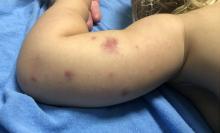
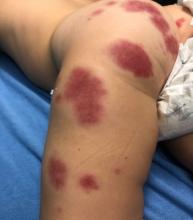
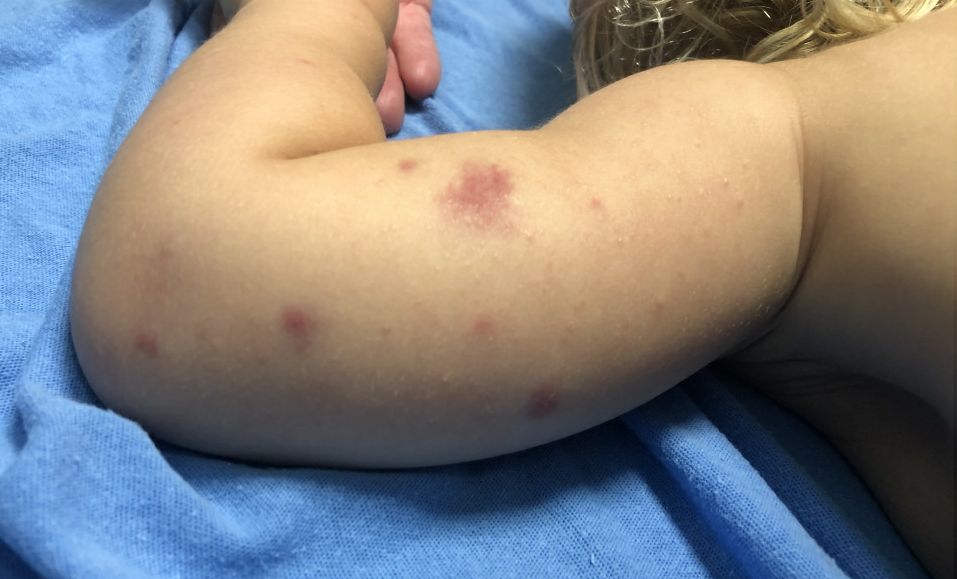
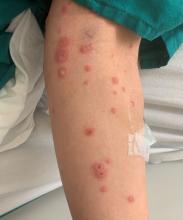
User login
The gap in cosmeceuticals education
Starting this month, I will be joining Dr. Leslie S. Baumann as a cocontributor to the Cosmeceutical Critique column, and since this is my first column, I would like to formally introduce myself. I am a cosmetic and general dermatologist in private practice in Miami and a longtime skin care enthusiast. My path toward becoming a dermatologist began when I was working in New York City, my hometown, as a scientific researcher, fulfilling my passion for scientific inquiry. After realizing that I most enjoyed applying discoveries made in the lab directly to patient care, I decided to pursue medical school at New York University before completing a dermatology residency at the University of Miami, serving as Chief Resident during my final year. Although I was born and raised in New York, staying in Miami was an obvious decision for me. In addition to the tropical weather and amazing lifestyle, the medical community in Miami supports adventure, creativity, and innovation, which are key aspects that drew me to the University of Miami and continue to drive my personal evolution in private practice.
I now practice at Baumann Cosmetic & Research Institute alongside my mentor, Dr. Baumann. I truly have my dream job – I get to talk skin care and do a wide array of cosmetics procedures, perform skin surgeries, and solve complex medical dermatology cases all in a day’s work. My career sits at the intersection of my passions for science, critical thinking, beauty, aesthetics, and most importantly, engaging with patients.
For my first column, I want to , and I will provide a simple framework to approach the design of skin care regimens and utilization of cosmeceuticals in practice.
The focus of a dermatology residency is on medical and surgical skills. We become experts in diagnosing and treating conditions ranging from life-threatening drug reactions like Stevens-Johnson Syndrome to complex diseases like dermatomyositis, utilizing medications and treatments ranging from cyclosporine and methotrexate to biologics and intravenous immunoglobulin, and performing advanced skin surgeries utilizing flaps and grafts to repair defects.
The discipline of cosmetic dermatology, let alone cosmeceuticals, accounts for a fraction of our didactic and hands-on training. I completed a top dermatology residency program that prepared me to treat any dermatologic condition; however, I honestly felt like I didn’t have a strong understanding of cosmeceuticals and skin care and how to integrate them with prescription therapies when I completed residency, which is a sentiment shared by residents across the country. I remember a study break while preparing for my final board exam when I went into a tailspin for an entire day trying to decode an ingredient list of a new “antiaging serum” and researching its mechanisms of action and the clinical data supporting the active ingredients in the serum, which included bakuchiol and a blend of peptides. As a dermatologist who likes to treat and provide recommendations based on scientific rationale and data to deliver the highest level of care, I admit that I felt insecure not being as knowledgeable about cosmeceuticals as I was about more complex dermatology treatments. As both a cosmetic and general dermatologist, discussing skin care and cosmeceuticals independent of or in conjunction with medical management occurs daily, and I recognized that becoming an expert in this area is essential to becoming a top, well-rounded dermatologist.
A gap in cosmeceutical education in dermatology residency
Multiple studies have established that the field of cosmetic dermatology comprises a fraction of dermatology residency training. In 2013, Kirby et al. published a survey of dermatology instructors and chief residents across the country and found that only 67% of responders reported having received formal lectures on cosmetic dermatology.1 In 2014, Bauer et al. published a survey of dermatology program directors assessing attitudes toward cosmetic dermatology and reported that only 38% of program directors believed that cosmetic dermatology should be a necessary aspect of residency training.2 A survey sent to dermatology residents published in 2012 found that among respondents, more than 58% of residency programs have an “encouraging or somewhat encouraging” attitude toward teaching cosmetic dermatology, yet 22% of programs had a “somewhat discouraging” or “discouraging” attitude.3 While these noted studies have focused on procedural aspects of cosmetic dermatology training, Feetham et al. surveyed dermatology residents and faculty to assess attitudes toward and training on skin care and cosmeceuticals specifically. Among resident respondents, most (74.5%) reported their education on skin care and cosmeceuticals has been “too little or nonexistent” during residency and 76.5% “agree or strongly agree” that it should be part of their education.4 In contrast, 60% of faculty reported resident education on skin care and cosmeceuticals is “just the right amount or too much” (P < .001).
In my personal experience as a resident, discussing skin care was emphasized when treating patients with eczema, contact dermatitis, acne, and hair disorders, but otherwise, the majority of skin care discussions relied on having a stock list of recommended cleansers, moisturizers, and sunscreens. In regards to cosmeceuticals for facial skin specifically, there were only a handful of instances in which alternative ingredients, such as vitamin C for hyperpigmentation, were discussed and specific brands were mentioned. Upon reflection, I wish I had more opportunity to see the clinical benefits of cosmeceuticals first hand, just like when I observe dupilumab clear patients with severe atopic dermatitis, rather than reading about it in textbooks and journals.
While one hypothesis for programs’ limited attention given to cosmetic training may be that it detracts from medical training, the survey by Bauer et al. found that residents did not feel less prepared (94.9%) or less interested (97.4%) in medical dermatology as a result of their cosmetic training.2 In addition, providers in an academic dermatology residency may limit discussions of skin care because of the high patient volume and because extensive skin care discussions will not impact insurance billings. Academic dermatology programs often service patients with more financial constraints, which further limits OTC cosmeceutical discussions. In my residency experience, I had the opportunity to regularly treat more severe and rare dermatologic cases than those I encounter in private practice; therefore, I spent more time focusing on systemic therapies, with fewer opportunities to dedicate time to cosmeceuticals.
Why skin care and cosmeceuticals should be an essential aspect of residency training
Discussing skin care and cosmeceuticals is a valuable aspect of medical and general dermatology, not just aesthetic dermatology. When treating general dermatologic conditions, guidance on proper skin care can improve both adherence and efficacy of medical treatments. For example, an acne study by de Lucas et al. demonstrated that adherence to adjuvant treatment of acne (such as the use of moisturizers) was associated not only with a 2.4-fold increase in the probability of adherence to pharmacological treatment, but also with a significant reduction in acne severity.5 Aside from skin care, cosmeceuticals themselves have efficacy in treating general dermatologic conditions. In the treatment of acne, topical niacinamide, a popular cosmeceutical ingredient, has been shown to have sebosuppressive and anti-inflammatory effects, addressing key aspects of acne pathogenesis.6 A double-blind study by Draelos et al. reported topical 2% niacinamide was effective in reducing the rate of sebum excretion in 50 Japanese patients over 4 weeks.6 In several double-blind studies that have compared twice daily application of 4% nicotinamide gel with the same application of 1% clindamycin gel in moderate inflammatory acne over 8 weeks, nicotinamide gel reduced the number of inflammatory papules and acne lesions to a level comparable with clindamycin gel.6 These studies support the use of niacinamide cosmeceutical products as an adjunctive treatment for acne.
With increased clinical data supporting cosmeceuticals, it can be expected that some cosmeceuticals will substitute traditional prescription medications in the dermatologists’ arsenal. For example, hydroquinone – both prescription strength and OTC 2% – is a workhorse in treating melasma; however, there is increasing interest in hydroquinone-free treatments, especially since OTC cosmeceuticals containing 2% hydroquinone were banned in 2020 because of safety concerns. Dermatologists will therefore need to provide guidance about hydroquinone alternatives for skin lightening, including soy, licorice extracts, kojic acid, arbutin, niacinamide, N-acetylglucosamine, and vitamin C, among others.7 Utilizing knowledge of a cosmeceutical’s mechanisms of action and clinical data, the dermatologist is in the best position to guide patients toward optimal ingredients and dispel cosmeceutical myths. Given that cosmeceuticals are not regulated by the Food and Drug Administration, it is even more important that the dermatologist serves as an authority on cosmeceuticals.
How to become a master skin care and cosmeceutical prescriber
A common pitfall I have observed among practitioners less experienced with aesthetic-focused skin care and cosmeceuticals is adapting a one-size-fits-all approach. In the one-size-fits-all approach, every patient concerned about aging gets the same vitamin C serum and retinoid, and every patient with hyperpigmentation gets the same hydroquinone prescription, for example. This approach, however, does not take into account unique differences in patients’ skin. Below
is the basic skin care framework that I follow, taught to me by Dr. Baumann. It utilizes an individualized approach based on the patient’s skin qualities to achieve optimal results.
Determine the patient’s skin type (dry vs. oily; sensitive vs. not sensitive; pigmentation issues vs. no hyperpigmentation; wrinkled and mature vs. nonwrinkled) and identify concerns (e.g., dark spots, redness, acne, dehydration).
Separate products into categories of cleansers, eye creams, moisturizers, sun protection, and treatments. Treatments refers to any additional products in a skin care regimen intended to ameliorate a particular condition (e.g., vitamin C for hyperpigmentation, retinoids for fine lines).
Choose products for each category in step 2 (cleansers, eye creams, moisturizers, sun protection, treatments) that are complementary to the patient’s skin type (determined in step 1) and aid the patient in meeting their particular skin goals. For example, a salicylic acid cleanser would be beneficial for a patient with oily skin and acne, but this same cleanser may be too drying and irritating for an acne patient with dry skin.
Ensure that chosen ingredients and products work together harmoniously. For example, while the acne patient may benefit from a salicylic acid cleanser and retinoid cream, using them in succession initially may be overly drying for some patients.
Spend the time to make sure patients understand the appropriate order of application and recognize when efficacy of a product is impacted by another product in the regimen. For example, a low pH cleanser can increase penetration of an ascorbic acid product that follows it in the regimen.
After establishing a basic skin care framework, the next step for beginners is learning about ingredients and their mechanisms of action and familiarizing themselves with scientific and clinical studies. Until cosmeceuticals become an integral part of the training curriculum, dermatologists can gain knowledge independently by reading literature and studies on cosmeceutical active ingredients and experimenting with consumer products. I look forward to regularly contributing to this column to further our awareness and understanding of the mechanisms of and data supporting cosmeceuticals so that we can better guide our patients.
Please feel free to email me at [email protected] or message me on Instagram @DrChloeGoldman with ideas that you would like me to address in this column.
Dr. Goldman is a dermatologist in private practice in Miami, and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a new general dermatology practice. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other relevant disclosures.
References
1. Kirby JS et al. J Am Acad Dermatol. 2013;68(2):e23-8.
2. Bauer et al. JAMA Dermatol. 2014;150(2):125-9.
3. Group A et al. Dermatol Surg. 2012;38(12):1975-80.
4. Feetham HJ et al. J Cosmet Dermatol. 2018;17(2):220-6.
5. de Lucas R et al. BMC Dermatol. 2015;15:17.
6. Araviiskaia E and Dreno BJ. Eur Acad Dermatol Venereol. 2016;30(6):926-35.
7. Leyden JJ et al. J Eur Acad Dermatol Venereol. 2011;25(10):1140-5.
Starting this month, I will be joining Dr. Leslie S. Baumann as a cocontributor to the Cosmeceutical Critique column, and since this is my first column, I would like to formally introduce myself. I am a cosmetic and general dermatologist in private practice in Miami and a longtime skin care enthusiast. My path toward becoming a dermatologist began when I was working in New York City, my hometown, as a scientific researcher, fulfilling my passion for scientific inquiry. After realizing that I most enjoyed applying discoveries made in the lab directly to patient care, I decided to pursue medical school at New York University before completing a dermatology residency at the University of Miami, serving as Chief Resident during my final year. Although I was born and raised in New York, staying in Miami was an obvious decision for me. In addition to the tropical weather and amazing lifestyle, the medical community in Miami supports adventure, creativity, and innovation, which are key aspects that drew me to the University of Miami and continue to drive my personal evolution in private practice.
I now practice at Baumann Cosmetic & Research Institute alongside my mentor, Dr. Baumann. I truly have my dream job – I get to talk skin care and do a wide array of cosmetics procedures, perform skin surgeries, and solve complex medical dermatology cases all in a day’s work. My career sits at the intersection of my passions for science, critical thinking, beauty, aesthetics, and most importantly, engaging with patients.
For my first column, I want to , and I will provide a simple framework to approach the design of skin care regimens and utilization of cosmeceuticals in practice.
The focus of a dermatology residency is on medical and surgical skills. We become experts in diagnosing and treating conditions ranging from life-threatening drug reactions like Stevens-Johnson Syndrome to complex diseases like dermatomyositis, utilizing medications and treatments ranging from cyclosporine and methotrexate to biologics and intravenous immunoglobulin, and performing advanced skin surgeries utilizing flaps and grafts to repair defects.
The discipline of cosmetic dermatology, let alone cosmeceuticals, accounts for a fraction of our didactic and hands-on training. I completed a top dermatology residency program that prepared me to treat any dermatologic condition; however, I honestly felt like I didn’t have a strong understanding of cosmeceuticals and skin care and how to integrate them with prescription therapies when I completed residency, which is a sentiment shared by residents across the country. I remember a study break while preparing for my final board exam when I went into a tailspin for an entire day trying to decode an ingredient list of a new “antiaging serum” and researching its mechanisms of action and the clinical data supporting the active ingredients in the serum, which included bakuchiol and a blend of peptides. As a dermatologist who likes to treat and provide recommendations based on scientific rationale and data to deliver the highest level of care, I admit that I felt insecure not being as knowledgeable about cosmeceuticals as I was about more complex dermatology treatments. As both a cosmetic and general dermatologist, discussing skin care and cosmeceuticals independent of or in conjunction with medical management occurs daily, and I recognized that becoming an expert in this area is essential to becoming a top, well-rounded dermatologist.
A gap in cosmeceutical education in dermatology residency
Multiple studies have established that the field of cosmetic dermatology comprises a fraction of dermatology residency training. In 2013, Kirby et al. published a survey of dermatology instructors and chief residents across the country and found that only 67% of responders reported having received formal lectures on cosmetic dermatology.1 In 2014, Bauer et al. published a survey of dermatology program directors assessing attitudes toward cosmetic dermatology and reported that only 38% of program directors believed that cosmetic dermatology should be a necessary aspect of residency training.2 A survey sent to dermatology residents published in 2012 found that among respondents, more than 58% of residency programs have an “encouraging or somewhat encouraging” attitude toward teaching cosmetic dermatology, yet 22% of programs had a “somewhat discouraging” or “discouraging” attitude.3 While these noted studies have focused on procedural aspects of cosmetic dermatology training, Feetham et al. surveyed dermatology residents and faculty to assess attitudes toward and training on skin care and cosmeceuticals specifically. Among resident respondents, most (74.5%) reported their education on skin care and cosmeceuticals has been “too little or nonexistent” during residency and 76.5% “agree or strongly agree” that it should be part of their education.4 In contrast, 60% of faculty reported resident education on skin care and cosmeceuticals is “just the right amount or too much” (P < .001).
