User login
President Biden’s ‘Cancer Moonshot’ to be relaunched
The “Cancer Moonshot” is about to be relaunched.
In a White House briefing, President Joe Biden announced that he is “reigniting” the initiative he spearheaded when he was vice president during the Obama administration.
During the livestreamed event, the president discussed his plans to bring a “fierce sense of urgency” to the fight against cancer and better support patients with cancer and their families.
He emphasized that cancer is one of the truly bipartisan issues. There is strong support from both “sides of the aisle,” he said, and he sees it as an issue that can bring the country together.
“We can do this. I promise you, we can do this. For all those we lost, for all those we miss. We can end cancer as we know it,” he said. “This is a presidential White House priority.”
The aim is to reduce the death rate from cancer by at least 50% over the next 25 years.
There is also a proposal to create the Advanced Research Projects Agency for Health, which would focus on driving cutting-edge innovation in health research.
Part of the plan is to assemble a “cancer cabinet” that includes 18 federal departments, agencies, and offices, including leaders from the departments of Health & Human Services, Veterans Affairs, Defense, Energy, and Agriculture.
At present, there are few details about the new program or how it will be funded.
Presumably more will be revealed at the Cancer Moonshot Summit being planned, as well as on a planned new website where people can track its progress.
President priority
Cancer Moonshot began back in 2016, when during his last State of the Union Address, former President Barack Obama announced the ambitious initiative. A few days later, Obama asked Congress for $1 billion to send cancer to the moon, and he put Biden, then vice president, in charge of “mission control” in the remaining months of the administration.
The new initiative will be headed by Danielle Carnival, PhD, who serves in the White House Office of Science and Technology Policy and has been appointed as White House Cancer Moonshot coordinator.
At the briefing, Mr. Biden and Vice President Kamala Harris spoke about losing family members to cancer. The president spoke about his eldest son, Beau, who died from brain cancer when he was 46 years old, while Ms. Harris spoke about her mother, Shyamala Gopalan, a breast cancer researcher who died of colon cancer in 2009.
Accolades but a bit of caution
The president’s speech was applauded by many cancer groups, both professional organizations and patient advocacy groups.
Karen E. Knudsen, PhD, chief executive officer of the American Cancer Society and its advocacy affiliate, the American Cancer Society Cancer Action Network, commended Mr. Biden for reigniting Cancer Moonshot.
“In 2022 alone, there will be an estimated 1.9 million people diagnosed with cancer and more than 600,000 people in the U.S. will die. Marshaling the resources of the federal government will be critical in our ability to reduce death and suffering from this disease,” she said.
The American Society for Radiation Oncology issued a press release, saying: “On behalf of radiation oncologists who treat people with cancer every day, we support the Biden-Harris administration’s move to drastically reduce the number of cancer deaths in the United States and improve the lives of people diagnosed with this disease.
“We believe the administration’s commitment to expand cancer prevention efforts and to increase equitable access to screenings and treatments will help mitigate some of the negative impact of the COVID-19 pandemic,” the society added.
At the American Association for Cancer Research, Chief Executive Officer Margaret Foti, MD, PhD, said she was thrilled to hear the announcement after the devastating interruptions in cancer research and patient care over the past 2 years.
“The reignited Cancer Moonshot will provide an important framework to help improve cancer prevention strategies, increase cancer screenings and early detection, reduce cancer disparities, and propel new lifesaving cures for patients with cancer,” she said.
However, increased funding from Congress will be needed for these goals to be achieved, she emphasized.
A version of this article first appeared on Medscape.com.
The “Cancer Moonshot” is about to be relaunched.
In a White House briefing, President Joe Biden announced that he is “reigniting” the initiative he spearheaded when he was vice president during the Obama administration.
During the livestreamed event, the president discussed his plans to bring a “fierce sense of urgency” to the fight against cancer and better support patients with cancer and their families.
He emphasized that cancer is one of the truly bipartisan issues. There is strong support from both “sides of the aisle,” he said, and he sees it as an issue that can bring the country together.
“We can do this. I promise you, we can do this. For all those we lost, for all those we miss. We can end cancer as we know it,” he said. “This is a presidential White House priority.”
The aim is to reduce the death rate from cancer by at least 50% over the next 25 years.
There is also a proposal to create the Advanced Research Projects Agency for Health, which would focus on driving cutting-edge innovation in health research.
Part of the plan is to assemble a “cancer cabinet” that includes 18 federal departments, agencies, and offices, including leaders from the departments of Health & Human Services, Veterans Affairs, Defense, Energy, and Agriculture.
At present, there are few details about the new program or how it will be funded.
Presumably more will be revealed at the Cancer Moonshot Summit being planned, as well as on a planned new website where people can track its progress.
President priority
Cancer Moonshot began back in 2016, when during his last State of the Union Address, former President Barack Obama announced the ambitious initiative. A few days later, Obama asked Congress for $1 billion to send cancer to the moon, and he put Biden, then vice president, in charge of “mission control” in the remaining months of the administration.
The new initiative will be headed by Danielle Carnival, PhD, who serves in the White House Office of Science and Technology Policy and has been appointed as White House Cancer Moonshot coordinator.
At the briefing, Mr. Biden and Vice President Kamala Harris spoke about losing family members to cancer. The president spoke about his eldest son, Beau, who died from brain cancer when he was 46 years old, while Ms. Harris spoke about her mother, Shyamala Gopalan, a breast cancer researcher who died of colon cancer in 2009.
Accolades but a bit of caution
The president’s speech was applauded by many cancer groups, both professional organizations and patient advocacy groups.
Karen E. Knudsen, PhD, chief executive officer of the American Cancer Society and its advocacy affiliate, the American Cancer Society Cancer Action Network, commended Mr. Biden for reigniting Cancer Moonshot.
“In 2022 alone, there will be an estimated 1.9 million people diagnosed with cancer and more than 600,000 people in the U.S. will die. Marshaling the resources of the federal government will be critical in our ability to reduce death and suffering from this disease,” she said.
The American Society for Radiation Oncology issued a press release, saying: “On behalf of radiation oncologists who treat people with cancer every day, we support the Biden-Harris administration’s move to drastically reduce the number of cancer deaths in the United States and improve the lives of people diagnosed with this disease.
“We believe the administration’s commitment to expand cancer prevention efforts and to increase equitable access to screenings and treatments will help mitigate some of the negative impact of the COVID-19 pandemic,” the society added.
At the American Association for Cancer Research, Chief Executive Officer Margaret Foti, MD, PhD, said she was thrilled to hear the announcement after the devastating interruptions in cancer research and patient care over the past 2 years.
“The reignited Cancer Moonshot will provide an important framework to help improve cancer prevention strategies, increase cancer screenings and early detection, reduce cancer disparities, and propel new lifesaving cures for patients with cancer,” she said.
However, increased funding from Congress will be needed for these goals to be achieved, she emphasized.
A version of this article first appeared on Medscape.com.
The “Cancer Moonshot” is about to be relaunched.
In a White House briefing, President Joe Biden announced that he is “reigniting” the initiative he spearheaded when he was vice president during the Obama administration.
During the livestreamed event, the president discussed his plans to bring a “fierce sense of urgency” to the fight against cancer and better support patients with cancer and their families.
He emphasized that cancer is one of the truly bipartisan issues. There is strong support from both “sides of the aisle,” he said, and he sees it as an issue that can bring the country together.
“We can do this. I promise you, we can do this. For all those we lost, for all those we miss. We can end cancer as we know it,” he said. “This is a presidential White House priority.”
The aim is to reduce the death rate from cancer by at least 50% over the next 25 years.
There is also a proposal to create the Advanced Research Projects Agency for Health, which would focus on driving cutting-edge innovation in health research.
Part of the plan is to assemble a “cancer cabinet” that includes 18 federal departments, agencies, and offices, including leaders from the departments of Health & Human Services, Veterans Affairs, Defense, Energy, and Agriculture.
At present, there are few details about the new program or how it will be funded.
Presumably more will be revealed at the Cancer Moonshot Summit being planned, as well as on a planned new website where people can track its progress.
President priority
Cancer Moonshot began back in 2016, when during his last State of the Union Address, former President Barack Obama announced the ambitious initiative. A few days later, Obama asked Congress for $1 billion to send cancer to the moon, and he put Biden, then vice president, in charge of “mission control” in the remaining months of the administration.
The new initiative will be headed by Danielle Carnival, PhD, who serves in the White House Office of Science and Technology Policy and has been appointed as White House Cancer Moonshot coordinator.
At the briefing, Mr. Biden and Vice President Kamala Harris spoke about losing family members to cancer. The president spoke about his eldest son, Beau, who died from brain cancer when he was 46 years old, while Ms. Harris spoke about her mother, Shyamala Gopalan, a breast cancer researcher who died of colon cancer in 2009.
Accolades but a bit of caution
The president’s speech was applauded by many cancer groups, both professional organizations and patient advocacy groups.
Karen E. Knudsen, PhD, chief executive officer of the American Cancer Society and its advocacy affiliate, the American Cancer Society Cancer Action Network, commended Mr. Biden for reigniting Cancer Moonshot.
“In 2022 alone, there will be an estimated 1.9 million people diagnosed with cancer and more than 600,000 people in the U.S. will die. Marshaling the resources of the federal government will be critical in our ability to reduce death and suffering from this disease,” she said.
The American Society for Radiation Oncology issued a press release, saying: “On behalf of radiation oncologists who treat people with cancer every day, we support the Biden-Harris administration’s move to drastically reduce the number of cancer deaths in the United States and improve the lives of people diagnosed with this disease.
“We believe the administration’s commitment to expand cancer prevention efforts and to increase equitable access to screenings and treatments will help mitigate some of the negative impact of the COVID-19 pandemic,” the society added.
At the American Association for Cancer Research, Chief Executive Officer Margaret Foti, MD, PhD, said she was thrilled to hear the announcement after the devastating interruptions in cancer research and patient care over the past 2 years.
“The reignited Cancer Moonshot will provide an important framework to help improve cancer prevention strategies, increase cancer screenings and early detection, reduce cancer disparities, and propel new lifesaving cures for patients with cancer,” she said.
However, increased funding from Congress will be needed for these goals to be achieved, she emphasized.
A version of this article first appeared on Medscape.com.
Endocrine Society and others to FDA: Restrict BPA
The chemical is used to make plastics in items such as food containers, pitchers, and inner linings of metal products. Small amounts of BPA can leak into food and beverages.
The petition points to a December 2021 report by the European Food Safety Authority titled: “Re-evaluation of the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs,” which summarizes evidence gathered since 2013.
It concludes that “there is a health concern from BPA exposure for all age groups.” Specific concerns include harm to the immune system and male and female reproductive systems.
Average American exposed to 5,000 times the safe level of BPA
The EFSA established a new “tolerable daily intake” of BPA of 0.04 ng/kg of body weight per day. By contrast, in 2014 the FDA estimated that the mean BPA intake for the U.S. population older than 2 years was 200 ng/kg bw/day and that the 90th percentile for BPA intake was 500 ng/kg of body weight per day.
