007/s12325-018-0824-8
32. Lean MEJ, Leslie WS, Barnes AC, et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019;7:344-355. doi: 10.1016/S2213-8587(19)30068-3
33. Rise MB, Pellerud A, Rygg LØ, et al. Making and maintaining lifestyle changes after participating in group based type 2 diabetes self-management educations: a qualitative study. PLoS One. 2013;8:e64009. doi: 10.1371/journal.pone.0064009
34. ADA Professional Practice Committee. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S125-S143. doi: 10.2337/dc22-S009
35. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive type 2 diabetes management algorithm—2020 executive summary. Endocr Pract. 2020;26:107-139. doi: 10.4158/CS-2019-0472
36. Metformin. Package insert. Bristol-Myers Squibb Company; 2017.
37. Invokana (canagliflozin). Package insert. Janssen Pharmaceuticals, Inc; 2020.
38. Farxiga (dapagliflozin). Package insert. AstraZeneca Pharmaceuticals LP; 2021.
39. Jardiance (empagliflozin). Package insert. Boehringer Ingelheim Pharmaceuticals, Inc; 2022.
40. Steglatro (ertugliflozin). Package insert. Merck & Co, Inc; 2021.
41. Trulicity (dulaglutide). Package insert. Lilly USA, LLC; 2022.
42. Byetta (exenatide). Package insert. AstraZeneca Canada Inc; 2022.
43. Bydureon (exenatide ER). Package insert. AstraZeneca Pharmaceuticals LP; 2022.
44. Victoza (liraglutide). Package insert. Novo Nordisk; 2022.
45. Adlyxin (lixisenatide). Package insert. Sanofi-Aventis US LLC; 2022.
46. Ozempic (semaglutide). Package insert. Novo Nordisk; 2022.
47. Alogliptin. Package insert. Takeda Pharmaceuticals USA, Inc; 2022.
48. Linagliptin. Package insert. Boehringer Ingelheim Pharmaceuticals, Inc; 2022.
49. Saxagliptin. Package insert. AstraZeneca Pharmaceuticals LP; 2019.
50. Januvia (sitagliptin). Package insert. Merck Sharp & Dohme LLC; 2022.
51. Glimepiride. Package insert. Sanofi-Aventis US LLC; 2009.
52. Glipizide. Package insert. Roerig; 2023.
53. Glyburide. Package insert. Sanofi-Aventis US LLC; 2009.
54. Pioglitazone. Package insert. Northstar Rx LLC; 2022.
55. Zelniker TA, Wiviott SD, Raz I, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31-39. doi: 10.1016/S0140-6736(18)32590-X
56. Zelniker TA, Wiviott SD, Raz I, et al. Comparison of the effects of glucagon-like peptide receptor agonists and sodium-glucose cotransporter 2 inhibitors for prevention of major adverse cardiovascular and renal outcomes in type 2 diabetes mellitus. Circulation. 2019;139:2022-2031. doi: 10.1161/CIRCULATIONAHA.118.038868
57. FDA. FDA Drug Safety Communication: FDA adds warnings about heart failure risk to labels of type 2 diabetes medicines containing saxagliptin and alogliptin. Accessed April 19, 2023. www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-adds-warnings-about-heart-failure-risk-labels-type-2-diabetes
58. Wilcox R, Bousser MG, Betteridge DJ, et al. Effects of pioglitazone in patients with type 2 diabetes with or without previous stroke: results from PROactive (PROspective pioglitAzone Clinical Trial In macroVascular Events 04). Stroke. 2007;38:865-873. doi: 10.1161/01.STR.0000257974.06317.49
59. Lingvay I, Hansen T, Macura S, et al. Superior weight loss with once-weekly semaglutide versus other glucagon-like peptide-1 receptor agonists is independent of gastrointestinal adverse events. BMJ Open Diabetes Res Care. 2020;8:e001706. doi: 10.1136/bmjdrc-2020-001706
60. Liu XY, Zhang N, Chen R, et al. Efficacy and safety of sodium-glucose cotransporter 2 inhibitors in type 2 diabetes: a meta-analysis of randomized controlled trials for 1 to 2 years. J Diabetes Complications. 2015;29:1295-1303. doi: 10.1016/j.jdiacomp.2015.07.011
61. Brown A, Guess N, Dornhorst A, et al. Insulin-associated weight gain in obese type 2 diabetes mellitus patients: what can be done? Diabetes Obes Metab. 2017;19:1655-1668. doi: 10.1111/dom.13009
62. Zhou X, Shrestha SS, Shao H, et al. Factors contributing to the rising national cost of glucose-lowering medicines for diabetes during 2005-2007 and 2015-2017. Diabetes Care. 2020;43:2396-2402. doi: 10.2337/dc19-2273
63. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi: 10.1161/CIR.0000000000000625
64. ADA Professional Practice Committee. Cardiovascular disease and risk management: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S144-S174. doi: 10.2337/dc22-S010
65. CDC. Adult immunization schedule by medical condition and other indication. 2022. Accessed April 19, 2023. www.cdc.gov/vaccines/schedules/hcp/imz/adult-conditions.htm
66. Semenkovich K, Brown ME, Svrakic DM, et al. Depression in type 2 diabetes mellitus: prevalence, impact, and treatment. Drugs. 2015;75:577-587. doi: 10.1007/s40265-015-0347-4
Nearly 40 antihyperglycemic agents have been approved by the US Food and Drug Administration (FDA) since the approval of human insulin in 1982.1 In addition, existing antihyperglycemic medications are constantly gaining FDA approval for new indications for common type 2 diabetes (T2D) comorbidities. For example, in addition to their glycemic benefits, the sodium-glucose cotransporter-2 (SGLT2) inhibitors have been approved for use in patients with T2D and established atherosclerotic cardiovascular disease (ASCVD) to reduce the risk for major adverse cardiovascular events (MACE; canagliflozin), risk for hospitalization for heart failure (dapagliflozin), and cardiovascular death (empagliflozin).2-4
The plethora of new agents and new data for existing agents, coupled with the annual release of guidelines from the American Diabetes Association (ADA) and practice recommendations from several other professional organizations,5-7 make it challenging for family physicians to stay current and provide the most up-to-date, evidence-based care. In this article, we provide advice on how to approach the screening, diagnosis, and evaluation of T2D, and on how to manage newly diagnosed T2D.
Screening, Dx, and evaluation: A quick review
Screening
Screening recommendations vary among professional organizations (TABLE 15,6,8). The US Preventive Services Task Force (USPSTF) recommends screening adults ages 35 to 70 years who are overweight or obese. Clinicians also can consider screening patients with a higher risk for diabetes.5 The ADA suggests screening all adults starting at 35 years, regardless of risk factors.8 Asymptomatic adults of any age with overweight or obesity and 1 or more risk factors should be screened.8

Making the diagnosis
The initial diagnosis of diabetes can be made by a fasting plasma glucose level ≥ 126 mg/dL (7.0 mmol/L); a 2-hour plasma glucose level ≥ 200 mg/dL (11.0 mmol/L) following an oral glucose tolerance test; or an A1C level ≥ 6.5%. Prioritize lab-drawn A1C measurements over point-of-care tests to diagnose T2D. In patients with classic symptoms of hyperglycemia, a random plasma glucose level ≥ 200 mg/dL (11.0 mmol/L) is also diagnostic. Generally, these tests are considered equally appropriate in screening for diabetes and may be used to detect prediabetes. In the absence of clear symptoms of hyperglycemia, the diagnosis of diabetes requires 2 abnormal screening test results, either via 1 blood sample (such as an abnormal A1C and glucose) or 2 separate blood samples of the same test. Further evaluation is advised if there is discordance between the 2 samples.8
Extended evaluations
Patients with newly diagnosed T2D require a thorough evaluation for comorbidities and complications of diabetes. Refer patients to an ophthalmologist for a dilated eye examination, with subsequent exams occurring every 1 to 2 years.6,9 Additional referrals for diabetes education, family planning for women of reproductive age, and dental, social, or mental health services may be clinically appropriate.9
Setting goals for glycemic control
Glycemic control is commonly monitored by the A1C level and by blood glucose monitoring either through traditional point-of-care glucometers or continuous glucose monitors (CGMs).10 Generally, CGMs provide more glycemic data than traditional glucometers and may cue patients to choose healthier dietary options and engage in physical exercise.11 Patients with T2D who use CGMs exhibit lower A1Cs, greater time in glycemic range, and reduced hypoglycemic episodes.11 Generally, CGMs are reserved for patients with type 1 diabetes and patients with T2D who use multiple daily injections, subcutaneous insulin infusions, or basal insulin only.12 Most professional organizations recommend that clinicians consider patient-specific factors to set individualized glycemic goals.6,10,13,14 For example, more stringent glycemic goals could be pursued for patients with longer life expectancy, shorter disease duration, absence of complications (eg, nephropathy, neuropathy, or cardiovascular disease), fewer comorbid conditions, lower hypoglycemia risk, or higher cognitive function.6
More specific A1C goals vary by professional organization. For nonpregnant adults, the ADA recommends an A1C goal of < 7% and a preprandial blood glucose level of 80 to 130 mg/dL (4.4-7.2 mmol/L).10 However, a lower A1C goal may be appropriate if it can be attained safely without causing hypoglycemia or other adverse effects.10 The AACE suggests an A1C goal of ≤ 6.5% and a fasting blood glucose level of < 110 mg/dL when it can be achieved safely.6 More stringent A1C goals may reduce long-term micro- and macrovascular complications—especially in patients with newly diagnosed T2D.10 While older studies such as the ACCORD trial found increased mortality in groups with more stringent glycemic targets, they did not include newer agents (SGLT2 inhibitors or glucagon-like peptide-1 [GLP-1] receptor agonists) that reduce cardiovascular events by mechanisms outside their glycemic-lowering effect. With these newer agents, more aggressive A1C goals can be targeted safely in select patients, particularly those with long life expectancy.10 Both the ADA and AACE recommend a less stringent A1C goal of 7% to 8% for patients with limited life expectancy or risks (eg, a history of hypoglycemia) that outweigh expected benefits.6,10
Continue to: Lifestyle modifications
Lifestyle modifications: As important as medication
Nutrition
The energy-dense Western diet, combined with sedentary behavior, are thought to be a primary cause of T2D.15 Therefore, include lifestyle modifications in the initial management of newly diagnosed T2D. Diets that replace carbohydrates with saturated and trans fats are related to increased mortality in patients with T2D.16 Increased consumption of vegetables, fruits, legumes, nuts, fish, cereal, and oils reduces concentrations of saturated and trans fats and increases dietary intake of monounsaturated fatty acids, fiber, antioxidants, and polyphenols.17
Increasing the intake of fiber, an undigestible carbohydrate, offers numerous benefits in T2D management. High-fiber diets can help regulate blood sugar and lipid levels, increase satiety, reduce inflammation, aid in weight management, and reduce premature mortality.18 Insoluble fiber, found in foods such as whole wheat flour, nuts, and cauliflower, helps food pass more quickly through the stomach and intestines and adds bulk to stool. Soluble fiber, found in foods such as chickpeas, lentils, and Brussels sprouts, absorbs water and forms a gel-like substance that protects nutrients from digestive enzymes and slows down digestion. The result is a more gradual rise in postprandial glucose levels and improved insulin sensitivity.19 Dietary fiber may produce short-chain fatty acids which in turn activate incretin secretion and stimulate a glucose-dependent release of insulin from the pancreas.20
Simple dietary substitutions, such as whole grains and legumes for white rice, can reduce fasting blood glucose and A1C levels.21 In a randomized controlled trial (RCT), increasing whole grain oat intake improved measures of glycemic control, reducing A1C by 1% at 1-year follow-up.19 Encourage patients with T2D to increase consumption of high-fiber foods and replace animal fats and refined grains with vegetable fats (eg, nuts, avocados, olives). Nutritional therapies should be individualized, taking into account personal preferences and cultural customs.22 Nutritional habits may be based on race/ethnicity, religion/spirituality, or even the city in which an individual resides. Nutrition recommendations should account for these differences as well as access to healthy foods. For instance, ethnic groups whose dietary patterns include tortillas could be counseled to choose high-fiber options such as corn instead of flour tortillas and to incorporate vegetables in place of high-fat foods. Additionally, ethnic groups who favor using animal fats in foods such as greens could be advised on ways to add flavor to vegetables without adding saturated fats. Taking this approach may lessen barriers to change and increase ability to make dietary modifications.23
Exercise
Encourage all patients with T2D to exercise regularly. The atherosclerotic plaques found in patients with T2D have increased inflammatory properties and result in worse cardiovascular outcomes compared with plaques in individuals without T2D.24 Regular exercise reduces levels of pro-inflammatory markers—C-reactive protein, interleukin (IL)-6, and tumor necrosis factor alpha—and increases levels of anti-inflammatory markers (IL-4 and IL-10).24 Regular exercise can improve body composition, physical fitness, lipid and glucose metabolism, and insulin sensitivity.25,26
A meta-analysis of RCTs demonstrated that structured exercise > 150 minutes per week resulted in A1C reductions of 0.89%,27 which is comparable to the effect of many oral antihyperglycemic medications.26 The Health Benefits of Aerobic and Resistance Training in individuals with T2D (HART-D) and Diabetes Aerobic and Resistance Exercise (DARE) studies demonstrated that combining endurance and resistance training was superior for improving glycemic control, cardiorespiratory fitness, and body composition, than using either type of training alone.25 Both the American College of Sports Medicine (ACSM) and the ADA recommend that adults engage in at least 150 total minutes of moderate-intensity aerobic activity per week and resistance training 2 to 3 times weekly.26 ACSM defines moderate-intensity exercise as 65% to 75% of maximal heart rate, a rating of perceived exertion of 3 to 4, or a step rate of 100 steps per minute.28
Continue to: Because of their longitudinal relationships...
Because of their longitudinal relationships with patients, family physicians are in an optimal position to assess a patient’s physical capacity level and provide individualized counseling. Several systematic reviews have demonstrated that counseling on exercise increases patients’ participation in physical activity.29 Encourage your patients with T2D to exercise regularly, considering each individual’s ability to engage in physical activity.
Weight loss
Include weight management in the initial treatment of patients with newly diagnosed T2D. Weight loss decreases hepatic glucose production and increases peripheral insulin sensitivity and insulin secretion.30 Moderate decreases in weight (5%-10%) can reduce complications related to diabetes, and sustained significant weight loss (> 10%) can potentially cause T2D remission (A1C < 6.5% after stopping diabetes medications).31,32
Diabetes self-management education supports patients by giving them tools for making and maintaining lifestyle changes. Understanding individual barriers to change and addressing these during motivational interviews is important. Through a qualitative interview study, participants in a diabetes self-management program revealed 4 factors that motivated them to maintain lifestyle changes: support from others, experiencing the impact of the changes they made, fear of T2D complications, and forming new habits.33 Family physicians are key in helping patients acquire knowledge and support to make the lifestyle modifications needed to manage newly diagnosed T2D.
Individualized pharmacotherapy considerations
For decades, the initial pharmacotherapeutic regimen for patients with newly diagnosed T2D considered the patient’s baseline A1C as a major driver for therapy. Metformin has been the mainstay in T2D treatment due to its clinical efficacy, minimal risk for hypoglycemia, and low cost. Regardless of the regimen, pharmacotherapy should be initiated at the time of T2D diagnosis in conjunction with the aforementioned lifestyle modifications.34
When selecting pharmacotherapy, practice guidelines recommend considering the efficacy and adverse effects of medications, patient-specific comorbidities, adherence, cost, and a patient’s lifestyle factors.34 Drug classes with pertinent information are listed in TABLE 2.34-54 After starting medication, monitor the A1C level every 3 months to determine whether therapy should be intensified. Patients should have their labs drawn ahead of the quarterly visit, or point-of-care measurements may be used to facilitate in-person patient–provider discussions.


Continue to: Consider patient-specific factors when starting pharmacotherapy
Consider patient-specific factors when starting pharmacotherapy
ASCVD. Regardless of baseline glycemic control, offer patients who have ASCVD, or who are at high risk for it, an SGLT2 inhibitor (canagliflozin, dapagliflozin, or empagliflozin) or a long-acting GLP-1 receptor agonist (dulaglutide, liraglutide, or semaglutide).34,35 SGLT2 inhibitors reduced the risk for MACE by 11% in patients with established ASCVD.55 They also reduced a composite outcome of cardiovascular death or hospitalization for heart failure by 23% in patients with or without ASCVD or heart failure at baseline.55 GLP-1 receptor agonists offer a similar reduction in MACE to SGLT2 inhibitors, but they do not have significant effects in heart failure.56 Thiazolidinediones (TZDs), saxagliptin, and alogliptin should be avoided in patients with heart failure.57 TZDs may reduce the risk for recurrent stroke in patients with T2D.58
Chronic kidney disease (CKD). As with ASCVD, prioritize SGLT2 inhibitors and GLP-1 receptor agonists in patients with CKD. While both classes reduced the risk for progression of kidney disease such as macroalbuminuria, SGLT2 inhibitors offer additional benefits in their reduction of the worsening of estimated glomerular filtration rate, end-stage kidney disease, and renal death.56
Obesity. Consider the effect of each drug class on weight when making initial treatment choices, taking special care to minimize weight gain and potentially promote weight loss.34 The ADA prefers GLP-1 receptor agonists, but also suggests SGLT2 inhibitors in these patients. While all GLP-1 receptor agonists have an impact on weight, weekly subcutaneous semaglutide offers the most pronounced weight loss of 2 to 7 kg over 56 weeks.59 SGLT2 inhibitors promote sustainable weight loss to a lesser degree, contributing to an average loss of 3 kg at 2 years.60 Weight gain is common in patients taking sulfonylureas (2.01-2.3 kg)31 and insulin (3-9 kg weight gain in the first year)61 and should be avoided in patients with T2D and obesity.34
Hypoglycemia risk. In addition to counseling patients on hypoglycemia management and prescribing glucagon rescue kits, offer medications with no or very low risk for hypoglycemia (eg, GLP-1 receptor agonists, SGLT2 inhibitors, dipeptidyl peptidase-4 inhibitors, and TZDs). Generally, avoid insulin and sulfonylureas in patients in whom hypoglycemia is a major concern (eg, older adults, individuals with labile blood glucose levels).34 Patients with reduced renal function are at higher risk for hypoglycemia with insulin or sulfonylureas due to reduced drug clearance. However, insulin is often the only treatment for patients with advanced renal disease. Pay close attention to insulin dosing in patients with advanced renal disease, which may necessitate lower doses and smaller dose adjustments due to this risk.
Social determinants of health. Medication access and cost is a major burden in T2D management and should be considered for every patient. Compared with the period of 2005 to 2007, the annual cost of diabetes medications for an individual in 2015 to 2017 increased by 147%, rising from $1106 to $2727 per year.62 This increase is driven by the cost of insulin and newer medications without generic options.62 Identify local resources in your community, such as patient assistance programs and pharmacies with reduced-price generic prescription programs, which may be useful for patients who are underinsured or uninsured.
Continue to: Even if cost weren't an issue...
