User login
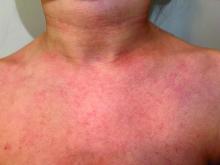
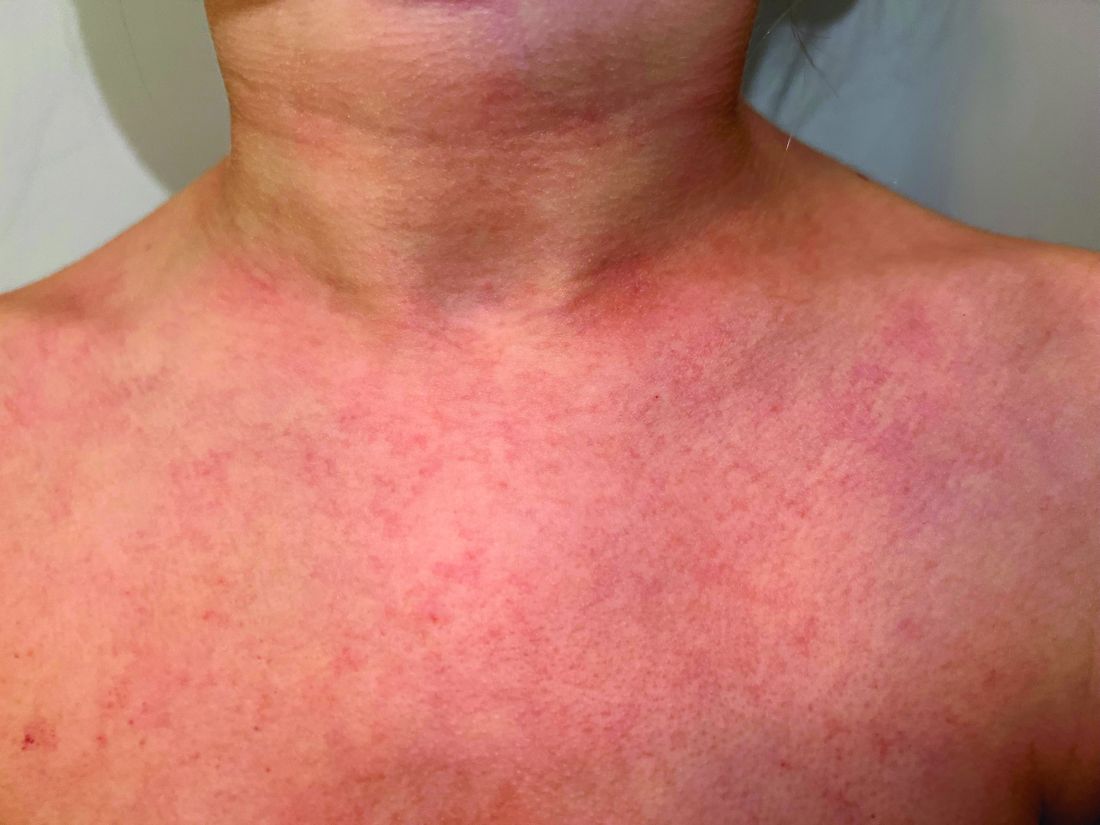
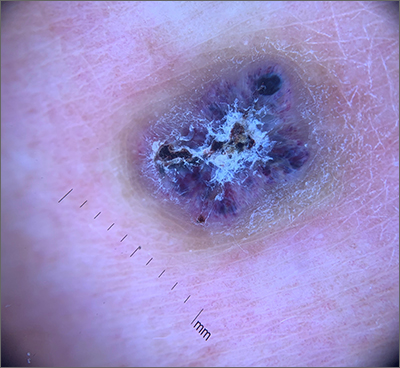
A 9-year old female presented with 1 day of fever, fatigue, and sore throat
This condition typically presents in the setting of Streptococcus pyogenes pharyngitis, or strep throat, and is spread via mucosal transfer in close proximity such as classrooms and nurseries. The dermatologic symptoms are a result of the endotoxin produced by S. pyogenes, which is part of the group A Strep bacteria. Clinically, the presentation can be differentiated from an allergic eruption by its relation to acute pharyngitis, insidious onset, and lack of confluence of the lesions. Diagnosis is supported by a throat culture and rapid strep test, although a rapid test lacks reliability in older patients who are less commonly affected and likely to be carriers. First-line treatment is penicillin or amoxicillin, but first-generation cephalosporins, clindamycin, or erythromycin are sufficient if the patient is allergic to penicillins. Prognosis worsens as time between onset and treatment increases, but is overall excellent now with the introduction of antibiotics and improved hygiene.
Scarlet fever is among a list of many common childhood rashes, and it can be difficult to differentiate between these pathologies on clinical presentation. A few notable childhood dermatologic eruptions include erythema infectiosum (fifth disease), roseola (exanthema subitum or sixth disease), and measles. These cases can be distinguished clinically by the age of the patient, distribution, and quality of the symptoms. Laboratory testing may be used to confirm the diagnosis.
Erythema infectiosum is known as fifth disease or slapped-cheek rash because it commonly presents on the cheeks as a pink, maculopapular rash in a reticular pattern. The disease is caused by parvovirus B19 and is accompanied by low fever, malaise, headache, sore throat, and nausea, which precedes the erythematous rash. The facial rash appears first and is followed by patchy eruptions on the extremities. Appearance of the rash typically indicates the patient is no longer contagious, and patients are treated symptomatically with NSAIDs and antihistamines for associated pruritus.
Roseola infantum is commonly caused by human herpesvirus 6 and is usually found in children 3 years and younger. The defining symptom is a high fever, which is paired with a mild cough, runny nose, and diarrhea. A maculopapular rash appears after the fever subsides, starting centrally and spreading outward to the extremities. Although this rash is similar to measles, they can be differentiated by the order of onset. The rash caused by measles begins on the face and mouth (Koplik spots) and moves downward. Additionally, the patient appears generally healthy and the disease is self-limiting in roseola, while patients with measles will appear more ill and require further attention. Measles is caused by the measles virus of the genus Morbillivirus and is highly contagious. It is spread via respiratory route presenting with fever, cough, coryza, and conjunctivitis followed by the rash. Fortunately, the measles vaccine is in widespread use, so cases have declined over the years.
Our patient had a positive strep test. Influenza and coronavirus tests were negative. She was started on daily amoxicillin and the rash resolved within 2 days of taking the antibiotics.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Tampa, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Allmon A et al.. Am Fam Physician. 2015 Aug 1;92(3):211-6.
Moss WJ. Lancet. 2017 Dec 2;390(10111):2490-502.
Mullins TB and Krishnamurthy K. Roseola Infantum, in “StatPearls.” Treasure Islan, Fla.: StatPearls Publishing, 2022.
Pardo S and Perera TB. Scarlet Fever, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2022.
This condition typically presents in the setting of Streptococcus pyogenes pharyngitis, or strep throat, and is spread via mucosal transfer in close proximity such as classrooms and nurseries. The dermatologic symptoms are a result of the endotoxin produced by S. pyogenes, which is part of the group A Strep bacteria. Clinically, the presentation can be differentiated from an allergic eruption by its relation to acute pharyngitis, insidious onset, and lack of confluence of the lesions. Diagnosis is supported by a throat culture and rapid strep test, although a rapid test lacks reliability in older patients who are less commonly affected and likely to be carriers. First-line treatment is penicillin or amoxicillin, but first-generation cephalosporins, clindamycin, or erythromycin are sufficient if the patient is allergic to penicillins. Prognosis worsens as time between onset and treatment increases, but is overall excellent now with the introduction of antibiotics and improved hygiene.
Scarlet fever is among a list of many common childhood rashes, and it can be difficult to differentiate between these pathologies on clinical presentation. A few notable childhood dermatologic eruptions include erythema infectiosum (fifth disease), roseola (exanthema subitum or sixth disease), and measles. These cases can be distinguished clinically by the age of the patient, distribution, and quality of the symptoms. Laboratory testing may be used to confirm the diagnosis.
Erythema infectiosum is known as fifth disease or slapped-cheek rash because it commonly presents on the cheeks as a pink, maculopapular rash in a reticular pattern. The disease is caused by parvovirus B19 and is accompanied by low fever, malaise, headache, sore throat, and nausea, which precedes the erythematous rash. The facial rash appears first and is followed by patchy eruptions on the extremities. Appearance of the rash typically indicates the patient is no longer contagious, and patients are treated symptomatically with NSAIDs and antihistamines for associated pruritus.
Roseola infantum is commonly caused by human herpesvirus 6 and is usually found in children 3 years and younger. The defining symptom is a high fever, which is paired with a mild cough, runny nose, and diarrhea. A maculopapular rash appears after the fever subsides, starting centrally and spreading outward to the extremities. Although this rash is similar to measles, they can be differentiated by the order of onset. The rash caused by measles begins on the face and mouth (Koplik spots) and moves downward. Additionally, the patient appears generally healthy and the disease is self-limiting in roseola, while patients with measles will appear more ill and require further attention. Measles is caused by the measles virus of the genus Morbillivirus and is highly contagious. It is spread via respiratory route presenting with fever, cough, coryza, and conjunctivitis followed by the rash. Fortunately, the measles vaccine is in widespread use, so cases have declined over the years.
Our patient had a positive strep test. Influenza and coronavirus tests were negative. She was started on daily amoxicillin and the rash resolved within 2 days of taking the antibiotics.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Tampa, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Allmon A et al.. Am Fam Physician. 2015 Aug 1;92(3):211-6.
Moss WJ. Lancet. 2017 Dec 2;390(10111):2490-502.
Mullins TB and Krishnamurthy K. Roseola Infantum, in “StatPearls.” Treasure Islan, Fla.: StatPearls Publishing, 2022.
Pardo S and Perera TB. Scarlet Fever, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2022.
This condition typically presents in the setting of Streptococcus pyogenes pharyngitis, or strep throat, and is spread via mucosal transfer in close proximity such as classrooms and nurseries. The dermatologic symptoms are a result of the endotoxin produced by S. pyogenes, which is part of the group A Strep bacteria. Clinically, the presentation can be differentiated from an allergic eruption by its relation to acute pharyngitis, insidious onset, and lack of confluence of the lesions. Diagnosis is supported by a throat culture and rapid strep test, although a rapid test lacks reliability in older patients who are less commonly affected and likely to be carriers. First-line treatment is penicillin or amoxicillin, but first-generation cephalosporins, clindamycin, or erythromycin are sufficient if the patient is allergic to penicillins. Prognosis worsens as time between onset and treatment increases, but is overall excellent now with the introduction of antibiotics and improved hygiene.
Scarlet fever is among a list of many common childhood rashes, and it can be difficult to differentiate between these pathologies on clinical presentation. A few notable childhood dermatologic eruptions include erythema infectiosum (fifth disease), roseola (exanthema subitum or sixth disease), and measles. These cases can be distinguished clinically by the age of the patient, distribution, and quality of the symptoms. Laboratory testing may be used to confirm the diagnosis.
Erythema infectiosum is known as fifth disease or slapped-cheek rash because it commonly presents on the cheeks as a pink, maculopapular rash in a reticular pattern. The disease is caused by parvovirus B19 and is accompanied by low fever, malaise, headache, sore throat, and nausea, which precedes the erythematous rash. The facial rash appears first and is followed by patchy eruptions on the extremities. Appearance of the rash typically indicates the patient is no longer contagious, and patients are treated symptomatically with NSAIDs and antihistamines for associated pruritus.
Roseola infantum is commonly caused by human herpesvirus 6 and is usually found in children 3 years and younger. The defining symptom is a high fever, which is paired with a mild cough, runny nose, and diarrhea. A maculopapular rash appears after the fever subsides, starting centrally and spreading outward to the extremities. Although this rash is similar to measles, they can be differentiated by the order of onset. The rash caused by measles begins on the face and mouth (Koplik spots) and moves downward. Additionally, the patient appears generally healthy and the disease is self-limiting in roseola, while patients with measles will appear more ill and require further attention. Measles is caused by the measles virus of the genus Morbillivirus and is highly contagious. It is spread via respiratory route presenting with fever, cough, coryza, and conjunctivitis followed by the rash. Fortunately, the measles vaccine is in widespread use, so cases have declined over the years.
Our patient had a positive strep test. Influenza and coronavirus tests were negative. She was started on daily amoxicillin and the rash resolved within 2 days of taking the antibiotics.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Tampa, and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Allmon A et al.. Am Fam Physician. 2015 Aug 1;92(3):211-6.
Moss WJ. Lancet. 2017 Dec 2;390(10111):2490-502.
Mullins TB and Krishnamurthy K. Roseola Infantum, in “StatPearls.” Treasure Islan, Fla.: StatPearls Publishing, 2022.
Pardo S and Perera TB. Scarlet Fever, in “StatPearls.” Treasure Island, Fla.: StatPearls Publishing, 2022.
Review gives weight to supplements for hair loss
because of small sample sizes, heterogeneity of hair loss types in study subjects, or other limitations.
The review, published online in JAMA Dermatology, notes that “Twelve of the 20 nutritional interventions had high-quality studies suggesting objectively evaluated effectiveness.”
It is “ground breaking,” in part because of its breadth and depth, said Eva Simmons-O’Brien, MD, a dermatologist in Towson, Md., who often recommends supplements for her patients with hair loss. “It basically kind of vindicates what some of us have been doing for a number of years in terms of treating hair loss,” she told this news organization. “It should hopefully make it more commonplace for dermatologists to consider using nutritional supplements as an adjuvant to treating hair loss,” added Dr. Simmons-O’Brien.
The review “is very helpful,” agreed Lynne J. Goldberg, MD, professor of dermatology and pathology and laboratory medicine at Boston University. Dr. Goldberg noted that many patients are already taking supplements and want to know whether they are safe and effective. The review “points out what the problems are; it talks about what the individual ingredients are and what they do, what the problems are; and it concluded that some people may find these helpful. Which is exactly what I tell my patients,” said Dr. Goldberg, who is also director of the Hair Clinic at Boston Medical Center.
