User login
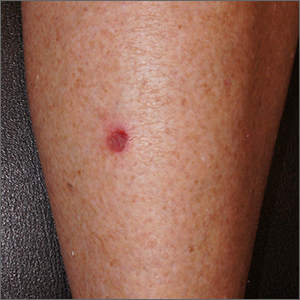
Small persistent leg wound

A leg ulcer may have many causes, including venous stasis, trauma, vasculitis, infection, or (as in this case) squamous cell carcinoma in situ (SCCis), aka Bowen’s Disease.
SCC and SCCis are common skin cancers that occur less frequently than basal cell carcinomas (BCCs).1 SCCis is normally scaly and hyperkeratotic, but it can manifest in rare cases as a chronic ulcer. Fair skin, long history of sun damage, and immunosuppression are significant risk factors for both SCCis and SCC.
While history and other clinical features may help narrow the diagnosis, a wound that does not heal despite treatments should be biopsied. Shave and punch biopsies are both excellent ways to diagnose an SCCis that has a classic appearance. However, ulcers and blisters can be caused by inflammatory processes (as in pyoderma gangrenosum or a fixed drug eruption) with characteristic findings deeper in the dermis; these lesions are better assessed with a punch biopsy.
In this case, a 4-mm punch biopsy was performed at the tissue edge and showed atypical keratinocytes limited to the epidermis. These atypical keratinocytes are associated with vesicle formation and ulcer, consistent with SCCis.
SCCis transforms into invasive disease in 3% to 5% of cases.2 Surgical treatment includes fusiform excision and electrodessication and curettage, both with cure rates that often exceed 90%.2,3 Nonsurgical options include topical 5-fluorouracil (67%-92% effective), topical imiquimod (75%-93%), and photodynamic therapy (52%-98%).4
Treatment choices depend on patient preference and provider capabilities. With surgical options there is the risk of bleeding and the need to care for a healing wound. Nonsurgical treatments can last longer and require topical treatment regimens and medications.
This patient opted for a fusiform excision and linear closure. She will continue to undergo serial skin evaluations twice a year for at least 2 years.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.
1. Lukowiak TM, Aizman L, Perz A, et al. Association of age, sex, race, and geographic region with variation of the ratio of basal cell to cutaneous squamous cell carcinomas in the United States. JAMA Dermatol. 2020;156:1192-1198. doi:10.1001/jamadermatol.2020.2571
2. Morton CA, Birnie AJ, Eedy DJ. British Association of Dermatologists’ guidelines for the management of squamous cell carcinoma in situ (Bowen's disease). Br J Dermatol. 2014;170:245-246. doi: 10.1111/bjd.12766
3. Veverka KK, Stratman EJ. Electrodesiccation and curettage for squamous cell carcinoma in situ: the effect of anatomic location on local recurrence. Dermatol Surg. 2023;49:821-824. doi: 10.1097/DSS.0000000000003855
4. Algarin, YA, Jambusaria-Pahlajani A. Ruiz E, et al. Advances in topical treatments of cutaneous malignancies. Am J Clin Dermatol. 2023;24:69-80. doi: 10.1007/s40257-022-00731-x

A leg ulcer may have many causes, including venous stasis, trauma, vasculitis, infection, or (as in this case) squamous cell carcinoma in situ (SCCis), aka Bowen’s Disease.
SCC and SCCis are common skin cancers that occur less frequently than basal cell carcinomas (BCCs).1 SCCis is normally scaly and hyperkeratotic, but it can manifest in rare cases as a chronic ulcer. Fair skin, long history of sun damage, and immunosuppression are significant risk factors for both SCCis and SCC.
While history and other clinical features may help narrow the diagnosis, a wound that does not heal despite treatments should be biopsied. Shave and punch biopsies are both excellent ways to diagnose an SCCis that has a classic appearance. However, ulcers and blisters can be caused by inflammatory processes (as in pyoderma gangrenosum or a fixed drug eruption) with characteristic findings deeper in the dermis; these lesions are better assessed with a punch biopsy.
In this case, a 4-mm punch biopsy was performed at the tissue edge and showed atypical keratinocytes limited to the epidermis. These atypical keratinocytes are associated with vesicle formation and ulcer, consistent with SCCis.
SCCis transforms into invasive disease in 3% to 5% of cases.2 Surgical treatment includes fusiform excision and electrodessication and curettage, both with cure rates that often exceed 90%.2,3 Nonsurgical options include topical 5-fluorouracil (67%-92% effective), topical imiquimod (75%-93%), and photodynamic therapy (52%-98%).4
Treatment choices depend on patient preference and provider capabilities. With surgical options there is the risk of bleeding and the need to care for a healing wound. Nonsurgical treatments can last longer and require topical treatment regimens and medications.
This patient opted for a fusiform excision and linear closure. She will continue to undergo serial skin evaluations twice a year for at least 2 years.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.

A leg ulcer may have many causes, including venous stasis, trauma, vasculitis, infection, or (as in this case) squamous cell carcinoma in situ (SCCis), aka Bowen’s Disease.
SCC and SCCis are common skin cancers that occur less frequently than basal cell carcinomas (BCCs).1 SCCis is normally scaly and hyperkeratotic, but it can manifest in rare cases as a chronic ulcer. Fair skin, long history of sun damage, and immunosuppression are significant risk factors for both SCCis and SCC.
While history and other clinical features may help narrow the diagnosis, a wound that does not heal despite treatments should be biopsied. Shave and punch biopsies are both excellent ways to diagnose an SCCis that has a classic appearance. However, ulcers and blisters can be caused by inflammatory processes (as in pyoderma gangrenosum or a fixed drug eruption) with characteristic findings deeper in the dermis; these lesions are better assessed with a punch biopsy.
In this case, a 4-mm punch biopsy was performed at the tissue edge and showed atypical keratinocytes limited to the epidermis. These atypical keratinocytes are associated with vesicle formation and ulcer, consistent with SCCis.
SCCis transforms into invasive disease in 3% to 5% of cases.2 Surgical treatment includes fusiform excision and electrodessication and curettage, both with cure rates that often exceed 90%.2,3 Nonsurgical options include topical 5-fluorouracil (67%-92% effective), topical imiquimod (75%-93%), and photodynamic therapy (52%-98%).4
Treatment choices depend on patient preference and provider capabilities. With surgical options there is the risk of bleeding and the need to care for a healing wound. Nonsurgical treatments can last longer and require topical treatment regimens and medications.
This patient opted for a fusiform excision and linear closure. She will continue to undergo serial skin evaluations twice a year for at least 2 years.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.
1. Lukowiak TM, Aizman L, Perz A, et al. Association of age, sex, race, and geographic region with variation of the ratio of basal cell to cutaneous squamous cell carcinomas in the United States. JAMA Dermatol. 2020;156:1192-1198. doi:10.1001/jamadermatol.2020.2571
2. Morton CA, Birnie AJ, Eedy DJ. British Association of Dermatologists’ guidelines for the management of squamous cell carcinoma in situ (Bowen's disease). Br J Dermatol. 2014;170:245-246. doi: 10.1111/bjd.12766
3. Veverka KK, Stratman EJ. Electrodesiccation and curettage for squamous cell carcinoma in situ: the effect of anatomic location on local recurrence. Dermatol Surg. 2023;49:821-824. doi: 10.1097/DSS.0000000000003855
4. Algarin, YA, Jambusaria-Pahlajani A. Ruiz E, et al. Advances in topical treatments of cutaneous malignancies. Am J Clin Dermatol. 2023;24:69-80. doi: 10.1007/s40257-022-00731-x
1. Lukowiak TM, Aizman L, Perz A, et al. Association of age, sex, race, and geographic region with variation of the ratio of basal cell to cutaneous squamous cell carcinomas in the United States. JAMA Dermatol. 2020;156:1192-1198. doi:10.1001/jamadermatol.2020.2571
2. Morton CA, Birnie AJ, Eedy DJ. British Association of Dermatologists’ guidelines for the management of squamous cell carcinoma in situ (Bowen's disease). Br J Dermatol. 2014;170:245-246. doi: 10.1111/bjd.12766
3. Veverka KK, Stratman EJ. Electrodesiccation and curettage for squamous cell carcinoma in situ: the effect of anatomic location on local recurrence. Dermatol Surg. 2023;49:821-824. doi: 10.1097/DSS.0000000000003855
4. Algarin, YA, Jambusaria-Pahlajani A. Ruiz E, et al. Advances in topical treatments of cutaneous malignancies. Am J Clin Dermatol. 2023;24:69-80. doi: 10.1007/s40257-022-00731-x
Disseminated Papules and Nodules on the Skin and Oral Mucosa in an Infant
The Diagnosis: Congenital Cutaneous Langerhans Cell Histiocytosis
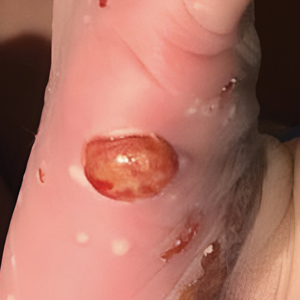
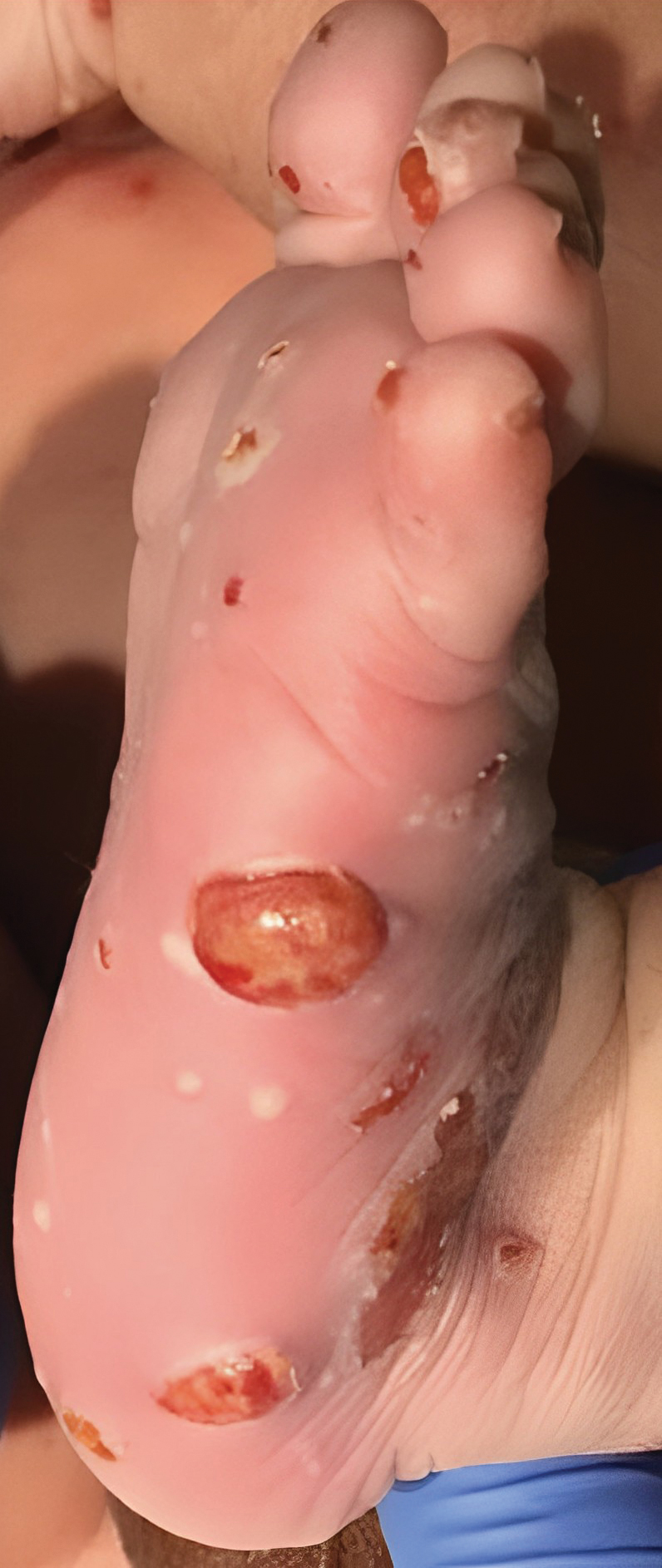
Although the infectious workup was positive for herpes simplex virus type 1 and cytomegalovirus antibodies, serologies for the rest of the TORCH (toxoplasmosis, other agents [syphilis, hepatitis B virus], rubella, cytomegalovirus) group of infections, as well as other bacterial, fungal, and viral infections, were negative. A skin biopsy from the right fifth toe showed a dense infiltrate of CD1a+ histiocytic cells with folded or kidney-shaped nuclei mixed with eosinophils, which was consistent with Langerhans cell histiocytosis (LCH) (Figure 1). Skin lesions were treated with hydrocortisone cream 2.5% and progressively faded over a few weeks.

Langerhans cell histiocytosis is a rare disorder with a variable clinical presentation depending on the sites affected and the extent of involvement. It can involve multiple organ systems, most commonly the skeletal system and the skin. Organ involvement is characterized by histiocyte infiltration. Acute disseminated multisystem disease most commonly is seen in children younger than 3 years.1
Congenital cutaneous LCH presents with variable skin lesions ranging from papules to vesicles, pustules, and ulcers, with onset at birth or in the neonatal period. Various morphologic traits of skin lesions have been described; the most common presentation is multiple red to yellow-brown, crusted papules with accompanying hemorrhage or erosion.1 Other cases have described an eczematous, seborrheic, diffuse eruption or erosive intertrigo. One case of a child with a solitary necrotic nodule on the scalp has been reported.2
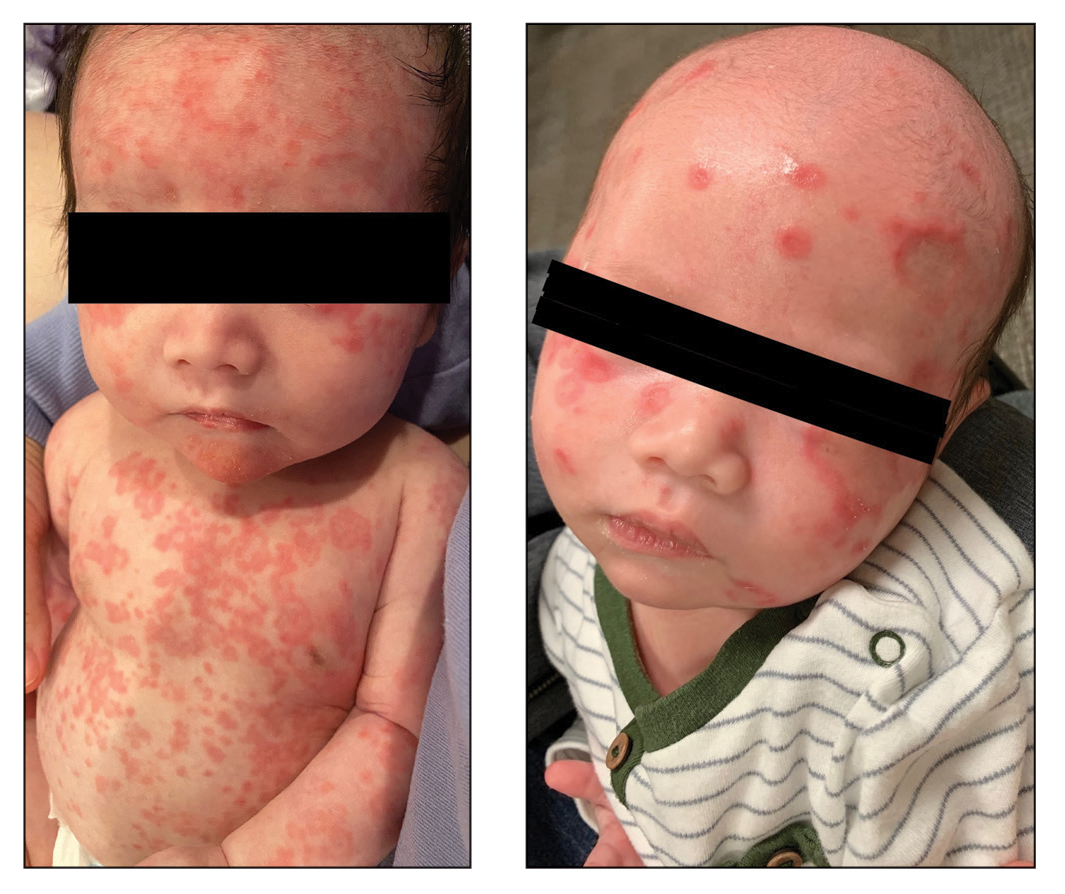
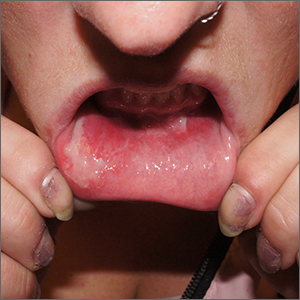
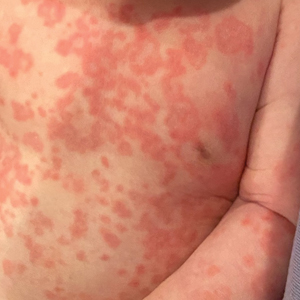
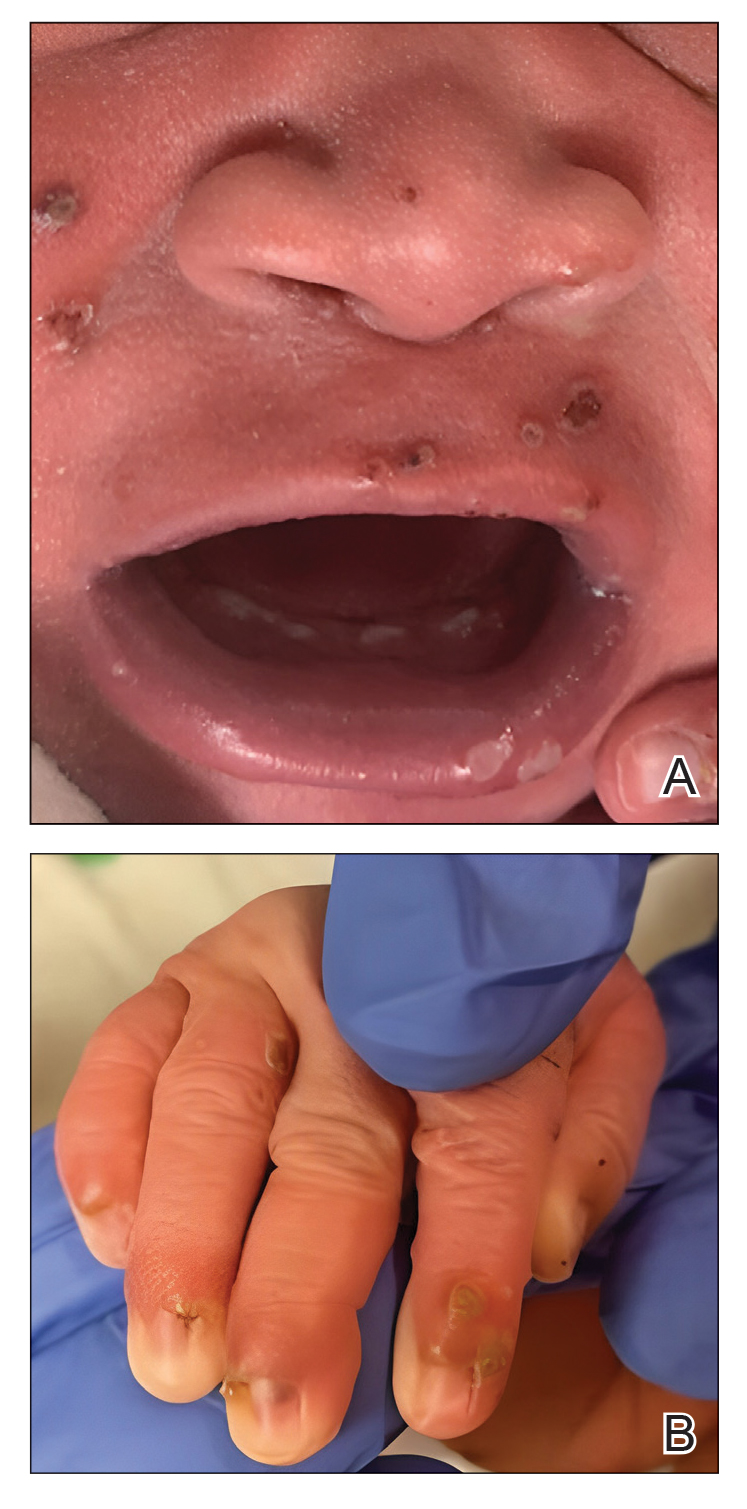
Our patient presented with disseminated, nonblanching, purple to dark red papules and nodules of the skin and oral mucosa, as well as nail dystrophy (Figure 2). However, LCH in a neonate can mimic other causes of congenital papulonodular eruptions. Red-brown papules and nodules with or without crusting in a newborn can be mistaken for erythema toxicum neonatorum, transient neonatal pustular melanosis, congenital leukemia cutis, neonatal erythropoiesis, disseminated neonatal hemangiomatosis, infantile acropustulosis, or congenital TORCH infections such as rubella or syphilis. When LCH presents as vesicles or eroded papules or nodules in a newborn, the differential diagnosis includes incontinentia pigmenti and hereditary epidermolysis bullosa.