Even if cost weren’t an issue, many medications such as insulin and GLP-1 receptor agonists should be kept refrigerated and are only stable at room temperature for a limited time. Medications that are stable at room temperature should be prioritized in patients with limited or inconsistent access to refrigeration or unstable housing who may find it difficult to store their medications appropriately.
Do not delay insulin initiation in patients with high baseline A1C
Whenever possible, a GLP-1 receptor agonist is the preferred injectable medication to insulin. Starting insulin introduces numerous risks, including hypoglycemia, weight gain, and stigma. However, in the patient with newly diagnosed T2D, choose basal insulin when the baseline hyperglycemia is severe,34 as indicated by:
- blood glucose > 300 mg/dL (16.7 mmol/L),
- A1C > 10% (86 mmol/mol),
- symptoms of hyperglycemia (polyuria or polydipsia), or
- evidence of catabolism (weight loss, hypertriglyceridemia, ketosis).
Basal insulin analogs are preferred over NPH given their reduced variability, dosing, and hypoglycemic risk.35 Mixed insulins may be used if a patient is unable to afford an insulin analog, which can be quite costly. However, extensive counseling on dosing and management of hypoglycemia is crucial to patient safety with these agents. The ADA recommends initiating 0.1 to 0.2 units/kg of basal insulin daily or 10 units daily.34 The AACE follows this recommendation for patients with baseline A1C < 8%, but it proposes a more aggressive initiation of 0.2 to 0.3 units/kg/d for patients with baseline A1C > 8%.35 Titrate the dose by 2 units every 3 days to reach the target fasting blood glucose level. As hyperglycemia resolves, simplify the regimen and transition to noninsulin options per the previously discussed considerations.
It’s not just about glycemic control
In addition to the direct effects of hyperglycemia, a T2D diagnosis introduces an increased risk for ASCVD, a reduced ability to fight infection, and heightened risk for depression. Order a lipid panel at the time of T2D diagnosis and initiate lipid management as needed (TABLE 335,63,64). Both the ADA and the American Heart Association recommend starting a moderate-intensity statin as primary prevention for all patients with T2D between 40 and 75 years of age regardless of the 10-year ASCVD risk.63 The AACE uses specific lipid targets and recommends moderate- to high-intensity statin therapy for patients with T2D.35 All recommendations by professional organizations list high-intensity statins for patients with established ASCVD.

It is also vital to recommend that patients with newly diagnosed T2D remain up to date on all indicated vaccinations. They should promptly receive the hepatitis B and pneumococcal vaccines if they have not already done so for a previous indication. COVID-19 and annual influenza vaccines also should be prioritized for these patients.65
Finally, patients with diabetes are twice as likely to develop depression than patients without diabetes.66 Individuals with T2D and depression exhibit poorer medication adherence, lifestyle choices, and glycemic control.66 Screen for and treat these issues in all patients with T2D across the course of the disease.
Overall, work closely with patients to support them in managing their new diagnosis with evidence-based pharmacologic and nonpharmacologic approaches. The importance of lifestyle changes including high-fiber diets, regular exercise, and weight loss should not be overlooked. Do not delay starting pharmacotherapy after diagnosing T2D and consider medication-specific and patient-specific factors to individualize therapy, improve adherence, and prevent complications.
CORRESPONDENCE
Jennie B. Jarrett, PharmD, MMedEd, 833 South Wood Street (MC 886), Chicago, IL 60612; [email protected]
Nearly 40 antihyperglycemic agents have been approved by the US Food and Drug Administration (FDA) since the approval of human insulin in 1982.1 In addition, existing antihyperglycemic medications are constantly gaining FDA approval for new indications for common type 2 diabetes (T2D) comorbidities. For example, in addition to their glycemic benefits, the sodium-glucose cotransporter-2 (SGLT2) inhibitors have been approved for use in patients with T2D and established atherosclerotic cardiovascular disease (ASCVD) to reduce the risk for major adverse cardiovascular events (MACE; canagliflozin), risk for hospitalization for heart failure (dapagliflozin), and cardiovascular death (empagliflozin).2-4
The plethora of new agents and new data for existing agents, coupled with the annual release of guidelines from the American Diabetes Association (ADA) and practice recommendations from several other professional organizations,5-7 make it challenging for family physicians to stay current and provide the most up-to-date, evidence-based care. In this article, we provide advice on how to approach the screening, diagnosis, and evaluation of T2D, and on how to manage newly diagnosed T2D.
Screening, Dx, and evaluation: A quick review
Screening
Screening recommendations vary among professional organizations (TABLE 15,6,8). The US Preventive Services Task Force (USPSTF) recommends screening adults ages 35 to 70 years who are overweight or obese. Clinicians also can consider screening patients with a higher risk for diabetes.5 The ADA suggests screening all adults starting at 35 years, regardless of risk factors.8 Asymptomatic adults of any age with overweight or obesity and 1 or more risk factors should be screened.8

Making the diagnosis
The initial diagnosis of diabetes can be made by a fasting plasma glucose level ≥ 126 mg/dL (7.0 mmol/L); a 2-hour plasma glucose level ≥ 200 mg/dL (11.0 mmol/L) following an oral glucose tolerance test; or an A1C level ≥ 6.5%. Prioritize lab-drawn A1C measurements over point-of-care tests to diagnose T2D. In patients with classic symptoms of hyperglycemia, a random plasma glucose level ≥ 200 mg/dL (11.0 mmol/L) is also diagnostic. Generally, these tests are considered equally appropriate in screening for diabetes and may be used to detect prediabetes. In the absence of clear symptoms of hyperglycemia, the diagnosis of diabetes requires 2 abnormal screening test results, either via 1 blood sample (such as an abnormal A1C and glucose) or 2 separate blood samples of the same test. Further evaluation is advised if there is discordance between the 2 samples.8
Extended evaluations
Patients with newly diagnosed T2D require a thorough evaluation for comorbidities and complications of diabetes. Refer patients to an ophthalmologist for a dilated eye examination, with subsequent exams occurring every 1 to 2 years.6,9 Additional referrals for diabetes education, family planning for women of reproductive age, and dental, social, or mental health services may be clinically appropriate.9
Setting goals for glycemic control
Glycemic control is commonly monitored by the A1C level and by blood glucose monitoring either through traditional point-of-care glucometers or continuous glucose monitors (CGMs).10 Generally, CGMs provide more glycemic data than traditional glucometers and may cue patients to choose healthier dietary options and engage in physical exercise.11 Patients with T2D who use CGMs exhibit lower A1Cs, greater time in glycemic range, and reduced hypoglycemic episodes.11 Generally, CGMs are reserved for patients with type 1 diabetes and patients with T2D who use multiple daily injections, subcutaneous insulin infusions, or basal insulin only.12 Most professional organizations recommend that clinicians consider patient-specific factors to set individualized glycemic goals.6,10,13,14 For example, more stringent glycemic goals could be pursued for patients with longer life expectancy, shorter disease duration, absence of complications (eg, nephropathy, neuropathy, or cardiovascular disease), fewer comorbid conditions, lower hypoglycemia risk, or higher cognitive function.6
More specific A1C goals vary by professional organization. For nonpregnant adults, the ADA recommends an A1C goal of < 7% and a preprandial blood glucose level of 80 to 130 mg/dL (4.4-7.2 mmol/L).10 However, a lower A1C goal may be appropriate if it can be attained safely without causing hypoglycemia or other adverse effects.10 The AACE suggests an A1C goal of ≤ 6.5% and a fasting blood glucose level of < 110 mg/dL when it can be achieved safely.6 More stringent A1C goals may reduce long-term micro- and macrovascular complications—especially in patients with newly diagnosed T2D.10 While older studies such as the ACCORD trial found increased mortality in groups with more stringent glycemic targets, they did not include newer agents (SGLT2 inhibitors or glucagon-like peptide-1 [GLP-1] receptor agonists) that reduce cardiovascular events by mechanisms outside their glycemic-lowering effect. With these newer agents, more aggressive A1C goals can be targeted safely in select patients, particularly those with long life expectancy.10 Both the ADA and AACE recommend a less stringent A1C goal of 7% to 8% for patients with limited life expectancy or risks (eg, a history of hypoglycemia) that outweigh expected benefits.6,10
Continue to: Lifestyle modifications
Lifestyle modifications: As important as medication
Nutrition
The energy-dense Western diet, combined with sedentary behavior, are thought to be a primary cause of T2D.15 Therefore, include lifestyle modifications in the initial management of newly diagnosed T2D. Diets that replace carbohydrates with saturated and trans fats are related to increased mortality in patients with T2D.16 Increased consumption of vegetables, fruits, legumes, nuts, fish, cereal, and oils reduces concentrations of saturated and trans fats and increases dietary intake of monounsaturated fatty acids, fiber, antioxidants, and polyphenols.17
Increasing the intake of fiber, an undigestible carbohydrate, offers numerous benefits in T2D management. High-fiber diets can help regulate blood sugar and lipid levels, increase satiety, reduce inflammation, aid in weight management, and reduce premature mortality.18 Insoluble fiber, found in foods such as whole wheat flour, nuts, and cauliflower, helps food pass more quickly through the stomach and intestines and adds bulk to stool. Soluble fiber, found in foods such as chickpeas, lentils, and Brussels sprouts, absorbs water and forms a gel-like substance that protects nutrients from digestive enzymes and slows down digestion. The result is a more gradual rise in postprandial glucose levels and improved insulin sensitivity.19 Dietary fiber may produce short-chain fatty acids which in turn activate incretin secretion and stimulate a glucose-dependent release of insulin from the pancreas.20
Simple dietary substitutions, such as whole grains and legumes for white rice, can reduce fasting blood glucose and A1C levels.21 In a randomized controlled trial (RCT), increasing whole grain oat intake improved measures of glycemic control, reducing A1C by 1% at 1-year follow-up.19 Encourage patients with T2D to increase consumption of high-fiber foods and replace animal fats and refined grains with vegetable fats (eg, nuts, avocados, olives). Nutritional therapies should be individualized, taking into account personal preferences and cultural customs.22 Nutritional habits may be based on race/ethnicity, religion/spirituality, or even the city in which an individual resides. Nutrition recommendations should account for these differences as well as access to healthy foods. For instance, ethnic groups whose dietary patterns include tortillas could be counseled to choose high-fiber options such as corn instead of flour tortillas and to incorporate vegetables in place of high-fat foods. Additionally, ethnic groups who favor using animal fats in foods such as greens could be advised on ways to add flavor to vegetables without adding saturated fats. Taking this approach may lessen barriers to change and increase ability to make dietary modifications.23
Exercise
Encourage all patients with T2D to exercise regularly. The atherosclerotic plaques found in patients with T2D have increased inflammatory properties and result in worse cardiovascular outcomes compared with plaques in individuals without T2D.24 Regular exercise reduces levels of pro-inflammatory markers—C-reactive protein, interleukin (IL)-6, and tumor necrosis factor alpha—and increases levels of anti-inflammatory markers (IL-4 and IL-10).24 Regular exercise can improve body composition, physical fitness, lipid and glucose metabolism, and insulin sensitivity.25,26
A meta-analysis of RCTs demonstrated that structured exercise > 150 minutes per week resulted in A1C reductions of 0.89%,27 which is comparable to the effect of many oral antihyperglycemic medications.26 The Health Benefits of Aerobic and Resistance Training in individuals with T2D (HART-D) and Diabetes Aerobic and Resistance Exercise (DARE) studies demonstrated that combining endurance and resistance training was superior for improving glycemic control, cardiorespiratory fitness, and body composition, than using either type of training alone.25 Both the American College of Sports Medicine (ACSM) and the ADA recommend that adults engage in at least 150 total minutes of moderate-intensity aerobic activity per week and resistance training 2 to 3 times weekly.26 ACSM defines moderate-intensity exercise as 65% to 75% of maximal heart rate, a rating of perceived exertion of 3 to 4, or a step rate of 100 steps per minute.28
Continue to: Because of their longitudinal relationships...
Because of their longitudinal relationships with patients, family physicians are in an optimal position to assess a patient’s physical capacity level and provide individualized counseling. Several systematic reviews have demonstrated that counseling on exercise increases patients’ participation in physical activity.29 Encourage your patients with T2D to exercise regularly, considering each individual’s ability to engage in physical activity.
Weight loss
Include weight management in the initial treatment of patients with newly diagnosed T2D. Weight loss decreases hepatic glucose production and increases peripheral insulin sensitivity and insulin secretion.30 Moderate decreases in weight (5%-10%) can reduce complications related to diabetes, and sustained significant weight loss (> 10%) can potentially cause T2D remission (A1C < 6.5% after stopping diabetes medications).31,32
Diabetes self-management education supports patients by giving them tools for making and maintaining lifestyle changes. Understanding individual barriers to change and addressing these during motivational interviews is important. Through a qualitative interview study, participants in a diabetes self-management program revealed 4 factors that motivated them to maintain lifestyle changes: support from others, experiencing the impact of the changes they made, fear of T2D complications, and forming new habits.33 Family physicians are key in helping patients acquire knowledge and support to make the lifestyle modifications needed to manage newly diagnosed T2D.
Individualized pharmacotherapy considerations
For decades, the initial pharmacotherapeutic regimen for patients with newly diagnosed T2D considered the patient’s baseline A1C as a major driver for therapy. Metformin has been the mainstay in T2D treatment due to its clinical efficacy, minimal risk for hypoglycemia, and low cost. Regardless of the regimen, pharmacotherapy should be initiated at the time of T2D diagnosis in conjunction with the aforementioned lifestyle modifications.34
When selecting pharmacotherapy, practice guidelines recommend considering the efficacy and adverse effects of medications, patient-specific comorbidities, adherence, cost, and a patient’s lifestyle factors.34 Drug classes with pertinent information are listed in TABLE 2.34-54 After starting medication, monitor the A1C level every 3 months to determine whether therapy should be intensified. Patients should have their labs drawn ahead of the quarterly visit, or point-of-care measurements may be used to facilitate in-person patient–provider discussions.


Continue to: Consider patient-specific factors when starting pharmacotherapy
Consider patient-specific factors when starting pharmacotherapy
ASCVD. Regardless of baseline glycemic control, offer patients who have ASCVD, or who are at high risk for it, an SGLT2 inhibitor (canagliflozin, dapagliflozin, or empagliflozin) or a long-acting GLP-1 receptor agonist (dulaglutide, liraglutide, or semaglutide).34,35 SGLT2 inhibitors reduced the risk for MACE by 11% in patients with established ASCVD.55 They also reduced a composite outcome of cardiovascular death or hospitalization for heart failure by 23% in patients with or without ASCVD or heart failure at baseline.55 GLP-1 receptor agonists offer a similar reduction in MACE to SGLT2 inhibitors, but they do not have significant effects in heart failure.56 Thiazolidinediones (TZDs), saxagliptin, and alogliptin should be avoided in patients with heart failure.57 TZDs may reduce the risk for recurrent stroke in patients with T2D.58
Chronic kidney disease (CKD). As with ASCVD, prioritize SGLT2 inhibitors and GLP-1 receptor agonists in patients with CKD. While both classes reduced the risk for progression of kidney disease such as macroalbuminuria, SGLT2 inhibitors offer additional benefits in their reduction of the worsening of estimated glomerular filtration rate, end-stage kidney disease, and renal death.56
Obesity. Consider the effect of each drug class on weight when making initial treatment choices, taking special care to minimize weight gain and potentially promote weight loss.34 The ADA prefers GLP-1 receptor agonists, but also suggests SGLT2 inhibitors in these patients. While all GLP-1 receptor agonists have an impact on weight, weekly subcutaneous semaglutide offers the most pronounced weight loss of 2 to 7 kg over 56 weeks.59 SGLT2 inhibitors promote sustainable weight loss to a lesser degree, contributing to an average loss of 3 kg at 2 years.60 Weight gain is common in patients taking sulfonylureas (2.01-2.3 kg)31 and insulin (3-9 kg weight gain in the first year)61 and should be avoided in patients with T2D and obesity.34
Hypoglycemia risk. In addition to counseling patients on hypoglycemia management and prescribing glucagon rescue kits, offer medications with no or very low risk for hypoglycemia (eg, GLP-1 receptor agonists, SGLT2 inhibitors, dipeptidyl peptidase-4 inhibitors, and TZDs). Generally, avoid insulin and sulfonylureas in patients in whom hypoglycemia is a major concern (eg, older adults, individuals with labile blood glucose levels).34 Patients with reduced renal function are at higher risk for hypoglycemia with insulin or sulfonylureas due to reduced drug clearance. However, insulin is often the only treatment for patients with advanced renal disease. Pay close attention to insulin dosing in patients with advanced renal disease, which may necessitate lower doses and smaller dose adjustments due to this risk.
Social determinants of health. Medication access and cost is a major burden in T2D management and should be considered for every patient. Compared with the period of 2005 to 2007, the annual cost of diabetes medications for an individual in 2015 to 2017 increased by 147%, rising from $1106 to $2727 per year.62 This increase is driven by the cost of insulin and newer medications without generic options.62 Identify local resources in your community, such as patient assistance programs and pharmacies with reduced-price generic prescription programs, which may be useful for patients who are underinsured or uninsured.
Continue to: Even if cost weren't an issue...
Even if cost weren’t an issue, many medications such as insulin and GLP-1 receptor agonists should be kept refrigerated and are only stable at room temperature for a limited time. Medications that are stable at room temperature should be prioritized in patients with limited or inconsistent access to refrigeration or unstable housing who may find it difficult to store their medications appropriately.
Do not delay insulin initiation in patients with high baseline A1C
Whenever possible, a GLP-1 receptor agonist is the preferred injectable medication to insulin. Starting insulin introduces numerous risks, including hypoglycemia, weight gain, and stigma. However, in the patient with newly diagnosed T2D, choose basal insulin when the baseline hyperglycemia is severe,34 as indicated by:
- blood glucose > 300 mg/dL (16.7 mmol/L),
- A1C > 10% (86 mmol/mol),
- symptoms of hyperglycemia (polyuria or polydipsia), or
- evidence of catabolism (weight loss, hypertriglyceridemia, ketosis).
Basal insulin analogs are preferred over NPH given their reduced variability, dosing, and hypoglycemic risk.35 Mixed insulins may be used if a patient is unable to afford an insulin analog, which can be quite costly. However, extensive counseling on dosing and management of hypoglycemia is crucial to patient safety with these agents. The ADA recommends initiating 0.1 to 0.2 units/kg of basal insulin daily or 10 units daily.34 The AACE follows this recommendation for patients with baseline A1C < 8%, but it proposes a more aggressive initiation of 0.2 to 0.3 units/kg/d for patients with baseline A1C > 8%.35 Titrate the dose by 2 units every 3 days to reach the target fasting blood glucose level. As hyperglycemia resolves, simplify the regimen and transition to noninsulin options per the previously discussed considerations.
