User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Inexperience Diagnosing Syphilis Adding to Higher Rates
With rates of syphilis rising quickly in the United States and elsewhere, clinicians are having to up their game when it comes to diagnosing and treating an infection that they may not be paying enough attention to.
More than 200,000 cases of syphilis were reported in the United States in 2022, which is the highest number since 1950 and is a 17.3% increase over 2021, according to the latest figures from the Centers for Disease Control and Prevention (CDC). The rate of infection has increased almost every year since a historic low in 2001.
And the trend is not limited to the United States. Last year, the infection rate in the United Kingdom hit a 50-year high, said David Mabey, BCh, DM, from the London School of Hygiene and Tropical Medicine. Syphilis and other sexually transmitted infections are also a major problem in low- and middle-income countries, he added, although good data are not always available.
Many of today’s healthcare professionals have little experience with the disease, shared Ina Park, MD, a sexually transmitted infections specialist at the University of California at San Francisco. “An entire generation of physicians — including myself — did not see any cases until we were well out of our training,” Dr. Park reported. “We’re really playing catch-up.”
A Centuries-Old Ailment
Dr. Park offered some advice on the challenges of diagnosing what can be an elusive infection at the Conference on Retroviruses and Opportunistic Infections (CROI) 2024 Annual Meeting in Denver. That advice boiled down to one simple rule: “Test, test, test.”
Because syphilis can mimic so many other conditions and can have long periods of latency, it can be easily missed or even misdiagnosed by experienced physicians, said Dr. Park. Clinicians need to keep it front of mind and have a lower threshold for testing, even if there are no obvious symptoms.
Following the CDC’s new recommendations for syphilis screening will help, she noted; every sexually active patient aged between 15 and 44 years who lives in a county with a syphilis infection rate of 4.6 per 100,000 people or higher should get the test. And clinicians should remain vigilant, even in areas with a lower prevalence. “If you can’t account for new symptoms in a sexually active patient, order a test,” said Dr. Park.
Complicated Cases
The lack of experience with syphilis affects not just diagnosis but also treatment, particularly for complex cases, said Khalil Ghanem, MD, PhD, from the Johns Hopkins University School of Medicine in Baltimore. “When you don’t have to deal with something for a while, you forget how to deal with it,” he added.
At CROI, Dr. Ghanem offered suggestions for how to navigate complicated cases of ocular syphilis, otic syphilis, and neurosyphilis, and how to interpret test results when a patient’s antigen titers are being “unruly.”
With potential ocular or otic syphilis, you shouldn’t wait for a specialist like an ophthalmologist to weigh in but instead refer the patient directly to the emergency department because of the risk that the symptoms may become irreversible and result in permanent blindness or deafness. “You don’t want to dilly-dally with those conditions,” Dr. Ghanem said.
Closely monitoring a patient’s rapid plasma regain and venereal disease research laboratory antigen levels is the only way to manage syphilis and to determine whether the infection is responding to treatment, he noted, but sometimes those titers “don’t do what you think they should be doing” and fail to decline or even go up after treatment.
“You don’t know if they went up because the patient was re-infected, or they developed neurosyphilis, or there was a problem at the lab,” he said. “It can be challenging to interpret.”
To decipher confusing test results, Dr. Ghanem recommended getting a detailed history to understand whether a patient is at risk for reinfection, whether there are signs of neurosyphilis or other complications, whether pregnancy is possible, and so on. “Based on the answers, you can determine what the most rational approach to treatment would be,” he shared.
Drug Shortages
Efforts to get the infection under control have become more complicated. Last summer, Pfizer announced that it had run out of penicillin G benzathine (Bicillin), an injectable, long-acting drug that is one of the main treatments for syphilis and the only one that can be given to pregnant people. Supplies for children ran out at the end of June 2023, and supplies for adults were gone by the end of September.
Because Pfizer is the only company that manufactures penicillin G benzathine, there is no one to pick up the slack in the short-term, so the shortage is expected to continue until at least the middle of 2024.
In response, the US Food and Drug Administration has temporarily allowed the use of benzylpenicillin benzathine (Extencilline), a French formulation that has not been approved in the United States, until supplies of penicillin G benzathine are stabilized.
The shortage has shone a spotlight on the important issue of a lack of alternatives for the treatment of syphilis during pregnancy, which increases the risk for congenital syphilis. “Hopefully, this pushes the National Institutes of Health and others to step up their game on studies for alternative drugs for use in pregnancy,” Dr. Ghanem said.
A version of this article appeared on Medscape.com.
With rates of syphilis rising quickly in the United States and elsewhere, clinicians are having to up their game when it comes to diagnosing and treating an infection that they may not be paying enough attention to.
More than 200,000 cases of syphilis were reported in the United States in 2022, which is the highest number since 1950 and is a 17.3% increase over 2021, according to the latest figures from the Centers for Disease Control and Prevention (CDC). The rate of infection has increased almost every year since a historic low in 2001.
And the trend is not limited to the United States. Last year, the infection rate in the United Kingdom hit a 50-year high, said David Mabey, BCh, DM, from the London School of Hygiene and Tropical Medicine. Syphilis and other sexually transmitted infections are also a major problem in low- and middle-income countries, he added, although good data are not always available.
Many of today’s healthcare professionals have little experience with the disease, shared Ina Park, MD, a sexually transmitted infections specialist at the University of California at San Francisco. “An entire generation of physicians — including myself — did not see any cases until we were well out of our training,” Dr. Park reported. “We’re really playing catch-up.”
A Centuries-Old Ailment
Dr. Park offered some advice on the challenges of diagnosing what can be an elusive infection at the Conference on Retroviruses and Opportunistic Infections (CROI) 2024 Annual Meeting in Denver. That advice boiled down to one simple rule: “Test, test, test.”
Because syphilis can mimic so many other conditions and can have long periods of latency, it can be easily missed or even misdiagnosed by experienced physicians, said Dr. Park. Clinicians need to keep it front of mind and have a lower threshold for testing, even if there are no obvious symptoms.
Following the CDC’s new recommendations for syphilis screening will help, she noted; every sexually active patient aged between 15 and 44 years who lives in a county with a syphilis infection rate of 4.6 per 100,000 people or higher should get the test. And clinicians should remain vigilant, even in areas with a lower prevalence. “If you can’t account for new symptoms in a sexually active patient, order a test,” said Dr. Park.
Complicated Cases
The lack of experience with syphilis affects not just diagnosis but also treatment, particularly for complex cases, said Khalil Ghanem, MD, PhD, from the Johns Hopkins University School of Medicine in Baltimore. “When you don’t have to deal with something for a while, you forget how to deal with it,” he added.
At CROI, Dr. Ghanem offered suggestions for how to navigate complicated cases of ocular syphilis, otic syphilis, and neurosyphilis, and how to interpret test results when a patient’s antigen titers are being “unruly.”
With potential ocular or otic syphilis, you shouldn’t wait for a specialist like an ophthalmologist to weigh in but instead refer the patient directly to the emergency department because of the risk that the symptoms may become irreversible and result in permanent blindness or deafness. “You don’t want to dilly-dally with those conditions,” Dr. Ghanem said.
Closely monitoring a patient’s rapid plasma regain and venereal disease research laboratory antigen levels is the only way to manage syphilis and to determine whether the infection is responding to treatment, he noted, but sometimes those titers “don’t do what you think they should be doing” and fail to decline or even go up after treatment.
“You don’t know if they went up because the patient was re-infected, or they developed neurosyphilis, or there was a problem at the lab,” he said. “It can be challenging to interpret.”
To decipher confusing test results, Dr. Ghanem recommended getting a detailed history to understand whether a patient is at risk for reinfection, whether there are signs of neurosyphilis or other complications, whether pregnancy is possible, and so on. “Based on the answers, you can determine what the most rational approach to treatment would be,” he shared.
Drug Shortages
Efforts to get the infection under control have become more complicated. Last summer, Pfizer announced that it had run out of penicillin G benzathine (Bicillin), an injectable, long-acting drug that is one of the main treatments for syphilis and the only one that can be given to pregnant people. Supplies for children ran out at the end of June 2023, and supplies for adults were gone by the end of September.
Because Pfizer is the only company that manufactures penicillin G benzathine, there is no one to pick up the slack in the short-term, so the shortage is expected to continue until at least the middle of 2024.
In response, the US Food and Drug Administration has temporarily allowed the use of benzylpenicillin benzathine (Extencilline), a French formulation that has not been approved in the United States, until supplies of penicillin G benzathine are stabilized.
The shortage has shone a spotlight on the important issue of a lack of alternatives for the treatment of syphilis during pregnancy, which increases the risk for congenital syphilis. “Hopefully, this pushes the National Institutes of Health and others to step up their game on studies for alternative drugs for use in pregnancy,” Dr. Ghanem said.
A version of this article appeared on Medscape.com.
With rates of syphilis rising quickly in the United States and elsewhere, clinicians are having to up their game when it comes to diagnosing and treating an infection that they may not be paying enough attention to.
More than 200,000 cases of syphilis were reported in the United States in 2022, which is the highest number since 1950 and is a 17.3% increase over 2021, according to the latest figures from the Centers for Disease Control and Prevention (CDC). The rate of infection has increased almost every year since a historic low in 2001.
And the trend is not limited to the United States. Last year, the infection rate in the United Kingdom hit a 50-year high, said David Mabey, BCh, DM, from the London School of Hygiene and Tropical Medicine. Syphilis and other sexually transmitted infections are also a major problem in low- and middle-income countries, he added, although good data are not always available.
Many of today’s healthcare professionals have little experience with the disease, shared Ina Park, MD, a sexually transmitted infections specialist at the University of California at San Francisco. “An entire generation of physicians — including myself — did not see any cases until we were well out of our training,” Dr. Park reported. “We’re really playing catch-up.”
A Centuries-Old Ailment
Dr. Park offered some advice on the challenges of diagnosing what can be an elusive infection at the Conference on Retroviruses and Opportunistic Infections (CROI) 2024 Annual Meeting in Denver. That advice boiled down to one simple rule: “Test, test, test.”
Because syphilis can mimic so many other conditions and can have long periods of latency, it can be easily missed or even misdiagnosed by experienced physicians, said Dr. Park. Clinicians need to keep it front of mind and have a lower threshold for testing, even if there are no obvious symptoms.
Following the CDC’s new recommendations for syphilis screening will help, she noted; every sexually active patient aged between 15 and 44 years who lives in a county with a syphilis infection rate of 4.6 per 100,000 people or higher should get the test. And clinicians should remain vigilant, even in areas with a lower prevalence. “If you can’t account for new symptoms in a sexually active patient, order a test,” said Dr. Park.
Complicated Cases
The lack of experience with syphilis affects not just diagnosis but also treatment, particularly for complex cases, said Khalil Ghanem, MD, PhD, from the Johns Hopkins University School of Medicine in Baltimore. “When you don’t have to deal with something for a while, you forget how to deal with it,” he added.
At CROI, Dr. Ghanem offered suggestions for how to navigate complicated cases of ocular syphilis, otic syphilis, and neurosyphilis, and how to interpret test results when a patient’s antigen titers are being “unruly.”
With potential ocular or otic syphilis, you shouldn’t wait for a specialist like an ophthalmologist to weigh in but instead refer the patient directly to the emergency department because of the risk that the symptoms may become irreversible and result in permanent blindness or deafness. “You don’t want to dilly-dally with those conditions,” Dr. Ghanem said.
Closely monitoring a patient’s rapid plasma regain and venereal disease research laboratory antigen levels is the only way to manage syphilis and to determine whether the infection is responding to treatment, he noted, but sometimes those titers “don’t do what you think they should be doing” and fail to decline or even go up after treatment.
“You don’t know if they went up because the patient was re-infected, or they developed neurosyphilis, or there was a problem at the lab,” he said. “It can be challenging to interpret.”
To decipher confusing test results, Dr. Ghanem recommended getting a detailed history to understand whether a patient is at risk for reinfection, whether there are signs of neurosyphilis or other complications, whether pregnancy is possible, and so on. “Based on the answers, you can determine what the most rational approach to treatment would be,” he shared.
Drug Shortages
Efforts to get the infection under control have become more complicated. Last summer, Pfizer announced that it had run out of penicillin G benzathine (Bicillin), an injectable, long-acting drug that is one of the main treatments for syphilis and the only one that can be given to pregnant people. Supplies for children ran out at the end of June 2023, and supplies for adults were gone by the end of September.
Because Pfizer is the only company that manufactures penicillin G benzathine, there is no one to pick up the slack in the short-term, so the shortage is expected to continue until at least the middle of 2024.
In response, the US Food and Drug Administration has temporarily allowed the use of benzylpenicillin benzathine (Extencilline), a French formulation that has not been approved in the United States, until supplies of penicillin G benzathine are stabilized.
The shortage has shone a spotlight on the important issue of a lack of alternatives for the treatment of syphilis during pregnancy, which increases the risk for congenital syphilis. “Hopefully, this pushes the National Institutes of Health and others to step up their game on studies for alternative drugs for use in pregnancy,” Dr. Ghanem said.
A version of this article appeared on Medscape.com.
Multimodal Treatment Found Effective for Overactive Bladder
TOPLINE:
A new study published in JAMA Network Open showed that an intervention including cognitive behavioral therapy improved the quality of life for women with overactive bladder (OAB).
METHODOLOGY:
- A total of 79 women with moderate to severe OAB were randomized to the control group or the intervention, which was composed of four 30-minute sessions using strategies including cognitive behavioral therapy (CBT).
- The first and second sessions provided education on OAB and CBT, lifestyle modifications such as limiting coffee intake, pelvic floor muscle training, and introduced exposure training.
- The third and fourth sessions continued exposure and pelvic floor muscle training and education on relapse prevention.
- Researchers assessed outcomes using the health-related quality of life (HRQOL), in which participants answered questions regarding their degree of distress, emotions, and physical and social limitations related to OAB symptoms.
TAKEAWAY:
- Participants who received the intervention on average improved in their HRQOL score by 12.6 points higher than those in the control group (usual care) from baseline to week 13 (between-group difference estimate, 12.6 [95% CI, 6.6-18.6] points; P < .001).
- The average age of participants was 63.5 years, and more than 87% of women in each group had moderate OAB.
- Patient-reported improvement and satisfaction scores were also more improved in the intervention group than in the control group; most participants in both groups had no change in the pharmacotherapy during the trial.
IN PRACTICE:
Urologists and other primary care clinicians who treat women with OAB may consider a multicomponent intervention that includes cognitive components and exposure-based bladder training or could refer to a cognitive behavioral therapist or pelvic floor physical therapist experienced in these techniques.
SOURCE:
Satoshi Funada, MD, PhD, and Takashi Kobayashi, MD, PhD, both with the Department of Urology at Kyoto University Graduate School of Medicine in Kyoto, Japan, are the corresponding authors. The study was published online in JAMA Network Open.
LIMITATIONS:
The trial was open label, and the use of a waiting list control group is known to produce greater differences between the two groups. The trial included patients both taking and not taking medication for OAB. The sample size was also relatively small, and the intervention was performed by a single clinician, possibly limiting the generalizability of results.
DISCLOSURES:
The study was funded by the Japan Society for the Promotion of Science (JSPS). Various study authors reported receiving grants from the Pfizer Health Research Foundation, AstraZeneca, and JSPS. Other study authors reported receiving personal fees from Eisai, Sawai Pharmaceutical, Statcom, and others. One author reported pending patents for intellectual properties for the Kokoro app licensed to Mitsubishi Tanabe Pharma.
A version of this article appeared on Medscape.com.
TOPLINE:
A new study published in JAMA Network Open showed that an intervention including cognitive behavioral therapy improved the quality of life for women with overactive bladder (OAB).
METHODOLOGY:
- A total of 79 women with moderate to severe OAB were randomized to the control group or the intervention, which was composed of four 30-minute sessions using strategies including cognitive behavioral therapy (CBT).
- The first and second sessions provided education on OAB and CBT, lifestyle modifications such as limiting coffee intake, pelvic floor muscle training, and introduced exposure training.
- The third and fourth sessions continued exposure and pelvic floor muscle training and education on relapse prevention.
- Researchers assessed outcomes using the health-related quality of life (HRQOL), in which participants answered questions regarding their degree of distress, emotions, and physical and social limitations related to OAB symptoms.
TAKEAWAY:
- Participants who received the intervention on average improved in their HRQOL score by 12.6 points higher than those in the control group (usual care) from baseline to week 13 (between-group difference estimate, 12.6 [95% CI, 6.6-18.6] points; P < .001).
- The average age of participants was 63.5 years, and more than 87% of women in each group had moderate OAB.
- Patient-reported improvement and satisfaction scores were also more improved in the intervention group than in the control group; most participants in both groups had no change in the pharmacotherapy during the trial.
IN PRACTICE:
Urologists and other primary care clinicians who treat women with OAB may consider a multicomponent intervention that includes cognitive components and exposure-based bladder training or could refer to a cognitive behavioral therapist or pelvic floor physical therapist experienced in these techniques.
SOURCE:
Satoshi Funada, MD, PhD, and Takashi Kobayashi, MD, PhD, both with the Department of Urology at Kyoto University Graduate School of Medicine in Kyoto, Japan, are the corresponding authors. The study was published online in JAMA Network Open.
LIMITATIONS:
The trial was open label, and the use of a waiting list control group is known to produce greater differences between the two groups. The trial included patients both taking and not taking medication for OAB. The sample size was also relatively small, and the intervention was performed by a single clinician, possibly limiting the generalizability of results.
DISCLOSURES:
The study was funded by the Japan Society for the Promotion of Science (JSPS). Various study authors reported receiving grants from the Pfizer Health Research Foundation, AstraZeneca, and JSPS. Other study authors reported receiving personal fees from Eisai, Sawai Pharmaceutical, Statcom, and others. One author reported pending patents for intellectual properties for the Kokoro app licensed to Mitsubishi Tanabe Pharma.
A version of this article appeared on Medscape.com.
TOPLINE:
A new study published in JAMA Network Open showed that an intervention including cognitive behavioral therapy improved the quality of life for women with overactive bladder (OAB).