In my personal experience as a resident, discussing skin care was emphasized when treating patients with eczema, contact dermatitis, acne, and hair disorders, but otherwise, the majority of skin care discussions relied on having a stock list of recommended cleansers, moisturizers, and sunscreens. In regards to cosmeceuticals for facial skin specifically, there were only a handful of instances in which alternative ingredients, such as vitamin C for hyperpigmentation, were discussed and specific brands were mentioned. Upon reflection, I wish I had more opportunity to see the clinical benefits of cosmeceuticals first hand, just like when I observe dupilumab clear patients with severe atopic dermatitis, rather than reading about it in textbooks and journals.
While one hypothesis for programs’ limited attention given to cosmetic training may be that it detracts from medical training, the survey by Bauer et al. found that residents did not feel less prepared (94.9%) or less interested (97.4%) in medical dermatology as a result of their cosmetic training.2 In addition, providers in an academic dermatology residency may limit discussions of skin care because of the high patient volume and because extensive skin care discussions will not impact insurance billings. Academic dermatology programs often service patients with more financial constraints, which further limits OTC cosmeceutical discussions. In my residency experience, I had the opportunity to regularly treat more severe and rare dermatologic cases than those I encounter in private practice; therefore, I spent more time focusing on systemic therapies, with fewer opportunities to dedicate time to cosmeceuticals.
Why skin care and cosmeceuticals should be an essential aspect of residency training
Discussing skin care and cosmeceuticals is a valuable aspect of medical and general dermatology, not just aesthetic dermatology. When treating general dermatologic conditions, guidance on proper skin care can improve both adherence and efficacy of medical treatments. For example, an acne study by de Lucas et al. demonstrated that adherence to adjuvant treatment of acne (such as the use of moisturizers) was associated not only with a 2.4-fold increase in the probability of adherence to pharmacological treatment, but also with a significant reduction in acne severity.5 Aside from skin care, cosmeceuticals themselves have efficacy in treating general dermatologic conditions. In the treatment of acne, topical niacinamide, a popular cosmeceutical ingredient, has been shown to have sebosuppressive and anti-inflammatory effects, addressing key aspects of acne pathogenesis.6 A double-blind study by Draelos et al. reported topical 2% niacinamide was effective in reducing the rate of sebum excretion in 50 Japanese patients over 4 weeks.6 In several double-blind studies that have compared twice daily application of 4% nicotinamide gel with the same application of 1% clindamycin gel in moderate inflammatory acne over 8 weeks, nicotinamide gel reduced the number of inflammatory papules and acne lesions to a level comparable with clindamycin gel.6 These studies support the use of niacinamide cosmeceutical products as an adjunctive treatment for acne.
With increased clinical data supporting cosmeceuticals, it can be expected that some cosmeceuticals will substitute traditional prescription medications in the dermatologists’ arsenal. For example, hydroquinone – both prescription strength and OTC 2% – is a workhorse in treating melasma; however, there is increasing interest in hydroquinone-free treatments, especially since OTC cosmeceuticals containing 2% hydroquinone were banned in 2020 because of safety concerns. Dermatologists will therefore need to provide guidance about hydroquinone alternatives for skin lightening, including soy, licorice extracts, kojic acid, arbutin, niacinamide, N-acetylglucosamine, and vitamin C, among others.7 Utilizing knowledge of a cosmeceutical’s mechanisms of action and clinical data, the dermatologist is in the best position to guide patients toward optimal ingredients and dispel cosmeceutical myths. Given that cosmeceuticals are not regulated by the Food and Drug Administration, it is even more important that the dermatologist serves as an authority on cosmeceuticals.
How to become a master skin care and cosmeceutical prescriber
A common pitfall I have observed among practitioners less experienced with aesthetic-focused skin care and cosmeceuticals is adapting a one-size-fits-all approach. In the one-size-fits-all approach, every patient concerned about aging gets the same vitamin C serum and retinoid, and every patient with hyperpigmentation gets the same hydroquinone prescription, for example. This approach, however, does not take into account unique differences in patients’ skin. Below
is the basic skin care framework that I follow, taught to me by Dr. Baumann. It utilizes an individualized approach based on the patient’s skin qualities to achieve optimal results.
Determine the patient’s skin type (dry vs. oily; sensitive vs. not sensitive; pigmentation issues vs. no hyperpigmentation; wrinkled and mature vs. nonwrinkled) and identify concerns (e.g., dark spots, redness, acne, dehydration).
Separate products into categories of cleansers, eye creams, moisturizers, sun protection, and treatments. Treatments refers to any additional products in a skin care regimen intended to ameliorate a particular condition (e.g., vitamin C for hyperpigmentation, retinoids for fine lines).
Choose products for each category in step 2 (cleansers, eye creams, moisturizers, sun protection, treatments) that are complementary to the patient’s skin type (determined in step 1) and aid the patient in meeting their particular skin goals. For example, a salicylic acid cleanser would be beneficial for a patient with oily skin and acne, but this same cleanser may be too drying and irritating for an acne patient with dry skin.
Ensure that chosen ingredients and products work together harmoniously. For example, while the acne patient may benefit from a salicylic acid cleanser and retinoid cream, using them in succession initially may be overly drying for some patients.
Spend the time to make sure patients understand the appropriate order of application and recognize when efficacy of a product is impacted by another product in the regimen. For example, a low pH cleanser can increase penetration of an ascorbic acid product that follows it in the regimen.
After establishing a basic skin care framework, the next step for beginners is learning about ingredients and their mechanisms of action and familiarizing themselves with scientific and clinical studies. Until cosmeceuticals become an integral part of the training curriculum, dermatologists can gain knowledge independently by reading literature and studies on cosmeceutical active ingredients and experimenting with consumer products. I look forward to regularly contributing to this column to further our awareness and understanding of the mechanisms of and data supporting cosmeceuticals so that we can better guide our patients.
Please feel free to email me at [email protected] or message me on Instagram @DrChloeGoldman with ideas that you would like me to address in this column.
Dr. Goldman is a dermatologist in private practice in Miami, and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a new general dermatology practice. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other relevant disclosures.
References
1. Kirby JS et al. J Am Acad Dermatol. 2013;68(2):e23-8.
2. Bauer et al. JAMA Dermatol. 2014;150(2):125-9.
3. Group A et al. Dermatol Surg. 2012;38(12):1975-80.
4. Feetham HJ et al. J Cosmet Dermatol. 2018;17(2):220-6.
5. de Lucas R et al. BMC Dermatol. 2015;15:17.
6. Araviiskaia E and Dreno BJ. Eur Acad Dermatol Venereol. 2016;30(6):926-35.
7. Leyden JJ et al. J Eur Acad Dermatol Venereol. 2011;25(10):1140-5.
Starting this month, I will be joining Dr. Leslie S. Baumann as a cocontributor to the Cosmeceutical Critique column, and since this is my first column, I would like to formally introduce myself. I am a cosmetic and general dermatologist in private practice in Miami and a longtime skin care enthusiast. My path toward becoming a dermatologist began when I was working in New York City, my hometown, as a scientific researcher, fulfilling my passion for scientific inquiry. After realizing that I most enjoyed applying discoveries made in the lab directly to patient care, I decided to pursue medical school at New York University before completing a dermatology residency at the University of Miami, serving as Chief Resident during my final year. Although I was born and raised in New York, staying in Miami was an obvious decision for me. In addition to the tropical weather and amazing lifestyle, the medical community in Miami supports adventure, creativity, and innovation, which are key aspects that drew me to the University of Miami and continue to drive my personal evolution in private practice.
I now practice at Baumann Cosmetic & Research Institute alongside my mentor, Dr. Baumann. I truly have my dream job – I get to talk skin care and do a wide array of cosmetics procedures, perform skin surgeries, and solve complex medical dermatology cases all in a day’s work. My career sits at the intersection of my passions for science, critical thinking, beauty, aesthetics, and most importantly, engaging with patients.
For my first column, I want to , and I will provide a simple framework to approach the design of skin care regimens and utilization of cosmeceuticals in practice.
The focus of a dermatology residency is on medical and surgical skills. We become experts in diagnosing and treating conditions ranging from life-threatening drug reactions like Stevens-Johnson Syndrome to complex diseases like dermatomyositis, utilizing medications and treatments ranging from cyclosporine and methotrexate to biologics and intravenous immunoglobulin, and performing advanced skin surgeries utilizing flaps and grafts to repair defects.
The discipline of cosmetic dermatology, let alone cosmeceuticals, accounts for a fraction of our didactic and hands-on training. I completed a top dermatology residency program that prepared me to treat any dermatologic condition; however, I honestly felt like I didn’t have a strong understanding of cosmeceuticals and skin care and how to integrate them with prescription therapies when I completed residency, which is a sentiment shared by residents across the country. I remember a study break while preparing for my final board exam when I went into a tailspin for an entire day trying to decode an ingredient list of a new “antiaging serum” and researching its mechanisms of action and the clinical data supporting the active ingredients in the serum, which included bakuchiol and a blend of peptides. As a dermatologist who likes to treat and provide recommendations based on scientific rationale and data to deliver the highest level of care, I admit that I felt insecure not being as knowledgeable about cosmeceuticals as I was about more complex dermatology treatments. As both a cosmetic and general dermatologist, discussing skin care and cosmeceuticals independent of or in conjunction with medical management occurs daily, and I recognized that becoming an expert in this area is essential to becoming a top, well-rounded dermatologist.
A gap in cosmeceutical education in dermatology residency
Multiple studies have established that the field of cosmetic dermatology comprises a fraction of dermatology residency training. In 2013, Kirby et al. published a survey of dermatology instructors and chief residents across the country and found that only 67% of responders reported having received formal lectures on cosmetic dermatology.1 In 2014, Bauer et al. published a survey of dermatology program directors assessing attitudes toward cosmetic dermatology and reported that only 38% of program directors believed that cosmetic dermatology should be a necessary aspect of residency training.2 A survey sent to dermatology residents published in 2012 found that among respondents, more than 58% of residency programs have an “encouraging or somewhat encouraging” attitude toward teaching cosmetic dermatology, yet 22% of programs had a “somewhat discouraging” or “discouraging” attitude.3 While these noted studies have focused on procedural aspects of cosmetic dermatology training, Feetham et al. surveyed dermatology residents and faculty to assess attitudes toward and training on skin care and cosmeceuticals specifically. Among resident respondents, most (74.5%) reported their education on skin care and cosmeceuticals has been “too little or nonexistent” during residency and 76.5% “agree or strongly agree” that it should be part of their education.4 In contrast, 60% of faculty reported resident education on skin care and cosmeceuticals is “just the right amount or too much” (P < .001).
In my personal experience as a resident, discussing skin care was emphasized when treating patients with eczema, contact dermatitis, acne, and hair disorders, but otherwise, the majority of skin care discussions relied on having a stock list of recommended cleansers, moisturizers, and sunscreens. In regards to cosmeceuticals for facial skin specifically, there were only a handful of instances in which alternative ingredients, such as vitamin C for hyperpigmentation, were discussed and specific brands were mentioned. Upon reflection, I wish I had more opportunity to see the clinical benefits of cosmeceuticals first hand, just like when I observe dupilumab clear patients with severe atopic dermatitis, rather than reading about it in textbooks and journals.
While one hypothesis for programs’ limited attention given to cosmetic training may be that it detracts from medical training, the survey by Bauer et al. found that residents did not feel less prepared (94.9%) or less interested (97.4%) in medical dermatology as a result of their cosmetic training.2 In addition, providers in an academic dermatology residency may limit discussions of skin care because of the high patient volume and because extensive skin care discussions will not impact insurance billings. Academic dermatology programs often service patients with more financial constraints, which further limits OTC cosmeceutical discussions. In my residency experience, I had the opportunity to regularly treat more severe and rare dermatologic cases than those I encounter in private practice; therefore, I spent more time focusing on systemic therapies, with fewer opportunities to dedicate time to cosmeceuticals.
Why skin care and cosmeceuticals should be an essential aspect of residency training
Discussing skin care and cosmeceuticals is a valuable aspect of medical and general dermatology, not just aesthetic dermatology. When treating general dermatologic conditions, guidance on proper skin care can improve both adherence and efficacy of medical treatments. For example, an acne study by de Lucas et al. demonstrated that adherence to adjuvant treatment of acne (such as the use of moisturizers) was associated not only with a 2.4-fold increase in the probability of adherence to pharmacological treatment, but also with a significant reduction in acne severity.5 Aside from skin care, cosmeceuticals themselves have efficacy in treating general dermatologic conditions. In the treatment of acne, topical niacinamide, a popular cosmeceutical ingredient, has been shown to have sebosuppressive and anti-inflammatory effects, addressing key aspects of acne pathogenesis.6 A double-blind study by Draelos et al. reported topical 2% niacinamide was effective in reducing the rate of sebum excretion in 50 Japanese patients over 4 weeks.6 In several double-blind studies that have compared twice daily application of 4% nicotinamide gel with the same application of 1% clindamycin gel in moderate inflammatory acne over 8 weeks, nicotinamide gel reduced the number of inflammatory papules and acne lesions to a level comparable with clindamycin gel.6 These studies support the use of niacinamide cosmeceutical products as an adjunctive treatment for acne.
With increased clinical data supporting cosmeceuticals, it can be expected that some cosmeceuticals will substitute traditional prescription medications in the dermatologists’ arsenal. For example, hydroquinone – both prescription strength and OTC 2% – is a workhorse in treating melasma; however, there is increasing interest in hydroquinone-free treatments, especially since OTC cosmeceuticals containing 2% hydroquinone were banned in 2020 because of safety concerns. Dermatologists will therefore need to provide guidance about hydroquinone alternatives for skin lightening, including soy, licorice extracts, kojic acid, arbutin, niacinamide, N-acetylglucosamine, and vitamin C, among others.7 Utilizing knowledge of a cosmeceutical’s mechanisms of action and clinical data, the dermatologist is in the best position to guide patients toward optimal ingredients and dispel cosmeceutical myths. Given that cosmeceuticals are not regulated by the Food and Drug Administration, it is even more important that the dermatologist serves as an authority on cosmeceuticals.
How to become a master skin care and cosmeceutical prescriber
A common pitfall I have observed among practitioners less experienced with aesthetic-focused skin care and cosmeceuticals is adapting a one-size-fits-all approach. In the one-size-fits-all approach, every patient concerned about aging gets the same vitamin C serum and retinoid, and every patient with hyperpigmentation gets the same hydroquinone prescription, for example. This approach, however, does not take into account unique differences in patients’ skin. Below
is the basic skin care framework that I follow, taught to me by Dr. Baumann. It utilizes an individualized approach based on the patient’s skin qualities to achieve optimal results.
Determine the patient’s skin type (dry vs. oily; sensitive vs. not sensitive; pigmentation issues vs. no hyperpigmentation; wrinkled and mature vs. nonwrinkled) and identify concerns (e.g., dark spots, redness, acne, dehydration).
Separate products into categories of cleansers, eye creams, moisturizers, sun protection, and treatments. Treatments refers to any additional products in a skin care regimen intended to ameliorate a particular condition (e.g., vitamin C for hyperpigmentation, retinoids for fine lines).
Choose products for each category in step 2 (cleansers, eye creams, moisturizers, sun protection, treatments) that are complementary to the patient’s skin type (determined in step 1) and aid the patient in meeting their particular skin goals. For example, a salicylic acid cleanser would be beneficial for a patient with oily skin and acne, but this same cleanser may be too drying and irritating for an acne patient with dry skin.
Ensure that chosen ingredients and products work together harmoniously. For example, while the acne patient may benefit from a salicylic acid cleanser and retinoid cream, using them in succession initially may be overly drying for some patients.
Spend the time to make sure patients understand the appropriate order of application and recognize when efficacy of a product is impacted by another product in the regimen. For example, a low pH cleanser can increase penetration of an ascorbic acid product that follows it in the regimen.