“Using FDA’s own exposure estimates, the average American is exposed to more than 5000 times the safe level of 0.04 ng BPA/kg [body weight per day] set by the EFSA expert panel. Without a doubt, these values constitute a high health risk and support the conclusion that uses of BPA are not safe ... Given the magnitude of the overexposure, we request an expedited review by FDA,” the petition reads.
In addition to the Endocrine Society, which has long warned about the dangers of endocrine-disrupting chemicals, other signatories to the petition include the Environmental Defense Fund, Breast Cancer Prevention Partners, Clean Water Action/Clean Water Fund, Consumer Reports, Environmental Working Group, Healthy Babies Bright Futures, and the former director of the National Institute of Environmental Health Sciences and National Toxicology Program.
In a statement, Endocrine Society BPA expert Heather Patisaul, PhD, of North Carolina University, Raleigh, said the report’s findings “are extremely concerning and prove the point that even very low levels of BPA exposure can be harmful and lead to issues with reproductive health, breast cancer risk, behavior, and metabolism.”
“The FDA needs to acknowledge the science behind endocrine-disrupting chemicals and act accordingly to protect public health,” she urged.
The FDA is expected to decide within the next few days whether to open a docket to accept comments.
A final decision could take 6 months or longer, an Endocrine Society spokesperson told this news organization.
A version of this article first appeared on Medscape.com.
The chemical is used to make plastics in items such as food containers, pitchers, and inner linings of metal products. Small amounts of BPA can leak into food and beverages.
The petition points to a December 2021 report by the European Food Safety Authority titled: “Re-evaluation of the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs,” which summarizes evidence gathered since 2013.
It concludes that “there is a health concern from BPA exposure for all age groups.” Specific concerns include harm to the immune system and male and female reproductive systems.
Average American exposed to 5,000 times the safe level of BPA
The EFSA established a new “tolerable daily intake” of BPA of 0.04 ng/kg of body weight per day. By contrast, in 2014 the FDA estimated that the mean BPA intake for the U.S. population older than 2 years was 200 ng/kg bw/day and that the 90th percentile for BPA intake was 500 ng/kg of body weight per day.
“Using FDA’s own exposure estimates, the average American is exposed to more than 5000 times the safe level of 0.04 ng BPA/kg [body weight per day] set by the EFSA expert panel. Without a doubt, these values constitute a high health risk and support the conclusion that uses of BPA are not safe ... Given the magnitude of the overexposure, we request an expedited review by FDA,” the petition reads.
In addition to the Endocrine Society, which has long warned about the dangers of endocrine-disrupting chemicals, other signatories to the petition include the Environmental Defense Fund, Breast Cancer Prevention Partners, Clean Water Action/Clean Water Fund, Consumer Reports, Environmental Working Group, Healthy Babies Bright Futures, and the former director of the National Institute of Environmental Health Sciences and National Toxicology Program.
In a statement, Endocrine Society BPA expert Heather Patisaul, PhD, of North Carolina University, Raleigh, said the report’s findings “are extremely concerning and prove the point that even very low levels of BPA exposure can be harmful and lead to issues with reproductive health, breast cancer risk, behavior, and metabolism.”
“The FDA needs to acknowledge the science behind endocrine-disrupting chemicals and act accordingly to protect public health,” she urged.
The FDA is expected to decide within the next few days whether to open a docket to accept comments.
A final decision could take 6 months or longer, an Endocrine Society spokesperson told this news organization.
A version of this article first appeared on Medscape.com.
The chemical is used to make plastics in items such as food containers, pitchers, and inner linings of metal products. Small amounts of BPA can leak into food and beverages.
The petition points to a December 2021 report by the European Food Safety Authority titled: “Re-evaluation of the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs,” which summarizes evidence gathered since 2013.
It concludes that “there is a health concern from BPA exposure for all age groups.” Specific concerns include harm to the immune system and male and female reproductive systems.
Average American exposed to 5,000 times the safe level of BPA
The EFSA established a new “tolerable daily intake” of BPA of 0.04 ng/kg of body weight per day. By contrast, in 2014 the FDA estimated that the mean BPA intake for the U.S. population older than 2 years was 200 ng/kg bw/day and that the 90th percentile for BPA intake was 500 ng/kg of body weight per day.
“Using FDA’s own exposure estimates, the average American is exposed to more than 5000 times the safe level of 0.04 ng BPA/kg [body weight per day] set by the EFSA expert panel. Without a doubt, these values constitute a high health risk and support the conclusion that uses of BPA are not safe ... Given the magnitude of the overexposure, we request an expedited review by FDA,” the petition reads.
In addition to the Endocrine Society, which has long warned about the dangers of endocrine-disrupting chemicals, other signatories to the petition include the Environmental Defense Fund, Breast Cancer Prevention Partners, Clean Water Action/Clean Water Fund, Consumer Reports, Environmental Working Group, Healthy Babies Bright Futures, and the former director of the National Institute of Environmental Health Sciences and National Toxicology Program.
In a statement, Endocrine Society BPA expert Heather Patisaul, PhD, of North Carolina University, Raleigh, said the report’s findings “are extremely concerning and prove the point that even very low levels of BPA exposure can be harmful and lead to issues with reproductive health, breast cancer risk, behavior, and metabolism.”
“The FDA needs to acknowledge the science behind endocrine-disrupting chemicals and act accordingly to protect public health,” she urged.
The FDA is expected to decide within the next few days whether to open a docket to accept comments.
A final decision could take 6 months or longer, an Endocrine Society spokesperson told this news organization.
A version of this article first appeared on Medscape.com.
Native American Tribes Settle ‘Epic’ Opioid Deal
Hundreds of Native American tribes have tentatively settled in what one of the lead attorneys describes as “an epic deal”: The top 3 pharmaceutical distributors in the US and Johnson & Johnson have agreed to pay $665 million for deceptive marketing practices and overdistribution of opioids. Native Americans were among those hardest hit by the opioid epidemic. Between 2006 and 2014, Native Americans were nearly 50% more likely than non-Natives to die of an opioid overdose. In 2014, they ranked number 1 for death by opioid overdose.
Overprescribing was rampant. In some areas, such as southwestern Virginia, eastern Kentucky, and Alabama, prescriptions were 5 to 6 times higher than the national average. The overprescribing was largely due to massive and aggressive billion-dollar marketing campaigns, which misrepresented the safety of opioid medications. Purdue Pharma, for instance, trained sales representatives to claim that the risk of addiction was “less than 1 percent.” In an interview with Smithsonian Magazine, Caleb Alexander, MD, codirector of Johns Hopkins’ Center for Drug Safety and Effectiveness, said, “When I was in residency training, we were taught that one needn’t worry about the addictive potential of opioids if a patient had true pain.” He said it was no accident that physicians were cultivated to overestimate the effectiveness for chronic, noncancer pain while underestimating the risks.
Native Americans were not only in the target group for prescriptions, but also apparently singularly targeted. “We were preyed upon,” said Chickasaw Nation Governor Bill Anoatubby in the Washington Post. “It was unconscionable.” A Washington Post analysis found that, between 2006 and 2014, opioid distributors shipped an average of 36 pills per person in the US. States in the so-called opioid belt (mostly Southern states), received an average of 60 to 66 pills per person. The distributors shipped 57 pills per person to Oklahoma, home to nearly 322,000 Native Americans. (The opioid death rate for Native Americans in Oklahoma from 2006 to 2014 was more than triple the nationwide rate for non-Natives.) In South Dakota as recently as 2015, enough opioids were prescribed to medicate every adult around-the-clock for 19 consecutive days. Native Americans comprise 9% of South Dakota’s population; however, almost 30% of the patients are being treated for opioid use disorder.
In the settlement, which is a first for tribes, McKesson, Cardinal Health, and AmerisourceBergen would pay $515 million over 7 years. Johnson & Johnson would contribute $150 million in 2 years to the federally recognized tribes. “This settlement is a real turning point in history,” said Lloyd Miller, one of the attorneys representing one-third of the litigating tribes.
But the money is still small compensation for ravaging millions of lives. “Flooding the Native community with Western medicine—sedating a population rather than seeking to understand its needs and challenges—is not an acceptable means of handling its trauma,” the Lakota People’s Law Project says in an article on its website. Thus, the money dispersal will be overseen by a panel of tribal health experts, to go toward programs that aid drug users and their communities.
The funds will be managed in a way that will consider the long-term damage, Native American leaders vow. Children, for instance, have not been exempt from the sequelae of the overprescribing. Foster care systems are “overrun” with children of addicted parents, the Law Project says, and the children are placed in homes outside the tribe. “In the long run, this has the potential to curtail tribal membership, break down familial lines, and degrade cultural values.”
Dealing with the problem has drained tribal resources—doubly strained by the COVID-19 epidemic. Chairman Douglas Yankton, of the Spirit Lake Nation in North Dakota, said in a statement, “The dollars that will flow to Tribes under this initial settlement will help fund crucial, on-reservation, culturally appropriate opioid treatment services.”
However, Chairman Kristopher Peters, of the Squaxin Island Tribe in Washington State, told the Washington Post, “There is no amount of money that’s going to solve the generational issues that have been created from this. Our hope is that we can use these funds to help revitalize our culture and help heal our people.”
Johnson & Johnson says it no longer sells prescription opioids in the US
Hundreds of Native American tribes have tentatively settled in what one of the lead attorneys describes as “an epic deal”: The top 3 pharmaceutical distributors in the US and Johnson & Johnson have agreed to pay $665 million for deceptive marketing practices and overdistribution of opioids. Native Americans were among those hardest hit by the opioid epidemic. Between 2006 and 2014, Native Americans were nearly 50% more likely than non-Natives to die of an opioid overdose. In 2014, they ranked number 1 for death by opioid overdose.
Overprescribing was rampant. In some areas, such as southwestern Virginia, eastern Kentucky, and Alabama, prescriptions were 5 to 6 times higher than the national average. The overprescribing was largely due to massive and aggressive billion-dollar marketing campaigns, which misrepresented the safety of opioid medications. Purdue Pharma, for instance, trained sales representatives to claim that the risk of addiction was “less than 1 percent.” In an interview with Smithsonian Magazine, Caleb Alexander, MD, codirector of Johns Hopkins’ Center for Drug Safety and Effectiveness, said, “When I was in residency training, we were taught that one needn’t worry about the addictive potential of opioids if a patient had true pain.” He said it was no accident that physicians were cultivated to overestimate the effectiveness for chronic, noncancer pain while underestimating the risks.
Native Americans were not only in the target group for prescriptions, but also apparently singularly targeted. “We were preyed upon,” said Chickasaw Nation Governor Bill Anoatubby in the Washington Post. “It was unconscionable.” A Washington Post analysis found that, between 2006 and 2014, opioid distributors shipped an average of 36 pills per person in the US. States in the so-called opioid belt (mostly Southern states), received an average of 60 to 66 pills per person. The distributors shipped 57 pills per person to Oklahoma, home to nearly 322,000 Native Americans. (The opioid death rate for Native Americans in Oklahoma from 2006 to 2014 was more than triple the nationwide rate for non-Natives.) In South Dakota as recently as 2015, enough opioids were prescribed to medicate every adult around-the-clock for 19 consecutive days. Native Americans comprise 9% of South Dakota’s population; however, almost 30% of the patients are being treated for opioid use disorder.