METHODOLOGY:
- A total of 79 women with moderate to severe OAB were randomized to the control group or the intervention, which was composed of four 30-minute sessions using strategies including cognitive behavioral therapy (CBT).
- The first and second sessions provided education on OAB and CBT, lifestyle modifications such as limiting coffee intake, pelvic floor muscle training, and introduced exposure training.
- The third and fourth sessions continued exposure and pelvic floor muscle training and education on relapse prevention.
- Researchers assessed outcomes using the health-related quality of life (HRQOL), in which participants answered questions regarding their degree of distress, emotions, and physical and social limitations related to OAB symptoms.
TAKEAWAY:
- Participants who received the intervention on average improved in their HRQOL score by 12.6 points higher than those in the control group (usual care) from baseline to week 13 (between-group difference estimate, 12.6 [95% CI, 6.6-18.6] points; P < .001).
- The average age of participants was 63.5 years, and more than 87% of women in each group had moderate OAB.
- Patient-reported improvement and satisfaction scores were also more improved in the intervention group than in the control group; most participants in both groups had no change in the pharmacotherapy during the trial.
IN PRACTICE:
Urologists and other primary care clinicians who treat women with OAB may consider a multicomponent intervention that includes cognitive components and exposure-based bladder training or could refer to a cognitive behavioral therapist or pelvic floor physical therapist experienced in these techniques.
SOURCE:
Satoshi Funada, MD, PhD, and Takashi Kobayashi, MD, PhD, both with the Department of Urology at Kyoto University Graduate School of Medicine in Kyoto, Japan, are the corresponding authors. The study was published online in JAMA Network Open.
LIMITATIONS:
The trial was open label, and the use of a waiting list control group is known to produce greater differences between the two groups. The trial included patients both taking and not taking medication for OAB. The sample size was also relatively small, and the intervention was performed by a single clinician, possibly limiting the generalizability of results.
DISCLOSURES:
The study was funded by the Japan Society for the Promotion of Science (JSPS). Various study authors reported receiving grants from the Pfizer Health Research Foundation, AstraZeneca, and JSPS. Other study authors reported receiving personal fees from Eisai, Sawai Pharmaceutical, Statcom, and others. One author reported pending patents for intellectual properties for the Kokoro app licensed to Mitsubishi Tanabe Pharma.
A version of this article appeared on Medscape.com.
Risk for Preterm Birth Stops Maternal RSV Vaccine Trial
A phase 3 trial of a maternal vaccine candidate for respiratory syncytial virus (RSV) has been stopped early because the risk for preterm births is higher in the candidate vaccine group than in the placebo group.
By the time enrollment was stopped on February 25, 2022 because of the safety signal of preterm birth, 5328 pregnant women had been vaccinated, about half of the intended 10,000 enrollees. Of these, 3557 received the candidate vaccine RSV prefusion F protein–based maternal vaccine, and another 1771 received a placebo.
Data from the trial, sponsored by GSK, were immediately made available when recruitment and vaccination were stopped, and investigation of the preterm birth risk followed. Results of that analysis, led by Ilse Dieussaert, IR, vice president for vaccine development at GSK in Wavre, Belgium, are published online on March 13 in The New England Journal of Medicine.
“We have discontinued our work on this RSV maternal candidate vaccine, and we are closing out all ongoing trials with the exception of the MAT-015 follow-on study to monitor subsequent pregnancies,” a GSK spokesperson said in an interview.
The trial was conducted in pregnant women aged 18-49 years to assess the efficacy and safety of the vaccine. The women were randomly assigned 2:1 to receive the candidate vaccine or placebo between 24 and 34 weeks’ gestation.
Preterm Births
The primary outcomes were any or severe medically assessed RSV-associated lower respiratory tract infection in infants from birth to 6 months and safety in infants from birth to 12 months.
According to the data, preterm birth occurred in 6.8% of the infants in the vaccine group and in 4.9% of those in the placebo group (relative risk [RR], 1.37; 95% CI, 1.08-1.74; P = .01). Neonatal death occurred in 0.4% in the vaccine group and 0.2% in the placebo group (RR, 2.16; 95% CI, 0.62-7.56; P = .23).
To date, only one RSV vaccine (Abrysvo, Pfizer) has been approved for use during pregnancy to protect infants from RSV-associated lower respiratory tract infection.
“It was a very big deal that this trial was stopped, and the new candidate won’t get approval.” said Aaron E. Glatt, MD, chair of the Department of Medicine and chief of Infectious Diseases and Hospital Epidemiologist at Mount Sinai South Nassau in Oceanside, New York.
Only One RSV Vaccine Approved in Pregnancy
Dr. Glatt pointed out the GSK vaccine is like the maternal vaccine that did get approved. “The data clearly show that there was a slight but increased risk in preterm labor,” Dr. Glatt said, “and while not as clearly shown, there was an increase in neonatal death in the group of very small numbers, but any neonatal death is of concern.”
The implications were disturbing, he added, “You’re giving this vaccine to prevent neonatal death.” Though the Pfizer vaccine that was granted approval had a very slight increase in premature birth, the risk wasn’t statistically significant, he pointed out, “and it showed similar benefits in preventing neonatal illness, which can be fatal.”
Dr. Glatt said that there is still a lingering concern with the approved vaccine, and he explained that most clinicians will give it closer to the end of the recommended time window of 34 weeks. “This way, even if there is a slight increase in premature term labor, you’re probably not going to have a serious outcome because the baby will be far enough along.”
A difference in the incidence of preterm birth between the experimental vaccine and placebo groups was predominantly found in low- and middle-income countries, according to Dieussaert’s team, “where approximately 50% of the trial population was enrolled and where the medical need for maternal RSV vaccines is the greatest.”
The RR was 1.56 (95% CI, 1.17-2.10) for low- and middle-income countries and 1.04 (95% CI, 0.68-1.58) for high-income countries.
“If a smaller percentage of participants from low- and middle-income countries had been enrolled in our trial, the RR for preterm birth in the vaccine group as compared with the placebo group might have been reduced in the overall trial population,” they reported.
The authors explained that the data do not reveal the cause of the higher risk for preterm birth in the vaccine group.
“We do not know what caused the signal,” the company’s spokesperson added. “GSK completed all the necessary steps of product development including preclinical toxicology studies and clinical studies in nonpregnant women prior to starting the studies in pregnant women. There were no safety signals identified in any of the earlier parts of the clinical testing. There have been no safety signals identified in the other phase 3 trials for this vaccine candidate.”
Researchers did not find a correlation between preterm births in the treatment vs control groups with gestational age at the time of vaccination or with particular vaccine clinical trial material lots, race, ethnicity, maternal smoking, alcohol consumption, body mass index, or time between study vaccination and delivery, the GSK spokesperson said.
The spokesperson noted that the halted vaccine is different from GSK’s currently approved adjuvanted RSV vaccine (Arexvy) for adults aged 60 years or older.
What’s Next for Other Vaccines
Maternal vaccines have been effective in preventing other diseases in infants, such as tetanus, influenza, and pertussis, but RSV is a very hard virus to make a vaccine for, Dr. Glatt shared.
The need is great to have more than one option for a maternal RSV vaccine, he added, to address any potential supply concerns.
“People have to realize how serious RSV can be in infants,” he said. “It can be a fatal disease. This can be a serious illness even in healthy children.”
A version of this article appeared on Medscape.com.
A phase 3 trial of a maternal vaccine candidate for respiratory syncytial virus (RSV) has been stopped early because the risk for preterm births is higher in the candidate vaccine group than in the placebo group.
By the time enrollment was stopped on February 25, 2022 because of the safety signal of preterm birth, 5328 pregnant women had been vaccinated, about half of the intended 10,000 enrollees. Of these, 3557 received the candidate vaccine RSV prefusion F protein–based maternal vaccine, and another 1771 received a placebo.
Data from the trial, sponsored by GSK, were immediately made available when recruitment and vaccination were stopped, and investigation of the preterm birth risk followed. Results of that analysis, led by Ilse Dieussaert, IR, vice president for vaccine development at GSK in Wavre, Belgium, are published online on March 13 in The New England Journal of Medicine.
“We have discontinued our work on this RSV maternal candidate vaccine, and we are closing out all ongoing trials with the exception of the MAT-015 follow-on study to monitor subsequent pregnancies,” a GSK spokesperson said in an interview.
The trial was conducted in pregnant women aged 18-49 years to assess the efficacy and safety of the vaccine. The women were randomly assigned 2:1 to receive the candidate vaccine or placebo between 24 and 34 weeks’ gestation.
Preterm Births
The primary outcomes were any or severe medically assessed RSV-associated lower respiratory tract infection in infants from birth to 6 months and safety in infants from birth to 12 months.
According to the data, preterm birth occurred in 6.8% of the infants in the vaccine group and in 4.9% of those in the placebo group (relative risk [RR], 1.37; 95% CI, 1.08-1.74; P = .01). Neonatal death occurred in 0.4% in the vaccine group and 0.2% in the placebo group (RR, 2.16; 95% CI, 0.62-7.56; P = .23).
To date, only one RSV vaccine (Abrysvo, Pfizer) has been approved for use during pregnancy to protect infants from RSV-associated lower respiratory tract infection.
“It was a very big deal that this trial was stopped, and the new candidate won’t get approval.” said Aaron E. Glatt, MD, chair of the Department of Medicine and chief of Infectious Diseases and Hospital Epidemiologist at Mount Sinai South Nassau in Oceanside, New York.
Only One RSV Vaccine Approved in Pregnancy
Dr. Glatt pointed out the GSK vaccine is like the maternal vaccine that did get approved. “The data clearly show that there was a slight but increased risk in preterm labor,” Dr. Glatt said, “and while not as clearly shown, there was an increase in neonatal death in the group of very small numbers, but any neonatal death is of concern.”
The implications were disturbing, he added, “You’re giving this vaccine to prevent neonatal death.” Though the Pfizer vaccine that was granted approval had a very slight increase in premature birth, the risk wasn’t statistically significant, he pointed out, “and it showed similar benefits in preventing neonatal illness, which can be fatal.”
Dr. Glatt said that there is still a lingering concern with the approved vaccine, and he explained that most clinicians will give it closer to the end of the recommended time window of 34 weeks. “This way, even if there is a slight increase in premature term labor, you’re probably not going to have a serious outcome because the baby will be far enough along.”
A difference in the incidence of preterm birth between the experimental vaccine and placebo groups was predominantly found in low- and middle-income countries, according to Dieussaert’s team, “where approximately 50% of the trial population was enrolled and where the medical need for maternal RSV vaccines is the greatest.”
The RR was 1.56 (95% CI, 1.17-2.10) for low- and middle-income countries and 1.04 (95% CI, 0.68-1.58) for high-income countries.
“If a smaller percentage of participants from low- and middle-income countries had been enrolled in our trial, the RR for preterm birth in the vaccine group as compared with the placebo group might have been reduced in the overall trial population,” they reported.
The authors explained that the data do not reveal the cause of the higher risk for preterm birth in the vaccine group.
“We do not know what caused the signal,” the company’s spokesperson added. “GSK completed all the necessary steps of product development including preclinical toxicology studies and clinical studies in nonpregnant women prior to starting the studies in pregnant women. There were no safety signals identified in any of the earlier parts of the clinical testing. There have been no safety signals identified in the other phase 3 trials for this vaccine candidate.”
Researchers did not find a correlation between preterm births in the treatment vs control groups with gestational age at the time of vaccination or with particular vaccine clinical trial material lots, race, ethnicity, maternal smoking, alcohol consumption, body mass index, or time between study vaccination and delivery, the GSK spokesperson said.
The spokesperson noted that the halted vaccine is different from GSK’s currently approved adjuvanted RSV vaccine (Arexvy) for adults aged 60 years or older.
What’s Next for Other Vaccines
Maternal vaccines have been effective in preventing other diseases in infants, such as tetanus, influenza, and pertussis, but RSV is a very hard virus to make a vaccine for, Dr. Glatt shared.
The need is great to have more than one option for a maternal RSV vaccine, he added, to address any potential supply concerns.
“People have to realize how serious RSV can be in infants,” he said. “It can be a fatal disease. This can be a serious illness even in healthy children.”
A version of this article appeared on Medscape.com.
A phase 3 trial of a maternal vaccine candidate for respiratory syncytial virus (RSV) has been stopped early because the risk for preterm births is higher in the candidate vaccine group than in the placebo group.
By the time enrollment was stopped on February 25, 2022 because of the safety signal of preterm birth, 5328 pregnant women had been vaccinated, about half of the intended 10,000 enrollees. Of these, 3557 received the candidate vaccine RSV prefusion F protein–based maternal vaccine, and another 1771 received a placebo.
Data from the trial, sponsored by GSK, were immediately made available when recruitment and vaccination were stopped, and investigation of the preterm birth risk followed. Results of that analysis, led by Ilse Dieussaert, IR, vice president for vaccine development at GSK in Wavre, Belgium, are published online on March 13 in The New England Journal of Medicine.
“We have discontinued our work on this RSV maternal candidate vaccine, and we are closing out all ongoing trials with the exception of the MAT-015 follow-on study to monitor subsequent pregnancies,” a GSK spokesperson said in an interview.
The trial was conducted in pregnant women aged 18-49 years to assess the efficacy and safety of the vaccine. The women were randomly assigned 2:1 to receive the candidate vaccine or placebo between 24 and 34 weeks’ gestation.
Preterm Births
The primary outcomes were any or severe medically assessed RSV-associated lower respiratory tract infection in infants from birth to 6 months and safety in infants from birth to 12 months.
According to the data, preterm birth occurred in 6.8% of the infants in the vaccine group and in 4.9% of those in the placebo group (relative risk [RR], 1.37; 95% CI, 1.08-1.74; P = .01). Neonatal death occurred in 0.4% in the vaccine group and 0.2% in the placebo group (RR, 2.16; 95% CI, 0.62-7.56; P = .23).
To date, only one RSV vaccine (Abrysvo, Pfizer) has been approved for use during pregnancy to protect infants from RSV-associated lower respiratory tract infection.
“It was a very big deal that this trial was stopped, and the new candidate won’t get approval.” said Aaron E. Glatt, MD, chair of the Department of Medicine and chief of Infectious Diseases and Hospital Epidemiologist at Mount Sinai South Nassau in Oceanside, New York.
Only One RSV Vaccine Approved in Pregnancy
Dr. Glatt pointed out the GSK vaccine is like the maternal vaccine that did get approved. “The data clearly show that there was a slight but increased risk in preterm labor,” Dr. Glatt said, “and while not as clearly shown, there was an increase in neonatal death in the group of very small numbers, but any neonatal death is of concern.”
The implications were disturbing, he added, “You’re giving this vaccine to prevent neonatal death.” Though the Pfizer vaccine that was granted approval had a very slight increase in premature birth, the risk wasn’t statistically significant, he pointed out, “and it showed similar benefits in preventing neonatal illness, which can be fatal.”
Dr. Glatt said that there is still a lingering concern with the approved vaccine, and he explained that most clinicians will give it closer to the end of the recommended time window of 34 weeks. “This way, even if there is a slight increase in premature term labor, you’re probably not going to have a serious outcome because the baby will be far enough along.”
A difference in the incidence of preterm birth between the experimental vaccine and placebo groups was predominantly found in low- and middle-income countries, according to Dieussaert’s team, “where approximately 50% of the trial population was enrolled and where the medical need for maternal RSV vaccines is the greatest.”
The RR was 1.56 (95% CI, 1.17-2.10) for low- and middle-income countries and 1.04 (95% CI, 0.68-1.58) for high-income countries.
“If a smaller percentage of participants from low- and middle-income countries had been enrolled in our trial, the RR for preterm birth in the vaccine group as compared with the placebo group might have been reduced in the overall trial population,” they reported.
The authors explained that the data do not reveal the cause of the higher risk for preterm birth in the vaccine group.
“We do not know what caused the signal,” the company’s spokesperson added. “GSK completed all the necessary steps of product development including preclinical toxicology studies and clinical studies in nonpregnant women prior to starting the studies in pregnant women. There were no safety signals identified in any of the earlier parts of the clinical testing. There have been no safety signals identified in the other phase 3 trials for this vaccine candidate.”
Researchers did not find a correlation between preterm births in the treatment vs control groups with gestational age at the time of vaccination or with particular vaccine clinical trial material lots, race, ethnicity, maternal smoking, alcohol consumption, body mass index, or time between study vaccination and delivery, the GSK spokesperson said.
The spokesperson noted that the halted vaccine is different from GSK’s currently approved adjuvanted RSV vaccine (Arexvy) for adults aged 60 years or older.
What’s Next for Other Vaccines
Maternal vaccines have been effective in preventing other diseases in infants, such as tetanus, influenza, and pertussis, but RSV is a very hard virus to make a vaccine for, Dr. Glatt shared.
The need is great to have more than one option for a maternal RSV vaccine, he added, to address any potential supply concerns.
“People have to realize how serious RSV can be in infants,” he said. “It can be a fatal disease. This can be a serious illness even in healthy children.”
A version of this article appeared on Medscape.com.
High Cesarean Rates Persist in Obesity Despite Standardized Protocols
NATIONAL HARBOR, MARYLAND — Implementation of a standardized induction of labor protocol had no significant effect on the rates of cesarean delivery in patients with obesity, based on data from more than 5000 individuals.
Previous research has shown that the risk for cesarean delivery increases by 5% with each 1-kg/m2 increase in body mass index (BMI) among nulliparous patients, said Melissa Riegel, MD, of the University of Pennsylvania, Philadelphia, in a presentation at the meeting sponsored by the Society for Maternal-Fetal Medicine. (abstract 82).
Research on the relationship between obesity and higher cesarean delivery rates “has been clouded by the inability to reduce variation in care,” Dr. Riegel said at the meeting sponsored by the Society for Maternal Fetal Medicine. Failed induction of labor (IOL) is a leading indicator for cesarean delivery, and cesarean delivery is 80% more likely in patients with obesity undergoing IOL than in normal-weight patients, Dr. Riegel said.
Possible explanations for these differences include provider factors such as variability in care management, conscious and unconscious biases, or physiologic differences in patients with obesity such as elevated hormones, differences in the labor curve, and higher doses of oxytocin and prostaglandins, Dr. Riegel said.