After establishing a basic skin care framework, the next step for beginners is learning about ingredients and their mechanisms of action and familiarizing themselves with scientific and clinical studies. Until cosmeceuticals become an integral part of the training curriculum, dermatologists can gain knowledge independently by reading literature and studies on cosmeceutical active ingredients and experimenting with consumer products. I look forward to regularly contributing to this column to further our awareness and understanding of the mechanisms of and data supporting cosmeceuticals so that we can better guide our patients.
Please feel free to email me at [email protected] or message me on Instagram @DrChloeGoldman with ideas that you would like me to address in this column.
Dr. Goldman is a dermatologist in private practice in Miami, and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a new general dermatology practice. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other relevant disclosures.
References
1. Kirby JS et al. J Am Acad Dermatol. 2013;68(2):e23-8.
2. Bauer et al. JAMA Dermatol. 2014;150(2):125-9.
3. Group A et al. Dermatol Surg. 2012;38(12):1975-80.
4. Feetham HJ et al. J Cosmet Dermatol. 2018;17(2):220-6.
5. de Lucas R et al. BMC Dermatol. 2015;15:17.
6. Araviiskaia E and Dreno BJ. Eur Acad Dermatol Venereol. 2016;30(6):926-35.
7. Leyden JJ et al. J Eur Acad Dermatol Venereol. 2011;25(10):1140-5.
Obstetrical care for gender diverse patients: A summary from the SMFM annual meeting
The purpose of this commentary is to provide a brief summary of discussions centering around reproductive health experiences and obstetrical care for gender-diverse patients from the recent Society of Maternal & Fetal Medicine meeting. Two presentations featured patient perspectives combined with physician lectures to provide a comprehensive outlook on unique reproductive care needs for this growing population.
One of the speakers, Trystan Reese, is a transgender activist, educator, and transgender male who chose to carry his own pregnancy and subsequently delivered his son in 2017. During the summit, he described many barriers that he faced during his pregnancy and offered providers suggestions on how to improve the care for members of the gender-diverse community seeking to start a family.
We often think of conception and pregnancy as experiences unique to one gender. This is simply not the case. In discussing preconceptual care and pregnancy, it is paramount for providers to make the distinction between gender identity and natal sex. Gender identity is an internal sense of self in relation to natal sex. Depending on this intrinsic feeling, people may identify as cisgender, transgender, or as a gender outside of the standard binary. Natal sex describes biologic characteristics such as chromosomal makeup, reproductive anatomy, and secondary sexual changes. In keeping these distinctions in mind, pregnancy is therefore exclusive to a person’s natal sex, not gender identity. One of the biggest challenges in caring for transgender patients who desire pregnancy, is the psychological distress related to the gendered notions surrounding this experience.1
There are many ways in which patients encounter unintentional marginalization within the medical system. For example, many electronic medical record systems don’t allow for pronouns or give error messages if the patient’s gender identity is different from their sex assigned at birth. Patients who attend prenatal appointments or birth classes are given documents that center around cisgender women and heterosexual relationships. The labor and delivery wards themselves typically include language such as “maternity,” and birth certificates have distinct “mother” and “father” denotations.1 Insurance coverage for prenatal care and delivery can be problematic if a patient who is assigned female at birth has changed their gender marker to “male” on their insurance card.
Many of these roadblocks can be ameliorated by utilizing more inclusive terminology. Terms such as “maternal” can be replaced with “pregnant patients, parent, or patients giving birth.” Names of maternity wards can be altered to perinatal units, which is more inclusive and more descriptive of the wide variety of patients that may experience childbirth and parenthood.1 Introducing “you-centered” language can also be helpful. Instead of saying “women may find ...” providers can try saying “patients may find ...” or “individuals may find.”1
Most of the medical and obstetrical care of gender-diverse patients is routine. Prenatal labs, aneuploidy screening, ultrasounds, and fetal surveillance do not differ between transgender and cisgender patients. However, the experience of pregnancy itself can significantly heighten feelings of dysphoria as it inherently leads to patients confronting aspects of their biological sex.2 Because of the teratogenic nature of testosterone, patients are required to stop taking testosterone prior to conception and for the duration of pregnancy. This can also heighten dysphoria and lead to increased rates of anxiety and depression.3
Many transgender patients can safely achieve a normal vaginal birth.4 A small survey of 41 people demonstrated that more transgender men who had taken testosterone were delivered by cesarean section (36% vs. 19%).3 Staff training is an important aspect of caring for a transgender patient in labor to ensure that all members of the labor unit are cognizant of appropriate name and pronoun usage. Another interesting, although unsurprising, fact is that many transgender gestational parents chose a community-based (out-of-hospital) birth according to a 2014 study.1 This is predominantly because of the discrimination patients face when delivering within a hospital setting.
Postpartum depression screening should be conducted prior to patients leaving the hospital and individualized during postpartum appointments. Reinitiation of testosterone can occur 4-6 weeks after delivery.1
While pregnancy can pose some unique challenges to gender-diverse individuals, these intricacies are not insurmountable. The result of pregnancy, regardless of one’s gender identity, is the same – parenthood. One patient’s description of his experience was particularly poignant: “Pregnancy and childbirth were very male experiences for me. When I birthed my children, I was born into fatherhood.”1 It is up to all providers to modify clinical settings, as well as our patient interactions and use of language, if we are to provide inclusion in obstetrics.1,5
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Brandt JS et al. “Understanding intersections: Care for transgender and gender diverse patient populations.” SMFM 2022 annual meeting. 2022 Feb 2.
2. Hoffkling A et al. BMC Pregnancy Childbirth. 2017 Nov 8;17(Suppl 2):332.
3. Light AD et al. Obstet Gynecol. 2014;124:1120-7.
4. Moseson H et al. Int J Transgend Health. 2021 Nov 17;22(1-2):30-41.
5. Brandt JS et al. Obstetrical care for trans*person, in “Trans*gynecology: Managing transgender patients in obstetrics and gynecology practice.” (Cambridge, England: Cambridge University Press, 2022).
The purpose of this commentary is to provide a brief summary of discussions centering around reproductive health experiences and obstetrical care for gender-diverse patients from the recent Society of Maternal & Fetal Medicine meeting. Two presentations featured patient perspectives combined with physician lectures to provide a comprehensive outlook on unique reproductive care needs for this growing population.
One of the speakers, Trystan Reese, is a transgender activist, educator, and transgender male who chose to carry his own pregnancy and subsequently delivered his son in 2017. During the summit, he described many barriers that he faced during his pregnancy and offered providers suggestions on how to improve the care for members of the gender-diverse community seeking to start a family.
We often think of conception and pregnancy as experiences unique to one gender. This is simply not the case. In discussing preconceptual care and pregnancy, it is paramount for providers to make the distinction between gender identity and natal sex. Gender identity is an internal sense of self in relation to natal sex. Depending on this intrinsic feeling, people may identify as cisgender, transgender, or as a gender outside of the standard binary. Natal sex describes biologic characteristics such as chromosomal makeup, reproductive anatomy, and secondary sexual changes. In keeping these distinctions in mind, pregnancy is therefore exclusive to a person’s natal sex, not gender identity. One of the biggest challenges in caring for transgender patients who desire pregnancy, is the psychological distress related to the gendered notions surrounding this experience.1
There are many ways in which patients encounter unintentional marginalization within the medical system. For example, many electronic medical record systems don’t allow for pronouns or give error messages if the patient’s gender identity is different from their sex assigned at birth. Patients who attend prenatal appointments or birth classes are given documents that center around cisgender women and heterosexual relationships. The labor and delivery wards themselves typically include language such as “maternity,” and birth certificates have distinct “mother” and “father” denotations.1 Insurance coverage for prenatal care and delivery can be problematic if a patient who is assigned female at birth has changed their gender marker to “male” on their insurance card.
Many of these roadblocks can be ameliorated by utilizing more inclusive terminology. Terms such as “maternal” can be replaced with “pregnant patients, parent, or patients giving birth.” Names of maternity wards can be altered to perinatal units, which is more inclusive and more descriptive of the wide variety of patients that may experience childbirth and parenthood.1 Introducing “you-centered” language can also be helpful. Instead of saying “women may find ...” providers can try saying “patients may find ...” or “individuals may find.”1
Most of the medical and obstetrical care of gender-diverse patients is routine. Prenatal labs, aneuploidy screening, ultrasounds, and fetal surveillance do not differ between transgender and cisgender patients. However, the experience of pregnancy itself can significantly heighten feelings of dysphoria as it inherently leads to patients confronting aspects of their biological sex.2 Because of the teratogenic nature of testosterone, patients are required to stop taking testosterone prior to conception and for the duration of pregnancy. This can also heighten dysphoria and lead to increased rates of anxiety and depression.3
Many transgender patients can safely achieve a normal vaginal birth.4 A small survey of 41 people demonstrated that more transgender men who had taken testosterone were delivered by cesarean section (36% vs. 19%).3 Staff training is an important aspect of caring for a transgender patient in labor to ensure that all members of the labor unit are cognizant of appropriate name and pronoun usage. Another interesting, although unsurprising, fact is that many transgender gestational parents chose a community-based (out-of-hospital) birth according to a 2014 study.1 This is predominantly because of the discrimination patients face when delivering within a hospital setting.
Postpartum depression screening should be conducted prior to patients leaving the hospital and individualized during postpartum appointments. Reinitiation of testosterone can occur 4-6 weeks after delivery.1
While pregnancy can pose some unique challenges to gender-diverse individuals, these intricacies are not insurmountable. The result of pregnancy, regardless of one’s gender identity, is the same – parenthood. One patient’s description of his experience was particularly poignant: “Pregnancy and childbirth were very male experiences for me. When I birthed my children, I was born into fatherhood.”1 It is up to all providers to modify clinical settings, as well as our patient interactions and use of language, if we are to provide inclusion in obstetrics.1,5
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Brandt JS et al. “Understanding intersections: Care for transgender and gender diverse patient populations.” SMFM 2022 annual meeting. 2022 Feb 2.
2. Hoffkling A et al. BMC Pregnancy Childbirth. 2017 Nov 8;17(Suppl 2):332.
3. Light AD et al. Obstet Gynecol. 2014;124:1120-7.
4. Moseson H et al. Int J Transgend Health. 2021 Nov 17;22(1-2):30-41.
5. Brandt JS et al. Obstetrical care for trans*person, in “Trans*gynecology: Managing transgender patients in obstetrics and gynecology practice.” (Cambridge, England: Cambridge University Press, 2022).
The purpose of this commentary is to provide a brief summary of discussions centering around reproductive health experiences and obstetrical care for gender-diverse patients from the recent Society of Maternal & Fetal Medicine meeting. Two presentations featured patient perspectives combined with physician lectures to provide a comprehensive outlook on unique reproductive care needs for this growing population.
One of the speakers, Trystan Reese, is a transgender activist, educator, and transgender male who chose to carry his own pregnancy and subsequently delivered his son in 2017. During the summit, he described many barriers that he faced during his pregnancy and offered providers suggestions on how to improve the care for members of the gender-diverse community seeking to start a family.
We often think of conception and pregnancy as experiences unique to one gender. This is simply not the case. In discussing preconceptual care and pregnancy, it is paramount for providers to make the distinction between gender identity and natal sex. Gender identity is an internal sense of self in relation to natal sex. Depending on this intrinsic feeling, people may identify as cisgender, transgender, or as a gender outside of the standard binary. Natal sex describes biologic characteristics such as chromosomal makeup, reproductive anatomy, and secondary sexual changes. In keeping these distinctions in mind, pregnancy is therefore exclusive to a person’s natal sex, not gender identity. One of the biggest challenges in caring for transgender patients who desire pregnancy, is the psychological distress related to the gendered notions surrounding this experience.1
There are many ways in which patients encounter unintentional marginalization within the medical system. For example, many electronic medical record systems don’t allow for pronouns or give error messages if the patient’s gender identity is different from their sex assigned at birth. Patients who attend prenatal appointments or birth classes are given documents that center around cisgender women and heterosexual relationships. The labor and delivery wards themselves typically include language such as “maternity,” and birth certificates have distinct “mother” and “father” denotations.1 Insurance coverage for prenatal care and delivery can be problematic if a patient who is assigned female at birth has changed their gender marker to “male” on their insurance card.
Many of these roadblocks can be ameliorated by utilizing more inclusive terminology. Terms such as “maternal” can be replaced with “pregnant patients, parent, or patients giving birth.” Names of maternity wards can be altered to perinatal units, which is more inclusive and more descriptive of the wide variety of patients that may experience childbirth and parenthood.1 Introducing “you-centered” language can also be helpful. Instead of saying “women may find ...” providers can try saying “patients may find ...” or “individuals may find.”1
Most of the medical and obstetrical care of gender-diverse patients is routine. Prenatal labs, aneuploidy screening, ultrasounds, and fetal surveillance do not differ between transgender and cisgender patients. However, the experience of pregnancy itself can significantly heighten feelings of dysphoria as it inherently leads to patients confronting aspects of their biological sex.2 Because of the teratogenic nature of testosterone, patients are required to stop taking testosterone prior to conception and for the duration of pregnancy. This can also heighten dysphoria and lead to increased rates of anxiety and depression.3
Many transgender patients can safely achieve a normal vaginal birth.4 A small survey of 41 people demonstrated that more transgender men who had taken testosterone were delivered by cesarean section (36% vs. 19%).3 Staff training is an important aspect of caring for a transgender patient in labor to ensure that all members of the labor unit are cognizant of appropriate name and pronoun usage. Another interesting, although unsurprising, fact is that many transgender gestational parents chose a community-based (out-of-hospital) birth according to a 2014 study.1 This is predominantly because of the discrimination patients face when delivering within a hospital setting.
Postpartum depression screening should be conducted prior to patients leaving the hospital and individualized during postpartum appointments. Reinitiation of testosterone can occur 4-6 weeks after delivery.1
While pregnancy can pose some unique challenges to gender-diverse individuals, these intricacies are not insurmountable. The result of pregnancy, regardless of one’s gender identity, is the same – parenthood. One patient’s description of his experience was particularly poignant: “Pregnancy and childbirth were very male experiences for me. When I birthed my children, I was born into fatherhood.”1 It is up to all providers to modify clinical settings, as well as our patient interactions and use of language, if we are to provide inclusion in obstetrics.1,5
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Brandt JS et al. “Understanding intersections: Care for transgender and gender diverse patient populations.” SMFM 2022 annual meeting. 2022 Feb 2.
2. Hoffkling A et al. BMC Pregnancy Childbirth. 2017 Nov 8;17(Suppl 2):332.
3. Light AD et al. Obstet Gynecol. 2014;124:1120-7.
4. Moseson H et al. Int J Transgend Health. 2021 Nov 17;22(1-2):30-41.
5. Brandt JS et al. Obstetrical care for trans*person, in “Trans*gynecology: Managing transgender patients in obstetrics and gynecology practice.” (Cambridge, England: Cambridge University Press, 2022).
Why is vitamin D hype so impervious to evidence?
The vitamin D story exudes teaching points: It offers a master class in critical appraisal, connecting the concepts of biologic plausibility, flawed surrogate markers, confounded observational studies, and slews of randomized controlled trials (RCTs) showing no benefits on health outcomes.
Yet despite the utter lack of benefit seen in trials, the hype continues. And the pandemic has only enhanced this hype as an onslaught of papers have reported the association of low vitamin D levels and COVID-19 disease.
My questions are simple: Why doesn’t the evidence persuade people? How many nonsignificant trials do we need before researchers stop studying vitamin D, doctors stop (routinely) measuring levels, and patients stop wasting money on the unhelpful supplement? What are the implications for this lack of persuasion?
Before exploring these questions, I want to set out that symptomatic vitamin deficiencies of any sort ought to be corrected.
Biologic plausibility and the pull of observational studies
It has long been known that vitamin D is crucial for bone health and that it can be produced in the skin with sun exposure. In the last decade, however, experts note that nearly every tissue and cell in our body has a vitamin D receptor. It then follows that if this many cells in the body can activate vitamin D, it must be vital for cardiovascular health, immune function, cancer prevention: basically, everything health related.
Oodles of observational studies have found that low serum levels of vitamin D correlate with higher mortality from all causes, cancer, cardiovascular disease, and now even COVID-19. Yet no matter the amount of statistical adjustment in these studies, we cannot know whether these associations are due to true causality.
The major issue is confounding: That is, people with low vitamin D levels have other conditions or diseases that lead to higher rates of ill health. Consider a patient with obesity, arthritis, and cognitive decline; this person is unlikely to do much exercise in the sun and may have low vitamin D levels. The low vitamin D level is simply a marker of overall poor health.
