User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
President Biden signs 10 new orders to help fight COVID-19

“For the past year, we couldn’t rely on the federal government to act with the urgency and focus and coordination we needed, and we have seen the tragic cost of that failure,” Mr. Biden said in remarks from the White House, unveiling his 198-page National Strategy for the COVID-19 Response and Pandemic Preparedness.
He said as many as 500,000 Americans will have died by February. “It’s going to take months for us to turn things around,” he said.
“Our national strategy is comprehensive – it’s based on science, not politics; it’s based on truth, not denial,” Mr. Biden said. He also promised to restore public trust, in part by having scientists and public health experts speak to the public. “That’s why you’ll be hearing a lot more from Dr. Fauci again, not from the president,” he said, adding that the experts will be “free from political interference.”
While the president’s executive orders can help accomplish some of the plan’s proposals, the majority will require new funding from Congress and will be included in the $1.9 trillion American Rescue package that Mr. Biden hopes legislators will approve.
Ten new orders
The 10 new pandemic-related orders Biden signed on Jan. 21 follow two he signed on his first day in office.
One establishes a COVID-19 Response Office responsible for coordinating the pandemic response across all federal departments and agencies and also reestablishes the White House Directorate on Global Health Security and Biodefense, which was disabled by the Trump administration.
The other order requires masks and physical distancing in all federal buildings, on all federal lands, and by federal employees and contractors.
Among the new orders will be directives that:
- Require individuals to also wear masks in airports and planes, and when using other modes of public transportation including trains, boats, and intercity buses, and also require international travelers to produce proof of a recent negative COVID-19 test prior to entry and to quarantine after entry.
- Federal agencies use all powers, including the Defense Production Act, to accelerate manufacturing and delivery of supplies such as N95 masks, gowns, gloves, swabs, reagents, pipette tips, rapid test kits, and nitrocellulose material for rapid antigen tests, and all equipment and material needed to accelerate manufacture, delivery, and administration of COVID-19 vaccine.
- Create a Pandemic Testing Board to expand supply and access, to promote more surge capacity, and to ensure equitable access to tests.
- Facilitate discovery, development, and trials of potential COVID-19 treatments, as well as expand access to programs that can meet the long-term health needs of those recovering from the disease.
- Facilitate more and better data sharing that will allow businesses, schools, hospitals, and individuals to make real-time decisions based on spread in their community.
- Direct the Education and Health & Human Services departments to provide schools and child-care operations guidance on how to reopen and operate safely.
- Direct the Occupational Safety and Health Administration (OSHA) to immediately release clear guidance for employers to help keep workers safe and to enforce health and safety requirements.
The plan also sets goals for vaccination – including 100 million shots in the administration’s first 100 days. President Biden had already previewed his goals for vaccination, including setting up mass vaccination sites and mobile vaccination sites. During his remarks, Mr. Biden said that he had already directed the Federal Emergency Management Agency (FEMA) to begin setting up the vaccination centers.
The administration is also going to look into improving reimbursement for giving vaccines. As a start, the HHS will ask the Centers for Medicare & Medicaid Services to consider if a higher rate “may more accurately compensate providers,” according to the Biden plan.
“But the brutal truth is it will take months before we can get the majority of Americans vaccinated,” said Mr. Biden.
As part of the goal of ensuring an equitable pandemic response, the president will sign an order that establishes a COVID-19 Health Equity Task Force. The task force is charged with providing recommendations for allocating resources and funding in communities with inequities in COVID-19 outcomes by race, ethnicity, geography, disability, and other considerations.
Finally, the administration has committed to being more transparent and sharing more information. The national plan calls for the federal government to conduct regular, expert-led, science-based public briefings and to release regular reports on the pandemic. The administration said it will launch massive science-based public information campaigns – in multiple languages – to educate Americans on masks, testing, and vaccines, and also work to counter misinformation and disinformation.
The American Academy of Family Physicians (AAFP) applauded Mr. Biden’s initiative. “If enacted, this bold legislative agenda will provide much-needed support to American families struggling during the pandemic – especially communities of color and those hardest hit by the virus,” Ada D. Stewart, MD, AAFP president, said in a statement.
Dr. Stewart also noted that family physicians “are uniquely positioned in their communities to educate patients, prioritize access, and coordinate administration of the COVID-19 vaccines,” and urged the administration to ensure that family physicians and staff be vaccinated as soon as possible, to help them “more safely provide care to their communities.”
A version of this article first appeared on Medscape.com.

“For the past year, we couldn’t rely on the federal government to act with the urgency and focus and coordination we needed, and we have seen the tragic cost of that failure,” Mr. Biden said in remarks from the White House, unveiling his 198-page National Strategy for the COVID-19 Response and Pandemic Preparedness.
He said as many as 500,000 Americans will have died by February. “It’s going to take months for us to turn things around,” he said.
“Our national strategy is comprehensive – it’s based on science, not politics; it’s based on truth, not denial,” Mr. Biden said. He also promised to restore public trust, in part by having scientists and public health experts speak to the public. “That’s why you’ll be hearing a lot more from Dr. Fauci again, not from the president,” he said, adding that the experts will be “free from political interference.”
While the president’s executive orders can help accomplish some of the plan’s proposals, the majority will require new funding from Congress and will be included in the $1.9 trillion American Rescue package that Mr. Biden hopes legislators will approve.
Ten new orders
The 10 new pandemic-related orders Biden signed on Jan. 21 follow two he signed on his first day in office.
One establishes a COVID-19 Response Office responsible for coordinating the pandemic response across all federal departments and agencies and also reestablishes the White House Directorate on Global Health Security and Biodefense, which was disabled by the Trump administration.
The other order requires masks and physical distancing in all federal buildings, on all federal lands, and by federal employees and contractors.
Among the new orders will be directives that:
- Require individuals to also wear masks in airports and planes, and when using other modes of public transportation including trains, boats, and intercity buses, and also require international travelers to produce proof of a recent negative COVID-19 test prior to entry and to quarantine after entry.
- Federal agencies use all powers, including the Defense Production Act, to accelerate manufacturing and delivery of supplies such as N95 masks, gowns, gloves, swabs, reagents, pipette tips, rapid test kits, and nitrocellulose material for rapid antigen tests, and all equipment and material needed to accelerate manufacture, delivery, and administration of COVID-19 vaccine.
- Create a Pandemic Testing Board to expand supply and access, to promote more surge capacity, and to ensure equitable access to tests.
- Facilitate discovery, development, and trials of potential COVID-19 treatments, as well as expand access to programs that can meet the long-term health needs of those recovering from the disease.
- Facilitate more and better data sharing that will allow businesses, schools, hospitals, and individuals to make real-time decisions based on spread in their community.
- Direct the Education and Health & Human Services departments to provide schools and child-care operations guidance on how to reopen and operate safely.
- Direct the Occupational Safety and Health Administration (OSHA) to immediately release clear guidance for employers to help keep workers safe and to enforce health and safety requirements.
The plan also sets goals for vaccination – including 100 million shots in the administration’s first 100 days. President Biden had already previewed his goals for vaccination, including setting up mass vaccination sites and mobile vaccination sites. During his remarks, Mr. Biden said that he had already directed the Federal Emergency Management Agency (FEMA) to begin setting up the vaccination centers.
The administration is also going to look into improving reimbursement for giving vaccines. As a start, the HHS will ask the Centers for Medicare & Medicaid Services to consider if a higher rate “may more accurately compensate providers,” according to the Biden plan.
“But the brutal truth is it will take months before we can get the majority of Americans vaccinated,” said Mr. Biden.
As part of the goal of ensuring an equitable pandemic response, the president will sign an order that establishes a COVID-19 Health Equity Task Force. The task force is charged with providing recommendations for allocating resources and funding in communities with inequities in COVID-19 outcomes by race, ethnicity, geography, disability, and other considerations.
Finally, the administration has committed to being more transparent and sharing more information. The national plan calls for the federal government to conduct regular, expert-led, science-based public briefings and to release regular reports on the pandemic. The administration said it will launch massive science-based public information campaigns – in multiple languages – to educate Americans on masks, testing, and vaccines, and also work to counter misinformation and disinformation.
The American Academy of Family Physicians (AAFP) applauded Mr. Biden’s initiative. “If enacted, this bold legislative agenda will provide much-needed support to American families struggling during the pandemic – especially communities of color and those hardest hit by the virus,” Ada D. Stewart, MD, AAFP president, said in a statement.
Dr. Stewart also noted that family physicians “are uniquely positioned in their communities to educate patients, prioritize access, and coordinate administration of the COVID-19 vaccines,” and urged the administration to ensure that family physicians and staff be vaccinated as soon as possible, to help them “more safely provide care to their communities.”
A version of this article first appeared on Medscape.com.

“For the past year, we couldn’t rely on the federal government to act with the urgency and focus and coordination we needed, and we have seen the tragic cost of that failure,” Mr. Biden said in remarks from the White House, unveiling his 198-page National Strategy for the COVID-19 Response and Pandemic Preparedness.
He said as many as 500,000 Americans will have died by February. “It’s going to take months for us to turn things around,” he said.
“Our national strategy is comprehensive – it’s based on science, not politics; it’s based on truth, not denial,” Mr. Biden said. He also promised to restore public trust, in part by having scientists and public health experts speak to the public. “That’s why you’ll be hearing a lot more from Dr. Fauci again, not from the president,” he said, adding that the experts will be “free from political interference.”
While the president’s executive orders can help accomplish some of the plan’s proposals, the majority will require new funding from Congress and will be included in the $1.9 trillion American Rescue package that Mr. Biden hopes legislators will approve.
Ten new orders
The 10 new pandemic-related orders Biden signed on Jan. 21 follow two he signed on his first day in office.
One establishes a COVID-19 Response Office responsible for coordinating the pandemic response across all federal departments and agencies and also reestablishes the White House Directorate on Global Health Security and Biodefense, which was disabled by the Trump administration.
The other order requires masks and physical distancing in all federal buildings, on all federal lands, and by federal employees and contractors.
Among the new orders will be directives that:
- Require individuals to also wear masks in airports and planes, and when using other modes of public transportation including trains, boats, and intercity buses, and also require international travelers to produce proof of a recent negative COVID-19 test prior to entry and to quarantine after entry.
- Federal agencies use all powers, including the Defense Production Act, to accelerate manufacturing and delivery of supplies such as N95 masks, gowns, gloves, swabs, reagents, pipette tips, rapid test kits, and nitrocellulose material for rapid antigen tests, and all equipment and material needed to accelerate manufacture, delivery, and administration of COVID-19 vaccine.
- Create a Pandemic Testing Board to expand supply and access, to promote more surge capacity, and to ensure equitable access to tests.
- Facilitate discovery, development, and trials of potential COVID-19 treatments, as well as expand access to programs that can meet the long-term health needs of those recovering from the disease.
- Facilitate more and better data sharing that will allow businesses, schools, hospitals, and individuals to make real-time decisions based on spread in their community.
- Direct the Education and Health & Human Services departments to provide schools and child-care operations guidance on how to reopen and operate safely.
- Direct the Occupational Safety and Health Administration (OSHA) to immediately release clear guidance for employers to help keep workers safe and to enforce health and safety requirements.
The plan also sets goals for vaccination – including 100 million shots in the administration’s first 100 days. President Biden had already previewed his goals for vaccination, including setting up mass vaccination sites and mobile vaccination sites. During his remarks, Mr. Biden said that he had already directed the Federal Emergency Management Agency (FEMA) to begin setting up the vaccination centers.
The administration is also going to look into improving reimbursement for giving vaccines. As a start, the HHS will ask the Centers for Medicare & Medicaid Services to consider if a higher rate “may more accurately compensate providers,” according to the Biden plan.
“But the brutal truth is it will take months before we can get the majority of Americans vaccinated,” said Mr. Biden.
As part of the goal of ensuring an equitable pandemic response, the president will sign an order that establishes a COVID-19 Health Equity Task Force. The task force is charged with providing recommendations for allocating resources and funding in communities with inequities in COVID-19 outcomes by race, ethnicity, geography, disability, and other considerations.
Finally, the administration has committed to being more transparent and sharing more information. The national plan calls for the federal government to conduct regular, expert-led, science-based public briefings and to release regular reports on the pandemic. The administration said it will launch massive science-based public information campaigns – in multiple languages – to educate Americans on masks, testing, and vaccines, and also work to counter misinformation and disinformation.
The American Academy of Family Physicians (AAFP) applauded Mr. Biden’s initiative. “If enacted, this bold legislative agenda will provide much-needed support to American families struggling during the pandemic – especially communities of color and those hardest hit by the virus,” Ada D. Stewart, MD, AAFP president, said in a statement.
Dr. Stewart also noted that family physicians “are uniquely positioned in their communities to educate patients, prioritize access, and coordinate administration of the COVID-19 vaccines,” and urged the administration to ensure that family physicians and staff be vaccinated as soon as possible, to help them “more safely provide care to their communities.”
A version of this article first appeared on Medscape.com.
Metformin treatment again linked to fewer deaths from COVID-19
People with type 2 diabetes who develop COVID-19 show a substantially reduced risk of dying if they are taking metformin, shows a study that adds to prior research indicating the drug might somehow play a role in reducing the severity of infection.
“Unlike several previous analyses, this was a study in a racially diverse population with a high proportion of Blacks/African Americans and [it] revealed that metformin treatment of diabetes prior to diagnosis with COVID-19 was associated with a dramatic threefold reduced mortality in subjects with type 2 diabetes, even after correcting for multiple covariates,” first author Anath Shalev, MD, of the Comprehensive Diabetes Center at the University of Alabama at Birmingham, said in an interview.
But Anne Peters, MD, a professor of clinical medicine at the University of Southern California, Los Angeles, said caution is needed when interpreting these findings.
“It’s hard to tease out the true effects because, for instance, those treated with insulin may be a sicker subset of patients with diabetes than those on metformin, or those with comorbidities such as renal insufficiency may not be treated with metformin” she said in an interview.
“In general, though, treatment obviously matters and people who are better treated tend to do better, so while I think this study raises the question of what role metformin plays in the risk of mortality and COVID-19, I don’t think it necessarily proves the association,” Dr. Peters asserted.
Diverse population
The new study, published this month in Frontiers of Endocrinology, included 25,326 individuals who were tested for COVID-19 at the University of Alabama at Birmingham Hospital between February and June 2020.
Overall, 2.4% tested positive for COVID-19 (n = 604), which the authors note is likely a low figure because screening included asymptomatic hospital staff and patients having elective procedures.
Black/African American patients had a significantly higher risk of COVID-19 positivity, compared with White patients (odds ratio, 2.6; P < .0001). Rates were also higher among those with hypertension (OR, 2.46), diabetes (OR, 2.11), and obesity (OR, 1.93), compared with those without each condition (all P < .0001).
The overall mortality rate in COVID-19-positive patients was 11%. Diabetes was associated with a dramatically increased risk of death (OR, 3.62; P < .0001), and remained an independent risk factor even after adjusting for age, race, sex, obesity, and hypertension.
Notably, the reduction in mortality among those with diabetes taking metformin prior to COVID-19 diagnosis was significant: 11% of those patients died, compared with 23% of those with diabetes not taking metformin (OR, 0.33; P = .021).
Similar findings reported across varied populations
The study adds to mounting research suggesting metformin could have a protective effect on COVID-19 mortality, including an early report from Wuhan, China, findings from the French CORONADO study, and a U.S. study linking treatment with decreased mortality among women with COVID-19.
Of note, the effects of metformin on mortality in the current study were observed in men and women alike, as well as in high-risk subgroups including African Americans.
“The fact that such similar results were obtained in different populations from around the world suggests that the observed reduction in mortality risk, associated with metformin use in subjects with type 2 diabetes and COVID-19, might be generalizable,” the authors wrote.
“Furthermore, these findings underline the importance of following general diabetes treatment and prevention guidelines and not delaying or discontinuing any metformin treatment,” they add.
Speculation of mechanisms includes anti-inflammatory effects
While the mechanisms behind metformin’s potential role in reducing mortality risk in COVID-19 are unknown, the authors note that the most obvious assumption – that improved glycemic control may be a key factor – is disputed by the study’s finding that blood glucose levels and hemoglobin A1c were not significantly different among COVID-19 survivors taking versus not taking metformin.
They point instead to metformin’s known anti-inflammatory and antithrombotic properties.
“We therefore hypothesize that, by exerting some of these effects, metformin might improve outcomes and we are now in the process of investigating this possibility further,” Dr. Shalev said.
Dr. Peters noted that anti-inflammatory properties, themselves, are not necessarily unique to metformin in the treatment of diabetes.
“Many other agents, such as sodium-glucose cotransporter 2 (SGLT2) inhibitors can reduce inflammation, so I don’t know if that would explain it, but it certainly could help,” she said. “[Reducing inflammation] is a hypothesis you see commonly with diabetes drugs, but I think there are also a lot of metabolic benefits from metformin.”
“It was fascinating that they had the A1c data and that survival with metformin didn’t appear to be as related to A1c levels as one might think,” she added.
Notably, a key advantage, should the effects and mechanisms be validated, is metformin’s high accessibility, Dr. Peters added.
“This doesn’t necessarily tell us what we can do to reduce the health care disparities surrounding COVID-19, but the fact that metformin is low cost and easily available is very important, so maybe it will help as we try to grapple with other risk factors.”
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People with type 2 diabetes who develop COVID-19 show a substantially reduced risk of dying if they are taking metformin, shows a study that adds to prior research indicating the drug might somehow play a role in reducing the severity of infection.
“Unlike several previous analyses, this was a study in a racially diverse population with a high proportion of Blacks/African Americans and [it] revealed that metformin treatment of diabetes prior to diagnosis with COVID-19 was associated with a dramatic threefold reduced mortality in subjects with type 2 diabetes, even after correcting for multiple covariates,” first author Anath Shalev, MD, of the Comprehensive Diabetes Center at the University of Alabama at Birmingham, said in an interview.
But Anne Peters, MD, a professor of clinical medicine at the University of Southern California, Los Angeles, said caution is needed when interpreting these findings.
“It’s hard to tease out the true effects because, for instance, those treated with insulin may be a sicker subset of patients with diabetes than those on metformin, or those with comorbidities such as renal insufficiency may not be treated with metformin” she said in an interview.
“In general, though, treatment obviously matters and people who are better treated tend to do better, so while I think this study raises the question of what role metformin plays in the risk of mortality and COVID-19, I don’t think it necessarily proves the association,” Dr. Peters asserted.
Diverse population
The new study, published this month in Frontiers of Endocrinology, included 25,326 individuals who were tested for COVID-19 at the University of Alabama at Birmingham Hospital between February and June 2020.
Overall, 2.4% tested positive for COVID-19 (n = 604), which the authors note is likely a low figure because screening included asymptomatic hospital staff and patients having elective procedures.
Black/African American patients had a significantly higher risk of COVID-19 positivity, compared with White patients (odds ratio, 2.6; P < .0001). Rates were also higher among those with hypertension (OR, 2.46), diabetes (OR, 2.11), and obesity (OR, 1.93), compared with those without each condition (all P < .0001).
The overall mortality rate in COVID-19-positive patients was 11%. Diabetes was associated with a dramatically increased risk of death (OR, 3.62; P < .0001), and remained an independent risk factor even after adjusting for age, race, sex, obesity, and hypertension.
Notably, the reduction in mortality among those with diabetes taking metformin prior to COVID-19 diagnosis was significant: 11% of those patients died, compared with 23% of those with diabetes not taking metformin (OR, 0.33; P = .021).
Similar findings reported across varied populations
The study adds to mounting research suggesting metformin could have a protective effect on COVID-19 mortality, including an early report from Wuhan, China, findings from the French CORONADO study, and a U.S. study linking treatment with decreased mortality among women with COVID-19.
Of note, the effects of metformin on mortality in the current study were observed in men and women alike, as well as in high-risk subgroups including African Americans.
