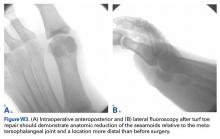
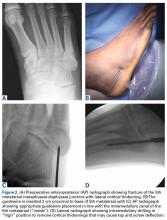
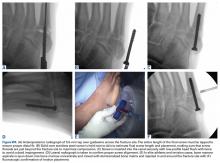
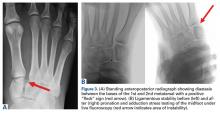
User login
Opioid Misuse Linked to Heroin Use in Study of Veterans
Researchers have found that veterans misusing opioids were 5.4 times more likely to use heroin than were veterans who did not misuse opioids. The study of 3,396 veterans found that 77% of heroin users reported previous misuse of opioids. The findings were reported in the Journal of Addiction and were based on an analysis of participants in the Veterans Aging Cohort Study (VACS).
Related: Veterans’ Health and Opioid Safety–Contexts, Risks, and Outreach Implications
“Our findings demonstrate a pattern of transitioning from nonmedical use of prescription opioids to heroin use that has only been demonstrated in select populations,” David Fiellin, Yale public health and medical professor and director of the VACS intervention group told a Brown University reporter. “Our findings are unique in that our sample of individuals consisted of patients who were receiving routine medical care for common medical conditions.”
Related:Call for App to Help Opioid Rehab
All study participants reported no lifetime use of heroin or nonmedical use of opioids at baseline. The authors analyzed VACS data of HIV-infected and an age/race/site-matched control group of HIV-uninfected veterans. Annual behavioral assessments were conducted and contained self-reported measures of nonmedical use of prescription opioids and heroin use.
In addition to analyzing opioid use, the study authors also examined the role of gender, race, and use of stimulant drugs in heroin use. Risk of heroin use was greater for men (2.6 times), stimulant drug users (2.1 times), and blacks (2 times).
Related: Joining Forces to Reduce Opioid-Related Death
“This paper shows that, as a general clinical practice, particularly for this population which does experience a lot of chronic pain and other risks for substance use including PTSD, screening for nonmedical painkiller use, whether you are prescribing an opioid or not, may be effective to prevent even more harmful transitions to heroin or other drugs,” Brandon Marshall, an assistant professor at the Brown University School of Public Health told the Brown University reporter.
Researchers have found that veterans misusing opioids were 5.4 times more likely to use heroin than were veterans who did not misuse opioids. The study of 3,396 veterans found that 77% of heroin users reported previous misuse of opioids. The findings were reported in the Journal of Addiction and were based on an analysis of participants in the Veterans Aging Cohort Study (VACS).
Related: Veterans’ Health and Opioid Safety–Contexts, Risks, and Outreach Implications
“Our findings demonstrate a pattern of transitioning from nonmedical use of prescription opioids to heroin use that has only been demonstrated in select populations,” David Fiellin, Yale public health and medical professor and director of the VACS intervention group told a Brown University reporter. “Our findings are unique in that our sample of individuals consisted of patients who were receiving routine medical care for common medical conditions.”
Related:Call for App to Help Opioid Rehab
All study participants reported no lifetime use of heroin or nonmedical use of opioids at baseline. The authors analyzed VACS data of HIV-infected and an age/race/site-matched control group of HIV-uninfected veterans. Annual behavioral assessments were conducted and contained self-reported measures of nonmedical use of prescription opioids and heroin use.
In addition to analyzing opioid use, the study authors also examined the role of gender, race, and use of stimulant drugs in heroin use. Risk of heroin use was greater for men (2.6 times), stimulant drug users (2.1 times), and blacks (2 times).
Related: Joining Forces to Reduce Opioid-Related Death
“This paper shows that, as a general clinical practice, particularly for this population which does experience a lot of chronic pain and other risks for substance use including PTSD, screening for nonmedical painkiller use, whether you are prescribing an opioid or not, may be effective to prevent even more harmful transitions to heroin or other drugs,” Brandon Marshall, an assistant professor at the Brown University School of Public Health told the Brown University reporter.
Researchers have found that veterans misusing opioids were 5.4 times more likely to use heroin than were veterans who did not misuse opioids. The study of 3,396 veterans found that 77% of heroin users reported previous misuse of opioids. The findings were reported in the Journal of Addiction and were based on an analysis of participants in the Veterans Aging Cohort Study (VACS).
Related: Veterans’ Health and Opioid Safety–Contexts, Risks, and Outreach Implications
“Our findings demonstrate a pattern of transitioning from nonmedical use of prescription opioids to heroin use that has only been demonstrated in select populations,” David Fiellin, Yale public health and medical professor and director of the VACS intervention group told a Brown University reporter. “Our findings are unique in that our sample of individuals consisted of patients who were receiving routine medical care for common medical conditions.”
Related:Call for App to Help Opioid Rehab
All study participants reported no lifetime use of heroin or nonmedical use of opioids at baseline. The authors analyzed VACS data of HIV-infected and an age/race/site-matched control group of HIV-uninfected veterans. Annual behavioral assessments were conducted and contained self-reported measures of nonmedical use of prescription opioids and heroin use.
In addition to analyzing opioid use, the study authors also examined the role of gender, race, and use of stimulant drugs in heroin use. Risk of heroin use was greater for men (2.6 times), stimulant drug users (2.1 times), and blacks (2 times).
Related: Joining Forces to Reduce Opioid-Related Death
“This paper shows that, as a general clinical practice, particularly for this population which does experience a lot of chronic pain and other risks for substance use including PTSD, screening for nonmedical painkiller use, whether you are prescribing an opioid or not, may be effective to prevent even more harmful transitions to heroin or other drugs,” Brandon Marshall, an assistant professor at the Brown University School of Public Health told the Brown University reporter.
Medical Issues in American Football: Eyes, Teeth, and Skin
Orthopedic conditions are only one of the many medical issues football team physicians may face. In this review, we cover the management of a few of the most common nonorthopedic medical issues football team physicians are likely to encounter, including eye injuries, dental concerns, and skin conditions.
Eye Injuries
More than 2.5 million eye injuries occur each year, with 50,000 people permanently losing part or all of their vision.1 Eye injuries account for over 600,000 yearly emergency department visits; over 30% of these eye injuries were attributed to a sports injury.1 Football is classified as high risk for eye injury, along with baseball, hockey, basketball, and lacrosse.2 Common eye injury mechanisms are categorized as blunt, penetrating, and radiating. Blunt injuries are most common.2 When evaluating an athlete on the sideline, relevant history would include the size of the object, the level of force, and the direction from which the impact occurred. An examination should include best-corrected visual acuity using an eye chart, confrontational visual fields, assessment of extraocular movements, assessment of red reflex, and pupil evaluation with a light source.2
Cornea Injuries
The outermost layer of the eye, the cornea, can be subject to blunt and penetrating injuries. Corneal abrasions often occur from mechanical trauma, such as one from the fingernail of an opposing player, that disrupts the integrity of the corneal epithelium. A corneal abrasion can be identified by applying fluorescein strips after application of a topical anesthetic. Abrasions appear fluorescent green when viewed with a cobalt blue light. If an abrasion is identified, management includes preventing infection and treating pain. Prophylactic topical antibiotics can be applied, particularly for contact lens wearers. Patching has not shown benefit in treatment of pain.3 The physician can consider using topical nonsteroidal anti-inflammatory drugs, such as diclofenac or ketorolac, with a soft contact lens to treat the pain.4 The patient should follow up frequently for monitoring for infection and healing.
Orbital Fractures
Orbital fractures should be considered when an object larger than the orbital opening, such as an elbow or knee, causes blunt trauma to the surrounding bony structures, or a digital poke occurs to the globe.5 The floor of the orbit and medial wall are thin bones that often break sacrificially to protect the globe from rupture. Examination findings may include diplopia, sunken globe, numbness in the distribution of infraorbital nerve, or periorbital emphysema.6 Urgent evaluation should be considered to rule out associated intraocular damage. Imaging and a physical examination can help guide surgical management, if indicated. The most common outcome after this injury is diplopia with upper field gaze.5
Retina Issues
Trauma to the face or head may result in a separation of the retina from the underlying retinal pigment epithelium and allow vitreous fluid to seep in and further separate the layers, causing a retinal detachment. Symptoms may include flashes of light (photopsia), floaters, and visual field defects. Emergent referral is indicated, as the outcomes from this condition are time-sensitive. Consider placing an eye shield to prevent any further pressure on the globe.
Globe Injuries and Rupture
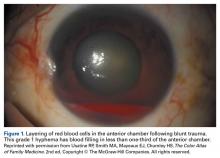
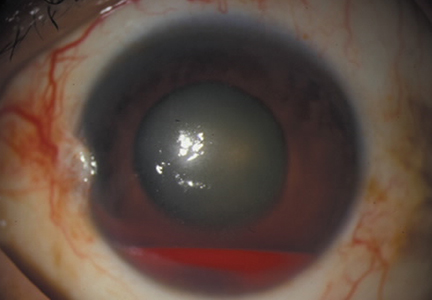
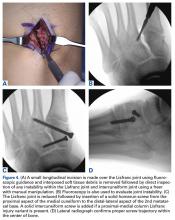
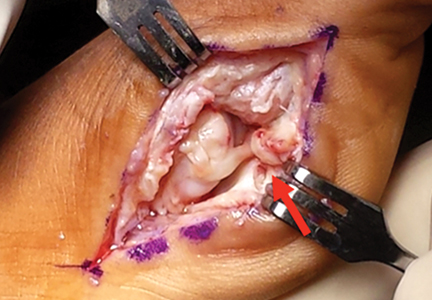
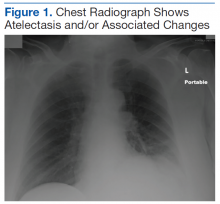
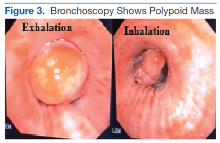
Another emergent ophthalmologic condition that can occur in football is globe rupture. Clinical findings usually prompt the clinician to consider this diagnosis. Hyphema (the collection of blood in the anterior chamber) may be seen in globe injuries. The most common clinical finding of athletes requiring hospitalization after an ocular injury is macroscopic hyphema (Figure 1).7-9
Prompt referral is warranted when there is a sudden decrease or change in vision, pain during movements, photophobia, and floaters and/or flashes.2 Consideration of return to play should take into account the patient’s vision and comfort level, which should not be masked by topical analgesics. Protective eyewear has been mandated in several sports, and has decreased the rate of eye injuries.10 Polycarbonate lenses of 3-mm thickness are recommended due to the significant comparable strength and impact-resistance.2 During the preparticipation physical for high-risk sports, the utilization of protective eyewear should be discussed.
Dental Concerns
Dental injuries may present a challenge for the sports medicine clinician. Contact injuries from elbows, fists, and other nonprojectile objects typically result in low-speed, lower-energy injuries, such as soft tissue lacerations and contusions. On the other hand, high-speed injuries occurring from balls, pucks, and sticks may result in more significant trauma. Baseball accounts for the highest percentage of sports-related dental injuries (40.2%), while basketball was second (20.2%) and football third (12.5%). Over 75% of these injuries occurred in males.11
On-field management of dental injuries should always start with the primary trauma survey, including assessment of the athlete’s airway, breathing, and circulatory function, as well as a targeted cervical spine evaluation. When obtaining a history, one should recognize the mechanism of injury and assess for signs of concomitant injuries, ie, respiratory compromise, concussion, leakage of cerebrospinal fluid, and teeth alignment. Findings from this initial evaluation may reveal critical conditions that will require management in addition to the dental injury.
Of central concern in managing dental trauma is preserving the viability of the injured structures. Therefore, much attention is paid to the pulpal and root vitality of injured teeth. The International Association of Dental Traumology Dental Trauma Guidelines recommend a biological approach to the urgent care of dental injuries:12
1. Stabilize the injury by carefully repositioning displaced entities and suturing soft tissue lacerations.
2. Eliminate or reduce the complications from bacterial contamination by rinsing and flushing with available liquids and use of chlorhexidine when possible.
3. Promote the opportunity for healing by replanting avulsed teeth and repositioning displaced teeth.
4. Make every effort to allow continued development of alveolar ridges in children.
Mouth guards are the single most effective prevention strategy for most contact sport dental injuries. One meta-analysis demonstrated a pooled 86% increased risk of orofacial injuries in nonusers.13
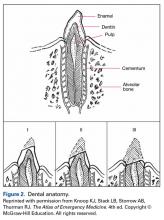
To review the anatomy (and injuries) of the tooth, one must consider the Ellis classification of enamel, dentin, and pulp injuries (Figure 2).
Tooth Subluxation
Tooth subluxations usually occur secondary to trauma and cause loosening of the tooth in its alveolar socket. A root fracture should be suspected in the setting of a subluxation. On exam, the tooth may be excessively mobile with gentle pressure. If unstable, immobilization with gauze packing or aluminum foil with dental follow-up is recommended.
Fractures
Ellis class I fractures are small chips in the enamel. There should be uniform color at the fracture site. A dental referral may be warranted to smooth rough enamel edges, but if no other injuries are present, these athletes may continue playing with some protection of the fractured surface. A mouth guard may be helpful to avoid mucosal lacerations.
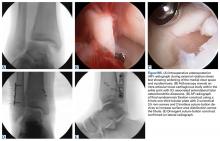
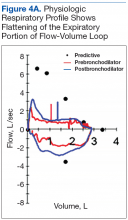
Ellis class II fractures often present with sensitivity to inhaled air and to hot and cold temperatures. Yellow dentin is visible at the fracture site (Figure 3).
Ellis class III fractures may also present with air and temperature sensitivity. Finger pressure may expose a large fracture. Pink or red pulp is visible at the fracture site. Wiping the fracture site with sterile gauze may reveal bleeding from the pulp. This is considered a dental emergency. Immediate restriction from contact sports participation and urgent dental evaluation is indicated for root canal and capping and to prevent abscess formation.
Tooth Avulsion
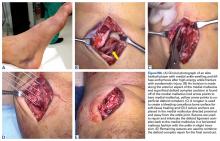
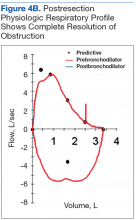
Tooth avulsions occur when a tooth is completely displaced from the socket (Figure 4).
Skin Issues
Dermatological issues are some of the most common medical conditions faced by a football team physician. Skin infections in particular can pose a significant challenge both diagnostically as well as from a clearance-to-play perspective, given the potential for infections to affect other participants, such as other members of the team. Skin infection rates vary by sport and age group, with one study reporting 28.56 infections per 100,000 athletic exposures in high school wrestlers, which was more than 10 times that of football.14 Still, football players are at a higher risk of skin infections given the contact nature of the sport and close person-to-person proximity. A precise diagnosis may be difficult early in the course of a skin eruption, and with differing guidelines from various professional societies, it may be best suited for medical personnel familiar with these conditions, such as a sports medicine physician or dermatologist, to manage these athletes. A thorough and systematic evaluation is recommended, as athletes are often treated with unnecessary antibiotics, which contributes to antibiotic resistance. Previous antibiotic use may also be a risk factor for developing community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA).15
Two terms sports medicine clinicians must be familiar with are “adequately protected” and “properly covered.” The National Collegiate Athletic Association (NCAA) defines a wound or skin condition as adequately protected when the condition is considered noninfectious, adequately treated by a healthcare provider, and is able to be properly covered. A skin infection is considered properly covered when the lesion is covered by a securely attached bandage or dressing that will contain all drainage and remain intact throughout the sport activity.16
Impetigo
Impetigo is often caused by Staphylococcus and Streptococcus subspecies. The classic presentation is a dry, honey-crusted lesion with an erythematous base. Culture or gram stain may be helpful, but treatment may be initiated on a clinical basis without these studies. Topical antibiotics may be used, but in the setting of multiple lesions or an outbreak, systemic (eg, oral) antibiotics are preferred. Oral antibiotics may also shorten the time to return to play. If not responsive to the initial treatment, MRSA should be considered. No new lesions for 48 hours and a minimum of 72 hours of therapy with no moist, exudative, or draining lesions are required prior to return to play. These lesions cannot be covered as the sole means of return to play.
Methicillin-Resistant Staphylococcus aureus
MRSA is one of the most challenging skin infections for the sports medicine clinician to manage. Several outbreaks have been reported in the high school, college, and professional settings.17-20 Standardized precautions and a proactive approach are key in preventing MRSA outbreaks. It appears that different activities within a given sport may contribute to MRSA risk. One study reported football linemen had the highest attack rate, while another study reported cornerbacks and wide receivers to have the highest rate of MRSA infections.17,20 The elbow area was the most common site infected in both studies.
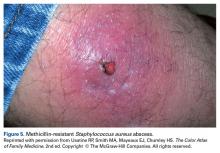
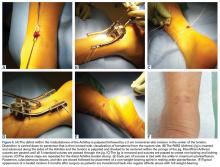
Abscesses are best initially managed by incision and drainage as well as obtaining wound cultures (Figure 5).
Preventative measures are thought to be useful, especially in the management of teams. The Centers for Disease Control and Prevention has published guidelines for both clinicians and patients. Precautions including hand washing; encouraging good overall hygiene; avoiding whirlpools; discouraging the sharing of towels, razors, and athletic gear; maintaining clean equipment/facilities; and encouraging early reporting of skin lesions.14,17,21,22 Isolated cases of MRSA do not need to be reported, but if more than one athlete is infected, one should notify the athletic training and team coaching staff. In the setting of an outbreak, the physician may need to notify local or state health agencies. No new lesions for 48 hours and a minimum of 72 hours of therapy with no moist, exudative, or draining lesions are required prior to returning to play. These lesions cannot be covered as the sole means of return to play.
Tinea Pedis
Tinea pedis is a common dermatophyte infection involving the feet and is most commonly caused by Trichophyton rubrum. Its distribution is usually interdigital or along the plantar surface of the foot. Topical antifungals with either allylamines or azoles are usually sufficient. Terbinafine has been shown to have a shorter duration of treatment. Athletes with tinea pedis are not restricted from sports participation during treatment, as long as the lesions are properly covered.
Tinea Corporis
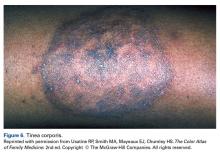
Tinea corporis is a common superficial fungal infection of the body. It classically presents as pruritic, annular lesions, with well-demarcated borders and central clearing (
Tinea Cruris
Commonly known as “jock-itch,” this fungal infection is often very pruritic and involves the groin or genital region. The area is also inflamed and scaly. Treatment usually consists of topical allylamines or azoles. Allylamines amines are often preferred, as they require a shorter duration of treatment. There are no specific guidelines on the return to play with these athletes. Clearance is at the team physician’s discretion, but usually there are no restrictions. Athletes with extensive lesions may need to be disqualified from contact sports activities.
Am J Orthop. 2016;45(6):377-382. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Owens PL, Mutter R. Emergency Department Visits Related to Eye Injuries, 2008. Agency for Healthcare Research and Quality Web site. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb112.pdf. Published May 2011. Accessed August 18, 2016.
2. Rodriguez JO, Lavina AM, Agarwai A. Prevention and treatment of common eye injuries in sports. Am Fam Physician. 2003;67(7):1481-1496.
3. Lim CH, Turner A, Lim BX. Patching for corneal abrasion. Cochrane Database Syst Rev. 2016;7:CD004764.
4. Weaver CS, Terrell KM. Evidence-based emergency medicine. Update: do ophthalmic nonsteroidal anti-inflammatory drugs reduce the pain associated with simple corneal abrasion without delaying healing? Ann Emerg Med. 2003;41(1):134-140.
5. Williams RJ 3rd, Marx RG, Barnes R, O’Brien SJ, Warren RF. Fractures about the orbit in professional American football players. Am J Sports Med. 2001;29(1):55-57.
6. Forrest LA, Schuller DE, Strauss RH. Management of orbital blow-out fractures. Case reports and discussion. Am J Sports Med. 1989;17(2):217-220.
7. Barr A, Baines PS, Desai P, MacEwen CJ. Ocular sports injuries: the current picture. Br J Sports Med. 2000;34(6):456-458.
8. Pokhrel PK, Loftus SA. Ocular emergencies. Am Fam Physician. 2007;76(6):829-836.
9. Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H. Eye Trauma—Hyphema. The Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013.
10. Lincoln AE, Caswell SV, Almquist JL, et al. Effectiveness of the women’s lacrosse protective eyewear mandate in the reduction of eye injuries. Am J Sports Med. 2012;40(3):611-614.
11. Stewart GB, Shields BJ, Fields S, Comstock RD, Smith GA. Consumer products and activities associated with dental injuries to children treated in United States emergency departments, 1990-2003. Dental Traumatol. 2009;25(4):399-405.
12. Bakland LK. Dental trauma guidelines. Pediatric Dent. 2013;35(2):106-108.
13. Knapik J, Marshall SW, Lee RB, et al. Mouthguards in sport activities: history, physical properties and Injury prevention effectiveness. Sports Med. 2007;37(2):117-144.
14. Ashack KA, Burton KA, Johnson TR, Currie DW, Comstock RD, Dellavalle RP. Skin infections among US high school athletes: a national survey. J Am Acad Dermatol. 2016;74(4):679-684.e1.
15. Ellis MW, Hospenthal DR, Dooley DP, Gray PJ, Murray CK. Natural history of community-acquired methicillin-resistant Staphylococcus aureus colonization and infection in soldiers. Clin Infect Dis. 2004;39(7):971-979.
16. The National Collegiate Athletic Association. 2014-15 NCAA Sports Medicine Handbook. http://www.ncaapublications.com/productdownloads/MD15.pdf. Revised June 2008. Accessed August 18, 2016.
17. Anderson BJ. The effectiveness of valacyclovir in preventing reactivation of herpes gladiatorum in wrestlers. Clin J Sport Med. 1999;9(2):86-90.
18. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55.
19. Jeffords MD, Batts K. Dermatology. In: O’Connor FG, Casa DJ, Davis BA, Pierre PS, Sallis RE, Wilder RP, eds. ACSM’s Sports Medicine: A Comprehensive Review. Riverwoods, IL: Wolters Kluwer; 2016:181-188.
20. Kazakova SV, Hageman JC, Matava M, et al. A clone of methicillin-resistant Staphylococcus aureus among professional football players. N Engl J Med. 2005;352(5):468-475.
21. Begier EM, Frenette K, Barrett NL, et al. A high-morbidity outbreak of methicillin-resistant Staphylococcus aureus among players on a college football team, facilitated by cosmetic body shaving and turf burns. Clin Infect Dis. 2004;39(10):1446-1453.
22. Geissler KE, Borchers JR. More than meets the eye: a rapidly progressive skin infection in a football player. Clin J Sport Med. 2015;25(3):e54-e56.
Orthopedic conditions are only one of the many medical issues football team physicians may face. In this review, we cover the management of a few of the most common nonorthopedic medical issues football team physicians are likely to encounter, including eye injuries, dental concerns, and skin conditions.
Eye Injuries
More than 2.5 million eye injuries occur each year, with 50,000 people permanently losing part or all of their vision.1 Eye injuries account for over 600,000 yearly emergency department visits; over 30% of these eye injuries were attributed to a sports injury.1 Football is classified as high risk for eye injury, along with baseball, hockey, basketball, and lacrosse.2 Common eye injury mechanisms are categorized as blunt, penetrating, and radiating. Blunt injuries are most common.2 When evaluating an athlete on the sideline, relevant history would include the size of the object, the level of force, and the direction from which the impact occurred. An examination should include best-corrected visual acuity using an eye chart, confrontational visual fields, assessment of extraocular movements, assessment of red reflex, and pupil evaluation with a light source.2
Cornea Injuries
The outermost layer of the eye, the cornea, can be subject to blunt and penetrating injuries. Corneal abrasions often occur from mechanical trauma, such as one from the fingernail of an opposing player, that disrupts the integrity of the corneal epithelium. A corneal abrasion can be identified by applying fluorescein strips after application of a topical anesthetic. Abrasions appear fluorescent green when viewed with a cobalt blue light. If an abrasion is identified, management includes preventing infection and treating pain. Prophylactic topical antibiotics can be applied, particularly for contact lens wearers. Patching has not shown benefit in treatment of pain.3 The physician can consider using topical nonsteroidal anti-inflammatory drugs, such as diclofenac or ketorolac, with a soft contact lens to treat the pain.4 The patient should follow up frequently for monitoring for infection and healing.
Orbital Fractures
Orbital fractures should be considered when an object larger than the orbital opening, such as an elbow or knee, causes blunt trauma to the surrounding bony structures, or a digital poke occurs to the globe.5 The floor of the orbit and medial wall are thin bones that often break sacrificially to protect the globe from rupture. Examination findings may include diplopia, sunken globe, numbness in the distribution of infraorbital nerve, or periorbital emphysema.6 Urgent evaluation should be considered to rule out associated intraocular damage. Imaging and a physical examination can help guide surgical management, if indicated. The most common outcome after this injury is diplopia with upper field gaze.5
Retina Issues
Trauma to the face or head may result in a separation of the retina from the underlying retinal pigment epithelium and allow vitreous fluid to seep in and further separate the layers, causing a retinal detachment. Symptoms may include flashes of light (photopsia), floaters, and visual field defects. Emergent referral is indicated, as the outcomes from this condition are time-sensitive. Consider placing an eye shield to prevent any further pressure on the globe.
Globe Injuries and Rupture
Another emergent ophthalmologic condition that can occur in football is globe rupture. Clinical findings usually prompt the clinician to consider this diagnosis. Hyphema (the collection of blood in the anterior chamber) may be seen in globe injuries. The most common clinical finding of athletes requiring hospitalization after an ocular injury is macroscopic hyphema (Figure 1).7-9
Prompt referral is warranted when there is a sudden decrease or change in vision, pain during movements, photophobia, and floaters and/or flashes.2 Consideration of return to play should take into account the patient’s vision and comfort level, which should not be masked by topical analgesics. Protective eyewear has been mandated in several sports, and has decreased the rate of eye injuries.10 Polycarbonate lenses of 3-mm thickness are recommended due to the significant comparable strength and impact-resistance.2 During the preparticipation physical for high-risk sports, the utilization of protective eyewear should be discussed.
Dental Concerns
Dental injuries may present a challenge for the sports medicine clinician. Contact injuries from elbows, fists, and other nonprojectile objects typically result in low-speed, lower-energy injuries, such as soft tissue lacerations and contusions. On the other hand, high-speed injuries occurring from balls, pucks, and sticks may result in more significant trauma. Baseball accounts for the highest percentage of sports-related dental injuries (40.2%), while basketball was second (20.2%) and football third (12.5%). Over 75% of these injuries occurred in males.11
On-field management of dental injuries should always start with the primary trauma survey, including assessment of the athlete’s airway, breathing, and circulatory function, as well as a targeted cervical spine evaluation. When obtaining a history, one should recognize the mechanism of injury and assess for signs of concomitant injuries, ie, respiratory compromise, concussion, leakage of cerebrospinal fluid, and teeth alignment. Findings from this initial evaluation may reveal critical conditions that will require management in addition to the dental injury.
Of central concern in managing dental trauma is preserving the viability of the injured structures. Therefore, much attention is paid to the pulpal and root vitality of injured teeth. The International Association of Dental Traumology Dental Trauma Guidelines recommend a biological approach to the urgent care of dental injuries:12
1. Stabilize the injury by carefully repositioning displaced entities and suturing soft tissue lacerations.
2. Eliminate or reduce the complications from bacterial contamination by rinsing and flushing with available liquids and use of chlorhexidine when possible.
3. Promote the opportunity for healing by replanting avulsed teeth and repositioning displaced teeth.
4. Make every effort to allow continued development of alveolar ridges in children.
Mouth guards are the single most effective prevention strategy for most contact sport dental injuries. One meta-analysis demonstrated a pooled 86% increased risk of orofacial injuries in nonusers.13
To review the anatomy (and injuries) of the tooth, one must consider the Ellis classification of enamel, dentin, and pulp injuries (Figure 2).
Tooth Subluxation
Tooth subluxations usually occur secondary to trauma and cause loosening of the tooth in its alveolar socket. A root fracture should be suspected in the setting of a subluxation. On exam, the tooth may be excessively mobile with gentle pressure. If unstable, immobilization with gauze packing or aluminum foil with dental follow-up is recommended.
Fractures
Ellis class I fractures are small chips in the enamel. There should be uniform color at the fracture site. A dental referral may be warranted to smooth rough enamel edges, but if no other injuries are present, these athletes may continue playing with some protection of the fractured surface. A mouth guard may be helpful to avoid mucosal lacerations.
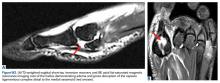
Ellis class II fractures often present with sensitivity to inhaled air and to hot and cold temperatures. Yellow dentin is visible at the fracture site (Figure 3).
Ellis class III fractures may also present with air and temperature sensitivity. Finger pressure may expose a large fracture. Pink or red pulp is visible at the fracture site. Wiping the fracture site with sterile gauze may reveal bleeding from the pulp. This is considered a dental emergency. Immediate restriction from contact sports participation and urgent dental evaluation is indicated for root canal and capping and to prevent abscess formation.
Tooth Avulsion
Tooth avulsions occur when a tooth is completely displaced from the socket (Figure 4).
Skin Issues
Dermatological issues are some of the most common medical conditions faced by a football team physician. Skin infections in particular can pose a significant challenge both diagnostically as well as from a clearance-to-play perspective, given the potential for infections to affect other participants, such as other members of the team. Skin infection rates vary by sport and age group, with one study reporting 28.56 infections per 100,000 athletic exposures in high school wrestlers, which was more than 10 times that of football.14 Still, football players are at a higher risk of skin infections given the contact nature of the sport and close person-to-person proximity. A precise diagnosis may be difficult early in the course of a skin eruption, and with differing guidelines from various professional societies, it may be best suited for medical personnel familiar with these conditions, such as a sports medicine physician or dermatologist, to manage these athletes. A thorough and systematic evaluation is recommended, as athletes are often treated with unnecessary antibiotics, which contributes to antibiotic resistance. Previous antibiotic use may also be a risk factor for developing community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA).15
Two terms sports medicine clinicians must be familiar with are “adequately protected” and “properly covered.” The National Collegiate Athletic Association (NCAA) defines a wound or skin condition as adequately protected when the condition is considered noninfectious, adequately treated by a healthcare provider, and is able to be properly covered. A skin infection is considered properly covered when the lesion is covered by a securely attached bandage or dressing that will contain all drainage and remain intact throughout the sport activity.16
Impetigo
Impetigo is often caused by Staphylococcus and Streptococcus subspecies. The classic presentation is a dry, honey-crusted lesion with an erythematous base. Culture or gram stain may be helpful, but treatment may be initiated on a clinical basis without these studies. Topical antibiotics may be used, but in the setting of multiple lesions or an outbreak, systemic (eg, oral) antibiotics are preferred. Oral antibiotics may also shorten the time to return to play. If not responsive to the initial treatment, MRSA should be considered. No new lesions for 48 hours and a minimum of 72 hours of therapy with no moist, exudative, or draining lesions are required prior to return to play. These lesions cannot be covered as the sole means of return to play.
Methicillin-Resistant Staphylococcus aureus
MRSA is one of the most challenging skin infections for the sports medicine clinician to manage. Several outbreaks have been reported in the high school, college, and professional settings.17-20 Standardized precautions and a proactive approach are key in preventing MRSA outbreaks. It appears that different activities within a given sport may contribute to MRSA risk. One study reported football linemen had the highest attack rate, while another study reported cornerbacks and wide receivers to have the highest rate of MRSA infections.17,20 The elbow area was the most common site infected in both studies.
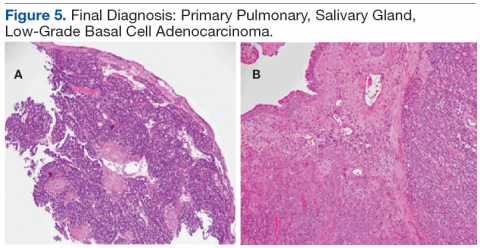
Abscesses are best initially managed by incision and drainage as well as obtaining wound cultures (Figure 5).
Preventative measures are thought to be useful, especially in the management of teams. The Centers for Disease Control and Prevention has published guidelines for both clinicians and patients. Precautions including hand washing; encouraging good overall hygiene; avoiding whirlpools; discouraging the sharing of towels, razors, and athletic gear; maintaining clean equipment/facilities; and encouraging early reporting of skin lesions.14,17,21,22 Isolated cases of MRSA do not need to be reported, but if more than one athlete is infected, one should notify the athletic training and team coaching staff. In the setting of an outbreak, the physician may need to notify local or state health agencies. No new lesions for 48 hours and a minimum of 72 hours of therapy with no moist, exudative, or draining lesions are required prior to returning to play. These lesions cannot be covered as the sole means of return to play.
Tinea Pedis
Tinea pedis is a common dermatophyte infection involving the feet and is most commonly caused by Trichophyton rubrum. Its distribution is usually interdigital or along the plantar surface of the foot. Topical antifungals with either allylamines or azoles are usually sufficient. Terbinafine has been shown to have a shorter duration of treatment. Athletes with tinea pedis are not restricted from sports participation during treatment, as long as the lesions are properly covered.
Tinea Corporis
Tinea corporis is a common superficial fungal infection of the body. It classically presents as pruritic, annular lesions, with well-demarcated borders and central clearing (
Tinea Cruris
Commonly known as “jock-itch,” this fungal infection is often very pruritic and involves the groin or genital region. The area is also inflamed and scaly. Treatment usually consists of topical allylamines or azoles. Allylamines amines are often preferred, as they require a shorter duration of treatment. There are no specific guidelines on the return to play with these athletes. Clearance is at the team physician’s discretion, but usually there are no restrictions. Athletes with extensive lesions may need to be disqualified from contact sports activities.
Am J Orthop. 2016;45(6):377-382. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
Orthopedic conditions are only one of the many medical issues football team physicians may face. In this review, we cover the management of a few of the most common nonorthopedic medical issues football team physicians are likely to encounter, including eye injuries, dental concerns, and skin conditions.
Eye Injuries
More than 2.5 million eye injuries occur each year, with 50,000 people permanently losing part or all of their vision.1 Eye injuries account for over 600,000 yearly emergency department visits; over 30% of these eye injuries were attributed to a sports injury.1 Football is classified as high risk for eye injury, along with baseball, hockey, basketball, and lacrosse.2 Common eye injury mechanisms are categorized as blunt, penetrating, and radiating. Blunt injuries are most common.2 When evaluating an athlete on the sideline, relevant history would include the size of the object, the level of force, and the direction from which the impact occurred. An examination should include best-corrected visual acuity using an eye chart, confrontational visual fields, assessment of extraocular movements, assessment of red reflex, and pupil evaluation with a light source.2
Cornea Injuries
The outermost layer of the eye, the cornea, can be subject to blunt and penetrating injuries. Corneal abrasions often occur from mechanical trauma, such as one from the fingernail of an opposing player, that disrupts the integrity of the corneal epithelium. A corneal abrasion can be identified by applying fluorescein strips after application of a topical anesthetic. Abrasions appear fluorescent green when viewed with a cobalt blue light. If an abrasion is identified, management includes preventing infection and treating pain. Prophylactic topical antibiotics can be applied, particularly for contact lens wearers. Patching has not shown benefit in treatment of pain.3 The physician can consider using topical nonsteroidal anti-inflammatory drugs, such as diclofenac or ketorolac, with a soft contact lens to treat the pain.4 The patient should follow up frequently for monitoring for infection and healing.
Orbital Fractures
Orbital fractures should be considered when an object larger than the orbital opening, such as an elbow or knee, causes blunt trauma to the surrounding bony structures, or a digital poke occurs to the globe.5 The floor of the orbit and medial wall are thin bones that often break sacrificially to protect the globe from rupture. Examination findings may include diplopia, sunken globe, numbness in the distribution of infraorbital nerve, or periorbital emphysema.6 Urgent evaluation should be considered to rule out associated intraocular damage. Imaging and a physical examination can help guide surgical management, if indicated. The most common outcome after this injury is diplopia with upper field gaze.5
Retina Issues
Trauma to the face or head may result in a separation of the retina from the underlying retinal pigment epithelium and allow vitreous fluid to seep in and further separate the layers, causing a retinal detachment. Symptoms may include flashes of light (photopsia), floaters, and visual field defects. Emergent referral is indicated, as the outcomes from this condition are time-sensitive. Consider placing an eye shield to prevent any further pressure on the globe.
Globe Injuries and Rupture
Another emergent ophthalmologic condition that can occur in football is globe rupture. Clinical findings usually prompt the clinician to consider this diagnosis. Hyphema (the collection of blood in the anterior chamber) may be seen in globe injuries. The most common clinical finding of athletes requiring hospitalization after an ocular injury is macroscopic hyphema (Figure 1).7-9
Prompt referral is warranted when there is a sudden decrease or change in vision, pain during movements, photophobia, and floaters and/or flashes.2 Consideration of return to play should take into account the patient’s vision and comfort level, which should not be masked by topical analgesics. Protective eyewear has been mandated in several sports, and has decreased the rate of eye injuries.10 Polycarbonate lenses of 3-mm thickness are recommended due to the significant comparable strength and impact-resistance.2 During the preparticipation physical for high-risk sports, the utilization of protective eyewear should be discussed.
Dental Concerns
Dental injuries may present a challenge for the sports medicine clinician. Contact injuries from elbows, fists, and other nonprojectile objects typically result in low-speed, lower-energy injuries, such as soft tissue lacerations and contusions. On the other hand, high-speed injuries occurring from balls, pucks, and sticks may result in more significant trauma. Baseball accounts for the highest percentage of sports-related dental injuries (40.2%), while basketball was second (20.2%) and football third (12.5%). Over 75% of these injuries occurred in males.11
On-field management of dental injuries should always start with the primary trauma survey, including assessment of the athlete’s airway, breathing, and circulatory function, as well as a targeted cervical spine evaluation. When obtaining a history, one should recognize the mechanism of injury and assess for signs of concomitant injuries, ie, respiratory compromise, concussion, leakage of cerebrospinal fluid, and teeth alignment. Findings from this initial evaluation may reveal critical conditions that will require management in addition to the dental injury.
Of central concern in managing dental trauma is preserving the viability of the injured structures. Therefore, much attention is paid to the pulpal and root vitality of injured teeth. The International Association of Dental Traumology Dental Trauma Guidelines recommend a biological approach to the urgent care of dental injuries:12
1. Stabilize the injury by carefully repositioning displaced entities and suturing soft tissue lacerations.
2. Eliminate or reduce the complications from bacterial contamination by rinsing and flushing with available liquids and use of chlorhexidine when possible.
3. Promote the opportunity for healing by replanting avulsed teeth and repositioning displaced teeth.
4. Make every effort to allow continued development of alveolar ridges in children.
Mouth guards are the single most effective prevention strategy for most contact sport dental injuries. One meta-analysis demonstrated a pooled 86% increased risk of orofacial injuries in nonusers.13
To review the anatomy (and injuries) of the tooth, one must consider the Ellis classification of enamel, dentin, and pulp injuries (Figure 2).
Tooth Subluxation
Tooth subluxations usually occur secondary to trauma and cause loosening of the tooth in its alveolar socket. A root fracture should be suspected in the setting of a subluxation. On exam, the tooth may be excessively mobile with gentle pressure. If unstable, immobilization with gauze packing or aluminum foil with dental follow-up is recommended.
Fractures
Ellis class I fractures are small chips in the enamel. There should be uniform color at the fracture site. A dental referral may be warranted to smooth rough enamel edges, but if no other injuries are present, these athletes may continue playing with some protection of the fractured surface. A mouth guard may be helpful to avoid mucosal lacerations.
Ellis class II fractures often present with sensitivity to inhaled air and to hot and cold temperatures. Yellow dentin is visible at the fracture site (Figure 3).
Ellis class III fractures may also present with air and temperature sensitivity. Finger pressure may expose a large fracture. Pink or red pulp is visible at the fracture site. Wiping the fracture site with sterile gauze may reveal bleeding from the pulp. This is considered a dental emergency. Immediate restriction from contact sports participation and urgent dental evaluation is indicated for root canal and capping and to prevent abscess formation.
Tooth Avulsion
Tooth avulsions occur when a tooth is completely displaced from the socket (Figure 4).
Skin Issues
Dermatological issues are some of the most common medical conditions faced by a football team physician. Skin infections in particular can pose a significant challenge both diagnostically as well as from a clearance-to-play perspective, given the potential for infections to affect other participants, such as other members of the team. Skin infection rates vary by sport and age group, with one study reporting 28.56 infections per 100,000 athletic exposures in high school wrestlers, which was more than 10 times that of football.14 Still, football players are at a higher risk of skin infections given the contact nature of the sport and close person-to-person proximity. A precise diagnosis may be difficult early in the course of a skin eruption, and with differing guidelines from various professional societies, it may be best suited for medical personnel familiar with these conditions, such as a sports medicine physician or dermatologist, to manage these athletes. A thorough and systematic evaluation is recommended, as athletes are often treated with unnecessary antibiotics, which contributes to antibiotic resistance. Previous antibiotic use may also be a risk factor for developing community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA).15
Two terms sports medicine clinicians must be familiar with are “adequately protected” and “properly covered.” The National Collegiate Athletic Association (NCAA) defines a wound or skin condition as adequately protected when the condition is considered noninfectious, adequately treated by a healthcare provider, and is able to be properly covered. A skin infection is considered properly covered when the lesion is covered by a securely attached bandage or dressing that will contain all drainage and remain intact throughout the sport activity.16
Impetigo
Impetigo is often caused by Staphylococcus and Streptococcus subspecies. The classic presentation is a dry, honey-crusted lesion with an erythematous base. Culture or gram stain may be helpful, but treatment may be initiated on a clinical basis without these studies. Topical antibiotics may be used, but in the setting of multiple lesions or an outbreak, systemic (eg, oral) antibiotics are preferred. Oral antibiotics may also shorten the time to return to play. If not responsive to the initial treatment, MRSA should be considered. No new lesions for 48 hours and a minimum of 72 hours of therapy with no moist, exudative, or draining lesions are required prior to return to play. These lesions cannot be covered as the sole means of return to play.
Methicillin-Resistant Staphylococcus aureus
MRSA is one of the most challenging skin infections for the sports medicine clinician to manage. Several outbreaks have been reported in the high school, college, and professional settings.17-20 Standardized precautions and a proactive approach are key in preventing MRSA outbreaks. It appears that different activities within a given sport may contribute to MRSA risk. One study reported football linemen had the highest attack rate, while another study reported cornerbacks and wide receivers to have the highest rate of MRSA infections.17,20 The elbow area was the most common site infected in both studies.
Abscesses are best initially managed by incision and drainage as well as obtaining wound cultures (Figure 5).
Preventative measures are thought to be useful, especially in the management of teams. The Centers for Disease Control and Prevention has published guidelines for both clinicians and patients. Precautions including hand washing; encouraging good overall hygiene; avoiding whirlpools; discouraging the sharing of towels, razors, and athletic gear; maintaining clean equipment/facilities; and encouraging early reporting of skin lesions.14,17,21,22 Isolated cases of MRSA do not need to be reported, but if more than one athlete is infected, one should notify the athletic training and team coaching staff. In the setting of an outbreak, the physician may need to notify local or state health agencies. No new lesions for 48 hours and a minimum of 72 hours of therapy with no moist, exudative, or draining lesions are required prior to returning to play. These lesions cannot be covered as the sole means of return to play.
Tinea Pedis
Tinea pedis is a common dermatophyte infection involving the feet and is most commonly caused by Trichophyton rubrum. Its distribution is usually interdigital or along the plantar surface of the foot. Topical antifungals with either allylamines or azoles are usually sufficient. Terbinafine has been shown to have a shorter duration of treatment. Athletes with tinea pedis are not restricted from sports participation during treatment, as long as the lesions are properly covered.
Tinea Corporis
Tinea corporis is a common superficial fungal infection of the body. It classically presents as pruritic, annular lesions, with well-demarcated borders and central clearing (
Tinea Cruris
Commonly known as “jock-itch,” this fungal infection is often very pruritic and involves the groin or genital region. The area is also inflamed and scaly. Treatment usually consists of topical allylamines or azoles. Allylamines amines are often preferred, as they require a shorter duration of treatment. There are no specific guidelines on the return to play with these athletes. Clearance is at the team physician’s discretion, but usually there are no restrictions. Athletes with extensive lesions may need to be disqualified from contact sports activities.
Am J Orthop. 2016;45(6):377-382. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Owens PL, Mutter R. Emergency Department Visits Related to Eye Injuries, 2008. Agency for Healthcare Research and Quality Web site. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb112.pdf. Published May 2011. Accessed August 18, 2016.
2. Rodriguez JO, Lavina AM, Agarwai A. Prevention and treatment of common eye injuries in sports. Am Fam Physician. 2003;67(7):1481-1496.
3. Lim CH, Turner A, Lim BX. Patching for corneal abrasion. Cochrane Database Syst Rev. 2016;7:CD004764.
4. Weaver CS, Terrell KM. Evidence-based emergency medicine. Update: do ophthalmic nonsteroidal anti-inflammatory drugs reduce the pain associated with simple corneal abrasion without delaying healing? Ann Emerg Med. 2003;41(1):134-140.
5. Williams RJ 3rd, Marx RG, Barnes R, O’Brien SJ, Warren RF. Fractures about the orbit in professional American football players. Am J Sports Med. 2001;29(1):55-57.
6. Forrest LA, Schuller DE, Strauss RH. Management of orbital blow-out fractures. Case reports and discussion. Am J Sports Med. 1989;17(2):217-220.
7. Barr A, Baines PS, Desai P, MacEwen CJ. Ocular sports injuries: the current picture. Br J Sports Med. 2000;34(6):456-458.
8. Pokhrel PK, Loftus SA. Ocular emergencies. Am Fam Physician. 2007;76(6):829-836.
9. Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H. Eye Trauma—Hyphema. The Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013.
10. Lincoln AE, Caswell SV, Almquist JL, et al. Effectiveness of the women’s lacrosse protective eyewear mandate in the reduction of eye injuries. Am J Sports Med. 2012;40(3):611-614.
11. Stewart GB, Shields BJ, Fields S, Comstock RD, Smith GA. Consumer products and activities associated with dental injuries to children treated in United States emergency departments, 1990-2003. Dental Traumatol. 2009;25(4):399-405.
12. Bakland LK. Dental trauma guidelines. Pediatric Dent. 2013;35(2):106-108.
13. Knapik J, Marshall SW, Lee RB, et al. Mouthguards in sport activities: history, physical properties and Injury prevention effectiveness. Sports Med. 2007;37(2):117-144.
14. Ashack KA, Burton KA, Johnson TR, Currie DW, Comstock RD, Dellavalle RP. Skin infections among US high school athletes: a national survey. J Am Acad Dermatol. 2016;74(4):679-684.e1.
15. Ellis MW, Hospenthal DR, Dooley DP, Gray PJ, Murray CK. Natural history of community-acquired methicillin-resistant Staphylococcus aureus colonization and infection in soldiers. Clin Infect Dis. 2004;39(7):971-979.
16. The National Collegiate Athletic Association. 2014-15 NCAA Sports Medicine Handbook. http://www.ncaapublications.com/productdownloads/MD15.pdf. Revised June 2008. Accessed August 18, 2016.
17. Anderson BJ. The effectiveness of valacyclovir in preventing reactivation of herpes gladiatorum in wrestlers. Clin J Sport Med. 1999;9(2):86-90.
18. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55.
19. Jeffords MD, Batts K. Dermatology. In: O’Connor FG, Casa DJ, Davis BA, Pierre PS, Sallis RE, Wilder RP, eds. ACSM’s Sports Medicine: A Comprehensive Review. Riverwoods, IL: Wolters Kluwer; 2016:181-188.
20. Kazakova SV, Hageman JC, Matava M, et al. A clone of methicillin-resistant Staphylococcus aureus among professional football players. N Engl J Med. 2005;352(5):468-475.
21. Begier EM, Frenette K, Barrett NL, et al. A high-morbidity outbreak of methicillin-resistant Staphylococcus aureus among players on a college football team, facilitated by cosmetic body shaving and turf burns. Clin Infect Dis. 2004;39(10):1446-1453.
22. Geissler KE, Borchers JR. More than meets the eye: a rapidly progressive skin infection in a football player. Clin J Sport Med. 2015;25(3):e54-e56.
1. Owens PL, Mutter R. Emergency Department Visits Related to Eye Injuries, 2008. Agency for Healthcare Research and Quality Web site. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb112.pdf. Published May 2011. Accessed August 18, 2016.
2. Rodriguez JO, Lavina AM, Agarwai A. Prevention and treatment of common eye injuries in sports. Am Fam Physician. 2003;67(7):1481-1496.
3. Lim CH, Turner A, Lim BX. Patching for corneal abrasion. Cochrane Database Syst Rev. 2016;7:CD004764.
4. Weaver CS, Terrell KM. Evidence-based emergency medicine. Update: do ophthalmic nonsteroidal anti-inflammatory drugs reduce the pain associated with simple corneal abrasion without delaying healing? Ann Emerg Med. 2003;41(1):134-140.
5. Williams RJ 3rd, Marx RG, Barnes R, O’Brien SJ, Warren RF. Fractures about the orbit in professional American football players. Am J Sports Med. 2001;29(1):55-57.
6. Forrest LA, Schuller DE, Strauss RH. Management of orbital blow-out fractures. Case reports and discussion. Am J Sports Med. 1989;17(2):217-220.
7. Barr A, Baines PS, Desai P, MacEwen CJ. Ocular sports injuries: the current picture. Br J Sports Med. 2000;34(6):456-458.
8. Pokhrel PK, Loftus SA. Ocular emergencies. Am Fam Physician. 2007;76(6):829-836.
9. Usatine RP, Smith MA, Mayeaux EJ Jr, Chumley H. Eye Trauma—Hyphema. The Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013.
10. Lincoln AE, Caswell SV, Almquist JL, et al. Effectiveness of the women’s lacrosse protective eyewear mandate in the reduction of eye injuries. Am J Sports Med. 2012;40(3):611-614.
11. Stewart GB, Shields BJ, Fields S, Comstock RD, Smith GA. Consumer products and activities associated with dental injuries to children treated in United States emergency departments, 1990-2003. Dental Traumatol. 2009;25(4):399-405.
12. Bakland LK. Dental trauma guidelines. Pediatric Dent. 2013;35(2):106-108.
13. Knapik J, Marshall SW, Lee RB, et al. Mouthguards in sport activities: history, physical properties and Injury prevention effectiveness. Sports Med. 2007;37(2):117-144.
14. Ashack KA, Burton KA, Johnson TR, Currie DW, Comstock RD, Dellavalle RP. Skin infections among US high school athletes: a national survey. J Am Acad Dermatol. 2016;74(4):679-684.e1.
15. Ellis MW, Hospenthal DR, Dooley DP, Gray PJ, Murray CK. Natural history of community-acquired methicillin-resistant Staphylococcus aureus colonization and infection in soldiers. Clin Infect Dis. 2004;39(7):971-979.
16. The National Collegiate Athletic Association. 2014-15 NCAA Sports Medicine Handbook. http://www.ncaapublications.com/productdownloads/MD15.pdf. Revised June 2008. Accessed August 18, 2016.
17. Anderson BJ. The effectiveness of valacyclovir in preventing reactivation of herpes gladiatorum in wrestlers. Clin J Sport Med. 1999;9(2):86-90.
18. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18-e55.
19. Jeffords MD, Batts K. Dermatology. In: O’Connor FG, Casa DJ, Davis BA, Pierre PS, Sallis RE, Wilder RP, eds. ACSM’s Sports Medicine: A Comprehensive Review. Riverwoods, IL: Wolters Kluwer; 2016:181-188.
20. Kazakova SV, Hageman JC, Matava M, et al. A clone of methicillin-resistant Staphylococcus aureus among professional football players. N Engl J Med. 2005;352(5):468-475.
21. Begier EM, Frenette K, Barrett NL, et al. A high-morbidity outbreak of methicillin-resistant Staphylococcus aureus among players on a college football team, facilitated by cosmetic body shaving and turf burns. Clin Infect Dis. 2004;39(10):1446-1453.
22. Geissler KE, Borchers JR. More than meets the eye: a rapidly progressive skin infection in a football player. Clin J Sport Med. 2015;25(3):e54-e56.
In My Athletic Trainer’s Bag
Editor’s Note: Doug Quon, MAT, ATC, PES, is the Assistant Athletic Trainer for the Washington Redskins. Click the PDF button below to view and download his list of the essential components of an athletic trainer’s bag for high school football
Editor’s Note: Doug Quon, MAT, ATC, PES, is the Assistant Athletic Trainer for the Washington Redskins. Click the PDF button below to view and download his list of the essential components of an athletic trainer’s bag for high school football
Editor’s Note: Doug Quon, MAT, ATC, PES, is the Assistant Athletic Trainer for the Washington Redskins. Click the PDF button below to view and download his list of the essential components of an athletic trainer’s bag for high school football
Knee Injuries in American Football: An Epidemiological Review
Football is one of the most popular sports in the United States. Every year more than 1 million high school males and over 60,000 collegiate males participate in organized football. The number of males who play football is greater than the combined number of males and females who participate in track and field or basketball.1 Football has the highest injury rate amongst popular American sports.2 From 2001 to 2005, there was an estimated 1.1 million emergency room visits as a direct result of football.3 Injuries are more likely to occur during games,1,2,4,5 more likely to require surgery,4 and more likely to end the player’s season or career when compared to other sports.6 Of those injuries that end seasons or careers, the knee is the most common culprit.6 This is of particular concern because knee injuries are most common in football.1,2,5,7 This article reviews the epidemiology of 4 of the most common knee injuries in American football: tears of the anterior cruciate ligament (ACL), medial collateral ligament (MCL), medial patellofemoral ligament (MPFL), and posterior cruciate ligament (PCL).
Anterior Cruciate Ligament
The ACL is the primary structure preventing anterior tibial translation. It is composed of 2 anatomic bundles: the anteromedial and posterolateral bundles. The ACL originates from the posteromedial portion of the lateral femoral condyle and inserts between and slightly anterior to the tibial intercondylar eminence. The bundles are named for their relative insertions onto the tibia.
Injury to the ACL occurs both through noncontact and contact mechanisms. Typical noncontact mechanism is a forceful valgus collapse with the knee close to full extension with combined external or internal rotation of the tibia.8 This is often the result of a sudden deceleration prior to a change in direction.9 Contact injuries to the ACL are the result of a direct blow to the knee causing valgus collapse.9 The majority of ACL injuries amongst all sports are a result of a noncontact mechanism. However, Dragoo and colleagues10 found the majority of football ACL injuries (55%-60%) were from contact. As a result, football players are 4 times more likely to sustain ACL injuries than in other sports.11
ACL injuries are associated with significant time loss from sport. At the high school level, they are the most likely injury to end a season or career.6 Because these are higher-energy injuries, they are frequently associated with damage to additional structures. ACL injuries that occur in football are associated with increased rates of meniscus, chondral, and multi-ligamentous injuries.12,13
The incidence of ACL injuries increases with level of competition. In high school athletes it is 11.1 per 100,000 athlete exposures (AE).1,11 In collegiate football, the rate increases to 14.2 to 18 per 100,000 AE.2,14 Though no incidence data per AE was found in our review of the literature, there were 219 ACL injuries in the National Football League (NFL) from 2010 to 2013.15 In addition, 14.2% of retired NFL athletes in one survey reported a history of ACL injury.16
The most common high-risk positions are running backs and linebackers. Brophy and colleagues17 found that 9.7% of running backs and 8.9% of linebackers participating in the NFL Combine had a history of ACL injury. This may be because both the running back and linebacker are involved in frequent high-energy collisions and often quickly change direction. Other studies have also identified running backs and linebackers as high risk, in addition to tight ends, wide receivers, and interior linemen.13,15,18
Treatment of choice for elite level athletes with ACL injury is reconstruction.19 Of those who undergo ACL reconstruction, the rate of return to play ranges from 63% to 80%.20-22 The average time to return to play is 9 to 13 months. The odds of making a successful return hinges on how successful the athlete was prior to injury. Factors such as prior game experience, position on depth chart, being on scholarship, and draft position for NFL athletes have all been shown to have a positive predictive value on a patient’s chance of returning from ACL reconstruction.20,21
Players who return have variable levels of success afterwards. In a study of NFL quarterbacks who sustained ACL injuries, 12 out of 13 were able to return to game action with no appreciable dropoff in performance based on in-game production.23 Carey and colleagues24 looked specifically at NFL wide receivers and running backs and found an 80% return to play rate but with an approximate decrease in production of one-third upon return. Furthermore, in the Multicenter Orthopaedic Outcomes Network (MOON) cohort study, only 43% of participants felt they returned to their preoperative level.22
Medial Collateral Ligament
The MCL consists of superficial and deep components. The superficial MCL is the primary restraint to valgus laxity at the knee. The superficial MCL has 1 femoral and 2 tibial attachments. The deep MCL is a thickening of the medial joint capsule and runs deep and parallel to the superficial MCL. The amount of medial joint gapping with a valgus force on examination is used to grade severity of MCL injuries. Grade I is a <5-mm opening; Grade II, 5- to 10-mm opening; and grade III, >10-mm opening.
The MCL is the most common knee injury in high school, collegiate, and professional football.1,18,25-28 Injuries are typically due to contact when a valgus force is applied to the knee.29 The annual incidence of MCL injuries amongst high school football players is 24.2 per 100,000 AE.1 The positions that appear to be at greatest risk for MCL injuries are offensive and defensive linemen.18,30-32 In a review of 5047 collegiate athletes participating in the NFL Combine from 1987 to 2000, 23% of offensive linemen had a history of MCL injury, compared to the overall rate of 16%.33 In a similar study, Bradley and colleagues18 performed medical histories on athletes invited to the 2005 NFL Combine and also found offensive linemen had the highest rate of MCL injury at 33%, compared to the overall rate of 22%. They reasonably hypothesized that “chop blocks” and other players “rolling up” on the outside of linemen’s knees were responsible for these injuries. Albright and colleagues32 found that prophylactic knee braces decreased the incidence of MCL injuries in collegiate offensive lineman. However, additional studies have not been able to reproduce these results and the use of prophylactic knee braces remains controversial.26
Treatment of MCL injuries depends upon the grade of injury, associated injuries, and anatomical location of injury. Management of MCL injuries is for the most part nonsurgical. In 1974, Ellsasser and colleagues34 were the first to publish data on nonoperative management of Grade I and Grade II injuries with immediate motion and rehabilitation instead of cast immobilization. They found 93% of patients returned to football in 3 to 8 weeks.34 Derscheid and Garrick27 observed nonoperative treatment of Grade I and II sprains in collegiate football players, with a time loss of 10.6 days and 19.5 days for Grade I and II injuries, respectively. Holden and colleagues35 evaluated nonoperative management of Grade I and II MCL injuries in collegiate football players and found an average return to play of 21 days.