The randomized controlled trials tell a clear story
There are hundreds of vitamin D RCTs. The results simplify into one sentence: Vitamin D supplements do not improve health outcomes.
Here is a short summary of some recent studies.
VITAL, a massive (N > 25,000) RCT with 5 years of follow-up, compared vitamin D supplements to placebo and found no differences in the primary endpoints of cancer or cardiac events. Rates of death from any cause were nearly identical. Crucially, in subgroup analyses, the effects did not vary according to vitamin D levels at baseline.
The D-Health investigators randomly assigned more than 21,000 adults to vitamin D or placebo and after 5.7 years of follow-up reported no differences in the primary endpoint of overall mortality. There also were no differences in cardiovascular disease mortality.
Then you have the Mendelian randomized studies, which some have called nature’s RCT. These studies take advantage of the fact that some people are born with gene variations that predispose to low vitamin D levels. More than 60 Mendelian randomization studies have evaluated the consequences of lifelong genetically lowered vitamin D levels on various outcomes; most of these have found null effects.
Then there are the meta-analyses and systematic reviews. I loved the conclusion of this review of systematic reviews from the BMJ (emphasis mine):
“Despite a few hundred systematic reviews and meta-analyses, highly convincing evidence of a clear role of vitamin D does not exist for any outcome, but associations with a selection of outcomes are probable.”
The failure to persuade
My original plan was to emphasize the power of the RCT. Despite strong associations of low vitamin D levels with poor outcomes, the trials show no benefit to treatment. This strongly suggests (or nearly proves) that low vitamin D levels are akin to premature ventricular complexes after myocardial infarction: a marker for risk but not a target for therapy.
But I now see the more important issue as why scientists, funders, clinicians, and patients are not persuaded by clear evidence. Every day in clinic I see patients on vitamin D supplements; the journals keep publishing vitamin D studies. The proponents of vitamin D remain positive. And lately there is outsized attention and hope that vitamin D will mitigate SARS-CoV2 infection – based only on observational data.
You might argue against this point by saying vitamin D is natural and relatively innocuous, so who cares?
I offer three rebuttals to that point: Opportunity costs, distraction, and the insidious danger of poor critical appraisal skills. If you are burning money on vitamin D research, there is less available to study other important issues. If a patient is distracted by low vitamin D levels, she may pay less attention to her high body mass index or hypertension. And on the matter of critical appraisal, trust in medicine requires clinicians to be competent in critical appraisal. And these days, what could be more important than trust in medical professionals?
One major reason for the failure of persuasion of evidence is spin – or language that distracts from the primary endpoint. Here are two (of many) examples:
A meta-analysis of 50 vitamin D trials set out to study mortality. The authors found no significant difference in that primary endpoint. But the second sentence in their conclusion was that vitamin D supplements reduced the risk for cancer deaths by 15%. That’s a secondary endpoint in a study with nonsignificance in the primary endpoint. That is spin. This meta-analysis was completed before the Australian D-Health trial found that cancer deaths were 15% higher in the vitamin D arm, a difference that did not reach statistical significance.
The following example is worse: The authors of the VITAL trial, which found that vitamin D supplements had no effect on the primary endpoint of invasive cancer or cardiovascular disease, published a secondary analysis of the trial looking at a different endpoint: A composite incidence of metastatic and fatal invasive total cancer. They reported a 0.4% lower rate for the vitamin D group, a difference that barely made statistical significance at a P value of .04.
But everyone knows the dangers of reanalyzing data with a new endpoint after you have seen the data. What’s more, even if this were a reasonable post hoc analysis, the results are neither clinically meaningful nor statistically robust. Yet the fatally flawed paper has been viewed 60,000 times and picked up by 48 news outlets.
Another way to distract from nonsignificant primary outcomes is to nitpick the trials. The vitamin D dose wasn’t high enough, for instance. This might persuade me if there were one or two vitamin D trials, but there are hundreds of trials and meta-analyses, and their results are consistently null.
Conclusion: No, it is not hopeless
A nihilist would argue that fighting spin is futile. They would say you can’t fight incentives and business models. The incentive structure to publish is strong, and the journals and media know vitamin D studies garner attention – which is their currency.
I am not a nihilist and believe strongly that we must continue to teach critical appraisal and numerical literacy.
In fact, I would speculate that decades of poor critical appraisal by the medical profession have fostered outsized hope and created erroneous norms.
Imagine a counter-factual world in which clinicians have taught society that the human body is unlike an engine that can be repaired by fixing one part (i.e., the vitamin D level), that magic bullets (insulin) are rare, that most treatments fail, or that you can’t rely on association studies to prove efficacy.
In this world, people would be immune from spin and hype.
The norm would be that pills, supplements, and procedures are not what delivers good health. What delivers health is an amalgam of good luck, healthy habits, and lots of time spent outside playing in the sun.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky., and is a writer and podcaster for Medscape. He espouses a conservative approach to medical practice. He participates in clinical research and writes often about the state of medical evidence. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
The vitamin D story exudes teaching points: It offers a master class in critical appraisal, connecting the concepts of biologic plausibility, flawed surrogate markers, confounded observational studies, and slews of randomized controlled trials (RCTs) showing no benefits on health outcomes.
Yet despite the utter lack of benefit seen in trials, the hype continues. And the pandemic has only enhanced this hype as an onslaught of papers have reported the association of low vitamin D levels and COVID-19 disease.
My questions are simple: Why doesn’t the evidence persuade people? How many nonsignificant trials do we need before researchers stop studying vitamin D, doctors stop (routinely) measuring levels, and patients stop wasting money on the unhelpful supplement? What are the implications for this lack of persuasion?
Before exploring these questions, I want to set out that symptomatic vitamin deficiencies of any sort ought to be corrected.
Biologic plausibility and the pull of observational studies
It has long been known that vitamin D is crucial for bone health and that it can be produced in the skin with sun exposure. In the last decade, however, experts note that nearly every tissue and cell in our body has a vitamin D receptor. It then follows that if this many cells in the body can activate vitamin D, it must be vital for cardiovascular health, immune function, cancer prevention: basically, everything health related.
Oodles of observational studies have found that low serum levels of vitamin D correlate with higher mortality from all causes, cancer, cardiovascular disease, and now even COVID-19. Yet no matter the amount of statistical adjustment in these studies, we cannot know whether these associations are due to true causality.
The major issue is confounding: That is, people with low vitamin D levels have other conditions or diseases that lead to higher rates of ill health. Consider a patient with obesity, arthritis, and cognitive decline; this person is unlikely to do much exercise in the sun and may have low vitamin D levels. The low vitamin D level is simply a marker of overall poor health.
The randomized controlled trials tell a clear story
There are hundreds of vitamin D RCTs. The results simplify into one sentence: Vitamin D supplements do not improve health outcomes.
Here is a short summary of some recent studies.
VITAL, a massive (N > 25,000) RCT with 5 years of follow-up, compared vitamin D supplements to placebo and found no differences in the primary endpoints of cancer or cardiac events. Rates of death from any cause were nearly identical. Crucially, in subgroup analyses, the effects did not vary according to vitamin D levels at baseline.
The D-Health investigators randomly assigned more than 21,000 adults to vitamin D or placebo and after 5.7 years of follow-up reported no differences in the primary endpoint of overall mortality. There also were no differences in cardiovascular disease mortality.
Then you have the Mendelian randomized studies, which some have called nature’s RCT. These studies take advantage of the fact that some people are born with gene variations that predispose to low vitamin D levels. More than 60 Mendelian randomization studies have evaluated the consequences of lifelong genetically lowered vitamin D levels on various outcomes; most of these have found null effects.
Then there are the meta-analyses and systematic reviews. I loved the conclusion of this review of systematic reviews from the BMJ (emphasis mine):
“Despite a few hundred systematic reviews and meta-analyses, highly convincing evidence of a clear role of vitamin D does not exist for any outcome, but associations with a selection of outcomes are probable.”
The failure to persuade
My original plan was to emphasize the power of the RCT. Despite strong associations of low vitamin D levels with poor outcomes, the trials show no benefit to treatment. This strongly suggests (or nearly proves) that low vitamin D levels are akin to premature ventricular complexes after myocardial infarction: a marker for risk but not a target for therapy.
But I now see the more important issue as why scientists, funders, clinicians, and patients are not persuaded by clear evidence. Every day in clinic I see patients on vitamin D supplements; the journals keep publishing vitamin D studies. The proponents of vitamin D remain positive. And lately there is outsized attention and hope that vitamin D will mitigate SARS-CoV2 infection – based only on observational data.
You might argue against this point by saying vitamin D is natural and relatively innocuous, so who cares?
I offer three rebuttals to that point: Opportunity costs, distraction, and the insidious danger of poor critical appraisal skills. If you are burning money on vitamin D research, there is less available to study other important issues. If a patient is distracted by low vitamin D levels, she may pay less attention to her high body mass index or hypertension. And on the matter of critical appraisal, trust in medicine requires clinicians to be competent in critical appraisal. And these days, what could be more important than trust in medical professionals?
One major reason for the failure of persuasion of evidence is spin – or language that distracts from the primary endpoint. Here are two (of many) examples:
A meta-analysis of 50 vitamin D trials set out to study mortality. The authors found no significant difference in that primary endpoint. But the second sentence in their conclusion was that vitamin D supplements reduced the risk for cancer deaths by 15%. That’s a secondary endpoint in a study with nonsignificance in the primary endpoint. That is spin. This meta-analysis was completed before the Australian D-Health trial found that cancer deaths were 15% higher in the vitamin D arm, a difference that did not reach statistical significance.
The following example is worse: The authors of the VITAL trial, which found that vitamin D supplements had no effect on the primary endpoint of invasive cancer or cardiovascular disease, published a secondary analysis of the trial looking at a different endpoint: A composite incidence of metastatic and fatal invasive total cancer. They reported a 0.4% lower rate for the vitamin D group, a difference that barely made statistical significance at a P value of .04.
But everyone knows the dangers of reanalyzing data with a new endpoint after you have seen the data. What’s more, even if this were a reasonable post hoc analysis, the results are neither clinically meaningful nor statistically robust. Yet the fatally flawed paper has been viewed 60,000 times and picked up by 48 news outlets.
Another way to distract from nonsignificant primary outcomes is to nitpick the trials. The vitamin D dose wasn’t high enough, for instance. This might persuade me if there were one or two vitamin D trials, but there are hundreds of trials and meta-analyses, and their results are consistently null.
Conclusion: No, it is not hopeless
A nihilist would argue that fighting spin is futile. They would say you can’t fight incentives and business models. The incentive structure to publish is strong, and the journals and media know vitamin D studies garner attention – which is their currency.
I am not a nihilist and believe strongly that we must continue to teach critical appraisal and numerical literacy.
In fact, I would speculate that decades of poor critical appraisal by the medical profession have fostered outsized hope and created erroneous norms.
Imagine a counter-factual world in which clinicians have taught society that the human body is unlike an engine that can be repaired by fixing one part (i.e., the vitamin D level), that magic bullets (insulin) are rare, that most treatments fail, or that you can’t rely on association studies to prove efficacy.
In this world, people would be immune from spin and hype.
The norm would be that pills, supplements, and procedures are not what delivers good health. What delivers health is an amalgam of good luck, healthy habits, and lots of time spent outside playing in the sun.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky., and is a writer and podcaster for Medscape. He espouses a conservative approach to medical practice. He participates in clinical research and writes often about the state of medical evidence. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
The vitamin D story exudes teaching points: It offers a master class in critical appraisal, connecting the concepts of biologic plausibility, flawed surrogate markers, confounded observational studies, and slews of randomized controlled trials (RCTs) showing no benefits on health outcomes.
Yet despite the utter lack of benefit seen in trials, the hype continues. And the pandemic has only enhanced this hype as an onslaught of papers have reported the association of low vitamin D levels and COVID-19 disease.
My questions are simple: Why doesn’t the evidence persuade people? How many nonsignificant trials do we need before researchers stop studying vitamin D, doctors stop (routinely) measuring levels, and patients stop wasting money on the unhelpful supplement? What are the implications for this lack of persuasion?
Before exploring these questions, I want to set out that symptomatic vitamin deficiencies of any sort ought to be corrected.
Biologic plausibility and the pull of observational studies
It has long been known that vitamin D is crucial for bone health and that it can be produced in the skin with sun exposure. In the last decade, however, experts note that nearly every tissue and cell in our body has a vitamin D receptor. It then follows that if this many cells in the body can activate vitamin D, it must be vital for cardiovascular health, immune function, cancer prevention: basically, everything health related.
Oodles of observational studies have found that low serum levels of vitamin D correlate with higher mortality from all causes, cancer, cardiovascular disease, and now even COVID-19. Yet no matter the amount of statistical adjustment in these studies, we cannot know whether these associations are due to true causality.
The major issue is confounding: That is, people with low vitamin D levels have other conditions or diseases that lead to higher rates of ill health. Consider a patient with obesity, arthritis, and cognitive decline; this person is unlikely to do much exercise in the sun and may have low vitamin D levels. The low vitamin D level is simply a marker of overall poor health.
The randomized controlled trials tell a clear story
There are hundreds of vitamin D RCTs. The results simplify into one sentence: Vitamin D supplements do not improve health outcomes.
Here is a short summary of some recent studies.
VITAL, a massive (N > 25,000) RCT with 5 years of follow-up, compared vitamin D supplements to placebo and found no differences in the primary endpoints of cancer or cardiac events. Rates of death from any cause were nearly identical. Crucially, in subgroup analyses, the effects did not vary according to vitamin D levels at baseline.
The D-Health investigators randomly assigned more than 21,000 adults to vitamin D or placebo and after 5.7 years of follow-up reported no differences in the primary endpoint of overall mortality. There also were no differences in cardiovascular disease mortality.
Then you have the Mendelian randomized studies, which some have called nature’s RCT. These studies take advantage of the fact that some people are born with gene variations that predispose to low vitamin D levels. More than 60 Mendelian randomization studies have evaluated the consequences of lifelong genetically lowered vitamin D levels on various outcomes; most of these have found null effects.
Then there are the meta-analyses and systematic reviews. I loved the conclusion of this review of systematic reviews from the BMJ (emphasis mine):
“Despite a few hundred systematic reviews and meta-analyses, highly convincing evidence of a clear role of vitamin D does not exist for any outcome, but associations with a selection of outcomes are probable.”
The failure to persuade
My original plan was to emphasize the power of the RCT. Despite strong associations of low vitamin D levels with poor outcomes, the trials show no benefit to treatment. This strongly suggests (or nearly proves) that low vitamin D levels are akin to premature ventricular complexes after myocardial infarction: a marker for risk but not a target for therapy.
But I now see the more important issue as why scientists, funders, clinicians, and patients are not persuaded by clear evidence. Every day in clinic I see patients on vitamin D supplements; the journals keep publishing vitamin D studies. The proponents of vitamin D remain positive. And lately there is outsized attention and hope that vitamin D will mitigate SARS-CoV2 infection – based only on observational data.
You might argue against this point by saying vitamin D is natural and relatively innocuous, so who cares?
I offer three rebuttals to that point: Opportunity costs, distraction, and the insidious danger of poor critical appraisal skills. If you are burning money on vitamin D research, there is less available to study other important issues. If a patient is distracted by low vitamin D levels, she may pay less attention to her high body mass index or hypertension. And on the matter of critical appraisal, trust in medicine requires clinicians to be competent in critical appraisal. And these days, what could be more important than trust in medical professionals?
One major reason for the failure of persuasion of evidence is spin – or language that distracts from the primary endpoint. Here are two (of many) examples:
A meta-analysis of 50 vitamin D trials set out to study mortality. The authors found no significant difference in that primary endpoint. But the second sentence in their conclusion was that vitamin D supplements reduced the risk for cancer deaths by 15%. That’s a secondary endpoint in a study with nonsignificance in the primary endpoint. That is spin. This meta-analysis was completed before the Australian D-Health trial found that cancer deaths were 15% higher in the vitamin D arm, a difference that did not reach statistical significance.
The following example is worse: The authors of the VITAL trial, which found that vitamin D supplements had no effect on the primary endpoint of invasive cancer or cardiovascular disease, published a secondary analysis of the trial looking at a different endpoint: A composite incidence of metastatic and fatal invasive total cancer. They reported a 0.4% lower rate for the vitamin D group, a difference that barely made statistical significance at a P value of .04.