In the settlement, which is a first for tribes, McKesson, Cardinal Health, and AmerisourceBergen would pay $515 million over 7 years. Johnson & Johnson would contribute $150 million in 2 years to the federally recognized tribes. “This settlement is a real turning point in history,” said Lloyd Miller, one of the attorneys representing one-third of the litigating tribes.
But the money is still small compensation for ravaging millions of lives. “Flooding the Native community with Western medicine—sedating a population rather than seeking to understand its needs and challenges—is not an acceptable means of handling its trauma,” the Lakota People’s Law Project says in an article on its website. Thus, the money dispersal will be overseen by a panel of tribal health experts, to go toward programs that aid drug users and their communities.
The funds will be managed in a way that will consider the long-term damage, Native American leaders vow. Children, for instance, have not been exempt from the sequelae of the overprescribing. Foster care systems are “overrun” with children of addicted parents, the Law Project says, and the children are placed in homes outside the tribe. “In the long run, this has the potential to curtail tribal membership, break down familial lines, and degrade cultural values.”
Dealing with the problem has drained tribal resources—doubly strained by the COVID-19 epidemic. Chairman Douglas Yankton, of the Spirit Lake Nation in North Dakota, said in a statement, “The dollars that will flow to Tribes under this initial settlement will help fund crucial, on-reservation, culturally appropriate opioid treatment services.”
However, Chairman Kristopher Peters, of the Squaxin Island Tribe in Washington State, told the Washington Post, “There is no amount of money that’s going to solve the generational issues that have been created from this. Our hope is that we can use these funds to help revitalize our culture and help heal our people.”
Johnson & Johnson says it no longer sells prescription opioids in the US
Hundreds of Native American tribes have tentatively settled in what one of the lead attorneys describes as “an epic deal”: The top 3 pharmaceutical distributors in the US and Johnson & Johnson have agreed to pay $665 million for deceptive marketing practices and overdistribution of opioids. Native Americans were among those hardest hit by the opioid epidemic. Between 2006 and 2014, Native Americans were nearly 50% more likely than non-Natives to die of an opioid overdose. In 2014, they ranked number 1 for death by opioid overdose.
Overprescribing was rampant. In some areas, such as southwestern Virginia, eastern Kentucky, and Alabama, prescriptions were 5 to 6 times higher than the national average. The overprescribing was largely due to massive and aggressive billion-dollar marketing campaigns, which misrepresented the safety of opioid medications. Purdue Pharma, for instance, trained sales representatives to claim that the risk of addiction was “less than 1 percent.” In an interview with Smithsonian Magazine, Caleb Alexander, MD, codirector of Johns Hopkins’ Center for Drug Safety and Effectiveness, said, “When I was in residency training, we were taught that one needn’t worry about the addictive potential of opioids if a patient had true pain.” He said it was no accident that physicians were cultivated to overestimate the effectiveness for chronic, noncancer pain while underestimating the risks.
Native Americans were not only in the target group for prescriptions, but also apparently singularly targeted. “We were preyed upon,” said Chickasaw Nation Governor Bill Anoatubby in the Washington Post. “It was unconscionable.” A Washington Post analysis found that, between 2006 and 2014, opioid distributors shipped an average of 36 pills per person in the US. States in the so-called opioid belt (mostly Southern states), received an average of 60 to 66 pills per person. The distributors shipped 57 pills per person to Oklahoma, home to nearly 322,000 Native Americans. (The opioid death rate for Native Americans in Oklahoma from 2006 to 2014 was more than triple the nationwide rate for non-Natives.) In South Dakota as recently as 2015, enough opioids were prescribed to medicate every adult around-the-clock for 19 consecutive days. Native Americans comprise 9% of South Dakota’s population; however, almost 30% of the patients are being treated for opioid use disorder.
In the settlement, which is a first for tribes, McKesson, Cardinal Health, and AmerisourceBergen would pay $515 million over 7 years. Johnson & Johnson would contribute $150 million in 2 years to the federally recognized tribes. “This settlement is a real turning point in history,” said Lloyd Miller, one of the attorneys representing one-third of the litigating tribes.
But the money is still small compensation for ravaging millions of lives. “Flooding the Native community with Western medicine—sedating a population rather than seeking to understand its needs and challenges—is not an acceptable means of handling its trauma,” the Lakota People’s Law Project says in an article on its website. Thus, the money dispersal will be overseen by a panel of tribal health experts, to go toward programs that aid drug users and their communities.
The funds will be managed in a way that will consider the long-term damage, Native American leaders vow. Children, for instance, have not been exempt from the sequelae of the overprescribing. Foster care systems are “overrun” with children of addicted parents, the Law Project says, and the children are placed in homes outside the tribe. “In the long run, this has the potential to curtail tribal membership, break down familial lines, and degrade cultural values.”
Dealing with the problem has drained tribal resources—doubly strained by the COVID-19 epidemic. Chairman Douglas Yankton, of the Spirit Lake Nation in North Dakota, said in a statement, “The dollars that will flow to Tribes under this initial settlement will help fund crucial, on-reservation, culturally appropriate opioid treatment services.”
However, Chairman Kristopher Peters, of the Squaxin Island Tribe in Washington State, told the Washington Post, “There is no amount of money that’s going to solve the generational issues that have been created from this. Our hope is that we can use these funds to help revitalize our culture and help heal our people.”
Johnson & Johnson says it no longer sells prescription opioids in the US
Community Care Program Lacks Essential Data for Health Care Decisions
In 2014, amidst stories of delays at Veterans Health Administration facilities, Congress established the Veterans Choice Program, which expanded access to private sector health care practitioners. When the program expired in 2018, lawmakers replaced it with the Veterans Community Care Program (VCCP) as part of the US Department of Veterans Affairs (VA) Maintaining Internal Systems and Strengthening Integrated Outside Networks Act (38 USC § 1703 MISSION Act). Since then, the VCCP has grown exponentially; 34% of current veteran health care visits are with private clinicians.
Along with broader private sector access, the MISSION Act also mandated the creation of quality-of-care standards for both VA and VCCP, and stipulated that data be compiled and made available to “provide covered veterans relevant comparative information to make informed decisions regarding their health care.” Two-and-a-half years later, data about the quality of VCCP care remains largely unknown.
Access to Care Website
In the lead up to the MISSION Act, the VA launched its Access to Care website, an online tool that publishes institutional performance data on key metrics so that veterans can make “more informed choices about where, when, and how they receive their health care.” Following the bill’s passage, the VA added a MISSION Act Quality Standards section, which includes results of 27 conventional quality measures for every VA facility. These scores are posted alongside data of regional facilities.
This trailblazing tool is exceedingly comprehensive. Yet, multiple website gaps compromise its utility for veterans deliberating whether to obtain VCCP care, including:
- Data isn’t about VCCP care. The hospitals are selected because they are local, not whether they participate in VCCP. Further, it appears that aggregate scores include non-VCCP facilities.
- Missing conditions/treatments. While the website contains quality scores for an ample range of procedures, it lacks information for many conditions that disproportionately affect veterans. A veteran with posttraumatic stress disorder (PTSD) or traumatic brain injury (TBI), for example, has no data to check.
- Skewed comparison population. Private sector practitioners primarily treat nonveteran patients, a population that is, on average, healthier and of higher socioeconomic status when compared with VA patients. Outcomes differ, for example, when patients have coexisting mental illness or homelessness. For VCCP scores to be beneficial for comparisons, they should derive from treated veterans or be accurately risk-adjusted.
- Tangential measures. The Institute of Medicine defined health care quality as “improvement of outcomes.” Patients considering health care options benefit from information about treatment effectiveness and symptom reduction. But because obtaining that quality data is labor intensive, proxy measures are substituted. For example, the measure advising smokers to quit is the closest the website comes to reporting on the quality of mental health care.
High-Performers
The VA initiated a second means to inform veterans about the quality of furnished care. Specifically, they guided third-party administrators (TPAs)—TriWest Healthcare Alliance and Optum—in creating algorithms designating that VCCP individual clinicians, practice groups, and hospitals can be deemed high performing providers (HPPs). The algorithms are calculated using a mix of Healthcare Effectiveness Data and Information Set (HEDIS), Physician Quality Reporting System (PQRS), and Blue Health Intelligence (BHI) primary and specialty care measures. The designations are intended to be accessible to local VA community care schedulers to connect veterans with HPPs.
Many aspects of the HPP system are not yet public, including the measures that comprise the algorithms and when the designations will become operational. From what is publicly discoverable about HPP designations, there are crucial gaps like those on the Access to Care website. Behavioral and mental health conditions, for instance, are intentionally excluded in HPP monitoring. HPP algorithms draw from care provided to the general population; an HPP’s patient panel may contain no veterans (with their common comorbidities) at all. Most limiting, there’s no expectation that VCCP clinicians be high performing. Of the 1.2 million program clinicians treating veterans as of November 2020, only a nominal 13.4% were HPP.
After studying the HPP system, VA Partnered Evidence-based Policy Resource Center acknowledged that “it remains unclear whether the quality metrics and referral system result in higher quality of care for VA patients or whether the program improves veteran health.”
Quality of VCCP Mental Health Treatment
The MISSION Act mandated the VA to “establish standards and requirements for the provision of care by non-VA health care practitioners in clinical areas for which the Department of Veterans Affairs has special expertise, including PTSD, military sexual trauma-related conditions (MST), and TBI.” This requirement arose from a recognition that mental health care provided in the private sector pales in comparison to the VA’s rigorous evidence-based training, consultation, case review and care delivery. For example, over 8500 VA clinicians have received training in evidence-based cognitive processing therapy and/or prolonged exposure therapy for PTSD.
The MISSION Act also mandated that VCCP providers must “fulfill training requirements established by the Secretary on how to deliver evidence-based treatments in the clinical areas for which the Department of Veterans Affairs has special expertise” before furnishing care pursuant to a contract with the VA. However, the VA elected to disregard the directive, and left it up to VCCP clinician’s discretion whether to obtain training or proficiency.
Two bills introduced in Congress in 2021 aim to uphold these vital mandates for the VCCP program. The Veterans’ Culturally Competent Care Act requires VCCP mental health practitioners to take courses on the evaluation and management of suicide, PTSD, TBI, and MST. The Lethal Means Safety Training Act aligns VCCP clinicians suicide prevention training with existing VA standards.
Recommendations to Assure the Quality of VCCP Care
With review and revision of VCCP quality standards now underway, the following remedial actions are recommended:
- VCCP metrics must be compiled using data on veterans’ care, not the general population, and be published on the Access to Care website. This indispensable information is published on the website for VA care but not for VCCP. Unless VCCP is required to track their veterans, apples-to-apples comparisons of quality of care will remain difficult to attain. Supplemental research that directly contrasts quality of VA to VCCP care should be posted. For example, a 2021 study of enrolled veterans brought by ambulance to VA or community emergency rooms found that all 170 VA medical centers had lower comparative death rates.