“For patients who are highly motivated and eager to try this, we’re hoping that this systematic review serves as a foundation to have a conversation,” study coauthor Arash Mostaghimi, MD, MPA, MPH, of the department of dermatology at Harvard Medical School, told this news organization. “When there’s medical uncertainty and the question is how much risk is one willing to take, the most important thing to do is to present the data and engage in shared decision-making with the patient,” noted Dr. Mostaghimi, who is also director of the inpatient dermatology consult service at Brigham and Women’s Hospital, Boston.
Surprising effectiveness
Going into the study, “we felt it would be likely that majority of nutritional supplements would either not be effective or not studied,” he said.
Dr. Mostaghimi and his coauthors conducted the study because so many patients take nutritional supplements to address hair loss, he said. An initial literature survey yielded more than 6,300 citations, but after screening and reviews, the authors included 30 articles for evaluation.
The review begins with a look at studies of saw palmetto (Serenoa repens), a botanical compound thought to inhibit the enzyme 5-alpha reductase (5AR), which converts testosterone to dihydroxytestosterone (DHT). DHT is a mediator of androgenic alopecia (AGA). The studies suggest that the compound might stabilize hair loss, “although its effect is likely less than that of finasteride,” write the authors. They also note that side effects associated with finasteride, such as sexual dysfunction, were also observed with saw palmetto “but to a lesser extent.”
For AGA, pumpkin seed oil may also be effective and a “potential alternative” to finasteride for AGA, and Forti5, a nutritional supplement that includes botanical 5AR inhibitors and other ingredients, had favorable effects in one study, the authors write. But neither has been compared to finasteride, and the Forti5 study lacked a control group.
The review also examines the micronutrients vitamin D, zinc, B vitamins, and antioxidants. Low levels of vitamin D have been associated with alopecia areata (AA), AGA, and telogen effluvium (TE) in some studies, and zinc deficiencies have been associated with TE, hair breakage, and thinning, according to the review. A single-arm vitamin D study showed improved results at 6 months for women with TE, but there was no control group and TE is self-resolving, the authors add. Studies in patients with normal zinc levels at baseline who had AA or hair loss showed significant hair regrowth and increased hair thickness and density, but the trials were a mishmash of controls and no controls and relied on self-perceived hair-loss data.
Larger more rigorous studies should be done to evaluate zinc’s effectiveness with AA, the authors comment.
Many patients take vitamin B7 (biotin) for hair loss. It has not been studied on its own but was an ingredient in some supplements in the review. Dr. Simmons-O’Brien said that biotin won’t result in new hair growth but that it can help strengthen the new hairs that grow as a result of other therapies. Both she and the study authors note that the Food and Drug Administration has warned against biotin supplementation because it can interfere with troponin and other test results.
The review also finds that immunomodulators –such as Chinese herbal extracts from paeony and glycyrrhizin – were effective in severe AA. Growth hormone modulators targeting deficiencies in insulin growth factor 1 or growth hormone are also promising. Studies of the modulators capsaicin and isoflavones – used topically – spurred hair growth, the authors write.
Products containing marine protein supplements, including Viviscal and Nourkrin, appeared effective in increasing hair counts in men and women, but the studies were funded by the manufacturer and were not well controlled. Side effects with Viviscal included bloating, according to the review.
The multi-ingredient supplements Nutrafol, Omni-Three, Apple Nutraceutical, and Lambdapil were also included in the review. Only Omni-Three showed no effectiveness, but studies of the other supplements had various limitations, including lack of controls and small sample sizes.
Complicated problem, multiple solutions
Given the many reasons for hair loss, multiple solutions are needed, the dermatologists note.
Dr. Mostaghimi said that he’s still a bit skeptical that supplements work as consistently as described or as well as described, given that he and his coauthors were unable to find any negative studies. In talking with patients who are taking supplements, he said that his first aim is to make sure they are safe. At least the supplements in the review have been studied for safety, he added.
He will encourage replacement of vitamin D or zinc or other vitamins or minerals if patients are deficient but said that he does not “actively encourage supplementation.”
Dr. Simmons-O’Brien said that, when evaluating patients with hair loss, she orders lab tests to determine whether the patient has anemia or a thyroid issue or deficiencies in vitamins or minerals or other nutritional deficiencies, asks about diet and styling practices, and takes a scalp biopsy. It is not uncommon to recommend supplementation on the basis of those findings, she added.
“As a hair-loss specialist, my job is to treat the patient at their level, in their framework, in their comfort zone,” said Dr. Goldberg. Some patients don’t want to take medications for hair loss, so she might recommend supplements in those cases but tells patients that they aren’t well studied.
She added that it can be hard to tell whether a supplement is working, particularly if it has multiple ingredients.
Dr. Mostaghimi reported consulting fees from Pfizer, Concert, Lilly, Hims and Hers, Equillium, AbbVie, Digital Diagnostics, and Bioniz and grants from Pfizer, all outside the submitted work. In addition, Dr. Mostaghimi disclosed that he is an associate editor of JAMA Dermatology but was not involved in any of the decisions regarding the review of the manuscript or its acceptance. No other disclosures were reported by the other study authors. Dr. Goldberg reported no disclosures. Dr. Simmons-O›Brien is a medical consultant for Isdin, but not for hair products.
A version of this article first appeared on Medscape.com.
because of small sample sizes, heterogeneity of hair loss types in study subjects, or other limitations.
The review, published online in JAMA Dermatology, notes that “Twelve of the 20 nutritional interventions had high-quality studies suggesting objectively evaluated effectiveness.”
It is “ground breaking,” in part because of its breadth and depth, said Eva Simmons-O’Brien, MD, a dermatologist in Towson, Md., who often recommends supplements for her patients with hair loss. “It basically kind of vindicates what some of us have been doing for a number of years in terms of treating hair loss,” she told this news organization. “It should hopefully make it more commonplace for dermatologists to consider using nutritional supplements as an adjuvant to treating hair loss,” added Dr. Simmons-O’Brien.
The review “is very helpful,” agreed Lynne J. Goldberg, MD, professor of dermatology and pathology and laboratory medicine at Boston University. Dr. Goldberg noted that many patients are already taking supplements and want to know whether they are safe and effective. The review “points out what the problems are; it talks about what the individual ingredients are and what they do, what the problems are; and it concluded that some people may find these helpful. Which is exactly what I tell my patients,” said Dr. Goldberg, who is also director of the Hair Clinic at Boston Medical Center.
“For patients who are highly motivated and eager to try this, we’re hoping that this systematic review serves as a foundation to have a conversation,” study coauthor Arash Mostaghimi, MD, MPA, MPH, of the department of dermatology at Harvard Medical School, told this news organization. “When there’s medical uncertainty and the question is how much risk is one willing to take, the most important thing to do is to present the data and engage in shared decision-making with the patient,” noted Dr. Mostaghimi, who is also director of the inpatient dermatology consult service at Brigham and Women’s Hospital, Boston.
Surprising effectiveness
Going into the study, “we felt it would be likely that majority of nutritional supplements would either not be effective or not studied,” he said.
Dr. Mostaghimi and his coauthors conducted the study because so many patients take nutritional supplements to address hair loss, he said. An initial literature survey yielded more than 6,300 citations, but after screening and reviews, the authors included 30 articles for evaluation.
The review begins with a look at studies of saw palmetto (Serenoa repens), a botanical compound thought to inhibit the enzyme 5-alpha reductase (5AR), which converts testosterone to dihydroxytestosterone (DHT). DHT is a mediator of androgenic alopecia (AGA). The studies suggest that the compound might stabilize hair loss, “although its effect is likely less than that of finasteride,” write the authors. They also note that side effects associated with finasteride, such as sexual dysfunction, were also observed with saw palmetto “but to a lesser extent.”
For AGA, pumpkin seed oil may also be effective and a “potential alternative” to finasteride for AGA, and Forti5, a nutritional supplement that includes botanical 5AR inhibitors and other ingredients, had favorable effects in one study, the authors write. But neither has been compared to finasteride, and the Forti5 study lacked a control group.
The review also examines the micronutrients vitamin D, zinc, B vitamins, and antioxidants. Low levels of vitamin D have been associated with alopecia areata (AA), AGA, and telogen effluvium (TE) in some studies, and zinc deficiencies have been associated with TE, hair breakage, and thinning, according to the review. A single-arm vitamin D study showed improved results at 6 months for women with TE, but there was no control group and TE is self-resolving, the authors add. Studies in patients with normal zinc levels at baseline who had AA or hair loss showed significant hair regrowth and increased hair thickness and density, but the trials were a mishmash of controls and no controls and relied on self-perceived hair-loss data.
Larger more rigorous studies should be done to evaluate zinc’s effectiveness with AA, the authors comment.
Many patients take vitamin B7 (biotin) for hair loss. It has not been studied on its own but was an ingredient in some supplements in the review. Dr. Simmons-O’Brien said that biotin won’t result in new hair growth but that it can help strengthen the new hairs that grow as a result of other therapies. Both she and the study authors note that the Food and Drug Administration has warned against biotin supplementation because it can interfere with troponin and other test results.
The review also finds that immunomodulators –such as Chinese herbal extracts from paeony and glycyrrhizin – were effective in severe AA. Growth hormone modulators targeting deficiencies in insulin growth factor 1 or growth hormone are also promising. Studies of the modulators capsaicin and isoflavones – used topically – spurred hair growth, the authors write.
Products containing marine protein supplements, including Viviscal and Nourkrin, appeared effective in increasing hair counts in men and women, but the studies were funded by the manufacturer and were not well controlled. Side effects with Viviscal included bloating, according to the review.
The multi-ingredient supplements Nutrafol, Omni-Three, Apple Nutraceutical, and Lambdapil were also included in the review. Only Omni-Three showed no effectiveness, but studies of the other supplements had various limitations, including lack of controls and small sample sizes.
Complicated problem, multiple solutions
Given the many reasons for hair loss, multiple solutions are needed, the dermatologists note.
Dr. Mostaghimi said that he’s still a bit skeptical that supplements work as consistently as described or as well as described, given that he and his coauthors were unable to find any negative studies. In talking with patients who are taking supplements, he said that his first aim is to make sure they are safe. At least the supplements in the review have been studied for safety, he added.
He will encourage replacement of vitamin D or zinc or other vitamins or minerals if patients are deficient but said that he does not “actively encourage supplementation.”
Dr. Simmons-O’Brien said that, when evaluating patients with hair loss, she orders lab tests to determine whether the patient has anemia or a thyroid issue or deficiencies in vitamins or minerals or other nutritional deficiencies, asks about diet and styling practices, and takes a scalp biopsy. It is not uncommon to recommend supplementation on the basis of those findings, she added.
“As a hair-loss specialist, my job is to treat the patient at their level, in their framework, in their comfort zone,” said Dr. Goldberg. Some patients don’t want to take medications for hair loss, so she might recommend supplements in those cases but tells patients that they aren’t well studied.
She added that it can be hard to tell whether a supplement is working, particularly if it has multiple ingredients.
Dr. Mostaghimi reported consulting fees from Pfizer, Concert, Lilly, Hims and Hers, Equillium, AbbVie, Digital Diagnostics, and Bioniz and grants from Pfizer, all outside the submitted work. In addition, Dr. Mostaghimi disclosed that he is an associate editor of JAMA Dermatology but was not involved in any of the decisions regarding the review of the manuscript or its acceptance. No other disclosures were reported by the other study authors. Dr. Goldberg reported no disclosures. Dr. Simmons-O›Brien is a medical consultant for Isdin, but not for hair products.
A version of this article first appeared on Medscape.com.
because of small sample sizes, heterogeneity of hair loss types in study subjects, or other limitations.
The review, published online in JAMA Dermatology, notes that “Twelve of the 20 nutritional interventions had high-quality studies suggesting objectively evaluated effectiveness.”
It is “ground breaking,” in part because of its breadth and depth, said Eva Simmons-O’Brien, MD, a dermatologist in Towson, Md., who often recommends supplements for her patients with hair loss. “It basically kind of vindicates what some of us have been doing for a number of years in terms of treating hair loss,” she told this news organization. “It should hopefully make it more commonplace for dermatologists to consider using nutritional supplements as an adjuvant to treating hair loss,” added Dr. Simmons-O’Brien.
The review “is very helpful,” agreed Lynne J. Goldberg, MD, professor of dermatology and pathology and laboratory medicine at Boston University. Dr. Goldberg noted that many patients are already taking supplements and want to know whether they are safe and effective. The review “points out what the problems are; it talks about what the individual ingredients are and what they do, what the problems are; and it concluded that some people may find these helpful. Which is exactly what I tell my patients,” said Dr. Goldberg, who is also director of the Hair Clinic at Boston Medical Center.
“For patients who are highly motivated and eager to try this, we’re hoping that this systematic review serves as a foundation to have a conversation,” study coauthor Arash Mostaghimi, MD, MPA, MPH, of the department of dermatology at Harvard Medical School, told this news organization. “When there’s medical uncertainty and the question is how much risk is one willing to take, the most important thing to do is to present the data and engage in shared decision-making with the patient,” noted Dr. Mostaghimi, who is also director of the inpatient dermatology consult service at Brigham and Women’s Hospital, Boston.
Surprising effectiveness
Going into the study, “we felt it would be likely that majority of nutritional supplements would either not be effective or not studied,” he said.
Dr. Mostaghimi and his coauthors conducted the study because so many patients take nutritional supplements to address hair loss, he said. An initial literature survey yielded more than 6,300 citations, but after screening and reviews, the authors included 30 articles for evaluation.
The review begins with a look at studies of saw palmetto (Serenoa repens), a botanical compound thought to inhibit the enzyme 5-alpha reductase (5AR), which converts testosterone to dihydroxytestosterone (DHT). DHT is a mediator of androgenic alopecia (AGA). The studies suggest that the compound might stabilize hair loss, “although its effect is likely less than that of finasteride,” write the authors. They also note that side effects associated with finasteride, such as sexual dysfunction, were also observed with saw palmetto “but to a lesser extent.”