“The fact that such similar results were obtained in different populations from around the world suggests that the observed reduction in mortality risk, associated with metformin use in subjects with type 2 diabetes and COVID-19, might be generalizable,” the authors wrote.
“Furthermore, these findings underline the importance of following general diabetes treatment and prevention guidelines and not delaying or discontinuing any metformin treatment,” they add.
Speculation of mechanisms includes anti-inflammatory effects
While the mechanisms behind metformin’s potential role in reducing mortality risk in COVID-19 are unknown, the authors note that the most obvious assumption – that improved glycemic control may be a key factor – is disputed by the study’s finding that blood glucose levels and hemoglobin A1c were not significantly different among COVID-19 survivors taking versus not taking metformin.
They point instead to metformin’s known anti-inflammatory and antithrombotic properties.
“We therefore hypothesize that, by exerting some of these effects, metformin might improve outcomes and we are now in the process of investigating this possibility further,” Dr. Shalev said.
Dr. Peters noted that anti-inflammatory properties, themselves, are not necessarily unique to metformin in the treatment of diabetes.
“Many other agents, such as sodium-glucose cotransporter 2 (SGLT2) inhibitors can reduce inflammation, so I don’t know if that would explain it, but it certainly could help,” she said. “[Reducing inflammation] is a hypothesis you see commonly with diabetes drugs, but I think there are also a lot of metabolic benefits from metformin.”
“It was fascinating that they had the A1c data and that survival with metformin didn’t appear to be as related to A1c levels as one might think,” she added.
Notably, a key advantage, should the effects and mechanisms be validated, is metformin’s high accessibility, Dr. Peters added.
“This doesn’t necessarily tell us what we can do to reduce the health care disparities surrounding COVID-19, but the fact that metformin is low cost and easily available is very important, so maybe it will help as we try to grapple with other risk factors.”
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People with type 2 diabetes who develop COVID-19 show a substantially reduced risk of dying if they are taking metformin, shows a study that adds to prior research indicating the drug might somehow play a role in reducing the severity of infection.
“Unlike several previous analyses, this was a study in a racially diverse population with a high proportion of Blacks/African Americans and [it] revealed that metformin treatment of diabetes prior to diagnosis with COVID-19 was associated with a dramatic threefold reduced mortality in subjects with type 2 diabetes, even after correcting for multiple covariates,” first author Anath Shalev, MD, of the Comprehensive Diabetes Center at the University of Alabama at Birmingham, said in an interview.
But Anne Peters, MD, a professor of clinical medicine at the University of Southern California, Los Angeles, said caution is needed when interpreting these findings.
“It’s hard to tease out the true effects because, for instance, those treated with insulin may be a sicker subset of patients with diabetes than those on metformin, or those with comorbidities such as renal insufficiency may not be treated with metformin” she said in an interview.
“In general, though, treatment obviously matters and people who are better treated tend to do better, so while I think this study raises the question of what role metformin plays in the risk of mortality and COVID-19, I don’t think it necessarily proves the association,” Dr. Peters asserted.
Diverse population
The new study, published this month in Frontiers of Endocrinology, included 25,326 individuals who were tested for COVID-19 at the University of Alabama at Birmingham Hospital between February and June 2020.
Overall, 2.4% tested positive for COVID-19 (n = 604), which the authors note is likely a low figure because screening included asymptomatic hospital staff and patients having elective procedures.
Black/African American patients had a significantly higher risk of COVID-19 positivity, compared with White patients (odds ratio, 2.6; P < .0001). Rates were also higher among those with hypertension (OR, 2.46), diabetes (OR, 2.11), and obesity (OR, 1.93), compared with those without each condition (all P < .0001).
The overall mortality rate in COVID-19-positive patients was 11%. Diabetes was associated with a dramatically increased risk of death (OR, 3.62; P < .0001), and remained an independent risk factor even after adjusting for age, race, sex, obesity, and hypertension.
Notably, the reduction in mortality among those with diabetes taking metformin prior to COVID-19 diagnosis was significant: 11% of those patients died, compared with 23% of those with diabetes not taking metformin (OR, 0.33; P = .021).
Similar findings reported across varied populations
The study adds to mounting research suggesting metformin could have a protective effect on COVID-19 mortality, including an early report from Wuhan, China, findings from the French CORONADO study, and a U.S. study linking treatment with decreased mortality among women with COVID-19.
Of note, the effects of metformin on mortality in the current study were observed in men and women alike, as well as in high-risk subgroups including African Americans.
“The fact that such similar results were obtained in different populations from around the world suggests that the observed reduction in mortality risk, associated with metformin use in subjects with type 2 diabetes and COVID-19, might be generalizable,” the authors wrote.
“Furthermore, these findings underline the importance of following general diabetes treatment and prevention guidelines and not delaying or discontinuing any metformin treatment,” they add.
Speculation of mechanisms includes anti-inflammatory effects
While the mechanisms behind metformin’s potential role in reducing mortality risk in COVID-19 are unknown, the authors note that the most obvious assumption – that improved glycemic control may be a key factor – is disputed by the study’s finding that blood glucose levels and hemoglobin A1c were not significantly different among COVID-19 survivors taking versus not taking metformin.
They point instead to metformin’s known anti-inflammatory and antithrombotic properties.
“We therefore hypothesize that, by exerting some of these effects, metformin might improve outcomes and we are now in the process of investigating this possibility further,” Dr. Shalev said.
Dr. Peters noted that anti-inflammatory properties, themselves, are not necessarily unique to metformin in the treatment of diabetes.
“Many other agents, such as sodium-glucose cotransporter 2 (SGLT2) inhibitors can reduce inflammation, so I don’t know if that would explain it, but it certainly could help,” she said. “[Reducing inflammation] is a hypothesis you see commonly with diabetes drugs, but I think there are also a lot of metabolic benefits from metformin.”
“It was fascinating that they had the A1c data and that survival with metformin didn’t appear to be as related to A1c levels as one might think,” she added.
Notably, a key advantage, should the effects and mechanisms be validated, is metformin’s high accessibility, Dr. Peters added.
“This doesn’t necessarily tell us what we can do to reduce the health care disparities surrounding COVID-19, but the fact that metformin is low cost and easily available is very important, so maybe it will help as we try to grapple with other risk factors.”
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Seven ways President Biden could now change health care
President Joe Biden has come into office after an unexpected shift in Congress. On Jan. 5, Democrats scored an upset by winning two U.S. Senate seats in runoff elections in Georgia, giving them control of the Senate.
Now the Democrats have control of all three levers of power – the Senate, the House, and the presidency – for the first time since the early years of the Obama administration.
How will President Biden use this new concentration of power to shape health care policy?
Democrats’ small majorities in both houses of Congress suggest that moderation and bipartisanship will be necessary to get things done. Moreover, Mr. Biden himself is calling for bipartisanship. “On this January day,” he said in his inauguration speech, “my whole soul is in this: Bringing America together, uniting our people, uniting our nation.”
Key health care actions that Mr. Biden could pursue include the following.
1. Passing a new COVID-19 relief bill
Above all, Mr. Biden is focused on overcoming the COVID-19 pandemic, which has been registering record deaths recently, and getting newly released vaccines to Americans.
“Dealing with the coronavirus pandemic is one of the most important battles our administration will face, and I will be informed by science and by experts,” the president said.
“There is no question that the pandemic is the highest priority for the Biden administration,” said Larry Levitt, executive vice president for health policy at the Henry J. Kaiser Family Foundation. “COVID will dominate the early weeks and months of this administration. His success rests, in particular, on improving the rollout of vaccines.”
Five days before his inauguration, the president-elect unveiled the American Rescue Plan, a massive, $1.9 trillion legislative package intended to hasten rollout of COVID-19 vaccines, improve COVID-19 testing, and provide financial help to businesses and individuals, among many other things.
The bill would add $1,400 to the recently passed $600 government relief payments for each American, amounting to a $2,000 check. It would also enact many non-COVID-19 measures, such as a $15-an-hour minimum wage and measures to bolster the Affordable Care Act (ACA).
If Democrats cannot reach a deal with the Republicans, they might turn the proposal into a reconciliation bill, which could then be passed with a simple majority. However, drafting a reconciliation bill is a long, complicated process that would require removing provisions that don’t meet the requirements of reconciliation, said Hazen Marshall, a Washington lobbyist and former staffer for Sen. Mitch McConnell.
Most importantly, Mr. Marshall said, reconciliation bills bring out diehard partisanship. “They involve a sledgehammer mentality,” he says. “You’re telling the other side that their views aren’t going to matter.” The final version of the ACA, for example, was passed as a reconciliation bill, with not one Republican vote.
In the Trump years, “the last four reconciliation bills did not get any votes from the minority,” added Rodney Whitlock, PhD, a political consultant at McDermott+Consulting, who worked 21 years for Republicans in the House. “When the majority chooses to use reconciliation, it is an admission that it has no interest in working with the minority.”
Hammering out a compromise will be tough, but Robert Pearl MD, former CEO of the Permanente Medical Group and a professor at Stanford (Calif.) University, said that if anyone can do it, it would be President Biden. Having served in the Senate for 36 years, “Biden knows Congress better than any president since Lyndon Johnson,” he said. “He can reach across the aisle and get legislation passed as much as anyone could these days.”
2. Restoring Obamacare
Mr. Biden has vowed to undo a gradual dismantling of the ACA that went on during the Trump administration through executive orders, rule-making, and new laws. “Reinvigorating the ACA was a central part of Biden’s platform as a candidate,” Mr. Levitt said.
Each Trump action against the ACA must be undone in the same way. Presidential orders must be met with presidential orders, regulations with regulations, and legislation with legislation.
The ACA is also being challenged in the Supreme Court. Republicans under Trump passed a law that reduced the penalty for not buying health insurance under the ACA to zero. Then a group of 20 states, led by Texas, filed a lawsuit asserting that this change makes the ACA unconstitutional.
The lawsuit was heard by the Supreme Court in November. From remarks made by the justices then, it appears that the court might well uphold the law when a verdict comes down in June.
But just in case, Mr. Biden wants Congress to enact a small penalty for not buying health insurance, which would remove the basis of the lawsuit.
Mr. Biden’s choice for secretary of Health and Human Services shows his level of commitment to protecting the ACA. His HHS nominee is California Attorney General Xavier Becerra, who led a group of 17 states defending the ACA in the current lawsuit.
In addition to undoing Trump’s changes, Mr. Biden plans to expand the ACA beyond the original legislation. The new COVID-19 bill contains provisions that would expand subsidies to buy insurance on the exchanges and would lower the maximum percentage of income that anyone has to pay for health insurance to 8.5%.
Dealing with Medicaid is also related to the ACA. In 2012, the Supreme Court struck down a mandate that states expand their Medicaid programs, with substantial funding from the federal government.
To date, 12 states still do not participate in the Medicaid expansion. To lure them into the expansion, the Democrat-controlled House last session passed a bill that would offer to pay the entire bill for the first 3 years of Medicaid expansion if they chose to enact an expansion.
3. Undoing other Trump actions in health care
In addition to changes in the ACA, Trump also enacted a number of other changes in health care that President Biden could undo. For example, Mr. Biden says he will reenter the World Health Organization (WHO) so that the United States could better coordinate a COVID-19 response with other nations. Trump exited the WHO with the stroke of a pen, and Mr. Biden can do the same in reverse.
Under Trump, the Centers for Medicare & Medicaid Services used waivers to weaken the ACA and allow states to alter their Medicaid programs. One waiver allows Georgia to leave the ACA exchanges and put brokers in charge of buying coverage. Other waivers allow states to transform federal Medicaid payments into block grants, which several states are planning to do.
The Trump CMS has allowed several states to use Medicaid waivers to add work requirements for Medicaid recipients. The courts have blocked the work rules so far, and the Biden CMS may decide to reverse these waivers or modify them.
“Undoing waivers is normally a fairly simple thing,” Mr. Levitt said. In January, however, the Trump CMS asked some waiver states to sign new contracts in which the CMS pledges not to end a waiver without 9 months’ notice. It’s unclear how many states signed such contracts and what obligation the Biden CMS has to enforce them.
The Trump CMS also stopped reimbursing insurers for waiving deductibles and copayments for low-income customers, as directed by the ACA. Without federal reimbursement, some insurers raised premiums by as much as 20% to cover the costs. It is unclear how the Biden CMS would tackle this change.
4. Negotiating lower drug prices
Allowing Medicare to negotiate drug prices, a major plank in Mr. Biden’s campaign, would seem like a slam dunk for the Democrats. This approach is backed by 89% of Americans, including 84% of Republicans, according to a Kaiser Family Foundation survey in December.
“With that level of support, it’s hard to go wrong politically on this issue,” Mr. Levitt said.
Many Republicans, however, do not favor negotiating drug prices, and the two parties continue to be far apart on how to control drug prices. Trump signed an action that allows Americans to buy cheaper drugs abroad, an approach that Mr. Biden also supports, but it is now tied up in the courts.
“A drug pricing bill has always been difficult to pass,” Dr. Whitlock said. “The issue is popular with the public, but change does not come easily. The drug lobby is one the strongest in Washington, and now it may be even stronger, since it was the drug companies that gave us the COVID vaccines.”
Dr. Whitlock said Republicans will want Democrats to compromise on drug pricing, but he doubts they will do so. The House passed a bill to negotiate drug prices last year, which never was voted on in the Senate. “It is difficult to imagine that the Democrats will be able to move rightward from that House bill,” Dr. Whitlock said. “Democrats are likely to stand pat on drug pricing.”
5. Introducing a public option
President Biden’s campaign proposal for a public option – health insurance offered by the federal government – and to lower the age for Medicare eligibility from 65 years to 60 years, resulted from a compromise between two factions of the Democratic party on how to expand coverage.
Although Mr. Biden and other moderates wanted to focus on fixing the ACA, Democrats led by Sen. Bernie Sanders of Vermont called for a single-payer system, dubbed “Medicare for all.” A public option was seen as the middle ground between the two camps.
“A public option would be a very controversial,” Dr. Whitlock said. Critics say it would pay at Medicare rates, which would reduce doctors’ reimbursements, and save very little money compared with a single-payer system.
Dr. Pearl sees similar problems with lowering the Medicare age. “This would be an expensive change that the federal government could not afford, particularly with all the spending on the pandemic,” he said. “And it would be tough on doctors and hospitals, because Medicare pays less than the private insurance payment they are now getting.”
“The public option is likely to get serious discussion within the Democratic caucus and get onto the Senate floor,” Mr. Levitt said. “The party won’t ignore it.” He notes that in the new Senate, Sen. Sanders chairs the budget committee, and from that position he is likely to push for expanding access to care.
Mr. Levitt says the Biden CMS might allow states to experiment with a statewide public option or even a single-payer model, but he concedes that states, with their budgets ravaged by COVID-19, do not currently have the money to launch such programs.
6. Reviving the CMS
Under President Obama, the CMS was the engine that implemented the ACA and shepherded wider use of value-based reimbursements, which reward providers for quality and outcomes rather than volume.
Under the Trump administration, CMS leadership continued to uphold value-based reimbursement, Dr. Pearl observed. “CMS leadership championed value-based payments, but they encountered a lot of pushback from doctors and hospitals and had to scale back their goals,” he said.
On the other hand, the Trump CMS took a 180-degree turn on the ACA and worked to take it apart. This took a toll on staff morale, according to Donald M. Berwick, MD, who ran the CMS under President Obama. “Many people in CMS did not feel supported during the Trump administration, and some of them left,” Dr. Berwick said.
The CMS needs experienced staff on board to write comprehensible rules and regulations that can overcome court challenges.
Having a fully functioning CMS also requires consistent leadership, which was a problem for Obama. When Mr. Obama nominated Dr. Berwick, 60 Senate votes were needed to confirm him, and Republicans would not vote for him. Mr. Obama eventually brought Dr. Berwick in as a recess appointment, but it meant he could serve for only 17 months.
Since then, Senate confirmation rules have changed so that only a simple majority is needed to confirm appointments. This is important for Biden’s nominees, Dr. Berwick said. “For a president, having your team in place means you are able to execute the policies you want,” he said. “You need to have consistent leadership.”
7. Potentially changing health care without Congress
Even with their newly won control of the Senate, the Democrats’ thin majorities in both houses of Congress may not be enough to pass much legislation if Republicans are solidly opposed.
Democrats in the House also have a narrow path this session in which to pass legislation. The Democratic leadership has an 11-vote majority, but it must contend with 15 moderate representatives in purple districts (where Democrats and Republicans have about equal support).
A bigger problem looms before the Democrats. In 2022, the party may well lose its majorities in both houses. Mr. Whitlock notes that the party of an incoming president normally loses seats in the first midterm election. “The last incoming president to keep both houses of Congress in his first midterm was Jimmy Carter,” he said.
If this happens, President Biden would have to govern without the support of Congress, which is what Barack Obama had to do through most of his presidency. As Mr. Obama’s vice president, Mr. Biden is well aware how that goes. Governing without Congress means relying on presidential orders and decrees.
In health care, Mr. Biden has a powerful policy-making tool, the Center for Medicare & Medicaid Innovation (CMMI). The CMMI was empowered by the ACA to initiate pilot programs for new payment models.
So far, the CMMI’s work has been mainly limited to accountable care organizations, bundled payments, and patient-centered medical homes, but it could also be used to enact new federal policies that would normally require Congressional action, Mr. Levitt said.
Conclusion
Expectations have been very high for what President Joe Biden can do in health care. He needs to unite a very divided political system to defeat a deadly pandemic, restore Obamacare, and sign landmark legislation, such as a drug-pricing bill.
But shepherding bills through Congress will be a challenge. “You need to have accountability, unity, and civility, which is a Herculean task,” Mr. Whitlock said. “You have to keep policies off the table that could blow up the bipartisanship.”
A version of this article first appeared on Medscape.com.
President Joe Biden has come into office after an unexpected shift in Congress. On Jan. 5, Democrats scored an upset by winning two U.S. Senate seats in runoff elections in Georgia, giving them control of the Senate.
Now the Democrats have control of all three levers of power – the Senate, the House, and the presidency – for the first time since the early years of the Obama administration.
How will President Biden use this new concentration of power to shape health care policy?
Democrats’ small majorities in both houses of Congress suggest that moderation and bipartisanship will be necessary to get things done. Moreover, Mr. Biden himself is calling for bipartisanship. “On this January day,” he said in his inauguration speech, “my whole soul is in this: Bringing America together, uniting our people, uniting our nation.”
Key health care actions that Mr. Biden could pursue include the following.
1. Passing a new COVID-19 relief bill
Above all, Mr. Biden is focused on overcoming the COVID-19 pandemic, which has been registering record deaths recently, and getting newly released vaccines to Americans.
“Dealing with the coronavirus pandemic is one of the most important battles our administration will face, and I will be informed by science and by experts,” the president said.
“There is no question that the pandemic is the highest priority for the Biden administration,” said Larry Levitt, executive vice president for health policy at the Henry J. Kaiser Family Foundation. “COVID will dominate the early weeks and months of this administration. His success rests, in particular, on improving the rollout of vaccines.”
Five days before his inauguration, the president-elect unveiled the American Rescue Plan, a massive, $1.9 trillion legislative package intended to hasten rollout of COVID-19 vaccines, improve COVID-19 testing, and provide financial help to businesses and individuals, among many other things.
The bill would add $1,400 to the recently passed $600 government relief payments for each American, amounting to a $2,000 check. It would also enact many non-COVID-19 measures, such as a $15-an-hour minimum wage and measures to bolster the Affordable Care Act (ACA).
If Democrats cannot reach a deal with the Republicans, they might turn the proposal into a reconciliation bill, which could then be passed with a simple majority. However, drafting a reconciliation bill is a long, complicated process that would require removing provisions that don’t meet the requirements of reconciliation, said Hazen Marshall, a Washington lobbyist and former staffer for Sen. Mitch McConnell.
