User login
Blind optimism only works in fantasy football. Time to get realistic
In the age of COVID, what exactly does it mean to be optimistic? I get this question quite a bit from virtually everyone I meet in one form or another through my work with the George Washington Resiliency and Well-Being Center in Washington, D.C. Giving a lecture on resilience and staying positive can be a significant challenge. Especially when we wake up to the news that 1 of every 100 older Americans has died secondary to COVID. The mind doesn’t really know how to process this type of loss. It is hard to maintain any form of a positive attitude when you’re still struggling just to accept the magnitude of what humanity has experienced over the past 2 years.
In “Resilience: The Science of Mastering Life’s Greatest Challenges,” (Cambridge, England: Cambridge University Press, 2018), Steven M. Southwick, MD, and Dennis S. Charney, MD, identify 10 critical factors associated with very resilient individuals. The authors based their work on science, personal experience, and interviews of people who have literally been through hell and back. One of the critical factors they identified is optimism.
“Optimism ignites resilience, providing energy to power the other resilience factors. It facilitates an active and creative approach to coping with challenging situations.”
Dr. Southwick and Dr. Charney are a lot smarter than me and far more patient to weave all this data together into a coherent story about optimism. Sounds like a damn good factor to focus a lecture on in my book! Slight problem: In my experience, many health professionals are already expert optimists. They literally eat, sleep, and sincerely breathe this stuff. So if we are going to talk about optimism, then we need to discuss realistic optimism.
How does realistic optimism differ from, say, blind optimism? Dr. Southwick and Dr. Charney’s review of the literature points to three features worthy of highlighting.
Realistic vs. blind optimism: Take-home points
- In realistic optimism, we notice the negative but don’t stay engaged with it. Realistic optimists moved on from problems that were not solvable.
- Blind optimism can lead to underestimating risk, overestimating abilities, and inadequate preparation.
Growing up in northeast Ohio, I can absolutely embrace the concept of realistic optimism. It’s overcast in Cleveland 8 months out of the year. To hope for 3 sunny days in a row in April is genuinely a fools’ errand. So you learn over time, the sun will shine; you just have to at times wait 3-4 months for it to occur.
From a skill perspective, realistic optimism could be conceptualized as a great mix of radical acceptance, emotion regulation, and focused problem solving. This is all fine, but to be realistically optimistic, we must first stop wishing for a better tomorrow. You may say, I don’t wish for or see rainbows and unicorns, et cetera, et cetera. Okay, so you don’t verbalize your wishes, but on a small level, you may engage in wishful thinking. Here are a few wishful thoughts that I would daydream about, which were not realistically optimistic at various points:
- “Once we get enough COVID-19 tests, things will improve.”
- “All we need to do is get vaccines, and then the new normal is right there.”
- “Once everyone gets the booster, then we got this thing beat.”
At this point, you could argue that I was engaged in blind optimism. I consider the above statements blind for a couple of reasons. They weren’t balanced (both positive and negative), didn’t have a clear definition of the outcome, and were more focused on external events I couldn’t control. These statements were the equivalent of wishes, and I don’t have a magic lamp with a genie, so I need to let go of my wishful thinking first. Let me rephrase that: I need to forcefully toss it into the sea of COVID variants and start figuring out how I’m going to tread water for another 6-12 months. So with this in mind, here are my initial thoughts on ways to navigate the next year of the pandemic:
- A multilayered form of protection gives me the best chance to survive the next 6 months of the pandemic.
- It will take time, but I’ll process the loss associated with a workplace that will never be the same.
- Until we have positivity test rates lower than 2% across the globe, COVID will remain a substantial disruption to humanity.
- I can’t bring back missed graduation or the first day of school, but I can share ways that I’ve countered and survived loneliness in my life with my children.
Okay, this is the starting point – hopefully not pessimistic, or blindly optimistic, just realistic. Now I can address other important topics, such as planning to rebuild my disappointing fantasy football team. I was No. 1 in our GW department of psychiatry fantasy football league until my star running back Derrick Henry went down. My residents will become attendings and still give me grief about this for many years to follow, and that is a very good thing.
Everyone be well and safe.
Dr. Norris is associate dean of student affairs; associate professor, department of psychiatry, George Washington University; chief wellness officer, GW Hospital, GW Medical Faculty Associates, and the GW School of Medicine and Health Sciences (GWU Medical Enterprise), Washington. He has disclosed having no relevant financial relationships. A version of this article first appeared on Medscape.com.
In the age of COVID, what exactly does it mean to be optimistic? I get this question quite a bit from virtually everyone I meet in one form or another through my work with the George Washington Resiliency and Well-Being Center in Washington, D.C. Giving a lecture on resilience and staying positive can be a significant challenge. Especially when we wake up to the news that 1 of every 100 older Americans has died secondary to COVID. The mind doesn’t really know how to process this type of loss. It is hard to maintain any form of a positive attitude when you’re still struggling just to accept the magnitude of what humanity has experienced over the past 2 years.
In “Resilience: The Science of Mastering Life’s Greatest Challenges,” (Cambridge, England: Cambridge University Press, 2018), Steven M. Southwick, MD, and Dennis S. Charney, MD, identify 10 critical factors associated with very resilient individuals. The authors based their work on science, personal experience, and interviews of people who have literally been through hell and back. One of the critical factors they identified is optimism.
“Optimism ignites resilience, providing energy to power the other resilience factors. It facilitates an active and creative approach to coping with challenging situations.”
Dr. Southwick and Dr. Charney are a lot smarter than me and far more patient to weave all this data together into a coherent story about optimism. Sounds like a damn good factor to focus a lecture on in my book! Slight problem: In my experience, many health professionals are already expert optimists. They literally eat, sleep, and sincerely breathe this stuff. So if we are going to talk about optimism, then we need to discuss realistic optimism.
How does realistic optimism differ from, say, blind optimism? Dr. Southwick and Dr. Charney’s review of the literature points to three features worthy of highlighting.
Realistic vs. blind optimism: Take-home points
- In realistic optimism, we notice the negative but don’t stay engaged with it. Realistic optimists moved on from problems that were not solvable.
- Blind optimism can lead to underestimating risk, overestimating abilities, and inadequate preparation.
Growing up in northeast Ohio, I can absolutely embrace the concept of realistic optimism. It’s overcast in Cleveland 8 months out of the year. To hope for 3 sunny days in a row in April is genuinely a fools’ errand. So you learn over time, the sun will shine; you just have to at times wait 3-4 months for it to occur.
From a skill perspective, realistic optimism could be conceptualized as a great mix of radical acceptance, emotion regulation, and focused problem solving. This is all fine, but to be realistically optimistic, we must first stop wishing for a better tomorrow. You may say, I don’t wish for or see rainbows and unicorns, et cetera, et cetera. Okay, so you don’t verbalize your wishes, but on a small level, you may engage in wishful thinking. Here are a few wishful thoughts that I would daydream about, which were not realistically optimistic at various points:
- “Once we get enough COVID-19 tests, things will improve.”
- “All we need to do is get vaccines, and then the new normal is right there.”
- “Once everyone gets the booster, then we got this thing beat.”
At this point, you could argue that I was engaged in blind optimism. I consider the above statements blind for a couple of reasons. They weren’t balanced (both positive and negative), didn’t have a clear definition of the outcome, and were more focused on external events I couldn’t control. These statements were the equivalent of wishes, and I don’t have a magic lamp with a genie, so I need to let go of my wishful thinking first. Let me rephrase that: I need to forcefully toss it into the sea of COVID variants and start figuring out how I’m going to tread water for another 6-12 months. So with this in mind, here are my initial thoughts on ways to navigate the next year of the pandemic:
- A multilayered form of protection gives me the best chance to survive the next 6 months of the pandemic.
- It will take time, but I’ll process the loss associated with a workplace that will never be the same.
- Until we have positivity test rates lower than 2% across the globe, COVID will remain a substantial disruption to humanity.
- I can’t bring back missed graduation or the first day of school, but I can share ways that I’ve countered and survived loneliness in my life with my children.
Okay, this is the starting point – hopefully not pessimistic, or blindly optimistic, just realistic. Now I can address other important topics, such as planning to rebuild my disappointing fantasy football team. I was No. 1 in our GW department of psychiatry fantasy football league until my star running back Derrick Henry went down. My residents will become attendings and still give me grief about this for many years to follow, and that is a very good thing.
Everyone be well and safe.
Dr. Norris is associate dean of student affairs; associate professor, department of psychiatry, George Washington University; chief wellness officer, GW Hospital, GW Medical Faculty Associates, and the GW School of Medicine and Health Sciences (GWU Medical Enterprise), Washington. He has disclosed having no relevant financial relationships. A version of this article first appeared on Medscape.com.
In the age of COVID, what exactly does it mean to be optimistic? I get this question quite a bit from virtually everyone I meet in one form or another through my work with the George Washington Resiliency and Well-Being Center in Washington, D.C. Giving a lecture on resilience and staying positive can be a significant challenge. Especially when we wake up to the news that 1 of every 100 older Americans has died secondary to COVID. The mind doesn’t really know how to process this type of loss. It is hard to maintain any form of a positive attitude when you’re still struggling just to accept the magnitude of what humanity has experienced over the past 2 years.
In “Resilience: The Science of Mastering Life’s Greatest Challenges,” (Cambridge, England: Cambridge University Press, 2018), Steven M. Southwick, MD, and Dennis S. Charney, MD, identify 10 critical factors associated with very resilient individuals. The authors based their work on science, personal experience, and interviews of people who have literally been through hell and back. One of the critical factors they identified is optimism.
“Optimism ignites resilience, providing energy to power the other resilience factors. It facilitates an active and creative approach to coping with challenging situations.”
Dr. Southwick and Dr. Charney are a lot smarter than me and far more patient to weave all this data together into a coherent story about optimism. Sounds like a damn good factor to focus a lecture on in my book! Slight problem: In my experience, many health professionals are already expert optimists. They literally eat, sleep, and sincerely breathe this stuff. So if we are going to talk about optimism, then we need to discuss realistic optimism.
How does realistic optimism differ from, say, blind optimism? Dr. Southwick and Dr. Charney’s review of the literature points to three features worthy of highlighting.
Realistic vs. blind optimism: Take-home points
- In realistic optimism, we notice the negative but don’t stay engaged with it. Realistic optimists moved on from problems that were not solvable.
- Blind optimism can lead to underestimating risk, overestimating abilities, and inadequate preparation.
Growing up in northeast Ohio, I can absolutely embrace the concept of realistic optimism. It’s overcast in Cleveland 8 months out of the year. To hope for 3 sunny days in a row in April is genuinely a fools’ errand. So you learn over time, the sun will shine; you just have to at times wait 3-4 months for it to occur.
From a skill perspective, realistic optimism could be conceptualized as a great mix of radical acceptance, emotion regulation, and focused problem solving. This is all fine, but to be realistically optimistic, we must first stop wishing for a better tomorrow. You may say, I don’t wish for or see rainbows and unicorns, et cetera, et cetera. Okay, so you don’t verbalize your wishes, but on a small level, you may engage in wishful thinking. Here are a few wishful thoughts that I would daydream about, which were not realistically optimistic at various points:
- “Once we get enough COVID-19 tests, things will improve.”
- “All we need to do is get vaccines, and then the new normal is right there.”
- “Once everyone gets the booster, then we got this thing beat.”
At this point, you could argue that I was engaged in blind optimism. I consider the above statements blind for a couple of reasons. They weren’t balanced (both positive and negative), didn’t have a clear definition of the outcome, and were more focused on external events I couldn’t control. These statements were the equivalent of wishes, and I don’t have a magic lamp with a genie, so I need to let go of my wishful thinking first. Let me rephrase that: I need to forcefully toss it into the sea of COVID variants and start figuring out how I’m going to tread water for another 6-12 months. So with this in mind, here are my initial thoughts on ways to navigate the next year of the pandemic:
- A multilayered form of protection gives me the best chance to survive the next 6 months of the pandemic.
- It will take time, but I’ll process the loss associated with a workplace that will never be the same.
- Until we have positivity test rates lower than 2% across the globe, COVID will remain a substantial disruption to humanity.
- I can’t bring back missed graduation or the first day of school, but I can share ways that I’ve countered and survived loneliness in my life with my children.
Okay, this is the starting point – hopefully not pessimistic, or blindly optimistic, just realistic. Now I can address other important topics, such as planning to rebuild my disappointing fantasy football team. I was No. 1 in our GW department of psychiatry fantasy football league until my star running back Derrick Henry went down. My residents will become attendings and still give me grief about this for many years to follow, and that is a very good thing.
Everyone be well and safe.
Dr. Norris is associate dean of student affairs; associate professor, department of psychiatry, George Washington University; chief wellness officer, GW Hospital, GW Medical Faculty Associates, and the GW School of Medicine and Health Sciences (GWU Medical Enterprise), Washington. He has disclosed having no relevant financial relationships. A version of this article first appeared on Medscape.com.
Treatment duration for acute otitis media – so many choices
Twenty years ago, the dilemma in treating acute otitis media (AOM) was which among 10-plus antibiotics to prescribe. A recent column discussed the evolving pathogen distribution in AOM and its effects on antibiotic choices.1 But here we consider treatment duration. Until the past decade, AOM treatment (except azithromycin) involved 10-day courses. But lately, 10-day antibiotic regimens for uncomplicated infections are disappearing. Shorter-course recommendations are the new norm because of the evolving clinical data showing that an appropriately chosen antibiotic (in partnership with host defenses and source control) resolves infection faster than was previously thought. Shorter courses make sense because of fewer adverse effects, less distortion of normal flora, and less likely induction of pathogen resistance. Table 4.12 in the newest 2021-2024 SOID Redbook lists three antibiotic durations for AOM, and actually there are more than that.
Why so many duration options? Clinical data show that not all AOM is alike and short courses work for subsets of AOM because, besides antibiotics, key elements in AOM resolution are host anatomy and immunity. Bacterial AOM results from a combination of refluxed pathogens in the middle ear being trapped when the eustachian tube malfunctions (infection occurs when middle ear plumbing gets stopped up). If the eustachian tube spontaneously drains and the host immune response slows/stops pathogen growth, no antibiotics are needed. Indeed, a sizable proportion of mild/moderate AOM episodes spontaneously resolve, particularly in children over 2 years old. So a high likelihood of spontaneous remission allows an initial 0-days duration option (watchful waiting) or delayed antibiotics (rescue prescriptions) for older children.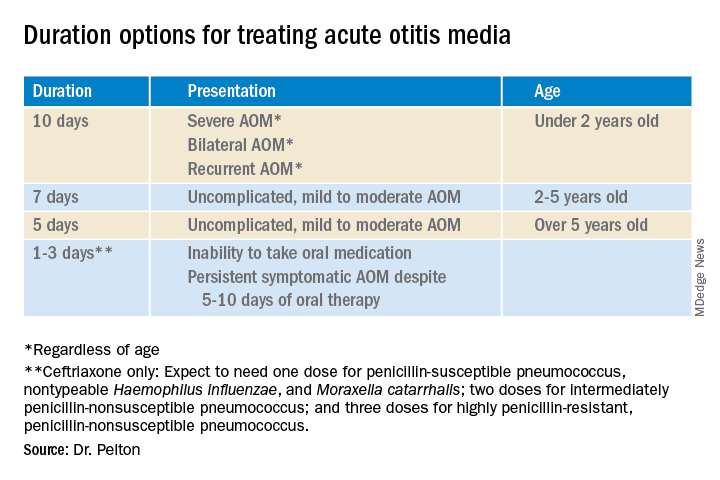
That said, when one chooses to initially prescribe antibiotics for AOM, different durations are recommended. Table 1 has my suggestions.
Data that gave me better microbiological understanding of why oral AOM trials less than 10 days were successful involved purulent AOM drainage from children who had pressure-equalizing (PE) tubes.2 The authors randomized children to either standard-dose amoxicillin-clavulanate or placebo. Of note, 95% of pathogens were susceptible to the antibiotic; 5% were pneumococcus intermediately resistant to penicillin. The authors sampled ear drainage daily for 7 days. Figure 1 shows that cultures remained positive in only around 5% of children by day 3-5 of antibiotics, but viable bacteria persisted through 7 days in over half of placebo recipients. Remember, both groups benefited from a form of source control (drainage of the middle ear via PE tubes). So, if antibiotics can do the job in 3-5 days, why continue antibiotics beyond 5 days?
Anatomy and severity. In children over 5 years old (reasonably mature eustachian tube anatomy) with nonrecurrent (no AOM in past month), nonsevere (no otalgia or high fever) AOM, 5 days is enough. But 2- to 5-year-olds (less mature anatomy) need 7 days and those <2 years old (least mature plumbing) need 10 days. Likewise, severe AOM usually warrants 10 days. Some experts recommend 10 days for bilateral AOM as well.
These age/severity differences make sense because failures are more frequent with:
1. Younger age.3 While not proven, my hypothesis is that “natural” source control (spontaneous internal draining the middle ear into the nasopharynx [NP]) is less frequent in younger children because they have less mature eustachian tube systems. Further, reflux of persisting NP organisms could restart a new AOM episode even if the original pathogen was eliminated by a short 5-day course.
2. Severe AOM. A rationale for longer courses in severe AOM (ear pain, high fever) is that high middle-ear pressures (indicated by degree of tympanic membrane bulging and ear pain) could impede antibiotic penetration, or that high initial bacterial loads (perhaps indicated by systemic fever) require more antibiotic. And finally, return to baseline eustachian tube function may take longer if severe AOM caused enhanced inflammation.
3. Recurrent AOM. (AOM within 1 prior month) – With recurrent AOM, the second “hit” to the eustachian tube may lead to more dysfunction, so a longer antibiotic course may be required to allow more complete source control and more time for more complete functional recovery after a repeated inflammatory injury.
4. Bilateral AOM. Two independent but infected sites mean twice the chance for failure. So, a longer course could allow more time for both sites to undergo “natural” source control.4
More bacteria – more antibiotic? So, is more antibiotic really needed for a higher bacterial load? In vitro this is known as the “inoculum effect,” particularly for beta-lactam drugs, for example, amoxicillin and cephalosporins. Laboratory susceptibility testing is performed with a specifically defined quantity of bacteria (105 bacteria/mL) and the minimum inhibitory concentration (MIC) is the lowest antibiotic concentration that stops bacterial growth. We know that drugs will likely fail if the MIC exceeds the achievable antibiotic concentration at the infection site. But is it as simple as just exceeding the MIC at the infection site? No, pharmacodynamics tell us that overall antibiotic exposure is also important. For example, to be successful, beta-lactam concentrations need to be above the MIC for 40%-50% of the day.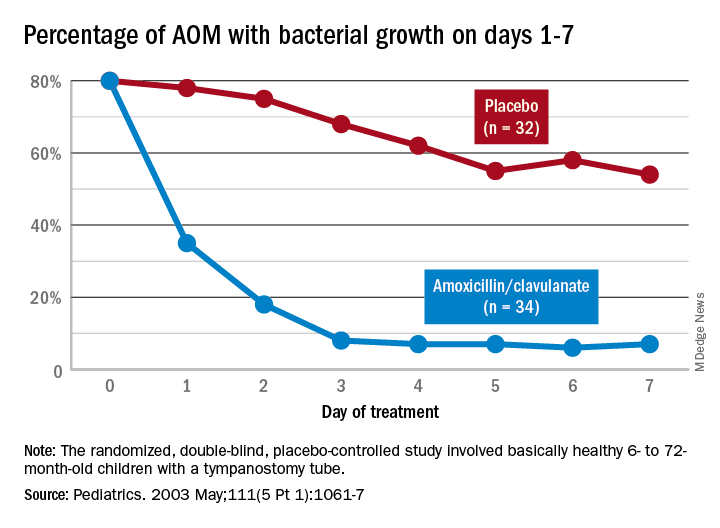
Higher MIC with higher bacterial load. Particularly for beta-lactams, testing with a quantity of bacteria >105/mL produces a higher MIC in vitro. This suggests that clinical failure could occur, even when our in vivo dosing leads to 40%-50% above the “standard” MIC that was obtained from testing the lab standard of 105/mL bacteria, when the infected site’s (middle ear) bacterial load is >105/mL (such higher bacterial loads occur in up to 30% of AOM).5 One way to negate inoculum effect is source control (drain the abscess or debridement), which reduces the bacterial load as well as allowing better antibiotic penetration– both favoring infection resolution. But with suboptimal source control, for example, the middle ear is not drained externally or internally, longer courses (more antibiotic exposure) could aid resolution. Whether the exposure can be administered as higher doses in fewer days or standard doses for more days is debatable but consider that a single parenteral dose of ceftriaxone successfully resolves AOM not attributable to penicillin-nonsusceptible pneumococcus.6Bottom line: Even though the number of potential antibiotics has contracted in the past 20 years, the need to individualize AOM treatment remains important and duration choices are more complex. Indeed, AOM comes in different flavors with patient age, clinical presentation, and episode frequency dictating the choice of duration.