For AGA, pumpkin seed oil may also be effective and a “potential alternative” to finasteride for AGA, and Forti5, a nutritional supplement that includes botanical 5AR inhibitors and other ingredients, had favorable effects in one study, the authors write. But neither has been compared to finasteride, and the Forti5 study lacked a control group.
The review also examines the micronutrients vitamin D, zinc, B vitamins, and antioxidants. Low levels of vitamin D have been associated with alopecia areata (AA), AGA, and telogen effluvium (TE) in some studies, and zinc deficiencies have been associated with TE, hair breakage, and thinning, according to the review. A single-arm vitamin D study showed improved results at 6 months for women with TE, but there was no control group and TE is self-resolving, the authors add. Studies in patients with normal zinc levels at baseline who had AA or hair loss showed significant hair regrowth and increased hair thickness and density, but the trials were a mishmash of controls and no controls and relied on self-perceived hair-loss data.
Larger more rigorous studies should be done to evaluate zinc’s effectiveness with AA, the authors comment.
Many patients take vitamin B7 (biotin) for hair loss. It has not been studied on its own but was an ingredient in some supplements in the review. Dr. Simmons-O’Brien said that biotin won’t result in new hair growth but that it can help strengthen the new hairs that grow as a result of other therapies. Both she and the study authors note that the Food and Drug Administration has warned against biotin supplementation because it can interfere with troponin and other test results.
The review also finds that immunomodulators –such as Chinese herbal extracts from paeony and glycyrrhizin – were effective in severe AA. Growth hormone modulators targeting deficiencies in insulin growth factor 1 or growth hormone are also promising. Studies of the modulators capsaicin and isoflavones – used topically – spurred hair growth, the authors write.
Products containing marine protein supplements, including Viviscal and Nourkrin, appeared effective in increasing hair counts in men and women, but the studies were funded by the manufacturer and were not well controlled. Side effects with Viviscal included bloating, according to the review.
The multi-ingredient supplements Nutrafol, Omni-Three, Apple Nutraceutical, and Lambdapil were also included in the review. Only Omni-Three showed no effectiveness, but studies of the other supplements had various limitations, including lack of controls and small sample sizes.
Complicated problem, multiple solutions
Given the many reasons for hair loss, multiple solutions are needed, the dermatologists note.
Dr. Mostaghimi said that he’s still a bit skeptical that supplements work as consistently as described or as well as described, given that he and his coauthors were unable to find any negative studies. In talking with patients who are taking supplements, he said that his first aim is to make sure they are safe. At least the supplements in the review have been studied for safety, he added.
He will encourage replacement of vitamin D or zinc or other vitamins or minerals if patients are deficient but said that he does not “actively encourage supplementation.”
Dr. Simmons-O’Brien said that, when evaluating patients with hair loss, she orders lab tests to determine whether the patient has anemia or a thyroid issue or deficiencies in vitamins or minerals or other nutritional deficiencies, asks about diet and styling practices, and takes a scalp biopsy. It is not uncommon to recommend supplementation on the basis of those findings, she added.
“As a hair-loss specialist, my job is to treat the patient at their level, in their framework, in their comfort zone,” said Dr. Goldberg. Some patients don’t want to take medications for hair loss, so she might recommend supplements in those cases but tells patients that they aren’t well studied.
She added that it can be hard to tell whether a supplement is working, particularly if it has multiple ingredients.
Dr. Mostaghimi reported consulting fees from Pfizer, Concert, Lilly, Hims and Hers, Equillium, AbbVie, Digital Diagnostics, and Bioniz and grants from Pfizer, all outside the submitted work. In addition, Dr. Mostaghimi disclosed that he is an associate editor of JAMA Dermatology but was not involved in any of the decisions regarding the review of the manuscript or its acceptance. No other disclosures were reported by the other study authors. Dr. Goldberg reported no disclosures. Dr. Simmons-O›Brien is a medical consultant for Isdin, but not for hair products.
A version of this article first appeared on Medscape.com.
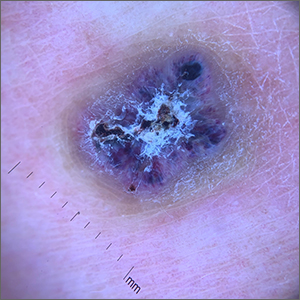
Knee lesion that bleeds
This combination of vascular features with excess keratin fit perfectly with the name of the diagnosis: angiokeratoma. The dark color of the lesion on magnification, or in this case with dermoscopy, showed the lacunar pattern of dilated vessels. The overlying keratin was likely accentuated because it was on an extensor surface; the rim of hyperpigmentation is common for these lesions.
Angiokeratomas result from dilation of the blood vessels underneath the epidermis. There are different inciting events that lead to the 5 different types of angiokeratomas. The overlying epidermal changes are secondary to the underlying process of capillary ectasia.1 This lesion was not part of a cluster, so it was characterized as a solitary angiokeratoma. Smaller lesions are usually less keratinized and are commonly seen on the scrotum and vulva, where there are usually multiple lesions (referred to as angiokeratoma of Fordyce).
Zaballos2 studied the dermoscopic characteristics of 32 solitary angiokeratomas and reported 6 findings in at least half of the solitary lesions. The most common features were dark lacunae in 94% of the lesions, white veil in 91%, and erythema in 69%. Peripheral erythema, red lacunae, and hemorrhagic crusts were all seen at a rate of 53%. The most common location was the lower extremities.
This patient’s previous pathology report from a shave biopsy was found, confirming that the original diagnosis was angiokeratoma. Since the patient’s lesion had not resolved and was symptomatic from minor trauma, he was scheduled to come back in for an elliptical excision to remove the lesion.
Image and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Schiller PI, Itin PH. Angiokeratomas: an update. Dermatology. 1996;193:275-282. doi: 10.1159/000246270
2. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318–325. doi:10.1001/archderm.143.3.318

This combination of vascular features with excess keratin fit perfectly with the name of the diagnosis: angiokeratoma. The dark color of the lesion on magnification, or in this case with dermoscopy, showed the lacunar pattern of dilated vessels. The overlying keratin was likely accentuated because it was on an extensor surface; the rim of hyperpigmentation is common for these lesions.
Angiokeratomas result from dilation of the blood vessels underneath the epidermis. There are different inciting events that lead to the 5 different types of angiokeratomas. The overlying epidermal changes are secondary to the underlying process of capillary ectasia.1 This lesion was not part of a cluster, so it was characterized as a solitary angiokeratoma. Smaller lesions are usually less keratinized and are commonly seen on the scrotum and vulva, where there are usually multiple lesions (referred to as angiokeratoma of Fordyce).
Zaballos2 studied the dermoscopic characteristics of 32 solitary angiokeratomas and reported 6 findings in at least half of the solitary lesions. The most common features were dark lacunae in 94% of the lesions, white veil in 91%, and erythema in 69%. Peripheral erythema, red lacunae, and hemorrhagic crusts were all seen at a rate of 53%. The most common location was the lower extremities.
This patient’s previous pathology report from a shave biopsy was found, confirming that the original diagnosis was angiokeratoma. Since the patient’s lesion had not resolved and was symptomatic from minor trauma, he was scheduled to come back in for an elliptical excision to remove the lesion.
Image and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

This combination of vascular features with excess keratin fit perfectly with the name of the diagnosis: angiokeratoma. The dark color of the lesion on magnification, or in this case with dermoscopy, showed the lacunar pattern of dilated vessels. The overlying keratin was likely accentuated because it was on an extensor surface; the rim of hyperpigmentation is common for these lesions.
Angiokeratomas result from dilation of the blood vessels underneath the epidermis. There are different inciting events that lead to the 5 different types of angiokeratomas. The overlying epidermal changes are secondary to the underlying process of capillary ectasia.1 This lesion was not part of a cluster, so it was characterized as a solitary angiokeratoma. Smaller lesions are usually less keratinized and are commonly seen on the scrotum and vulva, where there are usually multiple lesions (referred to as angiokeratoma of Fordyce).
Zaballos2 studied the dermoscopic characteristics of 32 solitary angiokeratomas and reported 6 findings in at least half of the solitary lesions. The most common features were dark lacunae in 94% of the lesions, white veil in 91%, and erythema in 69%. Peripheral erythema, red lacunae, and hemorrhagic crusts were all seen at a rate of 53%. The most common location was the lower extremities.
This patient’s previous pathology report from a shave biopsy was found, confirming that the original diagnosis was angiokeratoma. Since the patient’s lesion had not resolved and was symptomatic from minor trauma, he was scheduled to come back in for an elliptical excision to remove the lesion.
Image and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Schiller PI, Itin PH. Angiokeratomas: an update. Dermatology. 1996;193:275-282. doi: 10.1159/000246270
2. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318–325. doi:10.1001/archderm.143.3.318
1. Schiller PI, Itin PH. Angiokeratomas: an update. Dermatology. 1996;193:275-282. doi: 10.1159/000246270
2. Zaballos P, Daufí C, Puig S, et al. Dermoscopy of solitary angiokeratomas: a morphological study. Arch Dermatol. 2007;143:318–325. doi:10.1001/archderm.143.3.318
New melting hydrogel bandage could treat burn wounds faster, with less pain
Surgically debriding burn wounds can be tedious for doctors and excruciating for patients. To change that, bioengineers have created a new hydrogel formula that dissolves rapidly from wound sites, melting off in 6 minutes or less.
“The removal of dressings, with the current standard of care, is very hard and time-consuming. It becomes very painful for the patient. People are screaming, or they’re given a lot of opioids,” said senior author O. Berk Usta, PhD, of the Center for Engineering in Medicine and Surgery at Massachusetts General Hospital, Boston. “Those are the things we wanted to minimize: the pain and the time.”
Although beneficial for all patients, a short, painless bandage change would be a particular boon for younger patients. At the pediatric burns care center at Shriners Hospitals for Children (an MGH partner), researchers “observe a lot of children who go through therapy or treatment after burns,” said Dr. Usta. The team at MGH collaborated with scientists at Tufts University, Boston, with those patients in mind, setting out to create a new hydrogel that would transform burn wound care.
A better bandage
Hydrogels provide cooling relief to burn wounds and maintain a moist environment that can speed healing. There are currently hydrogel sheets and hydrogel-infused dressings, as well as gel that is applied directly to burn wounds before being covered with protective material. These dressings must be replaced frequently to prevent infections, but that can be unbearably painful and drawn out, as dressings often stick to wounds.
Mechanical debridement can be especially difficult for second-degree burn patients, whose wounds may still retain nerve endings. Debridement tends to also remove some healthy tissue and can damage newly formed tissue, slowing down healing.
“It can take up to 2, 3 hours, and it requires multiple people working on it,” said Dr. Usta.
The new hydrogel treatment can be applied directly to a wound and it forms a protective barrier around the site in 15 seconds. The hydrogel is then covered by a protective dressing until it needs to be changed.
“After you take off the protective covering, you add another solution, which dissolves the [hydrogel] dressing, so that it can be easily removed from the burn site,” Dr. Usta said.
The solution dissolves the hydrogel in 4-6 minutes.
Hybrid gels
Many hydrogels currently used for burn wounds feature physically cross-linked molecules. This makes them strong and capable of retaining moisture, but also difficult to dissolve. The researchers used a different approach.
“This is not physical cross-linking like the traditional approaches, but rather, softer covalent bonds between the different molecules. And that’s why, when you bring in another solution, the hydrogel dissolves away,” Dr. Usta said.
The new hydrogels rely on a supramolecular assembly: a network of synthetic polymers whose connections can be reversed more easily, meaning they can be dissolved quickly. Another standout feature of the new hydrogels is their hybrid composition, displaying characteristics of both liquids and solids. The polymers are knitted together into a mesh-like network that enables water retention, with the goal of maintaining the moist environment needed for wound healing.
The supramolecular assembly is also greener, Dr. Usta explained; traditional cross-linking approaches produce a lot of toxic by-products that could harm the environment.
And whereas traditional hydrogels can require a dozen chemistry steps to produce, the new hydrogels are ready after mixing two solutions, Dr. Usta explained. This makes them easy to prepare at bedside, ideal for treating large wounds in the ER or even on battlefields.
When tested in vitro, using skin cells, and in vivo, on mice, the new hydrogels were shown to be safe to use on wounds. Additional studies on mice, as well as large animals, will focus on safety and efficacy, and may be followed by human clinical trials, said Dr. Usta.
“The next phase of the project will be to look at whether these dressings will help wound healing by creating a moist environment,” said Dr. Usta.
The researchers are also exploring how to manufacture individual prewrapped hydrogels that could be applied in a clinical setting – or even in people’s homes. The consumer market is “another possibility,” said Dr. Usta, particularly among patients with “smaller, more superficial burns” or patients whose large burn wounds are still healing once they leave the hospital.
This research was supported by the National Institutes of Health, National Science Foundation, Massachusetts General Hospital Executive Committee on Research Interim Support Fund, and Shriners Hospitals.
A version of this article first appeared on Medscape.com.
Surgically debriding burn wounds can be tedious for doctors and excruciating for patients. To change that, bioengineers have created a new hydrogel formula that dissolves rapidly from wound sites, melting off in 6 minutes or less.
“The removal of dressings, with the current standard of care, is very hard and time-consuming. It becomes very painful for the patient. People are screaming, or they’re given a lot of opioids,” said senior author O. Berk Usta, PhD, of the Center for Engineering in Medicine and Surgery at Massachusetts General Hospital, Boston. “Those are the things we wanted to minimize: the pain and the time.”
Although beneficial for all patients, a short, painless bandage change would be a particular boon for younger patients. At the pediatric burns care center at Shriners Hospitals for Children (an MGH partner), researchers “observe a lot of children who go through therapy or treatment after burns,” said Dr. Usta. The team at MGH collaborated with scientists at Tufts University, Boston, with those patients in mind, setting out to create a new hydrogel that would transform burn wound care.
