User login
Pandemic colonoscopy restrictions may lead to worse CRC outcomes
For veterans, changes in colonoscopy screening caused by the COVID-19 pandemic may have increased risks of delayed colorectal cancer (CRC) diagnosis and could lead to worse CRC outcomes, based on data from more than 33,000 patients in the Veterans Health Administration.
After COVID-19 screening policies were implemented, a significantly lower rate of veterans with red-flag signs or symptoms for CRC underwent colonoscopy, lead author Joshua Demb, PhD, a cancer epidemiologist at the University of California, San Diego, reported at the annual Digestive Disease Week® (DDW).
“As a result of the COVID-19 pandemic, the Veterans Health Administration enacted risk mitigation and management strategies in March 2020, including postponement of nearly all colonoscopies,” the investigators reported. “Notably, this included veterans with red flag signs or symptoms for CRC, among whom delays in workup could increase risk for later-stage and fatal CRC, if present.”
To measure the effects of this policy change, Dr. Demb and colleagues performed a cohort study involving 33,804 veterans with red-flag signs or symptoms for CRC, including hematochezia, iron deficiency anemia, or abnormal guaiac fecal occult blood test or fecal immunochemical test (FIT). Veterans were divided into two cohorts based on date of first red flag diagnosis: either before the COVID-19 policy was implemented (April to October 2019; n = 19,472) or after (April to October 2020; n = 14,332), with an intervening 6-month washout period.
Primary outcomes were proportion completing colonoscopy and time to colonoscopy completion. Multivariable logistic regression incorporated a number of demographic and medical covariates, including race/ethnicity, sex, age, number of red-flag signs/symptoms, first red-flag sign/symptom, and others.
Before the COVID-19 policy change, 44% of individuals with red-flag signs or symptoms received a colonoscopy, compared with 32% after the policy was introduced (P < .01). Adjusted models showed that veterans in the COVID policy group were 42% less likely to receive a diagnostic colonoscopy than those in the prepolicy group (odds ratio, 0.58; 95% confidence interval, 0.55-0.61). While these findings showed greater likelihood of receiving a screening before the pandemic, postpolicy colonoscopies were conducted sooner, with a median time to procedure of 41 days, compared with 65 days before the pandemic (P < .01). Similar differences in screening rates between pre- and postpandemic groups were observed across all types of red flag signs and symptoms.
“Lower colonoscopy uptake was observed among individuals with red-flag signs/symptoms for CRC post- versus preimplementation of COVID-19 policies, suggesting increased future risk for delayed CRC diagnosis and adverse CRC outcomes,” the investigators concluded.
Prioritization may be needed to overcome backlog of colonoscopies
Jill Tinmouth, MD, PhD, lead scientist for ColonCancerCheck, Ontario’s organized colorectal cancer screening program, and a gastroenterologist and scientist at Sunnybrook Health Sciences Centre, Toronto, shared similar concerns about delayed diagnoses.
“We might expect these cancers to present ... at a more advanced stage, and that, as a result, the outcomes from these cancers could be worse,” Dr. Tinmouth said in an interview.
She also noted the change in colonoscopy timing.
“A particularly interesting finding was that, when a colonoscopy occurred, the time to colonoscopy was shorter during the COVID era than in the pre-COVID era,” Dr. Tinmouth said. “The authors suggested that this might be as a result of Veterans Health Administration policies implemented as a result of the pandemic that led to prioritization of more urgent procedures.”
According to Dr. Tinmouth, similar prioritization may be needed to catch up with the backlog of colonoscopies created by pandemic-related policy changes. In a recent study comparing two backlog management techniques, Dr. Tinmouth and colleagues concluded that redirecting low-yield colonoscopies to FIT without increasing hospital colonoscopy capacity could reduce time to recovery by more than half.
Even so, screening programs may be facing a long road to recovery.
“Recovery of the colonoscopy backlog is going to be a challenge that will take a while – maybe even years – to resolve,” Dr. Tinmouth said. “Jurisdictions/institutions that have a strong centralized intake or triage will likely be most successful in resolving the backlog quickly as they will be able to prioritize the most urgent cases, such as persons with an abnormal FIT or with symptoms, and to redirect persons scheduled for a ‘low-yield’ colonoscopy to have a FIT instead.” Ontario defines low-yield colonoscopies as primary screening for average-risk individuals and follow-up colonoscopies for patients with low-risk adenomas at baseline.
When asked about strategies to address future pandemics, Dr. Tinmouth said, “I think that two key learnings for me from this [pandemic] are: one, not to let our guard down, and to remain vigilant and prepared – in terms of monitoring, supply chain, equipment, etc.] ... and two to create a nimble and agile health system so that we are able to assess the challenges that the next pandemic brings and address them as quickly as possible.”The investigators and Dr. Tinmouth reported no conflicts of interest.
For veterans, changes in colonoscopy screening caused by the COVID-19 pandemic may have increased risks of delayed colorectal cancer (CRC) diagnosis and could lead to worse CRC outcomes, based on data from more than 33,000 patients in the Veterans Health Administration.
After COVID-19 screening policies were implemented, a significantly lower rate of veterans with red-flag signs or symptoms for CRC underwent colonoscopy, lead author Joshua Demb, PhD, a cancer epidemiologist at the University of California, San Diego, reported at the annual Digestive Disease Week® (DDW).
“As a result of the COVID-19 pandemic, the Veterans Health Administration enacted risk mitigation and management strategies in March 2020, including postponement of nearly all colonoscopies,” the investigators reported. “Notably, this included veterans with red flag signs or symptoms for CRC, among whom delays in workup could increase risk for later-stage and fatal CRC, if present.”
To measure the effects of this policy change, Dr. Demb and colleagues performed a cohort study involving 33,804 veterans with red-flag signs or symptoms for CRC, including hematochezia, iron deficiency anemia, or abnormal guaiac fecal occult blood test or fecal immunochemical test (FIT). Veterans were divided into two cohorts based on date of first red flag diagnosis: either before the COVID-19 policy was implemented (April to October 2019; n = 19,472) or after (April to October 2020; n = 14,332), with an intervening 6-month washout period.
Primary outcomes were proportion completing colonoscopy and time to colonoscopy completion. Multivariable logistic regression incorporated a number of demographic and medical covariates, including race/ethnicity, sex, age, number of red-flag signs/symptoms, first red-flag sign/symptom, and others.
Before the COVID-19 policy change, 44% of individuals with red-flag signs or symptoms received a colonoscopy, compared with 32% after the policy was introduced (P < .01). Adjusted models showed that veterans in the COVID policy group were 42% less likely to receive a diagnostic colonoscopy than those in the prepolicy group (odds ratio, 0.58; 95% confidence interval, 0.55-0.61). While these findings showed greater likelihood of receiving a screening before the pandemic, postpolicy colonoscopies were conducted sooner, with a median time to procedure of 41 days, compared with 65 days before the pandemic (P < .01). Similar differences in screening rates between pre- and postpandemic groups were observed across all types of red flag signs and symptoms.
“Lower colonoscopy uptake was observed among individuals with red-flag signs/symptoms for CRC post- versus preimplementation of COVID-19 policies, suggesting increased future risk for delayed CRC diagnosis and adverse CRC outcomes,” the investigators concluded.
Prioritization may be needed to overcome backlog of colonoscopies
Jill Tinmouth, MD, PhD, lead scientist for ColonCancerCheck, Ontario’s organized colorectal cancer screening program, and a gastroenterologist and scientist at Sunnybrook Health Sciences Centre, Toronto, shared similar concerns about delayed diagnoses.
“We might expect these cancers to present ... at a more advanced stage, and that, as a result, the outcomes from these cancers could be worse,” Dr. Tinmouth said in an interview.
She also noted the change in colonoscopy timing.
“A particularly interesting finding was that, when a colonoscopy occurred, the time to colonoscopy was shorter during the COVID era than in the pre-COVID era,” Dr. Tinmouth said. “The authors suggested that this might be as a result of Veterans Health Administration policies implemented as a result of the pandemic that led to prioritization of more urgent procedures.”
According to Dr. Tinmouth, similar prioritization may be needed to catch up with the backlog of colonoscopies created by pandemic-related policy changes. In a recent study comparing two backlog management techniques, Dr. Tinmouth and colleagues concluded that redirecting low-yield colonoscopies to FIT without increasing hospital colonoscopy capacity could reduce time to recovery by more than half.
Even so, screening programs may be facing a long road to recovery.
“Recovery of the colonoscopy backlog is going to be a challenge that will take a while – maybe even years – to resolve,” Dr. Tinmouth said. “Jurisdictions/institutions that have a strong centralized intake or triage will likely be most successful in resolving the backlog quickly as they will be able to prioritize the most urgent cases, such as persons with an abnormal FIT or with symptoms, and to redirect persons scheduled for a ‘low-yield’ colonoscopy to have a FIT instead.” Ontario defines low-yield colonoscopies as primary screening for average-risk individuals and follow-up colonoscopies for patients with low-risk adenomas at baseline.
When asked about strategies to address future pandemics, Dr. Tinmouth said, “I think that two key learnings for me from this [pandemic] are: one, not to let our guard down, and to remain vigilant and prepared – in terms of monitoring, supply chain, equipment, etc.] ... and two to create a nimble and agile health system so that we are able to assess the challenges that the next pandemic brings and address them as quickly as possible.”The investigators and Dr. Tinmouth reported no conflicts of interest.
For veterans, changes in colonoscopy screening caused by the COVID-19 pandemic may have increased risks of delayed colorectal cancer (CRC) diagnosis and could lead to worse CRC outcomes, based on data from more than 33,000 patients in the Veterans Health Administration.
After COVID-19 screening policies were implemented, a significantly lower rate of veterans with red-flag signs or symptoms for CRC underwent colonoscopy, lead author Joshua Demb, PhD, a cancer epidemiologist at the University of California, San Diego, reported at the annual Digestive Disease Week® (DDW).
“As a result of the COVID-19 pandemic, the Veterans Health Administration enacted risk mitigation and management strategies in March 2020, including postponement of nearly all colonoscopies,” the investigators reported. “Notably, this included veterans with red flag signs or symptoms for CRC, among whom delays in workup could increase risk for later-stage and fatal CRC, if present.”
To measure the effects of this policy change, Dr. Demb and colleagues performed a cohort study involving 33,804 veterans with red-flag signs or symptoms for CRC, including hematochezia, iron deficiency anemia, or abnormal guaiac fecal occult blood test or fecal immunochemical test (FIT). Veterans were divided into two cohorts based on date of first red flag diagnosis: either before the COVID-19 policy was implemented (April to October 2019; n = 19,472) or after (April to October 2020; n = 14,332), with an intervening 6-month washout period.
Primary outcomes were proportion completing colonoscopy and time to colonoscopy completion. Multivariable logistic regression incorporated a number of demographic and medical covariates, including race/ethnicity, sex, age, number of red-flag signs/symptoms, first red-flag sign/symptom, and others.
Before the COVID-19 policy change, 44% of individuals with red-flag signs or symptoms received a colonoscopy, compared with 32% after the policy was introduced (P < .01). Adjusted models showed that veterans in the COVID policy group were 42% less likely to receive a diagnostic colonoscopy than those in the prepolicy group (odds ratio, 0.58; 95% confidence interval, 0.55-0.61). While these findings showed greater likelihood of receiving a screening before the pandemic, postpolicy colonoscopies were conducted sooner, with a median time to procedure of 41 days, compared with 65 days before the pandemic (P < .01). Similar differences in screening rates between pre- and postpandemic groups were observed across all types of red flag signs and symptoms.
“Lower colonoscopy uptake was observed among individuals with red-flag signs/symptoms for CRC post- versus preimplementation of COVID-19 policies, suggesting increased future risk for delayed CRC diagnosis and adverse CRC outcomes,” the investigators concluded.
Prioritization may be needed to overcome backlog of colonoscopies
Jill Tinmouth, MD, PhD, lead scientist for ColonCancerCheck, Ontario’s organized colorectal cancer screening program, and a gastroenterologist and scientist at Sunnybrook Health Sciences Centre, Toronto, shared similar concerns about delayed diagnoses.
“We might expect these cancers to present ... at a more advanced stage, and that, as a result, the outcomes from these cancers could be worse,” Dr. Tinmouth said in an interview.
She also noted the change in colonoscopy timing.
“A particularly interesting finding was that, when a colonoscopy occurred, the time to colonoscopy was shorter during the COVID era than in the pre-COVID era,” Dr. Tinmouth said. “The authors suggested that this might be as a result of Veterans Health Administration policies implemented as a result of the pandemic that led to prioritization of more urgent procedures.”
According to Dr. Tinmouth, similar prioritization may be needed to catch up with the backlog of colonoscopies created by pandemic-related policy changes. In a recent study comparing two backlog management techniques, Dr. Tinmouth and colleagues concluded that redirecting low-yield colonoscopies to FIT without increasing hospital colonoscopy capacity could reduce time to recovery by more than half.
Even so, screening programs may be facing a long road to recovery.
“Recovery of the colonoscopy backlog is going to be a challenge that will take a while – maybe even years – to resolve,” Dr. Tinmouth said. “Jurisdictions/institutions that have a strong centralized intake or triage will likely be most successful in resolving the backlog quickly as they will be able to prioritize the most urgent cases, such as persons with an abnormal FIT or with symptoms, and to redirect persons scheduled for a ‘low-yield’ colonoscopy to have a FIT instead.” Ontario defines low-yield colonoscopies as primary screening for average-risk individuals and follow-up colonoscopies for patients with low-risk adenomas at baseline.
When asked about strategies to address future pandemics, Dr. Tinmouth said, “I think that two key learnings for me from this [pandemic] are: one, not to let our guard down, and to remain vigilant and prepared – in terms of monitoring, supply chain, equipment, etc.] ... and two to create a nimble and agile health system so that we are able to assess the challenges that the next pandemic brings and address them as quickly as possible.”The investigators and Dr. Tinmouth reported no conflicts of interest.
FROM DDW 2021
Discharge by Noon: Toward a Better Understanding of Benefits and Costs
Targeting “discharge before noon” (DBN) for hospitalized patients has been proposed as a way to improve hospital throughput and patient safety by reducing emergency department (ED) boarding and crowding. In this issue, Kirubarajan et al1 report no association between morning discharge and length of stay (LOS) for either the ED or hospitalization.1 This large (189,781 patients) 7-year study from seven quite different Canadian hospitals adds important data to a literature that remains divided about whether DBN helps or hurts hospital LOS and ED boarding.