It’s not just about glycemic control
In addition to the direct effects of hyperglycemia, a T2D diagnosis introduces an increased risk for ASCVD, a reduced ability to fight infection, and heightened risk for depression. Order a lipid panel at the time of T2D diagnosis and initiate lipid management as needed (TABLE 335,63,64). Both the ADA and the American Heart Association recommend starting a moderate-intensity statin as primary prevention for all patients with T2D between 40 and 75 years of age regardless of the 10-year ASCVD risk.63 The AACE uses specific lipid targets and recommends moderate- to high-intensity statin therapy for patients with T2D.35 All recommendations by professional organizations list high-intensity statins for patients with established ASCVD.

It is also vital to recommend that patients with newly diagnosed T2D remain up to date on all indicated vaccinations. They should promptly receive the hepatitis B and pneumococcal vaccines if they have not already done so for a previous indication. COVID-19 and annual influenza vaccines also should be prioritized for these patients.65
Finally, patients with diabetes are twice as likely to develop depression than patients without diabetes.66 Individuals with T2D and depression exhibit poorer medication adherence, lifestyle choices, and glycemic control.66 Screen for and treat these issues in all patients with T2D across the course of the disease.
Overall, work closely with patients to support them in managing their new diagnosis with evidence-based pharmacologic and nonpharmacologic approaches. The importance of lifestyle changes including high-fiber diets, regular exercise, and weight loss should not be overlooked. Do not delay starting pharmacotherapy after diagnosing T2D and consider medication-specific and patient-specific factors to individualize therapy, improve adherence, and prevent complications.
CORRESPONDENCE
Jennie B. Jarrett, PharmD, MMedEd, 833 South Wood Street (MC 886), Chicago, IL 60612; [email protected]
1. Dahlén AD, Dashi G, Maslov I, et al. Trends in antidiabetic drug discovery: FDA approved drugs, new drugs in clinical trials and global sales. Front Pharmacol. 2022;12. Accessed April 19, 2023. www.frontiersin.org/article/10.3389/fphar.2021.807548
2. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117-2128. doi: 10.1056/NEJMoa1504720
3. Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644-657. doi: 10.1056/NEJMoa1611925
4. Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347-357. doi: 10.1056/NEJMoa1812389
5. Davidson KW, Barry MJ, et al. Screening for prediabetes and type 2 diabetes: US Preventive Services Task Force recommendation statement. JAMA. 2021;326:736-743. doi: 10.1001/jama. 2021.12531
6. Handelsman Y, Bloomgarden ZT, Grunberger G, et al. American Association of Clinical Endocrinologists and American College of Endocrinology - clinical practice guidelines for developing a diabetes mellitus comprehensive care plan - 2015. Endocr Pract. 2015;21(suppl 1):1-87. doi: 10.4158/EP15672.GL
7. ADA. Introduction: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S1-S2. doi: 10.2337/dc22-Sint
8. ADA Professional Practice Committee. Classification and diagnosis of diabetes: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S17-S38. doi: 10.2337/dc22-S002
9. ADA Professional Practice Committee. Comprehensive medical evaluation and assessment of comorbidities: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S46-S59. doi: 10.2337/dc22-S004
10. ADA Professional Practice Committee. Glycemic targets: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S83-S96. doi: 10.2337/dc22-S006
11. Janapala RN, Jayaraj JS, Fathima N, et al. Continuous glucose monitoring versus self-monitoring of blood glucose in type 2 diabetes mellitus: a systematic review with meta-analysis. Cureus. 2019;11:e5634. doi: 10.7759/cureus.5634
12. ADA Professional Practice Committee. Diabetes technology: standards of medical care in diabetes - 2022. Diabetes Care. 2021;45(suppl 1):S97-S112. doi: 10.2337/dc22-S007
13. Qaseem A, Wilt TJ, Kansagara D, et al. Hemoglobin A1c targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: a guidance statement update from the American College of Physicians. Ann Intern Med. 2018;168:569-576. doi: 10.7326/M17-0939
14. Moran GM, Bakhai C, Song SH, et al, Guideline Committee. Type 2 diabetes: summary of updated NICE guidance. BMJ. 2022;377:o775. doi: 10.1136/bmj.o775
15. Kolb H, Martin S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017;15:131. doi: 10.1186/s12916-017-0901-x
16. McMacken M, Shah S. A plant-based diet for the prevention and treatment of type 2 diabetes. J Geriatr Cardiol. 2017;14:342-354. doi: 10.11909/j.issn.1671-5411.2017.05.009
17. Asif M. The prevention and control the type-2 diabetes by changing lifestyle and dietary pattern. J Educ Health Promot. 2014;3:1. doi: 10.4103/2277-9531.127541
18. Reynolds AN, Akerman AP, Mann J. Dietary fibre and whole grains in diabetes management: systematic review and meta-analyses. PLoS Med. 2020;17(3):e1003053. doi: 10.1371/journal.pmed.1003053
19. Li X, Cai X, Ma X, et al. Short- and long-term effects of wholegrain oat intake on weight management and glucolipid metabolism in overweight type-2 diabetics: a randomized control trial. Nutrients. 2016;8:549. doi: 10.3390/nu8090549
20. Fujii H, Iwase M, Ohkuma T, et al. Impact of dietary fiber intake on glycemic control, cardiovascular risk factors and chronic kidney disease in Japanese patients with type 2 diabetes mellitus: the Fukuoka Diabetes Registry. Nutr J. 2013;12:159. doi: 10.1186/1475-2891-12-159
21. Kim M, Jeung SR, Jeong TS, et al. Replacing with whole grains and legumes reduces Lp-PLA2 activities in plasma and PBMCs in patients with prediabetes or T2D. J Lipid Res. 2014;55:1762-1771. doi: 10.1194/jlr.M044834
22. Evert AB, Dennison M, Gardner CD, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731-754. doi: 10.2337/dci19-0014
23. Caballero AE. The “a to z” of managing type 2 diabetes in culturally diverse populations. Front Endocrinol. 2018;9:479. doi: 10.3389/fendo.2018.00479
24. Golbidi S, Badran M, Laher I. Antioxidant and anti-inflammatory effects of exercise in diabetic patients. Exp Diabetes Res. 2012; 2012:941868. doi: 10.1155/2012/941868
25. Karstoft K, Pedersen BK. Exercise and type 2 diabetes: focus on metabolism and inflammation. Immunol Cell Biol. 2016;94:146-150. doi: 10.1038/icb.2015.101
26. Dugan JA. Exercise recommendations for patients with type 2 diabetes. JAAPA. 2016;29:13-18. doi: 10.1097/01.JAA. 0000475460.77476.f6
27. Umpierre D, Ribeiro PA, Kramer CK, et al. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2011;305:1790–1799. doi: 10.1001/jama.2011.576
28. Zuhl M. Tips for monitoring aerobic exercise intensity. 2020. Accessed April 19, 2023. www.acsm.org/docs/default-source/files-for-resource-library/exercise-intensity-infographic.pdf? sfvrsn=f467c793_2
29. Williams A, Radford J, O’Brien J, Davison K. Type 2 diabetes and the medicine of exercise: the role of general practice in ensuring exercise is part of every patient’s plan. Aust J Gen Pract. 2020;49:189-193. doi: 10.31128/AJGP-09-19-5091
30. Grams J, Garvey WT. Weight loss and the prevention and treatment of type 2 diabetes using lifestyle therapy, pharmacotherapy, and bariatric surgery: mechanisms of action. Curr Obes Rep. 2015;4:287-302. doi: 10.1007/s13679-015-0155-x
31. Apovian CM, Okemah J, O’Neil PM. Body weight considerations in the management of type 2 diabetes. Adv Ther. 2019;36:44-58. doi: 10.1007/s12325-018-0824-8
32. Lean MEJ, Leslie WS, Barnes AC, et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019;7:344-355. doi: 10.1016/S2213-8587(19)30068-3
33. Rise MB, Pellerud A, Rygg LØ, et al. Making and maintaining lifestyle changes after participating in group based type 2 diabetes self-management educations: a qualitative study. PLoS One. 2013;8:e64009. doi: 10.1371/journal.pone.0064009
34. ADA Professional Practice Committee. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S125-S143. doi: 10.2337/dc22-S009
35. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive type 2 diabetes management algorithm—2020 executive summary. Endocr Pract. 2020;26:107-139. doi: 10.4158/CS-2019-0472
36. Metformin. Package insert. Bristol-Myers Squibb Company; 2017.
37. Invokana (canagliflozin). Package insert. Janssen Pharmaceuticals, Inc; 2020.
38. Farxiga (dapagliflozin). Package insert. AstraZeneca Pharmaceuticals LP; 2021.
39. Jardiance (empagliflozin). Package insert. Boehringer Ingelheim Pharmaceuticals, Inc; 2022.
40. Steglatro (ertugliflozin). Package insert. Merck & Co, Inc; 2021.
41. Trulicity (dulaglutide). Package insert. Lilly USA, LLC; 2022.
42. Byetta (exenatide). Package insert. AstraZeneca Canada Inc; 2022.
43. Bydureon (exenatide ER). Package insert. AstraZeneca Pharmaceuticals LP; 2022.
44. Victoza (liraglutide). Package insert. Novo Nordisk; 2022.
45. Adlyxin (lixisenatide). Package insert. Sanofi-Aventis US LLC; 2022.
46. Ozempic (semaglutide). Package insert. Novo Nordisk; 2022.
47. Alogliptin. Package insert. Takeda Pharmaceuticals USA, Inc; 2022.
48. Linagliptin. Package insert. Boehringer Ingelheim Pharmaceuticals, Inc; 2022.
49. Saxagliptin. Package insert. AstraZeneca Pharmaceuticals LP; 2019.
50. Januvia (sitagliptin). Package insert. Merck Sharp & Dohme LLC; 2022.
51. Glimepiride. Package insert. Sanofi-Aventis US LLC; 2009.
52. Glipizide. Package insert. Roerig; 2023.
53. Glyburide. Package insert. Sanofi-Aventis US LLC; 2009.
54. Pioglitazone. Package insert. Northstar Rx LLC; 2022.
55. Zelniker TA, Wiviott SD, Raz I, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31-39. doi: 10.1016/S0140-6736(18)32590-X
56. Zelniker TA, Wiviott SD, Raz I, et al. Comparison of the effects of glucagon-like peptide receptor agonists and sodium-glucose cotransporter 2 inhibitors for prevention of major adverse cardiovascular and renal outcomes in type 2 diabetes mellitus. Circulation. 2019;139:2022-2031. doi: 10.1161/CIRCULATIONAHA.118.038868
57. FDA. FDA Drug Safety Communication: FDA adds warnings about heart failure risk to labels of type 2 diabetes medicines containing saxagliptin and alogliptin. Accessed April 19, 2023. www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-adds-warnings-about-heart-failure-risk-labels-type-2-diabetes
58. Wilcox R, Bousser MG, Betteridge DJ, et al. Effects of pioglitazone in patients with type 2 diabetes with or without previous stroke: results from PROactive (PROspective pioglitAzone Clinical Trial In macroVascular Events 04). Stroke. 2007;38:865-873. doi: 10.1161/01.STR.0000257974.06317.49
59. Lingvay I, Hansen T, Macura S, et al. Superior weight loss with once-weekly semaglutide versus other glucagon-like peptide-1 receptor agonists is independent of gastrointestinal adverse events. BMJ Open Diabetes Res Care. 2020;8:e001706. doi: 10.1136/bmjdrc-2020-001706
60. Liu XY, Zhang N, Chen R, et al. Efficacy and safety of sodium-glucose cotransporter 2 inhibitors in type 2 diabetes: a meta-analysis of randomized controlled trials for 1 to 2 years. J Diabetes Complications. 2015;29:1295-1303. doi: 10.1016/j.jdiacomp.2015.07.011
61. Brown A, Guess N, Dornhorst A, et al. Insulin-associated weight gain in obese type 2 diabetes mellitus patients: what can be done? Diabetes Obes Metab. 2017;19:1655-1668. doi: 10.1111/dom.13009
62. Zhou X, Shrestha SS, Shao H, et al. Factors contributing to the rising national cost of glucose-lowering medicines for diabetes during 2005-2007 and 2015-2017. Diabetes Care. 2020;43:2396-2402. doi: 10.2337/dc19-2273
63. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi: 10.1161/CIR.0000000000000625
64. ADA Professional Practice Committee. Cardiovascular disease and risk management: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S144-S174. doi: 10.2337/dc22-S010
65. CDC. Adult immunization schedule by medical condition and other indication. 2022. Accessed April 19, 2023. www.cdc.gov/vaccines/schedules/hcp/imz/adult-conditions.htm
66. Semenkovich K, Brown ME, Svrakic DM, et al. Depression in type 2 diabetes mellitus: prevalence, impact, and treatment. Drugs. 2015;75:577-587. doi: 10.1007/s40265-015-0347-4
1. Dahlén AD, Dashi G, Maslov I, et al. Trends in antidiabetic drug discovery: FDA approved drugs, new drugs in clinical trials and global sales. Front Pharmacol. 2022;12. Accessed April 19, 2023. www.frontiersin.org/article/10.3389/fphar.2021.807548
2. Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117-2128. doi: 10.1056/NEJMoa1504720
3. Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644-657. doi: 10.1056/NEJMoa1611925
4. Wiviott SD, Raz I, Bonaca MP, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347-357. doi: 10.1056/NEJMoa1812389
5. Davidson KW, Barry MJ, et al. Screening for prediabetes and type 2 diabetes: US Preventive Services Task Force recommendation statement. JAMA. 2021;326:736-743. doi: 10.1001/jama. 2021.12531
6. Handelsman Y, Bloomgarden ZT, Grunberger G, et al. American Association of Clinical Endocrinologists and American College of Endocrinology - clinical practice guidelines for developing a diabetes mellitus comprehensive care plan - 2015. Endocr Pract. 2015;21(suppl 1):1-87. doi: 10.4158/EP15672.GL
7. ADA. Introduction: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S1-S2. doi: 10.2337/dc22-Sint
8. ADA Professional Practice Committee. Classification and diagnosis of diabetes: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S17-S38. doi: 10.2337/dc22-S002
9. ADA Professional Practice Committee. Comprehensive medical evaluation and assessment of comorbidities: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S46-S59. doi: 10.2337/dc22-S004
10. ADA Professional Practice Committee. Glycemic targets: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S83-S96. doi: 10.2337/dc22-S006
11. Janapala RN, Jayaraj JS, Fathima N, et al. Continuous glucose monitoring versus self-monitoring of blood glucose in type 2 diabetes mellitus: a systematic review with meta-analysis. Cureus. 2019;11:e5634. doi: 10.7759/cureus.5634
12. ADA Professional Practice Committee. Diabetes technology: standards of medical care in diabetes - 2022. Diabetes Care. 2021;45(suppl 1):S97-S112. doi: 10.2337/dc22-S007
13. Qaseem A, Wilt TJ, Kansagara D, et al. Hemoglobin A1c targets for glycemic control with pharmacologic therapy for nonpregnant adults with type 2 diabetes mellitus: a guidance statement update from the American College of Physicians. Ann Intern Med. 2018;168:569-576. doi: 10.7326/M17-0939
14. Moran GM, Bakhai C, Song SH, et al, Guideline Committee. Type 2 diabetes: summary of updated NICE guidance. BMJ. 2022;377:o775. doi: 10.1136/bmj.o775
15. Kolb H, Martin S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017;15:131. doi: 10.1186/s12916-017-0901-x
16. McMacken M, Shah S. A plant-based diet for the prevention and treatment of type 2 diabetes. J Geriatr Cardiol. 2017;14:342-354. doi: 10.11909/j.issn.1671-5411.2017.05.009
17. Asif M. The prevention and control the type-2 diabetes by changing lifestyle and dietary pattern. J Educ Health Promot. 2014;3:1. doi: 10.4103/2277-9531.127541
18. Reynolds AN, Akerman AP, Mann J. Dietary fibre and whole grains in diabetes management: systematic review and meta-analyses. PLoS Med. 2020;17(3):e1003053. doi: 10.1371/journal.pmed.1003053
19. Li X, Cai X, Ma X, et al. Short- and long-term effects of wholegrain oat intake on weight management and glucolipid metabolism in overweight type-2 diabetics: a randomized control trial. Nutrients. 2016;8:549. doi: 10.3390/nu8090549
20. Fujii H, Iwase M, Ohkuma T, et al. Impact of dietary fiber intake on glycemic control, cardiovascular risk factors and chronic kidney disease in Japanese patients with type 2 diabetes mellitus: the Fukuoka Diabetes Registry. Nutr J. 2013;12:159. doi: 10.1186/1475-2891-12-159
21. Kim M, Jeung SR, Jeong TS, et al. Replacing with whole grains and legumes reduces Lp-PLA2 activities in plasma and PBMCs in patients with prediabetes or T2D. J Lipid Res. 2014;55:1762-1771. doi: 10.1194/jlr.M044834
22. Evert AB, Dennison M, Gardner CD, et al. Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care. 2019;42:731-754. doi: 10.2337/dci19-0014
23. Caballero AE. The “a to z” of managing type 2 diabetes in culturally diverse populations. Front Endocrinol. 2018;9:479. doi: 10.3389/fendo.2018.00479
24. Golbidi S, Badran M, Laher I. Antioxidant and anti-inflammatory effects of exercise in diabetic patients. Exp Diabetes Res. 2012; 2012:941868. doi: 10.1155/2012/941868
25. Karstoft K, Pedersen BK. Exercise and type 2 diabetes: focus on metabolism and inflammation. Immunol Cell Biol. 2016;94:146-150. doi: 10.1038/icb.2015.101
26. Dugan JA. Exercise recommendations for patients with type 2 diabetes. JAAPA. 2016;29:13-18. doi: 10.1097/01.JAA. 0000475460.77476.f6
27. Umpierre D, Ribeiro PA, Kramer CK, et al. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA. 2011;305:1790–1799. doi: 10.1001/jama.2011.576
28. Zuhl M. Tips for monitoring aerobic exercise intensity. 2020. Accessed April 19, 2023. www.acsm.org/docs/default-source/files-for-resource-library/exercise-intensity-infographic.pdf? sfvrsn=f467c793_2
29. Williams A, Radford J, O’Brien J, Davison K. Type 2 diabetes and the medicine of exercise: the role of general practice in ensuring exercise is part of every patient’s plan. Aust J Gen Pract. 2020;49:189-193. doi: 10.31128/AJGP-09-19-5091
30. Grams J, Garvey WT. Weight loss and the prevention and treatment of type 2 diabetes using lifestyle therapy, pharmacotherapy, and bariatric surgery: mechanisms of action. Curr Obes Rep. 2015;4:287-302. doi: 10.1007/s13679-015-0155-x
31. Apovian CM, Okemah J, O’Neil PM. Body weight considerations in the management of type 2 diabetes. Adv Ther. 2019;36:44-58. doi: 10.1007/s12325-018-0824-8
32. Lean MEJ, Leslie WS, Barnes AC, et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019;7:344-355. doi: 10.1016/S2213-8587(19)30068-3
33. Rise MB, Pellerud A, Rygg LØ, et al. Making and maintaining lifestyle changes after participating in group based type 2 diabetes self-management educations: a qualitative study. PLoS One. 2013;8:e64009. doi: 10.1371/journal.pone.0064009
34. ADA Professional Practice Committee. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S125-S143. doi: 10.2337/dc22-S009
35. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive type 2 diabetes management algorithm—2020 executive summary. Endocr Pract. 2020;26:107-139. doi: 10.4158/CS-2019-0472