Dr. Christopher J. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. Email him at [email protected].
References
1. Pichichero ME. MDedge. 2022 Jan 11.
2. Ruohola A et al. Pediatrics. 2003;111(5):1061-7.
3. Hoberman A et al. N Engl J Med. 2016;375(25):2446-56.
4. Pichichero ME et al. Otolaryngol Head Neck Surg. 2001;124(4):381-7.
5. Harrison CJ et al. Pediatr Infect Dis. 1985;4(6):641-6.
6. Leibovitz E et al. Pediatr Infect Dis. 2000;19(11):1040-5.
Twenty years ago, the dilemma in treating acute otitis media (AOM) was which among 10-plus antibiotics to prescribe. A recent column discussed the evolving pathogen distribution in AOM and its effects on antibiotic choices.1 But here we consider treatment duration. Until the past decade, AOM treatment (except azithromycin) involved 10-day courses. But lately, 10-day antibiotic regimens for uncomplicated infections are disappearing. Shorter-course recommendations are the new norm because of the evolving clinical data showing that an appropriately chosen antibiotic (in partnership with host defenses and source control) resolves infection faster than was previously thought. Shorter courses make sense because of fewer adverse effects, less distortion of normal flora, and less likely induction of pathogen resistance. Table 4.12 in the newest 2021-2024 SOID Redbook lists three antibiotic durations for AOM, and actually there are more than that.
Why so many duration options? Clinical data show that not all AOM is alike and short courses work for subsets of AOM because, besides antibiotics, key elements in AOM resolution are host anatomy and immunity. Bacterial AOM results from a combination of refluxed pathogens in the middle ear being trapped when the eustachian tube malfunctions (infection occurs when middle ear plumbing gets stopped up). If the eustachian tube spontaneously drains and the host immune response slows/stops pathogen growth, no antibiotics are needed. Indeed, a sizable proportion of mild/moderate AOM episodes spontaneously resolve, particularly in children over 2 years old. So a high likelihood of spontaneous remission allows an initial 0-days duration option (watchful waiting) or delayed antibiotics (rescue prescriptions) for older children.
That said, when one chooses to initially prescribe antibiotics for AOM, different durations are recommended. Table 1 has my suggestions.
Data that gave me better microbiological understanding of why oral AOM trials less than 10 days were successful involved purulent AOM drainage from children who had pressure-equalizing (PE) tubes.2 The authors randomized children to either standard-dose amoxicillin-clavulanate or placebo. Of note, 95% of pathogens were susceptible to the antibiotic; 5% were pneumococcus intermediately resistant to penicillin. The authors sampled ear drainage daily for 7 days. Figure 1 shows that cultures remained positive in only around 5% of children by day 3-5 of antibiotics, but viable bacteria persisted through 7 days in over half of placebo recipients. Remember, both groups benefited from a form of source control (drainage of the middle ear via PE tubes). So, if antibiotics can do the job in 3-5 days, why continue antibiotics beyond 5 days?
Anatomy and severity. In children over 5 years old (reasonably mature eustachian tube anatomy) with nonrecurrent (no AOM in past month), nonsevere (no otalgia or high fever) AOM, 5 days is enough. But 2- to 5-year-olds (less mature anatomy) need 7 days and those <2 years old (least mature plumbing) need 10 days. Likewise, severe AOM usually warrants 10 days. Some experts recommend 10 days for bilateral AOM as well.
These age/severity differences make sense because failures are more frequent with:
1. Younger age.3 While not proven, my hypothesis is that “natural” source control (spontaneous internal draining the middle ear into the nasopharynx [NP]) is less frequent in younger children because they have less mature eustachian tube systems. Further, reflux of persisting NP organisms could restart a new AOM episode even if the original pathogen was eliminated by a short 5-day course.
2. Severe AOM. A rationale for longer courses in severe AOM (ear pain, high fever) is that high middle-ear pressures (indicated by degree of tympanic membrane bulging and ear pain) could impede antibiotic penetration, or that high initial bacterial loads (perhaps indicated by systemic fever) require more antibiotic. And finally, return to baseline eustachian tube function may take longer if severe AOM caused enhanced inflammation.
3. Recurrent AOM. (AOM within 1 prior month) – With recurrent AOM, the second “hit” to the eustachian tube may lead to more dysfunction, so a longer antibiotic course may be required to allow more complete source control and more time for more complete functional recovery after a repeated inflammatory injury.
4. Bilateral AOM. Two independent but infected sites mean twice the chance for failure. So, a longer course could allow more time for both sites to undergo “natural” source control.4
More bacteria – more antibiotic? So, is more antibiotic really needed for a higher bacterial load? In vitro this is known as the “inoculum effect,” particularly for beta-lactam drugs, for example, amoxicillin and cephalosporins. Laboratory susceptibility testing is performed with a specifically defined quantity of bacteria (105 bacteria/mL) and the minimum inhibitory concentration (MIC) is the lowest antibiotic concentration that stops bacterial growth. We know that drugs will likely fail if the MIC exceeds the achievable antibiotic concentration at the infection site. But is it as simple as just exceeding the MIC at the infection site? No, pharmacodynamics tell us that overall antibiotic exposure is also important. For example, to be successful, beta-lactam concentrations need to be above the MIC for 40%-50% of the day.
Higher MIC with higher bacterial load. Particularly for beta-lactams, testing with a quantity of bacteria >105/mL produces a higher MIC in vitro. This suggests that clinical failure could occur, even when our in vivo dosing leads to 40%-50% above the “standard” MIC that was obtained from testing the lab standard of 105/mL bacteria, when the infected site’s (middle ear) bacterial load is >105/mL (such higher bacterial loads occur in up to 30% of AOM).5 One way to negate inoculum effect is source control (drain the abscess or debridement), which reduces the bacterial load as well as allowing better antibiotic penetration– both favoring infection resolution. But with suboptimal source control, for example, the middle ear is not drained externally or internally, longer courses (more antibiotic exposure) could aid resolution. Whether the exposure can be administered as higher doses in fewer days or standard doses for more days is debatable but consider that a single parenteral dose of ceftriaxone successfully resolves AOM not attributable to penicillin-nonsusceptible pneumococcus.6Bottom line: Even though the number of potential antibiotics has contracted in the past 20 years, the need to individualize AOM treatment remains important and duration choices are more complex. Indeed, AOM comes in different flavors with patient age, clinical presentation, and episode frequency dictating the choice of duration.
Dr. Christopher J. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. Email him at [email protected].
References
1. Pichichero ME. MDedge. 2022 Jan 11.
2. Ruohola A et al. Pediatrics. 2003;111(5):1061-7.
3. Hoberman A et al. N Engl J Med. 2016;375(25):2446-56.
4. Pichichero ME et al. Otolaryngol Head Neck Surg. 2001;124(4):381-7.
5. Harrison CJ et al. Pediatr Infect Dis. 1985;4(6):641-6.
6. Leibovitz E et al. Pediatr Infect Dis. 2000;19(11):1040-5.
Twenty years ago, the dilemma in treating acute otitis media (AOM) was which among 10-plus antibiotics to prescribe. A recent column discussed the evolving pathogen distribution in AOM and its effects on antibiotic choices.1 But here we consider treatment duration. Until the past decade, AOM treatment (except azithromycin) involved 10-day courses. But lately, 10-day antibiotic regimens for uncomplicated infections are disappearing. Shorter-course recommendations are the new norm because of the evolving clinical data showing that an appropriately chosen antibiotic (in partnership with host defenses and source control) resolves infection faster than was previously thought. Shorter courses make sense because of fewer adverse effects, less distortion of normal flora, and less likely induction of pathogen resistance. Table 4.12 in the newest 2021-2024 SOID Redbook lists three antibiotic durations for AOM, and actually there are more than that.
Why so many duration options? Clinical data show that not all AOM is alike and short courses work for subsets of AOM because, besides antibiotics, key elements in AOM resolution are host anatomy and immunity. Bacterial AOM results from a combination of refluxed pathogens in the middle ear being trapped when the eustachian tube malfunctions (infection occurs when middle ear plumbing gets stopped up). If the eustachian tube spontaneously drains and the host immune response slows/stops pathogen growth, no antibiotics are needed. Indeed, a sizable proportion of mild/moderate AOM episodes spontaneously resolve, particularly in children over 2 years old. So a high likelihood of spontaneous remission allows an initial 0-days duration option (watchful waiting) or delayed antibiotics (rescue prescriptions) for older children.
That said, when one chooses to initially prescribe antibiotics for AOM, different durations are recommended. Table 1 has my suggestions.
Data that gave me better microbiological understanding of why oral AOM trials less than 10 days were successful involved purulent AOM drainage from children who had pressure-equalizing (PE) tubes.2 The authors randomized children to either standard-dose amoxicillin-clavulanate or placebo. Of note, 95% of pathogens were susceptible to the antibiotic; 5% were pneumococcus intermediately resistant to penicillin. The authors sampled ear drainage daily for 7 days. Figure 1 shows that cultures remained positive in only around 5% of children by day 3-5 of antibiotics, but viable bacteria persisted through 7 days in over half of placebo recipients. Remember, both groups benefited from a form of source control (drainage of the middle ear via PE tubes). So, if antibiotics can do the job in 3-5 days, why continue antibiotics beyond 5 days?
Anatomy and severity. In children over 5 years old (reasonably mature eustachian tube anatomy) with nonrecurrent (no AOM in past month), nonsevere (no otalgia or high fever) AOM, 5 days is enough. But 2- to 5-year-olds (less mature anatomy) need 7 days and those <2 years old (least mature plumbing) need 10 days. Likewise, severe AOM usually warrants 10 days. Some experts recommend 10 days for bilateral AOM as well.
These age/severity differences make sense because failures are more frequent with:
1. Younger age.3 While not proven, my hypothesis is that “natural” source control (spontaneous internal draining the middle ear into the nasopharynx [NP]) is less frequent in younger children because they have less mature eustachian tube systems. Further, reflux of persisting NP organisms could restart a new AOM episode even if the original pathogen was eliminated by a short 5-day course.
2. Severe AOM. A rationale for longer courses in severe AOM (ear pain, high fever) is that high middle-ear pressures (indicated by degree of tympanic membrane bulging and ear pain) could impede antibiotic penetration, or that high initial bacterial loads (perhaps indicated by systemic fever) require more antibiotic. And finally, return to baseline eustachian tube function may take longer if severe AOM caused enhanced inflammation.
3. Recurrent AOM. (AOM within 1 prior month) – With recurrent AOM, the second “hit” to the eustachian tube may lead to more dysfunction, so a longer antibiotic course may be required to allow more complete source control and more time for more complete functional recovery after a repeated inflammatory injury.
4. Bilateral AOM. Two independent but infected sites mean twice the chance for failure. So, a longer course could allow more time for both sites to undergo “natural” source control.4
More bacteria – more antibiotic? So, is more antibiotic really needed for a higher bacterial load? In vitro this is known as the “inoculum effect,” particularly for beta-lactam drugs, for example, amoxicillin and cephalosporins. Laboratory susceptibility testing is performed with a specifically defined quantity of bacteria (105 bacteria/mL) and the minimum inhibitory concentration (MIC) is the lowest antibiotic concentration that stops bacterial growth. We know that drugs will likely fail if the MIC exceeds the achievable antibiotic concentration at the infection site. But is it as simple as just exceeding the MIC at the infection site? No, pharmacodynamics tell us that overall antibiotic exposure is also important. For example, to be successful, beta-lactam concentrations need to be above the MIC for 40%-50% of the day.
Higher MIC with higher bacterial load. Particularly for beta-lactams, testing with a quantity of bacteria >105/mL produces a higher MIC in vitro. This suggests that clinical failure could occur, even when our in vivo dosing leads to 40%-50% above the “standard” MIC that was obtained from testing the lab standard of 105/mL bacteria, when the infected site’s (middle ear) bacterial load is >105/mL (such higher bacterial loads occur in up to 30% of AOM).5 One way to negate inoculum effect is source control (drain the abscess or debridement), which reduces the bacterial load as well as allowing better antibiotic penetration– both favoring infection resolution. But with suboptimal source control, for example, the middle ear is not drained externally or internally, longer courses (more antibiotic exposure) could aid resolution. Whether the exposure can be administered as higher doses in fewer days or standard doses for more days is debatable but consider that a single parenteral dose of ceftriaxone successfully resolves AOM not attributable to penicillin-nonsusceptible pneumococcus.6Bottom line: Even though the number of potential antibiotics has contracted in the past 20 years, the need to individualize AOM treatment remains important and duration choices are more complex. Indeed, AOM comes in different flavors with patient age, clinical presentation, and episode frequency dictating the choice of duration.
Dr. Christopher J. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospitals and Clinics in Kansas City, Mo. Email him at [email protected].
References
1. Pichichero ME. MDedge. 2022 Jan 11.
2. Ruohola A et al. Pediatrics. 2003;111(5):1061-7.
3. Hoberman A et al. N Engl J Med. 2016;375(25):2446-56.
4. Pichichero ME et al. Otolaryngol Head Neck Surg. 2001;124(4):381-7.
5. Harrison CJ et al. Pediatr Infect Dis. 1985;4(6):641-6.
6. Leibovitz E et al. Pediatr Infect Dis. 2000;19(11):1040-5.
Organ transplantation: Unvaccinated need not apply
I agree with most advice given by the affable TV character Ted Lasso. “Every choice is a chance,” he said. Pandemic-era physicians must now consider whether a politically motivated choice to decline COVID-19 vaccination should negatively affect the chance to receive an organ donation.
And in confronting these choices, we have a chance to educate the public on the complexities of the organ allocation process.
A well-informed patient’s personal choice should be honored, even if clinicians disagree, if it does not affect the well-being of others. For example, I once had a patient in acute leukemic crisis who declined blood products because she was a Jehovah’s Witness. She died. Her choice affected her longevity only.
Compare that decision with awarding an organ to an individual who has declined readily available protection of that organ. Weigh that choice against the fact that said protection is against an infectious disease that has killed over 5.5 million worldwide.
Some institutions stand strong, others hedge their bets
Admirably, Loyola University Health System understands that difference. They published a firm stand on transplant candidacy and COVID-19 vaccination status in the Journal of Heart and Lung Transplant. Daniel Dilling, MD, medical director of the lung transplantation program , and Mark Kuczewski, PhD, a professor of medical ethics at Loyola University Chicago, Maywood, Ill., wrote that: “We believe that requiring vaccination against COVID-19 should not be controversial when we focus strictly on established frameworks and practices surrounding eligibility for wait-listing to receive a solid organ transplant.”
The Cleveland Clinic apparently agrees. In October 2021, they denied a liver transplant to Michelle Vitullo of Ohio, whose daughter had been deemed “a perfect match.” Her daughter, also unvaccinated, stated: “Being denied for a nonmedical reason for someone’s beliefs that are different to yours, I mean that’s not how that should be.”
But vaccination status is a medical reason, given well-established data regarding increased mortality among the immunosuppressed. Ms. Vitullo then said: “We are trying to get to UPMC [University of Pittsburgh Medical Center] as they don’t require a vaccination.”
The public information page on transplant candidacy from UPMC reads (my italics): It is recommended that all transplant candidates, transplant recipients, and their household members receive COVID-19 vaccination when the vaccine is available to them. It is preferred that transplant candidates are vaccinated more than 2 weeks before transplantation.
I reached out to UPMC for clarification and was told by email that “we do not have a policy regarding COVID-19 vaccination requirement for current transplant candidates.” Houston Methodist shares the same agnostic stance.
Compare these opinions with Brigham and Women’s Hospital, where the requirements are resolute: “Like most other transplant programs across the country, the COVID-19 vaccine is one of several vaccines and lifestyle behaviors that are required for patients awaiting solid organ transplant.”
They add that “transplant candidates must also receive the seasonal influenza and hepatitis B vaccines, follow other healthy behaviors, and demonstrate they can commit to taking the required medications following transplant.”
In January 2022, Brigham and Women’s Hospital declared 31-year-old D.J. Ferguson ineligible for a heart transplant because he declined to be vaccinated against COVID-19. According to the New York Post and ABC News, his physicians resorted to left ventricular assist device support. His mother, Tracy Ferguson, is quoted as saying: “He’s not an antivaxxer. He has all of his vaccines.” I’ll just leave that right there.
Unfortunately, Michelle Vitullo’s obituary was published in December 2021. Regardless of whether she received her liver transplant, the outcome is tragic, and whatever you think of this family’s battle playing out in the glare of the national spotlight, their loss is no less devastating.
The directed-donation aspect of this case poses an interesting question. A news anchor asked the mother and daughter: “If you both accept the risks, why doesn’t the hospital just let you try?” The answers are obvious to us clinicians. Performing a transplantation in an unvaccinated patient could lead to their early death if they became infected because of their immunocompromised state, would open the door for transplantation of any patient who is unvaccinated for anything, including influenza and hepatitis B, which could result in the preventable waste of organs, and puts other vulnerable hospitalized patients at risk during the initial transplant stay and follow-up.
That’s not to mention the potential legal suit. Never has a consent form dissuaded any party from lodging an accusation of wrongful death or medical malpractice. In the face of strong data on higher mortality in unvaccinated, immunocompromised patients, a good lawyer could charge that the institution and transplant surgeons should have known better, regardless of the donor and recipient’s willingness to accept the risks.
The Vitullo and Ferguson cases are among many similar dilemmas surrounding transplant candidacy across the United States.
University of Virginia Health in Charlottesville denied 42-year-old Shamgar Connors a kidney transplant because he is unvaccinated, despite a previous COVID-19 infection. In October 2021, Leilani Lutali of Colorado was denied a kidney by UCHealth because she declined vaccination.
As Ted Lasso says: “There’s a bunch of crazy stuff on Twitter.”
Predictably, social media is full of public outcry. “Some cold-hearted people on here” tweeted one. “What if it was one of your loved ones who needed a transplant?” Another tweeted the Hippocratic oath with the comment that “They all swore under this noble ‘oat’, but I guess it’s been forgotten.” (This was followed with a photo of a box of Quaker Oats in a failed attempt at humor.) These discussions among the Twitterati highlight the depths of misunderstanding on organ transplantation.
To be fair, unless you have been personally involved in the decision-making process for transplant candidacy, there is little opportunity to be educated. I explain to my anxious patients and their families that a donor organ is like a fumbled football. There may be well over 100 patients at all levels of transplant status in many geographic locations diving for that same organ.
The transplant team is tasked with finding the best match, determining who is the sickest, assessing time for transport of that organ, and, above all, who will be the best steward of that organ.
Take heart transplantation, for instance. Approximately 3,500 patients in the United States are awaiting one each year. Instead of facing an almost certain death within 5 years, a transplant recipient has a chance at a median survival of 12-13 years. The cost of a heart transplant is approximately $1.38 million, according to Milliman, a consulting firm. This is “an incredibly resource intensive procedure,” including expenditures for transportation, antirejection medication, office visits, physician fees, ICU stays, rejection surveillance, and acute rejection therapies.
Transplant denial is nothing new
People get turned down for organ transplants all the time. My patient with end-stage dilated cardiomyopathy was denied a heart transplant when it was discovered that he had scores of outstanding parking tickets. This was seen as a surrogate for an inability to afford his antirejection medication.
Another patient swore that her positive cotinine levels were caused by endless hours at the bingo hall where second-hand smoke swirled. She was also denied. Many potential candidates who are in acute decline hold precariously to newfound sobriety. They are denied. A patient’s boyfriend told the transplant team that he couldn’t be relied upon to drive her to her appointments. She was denied.
Many people who engage in antisocial behaviors have no idea that these actions may result in the denial of an organ transplant should their future selves need one. These are hard lines, but everyone should agree that the odds of survival are heavily in favor of the consistently adherent.