Grade III injury treatment is more controversial. Indelicato and colleagues36 demonstrated successful nonoperative management of Grade III MCL injuries in collegiate football players, with an average return to play of 64.4 days. Jones and colleagues37 had similar success with high school football players, with an average return to play of 34 days. However, isolated Grade III injuries are rare and therefore treatment is likely to be dictated by concomitant injuries. Fetto and Marshall38 found that 78% of Grade III injuries were associated with an additional ligamentous injury. Of those additional injuries, 95% were ACL tears.
Finally, one must consider the location of the MCL injury. Injuries of the distal MCL at its tibial insertion may result in poor healing, as the ligament is displaced away from its insertion. Therefore, some authors recommend surgical management for these injuries.39,40
The patellofemoral joint is a complex structure in which the patella is stabilized within the trochlear groove of the femur by both bony and soft tissue structures. The MPFL is one of the most important soft tissue stabilizers. The MPFL is the primary restraint to lateral patellar translation within the first 20° of knee flexion, contributing to 60% of the total restraining force.41 The MPFL originates on the medial femoral condyle and inserts on the superomedial aspect of the patella.
Patellar instability is the subluxation or dislocation of the patella out of the trochlear groove. Patellar subluxation and dislocation account for approximately 3% of all knee injuries.42 Patella dislocations are more common in younger populations43-45 with the majority (52%-63%) occurring during sports.43,44,46 Mitchell and colleagues47 reported an incidence of 4.1 patellar subluxations/dislocations per 100,000 AE in high school football players.
Dislocation is most commonly the result of knee flexion with the tibia in a valgus position.44,48 The majority of patellar dislocations occur via a noncontact mechanism.44,48 However, the majority of these injuries in football are from contact (63%).47
Acute patellar dislocations are associated with more soft tissue damage than those with recurrent dislocations.46 In acute patella dislocations, the MPFL is almost always ruptured.44 In contrast, Fithian and colleagues46 found only 38% of recurrent dislocators had MPFL injury. As a result, it is thought that those with recurrent instability dislocate without trauma and do not have the same characteristics as those who dislocate from high-energy trauma in sport. Risk factors for atraumatic dislocation are numerous and have been well described in the literature.49 However, traumatic dislocators usually do not have risk factors.50
Traumatic patella dislocations are higher energy and are associated with chondral injury in up to 95%of cases 51 and osteochondral injury 58% to 76% of the time.52,53 In contrast, people with “articular hypermobility” are less likely to sustain articular damage.54 This concept is important when considering risk for recurrent patella dislocation. The literature reports a 17% to 50% rate of recurrent instability after acute patella dislocation.46,55,56 However, most studies do not distinguish between traumatic and atraumatic injuries. Because the majority of patellar dislocations in football occur through contact mechanisms, the rate of recurrent instability in these athletes may in fact be less than what is reported in the literature.
First-time patella dislocations are generally treated nonoperatively. Mitchell and colleagues47 reported that 72.6% of high school athletes with patella subluxation treated conservatively were able to return to sports within 3 weeks, compared to only 34.1% of those with patellar dislocations. In the same study, patellar dislocations were season-ending 37% of the time.47 Atkin and colleagues50 followed 74 patients treated conservatively for first-time patellar dislocation and noted 58% at 6 months still had difficulty in squatting, jumping, or cutting.
Those who have failed conservative management and have an additional dislocation are 7 times more likely to redislocate.46 Therefore, they are usually treated operatively with MPFL reconstruction. Return to sport ranges from 3 to 6 months,57 with 53% to 77.3% reporting return to their previous functionality.57-59 Overall, 84.1% of patients are able to return to sport with 1.2% risk of recurrent dislocation.60
Posterior Cruciate Ligament
The PCL is the primary posterior stabilizer of the knee.61,62 It consists of the anterolateral and posteromedial bundles, named by their insertion on the posterior tibial plateau. The larger, stronger anterolateral bundle is the primary restraint to posterior tibial translation.63
Due to the relative infrequency of PCL injuries, there is a paucity of epidemiological data on sports-related PCL injuries. These injuries in the literature are commonly found due to traffic accidents (45%-57%) or from sports (33%-40%).64,65 According to Swensen and colleagues,1 PCL injuries account for 2.4% of all high school sport knee injuries. In a cohort of 62 knees with PCL injuries, Patel and colleagues66 found football was the most common cause of injury (19.3%).
The most common mechanism of injury in athletes is knee hyperflexion or a direct blow to the tibia in a flexed knee.67 In football, contact mechanisms are the most common. In a 16-year review of the National Collegiate Athletic Association (NCAA) injury surveillance system, the incidence of contact PCL injuries during games were 7.3 times higher than noncontact.68 The most common activity was being tackled, which accounted for 22.9% of all PCL injuries.68
Due to the high energy of these injuries, isolated PCL injuries are rare. In one trauma center’s experience, 96.5% of PCL injuries had an additional ligament injury.64 In that study, injuries to the PCL were associated with posterolateral corner, ACL, and MCL injuries 62%, 46%, and 31% of the time, respectively.64,69
Because isolated PCL injuries are rare, clinicians must rely on a thorough history and physical examination when evaluating athletes with knee injuries. Classification of PCL injuries is based on the amount of posterior tibial translation in relation to the femur with the knee bent to 90°. Grade I is 1 to 5 mm; Grade II, 6 to 10 mm; and Grade III, >10 mm. If there is suspicion of a PCL injury, there should be a very low threshold for magnetic resonance imaging, given the high association with additional injuries.
Natural history of Grade I and II isolated PCL injuries is generally favorable compared to Grade III and multi-ligamentous injuries.70 As a result, isolated Grade I and II PCL injuries are generally treated nonoperatively. Treatment consists of physical therapy with emphasis on quadriceps strengthening. Return to play can be considered as early as 2 to 4 weeks from injury.71 Recent long-term data have shown successful conservative management of Grade I and II injuries with quadriceps strength to 97% of contralateral leg and full range of motion.72 However, there was 11% moderate to severe osteoarthritis in these patients at a mean follow-up of 14.3 years.72 Fowler and Messieh67 managed athletes with 7 isolated complete PCL tears and 5 partial tears nonoperatively, all of whom were able to return to sport without limitation. Parolie and Bergfeld73 managed 25 athletes with isolated PCL tears conservatively. In this study, 80% of athletes reported satisfaction and 68% returned to previous level of play.73 Neither of the aforementioned studies specify the grades of the injuries. Finally, Patel and colleagues66 managed 6 NFL athletes with Grade I and II injuries nonoperatively, and all were able to return to sport.
Treatment of isolated Grade III PCL injuries is more controversial, and no consensus exists in the literature. In an epidemiological study, Dick and colleagues68 found that only 39% of NCAA football athletes underwent surgery for their torn PCLs, compared to 79% of ACL injuries. However, their study makes no mention to the severity of these injuries. Numerous options exist for PCL reconstruction, with no consensus on the preferred method.
Conclusion
Knee injuries are the most common injury in football. Knowledge of the natural history of these injuries, as well as treatment options and expected outcomes, will help treating physicians educate their patients on the optimal treatment and manage return to play expectations.
Am J Orthop. 2016;45(6):368-373. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Swenson DM, Collins CL, Best TM, Flanigan DC, Fields SK, Comstock RD. Epidemiology of knee injuries among U.S. high school athletes, 2005/2006-2010/2011. Med Sci Sports Exerc. 2013;45(3):462-469.
2. Hootman JM, Dick R, Agel J. Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J Athl Train. 2007;42(2):311-319.
3. Mello MJ, Myers R, Christian JB, Palmisciano L, Linakis JG. Injuries in youth football: national emergency department visits during 2001-2005 for young and adolescent players. Acad Emerg Med. 2009;16(3):243-248.
4. Rechel JA, Collins CL, Comstock RD. Epidemiology of injuries requiring surgery among high school athletes in the United States, 2005 to 2010. J Trauma. 2011;71(4):982-989.
5. Ingram JG, Fields SK, Yard EE, Comstock RD. Epidemiology of knee injuries among boys and girls in US high school athletics. Am J Sports Med. 2008;36(6):1116-1122.
6. Tirabassi J, Brou L, Khodaee M, Lefort R, Fields SK, Comstock RD. Epidemiology of high school sports-related injuries resulting in medical disqualification: 2005-2006 through 2013-2014 academic years. Am J Sports Med. 2016 May 10. [Epub ahead of print]
7. Fernandez WG, Yard EE, Comstock RD. Epidemiology of lower extremity injuries among U.S. high school athletes. Acad Emerg Med. 2007;14(7):641-645.
8. Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. Am J Sports Med. 2004;32(4):1002-1012.
9. Boden BP, Dean GS, Feagin JA Jr, Garrett WE Jr. Mechanisms of anterior cruciate ligament injury. Orthopedics. 2000;23(6):573-578.
10. Dragoo JL, Braun HJ, Harris AH. The effect of playing surface on the incidence of ACL injuries in National Collegiate Athletic Association American Football. Knee. 2013;20(3):191-195.
11. Joseph AM, Collins CL, Henke NM, Yard EE, Fields SK, Comstock RD. A multisport epidemiologic comparison of anterior cruciate ligament injuries in high school athletics. J Athl Train. 2013;48(6):810-817.
12. Granan LP, Inacio MC, Maletis GB, Funahashi TT, Engebretsen L. Sport-specific injury pattern recorded during anterior cruciate ligament reconstruction. Am J Sports Med. 2013;41(12):2814-2818.
13. Bradley JP, Klimkiewicz JJ, Rytel MJ, Powell JW. Anterior cruciate ligament injuries in the National Football League: epidemiology and current treatment trends among team physicians. Arthroscopy. 2002;18(5):502-509.
14. Dragoo JL, Braun HJ, Durham JL, Chen MR, Harris AH. Incidence and risk factors for injuries to the anterior cruciate ligament in National Collegiate Athletic Association football: data from the 2004-2005 through 2008-2009 National Collegiate Athletic Association Injury Surveillance System. Am J Sports Med. 2012;40(5):990-995.
15. Dodson CC, Secrist ES, Bhat SB, Woods DP, Deluca PF. Anterior cruciate ligamenti in National Football League athletes from 2010 to 2013: a descriptive epidemiology study. Orthop J Sports Med. 2016;4(3):2325967116631949.
16. Golightly YM, Marshall SW, Callahan LF, Guskiewicz K. Early-onset arthritis in retired National Football League players. J Phys Act Health. 2009;6(5):638-643.
17. Brophy RH, Lyman S, Chehab EL, Barnes RP, Rodeo SA, Warren RF. Predictive value of prior injury on career in professional American football is affected by player position. Am J Sports Med. 2009;37(4):768-775.
18. Bradley J, Honkamp NJ, Jost P, West R, Norwig J, Kaplan LD. Incidence and variance of knee injuries in elite college football players. Am J Orthop. 2008;37(6):310-314.
19. Erickson BJ, Harris JD, Fillingham YA, et al. Anterior cruciate ligament reconstruction practice patterns by NFL and NCAA football team physicians. Arthroscopy. 2014;30(6):731-738.
20. Daruawalla JH, Greis PE, Hancock R; ASP Collaborative Group, Xerogeanes JW. Rates and determinants of return to play after anterior cruciate ligament reconstruction in NCAA Division 1 college football athletes: a study of the ACC, SEC, and PAC-12 conferences. Orthop J Sports Med. 2014;2(8):2325967114543901.
21. Shah VM, Andrews JR, Fleisig GS, McMichael CS, Lemak LJ. Return to play after anterior cruciate ligament reconstruction in National Football League athletes. Am J Sports Med. 2010;38(11):2233-2239.
22. McCullough KA, Phelps KD, Spindler KP, et al. Return to high school- and college-level football after anterior cruciate ligament reconstruction: a Multicenter Orthopaedic Outcomes Network (MOON) cohort study. Am J Sports Med. 2012;40(11):2523-2529.
23. Erickson BJ, Harris JD, Heninger JR, et al. Performance and return-to-sport after ACL reconstruction in NFL quarterbacks. Orthopedics. 2014;37(8):e728-e734.
24. Carey JL, Huffman GR, Parekh SG, Sennett BJ. Outcomes of anterior cruciate ligament injuries to running backs and wide receivers in the National Football League. Am J Sports Med. 2006;34(12):1911-1917.
25. Hershman EB, Anderson R, Bergfeld JA, et al. An analysis of specific lower extremity injury rates on grass and FieldTurf playing surfaces in National Football League Games: 2000-2009 seasons. Am J Sports Med. 2012;40(10):2200-2205.
26. Salata MJ, Gibbs AE, Sekiya JK. The effectiveness of prophylactic knee bracing in American football: a systematic review. Sports Health. 2010;2(5):375-379.
27. Derscheid GL, Garrick JG. Medial collateral ligament injuries in football. Nonoperative management of grade I and grade II sprains. Am J Sports Med. 1981;9(6):365-368.
28. Meyers MC, Barnhill BS. Incidence, causes, and severity of high school football injuries on FieldTurf versus natural grass: a 5-year prospective study. Am J Sports Med. 2004;32(7):1626-1638.
29. Lundblad M, Waldén M, Magnusson H, Karlsson J, Ekstrand J. The UEFA injury study: 11-year data concerning 346 MCL injuries and time to return to play. Br J Sports Med. 2013;47(12):759-762.
30. Hewson GF Jr, Mendini RA, Wang JB. Prophylactic knee bracing in college football. Am J Sports Med. 1986;14(4):262-266.
31. Rovere GD, Haupt HA, Yates CS. Prophylactic knee bracing in college football. Am J Sports Med. 1987;15(2):111-116.
32. Albright JP, Powell JW, Smith W, et al. Medial collateral ligament knee sprains in college football. Brace wear preferences and injury risk. Am J Sports Med. 1994;22(1):2-11.
33. Brophy RH, Barnes R, Rodeo SA, Warren RF. Prevalence of musculoskeletal disorders at the NFL Combine--trends from 1987 to 2000. Med Sci Sports Exerc. 2007;39(1):22-27.
34. Ellsasser JC, Reynolds FC, Omohundro JR. The non-operative treatment of collateral ligament injuries of the knee in professional football players. An analysis of seventy-four injuries treated non-operatively and twenty-four injuries treated surgically. J Bone Joint Surg Am. 1974;56(6):1185-1190.
35. Holden DL, Eggert AW, Butler JE. The nonoperative treatment of grade I and II medial collateral ligament injuries to the knee. Am J Sports Med. 1983;11(5):340-344.
36. Indelicato PA, Hermansdorfer J, Huegel M. Nonoperative management of complete tears of the medial collateral ligament of the knee in intercollegiate football players. Clin Orthop Relat Res. 1990;(256):174-177.
37. Jones RE, Henley MB, Francis P. Nonoperative management of isolated grade III collateral ligament injury in high school football players. Clin Orthop Relat Res. 1986;(213):137-140.
38. Fetto JF, Marshall JL. Medial collateral ligament injuries of the knee: a rationale for treatment. Clin Orthop Relat Res. 1978;(132):206-218.
39. Corten K, Hoser C, Fink C, Bellemans J. Case reports: a Stener-like lesion of the medial collateral ligament of the knee. Clin Orthop Relat Res. 2010;468(1):289-293.
40. Marchant MH Jr, Tibor LM, Sekiya JK, Hardaker WT Jr, Garrett WE Jr, Taylor DC. Management of medial-sided knee injuries, part 1: medial collateral ligament. Am J Sports Med. 2011;39(5):1102-1113.
41. Desio SM, Burks RT, Bachus KN. Soft tissue restraints to lateral patellar translation in the human knee. Am J Sports Med. 1998;26(1):59-65.
42. Casteleyn PP, Handelberg F. Arthroscopy in the diagnosis of occult dislocation of the patella. Acta Orthop Belg. 1989;55(3):381-383.
43. Waterman BR, Belmont PJ Jr, Owens BD. Patellar dislocation in the United States: role of sex, age, race, and athletic participation. J Knee Surg. 2012;25(1):51-57.
44. Sillanpää P, Mattila VM, Iivonen T, Visuri T, Pihlajamäki H. Incidence and risk factors of acute traumatic primary patellar dislocation. Med Sci Sports Exerc. 2008;40(4):606-611.
45. Hsiao M, Owens BD, Burks R, Sturdivant RX, Cameron KL. Incidence of acute traumatic patellar dislocation among active-duty United States military service members. Am J Sports Med. 2010;38(10):1997-2004.
46. Fithian DC, Paxton EW, Stone ML, et al. Epidemiology and natural history of acute patellar dislocation. Am J Sports Med. 2004;32(5):1114-1121.
47. Mitchell J, Magnussen RA, Collins CL, et al. Epidemiology of patellofemoral instability injuries among high school athletes in the United States. Am J Sports Med. 2015;43(7):1676-1682.
48. Nikku R, Nietosvaara Y, Aalto K, Kallio PE. The mechanism of primary patellar dislocation: trauma history of 126 patients. Acta Orthop. 2009;80(4):432-434.
49. Tsai CH, Hsu CJ, Hung CH, Hsu HC. Primary traumatic patellar dislocation. J Orthop Surg Res. 2012;7:21.
50. Atkin DM, Fithian DC, Marangi KS, Stone ML, Dobson BE, Mendelsohn C. Characteristics of patients with primary acute lateral patellar dislocation and their recovery within the first 6 months of injury. Am J Sports Med. 2000;28(4):472-479.
51. Nomura E, Inoue M, Kurimura M. Chondral and osteochondral injuries associated with acute patellar dislocation. Arthroscopy. 2003;19(7):717-721.
52. Kirsch MD, Fitzgerald SW, Friedman H, Rogers LF. Transient lateral patellar dislocation: diagnosis with MR imaging. AJR Am J Roentgenol. 1993;161(1):109-113.
53. Virolainen H, Visuri T, Kuusela T. Acute dislocation of the patella: MR findings. Radiology. 1993;189(1):243-246.
54. Stanitski CL. Articular hypermobility and chondral injury in patients with acute patellar dislocation. Am J Sports Med. 1995;23(2):146-150.
55. Mäenpää H, Huhtala H, Lehto MU. Recurrence after patellar dislocation. Redislocation in 37/75 patients followed for 6-24 years. Acta Orthop Scand. 1997;68(5):424-426.
56. Buchner M, Baudendistel B, Sabo D, Schmitt H. Acute traumatic primary patellar dislocation: long-term results comparing conservative and surgical treatment. Clin J Sport Med. 2005;15(2):62-66.
57. Fisher B, Nyland J, Brand E, Curtin B. Medial patellofemoral ligament reconstruction for recurrent patellar dislocation: a systematic review including rehabilitation and return-to-sports efficacy. Arthroscopy. 2010;26(10):1384-1394.
58. Lippacher S, Dreyhaupt J, Williams SR, Reichel H, Nelitz M. Reconstruction of the medial patellofemoral ligament: clinical outcomes and return to sports. Am J Sports Med. 2014;42(7):1661-1668.
59. Panni AS, Alam M, Cerciello S, Vasso M, Maffulli N. Medial patellofemoral ligament reconstruction with a divergent patellar transverse 2-tunnel technique. Am J Sports Med. 2011;39(12):2647-1655.
60. Schneider DK, Grawe B, Magnussen RA, et al. Outcomes after isolated medial patellofemoral ligament reconstruction for the treatment of recurrent lateral patellar dislocations: a systematic review and meta-analysis. Am J Sports Med. 2016 Feb 12. [Epub ahead of print]
61. Amis AA, Bull AM, Gupte CM, Hijazi I, Race A, Robinson JR. Biomechanics of the PCL and related structures: posterolateral, posteromedial and meniscofemoral ligaments. Knee Surg Sports Traumatol Arthrosc. 2003;11(5):271-281.
62. Fu FH, Harner CD, Johnson DL, Miller MD, Woo SL. Biomechanics of knee ligaments: basic concepts and clinical application. Instr Course Lect. 1994;43:137-148.
63. Markolf KL, Feeley BT, Tejwani SG, Martin DE, McAllister DR. Changes in knee laxity and ligament force after sectioning the posteromedial bundle of the posterior cruciate ligament. Arthroscopy. 2006; 22(10):1100-1106.
64. Ganelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526-529.
65. Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186-191.
66. Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT. The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J. 2007;3(2):137-146.
67. Fowler PJ, Messieh SS. Isolated posterior cruciate ligament injuries in athletes. Am J Sports Med. 1987;15(6):553-557.
68. Dick R, Ferrara MS, Agel J, et al. Descriptive epidemiology of collegiate men’s football injuries: National Collegiate Athletic Association Injury Surveillance System, 1988-1989 through 2003-2004. J Athl Train. 2007;42(2):221-233.
69. LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077-3092.
70. Torg JS, Barton TM, Pavlov H, Stine R. Natural history of the posterior cruciate ligament-deficient knee. Clin Orthop Relat Res. 1989(246):208-216.
71. Miller MD. Orthopaedic Knowledge Update: Sports Medicine 5. Rosemont, IL; American Academy of Orthopaedic Surgeons; 2016.
72. Shelbourne KD, Clark M, Gray T. Minimum 10-year follow-up of patients after an acute, isolated posterior cruciate ligament injury treated nonoperatively. Am J Sports Med. 2013;41(7):1526-1533.
73. Parolie JM, Bergfeld JA. Long-term results of nonoperative treatment of isolated posterior cruciate ligament injuries in the athlete. Am J Sports Med. 1986;14(1):35-38.
Football is one of the most popular sports in the United States. Every year more than 1 million high school males and over 60,000 collegiate males participate in organized football. The number of males who play football is greater than the combined number of males and females who participate in track and field or basketball.1 Football has the highest injury rate amongst popular American sports.2 From 2001 to 2005, there was an estimated 1.1 million emergency room visits as a direct result of football.3 Injuries are more likely to occur during games,1,2,4,5 more likely to require surgery,4 and more likely to end the player’s season or career when compared to other sports.6 Of those injuries that end seasons or careers, the knee is the most common culprit.6 This is of particular concern because knee injuries are most common in football.1,2,5,7 This article reviews the epidemiology of 4 of the most common knee injuries in American football: tears of the anterior cruciate ligament (ACL), medial collateral ligament (MCL), medial patellofemoral ligament (MPFL), and posterior cruciate ligament (PCL).
Anterior Cruciate Ligament
The ACL is the primary structure preventing anterior tibial translation. It is composed of 2 anatomic bundles: the anteromedial and posterolateral bundles. The ACL originates from the posteromedial portion of the lateral femoral condyle and inserts between and slightly anterior to the tibial intercondylar eminence. The bundles are named for their relative insertions onto the tibia.
Injury to the ACL occurs both through noncontact and contact mechanisms. Typical noncontact mechanism is a forceful valgus collapse with the knee close to full extension with combined external or internal rotation of the tibia.8 This is often the result of a sudden deceleration prior to a change in direction.9 Contact injuries to the ACL are the result of a direct blow to the knee causing valgus collapse.9 The majority of ACL injuries amongst all sports are a result of a noncontact mechanism. However, Dragoo and colleagues10 found the majority of football ACL injuries (55%-60%) were from contact. As a result, football players are 4 times more likely to sustain ACL injuries than in other sports.11
ACL injuries are associated with significant time loss from sport. At the high school level, they are the most likely injury to end a season or career.6 Because these are higher-energy injuries, they are frequently associated with damage to additional structures. ACL injuries that occur in football are associated with increased rates of meniscus, chondral, and multi-ligamentous injuries.12,13
The incidence of ACL injuries increases with level of competition. In high school athletes it is 11.1 per 100,000 athlete exposures (AE).1,11 In collegiate football, the rate increases to 14.2 to 18 per 100,000 AE.2,14 Though no incidence data per AE was found in our review of the literature, there were 219 ACL injuries in the National Football League (NFL) from 2010 to 2013.15 In addition, 14.2% of retired NFL athletes in one survey reported a history of ACL injury.16
The most common high-risk positions are running backs and linebackers. Brophy and colleagues17 found that 9.7% of running backs and 8.9% of linebackers participating in the NFL Combine had a history of ACL injury. This may be because both the running back and linebacker are involved in frequent high-energy collisions and often quickly change direction. Other studies have also identified running backs and linebackers as high risk, in addition to tight ends, wide receivers, and interior linemen.13,15,18
Treatment of choice for elite level athletes with ACL injury is reconstruction.19 Of those who undergo ACL reconstruction, the rate of return to play ranges from 63% to 80%.20-22 The average time to return to play is 9 to 13 months. The odds of making a successful return hinges on how successful the athlete was prior to injury. Factors such as prior game experience, position on depth chart, being on scholarship, and draft position for NFL athletes have all been shown to have a positive predictive value on a patient’s chance of returning from ACL reconstruction.20,21
Players who return have variable levels of success afterwards. In a study of NFL quarterbacks who sustained ACL injuries, 12 out of 13 were able to return to game action with no appreciable dropoff in performance based on in-game production.23 Carey and colleagues24 looked specifically at NFL wide receivers and running backs and found an 80% return to play rate but with an approximate decrease in production of one-third upon return. Furthermore, in the Multicenter Orthopaedic Outcomes Network (MOON) cohort study, only 43% of participants felt they returned to their preoperative level.22
Medial Collateral Ligament
The MCL consists of superficial and deep components. The superficial MCL is the primary restraint to valgus laxity at the knee. The superficial MCL has 1 femoral and 2 tibial attachments. The deep MCL is a thickening of the medial joint capsule and runs deep and parallel to the superficial MCL. The amount of medial joint gapping with a valgus force on examination is used to grade severity of MCL injuries. Grade I is a <5-mm opening; Grade II, 5- to 10-mm opening; and grade III, >10-mm opening.
The MCL is the most common knee injury in high school, collegiate, and professional football.1,18,25-28 Injuries are typically due to contact when a valgus force is applied to the knee.29 The annual incidence of MCL injuries amongst high school football players is 24.2 per 100,000 AE.1 The positions that appear to be at greatest risk for MCL injuries are offensive and defensive linemen.18,30-32 In a review of 5047 collegiate athletes participating in the NFL Combine from 1987 to 2000, 23% of offensive linemen had a history of MCL injury, compared to the overall rate of 16%.33 In a similar study, Bradley and colleagues18 performed medical histories on athletes invited to the 2005 NFL Combine and also found offensive linemen had the highest rate of MCL injury at 33%, compared to the overall rate of 22%. They reasonably hypothesized that “chop blocks” and other players “rolling up” on the outside of linemen’s knees were responsible for these injuries. Albright and colleagues32 found that prophylactic knee braces decreased the incidence of MCL injuries in collegiate offensive lineman. However, additional studies have not been able to reproduce these results and the use of prophylactic knee braces remains controversial.26
Treatment of MCL injuries depends upon the grade of injury, associated injuries, and anatomical location of injury. Management of MCL injuries is for the most part nonsurgical. In 1974, Ellsasser and colleagues34 were the first to publish data on nonoperative management of Grade I and Grade II injuries with immediate motion and rehabilitation instead of cast immobilization. They found 93% of patients returned to football in 3 to 8 weeks.34 Derscheid and Garrick27 observed nonoperative treatment of Grade I and II sprains in collegiate football players, with a time loss of 10.6 days and 19.5 days for Grade I and II injuries, respectively. Holden and colleagues35 evaluated nonoperative management of Grade I and II MCL injuries in collegiate football players and found an average return to play of 21 days.
Grade III injury treatment is more controversial. Indelicato and colleagues36 demonstrated successful nonoperative management of Grade III MCL injuries in collegiate football players, with an average return to play of 64.4 days. Jones and colleagues37 had similar success with high school football players, with an average return to play of 34 days. However, isolated Grade III injuries are rare and therefore treatment is likely to be dictated by concomitant injuries. Fetto and Marshall38 found that 78% of Grade III injuries were associated with an additional ligamentous injury. Of those additional injuries, 95% were ACL tears.
Finally, one must consider the location of the MCL injury. Injuries of the distal MCL at its tibial insertion may result in poor healing, as the ligament is displaced away from its insertion. Therefore, some authors recommend surgical management for these injuries.39,40
The patellofemoral joint is a complex structure in which the patella is stabilized within the trochlear groove of the femur by both bony and soft tissue structures. The MPFL is one of the most important soft tissue stabilizers. The MPFL is the primary restraint to lateral patellar translation within the first 20° of knee flexion, contributing to 60% of the total restraining force.41 The MPFL originates on the medial femoral condyle and inserts on the superomedial aspect of the patella.
Patellar instability is the subluxation or dislocation of the patella out of the trochlear groove. Patellar subluxation and dislocation account for approximately 3% of all knee injuries.42 Patella dislocations are more common in younger populations43-45 with the majority (52%-63%) occurring during sports.43,44,46 Mitchell and colleagues47 reported an incidence of 4.1 patellar subluxations/dislocations per 100,000 AE in high school football players.
Dislocation is most commonly the result of knee flexion with the tibia in a valgus position.44,48 The majority of patellar dislocations occur via a noncontact mechanism.44,48 However, the majority of these injuries in football are from contact (63%).47
Acute patellar dislocations are associated with more soft tissue damage than those with recurrent dislocations.46 In acute patella dislocations, the MPFL is almost always ruptured.44 In contrast, Fithian and colleagues46 found only 38% of recurrent dislocators had MPFL injury. As a result, it is thought that those with recurrent instability dislocate without trauma and do not have the same characteristics as those who dislocate from high-energy trauma in sport. Risk factors for atraumatic dislocation are numerous and have been well described in the literature.49 However, traumatic dislocators usually do not have risk factors.50
Traumatic patella dislocations are higher energy and are associated with chondral injury in up to 95%of cases 51 and osteochondral injury 58% to 76% of the time.52,53 In contrast, people with “articular hypermobility” are less likely to sustain articular damage.54 This concept is important when considering risk for recurrent patella dislocation. The literature reports a 17% to 50% rate of recurrent instability after acute patella dislocation.46,55,56 However, most studies do not distinguish between traumatic and atraumatic injuries. Because the majority of patellar dislocations in football occur through contact mechanisms, the rate of recurrent instability in these athletes may in fact be less than what is reported in the literature.
First-time patella dislocations are generally treated nonoperatively. Mitchell and colleagues47 reported that 72.6% of high school athletes with patella subluxation treated conservatively were able to return to sports within 3 weeks, compared to only 34.1% of those with patellar dislocations. In the same study, patellar dislocations were season-ending 37% of the time.47 Atkin and colleagues50 followed 74 patients treated conservatively for first-time patellar dislocation and noted 58% at 6 months still had difficulty in squatting, jumping, or cutting.
Those who have failed conservative management and have an additional dislocation are 7 times more likely to redislocate.46 Therefore, they are usually treated operatively with MPFL reconstruction. Return to sport ranges from 3 to 6 months,57 with 53% to 77.3% reporting return to their previous functionality.57-59 Overall, 84.1% of patients are able to return to sport with 1.2% risk of recurrent dislocation.60
Posterior Cruciate Ligament
The PCL is the primary posterior stabilizer of the knee.61,62 It consists of the anterolateral and posteromedial bundles, named by their insertion on the posterior tibial plateau. The larger, stronger anterolateral bundle is the primary restraint to posterior tibial translation.63
Due to the relative infrequency of PCL injuries, there is a paucity of epidemiological data on sports-related PCL injuries. These injuries in the literature are commonly found due to traffic accidents (45%-57%) or from sports (33%-40%).64,65 According to Swensen and colleagues,1 PCL injuries account for 2.4% of all high school sport knee injuries. In a cohort of 62 knees with PCL injuries, Patel and colleagues66 found football was the most common cause of injury (19.3%).
The most common mechanism of injury in athletes is knee hyperflexion or a direct blow to the tibia in a flexed knee.67 In football, contact mechanisms are the most common. In a 16-year review of the National Collegiate Athletic Association (NCAA) injury surveillance system, the incidence of contact PCL injuries during games were 7.3 times higher than noncontact.68 The most common activity was being tackled, which accounted for 22.9% of all PCL injuries.68
Due to the high energy of these injuries, isolated PCL injuries are rare. In one trauma center’s experience, 96.5% of PCL injuries had an additional ligament injury.64 In that study, injuries to the PCL were associated with posterolateral corner, ACL, and MCL injuries 62%, 46%, and 31% of the time, respectively.64,69
Because isolated PCL injuries are rare, clinicians must rely on a thorough history and physical examination when evaluating athletes with knee injuries. Classification of PCL injuries is based on the amount of posterior tibial translation in relation to the femur with the knee bent to 90°. Grade I is 1 to 5 mm; Grade II, 6 to 10 mm; and Grade III, >10 mm. If there is suspicion of a PCL injury, there should be a very low threshold for magnetic resonance imaging, given the high association with additional injuries.
Natural history of Grade I and II isolated PCL injuries is generally favorable compared to Grade III and multi-ligamentous injuries.70 As a result, isolated Grade I and II PCL injuries are generally treated nonoperatively. Treatment consists of physical therapy with emphasis on quadriceps strengthening. Return to play can be considered as early as 2 to 4 weeks from injury.71 Recent long-term data have shown successful conservative management of Grade I and II injuries with quadriceps strength to 97% of contralateral leg and full range of motion.72 However, there was 11% moderate to severe osteoarthritis in these patients at a mean follow-up of 14.3 years.72 Fowler and Messieh67 managed athletes with 7 isolated complete PCL tears and 5 partial tears nonoperatively, all of whom were able to return to sport without limitation. Parolie and Bergfeld73 managed 25 athletes with isolated PCL tears conservatively. In this study, 80% of athletes reported satisfaction and 68% returned to previous level of play.73 Neither of the aforementioned studies specify the grades of the injuries. Finally, Patel and colleagues66 managed 6 NFL athletes with Grade I and II injuries nonoperatively, and all were able to return to sport.
Treatment of isolated Grade III PCL injuries is more controversial, and no consensus exists in the literature. In an epidemiological study, Dick and colleagues68 found that only 39% of NCAA football athletes underwent surgery for their torn PCLs, compared to 79% of ACL injuries. However, their study makes no mention to the severity of these injuries. Numerous options exist for PCL reconstruction, with no consensus on the preferred method.
Conclusion
Knee injuries are the most common injury in football. Knowledge of the natural history of these injuries, as well as treatment options and expected outcomes, will help treating physicians educate their patients on the optimal treatment and manage return to play expectations.
Am J Orthop. 2016;45(6):368-373. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
Football is one of the most popular sports in the United States. Every year more than 1 million high school males and over 60,000 collegiate males participate in organized football. The number of males who play football is greater than the combined number of males and females who participate in track and field or basketball.1 Football has the highest injury rate amongst popular American sports.2 From 2001 to 2005, there was an estimated 1.1 million emergency room visits as a direct result of football.3 Injuries are more likely to occur during games,1,2,4,5 more likely to require surgery,4 and more likely to end the player’s season or career when compared to other sports.6 Of those injuries that end seasons or careers, the knee is the most common culprit.6 This is of particular concern because knee injuries are most common in football.1,2,5,7 This article reviews the epidemiology of 4 of the most common knee injuries in American football: tears of the anterior cruciate ligament (ACL), medial collateral ligament (MCL), medial patellofemoral ligament (MPFL), and posterior cruciate ligament (PCL).
Anterior Cruciate Ligament
The ACL is the primary structure preventing anterior tibial translation. It is composed of 2 anatomic bundles: the anteromedial and posterolateral bundles. The ACL originates from the posteromedial portion of the lateral femoral condyle and inserts between and slightly anterior to the tibial intercondylar eminence. The bundles are named for their relative insertions onto the tibia.
Injury to the ACL occurs both through noncontact and contact mechanisms. Typical noncontact mechanism is a forceful valgus collapse with the knee close to full extension with combined external or internal rotation of the tibia.8 This is often the result of a sudden deceleration prior to a change in direction.9 Contact injuries to the ACL are the result of a direct blow to the knee causing valgus collapse.9 The majority of ACL injuries amongst all sports are a result of a noncontact mechanism. However, Dragoo and colleagues10 found the majority of football ACL injuries (55%-60%) were from contact. As a result, football players are 4 times more likely to sustain ACL injuries than in other sports.11
ACL injuries are associated with significant time loss from sport. At the high school level, they are the most likely injury to end a season or career.6 Because these are higher-energy injuries, they are frequently associated with damage to additional structures. ACL injuries that occur in football are associated with increased rates of meniscus, chondral, and multi-ligamentous injuries.12,13
The incidence of ACL injuries increases with level of competition. In high school athletes it is 11.1 per 100,000 athlete exposures (AE).1,11 In collegiate football, the rate increases to 14.2 to 18 per 100,000 AE.2,14 Though no incidence data per AE was found in our review of the literature, there were 219 ACL injuries in the National Football League (NFL) from 2010 to 2013.15 In addition, 14.2% of retired NFL athletes in one survey reported a history of ACL injury.16
The most common high-risk positions are running backs and linebackers. Brophy and colleagues17 found that 9.7% of running backs and 8.9% of linebackers participating in the NFL Combine had a history of ACL injury. This may be because both the running back and linebacker are involved in frequent high-energy collisions and often quickly change direction. Other studies have also identified running backs and linebackers as high risk, in addition to tight ends, wide receivers, and interior linemen.13,15,18
Treatment of choice for elite level athletes with ACL injury is reconstruction.19 Of those who undergo ACL reconstruction, the rate of return to play ranges from 63% to 80%.20-22 The average time to return to play is 9 to 13 months. The odds of making a successful return hinges on how successful the athlete was prior to injury. Factors such as prior game experience, position on depth chart, being on scholarship, and draft position for NFL athletes have all been shown to have a positive predictive value on a patient’s chance of returning from ACL reconstruction.20,21
Players who return have variable levels of success afterwards. In a study of NFL quarterbacks who sustained ACL injuries, 12 out of 13 were able to return to game action with no appreciable dropoff in performance based on in-game production.23 Carey and colleagues24 looked specifically at NFL wide receivers and running backs and found an 80% return to play rate but with an approximate decrease in production of one-third upon return. Furthermore, in the Multicenter Orthopaedic Outcomes Network (MOON) cohort study, only 43% of participants felt they returned to their preoperative level.22
Medial Collateral Ligament
The MCL consists of superficial and deep components. The superficial MCL is the primary restraint to valgus laxity at the knee. The superficial MCL has 1 femoral and 2 tibial attachments. The deep MCL is a thickening of the medial joint capsule and runs deep and parallel to the superficial MCL. The amount of medial joint gapping with a valgus force on examination is used to grade severity of MCL injuries. Grade I is a <5-mm opening; Grade II, 5- to 10-mm opening; and grade III, >10-mm opening.
The MCL is the most common knee injury in high school, collegiate, and professional football.1,18,25-28 Injuries are typically due to contact when a valgus force is applied to the knee.29 The annual incidence of MCL injuries amongst high school football players is 24.2 per 100,000 AE.1 The positions that appear to be at greatest risk for MCL injuries are offensive and defensive linemen.18,30-32 In a review of 5047 collegiate athletes participating in the NFL Combine from 1987 to 2000, 23% of offensive linemen had a history of MCL injury, compared to the overall rate of 16%.33 In a similar study, Bradley and colleagues18 performed medical histories on athletes invited to the 2005 NFL Combine and also found offensive linemen had the highest rate of MCL injury at 33%, compared to the overall rate of 22%. They reasonably hypothesized that “chop blocks” and other players “rolling up” on the outside of linemen’s knees were responsible for these injuries. Albright and colleagues32 found that prophylactic knee braces decreased the incidence of MCL injuries in collegiate offensive lineman. However, additional studies have not been able to reproduce these results and the use of prophylactic knee braces remains controversial.26
Treatment of MCL injuries depends upon the grade of injury, associated injuries, and anatomical location of injury. Management of MCL injuries is for the most part nonsurgical. In 1974, Ellsasser and colleagues34 were the first to publish data on nonoperative management of Grade I and Grade II injuries with immediate motion and rehabilitation instead of cast immobilization. They found 93% of patients returned to football in 3 to 8 weeks.34 Derscheid and Garrick27 observed nonoperative treatment of Grade I and II sprains in collegiate football players, with a time loss of 10.6 days and 19.5 days for Grade I and II injuries, respectively. Holden and colleagues35 evaluated nonoperative management of Grade I and II MCL injuries in collegiate football players and found an average return to play of 21 days.
Grade III injury treatment is more controversial. Indelicato and colleagues36 demonstrated successful nonoperative management of Grade III MCL injuries in collegiate football players, with an average return to play of 64.4 days. Jones and colleagues37 had similar success with high school football players, with an average return to play of 34 days. However, isolated Grade III injuries are rare and therefore treatment is likely to be dictated by concomitant injuries. Fetto and Marshall38 found that 78% of Grade III injuries were associated with an additional ligamentous injury. Of those additional injuries, 95% were ACL tears.
Finally, one must consider the location of the MCL injury. Injuries of the distal MCL at its tibial insertion may result in poor healing, as the ligament is displaced away from its insertion. Therefore, some authors recommend surgical management for these injuries.39,40
The patellofemoral joint is a complex structure in which the patella is stabilized within the trochlear groove of the femur by both bony and soft tissue structures. The MPFL is one of the most important soft tissue stabilizers. The MPFL is the primary restraint to lateral patellar translation within the first 20° of knee flexion, contributing to 60% of the total restraining force.41 The MPFL originates on the medial femoral condyle and inserts on the superomedial aspect of the patella.
Patellar instability is the subluxation or dislocation of the patella out of the trochlear groove. Patellar subluxation and dislocation account for approximately 3% of all knee injuries.42 Patella dislocations are more common in younger populations43-45 with the majority (52%-63%) occurring during sports.43,44,46 Mitchell and colleagues47 reported an incidence of 4.1 patellar subluxations/dislocations per 100,000 AE in high school football players.
Dislocation is most commonly the result of knee flexion with the tibia in a valgus position.44,48 The majority of patellar dislocations occur via a noncontact mechanism.44,48 However, the majority of these injuries in football are from contact (63%).47
Acute patellar dislocations are associated with more soft tissue damage than those with recurrent dislocations.46 In acute patella dislocations, the MPFL is almost always ruptured.44 In contrast, Fithian and colleagues46 found only 38% of recurrent dislocators had MPFL injury. As a result, it is thought that those with recurrent instability dislocate without trauma and do not have the same characteristics as those who dislocate from high-energy trauma in sport. Risk factors for atraumatic dislocation are numerous and have been well described in the literature.49 However, traumatic dislocators usually do not have risk factors.50
Traumatic patella dislocations are higher energy and are associated with chondral injury in up to 95%of cases 51 and osteochondral injury 58% to 76% of the time.52,53 In contrast, people with “articular hypermobility” are less likely to sustain articular damage.54 This concept is important when considering risk for recurrent patella dislocation. The literature reports a 17% to 50% rate of recurrent instability after acute patella dislocation.46,55,56 However, most studies do not distinguish between traumatic and atraumatic injuries. Because the majority of patellar dislocations in football occur through contact mechanisms, the rate of recurrent instability in these athletes may in fact be less than what is reported in the literature.
First-time patella dislocations are generally treated nonoperatively. Mitchell and colleagues47 reported that 72.6% of high school athletes with patella subluxation treated conservatively were able to return to sports within 3 weeks, compared to only 34.1% of those with patellar dislocations. In the same study, patellar dislocations were season-ending 37% of the time.47 Atkin and colleagues50 followed 74 patients treated conservatively for first-time patellar dislocation and noted 58% at 6 months still had difficulty in squatting, jumping, or cutting.
Those who have failed conservative management and have an additional dislocation are 7 times more likely to redislocate.46 Therefore, they are usually treated operatively with MPFL reconstruction. Return to sport ranges from 3 to 6 months,57 with 53% to 77.3% reporting return to their previous functionality.57-59 Overall, 84.1% of patients are able to return to sport with 1.2% risk of recurrent dislocation.60
Posterior Cruciate Ligament
The PCL is the primary posterior stabilizer of the knee.61,62 It consists of the anterolateral and posteromedial bundles, named by their insertion on the posterior tibial plateau. The larger, stronger anterolateral bundle is the primary restraint to posterior tibial translation.63
Due to the relative infrequency of PCL injuries, there is a paucity of epidemiological data on sports-related PCL injuries. These injuries in the literature are commonly found due to traffic accidents (45%-57%) or from sports (33%-40%).64,65 According to Swensen and colleagues,1 PCL injuries account for 2.4% of all high school sport knee injuries. In a cohort of 62 knees with PCL injuries, Patel and colleagues66 found football was the most common cause of injury (19.3%).
The most common mechanism of injury in athletes is knee hyperflexion or a direct blow to the tibia in a flexed knee.67 In football, contact mechanisms are the most common. In a 16-year review of the National Collegiate Athletic Association (NCAA) injury surveillance system, the incidence of contact PCL injuries during games were 7.3 times higher than noncontact.68 The most common activity was being tackled, which accounted for 22.9% of all PCL injuries.68
Due to the high energy of these injuries, isolated PCL injuries are rare. In one trauma center’s experience, 96.5% of PCL injuries had an additional ligament injury.64 In that study, injuries to the PCL were associated with posterolateral corner, ACL, and MCL injuries 62%, 46%, and 31% of the time, respectively.64,69
Because isolated PCL injuries are rare, clinicians must rely on a thorough history and physical examination when evaluating athletes with knee injuries. Classification of PCL injuries is based on the amount of posterior tibial translation in relation to the femur with the knee bent to 90°. Grade I is 1 to 5 mm; Grade II, 6 to 10 mm; and Grade III, >10 mm. If there is suspicion of a PCL injury, there should be a very low threshold for magnetic resonance imaging, given the high association with additional injuries.
Natural history of Grade I and II isolated PCL injuries is generally favorable compared to Grade III and multi-ligamentous injuries.70 As a result, isolated Grade I and II PCL injuries are generally treated nonoperatively. Treatment consists of physical therapy with emphasis on quadriceps strengthening. Return to play can be considered as early as 2 to 4 weeks from injury.71 Recent long-term data have shown successful conservative management of Grade I and II injuries with quadriceps strength to 97% of contralateral leg and full range of motion.72 However, there was 11% moderate to severe osteoarthritis in these patients at a mean follow-up of 14.3 years.72 Fowler and Messieh67 managed athletes with 7 isolated complete PCL tears and 5 partial tears nonoperatively, all of whom were able to return to sport without limitation. Parolie and Bergfeld73 managed 25 athletes with isolated PCL tears conservatively. In this study, 80% of athletes reported satisfaction and 68% returned to previous level of play.73 Neither of the aforementioned studies specify the grades of the injuries. Finally, Patel and colleagues66 managed 6 NFL athletes with Grade I and II injuries nonoperatively, and all were able to return to sport.
Treatment of isolated Grade III PCL injuries is more controversial, and no consensus exists in the literature. In an epidemiological study, Dick and colleagues68 found that only 39% of NCAA football athletes underwent surgery for their torn PCLs, compared to 79% of ACL injuries. However, their study makes no mention to the severity of these injuries. Numerous options exist for PCL reconstruction, with no consensus on the preferred method.
Conclusion
Knee injuries are the most common injury in football. Knowledge of the natural history of these injuries, as well as treatment options and expected outcomes, will help treating physicians educate their patients on the optimal treatment and manage return to play expectations.
Am J Orthop. 2016;45(6):368-373. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Swenson DM, Collins CL, Best TM, Flanigan DC, Fields SK, Comstock RD. Epidemiology of knee injuries among U.S. high school athletes, 2005/2006-2010/2011. Med Sci Sports Exerc. 2013;45(3):462-469.
2. Hootman JM, Dick R, Agel J. Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J Athl Train. 2007;42(2):311-319.
3. Mello MJ, Myers R, Christian JB, Palmisciano L, Linakis JG. Injuries in youth football: national emergency department visits during 2001-2005 for young and adolescent players. Acad Emerg Med. 2009;16(3):243-248.
4. Rechel JA, Collins CL, Comstock RD. Epidemiology of injuries requiring surgery among high school athletes in the United States, 2005 to 2010. J Trauma. 2011;71(4):982-989.
5. Ingram JG, Fields SK, Yard EE, Comstock RD. Epidemiology of knee injuries among boys and girls in US high school athletics. Am J Sports Med. 2008;36(6):1116-1122.
6. Tirabassi J, Brou L, Khodaee M, Lefort R, Fields SK, Comstock RD. Epidemiology of high school sports-related injuries resulting in medical disqualification: 2005-2006 through 2013-2014 academic years. Am J Sports Med. 2016 May 10. [Epub ahead of print]
7. Fernandez WG, Yard EE, Comstock RD. Epidemiology of lower extremity injuries among U.S. high school athletes. Acad Emerg Med. 2007;14(7):641-645.
8. Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. Am J Sports Med. 2004;32(4):1002-1012.
9. Boden BP, Dean GS, Feagin JA Jr, Garrett WE Jr. Mechanisms of anterior cruciate ligament injury. Orthopedics. 2000;23(6):573-578.
10. Dragoo JL, Braun HJ, Harris AH. The effect of playing surface on the incidence of ACL injuries in National Collegiate Athletic Association American Football. Knee. 2013;20(3):191-195.
11. Joseph AM, Collins CL, Henke NM, Yard EE, Fields SK, Comstock RD. A multisport epidemiologic comparison of anterior cruciate ligament injuries in high school athletics. J Athl Train. 2013;48(6):810-817.
12. Granan LP, Inacio MC, Maletis GB, Funahashi TT, Engebretsen L. Sport-specific injury pattern recorded during anterior cruciate ligament reconstruction. Am J Sports Med. 2013;41(12):2814-2818.
13. Bradley JP, Klimkiewicz JJ, Rytel MJ, Powell JW. Anterior cruciate ligament injuries in the National Football League: epidemiology and current treatment trends among team physicians. Arthroscopy. 2002;18(5):502-509.
14. Dragoo JL, Braun HJ, Durham JL, Chen MR, Harris AH. Incidence and risk factors for injuries to the anterior cruciate ligament in National Collegiate Athletic Association football: data from the 2004-2005 through 2008-2009 National Collegiate Athletic Association Injury Surveillance System. Am J Sports Med. 2012;40(5):990-995.
15. Dodson CC, Secrist ES, Bhat SB, Woods DP, Deluca PF. Anterior cruciate ligamenti in National Football League athletes from 2010 to 2013: a descriptive epidemiology study. Orthop J Sports Med. 2016;4(3):2325967116631949.
16. Golightly YM, Marshall SW, Callahan LF, Guskiewicz K. Early-onset arthritis in retired National Football League players. J Phys Act Health. 2009;6(5):638-643.
17. Brophy RH, Lyman S, Chehab EL, Barnes RP, Rodeo SA, Warren RF. Predictive value of prior injury on career in professional American football is affected by player position. Am J Sports Med. 2009;37(4):768-775.
18. Bradley J, Honkamp NJ, Jost P, West R, Norwig J, Kaplan LD. Incidence and variance of knee injuries in elite college football players. Am J Orthop. 2008;37(6):310-314.
19. Erickson BJ, Harris JD, Fillingham YA, et al. Anterior cruciate ligament reconstruction practice patterns by NFL and NCAA football team physicians. Arthroscopy. 2014;30(6):731-738.
20. Daruawalla JH, Greis PE, Hancock R; ASP Collaborative Group, Xerogeanes JW. Rates and determinants of return to play after anterior cruciate ligament reconstruction in NCAA Division 1 college football athletes: a study of the ACC, SEC, and PAC-12 conferences. Orthop J Sports Med. 2014;2(8):2325967114543901.
21. Shah VM, Andrews JR, Fleisig GS, McMichael CS, Lemak LJ. Return to play after anterior cruciate ligament reconstruction in National Football League athletes. Am J Sports Med. 2010;38(11):2233-2239.
22. McCullough KA, Phelps KD, Spindler KP, et al. Return to high school- and college-level football after anterior cruciate ligament reconstruction: a Multicenter Orthopaedic Outcomes Network (MOON) cohort study. Am J Sports Med. 2012;40(11):2523-2529.
23. Erickson BJ, Harris JD, Heninger JR, et al. Performance and return-to-sport after ACL reconstruction in NFL quarterbacks. Orthopedics. 2014;37(8):e728-e734.
24. Carey JL, Huffman GR, Parekh SG, Sennett BJ. Outcomes of anterior cruciate ligament injuries to running backs and wide receivers in the National Football League. Am J Sports Med. 2006;34(12):1911-1917.
25. Hershman EB, Anderson R, Bergfeld JA, et al. An analysis of specific lower extremity injury rates on grass and FieldTurf playing surfaces in National Football League Games: 2000-2009 seasons. Am J Sports Med. 2012;40(10):2200-2205.
26. Salata MJ, Gibbs AE, Sekiya JK. The effectiveness of prophylactic knee bracing in American football: a systematic review. Sports Health. 2010;2(5):375-379.
27. Derscheid GL, Garrick JG. Medial collateral ligament injuries in football. Nonoperative management of grade I and grade II sprains. Am J Sports Med. 1981;9(6):365-368.
28. Meyers MC, Barnhill BS. Incidence, causes, and severity of high school football injuries on FieldTurf versus natural grass: a 5-year prospective study. Am J Sports Med. 2004;32(7):1626-1638.
29. Lundblad M, Waldén M, Magnusson H, Karlsson J, Ekstrand J. The UEFA injury study: 11-year data concerning 346 MCL injuries and time to return to play. Br J Sports Med. 2013;47(12):759-762.
30. Hewson GF Jr, Mendini RA, Wang JB. Prophylactic knee bracing in college football. Am J Sports Med. 1986;14(4):262-266.
31. Rovere GD, Haupt HA, Yates CS. Prophylactic knee bracing in college football. Am J Sports Med. 1987;15(2):111-116.
32. Albright JP, Powell JW, Smith W, et al. Medial collateral ligament knee sprains in college football. Brace wear preferences and injury risk. Am J Sports Med. 1994;22(1):2-11.
33. Brophy RH, Barnes R, Rodeo SA, Warren RF. Prevalence of musculoskeletal disorders at the NFL Combine--trends from 1987 to 2000. Med Sci Sports Exerc. 2007;39(1):22-27.
34. Ellsasser JC, Reynolds FC, Omohundro JR. The non-operative treatment of collateral ligament injuries of the knee in professional football players. An analysis of seventy-four injuries treated non-operatively and twenty-four injuries treated surgically. J Bone Joint Surg Am. 1974;56(6):1185-1190.
35. Holden DL, Eggert AW, Butler JE. The nonoperative treatment of grade I and II medial collateral ligament injuries to the knee. Am J Sports Med. 1983;11(5):340-344.
36. Indelicato PA, Hermansdorfer J, Huegel M. Nonoperative management of complete tears of the medial collateral ligament of the knee in intercollegiate football players. Clin Orthop Relat Res. 1990;(256):174-177.
37. Jones RE, Henley MB, Francis P. Nonoperative management of isolated grade III collateral ligament injury in high school football players. Clin Orthop Relat Res. 1986;(213):137-140.
38. Fetto JF, Marshall JL. Medial collateral ligament injuries of the knee: a rationale for treatment. Clin Orthop Relat Res. 1978;(132):206-218.
39. Corten K, Hoser C, Fink C, Bellemans J. Case reports: a Stener-like lesion of the medial collateral ligament of the knee. Clin Orthop Relat Res. 2010;468(1):289-293.
40. Marchant MH Jr, Tibor LM, Sekiya JK, Hardaker WT Jr, Garrett WE Jr, Taylor DC. Management of medial-sided knee injuries, part 1: medial collateral ligament. Am J Sports Med. 2011;39(5):1102-1113.
41. Desio SM, Burks RT, Bachus KN. Soft tissue restraints to lateral patellar translation in the human knee. Am J Sports Med. 1998;26(1):59-65.
42. Casteleyn PP, Handelberg F. Arthroscopy in the diagnosis of occult dislocation of the patella. Acta Orthop Belg. 1989;55(3):381-383.
43. Waterman BR, Belmont PJ Jr, Owens BD. Patellar dislocation in the United States: role of sex, age, race, and athletic participation. J Knee Surg. 2012;25(1):51-57.
44. Sillanpää P, Mattila VM, Iivonen T, Visuri T, Pihlajamäki H. Incidence and risk factors of acute traumatic primary patellar dislocation. Med Sci Sports Exerc. 2008;40(4):606-611.
45. Hsiao M, Owens BD, Burks R, Sturdivant RX, Cameron KL. Incidence of acute traumatic patellar dislocation among active-duty United States military service members. Am J Sports Med. 2010;38(10):1997-2004.
46. Fithian DC, Paxton EW, Stone ML, et al. Epidemiology and natural history of acute patellar dislocation. Am J Sports Med. 2004;32(5):1114-1121.
47. Mitchell J, Magnussen RA, Collins CL, et al. Epidemiology of patellofemoral instability injuries among high school athletes in the United States. Am J Sports Med. 2015;43(7):1676-1682.
48. Nikku R, Nietosvaara Y, Aalto K, Kallio PE. The mechanism of primary patellar dislocation: trauma history of 126 patients. Acta Orthop. 2009;80(4):432-434.
49. Tsai CH, Hsu CJ, Hung CH, Hsu HC. Primary traumatic patellar dislocation. J Orthop Surg Res. 2012;7:21.
50. Atkin DM, Fithian DC, Marangi KS, Stone ML, Dobson BE, Mendelsohn C. Characteristics of patients with primary acute lateral patellar dislocation and their recovery within the first 6 months of injury. Am J Sports Med. 2000;28(4):472-479.
51. Nomura E, Inoue M, Kurimura M. Chondral and osteochondral injuries associated with acute patellar dislocation. Arthroscopy. 2003;19(7):717-721.
52. Kirsch MD, Fitzgerald SW, Friedman H, Rogers LF. Transient lateral patellar dislocation: diagnosis with MR imaging. AJR Am J Roentgenol. 1993;161(1):109-113.
53. Virolainen H, Visuri T, Kuusela T. Acute dislocation of the patella: MR findings. Radiology. 1993;189(1):243-246.
54. Stanitski CL. Articular hypermobility and chondral injury in patients with acute patellar dislocation. Am J Sports Med. 1995;23(2):146-150.
55. Mäenpää H, Huhtala H, Lehto MU. Recurrence after patellar dislocation. Redislocation in 37/75 patients followed for 6-24 years. Acta Orthop Scand. 1997;68(5):424-426.
56. Buchner M, Baudendistel B, Sabo D, Schmitt H. Acute traumatic primary patellar dislocation: long-term results comparing conservative and surgical treatment. Clin J Sport Med. 2005;15(2):62-66.
57. Fisher B, Nyland J, Brand E, Curtin B. Medial patellofemoral ligament reconstruction for recurrent patellar dislocation: a systematic review including rehabilitation and return-to-sports efficacy. Arthroscopy. 2010;26(10):1384-1394.
58. Lippacher S, Dreyhaupt J, Williams SR, Reichel H, Nelitz M. Reconstruction of the medial patellofemoral ligament: clinical outcomes and return to sports. Am J Sports Med. 2014;42(7):1661-1668.