- VCCP providers should be held to the same quality standards as those applied to VA clinicians. In a 2020 critical issue update on implementation of the MISSION Act, major veterans service organizations (VSOs) recommended that competency, training, and quality standards for non-VA community clinicians must be equivalent to benchmarks expected of VA clinicians. That includes credentials, initial and follow-up training, diagnostic screening, care-delivery, and documentation standards. Enacting the Veterans’ Culturally Competent Care Act and the Lethal Means Safety Training Act would begin to meet the MISSION Act’s clear statutory language.
- The VA and VCCP should add quality information about major diagnostic categories. This will allow veterans to make informed decisions about their personal condition. For most health diagnoses, there is no searchable listing by disorder.
- Quality assessments should be realigned to focus on outcome measures. For prospective patients, outcome results provide the most meaningful basis for comparing and selecting clinicians. Proxy measures may have little bearing on whether veterans receive effective care. (As Albert Einstein’s famously observed, “Not everything that can be counted counts.”). Also, the specific measures used for a clinician’s HPP designation should be delineated.
- The VA must enforce the MISSION Act’s instruction to renew or cancel contracts based on demonstrated quality of care. As VSOs emphasized, “if the private sector is unwilling or unable to match the VA’s access and quality standards, the VA must consider whether it needs to find new community partners.”
Seventeen billion dollars is spent yearly on purchased health care whose quality remains indeterminate. Ironclad commitments are needed from Congress and the VA to ensure that the effectiveness of, and standards for, veterans care options in the private sector match that in the VA.
In 2014, amidst stories of delays at Veterans Health Administration facilities, Congress established the Veterans Choice Program, which expanded access to private sector health care practitioners. When the program expired in 2018, lawmakers replaced it with the Veterans Community Care Program (VCCP) as part of the US Department of Veterans Affairs (VA) Maintaining Internal Systems and Strengthening Integrated Outside Networks Act (38 USC § 1703 MISSION Act). Since then, the VCCP has grown exponentially; 34% of current veteran health care visits are with private clinicians.
Along with broader private sector access, the MISSION Act also mandated the creation of quality-of-care standards for both VA and VCCP, and stipulated that data be compiled and made available to “provide covered veterans relevant comparative information to make informed decisions regarding their health care.” Two-and-a-half years later, data about the quality of VCCP care remains largely unknown.
Access to Care Website
In the lead up to the MISSION Act, the VA launched its Access to Care website, an online tool that publishes institutional performance data on key metrics so that veterans can make “more informed choices about where, when, and how they receive their health care.” Following the bill’s passage, the VA added a MISSION Act Quality Standards section, which includes results of 27 conventional quality measures for every VA facility. These scores are posted alongside data of regional facilities.
This trailblazing tool is exceedingly comprehensive. Yet, multiple website gaps compromise its utility for veterans deliberating whether to obtain VCCP care, including:
- Data isn’t about VCCP care. The hospitals are selected because they are local, not whether they participate in VCCP. Further, it appears that aggregate scores include non-VCCP facilities.
- Missing conditions/treatments. While the website contains quality scores for an ample range of procedures, it lacks information for many conditions that disproportionately affect veterans. A veteran with posttraumatic stress disorder (PTSD) or traumatic brain injury (TBI), for example, has no data to check.
- Skewed comparison population. Private sector practitioners primarily treat nonveteran patients, a population that is, on average, healthier and of higher socioeconomic status when compared with VA patients. Outcomes differ, for example, when patients have coexisting mental illness or homelessness. For VCCP scores to be beneficial for comparisons, they should derive from treated veterans or be accurately risk-adjusted.
- Tangential measures. The Institute of Medicine defined health care quality as “improvement of outcomes.” Patients considering health care options benefit from information about treatment effectiveness and symptom reduction. But because obtaining that quality data is labor intensive, proxy measures are substituted. For example, the measure advising smokers to quit is the closest the website comes to reporting on the quality of mental health care.
High-Performers
The VA initiated a second means to inform veterans about the quality of furnished care. Specifically, they guided third-party administrators (TPAs)—TriWest Healthcare Alliance and Optum—in creating algorithms designating that VCCP individual clinicians, practice groups, and hospitals can be deemed high performing providers (HPPs). The algorithms are calculated using a mix of Healthcare Effectiveness Data and Information Set (HEDIS), Physician Quality Reporting System (PQRS), and Blue Health Intelligence (BHI) primary and specialty care measures. The designations are intended to be accessible to local VA community care schedulers to connect veterans with HPPs.
Many aspects of the HPP system are not yet public, including the measures that comprise the algorithms and when the designations will become operational. From what is publicly discoverable about HPP designations, there are crucial gaps like those on the Access to Care website. Behavioral and mental health conditions, for instance, are intentionally excluded in HPP monitoring. HPP algorithms draw from care provided to the general population; an HPP’s patient panel may contain no veterans (with their common comorbidities) at all. Most limiting, there’s no expectation that VCCP clinicians be high performing. Of the 1.2 million program clinicians treating veterans as of November 2020, only a nominal 13.4% were HPP.
After studying the HPP system, VA Partnered Evidence-based Policy Resource Center acknowledged that “it remains unclear whether the quality metrics and referral system result in higher quality of care for VA patients or whether the program improves veteran health.”
Quality of VCCP Mental Health Treatment
The MISSION Act mandated the VA to “establish standards and requirements for the provision of care by non-VA health care practitioners in clinical areas for which the Department of Veterans Affairs has special expertise, including PTSD, military sexual trauma-related conditions (MST), and TBI.” This requirement arose from a recognition that mental health care provided in the private sector pales in comparison to the VA’s rigorous evidence-based training, consultation, case review and care delivery. For example, over 8500 VA clinicians have received training in evidence-based cognitive processing therapy and/or prolonged exposure therapy for PTSD.
The MISSION Act also mandated that VCCP providers must “fulfill training requirements established by the Secretary on how to deliver evidence-based treatments in the clinical areas for which the Department of Veterans Affairs has special expertise” before furnishing care pursuant to a contract with the VA. However, the VA elected to disregard the directive, and left it up to VCCP clinician’s discretion whether to obtain training or proficiency.
Two bills introduced in Congress in 2021 aim to uphold these vital mandates for the VCCP program. The Veterans’ Culturally Competent Care Act requires VCCP mental health practitioners to take courses on the evaluation and management of suicide, PTSD, TBI, and MST. The Lethal Means Safety Training Act aligns VCCP clinicians suicide prevention training with existing VA standards.
Recommendations to Assure the Quality of VCCP Care
With review and revision of VCCP quality standards now underway, the following remedial actions are recommended:
- VCCP metrics must be compiled using data on veterans’ care, not the general population, and be published on the Access to Care website. This indispensable information is published on the website for VA care but not for VCCP. Unless VCCP is required to track their veterans, apples-to-apples comparisons of quality of care will remain difficult to attain. Supplemental research that directly contrasts quality of VA to VCCP care should be posted. For example, a 2021 study of enrolled veterans brought by ambulance to VA or community emergency rooms found that all 170 VA medical centers had lower comparative death rates.
- VCCP providers should be held to the same quality standards as those applied to VA clinicians. In a 2020 critical issue update on implementation of the MISSION Act, major veterans service organizations (VSOs) recommended that competency, training, and quality standards for non-VA community clinicians must be equivalent to benchmarks expected of VA clinicians. That includes credentials, initial and follow-up training, diagnostic screening, care-delivery, and documentation standards. Enacting the Veterans’ Culturally Competent Care Act and the Lethal Means Safety Training Act would begin to meet the MISSION Act’s clear statutory language.
- The VA and VCCP should add quality information about major diagnostic categories. This will allow veterans to make informed decisions about their personal condition. For most health diagnoses, there is no searchable listing by disorder.
- Quality assessments should be realigned to focus on outcome measures. For prospective patients, outcome results provide the most meaningful basis for comparing and selecting clinicians. Proxy measures may have little bearing on whether veterans receive effective care. (As Albert Einstein’s famously observed, “Not everything that can be counted counts.”). Also, the specific measures used for a clinician’s HPP designation should be delineated.
- The VA must enforce the MISSION Act’s instruction to renew or cancel contracts based on demonstrated quality of care. As VSOs emphasized, “if the private sector is unwilling or unable to match the VA’s access and quality standards, the VA must consider whether it needs to find new community partners.”
Seventeen billion dollars is spent yearly on purchased health care whose quality remains indeterminate. Ironclad commitments are needed from Congress and the VA to ensure that the effectiveness of, and standards for, veterans care options in the private sector match that in the VA.
In 2014, amidst stories of delays at Veterans Health Administration facilities, Congress established the Veterans Choice Program, which expanded access to private sector health care practitioners. When the program expired in 2018, lawmakers replaced it with the Veterans Community Care Program (VCCP) as part of the US Department of Veterans Affairs (VA) Maintaining Internal Systems and Strengthening Integrated Outside Networks Act (38 USC § 1703 MISSION Act). Since then, the VCCP has grown exponentially; 34% of current veteran health care visits are with private clinicians.
Along with broader private sector access, the MISSION Act also mandated the creation of quality-of-care standards for both VA and VCCP, and stipulated that data be compiled and made available to “provide covered veterans relevant comparative information to make informed decisions regarding their health care.” Two-and-a-half years later, data about the quality of VCCP care remains largely unknown.
Access to Care Website
In the lead up to the MISSION Act, the VA launched its Access to Care website, an online tool that publishes institutional performance data on key metrics so that veterans can make “more informed choices about where, when, and how they receive their health care.” Following the bill’s passage, the VA added a MISSION Act Quality Standards section, which includes results of 27 conventional quality measures for every VA facility. These scores are posted alongside data of regional facilities.
This trailblazing tool is exceedingly comprehensive. Yet, multiple website gaps compromise its utility for veterans deliberating whether to obtain VCCP care, including:
- Data isn’t about VCCP care. The hospitals are selected because they are local, not whether they participate in VCCP. Further, it appears that aggregate scores include non-VCCP facilities.
- Missing conditions/treatments. While the website contains quality scores for an ample range of procedures, it lacks information for many conditions that disproportionately affect veterans. A veteran with posttraumatic stress disorder (PTSD) or traumatic brain injury (TBI), for example, has no data to check.
- Skewed comparison population. Private sector practitioners primarily treat nonveteran patients, a population that is, on average, healthier and of higher socioeconomic status when compared with VA patients. Outcomes differ, for example, when patients have coexisting mental illness or homelessness. For VCCP scores to be beneficial for comparisons, they should derive from treated veterans or be accurately risk-adjusted.
- Tangential measures. The Institute of Medicine defined health care quality as “improvement of outcomes.” Patients considering health care options benefit from information about treatment effectiveness and symptom reduction. But because obtaining that quality data is labor intensive, proxy measures are substituted. For example, the measure advising smokers to quit is the closest the website comes to reporting on the quality of mental health care.
High-Performers
The VA initiated a second means to inform veterans about the quality of furnished care. Specifically, they guided third-party administrators (TPAs)—TriWest Healthcare Alliance and Optum—in creating algorithms designating that VCCP individual clinicians, practice groups, and hospitals can be deemed high performing providers (HPPs). The algorithms are calculated using a mix of Healthcare Effectiveness Data and Information Set (HEDIS), Physician Quality Reporting System (PQRS), and Blue Health Intelligence (BHI) primary and specialty care measures. The designations are intended to be accessible to local VA community care schedulers to connect veterans with HPPs.