Most importantly, Mr. Marshall said, reconciliation bills bring out diehard partisanship. “They involve a sledgehammer mentality,” he says. “You’re telling the other side that their views aren’t going to matter.” The final version of the ACA, for example, was passed as a reconciliation bill, with not one Republican vote.
In the Trump years, “the last four reconciliation bills did not get any votes from the minority,” added Rodney Whitlock, PhD, a political consultant at McDermott+Consulting, who worked 21 years for Republicans in the House. “When the majority chooses to use reconciliation, it is an admission that it has no interest in working with the minority.”
Hammering out a compromise will be tough, but Robert Pearl MD, former CEO of the Permanente Medical Group and a professor at Stanford (Calif.) University, said that if anyone can do it, it would be President Biden. Having served in the Senate for 36 years, “Biden knows Congress better than any president since Lyndon Johnson,” he said. “He can reach across the aisle and get legislation passed as much as anyone could these days.”
2. Restoring Obamacare
Mr. Biden has vowed to undo a gradual dismantling of the ACA that went on during the Trump administration through executive orders, rule-making, and new laws. “Reinvigorating the ACA was a central part of Biden’s platform as a candidate,” Mr. Levitt said.
Each Trump action against the ACA must be undone in the same way. Presidential orders must be met with presidential orders, regulations with regulations, and legislation with legislation.
The ACA is also being challenged in the Supreme Court. Republicans under Trump passed a law that reduced the penalty for not buying health insurance under the ACA to zero. Then a group of 20 states, led by Texas, filed a lawsuit asserting that this change makes the ACA unconstitutional.
The lawsuit was heard by the Supreme Court in November. From remarks made by the justices then, it appears that the court might well uphold the law when a verdict comes down in June.
But just in case, Mr. Biden wants Congress to enact a small penalty for not buying health insurance, which would remove the basis of the lawsuit.
Mr. Biden’s choice for secretary of Health and Human Services shows his level of commitment to protecting the ACA. His HHS nominee is California Attorney General Xavier Becerra, who led a group of 17 states defending the ACA in the current lawsuit.
In addition to undoing Trump’s changes, Mr. Biden plans to expand the ACA beyond the original legislation. The new COVID-19 bill contains provisions that would expand subsidies to buy insurance on the exchanges and would lower the maximum percentage of income that anyone has to pay for health insurance to 8.5%.
Dealing with Medicaid is also related to the ACA. In 2012, the Supreme Court struck down a mandate that states expand their Medicaid programs, with substantial funding from the federal government.
To date, 12 states still do not participate in the Medicaid expansion. To lure them into the expansion, the Democrat-controlled House last session passed a bill that would offer to pay the entire bill for the first 3 years of Medicaid expansion if they chose to enact an expansion.
3. Undoing other Trump actions in health care
In addition to changes in the ACA, Trump also enacted a number of other changes in health care that President Biden could undo. For example, Mr. Biden says he will reenter the World Health Organization (WHO) so that the United States could better coordinate a COVID-19 response with other nations. Trump exited the WHO with the stroke of a pen, and Mr. Biden can do the same in reverse.
Under Trump, the Centers for Medicare & Medicaid Services used waivers to weaken the ACA and allow states to alter their Medicaid programs. One waiver allows Georgia to leave the ACA exchanges and put brokers in charge of buying coverage. Other waivers allow states to transform federal Medicaid payments into block grants, which several states are planning to do.
The Trump CMS has allowed several states to use Medicaid waivers to add work requirements for Medicaid recipients. The courts have blocked the work rules so far, and the Biden CMS may decide to reverse these waivers or modify them.
“Undoing waivers is normally a fairly simple thing,” Mr. Levitt said. In January, however, the Trump CMS asked some waiver states to sign new contracts in which the CMS pledges not to end a waiver without 9 months’ notice. It’s unclear how many states signed such contracts and what obligation the Biden CMS has to enforce them.
The Trump CMS also stopped reimbursing insurers for waiving deductibles and copayments for low-income customers, as directed by the ACA. Without federal reimbursement, some insurers raised premiums by as much as 20% to cover the costs. It is unclear how the Biden CMS would tackle this change.
4. Negotiating lower drug prices
Allowing Medicare to negotiate drug prices, a major plank in Mr. Biden’s campaign, would seem like a slam dunk for the Democrats. This approach is backed by 89% of Americans, including 84% of Republicans, according to a Kaiser Family Foundation survey in December.
“With that level of support, it’s hard to go wrong politically on this issue,” Mr. Levitt said.
Many Republicans, however, do not favor negotiating drug prices, and the two parties continue to be far apart on how to control drug prices. Trump signed an action that allows Americans to buy cheaper drugs abroad, an approach that Mr. Biden also supports, but it is now tied up in the courts.
“A drug pricing bill has always been difficult to pass,” Dr. Whitlock said. “The issue is popular with the public, but change does not come easily. The drug lobby is one the strongest in Washington, and now it may be even stronger, since it was the drug companies that gave us the COVID vaccines.”
Dr. Whitlock said Republicans will want Democrats to compromise on drug pricing, but he doubts they will do so. The House passed a bill to negotiate drug prices last year, which never was voted on in the Senate. “It is difficult to imagine that the Democrats will be able to move rightward from that House bill,” Dr. Whitlock said. “Democrats are likely to stand pat on drug pricing.”
5. Introducing a public option
President Biden’s campaign proposal for a public option – health insurance offered by the federal government – and to lower the age for Medicare eligibility from 65 years to 60 years, resulted from a compromise between two factions of the Democratic party on how to expand coverage.
Although Mr. Biden and other moderates wanted to focus on fixing the ACA, Democrats led by Sen. Bernie Sanders of Vermont called for a single-payer system, dubbed “Medicare for all.” A public option was seen as the middle ground between the two camps.
“A public option would be a very controversial,” Dr. Whitlock said. Critics say it would pay at Medicare rates, which would reduce doctors’ reimbursements, and save very little money compared with a single-payer system.
Dr. Pearl sees similar problems with lowering the Medicare age. “This would be an expensive change that the federal government could not afford, particularly with all the spending on the pandemic,” he said. “And it would be tough on doctors and hospitals, because Medicare pays less than the private insurance payment they are now getting.”
“The public option is likely to get serious discussion within the Democratic caucus and get onto the Senate floor,” Mr. Levitt said. “The party won’t ignore it.” He notes that in the new Senate, Sen. Sanders chairs the budget committee, and from that position he is likely to push for expanding access to care.
Mr. Levitt says the Biden CMS might allow states to experiment with a statewide public option or even a single-payer model, but he concedes that states, with their budgets ravaged by COVID-19, do not currently have the money to launch such programs.
6. Reviving the CMS
Under President Obama, the CMS was the engine that implemented the ACA and shepherded wider use of value-based reimbursements, which reward providers for quality and outcomes rather than volume.
Under the Trump administration, CMS leadership continued to uphold value-based reimbursement, Dr. Pearl observed. “CMS leadership championed value-based payments, but they encountered a lot of pushback from doctors and hospitals and had to scale back their goals,” he said.
On the other hand, the Trump CMS took a 180-degree turn on the ACA and worked to take it apart. This took a toll on staff morale, according to Donald M. Berwick, MD, who ran the CMS under President Obama. “Many people in CMS did not feel supported during the Trump administration, and some of them left,” Dr. Berwick said.
The CMS needs experienced staff on board to write comprehensible rules and regulations that can overcome court challenges.
Having a fully functioning CMS also requires consistent leadership, which was a problem for Obama. When Mr. Obama nominated Dr. Berwick, 60 Senate votes were needed to confirm him, and Republicans would not vote for him. Mr. Obama eventually brought Dr. Berwick in as a recess appointment, but it meant he could serve for only 17 months.
Since then, Senate confirmation rules have changed so that only a simple majority is needed to confirm appointments. This is important for Biden’s nominees, Dr. Berwick said. “For a president, having your team in place means you are able to execute the policies you want,” he said. “You need to have consistent leadership.”
7. Potentially changing health care without Congress
Even with their newly won control of the Senate, the Democrats’ thin majorities in both houses of Congress may not be enough to pass much legislation if Republicans are solidly opposed.
Democrats in the House also have a narrow path this session in which to pass legislation. The Democratic leadership has an 11-vote majority, but it must contend with 15 moderate representatives in purple districts (where Democrats and Republicans have about equal support).
A bigger problem looms before the Democrats. In 2022, the party may well lose its majorities in both houses. Mr. Whitlock notes that the party of an incoming president normally loses seats in the first midterm election. “The last incoming president to keep both houses of Congress in his first midterm was Jimmy Carter,” he said.
If this happens, President Biden would have to govern without the support of Congress, which is what Barack Obama had to do through most of his presidency. As Mr. Obama’s vice president, Mr. Biden is well aware how that goes. Governing without Congress means relying on presidential orders and decrees.
In health care, Mr. Biden has a powerful policy-making tool, the Center for Medicare & Medicaid Innovation (CMMI). The CMMI was empowered by the ACA to initiate pilot programs for new payment models.
So far, the CMMI’s work has been mainly limited to accountable care organizations, bundled payments, and patient-centered medical homes, but it could also be used to enact new federal policies that would normally require Congressional action, Mr. Levitt said.
Conclusion
Expectations have been very high for what President Joe Biden can do in health care. He needs to unite a very divided political system to defeat a deadly pandemic, restore Obamacare, and sign landmark legislation, such as a drug-pricing bill.
But shepherding bills through Congress will be a challenge. “You need to have accountability, unity, and civility, which is a Herculean task,” Mr. Whitlock said. “You have to keep policies off the table that could blow up the bipartisanship.”
A version of this article first appeared on Medscape.com.
President Joe Biden has come into office after an unexpected shift in Congress. On Jan. 5, Democrats scored an upset by winning two U.S. Senate seats in runoff elections in Georgia, giving them control of the Senate.
Now the Democrats have control of all three levers of power – the Senate, the House, and the presidency – for the first time since the early years of the Obama administration.
How will President Biden use this new concentration of power to shape health care policy?
Democrats’ small majorities in both houses of Congress suggest that moderation and bipartisanship will be necessary to get things done. Moreover, Mr. Biden himself is calling for bipartisanship. “On this January day,” he said in his inauguration speech, “my whole soul is in this: Bringing America together, uniting our people, uniting our nation.”
Key health care actions that Mr. Biden could pursue include the following.
1. Passing a new COVID-19 relief bill
Above all, Mr. Biden is focused on overcoming the COVID-19 pandemic, which has been registering record deaths recently, and getting newly released vaccines to Americans.
“Dealing with the coronavirus pandemic is one of the most important battles our administration will face, and I will be informed by science and by experts,” the president said.
“There is no question that the pandemic is the highest priority for the Biden administration,” said Larry Levitt, executive vice president for health policy at the Henry J. Kaiser Family Foundation. “COVID will dominate the early weeks and months of this administration. His success rests, in particular, on improving the rollout of vaccines.”
Five days before his inauguration, the president-elect unveiled the American Rescue Plan, a massive, $1.9 trillion legislative package intended to hasten rollout of COVID-19 vaccines, improve COVID-19 testing, and provide financial help to businesses and individuals, among many other things.
The bill would add $1,400 to the recently passed $600 government relief payments for each American, amounting to a $2,000 check. It would also enact many non-COVID-19 measures, such as a $15-an-hour minimum wage and measures to bolster the Affordable Care Act (ACA).
If Democrats cannot reach a deal with the Republicans, they might turn the proposal into a reconciliation bill, which could then be passed with a simple majority. However, drafting a reconciliation bill is a long, complicated process that would require removing provisions that don’t meet the requirements of reconciliation, said Hazen Marshall, a Washington lobbyist and former staffer for Sen. Mitch McConnell.
Most importantly, Mr. Marshall said, reconciliation bills bring out diehard partisanship. “They involve a sledgehammer mentality,” he says. “You’re telling the other side that their views aren’t going to matter.” The final version of the ACA, for example, was passed as a reconciliation bill, with not one Republican vote.
In the Trump years, “the last four reconciliation bills did not get any votes from the minority,” added Rodney Whitlock, PhD, a political consultant at McDermott+Consulting, who worked 21 years for Republicans in the House. “When the majority chooses to use reconciliation, it is an admission that it has no interest in working with the minority.”
Hammering out a compromise will be tough, but Robert Pearl MD, former CEO of the Permanente Medical Group and a professor at Stanford (Calif.) University, said that if anyone can do it, it would be President Biden. Having served in the Senate for 36 years, “Biden knows Congress better than any president since Lyndon Johnson,” he said. “He can reach across the aisle and get legislation passed as much as anyone could these days.”
2. Restoring Obamacare
Mr. Biden has vowed to undo a gradual dismantling of the ACA that went on during the Trump administration through executive orders, rule-making, and new laws. “Reinvigorating the ACA was a central part of Biden’s platform as a candidate,” Mr. Levitt said.
Each Trump action against the ACA must be undone in the same way. Presidential orders must be met with presidential orders, regulations with regulations, and legislation with legislation.
The ACA is also being challenged in the Supreme Court. Republicans under Trump passed a law that reduced the penalty for not buying health insurance under the ACA to zero. Then a group of 20 states, led by Texas, filed a lawsuit asserting that this change makes the ACA unconstitutional.
The lawsuit was heard by the Supreme Court in November. From remarks made by the justices then, it appears that the court might well uphold the law when a verdict comes down in June.
But just in case, Mr. Biden wants Congress to enact a small penalty for not buying health insurance, which would remove the basis of the lawsuit.
Mr. Biden’s choice for secretary of Health and Human Services shows his level of commitment to protecting the ACA. His HHS nominee is California Attorney General Xavier Becerra, who led a group of 17 states defending the ACA in the current lawsuit.
In addition to undoing Trump’s changes, Mr. Biden plans to expand the ACA beyond the original legislation. The new COVID-19 bill contains provisions that would expand subsidies to buy insurance on the exchanges and would lower the maximum percentage of income that anyone has to pay for health insurance to 8.5%.
Dealing with Medicaid is also related to the ACA. In 2012, the Supreme Court struck down a mandate that states expand their Medicaid programs, with substantial funding from the federal government.
To date, 12 states still do not participate in the Medicaid expansion. To lure them into the expansion, the Democrat-controlled House last session passed a bill that would offer to pay the entire bill for the first 3 years of Medicaid expansion if they chose to enact an expansion.
3. Undoing other Trump actions in health care
In addition to changes in the ACA, Trump also enacted a number of other changes in health care that President Biden could undo. For example, Mr. Biden says he will reenter the World Health Organization (WHO) so that the United States could better coordinate a COVID-19 response with other nations. Trump exited the WHO with the stroke of a pen, and Mr. Biden can do the same in reverse.
Under Trump, the Centers for Medicare & Medicaid Services used waivers to weaken the ACA and allow states to alter their Medicaid programs. One waiver allows Georgia to leave the ACA exchanges and put brokers in charge of buying coverage. Other waivers allow states to transform federal Medicaid payments into block grants, which several states are planning to do.
The Trump CMS has allowed several states to use Medicaid waivers to add work requirements for Medicaid recipients. The courts have blocked the work rules so far, and the Biden CMS may decide to reverse these waivers or modify them.
“Undoing waivers is normally a fairly simple thing,” Mr. Levitt said. In January, however, the Trump CMS asked some waiver states to sign new contracts in which the CMS pledges not to end a waiver without 9 months’ notice. It’s unclear how many states signed such contracts and what obligation the Biden CMS has to enforce them.
The Trump CMS also stopped reimbursing insurers for waiving deductibles and copayments for low-income customers, as directed by the ACA. Without federal reimbursement, some insurers raised premiums by as much as 20% to cover the costs. It is unclear how the Biden CMS would tackle this change.
4. Negotiating lower drug prices
Allowing Medicare to negotiate drug prices, a major plank in Mr. Biden’s campaign, would seem like a slam dunk for the Democrats. This approach is backed by 89% of Americans, including 84% of Republicans, according to a Kaiser Family Foundation survey in December.
“With that level of support, it’s hard to go wrong politically on this issue,” Mr. Levitt said.
Many Republicans, however, do not favor negotiating drug prices, and the two parties continue to be far apart on how to control drug prices. Trump signed an action that allows Americans to buy cheaper drugs abroad, an approach that Mr. Biden also supports, but it is now tied up in the courts.
“A drug pricing bill has always been difficult to pass,” Dr. Whitlock said. “The issue is popular with the public, but change does not come easily. The drug lobby is one the strongest in Washington, and now it may be even stronger, since it was the drug companies that gave us the COVID vaccines.”
Dr. Whitlock said Republicans will want Democrats to compromise on drug pricing, but he doubts they will do so. The House passed a bill to negotiate drug prices last year, which never was voted on in the Senate. “It is difficult to imagine that the Democrats will be able to move rightward from that House bill,” Dr. Whitlock said. “Democrats are likely to stand pat on drug pricing.”
5. Introducing a public option
President Biden’s campaign proposal for a public option – health insurance offered by the federal government – and to lower the age for Medicare eligibility from 65 years to 60 years, resulted from a compromise between two factions of the Democratic party on how to expand coverage.
Although Mr. Biden and other moderates wanted to focus on fixing the ACA, Democrats led by Sen. Bernie Sanders of Vermont called for a single-payer system, dubbed “Medicare for all.” A public option was seen as the middle ground between the two camps.
“A public option would be a very controversial,” Dr. Whitlock said. Critics say it would pay at Medicare rates, which would reduce doctors’ reimbursements, and save very little money compared with a single-payer system.
Dr. Pearl sees similar problems with lowering the Medicare age. “This would be an expensive change that the federal government could not afford, particularly with all the spending on the pandemic,” he said. “And it would be tough on doctors and hospitals, because Medicare pays less than the private insurance payment they are now getting.”
“The public option is likely to get serious discussion within the Democratic caucus and get onto the Senate floor,” Mr. Levitt said. “The party won’t ignore it.” He notes that in the new Senate, Sen. Sanders chairs the budget committee, and from that position he is likely to push for expanding access to care.
Mr. Levitt says the Biden CMS might allow states to experiment with a statewide public option or even a single-payer model, but he concedes that states, with their budgets ravaged by COVID-19, do not currently have the money to launch such programs.
6. Reviving the CMS
Under President Obama, the CMS was the engine that implemented the ACA and shepherded wider use of value-based reimbursements, which reward providers for quality and outcomes rather than volume.
Under the Trump administration, CMS leadership continued to uphold value-based reimbursement, Dr. Pearl observed. “CMS leadership championed value-based payments, but they encountered a lot of pushback from doctors and hospitals and had to scale back their goals,” he said.
On the other hand, the Trump CMS took a 180-degree turn on the ACA and worked to take it apart. This took a toll on staff morale, according to Donald M. Berwick, MD, who ran the CMS under President Obama. “Many people in CMS did not feel supported during the Trump administration, and some of them left,” Dr. Berwick said.
The CMS needs experienced staff on board to write comprehensible rules and regulations that can overcome court challenges.
Having a fully functioning CMS also requires consistent leadership, which was a problem for Obama. When Mr. Obama nominated Dr. Berwick, 60 Senate votes were needed to confirm him, and Republicans would not vote for him. Mr. Obama eventually brought Dr. Berwick in as a recess appointment, but it meant he could serve for only 17 months.
Since then, Senate confirmation rules have changed so that only a simple majority is needed to confirm appointments. This is important for Biden’s nominees, Dr. Berwick said. “For a president, having your team in place means you are able to execute the policies you want,” he said. “You need to have consistent leadership.”
7. Potentially changing health care without Congress
Even with their newly won control of the Senate, the Democrats’ thin majorities in both houses of Congress may not be enough to pass much legislation if Republicans are solidly opposed.
Democrats in the House also have a narrow path this session in which to pass legislation. The Democratic leadership has an 11-vote majority, but it must contend with 15 moderate representatives in purple districts (where Democrats and Republicans have about equal support).
A bigger problem looms before the Democrats. In 2022, the party may well lose its majorities in both houses. Mr. Whitlock notes that the party of an incoming president normally loses seats in the first midterm election. “The last incoming president to keep both houses of Congress in his first midterm was Jimmy Carter,” he said.