59. Panni AS, Alam M, Cerciello S, Vasso M, Maffulli N. Medial patellofemoral ligament reconstruction with a divergent patellar transverse 2-tunnel technique. Am J Sports Med. 2011;39(12):2647-1655.
60. Schneider DK, Grawe B, Magnussen RA, et al. Outcomes after isolated medial patellofemoral ligament reconstruction for the treatment of recurrent lateral patellar dislocations: a systematic review and meta-analysis. Am J Sports Med. 2016 Feb 12. [Epub ahead of print]
61. Amis AA, Bull AM, Gupte CM, Hijazi I, Race A, Robinson JR. Biomechanics of the PCL and related structures: posterolateral, posteromedial and meniscofemoral ligaments. Knee Surg Sports Traumatol Arthrosc. 2003;11(5):271-281.
62. Fu FH, Harner CD, Johnson DL, Miller MD, Woo SL. Biomechanics of knee ligaments: basic concepts and clinical application. Instr Course Lect. 1994;43:137-148.
63. Markolf KL, Feeley BT, Tejwani SG, Martin DE, McAllister DR. Changes in knee laxity and ligament force after sectioning the posteromedial bundle of the posterior cruciate ligament. Arthroscopy. 2006; 22(10):1100-1106.
64. Ganelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526-529.
65. Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186-191.
66. Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT. The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J. 2007;3(2):137-146.
67. Fowler PJ, Messieh SS. Isolated posterior cruciate ligament injuries in athletes. Am J Sports Med. 1987;15(6):553-557.
68. Dick R, Ferrara MS, Agel J, et al. Descriptive epidemiology of collegiate men’s football injuries: National Collegiate Athletic Association Injury Surveillance System, 1988-1989 through 2003-2004. J Athl Train. 2007;42(2):221-233.
69. LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077-3092.
70. Torg JS, Barton TM, Pavlov H, Stine R. Natural history of the posterior cruciate ligament-deficient knee. Clin Orthop Relat Res. 1989(246):208-216.
71. Miller MD. Orthopaedic Knowledge Update: Sports Medicine 5. Rosemont, IL; American Academy of Orthopaedic Surgeons; 2016.
72. Shelbourne KD, Clark M, Gray T. Minimum 10-year follow-up of patients after an acute, isolated posterior cruciate ligament injury treated nonoperatively. Am J Sports Med. 2013;41(7):1526-1533.
73. Parolie JM, Bergfeld JA. Long-term results of nonoperative treatment of isolated posterior cruciate ligament injuries in the athlete. Am J Sports Med. 1986;14(1):35-38.
1. Swenson DM, Collins CL, Best TM, Flanigan DC, Fields SK, Comstock RD. Epidemiology of knee injuries among U.S. high school athletes, 2005/2006-2010/2011. Med Sci Sports Exerc. 2013;45(3):462-469.
2. Hootman JM, Dick R, Agel J. Epidemiology of collegiate injuries for 15 sports: summary and recommendations for injury prevention initiatives. J Athl Train. 2007;42(2):311-319.
3. Mello MJ, Myers R, Christian JB, Palmisciano L, Linakis JG. Injuries in youth football: national emergency department visits during 2001-2005 for young and adolescent players. Acad Emerg Med. 2009;16(3):243-248.
4. Rechel JA, Collins CL, Comstock RD. Epidemiology of injuries requiring surgery among high school athletes in the United States, 2005 to 2010. J Trauma. 2011;71(4):982-989.
5. Ingram JG, Fields SK, Yard EE, Comstock RD. Epidemiology of knee injuries among boys and girls in US high school athletics. Am J Sports Med. 2008;36(6):1116-1122.
6. Tirabassi J, Brou L, Khodaee M, Lefort R, Fields SK, Comstock RD. Epidemiology of high school sports-related injuries resulting in medical disqualification: 2005-2006 through 2013-2014 academic years. Am J Sports Med. 2016 May 10. [Epub ahead of print]
7. Fernandez WG, Yard EE, Comstock RD. Epidemiology of lower extremity injuries among U.S. high school athletes. Acad Emerg Med. 2007;14(7):641-645.
8. Olsen OE, Myklebust G, Engebretsen L, Bahr R. Injury mechanisms for anterior cruciate ligament injuries in team handball: a systematic video analysis. Am J Sports Med. 2004;32(4):1002-1012.
9. Boden BP, Dean GS, Feagin JA Jr, Garrett WE Jr. Mechanisms of anterior cruciate ligament injury. Orthopedics. 2000;23(6):573-578.
10. Dragoo JL, Braun HJ, Harris AH. The effect of playing surface on the incidence of ACL injuries in National Collegiate Athletic Association American Football. Knee. 2013;20(3):191-195.
11. Joseph AM, Collins CL, Henke NM, Yard EE, Fields SK, Comstock RD. A multisport epidemiologic comparison of anterior cruciate ligament injuries in high school athletics. J Athl Train. 2013;48(6):810-817.
12. Granan LP, Inacio MC, Maletis GB, Funahashi TT, Engebretsen L. Sport-specific injury pattern recorded during anterior cruciate ligament reconstruction. Am J Sports Med. 2013;41(12):2814-2818.
13. Bradley JP, Klimkiewicz JJ, Rytel MJ, Powell JW. Anterior cruciate ligament injuries in the National Football League: epidemiology and current treatment trends among team physicians. Arthroscopy. 2002;18(5):502-509.
14. Dragoo JL, Braun HJ, Durham JL, Chen MR, Harris AH. Incidence and risk factors for injuries to the anterior cruciate ligament in National Collegiate Athletic Association football: data from the 2004-2005 through 2008-2009 National Collegiate Athletic Association Injury Surveillance System. Am J Sports Med. 2012;40(5):990-995.
15. Dodson CC, Secrist ES, Bhat SB, Woods DP, Deluca PF. Anterior cruciate ligamenti in National Football League athletes from 2010 to 2013: a descriptive epidemiology study. Orthop J Sports Med. 2016;4(3):2325967116631949.
16. Golightly YM, Marshall SW, Callahan LF, Guskiewicz K. Early-onset arthritis in retired National Football League players. J Phys Act Health. 2009;6(5):638-643.
17. Brophy RH, Lyman S, Chehab EL, Barnes RP, Rodeo SA, Warren RF. Predictive value of prior injury on career in professional American football is affected by player position. Am J Sports Med. 2009;37(4):768-775.
18. Bradley J, Honkamp NJ, Jost P, West R, Norwig J, Kaplan LD. Incidence and variance of knee injuries in elite college football players. Am J Orthop. 2008;37(6):310-314.
19. Erickson BJ, Harris JD, Fillingham YA, et al. Anterior cruciate ligament reconstruction practice patterns by NFL and NCAA football team physicians. Arthroscopy. 2014;30(6):731-738.
20. Daruawalla JH, Greis PE, Hancock R; ASP Collaborative Group, Xerogeanes JW. Rates and determinants of return to play after anterior cruciate ligament reconstruction in NCAA Division 1 college football athletes: a study of the ACC, SEC, and PAC-12 conferences. Orthop J Sports Med. 2014;2(8):2325967114543901.
21. Shah VM, Andrews JR, Fleisig GS, McMichael CS, Lemak LJ. Return to play after anterior cruciate ligament reconstruction in National Football League athletes. Am J Sports Med. 2010;38(11):2233-2239.
22. McCullough KA, Phelps KD, Spindler KP, et al. Return to high school- and college-level football after anterior cruciate ligament reconstruction: a Multicenter Orthopaedic Outcomes Network (MOON) cohort study. Am J Sports Med. 2012;40(11):2523-2529.
23. Erickson BJ, Harris JD, Heninger JR, et al. Performance and return-to-sport after ACL reconstruction in NFL quarterbacks. Orthopedics. 2014;37(8):e728-e734.
24. Carey JL, Huffman GR, Parekh SG, Sennett BJ. Outcomes of anterior cruciate ligament injuries to running backs and wide receivers in the National Football League. Am J Sports Med. 2006;34(12):1911-1917.
25. Hershman EB, Anderson R, Bergfeld JA, et al. An analysis of specific lower extremity injury rates on grass and FieldTurf playing surfaces in National Football League Games: 2000-2009 seasons. Am J Sports Med. 2012;40(10):2200-2205.
26. Salata MJ, Gibbs AE, Sekiya JK. The effectiveness of prophylactic knee bracing in American football: a systematic review. Sports Health. 2010;2(5):375-379.
27. Derscheid GL, Garrick JG. Medial collateral ligament injuries in football. Nonoperative management of grade I and grade II sprains. Am J Sports Med. 1981;9(6):365-368.
28. Meyers MC, Barnhill BS. Incidence, causes, and severity of high school football injuries on FieldTurf versus natural grass: a 5-year prospective study. Am J Sports Med. 2004;32(7):1626-1638.
29. Lundblad M, Waldén M, Magnusson H, Karlsson J, Ekstrand J. The UEFA injury study: 11-year data concerning 346 MCL injuries and time to return to play. Br J Sports Med. 2013;47(12):759-762.
30. Hewson GF Jr, Mendini RA, Wang JB. Prophylactic knee bracing in college football. Am J Sports Med. 1986;14(4):262-266.
31. Rovere GD, Haupt HA, Yates CS. Prophylactic knee bracing in college football. Am J Sports Med. 1987;15(2):111-116.
32. Albright JP, Powell JW, Smith W, et al. Medial collateral ligament knee sprains in college football. Brace wear preferences and injury risk. Am J Sports Med. 1994;22(1):2-11.
33. Brophy RH, Barnes R, Rodeo SA, Warren RF. Prevalence of musculoskeletal disorders at the NFL Combine--trends from 1987 to 2000. Med Sci Sports Exerc. 2007;39(1):22-27.
34. Ellsasser JC, Reynolds FC, Omohundro JR. The non-operative treatment of collateral ligament injuries of the knee in professional football players. An analysis of seventy-four injuries treated non-operatively and twenty-four injuries treated surgically. J Bone Joint Surg Am. 1974;56(6):1185-1190.
35. Holden DL, Eggert AW, Butler JE. The nonoperative treatment of grade I and II medial collateral ligament injuries to the knee. Am J Sports Med. 1983;11(5):340-344.
36. Indelicato PA, Hermansdorfer J, Huegel M. Nonoperative management of complete tears of the medial collateral ligament of the knee in intercollegiate football players. Clin Orthop Relat Res. 1990;(256):174-177.
37. Jones RE, Henley MB, Francis P. Nonoperative management of isolated grade III collateral ligament injury in high school football players. Clin Orthop Relat Res. 1986;(213):137-140.
38. Fetto JF, Marshall JL. Medial collateral ligament injuries of the knee: a rationale for treatment. Clin Orthop Relat Res. 1978;(132):206-218.
39. Corten K, Hoser C, Fink C, Bellemans J. Case reports: a Stener-like lesion of the medial collateral ligament of the knee. Clin Orthop Relat Res. 2010;468(1):289-293.
40. Marchant MH Jr, Tibor LM, Sekiya JK, Hardaker WT Jr, Garrett WE Jr, Taylor DC. Management of medial-sided knee injuries, part 1: medial collateral ligament. Am J Sports Med. 2011;39(5):1102-1113.
41. Desio SM, Burks RT, Bachus KN. Soft tissue restraints to lateral patellar translation in the human knee. Am J Sports Med. 1998;26(1):59-65.
42. Casteleyn PP, Handelberg F. Arthroscopy in the diagnosis of occult dislocation of the patella. Acta Orthop Belg. 1989;55(3):381-383.
43. Waterman BR, Belmont PJ Jr, Owens BD. Patellar dislocation in the United States: role of sex, age, race, and athletic participation. J Knee Surg. 2012;25(1):51-57.
44. Sillanpää P, Mattila VM, Iivonen T, Visuri T, Pihlajamäki H. Incidence and risk factors of acute traumatic primary patellar dislocation. Med Sci Sports Exerc. 2008;40(4):606-611.
45. Hsiao M, Owens BD, Burks R, Sturdivant RX, Cameron KL. Incidence of acute traumatic patellar dislocation among active-duty United States military service members. Am J Sports Med. 2010;38(10):1997-2004.
46. Fithian DC, Paxton EW, Stone ML, et al. Epidemiology and natural history of acute patellar dislocation. Am J Sports Med. 2004;32(5):1114-1121.
47. Mitchell J, Magnussen RA, Collins CL, et al. Epidemiology of patellofemoral instability injuries among high school athletes in the United States. Am J Sports Med. 2015;43(7):1676-1682.
48. Nikku R, Nietosvaara Y, Aalto K, Kallio PE. The mechanism of primary patellar dislocation: trauma history of 126 patients. Acta Orthop. 2009;80(4):432-434.
49. Tsai CH, Hsu CJ, Hung CH, Hsu HC. Primary traumatic patellar dislocation. J Orthop Surg Res. 2012;7:21.
50. Atkin DM, Fithian DC, Marangi KS, Stone ML, Dobson BE, Mendelsohn C. Characteristics of patients with primary acute lateral patellar dislocation and their recovery within the first 6 months of injury. Am J Sports Med. 2000;28(4):472-479.
51. Nomura E, Inoue M, Kurimura M. Chondral and osteochondral injuries associated with acute patellar dislocation. Arthroscopy. 2003;19(7):717-721.
52. Kirsch MD, Fitzgerald SW, Friedman H, Rogers LF. Transient lateral patellar dislocation: diagnosis with MR imaging. AJR Am J Roentgenol. 1993;161(1):109-113.
53. Virolainen H, Visuri T, Kuusela T. Acute dislocation of the patella: MR findings. Radiology. 1993;189(1):243-246.
54. Stanitski CL. Articular hypermobility and chondral injury in patients with acute patellar dislocation. Am J Sports Med. 1995;23(2):146-150.
55. Mäenpää H, Huhtala H, Lehto MU. Recurrence after patellar dislocation. Redislocation in 37/75 patients followed for 6-24 years. Acta Orthop Scand. 1997;68(5):424-426.
56. Buchner M, Baudendistel B, Sabo D, Schmitt H. Acute traumatic primary patellar dislocation: long-term results comparing conservative and surgical treatment. Clin J Sport Med. 2005;15(2):62-66.
57. Fisher B, Nyland J, Brand E, Curtin B. Medial patellofemoral ligament reconstruction for recurrent patellar dislocation: a systematic review including rehabilitation and return-to-sports efficacy. Arthroscopy. 2010;26(10):1384-1394.
58. Lippacher S, Dreyhaupt J, Williams SR, Reichel H, Nelitz M. Reconstruction of the medial patellofemoral ligament: clinical outcomes and return to sports. Am J Sports Med. 2014;42(7):1661-1668.
59. Panni AS, Alam M, Cerciello S, Vasso M, Maffulli N. Medial patellofemoral ligament reconstruction with a divergent patellar transverse 2-tunnel technique. Am J Sports Med. 2011;39(12):2647-1655.
60. Schneider DK, Grawe B, Magnussen RA, et al. Outcomes after isolated medial patellofemoral ligament reconstruction for the treatment of recurrent lateral patellar dislocations: a systematic review and meta-analysis. Am J Sports Med. 2016 Feb 12. [Epub ahead of print]
61. Amis AA, Bull AM, Gupte CM, Hijazi I, Race A, Robinson JR. Biomechanics of the PCL and related structures: posterolateral, posteromedial and meniscofemoral ligaments. Knee Surg Sports Traumatol Arthrosc. 2003;11(5):271-281.
62. Fu FH, Harner CD, Johnson DL, Miller MD, Woo SL. Biomechanics of knee ligaments: basic concepts and clinical application. Instr Course Lect. 1994;43:137-148.
63. Markolf KL, Feeley BT, Tejwani SG, Martin DE, McAllister DR. Changes in knee laxity and ligament force after sectioning the posteromedial bundle of the posterior cruciate ligament. Arthroscopy. 2006; 22(10):1100-1106.
64. Ganelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526-529.
65. Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186-191.
66. Patel DV, Allen AA, Warren RF, Wickiewicz TL, Simonian PT. The nonoperative treatment of acute, isolated (partial or complete) posterior cruciate ligament-deficient knees: an intermediate-term follow-up study. HSS J. 2007;3(2):137-146.
67. Fowler PJ, Messieh SS. Isolated posterior cruciate ligament injuries in athletes. Am J Sports Med. 1987;15(6):553-557.
68. Dick R, Ferrara MS, Agel J, et al. Descriptive epidemiology of collegiate men’s football injuries: National Collegiate Athletic Association Injury Surveillance System, 1988-1989 through 2003-2004. J Athl Train. 2007;42(2):221-233.
69. LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077-3092.
70. Torg JS, Barton TM, Pavlov H, Stine R. Natural history of the posterior cruciate ligament-deficient knee. Clin Orthop Relat Res. 1989(246):208-216.
71. Miller MD. Orthopaedic Knowledge Update: Sports Medicine 5. Rosemont, IL; American Academy of Orthopaedic Surgeons; 2016.
72. Shelbourne KD, Clark M, Gray T. Minimum 10-year follow-up of patients after an acute, isolated posterior cruciate ligament injury treated nonoperatively. Am J Sports Med. 2013;41(7):1526-1533.
73. Parolie JM, Bergfeld JA. Long-term results of nonoperative treatment of isolated posterior cruciate ligament injuries in the athlete. Am J Sports Med. 1986;14(1):35-38.
Foot and Ankle Injuries in American Football
Foot and ankle injuries are common in American football, with injury rates significantly increasing over the past decade.1-5 Epidemiologic studies of collegiate football players have shown an annual incidence of foot and ankle injuries ranging from 9% to 39%,3,6 with as many as 72% of all collegiate players presenting to the National Football League (NFL) Combine with a history of a foot or ankle injury and 13% undergoing surgical treatment.5 Player position influences the rate and type of foot and ankle injury. Offensive and “skill position” players, including linemen, running backs, and wide receivers, are particularly susceptible to foot and ankle injuries due to high levels of force and torque placed on the distal extremity during running, cutting, and tackling. Shoe wear changes, playing field conditions, increasing player size and speed, and improved reporting of injuries are also contributing to increasing injury rates.
The interaction between player cleats and the playing surface is a central issue of foot and ankle injuries in football. Improved traction relates to performance, but increased subsequent torque on the lower extremity is associated with injury. While lateral ankle sprains are the most common foot and ankle injury experienced by football players,7 numerous other injuries can occur, including turf toe, Jones fractures, Lisfranc injuries, syndesmotic disruption, deltoid complex avulsion, and Achilles ruptures. It is important for physicians to be able to recognize, diagnose, and appropriately treat these injuries in players in order to expedite recovery, restore function, and help prevent future injury and long-term sequelae. This review focuses on updated treatment principles, surgical advances, and rehabilitation protocols for common football foot and ankle injuries.
Turf Toe
The term “turf toe” was first used in 1976 to refer to hyperextension injuries and plantar capsule-ligament sprains of the hallux metatarsophalangeal (MTP) joint that can lead to progressive cock-up deformity.8 While these injuries can occur on any surface and disrupt soft tissue balance with functional implications, predisposing factors include increasing playing surface hardness and decreasing shoe stiffness. In a classic scenario, the foot is fixed in equinus as an axial load is placed on the back of the heel, resulting in forced dorsiflexion of the hallux MTP joint.9 As the proximal phalanx extends, the sesamoids are drawn distally and the more dorsal portion of the metatarsal head articular surface bears the majority of the load, causing partial or complete tearing of the plantar plate with or without hallux MTP dislocation. Osteochondral lesions of the MTP joint and subchondral edema of the metatarsal head can occur concurrently as the proximal phalanx impacts or shears across the metatarsal head articular surface.
Clinical examination should focus on hallux swelling, alignment, and flexor hallucis longus (FHL) function along with vertical instability of the hallux MTP joint using a Lachman test. Radiographs should be evaluated for proximal migration of the sesamoids or diastasis (Figures W1A-W1C).
Indications for surgical intervention include loss of push-off strength, gross MTP instability, proximal migration of the sesamoids, and progressive hallux malalignment or clawing after immobilization. Cases can involve one or a combination of the following: (1) large capsular avulsion with unstable MTP joint; (2) diastasis of bipartite sesamoid; (3) diastasis of sesamoid fracture; (4) retraction of sesamoid; (5) traumatic hallux valgus deformity; (6) vertical instability (positive Lachman test); (7) loose body in MTP joint; or (8) chondral injury in MTP joint. The goal of surgery is the restoration of anatomy in order to restore normal function of the hallux MTP joint.
We have found that using dual medial and plantar incisions places less traction on the plantar medial cutaneous nerve, improves lateral exposure, and provides better wound healing. The medial capsulotomy extends from the metatarsal neck to the mid-phalanx to provide complete visualization of the sesamoid complex (Figures 1A-1F).
It is important to recognize that not all turf toe injuries involve pure hyperextension on artificial playing surfaces. In recent years, we have found an increasing rate of medial variant turf toe injuries in which a forceful valgus stress on the hallux leads to rupture of the medial collateral ligament, medial or plantar-medial capsule, and/or abductor halluces. Medial variant turf toe can lead to progressive hallux valgus and a traumatic bunion with a significant loss of push-off strength and difficulty with cutting maneuvers. Surgical treatment requires a modified McBride bunionectomy with adductor tenotomy and direct repair of the medial soft tissue defect.
Postoperative management is just as important as proper surgical technique for these injuries and involves a delicate balance between protecting the repair and starting early range of motion (ROM). Patients are immobilized non-weight-bearing (NWB) for 5 to 7 days maximum followed immediately with the initiation of passive hallux plantarflexion to keep the sesamoids moving. Active hallux plantarflexion is started at 4 weeks after surgery with active dorsiflexion from 6 to 8 weeks. Patients are transitioned to an accommodative shoe with stiff hallux insert 8 weeks postoperative with continued therapy focusing on hallux ROM. Running is initiated at 12 weeks and return to play (RTP) is typically allowed 4 months after surgery.
Jones Fractures
Jones fractures are fractures of the 5th metatarsal at the metaphyseal-diaphyseal junction, where there is a watershed area of decreased vascularity between the intramedullary nutrient and metaphyseal arteries. Current thought is that the rising rate of Jones fractures among football players is partially caused by the use of flexible, narrow cleats that do not provide enough stiffness and lateral support for the 5th metatarsal during running and cutting. Additionally, lateral overload from a baseline cavovarus foot posture with or without metatarsus adductus and/or skewfoot is thought to contribute to Jones fractures.10 Preoperative radiographs should be evaluated for fracture location, orientation, amount of cortical thickening, and overall geometry of the foot and 5th metatarsal. In elite athletes, the threshold for surgical intervention is decreasing in order to expedite healing and recovery and decrease re-fracture risk. This rationale is based on delayed union rates of 25% to 66%, nonunion rates of 7% to 28%,11 and re-fracture rates of up to 33% associated with nonoperative treatment.12 Nonoperative management is usually not feasible in the competitive athlete, as it typically involves a period of protected weight-bearing in a tall controlled ankle motion (CAM) boot for 6 to 8 weeks with serial radiographs to evaluate healing.
Our preference for surgical intervention involves percutaneous screw fixation with a “high and inside” starting point on fluoroscopy (Figures 2A-2D).
In career athletes, we augment the fracture site using a mixture of bone marrow aspirate concentrate (BMA) (Magellan, Arteriocyte Medical Systems) and demineralized bone matrix (DBM) (Mini Ignite, Wright Medical Technology) injected percutaneously in and around the fracture site under fluoroscopic guidance. Using this technique in a cohort of 25 NFL players treated operatively for Jones fractures, we found that 100% of athletes were able to RTP in the NFL in an average of 9.5 weeks.14 Two patients (7.5%) suffered re-fractures requiring revision surgery with iliac crest bone graft and repeat screw placement with a RTP after 15 weeks. We did not find an association between RTP and re-fracture rate.
The appropriate rehabilitation protocol for Jones fractures after surgery remains controversial and dependent on individual needs and abilities.15,16 For athletes in-season, we recommend a brief period of NWB for 1 to 2 weeks followed by toe-touch weight-bearing in a tall CAM boot for 2 to 4 weeks. After 4 weeks, patients begin gentle exercises on a stationary bike and pool therapy to reduce impact on the fracture site. Low-intensity pulsed ultrasound bone stimulators (Exogen, Bioventus) are frequently used directly over fracture site throughout the postoperative protocol as an adjuvant therapy. If clinically nontender over the fracture site, patients are allowed to begin running in modified protective shoe wear 4 weeks after surgery with an average RTP of 6 to 8 weeks. RTP is determined clinically, as radiographic union may not be evident for 12 to 16 weeks. Useful custom orthoses include turf toe plates with a cushioned lateral column and lateral heel wedge if hindfoot varus is present preoperatively.10 The solid intramedullary screw is left in place permanently.
In our experience, we have found the average re-fracture and nonunion rate to be approximately 8% across all athletes. Our observation that union rates do not appear to be related to RTP times suggests that underlying biology such as Vitamin D deficiency may play a larger role in union rates than previously thought. We have found that most Jones re-fractures occur in the first year after the initial injury, but can occur up to 2.5 years afterwards as well.14 For the management of symptomatic re-fractures and nonunions, the previous screw must be first removed. This can be difficult if the screw is bent or broken, and may require a lateral corticotomy of the metatarsal.
After hardware removal, we advocate open bone grafting of the fracture site using bone from the iliac crest retrieved with a small, percutaneous trephine. Re-fixation should be achieved using a larger, solid screw and postoperative adjuvants may include bone stimulators, Vitamin D and calcium supplemention, and possible teriparatide use (Forteo, Eli Lilly), depending on individual patient profile. In a cohort of 21 elite athletes treated for Jones fracture revision surgery with screw exchange and bone grafting, we found that 100% of patients had computed tomography (CT) evidence of union, with an average RTP of 12.3 weeks.17
Lisfranc Injuries
Lisfranc injuries include any bony or ligamentous damage that involves the tarsometatarsal (TMT) joints. While axial loading of a fixed, plantarflexed foot has traditionally been thought of as the most common mechanism of Lisfranc injury, we have found that noncontact twisting injuries leading to Lisfranc disruption are actually more common among NFL players. This mechanism is similar to noncontact turf toe and results in a purely ligamentous injury. We have found this to be particularly true in the case of defensive ends engaged with offensive linemen in which no axial loading or contact of the foot occurs. Clinically, patients often have painful weight-bearing, inability to perform a single limb heel rise, plantar ecchymosis, and swelling and point tenderness across the bases of the 1st and 2nd metatarsals.
It is critical to obtain comparison weight-bearing radiographs of both feet during initial work-up to look for evidence of instability. Subtle radiographic findings of Lisfranc injury include a bony “fleck” sign, compression fracture of the cuboid, and diastasis between the base of the 1st and 2nd metatarsals and/or medial and middle cuneiforms (Figures 3A, 3B).
The goal of surgical intervention is to obtain and maintain anatomic reduction of all unstable joints in order to restore a normal foot posture. One of the difficulties with Lisfranc injuries is that there are no exact diastasis parameters and individuals should be treated based on symptoms, functional needs, and degree of instability. It has been shown that 5 mm of displacement can have good long-term clinical results in select cases without surgery.18 For surgery, we recommend open reduction to remove interposed soft tissue debris and directly assess the articular surfaces (Figures 4A-4D).
Proximal-medial column Lisfranc injury variants are increasingly common among football players.20 In these injuries, the force of injury extends through the intercuneiform joint and exits out the naviculocuneiform joint, thus causing instability at multiple joints and an unstable 1st ray. Patients often have minimal clinical findings and normal radiographs and stress radiographs. MRI of the foot often reveals edema at the naviculocuneiform joint. Often patients fail to improve with nonoperative immobilization with continued inability to push off from the hallux. Unrecognized or untreated instability will lead to rapid deterioration of the naviculocuneiform joint. Surgical intervention requires a homerun screw and intercuneiform screw. We do not recommend primary arthrodesis in athletes due to significant risk of malunion and nonunion unless severe articular damage is present.
Patients are typically kept NWB in a splint for 2 weeks after surgery followed by NWB in a tall CAM from 3 to 4 weeks postoperative. Progressive weight-bearing and ROM exercises are initiated from 4 to 8 weeks, followed by return to accommodative shoe wear from 10 to 12 weeks. Hardware removal is performed 4 to 6 months after surgery, typically in the off-season to allow for 6 to 8 weeks or protected recovery afterwards. Premature hardware removal can lead to loss of reduction, particularly at the intercuneiform joints. All hardware crossing the TMT joints should be removed, while the homerun screw can be left in place in addition to the intercuneiform screw. RTP in football typically occurs 6 to 7 months after surgery. Final functional outcome is related to the adequacy of initial reduction and severity of the initial injury.21
Syndesmotic Disruption
Syndesmotic injuries comprise 1% to 18% of ankle sprains in the general population, but occur at much higher rates in football due to the increased rotation forces placed on the ankle during cutting and tackling. RTP after syndesmotic injury often takes twice as long when compared to isolated lateral ankle ligamentous injury.22 Missed injuries are common and if not treated properly can lead to chronic ankle instability and posttraumatic ankle arthritis.23 Syndesmotic injury can occur in isolation or with concomitant adjacent bony, cartilaginous, or ligamentous injuries. Therefore, clinical examination and imaging work-up are critical to successful management.
Syndesmotic injuries often result from an external rotation force applied to a hyperdorsiflexed ankle while the foot is planted. This mechanism causes the fibula to externally rotate while translating posteriorly and laterally, resulting in rupture of the anterior inferior tibiofibular ligament (AITFL) first, followed by the deep deltoid ligament, interosseous ligament (IOL), and lastly posterior talofibular ligament.24 Most syndesmotic injuries involve rupture of only the AITFL and IOL.25 Multiple clinical stress tests have been designed to assess syndesmotic stability, including the squeeze test, external rotation stress test, crossed-leg test, and fibula-translation test.26-29 However, no physical examination maneuver has been shown to reliably predict the presence or degree of syndesmotic injury and therefore imaging studies are necessary.30
Initial imaging should include standing radiographs of the affected ankle. An increase in the medial clear space between the medial malleolus and talus can occur with a combined syndesmotic and deltoid disruption. In the case of subtle syndesmotic injuries, contralateral comparison weight-bearing radiographs are recommended. CT and MRI can provide additional information, but these static imaging tests cannot identify instability. Fluoroscopic stress evaluation is beneficial but has a high false-negative rate in low-grade injuries and may not detect partial rupture of the AITFL and IOL.31 It has been shown that malrotation of as much as 30° of external rotation can occur if relying on intraoperative fluoroscopy alone.32 It has been our practice to recommend surgical reduction and stabilization for any syndesmotic injury with documented diastasis or instability seen on imaging and/or arthroscopy.
Nonoperative treatment is indicated for truly stable grade I syndesmotic injuries. This involves rest and immobilization followed by a progressive rehabilitation program consisting of stretching, strengthening, and proprioceptive exercises.33 After 1 week of protected weight-bearing in a cast or tall CAM boot, progression to full weight-bearing should occur over the following week. Active-assisted ankle ROM exercises and light proprioceptive training should then be initiated followed by sport-specific exercises 2 to 3 weeks after injury.
Arthroscopy can be a valuable diagnostic tool in the setting of subtle syndesmotic injury with negative radiographs, positive MRI for edema, and a protracted recovery course with vague pain (Figures W5A-W5E).
Implants are placed above the true syndesmotic joint (at least 15 mm above the tibial plafond) angled 30° posterior to anterior to follow the normal relationship of the fibula to the distal tibia in the incisura. Typically 2 suture-buttons are used, with the devices placed in a divergent fashion. We highly recommend the use of a fibular buttress plate with button placement in individuals returning to contact activity. This construct increases surface area distribution while preventing stress risers and the risk of fibula fractures. In a cadaver model with deliberate syndesmotic malreduction, suture-button stabilization resulted in decreased postoperative displacement as opposed to conventional screw fixation.34 Therefore, dynamic syndesmotic fixation may help to decrease the negative sequelae of iatrogenic clamp malreduction. Postoperative rehabilitation involves NWB in a cast or tall CAM boot for 4 weeks followed by ankle ROM exercises and progressive weight-bearing and physical therapy. Patients are transitioned to a lace-up ankle brace and athletic shoe from 6 to 12 weeks postoperative with increasing activity. Running and jumping is permitted 4 months after surgery with RTP typically at 6 to 7 months. Athletes who have had surgical stabilization for documented instability without any diastasis may engage in a more rapid recovery and RTP as symptoms and function allow.
Deltoid Complex Avulsion
Missed or neglected deltoid ligament injuries can lead to progressive chondral injury and joint degeneration. These injuries are often subtle and difficult to diagnose. An inability to perform a single limb heel rise, persistent pain with activity, and lack of normal functional improvement despite appropriate care are indicators of subtle ligament instability. These injuries often require an examination under anesthesia with combined ankle arthroscopy. Valgus stress testing of the ankle while directly visualizing the deltoid ligament from the anterolateral portal can reveal medial laxity in addition to potential osteochondral lesions along the anterolateral talar dome.
In American football players, we have observed that infolding and retraction of an avulsed superficial deltoid ligament complex after an ankle fracture, Maisonneuve injury, or severe high ankle sprain can be a source of persistent increased medial clear space, malreduction, and postoperative pain and medial instability. We have found that there is often complete avulsion of the superficial deltoid complex off the proximal aspect of the medial malleolus during high-energy ankle fractures in football players that is amenable to direct repair to bone (Figures W6A-W6E).
During surgical repair, an incision is made along the anterior aspect of the medial malleolus and the superficial deltoid ligament complex can often be found flipped and interposed in the medial gutter. A rongeur is used to create a bleeding cancellous bone surface for soft-tissue healing and 1 to 2 suture anchors are used to repair and imbricate the deltoid ligament complex back to the medial malleolus. The goal of these sutures is to repair the tibionavicular and tibial spring ligaments back to the medial malleolus. We believe that superficial deltoid complex avulsion during high-energy ankle fractures is a distinct injury pattern that should be recognized and may benefit from primary open repair.
We currently open explore every deltoid ligament complex in athletes with unstable syndesmotic injuries, as we believe that deltoid avulsion injuries are underrecognized and do not heal in an anatomic fashion if left alone. Postoperative recovery follows the same immobilization, progressive weight-bearing, and physical therapy protocol as that for syndesmotic disruption.
Achilles Ruptures
Acute midsubstance Achilles tendon ruptures are an increasingly common injury in patients 30 to 50 years of age, with more than 50% of all injuries occurring during basketball.36,37 Among NFL players, we have found that Achilles ruptures tend to occur at a higher rate during training camp, when athletes are deconditioned and quickly returning to explosive push-off activities. Physical examination should include a Thompson test, palpation of a gap within the tendon, and evaluation of resting ankle dorsiflexion in the affected extremity in the prone position with the knees bent. Lateral radiographs should be analyzed for the presence of a bony avulsion fragment indicative of an insertional avulsion injury or midsubstance calcium deposition reflecting chronic Achilles tendinosis, as both of these conditions will change surgical management. MRI is not recommended with acute midsubstance ruptures but may be helpful in the case of chronic ruptures or more proximal tears of the musculotendinous junction.
The management of acute midsubstance Achilles tendon ruptures is controversial, with no general consensus in the literature regarding nonoperative treatment, surgical repair, and ideal repair technique.36,38-42 American Academy of Orthopaedic Surgeons clinical practice guidelines report moderate evidence that nonoperative treatment of Achilles tendon ruptures has lower wound healing complications but higher rates of re-rupture.38,39 Additionally, limited incision approaches have been found to have fewer overall complications compared with traditional open repair. In an effort to reduce the incidence of postoperative wound complications while improving functional recovery, modern repair techniques focus on a limited incision repair using percutaneous suture insertion and management (PARS Achilles Jig System, Arthrex).36 The limited incision technique utilizes a 2-cm transverse incision and non-disposable jig with divergent needle passes and locking suture fixation options to secure and fixate both tendon ends with minimal dissection of skin, subcutaneous tissue, and paratenon. Limited incision repair is ideally performed within 2 weeks of the injury to ensure that both tendon ends are easy to identify, mobilize, and repair. An open repair is generally recommended for midsubstance ruptures more than 4 weeks old and cases of insertional rupture and Achilles tendinopathy.
In a cohort of 9 NFL players treated for midsubstance Achilles ruptures using the PARS technique, we found no re-ruptures, no wound complications, and no sural nerve issues after surgery.43 A comparative review of 270 cases of operatively treated Achilles tendon ruptures (101 PARS, 169 traditional open repair) showed that the PARS group had significantly shorter operative times and a higher number of patients able to return to baseline physical activities by 5 months compared to open repair.36 Although not statistically significant, the overall PARS complication rate was 5% while the open complication rate was 11%. The PARS group had no cases of sural neuritis or deep infection requiring reoperation. We currently use a limited incision technique for all acute midsubstance Achilles ruptures in athletes regardless of sport, patient size, or position played.
During surgery, a 2-cm transverse incision is made over the gap in the Achilles tendon and dissection is carried down to the rupture site with minimal manipulation of the skin (Figures 5A-5F).
A key aspect of postoperative recovery is avoiding excessive ankle dorsiflexion while the tendon is healing during the first 4 weeks after surgery, as this can lead to an elongated tendon with loss of push-off strength. Patients are kept in a plantarflexion splint NWB for 2 weeks after surgery. If the incision is healed at 2 weeks, sutures are removed and patients are transitioned into a NWB tall CAM boot for 2 weeks with gentle ankle ROM exercises. If there is any concern regarding wound healing status, sutures are maintained for an additional 1 to 2 weeks.
From 4 to 8 weeks after surgery, progressive weight-bearing with continued ankle ROM exercises is initiated with peel-away heel lifts (~2 cm thick total, 3 layers). Each layer of the heel lift is gradually removed as pain allows every 2 to 3 days with the goal of being full weight-bearing with the foot flat at 6 weeks postoperative. Physical therapy focusing on ankle ROM and gentle Achilles stretching and strengthening is also started 6 weeks after surgery. From 8 to 12 weeks postoperative, patients are transitioned out of the tall CAM boot into normal, accommodative shoe wear with full weight-bearing. We avoid ankle dorsiflexion past neutral until 12 weeks after surgery, as overlengthening of the Achilles complex and the subsequent loss of push-off power can be devastating to running athletes. Activity levels are increased as tolerated, with no running or jumping from 12 to 16 weeks with full release to all activities after 16 weeks. RTP often takes 5 to 6 months after surgery, depending on the position played.
Am J Orthop. 2016;45(6):358-367. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Canale ST, Cantler ED Jr, Sisk TD, Freeman BL 3rd. A chronicle of injuries of an American intercollegiate football team. Am J Sports Med. 1981;9(6):384-389.2. Robey JM, Blyth CS, Mueller FO. Athletic injuries. Application of epidemiologic methods. JAMA. 1971;217(2):184-189.
3. Saal JA. Common American football injuries. Sports Med. 1991;12(2):132-147.
4. Thompson N, Halpern B, Curl WW, et al. High school football injuries: evaluation. Am J Sports Med. 1987;15(2):117-124.
5. Kaplan LD, Jost PW, Honkamp N, Norwig J, West R, Bradley JP. Incidence and variance of foot and ankle injuries in elite college football players. Am J Orthop. 2011;40(1):40-44.
6. DeLee JC, Farney WC. Incidence of injury in Texas high school football. Am J Sports Med. 1992;20(5):575-580.
7. Brophy RH, Barnes R, Rodeo SA, Warren RF. Prevalence of musculoskeletal disorders at the NFL Combine--trends from 1987 to 2000. Med Sci Sports Exerc. 2007;39(1):22-27.
8. Bowers KD Jr, Martin RB. Turf-toe: a shoe-surface related football injury. Med Sci Sports. 1976;8(2):81-83.
9. McCormick JJ, Anderson RB. Turf toe: anatomy, diagnosis, and treatment. Sports Health. 2010;2(6):487-494.
10. Raikin SM, Slenker N, Ratigan B. The association of a varus hindfoot and fracture of the fifth metatarsal metaphyseal-diaphyseal junction: the Jones fracture. Am J Sports Med. 2008;36(7):1367-1372.
11. Title CI, Katchis SD. Traumatic foot and ankle injuries in the athlete. Orthop Clin North Am. 2002;33(3):587-598.
12. Quill GE Jr. Fractures of the proximal fifth metatarsal. Orthop Clin North Am. 1995;26(2):353-361.
13. Nunley JA, Glisson RR. A new option for intramedullary fixation of Jones fractures: the Charlotte Carolina Jones Fracture System. Foot Ankle Int. 2008;29(12):1216-1221.
14. Lareau CR, Hsu AR, Anderson RB. Return to play in National Football League players after operative Jones fracture treatment. Foot Ankle Int. 2016;37(1):8-16.
15. Larson CM, Almekinders LC, Taft TN, Garrett WE. Intramedullary screw fixation of Jones fractures. Analysis of failure. Am J Sports Med. 2002;30(1):55-60.
16. Portland G, Kelikian A, Kodros S. Acute surgical management of Jones’ fractures. Foot Ankle Int. 2003;24(11):829-833.
17. Hunt KJ, Anderson RB. Treatment of Jones fracture nonunions and refractures in the elite athlete: outcomes of intramedullary screw fixation with bone grafting. Am J Sports Med. 2011;39(9):1948-1954.
18. Nunley JA, Vertullo CJ. Classification, investigation, and management of midfoot sprains: Lisfranc injuries in the athlete. Am J Sports Med. 2002;30(6):871-878.
19. Alberta FG, Aronow MS, Barrero M, Diaz-Doran V, Sullivan RJ, Adams DJ. Ligamentous Lisfranc joint injuries: a biomechanical comparison of dorsal plate and transarticular screw fixation. Foot Ankle Int. 2005;26(6):462-473.
20. Ardoin GT, Anderson RB. Subtle Lisfranc injury. Tech Foot Ankle Surg. 2010;9(3):100-106.
21. Kuo RS, Tejwani NC, Digiovanni CW, et al. Outcome after open reduction and internal fixation of Lisfranc joint injuries. J Bone Joint Surg Am. 2000;82-A(11):1609-1618.
22. Wright RW, Barile RJ, Surprenant DA, Matava MJ. Ankle syndesmosis sprains in national hockey league players. Am J Sports Med. 2004;32(8):1941-1945.
23. Williams GN, Jones MH, Amendola A. Syndesmotic ankle sprains in athletes. Am J Sports Med. 2007;35(7):1197-1207.
24. Beumer A, Valstar ER, Garling EH, et al. Effects of ligament sectioning on the kinematics of the distal tibiofibular syndesmosis: a radiostereometric study of 10 cadaveric specimens based on presumed trauma mechanisms with suggestions for treatment. Acta Orthop. 2006;77(3):531-540.
25. McCollum GA, van den Bekerom MP, Kerkhoffs GM, Calder JD, van Dijk CN. Syndesmosis and deltoid ligament injuries in the athlete. Knee Surg Sports Traumatol Arthrosc. 2013;21(6):1328-1337.
26. Boytim MJ, Fischer DA, Neumann L. Syndesmotic ankle sprains. Am J Sports Med. 1991;19(3):294-298.
27. Nussbaum ED, Hosea TM, Sieler SD, Incremona BR, Kessler DE. Prospective evaluation of syndesmotic ankle sprains without diastasis. Am J Sports Med. 2001;29(1):31-35.
28. Kiter E, Bozkurt M. The crossed-leg test for examination of ankle syndesmosis injuries. Foot Ankle Int. 2005;26(2):187-188.
29. Beumer A, van Hemert WL, Swierstra BA, Jasper LE, Belkoff SM. A biomechanical evaluation of clinical stress tests for syndesmotic ankle instability. Foot Ankle Int. 2003;24(4):358-363.
30. Amendola A, Williams G, Foster D. Evidence-based approach to treatment of acute traumatic syndesmosis (high ankle) sprains. Sports Med Arthrosc. 2006;14(4):232-236.
31. Beumer A, Valstar ER, Garling EH, et al. External rotation stress imaging in syndesmotic injuries of the ankle: comparison of lateral radiography and radiostereometry in a cadaveric model. Acta Orthop Scand. 2003;74(2):201-205.
32. Marmor M, Hansen E, Han HK, Buckley J, Matityahu A. Limitations of standard fluoroscopy in detecting rotational malreduction of the syndesmosis in an ankle fracture model. Foot Ankle Int. 2011;32(6):616-622.
33. Williams GN, Allen EJ. Rehabilitation of syndesmotic (high) ankle sprains. Sports Health. 2010;2(6):460-470.
34. Westermann RW, Rungprai C, Goetz JE, Femino J, Amendola A, Phisitkul P. The effect of suture-button fixation on simulated syndesmotic malreduction: a cadaveric study. J Bone Joint Surg Am. 2014;96(20):1732-1738.
35. Hsu AR, Lareau CR, Anderson RB. Repair of acute superficial deltoid complex avulsion during ankle fracture fixation in National Football League players. Foot Ankle Int. 2015;36(11):1272-1278.
36. Hsu AR, Jones CP, Cohen BE, Davis WH, Ellington JK, Anderson RB. Clinical outcomes and complications of percutaneous Achilles repair system versus open technique for acute achilles tendon ruptures. Foot Ankle Int. 2015;36(11):1279-1286.
37. Raikin SM, Garras DN, Krapchev PV. Achilles tendon injuries in a United States population. Foot Ankle Int. 2013;34(4):475-480.
38. Chiodo CP, Glazebrook M, Bluman EM, et al. American Academy of Orthopaedic Surgeons clinical practice guideline on treatment of achilles tendon rupture. J Bone Joint Surg Am. 2010;92(14):2466-2468.
39. Chiodo CP, Glazebrook M, Bluman EM, et al. Diagnosis and treatment of acute achilles tendon rupture. J Am Acad Orthop Surg. 2010;18(8):503-510.
40. Khan RJ, Fick D, Keogh A, Crawford J, Brammar T, Parker M. Treatment of acute achilles tendon ruptures. A meta-analysis of randomized, controlled trials. J Bone Joint Surg Am. 2005;87(10):2202-2210.
41. Renninger CH, Kuhn K, Fellars T, Youngblood S, Bellamy J. Operative and nonoperative management of achilles tendon ruptures in active duty military population. Foot Ankle Int. 2016;37(3):269-273.
42. Khan RJ, Carey Smith RL. Surgical interventions for treating acute achilles tendon ruptures. Cochrane Database Syst Rev. 2010;(9):CD003674.
43. McCullough KA, Shaw CM, Anderson RB. Mini-open repair of achilles rupture in the national football league. J Surg Orthop Adv. 2014;23(4):179-183.
Foot and ankle injuries are common in American football, with injury rates significantly increasing over the past decade.1-5 Epidemiologic studies of collegiate football players have shown an annual incidence of foot and ankle injuries ranging from 9% to 39%,3,6 with as many as 72% of all collegiate players presenting to the National Football League (NFL) Combine with a history of a foot or ankle injury and 13% undergoing surgical treatment.5 Player position influences the rate and type of foot and ankle injury. Offensive and “skill position” players, including linemen, running backs, and wide receivers, are particularly susceptible to foot and ankle injuries due to high levels of force and torque placed on the distal extremity during running, cutting, and tackling. Shoe wear changes, playing field conditions, increasing player size and speed, and improved reporting of injuries are also contributing to increasing injury rates.
The interaction between player cleats and the playing surface is a central issue of foot and ankle injuries in football. Improved traction relates to performance, but increased subsequent torque on the lower extremity is associated with injury. While lateral ankle sprains are the most common foot and ankle injury experienced by football players,7 numerous other injuries can occur, including turf toe, Jones fractures, Lisfranc injuries, syndesmotic disruption, deltoid complex avulsion, and Achilles ruptures. It is important for physicians to be able to recognize, diagnose, and appropriately treat these injuries in players in order to expedite recovery, restore function, and help prevent future injury and long-term sequelae. This review focuses on updated treatment principles, surgical advances, and rehabilitation protocols for common football foot and ankle injuries.
Turf Toe
The term “turf toe” was first used in 1976 to refer to hyperextension injuries and plantar capsule-ligament sprains of the hallux metatarsophalangeal (MTP) joint that can lead to progressive cock-up deformity.8 While these injuries can occur on any surface and disrupt soft tissue balance with functional implications, predisposing factors include increasing playing surface hardness and decreasing shoe stiffness. In a classic scenario, the foot is fixed in equinus as an axial load is placed on the back of the heel, resulting in forced dorsiflexion of the hallux MTP joint.9 As the proximal phalanx extends, the sesamoids are drawn distally and the more dorsal portion of the metatarsal head articular surface bears the majority of the load, causing partial or complete tearing of the plantar plate with or without hallux MTP dislocation. Osteochondral lesions of the MTP joint and subchondral edema of the metatarsal head can occur concurrently as the proximal phalanx impacts or shears across the metatarsal head articular surface.
Clinical examination should focus on hallux swelling, alignment, and flexor hallucis longus (FHL) function along with vertical instability of the hallux MTP joint using a Lachman test. Radiographs should be evaluated for proximal migration of the sesamoids or diastasis (Figures W1A-W1C).
Indications for surgical intervention include loss of push-off strength, gross MTP instability, proximal migration of the sesamoids, and progressive hallux malalignment or clawing after immobilization. Cases can involve one or a combination of the following: (1) large capsular avulsion with unstable MTP joint; (2) diastasis of bipartite sesamoid; (3) diastasis of sesamoid fracture; (4) retraction of sesamoid; (5) traumatic hallux valgus deformity; (6) vertical instability (positive Lachman test); (7) loose body in MTP joint; or (8) chondral injury in MTP joint. The goal of surgery is the restoration of anatomy in order to restore normal function of the hallux MTP joint.
We have found that using dual medial and plantar incisions places less traction on the plantar medial cutaneous nerve, improves lateral exposure, and provides better wound healing. The medial capsulotomy extends from the metatarsal neck to the mid-phalanx to provide complete visualization of the sesamoid complex (Figures 1A-1F).
It is important to recognize that not all turf toe injuries involve pure hyperextension on artificial playing surfaces. In recent years, we have found an increasing rate of medial variant turf toe injuries in which a forceful valgus stress on the hallux leads to rupture of the medial collateral ligament, medial or plantar-medial capsule, and/or abductor halluces. Medial variant turf toe can lead to progressive hallux valgus and a traumatic bunion with a significant loss of push-off strength and difficulty with cutting maneuvers. Surgical treatment requires a modified McBride bunionectomy with adductor tenotomy and direct repair of the medial soft tissue defect.
Postoperative management is just as important as proper surgical technique for these injuries and involves a delicate balance between protecting the repair and starting early range of motion (ROM). Patients are immobilized non-weight-bearing (NWB) for 5 to 7 days maximum followed immediately with the initiation of passive hallux plantarflexion to keep the sesamoids moving. Active hallux plantarflexion is started at 4 weeks after surgery with active dorsiflexion from 6 to 8 weeks. Patients are transitioned to an accommodative shoe with stiff hallux insert 8 weeks postoperative with continued therapy focusing on hallux ROM. Running is initiated at 12 weeks and return to play (RTP) is typically allowed 4 months after surgery.
Jones Fractures
Jones fractures are fractures of the 5th metatarsal at the metaphyseal-diaphyseal junction, where there is a watershed area of decreased vascularity between the intramedullary nutrient and metaphyseal arteries. Current thought is that the rising rate of Jones fractures among football players is partially caused by the use of flexible, narrow cleats that do not provide enough stiffness and lateral support for the 5th metatarsal during running and cutting. Additionally, lateral overload from a baseline cavovarus foot posture with or without metatarsus adductus and/or skewfoot is thought to contribute to Jones fractures.10 Preoperative radiographs should be evaluated for fracture location, orientation, amount of cortical thickening, and overall geometry of the foot and 5th metatarsal. In elite athletes, the threshold for surgical intervention is decreasing in order to expedite healing and recovery and decrease re-fracture risk. This rationale is based on delayed union rates of 25% to 66%, nonunion rates of 7% to 28%,11 and re-fracture rates of up to 33% associated with nonoperative treatment.12 Nonoperative management is usually not feasible in the competitive athlete, as it typically involves a period of protected weight-bearing in a tall controlled ankle motion (CAM) boot for 6 to 8 weeks with serial radiographs to evaluate healing.
Our preference for surgical intervention involves percutaneous screw fixation with a “high and inside” starting point on fluoroscopy (Figures 2A-2D).
In career athletes, we augment the fracture site using a mixture of bone marrow aspirate concentrate (BMA) (Magellan, Arteriocyte Medical Systems) and demineralized bone matrix (DBM) (Mini Ignite, Wright Medical Technology) injected percutaneously in and around the fracture site under fluoroscopic guidance. Using this technique in a cohort of 25 NFL players treated operatively for Jones fractures, we found that 100% of athletes were able to RTP in the NFL in an average of 9.5 weeks.14 Two patients (7.5%) suffered re-fractures requiring revision surgery with iliac crest bone graft and repeat screw placement with a RTP after 15 weeks. We did not find an association between RTP and re-fracture rate.
The appropriate rehabilitation protocol for Jones fractures after surgery remains controversial and dependent on individual needs and abilities.15,16 For athletes in-season, we recommend a brief period of NWB for 1 to 2 weeks followed by toe-touch weight-bearing in a tall CAM boot for 2 to 4 weeks. After 4 weeks, patients begin gentle exercises on a stationary bike and pool therapy to reduce impact on the fracture site. Low-intensity pulsed ultrasound bone stimulators (Exogen, Bioventus) are frequently used directly over fracture site throughout the postoperative protocol as an adjuvant therapy. If clinically nontender over the fracture site, patients are allowed to begin running in modified protective shoe wear 4 weeks after surgery with an average RTP of 6 to 8 weeks. RTP is determined clinically, as radiographic union may not be evident for 12 to 16 weeks. Useful custom orthoses include turf toe plates with a cushioned lateral column and lateral heel wedge if hindfoot varus is present preoperatively.10 The solid intramedullary screw is left in place permanently.
In our experience, we have found the average re-fracture and nonunion rate to be approximately 8% across all athletes. Our observation that union rates do not appear to be related to RTP times suggests that underlying biology such as Vitamin D deficiency may play a larger role in union rates than previously thought. We have found that most Jones re-fractures occur in the first year after the initial injury, but can occur up to 2.5 years afterwards as well.14 For the management of symptomatic re-fractures and nonunions, the previous screw must be first removed. This can be difficult if the screw is bent or broken, and may require a lateral corticotomy of the metatarsal.
After hardware removal, we advocate open bone grafting of the fracture site using bone from the iliac crest retrieved with a small, percutaneous trephine. Re-fixation should be achieved using a larger, solid screw and postoperative adjuvants may include bone stimulators, Vitamin D and calcium supplemention, and possible teriparatide use (Forteo, Eli Lilly), depending on individual patient profile. In a cohort of 21 elite athletes treated for Jones fracture revision surgery with screw exchange and bone grafting, we found that 100% of patients had computed tomography (CT) evidence of union, with an average RTP of 12.3 weeks.17
Lisfranc Injuries
Lisfranc injuries include any bony or ligamentous damage that involves the tarsometatarsal (TMT) joints. While axial loading of a fixed, plantarflexed foot has traditionally been thought of as the most common mechanism of Lisfranc injury, we have found that noncontact twisting injuries leading to Lisfranc disruption are actually more common among NFL players. This mechanism is similar to noncontact turf toe and results in a purely ligamentous injury. We have found this to be particularly true in the case of defensive ends engaged with offensive linemen in which no axial loading or contact of the foot occurs. Clinically, patients often have painful weight-bearing, inability to perform a single limb heel rise, plantar ecchymosis, and swelling and point tenderness across the bases of the 1st and 2nd metatarsals.
It is critical to obtain comparison weight-bearing radiographs of both feet during initial work-up to look for evidence of instability. Subtle radiographic findings of Lisfranc injury include a bony “fleck” sign, compression fracture of the cuboid, and diastasis between the base of the 1st and 2nd metatarsals and/or medial and middle cuneiforms (Figures 3A, 3B).
The goal of surgical intervention is to obtain and maintain anatomic reduction of all unstable joints in order to restore a normal foot posture. One of the difficulties with Lisfranc injuries is that there are no exact diastasis parameters and individuals should be treated based on symptoms, functional needs, and degree of instability. It has been shown that 5 mm of displacement can have good long-term clinical results in select cases without surgery.18 For surgery, we recommend open reduction to remove interposed soft tissue debris and directly assess the articular surfaces (Figures 4A-4D).
Proximal-medial column Lisfranc injury variants are increasingly common among football players.20 In these injuries, the force of injury extends through the intercuneiform joint and exits out the naviculocuneiform joint, thus causing instability at multiple joints and an unstable 1st ray. Patients often have minimal clinical findings and normal radiographs and stress radiographs. MRI of the foot often reveals edema at the naviculocuneiform joint. Often patients fail to improve with nonoperative immobilization with continued inability to push off from the hallux. Unrecognized or untreated instability will lead to rapid deterioration of the naviculocuneiform joint. Surgical intervention requires a homerun screw and intercuneiform screw. We do not recommend primary arthrodesis in athletes due to significant risk of malunion and nonunion unless severe articular damage is present.
Patients are typically kept NWB in a splint for 2 weeks after surgery followed by NWB in a tall CAM from 3 to 4 weeks postoperative. Progressive weight-bearing and ROM exercises are initiated from 4 to 8 weeks, followed by return to accommodative shoe wear from 10 to 12 weeks. Hardware removal is performed 4 to 6 months after surgery, typically in the off-season to allow for 6 to 8 weeks or protected recovery afterwards. Premature hardware removal can lead to loss of reduction, particularly at the intercuneiform joints. All hardware crossing the TMT joints should be removed, while the homerun screw can be left in place in addition to the intercuneiform screw. RTP in football typically occurs 6 to 7 months after surgery. Final functional outcome is related to the adequacy of initial reduction and severity of the initial injury.21
Syndesmotic Disruption
Syndesmotic injuries comprise 1% to 18% of ankle sprains in the general population, but occur at much higher rates in football due to the increased rotation forces placed on the ankle during cutting and tackling. RTP after syndesmotic injury often takes twice as long when compared to isolated lateral ankle ligamentous injury.22 Missed injuries are common and if not treated properly can lead to chronic ankle instability and posttraumatic ankle arthritis.23 Syndesmotic injury can occur in isolation or with concomitant adjacent bony, cartilaginous, or ligamentous injuries. Therefore, clinical examination and imaging work-up are critical to successful management.