But everyone knows the dangers of reanalyzing data with a new endpoint after you have seen the data. What’s more, even if this were a reasonable post hoc analysis, the results are neither clinically meaningful nor statistically robust. Yet the fatally flawed paper has been viewed 60,000 times and picked up by 48 news outlets.
Another way to distract from nonsignificant primary outcomes is to nitpick the trials. The vitamin D dose wasn’t high enough, for instance. This might persuade me if there were one or two vitamin D trials, but there are hundreds of trials and meta-analyses, and their results are consistently null.
Conclusion: No, it is not hopeless
A nihilist would argue that fighting spin is futile. They would say you can’t fight incentives and business models. The incentive structure to publish is strong, and the journals and media know vitamin D studies garner attention – which is their currency.
I am not a nihilist and believe strongly that we must continue to teach critical appraisal and numerical literacy.
In fact, I would speculate that decades of poor critical appraisal by the medical profession have fostered outsized hope and created erroneous norms.
Imagine a counter-factual world in which clinicians have taught society that the human body is unlike an engine that can be repaired by fixing one part (i.e., the vitamin D level), that magic bullets (insulin) are rare, that most treatments fail, or that you can’t rely on association studies to prove efficacy.
In this world, people would be immune from spin and hype.
The norm would be that pills, supplements, and procedures are not what delivers good health. What delivers health is an amalgam of good luck, healthy habits, and lots of time spent outside playing in the sun.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky., and is a writer and podcaster for Medscape. He espouses a conservative approach to medical practice. He participates in clinical research and writes often about the state of medical evidence. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Practice valuation
.
Too often, physicians are not receiving a fair return on the equity they have worked so hard to build over several decades, either because they have waited too long and must accept what is offered, or because they simply take the buyer’s word for their practice’s value. Don’t put yourself in either of those positions, and don’t entertain any offers until you obtain an objective appraisal from a neutral party.
Of course, a medical practice is trickier to value than an ordinary business, and usually requires the services of an experienced professional appraiser. Entire books have been written about the process, so I can’t hope to cover it completely in 750 words; but three basic yardsticks are essential for determining the equity, or book value, of a practice:
- Tangible assets. Equipment, cash, accounts receivable, and other property owned by the practice.
- Liabilities. Accounts payable, outstanding loans, and anything else owed to others.
- Intangible assets. Sometimes called “good will” – the reputation of the physicians, the location and name recognition of the practice, the loyalty and volume of patients, and other, well, intangibles.
Valuing tangible assets is comparatively straightforward, but there are several ways to do it, and when reviewing a practice appraisal you should ask which of them was used. Depreciated value is the book value of equipment and supplies as determined by their purchase price, less the amount their value has decreased since purchase. Remaining useful life value estimates how long the equipment can be expected to last. Market (or replacement) value is the amount it would cost on the open market to replace all equipment and supplies.
Intangible assets are more difficult to value. Many components are analyzed, including location, interior and exterior decor, accessibility to patients, age and functional status of equipment, systems in place to promote efficiency, reasons why patients come back (if in fact they do), and the overall reputation of the practice in the community. Other important factors include the “payer mix” (what percentage pays cash, how many third-party contracts are in place and how well they pay, etcetera), the extent and strength of the referral base, and the presence of supplemental income streams, such as clinical research.
It is also important to determine to what extent intangible assets are transferable. For example, unique skills with a laser, neurotoxins, or filler substances, or extraordinary personal charisma, may increase your practice’s value to you, but they are worthless to the next owner, and he or she will be unwilling to pay for them unless your services become part of the deal.
Once again there are many ways to estimate intangible asset value, and once again you should ask which were used. Cash flow analysis works on the assumption that cash flow is a measure of intangible value. Capitalization of earnings puts a value, or capitalization, on the practice’s income streams using a variety of assumptions. Guideline comparison uses various databases to compare your practice with other, similar ones that have changed hands in the past.
Two newer techniques that some consider a better estimate of intangible assets are the replacement method, which estimates the costs of starting the practice over again in the current market; and the excess earnings method, which measures how far above average your practice’s earnings (and thus its overall value) are.
Asset-based valuation is the most popular, but by no means the only method available. Income-based valuation looks at the source and strength of a practice’s income stream as a creator of value, as well as whether or not its income stream under a different owner would mirror its present one. This in turn becomes the basis for an understanding of the fair market value of both tangible and intangible assets. Market valuation combines the asset-based and income-based approaches, along with an analysis of sales and mergers of comparable practices in the community, to determine the value of a practice in its local market.
Whatever methods are used, it is important that the appraisal be done by an experienced and independent financial consultant, that all techniques used in the valuation be divulged and explained, and that documentation is supplied to support the conclusions reached. This is especially important if the appraisal will be relied upon in the sale or merger of the practice.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
.
Too often, physicians are not receiving a fair return on the equity they have worked so hard to build over several decades, either because they have waited too long and must accept what is offered, or because they simply take the buyer’s word for their practice’s value. Don’t put yourself in either of those positions, and don’t entertain any offers until you obtain an objective appraisal from a neutral party.
Of course, a medical practice is trickier to value than an ordinary business, and usually requires the services of an experienced professional appraiser. Entire books have been written about the process, so I can’t hope to cover it completely in 750 words; but three basic yardsticks are essential for determining the equity, or book value, of a practice:
- Tangible assets. Equipment, cash, accounts receivable, and other property owned by the practice.
- Liabilities. Accounts payable, outstanding loans, and anything else owed to others.
- Intangible assets. Sometimes called “good will” – the reputation of the physicians, the location and name recognition of the practice, the loyalty and volume of patients, and other, well, intangibles.
Valuing tangible assets is comparatively straightforward, but there are several ways to do it, and when reviewing a practice appraisal you should ask which of them was used. Depreciated value is the book value of equipment and supplies as determined by their purchase price, less the amount their value has decreased since purchase. Remaining useful life value estimates how long the equipment can be expected to last. Market (or replacement) value is the amount it would cost on the open market to replace all equipment and supplies.
Intangible assets are more difficult to value. Many components are analyzed, including location, interior and exterior decor, accessibility to patients, age and functional status of equipment, systems in place to promote efficiency, reasons why patients come back (if in fact they do), and the overall reputation of the practice in the community. Other important factors include the “payer mix” (what percentage pays cash, how many third-party contracts are in place and how well they pay, etcetera), the extent and strength of the referral base, and the presence of supplemental income streams, such as clinical research.
It is also important to determine to what extent intangible assets are transferable. For example, unique skills with a laser, neurotoxins, or filler substances, or extraordinary personal charisma, may increase your practice’s value to you, but they are worthless to the next owner, and he or she will be unwilling to pay for them unless your services become part of the deal.
Once again there are many ways to estimate intangible asset value, and once again you should ask which were used. Cash flow analysis works on the assumption that cash flow is a measure of intangible value. Capitalization of earnings puts a value, or capitalization, on the practice’s income streams using a variety of assumptions. Guideline comparison uses various databases to compare your practice with other, similar ones that have changed hands in the past.
Two newer techniques that some consider a better estimate of intangible assets are the replacement method, which estimates the costs of starting the practice over again in the current market; and the excess earnings method, which measures how far above average your practice’s earnings (and thus its overall value) are.
Asset-based valuation is the most popular, but by no means the only method available. Income-based valuation looks at the source and strength of a practice’s income stream as a creator of value, as well as whether or not its income stream under a different owner would mirror its present one. This in turn becomes the basis for an understanding of the fair market value of both tangible and intangible assets. Market valuation combines the asset-based and income-based approaches, along with an analysis of sales and mergers of comparable practices in the community, to determine the value of a practice in its local market.
Whatever methods are used, it is important that the appraisal be done by an experienced and independent financial consultant, that all techniques used in the valuation be divulged and explained, and that documentation is supplied to support the conclusions reached. This is especially important if the appraisal will be relied upon in the sale or merger of the practice.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
.
Too often, physicians are not receiving a fair return on the equity they have worked so hard to build over several decades, either because they have waited too long and must accept what is offered, or because they simply take the buyer’s word for their practice’s value. Don’t put yourself in either of those positions, and don’t entertain any offers until you obtain an objective appraisal from a neutral party.
Of course, a medical practice is trickier to value than an ordinary business, and usually requires the services of an experienced professional appraiser. Entire books have been written about the process, so I can’t hope to cover it completely in 750 words; but three basic yardsticks are essential for determining the equity, or book value, of a practice:
- Tangible assets. Equipment, cash, accounts receivable, and other property owned by the practice.
- Liabilities. Accounts payable, outstanding loans, and anything else owed to others.
- Intangible assets. Sometimes called “good will” – the reputation of the physicians, the location and name recognition of the practice, the loyalty and volume of patients, and other, well, intangibles.
Valuing tangible assets is comparatively straightforward, but there are several ways to do it, and when reviewing a practice appraisal you should ask which of them was used. Depreciated value is the book value of equipment and supplies as determined by their purchase price, less the amount their value has decreased since purchase. Remaining useful life value estimates how long the equipment can be expected to last. Market (or replacement) value is the amount it would cost on the open market to replace all equipment and supplies.
Intangible assets are more difficult to value. Many components are analyzed, including location, interior and exterior decor, accessibility to patients, age and functional status of equipment, systems in place to promote efficiency, reasons why patients come back (if in fact they do), and the overall reputation of the practice in the community. Other important factors include the “payer mix” (what percentage pays cash, how many third-party contracts are in place and how well they pay, etcetera), the extent and strength of the referral base, and the presence of supplemental income streams, such as clinical research.
It is also important to determine to what extent intangible assets are transferable. For example, unique skills with a laser, neurotoxins, or filler substances, or extraordinary personal charisma, may increase your practice’s value to you, but they are worthless to the next owner, and he or she will be unwilling to pay for them unless your services become part of the deal.
Once again there are many ways to estimate intangible asset value, and once again you should ask which were used. Cash flow analysis works on the assumption that cash flow is a measure of intangible value. Capitalization of earnings puts a value, or capitalization, on the practice’s income streams using a variety of assumptions. Guideline comparison uses various databases to compare your practice with other, similar ones that have changed hands in the past.
Two newer techniques that some consider a better estimate of intangible assets are the replacement method, which estimates the costs of starting the practice over again in the current market; and the excess earnings method, which measures how far above average your practice’s earnings (and thus its overall value) are.
Asset-based valuation is the most popular, but by no means the only method available. Income-based valuation looks at the source and strength of a practice’s income stream as a creator of value, as well as whether or not its income stream under a different owner would mirror its present one. This in turn becomes the basis for an understanding of the fair market value of both tangible and intangible assets. Market valuation combines the asset-based and income-based approaches, along with an analysis of sales and mergers of comparable practices in the community, to determine the value of a practice in its local market.
Whatever methods are used, it is important that the appraisal be done by an experienced and independent financial consultant, that all techniques used in the valuation be divulged and explained, and that documentation is supplied to support the conclusions reached. This is especially important if the appraisal will be relied upon in the sale or merger of the practice.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
To a perfect day
Motionless, every Olympic skater starts off perfectly. Once the music starts, it’s up to them whether they will continue on perfectly or not. In this way, you’re just like an Olympic skater. Each day, a skating program. The music starts the moment your foot touches the floor in the morning. It’s up to you if the rest of the day will continue on flawlessly or not. To this point, I’ve yet to have a perfect day.
If I’m honest, my “perfect day” streak typically ends once I’ve made coffee. By then, I’ll have spilled a few grains of grounds or clinked mugs together when taking one from the cupboard. (D’oh!) Hardly ever can I make it to backing out of the driveway, let alone through a patient encounter. I’ve had a few procedures that when complete I’ve thought, “well, that looks great.” I can remember encounters that went brilliantly despite a high technical difficulty. I’ve also tagged a 7-iron shot 160 downwind yards to within inches of the cup. But I’ve hardly ever done anything in my life perfectly.
What does it mean to be perfect? Well, there have been 23 perfect baseball games. In 1972, the Miami Dolphins had the only perfect NFL season, 14-0 (although my 2007 Patriots went 18-0 before losing to the – ugh – Giants). Every year, several hundred students score a perfect 1600 on the SAT. In an underground vault somewhere in France is a perfect sphere, a perfectly spherical 1-kg mass of pure silicon. There are at least 51 perfect numbers. And model Bella Hadid’s exactly 1.62-ratioed face is said to be perfectly beautiful. But yet, U.S. skater Nathan Chen’s seemingly flawless 113.97-point short program in Beijing, still imperfect.
Attempting a perfect day or perfect surgery or a perfect pour over coffee is a fun game, but perfectionism has an insidious side. Some of us feel this way every day: We must do it exactly right, every time. Even an insignificant imperfection or error feels like failure. A 3.90 GPA is a fail. 515 on the MCAT, not nearly good enough. For them, the burden of perfection is crushing. It is hard for some to recognize that even if your performance could not be improved, the outcome can still be flawed. A chip in the ice, a patient showing up late, an interviewer with an agenda, a missed referee call can all flub up an otherwise flawless day. It isn’t necessary to abandon hope, all ye who live in the real world. Although achieving perfection is usually impossible, reward comes from the pursuit of perfection, not from holding it. It is called perfectionistic striving and in contrast to perfectionistic concerns, it is associated with resilience and positive mood. To do so you must combine giving your all with acceptance of whatever the outcome.
Keith Jarrett is one of the greatest jazz pianists of all time. He is a true perfectionist, precise in his standards and exacting in expectations. In 1975 in Cologne, Germany, he agreed to play at the behest of a teenage girl who arranged to have him perform at the opera house. Except, there was a miscommunication and only a small, broken rehearsal piano was available. As the story goes, she approached him as he waited to be taken back to his hotel, the concert was canceled and she somehow convinced him to play on the nearly unplayable instrument. The result is the Köln Concert, one of the greatest jazz performances in history. It was perfectly imperfect.
Yes, even the 1-kg sphere has femtogram quantities of other elements mixed in – the universal standard for perfect is itself, imperfect. It doesn’t matter. It’s the pursuit of such that makes life worthwhile. There’s always tomorrow. Have your coffee grinders ready.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected]
Motionless, every Olympic skater starts off perfectly. Once the music starts, it’s up to them whether they will continue on perfectly or not. In this way, you’re just like an Olympic skater. Each day, a skating program. The music starts the moment your foot touches the floor in the morning. It’s up to you if the rest of the day will continue on flawlessly or not. To this point, I’ve yet to have a perfect day.
If I’m honest, my “perfect day” streak typically ends once I’ve made coffee. By then, I’ll have spilled a few grains of grounds or clinked mugs together when taking one from the cupboard. (D’oh!) Hardly ever can I make it to backing out of the driveway, let alone through a patient encounter. I’ve had a few procedures that when complete I’ve thought, “well, that looks great.” I can remember encounters that went brilliantly despite a high technical difficulty. I’ve also tagged a 7-iron shot 160 downwind yards to within inches of the cup. But I’ve hardly ever done anything in my life perfectly.
What does it mean to be perfect? Well, there have been 23 perfect baseball games. In 1972, the Miami Dolphins had the only perfect NFL season, 14-0 (although my 2007 Patriots went 18-0 before losing to the – ugh – Giants). Every year, several hundred students score a perfect 1600 on the SAT. In an underground vault somewhere in France is a perfect sphere, a perfectly spherical 1-kg mass of pure silicon. There are at least 51 perfect numbers. And model Bella Hadid’s exactly 1.62-ratioed face is said to be perfectly beautiful. But yet, U.S. skater Nathan Chen’s seemingly flawless 113.97-point short program in Beijing, still imperfect.
Attempting a perfect day or perfect surgery or a perfect pour over coffee is a fun game, but perfectionism has an insidious side. Some of us feel this way every day: We must do it exactly right, every time. Even an insignificant imperfection or error feels like failure. A 3.90 GPA is a fail. 515 on the MCAT, not nearly good enough. For them, the burden of perfection is crushing. It is hard for some to recognize that even if your performance could not be improved, the outcome can still be flawed. A chip in the ice, a patient showing up late, an interviewer with an agenda, a missed referee call can all flub up an otherwise flawless day. It isn’t necessary to abandon hope, all ye who live in the real world. Although achieving perfection is usually impossible, reward comes from the pursuit of perfection, not from holding it. It is called perfectionistic striving and in contrast to perfectionistic concerns, it is associated with resilience and positive mood. To do so you must combine giving your all with acceptance of whatever the outcome.