36. Metformin. Package insert. Bristol-Myers Squibb Company; 2017.
37. Invokana (canagliflozin). Package insert. Janssen Pharmaceuticals, Inc; 2020.
38. Farxiga (dapagliflozin). Package insert. AstraZeneca Pharmaceuticals LP; 2021.
39. Jardiance (empagliflozin). Package insert. Boehringer Ingelheim Pharmaceuticals, Inc; 2022.
40. Steglatro (ertugliflozin). Package insert. Merck & Co, Inc; 2021.
41. Trulicity (dulaglutide). Package insert. Lilly USA, LLC; 2022.
42. Byetta (exenatide). Package insert. AstraZeneca Canada Inc; 2022.
43. Bydureon (exenatide ER). Package insert. AstraZeneca Pharmaceuticals LP; 2022.
44. Victoza (liraglutide). Package insert. Novo Nordisk; 2022.
45. Adlyxin (lixisenatide). Package insert. Sanofi-Aventis US LLC; 2022.
46. Ozempic (semaglutide). Package insert. Novo Nordisk; 2022.
47. Alogliptin. Package insert. Takeda Pharmaceuticals USA, Inc; 2022.
48. Linagliptin. Package insert. Boehringer Ingelheim Pharmaceuticals, Inc; 2022.
49. Saxagliptin. Package insert. AstraZeneca Pharmaceuticals LP; 2019.
50. Januvia (sitagliptin). Package insert. Merck Sharp & Dohme LLC; 2022.
51. Glimepiride. Package insert. Sanofi-Aventis US LLC; 2009.
52. Glipizide. Package insert. Roerig; 2023.
53. Glyburide. Package insert. Sanofi-Aventis US LLC; 2009.
54. Pioglitazone. Package insert. Northstar Rx LLC; 2022.
55. Zelniker TA, Wiviott SD, Raz I, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31-39. doi: 10.1016/S0140-6736(18)32590-X
56. Zelniker TA, Wiviott SD, Raz I, et al. Comparison of the effects of glucagon-like peptide receptor agonists and sodium-glucose cotransporter 2 inhibitors for prevention of major adverse cardiovascular and renal outcomes in type 2 diabetes mellitus. Circulation. 2019;139:2022-2031. doi: 10.1161/CIRCULATIONAHA.118.038868
57. FDA. FDA Drug Safety Communication: FDA adds warnings about heart failure risk to labels of type 2 diabetes medicines containing saxagliptin and alogliptin. Accessed April 19, 2023. www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-adds-warnings-about-heart-failure-risk-labels-type-2-diabetes
58. Wilcox R, Bousser MG, Betteridge DJ, et al. Effects of pioglitazone in patients with type 2 diabetes with or without previous stroke: results from PROactive (PROspective pioglitAzone Clinical Trial In macroVascular Events 04). Stroke. 2007;38:865-873. doi: 10.1161/01.STR.0000257974.06317.49
59. Lingvay I, Hansen T, Macura S, et al. Superior weight loss with once-weekly semaglutide versus other glucagon-like peptide-1 receptor agonists is independent of gastrointestinal adverse events. BMJ Open Diabetes Res Care. 2020;8:e001706. doi: 10.1136/bmjdrc-2020-001706
60. Liu XY, Zhang N, Chen R, et al. Efficacy and safety of sodium-glucose cotransporter 2 inhibitors in type 2 diabetes: a meta-analysis of randomized controlled trials for 1 to 2 years. J Diabetes Complications. 2015;29:1295-1303. doi: 10.1016/j.jdiacomp.2015.07.011
61. Brown A, Guess N, Dornhorst A, et al. Insulin-associated weight gain in obese type 2 diabetes mellitus patients: what can be done? Diabetes Obes Metab. 2017;19:1655-1668. doi: 10.1111/dom.13009
62. Zhou X, Shrestha SS, Shao H, et al. Factors contributing to the rising national cost of glucose-lowering medicines for diabetes during 2005-2007 and 2015-2017. Diabetes Care. 2020;43:2396-2402. doi: 10.2337/dc19-2273
63. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi: 10.1161/CIR.0000000000000625
64. ADA Professional Practice Committee. Cardiovascular disease and risk management: standards of medical care in diabetes—2022. Diabetes Care. 2021;45(suppl 1):S144-S174. doi: 10.2337/dc22-S010
65. CDC. Adult immunization schedule by medical condition and other indication. 2022. Accessed April 19, 2023. www.cdc.gov/vaccines/schedules/hcp/imz/adult-conditions.htm
66. Semenkovich K, Brown ME, Svrakic DM, et al. Depression in type 2 diabetes mellitus: prevalence, impact, and treatment. Drugs. 2015;75:577-587. doi: 10.1007/s40265-015-0347-4
PRACTICE RECOMMENDATIONS
› Individualize lifestyle modifications, considering personal and cultural experiences, health literacy, access to healthy foods, willingness and ability to make behavior changes, and barriers to change. C
› Initiate medication therapy at diagnosis, considering medication efficacy and cost, hypoglycemia risk, weight effects, benefits in cardiovascular and kidney disease, and patient-specific comorbidities. C
› Start basal insulin as first-line therapy in patients with severe baseline hyperglycemia, symptoms of hyperglycemia, or evidence of catabolism. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Which patients might benefit from platelet-rich plasma?
Platelet-rich plasma (PRP) injections have become a popular treatment option in a variety of specialties including sports medicine, maxillofacial surgery, dermatology, cosmetology, and reproductive medicine.1 PRP is an autologous blood product derived from whole blood, using a centrifuge to isolate a concentrated layer of platelets. The a-granules in platelets release transforming growth factor b 1, vascular endothelial growth factor, platelet-derived growth factor, basic fibroblast growth factor, epidermal growth factor, insulin-like growth factor 1, and other mediatorsthat enhance the natural healing process.2
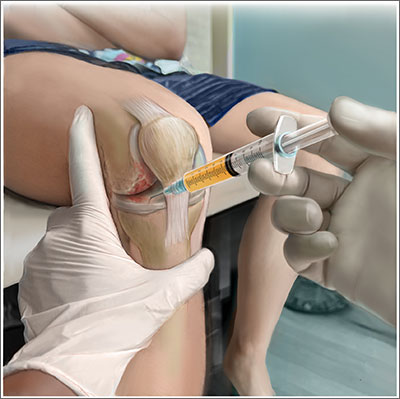
When patients ask. Familiarity with the use of PRP to treat specific musculoskeletal (MSK) conditions is essential for family physicians who frequently are asked by patients about whether PRP is right for them. These patients may have experienced failure of medication therapy or declined surgical intervention, or may not be surgical candidates. This review details the evidence surrounding common intra-articular and extra-articular applications of PRP. But first, a word about how PRP is prepared, its contraindications, and costs.
Preparation and types of PRP
Although there are many commercial systems for preparing PRP, there is no consensus on the optimal formulation.2 Other terms for PRP, such as autologous concentrated platelets and super-concentrated platelets, are based on concentration of red blood cells, leukocytes, and fibrin.3 PRP therapies usually are categorized as leukocyte-rich PRP (LR-PRP) or leukocyte-poor PRP (LP-PRP), based on neutrophil concentrations that are above and below baseline.2 Leukocyte concentration is one of the most debated topics in PRP therapy.4
Common commercially available preparation systems produce platelet concentrations between 3 to 6 times the baseline platelet count.5 Although there is no universally agreed upon PRP formulation, studies have shown 2 centrifugation cycles (“double-spun” or “dual centrifugation”) that yield platelet concentrations between 1.8 to 1.9 times the baseline values significantly improve MSK conditions.6-8
For MSK purposes, PRP may be injected into intratendinous, peritendinous, and intra-articular spaces. Currently, there is no consensus regarding injection frequency. Many studies have incorporated single-injection protocols, while some have used 2 to 3 injections repeated over several weeks to months. PRP commonly is injected at point-of-care without requiring storage.
Contraindications. PRP has been shown to be safe, with most adverse effects attributed to local injection site pain, bleeding, swelling, and bruising.9
Contraindications to PRP include active malignancy or recent remission from malignancy with the exception of nonmetastatic skin tumors.10 PRP is not recommended for patients with an allergy to manufacturing components (eg, dimethyl sulfoxide), thrombocytopenia, nonsteroidal anti-inflammatory drug use within 2 weeks, active infection causing fever, and local infection at the injection site.10 Since local anesthetics may impair platelet function, they should not be given at the same injection site as PRP.10
Continue to: Cost
Cost. PRP is not covered by most insurance plans.11,12 The cost for PRP may range from $500 to $2500 for a single injection.12
Evidence-based summary by condition
Knee osteoarthritis
❯❯❯ Consider using PRP
Knee osteoarthritis (OA) is a common cause of pain and disability. Treatment options include physical therapy, pharmacotherapy, and surgery. PRP has gained popularity as a nonsurgical option. A recent meta-analysis by Costa et al13 of 40 studies with 3035 participants comparing intra-articular PRP with hyaluronic acid (HA), corticosteroid, and saline injections, found that PRP appears to be more effective or as effective as other nonsurgical modalities. However, due to study heterogeneity and high risk for bias, the authors could not recommend PRP for knee OA in clinical practice.13
Despite Costa et al’s findings, reproducible data have demonstrated the superiority of PRP over other nonsurgical treatment options for knee OA. A 2021 systematic review and meta-analysis of 18 randomized controlled trials (RCTs; N = 811) by Belk et al6 comparing PRP to HA injections showed a higher mean improvement in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores in the PRP group compared to the HA group (44.7% vs 12.6%, respectively; P < .01).6 Six of 11 studies using the visual analog scale (VAS) for pain reported significantly less pain in the PRP group compared to the HA group (P < .05).6 The mean follow-up time was 11.1 months.6 Three of 6 studies reported improved subjective International Knee Documentation Committee (IKDC) scores (range from 0-100, with higher scores representing higher levels of function and lower levels of symptoms) in the PRP group compared to the HA group: 75.7 ± 15.1 vs 65.6 ± 16.9 (P = .004); 65.5 ± 3.6 vs 55.8 ± 3.8 (P = .01); and 60.8 ± 9.8 vs 48.4 ± 6.2 (P < .05).6 There was concern for moderate-to-high heterogeneity.6
Other systematic reviews and meta-analyses found similar efficacy of PRP for knee OA, including improved WOMAC scores and patient-reported outcomes (eg, pain, physical function, stiffness) compared to other injectable options.14,15 A systematic review of 14 RCTs (N = 1423) by Shen et al15 showed improved WOMAC scores at 3 months (mean differences [MD] = –14.53; 95% CI, –29.97 to –7.09; P < .001), 6 months
Despite a lack of consensus regarding the optimal preparation of PRP for knee OA, another recent RCT (N = 192) found significant improvement in mean subjective IKDC scores in the LR-PRP group (45.5 ± 15.5 to 60.7 ± 21.1; P < .0005) and the LP-PRP group (46.8 ± 15.8 to 62.9 ± 19.9; P < .0005), indicating efficacy regardless of PRP type.4
Continue to: Ankle osteoarthritis
Ankle osteoarthritis
❯ ❯ ❯ Additional research is needed
Ankle OA affects 3.4% of all adults and is more common in the younger population than knee or hip OA.16 An RCT (N = 100) investigating PRP vs placebo (saline) injections showed no statistically significant difference in American Orthopedic Foot and Ankle Society scores evaluating pain and function over 26 weeks (–2 points; 95% CI, –5 to 1; P = .16).16 Limitations to this study include its small sample size and the PRP formulation used. (The intervention group received 2 injections of 2 mL of PRP, and the platelet concentration was not reported.)16
Hip osteoarthritis
❯ ❯ ❯ Additional research is needed
Symptomatic hip OA occurs in 40% of adults older than 65 years, with a higher prevalence in women.18 Currently, corticosteroid injections are the only intra-articular therapy recommended by international guidelines for hip OA.19 A systematic review and meta-analysis comparing PRP to HA injections that included 4 RCTs (N = 303) showed a statistically significant reduction in VAS scores at 2 months in the PRP group compared to the HA group (weighted mean difference [WMD] = –0.376; 95% CI, –0.614 to –0.138; P = .002).18 However, there were no significant differences in VAS scores between the PRP and HA groups at 6 months (WMD = –0.141; 95% CI, –0.401 to 0.119; P = .289) and 12 months (WMD = –0.083; 95% CI, –0.343 to 0.117; P = .534). Likewise, no significant differences were found in WOMAC scores at 6 months (WMD = –2.841; 95% CI, –6.248 to 0.565; P = .102) and 12 months (WMD = –3.134; 95% CI, –6.624 to 0.356; P = .078) and Harris Hip Scores (HHS) at 6 months (WMD = 2.782; 95% CI, –6.639 to 12.203; P =.563) and 12 months (WMD = 0.706; 95% CI, –6.333 to 7.745; P = .844).18
A systematic review of 6 RCTs (N = 408) by Belk et al20 comparing PRP to HA for hip OA found similar short-term improvements in WOMAC scores (standardized mean differences [SMD] = 0.27; 95% CI, –0.05 to 0.59; P = .09), VAS scores (MD = 0.59; 95% CI, –0.741 to 1.92; P = .39), and HHS (MD = -0.81; 95% CI, –10.06 to 8.43; P = .93).The average follow-up time was 12.2 and 11.9 months for the PRP and HA groups, respectively.20
LR-PRP, which was used in 1 of the 6 RCTs, showed improvement in VAS scores and HHS from baseline, but no significant difference compared to HA at the latest follow-up.20 A pooled subanalysis of the 3 studies that used LP-PRP found no difference in WOMAC scores between the PRP and HA groups (SMD = 0.42; 95% CI, –0.01 to 0.86; P = .06).20 Future studies comparing the efficacy of intra-articular steroid vs PRP for hip OA would be beneficial.18
Continue to: Rotator cuff tendinopathy
Rotator cuff tendinopathy
❯ ❯ ❯ Consider PRP for short-term pain relief
Painful conditions of the rotator cuff include impingement syndrome, tendonitis, and partial and complete tears. A 2021 RCT (N = 58) by Dadgostar et al21 comparing PRP injection to corticosteroid therapy (methylprednisolone and lidocaine) for the treatment of rotator cuff tendinopathy showed significant improvement in VAS scores at 3 months in the PRP group compared to the corticosteroid group (6.66
Another RCT (N = 99) by Kwong et al22 comparing PRP to corticosteroids found similar short-term advantages of LP-PRP with an improved VAS score (–13.6 vs 0.4; P = .03), American Shoulder and Elbow Surgeons score (13.0 vs 2.9; P = .02), and Western Ontario Rotator Cuff Index score (16.8 vs 5.8; P = .03).However, there was no long-term benefit of PRP over corticosteroids found at 12 months.22
A 2021 systematic review and meta-analysis by Hamid et al23 that included 8 RCTs (N = 976) favored PRP over control (no injection, saline injections, and/or shoulder rehabilitation) with improved VAS scores at 12 months (SMD = –0.5; 95% CI, –0.7 to –0.2; P < .001).The evidence on functional outcome was mixed. Data pooled from 2 studies (n = 228) found better Shoulder Pain and Disability Index (SPADI) scores compared to controls at 3- and 6-month follow-ups. However, there were no significant differences in Disabilities of the Arm, Shoulder and Hand (DASH) scores between the 2 groups.23
Patellar tendinopathy
❯ ❯ ❯ Consider using PRP for return to sport
Patellar tendinopathy, a common MSK condition encountered in the primary care setting, has an overall prevalence of 22% in elite athletes at some point in their career.24 Nonsurgical management options include rest, ice, eccentric and isometric exercises, anti-inflammatory drugs, extracorporeal shock wave therapy (ESWT), and dry needling (DN).
A 2014 RCT (N = 23) evaluating DN vs PRP for patellar tendinopathy favored PRP with improved VAS scores (mean ± SD = 25.4 ± 23.2 points; P = .01 vs 5.2 ± 12.5 points; P = .20) at 12 weeks (P = .02). However, at ≥ 26 weeks, the improvement in pain and function scores was similar between the DN and PRP groups (33.2 ± 14.0 points; P = .001 vs 28.9 ± 25.2 points; P = .01). Notably, there was significantly more improvement in the PRP group at 12 weeks (P = .02) but not at 26 weeks (P = .66).25
Continue to: Another perspective study...
Another prospective study (N = 31) comparing PRP to physiotherapy showed a greater improvement in sport activity level reflected by the Tegner score in the PRP group (percentage improvement, 39 ± 22%) compared to control (20 ± 27%; P = .048) at 6 months.7
A recent RCT (N = 20) revealed improved VAS scores at 6 months with rehabilitation paired with either bone marrow mesenchymal stem cells (BM-MSC) or LP-PRP when compared with baseline (BM-MSC group: 4.23 ± 2.13 to 2.52 ± 2.37; P = .0621; LP-PRP group: 3.10 ± 1.20 to 1.13 ± 1.25; P = .0083). Pain was significantly reduced during sport play in both groups at 6 months when compared with baseline (BM-MSC group: 6.91 ± 1.11 to 3.06 ± 2.89, P = .0049; PRP group: 7.03 ± 1.42 to 1.94 ± 1.24, P = .0001).26
A 2019 systematic review and meta-analysis (N = 2530) demonstrated greater improvements in Victorian Institute of Sport Assessment scale for patellar tendinopathy (VISA-P) with multiple injections of PRP (38.7 points; 95% CI, 26.3-51.2 points) compared to single injections of PRP (24.3 points; 95% CI, 18.2-30.5 points), eccentric exercise (28.3 points; 95% CI, 18.9-37.8 points) and ESWT (27.4 points; 95% CI, 10.0-39.8 points) after 6 months.27 In contrast, an RCT (n = 57) comparing a single injection of LR-PRP or LP-PRP was no more effective than a single injection of saline for improvement in mean VISA-P scores (P > .05) at 1 year.28
Lateral epicondylitis
❯ ❯ ❯ Consider using PRP
Lateral epicondylitis (“tennis elbow”) is caused by overuse of the elbow extensors at the site of the lateral epicondyle. Chronic lateral epicondylosis involves tissue degeneration and microtrauma.Most cases of epicondylar tendinopathies are treated nonoperatively, with corticosteroid injections being a mainstay of treatment despite their short-term benefit29 and potential to deteriorate connective tissue over time. Recent studies suggest PRP therapy for epicondylitis and epicondylosis may increase long-term pain relief and improve function.
A 2017 systematic review and meta-analysis of 16 RCTs (N = 1018) concluded PRP was more efficacious than control injections (bupivacaine) for pain reduction in tendinopathies (effect size = 0.47; 95% CI, 0.22-0.72).30 In the review, lateral epicondylitis was evaluated in 12 studies and was most responsive to PRP (effect size = 0.57) when compared to control injection.30 In another systematic review (5 RCTs; 250 patients), corticosteroid injections improved pain within the first 6 weeks of treatment. However, PRP outperformed corticosteroid in VAS scores (21.3 ± 28.1 vs 42.4 ± 26.8) and DASH scores (17.6 ± 24.0 vs 36.5 ± 23.8) (P < .001) at 2 years.31
Continue to: A 2022 systematic review...