If this happens, President Biden would have to govern without the support of Congress, which is what Barack Obama had to do through most of his presidency. As Mr. Obama’s vice president, Mr. Biden is well aware how that goes. Governing without Congress means relying on presidential orders and decrees.
In health care, Mr. Biden has a powerful policy-making tool, the Center for Medicare & Medicaid Innovation (CMMI). The CMMI was empowered by the ACA to initiate pilot programs for new payment models.
So far, the CMMI’s work has been mainly limited to accountable care organizations, bundled payments, and patient-centered medical homes, but it could also be used to enact new federal policies that would normally require Congressional action, Mr. Levitt said.
Conclusion
Expectations have been very high for what President Joe Biden can do in health care. He needs to unite a very divided political system to defeat a deadly pandemic, restore Obamacare, and sign landmark legislation, such as a drug-pricing bill.
But shepherding bills through Congress will be a challenge. “You need to have accountability, unity, and civility, which is a Herculean task,” Mr. Whitlock said. “You have to keep policies off the table that could blow up the bipartisanship.”
A version of this article first appeared on Medscape.com.
Reproductive psychiatry in 2021: Old questions and new challenges
Across this period of the pandemic, we’ve spent considerable attention focusing on adaptations to clinical care for pregnant and postpartum women across the board. From the start, this has included a shift to telemedicine for the majority of our patients who come to see us with psychiatric disorders either before, during, or after pregnancy.
Specific issues for perinatal patients since the early days of COVID-19 have included the shifts in women’s plans with respect to delivery as well as the limitation on women’s ability to configure the types of support that they had originally planned on with family, friends, and others during delivery. Telemedicine again helped, at least in part, to fill that void by having online digital support by individuals or groups for both pregnant and postpartum women. These supports were always available, but quickly scaled up during the first 6-9 months of the pandemic and have likely seen their greatest increase in participation in the history of support groups for pregnant and postpartum women.
Similarly, at our own center, we have seen a dramatic increase across the last 10 months in requests for consultation by women with psychiatric disorders who have hopes and plans to conceive, to those who are pregnant or post partum and who are trying to sustain emotional well-being despite the added burden of the pandemic. As we heard similar stories regarding interactions with perinatal patients from reproductive psychiatrists across the country, my colleagues and I had to set up an additional resource, Virtual Rounds at the Center for Women’s Mental Health at Massachusetts General Hospital, Boston, which has been mentioned in previous columns, which has only grown during the last 6 months of the pandemic. We have colleagues across the country joining us from 2 p.m. to 3 p.m. on Wednesdays after our own faculty rounds, where we perform case reviews of our own patients, and invite our colleagues to share cases that are then reviewed with expert panelists together with our own faculty in a collaborative environment. Feedback from the community of clinicians has indicated that these virtual rounds have been invaluable to their efforts in taking care of women with perinatal psychiatric issues, particularly during the pandemic.
Of particular note during consultations on our service is the number of women coming to see us for questions about the reproductive safety of the medications on which they are maintained. Hundreds of women present to the center each year for the most up-to-date information regarding the reproductive safety of the most commonly used psychiatric medications in reproductive-age women, including antidepressants (SSRIs, serotonin norepinephrine reuptake inhibitor), mood stabilizers, lithium, lamotrigine, and atypical antipsychotics, as well as other medicines used to treat symptoms that have been a particular issue during the pandemic, such as insomnia and anxiety (benzodiazepines, nonbenzodiazepines, sedative hypnotics, and medicines such as gabapentin).
While consultation regarding risk of fetal exposure to psychotropics has been the cornerstone of our clinical work for 25 years, it has taken on a particularly critical dimension during the pandemic given the wish that women stay euthymic during the pandemic to limit the possibility of patients needing to present in a clinical space that would increase their risk for COVID-19, and to also minimize their risk for postpartum depression. (Psychiatric disorder during pregnancy remains the strongest predictor of emergence or worsening of underlying illness during the postpartum period.)
It is also noteworthy that, during a pandemic year, publications in reproductive psychiatry have been numerous, and we continue to make an effort at our own center to keep up with this and to share with our colleagues our impression of that literature using the weekly blog at womensmentalhealth.org. Last year brought the largest audience to the blog and visits to womensmentalhealth.org in the history of our center.
A case recently at our center presents a unique opportunity to review a confusing question in reproductive psychiatry over the last 15 years. A woman with a longstanding history of mixed anxiety and depression recently came to see us on a regimen of escitalopram and low-dose benzodiazepine. She was doing well, and she and her husband of 4 years were hopeful about starting to try to conceive despite the pandemic. We reviewed the reproductive safety data of the medicines on which she was maintained and made plans to follow-up as her plans galvanized. She notified me several months later that she had become pregnant but had experienced an early miscarriage. The patient was obviously upset and, as she reflected on her decision to maintain treatment with SSRI during her attempts to conceive and across a very early pregnancy, she queried about the extent to which her SSRI use might have contributed to her miscarriage.
The question about the possible association of antidepressant use during pregnancy and increased risk for miscarriage goes back at least 15 years when there were reports of an increased risk of miscarriage in women taking SSRIs during pregnancy. In that early work, there was an apparent increase in miscarriage in women taking SSRIs relative to a control group, but the rate did not exceed the prevalence of miscarriage in the general population. Since those early reports, we are lucky to have had multiple investigators look very closely at this issue, including one meta-analysis of 11 studies done approximately 8 years ago that failed to show an increased risk of miscarriage in the setting of first trimester exposure to SSRIs.
What has been most problematic methodologically, however, has been the failure to account for the potential role of depression in models that predict risk. A subsequent large epidemiologic study from Denmark evaluating over a million women has looked at this question further. The authors found a slightly increased risk of spontaneous abortion associated with the use of antidepressants (12.0% in women with antidepressant exposure vs. 11.1% in women with no exposure). However, looking only at women with a diagnosis of depression, the adjusted risk ratio for spontaneous abortion after any antidepressant exposure was 1.00 (95% confidence interval, 0.80-1.24). Thus, the researchers concluded that exposure to depression – but not exposure to antidepressant – is associated with a slightly higher risk of miscarriage.
Even more recently, a follow-up study examining this question supports the large epidemiologic study by Kjaersgaard and colleagues. For most readers, this effectively answers this very important question for women about rates of miscarriage associated with fetal exposure to SSRIs.
For the patient who presented at the center, reassuring her with this information felt particularly good, especially within the context of the pandemic. After several months of trying to conceive, she again became pregnant and delivered without difficulty. What was palpable in that clinical scenario, as it relates to the practice of reproductive psychiatry during the pandemic, is the even-greater emotional valence that questions about using psychiatric medications during pregnancy has taken on across these past months. While attention and thoughtful consideration about the relative risks of using psychiatric medications during pregnancy should be standard clinical practice, the level of anxiety associated with decisions to sustain or to discontinue treatment during pregnancy seems to have increased for some patients during the pandemic.
Even as the COVID-19 vaccine initiative across the United States is rolled out, 2021 will continue to be a complicated time for women and families. We still need to be vigilant. In addition to screening for perinatal depression during pregnancy and the postpartum period, we should be equally mindful of screening and treating perinatal anxiety, particularly during this challenging time. The challenge to keep pregnant and postpartum women well is perhaps even greater now, 10 months into the pandemic, than it was when we were in crisis mode in March 2020. As clinicians, we need to mobilize the spectrum of both pharmacologic and nonpharmacologic treatment options to sustain emotional well-being among women planning to conceive as well as those who are pregnant or postpartum as we navigate our way to safer times.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Across this period of the pandemic, we’ve spent considerable attention focusing on adaptations to clinical care for pregnant and postpartum women across the board. From the start, this has included a shift to telemedicine for the majority of our patients who come to see us with psychiatric disorders either before, during, or after pregnancy.
Specific issues for perinatal patients since the early days of COVID-19 have included the shifts in women’s plans with respect to delivery as well as the limitation on women’s ability to configure the types of support that they had originally planned on with family, friends, and others during delivery. Telemedicine again helped, at least in part, to fill that void by having online digital support by individuals or groups for both pregnant and postpartum women. These supports were always available, but quickly scaled up during the first 6-9 months of the pandemic and have likely seen their greatest increase in participation in the history of support groups for pregnant and postpartum women.
Similarly, at our own center, we have seen a dramatic increase across the last 10 months in requests for consultation by women with psychiatric disorders who have hopes and plans to conceive, to those who are pregnant or post partum and who are trying to sustain emotional well-being despite the added burden of the pandemic. As we heard similar stories regarding interactions with perinatal patients from reproductive psychiatrists across the country, my colleagues and I had to set up an additional resource, Virtual Rounds at the Center for Women’s Mental Health at Massachusetts General Hospital, Boston, which has been mentioned in previous columns, which has only grown during the last 6 months of the pandemic. We have colleagues across the country joining us from 2 p.m. to 3 p.m. on Wednesdays after our own faculty rounds, where we perform case reviews of our own patients, and invite our colleagues to share cases that are then reviewed with expert panelists together with our own faculty in a collaborative environment. Feedback from the community of clinicians has indicated that these virtual rounds have been invaluable to their efforts in taking care of women with perinatal psychiatric issues, particularly during the pandemic.
Of particular note during consultations on our service is the number of women coming to see us for questions about the reproductive safety of the medications on which they are maintained. Hundreds of women present to the center each year for the most up-to-date information regarding the reproductive safety of the most commonly used psychiatric medications in reproductive-age women, including antidepressants (SSRIs, serotonin norepinephrine reuptake inhibitor), mood stabilizers, lithium, lamotrigine, and atypical antipsychotics, as well as other medicines used to treat symptoms that have been a particular issue during the pandemic, such as insomnia and anxiety (benzodiazepines, nonbenzodiazepines, sedative hypnotics, and medicines such as gabapentin).
While consultation regarding risk of fetal exposure to psychotropics has been the cornerstone of our clinical work for 25 years, it has taken on a particularly critical dimension during the pandemic given the wish that women stay euthymic during the pandemic to limit the possibility of patients needing to present in a clinical space that would increase their risk for COVID-19, and to also minimize their risk for postpartum depression. (Psychiatric disorder during pregnancy remains the strongest predictor of emergence or worsening of underlying illness during the postpartum period.)
It is also noteworthy that, during a pandemic year, publications in reproductive psychiatry have been numerous, and we continue to make an effort at our own center to keep up with this and to share with our colleagues our impression of that literature using the weekly blog at womensmentalhealth.org. Last year brought the largest audience to the blog and visits to womensmentalhealth.org in the history of our center.
A case recently at our center presents a unique opportunity to review a confusing question in reproductive psychiatry over the last 15 years. A woman with a longstanding history of mixed anxiety and depression recently came to see us on a regimen of escitalopram and low-dose benzodiazepine. She was doing well, and she and her husband of 4 years were hopeful about starting to try to conceive despite the pandemic. We reviewed the reproductive safety data of the medicines on which she was maintained and made plans to follow-up as her plans galvanized. She notified me several months later that she had become pregnant but had experienced an early miscarriage. The patient was obviously upset and, as she reflected on her decision to maintain treatment with SSRI during her attempts to conceive and across a very early pregnancy, she queried about the extent to which her SSRI use might have contributed to her miscarriage.
The question about the possible association of antidepressant use during pregnancy and increased risk for miscarriage goes back at least 15 years when there were reports of an increased risk of miscarriage in women taking SSRIs during pregnancy. In that early work, there was an apparent increase in miscarriage in women taking SSRIs relative to a control group, but the rate did not exceed the prevalence of miscarriage in the general population. Since those early reports, we are lucky to have had multiple investigators look very closely at this issue, including one meta-analysis of 11 studies done approximately 8 years ago that failed to show an increased risk of miscarriage in the setting of first trimester exposure to SSRIs.
What has been most problematic methodologically, however, has been the failure to account for the potential role of depression in models that predict risk. A subsequent large epidemiologic study from Denmark evaluating over a million women has looked at this question further. The authors found a slightly increased risk of spontaneous abortion associated with the use of antidepressants (12.0% in women with antidepressant exposure vs. 11.1% in women with no exposure). However, looking only at women with a diagnosis of depression, the adjusted risk ratio for spontaneous abortion after any antidepressant exposure was 1.00 (95% confidence interval, 0.80-1.24). Thus, the researchers concluded that exposure to depression – but not exposure to antidepressant – is associated with a slightly higher risk of miscarriage.
Even more recently, a follow-up study examining this question supports the large epidemiologic study by Kjaersgaard and colleagues. For most readers, this effectively answers this very important question for women about rates of miscarriage associated with fetal exposure to SSRIs.
For the patient who presented at the center, reassuring her with this information felt particularly good, especially within the context of the pandemic. After several months of trying to conceive, she again became pregnant and delivered without difficulty. What was palpable in that clinical scenario, as it relates to the practice of reproductive psychiatry during the pandemic, is the even-greater emotional valence that questions about using psychiatric medications during pregnancy has taken on across these past months. While attention and thoughtful consideration about the relative risks of using psychiatric medications during pregnancy should be standard clinical practice, the level of anxiety associated with decisions to sustain or to discontinue treatment during pregnancy seems to have increased for some patients during the pandemic.
Even as the COVID-19 vaccine initiative across the United States is rolled out, 2021 will continue to be a complicated time for women and families. We still need to be vigilant. In addition to screening for perinatal depression during pregnancy and the postpartum period, we should be equally mindful of screening and treating perinatal anxiety, particularly during this challenging time. The challenge to keep pregnant and postpartum women well is perhaps even greater now, 10 months into the pandemic, than it was when we were in crisis mode in March 2020. As clinicians, we need to mobilize the spectrum of both pharmacologic and nonpharmacologic treatment options to sustain emotional well-being among women planning to conceive as well as those who are pregnant or postpartum as we navigate our way to safer times.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Across this period of the pandemic, we’ve spent considerable attention focusing on adaptations to clinical care for pregnant and postpartum women across the board. From the start, this has included a shift to telemedicine for the majority of our patients who come to see us with psychiatric disorders either before, during, or after pregnancy.
Specific issues for perinatal patients since the early days of COVID-19 have included the shifts in women’s plans with respect to delivery as well as the limitation on women’s ability to configure the types of support that they had originally planned on with family, friends, and others during delivery. Telemedicine again helped, at least in part, to fill that void by having online digital support by individuals or groups for both pregnant and postpartum women. These supports were always available, but quickly scaled up during the first 6-9 months of the pandemic and have likely seen their greatest increase in participation in the history of support groups for pregnant and postpartum women.
Similarly, at our own center, we have seen a dramatic increase across the last 10 months in requests for consultation by women with psychiatric disorders who have hopes and plans to conceive, to those who are pregnant or post partum and who are trying to sustain emotional well-being despite the added burden of the pandemic. As we heard similar stories regarding interactions with perinatal patients from reproductive psychiatrists across the country, my colleagues and I had to set up an additional resource, Virtual Rounds at the Center for Women’s Mental Health at Massachusetts General Hospital, Boston, which has been mentioned in previous columns, which has only grown during the last 6 months of the pandemic. We have colleagues across the country joining us from 2 p.m. to 3 p.m. on Wednesdays after our own faculty rounds, where we perform case reviews of our own patients, and invite our colleagues to share cases that are then reviewed with expert panelists together with our own faculty in a collaborative environment. Feedback from the community of clinicians has indicated that these virtual rounds have been invaluable to their efforts in taking care of women with perinatal psychiatric issues, particularly during the pandemic.
Of particular note during consultations on our service is the number of women coming to see us for questions about the reproductive safety of the medications on which they are maintained. Hundreds of women present to the center each year for the most up-to-date information regarding the reproductive safety of the most commonly used psychiatric medications in reproductive-age women, including antidepressants (SSRIs, serotonin norepinephrine reuptake inhibitor), mood stabilizers, lithium, lamotrigine, and atypical antipsychotics, as well as other medicines used to treat symptoms that have been a particular issue during the pandemic, such as insomnia and anxiety (benzodiazepines, nonbenzodiazepines, sedative hypnotics, and medicines such as gabapentin).
While consultation regarding risk of fetal exposure to psychotropics has been the cornerstone of our clinical work for 25 years, it has taken on a particularly critical dimension during the pandemic given the wish that women stay euthymic during the pandemic to limit the possibility of patients needing to present in a clinical space that would increase their risk for COVID-19, and to also minimize their risk for postpartum depression. (Psychiatric disorder during pregnancy remains the strongest predictor of emergence or worsening of underlying illness during the postpartum period.)
It is also noteworthy that, during a pandemic year, publications in reproductive psychiatry have been numerous, and we continue to make an effort at our own center to keep up with this and to share with our colleagues our impression of that literature using the weekly blog at womensmentalhealth.org. Last year brought the largest audience to the blog and visits to womensmentalhealth.org in the history of our center.
A case recently at our center presents a unique opportunity to review a confusing question in reproductive psychiatry over the last 15 years. A woman with a longstanding history of mixed anxiety and depression recently came to see us on a regimen of escitalopram and low-dose benzodiazepine. She was doing well, and she and her husband of 4 years were hopeful about starting to try to conceive despite the pandemic. We reviewed the reproductive safety data of the medicines on which she was maintained and made plans to follow-up as her plans galvanized. She notified me several months later that she had become pregnant but had experienced an early miscarriage. The patient was obviously upset and, as she reflected on her decision to maintain treatment with SSRI during her attempts to conceive and across a very early pregnancy, she queried about the extent to which her SSRI use might have contributed to her miscarriage.
The question about the possible association of antidepressant use during pregnancy and increased risk for miscarriage goes back at least 15 years when there were reports of an increased risk of miscarriage in women taking SSRIs during pregnancy. In that early work, there was an apparent increase in miscarriage in women taking SSRIs relative to a control group, but the rate did not exceed the prevalence of miscarriage in the general population. Since those early reports, we are lucky to have had multiple investigators look very closely at this issue, including one meta-analysis of 11 studies done approximately 8 years ago that failed to show an increased risk of miscarriage in the setting of first trimester exposure to SSRIs.
What has been most problematic methodologically, however, has been the failure to account for the potential role of depression in models that predict risk. A subsequent large epidemiologic study from Denmark evaluating over a million women has looked at this question further. The authors found a slightly increased risk of spontaneous abortion associated with the use of antidepressants (12.0% in women with antidepressant exposure vs. 11.1% in women with no exposure). However, looking only at women with a diagnosis of depression, the adjusted risk ratio for spontaneous abortion after any antidepressant exposure was 1.00 (95% confidence interval, 0.80-1.24). Thus, the researchers concluded that exposure to depression – but not exposure to antidepressant – is associated with a slightly higher risk of miscarriage.
Even more recently, a follow-up study examining this question supports the large epidemiologic study by Kjaersgaard and colleagues. For most readers, this effectively answers this very important question for women about rates of miscarriage associated with fetal exposure to SSRIs.
For the patient who presented at the center, reassuring her with this information felt particularly good, especially within the context of the pandemic. After several months of trying to conceive, she again became pregnant and delivered without difficulty. What was palpable in that clinical scenario, as it relates to the practice of reproductive psychiatry during the pandemic, is the even-greater emotional valence that questions about using psychiatric medications during pregnancy has taken on across these past months. While attention and thoughtful consideration about the relative risks of using psychiatric medications during pregnancy should be standard clinical practice, the level of anxiety associated with decisions to sustain or to discontinue treatment during pregnancy seems to have increased for some patients during the pandemic.
Even as the COVID-19 vaccine initiative across the United States is rolled out, 2021 will continue to be a complicated time for women and families. We still need to be vigilant. In addition to screening for perinatal depression during pregnancy and the postpartum period, we should be equally mindful of screening and treating perinatal anxiety, particularly during this challenging time. The challenge to keep pregnant and postpartum women well is perhaps even greater now, 10 months into the pandemic, than it was when we were in crisis mode in March 2020. As clinicians, we need to mobilize the spectrum of both pharmacologic and nonpharmacologic treatment options to sustain emotional well-being among women planning to conceive as well as those who are pregnant or postpartum as we navigate our way to safer times.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Repeated ketamine infusions linked to rapid relief of PTSD
Repeated intravenous infusions of ketamine provide rapid relief for patients with posttraumatic stress disorder, new research suggests.