Keith Jarrett is one of the greatest jazz pianists of all time. He is a true perfectionist, precise in his standards and exacting in expectations. In 1975 in Cologne, Germany, he agreed to play at the behest of a teenage girl who arranged to have him perform at the opera house. Except, there was a miscommunication and only a small, broken rehearsal piano was available. As the story goes, she approached him as he waited to be taken back to his hotel, the concert was canceled and she somehow convinced him to play on the nearly unplayable instrument. The result is the Köln Concert, one of the greatest jazz performances in history. It was perfectly imperfect.
Yes, even the 1-kg sphere has femtogram quantities of other elements mixed in – the universal standard for perfect is itself, imperfect. It doesn’t matter. It’s the pursuit of such that makes life worthwhile. There’s always tomorrow. Have your coffee grinders ready.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected]
Motionless, every Olympic skater starts off perfectly. Once the music starts, it’s up to them whether they will continue on perfectly or not. In this way, you’re just like an Olympic skater. Each day, a skating program. The music starts the moment your foot touches the floor in the morning. It’s up to you if the rest of the day will continue on flawlessly or not. To this point, I’ve yet to have a perfect day.
If I’m honest, my “perfect day” streak typically ends once I’ve made coffee. By then, I’ll have spilled a few grains of grounds or clinked mugs together when taking one from the cupboard. (D’oh!) Hardly ever can I make it to backing out of the driveway, let alone through a patient encounter. I’ve had a few procedures that when complete I’ve thought, “well, that looks great.” I can remember encounters that went brilliantly despite a high technical difficulty. I’ve also tagged a 7-iron shot 160 downwind yards to within inches of the cup. But I’ve hardly ever done anything in my life perfectly.
What does it mean to be perfect? Well, there have been 23 perfect baseball games. In 1972, the Miami Dolphins had the only perfect NFL season, 14-0 (although my 2007 Patriots went 18-0 before losing to the – ugh – Giants). Every year, several hundred students score a perfect 1600 on the SAT. In an underground vault somewhere in France is a perfect sphere, a perfectly spherical 1-kg mass of pure silicon. There are at least 51 perfect numbers. And model Bella Hadid’s exactly 1.62-ratioed face is said to be perfectly beautiful. But yet, U.S. skater Nathan Chen’s seemingly flawless 113.97-point short program in Beijing, still imperfect.
Attempting a perfect day or perfect surgery or a perfect pour over coffee is a fun game, but perfectionism has an insidious side. Some of us feel this way every day: We must do it exactly right, every time. Even an insignificant imperfection or error feels like failure. A 3.90 GPA is a fail. 515 on the MCAT, not nearly good enough. For them, the burden of perfection is crushing. It is hard for some to recognize that even if your performance could not be improved, the outcome can still be flawed. A chip in the ice, a patient showing up late, an interviewer with an agenda, a missed referee call can all flub up an otherwise flawless day. It isn’t necessary to abandon hope, all ye who live in the real world. Although achieving perfection is usually impossible, reward comes from the pursuit of perfection, not from holding it. It is called perfectionistic striving and in contrast to perfectionistic concerns, it is associated with resilience and positive mood. To do so you must combine giving your all with acceptance of whatever the outcome.
Keith Jarrett is one of the greatest jazz pianists of all time. He is a true perfectionist, precise in his standards and exacting in expectations. In 1975 in Cologne, Germany, he agreed to play at the behest of a teenage girl who arranged to have him perform at the opera house. Except, there was a miscommunication and only a small, broken rehearsal piano was available. As the story goes, she approached him as he waited to be taken back to his hotel, the concert was canceled and she somehow convinced him to play on the nearly unplayable instrument. The result is the Köln Concert, one of the greatest jazz performances in history. It was perfectly imperfect.
Yes, even the 1-kg sphere has femtogram quantities of other elements mixed in – the universal standard for perfect is itself, imperfect. It doesn’t matter. It’s the pursuit of such that makes life worthwhile. There’s always tomorrow. Have your coffee grinders ready.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected]
Tips for connecting with your patients
It is a tough time to be a doctor. With the stresses of the pandemic, the continued unfettered rise of insurance company BS, and so many medical groups being bought up that we often don’t even know who makes the decisions, the patient can sometimes be hidden in the equation.
Be curious
When physicians are curious about why patients have symptoms, how those symptoms will affect their lives, and how worried the patient is about them, patients feel cared about.
Ascertaining how concerned patients are about their symptoms will help you make decisions on whether symptoms you are not concerned about actually need to be treated.
Limit use of EHRs when possible
Use of the electronic health record during visits is essential, but focusing on it too much can put a barrier between the physician and the patient.
Marmor and colleagues found there is an inverse relationship between time spent on the EHR by a patient’s physician and the patient’s satisfaction.1
Eye contact with the patient is important, especially when patients are sharing concerns they are scared about and upsetting experiences. There can be awkward pauses when looking things up on the EHR. Fill those pauses by explaining to the patient what you are doing, or chatting with the patient.
Consider teaching medical students
When a medical student works with you, it doubles the time the patient gets with a concerned listener. Students also can do a great job with timely follow-up and checking in with worried patients.
By having the student present in the clinic room, with the patient present, the patient can really feel heard. The student shares all the details the patient shared, and now their physician is hearing an organized, thoughtful report of the patients concerns.
In fact, I was involved in a study that showed that patients preferred in room presentations, and that they were more satisfied when students presented in the room.2
Use healing words
Some words carry loaded emotions. The word chronic, for example, has negative connotations, whereas the term persisting does not.
I will often ask patients how long they have been suffering from a symptom to imply my concern for what they are going through. The term “chief complaint” is outdated, and upsets patients when they see it in their medical record.
As a patient of mine once said to me: “I never complained about that problem, I just brought it to your attention.” No one wants to be seen as a complainer. Substituting the word concern for complaint works well.
Explain as you examine
People love to hear the term normal. When you are examining a patient, let them know when findings are normal.
I also find it helpful to explain to patients why I am doing certain physical exam maneuvers. This helps them assess how thorough we are in our thought process.
When patients feel their physicians are thorough, they have more confidence in them.
In summary
- Be curious.
- Do not overly focus on the EHR.
- Consider teaching a medical student.
- Be careful of word choice.
- “Overexplain” the physical exam.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Marmor RA et al. Appl Clin Inform. 2018 Jan;9(1):11-4.
2. Rogers HD et al. Acad Med. 2003 Sep;78(9):945-9.
It is a tough time to be a doctor. With the stresses of the pandemic, the continued unfettered rise of insurance company BS, and so many medical groups being bought up that we often don’t even know who makes the decisions, the patient can sometimes be hidden in the equation.
Be curious
When physicians are curious about why patients have symptoms, how those symptoms will affect their lives, and how worried the patient is about them, patients feel cared about.
Ascertaining how concerned patients are about their symptoms will help you make decisions on whether symptoms you are not concerned about actually need to be treated.
Limit use of EHRs when possible
Use of the electronic health record during visits is essential, but focusing on it too much can put a barrier between the physician and the patient.
Marmor and colleagues found there is an inverse relationship between time spent on the EHR by a patient’s physician and the patient’s satisfaction.1
Eye contact with the patient is important, especially when patients are sharing concerns they are scared about and upsetting experiences. There can be awkward pauses when looking things up on the EHR. Fill those pauses by explaining to the patient what you are doing, or chatting with the patient.
Consider teaching medical students
When a medical student works with you, it doubles the time the patient gets with a concerned listener. Students also can do a great job with timely follow-up and checking in with worried patients.
By having the student present in the clinic room, with the patient present, the patient can really feel heard. The student shares all the details the patient shared, and now their physician is hearing an organized, thoughtful report of the patients concerns.
In fact, I was involved in a study that showed that patients preferred in room presentations, and that they were more satisfied when students presented in the room.2
Use healing words
Some words carry loaded emotions. The word chronic, for example, has negative connotations, whereas the term persisting does not.
I will often ask patients how long they have been suffering from a symptom to imply my concern for what they are going through. The term “chief complaint” is outdated, and upsets patients when they see it in their medical record.
As a patient of mine once said to me: “I never complained about that problem, I just brought it to your attention.” No one wants to be seen as a complainer. Substituting the word concern for complaint works well.
Explain as you examine
People love to hear the term normal. When you are examining a patient, let them know when findings are normal.
I also find it helpful to explain to patients why I am doing certain physical exam maneuvers. This helps them assess how thorough we are in our thought process.
When patients feel their physicians are thorough, they have more confidence in them.
In summary
- Be curious.
- Do not overly focus on the EHR.
- Consider teaching a medical student.
- Be careful of word choice.
- “Overexplain” the physical exam.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Marmor RA et al. Appl Clin Inform. 2018 Jan;9(1):11-4.
2. Rogers HD et al. Acad Med. 2003 Sep;78(9):945-9.
It is a tough time to be a doctor. With the stresses of the pandemic, the continued unfettered rise of insurance company BS, and so many medical groups being bought up that we often don’t even know who makes the decisions, the patient can sometimes be hidden in the equation.
Be curious
When physicians are curious about why patients have symptoms, how those symptoms will affect their lives, and how worried the patient is about them, patients feel cared about.
Ascertaining how concerned patients are about their symptoms will help you make decisions on whether symptoms you are not concerned about actually need to be treated.
Limit use of EHRs when possible
Use of the electronic health record during visits is essential, but focusing on it too much can put a barrier between the physician and the patient.
Marmor and colleagues found there is an inverse relationship between time spent on the EHR by a patient’s physician and the patient’s satisfaction.1
Eye contact with the patient is important, especially when patients are sharing concerns they are scared about and upsetting experiences. There can be awkward pauses when looking things up on the EHR. Fill those pauses by explaining to the patient what you are doing, or chatting with the patient.
Consider teaching medical students
When a medical student works with you, it doubles the time the patient gets with a concerned listener. Students also can do a great job with timely follow-up and checking in with worried patients.
By having the student present in the clinic room, with the patient present, the patient can really feel heard. The student shares all the details the patient shared, and now their physician is hearing an organized, thoughtful report of the patients concerns.
In fact, I was involved in a study that showed that patients preferred in room presentations, and that they were more satisfied when students presented in the room.2
Use healing words
Some words carry loaded emotions. The word chronic, for example, has negative connotations, whereas the term persisting does not.
I will often ask patients how long they have been suffering from a symptom to imply my concern for what they are going through. The term “chief complaint” is outdated, and upsets patients when they see it in their medical record.
As a patient of mine once said to me: “I never complained about that problem, I just brought it to your attention.” No one wants to be seen as a complainer. Substituting the word concern for complaint works well.
Explain as you examine
People love to hear the term normal. When you are examining a patient, let them know when findings are normal.
I also find it helpful to explain to patients why I am doing certain physical exam maneuvers. This helps them assess how thorough we are in our thought process.
When patients feel their physicians are thorough, they have more confidence in them.
In summary
- Be curious.
- Do not overly focus on the EHR.
- Consider teaching a medical student.
- Be careful of word choice.
- “Overexplain” the physical exam.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Marmor RA et al. Appl Clin Inform. 2018 Jan;9(1):11-4.
2. Rogers HD et al. Acad Med. 2003 Sep;78(9):945-9.
A 19-month-old vaccinated female with 2 days of rash
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
Acute hemorrhagic edema of infancy (AHEI) is a leukocytoclastic vasculitis that typically affects children between 4 months and 2 years of age.1 Etiology is unknown but the majority of cases are preceded by infections, vaccinations, or certain medications.2
AHEI is a self-limited disease that runs a benign course with spontaneous resolution within days to 3 weeks.3 Classic presentation involves acute onset of fever, purpura, ecchymosis, and inflammatory edema. Edema is often the first sign, and may involve the face, ears, scrotum, or extremities. Hemorrhagic lesions may vary in size but often coalesce and present in a distinctive “cockade” or rosette pattern with scalloped borders. Systemic manifestations are rare, but renal and joint involvement may occur.4 Despite the dramatic and sometimes extensive appearance of the dermatologic manifestations, patients with AHEI are usually not in significant distress.
Diagnosis is clinical, but skin biopsy may show leukocytoclastic vasculitis of the superficial small vessels with infiltrations of neutrophils, extravasation of red blood cells, and fibrinoid necrosis.5 In most cases, immunofluorescence is negative for perivascular IgA deposition. Treatment is symptomatic as the disease resolves spontaneously. Recurrence is uncommon but may occur, and usually occurs early.
What is on the differential?
Kawasaki disease. Similar to AHEI, patients with Kawasaki disease also may present with facial and extremity edema. However, patients with Kawasaki disease appear sicker, have associated lymphadenopathy, conjunctivitis, and fever longer than 5 days. The lack of elevated inflammatory markers, acute-onset, classic dermatologic lesions, and nontoxic appearance in our patient rule out Kawasaki disease and make AHEI more likely.
IgA vasculitis/Henoch-Schönlein purpura. The distinction between AHEI and Henoch-Schönlein purpura is among the most challenging. AHEI commonly afflicts younger children ranging from 4 months to 2 years, whereas Henoch-Schönlein purpura occurs in older children from 3 to 6 years of age. Visceral involvement is rare in AHEI, but frequently presents in Henoch-Schönlein purpura with gastrointestinal and renal complications. Although our patient had both mild renal involvement and a distribution primarily on the buttocks and lower limbs, similar to the classic distribution of Henoch-Schönlein purpura, the younger age and lack of gastrointestinal and arthritic manifestations make AHEI more likely.
Gianotti-Crosti syndrome. Gianotti-Crosti syndrome, also known as papulovesicular acrodermatitis of childhood, mainly affects children between the ages of 6 months and 12 years. Like AHEI, Gianotti-Crosti is a self-limiting condition likely triggered by viral infection or immunization. However, Gianotti-Crosti is characterized by a papular rash that may last for several weeks. Neither AHEI nor Gianotti-Crosti are pruritic, but patients with Gianotti-Crosti tend to have either inguinal or axillary lymphadenopathy. Our patient’s large, coalescing dusky red patches and edematous plaques without lymphadenopathy are more consistent with AHEI.
Erythema multiforme. Erythema multiforme is an acute, immune-mediated condition characterized by distinctive target-like lesions on the skin often accompanied by erosions or bullae. Unlike AHEI, erythema multiforme can involve the oral, genital, and/or ocular mucosae. Erythema multiforme is rare before the age of 4 years. Although the targetoid or annular purpuric configuration of erythema multiforme may present similarly to AHEI in some cases, the young age of our patient and the lack of mucosal involvement make AHEI more likely.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Ms. Kleinman is a pediatric dermatology research associate at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Neither Dr. Matiz nor Ms. Kleinman has any relevant financial disclosures.
References
1. Savino F et al. Pediatr Dermatol. 2013;30(6):e149-e152.
2. Carboni E et al. F1000Res. 2019;8:1771. 2019 Oct 17.
3. Fiore E et al. J Am Acad Dermatol. 2008;59(4):684-95.
4. Watanabe T and Sato Y. Pediatr Nephrol. 2007;22(11):1979-81.
5. Cunha DF et al. Autops Case Rep. 2015;5(3):37-41.
A 34-year-old male presented with 10 days of a pruritic rash
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Late-onset recurrence in breast cancer: Implications for women’s health clinicians in survivorship care
Improved treatments for breast cancer (BC) and effective screening programs have resulted in a BC mortality rate reduction of 41% since 1989.1 Because BC is the leading cause of cancer in women, these mortality improvements have resulted in more than 3 million BC survivors in the United States.2,3 With longer-term survival, there is increasing interest in late-onset recurrences.4,5 A recent study has provided an improved understanding of the risk of lateonset recurrence in women with 10 years of disease-free survival, an important finding for women’s health providers because oncologists do not typically follow survivors after 10 years of disease-free survival.4
Recent study looks at incidence of late-onset recurrence
Pederson and colleagues evaluated all patients diagnosed with BC in Denmark from 1987 through 2004.4 Those patients without evidence of recurrence at 10 years were then followed utilizing population-based linked registries to identify patients who subsequently developed a local, regional, or distant late-onset recurrence. The authors evaluated the frequency of late recurrence and identified associations with demographic and tumor characteristics.