A 2022 systematic review and meta-analysis (26 studies; N = 1040) comparing scores at baseline vs 2 years post-PRP showed improvement in VAS scores (7.4 ± 1.30 vs 3.71 ± 2.35; P < .001), DASH scores (60.8 ± 12.5 vs 13.0 ± 18.5; P < .001), Patient-Rated Tennis Elbow Evaluation (55.6 ± 14.7 vs 48.8 ± 4.1; P < .001), and Mayo Clinic Performance Index (55.5 ± 6.1 vs 93.0 ± 6.7; P < .001).32
Regarding the therapeutic effects of different PRP types in lateral epicondylitis, a 2022 systematic review of 33 studies (N = 2420) found improved function and pain relief with LR-PRP and LP-PRP with no significant differences.33 Pretreatment VAS scores in the LR-PRP group, which ranged from 6.1 to 8.0, improved to 1.5 to 4.0 at 3 months and 0.6 to 3.3 after 1 year.33 Similarly, pretreatment VAS scores in the LP-PRP group, which ranged from 4.2 to 8.4, improved to 1.6 to 5.9 at 3 months and 0.7 to 2.7 after 1 year.34 DASH scores also improved in the LR-PRP and LP-PRP groups, with pretreatment scores (LR-PRP, 47.0 to 54.3; LP-PRP, 30.0 to 67.7) improving to 20.0 to 22.0 and 5.5 to 19.0, respectively, at 1 year.33
Achilles tendinopathy
❯ ❯ ❯ Do not use PRP; evidence is lacking
Achilles tendinopathy, caused by chronic overuse and overload resulting in microtrauma and poor tissue healing, typically occurs in the most poorly vascularized portion of the tendon and is common in runners. First-line treatments for Achilles tendinopathy include eccentric strength training and anti-inflammatory drugs.34,35 Corticosteroid injections are not recommended, given concern for degraded tendon tissue over time and worse function.34
A 2020 systematic review of 11 randomized and nonrandomized clinical trials (N = 406) found PRP improved Victorian Institute of Sports Assessment—Achilles (VISA-A) scores at 24 weeks compared to other nonsurgical treatment options (41.2 vs 70.12; P < .018).34 However, a higher-quality 2021 systematic review and meta-analysis of 4 RCTs (N = 170) comparing PRP injections with placebo showed no significant difference in VISA-A scores at 3 months (0.23; 95% CI, –0.45 to 0.91), 6 months (0.83; 95% CI, –0.26 to 1.92), and 12 months (0.83; 95% CI, –0.77 to 2.44).36 Therefore, further studies are warranted to evaluate the benefit of PRP injections for Achilles tendinopathy.
Conclusions
While high-quality studies support the use of PRP for knee OA and lateral epicondylitis, they have a moderate-to-high risk for bias. Several RCTs show that PRP provides superior short-term pain relief and range of motion compared to corticosteroids for rotator cuff tendinopathy. Multiple injections of PRP for patellar tendinopathy may accelerate return to sport and improve symptoms over the long term. However, current evidence does not support PRP therapy for Achilles tendinopathy. Given variability in PRP preparation, an accurate interpretation of the literature regarding its use in MSK conditions is recommended (TABLE4,6,7,14-18,20-23,25-28,30-34,36).
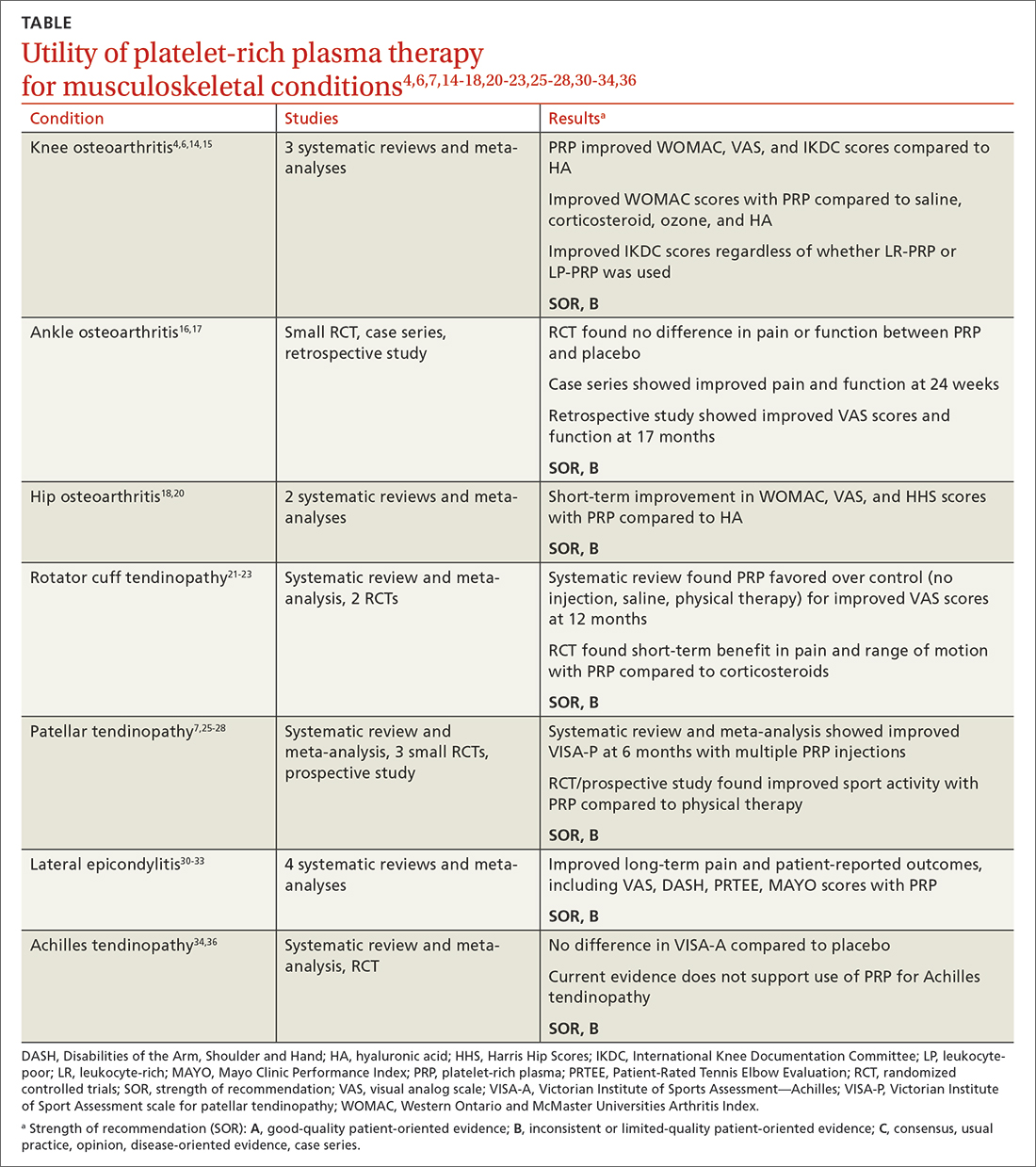
Continue to: Concerning the effectiveness of PRP...
Concerning the effectiveness of PRP, it is important to consider early publication bias. Although recent studies have shown its benefits,6,14,15,37 additional studies comparing PRP to placebo will help demonstrate its efficacy. Interestingly, a literature search by Bar-Or et al38 found intra-articular saline may have a therapeutic effect on knee OA and confound findings when used as a placebo.
Recognizing the presence or lack of clinically significant improvement in the literature is important. For example, while some recent studies have shown PRP exceeds the minimal clinically significant difference for knee OA and lateral epicondylitis, others have not.32,37 A 2021 systematic review of 11 clinical practice guidelines for the use of PRP in knee OA found that 9 were “uncertain or unable to make a recommendation” and 2 recommended against it.39
In its 2021 position statement for the responsible use of regenerative medicine, the American Medical Society for Sports Medicine includes guidance on integrating orthobiologics into clinical practice. The guideline emphasizes informed consent and provides an evidence-based rationale for using PRP in certain patient populations (lateral epicondylitis and younger patients with mild-to-moderate knee OA), recommending its use only after exhausting other conservative options.40 Patients should be referred to physicians with experience using PRP and image-guided procedures.
CORRESPONDENCE
Gregory D. Bentz Jr, MD, 3640 High Street Suite 3B, Portsmouth, VA 23707; [email protected]
1. Cecerska-Heryć E, Goszka M, Serwin N, et al. Applications of the regenerative capacity of platelets in modern medicine. Cytokine Growth Factor Rev. 2022;64:84-94. doi: 10.1016/j.cytogfr.2021.11.003
2. Le ADK, Enweze L, DeBaun MR, et al. Current clinical recommendations for use of platelet-rich plasma. Curr Rev Musculoskelet Med. 2018;11:624-634. doi: 10.1007/s12178-018-9527-7
3. Everts P, Onishi K, Jayaram P, et al. Platelet-rich plasma: new performance understandings and therapeutic considerations in 2020. Int J Mol Sci. 2020;21:7794. doi: 10.3390/ijms21207794
4. Di Martino A, Boffa A, Andriolo L, et al. Leukocyte-rich versus leukocyte-poor platelet-rich plasma for the treatment of knee osteoarthritis: a double-blind randomized trial. Am J Sports Med. 2022;50:609-617. doi: 10.1177/03635465211064303
5. Mariani E, Pulsatelli L. Platelet concentrates in musculoskeletal medicine. Int J Mol Sci. 2020;21:1328. doi: 10.3390/ijms21041328
6. Belk JW, Kraeutler MJ, Houck DA, et al. Platelet-rich plasma versus hyaluronic acid for knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Am J Sports Med. 2021;49:249-260. doi: 10.1177/0363546520909397
7. Filardo G, Kon E, Della Villa S, et al. Use of platelet-rich plasma for the treatment of refractory jumper’s knee. Int Orthop. 2010;34:909-915. doi: 10.1007/s00264-009-0845-7
8. Kon E, Filardo G, Delcogliano M, et al. Platelet-rich plasma: new clinical application: a pilot study for treatment of jumper’s knee. Injury. 2009;40:598-603. doi: 10.1016/j.injury.2008.11.026
9. Kanchanatawan W, Arirachakaran A, Chaijenkij K, et al. Short-term outcomes of platelet-rich plasma injection for treatment of osteoarthritis of the knee. Knee Surg Sports Traumatol Arthrosc. 2016;24:1665-1677. doi: 10.1007/s00167-015-3784-4
10. Cook J, Young M. Biologic therapies for tendon and muscle injury. UpToDate. Updated August 11, 2022. Accessed May 23, 2023. www.uptodate.com/contents/biologic-therapies-for-tendon-and-muscle-injury
11. Bendich I, Rubenstein WJ, Cole BJ, et al. What is the appropriate price for platelet-rich plasma injections for knee osteoarthritis? A cost-effectiveness analysis based on evidence from Level I randomized controlled trials. Arthroscopy. 2020;36:1983-1991.e1. doi: 10.1016/j.arthro.2020.02.004
12. Jones IA, Togashi RC, Thomas Vangsness C Jr. The economics and regulation of PRP in the evolving field of orthopedic biologics. Curr Rev Musculoskelet Med. 2018;11:558-565. doi: 10.1007/s12178-018-9514-z
13. Costa LAV, Lenza M, Irrgang JJ, et al. How does platelet-rich plasma compare clinically to other therapies in the treatment of knee osteoarthritis? A systematic review and meta-analysis. Am J Sports Med. 2023;51:1074-1086 doi: 10.1177/03635465211062243
14. Meheux CJ, McCulloch PC, Lintner DM, et al. Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy. 2016;32:495-505. doi: 10.1016/j.arthro.2015.08.005
15. Shen L, Yuan T, Chen S, et al. The temporal effect of platelet-rich plasma on pain and physical function in the treatment of knee osteoarthritis: systematic review and meta-analysis of randomized controlled trials. J Orthop Surg Res. 2017;12:16. doi: 10.1186/s13018-017-0521-3
16. Paget LDA, Reurink G, de Vos RJ, et al; PRIMA Study Group. Effect of platelet-rich plasma injections vs. placebo on ankle symptoms and function in patients with ankle osteoarthritis: a randomized clinical trial. JAMA. 2021;326:1595-1605. doi: 10.1001/jama.2021.16602
17. Evans A, Ibrahim M, Pope R, et al. Treating hand and foot osteoarthritis using a patient’s own blood: a systematic review and meta-analysis of platelet-rich plasma. J Orthop. 2020;18:226-236. doi: 10.1016/j.jor.2020.01.037
18. Ye Y, Zhou X, Mao S, et al. Platelet rich plasma versus hyaluronic acid in patients with hip osteoarthritis: a meta-analysis of randomized controlled trials. Int J Surg. 2018;53:279-287. doi: 10.1016/j.ijsu.2018.03.078.
19. Berney M, McCarroll P, Glynn L, et al. Platelet-rich plasma injections for hip osteoarthritis: a review of the evidence. Ir J Med Sci. 2021;190:1021-1025. doi: 10.1007/s11845-020-02388-z
20. Belk JW, Houck DA, Littlefield CP, et al. Platelet-rich plasma versus hyaluronic acid for hip osteoarthritis yields similarly beneficial short-term clinical outcomes: a systematic review and meta-analysis of Level I and II randomized controlled trials. Arthroscopy. 2022;38:2035-2046. doi: 10.1016/j.arthro.2021.11.005
21. Dadgostar H, Fahimipour F, Pahlevan Sabagh A, et al. Corticosteroids or platelet-rich plasma injections for rotator cuff tendinopathy: a randomized clinical trial study. J Orthop Surg Res. 2021;16:333. doi: 10.1186/s13018-021-02470-x
22. Kwong CA, Woodmass JM, Gusnowski EM, et al. Platelet-rich plasma in patients with partial-thickness rotator cuff tears or tendinopathy leads to significantly improved short-term pain relief and function compared with corticosteroid injection: a double-blind randomized controlled trial. Arthroscopy. 2021;37:510-517. doi: 10.1016/j.arthro.2020.10.037
23. A Hamid MS, Sazlina SG. Platelet-rich plasma for rotator cuff tendinopathy: a systematic review and meta-analysis. PLoS One. 2021;16:e0251111. doi: 10.1371/journal.pone.0251111
24. Lian OB, Engebretsen L, Bahr R. Prevalence of jumper’s knee among elite athletes from different sports: a cross-sectional study. Am J Sports Med. 2005;33:561-567. doi: 10.1177/0363546504270454
25. Dragoo JL, Wasterlain AS, Braun HJ, et al. Platelet-rich plasma as a treatment for patellar tendinopathy: a double-blind, randomized controlled trial. Am J Sports Med. 2014;42:610-618. doi: 10.1177/0363546513518416.
26. Rodas G, Soler-Rich R, Rius-Tarruella J, et al. Effect of autologous expanded bone marrow mesenchymal stem cells or leukocyte-poor platelet-rich plasma in chronic patellar tendinopathy (with gap >3 mm): preliminary outcomes after 6 months of a double-blind, randomized, prospective study. Am J Sports Med. 2021;49:1492-1504. doi: 10.1177/0363546521998725
27. Andriolo L, Altamura SA, Reale D, et al. Nonsurgical treatments of patellar tendinopathy: multiple injections of platelet-rich plasma are a suitable option: a systematic review and meta-analysis. Am J Sports Med. 2019;47:1001-1018. doi: 10.1177/0363546518759674
28. Scott A, LaPrade RF, Harmon KG, et al. Platelet-rich plasma for patellar tendinopathy: a randomized controlled trial of leukocyte-rich PRP or leukocyte-poor PRP versus saline. Am J Sports Med. 2019;47:1654-1661. doi: 10.1177/0363546519837954
29. Kemp JA, Olson MA, Tao MA, et al. Platelet-rich plasma versus corticosteroid injection for the treatment of lateral epicondylitis: a systematic review of systematic reviews. Int J Sports Phys Ther. 2021;16:597-605. doi: 10.26603/001c.24148
30. Miller LE, Parrish WR, Roides B, et al. Efficacy of platelet-rich plasma injections for symptomatic tendinopathy: systematic review and meta-analysis of randomised injection-controlled trials. BMJ Open Sport Exerc Med. 2017;3:e000237. doi: 10.1136/bmjsem-2017- 000237
31. Ben-Nafa W, Munro W. The effect of corticosteroid versus platelet-rich plasma injection therapies for the management of lateral epicondylitis: a systematic review. SICOT J. 2018;4:11.
32. Niemiec P, Szyluk K, Jarosz A, et al. Effectiveness of platelet-rich plasma for lateral epicondylitis: a systematic review and meta-analysis based on achievement of minimal clinically important difference. Orthop J Sports Med. 2022;10:23259671221086920. doi: 10.1177/23259671221086920
33. Li S, Yang G, Zhang H, et al. A systematic review on the efficacy of different types of platelet-rich plasma in the management of lateral epicondylitis. J Shoulder Elbow Surg. 2022;311533-1544. doi: 10.1016/j.jse.2022.02.017.
34. Madhi MI, Yausep OE, Khamdan K, et al. The use of PRP in treatment of Achilles tendinopathy: a systematic review of literature. Study design: systematic review of literature. Ann Med Surg (Lond). 2020;55:320-326. doi: 10.1016/j.amsu.2020.04.042
35. Loppini M, Maffulli N. Conservative management of tendinopathy: an evidence-based approach. Muscles Ligaments Tendons J. 2012;1:134-137.
36. Nauwelaers AK, Van Oost L, Peers K. Evidence for the use of PRP in chronic midsubstance Achilles tendinopathy: a systematic review with meta-analysis. Foot Ankle Surg. 2021;27:486-495. doi: 10.1016/j.fas.2020.07.009
37. Dai WL, Zhou AG, Zhang H, et al. Efficacy of platelet-rich plasma in the treatment of knee osteoarthritis: a meta-analysis of randomized controlled trials. Arthroscopy. 2017;33:659-670.e1. doi: 10.1016/j.arthro.2016.09.024
38. Bar-Or D, Rael LT, Brody EN. Use of saline as a placebo in intra-articular injections in osteoarthritis: potential contributions to nociceptive pain relief. Open Rheumatol J. 2017;11:16-22. doi: 10.2174/1874312901711010016
39. Phillips M, Bhandari M, Grant J, et al. A systematic review of current clinical practice guidelines on intra-articular hyaluronic acid, corticosteroid, and platelet-rich plasma injection for knee osteoarthritis: an international perspective. Orthop J Sports Med. 2021;9:23259671211030272. doi: 10.1177/23259671211030272
40. Finnoff JT, Awan TM, Borg-Stein J, et al. American Medical Society for Sports Medicine position statement: principles for the responsible use of regenerative medicine in sports medicine. Clin J Sport Med. 2021;31:530-541. doi: 10.1097/JSM.0000000000000973
Platelet-rich plasma (PRP) injections have become a popular treatment option in a variety of specialties including sports medicine, maxillofacial surgery, dermatology, cosmetology, and reproductive medicine.1 PRP is an autologous blood product derived from whole blood, using a centrifuge to isolate a concentrated layer of platelets. The a-granules in platelets release transforming growth factor b 1, vascular endothelial growth factor, platelet-derived growth factor, basic fibroblast growth factor, epidermal growth factor, insulin-like growth factor 1, and other mediatorsthat enhance the natural healing process.2

When patients ask. Familiarity with the use of PRP to treat specific musculoskeletal (MSK) conditions is essential for family physicians who frequently are asked by patients about whether PRP is right for them. These patients may have experienced failure of medication therapy or declined surgical intervention, or may not be surgical candidates. This review details the evidence surrounding common intra-articular and extra-articular applications of PRP. But first, a word about how PRP is prepared, its contraindications, and costs.