Many aspects of the HPP system are not yet public, including the measures that comprise the algorithms and when the designations will become operational. From what is publicly discoverable about HPP designations, there are crucial gaps like those on the Access to Care website. Behavioral and mental health conditions, for instance, are intentionally excluded in HPP monitoring. HPP algorithms draw from care provided to the general population; an HPP’s patient panel may contain no veterans (with their common comorbidities) at all. Most limiting, there’s no expectation that VCCP clinicians be high performing. Of the 1.2 million program clinicians treating veterans as of November 2020, only a nominal 13.4% were HPP.
After studying the HPP system, VA Partnered Evidence-based Policy Resource Center acknowledged that “it remains unclear whether the quality metrics and referral system result in higher quality of care for VA patients or whether the program improves veteran health.”
Quality of VCCP Mental Health Treatment
The MISSION Act mandated the VA to “establish standards and requirements for the provision of care by non-VA health care practitioners in clinical areas for which the Department of Veterans Affairs has special expertise, including PTSD, military sexual trauma-related conditions (MST), and TBI.” This requirement arose from a recognition that mental health care provided in the private sector pales in comparison to the VA’s rigorous evidence-based training, consultation, case review and care delivery. For example, over 8500 VA clinicians have received training in evidence-based cognitive processing therapy and/or prolonged exposure therapy for PTSD.
The MISSION Act also mandated that VCCP providers must “fulfill training requirements established by the Secretary on how to deliver evidence-based treatments in the clinical areas for which the Department of Veterans Affairs has special expertise” before furnishing care pursuant to a contract with the VA. However, the VA elected to disregard the directive, and left it up to VCCP clinician’s discretion whether to obtain training or proficiency.
Two bills introduced in Congress in 2021 aim to uphold these vital mandates for the VCCP program. The Veterans’ Culturally Competent Care Act requires VCCP mental health practitioners to take courses on the evaluation and management of suicide, PTSD, TBI, and MST. The Lethal Means Safety Training Act aligns VCCP clinicians suicide prevention training with existing VA standards.
Recommendations to Assure the Quality of VCCP Care
With review and revision of VCCP quality standards now underway, the following remedial actions are recommended:
- VCCP metrics must be compiled using data on veterans’ care, not the general population, and be published on the Access to Care website. This indispensable information is published on the website for VA care but not for VCCP. Unless VCCP is required to track their veterans, apples-to-apples comparisons of quality of care will remain difficult to attain. Supplemental research that directly contrasts quality of VA to VCCP care should be posted. For example, a 2021 study of enrolled veterans brought by ambulance to VA or community emergency rooms found that all 170 VA medical centers had lower comparative death rates.
- VCCP providers should be held to the same quality standards as those applied to VA clinicians. In a 2020 critical issue update on implementation of the MISSION Act, major veterans service organizations (VSOs) recommended that competency, training, and quality standards for non-VA community clinicians must be equivalent to benchmarks expected of VA clinicians. That includes credentials, initial and follow-up training, diagnostic screening, care-delivery, and documentation standards. Enacting the Veterans’ Culturally Competent Care Act and the Lethal Means Safety Training Act would begin to meet the MISSION Act’s clear statutory language.
- The VA and VCCP should add quality information about major diagnostic categories. This will allow veterans to make informed decisions about their personal condition. For most health diagnoses, there is no searchable listing by disorder.
- Quality assessments should be realigned to focus on outcome measures. For prospective patients, outcome results provide the most meaningful basis for comparing and selecting clinicians. Proxy measures may have little bearing on whether veterans receive effective care. (As Albert Einstein’s famously observed, “Not everything that can be counted counts.”). Also, the specific measures used for a clinician’s HPP designation should be delineated.
- The VA must enforce the MISSION Act’s instruction to renew or cancel contracts based on demonstrated quality of care. As VSOs emphasized, “if the private sector is unwilling or unable to match the VA’s access and quality standards, the VA must consider whether it needs to find new community partners.”
Seventeen billion dollars is spent yearly on purchased health care whose quality remains indeterminate. Ironclad commitments are needed from Congress and the VA to ensure that the effectiveness of, and standards for, veterans care options in the private sector match that in the VA.
What Federal Practitioners Need to Know About the National Practitioner Data Bank
Not all federal practitioners know about the National Practitioner Data Bank (NPDB), a federal web-based repository of reports containing information on medical malpractice payments and certain adverse actions related to health care practitioners, providers, and suppliers. This article explains how NPDB statutes and regulations specifically affect federal health care practitioners, which may differ from how the rules affect practitioners in the private sector.1
National Practitioner Data Bank
Established by Congress in 1986, the NPDB contains information health care organizations need to make informed decisions about the health care practitionerss they license, credential, and hire. Federal regulations authorize eligible entities, including government agencies, to report to and query the NPDB. Individuals and organizations that are subjects of these reports have access to their own information. The reports are confidential and not available to the public. The NPDB currently contains > 1.6 million reports.2
Federal Agencies Queries
A query is a search for information in the NPDB regarding a health care practitioners or organization. Some federal agencies are permitted to query the NPDB, and all hospitals, including federal hospitals, are required to query. Agencies administering government health care programs (including private entities administering such programs under contract), federal law enforcement officials and agencies, and federal agencies responsible for the licensing or certification of health care practitioners, health care providers, or health care suppliers may query NPDB. Information received in response to queries includes, among other actions, licensure and certification actions taken by states, medical malpractice payment information, federal licensing and certification actions, and adverse privileging actions.3
Federal Reporting Requirements
Federal government agencies must report exclusions (described below), adjudicated actions, civil judgments, and criminal convictions concerning health care practitioners, providers, or suppliers. The following provides detailed information about the actions federal government agencies are required to report.
Adjudicated Actions or Decisions
Adjudicated actions or decisions are formal or official final actions.3 They include, but are not limited to, personnel-related actions such as suspensions without pay, reductions in pay, reductions in grade for cause, terminations, or other comparable actions. To be reportable, adjudicated actions or decisions must include due process mechanisms. Whether the subject of a report elects not to use the due process mechanism is immaterial as long as such a process is available to the subject before the adjudicated action or decision is made final. In general, if an adjudicated action or decision follows an agency’s established administrative procedures and those procedures ensure that due process is available to the subject, the due process requirement is satisfied. This definition specifically excludes clinical privileging actions taken by federal government agencies, which are described in appropriate memorandums of understanding.
Exclusions
An exclusion is a temporary or permanent debarment of an individual or organization from participation in a federal health-related program, such that items or services furnished by the individual or organization will not be reimbursed under the federal program.3
Civil Judgments and Criminal Convictions
Health care–related civil judgments and settlements must be reported.However, settlements in which no findings of liability have been made are not reportable.3 Health care–related criminal convictions prosecuted by federal government agencies in federal court must be reported to the NPDB. Pleas of guilt and nolo contendere, or no contest, by individuals or organizations also are reportable.3
In addition, final adverse licensure and certification actions are those taken against health care practitioners, providers, or suppliers, regardless of whether the final adverse action is the subject of a pending appeal.3 These must be reported.
Additional Reporting Requirements
Federal hospitals or federal government agencies administering health care services may have additional reporting requirements besides reporting adjudicated actions, exclusions, civil judgments, and criminal convictions. They may include submitting reports under a memorandum of understanding on clinical privileges actions and medical malpractice payments.3 The US Department of Health and Human Services (HHS) has entered into memorandums of understanding with the US Department of Defense and the US Department of Veteran Affairs to ensure their participation in the NPDB system. Federal hospitals should refer to applicable memorandums of understanding and agency-specific policies for guidance on carrying out their reporting responsibilities.4
Responding to a Report
The NPDB sends a letter to health care practitioners when an organization submits a report about the practitioner. The letter has the report number and a password is required to view the report.2 Health care practitioners also can order a self-query online to view any reports on them in the NPDB.
The subject of the report can also add a statement and dispute the report. The statement is an opportunity to provide additional information the subject would like to have included in the report. If the subject disagrees with the accuracy of a report or believes it does not meet NPDB reporting requirements, it can be disputed. The dispute will become part of the report. When the subject adds a statement or dispute, the NPDB notifies the reporting organization and all organizations that received the report within the previous 3 years of the report activity.
Health care practitioners must contact the reporting organization to try to resolve their dispute. If the subject of the report has contacted or tried to contact the reporting organization and could not resolve the dispute after 60 days, or if, within the 60-day period, the organization informs the subject that it will not modify the report, that individual may request dispute resolution.Requesting dispute resolution does not remove the report from the NPDB.
Dispute Resolution
Dispute resolution is a request for the HHS secretary to review the report. The secretary authorizes the Division of Practitioner Data Bank (DPDB) to conduct this review. The DPDB is responsible for oversight of the NPDB. The subject of the report will need to submit relevant supporting documentation to request dispute resolution. This documentation should show that the information in the report is not accurate or that the action is not reportable. Also, proof should be included that the subject contacted or attempted to contact the reporting organization. Submitting large volumes or extraneous documentation can delay the review process.
A dispute resolution manager will review the case and send the reporting organization a request for information if needed. The DPDB will send the subject of the report a courtesy copy of all correspondence. The dispute resolution timeline varies, as the DPDB reviews disputes in the order they are received. It completes a fair and thorough review based on the unique circumstances of each case and will review the case as soon as possible. Once the DPDB receives documentation from the subject and the reporting organization, it reviews the documentation to determine whether the report accurately reflects the record.
The DPDB decides to either maintain the report as is, correct it, or remove it from the NPDB. Once the process is complete, the dispute resolution manager sends a decision letter to the subject of the report and the reporting organization. The dispute resolution decision will appear in the report.
Regulations strictly limit the DPDB’s jurisdiction for reviewing disputed reports. It may only review the following: whether the report was submitted in accordance with reporting requirements, whether the reporting organization was eligible to report the information, and whether the report accurately depicts the action taken by the reporting organization and the basis for the action the reporting organization cited, as shown in the organization’s written record. The subject of the report must resolve any other issues with the reporting organization.
Under the dispute resolution review process, the DPDB cannot conduct an independent review of the merits of the action taken by the reporting organization, review the due process provided by the organization, or substitute its judgment for that of the reporting organization.2 The DPDB does not examine whether the subject of a report was informed of an ongoing investigation. The DPDB does not examine civil rights issues such as claims of discrimination or harassment in the work environment. Practitioners can find additional information at www.npdb.hrsa.gov.