A better bandage
Hydrogels provide cooling relief to burn wounds and maintain a moist environment that can speed healing. There are currently hydrogel sheets and hydrogel-infused dressings, as well as gel that is applied directly to burn wounds before being covered with protective material. These dressings must be replaced frequently to prevent infections, but that can be unbearably painful and drawn out, as dressings often stick to wounds.
Mechanical debridement can be especially difficult for second-degree burn patients, whose wounds may still retain nerve endings. Debridement tends to also remove some healthy tissue and can damage newly formed tissue, slowing down healing.
“It can take up to 2, 3 hours, and it requires multiple people working on it,” said Dr. Usta.
The new hydrogel treatment can be applied directly to a wound and it forms a protective barrier around the site in 15 seconds. The hydrogel is then covered by a protective dressing until it needs to be changed.
“After you take off the protective covering, you add another solution, which dissolves the [hydrogel] dressing, so that it can be easily removed from the burn site,” Dr. Usta said.
The solution dissolves the hydrogel in 4-6 minutes.
Hybrid gels
Many hydrogels currently used for burn wounds feature physically cross-linked molecules. This makes them strong and capable of retaining moisture, but also difficult to dissolve. The researchers used a different approach.
“This is not physical cross-linking like the traditional approaches, but rather, softer covalent bonds between the different molecules. And that’s why, when you bring in another solution, the hydrogel dissolves away,” Dr. Usta said.
The new hydrogels rely on a supramolecular assembly: a network of synthetic polymers whose connections can be reversed more easily, meaning they can be dissolved quickly. Another standout feature of the new hydrogels is their hybrid composition, displaying characteristics of both liquids and solids. The polymers are knitted together into a mesh-like network that enables water retention, with the goal of maintaining the moist environment needed for wound healing.
The supramolecular assembly is also greener, Dr. Usta explained; traditional cross-linking approaches produce a lot of toxic by-products that could harm the environment.
And whereas traditional hydrogels can require a dozen chemistry steps to produce, the new hydrogels are ready after mixing two solutions, Dr. Usta explained. This makes them easy to prepare at bedside, ideal for treating large wounds in the ER or even on battlefields.
When tested in vitro, using skin cells, and in vivo, on mice, the new hydrogels were shown to be safe to use on wounds. Additional studies on mice, as well as large animals, will focus on safety and efficacy, and may be followed by human clinical trials, said Dr. Usta.
“The next phase of the project will be to look at whether these dressings will help wound healing by creating a moist environment,” said Dr. Usta.
The researchers are also exploring how to manufacture individual prewrapped hydrogels that could be applied in a clinical setting – or even in people’s homes. The consumer market is “another possibility,” said Dr. Usta, particularly among patients with “smaller, more superficial burns” or patients whose large burn wounds are still healing once they leave the hospital.
This research was supported by the National Institutes of Health, National Science Foundation, Massachusetts General Hospital Executive Committee on Research Interim Support Fund, and Shriners Hospitals.
A version of this article first appeared on Medscape.com.
Surgically debriding burn wounds can be tedious for doctors and excruciating for patients. To change that, bioengineers have created a new hydrogel formula that dissolves rapidly from wound sites, melting off in 6 minutes or less.
“The removal of dressings, with the current standard of care, is very hard and time-consuming. It becomes very painful for the patient. People are screaming, or they’re given a lot of opioids,” said senior author O. Berk Usta, PhD, of the Center for Engineering in Medicine and Surgery at Massachusetts General Hospital, Boston. “Those are the things we wanted to minimize: the pain and the time.”
Although beneficial for all patients, a short, painless bandage change would be a particular boon for younger patients. At the pediatric burns care center at Shriners Hospitals for Children (an MGH partner), researchers “observe a lot of children who go through therapy or treatment after burns,” said Dr. Usta. The team at MGH collaborated with scientists at Tufts University, Boston, with those patients in mind, setting out to create a new hydrogel that would transform burn wound care.
A better bandage
Hydrogels provide cooling relief to burn wounds and maintain a moist environment that can speed healing. There are currently hydrogel sheets and hydrogel-infused dressings, as well as gel that is applied directly to burn wounds before being covered with protective material. These dressings must be replaced frequently to prevent infections, but that can be unbearably painful and drawn out, as dressings often stick to wounds.
Mechanical debridement can be especially difficult for second-degree burn patients, whose wounds may still retain nerve endings. Debridement tends to also remove some healthy tissue and can damage newly formed tissue, slowing down healing.
“It can take up to 2, 3 hours, and it requires multiple people working on it,” said Dr. Usta.
The new hydrogel treatment can be applied directly to a wound and it forms a protective barrier around the site in 15 seconds. The hydrogel is then covered by a protective dressing until it needs to be changed.
“After you take off the protective covering, you add another solution, which dissolves the [hydrogel] dressing, so that it can be easily removed from the burn site,” Dr. Usta said.
The solution dissolves the hydrogel in 4-6 minutes.
Hybrid gels
Many hydrogels currently used for burn wounds feature physically cross-linked molecules. This makes them strong and capable of retaining moisture, but also difficult to dissolve. The researchers used a different approach.
“This is not physical cross-linking like the traditional approaches, but rather, softer covalent bonds between the different molecules. And that’s why, when you bring in another solution, the hydrogel dissolves away,” Dr. Usta said.
The new hydrogels rely on a supramolecular assembly: a network of synthetic polymers whose connections can be reversed more easily, meaning they can be dissolved quickly. Another standout feature of the new hydrogels is their hybrid composition, displaying characteristics of both liquids and solids. The polymers are knitted together into a mesh-like network that enables water retention, with the goal of maintaining the moist environment needed for wound healing.
The supramolecular assembly is also greener, Dr. Usta explained; traditional cross-linking approaches produce a lot of toxic by-products that could harm the environment.
And whereas traditional hydrogels can require a dozen chemistry steps to produce, the new hydrogels are ready after mixing two solutions, Dr. Usta explained. This makes them easy to prepare at bedside, ideal for treating large wounds in the ER or even on battlefields.
When tested in vitro, using skin cells, and in vivo, on mice, the new hydrogels were shown to be safe to use on wounds. Additional studies on mice, as well as large animals, will focus on safety and efficacy, and may be followed by human clinical trials, said Dr. Usta.
“The next phase of the project will be to look at whether these dressings will help wound healing by creating a moist environment,” said Dr. Usta.
The researchers are also exploring how to manufacture individual prewrapped hydrogels that could be applied in a clinical setting – or even in people’s homes. The consumer market is “another possibility,” said Dr. Usta, particularly among patients with “smaller, more superficial burns” or patients whose large burn wounds are still healing once they leave the hospital.
This research was supported by the National Institutes of Health, National Science Foundation, Massachusetts General Hospital Executive Committee on Research Interim Support Fund, and Shriners Hospitals.
A version of this article first appeared on Medscape.com.
FROM BIOACTIVE MATERIALS
Consider quality of life, comorbidities in hidradenitis suppurativa
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Pandemic caused treatment delay for half of patients with CTCL, study finds
showed. However, among patients with CTCL diagnosed with COVID-19 during that time, no cases were acquired from outpatient visits.
“Delays in therapy for patients with cutaneous lymphomas should likely be avoided,” two of the study authors, Larisa J. Geskin, MD, of the department of dermatology at Columbia University Irving Medical Center, New York, and Bradley D. Kwinta, a medical student at Columbia University, told this news organization in a combined response via email.
“Continuing treatment and maintenance therapy appears critical to avoiding disease progression, highlighting the importance of maintenance therapy in CTCL,” they said. “These patients can be safely treated according to established treatment protocols while practicing physical distancing and using personal protective equipment without significantly increasing their risk of COVID-19 infection.”
The United States Cutaneous Lymphoma Consortium and the European Organization for Research and Treatment of Cancer developed emergency guidelines for the management of patients with cutaneous lymphomas during the pandemic to ensure patient safety, and the International Society for Cutaneous Lymphomas created an International Cutaneous Lymphomas Pandemic Section to collect data to assess the impact of these guidelines.
“Using this data, we can determine if these measures were effective in preventing COVID-19 infection, what the impact was of maintenance therapy, and how delays in treatment affected disease outcomes in CTCL patients,” the authors and their colleagues wrote in the study, which was published in the Journal of the American Academy of Dermatology.
They retrospectively analyzed data from the electronic medical records of 149 patients with CTCL who were being managed at one of nine international academic medical centers in seven countries from March to October 2020. Slightly more than half (56%) were male, 70% were White, 18% were Black, 52% had stage IA-IIA disease, and 19% acquired COVID-19 during the study period.
Of the 149 patients, 79 (53%) experienced a mean treatment delay of 3.2 months (range, 10 days to 10 months). After adjusting for age, race, biological sex, COVID-19 status, and disease stage, treatment delay was associated with a significant risk of disease relapse or progression across all stages (odds ratio, 5.00; P < .001). Specifically, for each additional month that a patient experienced treatment delay, the odds of disease progression increased by 37% (OR, 1.37; P < .001).
A total of 28 patients with CTCL (19%) were diagnosed with COVID-19, but none were acquired from outpatient office visits. Patients who contracted COVID-19 did not have a statistically significant increase in odds of disease progression, compared with COVID-negative patients (OR, 0.41; P = .07).
According to Dr. Geskin, who is also director of the Comprehensive Skin Cancer Center in the division of cutaneous oncology in the department of dermatology at Columbia, and Mr. Kwinta, no clinical trials exist to inform maintenance protocols in patients with cutaneous lymphomas. “There are also no randomized and controlled observational studies that demonstrate the impact that therapy delay may have on disease outcomes,” they said in the email. “In fact, the need for maintenance therapy for CTCL is often debated. Our findings demonstrate the importance of continuing treatment and the use of maintenance therapy in avoiding disease progression in these incurable lymphomas.”
They acknowledged certain limitations of the study, including its retrospective observational design. “Therefore, we cannot establish a definitive causal link between treatment delay and disease progression,” they said. “Our cohort of patients were on various and often multiple therapies, making it hard to extrapolate our data to discern which maintenance therapies were most effective in preventing disease progression.”
In addition, their data only includes patients from March to October 2020, “before the discovery of new variants and the development of COVID-19 vaccines,” they added. “Additional studies would be required to draw conclusions on how COVID-19 vaccines may affect patients with CTCL, including outcomes in the setting of new variants.”
The authors reported having no financial disclosures.
showed. However, among patients with CTCL diagnosed with COVID-19 during that time, no cases were acquired from outpatient visits.
“Delays in therapy for patients with cutaneous lymphomas should likely be avoided,” two of the study authors, Larisa J. Geskin, MD, of the department of dermatology at Columbia University Irving Medical Center, New York, and Bradley D. Kwinta, a medical student at Columbia University, told this news organization in a combined response via email.
“Continuing treatment and maintenance therapy appears critical to avoiding disease progression, highlighting the importance of maintenance therapy in CTCL,” they said. “These patients can be safely treated according to established treatment protocols while practicing physical distancing and using personal protective equipment without significantly increasing their risk of COVID-19 infection.”
The United States Cutaneous Lymphoma Consortium and the European Organization for Research and Treatment of Cancer developed emergency guidelines for the management of patients with cutaneous lymphomas during the pandemic to ensure patient safety, and the International Society for Cutaneous Lymphomas created an International Cutaneous Lymphomas Pandemic Section to collect data to assess the impact of these guidelines.
“Using this data, we can determine if these measures were effective in preventing COVID-19 infection, what the impact was of maintenance therapy, and how delays in treatment affected disease outcomes in CTCL patients,” the authors and their colleagues wrote in the study, which was published in the Journal of the American Academy of Dermatology.
They retrospectively analyzed data from the electronic medical records of 149 patients with CTCL who were being managed at one of nine international academic medical centers in seven countries from March to October 2020. Slightly more than half (56%) were male, 70% were White, 18% were Black, 52% had stage IA-IIA disease, and 19% acquired COVID-19 during the study period.
Of the 149 patients, 79 (53%) experienced a mean treatment delay of 3.2 months (range, 10 days to 10 months). After adjusting for age, race, biological sex, COVID-19 status, and disease stage, treatment delay was associated with a significant risk of disease relapse or progression across all stages (odds ratio, 5.00; P < .001). Specifically, for each additional month that a patient experienced treatment delay, the odds of disease progression increased by 37% (OR, 1.37; P < .001).
A total of 28 patients with CTCL (19%) were diagnosed with COVID-19, but none were acquired from outpatient office visits. Patients who contracted COVID-19 did not have a statistically significant increase in odds of disease progression, compared with COVID-negative patients (OR, 0.41; P = .07).
According to Dr. Geskin, who is also director of the Comprehensive Skin Cancer Center in the division of cutaneous oncology in the department of dermatology at Columbia, and Mr. Kwinta, no clinical trials exist to inform maintenance protocols in patients with cutaneous lymphomas. “There are also no randomized and controlled observational studies that demonstrate the impact that therapy delay may have on disease outcomes,” they said in the email. “In fact, the need for maintenance therapy for CTCL is often debated. Our findings demonstrate the importance of continuing treatment and the use of maintenance therapy in avoiding disease progression in these incurable lymphomas.”
They acknowledged certain limitations of the study, including its retrospective observational design. “Therefore, we cannot establish a definitive causal link between treatment delay and disease progression,” they said. “Our cohort of patients were on various and often multiple therapies, making it hard to extrapolate our data to discern which maintenance therapies were most effective in preventing disease progression.”
In addition, their data only includes patients from March to October 2020, “before the discovery of new variants and the development of COVID-19 vaccines,” they added. “Additional studies would be required to draw conclusions on how COVID-19 vaccines may affect patients with CTCL, including outcomes in the setting of new variants.”
The authors reported having no financial disclosures.
showed. However, among patients with CTCL diagnosed with COVID-19 during that time, no cases were acquired from outpatient visits.
“Delays in therapy for patients with cutaneous lymphomas should likely be avoided,” two of the study authors, Larisa J. Geskin, MD, of the department of dermatology at Columbia University Irving Medical Center, New York, and Bradley D. Kwinta, a medical student at Columbia University, told this news organization in a combined response via email.