Langerhans cell histiocytosis may even present with a classic blueberry muffin rash that can lead clinicians to consider cutaneous metastasis from various hematologic malignancies or the more common TORCH infections. Several diagnostic tests can be performed to clarify the diagnosis, including bacterial and viral cultures and stains, serology, immunohistochemistry, flow cytometry, bone marrow aspiration, or skin biopsy.3 Langerhans cell histiocytosis is diagnosed with a combination of histology, immunohistochemistry, and clinical presentation; however, a skin biopsy is crucial. Tissue should be taken from the most easily accessible yet representative lesion. The characteristic appearance of LCH lesions is described as a dense infiltrate of histiocytic cells mixed with numerous eosinophils in the dermis.1 Histiocytes usually have folded nuclei and eosinophilic cytoplasm or kidney-shaped nuclei with prominent nucleoli. Positive CD1a and/or CD207 (Langerin) staining of the cells is required for definitive diagnosis.4 After diagnosis, it is important to obtain baseline laboratory and radiographic studies to determine the extent of systemic involvement.
Treatment of congenital LCH is tailored to the extent of organ involvement. The dermatologic manifestations resolve without medications in many cases. However, true self-resolving LCH can only be diagnosed retrospectively after a full evaluation for other sites of disease. Disseminated disease can be life-threatening and requires more active management. In cases of skin-limited disease, therapies include topical steroids, nitrogen mustard, or imiquimod; surgical resection of isolated lesions; phototherapy; or systemic therapies such as methotrexate, 6-mercaptopurine, vinblastine/vincristine, cladribine, and/or cytarabine. Symptomatic patients initially are treated with methotrexate and 6-mercaptopurine.5 Asymptomatic infants with skin-limited involvement can be managed with topical treatments.
Our patient had skin-limited disease. Abdominal ultrasonography, skeletal survey, and magnetic resonance imaging of the brain revealed no abnormalities. The patient’s family was advised to monitor him for reoccurrence of the skin lesions and to continue close follow-up with hematology and dermatology. Although congenital LCH often is self-resolving, extensive skin involvement increases the risk for internal organ involvement for several years.6 These patients require long-term follow-up for potential musculoskeletal, ophthalmologic, endocrine, hepatic, and/or pulmonary disease.
- Pan Y, Zeng X, Ge J, et al. Congenital self-healing Langerhans cell histiocytosis: clinical and pathological characteristics. Int J Clin Exp Pathol. 2019;12:2275-2278.
- Morren MA, Vanden Broecke K, Vangeebergen L, et al. Diverse cutaneous presentations of Langerhans cell histiocytosis in children: a retrospective cohort study. Pediatr Blood Cancer. 2016;63:486-492. doi:10.1002/pbc.25834
- Krooks J, Minkov M, Weatherall AG. Langerhans cell histiocytosis in children: diagnosis, differential diagnosis, treatment, sequelae, and standardized follow-up. J Am Acad Dermatol. 2018;78:1047-1056. doi:10.1016/j.jaad.2017.05.060
- Haupt R, Minkov M, Astigarraga I, et al. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184. doi:10.1002/pbc.24367
- Allen CE, Ladisch S, McClain KL. How I treat Langerhans cell histiocytosis. Blood. 2015;126:26-35. doi:10.1182/blood-2014-12-569301
- Jezierska M, Stefanowicz J, Romanowicz G, et al. Langerhans cell histiocytosis in children—a disease with many faces. recent advances in pathogenesis, diagnostic examinations and treatment. Postepy Dermatol Alergol. 2018;35:6-17. doi:10.5114/pdia.2017.67095
The Diagnosis: Congenital Cutaneous Langerhans Cell Histiocytosis
Although the infectious workup was positive for herpes simplex virus type 1 and cytomegalovirus antibodies, serologies for the rest of the TORCH (toxoplasmosis, other agents [syphilis, hepatitis B virus], rubella, cytomegalovirus) group of infections, as well as other bacterial, fungal, and viral infections, were negative. A skin biopsy from the right fifth toe showed a dense infiltrate of CD1a+ histiocytic cells with folded or kidney-shaped nuclei mixed with eosinophils, which was consistent with Langerhans cell histiocytosis (LCH) (Figure 1). Skin lesions were treated with hydrocortisone cream 2.5% and progressively faded over a few weeks.

Langerhans cell histiocytosis is a rare disorder with a variable clinical presentation depending on the sites affected and the extent of involvement. It can involve multiple organ systems, most commonly the skeletal system and the skin. Organ involvement is characterized by histiocyte infiltration. Acute disseminated multisystem disease most commonly is seen in children younger than 3 years.1
Congenital cutaneous LCH presents with variable skin lesions ranging from papules to vesicles, pustules, and ulcers, with onset at birth or in the neonatal period. Various morphologic traits of skin lesions have been described; the most common presentation is multiple red to yellow-brown, crusted papules with accompanying hemorrhage or erosion.1 Other cases have described an eczematous, seborrheic, diffuse eruption or erosive intertrigo. One case of a child with a solitary necrotic nodule on the scalp has been reported.2
Our patient presented with disseminated, nonblanching, purple to dark red papules and nodules of the skin and oral mucosa, as well as nail dystrophy (Figure 2). However, LCH in a neonate can mimic other causes of congenital papulonodular eruptions. Red-brown papules and nodules with or without crusting in a newborn can be mistaken for erythema toxicum neonatorum, transient neonatal pustular melanosis, congenital leukemia cutis, neonatal erythropoiesis, disseminated neonatal hemangiomatosis, infantile acropustulosis, or congenital TORCH infections such as rubella or syphilis. When LCH presents as vesicles or eroded papules or nodules in a newborn, the differential diagnosis includes incontinentia pigmenti and hereditary epidermolysis bullosa.

Langerhans cell histiocytosis may even present with a classic blueberry muffin rash that can lead clinicians to consider cutaneous metastasis from various hematologic malignancies or the more common TORCH infections. Several diagnostic tests can be performed to clarify the diagnosis, including bacterial and viral cultures and stains, serology, immunohistochemistry, flow cytometry, bone marrow aspiration, or skin biopsy.3 Langerhans cell histiocytosis is diagnosed with a combination of histology, immunohistochemistry, and clinical presentation; however, a skin biopsy is crucial. Tissue should be taken from the most easily accessible yet representative lesion. The characteristic appearance of LCH lesions is described as a dense infiltrate of histiocytic cells mixed with numerous eosinophils in the dermis.1 Histiocytes usually have folded nuclei and eosinophilic cytoplasm or kidney-shaped nuclei with prominent nucleoli. Positive CD1a and/or CD207 (Langerin) staining of the cells is required for definitive diagnosis.4 After diagnosis, it is important to obtain baseline laboratory and radiographic studies to determine the extent of systemic involvement.
Treatment of congenital LCH is tailored to the extent of organ involvement. The dermatologic manifestations resolve without medications in many cases. However, true self-resolving LCH can only be diagnosed retrospectively after a full evaluation for other sites of disease. Disseminated disease can be life-threatening and requires more active management. In cases of skin-limited disease, therapies include topical steroids, nitrogen mustard, or imiquimod; surgical resection of isolated lesions; phototherapy; or systemic therapies such as methotrexate, 6-mercaptopurine, vinblastine/vincristine, cladribine, and/or cytarabine. Symptomatic patients initially are treated with methotrexate and 6-mercaptopurine.5 Asymptomatic infants with skin-limited involvement can be managed with topical treatments.
Our patient had skin-limited disease. Abdominal ultrasonography, skeletal survey, and magnetic resonance imaging of the brain revealed no abnormalities. The patient’s family was advised to monitor him for reoccurrence of the skin lesions and to continue close follow-up with hematology and dermatology. Although congenital LCH often is self-resolving, extensive skin involvement increases the risk for internal organ involvement for several years.6 These patients require long-term follow-up for potential musculoskeletal, ophthalmologic, endocrine, hepatic, and/or pulmonary disease.
The Diagnosis: Congenital Cutaneous Langerhans Cell Histiocytosis
Although the infectious workup was positive for herpes simplex virus type 1 and cytomegalovirus antibodies, serologies for the rest of the TORCH (toxoplasmosis, other agents [syphilis, hepatitis B virus], rubella, cytomegalovirus) group of infections, as well as other bacterial, fungal, and viral infections, were negative. A skin biopsy from the right fifth toe showed a dense infiltrate of CD1a+ histiocytic cells with folded or kidney-shaped nuclei mixed with eosinophils, which was consistent with Langerhans cell histiocytosis (LCH) (Figure 1). Skin lesions were treated with hydrocortisone cream 2.5% and progressively faded over a few weeks.

Langerhans cell histiocytosis is a rare disorder with a variable clinical presentation depending on the sites affected and the extent of involvement. It can involve multiple organ systems, most commonly the skeletal system and the skin. Organ involvement is characterized by histiocyte infiltration. Acute disseminated multisystem disease most commonly is seen in children younger than 3 years.1
Congenital cutaneous LCH presents with variable skin lesions ranging from papules to vesicles, pustules, and ulcers, with onset at birth or in the neonatal period. Various morphologic traits of skin lesions have been described; the most common presentation is multiple red to yellow-brown, crusted papules with accompanying hemorrhage or erosion.1 Other cases have described an eczematous, seborrheic, diffuse eruption or erosive intertrigo. One case of a child with a solitary necrotic nodule on the scalp has been reported.2
Our patient presented with disseminated, nonblanching, purple to dark red papules and nodules of the skin and oral mucosa, as well as nail dystrophy (Figure 2). However, LCH in a neonate can mimic other causes of congenital papulonodular eruptions. Red-brown papules and nodules with or without crusting in a newborn can be mistaken for erythema toxicum neonatorum, transient neonatal pustular melanosis, congenital leukemia cutis, neonatal erythropoiesis, disseminated neonatal hemangiomatosis, infantile acropustulosis, or congenital TORCH infections such as rubella or syphilis. When LCH presents as vesicles or eroded papules or nodules in a newborn, the differential diagnosis includes incontinentia pigmenti and hereditary epidermolysis bullosa.

Langerhans cell histiocytosis may even present with a classic blueberry muffin rash that can lead clinicians to consider cutaneous metastasis from various hematologic malignancies or the more common TORCH infections. Several diagnostic tests can be performed to clarify the diagnosis, including bacterial and viral cultures and stains, serology, immunohistochemistry, flow cytometry, bone marrow aspiration, or skin biopsy.3 Langerhans cell histiocytosis is diagnosed with a combination of histology, immunohistochemistry, and clinical presentation; however, a skin biopsy is crucial. Tissue should be taken from the most easily accessible yet representative lesion. The characteristic appearance of LCH lesions is described as a dense infiltrate of histiocytic cells mixed with numerous eosinophils in the dermis.1 Histiocytes usually have folded nuclei and eosinophilic cytoplasm or kidney-shaped nuclei with prominent nucleoli. Positive CD1a and/or CD207 (Langerin) staining of the cells is required for definitive diagnosis.4 After diagnosis, it is important to obtain baseline laboratory and radiographic studies to determine the extent of systemic involvement.
Treatment of congenital LCH is tailored to the extent of organ involvement. The dermatologic manifestations resolve without medications in many cases. However, true self-resolving LCH can only be diagnosed retrospectively after a full evaluation for other sites of disease. Disseminated disease can be life-threatening and requires more active management. In cases of skin-limited disease, therapies include topical steroids, nitrogen mustard, or imiquimod; surgical resection of isolated lesions; phototherapy; or systemic therapies such as methotrexate, 6-mercaptopurine, vinblastine/vincristine, cladribine, and/or cytarabine. Symptomatic patients initially are treated with methotrexate and 6-mercaptopurine.5 Asymptomatic infants with skin-limited involvement can be managed with topical treatments.
Our patient had skin-limited disease. Abdominal ultrasonography, skeletal survey, and magnetic resonance imaging of the brain revealed no abnormalities. The patient’s family was advised to monitor him for reoccurrence of the skin lesions and to continue close follow-up with hematology and dermatology. Although congenital LCH often is self-resolving, extensive skin involvement increases the risk for internal organ involvement for several years.6 These patients require long-term follow-up for potential musculoskeletal, ophthalmologic, endocrine, hepatic, and/or pulmonary disease.
- Pan Y, Zeng X, Ge J, et al. Congenital self-healing Langerhans cell histiocytosis: clinical and pathological characteristics. Int J Clin Exp Pathol. 2019;12:2275-2278.
- Morren MA, Vanden Broecke K, Vangeebergen L, et al. Diverse cutaneous presentations of Langerhans cell histiocytosis in children: a retrospective cohort study. Pediatr Blood Cancer. 2016;63:486-492. doi:10.1002/pbc.25834
- Krooks J, Minkov M, Weatherall AG. Langerhans cell histiocytosis in children: diagnosis, differential diagnosis, treatment, sequelae, and standardized follow-up. J Am Acad Dermatol. 2018;78:1047-1056. doi:10.1016/j.jaad.2017.05.060
- Haupt R, Minkov M, Astigarraga I, et al. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184. doi:10.1002/pbc.24367
- Allen CE, Ladisch S, McClain KL. How I treat Langerhans cell histiocytosis. Blood. 2015;126:26-35. doi:10.1182/blood-2014-12-569301
- Jezierska M, Stefanowicz J, Romanowicz G, et al. Langerhans cell histiocytosis in children—a disease with many faces. recent advances in pathogenesis, diagnostic examinations and treatment. Postepy Dermatol Alergol. 2018;35:6-17. doi:10.5114/pdia.2017.67095
- Pan Y, Zeng X, Ge J, et al. Congenital self-healing Langerhans cell histiocytosis: clinical and pathological characteristics. Int J Clin Exp Pathol. 2019;12:2275-2278.
- Morren MA, Vanden Broecke K, Vangeebergen L, et al. Diverse cutaneous presentations of Langerhans cell histiocytosis in children: a retrospective cohort study. Pediatr Blood Cancer. 2016;63:486-492. doi:10.1002/pbc.25834
- Krooks J, Minkov M, Weatherall AG. Langerhans cell histiocytosis in children: diagnosis, differential diagnosis, treatment, sequelae, and standardized follow-up. J Am Acad Dermatol. 2018;78:1047-1056. doi:10.1016/j.jaad.2017.05.060
- Haupt R, Minkov M, Astigarraga I, et al. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184. doi:10.1002/pbc.24367
- Allen CE, Ladisch S, McClain KL. How I treat Langerhans cell histiocytosis. Blood. 2015;126:26-35. doi:10.1182/blood-2014-12-569301
- Jezierska M, Stefanowicz J, Romanowicz G, et al. Langerhans cell histiocytosis in children—a disease with many faces. recent advances in pathogenesis, diagnostic examinations and treatment. Postepy Dermatol Alergol. 2018;35:6-17. doi:10.5114/pdia.2017.67095
A 38-week-old infant boy presented at birth with disseminated, nonblanching, purple to dark red papules and nodules on the skin and oral mucosa. He was born spontaneously after an uncomplicated pregnancy. The mother experienced an episode of oral herpes simplex virus during pregnancy. The infant was otherwise healthy. Laboratory tests including a complete blood cell count and routine serum biochemical analyses were within reference range; however, an infectious workup was positive for herpes simplex virus type 1 and cytomegalovirus antibodies. Ophthalmologic and auditory screenings were normal.
Ruxolitinib for vitiligo: Experts share experiences from first year
.
The Food and Drug Administration approved the cream formulation of ruxolitinib (Opzelura), a JAK inhibitor, for repigmentation of nonsegmental vitiligo in July 2022 for people aged 12 years and older.
Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, said that he likes to use ruxolitinib cream in combination with other treatments.
“In the real world with vitiligo patients, we’re oftentimes doing combinatorial therapy anyway. So phototherapy, specifically, narrow-band UVB, is something that we have a lot of clinical evidence for over the years, and it’s a modality that can combine with topical steroids and topical calcineurin inhibitors.”
He said trials to study combinations will yield better guidance on optimal use of ruxolitinib cream. “In general, vitiligo patients can really benefit from phototherapy,” he said in an interview. (Labeling recommends against combination with other JAK inhibitors, biologics, or potent immunosuppressants, such as azathioprine or cyclosporine.)
This first year has shown that ruxolitinib is an effective option, but counseling patients to expect slow improvement is important so that patients stick with it, he noted.
Documenting what treatments patients with vitiligo have used before is important, he said, as is counseling patients that ruxolitinib is approved only for use on up to 10% of a person’s body surface area. (Product labeling recommends that a thin layer be applied twice a day to affected areas up to 10% of body surface area.)
Ruxolitinib has brought a “louder voice” to vitiligo and has opened up options for patients with the disease, Dr. Chovatiya said. “Having the ability to topically treat people who have very extensive disease really gives us a lot more flexibility than we have had before.”
Good experiences with payers at safety-net hospital
Candrice R. Heath, MD, assistant professor of dermatology at Temple University, Philadelphia, said that real-world experience with topical ruxolitinib will be more evident after its been on the market for 18-24 months.
Dr. Heath said she, too, encourages use of narrow-band UVB phototherapy in conjunction with the treatment.
From an insurance reimbursement standpoint, she said that she is glad that there have been fewer hurdles in getting ruxolitinib to patients than she has experienced with other medications.
In her safety-net hospital, she told this news organization, she sees patients with many types of insurance, but most have Medicaid. “So, I’m always expecting the step therapies, denials, pushbacks, etc.,” she said. But the path has been smoother for ruxolitinib coverage, she noted.
Her colleagues are committed to documenting everything the patient has tried, she added, and that helps with prior authorization.
Dr. Heath said that pointing out to insurers that ruxolitinib is the only approved treatment for repigmentation helps facilitate coverage.
“The science is advancing, and I’m happy to be practicing during a time when we actually have something approved for vitiligo,” she said. But she pointed out that phototherapy often is not covered for vitiligo, “which is horrible, when it is readily approved for psoriasis and atopic dermatitis.”
To document progress, Dr. Heath said that she always takes photographs of her patients with vitiligo because “the pictures remind us how far we have come.”
Data spotlight success in adolescents
Data from two trials give a clinical picture of the drug’s safety and efficacy in younger patients.
Adolescents had particularly good results in the first year with ruxolitinib, according to pooled phase 3 data from TRuE-V1 and TRuE-V2, this news organization reported.
The findings, presented at the 25th World Congress of Dermatology in Singapore, indicate that more than half of the participants achieved at least a 50% improvement from baseline in the total Vitiligo Area Scoring Index (T-VASI50) at 52 weeks.
The percentages of young patients aged 12-17 years taking twice-daily ruxolitinib who achieved T-VASI 50 at weeks 12, 24, and 52 were 11.5%, 26.9%, and 57.7%, respectively. The corresponding percentages for all in the study population were 10.7%, 22.7%, and 44.4%, respectively.
At the meeting, the presenter, Julien Seneschal, MD, PhD, professor of dermatology and head of the vitiligo and pigmentary disorders clinic at the University of Bordeaux, France, said, “This suggests that younger patients can respond better to the treatment.” He noted, however, that there were few adolescents in the studies.
New excitement in the field
Daniel Gutierrez, MD, assistant professor of dermatology at New York University, said the treatment has brought new excitement to the field.