Syndesmotic injuries often result from an external rotation force applied to a hyperdorsiflexed ankle while the foot is planted. This mechanism causes the fibula to externally rotate while translating posteriorly and laterally, resulting in rupture of the anterior inferior tibiofibular ligament (AITFL) first, followed by the deep deltoid ligament, interosseous ligament (IOL), and lastly posterior talofibular ligament.24 Most syndesmotic injuries involve rupture of only the AITFL and IOL.25 Multiple clinical stress tests have been designed to assess syndesmotic stability, including the squeeze test, external rotation stress test, crossed-leg test, and fibula-translation test.26-29 However, no physical examination maneuver has been shown to reliably predict the presence or degree of syndesmotic injury and therefore imaging studies are necessary.30
Initial imaging should include standing radiographs of the affected ankle. An increase in the medial clear space between the medial malleolus and talus can occur with a combined syndesmotic and deltoid disruption. In the case of subtle syndesmotic injuries, contralateral comparison weight-bearing radiographs are recommended. CT and MRI can provide additional information, but these static imaging tests cannot identify instability. Fluoroscopic stress evaluation is beneficial but has a high false-negative rate in low-grade injuries and may not detect partial rupture of the AITFL and IOL.31 It has been shown that malrotation of as much as 30° of external rotation can occur if relying on intraoperative fluoroscopy alone.32 It has been our practice to recommend surgical reduction and stabilization for any syndesmotic injury with documented diastasis or instability seen on imaging and/or arthroscopy.
Nonoperative treatment is indicated for truly stable grade I syndesmotic injuries. This involves rest and immobilization followed by a progressive rehabilitation program consisting of stretching, strengthening, and proprioceptive exercises.33 After 1 week of protected weight-bearing in a cast or tall CAM boot, progression to full weight-bearing should occur over the following week. Active-assisted ankle ROM exercises and light proprioceptive training should then be initiated followed by sport-specific exercises 2 to 3 weeks after injury.
Arthroscopy can be a valuable diagnostic tool in the setting of subtle syndesmotic injury with negative radiographs, positive MRI for edema, and a protracted recovery course with vague pain (Figures W5A-W5E).
Implants are placed above the true syndesmotic joint (at least 15 mm above the tibial plafond) angled 30° posterior to anterior to follow the normal relationship of the fibula to the distal tibia in the incisura. Typically 2 suture-buttons are used, with the devices placed in a divergent fashion. We highly recommend the use of a fibular buttress plate with button placement in individuals returning to contact activity. This construct increases surface area distribution while preventing stress risers and the risk of fibula fractures. In a cadaver model with deliberate syndesmotic malreduction, suture-button stabilization resulted in decreased postoperative displacement as opposed to conventional screw fixation.34 Therefore, dynamic syndesmotic fixation may help to decrease the negative sequelae of iatrogenic clamp malreduction. Postoperative rehabilitation involves NWB in a cast or tall CAM boot for 4 weeks followed by ankle ROM exercises and progressive weight-bearing and physical therapy. Patients are transitioned to a lace-up ankle brace and athletic shoe from 6 to 12 weeks postoperative with increasing activity. Running and jumping is permitted 4 months after surgery with RTP typically at 6 to 7 months. Athletes who have had surgical stabilization for documented instability without any diastasis may engage in a more rapid recovery and RTP as symptoms and function allow.
Deltoid Complex Avulsion
Missed or neglected deltoid ligament injuries can lead to progressive chondral injury and joint degeneration. These injuries are often subtle and difficult to diagnose. An inability to perform a single limb heel rise, persistent pain with activity, and lack of normal functional improvement despite appropriate care are indicators of subtle ligament instability. These injuries often require an examination under anesthesia with combined ankle arthroscopy. Valgus stress testing of the ankle while directly visualizing the deltoid ligament from the anterolateral portal can reveal medial laxity in addition to potential osteochondral lesions along the anterolateral talar dome.
In American football players, we have observed that infolding and retraction of an avulsed superficial deltoid ligament complex after an ankle fracture, Maisonneuve injury, or severe high ankle sprain can be a source of persistent increased medial clear space, malreduction, and postoperative pain and medial instability. We have found that there is often complete avulsion of the superficial deltoid complex off the proximal aspect of the medial malleolus during high-energy ankle fractures in football players that is amenable to direct repair to bone (Figures W6A-W6E).
During surgical repair, an incision is made along the anterior aspect of the medial malleolus and the superficial deltoid ligament complex can often be found flipped and interposed in the medial gutter. A rongeur is used to create a bleeding cancellous bone surface for soft-tissue healing and 1 to 2 suture anchors are used to repair and imbricate the deltoid ligament complex back to the medial malleolus. The goal of these sutures is to repair the tibionavicular and tibial spring ligaments back to the medial malleolus. We believe that superficial deltoid complex avulsion during high-energy ankle fractures is a distinct injury pattern that should be recognized and may benefit from primary open repair.
We currently open explore every deltoid ligament complex in athletes with unstable syndesmotic injuries, as we believe that deltoid avulsion injuries are underrecognized and do not heal in an anatomic fashion if left alone. Postoperative recovery follows the same immobilization, progressive weight-bearing, and physical therapy protocol as that for syndesmotic disruption.
Achilles Ruptures
Acute midsubstance Achilles tendon ruptures are an increasingly common injury in patients 30 to 50 years of age, with more than 50% of all injuries occurring during basketball.36,37 Among NFL players, we have found that Achilles ruptures tend to occur at a higher rate during training camp, when athletes are deconditioned and quickly returning to explosive push-off activities. Physical examination should include a Thompson test, palpation of a gap within the tendon, and evaluation of resting ankle dorsiflexion in the affected extremity in the prone position with the knees bent. Lateral radiographs should be analyzed for the presence of a bony avulsion fragment indicative of an insertional avulsion injury or midsubstance calcium deposition reflecting chronic Achilles tendinosis, as both of these conditions will change surgical management. MRI is not recommended with acute midsubstance ruptures but may be helpful in the case of chronic ruptures or more proximal tears of the musculotendinous junction.
The management of acute midsubstance Achilles tendon ruptures is controversial, with no general consensus in the literature regarding nonoperative treatment, surgical repair, and ideal repair technique.36,38-42 American Academy of Orthopaedic Surgeons clinical practice guidelines report moderate evidence that nonoperative treatment of Achilles tendon ruptures has lower wound healing complications but higher rates of re-rupture.38,39 Additionally, limited incision approaches have been found to have fewer overall complications compared with traditional open repair. In an effort to reduce the incidence of postoperative wound complications while improving functional recovery, modern repair techniques focus on a limited incision repair using percutaneous suture insertion and management (PARS Achilles Jig System, Arthrex).36 The limited incision technique utilizes a 2-cm transverse incision and non-disposable jig with divergent needle passes and locking suture fixation options to secure and fixate both tendon ends with minimal dissection of skin, subcutaneous tissue, and paratenon. Limited incision repair is ideally performed within 2 weeks of the injury to ensure that both tendon ends are easy to identify, mobilize, and repair. An open repair is generally recommended for midsubstance ruptures more than 4 weeks old and cases of insertional rupture and Achilles tendinopathy.
In a cohort of 9 NFL players treated for midsubstance Achilles ruptures using the PARS technique, we found no re-ruptures, no wound complications, and no sural nerve issues after surgery.43 A comparative review of 270 cases of operatively treated Achilles tendon ruptures (101 PARS, 169 traditional open repair) showed that the PARS group had significantly shorter operative times and a higher number of patients able to return to baseline physical activities by 5 months compared to open repair.36 Although not statistically significant, the overall PARS complication rate was 5% while the open complication rate was 11%. The PARS group had no cases of sural neuritis or deep infection requiring reoperation. We currently use a limited incision technique for all acute midsubstance Achilles ruptures in athletes regardless of sport, patient size, or position played.
During surgery, a 2-cm transverse incision is made over the gap in the Achilles tendon and dissection is carried down to the rupture site with minimal manipulation of the skin (Figures 5A-5F).
A key aspect of postoperative recovery is avoiding excessive ankle dorsiflexion while the tendon is healing during the first 4 weeks after surgery, as this can lead to an elongated tendon with loss of push-off strength. Patients are kept in a plantarflexion splint NWB for 2 weeks after surgery. If the incision is healed at 2 weeks, sutures are removed and patients are transitioned into a NWB tall CAM boot for 2 weeks with gentle ankle ROM exercises. If there is any concern regarding wound healing status, sutures are maintained for an additional 1 to 2 weeks.
From 4 to 8 weeks after surgery, progressive weight-bearing with continued ankle ROM exercises is initiated with peel-away heel lifts (~2 cm thick total, 3 layers). Each layer of the heel lift is gradually removed as pain allows every 2 to 3 days with the goal of being full weight-bearing with the foot flat at 6 weeks postoperative. Physical therapy focusing on ankle ROM and gentle Achilles stretching and strengthening is also started 6 weeks after surgery. From 8 to 12 weeks postoperative, patients are transitioned out of the tall CAM boot into normal, accommodative shoe wear with full weight-bearing. We avoid ankle dorsiflexion past neutral until 12 weeks after surgery, as overlengthening of the Achilles complex and the subsequent loss of push-off power can be devastating to running athletes. Activity levels are increased as tolerated, with no running or jumping from 12 to 16 weeks with full release to all activities after 16 weeks. RTP often takes 5 to 6 months after surgery, depending on the position played.
Am J Orthop. 2016;45(6):358-367. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
Foot and ankle injuries are common in American football, with injury rates significantly increasing over the past decade.1-5 Epidemiologic studies of collegiate football players have shown an annual incidence of foot and ankle injuries ranging from 9% to 39%,3,6 with as many as 72% of all collegiate players presenting to the National Football League (NFL) Combine with a history of a foot or ankle injury and 13% undergoing surgical treatment.5 Player position influences the rate and type of foot and ankle injury. Offensive and “skill position” players, including linemen, running backs, and wide receivers, are particularly susceptible to foot and ankle injuries due to high levels of force and torque placed on the distal extremity during running, cutting, and tackling. Shoe wear changes, playing field conditions, increasing player size and speed, and improved reporting of injuries are also contributing to increasing injury rates.
The interaction between player cleats and the playing surface is a central issue of foot and ankle injuries in football. Improved traction relates to performance, but increased subsequent torque on the lower extremity is associated with injury. While lateral ankle sprains are the most common foot and ankle injury experienced by football players,7 numerous other injuries can occur, including turf toe, Jones fractures, Lisfranc injuries, syndesmotic disruption, deltoid complex avulsion, and Achilles ruptures. It is important for physicians to be able to recognize, diagnose, and appropriately treat these injuries in players in order to expedite recovery, restore function, and help prevent future injury and long-term sequelae. This review focuses on updated treatment principles, surgical advances, and rehabilitation protocols for common football foot and ankle injuries.
Turf Toe
The term “turf toe” was first used in 1976 to refer to hyperextension injuries and plantar capsule-ligament sprains of the hallux metatarsophalangeal (MTP) joint that can lead to progressive cock-up deformity.8 While these injuries can occur on any surface and disrupt soft tissue balance with functional implications, predisposing factors include increasing playing surface hardness and decreasing shoe stiffness. In a classic scenario, the foot is fixed in equinus as an axial load is placed on the back of the heel, resulting in forced dorsiflexion of the hallux MTP joint.9 As the proximal phalanx extends, the sesamoids are drawn distally and the more dorsal portion of the metatarsal head articular surface bears the majority of the load, causing partial or complete tearing of the plantar plate with or without hallux MTP dislocation. Osteochondral lesions of the MTP joint and subchondral edema of the metatarsal head can occur concurrently as the proximal phalanx impacts or shears across the metatarsal head articular surface.
Clinical examination should focus on hallux swelling, alignment, and flexor hallucis longus (FHL) function along with vertical instability of the hallux MTP joint using a Lachman test. Radiographs should be evaluated for proximal migration of the sesamoids or diastasis (Figures W1A-W1C).
Indications for surgical intervention include loss of push-off strength, gross MTP instability, proximal migration of the sesamoids, and progressive hallux malalignment or clawing after immobilization. Cases can involve one or a combination of the following: (1) large capsular avulsion with unstable MTP joint; (2) diastasis of bipartite sesamoid; (3) diastasis of sesamoid fracture; (4) retraction of sesamoid; (5) traumatic hallux valgus deformity; (6) vertical instability (positive Lachman test); (7) loose body in MTP joint; or (8) chondral injury in MTP joint. The goal of surgery is the restoration of anatomy in order to restore normal function of the hallux MTP joint.
We have found that using dual medial and plantar incisions places less traction on the plantar medial cutaneous nerve, improves lateral exposure, and provides better wound healing. The medial capsulotomy extends from the metatarsal neck to the mid-phalanx to provide complete visualization of the sesamoid complex (Figures 1A-1F).
It is important to recognize that not all turf toe injuries involve pure hyperextension on artificial playing surfaces. In recent years, we have found an increasing rate of medial variant turf toe injuries in which a forceful valgus stress on the hallux leads to rupture of the medial collateral ligament, medial or plantar-medial capsule, and/or abductor halluces. Medial variant turf toe can lead to progressive hallux valgus and a traumatic bunion with a significant loss of push-off strength and difficulty with cutting maneuvers. Surgical treatment requires a modified McBride bunionectomy with adductor tenotomy and direct repair of the medial soft tissue defect.
Postoperative management is just as important as proper surgical technique for these injuries and involves a delicate balance between protecting the repair and starting early range of motion (ROM). Patients are immobilized non-weight-bearing (NWB) for 5 to 7 days maximum followed immediately with the initiation of passive hallux plantarflexion to keep the sesamoids moving. Active hallux plantarflexion is started at 4 weeks after surgery with active dorsiflexion from 6 to 8 weeks. Patients are transitioned to an accommodative shoe with stiff hallux insert 8 weeks postoperative with continued therapy focusing on hallux ROM. Running is initiated at 12 weeks and return to play (RTP) is typically allowed 4 months after surgery.
Jones Fractures
Jones fractures are fractures of the 5th metatarsal at the metaphyseal-diaphyseal junction, where there is a watershed area of decreased vascularity between the intramedullary nutrient and metaphyseal arteries. Current thought is that the rising rate of Jones fractures among football players is partially caused by the use of flexible, narrow cleats that do not provide enough stiffness and lateral support for the 5th metatarsal during running and cutting. Additionally, lateral overload from a baseline cavovarus foot posture with or without metatarsus adductus and/or skewfoot is thought to contribute to Jones fractures.10 Preoperative radiographs should be evaluated for fracture location, orientation, amount of cortical thickening, and overall geometry of the foot and 5th metatarsal. In elite athletes, the threshold for surgical intervention is decreasing in order to expedite healing and recovery and decrease re-fracture risk. This rationale is based on delayed union rates of 25% to 66%, nonunion rates of 7% to 28%,11 and re-fracture rates of up to 33% associated with nonoperative treatment.12 Nonoperative management is usually not feasible in the competitive athlete, as it typically involves a period of protected weight-bearing in a tall controlled ankle motion (CAM) boot for 6 to 8 weeks with serial radiographs to evaluate healing.
Our preference for surgical intervention involves percutaneous screw fixation with a “high and inside” starting point on fluoroscopy (Figures 2A-2D).
In career athletes, we augment the fracture site using a mixture of bone marrow aspirate concentrate (BMA) (Magellan, Arteriocyte Medical Systems) and demineralized bone matrix (DBM) (Mini Ignite, Wright Medical Technology) injected percutaneously in and around the fracture site under fluoroscopic guidance. Using this technique in a cohort of 25 NFL players treated operatively for Jones fractures, we found that 100% of athletes were able to RTP in the NFL in an average of 9.5 weeks.14 Two patients (7.5%) suffered re-fractures requiring revision surgery with iliac crest bone graft and repeat screw placement with a RTP after 15 weeks. We did not find an association between RTP and re-fracture rate.
The appropriate rehabilitation protocol for Jones fractures after surgery remains controversial and dependent on individual needs and abilities.15,16 For athletes in-season, we recommend a brief period of NWB for 1 to 2 weeks followed by toe-touch weight-bearing in a tall CAM boot for 2 to 4 weeks. After 4 weeks, patients begin gentle exercises on a stationary bike and pool therapy to reduce impact on the fracture site. Low-intensity pulsed ultrasound bone stimulators (Exogen, Bioventus) are frequently used directly over fracture site throughout the postoperative protocol as an adjuvant therapy. If clinically nontender over the fracture site, patients are allowed to begin running in modified protective shoe wear 4 weeks after surgery with an average RTP of 6 to 8 weeks. RTP is determined clinically, as radiographic union may not be evident for 12 to 16 weeks. Useful custom orthoses include turf toe plates with a cushioned lateral column and lateral heel wedge if hindfoot varus is present preoperatively.10 The solid intramedullary screw is left in place permanently.
In our experience, we have found the average re-fracture and nonunion rate to be approximately 8% across all athletes. Our observation that union rates do not appear to be related to RTP times suggests that underlying biology such as Vitamin D deficiency may play a larger role in union rates than previously thought. We have found that most Jones re-fractures occur in the first year after the initial injury, but can occur up to 2.5 years afterwards as well.14 For the management of symptomatic re-fractures and nonunions, the previous screw must be first removed. This can be difficult if the screw is bent or broken, and may require a lateral corticotomy of the metatarsal.
After hardware removal, we advocate open bone grafting of the fracture site using bone from the iliac crest retrieved with a small, percutaneous trephine. Re-fixation should be achieved using a larger, solid screw and postoperative adjuvants may include bone stimulators, Vitamin D and calcium supplemention, and possible teriparatide use (Forteo, Eli Lilly), depending on individual patient profile. In a cohort of 21 elite athletes treated for Jones fracture revision surgery with screw exchange and bone grafting, we found that 100% of patients had computed tomography (CT) evidence of union, with an average RTP of 12.3 weeks.17
Lisfranc Injuries
Lisfranc injuries include any bony or ligamentous damage that involves the tarsometatarsal (TMT) joints. While axial loading of a fixed, plantarflexed foot has traditionally been thought of as the most common mechanism of Lisfranc injury, we have found that noncontact twisting injuries leading to Lisfranc disruption are actually more common among NFL players. This mechanism is similar to noncontact turf toe and results in a purely ligamentous injury. We have found this to be particularly true in the case of defensive ends engaged with offensive linemen in which no axial loading or contact of the foot occurs. Clinically, patients often have painful weight-bearing, inability to perform a single limb heel rise, plantar ecchymosis, and swelling and point tenderness across the bases of the 1st and 2nd metatarsals.
It is critical to obtain comparison weight-bearing radiographs of both feet during initial work-up to look for evidence of instability. Subtle radiographic findings of Lisfranc injury include a bony “fleck” sign, compression fracture of the cuboid, and diastasis between the base of the 1st and 2nd metatarsals and/or medial and middle cuneiforms (Figures 3A, 3B).
The goal of surgical intervention is to obtain and maintain anatomic reduction of all unstable joints in order to restore a normal foot posture. One of the difficulties with Lisfranc injuries is that there are no exact diastasis parameters and individuals should be treated based on symptoms, functional needs, and degree of instability. It has been shown that 5 mm of displacement can have good long-term clinical results in select cases without surgery.18 For surgery, we recommend open reduction to remove interposed soft tissue debris and directly assess the articular surfaces (Figures 4A-4D).
Proximal-medial column Lisfranc injury variants are increasingly common among football players.20 In these injuries, the force of injury extends through the intercuneiform joint and exits out the naviculocuneiform joint, thus causing instability at multiple joints and an unstable 1st ray. Patients often have minimal clinical findings and normal radiographs and stress radiographs. MRI of the foot often reveals edema at the naviculocuneiform joint. Often patients fail to improve with nonoperative immobilization with continued inability to push off from the hallux. Unrecognized or untreated instability will lead to rapid deterioration of the naviculocuneiform joint. Surgical intervention requires a homerun screw and intercuneiform screw. We do not recommend primary arthrodesis in athletes due to significant risk of malunion and nonunion unless severe articular damage is present.
Patients are typically kept NWB in a splint for 2 weeks after surgery followed by NWB in a tall CAM from 3 to 4 weeks postoperative. Progressive weight-bearing and ROM exercises are initiated from 4 to 8 weeks, followed by return to accommodative shoe wear from 10 to 12 weeks. Hardware removal is performed 4 to 6 months after surgery, typically in the off-season to allow for 6 to 8 weeks or protected recovery afterwards. Premature hardware removal can lead to loss of reduction, particularly at the intercuneiform joints. All hardware crossing the TMT joints should be removed, while the homerun screw can be left in place in addition to the intercuneiform screw. RTP in football typically occurs 6 to 7 months after surgery. Final functional outcome is related to the adequacy of initial reduction and severity of the initial injury.21
Syndesmotic Disruption
Syndesmotic injuries comprise 1% to 18% of ankle sprains in the general population, but occur at much higher rates in football due to the increased rotation forces placed on the ankle during cutting and tackling. RTP after syndesmotic injury often takes twice as long when compared to isolated lateral ankle ligamentous injury.22 Missed injuries are common and if not treated properly can lead to chronic ankle instability and posttraumatic ankle arthritis.23 Syndesmotic injury can occur in isolation or with concomitant adjacent bony, cartilaginous, or ligamentous injuries. Therefore, clinical examination and imaging work-up are critical to successful management.
Syndesmotic injuries often result from an external rotation force applied to a hyperdorsiflexed ankle while the foot is planted. This mechanism causes the fibula to externally rotate while translating posteriorly and laterally, resulting in rupture of the anterior inferior tibiofibular ligament (AITFL) first, followed by the deep deltoid ligament, interosseous ligament (IOL), and lastly posterior talofibular ligament.24 Most syndesmotic injuries involve rupture of only the AITFL and IOL.25 Multiple clinical stress tests have been designed to assess syndesmotic stability, including the squeeze test, external rotation stress test, crossed-leg test, and fibula-translation test.26-29 However, no physical examination maneuver has been shown to reliably predict the presence or degree of syndesmotic injury and therefore imaging studies are necessary.30
Initial imaging should include standing radiographs of the affected ankle. An increase in the medial clear space between the medial malleolus and talus can occur with a combined syndesmotic and deltoid disruption. In the case of subtle syndesmotic injuries, contralateral comparison weight-bearing radiographs are recommended. CT and MRI can provide additional information, but these static imaging tests cannot identify instability. Fluoroscopic stress evaluation is beneficial but has a high false-negative rate in low-grade injuries and may not detect partial rupture of the AITFL and IOL.31 It has been shown that malrotation of as much as 30° of external rotation can occur if relying on intraoperative fluoroscopy alone.32 It has been our practice to recommend surgical reduction and stabilization for any syndesmotic injury with documented diastasis or instability seen on imaging and/or arthroscopy.
Nonoperative treatment is indicated for truly stable grade I syndesmotic injuries. This involves rest and immobilization followed by a progressive rehabilitation program consisting of stretching, strengthening, and proprioceptive exercises.33 After 1 week of protected weight-bearing in a cast or tall CAM boot, progression to full weight-bearing should occur over the following week. Active-assisted ankle ROM exercises and light proprioceptive training should then be initiated followed by sport-specific exercises 2 to 3 weeks after injury.
Arthroscopy can be a valuable diagnostic tool in the setting of subtle syndesmotic injury with negative radiographs, positive MRI for edema, and a protracted recovery course with vague pain (Figures W5A-W5E).
Implants are placed above the true syndesmotic joint (at least 15 mm above the tibial plafond) angled 30° posterior to anterior to follow the normal relationship of the fibula to the distal tibia in the incisura. Typically 2 suture-buttons are used, with the devices placed in a divergent fashion. We highly recommend the use of a fibular buttress plate with button placement in individuals returning to contact activity. This construct increases surface area distribution while preventing stress risers and the risk of fibula fractures. In a cadaver model with deliberate syndesmotic malreduction, suture-button stabilization resulted in decreased postoperative displacement as opposed to conventional screw fixation.34 Therefore, dynamic syndesmotic fixation may help to decrease the negative sequelae of iatrogenic clamp malreduction. Postoperative rehabilitation involves NWB in a cast or tall CAM boot for 4 weeks followed by ankle ROM exercises and progressive weight-bearing and physical therapy. Patients are transitioned to a lace-up ankle brace and athletic shoe from 6 to 12 weeks postoperative with increasing activity. Running and jumping is permitted 4 months after surgery with RTP typically at 6 to 7 months. Athletes who have had surgical stabilization for documented instability without any diastasis may engage in a more rapid recovery and RTP as symptoms and function allow.
Deltoid Complex Avulsion
Missed or neglected deltoid ligament injuries can lead to progressive chondral injury and joint degeneration. These injuries are often subtle and difficult to diagnose. An inability to perform a single limb heel rise, persistent pain with activity, and lack of normal functional improvement despite appropriate care are indicators of subtle ligament instability. These injuries often require an examination under anesthesia with combined ankle arthroscopy. Valgus stress testing of the ankle while directly visualizing the deltoid ligament from the anterolateral portal can reveal medial laxity in addition to potential osteochondral lesions along the anterolateral talar dome.
In American football players, we have observed that infolding and retraction of an avulsed superficial deltoid ligament complex after an ankle fracture, Maisonneuve injury, or severe high ankle sprain can be a source of persistent increased medial clear space, malreduction, and postoperative pain and medial instability. We have found that there is often complete avulsion of the superficial deltoid complex off the proximal aspect of the medial malleolus during high-energy ankle fractures in football players that is amenable to direct repair to bone (Figures W6A-W6E).
During surgical repair, an incision is made along the anterior aspect of the medial malleolus and the superficial deltoid ligament complex can often be found flipped and interposed in the medial gutter. A rongeur is used to create a bleeding cancellous bone surface for soft-tissue healing and 1 to 2 suture anchors are used to repair and imbricate the deltoid ligament complex back to the medial malleolus. The goal of these sutures is to repair the tibionavicular and tibial spring ligaments back to the medial malleolus. We believe that superficial deltoid complex avulsion during high-energy ankle fractures is a distinct injury pattern that should be recognized and may benefit from primary open repair.
We currently open explore every deltoid ligament complex in athletes with unstable syndesmotic injuries, as we believe that deltoid avulsion injuries are underrecognized and do not heal in an anatomic fashion if left alone. Postoperative recovery follows the same immobilization, progressive weight-bearing, and physical therapy protocol as that for syndesmotic disruption.
Achilles Ruptures
Acute midsubstance Achilles tendon ruptures are an increasingly common injury in patients 30 to 50 years of age, with more than 50% of all injuries occurring during basketball.36,37 Among NFL players, we have found that Achilles ruptures tend to occur at a higher rate during training camp, when athletes are deconditioned and quickly returning to explosive push-off activities. Physical examination should include a Thompson test, palpation of a gap within the tendon, and evaluation of resting ankle dorsiflexion in the affected extremity in the prone position with the knees bent. Lateral radiographs should be analyzed for the presence of a bony avulsion fragment indicative of an insertional avulsion injury or midsubstance calcium deposition reflecting chronic Achilles tendinosis, as both of these conditions will change surgical management. MRI is not recommended with acute midsubstance ruptures but may be helpful in the case of chronic ruptures or more proximal tears of the musculotendinous junction.
The management of acute midsubstance Achilles tendon ruptures is controversial, with no general consensus in the literature regarding nonoperative treatment, surgical repair, and ideal repair technique.36,38-42 American Academy of Orthopaedic Surgeons clinical practice guidelines report moderate evidence that nonoperative treatment of Achilles tendon ruptures has lower wound healing complications but higher rates of re-rupture.38,39 Additionally, limited incision approaches have been found to have fewer overall complications compared with traditional open repair. In an effort to reduce the incidence of postoperative wound complications while improving functional recovery, modern repair techniques focus on a limited incision repair using percutaneous suture insertion and management (PARS Achilles Jig System, Arthrex).36 The limited incision technique utilizes a 2-cm transverse incision and non-disposable jig with divergent needle passes and locking suture fixation options to secure and fixate both tendon ends with minimal dissection of skin, subcutaneous tissue, and paratenon. Limited incision repair is ideally performed within 2 weeks of the injury to ensure that both tendon ends are easy to identify, mobilize, and repair. An open repair is generally recommended for midsubstance ruptures more than 4 weeks old and cases of insertional rupture and Achilles tendinopathy.
In a cohort of 9 NFL players treated for midsubstance Achilles ruptures using the PARS technique, we found no re-ruptures, no wound complications, and no sural nerve issues after surgery.43 A comparative review of 270 cases of operatively treated Achilles tendon ruptures (101 PARS, 169 traditional open repair) showed that the PARS group had significantly shorter operative times and a higher number of patients able to return to baseline physical activities by 5 months compared to open repair.36 Although not statistically significant, the overall PARS complication rate was 5% while the open complication rate was 11%. The PARS group had no cases of sural neuritis or deep infection requiring reoperation. We currently use a limited incision technique for all acute midsubstance Achilles ruptures in athletes regardless of sport, patient size, or position played.
During surgery, a 2-cm transverse incision is made over the gap in the Achilles tendon and dissection is carried down to the rupture site with minimal manipulation of the skin (Figures 5A-5F).
A key aspect of postoperative recovery is avoiding excessive ankle dorsiflexion while the tendon is healing during the first 4 weeks after surgery, as this can lead to an elongated tendon with loss of push-off strength. Patients are kept in a plantarflexion splint NWB for 2 weeks after surgery. If the incision is healed at 2 weeks, sutures are removed and patients are transitioned into a NWB tall CAM boot for 2 weeks with gentle ankle ROM exercises. If there is any concern regarding wound healing status, sutures are maintained for an additional 1 to 2 weeks.
From 4 to 8 weeks after surgery, progressive weight-bearing with continued ankle ROM exercises is initiated with peel-away heel lifts (~2 cm thick total, 3 layers). Each layer of the heel lift is gradually removed as pain allows every 2 to 3 days with the goal of being full weight-bearing with the foot flat at 6 weeks postoperative. Physical therapy focusing on ankle ROM and gentle Achilles stretching and strengthening is also started 6 weeks after surgery. From 8 to 12 weeks postoperative, patients are transitioned out of the tall CAM boot into normal, accommodative shoe wear with full weight-bearing. We avoid ankle dorsiflexion past neutral until 12 weeks after surgery, as overlengthening of the Achilles complex and the subsequent loss of push-off power can be devastating to running athletes. Activity levels are increased as tolerated, with no running or jumping from 12 to 16 weeks with full release to all activities after 16 weeks. RTP often takes 5 to 6 months after surgery, depending on the position played.
Am J Orthop. 2016;45(6):358-367. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Canale ST, Cantler ED Jr, Sisk TD, Freeman BL 3rd. A chronicle of injuries of an American intercollegiate football team. Am J Sports Med. 1981;9(6):384-389.2. Robey JM, Blyth CS, Mueller FO. Athletic injuries. Application of epidemiologic methods. JAMA. 1971;217(2):184-189.
3. Saal JA. Common American football injuries. Sports Med. 1991;12(2):132-147.
4. Thompson N, Halpern B, Curl WW, et al. High school football injuries: evaluation. Am J Sports Med. 1987;15(2):117-124.
5. Kaplan LD, Jost PW, Honkamp N, Norwig J, West R, Bradley JP. Incidence and variance of foot and ankle injuries in elite college football players. Am J Orthop. 2011;40(1):40-44.
6. DeLee JC, Farney WC. Incidence of injury in Texas high school football. Am J Sports Med. 1992;20(5):575-580.
7. Brophy RH, Barnes R, Rodeo SA, Warren RF. Prevalence of musculoskeletal disorders at the NFL Combine--trends from 1987 to 2000. Med Sci Sports Exerc. 2007;39(1):22-27.
8. Bowers KD Jr, Martin RB. Turf-toe: a shoe-surface related football injury. Med Sci Sports. 1976;8(2):81-83.
9. McCormick JJ, Anderson RB. Turf toe: anatomy, diagnosis, and treatment. Sports Health. 2010;2(6):487-494.
10. Raikin SM, Slenker N, Ratigan B. The association of a varus hindfoot and fracture of the fifth metatarsal metaphyseal-diaphyseal junction: the Jones fracture. Am J Sports Med. 2008;36(7):1367-1372.
11. Title CI, Katchis SD. Traumatic foot and ankle injuries in the athlete. Orthop Clin North Am. 2002;33(3):587-598.
12. Quill GE Jr. Fractures of the proximal fifth metatarsal. Orthop Clin North Am. 1995;26(2):353-361.
13. Nunley JA, Glisson RR. A new option for intramedullary fixation of Jones fractures: the Charlotte Carolina Jones Fracture System. Foot Ankle Int. 2008;29(12):1216-1221.
14. Lareau CR, Hsu AR, Anderson RB. Return to play in National Football League players after operative Jones fracture treatment. Foot Ankle Int. 2016;37(1):8-16.
15. Larson CM, Almekinders LC, Taft TN, Garrett WE. Intramedullary screw fixation of Jones fractures. Analysis of failure. Am J Sports Med. 2002;30(1):55-60.
16. Portland G, Kelikian A, Kodros S. Acute surgical management of Jones’ fractures. Foot Ankle Int. 2003;24(11):829-833.
17. Hunt KJ, Anderson RB. Treatment of Jones fracture nonunions and refractures in the elite athlete: outcomes of intramedullary screw fixation with bone grafting. Am J Sports Med. 2011;39(9):1948-1954.
18. Nunley JA, Vertullo CJ. Classification, investigation, and management of midfoot sprains: Lisfranc injuries in the athlete. Am J Sports Med. 2002;30(6):871-878.
19. Alberta FG, Aronow MS, Barrero M, Diaz-Doran V, Sullivan RJ, Adams DJ. Ligamentous Lisfranc joint injuries: a biomechanical comparison of dorsal plate and transarticular screw fixation. Foot Ankle Int. 2005;26(6):462-473.
20. Ardoin GT, Anderson RB. Subtle Lisfranc injury. Tech Foot Ankle Surg. 2010;9(3):100-106.
21. Kuo RS, Tejwani NC, Digiovanni CW, et al. Outcome after open reduction and internal fixation of Lisfranc joint injuries. J Bone Joint Surg Am. 2000;82-A(11):1609-1618.
22. Wright RW, Barile RJ, Surprenant DA, Matava MJ. Ankle syndesmosis sprains in national hockey league players. Am J Sports Med. 2004;32(8):1941-1945.
23. Williams GN, Jones MH, Amendola A. Syndesmotic ankle sprains in athletes. Am J Sports Med. 2007;35(7):1197-1207.
24. Beumer A, Valstar ER, Garling EH, et al. Effects of ligament sectioning on the kinematics of the distal tibiofibular syndesmosis: a radiostereometric study of 10 cadaveric specimens based on presumed trauma mechanisms with suggestions for treatment. Acta Orthop. 2006;77(3):531-540.
25. McCollum GA, van den Bekerom MP, Kerkhoffs GM, Calder JD, van Dijk CN. Syndesmosis and deltoid ligament injuries in the athlete. Knee Surg Sports Traumatol Arthrosc. 2013;21(6):1328-1337.
26. Boytim MJ, Fischer DA, Neumann L. Syndesmotic ankle sprains. Am J Sports Med. 1991;19(3):294-298.
27. Nussbaum ED, Hosea TM, Sieler SD, Incremona BR, Kessler DE. Prospective evaluation of syndesmotic ankle sprains without diastasis. Am J Sports Med. 2001;29(1):31-35.
28. Kiter E, Bozkurt M. The crossed-leg test for examination of ankle syndesmosis injuries. Foot Ankle Int. 2005;26(2):187-188.
29. Beumer A, van Hemert WL, Swierstra BA, Jasper LE, Belkoff SM. A biomechanical evaluation of clinical stress tests for syndesmotic ankle instability. Foot Ankle Int. 2003;24(4):358-363.
30. Amendola A, Williams G, Foster D. Evidence-based approach to treatment of acute traumatic syndesmosis (high ankle) sprains. Sports Med Arthrosc. 2006;14(4):232-236.
31. Beumer A, Valstar ER, Garling EH, et al. External rotation stress imaging in syndesmotic injuries of the ankle: comparison of lateral radiography and radiostereometry in a cadaveric model. Acta Orthop Scand. 2003;74(2):201-205.
32. Marmor M, Hansen E, Han HK, Buckley J, Matityahu A. Limitations of standard fluoroscopy in detecting rotational malreduction of the syndesmosis in an ankle fracture model. Foot Ankle Int. 2011;32(6):616-622.
33. Williams GN, Allen EJ. Rehabilitation of syndesmotic (high) ankle sprains. Sports Health. 2010;2(6):460-470.
34. Westermann RW, Rungprai C, Goetz JE, Femino J, Amendola A, Phisitkul P. The effect of suture-button fixation on simulated syndesmotic malreduction: a cadaveric study. J Bone Joint Surg Am. 2014;96(20):1732-1738.
35. Hsu AR, Lareau CR, Anderson RB. Repair of acute superficial deltoid complex avulsion during ankle fracture fixation in National Football League players. Foot Ankle Int. 2015;36(11):1272-1278.
36. Hsu AR, Jones CP, Cohen BE, Davis WH, Ellington JK, Anderson RB. Clinical outcomes and complications of percutaneous Achilles repair system versus open technique for acute achilles tendon ruptures. Foot Ankle Int. 2015;36(11):1279-1286.
37. Raikin SM, Garras DN, Krapchev PV. Achilles tendon injuries in a United States population. Foot Ankle Int. 2013;34(4):475-480.
38. Chiodo CP, Glazebrook M, Bluman EM, et al. American Academy of Orthopaedic Surgeons clinical practice guideline on treatment of achilles tendon rupture. J Bone Joint Surg Am. 2010;92(14):2466-2468.
39. Chiodo CP, Glazebrook M, Bluman EM, et al. Diagnosis and treatment of acute achilles tendon rupture. J Am Acad Orthop Surg. 2010;18(8):503-510.
40. Khan RJ, Fick D, Keogh A, Crawford J, Brammar T, Parker M. Treatment of acute achilles tendon ruptures. A meta-analysis of randomized, controlled trials. J Bone Joint Surg Am. 2005;87(10):2202-2210.
41. Renninger CH, Kuhn K, Fellars T, Youngblood S, Bellamy J. Operative and nonoperative management of achilles tendon ruptures in active duty military population. Foot Ankle Int. 2016;37(3):269-273.
42. Khan RJ, Carey Smith RL. Surgical interventions for treating acute achilles tendon ruptures. Cochrane Database Syst Rev. 2010;(9):CD003674.
43. McCullough KA, Shaw CM, Anderson RB. Mini-open repair of achilles rupture in the national football league. J Surg Orthop Adv. 2014;23(4):179-183.
1. Canale ST, Cantler ED Jr, Sisk TD, Freeman BL 3rd. A chronicle of injuries of an American intercollegiate football team. Am J Sports Med. 1981;9(6):384-389.2. Robey JM, Blyth CS, Mueller FO. Athletic injuries. Application of epidemiologic methods. JAMA. 1971;217(2):184-189.
3. Saal JA. Common American football injuries. Sports Med. 1991;12(2):132-147.
4. Thompson N, Halpern B, Curl WW, et al. High school football injuries: evaluation. Am J Sports Med. 1987;15(2):117-124.
5. Kaplan LD, Jost PW, Honkamp N, Norwig J, West R, Bradley JP. Incidence and variance of foot and ankle injuries in elite college football players. Am J Orthop. 2011;40(1):40-44.
6. DeLee JC, Farney WC. Incidence of injury in Texas high school football. Am J Sports Med. 1992;20(5):575-580.
7. Brophy RH, Barnes R, Rodeo SA, Warren RF. Prevalence of musculoskeletal disorders at the NFL Combine--trends from 1987 to 2000. Med Sci Sports Exerc. 2007;39(1):22-27.
8. Bowers KD Jr, Martin RB. Turf-toe: a shoe-surface related football injury. Med Sci Sports. 1976;8(2):81-83.
9. McCormick JJ, Anderson RB. Turf toe: anatomy, diagnosis, and treatment. Sports Health. 2010;2(6):487-494.
10. Raikin SM, Slenker N, Ratigan B. The association of a varus hindfoot and fracture of the fifth metatarsal metaphyseal-diaphyseal junction: the Jones fracture. Am J Sports Med. 2008;36(7):1367-1372.
11. Title CI, Katchis SD. Traumatic foot and ankle injuries in the athlete. Orthop Clin North Am. 2002;33(3):587-598.
12. Quill GE Jr. Fractures of the proximal fifth metatarsal. Orthop Clin North Am. 1995;26(2):353-361.
13. Nunley JA, Glisson RR. A new option for intramedullary fixation of Jones fractures: the Charlotte Carolina Jones Fracture System. Foot Ankle Int. 2008;29(12):1216-1221.
14. Lareau CR, Hsu AR, Anderson RB. Return to play in National Football League players after operative Jones fracture treatment. Foot Ankle Int. 2016;37(1):8-16.
15. Larson CM, Almekinders LC, Taft TN, Garrett WE. Intramedullary screw fixation of Jones fractures. Analysis of failure. Am J Sports Med. 2002;30(1):55-60.
16. Portland G, Kelikian A, Kodros S. Acute surgical management of Jones’ fractures. Foot Ankle Int. 2003;24(11):829-833.
17. Hunt KJ, Anderson RB. Treatment of Jones fracture nonunions and refractures in the elite athlete: outcomes of intramedullary screw fixation with bone grafting. Am J Sports Med. 2011;39(9):1948-1954.
18. Nunley JA, Vertullo CJ. Classification, investigation, and management of midfoot sprains: Lisfranc injuries in the athlete. Am J Sports Med. 2002;30(6):871-878.
19. Alberta FG, Aronow MS, Barrero M, Diaz-Doran V, Sullivan RJ, Adams DJ. Ligamentous Lisfranc joint injuries: a biomechanical comparison of dorsal plate and transarticular screw fixation. Foot Ankle Int. 2005;26(6):462-473.
20. Ardoin GT, Anderson RB. Subtle Lisfranc injury. Tech Foot Ankle Surg. 2010;9(3):100-106.
21. Kuo RS, Tejwani NC, Digiovanni CW, et al. Outcome after open reduction and internal fixation of Lisfranc joint injuries. J Bone Joint Surg Am. 2000;82-A(11):1609-1618.
22. Wright RW, Barile RJ, Surprenant DA, Matava MJ. Ankle syndesmosis sprains in national hockey league players. Am J Sports Med. 2004;32(8):1941-1945.
23. Williams GN, Jones MH, Amendola A. Syndesmotic ankle sprains in athletes. Am J Sports Med. 2007;35(7):1197-1207.
24. Beumer A, Valstar ER, Garling EH, et al. Effects of ligament sectioning on the kinematics of the distal tibiofibular syndesmosis: a radiostereometric study of 10 cadaveric specimens based on presumed trauma mechanisms with suggestions for treatment. Acta Orthop. 2006;77(3):531-540.
25. McCollum GA, van den Bekerom MP, Kerkhoffs GM, Calder JD, van Dijk CN. Syndesmosis and deltoid ligament injuries in the athlete. Knee Surg Sports Traumatol Arthrosc. 2013;21(6):1328-1337.
26. Boytim MJ, Fischer DA, Neumann L. Syndesmotic ankle sprains. Am J Sports Med. 1991;19(3):294-298.
27. Nussbaum ED, Hosea TM, Sieler SD, Incremona BR, Kessler DE. Prospective evaluation of syndesmotic ankle sprains without diastasis. Am J Sports Med. 2001;29(1):31-35.
28. Kiter E, Bozkurt M. The crossed-leg test for examination of ankle syndesmosis injuries. Foot Ankle Int. 2005;26(2):187-188.
29. Beumer A, van Hemert WL, Swierstra BA, Jasper LE, Belkoff SM. A biomechanical evaluation of clinical stress tests for syndesmotic ankle instability. Foot Ankle Int. 2003;24(4):358-363.
30. Amendola A, Williams G, Foster D. Evidence-based approach to treatment of acute traumatic syndesmosis (high ankle) sprains. Sports Med Arthrosc. 2006;14(4):232-236.
31. Beumer A, Valstar ER, Garling EH, et al. External rotation stress imaging in syndesmotic injuries of the ankle: comparison of lateral radiography and radiostereometry in a cadaveric model. Acta Orthop Scand. 2003;74(2):201-205.
32. Marmor M, Hansen E, Han HK, Buckley J, Matityahu A. Limitations of standard fluoroscopy in detecting rotational malreduction of the syndesmosis in an ankle fracture model. Foot Ankle Int. 2011;32(6):616-622.
33. Williams GN, Allen EJ. Rehabilitation of syndesmotic (high) ankle sprains. Sports Health. 2010;2(6):460-470.
34. Westermann RW, Rungprai C, Goetz JE, Femino J, Amendola A, Phisitkul P. The effect of suture-button fixation on simulated syndesmotic malreduction: a cadaveric study. J Bone Joint Surg Am. 2014;96(20):1732-1738.
35. Hsu AR, Lareau CR, Anderson RB. Repair of acute superficial deltoid complex avulsion during ankle fracture fixation in National Football League players. Foot Ankle Int. 2015;36(11):1272-1278.
36. Hsu AR, Jones CP, Cohen BE, Davis WH, Ellington JK, Anderson RB. Clinical outcomes and complications of percutaneous Achilles repair system versus open technique for acute achilles tendon ruptures. Foot Ankle Int. 2015;36(11):1279-1286.
37. Raikin SM, Garras DN, Krapchev PV. Achilles tendon injuries in a United States population. Foot Ankle Int. 2013;34(4):475-480.
38. Chiodo CP, Glazebrook M, Bluman EM, et al. American Academy of Orthopaedic Surgeons clinical practice guideline on treatment of achilles tendon rupture. J Bone Joint Surg Am. 2010;92(14):2466-2468.
39. Chiodo CP, Glazebrook M, Bluman EM, et al. Diagnosis and treatment of acute achilles tendon rupture. J Am Acad Orthop Surg. 2010;18(8):503-510.
40. Khan RJ, Fick D, Keogh A, Crawford J, Brammar T, Parker M. Treatment of acute achilles tendon ruptures. A meta-analysis of randomized, controlled trials. J Bone Joint Surg Am. 2005;87(10):2202-2210.
41. Renninger CH, Kuhn K, Fellars T, Youngblood S, Bellamy J. Operative and nonoperative management of achilles tendon ruptures in active duty military population. Foot Ankle Int. 2016;37(3):269-273.
42. Khan RJ, Carey Smith RL. Surgical interventions for treating acute achilles tendon ruptures. Cochrane Database Syst Rev. 2010;(9):CD003674.
43. McCullough KA, Shaw CM, Anderson RB. Mini-open repair of achilles rupture in the national football league. J Surg Orthop Adv. 2014;23(4):179-183.
Concussions in American Football
Football is an important component of American culture, with approximately 3 million youth athletes, 1.1 million high school athletes, and 100,000 college athletes participating each year.1 Participation in football provides athletes with physical, social, psychological, and academic benefits. Despite these benefits, widespread focus has been placed on the safety of football due to the risk for sport-related concussion (SRC) and potentially long-term effects; however, little recognition has been given to the advancements in concussion management across time and occurrence of concussions during most life activities. Although it is reasonable for concerns to be presented, it is important to better understand SRC and the current factors leading to prolonged recoveries, increased risk for injury, and potentially long-term effects.
What Is a Concussion?
Concussions occur after sustaining direct or indirect injury to the head or other parts of the body, as long as the injury force is transmitted to the head. Athletes often experience physical, cognitive, emotional, and sleep-related symptoms post-concussion secondary to an “energy crisis” within the brain.2 The energy crisis occurs as the result of transient neurological dysfunction triggered by changes in the brain (eg, release of neurotransmitters, impaired axonal function).2,3 Concussion is undetectable with traditional imaging; however, advanced imaging techniques (eg, diffuse tensor imaging) have shown progress in assessing axonal injury.3 Symptom duration post-concussion is highly variable due to individual differences; a recent study showed recovery took 3 to 4 weeks for memory and symptoms.4,5
Previous Concussion Management
Identification techniques and return-to-play guidelines for concussion have significantly changed across time. In the past, concussion grading scales were utilized for diagnosis and return to play was possible within the same contest.6,7 It has since been recognized that initial concussion severity makes it difficult to predict recovery.3 For example, research revealed memory decline and increased symptoms 36 hours post-injury for athletes with a grade 1 concussion (ie, transient confusion, no loss of consciousness, concussion symptoms or mental status changes that resolve within 15 minutes of injury) compared to baseline.7 Another study found duration of mental status changes to be related to slower symptom resolution and memory impairment 36 hours to 7 days post-injury.6 Consequently, return to play within the same contest was likely too liberal. Guidelines today recommend immediate removal from play with suspected SRC. Nevertheless, the “play through pain” culture has led athletes to continue playing after SRC, contributing to prolonged recoveries and potentially long-term effects.
Current Concussion Management: Continued Concerns and Areas of Improvement
Despite increased awareness of concussions, recent estimates revealed high rates (ie, 27:1 ratio for general players) of underreporting in college football, particularly amongst offensive linemen.8 Researchers have studied recovery implications for remaining in play, with one study revealing a 2.2 times greater risk for prolonged recovery in college athletes with delayed vs immediate removal.9 Another similar study discovered an 8.8 times greater risk for prolonged recovery in adolescent and young adult athletes not removed vs removed from play.10 Further analysis found remaining in play to be the greatest risk factor for prolonged recovery compared to other previously studied risk factors (eg, age, sex, posttraumatic migraine).10 Additionally, significant differences in neurocognitive data were seen between the “removed” and “not removed” groups for verbal memory, visual memory, processing speed, and reaction time at 1 to 7 days and 8 to 30 days.10 The recovery implications of remaining in play and the additional risk for second impact syndrome (SIS), or repeat concussion when recovering from another injury, emphasizes the need for further education efforts amongst athletes to encourage immediate reporting of injury.11
Sideline Assessment
Sideline assessment has become a vital component of concussion management to rule out concussion and/or significant injury other than concussion. Assessment should include observation, cognitive/balance testing, neurologic examination, and possible exertion testing to ensure a comprehensive evaluation of all areas of potential dysfunction.12 Indications for emergency department evaluation include suspicion for cervical spine injury, intracranial hemorrhage, or skull fracture as well as prolonged loss of consciousness, high-risk mechanisms, posttraumatic seizure(s), and/or significant worsening of symptoms.12
Observation
On the sideline, it is important to identify any immediate signs of injury (ie, loss of consciousness, anterograde/retrograde amnesia, and disorientation/confusion). Since immediate signs are not always present, it is important to be aware of the most commonly reported symptoms, including headache, difficulty concentrating, fatigue, drowsiness, and dizziness.13 If symptoms are not reported by the athlete, balance problems, lack of coordination, increased emotionality, and difficulty following instructions may be observed during play.12
On-Field Assessment
Cognitive and balance testing are essential in determining if an athlete has sustained a concussion. Immediate declines in memory, concentration abilities, and balance abilities are common. Given limitations in administering long testing batteries on the sideline, brief standardized tests such as the Standardized Assessment of Concussion (SAC), Balance Error Scoring System (BESS), and Sport Concussion Assessment Tool (SCAT) are commonly utilized. Identification of cognitive and/or balance abnormalities can help the athlete recognize deficits following injury.12 Balance problems are experienced due to abnormalities in sensory organization and generally resolve during the acute recovery period.14,15 Cognitive difficulties typically persist longer than balance problems, though duration varies widely.
Neurologic Evaluation
A neurologic evaluation including cranial nerve testing and evaluation of motor-sensory function (ie, assessment for the strength and sensation of upper and lower extremities) is important to identify focal deficits (ie, sensation changes, loss of fine motor control) indicative of serious intracranial pathology.12 Additionally, clinicians have suggested inclusion of vestibular and oculomotor assessments due to frequent dysfunction post-concussion.12,15,16 Examination of the vestibular/oculomotor systems through tools such as the Vestibular/Ocular Motor Screening (VOMS) assessment (assesses both the vestibular and oculomotor systems) and King-Devick Test (primarily assesses saccadic eye movements) can elicit symptoms that may not present immediately. If assessment appears normal, exertion testing can be utilized to determine if symptoms are provoked through physical exercise that should include cardio, dynamic, and sport-specific activities to stress the vestibular system.12
Risk Factors for Injury and Prolonged Recovery
Medical professionals must consider the presence of risk factors when managing concussion in order to make appropriate treatment recommendations and return-to-play decisions. Research has demonstrated the role of female gender, learning disability, attention-deficit/hyperactivity disorder, psychiatric history, young age, motion sickness, sleep problems, somatization, concussion history, on-field dizziness, posttraumatic migraine, and fogginess in increased risk for injury and/or prolonged recovery.17-25 Additionally, athletes with ongoing symptoms from a previous injury are at risk for sustaining another injury.
Acute Home Concussion Management
Although strict rest has been recommended post-concussion, recent research evaluating strict rest vs usual care for adolescents revealed greater symptom reports and longer recovery periods for the strict rest group.26 Based on these findings and emphasis for regulation within the migraine literature (due to the common pathophysiology between migraine and concussion27), we recommend that athletes follow a regulated daily schedule post-concussion including: 1) regular sleep-wake schedule with avoidance of naps, 2) regular meals, 3) adequate fluid hydration, 4) light noncontact physical activity (ie, walking, with progressions recommended by a physician), and 5) stress management techniques. Use of these strategies immediately can help in preventing against increased symptoms and stress, and decreases the need for medication in select cases. Additionally, over-the-counter medications should be limited to 2 to 3 doses per week to avoid rebound headaches.28
In-Office Concussion Management
Athletes diagnosed with SRC will experience different symptoms based on the injury mechanism, risk factors, and management approach. Comprehensive evaluation should include assessment of risk factors, injury details, symptoms, neurocognitive functioning, vestibular/oculomotor dysfunction, tolerance of physical exertion, balance functioning, and cervical spine integrity (if necessary).29,30 Due to individual differences and the heterogeneous symptom profiles, concussion management must move beyond a “one size fits all” approach to avoid nonspecific treatment strategies and consequently prolonged recoveries.29 Clinicians and researchers at University of Pittsburgh Medical Center have identified 6 concussion clinical profiles (ie, vestibular, ocular, posttraumatic migraine, cervical, anxiety/mood, and cognitive/fatigue) that are generally identifiable 48 hours after injury.29,30 Identification of the clinical profile(s) through a comprehensive evaluation guides the development of individualized treatment plans and targeted rehabilitation strategies.29,30
Vestibular. The vestibular system is responsible for stabilizing vision while the head moves and balance control.15 Athletes can experience central and/or peripheral vestibular dysfunction to include benign paroxysmal positional vertigo (BPPV), visual motion sensitivity, vestibular ocular reflex impairment, and balance impairment.30,31 Symptoms typically include dizziness, impaired balance, blurry vision, difficulty focusing, and environmental sensitivity.15,29,30 Potential treatment options include vestibular rehabilitation, exertion therapy, and school/work accommodations.
Ocular. The oculomotor system is responsible for control of eye movements. Athletes can experience many different posttraumatic vision changes, including convergence problems, eye-tracking difficulties, refractive error, difficulty with pursuits/saccades, and accommodation insufficiency. Symptoms typically include light sensitivity, blurred vision, double vision, headaches, fatigue, and memory difficulties.15,29,30 Potential treatment options include vision therapy, vestibular rehabilitation, and school/work accommodations.32
Posttraumatic Migraine. Headache, the most common post-concussion symptom, can persist and meet criteria for posttraumatic migraine (ie, unilateral headache with accompanying nausea and/or photophobia and phonophobia).29,30,33 Implementation of a routine schedule, daily physical activity, exertion therapy, pharmacologic intervention, and school/work accommodations are potential treatment options.
Cervical. The cervical spine can be injured during whiplash-type injuries. Therefore, determining the location, onset, and typical exacerbations of pain can be helpful in identifying cervical involvement.29,30 Symptoms typically include headaches, neck pain, numbness, and tingling. Evaluation and therapy by a certified physical therapist and pharmacologic intervention (eg, muscle relaxants) are potential treatment options. 29,30
Anxiety/Mood. Anxiety, or worry and fear about everyday situations, is common post-concussion and can sometimes be related to ongoing vestibular impairment. Symptoms typically include ruminative thoughts, avoidance of specific situations, hypervigilance, feelings of being overwhelmed, and difficulty falling asleep.29,30 Potential treatment options include implementation of a routine schedule, exposure to provocative situations, psychotherapy, pharmacologic intervention, and school/work accommodations.34
Cognitive/Fatigue. A global concussion factor (including cognitive, fatigue, and migraine symptoms) has been identified within 1 to 7 days of injury. Although this factor of symptoms generally resolves during the acute recovery period, it persists in select cases.13 Symptoms typically include fatigue, decreased energy levels, nonspecific headaches, potential sleep disruption, increased symptoms towards the end of the day, difficulty concentrating, and increased headache with cognitive activities.29,30,35 Routine schedule, daily physical activity, exertion therapy, pharmacologic intervention (eg, amantadine), and school/work accommodations are potential treatment options.30
Conclusion
Advancements in SRC management warrant change in the conversations regarding concussion in football. Specifically, conversations should address the current understanding of concussion and improvements in the safety of football through stricter concussion guidelines, detailed sideline evaluations, recognition of risk factors, improved acute management, and identification of concussion profiles that help to direct individualized treatment plans and targeted rehabilitation strategies. The biggest concerns related to concussions in football include underreporting of injury, premature return to play, and receiving routine rather than individualized treatment. Therefore, to further improve the safety of football and management of concussion it is essential that future efforts focus on the following 6 areas:
Education: Improved understanding of concussion is imperative to reducing poor outcomes and widespread concerns.
Immediate reporting: Reporting of concussion must be expected and encouraged through consistent responses by coaches to reduce underreporting and fear of reporting in athletes.
Prevention techniques: Athletes must be taught proper form and playing techniques to reduce the risk for concussion. Proper form and technique should be incentivized.
Targeted treatment: Individualized treatment plans and targeted rehabilitation strategies must be developed based on the identified clinical profile(s) to avoid nonspecific treatment recommendations.
Multidisciplinary treatment teams: Given the heterogeneous symptoms profiles and need for care provided by different medical specialties, multidisciplinary teams are essential.
Remain current: With the progress in understanding concussion, providers must remain vigilant of future advances in concussion management to further improve the safety of football.
Am J Orthop. 2016;45(6):352-356. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Dompier TP, Kerr ZY, Marshall SW, et al. Incidence of concussion during practice and games in youth, high school, and collegiate American football players. JAMA Pediatrics. 2015;169(7):659-665.
2. Giza C, Hovda D. The new neurometabolic cascade of concussion
3. Barkhoudarian G, Hovda DA, Giza CC. The molecular pathophysiology of concussive brain injury - an update. Phys Med Rehabil Clin N Am. 2016;27:373-393.
4. Henry L, Elbin R, Collins M, Marchetti G, Kontos A. Examining recovery trajectories after sport-related concussion with a multimodal clinical assessment approach. Neurosurgery. 2016;78(2):232-241.
5. McCrory P, Meeuwisse WH, Aubry M, et al. Consensus statement on concussion in sport: the 4th International Conference on Concussion in Sport held in Zurich, November 2012. Brit J Sports Med. 2013;47(5):250-258.
6. Lovell MR, Collins MW, Iverson GL, et al. Recovery from mild concussion in high school athletes. J Neurosurg. 2003;98(2):296-301.
7. Lovell MR, Collins MW, Iverson GL, Johnston KM, Bradley JP. Grade 1 or “ding” concussions in high school athletes. Am J Sports Med. 2004;32(1):47-54.