“Continuing treatment and maintenance therapy appears critical to avoiding disease progression, highlighting the importance of maintenance therapy in CTCL,” they said. “These patients can be safely treated according to established treatment protocols while practicing physical distancing and using personal protective equipment without significantly increasing their risk of COVID-19 infection.”
The United States Cutaneous Lymphoma Consortium and the European Organization for Research and Treatment of Cancer developed emergency guidelines for the management of patients with cutaneous lymphomas during the pandemic to ensure patient safety, and the International Society for Cutaneous Lymphomas created an International Cutaneous Lymphomas Pandemic Section to collect data to assess the impact of these guidelines.
“Using this data, we can determine if these measures were effective in preventing COVID-19 infection, what the impact was of maintenance therapy, and how delays in treatment affected disease outcomes in CTCL patients,” the authors and their colleagues wrote in the study, which was published in the Journal of the American Academy of Dermatology.
They retrospectively analyzed data from the electronic medical records of 149 patients with CTCL who were being managed at one of nine international academic medical centers in seven countries from March to October 2020. Slightly more than half (56%) were male, 70% were White, 18% were Black, 52% had stage IA-IIA disease, and 19% acquired COVID-19 during the study period.
Of the 149 patients, 79 (53%) experienced a mean treatment delay of 3.2 months (range, 10 days to 10 months). After adjusting for age, race, biological sex, COVID-19 status, and disease stage, treatment delay was associated with a significant risk of disease relapse or progression across all stages (odds ratio, 5.00; P < .001). Specifically, for each additional month that a patient experienced treatment delay, the odds of disease progression increased by 37% (OR, 1.37; P < .001).
A total of 28 patients with CTCL (19%) were diagnosed with COVID-19, but none were acquired from outpatient office visits. Patients who contracted COVID-19 did not have a statistically significant increase in odds of disease progression, compared with COVID-negative patients (OR, 0.41; P = .07).
According to Dr. Geskin, who is also director of the Comprehensive Skin Cancer Center in the division of cutaneous oncology in the department of dermatology at Columbia, and Mr. Kwinta, no clinical trials exist to inform maintenance protocols in patients with cutaneous lymphomas. “There are also no randomized and controlled observational studies that demonstrate the impact that therapy delay may have on disease outcomes,” they said in the email. “In fact, the need for maintenance therapy for CTCL is often debated. Our findings demonstrate the importance of continuing treatment and the use of maintenance therapy in avoiding disease progression in these incurable lymphomas.”
They acknowledged certain limitations of the study, including its retrospective observational design. “Therefore, we cannot establish a definitive causal link between treatment delay and disease progression,” they said. “Our cohort of patients were on various and often multiple therapies, making it hard to extrapolate our data to discern which maintenance therapies were most effective in preventing disease progression.”
In addition, their data only includes patients from March to October 2020, “before the discovery of new variants and the development of COVID-19 vaccines,” they added. “Additional studies would be required to draw conclusions on how COVID-19 vaccines may affect patients with CTCL, including outcomes in the setting of new variants.”
The authors reported having no financial disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
HS treated in EDs not followed up in derm clinics, study finds
according to a study of a large national administrative data set.
“Patients with HS presenting to the ED for their disease exhibited high rates of ED return with low rates of dermatology follow-up after an initial ED visit,” lead study author Cynthia X. Wang, MD, MPHS, and colleagues in the division of dermatology and the Institute for Informatics, Washington University in St. Louis, wrote in JAMA Dermatology.
Patients who received opioid prescriptions and patients on Medicaid were more likely to return to the ED, they noted.
HS, a debilitating skin disease involving chronic follicular inflammation, frequently affects the axilla, anogenital, and inframammary areas, with painful nodules, abscesses, and sinus tracts that can form scars, the authors wrote. HS is linked with comorbidities including inflammatory bowel disease, obesity, diabetes, substance use, and psychiatric conditions, and is often misdiagnosed for an estimated 7-10 years.
In the retrospective cohort study, Dr. Wang and colleagues collected data from nationwide commercial and Medicaid databases. They included patients aged 18 to 64 years with two or more claims for HS and with at least one ED visit not resulting in admission for HS or a defined proxy (such as a related diagnosis of folliculitis, in a location typical for HS) between 2010 and 2019.
The median age of the 20,269 patients in the study was 32 years; most (82.9%) were female, and nearly 37% had commercial insurance and 63.2% had Medicaid. About half the patients on Medicaid were Black and 36.2% were White (race and ethnicity data were not available for commercially insured patients). In both insurance groups, the rates of comorbidities were high, including 22.5% with obesity and 11.9% with diabetes.
The researchers found that, at the index ED visit, 48.0% of patients had incision and drainage performed (51% among those with commercial insurance vs. 46.3% of those with Medicaid; P < .001); 72.6% of patients filled an oral antibiotic prescription within 7 days, with similar percentages in both insurance groups; and 48.9% filled an oral opioid medication prescription within 7 days (46.5% for commercial insurance vs. 50.3% for Medicaid; P < .001).
Regarding follow-up care, the investigators found that 17.2% of patients had at least one return ED visit for HS or proxy within 30 days (15.7% for commercial vs. 18.1% for Medicaid; P < .001), while 2.4% had a dermatology visit (5.3% for commercial vs. 0.7% for Medicaid; P < .001). In addition, 34% of patients had at least one return ED visit for HS or proxy within 180 days (27.2% for commercial insurance vs. 38% for Medicaid; P < .001), while 6.8% had a dermatology visit (14.1% for commercial vs. 2.5% for Medicaid; P < .001).
Patients with an opioid prescribed within 7 days of the ED visit were more likely to return to the ED, within 30 days (odds ratio, 1.67; 95% confidence interval, 1.54-1.80; P < .001), and within 180 days (OR, 1.48; 95% CI, 1.39-1.58; P < .001). But they were less likely to have dermatology follow-up within 30 days (OR, 0.78; 95% CI, 0.64-0.95; P = .01) and within 180 days (OR, 0.81; 95% CI, 0.71-0.91; P < .001).
Medicaid patients were more likely to return to the ED within 30 days (OR, 1.12; 95% CI, 1.03-1.22; P = .009) and within 180 days (OR, 1.48; 95% CI, 1.38-1.58; P < .001). But they were less likely to receive outpatient dermatology follow-up care within 30 days (OR, 0.12; 95% CI, 0.09-0.15; P < .001) and within 180 days (OR, 0.16; 95% CI, 0.14-0.18; P < .001).
“This study highlights potential areas of action to improve care for patients with HS,” the authors concluded, including cross-specialty education and interventions, and focus on patients most likely to return to the ED for care.
Findings are not surprising
Christopher Sayed, MD, professor of dermatology at the University of North Carolina at Chapel Hill, said in an interview that the study results are expected. “Many patients have trouble establishing care with a dermatologist familiar with HS, so they seek more fragmented care at urgent cares and EDs,” he said.
“Some dermatologists are not familiar with HS or don’t accept insurance such as Medicaid,” Dr. Sayed added. “Many emergency room providers may not recognize that medical therapy for HS has evolved in a way that makes referral to a dermatologist more essential than ever. They may tell patients there is nothing else to do but return to the ED for the next flare. “Emergency medicine and dermatology training programs need to educate providers about appropriate long-term HS management.”
In an interview, Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital in New York and assistant professor of emergency medicine at Hofstra University, Hempstead, N.Y., said that the study describes a reality he and his colleagues know too well.
“The study gives a true snapshot of the disarray and inequality that exists for patients disproportionately affected by HS. Those who are African American and low income suffer from lack of HS primary dermatologic care and follow-up at much higher rates than do other demographic groups,” he said.
Doctors would like to see the current situation change, Dr. Glatter noted. “It’s frustrating for emergency physicians and for dermatologists, who know that optimal follow-up care for this chronic and disabling disease should be with a dermatologist (and other surgical specialists if necessary).
“It’s a broken system. Patients can’t get appointments in nonacademic private settings because the bulk of dermatologists will not accept Medicaid. And many academic practices will not see these patients, either,” he commented. “We end up becoming a safety net of care.”
Replace the broken system with an integrated process
A solution to address the problem would be to set up follow-up dermatology appointments when patients arrive in EDs during and after normal business hours, Dr. Glatter suggested. “Developing a coordinated, structured, streamlined process requires buy-in from all stakeholders, including private dermatologists, academic dermatology clinics, and the government.”
Having the Centers for Medicare & Medicaid Services study interventions for high utilizers of EDs for HS would also help with “the development of economic and logistical changes, including provider reimbursement and allocation of funds to address this ongoing disparity in care,” he added.
Ideally, larger health care systems could collaborate with academic and nonacademic dermatologists to design a referral network that cares for all uninsured or underinsured patients, he said. “Balancing patient care and improved outcomes – while working on a framework for reimbursement – would be in everyone’s best interest.”
The study was partially funded by a grant from the National Center for Advancing Translational Sciences of the National Institutes of Health. One author reported financial involvements with multiple pharmaceutical companies. Dr. Wang, the remaining coauthors, as well as Dr. Sayed and Dr. Glatter reported no conflicts of interest with the study. Dr. Glatter is an editor and columnist at Medscape.
according to a study of a large national administrative data set.
“Patients with HS presenting to the ED for their disease exhibited high rates of ED return with low rates of dermatology follow-up after an initial ED visit,” lead study author Cynthia X. Wang, MD, MPHS, and colleagues in the division of dermatology and the Institute for Informatics, Washington University in St. Louis, wrote in JAMA Dermatology.
Patients who received opioid prescriptions and patients on Medicaid were more likely to return to the ED, they noted.
HS, a debilitating skin disease involving chronic follicular inflammation, frequently affects the axilla, anogenital, and inframammary areas, with painful nodules, abscesses, and sinus tracts that can form scars, the authors wrote. HS is linked with comorbidities including inflammatory bowel disease, obesity, diabetes, substance use, and psychiatric conditions, and is often misdiagnosed for an estimated 7-10 years.
In the retrospective cohort study, Dr. Wang and colleagues collected data from nationwide commercial and Medicaid databases. They included patients aged 18 to 64 years with two or more claims for HS and with at least one ED visit not resulting in admission for HS or a defined proxy (such as a related diagnosis of folliculitis, in a location typical for HS) between 2010 and 2019.
The median age of the 20,269 patients in the study was 32 years; most (82.9%) were female, and nearly 37% had commercial insurance and 63.2% had Medicaid. About half the patients on Medicaid were Black and 36.2% were White (race and ethnicity data were not available for commercially insured patients). In both insurance groups, the rates of comorbidities were high, including 22.5% with obesity and 11.9% with diabetes.
The researchers found that, at the index ED visit, 48.0% of patients had incision and drainage performed (51% among those with commercial insurance vs. 46.3% of those with Medicaid; P < .001); 72.6% of patients filled an oral antibiotic prescription within 7 days, with similar percentages in both insurance groups; and 48.9% filled an oral opioid medication prescription within 7 days (46.5% for commercial insurance vs. 50.3% for Medicaid; P < .001).
Regarding follow-up care, the investigators found that 17.2% of patients had at least one return ED visit for HS or proxy within 30 days (15.7% for commercial vs. 18.1% for Medicaid; P < .001), while 2.4% had a dermatology visit (5.3% for commercial vs. 0.7% for Medicaid; P < .001). In addition, 34% of patients had at least one return ED visit for HS or proxy within 180 days (27.2% for commercial insurance vs. 38% for Medicaid; P < .001), while 6.8% had a dermatology visit (14.1% for commercial vs. 2.5% for Medicaid; P < .001).
Patients with an opioid prescribed within 7 days of the ED visit were more likely to return to the ED, within 30 days (odds ratio, 1.67; 95% confidence interval, 1.54-1.80; P < .001), and within 180 days (OR, 1.48; 95% CI, 1.39-1.58; P < .001). But they were less likely to have dermatology follow-up within 30 days (OR, 0.78; 95% CI, 0.64-0.95; P = .01) and within 180 days (OR, 0.81; 95% CI, 0.71-0.91; P < .001).
Medicaid patients were more likely to return to the ED within 30 days (OR, 1.12; 95% CI, 1.03-1.22; P = .009) and within 180 days (OR, 1.48; 95% CI, 1.38-1.58; P < .001). But they were less likely to receive outpatient dermatology follow-up care within 30 days (OR, 0.12; 95% CI, 0.09-0.15; P < .001) and within 180 days (OR, 0.16; 95% CI, 0.14-0.18; P < .001).
“This study highlights potential areas of action to improve care for patients with HS,” the authors concluded, including cross-specialty education and interventions, and focus on patients most likely to return to the ED for care.
Findings are not surprising
Christopher Sayed, MD, professor of dermatology at the University of North Carolina at Chapel Hill, said in an interview that the study results are expected. “Many patients have trouble establishing care with a dermatologist familiar with HS, so they seek more fragmented care at urgent cares and EDs,” he said.
“Some dermatologists are not familiar with HS or don’t accept insurance such as Medicaid,” Dr. Sayed added. “Many emergency room providers may not recognize that medical therapy for HS has evolved in a way that makes referral to a dermatologist more essential than ever. They may tell patients there is nothing else to do but return to the ED for the next flare. “Emergency medicine and dermatology training programs need to educate providers about appropriate long-term HS management.”
In an interview, Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital in New York and assistant professor of emergency medicine at Hofstra University, Hempstead, N.Y., said that the study describes a reality he and his colleagues know too well.
“The study gives a true snapshot of the disarray and inequality that exists for patients disproportionately affected by HS. Those who are African American and low income suffer from lack of HS primary dermatologic care and follow-up at much higher rates than do other demographic groups,” he said.
Doctors would like to see the current situation change, Dr. Glatter noted. “It’s frustrating for emergency physicians and for dermatologists, who know that optimal follow-up care for this chronic and disabling disease should be with a dermatologist (and other surgical specialists if necessary).
“It’s a broken system. Patients can’t get appointments in nonacademic private settings because the bulk of dermatologists will not accept Medicaid. And many academic practices will not see these patients, either,” he commented. “We end up becoming a safety net of care.”