In what investigators are calling the first randomized controlled trial of repeated ketamine administration for chronic PTSD, 30 patients received six infusions of ketamine or midazolam (used as a psychoactive placebo) over 2 consecutive weeks.
Between baseline and week 2, those receiving ketamine showed significantly greater improvement than those receiving midazolam. Total scores on the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5) for the first group were almost 12 points lower than the latter group at week 2, meeting the study’s primary outcome measure.
In addition, 67% vs. 20% of the patients, respectively, were considered to be treatment responders; time to loss of response for those in the ketamine group was 28 days.
Although the overall findings were as expected, “what was surprising was how robust the results were,” lead author Adriana Feder, MD, associate professor of psychiatry, Icahn School of Medicine, Mount Sinai, New York, told this news organization.
It was also a bit surprising that, in a study of just 30 participants, “we were able to show such a clear difference” between the two treatment groups, said Dr. Feder, who is also a coinventor on issued patents for the use of ketamine as therapy for PTSD, and codirector of the Ehrenkranz Lab for the Study of Human Resilience at Mount Sinai.
The findings were published online Jan. 5 in the American Journal of Psychiatry.
Unmet need
Ketamine is a glutamate N-methyl-D-aspartate (NMDA) receptor antagonist that was first approved by the U.S. Food and Drug Administration for anesthetic use in 1970. It has also been shown to be effective for treatment-resistant depression.
PTSD has a lifetime prevalence of about 6% in the United States. “While trauma-focused psychotherapies have the most empirical support, they are limited by significant rates of nonresponse, partial response, and treatment dropout,” the investigators write. Also, there are “few available pharmacotherapies for PTSD, and their efficacy is insufficient,” they add.
“There’s a real need for new treatment interventions that are effective for PTSD and also work rapidly, because it can take weeks to months for currently available treatments to work for PTSD,” Dr. Feder said.
The researchers previously conducted a “proof-of-concept” randomized controlled trial of single infusions of ketamine for chronic PTSD. Results published in 2014 in JAMA Psychiatry showed significant reduction in PTSD symptoms 24 hours after infusion.
For the current study, the investigative team wanted to assess whether ketamine was viable as a longer-term treatment.
“We were encouraged by our initial promising findings” of the earlier trial, Dr. Feder said. “We wanted to do the second study to see if ketamine really works for PTSD, to see if we could replicate the rapid improvement and also examine whether a course of six infusions over 2 weeks could maintain the improvement.”
Thirty patients (aged 18-70; mean age, 39 years) with chronic PTSD from civilian or military trauma were enrolled (mean PTSD duration, 15 years).
The most cited primary trauma was sexual assault or molestation (n = 13), followed by physical assault or abuse (n = 8), witnessing a violent assault or death (n = 4), witnessing the 9/11 attacks (n = 3), and combat exposure (n = 2).
During the 2-week treatment phase, half of the patients were randomly assigned to receive six infusions of ketamine hydrochloride at a dose of 0.5 mg/kg (86.7% women; mean CAPS-5 score, 42), while the other half received six infusions of midazolam at a dose of 0.045 mg/kg (66.7% women; mean CAPS-5 score, 40).
In addition to the primary outcome measure of 2-week changes on the CAPS-5, secondary outcomes included score changes on the Montgomery-Åsberg Depression Rating Scale (MADRS) and the Impact of Event Scale-Revised (IES-R).
Treatment response was defined as a 30% or more improvement in symptoms on the CAPS-5. A number of measures were also used to assess potential treatment-related adverse events (AEs).
Safe, effective
Results showed significantly lower total CAPS-5 scores for the ketamine group vs. the midazolam group at week 1 (score difference, 8.8 points; P = .03) and at week 2 (score difference, 11.88 points; P = .004).
Those receiving ketamine also showed improvements in three of the four PTSD symptom clusters on the CAPS-5: avoidance (P < .0001), negative mood and cognitions (P = .02), and intrusions (P = .03). The fourth symptom cluster – arousal and reactivity – did not show a significant improvement.
In addition, the ketamine group showed significantly greater improvement scores on the MADRS at both week 1 and week 2.
Treatment response at 2 weeks was achieved by 10 members of the ketamine group and by three members of the midazolam group (P = .03).
Secondary analyses showed rapid improvement in the treatment responders within the ketamine group, with a mean change of 26 points on the total IES-R score between baseline and 24 hours after their first infusion, and a mean change of 13.4 points on the MADRS total past-24-hour score, a 53% improvement on average.
“A response at 2 weeks is very rapid but they got better sometimes within the first day,” Dr. Feder noted.
There were no serious AEs reported. Although some dissociative symptoms occurred during ketamine infusions, with the highest levels reported at the end of the infusion, these symptoms had resolved by the next assessment, conducted 2 hours after infusion.
The most frequently reported AE in the ketamine group, compared with midazolam, after the start of infusions was blurred vision (53% vs. 0%), followed by dizziness (33% vs. 13%), fatigue (33% vs. 87%), headache (27% vs. 13%), and nausea or vomiting (20% vs. 7%).
‘Large-magnitude improvement’
The overall findings show that, in this patient population, “repeated intravenous ketamine infusions administered over 2 weeks were associated with a large-magnitude, clinically significant improvement in PTSD symptoms,” the investigators write.
The results “were very satisfying,” added Dr. Feder. “It was heartening also to hear what some of the participants would say. Some told us about how their symptoms and feelings had changed during the course of treatment with ketamine, where they felt stronger and better able to cope with their trauma and memories.”
She noted, however, that this was not a study designed to specifically assess ketamine in treatment-resistant PTSD. “Some patients had had multiple treatments before that hadn’t worked, while others had not received treatment before. Efficacy for treatment-resistant PTSD is an important question for future research,” Dr. Feder said.
Other areas worth future exploration include treatment efficacy in patients with different types of trauma and whether outcomes can last longer in patients receiving ketamine plus psychotherapy treatment, she noted.
“I don’t want to ignore the fact that currently available treatments work for a number of people with chronic PTSD. But because there are many more for whom [the treatments] don’t work, or they’re insufficiently helped by those treatments, this is certainly one potentially very promising approach that can be added” to a clinician’s toolbox, Dr. Feder said.
Speaks to clinical utility
Commenting for this news organization, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University, New Haven, Connecticut, called this a “very solid and well-designed” study.
“It definitely builds on what’s been found in the past, but it’s a critical piece of information speaking to the clinical utility of this treatment for PTSD,” said Dr. Sanacora, who is also director of the Yale Depression Research Program and was not involved with the current research.
He agreed with the investigators that PTSD has long been a condition that is difficult to treat.
“It’s an area that has a great unmet need for treatment options. Beyond that, as ketamine is becoming more widely used, there’s increasing demand for off-label uses. This [study] actually provides some evidence that there may be efficacy there,” Dr. Sanacora said.
Although he cautioned that this was a small study, and thus further research with a larger patient population will be needed, it provides a compelling foundation to build upon.
“This study provides clear evidence to support a larger study to really give a definitive statement on the efficacy and safety of its use for PTSD. I don’t think this is the study that provides that definitive evidence, but it is a very strong indication, and it very strongly supports the initiation of a large study to address that,” said Dr. Sanacora.
He noted that, although he’s used the term “cautious optimism” for studies in the past, he has “real optimism” that ketamine will be effective for PTSD based on the results of this current study.
“We still need some more data to really convince us of that before we can say with any clear statement that it is effective and safe, but I’m very optimistic,” Dr. Sanacora concluded.
The study was funded by the Brain and Behavior Research Foundation, Mount Sinai Innovation Partners and the Mount Sinai i3 Accelerator, Gerald and Glenda Greenwald, and the Ehrenkranz Laboratory for Human Resilience. Dr. Feder is a coinventor on issued patents for the use of ketamine as therapy for PTSD. A list of all disclosures for the other study authors are listed in the original article.
A version of this article first appeared on Medscape.com.
Repeated intravenous infusions of ketamine provide rapid relief for patients with posttraumatic stress disorder, new research suggests.
In what investigators are calling the first randomized controlled trial of repeated ketamine administration for chronic PTSD, 30 patients received six infusions of ketamine or midazolam (used as a psychoactive placebo) over 2 consecutive weeks.
Between baseline and week 2, those receiving ketamine showed significantly greater improvement than those receiving midazolam. Total scores on the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5) for the first group were almost 12 points lower than the latter group at week 2, meeting the study’s primary outcome measure.
In addition, 67% vs. 20% of the patients, respectively, were considered to be treatment responders; time to loss of response for those in the ketamine group was 28 days.
Although the overall findings were as expected, “what was surprising was how robust the results were,” lead author Adriana Feder, MD, associate professor of psychiatry, Icahn School of Medicine, Mount Sinai, New York, told this news organization.
It was also a bit surprising that, in a study of just 30 participants, “we were able to show such a clear difference” between the two treatment groups, said Dr. Feder, who is also a coinventor on issued patents for the use of ketamine as therapy for PTSD, and codirector of the Ehrenkranz Lab for the Study of Human Resilience at Mount Sinai.
The findings were published online Jan. 5 in the American Journal of Psychiatry.
Unmet need
Ketamine is a glutamate N-methyl-D-aspartate (NMDA) receptor antagonist that was first approved by the U.S. Food and Drug Administration for anesthetic use in 1970. It has also been shown to be effective for treatment-resistant depression.
PTSD has a lifetime prevalence of about 6% in the United States. “While trauma-focused psychotherapies have the most empirical support, they are limited by significant rates of nonresponse, partial response, and treatment dropout,” the investigators write. Also, there are “few available pharmacotherapies for PTSD, and their efficacy is insufficient,” they add.
“There’s a real need for new treatment interventions that are effective for PTSD and also work rapidly, because it can take weeks to months for currently available treatments to work for PTSD,” Dr. Feder said.
The researchers previously conducted a “proof-of-concept” randomized controlled trial of single infusions of ketamine for chronic PTSD. Results published in 2014 in JAMA Psychiatry showed significant reduction in PTSD symptoms 24 hours after infusion.
For the current study, the investigative team wanted to assess whether ketamine was viable as a longer-term treatment.
“We were encouraged by our initial promising findings” of the earlier trial, Dr. Feder said. “We wanted to do the second study to see if ketamine really works for PTSD, to see if we could replicate the rapid improvement and also examine whether a course of six infusions over 2 weeks could maintain the improvement.”
Thirty patients (aged 18-70; mean age, 39 years) with chronic PTSD from civilian or military trauma were enrolled (mean PTSD duration, 15 years).
The most cited primary trauma was sexual assault or molestation (n = 13), followed by physical assault or abuse (n = 8), witnessing a violent assault or death (n = 4), witnessing the 9/11 attacks (n = 3), and combat exposure (n = 2).
During the 2-week treatment phase, half of the patients were randomly assigned to receive six infusions of ketamine hydrochloride at a dose of 0.5 mg/kg (86.7% women; mean CAPS-5 score, 42), while the other half received six infusions of midazolam at a dose of 0.045 mg/kg (66.7% women; mean CAPS-5 score, 40).
In addition to the primary outcome measure of 2-week changes on the CAPS-5, secondary outcomes included score changes on the Montgomery-Åsberg Depression Rating Scale (MADRS) and the Impact of Event Scale-Revised (IES-R).
Treatment response was defined as a 30% or more improvement in symptoms on the CAPS-5. A number of measures were also used to assess potential treatment-related adverse events (AEs).
Safe, effective
Results showed significantly lower total CAPS-5 scores for the ketamine group vs. the midazolam group at week 1 (score difference, 8.8 points; P = .03) and at week 2 (score difference, 11.88 points; P = .004).
Those receiving ketamine also showed improvements in three of the four PTSD symptom clusters on the CAPS-5: avoidance (P < .0001), negative mood and cognitions (P = .02), and intrusions (P = .03). The fourth symptom cluster – arousal and reactivity – did not show a significant improvement.
In addition, the ketamine group showed significantly greater improvement scores on the MADRS at both week 1 and week 2.
Treatment response at 2 weeks was achieved by 10 members of the ketamine group and by three members of the midazolam group (P = .03).
Secondary analyses showed rapid improvement in the treatment responders within the ketamine group, with a mean change of 26 points on the total IES-R score between baseline and 24 hours after their first infusion, and a mean change of 13.4 points on the MADRS total past-24-hour score, a 53% improvement on average.
“A response at 2 weeks is very rapid but they got better sometimes within the first day,” Dr. Feder noted.
There were no serious AEs reported. Although some dissociative symptoms occurred during ketamine infusions, with the highest levels reported at the end of the infusion, these symptoms had resolved by the next assessment, conducted 2 hours after infusion.
The most frequently reported AE in the ketamine group, compared with midazolam, after the start of infusions was blurred vision (53% vs. 0%), followed by dizziness (33% vs. 13%), fatigue (33% vs. 87%), headache (27% vs. 13%), and nausea or vomiting (20% vs. 7%).
‘Large-magnitude improvement’
The overall findings show that, in this patient population, “repeated intravenous ketamine infusions administered over 2 weeks were associated with a large-magnitude, clinically significant improvement in PTSD symptoms,” the investigators write.
The results “were very satisfying,” added Dr. Feder. “It was heartening also to hear what some of the participants would say. Some told us about how their symptoms and feelings had changed during the course of treatment with ketamine, where they felt stronger and better able to cope with their trauma and memories.”
She noted, however, that this was not a study designed to specifically assess ketamine in treatment-resistant PTSD. “Some patients had had multiple treatments before that hadn’t worked, while others had not received treatment before. Efficacy for treatment-resistant PTSD is an important question for future research,” Dr. Feder said.
Other areas worth future exploration include treatment efficacy in patients with different types of trauma and whether outcomes can last longer in patients receiving ketamine plus psychotherapy treatment, she noted.
“I don’t want to ignore the fact that currently available treatments work for a number of people with chronic PTSD. But because there are many more for whom [the treatments] don’t work, or they’re insufficiently helped by those treatments, this is certainly one potentially very promising approach that can be added” to a clinician’s toolbox, Dr. Feder said.
Speaks to clinical utility
Commenting for this news organization, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University, New Haven, Connecticut, called this a “very solid and well-designed” study.
“It definitely builds on what’s been found in the past, but it’s a critical piece of information speaking to the clinical utility of this treatment for PTSD,” said Dr. Sanacora, who is also director of the Yale Depression Research Program and was not involved with the current research.
He agreed with the investigators that PTSD has long been a condition that is difficult to treat.
“It’s an area that has a great unmet need for treatment options. Beyond that, as ketamine is becoming more widely used, there’s increasing demand for off-label uses. This [study] actually provides some evidence that there may be efficacy there,” Dr. Sanacora said.
Although he cautioned that this was a small study, and thus further research with a larger patient population will be needed, it provides a compelling foundation to build upon.
“This study provides clear evidence to support a larger study to really give a definitive statement on the efficacy and safety of its use for PTSD. I don’t think this is the study that provides that definitive evidence, but it is a very strong indication, and it very strongly supports the initiation of a large study to address that,” said Dr. Sanacora.
He noted that, although he’s used the term “cautious optimism” for studies in the past, he has “real optimism” that ketamine will be effective for PTSD based on the results of this current study.
“We still need some more data to really convince us of that before we can say with any clear statement that it is effective and safe, but I’m very optimistic,” Dr. Sanacora concluded.
The study was funded by the Brain and Behavior Research Foundation, Mount Sinai Innovation Partners and the Mount Sinai i3 Accelerator, Gerald and Glenda Greenwald, and the Ehrenkranz Laboratory for Human Resilience. Dr. Feder is a coinventor on issued patents for the use of ketamine as therapy for PTSD. A list of all disclosures for the other study authors are listed in the original article.
A version of this article first appeared on Medscape.com.
Repeated intravenous infusions of ketamine provide rapid relief for patients with posttraumatic stress disorder, new research suggests.
In what investigators are calling the first randomized controlled trial of repeated ketamine administration for chronic PTSD, 30 patients received six infusions of ketamine or midazolam (used as a psychoactive placebo) over 2 consecutive weeks.
Between baseline and week 2, those receiving ketamine showed significantly greater improvement than those receiving midazolam. Total scores on the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5) for the first group were almost 12 points lower than the latter group at week 2, meeting the study’s primary outcome measure.
In addition, 67% vs. 20% of the patients, respectively, were considered to be treatment responders; time to loss of response for those in the ketamine group was 28 days.
Although the overall findings were as expected, “what was surprising was how robust the results were,” lead author Adriana Feder, MD, associate professor of psychiatry, Icahn School of Medicine, Mount Sinai, New York, told this news organization.
It was also a bit surprising that, in a study of just 30 participants, “we were able to show such a clear difference” between the two treatment groups, said Dr. Feder, who is also a coinventor on issued patents for the use of ketamine as therapy for PTSD, and codirector of the Ehrenkranz Lab for the Study of Human Resilience at Mount Sinai.
The findings were published online Jan. 5 in the American Journal of Psychiatry.
Unmet need
Ketamine is a glutamate N-methyl-D-aspartate (NMDA) receptor antagonist that was first approved by the U.S. Food and Drug Administration for anesthetic use in 1970. It has also been shown to be effective for treatment-resistant depression.
PTSD has a lifetime prevalence of about 6% in the United States. “While trauma-focused psychotherapies have the most empirical support, they are limited by significant rates of nonresponse, partial response, and treatment dropout,” the investigators write. Also, there are “few available pharmacotherapies for PTSD, and their efficacy is insufficient,” they add.
“There’s a real need for new treatment interventions that are effective for PTSD and also work rapidly, because it can take weeks to months for currently available treatments to work for PTSD,” Dr. Feder said.
The researchers previously conducted a “proof-of-concept” randomized controlled trial of single infusions of ketamine for chronic PTSD. Results published in 2014 in JAMA Psychiatry showed significant reduction in PTSD symptoms 24 hours after infusion.
For the current study, the investigative team wanted to assess whether ketamine was viable as a longer-term treatment.
“We were encouraged by our initial promising findings” of the earlier trial, Dr. Feder said. “We wanted to do the second study to see if ketamine really works for PTSD, to see if we could replicate the rapid improvement and also examine whether a course of six infusions over 2 weeks could maintain the improvement.”
Thirty patients (aged 18-70; mean age, 39 years) with chronic PTSD from civilian or military trauma were enrolled (mean PTSD duration, 15 years).
The most cited primary trauma was sexual assault or molestation (n = 13), followed by physical assault or abuse (n = 8), witnessing a violent assault or death (n = 4), witnessing the 9/11 attacks (n = 3), and combat exposure (n = 2).
During the 2-week treatment phase, half of the patients were randomly assigned to receive six infusions of ketamine hydrochloride at a dose of 0.5 mg/kg (86.7% women; mean CAPS-5 score, 42), while the other half received six infusions of midazolam at a dose of 0.045 mg/kg (66.7% women; mean CAPS-5 score, 40).
In addition to the primary outcome measure of 2-week changes on the CAPS-5, secondary outcomes included score changes on the Montgomery-Åsberg Depression Rating Scale (MADRS) and the Impact of Event Scale-Revised (IES-R).
Treatment response was defined as a 30% or more improvement in symptoms on the CAPS-5. A number of measures were also used to assess potential treatment-related adverse events (AEs).
Safe, effective
Results showed significantly lower total CAPS-5 scores for the ketamine group vs. the midazolam group at week 1 (score difference, 8.8 points; P = .03) and at week 2 (score difference, 11.88 points; P = .004).
Those receiving ketamine also showed improvements in three of the four PTSD symptom clusters on the CAPS-5: avoidance (P < .0001), negative mood and cognitions (P = .02), and intrusions (P = .03). The fourth symptom cluster – arousal and reactivity – did not show a significant improvement.