We should take this opportunity to educate the public on how complicated obtaining an organ transplant can be. More than 6,000 people die each year waiting for an organ because of the supply-and-demand disparities in the transplantation arena. I’m willing to bet that many of the loudest protesters in favor of unvaccinated transplant recipients have not signed the organ donor box on the back of their driver’s license. This conversation is an opportunity to change that and remind people that organ donation may be their only opportunity to save a fellow human’s life.
Again, to quote Ted Lasso: “If you care about someone and you got a little love in your heart, there ain’t nothing you can’t get through together.” That philosophy should apply to the tasks of selecting the best organ donors as well as the best organ recipients.
And every organ should go to the one who will honor their donor and their donor’s family by taking the best care of that ultimate gift of life, including being vaccinated against COVID-19.
Dr. Walton-Shirley is a native Kentuckian who retired from full-time invasive cardiology. She enjoys locums work in Montana and is a champion of physician rights and patient safety. She disclosed no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
I agree with most advice given by the affable TV character Ted Lasso. “Every choice is a chance,” he said. Pandemic-era physicians must now consider whether a politically motivated choice to decline COVID-19 vaccination should negatively affect the chance to receive an organ donation.
And in confronting these choices, we have a chance to educate the public on the complexities of the organ allocation process.
A well-informed patient’s personal choice should be honored, even if clinicians disagree, if it does not affect the well-being of others. For example, I once had a patient in acute leukemic crisis who declined blood products because she was a Jehovah’s Witness. She died. Her choice affected her longevity only.
Compare that decision with awarding an organ to an individual who has declined readily available protection of that organ. Weigh that choice against the fact that said protection is against an infectious disease that has killed over 5.5 million worldwide.
Some institutions stand strong, others hedge their bets
Admirably, Loyola University Health System understands that difference. They published a firm stand on transplant candidacy and COVID-19 vaccination status in the Journal of Heart and Lung Transplant. Daniel Dilling, MD, medical director of the lung transplantation program , and Mark Kuczewski, PhD, a professor of medical ethics at Loyola University Chicago, Maywood, Ill., wrote that: “We believe that requiring vaccination against COVID-19 should not be controversial when we focus strictly on established frameworks and practices surrounding eligibility for wait-listing to receive a solid organ transplant.”
The Cleveland Clinic apparently agrees. In October 2021, they denied a liver transplant to Michelle Vitullo of Ohio, whose daughter had been deemed “a perfect match.” Her daughter, also unvaccinated, stated: “Being denied for a nonmedical reason for someone’s beliefs that are different to yours, I mean that’s not how that should be.”
But vaccination status is a medical reason, given well-established data regarding increased mortality among the immunosuppressed. Ms. Vitullo then said: “We are trying to get to UPMC [University of Pittsburgh Medical Center] as they don’t require a vaccination.”
The public information page on transplant candidacy from UPMC reads (my italics): It is recommended that all transplant candidates, transplant recipients, and their household members receive COVID-19 vaccination when the vaccine is available to them. It is preferred that transplant candidates are vaccinated more than 2 weeks before transplantation.
I reached out to UPMC for clarification and was told by email that “we do not have a policy regarding COVID-19 vaccination requirement for current transplant candidates.” Houston Methodist shares the same agnostic stance.
Compare these opinions with Brigham and Women’s Hospital, where the requirements are resolute: “Like most other transplant programs across the country, the COVID-19 vaccine is one of several vaccines and lifestyle behaviors that are required for patients awaiting solid organ transplant.”
They add that “transplant candidates must also receive the seasonal influenza and hepatitis B vaccines, follow other healthy behaviors, and demonstrate they can commit to taking the required medications following transplant.”
In January 2022, Brigham and Women’s Hospital declared 31-year-old D.J. Ferguson ineligible for a heart transplant because he declined to be vaccinated against COVID-19. According to the New York Post and ABC News, his physicians resorted to left ventricular assist device support. His mother, Tracy Ferguson, is quoted as saying: “He’s not an antivaxxer. He has all of his vaccines.” I’ll just leave that right there.
Unfortunately, Michelle Vitullo’s obituary was published in December 2021. Regardless of whether she received her liver transplant, the outcome is tragic, and whatever you think of this family’s battle playing out in the glare of the national spotlight, their loss is no less devastating.
The directed-donation aspect of this case poses an interesting question. A news anchor asked the mother and daughter: “If you both accept the risks, why doesn’t the hospital just let you try?” The answers are obvious to us clinicians. Performing a transplantation in an unvaccinated patient could lead to their early death if they became infected because of their immunocompromised state, would open the door for transplantation of any patient who is unvaccinated for anything, including influenza and hepatitis B, which could result in the preventable waste of organs, and puts other vulnerable hospitalized patients at risk during the initial transplant stay and follow-up.
That’s not to mention the potential legal suit. Never has a consent form dissuaded any party from lodging an accusation of wrongful death or medical malpractice. In the face of strong data on higher mortality in unvaccinated, immunocompromised patients, a good lawyer could charge that the institution and transplant surgeons should have known better, regardless of the donor and recipient’s willingness to accept the risks.
The Vitullo and Ferguson cases are among many similar dilemmas surrounding transplant candidacy across the United States.
University of Virginia Health in Charlottesville denied 42-year-old Shamgar Connors a kidney transplant because he is unvaccinated, despite a previous COVID-19 infection. In October 2021, Leilani Lutali of Colorado was denied a kidney by UCHealth because she declined vaccination.
As Ted Lasso says: “There’s a bunch of crazy stuff on Twitter.”
Predictably, social media is full of public outcry. “Some cold-hearted people on here” tweeted one. “What if it was one of your loved ones who needed a transplant?” Another tweeted the Hippocratic oath with the comment that “They all swore under this noble ‘oat’, but I guess it’s been forgotten.” (This was followed with a photo of a box of Quaker Oats in a failed attempt at humor.) These discussions among the Twitterati highlight the depths of misunderstanding on organ transplantation.
To be fair, unless you have been personally involved in the decision-making process for transplant candidacy, there is little opportunity to be educated. I explain to my anxious patients and their families that a donor organ is like a fumbled football. There may be well over 100 patients at all levels of transplant status in many geographic locations diving for that same organ.
The transplant team is tasked with finding the best match, determining who is the sickest, assessing time for transport of that organ, and, above all, who will be the best steward of that organ.
Take heart transplantation, for instance. Approximately 3,500 patients in the United States are awaiting one each year. Instead of facing an almost certain death within 5 years, a transplant recipient has a chance at a median survival of 12-13 years. The cost of a heart transplant is approximately $1.38 million, according to Milliman, a consulting firm. This is “an incredibly resource intensive procedure,” including expenditures for transportation, antirejection medication, office visits, physician fees, ICU stays, rejection surveillance, and acute rejection therapies.
Transplant denial is nothing new
People get turned down for organ transplants all the time. My patient with end-stage dilated cardiomyopathy was denied a heart transplant when it was discovered that he had scores of outstanding parking tickets. This was seen as a surrogate for an inability to afford his antirejection medication.
Another patient swore that her positive cotinine levels were caused by endless hours at the bingo hall where second-hand smoke swirled. She was also denied. Many potential candidates who are in acute decline hold precariously to newfound sobriety. They are denied. A patient’s boyfriend told the transplant team that he couldn’t be relied upon to drive her to her appointments. She was denied.
Many people who engage in antisocial behaviors have no idea that these actions may result in the denial of an organ transplant should their future selves need one. These are hard lines, but everyone should agree that the odds of survival are heavily in favor of the consistently adherent.
We should take this opportunity to educate the public on how complicated obtaining an organ transplant can be. More than 6,000 people die each year waiting for an organ because of the supply-and-demand disparities in the transplantation arena. I’m willing to bet that many of the loudest protesters in favor of unvaccinated transplant recipients have not signed the organ donor box on the back of their driver’s license. This conversation is an opportunity to change that and remind people that organ donation may be their only opportunity to save a fellow human’s life.
Again, to quote Ted Lasso: “If you care about someone and you got a little love in your heart, there ain’t nothing you can’t get through together.” That philosophy should apply to the tasks of selecting the best organ donors as well as the best organ recipients.
And every organ should go to the one who will honor their donor and their donor’s family by taking the best care of that ultimate gift of life, including being vaccinated against COVID-19.
Dr. Walton-Shirley is a native Kentuckian who retired from full-time invasive cardiology. She enjoys locums work in Montana and is a champion of physician rights and patient safety. She disclosed no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
I agree with most advice given by the affable TV character Ted Lasso. “Every choice is a chance,” he said. Pandemic-era physicians must now consider whether a politically motivated choice to decline COVID-19 vaccination should negatively affect the chance to receive an organ donation.
And in confronting these choices, we have a chance to educate the public on the complexities of the organ allocation process.
A well-informed patient’s personal choice should be honored, even if clinicians disagree, if it does not affect the well-being of others. For example, I once had a patient in acute leukemic crisis who declined blood products because she was a Jehovah’s Witness. She died. Her choice affected her longevity only.
Compare that decision with awarding an organ to an individual who has declined readily available protection of that organ. Weigh that choice against the fact that said protection is against an infectious disease that has killed over 5.5 million worldwide.
Some institutions stand strong, others hedge their bets
Admirably, Loyola University Health System understands that difference. They published a firm stand on transplant candidacy and COVID-19 vaccination status in the Journal of Heart and Lung Transplant. Daniel Dilling, MD, medical director of the lung transplantation program , and Mark Kuczewski, PhD, a professor of medical ethics at Loyola University Chicago, Maywood, Ill., wrote that: “We believe that requiring vaccination against COVID-19 should not be controversial when we focus strictly on established frameworks and practices surrounding eligibility for wait-listing to receive a solid organ transplant.”
The Cleveland Clinic apparently agrees. In October 2021, they denied a liver transplant to Michelle Vitullo of Ohio, whose daughter had been deemed “a perfect match.” Her daughter, also unvaccinated, stated: “Being denied for a nonmedical reason for someone’s beliefs that are different to yours, I mean that’s not how that should be.”
But vaccination status is a medical reason, given well-established data regarding increased mortality among the immunosuppressed. Ms. Vitullo then said: “We are trying to get to UPMC [University of Pittsburgh Medical Center] as they don’t require a vaccination.”
The public information page on transplant candidacy from UPMC reads (my italics): It is recommended that all transplant candidates, transplant recipients, and their household members receive COVID-19 vaccination when the vaccine is available to them. It is preferred that transplant candidates are vaccinated more than 2 weeks before transplantation.
I reached out to UPMC for clarification and was told by email that “we do not have a policy regarding COVID-19 vaccination requirement for current transplant candidates.” Houston Methodist shares the same agnostic stance.
Compare these opinions with Brigham and Women’s Hospital, where the requirements are resolute: “Like most other transplant programs across the country, the COVID-19 vaccine is one of several vaccines and lifestyle behaviors that are required for patients awaiting solid organ transplant.”
They add that “transplant candidates must also receive the seasonal influenza and hepatitis B vaccines, follow other healthy behaviors, and demonstrate they can commit to taking the required medications following transplant.”
In January 2022, Brigham and Women’s Hospital declared 31-year-old D.J. Ferguson ineligible for a heart transplant because he declined to be vaccinated against COVID-19. According to the New York Post and ABC News, his physicians resorted to left ventricular assist device support. His mother, Tracy Ferguson, is quoted as saying: “He’s not an antivaxxer. He has all of his vaccines.” I’ll just leave that right there.
Unfortunately, Michelle Vitullo’s obituary was published in December 2021. Regardless of whether she received her liver transplant, the outcome is tragic, and whatever you think of this family’s battle playing out in the glare of the national spotlight, their loss is no less devastating.
The directed-donation aspect of this case poses an interesting question. A news anchor asked the mother and daughter: “If you both accept the risks, why doesn’t the hospital just let you try?” The answers are obvious to us clinicians. Performing a transplantation in an unvaccinated patient could lead to their early death if they became infected because of their immunocompromised state, would open the door for transplantation of any patient who is unvaccinated for anything, including influenza and hepatitis B, which could result in the preventable waste of organs, and puts other vulnerable hospitalized patients at risk during the initial transplant stay and follow-up.
That’s not to mention the potential legal suit. Never has a consent form dissuaded any party from lodging an accusation of wrongful death or medical malpractice. In the face of strong data on higher mortality in unvaccinated, immunocompromised patients, a good lawyer could charge that the institution and transplant surgeons should have known better, regardless of the donor and recipient’s willingness to accept the risks.
The Vitullo and Ferguson cases are among many similar dilemmas surrounding transplant candidacy across the United States.
University of Virginia Health in Charlottesville denied 42-year-old Shamgar Connors a kidney transplant because he is unvaccinated, despite a previous COVID-19 infection. In October 2021, Leilani Lutali of Colorado was denied a kidney by UCHealth because she declined vaccination.
As Ted Lasso says: “There’s a bunch of crazy stuff on Twitter.”
Predictably, social media is full of public outcry. “Some cold-hearted people on here” tweeted one. “What if it was one of your loved ones who needed a transplant?” Another tweeted the Hippocratic oath with the comment that “They all swore under this noble ‘oat’, but I guess it’s been forgotten.” (This was followed with a photo of a box of Quaker Oats in a failed attempt at humor.) These discussions among the Twitterati highlight the depths of misunderstanding on organ transplantation.
To be fair, unless you have been personally involved in the decision-making process for transplant candidacy, there is little opportunity to be educated. I explain to my anxious patients and their families that a donor organ is like a fumbled football. There may be well over 100 patients at all levels of transplant status in many geographic locations diving for that same organ.
The transplant team is tasked with finding the best match, determining who is the sickest, assessing time for transport of that organ, and, above all, who will be the best steward of that organ.
Take heart transplantation, for instance. Approximately 3,500 patients in the United States are awaiting one each year. Instead of facing an almost certain death within 5 years, a transplant recipient has a chance at a median survival of 12-13 years. The cost of a heart transplant is approximately $1.38 million, according to Milliman, a consulting firm. This is “an incredibly resource intensive procedure,” including expenditures for transportation, antirejection medication, office visits, physician fees, ICU stays, rejection surveillance, and acute rejection therapies.
Transplant denial is nothing new
People get turned down for organ transplants all the time. My patient with end-stage dilated cardiomyopathy was denied a heart transplant when it was discovered that he had scores of outstanding parking tickets. This was seen as a surrogate for an inability to afford his antirejection medication.
Another patient swore that her positive cotinine levels were caused by endless hours at the bingo hall where second-hand smoke swirled. She was also denied. Many potential candidates who are in acute decline hold precariously to newfound sobriety. They are denied. A patient’s boyfriend told the transplant team that he couldn’t be relied upon to drive her to her appointments. She was denied.
Many people who engage in antisocial behaviors have no idea that these actions may result in the denial of an organ transplant should their future selves need one. These are hard lines, but everyone should agree that the odds of survival are heavily in favor of the consistently adherent.
We should take this opportunity to educate the public on how complicated obtaining an organ transplant can be. More than 6,000 people die each year waiting for an organ because of the supply-and-demand disparities in the transplantation arena. I’m willing to bet that many of the loudest protesters in favor of unvaccinated transplant recipients have not signed the organ donor box on the back of their driver’s license. This conversation is an opportunity to change that and remind people that organ donation may be their only opportunity to save a fellow human’s life.
Again, to quote Ted Lasso: “If you care about someone and you got a little love in your heart, there ain’t nothing you can’t get through together.” That philosophy should apply to the tasks of selecting the best organ donors as well as the best organ recipients.
And every organ should go to the one who will honor their donor and their donor’s family by taking the best care of that ultimate gift of life, including being vaccinated against COVID-19.
Dr. Walton-Shirley is a native Kentuckian who retired from full-time invasive cardiology. She enjoys locums work in Montana and is a champion of physician rights and patient safety. She disclosed no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
ctDNA shows promise for assessing lung cancer treatment response
This transcript has been edited for clarity. A version of this article first appeared on Medscape.com.
Hello. This is Mark Kris from Memorial Sloan Kettering, talking today about circulating tumor DNA (ctDNA), an emerging technology for use in perioperative patients. Recently, there have been a number of presentations about the use of ctDNA measurements in patients receiving pre- or postoperative therapies. These are critical therapies because they are given with the intention of improving the chance for cure.
All three of the presentations I’m going to mention have one thing in common: They used the so-called tumor-informed panel. That technology is going to become very important, as shown in these presentations.
I made one of these presentations at the European Society for Medical Oncology Immuno-Oncology virtual meeting in Geneva. In our study, we were able to find genes in the majority of patients who had tumor tissue available. These patients were preoperative surgical candidates. In 72% of these, we were able to find and track ctDNA. When we tracked the DNA in the blood, we saw that the falling levels of DNA were associated with shrinkages of the cancer radiographically – the degree of shrinkage seen in this case in the neoadjuvant examination at the time of surgery and examining the resection specimen after neoadjuvant therapy. Ultimately, the major pathologic responses were associated with clearing or falling DNA as well. Perhaps the most interesting observation is that when you put this DNA information together with the major pathologic response information, all of the patients who had clearance of ctDNA and had a major pathologic response were disease free. I believe that eventually we will use this ctDNA data in conjunction with other measures of benefit to reach a more precise assessment of therapy benefit, and eventually it may be helpful for prognosis as well.
Two other studies also used this technology. One was earlier this year, presented by Patrick Forde at the American Association for Cancer Research meeting. They associated changes in ctDNA using another tumor-informed assay. In that study, using the Archer assay, they were able to show that the ctDNA clearance was associated with a complete pathologic response. So again, combining this information provides a more precise measurement of the benefit of therapy.
Another presentation at ESMO Immuno-Oncology, by Caicun Zhou, looked at the Natera assay, another tumor-informed assay, in a trial of adjuvant atezolizumab. This group showed that patients who had clearance of their ctDNA after surgery had the greatest benefit from subsequent atezolizumab therapy. And even those patients who did not have clearance experienced some benefit of the atezolizumab therapy. In addition, they assessed the degree of benefit associated with whether or not PD-L1 was present. Those patients who had PD-L1 expression experienced the greatest benefit from the atezolizumab. For patients who didn’t have PD-L1 expression, where you wouldn’t expect atezolizumab to have this greater benefit, they didn’t see it.
I believe that ctDNA-informed testing will become more and more useful, both in clinical trials and ultimately in the care of patients with early-stage lung cancers. These tumor-informed assays are going to be standards of care and provide physicians and patients a better estimate of the effectiveness of therapy going forward.
Dr. Kris is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York. He reported serving as a consultant and/or adviser for AstraZeneca, Daiichi Sankyo, and Pfizer, and has received payments for various services from Genentech.
This transcript has been edited for clarity. A version of this article first appeared on Medscape.com.
Hello. This is Mark Kris from Memorial Sloan Kettering, talking today about circulating tumor DNA (ctDNA), an emerging technology for use in perioperative patients. Recently, there have been a number of presentations about the use of ctDNA measurements in patients receiving pre- or postoperative therapies. These are critical therapies because they are given with the intention of improving the chance for cure.
All three of the presentations I’m going to mention have one thing in common: They used the so-called tumor-informed panel. That technology is going to become very important, as shown in these presentations.
I made one of these presentations at the European Society for Medical Oncology Immuno-Oncology virtual meeting in Geneva. In our study, we were able to find genes in the majority of patients who had tumor tissue available. These patients were preoperative surgical candidates. In 72% of these, we were able to find and track ctDNA. When we tracked the DNA in the blood, we saw that the falling levels of DNA were associated with shrinkages of the cancer radiographically – the degree of shrinkage seen in this case in the neoadjuvant examination at the time of surgery and examining the resection specimen after neoadjuvant therapy. Ultimately, the major pathologic responses were associated with clearing or falling DNA as well. Perhaps the most interesting observation is that when you put this DNA information together with the major pathologic response information, all of the patients who had clearance of ctDNA and had a major pathologic response were disease free. I believe that eventually we will use this ctDNA data in conjunction with other measures of benefit to reach a more precise assessment of therapy benefit, and eventually it may be helpful for prognosis as well.
Two other studies also used this technology. One was earlier this year, presented by Patrick Forde at the American Association for Cancer Research meeting. They associated changes in ctDNA using another tumor-informed assay. In that study, using the Archer assay, they were able to show that the ctDNA clearance was associated with a complete pathologic response. So again, combining this information provides a more precise measurement of the benefit of therapy.