8. Baugh CM, Kroshus E, Daneshvar DH, Filali NA, Hiscox MJ, Glantz LH. Concussion management in United States college sports: compliance with National Collegiate Athletic Association concussion policy and areas for improvement. Am J Sports Med. 2015;43(1):47-56.
9. Asken BM, McCrea MA, Clugston JR, Snyder AR, Houck ZM, Bauer RM. “Playing through it”: Delayed reporting and removal from athletic activity after concussion predicts prolonged recovery. J Athl Train. 2016;51(4):329-335.
10. Elbin RJ, Sufrinko A, Schatz P, et al. Athletes that continue to play with concussion demonstrate worse recovery outcomes than athletes immediately removed from play. J Pediatr. In press.
11. Signoretti S, Lazzarino G, Tavazzi B, Vagnozzi R. The pathophysiology of concussion. PM R. 2011;3(10 Suppl 2):S359-S368.
12. Bloom J, Blount JG. Sideline evaluation of concussion. UpToDate. 2016. http://www.uptodate.com/contents/sideline-evaluation-of-concussion. Accessed July 13, 2016.
13. Kontos AP, Elbin RJ, Schatz P, et al. A revised factor structure for the post-concussion symptom scale: baseline and postconcussion factors. Am J Sports Med. 2012;40(10):2375-2384.
14. Guskiewicz KM, Ross SE, Marshall SW. Postural stability and neuropsychological deficits after concussion in collegiate athletes. J Athl Train. 2001;36(3):263.
15. Mucha A, Collins MW, Elbin R, et al. A brief Vestibular/Ocular Motor Screening (VOMS) assessment to evaluate concussions preliminary findings. Am J Sports Med. 2014;42(10):2479-2486.
16. Bloom J. Vestibular and ocular motor assessments: Important pieces to the concussion puzzle. Athletic Training and Sports Health Care. 2013;5(6):246-248.
17. Covassin T, Elbin R, Harris W, Parker T, Kontos A. The role of age and sex in symptoms, neurocognitive performance, and postural stability in athletes after concussion. Am J Sports Med. 2012;40(6):1303-1312.
18. Kontos A, Sufrinko A, Elbin R, Puskar A, Collins M. Reliability and associated risk factors for performance on the Vestibular/Ocular Motor Screening (VOMS) tool in healthy collegiate athletes. Am J Sports Med. 2016;44(6):1400-1406.
19. Guskiewicz KM, McCrea M, Marshall SW, et al. Cumulative effects associated with recurrent concussion in collegiate football players: the NCAA Concussion Study. JAMA. 2003;290(19):2549-2555.
20. Lau B, Lovell MR, Collins MW, Pardini J. Neurocognitive and symptom predictors of recovery in high school athletes. Clin J Sport Med. 2009;19(3):216-221.
21. Lau BC, Kontos AP, Collins MW, Mucha A, Lovell MR. Which on-field signs/symptoms predict protracted recovery from sport-related concussion among high school football players? Am J Sports Med. 2011;39(11):2311-2318.
22. Mihalik JP, Register-Mihalik J, Kerr ZY, Marshall SW, McCrea MC, Guskiewicz KM. Recovery of posttraumatic migraine characteristics in patients after mild traumatic brain injury. Am J Sports Med. 2013;41(7):1490-1496.
23. Covassin T, Moran R, Elbin RJ. Sex differences in reported concussion injury rates and time loss from participation: An update of the National Collegiate Athletic Association injury surveillance program from 2004-2005 through 2008-2009. J Athl Train. 2016;51(3):189-194.
24. Root JM, Zuckerbraun NS, Wang L, et al. History of somatization is associated with prolonged recovery from concussion. J Pediatr. 2016;174:39-44.
25. Sufrinko A, Pearce K, Elbin RJ, et al. The effect of preinjury sleep difficulties on neurocognitive impairment and symptoms after sport-related concussion. Am J Sports Med. 2015;43(4):830-838.
26. Thomas DG, Apps JN, Hoffmann RG, McCrea M, Hammeke T. Benefits of strict rest after acute concussion: a randomized controlled trial. Pediatrics. 2015;135(2):213-223.
27. Choe M, Blume H. Pediatric posttraumatic Headache: a review. J Child Neurol. 2016;31(1):76-85.
28. Tepper SJ, Tepper DE. Breaking the cycle of medication overuse. Cleve Clin J Med. 2010;77(4):236-242.
29. Collins M, Kontos A, Reynolds E, Murawski C, Fu F. A comprehensive, targeted approach to the clinical care of athletes following sport-related concussion. Knee Surg Sports Traumatol Arthrosc. 2014;22(2):235-246.
30. Reynolds E, Collins MW, Mucha A, Troutman-Ensecki C. Establishing a clinical service for the management of sports-related concussions. Neurosurgery. 2014;75 Suppl 4:S71-S81.
31. Broglio SP, Collins MW, Williams RM, Mucha A, Kontos AP. Current and emerging rehabilitation for concussion: a review of the evidence. Clin Sports Med. 2015;34(2):213-231.
32. Master C, Scheiman M, Gallaway M, et al. Vision diagnoses are common after concussion in adolescents. Clin Pediatr (Phila). 2016;55(3):260-267.
33. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia. 2013;33(9):629-808.
34. Kontos A, Deitrick JM, Reynolds E. Mental health implication and consequences following sport-related concussion. Brit J Sports Med. 2016;50(3):139-140.
35. Kontos AP, Covassin T, Elbin R, Parker T. Depression and neurocognitive performance after concussion among male and female high school and collegiate athletes. Arch Phys Med Rehabil. 2012;93(10):1751-1756.
Football is an important component of American culture, with approximately 3 million youth athletes, 1.1 million high school athletes, and 100,000 college athletes participating each year.1 Participation in football provides athletes with physical, social, psychological, and academic benefits. Despite these benefits, widespread focus has been placed on the safety of football due to the risk for sport-related concussion (SRC) and potentially long-term effects; however, little recognition has been given to the advancements in concussion management across time and occurrence of concussions during most life activities. Although it is reasonable for concerns to be presented, it is important to better understand SRC and the current factors leading to prolonged recoveries, increased risk for injury, and potentially long-term effects.
What Is a Concussion?
Concussions occur after sustaining direct or indirect injury to the head or other parts of the body, as long as the injury force is transmitted to the head. Athletes often experience physical, cognitive, emotional, and sleep-related symptoms post-concussion secondary to an “energy crisis” within the brain.2 The energy crisis occurs as the result of transient neurological dysfunction triggered by changes in the brain (eg, release of neurotransmitters, impaired axonal function).2,3 Concussion is undetectable with traditional imaging; however, advanced imaging techniques (eg, diffuse tensor imaging) have shown progress in assessing axonal injury.3 Symptom duration post-concussion is highly variable due to individual differences; a recent study showed recovery took 3 to 4 weeks for memory and symptoms.4,5
Previous Concussion Management
Identification techniques and return-to-play guidelines for concussion have significantly changed across time. In the past, concussion grading scales were utilized for diagnosis and return to play was possible within the same contest.6,7 It has since been recognized that initial concussion severity makes it difficult to predict recovery.3 For example, research revealed memory decline and increased symptoms 36 hours post-injury for athletes with a grade 1 concussion (ie, transient confusion, no loss of consciousness, concussion symptoms or mental status changes that resolve within 15 minutes of injury) compared to baseline.7 Another study found duration of mental status changes to be related to slower symptom resolution and memory impairment 36 hours to 7 days post-injury.6 Consequently, return to play within the same contest was likely too liberal. Guidelines today recommend immediate removal from play with suspected SRC. Nevertheless, the “play through pain” culture has led athletes to continue playing after SRC, contributing to prolonged recoveries and potentially long-term effects.
Current Concussion Management: Continued Concerns and Areas of Improvement
Despite increased awareness of concussions, recent estimates revealed high rates (ie, 27:1 ratio for general players) of underreporting in college football, particularly amongst offensive linemen.8 Researchers have studied recovery implications for remaining in play, with one study revealing a 2.2 times greater risk for prolonged recovery in college athletes with delayed vs immediate removal.9 Another similar study discovered an 8.8 times greater risk for prolonged recovery in adolescent and young adult athletes not removed vs removed from play.10 Further analysis found remaining in play to be the greatest risk factor for prolonged recovery compared to other previously studied risk factors (eg, age, sex, posttraumatic migraine).10 Additionally, significant differences in neurocognitive data were seen between the “removed” and “not removed” groups for verbal memory, visual memory, processing speed, and reaction time at 1 to 7 days and 8 to 30 days.10 The recovery implications of remaining in play and the additional risk for second impact syndrome (SIS), or repeat concussion when recovering from another injury, emphasizes the need for further education efforts amongst athletes to encourage immediate reporting of injury.11
Sideline Assessment
Sideline assessment has become a vital component of concussion management to rule out concussion and/or significant injury other than concussion. Assessment should include observation, cognitive/balance testing, neurologic examination, and possible exertion testing to ensure a comprehensive evaluation of all areas of potential dysfunction.12 Indications for emergency department evaluation include suspicion for cervical spine injury, intracranial hemorrhage, or skull fracture as well as prolonged loss of consciousness, high-risk mechanisms, posttraumatic seizure(s), and/or significant worsening of symptoms.12
Observation
On the sideline, it is important to identify any immediate signs of injury (ie, loss of consciousness, anterograde/retrograde amnesia, and disorientation/confusion). Since immediate signs are not always present, it is important to be aware of the most commonly reported symptoms, including headache, difficulty concentrating, fatigue, drowsiness, and dizziness.13 If symptoms are not reported by the athlete, balance problems, lack of coordination, increased emotionality, and difficulty following instructions may be observed during play.12
On-Field Assessment
Cognitive and balance testing are essential in determining if an athlete has sustained a concussion. Immediate declines in memory, concentration abilities, and balance abilities are common. Given limitations in administering long testing batteries on the sideline, brief standardized tests such as the Standardized Assessment of Concussion (SAC), Balance Error Scoring System (BESS), and Sport Concussion Assessment Tool (SCAT) are commonly utilized. Identification of cognitive and/or balance abnormalities can help the athlete recognize deficits following injury.12 Balance problems are experienced due to abnormalities in sensory organization and generally resolve during the acute recovery period.14,15 Cognitive difficulties typically persist longer than balance problems, though duration varies widely.
Neurologic Evaluation
A neurologic evaluation including cranial nerve testing and evaluation of motor-sensory function (ie, assessment for the strength and sensation of upper and lower extremities) is important to identify focal deficits (ie, sensation changes, loss of fine motor control) indicative of serious intracranial pathology.12 Additionally, clinicians have suggested inclusion of vestibular and oculomotor assessments due to frequent dysfunction post-concussion.12,15,16 Examination of the vestibular/oculomotor systems through tools such as the Vestibular/Ocular Motor Screening (VOMS) assessment (assesses both the vestibular and oculomotor systems) and King-Devick Test (primarily assesses saccadic eye movements) can elicit symptoms that may not present immediately. If assessment appears normal, exertion testing can be utilized to determine if symptoms are provoked through physical exercise that should include cardio, dynamic, and sport-specific activities to stress the vestibular system.12
Risk Factors for Injury and Prolonged Recovery
Medical professionals must consider the presence of risk factors when managing concussion in order to make appropriate treatment recommendations and return-to-play decisions. Research has demonstrated the role of female gender, learning disability, attention-deficit/hyperactivity disorder, psychiatric history, young age, motion sickness, sleep problems, somatization, concussion history, on-field dizziness, posttraumatic migraine, and fogginess in increased risk for injury and/or prolonged recovery.17-25 Additionally, athletes with ongoing symptoms from a previous injury are at risk for sustaining another injury.
Acute Home Concussion Management
Although strict rest has been recommended post-concussion, recent research evaluating strict rest vs usual care for adolescents revealed greater symptom reports and longer recovery periods for the strict rest group.26 Based on these findings and emphasis for regulation within the migraine literature (due to the common pathophysiology between migraine and concussion27), we recommend that athletes follow a regulated daily schedule post-concussion including: 1) regular sleep-wake schedule with avoidance of naps, 2) regular meals, 3) adequate fluid hydration, 4) light noncontact physical activity (ie, walking, with progressions recommended by a physician), and 5) stress management techniques. Use of these strategies immediately can help in preventing against increased symptoms and stress, and decreases the need for medication in select cases. Additionally, over-the-counter medications should be limited to 2 to 3 doses per week to avoid rebound headaches.28
In-Office Concussion Management
Athletes diagnosed with SRC will experience different symptoms based on the injury mechanism, risk factors, and management approach. Comprehensive evaluation should include assessment of risk factors, injury details, symptoms, neurocognitive functioning, vestibular/oculomotor dysfunction, tolerance of physical exertion, balance functioning, and cervical spine integrity (if necessary).29,30 Due to individual differences and the heterogeneous symptom profiles, concussion management must move beyond a “one size fits all” approach to avoid nonspecific treatment strategies and consequently prolonged recoveries.29 Clinicians and researchers at University of Pittsburgh Medical Center have identified 6 concussion clinical profiles (ie, vestibular, ocular, posttraumatic migraine, cervical, anxiety/mood, and cognitive/fatigue) that are generally identifiable 48 hours after injury.29,30 Identification of the clinical profile(s) through a comprehensive evaluation guides the development of individualized treatment plans and targeted rehabilitation strategies.29,30
Vestibular. The vestibular system is responsible for stabilizing vision while the head moves and balance control.15 Athletes can experience central and/or peripheral vestibular dysfunction to include benign paroxysmal positional vertigo (BPPV), visual motion sensitivity, vestibular ocular reflex impairment, and balance impairment.30,31 Symptoms typically include dizziness, impaired balance, blurry vision, difficulty focusing, and environmental sensitivity.15,29,30 Potential treatment options include vestibular rehabilitation, exertion therapy, and school/work accommodations.
Ocular. The oculomotor system is responsible for control of eye movements. Athletes can experience many different posttraumatic vision changes, including convergence problems, eye-tracking difficulties, refractive error, difficulty with pursuits/saccades, and accommodation insufficiency. Symptoms typically include light sensitivity, blurred vision, double vision, headaches, fatigue, and memory difficulties.15,29,30 Potential treatment options include vision therapy, vestibular rehabilitation, and school/work accommodations.32
Posttraumatic Migraine. Headache, the most common post-concussion symptom, can persist and meet criteria for posttraumatic migraine (ie, unilateral headache with accompanying nausea and/or photophobia and phonophobia).29,30,33 Implementation of a routine schedule, daily physical activity, exertion therapy, pharmacologic intervention, and school/work accommodations are potential treatment options.
Cervical. The cervical spine can be injured during whiplash-type injuries. Therefore, determining the location, onset, and typical exacerbations of pain can be helpful in identifying cervical involvement.29,30 Symptoms typically include headaches, neck pain, numbness, and tingling. Evaluation and therapy by a certified physical therapist and pharmacologic intervention (eg, muscle relaxants) are potential treatment options. 29,30
Anxiety/Mood. Anxiety, or worry and fear about everyday situations, is common post-concussion and can sometimes be related to ongoing vestibular impairment. Symptoms typically include ruminative thoughts, avoidance of specific situations, hypervigilance, feelings of being overwhelmed, and difficulty falling asleep.29,30 Potential treatment options include implementation of a routine schedule, exposure to provocative situations, psychotherapy, pharmacologic intervention, and school/work accommodations.34
Cognitive/Fatigue. A global concussion factor (including cognitive, fatigue, and migraine symptoms) has been identified within 1 to 7 days of injury. Although this factor of symptoms generally resolves during the acute recovery period, it persists in select cases.13 Symptoms typically include fatigue, decreased energy levels, nonspecific headaches, potential sleep disruption, increased symptoms towards the end of the day, difficulty concentrating, and increased headache with cognitive activities.29,30,35 Routine schedule, daily physical activity, exertion therapy, pharmacologic intervention (eg, amantadine), and school/work accommodations are potential treatment options.30
Conclusion
Advancements in SRC management warrant change in the conversations regarding concussion in football. Specifically, conversations should address the current understanding of concussion and improvements in the safety of football through stricter concussion guidelines, detailed sideline evaluations, recognition of risk factors, improved acute management, and identification of concussion profiles that help to direct individualized treatment plans and targeted rehabilitation strategies. The biggest concerns related to concussions in football include underreporting of injury, premature return to play, and receiving routine rather than individualized treatment. Therefore, to further improve the safety of football and management of concussion it is essential that future efforts focus on the following 6 areas:
Education: Improved understanding of concussion is imperative to reducing poor outcomes and widespread concerns.
Immediate reporting: Reporting of concussion must be expected and encouraged through consistent responses by coaches to reduce underreporting and fear of reporting in athletes.
Prevention techniques: Athletes must be taught proper form and playing techniques to reduce the risk for concussion. Proper form and technique should be incentivized.
Targeted treatment: Individualized treatment plans and targeted rehabilitation strategies must be developed based on the identified clinical profile(s) to avoid nonspecific treatment recommendations.
Multidisciplinary treatment teams: Given the heterogeneous symptoms profiles and need for care provided by different medical specialties, multidisciplinary teams are essential.
Remain current: With the progress in understanding concussion, providers must remain vigilant of future advances in concussion management to further improve the safety of football.
Am J Orthop. 2016;45(6):352-356. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
Football is an important component of American culture, with approximately 3 million youth athletes, 1.1 million high school athletes, and 100,000 college athletes participating each year.1 Participation in football provides athletes with physical, social, psychological, and academic benefits. Despite these benefits, widespread focus has been placed on the safety of football due to the risk for sport-related concussion (SRC) and potentially long-term effects; however, little recognition has been given to the advancements in concussion management across time and occurrence of concussions during most life activities. Although it is reasonable for concerns to be presented, it is important to better understand SRC and the current factors leading to prolonged recoveries, increased risk for injury, and potentially long-term effects.
What Is a Concussion?
Concussions occur after sustaining direct or indirect injury to the head or other parts of the body, as long as the injury force is transmitted to the head. Athletes often experience physical, cognitive, emotional, and sleep-related symptoms post-concussion secondary to an “energy crisis” within the brain.2 The energy crisis occurs as the result of transient neurological dysfunction triggered by changes in the brain (eg, release of neurotransmitters, impaired axonal function).2,3 Concussion is undetectable with traditional imaging; however, advanced imaging techniques (eg, diffuse tensor imaging) have shown progress in assessing axonal injury.3 Symptom duration post-concussion is highly variable due to individual differences; a recent study showed recovery took 3 to 4 weeks for memory and symptoms.4,5
Previous Concussion Management
Identification techniques and return-to-play guidelines for concussion have significantly changed across time. In the past, concussion grading scales were utilized for diagnosis and return to play was possible within the same contest.6,7 It has since been recognized that initial concussion severity makes it difficult to predict recovery.3 For example, research revealed memory decline and increased symptoms 36 hours post-injury for athletes with a grade 1 concussion (ie, transient confusion, no loss of consciousness, concussion symptoms or mental status changes that resolve within 15 minutes of injury) compared to baseline.7 Another study found duration of mental status changes to be related to slower symptom resolution and memory impairment 36 hours to 7 days post-injury.6 Consequently, return to play within the same contest was likely too liberal. Guidelines today recommend immediate removal from play with suspected SRC. Nevertheless, the “play through pain” culture has led athletes to continue playing after SRC, contributing to prolonged recoveries and potentially long-term effects.
Current Concussion Management: Continued Concerns and Areas of Improvement
Despite increased awareness of concussions, recent estimates revealed high rates (ie, 27:1 ratio for general players) of underreporting in college football, particularly amongst offensive linemen.8 Researchers have studied recovery implications for remaining in play, with one study revealing a 2.2 times greater risk for prolonged recovery in college athletes with delayed vs immediate removal.9 Another similar study discovered an 8.8 times greater risk for prolonged recovery in adolescent and young adult athletes not removed vs removed from play.10 Further analysis found remaining in play to be the greatest risk factor for prolonged recovery compared to other previously studied risk factors (eg, age, sex, posttraumatic migraine).10 Additionally, significant differences in neurocognitive data were seen between the “removed” and “not removed” groups for verbal memory, visual memory, processing speed, and reaction time at 1 to 7 days and 8 to 30 days.10 The recovery implications of remaining in play and the additional risk for second impact syndrome (SIS), or repeat concussion when recovering from another injury, emphasizes the need for further education efforts amongst athletes to encourage immediate reporting of injury.11
Sideline Assessment
Sideline assessment has become a vital component of concussion management to rule out concussion and/or significant injury other than concussion. Assessment should include observation, cognitive/balance testing, neurologic examination, and possible exertion testing to ensure a comprehensive evaluation of all areas of potential dysfunction.12 Indications for emergency department evaluation include suspicion for cervical spine injury, intracranial hemorrhage, or skull fracture as well as prolonged loss of consciousness, high-risk mechanisms, posttraumatic seizure(s), and/or significant worsening of symptoms.12
Observation
On the sideline, it is important to identify any immediate signs of injury (ie, loss of consciousness, anterograde/retrograde amnesia, and disorientation/confusion). Since immediate signs are not always present, it is important to be aware of the most commonly reported symptoms, including headache, difficulty concentrating, fatigue, drowsiness, and dizziness.13 If symptoms are not reported by the athlete, balance problems, lack of coordination, increased emotionality, and difficulty following instructions may be observed during play.12
On-Field Assessment
Cognitive and balance testing are essential in determining if an athlete has sustained a concussion. Immediate declines in memory, concentration abilities, and balance abilities are common. Given limitations in administering long testing batteries on the sideline, brief standardized tests such as the Standardized Assessment of Concussion (SAC), Balance Error Scoring System (BESS), and Sport Concussion Assessment Tool (SCAT) are commonly utilized. Identification of cognitive and/or balance abnormalities can help the athlete recognize deficits following injury.12 Balance problems are experienced due to abnormalities in sensory organization and generally resolve during the acute recovery period.14,15 Cognitive difficulties typically persist longer than balance problems, though duration varies widely.
Neurologic Evaluation
A neurologic evaluation including cranial nerve testing and evaluation of motor-sensory function (ie, assessment for the strength and sensation of upper and lower extremities) is important to identify focal deficits (ie, sensation changes, loss of fine motor control) indicative of serious intracranial pathology.12 Additionally, clinicians have suggested inclusion of vestibular and oculomotor assessments due to frequent dysfunction post-concussion.12,15,16 Examination of the vestibular/oculomotor systems through tools such as the Vestibular/Ocular Motor Screening (VOMS) assessment (assesses both the vestibular and oculomotor systems) and King-Devick Test (primarily assesses saccadic eye movements) can elicit symptoms that may not present immediately. If assessment appears normal, exertion testing can be utilized to determine if symptoms are provoked through physical exercise that should include cardio, dynamic, and sport-specific activities to stress the vestibular system.12
Risk Factors for Injury and Prolonged Recovery
Medical professionals must consider the presence of risk factors when managing concussion in order to make appropriate treatment recommendations and return-to-play decisions. Research has demonstrated the role of female gender, learning disability, attention-deficit/hyperactivity disorder, psychiatric history, young age, motion sickness, sleep problems, somatization, concussion history, on-field dizziness, posttraumatic migraine, and fogginess in increased risk for injury and/or prolonged recovery.17-25 Additionally, athletes with ongoing symptoms from a previous injury are at risk for sustaining another injury.
Acute Home Concussion Management
Although strict rest has been recommended post-concussion, recent research evaluating strict rest vs usual care for adolescents revealed greater symptom reports and longer recovery periods for the strict rest group.26 Based on these findings and emphasis for regulation within the migraine literature (due to the common pathophysiology between migraine and concussion27), we recommend that athletes follow a regulated daily schedule post-concussion including: 1) regular sleep-wake schedule with avoidance of naps, 2) regular meals, 3) adequate fluid hydration, 4) light noncontact physical activity (ie, walking, with progressions recommended by a physician), and 5) stress management techniques. Use of these strategies immediately can help in preventing against increased symptoms and stress, and decreases the need for medication in select cases. Additionally, over-the-counter medications should be limited to 2 to 3 doses per week to avoid rebound headaches.28
In-Office Concussion Management
Athletes diagnosed with SRC will experience different symptoms based on the injury mechanism, risk factors, and management approach. Comprehensive evaluation should include assessment of risk factors, injury details, symptoms, neurocognitive functioning, vestibular/oculomotor dysfunction, tolerance of physical exertion, balance functioning, and cervical spine integrity (if necessary).29,30 Due to individual differences and the heterogeneous symptom profiles, concussion management must move beyond a “one size fits all” approach to avoid nonspecific treatment strategies and consequently prolonged recoveries.29 Clinicians and researchers at University of Pittsburgh Medical Center have identified 6 concussion clinical profiles (ie, vestibular, ocular, posttraumatic migraine, cervical, anxiety/mood, and cognitive/fatigue) that are generally identifiable 48 hours after injury.29,30 Identification of the clinical profile(s) through a comprehensive evaluation guides the development of individualized treatment plans and targeted rehabilitation strategies.29,30
Vestibular. The vestibular system is responsible for stabilizing vision while the head moves and balance control.15 Athletes can experience central and/or peripheral vestibular dysfunction to include benign paroxysmal positional vertigo (BPPV), visual motion sensitivity, vestibular ocular reflex impairment, and balance impairment.30,31 Symptoms typically include dizziness, impaired balance, blurry vision, difficulty focusing, and environmental sensitivity.15,29,30 Potential treatment options include vestibular rehabilitation, exertion therapy, and school/work accommodations.
Ocular. The oculomotor system is responsible for control of eye movements. Athletes can experience many different posttraumatic vision changes, including convergence problems, eye-tracking difficulties, refractive error, difficulty with pursuits/saccades, and accommodation insufficiency. Symptoms typically include light sensitivity, blurred vision, double vision, headaches, fatigue, and memory difficulties.15,29,30 Potential treatment options include vision therapy, vestibular rehabilitation, and school/work accommodations.32
Posttraumatic Migraine. Headache, the most common post-concussion symptom, can persist and meet criteria for posttraumatic migraine (ie, unilateral headache with accompanying nausea and/or photophobia and phonophobia).29,30,33 Implementation of a routine schedule, daily physical activity, exertion therapy, pharmacologic intervention, and school/work accommodations are potential treatment options.
Cervical. The cervical spine can be injured during whiplash-type injuries. Therefore, determining the location, onset, and typical exacerbations of pain can be helpful in identifying cervical involvement.29,30 Symptoms typically include headaches, neck pain, numbness, and tingling. Evaluation and therapy by a certified physical therapist and pharmacologic intervention (eg, muscle relaxants) are potential treatment options. 29,30
Anxiety/Mood. Anxiety, or worry and fear about everyday situations, is common post-concussion and can sometimes be related to ongoing vestibular impairment. Symptoms typically include ruminative thoughts, avoidance of specific situations, hypervigilance, feelings of being overwhelmed, and difficulty falling asleep.29,30 Potential treatment options include implementation of a routine schedule, exposure to provocative situations, psychotherapy, pharmacologic intervention, and school/work accommodations.34
Cognitive/Fatigue. A global concussion factor (including cognitive, fatigue, and migraine symptoms) has been identified within 1 to 7 days of injury. Although this factor of symptoms generally resolves during the acute recovery period, it persists in select cases.13 Symptoms typically include fatigue, decreased energy levels, nonspecific headaches, potential sleep disruption, increased symptoms towards the end of the day, difficulty concentrating, and increased headache with cognitive activities.29,30,35 Routine schedule, daily physical activity, exertion therapy, pharmacologic intervention (eg, amantadine), and school/work accommodations are potential treatment options.30
Conclusion
Advancements in SRC management warrant change in the conversations regarding concussion in football. Specifically, conversations should address the current understanding of concussion and improvements in the safety of football through stricter concussion guidelines, detailed sideline evaluations, recognition of risk factors, improved acute management, and identification of concussion profiles that help to direct individualized treatment plans and targeted rehabilitation strategies. The biggest concerns related to concussions in football include underreporting of injury, premature return to play, and receiving routine rather than individualized treatment. Therefore, to further improve the safety of football and management of concussion it is essential that future efforts focus on the following 6 areas:
Education: Improved understanding of concussion is imperative to reducing poor outcomes and widespread concerns.
Immediate reporting: Reporting of concussion must be expected and encouraged through consistent responses by coaches to reduce underreporting and fear of reporting in athletes.
Prevention techniques: Athletes must be taught proper form and playing techniques to reduce the risk for concussion. Proper form and technique should be incentivized.
Targeted treatment: Individualized treatment plans and targeted rehabilitation strategies must be developed based on the identified clinical profile(s) to avoid nonspecific treatment recommendations.
Multidisciplinary treatment teams: Given the heterogeneous symptoms profiles and need for care provided by different medical specialties, multidisciplinary teams are essential.
Remain current: With the progress in understanding concussion, providers must remain vigilant of future advances in concussion management to further improve the safety of football.
Am J Orthop. 2016;45(6):352-356. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Dompier TP, Kerr ZY, Marshall SW, et al. Incidence of concussion during practice and games in youth, high school, and collegiate American football players. JAMA Pediatrics. 2015;169(7):659-665.
2. Giza C, Hovda D. The new neurometabolic cascade of concussion
3. Barkhoudarian G, Hovda DA, Giza CC. The molecular pathophysiology of concussive brain injury - an update. Phys Med Rehabil Clin N Am. 2016;27:373-393.
4. Henry L, Elbin R, Collins M, Marchetti G, Kontos A. Examining recovery trajectories after sport-related concussion with a multimodal clinical assessment approach. Neurosurgery. 2016;78(2):232-241.
5. McCrory P, Meeuwisse WH, Aubry M, et al. Consensus statement on concussion in sport: the 4th International Conference on Concussion in Sport held in Zurich, November 2012. Brit J Sports Med. 2013;47(5):250-258.
6. Lovell MR, Collins MW, Iverson GL, et al. Recovery from mild concussion in high school athletes. J Neurosurg. 2003;98(2):296-301.
7. Lovell MR, Collins MW, Iverson GL, Johnston KM, Bradley JP. Grade 1 or “ding” concussions in high school athletes. Am J Sports Med. 2004;32(1):47-54.
8. Baugh CM, Kroshus E, Daneshvar DH, Filali NA, Hiscox MJ, Glantz LH. Concussion management in United States college sports: compliance with National Collegiate Athletic Association concussion policy and areas for improvement. Am J Sports Med. 2015;43(1):47-56.
9. Asken BM, McCrea MA, Clugston JR, Snyder AR, Houck ZM, Bauer RM. “Playing through it”: Delayed reporting and removal from athletic activity after concussion predicts prolonged recovery. J Athl Train. 2016;51(4):329-335.
10. Elbin RJ, Sufrinko A, Schatz P, et al. Athletes that continue to play with concussion demonstrate worse recovery outcomes than athletes immediately removed from play. J Pediatr. In press.
11. Signoretti S, Lazzarino G, Tavazzi B, Vagnozzi R. The pathophysiology of concussion. PM R. 2011;3(10 Suppl 2):S359-S368.
12. Bloom J, Blount JG. Sideline evaluation of concussion. UpToDate. 2016. http://www.uptodate.com/contents/sideline-evaluation-of-concussion. Accessed July 13, 2016.
13. Kontos AP, Elbin RJ, Schatz P, et al. A revised factor structure for the post-concussion symptom scale: baseline and postconcussion factors. Am J Sports Med. 2012;40(10):2375-2384.
14. Guskiewicz KM, Ross SE, Marshall SW. Postural stability and neuropsychological deficits after concussion in collegiate athletes. J Athl Train. 2001;36(3):263.
15. Mucha A, Collins MW, Elbin R, et al. A brief Vestibular/Ocular Motor Screening (VOMS) assessment to evaluate concussions preliminary findings. Am J Sports Med. 2014;42(10):2479-2486.
16. Bloom J. Vestibular and ocular motor assessments: Important pieces to the concussion puzzle. Athletic Training and Sports Health Care. 2013;5(6):246-248.
17. Covassin T, Elbin R, Harris W, Parker T, Kontos A. The role of age and sex in symptoms, neurocognitive performance, and postural stability in athletes after concussion. Am J Sports Med. 2012;40(6):1303-1312.
18. Kontos A, Sufrinko A, Elbin R, Puskar A, Collins M. Reliability and associated risk factors for performance on the Vestibular/Ocular Motor Screening (VOMS) tool in healthy collegiate athletes. Am J Sports Med. 2016;44(6):1400-1406.
19. Guskiewicz KM, McCrea M, Marshall SW, et al. Cumulative effects associated with recurrent concussion in collegiate football players: the NCAA Concussion Study. JAMA. 2003;290(19):2549-2555.
20. Lau B, Lovell MR, Collins MW, Pardini J. Neurocognitive and symptom predictors of recovery in high school athletes. Clin J Sport Med. 2009;19(3):216-221.
21. Lau BC, Kontos AP, Collins MW, Mucha A, Lovell MR. Which on-field signs/symptoms predict protracted recovery from sport-related concussion among high school football players? Am J Sports Med. 2011;39(11):2311-2318.
22. Mihalik JP, Register-Mihalik J, Kerr ZY, Marshall SW, McCrea MC, Guskiewicz KM. Recovery of posttraumatic migraine characteristics in patients after mild traumatic brain injury. Am J Sports Med. 2013;41(7):1490-1496.
23. Covassin T, Moran R, Elbin RJ. Sex differences in reported concussion injury rates and time loss from participation: An update of the National Collegiate Athletic Association injury surveillance program from 2004-2005 through 2008-2009. J Athl Train. 2016;51(3):189-194.
24. Root JM, Zuckerbraun NS, Wang L, et al. History of somatization is associated with prolonged recovery from concussion. J Pediatr. 2016;174:39-44.
25. Sufrinko A, Pearce K, Elbin RJ, et al. The effect of preinjury sleep difficulties on neurocognitive impairment and symptoms after sport-related concussion. Am J Sports Med. 2015;43(4):830-838.
26. Thomas DG, Apps JN, Hoffmann RG, McCrea M, Hammeke T. Benefits of strict rest after acute concussion: a randomized controlled trial. Pediatrics. 2015;135(2):213-223.
27. Choe M, Blume H. Pediatric posttraumatic Headache: a review. J Child Neurol. 2016;31(1):76-85.
28. Tepper SJ, Tepper DE. Breaking the cycle of medication overuse. Cleve Clin J Med. 2010;77(4):236-242.
29. Collins M, Kontos A, Reynolds E, Murawski C, Fu F. A comprehensive, targeted approach to the clinical care of athletes following sport-related concussion. Knee Surg Sports Traumatol Arthrosc. 2014;22(2):235-246.
30. Reynolds E, Collins MW, Mucha A, Troutman-Ensecki C. Establishing a clinical service for the management of sports-related concussions. Neurosurgery. 2014;75 Suppl 4:S71-S81.
31. Broglio SP, Collins MW, Williams RM, Mucha A, Kontos AP. Current and emerging rehabilitation for concussion: a review of the evidence. Clin Sports Med. 2015;34(2):213-231.
32. Master C, Scheiman M, Gallaway M, et al. Vision diagnoses are common after concussion in adolescents. Clin Pediatr (Phila). 2016;55(3):260-267.
33. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia. 2013;33(9):629-808.
34. Kontos A, Deitrick JM, Reynolds E. Mental health implication and consequences following sport-related concussion. Brit J Sports Med. 2016;50(3):139-140.
35. Kontos AP, Covassin T, Elbin R, Parker T. Depression and neurocognitive performance after concussion among male and female high school and collegiate athletes. Arch Phys Med Rehabil. 2012;93(10):1751-1756.
1. Dompier TP, Kerr ZY, Marshall SW, et al. Incidence of concussion during practice and games in youth, high school, and collegiate American football players. JAMA Pediatrics. 2015;169(7):659-665.
2. Giza C, Hovda D. The new neurometabolic cascade of concussion
3. Barkhoudarian G, Hovda DA, Giza CC. The molecular pathophysiology of concussive brain injury - an update. Phys Med Rehabil Clin N Am. 2016;27:373-393.
4. Henry L, Elbin R, Collins M, Marchetti G, Kontos A. Examining recovery trajectories after sport-related concussion with a multimodal clinical assessment approach. Neurosurgery. 2016;78(2):232-241.
5. McCrory P, Meeuwisse WH, Aubry M, et al. Consensus statement on concussion in sport: the 4th International Conference on Concussion in Sport held in Zurich, November 2012. Brit J Sports Med. 2013;47(5):250-258.
6. Lovell MR, Collins MW, Iverson GL, et al. Recovery from mild concussion in high school athletes. J Neurosurg. 2003;98(2):296-301.
7. Lovell MR, Collins MW, Iverson GL, Johnston KM, Bradley JP. Grade 1 or “ding” concussions in high school athletes. Am J Sports Med. 2004;32(1):47-54.
8. Baugh CM, Kroshus E, Daneshvar DH, Filali NA, Hiscox MJ, Glantz LH. Concussion management in United States college sports: compliance with National Collegiate Athletic Association concussion policy and areas for improvement. Am J Sports Med. 2015;43(1):47-56.
9. Asken BM, McCrea MA, Clugston JR, Snyder AR, Houck ZM, Bauer RM. “Playing through it”: Delayed reporting and removal from athletic activity after concussion predicts prolonged recovery. J Athl Train. 2016;51(4):329-335.
10. Elbin RJ, Sufrinko A, Schatz P, et al. Athletes that continue to play with concussion demonstrate worse recovery outcomes than athletes immediately removed from play. J Pediatr. In press.
11. Signoretti S, Lazzarino G, Tavazzi B, Vagnozzi R. The pathophysiology of concussion. PM R. 2011;3(10 Suppl 2):S359-S368.
12. Bloom J, Blount JG. Sideline evaluation of concussion. UpToDate. 2016. http://www.uptodate.com/contents/sideline-evaluation-of-concussion. Accessed July 13, 2016.
13. Kontos AP, Elbin RJ, Schatz P, et al. A revised factor structure for the post-concussion symptom scale: baseline and postconcussion factors. Am J Sports Med. 2012;40(10):2375-2384.
14. Guskiewicz KM, Ross SE, Marshall SW. Postural stability and neuropsychological deficits after concussion in collegiate athletes. J Athl Train. 2001;36(3):263.
15. Mucha A, Collins MW, Elbin R, et al. A brief Vestibular/Ocular Motor Screening (VOMS) assessment to evaluate concussions preliminary findings. Am J Sports Med. 2014;42(10):2479-2486.
16. Bloom J. Vestibular and ocular motor assessments: Important pieces to the concussion puzzle. Athletic Training and Sports Health Care. 2013;5(6):246-248.
17. Covassin T, Elbin R, Harris W, Parker T, Kontos A. The role of age and sex in symptoms, neurocognitive performance, and postural stability in athletes after concussion. Am J Sports Med. 2012;40(6):1303-1312.
18. Kontos A, Sufrinko A, Elbin R, Puskar A, Collins M. Reliability and associated risk factors for performance on the Vestibular/Ocular Motor Screening (VOMS) tool in healthy collegiate athletes. Am J Sports Med. 2016;44(6):1400-1406.
19. Guskiewicz KM, McCrea M, Marshall SW, et al. Cumulative effects associated with recurrent concussion in collegiate football players: the NCAA Concussion Study. JAMA. 2003;290(19):2549-2555.
20. Lau B, Lovell MR, Collins MW, Pardini J. Neurocognitive and symptom predictors of recovery in high school athletes. Clin J Sport Med. 2009;19(3):216-221.
21. Lau BC, Kontos AP, Collins MW, Mucha A, Lovell MR. Which on-field signs/symptoms predict protracted recovery from sport-related concussion among high school football players? Am J Sports Med. 2011;39(11):2311-2318.
22. Mihalik JP, Register-Mihalik J, Kerr ZY, Marshall SW, McCrea MC, Guskiewicz KM. Recovery of posttraumatic migraine characteristics in patients after mild traumatic brain injury. Am J Sports Med. 2013;41(7):1490-1496.
23. Covassin T, Moran R, Elbin RJ. Sex differences in reported concussion injury rates and time loss from participation: An update of the National Collegiate Athletic Association injury surveillance program from 2004-2005 through 2008-2009. J Athl Train. 2016;51(3):189-194.
24. Root JM, Zuckerbraun NS, Wang L, et al. History of somatization is associated with prolonged recovery from concussion. J Pediatr. 2016;174:39-44.
25. Sufrinko A, Pearce K, Elbin RJ, et al. The effect of preinjury sleep difficulties on neurocognitive impairment and symptoms after sport-related concussion. Am J Sports Med. 2015;43(4):830-838.
26. Thomas DG, Apps JN, Hoffmann RG, McCrea M, Hammeke T. Benefits of strict rest after acute concussion: a randomized controlled trial. Pediatrics. 2015;135(2):213-223.
27. Choe M, Blume H. Pediatric posttraumatic Headache: a review. J Child Neurol. 2016;31(1):76-85.
28. Tepper SJ, Tepper DE. Breaking the cycle of medication overuse. Cleve Clin J Med. 2010;77(4):236-242.
29. Collins M, Kontos A, Reynolds E, Murawski C, Fu F. A comprehensive, targeted approach to the clinical care of athletes following sport-related concussion. Knee Surg Sports Traumatol Arthrosc. 2014;22(2):235-246.
30. Reynolds E, Collins MW, Mucha A, Troutman-Ensecki C. Establishing a clinical service for the management of sports-related concussions. Neurosurgery. 2014;75 Suppl 4:S71-S81.
31. Broglio SP, Collins MW, Williams RM, Mucha A, Kontos AP. Current and emerging rehabilitation for concussion: a review of the evidence. Clin Sports Med. 2015;34(2):213-231.
32. Master C, Scheiman M, Gallaway M, et al. Vision diagnoses are common after concussion in adolescents. Clin Pediatr (Phila). 2016;55(3):260-267.
33. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia. 2013;33(9):629-808.
34. Kontos A, Deitrick JM, Reynolds E. Mental health implication and consequences following sport-related concussion. Brit J Sports Med. 2016;50(3):139-140.
35. Kontos AP, Covassin T, Elbin R, Parker T. Depression and neurocognitive performance after concussion among male and female high school and collegiate athletes. Arch Phys Med Rehabil. 2012;93(10):1751-1756.
Exertional Heat Stroke and American Football: What the Team Physician Needs to Know
Football, one of the most popular sports in the United States, is additionally recognized as a leading contributor to sports injury secondary to the contact collision nature of the endeavor. There are an estimated 1.1 million high school football players with another 100,000 participants combined in the National Football League (NFL), college, junior college, Arena Football League, and semipro levels of play.1 USA Football estimates that an additional 3 million youth participate in community football leagues.1 The National Center for Catastrophic Sports Injury Research recently calculated a fatality rate of 0.14 per 100,000 participants in 2014 for the 4.2 million who play football at all levels—and 0.45 per 100,000 in high school.1 While direct deaths from head and spine injury remain a significant contributor to the number of catastrophic injuries, indirect deaths (systemic failure) predominate. Exertional heat stroke (EHS) has emerged as one of the leading indirect causes of death in high school and collegiate football. Boden and colleagues2 reported that high school and college football players sustain approximately 12 fatalities annually, with indirect systemic causes being twice as common as direct blunt trauma.2The most common indirect causes identified included cardiac failure, heat illness, and complications of sickle cell trait (SCT). It was also noted that the risk of SCT, heat-related, and cardiac deaths increased during the second decade of the study, indicating these conditions may require a greater emphasis on diagnosis, treatment, and prevention. This review details for the team physician the unique challenge of exercising in the heat to the football player, and the prevention, diagnosis, management and return-to-play issues pertinent to exertional heat illness (EHI).
The Challenge
EHS represents the most severe manifestation of EHI—a gamut of diseases commonly encountered during the hot summer months when American football season begins. The breadth of EHI includes several important clinical diagnoses: exercise-associated muscle cramps (heat cramps); heat exhaustion with and without syncope; heat injury with evidence of end organ injury (eg, rhabdomyolysis); and EHS. EHS is defined as “a form of hyperthermia associated with a systemic inflammatory response leading to a syndrome of multi-organ dysfunction in which encephalopathy predominates.”3 EHS, if left untreated, or even if clinical treatment is delayed, may result in significant end organ morbidity and/or mortality.
During exercise, the human thermoregulatory system mitigates heat gain by increasing skin blood flow and sweating, causing an increased dissipation of heat to the surrounding environment by leveraging conduction, convection, and evaporation.4,5 Elevated environmental temperatures, increased humidity, and dehydration can impede the body’s ability to dissipate heat at a rate needed to maintain thermoregulation. This imbalance can result in hyperthermia secondary to uncompensated heat stress,5 which in turn can lead to EHI. Football players have unique challenges that make them particularly vulnerable to EHI. The summer heat during early-season participation and the requirement for equipment that covers nearly 60% of body surfaces pose increased risk of volume losses and hyperthermia that trigger the onset of EHI.6 Football athletes’ body compositions and physical size are additional contributing risk factors; the relatively high muscle and fat content increase thermogenicity, which require their bodies to dissipate more heat.7
An estimated 9000 cases of EHI occur annually across all high school sports,8 with an incidence of 1.6:100,000 athlete-exposures.8,9 Studies have demonstrated, however, that EHI occurs in football 11.4 times more often than in all other high school sports combined.10 The incidence of nonfatal EHI in all levels of football is 4.42-5:100,000.8,9 Between 2000 and 2014, 41 football players died from EHS.1 In football, approximately 75% of all EHI events occurred during practices, while only 25% of incidents occurred during games.8
Given these potentially deadly consequences, it is important that football team physicians are not only alert to the early symptoms of heat illness and prepared to intervene to prevent the progression to EHS, but are critical leaders in educating coaches and players in evidence-based EHI prevention practices and policies.
Prevention
EHS is a preventable condition, arguably the most common cause of preventable nontraumatic exertional death in young athletes in the United States. Close attention to mitigating risk factors should begin prior to the onset of preseason practice and continue through the early season, where athletes are at the highest risk of developing heat illness.
Primary Prevention
Primary prevention is fundamental to minimizing the occurrences of EHI. It focuses on the following methods: recognition of inherent risk factors, acclimatization, hydration, and avoidance of inciting substances (including supplements).
Pre-Participation Examination. The purpose of the pre-participation examination (PPE) is to maximize an athlete’s safety by identifying medical conditions that place the athlete at risk.11,12 The Preparticipation Physical Evaluation, 4th edition, the most widely used consensus publication, specifically queries if an athlete has a previous history of heat injury. However, it only indirectly addresses intrinsic risk factors that may predispose an athlete to EHI who has never had an EHI before. Therefore, providers should take the opportunity of the PPE to inquire about additional risk factors that may make an athlete high risk for sustaining a heat injury. Common risk factors for EHI are listed in Table 1.
Heat Acclimatization. The risk of EHI escalates significantly when athletes are subjected to multiple stressors during periods of heat exposure, such as sudden increases in intensity or duration of exercise; prolonged new exposures to heat; dehydration; and sleep loss.5 When football season begins in late summer, athletes are least conditioned as temperatures reach their seasonal peak, causing increased risk of EHI.15 Planning for heat acclimatization is vital for all athletes who exercise in hot environments. Acclimatization procedures place progressively mounting physiologic strains on the body to improve athletes’ ability to dissipate heat, diminishing thermoregulatory and cardiovascular exertion.4,5 Acclimatization begins with expansion of plasma volume on days 3 to 6, causing improvements in cardiac efficiency and resulting in an overall decrease in basal internal body temperature.4,5,15 This process results in improvements in heat tolerance and exercise performance, evolving over 10 to 14 days of gradual escalation of exercise intensity and duration.5,10,11,16 However, poor fitness levels and extreme temperatures can prolong this period, requiring up to 2 to 3 months to fully take effect.5,7
The National Athletic Trainers Association (NATA) and National Collegiate Athletic Association (NCAA) have released consensus guidelines regarding heat acclimatization protocols for football athletes at the high school and college levels (Tables 3 and 4). Each of these guidelines involves an initial period without use of protective equipment, followed by a gradual addition of further equipment.11,16
Secondary Prevention
Despite physicians’ best efforts to prevent all cases of EHI, athletes will still experience the effects of exercise-induced hyperthermia. The goal of secondary prevention is to slow the progression of this hyperthermia so that it does not progress to more dangerous EHI.
Hydration. Dehydration is an important risk factor for EHI. Sweat maintains thermoregulation by dissipating heat generated during exercise; however, it also contributes to body water losses. Furthermore, intravascular depletion decreases stroke volume, thereby increasing cardiovascular strain. It is estimated that for every 1% loss in body mass from dehydration, body temperature rises 0.22°C in comparison to a euhydrated state.6 Dehydration occurs more rapidly in hot environments, as fluid is lost through increased sweat production.7 After approximately 6% to 10% body weight volume loss, cardiac output cannot be maintained, diminishing sweat production and blood flow to both skin and muscle and causing diminished performance and a significant risk of heat exhaustion.7 If left unchecked, these physiologic changes result in further elevations in body temperature and increased cardiovascular strain, ultimately placing the athlete at significant risk for development of EHS.
Adequate hydration to maintain euvolemia is an important step in avoiding possible EHI. Multiple studies have shown that football players experience a baseline hypovolemia during their competition season,6 a deficit that is most marked during the first week of practices.17 This deficit is multifactorial, as football players expend a significant amount of fluid through sweat, are not able to adequately replace these losses during practice, and do not appropriately hydrate off the field.6,18 Some players, especially linemen, sweat at a higher rate than their teammates, posing a possible risk of significant dehydration.6 Coaches and players alike should be educated on the importance of adequate hydration to meet their fluid needs.
The goal of hydration during exercise is to prevent large fluid losses that can adversely affect performance and increase risk of EHI;6 it may be unrealistic to replace all fluid losses during the practice period. Instead, athletes should target complete volume replacement over the post-exercise period.6 Some recommend hydrating based upon thirst drive; however, thirst is activated following a volume loss of approximately 2% body mass, the same degree of losses that place athletes at an increased risk for performance impairment and EHI.4,6,11,12 Individuals should have access to fluids throughout practice and competition and be encouraged to hydrate as needed.6,12,15 Furthermore, staff should modify their practices based upon WBGT and acclimatization status to provide more frequent hydration breaks.
Hyperhydration and Salt Intake. Of note, there are inherent risks to hyperhydration. Athletes with low sweat rates have an increased risk of overhydration and the development of exercise-associated hyponatremia (EAH),6 a condition whose presentation is very similar to EHS. In addition, inadequate sodium intake and excessive sweating can also contribute to the development of EAH. EAH has been implicated in the deaths of 2 football players in 2014.1,6 Establishing team hydration guidelines and educating players and staff on appropriate hydration and dietary salt intake is essential to reduce the risk of both dehydration and hyperhydration and their complications.6Intra-Event Cooling. During exercise, team physicians can employ strategies for cooling athletes during exertion to mitigate their risk of EHI by decreasing thermal and cardiovascular strain.4,19 Cooling during exercise is hypothesized to allow for accelerated heat dissipation, where heat is lost from the body more effectively. This accelerated loss enables athletes to maintain a higher heat storage capacity over the duration of exercise, avoiding uncompensated heat stresses that ultimately cause EHI.19
Some intra-event cooling strategies include the use of cooling garments, cooling packs, and cold water/slurry ingestion. Cooling garments lower skin temperature, which in turn can decrease thermoregulatory strains;4 a recent meta-analysis of intra-event cooling modalities revealed that wearing an ice vest during exercise resulted in the greatest decrease in thermal heat strain.19 Internal cooling strategies—namely ingestion of cold fluids/ice slurry—have shown some mild benefit in decreasing internal temperatures; however, some studies have demonstrated some decrease in sweat production associated with cold oral intake used in isolation.19 Overall, studies have shown that combining external (cooling clothing, ice packs, fanning) and internal (cold water, ice slurry) cooling methods result in a greater cooling effect than a use of a single method.4
Tertiary Prevention
The goal of tertiary prevention is to mitigate the risk of long-term adverse outcomes following an EHS event. The most effective means of reducing risk for morbidity and mortality is rapid identification and treatment of EHS as well as close evaluation of an athlete’s return to activity in heat. This process is spearheaded by an effective and well-rehearsed emergency action plan.
Diagnosis and Management
Rapid identification and treatment of EHS is crucial to minimizing the risk of poor outcomes.7 Any delay in the treatment of EHS can dramatically increase the likelihood of associated morbidity and mortality.20
EHS is diagnosed by an elevated rectal temperature ≥40°C (104°F) and associated central nervous system (CNS) dysfunction.21 EHS should be strongly suspected in any athlete exercising in heat who exhibits signs of CNS dysfunction, including disorientation, confusion, dizziness, erratic behavior, irritability, headache, loss of coordination, delirium, collapse, or seizures.7,12,15 EHS may also present with symptoms of heat exhaustion, including fatigue, hyperventilation, tachycardia, vomiting, diarrhea, and hypotension.7,12,15
Rectal temperature should be taken for any athlete with suspected EHS, as other modalities—oral, skin, axillary, and aural—can be inaccurate and easily modified by ambient confounders such as ambient and skin temperature, athlete hyperventilation, and consumption of liquids.7,11,12 Athletes exhibiting CNS symptoms with moderately elevated rectal temperatures that do not exceed 40°C should also be assumed to be suffering from EHS and treated with rapid cooling.11 On the other hand, athletes with CNS symptoms who are normothermic should be assumed to have EAH until ruled out by electrolyte assessment; IV fluids should be at no more than keep vein open (KVO) pending this determination.11 In some cases, an athlete may initially present with altered mental status but return to “normal.” However, this improvement may represent a “lucid period”; evaluation should continue with rectal temperature and treatment, as EHS in these cases may progress quickly.15
Treatment is centered on rapid, whole body cooling initiated at the first sign of heat illness.7,22 The goal of treatment is to achieve a rectal temperature <38.9°C within 30 minutes of the onset of EHS.15 Upon diagnosis, the athlete should be quickly placed in a tub of ice water to facilitate cold water immersion (CWI) therapy. Some guidelines suggest the athlete’s clothing be removed to potentiate evaporative cooling during CWI;12 however, cooling should not be delayed due to difficulties in removing equipment. CWI, where a heat stroke victim is submerged in ice water up to their neck while water is continuously circulated, is generally considered to be the gold standard treatment as it is the modality with the highest recorded cooling rates and the lowest rate of morbidity and mortality.7,20,21 Multiple studies of CWI have shown that survival nears 100% when aggressive cooling starts within 5 minutes of collapse or identification of EHS.20,21,22
If whole body CWI is unavailable, alternative methods of rapid cooling should be employed. Partial CWI, with torso immersion being preferable to the extremities, has been shown to achieve an acceptable rate of cooling to achieve sufficient drops in internal body temperature.20,23 However, one popular treatment—applying ice packs to the whole body, in particular to the groin and axillae—has not been shown to be sufficient to achieve standard cooling goals.20 None of these methods have been shown to be as effective as CWI.23
Intravenous access should be initiated with fluid resuscitation dictated by the provider’s assessment. Normal saline is recommended as the resuscitative fluid of choice, with the rate dictated by clinical judgment and adjusted as guided by electrolyte determination and clinical response. It cannot be overstated that in normothermic patients with confusion, EAH is the diagnosis of exclusion and aggressive fluid resuscitation should be withheld until electrolyte determination.
Once rectal temperature descends appropriately (~38.9°C), the cooling process should stop and the individual should be transported to a hospital for further observation20 and evaluation of possible sequelae, including rhabdomyolysis and renal injury, cardiac dysfunction and arrhythmia, severe electrolyte abnormalities, acute respiratory distress syndrome, lactic acidosis, and other forms of end-organ failure (Figure).
Rapid cooling is more crucial than transport; transport poses a risk of delayed cooling, which can dramatically increase an individual’s risk of morbidity and mortality.20,23 In situations where a patient can be cooled on-site, physicians should pursue cooling before transporting the patient to a medical treatment facility.
Emergency Action Plan
Team physicians should be proactive in developing an emergency action plan to address possible EHS events. These plans should be site-specific, addressing procedures for all practice and home competition locations.12 All competition venues should have a CWI tub on-site in events where there is an increased risk of EHS.12,15,20 This tub should be set up and functional for all high-risk activities, including practices.12
Following recognition of a potential case of EHS, treatment teams should have procedures in place to transport athletes to the treatment area, obtain rectal temperature, initiate rapid cooling, and stabilize the athlete for transport to an emergency department (ED) for further evaluation.12,15 A written record of treatments and medications provided during athlete stabilization should be maintained and transported with the athlete to the ED.15 A list of helpful equipment and supplies for treatment of EHS can be found in Table 5.
EHS is a unique life-threatening situation where it is best to treat the patient on the sideline before transport.15 Those athletes transported before cooling risk spending an increased amount of time above critical temperatures for cell damage, which has been associated with increased morbidity and mortality. This mantra of “cool first, transport second” cannot be overemphasized, as those individuals with EHS who present to the ED with a persisting rectal temperature >41°F may risk up to an 80% mortality rate.24 Conversely, a recent large, retrospective study of 274 EHS events sustained during the Falmouth Road Race found a 100% survival rate when athletes were rapidly identified via rectal thermometry and treated with aggressive, rapid cooling through CWI.21
Return to Play
Perhaps the most challenging and important role the team physician has is determining an athlete’s return to play following EHI, as there currently are no evidence-based guidelines for return to activity for these athletes.7 The decisions surrounding return to play are highly individualized, as recovery from EHS and heat injury is associated with the duration of internal body temperature elevation above the critical level (40°C).7,20 Guidelines for return to activity following recovery from EHI differ among experts and institutions.7,25 The general consensus from these guidelines is that, at minimum, athletes should not participate in any physical activity until they are asymptomatic and all blood tests have normalized.11 Following this asymptomatic period, most guidelines advocate for a slow, deliberate return to activity.11 The American College of Sports Medicine (ACSM) offers one reasonable approach to the returning athlete following EHS:7
- No exercise for at least 7 days following release from medical care.
- Follow-up with a physician 1 week after release from medical care for physical examination and any warranted lab or radiologic studies (based upon organ systems affected during EHS).
- Once cleared to return to activity, the athlete begins exercise in a cool environment, gradually increasing the duration, intensity, and heat exposure over 2 weeks to demonstrate heat tolerance and acclimatization.
- Athletes who cannot resume vigorous activity due to recurrent symptoms (eg, excessive fatigue) should be reevaluated after 4 weeks. Laboratory exercise-heat tolerance testing may be useful in this setting.
- The athlete may resume full competition once they are able to participate in full training in the heat for 2 to 4 weeks without adverse effects.
Heat tolerance testing (HTT) in these athletes remains controversial.5 26 The ACSM recommends that HTT be considered only for those unable to return to vigorous activity after a suitable period (approximately 4 weeks). In contrast, the Israeli Defense Force (IDF) uses HTT to evaluate soldiers following EHS to guide decision-making about return to duty.27 The IDF HTT assumes that individuals will respond differently to heat stresses. They identify individuals who are “heat intolerant” as being unable to tolerate specific heat challenges, indicated by increases in body temperature occurring more rapidly than normal responders under identical environmental and exercise conditions. However, despite being used for more than 30 years, there is no clear evidence that HTT adequately predicts who will experience subsequent episodes of EHS.