In addition, the ketamine group showed significantly greater improvement scores on the MADRS at both week 1 and week 2.
Treatment response at 2 weeks was achieved by 10 members of the ketamine group and by three members of the midazolam group (P = .03).
Secondary analyses showed rapid improvement in the treatment responders within the ketamine group, with a mean change of 26 points on the total IES-R score between baseline and 24 hours after their first infusion, and a mean change of 13.4 points on the MADRS total past-24-hour score, a 53% improvement on average.
“A response at 2 weeks is very rapid but they got better sometimes within the first day,” Dr. Feder noted.
There were no serious AEs reported. Although some dissociative symptoms occurred during ketamine infusions, with the highest levels reported at the end of the infusion, these symptoms had resolved by the next assessment, conducted 2 hours after infusion.
The most frequently reported AE in the ketamine group, compared with midazolam, after the start of infusions was blurred vision (53% vs. 0%), followed by dizziness (33% vs. 13%), fatigue (33% vs. 87%), headache (27% vs. 13%), and nausea or vomiting (20% vs. 7%).
‘Large-magnitude improvement’
The overall findings show that, in this patient population, “repeated intravenous ketamine infusions administered over 2 weeks were associated with a large-magnitude, clinically significant improvement in PTSD symptoms,” the investigators write.
The results “were very satisfying,” added Dr. Feder. “It was heartening also to hear what some of the participants would say. Some told us about how their symptoms and feelings had changed during the course of treatment with ketamine, where they felt stronger and better able to cope with their trauma and memories.”
She noted, however, that this was not a study designed to specifically assess ketamine in treatment-resistant PTSD. “Some patients had had multiple treatments before that hadn’t worked, while others had not received treatment before. Efficacy for treatment-resistant PTSD is an important question for future research,” Dr. Feder said.
Other areas worth future exploration include treatment efficacy in patients with different types of trauma and whether outcomes can last longer in patients receiving ketamine plus psychotherapy treatment, she noted.
“I don’t want to ignore the fact that currently available treatments work for a number of people with chronic PTSD. But because there are many more for whom [the treatments] don’t work, or they’re insufficiently helped by those treatments, this is certainly one potentially very promising approach that can be added” to a clinician’s toolbox, Dr. Feder said.
Speaks to clinical utility
Commenting for this news organization, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University, New Haven, Connecticut, called this a “very solid and well-designed” study.
“It definitely builds on what’s been found in the past, but it’s a critical piece of information speaking to the clinical utility of this treatment for PTSD,” said Dr. Sanacora, who is also director of the Yale Depression Research Program and was not involved with the current research.
He agreed with the investigators that PTSD has long been a condition that is difficult to treat.
“It’s an area that has a great unmet need for treatment options. Beyond that, as ketamine is becoming more widely used, there’s increasing demand for off-label uses. This [study] actually provides some evidence that there may be efficacy there,” Dr. Sanacora said.
Although he cautioned that this was a small study, and thus further research with a larger patient population will be needed, it provides a compelling foundation to build upon.
“This study provides clear evidence to support a larger study to really give a definitive statement on the efficacy and safety of its use for PTSD. I don’t think this is the study that provides that definitive evidence, but it is a very strong indication, and it very strongly supports the initiation of a large study to address that,” said Dr. Sanacora.
He noted that, although he’s used the term “cautious optimism” for studies in the past, he has “real optimism” that ketamine will be effective for PTSD based on the results of this current study.
“We still need some more data to really convince us of that before we can say with any clear statement that it is effective and safe, but I’m very optimistic,” Dr. Sanacora concluded.
The study was funded by the Brain and Behavior Research Foundation, Mount Sinai Innovation Partners and the Mount Sinai i3 Accelerator, Gerald and Glenda Greenwald, and the Ehrenkranz Laboratory for Human Resilience. Dr. Feder is a coinventor on issued patents for the use of ketamine as therapy for PTSD. A list of all disclosures for the other study authors are listed in the original article.
A version of this article first appeared on Medscape.com.
PCPs play a small part in low-value care spending
according to a brief report published online Jan. 18 in Annals of Internal Medicine.
However, one expert said there are better ways to curb low-value care than focusing on which specialties are guilty of the practice.
Analyzing a 20% random sample of Medicare Part B claims, Aaron Baum, PhD, with the Icahn School of Medicine at Mount Sinai, New York, and colleagues found that the services primary care physicians performed or ordered made up on average 8.3% of the low-value care their patients received (interquartile range, 3.9%-15.1%; 95th percentile, 35.6%) and their referrals made up 15.4% (IQR, 6.3%-26.4%; 95th percentile, 44.6%).
By specialty, cardiology had the worst record with 27% of all spending on low-value services ($1.8 billion) attributed to that specialty. Yet, of the 25 highest-spending specialties in the report, 12 of them were associated with 1% or less than 1% each of all low-value spending, indicating the waste was widely distributed.
Dr. Baum said in an interview that though there are some PCPs guilty of high spending on low-value services, overall, most primary care physicians’ low-value services add up to only 0.3% of Part B spending. He noted that Part B spending is about one-third of all Medicare spending.
Primary care is often thought to be at the core of care management and spending and PCPs are often seen as the gatekeepers, but this analysis suggests that efforts to make big differences in curtailing low-value spending might be more effective elsewhere.
“There’s only so much spending you can reduce by changing primary care physicians’ services that they directly perform,” Dr. Baum said.
Low-value care is costly, can be harmful
Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, said in an interview that the report adds confirmation to previous research that has consistently shown low-value care is “extremely common, very costly, and provided by primary care providers and specialists alike.” He noted that it can also be harmful.
“The math is simple,” he said. “If we want to improve coverage and lower patient costs for essential services like visits, diagnostic tests, and drugs, we have to reduce spending on those services that do not make Americans any healthier.”
The study ranked 31 clinical services judged to be low value by physician societies, Medicare and clinical guidelines, and their use among beneficiaries enrolled between 2007 and 2014. Here’s how the top six low-value services compare.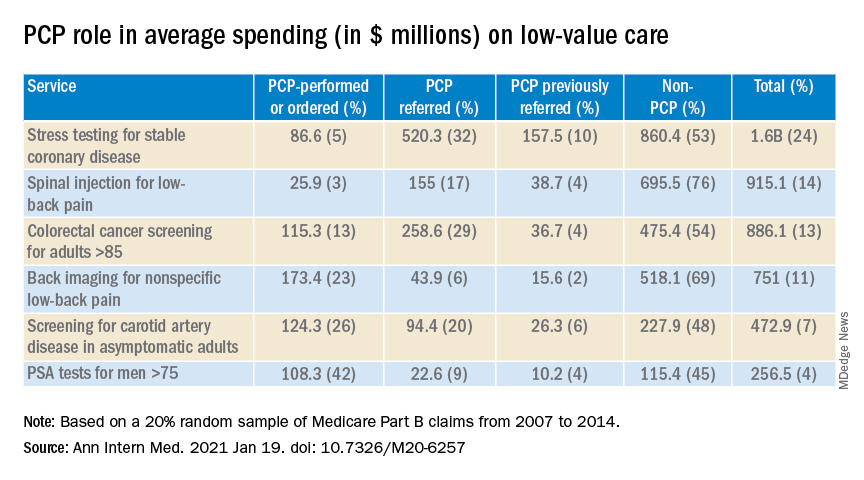
Dr. Fendrick said a weakness of the paper is the years of the data (2007-2014). Some of the criteria around low-value care have changed since then. The age that a prostate-specific antigen test becomes low-value is now 70 years, for instance, instead of 75. He added that some of the figures attributed to non-PCP providers appear out of date.
Dr. Fendrick said, “I understand that there are Medicare patients who end up at a gastroenterologist or surgeon’s office to get colorectal cancer screening, but it would be very hard for me to believe that half of stress tests and over half of colon cancer screening over [age] 85 [years] and half of PSA for people over 75 did not have some type of referring clinicians involved. I certainly don’t think that would be the case in 2020-2021.”
Dr. Baum said those years were the latest years available for the data points needed for this analysis, but he and his colleagues were working to update the data for future publication.
Dr. Fendrick said not much has changed in recent years in terms of waste on low-value care, even with campaigns such as Choosing Wisely dedicated to identifying low-value services or procedures in each specialty.
“I believe there’s not a particular group of clinicians one way or the other who are actually doing any better now than they were 7 years ago,” he said. He would rather focus less on which specialties are associated with the most low-value care and more on the underlying policies that encourage low-value care.
“If you’re going to get paid for doing a stress test and get paid nothing or significantly less if you don’t, the incentives are in the wrong direction,” he said.
Dr. Fendrick said the pandemic era provides an opportunity to eliminate low-value care because use of those services has dropped drastically as resources have been diverted to COVID-19 patients and many services have been delayed or canceled.
He said he has been pushing an approach that providers should be paid more after the pandemic “to do the things we want them to do.”
As an example, he said, instead of paying $886 million on colonoscopies for people over the age of 85, “why don’t we put a policy in place that would make it better for patients by lowering cost sharing and better for providers by paying them more to do the service on the people who need it as opposed to the people who don’t?”
The research was funded by the American Board of Family Medicine Foundation. Dr. Baum and a coauthor reported receiving personal fees from American Board of Family Medicine Foundation during the conduct of the study. Another coauthor reported receiving personal fees from Collective Health, HealthRight 360, PLOS Medicine, and the New England Journal of Medicine, outside the submitted work. Dr. Fendrick disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a brief report published online Jan. 18 in Annals of Internal Medicine.
However, one expert said there are better ways to curb low-value care than focusing on which specialties are guilty of the practice.
Analyzing a 20% random sample of Medicare Part B claims, Aaron Baum, PhD, with the Icahn School of Medicine at Mount Sinai, New York, and colleagues found that the services primary care physicians performed or ordered made up on average 8.3% of the low-value care their patients received (interquartile range, 3.9%-15.1%; 95th percentile, 35.6%) and their referrals made up 15.4% (IQR, 6.3%-26.4%; 95th percentile, 44.6%).
By specialty, cardiology had the worst record with 27% of all spending on low-value services ($1.8 billion) attributed to that specialty. Yet, of the 25 highest-spending specialties in the report, 12 of them were associated with 1% or less than 1% each of all low-value spending, indicating the waste was widely distributed.
Dr. Baum said in an interview that though there are some PCPs guilty of high spending on low-value services, overall, most primary care physicians’ low-value services add up to only 0.3% of Part B spending. He noted that Part B spending is about one-third of all Medicare spending.
Primary care is often thought to be at the core of care management and spending and PCPs are often seen as the gatekeepers, but this analysis suggests that efforts to make big differences in curtailing low-value spending might be more effective elsewhere.
“There’s only so much spending you can reduce by changing primary care physicians’ services that they directly perform,” Dr. Baum said.
Low-value care is costly, can be harmful
Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, said in an interview that the report adds confirmation to previous research that has consistently shown low-value care is “extremely common, very costly, and provided by primary care providers and specialists alike.” He noted that it can also be harmful.
“The math is simple,” he said. “If we want to improve coverage and lower patient costs for essential services like visits, diagnostic tests, and drugs, we have to reduce spending on those services that do not make Americans any healthier.”
The study ranked 31 clinical services judged to be low value by physician societies, Medicare and clinical guidelines, and their use among beneficiaries enrolled between 2007 and 2014. Here’s how the top six low-value services compare.
Dr. Fendrick said a weakness of the paper is the years of the data (2007-2014). Some of the criteria around low-value care have changed since then. The age that a prostate-specific antigen test becomes low-value is now 70 years, for instance, instead of 75. He added that some of the figures attributed to non-PCP providers appear out of date.
Dr. Fendrick said, “I understand that there are Medicare patients who end up at a gastroenterologist or surgeon’s office to get colorectal cancer screening, but it would be very hard for me to believe that half of stress tests and over half of colon cancer screening over [age] 85 [years] and half of PSA for people over 75 did not have some type of referring clinicians involved. I certainly don’t think that would be the case in 2020-2021.”
Dr. Baum said those years were the latest years available for the data points needed for this analysis, but he and his colleagues were working to update the data for future publication.
Dr. Fendrick said not much has changed in recent years in terms of waste on low-value care, even with campaigns such as Choosing Wisely dedicated to identifying low-value services or procedures in each specialty.
“I believe there’s not a particular group of clinicians one way or the other who are actually doing any better now than they were 7 years ago,” he said. He would rather focus less on which specialties are associated with the most low-value care and more on the underlying policies that encourage low-value care.
“If you’re going to get paid for doing a stress test and get paid nothing or significantly less if you don’t, the incentives are in the wrong direction,” he said.
Dr. Fendrick said the pandemic era provides an opportunity to eliminate low-value care because use of those services has dropped drastically as resources have been diverted to COVID-19 patients and many services have been delayed or canceled.
He said he has been pushing an approach that providers should be paid more after the pandemic “to do the things we want them to do.”
As an example, he said, instead of paying $886 million on colonoscopies for people over the age of 85, “why don’t we put a policy in place that would make it better for patients by lowering cost sharing and better for providers by paying them more to do the service on the people who need it as opposed to the people who don’t?”
The research was funded by the American Board of Family Medicine Foundation. Dr. Baum and a coauthor reported receiving personal fees from American Board of Family Medicine Foundation during the conduct of the study. Another coauthor reported receiving personal fees from Collective Health, HealthRight 360, PLOS Medicine, and the New England Journal of Medicine, outside the submitted work. Dr. Fendrick disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a brief report published online Jan. 18 in Annals of Internal Medicine.
However, one expert said there are better ways to curb low-value care than focusing on which specialties are guilty of the practice.
Analyzing a 20% random sample of Medicare Part B claims, Aaron Baum, PhD, with the Icahn School of Medicine at Mount Sinai, New York, and colleagues found that the services primary care physicians performed or ordered made up on average 8.3% of the low-value care their patients received (interquartile range, 3.9%-15.1%; 95th percentile, 35.6%) and their referrals made up 15.4% (IQR, 6.3%-26.4%; 95th percentile, 44.6%).
By specialty, cardiology had the worst record with 27% of all spending on low-value services ($1.8 billion) attributed to that specialty. Yet, of the 25 highest-spending specialties in the report, 12 of them were associated with 1% or less than 1% each of all low-value spending, indicating the waste was widely distributed.
Dr. Baum said in an interview that though there are some PCPs guilty of high spending on low-value services, overall, most primary care physicians’ low-value services add up to only 0.3% of Part B spending. He noted that Part B spending is about one-third of all Medicare spending.
Primary care is often thought to be at the core of care management and spending and PCPs are often seen as the gatekeepers, but this analysis suggests that efforts to make big differences in curtailing low-value spending might be more effective elsewhere.
“There’s only so much spending you can reduce by changing primary care physicians’ services that they directly perform,” Dr. Baum said.
Low-value care is costly, can be harmful
Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, said in an interview that the report adds confirmation to previous research that has consistently shown low-value care is “extremely common, very costly, and provided by primary care providers and specialists alike.” He noted that it can also be harmful.
“The math is simple,” he said. “If we want to improve coverage and lower patient costs for essential services like visits, diagnostic tests, and drugs, we have to reduce spending on those services that do not make Americans any healthier.”
The study ranked 31 clinical services judged to be low value by physician societies, Medicare and clinical guidelines, and their use among beneficiaries enrolled between 2007 and 2014. Here’s how the top six low-value services compare.
Dr. Fendrick said a weakness of the paper is the years of the data (2007-2014). Some of the criteria around low-value care have changed since then. The age that a prostate-specific antigen test becomes low-value is now 70 years, for instance, instead of 75. He added that some of the figures attributed to non-PCP providers appear out of date.
Dr. Fendrick said, “I understand that there are Medicare patients who end up at a gastroenterologist or surgeon’s office to get colorectal cancer screening, but it would be very hard for me to believe that half of stress tests and over half of colon cancer screening over [age] 85 [years] and half of PSA for people over 75 did not have some type of referring clinicians involved. I certainly don’t think that would be the case in 2020-2021.”
Dr. Baum said those years were the latest years available for the data points needed for this analysis, but he and his colleagues were working to update the data for future publication.
Dr. Fendrick said not much has changed in recent years in terms of waste on low-value care, even with campaigns such as Choosing Wisely dedicated to identifying low-value services or procedures in each specialty.
“I believe there’s not a particular group of clinicians one way or the other who are actually doing any better now than they were 7 years ago,” he said. He would rather focus less on which specialties are associated with the most low-value care and more on the underlying policies that encourage low-value care.
“If you’re going to get paid for doing a stress test and get paid nothing or significantly less if you don’t, the incentives are in the wrong direction,” he said.
Dr. Fendrick said the pandemic era provides an opportunity to eliminate low-value care because use of those services has dropped drastically as resources have been diverted to COVID-19 patients and many services have been delayed or canceled.
He said he has been pushing an approach that providers should be paid more after the pandemic “to do the things we want them to do.”
As an example, he said, instead of paying $886 million on colonoscopies for people over the age of 85, “why don’t we put a policy in place that would make it better for patients by lowering cost sharing and better for providers by paying them more to do the service on the people who need it as opposed to the people who don’t?”
The research was funded by the American Board of Family Medicine Foundation. Dr. Baum and a coauthor reported receiving personal fees from American Board of Family Medicine Foundation during the conduct of the study. Another coauthor reported receiving personal fees from Collective Health, HealthRight 360, PLOS Medicine, and the New England Journal of Medicine, outside the submitted work. Dr. Fendrick disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patient Handout: Safe practices during the COVID-19 pandemic
In addition to sharing this handout (see PDF link) with your patients, Dr. Gupta also recommends advising them to watch the video Hand-washing Steps Using the WHO Technique, which is available at https://youtu.be/IisgnbMfKvI
In addition to sharing this handout (see PDF link) with your patients, Dr. Gupta also recommends advising them to watch the video Hand-washing Steps Using the WHO Technique, which is available at https://youtu.be/IisgnbMfKvI
In addition to sharing this handout (see PDF link) with your patients, Dr. Gupta also recommends advising them to watch the video Hand-washing Steps Using the WHO Technique, which is available at https://youtu.be/IisgnbMfKvI
President Biden kicks off health agenda with COVID actions, WHO outreach
President Joe Biden kicked off his new administration Jan. 20 with an immediate focus on attempts to stop the spread of COVID-19, including closer coordination with other nations.
Mr. Biden signed 17 executive orders, memoranda, and directives addressing not only the pandemic but also economic concerns, climate change, and racial inequity.
At the top of the list of actions was what his transition team called a “100 Days Masking Challenge.” Mr. Biden issued an executive order requiring masks and physical distancing in all federal buildings, on all federal lands, and by federal employees and contractors.
The president also halted the Trump administration’s process of withdrawing from the World Health Organization. Instead, Mr. Biden named Anthony Fauci, MD, the director of the National Institute for Allergy and Infectious Diseases, as the head of a delegation to participate in the WHO executive board meeting that is being held this week.
Mr. Biden also signed an executive order creating the position of COVID-19 response coordinator, which will report directly to the president and be responsible for coordinating all elements of the COVID-19 response across government, including the production and distribution of vaccines and medical supplies.
The newly inaugurated president also intends to restore the National Security Council’s Directorate for Global Health Security and Biodefense, which will aid in the response to the pandemic, his transition team said.
The American Medical Association was among the first to commend the first-day actions.
“Defeating COVID-19 requires bold, coordinated federal leadership and strong adherence to the public health steps we know stop the spread of this virus – wearing masks, practicing physical distancing, and washing hands,” said AMA President Susan R. Bailey, MD in a news release. “We are pleased by the Biden administration’s steps today, including universal mask wearing within federal jurisdictions, providing federal leadership for COVID-19 response, and reengaging with the World Health Organization. Taking these actions on day 1 of the administration sends the right message – that our nation is laser focused on stopping the ravages of COVID-19.”
A version of this article first appeared on Medscape.com.