Dr. Riegel and colleagues hypothesized that differences in cesarean delivery rates would persist despite a standardized labor induction protocol, thereby supporting the effects of factors other than variations in care on increased cesarean delivery risk after IOL in patients with obesity.
The researchers reviewed data from two sites comparing 2-year periods before and after implementation of an IOL protocol from 2018 to 2022. The study population included nulliparous women with singleton pregnancies at term who underwent IOL with intact membranes and unfavorable cervices, and had a BMI of at least 30 kg/m2 at delivery. The preimplementation group (PRE) included 2480 individuals and the postimplementation group (POST) included 2651 individuals. Patients were divided into weight classes based on BMI: 30-34.9; 35-39.9; ≥40.
The standardized protocol consisted of active labor management with cervical exams, with an amniotomy by the time of the first exam with 4 cm or greater cervical dilation, and further intervention with medication such as oxytocin or an intrauterine pressure catheter if no cervical change was noted after 2 hours.
In a multivariate analysis, the overall cesarean delivery rate was 24.9% before the protocol implementation and 26.0% in the postimplementation group. There were no differences in the risk of cesarean delivery in any obesity class from the PRE to POST period.
In addition, no significant differences appeared in the secondary outcomes of duration of labor, maternal morbidity, or neonatal morbidity, Dr. Riegel said. Nonreassuring fetal heart rate tracing was the most common reason for cesarean delivery across all obesity classes and the PRE and POST groups.
Study limitations included the use of data from only two sites, but the results were strengthened by the large sample size, said Dr. Reigel. The results indicate that reducing variation in IOL management had no significant effect on the relationship between obesity and cesarean delivery and support underlying physiologic explanations, she said.
Making the Case for Physiology
“By standardizing induction practices, we were able to minimize differences in care and better answer why the increased cesarean delivery rate exists in this patient population,” Dr. Riegel said in an interview. The findings were in line with the primary hypothesis that standardized induction would not affect cesarean delivery rates in patients with obesity, she said. Instead, the findings support potential physiologic differences as “the driving force behind this relationship,” she added.
Looking ahead, “There is a role for translational work to investigate the specific biological changes in patients with obesity that might contribute to an increased risk of cesarean delivery and there is also a role for investigating the effectiveness of different labor induction interventions specifically in patients with obesity,” Dr. Riegel said.
Different Induction Protocols Needed for Obese Patients?
“Given that severe maternal morbidity and mortality are continuing to increase in the United States, this study is critical, as we know that both cesarean delivery and obesity are driving factors in increasing maternal morbidity,” said Marissa Platner, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, Georgia, in an interview.
However, the novel takeaway message from the current study is that patients with obesity were more likely to require cesarean delivery even with a protocol in which variation in labor induction techniques are minimized, said Dr. Platner, who was not involved in the study. “This leads to the question of [whether] we should have different standards or protocols for our patients with obesity, as well as a need for clear counseling for these patients early on in pregnancy,” she said.
As for further research, “It would be interesting to see if the risk of cesarean delivery changed based on class of obesity, and the primary drivers of cesarean delivery in this study,” Dr. Platner said. “Additionally, it would be helpful to know how much pitocin was needed for patients, based on their BMI category, to achieve successful vaginal delivery,” she noted.
The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Development. The researchers had no financial conflicts to disclose. Dr. Platner had no financial conflicts to disclose.
NATIONAL HARBOR, MARYLAND — Implementation of a standardized induction of labor protocol had no significant effect on the rates of cesarean delivery in patients with obesity, based on data from more than 5000 individuals.
Previous research has shown that the risk for cesarean delivery increases by 5% with each 1-kg/m2 increase in body mass index (BMI) among nulliparous patients, said Melissa Riegel, MD, of the University of Pennsylvania, Philadelphia, in a presentation at the meeting sponsored by the Society for Maternal-Fetal Medicine. (abstract 82).
Research on the relationship between obesity and higher cesarean delivery rates “has been clouded by the inability to reduce variation in care,” Dr. Riegel said at the meeting sponsored by the Society for Maternal Fetal Medicine. Failed induction of labor (IOL) is a leading indicator for cesarean delivery, and cesarean delivery is 80% more likely in patients with obesity undergoing IOL than in normal-weight patients, Dr. Riegel said.
Possible explanations for these differences include provider factors such as variability in care management, conscious and unconscious biases, or physiologic differences in patients with obesity such as elevated hormones, differences in the labor curve, and higher doses of oxytocin and prostaglandins, Dr. Riegel said.
Dr. Riegel and colleagues hypothesized that differences in cesarean delivery rates would persist despite a standardized labor induction protocol, thereby supporting the effects of factors other than variations in care on increased cesarean delivery risk after IOL in patients with obesity.
The researchers reviewed data from two sites comparing 2-year periods before and after implementation of an IOL protocol from 2018 to 2022. The study population included nulliparous women with singleton pregnancies at term who underwent IOL with intact membranes and unfavorable cervices, and had a BMI of at least 30 kg/m2 at delivery. The preimplementation group (PRE) included 2480 individuals and the postimplementation group (POST) included 2651 individuals. Patients were divided into weight classes based on BMI: 30-34.9; 35-39.9; ≥40.
The standardized protocol consisted of active labor management with cervical exams, with an amniotomy by the time of the first exam with 4 cm or greater cervical dilation, and further intervention with medication such as oxytocin or an intrauterine pressure catheter if no cervical change was noted after 2 hours.
In a multivariate analysis, the overall cesarean delivery rate was 24.9% before the protocol implementation and 26.0% in the postimplementation group. There were no differences in the risk of cesarean delivery in any obesity class from the PRE to POST period.
In addition, no significant differences appeared in the secondary outcomes of duration of labor, maternal morbidity, or neonatal morbidity, Dr. Riegel said. Nonreassuring fetal heart rate tracing was the most common reason for cesarean delivery across all obesity classes and the PRE and POST groups.
Study limitations included the use of data from only two sites, but the results were strengthened by the large sample size, said Dr. Reigel. The results indicate that reducing variation in IOL management had no significant effect on the relationship between obesity and cesarean delivery and support underlying physiologic explanations, she said.
Making the Case for Physiology
“By standardizing induction practices, we were able to minimize differences in care and better answer why the increased cesarean delivery rate exists in this patient population,” Dr. Riegel said in an interview. The findings were in line with the primary hypothesis that standardized induction would not affect cesarean delivery rates in patients with obesity, she said. Instead, the findings support potential physiologic differences as “the driving force behind this relationship,” she added.
Looking ahead, “There is a role for translational work to investigate the specific biological changes in patients with obesity that might contribute to an increased risk of cesarean delivery and there is also a role for investigating the effectiveness of different labor induction interventions specifically in patients with obesity,” Dr. Riegel said.
Different Induction Protocols Needed for Obese Patients?
“Given that severe maternal morbidity and mortality are continuing to increase in the United States, this study is critical, as we know that both cesarean delivery and obesity are driving factors in increasing maternal morbidity,” said Marissa Platner, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, Georgia, in an interview.
However, the novel takeaway message from the current study is that patients with obesity were more likely to require cesarean delivery even with a protocol in which variation in labor induction techniques are minimized, said Dr. Platner, who was not involved in the study. “This leads to the question of [whether] we should have different standards or protocols for our patients with obesity, as well as a need for clear counseling for these patients early on in pregnancy,” she said.
As for further research, “It would be interesting to see if the risk of cesarean delivery changed based on class of obesity, and the primary drivers of cesarean delivery in this study,” Dr. Platner said. “Additionally, it would be helpful to know how much pitocin was needed for patients, based on their BMI category, to achieve successful vaginal delivery,” she noted.
The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Development. The researchers had no financial conflicts to disclose. Dr. Platner had no financial conflicts to disclose.
NATIONAL HARBOR, MARYLAND — Implementation of a standardized induction of labor protocol had no significant effect on the rates of cesarean delivery in patients with obesity, based on data from more than 5000 individuals.
Previous research has shown that the risk for cesarean delivery increases by 5% with each 1-kg/m2 increase in body mass index (BMI) among nulliparous patients, said Melissa Riegel, MD, of the University of Pennsylvania, Philadelphia, in a presentation at the meeting sponsored by the Society for Maternal-Fetal Medicine. (abstract 82).
Research on the relationship between obesity and higher cesarean delivery rates “has been clouded by the inability to reduce variation in care,” Dr. Riegel said at the meeting sponsored by the Society for Maternal Fetal Medicine. Failed induction of labor (IOL) is a leading indicator for cesarean delivery, and cesarean delivery is 80% more likely in patients with obesity undergoing IOL than in normal-weight patients, Dr. Riegel said.
Possible explanations for these differences include provider factors such as variability in care management, conscious and unconscious biases, or physiologic differences in patients with obesity such as elevated hormones, differences in the labor curve, and higher doses of oxytocin and prostaglandins, Dr. Riegel said.
Dr. Riegel and colleagues hypothesized that differences in cesarean delivery rates would persist despite a standardized labor induction protocol, thereby supporting the effects of factors other than variations in care on increased cesarean delivery risk after IOL in patients with obesity.
The researchers reviewed data from two sites comparing 2-year periods before and after implementation of an IOL protocol from 2018 to 2022. The study population included nulliparous women with singleton pregnancies at term who underwent IOL with intact membranes and unfavorable cervices, and had a BMI of at least 30 kg/m2 at delivery. The preimplementation group (PRE) included 2480 individuals and the postimplementation group (POST) included 2651 individuals. Patients were divided into weight classes based on BMI: 30-34.9; 35-39.9; ≥40.
The standardized protocol consisted of active labor management with cervical exams, with an amniotomy by the time of the first exam with 4 cm or greater cervical dilation, and further intervention with medication such as oxytocin or an intrauterine pressure catheter if no cervical change was noted after 2 hours.
In a multivariate analysis, the overall cesarean delivery rate was 24.9% before the protocol implementation and 26.0% in the postimplementation group. There were no differences in the risk of cesarean delivery in any obesity class from the PRE to POST period.
In addition, no significant differences appeared in the secondary outcomes of duration of labor, maternal morbidity, or neonatal morbidity, Dr. Riegel said. Nonreassuring fetal heart rate tracing was the most common reason for cesarean delivery across all obesity classes and the PRE and POST groups.
Study limitations included the use of data from only two sites, but the results were strengthened by the large sample size, said Dr. Reigel. The results indicate that reducing variation in IOL management had no significant effect on the relationship between obesity and cesarean delivery and support underlying physiologic explanations, she said.
Making the Case for Physiology
“By standardizing induction practices, we were able to minimize differences in care and better answer why the increased cesarean delivery rate exists in this patient population,” Dr. Riegel said in an interview. The findings were in line with the primary hypothesis that standardized induction would not affect cesarean delivery rates in patients with obesity, she said. Instead, the findings support potential physiologic differences as “the driving force behind this relationship,” she added.
Looking ahead, “There is a role for translational work to investigate the specific biological changes in patients with obesity that might contribute to an increased risk of cesarean delivery and there is also a role for investigating the effectiveness of different labor induction interventions specifically in patients with obesity,” Dr. Riegel said.
Different Induction Protocols Needed for Obese Patients?
“Given that severe maternal morbidity and mortality are continuing to increase in the United States, this study is critical, as we know that both cesarean delivery and obesity are driving factors in increasing maternal morbidity,” said Marissa Platner, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, Georgia, in an interview.
However, the novel takeaway message from the current study is that patients with obesity were more likely to require cesarean delivery even with a protocol in which variation in labor induction techniques are minimized, said Dr. Platner, who was not involved in the study. “This leads to the question of [whether] we should have different standards or protocols for our patients with obesity, as well as a need for clear counseling for these patients early on in pregnancy,” she said.
As for further research, “It would be interesting to see if the risk of cesarean delivery changed based on class of obesity, and the primary drivers of cesarean delivery in this study,” Dr. Platner said. “Additionally, it would be helpful to know how much pitocin was needed for patients, based on their BMI category, to achieve successful vaginal delivery,” she noted.
The study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Development. The researchers had no financial conflicts to disclose. Dr. Platner had no financial conflicts to disclose.
AT THE PREGNANCY MEETING
Medicare Doc Pay Cut Eased, but When Will Serious Revisions Come?
President Joe Biden on March 9 signed into law a measure that softened — but did not completely eliminate — a 2024 cut in a key rate used to determine how physicians are paid for treating Medicare patients.
While physician groups hailed the move as partial relief, they say they’ll continue to press for broader changes in the Medicare physician fee schedule.
The Medicare provision was tucked into a larger spending package approved by the US House and Senate.
The American Academy of Family Physicians (AAFP), the American Medical Association (AMA), and other groups have lobbied Congress for months to undo a 3.4% cut in the base rate, or conversion factor, in the physician fee schedule for 2024.
The conversion factor is used in calculations to determine reimbursement for myriad other services. Federal Medicare officials said the cut would mean a 1.25% decrease in overall payments in 2024, compared with 2023.
“With the passage of this legislation, Congress has offset 2.93% of that payment cut,” said Steven P. Furr, MD, AAFP’s president in a statement. “We appreciate this temporary measure but continue to urge Congress to advance comprehensive, long-term Medicare payment reform.”
In a statement, Representative Larry Bucshon, MD (R-IN), said the payment cut could not be completely eliminated because of budget constraints.
The Medicare physician fee schedule covers much of the care clinicians provide to people older than 65 and those with disabilities. It covers about 8000 different types of services, ranging from office visits to surgical procedures, imaging, and tests, according to the Medicare Payment Advisory Commission (MedPAC).
Along with physicians, the fee schedule sets payments for nurse practitioners, physician assistants, podiatrists, physical therapists, psychologists, and other clinicians.
In 2021, the Medicare program and its beneficiaries paid $92.8 billion for services provided by almost 1.3 million clinicians, MedPAC said.
Larger Changes Ahead?
Rep. Bucshon is among the physicians serving in the House who are pressing for a permanent revamp of the Medicare physician fee schedule. He cosponsored a bill (HR 2474) that would peg future annual increases in the physician fee schedule to the Medicare Economic Index, which would reflect inflation’s effect.
In April, more than 120 state and national medical groups signed onto an AMA-led letter urging Congress to pass this bill.
The measure is a key priority for the AMA. The organization reached out repeatedly last year to federal officials about it through its own in-house lobbyists, this news organization found through a review of congressional lobbying forms submitted by AMA.
These required disclosure forms reveal how much AMA and other organizations spend each quarter to appeal to members of Congress and federal agencies on specific issues. The disclosure forms do not include a detailed accounting of spending on each issue.
But they do show which issues are priorities for an organization. AMA’s in-house lobbyists reported raising dozens of issues in 2024 within contacts in Congress and federal agencies. These issues included abortion access, maternal health, physician burnout, and potential for bias in clinical use of algorithms, as well as Medicare payment for physicians.
AMA reported spending estimated cost of $20.6 million. (AMA spent $6.7 million in the first quarter, $4.75 million in the second quarter, $3.42 million in the third quarter, and $5.74 million in the fourth quarter.)
In a March 6 statement, Jesse M. Ehrenfeld, MD, MPH, AMA president, urged Congress to turn to more serious consideration of Medicare physician pay beyond short-term tweaks attached to other larger bills.
“As physicians, we are trained to run toward emergencies. We urge Congress to do the same,” Dr. Ehrenfeld said. “We encourage Congress to act if this policy decision is an emergency because — in fact — it is. It is well past time to put an end to stopgap measures that fail to address the underlying causes of the continuing decline in Medicare physician payments.”
There’s bipartisan interest in a revamp of the physician fee schedule amid widespread criticism of the last such overhaul, the Medicare Access and CHIP Reauthorization Act of 2015.
For example, Senate Budget Chairman Sheldon Whitehouse (D-RI) has proposed the creation of a technical advisory committee to improve how Medicare sets the physician fee schedule. The existing fee schedule provides too little money for primary care services and primary care provider pay, contributing to shortages, Sen. Whitehouse said.
Sen. Whitehouse on March 6 held a hearing on ways to beef up US primary care. Among the experts who appeared was Amol Navathe, MD, PhD, of the University of Pennsylvania, Philadelphia, Pennsylvania.
Dr. Navathe said the current Medicare physician fee schedule tilts in favor of procedural services, leading to “underinvestment in cognitive, diagnostic, and supportive services such as primary care.”
In addition, much of what primary care clinicians do, “such as addressing social challenges, is not included in the codes of the fee schedule itself,” said Dr. Navathe, who also serves as the vice chairman of MedPAC.
It’s unclear when Congress will attempt a serious revision to the Medicare physician fee schedule. Lawmakers are unlikely to take on such a major challenge in this election year.
There would be significant opposition and challenges for lawmakers in trying to clear a bill that added an inflation adjustment for what’s already seen as an imperfect physician fee schedule, said Mark E. Miller, PhD, executive vice president of healthcare at the philanthropy Arnold Ventures, which studies how payment decisions affect medical care.
“That bill could cost a lot of money and raise a lot of questions,” Dr. Miller said.
A version of this article appeared on Medscape.com.
President Joe Biden on March 9 signed into law a measure that softened — but did not completely eliminate — a 2024 cut in a key rate used to determine how physicians are paid for treating Medicare patients.
While physician groups hailed the move as partial relief, they say they’ll continue to press for broader changes in the Medicare physician fee schedule.
The Medicare provision was tucked into a larger spending package approved by the US House and Senate.
The American Academy of Family Physicians (AAFP), the American Medical Association (AMA), and other groups have lobbied Congress for months to undo a 3.4% cut in the base rate, or conversion factor, in the physician fee schedule for 2024.
The conversion factor is used in calculations to determine reimbursement for myriad other services. Federal Medicare officials said the cut would mean a 1.25% decrease in overall payments in 2024, compared with 2023.
“With the passage of this legislation, Congress has offset 2.93% of that payment cut,” said Steven P. Furr, MD, AAFP’s president in a statement. “We appreciate this temporary measure but continue to urge Congress to advance comprehensive, long-term Medicare payment reform.”
In a statement, Representative Larry Bucshon, MD (R-IN), said the payment cut could not be completely eliminated because of budget constraints.