“Patients with vitiligo are very motivated to treat their disease,” he said, because it typically is on the face and other highly visual areas, which can affect their overall perception of self.
Previously, he noted in an interview, the only FDA-approved treatment was monobenzone, but that was for depigmentation rather than repigmentation.
Otherwise, treatments were being used off label, and patients were receiving compounded formulations that often weren’t covered by insurance and often had shorter shelf life.
He said that he still occasionally gets denials from payers who consider vitiligo a cosmetic condition.
“I’ve had more luck with insurance, at least in the New York State area.” He added that sometimes payers require use of a topical calcineurin inhibitor for about 12 weeks before they will cover ruxolitinib.
Dr. Gutierrez also recommends using phototherapy with topical ruxolitinib “because they work on slightly different pathways.”
When he starts patients on a new therapy such as ruxolitinib, he asks them to come back in 3 months, and often by then, progress is evident. Facial areas show the most response, he said, while hands and feet are less likely to show significant improvement.
He said that it’s important for physicians and patients to know that improvements can take weeks or months to be noticeable. “I tell patients not to give up,” he added.
Showing the patients pictures from the current appointment and comparing them with pictures from previous appointments can help them better understand their progress, he said.
Lead investigator adds observations
David Rosmarin, MD, chair of the department of dermatology at Indiana University, Indianapolis, was the lead investigator of the pivotal TruE-V1 and TruE-V2 trials for vitiligo. In that role, he has been treating vitiligo patients with topical ruxolitinib since 2015.
In an interview, he said that many patients “don’t hit their optimal results at 3 months, 6 months, even the year mark. With continued use, many can see continued benefit.”
Other patients, he said, don’t respond within the first 6 months but with continued use may eventually respond, he said.
“Unfortunately, we have no way of knowing, based on clinical characteristics or baseline demographics, whether a patient will be a delayed responder or not or an early responder,” Dr. Rosmarin added.
He provided several observations about people who have stopped taking the medication.
“When people stop,” he said, “some maintain their response, but some start to depigment again. Again, we have no way of predicting who will be in which category.”
He said that once patients have hit their desired response, he usually advises them to taper down to maybe twice a week or to stop treatment, but if they see any recurrence, they should start reusing the medicine.
“We have some patients who have gone 6 or 7 years now before they had a recurrence, but others may start to depigment again in 2 to 3 months,” Dr. Rosmarin said.
As for phototherapy, he said, the combination with topical ruxolitinib is being studied.
“We think the combination is synergistic and better than either alone, but we’re still waiting for data to prove that,” he said.
In his practice, he offers patients the option either to use just ruxolitinib cream or the combination early on. Many patients, because of convenience, say they’ll first try the cream to see if that works.
“The challenge with light [therapy] is that it can be very inconvenient,” he said. Patients have to live close to a phototherapy unit to receive therapy 2-3 times a week or have a phototherapy product in their home.
Next in the pipeline
Experts say the progress doesn’t stop with ruxolitinib cream. Current trials of several medications show there’s more to come for patients with vitiligo.
Dr. Chovatiya said that next up may be oral ritlecitinib (Litfulo), a JAK inhibitor that was approved for severe alopecia areata in June for people aged 12 years and older. Phase 2 results have been published for its use with vitiligo.
“This would be an oral medication that may be able to help people with much more extensive disease as far as vitiligo goes,” he said, adding that he expects approval for a vitiligo indication within a few years.
He pointed out that longer-term safety data will be available because it is already on the market for alopecia.
Upadacitinib (Rinvoq), an oral JAK inhibitor, is approved for atopic dermatitis but is being studied for vitiligo as well, he noted. “I’m very excited to see what that holds for patients as well,” Dr. Chovatiya said.
Dr. Gutierrez said that he is excited about oral JAK inhibitors but sees potential in finding new ways to transplant melanocytes into areas where there are none.
The pigmentation field has seen new energy since last year’s approval, he said, particularly among people of color.
“We have new options for vitiligo that were lacking compared with other conditions, such as atopic dermatitis and psoriasis,” he said. “Hopefully, there will be more promising breakthroughs.”
Dr. Rosmarin is the chief investigator for the pivotal trials that led to FDA approval of ruxolitinib. He disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena, Boehringer Ingelheim, Bristol-Meyers Squibb, Celgene, Concert, CSL Behring, Dermavant, Dermira, Galderma, Incyte, Janssen, Kyowa Kirin, Lilly, Merck, Novartis, Pfizer, Regeneron, Revolo Biotherapeutics, Sanofi, Sun Pharmaceuticals, UCB, and Viela Bio. Dr. Chovatiya disclosed ties with AbbVie, Arcutis, Arena, Argenx, Beiersdorf, Bristol-Myers Squibb, Dermavant, Eli Lilly, EPI Health, Incyte, LEO Pharma, L’Oréal, National Eczema Association, Pfizer, Regeneron, Sanofi, and UCB. Dr. Heath and Dr. Gutierrez report no relevant financial relationships.
A version of this article appeared on Medscape.com.
.
The Food and Drug Administration approved the cream formulation of ruxolitinib (Opzelura), a JAK inhibitor, for repigmentation of nonsegmental vitiligo in July 2022 for people aged 12 years and older.
Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, said that he likes to use ruxolitinib cream in combination with other treatments.
“In the real world with vitiligo patients, we’re oftentimes doing combinatorial therapy anyway. So phototherapy, specifically, narrow-band UVB, is something that we have a lot of clinical evidence for over the years, and it’s a modality that can combine with topical steroids and topical calcineurin inhibitors.”
He said trials to study combinations will yield better guidance on optimal use of ruxolitinib cream. “In general, vitiligo patients can really benefit from phototherapy,” he said in an interview. (Labeling recommends against combination with other JAK inhibitors, biologics, or potent immunosuppressants, such as azathioprine or cyclosporine.)
This first year has shown that ruxolitinib is an effective option, but counseling patients to expect slow improvement is important so that patients stick with it, he noted.
Documenting what treatments patients with vitiligo have used before is important, he said, as is counseling patients that ruxolitinib is approved only for use on up to 10% of a person’s body surface area. (Product labeling recommends that a thin layer be applied twice a day to affected areas up to 10% of body surface area.)
Ruxolitinib has brought a “louder voice” to vitiligo and has opened up options for patients with the disease, Dr. Chovatiya said. “Having the ability to topically treat people who have very extensive disease really gives us a lot more flexibility than we have had before.”
Good experiences with payers at safety-net hospital
Candrice R. Heath, MD, assistant professor of dermatology at Temple University, Philadelphia, said that real-world experience with topical ruxolitinib will be more evident after its been on the market for 18-24 months.
Dr. Heath said she, too, encourages use of narrow-band UVB phototherapy in conjunction with the treatment.
From an insurance reimbursement standpoint, she said that she is glad that there have been fewer hurdles in getting ruxolitinib to patients than she has experienced with other medications.
In her safety-net hospital, she told this news organization, she sees patients with many types of insurance, but most have Medicaid. “So, I’m always expecting the step therapies, denials, pushbacks, etc.,” she said. But the path has been smoother for ruxolitinib coverage, she noted.
Her colleagues are committed to documenting everything the patient has tried, she added, and that helps with prior authorization.
Dr. Heath said that pointing out to insurers that ruxolitinib is the only approved treatment for repigmentation helps facilitate coverage.
“The science is advancing, and I’m happy to be practicing during a time when we actually have something approved for vitiligo,” she said. But she pointed out that phototherapy often is not covered for vitiligo, “which is horrible, when it is readily approved for psoriasis and atopic dermatitis.”
To document progress, Dr. Heath said that she always takes photographs of her patients with vitiligo because “the pictures remind us how far we have come.”
Data spotlight success in adolescents
Data from two trials give a clinical picture of the drug’s safety and efficacy in younger patients.
Adolescents had particularly good results in the first year with ruxolitinib, according to pooled phase 3 data from TRuE-V1 and TRuE-V2, this news organization reported.
The findings, presented at the 25th World Congress of Dermatology in Singapore, indicate that more than half of the participants achieved at least a 50% improvement from baseline in the total Vitiligo Area Scoring Index (T-VASI50) at 52 weeks.
The percentages of young patients aged 12-17 years taking twice-daily ruxolitinib who achieved T-VASI 50 at weeks 12, 24, and 52 were 11.5%, 26.9%, and 57.7%, respectively. The corresponding percentages for all in the study population were 10.7%, 22.7%, and 44.4%, respectively.
At the meeting, the presenter, Julien Seneschal, MD, PhD, professor of dermatology and head of the vitiligo and pigmentary disorders clinic at the University of Bordeaux, France, said, “This suggests that younger patients can respond better to the treatment.” He noted, however, that there were few adolescents in the studies.
New excitement in the field
Daniel Gutierrez, MD, assistant professor of dermatology at New York University, said the treatment has brought new excitement to the field.
“Patients with vitiligo are very motivated to treat their disease,” he said, because it typically is on the face and other highly visual areas, which can affect their overall perception of self.
Previously, he noted in an interview, the only FDA-approved treatment was monobenzone, but that was for depigmentation rather than repigmentation.
Otherwise, treatments were being used off label, and patients were receiving compounded formulations that often weren’t covered by insurance and often had shorter shelf life.
He said that he still occasionally gets denials from payers who consider vitiligo a cosmetic condition.
“I’ve had more luck with insurance, at least in the New York State area.” He added that sometimes payers require use of a topical calcineurin inhibitor for about 12 weeks before they will cover ruxolitinib.
Dr. Gutierrez also recommends using phototherapy with topical ruxolitinib “because they work on slightly different pathways.”
When he starts patients on a new therapy such as ruxolitinib, he asks them to come back in 3 months, and often by then, progress is evident. Facial areas show the most response, he said, while hands and feet are less likely to show significant improvement.
He said that it’s important for physicians and patients to know that improvements can take weeks or months to be noticeable. “I tell patients not to give up,” he added.
Showing the patients pictures from the current appointment and comparing them with pictures from previous appointments can help them better understand their progress, he said.
Lead investigator adds observations
David Rosmarin, MD, chair of the department of dermatology at Indiana University, Indianapolis, was the lead investigator of the pivotal TruE-V1 and TruE-V2 trials for vitiligo. In that role, he has been treating vitiligo patients with topical ruxolitinib since 2015.
In an interview, he said that many patients “don’t hit their optimal results at 3 months, 6 months, even the year mark. With continued use, many can see continued benefit.”
Other patients, he said, don’t respond within the first 6 months but with continued use may eventually respond, he said.
“Unfortunately, we have no way of knowing, based on clinical characteristics or baseline demographics, whether a patient will be a delayed responder or not or an early responder,” Dr. Rosmarin added.
He provided several observations about people who have stopped taking the medication.
“When people stop,” he said, “some maintain their response, but some start to depigment again. Again, we have no way of predicting who will be in which category.”
He said that once patients have hit their desired response, he usually advises them to taper down to maybe twice a week or to stop treatment, but if they see any recurrence, they should start reusing the medicine.
“We have some patients who have gone 6 or 7 years now before they had a recurrence, but others may start to depigment again in 2 to 3 months,” Dr. Rosmarin said.
As for phototherapy, he said, the combination with topical ruxolitinib is being studied.
“We think the combination is synergistic and better than either alone, but we’re still waiting for data to prove that,” he said.
In his practice, he offers patients the option either to use just ruxolitinib cream or the combination early on. Many patients, because of convenience, say they’ll first try the cream to see if that works.
“The challenge with light [therapy] is that it can be very inconvenient,” he said. Patients have to live close to a phototherapy unit to receive therapy 2-3 times a week or have a phototherapy product in their home.
Next in the pipeline
Experts say the progress doesn’t stop with ruxolitinib cream. Current trials of several medications show there’s more to come for patients with vitiligo.
Dr. Chovatiya said that next up may be oral ritlecitinib (Litfulo), a JAK inhibitor that was approved for severe alopecia areata in June for people aged 12 years and older. Phase 2 results have been published for its use with vitiligo.
“This would be an oral medication that may be able to help people with much more extensive disease as far as vitiligo goes,” he said, adding that he expects approval for a vitiligo indication within a few years.
He pointed out that longer-term safety data will be available because it is already on the market for alopecia.
Upadacitinib (Rinvoq), an oral JAK inhibitor, is approved for atopic dermatitis but is being studied for vitiligo as well, he noted. “I’m very excited to see what that holds for patients as well,” Dr. Chovatiya said.
Dr. Gutierrez said that he is excited about oral JAK inhibitors but sees potential in finding new ways to transplant melanocytes into areas where there are none.
The pigmentation field has seen new energy since last year’s approval, he said, particularly among people of color.
“We have new options for vitiligo that were lacking compared with other conditions, such as atopic dermatitis and psoriasis,” he said. “Hopefully, there will be more promising breakthroughs.”
Dr. Rosmarin is the chief investigator for the pivotal trials that led to FDA approval of ruxolitinib. He disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena, Boehringer Ingelheim, Bristol-Meyers Squibb, Celgene, Concert, CSL Behring, Dermavant, Dermira, Galderma, Incyte, Janssen, Kyowa Kirin, Lilly, Merck, Novartis, Pfizer, Regeneron, Revolo Biotherapeutics, Sanofi, Sun Pharmaceuticals, UCB, and Viela Bio. Dr. Chovatiya disclosed ties with AbbVie, Arcutis, Arena, Argenx, Beiersdorf, Bristol-Myers Squibb, Dermavant, Eli Lilly, EPI Health, Incyte, LEO Pharma, L’Oréal, National Eczema Association, Pfizer, Regeneron, Sanofi, and UCB. Dr. Heath and Dr. Gutierrez report no relevant financial relationships.
A version of this article appeared on Medscape.com.
.
The Food and Drug Administration approved the cream formulation of ruxolitinib (Opzelura), a JAK inhibitor, for repigmentation of nonsegmental vitiligo in July 2022 for people aged 12 years and older.
Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, said that he likes to use ruxolitinib cream in combination with other treatments.
“In the real world with vitiligo patients, we’re oftentimes doing combinatorial therapy anyway. So phototherapy, specifically, narrow-band UVB, is something that we have a lot of clinical evidence for over the years, and it’s a modality that can combine with topical steroids and topical calcineurin inhibitors.”
He said trials to study combinations will yield better guidance on optimal use of ruxolitinib cream. “In general, vitiligo patients can really benefit from phototherapy,” he said in an interview. (Labeling recommends against combination with other JAK inhibitors, biologics, or potent immunosuppressants, such as azathioprine or cyclosporine.)
This first year has shown that ruxolitinib is an effective option, but counseling patients to expect slow improvement is important so that patients stick with it, he noted.
Documenting what treatments patients with vitiligo have used before is important, he said, as is counseling patients that ruxolitinib is approved only for use on up to 10% of a person’s body surface area. (Product labeling recommends that a thin layer be applied twice a day to affected areas up to 10% of body surface area.)
Ruxolitinib has brought a “louder voice” to vitiligo and has opened up options for patients with the disease, Dr. Chovatiya said. “Having the ability to topically treat people who have very extensive disease really gives us a lot more flexibility than we have had before.”
Good experiences with payers at safety-net hospital
Candrice R. Heath, MD, assistant professor of dermatology at Temple University, Philadelphia, said that real-world experience with topical ruxolitinib will be more evident after its been on the market for 18-24 months.
Dr. Heath said she, too, encourages use of narrow-band UVB phototherapy in conjunction with the treatment.
From an insurance reimbursement standpoint, she said that she is glad that there have been fewer hurdles in getting ruxolitinib to patients than she has experienced with other medications.
In her safety-net hospital, she told this news organization, she sees patients with many types of insurance, but most have Medicaid. “So, I’m always expecting the step therapies, denials, pushbacks, etc.,” she said. But the path has been smoother for ruxolitinib coverage, she noted.
Her colleagues are committed to documenting everything the patient has tried, she added, and that helps with prior authorization.
Dr. Heath said that pointing out to insurers that ruxolitinib is the only approved treatment for repigmentation helps facilitate coverage.
“The science is advancing, and I’m happy to be practicing during a time when we actually have something approved for vitiligo,” she said. But she pointed out that phototherapy often is not covered for vitiligo, “which is horrible, when it is readily approved for psoriasis and atopic dermatitis.”
To document progress, Dr. Heath said that she always takes photographs of her patients with vitiligo because “the pictures remind us how far we have come.”
Data spotlight success in adolescents
Data from two trials give a clinical picture of the drug’s safety and efficacy in younger patients.
Adolescents had particularly good results in the first year with ruxolitinib, according to pooled phase 3 data from TRuE-V1 and TRuE-V2, this news organization reported.
The findings, presented at the 25th World Congress of Dermatology in Singapore, indicate that more than half of the participants achieved at least a 50% improvement from baseline in the total Vitiligo Area Scoring Index (T-VASI50) at 52 weeks.
The percentages of young patients aged 12-17 years taking twice-daily ruxolitinib who achieved T-VASI 50 at weeks 12, 24, and 52 were 11.5%, 26.9%, and 57.7%, respectively. The corresponding percentages for all in the study population were 10.7%, 22.7%, and 44.4%, respectively.
At the meeting, the presenter, Julien Seneschal, MD, PhD, professor of dermatology and head of the vitiligo and pigmentary disorders clinic at the University of Bordeaux, France, said, “This suggests that younger patients can respond better to the treatment.” He noted, however, that there were few adolescents in the studies.
New excitement in the field
Daniel Gutierrez, MD, assistant professor of dermatology at New York University, said the treatment has brought new excitement to the field.
“Patients with vitiligo are very motivated to treat their disease,” he said, because it typically is on the face and other highly visual areas, which can affect their overall perception of self.
Previously, he noted in an interview, the only FDA-approved treatment was monobenzone, but that was for depigmentation rather than repigmentation.
Otherwise, treatments were being used off label, and patients were receiving compounded formulations that often weren’t covered by insurance and often had shorter shelf life.
He said that he still occasionally gets denials from payers who consider vitiligo a cosmetic condition.
“I’ve had more luck with insurance, at least in the New York State area.” He added that sometimes payers require use of a topical calcineurin inhibitor for about 12 weeks before they will cover ruxolitinib.
Dr. Gutierrez also recommends using phototherapy with topical ruxolitinib “because they work on slightly different pathways.”
When he starts patients on a new therapy such as ruxolitinib, he asks them to come back in 3 months, and often by then, progress is evident. Facial areas show the most response, he said, while hands and feet are less likely to show significant improvement.
He said that it’s important for physicians and patients to know that improvements can take weeks or months to be noticeable. “I tell patients not to give up,” he added.
Showing the patients pictures from the current appointment and comparing them with pictures from previous appointments can help them better understand their progress, he said.
Lead investigator adds observations
David Rosmarin, MD, chair of the department of dermatology at Indiana University, Indianapolis, was the lead investigator of the pivotal TruE-V1 and TruE-V2 trials for vitiligo. In that role, he has been treating vitiligo patients with topical ruxolitinib since 2015.
In an interview, he said that many patients “don’t hit their optimal results at 3 months, 6 months, even the year mark. With continued use, many can see continued benefit.”
Other patients, he said, don’t respond within the first 6 months but with continued use may eventually respond, he said.
“Unfortunately, we have no way of knowing, based on clinical characteristics or baseline demographics, whether a patient will be a delayed responder or not or an early responder,” Dr. Rosmarin added.
He provided several observations about people who have stopped taking the medication.
“When people stop,” he said, “some maintain their response, but some start to depigment again. Again, we have no way of predicting who will be in which category.”
He said that once patients have hit their desired response, he usually advises them to taper down to maybe twice a week or to stop treatment, but if they see any recurrence, they should start reusing the medicine.
“We have some patients who have gone 6 or 7 years now before they had a recurrence, but others may start to depigment again in 2 to 3 months,” Dr. Rosmarin said.
As for phototherapy, he said, the combination with topical ruxolitinib is being studied.
“We think the combination is synergistic and better than either alone, but we’re still waiting for data to prove that,” he said.
In his practice, he offers patients the option either to use just ruxolitinib cream or the combination early on. Many patients, because of convenience, say they’ll first try the cream to see if that works.
“The challenge with light [therapy] is that it can be very inconvenient,” he said. Patients have to live close to a phototherapy unit to receive therapy 2-3 times a week or have a phototherapy product in their home.