Conclusion
While the recognized cornerstone of being a team physician is the provision of medical care, the ACSM Team Physician Consensus Statement28 further delineates the medical and administrative responsibilities as both (1) understanding medical management and prevention of injury and illness in athletes; and (2) awareness of or involvement in the development and rehearsal of an emergency action plan. These tenets are critical for the team physician who accepts the responsibility to cover sports at the high school level or higher. Football team physicians play an essential role in mitigating risk of EHI in their athletes. Through development and execution of both comprehensive prevention strategies and emergency action plans, physicians can work to minimize athletes’ risk of both developing and experiencing significant adverse outcomes from an EHI.
Am J Orthop. 2016;45(6):340-348. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Kucera KL, Klossner D, Colgate B, Cantu RC. Annual Survey of Football Injury Research: 1931-2014. National Center for Catastrophic Sport Injury Research Web site. https://nccsir.unc.edu/files/2013/10/Annual-Football-2014-Fatalities-Final.pdf. Accessed May 31, 2016.
2. Boden BP, Breit I, Beachler JA, Williams A, Mueller FO. Fatalities in high school and college football players. Am J Sports Med. 2013;41(5):1108-1116.
3. Bouchama A, Knochel JP. Heat stroke. N Engl J Med. 2002;346(25):1978-1988.
4. Racinais S, Alonso JM, Coutts AJ, et al. Consensus recommendations on training and competing in the heat. Scand J Med Sci Sports. 2015;25 Suppl 1:6-19.
5. Pryor RR, Casa DJ, Adams WM, et al. Maximizing athletic performance in the heat. Strength Cond J. 2013;35(6):24-33.
6. Adams WM, Casa DJ. Hydration for football athletes. Sports Sci Exchange. 2015;28(141):1-5.
7. American College of Sports Medicine, Armstrong LE, Casa DJ, et al. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc. 2007;39(3):556-572.
8. Yard EE, Gilchrist J, Haileyesus T, et al. Heat illness among high school athletes--United States, 2005-2009. J Safety Res. 2010;41(6):471-474.
9. Huffman EA, Yard EE, Fields SK, Collins CL, Comstock RD. Epidemiology of rare injuries and conditions among United States high school athletes during the 2005-2006 and 2006-2007 school years. J Athl Train. 2008;43(6):624-630.
10. Kerr ZY, Casa DJ, Marshall SW, Comstock RD. Epidemiology of exertional heat illness among U.S. high school athletes. Am J Prev Med. 2013;44(1):8-14.
11. Casa DJ, DeMartini JK, Bergeron MF, et al. National Athletic Trainers’ Association position statement: exertional heat illnesses. J Athl Train. 2015;50(9):986-1000.
12. Casa DJ, Almquist J, Anderson SA. The inter-association task force for preventing sudden death in secondary school athletics programs: best-practices recommendations. J Athl Train. 2013;48(4):546-553.
13. Gardner JW, Kark JA, Karnei K, et al. Risk factors predicting exertional heat illness in male Marine Corps recruits. Med Sci Sports Exerc. 1996;28(8):939-944.
14. Gundstein AJ, Ramseyer C, Zhao F, et al. A retrospective analysis of American football hyperthermia deaths in the United States. Int J Biometerol. 2012;56(1):11-20.
15. Armstrong LE, Johnson EC, Casa DJ, et al. The American football uniform: uncompensable heat stress and hyperthermic exhaustion. J Athl Train. 2010;45(2):117-127.
16. Casa DJ, Csillan D; Inter-Association Task Force for Preseason Secondary School Athletics Participants, et al. Preseason heat-acclimatization guidelines for secondary school athletics. J Athl Train. 2009;44(3):332-333.
17. Godek SF, Godek JJ, Bartolozzi AR. Hydration status in college football players during consecutive days of twice-a-day preseason practices. Am J Sports Med. 2005;33(6):843-851.
18. Stover EA, Zachwieja J, Stofan J, Murray R, Horswill CA. Consistently high urine specific gravity in adolescent American football players and the impact of an acute drinking strategy. Int J Sports Med. 2006;27(4):330-335.
19. Bongers CC, Thijssen DH, Veltmeijer MTW, Hopman MT, Eijsvogels TM. Precooling and percooling (cooling during exercise) both improve performance in the heat: a meta-analytical review. Br J Sports Med. 2015;49(6):377-384.
20. Casa DJ, McDermott BP, Lee EC, Yeargin SW, Armstrong LE, Maresh CM. Cold water immersion: the gold standard for exertional heatstroke treatment. Exerc Sport Sci Rev. 2007;35(3):141-149.
21. DeMartini JK, Casa DJ, Stearns R, et al. Effectiveness of cold water immersion in the treatment of exertional heat stroke at the Falmouth Road Race. Med Sci Sports Exerc. 2015;47(2):240-245.
22. Casa DJ, Kenny GP, Taylor NA. Immersion treatment for exertional hyperthermia: cold or temperate water? Med Sci Sports Exerc. 2010;42(7):1246-1252.
23. Casa DJ, Armstrong LE, Kenny GP, O’Connor FG, Huggins RA. Exertional heat stroke: new concepts regarding cause and care. Curr Sports Med Rep. 2012;11(3):115-122.
24. Argaud L, Ferry T, Le QH, et al. Short- and long-term outcomes of heat stroke following the 2003 heat wave in Lyon, France. Arch Intern Med. 2007;167(20):2177-2183.
25. O’Connor FG, Casa DJ, Bergeron MF, et al. American College of Sports Medicine Roundtable on exertional heat stroke--return to duty/return to play: conference proceedings. Curr Sports Med Rep. 2010;9(5):314-321.
26. Kazman JB, Heled Y, Lisman PJ, Druyan A, Deuster PA, O’Connor FG. Exertional heat illness: the role of heat tolerance testing. Curr Sports Med Rep. 2013;12(2):101-105.
27. Moran DS, Heled Y, Still L, Laor A, Shapiro Y. Assessment of heat tolerance for post exertional heat stroke individuals. Med Sci Monit. 2004;10(6):CR252-CR257.
28. Herring SA, Kibler WB, Putukian M. Team Physician Consensus Statement: 2013 update. Med Sci Sports Exerc. 2013;45(8):1618-1622.
29. Heat stroke treatment. Korey Stringer Institute University of Connecticut Web site. http://ksi.uconn.edu/emergency-conditions/heat-illnesses/exertional-heat-stroke/heat-stroke-treatment/. Accessed June 14, 2016.
30. Headquarters, Department of the Army and the Air Force. Heat Stress Control and Heat Casualty Management. Technical Bulletin Medical 507. http://www.dir.ca.gov/oshsb/documents/Heat_illness_prevention_tbmed507.pdf. Published March 7, 2003. Accessed June 14, 2016.
Football, one of the most popular sports in the United States, is additionally recognized as a leading contributor to sports injury secondary to the contact collision nature of the endeavor. There are an estimated 1.1 million high school football players with another 100,000 participants combined in the National Football League (NFL), college, junior college, Arena Football League, and semipro levels of play.1 USA Football estimates that an additional 3 million youth participate in community football leagues.1 The National Center for Catastrophic Sports Injury Research recently calculated a fatality rate of 0.14 per 100,000 participants in 2014 for the 4.2 million who play football at all levels—and 0.45 per 100,000 in high school.1 While direct deaths from head and spine injury remain a significant contributor to the number of catastrophic injuries, indirect deaths (systemic failure) predominate. Exertional heat stroke (EHS) has emerged as one of the leading indirect causes of death in high school and collegiate football. Boden and colleagues2 reported that high school and college football players sustain approximately 12 fatalities annually, with indirect systemic causes being twice as common as direct blunt trauma.2The most common indirect causes identified included cardiac failure, heat illness, and complications of sickle cell trait (SCT). It was also noted that the risk of SCT, heat-related, and cardiac deaths increased during the second decade of the study, indicating these conditions may require a greater emphasis on diagnosis, treatment, and prevention. This review details for the team physician the unique challenge of exercising in the heat to the football player, and the prevention, diagnosis, management and return-to-play issues pertinent to exertional heat illness (EHI).
The Challenge
EHS represents the most severe manifestation of EHI—a gamut of diseases commonly encountered during the hot summer months when American football season begins. The breadth of EHI includes several important clinical diagnoses: exercise-associated muscle cramps (heat cramps); heat exhaustion with and without syncope; heat injury with evidence of end organ injury (eg, rhabdomyolysis); and EHS. EHS is defined as “a form of hyperthermia associated with a systemic inflammatory response leading to a syndrome of multi-organ dysfunction in which encephalopathy predominates.”3 EHS, if left untreated, or even if clinical treatment is delayed, may result in significant end organ morbidity and/or mortality.
During exercise, the human thermoregulatory system mitigates heat gain by increasing skin blood flow and sweating, causing an increased dissipation of heat to the surrounding environment by leveraging conduction, convection, and evaporation.4,5 Elevated environmental temperatures, increased humidity, and dehydration can impede the body’s ability to dissipate heat at a rate needed to maintain thermoregulation. This imbalance can result in hyperthermia secondary to uncompensated heat stress,5 which in turn can lead to EHI. Football players have unique challenges that make them particularly vulnerable to EHI. The summer heat during early-season participation and the requirement for equipment that covers nearly 60% of body surfaces pose increased risk of volume losses and hyperthermia that trigger the onset of EHI.6 Football athletes’ body compositions and physical size are additional contributing risk factors; the relatively high muscle and fat content increase thermogenicity, which require their bodies to dissipate more heat.7
An estimated 9000 cases of EHI occur annually across all high school sports,8 with an incidence of 1.6:100,000 athlete-exposures.8,9 Studies have demonstrated, however, that EHI occurs in football 11.4 times more often than in all other high school sports combined.10 The incidence of nonfatal EHI in all levels of football is 4.42-5:100,000.8,9 Between 2000 and 2014, 41 football players died from EHS.1 In football, approximately 75% of all EHI events occurred during practices, while only 25% of incidents occurred during games.8
Given these potentially deadly consequences, it is important that football team physicians are not only alert to the early symptoms of heat illness and prepared to intervene to prevent the progression to EHS, but are critical leaders in educating coaches and players in evidence-based EHI prevention practices and policies.
Prevention
EHS is a preventable condition, arguably the most common cause of preventable nontraumatic exertional death in young athletes in the United States. Close attention to mitigating risk factors should begin prior to the onset of preseason practice and continue through the early season, where athletes are at the highest risk of developing heat illness.
Primary Prevention
Primary prevention is fundamental to minimizing the occurrences of EHI. It focuses on the following methods: recognition of inherent risk factors, acclimatization, hydration, and avoidance of inciting substances (including supplements).
Pre-Participation Examination. The purpose of the pre-participation examination (PPE) is to maximize an athlete’s safety by identifying medical conditions that place the athlete at risk.11,12 The Preparticipation Physical Evaluation, 4th edition, the most widely used consensus publication, specifically queries if an athlete has a previous history of heat injury. However, it only indirectly addresses intrinsic risk factors that may predispose an athlete to EHI who has never had an EHI before. Therefore, providers should take the opportunity of the PPE to inquire about additional risk factors that may make an athlete high risk for sustaining a heat injury. Common risk factors for EHI are listed in Table 1.
Heat Acclimatization. The risk of EHI escalates significantly when athletes are subjected to multiple stressors during periods of heat exposure, such as sudden increases in intensity or duration of exercise; prolonged new exposures to heat; dehydration; and sleep loss.5 When football season begins in late summer, athletes are least conditioned as temperatures reach their seasonal peak, causing increased risk of EHI.15 Planning for heat acclimatization is vital for all athletes who exercise in hot environments. Acclimatization procedures place progressively mounting physiologic strains on the body to improve athletes’ ability to dissipate heat, diminishing thermoregulatory and cardiovascular exertion.4,5 Acclimatization begins with expansion of plasma volume on days 3 to 6, causing improvements in cardiac efficiency and resulting in an overall decrease in basal internal body temperature.4,5,15 This process results in improvements in heat tolerance and exercise performance, evolving over 10 to 14 days of gradual escalation of exercise intensity and duration.5,10,11,16 However, poor fitness levels and extreme temperatures can prolong this period, requiring up to 2 to 3 months to fully take effect.5,7
The National Athletic Trainers Association (NATA) and National Collegiate Athletic Association (NCAA) have released consensus guidelines regarding heat acclimatization protocols for football athletes at the high school and college levels (Tables 3 and 4). Each of these guidelines involves an initial period without use of protective equipment, followed by a gradual addition of further equipment.11,16
Secondary Prevention
Despite physicians’ best efforts to prevent all cases of EHI, athletes will still experience the effects of exercise-induced hyperthermia. The goal of secondary prevention is to slow the progression of this hyperthermia so that it does not progress to more dangerous EHI.
Hydration. Dehydration is an important risk factor for EHI. Sweat maintains thermoregulation by dissipating heat generated during exercise; however, it also contributes to body water losses. Furthermore, intravascular depletion decreases stroke volume, thereby increasing cardiovascular strain. It is estimated that for every 1% loss in body mass from dehydration, body temperature rises 0.22°C in comparison to a euhydrated state.6 Dehydration occurs more rapidly in hot environments, as fluid is lost through increased sweat production.7 After approximately 6% to 10% body weight volume loss, cardiac output cannot be maintained, diminishing sweat production and blood flow to both skin and muscle and causing diminished performance and a significant risk of heat exhaustion.7 If left unchecked, these physiologic changes result in further elevations in body temperature and increased cardiovascular strain, ultimately placing the athlete at significant risk for development of EHS.
Adequate hydration to maintain euvolemia is an important step in avoiding possible EHI. Multiple studies have shown that football players experience a baseline hypovolemia during their competition season,6 a deficit that is most marked during the first week of practices.17 This deficit is multifactorial, as football players expend a significant amount of fluid through sweat, are not able to adequately replace these losses during practice, and do not appropriately hydrate off the field.6,18 Some players, especially linemen, sweat at a higher rate than their teammates, posing a possible risk of significant dehydration.6 Coaches and players alike should be educated on the importance of adequate hydration to meet their fluid needs.
The goal of hydration during exercise is to prevent large fluid losses that can adversely affect performance and increase risk of EHI;6 it may be unrealistic to replace all fluid losses during the practice period. Instead, athletes should target complete volume replacement over the post-exercise period.6 Some recommend hydrating based upon thirst drive; however, thirst is activated following a volume loss of approximately 2% body mass, the same degree of losses that place athletes at an increased risk for performance impairment and EHI.4,6,11,12 Individuals should have access to fluids throughout practice and competition and be encouraged to hydrate as needed.6,12,15 Furthermore, staff should modify their practices based upon WBGT and acclimatization status to provide more frequent hydration breaks.
Hyperhydration and Salt Intake. Of note, there are inherent risks to hyperhydration. Athletes with low sweat rates have an increased risk of overhydration and the development of exercise-associated hyponatremia (EAH),6 a condition whose presentation is very similar to EHS. In addition, inadequate sodium intake and excessive sweating can also contribute to the development of EAH. EAH has been implicated in the deaths of 2 football players in 2014.1,6 Establishing team hydration guidelines and educating players and staff on appropriate hydration and dietary salt intake is essential to reduce the risk of both dehydration and hyperhydration and their complications.6Intra-Event Cooling. During exercise, team physicians can employ strategies for cooling athletes during exertion to mitigate their risk of EHI by decreasing thermal and cardiovascular strain.4,19 Cooling during exercise is hypothesized to allow for accelerated heat dissipation, where heat is lost from the body more effectively. This accelerated loss enables athletes to maintain a higher heat storage capacity over the duration of exercise, avoiding uncompensated heat stresses that ultimately cause EHI.19
Some intra-event cooling strategies include the use of cooling garments, cooling packs, and cold water/slurry ingestion. Cooling garments lower skin temperature, which in turn can decrease thermoregulatory strains;4 a recent meta-analysis of intra-event cooling modalities revealed that wearing an ice vest during exercise resulted in the greatest decrease in thermal heat strain.19 Internal cooling strategies—namely ingestion of cold fluids/ice slurry—have shown some mild benefit in decreasing internal temperatures; however, some studies have demonstrated some decrease in sweat production associated with cold oral intake used in isolation.19 Overall, studies have shown that combining external (cooling clothing, ice packs, fanning) and internal (cold water, ice slurry) cooling methods result in a greater cooling effect than a use of a single method.4
Tertiary Prevention
The goal of tertiary prevention is to mitigate the risk of long-term adverse outcomes following an EHS event. The most effective means of reducing risk for morbidity and mortality is rapid identification and treatment of EHS as well as close evaluation of an athlete’s return to activity in heat. This process is spearheaded by an effective and well-rehearsed emergency action plan.
Diagnosis and Management
Rapid identification and treatment of EHS is crucial to minimizing the risk of poor outcomes.7 Any delay in the treatment of EHS can dramatically increase the likelihood of associated morbidity and mortality.20
EHS is diagnosed by an elevated rectal temperature ≥40°C (104°F) and associated central nervous system (CNS) dysfunction.21 EHS should be strongly suspected in any athlete exercising in heat who exhibits signs of CNS dysfunction, including disorientation, confusion, dizziness, erratic behavior, irritability, headache, loss of coordination, delirium, collapse, or seizures.7,12,15 EHS may also present with symptoms of heat exhaustion, including fatigue, hyperventilation, tachycardia, vomiting, diarrhea, and hypotension.7,12,15
Rectal temperature should be taken for any athlete with suspected EHS, as other modalities—oral, skin, axillary, and aural—can be inaccurate and easily modified by ambient confounders such as ambient and skin temperature, athlete hyperventilation, and consumption of liquids.7,11,12 Athletes exhibiting CNS symptoms with moderately elevated rectal temperatures that do not exceed 40°C should also be assumed to be suffering from EHS and treated with rapid cooling.11 On the other hand, athletes with CNS symptoms who are normothermic should be assumed to have EAH until ruled out by electrolyte assessment; IV fluids should be at no more than keep vein open (KVO) pending this determination.11 In some cases, an athlete may initially present with altered mental status but return to “normal.” However, this improvement may represent a “lucid period”; evaluation should continue with rectal temperature and treatment, as EHS in these cases may progress quickly.15
Treatment is centered on rapid, whole body cooling initiated at the first sign of heat illness.7,22 The goal of treatment is to achieve a rectal temperature <38.9°C within 30 minutes of the onset of EHS.15 Upon diagnosis, the athlete should be quickly placed in a tub of ice water to facilitate cold water immersion (CWI) therapy. Some guidelines suggest the athlete’s clothing be removed to potentiate evaporative cooling during CWI;12 however, cooling should not be delayed due to difficulties in removing equipment. CWI, where a heat stroke victim is submerged in ice water up to their neck while water is continuously circulated, is generally considered to be the gold standard treatment as it is the modality with the highest recorded cooling rates and the lowest rate of morbidity and mortality.7,20,21 Multiple studies of CWI have shown that survival nears 100% when aggressive cooling starts within 5 minutes of collapse or identification of EHS.20,21,22
If whole body CWI is unavailable, alternative methods of rapid cooling should be employed. Partial CWI, with torso immersion being preferable to the extremities, has been shown to achieve an acceptable rate of cooling to achieve sufficient drops in internal body temperature.20,23 However, one popular treatment—applying ice packs to the whole body, in particular to the groin and axillae—has not been shown to be sufficient to achieve standard cooling goals.20 None of these methods have been shown to be as effective as CWI.23
Intravenous access should be initiated with fluid resuscitation dictated by the provider’s assessment. Normal saline is recommended as the resuscitative fluid of choice, with the rate dictated by clinical judgment and adjusted as guided by electrolyte determination and clinical response. It cannot be overstated that in normothermic patients with confusion, EAH is the diagnosis of exclusion and aggressive fluid resuscitation should be withheld until electrolyte determination.
Once rectal temperature descends appropriately (~38.9°C), the cooling process should stop and the individual should be transported to a hospital for further observation20 and evaluation of possible sequelae, including rhabdomyolysis and renal injury, cardiac dysfunction and arrhythmia, severe electrolyte abnormalities, acute respiratory distress syndrome, lactic acidosis, and other forms of end-organ failure (Figure).
Rapid cooling is more crucial than transport; transport poses a risk of delayed cooling, which can dramatically increase an individual’s risk of morbidity and mortality.20,23 In situations where a patient can be cooled on-site, physicians should pursue cooling before transporting the patient to a medical treatment facility.
Emergency Action Plan
Team physicians should be proactive in developing an emergency action plan to address possible EHS events. These plans should be site-specific, addressing procedures for all practice and home competition locations.12 All competition venues should have a CWI tub on-site in events where there is an increased risk of EHS.12,15,20 This tub should be set up and functional for all high-risk activities, including practices.12
Following recognition of a potential case of EHS, treatment teams should have procedures in place to transport athletes to the treatment area, obtain rectal temperature, initiate rapid cooling, and stabilize the athlete for transport to an emergency department (ED) for further evaluation.12,15 A written record of treatments and medications provided during athlete stabilization should be maintained and transported with the athlete to the ED.15 A list of helpful equipment and supplies for treatment of EHS can be found in Table 5.
EHS is a unique life-threatening situation where it is best to treat the patient on the sideline before transport.15 Those athletes transported before cooling risk spending an increased amount of time above critical temperatures for cell damage, which has been associated with increased morbidity and mortality. This mantra of “cool first, transport second” cannot be overemphasized, as those individuals with EHS who present to the ED with a persisting rectal temperature >41°F may risk up to an 80% mortality rate.24 Conversely, a recent large, retrospective study of 274 EHS events sustained during the Falmouth Road Race found a 100% survival rate when athletes were rapidly identified via rectal thermometry and treated with aggressive, rapid cooling through CWI.21
Return to Play
Perhaps the most challenging and important role the team physician has is determining an athlete’s return to play following EHI, as there currently are no evidence-based guidelines for return to activity for these athletes.7 The decisions surrounding return to play are highly individualized, as recovery from EHS and heat injury is associated with the duration of internal body temperature elevation above the critical level (40°C).7,20 Guidelines for return to activity following recovery from EHI differ among experts and institutions.7,25 The general consensus from these guidelines is that, at minimum, athletes should not participate in any physical activity until they are asymptomatic and all blood tests have normalized.11 Following this asymptomatic period, most guidelines advocate for a slow, deliberate return to activity.11 The American College of Sports Medicine (ACSM) offers one reasonable approach to the returning athlete following EHS:7
- No exercise for at least 7 days following release from medical care.
- Follow-up with a physician 1 week after release from medical care for physical examination and any warranted lab or radiologic studies (based upon organ systems affected during EHS).
- Once cleared to return to activity, the athlete begins exercise in a cool environment, gradually increasing the duration, intensity, and heat exposure over 2 weeks to demonstrate heat tolerance and acclimatization.
- Athletes who cannot resume vigorous activity due to recurrent symptoms (eg, excessive fatigue) should be reevaluated after 4 weeks. Laboratory exercise-heat tolerance testing may be useful in this setting.
- The athlete may resume full competition once they are able to participate in full training in the heat for 2 to 4 weeks without adverse effects.
Heat tolerance testing (HTT) in these athletes remains controversial.5 26 The ACSM recommends that HTT be considered only for those unable to return to vigorous activity after a suitable period (approximately 4 weeks). In contrast, the Israeli Defense Force (IDF) uses HTT to evaluate soldiers following EHS to guide decision-making about return to duty.27 The IDF HTT assumes that individuals will respond differently to heat stresses. They identify individuals who are “heat intolerant” as being unable to tolerate specific heat challenges, indicated by increases in body temperature occurring more rapidly than normal responders under identical environmental and exercise conditions. However, despite being used for more than 30 years, there is no clear evidence that HTT adequately predicts who will experience subsequent episodes of EHS.
Conclusion
While the recognized cornerstone of being a team physician is the provision of medical care, the ACSM Team Physician Consensus Statement28 further delineates the medical and administrative responsibilities as both (1) understanding medical management and prevention of injury and illness in athletes; and (2) awareness of or involvement in the development and rehearsal of an emergency action plan. These tenets are critical for the team physician who accepts the responsibility to cover sports at the high school level or higher. Football team physicians play an essential role in mitigating risk of EHI in their athletes. Through development and execution of both comprehensive prevention strategies and emergency action plans, physicians can work to minimize athletes’ risk of both developing and experiencing significant adverse outcomes from an EHI.
Am J Orthop. 2016;45(6):340-348. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
Football, one of the most popular sports in the United States, is additionally recognized as a leading contributor to sports injury secondary to the contact collision nature of the endeavor. There are an estimated 1.1 million high school football players with another 100,000 participants combined in the National Football League (NFL), college, junior college, Arena Football League, and semipro levels of play.1 USA Football estimates that an additional 3 million youth participate in community football leagues.1 The National Center for Catastrophic Sports Injury Research recently calculated a fatality rate of 0.14 per 100,000 participants in 2014 for the 4.2 million who play football at all levels—and 0.45 per 100,000 in high school.1 While direct deaths from head and spine injury remain a significant contributor to the number of catastrophic injuries, indirect deaths (systemic failure) predominate. Exertional heat stroke (EHS) has emerged as one of the leading indirect causes of death in high school and collegiate football. Boden and colleagues2 reported that high school and college football players sustain approximately 12 fatalities annually, with indirect systemic causes being twice as common as direct blunt trauma.2The most common indirect causes identified included cardiac failure, heat illness, and complications of sickle cell trait (SCT). It was also noted that the risk of SCT, heat-related, and cardiac deaths increased during the second decade of the study, indicating these conditions may require a greater emphasis on diagnosis, treatment, and prevention. This review details for the team physician the unique challenge of exercising in the heat to the football player, and the prevention, diagnosis, management and return-to-play issues pertinent to exertional heat illness (EHI).
The Challenge
EHS represents the most severe manifestation of EHI—a gamut of diseases commonly encountered during the hot summer months when American football season begins. The breadth of EHI includes several important clinical diagnoses: exercise-associated muscle cramps (heat cramps); heat exhaustion with and without syncope; heat injury with evidence of end organ injury (eg, rhabdomyolysis); and EHS. EHS is defined as “a form of hyperthermia associated with a systemic inflammatory response leading to a syndrome of multi-organ dysfunction in which encephalopathy predominates.”3 EHS, if left untreated, or even if clinical treatment is delayed, may result in significant end organ morbidity and/or mortality.
During exercise, the human thermoregulatory system mitigates heat gain by increasing skin blood flow and sweating, causing an increased dissipation of heat to the surrounding environment by leveraging conduction, convection, and evaporation.4,5 Elevated environmental temperatures, increased humidity, and dehydration can impede the body’s ability to dissipate heat at a rate needed to maintain thermoregulation. This imbalance can result in hyperthermia secondary to uncompensated heat stress,5 which in turn can lead to EHI. Football players have unique challenges that make them particularly vulnerable to EHI. The summer heat during early-season participation and the requirement for equipment that covers nearly 60% of body surfaces pose increased risk of volume losses and hyperthermia that trigger the onset of EHI.6 Football athletes’ body compositions and physical size are additional contributing risk factors; the relatively high muscle and fat content increase thermogenicity, which require their bodies to dissipate more heat.7
An estimated 9000 cases of EHI occur annually across all high school sports,8 with an incidence of 1.6:100,000 athlete-exposures.8,9 Studies have demonstrated, however, that EHI occurs in football 11.4 times more often than in all other high school sports combined.10 The incidence of nonfatal EHI in all levels of football is 4.42-5:100,000.8,9 Between 2000 and 2014, 41 football players died from EHS.1 In football, approximately 75% of all EHI events occurred during practices, while only 25% of incidents occurred during games.8
Given these potentially deadly consequences, it is important that football team physicians are not only alert to the early symptoms of heat illness and prepared to intervene to prevent the progression to EHS, but are critical leaders in educating coaches and players in evidence-based EHI prevention practices and policies.
Prevention
EHS is a preventable condition, arguably the most common cause of preventable nontraumatic exertional death in young athletes in the United States. Close attention to mitigating risk factors should begin prior to the onset of preseason practice and continue through the early season, where athletes are at the highest risk of developing heat illness.
Primary Prevention
Primary prevention is fundamental to minimizing the occurrences of EHI. It focuses on the following methods: recognition of inherent risk factors, acclimatization, hydration, and avoidance of inciting substances (including supplements).
Pre-Participation Examination. The purpose of the pre-participation examination (PPE) is to maximize an athlete’s safety by identifying medical conditions that place the athlete at risk.11,12 The Preparticipation Physical Evaluation, 4th edition, the most widely used consensus publication, specifically queries if an athlete has a previous history of heat injury. However, it only indirectly addresses intrinsic risk factors that may predispose an athlete to EHI who has never had an EHI before. Therefore, providers should take the opportunity of the PPE to inquire about additional risk factors that may make an athlete high risk for sustaining a heat injury. Common risk factors for EHI are listed in Table 1.
Heat Acclimatization. The risk of EHI escalates significantly when athletes are subjected to multiple stressors during periods of heat exposure, such as sudden increases in intensity or duration of exercise; prolonged new exposures to heat; dehydration; and sleep loss.5 When football season begins in late summer, athletes are least conditioned as temperatures reach their seasonal peak, causing increased risk of EHI.15 Planning for heat acclimatization is vital for all athletes who exercise in hot environments. Acclimatization procedures place progressively mounting physiologic strains on the body to improve athletes’ ability to dissipate heat, diminishing thermoregulatory and cardiovascular exertion.4,5 Acclimatization begins with expansion of plasma volume on days 3 to 6, causing improvements in cardiac efficiency and resulting in an overall decrease in basal internal body temperature.4,5,15 This process results in improvements in heat tolerance and exercise performance, evolving over 10 to 14 days of gradual escalation of exercise intensity and duration.5,10,11,16 However, poor fitness levels and extreme temperatures can prolong this period, requiring up to 2 to 3 months to fully take effect.5,7
The National Athletic Trainers Association (NATA) and National Collegiate Athletic Association (NCAA) have released consensus guidelines regarding heat acclimatization protocols for football athletes at the high school and college levels (Tables 3 and 4). Each of these guidelines involves an initial period without use of protective equipment, followed by a gradual addition of further equipment.11,16
Secondary Prevention
Despite physicians’ best efforts to prevent all cases of EHI, athletes will still experience the effects of exercise-induced hyperthermia. The goal of secondary prevention is to slow the progression of this hyperthermia so that it does not progress to more dangerous EHI.
Hydration. Dehydration is an important risk factor for EHI. Sweat maintains thermoregulation by dissipating heat generated during exercise; however, it also contributes to body water losses. Furthermore, intravascular depletion decreases stroke volume, thereby increasing cardiovascular strain. It is estimated that for every 1% loss in body mass from dehydration, body temperature rises 0.22°C in comparison to a euhydrated state.6 Dehydration occurs more rapidly in hot environments, as fluid is lost through increased sweat production.7 After approximately 6% to 10% body weight volume loss, cardiac output cannot be maintained, diminishing sweat production and blood flow to both skin and muscle and causing diminished performance and a significant risk of heat exhaustion.7 If left unchecked, these physiologic changes result in further elevations in body temperature and increased cardiovascular strain, ultimately placing the athlete at significant risk for development of EHS.
Adequate hydration to maintain euvolemia is an important step in avoiding possible EHI. Multiple studies have shown that football players experience a baseline hypovolemia during their competition season,6 a deficit that is most marked during the first week of practices.17 This deficit is multifactorial, as football players expend a significant amount of fluid through sweat, are not able to adequately replace these losses during practice, and do not appropriately hydrate off the field.6,18 Some players, especially linemen, sweat at a higher rate than their teammates, posing a possible risk of significant dehydration.6 Coaches and players alike should be educated on the importance of adequate hydration to meet their fluid needs.
The goal of hydration during exercise is to prevent large fluid losses that can adversely affect performance and increase risk of EHI;6 it may be unrealistic to replace all fluid losses during the practice period. Instead, athletes should target complete volume replacement over the post-exercise period.6 Some recommend hydrating based upon thirst drive; however, thirst is activated following a volume loss of approximately 2% body mass, the same degree of losses that place athletes at an increased risk for performance impairment and EHI.4,6,11,12 Individuals should have access to fluids throughout practice and competition and be encouraged to hydrate as needed.6,12,15 Furthermore, staff should modify their practices based upon WBGT and acclimatization status to provide more frequent hydration breaks.
Hyperhydration and Salt Intake. Of note, there are inherent risks to hyperhydration. Athletes with low sweat rates have an increased risk of overhydration and the development of exercise-associated hyponatremia (EAH),6 a condition whose presentation is very similar to EHS. In addition, inadequate sodium intake and excessive sweating can also contribute to the development of EAH. EAH has been implicated in the deaths of 2 football players in 2014.1,6 Establishing team hydration guidelines and educating players and staff on appropriate hydration and dietary salt intake is essential to reduce the risk of both dehydration and hyperhydration and their complications.6Intra-Event Cooling. During exercise, team physicians can employ strategies for cooling athletes during exertion to mitigate their risk of EHI by decreasing thermal and cardiovascular strain.4,19 Cooling during exercise is hypothesized to allow for accelerated heat dissipation, where heat is lost from the body more effectively. This accelerated loss enables athletes to maintain a higher heat storage capacity over the duration of exercise, avoiding uncompensated heat stresses that ultimately cause EHI.19
Some intra-event cooling strategies include the use of cooling garments, cooling packs, and cold water/slurry ingestion. Cooling garments lower skin temperature, which in turn can decrease thermoregulatory strains;4 a recent meta-analysis of intra-event cooling modalities revealed that wearing an ice vest during exercise resulted in the greatest decrease in thermal heat strain.19 Internal cooling strategies—namely ingestion of cold fluids/ice slurry—have shown some mild benefit in decreasing internal temperatures; however, some studies have demonstrated some decrease in sweat production associated with cold oral intake used in isolation.19 Overall, studies have shown that combining external (cooling clothing, ice packs, fanning) and internal (cold water, ice slurry) cooling methods result in a greater cooling effect than a use of a single method.4
Tertiary Prevention
The goal of tertiary prevention is to mitigate the risk of long-term adverse outcomes following an EHS event. The most effective means of reducing risk for morbidity and mortality is rapid identification and treatment of EHS as well as close evaluation of an athlete’s return to activity in heat. This process is spearheaded by an effective and well-rehearsed emergency action plan.
Diagnosis and Management
Rapid identification and treatment of EHS is crucial to minimizing the risk of poor outcomes.7 Any delay in the treatment of EHS can dramatically increase the likelihood of associated morbidity and mortality.20
EHS is diagnosed by an elevated rectal temperature ≥40°C (104°F) and associated central nervous system (CNS) dysfunction.21 EHS should be strongly suspected in any athlete exercising in heat who exhibits signs of CNS dysfunction, including disorientation, confusion, dizziness, erratic behavior, irritability, headache, loss of coordination, delirium, collapse, or seizures.7,12,15 EHS may also present with symptoms of heat exhaustion, including fatigue, hyperventilation, tachycardia, vomiting, diarrhea, and hypotension.7,12,15
Rectal temperature should be taken for any athlete with suspected EHS, as other modalities—oral, skin, axillary, and aural—can be inaccurate and easily modified by ambient confounders such as ambient and skin temperature, athlete hyperventilation, and consumption of liquids.7,11,12 Athletes exhibiting CNS symptoms with moderately elevated rectal temperatures that do not exceed 40°C should also be assumed to be suffering from EHS and treated with rapid cooling.11 On the other hand, athletes with CNS symptoms who are normothermic should be assumed to have EAH until ruled out by electrolyte assessment; IV fluids should be at no more than keep vein open (KVO) pending this determination.11 In some cases, an athlete may initially present with altered mental status but return to “normal.” However, this improvement may represent a “lucid period”; evaluation should continue with rectal temperature and treatment, as EHS in these cases may progress quickly.15
Treatment is centered on rapid, whole body cooling initiated at the first sign of heat illness.7,22 The goal of treatment is to achieve a rectal temperature <38.9°C within 30 minutes of the onset of EHS.15 Upon diagnosis, the athlete should be quickly placed in a tub of ice water to facilitate cold water immersion (CWI) therapy. Some guidelines suggest the athlete’s clothing be removed to potentiate evaporative cooling during CWI;12 however, cooling should not be delayed due to difficulties in removing equipment. CWI, where a heat stroke victim is submerged in ice water up to their neck while water is continuously circulated, is generally considered to be the gold standard treatment as it is the modality with the highest recorded cooling rates and the lowest rate of morbidity and mortality.7,20,21 Multiple studies of CWI have shown that survival nears 100% when aggressive cooling starts within 5 minutes of collapse or identification of EHS.20,21,22
If whole body CWI is unavailable, alternative methods of rapid cooling should be employed. Partial CWI, with torso immersion being preferable to the extremities, has been shown to achieve an acceptable rate of cooling to achieve sufficient drops in internal body temperature.20,23 However, one popular treatment—applying ice packs to the whole body, in particular to the groin and axillae—has not been shown to be sufficient to achieve standard cooling goals.20 None of these methods have been shown to be as effective as CWI.23
Intravenous access should be initiated with fluid resuscitation dictated by the provider’s assessment. Normal saline is recommended as the resuscitative fluid of choice, with the rate dictated by clinical judgment and adjusted as guided by electrolyte determination and clinical response. It cannot be overstated that in normothermic patients with confusion, EAH is the diagnosis of exclusion and aggressive fluid resuscitation should be withheld until electrolyte determination.
Once rectal temperature descends appropriately (~38.9°C), the cooling process should stop and the individual should be transported to a hospital for further observation20 and evaluation of possible sequelae, including rhabdomyolysis and renal injury, cardiac dysfunction and arrhythmia, severe electrolyte abnormalities, acute respiratory distress syndrome, lactic acidosis, and other forms of end-organ failure (Figure).
Rapid cooling is more crucial than transport; transport poses a risk of delayed cooling, which can dramatically increase an individual’s risk of morbidity and mortality.20,23 In situations where a patient can be cooled on-site, physicians should pursue cooling before transporting the patient to a medical treatment facility.
Emergency Action Plan
Team physicians should be proactive in developing an emergency action plan to address possible EHS events. These plans should be site-specific, addressing procedures for all practice and home competition locations.12 All competition venues should have a CWI tub on-site in events where there is an increased risk of EHS.12,15,20 This tub should be set up and functional for all high-risk activities, including practices.12
Following recognition of a potential case of EHS, treatment teams should have procedures in place to transport athletes to the treatment area, obtain rectal temperature, initiate rapid cooling, and stabilize the athlete for transport to an emergency department (ED) for further evaluation.12,15 A written record of treatments and medications provided during athlete stabilization should be maintained and transported with the athlete to the ED.15 A list of helpful equipment and supplies for treatment of EHS can be found in Table 5.
EHS is a unique life-threatening situation where it is best to treat the patient on the sideline before transport.15 Those athletes transported before cooling risk spending an increased amount of time above critical temperatures for cell damage, which has been associated with increased morbidity and mortality. This mantra of “cool first, transport second” cannot be overemphasized, as those individuals with EHS who present to the ED with a persisting rectal temperature >41°F may risk up to an 80% mortality rate.24 Conversely, a recent large, retrospective study of 274 EHS events sustained during the Falmouth Road Race found a 100% survival rate when athletes were rapidly identified via rectal thermometry and treated with aggressive, rapid cooling through CWI.21
Return to Play
Perhaps the most challenging and important role the team physician has is determining an athlete’s return to play following EHI, as there currently are no evidence-based guidelines for return to activity for these athletes.7 The decisions surrounding return to play are highly individualized, as recovery from EHS and heat injury is associated with the duration of internal body temperature elevation above the critical level (40°C).7,20 Guidelines for return to activity following recovery from EHI differ among experts and institutions.7,25 The general consensus from these guidelines is that, at minimum, athletes should not participate in any physical activity until they are asymptomatic and all blood tests have normalized.11 Following this asymptomatic period, most guidelines advocate for a slow, deliberate return to activity.11 The American College of Sports Medicine (ACSM) offers one reasonable approach to the returning athlete following EHS:7
- No exercise for at least 7 days following release from medical care.
- Follow-up with a physician 1 week after release from medical care for physical examination and any warranted lab or radiologic studies (based upon organ systems affected during EHS).
- Once cleared to return to activity, the athlete begins exercise in a cool environment, gradually increasing the duration, intensity, and heat exposure over 2 weeks to demonstrate heat tolerance and acclimatization.
- Athletes who cannot resume vigorous activity due to recurrent symptoms (eg, excessive fatigue) should be reevaluated after 4 weeks. Laboratory exercise-heat tolerance testing may be useful in this setting.
- The athlete may resume full competition once they are able to participate in full training in the heat for 2 to 4 weeks without adverse effects.
Heat tolerance testing (HTT) in these athletes remains controversial.5 26 The ACSM recommends that HTT be considered only for those unable to return to vigorous activity after a suitable period (approximately 4 weeks). In contrast, the Israeli Defense Force (IDF) uses HTT to evaluate soldiers following EHS to guide decision-making about return to duty.27 The IDF HTT assumes that individuals will respond differently to heat stresses. They identify individuals who are “heat intolerant” as being unable to tolerate specific heat challenges, indicated by increases in body temperature occurring more rapidly than normal responders under identical environmental and exercise conditions. However, despite being used for more than 30 years, there is no clear evidence that HTT adequately predicts who will experience subsequent episodes of EHS.
Conclusion
While the recognized cornerstone of being a team physician is the provision of medical care, the ACSM Team Physician Consensus Statement28 further delineates the medical and administrative responsibilities as both (1) understanding medical management and prevention of injury and illness in athletes; and (2) awareness of or involvement in the development and rehearsal of an emergency action plan. These tenets are critical for the team physician who accepts the responsibility to cover sports at the high school level or higher. Football team physicians play an essential role in mitigating risk of EHI in their athletes. Through development and execution of both comprehensive prevention strategies and emergency action plans, physicians can work to minimize athletes’ risk of both developing and experiencing significant adverse outcomes from an EHI.
Am J Orthop. 2016;45(6):340-348. Copyright Frontline Medical Communications Inc. 2016. All rights reserved.
1. Kucera KL, Klossner D, Colgate B, Cantu RC. Annual Survey of Football Injury Research: 1931-2014. National Center for Catastrophic Sport Injury Research Web site. https://nccsir.unc.edu/files/2013/10/Annual-Football-2014-Fatalities-Final.pdf. Accessed May 31, 2016.
2. Boden BP, Breit I, Beachler JA, Williams A, Mueller FO. Fatalities in high school and college football players. Am J Sports Med. 2013;41(5):1108-1116.
3. Bouchama A, Knochel JP. Heat stroke. N Engl J Med. 2002;346(25):1978-1988.
4. Racinais S, Alonso JM, Coutts AJ, et al. Consensus recommendations on training and competing in the heat. Scand J Med Sci Sports. 2015;25 Suppl 1:6-19.
5. Pryor RR, Casa DJ, Adams WM, et al. Maximizing athletic performance in the heat. Strength Cond J. 2013;35(6):24-33.
6. Adams WM, Casa DJ. Hydration for football athletes. Sports Sci Exchange. 2015;28(141):1-5.
7. American College of Sports Medicine, Armstrong LE, Casa DJ, et al. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc. 2007;39(3):556-572.
8. Yard EE, Gilchrist J, Haileyesus T, et al. Heat illness among high school athletes--United States, 2005-2009. J Safety Res. 2010;41(6):471-474.
9. Huffman EA, Yard EE, Fields SK, Collins CL, Comstock RD. Epidemiology of rare injuries and conditions among United States high school athletes during the 2005-2006 and 2006-2007 school years. J Athl Train. 2008;43(6):624-630.
10. Kerr ZY, Casa DJ, Marshall SW, Comstock RD. Epidemiology of exertional heat illness among U.S. high school athletes. Am J Prev Med. 2013;44(1):8-14.
11. Casa DJ, DeMartini JK, Bergeron MF, et al. National Athletic Trainers’ Association position statement: exertional heat illnesses. J Athl Train. 2015;50(9):986-1000.
12. Casa DJ, Almquist J, Anderson SA. The inter-association task force for preventing sudden death in secondary school athletics programs: best-practices recommendations. J Athl Train. 2013;48(4):546-553.
13. Gardner JW, Kark JA, Karnei K, et al. Risk factors predicting exertional heat illness in male Marine Corps recruits. Med Sci Sports Exerc. 1996;28(8):939-944.
14. Gundstein AJ, Ramseyer C, Zhao F, et al. A retrospective analysis of American football hyperthermia deaths in the United States. Int J Biometerol. 2012;56(1):11-20.
15. Armstrong LE, Johnson EC, Casa DJ, et al. The American football uniform: uncompensable heat stress and hyperthermic exhaustion. J Athl Train. 2010;45(2):117-127.
16. Casa DJ, Csillan D; Inter-Association Task Force for Preseason Secondary School Athletics Participants, et al. Preseason heat-acclimatization guidelines for secondary school athletics. J Athl Train. 2009;44(3):332-333.
17. Godek SF, Godek JJ, Bartolozzi AR. Hydration status in college football players during consecutive days of twice-a-day preseason practices. Am J Sports Med. 2005;33(6):843-851.
18. Stover EA, Zachwieja J, Stofan J, Murray R, Horswill CA. Consistently high urine specific gravity in adolescent American football players and the impact of an acute drinking strategy. Int J Sports Med. 2006;27(4):330-335.
19. Bongers CC, Thijssen DH, Veltmeijer MTW, Hopman MT, Eijsvogels TM. Precooling and percooling (cooling during exercise) both improve performance in the heat: a meta-analytical review. Br J Sports Med. 2015;49(6):377-384.
20. Casa DJ, McDermott BP, Lee EC, Yeargin SW, Armstrong LE, Maresh CM. Cold water immersion: the gold standard for exertional heatstroke treatment. Exerc Sport Sci Rev. 2007;35(3):141-149.
21. DeMartini JK, Casa DJ, Stearns R, et al. Effectiveness of cold water immersion in the treatment of exertional heat stroke at the Falmouth Road Race. Med Sci Sports Exerc. 2015;47(2):240-245.
22. Casa DJ, Kenny GP, Taylor NA. Immersion treatment for exertional hyperthermia: cold or temperate water? Med Sci Sports Exerc. 2010;42(7):1246-1252.
23. Casa DJ, Armstrong LE, Kenny GP, O’Connor FG, Huggins RA. Exertional heat stroke: new concepts regarding cause and care. Curr Sports Med Rep. 2012;11(3):115-122.
24. Argaud L, Ferry T, Le QH, et al. Short- and long-term outcomes of heat stroke following the 2003 heat wave in Lyon, France. Arch Intern Med. 2007;167(20):2177-2183.
25. O’Connor FG, Casa DJ, Bergeron MF, et al. American College of Sports Medicine Roundtable on exertional heat stroke--return to duty/return to play: conference proceedings. Curr Sports Med Rep. 2010;9(5):314-321.
26. Kazman JB, Heled Y, Lisman PJ, Druyan A, Deuster PA, O’Connor FG. Exertional heat illness: the role of heat tolerance testing. Curr Sports Med Rep. 2013;12(2):101-105.
27. Moran DS, Heled Y, Still L, Laor A, Shapiro Y. Assessment of heat tolerance for post exertional heat stroke individuals. Med Sci Monit. 2004;10(6):CR252-CR257.
28. Herring SA, Kibler WB, Putukian M. Team Physician Consensus Statement: 2013 update. Med Sci Sports Exerc. 2013;45(8):1618-1622.
29. Heat stroke treatment. Korey Stringer Institute University of Connecticut Web site. http://ksi.uconn.edu/emergency-conditions/heat-illnesses/exertional-heat-stroke/heat-stroke-treatment/. Accessed June 14, 2016.
30. Headquarters, Department of the Army and the Air Force. Heat Stress Control and Heat Casualty Management. Technical Bulletin Medical 507. http://www.dir.ca.gov/oshsb/documents/Heat_illness_prevention_tbmed507.pdf. Published March 7, 2003. Accessed June 14, 2016.
1. Kucera KL, Klossner D, Colgate B, Cantu RC. Annual Survey of Football Injury Research: 1931-2014. National Center for Catastrophic Sport Injury Research Web site. https://nccsir.unc.edu/files/2013/10/Annual-Football-2014-Fatalities-Final.pdf. Accessed May 31, 2016.
2. Boden BP, Breit I, Beachler JA, Williams A, Mueller FO. Fatalities in high school and college football players. Am J Sports Med. 2013;41(5):1108-1116.
3. Bouchama A, Knochel JP. Heat stroke. N Engl J Med. 2002;346(25):1978-1988.
4. Racinais S, Alonso JM, Coutts AJ, et al. Consensus recommendations on training and competing in the heat. Scand J Med Sci Sports. 2015;25 Suppl 1:6-19.
5. Pryor RR, Casa DJ, Adams WM, et al. Maximizing athletic performance in the heat. Strength Cond J. 2013;35(6):24-33.
6. Adams WM, Casa DJ. Hydration for football athletes. Sports Sci Exchange. 2015;28(141):1-5.
7. American College of Sports Medicine, Armstrong LE, Casa DJ, et al. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc. 2007;39(3):556-572.
8. Yard EE, Gilchrist J, Haileyesus T, et al. Heat illness among high school athletes--United States, 2005-2009. J Safety Res. 2010;41(6):471-474.
9. Huffman EA, Yard EE, Fields SK, Collins CL, Comstock RD. Epidemiology of rare injuries and conditions among United States high school athletes during the 2005-2006 and 2006-2007 school years. J Athl Train. 2008;43(6):624-630.
10. Kerr ZY, Casa DJ, Marshall SW, Comstock RD. Epidemiology of exertional heat illness among U.S. high school athletes. Am J Prev Med. 2013;44(1):8-14.
11. Casa DJ, DeMartini JK, Bergeron MF, et al. National Athletic Trainers’ Association position statement: exertional heat illnesses. J Athl Train. 2015;50(9):986-1000.
12. Casa DJ, Almquist J, Anderson SA. The inter-association task force for preventing sudden death in secondary school athletics programs: best-practices recommendations. J Athl Train. 2013;48(4):546-553.
13. Gardner JW, Kark JA, Karnei K, et al. Risk factors predicting exertional heat illness in male Marine Corps recruits. Med Sci Sports Exerc. 1996;28(8):939-944.
14. Gundstein AJ, Ramseyer C, Zhao F, et al. A retrospective analysis of American football hyperthermia deaths in the United States. Int J Biometerol. 2012;56(1):11-20.
15. Armstrong LE, Johnson EC, Casa DJ, et al. The American football uniform: uncompensable heat stress and hyperthermic exhaustion. J Athl Train. 2010;45(2):117-127.
16. Casa DJ, Csillan D; Inter-Association Task Force for Preseason Secondary School Athletics Participants, et al. Preseason heat-acclimatization guidelines for secondary school athletics. J Athl Train. 2009;44(3):332-333.
17. Godek SF, Godek JJ, Bartolozzi AR. Hydration status in college football players during consecutive days of twice-a-day preseason practices. Am J Sports Med. 2005;33(6):843-851.
18. Stover EA, Zachwieja J, Stofan J, Murray R, Horswill CA. Consistently high urine specific gravity in adolescent American football players and the impact of an acute drinking strategy. Int J Sports Med. 2006;27(4):330-335.
19. Bongers CC, Thijssen DH, Veltmeijer MTW, Hopman MT, Eijsvogels TM. Precooling and percooling (cooling during exercise) both improve performance in the heat: a meta-analytical review. Br J Sports Med. 2015;49(6):377-384.
20. Casa DJ, McDermott BP, Lee EC, Yeargin SW, Armstrong LE, Maresh CM. Cold water immersion: the gold standard for exertional heatstroke treatment. Exerc Sport Sci Rev. 2007;35(3):141-149.
21. DeMartini JK, Casa DJ, Stearns R, et al. Effectiveness of cold water immersion in the treatment of exertional heat stroke at the Falmouth Road Race. Med Sci Sports Exerc. 2015;47(2):240-245.
22. Casa DJ, Kenny GP, Taylor NA. Immersion treatment for exertional hyperthermia: cold or temperate water? Med Sci Sports Exerc. 2010;42(7):1246-1252.
23. Casa DJ, Armstrong LE, Kenny GP, O’Connor FG, Huggins RA. Exertional heat stroke: new concepts regarding cause and care. Curr Sports Med Rep. 2012;11(3):115-122.
24. Argaud L, Ferry T, Le QH, et al. Short- and long-term outcomes of heat stroke following the 2003 heat wave in Lyon, France. Arch Intern Med. 2007;167(20):2177-2183.
25. O’Connor FG, Casa DJ, Bergeron MF, et al. American College of Sports Medicine Roundtable on exertional heat stroke--return to duty/return to play: conference proceedings. Curr Sports Med Rep. 2010;9(5):314-321.
26. Kazman JB, Heled Y, Lisman PJ, Druyan A, Deuster PA, O’Connor FG. Exertional heat illness: the role of heat tolerance testing. Curr Sports Med Rep. 2013;12(2):101-105.
27. Moran DS, Heled Y, Still L, Laor A, Shapiro Y. Assessment of heat tolerance for post exertional heat stroke individuals. Med Sci Monit. 2004;10(6):CR252-CR257.
28. Herring SA, Kibler WB, Putukian M. Team Physician Consensus Statement: 2013 update. Med Sci Sports Exerc. 2013;45(8):1618-1622.
29. Heat stroke treatment. Korey Stringer Institute University of Connecticut Web site. http://ksi.uconn.edu/emergency-conditions/heat-illnesses/exertional-heat-stroke/heat-stroke-treatment/. Accessed June 14, 2016.
30. Headquarters, Department of the Army and the Air Force. Heat Stress Control and Heat Casualty Management. Technical Bulletin Medical 507. http://www.dir.ca.gov/oshsb/documents/Heat_illness_prevention_tbmed507.pdf. Published March 7, 2003. Accessed June 14, 2016.
2016 Update on female sexual dysfunction
The age-adjusted prevalence of any sexual problem is 43% among US women. A full 22% of these women experience sexually related personal distress.1 With publication of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition2 has come a shift in classification and, at times, management approach for reported female sexual dysfunction. When women report to their clinicians decreased sexual desire or arousal or pain at penetration, the management is no longer guided by a linear model of sexual response (excitation, plateau, orgasm, and resolution) but rather by a more nuanced and complex biopsychosocial approach. In this model, diagnosis and management strategies to address bothersome sexual concerns consider the whole woman in the context of her physical and psychosocial health. The patient’s age, medical history, and relationship status are among the factors that could affect management of the problem. In an effort to explore this management approach, I used this Update on Female Sexual Dysfunction as an opportunity to convene a roundtable of several experts, representing varying backgrounds and practice vantage points, to discuss 5 cases of sexual problems that you as a busy clinician may encounter in your practice.

Genital atrophy in a sexually inactive 61-year-old woman
Barbara S. Levy, MD: Two years after her husband's death, which followed several years of illness, your 61-year-old patient mentions at her well woman visit that she anticipates becoming sexually active again. She has not used systemic or vaginal hormone therapy. During pelvic examination, atrophic external genital changes are present, and use of an ultrathin (thinner than a Pederson) speculum reveals vaginal epithelial atrophic changes. A single-digit bimanual exam can be performed with moderate patient discomfort; the patient cannot tolerate a 2-digit bimanual exam. She expresses concern about being able to engage in penile/vaginal sexual intercourse.
Dr. Kaunitz, what is important for you to ask this patient, and what concerns you most on her physical exam?
Andrew M. Kaunitz, MD: First, it is important to recognize the patient's expectations and desires. As the case suggests, but further questioning could clarify, she would like to be able to comfortably engage in sexual intercourse with a new partner, but penetration may be difficult (and definitely painful for her) unless treatment is pursued. This combination of mucosal and vestibular atrophic changes (genitourinary syndrome of menopause [GSM], or vulvovaginal atrophy [VVA]) plus the absence of penetration for many years can be a double whammy situation for menopausal women. In this case it has led to extensive contracture of the introitus, and if it is not addressed will cause sexual dysfunction.
Dr. Levy: In addition we need to clarify whether or not a history of breast cancer or some other thing may impact the care we provide. How would you approach talking with this patient in order to manage her care?
Dr. Kaunitz: One step is to see how motivated she is to address this, as it is not something that, as gynecologists, we can snap our fingers and the situation will be resolved. If the patient is motivated to treat the atrophic changes with medical treatment, in the form of low-dose vaginal estrogen, and dilation, either on her own if she's highly motivated to do so, or in my practice more commonly with the support of a women's physical therapist, over time she should be able to comfortably engage in sexual intercourse with penetration. If this is what she wants, we can help steer her in the right direction.
Sheryl Kingsberg, PhD: You know that this woman is motivated by virtue of her initiating the topic herself. Patients are often embarrassed talking about sexual issues, or they are not sure that their gynecologist is comfortable with it. After all, they think, if this is the right place to discuss sexual problems, why didn't he or she ask me? Clinicians must be aware that it is their responsibility to ask about sexual function and not leave it for the patient to open the door.
Dr. Kaunitz: Great point.
Cheryl B. Iglesia, MD: Gratefully, a lot of the atrophic changes this patient demonstrates are reversible. However, other autoimmune diseases (eg, lichen planus, which can affect the vaginal epithelium, or lichen sclerosus, which can affect the clitoris, labia, and vulva) can also cause constriction, and in severe cases, complete obliteration of the vagina and introitus. Women may not be sexually active, and for each annual exam their clinician uses a smaller and smaller speculum--to the point that they cannot even access the cervix anymore--and the vagina can close off. Clinicians may not realize that you need something other than estrogen; with lichen planus you need steroid suppository treatment, and with lichen sclerosus you need topical steroid treatment. So these autoimmune conditions should also be in the differential and, with appropriate treatment, the sexual effects can be reversible.
Michael Krychman, MD: I agree. The vulva can be a great mimicker and, according to the history and physical exam, at some point a vulvoscopy, and even potential biopsies, may be warranted as clinically indicated.
The concept of a comprehensive approach, as Dr. Kingsberg had previously mentioned, involves not only sexual medicine but also evaluating the patient's biopsychosocial variables that may impact her condition. We also need to set realistic expectations. Some women may benefit from off-label use of medications besides estrogen, including topical testosterone. Informed consent is very important with these treatments. I also have had much clinical success with intravaginal diazepam/lorazepam for pelvic floor hypertonus.
In addition, certainly I agree that pelvic floor physical therapy (PT) is a vital treatment component for this patient and, not to diminish its importance, but many women cannot afford, nor do they have the time or opportunity, to go to pelvic floor PT. As clinicians, we can develop and implement effective programs, even in the office, to educate the patient to help herself as well.
Dr. Kaunitz: Absolutely. Also, if, in a clinical setting consistent with atrophic changes, an ObGyn physician is comfortable that vulvovaginal changes noted on exam represent GSM/atrophic changes, I do not feel vulvoscopy is warranted.