Preparation and types of PRP
Although there are many commercial systems for preparing PRP, there is no consensus on the optimal formulation.2 Other terms for PRP, such as autologous concentrated platelets and super-concentrated platelets, are based on concentration of red blood cells, leukocytes, and fibrin.3 PRP therapies usually are categorized as leukocyte-rich PRP (LR-PRP) or leukocyte-poor PRP (LP-PRP), based on neutrophil concentrations that are above and below baseline.2 Leukocyte concentration is one of the most debated topics in PRP therapy.4
Common commercially available preparation systems produce platelet concentrations between 3 to 6 times the baseline platelet count.5 Although there is no universally agreed upon PRP formulation, studies have shown 2 centrifugation cycles (“double-spun” or “dual centrifugation”) that yield platelet concentrations between 1.8 to 1.9 times the baseline values significantly improve MSK conditions.6-8
For MSK purposes, PRP may be injected into intratendinous, peritendinous, and intra-articular spaces. Currently, there is no consensus regarding injection frequency. Many studies have incorporated single-injection protocols, while some have used 2 to 3 injections repeated over several weeks to months. PRP commonly is injected at point-of-care without requiring storage.
Contraindications. PRP has been shown to be safe, with most adverse effects attributed to local injection site pain, bleeding, swelling, and bruising.9
Contraindications to PRP include active malignancy or recent remission from malignancy with the exception of nonmetastatic skin tumors.10 PRP is not recommended for patients with an allergy to manufacturing components (eg, dimethyl sulfoxide), thrombocytopenia, nonsteroidal anti-inflammatory drug use within 2 weeks, active infection causing fever, and local infection at the injection site.10 Since local anesthetics may impair platelet function, they should not be given at the same injection site as PRP.10
Continue to: Cost
Cost. PRP is not covered by most insurance plans.11,12 The cost for PRP may range from $500 to $2500 for a single injection.12
Evidence-based summary by condition
Knee osteoarthritis
❯❯❯ Consider using PRP
Knee osteoarthritis (OA) is a common cause of pain and disability. Treatment options include physical therapy, pharmacotherapy, and surgery. PRP has gained popularity as a nonsurgical option. A recent meta-analysis by Costa et al13 of 40 studies with 3035 participants comparing intra-articular PRP with hyaluronic acid (HA), corticosteroid, and saline injections, found that PRP appears to be more effective or as effective as other nonsurgical modalities. However, due to study heterogeneity and high risk for bias, the authors could not recommend PRP for knee OA in clinical practice.13
Despite Costa et al’s findings, reproducible data have demonstrated the superiority of PRP over other nonsurgical treatment options for knee OA. A 2021 systematic review and meta-analysis of 18 randomized controlled trials (RCTs; N = 811) by Belk et al6 comparing PRP to HA injections showed a higher mean improvement in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores in the PRP group compared to the HA group (44.7% vs 12.6%, respectively; P < .01).6 Six of 11 studies using the visual analog scale (VAS) for pain reported significantly less pain in the PRP group compared to the HA group (P < .05).6 The mean follow-up time was 11.1 months.6 Three of 6 studies reported improved subjective International Knee Documentation Committee (IKDC) scores (range from 0-100, with higher scores representing higher levels of function and lower levels of symptoms) in the PRP group compared to the HA group: 75.7 ± 15.1 vs 65.6 ± 16.9 (P = .004); 65.5 ± 3.6 vs 55.8 ± 3.8 (P = .01); and 60.8 ± 9.8 vs 48.4 ± 6.2 (P < .05).6 There was concern for moderate-to-high heterogeneity.6
Other systematic reviews and meta-analyses found similar efficacy of PRP for knee OA, including improved WOMAC scores and patient-reported outcomes (eg, pain, physical function, stiffness) compared to other injectable options.14,15 A systematic review of 14 RCTs (N = 1423) by Shen et al15 showed improved WOMAC scores at 3 months (mean differences [MD] = –14.53; 95% CI, –29.97 to –7.09; P < .001), 6 months
Despite a lack of consensus regarding the optimal preparation of PRP for knee OA, another recent RCT (N = 192) found significant improvement in mean subjective IKDC scores in the LR-PRP group (45.5 ± 15.5 to 60.7 ± 21.1; P < .0005) and the LP-PRP group (46.8 ± 15.8 to 62.9 ± 19.9; P < .0005), indicating efficacy regardless of PRP type.4
Continue to: Ankle osteoarthritis
Ankle osteoarthritis
❯ ❯ ❯ Additional research is needed
Ankle OA affects 3.4% of all adults and is more common in the younger population than knee or hip OA.16 An RCT (N = 100) investigating PRP vs placebo (saline) injections showed no statistically significant difference in American Orthopedic Foot and Ankle Society scores evaluating pain and function over 26 weeks (–2 points; 95% CI, –5 to 1; P = .16).16 Limitations to this study include its small sample size and the PRP formulation used. (The intervention group received 2 injections of 2 mL of PRP, and the platelet concentration was not reported.)16
Hip osteoarthritis
❯ ❯ ❯ Additional research is needed
Symptomatic hip OA occurs in 40% of adults older than 65 years, with a higher prevalence in women.18 Currently, corticosteroid injections are the only intra-articular therapy recommended by international guidelines for hip OA.19 A systematic review and meta-analysis comparing PRP to HA injections that included 4 RCTs (N = 303) showed a statistically significant reduction in VAS scores at 2 months in the PRP group compared to the HA group (weighted mean difference [WMD] = –0.376; 95% CI, –0.614 to –0.138; P = .002).18 However, there were no significant differences in VAS scores between the PRP and HA groups at 6 months (WMD = –0.141; 95% CI, –0.401 to 0.119; P = .289) and 12 months (WMD = –0.083; 95% CI, –0.343 to 0.117; P = .534). Likewise, no significant differences were found in WOMAC scores at 6 months (WMD = –2.841; 95% CI, –6.248 to 0.565; P = .102) and 12 months (WMD = –3.134; 95% CI, –6.624 to 0.356; P = .078) and Harris Hip Scores (HHS) at 6 months (WMD = 2.782; 95% CI, –6.639 to 12.203; P =.563) and 12 months (WMD = 0.706; 95% CI, –6.333 to 7.745; P = .844).18
A systematic review of 6 RCTs (N = 408) by Belk et al20 comparing PRP to HA for hip OA found similar short-term improvements in WOMAC scores (standardized mean differences [SMD] = 0.27; 95% CI, –0.05 to 0.59; P = .09), VAS scores (MD = 0.59; 95% CI, –0.741 to 1.92; P = .39), and HHS (MD = -0.81; 95% CI, –10.06 to 8.43; P = .93).The average follow-up time was 12.2 and 11.9 months for the PRP and HA groups, respectively.20
LR-PRP, which was used in 1 of the 6 RCTs, showed improvement in VAS scores and HHS from baseline, but no significant difference compared to HA at the latest follow-up.20 A pooled subanalysis of the 3 studies that used LP-PRP found no difference in WOMAC scores between the PRP and HA groups (SMD = 0.42; 95% CI, –0.01 to 0.86; P = .06).20 Future studies comparing the efficacy of intra-articular steroid vs PRP for hip OA would be beneficial.18
Continue to: Rotator cuff tendinopathy
Rotator cuff tendinopathy
❯ ❯ ❯ Consider PRP for short-term pain relief
Painful conditions of the rotator cuff include impingement syndrome, tendonitis, and partial and complete tears. A 2021 RCT (N = 58) by Dadgostar et al21 comparing PRP injection to corticosteroid therapy (methylprednisolone and lidocaine) for the treatment of rotator cuff tendinopathy showed significant improvement in VAS scores at 3 months in the PRP group compared to the corticosteroid group (6.66
Another RCT (N = 99) by Kwong et al22 comparing PRP to corticosteroids found similar short-term advantages of LP-PRP with an improved VAS score (–13.6 vs 0.4; P = .03), American Shoulder and Elbow Surgeons score (13.0 vs 2.9; P = .02), and Western Ontario Rotator Cuff Index score (16.8 vs 5.8; P = .03).However, there was no long-term benefit of PRP over corticosteroids found at 12 months.22
A 2021 systematic review and meta-analysis by Hamid et al23 that included 8 RCTs (N = 976) favored PRP over control (no injection, saline injections, and/or shoulder rehabilitation) with improved VAS scores at 12 months (SMD = –0.5; 95% CI, –0.7 to –0.2; P < .001).The evidence on functional outcome was mixed. Data pooled from 2 studies (n = 228) found better Shoulder Pain and Disability Index (SPADI) scores compared to controls at 3- and 6-month follow-ups. However, there were no significant differences in Disabilities of the Arm, Shoulder and Hand (DASH) scores between the 2 groups.23
Patellar tendinopathy
❯ ❯ ❯ Consider using PRP for return to sport
Patellar tendinopathy, a common MSK condition encountered in the primary care setting, has an overall prevalence of 22% in elite athletes at some point in their career.24 Nonsurgical management options include rest, ice, eccentric and isometric exercises, anti-inflammatory drugs, extracorporeal shock wave therapy (ESWT), and dry needling (DN).
A 2014 RCT (N = 23) evaluating DN vs PRP for patellar tendinopathy favored PRP with improved VAS scores (mean ± SD = 25.4 ± 23.2 points; P = .01 vs 5.2 ± 12.5 points; P = .20) at 12 weeks (P = .02). However, at ≥ 26 weeks, the improvement in pain and function scores was similar between the DN and PRP groups (33.2 ± 14.0 points; P = .001 vs 28.9 ± 25.2 points; P = .01). Notably, there was significantly more improvement in the PRP group at 12 weeks (P = .02) but not at 26 weeks (P = .66).25
Continue to: Another perspective study...
Another prospective study (N = 31) comparing PRP to physiotherapy showed a greater improvement in sport activity level reflected by the Tegner score in the PRP group (percentage improvement, 39 ± 22%) compared to control (20 ± 27%; P = .048) at 6 months.7
A recent RCT (N = 20) revealed improved VAS scores at 6 months with rehabilitation paired with either bone marrow mesenchymal stem cells (BM-MSC) or LP-PRP when compared with baseline (BM-MSC group: 4.23 ± 2.13 to 2.52 ± 2.37; P = .0621; LP-PRP group: 3.10 ± 1.20 to 1.13 ± 1.25; P = .0083). Pain was significantly reduced during sport play in both groups at 6 months when compared with baseline (BM-MSC group: 6.91 ± 1.11 to 3.06 ± 2.89, P = .0049; PRP group: 7.03 ± 1.42 to 1.94 ± 1.24, P = .0001).26
A 2019 systematic review and meta-analysis (N = 2530) demonstrated greater improvements in Victorian Institute of Sport Assessment scale for patellar tendinopathy (VISA-P) with multiple injections of PRP (38.7 points; 95% CI, 26.3-51.2 points) compared to single injections of PRP (24.3 points; 95% CI, 18.2-30.5 points), eccentric exercise (28.3 points; 95% CI, 18.9-37.8 points) and ESWT (27.4 points; 95% CI, 10.0-39.8 points) after 6 months.27 In contrast, an RCT (n = 57) comparing a single injection of LR-PRP or LP-PRP was no more effective than a single injection of saline for improvement in mean VISA-P scores (P > .05) at 1 year.28
Lateral epicondylitis
❯ ❯ ❯ Consider using PRP
Lateral epicondylitis (“tennis elbow”) is caused by overuse of the elbow extensors at the site of the lateral epicondyle. Chronic lateral epicondylosis involves tissue degeneration and microtrauma.Most cases of epicondylar tendinopathies are treated nonoperatively, with corticosteroid injections being a mainstay of treatment despite their short-term benefit29 and potential to deteriorate connective tissue over time. Recent studies suggest PRP therapy for epicondylitis and epicondylosis may increase long-term pain relief and improve function.
A 2017 systematic review and meta-analysis of 16 RCTs (N = 1018) concluded PRP was more efficacious than control injections (bupivacaine) for pain reduction in tendinopathies (effect size = 0.47; 95% CI, 0.22-0.72).30 In the review, lateral epicondylitis was evaluated in 12 studies and was most responsive to PRP (effect size = 0.57) when compared to control injection.30 In another systematic review (5 RCTs; 250 patients), corticosteroid injections improved pain within the first 6 weeks of treatment. However, PRP outperformed corticosteroid in VAS scores (21.3 ± 28.1 vs 42.4 ± 26.8) and DASH scores (17.6 ± 24.0 vs 36.5 ± 23.8) (P < .001) at 2 years.31
Continue to: A 2022 systematic review...
A 2022 systematic review and meta-analysis (26 studies; N = 1040) comparing scores at baseline vs 2 years post-PRP showed improvement in VAS scores (7.4 ± 1.30 vs 3.71 ± 2.35; P < .001), DASH scores (60.8 ± 12.5 vs 13.0 ± 18.5; P < .001), Patient-Rated Tennis Elbow Evaluation (55.6 ± 14.7 vs 48.8 ± 4.1; P < .001), and Mayo Clinic Performance Index (55.5 ± 6.1 vs 93.0 ± 6.7; P < .001).32
Regarding the therapeutic effects of different PRP types in lateral epicondylitis, a 2022 systematic review of 33 studies (N = 2420) found improved function and pain relief with LR-PRP and LP-PRP with no significant differences.33 Pretreatment VAS scores in the LR-PRP group, which ranged from 6.1 to 8.0, improved to 1.5 to 4.0 at 3 months and 0.6 to 3.3 after 1 year.33 Similarly, pretreatment VAS scores in the LP-PRP group, which ranged from 4.2 to 8.4, improved to 1.6 to 5.9 at 3 months and 0.7 to 2.7 after 1 year.34 DASH scores also improved in the LR-PRP and LP-PRP groups, with pretreatment scores (LR-PRP, 47.0 to 54.3; LP-PRP, 30.0 to 67.7) improving to 20.0 to 22.0 and 5.5 to 19.0, respectively, at 1 year.33
Achilles tendinopathy
❯ ❯ ❯ Do not use PRP; evidence is lacking
Achilles tendinopathy, caused by chronic overuse and overload resulting in microtrauma and poor tissue healing, typically occurs in the most poorly vascularized portion of the tendon and is common in runners. First-line treatments for Achilles tendinopathy include eccentric strength training and anti-inflammatory drugs.34,35 Corticosteroid injections are not recommended, given concern for degraded tendon tissue over time and worse function.34
A 2020 systematic review of 11 randomized and nonrandomized clinical trials (N = 406) found PRP improved Victorian Institute of Sports Assessment—Achilles (VISA-A) scores at 24 weeks compared to other nonsurgical treatment options (41.2 vs 70.12; P < .018).34 However, a higher-quality 2021 systematic review and meta-analysis of 4 RCTs (N = 170) comparing PRP injections with placebo showed no significant difference in VISA-A scores at 3 months (0.23; 95% CI, –0.45 to 0.91), 6 months (0.83; 95% CI, –0.26 to 1.92), and 12 months (0.83; 95% CI, –0.77 to 2.44).36 Therefore, further studies are warranted to evaluate the benefit of PRP injections for Achilles tendinopathy.
Conclusions
While high-quality studies support the use of PRP for knee OA and lateral epicondylitis, they have a moderate-to-high risk for bias. Several RCTs show that PRP provides superior short-term pain relief and range of motion compared to corticosteroids for rotator cuff tendinopathy. Multiple injections of PRP for patellar tendinopathy may accelerate return to sport and improve symptoms over the long term. However, current evidence does not support PRP therapy for Achilles tendinopathy. Given variability in PRP preparation, an accurate interpretation of the literature regarding its use in MSK conditions is recommended (TABLE4,6,7,14-18,20-23,25-28,30-34,36).

Continue to: Concerning the effectiveness of PRP...
Concerning the effectiveness of PRP, it is important to consider early publication bias. Although recent studies have shown its benefits,6,14,15,37 additional studies comparing PRP to placebo will help demonstrate its efficacy. Interestingly, a literature search by Bar-Or et al38 found intra-articular saline may have a therapeutic effect on knee OA and confound findings when used as a placebo.
Recognizing the presence or lack of clinically significant improvement in the literature is important. For example, while some recent studies have shown PRP exceeds the minimal clinically significant difference for knee OA and lateral epicondylitis, others have not.32,37 A 2021 systematic review of 11 clinical practice guidelines for the use of PRP in knee OA found that 9 were “uncertain or unable to make a recommendation” and 2 recommended against it.39
In its 2021 position statement for the responsible use of regenerative medicine, the American Medical Society for Sports Medicine includes guidance on integrating orthobiologics into clinical practice. The guideline emphasizes informed consent and provides an evidence-based rationale for using PRP in certain patient populations (lateral epicondylitis and younger patients with mild-to-moderate knee OA), recommending its use only after exhausting other conservative options.40 Patients should be referred to physicians with experience using PRP and image-guided procedures.
CORRESPONDENCE
Gregory D. Bentz Jr, MD, 3640 High Street Suite 3B, Portsmouth, VA 23707; [email protected]
Platelet-rich plasma (PRP) injections have become a popular treatment option in a variety of specialties including sports medicine, maxillofacial surgery, dermatology, cosmetology, and reproductive medicine.1 PRP is an autologous blood product derived from whole blood, using a centrifuge to isolate a concentrated layer of platelets. The a-granules in platelets release transforming growth factor b 1, vascular endothelial growth factor, platelet-derived growth factor, basic fibroblast growth factor, epidermal growth factor, insulin-like growth factor 1, and other mediatorsthat enhance the natural healing process.2

When patients ask. Familiarity with the use of PRP to treat specific musculoskeletal (MSK) conditions is essential for family physicians who frequently are asked by patients about whether PRP is right for them. These patients may have experienced failure of medication therapy or declined surgical intervention, or may not be surgical candidates. This review details the evidence surrounding common intra-articular and extra-articular applications of PRP. But first, a word about how PRP is prepared, its contraindications, and costs.