Unlike trials reporting interventions to encourage DBN, this observational study was unique in that it took each day as the unit of observation. This method cleverly allowed the authors to examine whether days with more discharges before noon conferred a lower mean ED and inpatient LOS among patients admitted on those days. Their approach appropriately reframes the central issue as one of patient flow.
Kirubarajan et al’s most notable, and perhaps surprising, finding is the lack of association between morning discharge and ED LOS. Computer modeling supports the hypothesis that ED throughput will improve on days with earlier inpatient bed availability.2 Several studies have also noted earlier ED departure times and decreased ED wait times after implementing interventions to promote DBN.3 Why might the authors’ findings contradict previous studies? Their outcomes may in part be due to high ED LOS (>14 hours), exceeding Canadian published targets and reports from the United States.4,5 Problems relating to ED resources, practice, and hospital census may have overwhelmed DBN as factors in boarding. The interpretation of their findings is limited by the authors’ decision to report only ED LOS, rather than including the time between a decision to admit and ED departure (boarding time).
While early studies that focused on interventions to promote DBN noted decreased inpatient LOS after their implementation, later studies found no effect or even an increase in LOS for general internal medicine patients. Concerns have been raised about the confounding effect of concurrent initiatives aimed at improving LOS as well as misaligned incentives to delay discharge to the following morning. As the number of conflicting studies mounts, and with the current report in hand, it is tempting to conclude that for the DBN evidence base as a whole, we are observing random variation around no effect.
With growing doubt about benefits of morning discharge, perhaps we should turn our attention away from the question of how to increase DBN and consider instead why and at what cost. Hospitals are delicate organisms; a singular focus on one metric will undoubtedly impact others. Does the effort to discharge before noon consume valuable morning hours and detract from the care of other patients? Are patients held overnight unnecessarily to comply with DBN? Are there consequences in patient, nursing, or trainee satisfaction? Is bedside teaching affected?
And as concepts of patient-centered care are increasingly valued, we may ask whether DBN is such a concept, or is it rather an increasingly dubious strategy aimed at regularizing hospital operations? The need for a more holistic assessment of “discharge quality” is apparent. Instead of focusing on a particular hour, initiatives should determine the “best, earliest discharge time” for each patient and align multidisciplinary efforts toward this patient-centered goal. Such efforts are already underway in pediatric hospitals, where fixed discharge times are being replaced by discharge milestones embedded into the electronic medical record.6 An instrument to track “discharge readiness” such as this one, paired with ongoing analysis of the barriers to timely discharge, might better facilitate throughput by targeting the entire admission, rather than concentrating pressure on its final hours.
1. Kirubarajan A, Shin S, Fralick M, Kwan Jet al. Morning discharges and patient length-of-stay in inpatient general internal medicine. J Hosp Med. 2021;16(6):334-338. https://doi.org/ 10.12788/jhm.3605
2. Powell ES, Khare RK, Venkatesh AK, Van Roo BD, Adams JG, Reinhardt G. The relationship between inpatient discharge timing and emergency department boarding. J Emerg Med. 2012;42(2):186-196. https://doi.org/10.1016/j.jemermed.2010.06.028
3. Wertheimer B, Jacobs RE, Iturrate E, Bailey M, Hochman K. Discharge before noon: effect on throughput and sustainability. J Hosp Med. 2015;10(10):664-669. https://doi.org/10.1002/jhm.2412
4. Fee C, Burstin H, Maselli JH, Hsia RY. Association of emergency department length of stay with safety-net status. JAMA. 2012;307(5):476-482. https://doi.org/10.1001/jama.2012.41
5. Ontario wait times. Ontario Ministry of Health and Ministry of Long-Term Care. Accessed February 17, 2021. http://www.health.gov.on.ca/en/pro/programs/waittimes/edrs/targets.aspx
6. White CM, Statile AM, White DL, et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428-436. https://doi.org/10.1136/bmjqs-2013-002556
Targeting “discharge before noon” (DBN) for hospitalized patients has been proposed as a way to improve hospital throughput and patient safety by reducing emergency department (ED) boarding and crowding. In this issue, Kirubarajan et al1 report no association between morning discharge and length of stay (LOS) for either the ED or hospitalization.1 This large (189,781 patients) 7-year study from seven quite different Canadian hospitals adds important data to a literature that remains divided about whether DBN helps or hurts hospital LOS and ED boarding.
Unlike trials reporting interventions to encourage DBN, this observational study was unique in that it took each day as the unit of observation. This method cleverly allowed the authors to examine whether days with more discharges before noon conferred a lower mean ED and inpatient LOS among patients admitted on those days. Their approach appropriately reframes the central issue as one of patient flow.
Kirubarajan et al’s most notable, and perhaps surprising, finding is the lack of association between morning discharge and ED LOS. Computer modeling supports the hypothesis that ED throughput will improve on days with earlier inpatient bed availability.2 Several studies have also noted earlier ED departure times and decreased ED wait times after implementing interventions to promote DBN.3 Why might the authors’ findings contradict previous studies? Their outcomes may in part be due to high ED LOS (>14 hours), exceeding Canadian published targets and reports from the United States.4,5 Problems relating to ED resources, practice, and hospital census may have overwhelmed DBN as factors in boarding. The interpretation of their findings is limited by the authors’ decision to report only ED LOS, rather than including the time between a decision to admit and ED departure (boarding time).
While early studies that focused on interventions to promote DBN noted decreased inpatient LOS after their implementation, later studies found no effect or even an increase in LOS for general internal medicine patients. Concerns have been raised about the confounding effect of concurrent initiatives aimed at improving LOS as well as misaligned incentives to delay discharge to the following morning. As the number of conflicting studies mounts, and with the current report in hand, it is tempting to conclude that for the DBN evidence base as a whole, we are observing random variation around no effect.
With growing doubt about benefits of morning discharge, perhaps we should turn our attention away from the question of how to increase DBN and consider instead why and at what cost. Hospitals are delicate organisms; a singular focus on one metric will undoubtedly impact others. Does the effort to discharge before noon consume valuable morning hours and detract from the care of other patients? Are patients held overnight unnecessarily to comply with DBN? Are there consequences in patient, nursing, or trainee satisfaction? Is bedside teaching affected?
And as concepts of patient-centered care are increasingly valued, we may ask whether DBN is such a concept, or is it rather an increasingly dubious strategy aimed at regularizing hospital operations? The need for a more holistic assessment of “discharge quality” is apparent. Instead of focusing on a particular hour, initiatives should determine the “best, earliest discharge time” for each patient and align multidisciplinary efforts toward this patient-centered goal. Such efforts are already underway in pediatric hospitals, where fixed discharge times are being replaced by discharge milestones embedded into the electronic medical record.6 An instrument to track “discharge readiness” such as this one, paired with ongoing analysis of the barriers to timely discharge, might better facilitate throughput by targeting the entire admission, rather than concentrating pressure on its final hours.
Targeting “discharge before noon” (DBN) for hospitalized patients has been proposed as a way to improve hospital throughput and patient safety by reducing emergency department (ED) boarding and crowding. In this issue, Kirubarajan et al1 report no association between morning discharge and length of stay (LOS) for either the ED or hospitalization.1 This large (189,781 patients) 7-year study from seven quite different Canadian hospitals adds important data to a literature that remains divided about whether DBN helps or hurts hospital LOS and ED boarding.
Unlike trials reporting interventions to encourage DBN, this observational study was unique in that it took each day as the unit of observation. This method cleverly allowed the authors to examine whether days with more discharges before noon conferred a lower mean ED and inpatient LOS among patients admitted on those days. Their approach appropriately reframes the central issue as one of patient flow.
Kirubarajan et al’s most notable, and perhaps surprising, finding is the lack of association between morning discharge and ED LOS. Computer modeling supports the hypothesis that ED throughput will improve on days with earlier inpatient bed availability.2 Several studies have also noted earlier ED departure times and decreased ED wait times after implementing interventions to promote DBN.3 Why might the authors’ findings contradict previous studies? Their outcomes may in part be due to high ED LOS (>14 hours), exceeding Canadian published targets and reports from the United States.4,5 Problems relating to ED resources, practice, and hospital census may have overwhelmed DBN as factors in boarding. The interpretation of their findings is limited by the authors’ decision to report only ED LOS, rather than including the time between a decision to admit and ED departure (boarding time).
While early studies that focused on interventions to promote DBN noted decreased inpatient LOS after their implementation, later studies found no effect or even an increase in LOS for general internal medicine patients. Concerns have been raised about the confounding effect of concurrent initiatives aimed at improving LOS as well as misaligned incentives to delay discharge to the following morning. As the number of conflicting studies mounts, and with the current report in hand, it is tempting to conclude that for the DBN evidence base as a whole, we are observing random variation around no effect.
With growing doubt about benefits of morning discharge, perhaps we should turn our attention away from the question of how to increase DBN and consider instead why and at what cost. Hospitals are delicate organisms; a singular focus on one metric will undoubtedly impact others. Does the effort to discharge before noon consume valuable morning hours and detract from the care of other patients? Are patients held overnight unnecessarily to comply with DBN? Are there consequences in patient, nursing, or trainee satisfaction? Is bedside teaching affected?
And as concepts of patient-centered care are increasingly valued, we may ask whether DBN is such a concept, or is it rather an increasingly dubious strategy aimed at regularizing hospital operations? The need for a more holistic assessment of “discharge quality” is apparent. Instead of focusing on a particular hour, initiatives should determine the “best, earliest discharge time” for each patient and align multidisciplinary efforts toward this patient-centered goal. Such efforts are already underway in pediatric hospitals, where fixed discharge times are being replaced by discharge milestones embedded into the electronic medical record.6 An instrument to track “discharge readiness” such as this one, paired with ongoing analysis of the barriers to timely discharge, might better facilitate throughput by targeting the entire admission, rather than concentrating pressure on its final hours.
1. Kirubarajan A, Shin S, Fralick M, Kwan Jet al. Morning discharges and patient length-of-stay in inpatient general internal medicine. J Hosp Med. 2021;16(6):334-338. https://doi.org/ 10.12788/jhm.3605
2. Powell ES, Khare RK, Venkatesh AK, Van Roo BD, Adams JG, Reinhardt G. The relationship between inpatient discharge timing and emergency department boarding. J Emerg Med. 2012;42(2):186-196. https://doi.org/10.1016/j.jemermed.2010.06.028
3. Wertheimer B, Jacobs RE, Iturrate E, Bailey M, Hochman K. Discharge before noon: effect on throughput and sustainability. J Hosp Med. 2015;10(10):664-669. https://doi.org/10.1002/jhm.2412
4. Fee C, Burstin H, Maselli JH, Hsia RY. Association of emergency department length of stay with safety-net status. JAMA. 2012;307(5):476-482. https://doi.org/10.1001/jama.2012.41
5. Ontario wait times. Ontario Ministry of Health and Ministry of Long-Term Care. Accessed February 17, 2021. http://www.health.gov.on.ca/en/pro/programs/waittimes/edrs/targets.aspx
6. White CM, Statile AM, White DL, et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428-436. https://doi.org/10.1136/bmjqs-2013-002556
1. Kirubarajan A, Shin S, Fralick M, Kwan Jet al. Morning discharges and patient length-of-stay in inpatient general internal medicine. J Hosp Med. 2021;16(6):334-338. https://doi.org/ 10.12788/jhm.3605
2. Powell ES, Khare RK, Venkatesh AK, Van Roo BD, Adams JG, Reinhardt G. The relationship between inpatient discharge timing and emergency department boarding. J Emerg Med. 2012;42(2):186-196. https://doi.org/10.1016/j.jemermed.2010.06.028
3. Wertheimer B, Jacobs RE, Iturrate E, Bailey M, Hochman K. Discharge before noon: effect on throughput and sustainability. J Hosp Med. 2015;10(10):664-669. https://doi.org/10.1002/jhm.2412
4. Fee C, Burstin H, Maselli JH, Hsia RY. Association of emergency department length of stay with safety-net status. JAMA. 2012;307(5):476-482. https://doi.org/10.1001/jama.2012.41
5. Ontario wait times. Ontario Ministry of Health and Ministry of Long-Term Care. Accessed February 17, 2021. http://www.health.gov.on.ca/en/pro/programs/waittimes/edrs/targets.aspx
6. White CM, Statile AM, White DL, et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428-436. https://doi.org/10.1136/bmjqs-2013-002556
© 2021 Society of Hospital Medicine
Are You Thinking What I’m Thinking? The Case for Shared Mental Models in Hospital Discharges
Hospital discharge is a complex, multi-stakeholder event, and evidence suggests that the quality of that transition directly relates to mortality, readmissions, and postdischarge quality of life and functional status.1 The Centers for Medicare & Medicaid Services call for team-based and patient-centered discharge planning,2 yet the process for achieving this is poorly defined.
In this issue of the Journal of Hospital Medicine, Manges et al3 use shared mental models (SMM) as a conceptual framework to describe differences in how care team members and patients perceive hospital discharge readiness. While our understanding of factors associated with safe and patient-centered hospital discharges is still growing, the authors focus on one critical component: lack of agreement between patients and interprofessional teams regarding discharge readiness.
Manges et al3 measured whether interprofessional team members agree, or converge, on their assessment of a patient’s discharge readiness (team-SMM convergence) and whether that assessment converges with the patient’s self-assessment (team-patient SMM convergence). They found good team-SMM convergence regarding the patient’s discharge readiness, yet teams overestimated readiness compared with the patient’s self-assessment nearly half (48.4%) of the time. A clinical trial found that clinician assessments of discharge readiness were poorly predictive of readmissions unless they were combined with a patient’s self-assessment.4 Manges et al’s study findings, while of limited generalizability, enhance our understanding of a potential gap in achieving patient-centered care as outlined in the Institute of Medicine’s Crossing the Quality Chasm,5 which urges clinicians to see patients and families as partners in improving care.
The authors also found that higher team-patient convergence was associated with teams that reported high-quality teamwork and those having more baccalaureate degree−educated nurses (BSN). While Manges et al3 did not elucidate the mechanism by which this occurs, their findings align with existing literature showing that patients receiving care from a higher proportion of BSN-prepared nurses experience an 18.7% reduction in odds of readmission.6 Further research investigating the link between team communication, registered nurse education, and discharge outcomes may reveal additional opportunities for interventions to improve discharge quality.