What they found
A total of 36,920 patients were diagnosed with BC in Denmark between 1987-2004, of whom 20,315 (55%) were identified as disease free for at least 10 years. Late-onset recurrence occurred in 2,595 (12.8%) with the strongest associations of recurrence seen in patients who had a tumor size >2 cm and lymph node‒positive (involving 4 or more nodes) disease (24.6%), compared with 12.7% in patients with tumors <2 cm and node-negative disease. Several other factors were associated with a higher risk of late-onset recurrence and are included in the TABLE. Half of the recurrences occurred between 10 and 15 years after the primary diagnosis.

Prior research
These findings are consistent with another recent study showing that BC patients have a 1% to 2%/year risk of recurrence after 10 disease-free years.5 Strengths of this study include:
- population-based, including all women with BC
- long-term follow-up for up to 32 years
- universal health care in Denmark, which results in robust and linked databases and very few missing data points.
There were two notable weaknesses to consider:
- Treatment regimens changed considerably during the time frame of the study (1997-2018), particularly the duration of tamoxifen use in patients with HR-positive disease. In this study nearly all patients received 5 years or less of tamoxifen. Since the mid-2010s, 10 years of hormonal adjuvant therapy has become routine in HR-positive BC, which reduces recurrences, including late-onset recurrence.6 The effect of 10 years of tamoxifen would very likely have resulted in less late-onset recurrence in the HR-positive population in this study.
- There is a lack of racial diversity in the Danish population, and the study findings may not translate to Black patients who have a higher frequency of triple-negative BC with a different risk of late-onset recurrence.7
Practice takeaways
Cancer surveillance. There are 3+ million BC survivors in the United States, and a 55%+ likelihood that they will be disease free for 10 years. This is clearly an important population to the women’s health care provider. This study, and previous research, suggests that among 10-year-disease-free survivors, 1% to 2% will recur annually, with higher rates amongst HR-positive, lymph-node positive women under age 40, and in the first 5 years following the 10-year post–initial diagnosis mark, so ongoing surveillance is imperative. Annual clinical breast examinations along with annual (not biennial) mammography should be performed.8 Digital breast tomosynthesis has improved specificity and sensitivity for BC detection and is the preferred modality when it is available.
Management of menopausal symptoms. These findings also have implications for menopausal hormone therapy for patients with symptoms. Because HR-positive patients have an increased risk of late-onset recurrence, nonhormonal therapies should be considered as first-line therapy for patients with menopausal symptoms. If hormone therapy is being considered, providers and patients should use shared decision making to balance the potential benefits (reduction in symptoms, possible cardiovascular benefits, and reduction in bone loss) with the risks (increased risk of recurrence and venous thromboembolism), even if patients are remote from the original diagnosis (ie, 10-year disease-free survival).
Topical estrogen therapies would be preferred for patients with significant urogenital atrophic symptoms who fail nonhormonal therapies due to substantially less systemic absorption and the lack of need to add a progestin.9,10 If oral therapy is being considered, I carefully counsel these women about the likely increased risk of recurrence and, if possible, include their breast oncologist in the discussion. ●
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33. doi: 10.3322/caac.21654.
- de Moor JS, Mariotto AB, Parry C, et al. Cancer survivors in the United States: prevalence across the survivorship trajectory and implications for care. Cancer Epidemiol Biomarkers Prev. 2013;22:561- 570. doi: 10.1158/1055-9965.EPI-12-1356.
- Carreira H, Williams R, Funston G, et al. Associations between breast cancer survivorship and adverse mental health outcomes: a matched population-based cohort study in the United Kingdom. PLOS Med. 2021;18:e1003504. doi: 10.1371/journal.pmed.1003504.
- Pedersen RN, Esen BÖ, Mellemkjær L, et al. The incidence of breast cancer recurrence 10-32 years after primary diagnosis. J Natl Cancer Inst. November 8, 2021. doi: 10.1093/jnci/djab202.
- Pan H, Gray R, Braybrooke J, et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377:1836- 1846. doi: 10.1056/NEJMoa1701830.
- Davies C, Pan H, Godwin J, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. doi: 10.1016/S0140-6736(12)61963-1.
- Scott LC, Mobley LR, Kuo TM, et al. Update on triple‐negative breast cancer disparities for the United States: a population‐based study from the United States Cancer Statistics database, 2010 through 2014. Cancer. 2019;125:3412-3417. doi: 10.1002/cncr.32207.
- NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. 2021; Version 2.2022.
- Crandall CJ, Diamant A, Santoro N. Safety of vaginal estrogens: a systematic review. Menopause. 2020;27:339-360. doi: 10.1097 /GME.0000000000001468.
- Treatment of urogenital symptoms in individuals with a history of estrogen-dependent breast cancer: clinical consensus. Obstet Gynecol. 2021;138:950-960. doi: 10.1097/AOG .0000000000004601.
Improved treatments for breast cancer (BC) and effective screening programs have resulted in a BC mortality rate reduction of 41% since 1989.1 Because BC is the leading cause of cancer in women, these mortality improvements have resulted in more than 3 million BC survivors in the United States.2,3 With longer-term survival, there is increasing interest in late-onset recurrences.4,5 A recent study has provided an improved understanding of the risk of lateonset recurrence in women with 10 years of disease-free survival, an important finding for women’s health providers because oncologists do not typically follow survivors after 10 years of disease-free survival.4
Recent study looks at incidence of late-onset recurrence
Pederson and colleagues evaluated all patients diagnosed with BC in Denmark from 1987 through 2004.4 Those patients without evidence of recurrence at 10 years were then followed utilizing population-based linked registries to identify patients who subsequently developed a local, regional, or distant late-onset recurrence. The authors evaluated the frequency of late recurrence and identified associations with demographic and tumor characteristics.
What they found
A total of 36,920 patients were diagnosed with BC in Denmark between 1987-2004, of whom 20,315 (55%) were identified as disease free for at least 10 years. Late-onset recurrence occurred in 2,595 (12.8%) with the strongest associations of recurrence seen in patients who had a tumor size >2 cm and lymph node‒positive (involving 4 or more nodes) disease (24.6%), compared with 12.7% in patients with tumors <2 cm and node-negative disease. Several other factors were associated with a higher risk of late-onset recurrence and are included in the TABLE. Half of the recurrences occurred between 10 and 15 years after the primary diagnosis.

Prior research
These findings are consistent with another recent study showing that BC patients have a 1% to 2%/year risk of recurrence after 10 disease-free years.5 Strengths of this study include:
- population-based, including all women with BC
- long-term follow-up for up to 32 years
- universal health care in Denmark, which results in robust and linked databases and very few missing data points.
There were two notable weaknesses to consider:
- Treatment regimens changed considerably during the time frame of the study (1997-2018), particularly the duration of tamoxifen use in patients with HR-positive disease. In this study nearly all patients received 5 years or less of tamoxifen. Since the mid-2010s, 10 years of hormonal adjuvant therapy has become routine in HR-positive BC, which reduces recurrences, including late-onset recurrence.6 The effect of 10 years of tamoxifen would very likely have resulted in less late-onset recurrence in the HR-positive population in this study.
- There is a lack of racial diversity in the Danish population, and the study findings may not translate to Black patients who have a higher frequency of triple-negative BC with a different risk of late-onset recurrence.7
Practice takeaways
Cancer surveillance. There are 3+ million BC survivors in the United States, and a 55%+ likelihood that they will be disease free for 10 years. This is clearly an important population to the women’s health care provider. This study, and previous research, suggests that among 10-year-disease-free survivors, 1% to 2% will recur annually, with higher rates amongst HR-positive, lymph-node positive women under age 40, and in the first 5 years following the 10-year post–initial diagnosis mark, so ongoing surveillance is imperative. Annual clinical breast examinations along with annual (not biennial) mammography should be performed.8 Digital breast tomosynthesis has improved specificity and sensitivity for BC detection and is the preferred modality when it is available.
Management of menopausal symptoms. These findings also have implications for menopausal hormone therapy for patients with symptoms. Because HR-positive patients have an increased risk of late-onset recurrence, nonhormonal therapies should be considered as first-line therapy for patients with menopausal symptoms. If hormone therapy is being considered, providers and patients should use shared decision making to balance the potential benefits (reduction in symptoms, possible cardiovascular benefits, and reduction in bone loss) with the risks (increased risk of recurrence and venous thromboembolism), even if patients are remote from the original diagnosis (ie, 10-year disease-free survival).
Topical estrogen therapies would be preferred for patients with significant urogenital atrophic symptoms who fail nonhormonal therapies due to substantially less systemic absorption and the lack of need to add a progestin.9,10 If oral therapy is being considered, I carefully counsel these women about the likely increased risk of recurrence and, if possible, include their breast oncologist in the discussion. ●
Improved treatments for breast cancer (BC) and effective screening programs have resulted in a BC mortality rate reduction of 41% since 1989.1 Because BC is the leading cause of cancer in women, these mortality improvements have resulted in more than 3 million BC survivors in the United States.2,3 With longer-term survival, there is increasing interest in late-onset recurrences.4,5 A recent study has provided an improved understanding of the risk of lateonset recurrence in women with 10 years of disease-free survival, an important finding for women’s health providers because oncologists do not typically follow survivors after 10 years of disease-free survival.4
Recent study looks at incidence of late-onset recurrence
Pederson and colleagues evaluated all patients diagnosed with BC in Denmark from 1987 through 2004.4 Those patients without evidence of recurrence at 10 years were then followed utilizing population-based linked registries to identify patients who subsequently developed a local, regional, or distant late-onset recurrence. The authors evaluated the frequency of late recurrence and identified associations with demographic and tumor characteristics.
What they found
A total of 36,920 patients were diagnosed with BC in Denmark between 1987-2004, of whom 20,315 (55%) were identified as disease free for at least 10 years. Late-onset recurrence occurred in 2,595 (12.8%) with the strongest associations of recurrence seen in patients who had a tumor size >2 cm and lymph node‒positive (involving 4 or more nodes) disease (24.6%), compared with 12.7% in patients with tumors <2 cm and node-negative disease. Several other factors were associated with a higher risk of late-onset recurrence and are included in the TABLE. Half of the recurrences occurred between 10 and 15 years after the primary diagnosis.

Prior research
These findings are consistent with another recent study showing that BC patients have a 1% to 2%/year risk of recurrence after 10 disease-free years.5 Strengths of this study include:
- population-based, including all women with BC
- long-term follow-up for up to 32 years
- universal health care in Denmark, which results in robust and linked databases and very few missing data points.
There were two notable weaknesses to consider:
- Treatment regimens changed considerably during the time frame of the study (1997-2018), particularly the duration of tamoxifen use in patients with HR-positive disease. In this study nearly all patients received 5 years or less of tamoxifen. Since the mid-2010s, 10 years of hormonal adjuvant therapy has become routine in HR-positive BC, which reduces recurrences, including late-onset recurrence.6 The effect of 10 years of tamoxifen would very likely have resulted in less late-onset recurrence in the HR-positive population in this study.
- There is a lack of racial diversity in the Danish population, and the study findings may not translate to Black patients who have a higher frequency of triple-negative BC with a different risk of late-onset recurrence.7
Practice takeaways
Cancer surveillance. There are 3+ million BC survivors in the United States, and a 55%+ likelihood that they will be disease free for 10 years. This is clearly an important population to the women’s health care provider. This study, and previous research, suggests that among 10-year-disease-free survivors, 1% to 2% will recur annually, with higher rates amongst HR-positive, lymph-node positive women under age 40, and in the first 5 years following the 10-year post–initial diagnosis mark, so ongoing surveillance is imperative. Annual clinical breast examinations along with annual (not biennial) mammography should be performed.8 Digital breast tomosynthesis has improved specificity and sensitivity for BC detection and is the preferred modality when it is available.
Management of menopausal symptoms. These findings also have implications for menopausal hormone therapy for patients with symptoms. Because HR-positive patients have an increased risk of late-onset recurrence, nonhormonal therapies should be considered as first-line therapy for patients with menopausal symptoms. If hormone therapy is being considered, providers and patients should use shared decision making to balance the potential benefits (reduction in symptoms, possible cardiovascular benefits, and reduction in bone loss) with the risks (increased risk of recurrence and venous thromboembolism), even if patients are remote from the original diagnosis (ie, 10-year disease-free survival).
Topical estrogen therapies would be preferred for patients with significant urogenital atrophic symptoms who fail nonhormonal therapies due to substantially less systemic absorption and the lack of need to add a progestin.9,10 If oral therapy is being considered, I carefully counsel these women about the likely increased risk of recurrence and, if possible, include their breast oncologist in the discussion. ●
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33. doi: 10.3322/caac.21654.
- de Moor JS, Mariotto AB, Parry C, et al. Cancer survivors in the United States: prevalence across the survivorship trajectory and implications for care. Cancer Epidemiol Biomarkers Prev. 2013;22:561- 570. doi: 10.1158/1055-9965.EPI-12-1356.
- Carreira H, Williams R, Funston G, et al. Associations between breast cancer survivorship and adverse mental health outcomes: a matched population-based cohort study in the United Kingdom. PLOS Med. 2021;18:e1003504. doi: 10.1371/journal.pmed.1003504.
- Pedersen RN, Esen BÖ, Mellemkjær L, et al. The incidence of breast cancer recurrence 10-32 years after primary diagnosis. J Natl Cancer Inst. November 8, 2021. doi: 10.1093/jnci/djab202.
- Pan H, Gray R, Braybrooke J, et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377:1836- 1846. doi: 10.1056/NEJMoa1701830.
- Davies C, Pan H, Godwin J, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. doi: 10.1016/S0140-6736(12)61963-1.
- Scott LC, Mobley LR, Kuo TM, et al. Update on triple‐negative breast cancer disparities for the United States: a population‐based study from the United States Cancer Statistics database, 2010 through 2014. Cancer. 2019;125:3412-3417. doi: 10.1002/cncr.32207.
- NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. 2021; Version 2.2022.
- Crandall CJ, Diamant A, Santoro N. Safety of vaginal estrogens: a systematic review. Menopause. 2020;27:339-360. doi: 10.1097 /GME.0000000000001468.
- Treatment of urogenital symptoms in individuals with a history of estrogen-dependent breast cancer: clinical consensus. Obstet Gynecol. 2021;138:950-960. doi: 10.1097/AOG .0000000000004601.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7-33. doi: 10.3322/caac.21654.
- de Moor JS, Mariotto AB, Parry C, et al. Cancer survivors in the United States: prevalence across the survivorship trajectory and implications for care. Cancer Epidemiol Biomarkers Prev. 2013;22:561- 570. doi: 10.1158/1055-9965.EPI-12-1356.
- Carreira H, Williams R, Funston G, et al. Associations between breast cancer survivorship and adverse mental health outcomes: a matched population-based cohort study in the United Kingdom. PLOS Med. 2021;18:e1003504. doi: 10.1371/journal.pmed.1003504.
- Pedersen RN, Esen BÖ, Mellemkjær L, et al. The incidence of breast cancer recurrence 10-32 years after primary diagnosis. J Natl Cancer Inst. November 8, 2021. doi: 10.1093/jnci/djab202.
- Pan H, Gray R, Braybrooke J, et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377:1836- 1846. doi: 10.1056/NEJMoa1701830.
- Davies C, Pan H, Godwin J, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. doi: 10.1016/S0140-6736(12)61963-1.
- Scott LC, Mobley LR, Kuo TM, et al. Update on triple‐negative breast cancer disparities for the United States: a population‐based study from the United States Cancer Statistics database, 2010 through 2014. Cancer. 2019;125:3412-3417. doi: 10.1002/cncr.32207.
- NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. 2021; Version 2.2022.
- Crandall CJ, Diamant A, Santoro N. Safety of vaginal estrogens: a systematic review. Menopause. 2020;27:339-360. doi: 10.1097 /GME.0000000000001468.
- Treatment of urogenital symptoms in individuals with a history of estrogen-dependent breast cancer: clinical consensus. Obstet Gynecol. 2021;138:950-960. doi: 10.1097/AOG .0000000000004601.
Growth hormone therapy for certain children may help them reach their potential
“Dr. Lilley, you’ll always be my favorite doctor; you helped me grow.”
These were the parting words from the last patient that I treated during my endocrinology fellowship. I had watched this young man grow from a prepubertal 17-year-old to a young man who had reached his predicted family height as I treated his delayed puberty caused by Kallmann syndrome, a problem that had been missed for years. It was the appropriate bookend for my chosen specialty.
Watching children grow and develop into who they were meant to be is one of my favorite things about endocrinology, as well as forming meaningful relationships with families. Treating detectable deficiencies in logical and measurable ways is also extremely satisfying.