Another presentation at ESMO Immuno-Oncology, by Caicun Zhou, looked at the Natera assay, another tumor-informed assay, in a trial of adjuvant atezolizumab. This group showed that patients who had clearance of their ctDNA after surgery had the greatest benefit from subsequent atezolizumab therapy. And even those patients who did not have clearance experienced some benefit of the atezolizumab therapy. In addition, they assessed the degree of benefit associated with whether or not PD-L1 was present. Those patients who had PD-L1 expression experienced the greatest benefit from the atezolizumab. For patients who didn’t have PD-L1 expression, where you wouldn’t expect atezolizumab to have this greater benefit, they didn’t see it.
I believe that ctDNA-informed testing will become more and more useful, both in clinical trials and ultimately in the care of patients with early-stage lung cancers. These tumor-informed assays are going to be standards of care and provide physicians and patients a better estimate of the effectiveness of therapy going forward.
Dr. Kris is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York. He reported serving as a consultant and/or adviser for AstraZeneca, Daiichi Sankyo, and Pfizer, and has received payments for various services from Genentech.
This transcript has been edited for clarity. A version of this article first appeared on Medscape.com.
Hello. This is Mark Kris from Memorial Sloan Kettering, talking today about circulating tumor DNA (ctDNA), an emerging technology for use in perioperative patients. Recently, there have been a number of presentations about the use of ctDNA measurements in patients receiving pre- or postoperative therapies. These are critical therapies because they are given with the intention of improving the chance for cure.
All three of the presentations I’m going to mention have one thing in common: They used the so-called tumor-informed panel. That technology is going to become very important, as shown in these presentations.
I made one of these presentations at the European Society for Medical Oncology Immuno-Oncology virtual meeting in Geneva. In our study, we were able to find genes in the majority of patients who had tumor tissue available. These patients were preoperative surgical candidates. In 72% of these, we were able to find and track ctDNA. When we tracked the DNA in the blood, we saw that the falling levels of DNA were associated with shrinkages of the cancer radiographically – the degree of shrinkage seen in this case in the neoadjuvant examination at the time of surgery and examining the resection specimen after neoadjuvant therapy. Ultimately, the major pathologic responses were associated with clearing or falling DNA as well. Perhaps the most interesting observation is that when you put this DNA information together with the major pathologic response information, all of the patients who had clearance of ctDNA and had a major pathologic response were disease free. I believe that eventually we will use this ctDNA data in conjunction with other measures of benefit to reach a more precise assessment of therapy benefit, and eventually it may be helpful for prognosis as well.
Two other studies also used this technology. One was earlier this year, presented by Patrick Forde at the American Association for Cancer Research meeting. They associated changes in ctDNA using another tumor-informed assay. In that study, using the Archer assay, they were able to show that the ctDNA clearance was associated with a complete pathologic response. So again, combining this information provides a more precise measurement of the benefit of therapy.
Another presentation at ESMO Immuno-Oncology, by Caicun Zhou, looked at the Natera assay, another tumor-informed assay, in a trial of adjuvant atezolizumab. This group showed that patients who had clearance of their ctDNA after surgery had the greatest benefit from subsequent atezolizumab therapy. And even those patients who did not have clearance experienced some benefit of the atezolizumab therapy. In addition, they assessed the degree of benefit associated with whether or not PD-L1 was present. Those patients who had PD-L1 expression experienced the greatest benefit from the atezolizumab. For patients who didn’t have PD-L1 expression, where you wouldn’t expect atezolizumab to have this greater benefit, they didn’t see it.
I believe that ctDNA-informed testing will become more and more useful, both in clinical trials and ultimately in the care of patients with early-stage lung cancers. These tumor-informed assays are going to be standards of care and provide physicians and patients a better estimate of the effectiveness of therapy going forward.
Dr. Kris is chief of the thoracic oncology service and the William and Joy Ruane Chair in Thoracic Oncology at Memorial Sloan Kettering Cancer Center in New York. He reported serving as a consultant and/or adviser for AstraZeneca, Daiichi Sankyo, and Pfizer, and has received payments for various services from Genentech.
Did you know these things about nicotine? Your patients don’t
When asked, young people report that their reasons for starting smoking include rebellion, a new thing to try, and a peer social activity, among others. While you recognize these as developmentally expected drives, it is frustrating and scary that youth don’t realize how their brains are especially sensitive to permanent changes from nicotine.
Smoking even five packs of cigarettes is enough to cause addiction in youth; an influence as powerful as for cocaine or heroin. One pod of a vaping device delivers as much nicotine as one to five packs of cigarettes, depending on the strength and brand. There are no standards for this content and youth often are unaware of any nicotine and chemicals in vapes. Over 90% of adult smokers started before age 18, some as young as 6, mainly because quitting is so difficult. Cigarettes and vaping are not the only sources of nicotine used by youth; others are oral tobacco (chewing tobacco and dip), cigars, pipes, snus (between cheek and gum), hookahs, electronic devices, bidis (tobacco in a tendu leaf), kreteks (tobacco with cloves), and dissolvable tobacco products. Many youth use both cigarettes and noncigarette tobacco.
Given these predispositions, short-term COVID-19 and asthma exacerbation, and the long-lasting detriment of smoking on neurological, cardiac, pulmonary, and emotional health, actually the “leading preventable cause of death,” our job as pediatric providers is to do our best to prevent smoking/vaping or help our patients quit. But adolescent development is notoriously characterized by short-term thinking and feeling immune from long-term health consequences. So what approach has the best results? Focus on aspects of smoking important to the youth now, such as sports performance, bad breath, social stigma, insomnia, cost, lack of benefit for weight loss, and hazardous waste produced. Add to that loss of independence and being manipulated by Big Business by getting them (and targeted minorities) hooked may be salient in our discussion.
Even a brief 3-minute discussion using the AAC (Ask/Assess, Advise, Connect) format has shown effectiveness in getting teens and adults to quit smoking. Our assessment needs to include asking the extent of current use and symptoms of dependence to inform the treatment plan. We need to use their trust in us to advise that quitting is the best thing they can do for their health.
If the youth’s readiness stage is “thinking about stopping” nicotine, our motivational interview–style discussion of pros and cons could include asking “How important is it to you to stop?” and “What are some things that would help you?” If they are open to trying to stop, advise them to set a quit date within 2 weeks and suggest reducing gradually before then (and schedule follow-up). The plan needs to include dealing with the inevitable urges by finding ways to avoid current triggers to smoke (e.g., certain school bathrooms, people drinking or smoking, or stress over homework, conflict at home, etc.). Encourage exercise and meditation to distract and deal with the anxiety; asking family to quit; having a snack handy (such as sugarless gum or sunflower seeds) for when oral cravings develop; and setting rewards for early days of smoke-free success. We need to inform youth that using e-cigs actually reduces rates of success in quitting.
We need to warn youth of the withdrawal symptoms and their usual course when quitting: cravings each lasting 15-20 minutes (starting at 1/2-4 hours); restlessness, sadness, hopelessness (10 hours); irritability, trouble concentrating, insomnia, hunger and weight gain (5-10 pounds over 2 weeks, starting 24 hrs); headaches, dizziness, fatigue (starting 2 days); and anxiety (starting 3 days). There tends to be less brain fog, and less hunger after 2-4 weeks, but depression, anxiety, irritability, cough, constipation, and even suicidal thoughts may last weeks to months. Sounds nasty, right? No wonder quitting is so hard.
Support is crucial to quitting and staying off nicotine. You can provide this but, in addition to friends and family, we should connect youth to free ongoing phone counselors (1-800-QUIT-NOW or 877-44U-QUIT for Spanish), text services (text QUIT to 47848), apps (quit START), or community support.
While behavioral treatments are best for youth with minimal to mild dependence, risk of relapse is minimized with fewer withdrawal symptoms, thus the role for nicotine replacement therapy (NRT) for those with moderate to strong dependence and to help anyone ad lib with cravings. NRT is recommended by the American Academy of Pediatrics (AAP) to supplement counseling, although NRT is not Food and Drug Administration approved and requires a prescription for those under 18.
How can we determine the degree of dependence? Smoking more than 15 cigarettes per day (or vape equivalent) and inhaling even “seldom” counts as “moderate” dependence and more than 26 with difficulty refraining in several situations as “substantial” in the Fagerstrom Tolerance test. Early morning smoking is asked about, important to which NRT to use (gum or lozenge for faster onset). The Hooked on Nicotine Checklist assesses “loss of autonomy” over smoking by any “yes” item and is incorporated in the CRAFFT screen. The recommended dose of NRT and length of weaning is greater in substantial addiction versus moderate. Besides gum, lozenges, patch, inhaler, and nasal spray, you can prescribe bupropion (Wellbutrin or Zyban) or varenicline (Chantix), making note of the black box suicide warning. Combining NRTs is similarly effective compared with varenicline.
Relapse after quitting is more common than not. As for any chronic condition, in relapse we need to query adherence, and consider increasing NRT dose or wean duration, even years. Discussion should have a positive focus on “what was learned” from past attempts in making a new plan that incorporates Relevance, Risks, Rewards, Roadblocks, and Repetition.
Many youth smokers start because their parents smoke. While addressing adults may seem out of scope, we often treat parents when managing scabies, pinworms, meningococcal disease, and even depression for the benefit of the child. The AAP recommends prescribing NRT for parents, when needed.
Nicotine dependence is a chronic relapsing condition with comorbidities of substance use and psychiatric disorders that requires similar monitoring and support as for other chronic conditions we manage and is more likely to shorten lifespan than many.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Reference
Clinical practice policy to protect children from tobacco, nicotine, and tobacco smoke, Pediatrics 2015;136(5):1008-17. doi: 10.1542/peds.2015-31088.
When asked, young people report that their reasons for starting smoking include rebellion, a new thing to try, and a peer social activity, among others. While you recognize these as developmentally expected drives, it is frustrating and scary that youth don’t realize how their brains are especially sensitive to permanent changes from nicotine.
Smoking even five packs of cigarettes is enough to cause addiction in youth; an influence as powerful as for cocaine or heroin. One pod of a vaping device delivers as much nicotine as one to five packs of cigarettes, depending on the strength and brand. There are no standards for this content and youth often are unaware of any nicotine and chemicals in vapes. Over 90% of adult smokers started before age 18, some as young as 6, mainly because quitting is so difficult. Cigarettes and vaping are not the only sources of nicotine used by youth; others are oral tobacco (chewing tobacco and dip), cigars, pipes, snus (between cheek and gum), hookahs, electronic devices, bidis (tobacco in a tendu leaf), kreteks (tobacco with cloves), and dissolvable tobacco products. Many youth use both cigarettes and noncigarette tobacco.
Given these predispositions, short-term COVID-19 and asthma exacerbation, and the long-lasting detriment of smoking on neurological, cardiac, pulmonary, and emotional health, actually the “leading preventable cause of death,” our job as pediatric providers is to do our best to prevent smoking/vaping or help our patients quit. But adolescent development is notoriously characterized by short-term thinking and feeling immune from long-term health consequences. So what approach has the best results? Focus on aspects of smoking important to the youth now, such as sports performance, bad breath, social stigma, insomnia, cost, lack of benefit for weight loss, and hazardous waste produced. Add to that loss of independence and being manipulated by Big Business by getting them (and targeted minorities) hooked may be salient in our discussion.
Even a brief 3-minute discussion using the AAC (Ask/Assess, Advise, Connect) format has shown effectiveness in getting teens and adults to quit smoking. Our assessment needs to include asking the extent of current use and symptoms of dependence to inform the treatment plan. We need to use their trust in us to advise that quitting is the best thing they can do for their health.
If the youth’s readiness stage is “thinking about stopping” nicotine, our motivational interview–style discussion of pros and cons could include asking “How important is it to you to stop?” and “What are some things that would help you?” If they are open to trying to stop, advise them to set a quit date within 2 weeks and suggest reducing gradually before then (and schedule follow-up). The plan needs to include dealing with the inevitable urges by finding ways to avoid current triggers to smoke (e.g., certain school bathrooms, people drinking or smoking, or stress over homework, conflict at home, etc.). Encourage exercise and meditation to distract and deal with the anxiety; asking family to quit; having a snack handy (such as sugarless gum or sunflower seeds) for when oral cravings develop; and setting rewards for early days of smoke-free success. We need to inform youth that using e-cigs actually reduces rates of success in quitting.
We need to warn youth of the withdrawal symptoms and their usual course when quitting: cravings each lasting 15-20 minutes (starting at 1/2-4 hours); restlessness, sadness, hopelessness (10 hours); irritability, trouble concentrating, insomnia, hunger and weight gain (5-10 pounds over 2 weeks, starting 24 hrs); headaches, dizziness, fatigue (starting 2 days); and anxiety (starting 3 days). There tends to be less brain fog, and less hunger after 2-4 weeks, but depression, anxiety, irritability, cough, constipation, and even suicidal thoughts may last weeks to months. Sounds nasty, right? No wonder quitting is so hard.
Support is crucial to quitting and staying off nicotine. You can provide this but, in addition to friends and family, we should connect youth to free ongoing phone counselors (1-800-QUIT-NOW or 877-44U-QUIT for Spanish), text services (text QUIT to 47848), apps (quit START), or community support.
While behavioral treatments are best for youth with minimal to mild dependence, risk of relapse is minimized with fewer withdrawal symptoms, thus the role for nicotine replacement therapy (NRT) for those with moderate to strong dependence and to help anyone ad lib with cravings. NRT is recommended by the American Academy of Pediatrics (AAP) to supplement counseling, although NRT is not Food and Drug Administration approved and requires a prescription for those under 18.
How can we determine the degree of dependence? Smoking more than 15 cigarettes per day (or vape equivalent) and inhaling even “seldom” counts as “moderate” dependence and more than 26 with difficulty refraining in several situations as “substantial” in the Fagerstrom Tolerance test. Early morning smoking is asked about, important to which NRT to use (gum or lozenge for faster onset). The Hooked on Nicotine Checklist assesses “loss of autonomy” over smoking by any “yes” item and is incorporated in the CRAFFT screen. The recommended dose of NRT and length of weaning is greater in substantial addiction versus moderate. Besides gum, lozenges, patch, inhaler, and nasal spray, you can prescribe bupropion (Wellbutrin or Zyban) or varenicline (Chantix), making note of the black box suicide warning. Combining NRTs is similarly effective compared with varenicline.
Relapse after quitting is more common than not. As for any chronic condition, in relapse we need to query adherence, and consider increasing NRT dose or wean duration, even years. Discussion should have a positive focus on “what was learned” from past attempts in making a new plan that incorporates Relevance, Risks, Rewards, Roadblocks, and Repetition.
Many youth smokers start because their parents smoke. While addressing adults may seem out of scope, we often treat parents when managing scabies, pinworms, meningococcal disease, and even depression for the benefit of the child. The AAP recommends prescribing NRT for parents, when needed.
Nicotine dependence is a chronic relapsing condition with comorbidities of substance use and psychiatric disorders that requires similar monitoring and support as for other chronic conditions we manage and is more likely to shorten lifespan than many.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Reference
Clinical practice policy to protect children from tobacco, nicotine, and tobacco smoke, Pediatrics 2015;136(5):1008-17. doi: 10.1542/peds.2015-31088.
When asked, young people report that their reasons for starting smoking include rebellion, a new thing to try, and a peer social activity, among others. While you recognize these as developmentally expected drives, it is frustrating and scary that youth don’t realize how their brains are especially sensitive to permanent changes from nicotine.
Smoking even five packs of cigarettes is enough to cause addiction in youth; an influence as powerful as for cocaine or heroin. One pod of a vaping device delivers as much nicotine as one to five packs of cigarettes, depending on the strength and brand. There are no standards for this content and youth often are unaware of any nicotine and chemicals in vapes. Over 90% of adult smokers started before age 18, some as young as 6, mainly because quitting is so difficult. Cigarettes and vaping are not the only sources of nicotine used by youth; others are oral tobacco (chewing tobacco and dip), cigars, pipes, snus (between cheek and gum), hookahs, electronic devices, bidis (tobacco in a tendu leaf), kreteks (tobacco with cloves), and dissolvable tobacco products. Many youth use both cigarettes and noncigarette tobacco.
Given these predispositions, short-term COVID-19 and asthma exacerbation, and the long-lasting detriment of smoking on neurological, cardiac, pulmonary, and emotional health, actually the “leading preventable cause of death,” our job as pediatric providers is to do our best to prevent smoking/vaping or help our patients quit. But adolescent development is notoriously characterized by short-term thinking and feeling immune from long-term health consequences. So what approach has the best results? Focus on aspects of smoking important to the youth now, such as sports performance, bad breath, social stigma, insomnia, cost, lack of benefit for weight loss, and hazardous waste produced. Add to that loss of independence and being manipulated by Big Business by getting them (and targeted minorities) hooked may be salient in our discussion.
Even a brief 3-minute discussion using the AAC (Ask/Assess, Advise, Connect) format has shown effectiveness in getting teens and adults to quit smoking. Our assessment needs to include asking the extent of current use and symptoms of dependence to inform the treatment plan. We need to use their trust in us to advise that quitting is the best thing they can do for their health.
If the youth’s readiness stage is “thinking about stopping” nicotine, our motivational interview–style discussion of pros and cons could include asking “How important is it to you to stop?” and “What are some things that would help you?” If they are open to trying to stop, advise them to set a quit date within 2 weeks and suggest reducing gradually before then (and schedule follow-up). The plan needs to include dealing with the inevitable urges by finding ways to avoid current triggers to smoke (e.g., certain school bathrooms, people drinking or smoking, or stress over homework, conflict at home, etc.). Encourage exercise and meditation to distract and deal with the anxiety; asking family to quit; having a snack handy (such as sugarless gum or sunflower seeds) for when oral cravings develop; and setting rewards for early days of smoke-free success. We need to inform youth that using e-cigs actually reduces rates of success in quitting.
We need to warn youth of the withdrawal symptoms and their usual course when quitting: cravings each lasting 15-20 minutes (starting at 1/2-4 hours); restlessness, sadness, hopelessness (10 hours); irritability, trouble concentrating, insomnia, hunger and weight gain (5-10 pounds over 2 weeks, starting 24 hrs); headaches, dizziness, fatigue (starting 2 days); and anxiety (starting 3 days). There tends to be less brain fog, and less hunger after 2-4 weeks, but depression, anxiety, irritability, cough, constipation, and even suicidal thoughts may last weeks to months. Sounds nasty, right? No wonder quitting is so hard.
Support is crucial to quitting and staying off nicotine. You can provide this but, in addition to friends and family, we should connect youth to free ongoing phone counselors (1-800-QUIT-NOW or 877-44U-QUIT for Spanish), text services (text QUIT to 47848), apps (quit START), or community support.
While behavioral treatments are best for youth with minimal to mild dependence, risk of relapse is minimized with fewer withdrawal symptoms, thus the role for nicotine replacement therapy (NRT) for those with moderate to strong dependence and to help anyone ad lib with cravings. NRT is recommended by the American Academy of Pediatrics (AAP) to supplement counseling, although NRT is not Food and Drug Administration approved and requires a prescription for those under 18.
How can we determine the degree of dependence? Smoking more than 15 cigarettes per day (or vape equivalent) and inhaling even “seldom” counts as “moderate” dependence and more than 26 with difficulty refraining in several situations as “substantial” in the Fagerstrom Tolerance test. Early morning smoking is asked about, important to which NRT to use (gum or lozenge for faster onset). The Hooked on Nicotine Checklist assesses “loss of autonomy” over smoking by any “yes” item and is incorporated in the CRAFFT screen. The recommended dose of NRT and length of weaning is greater in substantial addiction versus moderate. Besides gum, lozenges, patch, inhaler, and nasal spray, you can prescribe bupropion (Wellbutrin or Zyban) or varenicline (Chantix), making note of the black box suicide warning. Combining NRTs is similarly effective compared with varenicline.
Relapse after quitting is more common than not. As for any chronic condition, in relapse we need to query adherence, and consider increasing NRT dose or wean duration, even years. Discussion should have a positive focus on “what was learned” from past attempts in making a new plan that incorporates Relevance, Risks, Rewards, Roadblocks, and Repetition.
Many youth smokers start because their parents smoke. While addressing adults may seem out of scope, we often treat parents when managing scabies, pinworms, meningococcal disease, and even depression for the benefit of the child. The AAP recommends prescribing NRT for parents, when needed.
Nicotine dependence is a chronic relapsing condition with comorbidities of substance use and psychiatric disorders that requires similar monitoring and support as for other chronic conditions we manage and is more likely to shorten lifespan than many.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Reference
Clinical practice policy to protect children from tobacco, nicotine, and tobacco smoke, Pediatrics 2015;136(5):1008-17. doi: 10.1542/peds.2015-31088.