The lack of patient outcomes and the limited diversity of the patient population are substantial limitations of the study. The authors did not assess the relationship between SMMs and important outcomes like readmission or adverse events. Furthermore, most of the patients were White and English-speaking, precluding assessment of factors that disproportionately impact patient populations that already experience disparities in a multitude of health outcomes.
In summary, Manges et al3 highlight challenges and opportunities in optimizing clinician communication and ensuring that the team’s and the patient’s self-assessments align and inform discharge planning. Their findings suggest the theoretical framework of SMM holds promise in identifying and evaluating some of the complex determinants involved in high-quality, patient-centered hospital discharges.
1. Naylor MD, Brooten DA, Campbell RL, Maislin G, McCauley KM, Schwartz JS. Transitional care of older adults hospitalized with heart failure: a randomized, controlled trial. J Am Geriatr Soc. 2004;52(5):675-684. https://doi.org/10.1111/j.1532-5415.2004.52202.x
2. Centers for Medicare & Medicaid Services. Medicare and Medicaid programs; revisions to requirements for discharge planning for hospitals, critical access hospitals, and home health agencies, and hospital and critical access hospital changes to promote innovation, flexibility, and improvement in patient care. Fed Regist. 2019;84(189):51836-51884. https://www.govinfo.gov/content/pkg/FR-2019-09-30/pdf/2019-20732.pdf
3. Manges KA, Wallace AS, Groves PS, Schapira MM, Burke RE. Ready to go home? Assessment of shared mental models of the patient and discharging team regarding readiness for hospital discharge. J Hosp Med. 2020;16(6):326-332. https://doi.org/10.12788/jhm.3464
4. Weiss ME, Yakusheva O, Bobay KL, et al. Effect of implementing discharge readiness assessment in adult medical-surgical units on 30-day return to hospital: the READI randomized clinical trial. JAMA Netw open. 2019;2(1):e187387. https://doi.org/10.1001/jamanetworkopen.2018.7387
5. Institute of Medicine Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. National Academies Press; 2001.
6. Yakusheva O, Lindrooth R, Weiss M. Economic evaluation of the 80% baccalaureate nurse workforce recommendation: a patient-level analysis. Med Care. 2014;52(10):864-869. https://doi.org/10.1097/MLR.0000000000000189
Hospital discharge is a complex, multi-stakeholder event, and evidence suggests that the quality of that transition directly relates to mortality, readmissions, and postdischarge quality of life and functional status.1 The Centers for Medicare & Medicaid Services call for team-based and patient-centered discharge planning,2 yet the process for achieving this is poorly defined.
In this issue of the Journal of Hospital Medicine, Manges et al3 use shared mental models (SMM) as a conceptual framework to describe differences in how care team members and patients perceive hospital discharge readiness. While our understanding of factors associated with safe and patient-centered hospital discharges is still growing, the authors focus on one critical component: lack of agreement between patients and interprofessional teams regarding discharge readiness.
Manges et al3 measured whether interprofessional team members agree, or converge, on their assessment of a patient’s discharge readiness (team-SMM convergence) and whether that assessment converges with the patient’s self-assessment (team-patient SMM convergence). They found good team-SMM convergence regarding the patient’s discharge readiness, yet teams overestimated readiness compared with the patient’s self-assessment nearly half (48.4%) of the time. A clinical trial found that clinician assessments of discharge readiness were poorly predictive of readmissions unless they were combined with a patient’s self-assessment.4 Manges et al’s study findings, while of limited generalizability, enhance our understanding of a potential gap in achieving patient-centered care as outlined in the Institute of Medicine’s Crossing the Quality Chasm,5 which urges clinicians to see patients and families as partners in improving care.
The authors also found that higher team-patient convergence was associated with teams that reported high-quality teamwork and those having more baccalaureate degree−educated nurses (BSN). While Manges et al3 did not elucidate the mechanism by which this occurs, their findings align with existing literature showing that patients receiving care from a higher proportion of BSN-prepared nurses experience an 18.7% reduction in odds of readmission.6 Further research investigating the link between team communication, registered nurse education, and discharge outcomes may reveal additional opportunities for interventions to improve discharge quality.
The lack of patient outcomes and the limited diversity of the patient population are substantial limitations of the study. The authors did not assess the relationship between SMMs and important outcomes like readmission or adverse events. Furthermore, most of the patients were White and English-speaking, precluding assessment of factors that disproportionately impact patient populations that already experience disparities in a multitude of health outcomes.
In summary, Manges et al3 highlight challenges and opportunities in optimizing clinician communication and ensuring that the team’s and the patient’s self-assessments align and inform discharge planning. Their findings suggest the theoretical framework of SMM holds promise in identifying and evaluating some of the complex determinants involved in high-quality, patient-centered hospital discharges.
Hospital discharge is a complex, multi-stakeholder event, and evidence suggests that the quality of that transition directly relates to mortality, readmissions, and postdischarge quality of life and functional status.1 The Centers for Medicare & Medicaid Services call for team-based and patient-centered discharge planning,2 yet the process for achieving this is poorly defined.
In this issue of the Journal of Hospital Medicine, Manges et al3 use shared mental models (SMM) as a conceptual framework to describe differences in how care team members and patients perceive hospital discharge readiness. While our understanding of factors associated with safe and patient-centered hospital discharges is still growing, the authors focus on one critical component: lack of agreement between patients and interprofessional teams regarding discharge readiness.
Manges et al3 measured whether interprofessional team members agree, or converge, on their assessment of a patient’s discharge readiness (team-SMM convergence) and whether that assessment converges with the patient’s self-assessment (team-patient SMM convergence). They found good team-SMM convergence regarding the patient’s discharge readiness, yet teams overestimated readiness compared with the patient’s self-assessment nearly half (48.4%) of the time. A clinical trial found that clinician assessments of discharge readiness were poorly predictive of readmissions unless they were combined with a patient’s self-assessment.4 Manges et al’s study findings, while of limited generalizability, enhance our understanding of a potential gap in achieving patient-centered care as outlined in the Institute of Medicine’s Crossing the Quality Chasm,5 which urges clinicians to see patients and families as partners in improving care.
The authors also found that higher team-patient convergence was associated with teams that reported high-quality teamwork and those having more baccalaureate degree−educated nurses (BSN). While Manges et al3 did not elucidate the mechanism by which this occurs, their findings align with existing literature showing that patients receiving care from a higher proportion of BSN-prepared nurses experience an 18.7% reduction in odds of readmission.6 Further research investigating the link between team communication, registered nurse education, and discharge outcomes may reveal additional opportunities for interventions to improve discharge quality.
The lack of patient outcomes and the limited diversity of the patient population are substantial limitations of the study. The authors did not assess the relationship between SMMs and important outcomes like readmission or adverse events. Furthermore, most of the patients were White and English-speaking, precluding assessment of factors that disproportionately impact patient populations that already experience disparities in a multitude of health outcomes.
In summary, Manges et al3 highlight challenges and opportunities in optimizing clinician communication and ensuring that the team’s and the patient’s self-assessments align and inform discharge planning. Their findings suggest the theoretical framework of SMM holds promise in identifying and evaluating some of the complex determinants involved in high-quality, patient-centered hospital discharges.
1. Naylor MD, Brooten DA, Campbell RL, Maislin G, McCauley KM, Schwartz JS. Transitional care of older adults hospitalized with heart failure: a randomized, controlled trial. J Am Geriatr Soc. 2004;52(5):675-684. https://doi.org/10.1111/j.1532-5415.2004.52202.x
2. Centers for Medicare & Medicaid Services. Medicare and Medicaid programs; revisions to requirements for discharge planning for hospitals, critical access hospitals, and home health agencies, and hospital and critical access hospital changes to promote innovation, flexibility, and improvement in patient care. Fed Regist. 2019;84(189):51836-51884. https://www.govinfo.gov/content/pkg/FR-2019-09-30/pdf/2019-20732.pdf
3. Manges KA, Wallace AS, Groves PS, Schapira MM, Burke RE. Ready to go home? Assessment of shared mental models of the patient and discharging team regarding readiness for hospital discharge. J Hosp Med. 2020;16(6):326-332. https://doi.org/10.12788/jhm.3464
4. Weiss ME, Yakusheva O, Bobay KL, et al. Effect of implementing discharge readiness assessment in adult medical-surgical units on 30-day return to hospital: the READI randomized clinical trial. JAMA Netw open. 2019;2(1):e187387. https://doi.org/10.1001/jamanetworkopen.2018.7387
5. Institute of Medicine Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. National Academies Press; 2001.
6. Yakusheva O, Lindrooth R, Weiss M. Economic evaluation of the 80% baccalaureate nurse workforce recommendation: a patient-level analysis. Med Care. 2014;52(10):864-869. https://doi.org/10.1097/MLR.0000000000000189
1. Naylor MD, Brooten DA, Campbell RL, Maislin G, McCauley KM, Schwartz JS. Transitional care of older adults hospitalized with heart failure: a randomized, controlled trial. J Am Geriatr Soc. 2004;52(5):675-684. https://doi.org/10.1111/j.1532-5415.2004.52202.x
2. Centers for Medicare & Medicaid Services. Medicare and Medicaid programs; revisions to requirements for discharge planning for hospitals, critical access hospitals, and home health agencies, and hospital and critical access hospital changes to promote innovation, flexibility, and improvement in patient care. Fed Regist. 2019;84(189):51836-51884. https://www.govinfo.gov/content/pkg/FR-2019-09-30/pdf/2019-20732.pdf
3. Manges KA, Wallace AS, Groves PS, Schapira MM, Burke RE. Ready to go home? Assessment of shared mental models of the patient and discharging team regarding readiness for hospital discharge. J Hosp Med. 2020;16(6):326-332. https://doi.org/10.12788/jhm.3464
4. Weiss ME, Yakusheva O, Bobay KL, et al. Effect of implementing discharge readiness assessment in adult medical-surgical units on 30-day return to hospital: the READI randomized clinical trial. JAMA Netw open. 2019;2(1):e187387. https://doi.org/10.1001/jamanetworkopen.2018.7387
5. Institute of Medicine Committee on Quality of Health Care in America. Crossing the Quality Chasm: A New Health System for the 21st Century. National Academies Press; 2001.
6. Yakusheva O, Lindrooth R, Weiss M. Economic evaluation of the 80% baccalaureate nurse workforce recommendation: a patient-level analysis. Med Care. 2014;52(10):864-869. https://doi.org/10.1097/MLR.0000000000000189
© 2021 Society of Hospital Medicine
Complete pelvic peritonectomy
Lower SARS-CoV-2 vaccine responses seen in patients with immune-mediated inflammatory diseases
Ten percent of patients with immune-mediated inflammatory diseases (IMIDs) fail to respond properly to COVID-19 vaccinations regardless of medication, researchers report, and small new studies suggest those on methotrexate and rituximab may be especially vulnerable to vaccine failure.
Even so, it’s still crucially vital for patients with IMIDs to get vaccinated and for clinicians to follow recommendations to temporarily withhold certain medications around the time of vaccination, rheumatologist Anne R. Bass, MD, of Weill Cornell Medicine and the Hospital for Special Surgery, New York, said in an interview. “We’re not making any significant adjustments,” added Dr. Bass, a coauthor of the American College of Rheumatology’s COVID-19 vaccination guidelines for patients with rheumatic and musculoskeletal diseases.
The findings appear in a trio of studies in Annals of the Rheumatic Diseases. The most recent study, which appeared May 25, 2021, found that more than one-third of patients with IMIDs who took methotrexate didn’t produce adequate antibody levels after vaccination versus 10% of those in other groups. (P < .001) A May 11 study found that 20 of 30 patients with rheumatic diseases on rituximab failed to respond to vaccination. And a May 6 study reported that immune responses against SARS-CoV-2 are “somewhat delayed and reduced” in patients with IMID, with 99.5% of a control group developing neutralizing antibody activity after vaccination versus 90% of those with IMID (P = .0008).
Development of neutralizing antibodies somewhat delayed and reduced
Team members were surprised by the high number of vaccine nonresponders in the May 6 IMID study, coauthor Georg Schett, MD, of Germany’s Friedrich-Alexander University Erlangen-Nuremberg and University Hospital Erlangen, said in an interview.
The researchers compared two groups of patients who had no history of COVID-19 and received COVID-19 vaccinations, mostly two shots of the Pfizer-BioNTech vaccine (96%): 84 with IMID (mean age, 53.1 years; 65.5% females) and 182 healthy controls (mean age, 40.8 years; 57.1% females).
The patients with IMID most commonly had spondyloarthritis (32.1%), RA (29.8%), inflammatory bowel disease (9.5%), and psoriasis (9.5%). Nearly 43% of the patients were treated with biologic and targeted synthetic disease-modifying antirheumatic drugs and 23.9% with conventional synthetic DMARDSs. Another 29% were not treated.
All of the controls developed anti–SARS-CoV-2 IgG, but 6% of the patients with IMID did not (P = .003). The gap in development of neutralizing antibodies was even higher: 99.5% of the controls developed neutralizing antibody activity versus 90% of the IMID group. “Neutralizing antibodies are more relevant because the test shows how much the antibodies interfere with the binding of SARS-CoV-2 proteins to the receptor,” Dr. Schett said.
The study authors concluded that “our study provides evidence that, while vaccination against SARS-CoV-2 is well tolerated and even associated with lower incidence of side effects in patients with IMID, its efficacy is somewhat delayed and reduced. Nonetheless, the data also show that, in principle, patients with IMID respond to SARS-CoV-2 vaccination, supporting an aggressive vaccination strategy.”
Lowered antibody response to vaccination for some methotrexate users
In the newer study, led by Rebecca H. Haberman, MD, of New York University Langone Health, researchers examined COVID-19 vaccine response in cohorts in New York City and Erlangen, Germany.
The New York cohort included 25 patients with IMID who were taking methotrexate by itself or with other immunomodulatory medications (mean age, 63.2 years), 26 with IMID who were on anticytokine therapy and/or other oral immunomodulators (mean age, 49.1 years) and 26 healthy controls (mean age, 49.2 years). Most patients with IMID had psoriasis/psoriatic arthritis or RA.