Dr. Levy: In conclusion, we need to be aware that pelvic floor PT may not be available everywhere and that a woman's own digits and her partner can also be incorporated into this treatment.
Something that we have all talked about in other venues, but have not looked at in the larger sphere here, is whether there is value to seeing women annually and performing pelvic exams. As Dr. Kingsberg mentioned, this is a highly motivated patient. We have many patients out there who are silent sufferers. The physical exam is an opportunity for us to recognize and address this problem.
Dyspareunia and low sexual desire in a breast cancer survivor
Dr. Levy: In this case, a 36-year-old woman with BRCA1−positive breast cancer has vaginal dryness, painful intercourse, and lowered sexual interest since her treatment, which included chemotherapy after bilateral mastectomies. She has a bilateral salpingo-oophorectomy(BSO) scheduled for primary prevention of her ovarian cancer risk.
Dr. Kingsberg, what is important for you to know to help guide case management?
Dr. Kingsberg: This woman is actually presenting with 2 sexual problems: dyspareunia, which is probably secondary to VVA or GSM, and low sexual desire. Key questions are: 1) When was symptom onset--acquired after treatment or lifelong? 2) Did she develop the dyspareunia and as a result of having pain during sex lost desire to have sex? Or, did she lose desire and then, without it, had no arousal and therefore pain with penetration developed? It also could be that she has 2 distinct problems, VVA and hypoactive sexual desire disorder (HSDD), in which case you need to think about treating both. Finally, we do not actually know if she is having penetrative intercourse or even if she has a partner.
A vulvovaginal exam would give clues as to whether she has VVA, and hormone levels would indicate if she now has chemo-induced menopause. If she is not in menopause now, she certainly is going to be with her BSO. The hormonal changes due to menopause actually can be primarily responsible for both the dyspareunia and HSDD. Management of both symptoms really needs to be based on shared decision making with the patient--with which treatment for which conditions coming first, based on what is causing her the most distress.
I would encourage this woman to treat her VVA since GSM does have long-term physiologic consequences if untreated. The American College of Obstetricians and Gynecologists (ACOG) recommends nonhormonal treatments as first-line treatments, with vaginal estrogen considered if these therapies fail.3 If lubricants and moisturizers and other nonhormonal options are not sufficient, you could consider local estrogen, even though she is a breast cancer survivor, as well as ospemiphene.
If she is distressed by her loss of sexual desire, you can choose to treat her for HSDD. Flibanserin is the first FDA-approved treatment for HSDD. It is only approved in premenopausal women, so it would be considered off-label use if she is postmenopausal (even though she is quite young). You also could consider exogenous testosterone off-label, after consulting with her oncologist.
In addition to the obvious physiologic etiology of her pain and her low desire, the biopsychosocial aspects to consider are: 1) changes to her body image, as she has had bilateral mastectomies, 2) her anxiety about the cancer diagnosis, and 3) concerns about her relationship if she has one--her partner's reactions to her illness and the quality of the relationship outside the bedroom.
Dr. Iglesia: I am seeing here in our nation's capital a lot of advertisements for laser therapy for GSM. I caution women about this because providers are charging a lot of money for this therapy when we do not have long-term safety and effectiveness data for it.
Our group is currently conducting a randomized controlled trial, looking at vaginal estrogen cream versus laser therapy for GSM here at Medstar Health--one of the first in the country as part of a multisite trial. But the North American Menopause Society (NAMS) has come out with a pretty strong statement,4 as has ACOG,5 on this therapy, and I caution people about overzealously offering a very costly procedure targeted to a very vulnerable population, especially to women with personal histories of estrogen-sensitive cancers.
Dr. Krychman: I agree. Very often cancer patients are preyed upon by those offering emerging unproven technologies or medications. We have to work as a coordinated comprehensive team, whether it's a sexual medicine expert, psychologist, urogynecologist, gynecologist, or oncologist, and incorporate the patient's needs and expectations and risk tolerance coupled with treatment efficacy and safety.
Dr. Levy: This was a complex case. The biopsychosocial model is critical here. It's important that we are not siloes in our medical management approach and that we try to help this patient embrace the complexity of her situation. It's not only that she has cancer at age 36; there is a possible guilt factor if she has children and passed that gene on.
Another point that we began to talk about is the fact that in this country we tend to be early adopters of new technology. In our discussion with patients, we should focus on what we know and the risk of the unknowns related to some of the treatment options. But let's discuss lasers a little more later on.
Diminished arousal and orgasmic intensity in a patient taking SSRIs
Dr. Levy: In this next case, a 44-year-old woman in a 15-year marriage notices a change in her orgasmic intensity and latency. She has a supportive husband, and they are attentive to each other's sexual needs. However, she notices a change in her arousal and orgasmic intensity, which has diminished over the last year. She reports that the time to orgasm or latency has increased and both she and her partner are frustrated and getting concerned. She has a history of depression that has been managed by selective serotonin reuptake inhibitors for the past 5 years and has no depressive symptoms currently.
Dr. Krychman, what are you considering before beginning to talk with this patient?
Dr. Krychman: My approach really is a comprehensive one, looking not only at the underlying medical issues but also at the psychological and dynamic relationship facets. We of course also want to look at medications: Has she changed her dose or the timing of when she takes it? Is this a new onset? Finally, we want to know why this is coming to the forefront now. Is it because it is getting worse, or is it because there is some significant issue that is going on in the relationship?
Regarding the physical exam, it is important to rule out underlying genital pelvic pathology. Young women can get changes in the integrity of the pelvic floor, in what I would call the orgasmic matrix--the clitoral tissue, the body, the crura (or arms of the clitoris)--we want to examine and be reassured that her genital anatomy is normal and that there is no underlying pathology that could signal an underlying abnormal hormonal profile. Young women certainly can get lowered estrogen effects at the genital/pelvic tissues (including the labia and vulva), and intravaginally as well. Sometimes women will have pelvic floor hypertonus, as we see with other urinary issues. A thorough pelvic exam is quite vital.
Let's not forget the body that is attached to the genitals; we want to rule out chronic medical disease that may impact her: hypertension, diabetes, or hypercholesterolemia. Untreated, these conditions may directly impact the arousal physiologic mechanisms.
Dr. Levy: In doing this patient's physical exam I would be looking for significant weight gain, and even asking about her partner's weight. Body image can be a huge issue. If she has a history of depression, if she is suffering from a body image problem, she can be feeling unattractive. In my experience this can be a common thing to affect women in their mid-40s.
How would you manage this case?
Dr. Krychman: It is important to divide it up in terms of a conservative to aggressive approach. We want to find out about the relationship. For instance, is the sexual dynamic scripted (ie, boring and predictable)? Is she distracted and frustrated or is she getting enough of the type of stimulation that she likes and enjoys? There certainly are a lot of new devices that are available, whether a self-stimulator or vibrator, the Fiera, or other stimulating devices, that may be important to incorporate into the sexual repertoire. If there is underlying pathology, we want to evaluate and treat that. She may need to be primed, so to speak, with systemic hormones. And does she have issues related to other effects of hormonal deprivation, even local effects? Does she have clitoral atrophy?
There are neutraceuticals that are currently available, whether topical arousal gels or ointments, and we as clinicians need to be critical and evaluate their benefit/risk and look at the data concerning these products. In addition, women who have changes in arousal and in orgasmic intensity and latency may be very frustrated. They describe it as climbing up to a peak but never getting over the top, and this frustration may lead to participant spectatoring, so incorporating a certified sex therapist or counselor is sometimes very critical.
Finally, there are a lot of snake oils, charmers, and charlatan unproven procedures--injecting fillers or other substances into the clitoris are a few examples. I would be a critical clinician, examine the evidence, look at the benefit/risk before advocating an intervention that does not have good clinical data to support its use--a comprehensive approach of sexual medicine as well as sexual psychology.
Dr. Kingsberg: Additionally, we know she is in a long-term relationship--15 years; we want to acknowledge the partner. We talked about the partner's weight, but what about his erectile function? Does he have changes in sexual function that are affecting her, and she is the one carrying the "symptom"?
Looking at each piece separately helps a clinician from getting overwhelmed by the patient who comes in reporting distress with orgasmic dysfunction. We have no pharmacologic FDA-approved treatments, so it can feel off-putting for a clinician to try to fix the reported issue. Looking at each component to help her figure out the underlying cause can be helpful.
Dr. Iglesia: With aging, there can be changes in blood flow, not to mention the hormonal and even peripheral nerve changes, that require more stimulation in order to achieve the desired response. I echo concern about expensive procedures being offered with no evidence, such as the "O" or "G" shot, that can cost up to thousands of dollars.
The other procedure that gives me a lot of angst is clitoral unhooding. The 3 parts of the clitoris are sensitive in terms of innervation and blood flow, and cutting around that delicate tissue goes against the surgical principles required for preserving nerves and blood flow.
New onset pain post−prolapse surgery with TOT sling placement
Dr. Levy: For this case, let's consider a 42-year-old woman (P3) who is 6 months post− vaginal hysterectomy. The surgery included ovarian preservation combined with anterior and posterior repair for prolapse as well as apical uterosacral ligament suspension for stage 2 uterovaginal prolapse. A transobturator sling was used.
Extensive preop evaluation was performed, with confirmed symptomatic prolapse. She had no stress incontinence symptoms but did have confirmed occult stress incontinence.
Surgery was uneventful. She resumed intercourse at 8 weeks, but she now has pain with both initial entry and deep penetration. Lubricants and changes in position have not helped. She is in a stable relationship with her husband of 17 years, and she is worried that the sling mesh might be the culprit. On exam, she has no atrophy, pH is 4.5, vaginal length is 8 cm, and there is no prolapse. There is no mesh exposure noted, although she reports slight tenderness with palpation of the right sling arm beneath the right pubic bone.
Dr. Iglesia, what are the patient history questions important to ask here?
Dr. Iglesia: This is not an uncommon scenario--elective surgical correction of occult or latent stress incontinence after surgical correction for pelvic organ prolapse. Now this patient here has no more prolapse complaints; however, she has a new symptom. There are many different causes of dyspareunia; we cannot just assume it is the sling mesh (although with all the legal representation advertisements for those who have had mesh placed, it can certainly be at the top of the patient's mind, causing anxiety and fear).
Multiple trials have looked at prophylactic surgery for incontinence at the time of prolapse repairs. This woman happened to be one of those patients who did not have incontinence symptoms, and they put a sling in. A recent large trial examined women with vaginal prolapse who underwent hysterectomy and suspension.6 (They compared 2 different suspensions.) What is interesting is that 25% of women with prolapse do have baseline pain. However, at 24 months, de novo pain can occur in 10% of women--just from the apical suspension. So, here, it could be the prolapse suspension. Or, in terms of the transobturator sling, long-term data do tell us that the de novo dyspareunia rate ranges on the magnitude of 1% to 9%.7 What is important here is figuring out the cause of the dyspareunia.
Dr. Levy: One of the important points you raised already was that 25% of these women have preoperative pain. So figuring out what her functioning was before surgery and incorporating that into our assessment postop would be pretty important I would think.
Dr. Iglesia: Yes, you need to understand what her typical encounter was before the surgery and how things have changed now that the prolapse is not in the way. Changes obviously can occur with scar tissue, which over time will improve. If she is perimenopausal and starts to get epithelial changes, we can fix that. The question then becomes, "Is the pain emanating from the mesh?"
When examining this patient, it is not uncommon for me to be able to feel "banjo" strings if the mesh is too tight or close to the surface. It is not exposed but it's palpable, and the patient may feel a ridge during penetration. You can ask the patient if pain occurs with different penetration positions. In addition, explore associated neurologic symptoms (numbness or muscle pain in the thigh).
Dr. Kingsberg: There were 2 different sources of pain--on initial entry and at deep penetration. You want to make sure you address both. Importantly, did one precede the other? For instance, if women have pain with penetration they can then end up with an arousal disorder (the length of the vagina cannot increase as much as it might otherwise) and dystonia secondary to the pain with penetration. The timing of the pain--did it all happen at the same time, or did she start out with pain at one point and did it move to something else--is another critical piece of the history.
Dr. Iglesia: It does take a detailed history and physical exam to identify myofascial trigger points. In this case there seems to be pinpoint tenderness directly on the part of the mesh that is not exposed on the right. There are people who feel you should remove the whole mesh, including the arms. Others feel, okay, we can work on these trigger points, with injections, physical therapy, extra lubrication, and neuromodulatory medications. Only then would they think about potentially excising the sling or a portion thereof.
Dr. Krychman: Keep in mind that, even if you do remove the sling, her pain may not subside, if it is secondary to an underlying issue. Because of media sensationalism, she could be focusing on the sling. It is important to set realistic expectations. I often see vulvar pathology or even provoked vestibulodynia that can present with a deep dyspareunia. The concept of collision dyspareunia or introital discomfort or pain on insertion has far reaching implications. We need to look at the patient in totality, ruling out underlying issues related to the bladder, even the colon.
Patient inquires about the benefits of laser treatment for vaginal health
Dr. Levy: Let's move to this last case: A 47-year-old patient who reports lack of sexual satisfaction and attributes it to a loose vagina says, "I've heard about surgery and laser treatment and radiofrequency devices for vaginal health. What are the benefits and risks of these procedures, and will they correct the issues that I'm experiencing?"
How do you approach this patient?
Dr. Krychman: I want to know why this is of paramount importance to her. Is this her actual complaint or is it society's unrealistic expectation of sexual pleasure and performance placed upon her? Or is this a relationship issue surfacing, compounded by physiologic changes? With good communication techniques, like "ask, tell, ask" or effective use of silence (not interrupting), patients will lead us to the reason and the rationale.
Dr. Levy: I have seen a lot of advertising to women, now showing pictures of genitalia, perhaps creating an expectation that we should look infantile in some way. We are creating a sense of beauty and acceptablevisualization of the vagina and vulva that are completely unrealistic. It's fascinating on one hand but it is also disturbing in that some of the direct-to-consumer marketing going on is creating a sense of unease in women who are otherwise perfectly satisfied. Now they take a look at their sagging skin, maybe after having 2 or 3 children, and although they may not look the same they function not so badly perhaps. I think we are creating a distress and an illness model that is interesting to discuss.
Dr. Iglesia, you are in the midst of a randomized trial, giving an informed consent to participants about the expectations for this potential intervention. How do you explain to women what these laser and radiofrequency devices are expected to do and why they might or might not work?
Dr. Iglesia: We are doing a randomized trial for menopausal women who have GSM. We are comparing estrogen cream with fractional CO2 laser therapy. I also am involved in another randomized trial for lichen sclerosus. I am not involved in the cosmetic use of a laser for people who feel their vaginas are just "loose." Like you, Dr. Levy, I am very concerned about the images that women are seeing of the idealized vulva and vagina, and about the rise of cosmetic gynecology, much of which is being performed by plastic surgeons or dermatologists.
A recent article looked at the number of women who are doing pubic hair grooming; the prevalance is about 80% here in America.8 So people have a clear view of what is down there, and then they compare it to what they see in pornographic images on the Internet and want to look like a Barbie doll. That is disturbing because women, particularly young women, do not realize what happens with GSM changes to the vulva and vagina. On the other hand, these laser machines are very expensive, and some doctors are charging thousands of dollars and promising cosmetic and functional results for which we lack long-term, comparative data.
The laser that we are studying is one by Cynosure called Mona Lisa, which works with fractional CO2 and has very low depth of penetration. The concept is that, with microdot therapy (on the order of micrometers), pinpoint destruction will foster regeneration of new collagen and blood flow to the vagina and vulva. We are still in the midst of analyzing this.
Dr. Krychman: I caution people not to lump devices together. There is a significant difference between laser and radiofrequency--especially in the depth of tissue penetration, and level of evidence as well. There are companies performing randomized clinical trials, with well designed sham controls, and have demonstrated clinical efficacy. We need to be cautious of a procedure that is saying it's the best thing since sliced bread, curing interstitial cystitis, dyspareunia, and lichen sclerosus and improving orgasm, lubrication, and arousal. These far reaching, off-label claims are concerning and misleading.
Dr. Levy: I think the important things are 1) shared decision making with the patient and 2) disclosure of what we do not know, which are the long-term results and outcomes and possible downstream negative effects of some of these treatments, since the data we have are so short term.
Dr. Kingsberg: Basics are important. You talked about the pressure for cosmetic appearance, but is that really what is going on for this particular patient? Is she describing a sexual dysfunction when she talks about lack of satisfaction? You need to operationally define that term. Does she have problems with arousal or orgasm or desire and those are what underlie her lack of satisfaction? Is the key to management helping her come to terms with body image issues or to treat a sexual dysfunction? If it truly is a sexual dysfunction, then you can have the shared decision making on preferred treatment approach.
Dr. Levy: This has been an enlightening discussion. Thank all of you for your expertise and clinical acumen.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112(5):970−978.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM), Fifth Edition. American Psychiatric Association Publishing: Arlington, VA; 2013.
- American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, Farrell R. ACOG Committee Opinion No. 659: The Use of Vaginal Estrogen in Women With a History of Estrogen-Dependent Breast Cancer. Obstet Gynecol. 2016;127(3):e93−e96.
- Krychman ML, Shifren JL, Liu JH, Kingsberg SL, Utian WH. The North American Menopause Society (NAMS). NAMS Menopause e-Consult: Laser treatment safe for vulvovaginal atrophy? 2015;11(3). http://www.medscape.com/viewarticle/846960. Accessed August 17, 2016.
- The American College of Obstetricians and Gynecologists and The American Congress of Obstetricians and Gynecologists. Fractional laser treatment of vulvovaginal atrophy and U.S. Food and Drug Administration clearance: Position Statement. http://www.acog.org/Resources-And-Publications/Position-Statements/Fractional-Laser-Treatment-of-Vulvovaginal-Atrophy-and-US-Food-and-Drug-Administration-Clearance. Published May 2016. Accessed August 17, 2016.
- Lukacz ES, Warren LK, Richter HE, et al. Quality of life and sexual function 2 years after vaginal surgery for prolapse. Obstet Gynecol. 2016;127(6):1071−1079.
- Brubaker L, Chiang S, Zyczynski H, et al. The impact of stress incontinence surgery on female sexual function. Am J Obstet Gynecol. 2009;200(5):562.e1.
- Rowen TS, Gaither TW, Awad MA, Osterberg EC, Shindel AW, Breyer BN. Pubic hair grooming prevalence and motivation among women in the United States [published online ahead of print June 29, 2016]. JAMA Dermatol. doi:10.1001/jamader matol.2016.2154.
The age-adjusted prevalence of any sexual problem is 43% among US women. A full 22% of these women experience sexually related personal distress.1 With publication of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition2 has come a shift in classification and, at times, management approach for reported female sexual dysfunction. When women report to their clinicians decreased sexual desire or arousal or pain at penetration, the management is no longer guided by a linear model of sexual response (excitation, plateau, orgasm, and resolution) but rather by a more nuanced and complex biopsychosocial approach. In this model, diagnosis and management strategies to address bothersome sexual concerns consider the whole woman in the context of her physical and psychosocial health. The patient’s age, medical history, and relationship status are among the factors that could affect management of the problem. In an effort to explore this management approach, I used this Update on Female Sexual Dysfunction as an opportunity to convene a roundtable of several experts, representing varying backgrounds and practice vantage points, to discuss 5 cases of sexual problems that you as a busy clinician may encounter in your practice.

Genital atrophy in a sexually inactive 61-year-old woman
Barbara S. Levy, MD: Two years after her husband's death, which followed several years of illness, your 61-year-old patient mentions at her well woman visit that she anticipates becoming sexually active again. She has not used systemic or vaginal hormone therapy. During pelvic examination, atrophic external genital changes are present, and use of an ultrathin (thinner than a Pederson) speculum reveals vaginal epithelial atrophic changes. A single-digit bimanual exam can be performed with moderate patient discomfort; the patient cannot tolerate a 2-digit bimanual exam. She expresses concern about being able to engage in penile/vaginal sexual intercourse.
Dr. Kaunitz, what is important for you to ask this patient, and what concerns you most on her physical exam?
Andrew M. Kaunitz, MD: First, it is important to recognize the patient's expectations and desires. As the case suggests, but further questioning could clarify, she would like to be able to comfortably engage in sexual intercourse with a new partner, but penetration may be difficult (and definitely painful for her) unless treatment is pursued. This combination of mucosal and vestibular atrophic changes (genitourinary syndrome of menopause [GSM], or vulvovaginal atrophy [VVA]) plus the absence of penetration for many years can be a double whammy situation for menopausal women. In this case it has led to extensive contracture of the introitus, and if it is not addressed will cause sexual dysfunction.
Dr. Levy: In addition we need to clarify whether or not a history of breast cancer or some other thing may impact the care we provide. How would you approach talking with this patient in order to manage her care?
Dr. Kaunitz: One step is to see how motivated she is to address this, as it is not something that, as gynecologists, we can snap our fingers and the situation will be resolved. If the patient is motivated to treat the atrophic changes with medical treatment, in the form of low-dose vaginal estrogen, and dilation, either on her own if she's highly motivated to do so, or in my practice more commonly with the support of a women's physical therapist, over time she should be able to comfortably engage in sexual intercourse with penetration. If this is what she wants, we can help steer her in the right direction.
Sheryl Kingsberg, PhD: You know that this woman is motivated by virtue of her initiating the topic herself. Patients are often embarrassed talking about sexual issues, or they are not sure that their gynecologist is comfortable with it. After all, they think, if this is the right place to discuss sexual problems, why didn't he or she ask me? Clinicians must be aware that it is their responsibility to ask about sexual function and not leave it for the patient to open the door.
Dr. Kaunitz: Great point.
Cheryl B. Iglesia, MD: Gratefully, a lot of the atrophic changes this patient demonstrates are reversible. However, other autoimmune diseases (eg, lichen planus, which can affect the vaginal epithelium, or lichen sclerosus, which can affect the clitoris, labia, and vulva) can also cause constriction, and in severe cases, complete obliteration of the vagina and introitus. Women may not be sexually active, and for each annual exam their clinician uses a smaller and smaller speculum--to the point that they cannot even access the cervix anymore--and the vagina can close off. Clinicians may not realize that you need something other than estrogen; with lichen planus you need steroid suppository treatment, and with lichen sclerosus you need topical steroid treatment. So these autoimmune conditions should also be in the differential and, with appropriate treatment, the sexual effects can be reversible.
Michael Krychman, MD: I agree. The vulva can be a great mimicker and, according to the history and physical exam, at some point a vulvoscopy, and even potential biopsies, may be warranted as clinically indicated.
The concept of a comprehensive approach, as Dr. Kingsberg had previously mentioned, involves not only sexual medicine but also evaluating the patient's biopsychosocial variables that may impact her condition. We also need to set realistic expectations. Some women may benefit from off-label use of medications besides estrogen, including topical testosterone. Informed consent is very important with these treatments. I also have had much clinical success with intravaginal diazepam/lorazepam for pelvic floor hypertonus.
In addition, certainly I agree that pelvic floor physical therapy (PT) is a vital treatment component for this patient and, not to diminish its importance, but many women cannot afford, nor do they have the time or opportunity, to go to pelvic floor PT. As clinicians, we can develop and implement effective programs, even in the office, to educate the patient to help herself as well.
Dr. Kaunitz: Absolutely. Also, if, in a clinical setting consistent with atrophic changes, an ObGyn physician is comfortable that vulvovaginal changes noted on exam represent GSM/atrophic changes, I do not feel vulvoscopy is warranted.
Dr. Levy: In conclusion, we need to be aware that pelvic floor PT may not be available everywhere and that a woman's own digits and her partner can also be incorporated into this treatment.
Something that we have all talked about in other venues, but have not looked at in the larger sphere here, is whether there is value to seeing women annually and performing pelvic exams. As Dr. Kingsberg mentioned, this is a highly motivated patient. We have many patients out there who are silent sufferers. The physical exam is an opportunity for us to recognize and address this problem.

Dyspareunia and low sexual desire in a breast cancer survivor
Dr. Levy: In this case, a 36-year-old woman with BRCA1−positive breast cancer has vaginal dryness, painful intercourse, and lowered sexual interest since her treatment, which included chemotherapy after bilateral mastectomies. She has a bilateral salpingo-oophorectomy(BSO) scheduled for primary prevention of her ovarian cancer risk.
Dr. Kingsberg, what is important for you to know to help guide case management?
Dr. Kingsberg: This woman is actually presenting with 2 sexual problems: dyspareunia, which is probably secondary to VVA or GSM, and low sexual desire. Key questions are: 1) When was symptom onset--acquired after treatment or lifelong? 2) Did she develop the dyspareunia and as a result of having pain during sex lost desire to have sex? Or, did she lose desire and then, without it, had no arousal and therefore pain with penetration developed? It also could be that she has 2 distinct problems, VVA and hypoactive sexual desire disorder (HSDD), in which case you need to think about treating both. Finally, we do not actually know if she is having penetrative intercourse or even if she has a partner.
A vulvovaginal exam would give clues as to whether she has VVA, and hormone levels would indicate if she now has chemo-induced menopause. If she is not in menopause now, she certainly is going to be with her BSO. The hormonal changes due to menopause actually can be primarily responsible for both the dyspareunia and HSDD. Management of both symptoms really needs to be based on shared decision making with the patient--with which treatment for which conditions coming first, based on what is causing her the most distress.
I would encourage this woman to treat her VVA since GSM does have long-term physiologic consequences if untreated. The American College of Obstetricians and Gynecologists (ACOG) recommends nonhormonal treatments as first-line treatments, with vaginal estrogen considered if these therapies fail.3 If lubricants and moisturizers and other nonhormonal options are not sufficient, you could consider local estrogen, even though she is a breast cancer survivor, as well as ospemiphene.
If she is distressed by her loss of sexual desire, you can choose to treat her for HSDD. Flibanserin is the first FDA-approved treatment for HSDD. It is only approved in premenopausal women, so it would be considered off-label use if she is postmenopausal (even though she is quite young). You also could consider exogenous testosterone off-label, after consulting with her oncologist.
In addition to the obvious physiologic etiology of her pain and her low desire, the biopsychosocial aspects to consider are: 1) changes to her body image, as she has had bilateral mastectomies, 2) her anxiety about the cancer diagnosis, and 3) concerns about her relationship if she has one--her partner's reactions to her illness and the quality of the relationship outside the bedroom.
Dr. Iglesia: I am seeing here in our nation's capital a lot of advertisements for laser therapy for GSM. I caution women about this because providers are charging a lot of money for this therapy when we do not have long-term safety and effectiveness data for it.
Our group is currently conducting a randomized controlled trial, looking at vaginal estrogen cream versus laser therapy for GSM here at Medstar Health--one of the first in the country as part of a multisite trial. But the North American Menopause Society (NAMS) has come out with a pretty strong statement,4 as has ACOG,5 on this therapy, and I caution people about overzealously offering a very costly procedure targeted to a very vulnerable population, especially to women with personal histories of estrogen-sensitive cancers.
Dr. Krychman: I agree. Very often cancer patients are preyed upon by those offering emerging unproven technologies or medications. We have to work as a coordinated comprehensive team, whether it's a sexual medicine expert, psychologist, urogynecologist, gynecologist, or oncologist, and incorporate the patient's needs and expectations and risk tolerance coupled with treatment efficacy and safety.
Dr. Levy: This was a complex case. The biopsychosocial model is critical here. It's important that we are not siloes in our medical management approach and that we try to help this patient embrace the complexity of her situation. It's not only that she has cancer at age 36; there is a possible guilt factor if she has children and passed that gene on.
Another point that we began to talk about is the fact that in this country we tend to be early adopters of new technology. In our discussion with patients, we should focus on what we know and the risk of the unknowns related to some of the treatment options. But let's discuss lasers a little more later on.
Diminished arousal and orgasmic intensity in a patient taking SSRIs
Dr. Levy: In this next case, a 44-year-old woman in a 15-year marriage notices a change in her orgasmic intensity and latency. She has a supportive husband, and they are attentive to each other's sexual needs. However, she notices a change in her arousal and orgasmic intensity, which has diminished over the last year. She reports that the time to orgasm or latency has increased and both she and her partner are frustrated and getting concerned. She has a history of depression that has been managed by selective serotonin reuptake inhibitors for the past 5 years and has no depressive symptoms currently.
Dr. Krychman, what are you considering before beginning to talk with this patient?
Dr. Krychman: My approach really is a comprehensive one, looking not only at the underlying medical issues but also at the psychological and dynamic relationship facets. We of course also want to look at medications: Has she changed her dose or the timing of when she takes it? Is this a new onset? Finally, we want to know why this is coming to the forefront now. Is it because it is getting worse, or is it because there is some significant issue that is going on in the relationship?
Regarding the physical exam, it is important to rule out underlying genital pelvic pathology. Young women can get changes in the integrity of the pelvic floor, in what I would call the orgasmic matrix--the clitoral tissue, the body, the crura (or arms of the clitoris)--we want to examine and be reassured that her genital anatomy is normal and that there is no underlying pathology that could signal an underlying abnormal hormonal profile. Young women certainly can get lowered estrogen effects at the genital/pelvic tissues (including the labia and vulva), and intravaginally as well. Sometimes women will have pelvic floor hypertonus, as we see with other urinary issues. A thorough pelvic exam is quite vital.
Let's not forget the body that is attached to the genitals; we want to rule out chronic medical disease that may impact her: hypertension, diabetes, or hypercholesterolemia. Untreated, these conditions may directly impact the arousal physiologic mechanisms.
Dr. Levy: In doing this patient's physical exam I would be looking for significant weight gain, and even asking about her partner's weight. Body image can be a huge issue. If she has a history of depression, if she is suffering from a body image problem, she can be feeling unattractive. In my experience this can be a common thing to affect women in their mid-40s.
How would you manage this case?
Dr. Krychman: It is important to divide it up in terms of a conservative to aggressive approach. We want to find out about the relationship. For instance, is the sexual dynamic scripted (ie, boring and predictable)? Is she distracted and frustrated or is she getting enough of the type of stimulation that she likes and enjoys? There certainly are a lot of new devices that are available, whether a self-stimulator or vibrator, the Fiera, or other stimulating devices, that may be important to incorporate into the sexual repertoire. If there is underlying pathology, we want to evaluate and treat that. She may need to be primed, so to speak, with systemic hormones. And does she have issues related to other effects of hormonal deprivation, even local effects? Does she have clitoral atrophy?
There are neutraceuticals that are currently available, whether topical arousal gels or ointments, and we as clinicians need to be critical and evaluate their benefit/risk and look at the data concerning these products. In addition, women who have changes in arousal and in orgasmic intensity and latency may be very frustrated. They describe it as climbing up to a peak but never getting over the top, and this frustration may lead to participant spectatoring, so incorporating a certified sex therapist or counselor is sometimes very critical.
Finally, there are a lot of snake oils, charmers, and charlatan unproven procedures--injecting fillers or other substances into the clitoris are a few examples. I would be a critical clinician, examine the evidence, look at the benefit/risk before advocating an intervention that does not have good clinical data to support its use--a comprehensive approach of sexual medicine as well as sexual psychology.
Dr. Kingsberg: Additionally, we know she is in a long-term relationship--15 years; we want to acknowledge the partner. We talked about the partner's weight, but what about his erectile function? Does he have changes in sexual function that are affecting her, and she is the one carrying the "symptom"?
Looking at each piece separately helps a clinician from getting overwhelmed by the patient who comes in reporting distress with orgasmic dysfunction. We have no pharmacologic FDA-approved treatments, so it can feel off-putting for a clinician to try to fix the reported issue. Looking at each component to help her figure out the underlying cause can be helpful.
Dr. Iglesia: With aging, there can be changes in blood flow, not to mention the hormonal and even peripheral nerve changes, that require more stimulation in order to achieve the desired response. I echo concern about expensive procedures being offered with no evidence, such as the "O" or "G" shot, that can cost up to thousands of dollars.
The other procedure that gives me a lot of angst is clitoral unhooding. The 3 parts of the clitoris are sensitive in terms of innervation and blood flow, and cutting around that delicate tissue goes against the surgical principles required for preserving nerves and blood flow.
New onset pain post−prolapse surgery with TOT sling placement
Dr. Levy: For this case, let's consider a 42-year-old woman (P3) who is 6 months post− vaginal hysterectomy. The surgery included ovarian preservation combined with anterior and posterior repair for prolapse as well as apical uterosacral ligament suspension for stage 2 uterovaginal prolapse. A transobturator sling was used.
Extensive preop evaluation was performed, with confirmed symptomatic prolapse. She had no stress incontinence symptoms but did have confirmed occult stress incontinence.
Surgery was uneventful. She resumed intercourse at 8 weeks, but she now has pain with both initial entry and deep penetration. Lubricants and changes in position have not helped. She is in a stable relationship with her husband of 17 years, and she is worried that the sling mesh might be the culprit. On exam, she has no atrophy, pH is 4.5, vaginal length is 8 cm, and there is no prolapse. There is no mesh exposure noted, although she reports slight tenderness with palpation of the right sling arm beneath the right pubic bone.
Dr. Iglesia, what are the patient history questions important to ask here?
Dr. Iglesia: This is not an uncommon scenario--elective surgical correction of occult or latent stress incontinence after surgical correction for pelvic organ prolapse. Now this patient here has no more prolapse complaints; however, she has a new symptom. There are many different causes of dyspareunia; we cannot just assume it is the sling mesh (although with all the legal representation advertisements for those who have had mesh placed, it can certainly be at the top of the patient's mind, causing anxiety and fear).
Multiple trials have looked at prophylactic surgery for incontinence at the time of prolapse repairs. This woman happened to be one of those patients who did not have incontinence symptoms, and they put a sling in. A recent large trial examined women with vaginal prolapse who underwent hysterectomy and suspension.6 (They compared 2 different suspensions.) What is interesting is that 25% of women with prolapse do have baseline pain. However, at 24 months, de novo pain can occur in 10% of women--just from the apical suspension. So, here, it could be the prolapse suspension. Or, in terms of the transobturator sling, long-term data do tell us that the de novo dyspareunia rate ranges on the magnitude of 1% to 9%.7 What is important here is figuring out the cause of the dyspareunia.
Dr. Levy: One of the important points you raised already was that 25% of these women have preoperative pain. So figuring out what her functioning was before surgery and incorporating that into our assessment postop would be pretty important I would think.
Dr. Iglesia: Yes, you need to understand what her typical encounter was before the surgery and how things have changed now that the prolapse is not in the way. Changes obviously can occur with scar tissue, which over time will improve. If she is perimenopausal and starts to get epithelial changes, we can fix that. The question then becomes, "Is the pain emanating from the mesh?"
When examining this patient, it is not uncommon for me to be able to feel "banjo" strings if the mesh is too tight or close to the surface. It is not exposed but it's palpable, and the patient may feel a ridge during penetration. You can ask the patient if pain occurs with different penetration positions. In addition, explore associated neurologic symptoms (numbness or muscle pain in the thigh).
Dr. Kingsberg: There were 2 different sources of pain--on initial entry and at deep penetration. You want to make sure you address both. Importantly, did one precede the other? For instance, if women have pain with penetration they can then end up with an arousal disorder (the length of the vagina cannot increase as much as it might otherwise) and dystonia secondary to the pain with penetration. The timing of the pain--did it all happen at the same time, or did she start out with pain at one point and did it move to something else--is another critical piece of the history.
Dr. Iglesia: It does take a detailed history and physical exam to identify myofascial trigger points. In this case there seems to be pinpoint tenderness directly on the part of the mesh that is not exposed on the right. There are people who feel you should remove the whole mesh, including the arms. Others feel, okay, we can work on these trigger points, with injections, physical therapy, extra lubrication, and neuromodulatory medications. Only then would they think about potentially excising the sling or a portion thereof.
Dr. Krychman: Keep in mind that, even if you do remove the sling, her pain may not subside, if it is secondary to an underlying issue. Because of media sensationalism, she could be focusing on the sling. It is important to set realistic expectations. I often see vulvar pathology or even provoked vestibulodynia that can present with a deep dyspareunia. The concept of collision dyspareunia or introital discomfort or pain on insertion has far reaching implications. We need to look at the patient in totality, ruling out underlying issues related to the bladder, even the colon.
Patient inquires about the benefits of laser treatment for vaginal health
Dr. Levy: Let's move to this last case: A 47-year-old patient who reports lack of sexual satisfaction and attributes it to a loose vagina says, "I've heard about surgery and laser treatment and radiofrequency devices for vaginal health. What are the benefits and risks of these procedures, and will they correct the issues that I'm experiencing?"
How do you approach this patient?
Dr. Krychman: I want to know why this is of paramount importance to her. Is this her actual complaint or is it society's unrealistic expectation of sexual pleasure and performance placed upon her? Or is this a relationship issue surfacing, compounded by physiologic changes? With good communication techniques, like "ask, tell, ask" or effective use of silence (not interrupting), patients will lead us to the reason and the rationale.
Dr. Levy: I have seen a lot of advertising to women, now showing pictures of genitalia, perhaps creating an expectation that we should look infantile in some way. We are creating a sense of beauty and acceptablevisualization of the vagina and vulva that are completely unrealistic. It's fascinating on one hand but it is also disturbing in that some of the direct-to-consumer marketing going on is creating a sense of unease in women who are otherwise perfectly satisfied. Now they take a look at their sagging skin, maybe after having 2 or 3 children, and although they may not look the same they function not so badly perhaps. I think we are creating a distress and an illness model that is interesting to discuss.
Dr. Iglesia, you are in the midst of a randomized trial, giving an informed consent to participants about the expectations for this potential intervention. How do you explain to women what these laser and radiofrequency devices are expected to do and why they might or might not work?
Dr. Iglesia: We are doing a randomized trial for menopausal women who have GSM. We are comparing estrogen cream with fractional CO2 laser therapy. I also am involved in another randomized trial for lichen sclerosus. I am not involved in the cosmetic use of a laser for people who feel their vaginas are just "loose." Like you, Dr. Levy, I am very concerned about the images that women are seeing of the idealized vulva and vagina, and about the rise of cosmetic gynecology, much of which is being performed by plastic surgeons or dermatologists.
A recent article looked at the number of women who are doing pubic hair grooming; the prevalance is about 80% here in America.8 So people have a clear view of what is down there, and then they compare it to what they see in pornographic images on the Internet and want to look like a Barbie doll. That is disturbing because women, particularly young women, do not realize what happens with GSM changes to the vulva and vagina. On the other hand, these laser machines are very expensive, and some doctors are charging thousands of dollars and promising cosmetic and functional results for which we lack long-term, comparative data.
The laser that we are studying is one by Cynosure called Mona Lisa, which works with fractional CO2 and has very low depth of penetration. The concept is that, with microdot therapy (on the order of micrometers), pinpoint destruction will foster regeneration of new collagen and blood flow to the vagina and vulva. We are still in the midst of analyzing this.
Dr. Krychman: I caution people not to lump devices together. There is a significant difference between laser and radiofrequency--especially in the depth of tissue penetration, and level of evidence as well. There are companies performing randomized clinical trials, with well designed sham controls, and have demonstrated clinical efficacy. We need to be cautious of a procedure that is saying it's the best thing since sliced bread, curing interstitial cystitis, dyspareunia, and lichen sclerosus and improving orgasm, lubrication, and arousal. These far reaching, off-label claims are concerning and misleading.
Dr. Levy: I think the important things are 1) shared decision making with the patient and 2) disclosure of what we do not know, which are the long-term results and outcomes and possible downstream negative effects of some of these treatments, since the data we have are so short term.
Dr. Kingsberg: Basics are important. You talked about the pressure for cosmetic appearance, but is that really what is going on for this particular patient? Is she describing a sexual dysfunction when she talks about lack of satisfaction? You need to operationally define that term. Does she have problems with arousal or orgasm or desire and those are what underlie her lack of satisfaction? Is the key to management helping her come to terms with body image issues or to treat a sexual dysfunction? If it truly is a sexual dysfunction, then you can have the shared decision making on preferred treatment approach.
Dr. Levy: This has been an enlightening discussion. Thank all of you for your expertise and clinical acumen.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The age-adjusted prevalence of any sexual problem is 43% among US women. A full 22% of these women experience sexually related personal distress.1 With publication of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition2 has come a shift in classification and, at times, management approach for reported female sexual dysfunction. When women report to their clinicians decreased sexual desire or arousal or pain at penetration, the management is no longer guided by a linear model of sexual response (excitation, plateau, orgasm, and resolution) but rather by a more nuanced and complex biopsychosocial approach. In this model, diagnosis and management strategies to address bothersome sexual concerns consider the whole woman in the context of her physical and psychosocial health. The patient’s age, medical history, and relationship status are among the factors that could affect management of the problem. In an effort to explore this management approach, I used this Update on Female Sexual Dysfunction as an opportunity to convene a roundtable of several experts, representing varying backgrounds and practice vantage points, to discuss 5 cases of sexual problems that you as a busy clinician may encounter in your practice.

Genital atrophy in a sexually inactive 61-year-old woman
Barbara S. Levy, MD: Two years after her husband's death, which followed several years of illness, your 61-year-old patient mentions at her well woman visit that she anticipates becoming sexually active again. She has not used systemic or vaginal hormone therapy. During pelvic examination, atrophic external genital changes are present, and use of an ultrathin (thinner than a Pederson) speculum reveals vaginal epithelial atrophic changes. A single-digit bimanual exam can be performed with moderate patient discomfort; the patient cannot tolerate a 2-digit bimanual exam. She expresses concern about being able to engage in penile/vaginal sexual intercourse.
Dr. Kaunitz, what is important for you to ask this patient, and what concerns you most on her physical exam?
Andrew M. Kaunitz, MD: First, it is important to recognize the patient's expectations and desires. As the case suggests, but further questioning could clarify, she would like to be able to comfortably engage in sexual intercourse with a new partner, but penetration may be difficult (and definitely painful for her) unless treatment is pursued. This combination of mucosal and vestibular atrophic changes (genitourinary syndrome of menopause [GSM], or vulvovaginal atrophy [VVA]) plus the absence of penetration for many years can be a double whammy situation for menopausal women. In this case it has led to extensive contracture of the introitus, and if it is not addressed will cause sexual dysfunction.
Dr. Levy: In addition we need to clarify whether or not a history of breast cancer or some other thing may impact the care we provide. How would you approach talking with this patient in order to manage her care?
Dr. Kaunitz: One step is to see how motivated she is to address this, as it is not something that, as gynecologists, we can snap our fingers and the situation will be resolved. If the patient is motivated to treat the atrophic changes with medical treatment, in the form of low-dose vaginal estrogen, and dilation, either on her own if she's highly motivated to do so, or in my practice more commonly with the support of a women's physical therapist, over time she should be able to comfortably engage in sexual intercourse with penetration. If this is what she wants, we can help steer her in the right direction.
Sheryl Kingsberg, PhD: You know that this woman is motivated by virtue of her initiating the topic herself. Patients are often embarrassed talking about sexual issues, or they are not sure that their gynecologist is comfortable with it. After all, they think, if this is the right place to discuss sexual problems, why didn't he or she ask me? Clinicians must be aware that it is their responsibility to ask about sexual function and not leave it for the patient to open the door.
Dr. Kaunitz: Great point.
Cheryl B. Iglesia, MD: Gratefully, a lot of the atrophic changes this patient demonstrates are reversible. However, other autoimmune diseases (eg, lichen planus, which can affect the vaginal epithelium, or lichen sclerosus, which can affect the clitoris, labia, and vulva) can also cause constriction, and in severe cases, complete obliteration of the vagina and introitus. Women may not be sexually active, and for each annual exam their clinician uses a smaller and smaller speculum--to the point that they cannot even access the cervix anymore--and the vagina can close off. Clinicians may not realize that you need something other than estrogen; with lichen planus you need steroid suppository treatment, and with lichen sclerosus you need topical steroid treatment. So these autoimmune conditions should also be in the differential and, with appropriate treatment, the sexual effects can be reversible.
Michael Krychman, MD: I agree. The vulva can be a great mimicker and, according to the history and physical exam, at some point a vulvoscopy, and even potential biopsies, may be warranted as clinically indicated.
The concept of a comprehensive approach, as Dr. Kingsberg had previously mentioned, involves not only sexual medicine but also evaluating the patient's biopsychosocial variables that may impact her condition. We also need to set realistic expectations. Some women may benefit from off-label use of medications besides estrogen, including topical testosterone. Informed consent is very important with these treatments. I also have had much clinical success with intravaginal diazepam/lorazepam for pelvic floor hypertonus.
In addition, certainly I agree that pelvic floor physical therapy (PT) is a vital treatment component for this patient and, not to diminish its importance, but many women cannot afford, nor do they have the time or opportunity, to go to pelvic floor PT. As clinicians, we can develop and implement effective programs, even in the office, to educate the patient to help herself as well.
Dr. Kaunitz: Absolutely. Also, if, in a clinical setting consistent with atrophic changes, an ObGyn physician is comfortable that vulvovaginal changes noted on exam represent GSM/atrophic changes, I do not feel vulvoscopy is warranted.
Dr. Levy: In conclusion, we need to be aware that pelvic floor PT may not be available everywhere and that a woman's own digits and her partner can also be incorporated into this treatment.
Something that we have all talked about in other venues, but have not looked at in the larger sphere here, is whether there is value to seeing women annually and performing pelvic exams. As Dr. Kingsberg mentioned, this is a highly motivated patient. We have many patients out there who are silent sufferers. The physical exam is an opportunity for us to recognize and address this problem.

Dyspareunia and low sexual desire in a breast cancer survivor
Dr. Levy: In this case, a 36-year-old woman with BRCA1−positive breast cancer has vaginal dryness, painful intercourse, and lowered sexual interest since her treatment, which included chemotherapy after bilateral mastectomies. She has a bilateral salpingo-oophorectomy(BSO) scheduled for primary prevention of her ovarian cancer risk.
Dr. Kingsberg, what is important for you to know to help guide case management?
Dr. Kingsberg: This woman is actually presenting with 2 sexual problems: dyspareunia, which is probably secondary to VVA or GSM, and low sexual desire. Key questions are: 1) When was symptom onset--acquired after treatment or lifelong? 2) Did she develop the dyspareunia and as a result of having pain during sex lost desire to have sex? Or, did she lose desire and then, without it, had no arousal and therefore pain with penetration developed? It also could be that she has 2 distinct problems, VVA and hypoactive sexual desire disorder (HSDD), in which case you need to think about treating both. Finally, we do not actually know if she is having penetrative intercourse or even if she has a partner.
A vulvovaginal exam would give clues as to whether she has VVA, and hormone levels would indicate if she now has chemo-induced menopause. If she is not in menopause now, she certainly is going to be with her BSO. The hormonal changes due to menopause actually can be primarily responsible for both the dyspareunia and HSDD. Management of both symptoms really needs to be based on shared decision making with the patient--with which treatment for which conditions coming first, based on what is causing her the most distress.
I would encourage this woman to treat her VVA since GSM does have long-term physiologic consequences if untreated. The American College of Obstetricians and Gynecologists (ACOG) recommends nonhormonal treatments as first-line treatments, with vaginal estrogen considered if these therapies fail.3 If lubricants and moisturizers and other nonhormonal options are not sufficient, you could consider local estrogen, even though she is a breast cancer survivor, as well as ospemiphene.
If she is distressed by her loss of sexual desire, you can choose to treat her for HSDD. Flibanserin is the first FDA-approved treatment for HSDD. It is only approved in premenopausal women, so it would be considered off-label use if she is postmenopausal (even though she is quite young). You also could consider exogenous testosterone off-label, after consulting with her oncologist.
In addition to the obvious physiologic etiology of her pain and her low desire, the biopsychosocial aspects to consider are: 1) changes to her body image, as she has had bilateral mastectomies, 2) her anxiety about the cancer diagnosis, and 3) concerns about her relationship if she has one--her partner's reactions to her illness and the quality of the relationship outside the bedroom.
Dr. Iglesia: I am seeing here in our nation's capital a lot of advertisements for laser therapy for GSM. I caution women about this because providers are charging a lot of money for this therapy when we do not have long-term safety and effectiveness data for it.
Our group is currently conducting a randomized controlled trial, looking at vaginal estrogen cream versus laser therapy for GSM here at Medstar Health--one of the first in the country as part of a multisite trial. But the North American Menopause Society (NAMS) has come out with a pretty strong statement,4 as has ACOG,5 on this therapy, and I caution people about overzealously offering a very costly procedure targeted to a very vulnerable population, especially to women with personal histories of estrogen-sensitive cancers.
Dr. Krychman: I agree. Very often cancer patients are preyed upon by those offering emerging unproven technologies or medications. We have to work as a coordinated comprehensive team, whether it's a sexual medicine expert, psychologist, urogynecologist, gynecologist, or oncologist, and incorporate the patient's needs and expectations and risk tolerance coupled with treatment efficacy and safety.
Dr. Levy: This was a complex case. The biopsychosocial model is critical here. It's important that we are not siloes in our medical management approach and that we try to help this patient embrace the complexity of her situation. It's not only that she has cancer at age 36; there is a possible guilt factor if she has children and passed that gene on.
Another point that we began to talk about is the fact that in this country we tend to be early adopters of new technology. In our discussion with patients, we should focus on what we know and the risk of the unknowns related to some of the treatment options. But let's discuss lasers a little more later on.
Diminished arousal and orgasmic intensity in a patient taking SSRIs
Dr. Levy: In this next case, a 44-year-old woman in a 15-year marriage notices a change in her orgasmic intensity and latency. She has a supportive husband, and they are attentive to each other's sexual needs. However, she notices a change in her arousal and orgasmic intensity, which has diminished over the last year. She reports that the time to orgasm or latency has increased and both she and her partner are frustrated and getting concerned. She has a history of depression that has been managed by selective serotonin reuptake inhibitors for the past 5 years and has no depressive symptoms currently.
Dr. Krychman, what are you considering before beginning to talk with this patient?
Dr. Krychman: My approach really is a comprehensive one, looking not only at the underlying medical issues but also at the psychological and dynamic relationship facets. We of course also want to look at medications: Has she changed her dose or the timing of when she takes it? Is this a new onset? Finally, we want to know why this is coming to the forefront now. Is it because it is getting worse, or is it because there is some significant issue that is going on in the relationship?
Regarding the physical exam, it is important to rule out underlying genital pelvic pathology. Young women can get changes in the integrity of the pelvic floor, in what I would call the orgasmic matrix--the clitoral tissue, the body, the crura (or arms of the clitoris)--we want to examine and be reassured that her genital anatomy is normal and that there is no underlying pathology that could signal an underlying abnormal hormonal profile. Young women certainly can get lowered estrogen effects at the genital/pelvic tissues (including the labia and vulva), and intravaginally as well. Sometimes women will have pelvic floor hypertonus, as we see with other urinary issues. A thorough pelvic exam is quite vital.
Let's not forget the body that is attached to the genitals; we want to rule out chronic medical disease that may impact her: hypertension, diabetes, or hypercholesterolemia. Untreated, these conditions may directly impact the arousal physiologic mechanisms.
Dr. Levy: In doing this patient's physical exam I would be looking for significant weight gain, and even asking about her partner's weight. Body image can be a huge issue. If she has a history of depression, if she is suffering from a body image problem, she can be feeling unattractive. In my experience this can be a common thing to affect women in their mid-40s.
How would you manage this case?
Dr. Krychman: It is important to divide it up in terms of a conservative to aggressive approach. We want to find out about the relationship. For instance, is the sexual dynamic scripted (ie, boring and predictable)? Is she distracted and frustrated or is she getting enough of the type of stimulation that she likes and enjoys? There certainly are a lot of new devices that are available, whether a self-stimulator or vibrator, the Fiera, or other stimulating devices, that may be important to incorporate into the sexual repertoire. If there is underlying pathology, we want to evaluate and treat that. She may need to be primed, so to speak, with systemic hormones. And does she have issues related to other effects of hormonal deprivation, even local effects? Does she have clitoral atrophy?
There are neutraceuticals that are currently available, whether topical arousal gels or ointments, and we as clinicians need to be critical and evaluate their benefit/risk and look at the data concerning these products. In addition, women who have changes in arousal and in orgasmic intensity and latency may be very frustrated. They describe it as climbing up to a peak but never getting over the top, and this frustration may lead to participant spectatoring, so incorporating a certified sex therapist or counselor is sometimes very critical.
Finally, there are a lot of snake oils, charmers, and charlatan unproven procedures--injecting fillers or other substances into the clitoris are a few examples. I would be a critical clinician, examine the evidence, look at the benefit/risk before advocating an intervention that does not have good clinical data to support its use--a comprehensive approach of sexual medicine as well as sexual psychology.
Dr. Kingsberg: Additionally, we know she is in a long-term relationship--15 years; we want to acknowledge the partner. We talked about the partner's weight, but what about his erectile function? Does he have changes in sexual function that are affecting her, and she is the one carrying the "symptom"?
Looking at each piece separately helps a clinician from getting overwhelmed by the patient who comes in reporting distress with orgasmic dysfunction. We have no pharmacologic FDA-approved treatments, so it can feel off-putting for a clinician to try to fix the reported issue. Looking at each component to help her figure out the underlying cause can be helpful.
Dr. Iglesia: With aging, there can be changes in blood flow, not to mention the hormonal and even peripheral nerve changes, that require more stimulation in order to achieve the desired response. I echo concern about expensive procedures being offered with no evidence, such as the "O" or "G" shot, that can cost up to thousands of dollars.
The other procedure that gives me a lot of angst is clitoral unhooding. The 3 parts of the clitoris are sensitive in terms of innervation and blood flow, and cutting around that delicate tissue goes against the surgical principles required for preserving nerves and blood flow.
New onset pain post−prolapse surgery with TOT sling placement
Dr. Levy: For this case, let's consider a 42-year-old woman (P3) who is 6 months post− vaginal hysterectomy. The surgery included ovarian preservation combined with anterior and posterior repair for prolapse as well as apical uterosacral ligament suspension for stage 2 uterovaginal prolapse. A transobturator sling was used.
Extensive preop evaluation was performed, with confirmed symptomatic prolapse. She had no stress incontinence symptoms but did have confirmed occult stress incontinence.
Surgery was uneventful. She resumed intercourse at 8 weeks, but she now has pain with both initial entry and deep penetration. Lubricants and changes in position have not helped. She is in a stable relationship with her husband of 17 years, and she is worried that the sling mesh might be the culprit. On exam, she has no atrophy, pH is 4.5, vaginal length is 8 cm, and there is no prolapse. There is no mesh exposure noted, although she reports slight tenderness with palpation of the right sling arm beneath the right pubic bone.
Dr. Iglesia, what are the patient history questions important to ask here?
Dr. Iglesia: This is not an uncommon scenario--elective surgical correction of occult or latent stress incontinence after surgical correction for pelvic organ prolapse. Now this patient here has no more prolapse complaints; however, she has a new symptom. There are many different causes of dyspareunia; we cannot just assume it is the sling mesh (although with all the legal representation advertisements for those who have had mesh placed, it can certainly be at the top of the patient's mind, causing anxiety and fear).
Multiple trials have looked at prophylactic surgery for incontinence at the time of prolapse repairs. This woman happened to be one of those patients who did not have incontinence symptoms, and they put a sling in. A recent large trial examined women with vaginal prolapse who underwent hysterectomy and suspension.6 (They compared 2 different suspensions.) What is interesting is that 25% of women with prolapse do have baseline pain. However, at 24 months, de novo pain can occur in 10% of women--just from the apical suspension. So, here, it could be the prolapse suspension. Or, in terms of the transobturator sling, long-term data do tell us that the de novo dyspareunia rate ranges on the magnitude of 1% to 9%.7 What is important here is figuring out the cause of the dyspareunia.
Dr. Levy: One of the important points you raised already was that 25% of these women have preoperative pain. So figuring out what her functioning was before surgery and incorporating that into our assessment postop would be pretty important I would think.
Dr. Iglesia: Yes, you need to understand what her typical encounter was before the surgery and how things have changed now that the prolapse is not in the way. Changes obviously can occur with scar tissue, which over time will improve. If she is perimenopausal and starts to get epithelial changes, we can fix that. The question then becomes, "Is the pain emanating from the mesh?"
When examining this patient, it is not uncommon for me to be able to feel "banjo" strings if the mesh is too tight or close to the surface. It is not exposed but it's palpable, and the patient may feel a ridge during penetration. You can ask the patient if pain occurs with different penetration positions. In addition, explore associated neurologic symptoms (numbness or muscle pain in the thigh).
Dr. Kingsberg: There were 2 different sources of pain--on initial entry and at deep penetration. You want to make sure you address both. Importantly, did one precede the other? For instance, if women have pain with penetration they can then end up with an arousal disorder (the length of the vagina cannot increase as much as it might otherwise) and dystonia secondary to the pain with penetration. The timing of the pain--did it all happen at the same time, or did she start out with pain at one point and did it move to something else--is another critical piece of the history.
Dr. Iglesia: It does take a detailed history and physical exam to identify myofascial trigger points. In this case there seems to be pinpoint tenderness directly on the part of the mesh that is not exposed on the right. There are people who feel you should remove the whole mesh, including the arms. Others feel, okay, we can work on these trigger points, with injections, physical therapy, extra lubrication, and neuromodulatory medications. Only then would they think about potentially excising the sling or a portion thereof.
Dr. Krychman: Keep in mind that, even if you do remove the sling, her pain may not subside, if it is secondary to an underlying issue. Because of media sensationalism, she could be focusing on the sling. It is important to set realistic expectations. I often see vulvar pathology or even provoked vestibulodynia that can present with a deep dyspareunia. The concept of collision dyspareunia or introital discomfort or pain on insertion has far reaching implications. We need to look at the patient in totality, ruling out underlying issues related to the bladder, even the colon.
Patient inquires about the benefits of laser treatment for vaginal health
Dr. Levy: Let's move to this last case: A 47-year-old patient who reports lack of sexual satisfaction and attributes it to a loose vagina says, "I've heard about surgery and laser treatment and radiofrequency devices for vaginal health. What are the benefits and risks of these procedures, and will they correct the issues that I'm experiencing?"
How do you approach this patient?
Dr. Krychman: I want to know why this is of paramount importance to her. Is this her actual complaint or is it society's unrealistic expectation of sexual pleasure and performance placed upon her? Or is this a relationship issue surfacing, compounded by physiologic changes? With good communication techniques, like "ask, tell, ask" or effective use of silence (not interrupting), patients will lead us to the reason and the rationale.
Dr. Levy: I have seen a lot of advertising to women, now showing pictures of genitalia, perhaps creating an expectation that we should look infantile in some way. We are creating a sense of beauty and acceptablevisualization of the vagina and vulva that are completely unrealistic. It's fascinating on one hand but it is also disturbing in that some of the direct-to-consumer marketing going on is creating a sense of unease in women who are otherwise perfectly satisfied. Now they take a look at their sagging skin, maybe after having 2 or 3 children, and although they may not look the same they function not so badly perhaps. I think we are creating a distress and an illness model that is interesting to discuss.