Too little testosterone? That’s a problem I can solve. Too much thyroid hormone? There’s a blocker for that! Endocrinology can be a straightforward field, and when all goes well, everyone leaves happy.
Except when they don’t.
Gatekeepers for treatment for children’s growth
“Nice to meet you. We’re here to get growth hormone.”
“We’re here because his pediatrician made us come. We’ve already decided we’re not going to put hormones into his body.”
These are common statements I hear when I first meet new patients whose parents are concerned about their children’s growth. Pediatric endocrinologists, after all, are the usual gatekeepers for this treatment.
Growth hormone (GH) often makes the news for controversial reasons – most commonly for its abuse by elite athletes hoping to exploit its anabolic effects – causing parents to have varied opinions about its possible use in their children.
Some refuse endocrinology referrals at all owing to concerns that we will push daily injections on their children. Others demand referrals for their children of average height, hoping for every perceived advantage.
GH deficiency (GHD) – a condition where the pituitary gland fails to produce enough GH – can occur because of congenital pituitary malformations; anatomic, surgical, or traumatic interruptions to the gland; or enzyme deficiencies leading to faulty production.
GHD is just one reason for poor growth, however.
Growth is one of the most important indicators of health in children. A waning growth rate may be an early symptom of serious problems. In my clinic, I’ve diagnosed severe hypothyroidism in a marathon runner, a brain tumor, celiac disease in a teenager with no gastrointestinal complaints, autoimmune hepatitis, and several other diseases needing treatment in children who show no symptoms other than poor growth.
Barriers to normal growth
Sometimes, the die is cast for children to have barriers to normal growth. Several genetic conditions can lead to poor GH production or response, and GH treatment is often necessary to approximate normal height.
These may include:
- Turner syndrome (in females who are missing an X chromosome in whole or part) should be considered in every girl with abnormally short stature; mosaic forms of the condition may be subtle and lack classic features.
- Noonan syndrome is important to detect owing to the possibility of cardiac or renal malformations that may also occur in this condition, caused by a mutation in one of the genes in the RAS-MAPK pathway.
- Russell-Silver syndrome can cause intrauterine GH restriction and has been traced to uniparental disomy of chromosome 7 or duplications, mutations, or methylation defects in chromosome 11.
- Individuals with Prader-Willi syndrome, which is characterized by low muscle tone, hyperphagia, and hypogonadism, have demonstrated dramatic benefits from GH therapy, primarily in maintaining a normal body mass index.
Children who are small for their gestational age may be GH resistant, and those who do not catch up to their growth curve by the age of 2 years may require GH treatment to reach their height potential.
GH therapy isn’t entirely benign. Rare adverse effects of overtreatment can include slipped capital femoral epiphysis (a fracture to the growth plate) and pseudotumor cerebri (idiopathic intracranial hypertension).
Overtreatment can cause acromegaly, which results in coarsened features and large hands and feet.
When is GH therapy warranted?
“Growth hormone therapy has been denied by her insurer. They want you to fill out an appeal.”
Insurance approval in the United States can be a herculean effort because GH is expensive: Out-of-pocket costs are prohibitive for most people without insurance assistance, ranging from $7,000 to $30,000 annually.
Pediatric endocrinologists aren’t in the business of cosmetic endocrinology. Treatment of idiopathic short stature has been controversial since this became an indication for GH treatment.
GH isn’t always necessary. Diagnosing the underlying cause for poor growth is the most important step.
Often, we find constitutional delay of growth and puberty, or “late bloomers.” This condition is characterized by a delayed bone age (growth plates more open than expected for age) and delayed pubertal onset. These children will often reach a normal height despite starting as some of the smallest of their peers.
However, GH plays other roles in the body than simply propelling height. Children with congenital GHD will require GH treatment to prevent hypoglycemia, especially in infancy.
GH is needed even in adults with fused growth plates for normal lipid metabolism, bone accrual, and maintaining normal muscle mass.
I have noticed marked improvements in muscle tone in many children with developmental delays who are treated with GH, and research supports cognitive benefits for certain populations.
The most common regimens for GH focus on treatment via subcutaneous injection nightly, when GH is naturally produced; sometimes, injections are given six nights out of seven to provide a break or for splitting time between households.
Newer once-weekly formulations have recently received approval, as reported by this news organization, and are coming into use.
Pediatric endocrinologists measure height and follow growth factors closely with visits every 3-6 months. GH levels are not useful outside of provocative diagnostic (stimulation) testing.
Insulinlike growth factor 1 or insulinlike growth factor binding protein levels are analyzed per Tanner stage of puberty to assess appropriate response and to make dose adjustments.
Annual standardized films of the left hand help predict progress and anticipated adult height. Treatment usually persists through puberty until growth plates are closed; if true GHD is noticed, much smaller doses are continued through adulthood.
Regardless, conversations about GH happen with your friendly local pediatric endocrinologist.
We are thrilled to help shepherd patients through their growing age to meet their potential. For more information about GH treatment for children, the MAGIC Foundation is the perfect place to start.
Dr. Lilley is director of the pediatric diabetes and lipid program, Mississippi Center for Advanced Medicine, Madison. She disclosed no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
“Dr. Lilley, you’ll always be my favorite doctor; you helped me grow.”
These were the parting words from the last patient that I treated during my endocrinology fellowship. I had watched this young man grow from a prepubertal 17-year-old to a young man who had reached his predicted family height as I treated his delayed puberty caused by Kallmann syndrome, a problem that had been missed for years. It was the appropriate bookend for my chosen specialty.
Watching children grow and develop into who they were meant to be is one of my favorite things about endocrinology, as well as forming meaningful relationships with families. Treating detectable deficiencies in logical and measurable ways is also extremely satisfying.
Too little testosterone? That’s a problem I can solve. Too much thyroid hormone? There’s a blocker for that! Endocrinology can be a straightforward field, and when all goes well, everyone leaves happy.
Except when they don’t.
Gatekeepers for treatment for children’s growth
“Nice to meet you. We’re here to get growth hormone.”
“We’re here because his pediatrician made us come. We’ve already decided we’re not going to put hormones into his body.”
These are common statements I hear when I first meet new patients whose parents are concerned about their children’s growth. Pediatric endocrinologists, after all, are the usual gatekeepers for this treatment.
Growth hormone (GH) often makes the news for controversial reasons – most commonly for its abuse by elite athletes hoping to exploit its anabolic effects – causing parents to have varied opinions about its possible use in their children.
Some refuse endocrinology referrals at all owing to concerns that we will push daily injections on their children. Others demand referrals for their children of average height, hoping for every perceived advantage.
GH deficiency (GHD) – a condition where the pituitary gland fails to produce enough GH – can occur because of congenital pituitary malformations; anatomic, surgical, or traumatic interruptions to the gland; or enzyme deficiencies leading to faulty production.
GHD is just one reason for poor growth, however.
Growth is one of the most important indicators of health in children. A waning growth rate may be an early symptom of serious problems. In my clinic, I’ve diagnosed severe hypothyroidism in a marathon runner, a brain tumor, celiac disease in a teenager with no gastrointestinal complaints, autoimmune hepatitis, and several other diseases needing treatment in children who show no symptoms other than poor growth.
Barriers to normal growth
Sometimes, the die is cast for children to have barriers to normal growth. Several genetic conditions can lead to poor GH production or response, and GH treatment is often necessary to approximate normal height.
These may include:
- Turner syndrome (in females who are missing an X chromosome in whole or part) should be considered in every girl with abnormally short stature; mosaic forms of the condition may be subtle and lack classic features.
- Noonan syndrome is important to detect owing to the possibility of cardiac or renal malformations that may also occur in this condition, caused by a mutation in one of the genes in the RAS-MAPK pathway.
- Russell-Silver syndrome can cause intrauterine GH restriction and has been traced to uniparental disomy of chromosome 7 or duplications, mutations, or methylation defects in chromosome 11.
- Individuals with Prader-Willi syndrome, which is characterized by low muscle tone, hyperphagia, and hypogonadism, have demonstrated dramatic benefits from GH therapy, primarily in maintaining a normal body mass index.
Children who are small for their gestational age may be GH resistant, and those who do not catch up to their growth curve by the age of 2 years may require GH treatment to reach their height potential.
GH therapy isn’t entirely benign. Rare adverse effects of overtreatment can include slipped capital femoral epiphysis (a fracture to the growth plate) and pseudotumor cerebri (idiopathic intracranial hypertension).
Overtreatment can cause acromegaly, which results in coarsened features and large hands and feet.
When is GH therapy warranted?
“Growth hormone therapy has been denied by her insurer. They want you to fill out an appeal.”
Insurance approval in the United States can be a herculean effort because GH is expensive: Out-of-pocket costs are prohibitive for most people without insurance assistance, ranging from $7,000 to $30,000 annually.
Pediatric endocrinologists aren’t in the business of cosmetic endocrinology. Treatment of idiopathic short stature has been controversial since this became an indication for GH treatment.
GH isn’t always necessary. Diagnosing the underlying cause for poor growth is the most important step.
Often, we find constitutional delay of growth and puberty, or “late bloomers.” This condition is characterized by a delayed bone age (growth plates more open than expected for age) and delayed pubertal onset. These children will often reach a normal height despite starting as some of the smallest of their peers.
However, GH plays other roles in the body than simply propelling height. Children with congenital GHD will require GH treatment to prevent hypoglycemia, especially in infancy.
GH is needed even in adults with fused growth plates for normal lipid metabolism, bone accrual, and maintaining normal muscle mass.
I have noticed marked improvements in muscle tone in many children with developmental delays who are treated with GH, and research supports cognitive benefits for certain populations.
The most common regimens for GH focus on treatment via subcutaneous injection nightly, when GH is naturally produced; sometimes, injections are given six nights out of seven to provide a break or for splitting time between households.
Newer once-weekly formulations have recently received approval, as reported by this news organization, and are coming into use.
Pediatric endocrinologists measure height and follow growth factors closely with visits every 3-6 months. GH levels are not useful outside of provocative diagnostic (stimulation) testing.
Insulinlike growth factor 1 or insulinlike growth factor binding protein levels are analyzed per Tanner stage of puberty to assess appropriate response and to make dose adjustments.
Annual standardized films of the left hand help predict progress and anticipated adult height. Treatment usually persists through puberty until growth plates are closed; if true GHD is noticed, much smaller doses are continued through adulthood.
Regardless, conversations about GH happen with your friendly local pediatric endocrinologist.
We are thrilled to help shepherd patients through their growing age to meet their potential. For more information about GH treatment for children, the MAGIC Foundation is the perfect place to start.
Dr. Lilley is director of the pediatric diabetes and lipid program, Mississippi Center for Advanced Medicine, Madison. She disclosed no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
“Dr. Lilley, you’ll always be my favorite doctor; you helped me grow.”
These were the parting words from the last patient that I treated during my endocrinology fellowship. I had watched this young man grow from a prepubertal 17-year-old to a young man who had reached his predicted family height as I treated his delayed puberty caused by Kallmann syndrome, a problem that had been missed for years. It was the appropriate bookend for my chosen specialty.
Watching children grow and develop into who they were meant to be is one of my favorite things about endocrinology, as well as forming meaningful relationships with families. Treating detectable deficiencies in logical and measurable ways is also extremely satisfying.
Too little testosterone? That’s a problem I can solve. Too much thyroid hormone? There’s a blocker for that! Endocrinology can be a straightforward field, and when all goes well, everyone leaves happy.
Except when they don’t.
Gatekeepers for treatment for children’s growth
“Nice to meet you. We’re here to get growth hormone.”
“We’re here because his pediatrician made us come. We’ve already decided we’re not going to put hormones into his body.”
These are common statements I hear when I first meet new patients whose parents are concerned about their children’s growth. Pediatric endocrinologists, after all, are the usual gatekeepers for this treatment.
Growth hormone (GH) often makes the news for controversial reasons – most commonly for its abuse by elite athletes hoping to exploit its anabolic effects – causing parents to have varied opinions about its possible use in their children.
Some refuse endocrinology referrals at all owing to concerns that we will push daily injections on their children. Others demand referrals for their children of average height, hoping for every perceived advantage.
GH deficiency (GHD) – a condition where the pituitary gland fails to produce enough GH – can occur because of congenital pituitary malformations; anatomic, surgical, or traumatic interruptions to the gland; or enzyme deficiencies leading to faulty production.
GHD is just one reason for poor growth, however.
Growth is one of the most important indicators of health in children. A waning growth rate may be an early symptom of serious problems. In my clinic, I’ve diagnosed severe hypothyroidism in a marathon runner, a brain tumor, celiac disease in a teenager with no gastrointestinal complaints, autoimmune hepatitis, and several other diseases needing treatment in children who show no symptoms other than poor growth.
Barriers to normal growth
Sometimes, the die is cast for children to have barriers to normal growth. Several genetic conditions can lead to poor GH production or response, and GH treatment is often necessary to approximate normal height.
These may include:
- Turner syndrome (in females who are missing an X chromosome in whole or part) should be considered in every girl with abnormally short stature; mosaic forms of the condition may be subtle and lack classic features.
- Noonan syndrome is important to detect owing to the possibility of cardiac or renal malformations that may also occur in this condition, caused by a mutation in one of the genes in the RAS-MAPK pathway.
- Russell-Silver syndrome can cause intrauterine GH restriction and has been traced to uniparental disomy of chromosome 7 or duplications, mutations, or methylation defects in chromosome 11.
- Individuals with Prader-Willi syndrome, which is characterized by low muscle tone, hyperphagia, and hypogonadism, have demonstrated dramatic benefits from GH therapy, primarily in maintaining a normal body mass index.
Children who are small for their gestational age may be GH resistant, and those who do not catch up to their growth curve by the age of 2 years may require GH treatment to reach their height potential.
GH therapy isn’t entirely benign. Rare adverse effects of overtreatment can include slipped capital femoral epiphysis (a fracture to the growth plate) and pseudotumor cerebri (idiopathic intracranial hypertension).
Overtreatment can cause acromegaly, which results in coarsened features and large hands and feet.
When is GH therapy warranted?
“Growth hormone therapy has been denied by her insurer. They want you to fill out an appeal.”
Insurance approval in the United States can be a herculean effort because GH is expensive: Out-of-pocket costs are prohibitive for most people without insurance assistance, ranging from $7,000 to $30,000 annually.
Pediatric endocrinologists aren’t in the business of cosmetic endocrinology. Treatment of idiopathic short stature has been controversial since this became an indication for GH treatment.
GH isn’t always necessary. Diagnosing the underlying cause for poor growth is the most important step.
Often, we find constitutional delay of growth and puberty, or “late bloomers.” This condition is characterized by a delayed bone age (growth plates more open than expected for age) and delayed pubertal onset. These children will often reach a normal height despite starting as some of the smallest of their peers.
However, GH plays other roles in the body than simply propelling height. Children with congenital GHD will require GH treatment to prevent hypoglycemia, especially in infancy.
GH is needed even in adults with fused growth plates for normal lipid metabolism, bone accrual, and maintaining normal muscle mass.
I have noticed marked improvements in muscle tone in many children with developmental delays who are treated with GH, and research supports cognitive benefits for certain populations.
The most common regimens for GH focus on treatment via subcutaneous injection nightly, when GH is naturally produced; sometimes, injections are given six nights out of seven to provide a break or for splitting time between households.
Newer once-weekly formulations have recently received approval, as reported by this news organization, and are coming into use.
Pediatric endocrinologists measure height and follow growth factors closely with visits every 3-6 months. GH levels are not useful outside of provocative diagnostic (stimulation) testing.
Insulinlike growth factor 1 or insulinlike growth factor binding protein levels are analyzed per Tanner stage of puberty to assess appropriate response and to make dose adjustments.
Annual standardized films of the left hand help predict progress and anticipated adult height. Treatment usually persists through puberty until growth plates are closed; if true GHD is noticed, much smaller doses are continued through adulthood.
Regardless, conversations about GH happen with your friendly local pediatric endocrinologist.
We are thrilled to help shepherd patients through their growing age to meet their potential. For more information about GH treatment for children, the MAGIC Foundation is the perfect place to start.
Dr. Lilley is director of the pediatric diabetes and lipid program, Mississippi Center for Advanced Medicine, Madison. She disclosed no relevant conflicts of interest. A version of this article first appeared on Medscape.com.