Preparation and types of PRP
Although there are many commercial systems for preparing PRP, there is no consensus on the optimal formulation.2 Other terms for PRP, such as autologous concentrated platelets and super-concentrated platelets, are based on concentration of red blood cells, leukocytes, and fibrin.3 PRP therapies usually are categorized as leukocyte-rich PRP (LR-PRP) or leukocyte-poor PRP (LP-PRP), based on neutrophil concentrations that are above and below baseline.2 Leukocyte concentration is one of the most debated topics in PRP therapy.4
Common commercially available preparation systems produce platelet concentrations between 3 to 6 times the baseline platelet count.5 Although there is no universally agreed upon PRP formulation, studies have shown 2 centrifugation cycles (“double-spun” or “dual centrifugation”) that yield platelet concentrations between 1.8 to 1.9 times the baseline values significantly improve MSK conditions.6-8
For MSK purposes, PRP may be injected into intratendinous, peritendinous, and intra-articular spaces. Currently, there is no consensus regarding injection frequency. Many studies have incorporated single-injection protocols, while some have used 2 to 3 injections repeated over several weeks to months. PRP commonly is injected at point-of-care without requiring storage.
Contraindications. PRP has been shown to be safe, with most adverse effects attributed to local injection site pain, bleeding, swelling, and bruising.9
Contraindications to PRP include active malignancy or recent remission from malignancy with the exception of nonmetastatic skin tumors.10 PRP is not recommended for patients with an allergy to manufacturing components (eg, dimethyl sulfoxide), thrombocytopenia, nonsteroidal anti-inflammatory drug use within 2 weeks, active infection causing fever, and local infection at the injection site.10 Since local anesthetics may impair platelet function, they should not be given at the same injection site as PRP.10
Continue to: Cost
Cost. PRP is not covered by most insurance plans.11,12 The cost for PRP may range from $500 to $2500 for a single injection.12
Evidence-based summary by condition
Knee osteoarthritis
❯❯❯ Consider using PRP
Knee osteoarthritis (OA) is a common cause of pain and disability. Treatment options include physical therapy, pharmacotherapy, and surgery. PRP has gained popularity as a nonsurgical option. A recent meta-analysis by Costa et al13 of 40 studies with 3035 participants comparing intra-articular PRP with hyaluronic acid (HA), corticosteroid, and saline injections, found that PRP appears to be more effective or as effective as other nonsurgical modalities. However, due to study heterogeneity and high risk for bias, the authors could not recommend PRP for knee OA in clinical practice.13
Despite Costa et al’s findings, reproducible data have demonstrated the superiority of PRP over other nonsurgical treatment options for knee OA. A 2021 systematic review and meta-analysis of 18 randomized controlled trials (RCTs; N = 811) by Belk et al6 comparing PRP to HA injections showed a higher mean improvement in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores in the PRP group compared to the HA group (44.7% vs 12.6%, respectively; P < .01).6 Six of 11 studies using the visual analog scale (VAS) for pain reported significantly less pain in the PRP group compared to the HA group (P < .05).6 The mean follow-up time was 11.1 months.6 Three of 6 studies reported improved subjective International Knee Documentation Committee (IKDC) scores (range from 0-100, with higher scores representing higher levels of function and lower levels of symptoms) in the PRP group compared to the HA group: 75.7 ± 15.1 vs 65.6 ± 16.9 (P = .004); 65.5 ± 3.6 vs 55.8 ± 3.8 (P = .01); and 60.8 ± 9.8 vs 48.4 ± 6.2 (P < .05).6 There was concern for moderate-to-high heterogeneity.6
Other systematic reviews and meta-analyses found similar efficacy of PRP for knee OA, including improved WOMAC scores and patient-reported outcomes (eg, pain, physical function, stiffness) compared to other injectable options.14,15 A systematic review of 14 RCTs (N = 1423) by Shen et al15 showed improved WOMAC scores at 3 months (mean differences [MD] = –14.53; 95% CI, –29.97 to –7.09; P < .001), 6 months
Despite a lack of consensus regarding the optimal preparation of PRP for knee OA, another recent RCT (N = 192) found significant improvement in mean subjective IKDC scores in the LR-PRP group (45.5 ± 15.5 to 60.7 ± 21.1; P < .0005) and the LP-PRP group (46.8 ± 15.8 to 62.9 ± 19.9; P < .0005), indicating efficacy regardless of PRP type.4
Continue to: Ankle osteoarthritis
Ankle osteoarthritis
❯ ❯ ❯ Additional research is needed
Ankle OA affects 3.4% of all adults and is more common in the younger population than knee or hip OA.16 An RCT (N = 100) investigating PRP vs placebo (saline) injections showed no statistically significant difference in American Orthopedic Foot and Ankle Society scores evaluating pain and function over 26 weeks (–2 points; 95% CI, –5 to 1; P = .16).16 Limitations to this study include its small sample size and the PRP formulation used. (The intervention group received 2 injections of 2 mL of PRP, and the platelet concentration was not reported.)16
Hip osteoarthritis
❯ ❯ ❯ Additional research is needed
Symptomatic hip OA occurs in 40% of adults older than 65 years, with a higher prevalence in women.18 Currently, corticosteroid injections are the only intra-articular therapy recommended by international guidelines for hip OA.19 A systematic review and meta-analysis comparing PRP to HA injections that included 4 RCTs (N = 303) showed a statistically significant reduction in VAS scores at 2 months in the PRP group compared to the HA group (weighted mean difference [WMD] = –0.376; 95% CI, –0.614 to –0.138; P = .002).18 However, there were no significant differences in VAS scores between the PRP and HA groups at 6 months (WMD = –0.141; 95% CI, –0.401 to 0.119; P = .289) and 12 months (WMD = –0.083; 95% CI, –0.343 to 0.117; P = .534). Likewise, no significant differences were found in WOMAC scores at 6 months (WMD = –2.841; 95% CI, –6.248 to 0.565; P = .102) and 12 months (WMD = –3.134; 95% CI, –6.624 to 0.356; P = .078) and Harris Hip Scores (HHS) at 6 months (WMD = 2.782; 95% CI, –6.639 to 12.203; P =.563) and 12 months (WMD = 0.706; 95% CI, –6.333 to 7.745; P = .844).18
A systematic review of 6 RCTs (N = 408) by Belk et al20 comparing PRP to HA for hip OA found similar short-term improvements in WOMAC scores (standardized mean differences [SMD] = 0.27; 95% CI, –0.05 to 0.59; P = .09), VAS scores (MD = 0.59; 95% CI, –0.741 to 1.92; P = .39), and HHS (MD = -0.81; 95% CI, –10.06 to 8.43; P = .93).The average follow-up time was 12.2 and 11.9 months for the PRP and HA groups, respectively.20
LR-PRP, which was used in 1 of the 6 RCTs, showed improvement in VAS scores and HHS from baseline, but no significant difference compared to HA at the latest follow-up.20 A pooled subanalysis of the 3 studies that used LP-PRP found no difference in WOMAC scores between the PRP and HA groups (SMD = 0.42; 95% CI, –0.01 to 0.86; P = .06).20 Future studies comparing the efficacy of intra-articular steroid vs PRP for hip OA would be beneficial.18
Continue to: Rotator cuff tendinopathy
Rotator cuff tendinopathy
❯ ❯ ❯ Consider PRP for short-term pain relief
Painful conditions of the rotator cuff include impingement syndrome, tendonitis, and partial and complete tears. A 2021 RCT (N = 58) by Dadgostar et al21 comparing PRP injection to corticosteroid therapy (methylprednisolone and lidocaine) for the treatment of rotator cuff tendinopathy showed significant improvement in VAS scores at 3 months in the PRP group compared to the corticosteroid group (6.66
Another RCT (N = 99) by Kwong et al22 comparing PRP to corticosteroids found similar short-term advantages of LP-PRP with an improved VAS score (–13.6 vs 0.4; P = .03), American Shoulder and Elbow Surgeons score (13.0 vs 2.9; P = .02), and Western Ontario Rotator Cuff Index score (16.8 vs 5.8; P = .03).However, there was no long-term benefit of PRP over corticosteroids found at 12 months.22
A 2021 systematic review and meta-analysis by Hamid et al23 that included 8 RCTs (N = 976) favored PRP over control (no injection, saline injections, and/or shoulder rehabilitation) with improved VAS scores at 12 months (SMD = –0.5; 95% CI, –0.7 to –0.2; P < .001).The evidence on functional outcome was mixed. Data pooled from 2 studies (n = 228) found better Shoulder Pain and Disability Index (SPADI) scores compared to controls at 3- and 6-month follow-ups. However, there were no significant differences in Disabilities of the Arm, Shoulder and Hand (DASH) scores between the 2 groups.23
Patellar tendinopathy
❯ ❯ ❯ Consider using PRP for return to sport
Patellar tendinopathy, a common MSK condition encountered in the primary care setting, has an overall prevalence of 22% in elite athletes at some point in their career.24 Nonsurgical management options include rest, ice, eccentric and isometric exercises, anti-inflammatory drugs, extracorporeal shock wave therapy (ESWT), and dry needling (DN).
A 2014 RCT (N = 23) evaluating DN vs PRP for patellar tendinopathy favored PRP with improved VAS scores (mean ± SD = 25.4 ± 23.2 points; P = .01 vs 5.2 ± 12.5 points; P = .20) at 12 weeks (P = .02). However, at ≥ 26 weeks, the improvement in pain and function scores was similar between the DN and PRP groups (33.2 ± 14.0 points; P = .001 vs 28.9 ± 25.2 points; P = .01). Notably, there was significantly more improvement in the PRP group at 12 weeks (P = .02) but not at 26 weeks (P = .66).25
Continue to: Another perspective study...
Another prospective study (N = 31) comparing PRP to physiotherapy showed a greater improvement in sport activity level reflected by the Tegner score in the PRP group (percentage improvement, 39 ± 22%) compared to control (20 ± 27%; P = .048) at 6 months.7
A recent RCT (N = 20) revealed improved VAS scores at 6 months with rehabilitation paired with either bone marrow mesenchymal stem cells (BM-MSC) or LP-PRP when compared with baseline (BM-MSC group: 4.23 ± 2.13 to 2.52 ± 2.37; P = .0621; LP-PRP group: 3.10 ± 1.20 to 1.13 ± 1.25; P = .0083). Pain was significantly reduced during sport play in both groups at 6 months when compared with baseline (BM-MSC group: 6.91 ± 1.11 to 3.06 ± 2.89, P = .0049; PRP group: 7.03 ± 1.42 to 1.94 ± 1.24, P = .0001).26
A 2019 systematic review and meta-analysis (N = 2530) demonstrated greater improvements in Victorian Institute of Sport Assessment scale for patellar tendinopathy (VISA-P) with multiple injections of PRP (38.7 points; 95% CI, 26.3-51.2 points) compared to single injections of PRP (24.3 points; 95% CI, 18.2-30.5 points), eccentric exercise (28.3 points; 95% CI, 18.9-37.8 points) and ESWT (27.4 points; 95% CI, 10.0-39.8 points) after 6 months.27 In contrast, an RCT (n = 57) comparing a single injection of LR-PRP or LP-PRP was no more effective than a single injection of saline for improvement in mean VISA-P scores (P > .05) at 1 year.28
Lateral epicondylitis
❯ ❯ ❯ Consider using PRP
Lateral epicondylitis (“tennis elbow”) is caused by overuse of the elbow extensors at the site of the lateral epicondyle. Chronic lateral epicondylosis involves tissue degeneration and microtrauma.Most cases of epicondylar tendinopathies are treated nonoperatively, with corticosteroid injections being a mainstay of treatment despite their short-term benefit29 and potential to deteriorate connective tissue over time. Recent studies suggest PRP therapy for epicondylitis and epicondylosis may increase long-term pain relief and improve function.
A 2017 systematic review and meta-analysis of 16 RCTs (N = 1018) concluded PRP was more efficacious than control injections (bupivacaine) for pain reduction in tendinopathies (effect size = 0.47; 95% CI, 0.22-0.72).30 In the review, lateral epicondylitis was evaluated in 12 studies and was most responsive to PRP (effect size = 0.57) when compared to control injection.30 In another systematic review (5 RCTs; 250 patients), corticosteroid injections improved pain within the first 6 weeks of treatment. However, PRP outperformed corticosteroid in VAS scores (21.3 ± 28.1 vs 42.4 ± 26.8) and DASH scores (17.6 ± 24.0 vs 36.5 ± 23.8) (P < .001) at 2 years.31
Continue to: A 2022 systematic review...
A 2022 systematic review and meta-analysis (26 studies; N = 1040) comparing scores at baseline vs 2 years post-PRP showed improvement in VAS scores (7.4 ± 1.30 vs 3.71 ± 2.35; P < .001), DASH scores (60.8 ± 12.5 vs 13.0 ± 18.5; P < .001), Patient-Rated Tennis Elbow Evaluation (55.6 ± 14.7 vs 48.8 ± 4.1; P < .001), and Mayo Clinic Performance Index (55.5 ± 6.1 vs 93.0 ± 6.7; P < .001).32
Regarding the therapeutic effects of different PRP types in lateral epicondylitis, a 2022 systematic review of 33 studies (N = 2420) found improved function and pain relief with LR-PRP and LP-PRP with no significant differences.33 Pretreatment VAS scores in the LR-PRP group, which ranged from 6.1 to 8.0, improved to 1.5 to 4.0 at 3 months and 0.6 to 3.3 after 1 year.33 Similarly, pretreatment VAS scores in the LP-PRP group, which ranged from 4.2 to 8.4, improved to 1.6 to 5.9 at 3 months and 0.7 to 2.7 after 1 year.34 DASH scores also improved in the LR-PRP and LP-PRP groups, with pretreatment scores (LR-PRP, 47.0 to 54.3; LP-PRP, 30.0 to 67.7) improving to 20.0 to 22.0 and 5.5 to 19.0, respectively, at 1 year.33
Achilles tendinopathy
❯ ❯ ❯ Do not use PRP; evidence is lacking
Achilles tendinopathy, caused by chronic overuse and overload resulting in microtrauma and poor tissue healing, typically occurs in the most poorly vascularized portion of the tendon and is common in runners. First-line treatments for Achilles tendinopathy include eccentric strength training and anti-inflammatory drugs.34,35 Corticosteroid injections are not recommended, given concern for degraded tendon tissue over time and worse function.34
A 2020 systematic review of 11 randomized and nonrandomized clinical trials (N = 406) found PRP improved Victorian Institute of Sports Assessment—Achilles (VISA-A) scores at 24 weeks compared to other nonsurgical treatment options (41.2 vs 70.12; P < .018).34 However, a higher-quality 2021 systematic review and meta-analysis of 4 RCTs (N = 170) comparing PRP injections with placebo showed no significant difference in VISA-A scores at 3 months (0.23; 95% CI, –0.45 to 0.91), 6 months (0.83; 95% CI, –0.26 to 1.92), and 12 months (0.83; 95% CI, –0.77 to 2.44).36 Therefore, further studies are warranted to evaluate the benefit of PRP injections for Achilles tendinopathy.
Conclusions
While high-quality studies support the use of PRP for knee OA and lateral epicondylitis, they have a moderate-to-high risk for bias. Several RCTs show that PRP provides superior short-term pain relief and range of motion compared to corticosteroids for rotator cuff tendinopathy. Multiple injections of PRP for patellar tendinopathy may accelerate return to sport and improve symptoms over the long term. However, current evidence does not support PRP therapy for Achilles tendinopathy. Given variability in PRP preparation, an accurate interpretation of the literature regarding its use in MSK conditions is recommended (TABLE4,6,7,14-18,20-23,25-28,30-34,36).

Continue to: Concerning the effectiveness of PRP...
Concerning the effectiveness of PRP, it is important to consider early publication bias. Although recent studies have shown its benefits,6,14,15,37 additional studies comparing PRP to placebo will help demonstrate its efficacy. Interestingly, a literature search by Bar-Or et al38 found intra-articular saline may have a therapeutic effect on knee OA and confound findings when used as a placebo.
Recognizing the presence or lack of clinically significant improvement in the literature is important. For example, while some recent studies have shown PRP exceeds the minimal clinically significant difference for knee OA and lateral epicondylitis, others have not.32,37 A 2021 systematic review of 11 clinical practice guidelines for the use of PRP in knee OA found that 9 were “uncertain or unable to make a recommendation” and 2 recommended against it.39
In its 2021 position statement for the responsible use of regenerative medicine, the American Medical Society for Sports Medicine includes guidance on integrating orthobiologics into clinical practice. The guideline emphasizes informed consent and provides an evidence-based rationale for using PRP in certain patient populations (lateral epicondylitis and younger patients with mild-to-moderate knee OA), recommending its use only after exhausting other conservative options.40 Patients should be referred to physicians with experience using PRP and image-guided procedures.
CORRESPONDENCE
Gregory D. Bentz Jr, MD, 3640 High Street Suite 3B, Portsmouth, VA 23707; [email protected]
1. Cecerska-Heryć E, Goszka M, Serwin N, et al. Applications of the regenerative capacity of platelets in modern medicine. Cytokine Growth Factor Rev. 2022;64:84-94. doi: 10.1016/j.cytogfr.2021.11.003
2. Le ADK, Enweze L, DeBaun MR, et al. Current clinical recommendations for use of platelet-rich plasma. Curr Rev Musculoskelet Med. 2018;11:624-634. doi: 10.1007/s12178-018-9527-7
3. Everts P, Onishi K, Jayaram P, et al. Platelet-rich plasma: new performance understandings and therapeutic considerations in 2020. Int J Mol Sci. 2020;21:7794. doi: 10.3390/ijms21207794
4. Di Martino A, Boffa A, Andriolo L, et al. Leukocyte-rich versus leukocyte-poor platelet-rich plasma for the treatment of knee osteoarthritis: a double-blind randomized trial. Am J Sports Med. 2022;50:609-617. doi: 10.1177/03635465211064303
5. Mariani E, Pulsatelli L. Platelet concentrates in musculoskeletal medicine. Int J Mol Sci. 2020;21:1328. doi: 10.3390/ijms21041328
6. Belk JW, Kraeutler MJ, Houck DA, et al. Platelet-rich plasma versus hyaluronic acid for knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Am J Sports Med. 2021;49:249-260. doi: 10.1177/0363546520909397
7. Filardo G, Kon E, Della Villa S, et al. Use of platelet-rich plasma for the treatment of refractory jumper’s knee. Int Orthop. 2010;34:909-915. doi: 10.1007/s00264-009-0845-7
8. Kon E, Filardo G, Delcogliano M, et al. Platelet-rich plasma: new clinical application: a pilot study for treatment of jumper’s knee. Injury. 2009;40:598-603. doi: 10.1016/j.injury.2008.11.026
9. Kanchanatawan W, Arirachakaran A, Chaijenkij K, et al. Short-term outcomes of platelet-rich plasma injection for treatment of osteoarthritis of the knee. Knee Surg Sports Traumatol Arthrosc. 2016;24:1665-1677. doi: 10.1007/s00167-015-3784-4
10. Cook J, Young M. Biologic therapies for tendon and muscle injury. UpToDate. Updated August 11, 2022. Accessed May 23, 2023. www.uptodate.com/contents/biologic-therapies-for-tendon-and-muscle-injury
11. Bendich I, Rubenstein WJ, Cole BJ, et al. What is the appropriate price for platelet-rich plasma injections for knee osteoarthritis? A cost-effectiveness analysis based on evidence from Level I randomized controlled trials. Arthroscopy. 2020;36:1983-1991.e1. doi: 10.1016/j.arthro.2020.02.004
12. Jones IA, Togashi RC, Thomas Vangsness C Jr. The economics and regulation of PRP in the evolving field of orthopedic biologics. Curr Rev Musculoskelet Med. 2018;11:558-565. doi: 10.1007/s12178-018-9514-z
13. Costa LAV, Lenza M, Irrgang JJ, et al. How does platelet-rich plasma compare clinically to other therapies in the treatment of knee osteoarthritis? A systematic review and meta-analysis. Am J Sports Med. 2023;51:1074-1086 doi: 10.1177/03635465211062243
14. Meheux CJ, McCulloch PC, Lintner DM, et al. Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy. 2016;32:495-505. doi: 10.1016/j.arthro.2015.08.005
15. Shen L, Yuan T, Chen S, et al. The temporal effect of platelet-rich plasma on pain and physical function in the treatment of knee osteoarthritis: systematic review and meta-analysis of randomized controlled trials. J Orthop Surg Res. 2017;12:16. doi: 10.1186/s13018-017-0521-3
16. Paget LDA, Reurink G, de Vos RJ, et al; PRIMA Study Group. Effect of platelet-rich plasma injections vs. placebo on ankle symptoms and function in patients with ankle osteoarthritis: a randomized clinical trial. JAMA. 2021;326:1595-1605. doi: 10.1001/jama.2021.16602
17. Evans A, Ibrahim M, Pope R, et al. Treating hand and foot osteoarthritis using a patient’s own blood: a systematic review and meta-analysis of platelet-rich plasma. J Orthop. 2020;18:226-236. doi: 10.1016/j.jor.2020.01.037
18. Ye Y, Zhou X, Mao S, et al. Platelet rich plasma versus hyaluronic acid in patients with hip osteoarthritis: a meta-analysis of randomized controlled trials. Int J Surg. 2018;53:279-287. doi: 10.1016/j.ijsu.2018.03.078.