Dietary recommendations for inflammatory rheumatic diseases
This interview is a translation of a video blog that first appeared on Medscape France. It has been edited for clarity.
Weight loss, omega-3 supplements, the Mediterranean diet? What about exclusion diets? Jérémie Sellam, MD, PhD, from Saint-Antoine Hospital in Paris, summarizes the key points of the first set of dietary recommendations of the French Society for Rheumatology.
Transcript
Jérémie Sellam, MD, PhD: Hello, everyone. I’m Professor Jérémie Sellam. I’m a rheumatologist at Saint-Antoine Hospital, which is affiliated with the Sorbonne University in Paris. And I was fortunate enough to coordinate France’s first set of dietary recommendations – in fact, the world’s first set of dietary recommendations – for patients with chronic inflammatory rheumatic diseases. I worked on this project with Claire Daien, MD, PhD, who’s a rheumatologist at Montpellier University Hospital.
The idea of coming up with dietary recommendations for patients with inflammatory rheumatic diseases came, quite simply, from our clinical practice. We see that when patients learn they have polyarthritis or spondyloarthritis, they start to experiment with different diets. Many patients start exclusion diets and experiment in all sorts of ways with the food they eat. And although rheumatologists have been able to find some information here and there in the literature, they’ve been pretty much on their own when trying to come up with advice to give their patients. It was to address this issue that [Dr.] Daien and I set out to form a working group. Because when patients aren’t able to get sound advice and authoritative guidance from their doctors, medical associations, or patient advocacy organizations, they often look for information online, and that information is not always reliable or validated.
This group was made up of rheumatologists, some who work at hospitals and others in private practice. Also involved were physician nutrition specialists and registered dietitians. Operating under the auspices of the French Society for Rheumatology, these multidisciplinary experts conducted out a systematic literature review for the purpose of establishing and drafting recommendations. The result was a declaration of eight general principles and nine recommendations.
General principles
The first of the general principles states that nutritional advice is not a substitute for the pharmacologic treatment of chronic inflammatory rheumatic diseases. As you know, whether it’s methotrexate or biologics, pharmacologic treatments are essential for the proper management of chronic inflammatory rheumatic diseases. We know that these medications have an anti-inflammatory effect, reduce pain, and – particularly in the case of rheumatoid arthritis – have a structural effect. In other words, they prevent joint deterioration and destruction. Now, I can tell you that there’s currently no diet, and no dietary supplement, that has proven to be structurally effective. So, yes, dietary intervention might turn out to be promising for patients with chronic inflammatory rheumatic diseases, but pharmacologic treatment must still be part of the picture.
Another general principle emphasizes that dietary intervention is a way for patients to be actively involved in the overall care of their disease, beyond just taking their medication. We know that patients, when they suffer from chronic diseases, are looking for something more, beyond just taking medications. Encouraging them to take an interest in their diet, asking them about what they eat, giving them advice, and supporting their desire to become involved in this aspect of their treatment plan can give them a sense of empowerment.
Dietary interventions can have articular effects, and I’m going to speak about which interventions you can propose, but also which can be beneficial in terms of cardiovascular health and bone health. All of this is based on the literature. In these recommendations, we’ve taken into account not only laboratory experiments – where this or that diet is given to a mouse with arthritis – but also reviewed randomized controlled trials that compare an intervention group with a control group. This is the benchmark we used to determine whether or not a diet should be recommended.
The recommendations
As for the recommendations themselves, we wanted to start off by emphasizing weight loss and what can be called weight-loss support. There’s a link between obesity and the risk of developing rheumatoid arthritis, and also psoriatic arthropathy. And the more overweight a patient is, the more active their disease. In other words, patients with obesity are going to experience more pain, more instances of wakefulness, and more morning stiffness than their normal-weight peers. They’re also going to show symptoms that suggest that disease activity is not controlled well.
Several randomized controlled studies have shown that weight loss will improve systemic joint symptoms. In one particular study, patients with psoriatic arthropathy were started on [tumor necrosis factor] inhibitor therapy and one group followed a prescribed diet and the other had no restrictions on eating. More patients in the diet group than in the no-diet group achieved minimal disease activity. Of course, in some cases – for example, patients with complicated morbid obesity – it might be necessary to have a discussion about bariatric surgery.
But practically speaking, how does one proceed? First of all, patients should be weighed at each visit and, if they’re overweight or obese, the subject should be broached. But even after that conversation, the reality remains that it’s not easy to lose weight. So in the recommendations, we focused on the fact that it shouldn’t be left to the rheumatologist or treating physician alone to handle this challenging aspect of treatment. They should incorporate dietary and nutritional care by reaching out to a dietician or, in the case of complicated obesity – especially when the BMI is higher than 35 kg/m2 – they can refer patients to a nutrition expert who can manage the patient’s obesity, come up with a weight-loss plan, and handle any complications that might arise.
We don’t speak about a low-calorie diet in the recommendations because a diet has a beginning and an end and, quite often, patients regain weight after stopping a diet. Instead, we speak about weight-loss support to point out that weight loss maintained through dietary changes brings about long-term control of disease activity.
In addition, we make two positive recommendations, which overlap, that can help patients control their disease: a Mediterranean diet and omega-3 supplements. One study showed that after participants with rheumatoid arthritis followed the Mediterranean diet for 1 year, those who also took omega-3 fish oil supplements were twice as likely to achieve remission (40% vs. 20%). This explains the interest in having omega-3 as part of the diet. Other studies have shown a broad benefit of the Mediterranean diet.
We know this diet: Fish, especially fatty fish; meat, but not every day, and white meat is best; and fruits and vegetables. In addition, exercise and stay hydrated. All of this can help patients who want to use diet as a means to control their disease. And, as I said earlier, studies have shown that omega-3 supplements have beneficial effects. These are essential polyunsaturated fatty acids, which can help control the disease and joint symptoms.
We also provide some exclusionary recommendations. Not all studies are done well, but it’s clear that there are no major benefits – in fact, no benefit at all – from vegan diets, gluten-free diets, or dairy-free diets. And with these diets, patients run the risk of developing deficiencies, so it’s important that patients are aware of this. We also have to keep in mind that exclusion diets can increase social isolation. Patients need to take part in meals; such gatherings are times for sharing and having social interactions. And I would say that they must be told that there are no data in the literature in support of these diets. But if they ever insist on this kind of intervention, I think that it’s better to advise them to do it under the supervision of a dietician and nutritionist, especially to prevent the development of deficiencies. We’re talking about deficiencies in things like calcium, vitamin B12, and selenium.
Conclusion
As you can see, we have positive recommendations when the patient wants to do something beyond pharmacologic treatment: the Mediterranean diet and omega-3 supplements. And we have negative recommendations, marked by a warning about the risk of developing deficiencies. But I think we all understand the importance of paying close attention to how our patients are experimenting with food. Their diets and eating habits can give us ideas for research and reviews that could allow us to deepen our understanding of the effect of diet on disease, because currently, the quality of the data on some of the diets and types of dietary interventions out there is rather tenuous.
Thank you for listening. I’d also like to thank Claire Daien, MD, PhD, for conducting this project with me so that we could come up with all of these recommendations. I’m also grateful to the following nutrition societies and associations who were our partners: the French Society of Nutrition, the French-Speaking Society of Clinical Nutrition and Metabolism, the French Association for the Study of Obesity, and the French Association of Dieticians and Nutritionists. And patient associations, too, must be recognized, as some of their members participated: the French National Association Against Rheumatoid Arthritis, the French Spondyloarthritis Association, and the French Association for Polyarthritis and Chronic Inflammatory Rheumatic Diseases.
This interview is a translation of a video blog that first appeared on Medscape France. It has been edited for clarity.
Weight loss, omega-3 supplements, the Mediterranean diet? What about exclusion diets? Jérémie Sellam, MD, PhD, from Saint-Antoine Hospital in Paris, summarizes the key points of the first set of dietary recommendations of the French Society for Rheumatology.
Transcript
Jérémie Sellam, MD, PhD: Hello, everyone. I’m Professor Jérémie Sellam. I’m a rheumatologist at Saint-Antoine Hospital, which is affiliated with the Sorbonne University in Paris. And I was fortunate enough to coordinate France’s first set of dietary recommendations – in fact, the world’s first set of dietary recommendations – for patients with chronic inflammatory rheumatic diseases. I worked on this project with Claire Daien, MD, PhD, who’s a rheumatologist at Montpellier University Hospital.
The idea of coming up with dietary recommendations for patients with inflammatory rheumatic diseases came, quite simply, from our clinical practice. We see that when patients learn they have polyarthritis or spondyloarthritis, they start to experiment with different diets. Many patients start exclusion diets and experiment in all sorts of ways with the food they eat. And although rheumatologists have been able to find some information here and there in the literature, they’ve been pretty much on their own when trying to come up with advice to give their patients. It was to address this issue that [Dr.] Daien and I set out to form a working group. Because when patients aren’t able to get sound advice and authoritative guidance from their doctors, medical associations, or patient advocacy organizations, they often look for information online, and that information is not always reliable or validated.
This group was made up of rheumatologists, some who work at hospitals and others in private practice. Also involved were physician nutrition specialists and registered dietitians. Operating under the auspices of the French Society for Rheumatology, these multidisciplinary experts conducted out a systematic literature review for the purpose of establishing and drafting recommendations. The result was a declaration of eight general principles and nine recommendations.
General principles
The first of the general principles states that nutritional advice is not a substitute for the pharmacologic treatment of chronic inflammatory rheumatic diseases. As you know, whether it’s methotrexate or biologics, pharmacologic treatments are essential for the proper management of chronic inflammatory rheumatic diseases. We know that these medications have an anti-inflammatory effect, reduce pain, and – particularly in the case of rheumatoid arthritis – have a structural effect. In other words, they prevent joint deterioration and destruction. Now, I can tell you that there’s currently no diet, and no dietary supplement, that has proven to be structurally effective. So, yes, dietary intervention might turn out to be promising for patients with chronic inflammatory rheumatic diseases, but pharmacologic treatment must still be part of the picture.
Another general principle emphasizes that dietary intervention is a way for patients to be actively involved in the overall care of their disease, beyond just taking their medication. We know that patients, when they suffer from chronic diseases, are looking for something more, beyond just taking medications. Encouraging them to take an interest in their diet, asking them about what they eat, giving them advice, and supporting their desire to become involved in this aspect of their treatment plan can give them a sense of empowerment.
Dietary interventions can have articular effects, and I’m going to speak about which interventions you can propose, but also which can be beneficial in terms of cardiovascular health and bone health. All of this is based on the literature. In these recommendations, we’ve taken into account not only laboratory experiments – where this or that diet is given to a mouse with arthritis – but also reviewed randomized controlled trials that compare an intervention group with a control group. This is the benchmark we used to determine whether or not a diet should be recommended.
The recommendations
As for the recommendations themselves, we wanted to start off by emphasizing weight loss and what can be called weight-loss support. There’s a link between obesity and the risk of developing rheumatoid arthritis, and also psoriatic arthropathy. And the more overweight a patient is, the more active their disease. In other words, patients with obesity are going to experience more pain, more instances of wakefulness, and more morning stiffness than their normal-weight peers. They’re also going to show symptoms that suggest that disease activity is not controlled well.
Several randomized controlled studies have shown that weight loss will improve systemic joint symptoms. In one particular study, patients with psoriatic arthropathy were started on [tumor necrosis factor] inhibitor therapy and one group followed a prescribed diet and the other had no restrictions on eating. More patients in the diet group than in the no-diet group achieved minimal disease activity. Of course, in some cases – for example, patients with complicated morbid obesity – it might be necessary to have a discussion about bariatric surgery.
But practically speaking, how does one proceed? First of all, patients should be weighed at each visit and, if they’re overweight or obese, the subject should be broached. But even after that conversation, the reality remains that it’s not easy to lose weight. So in the recommendations, we focused on the fact that it shouldn’t be left to the rheumatologist or treating physician alone to handle this challenging aspect of treatment. They should incorporate dietary and nutritional care by reaching out to a dietician or, in the case of complicated obesity – especially when the BMI is higher than 35 kg/m2 – they can refer patients to a nutrition expert who can manage the patient’s obesity, come up with a weight-loss plan, and handle any complications that might arise.
We don’t speak about a low-calorie diet in the recommendations because a diet has a beginning and an end and, quite often, patients regain weight after stopping a diet. Instead, we speak about weight-loss support to point out that weight loss maintained through dietary changes brings about long-term control of disease activity.
In addition, we make two positive recommendations, which overlap, that can help patients control their disease: a Mediterranean diet and omega-3 supplements. One study showed that after participants with rheumatoid arthritis followed the Mediterranean diet for 1 year, those who also took omega-3 fish oil supplements were twice as likely to achieve remission (40% vs. 20%). This explains the interest in having omega-3 as part of the diet. Other studies have shown a broad benefit of the Mediterranean diet.
We know this diet: Fish, especially fatty fish; meat, but not every day, and white meat is best; and fruits and vegetables. In addition, exercise and stay hydrated. All of this can help patients who want to use diet as a means to control their disease. And, as I said earlier, studies have shown that omega-3 supplements have beneficial effects. These are essential polyunsaturated fatty acids, which can help control the disease and joint symptoms.
We also provide some exclusionary recommendations. Not all studies are done well, but it’s clear that there are no major benefits – in fact, no benefit at all – from vegan diets, gluten-free diets, or dairy-free diets. And with these diets, patients run the risk of developing deficiencies, so it’s important that patients are aware of this. We also have to keep in mind that exclusion diets can increase social isolation. Patients need to take part in meals; such gatherings are times for sharing and having social interactions. And I would say that they must be told that there are no data in the literature in support of these diets. But if they ever insist on this kind of intervention, I think that it’s better to advise them to do it under the supervision of a dietician and nutritionist, especially to prevent the development of deficiencies. We’re talking about deficiencies in things like calcium, vitamin B12, and selenium.
Conclusion
As you can see, we have positive recommendations when the patient wants to do something beyond pharmacologic treatment: the Mediterranean diet and omega-3 supplements. And we have negative recommendations, marked by a warning about the risk of developing deficiencies. But I think we all understand the importance of paying close attention to how our patients are experimenting with food. Their diets and eating habits can give us ideas for research and reviews that could allow us to deepen our understanding of the effect of diet on disease, because currently, the quality of the data on some of the diets and types of dietary interventions out there is rather tenuous.
Thank you for listening. I’d also like to thank Claire Daien, MD, PhD, for conducting this project with me so that we could come up with all of these recommendations. I’m also grateful to the following nutrition societies and associations who were our partners: the French Society of Nutrition, the French-Speaking Society of Clinical Nutrition and Metabolism, the French Association for the Study of Obesity, and the French Association of Dieticians and Nutritionists. And patient associations, too, must be recognized, as some of their members participated: the French National Association Against Rheumatoid Arthritis, the French Spondyloarthritis Association, and the French Association for Polyarthritis and Chronic Inflammatory Rheumatic Diseases.
This interview is a translation of a video blog that first appeared on Medscape France. It has been edited for clarity.
Weight loss, omega-3 supplements, the Mediterranean diet? What about exclusion diets? Jérémie Sellam, MD, PhD, from Saint-Antoine Hospital in Paris, summarizes the key points of the first set of dietary recommendations of the French Society for Rheumatology.
Transcript
Jérémie Sellam, MD, PhD: Hello, everyone. I’m Professor Jérémie Sellam. I’m a rheumatologist at Saint-Antoine Hospital, which is affiliated with the Sorbonne University in Paris. And I was fortunate enough to coordinate France’s first set of dietary recommendations – in fact, the world’s first set of dietary recommendations – for patients with chronic inflammatory rheumatic diseases. I worked on this project with Claire Daien, MD, PhD, who’s a rheumatologist at Montpellier University Hospital.
The idea of coming up with dietary recommendations for patients with inflammatory rheumatic diseases came, quite simply, from our clinical practice. We see that when patients learn they have polyarthritis or spondyloarthritis, they start to experiment with different diets. Many patients start exclusion diets and experiment in all sorts of ways with the food they eat. And although rheumatologists have been able to find some information here and there in the literature, they’ve been pretty much on their own when trying to come up with advice to give their patients. It was to address this issue that [Dr.] Daien and I set out to form a working group. Because when patients aren’t able to get sound advice and authoritative guidance from their doctors, medical associations, or patient advocacy organizations, they often look for information online, and that information is not always reliable or validated.
This group was made up of rheumatologists, some who work at hospitals and others in private practice. Also involved were physician nutrition specialists and registered dietitians. Operating under the auspices of the French Society for Rheumatology, these multidisciplinary experts conducted out a systematic literature review for the purpose of establishing and drafting recommendations. The result was a declaration of eight general principles and nine recommendations.
General principles
The first of the general principles states that nutritional advice is not a substitute for the pharmacologic treatment of chronic inflammatory rheumatic diseases. As you know, whether it’s methotrexate or biologics, pharmacologic treatments are essential for the proper management of chronic inflammatory rheumatic diseases. We know that these medications have an anti-inflammatory effect, reduce pain, and – particularly in the case of rheumatoid arthritis – have a structural effect. In other words, they prevent joint deterioration and destruction. Now, I can tell you that there’s currently no diet, and no dietary supplement, that has proven to be structurally effective. So, yes, dietary intervention might turn out to be promising for patients with chronic inflammatory rheumatic diseases, but pharmacologic treatment must still be part of the picture.
Another general principle emphasizes that dietary intervention is a way for patients to be actively involved in the overall care of their disease, beyond just taking their medication. We know that patients, when they suffer from chronic diseases, are looking for something more, beyond just taking medications. Encouraging them to take an interest in their diet, asking them about what they eat, giving them advice, and supporting their desire to become involved in this aspect of their treatment plan can give them a sense of empowerment.
Dietary interventions can have articular effects, and I’m going to speak about which interventions you can propose, but also which can be beneficial in terms of cardiovascular health and bone health. All of this is based on the literature. In these recommendations, we’ve taken into account not only laboratory experiments – where this or that diet is given to a mouse with arthritis – but also reviewed randomized controlled trials that compare an intervention group with a control group. This is the benchmark we used to determine whether or not a diet should be recommended.
The recommendations
As for the recommendations themselves, we wanted to start off by emphasizing weight loss and what can be called weight-loss support. There’s a link between obesity and the risk of developing rheumatoid arthritis, and also psoriatic arthropathy. And the more overweight a patient is, the more active their disease. In other words, patients with obesity are going to experience more pain, more instances of wakefulness, and more morning stiffness than their normal-weight peers. They’re also going to show symptoms that suggest that disease activity is not controlled well.
Several randomized controlled studies have shown that weight loss will improve systemic joint symptoms. In one particular study, patients with psoriatic arthropathy were started on [tumor necrosis factor] inhibitor therapy and one group followed a prescribed diet and the other had no restrictions on eating. More patients in the diet group than in the no-diet group achieved minimal disease activity. Of course, in some cases – for example, patients with complicated morbid obesity – it might be necessary to have a discussion about bariatric surgery.
But practically speaking, how does one proceed? First of all, patients should be weighed at each visit and, if they’re overweight or obese, the subject should be broached. But even after that conversation, the reality remains that it’s not easy to lose weight. So in the recommendations, we focused on the fact that it shouldn’t be left to the rheumatologist or treating physician alone to handle this challenging aspect of treatment. They should incorporate dietary and nutritional care by reaching out to a dietician or, in the case of complicated obesity – especially when the BMI is higher than 35 kg/m2 – they can refer patients to a nutrition expert who can manage the patient’s obesity, come up with a weight-loss plan, and handle any complications that might arise.
We don’t speak about a low-calorie diet in the recommendations because a diet has a beginning and an end and, quite often, patients regain weight after stopping a diet. Instead, we speak about weight-loss support to point out that weight loss maintained through dietary changes brings about long-term control of disease activity.
In addition, we make two positive recommendations, which overlap, that can help patients control their disease: a Mediterranean diet and omega-3 supplements. One study showed that after participants with rheumatoid arthritis followed the Mediterranean diet for 1 year, those who also took omega-3 fish oil supplements were twice as likely to achieve remission (40% vs. 20%). This explains the interest in having omega-3 as part of the diet. Other studies have shown a broad benefit of the Mediterranean diet.
We know this diet: Fish, especially fatty fish; meat, but not every day, and white meat is best; and fruits and vegetables. In addition, exercise and stay hydrated. All of this can help patients who want to use diet as a means to control their disease. And, as I said earlier, studies have shown that omega-3 supplements have beneficial effects. These are essential polyunsaturated fatty acids, which can help control the disease and joint symptoms.