The German validation cohort included 182 healthy subjects (mean age, 45.0 years), 11 subjects with IMID who received TNF inhibitor monotherapy (mean age, 40.8 years), and 20 subjects with IMID on methotrexate monotherapy (mean age, 54.5 years).
In the New York cohort, 96.1% of healthy controls showed “adequate humoral immune response,” along with 92.3% of patients with IMID who weren’t taking methotrexate. However, those on methotrexate had a lower rate of adequate response (72.0%), and the gap persisted even after researchers removed those who showed signs of previous COVID-19 infection (P = .045).
In the German cohort, 98.3% of healthy cohorts and 90.9% of patients with IMID who didn’t receive methotrexate reached an “adequate” humoral response versus just half (50.0%) of those who were taking methotrexate.
When both cohorts are combined, over 90% of the healthy subjects and the patients with IMID on biologic treatments (mainly TNF blockers, n = 37) showed “robust” antibody response. However, only 62% of patients with IMID who took methotrexate (n = 45) reached an “adequate” level of response. The methotrexate gap remained after researchers accounted for differences in age among the cohorts.
What’s going on? “We think that the underlying chronic immune stimulation in autoimmune patients may cause T-cell exhaustion and thus blunts the immune response,” said Dr. Schett, who’s also a coauthor of this study. “In addition, specific drugs such as methotrexate could additionally impair the immune response.”
Still, the findings “reiterate that vaccinations are safe and effective, which is what the recommendations state,” he said, adding that more testing of vaccination immune response is wise.
Insights into vaccine response while on rituximab
Two more reports, also published in Annals of the Rheumatic Diseases, offer insight into vaccine response in patients with IMID who take rituximab.
In one report, published May 11, U.S. researchers retrospectively tracked 89 rheumatic disease patients (76% female; mean age, 61) at a single clinic who’d received at least one dose of a COVID-19 vaccine. Of those, 21 patients showed no sign of vaccine antibody response, and 20 of them were in the group taking rituximab. (The other patient was taking belimumab.) Another 10 patients taking rituximab did show a response.
“Longer duration from most recent rituximab exposure was associated with a greater likelihood of response,” the report’s authors wrote. “The results suggest that time from last rituximab exposure is an important consideration in maximizing the likelihood of a serological response, but this likely is related to the substantial variation in the period of B-cell depletion following rituximab.”
Finally, an Austrian report published May 6 examined COVID-19 vaccine immune response in five patients who were taking rituximab (four with other drugs such as methotrexate and prednisone). Researchers compared them with eight healthy controls, half who’d been vaccinated.
The researchers found evidence that rituximab “may not have to preclude SARS-CoV-2 vaccination, since a cellular immune response will be mounted even in the absence of circulating B cells. Alternatively, in patients with stable disease, delaying [rituximab] treatment until after the second vaccination may be warranted and, therefore, vaccines with a short interval between first and second vaccination or those showing full protection after a single vaccination may be preferable. Importantly, in the presence of circulating B cells also a humoral immune response may be expected despite prior [rituximab] therapy.”
Dr. Bass said the findings reflect growing awareness that “patients with autoimmune disease, especially when they’re on immunosuppressant medications, don’t quite have as optimal responses to the vaccinations.” However, she said, the vaccines are so potent that they’re likely to still have significant efficacy in these patients even if there’s a reduction in response.
What’s next? Dr. Schett said “testing immune response to vaccination is important for patients with autoimmune disease. Some of them may need a third vaccination.”
The American College of Rheumatology’s COVID-19 vaccination guidelines do not recommend third vaccinations or postvaccination immune testing at this time. However, Dr. Bass, one of the coauthors of the recommendations, said it’s likely that postvaccination immune testing and booster shots will become routine.
Dr. Bass reported no relevant disclosures. Dr. Schett reported receiving consulting fees from AbbVie. The May 6 German vaccine study was funded by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, the ERC Synergy grant 4D Nanoscope, the IMI funded project RTCure, the Emerging Fields Initiative MIRACLE of the Friedrich-Alexander-Universität Erlangen-Nürnberg, the Schreiber Stiftung, and the Else Kröner-Memorial Scholarship. The study authors reported no disclosures. The May 25 study of German and American cohorts was funded by the National Institute of Arthritis and Musculoskletal and Skin Diseases, National Institute of Allergy and Infectious Diseases, Rheumatology Research Foundation, Bloomberg Philanthropies COVID-19 Initiative, Pfizer COVID-19 Competitive Grant Program, Beatrice Snyder Foundation, Riley Family Foundation, National Psoriasis Foundation, and Deutsche Forschungsgemeinschaft. The authors reported a range of financial relationships with pharmaceutical companies. No specific funding was reported for the other two studies mentioned.
Ten percent of patients with immune-mediated inflammatory diseases (IMIDs) fail to respond properly to COVID-19 vaccinations regardless of medication, researchers report, and small new studies suggest those on methotrexate and rituximab may be especially vulnerable to vaccine failure.
Even so, it’s still crucially vital for patients with IMIDs to get vaccinated and for clinicians to follow recommendations to temporarily withhold certain medications around the time of vaccination, rheumatologist Anne R. Bass, MD, of Weill Cornell Medicine and the Hospital for Special Surgery, New York, said in an interview. “We’re not making any significant adjustments,” added Dr. Bass, a coauthor of the American College of Rheumatology’s COVID-19 vaccination guidelines for patients with rheumatic and musculoskeletal diseases.
The findings appear in a trio of studies in Annals of the Rheumatic Diseases. The most recent study, which appeared May 25, 2021, found that more than one-third of patients with IMIDs who took methotrexate didn’t produce adequate antibody levels after vaccination versus 10% of those in other groups. (P < .001) A May 11 study found that 20 of 30 patients with rheumatic diseases on rituximab failed to respond to vaccination. And a May 6 study reported that immune responses against SARS-CoV-2 are “somewhat delayed and reduced” in patients with IMID, with 99.5% of a control group developing neutralizing antibody activity after vaccination versus 90% of those with IMID (P = .0008).
Development of neutralizing antibodies somewhat delayed and reduced
Team members were surprised by the high number of vaccine nonresponders in the May 6 IMID study, coauthor Georg Schett, MD, of Germany’s Friedrich-Alexander University Erlangen-Nuremberg and University Hospital Erlangen, said in an interview.
The researchers compared two groups of patients who had no history of COVID-19 and received COVID-19 vaccinations, mostly two shots of the Pfizer-BioNTech vaccine (96%): 84 with IMID (mean age, 53.1 years; 65.5% females) and 182 healthy controls (mean age, 40.8 years; 57.1% females).
The patients with IMID most commonly had spondyloarthritis (32.1%), RA (29.8%), inflammatory bowel disease (9.5%), and psoriasis (9.5%). Nearly 43% of the patients were treated with biologic and targeted synthetic disease-modifying antirheumatic drugs and 23.9% with conventional synthetic DMARDSs. Another 29% were not treated.
All of the controls developed anti–SARS-CoV-2 IgG, but 6% of the patients with IMID did not (P = .003). The gap in development of neutralizing antibodies was even higher: 99.5% of the controls developed neutralizing antibody activity versus 90% of the IMID group. “Neutralizing antibodies are more relevant because the test shows how much the antibodies interfere with the binding of SARS-CoV-2 proteins to the receptor,” Dr. Schett said.
The study authors concluded that “our study provides evidence that, while vaccination against SARS-CoV-2 is well tolerated and even associated with lower incidence of side effects in patients with IMID, its efficacy is somewhat delayed and reduced. Nonetheless, the data also show that, in principle, patients with IMID respond to SARS-CoV-2 vaccination, supporting an aggressive vaccination strategy.”
Lowered antibody response to vaccination for some methotrexate users
In the newer study, led by Rebecca H. Haberman, MD, of New York University Langone Health, researchers examined COVID-19 vaccine response in cohorts in New York City and Erlangen, Germany.
The New York cohort included 25 patients with IMID who were taking methotrexate by itself or with other immunomodulatory medications (mean age, 63.2 years), 26 with IMID who were on anticytokine therapy and/or other oral immunomodulators (mean age, 49.1 years) and 26 healthy controls (mean age, 49.2 years). Most patients with IMID had psoriasis/psoriatic arthritis or RA.
The German validation cohort included 182 healthy subjects (mean age, 45.0 years), 11 subjects with IMID who received TNF inhibitor monotherapy (mean age, 40.8 years), and 20 subjects with IMID on methotrexate monotherapy (mean age, 54.5 years).
In the New York cohort, 96.1% of healthy controls showed “adequate humoral immune response,” along with 92.3% of patients with IMID who weren’t taking methotrexate. However, those on methotrexate had a lower rate of adequate response (72.0%), and the gap persisted even after researchers removed those who showed signs of previous COVID-19 infection (P = .045).
In the German cohort, 98.3% of healthy cohorts and 90.9% of patients with IMID who didn’t receive methotrexate reached an “adequate” humoral response versus just half (50.0%) of those who were taking methotrexate.
When both cohorts are combined, over 90% of the healthy subjects and the patients with IMID on biologic treatments (mainly TNF blockers, n = 37) showed “robust” antibody response. However, only 62% of patients with IMID who took methotrexate (n = 45) reached an “adequate” level of response. The methotrexate gap remained after researchers accounted for differences in age among the cohorts.
What’s going on? “We think that the underlying chronic immune stimulation in autoimmune patients may cause T-cell exhaustion and thus blunts the immune response,” said Dr. Schett, who’s also a coauthor of this study. “In addition, specific drugs such as methotrexate could additionally impair the immune response.”
Still, the findings “reiterate that vaccinations are safe and effective, which is what the recommendations state,” he said, adding that more testing of vaccination immune response is wise.
Insights into vaccine response while on rituximab
Two more reports, also published in Annals of the Rheumatic Diseases, offer insight into vaccine response in patients with IMID who take rituximab.
In one report, published May 11, U.S. researchers retrospectively tracked 89 rheumatic disease patients (76% female; mean age, 61) at a single clinic who’d received at least one dose of a COVID-19 vaccine. Of those, 21 patients showed no sign of vaccine antibody response, and 20 of them were in the group taking rituximab. (The other patient was taking belimumab.) Another 10 patients taking rituximab did show a response.
“Longer duration from most recent rituximab exposure was associated with a greater likelihood of response,” the report’s authors wrote. “The results suggest that time from last rituximab exposure is an important consideration in maximizing the likelihood of a serological response, but this likely is related to the substantial variation in the period of B-cell depletion following rituximab.”
Finally, an Austrian report published May 6 examined COVID-19 vaccine immune response in five patients who were taking rituximab (four with other drugs such as methotrexate and prednisone). Researchers compared them with eight healthy controls, half who’d been vaccinated.
The researchers found evidence that rituximab “may not have to preclude SARS-CoV-2 vaccination, since a cellular immune response will be mounted even in the absence of circulating B cells. Alternatively, in patients with stable disease, delaying [rituximab] treatment until after the second vaccination may be warranted and, therefore, vaccines with a short interval between first and second vaccination or those showing full protection after a single vaccination may be preferable. Importantly, in the presence of circulating B cells also a humoral immune response may be expected despite prior [rituximab] therapy.”
Dr. Bass said the findings reflect growing awareness that “patients with autoimmune disease, especially when they’re on immunosuppressant medications, don’t quite have as optimal responses to the vaccinations.” However, she said, the vaccines are so potent that they’re likely to still have significant efficacy in these patients even if there’s a reduction in response.
What’s next? Dr. Schett said “testing immune response to vaccination is important for patients with autoimmune disease. Some of them may need a third vaccination.”
The American College of Rheumatology’s COVID-19 vaccination guidelines do not recommend third vaccinations or postvaccination immune testing at this time. However, Dr. Bass, one of the coauthors of the recommendations, said it’s likely that postvaccination immune testing and booster shots will become routine.
Dr. Bass reported no relevant disclosures. Dr. Schett reported receiving consulting fees from AbbVie. The May 6 German vaccine study was funded by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, the ERC Synergy grant 4D Nanoscope, the IMI funded project RTCure, the Emerging Fields Initiative MIRACLE of the Friedrich-Alexander-Universität Erlangen-Nürnberg, the Schreiber Stiftung, and the Else Kröner-Memorial Scholarship. The study authors reported no disclosures. The May 25 study of German and American cohorts was funded by the National Institute of Arthritis and Musculoskletal and Skin Diseases, National Institute of Allergy and Infectious Diseases, Rheumatology Research Foundation, Bloomberg Philanthropies COVID-19 Initiative, Pfizer COVID-19 Competitive Grant Program, Beatrice Snyder Foundation, Riley Family Foundation, National Psoriasis Foundation, and Deutsche Forschungsgemeinschaft. The authors reported a range of financial relationships with pharmaceutical companies. No specific funding was reported for the other two studies mentioned.
Ten percent of patients with immune-mediated inflammatory diseases (IMIDs) fail to respond properly to COVID-19 vaccinations regardless of medication, researchers report, and small new studies suggest those on methotrexate and rituximab may be especially vulnerable to vaccine failure.
Even so, it’s still crucially vital for patients with IMIDs to get vaccinated and for clinicians to follow recommendations to temporarily withhold certain medications around the time of vaccination, rheumatologist Anne R. Bass, MD, of Weill Cornell Medicine and the Hospital for Special Surgery, New York, said in an interview. “We’re not making any significant adjustments,” added Dr. Bass, a coauthor of the American College of Rheumatology’s COVID-19 vaccination guidelines for patients with rheumatic and musculoskeletal diseases.
The findings appear in a trio of studies in Annals of the Rheumatic Diseases. The most recent study, which appeared May 25, 2021, found that more than one-third of patients with IMIDs who took methotrexate didn’t produce adequate antibody levels after vaccination versus 10% of those in other groups. (P < .001) A May 11 study found that 20 of 30 patients with rheumatic diseases on rituximab failed to respond to vaccination. And a May 6 study reported that immune responses against SARS-CoV-2 are “somewhat delayed and reduced” in patients with IMID, with 99.5% of a control group developing neutralizing antibody activity after vaccination versus 90% of those with IMID (P = .0008).
Development of neutralizing antibodies somewhat delayed and reduced
Team members were surprised by the high number of vaccine nonresponders in the May 6 IMID study, coauthor Georg Schett, MD, of Germany’s Friedrich-Alexander University Erlangen-Nuremberg and University Hospital Erlangen, said in an interview.