The Medicare physician fee schedule covers much of the care clinicians provide to people older than 65 and those with disabilities. It covers about 8000 different types of services, ranging from office visits to surgical procedures, imaging, and tests, according to the Medicare Payment Advisory Commission (MedPAC).
Along with physicians, the fee schedule sets payments for nurse practitioners, physician assistants, podiatrists, physical therapists, psychologists, and other clinicians.
In 2021, the Medicare program and its beneficiaries paid $92.8 billion for services provided by almost 1.3 million clinicians, MedPAC said.
Larger Changes Ahead?
Rep. Bucshon is among the physicians serving in the House who are pressing for a permanent revamp of the Medicare physician fee schedule. He cosponsored a bill (HR 2474) that would peg future annual increases in the physician fee schedule to the Medicare Economic Index, which would reflect inflation’s effect.
In April, more than 120 state and national medical groups signed onto an AMA-led letter urging Congress to pass this bill.
The measure is a key priority for the AMA. The organization reached out repeatedly last year to federal officials about it through its own in-house lobbyists, this news organization found through a review of congressional lobbying forms submitted by AMA.
These required disclosure forms reveal how much AMA and other organizations spend each quarter to appeal to members of Congress and federal agencies on specific issues. The disclosure forms do not include a detailed accounting of spending on each issue.
But they do show which issues are priorities for an organization. AMA’s in-house lobbyists reported raising dozens of issues in 2024 within contacts in Congress and federal agencies. These issues included abortion access, maternal health, physician burnout, and potential for bias in clinical use of algorithms, as well as Medicare payment for physicians.
AMA reported spending estimated cost of $20.6 million. (AMA spent $6.7 million in the first quarter, $4.75 million in the second quarter, $3.42 million in the third quarter, and $5.74 million in the fourth quarter.)
In a March 6 statement, Jesse M. Ehrenfeld, MD, MPH, AMA president, urged Congress to turn to more serious consideration of Medicare physician pay beyond short-term tweaks attached to other larger bills.
“As physicians, we are trained to run toward emergencies. We urge Congress to do the same,” Dr. Ehrenfeld said. “We encourage Congress to act if this policy decision is an emergency because — in fact — it is. It is well past time to put an end to stopgap measures that fail to address the underlying causes of the continuing decline in Medicare physician payments.”
There’s bipartisan interest in a revamp of the physician fee schedule amid widespread criticism of the last such overhaul, the Medicare Access and CHIP Reauthorization Act of 2015.
For example, Senate Budget Chairman Sheldon Whitehouse (D-RI) has proposed the creation of a technical advisory committee to improve how Medicare sets the physician fee schedule. The existing fee schedule provides too little money for primary care services and primary care provider pay, contributing to shortages, Sen. Whitehouse said.
Sen. Whitehouse on March 6 held a hearing on ways to beef up US primary care. Among the experts who appeared was Amol Navathe, MD, PhD, of the University of Pennsylvania, Philadelphia, Pennsylvania.
Dr. Navathe said the current Medicare physician fee schedule tilts in favor of procedural services, leading to “underinvestment in cognitive, diagnostic, and supportive services such as primary care.”
In addition, much of what primary care clinicians do, “such as addressing social challenges, is not included in the codes of the fee schedule itself,” said Dr. Navathe, who also serves as the vice chairman of MedPAC.
It’s unclear when Congress will attempt a serious revision to the Medicare physician fee schedule. Lawmakers are unlikely to take on such a major challenge in this election year.
There would be significant opposition and challenges for lawmakers in trying to clear a bill that added an inflation adjustment for what’s already seen as an imperfect physician fee schedule, said Mark E. Miller, PhD, executive vice president of healthcare at the philanthropy Arnold Ventures, which studies how payment decisions affect medical care.
“That bill could cost a lot of money and raise a lot of questions,” Dr. Miller said.
A version of this article appeared on Medscape.com.
President Joe Biden on March 9 signed into law a measure that softened — but did not completely eliminate — a 2024 cut in a key rate used to determine how physicians are paid for treating Medicare patients.
While physician groups hailed the move as partial relief, they say they’ll continue to press for broader changes in the Medicare physician fee schedule.
The Medicare provision was tucked into a larger spending package approved by the US House and Senate.
The American Academy of Family Physicians (AAFP), the American Medical Association (AMA), and other groups have lobbied Congress for months to undo a 3.4% cut in the base rate, or conversion factor, in the physician fee schedule for 2024.
The conversion factor is used in calculations to determine reimbursement for myriad other services. Federal Medicare officials said the cut would mean a 1.25% decrease in overall payments in 2024, compared with 2023.
“With the passage of this legislation, Congress has offset 2.93% of that payment cut,” said Steven P. Furr, MD, AAFP’s president in a statement. “We appreciate this temporary measure but continue to urge Congress to advance comprehensive, long-term Medicare payment reform.”
In a statement, Representative Larry Bucshon, MD (R-IN), said the payment cut could not be completely eliminated because of budget constraints.
The Medicare physician fee schedule covers much of the care clinicians provide to people older than 65 and those with disabilities. It covers about 8000 different types of services, ranging from office visits to surgical procedures, imaging, and tests, according to the Medicare Payment Advisory Commission (MedPAC).
Along with physicians, the fee schedule sets payments for nurse practitioners, physician assistants, podiatrists, physical therapists, psychologists, and other clinicians.
In 2021, the Medicare program and its beneficiaries paid $92.8 billion for services provided by almost 1.3 million clinicians, MedPAC said.
Larger Changes Ahead?
Rep. Bucshon is among the physicians serving in the House who are pressing for a permanent revamp of the Medicare physician fee schedule. He cosponsored a bill (HR 2474) that would peg future annual increases in the physician fee schedule to the Medicare Economic Index, which would reflect inflation’s effect.
In April, more than 120 state and national medical groups signed onto an AMA-led letter urging Congress to pass this bill.
The measure is a key priority for the AMA. The organization reached out repeatedly last year to federal officials about it through its own in-house lobbyists, this news organization found through a review of congressional lobbying forms submitted by AMA.
These required disclosure forms reveal how much AMA and other organizations spend each quarter to appeal to members of Congress and federal agencies on specific issues. The disclosure forms do not include a detailed accounting of spending on each issue.
But they do show which issues are priorities for an organization. AMA’s in-house lobbyists reported raising dozens of issues in 2024 within contacts in Congress and federal agencies. These issues included abortion access, maternal health, physician burnout, and potential for bias in clinical use of algorithms, as well as Medicare payment for physicians.
AMA reported spending estimated cost of $20.6 million. (AMA spent $6.7 million in the first quarter, $4.75 million in the second quarter, $3.42 million in the third quarter, and $5.74 million in the fourth quarter.)
In a March 6 statement, Jesse M. Ehrenfeld, MD, MPH, AMA president, urged Congress to turn to more serious consideration of Medicare physician pay beyond short-term tweaks attached to other larger bills.
“As physicians, we are trained to run toward emergencies. We urge Congress to do the same,” Dr. Ehrenfeld said. “We encourage Congress to act if this policy decision is an emergency because — in fact — it is. It is well past time to put an end to stopgap measures that fail to address the underlying causes of the continuing decline in Medicare physician payments.”
There’s bipartisan interest in a revamp of the physician fee schedule amid widespread criticism of the last such overhaul, the Medicare Access and CHIP Reauthorization Act of 2015.
For example, Senate Budget Chairman Sheldon Whitehouse (D-RI) has proposed the creation of a technical advisory committee to improve how Medicare sets the physician fee schedule. The existing fee schedule provides too little money for primary care services and primary care provider pay, contributing to shortages, Sen. Whitehouse said.
Sen. Whitehouse on March 6 held a hearing on ways to beef up US primary care. Among the experts who appeared was Amol Navathe, MD, PhD, of the University of Pennsylvania, Philadelphia, Pennsylvania.
Dr. Navathe said the current Medicare physician fee schedule tilts in favor of procedural services, leading to “underinvestment in cognitive, diagnostic, and supportive services such as primary care.”
In addition, much of what primary care clinicians do, “such as addressing social challenges, is not included in the codes of the fee schedule itself,” said Dr. Navathe, who also serves as the vice chairman of MedPAC.
It’s unclear when Congress will attempt a serious revision to the Medicare physician fee schedule. Lawmakers are unlikely to take on such a major challenge in this election year.
There would be significant opposition and challenges for lawmakers in trying to clear a bill that added an inflation adjustment for what’s already seen as an imperfect physician fee schedule, said Mark E. Miller, PhD, executive vice president of healthcare at the philanthropy Arnold Ventures, which studies how payment decisions affect medical care.
“That bill could cost a lot of money and raise a lot of questions,” Dr. Miller said.
A version of this article appeared on Medscape.com.
Vitamin D Supplements May Be a Double-Edged Sword
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
Imagine, if you will, the great Cathedral of Our Lady of Correlation. You walk through the majestic oak doors depicting the link between ice cream sales and shark attacks, past the rose window depicting the cardiovascular benefits of red wine, and down the aisles frescoed in dramatic images showing how Facebook usage is associated with less life satisfaction. And then you reach the altar, the holy of holies where, emblazoned in shimmering pyrite, you see the patron saint of this church: vitamin D.
Yes, if you’ve watched this space, then you know that I have little truck with the wildly popular supplement. In all of clinical research, I believe that there is no molecule with stronger data for correlation and weaker data for causation.
Low serum vitamin D levels have been linked to higher risks for heart disease, cancer, falls, COVID, dementia, C diff, and others. And yet, when we do randomized trials of vitamin D supplementation — the thing that can prove that the low level was causally linked to the outcome of interest — we get negative results.
Trials aren’t perfect, of course, and we’ll talk in a moment about a big one that had some issues. But we are at a point where we need to either be vitamin D apologists, saying, “Forget what those lying RCTs tell you and buy this supplement” — an $800 million-a-year industry, by the way — or conclude that vitamin D levels are a convenient marker of various lifestyle factors that are associated with better outcomes: markers of exercise, getting outside, eating a varied diet.
Or perhaps vitamin D supplements have real effects. It’s just that the beneficial effects are matched by the harmful ones. Stay tuned.
The Women’s Health Initiative remains among the largest randomized trials of vitamin D and calcium supplementation ever conducted — and a major contributor to the negative outcomes of vitamin D trials.
But if you dig into the inclusion and exclusion criteria for this trial, you’ll find that individuals were allowed to continue taking vitamins and supplements while they were in the trial, regardless of their randomization status. In fact, the majority took supplements at baseline, and more took supplements over time.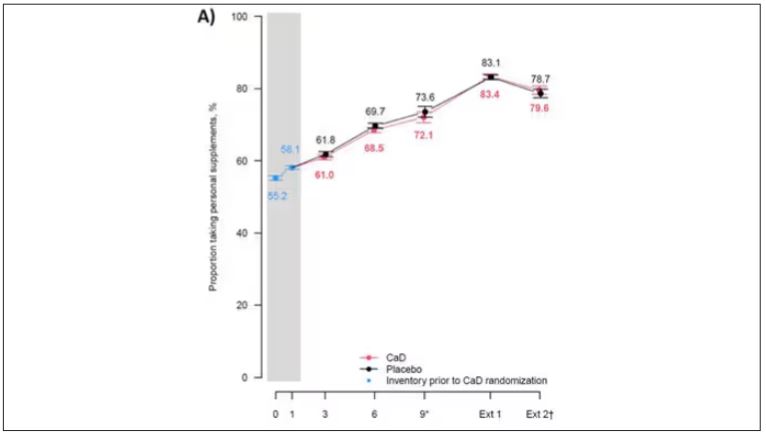
That means, of course, that people in the placebo group, who were getting sugar pills instead of vitamin D and calcium, may have been taking vitamin D and calcium on the side. That would certainly bias the results of the trial toward the null, which is what the primary analyses showed. To wit, the original analysis of the Women’s Health Initiative trial showed no effect of randomization to vitamin D supplementation on improving cancer or cardiovascular outcomes.
But the Women’s Health Initiative trial started 30 years ago. Today, with the benefit of decades of follow-up, we can re-investigate — and perhaps re-litigate — those findings, courtesy of this study, “Long-Term Effect of Randomization to Calcium and Vitamin D Supplementation on Health in Older Women” appearing in Annals of Internal Medicine.
Dr Cynthia Thomson, of the Mel and Enid Zuckerman College of Public Health at the University of Arizona, and colleagues led this updated analysis focused on two findings that had been hinted at, but not statistically confirmed, in other vitamin D studies: a potential for the supplement to reduce the risk for cancer, and a potential for it to increase the risk for heart disease.
The randomized trial itself only lasted 7 years. What we are seeing in this analysis of 36,282 women is outcomes that happened at any time from randomization to the end of 2023 — around 20 years after the randomization to supplementation stopped. But, the researchers would argue, that’s probably okay. Cancer and heart disease take time to develop; we see lung cancer long after people stop smoking. So a history of consistent vitamin D supplementation may indeed be protective — or harmful.
Here are the top-line results. Those randomized to vitamin D and calcium supplementation had a 7% reduction in the rate of death from cancer, driven primarily by a reduction in colorectal cancer. This was statistically significant. Also statistically significant? Those randomized to supplementation had a 6% increase in the rate of death from cardiovascular disease. Put those findings together and what do you get? Stone-cold nothing, in terms of overall mortality.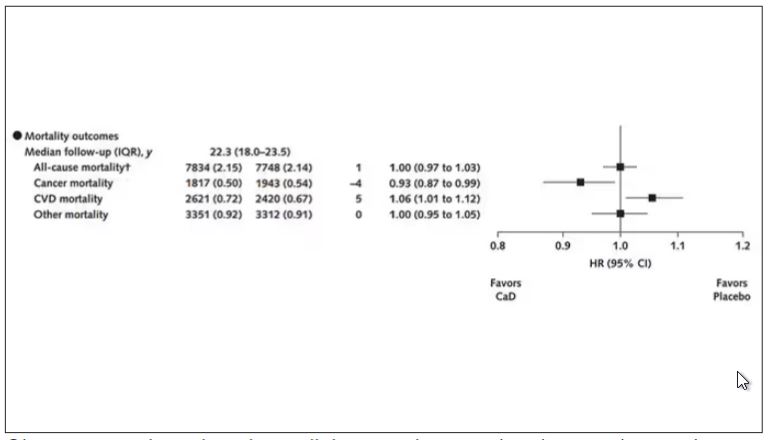
Okay, you say, but what about all that supplementation that was happening outside of the context of the trial, biasing our results toward the null?
The researchers finally clue us in.
First of all, I’ll tell you that, yes, people who were supplementing outside of the trial had higher baseline vitamin D levels — a median of 54.5 nmol/L vs 32.8 nmol/L. This may be because they were supplementing with vitamin D, but it could also be because people who take supplements tend to do other healthy things — another correlation to add to the great cathedral.
To get a better view of the real effects of randomization, the authors restricted the analysis to just those who did not use outside supplements. If vitamin D supplements help, then these are the people they should help. This group had about a 11% reduction in the incidence of cancer — statistically significant — and a 7% reduction in cancer mortality that did not meet the bar for statistical significance.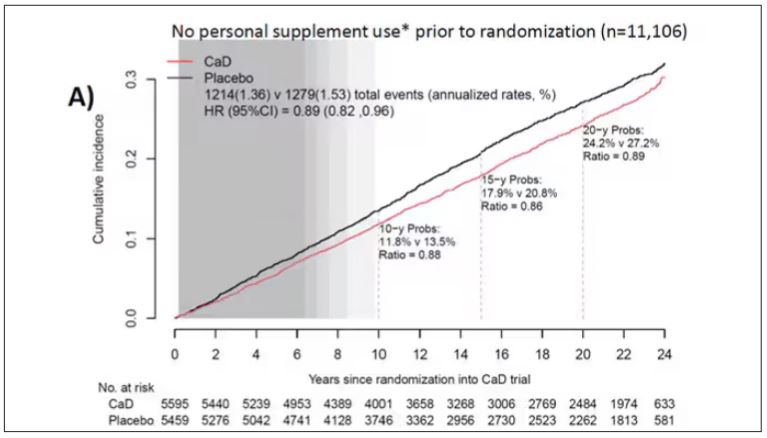
There was no increase in cardiovascular disease among this group. But this small effect on cancer was nowhere near enough to significantly reduce the rate of all-cause mortality.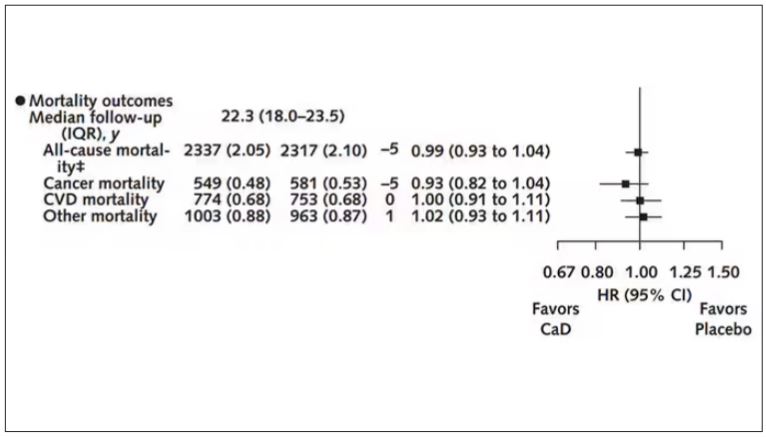
Among those using supplements, vitamin D supplementation didn’t really move the needle on any outcome.
I know what you’re thinking: How many of these women were vitamin D deficient when we got started? These results may simply be telling us that people who have normal vitamin D levels are fine to go without supplementation.
Nearly three fourths of women who were not taking supplements entered the trial with vitamin D levels below the 50 nmol/L cutoff that the authors suggest would qualify for deficiency. Around half of those who used supplements were deficient. And yet, frustratingly, I could not find data on the effect of randomization to supplementation stratified by baseline vitamin D level. I even reached out to Dr Thomson to ask about this. She replied, “We did not stratify on baseline values because the numbers are too small statistically to test this.” Sorry.