We also provide some exclusionary recommendations. Not all studies are done well, but it’s clear that there are no major benefits – in fact, no benefit at all – from vegan diets, gluten-free diets, or dairy-free diets. And with these diets, patients run the risk of developing deficiencies, so it’s important that patients are aware of this. We also have to keep in mind that exclusion diets can increase social isolation. Patients need to take part in meals; such gatherings are times for sharing and having social interactions. And I would say that they must be told that there are no data in the literature in support of these diets. But if they ever insist on this kind of intervention, I think that it’s better to advise them to do it under the supervision of a dietician and nutritionist, especially to prevent the development of deficiencies. We’re talking about deficiencies in things like calcium, vitamin B12, and selenium.
Conclusion
As you can see, we have positive recommendations when the patient wants to do something beyond pharmacologic treatment: the Mediterranean diet and omega-3 supplements. And we have negative recommendations, marked by a warning about the risk of developing deficiencies. But I think we all understand the importance of paying close attention to how our patients are experimenting with food. Their diets and eating habits can give us ideas for research and reviews that could allow us to deepen our understanding of the effect of diet on disease, because currently, the quality of the data on some of the diets and types of dietary interventions out there is rather tenuous.
Thank you for listening. I’d also like to thank Claire Daien, MD, PhD, for conducting this project with me so that we could come up with all of these recommendations. I’m also grateful to the following nutrition societies and associations who were our partners: the French Society of Nutrition, the French-Speaking Society of Clinical Nutrition and Metabolism, the French Association for the Study of Obesity, and the French Association of Dieticians and Nutritionists. And patient associations, too, must be recognized, as some of their members participated: the French National Association Against Rheumatoid Arthritis, the French Spondyloarthritis Association, and the French Association for Polyarthritis and Chronic Inflammatory Rheumatic Diseases.
Docs react: NyQuil chicken and endless eye mucus
It’s the season of love. In that spirit, Lean in and get a whiff of the latest good, bad, and ugly videos making the rounds on the internet’s most perplexing platform. But don’t get too close; these videos are especially ripe.
The bad: NyQuil chicken
You know something bad has happened when your TikTok search ends with a warning from the app that says “Learn how to recognize harmful trends and hoaxes.” That’s what shows up now when you try to find out what the “NyQuil chicken” or “sleepy chicken” trend is (or was) all about.
TikTok videos, including this one from TikTok user @janelleandkate, show users trying out a trend meant to cook up a meal that will also cure your cold symptoms. The trend involves cooking chicken in a pan full of the cold and flu medicine NyQuil. The NyQuil chicken idea stems from a Twitter meme from 2017, so it is possible that some of the recent videos are fake (blue food coloring is easy to get, people).
However, in the instance that people believe the videos to be real and want to try the trend out, it is important to warn that this shouldn’t be attempted.
Aaron Hartman, MD, assistant clinical professor of family medicine at Virginia Commonwealth University, told the website Mic about the trend’s dangers: “When you cook cough medicine like NyQuil, however, you boil off the water and alcohol in it, leaving the chicken saturated with a super concentrated amount of drugs in the meat. If you ate one of those cutlets completely cooked, it’d be as if you’re actually consuming a quarter to half a bottle of NyQuil.”
And that’s not good for anyone. What ever happened to an old fashioned herb marinade?
The good: Can you fart yourself blind? Doc explains
It’s something we’ve all wondered about, right?
TikTok and YouTube’s mainstay plastic surgeon Anthony Youn, MD, took it upon himself to reply to a comment saying “I once farted so hard I went blind for 3 minutes.” This phenomenon, according to Dr. Youn, is very rare, but not impossible, though we wouldn’t exactly want to try it for ourselves.
In the humorous (but very informative!) video, Dr. Youn explains that particularly pungent flatulence can contain large amounts of hydrogen sulfide, a gas that is known for smelling like rotten eggs. According to the Occupational Safety and Health Administration, hydrogen sulfide is produced in a number of industries, like oil and gas refining, mining, and paper processing. Exposure to higher concentrations of hydrogen sulfide can be dangerous, with prolonged exposure at a 2-5 parts per million (ppm) concentration causing nausea, headaches, and airway problems in some asthma patients. At very high concentrations, it can be fatal.
Thankfully, a person’s gas is not at all that dangerous. When it comes to the commentor’s claim, Dr. Youn says that something else hydrogen sulfide can do is reduce blood pressure.
“If it reduces blood pressure to the central retinal artery,” Dr. Youn says, “your silent but deadly toot could theoretically make you go blind.”
Thank goodness we can lay that question to rest.
The ugly: Eye boogers from hell
Get a look at this!
This video from @mikaylaadiorr has amassed over 8 million likes and over 89,000 comments, and shows someone, who we can assume is Mikayla, pulling some sort of long string-like material out of the corner of her eye. It’s like a clown’s never-ending handkerchief, only goopy.
These mucus eye strings are caused by untreated eye conditions, like dry eye or pink eye (conjunctivitis), but pulling the mucus out is actually a symptom of what is called mucus fishing syndrome. As you know, our eyes are covered in layers of mucus and tears, which keeps our eyeballs lubricated and also protects us from bacteria and viruses. It’s possible to dry out the eyes by pulling some mucus off, but our eyes aren’t big fans of that, so they’ll create more mucus to keep from drying out.
A person who might get a bit addicted to pulling the strings out has likely developed mucus fishing syndrome, which is considered a body-focused repetitive behavior (BFRB); other BFRBs include skin-picking (dermatillomania) or picking hairs out (trichotillomania).
Popular TikToker and Oregon ophthalmologist Will Flanary, MD, aka Dr. Glaucomflecken, responded to the videos, which have been encouraging others to try it.
“This is called mucus fishing syndrome,” the ophthalmologist explained via text captions in his video. “The trauma from pulling mucus out of your eye causes more mucus to form. You get caught in a never-ending cycle that gets worse over time. So…stop it.”
Fingers off the mucus, people.
A version of this article first appeared on Medscape.com.
It’s the season of love. In that spirit, Lean in and get a whiff of the latest good, bad, and ugly videos making the rounds on the internet’s most perplexing platform. But don’t get too close; these videos are especially ripe.
The bad: NyQuil chicken
You know something bad has happened when your TikTok search ends with a warning from the app that says “Learn how to recognize harmful trends and hoaxes.” That’s what shows up now when you try to find out what the “NyQuil chicken” or “sleepy chicken” trend is (or was) all about.
TikTok videos, including this one from TikTok user @janelleandkate, show users trying out a trend meant to cook up a meal that will also cure your cold symptoms. The trend involves cooking chicken in a pan full of the cold and flu medicine NyQuil. The NyQuil chicken idea stems from a Twitter meme from 2017, so it is possible that some of the recent videos are fake (blue food coloring is easy to get, people).
However, in the instance that people believe the videos to be real and want to try the trend out, it is important to warn that this shouldn’t be attempted.
Aaron Hartman, MD, assistant clinical professor of family medicine at Virginia Commonwealth University, told the website Mic about the trend’s dangers: “When you cook cough medicine like NyQuil, however, you boil off the water and alcohol in it, leaving the chicken saturated with a super concentrated amount of drugs in the meat. If you ate one of those cutlets completely cooked, it’d be as if you’re actually consuming a quarter to half a bottle of NyQuil.”
And that’s not good for anyone. What ever happened to an old fashioned herb marinade?
The good: Can you fart yourself blind? Doc explains
It’s something we’ve all wondered about, right?
TikTok and YouTube’s mainstay plastic surgeon Anthony Youn, MD, took it upon himself to reply to a comment saying “I once farted so hard I went blind for 3 minutes.” This phenomenon, according to Dr. Youn, is very rare, but not impossible, though we wouldn’t exactly want to try it for ourselves.
In the humorous (but very informative!) video, Dr. Youn explains that particularly pungent flatulence can contain large amounts of hydrogen sulfide, a gas that is known for smelling like rotten eggs. According to the Occupational Safety and Health Administration, hydrogen sulfide is produced in a number of industries, like oil and gas refining, mining, and paper processing. Exposure to higher concentrations of hydrogen sulfide can be dangerous, with prolonged exposure at a 2-5 parts per million (ppm) concentration causing nausea, headaches, and airway problems in some asthma patients. At very high concentrations, it can be fatal.
Thankfully, a person’s gas is not at all that dangerous. When it comes to the commentor’s claim, Dr. Youn says that something else hydrogen sulfide can do is reduce blood pressure.
“If it reduces blood pressure to the central retinal artery,” Dr. Youn says, “your silent but deadly toot could theoretically make you go blind.”
Thank goodness we can lay that question to rest.
The ugly: Eye boogers from hell
Get a look at this!
This video from @mikaylaadiorr has amassed over 8 million likes and over 89,000 comments, and shows someone, who we can assume is Mikayla, pulling some sort of long string-like material out of the corner of her eye. It’s like a clown’s never-ending handkerchief, only goopy.
These mucus eye strings are caused by untreated eye conditions, like dry eye or pink eye (conjunctivitis), but pulling the mucus out is actually a symptom of what is called mucus fishing syndrome. As you know, our eyes are covered in layers of mucus and tears, which keeps our eyeballs lubricated and also protects us from bacteria and viruses. It’s possible to dry out the eyes by pulling some mucus off, but our eyes aren’t big fans of that, so they’ll create more mucus to keep from drying out.
A person who might get a bit addicted to pulling the strings out has likely developed mucus fishing syndrome, which is considered a body-focused repetitive behavior (BFRB); other BFRBs include skin-picking (dermatillomania) or picking hairs out (trichotillomania).
Popular TikToker and Oregon ophthalmologist Will Flanary, MD, aka Dr. Glaucomflecken, responded to the videos, which have been encouraging others to try it.
“This is called mucus fishing syndrome,” the ophthalmologist explained via text captions in his video. “The trauma from pulling mucus out of your eye causes more mucus to form. You get caught in a never-ending cycle that gets worse over time. So…stop it.”
Fingers off the mucus, people.
A version of this article first appeared on Medscape.com.
It’s the season of love. In that spirit, Lean in and get a whiff of the latest good, bad, and ugly videos making the rounds on the internet’s most perplexing platform. But don’t get too close; these videos are especially ripe.
The bad: NyQuil chicken
You know something bad has happened when your TikTok search ends with a warning from the app that says “Learn how to recognize harmful trends and hoaxes.” That’s what shows up now when you try to find out what the “NyQuil chicken” or “sleepy chicken” trend is (or was) all about.
TikTok videos, including this one from TikTok user @janelleandkate, show users trying out a trend meant to cook up a meal that will also cure your cold symptoms. The trend involves cooking chicken in a pan full of the cold and flu medicine NyQuil. The NyQuil chicken idea stems from a Twitter meme from 2017, so it is possible that some of the recent videos are fake (blue food coloring is easy to get, people).
However, in the instance that people believe the videos to be real and want to try the trend out, it is important to warn that this shouldn’t be attempted.
Aaron Hartman, MD, assistant clinical professor of family medicine at Virginia Commonwealth University, told the website Mic about the trend’s dangers: “When you cook cough medicine like NyQuil, however, you boil off the water and alcohol in it, leaving the chicken saturated with a super concentrated amount of drugs in the meat. If you ate one of those cutlets completely cooked, it’d be as if you’re actually consuming a quarter to half a bottle of NyQuil.”
And that’s not good for anyone. What ever happened to an old fashioned herb marinade?
The good: Can you fart yourself blind? Doc explains
It’s something we’ve all wondered about, right?
TikTok and YouTube’s mainstay plastic surgeon Anthony Youn, MD, took it upon himself to reply to a comment saying “I once farted so hard I went blind for 3 minutes.” This phenomenon, according to Dr. Youn, is very rare, but not impossible, though we wouldn’t exactly want to try it for ourselves.
In the humorous (but very informative!) video, Dr. Youn explains that particularly pungent flatulence can contain large amounts of hydrogen sulfide, a gas that is known for smelling like rotten eggs. According to the Occupational Safety and Health Administration, hydrogen sulfide is produced in a number of industries, like oil and gas refining, mining, and paper processing. Exposure to higher concentrations of hydrogen sulfide can be dangerous, with prolonged exposure at a 2-5 parts per million (ppm) concentration causing nausea, headaches, and airway problems in some asthma patients. At very high concentrations, it can be fatal.
Thankfully, a person’s gas is not at all that dangerous. When it comes to the commentor’s claim, Dr. Youn says that something else hydrogen sulfide can do is reduce blood pressure.
“If it reduces blood pressure to the central retinal artery,” Dr. Youn says, “your silent but deadly toot could theoretically make you go blind.”
Thank goodness we can lay that question to rest.
The ugly: Eye boogers from hell
Get a look at this!
This video from @mikaylaadiorr has amassed over 8 million likes and over 89,000 comments, and shows someone, who we can assume is Mikayla, pulling some sort of long string-like material out of the corner of her eye. It’s like a clown’s never-ending handkerchief, only goopy.
These mucus eye strings are caused by untreated eye conditions, like dry eye or pink eye (conjunctivitis), but pulling the mucus out is actually a symptom of what is called mucus fishing syndrome. As you know, our eyes are covered in layers of mucus and tears, which keeps our eyeballs lubricated and also protects us from bacteria and viruses. It’s possible to dry out the eyes by pulling some mucus off, but our eyes aren’t big fans of that, so they’ll create more mucus to keep from drying out.
A person who might get a bit addicted to pulling the strings out has likely developed mucus fishing syndrome, which is considered a body-focused repetitive behavior (BFRB); other BFRBs include skin-picking (dermatillomania) or picking hairs out (trichotillomania).
Popular TikToker and Oregon ophthalmologist Will Flanary, MD, aka Dr. Glaucomflecken, responded to the videos, which have been encouraging others to try it.
“This is called mucus fishing syndrome,” the ophthalmologist explained via text captions in his video. “The trauma from pulling mucus out of your eye causes more mucus to form. You get caught in a never-ending cycle that gets worse over time. So…stop it.”
Fingers off the mucus, people.
A version of this article first appeared on Medscape.com.
Enough is enough: the pandemic and loss of female oncologists
Imagine this: As a young girl, you decide you want to become a doctor when you grow up. You spend countless hours studying, researching, and volunteering to eventually make it into medical school. Four years later, you graduate top of your class and match into your first-choice residency program. You are so proud of yourself!
During your last year of residency, a pandemic takes the entire world by storm. You persevere through your last 14 months of residency that included additional time in the ICU, not seeing your colleagues, and interviewing for your new job all from your own living room. After all of this, you finally get to start doing what you have been waiting to do for the past decade: train with the brilliant minds in hematology and oncology.
All of a sudden, You start to question: If these incredible women have decided that the sacrifice this career requires is too much, then (1) How will I survive? and (2) Did I make a huge mistake in my career decision? Spoiler alert: This girl is me.
The World Health Organization defines burnout as a “syndrome conceptualized as resulting from chronic workplace stress that has not been successfully managed. It is characterized by energy depletion or exhaustion, increased mental distance from one’s job, and reduced professional efficacy.”
We know that 33% of oncologists are feeling burned out right now, according to the Medscape National Physician Burnout & Suicide Report 2021. Of the 51% of female physicians that are burned out, work-life balance has been identified as the biggest workplace concern to them. Research has shown that hours per week devoted to direct patient care is the dominant predictor of burnout for practicing oncologists. But in academic oncology, that is followed by grant deadlines, manuscript rejections, and the constant reminders that you are a new face in oncology, a specialty that was previously male-dominated.
In less than a year, we have had several key female oncologists leave our cancer center. While some made the decision to retire early, two of them chose to pivot their careers and leave clinical medicine to assist with drug development and clinical trials. Although this is extremely important work for cancer care, I was shocked to hear that these amazing and successful clinicians were choosing to remove all direct patient care from their practice, when for many of them, patient care was what motivated them to pursue medicine in the first place. They were loved by their patients, respected as researchers, and well known as educators within the division.
One shared that she no longer felt like she could be a good mother, wife, or daughter with what was currently being demanded of her to have a successful academic career. In hearing this news, I was saddened to have to say goodbye to a mentor of mine and immediately started second-guessing my career choice. I felt that my goal of having an impactful career and prosperous home life was not only unattainable but potentially unrealistic.
While we know that female physicians already experience a greater degree of burnout, the pandemic has only added fuel to the fire. This is especially true in cancer care. It has been estimated that new cancer diagnosis have decreased by as much as 23% since the beginning of the pandemic. This delay in diagnosis will lead to patients presenting with more advanced disease, busier clinic schedules, and worsened clinical outcomes for years to come. With no end in sight, I worry what this will mean for women currently in oncology, in addition to those in training or deciding if they should pursue this as a career.
Extrapolating evidence from prior epidemics, physicians are at increased risk for burnout due to immediate and long-term effects from this pandemic. We need to act now to not only continue addressing previously existing individual and organizational causes of burnout but also develop strategies to provide support for the COVID-19–specific impacts on oncologists’ well-being. An editorial published by the American Society of Clinical Oncology provides helpful suggestions on how to do this.
A recent cross-sectional survey found that 22% of academic female oncologists were likely or very likely to pursue a career outside of academia in the next 5 years. Losing these women would be detrimental to the field. This would mean a significant number of patients losing their long-term oncologists with whom they have years of care, trainees losing their professional and research mentors to guide and help mold them into successful independent practitioners and researchers, and arguably most important, little girls losing role models to show them that regardless of their gender, they can become an oncologist.Dr. Poterala is a current hematology and oncology fellow at the University of Wisconsin Carbone Cancer Center, Madison. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Imagine this: As a young girl, you decide you want to become a doctor when you grow up. You spend countless hours studying, researching, and volunteering to eventually make it into medical school. Four years later, you graduate top of your class and match into your first-choice residency program. You are so proud of yourself!
During your last year of residency, a pandemic takes the entire world by storm. You persevere through your last 14 months of residency that included additional time in the ICU, not seeing your colleagues, and interviewing for your new job all from your own living room. After all of this, you finally get to start doing what you have been waiting to do for the past decade: train with the brilliant minds in hematology and oncology.
All of a sudden, You start to question: If these incredible women have decided that the sacrifice this career requires is too much, then (1) How will I survive? and (2) Did I make a huge mistake in my career decision? Spoiler alert: This girl is me.
The World Health Organization defines burnout as a “syndrome conceptualized as resulting from chronic workplace stress that has not been successfully managed. It is characterized by energy depletion or exhaustion, increased mental distance from one’s job, and reduced professional efficacy.”
We know that 33% of oncologists are feeling burned out right now, according to the Medscape National Physician Burnout & Suicide Report 2021. Of the 51% of female physicians that are burned out, work-life balance has been identified as the biggest workplace concern to them. Research has shown that hours per week devoted to direct patient care is the dominant predictor of burnout for practicing oncologists. But in academic oncology, that is followed by grant deadlines, manuscript rejections, and the constant reminders that you are a new face in oncology, a specialty that was previously male-dominated.
In less than a year, we have had several key female oncologists leave our cancer center. While some made the decision to retire early, two of them chose to pivot their careers and leave clinical medicine to assist with drug development and clinical trials. Although this is extremely important work for cancer care, I was shocked to hear that these amazing and successful clinicians were choosing to remove all direct patient care from their practice, when for many of them, patient care was what motivated them to pursue medicine in the first place. They were loved by their patients, respected as researchers, and well known as educators within the division.
One shared that she no longer felt like she could be a good mother, wife, or daughter with what was currently being demanded of her to have a successful academic career. In hearing this news, I was saddened to have to say goodbye to a mentor of mine and immediately started second-guessing my career choice. I felt that my goal of having an impactful career and prosperous home life was not only unattainable but potentially unrealistic.
While we know that female physicians already experience a greater degree of burnout, the pandemic has only added fuel to the fire. This is especially true in cancer care. It has been estimated that new cancer diagnosis have decreased by as much as 23% since the beginning of the pandemic. This delay in diagnosis will lead to patients presenting with more advanced disease, busier clinic schedules, and worsened clinical outcomes for years to come. With no end in sight, I worry what this will mean for women currently in oncology, in addition to those in training or deciding if they should pursue this as a career.
Extrapolating evidence from prior epidemics, physicians are at increased risk for burnout due to immediate and long-term effects from this pandemic. We need to act now to not only continue addressing previously existing individual and organizational causes of burnout but also develop strategies to provide support for the COVID-19–specific impacts on oncologists’ well-being. An editorial published by the American Society of Clinical Oncology provides helpful suggestions on how to do this.