Next in the pipeline
Experts say the progress doesn’t stop with ruxolitinib cream. Current trials of several medications show there’s more to come for patients with vitiligo.
Dr. Chovatiya said that next up may be oral ritlecitinib (Litfulo), a JAK inhibitor that was approved for severe alopecia areata in June for people aged 12 years and older. Phase 2 results have been published for its use with vitiligo.
“This would be an oral medication that may be able to help people with much more extensive disease as far as vitiligo goes,” he said, adding that he expects approval for a vitiligo indication within a few years.
He pointed out that longer-term safety data will be available because it is already on the market for alopecia.
Upadacitinib (Rinvoq), an oral JAK inhibitor, is approved for atopic dermatitis but is being studied for vitiligo as well, he noted. “I’m very excited to see what that holds for patients as well,” Dr. Chovatiya said.
Dr. Gutierrez said that he is excited about oral JAK inhibitors but sees potential in finding new ways to transplant melanocytes into areas where there are none.
The pigmentation field has seen new energy since last year’s approval, he said, particularly among people of color.
“We have new options for vitiligo that were lacking compared with other conditions, such as atopic dermatitis and psoriasis,” he said. “Hopefully, there will be more promising breakthroughs.”
Dr. Rosmarin is the chief investigator for the pivotal trials that led to FDA approval of ruxolitinib. He disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena, Boehringer Ingelheim, Bristol-Meyers Squibb, Celgene, Concert, CSL Behring, Dermavant, Dermira, Galderma, Incyte, Janssen, Kyowa Kirin, Lilly, Merck, Novartis, Pfizer, Regeneron, Revolo Biotherapeutics, Sanofi, Sun Pharmaceuticals, UCB, and Viela Bio. Dr. Chovatiya disclosed ties with AbbVie, Arcutis, Arena, Argenx, Beiersdorf, Bristol-Myers Squibb, Dermavant, Eli Lilly, EPI Health, Incyte, LEO Pharma, L’Oréal, National Eczema Association, Pfizer, Regeneron, Sanofi, and UCB. Dr. Heath and Dr. Gutierrez report no relevant financial relationships.
A version of this article appeared on Medscape.com.
FDA to step up oversight of cosmetics, assess ‘forever chemicals’
They are also preparing to assess potential risks of so-called forever chemicals in these products.
The Food and Drug Administration last year gained new authority over cosmetics when Congress passed the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) by adding this bill to a December budget package.
“On average, consumers in the U.S. use six to 12 cosmetics products daily. But, until recently the FDA didn’t have the authority to require manufacturers to submit cosmetic product listings, including a list of ingredients used in these products, or register the facilities where they were produced,” Namandjé Bumpus, PhD, FDA’s chief scientist, said in a press release.
In the statement, the FDA announced the release of a draft guidance document that is intended to help companies comply with the transparency requirements slated to kick in this December. The agency is accepting comments on this draft guidance through Sept. 7.
“Later this year, registration and listing of cosmetic product facilities and products will become a requirement, making information about cosmetic products, including the ingredients used in products and the facilities where they are produced, readily available to the agency,” Dr. Bumpus said.
The products, according to the FDA statement, include makeup, nail polishes, shaving creams, other grooming products, perfumes, face and body cleansers, hair products, moisturizers, and other skin care items.
MoCRA “represents a sea change in how FDA regulates the cosmetics industry,” attorneys Frederick R. Ball, Alyson Walker Lotman, and Kelly A. Bonner, wrote in an article for the Food and Drug Law Institute published in spring 2023.
The FDA has called the MoCRA law “the most significant expansion” of its authority to regulate cosmetics since the Federal Food, Drug, and Cosmetic Act was passed in 1938.
The agency is in the process of expanding its staff to carry out newly authorized duties, including the tracking of adverse events. The FDA budget request for fiscal 2024, which begins Oct. 1, seeks $5 million for work needed to implement MoCRA.
PFAS, or ‘forever chemicals’
Some of the requested FDA funding is intended to prepare the agency to assess the use of per-and polyfluoroalkyl substances (PFAS) in cosmetics.
MoCRA sets a 3-year deadline for the FDA to issue an assessment of the use and potential risks of PFAS in cosmetics products. PFAS are sometimes added as ingredients in some cosmetic products, including lotions, cleansers, nail polish, shaving cream, foundation, lipstick, eyeliner, eyeshadow, and mascara, according to the FDA. Sometimes the presence of PFAS in cosmetics is unintentional and is the result of impurities in raw materials or is due to the breakdown of ingredients, the FDA said.
The FDA’s website says that so far, the available research doesn’t allow for “definitive conclusions about the potential health risks of PFAS in cosmetics.”
The Centers for Disease Control and Prevention has stated that research has suggested potential links between high levels of certain PFAS, in general, with increased cholesterol levels, changes in liver enzyme levels, increased risk of hypertension or preeclampsia in pregnant women, and increased risk of kidney or testicular cancer.
PFAS compounds often are used to resist grease, oil, water, and heat in industrial settings. They are used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they can reduce friction, according to a National Academies of Sciences, Engineering, and Medicine report on PFAS that was issued last year.
PFAS are known as “forever chemicals” because they contain a carbon-fluorine bond, which does not break naturally. Even when PFAS are transformed in the body, they can assume other forms of PFAS that preserve the troublesome carbon-fluorine bond. With PFAS, the human body is confronted with a substance it doesn’t have the tools to process.
This is in contrast to proteins and carbohydrates, which are in a sense prepackaged for relatively easy disassembly in the human body. Many of these compounds have weak links that enzymes and stomach acid can take apart, such as sulfur-to-sulfur (disulfide) bonds. That’s why protein-based biotech drugs are injected instead of administered as pills. The ultimate goal of this digestion is for the body to gain energy from these compounds.
But with PFAS, the body faces the challenge of carbon-fluorine bonds that are very hard to break down, and there is no payoff for these efforts, Graham F. Peaslee, PhD, professor of physics at the University of Notre Dame (Indiana), told this news organization.
“Nothing will naturally eat it because when you break the bond, it’s like eating celery,” he said. “You use more calories to eat the celery than you gain back from it.”
Interest from a U.S. senator
Dr. Peaslee was one of the authors of a 2021 article about PFAS in cosmetics that appeared in the journal Environmental Science and Technology Letters.
In the article, Dr. Peaslee and colleagues reported on their screening of 231 cosmetic products purchased in the United States and Canada using particle-induced gamma-ray emission spectroscopy. They found cases of undisclosed PFAS in cosmetic products. Foundations, mascaras, and lip products were noted as being especially problematic.
Sen. Susan Collins (R-ME) cited Dr. Peaslee’s article in a 2021 floor speech as she argued for having the FDA ban the intentional addition of PFAS to cosmetics.
“The findings of this study are particularly alarming, as many of these products are subject to direct human exposure,” Sen. Collins said. “For example, lipstick is often inadvertently ingested, and mascara is sometimes absorbed through tear ducts.”
In addition, workers at cosmetics plants may be exposed to PFAS and discarded cosmetics that have these compounds, which could potentially contaminate drinking water, Sen. Collins said. In 2021, she introduced legislation seeking a ban on PFAS that are intentionally added to cosmetics. That legislation did not advance through the Senate.
But the Senate Appropriations Committee, on which Sen. Collins is the ranking Republican, wants the FDA to keep a ban on PFAS in mind.
The Senate Agriculture Appropriations subcommittee, which oversees the FDA’s budget, raised the issue of PFAS and cosmetics in a June report. The FDA should develop a plan outlining research needed to inform “regulatory decision making, including potential development of a proposed rule to ban intentionally added PFAS substances in cosmetics,” the subcommittee said.
A version of this article first appeared on Medscape.com.
They are also preparing to assess potential risks of so-called forever chemicals in these products.
The Food and Drug Administration last year gained new authority over cosmetics when Congress passed the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) by adding this bill to a December budget package.
“On average, consumers in the U.S. use six to 12 cosmetics products daily. But, until recently the FDA didn’t have the authority to require manufacturers to submit cosmetic product listings, including a list of ingredients used in these products, or register the facilities where they were produced,” Namandjé Bumpus, PhD, FDA’s chief scientist, said in a press release.
In the statement, the FDA announced the release of a draft guidance document that is intended to help companies comply with the transparency requirements slated to kick in this December. The agency is accepting comments on this draft guidance through Sept. 7.
“Later this year, registration and listing of cosmetic product facilities and products will become a requirement, making information about cosmetic products, including the ingredients used in products and the facilities where they are produced, readily available to the agency,” Dr. Bumpus said.
The products, according to the FDA statement, include makeup, nail polishes, shaving creams, other grooming products, perfumes, face and body cleansers, hair products, moisturizers, and other skin care items.
MoCRA “represents a sea change in how FDA regulates the cosmetics industry,” attorneys Frederick R. Ball, Alyson Walker Lotman, and Kelly A. Bonner, wrote in an article for the Food and Drug Law Institute published in spring 2023.
The FDA has called the MoCRA law “the most significant expansion” of its authority to regulate cosmetics since the Federal Food, Drug, and Cosmetic Act was passed in 1938.
The agency is in the process of expanding its staff to carry out newly authorized duties, including the tracking of adverse events. The FDA budget request for fiscal 2024, which begins Oct. 1, seeks $5 million for work needed to implement MoCRA.
PFAS, or ‘forever chemicals’
Some of the requested FDA funding is intended to prepare the agency to assess the use of per-and polyfluoroalkyl substances (PFAS) in cosmetics.
MoCRA sets a 3-year deadline for the FDA to issue an assessment of the use and potential risks of PFAS in cosmetics products. PFAS are sometimes added as ingredients in some cosmetic products, including lotions, cleansers, nail polish, shaving cream, foundation, lipstick, eyeliner, eyeshadow, and mascara, according to the FDA. Sometimes the presence of PFAS in cosmetics is unintentional and is the result of impurities in raw materials or is due to the breakdown of ingredients, the FDA said.
The FDA’s website says that so far, the available research doesn’t allow for “definitive conclusions about the potential health risks of PFAS in cosmetics.”
The Centers for Disease Control and Prevention has stated that research has suggested potential links between high levels of certain PFAS, in general, with increased cholesterol levels, changes in liver enzyme levels, increased risk of hypertension or preeclampsia in pregnant women, and increased risk of kidney or testicular cancer.
PFAS compounds often are used to resist grease, oil, water, and heat in industrial settings. They are used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they can reduce friction, according to a National Academies of Sciences, Engineering, and Medicine report on PFAS that was issued last year.
PFAS are known as “forever chemicals” because they contain a carbon-fluorine bond, which does not break naturally. Even when PFAS are transformed in the body, they can assume other forms of PFAS that preserve the troublesome carbon-fluorine bond. With PFAS, the human body is confronted with a substance it doesn’t have the tools to process.
This is in contrast to proteins and carbohydrates, which are in a sense prepackaged for relatively easy disassembly in the human body. Many of these compounds have weak links that enzymes and stomach acid can take apart, such as sulfur-to-sulfur (disulfide) bonds. That’s why protein-based biotech drugs are injected instead of administered as pills. The ultimate goal of this digestion is for the body to gain energy from these compounds.
But with PFAS, the body faces the challenge of carbon-fluorine bonds that are very hard to break down, and there is no payoff for these efforts, Graham F. Peaslee, PhD, professor of physics at the University of Notre Dame (Indiana), told this news organization.
“Nothing will naturally eat it because when you break the bond, it’s like eating celery,” he said. “You use more calories to eat the celery than you gain back from it.”
Interest from a U.S. senator
Dr. Peaslee was one of the authors of a 2021 article about PFAS in cosmetics that appeared in the journal Environmental Science and Technology Letters.
In the article, Dr. Peaslee and colleagues reported on their screening of 231 cosmetic products purchased in the United States and Canada using particle-induced gamma-ray emission spectroscopy. They found cases of undisclosed PFAS in cosmetic products. Foundations, mascaras, and lip products were noted as being especially problematic.
Sen. Susan Collins (R-ME) cited Dr. Peaslee’s article in a 2021 floor speech as she argued for having the FDA ban the intentional addition of PFAS to cosmetics.
“The findings of this study are particularly alarming, as many of these products are subject to direct human exposure,” Sen. Collins said. “For example, lipstick is often inadvertently ingested, and mascara is sometimes absorbed through tear ducts.”
In addition, workers at cosmetics plants may be exposed to PFAS and discarded cosmetics that have these compounds, which could potentially contaminate drinking water, Sen. Collins said. In 2021, she introduced legislation seeking a ban on PFAS that are intentionally added to cosmetics. That legislation did not advance through the Senate.
But the Senate Appropriations Committee, on which Sen. Collins is the ranking Republican, wants the FDA to keep a ban on PFAS in mind.
The Senate Agriculture Appropriations subcommittee, which oversees the FDA’s budget, raised the issue of PFAS and cosmetics in a June report. The FDA should develop a plan outlining research needed to inform “regulatory decision making, including potential development of a proposed rule to ban intentionally added PFAS substances in cosmetics,” the subcommittee said.
A version of this article first appeared on Medscape.com.
They are also preparing to assess potential risks of so-called forever chemicals in these products.
The Food and Drug Administration last year gained new authority over cosmetics when Congress passed the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) by adding this bill to a December budget package.
“On average, consumers in the U.S. use six to 12 cosmetics products daily. But, until recently the FDA didn’t have the authority to require manufacturers to submit cosmetic product listings, including a list of ingredients used in these products, or register the facilities where they were produced,” Namandjé Bumpus, PhD, FDA’s chief scientist, said in a press release.
In the statement, the FDA announced the release of a draft guidance document that is intended to help companies comply with the transparency requirements slated to kick in this December. The agency is accepting comments on this draft guidance through Sept. 7.
“Later this year, registration and listing of cosmetic product facilities and products will become a requirement, making information about cosmetic products, including the ingredients used in products and the facilities where they are produced, readily available to the agency,” Dr. Bumpus said.
The products, according to the FDA statement, include makeup, nail polishes, shaving creams, other grooming products, perfumes, face and body cleansers, hair products, moisturizers, and other skin care items.
MoCRA “represents a sea change in how FDA regulates the cosmetics industry,” attorneys Frederick R. Ball, Alyson Walker Lotman, and Kelly A. Bonner, wrote in an article for the Food and Drug Law Institute published in spring 2023.
The FDA has called the MoCRA law “the most significant expansion” of its authority to regulate cosmetics since the Federal Food, Drug, and Cosmetic Act was passed in 1938.
The agency is in the process of expanding its staff to carry out newly authorized duties, including the tracking of adverse events. The FDA budget request for fiscal 2024, which begins Oct. 1, seeks $5 million for work needed to implement MoCRA.
PFAS, or ‘forever chemicals’
Some of the requested FDA funding is intended to prepare the agency to assess the use of per-and polyfluoroalkyl substances (PFAS) in cosmetics.
MoCRA sets a 3-year deadline for the FDA to issue an assessment of the use and potential risks of PFAS in cosmetics products. PFAS are sometimes added as ingredients in some cosmetic products, including lotions, cleansers, nail polish, shaving cream, foundation, lipstick, eyeliner, eyeshadow, and mascara, according to the FDA. Sometimes the presence of PFAS in cosmetics is unintentional and is the result of impurities in raw materials or is due to the breakdown of ingredients, the FDA said.
The FDA’s website says that so far, the available research doesn’t allow for “definitive conclusions about the potential health risks of PFAS in cosmetics.”
The Centers for Disease Control and Prevention has stated that research has suggested potential links between high levels of certain PFAS, in general, with increased cholesterol levels, changes in liver enzyme levels, increased risk of hypertension or preeclampsia in pregnant women, and increased risk of kidney or testicular cancer.
PFAS compounds often are used to resist grease, oil, water, and heat in industrial settings. They are used in thousands of products, from nonstick cookware to firefighting foams and protective gear, because they can reduce friction, according to a National Academies of Sciences, Engineering, and Medicine report on PFAS that was issued last year.
PFAS are known as “forever chemicals” because they contain a carbon-fluorine bond, which does not break naturally. Even when PFAS are transformed in the body, they can assume other forms of PFAS that preserve the troublesome carbon-fluorine bond. With PFAS, the human body is confronted with a substance it doesn’t have the tools to process.
This is in contrast to proteins and carbohydrates, which are in a sense prepackaged for relatively easy disassembly in the human body. Many of these compounds have weak links that enzymes and stomach acid can take apart, such as sulfur-to-sulfur (disulfide) bonds. That’s why protein-based biotech drugs are injected instead of administered as pills. The ultimate goal of this digestion is for the body to gain energy from these compounds.
But with PFAS, the body faces the challenge of carbon-fluorine bonds that are very hard to break down, and there is no payoff for these efforts, Graham F. Peaslee, PhD, professor of physics at the University of Notre Dame (Indiana), told this news organization.
“Nothing will naturally eat it because when you break the bond, it’s like eating celery,” he said. “You use more calories to eat the celery than you gain back from it.”
Interest from a U.S. senator
Dr. Peaslee was one of the authors of a 2021 article about PFAS in cosmetics that appeared in the journal Environmental Science and Technology Letters.
In the article, Dr. Peaslee and colleagues reported on their screening of 231 cosmetic products purchased in the United States and Canada using particle-induced gamma-ray emission spectroscopy. They found cases of undisclosed PFAS in cosmetic products. Foundations, mascaras, and lip products were noted as being especially problematic.
Sen. Susan Collins (R-ME) cited Dr. Peaslee’s article in a 2021 floor speech as she argued for having the FDA ban the intentional addition of PFAS to cosmetics.
“The findings of this study are particularly alarming, as many of these products are subject to direct human exposure,” Sen. Collins said. “For example, lipstick is often inadvertently ingested, and mascara is sometimes absorbed through tear ducts.”
In addition, workers at cosmetics plants may be exposed to PFAS and discarded cosmetics that have these compounds, which could potentially contaminate drinking water, Sen. Collins said. In 2021, she introduced legislation seeking a ban on PFAS that are intentionally added to cosmetics. That legislation did not advance through the Senate.
But the Senate Appropriations Committee, on which Sen. Collins is the ranking Republican, wants the FDA to keep a ban on PFAS in mind.
The Senate Agriculture Appropriations subcommittee, which oversees the FDA’s budget, raised the issue of PFAS and cosmetics in a June report. The FDA should develop a plan outlining research needed to inform “regulatory decision making, including potential development of a proposed rule to ban intentionally added PFAS substances in cosmetics,” the subcommittee said.
A version of this article first appeared on Medscape.com.
Inner lip erosions
The patient was having a flare of pemphigus vulgaris (PV), a rare and sometimes life-threatening acquired autoimmune blistering disease that affects the skin and/or mucosa. Ashkenazi Jewish patients and patients from Mediterranean and Middle Eastern countries are more likely to be affected.
In PV, acquired autoantibodies target the desmosomes that connect epithelial cells together, weakening the intercellular adhesion. It can affect skin, mucosa, or both. Patients present with fragile bullae or ulcers. The connections between the cells are often so damaged that rubbing on the skin creates a new blister called “Nikolsky sign.” In the mouth, bullae erode rapidly. Look for disease affecting the ocular conjunctiva or sclera, as well. PV can also occasionally affect the nasopharynx and esophagus, usually manifesting as hemoptysis, dysphagia, and nosebleeds with ulcer seen on endoscopy or otolaryngoscopy.
Although PV is often severe (and can warrant hospitalization when significant body surface area is involved), some patients may have few active lesions and can be managed safely as outpatients.
The diagnosis requires 2 biopsies and serum for indirect immunofluorescence. One biopsy (either by punch or shave to the upper dermis) is taken from the edge of a bulla or ulcer. Another biopsy (by punch or shave) is taken from nearby normal-looking skin or mucosa for testing the direct immunofluorescence pattern. In the mucosa, a punch biopsy may be left open or closed with absorbable sutures. A serum sample is taken for indirect immunofluorescence to differentiate pemphigus vulgaris from other forms of pemphigus.1
PV is treated by suppressing the immune system. Focal disease may be treated with super-potent topical steroids, including clobetasol 0.05% ointment. Even in the mouth, topical clobetasol 0.05% may be used off-label twice daily until control is achieved. When topical treatment is used in the mouth, advise patients to apply the clobetasol ointment to a piece of gauze and place the gauze (ointment side down) over affected areas for 20 to 30 minutes twice daily.