1. US Department of Health and Human Services, National Practitioner Data Bank. NPDB guidebook. Updated October 2018. Accessed December 16, 2021. https://www.npdb.hrsa.gov/resources/aboutGuidebooks.jsp
2. US Department of Health and Human Services, National Practitioner Data Bank. A practitioner’s guide to the NPDB. Updated February 2021. Accessed December 16, 2021. https://www.npdb.hrsa.gov/pract/practGuide.jsp
3. US Department of Health and Human Services, National Practitioner Data Bank. Federal hospitals and federal government agencies. Accessed December 16, 2021. https://www.npdb.hrsa.gov/orgs/federalAgencies.jsp
4. US Department of Health and Human Services, National Practitioner Data Bank. Federal hospitals. Accessed December 16, 2021. https://www.npdb.hrsa.gov/orgs/federalHospitals.jsp
Not all federal practitioners know about the National Practitioner Data Bank (NPDB), a federal web-based repository of reports containing information on medical malpractice payments and certain adverse actions related to health care practitioners, providers, and suppliers. This article explains how NPDB statutes and regulations specifically affect federal health care practitioners, which may differ from how the rules affect practitioners in the private sector.1
National Practitioner Data Bank
Established by Congress in 1986, the NPDB contains information health care organizations need to make informed decisions about the health care practitionerss they license, credential, and hire. Federal regulations authorize eligible entities, including government agencies, to report to and query the NPDB. Individuals and organizations that are subjects of these reports have access to their own information. The reports are confidential and not available to the public. The NPDB currently contains > 1.6 million reports.2
Federal Agencies Queries
A query is a search for information in the NPDB regarding a health care practitioners or organization. Some federal agencies are permitted to query the NPDB, and all hospitals, including federal hospitals, are required to query. Agencies administering government health care programs (including private entities administering such programs under contract), federal law enforcement officials and agencies, and federal agencies responsible for the licensing or certification of health care practitioners, health care providers, or health care suppliers may query NPDB. Information received in response to queries includes, among other actions, licensure and certification actions taken by states, medical malpractice payment information, federal licensing and certification actions, and adverse privileging actions.3
Federal Reporting Requirements
Federal government agencies must report exclusions (described below), adjudicated actions, civil judgments, and criminal convictions concerning health care practitioners, providers, or suppliers. The following provides detailed information about the actions federal government agencies are required to report.
Adjudicated Actions or Decisions
Adjudicated actions or decisions are formal or official final actions.3 They include, but are not limited to, personnel-related actions such as suspensions without pay, reductions in pay, reductions in grade for cause, terminations, or other comparable actions. To be reportable, adjudicated actions or decisions must include due process mechanisms. Whether the subject of a report elects not to use the due process mechanism is immaterial as long as such a process is available to the subject before the adjudicated action or decision is made final. In general, if an adjudicated action or decision follows an agency’s established administrative procedures and those procedures ensure that due process is available to the subject, the due process requirement is satisfied. This definition specifically excludes clinical privileging actions taken by federal government agencies, which are described in appropriate memorandums of understanding.
Exclusions
An exclusion is a temporary or permanent debarment of an individual or organization from participation in a federal health-related program, such that items or services furnished by the individual or organization will not be reimbursed under the federal program.3
Civil Judgments and Criminal Convictions
Health care–related civil judgments and settlements must be reported.However, settlements in which no findings of liability have been made are not reportable.3 Health care–related criminal convictions prosecuted by federal government agencies in federal court must be reported to the NPDB. Pleas of guilt and nolo contendere, or no contest, by individuals or organizations also are reportable.3
In addition, final adverse licensure and certification actions are those taken against health care practitioners, providers, or suppliers, regardless of whether the final adverse action is the subject of a pending appeal.3 These must be reported.
Additional Reporting Requirements
Federal hospitals or federal government agencies administering health care services may have additional reporting requirements besides reporting adjudicated actions, exclusions, civil judgments, and criminal convictions. They may include submitting reports under a memorandum of understanding on clinical privileges actions and medical malpractice payments.3 The US Department of Health and Human Services (HHS) has entered into memorandums of understanding with the US Department of Defense and the US Department of Veteran Affairs to ensure their participation in the NPDB system. Federal hospitals should refer to applicable memorandums of understanding and agency-specific policies for guidance on carrying out their reporting responsibilities.4
Responding to a Report
The NPDB sends a letter to health care practitioners when an organization submits a report about the practitioner. The letter has the report number and a password is required to view the report.2 Health care practitioners also can order a self-query online to view any reports on them in the NPDB.
The subject of the report can also add a statement and dispute the report. The statement is an opportunity to provide additional information the subject would like to have included in the report. If the subject disagrees with the accuracy of a report or believes it does not meet NPDB reporting requirements, it can be disputed. The dispute will become part of the report. When the subject adds a statement or dispute, the NPDB notifies the reporting organization and all organizations that received the report within the previous 3 years of the report activity.
Health care practitioners must contact the reporting organization to try to resolve their dispute. If the subject of the report has contacted or tried to contact the reporting organization and could not resolve the dispute after 60 days, or if, within the 60-day period, the organization informs the subject that it will not modify the report, that individual may request dispute resolution.Requesting dispute resolution does not remove the report from the NPDB.
Dispute Resolution
Dispute resolution is a request for the HHS secretary to review the report. The secretary authorizes the Division of Practitioner Data Bank (DPDB) to conduct this review. The DPDB is responsible for oversight of the NPDB. The subject of the report will need to submit relevant supporting documentation to request dispute resolution. This documentation should show that the information in the report is not accurate or that the action is not reportable. Also, proof should be included that the subject contacted or attempted to contact the reporting organization. Submitting large volumes or extraneous documentation can delay the review process.
A dispute resolution manager will review the case and send the reporting organization a request for information if needed. The DPDB will send the subject of the report a courtesy copy of all correspondence. The dispute resolution timeline varies, as the DPDB reviews disputes in the order they are received. It completes a fair and thorough review based on the unique circumstances of each case and will review the case as soon as possible. Once the DPDB receives documentation from the subject and the reporting organization, it reviews the documentation to determine whether the report accurately reflects the record.
The DPDB decides to either maintain the report as is, correct it, or remove it from the NPDB. Once the process is complete, the dispute resolution manager sends a decision letter to the subject of the report and the reporting organization. The dispute resolution decision will appear in the report.
Regulations strictly limit the DPDB’s jurisdiction for reviewing disputed reports. It may only review the following: whether the report was submitted in accordance with reporting requirements, whether the reporting organization was eligible to report the information, and whether the report accurately depicts the action taken by the reporting organization and the basis for the action the reporting organization cited, as shown in the organization’s written record. The subject of the report must resolve any other issues with the reporting organization.
Under the dispute resolution review process, the DPDB cannot conduct an independent review of the merits of the action taken by the reporting organization, review the due process provided by the organization, or substitute its judgment for that of the reporting organization.2 The DPDB does not examine whether the subject of a report was informed of an ongoing investigation. The DPDB does not examine civil rights issues such as claims of discrimination or harassment in the work environment. Practitioners can find additional information at www.npdb.hrsa.gov.
Not all federal practitioners know about the National Practitioner Data Bank (NPDB), a federal web-based repository of reports containing information on medical malpractice payments and certain adverse actions related to health care practitioners, providers, and suppliers. This article explains how NPDB statutes and regulations specifically affect federal health care practitioners, which may differ from how the rules affect practitioners in the private sector.1
National Practitioner Data Bank
Established by Congress in 1986, the NPDB contains information health care organizations need to make informed decisions about the health care practitionerss they license, credential, and hire. Federal regulations authorize eligible entities, including government agencies, to report to and query the NPDB. Individuals and organizations that are subjects of these reports have access to their own information. The reports are confidential and not available to the public. The NPDB currently contains > 1.6 million reports.2
Federal Agencies Queries
A query is a search for information in the NPDB regarding a health care practitioners or organization. Some federal agencies are permitted to query the NPDB, and all hospitals, including federal hospitals, are required to query. Agencies administering government health care programs (including private entities administering such programs under contract), federal law enforcement officials and agencies, and federal agencies responsible for the licensing or certification of health care practitioners, health care providers, or health care suppliers may query NPDB. Information received in response to queries includes, among other actions, licensure and certification actions taken by states, medical malpractice payment information, federal licensing and certification actions, and adverse privileging actions.3
Federal Reporting Requirements
Federal government agencies must report exclusions (described below), adjudicated actions, civil judgments, and criminal convictions concerning health care practitioners, providers, or suppliers. The following provides detailed information about the actions federal government agencies are required to report.
Adjudicated Actions or Decisions
Adjudicated actions or decisions are formal or official final actions.3 They include, but are not limited to, personnel-related actions such as suspensions without pay, reductions in pay, reductions in grade for cause, terminations, or other comparable actions. To be reportable, adjudicated actions or decisions must include due process mechanisms. Whether the subject of a report elects not to use the due process mechanism is immaterial as long as such a process is available to the subject before the adjudicated action or decision is made final. In general, if an adjudicated action or decision follows an agency’s established administrative procedures and those procedures ensure that due process is available to the subject, the due process requirement is satisfied. This definition specifically excludes clinical privileging actions taken by federal government agencies, which are described in appropriate memorandums of understanding.
Exclusions
An exclusion is a temporary or permanent debarment of an individual or organization from participation in a federal health-related program, such that items or services furnished by the individual or organization will not be reimbursed under the federal program.3
Civil Judgments and Criminal Convictions
Health care–related civil judgments and settlements must be reported.However, settlements in which no findings of liability have been made are not reportable.3 Health care–related criminal convictions prosecuted by federal government agencies in federal court must be reported to the NPDB. Pleas of guilt and nolo contendere, or no contest, by individuals or organizations also are reportable.3
In addition, final adverse licensure and certification actions are those taken against health care practitioners, providers, or suppliers, regardless of whether the final adverse action is the subject of a pending appeal.3 These must be reported.
Additional Reporting Requirements
Federal hospitals or federal government agencies administering health care services may have additional reporting requirements besides reporting adjudicated actions, exclusions, civil judgments, and criminal convictions. They may include submitting reports under a memorandum of understanding on clinical privileges actions and medical malpractice payments.3 The US Department of Health and Human Services (HHS) has entered into memorandums of understanding with the US Department of Defense and the US Department of Veteran Affairs to ensure their participation in the NPDB system. Federal hospitals should refer to applicable memorandums of understanding and agency-specific policies for guidance on carrying out their reporting responsibilities.4
Responding to a Report
The NPDB sends a letter to health care practitioners when an organization submits a report about the practitioner. The letter has the report number and a password is required to view the report.2 Health care practitioners also can order a self-query online to view any reports on them in the NPDB.
The subject of the report can also add a statement and dispute the report. The statement is an opportunity to provide additional information the subject would like to have included in the report. If the subject disagrees with the accuracy of a report or believes it does not meet NPDB reporting requirements, it can be disputed. The dispute will become part of the report. When the subject adds a statement or dispute, the NPDB notifies the reporting organization and all organizations that received the report within the previous 3 years of the report activity.
Health care practitioners must contact the reporting organization to try to resolve their dispute. If the subject of the report has contacted or tried to contact the reporting organization and could not resolve the dispute after 60 days, or if, within the 60-day period, the organization informs the subject that it will not modify the report, that individual may request dispute resolution.Requesting dispute resolution does not remove the report from the NPDB.
Dispute Resolution
Dispute resolution is a request for the HHS secretary to review the report. The secretary authorizes the Division of Practitioner Data Bank (DPDB) to conduct this review. The DPDB is responsible for oversight of the NPDB. The subject of the report will need to submit relevant supporting documentation to request dispute resolution. This documentation should show that the information in the report is not accurate or that the action is not reportable. Also, proof should be included that the subject contacted or attempted to contact the reporting organization. Submitting large volumes or extraneous documentation can delay the review process.