A recent cross-sectional survey found that 22% of academic female oncologists were likely or very likely to pursue a career outside of academia in the next 5 years. Losing these women would be detrimental to the field. This would mean a significant number of patients losing their long-term oncologists with whom they have years of care, trainees losing their professional and research mentors to guide and help mold them into successful independent practitioners and researchers, and arguably most important, little girls losing role models to show them that regardless of their gender, they can become an oncologist.Dr. Poterala is a current hematology and oncology fellow at the University of Wisconsin Carbone Cancer Center, Madison. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Imagine this: As a young girl, you decide you want to become a doctor when you grow up. You spend countless hours studying, researching, and volunteering to eventually make it into medical school. Four years later, you graduate top of your class and match into your first-choice residency program. You are so proud of yourself!
During your last year of residency, a pandemic takes the entire world by storm. You persevere through your last 14 months of residency that included additional time in the ICU, not seeing your colleagues, and interviewing for your new job all from your own living room. After all of this, you finally get to start doing what you have been waiting to do for the past decade: train with the brilliant minds in hematology and oncology.
All of a sudden, You start to question: If these incredible women have decided that the sacrifice this career requires is too much, then (1) How will I survive? and (2) Did I make a huge mistake in my career decision? Spoiler alert: This girl is me.
The World Health Organization defines burnout as a “syndrome conceptualized as resulting from chronic workplace stress that has not been successfully managed. It is characterized by energy depletion or exhaustion, increased mental distance from one’s job, and reduced professional efficacy.”
We know that 33% of oncologists are feeling burned out right now, according to the Medscape National Physician Burnout & Suicide Report 2021. Of the 51% of female physicians that are burned out, work-life balance has been identified as the biggest workplace concern to them. Research has shown that hours per week devoted to direct patient care is the dominant predictor of burnout for practicing oncologists. But in academic oncology, that is followed by grant deadlines, manuscript rejections, and the constant reminders that you are a new face in oncology, a specialty that was previously male-dominated.
In less than a year, we have had several key female oncologists leave our cancer center. While some made the decision to retire early, two of them chose to pivot their careers and leave clinical medicine to assist with drug development and clinical trials. Although this is extremely important work for cancer care, I was shocked to hear that these amazing and successful clinicians were choosing to remove all direct patient care from their practice, when for many of them, patient care was what motivated them to pursue medicine in the first place. They were loved by their patients, respected as researchers, and well known as educators within the division.
One shared that she no longer felt like she could be a good mother, wife, or daughter with what was currently being demanded of her to have a successful academic career. In hearing this news, I was saddened to have to say goodbye to a mentor of mine and immediately started second-guessing my career choice. I felt that my goal of having an impactful career and prosperous home life was not only unattainable but potentially unrealistic.
While we know that female physicians already experience a greater degree of burnout, the pandemic has only added fuel to the fire. This is especially true in cancer care. It has been estimated that new cancer diagnosis have decreased by as much as 23% since the beginning of the pandemic. This delay in diagnosis will lead to patients presenting with more advanced disease, busier clinic schedules, and worsened clinical outcomes for years to come. With no end in sight, I worry what this will mean for women currently in oncology, in addition to those in training or deciding if they should pursue this as a career.
Extrapolating evidence from prior epidemics, physicians are at increased risk for burnout due to immediate and long-term effects from this pandemic. We need to act now to not only continue addressing previously existing individual and organizational causes of burnout but also develop strategies to provide support for the COVID-19–specific impacts on oncologists’ well-being. An editorial published by the American Society of Clinical Oncology provides helpful suggestions on how to do this.
A recent cross-sectional survey found that 22% of academic female oncologists were likely or very likely to pursue a career outside of academia in the next 5 years. Losing these women would be detrimental to the field. This would mean a significant number of patients losing their long-term oncologists with whom they have years of care, trainees losing their professional and research mentors to guide and help mold them into successful independent practitioners and researchers, and arguably most important, little girls losing role models to show them that regardless of their gender, they can become an oncologist.Dr. Poterala is a current hematology and oncology fellow at the University of Wisconsin Carbone Cancer Center, Madison. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Machine Learning: the Future of Total Knee Replacement
Total knee replacement (TKR) is one of the most common surgeries worldwide, with > 1 million performed last year. Many patients have seen tremendous benefit from TKR; however, studies have shown that up to 20% of patients are not satisfied with the results of this procedure.1,2 This equates to about 200,000 patients worldwide every year who are dissatisfied. This is a huge concern to patients, surgeons, implant manufacturers, hospitals, and health care payers.
Many attempts to improve satisfaction in TKR have been tried, including computer navigation, minimally invasive surgery, rotating platform prostheses, gender-specific implants, different materials, changes in pain management, and revised postoperative rehabilitation.3-7 However, these efforts show no significant improvement in satisfaction.
The most common method of TKR today involves using a long rod placed through a drill hole in the femur. Standardized cuts on the femur and tibia are made through metal cutting blocks. Only metal mechanical instruments are used to perform the surgery, and all patients are aligned the same. However, anatomic studies have shown that patient anatomy in 3 dimensions (3D) varies widely from patient to patient.8 Our current technique seems far removed from modern engineering, where we now see extensive use of artificial intelligence (AI) to improve outcomes.
Machine learning (ML) is considered a subset of AI that involves the use of various computer algorithms. ML allows the computer to learn and continually improve analysis of data. Large sets of inputs and outputs are used to train the machine to make autonomous recommendations or decisions.9,10
Seven years ago, our team at the Phoenix Veteran Affairs Medical Center in Arizona published a randomized controlled trial evaluating a new, individualized alignment technique for TKR.11 This method used 3D-printed guides made from an MRI of an individual patient’s knee. Instead of aligning all knee replacements the same, each patient was aligned according to their unique anatomy. Compared with the conventional alignment technique, the newer technique showed significant improvement in all outcome scores and range of motion at 2 years postsurgery. There has been a great deal of interest in individualizing TKR, and many articles and techniques have followed.12
Our surgical technique has evolved since publishing our trial. Currently, knee X-rays are digitally templated for each patient. Understanding the patient’s preoperative alignment can then assist in planning a TKR in 3D. A plastic 3D-printed guide is manufactured in Belgium, shipped to the US, sterilized, and used in surgery. These guides fit accurately on the patient’s anatomy and allow precise angles and depth of resection for each surgical bone cut. Our research has shown that these guides are accurate to within 0.5° and 0.5 mm for the bone cuts performed in surgery. After surgery, we track patient-reported outcomes (PROs), which can then be used in ML or logistic regression analysis to determine alignment factors that contribute to the best outcome.13
Soon, use of a robot will take the place of the templating and preplanning, allowing the 3D plan to be immediately produced in surgery by the software installed in the robot.14-16 Each patient’s preoperative alignment can then be immediately compared with the postoperative result, and smartphone technology can allow a patient to input their PRO after the surgery is healed.17
Collecting all this information in a large database can allow ML analyses of the outcomes and individual alignment.14-17 As the factors contributing to the best clinical results are determined, the computer can be programmed to learn how to make the best recommendations for alignment of each patient, which can be incorporated into the robotic platform for each surgery. Also pre- and postoperative factors can be added to the ML platform so we can identify the best preoperative patient parameters, anticoagulation program postoperative rehabilitation program, etc, to help drive higher PROs and satisfaction.
Multiple surgical robots for TKR are now on the market. Orthopedic literature includes ML algorithms to improve outcomes after total hip arthroplasty.18 The EHR can be used to develop models to predict poor outcomes after TKR. Integrating these models into clinical decision support could improve patient selection, education, and satisfaction.19 AI for adult spinal surgery using predictive analytics can help surgeons better inform patients about outcomes after corrective surgery.20,21
With worldwide TKRs expected to exceed 3 million over the next decade, ML using large databases, robotic surgery, and PROs could be key to improving our TKR outcomes.22 This form of AI may reduce the large number of patients currently not satisfied with their knee replacement.
1. Baker PN, van der Meulen JH, Lewsey J, Gregg PJ; National Joint Registry for England and Wales. The role of pain and function in determining patient satisfaction after total knee replacement. Data from the National Joint Registry for England and Wales. J Bone Joint Surg Br. 2007;89(7):893-900. doi:10.1302/0301-620X.89B7.19091
2. Noble PC, Conditt MA, Cook KF, Mathis KB. The John Insall Award: patient expectations affect satisfaction with total knee arthroplasty. Clin Orthop Relat Res. 2006;452:35-43. doi:10.1097/01.blo.0000238825.63648.1e
3. Matziolis G, Krocker D, Weiss U, Tohtz S, Perka C. A prospective, randomized study of computer-assisted and conventional total knee arthroplasty. Three-dimensional evaluation of implant alignment and rotation. J Bone Joint Surg Am. 2007;89(2):236-243. doi:10.2106/JBJS.F.00386
4. Stulberg SD, Yaffe MA, Koo SS. Computer-assisted surgery versus manual total knee arthroplasty: a case-controlled study. J Bone Joint Surg Am. 2006;88(suppl 4):47-54. doi:10.2106/JBJS.F.00698
5. Kalisvaart MM, Pagnano MW, Trousdale RT, Stuart MJ, Hanssen AD. Randomized clinical trial of rotating-platform and fixed-bearing total knee arthroplasty: no clinically detectable differences at five years. J Bone Joint Surg Am. 2012;94(6):481-489. doi:10.2106/JBJS.K.00315
6. Wülker N, Lambermont JP, Sacchetti L, Lazaró JG, Nardi J. A prospective randomized study of minimally invasive total knee arthroplasty compared with conventional surgery. J Bone Joint Surg Am. 2010;92(7):1584-1590. doi:10.2106/JBJS.H.01070
7. Thomsen MG, Husted H, Bencke J, Curtis D, Holm G, Troelsen A. Do we need a gender-specific total knee replacement? A randomised controlled trial comparing a high-flex and a gender-specific posterior design. J Bone Joint Surg Br. 2012;94(6):787-792. doi:10.1302/0301-620X.94B6.28781
8. Eckhoff D, Hogan C, DiMatteo L, Robinson M, Bach J. Difference between the epicondylar and cylindrical axis of the knee. Clin Orthop Relat Res. 2007;461:238-244. doi:10.1097/BLO.0b013e318112416b
9. Martin RK, Ley C, Pareek A, Groll A, Tischer T, Seil R. Artificial intelligence and machine learning: an introduction for orthopaedic surgeons [published online ahead of print, 2021 Sep 15]. Knee Surg Sports Traumatol Arthrosc. 2021;10.1007/s00167-021-06741-2. doi:10.1007/s00167-021-06741-2
10. Helm JM, Swiergosz AM, Haeberle HS, et al. Machine Learning and Artificial Intelligence: Definitions, Applications, and Future Directions. Curr Rev Musculoskelet Med. 2020;13(1):69-76. doi:10.1007/s12178-020-09600-8
11. Dossett HG, Estrada NA, Swartz GJ, LeFevre GW, Kwasman BG. A randomised controlled trial of kinematically and mechanically aligned total knee replacements: two-year clinical results. Bone Joint J. 2014;96-B(7):907-913. doi:10.1302/0301-620X.96B7.32812
12. Rivière C, Iranpour F, Auvinet E, et al. Alignment options for total knee arthroplasty: a systematic review. Orthop Traumatol Surg Res. 2017;103(7):1047-1056. doi:10.1016/j.otsr.2017.07.010
13. Dossett HG. High reliability in total knee replacement surgery: is it possible? Orthop Proc. 2018;95-B(suppl 34):292-293.
14. Schock J, Truhn D, Abrar DB, et al. Automated analysis of alignment in long-leg radiographs by using a fully automated support system based on artificial intelligence. Radiol: Artif Intell. Dec 23, 2020;3(2). doi:10.1148/ryai.2020200198
15. Cabitza F, Locoro A, Banfi G. Machine learning in orthopedics: a literature review. Front Bioeng Biotechnol. 2018;6:75. Published 2018 Jun 27. doi:10.3389/fbioe.2018.00075
16. von Schacky CE, Wilhelm NJ, Schäfer VS, et al. Multitask deep learning for segmentation and classification of primary bone tumors on radiographs. Radiology. 2021;301(2):398-406. doi:10.1148/radiol.2021204531
17. Myers TG, Ramkumar PN, Ricciardi BF, Urish KL, Kipper J, Ketonis C. Artificial intelligence and orthopaedics: an introduction for clinicians. J Bone Joint Surg Am. 2020;102(9):830-840. doi:10.2106/JBJS.19.01128
18. Kunze KN, Karhade AV, Sadauskas AJ, Schwab JH, Levine BR. Development of machine learning algorithms to predict clinically meaningful improvement for the patient-reported health state after total hip arthroplasty. J Arthroplasty. 2020;35(8):2119-2123. doi:10.1016/j.arth.2020.03.019
19. Harris AHS, Kuo AC, Bowe TR, Manfredi L, Lalani NF, Giori NJ. Can machine learning methods produce accurate and easy-to-use preoperative prediction models of one-year improvements in pain and functioning after knee arthroplasty? J Arthroplasty. 2021;36(1):112-117.e6. doi:10.1016/j.arth.2020.07.026
20. Rasouli JJ, Shao J, Neifert S, et al. Artificial intelligence and robotics in spine surgery. Global Spine J. 2021;11(4):556-564. doi:10.1177/2192568220915718
21. Joshi RS, Haddad AF, Lau D, Ames CP. Artificial intelligence for adult spinal deformity. Neurospine. 2019;16(4):686-694. doi:10.14245/ns.1938414.207
22. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
Total knee replacement (TKR) is one of the most common surgeries worldwide, with > 1 million performed last year. Many patients have seen tremendous benefit from TKR; however, studies have shown that up to 20% of patients are not satisfied with the results of this procedure.1,2 This equates to about 200,000 patients worldwide every year who are dissatisfied. This is a huge concern to patients, surgeons, implant manufacturers, hospitals, and health care payers.
Many attempts to improve satisfaction in TKR have been tried, including computer navigation, minimally invasive surgery, rotating platform prostheses, gender-specific implants, different materials, changes in pain management, and revised postoperative rehabilitation.3-7 However, these efforts show no significant improvement in satisfaction.
The most common method of TKR today involves using a long rod placed through a drill hole in the femur. Standardized cuts on the femur and tibia are made through metal cutting blocks. Only metal mechanical instruments are used to perform the surgery, and all patients are aligned the same. However, anatomic studies have shown that patient anatomy in 3 dimensions (3D) varies widely from patient to patient.8 Our current technique seems far removed from modern engineering, where we now see extensive use of artificial intelligence (AI) to improve outcomes.
Machine learning (ML) is considered a subset of AI that involves the use of various computer algorithms. ML allows the computer to learn and continually improve analysis of data. Large sets of inputs and outputs are used to train the machine to make autonomous recommendations or decisions.9,10
Seven years ago, our team at the Phoenix Veteran Affairs Medical Center in Arizona published a randomized controlled trial evaluating a new, individualized alignment technique for TKR.11 This method used 3D-printed guides made from an MRI of an individual patient’s knee. Instead of aligning all knee replacements the same, each patient was aligned according to their unique anatomy. Compared with the conventional alignment technique, the newer technique showed significant improvement in all outcome scores and range of motion at 2 years postsurgery. There has been a great deal of interest in individualizing TKR, and many articles and techniques have followed.12
Our surgical technique has evolved since publishing our trial. Currently, knee X-rays are digitally templated for each patient. Understanding the patient’s preoperative alignment can then assist in planning a TKR in 3D. A plastic 3D-printed guide is manufactured in Belgium, shipped to the US, sterilized, and used in surgery. These guides fit accurately on the patient’s anatomy and allow precise angles and depth of resection for each surgical bone cut. Our research has shown that these guides are accurate to within 0.5° and 0.5 mm for the bone cuts performed in surgery. After surgery, we track patient-reported outcomes (PROs), which can then be used in ML or logistic regression analysis to determine alignment factors that contribute to the best outcome.13
Soon, use of a robot will take the place of the templating and preplanning, allowing the 3D plan to be immediately produced in surgery by the software installed in the robot.14-16 Each patient’s preoperative alignment can then be immediately compared with the postoperative result, and smartphone technology can allow a patient to input their PRO after the surgery is healed.17
Collecting all this information in a large database can allow ML analyses of the outcomes and individual alignment.14-17 As the factors contributing to the best clinical results are determined, the computer can be programmed to learn how to make the best recommendations for alignment of each patient, which can be incorporated into the robotic platform for each surgery. Also pre- and postoperative factors can be added to the ML platform so we can identify the best preoperative patient parameters, anticoagulation program postoperative rehabilitation program, etc, to help drive higher PROs and satisfaction.
Multiple surgical robots for TKR are now on the market. Orthopedic literature includes ML algorithms to improve outcomes after total hip arthroplasty.18 The EHR can be used to develop models to predict poor outcomes after TKR. Integrating these models into clinical decision support could improve patient selection, education, and satisfaction.19 AI for adult spinal surgery using predictive analytics can help surgeons better inform patients about outcomes after corrective surgery.20,21
With worldwide TKRs expected to exceed 3 million over the next decade, ML using large databases, robotic surgery, and PROs could be key to improving our TKR outcomes.22 This form of AI may reduce the large number of patients currently not satisfied with their knee replacement.
Total knee replacement (TKR) is one of the most common surgeries worldwide, with > 1 million performed last year. Many patients have seen tremendous benefit from TKR; however, studies have shown that up to 20% of patients are not satisfied with the results of this procedure.1,2 This equates to about 200,000 patients worldwide every year who are dissatisfied. This is a huge concern to patients, surgeons, implant manufacturers, hospitals, and health care payers.
Many attempts to improve satisfaction in TKR have been tried, including computer navigation, minimally invasive surgery, rotating platform prostheses, gender-specific implants, different materials, changes in pain management, and revised postoperative rehabilitation.3-7 However, these efforts show no significant improvement in satisfaction.
The most common method of TKR today involves using a long rod placed through a drill hole in the femur. Standardized cuts on the femur and tibia are made through metal cutting blocks. Only metal mechanical instruments are used to perform the surgery, and all patients are aligned the same. However, anatomic studies have shown that patient anatomy in 3 dimensions (3D) varies widely from patient to patient.8 Our current technique seems far removed from modern engineering, where we now see extensive use of artificial intelligence (AI) to improve outcomes.
Machine learning (ML) is considered a subset of AI that involves the use of various computer algorithms. ML allows the computer to learn and continually improve analysis of data. Large sets of inputs and outputs are used to train the machine to make autonomous recommendations or decisions.9,10
Seven years ago, our team at the Phoenix Veteran Affairs Medical Center in Arizona published a randomized controlled trial evaluating a new, individualized alignment technique for TKR.11 This method used 3D-printed guides made from an MRI of an individual patient’s knee. Instead of aligning all knee replacements the same, each patient was aligned according to their unique anatomy. Compared with the conventional alignment technique, the newer technique showed significant improvement in all outcome scores and range of motion at 2 years postsurgery. There has been a great deal of interest in individualizing TKR, and many articles and techniques have followed.12
Our surgical technique has evolved since publishing our trial. Currently, knee X-rays are digitally templated for each patient. Understanding the patient’s preoperative alignment can then assist in planning a TKR in 3D. A plastic 3D-printed guide is manufactured in Belgium, shipped to the US, sterilized, and used in surgery. These guides fit accurately on the patient’s anatomy and allow precise angles and depth of resection for each surgical bone cut. Our research has shown that these guides are accurate to within 0.5° and 0.5 mm for the bone cuts performed in surgery. After surgery, we track patient-reported outcomes (PROs), which can then be used in ML or logistic regression analysis to determine alignment factors that contribute to the best outcome.13
Soon, use of a robot will take the place of the templating and preplanning, allowing the 3D plan to be immediately produced in surgery by the software installed in the robot.14-16 Each patient’s preoperative alignment can then be immediately compared with the postoperative result, and smartphone technology can allow a patient to input their PRO after the surgery is healed.17
Collecting all this information in a large database can allow ML analyses of the outcomes and individual alignment.14-17 As the factors contributing to the best clinical results are determined, the computer can be programmed to learn how to make the best recommendations for alignment of each patient, which can be incorporated into the robotic platform for each surgery. Also pre- and postoperative factors can be added to the ML platform so we can identify the best preoperative patient parameters, anticoagulation program postoperative rehabilitation program, etc, to help drive higher PROs and satisfaction.