Patients with widespread or severe disease should be hospitalized. In severe cases, supportive wound care is provided, and treatment is aimed at immunosuppression. Systemic options include high-dose prednisone 0.5 to 1 mg/kg daily until clear, a steroid-sparing immunosuppressant such as mycophenolate mofetil up to 1000 mg bid, or rituximab in 1 of several regimens.
Three years prior to this patient’s visit, she had been successfully treated for PV with a course of rituximab. To treat the current flare, she was started on prednisone 60 mg/d. In addition, the plan was for her to complete 2 infusions of 1000 mg rituximab 2 weeks apart.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.
1. Didona, D, Schmidt, MF, Maglie, R, et al. Pemphigus and pemphigoids: clinical presentation, diagnosis and therapy. J Dtsch Dermatol Ges. 2023;1-20. doi: 10.1111/ddg.15174

The patient was having a flare of pemphigus vulgaris (PV), a rare and sometimes life-threatening acquired autoimmune blistering disease that affects the skin and/or mucosa. Ashkenazi Jewish patients and patients from Mediterranean and Middle Eastern countries are more likely to be affected.
In PV, acquired autoantibodies target the desmosomes that connect epithelial cells together, weakening the intercellular adhesion. It can affect skin, mucosa, or both. Patients present with fragile bullae or ulcers. The connections between the cells are often so damaged that rubbing on the skin creates a new blister called “Nikolsky sign.” In the mouth, bullae erode rapidly. Look for disease affecting the ocular conjunctiva or sclera, as well. PV can also occasionally affect the nasopharynx and esophagus, usually manifesting as hemoptysis, dysphagia, and nosebleeds with ulcer seen on endoscopy or otolaryngoscopy.
Although PV is often severe (and can warrant hospitalization when significant body surface area is involved), some patients may have few active lesions and can be managed safely as outpatients.
The diagnosis requires 2 biopsies and serum for indirect immunofluorescence. One biopsy (either by punch or shave to the upper dermis) is taken from the edge of a bulla or ulcer. Another biopsy (by punch or shave) is taken from nearby normal-looking skin or mucosa for testing the direct immunofluorescence pattern. In the mucosa, a punch biopsy may be left open or closed with absorbable sutures. A serum sample is taken for indirect immunofluorescence to differentiate pemphigus vulgaris from other forms of pemphigus.1
PV is treated by suppressing the immune system. Focal disease may be treated with super-potent topical steroids, including clobetasol 0.05% ointment. Even in the mouth, topical clobetasol 0.05% may be used off-label twice daily until control is achieved. When topical treatment is used in the mouth, advise patients to apply the clobetasol ointment to a piece of gauze and place the gauze (ointment side down) over affected areas for 20 to 30 minutes twice daily.
Patients with widespread or severe disease should be hospitalized. In severe cases, supportive wound care is provided, and treatment is aimed at immunosuppression. Systemic options include high-dose prednisone 0.5 to 1 mg/kg daily until clear, a steroid-sparing immunosuppressant such as mycophenolate mofetil up to 1000 mg bid, or rituximab in 1 of several regimens.
Three years prior to this patient’s visit, she had been successfully treated for PV with a course of rituximab. To treat the current flare, she was started on prednisone 60 mg/d. In addition, the plan was for her to complete 2 infusions of 1000 mg rituximab 2 weeks apart.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.

The patient was having a flare of pemphigus vulgaris (PV), a rare and sometimes life-threatening acquired autoimmune blistering disease that affects the skin and/or mucosa. Ashkenazi Jewish patients and patients from Mediterranean and Middle Eastern countries are more likely to be affected.
In PV, acquired autoantibodies target the desmosomes that connect epithelial cells together, weakening the intercellular adhesion. It can affect skin, mucosa, or both. Patients present with fragile bullae or ulcers. The connections between the cells are often so damaged that rubbing on the skin creates a new blister called “Nikolsky sign.” In the mouth, bullae erode rapidly. Look for disease affecting the ocular conjunctiva or sclera, as well. PV can also occasionally affect the nasopharynx and esophagus, usually manifesting as hemoptysis, dysphagia, and nosebleeds with ulcer seen on endoscopy or otolaryngoscopy.
Although PV is often severe (and can warrant hospitalization when significant body surface area is involved), some patients may have few active lesions and can be managed safely as outpatients.
The diagnosis requires 2 biopsies and serum for indirect immunofluorescence. One biopsy (either by punch or shave to the upper dermis) is taken from the edge of a bulla or ulcer. Another biopsy (by punch or shave) is taken from nearby normal-looking skin or mucosa for testing the direct immunofluorescence pattern. In the mucosa, a punch biopsy may be left open or closed with absorbable sutures. A serum sample is taken for indirect immunofluorescence to differentiate pemphigus vulgaris from other forms of pemphigus.1
PV is treated by suppressing the immune system. Focal disease may be treated with super-potent topical steroids, including clobetasol 0.05% ointment. Even in the mouth, topical clobetasol 0.05% may be used off-label twice daily until control is achieved. When topical treatment is used in the mouth, advise patients to apply the clobetasol ointment to a piece of gauze and place the gauze (ointment side down) over affected areas for 20 to 30 minutes twice daily.
Patients with widespread or severe disease should be hospitalized. In severe cases, supportive wound care is provided, and treatment is aimed at immunosuppression. Systemic options include high-dose prednisone 0.5 to 1 mg/kg daily until clear, a steroid-sparing immunosuppressant such as mycophenolate mofetil up to 1000 mg bid, or rituximab in 1 of several regimens.
Three years prior to this patient’s visit, she had been successfully treated for PV with a course of rituximab. To treat the current flare, she was started on prednisone 60 mg/d. In addition, the plan was for her to complete 2 infusions of 1000 mg rituximab 2 weeks apart.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.
1. Didona, D, Schmidt, MF, Maglie, R, et al. Pemphigus and pemphigoids: clinical presentation, diagnosis and therapy. J Dtsch Dermatol Ges. 2023;1-20. doi: 10.1111/ddg.15174
1. Didona, D, Schmidt, MF, Maglie, R, et al. Pemphigus and pemphigoids: clinical presentation, diagnosis and therapy. J Dtsch Dermatol Ges. 2023;1-20. doi: 10.1111/ddg.15174
Atopic dermatitis may be a risk factor for GBS colonization in pregnancy
suggest.
“The rate of GBS colonization among pregnant females with a history of AD has not been previously reported, but AD could be a risk factor for maternal carriage of GBS,” corresponding author David J. Margolis, MD, PhD, of the department of dermatology at the University of Pennsylvania, Philadelphia, and colleagues wrote in the study, which was published as a letter to the editor online in the Journal of Investigative Dermatology. “GBS reporting in a large administrative database represents a unique opportunity to conduct a population-based evaluation of GBS carriage with AD. Understanding this association could expand our understanding of microbial changes associated with AD,” they noted.
To determine if an association between GBS and AD in pregnant women exists, the researchers performed a cross-sectional study using a random sample from an Optum administrative database of pregnant women who had vaginal deliveries between May of 2007 and September 2021. The primary outcome of interest was the presence of GBS based on American College of Obstetricians and Gynecologists–recommended codes for GBS during 36 0/7 to 37 6/7 weeks of pregnancy. They used descriptive statistics to summarize categorical and continuous variables as proportions and means, and logistic regression to examine the association between AD and GBS status.
The cohort included 566,467 pregnant women with an average age of 38.8 years. Of these, 2.9% had a diagnosis of AD or a history of AD, and 24.9% had diagnoses of asthma, seasonal allergies, or both. Women with AD had an increased odds ratio of asthma (OR, 2.55), seasonal allergies (OR, 3.39), or both (OR, 5.35), compared with those without AD.
GBS was reported in 20.6% of the cohort. The median time of follow-up for those with and without GBS was 494 days and 468 days, respectively (P = .134). Among the women with AD, 24.1% had GBS, compared with 20.51% of the women without AD (P <.0001), which translated into an OR of 1.23 (95% confidence interval, 1.18-1.27).
Among the women with GBS, the OR of asthma was 1.08 (95% CI, 1.06-1.10) and was 1.07 (95% CI, 1.05-1.09) among those with seasonal allergies. When adjusted for potential confounders, these findings did not change substantively.
“It is not apparent why pregnant females with AD are more likely to specifically carry GBS,” the authors wrote. “However, several studies have shown that individuals with AD are more likely to carry [Staphylococcus] aureus and that individuals with AD might be deficient in host defenses against S. aureus and other pathogens,” they added.
“Individuals with AD frequently receive antibiotics as part of their AD treatment and this might alter their resident microbiome. Carriage rates may be enhanced by the inhibition of an important barrier protein called filaggrin (FLG) and FLG loss of function genetic variation is known to decrease barrier proteins thought to inhibit the colonization of S. aureus and other pathogens,” the researchers wrote.
They acknowledged certain limitations of their study, including its reliance on an administrative database that does not contain information on past disease.
Asked to comment on the results, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the study, characterized AD as “the poster child for cutaneous dysbiosis – an altered petri dish, so to speak, [that] facilitates survival of the few, leading to decreased microbial diversity that can both enable potential pathogen invasion and immune dysregulation.”
Though it’s not surprising that pregnant AD patients have dysbiosis, the focus on GBS, “which can be a bad actor in the perinatal period, is an interesting connection,” he said. “Will this change practices? Pregnant women should be screened for GBS regardless, but maybe more attention or counseling can be offered to AD patients about the importance of screening. Would decolonization regimens be employed early in pregnancy? This study can’t answer that but certainly raises good questions.”
Dr. Margolis disclosed that he is or recently has been a consultant for Pfizer, Leo, and Sanofi with respect to studies of atopic dermatitis and served on an advisory board for the National Eczema Association. Another author disclosed receiving grants from companies related to work with AD; other authors had no disclosures. Dr. Friedman reported having no relevant disclosures.
suggest.
“The rate of GBS colonization among pregnant females with a history of AD has not been previously reported, but AD could be a risk factor for maternal carriage of GBS,” corresponding author David J. Margolis, MD, PhD, of the department of dermatology at the University of Pennsylvania, Philadelphia, and colleagues wrote in the study, which was published as a letter to the editor online in the Journal of Investigative Dermatology. “GBS reporting in a large administrative database represents a unique opportunity to conduct a population-based evaluation of GBS carriage with AD. Understanding this association could expand our understanding of microbial changes associated with AD,” they noted.
To determine if an association between GBS and AD in pregnant women exists, the researchers performed a cross-sectional study using a random sample from an Optum administrative database of pregnant women who had vaginal deliveries between May of 2007 and September 2021. The primary outcome of interest was the presence of GBS based on American College of Obstetricians and Gynecologists–recommended codes for GBS during 36 0/7 to 37 6/7 weeks of pregnancy. They used descriptive statistics to summarize categorical and continuous variables as proportions and means, and logistic regression to examine the association between AD and GBS status.
The cohort included 566,467 pregnant women with an average age of 38.8 years. Of these, 2.9% had a diagnosis of AD or a history of AD, and 24.9% had diagnoses of asthma, seasonal allergies, or both. Women with AD had an increased odds ratio of asthma (OR, 2.55), seasonal allergies (OR, 3.39), or both (OR, 5.35), compared with those without AD.
GBS was reported in 20.6% of the cohort. The median time of follow-up for those with and without GBS was 494 days and 468 days, respectively (P = .134). Among the women with AD, 24.1% had GBS, compared with 20.51% of the women without AD (P <.0001), which translated into an OR of 1.23 (95% confidence interval, 1.18-1.27).
Among the women with GBS, the OR of asthma was 1.08 (95% CI, 1.06-1.10) and was 1.07 (95% CI, 1.05-1.09) among those with seasonal allergies. When adjusted for potential confounders, these findings did not change substantively.
“It is not apparent why pregnant females with AD are more likely to specifically carry GBS,” the authors wrote. “However, several studies have shown that individuals with AD are more likely to carry [Staphylococcus] aureus and that individuals with AD might be deficient in host defenses against S. aureus and other pathogens,” they added.
“Individuals with AD frequently receive antibiotics as part of their AD treatment and this might alter their resident microbiome. Carriage rates may be enhanced by the inhibition of an important barrier protein called filaggrin (FLG) and FLG loss of function genetic variation is known to decrease barrier proteins thought to inhibit the colonization of S. aureus and other pathogens,” the researchers wrote.
They acknowledged certain limitations of their study, including its reliance on an administrative database that does not contain information on past disease.
Asked to comment on the results, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the study, characterized AD as “the poster child for cutaneous dysbiosis – an altered petri dish, so to speak, [that] facilitates survival of the few, leading to decreased microbial diversity that can both enable potential pathogen invasion and immune dysregulation.”
Though it’s not surprising that pregnant AD patients have dysbiosis, the focus on GBS, “which can be a bad actor in the perinatal period, is an interesting connection,” he said. “Will this change practices? Pregnant women should be screened for GBS regardless, but maybe more attention or counseling can be offered to AD patients about the importance of screening. Would decolonization regimens be employed early in pregnancy? This study can’t answer that but certainly raises good questions.”
Dr. Margolis disclosed that he is or recently has been a consultant for Pfizer, Leo, and Sanofi with respect to studies of atopic dermatitis and served on an advisory board for the National Eczema Association. Another author disclosed receiving grants from companies related to work with AD; other authors had no disclosures. Dr. Friedman reported having no relevant disclosures.
suggest.
“The rate of GBS colonization among pregnant females with a history of AD has not been previously reported, but AD could be a risk factor for maternal carriage of GBS,” corresponding author David J. Margolis, MD, PhD, of the department of dermatology at the University of Pennsylvania, Philadelphia, and colleagues wrote in the study, which was published as a letter to the editor online in the Journal of Investigative Dermatology. “GBS reporting in a large administrative database represents a unique opportunity to conduct a population-based evaluation of GBS carriage with AD. Understanding this association could expand our understanding of microbial changes associated with AD,” they noted.
To determine if an association between GBS and AD in pregnant women exists, the researchers performed a cross-sectional study using a random sample from an Optum administrative database of pregnant women who had vaginal deliveries between May of 2007 and September 2021. The primary outcome of interest was the presence of GBS based on American College of Obstetricians and Gynecologists–recommended codes for GBS during 36 0/7 to 37 6/7 weeks of pregnancy. They used descriptive statistics to summarize categorical and continuous variables as proportions and means, and logistic regression to examine the association between AD and GBS status.
The cohort included 566,467 pregnant women with an average age of 38.8 years. Of these, 2.9% had a diagnosis of AD or a history of AD, and 24.9% had diagnoses of asthma, seasonal allergies, or both. Women with AD had an increased odds ratio of asthma (OR, 2.55), seasonal allergies (OR, 3.39), or both (OR, 5.35), compared with those without AD.
GBS was reported in 20.6% of the cohort. The median time of follow-up for those with and without GBS was 494 days and 468 days, respectively (P = .134). Among the women with AD, 24.1% had GBS, compared with 20.51% of the women without AD (P <.0001), which translated into an OR of 1.23 (95% confidence interval, 1.18-1.27).
Among the women with GBS, the OR of asthma was 1.08 (95% CI, 1.06-1.10) and was 1.07 (95% CI, 1.05-1.09) among those with seasonal allergies. When adjusted for potential confounders, these findings did not change substantively.
“It is not apparent why pregnant females with AD are more likely to specifically carry GBS,” the authors wrote. “However, several studies have shown that individuals with AD are more likely to carry [Staphylococcus] aureus and that individuals with AD might be deficient in host defenses against S. aureus and other pathogens,” they added.
“Individuals with AD frequently receive antibiotics as part of their AD treatment and this might alter their resident microbiome. Carriage rates may be enhanced by the inhibition of an important barrier protein called filaggrin (FLG) and FLG loss of function genetic variation is known to decrease barrier proteins thought to inhibit the colonization of S. aureus and other pathogens,” the researchers wrote.
They acknowledged certain limitations of their study, including its reliance on an administrative database that does not contain information on past disease.
Asked to comment on the results, Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was not involved with the study, characterized AD as “the poster child for cutaneous dysbiosis – an altered petri dish, so to speak, [that] facilitates survival of the few, leading to decreased microbial diversity that can both enable potential pathogen invasion and immune dysregulation.”
Though it’s not surprising that pregnant AD patients have dysbiosis, the focus on GBS, “which can be a bad actor in the perinatal period, is an interesting connection,” he said. “Will this change practices? Pregnant women should be screened for GBS regardless, but maybe more attention or counseling can be offered to AD patients about the importance of screening. Would decolonization regimens be employed early in pregnancy? This study can’t answer that but certainly raises good questions.”
Dr. Margolis disclosed that he is or recently has been a consultant for Pfizer, Leo, and Sanofi with respect to studies of atopic dermatitis and served on an advisory board for the National Eczema Association. Another author disclosed receiving grants from companies related to work with AD; other authors had no disclosures. Dr. Friedman reported having no relevant disclosures.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Mohs found to confer survival benefit in localized Merkel cell carcinoma
results from a national retrospective cohort study suggest.
The study found that, in patients with pathologically confirmed, localized T1/T2 MCC, “treatment with MMS was associated with an approximately 40% reduction in hazard of death compared with WLE,” reported John A. Carucci, MD, PhD, and colleagues in the department of dermatology at NYU Langone Health, New York. The results provide “preliminary data suggesting that treatment of localized, early-stage MCC with MMS may result in the most optimal patient survival outcomes for this aggressive form of skin cancer,” they added. The study was published online in JAMA Dermatology.
“Although data for keratinocytic nonmelanoma skin cancers have been definitive in demonstrating the advantage of peripheral and deep en face margin assessment over conventional WLE or NME [narrow-margin excision], the data for MCC, likely because of the disease’s rarity and limitations of available data sets, have been mixed,” they wrote.
Results from national studies published in the Journal of the National Cancer Institute and the Journal of the American Academy of Dermatology found no difference in survival among patients with localized MCC treated with WLE versus MMS. “However, these studies did not have confirmed pathologic node status, a substantial limitation considering that clinically node-negative cases of localized MCC have sentinel lymph node positivity rates ranging from 25% to 40%,” the authors noted.
To evaluate the association of the surgical excision modality and patient survival for pathologically confirmed localized T1/T2 MCC, Dr. Carucci and coauthors examined a cohort of 2,313 patients from the National Cancer Database with T1/T2 MCC diagnosed between Jan. 1, 2004, and Dec. 31, 2018, with pathologically confirmed, negative regional lymph nodes and treated with surgery. Their mean age was 71 years and 57.9% were male. Of the 2,313 patients, 1,452 underwent WLE, 104 underwent MMS, and 757 underwent NME.
The unadjusted analysis revealed that, compared with WLE, excision with MMS had the best unadjusted mean survival rates: 87.4% versus 86.1%, respectively, at 3 years, 84.5% versus 76.9% at 5 years, and 81.8% versus 60.9% at 10 years. Patients treated with NME had similar mean survival rates as those treated with WLE: 84.8% at 3 years, 78.3% at 5 years, and 60.8% at 10 years.
Multivariable survival analysis demonstrated that treatment with MMS was associated with significantly improved survival, compared with WLE (hazard ratio, 0.59; 95% CI, 0.36-0.97; P = .04).
“These data suggest that MMS may provide a survival benefit in the treatment of localized MCC, although further prospective work studying this issue is required,” the authors concluded. “Future directions may also focus on elucidating the benefit of adjuvant radiotherapy in localized cases treated with MMS.”
They acknowledged certain limitations of the study, including the fewer numbers of patients receiving MMS surgery, lack of randomization, and potential for selection bias.
In an interview, Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, who was asked to comment on the study, said that the field of MCC “has undergone rapid and robust transformation over the past 20 years. These changes encompass advancements in diagnosing the condition, identifying linked viruses, and developing systemic treatments.”
The study findings “imply that comprehensive assessment of histologic margins might offer advantages beyond minimizing scars, minimizing functional impact, and reducing the likelihood of local recurrence,” he said.
“It’s beyond doubt,” he added, that the study “furnishes us with yet another set of real-world insights that will undoubtedly influence patient outcomes. These insights serve to bring clarity to the ways in which we can deliver precisely targeted surgical treatment with durable outcomes for localized MCC.”