The researchers compared two groups of patients who had no history of COVID-19 and received COVID-19 vaccinations, mostly two shots of the Pfizer-BioNTech vaccine (96%): 84 with IMID (mean age, 53.1 years; 65.5% females) and 182 healthy controls (mean age, 40.8 years; 57.1% females).
The patients with IMID most commonly had spondyloarthritis (32.1%), RA (29.8%), inflammatory bowel disease (9.5%), and psoriasis (9.5%). Nearly 43% of the patients were treated with biologic and targeted synthetic disease-modifying antirheumatic drugs and 23.9% with conventional synthetic DMARDSs. Another 29% were not treated.
All of the controls developed anti–SARS-CoV-2 IgG, but 6% of the patients with IMID did not (P = .003). The gap in development of neutralizing antibodies was even higher: 99.5% of the controls developed neutralizing antibody activity versus 90% of the IMID group. “Neutralizing antibodies are more relevant because the test shows how much the antibodies interfere with the binding of SARS-CoV-2 proteins to the receptor,” Dr. Schett said.
The study authors concluded that “our study provides evidence that, while vaccination against SARS-CoV-2 is well tolerated and even associated with lower incidence of side effects in patients with IMID, its efficacy is somewhat delayed and reduced. Nonetheless, the data also show that, in principle, patients with IMID respond to SARS-CoV-2 vaccination, supporting an aggressive vaccination strategy.”
Lowered antibody response to vaccination for some methotrexate users
In the newer study, led by Rebecca H. Haberman, MD, of New York University Langone Health, researchers examined COVID-19 vaccine response in cohorts in New York City and Erlangen, Germany.
The New York cohort included 25 patients with IMID who were taking methotrexate by itself or with other immunomodulatory medications (mean age, 63.2 years), 26 with IMID who were on anticytokine therapy and/or other oral immunomodulators (mean age, 49.1 years) and 26 healthy controls (mean age, 49.2 years). Most patients with IMID had psoriasis/psoriatic arthritis or RA.
The German validation cohort included 182 healthy subjects (mean age, 45.0 years), 11 subjects with IMID who received TNF inhibitor monotherapy (mean age, 40.8 years), and 20 subjects with IMID on methotrexate monotherapy (mean age, 54.5 years).
In the New York cohort, 96.1% of healthy controls showed “adequate humoral immune response,” along with 92.3% of patients with IMID who weren’t taking methotrexate. However, those on methotrexate had a lower rate of adequate response (72.0%), and the gap persisted even after researchers removed those who showed signs of previous COVID-19 infection (P = .045).
In the German cohort, 98.3% of healthy cohorts and 90.9% of patients with IMID who didn’t receive methotrexate reached an “adequate” humoral response versus just half (50.0%) of those who were taking methotrexate.
When both cohorts are combined, over 90% of the healthy subjects and the patients with IMID on biologic treatments (mainly TNF blockers, n = 37) showed “robust” antibody response. However, only 62% of patients with IMID who took methotrexate (n = 45) reached an “adequate” level of response. The methotrexate gap remained after researchers accounted for differences in age among the cohorts.
What’s going on? “We think that the underlying chronic immune stimulation in autoimmune patients may cause T-cell exhaustion and thus blunts the immune response,” said Dr. Schett, who’s also a coauthor of this study. “In addition, specific drugs such as methotrexate could additionally impair the immune response.”
Still, the findings “reiterate that vaccinations are safe and effective, which is what the recommendations state,” he said, adding that more testing of vaccination immune response is wise.
Insights into vaccine response while on rituximab
Two more reports, also published in Annals of the Rheumatic Diseases, offer insight into vaccine response in patients with IMID who take rituximab.
In one report, published May 11, U.S. researchers retrospectively tracked 89 rheumatic disease patients (76% female; mean age, 61) at a single clinic who’d received at least one dose of a COVID-19 vaccine. Of those, 21 patients showed no sign of vaccine antibody response, and 20 of them were in the group taking rituximab. (The other patient was taking belimumab.) Another 10 patients taking rituximab did show a response.
“Longer duration from most recent rituximab exposure was associated with a greater likelihood of response,” the report’s authors wrote. “The results suggest that time from last rituximab exposure is an important consideration in maximizing the likelihood of a serological response, but this likely is related to the substantial variation in the period of B-cell depletion following rituximab.”
Finally, an Austrian report published May 6 examined COVID-19 vaccine immune response in five patients who were taking rituximab (four with other drugs such as methotrexate and prednisone). Researchers compared them with eight healthy controls, half who’d been vaccinated.
The researchers found evidence that rituximab “may not have to preclude SARS-CoV-2 vaccination, since a cellular immune response will be mounted even in the absence of circulating B cells. Alternatively, in patients with stable disease, delaying [rituximab] treatment until after the second vaccination may be warranted and, therefore, vaccines with a short interval between first and second vaccination or those showing full protection after a single vaccination may be preferable. Importantly, in the presence of circulating B cells also a humoral immune response may be expected despite prior [rituximab] therapy.”
Dr. Bass said the findings reflect growing awareness that “patients with autoimmune disease, especially when they’re on immunosuppressant medications, don’t quite have as optimal responses to the vaccinations.” However, she said, the vaccines are so potent that they’re likely to still have significant efficacy in these patients even if there’s a reduction in response.
What’s next? Dr. Schett said “testing immune response to vaccination is important for patients with autoimmune disease. Some of them may need a third vaccination.”
The American College of Rheumatology’s COVID-19 vaccination guidelines do not recommend third vaccinations or postvaccination immune testing at this time. However, Dr. Bass, one of the coauthors of the recommendations, said it’s likely that postvaccination immune testing and booster shots will become routine.
Dr. Bass reported no relevant disclosures. Dr. Schett reported receiving consulting fees from AbbVie. The May 6 German vaccine study was funded by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, the ERC Synergy grant 4D Nanoscope, the IMI funded project RTCure, the Emerging Fields Initiative MIRACLE of the Friedrich-Alexander-Universität Erlangen-Nürnberg, the Schreiber Stiftung, and the Else Kröner-Memorial Scholarship. The study authors reported no disclosures. The May 25 study of German and American cohorts was funded by the National Institute of Arthritis and Musculoskletal and Skin Diseases, National Institute of Allergy and Infectious Diseases, Rheumatology Research Foundation, Bloomberg Philanthropies COVID-19 Initiative, Pfizer COVID-19 Competitive Grant Program, Beatrice Snyder Foundation, Riley Family Foundation, National Psoriasis Foundation, and Deutsche Forschungsgemeinschaft. The authors reported a range of financial relationships with pharmaceutical companies. No specific funding was reported for the other two studies mentioned.
FROM ANNALS OF THE RHEUMATIC DISEASES
New allergy guidelines call for end to food bans in schools
Children with food allergies often require diligent monitoring and a restricted diet to reduce allergic attacks, but there is little evidence available to support so-called “food bans” at schools and childcare centers.
Instead, a new practice guideline published earlier this month in the Journal of Allergy and Clinical Immunology calls for better allergy management training for staff, as well as increased epinephrine availability in educational environments. The guidelines were developed by an international panel of clinicians, school personnel, and parents.
The guidance at a glance
Rather than creating site-wide food prohibitions on nuts, dairy, and other allergenic foods, the practice guidance recommends centers and schools use “common-sense approaches” to reduce allergic reaction risk among school-aged children. According to the guideline authors, these strategies could include promoting handwashing, providing adult supervision during snacks and meals, and cleaning surfaces where food is either eaten or prepared.
Additionally, the new evidence-based guidance calls for schools and childcare centers to teach school personnel to recognize, prevent, and respond appropriately to food-related allergic reactions when they do occur.
The guidance also recommends that educational institutions require an up-to-date allergy ‘action plan’ from parents, designed for their children with allergies. These action plans can be integrated into the training of teachers and nurses to help manage potential allergic reactions.
Moreover, the guidance suggests schools should keep unassigned epinephrine autoinjectors in stock, both on site and even when traveling, where laws permit, rather than requiring students with allergies to bring in their own autoinjectors. Ultimately, this represents a more proactive approach to treating anaphylaxis, particularly in settings where treatment is urgently needed, such as when students are away from campus and participating in a school-designated trip or event.
Expert perspectives
Jennifer A. Dantzer, MD, MHS, allergist-immunologist and assistant professor of pediatrics at Johns Hopkins University, Baltimore, told this news organization via email that the practice guidelines offer an important starting point for ensuring quality of life of students, parents, and other school personnel.
While the Centers for Disease Control and Prevention published voluntary guidance for managing food allergies in schools back in 2013, there has since “been a lack of universal policies and procedures to manage the risk of allergic reactions in schools,” explained Dr. Dantzer. “The new guidelines are a good first step of using available evidence and all the key stakeholders, clinicians, school personnel, and families to figure out the best way to keep children with food allergies safe at school.”
Dr. Dantzer wasn’t involved in the creation of the new practice guidelines, but she shared how her clinical experience reinforces the need for the evidence-based recommendations. “Every single week we talk with families, both in clinic and in our research studies, about living with food allergies, and we recognize that every child is different,” she said. “We constantly work to advocate for each individual child with food allergies.”
Pediatric allergist Malika Gupta, MBBS, MD, said in an interview via email that the guidelines could assist in the creation of new nationwide policies for food allergy management at schools. “Also, the guidelines are labeled ‘conditional,’ which gives policymakers the ability to adapt to their specific circumstances and individuals, as well as make modifications according to regional trends,” she added.
Dr. Gupta, a clinical assistant professor in the Division of Allergy and Clinical Immunology at the University of Michigan, Ann Arbor, echoed the guideline panel’s sentiments regarding food bans, explaining that prohibiting certain foods could lend a “false sense of security” and could also “promote bullying and a sense of isolation for the food-allergic child.” In spite of the lack of evidence supporting food bans, Dr. Gupta noted that these bans can give families a sense of control and security. Ideally, more research should be performed to determine whether food bans actually work, she added.
In addition to promoting the new guidelines, allergists and pediatricians can also implement proactive allergy reaction mitigation strategies that work with school systems, according to Dr. Gupta. “In-clinic, we ensure all families have food allergy action plans for school and current epinephrine auto-injectors,” she said. “We also often have our food allergy nurses educate schools when food allergy awareness is a concern.”
Many of the 25 authors of the food allergy guidelines disclosed relevant financial relationships. The full list is with the original article. According to a footnote within the guidelines, “Panel members who were deemed to have a real, perceived, or potential conflict of interest were asked to abstain from voting on recommendations related to that interest.”
A version of this article first appeared on Medscape.com.
Children with food allergies often require diligent monitoring and a restricted diet to reduce allergic attacks, but there is little evidence available to support so-called “food bans” at schools and childcare centers.
Instead, a new practice guideline published earlier this month in the Journal of Allergy and Clinical Immunology calls for better allergy management training for staff, as well as increased epinephrine availability in educational environments. The guidelines were developed by an international panel of clinicians, school personnel, and parents.
The guidance at a glance
Rather than creating site-wide food prohibitions on nuts, dairy, and other allergenic foods, the practice guidance recommends centers and schools use “common-sense approaches” to reduce allergic reaction risk among school-aged children. According to the guideline authors, these strategies could include promoting handwashing, providing adult supervision during snacks and meals, and cleaning surfaces where food is either eaten or prepared.
Additionally, the new evidence-based guidance calls for schools and childcare centers to teach school personnel to recognize, prevent, and respond appropriately to food-related allergic reactions when they do occur.
The guidance also recommends that educational institutions require an up-to-date allergy ‘action plan’ from parents, designed for their children with allergies. These action plans can be integrated into the training of teachers and nurses to help manage potential allergic reactions.
Moreover, the guidance suggests schools should keep unassigned epinephrine autoinjectors in stock, both on site and even when traveling, where laws permit, rather than requiring students with allergies to bring in their own autoinjectors. Ultimately, this represents a more proactive approach to treating anaphylaxis, particularly in settings where treatment is urgently needed, such as when students are away from campus and participating in a school-designated trip or event.
Expert perspectives
Jennifer A. Dantzer, MD, MHS, allergist-immunologist and assistant professor of pediatrics at Johns Hopkins University, Baltimore, told this news organization via email that the practice guidelines offer an important starting point for ensuring quality of life of students, parents, and other school personnel.
While the Centers for Disease Control and Prevention published voluntary guidance for managing food allergies in schools back in 2013, there has since “been a lack of universal policies and procedures to manage the risk of allergic reactions in schools,” explained Dr. Dantzer. “The new guidelines are a good first step of using available evidence and all the key stakeholders, clinicians, school personnel, and families to figure out the best way to keep children with food allergies safe at school.”
Dr. Dantzer wasn’t involved in the creation of the new practice guidelines, but she shared how her clinical experience reinforces the need for the evidence-based recommendations. “Every single week we talk with families, both in clinic and in our research studies, about living with food allergies, and we recognize that every child is different,” she said. “We constantly work to advocate for each individual child with food allergies.”
Pediatric allergist Malika Gupta, MBBS, MD, said in an interview via email that the guidelines could assist in the creation of new nationwide policies for food allergy management at schools. “Also, the guidelines are labeled ‘conditional,’ which gives policymakers the ability to adapt to their specific circumstances and individuals, as well as make modifications according to regional trends,” she added.
Dr. Gupta, a clinical assistant professor in the Division of Allergy and Clinical Immunology at the University of Michigan, Ann Arbor, echoed the guideline panel’s sentiments regarding food bans, explaining that prohibiting certain foods could lend a “false sense of security” and could also “promote bullying and a sense of isolation for the food-allergic child.” In spite of the lack of evidence supporting food bans, Dr. Gupta noted that these bans can give families a sense of control and security. Ideally, more research should be performed to determine whether food bans actually work, she added.
In addition to promoting the new guidelines, allergists and pediatricians can also implement proactive allergy reaction mitigation strategies that work with school systems, according to Dr. Gupta. “In-clinic, we ensure all families have food allergy action plans for school and current epinephrine auto-injectors,” she said. “We also often have our food allergy nurses educate schools when food allergy awareness is a concern.”
Many of the 25 authors of the food allergy guidelines disclosed relevant financial relationships. The full list is with the original article. According to a footnote within the guidelines, “Panel members who were deemed to have a real, perceived, or potential conflict of interest were asked to abstain from voting on recommendations related to that interest.”
A version of this article first appeared on Medscape.com.