President Joe Biden kicked off his new administration Jan. 20 with an immediate focus on attempts to stop the spread of COVID-19, including closer coordination with other nations.
Mr. Biden signed 17 executive orders, memoranda, and directives addressing not only the pandemic but also economic concerns, climate change, and racial inequity.
At the top of the list of actions was what his transition team called a “100 Days Masking Challenge.” Mr. Biden issued an executive order requiring masks and physical distancing in all federal buildings, on all federal lands, and by federal employees and contractors.
The president also halted the Trump administration’s process of withdrawing from the World Health Organization. Instead, Mr. Biden named Anthony Fauci, MD, the director of the National Institute for Allergy and Infectious Diseases, as the head of a delegation to participate in the WHO executive board meeting that is being held this week.
Mr. Biden also signed an executive order creating the position of COVID-19 response coordinator, which will report directly to the president and be responsible for coordinating all elements of the COVID-19 response across government, including the production and distribution of vaccines and medical supplies.
The newly inaugurated president also intends to restore the National Security Council’s Directorate for Global Health Security and Biodefense, which will aid in the response to the pandemic, his transition team said.
The American Medical Association was among the first to commend the first-day actions.
“Defeating COVID-19 requires bold, coordinated federal leadership and strong adherence to the public health steps we know stop the spread of this virus – wearing masks, practicing physical distancing, and washing hands,” said AMA President Susan R. Bailey, MD in a news release. “We are pleased by the Biden administration’s steps today, including universal mask wearing within federal jurisdictions, providing federal leadership for COVID-19 response, and reengaging with the World Health Organization. Taking these actions on day 1 of the administration sends the right message – that our nation is laser focused on stopping the ravages of COVID-19.”
A version of this article first appeared on Medscape.com.
President Joe Biden kicked off his new administration Jan. 20 with an immediate focus on attempts to stop the spread of COVID-19, including closer coordination with other nations.
Mr. Biden signed 17 executive orders, memoranda, and directives addressing not only the pandemic but also economic concerns, climate change, and racial inequity.
At the top of the list of actions was what his transition team called a “100 Days Masking Challenge.” Mr. Biden issued an executive order requiring masks and physical distancing in all federal buildings, on all federal lands, and by federal employees and contractors.
The president also halted the Trump administration’s process of withdrawing from the World Health Organization. Instead, Mr. Biden named Anthony Fauci, MD, the director of the National Institute for Allergy and Infectious Diseases, as the head of a delegation to participate in the WHO executive board meeting that is being held this week.
Mr. Biden also signed an executive order creating the position of COVID-19 response coordinator, which will report directly to the president and be responsible for coordinating all elements of the COVID-19 response across government, including the production and distribution of vaccines and medical supplies.
The newly inaugurated president also intends to restore the National Security Council’s Directorate for Global Health Security and Biodefense, which will aid in the response to the pandemic, his transition team said.
The American Medical Association was among the first to commend the first-day actions.
“Defeating COVID-19 requires bold, coordinated federal leadership and strong adherence to the public health steps we know stop the spread of this virus – wearing masks, practicing physical distancing, and washing hands,” said AMA President Susan R. Bailey, MD in a news release. “We are pleased by the Biden administration’s steps today, including universal mask wearing within federal jurisdictions, providing federal leadership for COVID-19 response, and reengaging with the World Health Organization. Taking these actions on day 1 of the administration sends the right message – that our nation is laser focused on stopping the ravages of COVID-19.”
A version of this article first appeared on Medscape.com.
APA apologizes for past support of racism in psychiatry
The American Psychiatric Association has issued a formal apology for its past support of structural racism in psychiatry.
The apology, issued Jan. 18, coincided with the federal holiday honoring the life and work of civil rights activist Dr. Martin Luther King Jr.
“We apologize for our role in perpetrating structural racism in this country, and we hope to begin to make amends for APA’s and psychiatry’s history of actions, intentional and not, that hurt Black, indigenous, and people of color,” APA President Jeffrey Geller, MD, MPH, said in a statement.
The apology was written and issued by the APA Board of Trustees. It acknowledges practices and events in psychiatry that contributed to racial inequality, and expresses the organization’s commitment to developing antiracist policies that promote equity in mental health for all.
“This apology is one important step we needed to take to move forward to a more equitable future. The board is issuing this document on Martin Luther King Jr. Day, because we hope that it honors his life’s work of reconciliation and equality. We do not take that legacy or his call to action lightly and will continue our important work,” said Dr. Geller.
One involved the Eastern State Hospital in Williamsburg, Va., the nation’s first psychiatric care facility, founded in 1773.
Eastern State, which for a time in the 1800s was called the Eastern Lunatic Asylum, was not segregated when founded. However, 70 years later, when the 13 founders of what is now the APA met to discuss improvements in mental health care delivery, the treatment system they created and the organization they founded aligned with that era’s racist social and political policies. In this system, Black patients received psychiatric care separately from White patients, the APA said.
The APA also acknowledged failing to act in Black Americans’ best interest at critical points in the United States’ sociopolitical evolution throughout the 19th and 20th centuries.
“This inactivity was notably evident while white supremacists lynched Black people during the Reconstruction Era as well as when Jim Crow segregation was in effect, which led to ‘separate but equal’ standards of care starting in 1896,” the APA said.
Later, the APA failed to declare support for Brown v. Board of Education of Topeka in 1954, along with further major civil rights legislation designed to improve social and psychological conditions for Black people, the organization admitted.
Throughout the decades that followed, psychiatric misdiagnosis among Black, indigenous, and people of color populations were also common, the APA acknowledged.
For example, late 20th century psychiatrists commonly attributed their minority patients’ frustrations to schizophrenia, while categorizing similar behaviors as “neuroticism” in White patients.
The APA pointed to one study which found that APA members diagnosed more Black than White patients with schizophrenia, even when both had otherwise identical clinical presentations.
“This reveals the basis for embedded discrimination within psychiatry that has contributed to reduced quality of care” for Black, indigenous, and people of color, and “perpetuation of dangerous stereotypes,” the APA said.
Saul Levin, MD, the APA’s medical director and CEO, said the Board of Trustees has taken “an important step in issuing this apology. The APA administration is committed to working toward inclusion, health equity, and fairness that everyone deserves.”
The APA Board of Trustees began drafting the apology late last year after it concluded that events and persistent inequities in health care and psychiatry had highlighted an organizational need for action.
The APA’s Presidential Task Force on Structural Racism is continuing with efforts to educate and engage members on the issue and implement changes within the organization.
A version of this article first appeared on Medscape.com.
The American Psychiatric Association has issued a formal apology for its past support of structural racism in psychiatry.
The apology, issued Jan. 18, coincided with the federal holiday honoring the life and work of civil rights activist Dr. Martin Luther King Jr.
“We apologize for our role in perpetrating structural racism in this country, and we hope to begin to make amends for APA’s and psychiatry’s history of actions, intentional and not, that hurt Black, indigenous, and people of color,” APA President Jeffrey Geller, MD, MPH, said in a statement.
The apology was written and issued by the APA Board of Trustees. It acknowledges practices and events in psychiatry that contributed to racial inequality, and expresses the organization’s commitment to developing antiracist policies that promote equity in mental health for all.
“This apology is one important step we needed to take to move forward to a more equitable future. The board is issuing this document on Martin Luther King Jr. Day, because we hope that it honors his life’s work of reconciliation and equality. We do not take that legacy or his call to action lightly and will continue our important work,” said Dr. Geller.
One involved the Eastern State Hospital in Williamsburg, Va., the nation’s first psychiatric care facility, founded in 1773.
Eastern State, which for a time in the 1800s was called the Eastern Lunatic Asylum, was not segregated when founded. However, 70 years later, when the 13 founders of what is now the APA met to discuss improvements in mental health care delivery, the treatment system they created and the organization they founded aligned with that era’s racist social and political policies. In this system, Black patients received psychiatric care separately from White patients, the APA said.
The APA also acknowledged failing to act in Black Americans’ best interest at critical points in the United States’ sociopolitical evolution throughout the 19th and 20th centuries.
“This inactivity was notably evident while white supremacists lynched Black people during the Reconstruction Era as well as when Jim Crow segregation was in effect, which led to ‘separate but equal’ standards of care starting in 1896,” the APA said.
Later, the APA failed to declare support for Brown v. Board of Education of Topeka in 1954, along with further major civil rights legislation designed to improve social and psychological conditions for Black people, the organization admitted.
Throughout the decades that followed, psychiatric misdiagnosis among Black, indigenous, and people of color populations were also common, the APA acknowledged.
For example, late 20th century psychiatrists commonly attributed their minority patients’ frustrations to schizophrenia, while categorizing similar behaviors as “neuroticism” in White patients.
The APA pointed to one study which found that APA members diagnosed more Black than White patients with schizophrenia, even when both had otherwise identical clinical presentations.
“This reveals the basis for embedded discrimination within psychiatry that has contributed to reduced quality of care” for Black, indigenous, and people of color, and “perpetuation of dangerous stereotypes,” the APA said.
Saul Levin, MD, the APA’s medical director and CEO, said the Board of Trustees has taken “an important step in issuing this apology. The APA administration is committed to working toward inclusion, health equity, and fairness that everyone deserves.”
The APA Board of Trustees began drafting the apology late last year after it concluded that events and persistent inequities in health care and psychiatry had highlighted an organizational need for action.
The APA’s Presidential Task Force on Structural Racism is continuing with efforts to educate and engage members on the issue and implement changes within the organization.
A version of this article first appeared on Medscape.com.
The American Psychiatric Association has issued a formal apology for its past support of structural racism in psychiatry.
The apology, issued Jan. 18, coincided with the federal holiday honoring the life and work of civil rights activist Dr. Martin Luther King Jr.
“We apologize for our role in perpetrating structural racism in this country, and we hope to begin to make amends for APA’s and psychiatry’s history of actions, intentional and not, that hurt Black, indigenous, and people of color,” APA President Jeffrey Geller, MD, MPH, said in a statement.
The apology was written and issued by the APA Board of Trustees. It acknowledges practices and events in psychiatry that contributed to racial inequality, and expresses the organization’s commitment to developing antiracist policies that promote equity in mental health for all.
“This apology is one important step we needed to take to move forward to a more equitable future. The board is issuing this document on Martin Luther King Jr. Day, because we hope that it honors his life’s work of reconciliation and equality. We do not take that legacy or his call to action lightly and will continue our important work,” said Dr. Geller.
One involved the Eastern State Hospital in Williamsburg, Va., the nation’s first psychiatric care facility, founded in 1773.
Eastern State, which for a time in the 1800s was called the Eastern Lunatic Asylum, was not segregated when founded. However, 70 years later, when the 13 founders of what is now the APA met to discuss improvements in mental health care delivery, the treatment system they created and the organization they founded aligned with that era’s racist social and political policies. In this system, Black patients received psychiatric care separately from White patients, the APA said.
The APA also acknowledged failing to act in Black Americans’ best interest at critical points in the United States’ sociopolitical evolution throughout the 19th and 20th centuries.
“This inactivity was notably evident while white supremacists lynched Black people during the Reconstruction Era as well as when Jim Crow segregation was in effect, which led to ‘separate but equal’ standards of care starting in 1896,” the APA said.
Later, the APA failed to declare support for Brown v. Board of Education of Topeka in 1954, along with further major civil rights legislation designed to improve social and psychological conditions for Black people, the organization admitted.
Throughout the decades that followed, psychiatric misdiagnosis among Black, indigenous, and people of color populations were also common, the APA acknowledged.
For example, late 20th century psychiatrists commonly attributed their minority patients’ frustrations to schizophrenia, while categorizing similar behaviors as “neuroticism” in White patients.
The APA pointed to one study which found that APA members diagnosed more Black than White patients with schizophrenia, even when both had otherwise identical clinical presentations.
“This reveals the basis for embedded discrimination within psychiatry that has contributed to reduced quality of care” for Black, indigenous, and people of color, and “perpetuation of dangerous stereotypes,” the APA said.
Saul Levin, MD, the APA’s medical director and CEO, said the Board of Trustees has taken “an important step in issuing this apology. The APA administration is committed to working toward inclusion, health equity, and fairness that everyone deserves.”
The APA Board of Trustees began drafting the apology late last year after it concluded that events and persistent inequities in health care and psychiatry had highlighted an organizational need for action.
The APA’s Presidential Task Force on Structural Racism is continuing with efforts to educate and engage members on the issue and implement changes within the organization.
A version of this article first appeared on Medscape.com.
Give women's mental health a seat at the health care table
Why it’s time for women’s mental health to be recognized as the subspecialty it already is
It wasn’t until I (Dr. Leistikow) finished my psychiatry residency that I realized the training I had received in women’s mental health was unusual. It was simply a required experience for PGY-3 residents at Johns Hopkins University, Baltimore.
All of us, regardless of interest, spent 1 afternoon a week over 6 months caring for patients in a specialty psychiatric clinic for women (run by Dr. Payne and Dr. Osborne). We discussed cases and received didactics on such topics as risk factors for postpartum depression; the risks of untreated mental illness in pregnancy, compared with the risks of various psychiatric medications; how to choose and dose medications for bipolar disorder as blood levels change across pregnancy; which resources to consult to determine the amounts and risks of various medications passed on in breast milk; and how to diagnose and treat premenstrual dysphoric disorder, to name a few lecture subjects.
By the time we were done, all residents had received more than 20 hours of teaching about how to treat mental illness in women across the reproductive life cycle. This was 20 hours more than is currently required by the American College of Graduate Medical Education, the accrediting body for all residencies, including psychiatry.1 It is time for that to change.
Women’s need for psychiatric treatment that addresses reproductive transitions is not new; it is as old as time. Not only do women who previously needed psychiatric treatment continue to need treatment when they get pregnant or are breastfeeding, but it is now well recognized that times of reproductive transition or flux – whether premenstrual, post partum, or perimenopausal – confer increased risk for both new-onset and exacerbations of prior mental illnesses.
What has changed is psychiatry’s ability to finally meet that need. Previously, despite the fact that women make up the majority of patients presenting for treatment, that nearly all women will menstruate and go through menopause, and that more than 80% of American women will have at least one pregnancy during their lifetime,psychiatrists practice as if these reproductive transitions were unfortunate blips getting in the doctor’s way.2 We mostly threw up our hands when our patients became pregnant, reflexively stopped all medications, and expected women to suffer for the sake of their babies.
with a large and growing research base, with both agreed-upon best practices and evolving standards of care informed by and responsive to the scientific literature. We now know that untreated maternal psychiatric illness carries its own risks for infants both before and after delivery; that many maternal pharmacologic treatments are lower risk for infants than previously thought; that protecting and treating women’s mental health in pregnancy has benefits for women, their babies, and the families that depend on them; and that there is now a growing evidence base informing both new and older treatments and enabling women and their doctors to make complex decisions balancing risk and benefit across the life cycle.
Many psychiatrists-in-training are hungry for this knowledge. At last count, in the United States alone, there were 16 women’s mental health fellowships available, up from just 3 in 2008.3 The problem is that none of them are accredited or funded by the ACGME, because reproductive psychiatry (here used interchangeably with the term women’s mental health) has not been officially recognized as a subspecialty. This means that current funding frequently rests on philanthropy, which often cannot be sustained, and clinical billing, which gives fellows in some programs such heavy clinical responsibilities that little time is left for scholarly work. Lack of subspecialty status also blocks numerous important downstream effects that would flow from this recognition.
Reproductive psychiatry clearly already meets criteria laid out by the American Board of Medical Specialties for defining a subspecialty field. As argued elsewhere, it has a distinct patient population with definable care needs and a standalone body of scientific medical knowledge as well as a national (and international) community of experts that has already done much to improve women’s access to care they desperately need.4 It also meets the ACGME’s criteria for a new subspecialty except for approval by the American Board of Psychiatry and Neurology.5 Finally, it also meets the requirements of the ABPN except for having 25 fellowship programs with 50 fellowship positions and 50 trainees per year completing fellowships, a challenging Catch-22 without the necessary funding that would accrue from accreditation.6
Despite growing awareness and demand, there remains a shortage of psychiatrists trained to treat women during times of reproductive transition and to pass their recommendations and knowledge on to their primary care and ob.gyn. colleagues. What official recognition would bring, in addition to funding for fellowships post residency, is a guaranteed seat at the table in psychiatry residencies, in terms of a required number of hours devoted to these topics for trainees, ensuring that all graduating psychiatrists have at least some exposure to the knowledge and practices so material to their patients.
It isn’t enough to wait for residencies to see the writing on the wall and voluntarily carve out a slice of pie devoted to women’s mental health from the limited time and resources available to train residents. A 2017 survey of psychiatry residency program training directors found that 23%, or almost a quarter of programs that responded, offered no reproductive psychiatry training at all, that 49% required 5 hours or less across all 4 years of training, and that 75% of programs had no required clinical exposure to reproductive psychiatry patients.7 Despite the fact that 87% of training directors surveyed agreed either that reproductive psychiatry was “an important area of education” or a subject general residents should be competent in, ACGME-recognized specialties take precedence.
A system so patchy and insufficient won’t do. It’s not good enough for the trainees who frequently have to look outside of their own institutions for the training they know they need. It’s not good enough for the pregnant or postpartum patient looking for evidence-based advice, who is currently left on her own to determine, prior to booking an appointment, whether a specific psychiatrist has received any training relevant to treating her. Adding reproductive psychiatry to the topics a graduating psychiatrist must have some proficiency in also signals to recent graduates and experienced attendings, as well as the relevant examining boards and producers of continuing medical education content, that women’s mental health is no longer a fringe topic but rather foundational to all practicing psychiatrists.
The oil needed to prime this pump is official recognition of the subspecialty that reproductive psychiatry already is. The women’s mental health community is ready. The research base is well established and growing exponentially. The number of women’s mental health fellowships is healthy and would increase significantly with ACGME funding. Psychiatry residency training programs can turn to recent graduates of these fellowships as well as their own faculty with reproductive psychiatry experience to teach trainees. In addition, the National Curriculum in Reproductive Psychiatry, over the last 4 years, has created a repository of free online modules dedicated to facilitating this type of training, with case discussions across numerous topics for use by both educators and trainees. The American Psychiatric Association recently formed the Committee on Women’s Mental Health in 2020 and will be publishing a textbook based on work done by the NCRP within the coming year.
Imagine the changed world that would open to all psychiatrists if reproductive psychiatry were given the credentials it deserves. When writing prescriptions, we would view pregnancy as the potential outcome it is in any woman of reproductive age, given that 50% of pregnancies are unplanned, and let women know ahead of time how to think about possible fetal effects rather than waiting for their panicked phone messages or hearing that they have stopped their medications abruptly. We would work to identify our patient’s individual risk factors for postpartum depression predelivery to reduce that risk and prevent or limit illness. We would plan ahead for close follow-up post partum during the window of greatest risk, rather than expecting women to drop out of care while taking care of their infants or languish on scheduling waiting lists. We would feel confident in giving evidence-based advice to our patients around times of reproductive transition across the life cycle, but especially in pregnancy and lactation, empowering women to make healthy decisions for themselves and their families, no longer abandoning them just when they need us most.
References
1. ACGME Program Requirements for Graduate Medical Education in Psychiatry. Accreditation Counsel for Graduate Medical Education. 2020 Jul 1.
2. Livingston G. “They’re waiting longer, but U.S. women today more likely to have children than a decade ago.” Pew Research Center’s Social & Demographic Trends Project. pewsocialtrends.org. 2018 Jan 18.