Patricia M. Richey, MD, director of Mohs surgery at Boston University, who was also asked to comment on the study, added that, because of the nature of the National Cancer Database, “the authors of this study were unfortunately unable to report disease-specific survival or immunosuppression status. That being said, the preliminary data presented are convincing and should result in us further exploring this topic, as well as readdressing and questioning related issues such as whether or not adjuvant radiotherapy is truly beneficial in cases with histologic clearance via Mohs.”
Dr. Carucci reported receiving grant funding from Regeneron for investigator-initiated basic research. No other author disclosures were reported. Neither Dr. Blalock nor Dr. Richey had relevant disclosures.
results from a national retrospective cohort study suggest.
The study found that, in patients with pathologically confirmed, localized T1/T2 MCC, “treatment with MMS was associated with an approximately 40% reduction in hazard of death compared with WLE,” reported John A. Carucci, MD, PhD, and colleagues in the department of dermatology at NYU Langone Health, New York. The results provide “preliminary data suggesting that treatment of localized, early-stage MCC with MMS may result in the most optimal patient survival outcomes for this aggressive form of skin cancer,” they added. The study was published online in JAMA Dermatology.
“Although data for keratinocytic nonmelanoma skin cancers have been definitive in demonstrating the advantage of peripheral and deep en face margin assessment over conventional WLE or NME [narrow-margin excision], the data for MCC, likely because of the disease’s rarity and limitations of available data sets, have been mixed,” they wrote.
Results from national studies published in the Journal of the National Cancer Institute and the Journal of the American Academy of Dermatology found no difference in survival among patients with localized MCC treated with WLE versus MMS. “However, these studies did not have confirmed pathologic node status, a substantial limitation considering that clinically node-negative cases of localized MCC have sentinel lymph node positivity rates ranging from 25% to 40%,” the authors noted.
To evaluate the association of the surgical excision modality and patient survival for pathologically confirmed localized T1/T2 MCC, Dr. Carucci and coauthors examined a cohort of 2,313 patients from the National Cancer Database with T1/T2 MCC diagnosed between Jan. 1, 2004, and Dec. 31, 2018, with pathologically confirmed, negative regional lymph nodes and treated with surgery. Their mean age was 71 years and 57.9% were male. Of the 2,313 patients, 1,452 underwent WLE, 104 underwent MMS, and 757 underwent NME.
The unadjusted analysis revealed that, compared with WLE, excision with MMS had the best unadjusted mean survival rates: 87.4% versus 86.1%, respectively, at 3 years, 84.5% versus 76.9% at 5 years, and 81.8% versus 60.9% at 10 years. Patients treated with NME had similar mean survival rates as those treated with WLE: 84.8% at 3 years, 78.3% at 5 years, and 60.8% at 10 years.
Multivariable survival analysis demonstrated that treatment with MMS was associated with significantly improved survival, compared with WLE (hazard ratio, 0.59; 95% CI, 0.36-0.97; P = .04).
“These data suggest that MMS may provide a survival benefit in the treatment of localized MCC, although further prospective work studying this issue is required,” the authors concluded. “Future directions may also focus on elucidating the benefit of adjuvant radiotherapy in localized cases treated with MMS.”
They acknowledged certain limitations of the study, including the fewer numbers of patients receiving MMS surgery, lack of randomization, and potential for selection bias.
In an interview, Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, who was asked to comment on the study, said that the field of MCC “has undergone rapid and robust transformation over the past 20 years. These changes encompass advancements in diagnosing the condition, identifying linked viruses, and developing systemic treatments.”
The study findings “imply that comprehensive assessment of histologic margins might offer advantages beyond minimizing scars, minimizing functional impact, and reducing the likelihood of local recurrence,” he said.
“It’s beyond doubt,” he added, that the study “furnishes us with yet another set of real-world insights that will undoubtedly influence patient outcomes. These insights serve to bring clarity to the ways in which we can deliver precisely targeted surgical treatment with durable outcomes for localized MCC.”
Patricia M. Richey, MD, director of Mohs surgery at Boston University, who was also asked to comment on the study, added that, because of the nature of the National Cancer Database, “the authors of this study were unfortunately unable to report disease-specific survival or immunosuppression status. That being said, the preliminary data presented are convincing and should result in us further exploring this topic, as well as readdressing and questioning related issues such as whether or not adjuvant radiotherapy is truly beneficial in cases with histologic clearance via Mohs.”
Dr. Carucci reported receiving grant funding from Regeneron for investigator-initiated basic research. No other author disclosures were reported. Neither Dr. Blalock nor Dr. Richey had relevant disclosures.
results from a national retrospective cohort study suggest.
The study found that, in patients with pathologically confirmed, localized T1/T2 MCC, “treatment with MMS was associated with an approximately 40% reduction in hazard of death compared with WLE,” reported John A. Carucci, MD, PhD, and colleagues in the department of dermatology at NYU Langone Health, New York. The results provide “preliminary data suggesting that treatment of localized, early-stage MCC with MMS may result in the most optimal patient survival outcomes for this aggressive form of skin cancer,” they added. The study was published online in JAMA Dermatology.
“Although data for keratinocytic nonmelanoma skin cancers have been definitive in demonstrating the advantage of peripheral and deep en face margin assessment over conventional WLE or NME [narrow-margin excision], the data for MCC, likely because of the disease’s rarity and limitations of available data sets, have been mixed,” they wrote.
Results from national studies published in the Journal of the National Cancer Institute and the Journal of the American Academy of Dermatology found no difference in survival among patients with localized MCC treated with WLE versus MMS. “However, these studies did not have confirmed pathologic node status, a substantial limitation considering that clinically node-negative cases of localized MCC have sentinel lymph node positivity rates ranging from 25% to 40%,” the authors noted.
To evaluate the association of the surgical excision modality and patient survival for pathologically confirmed localized T1/T2 MCC, Dr. Carucci and coauthors examined a cohort of 2,313 patients from the National Cancer Database with T1/T2 MCC diagnosed between Jan. 1, 2004, and Dec. 31, 2018, with pathologically confirmed, negative regional lymph nodes and treated with surgery. Their mean age was 71 years and 57.9% were male. Of the 2,313 patients, 1,452 underwent WLE, 104 underwent MMS, and 757 underwent NME.
The unadjusted analysis revealed that, compared with WLE, excision with MMS had the best unadjusted mean survival rates: 87.4% versus 86.1%, respectively, at 3 years, 84.5% versus 76.9% at 5 years, and 81.8% versus 60.9% at 10 years. Patients treated with NME had similar mean survival rates as those treated with WLE: 84.8% at 3 years, 78.3% at 5 years, and 60.8% at 10 years.
Multivariable survival analysis demonstrated that treatment with MMS was associated with significantly improved survival, compared with WLE (hazard ratio, 0.59; 95% CI, 0.36-0.97; P = .04).
“These data suggest that MMS may provide a survival benefit in the treatment of localized MCC, although further prospective work studying this issue is required,” the authors concluded. “Future directions may also focus on elucidating the benefit of adjuvant radiotherapy in localized cases treated with MMS.”
They acknowledged certain limitations of the study, including the fewer numbers of patients receiving MMS surgery, lack of randomization, and potential for selection bias.
In an interview, Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, who was asked to comment on the study, said that the field of MCC “has undergone rapid and robust transformation over the past 20 years. These changes encompass advancements in diagnosing the condition, identifying linked viruses, and developing systemic treatments.”
The study findings “imply that comprehensive assessment of histologic margins might offer advantages beyond minimizing scars, minimizing functional impact, and reducing the likelihood of local recurrence,” he said.
“It’s beyond doubt,” he added, that the study “furnishes us with yet another set of real-world insights that will undoubtedly influence patient outcomes. These insights serve to bring clarity to the ways in which we can deliver precisely targeted surgical treatment with durable outcomes for localized MCC.”
Patricia M. Richey, MD, director of Mohs surgery at Boston University, who was also asked to comment on the study, added that, because of the nature of the National Cancer Database, “the authors of this study were unfortunately unable to report disease-specific survival or immunosuppression status. That being said, the preliminary data presented are convincing and should result in us further exploring this topic, as well as readdressing and questioning related issues such as whether or not adjuvant radiotherapy is truly beneficial in cases with histologic clearance via Mohs.”
Dr. Carucci reported receiving grant funding from Regeneron for investigator-initiated basic research. No other author disclosures were reported. Neither Dr. Blalock nor Dr. Richey had relevant disclosures.
FROM JAMA DERMATOLOGY
Consider housing insecurity, other issues when managing challenging skin diseases in children, expert says
ASHEVILLE, N.C. – , according to a pediatric dermatologist who addressed the annual meeting of the Society for Pediatric Dermatology.
As a general principle for treating chronic skin conditions in children who are not doing well, it is reasonable to draw out information about a patient’s access to adequate housing, nutrition, and other basic needs, George Hightower, MD, PhD, of the division of pediatric and adolescent dermatology, University of California, San Diego, said at the meeting.
“We need conversations about where patients play, learn, and rest their heads at night,” said Dr. Hightower, who conducts research in this area. Fundamental components of well-being, such as stable housing and secure access to nutrition “are inseparable” from a child’s health, he noted.
“What are the stakes?” he asked. For many children, these factors might mean the difference between effective and poor control of the diseases for which the patient is seeking care.
To illustrate the point, Dr. Hightower used hidradenitis suppurativa (HS), a disease that appears to be on the rise among adolescents, as an example of why patient circumstances matter and should be considered. A complex disorder that is more prevalent in resource-poor communities, HS is difficult to control, often requiring extended periods of treatment with medications that can involve complex dosing or regular infusions.
“There is a need for medical providers to help the patient plan for this chronic illness,” said Dr. Hightower, referring to the importance of close follow-up. In adolescents, HS can be sufficiently disruptive from both the physical and psychological perspective that poor control can “derail future aspirations” by complicating educational endeavors and social interactions.
Dr. Hightower acknowledged that simply documenting housing insecurity or other issues does not solve these problems, but he does believe that developing a sensitivity to these obstacles to health care is a first step. It is a process that should permeate into medical training, health care research, and strategies to improve outcomes.
“The connections between fair housing and clinical practice may appear tenuous and inconsequential to the care provided by medical specialists,” Dr. Hightower said, but he emphasized that there are clear consequences when these factors contribute to inadequate control of such diseases as HS. As a source of missed appointments and disjointed care, an unstable home life can be an important barrier to disease control – and because of scarring nodules, fistulae, pain, school absences, and social isolation, complications can be dire.
Solutions to insecure housing are not typically available to an individual clinician, but the awareness that this can be a factor can help both physicians and patients begin to think about the role this plays in impairing recovery and what solutions might be found to modify the impact. Awareness not just among individual clinicians but a broader consortium of those working to improve health care outcomes is needed to “challenge the way we are doing medicine,” he said.
While conversations about the social determinants of health, including access to resources within patients’ neighborhoods, schools, and environment, can demonstrate concern about how to address obstacles, it can also be part of a reorientation to think beyond treatment for the underlying pathology alone. Eliciting trust and emphasizing the importance of environmental barriers to adequate care can be positive steps on the path to solutions.
Participatory action research
Relevant to this orientation, Dr. Hightower spoke about participatory action research (PAR), which provides a framework for patients to participate in the planning of clinical studies to effect change, not just serve as subjects in these studies.
The assumption of PAR is that “all people have valuable knowledge about their lives and experiences,” Dr. Hightower said. From this assumption, individuals who have been historically marginalized by race, income, or other factors can help define the problems from the patient’s perspective and, from there, create studies to seek solutions.
PAR is consistent with a patient-centered approach to medical care, which Dr. Hightower called “the future of medicine.” It involves a big-picture approach to look beyond disease pathology and symptoms to factors that might be creating susceptibility to disease and undermining health care.
Organized medicine alone cannot solve the cause of social inequities leading to disparate risks for disease and risks of inadequate health care, but Dr. Hightower argued that these inequities should not be ignored. He believes medical trainees should learn how to elicit information about the barriers to adequate health care and be aware of solutions, such as fair housing policies.
While he believes that PAR is an example of a pathway to problem solving, he suggested that a comprehensive approach requires an effective method of communication between providers and patients that would lead to a collaborative and mutually reinforcing approach.
“How do we ensure that individuals from communities most impacted by health disparities are treated fairly and empowered to address these disparities?” Dr. Hightower asked. He said that this is the direction of his own research and the issues that inhibit adequate treatment of many dermatologic diseases, as well as other types of disease, in childhood.
Craig Burkhart, MD, director of a private pediatric and adolescent dermatology practice in Cary, N.C., said that Dr. Hightower’s message is relevant. The value of considering and addressing the psychological well-being of patients of any age is not a new concept, but he acknowledged that he, for one, has not routinely inquired about obstacles to follow-up care if there is a signal that this might be an issue.
“As dermatologists, we focus on the acute complaints. We want to make the patient better,” said Dr. Burkhart, who moderated the session in which Dr. Hightower spoke. He agreed with Dr. Hightower that environmental factors make a difference on the road to recovery for a patient, and his presentation was a good reminder, he said, to consider the patient’s circumstances when response to treatment is inadequate, particularly in chronic diseases like HS, for which comprehensive care and close follow-up are needed.
Dr. Hightower and Dr. Burkhart report no potential conflicts of interest.
ASHEVILLE, N.C. – , according to a pediatric dermatologist who addressed the annual meeting of the Society for Pediatric Dermatology.
As a general principle for treating chronic skin conditions in children who are not doing well, it is reasonable to draw out information about a patient’s access to adequate housing, nutrition, and other basic needs, George Hightower, MD, PhD, of the division of pediatric and adolescent dermatology, University of California, San Diego, said at the meeting.
“We need conversations about where patients play, learn, and rest their heads at night,” said Dr. Hightower, who conducts research in this area. Fundamental components of well-being, such as stable housing and secure access to nutrition “are inseparable” from a child’s health, he noted.
“What are the stakes?” he asked. For many children, these factors might mean the difference between effective and poor control of the diseases for which the patient is seeking care.
To illustrate the point, Dr. Hightower used hidradenitis suppurativa (HS), a disease that appears to be on the rise among adolescents, as an example of why patient circumstances matter and should be considered. A complex disorder that is more prevalent in resource-poor communities, HS is difficult to control, often requiring extended periods of treatment with medications that can involve complex dosing or regular infusions.
“There is a need for medical providers to help the patient plan for this chronic illness,” said Dr. Hightower, referring to the importance of close follow-up. In adolescents, HS can be sufficiently disruptive from both the physical and psychological perspective that poor control can “derail future aspirations” by complicating educational endeavors and social interactions.
Dr. Hightower acknowledged that simply documenting housing insecurity or other issues does not solve these problems, but he does believe that developing a sensitivity to these obstacles to health care is a first step. It is a process that should permeate into medical training, health care research, and strategies to improve outcomes.
“The connections between fair housing and clinical practice may appear tenuous and inconsequential to the care provided by medical specialists,” Dr. Hightower said, but he emphasized that there are clear consequences when these factors contribute to inadequate control of such diseases as HS. As a source of missed appointments and disjointed care, an unstable home life can be an important barrier to disease control – and because of scarring nodules, fistulae, pain, school absences, and social isolation, complications can be dire.
Solutions to insecure housing are not typically available to an individual clinician, but the awareness that this can be a factor can help both physicians and patients begin to think about the role this plays in impairing recovery and what solutions might be found to modify the impact. Awareness not just among individual clinicians but a broader consortium of those working to improve health care outcomes is needed to “challenge the way we are doing medicine,” he said.
While conversations about the social determinants of health, including access to resources within patients’ neighborhoods, schools, and environment, can demonstrate concern about how to address obstacles, it can also be part of a reorientation to think beyond treatment for the underlying pathology alone. Eliciting trust and emphasizing the importance of environmental barriers to adequate care can be positive steps on the path to solutions.
Participatory action research
Relevant to this orientation, Dr. Hightower spoke about participatory action research (PAR), which provides a framework for patients to participate in the planning of clinical studies to effect change, not just serve as subjects in these studies.
The assumption of PAR is that “all people have valuable knowledge about their lives and experiences,” Dr. Hightower said. From this assumption, individuals who have been historically marginalized by race, income, or other factors can help define the problems from the patient’s perspective and, from there, create studies to seek solutions.
PAR is consistent with a patient-centered approach to medical care, which Dr. Hightower called “the future of medicine.” It involves a big-picture approach to look beyond disease pathology and symptoms to factors that might be creating susceptibility to disease and undermining health care.
Organized medicine alone cannot solve the cause of social inequities leading to disparate risks for disease and risks of inadequate health care, but Dr. Hightower argued that these inequities should not be ignored. He believes medical trainees should learn how to elicit information about the barriers to adequate health care and be aware of solutions, such as fair housing policies.
While he believes that PAR is an example of a pathway to problem solving, he suggested that a comprehensive approach requires an effective method of communication between providers and patients that would lead to a collaborative and mutually reinforcing approach.
“How do we ensure that individuals from communities most impacted by health disparities are treated fairly and empowered to address these disparities?” Dr. Hightower asked. He said that this is the direction of his own research and the issues that inhibit adequate treatment of many dermatologic diseases, as well as other types of disease, in childhood.
Craig Burkhart, MD, director of a private pediatric and adolescent dermatology practice in Cary, N.C., said that Dr. Hightower’s message is relevant. The value of considering and addressing the psychological well-being of patients of any age is not a new concept, but he acknowledged that he, for one, has not routinely inquired about obstacles to follow-up care if there is a signal that this might be an issue.
“As dermatologists, we focus on the acute complaints. We want to make the patient better,” said Dr. Burkhart, who moderated the session in which Dr. Hightower spoke. He agreed with Dr. Hightower that environmental factors make a difference on the road to recovery for a patient, and his presentation was a good reminder, he said, to consider the patient’s circumstances when response to treatment is inadequate, particularly in chronic diseases like HS, for which comprehensive care and close follow-up are needed.
Dr. Hightower and Dr. Burkhart report no potential conflicts of interest.
ASHEVILLE, N.C. – , according to a pediatric dermatologist who addressed the annual meeting of the Society for Pediatric Dermatology.
As a general principle for treating chronic skin conditions in children who are not doing well, it is reasonable to draw out information about a patient’s access to adequate housing, nutrition, and other basic needs, George Hightower, MD, PhD, of the division of pediatric and adolescent dermatology, University of California, San Diego, said at the meeting.
“We need conversations about where patients play, learn, and rest their heads at night,” said Dr. Hightower, who conducts research in this area. Fundamental components of well-being, such as stable housing and secure access to nutrition “are inseparable” from a child’s health, he noted.
“What are the stakes?” he asked. For many children, these factors might mean the difference between effective and poor control of the diseases for which the patient is seeking care.
To illustrate the point, Dr. Hightower used hidradenitis suppurativa (HS), a disease that appears to be on the rise among adolescents, as an example of why patient circumstances matter and should be considered. A complex disorder that is more prevalent in resource-poor communities, HS is difficult to control, often requiring extended periods of treatment with medications that can involve complex dosing or regular infusions.
“There is a need for medical providers to help the patient plan for this chronic illness,” said Dr. Hightower, referring to the importance of close follow-up. In adolescents, HS can be sufficiently disruptive from both the physical and psychological perspective that poor control can “derail future aspirations” by complicating educational endeavors and social interactions.
Dr. Hightower acknowledged that simply documenting housing insecurity or other issues does not solve these problems, but he does believe that developing a sensitivity to these obstacles to health care is a first step. It is a process that should permeate into medical training, health care research, and strategies to improve outcomes.
“The connections between fair housing and clinical practice may appear tenuous and inconsequential to the care provided by medical specialists,” Dr. Hightower said, but he emphasized that there are clear consequences when these factors contribute to inadequate control of such diseases as HS. As a source of missed appointments and disjointed care, an unstable home life can be an important barrier to disease control – and because of scarring nodules, fistulae, pain, school absences, and social isolation, complications can be dire.
Solutions to insecure housing are not typically available to an individual clinician, but the awareness that this can be a factor can help both physicians and patients begin to think about the role this plays in impairing recovery and what solutions might be found to modify the impact. Awareness not just among individual clinicians but a broader consortium of those working to improve health care outcomes is needed to “challenge the way we are doing medicine,” he said.
While conversations about the social determinants of health, including access to resources within patients’ neighborhoods, schools, and environment, can demonstrate concern about how to address obstacles, it can also be part of a reorientation to think beyond treatment for the underlying pathology alone. Eliciting trust and emphasizing the importance of environmental barriers to adequate care can be positive steps on the path to solutions.