Children with food allergies often require diligent monitoring and a restricted diet to reduce allergic attacks, but there is little evidence available to support so-called “food bans” at schools and childcare centers.
Instead, a new practice guideline published earlier this month in the Journal of Allergy and Clinical Immunology calls for better allergy management training for staff, as well as increased epinephrine availability in educational environments. The guidelines were developed by an international panel of clinicians, school personnel, and parents.
The guidance at a glance
Rather than creating site-wide food prohibitions on nuts, dairy, and other allergenic foods, the practice guidance recommends centers and schools use “common-sense approaches” to reduce allergic reaction risk among school-aged children. According to the guideline authors, these strategies could include promoting handwashing, providing adult supervision during snacks and meals, and cleaning surfaces where food is either eaten or prepared.
Additionally, the new evidence-based guidance calls for schools and childcare centers to teach school personnel to recognize, prevent, and respond appropriately to food-related allergic reactions when they do occur.
The guidance also recommends that educational institutions require an up-to-date allergy ‘action plan’ from parents, designed for their children with allergies. These action plans can be integrated into the training of teachers and nurses to help manage potential allergic reactions.
Moreover, the guidance suggests schools should keep unassigned epinephrine autoinjectors in stock, both on site and even when traveling, where laws permit, rather than requiring students with allergies to bring in their own autoinjectors. Ultimately, this represents a more proactive approach to treating anaphylaxis, particularly in settings where treatment is urgently needed, such as when students are away from campus and participating in a school-designated trip or event.
Expert perspectives
Jennifer A. Dantzer, MD, MHS, allergist-immunologist and assistant professor of pediatrics at Johns Hopkins University, Baltimore, told this news organization via email that the practice guidelines offer an important starting point for ensuring quality of life of students, parents, and other school personnel.
While the Centers for Disease Control and Prevention published voluntary guidance for managing food allergies in schools back in 2013, there has since “been a lack of universal policies and procedures to manage the risk of allergic reactions in schools,” explained Dr. Dantzer. “The new guidelines are a good first step of using available evidence and all the key stakeholders, clinicians, school personnel, and families to figure out the best way to keep children with food allergies safe at school.”
Dr. Dantzer wasn’t involved in the creation of the new practice guidelines, but she shared how her clinical experience reinforces the need for the evidence-based recommendations. “Every single week we talk with families, both in clinic and in our research studies, about living with food allergies, and we recognize that every child is different,” she said. “We constantly work to advocate for each individual child with food allergies.”
Pediatric allergist Malika Gupta, MBBS, MD, said in an interview via email that the guidelines could assist in the creation of new nationwide policies for food allergy management at schools. “Also, the guidelines are labeled ‘conditional,’ which gives policymakers the ability to adapt to their specific circumstances and individuals, as well as make modifications according to regional trends,” she added.
Dr. Gupta, a clinical assistant professor in the Division of Allergy and Clinical Immunology at the University of Michigan, Ann Arbor, echoed the guideline panel’s sentiments regarding food bans, explaining that prohibiting certain foods could lend a “false sense of security” and could also “promote bullying and a sense of isolation for the food-allergic child.” In spite of the lack of evidence supporting food bans, Dr. Gupta noted that these bans can give families a sense of control and security. Ideally, more research should be performed to determine whether food bans actually work, she added.
In addition to promoting the new guidelines, allergists and pediatricians can also implement proactive allergy reaction mitigation strategies that work with school systems, according to Dr. Gupta. “In-clinic, we ensure all families have food allergy action plans for school and current epinephrine auto-injectors,” she said. “We also often have our food allergy nurses educate schools when food allergy awareness is a concern.”
Many of the 25 authors of the food allergy guidelines disclosed relevant financial relationships. The full list is with the original article. According to a footnote within the guidelines, “Panel members who were deemed to have a real, perceived, or potential conflict of interest were asked to abstain from voting on recommendations related to that interest.”
A version of this article first appeared on Medscape.com.
COVID-19 vaccination rate rising quickly among adolescents
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.
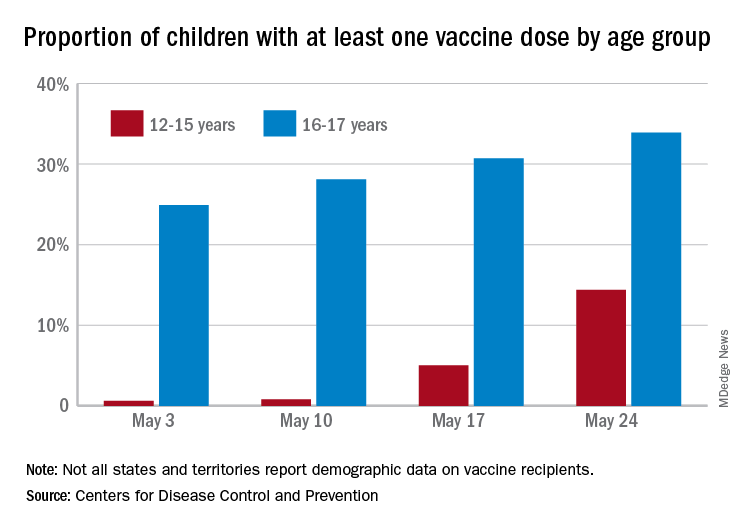
As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.

As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.

As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
COVID-19 vaccination and pregnancy: Benefits outweigh the risks, for now
Vaccines have been a lifesaving public health measure since 1000 CE, when the Chinese first used smallpox inoculations to induce immunity.1 Work by pioneers such as Edward Jenner, Louis Pasteur, and Maurice Hilleman has averted countless millions of vaccine-preventable illnesses and deaths, and vaccines have become a routine part of health maintenance throughout the human life cycle.
Pregnant patients who receive vaccines often have an added benefit of protection provided to their infants through passive transfer of antibodies. Several vaccine platforms have been utilized in pregnancy with well-documented improvements in maternal and obstetric outcomes as well as improved neonatal outcomes in the first several months of life.
Risks of COVID-19 in pregnancy
The COVID-19 pandemic placed a spotlight on medically at-risk groups. Pregnant women are 3 times more likely to require admission to the intensive care unit, have increased requirement for extracorporeal membrane oxygenation treatment, and are up to 70% more likely to die than nonpregnant peers—and this risk increases with the presence of additional comorbidities.
In the case of COVID-19, vaccination trials that have shaped worldwide clinical practice unfortunately followed the historical trend of excluding pregnant patients from participation. This has required clinicians to guide their patients through the decision of whether or not to accept vaccination without having the same reassurances regarding safety and effectiveness afforded to their nonpregnant counterparts. With more than 86,000 pregnant women infected with COVID-19 through April 19, 2021, this lack of information regarding vaccine safety in pregnancy is a significant public health gap.2
COVID-19 vaccines
The current COVID-19 vaccines approved for use in the United States under an Emergency Use Authorization issued by the US Food and Drug Administration are nonreplicating and thus cannot cause infection in the mother or fetus. These are the Pfizer-BioNTech mRNA vaccine, the Moderna mRNA-1273 vaccine, and the Janssen Biotech Inc. monovalent vaccine. Furthermore, in animal studies that included the Pfizer-BioNTech, Moderna, or Janssen COVID-19 vaccines, no fetal, embryonal, female reproductive, or postnatal development safety concerns were demonstrated.
As of April 19, 2021, 94,335 pregnant women had received a COVID-19 vaccination, and 4,622 of these enrolled in the Centers for Disease Control and Prevention’s (CDC) V-safe Vaccine Pregnancy Registry.3 The data reported noted no unexpected pregnancy or infant outcomes related to COVID-19 vaccination in pregnancy. Adverse effects of the vaccine were similar to those in nonpregnant cohorts. Additionally, emerging data suggest passage of immunity to neonates, with maternal antibodies demonstrated in cord blood at time of delivery as well as in breast milk.4 To date, these data mainly have come from women immunized with the Moderna and Pfizer-BioNTech mRNA vaccines.
Counseling pregnant patients
Our counseling aligns with that of the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, and the CDC’s Advisory Committee on Immunization Practices in that COVID-19 vaccination should not be withheld from pregnant patients or patients who want to become pregnant. In pregnant patients with comorbidities that place them at higher risk for severe COVID-19 infection, all available formulations of the COVID-19 vaccination should be strongly considered.
As evidence for vaccination safety continues to emerge, patients should continue to discuss their individual needs for vaccination in a shared decision-making format with their obstetric providers.
Boylston A. The origins of inoculation. J R Soc Med. 2012;105:309-313.
Centers for Disease Control and Prevention. COVID data tracker. Data on COVID-19 during pregnancy: severity of maternal illness. https://covid.cdc.gov/covid-data-tracker/#pregnant-population. Accessed April 19, 2021.
Centers for Disease Control and Prevention. V-safe COVID-19 Vaccine Pregnancy Registry. https://www.cdc.gov/coronavirus/2019- ncov/vaccines/safety/vsafepregnancyregistry.html. Updated May 3, 2021. Accessed April 19, 2021.
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021;S0002-9378(21)00187-3. doi: 10.1016/j.ajog.2021.03.023
Vaccines have been a lifesaving public health measure since 1000 CE, when the Chinese first used smallpox inoculations to induce immunity.1 Work by pioneers such as Edward Jenner, Louis Pasteur, and Maurice Hilleman has averted countless millions of vaccine-preventable illnesses and deaths, and vaccines have become a routine part of health maintenance throughout the human life cycle.
Pregnant patients who receive vaccines often have an added benefit of protection provided to their infants through passive transfer of antibodies. Several vaccine platforms have been utilized in pregnancy with well-documented improvements in maternal and obstetric outcomes as well as improved neonatal outcomes in the first several months of life.
Risks of COVID-19 in pregnancy
The COVID-19 pandemic placed a spotlight on medically at-risk groups. Pregnant women are 3 times more likely to require admission to the intensive care unit, have increased requirement for extracorporeal membrane oxygenation treatment, and are up to 70% more likely to die than nonpregnant peers—and this risk increases with the presence of additional comorbidities.
In the case of COVID-19, vaccination trials that have shaped worldwide clinical practice unfortunately followed the historical trend of excluding pregnant patients from participation. This has required clinicians to guide their patients through the decision of whether or not to accept vaccination without having the same reassurances regarding safety and effectiveness afforded to their nonpregnant counterparts. With more than 86,000 pregnant women infected with COVID-19 through April 19, 2021, this lack of information regarding vaccine safety in pregnancy is a significant public health gap.2
COVID-19 vaccines
The current COVID-19 vaccines approved for use in the United States under an Emergency Use Authorization issued by the US Food and Drug Administration are nonreplicating and thus cannot cause infection in the mother or fetus. These are the Pfizer-BioNTech mRNA vaccine, the Moderna mRNA-1273 vaccine, and the Janssen Biotech Inc. monovalent vaccine. Furthermore, in animal studies that included the Pfizer-BioNTech, Moderna, or Janssen COVID-19 vaccines, no fetal, embryonal, female reproductive, or postnatal development safety concerns were demonstrated.
As of April 19, 2021, 94,335 pregnant women had received a COVID-19 vaccination, and 4,622 of these enrolled in the Centers for Disease Control and Prevention’s (CDC) V-safe Vaccine Pregnancy Registry.3 The data reported noted no unexpected pregnancy or infant outcomes related to COVID-19 vaccination in pregnancy. Adverse effects of the vaccine were similar to those in nonpregnant cohorts. Additionally, emerging data suggest passage of immunity to neonates, with maternal antibodies demonstrated in cord blood at time of delivery as well as in breast milk.4 To date, these data mainly have come from women immunized with the Moderna and Pfizer-BioNTech mRNA vaccines.
Counseling pregnant patients
Our counseling aligns with that of the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, and the CDC’s Advisory Committee on Immunization Practices in that COVID-19 vaccination should not be withheld from pregnant patients or patients who want to become pregnant. In pregnant patients with comorbidities that place them at higher risk for severe COVID-19 infection, all available formulations of the COVID-19 vaccination should be strongly considered.
As evidence for vaccination safety continues to emerge, patients should continue to discuss their individual needs for vaccination in a shared decision-making format with their obstetric providers.
Vaccines have been a lifesaving public health measure since 1000 CE, when the Chinese first used smallpox inoculations to induce immunity.1 Work by pioneers such as Edward Jenner, Louis Pasteur, and Maurice Hilleman has averted countless millions of vaccine-preventable illnesses and deaths, and vaccines have become a routine part of health maintenance throughout the human life cycle.
Pregnant patients who receive vaccines often have an added benefit of protection provided to their infants through passive transfer of antibodies. Several vaccine platforms have been utilized in pregnancy with well-documented improvements in maternal and obstetric outcomes as well as improved neonatal outcomes in the first several months of life.
Risks of COVID-19 in pregnancy
The COVID-19 pandemic placed a spotlight on medically at-risk groups. Pregnant women are 3 times more likely to require admission to the intensive care unit, have increased requirement for extracorporeal membrane oxygenation treatment, and are up to 70% more likely to die than nonpregnant peers—and this risk increases with the presence of additional comorbidities.
In the case of COVID-19, vaccination trials that have shaped worldwide clinical practice unfortunately followed the historical trend of excluding pregnant patients from participation. This has required clinicians to guide their patients through the decision of whether or not to accept vaccination without having the same reassurances regarding safety and effectiveness afforded to their nonpregnant counterparts. With more than 86,000 pregnant women infected with COVID-19 through April 19, 2021, this lack of information regarding vaccine safety in pregnancy is a significant public health gap.2
COVID-19 vaccines
The current COVID-19 vaccines approved for use in the United States under an Emergency Use Authorization issued by the US Food and Drug Administration are nonreplicating and thus cannot cause infection in the mother or fetus. These are the Pfizer-BioNTech mRNA vaccine, the Moderna mRNA-1273 vaccine, and the Janssen Biotech Inc. monovalent vaccine. Furthermore, in animal studies that included the Pfizer-BioNTech, Moderna, or Janssen COVID-19 vaccines, no fetal, embryonal, female reproductive, or postnatal development safety concerns were demonstrated.
As of April 19, 2021, 94,335 pregnant women had received a COVID-19 vaccination, and 4,622 of these enrolled in the Centers for Disease Control and Prevention’s (CDC) V-safe Vaccine Pregnancy Registry.3 The data reported noted no unexpected pregnancy or infant outcomes related to COVID-19 vaccination in pregnancy. Adverse effects of the vaccine were similar to those in nonpregnant cohorts. Additionally, emerging data suggest passage of immunity to neonates, with maternal antibodies demonstrated in cord blood at time of delivery as well as in breast milk.4 To date, these data mainly have come from women immunized with the Moderna and Pfizer-BioNTech mRNA vaccines.