A dispute resolution manager will review the case and send the reporting organization a request for information if needed. The DPDB will send the subject of the report a courtesy copy of all correspondence. The dispute resolution timeline varies, as the DPDB reviews disputes in the order they are received. It completes a fair and thorough review based on the unique circumstances of each case and will review the case as soon as possible. Once the DPDB receives documentation from the subject and the reporting organization, it reviews the documentation to determine whether the report accurately reflects the record.
The DPDB decides to either maintain the report as is, correct it, or remove it from the NPDB. Once the process is complete, the dispute resolution manager sends a decision letter to the subject of the report and the reporting organization. The dispute resolution decision will appear in the report.
Regulations strictly limit the DPDB’s jurisdiction for reviewing disputed reports. It may only review the following: whether the report was submitted in accordance with reporting requirements, whether the reporting organization was eligible to report the information, and whether the report accurately depicts the action taken by the reporting organization and the basis for the action the reporting organization cited, as shown in the organization’s written record. The subject of the report must resolve any other issues with the reporting organization.
Under the dispute resolution review process, the DPDB cannot conduct an independent review of the merits of the action taken by the reporting organization, review the due process provided by the organization, or substitute its judgment for that of the reporting organization.2 The DPDB does not examine whether the subject of a report was informed of an ongoing investigation. The DPDB does not examine civil rights issues such as claims of discrimination or harassment in the work environment. Practitioners can find additional information at www.npdb.hrsa.gov.
1. US Department of Health and Human Services, National Practitioner Data Bank. NPDB guidebook. Updated October 2018. Accessed December 16, 2021. https://www.npdb.hrsa.gov/resources/aboutGuidebooks.jsp
2. US Department of Health and Human Services, National Practitioner Data Bank. A practitioner’s guide to the NPDB. Updated February 2021. Accessed December 16, 2021. https://www.npdb.hrsa.gov/pract/practGuide.jsp
3. US Department of Health and Human Services, National Practitioner Data Bank. Federal hospitals and federal government agencies. Accessed December 16, 2021. https://www.npdb.hrsa.gov/orgs/federalAgencies.jsp
4. US Department of Health and Human Services, National Practitioner Data Bank. Federal hospitals. Accessed December 16, 2021. https://www.npdb.hrsa.gov/orgs/federalHospitals.jsp
1. US Department of Health and Human Services, National Practitioner Data Bank. NPDB guidebook. Updated October 2018. Accessed December 16, 2021. https://www.npdb.hrsa.gov/resources/aboutGuidebooks.jsp
2. US Department of Health and Human Services, National Practitioner Data Bank. A practitioner’s guide to the NPDB. Updated February 2021. Accessed December 16, 2021. https://www.npdb.hrsa.gov/pract/practGuide.jsp
3. US Department of Health and Human Services, National Practitioner Data Bank. Federal hospitals and federal government agencies. Accessed December 16, 2021. https://www.npdb.hrsa.gov/orgs/federalAgencies.jsp
4. US Department of Health and Human Services, National Practitioner Data Bank. Federal hospitals. Accessed December 16, 2021. https://www.npdb.hrsa.gov/orgs/federalHospitals.jsp
The Balance of Truth-Telling and Respect for Confidentiality: The Ethics of Case Reports
Medical case reports are as old as the healing profession itself.1 These ancient medical stories have a modern definition: “A case report is a narrative that describes, for medical, scientific or educational purposes, a medical problem experienced by one or more patients.”2 Case report experts describe the 3-fold purposes of this type of research: as a mainstay of education; a harbinger of emerging illnesses; and an appraiser of new interventions. Case-based education has long been a pillar of health professions education: Nurses, doctors, and allied health professionals are taught and learn through reading and discussing with their teachers and each other about cases of their own patients and of those in the literature.3 Case reports also have helped identify and raise awareness of new diseases and rare conditions, such as HIV.4 Finally, case reports have alerted regulatory agencies and the medical community about medication adverse effects, such as birth defects from thalidomide.5
Case reports also have been criticized on both scientific and ethical grounds. Critics argue that many case reports often lack the rigor and consistency of other types of research.6 Three recent trends in medical publication have strengthened the validity of these criticisms: the increase in the popularity of case reports; the corresponding increase in submissions to journals, including Federal Practitioner; and the rise of predatory publishers.7,8
The ethical scrutiny of case reports discussed in this column focuses on the tension between providing readers with adequate, accurate information to fulfil the goals of case reports while also protecting patient confidentiality. The latter issue during most of the history of medicine was not considered by health care professionals when the prevailing paternalism supported a professional-oriented approach to health care. The rise of bioethics in the 1960s and 1970s began the shift toward patient autonomy in medical decision making and patient rights to control their protected health information that rendered case reports ethically problematic.
To address both changes in ethical standards and scientific limitations, a committee of clinicians, researchers, and journal editors formed the Case Report (CARE) group.2,8 The group undertook an effort to improve the quality of case reports. From 2011 to 2012, they developed the CARE guidelines for clinical case reporting. The guidance took the form of a Statement and Checklist presented at the 2013 International Congress on Peer Review and Biomedical Publication. Since their presentation, multiple prestigious medical journals in many countries have implemented these recommendations.
As part of an overall effort to raise the ethical caliber of our own journal, Federal Practitioner will begin to implement the CARE guidelines for case reports for all future submissions. Use of the CARE recommendations will help prospective authors enhance the scientific value and ethical caliber of case reports submitted to the journal as well as assist the Federal Practitioner editorial team, editorial board, and peer reviewers to evaluate submissions more judiciously.
An essential part of the CARE guidelines is that the patient who is the subject of the case report provide informed consent for the publication of their personal narrative. The CARE group considers this an “ethical duty” of authors and editors alike. In “exceptional circumstances” such as if the patient is a minor or permanently incapacitated, a guardian or relative may grant consent. In the rare event that even with exhaustive attempts, if informed consent cannot be obtained from a patient or their representative, then the authors of the case report must submit a statement to this effect.4 Some journals may require that the authors obtain the approval of an institutional review board or the permission of an ethics or other institutional committee or a privacy officer.2
Requesting the patient’s consent is an extension of the shared decision making that is now a best practice in clinical care into the arena of research, making the patient or their representative a partner in the work. Ethicists have recommended inviting patients or relatives to read a draft of the case report and agree to its publication or request specific modifications to the manuscript. The CARE group rightly points out that with the rise of open notes in medical documentation, patients increasingly have access to their charts in near or real time.2 Gone are the days of Sir William Osler when only doctors read medical journals and all of these technical developments as well as standards of research and social changes in the practitioner-patient relationship make it imperative that writers and editors join together to make case reports more transparent, accurate, and consistent.7
An additional step to protect patient privacy is the requirement that authors either de-identify potentially identifiable health information, such as age, birth, death, admission, and discharge dates, or in some instances obtain separate consent for the release of that protected data.8 These restrictions constitute a challenge to case report authors who in some instances may consider these same facts critical to the integrity of the case presentation that have made some scholars doubt their continued viability. After all, the contribution of the case to the medical literature often lies in its very particularity. Conversely, no matter how frustrated we might become during writing a case report, we would not want to see our own protected health information or that of our family on a website or in print without our knowledge or approval. Indeed, the International Committee of Medical Journal Editors states that “If identifying characteristics are de-identified, authors should provide assurance, and editors should so note, that such changes do not distort scientific meaning.”9
However, the exponential growth of the internet, the spread of social media, and the ubiquity of a plethora of electronic devices, which prior generations of writers and readers could not even imagine, make these limitations necessary to protect patient privacy and the public’s trust in health care professionals. The CARE guidelines can help authors of case reports hone the art of anonymizing the protected health information of subjects of case reports, such as ethnicity and occupation, while accurately conveying the clinical specifics of the case that make it valuable to students and colleagues.
We at Federal Practitioner recognize there is a real tension between truth-telling in case report publication and respect for patient confidentiality that will never be perfectly achieved, but is one that is important for medical knowledge, making it worthy of the continuous efforts of authors and editors to negotiate.
1. Nissen T, Wynn R. The history of the case report: a selective review. JRSM Open. 2014;5(4):2054270414523410. Published 2014 Mar 12. doi:10.1177/2054270414523410
2. Gagnier JJ, Kienle G, Altman DG, et al. The CARE guidelines: consensus-based clinical case reporting guideline development. BMJ Case Rep. 2013;2013:bcr2013201554. Published 2013 Oct 23. doi:10.1136/bcr-2013-201554
3. McLean SF. Case-based learning and its application in medical and health-care fields: a review of worldwide literature. J Med Educ Curric Dev. 2016;3:JMECD.S20377. Published 2016 Apr 27. doi:10.4137/JMECD.S20377
4. Centers for Disease Control (CDC). Pneumocystis pneumonia—Los Angeles. MMWR Morb Mortal Wkly Rep. 1981;30(21):250-252.
5. McBride WG. Thalidomide and congenital abnormalities. Lancet 1961;278(7216):1358. doi:10.1016/S0140-6736(61)90927-8
6. Vandenbroucke JP. In defense of case reports and case series. Ann Intern Med. 2001;134(4):330-334. doi:10.7326/0003-4819-134-4-200102200-00017
7. Rosoff PM. Can the case report withstand ethical scrutiny? Hastings Cent Rep. 2019;49(6):17-21. doi:10.1002/hast.1065
8. Riley DS, Barber MS, Kienle GS, et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol. 2017;89:218-235. doi:10.1016/j.jclinepi.2017.04.026
9. International Committee of Medical Journal Editors. Recommendations for the conduct, reporting, editing, and publication of scholarly work in medical journals. Updated December 2021. Accessed January 31, 2022. http://www.icmje.org/news-and-editorials/new_journal_dec2021.html
Medical case reports are as old as the healing profession itself.1 These ancient medical stories have a modern definition: “A case report is a narrative that describes, for medical, scientific or educational purposes, a medical problem experienced by one or more patients.”2 Case report experts describe the 3-fold purposes of this type of research: as a mainstay of education; a harbinger of emerging illnesses; and an appraiser of new interventions. Case-based education has long been a pillar of health professions education: Nurses, doctors, and allied health professionals are taught and learn through reading and discussing with their teachers and each other about cases of their own patients and of those in the literature.3 Case reports also have helped identify and raise awareness of new diseases and rare conditions, such as HIV.4 Finally, case reports have alerted regulatory agencies and the medical community about medication adverse effects, such as birth defects from thalidomide.5
Case reports also have been criticized on both scientific and ethical grounds. Critics argue that many case reports often lack the rigor and consistency of other types of research.6 Three recent trends in medical publication have strengthened the validity of these criticisms: the increase in the popularity of case reports; the corresponding increase in submissions to journals, including Federal Practitioner; and the rise of predatory publishers.7,8
The ethical scrutiny of case reports discussed in this column focuses on the tension between providing readers with adequate, accurate information to fulfil the goals of case reports while also protecting patient confidentiality. The latter issue during most of the history of medicine was not considered by health care professionals when the prevailing paternalism supported a professional-oriented approach to health care. The rise of bioethics in the 1960s and 1970s began the shift toward patient autonomy in medical decision making and patient rights to control their protected health information that rendered case reports ethically problematic.