Participatory action research
Relevant to this orientation, Dr. Hightower spoke about participatory action research (PAR), which provides a framework for patients to participate in the planning of clinical studies to effect change, not just serve as subjects in these studies.
The assumption of PAR is that “all people have valuable knowledge about their lives and experiences,” Dr. Hightower said. From this assumption, individuals who have been historically marginalized by race, income, or other factors can help define the problems from the patient’s perspective and, from there, create studies to seek solutions.
PAR is consistent with a patient-centered approach to medical care, which Dr. Hightower called “the future of medicine.” It involves a big-picture approach to look beyond disease pathology and symptoms to factors that might be creating susceptibility to disease and undermining health care.
Organized medicine alone cannot solve the cause of social inequities leading to disparate risks for disease and risks of inadequate health care, but Dr. Hightower argued that these inequities should not be ignored. He believes medical trainees should learn how to elicit information about the barriers to adequate health care and be aware of solutions, such as fair housing policies.
While he believes that PAR is an example of a pathway to problem solving, he suggested that a comprehensive approach requires an effective method of communication between providers and patients that would lead to a collaborative and mutually reinforcing approach.
“How do we ensure that individuals from communities most impacted by health disparities are treated fairly and empowered to address these disparities?” Dr. Hightower asked. He said that this is the direction of his own research and the issues that inhibit adequate treatment of many dermatologic diseases, as well as other types of disease, in childhood.
Craig Burkhart, MD, director of a private pediatric and adolescent dermatology practice in Cary, N.C., said that Dr. Hightower’s message is relevant. The value of considering and addressing the psychological well-being of patients of any age is not a new concept, but he acknowledged that he, for one, has not routinely inquired about obstacles to follow-up care if there is a signal that this might be an issue.
“As dermatologists, we focus on the acute complaints. We want to make the patient better,” said Dr. Burkhart, who moderated the session in which Dr. Hightower spoke. He agreed with Dr. Hightower that environmental factors make a difference on the road to recovery for a patient, and his presentation was a good reminder, he said, to consider the patient’s circumstances when response to treatment is inadequate, particularly in chronic diseases like HS, for which comprehensive care and close follow-up are needed.
Dr. Hightower and Dr. Burkhart report no potential conflicts of interest.
AT SPD 2023
Dupilumab gains off-label uses as clinicians turn to drug for more indications
.
The drug, marketed as Dupixent, is currently approved in the United States to treat atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis, eosinophilic esophagitis, and prurigo nodularis in adults. Dupilumab is also approved to treat eosinophilic esophagitis in patients aged 12 years and older and atopic dermatitis and asthma in some patients as young as age 6 months.
As the roster of approved and off-label indications grows, skin specialists said, pediatricians and other primary care providers should become familiar with the drug – given the increasing likelihood that their patients may be taking the medication.
The U.S. Food and Drug Administration first approved dupilumab in 2017 for eczema and has continued to add new treatment indications, the most recent being for prurigo nodularis, in 2022. Sanofi, which markets the drug with Regeneron, announced in April 2022 that some 430,000 patients worldwide were taking the drug – a figure it hoped to raise by 1.5 million by 2025.
A well-tolerated – if expensive – drug
Dupilumab, an interleukin-4 (IL-4) receptor alpha-antagonist biologic, blocks both IL-4 and IL-13 signaling, Marlys Fassett, MD, PhD, associate professor of dermatology at the University of California, San Francisco, told this news organization.
Dr. Fassett said she prescribes the drug off label for chronic idiopathic urticaria, including in older patients, and finds that the side effects in older patients are similar to those in younger people. The medication costs $36,000 per year, although some patients can get it more cheaply.
“Dupixent is a super-safe drug because it doesn’t immunosuppress any other part of the immune system, so you still have good antibacterial, antiviral, and antifungal immunity,” she added. “That makes perfect sense as a biological mechanism, and it’s been found safe in clinical trials.”
Case reports of potential adverse reactions to dupilumab have included ocular surface disease, lichen planus, and rash on the face and neck.
“We’re still learning about complications and are watching patients carefully,” said Marissa J. Perman, MD, section chief of dermatology at Children’s Hospital of Philadelphia.
Many people with atopic dermatitis also have other allergic conditions, such as contact dermatitis, asthma, prurigo nodularis, allergic rhinitis, and seasonal allergies. Each of these conditions has a pathway that depends on IL-4 receptors, Dr. Fassett said.
“It’s amazing how many conditions Dupixent improves. Sometimes we prescribe on-label Dupixent for atopic dermatitis, and inadvertently, the drug also improves that patient’s other, off-label conditions,” Dr. Fassett said. “I think that’s the best evidence that Dupixent works in these off-label cases.”
Lindsay C. Strowd, MD, associate professor of dermatology at Wake Forest University, Winston-Salem, N.C., said she uses off-label dupilumab to treat bullous pemphigoid and intense pruritus of unknown etiology.
“And several times I have treated drug reaction with eosinophilia and systemic symptoms, a rare adverse drug reaction that causes a rash and eosinophilia,” Dr. Strowd added.
Tissa Hata, MD, professor of medicine and clinical service chief at the University of California, San Diego, mainly treats elderly patients. She uses dupilumab to treat bullous pemphigoid and chronic pruritus. “There have been reports of using Dupixent to treat adult alopecia areata, chronic urticaria, localized scleroderma, and even keloids,” she told this news organization.
As a pediatric dermatologist, Dr. Perman treats children with atopic dermatitis as young as 3 months of age. She also uses dupilumab for alopecia areata, graft vs. host disease, and pruritus not otherwise specified.
Conjunctivitis and facial redness are two side effects Dr. Fassett sometimes sees with dupilumab. They occur similarly with all conditions and in all age groups. “We don’t know why they occur, and we don’t always know how to alleviate them,” she said. “So a small number of patients stop using Dupixent because they can’t tolerate those two side effects.
“We’re not worried about infection risk,” Dr. Fassett said. “Your patients may have heard of dupilumab as an immunosuppressant, but its immunosuppression is very focused. You can reassure them that they’re not at increased risk for viral or bacterial infections when they’re on this drug.”
“I don’t think there are any different safety signals to watch for with on-label vs. off-label Dupixent use,” Dr. Strowd added. “In general, the medicine is very safe.”
Dr. Hata said she is impressed with dupilumab’s safety in her elderly patients. All her patients older than 85 years who have taken the drug for bullous pemphigoid have tolerated it well, she said.
“Dupixent seems to be a safe alternative for elderly patients with pruritus because they often cannot tolerate sedating antihistamines due to the risk of falling,” Dr. Hata said. “And UV therapy may be difficult for elderly patients due to problems with transport.”
Although some of Dr. Hata’s elderly patients with atopic dermatitis have discontinued use of the drug after developing conjunctivitis, none taking the drug off label have discontinued it because of side effects, she noted.
“Dupixent manages the condition, but it is not a cure,” Dr. Fassett noted. “Based on the current data, we think it’s safe and effective to take long term, potentially for life.”
Making injections less bothersome
Dupilumab is injected subcutaneously from a single-dose prefilled syringe or a prefilled pen (syringe hidden in an opaque sheath), typically in the thigh, arm, abdomen, or buttocks. According to Sanofi and Regeneron, patients receive dupilumab injections every 2 to 4 weeks in doses based on their age and weight.
“The medication is somewhat viscous, so taking the syringe or pen out of the refrigerator ahead of time to warm it up can make the experience less painful,” Dr. Strowd advised. “For pediatric patients, I sometimes prescribe topical lidocaine applied 30 minutes before injection.”
Dr. Hata suggested icing the skin prior to injecting or distracting the patient by tapping a different area of the skin.
For her pediatric patients, Dr. Perman said she uses “lots of distraction, EMLA cream, and having one person hold the child while a second person injects.”
Clinic and pharmacy staff may show patients how to inject properly, Dr. Fassett added; and the product website provides injection tutorials.
Off-label dupixent can be expensive, difficult to obtain
The list price per injection, regardless of dose, is around $1,800. But according to the company’s website, most patients have health insurance or qualify for other assistance, so “very few patients pay the list price.”
Even so, “due to cost and insurance coverage hurdles, obtaining Dupixent for off-label use can be difficult,” Dr. Strowd said.
“In academic medicine, we can obtain drugs for our patients that community doctors may not get approval for,” Dr. Fassett added. “Community doctors can use information in the medical literature and in news articles to press insurance companies to spend money to provide their patients with Dupixent.”
The experts who commented have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
.
The drug, marketed as Dupixent, is currently approved in the United States to treat atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis, eosinophilic esophagitis, and prurigo nodularis in adults. Dupilumab is also approved to treat eosinophilic esophagitis in patients aged 12 years and older and atopic dermatitis and asthma in some patients as young as age 6 months.
As the roster of approved and off-label indications grows, skin specialists said, pediatricians and other primary care providers should become familiar with the drug – given the increasing likelihood that their patients may be taking the medication.
The U.S. Food and Drug Administration first approved dupilumab in 2017 for eczema and has continued to add new treatment indications, the most recent being for prurigo nodularis, in 2022. Sanofi, which markets the drug with Regeneron, announced in April 2022 that some 430,000 patients worldwide were taking the drug – a figure it hoped to raise by 1.5 million by 2025.
A well-tolerated – if expensive – drug
Dupilumab, an interleukin-4 (IL-4) receptor alpha-antagonist biologic, blocks both IL-4 and IL-13 signaling, Marlys Fassett, MD, PhD, associate professor of dermatology at the University of California, San Francisco, told this news organization.
Dr. Fassett said she prescribes the drug off label for chronic idiopathic urticaria, including in older patients, and finds that the side effects in older patients are similar to those in younger people. The medication costs $36,000 per year, although some patients can get it more cheaply.
“Dupixent is a super-safe drug because it doesn’t immunosuppress any other part of the immune system, so you still have good antibacterial, antiviral, and antifungal immunity,” she added. “That makes perfect sense as a biological mechanism, and it’s been found safe in clinical trials.”
Case reports of potential adverse reactions to dupilumab have included ocular surface disease, lichen planus, and rash on the face and neck.
“We’re still learning about complications and are watching patients carefully,” said Marissa J. Perman, MD, section chief of dermatology at Children’s Hospital of Philadelphia.
Many people with atopic dermatitis also have other allergic conditions, such as contact dermatitis, asthma, prurigo nodularis, allergic rhinitis, and seasonal allergies. Each of these conditions has a pathway that depends on IL-4 receptors, Dr. Fassett said.
“It’s amazing how many conditions Dupixent improves. Sometimes we prescribe on-label Dupixent for atopic dermatitis, and inadvertently, the drug also improves that patient’s other, off-label conditions,” Dr. Fassett said. “I think that’s the best evidence that Dupixent works in these off-label cases.”
Lindsay C. Strowd, MD, associate professor of dermatology at Wake Forest University, Winston-Salem, N.C., said she uses off-label dupilumab to treat bullous pemphigoid and intense pruritus of unknown etiology.
“And several times I have treated drug reaction with eosinophilia and systemic symptoms, a rare adverse drug reaction that causes a rash and eosinophilia,” Dr. Strowd added.
Tissa Hata, MD, professor of medicine and clinical service chief at the University of California, San Diego, mainly treats elderly patients. She uses dupilumab to treat bullous pemphigoid and chronic pruritus. “There have been reports of using Dupixent to treat adult alopecia areata, chronic urticaria, localized scleroderma, and even keloids,” she told this news organization.
As a pediatric dermatologist, Dr. Perman treats children with atopic dermatitis as young as 3 months of age. She also uses dupilumab for alopecia areata, graft vs. host disease, and pruritus not otherwise specified.
Conjunctivitis and facial redness are two side effects Dr. Fassett sometimes sees with dupilumab. They occur similarly with all conditions and in all age groups. “We don’t know why they occur, and we don’t always know how to alleviate them,” she said. “So a small number of patients stop using Dupixent because they can’t tolerate those two side effects.
“We’re not worried about infection risk,” Dr. Fassett said. “Your patients may have heard of dupilumab as an immunosuppressant, but its immunosuppression is very focused. You can reassure them that they’re not at increased risk for viral or bacterial infections when they’re on this drug.”
“I don’t think there are any different safety signals to watch for with on-label vs. off-label Dupixent use,” Dr. Strowd added. “In general, the medicine is very safe.”
Dr. Hata said she is impressed with dupilumab’s safety in her elderly patients. All her patients older than 85 years who have taken the drug for bullous pemphigoid have tolerated it well, she said.
“Dupixent seems to be a safe alternative for elderly patients with pruritus because they often cannot tolerate sedating antihistamines due to the risk of falling,” Dr. Hata said. “And UV therapy may be difficult for elderly patients due to problems with transport.”
Although some of Dr. Hata’s elderly patients with atopic dermatitis have discontinued use of the drug after developing conjunctivitis, none taking the drug off label have discontinued it because of side effects, she noted.
“Dupixent manages the condition, but it is not a cure,” Dr. Fassett noted. “Based on the current data, we think it’s safe and effective to take long term, potentially for life.”
Making injections less bothersome
Dupilumab is injected subcutaneously from a single-dose prefilled syringe or a prefilled pen (syringe hidden in an opaque sheath), typically in the thigh, arm, abdomen, or buttocks. According to Sanofi and Regeneron, patients receive dupilumab injections every 2 to 4 weeks in doses based on their age and weight.
“The medication is somewhat viscous, so taking the syringe or pen out of the refrigerator ahead of time to warm it up can make the experience less painful,” Dr. Strowd advised. “For pediatric patients, I sometimes prescribe topical lidocaine applied 30 minutes before injection.”
Dr. Hata suggested icing the skin prior to injecting or distracting the patient by tapping a different area of the skin.
For her pediatric patients, Dr. Perman said she uses “lots of distraction, EMLA cream, and having one person hold the child while a second person injects.”
Clinic and pharmacy staff may show patients how to inject properly, Dr. Fassett added; and the product website provides injection tutorials.
Off-label dupixent can be expensive, difficult to obtain
The list price per injection, regardless of dose, is around $1,800. But according to the company’s website, most patients have health insurance or qualify for other assistance, so “very few patients pay the list price.”
Even so, “due to cost and insurance coverage hurdles, obtaining Dupixent for off-label use can be difficult,” Dr. Strowd said.
“In academic medicine, we can obtain drugs for our patients that community doctors may not get approval for,” Dr. Fassett added. “Community doctors can use information in the medical literature and in news articles to press insurance companies to spend money to provide their patients with Dupixent.”
The experts who commented have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
.
The drug, marketed as Dupixent, is currently approved in the United States to treat atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis, eosinophilic esophagitis, and prurigo nodularis in adults. Dupilumab is also approved to treat eosinophilic esophagitis in patients aged 12 years and older and atopic dermatitis and asthma in some patients as young as age 6 months.
As the roster of approved and off-label indications grows, skin specialists said, pediatricians and other primary care providers should become familiar with the drug – given the increasing likelihood that their patients may be taking the medication.
The U.S. Food and Drug Administration first approved dupilumab in 2017 for eczema and has continued to add new treatment indications, the most recent being for prurigo nodularis, in 2022. Sanofi, which markets the drug with Regeneron, announced in April 2022 that some 430,000 patients worldwide were taking the drug – a figure it hoped to raise by 1.5 million by 2025.
A well-tolerated – if expensive – drug
Dupilumab, an interleukin-4 (IL-4) receptor alpha-antagonist biologic, blocks both IL-4 and IL-13 signaling, Marlys Fassett, MD, PhD, associate professor of dermatology at the University of California, San Francisco, told this news organization.
Dr. Fassett said she prescribes the drug off label for chronic idiopathic urticaria, including in older patients, and finds that the side effects in older patients are similar to those in younger people. The medication costs $36,000 per year, although some patients can get it more cheaply.
“Dupixent is a super-safe drug because it doesn’t immunosuppress any other part of the immune system, so you still have good antibacterial, antiviral, and antifungal immunity,” she added. “That makes perfect sense as a biological mechanism, and it’s been found safe in clinical trials.”
Case reports of potential adverse reactions to dupilumab have included ocular surface disease, lichen planus, and rash on the face and neck.
“We’re still learning about complications and are watching patients carefully,” said Marissa J. Perman, MD, section chief of dermatology at Children’s Hospital of Philadelphia.
Many people with atopic dermatitis also have other allergic conditions, such as contact dermatitis, asthma, prurigo nodularis, allergic rhinitis, and seasonal allergies. Each of these conditions has a pathway that depends on IL-4 receptors, Dr. Fassett said.
“It’s amazing how many conditions Dupixent improves. Sometimes we prescribe on-label Dupixent for atopic dermatitis, and inadvertently, the drug also improves that patient’s other, off-label conditions,” Dr. Fassett said. “I think that’s the best evidence that Dupixent works in these off-label cases.”
Lindsay C. Strowd, MD, associate professor of dermatology at Wake Forest University, Winston-Salem, N.C., said she uses off-label dupilumab to treat bullous pemphigoid and intense pruritus of unknown etiology.
“And several times I have treated drug reaction with eosinophilia and systemic symptoms, a rare adverse drug reaction that causes a rash and eosinophilia,” Dr. Strowd added.
Tissa Hata, MD, professor of medicine and clinical service chief at the University of California, San Diego, mainly treats elderly patients. She uses dupilumab to treat bullous pemphigoid and chronic pruritus. “There have been reports of using Dupixent to treat adult alopecia areata, chronic urticaria, localized scleroderma, and even keloids,” she told this news organization.
As a pediatric dermatologist, Dr. Perman treats children with atopic dermatitis as young as 3 months of age. She also uses dupilumab for alopecia areata, graft vs. host disease, and pruritus not otherwise specified.
Conjunctivitis and facial redness are two side effects Dr. Fassett sometimes sees with dupilumab. They occur similarly with all conditions and in all age groups. “We don’t know why they occur, and we don’t always know how to alleviate them,” she said. “So a small number of patients stop using Dupixent because they can’t tolerate those two side effects.
“We’re not worried about infection risk,” Dr. Fassett said. “Your patients may have heard of dupilumab as an immunosuppressant, but its immunosuppression is very focused. You can reassure them that they’re not at increased risk for viral or bacterial infections when they’re on this drug.”
“I don’t think there are any different safety signals to watch for with on-label vs. off-label Dupixent use,” Dr. Strowd added. “In general, the medicine is very safe.”
Dr. Hata said she is impressed with dupilumab’s safety in her elderly patients. All her patients older than 85 years who have taken the drug for bullous pemphigoid have tolerated it well, she said.
“Dupixent seems to be a safe alternative for elderly patients with pruritus because they often cannot tolerate sedating antihistamines due to the risk of falling,” Dr. Hata said. “And UV therapy may be difficult for elderly patients due to problems with transport.”
Although some of Dr. Hata’s elderly patients with atopic dermatitis have discontinued use of the drug after developing conjunctivitis, none taking the drug off label have discontinued it because of side effects, she noted.
“Dupixent manages the condition, but it is not a cure,” Dr. Fassett noted. “Based on the current data, we think it’s safe and effective to take long term, potentially for life.”
Making injections less bothersome
Dupilumab is injected subcutaneously from a single-dose prefilled syringe or a prefilled pen (syringe hidden in an opaque sheath), typically in the thigh, arm, abdomen, or buttocks. According to Sanofi and Regeneron, patients receive dupilumab injections every 2 to 4 weeks in doses based on their age and weight.
“The medication is somewhat viscous, so taking the syringe or pen out of the refrigerator ahead of time to warm it up can make the experience less painful,” Dr. Strowd advised. “For pediatric patients, I sometimes prescribe topical lidocaine applied 30 minutes before injection.”
Dr. Hata suggested icing the skin prior to injecting or distracting the patient by tapping a different area of the skin.
For her pediatric patients, Dr. Perman said she uses “lots of distraction, EMLA cream, and having one person hold the child while a second person injects.”
Clinic and pharmacy staff may show patients how to inject properly, Dr. Fassett added; and the product website provides injection tutorials.
Off-label dupixent can be expensive, difficult to obtain
The list price per injection, regardless of dose, is around $1,800. But according to the company’s website, most patients have health insurance or qualify for other assistance, so “very few patients pay the list price.”
Even so, “due to cost and insurance coverage hurdles, obtaining Dupixent for off-label use can be difficult,” Dr. Strowd said.