Multiple surgical robots for TKR are now on the market. Orthopedic literature includes ML algorithms to improve outcomes after total hip arthroplasty.18 The EHR can be used to develop models to predict poor outcomes after TKR. Integrating these models into clinical decision support could improve patient selection, education, and satisfaction.19 AI for adult spinal surgery using predictive analytics can help surgeons better inform patients about outcomes after corrective surgery.20,21
With worldwide TKRs expected to exceed 3 million over the next decade, ML using large databases, robotic surgery, and PROs could be key to improving our TKR outcomes.22 This form of AI may reduce the large number of patients currently not satisfied with their knee replacement.
1. Baker PN, van der Meulen JH, Lewsey J, Gregg PJ; National Joint Registry for England and Wales. The role of pain and function in determining patient satisfaction after total knee replacement. Data from the National Joint Registry for England and Wales. J Bone Joint Surg Br. 2007;89(7):893-900. doi:10.1302/0301-620X.89B7.19091
2. Noble PC, Conditt MA, Cook KF, Mathis KB. The John Insall Award: patient expectations affect satisfaction with total knee arthroplasty. Clin Orthop Relat Res. 2006;452:35-43. doi:10.1097/01.blo.0000238825.63648.1e
3. Matziolis G, Krocker D, Weiss U, Tohtz S, Perka C. A prospective, randomized study of computer-assisted and conventional total knee arthroplasty. Three-dimensional evaluation of implant alignment and rotation. J Bone Joint Surg Am. 2007;89(2):236-243. doi:10.2106/JBJS.F.00386
4. Stulberg SD, Yaffe MA, Koo SS. Computer-assisted surgery versus manual total knee arthroplasty: a case-controlled study. J Bone Joint Surg Am. 2006;88(suppl 4):47-54. doi:10.2106/JBJS.F.00698
5. Kalisvaart MM, Pagnano MW, Trousdale RT, Stuart MJ, Hanssen AD. Randomized clinical trial of rotating-platform and fixed-bearing total knee arthroplasty: no clinically detectable differences at five years. J Bone Joint Surg Am. 2012;94(6):481-489. doi:10.2106/JBJS.K.00315
6. Wülker N, Lambermont JP, Sacchetti L, Lazaró JG, Nardi J. A prospective randomized study of minimally invasive total knee arthroplasty compared with conventional surgery. J Bone Joint Surg Am. 2010;92(7):1584-1590. doi:10.2106/JBJS.H.01070
7. Thomsen MG, Husted H, Bencke J, Curtis D, Holm G, Troelsen A. Do we need a gender-specific total knee replacement? A randomised controlled trial comparing a high-flex and a gender-specific posterior design. J Bone Joint Surg Br. 2012;94(6):787-792. doi:10.1302/0301-620X.94B6.28781
8. Eckhoff D, Hogan C, DiMatteo L, Robinson M, Bach J. Difference between the epicondylar and cylindrical axis of the knee. Clin Orthop Relat Res. 2007;461:238-244. doi:10.1097/BLO.0b013e318112416b
9. Martin RK, Ley C, Pareek A, Groll A, Tischer T, Seil R. Artificial intelligence and machine learning: an introduction for orthopaedic surgeons [published online ahead of print, 2021 Sep 15]. Knee Surg Sports Traumatol Arthrosc. 2021;10.1007/s00167-021-06741-2. doi:10.1007/s00167-021-06741-2
10. Helm JM, Swiergosz AM, Haeberle HS, et al. Machine Learning and Artificial Intelligence: Definitions, Applications, and Future Directions. Curr Rev Musculoskelet Med. 2020;13(1):69-76. doi:10.1007/s12178-020-09600-8
11. Dossett HG, Estrada NA, Swartz GJ, LeFevre GW, Kwasman BG. A randomised controlled trial of kinematically and mechanically aligned total knee replacements: two-year clinical results. Bone Joint J. 2014;96-B(7):907-913. doi:10.1302/0301-620X.96B7.32812
12. Rivière C, Iranpour F, Auvinet E, et al. Alignment options for total knee arthroplasty: a systematic review. Orthop Traumatol Surg Res. 2017;103(7):1047-1056. doi:10.1016/j.otsr.2017.07.010
13. Dossett HG. High reliability in total knee replacement surgery: is it possible? Orthop Proc. 2018;95-B(suppl 34):292-293.
14. Schock J, Truhn D, Abrar DB, et al. Automated analysis of alignment in long-leg radiographs by using a fully automated support system based on artificial intelligence. Radiol: Artif Intell. Dec 23, 2020;3(2). doi:10.1148/ryai.2020200198
15. Cabitza F, Locoro A, Banfi G. Machine learning in orthopedics: a literature review. Front Bioeng Biotechnol. 2018;6:75. Published 2018 Jun 27. doi:10.3389/fbioe.2018.00075
16. von Schacky CE, Wilhelm NJ, Schäfer VS, et al. Multitask deep learning for segmentation and classification of primary bone tumors on radiographs. Radiology. 2021;301(2):398-406. doi:10.1148/radiol.2021204531
17. Myers TG, Ramkumar PN, Ricciardi BF, Urish KL, Kipper J, Ketonis C. Artificial intelligence and orthopaedics: an introduction for clinicians. J Bone Joint Surg Am. 2020;102(9):830-840. doi:10.2106/JBJS.19.01128
18. Kunze KN, Karhade AV, Sadauskas AJ, Schwab JH, Levine BR. Development of machine learning algorithms to predict clinically meaningful improvement for the patient-reported health state after total hip arthroplasty. J Arthroplasty. 2020;35(8):2119-2123. doi:10.1016/j.arth.2020.03.019
19. Harris AHS, Kuo AC, Bowe TR, Manfredi L, Lalani NF, Giori NJ. Can machine learning methods produce accurate and easy-to-use preoperative prediction models of one-year improvements in pain and functioning after knee arthroplasty? J Arthroplasty. 2021;36(1):112-117.e6. doi:10.1016/j.arth.2020.07.026
20. Rasouli JJ, Shao J, Neifert S, et al. Artificial intelligence and robotics in spine surgery. Global Spine J. 2021;11(4):556-564. doi:10.1177/2192568220915718
21. Joshi RS, Haddad AF, Lau D, Ames CP. Artificial intelligence for adult spinal deformity. Neurospine. 2019;16(4):686-694. doi:10.14245/ns.1938414.207
22. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
1. Baker PN, van der Meulen JH, Lewsey J, Gregg PJ; National Joint Registry for England and Wales. The role of pain and function in determining patient satisfaction after total knee replacement. Data from the National Joint Registry for England and Wales. J Bone Joint Surg Br. 2007;89(7):893-900. doi:10.1302/0301-620X.89B7.19091
2. Noble PC, Conditt MA, Cook KF, Mathis KB. The John Insall Award: patient expectations affect satisfaction with total knee arthroplasty. Clin Orthop Relat Res. 2006;452:35-43. doi:10.1097/01.blo.0000238825.63648.1e
3. Matziolis G, Krocker D, Weiss U, Tohtz S, Perka C. A prospective, randomized study of computer-assisted and conventional total knee arthroplasty. Three-dimensional evaluation of implant alignment and rotation. J Bone Joint Surg Am. 2007;89(2):236-243. doi:10.2106/JBJS.F.00386
4. Stulberg SD, Yaffe MA, Koo SS. Computer-assisted surgery versus manual total knee arthroplasty: a case-controlled study. J Bone Joint Surg Am. 2006;88(suppl 4):47-54. doi:10.2106/JBJS.F.00698
5. Kalisvaart MM, Pagnano MW, Trousdale RT, Stuart MJ, Hanssen AD. Randomized clinical trial of rotating-platform and fixed-bearing total knee arthroplasty: no clinically detectable differences at five years. J Bone Joint Surg Am. 2012;94(6):481-489. doi:10.2106/JBJS.K.00315
6. Wülker N, Lambermont JP, Sacchetti L, Lazaró JG, Nardi J. A prospective randomized study of minimally invasive total knee arthroplasty compared with conventional surgery. J Bone Joint Surg Am. 2010;92(7):1584-1590. doi:10.2106/JBJS.H.01070
7. Thomsen MG, Husted H, Bencke J, Curtis D, Holm G, Troelsen A. Do we need a gender-specific total knee replacement? A randomised controlled trial comparing a high-flex and a gender-specific posterior design. J Bone Joint Surg Br. 2012;94(6):787-792. doi:10.1302/0301-620X.94B6.28781
8. Eckhoff D, Hogan C, DiMatteo L, Robinson M, Bach J. Difference between the epicondylar and cylindrical axis of the knee. Clin Orthop Relat Res. 2007;461:238-244. doi:10.1097/BLO.0b013e318112416b
9. Martin RK, Ley C, Pareek A, Groll A, Tischer T, Seil R. Artificial intelligence and machine learning: an introduction for orthopaedic surgeons [published online ahead of print, 2021 Sep 15]. Knee Surg Sports Traumatol Arthrosc. 2021;10.1007/s00167-021-06741-2. doi:10.1007/s00167-021-06741-2
10. Helm JM, Swiergosz AM, Haeberle HS, et al. Machine Learning and Artificial Intelligence: Definitions, Applications, and Future Directions. Curr Rev Musculoskelet Med. 2020;13(1):69-76. doi:10.1007/s12178-020-09600-8
11. Dossett HG, Estrada NA, Swartz GJ, LeFevre GW, Kwasman BG. A randomised controlled trial of kinematically and mechanically aligned total knee replacements: two-year clinical results. Bone Joint J. 2014;96-B(7):907-913. doi:10.1302/0301-620X.96B7.32812
12. Rivière C, Iranpour F, Auvinet E, et al. Alignment options for total knee arthroplasty: a systematic review. Orthop Traumatol Surg Res. 2017;103(7):1047-1056. doi:10.1016/j.otsr.2017.07.010
13. Dossett HG. High reliability in total knee replacement surgery: is it possible? Orthop Proc. 2018;95-B(suppl 34):292-293.
14. Schock J, Truhn D, Abrar DB, et al. Automated analysis of alignment in long-leg radiographs by using a fully automated support system based on artificial intelligence. Radiol: Artif Intell. Dec 23, 2020;3(2). doi:10.1148/ryai.2020200198
15. Cabitza F, Locoro A, Banfi G. Machine learning in orthopedics: a literature review. Front Bioeng Biotechnol. 2018;6:75. Published 2018 Jun 27. doi:10.3389/fbioe.2018.00075
16. von Schacky CE, Wilhelm NJ, Schäfer VS, et al. Multitask deep learning for segmentation and classification of primary bone tumors on radiographs. Radiology. 2021;301(2):398-406. doi:10.1148/radiol.2021204531
17. Myers TG, Ramkumar PN, Ricciardi BF, Urish KL, Kipper J, Ketonis C. Artificial intelligence and orthopaedics: an introduction for clinicians. J Bone Joint Surg Am. 2020;102(9):830-840. doi:10.2106/JBJS.19.01128
18. Kunze KN, Karhade AV, Sadauskas AJ, Schwab JH, Levine BR. Development of machine learning algorithms to predict clinically meaningful improvement for the patient-reported health state after total hip arthroplasty. J Arthroplasty. 2020;35(8):2119-2123. doi:10.1016/j.arth.2020.03.019
19. Harris AHS, Kuo AC, Bowe TR, Manfredi L, Lalani NF, Giori NJ. Can machine learning methods produce accurate and easy-to-use preoperative prediction models of one-year improvements in pain and functioning after knee arthroplasty? J Arthroplasty. 2021;36(1):112-117.e6. doi:10.1016/j.arth.2020.07.026
20. Rasouli JJ, Shao J, Neifert S, et al. Artificial intelligence and robotics in spine surgery. Global Spine J. 2021;11(4):556-564. doi:10.1177/2192568220915718
21. Joshi RS, Haddad AF, Lau D, Ames CP. Artificial intelligence for adult spinal deformity. Neurospine. 2019;16(4):686-694. doi:10.14245/ns.1938414.207
22. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785. doi:10.2106/JBJS.F.00222
Announcement: Thank You to Our Cutis Reviewers
Cutis Reviewers, JANUARY TO DECEMBER 2021
Jennifer L. Adams, MD
Brandon Adler, MD
Iris Ahronowitz, MD
Abdullah Aleisa, MD
Justin Bandino, MD
Robert Baran, MD
Naiara S. Barbosa, MD
Kristina R. Burke, MD
Craig Burkhart, MD
Jeffrey P. Callen, MD
Charles Camisa, MD
Ashley B. Crew, MD
Zoe Diana Draelos, MD
Joseph S. Eastern, MD
Nada Elbuluk, MD, MSc
Joseph C. English III, MD
Tammie C. Ferringer, MD
Bahar F. Firoz, MD, MPH
John R. Griffin, MD
Kristi Hawley, DO
Thomas N. Helm, MD
Stephen Ellsworth Helms, MD
Brian P. Hibler, MD
Ranella J. Hirsch, MD
Jenny Hu, MD, MPH
Erick Jacobson-Dunlop, MD
William D. James, MD
Camilla K. Janniger, MD
Ronald Belle Johnston Jr, MD
Theodora Karagounis, MD
Michael Kasperkiewicz, MD
Brett H. Keeling, MD
Chesahna Kindred, MD, MBA
Christina Nicole Kraus, MD
Eun Ji Kwon, MD
Eden Lake, MD
Nicholas Logemann, DO
Michele S. Maroon, MD
Cathy Massoud, MD
Elizabeth I. McBurney, MD
Lynn J. McKinley-Grant, MD
Amy J. McMichael, MD
Darius R. Mehregan, MD
Jon Hamilton Meyerle, MD
Robert Micheletti, MD
Binh Ngo, MD
Anh Nguyen, MD
Josephine Nguyen, MD, MHCDS
Joe Niamtu III, DMD
Rajiv I. Nijhawan, MD
Michael A. Nowak, MD
Maria T. Ochoa, MD
Chika Ohata, MD
Lawrence C. Parish, MD
Marion Apter Quinn, MD
Phoebe Rich, MD
Maureen Riegert, MD
Bethany Rohr, MD
Lorraine L. Rosamilia, MD
Ted Rosen, MD
Robert I. Rudolph, MD
Elizabeth K. Satter, MD, MPH
Robert A. Schwartz, MD, MPH
Michael J. Scott III, DO, MD, MPH
Jane Scribner, MD
Bridget E. Shields, MD
Daniel Mark Siegel, MD, MS
Nanette B. Silverberg, MD
Steven Brett Sloan, MD
Leonard Sperling, MD
Stephen P. Stone, MD
Brian L. Swick, MD
Susan C. Taylor, MD
Manuel Valdebran, MD
Richard F. Wagner Jr, MD
Emily Wong, MD
Julie Woodward, MD
Scott Worswick, MD
Julia Wu, MD
If you are interested in serving as a reviewer, please email the Editorial Office at [email protected].
Cutis Reviewers, JANUARY TO DECEMBER 2021
Jennifer L. Adams, MD
Brandon Adler, MD
Iris Ahronowitz, MD
Abdullah Aleisa, MD
Justin Bandino, MD
Robert Baran, MD
Naiara S. Barbosa, MD
Kristina R. Burke, MD
Craig Burkhart, MD
Jeffrey P. Callen, MD
Charles Camisa, MD
Ashley B. Crew, MD
Zoe Diana Draelos, MD
Joseph S. Eastern, MD
Nada Elbuluk, MD, MSc
Joseph C. English III, MD
Tammie C. Ferringer, MD
Bahar F. Firoz, MD, MPH
John R. Griffin, MD
Kristi Hawley, DO
Thomas N. Helm, MD
Stephen Ellsworth Helms, MD
Brian P. Hibler, MD
Ranella J. Hirsch, MD
Jenny Hu, MD, MPH
Erick Jacobson-Dunlop, MD
William D. James, MD
Camilla K. Janniger, MD
Ronald Belle Johnston Jr, MD
Theodora Karagounis, MD
Michael Kasperkiewicz, MD
Brett H. Keeling, MD
Chesahna Kindred, MD, MBA
Christina Nicole Kraus, MD
Eun Ji Kwon, MD
Eden Lake, MD
Nicholas Logemann, DO
Michele S. Maroon, MD
Cathy Massoud, MD
Elizabeth I. McBurney, MD
Lynn J. McKinley-Grant, MD
Amy J. McMichael, MD
Darius R. Mehregan, MD
Jon Hamilton Meyerle, MD
Robert Micheletti, MD
Binh Ngo, MD
Anh Nguyen, MD
Josephine Nguyen, MD, MHCDS
Joe Niamtu III, DMD
Rajiv I. Nijhawan, MD
Michael A. Nowak, MD
Maria T. Ochoa, MD
Chika Ohata, MD
Lawrence C. Parish, MD
Marion Apter Quinn, MD
Phoebe Rich, MD
Maureen Riegert, MD
Bethany Rohr, MD
Lorraine L. Rosamilia, MD
Ted Rosen, MD
Robert I. Rudolph, MD
Elizabeth K. Satter, MD, MPH
Robert A. Schwartz, MD, MPH
Michael J. Scott III, DO, MD, MPH
Jane Scribner, MD
Bridget E. Shields, MD
Daniel Mark Siegel, MD, MS
Nanette B. Silverberg, MD
Steven Brett Sloan, MD
Leonard Sperling, MD
Stephen P. Stone, MD
Brian L. Swick, MD
Susan C. Taylor, MD
Manuel Valdebran, MD
Richard F. Wagner Jr, MD
Emily Wong, MD
Julie Woodward, MD
Scott Worswick, MD
Julia Wu, MD
If you are interested in serving as a reviewer, please email the Editorial Office at [email protected].
Cutis Reviewers, JANUARY TO DECEMBER 2021
Jennifer L. Adams, MD
Brandon Adler, MD
Iris Ahronowitz, MD
Abdullah Aleisa, MD
Justin Bandino, MD
Robert Baran, MD
Naiara S. Barbosa, MD
Kristina R. Burke, MD
Craig Burkhart, MD
Jeffrey P. Callen, MD
Charles Camisa, MD
Ashley B. Crew, MD
Zoe Diana Draelos, MD
Joseph S. Eastern, MD
Nada Elbuluk, MD, MSc
Joseph C. English III, MD
Tammie C. Ferringer, MD
Bahar F. Firoz, MD, MPH
John R. Griffin, MD
Kristi Hawley, DO
Thomas N. Helm, MD
Stephen Ellsworth Helms, MD
Brian P. Hibler, MD
Ranella J. Hirsch, MD
Jenny Hu, MD, MPH
Erick Jacobson-Dunlop, MD
William D. James, MD
Camilla K. Janniger, MD
Ronald Belle Johnston Jr, MD
Theodora Karagounis, MD
Michael Kasperkiewicz, MD
Brett H. Keeling, MD
Chesahna Kindred, MD, MBA
Christina Nicole Kraus, MD
Eun Ji Kwon, MD
Eden Lake, MD
Nicholas Logemann, DO
Michele S. Maroon, MD
Cathy Massoud, MD
Elizabeth I. McBurney, MD
Lynn J. McKinley-Grant, MD
Amy J. McMichael, MD
Darius R. Mehregan, MD
Jon Hamilton Meyerle, MD
Robert Micheletti, MD
Binh Ngo, MD
Anh Nguyen, MD
Josephine Nguyen, MD, MHCDS
Joe Niamtu III, DMD
Rajiv I. Nijhawan, MD
Michael A. Nowak, MD
Maria T. Ochoa, MD
Chika Ohata, MD
Lawrence C. Parish, MD
Marion Apter Quinn, MD
Phoebe Rich, MD
Maureen Riegert, MD
Bethany Rohr, MD
Lorraine L. Rosamilia, MD
Ted Rosen, MD
Robert I. Rudolph, MD
Elizabeth K. Satter, MD, MPH
Robert A. Schwartz, MD, MPH
Michael J. Scott III, DO, MD, MPH
Jane Scribner, MD
Bridget E. Shields, MD
Daniel Mark Siegel, MD, MS
Nanette B. Silverberg, MD
Steven Brett Sloan, MD
Leonard Sperling, MD
Stephen P. Stone, MD
Brian L. Swick, MD
Susan C. Taylor, MD
Manuel Valdebran, MD
Richard F. Wagner Jr, MD
Emily Wong, MD
Julie Woodward, MD
Scott Worswick, MD
Julia Wu, MD
If you are interested in serving as a reviewer, please email the Editorial Office at [email protected].