In the meantime, I can tell you that for your “average woman,” vitamin D supplementation likely has no effect on mortality. It might modestly reduce the risk for certain cancers while increasing the risk for heart disease (probably through coronary calcification). So, there might be some room for personalization here. Perhaps women with a strong family history of cancer or other risk factors would do better with supplements, and those with a high risk for heart disease would do worse. Seems like a strategy that could be tested in a clinical trial. But maybe we could ask the participants to give up their extracurricular supplement use before they enter the trial. F. Perry Wilson, MD, MSCE, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and his book, How Medicine Works and When It Doesn’t, is available now.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
Imagine, if you will, the great Cathedral of Our Lady of Correlation. You walk through the majestic oak doors depicting the link between ice cream sales and shark attacks, past the rose window depicting the cardiovascular benefits of red wine, and down the aisles frescoed in dramatic images showing how Facebook usage is associated with less life satisfaction. And then you reach the altar, the holy of holies where, emblazoned in shimmering pyrite, you see the patron saint of this church: vitamin D.
Yes, if you’ve watched this space, then you know that I have little truck with the wildly popular supplement. In all of clinical research, I believe that there is no molecule with stronger data for correlation and weaker data for causation.
Low serum vitamin D levels have been linked to higher risks for heart disease, cancer, falls, COVID, dementia, C diff, and others. And yet, when we do randomized trials of vitamin D supplementation — the thing that can prove that the low level was causally linked to the outcome of interest — we get negative results.
Trials aren’t perfect, of course, and we’ll talk in a moment about a big one that had some issues. But we are at a point where we need to either be vitamin D apologists, saying, “Forget what those lying RCTs tell you and buy this supplement” — an $800 million-a-year industry, by the way — or conclude that vitamin D levels are a convenient marker of various lifestyle factors that are associated with better outcomes: markers of exercise, getting outside, eating a varied diet.
Or perhaps vitamin D supplements have real effects. It’s just that the beneficial effects are matched by the harmful ones. Stay tuned.
The Women’s Health Initiative remains among the largest randomized trials of vitamin D and calcium supplementation ever conducted — and a major contributor to the negative outcomes of vitamin D trials.
But if you dig into the inclusion and exclusion criteria for this trial, you’ll find that individuals were allowed to continue taking vitamins and supplements while they were in the trial, regardless of their randomization status. In fact, the majority took supplements at baseline, and more took supplements over time.
That means, of course, that people in the placebo group, who were getting sugar pills instead of vitamin D and calcium, may have been taking vitamin D and calcium on the side. That would certainly bias the results of the trial toward the null, which is what the primary analyses showed. To wit, the original analysis of the Women’s Health Initiative trial showed no effect of randomization to vitamin D supplementation on improving cancer or cardiovascular outcomes.
But the Women’s Health Initiative trial started 30 years ago. Today, with the benefit of decades of follow-up, we can re-investigate — and perhaps re-litigate — those findings, courtesy of this study, “Long-Term Effect of Randomization to Calcium and Vitamin D Supplementation on Health in Older Women” appearing in Annals of Internal Medicine.
Dr Cynthia Thomson, of the Mel and Enid Zuckerman College of Public Health at the University of Arizona, and colleagues led this updated analysis focused on two findings that had been hinted at, but not statistically confirmed, in other vitamin D studies: a potential for the supplement to reduce the risk for cancer, and a potential for it to increase the risk for heart disease.
The randomized trial itself only lasted 7 years. What we are seeing in this analysis of 36,282 women is outcomes that happened at any time from randomization to the end of 2023 — around 20 years after the randomization to supplementation stopped. But, the researchers would argue, that’s probably okay. Cancer and heart disease take time to develop; we see lung cancer long after people stop smoking. So a history of consistent vitamin D supplementation may indeed be protective — or harmful.
Here are the top-line results. Those randomized to vitamin D and calcium supplementation had a 7% reduction in the rate of death from cancer, driven primarily by a reduction in colorectal cancer. This was statistically significant. Also statistically significant? Those randomized to supplementation had a 6% increase in the rate of death from cardiovascular disease. Put those findings together and what do you get? Stone-cold nothing, in terms of overall mortality.
Okay, you say, but what about all that supplementation that was happening outside of the context of the trial, biasing our results toward the null?
The researchers finally clue us in.
First of all, I’ll tell you that, yes, people who were supplementing outside of the trial had higher baseline vitamin D levels — a median of 54.5 nmol/L vs 32.8 nmol/L. This may be because they were supplementing with vitamin D, but it could also be because people who take supplements tend to do other healthy things — another correlation to add to the great cathedral.
To get a better view of the real effects of randomization, the authors restricted the analysis to just those who did not use outside supplements. If vitamin D supplements help, then these are the people they should help. This group had about a 11% reduction in the incidence of cancer — statistically significant — and a 7% reduction in cancer mortality that did not meet the bar for statistical significance.
There was no increase in cardiovascular disease among this group. But this small effect on cancer was nowhere near enough to significantly reduce the rate of all-cause mortality.
Among those using supplements, vitamin D supplementation didn’t really move the needle on any outcome.
I know what you’re thinking: How many of these women were vitamin D deficient when we got started? These results may simply be telling us that people who have normal vitamin D levels are fine to go without supplementation.
Nearly three fourths of women who were not taking supplements entered the trial with vitamin D levels below the 50 nmol/L cutoff that the authors suggest would qualify for deficiency. Around half of those who used supplements were deficient. And yet, frustratingly, I could not find data on the effect of randomization to supplementation stratified by baseline vitamin D level. I even reached out to Dr Thomson to ask about this. She replied, “We did not stratify on baseline values because the numbers are too small statistically to test this.” Sorry.
In the meantime, I can tell you that for your “average woman,” vitamin D supplementation likely has no effect on mortality. It might modestly reduce the risk for certain cancers while increasing the risk for heart disease (probably through coronary calcification). So, there might be some room for personalization here. Perhaps women with a strong family history of cancer or other risk factors would do better with supplements, and those with a high risk for heart disease would do worse. Seems like a strategy that could be tested in a clinical trial. But maybe we could ask the participants to give up their extracurricular supplement use before they enter the trial. F. Perry Wilson, MD, MSCE, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and his book, How Medicine Works and When It Doesn’t, is available now.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
Imagine, if you will, the great Cathedral of Our Lady of Correlation. You walk through the majestic oak doors depicting the link between ice cream sales and shark attacks, past the rose window depicting the cardiovascular benefits of red wine, and down the aisles frescoed in dramatic images showing how Facebook usage is associated with less life satisfaction. And then you reach the altar, the holy of holies where, emblazoned in shimmering pyrite, you see the patron saint of this church: vitamin D.
Yes, if you’ve watched this space, then you know that I have little truck with the wildly popular supplement. In all of clinical research, I believe that there is no molecule with stronger data for correlation and weaker data for causation.
Low serum vitamin D levels have been linked to higher risks for heart disease, cancer, falls, COVID, dementia, C diff, and others. And yet, when we do randomized trials of vitamin D supplementation — the thing that can prove that the low level was causally linked to the outcome of interest — we get negative results.
Trials aren’t perfect, of course, and we’ll talk in a moment about a big one that had some issues. But we are at a point where we need to either be vitamin D apologists, saying, “Forget what those lying RCTs tell you and buy this supplement” — an $800 million-a-year industry, by the way — or conclude that vitamin D levels are a convenient marker of various lifestyle factors that are associated with better outcomes: markers of exercise, getting outside, eating a varied diet.
Or perhaps vitamin D supplements have real effects. It’s just that the beneficial effects are matched by the harmful ones. Stay tuned.
The Women’s Health Initiative remains among the largest randomized trials of vitamin D and calcium supplementation ever conducted — and a major contributor to the negative outcomes of vitamin D trials.
But if you dig into the inclusion and exclusion criteria for this trial, you’ll find that individuals were allowed to continue taking vitamins and supplements while they were in the trial, regardless of their randomization status. In fact, the majority took supplements at baseline, and more took supplements over time.
That means, of course, that people in the placebo group, who were getting sugar pills instead of vitamin D and calcium, may have been taking vitamin D and calcium on the side. That would certainly bias the results of the trial toward the null, which is what the primary analyses showed. To wit, the original analysis of the Women’s Health Initiative trial showed no effect of randomization to vitamin D supplementation on improving cancer or cardiovascular outcomes.
But the Women’s Health Initiative trial started 30 years ago. Today, with the benefit of decades of follow-up, we can re-investigate — and perhaps re-litigate — those findings, courtesy of this study, “Long-Term Effect of Randomization to Calcium and Vitamin D Supplementation on Health in Older Women” appearing in Annals of Internal Medicine.
Dr Cynthia Thomson, of the Mel and Enid Zuckerman College of Public Health at the University of Arizona, and colleagues led this updated analysis focused on two findings that had been hinted at, but not statistically confirmed, in other vitamin D studies: a potential for the supplement to reduce the risk for cancer, and a potential for it to increase the risk for heart disease.
The randomized trial itself only lasted 7 years. What we are seeing in this analysis of 36,282 women is outcomes that happened at any time from randomization to the end of 2023 — around 20 years after the randomization to supplementation stopped. But, the researchers would argue, that’s probably okay. Cancer and heart disease take time to develop; we see lung cancer long after people stop smoking. So a history of consistent vitamin D supplementation may indeed be protective — or harmful.
Here are the top-line results. Those randomized to vitamin D and calcium supplementation had a 7% reduction in the rate of death from cancer, driven primarily by a reduction in colorectal cancer. This was statistically significant. Also statistically significant? Those randomized to supplementation had a 6% increase in the rate of death from cardiovascular disease. Put those findings together and what do you get? Stone-cold nothing, in terms of overall mortality.
Okay, you say, but what about all that supplementation that was happening outside of the context of the trial, biasing our results toward the null?
The researchers finally clue us in.
First of all, I’ll tell you that, yes, people who were supplementing outside of the trial had higher baseline vitamin D levels — a median of 54.5 nmol/L vs 32.8 nmol/L. This may be because they were supplementing with vitamin D, but it could also be because people who take supplements tend to do other healthy things — another correlation to add to the great cathedral.
To get a better view of the real effects of randomization, the authors restricted the analysis to just those who did not use outside supplements. If vitamin D supplements help, then these are the people they should help. This group had about a 11% reduction in the incidence of cancer — statistically significant — and a 7% reduction in cancer mortality that did not meet the bar for statistical significance.
There was no increase in cardiovascular disease among this group. But this small effect on cancer was nowhere near enough to significantly reduce the rate of all-cause mortality.
Among those using supplements, vitamin D supplementation didn’t really move the needle on any outcome.
I know what you’re thinking: How many of these women were vitamin D deficient when we got started? These results may simply be telling us that people who have normal vitamin D levels are fine to go without supplementation.
Nearly three fourths of women who were not taking supplements entered the trial with vitamin D levels below the 50 nmol/L cutoff that the authors suggest would qualify for deficiency. Around half of those who used supplements were deficient. And yet, frustratingly, I could not find data on the effect of randomization to supplementation stratified by baseline vitamin D level. I even reached out to Dr Thomson to ask about this. She replied, “We did not stratify on baseline values because the numbers are too small statistically to test this.” Sorry.
In the meantime, I can tell you that for your “average woman,” vitamin D supplementation likely has no effect on mortality. It might modestly reduce the risk for certain cancers while increasing the risk for heart disease (probably through coronary calcification). So, there might be some room for personalization here. Perhaps women with a strong family history of cancer or other risk factors would do better with supplements, and those with a high risk for heart disease would do worse. Seems like a strategy that could be tested in a clinical trial. But maybe we could ask the participants to give up their extracurricular supplement use before they enter the trial. F. Perry Wilson, MD, MSCE, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and his book, How Medicine Works and When It Doesn’t, is available now.
Long-Term Calcium and Vitamin D: Cancer Deaths Down, CVD Deaths Up in Older Women?
Some doctors may be scratching their heads over a new analysis reporting that combined calcium and vitamin D (CaD) supplements appear to be associated with a slight 6% increase in cardiovascular (CVD) mortality, a slight 7% decrease in cancer risk, and no effect on osteoporotic fracture in postmenopausal women.
The study, in Annals of Internal Medicine, found no effect of supplementation on all-cause mortality.
The findings emerged from an analysis of more than 20 years’ follow-up data on a randomized trial in postmenopausal women conducted as part of the Women’s Health Initiative (WHI).
Cynthia A. Thomson, PhD, RD, first author and cancer prevention scientist at the Arizona Cancer Center and a professor of health promotion sciences at the University of Arizona in Tucson said the findings recommend individualized assessment of the need for supplements for older women as they consider them in hopes of preventing fractures.
“Evaluate your patients individually and understand that there are some who may benefit from supplementation, for example, in terms of reducing colorectal cancer mortality,” Dr. Thomson said in an interview. The approach should be nuanced. “If you check the adequacy of vitamin D and calcium in their diets, supplementation may not be needed.” She added that supplementation is best considered in the context of a woman’s overall health profile, including risk factors for fracture, heart disease, and cancer, especially colorectal cancer (CRC).
Study Details
The investigators conducted postintervention follow-up of the WHI’s 7-year multicenter randomized intervention trial of CaD vs placebo.
Since existing evidence of long-term health outcomes was limited, the trial, begun in 1999 and closed in 2005, enrolled 36,282 postmenopausal women (mean age 62) with no history of breast or colorectal cancer. They were randomly assigned 1:1 to supplementation with 1000 mg of calcium carbonate (400 mg elemental calcium) plus 400 IU of vitamin D3 daily or placebo, taken twice daily in half doses.
Study outcomes were incidence of CRC, total and invasive breast cancer; disease-specific and all-cause mortality; total CVD; and hip fracture measured through December 2020, with analyses stratified by personal supplement usage.
Cancer. CaD was associated with reduced incident total cancer, CRC, and invasive breast cancer — notably among participants not taking CaD before randomization. Cancer incidence estimates varied widely, the authors noted, when stratified by supplement use before randomization. Noting that CaD seemed to have more cancer-related impact in those without prior supplementation, the authors suggested supplementation may affect cancer biology primarily by augmenting nutrient insufficiency.
An estimated 7% reduction in cancer mortality was observed after a median cumulative follow-up of 22.3 years: 1817 vs 1943 deaths (hazard ratio, 0.93; 95% CI, 0.87-0.99).
CVD. An estimated 6% increase in CVD mortality was seen in the CaD group: 2621 vs 2420 deaths (HR, 1.06; 95% CI, 1.01-1.12). Pretrial supplement users were found to be at higher CVD risk.
Hip fracture. No effect on hip fracture risk was measured, but the authors cautioned that hip fracture and CVD outcomes were available only for a subset of participants, and the effects of calcium alone vs vitamin D alone vs the combination could not be disentangled.
In a small subgroup analysis, some CaD users were seen to respond in terms of bone mineral density but since only 4 of the study’s 40 sites collected such information, the study was underpowered to examine the effect. ”Many other studies, however, show a response to supplementation in women who already have bone mineral deficits,” Dr. Thomson said.
The Calcification Question
One of the possible mechanisms of harm is that high-dose calcium supplements can increase the rate of blood coagulation and promote vascular calcification, said Emma Laing, PhD, RD, director of dietetics at the University of Georgia in Athens and a spokesperson for the Chicago-based Academy of Nutrition and Dietetics.
“Other factors that should be considered when determining a patient’s CVD risk are race, genetic predisposition, medical and social history, response to stress, and lifestyle behaviors, as well as the length of time supplements have been consumed,” added Dr. Laing, who was not involved in the WHI analysis.
“We asked ourselves if CaD supplements might contribute to calcification of the coronary arteries, since some believe this to be the case, although the literature is mixed,” said Dr. Thomson.
“So we did a shorter ancillary study in a small sample of several hundred [women] to see if there was any increase in calcification” and no difference was seen on imaging across the two arms. “However, women who were already on supplements before entering the study seemed to be at higher CVD risk,” she said.
Added study coauthor JoAnn E. Manson, MD, DrPH, chief of the division of preventive medicine at Brigham and Women’s Hospital and professor of women’s health at Harvard Medical School, both in Boston: “With no increase or decrease in coronary artery calcium at the end of the trial, we don’t believe starting or continuing calcium/vitamin D supplements should require screening for coronary artery disease.”
Some randomized trials and systematic reviews, however, have observed an increased risk of CVD in healthy patients on calcium supplements, with one Korean meta-analysis reporting a 15% increase in CVD risk in healthy postmenopausal women taking calcium supplements. Another meta-analysis found a link between calcium supplements and a greater risk of various cardiovascular outcomes, especially myocardial infarction.
Vitamin D Supplementation
As for vitamin D only supplementation, an updated meta-analysis including more than 83,000 individuals showed that it confers no cardiovascular protection and is therefore not indicated for this purpose.
Practice Considerations
Offering an outsider’s perspective, Sarah G. Candler, MD, MPH, an internist in Houston specializing in primary care for older high-risk adults, said: “Unfortunately, this latest study continues the trend of creating more questions than answers. If the adverse outcome of CVD death is a result of supplementation, it is unclear if this is due to the vitamin D, the calcium, or both. And it is unclear if this is dose dependent, time dependent, or due to concurrent risk factors unique to certain populations.
“It is recommended that patients at risk of osteoporosis based on age, sex, medications, and lifestyle be screened for osteoporosis and treated accordingly, including supplementation with CaD,” Dr. Candler said. “It remains unclear whether supplementation with CaD in the absence of osteoporosis and osteopenia is net beneficial or harmful, and at this time I would not recommend it to my patients.”
Added Dr. Manson: “The very small increase seen in cardiovascular mortality wouldn’t be a reason to discontinue supplementation among women who have been advised by their healthcare providers to take these supplements for bone health or other purposes.
“Among those at usual risk of fracture, we recommend trying to obtain adequate calcium and vitamin D from food sources first and to use supplements only for the purpose of filling gaps in intake,” Dr. Manson continued. Overall, the findings support the national recommended dietary allowances for daily calcium intake of 1200 mg and daily vitamin D intake of 600-800 IU among postmenopausal women for maintenance of bone health, she said.
While a 2022 study found that vitamin D supplementation alone did not prevent fractures in healthy adults, other research has shown that a calcium/vitamin D combination is more likely to protect the skeleton.