Dr. Iglesia, you are in the midst of a randomized trial, giving an informed consent to participants about the expectations for this potential intervention. How do you explain to women what these laser and radiofrequency devices are expected to do and why they might or might not work?
Dr. Iglesia: We are doing a randomized trial for menopausal women who have GSM. We are comparing estrogen cream with fractional CO2 laser therapy. I also am involved in another randomized trial for lichen sclerosus. I am not involved in the cosmetic use of a laser for people who feel their vaginas are just "loose." Like you, Dr. Levy, I am very concerned about the images that women are seeing of the idealized vulva and vagina, and about the rise of cosmetic gynecology, much of which is being performed by plastic surgeons or dermatologists.
A recent article looked at the number of women who are doing pubic hair grooming; the prevalance is about 80% here in America.8 So people have a clear view of what is down there, and then they compare it to what they see in pornographic images on the Internet and want to look like a Barbie doll. That is disturbing because women, particularly young women, do not realize what happens with GSM changes to the vulva and vagina. On the other hand, these laser machines are very expensive, and some doctors are charging thousands of dollars and promising cosmetic and functional results for which we lack long-term, comparative data.
The laser that we are studying is one by Cynosure called Mona Lisa, which works with fractional CO2 and has very low depth of penetration. The concept is that, with microdot therapy (on the order of micrometers), pinpoint destruction will foster regeneration of new collagen and blood flow to the vagina and vulva. We are still in the midst of analyzing this.
Dr. Krychman: I caution people not to lump devices together. There is a significant difference between laser and radiofrequency--especially in the depth of tissue penetration, and level of evidence as well. There are companies performing randomized clinical trials, with well designed sham controls, and have demonstrated clinical efficacy. We need to be cautious of a procedure that is saying it's the best thing since sliced bread, curing interstitial cystitis, dyspareunia, and lichen sclerosus and improving orgasm, lubrication, and arousal. These far reaching, off-label claims are concerning and misleading.
Dr. Levy: I think the important things are 1) shared decision making with the patient and 2) disclosure of what we do not know, which are the long-term results and outcomes and possible downstream negative effects of some of these treatments, since the data we have are so short term.
Dr. Kingsberg: Basics are important. You talked about the pressure for cosmetic appearance, but is that really what is going on for this particular patient? Is she describing a sexual dysfunction when she talks about lack of satisfaction? You need to operationally define that term. Does she have problems with arousal or orgasm or desire and those are what underlie her lack of satisfaction? Is the key to management helping her come to terms with body image issues or to treat a sexual dysfunction? If it truly is a sexual dysfunction, then you can have the shared decision making on preferred treatment approach.
Dr. Levy: This has been an enlightening discussion. Thank all of you for your expertise and clinical acumen.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112(5):970−978.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM), Fifth Edition. American Psychiatric Association Publishing: Arlington, VA; 2013.
- American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, Farrell R. ACOG Committee Opinion No. 659: The Use of Vaginal Estrogen in Women With a History of Estrogen-Dependent Breast Cancer. Obstet Gynecol. 2016;127(3):e93−e96.
- Krychman ML, Shifren JL, Liu JH, Kingsberg SL, Utian WH. The North American Menopause Society (NAMS). NAMS Menopause e-Consult: Laser treatment safe for vulvovaginal atrophy? 2015;11(3). http://www.medscape.com/viewarticle/846960. Accessed August 17, 2016.
- The American College of Obstetricians and Gynecologists and The American Congress of Obstetricians and Gynecologists. Fractional laser treatment of vulvovaginal atrophy and U.S. Food and Drug Administration clearance: Position Statement. http://www.acog.org/Resources-And-Publications/Position-Statements/Fractional-Laser-Treatment-of-Vulvovaginal-Atrophy-and-US-Food-and-Drug-Administration-Clearance. Published May 2016. Accessed August 17, 2016.
- Lukacz ES, Warren LK, Richter HE, et al. Quality of life and sexual function 2 years after vaginal surgery for prolapse. Obstet Gynecol. 2016;127(6):1071−1079.
- Brubaker L, Chiang S, Zyczynski H, et al. The impact of stress incontinence surgery on female sexual function. Am J Obstet Gynecol. 2009;200(5):562.e1.
- Rowen TS, Gaither TW, Awad MA, Osterberg EC, Shindel AW, Breyer BN. Pubic hair grooming prevalence and motivation among women in the United States [published online ahead of print June 29, 2016]. JAMA Dermatol. doi:10.1001/jamader matol.2016.2154.
- Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112(5):970−978.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM), Fifth Edition. American Psychiatric Association Publishing: Arlington, VA; 2013.
- American College of Obstetricians and Gynecologists’ Committee on Gynecologic Practice, Farrell R. ACOG Committee Opinion No. 659: The Use of Vaginal Estrogen in Women With a History of Estrogen-Dependent Breast Cancer. Obstet Gynecol. 2016;127(3):e93−e96.
- Krychman ML, Shifren JL, Liu JH, Kingsberg SL, Utian WH. The North American Menopause Society (NAMS). NAMS Menopause e-Consult: Laser treatment safe for vulvovaginal atrophy? 2015;11(3). http://www.medscape.com/viewarticle/846960. Accessed August 17, 2016.
- The American College of Obstetricians and Gynecologists and The American Congress of Obstetricians and Gynecologists. Fractional laser treatment of vulvovaginal atrophy and U.S. Food and Drug Administration clearance: Position Statement. http://www.acog.org/Resources-And-Publications/Position-Statements/Fractional-Laser-Treatment-of-Vulvovaginal-Atrophy-and-US-Food-and-Drug-Administration-Clearance. Published May 2016. Accessed August 17, 2016.
- Lukacz ES, Warren LK, Richter HE, et al. Quality of life and sexual function 2 years after vaginal surgery for prolapse. Obstet Gynecol. 2016;127(6):1071−1079.
- Brubaker L, Chiang S, Zyczynski H, et al. The impact of stress incontinence surgery on female sexual function. Am J Obstet Gynecol. 2009;200(5):562.e1.
- Rowen TS, Gaither TW, Awad MA, Osterberg EC, Shindel AW, Breyer BN. Pubic hair grooming prevalence and motivation among women in the United States [published online ahead of print June 29, 2016]. JAMA Dermatol. doi:10.1001/jamader matol.2016.2154.
In This Article
- This roundtable's expert panel
- Dyspareunia and low sexual desire in a breast cancer survivor
- Laser treatment and vaginal health
Can Serum Free Light Chains Be Used for the Early Diagnosis of Monoclonal Immunoglobulin-Secreting B-Cell and Plasma-Cell Diseases? (FULL)
Patients who are undergoing multiple myeloma screening with serum protein electrophoresis and immunofixation, especially those with renal failure, also should receive serum free light chain testing to increase specificity and reduce false-negatives.
Multiple myeloma (MM) is a devastating disease with an estimated 26,850 new cases in 2015 according to Surveillance, Epidemiology, and End Results data and no definitive chemotherapeutic cure.1 In 97% of cases, MM is defined by monoclonal hypergammaglobulinemia, in which a malignant plasma cell clone secretes a monoclonal globulin; the remaining cases are nonsecretors.2 Each pathologically produced clonal globulin contains 2 heavy chains attached by disulfide linkage and 2 light chains. Unchecked plasma cell production is what later causes the symptoms of renal failure, bone destruction, and anemia.
The rate of MM is disproportionately high in the veteran population, and the VA health care system provides care for many of these patients. The higher rate is likely secondary to the predominantly male population, which has higher MM rates, and has been linked to Agent Orange exposure in Vietnam. As MM is not easy to diagnose, any algorithm or testing method would be of great benefit to this population.
The gold standard for MM detection remains serum protein electrophoresis (SPEP) with immunofixation (IFE), but other detection methods have been emerging. The method of serum free light chain (SFLC) assay has become more readily available, and its incorporation into diagnostic guidelines has become more apparent but is not universal.3
In the case series reported in this article, SPEP/IFE and SFLC assays were used to test 207 patients from the VA New York Harbor Healthcare System (VANYHHS). All these patients had a clinical context for MM testing.
Methods
In this retrospective study, the authors reviewed the charts of VANYHHS patients who were being treated for conditions that prompted SPEP/IFE and λ and κ SFLC analysis between December 2013 and March 2014. The study was exempt from institutional review board approval.
The SPEP/IFE analysis was performed with an automated electrophoresis machine (Sebia Electrophoresis), and the SFLC analysis was performed with an automated SFLC assay (Freelite). Sensitivity, specificity, and positive and negative predictive values were calculated using SPEP/IFE as the gold standard and SFLC κ-to-λ ratio asthe test method. Patients with a positive κ-to-λ ratio but negative SPEP were considered false-positives. These patients’ SFLC analyses were further analyzed in an effort to evaluate use of the κ-to-λ ratio as an early tumor marker.
The κ reference range used was 3.3 to 19.4 mg/L, and the λ reference range used was 5.7 to 26.3 mg/L.4 The traditional reference range for the κ-to-λ ratio is 0.26 to 1.65.5
Results
Of the 207 patients in this study, 205 were men. Mean age was 69 years (range, 28-97 years). Mean serum urea nitrogen level was 8.75 mmol/L (range, 2.86-38.21 mmol/L), and mean creatinine level was 140.59 μmol/L (range, 44.21-1503.14 μmol/L). Mean κ was 49.82 mg/L (range, 4.6-700.96 mg/L), and mean λ was 54.27 mg/L (range, 3-1,750 mg/L). Table 1 compares the SPEP and SFLC data. Sensitivity was 67%, specificity was 85%, positive predictive value was 58%, and negative predictive value was 89%. Concordance of the 2 methods was 80%. The false-positive group was followed up 16 months later to check for diagnosis of disease. Two of the 24 patients in this quadrant were later diagnosed with MM (Table 1).
One of the patients with MM was an 82-year-old African American man with a history of hypertension, diabetes, and prostate cancer (Gleason 4 + 4 = 8/10). He presented to VANYHHS after a fall in which he sustained a pathologic fracture of the left acromion. Recurrent prostate cancer was initially suspected, and nuclear bone scintigraphy revealed increased uptake in the left shoulder and the posterior ninth rib. Results of computed tomography-guided biopsy showed the rib lesion packed with plasma cells and consistent with MM. Immunohistochemical analysis was positive for CD138 and κ in the malignant plasma cells. Initial SPEP performed before the biopsy showed an acute phase reaction with hypogammaglobulinemia, and SPEP after the biopsy showed an increased α-2 band but no monoclonal gammaglobulinopathy. The initial κ of 42.18 mg/L (κ-to-λ ratio, 4.01) was up to 67.53 mg/L 4 months later.
The other patient with MM was a 91-year-old man who had coronary artery disease after undergoing coronary artery bypass grafting in 1993, sick sinus syndrome after pacemaker implantation, hypertension, and anemia. He initially presented to the geriatrics clinic with polyneuropathy, which prompted SPEP and SFLC analysis. SPEP results showed a normal electrophoretic pattern, but κ increased to 47.52 mg/L (κ-to-λ ratio, 2.63). The decision was made to monitor the patient in the hematology clinic. Subsequent κ chain analysis revealed an increase to 59.50 mg/L. A repeat SPEP, performed 1 year after the first SPEP, revealed monoclonal immunoglobulin A on IFE.
Of the 24 patients with false-positive results, 16 had moderate-to-severe kidney disease (stage IIIa-IV).6All patients in this quadrant were men; their mean age was 75 years, and their mean creatinine level was 182.15 μmol/L. Further laboratory data are listed in Table 2.
The patient whose biopsy results led to an MM diagnosis and the patient whose IFE led to a gammopathy diagnosis both maintained a glomerular filtration rate within normal limits. The Figure shows the κ-to-λ ratios of this quadrant logarithmically.
Discussion
Use of SFLC analysis as a supplement to serum and urine protein electrophoresis has been investigated and accepted in the recent literature.3,4,7,8 Use of light chains as a method of earlier or alternative detection has not been proved. In the present study of 207 patients, comparisons showed that more traditional MM detection methods and SFLC analysis are largely concordant. The 2 patients with MM and negative electrophoretic patterns provided a clear indication of the potential benefit of SFLC analysis in the diagnosis of secretory and nonsecretory myeloma.
In 2014, Kim and colleagues compared 2 SFLC assays (Freelite, N Latex) to each other and to SPEP in a 120-patient population.9 The Freelite results in their study correlated closely with VA population findings (κ-to-λ ratio sensitivity and specificity: 72.2% and 93.6%, respectively). N Latex, the newer SFLC assay, had lower sensitivity (64.6%) and higher specificity (100%). With application of the extended reference range (0.37-3.1) proposed by Hutchison and colleagues for use in patients with renal failure, SFLC becomes a more statistically powerful tool.5
The patients who tested false-positive had higher mean creatinine levels, and 16 had renal insufficiency. The 2 false-positive patients were later found to have clinical myeloma and were within the normal range of renal function. Of the 16 patients with an abnormal κ-to-λ ratio and renal failure, 15 would be within the revised normal reference range, leaving 9 false-positives, 2 of whom eventually were found to have disease. With the application of the extended light chain range (as per Hutchison) for those patients with renal failure, 15 of the original 24 false-positives became true-negatives. Two of the false-positives become true-positives after they were subsequently diagnosed. Therefore, SFLC analysis detected disease in 22% of the revised false-positives when SPEP could not.
Table 2 lists the revised data after follow-up and renal failure correction. The strongest aspect of SFLC analysis remains its 95% specificity; its 69% sensitivity remains relatively constant. The test’s positive predictive value is 84%, and its negative predictive value is 90%. In veteran and other at-risk populations, SFLC analysis proves to be a very powerful tool on its own.
Conclusion
Both patient cases described in this article demonstrate the usefulness of SFLC analysis as an adjunct to SPEP. The authors propose SFLC testing for all patients who are undergoing MM screening with SPEP/IFE. In patients with renal failure, the expanded reference range seems to reduce erroneous false-positive results. Patients who have abnormal ratios should be followed up in clinic with repeat MM testing. It seems clear that, at the very least, SFLC analysis is a necessary adjunct to SPEP testing. However, SFLC stands on its own merit as well.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. National Cancer Institute, Surveillance, Epidemiology, and End Results (SEER) Program. SEER website. http://seer.cancer.gov/statfacts/html/mulmy.html. Accessed July 11, 2016.
2. Kyle RA, Gertz MA, Witzig TE, et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc. 2003;78(1):21-33.
3. Dimopoulos M, Kyle R, Fermand JP, et al; International Myeloma Workshop Consensus Panel 3. Consensus recommendations for standard investigative workup: report of the International Myeloma Workshop Consensus Panel 3. Blood. 2011;117(18):4701-4705.
4. Katzmann JA, Clark RJ, Abraham RS, et al. Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin Chem. 2002;48(9):1437-1444.
5. Hutchison CA, Plant T, Drayson M, et al. Serum free light chain measurement
aids the diagnosis of myeloma in patients with severe renal failure.
BMC Nephrol. 2008;9:11.
6. Levey AS, Stevens LA, Schmid CH, et al; CKD-EPI (Chronic Kidney Disease
Epidemiology Collaboration). A new equation to estimate glomerular filtration
rate. Ann Intern Med. 2009;150(9):604-612.
7. McTaggart MP, Lindsay J, Kearney EM. Replacing urine protein electrophoresis
with serum free light chain analysis as a first-line test for detecting plasma
cell disorders offers increased diagnostic accuracy and potential health benefit
to patients. Am J Clin Pathol. 2013;140(6):890-897.
8. Abadie JM, Bankson DD. Assessment of serum free light chain assays for
plasma cell disorder screening in a Veterans Affairs population. Ann Clin Lab
Sci. 2006;36(2):157-162.
9. Kim HS, Kim HS, Shin KS, et al. Clinical comparisons of two free light chain
assays to immunofixation electrophoresis for detecting monoclonal gammopathy.
Biomed Res Int. 2014;2014:647238.
Note: Page numbers differ between the print issue and digital edition.
Patients who are undergoing multiple myeloma screening with serum protein electrophoresis and immunofixation, especially those with renal failure, also should receive serum free light chain testing to increase specificity and reduce false-negatives.
Multiple myeloma (MM) is a devastating disease with an estimated 26,850 new cases in 2015 according to Surveillance, Epidemiology, and End Results data and no definitive chemotherapeutic cure.1 In 97% of cases, MM is defined by monoclonal hypergammaglobulinemia, in which a malignant plasma cell clone secretes a monoclonal globulin; the remaining cases are nonsecretors.2 Each pathologically produced clonal globulin contains 2 heavy chains attached by disulfide linkage and 2 light chains. Unchecked plasma cell production is what later causes the symptoms of renal failure, bone destruction, and anemia.
The rate of MM is disproportionately high in the veteran population, and the VA health care system provides care for many of these patients. The higher rate is likely secondary to the predominantly male population, which has higher MM rates, and has been linked to Agent Orange exposure in Vietnam. As MM is not easy to diagnose, any algorithm or testing method would be of great benefit to this population.
The gold standard for MM detection remains serum protein electrophoresis (SPEP) with immunofixation (IFE), but other detection methods have been emerging. The method of serum free light chain (SFLC) assay has become more readily available, and its incorporation into diagnostic guidelines has become more apparent but is not universal.3
In the case series reported in this article, SPEP/IFE and SFLC assays were used to test 207 patients from the VA New York Harbor Healthcare System (VANYHHS). All these patients had a clinical context for MM testing.
Methods
In this retrospective study, the authors reviewed the charts of VANYHHS patients who were being treated for conditions that prompted SPEP/IFE and λ and κ SFLC analysis between December 2013 and March 2014. The study was exempt from institutional review board approval.
The SPEP/IFE analysis was performed with an automated electrophoresis machine (Sebia Electrophoresis), and the SFLC analysis was performed with an automated SFLC assay (Freelite). Sensitivity, specificity, and positive and negative predictive values were calculated using SPEP/IFE as the gold standard and SFLC κ-to-λ ratio asthe test method. Patients with a positive κ-to-λ ratio but negative SPEP were considered false-positives. These patients’ SFLC analyses were further analyzed in an effort to evaluate use of the κ-to-λ ratio as an early tumor marker.
The κ reference range used was 3.3 to 19.4 mg/L, and the λ reference range used was 5.7 to 26.3 mg/L.4 The traditional reference range for the κ-to-λ ratio is 0.26 to 1.65.5
Results
Of the 207 patients in this study, 205 were men. Mean age was 69 years (range, 28-97 years). Mean serum urea nitrogen level was 8.75 mmol/L (range, 2.86-38.21 mmol/L), and mean creatinine level was 140.59 μmol/L (range, 44.21-1503.14 μmol/L). Mean κ was 49.82 mg/L (range, 4.6-700.96 mg/L), and mean λ was 54.27 mg/L (range, 3-1,750 mg/L). Table 1 compares the SPEP and SFLC data. Sensitivity was 67%, specificity was 85%, positive predictive value was 58%, and negative predictive value was 89%. Concordance of the 2 methods was 80%. The false-positive group was followed up 16 months later to check for diagnosis of disease. Two of the 24 patients in this quadrant were later diagnosed with MM (Table 1).
One of the patients with MM was an 82-year-old African American man with a history of hypertension, diabetes, and prostate cancer (Gleason 4 + 4 = 8/10). He presented to VANYHHS after a fall in which he sustained a pathologic fracture of the left acromion. Recurrent prostate cancer was initially suspected, and nuclear bone scintigraphy revealed increased uptake in the left shoulder and the posterior ninth rib. Results of computed tomography-guided biopsy showed the rib lesion packed with plasma cells and consistent with MM. Immunohistochemical analysis was positive for CD138 and κ in the malignant plasma cells. Initial SPEP performed before the biopsy showed an acute phase reaction with hypogammaglobulinemia, and SPEP after the biopsy showed an increased α-2 band but no monoclonal gammaglobulinopathy. The initial κ of 42.18 mg/L (κ-to-λ ratio, 4.01) was up to 67.53 mg/L 4 months later.
The other patient with MM was a 91-year-old man who had coronary artery disease after undergoing coronary artery bypass grafting in 1993, sick sinus syndrome after pacemaker implantation, hypertension, and anemia. He initially presented to the geriatrics clinic with polyneuropathy, which prompted SPEP and SFLC analysis. SPEP results showed a normal electrophoretic pattern, but κ increased to 47.52 mg/L (κ-to-λ ratio, 2.63). The decision was made to monitor the patient in the hematology clinic. Subsequent κ chain analysis revealed an increase to 59.50 mg/L. A repeat SPEP, performed 1 year after the first SPEP, revealed monoclonal immunoglobulin A on IFE.
Of the 24 patients with false-positive results, 16 had moderate-to-severe kidney disease (stage IIIa-IV).6All patients in this quadrant were men; their mean age was 75 years, and their mean creatinine level was 182.15 μmol/L. Further laboratory data are listed in Table 2.
The patient whose biopsy results led to an MM diagnosis and the patient whose IFE led to a gammopathy diagnosis both maintained a glomerular filtration rate within normal limits. The Figure shows the κ-to-λ ratios of this quadrant logarithmically.
Discussion
Use of SFLC analysis as a supplement to serum and urine protein electrophoresis has been investigated and accepted in the recent literature.3,4,7,8 Use of light chains as a method of earlier or alternative detection has not been proved. In the present study of 207 patients, comparisons showed that more traditional MM detection methods and SFLC analysis are largely concordant. The 2 patients with MM and negative electrophoretic patterns provided a clear indication of the potential benefit of SFLC analysis in the diagnosis of secretory and nonsecretory myeloma.
In 2014, Kim and colleagues compared 2 SFLC assays (Freelite, N Latex) to each other and to SPEP in a 120-patient population.9 The Freelite results in their study correlated closely with VA population findings (κ-to-λ ratio sensitivity and specificity: 72.2% and 93.6%, respectively). N Latex, the newer SFLC assay, had lower sensitivity (64.6%) and higher specificity (100%). With application of the extended reference range (0.37-3.1) proposed by Hutchison and colleagues for use in patients with renal failure, SFLC becomes a more statistically powerful tool.5
The patients who tested false-positive had higher mean creatinine levels, and 16 had renal insufficiency. The 2 false-positive patients were later found to have clinical myeloma and were within the normal range of renal function. Of the 16 patients with an abnormal κ-to-λ ratio and renal failure, 15 would be within the revised normal reference range, leaving 9 false-positives, 2 of whom eventually were found to have disease. With the application of the extended light chain range (as per Hutchison) for those patients with renal failure, 15 of the original 24 false-positives became true-negatives. Two of the false-positives become true-positives after they were subsequently diagnosed. Therefore, SFLC analysis detected disease in 22% of the revised false-positives when SPEP could not.
Table 2 lists the revised data after follow-up and renal failure correction. The strongest aspect of SFLC analysis remains its 95% specificity; its 69% sensitivity remains relatively constant. The test’s positive predictive value is 84%, and its negative predictive value is 90%. In veteran and other at-risk populations, SFLC analysis proves to be a very powerful tool on its own.
Conclusion
Both patient cases described in this article demonstrate the usefulness of SFLC analysis as an adjunct to SPEP. The authors propose SFLC testing for all patients who are undergoing MM screening with SPEP/IFE. In patients with renal failure, the expanded reference range seems to reduce erroneous false-positive results. Patients who have abnormal ratios should be followed up in clinic with repeat MM testing. It seems clear that, at the very least, SFLC analysis is a necessary adjunct to SPEP testing. However, SFLC stands on its own merit as well.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
Patients who are undergoing multiple myeloma screening with serum protein electrophoresis and immunofixation, especially those with renal failure, also should receive serum free light chain testing to increase specificity and reduce false-negatives.
Multiple myeloma (MM) is a devastating disease with an estimated 26,850 new cases in 2015 according to Surveillance, Epidemiology, and End Results data and no definitive chemotherapeutic cure.1 In 97% of cases, MM is defined by monoclonal hypergammaglobulinemia, in which a malignant plasma cell clone secretes a monoclonal globulin; the remaining cases are nonsecretors.2 Each pathologically produced clonal globulin contains 2 heavy chains attached by disulfide linkage and 2 light chains. Unchecked plasma cell production is what later causes the symptoms of renal failure, bone destruction, and anemia.
The rate of MM is disproportionately high in the veteran population, and the VA health care system provides care for many of these patients. The higher rate is likely secondary to the predominantly male population, which has higher MM rates, and has been linked to Agent Orange exposure in Vietnam. As MM is not easy to diagnose, any algorithm or testing method would be of great benefit to this population.
The gold standard for MM detection remains serum protein electrophoresis (SPEP) with immunofixation (IFE), but other detection methods have been emerging. The method of serum free light chain (SFLC) assay has become more readily available, and its incorporation into diagnostic guidelines has become more apparent but is not universal.3
In the case series reported in this article, SPEP/IFE and SFLC assays were used to test 207 patients from the VA New York Harbor Healthcare System (VANYHHS). All these patients had a clinical context for MM testing.
Methods
In this retrospective study, the authors reviewed the charts of VANYHHS patients who were being treated for conditions that prompted SPEP/IFE and λ and κ SFLC analysis between December 2013 and March 2014. The study was exempt from institutional review board approval.
The SPEP/IFE analysis was performed with an automated electrophoresis machine (Sebia Electrophoresis), and the SFLC analysis was performed with an automated SFLC assay (Freelite). Sensitivity, specificity, and positive and negative predictive values were calculated using SPEP/IFE as the gold standard and SFLC κ-to-λ ratio asthe test method. Patients with a positive κ-to-λ ratio but negative SPEP were considered false-positives. These patients’ SFLC analyses were further analyzed in an effort to evaluate use of the κ-to-λ ratio as an early tumor marker.
The κ reference range used was 3.3 to 19.4 mg/L, and the λ reference range used was 5.7 to 26.3 mg/L.4 The traditional reference range for the κ-to-λ ratio is 0.26 to 1.65.5
Results
Of the 207 patients in this study, 205 were men. Mean age was 69 years (range, 28-97 years). Mean serum urea nitrogen level was 8.75 mmol/L (range, 2.86-38.21 mmol/L), and mean creatinine level was 140.59 μmol/L (range, 44.21-1503.14 μmol/L). Mean κ was 49.82 mg/L (range, 4.6-700.96 mg/L), and mean λ was 54.27 mg/L (range, 3-1,750 mg/L). Table 1 compares the SPEP and SFLC data. Sensitivity was 67%, specificity was 85%, positive predictive value was 58%, and negative predictive value was 89%. Concordance of the 2 methods was 80%. The false-positive group was followed up 16 months later to check for diagnosis of disease. Two of the 24 patients in this quadrant were later diagnosed with MM (Table 1).
One of the patients with MM was an 82-year-old African American man with a history of hypertension, diabetes, and prostate cancer (Gleason 4 + 4 = 8/10). He presented to VANYHHS after a fall in which he sustained a pathologic fracture of the left acromion. Recurrent prostate cancer was initially suspected, and nuclear bone scintigraphy revealed increased uptake in the left shoulder and the posterior ninth rib. Results of computed tomography-guided biopsy showed the rib lesion packed with plasma cells and consistent with MM. Immunohistochemical analysis was positive for CD138 and κ in the malignant plasma cells. Initial SPEP performed before the biopsy showed an acute phase reaction with hypogammaglobulinemia, and SPEP after the biopsy showed an increased α-2 band but no monoclonal gammaglobulinopathy. The initial κ of 42.18 mg/L (κ-to-λ ratio, 4.01) was up to 67.53 mg/L 4 months later.
The other patient with MM was a 91-year-old man who had coronary artery disease after undergoing coronary artery bypass grafting in 1993, sick sinus syndrome after pacemaker implantation, hypertension, and anemia. He initially presented to the geriatrics clinic with polyneuropathy, which prompted SPEP and SFLC analysis. SPEP results showed a normal electrophoretic pattern, but κ increased to 47.52 mg/L (κ-to-λ ratio, 2.63). The decision was made to monitor the patient in the hematology clinic. Subsequent κ chain analysis revealed an increase to 59.50 mg/L. A repeat SPEP, performed 1 year after the first SPEP, revealed monoclonal immunoglobulin A on IFE.
Of the 24 patients with false-positive results, 16 had moderate-to-severe kidney disease (stage IIIa-IV).6All patients in this quadrant were men; their mean age was 75 years, and their mean creatinine level was 182.15 μmol/L. Further laboratory data are listed in Table 2.
The patient whose biopsy results led to an MM diagnosis and the patient whose IFE led to a gammopathy diagnosis both maintained a glomerular filtration rate within normal limits. The Figure shows the κ-to-λ ratios of this quadrant logarithmically.
Discussion
Use of SFLC analysis as a supplement to serum and urine protein electrophoresis has been investigated and accepted in the recent literature.3,4,7,8 Use of light chains as a method of earlier or alternative detection has not been proved. In the present study of 207 patients, comparisons showed that more traditional MM detection methods and SFLC analysis are largely concordant. The 2 patients with MM and negative electrophoretic patterns provided a clear indication of the potential benefit of SFLC analysis in the diagnosis of secretory and nonsecretory myeloma.
In 2014, Kim and colleagues compared 2 SFLC assays (Freelite, N Latex) to each other and to SPEP in a 120-patient population.9 The Freelite results in their study correlated closely with VA population findings (κ-to-λ ratio sensitivity and specificity: 72.2% and 93.6%, respectively). N Latex, the newer SFLC assay, had lower sensitivity (64.6%) and higher specificity (100%). With application of the extended reference range (0.37-3.1) proposed by Hutchison and colleagues for use in patients with renal failure, SFLC becomes a more statistically powerful tool.5
The patients who tested false-positive had higher mean creatinine levels, and 16 had renal insufficiency. The 2 false-positive patients were later found to have clinical myeloma and were within the normal range of renal function. Of the 16 patients with an abnormal κ-to-λ ratio and renal failure, 15 would be within the revised normal reference range, leaving 9 false-positives, 2 of whom eventually were found to have disease. With the application of the extended light chain range (as per Hutchison) for those patients with renal failure, 15 of the original 24 false-positives became true-negatives. Two of the false-positives become true-positives after they were subsequently diagnosed. Therefore, SFLC analysis detected disease in 22% of the revised false-positives when SPEP could not.
Table 2 lists the revised data after follow-up and renal failure correction. The strongest aspect of SFLC analysis remains its 95% specificity; its 69% sensitivity remains relatively constant. The test’s positive predictive value is 84%, and its negative predictive value is 90%. In veteran and other at-risk populations, SFLC analysis proves to be a very powerful tool on its own.
Conclusion
Both patient cases described in this article demonstrate the usefulness of SFLC analysis as an adjunct to SPEP. The authors propose SFLC testing for all patients who are undergoing MM screening with SPEP/IFE. In patients with renal failure, the expanded reference range seems to reduce erroneous false-positive results. Patients who have abnormal ratios should be followed up in clinic with repeat MM testing. It seems clear that, at the very least, SFLC analysis is a necessary adjunct to SPEP testing. However, SFLC stands on its own merit as well.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. National Cancer Institute, Surveillance, Epidemiology, and End Results (SEER) Program. SEER website. http://seer.cancer.gov/statfacts/html/mulmy.html. Accessed July 11, 2016.
2. Kyle RA, Gertz MA, Witzig TE, et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc. 2003;78(1):21-33.
3. Dimopoulos M, Kyle R, Fermand JP, et al; International Myeloma Workshop Consensus Panel 3. Consensus recommendations for standard investigative workup: report of the International Myeloma Workshop Consensus Panel 3. Blood. 2011;117(18):4701-4705.
4. Katzmann JA, Clark RJ, Abraham RS, et al. Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin Chem. 2002;48(9):1437-1444.
5. Hutchison CA, Plant T, Drayson M, et al. Serum free light chain measurement
aids the diagnosis of myeloma in patients with severe renal failure.
BMC Nephrol. 2008;9:11.
6. Levey AS, Stevens LA, Schmid CH, et al; CKD-EPI (Chronic Kidney Disease
Epidemiology Collaboration). A new equation to estimate glomerular filtration
rate. Ann Intern Med. 2009;150(9):604-612.
7. McTaggart MP, Lindsay J, Kearney EM. Replacing urine protein electrophoresis
with serum free light chain analysis as a first-line test for detecting plasma
cell disorders offers increased diagnostic accuracy and potential health benefit
to patients. Am J Clin Pathol. 2013;140(6):890-897.
8. Abadie JM, Bankson DD. Assessment of serum free light chain assays for
plasma cell disorder screening in a Veterans Affairs population. Ann Clin Lab
Sci. 2006;36(2):157-162.
9. Kim HS, Kim HS, Shin KS, et al. Clinical comparisons of two free light chain
assays to immunofixation electrophoresis for detecting monoclonal gammopathy.
Biomed Res Int. 2014;2014:647238.
Note: Page numbers differ between the print issue and digital edition.
1. National Cancer Institute, Surveillance, Epidemiology, and End Results (SEER) Program. SEER website. http://seer.cancer.gov/statfacts/html/mulmy.html. Accessed July 11, 2016.
2. Kyle RA, Gertz MA, Witzig TE, et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc. 2003;78(1):21-33.
3. Dimopoulos M, Kyle R, Fermand JP, et al; International Myeloma Workshop Consensus Panel 3. Consensus recommendations for standard investigative workup: report of the International Myeloma Workshop Consensus Panel 3. Blood. 2011;117(18):4701-4705.
4. Katzmann JA, Clark RJ, Abraham RS, et al. Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin Chem. 2002;48(9):1437-1444.
5. Hutchison CA, Plant T, Drayson M, et al. Serum free light chain measurement
aids the diagnosis of myeloma in patients with severe renal failure.
BMC Nephrol. 2008;9:11.
6. Levey AS, Stevens LA, Schmid CH, et al; CKD-EPI (Chronic Kidney Disease
Epidemiology Collaboration). A new equation to estimate glomerular filtration
rate. Ann Intern Med. 2009;150(9):604-612.
7. McTaggart MP, Lindsay J, Kearney EM. Replacing urine protein electrophoresis
with serum free light chain analysis as a first-line test for detecting plasma
cell disorders offers increased diagnostic accuracy and potential health benefit
to patients. Am J Clin Pathol. 2013;140(6):890-897.
8. Abadie JM, Bankson DD. Assessment of serum free light chain assays for
plasma cell disorder screening in a Veterans Affairs population. Ann Clin Lab
Sci. 2006;36(2):157-162.
9. Kim HS, Kim HS, Shin KS, et al. Clinical comparisons of two free light chain
assays to immunofixation electrophoresis for detecting monoclonal gammopathy.
Biomed Res Int. 2014;2014:647238.
Note: Page numbers differ between the print issue and digital edition.
An Unusual Cause of Shortness of Breath: Primary Tracheal Basal Cell Adenocarcinoma
Salivary gland lung tumors are extremely rare intrathoracic malignancies, accounting for only 0.2% of all lung tumors.1 It has been postulated that these lung tumors arise from pluripotential cells in the epithelium of the submucosal bronchial glands and usually present as endoluminal lesions. The cause of salivary gland tumors is unclear. They seem to be unrelated to exposure to smoking, air pollutants, or other chemicals.2 Associated symptoms are generally related to endoluminal obstruction by the tumors, which are centrally located. Thus, presenting symptoms commonly include chronic cough, progressive dyspnea, hoarseness, wheezing, and occasional hemoptysis.3 Chest radiographs seem normal in most cases except those in which obstruction is present. Computed tomography usually shows well-defined endotracheal or endobronchial lesions that are lobulated, polypoid, or smooth, without infiltration into surrounding tissues.
Although basal cell adenocarcinoma (BCAC) was included in World Health Organization’s (WHO) Pathology and Genetics of Head and Neck Tumours in 1991, its definition—“an epithelial neoplasm that has cytological characteristics of basal cell adenoma (BCA), but a morphologic growth pattern indicative of malignancy”—did not appear until 2005.4 Basal cell adenocarcinoma generally is classified as a low-grade malignancy with a good long-term prognosis. It usually affects parotid and submandibular minor salivary glands.5
Histologic differentiation of BCAC and BCA is difficult and depends on whether local structures have been invaded or on which histologic features of perineural or vascular invasion are present.6 Basal cell adenocarcinoma also shows strong immunoreactivity to cytokeratin 7 (CK-7) and variable myoepithelial staining with S100.7 Because of the prognosis and potential treatment differences involved, BCAC must be differentiated from other basaloid cell tumors, such as BCA, adenoid cystic carcinoma, polymorphous low-grade adenocarcinoma, myoepithelial tumor, epithelial-myoepithelial carcinoma, and basaloid squamous cell carcinoma (SCC).8 Surgical excision is the primary treatment of choice.5 Rare in the salivary glands, BCA and BCAC are even rarer in salivary gland tissue outside the head and neck region. The authors report on a case of BCAC of the salivary gland tissue in the trachea.
Case Report
An 84-year-old man with diabetes mellitus and hypertension and a nonsmoker presented to the emergency department of the VA Caribbean Healthcare System in San Juan, Puerto Rico, with a dry cough and shortness of breath lasting 1 week, which worsened the day before
presentation. On physical examination, the patient was afebrile and in no respiratory distress, and his vital signs were within normal limits. There was no barrel chest, no prolonged expiratory phase, and occasional wheezing more prominent on the left side. A chest radiograph showed atelectasis and/or associated changes (Figure 1).
After the initial biopsy results led to a provisional diagnosis of basaloid neoplasm with squamous features, the patient underwent rigid bronchoscopic tracheal tumor debridement followed by cryotherapy at the base of the tumor.
Discussion
Primary tracheal tumors are rare, accounting for < 1% of all malignancies.9,10 According to the National Cancer Institute Surveillance, Epidemiology, and End Results database, the rate of new cases of primary carcinoma of the trachea was 2.6 per 1 million people per year.11 Of all primary tumors of the trachea, 80% are malignant9,10; the rest vary widely and include both malignant and benign histotypes.11
The abundant minor salivary gland tissue in the upper respiratory tract can potentially develop neoplasia the same as the wide spectrum seen in head and neck salivary gland tumors. The most recent (2004) WHO Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart includes a brief section on salivary gland tumors and mentions only mucoepidermoid carcinoma, adenoid cystic carcinoma, myoepithelial carcinoma, epithelial-myoepithelial carcinoma, pleomorphic adenoma, and carcinoma ex-pleomorphic adenoma.12 However, there are reports of other salivary gland tumors in the tracheopulmonary location.
García and colleagues recently described a case of hyalinizing clear cell carcinoma with its defining EWSR1-ATF1 (Ewing sarcoma breakpoint region 1–activating transcription factor 1) fusion transcript.13 Similarly, no tracheopulmonary BCAC cases have been reported in the English-language literature since 2004, when Damiani and colleagues described a case of basal adenocarcinoma of the lung.14 In 2012, Yamada and colleagues reported a case of “basaloid carcinoma with central cavitation,”15 which showed a peculiar immunohistochemical profile similar
to the tumor described in the present article—suggestive of a myoepithelial origin if the morphologic architectural features match.
Salivary gland tumors are prone to manifest the squamous phenotype. Depending on the biopsy modality, these squamous areas (squamous eddies) can be sampled and can lead to the erroneous diagnosis of SCC. However, the histologic features of squamous malignancy are
lacking in these squamous components, and the pathologist should be able to distinguish additional phenotypes that can generate a wider differential diagnosis.
On follow-up, this patient has maintained satisfactory respiratory function. Starting 2 years after the initial resection, he has had annual checkups. Recent imaging and bronchoscopy have revealed tumor recurrence in the same site. Because of the patient’s extensive comorbidities, surgical intervention has been deferred in favor of cryotherapy, a less aggressive therapy.
Author disclosure
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. Moran CA. Primary salivary gland-type tumors of the lung. Semin Diagn Pathol.
1995;12(2):106-122.
2. Molina JR, Aubry MC, Lewis JE, et al. Primary salivary gland-type lung cancer: spectrum of clinical presentation, histopathologic and prognostic factors. Cancer. 2007;110(10):2253-2259.
3. Brutinel WM, Cortese DA, McDougall JC, Gillio RG, Bergstralh EJ. A two-year experience with the neodymium-YAG laser in endobronchial obstruction. Chest. 1987;91(2):159-165.
4. Barnes L, Eveson JW, Reichart P, Sidransky D, eds. World Health Organization Classification of Tumours. Pathology and Genetics of Head and Neck Tumours. Lyon, France: IARC Press; 2005. International Agency for Research on Cancer website. https://www.iarc.fr/en/publications/pdfs-online/pat-gen/bb9/BB9.pdf. Accessed June 16, 2016.
5. Sarath PV, Kannan N, Patil R, Manne RK, Swapna B, Suneel Kumar KV. Basal cell adenocarcinoma of the minor salivary glands involving palate and maxillary sinus. J Clin Imaging Sci. 2013;3(suppl 1):4.
6. Farrell T, Chang YL. Basal cell adenocarcinoma of minor salivary glands. Arch Pathol Lab Med. 2007;131(10):1602-1604.
7. Jung MJ, Roh JL, Choi SH, et al. Basal cell adenocarcinoma of the salivary gland: a morphological and immunohistochemical comparison with basal cell adenoma with and without capsular invasion. Diagn Pathol. 2013;8:171.
8. Jäkel KT, Löning T. Differential diagnosis of basaloid salivary gland tumors [in German]. Pathologe. 2004;25(1):46-55.
9. Urdaneta AI, Yu JB, Wilson LD. Population based cancer registry analysis of primary tracheal carcinoma. Am J Clin Oncol. 2011;34(1):32-37.
10. Varadhachary GR, Rabe MN. Cancer of unknown primary site. N Engl J Med. 2014;371(8):757-765.
11. Pelosi G, Fraggetta F, Maffini F, Solli P, Cavallon A, Viale G. Pulmonary epithelialmyoepithelial tumor of unproven malignant potential: report of a case and review
of the literature. Mod Pathol. 2001;14(5):521-526.
12. Travis WD, Brambilla E, Muller-Hermelink HK, Harris CC, eds. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon, France: IARC Press; 2004. International Agency for Research on Cancer website. https://www.iarc.fr/en/publications/pdfs-online /pat-gen/bb10/BB10.pdf. Accessed June 29, 2016.
13. García JJ, Jin L, Jackson SB, et al. Primary pulmonary hyalinizing clear cell carcinoma of bronchial submucosal gland origin. Hum Pathol. 2015;46(3):471-475.
14. Damiani S, Magrini E, Farnedi A, Pession A. Basal cell (myoepithelial) adenocarcinoma of the lung. First case with cytogenetic findings. Histopathology. 2004;45(4):422-424.
15. Yamada S, Noguchi H, Nabeshima A, et al. Basaloid carcinoma of the lung associated with central cavitation: a unique surgical case focusing on cytological and immunohistochemical findings. Diagn Pathol. 2012;7:175-180.
Note: Page numbers differ between the print issue and digital edition.
Salivary gland lung tumors are extremely rare intrathoracic malignancies, accounting for only 0.2% of all lung tumors.1 It has been postulated that these lung tumors arise from pluripotential cells in the epithelium of the submucosal bronchial glands and usually present as endoluminal lesions. The cause of salivary gland tumors is unclear. They seem to be unrelated to exposure to smoking, air pollutants, or other chemicals.2 Associated symptoms are generally related to endoluminal obstruction by the tumors, which are centrally located. Thus, presenting symptoms commonly include chronic cough, progressive dyspnea, hoarseness, wheezing, and occasional hemoptysis.3 Chest radiographs seem normal in most cases except those in which obstruction is present. Computed tomography usually shows well-defined endotracheal or endobronchial lesions that are lobulated, polypoid, or smooth, without infiltration into surrounding tissues.
Although basal cell adenocarcinoma (BCAC) was included in World Health Organization’s (WHO) Pathology and Genetics of Head and Neck Tumours in 1991, its definition—“an epithelial neoplasm that has cytological characteristics of basal cell adenoma (BCA), but a morphologic growth pattern indicative of malignancy”—did not appear until 2005.4 Basal cell adenocarcinoma generally is classified as a low-grade malignancy with a good long-term prognosis. It usually affects parotid and submandibular minor salivary glands.5
Histologic differentiation of BCAC and BCA is difficult and depends on whether local structures have been invaded or on which histologic features of perineural or vascular invasion are present.6 Basal cell adenocarcinoma also shows strong immunoreactivity to cytokeratin 7 (CK-7) and variable myoepithelial staining with S100.7 Because of the prognosis and potential treatment differences involved, BCAC must be differentiated from other basaloid cell tumors, such as BCA, adenoid cystic carcinoma, polymorphous low-grade adenocarcinoma, myoepithelial tumor, epithelial-myoepithelial carcinoma, and basaloid squamous cell carcinoma (SCC).8 Surgical excision is the primary treatment of choice.5 Rare in the salivary glands, BCA and BCAC are even rarer in salivary gland tissue outside the head and neck region. The authors report on a case of BCAC of the salivary gland tissue in the trachea.
Case Report
An 84-year-old man with diabetes mellitus and hypertension and a nonsmoker presented to the emergency department of the VA Caribbean Healthcare System in San Juan, Puerto Rico, with a dry cough and shortness of breath lasting 1 week, which worsened the day before
presentation. On physical examination, the patient was afebrile and in no respiratory distress, and his vital signs were within normal limits. There was no barrel chest, no prolonged expiratory phase, and occasional wheezing more prominent on the left side. A chest radiograph showed atelectasis and/or associated changes (Figure 1).
After the initial biopsy results led to a provisional diagnosis of basaloid neoplasm with squamous features, the patient underwent rigid bronchoscopic tracheal tumor debridement followed by cryotherapy at the base of the tumor.
Discussion
Primary tracheal tumors are rare, accounting for < 1% of all malignancies.9,10 According to the National Cancer Institute Surveillance, Epidemiology, and End Results database, the rate of new cases of primary carcinoma of the trachea was 2.6 per 1 million people per year.11 Of all primary tumors of the trachea, 80% are malignant9,10; the rest vary widely and include both malignant and benign histotypes.11
The abundant minor salivary gland tissue in the upper respiratory tract can potentially develop neoplasia the same as the wide spectrum seen in head and neck salivary gland tumors. The most recent (2004) WHO Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart includes a brief section on salivary gland tumors and mentions only mucoepidermoid carcinoma, adenoid cystic carcinoma, myoepithelial carcinoma, epithelial-myoepithelial carcinoma, pleomorphic adenoma, and carcinoma ex-pleomorphic adenoma.12 However, there are reports of other salivary gland tumors in the tracheopulmonary location.
García and colleagues recently described a case of hyalinizing clear cell carcinoma with its defining EWSR1-ATF1 (Ewing sarcoma breakpoint region 1–activating transcription factor 1) fusion transcript.13 Similarly, no tracheopulmonary BCAC cases have been reported in the English-language literature since 2004, when Damiani and colleagues described a case of basal adenocarcinoma of the lung.14 In 2012, Yamada and colleagues reported a case of “basaloid carcinoma with central cavitation,”15 which showed a peculiar immunohistochemical profile similar
to the tumor described in the present article—suggestive of a myoepithelial origin if the morphologic architectural features match.
Salivary gland tumors are prone to manifest the squamous phenotype. Depending on the biopsy modality, these squamous areas (squamous eddies) can be sampled and can lead to the erroneous diagnosis of SCC. However, the histologic features of squamous malignancy are
lacking in these squamous components, and the pathologist should be able to distinguish additional phenotypes that can generate a wider differential diagnosis.
On follow-up, this patient has maintained satisfactory respiratory function. Starting 2 years after the initial resection, he has had annual checkups. Recent imaging and bronchoscopy have revealed tumor recurrence in the same site. Because of the patient’s extensive comorbidities, surgical intervention has been deferred in favor of cryotherapy, a less aggressive therapy.
Author disclosure
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
Salivary gland lung tumors are extremely rare intrathoracic malignancies, accounting for only 0.2% of all lung tumors.1 It has been postulated that these lung tumors arise from pluripotential cells in the epithelium of the submucosal bronchial glands and usually present as endoluminal lesions. The cause of salivary gland tumors is unclear. They seem to be unrelated to exposure to smoking, air pollutants, or other chemicals.2 Associated symptoms are generally related to endoluminal obstruction by the tumors, which are centrally located. Thus, presenting symptoms commonly include chronic cough, progressive dyspnea, hoarseness, wheezing, and occasional hemoptysis.3 Chest radiographs seem normal in most cases except those in which obstruction is present. Computed tomography usually shows well-defined endotracheal or endobronchial lesions that are lobulated, polypoid, or smooth, without infiltration into surrounding tissues.
Although basal cell adenocarcinoma (BCAC) was included in World Health Organization’s (WHO) Pathology and Genetics of Head and Neck Tumours in 1991, its definition—“an epithelial neoplasm that has cytological characteristics of basal cell adenoma (BCA), but a morphologic growth pattern indicative of malignancy”—did not appear until 2005.4 Basal cell adenocarcinoma generally is classified as a low-grade malignancy with a good long-term prognosis. It usually affects parotid and submandibular minor salivary glands.5
Histologic differentiation of BCAC and BCA is difficult and depends on whether local structures have been invaded or on which histologic features of perineural or vascular invasion are present.6 Basal cell adenocarcinoma also shows strong immunoreactivity to cytokeratin 7 (CK-7) and variable myoepithelial staining with S100.7 Because of the prognosis and potential treatment differences involved, BCAC must be differentiated from other basaloid cell tumors, such as BCA, adenoid cystic carcinoma, polymorphous low-grade adenocarcinoma, myoepithelial tumor, epithelial-myoepithelial carcinoma, and basaloid squamous cell carcinoma (SCC).8 Surgical excision is the primary treatment of choice.5 Rare in the salivary glands, BCA and BCAC are even rarer in salivary gland tissue outside the head and neck region. The authors report on a case of BCAC of the salivary gland tissue in the trachea.
Case Report
An 84-year-old man with diabetes mellitus and hypertension and a nonsmoker presented to the emergency department of the VA Caribbean Healthcare System in San Juan, Puerto Rico, with a dry cough and shortness of breath lasting 1 week, which worsened the day before
presentation. On physical examination, the patient was afebrile and in no respiratory distress, and his vital signs were within normal limits. There was no barrel chest, no prolonged expiratory phase, and occasional wheezing more prominent on the left side. A chest radiograph showed atelectasis and/or associated changes (Figure 1).
After the initial biopsy results led to a provisional diagnosis of basaloid neoplasm with squamous features, the patient underwent rigid bronchoscopic tracheal tumor debridement followed by cryotherapy at the base of the tumor.
Discussion
Primary tracheal tumors are rare, accounting for < 1% of all malignancies.9,10 According to the National Cancer Institute Surveillance, Epidemiology, and End Results database, the rate of new cases of primary carcinoma of the trachea was 2.6 per 1 million people per year.11 Of all primary tumors of the trachea, 80% are malignant9,10; the rest vary widely and include both malignant and benign histotypes.11
The abundant minor salivary gland tissue in the upper respiratory tract can potentially develop neoplasia the same as the wide spectrum seen in head and neck salivary gland tumors. The most recent (2004) WHO Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart includes a brief section on salivary gland tumors and mentions only mucoepidermoid carcinoma, adenoid cystic carcinoma, myoepithelial carcinoma, epithelial-myoepithelial carcinoma, pleomorphic adenoma, and carcinoma ex-pleomorphic adenoma.12 However, there are reports of other salivary gland tumors in the tracheopulmonary location.
García and colleagues recently described a case of hyalinizing clear cell carcinoma with its defining EWSR1-ATF1 (Ewing sarcoma breakpoint region 1–activating transcription factor 1) fusion transcript.13 Similarly, no tracheopulmonary BCAC cases have been reported in the English-language literature since 2004, when Damiani and colleagues described a case of basal adenocarcinoma of the lung.14 In 2012, Yamada and colleagues reported a case of “basaloid carcinoma with central cavitation,”15 which showed a peculiar immunohistochemical profile similar
to the tumor described in the present article—suggestive of a myoepithelial origin if the morphologic architectural features match.
Salivary gland tumors are prone to manifest the squamous phenotype. Depending on the biopsy modality, these squamous areas (squamous eddies) can be sampled and can lead to the erroneous diagnosis of SCC. However, the histologic features of squamous malignancy are
lacking in these squamous components, and the pathologist should be able to distinguish additional phenotypes that can generate a wider differential diagnosis.
On follow-up, this patient has maintained satisfactory respiratory function. Starting 2 years after the initial resection, he has had annual checkups. Recent imaging and bronchoscopy have revealed tumor recurrence in the same site. Because of the patient’s extensive comorbidities, surgical intervention has been deferred in favor of cryotherapy, a less aggressive therapy.
Author disclosure
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. Moran CA. Primary salivary gland-type tumors of the lung. Semin Diagn Pathol.
1995;12(2):106-122.
2. Molina JR, Aubry MC, Lewis JE, et al. Primary salivary gland-type lung cancer: spectrum of clinical presentation, histopathologic and prognostic factors. Cancer. 2007;110(10):2253-2259.
3. Brutinel WM, Cortese DA, McDougall JC, Gillio RG, Bergstralh EJ. A two-year experience with the neodymium-YAG laser in endobronchial obstruction. Chest. 1987;91(2):159-165.
4. Barnes L, Eveson JW, Reichart P, Sidransky D, eds. World Health Organization Classification of Tumours. Pathology and Genetics of Head and Neck Tumours. Lyon, France: IARC Press; 2005. International Agency for Research on Cancer website. https://www.iarc.fr/en/publications/pdfs-online/pat-gen/bb9/BB9.pdf. Accessed June 16, 2016.
5. Sarath PV, Kannan N, Patil R, Manne RK, Swapna B, Suneel Kumar KV. Basal cell adenocarcinoma of the minor salivary glands involving palate and maxillary sinus. J Clin Imaging Sci. 2013;3(suppl 1):4.
6. Farrell T, Chang YL. Basal cell adenocarcinoma of minor salivary glands. Arch Pathol Lab Med. 2007;131(10):1602-1604.
7. Jung MJ, Roh JL, Choi SH, et al. Basal cell adenocarcinoma of the salivary gland: a morphological and immunohistochemical comparison with basal cell adenoma with and without capsular invasion. Diagn Pathol. 2013;8:171.
8. Jäkel KT, Löning T. Differential diagnosis of basaloid salivary gland tumors [in German]. Pathologe. 2004;25(1):46-55.
9. Urdaneta AI, Yu JB, Wilson LD. Population based cancer registry analysis of primary tracheal carcinoma. Am J Clin Oncol. 2011;34(1):32-37.
10. Varadhachary GR, Rabe MN. Cancer of unknown primary site. N Engl J Med. 2014;371(8):757-765.
11. Pelosi G, Fraggetta F, Maffini F, Solli P, Cavallon A, Viale G. Pulmonary epithelialmyoepithelial tumor of unproven malignant potential: report of a case and review
of the literature. Mod Pathol. 2001;14(5):521-526.
12. Travis WD, Brambilla E, Muller-Hermelink HK, Harris CC, eds. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon, France: IARC Press; 2004. International Agency for Research on Cancer website. https://www.iarc.fr/en/publications/pdfs-online /pat-gen/bb10/BB10.pdf. Accessed June 29, 2016.
13. García JJ, Jin L, Jackson SB, et al. Primary pulmonary hyalinizing clear cell carcinoma of bronchial submucosal gland origin. Hum Pathol. 2015;46(3):471-475.
14. Damiani S, Magrini E, Farnedi A, Pession A. Basal cell (myoepithelial) adenocarcinoma of the lung. First case with cytogenetic findings. Histopathology. 2004;45(4):422-424.
15. Yamada S, Noguchi H, Nabeshima A, et al. Basaloid carcinoma of the lung associated with central cavitation: a unique surgical case focusing on cytological and immunohistochemical findings. Diagn Pathol. 2012;7:175-180.
Note: Page numbers differ between the print issue and digital edition.
1. Moran CA. Primary salivary gland-type tumors of the lung. Semin Diagn Pathol.
1995;12(2):106-122.
2. Molina JR, Aubry MC, Lewis JE, et al. Primary salivary gland-type lung cancer: spectrum of clinical presentation, histopathologic and prognostic factors. Cancer. 2007;110(10):2253-2259.
3. Brutinel WM, Cortese DA, McDougall JC, Gillio RG, Bergstralh EJ. A two-year experience with the neodymium-YAG laser in endobronchial obstruction. Chest. 1987;91(2):159-165.
4. Barnes L, Eveson JW, Reichart P, Sidransky D, eds. World Health Organization Classification of Tumours. Pathology and Genetics of Head and Neck Tumours. Lyon, France: IARC Press; 2005. International Agency for Research on Cancer website. https://www.iarc.fr/en/publications/pdfs-online/pat-gen/bb9/BB9.pdf. Accessed June 16, 2016.
5. Sarath PV, Kannan N, Patil R, Manne RK, Swapna B, Suneel Kumar KV. Basal cell adenocarcinoma of the minor salivary glands involving palate and maxillary sinus. J Clin Imaging Sci. 2013;3(suppl 1):4.
6. Farrell T, Chang YL. Basal cell adenocarcinoma of minor salivary glands. Arch Pathol Lab Med. 2007;131(10):1602-1604.
7. Jung MJ, Roh JL, Choi SH, et al. Basal cell adenocarcinoma of the salivary gland: a morphological and immunohistochemical comparison with basal cell adenoma with and without capsular invasion. Diagn Pathol. 2013;8:171.
8. Jäkel KT, Löning T. Differential diagnosis of basaloid salivary gland tumors [in German]. Pathologe. 2004;25(1):46-55.
9. Urdaneta AI, Yu JB, Wilson LD. Population based cancer registry analysis of primary tracheal carcinoma. Am J Clin Oncol. 2011;34(1):32-37.
10. Varadhachary GR, Rabe MN. Cancer of unknown primary site. N Engl J Med. 2014;371(8):757-765.
11. Pelosi G, Fraggetta F, Maffini F, Solli P, Cavallon A, Viale G. Pulmonary epithelialmyoepithelial tumor of unproven malignant potential: report of a case and review
of the literature. Mod Pathol. 2001;14(5):521-526.
12. Travis WD, Brambilla E, Muller-Hermelink HK, Harris CC, eds. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon, France: IARC Press; 2004. International Agency for Research on Cancer website. https://www.iarc.fr/en/publications/pdfs-online /pat-gen/bb10/BB10.pdf. Accessed June 29, 2016.
13. García JJ, Jin L, Jackson SB, et al. Primary pulmonary hyalinizing clear cell carcinoma of bronchial submucosal gland origin. Hum Pathol. 2015;46(3):471-475.
14. Damiani S, Magrini E, Farnedi A, Pession A. Basal cell (myoepithelial) adenocarcinoma of the lung. First case with cytogenetic findings. Histopathology. 2004;45(4):422-424.
15. Yamada S, Noguchi H, Nabeshima A, et al. Basaloid carcinoma of the lung associated with central cavitation: a unique surgical case focusing on cytological and immunohistochemical findings. Diagn Pathol. 2012;7:175-180.
Note: Page numbers differ between the print issue and digital edition.