Replace the broken system with an integrated process
A solution to address the problem would be to set up follow-up dermatology appointments when patients arrive in EDs during and after normal business hours, Dr. Glatter suggested. “Developing a coordinated, structured, streamlined process requires buy-in from all stakeholders, including private dermatologists, academic dermatology clinics, and the government.”
Having the Centers for Medicare & Medicaid Services study interventions for high utilizers of EDs for HS would also help with “the development of economic and logistical changes, including provider reimbursement and allocation of funds to address this ongoing disparity in care,” he added.
Ideally, larger health care systems could collaborate with academic and nonacademic dermatologists to design a referral network that cares for all uninsured or underinsured patients, he said. “Balancing patient care and improved outcomes – while working on a framework for reimbursement – would be in everyone’s best interest.”
The study was partially funded by a grant from the National Center for Advancing Translational Sciences of the National Institutes of Health. One author reported financial involvements with multiple pharmaceutical companies. Dr. Wang, the remaining coauthors, as well as Dr. Sayed and Dr. Glatter reported no conflicts of interest with the study. Dr. Glatter is an editor and columnist at Medscape.
according to a study of a large national administrative data set.
“Patients with HS presenting to the ED for their disease exhibited high rates of ED return with low rates of dermatology follow-up after an initial ED visit,” lead study author Cynthia X. Wang, MD, MPHS, and colleagues in the division of dermatology and the Institute for Informatics, Washington University in St. Louis, wrote in JAMA Dermatology.
Patients who received opioid prescriptions and patients on Medicaid were more likely to return to the ED, they noted.
HS, a debilitating skin disease involving chronic follicular inflammation, frequently affects the axilla, anogenital, and inframammary areas, with painful nodules, abscesses, and sinus tracts that can form scars, the authors wrote. HS is linked with comorbidities including inflammatory bowel disease, obesity, diabetes, substance use, and psychiatric conditions, and is often misdiagnosed for an estimated 7-10 years.
In the retrospective cohort study, Dr. Wang and colleagues collected data from nationwide commercial and Medicaid databases. They included patients aged 18 to 64 years with two or more claims for HS and with at least one ED visit not resulting in admission for HS or a defined proxy (such as a related diagnosis of folliculitis, in a location typical for HS) between 2010 and 2019.
The median age of the 20,269 patients in the study was 32 years; most (82.9%) were female, and nearly 37% had commercial insurance and 63.2% had Medicaid. About half the patients on Medicaid were Black and 36.2% were White (race and ethnicity data were not available for commercially insured patients). In both insurance groups, the rates of comorbidities were high, including 22.5% with obesity and 11.9% with diabetes.
The researchers found that, at the index ED visit, 48.0% of patients had incision and drainage performed (51% among those with commercial insurance vs. 46.3% of those with Medicaid; P < .001); 72.6% of patients filled an oral antibiotic prescription within 7 days, with similar percentages in both insurance groups; and 48.9% filled an oral opioid medication prescription within 7 days (46.5% for commercial insurance vs. 50.3% for Medicaid; P < .001).
Regarding follow-up care, the investigators found that 17.2% of patients had at least one return ED visit for HS or proxy within 30 days (15.7% for commercial vs. 18.1% for Medicaid; P < .001), while 2.4% had a dermatology visit (5.3% for commercial vs. 0.7% for Medicaid; P < .001). In addition, 34% of patients had at least one return ED visit for HS or proxy within 180 days (27.2% for commercial insurance vs. 38% for Medicaid; P < .001), while 6.8% had a dermatology visit (14.1% for commercial vs. 2.5% for Medicaid; P < .001).
Patients with an opioid prescribed within 7 days of the ED visit were more likely to return to the ED, within 30 days (odds ratio, 1.67; 95% confidence interval, 1.54-1.80; P < .001), and within 180 days (OR, 1.48; 95% CI, 1.39-1.58; P < .001). But they were less likely to have dermatology follow-up within 30 days (OR, 0.78; 95% CI, 0.64-0.95; P = .01) and within 180 days (OR, 0.81; 95% CI, 0.71-0.91; P < .001).
Medicaid patients were more likely to return to the ED within 30 days (OR, 1.12; 95% CI, 1.03-1.22; P = .009) and within 180 days (OR, 1.48; 95% CI, 1.38-1.58; P < .001). But they were less likely to receive outpatient dermatology follow-up care within 30 days (OR, 0.12; 95% CI, 0.09-0.15; P < .001) and within 180 days (OR, 0.16; 95% CI, 0.14-0.18; P < .001).
“This study highlights potential areas of action to improve care for patients with HS,” the authors concluded, including cross-specialty education and interventions, and focus on patients most likely to return to the ED for care.
Findings are not surprising
Christopher Sayed, MD, professor of dermatology at the University of North Carolina at Chapel Hill, said in an interview that the study results are expected. “Many patients have trouble establishing care with a dermatologist familiar with HS, so they seek more fragmented care at urgent cares and EDs,” he said.
“Some dermatologists are not familiar with HS or don’t accept insurance such as Medicaid,” Dr. Sayed added. “Many emergency room providers may not recognize that medical therapy for HS has evolved in a way that makes referral to a dermatologist more essential than ever. They may tell patients there is nothing else to do but return to the ED for the next flare. “Emergency medicine and dermatology training programs need to educate providers about appropriate long-term HS management.”
In an interview, Robert Glatter, MD, an emergency medicine physician at Lenox Hill Hospital in New York and assistant professor of emergency medicine at Hofstra University, Hempstead, N.Y., said that the study describes a reality he and his colleagues know too well.
“The study gives a true snapshot of the disarray and inequality that exists for patients disproportionately affected by HS. Those who are African American and low income suffer from lack of HS primary dermatologic care and follow-up at much higher rates than do other demographic groups,” he said.
Doctors would like to see the current situation change, Dr. Glatter noted. “It’s frustrating for emergency physicians and for dermatologists, who know that optimal follow-up care for this chronic and disabling disease should be with a dermatologist (and other surgical specialists if necessary).
“It’s a broken system. Patients can’t get appointments in nonacademic private settings because the bulk of dermatologists will not accept Medicaid. And many academic practices will not see these patients, either,” he commented. “We end up becoming a safety net of care.”
Replace the broken system with an integrated process
A solution to address the problem would be to set up follow-up dermatology appointments when patients arrive in EDs during and after normal business hours, Dr. Glatter suggested. “Developing a coordinated, structured, streamlined process requires buy-in from all stakeholders, including private dermatologists, academic dermatology clinics, and the government.”
Having the Centers for Medicare & Medicaid Services study interventions for high utilizers of EDs for HS would also help with “the development of economic and logistical changes, including provider reimbursement and allocation of funds to address this ongoing disparity in care,” he added.
Ideally, larger health care systems could collaborate with academic and nonacademic dermatologists to design a referral network that cares for all uninsured or underinsured patients, he said. “Balancing patient care and improved outcomes – while working on a framework for reimbursement – would be in everyone’s best interest.”
The study was partially funded by a grant from the National Center for Advancing Translational Sciences of the National Institutes of Health. One author reported financial involvements with multiple pharmaceutical companies. Dr. Wang, the remaining coauthors, as well as Dr. Sayed and Dr. Glatter reported no conflicts of interest with the study. Dr. Glatter is an editor and columnist at Medscape.
FROM JAMA DERMATOLOGY
Dark spot near ear

Stable slate gray to blue lesions that are asymptomatic raise the possibility of a blue nevus, also known as dermal dendritic melanocytic proliferations. In this case, dermoscopy confirmed a uniform dark color with no signs suggestive of melanoma or pigmented basal cell carcinoma (BCC).
Blue nevi are the result of a benign localized proliferation of dermal dendritic melanocytes. The blue color is due to the increased pigment deep in the dermis that reflects the blue shorter wavelength light while absorbing longer wavelengths.1 In this author’s experience, these “blue” lesions usually appear to be more gray (as was the case with this individual). Dermoscopy shows a steel blue homogenous pigmentation.2
It is helpful to use dermoscopy to screen for an atypical pigment network, regression of pigmentation, or abnormal pigmentation; these are signs of atypical nevi and melanoma. It is also important to look for arborizing blood vessels and leaf-like structures that can be seen in pigmented BCCs. Both melanoma and pigmented BCCs can appear as circumscribed dark lesions.
Reassuring factors for blue nevi are lesions that are stable in size and color over time, asymptomatic, and have not bled nor shown signs of erosion. If the diagnosis is in doubt, excise the lesion in its entirety for definitive pathology. Since the melanocytes are typically deeper in blue nevi than in most other nevi, a deep shave technique may not remove the lesion in its entirety. A deeper than usual shave (or, if feasible, a full-thickness excision) may return better results with quicker healing.
This patient was advised of the benign nature of a blue nevus. He was counseled to watch for any changes in the lesion and to return for reevaluation if symptoms or changes occurred.
Image and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Murali R, McCarthy SW, Scolyer RA. Blue nevi and related lesions: a review highlighting atypical and newly described variants, distinguishing features and diagnostic pitfalls. Adv Anat Pathol. 2009;16:365-382. doi: 10.1097/PAP.0b013e3181bb6b53
2. Longo C, Scope A, Lallas A, et al. Blue lesions. Dermatol Clin. 2013;31:637-647, ix. doi: 10.1016/j.det.2013.07.001

Stable slate gray to blue lesions that are asymptomatic raise the possibility of a blue nevus, also known as dermal dendritic melanocytic proliferations. In this case, dermoscopy confirmed a uniform dark color with no signs suggestive of melanoma or pigmented basal cell carcinoma (BCC).
Blue nevi are the result of a benign localized proliferation of dermal dendritic melanocytes. The blue color is due to the increased pigment deep in the dermis that reflects the blue shorter wavelength light while absorbing longer wavelengths.1 In this author’s experience, these “blue” lesions usually appear to be more gray (as was the case with this individual). Dermoscopy shows a steel blue homogenous pigmentation.2
It is helpful to use dermoscopy to screen for an atypical pigment network, regression of pigmentation, or abnormal pigmentation; these are signs of atypical nevi and melanoma. It is also important to look for arborizing blood vessels and leaf-like structures that can be seen in pigmented BCCs. Both melanoma and pigmented BCCs can appear as circumscribed dark lesions.
Reassuring factors for blue nevi are lesions that are stable in size and color over time, asymptomatic, and have not bled nor shown signs of erosion. If the diagnosis is in doubt, excise the lesion in its entirety for definitive pathology. Since the melanocytes are typically deeper in blue nevi than in most other nevi, a deep shave technique may not remove the lesion in its entirety. A deeper than usual shave (or, if feasible, a full-thickness excision) may return better results with quicker healing.
This patient was advised of the benign nature of a blue nevus. He was counseled to watch for any changes in the lesion and to return for reevaluation if symptoms or changes occurred.
Image and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

Stable slate gray to blue lesions that are asymptomatic raise the possibility of a blue nevus, also known as dermal dendritic melanocytic proliferations. In this case, dermoscopy confirmed a uniform dark color with no signs suggestive of melanoma or pigmented basal cell carcinoma (BCC).
Blue nevi are the result of a benign localized proliferation of dermal dendritic melanocytes. The blue color is due to the increased pigment deep in the dermis that reflects the blue shorter wavelength light while absorbing longer wavelengths.1 In this author’s experience, these “blue” lesions usually appear to be more gray (as was the case with this individual). Dermoscopy shows a steel blue homogenous pigmentation.2
It is helpful to use dermoscopy to screen for an atypical pigment network, regression of pigmentation, or abnormal pigmentation; these are signs of atypical nevi and melanoma. It is also important to look for arborizing blood vessels and leaf-like structures that can be seen in pigmented BCCs. Both melanoma and pigmented BCCs can appear as circumscribed dark lesions.
Reassuring factors for blue nevi are lesions that are stable in size and color over time, asymptomatic, and have not bled nor shown signs of erosion. If the diagnosis is in doubt, excise the lesion in its entirety for definitive pathology. Since the melanocytes are typically deeper in blue nevi than in most other nevi, a deep shave technique may not remove the lesion in its entirety. A deeper than usual shave (or, if feasible, a full-thickness excision) may return better results with quicker healing.
This patient was advised of the benign nature of a blue nevus. He was counseled to watch for any changes in the lesion and to return for reevaluation if symptoms or changes occurred.
Image and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Murali R, McCarthy SW, Scolyer RA. Blue nevi and related lesions: a review highlighting atypical and newly described variants, distinguishing features and diagnostic pitfalls. Adv Anat Pathol. 2009;16:365-382. doi: 10.1097/PAP.0b013e3181bb6b53
2. Longo C, Scope A, Lallas A, et al. Blue lesions. Dermatol Clin. 2013;31:637-647, ix. doi: 10.1016/j.det.2013.07.001
1. Murali R, McCarthy SW, Scolyer RA. Blue nevi and related lesions: a review highlighting atypical and newly described variants, distinguishing features and diagnostic pitfalls. Adv Anat Pathol. 2009;16:365-382. doi: 10.1097/PAP.0b013e3181bb6b53
2. Longo C, Scope A, Lallas A, et al. Blue lesions. Dermatol Clin. 2013;31:637-647, ix. doi: 10.1016/j.det.2013.07.001
Consider radiologic imaging for high-risk cutaneous SCC, expert advises
DENVER –
In a study published in 2020, Emily Ruiz, MD, MPH, and colleagues identified 87 CSCC tumors in 83 patients who underwent baseline or surveillance imaging primary at the Brigham and Women’s Hospital Mohs Surgery Clinic and the Dana-Farber Cancer Institute High-Risk Skin Cancer Clinic, both in Boston, from Jan. 1, 2017, to June 1, 2019. Of the 87 primary CSCCs, 48 (58%) underwent surveillance imaging. The researchers found that imaging detected additional disease in 26 patients, or 30% of cases, “whether that be nodal metastasis, local invasion beyond what was clinically accepted, or in-transit disease,” Dr. Ruiz, academic director of the Mohs and Dermatologic Surgery Center at Brigham and Women’s, said during the annual meeting of the American Society for Dermatologic Surgery. “But if you look at the 16 nodal metastases in this cohort, all were picked up on imaging and not on clinical exam.”