3. Nagle-Yang S et al. Acad Psychiatry. 2018 Apr;42(2):202-6.
4. Payne JL. Int Rev Psychiatry. 2019 May;31(3):207-9.
5. Accreditation Council for Graduate Medical Education Policies and Procedures. 2020 Sep 26.
6. American Board of Psychiatry and Neurology. Requirements for Subspecialty Recognition, Attachment A. 2008.
7. Osborne LM et al. Acad Psychiatry. 2018 Apr;42(2):197-201.
Dr. Leistikow is a reproductive psychiatrist and clinical assistant professor in the department of psychiatry at the University of Maryland, Baltimore, where she sees patients and helps train residents and fellows. She is on the education committee of the National Curriculum in Reproductive Psychiatry (NCRPtraining.org) and has written about women’s mental health for textbooks, scientific journals and on her private practice blog at www.womenspsychiatrybaltimore.com. Dr. Leistikow has no conflicts of interest.
Dr. Payne is associate professor of psychiatry and behavioral sciences and director of the Women’s Mood Disorders Center at Johns Hopkins University, Baltimore. In addition to providing outstanding clinical care for women with mood disorders, she conducts research into the genetic, biological, and environmental factors involved in postpartum depression. She and her colleagues have recently identified two epigenetic biomarkers of postpartum depression and are working hard to replicate this work with National Institutes of Health funding. Most recently, she was appointed to the American Psychiatric Association’s committee on women’s mental health and is serving as president-elect for both the Marcé of North America and the International Marcé Perinatal Mental Health Societies. She disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for Sage Therapeutics and Janssen Pharmaceuticals.
Dr. Osborne is associate professor of psychiatry and behavioral sciences and of gynecology and obstetrics at Johns Hopkins University, where she directs a postdoctoral fellowship program in reproductive psychiatry. She is an expert on the diagnosis and treatment of mood and anxiety disorders during pregnancy, the post partum, the premenstrual period, and perimenopause. Her work is supported by the Brain and Behavior Foundation, the Doris Duke Foundation, the American Board of Psychiatry and Neurology, and the National Institute of Mental Health. She has no conflicts of interest.
Why it’s time for women’s mental health to be recognized as the subspecialty it already is
Why it’s time for women’s mental health to be recognized as the subspecialty it already is
It wasn’t until I (Dr. Leistikow) finished my psychiatry residency that I realized the training I had received in women’s mental health was unusual. It was simply a required experience for PGY-3 residents at Johns Hopkins University, Baltimore.
All of us, regardless of interest, spent 1 afternoon a week over 6 months caring for patients in a specialty psychiatric clinic for women (run by Dr. Payne and Dr. Osborne). We discussed cases and received didactics on such topics as risk factors for postpartum depression; the risks of untreated mental illness in pregnancy, compared with the risks of various psychiatric medications; how to choose and dose medications for bipolar disorder as blood levels change across pregnancy; which resources to consult to determine the amounts and risks of various medications passed on in breast milk; and how to diagnose and treat premenstrual dysphoric disorder, to name a few lecture subjects.
By the time we were done, all residents had received more than 20 hours of teaching about how to treat mental illness in women across the reproductive life cycle. This was 20 hours more than is currently required by the American College of Graduate Medical Education, the accrediting body for all residencies, including psychiatry.1 It is time for that to change.
Women’s need for psychiatric treatment that addresses reproductive transitions is not new; it is as old as time. Not only do women who previously needed psychiatric treatment continue to need treatment when they get pregnant or are breastfeeding, but it is now well recognized that times of reproductive transition or flux – whether premenstrual, post partum, or perimenopausal – confer increased risk for both new-onset and exacerbations of prior mental illnesses.
What has changed is psychiatry’s ability to finally meet that need. Previously, despite the fact that women make up the majority of patients presenting for treatment, that nearly all women will menstruate and go through menopause, and that more than 80% of American women will have at least one pregnancy during their lifetime,psychiatrists practice as if these reproductive transitions were unfortunate blips getting in the doctor’s way.2 We mostly threw up our hands when our patients became pregnant, reflexively stopped all medications, and expected women to suffer for the sake of their babies.
with a large and growing research base, with both agreed-upon best practices and evolving standards of care informed by and responsive to the scientific literature. We now know that untreated maternal psychiatric illness carries its own risks for infants both before and after delivery; that many maternal pharmacologic treatments are lower risk for infants than previously thought; that protecting and treating women’s mental health in pregnancy has benefits for women, their babies, and the families that depend on them; and that there is now a growing evidence base informing both new and older treatments and enabling women and their doctors to make complex decisions balancing risk and benefit across the life cycle.
Many psychiatrists-in-training are hungry for this knowledge. At last count, in the United States alone, there were 16 women’s mental health fellowships available, up from just 3 in 2008.3 The problem is that none of them are accredited or funded by the ACGME, because reproductive psychiatry (here used interchangeably with the term women’s mental health) has not been officially recognized as a subspecialty. This means that current funding frequently rests on philanthropy, which often cannot be sustained, and clinical billing, which gives fellows in some programs such heavy clinical responsibilities that little time is left for scholarly work. Lack of subspecialty status also blocks numerous important downstream effects that would flow from this recognition.
Reproductive psychiatry clearly already meets criteria laid out by the American Board of Medical Specialties for defining a subspecialty field. As argued elsewhere, it has a distinct patient population with definable care needs and a standalone body of scientific medical knowledge as well as a national (and international) community of experts that has already done much to improve women’s access to care they desperately need.4 It also meets the ACGME’s criteria for a new subspecialty except for approval by the American Board of Psychiatry and Neurology.5 Finally, it also meets the requirements of the ABPN except for having 25 fellowship programs with 50 fellowship positions and 50 trainees per year completing fellowships, a challenging Catch-22 without the necessary funding that would accrue from accreditation.6
Despite growing awareness and demand, there remains a shortage of psychiatrists trained to treat women during times of reproductive transition and to pass their recommendations and knowledge on to their primary care and ob.gyn. colleagues. What official recognition would bring, in addition to funding for fellowships post residency, is a guaranteed seat at the table in psychiatry residencies, in terms of a required number of hours devoted to these topics for trainees, ensuring that all graduating psychiatrists have at least some exposure to the knowledge and practices so material to their patients.
It isn’t enough to wait for residencies to see the writing on the wall and voluntarily carve out a slice of pie devoted to women’s mental health from the limited time and resources available to train residents. A 2017 survey of psychiatry residency program training directors found that 23%, or almost a quarter of programs that responded, offered no reproductive psychiatry training at all, that 49% required 5 hours or less across all 4 years of training, and that 75% of programs had no required clinical exposure to reproductive psychiatry patients.7 Despite the fact that 87% of training directors surveyed agreed either that reproductive psychiatry was “an important area of education” or a subject general residents should be competent in, ACGME-recognized specialties take precedence.
A system so patchy and insufficient won’t do. It’s not good enough for the trainees who frequently have to look outside of their own institutions for the training they know they need. It’s not good enough for the pregnant or postpartum patient looking for evidence-based advice, who is currently left on her own to determine, prior to booking an appointment, whether a specific psychiatrist has received any training relevant to treating her. Adding reproductive psychiatry to the topics a graduating psychiatrist must have some proficiency in also signals to recent graduates and experienced attendings, as well as the relevant examining boards and producers of continuing medical education content, that women’s mental health is no longer a fringe topic but rather foundational to all practicing psychiatrists.
The oil needed to prime this pump is official recognition of the subspecialty that reproductive psychiatry already is. The women’s mental health community is ready. The research base is well established and growing exponentially. The number of women’s mental health fellowships is healthy and would increase significantly with ACGME funding. Psychiatry residency training programs can turn to recent graduates of these fellowships as well as their own faculty with reproductive psychiatry experience to teach trainees. In addition, the National Curriculum in Reproductive Psychiatry, over the last 4 years, has created a repository of free online modules dedicated to facilitating this type of training, with case discussions across numerous topics for use by both educators and trainees. The American Psychiatric Association recently formed the Committee on Women’s Mental Health in 2020 and will be publishing a textbook based on work done by the NCRP within the coming year.
Imagine the changed world that would open to all psychiatrists if reproductive psychiatry were given the credentials it deserves. When writing prescriptions, we would view pregnancy as the potential outcome it is in any woman of reproductive age, given that 50% of pregnancies are unplanned, and let women know ahead of time how to think about possible fetal effects rather than waiting for their panicked phone messages or hearing that they have stopped their medications abruptly. We would work to identify our patient’s individual risk factors for postpartum depression predelivery to reduce that risk and prevent or limit illness. We would plan ahead for close follow-up post partum during the window of greatest risk, rather than expecting women to drop out of care while taking care of their infants or languish on scheduling waiting lists. We would feel confident in giving evidence-based advice to our patients around times of reproductive transition across the life cycle, but especially in pregnancy and lactation, empowering women to make healthy decisions for themselves and their families, no longer abandoning them just when they need us most.
References
1. ACGME Program Requirements for Graduate Medical Education in Psychiatry. Accreditation Counsel for Graduate Medical Education. 2020 Jul 1.
2. Livingston G. “They’re waiting longer, but U.S. women today more likely to have children than a decade ago.” Pew Research Center’s Social & Demographic Trends Project. pewsocialtrends.org. 2018 Jan 18.
3. Nagle-Yang S et al. Acad Psychiatry. 2018 Apr;42(2):202-6.
4. Payne JL. Int Rev Psychiatry. 2019 May;31(3):207-9.
5. Accreditation Council for Graduate Medical Education Policies and Procedures. 2020 Sep 26.
6. American Board of Psychiatry and Neurology. Requirements for Subspecialty Recognition, Attachment A. 2008.
7. Osborne LM et al. Acad Psychiatry. 2018 Apr;42(2):197-201.
Dr. Leistikow is a reproductive psychiatrist and clinical assistant professor in the department of psychiatry at the University of Maryland, Baltimore, where she sees patients and helps train residents and fellows. She is on the education committee of the National Curriculum in Reproductive Psychiatry (NCRPtraining.org) and has written about women’s mental health for textbooks, scientific journals and on her private practice blog at www.womenspsychiatrybaltimore.com. Dr. Leistikow has no conflicts of interest.
Dr. Payne is associate professor of psychiatry and behavioral sciences and director of the Women’s Mood Disorders Center at Johns Hopkins University, Baltimore. In addition to providing outstanding clinical care for women with mood disorders, she conducts research into the genetic, biological, and environmental factors involved in postpartum depression. She and her colleagues have recently identified two epigenetic biomarkers of postpartum depression and are working hard to replicate this work with National Institutes of Health funding. Most recently, she was appointed to the American Psychiatric Association’s committee on women’s mental health and is serving as president-elect for both the Marcé of North America and the International Marcé Perinatal Mental Health Societies. She disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for Sage Therapeutics and Janssen Pharmaceuticals.
Dr. Osborne is associate professor of psychiatry and behavioral sciences and of gynecology and obstetrics at Johns Hopkins University, where she directs a postdoctoral fellowship program in reproductive psychiatry. She is an expert on the diagnosis and treatment of mood and anxiety disorders during pregnancy, the post partum, the premenstrual period, and perimenopause. Her work is supported by the Brain and Behavior Foundation, the Doris Duke Foundation, the American Board of Psychiatry and Neurology, and the National Institute of Mental Health. She has no conflicts of interest.
It wasn’t until I (Dr. Leistikow) finished my psychiatry residency that I realized the training I had received in women’s mental health was unusual. It was simply a required experience for PGY-3 residents at Johns Hopkins University, Baltimore.
All of us, regardless of interest, spent 1 afternoon a week over 6 months caring for patients in a specialty psychiatric clinic for women (run by Dr. Payne and Dr. Osborne). We discussed cases and received didactics on such topics as risk factors for postpartum depression; the risks of untreated mental illness in pregnancy, compared with the risks of various psychiatric medications; how to choose and dose medications for bipolar disorder as blood levels change across pregnancy; which resources to consult to determine the amounts and risks of various medications passed on in breast milk; and how to diagnose and treat premenstrual dysphoric disorder, to name a few lecture subjects.
By the time we were done, all residents had received more than 20 hours of teaching about how to treat mental illness in women across the reproductive life cycle. This was 20 hours more than is currently required by the American College of Graduate Medical Education, the accrediting body for all residencies, including psychiatry.1 It is time for that to change.
Women’s need for psychiatric treatment that addresses reproductive transitions is not new; it is as old as time. Not only do women who previously needed psychiatric treatment continue to need treatment when they get pregnant or are breastfeeding, but it is now well recognized that times of reproductive transition or flux – whether premenstrual, post partum, or perimenopausal – confer increased risk for both new-onset and exacerbations of prior mental illnesses.
What has changed is psychiatry’s ability to finally meet that need. Previously, despite the fact that women make up the majority of patients presenting for treatment, that nearly all women will menstruate and go through menopause, and that more than 80% of American women will have at least one pregnancy during their lifetime,psychiatrists practice as if these reproductive transitions were unfortunate blips getting in the doctor’s way.2 We mostly threw up our hands when our patients became pregnant, reflexively stopped all medications, and expected women to suffer for the sake of their babies.
with a large and growing research base, with both agreed-upon best practices and evolving standards of care informed by and responsive to the scientific literature. We now know that untreated maternal psychiatric illness carries its own risks for infants both before and after delivery; that many maternal pharmacologic treatments are lower risk for infants than previously thought; that protecting and treating women’s mental health in pregnancy has benefits for women, their babies, and the families that depend on them; and that there is now a growing evidence base informing both new and older treatments and enabling women and their doctors to make complex decisions balancing risk and benefit across the life cycle.
Many psychiatrists-in-training are hungry for this knowledge. At last count, in the United States alone, there were 16 women’s mental health fellowships available, up from just 3 in 2008.3 The problem is that none of them are accredited or funded by the ACGME, because reproductive psychiatry (here used interchangeably with the term women’s mental health) has not been officially recognized as a subspecialty. This means that current funding frequently rests on philanthropy, which often cannot be sustained, and clinical billing, which gives fellows in some programs such heavy clinical responsibilities that little time is left for scholarly work. Lack of subspecialty status also blocks numerous important downstream effects that would flow from this recognition.
Reproductive psychiatry clearly already meets criteria laid out by the American Board of Medical Specialties for defining a subspecialty field. As argued elsewhere, it has a distinct patient population with definable care needs and a standalone body of scientific medical knowledge as well as a national (and international) community of experts that has already done much to improve women’s access to care they desperately need.4 It also meets the ACGME’s criteria for a new subspecialty except for approval by the American Board of Psychiatry and Neurology.5 Finally, it also meets the requirements of the ABPN except for having 25 fellowship programs with 50 fellowship positions and 50 trainees per year completing fellowships, a challenging Catch-22 without the necessary funding that would accrue from accreditation.6
Despite growing awareness and demand, there remains a shortage of psychiatrists trained to treat women during times of reproductive transition and to pass their recommendations and knowledge on to their primary care and ob.gyn. colleagues. What official recognition would bring, in addition to funding for fellowships post residency, is a guaranteed seat at the table in psychiatry residencies, in terms of a required number of hours devoted to these topics for trainees, ensuring that all graduating psychiatrists have at least some exposure to the knowledge and practices so material to their patients.
It isn’t enough to wait for residencies to see the writing on the wall and voluntarily carve out a slice of pie devoted to women’s mental health from the limited time and resources available to train residents. A 2017 survey of psychiatry residency program training directors found that 23%, or almost a quarter of programs that responded, offered no reproductive psychiatry training at all, that 49% required 5 hours or less across all 4 years of training, and that 75% of programs had no required clinical exposure to reproductive psychiatry patients.7 Despite the fact that 87% of training directors surveyed agreed either that reproductive psychiatry was “an important area of education” or a subject general residents should be competent in, ACGME-recognized specialties take precedence.
A system so patchy and insufficient won’t do. It’s not good enough for the trainees who frequently have to look outside of their own institutions for the training they know they need. It’s not good enough for the pregnant or postpartum patient looking for evidence-based advice, who is currently left on her own to determine, prior to booking an appointment, whether a specific psychiatrist has received any training relevant to treating her. Adding reproductive psychiatry to the topics a graduating psychiatrist must have some proficiency in also signals to recent graduates and experienced attendings, as well as the relevant examining boards and producers of continuing medical education content, that women’s mental health is no longer a fringe topic but rather foundational to all practicing psychiatrists.
The oil needed to prime this pump is official recognition of the subspecialty that reproductive psychiatry already is. The women’s mental health community is ready. The research base is well established and growing exponentially. The number of women’s mental health fellowships is healthy and would increase significantly with ACGME funding. Psychiatry residency training programs can turn to recent graduates of these fellowships as well as their own faculty with reproductive psychiatry experience to teach trainees. In addition, the National Curriculum in Reproductive Psychiatry, over the last 4 years, has created a repository of free online modules dedicated to facilitating this type of training, with case discussions across numerous topics for use by both educators and trainees. The American Psychiatric Association recently formed the Committee on Women’s Mental Health in 2020 and will be publishing a textbook based on work done by the NCRP within the coming year.
Imagine the changed world that would open to all psychiatrists if reproductive psychiatry were given the credentials it deserves. When writing prescriptions, we would view pregnancy as the potential outcome it is in any woman of reproductive age, given that 50% of pregnancies are unplanned, and let women know ahead of time how to think about possible fetal effects rather than waiting for their panicked phone messages or hearing that they have stopped their medications abruptly. We would work to identify our patient’s individual risk factors for postpartum depression predelivery to reduce that risk and prevent or limit illness. We would plan ahead for close follow-up post partum during the window of greatest risk, rather than expecting women to drop out of care while taking care of their infants or languish on scheduling waiting lists. We would feel confident in giving evidence-based advice to our patients around times of reproductive transition across the life cycle, but especially in pregnancy and lactation, empowering women to make healthy decisions for themselves and their families, no longer abandoning them just when they need us most.
References
1. ACGME Program Requirements for Graduate Medical Education in Psychiatry. Accreditation Counsel for Graduate Medical Education. 2020 Jul 1.
2. Livingston G. “They’re waiting longer, but U.S. women today more likely to have children than a decade ago.” Pew Research Center’s Social & Demographic Trends Project. pewsocialtrends.org. 2018 Jan 18.
3. Nagle-Yang S et al. Acad Psychiatry. 2018 Apr;42(2):202-6.
4. Payne JL. Int Rev Psychiatry. 2019 May;31(3):207-9.
5. Accreditation Council for Graduate Medical Education Policies and Procedures. 2020 Sep 26.
6. American Board of Psychiatry and Neurology. Requirements for Subspecialty Recognition, Attachment A. 2008.
7. Osborne LM et al. Acad Psychiatry. 2018 Apr;42(2):197-201.
Dr. Leistikow is a reproductive psychiatrist and clinical assistant professor in the department of psychiatry at the University of Maryland, Baltimore, where she sees patients and helps train residents and fellows. She is on the education committee of the National Curriculum in Reproductive Psychiatry (NCRPtraining.org) and has written about women’s mental health for textbooks, scientific journals and on her private practice blog at www.womenspsychiatrybaltimore.com. Dr. Leistikow has no conflicts of interest.
Dr. Payne is associate professor of psychiatry and behavioral sciences and director of the Women’s Mood Disorders Center at Johns Hopkins University, Baltimore. In addition to providing outstanding clinical care for women with mood disorders, she conducts research into the genetic, biological, and environmental factors involved in postpartum depression. She and her colleagues have recently identified two epigenetic biomarkers of postpartum depression and are working hard to replicate this work with National Institutes of Health funding. Most recently, she was appointed to the American Psychiatric Association’s committee on women’s mental health and is serving as president-elect for both the Marcé of North America and the International Marcé Perinatal Mental Health Societies. She disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, adviser, consultant, or trustee for Sage Therapeutics and Janssen Pharmaceuticals.
Dr. Osborne is associate professor of psychiatry and behavioral sciences and of gynecology and obstetrics at Johns Hopkins University, where she directs a postdoctoral fellowship program in reproductive psychiatry. She is an expert on the diagnosis and treatment of mood and anxiety disorders during pregnancy, the post partum, the premenstrual period, and perimenopause. Her work is supported by the Brain and Behavior Foundation, the Doris Duke Foundation, the American Board of Psychiatry and Neurology, and the National Institute of Mental Health. She has no conflicts of interest.