19. Berney M, McCarroll P, Glynn L, et al. Platelet-rich plasma injections for hip osteoarthritis: a review of the evidence. Ir J Med Sci. 2021;190:1021-1025. doi: 10.1007/s11845-020-02388-z
20. Belk JW, Houck DA, Littlefield CP, et al. Platelet-rich plasma versus hyaluronic acid for hip osteoarthritis yields similarly beneficial short-term clinical outcomes: a systematic review and meta-analysis of Level I and II randomized controlled trials. Arthroscopy. 2022;38:2035-2046. doi: 10.1016/j.arthro.2021.11.005
21. Dadgostar H, Fahimipour F, Pahlevan Sabagh A, et al. Corticosteroids or platelet-rich plasma injections for rotator cuff tendinopathy: a randomized clinical trial study. J Orthop Surg Res. 2021;16:333. doi: 10.1186/s13018-021-02470-x
22. Kwong CA, Woodmass JM, Gusnowski EM, et al. Platelet-rich plasma in patients with partial-thickness rotator cuff tears or tendinopathy leads to significantly improved short-term pain relief and function compared with corticosteroid injection: a double-blind randomized controlled trial. Arthroscopy. 2021;37:510-517. doi: 10.1016/j.arthro.2020.10.037
23. A Hamid MS, Sazlina SG. Platelet-rich plasma for rotator cuff tendinopathy: a systematic review and meta-analysis. PLoS One. 2021;16:e0251111. doi: 10.1371/journal.pone.0251111
24. Lian OB, Engebretsen L, Bahr R. Prevalence of jumper’s knee among elite athletes from different sports: a cross-sectional study. Am J Sports Med. 2005;33:561-567. doi: 10.1177/0363546504270454
25. Dragoo JL, Wasterlain AS, Braun HJ, et al. Platelet-rich plasma as a treatment for patellar tendinopathy: a double-blind, randomized controlled trial. Am J Sports Med. 2014;42:610-618. doi: 10.1177/0363546513518416.
26. Rodas G, Soler-Rich R, Rius-Tarruella J, et al. Effect of autologous expanded bone marrow mesenchymal stem cells or leukocyte-poor platelet-rich plasma in chronic patellar tendinopathy (with gap >3 mm): preliminary outcomes after 6 months of a double-blind, randomized, prospective study. Am J Sports Med. 2021;49:1492-1504. doi: 10.1177/0363546521998725
27. Andriolo L, Altamura SA, Reale D, et al. Nonsurgical treatments of patellar tendinopathy: multiple injections of platelet-rich plasma are a suitable option: a systematic review and meta-analysis. Am J Sports Med. 2019;47:1001-1018. doi: 10.1177/0363546518759674
28. Scott A, LaPrade RF, Harmon KG, et al. Platelet-rich plasma for patellar tendinopathy: a randomized controlled trial of leukocyte-rich PRP or leukocyte-poor PRP versus saline. Am J Sports Med. 2019;47:1654-1661. doi: 10.1177/0363546519837954
29. Kemp JA, Olson MA, Tao MA, et al. Platelet-rich plasma versus corticosteroid injection for the treatment of lateral epicondylitis: a systematic review of systematic reviews. Int J Sports Phys Ther. 2021;16:597-605. doi: 10.26603/001c.24148
30. Miller LE, Parrish WR, Roides B, et al. Efficacy of platelet-rich plasma injections for symptomatic tendinopathy: systematic review and meta-analysis of randomised injection-controlled trials. BMJ Open Sport Exerc Med. 2017;3:e000237. doi: 10.1136/bmjsem-2017- 000237
31. Ben-Nafa W, Munro W. The effect of corticosteroid versus platelet-rich plasma injection therapies for the management of lateral epicondylitis: a systematic review. SICOT J. 2018;4:11.
32. Niemiec P, Szyluk K, Jarosz A, et al. Effectiveness of platelet-rich plasma for lateral epicondylitis: a systematic review and meta-analysis based on achievement of minimal clinically important difference. Orthop J Sports Med. 2022;10:23259671221086920. doi: 10.1177/23259671221086920
33. Li S, Yang G, Zhang H, et al. A systematic review on the efficacy of different types of platelet-rich plasma in the management of lateral epicondylitis. J Shoulder Elbow Surg. 2022;311533-1544. doi: 10.1016/j.jse.2022.02.017.
34. Madhi MI, Yausep OE, Khamdan K, et al. The use of PRP in treatment of Achilles tendinopathy: a systematic review of literature. Study design: systematic review of literature. Ann Med Surg (Lond). 2020;55:320-326. doi: 10.1016/j.amsu.2020.04.042
35. Loppini M, Maffulli N. Conservative management of tendinopathy: an evidence-based approach. Muscles Ligaments Tendons J. 2012;1:134-137.
36. Nauwelaers AK, Van Oost L, Peers K. Evidence for the use of PRP in chronic midsubstance Achilles tendinopathy: a systematic review with meta-analysis. Foot Ankle Surg. 2021;27:486-495. doi: 10.1016/j.fas.2020.07.009
37. Dai WL, Zhou AG, Zhang H, et al. Efficacy of platelet-rich plasma in the treatment of knee osteoarthritis: a meta-analysis of randomized controlled trials. Arthroscopy. 2017;33:659-670.e1. doi: 10.1016/j.arthro.2016.09.024
38. Bar-Or D, Rael LT, Brody EN. Use of saline as a placebo in intra-articular injections in osteoarthritis: potential contributions to nociceptive pain relief. Open Rheumatol J. 2017;11:16-22. doi: 10.2174/1874312901711010016
39. Phillips M, Bhandari M, Grant J, et al. A systematic review of current clinical practice guidelines on intra-articular hyaluronic acid, corticosteroid, and platelet-rich plasma injection for knee osteoarthritis: an international perspective. Orthop J Sports Med. 2021;9:23259671211030272. doi: 10.1177/23259671211030272
40. Finnoff JT, Awan TM, Borg-Stein J, et al. American Medical Society for Sports Medicine position statement: principles for the responsible use of regenerative medicine in sports medicine. Clin J Sport Med. 2021;31:530-541. doi: 10.1097/JSM.0000000000000973
1. Cecerska-Heryć E, Goszka M, Serwin N, et al. Applications of the regenerative capacity of platelets in modern medicine. Cytokine Growth Factor Rev. 2022;64:84-94. doi: 10.1016/j.cytogfr.2021.11.003
2. Le ADK, Enweze L, DeBaun MR, et al. Current clinical recommendations for use of platelet-rich plasma. Curr Rev Musculoskelet Med. 2018;11:624-634. doi: 10.1007/s12178-018-9527-7
3. Everts P, Onishi K, Jayaram P, et al. Platelet-rich plasma: new performance understandings and therapeutic considerations in 2020. Int J Mol Sci. 2020;21:7794. doi: 10.3390/ijms21207794
4. Di Martino A, Boffa A, Andriolo L, et al. Leukocyte-rich versus leukocyte-poor platelet-rich plasma for the treatment of knee osteoarthritis: a double-blind randomized trial. Am J Sports Med. 2022;50:609-617. doi: 10.1177/03635465211064303
5. Mariani E, Pulsatelli L. Platelet concentrates in musculoskeletal medicine. Int J Mol Sci. 2020;21:1328. doi: 10.3390/ijms21041328
6. Belk JW, Kraeutler MJ, Houck DA, et al. Platelet-rich plasma versus hyaluronic acid for knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Am J Sports Med. 2021;49:249-260. doi: 10.1177/0363546520909397
7. Filardo G, Kon E, Della Villa S, et al. Use of platelet-rich plasma for the treatment of refractory jumper’s knee. Int Orthop. 2010;34:909-915. doi: 10.1007/s00264-009-0845-7
8. Kon E, Filardo G, Delcogliano M, et al. Platelet-rich plasma: new clinical application: a pilot study for treatment of jumper’s knee. Injury. 2009;40:598-603. doi: 10.1016/j.injury.2008.11.026
9. Kanchanatawan W, Arirachakaran A, Chaijenkij K, et al. Short-term outcomes of platelet-rich plasma injection for treatment of osteoarthritis of the knee. Knee Surg Sports Traumatol Arthrosc. 2016;24:1665-1677. doi: 10.1007/s00167-015-3784-4
10. Cook J, Young M. Biologic therapies for tendon and muscle injury. UpToDate. Updated August 11, 2022. Accessed May 23, 2023. www.uptodate.com/contents/biologic-therapies-for-tendon-and-muscle-injury
11. Bendich I, Rubenstein WJ, Cole BJ, et al. What is the appropriate price for platelet-rich plasma injections for knee osteoarthritis? A cost-effectiveness analysis based on evidence from Level I randomized controlled trials. Arthroscopy. 2020;36:1983-1991.e1. doi: 10.1016/j.arthro.2020.02.004
12. Jones IA, Togashi RC, Thomas Vangsness C Jr. The economics and regulation of PRP in the evolving field of orthopedic biologics. Curr Rev Musculoskelet Med. 2018;11:558-565. doi: 10.1007/s12178-018-9514-z
13. Costa LAV, Lenza M, Irrgang JJ, et al. How does platelet-rich plasma compare clinically to other therapies in the treatment of knee osteoarthritis? A systematic review and meta-analysis. Am J Sports Med. 2023;51:1074-1086 doi: 10.1177/03635465211062243
14. Meheux CJ, McCulloch PC, Lintner DM, et al. Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy. 2016;32:495-505. doi: 10.1016/j.arthro.2015.08.005
15. Shen L, Yuan T, Chen S, et al. The temporal effect of platelet-rich plasma on pain and physical function in the treatment of knee osteoarthritis: systematic review and meta-analysis of randomized controlled trials. J Orthop Surg Res. 2017;12:16. doi: 10.1186/s13018-017-0521-3
16. Paget LDA, Reurink G, de Vos RJ, et al; PRIMA Study Group. Effect of platelet-rich plasma injections vs. placebo on ankle symptoms and function in patients with ankle osteoarthritis: a randomized clinical trial. JAMA. 2021;326:1595-1605. doi: 10.1001/jama.2021.16602
17. Evans A, Ibrahim M, Pope R, et al. Treating hand and foot osteoarthritis using a patient’s own blood: a systematic review and meta-analysis of platelet-rich plasma. J Orthop. 2020;18:226-236. doi: 10.1016/j.jor.2020.01.037
18. Ye Y, Zhou X, Mao S, et al. Platelet rich plasma versus hyaluronic acid in patients with hip osteoarthritis: a meta-analysis of randomized controlled trials. Int J Surg. 2018;53:279-287. doi: 10.1016/j.ijsu.2018.03.078.
19. Berney M, McCarroll P, Glynn L, et al. Platelet-rich plasma injections for hip osteoarthritis: a review of the evidence. Ir J Med Sci. 2021;190:1021-1025. doi: 10.1007/s11845-020-02388-z
20. Belk JW, Houck DA, Littlefield CP, et al. Platelet-rich plasma versus hyaluronic acid for hip osteoarthritis yields similarly beneficial short-term clinical outcomes: a systematic review and meta-analysis of Level I and II randomized controlled trials. Arthroscopy. 2022;38:2035-2046. doi: 10.1016/j.arthro.2021.11.005
21. Dadgostar H, Fahimipour F, Pahlevan Sabagh A, et al. Corticosteroids or platelet-rich plasma injections for rotator cuff tendinopathy: a randomized clinical trial study. J Orthop Surg Res. 2021;16:333. doi: 10.1186/s13018-021-02470-x
22. Kwong CA, Woodmass JM, Gusnowski EM, et al. Platelet-rich plasma in patients with partial-thickness rotator cuff tears or tendinopathy leads to significantly improved short-term pain relief and function compared with corticosteroid injection: a double-blind randomized controlled trial. Arthroscopy. 2021;37:510-517. doi: 10.1016/j.arthro.2020.10.037
23. A Hamid MS, Sazlina SG. Platelet-rich plasma for rotator cuff tendinopathy: a systematic review and meta-analysis. PLoS One. 2021;16:e0251111. doi: 10.1371/journal.pone.0251111
24. Lian OB, Engebretsen L, Bahr R. Prevalence of jumper’s knee among elite athletes from different sports: a cross-sectional study. Am J Sports Med. 2005;33:561-567. doi: 10.1177/0363546504270454
25. Dragoo JL, Wasterlain AS, Braun HJ, et al. Platelet-rich plasma as a treatment for patellar tendinopathy: a double-blind, randomized controlled trial. Am J Sports Med. 2014;42:610-618. doi: 10.1177/0363546513518416.
26. Rodas G, Soler-Rich R, Rius-Tarruella J, et al. Effect of autologous expanded bone marrow mesenchymal stem cells or leukocyte-poor platelet-rich plasma in chronic patellar tendinopathy (with gap >3 mm): preliminary outcomes after 6 months of a double-blind, randomized, prospective study. Am J Sports Med. 2021;49:1492-1504. doi: 10.1177/0363546521998725
27. Andriolo L, Altamura SA, Reale D, et al. Nonsurgical treatments of patellar tendinopathy: multiple injections of platelet-rich plasma are a suitable option: a systematic review and meta-analysis. Am J Sports Med. 2019;47:1001-1018. doi: 10.1177/0363546518759674
28. Scott A, LaPrade RF, Harmon KG, et al. Platelet-rich plasma for patellar tendinopathy: a randomized controlled trial of leukocyte-rich PRP or leukocyte-poor PRP versus saline. Am J Sports Med. 2019;47:1654-1661. doi: 10.1177/0363546519837954
29. Kemp JA, Olson MA, Tao MA, et al. Platelet-rich plasma versus corticosteroid injection for the treatment of lateral epicondylitis: a systematic review of systematic reviews. Int J Sports Phys Ther. 2021;16:597-605. doi: 10.26603/001c.24148
30. Miller LE, Parrish WR, Roides B, et al. Efficacy of platelet-rich plasma injections for symptomatic tendinopathy: systematic review and meta-analysis of randomised injection-controlled trials. BMJ Open Sport Exerc Med. 2017;3:e000237. doi: 10.1136/bmjsem-2017- 000237
31. Ben-Nafa W, Munro W. The effect of corticosteroid versus platelet-rich plasma injection therapies for the management of lateral epicondylitis: a systematic review. SICOT J. 2018;4:11.
32. Niemiec P, Szyluk K, Jarosz A, et al. Effectiveness of platelet-rich plasma for lateral epicondylitis: a systematic review and meta-analysis based on achievement of minimal clinically important difference. Orthop J Sports Med. 2022;10:23259671221086920. doi: 10.1177/23259671221086920
33. Li S, Yang G, Zhang H, et al. A systematic review on the efficacy of different types of platelet-rich plasma in the management of lateral epicondylitis. J Shoulder Elbow Surg. 2022;311533-1544. doi: 10.1016/j.jse.2022.02.017.
34. Madhi MI, Yausep OE, Khamdan K, et al. The use of PRP in treatment of Achilles tendinopathy: a systematic review of literature. Study design: systematic review of literature. Ann Med Surg (Lond). 2020;55:320-326. doi: 10.1016/j.amsu.2020.04.042
35. Loppini M, Maffulli N. Conservative management of tendinopathy: an evidence-based approach. Muscles Ligaments Tendons J. 2012;1:134-137.
36. Nauwelaers AK, Van Oost L, Peers K. Evidence for the use of PRP in chronic midsubstance Achilles tendinopathy: a systematic review with meta-analysis. Foot Ankle Surg. 2021;27:486-495. doi: 10.1016/j.fas.2020.07.009
37. Dai WL, Zhou AG, Zhang H, et al. Efficacy of platelet-rich plasma in the treatment of knee osteoarthritis: a meta-analysis of randomized controlled trials. Arthroscopy. 2017;33:659-670.e1. doi: 10.1016/j.arthro.2016.09.024
38. Bar-Or D, Rael LT, Brody EN. Use of saline as a placebo in intra-articular injections in osteoarthritis: potential contributions to nociceptive pain relief. Open Rheumatol J. 2017;11:16-22. doi: 10.2174/1874312901711010016
39. Phillips M, Bhandari M, Grant J, et al. A systematic review of current clinical practice guidelines on intra-articular hyaluronic acid, corticosteroid, and platelet-rich plasma injection for knee osteoarthritis: an international perspective. Orthop J Sports Med. 2021;9:23259671211030272. doi: 10.1177/23259671211030272
40. Finnoff JT, Awan TM, Borg-Stein J, et al. American Medical Society for Sports Medicine position statement: principles for the responsible use of regenerative medicine in sports medicine. Clin J Sport Med. 2021;31:530-541. doi: 10.1097/JSM.0000000000000973
PRACTICE RECOMMENDATIONS
› Consider plateletrich plasma (PRP) for conservative management of knee osteoarthritis and lateral epicondylitis. B
› Consider giving multiple injections of PRP for longterm pain relief and expedited return to sport in patellar tendinopathy. B
› Do not use PRP for Achilles tendinopathy due to a lack of clinical evidence. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series