“In academic medicine, we can obtain drugs for our patients that community doctors may not get approval for,” Dr. Fassett added. “Community doctors can use information in the medical literature and in news articles to press insurance companies to spend money to provide their patients with Dupixent.”
The experts who commented have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Diffuse Annular Plaques in an Infant
The Diagnosis: Neonatal Lupus Erythematosus
A review of the medical records of the patient’s mother from her first pregnancy revealed positive anti-Ro/SSA (Sjögren syndrome A) (>8.0 U [reference range <1.0 U]) and anti-La/SSB (Sjögren syndrome B) antibodies (>8.0 U [reference range <1.0 U]), which were reconfirmed during her pregnancy with our patient (the second child). The patient’s older brother was diagnosed with neonatal lupus erythematosus (NLE) 2 years prior at 1 month of age; therefore, the mother took hydroxychloroquine during the pregnancy with the second child to help prevent heart block if the child was diagnosed with NLE. Given the family history, positive antibodies in the mother, and clinical presentation, our patient was diagnosed with NLE. He was referred to a pediatric cardiologist and pediatrician to continue the workup of systemic manifestations of NLE and to rule out the presence of congenital heart block. The rash resolved 6 months after the initial presentation, and he did not develop any systemic manifestations of NLE.
Neonatal lupus erythematosus is a rare acquired autoimmune disorder caused by the placental transfer of anti-Ro/SSA and anti-La/SSB antibodies and less commonly anti-U1 ribonucleoprotein antinuclear autoantibodies.1,2 Approximately 1% to 2% of mothers with these positive antibodies will have infants affected with NLE.2 The annual prevalence of NLE in the United States is approximately 1 in 20,000 live births. Mothers of children with NLE most commonly have clinical Sjögren syndrome; however, anti-Ro/SSA and anti-LA/SSB antibodies may be present in 0.1% to 1.5% of healthy women, and 25% to 60% of women with autoimmune disease may be asymptomatic.1 As demonstrated in our case, when there is a family history of NLE in an infant from an earlier pregnancy, the risk for NLE increases to 17% to 20% in subsequent pregnancies1,3 and up to 25% in subsequent pregnancies if the initial child was diagnosed with a congenital heart block in the setting of NLE.1
Neonatal lupus erythematosus classically presents as annular erythematous macules and plaques with central scaling, telangictasia, atrophy, and pigmentary changes. It may start on the scalp and face and spread caudally.1,2 Patients may develop these lesions after UV exposure, and 80% of infants may not have dermatologic findings at birth. Importantly, 40% to 60% of mothers may be asymptomatic at the time of presentation of their child’s NLE.1 The diagnosis can be confirmed via antibody testing in the mother and/or infant. If performed, a punch biopsy shows interface dermatitis, vacuolar degeneration, and possible periadnexal lymphocytic infiltrates on histopathology.1,2
Management of cutaneous NLE includes sun protection (eg, application of sunscreen) and topical corticosteroids. Most dermatologic manifestations of NLE are transient, resolving after clearance of maternal IgG antibodies in 6 to 9 months; however, some telangiectasia, dyspigmentation, and atrophic scarring may persist.1-3
Neonatal lupus erythematosus also may have hepatobiliary, cardiac, hematologic, and less commonly neurologic manifestations. Hepatobiliary manifestations usually present as hepatomegaly or asymptomatic elevated transaminases or γ-glutamyl transferase.1,3 Approximately 10% to 20% of infants with NLE may present with transient anemia and thrombocytopenia.1 Cardiac manifestations are permanent and may require pacemaker implantation.1,3 The incidence of a congenital heart block in infants with NLE is 15% to 30%.3 Cardiac NLE most commonly injures the conductive tissue, leading to a congenital atrioventricular block. The development of a congenital heart block develops in the 18th to 24th week of gestation. Manifestations of a more advanced condition can include dilation of the ascending aorta and dilated cardiomyopathy.1 As such, patients need to be followed by a pediatric cardiologist for monitoring and treatment of any cardiac manifestations.
The overall prognosis of infants affected with NLE varies. Cardiac involvement is associated with a poor prognosis, while isolated cutaneous involvement requires little treatment and portends a favorable prognosis. It is critical for dermatologists to recognize NLE to refer patients to appropriate specialists to investigate and further monitor possible extracutaneous manifestations. With an understanding of the increased risk for a congenital heart block and NLE in subsequent pregnancies, mothers with positive anti-Ro/La antibodies should receive timely counseling and screening. In expectant mothers with suspected autoimmune disease, testing for antinuclear antibodies and SSA and SSB antibodies can be considered, as administration of hydroxychloroquine or prenatal systemic corticosteroids has proven to be effective in preventing a congenital heart block.1 Our patient was followed by pediatric cardiology and was not found to have a congenital heart block.
The differential diagnosis includes other causes of annular erythema in infants, as NLE can mimic several conditions. Tinea corporis may present as scaly annular plaques with central clearing; however, it rarely is encountered fulminantly in neonates.4 Erythema multiforme is a mucocutaneous hypersensitivy reaction distinguished by targetoid morphology.5 It is an exceedingly rare diagnosis in neonates; the average pediatric age of onset is 5.6 years.6 Erythema multiforme often is associated with an infection, most commonly herpes simplex virus,5 and mucosal involvement is common.6 Urticaria multiforme (also known as acute annular urticaria) is a benign disease that appears between 2 months to 3 years of age with blanchable urticarial plaques that likely are triggered by viral or bacterial infections, antibiotics, or vaccines.6 Specific lesions usually will resolve within 24 hours. Annular erythema of infancy is a benign and asymptomatic gyrate erythema that presents as annular plaques with palpable borders that spread centrifugally in patients younger than 1 year. Notably, lesions should periodically fade and may reappear cyclically for months to years. Evaluation for underlying disease usually is negative.6
- Derdulska JM, Rudnicka L, Szykut-Badaczewska A, et al. Neonatal lupus erythematosus—practical guidelines. J Perinat Med. 2021;49:529-538. doi:10.1515/jpm-2020-0543
- Wu J, Berk-Krauss J, Glick SA. Neonatal lupus erythematosus. JAMA Dermatol. 2021;157:590. doi:10.1001/jamadermatol.2021.0041
- Hon KL, Leung AK. Neonatal lupus erythematosus. Autoimmune Dis. 2012;2012:301274. doi:10.1155/2012/301274
- Khare AK, Gupta LK, Mittal A, et al. Neonatal tinea corporis. Indian J Dermatol. 2010;55:201. doi:10.4103/0019-5154.6274
- Ang-Tiu CU, Nicolas ME. Erythema multiforme in a 25-day old neonate. Pediatr Dermatol. 2013;30:E118-E120. doi:10.1111 /j.1525-1470.2012.01873.x
- Agnihotri G, Tsoukas MM. Annular skin lesions in infancy [published online February 3, 2022]. Clin Dermatol. 2022;40:505-512. doi:10.1016/j.clindermatol.2021.12.011
The Diagnosis: Neonatal Lupus Erythematosus
A review of the medical records of the patient’s mother from her first pregnancy revealed positive anti-Ro/SSA (Sjögren syndrome A) (>8.0 U [reference range <1.0 U]) and anti-La/SSB (Sjögren syndrome B) antibodies (>8.0 U [reference range <1.0 U]), which were reconfirmed during her pregnancy with our patient (the second child). The patient’s older brother was diagnosed with neonatal lupus erythematosus (NLE) 2 years prior at 1 month of age; therefore, the mother took hydroxychloroquine during the pregnancy with the second child to help prevent heart block if the child was diagnosed with NLE. Given the family history, positive antibodies in the mother, and clinical presentation, our patient was diagnosed with NLE. He was referred to a pediatric cardiologist and pediatrician to continue the workup of systemic manifestations of NLE and to rule out the presence of congenital heart block. The rash resolved 6 months after the initial presentation, and he did not develop any systemic manifestations of NLE.
Neonatal lupus erythematosus is a rare acquired autoimmune disorder caused by the placental transfer of anti-Ro/SSA and anti-La/SSB antibodies and less commonly anti-U1 ribonucleoprotein antinuclear autoantibodies.1,2 Approximately 1% to 2% of mothers with these positive antibodies will have infants affected with NLE.2 The annual prevalence of NLE in the United States is approximately 1 in 20,000 live births. Mothers of children with NLE most commonly have clinical Sjögren syndrome; however, anti-Ro/SSA and anti-LA/SSB antibodies may be present in 0.1% to 1.5% of healthy women, and 25% to 60% of women with autoimmune disease may be asymptomatic.1 As demonstrated in our case, when there is a family history of NLE in an infant from an earlier pregnancy, the risk for NLE increases to 17% to 20% in subsequent pregnancies1,3 and up to 25% in subsequent pregnancies if the initial child was diagnosed with a congenital heart block in the setting of NLE.1
Neonatal lupus erythematosus classically presents as annular erythematous macules and plaques with central scaling, telangictasia, atrophy, and pigmentary changes. It may start on the scalp and face and spread caudally.1,2 Patients may develop these lesions after UV exposure, and 80% of infants may not have dermatologic findings at birth. Importantly, 40% to 60% of mothers may be asymptomatic at the time of presentation of their child’s NLE.1 The diagnosis can be confirmed via antibody testing in the mother and/or infant. If performed, a punch biopsy shows interface dermatitis, vacuolar degeneration, and possible periadnexal lymphocytic infiltrates on histopathology.1,2
Management of cutaneous NLE includes sun protection (eg, application of sunscreen) and topical corticosteroids. Most dermatologic manifestations of NLE are transient, resolving after clearance of maternal IgG antibodies in 6 to 9 months; however, some telangiectasia, dyspigmentation, and atrophic scarring may persist.1-3
Neonatal lupus erythematosus also may have hepatobiliary, cardiac, hematologic, and less commonly neurologic manifestations. Hepatobiliary manifestations usually present as hepatomegaly or asymptomatic elevated transaminases or γ-glutamyl transferase.1,3 Approximately 10% to 20% of infants with NLE may present with transient anemia and thrombocytopenia.1 Cardiac manifestations are permanent and may require pacemaker implantation.1,3 The incidence of a congenital heart block in infants with NLE is 15% to 30%.3 Cardiac NLE most commonly injures the conductive tissue, leading to a congenital atrioventricular block. The development of a congenital heart block develops in the 18th to 24th week of gestation. Manifestations of a more advanced condition can include dilation of the ascending aorta and dilated cardiomyopathy.1 As such, patients need to be followed by a pediatric cardiologist for monitoring and treatment of any cardiac manifestations.
The overall prognosis of infants affected with NLE varies. Cardiac involvement is associated with a poor prognosis, while isolated cutaneous involvement requires little treatment and portends a favorable prognosis. It is critical for dermatologists to recognize NLE to refer patients to appropriate specialists to investigate and further monitor possible extracutaneous manifestations. With an understanding of the increased risk for a congenital heart block and NLE in subsequent pregnancies, mothers with positive anti-Ro/La antibodies should receive timely counseling and screening. In expectant mothers with suspected autoimmune disease, testing for antinuclear antibodies and SSA and SSB antibodies can be considered, as administration of hydroxychloroquine or prenatal systemic corticosteroids has proven to be effective in preventing a congenital heart block.1 Our patient was followed by pediatric cardiology and was not found to have a congenital heart block.
The differential diagnosis includes other causes of annular erythema in infants, as NLE can mimic several conditions. Tinea corporis may present as scaly annular plaques with central clearing; however, it rarely is encountered fulminantly in neonates.4 Erythema multiforme is a mucocutaneous hypersensitivy reaction distinguished by targetoid morphology.5 It is an exceedingly rare diagnosis in neonates; the average pediatric age of onset is 5.6 years.6 Erythema multiforme often is associated with an infection, most commonly herpes simplex virus,5 and mucosal involvement is common.6 Urticaria multiforme (also known as acute annular urticaria) is a benign disease that appears between 2 months to 3 years of age with blanchable urticarial plaques that likely are triggered by viral or bacterial infections, antibiotics, or vaccines.6 Specific lesions usually will resolve within 24 hours. Annular erythema of infancy is a benign and asymptomatic gyrate erythema that presents as annular plaques with palpable borders that spread centrifugally in patients younger than 1 year. Notably, lesions should periodically fade and may reappear cyclically for months to years. Evaluation for underlying disease usually is negative.6
The Diagnosis: Neonatal Lupus Erythematosus
A review of the medical records of the patient’s mother from her first pregnancy revealed positive anti-Ro/SSA (Sjögren syndrome A) (>8.0 U [reference range <1.0 U]) and anti-La/SSB (Sjögren syndrome B) antibodies (>8.0 U [reference range <1.0 U]), which were reconfirmed during her pregnancy with our patient (the second child). The patient’s older brother was diagnosed with neonatal lupus erythematosus (NLE) 2 years prior at 1 month of age; therefore, the mother took hydroxychloroquine during the pregnancy with the second child to help prevent heart block if the child was diagnosed with NLE. Given the family history, positive antibodies in the mother, and clinical presentation, our patient was diagnosed with NLE. He was referred to a pediatric cardiologist and pediatrician to continue the workup of systemic manifestations of NLE and to rule out the presence of congenital heart block. The rash resolved 6 months after the initial presentation, and he did not develop any systemic manifestations of NLE.
Neonatal lupus erythematosus is a rare acquired autoimmune disorder caused by the placental transfer of anti-Ro/SSA and anti-La/SSB antibodies and less commonly anti-U1 ribonucleoprotein antinuclear autoantibodies.1,2 Approximately 1% to 2% of mothers with these positive antibodies will have infants affected with NLE.2 The annual prevalence of NLE in the United States is approximately 1 in 20,000 live births. Mothers of children with NLE most commonly have clinical Sjögren syndrome; however, anti-Ro/SSA and anti-LA/SSB antibodies may be present in 0.1% to 1.5% of healthy women, and 25% to 60% of women with autoimmune disease may be asymptomatic.1 As demonstrated in our case, when there is a family history of NLE in an infant from an earlier pregnancy, the risk for NLE increases to 17% to 20% in subsequent pregnancies1,3 and up to 25% in subsequent pregnancies if the initial child was diagnosed with a congenital heart block in the setting of NLE.1
Neonatal lupus erythematosus classically presents as annular erythematous macules and plaques with central scaling, telangictasia, atrophy, and pigmentary changes. It may start on the scalp and face and spread caudally.1,2 Patients may develop these lesions after UV exposure, and 80% of infants may not have dermatologic findings at birth. Importantly, 40% to 60% of mothers may be asymptomatic at the time of presentation of their child’s NLE.1 The diagnosis can be confirmed via antibody testing in the mother and/or infant. If performed, a punch biopsy shows interface dermatitis, vacuolar degeneration, and possible periadnexal lymphocytic infiltrates on histopathology.1,2
Management of cutaneous NLE includes sun protection (eg, application of sunscreen) and topical corticosteroids. Most dermatologic manifestations of NLE are transient, resolving after clearance of maternal IgG antibodies in 6 to 9 months; however, some telangiectasia, dyspigmentation, and atrophic scarring may persist.1-3
Neonatal lupus erythematosus also may have hepatobiliary, cardiac, hematologic, and less commonly neurologic manifestations. Hepatobiliary manifestations usually present as hepatomegaly or asymptomatic elevated transaminases or γ-glutamyl transferase.1,3 Approximately 10% to 20% of infants with NLE may present with transient anemia and thrombocytopenia.1 Cardiac manifestations are permanent and may require pacemaker implantation.1,3 The incidence of a congenital heart block in infants with NLE is 15% to 30%.3 Cardiac NLE most commonly injures the conductive tissue, leading to a congenital atrioventricular block. The development of a congenital heart block develops in the 18th to 24th week of gestation. Manifestations of a more advanced condition can include dilation of the ascending aorta and dilated cardiomyopathy.1 As such, patients need to be followed by a pediatric cardiologist for monitoring and treatment of any cardiac manifestations.
The overall prognosis of infants affected with NLE varies. Cardiac involvement is associated with a poor prognosis, while isolated cutaneous involvement requires little treatment and portends a favorable prognosis. It is critical for dermatologists to recognize NLE to refer patients to appropriate specialists to investigate and further monitor possible extracutaneous manifestations. With an understanding of the increased risk for a congenital heart block and NLE in subsequent pregnancies, mothers with positive anti-Ro/La antibodies should receive timely counseling and screening. In expectant mothers with suspected autoimmune disease, testing for antinuclear antibodies and SSA and SSB antibodies can be considered, as administration of hydroxychloroquine or prenatal systemic corticosteroids has proven to be effective in preventing a congenital heart block.1 Our patient was followed by pediatric cardiology and was not found to have a congenital heart block.
The differential diagnosis includes other causes of annular erythema in infants, as NLE can mimic several conditions. Tinea corporis may present as scaly annular plaques with central clearing; however, it rarely is encountered fulminantly in neonates.4 Erythema multiforme is a mucocutaneous hypersensitivy reaction distinguished by targetoid morphology.5 It is an exceedingly rare diagnosis in neonates; the average pediatric age of onset is 5.6 years.6 Erythema multiforme often is associated with an infection, most commonly herpes simplex virus,5 and mucosal involvement is common.6 Urticaria multiforme (also known as acute annular urticaria) is a benign disease that appears between 2 months to 3 years of age with blanchable urticarial plaques that likely are triggered by viral or bacterial infections, antibiotics, or vaccines.6 Specific lesions usually will resolve within 24 hours. Annular erythema of infancy is a benign and asymptomatic gyrate erythema that presents as annular plaques with palpable borders that spread centrifugally in patients younger than 1 year. Notably, lesions should periodically fade and may reappear cyclically for months to years. Evaluation for underlying disease usually is negative.6
- Derdulska JM, Rudnicka L, Szykut-Badaczewska A, et al. Neonatal lupus erythematosus—practical guidelines. J Perinat Med. 2021;49:529-538. doi:10.1515/jpm-2020-0543
- Wu J, Berk-Krauss J, Glick SA. Neonatal lupus erythematosus. JAMA Dermatol. 2021;157:590. doi:10.1001/jamadermatol.2021.0041
- Hon KL, Leung AK. Neonatal lupus erythematosus. Autoimmune Dis. 2012;2012:301274. doi:10.1155/2012/301274
- Khare AK, Gupta LK, Mittal A, et al. Neonatal tinea corporis. Indian J Dermatol. 2010;55:201. doi:10.4103/0019-5154.6274
- Ang-Tiu CU, Nicolas ME. Erythema multiforme in a 25-day old neonate. Pediatr Dermatol. 2013;30:E118-E120. doi:10.1111 /j.1525-1470.2012.01873.x
- Agnihotri G, Tsoukas MM. Annular skin lesions in infancy [published online February 3, 2022]. Clin Dermatol. 2022;40:505-512. doi:10.1016/j.clindermatol.2021.12.011
- Derdulska JM, Rudnicka L, Szykut-Badaczewska A, et al. Neonatal lupus erythematosus—practical guidelines. J Perinat Med. 2021;49:529-538. doi:10.1515/jpm-2020-0543
- Wu J, Berk-Krauss J, Glick SA. Neonatal lupus erythematosus. JAMA Dermatol. 2021;157:590. doi:10.1001/jamadermatol.2021.0041
- Hon KL, Leung AK. Neonatal lupus erythematosus. Autoimmune Dis. 2012;2012:301274. doi:10.1155/2012/301274
- Khare AK, Gupta LK, Mittal A, et al. Neonatal tinea corporis. Indian J Dermatol. 2010;55:201. doi:10.4103/0019-5154.6274
- Ang-Tiu CU, Nicolas ME. Erythema multiforme in a 25-day old neonate. Pediatr Dermatol. 2013;30:E118-E120. doi:10.1111 /j.1525-1470.2012.01873.x
- Agnihotri G, Tsoukas MM. Annular skin lesions in infancy [published online February 3, 2022]. Clin Dermatol. 2022;40:505-512. doi:10.1016/j.clindermatol.2021.12.011
A 5-week-old infant boy presented with a rash at birth (left). The pregnancy was full term without complications, and he was otherwise healthy. A family history revealed that his older brother developed a similar rash 2 weeks after birth (right). Physical examination revealed polycyclic annular patches with an erythematous border and central clearing diffusely located on the trunk, extremities, scalp, and face with periorbital edema.