Counseling pregnant patients
Our counseling aligns with that of the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, and the CDC’s Advisory Committee on Immunization Practices in that COVID-19 vaccination should not be withheld from pregnant patients or patients who want to become pregnant. In pregnant patients with comorbidities that place them at higher risk for severe COVID-19 infection, all available formulations of the COVID-19 vaccination should be strongly considered.
As evidence for vaccination safety continues to emerge, patients should continue to discuss their individual needs for vaccination in a shared decision-making format with their obstetric providers.
Boylston A. The origins of inoculation. J R Soc Med. 2012;105:309-313.
Centers for Disease Control and Prevention. COVID data tracker. Data on COVID-19 during pregnancy: severity of maternal illness. https://covid.cdc.gov/covid-data-tracker/#pregnant-population. Accessed April 19, 2021.
Centers for Disease Control and Prevention. V-safe COVID-19 Vaccine Pregnancy Registry. https://www.cdc.gov/coronavirus/2019- ncov/vaccines/safety/vsafepregnancyregistry.html. Updated May 3, 2021. Accessed April 19, 2021.
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021;S0002-9378(21)00187-3. doi: 10.1016/j.ajog.2021.03.023
Boylston A. The origins of inoculation. J R Soc Med. 2012;105:309-313.
Centers for Disease Control and Prevention. COVID data tracker. Data on COVID-19 during pregnancy: severity of maternal illness. https://covid.cdc.gov/covid-data-tracker/#pregnant-population. Accessed April 19, 2021.
Centers for Disease Control and Prevention. V-safe COVID-19 Vaccine Pregnancy Registry. https://www.cdc.gov/coronavirus/2019- ncov/vaccines/safety/vsafepregnancyregistry.html. Updated May 3, 2021. Accessed April 19, 2021.
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021;S0002-9378(21)00187-3. doi: 10.1016/j.ajog.2021.03.023
Tezepelumab reduces serious exacerbations in severe asthma
Results from the NAVIGATOR study of tezepelumab showed that treatment of adults and adolescents with severe, uncontrolled asthma with the new biologic led to a large reduction in exacerbations requiring hospital stays and ED visits.
Tezepelumab, codeveloped by Amgen and AstraZeneca, has a novel mechanism of action. It blocks thymic stromal lymphopoietin (TSLP), which is a cytokine produced by epithelial cells. TSLP levels correlate with airway obstruction, severity of disease, and glucocorticoid resistance. TSLP is involved in T2 inflammation within the airway, but also plays a role in the interactions between airway cells and immune cells, which doesn’t rely only solely on T2 inflammation. That broad mechanism of action distinguishes tezepelumab from most other biologics for the treatment of asthma, which are more targeted.
“By working at the top of the cascade, tezepelumab helps stop inflammation at a key source. Clinical trials with tezepelumab showed a clinical benefit in patients irrespective of their baseline biomarker level, including patients with low eosinophil levels at baseline,” said Jean-Pierre Llanos-Ackert, MD, who is executive medical director and global medical affairs lead for tezepelumab at Amgen.
The primary endpoint data look robust, according to Praveen Akuthota, MD, who is an associate professor of medicine at the University of California, San Diego, and comoderated the session at the American Thoracic Society’s virtual international conference, where the research was presented. The study was also published on May 13, 2021, in the New England Journal of Medicine. The conference session included updated results.
The drug holds promise, but more study is needed. “The question really will be, how is this drug different from the existing biologics? How much better is this drug in patients who have borderline T2 biomarkers, or even low T2. The study does show some efficacy in patients whose T2 signals may not be as robust. We’ll have to see with ongoing longitudinal data, how this drug positions, compared to the other agents. It’s obviously exciting, though, to have another option, given that we know what our current armamentarium of agents there are still nonresponders,” said Dr. Akuthota in an interview.
The other comoderator in the session, Laura Crotty Alexander, MD, commented: “It seems like it might work possibly even better than some that are directly covering one pathway only. Hopefully, this agent will be efficacious in a broader population than some of the more targeted biologics.” Dr. Alexander is an associate professor of medicine at the University of California, San Diego, and section chief of pulmonary critical care at the Veterans Affairs San Diego Healthcare System.
She pointed out that physicians often think of asthma patients in broad brush terms, as high or low T2, or T2 high and Th1 or neutrophilic or obese, but many patients present a more complicated picture. “There is some overlap across those phenotypes, such that an agent that works really well for one group doesn’t mean that it won’t have an impact, especially clinically, on some of these other phenotypes,” said Dr. Alexander.
Dr. Akuthota agreed. “Having options for patients whose biomarkers are not maybe as clear is, I think, important.”
Promising results
The study included 1,059 patients aged 12-80 who received 210 mg tezepelumab or placebo. Over 52 weeks, the treatment group had a 79% reduction in exacerbations requiring hospitalization or an ED visit, compared with placebo (rate ratio, 0.21; 95% confidence interval, 0.12-0.37), and an 85% reduction in exacerbations requiring hospitalization (RR, 0.15; 95% CI, 0.07-0.33). The drug increased the time to first exacerbation requiring hospitalization that required hospitalization or an ED visit, reducing risk by 65% (hazard ratio, 0.35; 95% CI, 0.22-0.56).
Fewer patients in the treatment group than placebo used asthma-related health care resources, including: ED visits (32 vs. 94), unscheduled visit to a specialist (285 vs. 406), telephone calls to a health care provider (234 vs. 599), ambulance transport (5 vs. 22), and home visits from a health care provider (18 vs. 22). Fewer patients in the tezepelumab group had hospital stays (3.2% vs. 7.0%), and they had a lower total number of hospital days (108 vs. 497) and days in the ICU (0 vs. 31).
The study was funded by Amgen and AstraZeneca. Dr. Llanos-Ackert is an employee of Amgen. Dr. Alexander has no relevant financial disclosures. Dr. Akuthota has consulted for AstraZeneca and participated in their clinical trials.
Results from the NAVIGATOR study of tezepelumab showed that treatment of adults and adolescents with severe, uncontrolled asthma with the new biologic led to a large reduction in exacerbations requiring hospital stays and ED visits.
Tezepelumab, codeveloped by Amgen and AstraZeneca, has a novel mechanism of action. It blocks thymic stromal lymphopoietin (TSLP), which is a cytokine produced by epithelial cells. TSLP levels correlate with airway obstruction, severity of disease, and glucocorticoid resistance. TSLP is involved in T2 inflammation within the airway, but also plays a role in the interactions between airway cells and immune cells, which doesn’t rely only solely on T2 inflammation. That broad mechanism of action distinguishes tezepelumab from most other biologics for the treatment of asthma, which are more targeted.
“By working at the top of the cascade, tezepelumab helps stop inflammation at a key source. Clinical trials with tezepelumab showed a clinical benefit in patients irrespective of their baseline biomarker level, including patients with low eosinophil levels at baseline,” said Jean-Pierre Llanos-Ackert, MD, who is executive medical director and global medical affairs lead for tezepelumab at Amgen.
The primary endpoint data look robust, according to Praveen Akuthota, MD, who is an associate professor of medicine at the University of California, San Diego, and comoderated the session at the American Thoracic Society’s virtual international conference, where the research was presented. The study was also published on May 13, 2021, in the New England Journal of Medicine. The conference session included updated results.
The drug holds promise, but more study is needed. “The question really will be, how is this drug different from the existing biologics? How much better is this drug in patients who have borderline T2 biomarkers, or even low T2. The study does show some efficacy in patients whose T2 signals may not be as robust. We’ll have to see with ongoing longitudinal data, how this drug positions, compared to the other agents. It’s obviously exciting, though, to have another option, given that we know what our current armamentarium of agents there are still nonresponders,” said Dr. Akuthota in an interview.
The other comoderator in the session, Laura Crotty Alexander, MD, commented: “It seems like it might work possibly even better than some that are directly covering one pathway only. Hopefully, this agent will be efficacious in a broader population than some of the more targeted biologics.” Dr. Alexander is an associate professor of medicine at the University of California, San Diego, and section chief of pulmonary critical care at the Veterans Affairs San Diego Healthcare System.
She pointed out that physicians often think of asthma patients in broad brush terms, as high or low T2, or T2 high and Th1 or neutrophilic or obese, but many patients present a more complicated picture. “There is some overlap across those phenotypes, such that an agent that works really well for one group doesn’t mean that it won’t have an impact, especially clinically, on some of these other phenotypes,” said Dr. Alexander.
Dr. Akuthota agreed. “Having options for patients whose biomarkers are not maybe as clear is, I think, important.”
Promising results
The study included 1,059 patients aged 12-80 who received 210 mg tezepelumab or placebo. Over 52 weeks, the treatment group had a 79% reduction in exacerbations requiring hospitalization or an ED visit, compared with placebo (rate ratio, 0.21; 95% confidence interval, 0.12-0.37), and an 85% reduction in exacerbations requiring hospitalization (RR, 0.15; 95% CI, 0.07-0.33). The drug increased the time to first exacerbation requiring hospitalization that required hospitalization or an ED visit, reducing risk by 65% (hazard ratio, 0.35; 95% CI, 0.22-0.56).
Fewer patients in the treatment group than placebo used asthma-related health care resources, including: ED visits (32 vs. 94), unscheduled visit to a specialist (285 vs. 406), telephone calls to a health care provider (234 vs. 599), ambulance transport (5 vs. 22), and home visits from a health care provider (18 vs. 22). Fewer patients in the tezepelumab group had hospital stays (3.2% vs. 7.0%), and they had a lower total number of hospital days (108 vs. 497) and days in the ICU (0 vs. 31).
The study was funded by Amgen and AstraZeneca. Dr. Llanos-Ackert is an employee of Amgen. Dr. Alexander has no relevant financial disclosures. Dr. Akuthota has consulted for AstraZeneca and participated in their clinical trials.
Results from the NAVIGATOR study of tezepelumab showed that treatment of adults and adolescents with severe, uncontrolled asthma with the new biologic led to a large reduction in exacerbations requiring hospital stays and ED visits.
Tezepelumab, codeveloped by Amgen and AstraZeneca, has a novel mechanism of action. It blocks thymic stromal lymphopoietin (TSLP), which is a cytokine produced by epithelial cells. TSLP levels correlate with airway obstruction, severity of disease, and glucocorticoid resistance. TSLP is involved in T2 inflammation within the airway, but also plays a role in the interactions between airway cells and immune cells, which doesn’t rely only solely on T2 inflammation. That broad mechanism of action distinguishes tezepelumab from most other biologics for the treatment of asthma, which are more targeted.
“By working at the top of the cascade, tezepelumab helps stop inflammation at a key source. Clinical trials with tezepelumab showed a clinical benefit in patients irrespective of their baseline biomarker level, including patients with low eosinophil levels at baseline,” said Jean-Pierre Llanos-Ackert, MD, who is executive medical director and global medical affairs lead for tezepelumab at Amgen.
The primary endpoint data look robust, according to Praveen Akuthota, MD, who is an associate professor of medicine at the University of California, San Diego, and comoderated the session at the American Thoracic Society’s virtual international conference, where the research was presented. The study was also published on May 13, 2021, in the New England Journal of Medicine. The conference session included updated results.
The drug holds promise, but more study is needed. “The question really will be, how is this drug different from the existing biologics? How much better is this drug in patients who have borderline T2 biomarkers, or even low T2. The study does show some efficacy in patients whose T2 signals may not be as robust. We’ll have to see with ongoing longitudinal data, how this drug positions, compared to the other agents. It’s obviously exciting, though, to have another option, given that we know what our current armamentarium of agents there are still nonresponders,” said Dr. Akuthota in an interview.
The other comoderator in the session, Laura Crotty Alexander, MD, commented: “It seems like it might work possibly even better than some that are directly covering one pathway only. Hopefully, this agent will be efficacious in a broader population than some of the more targeted biologics.” Dr. Alexander is an associate professor of medicine at the University of California, San Diego, and section chief of pulmonary critical care at the Veterans Affairs San Diego Healthcare System.
She pointed out that physicians often think of asthma patients in broad brush terms, as high or low T2, or T2 high and Th1 or neutrophilic or obese, but many patients present a more complicated picture. “There is some overlap across those phenotypes, such that an agent that works really well for one group doesn’t mean that it won’t have an impact, especially clinically, on some of these other phenotypes,” said Dr. Alexander.
Dr. Akuthota agreed. “Having options for patients whose biomarkers are not maybe as clear is, I think, important.”
Promising results
The study included 1,059 patients aged 12-80 who received 210 mg tezepelumab or placebo. Over 52 weeks, the treatment group had a 79% reduction in exacerbations requiring hospitalization or an ED visit, compared with placebo (rate ratio, 0.21; 95% confidence interval, 0.12-0.37), and an 85% reduction in exacerbations requiring hospitalization (RR, 0.15; 95% CI, 0.07-0.33). The drug increased the time to first exacerbation requiring hospitalization that required hospitalization or an ED visit, reducing risk by 65% (hazard ratio, 0.35; 95% CI, 0.22-0.56).
Fewer patients in the treatment group than placebo used asthma-related health care resources, including: ED visits (32 vs. 94), unscheduled visit to a specialist (285 vs. 406), telephone calls to a health care provider (234 vs. 599), ambulance transport (5 vs. 22), and home visits from a health care provider (18 vs. 22). Fewer patients in the tezepelumab group had hospital stays (3.2% vs. 7.0%), and they had a lower total number of hospital days (108 vs. 497) and days in the ICU (0 vs. 31).
The study was funded by Amgen and AstraZeneca. Dr. Llanos-Ackert is an employee of Amgen. Dr. Alexander has no relevant financial disclosures. Dr. Akuthota has consulted for AstraZeneca and participated in their clinical trials.
FROM ATS 2021
Mild cortisol excess increases mortality in adrenal incidentaloma
Mortality is two to three times higher in patients with adrenal incidentalomas who have autonomous cortisol secretion levels of 83 nmol/L (3 µg/dL) or more after a 1 mg dexamethasone suppression test (DST), compared with those with levels below this, new research finds.