Since publication of these results, Dr. Ruiz routinely considers baseline radiologic imaging in T2b and T3 tumors; borderline T2a tumors (which she said they are now calling “T2a high,” for those who have one risk factor plus another intermediate risk factor),” and T2a tumors in patients who are profoundly immunosuppressed.
“My preference is to always do [the imaging] before treatment unless I’m up-staging them during surgery,” said Dr. Ruiz, who also directs the High-Risk Skin Cancer Clinic at Dana Farber. “We have picked up nodal metastases before surgery, which enables us to create a good therapeutic plan for our patients before we start operating. Then we image them every 6 months or so for about 2 years. Sometimes we will extend that out to 3 years.”
Some clinicians use sentinel lymph node biopsy (SLNB) as a diagnostic test, but there are mixed results about its prognostic significance. A retrospective observational study of 720 patients with CSCC found that SLNB provided no benefit regarding further metastasis or tumor-specific survival, compared with those who received routine observation and follow-up, “but head and neck surgeons in the U.S. are putting together some prospective data from multiple centers,” Dr. Ruiz said. “I think in the coming years, you will have more multicenter data to inform us as to whether to do SLNB or not.”
Surgery may be the mainstay of treatment for resectable SCC, but the emerging role of neoadjuvant therapeutics is changing the way oncologists treat these tumors. For example, in a phase 2 trial recently published in the New England Journal of Medicine, 79 patients with stage II-IV CSCC received up to four doses of immunotherapy with the programmed death receptor–1 (PD-1) blocker cemiplimab administered every 3 weeks. The primary endpoint was a pathologic complete response, defined as the absence of viable tumor cells in the surgical specimen at a central laboratory. The researchers observed that 68% of patients had an objective response.
“These were patients with localized tumors that were either very aggressive or had nodal metastases,” said Dr, Ruiz, who was the site primary investigator at Dana Farber and a coauthor of the NEJM study. “This has altered the way we approach treating our larger tumors that could be resectable but have a lot of disease either locally or in the nodal basin. We think that we can shrink down the tumor and make it easier to resect, but also there is the possibility or improving outcomes.”
At Brigham and Women’s and the Dana Farber, she and her colleagues consider immunotherapy for multiple recurrent tumors that have been previously irradiated; cases of large tumor burden locally or in the nodal basin; tumors that have a complex surgical plan; cases where there is a low likelihood of achieving clear surgical margins; and cases of in-transit disease.
“We use two to four doses of immunotherapy prior to surgery and assess the tumor response after two doses both clinically and radiologically,” she said. “If the tumor continues to grow, we would do surgery sooner.”
The side-effect profile of immunotherapy is another consideration. “Some patients are not appropriate for a neoadjuvant immunotherapy approach, such as transplant patients,” she said.
According to the latest National Comprehensive Cancer Network guidelines, surgery with or without adjuvant radiation is the current standard of care for treating CSCC. These guidelines were developed without much data to support the use of radiation, but a 20-year retrospective cohort study at Brigham and Women’s Hospital and the Cleveland Clinic Foundation found that adjuvant radiation following margin resection in high T-stage CSCC cut the risk of local and locoregional recurrence in half.
“This is something that radiation oncologists have told us for years, but there was no data to support it, so it was nice to see that borne out in clinical data,” said Dr. Ruiz, the study’s lead author. The 10% risk of local recurrence observed in the study “may not be high enough for some of our older patients, so we wanted to see if we could identify a group of high tumors that had higher risk of local recurrence,” she said. They found that patients who had a greater than 20% risk of poor outcome were those with recurrent tumors, those with tumors 6 cm or greater in size, and those with all four BWH risk factors (tumor diameter ≥ 2 cm, poorly differentiated histology, perineural invasion ≥ 0.1 mm, or tumor invasion beyond fat excluding bone invasion).
“Those risks were also cut in half if you added radiation,” she said. “So, the way I now approach counseling patients is, I try to estimate their baseline risk as best I can based on the tumor itself. I tell them that if they want to do adjuvant radiation it would cut the risk in half. Some patients are too frail and want to pass on it, while others are very interested.”
Of patients who did not receive radiation but had a disease recurrence, just under half of tumors were salvageable, about 25% died of their disease, and 23% had persistent disease. “I think this does support using radiation earlier on for the appropriate patient,” Dr. Ruiz said. “I consider the baseline risks [and] balance that with the patient’s comorbidities.”
Limited data exists on adjuvant immunotherapy for CSCC, but two ongoing randomized prospective clinical trials underway are studying the PD-1 inhibitors cemiplimab and pembrolizumab versus placebo. “We don’t have data yet, but prior to randomization, patients undergo surgery with macroscopic gross resection of all disease,” Dr. Ruiz said. “All tumors receive ART [adjuvant radiation therapy] prior to randomization”
Dr. Ruiz disclosed that she is a consultant for Sanofi, Regeneron, Genentech, and Jaunce Therapeutics. She is also a member of the advisory board for Checkpoint Therapeutics and is an investigator for Merck, Sanofi, and Regeneron.
DENVER –
In a study published in 2020, Emily Ruiz, MD, MPH, and colleagues identified 87 CSCC tumors in 83 patients who underwent baseline or surveillance imaging primary at the Brigham and Women’s Hospital Mohs Surgery Clinic and the Dana-Farber Cancer Institute High-Risk Skin Cancer Clinic, both in Boston, from Jan. 1, 2017, to June 1, 2019. Of the 87 primary CSCCs, 48 (58%) underwent surveillance imaging. The researchers found that imaging detected additional disease in 26 patients, or 30% of cases, “whether that be nodal metastasis, local invasion beyond what was clinically accepted, or in-transit disease,” Dr. Ruiz, academic director of the Mohs and Dermatologic Surgery Center at Brigham and Women’s, said during the annual meeting of the American Society for Dermatologic Surgery. “But if you look at the 16 nodal metastases in this cohort, all were picked up on imaging and not on clinical exam.”
Since publication of these results, Dr. Ruiz routinely considers baseline radiologic imaging in T2b and T3 tumors; borderline T2a tumors (which she said they are now calling “T2a high,” for those who have one risk factor plus another intermediate risk factor),” and T2a tumors in patients who are profoundly immunosuppressed.
“My preference is to always do [the imaging] before treatment unless I’m up-staging them during surgery,” said Dr. Ruiz, who also directs the High-Risk Skin Cancer Clinic at Dana Farber. “We have picked up nodal metastases before surgery, which enables us to create a good therapeutic plan for our patients before we start operating. Then we image them every 6 months or so for about 2 years. Sometimes we will extend that out to 3 years.”
Some clinicians use sentinel lymph node biopsy (SLNB) as a diagnostic test, but there are mixed results about its prognostic significance. A retrospective observational study of 720 patients with CSCC found that SLNB provided no benefit regarding further metastasis or tumor-specific survival, compared with those who received routine observation and follow-up, “but head and neck surgeons in the U.S. are putting together some prospective data from multiple centers,” Dr. Ruiz said. “I think in the coming years, you will have more multicenter data to inform us as to whether to do SLNB or not.”
Surgery may be the mainstay of treatment for resectable SCC, but the emerging role of neoadjuvant therapeutics is changing the way oncologists treat these tumors. For example, in a phase 2 trial recently published in the New England Journal of Medicine, 79 patients with stage II-IV CSCC received up to four doses of immunotherapy with the programmed death receptor–1 (PD-1) blocker cemiplimab administered every 3 weeks. The primary endpoint was a pathologic complete response, defined as the absence of viable tumor cells in the surgical specimen at a central laboratory. The researchers observed that 68% of patients had an objective response.
“These were patients with localized tumors that were either very aggressive or had nodal metastases,” said Dr, Ruiz, who was the site primary investigator at Dana Farber and a coauthor of the NEJM study. “This has altered the way we approach treating our larger tumors that could be resectable but have a lot of disease either locally or in the nodal basin. We think that we can shrink down the tumor and make it easier to resect, but also there is the possibility or improving outcomes.”
At Brigham and Women’s and the Dana Farber, she and her colleagues consider immunotherapy for multiple recurrent tumors that have been previously irradiated; cases of large tumor burden locally or in the nodal basin; tumors that have a complex surgical plan; cases where there is a low likelihood of achieving clear surgical margins; and cases of in-transit disease.
“We use two to four doses of immunotherapy prior to surgery and assess the tumor response after two doses both clinically and radiologically,” she said. “If the tumor continues to grow, we would do surgery sooner.”
The side-effect profile of immunotherapy is another consideration. “Some patients are not appropriate for a neoadjuvant immunotherapy approach, such as transplant patients,” she said.
According to the latest National Comprehensive Cancer Network guidelines, surgery with or without adjuvant radiation is the current standard of care for treating CSCC. These guidelines were developed without much data to support the use of radiation, but a 20-year retrospective cohort study at Brigham and Women’s Hospital and the Cleveland Clinic Foundation found that adjuvant radiation following margin resection in high T-stage CSCC cut the risk of local and locoregional recurrence in half.
“This is something that radiation oncologists have told us for years, but there was no data to support it, so it was nice to see that borne out in clinical data,” said Dr. Ruiz, the study’s lead author. The 10% risk of local recurrence observed in the study “may not be high enough for some of our older patients, so we wanted to see if we could identify a group of high tumors that had higher risk of local recurrence,” she said. They found that patients who had a greater than 20% risk of poor outcome were those with recurrent tumors, those with tumors 6 cm or greater in size, and those with all four BWH risk factors (tumor diameter ≥ 2 cm, poorly differentiated histology, perineural invasion ≥ 0.1 mm, or tumor invasion beyond fat excluding bone invasion).
“Those risks were also cut in half if you added radiation,” she said. “So, the way I now approach counseling patients is, I try to estimate their baseline risk as best I can based on the tumor itself. I tell them that if they want to do adjuvant radiation it would cut the risk in half. Some patients are too frail and want to pass on it, while others are very interested.”
Of patients who did not receive radiation but had a disease recurrence, just under half of tumors were salvageable, about 25% died of their disease, and 23% had persistent disease. “I think this does support using radiation earlier on for the appropriate patient,” Dr. Ruiz said. “I consider the baseline risks [and] balance that with the patient’s comorbidities.”
Limited data exists on adjuvant immunotherapy for CSCC, but two ongoing randomized prospective clinical trials underway are studying the PD-1 inhibitors cemiplimab and pembrolizumab versus placebo. “We don’t have data yet, but prior to randomization, patients undergo surgery with macroscopic gross resection of all disease,” Dr. Ruiz said. “All tumors receive ART [adjuvant radiation therapy] prior to randomization”
Dr. Ruiz disclosed that she is a consultant for Sanofi, Regeneron, Genentech, and Jaunce Therapeutics. She is also a member of the advisory board for Checkpoint Therapeutics and is an investigator for Merck, Sanofi, and Regeneron.
DENVER –
In a study published in 2020, Emily Ruiz, MD, MPH, and colleagues identified 87 CSCC tumors in 83 patients who underwent baseline or surveillance imaging primary at the Brigham and Women’s Hospital Mohs Surgery Clinic and the Dana-Farber Cancer Institute High-Risk Skin Cancer Clinic, both in Boston, from Jan. 1, 2017, to June 1, 2019. Of the 87 primary CSCCs, 48 (58%) underwent surveillance imaging. The researchers found that imaging detected additional disease in 26 patients, or 30% of cases, “whether that be nodal metastasis, local invasion beyond what was clinically accepted, or in-transit disease,” Dr. Ruiz, academic director of the Mohs and Dermatologic Surgery Center at Brigham and Women’s, said during the annual meeting of the American Society for Dermatologic Surgery. “But if you look at the 16 nodal metastases in this cohort, all were picked up on imaging and not on clinical exam.”
Since publication of these results, Dr. Ruiz routinely considers baseline radiologic imaging in T2b and T3 tumors; borderline T2a tumors (which she said they are now calling “T2a high,” for those who have one risk factor plus another intermediate risk factor),” and T2a tumors in patients who are profoundly immunosuppressed.
“My preference is to always do [the imaging] before treatment unless I’m up-staging them during surgery,” said Dr. Ruiz, who also directs the High-Risk Skin Cancer Clinic at Dana Farber. “We have picked up nodal metastases before surgery, which enables us to create a good therapeutic plan for our patients before we start operating. Then we image them every 6 months or so for about 2 years. Sometimes we will extend that out to 3 years.”
Some clinicians use sentinel lymph node biopsy (SLNB) as a diagnostic test, but there are mixed results about its prognostic significance. A retrospective observational study of 720 patients with CSCC found that SLNB provided no benefit regarding further metastasis or tumor-specific survival, compared with those who received routine observation and follow-up, “but head and neck surgeons in the U.S. are putting together some prospective data from multiple centers,” Dr. Ruiz said. “I think in the coming years, you will have more multicenter data to inform us as to whether to do SLNB or not.”
Surgery may be the mainstay of treatment for resectable SCC, but the emerging role of neoadjuvant therapeutics is changing the way oncologists treat these tumors. For example, in a phase 2 trial recently published in the New England Journal of Medicine, 79 patients with stage II-IV CSCC received up to four doses of immunotherapy with the programmed death receptor–1 (PD-1) blocker cemiplimab administered every 3 weeks. The primary endpoint was a pathologic complete response, defined as the absence of viable tumor cells in the surgical specimen at a central laboratory. The researchers observed that 68% of patients had an objective response.
“These were patients with localized tumors that were either very aggressive or had nodal metastases,” said Dr, Ruiz, who was the site primary investigator at Dana Farber and a coauthor of the NEJM study. “This has altered the way we approach treating our larger tumors that could be resectable but have a lot of disease either locally or in the nodal basin. We think that we can shrink down the tumor and make it easier to resect, but also there is the possibility or improving outcomes.”