“Patients at risk for fractures will probably benefit from calcium and/or vitamin D supplementation if they do not meet dietary intake requirements, have malabsorption syndromes, are taking medications that affect nutrient absorption, or if they are older and not regularly exposed to sunlight,” said Dr. Laing. “A combination of biochemical, imaging, functional, and dietary intake data can help determine if a supplement is warranted.”
She stressed that additional research is needed in more diverse populations before changing practice guidelines. “However, doctors should continue to weigh the risks and benefits of prescribing supplements for each patient.”
The WHI program is funded by the National Heart, Lung, and Blood Institute. Dr. Thomson disclosed no competing interests. Dr. Manson reported a relationship with Mars Edge. Multiple authors reported grant support from government funding agencies. The outside commentators had no relevant competing interests to disclose.
Some doctors may be scratching their heads over a new analysis reporting that combined calcium and vitamin D (CaD) supplements appear to be associated with a slight 6% increase in cardiovascular (CVD) mortality, a slight 7% decrease in cancer risk, and no effect on osteoporotic fracture in postmenopausal women.
The study, in Annals of Internal Medicine, found no effect of supplementation on all-cause mortality.
The findings emerged from an analysis of more than 20 years’ follow-up data on a randomized trial in postmenopausal women conducted as part of the Women’s Health Initiative (WHI).
Cynthia A. Thomson, PhD, RD, first author and cancer prevention scientist at the Arizona Cancer Center and a professor of health promotion sciences at the University of Arizona in Tucson said the findings recommend individualized assessment of the need for supplements for older women as they consider them in hopes of preventing fractures.
“Evaluate your patients individually and understand that there are some who may benefit from supplementation, for example, in terms of reducing colorectal cancer mortality,” Dr. Thomson said in an interview. The approach should be nuanced. “If you check the adequacy of vitamin D and calcium in their diets, supplementation may not be needed.” She added that supplementation is best considered in the context of a woman’s overall health profile, including risk factors for fracture, heart disease, and cancer, especially colorectal cancer (CRC).
Study Details
The investigators conducted postintervention follow-up of the WHI’s 7-year multicenter randomized intervention trial of CaD vs placebo.
Since existing evidence of long-term health outcomes was limited, the trial, begun in 1999 and closed in 2005, enrolled 36,282 postmenopausal women (mean age 62) with no history of breast or colorectal cancer. They were randomly assigned 1:1 to supplementation with 1000 mg of calcium carbonate (400 mg elemental calcium) plus 400 IU of vitamin D3 daily or placebo, taken twice daily in half doses.
Study outcomes were incidence of CRC, total and invasive breast cancer; disease-specific and all-cause mortality; total CVD; and hip fracture measured through December 2020, with analyses stratified by personal supplement usage.
Cancer. CaD was associated with reduced incident total cancer, CRC, and invasive breast cancer — notably among participants not taking CaD before randomization. Cancer incidence estimates varied widely, the authors noted, when stratified by supplement use before randomization. Noting that CaD seemed to have more cancer-related impact in those without prior supplementation, the authors suggested supplementation may affect cancer biology primarily by augmenting nutrient insufficiency.
An estimated 7% reduction in cancer mortality was observed after a median cumulative follow-up of 22.3 years: 1817 vs 1943 deaths (hazard ratio, 0.93; 95% CI, 0.87-0.99).
CVD. An estimated 6% increase in CVD mortality was seen in the CaD group: 2621 vs 2420 deaths (HR, 1.06; 95% CI, 1.01-1.12). Pretrial supplement users were found to be at higher CVD risk.
Hip fracture. No effect on hip fracture risk was measured, but the authors cautioned that hip fracture and CVD outcomes were available only for a subset of participants, and the effects of calcium alone vs vitamin D alone vs the combination could not be disentangled.
In a small subgroup analysis, some CaD users were seen to respond in terms of bone mineral density but since only 4 of the study’s 40 sites collected such information, the study was underpowered to examine the effect. ”Many other studies, however, show a response to supplementation in women who already have bone mineral deficits,” Dr. Thomson said.
The Calcification Question
One of the possible mechanisms of harm is that high-dose calcium supplements can increase the rate of blood coagulation and promote vascular calcification, said Emma Laing, PhD, RD, director of dietetics at the University of Georgia in Athens and a spokesperson for the Chicago-based Academy of Nutrition and Dietetics.
“Other factors that should be considered when determining a patient’s CVD risk are race, genetic predisposition, medical and social history, response to stress, and lifestyle behaviors, as well as the length of time supplements have been consumed,” added Dr. Laing, who was not involved in the WHI analysis.
“We asked ourselves if CaD supplements might contribute to calcification of the coronary arteries, since some believe this to be the case, although the literature is mixed,” said Dr. Thomson.
“So we did a shorter ancillary study in a small sample of several hundred [women] to see if there was any increase in calcification” and no difference was seen on imaging across the two arms. “However, women who were already on supplements before entering the study seemed to be at higher CVD risk,” she said.
Added study coauthor JoAnn E. Manson, MD, DrPH, chief of the division of preventive medicine at Brigham and Women’s Hospital and professor of women’s health at Harvard Medical School, both in Boston: “With no increase or decrease in coronary artery calcium at the end of the trial, we don’t believe starting or continuing calcium/vitamin D supplements should require screening for coronary artery disease.”
Some randomized trials and systematic reviews, however, have observed an increased risk of CVD in healthy patients on calcium supplements, with one Korean meta-analysis reporting a 15% increase in CVD risk in healthy postmenopausal women taking calcium supplements. Another meta-analysis found a link between calcium supplements and a greater risk of various cardiovascular outcomes, especially myocardial infarction.
Vitamin D Supplementation
As for vitamin D only supplementation, an updated meta-analysis including more than 83,000 individuals showed that it confers no cardiovascular protection and is therefore not indicated for this purpose.
Practice Considerations
Offering an outsider’s perspective, Sarah G. Candler, MD, MPH, an internist in Houston specializing in primary care for older high-risk adults, said: “Unfortunately, this latest study continues the trend of creating more questions than answers. If the adverse outcome of CVD death is a result of supplementation, it is unclear if this is due to the vitamin D, the calcium, or both. And it is unclear if this is dose dependent, time dependent, or due to concurrent risk factors unique to certain populations.
“It is recommended that patients at risk of osteoporosis based on age, sex, medications, and lifestyle be screened for osteoporosis and treated accordingly, including supplementation with CaD,” Dr. Candler said. “It remains unclear whether supplementation with CaD in the absence of osteoporosis and osteopenia is net beneficial or harmful, and at this time I would not recommend it to my patients.”
Added Dr. Manson: “The very small increase seen in cardiovascular mortality wouldn’t be a reason to discontinue supplementation among women who have been advised by their healthcare providers to take these supplements for bone health or other purposes.
“Among those at usual risk of fracture, we recommend trying to obtain adequate calcium and vitamin D from food sources first and to use supplements only for the purpose of filling gaps in intake,” Dr. Manson continued. Overall, the findings support the national recommended dietary allowances for daily calcium intake of 1200 mg and daily vitamin D intake of 600-800 IU among postmenopausal women for maintenance of bone health, she said.
While a 2022 study found that vitamin D supplementation alone did not prevent fractures in healthy adults, other research has shown that a calcium/vitamin D combination is more likely to protect the skeleton.
“Patients at risk for fractures will probably benefit from calcium and/or vitamin D supplementation if they do not meet dietary intake requirements, have malabsorption syndromes, are taking medications that affect nutrient absorption, or if they are older and not regularly exposed to sunlight,” said Dr. Laing. “A combination of biochemical, imaging, functional, and dietary intake data can help determine if a supplement is warranted.”
She stressed that additional research is needed in more diverse populations before changing practice guidelines. “However, doctors should continue to weigh the risks and benefits of prescribing supplements for each patient.”
The WHI program is funded by the National Heart, Lung, and Blood Institute. Dr. Thomson disclosed no competing interests. Dr. Manson reported a relationship with Mars Edge. Multiple authors reported grant support from government funding agencies. The outside commentators had no relevant competing interests to disclose.
Some doctors may be scratching their heads over a new analysis reporting that combined calcium and vitamin D (CaD) supplements appear to be associated with a slight 6% increase in cardiovascular (CVD) mortality, a slight 7% decrease in cancer risk, and no effect on osteoporotic fracture in postmenopausal women.
The study, in Annals of Internal Medicine, found no effect of supplementation on all-cause mortality.
The findings emerged from an analysis of more than 20 years’ follow-up data on a randomized trial in postmenopausal women conducted as part of the Women’s Health Initiative (WHI).
Cynthia A. Thomson, PhD, RD, first author and cancer prevention scientist at the Arizona Cancer Center and a professor of health promotion sciences at the University of Arizona in Tucson said the findings recommend individualized assessment of the need for supplements for older women as they consider them in hopes of preventing fractures.
“Evaluate your patients individually and understand that there are some who may benefit from supplementation, for example, in terms of reducing colorectal cancer mortality,” Dr. Thomson said in an interview. The approach should be nuanced. “If you check the adequacy of vitamin D and calcium in their diets, supplementation may not be needed.” She added that supplementation is best considered in the context of a woman’s overall health profile, including risk factors for fracture, heart disease, and cancer, especially colorectal cancer (CRC).
Study Details
The investigators conducted postintervention follow-up of the WHI’s 7-year multicenter randomized intervention trial of CaD vs placebo.
Since existing evidence of long-term health outcomes was limited, the trial, begun in 1999 and closed in 2005, enrolled 36,282 postmenopausal women (mean age 62) with no history of breast or colorectal cancer. They were randomly assigned 1:1 to supplementation with 1000 mg of calcium carbonate (400 mg elemental calcium) plus 400 IU of vitamin D3 daily or placebo, taken twice daily in half doses.
Study outcomes were incidence of CRC, total and invasive breast cancer; disease-specific and all-cause mortality; total CVD; and hip fracture measured through December 2020, with analyses stratified by personal supplement usage.
Cancer. CaD was associated with reduced incident total cancer, CRC, and invasive breast cancer — notably among participants not taking CaD before randomization. Cancer incidence estimates varied widely, the authors noted, when stratified by supplement use before randomization. Noting that CaD seemed to have more cancer-related impact in those without prior supplementation, the authors suggested supplementation may affect cancer biology primarily by augmenting nutrient insufficiency.
An estimated 7% reduction in cancer mortality was observed after a median cumulative follow-up of 22.3 years: 1817 vs 1943 deaths (hazard ratio, 0.93; 95% CI, 0.87-0.99).
CVD. An estimated 6% increase in CVD mortality was seen in the CaD group: 2621 vs 2420 deaths (HR, 1.06; 95% CI, 1.01-1.12). Pretrial supplement users were found to be at higher CVD risk.
Hip fracture. No effect on hip fracture risk was measured, but the authors cautioned that hip fracture and CVD outcomes were available only for a subset of participants, and the effects of calcium alone vs vitamin D alone vs the combination could not be disentangled.
In a small subgroup analysis, some CaD users were seen to respond in terms of bone mineral density but since only 4 of the study’s 40 sites collected such information, the study was underpowered to examine the effect. ”Many other studies, however, show a response to supplementation in women who already have bone mineral deficits,” Dr. Thomson said.
The Calcification Question
One of the possible mechanisms of harm is that high-dose calcium supplements can increase the rate of blood coagulation and promote vascular calcification, said Emma Laing, PhD, RD, director of dietetics at the University of Georgia in Athens and a spokesperson for the Chicago-based Academy of Nutrition and Dietetics.
“Other factors that should be considered when determining a patient’s CVD risk are race, genetic predisposition, medical and social history, response to stress, and lifestyle behaviors, as well as the length of time supplements have been consumed,” added Dr. Laing, who was not involved in the WHI analysis.
“We asked ourselves if CaD supplements might contribute to calcification of the coronary arteries, since some believe this to be the case, although the literature is mixed,” said Dr. Thomson.
“So we did a shorter ancillary study in a small sample of several hundred [women] to see if there was any increase in calcification” and no difference was seen on imaging across the two arms. “However, women who were already on supplements before entering the study seemed to be at higher CVD risk,” she said.
Added study coauthor JoAnn E. Manson, MD, DrPH, chief of the division of preventive medicine at Brigham and Women’s Hospital and professor of women’s health at Harvard Medical School, both in Boston: “With no increase or decrease in coronary artery calcium at the end of the trial, we don’t believe starting or continuing calcium/vitamin D supplements should require screening for coronary artery disease.”
Some randomized trials and systematic reviews, however, have observed an increased risk of CVD in healthy patients on calcium supplements, with one Korean meta-analysis reporting a 15% increase in CVD risk in healthy postmenopausal women taking calcium supplements. Another meta-analysis found a link between calcium supplements and a greater risk of various cardiovascular outcomes, especially myocardial infarction.
Vitamin D Supplementation
As for vitamin D only supplementation, an updated meta-analysis including more than 83,000 individuals showed that it confers no cardiovascular protection and is therefore not indicated for this purpose.
Practice Considerations
Offering an outsider’s perspective, Sarah G. Candler, MD, MPH, an internist in Houston specializing in primary care for older high-risk adults, said: “Unfortunately, this latest study continues the trend of creating more questions than answers. If the adverse outcome of CVD death is a result of supplementation, it is unclear if this is due to the vitamin D, the calcium, or both. And it is unclear if this is dose dependent, time dependent, or due to concurrent risk factors unique to certain populations.
“It is recommended that patients at risk of osteoporosis based on age, sex, medications, and lifestyle be screened for osteoporosis and treated accordingly, including supplementation with CaD,” Dr. Candler said. “It remains unclear whether supplementation with CaD in the absence of osteoporosis and osteopenia is net beneficial or harmful, and at this time I would not recommend it to my patients.”
Added Dr. Manson: “The very small increase seen in cardiovascular mortality wouldn’t be a reason to discontinue supplementation among women who have been advised by their healthcare providers to take these supplements for bone health or other purposes.
“Among those at usual risk of fracture, we recommend trying to obtain adequate calcium and vitamin D from food sources first and to use supplements only for the purpose of filling gaps in intake,” Dr. Manson continued. Overall, the findings support the national recommended dietary allowances for daily calcium intake of 1200 mg and daily vitamin D intake of 600-800 IU among postmenopausal women for maintenance of bone health, she said.
While a 2022 study found that vitamin D supplementation alone did not prevent fractures in healthy adults, other research has shown that a calcium/vitamin D combination is more likely to protect the skeleton.
“Patients at risk for fractures will probably benefit from calcium and/or vitamin D supplementation if they do not meet dietary intake requirements, have malabsorption syndromes, are taking medications that affect nutrient absorption, or if they are older and not regularly exposed to sunlight,” said Dr. Laing. “A combination of biochemical, imaging, functional, and dietary intake data can help determine if a supplement is warranted.”
She stressed that additional research is needed in more diverse populations before changing practice guidelines. “However, doctors should continue to weigh the risks and benefits of prescribing supplements for each patient.”
The WHI program is funded by the National Heart, Lung, and Blood Institute. Dr. Thomson disclosed no competing interests. Dr. Manson reported a relationship with Mars Edge. Multiple authors reported grant support from government funding agencies. The outside commentators had no relevant competing interests to disclose.
FROM ANNALS OF INTERNAL MEDICINE
ASTRO Pushes Return to Direct Supervision in RT: Needed or ‘Babysitting’?
Although serious errors during virtual supervision are rare, ASTRO said radiation treatments (RT) should be done with a radiation oncologist on site to ensure high-quality care. But some radiation oncologists do not agree with the proposal to move back to direct in-person supervision only.
Changes to Direct Supervision
Most radiation oncology treatments are delivered in an outpatient setting under a physician’s direction and control.
During the COVID-19 pandemic when social distancing mandates were in place, CMS temporarily changed the definition of “direct supervision” to include telehealth, specifying that a physician must be immediately available to assist and direct a procedure virtually using real-time audio and video. In other words, a physician did not need to be physically present in the room when the treatment was being performed.
CMS has extended this rule until the end of 2024 and is considering making it a permanent change. In the Calendar Year (CY) 2024 Medicare Physician Fee Schedule (PFS) Final Rule, CMS asked for comments on whether to extend the rule.
“We received input from interested parties on potential patient safety or quality concerns when direct supervision occurs virtually, which we will consider for future rulemaking,” a CMS spokesperson told this news organization. “CMS is currently considering the best approach that will protect patient access and safety as well as quality of care and program integrity concerns following CY 2024.”
CMS also noted its concerns that an abrupt transition back to requiring a physician’s physical presence could interrupt care from practitioners who have established new patterns of practice with telehealth.
What Are ASTRO’s Concerns?
Late last month, ASTRO sent CMS a letter, asking the agency to change the rules back to direct in-person supervision for all radiation services, citing that virtual supervision jeopardizes patient safety and quality of care.
Jeff Michalski, MD, MBA, chair of the ASTRO Board of Directors, said in an interview that radiation oncologists should be physically present to supervise the treatments.
“ASTRO is concerned that blanket policies of general or virtual supervision could lead to patients not having direct, in-person access to their doctors’ care,” he said. “While serious errors are rare, real-world experiences of radiation oncologists across practice settings demonstrate how an in-person radiation oncology physician is best suited to ensure high-quality care.”
What Do Radiation Oncologists Think?
According to ASTRO, most radiation oncologists would agree that in-person supervision is best for patients.
But that might not be the case.
Radiation oncologists took to X (formerly Twitter) to voice their opinions about ASTRO’s letter.
Jason Beckta, MD, PhD, of Rutland Regional’s Foley Cancer Center, Vermont, said “the February 26th ASTRO letter reads like an Onion article.”
“I’m struggling to understand the Luddite-level myopia around this topic,” he said in another tweet. “Virtual direct/outpatient general supervision has done nothing but boost my productivity and in particular, face-to-face patient contact.”
Join Y. Luh, MD, with the Providence Medical Network in Eureka, California, said he understands the challenges faced by clinicians working in more isolated rural settings. “For them, it’s either having virtual supervision or closing the center,” Dr. Luh said.