Autonomous cortisol secretion (ACS) has been linked to hypertension, cardiovascular disease, type 2 diabetes, and early mortality, and risks vary by cortisol level.
“To adequately decide whether treatment should be surgery or medical management of possible complications, it is essential to know the risk associated with the actual level of ACS,” write Albin Kjellbom, MD, of Skåne University Hospital, Lund, Sweden, and colleagues, in their article published May 25 in Annals of Internal Medicine.
Asked to comment, Salila Kurra, MD, of the Columbia adrenal center, Columbia University, New York, told this news organization that “this idea that mild cortisol excess that doesn’t meet the threshold for overt Cushing’s can, in and of itself, cause increased morbidity and mortality is something people have been thinking about for many years now.”
“But there isn’t very clear guidance on exactly what to do in that situation, whether the incidentaloma should be removed, medically managed, or the patient should just be watched ... It may be clinically significant, but the way to sort that out is to do other testing.”
Most deaths were from cardiovascular disease or cancer
Adrenal lesions are found incidentally in approximately 2% to 7% of the adult population who undergo abdominal imaging, and up to a third of those have ACS in the absence of clinical signs of Cushing syndrome.
European guidelines state that a plasma cortisol level of 138 nmol/L (5 µg/dL) or greater following DST defines ACS, and a level less than 50 nmol/L (1.8 µg/dL) rules it out, while values 50-137 (1.8-5 µg/dL) are deemed “possible” ACS.
For their study, the authors retrospectively analyzed 1,048 consecutive patients with adrenal incidentalomas seen at two Swedish hospitals between 2005 and September 2015 who were followed for up to 14 years.
The patients were a median age of 64.9 years, and 58.5% were women.
At baseline, 45.1% had a cortisol level of 50 nmol/L (1.8 µg/dL) or higher following DST, 52.9% had hypertension, 18.7% had diabetes, and 20.6% had a medical history of one or more cardiovascular events. A total of 54 patients underwent adrenalectomy, eight of them more than 2 years after the DST.
Researchers found a linear increase in mortality risk with increasing cortisol values up to 200 nmol/L (7.25 µg/dL) following DST.
Over 14 years, 16.2% (170 patients) died. Compared with cortisol less than 50 nmol/L (1.8 µg/dL) following DST, adjusted hazard ratios for mortality were 2.30 and 3.04 for cortisol levels 83 to 137 nmol/L (3-5 µg/dL) and 138 nmol/L (5 µg/dL) or greater, respectively, and both were significant.
Among the patients who died, causes of death were cardiovascular disease in 38%, cancer in 30%, infection in 4%, and other diseases in 28%.
Patients with post-DST cortisol levels of 83 nmol/L (3 µg/dL) or higher had increased cardiovascular mortality, while those with levels of 50-82 nmol/L (1.8-3.0 µg/dL) did not. In contrast, mortality rates from cancer, infection, and other diseases didn’t vary across groups.
Implications: Further testing, prospective studies needed
“The increase in mortality associated with cortisol DST values of 83 nmol/L or higher has implications,” the authors say.
“We suggest [medical] treatment of known cardiovascular risk factors in these patients and incorporation of our results in the decision about which patients to recommend for adrenalectomy.”
In contrast, ACS with lower cortisol (<83 nmol/L or 3 µg/dL) following DST “is not associated with clinically relevant increased mortality within 5 to 10 years,” they observe.
Dr. Kurra said she would perform further testing for any patient with an adrenal incidentaloma and a cortisol level 50-137 nmol/L (1.8-5 µg/dL) following DST: Specifically, a dehydroepiandrosterone sulfate (DHEAS) test.
“If DHEAS is low and the patient has metabolic complications, then I will work them up more, with adrenocorticotropin (ACTH) and 24-hour urine and go down that path of looking for the extent of overproduction of cortisol.”
She recommended an algorithm published in 2017 of an age- and sex-adjusted DHEAS ratio that provides a sensitive and specific screening test for subclinical hypercortisolism in patients with adrenal incidentalomas.
In further analyses by Dr. Kjellbom and colleagues into incidentaloma size, bilateralism, basal ACTH less than 2.0 pmol/L, or DHEAS less than 1.04 mmol/L, only DHEAS significantly predicted mortality.
“This should be studied further, specific to sex, age, and [post-DST]-cortisol strata,” Dr. Kjellbom and colleagues say.
In conclusion, Dr. Kurra said the new data “confirm something that people have postulated. But because it’s a retrospective review, we need prospective studies. It is an interesting finding that needs further study before we can change clinical practice.”
The study was funded by unrestricted grants from the Lisa and Johan Grönberg Foundation and the Gyllenstiernska Krapperup Foundation. Dr. Kjellbom and Dr. Kurra have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mortality is two to three times higher in patients with adrenal incidentalomas who have autonomous cortisol secretion levels of 83 nmol/L (3 µg/dL) or more after a 1 mg dexamethasone suppression test (DST), compared with those with levels below this, new research finds.
Autonomous cortisol secretion (ACS) has been linked to hypertension, cardiovascular disease, type 2 diabetes, and early mortality, and risks vary by cortisol level.
“To adequately decide whether treatment should be surgery or medical management of possible complications, it is essential to know the risk associated with the actual level of ACS,” write Albin Kjellbom, MD, of Skåne University Hospital, Lund, Sweden, and colleagues, in their article published May 25 in Annals of Internal Medicine.
Asked to comment, Salila Kurra, MD, of the Columbia adrenal center, Columbia University, New York, told this news organization that “this idea that mild cortisol excess that doesn’t meet the threshold for overt Cushing’s can, in and of itself, cause increased morbidity and mortality is something people have been thinking about for many years now.”
“But there isn’t very clear guidance on exactly what to do in that situation, whether the incidentaloma should be removed, medically managed, or the patient should just be watched ... It may be clinically significant, but the way to sort that out is to do other testing.”
Most deaths were from cardiovascular disease or cancer
Adrenal lesions are found incidentally in approximately 2% to 7% of the adult population who undergo abdominal imaging, and up to a third of those have ACS in the absence of clinical signs of Cushing syndrome.
European guidelines state that a plasma cortisol level of 138 nmol/L (5 µg/dL) or greater following DST defines ACS, and a level less than 50 nmol/L (1.8 µg/dL) rules it out, while values 50-137 (1.8-5 µg/dL) are deemed “possible” ACS.
For their study, the authors retrospectively analyzed 1,048 consecutive patients with adrenal incidentalomas seen at two Swedish hospitals between 2005 and September 2015 who were followed for up to 14 years.
The patients were a median age of 64.9 years, and 58.5% were women.
At baseline, 45.1% had a cortisol level of 50 nmol/L (1.8 µg/dL) or higher following DST, 52.9% had hypertension, 18.7% had diabetes, and 20.6% had a medical history of one or more cardiovascular events. A total of 54 patients underwent adrenalectomy, eight of them more than 2 years after the DST.
Researchers found a linear increase in mortality risk with increasing cortisol values up to 200 nmol/L (7.25 µg/dL) following DST.
Over 14 years, 16.2% (170 patients) died. Compared with cortisol less than 50 nmol/L (1.8 µg/dL) following DST, adjusted hazard ratios for mortality were 2.30 and 3.04 for cortisol levels 83 to 137 nmol/L (3-5 µg/dL) and 138 nmol/L (5 µg/dL) or greater, respectively, and both were significant.
Among the patients who died, causes of death were cardiovascular disease in 38%, cancer in 30%, infection in 4%, and other diseases in 28%.
Patients with post-DST cortisol levels of 83 nmol/L (3 µg/dL) or higher had increased cardiovascular mortality, while those with levels of 50-82 nmol/L (1.8-3.0 µg/dL) did not. In contrast, mortality rates from cancer, infection, and other diseases didn’t vary across groups.
Implications: Further testing, prospective studies needed
“The increase in mortality associated with cortisol DST values of 83 nmol/L or higher has implications,” the authors say.
“We suggest [medical] treatment of known cardiovascular risk factors in these patients and incorporation of our results in the decision about which patients to recommend for adrenalectomy.”
In contrast, ACS with lower cortisol (<83 nmol/L or 3 µg/dL) following DST “is not associated with clinically relevant increased mortality within 5 to 10 years,” they observe.
Dr. Kurra said she would perform further testing for any patient with an adrenal incidentaloma and a cortisol level 50-137 nmol/L (1.8-5 µg/dL) following DST: Specifically, a dehydroepiandrosterone sulfate (DHEAS) test.
“If DHEAS is low and the patient has metabolic complications, then I will work them up more, with adrenocorticotropin (ACTH) and 24-hour urine and go down that path of looking for the extent of overproduction of cortisol.”
She recommended an algorithm published in 2017 of an age- and sex-adjusted DHEAS ratio that provides a sensitive and specific screening test for subclinical hypercortisolism in patients with adrenal incidentalomas.
In further analyses by Dr. Kjellbom and colleagues into incidentaloma size, bilateralism, basal ACTH less than 2.0 pmol/L, or DHEAS less than 1.04 mmol/L, only DHEAS significantly predicted mortality.
“This should be studied further, specific to sex, age, and [post-DST]-cortisol strata,” Dr. Kjellbom and colleagues say.
In conclusion, Dr. Kurra said the new data “confirm something that people have postulated. But because it’s a retrospective review, we need prospective studies. It is an interesting finding that needs further study before we can change clinical practice.”
The study was funded by unrestricted grants from the Lisa and Johan Grönberg Foundation and the Gyllenstiernska Krapperup Foundation. Dr. Kjellbom and Dr. Kurra have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mortality is two to three times higher in patients with adrenal incidentalomas who have autonomous cortisol secretion levels of 83 nmol/L (3 µg/dL) or more after a 1 mg dexamethasone suppression test (DST), compared with those with levels below this, new research finds.
Autonomous cortisol secretion (ACS) has been linked to hypertension, cardiovascular disease, type 2 diabetes, and early mortality, and risks vary by cortisol level.
“To adequately decide whether treatment should be surgery or medical management of possible complications, it is essential to know the risk associated with the actual level of ACS,” write Albin Kjellbom, MD, of Skåne University Hospital, Lund, Sweden, and colleagues, in their article published May 25 in Annals of Internal Medicine.
Asked to comment, Salila Kurra, MD, of the Columbia adrenal center, Columbia University, New York, told this news organization that “this idea that mild cortisol excess that doesn’t meet the threshold for overt Cushing’s can, in and of itself, cause increased morbidity and mortality is something people have been thinking about for many years now.”
“But there isn’t very clear guidance on exactly what to do in that situation, whether the incidentaloma should be removed, medically managed, or the patient should just be watched ... It may be clinically significant, but the way to sort that out is to do other testing.”
Most deaths were from cardiovascular disease or cancer
Adrenal lesions are found incidentally in approximately 2% to 7% of the adult population who undergo abdominal imaging, and up to a third of those have ACS in the absence of clinical signs of Cushing syndrome.
European guidelines state that a plasma cortisol level of 138 nmol/L (5 µg/dL) or greater following DST defines ACS, and a level less than 50 nmol/L (1.8 µg/dL) rules it out, while values 50-137 (1.8-5 µg/dL) are deemed “possible” ACS.
For their study, the authors retrospectively analyzed 1,048 consecutive patients with adrenal incidentalomas seen at two Swedish hospitals between 2005 and September 2015 who were followed for up to 14 years.
The patients were a median age of 64.9 years, and 58.5% were women.
At baseline, 45.1% had a cortisol level of 50 nmol/L (1.8 µg/dL) or higher following DST, 52.9% had hypertension, 18.7% had diabetes, and 20.6% had a medical history of one or more cardiovascular events. A total of 54 patients underwent adrenalectomy, eight of them more than 2 years after the DST.
Researchers found a linear increase in mortality risk with increasing cortisol values up to 200 nmol/L (7.25 µg/dL) following DST.
Over 14 years, 16.2% (170 patients) died. Compared with cortisol less than 50 nmol/L (1.8 µg/dL) following DST, adjusted hazard ratios for mortality were 2.30 and 3.04 for cortisol levels 83 to 137 nmol/L (3-5 µg/dL) and 138 nmol/L (5 µg/dL) or greater, respectively, and both were significant.
Among the patients who died, causes of death were cardiovascular disease in 38%, cancer in 30%, infection in 4%, and other diseases in 28%.
Patients with post-DST cortisol levels of 83 nmol/L (3 µg/dL) or higher had increased cardiovascular mortality, while those with levels of 50-82 nmol/L (1.8-3.0 µg/dL) did not. In contrast, mortality rates from cancer, infection, and other diseases didn’t vary across groups.
Implications: Further testing, prospective studies needed
“The increase in mortality associated with cortisol DST values of 83 nmol/L or higher has implications,” the authors say.
“We suggest [medical] treatment of known cardiovascular risk factors in these patients and incorporation of our results in the decision about which patients to recommend for adrenalectomy.”
In contrast, ACS with lower cortisol (<83 nmol/L or 3 µg/dL) following DST “is not associated with clinically relevant increased mortality within 5 to 10 years,” they observe.
Dr. Kurra said she would perform further testing for any patient with an adrenal incidentaloma and a cortisol level 50-137 nmol/L (1.8-5 µg/dL) following DST: Specifically, a dehydroepiandrosterone sulfate (DHEAS) test.
“If DHEAS is low and the patient has metabolic complications, then I will work them up more, with adrenocorticotropin (ACTH) and 24-hour urine and go down that path of looking for the extent of overproduction of cortisol.”
She recommended an algorithm published in 2017 of an age- and sex-adjusted DHEAS ratio that provides a sensitive and specific screening test for subclinical hypercortisolism in patients with adrenal incidentalomas.
In further analyses by Dr. Kjellbom and colleagues into incidentaloma size, bilateralism, basal ACTH less than 2.0 pmol/L, or DHEAS less than 1.04 mmol/L, only DHEAS significantly predicted mortality.
“This should be studied further, specific to sex, age, and [post-DST]-cortisol strata,” Dr. Kjellbom and colleagues say.
In conclusion, Dr. Kurra said the new data “confirm something that people have postulated. But because it’s a retrospective review, we need prospective studies. It is an interesting finding that needs further study before we can change clinical practice.”
The study was funded by unrestricted grants from the Lisa and Johan Grönberg Foundation and the Gyllenstiernska Krapperup Foundation. Dr. Kjellbom and Dr. Kurra have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.