At Brigham and Women’s and the Dana Farber, she and her colleagues consider immunotherapy for multiple recurrent tumors that have been previously irradiated; cases of large tumor burden locally or in the nodal basin; tumors that have a complex surgical plan; cases where there is a low likelihood of achieving clear surgical margins; and cases of in-transit disease.
“We use two to four doses of immunotherapy prior to surgery and assess the tumor response after two doses both clinically and radiologically,” she said. “If the tumor continues to grow, we would do surgery sooner.”
The side-effect profile of immunotherapy is another consideration. “Some patients are not appropriate for a neoadjuvant immunotherapy approach, such as transplant patients,” she said.
According to the latest National Comprehensive Cancer Network guidelines, surgery with or without adjuvant radiation is the current standard of care for treating CSCC. These guidelines were developed without much data to support the use of radiation, but a 20-year retrospective cohort study at Brigham and Women’s Hospital and the Cleveland Clinic Foundation found that adjuvant radiation following margin resection in high T-stage CSCC cut the risk of local and locoregional recurrence in half.
“This is something that radiation oncologists have told us for years, but there was no data to support it, so it was nice to see that borne out in clinical data,” said Dr. Ruiz, the study’s lead author. The 10% risk of local recurrence observed in the study “may not be high enough for some of our older patients, so we wanted to see if we could identify a group of high tumors that had higher risk of local recurrence,” she said. They found that patients who had a greater than 20% risk of poor outcome were those with recurrent tumors, those with tumors 6 cm or greater in size, and those with all four BWH risk factors (tumor diameter ≥ 2 cm, poorly differentiated histology, perineural invasion ≥ 0.1 mm, or tumor invasion beyond fat excluding bone invasion).
“Those risks were also cut in half if you added radiation,” she said. “So, the way I now approach counseling patients is, I try to estimate their baseline risk as best I can based on the tumor itself. I tell them that if they want to do adjuvant radiation it would cut the risk in half. Some patients are too frail and want to pass on it, while others are very interested.”
Of patients who did not receive radiation but had a disease recurrence, just under half of tumors were salvageable, about 25% died of their disease, and 23% had persistent disease. “I think this does support using radiation earlier on for the appropriate patient,” Dr. Ruiz said. “I consider the baseline risks [and] balance that with the patient’s comorbidities.”
Limited data exists on adjuvant immunotherapy for CSCC, but two ongoing randomized prospective clinical trials underway are studying the PD-1 inhibitors cemiplimab and pembrolizumab versus placebo. “We don’t have data yet, but prior to randomization, patients undergo surgery with macroscopic gross resection of all disease,” Dr. Ruiz said. “All tumors receive ART [adjuvant radiation therapy] prior to randomization”
Dr. Ruiz disclosed that she is a consultant for Sanofi, Regeneron, Genentech, and Jaunce Therapeutics. She is also a member of the advisory board for Checkpoint Therapeutics and is an investigator for Merck, Sanofi, and Regeneron.
AT ASDS 2022
Brepocitinib improves symptoms of mild to moderate AD in phase 2b trial
compared with a group that received vehicle, in a recently published study..
The investigators said that brepocitinib, an investigational dual tyrosine kinase 2 (TYK2) and Janus kinase 1 (JAK1) inhibitor, was effective and well tolerated in patients with mild to moderate AD based on improvements in multiple measures, including Eczema Area and Severity Index (EASI) total score and Investigator Global Assessment (IGA) responder rates. Brepocitinib also reduced pruritus symptoms as early as 2 days after the start of treatment, they noted.
“This study supports the further evaluation of topical brepocitinib as a novel treatment for mild to moderate AD,” Megan N. Landis, MD, of the department of medicine at the University of Louisville (Ky.) and colleagues wrote in the study published in the British Journal of Dermatology.
They evaluated brepocitinib in a phase 2b, double-blind, dose-ranging study where 292 patients were randomized to receive brepocitinib once daily (brepocitinib 0.1%, 0.3%, 1.0%, 3.0%) or twice daily (brepocitinib 0.3%, 1.0%), or vehicle for 6 weeks. At 6 weeks, the researchers assessed EASI total score as a primary outcome, an IGA score of 0 or 1 as a secondary outcome. The mean age of the patients was 40 years (range, 13-74), almost 60% were White, 17.5% were Black, and about 20% were Asian.
Compared with the corresponding once-daily vehicle group (least squares mean reduction of –44.4; 90% confidence interval, –57.3 to –31.6) and the twice-daily vehicle group (LSM, –47.6; 90% CI, –57.5 to –37.7) , the brepocitinib 1% once-daily group (LSM, –70.1; 90% CI, –82.1 to –58.0) and twice-daily group (LSM, –75.0; 90% CI, –83.8 to –66.2) had significant percentage reductions in EASI total score compared with baseline at 6 weeks. Patients in the other brepocitinib dose groups had nonsignificant reductions in EASI from baseline.
Regarding secondary outcomes, a significantly higher percentage of patients in five of the six active treatment groups achieved an IGA score of 0 or 1 and at least a 2-point reduction in IGA score in the once-daily brepocitinib 0.1% group (29.7%; 90% CI, 18.5%-43.3%), 0.3% group (33.3%; 90% CI, 21.3%-47.0%), 1.0% group (40.5%; 90% CI, 28.0%-54.4%), 3.0% group (44.4%; 90% CI, 30.2%-59.1%), and brepocitinib 0.3% twice-daily group (33.3%; 90% CI, 21.3%-47.0%) compared with the once-daily (10.8%; 90% CI, 4.8%-22.2%) and twice-daily (13.9%; 90% CI, 6.9%-25.4%) vehicle groups.
The study authors noted that 37.0% of patients overall experienced treatment-emergent adverse events (TEAEs), with most TEAEs occurring in the once-daily vehicle (48.6%), twice-daily vehicle (47.2%), and brepocitinib 0.1% (45.9%) groups. Adverse events were not considered dose dependent, and no group had any serious TEAEs or deaths.
Nasopharyngitis and worsening AD were the most common TEAEs reported, with about 8% of those in the vehicle groups experiencing worsening AD.
Brepocitinib is also currently being developed as a treatment for dermatomyositis, systemic lupus erythematosus, hidradenitis suppurativa, and noninfectious uveitis by Priovant Therapeutics, a company founded by Pfizer and Roivant Sciences.
In September 2021, the Food and Drug Administration approved topical ruxolitinib cream for the treatment of patients with mild to moderate atopic dermatitis aged 12 years and older, the first topical JAK inhibitor approved for AD.
This study was sponsored by Pfizer. The authors reported personal and institutional relationships in the form of investigator positions, fees, honoraria, research grants, employee positions, and holding stock or shares for a variety of pharmaceutical, life science, and biotechnology companies.
compared with a group that received vehicle, in a recently published study..
The investigators said that brepocitinib, an investigational dual tyrosine kinase 2 (TYK2) and Janus kinase 1 (JAK1) inhibitor, was effective and well tolerated in patients with mild to moderate AD based on improvements in multiple measures, including Eczema Area and Severity Index (EASI) total score and Investigator Global Assessment (IGA) responder rates. Brepocitinib also reduced pruritus symptoms as early as 2 days after the start of treatment, they noted.
“This study supports the further evaluation of topical brepocitinib as a novel treatment for mild to moderate AD,” Megan N. Landis, MD, of the department of medicine at the University of Louisville (Ky.) and colleagues wrote in the study published in the British Journal of Dermatology.
They evaluated brepocitinib in a phase 2b, double-blind, dose-ranging study where 292 patients were randomized to receive brepocitinib once daily (brepocitinib 0.1%, 0.3%, 1.0%, 3.0%) or twice daily (brepocitinib 0.3%, 1.0%), or vehicle for 6 weeks. At 6 weeks, the researchers assessed EASI total score as a primary outcome, an IGA score of 0 or 1 as a secondary outcome. The mean age of the patients was 40 years (range, 13-74), almost 60% were White, 17.5% were Black, and about 20% were Asian.
Compared with the corresponding once-daily vehicle group (least squares mean reduction of –44.4; 90% confidence interval, –57.3 to –31.6) and the twice-daily vehicle group (LSM, –47.6; 90% CI, –57.5 to –37.7) , the brepocitinib 1% once-daily group (LSM, –70.1; 90% CI, –82.1 to –58.0) and twice-daily group (LSM, –75.0; 90% CI, –83.8 to –66.2) had significant percentage reductions in EASI total score compared with baseline at 6 weeks. Patients in the other brepocitinib dose groups had nonsignificant reductions in EASI from baseline.
Regarding secondary outcomes, a significantly higher percentage of patients in five of the six active treatment groups achieved an IGA score of 0 or 1 and at least a 2-point reduction in IGA score in the once-daily brepocitinib 0.1% group (29.7%; 90% CI, 18.5%-43.3%), 0.3% group (33.3%; 90% CI, 21.3%-47.0%), 1.0% group (40.5%; 90% CI, 28.0%-54.4%), 3.0% group (44.4%; 90% CI, 30.2%-59.1%), and brepocitinib 0.3% twice-daily group (33.3%; 90% CI, 21.3%-47.0%) compared with the once-daily (10.8%; 90% CI, 4.8%-22.2%) and twice-daily (13.9%; 90% CI, 6.9%-25.4%) vehicle groups.
The study authors noted that 37.0% of patients overall experienced treatment-emergent adverse events (TEAEs), with most TEAEs occurring in the once-daily vehicle (48.6%), twice-daily vehicle (47.2%), and brepocitinib 0.1% (45.9%) groups. Adverse events were not considered dose dependent, and no group had any serious TEAEs or deaths.
Nasopharyngitis and worsening AD were the most common TEAEs reported, with about 8% of those in the vehicle groups experiencing worsening AD.
Brepocitinib is also currently being developed as a treatment for dermatomyositis, systemic lupus erythematosus, hidradenitis suppurativa, and noninfectious uveitis by Priovant Therapeutics, a company founded by Pfizer and Roivant Sciences.
In September 2021, the Food and Drug Administration approved topical ruxolitinib cream for the treatment of patients with mild to moderate atopic dermatitis aged 12 years and older, the first topical JAK inhibitor approved for AD.
This study was sponsored by Pfizer. The authors reported personal and institutional relationships in the form of investigator positions, fees, honoraria, research grants, employee positions, and holding stock or shares for a variety of pharmaceutical, life science, and biotechnology companies.
compared with a group that received vehicle, in a recently published study..
The investigators said that brepocitinib, an investigational dual tyrosine kinase 2 (TYK2) and Janus kinase 1 (JAK1) inhibitor, was effective and well tolerated in patients with mild to moderate AD based on improvements in multiple measures, including Eczema Area and Severity Index (EASI) total score and Investigator Global Assessment (IGA) responder rates. Brepocitinib also reduced pruritus symptoms as early as 2 days after the start of treatment, they noted.
“This study supports the further evaluation of topical brepocitinib as a novel treatment for mild to moderate AD,” Megan N. Landis, MD, of the department of medicine at the University of Louisville (Ky.) and colleagues wrote in the study published in the British Journal of Dermatology.
They evaluated brepocitinib in a phase 2b, double-blind, dose-ranging study where 292 patients were randomized to receive brepocitinib once daily (brepocitinib 0.1%, 0.3%, 1.0%, 3.0%) or twice daily (brepocitinib 0.3%, 1.0%), or vehicle for 6 weeks. At 6 weeks, the researchers assessed EASI total score as a primary outcome, an IGA score of 0 or 1 as a secondary outcome. The mean age of the patients was 40 years (range, 13-74), almost 60% were White, 17.5% were Black, and about 20% were Asian.
Compared with the corresponding once-daily vehicle group (least squares mean reduction of –44.4; 90% confidence interval, –57.3 to –31.6) and the twice-daily vehicle group (LSM, –47.6; 90% CI, –57.5 to –37.7) , the brepocitinib 1% once-daily group (LSM, –70.1; 90% CI, –82.1 to –58.0) and twice-daily group (LSM, –75.0; 90% CI, –83.8 to –66.2) had significant percentage reductions in EASI total score compared with baseline at 6 weeks. Patients in the other brepocitinib dose groups had nonsignificant reductions in EASI from baseline.
Regarding secondary outcomes, a significantly higher percentage of patients in five of the six active treatment groups achieved an IGA score of 0 or 1 and at least a 2-point reduction in IGA score in the once-daily brepocitinib 0.1% group (29.7%; 90% CI, 18.5%-43.3%), 0.3% group (33.3%; 90% CI, 21.3%-47.0%), 1.0% group (40.5%; 90% CI, 28.0%-54.4%), 3.0% group (44.4%; 90% CI, 30.2%-59.1%), and brepocitinib 0.3% twice-daily group (33.3%; 90% CI, 21.3%-47.0%) compared with the once-daily (10.8%; 90% CI, 4.8%-22.2%) and twice-daily (13.9%; 90% CI, 6.9%-25.4%) vehicle groups.
The study authors noted that 37.0% of patients overall experienced treatment-emergent adverse events (TEAEs), with most TEAEs occurring in the once-daily vehicle (48.6%), twice-daily vehicle (47.2%), and brepocitinib 0.1% (45.9%) groups. Adverse events were not considered dose dependent, and no group had any serious TEAEs or deaths.
Nasopharyngitis and worsening AD were the most common TEAEs reported, with about 8% of those in the vehicle groups experiencing worsening AD.
Brepocitinib is also currently being developed as a treatment for dermatomyositis, systemic lupus erythematosus, hidradenitis suppurativa, and noninfectious uveitis by Priovant Therapeutics, a company founded by Pfizer and Roivant Sciences.
In September 2021, the Food and Drug Administration approved topical ruxolitinib cream for the treatment of patients with mild to moderate atopic dermatitis aged 12 years and older, the first topical JAK inhibitor approved for AD.
This study was sponsored by Pfizer. The authors reported personal and institutional relationships in the form of investigator positions, fees, honoraria, research grants, employee positions, and holding stock or shares for a variety of pharmaceutical, life science, and biotechnology companies.
FROM BRITISH JOURNAL OF DERMATOLOGY