“Virtual care is definitely at my clinic and is not only an option but is critical to my patients who are 2+ snowy, mountainous hours away,” Dr. Luh wrote. “But I’m still in the clinic directly supervising treatments.”
Sidney Roberts, MD, with the CHI St. Luke’s Health-Memorial, Texas, tweeted that supervision does require some face-to-face care but contended that “babysitting trained therapists for every routine treatment is a farce.”
Another issue Dr. Luh brought up is reimbursement for virtual supervision, noting that “the elephant in the room is whether that level of service should be reimbursed at the same rate. Reimbursement has not changed — but will it stay that way?”
ASTRO has acknowledged that radiation oncologists will have varying opinions and says it is working to balance these challenges.
CMS has not reached a decision on whether the change will be implemented permanently. The organization will assess concern, patient safety, and quality of care at the end of the year.
A version of this article first appeared on Medscape.com
Although serious errors during virtual supervision are rare, ASTRO said radiation treatments (RT) should be done with a radiation oncologist on site to ensure high-quality care. But some radiation oncologists do not agree with the proposal to move back to direct in-person supervision only.
Changes to Direct Supervision
Most radiation oncology treatments are delivered in an outpatient setting under a physician’s direction and control.
During the COVID-19 pandemic when social distancing mandates were in place, CMS temporarily changed the definition of “direct supervision” to include telehealth, specifying that a physician must be immediately available to assist and direct a procedure virtually using real-time audio and video. In other words, a physician did not need to be physically present in the room when the treatment was being performed.
CMS has extended this rule until the end of 2024 and is considering making it a permanent change. In the Calendar Year (CY) 2024 Medicare Physician Fee Schedule (PFS) Final Rule, CMS asked for comments on whether to extend the rule.
“We received input from interested parties on potential patient safety or quality concerns when direct supervision occurs virtually, which we will consider for future rulemaking,” a CMS spokesperson told this news organization. “CMS is currently considering the best approach that will protect patient access and safety as well as quality of care and program integrity concerns following CY 2024.”
CMS also noted its concerns that an abrupt transition back to requiring a physician’s physical presence could interrupt care from practitioners who have established new patterns of practice with telehealth.
What Are ASTRO’s Concerns?
Late last month, ASTRO sent CMS a letter, asking the agency to change the rules back to direct in-person supervision for all radiation services, citing that virtual supervision jeopardizes patient safety and quality of care.
Jeff Michalski, MD, MBA, chair of the ASTRO Board of Directors, said in an interview that radiation oncologists should be physically present to supervise the treatments.
“ASTRO is concerned that blanket policies of general or virtual supervision could lead to patients not having direct, in-person access to their doctors’ care,” he said. “While serious errors are rare, real-world experiences of radiation oncologists across practice settings demonstrate how an in-person radiation oncology physician is best suited to ensure high-quality care.”
What Do Radiation Oncologists Think?
According to ASTRO, most radiation oncologists would agree that in-person supervision is best for patients.
But that might not be the case.
Radiation oncologists took to X (formerly Twitter) to voice their opinions about ASTRO’s letter.
Jason Beckta, MD, PhD, of Rutland Regional’s Foley Cancer Center, Vermont, said “the February 26th ASTRO letter reads like an Onion article.”
“I’m struggling to understand the Luddite-level myopia around this topic,” he said in another tweet. “Virtual direct/outpatient general supervision has done nothing but boost my productivity and in particular, face-to-face patient contact.”
Join Y. Luh, MD, with the Providence Medical Network in Eureka, California, said he understands the challenges faced by clinicians working in more isolated rural settings. “For them, it’s either having virtual supervision or closing the center,” Dr. Luh said.
“Virtual care is definitely at my clinic and is not only an option but is critical to my patients who are 2+ snowy, mountainous hours away,” Dr. Luh wrote. “But I’m still in the clinic directly supervising treatments.”
Sidney Roberts, MD, with the CHI St. Luke’s Health-Memorial, Texas, tweeted that supervision does require some face-to-face care but contended that “babysitting trained therapists for every routine treatment is a farce.”
Another issue Dr. Luh brought up is reimbursement for virtual supervision, noting that “the elephant in the room is whether that level of service should be reimbursed at the same rate. Reimbursement has not changed — but will it stay that way?”
ASTRO has acknowledged that radiation oncologists will have varying opinions and says it is working to balance these challenges.
CMS has not reached a decision on whether the change will be implemented permanently. The organization will assess concern, patient safety, and quality of care at the end of the year.
A version of this article first appeared on Medscape.com
Although serious errors during virtual supervision are rare, ASTRO said radiation treatments (RT) should be done with a radiation oncologist on site to ensure high-quality care. But some radiation oncologists do not agree with the proposal to move back to direct in-person supervision only.
Changes to Direct Supervision
Most radiation oncology treatments are delivered in an outpatient setting under a physician’s direction and control.
During the COVID-19 pandemic when social distancing mandates were in place, CMS temporarily changed the definition of “direct supervision” to include telehealth, specifying that a physician must be immediately available to assist and direct a procedure virtually using real-time audio and video. In other words, a physician did not need to be physically present in the room when the treatment was being performed.
CMS has extended this rule until the end of 2024 and is considering making it a permanent change. In the Calendar Year (CY) 2024 Medicare Physician Fee Schedule (PFS) Final Rule, CMS asked for comments on whether to extend the rule.
“We received input from interested parties on potential patient safety or quality concerns when direct supervision occurs virtually, which we will consider for future rulemaking,” a CMS spokesperson told this news organization. “CMS is currently considering the best approach that will protect patient access and safety as well as quality of care and program integrity concerns following CY 2024.”
CMS also noted its concerns that an abrupt transition back to requiring a physician’s physical presence could interrupt care from practitioners who have established new patterns of practice with telehealth.
What Are ASTRO’s Concerns?
Late last month, ASTRO sent CMS a letter, asking the agency to change the rules back to direct in-person supervision for all radiation services, citing that virtual supervision jeopardizes patient safety and quality of care.
Jeff Michalski, MD, MBA, chair of the ASTRO Board of Directors, said in an interview that radiation oncologists should be physically present to supervise the treatments.
“ASTRO is concerned that blanket policies of general or virtual supervision could lead to patients not having direct, in-person access to their doctors’ care,” he said. “While serious errors are rare, real-world experiences of radiation oncologists across practice settings demonstrate how an in-person radiation oncology physician is best suited to ensure high-quality care.”
What Do Radiation Oncologists Think?
According to ASTRO, most radiation oncologists would agree that in-person supervision is best for patients.
But that might not be the case.
Radiation oncologists took to X (formerly Twitter) to voice their opinions about ASTRO’s letter.
Jason Beckta, MD, PhD, of Rutland Regional’s Foley Cancer Center, Vermont, said “the February 26th ASTRO letter reads like an Onion article.”
“I’m struggling to understand the Luddite-level myopia around this topic,” he said in another tweet. “Virtual direct/outpatient general supervision has done nothing but boost my productivity and in particular, face-to-face patient contact.”
Join Y. Luh, MD, with the Providence Medical Network in Eureka, California, said he understands the challenges faced by clinicians working in more isolated rural settings. “For them, it’s either having virtual supervision or closing the center,” Dr. Luh said.
“Virtual care is definitely at my clinic and is not only an option but is critical to my patients who are 2+ snowy, mountainous hours away,” Dr. Luh wrote. “But I’m still in the clinic directly supervising treatments.”
Sidney Roberts, MD, with the CHI St. Luke’s Health-Memorial, Texas, tweeted that supervision does require some face-to-face care but contended that “babysitting trained therapists for every routine treatment is a farce.”
Another issue Dr. Luh brought up is reimbursement for virtual supervision, noting that “the elephant in the room is whether that level of service should be reimbursed at the same rate. Reimbursement has not changed — but will it stay that way?”
ASTRO has acknowledged that radiation oncologists will have varying opinions and says it is working to balance these challenges.
CMS has not reached a decision on whether the change will be implemented permanently. The organization will assess concern, patient safety, and quality of care at the end of the year.
A version of this article first appeared on Medscape.com
Skin Infections in Pregnant Women: Many Drugs Safe, but Not All
SAN DIEGO — . However, several drugs should be avoided or used with caution because of potential risks during pregnancy.
When treating bacterial infections in pregnant women, there are many options, “especially for the sort of short-term antibiotic use that we tend to use for treating infections,” said Jenny Murase, MD, of the Palo Alto Foundation Medical Group and the University of California San Francisco.
During a presentation on treating infections in pregnant patients, she made the following recommendations for treating pyogenic infections:
- Impetigo: First-line treatments are topical mupirocin, oral first-generation cephalosporins, and oral dicloxacillin.
- Cellulitis: Recommended treatments are oral or intravenous penicillin, oral first-generation cephalosporins, and oral dicloxacillin.
- Methicillin-resistant Staphylococcus aureus (MRSA): “Clindamycin is first-line, dependent on bacteria culture and sensitivities,” and because of its safety, “it’s a really good choice for a pregnant woman.” Dr. Murase said. However, be aware of potential inducible resistance and test for the erm gene, she said.
- Abscesses: Incision and drainage are recommended. “Whenever we’re managing a patient with a condition during pregnancy, we want to try to use nonmedications when possible,” Dr. Murase said. “No antibiotic is necessary unless the abscess is greater than 5 cm or if it’s greater than 2 cm with erythema around the abscess.”
- Tuberculosis: The best strategy is rifampin, but peripartum vitamin K prophylaxis for mother and fetus should be used, she said.
General Infections
With regard to antibiotics to treat general infections — for instance, if a patient with atopic dermatitis has a secondary skin infection — Dr. Murase recommended first-line oral antibiotic therapy with penicillin, first-generation cephalosporins, or dicloxacillin. For second-line therapy, erythromycin is the preferred macrolide over azithromycin and clarithromycin, she said.
She noted that there is an increased risk for atrial/ventricular septal defects and pyloric stenosis associated with the use of erythromycin when used during the first trimester of pregnancy. In addition, erythromycin estolate increases the risk of liver toxicity, while erythromycin base and erythromycin ethylsuccinate do not.
Sulfonamides are a second-line line choice up until the third trimester. If given to a patient in the first trimester, she said, “make sure that they are supplementing with folic acid efficiently, at least 0.5 mg a day.” During the peripartum period they are contraindicated, as they pose a risk for hemolytic anemia, hyperbilirubinemia, and kernicterus.
The combination drug trimethoprim/sulfamethoxazole is a second-line choice for complicated infections because of the associated risk for low birth weight and prematurity, Dr. Murase said.
Quinolones are also a second-line option during pregnancy she said, and ciprofloxacin and norfloxacin have been studied the most. “If you have to choose a quinolone for a complicated infection in pregnancy, those would be the quinolones of choice,” Dr. Murase said.
Considering the bad reputation of tetracyclines in pregnancy, dermatologists may be surprised to learn that they are considered a second-line therapy up to 14 weeks’ gestation, she said. After that time, however, they’re contraindicated because of bone growth inhibition, teeth discoloration, and maternal hepatitis.
Fungal Infections
As for fungal infections, clotrimazole is the first choice for topical treatment of tinea corporis, followed by miconazole and then ketoconazole, according to Dr. Murase. There are limited data for topical terbinafine, naftifine, and ciclopirox during pregnancy she noted, but they are likely safe.
There is also limited data about these drugs when used for topical treatment of candidiasis during pregnancy. Nystatin is safe, but less effective than other options, Dr. Murase said. Other options include clotrimazole, miconazole, and ketoconazole, which, in animals exposed to high doses, have not been associated with defects, and topical gentian violet (0.5%-1% solution), she noted.
For topical treatment of tinea versicolor during pregnancy, limited application of clotrimazole or miconazole is considered safe, and zinc pyrithione soap or topical benzoyl peroxide soap can be used for more widespread areas.
Dr. Murase recommended caution when using selenium sulfide since poisoning has been linked to miscarriages, she said. Limited application appears to be safe, “so make sure that the patient is using it on smaller body surface areas.”
As for systemic antifungal treatments, fluconazole, ketoconazole, and itraconazole should be avoided in pregnancy because of the risks of craniosynostosis, congenital heart defects, and skeletal anomalies, Dr. Murase said. However, she referred to a study that found no increased risk of congenital malformations with fluconazole during the first trimester, and a patient could be reassured if, for example, she was treated for a yeast infection before she knew she was pregnant, she said.
Griseofulvin is not recommended during pregnancy, but a 2020 study suggests that terbinafine is safe, she said. In that study, oral or topical terbinafine did not appear to be associated with an increased risk for spontaneous abortion or major malformations. “Certainly, we can wait until after the pregnancy to treat onychomycosis. But I have had situations that even in spite of regular topical therapy, pregnant patients needed to take some kind of oral agent” because of severe itching.
Viral Infections
For herpes simplex, acyclovir is the top choice, and famciclovir and valacyclovir (Valtrex) are likely safe, but daily prophylaxis is not recommended during pregnancy, Dr. Murase said.
Because of a lack of data, podofilox, cantharidin, and imiquimod for treating human papillomavirus (HPV) should be avoided, she said. Podophyllin is extremely dangerous in pregnancy and has been linked to maternal and fetal deaths, and malformations, and is contraindicated in pregnancy, she added.
Instead, liquid nitrogen is the treatment of choice for HPV in pregnant patients, she said.
Trichloracetic acid is the treatment of choice for condylomata acuminata, and squaric acid or intralesional Candida antigen injection for periungual verrucas can be used, she said, and limited applications of salicylic acid are considered safe.
Dr. Murase highlighted a 2014 paper that she coauthored on the safety of dermatologic medications during pregnancy, noting that an updated report will be published later this year.
Dr. Murase disclosed relationships with Regeneron and UCB (speaker), Sanofi/Regeneron and Bristol-Myers Squibb (advisory board), and UCB, AbbVie, and UpToDate (consulting).
A version of this article appeared on Medscape.com.
SAN DIEGO — . However, several drugs should be avoided or used with caution because of potential risks during pregnancy.
When treating bacterial infections in pregnant women, there are many options, “especially for the sort of short-term antibiotic use that we tend to use for treating infections,” said Jenny Murase, MD, of the Palo Alto Foundation Medical Group and the University of California San Francisco.
During a presentation on treating infections in pregnant patients, she made the following recommendations for treating pyogenic infections:
- Impetigo: First-line treatments are topical mupirocin, oral first-generation cephalosporins, and oral dicloxacillin.
- Cellulitis: Recommended treatments are oral or intravenous penicillin, oral first-generation cephalosporins, and oral dicloxacillin.
- Methicillin-resistant Staphylococcus aureus (MRSA): “Clindamycin is first-line, dependent on bacteria culture and sensitivities,” and because of its safety, “it’s a really good choice for a pregnant woman.” Dr. Murase said. However, be aware of potential inducible resistance and test for the erm gene, she said.
- Abscesses: Incision and drainage are recommended. “Whenever we’re managing a patient with a condition during pregnancy, we want to try to use nonmedications when possible,” Dr. Murase said. “No antibiotic is necessary unless the abscess is greater than 5 cm or if it’s greater than 2 cm with erythema around the abscess.”
- Tuberculosis: The best strategy is rifampin, but peripartum vitamin K prophylaxis for mother and fetus should be used, she said.
General Infections
With regard to antibiotics to treat general infections — for instance, if a patient with atopic dermatitis has a secondary skin infection — Dr. Murase recommended first-line oral antibiotic therapy with penicillin, first-generation cephalosporins, or dicloxacillin. For second-line therapy, erythromycin is the preferred macrolide over azithromycin and clarithromycin, she said.
She noted that there is an increased risk for atrial/ventricular septal defects and pyloric stenosis associated with the use of erythromycin when used during the first trimester of pregnancy. In addition, erythromycin estolate increases the risk of liver toxicity, while erythromycin base and erythromycin ethylsuccinate do not.
Sulfonamides are a second-line line choice up until the third trimester. If given to a patient in the first trimester, she said, “make sure that they are supplementing with folic acid efficiently, at least 0.5 mg a day.” During the peripartum period they are contraindicated, as they pose a risk for hemolytic anemia, hyperbilirubinemia, and kernicterus.
The combination drug trimethoprim/sulfamethoxazole is a second-line choice for complicated infections because of the associated risk for low birth weight and prematurity, Dr. Murase said.
Quinolones are also a second-line option during pregnancy she said, and ciprofloxacin and norfloxacin have been studied the most. “If you have to choose a quinolone for a complicated infection in pregnancy, those would be the quinolones of choice,” Dr. Murase said.
Considering the bad reputation of tetracyclines in pregnancy, dermatologists may be surprised to learn that they are considered a second-line therapy up to 14 weeks’ gestation, she said. After that time, however, they’re contraindicated because of bone growth inhibition, teeth discoloration, and maternal hepatitis.
Fungal Infections
As for fungal infections, clotrimazole is the first choice for topical treatment of tinea corporis, followed by miconazole and then ketoconazole, according to Dr. Murase. There are limited data for topical terbinafine, naftifine, and ciclopirox during pregnancy she noted, but they are likely safe.
There is also limited data about these drugs when used for topical treatment of candidiasis during pregnancy. Nystatin is safe, but less effective than other options, Dr. Murase said. Other options include clotrimazole, miconazole, and ketoconazole, which, in animals exposed to high doses, have not been associated with defects, and topical gentian violet (0.5%-1% solution), she noted.
For topical treatment of tinea versicolor during pregnancy, limited application of clotrimazole or miconazole is considered safe, and zinc pyrithione soap or topical benzoyl peroxide soap can be used for more widespread areas.
Dr. Murase recommended caution when using selenium sulfide since poisoning has been linked to miscarriages, she said. Limited application appears to be safe, “so make sure that the patient is using it on smaller body surface areas.”
As for systemic antifungal treatments, fluconazole, ketoconazole, and itraconazole should be avoided in pregnancy because of the risks of craniosynostosis, congenital heart defects, and skeletal anomalies, Dr. Murase said. However, she referred to a study that found no increased risk of congenital malformations with fluconazole during the first trimester, and a patient could be reassured if, for example, she was treated for a yeast infection before she knew she was pregnant, she said.
Griseofulvin is not recommended during pregnancy, but a 2020 study suggests that terbinafine is safe, she said. In that study, oral or topical terbinafine did not appear to be associated with an increased risk for spontaneous abortion or major malformations. “Certainly, we can wait until after the pregnancy to treat onychomycosis. But I have had situations that even in spite of regular topical therapy, pregnant patients needed to take some kind of oral agent” because of severe itching.
Viral Infections
For herpes simplex, acyclovir is the top choice, and famciclovir and valacyclovir (Valtrex) are likely safe, but daily prophylaxis is not recommended during pregnancy, Dr. Murase said.
Because of a lack of data, podofilox, cantharidin, and imiquimod for treating human papillomavirus (HPV) should be avoided, she said. Podophyllin is extremely dangerous in pregnancy and has been linked to maternal and fetal deaths, and malformations, and is contraindicated in pregnancy, she added.
Instead, liquid nitrogen is the treatment of choice for HPV in pregnant patients, she said.
Trichloracetic acid is the treatment of choice for condylomata acuminata, and squaric acid or intralesional Candida antigen injection for periungual verrucas can be used, she said, and limited applications of salicylic acid are considered safe.
Dr. Murase highlighted a 2014 paper that she coauthored on the safety of dermatologic medications during pregnancy, noting that an updated report will be published later this year.
Dr. Murase disclosed relationships with Regeneron and UCB (speaker), Sanofi/Regeneron and Bristol-Myers Squibb (advisory board), and UCB, AbbVie, and UpToDate (consulting).
A version of this article appeared on Medscape.com.
SAN DIEGO — . However, several drugs should be avoided or used with caution because of potential risks during pregnancy.
When treating bacterial infections in pregnant women, there are many options, “especially for the sort of short-term antibiotic use that we tend to use for treating infections,” said Jenny Murase, MD, of the Palo Alto Foundation Medical Group and the University of California San Francisco.
During a presentation on treating infections in pregnant patients, she made the following recommendations for treating pyogenic infections:
- Impetigo: First-line treatments are topical mupirocin, oral first-generation cephalosporins, and oral dicloxacillin.
- Cellulitis: Recommended treatments are oral or intravenous penicillin, oral first-generation cephalosporins, and oral dicloxacillin.
- Methicillin-resistant Staphylococcus aureus (MRSA): “Clindamycin is first-line, dependent on bacteria culture and sensitivities,” and because of its safety, “it’s a really good choice for a pregnant woman.” Dr. Murase said. However, be aware of potential inducible resistance and test for the erm gene, she said.
- Abscesses: Incision and drainage are recommended. “Whenever we’re managing a patient with a condition during pregnancy, we want to try to use nonmedications when possible,” Dr. Murase said. “No antibiotic is necessary unless the abscess is greater than 5 cm or if it’s greater than 2 cm with erythema around the abscess.”
- Tuberculosis: The best strategy is rifampin, but peripartum vitamin K prophylaxis for mother and fetus should be used, she said.
General Infections
With regard to antibiotics to treat general infections — for instance, if a patient with atopic dermatitis has a secondary skin infection — Dr. Murase recommended first-line oral antibiotic therapy with penicillin, first-generation cephalosporins, or dicloxacillin. For second-line therapy, erythromycin is the preferred macrolide over azithromycin and clarithromycin, she said.
She noted that there is an increased risk for atrial/ventricular septal defects and pyloric stenosis associated with the use of erythromycin when used during the first trimester of pregnancy. In addition, erythromycin estolate increases the risk of liver toxicity, while erythromycin base and erythromycin ethylsuccinate do not.
Sulfonamides are a second-line line choice up until the third trimester. If given to a patient in the first trimester, she said, “make sure that they are supplementing with folic acid efficiently, at least 0.5 mg a day.” During the peripartum period they are contraindicated, as they pose a risk for hemolytic anemia, hyperbilirubinemia, and kernicterus.
The combination drug trimethoprim/sulfamethoxazole is a second-line choice for complicated infections because of the associated risk for low birth weight and prematurity, Dr. Murase said.
Quinolones are also a second-line option during pregnancy she said, and ciprofloxacin and norfloxacin have been studied the most. “If you have to choose a quinolone for a complicated infection in pregnancy, those would be the quinolones of choice,” Dr. Murase said.
Considering the bad reputation of tetracyclines in pregnancy, dermatologists may be surprised to learn that they are considered a second-line therapy up to 14 weeks’ gestation, she said. After that time, however, they’re contraindicated because of bone growth inhibition, teeth discoloration, and maternal hepatitis.
Fungal Infections
As for fungal infections, clotrimazole is the first choice for topical treatment of tinea corporis, followed by miconazole and then ketoconazole, according to Dr. Murase. There are limited data for topical terbinafine, naftifine, and ciclopirox during pregnancy she noted, but they are likely safe.
There is also limited data about these drugs when used for topical treatment of candidiasis during pregnancy. Nystatin is safe, but less effective than other options, Dr. Murase said. Other options include clotrimazole, miconazole, and ketoconazole, which, in animals exposed to high doses, have not been associated with defects, and topical gentian violet (0.5%-1% solution), she noted.
For topical treatment of tinea versicolor during pregnancy, limited application of clotrimazole or miconazole is considered safe, and zinc pyrithione soap or topical benzoyl peroxide soap can be used for more widespread areas.
Dr. Murase recommended caution when using selenium sulfide since poisoning has been linked to miscarriages, she said. Limited application appears to be safe, “so make sure that the patient is using it on smaller body surface areas.”
As for systemic antifungal treatments, fluconazole, ketoconazole, and itraconazole should be avoided in pregnancy because of the risks of craniosynostosis, congenital heart defects, and skeletal anomalies, Dr. Murase said. However, she referred to a study that found no increased risk of congenital malformations with fluconazole during the first trimester, and a patient could be reassured if, for example, she was treated for a yeast infection before she knew she was pregnant, she said.
Griseofulvin is not recommended during pregnancy, but a 2020 study suggests that terbinafine is safe, she said. In that study, oral or topical terbinafine did not appear to be associated with an increased risk for spontaneous abortion or major malformations. “Certainly, we can wait until after the pregnancy to treat onychomycosis. But I have had situations that even in spite of regular topical therapy, pregnant patients needed to take some kind of oral agent” because of severe itching.
Viral Infections
For herpes simplex, acyclovir is the top choice, and famciclovir and valacyclovir (Valtrex) are likely safe, but daily prophylaxis is not recommended during pregnancy, Dr. Murase said.
Because of a lack of data, podofilox, cantharidin, and imiquimod for treating human papillomavirus (HPV) should be avoided, she said. Podophyllin is extremely dangerous in pregnancy and has been linked to maternal and fetal deaths, and malformations, and is contraindicated in pregnancy, she added.
Instead, liquid nitrogen is the treatment of choice for HPV in pregnant patients, she said.
Trichloracetic acid is the treatment of choice for condylomata acuminata, and squaric acid or intralesional Candida antigen injection for periungual verrucas can be used, she said, and limited applications of salicylic acid are considered safe.
Dr. Murase highlighted a 2014 paper that she coauthored on the safety of dermatologic medications during pregnancy, noting that an updated report will be published later this year.
Dr. Murase disclosed relationships with Regeneron and UCB (speaker), Sanofi/Regeneron and Bristol-Myers Squibb (advisory board), and UCB, AbbVie, and UpToDate (consulting).
A version of this article appeared on Medscape.com.
FROM AAD 2024
Does worsening metabolic syndrome increase the risk of developing cancer?
The conditions that comprise metabolic syndrome (high blood pressure, high blood sugar, increased abdominal adiposity, and high cholesterol and triglycerides) have been associated with an increased risk of diseases, including heart disease, stroke, and type 2 diabetes, wrote Li Deng, PhD, of Capital Medical University, Beijing, China, and colleagues.
A systematic review and meta-analysis published in Diabetes Care in 2012 showed an association between the presence of metabolic syndrome and an increased risk of various cancers including liver, bladder, pancreatic, breast, and colorectal.
More recently, a 2019 study published in Diabetes showed evidence of increased risk for certain cancers (pancreatic, kidney, uterine, cervical) but no increased risk for cancer overall.
However, the reasons for this association between metabolic syndrome and cancer remain unclear, and the effect of the fluctuating nature of metabolic syndrome over time on long-term cancer risk has not been explored, the researchers wrote.
What Does New Study Add to Other Research on Metabolic Syndrome and Cancer Risk?
In the new study, published in Cancer on March 11 (doi: 10.1002/cncr.35235), 44,115 adults in China were separated into four trajectories based on metabolic syndrome scores for the period from 2006 to 2010. The scores were based on clinical evidence of metabolic syndrome, defined using the International Diabetes Federation criteria of central obesity and the presence of at least two other factors including increased triglycerides, decreased HDL cholesterol, high blood pressure (or treatment for previously diagnosed hypertension), and increased fasting plasma glucose (or previous diagnosis of type 2 diabetes).
The average age of the participants was 49 years. The four trajectories of metabolic syndrome were low-stable (10.56% of participants), moderate-low (40.84%), moderate-high (41.46%), and elevated-increasing (7.14%), based on trends from the individuals’ initial physical exams on entering the study.
Over a median follow-up period of 9.4 years (from 2010 to 2021), 2,271 cancer diagnoses were reported in the study population. Those with an elevated-increasing metabolic syndrome trajectory had 1.3 times the risk of any cancer compared with those in the low-stable group. Risk for breast cancer, endometrial cancer, kidney cancer, colorectal cancer, and liver cancer in the highest trajectory group were 2.1, 3.3, 4.5, 2.5, and 1.6 times higher, respectively, compared to the lowest group. The increased risk in the elevated-trajectory group for all cancer types persisted when the low-stable, moderate-low, and moderate-high trajectory pattern groups were combined.
The researchers also examined the impact of chronic inflammation and found that individuals with persistently high metabolic syndrome scores and concurrent chronic inflammation had the highest risks of breast, endometrial, colon, and liver cancer. However, individuals with persistently high metabolic syndrome scores and no concurrent chronic inflammation had the highest risk of kidney cancer.
What Are the Limitations of This Research?
The researchers of the current study acknowledged the lack of information on other causes of cancer, including dietary habits, hepatitis C infection, and Helicobacter pylori infection. Other limitations include the focus only on individuals from a single community of mainly middle-aged men in China that may not generalize to other populations.
Also, the metabolic syndrome trajectories did not change much over time, which may be related to the short 4-year study period.
What Is the Takeaway Message for Clinical Practice?
The results suggest that monitoring and managing metabolic syndrome could help reduce cancer risk, the researchers concluded.
“This research suggests that proactive and continuous management of metabolic syndrome may serve as an essential strategy in preventing cancer,” senior author Han-Ping Shi, MD, PhD, of Capital Medical University in Beijing, said in a press release accompanying the study.
More research is needed to assess the impact of these interventions on cancer risk, he noted. However, the data from the current study can guide future research that may lead to more targeted treatments and more effective preventive strategies, he said in a statement.
The study was supported by the National Key Research and Development Program of China. The researchers had no financial conflicts to disclose.
The conditions that comprise metabolic syndrome (high blood pressure, high blood sugar, increased abdominal adiposity, and high cholesterol and triglycerides) have been associated with an increased risk of diseases, including heart disease, stroke, and type 2 diabetes, wrote Li Deng, PhD, of Capital Medical University, Beijing, China, and colleagues.
A systematic review and meta-analysis published in Diabetes Care in 2012 showed an association between the presence of metabolic syndrome and an increased risk of various cancers including liver, bladder, pancreatic, breast, and colorectal.
More recently, a 2019 study published in Diabetes showed evidence of increased risk for certain cancers (pancreatic, kidney, uterine, cervical) but no increased risk for cancer overall.
However, the reasons for this association between metabolic syndrome and cancer remain unclear, and the effect of the fluctuating nature of metabolic syndrome over time on long-term cancer risk has not been explored, the researchers wrote.
What Does New Study Add to Other Research on Metabolic Syndrome and Cancer Risk?
In the new study, published in Cancer on March 11 (doi: 10.1002/cncr.35235), 44,115 adults in China were separated into four trajectories based on metabolic syndrome scores for the period from 2006 to 2010. The scores were based on clinical evidence of metabolic syndrome, defined using the International Diabetes Federation criteria of central obesity and the presence of at least two other factors including increased triglycerides, decreased HDL cholesterol, high blood pressure (or treatment for previously diagnosed hypertension), and increased fasting plasma glucose (or previous diagnosis of type 2 diabetes).
The average age of the participants was 49 years. The four trajectories of metabolic syndrome were low-stable (10.56% of participants), moderate-low (40.84%), moderate-high (41.46%), and elevated-increasing (7.14%), based on trends from the individuals’ initial physical exams on entering the study.
Over a median follow-up period of 9.4 years (from 2010 to 2021), 2,271 cancer diagnoses were reported in the study population. Those with an elevated-increasing metabolic syndrome trajectory had 1.3 times the risk of any cancer compared with those in the low-stable group. Risk for breast cancer, endometrial cancer, kidney cancer, colorectal cancer, and liver cancer in the highest trajectory group were 2.1, 3.3, 4.5, 2.5, and 1.6 times higher, respectively, compared to the lowest group. The increased risk in the elevated-trajectory group for all cancer types persisted when the low-stable, moderate-low, and moderate-high trajectory pattern groups were combined.
The researchers also examined the impact of chronic inflammation and found that individuals with persistently high metabolic syndrome scores and concurrent chronic inflammation had the highest risks of breast, endometrial, colon, and liver cancer. However, individuals with persistently high metabolic syndrome scores and no concurrent chronic inflammation had the highest risk of kidney cancer.
What Are the Limitations of This Research?
The researchers of the current study acknowledged the lack of information on other causes of cancer, including dietary habits, hepatitis C infection, and Helicobacter pylori infection. Other limitations include the focus only on individuals from a single community of mainly middle-aged men in China that may not generalize to other populations.
Also, the metabolic syndrome trajectories did not change much over time, which may be related to the short 4-year study period.
What Is the Takeaway Message for Clinical Practice?
The results suggest that monitoring and managing metabolic syndrome could help reduce cancer risk, the researchers concluded.
“This research suggests that proactive and continuous management of metabolic syndrome may serve as an essential strategy in preventing cancer,” senior author Han-Ping Shi, MD, PhD, of Capital Medical University in Beijing, said in a press release accompanying the study.
More research is needed to assess the impact of these interventions on cancer risk, he noted. However, the data from the current study can guide future research that may lead to more targeted treatments and more effective preventive strategies, he said in a statement.
The study was supported by the National Key Research and Development Program of China. The researchers had no financial conflicts to disclose.
The conditions that comprise metabolic syndrome (high blood pressure, high blood sugar, increased abdominal adiposity, and high cholesterol and triglycerides) have been associated with an increased risk of diseases, including heart disease, stroke, and type 2 diabetes, wrote Li Deng, PhD, of Capital Medical University, Beijing, China, and colleagues.
A systematic review and meta-analysis published in Diabetes Care in 2012 showed an association between the presence of metabolic syndrome and an increased risk of various cancers including liver, bladder, pancreatic, breast, and colorectal.
More recently, a 2019 study published in Diabetes showed evidence of increased risk for certain cancers (pancreatic, kidney, uterine, cervical) but no increased risk for cancer overall.
However, the reasons for this association between metabolic syndrome and cancer remain unclear, and the effect of the fluctuating nature of metabolic syndrome over time on long-term cancer risk has not been explored, the researchers wrote.
What Does New Study Add to Other Research on Metabolic Syndrome and Cancer Risk?
In the new study, published in Cancer on March 11 (doi: 10.1002/cncr.35235), 44,115 adults in China were separated into four trajectories based on metabolic syndrome scores for the period from 2006 to 2010. The scores were based on clinical evidence of metabolic syndrome, defined using the International Diabetes Federation criteria of central obesity and the presence of at least two other factors including increased triglycerides, decreased HDL cholesterol, high blood pressure (or treatment for previously diagnosed hypertension), and increased fasting plasma glucose (or previous diagnosis of type 2 diabetes).
The average age of the participants was 49 years. The four trajectories of metabolic syndrome were low-stable (10.56% of participants), moderate-low (40.84%), moderate-high (41.46%), and elevated-increasing (7.14%), based on trends from the individuals’ initial physical exams on entering the study.
Over a median follow-up period of 9.4 years (from 2010 to 2021), 2,271 cancer diagnoses were reported in the study population. Those with an elevated-increasing metabolic syndrome trajectory had 1.3 times the risk of any cancer compared with those in the low-stable group. Risk for breast cancer, endometrial cancer, kidney cancer, colorectal cancer, and liver cancer in the highest trajectory group were 2.1, 3.3, 4.5, 2.5, and 1.6 times higher, respectively, compared to the lowest group. The increased risk in the elevated-trajectory group for all cancer types persisted when the low-stable, moderate-low, and moderate-high trajectory pattern groups were combined.
The researchers also examined the impact of chronic inflammation and found that individuals with persistently high metabolic syndrome scores and concurrent chronic inflammation had the highest risks of breast, endometrial, colon, and liver cancer. However, individuals with persistently high metabolic syndrome scores and no concurrent chronic inflammation had the highest risk of kidney cancer.
What Are the Limitations of This Research?
The researchers of the current study acknowledged the lack of information on other causes of cancer, including dietary habits, hepatitis C infection, and Helicobacter pylori infection. Other limitations include the focus only on individuals from a single community of mainly middle-aged men in China that may not generalize to other populations.
Also, the metabolic syndrome trajectories did not change much over time, which may be related to the short 4-year study period.
What Is the Takeaway Message for Clinical Practice?
The results suggest that monitoring and managing metabolic syndrome could help reduce cancer risk, the researchers concluded.
“This research suggests that proactive and continuous management of metabolic syndrome may serve as an essential strategy in preventing cancer,” senior author Han-Ping Shi, MD, PhD, of Capital Medical University in Beijing, said in a press release accompanying the study.
More research is needed to assess the impact of these interventions on cancer risk, he noted. However, the data from the current study can guide future research that may lead to more targeted treatments and more effective preventive strategies, he said in a statement.
The study was supported by the National Key Research and Development Program of China. The researchers had no financial conflicts to disclose.
FROM CANCER