To address both changes in ethical standards and scientific limitations, a committee of clinicians, researchers, and journal editors formed the Case Report (CARE) group.2,8 The group undertook an effort to improve the quality of case reports. From 2011 to 2012, they developed the CARE guidelines for clinical case reporting. The guidance took the form of a Statement and Checklist presented at the 2013 International Congress on Peer Review and Biomedical Publication. Since their presentation, multiple prestigious medical journals in many countries have implemented these recommendations.
As part of an overall effort to raise the ethical caliber of our own journal, Federal Practitioner will begin to implement the CARE guidelines for case reports for all future submissions. Use of the CARE recommendations will help prospective authors enhance the scientific value and ethical caliber of case reports submitted to the journal as well as assist the Federal Practitioner editorial team, editorial board, and peer reviewers to evaluate submissions more judiciously.
An essential part of the CARE guidelines is that the patient who is the subject of the case report provide informed consent for the publication of their personal narrative. The CARE group considers this an “ethical duty” of authors and editors alike. In “exceptional circumstances” such as if the patient is a minor or permanently incapacitated, a guardian or relative may grant consent. In the rare event that even with exhaustive attempts, if informed consent cannot be obtained from a patient or their representative, then the authors of the case report must submit a statement to this effect.4 Some journals may require that the authors obtain the approval of an institutional review board or the permission of an ethics or other institutional committee or a privacy officer.2
Requesting the patient’s consent is an extension of the shared decision making that is now a best practice in clinical care into the arena of research, making the patient or their representative a partner in the work. Ethicists have recommended inviting patients or relatives to read a draft of the case report and agree to its publication or request specific modifications to the manuscript. The CARE group rightly points out that with the rise of open notes in medical documentation, patients increasingly have access to their charts in near or real time.2 Gone are the days of Sir William Osler when only doctors read medical journals and all of these technical developments as well as standards of research and social changes in the practitioner-patient relationship make it imperative that writers and editors join together to make case reports more transparent, accurate, and consistent.7
An additional step to protect patient privacy is the requirement that authors either de-identify potentially identifiable health information, such as age, birth, death, admission, and discharge dates, or in some instances obtain separate consent for the release of that protected data.8 These restrictions constitute a challenge to case report authors who in some instances may consider these same facts critical to the integrity of the case presentation that have made some scholars doubt their continued viability. After all, the contribution of the case to the medical literature often lies in its very particularity. Conversely, no matter how frustrated we might become during writing a case report, we would not want to see our own protected health information or that of our family on a website or in print without our knowledge or approval. Indeed, the International Committee of Medical Journal Editors states that “If identifying characteristics are de-identified, authors should provide assurance, and editors should so note, that such changes do not distort scientific meaning.”9
However, the exponential growth of the internet, the spread of social media, and the ubiquity of a plethora of electronic devices, which prior generations of writers and readers could not even imagine, make these limitations necessary to protect patient privacy and the public’s trust in health care professionals. The CARE guidelines can help authors of case reports hone the art of anonymizing the protected health information of subjects of case reports, such as ethnicity and occupation, while accurately conveying the clinical specifics of the case that make it valuable to students and colleagues.
We at Federal Practitioner recognize there is a real tension between truth-telling in case report publication and respect for patient confidentiality that will never be perfectly achieved, but is one that is important for medical knowledge, making it worthy of the continuous efforts of authors and editors to negotiate.
Medical case reports are as old as the healing profession itself.1 These ancient medical stories have a modern definition: “A case report is a narrative that describes, for medical, scientific or educational purposes, a medical problem experienced by one or more patients.”2 Case report experts describe the 3-fold purposes of this type of research: as a mainstay of education; a harbinger of emerging illnesses; and an appraiser of new interventions. Case-based education has long been a pillar of health professions education: Nurses, doctors, and allied health professionals are taught and learn through reading and discussing with their teachers and each other about cases of their own patients and of those in the literature.3 Case reports also have helped identify and raise awareness of new diseases and rare conditions, such as HIV.4 Finally, case reports have alerted regulatory agencies and the medical community about medication adverse effects, such as birth defects from thalidomide.5
Case reports also have been criticized on both scientific and ethical grounds. Critics argue that many case reports often lack the rigor and consistency of other types of research.6 Three recent trends in medical publication have strengthened the validity of these criticisms: the increase in the popularity of case reports; the corresponding increase in submissions to journals, including Federal Practitioner; and the rise of predatory publishers.7,8
The ethical scrutiny of case reports discussed in this column focuses on the tension between providing readers with adequate, accurate information to fulfil the goals of case reports while also protecting patient confidentiality. The latter issue during most of the history of medicine was not considered by health care professionals when the prevailing paternalism supported a professional-oriented approach to health care. The rise of bioethics in the 1960s and 1970s began the shift toward patient autonomy in medical decision making and patient rights to control their protected health information that rendered case reports ethically problematic.
To address both changes in ethical standards and scientific limitations, a committee of clinicians, researchers, and journal editors formed the Case Report (CARE) group.2,8 The group undertook an effort to improve the quality of case reports. From 2011 to 2012, they developed the CARE guidelines for clinical case reporting. The guidance took the form of a Statement and Checklist presented at the 2013 International Congress on Peer Review and Biomedical Publication. Since their presentation, multiple prestigious medical journals in many countries have implemented these recommendations.
As part of an overall effort to raise the ethical caliber of our own journal, Federal Practitioner will begin to implement the CARE guidelines for case reports for all future submissions. Use of the CARE recommendations will help prospective authors enhance the scientific value and ethical caliber of case reports submitted to the journal as well as assist the Federal Practitioner editorial team, editorial board, and peer reviewers to evaluate submissions more judiciously.
An essential part of the CARE guidelines is that the patient who is the subject of the case report provide informed consent for the publication of their personal narrative. The CARE group considers this an “ethical duty” of authors and editors alike. In “exceptional circumstances” such as if the patient is a minor or permanently incapacitated, a guardian or relative may grant consent. In the rare event that even with exhaustive attempts, if informed consent cannot be obtained from a patient or their representative, then the authors of the case report must submit a statement to this effect.4 Some journals may require that the authors obtain the approval of an institutional review board or the permission of an ethics or other institutional committee or a privacy officer.2
Requesting the patient’s consent is an extension of the shared decision making that is now a best practice in clinical care into the arena of research, making the patient or their representative a partner in the work. Ethicists have recommended inviting patients or relatives to read a draft of the case report and agree to its publication or request specific modifications to the manuscript. The CARE group rightly points out that with the rise of open notes in medical documentation, patients increasingly have access to their charts in near or real time.2 Gone are the days of Sir William Osler when only doctors read medical journals and all of these technical developments as well as standards of research and social changes in the practitioner-patient relationship make it imperative that writers and editors join together to make case reports more transparent, accurate, and consistent.7
An additional step to protect patient privacy is the requirement that authors either de-identify potentially identifiable health information, such as age, birth, death, admission, and discharge dates, or in some instances obtain separate consent for the release of that protected data.8 These restrictions constitute a challenge to case report authors who in some instances may consider these same facts critical to the integrity of the case presentation that have made some scholars doubt their continued viability. After all, the contribution of the case to the medical literature often lies in its very particularity. Conversely, no matter how frustrated we might become during writing a case report, we would not want to see our own protected health information or that of our family on a website or in print without our knowledge or approval. Indeed, the International Committee of Medical Journal Editors states that “If identifying characteristics are de-identified, authors should provide assurance, and editors should so note, that such changes do not distort scientific meaning.”9
However, the exponential growth of the internet, the spread of social media, and the ubiquity of a plethora of electronic devices, which prior generations of writers and readers could not even imagine, make these limitations necessary to protect patient privacy and the public’s trust in health care professionals. The CARE guidelines can help authors of case reports hone the art of anonymizing the protected health information of subjects of case reports, such as ethnicity and occupation, while accurately conveying the clinical specifics of the case that make it valuable to students and colleagues.
We at Federal Practitioner recognize there is a real tension between truth-telling in case report publication and respect for patient confidentiality that will never be perfectly achieved, but is one that is important for medical knowledge, making it worthy of the continuous efforts of authors and editors to negotiate.
1. Nissen T, Wynn R. The history of the case report: a selective review. JRSM Open. 2014;5(4):2054270414523410. Published 2014 Mar 12. doi:10.1177/2054270414523410
2. Gagnier JJ, Kienle G, Altman DG, et al. The CARE guidelines: consensus-based clinical case reporting guideline development. BMJ Case Rep. 2013;2013:bcr2013201554. Published 2013 Oct 23. doi:10.1136/bcr-2013-201554
3. McLean SF. Case-based learning and its application in medical and health-care fields: a review of worldwide literature. J Med Educ Curric Dev. 2016;3:JMECD.S20377. Published 2016 Apr 27. doi:10.4137/JMECD.S20377
4. Centers for Disease Control (CDC). Pneumocystis pneumonia—Los Angeles. MMWR Morb Mortal Wkly Rep. 1981;30(21):250-252.
5. McBride WG. Thalidomide and congenital abnormalities. Lancet 1961;278(7216):1358. doi:10.1016/S0140-6736(61)90927-8
6. Vandenbroucke JP. In defense of case reports and case series. Ann Intern Med. 2001;134(4):330-334. doi:10.7326/0003-4819-134-4-200102200-00017
7. Rosoff PM. Can the case report withstand ethical scrutiny? Hastings Cent Rep. 2019;49(6):17-21. doi:10.1002/hast.1065
8. Riley DS, Barber MS, Kienle GS, et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol. 2017;89:218-235. doi:10.1016/j.jclinepi.2017.04.026
9. International Committee of Medical Journal Editors. Recommendations for the conduct, reporting, editing, and publication of scholarly work in medical journals. Updated December 2021. Accessed January 31, 2022. http://www.icmje.org/news-and-editorials/new_journal_dec2021.html
1. Nissen T, Wynn R. The history of the case report: a selective review. JRSM Open. 2014;5(4):2054270414523410. Published 2014 Mar 12. doi:10.1177/2054270414523410
2. Gagnier JJ, Kienle G, Altman DG, et al. The CARE guidelines: consensus-based clinical case reporting guideline development. BMJ Case Rep. 2013;2013:bcr2013201554. Published 2013 Oct 23. doi:10.1136/bcr-2013-201554
3. McLean SF. Case-based learning and its application in medical and health-care fields: a review of worldwide literature. J Med Educ Curric Dev. 2016;3:JMECD.S20377. Published 2016 Apr 27. doi:10.4137/JMECD.S20377
4. Centers for Disease Control (CDC). Pneumocystis pneumonia—Los Angeles. MMWR Morb Mortal Wkly Rep. 1981;30(21):250-252.
5. McBride WG. Thalidomide and congenital abnormalities. Lancet 1961;278(7216):1358. doi:10.1016/S0140-6736(61)90927-8
6. Vandenbroucke JP. In defense of case reports and case series. Ann Intern Med. 2001;134(4):330-334. doi:10.7326/0003-4819-134-4-200102200-00017
7. Rosoff PM. Can the case report withstand ethical scrutiny? Hastings Cent Rep. 2019;49(6):17-21. doi:10.1002/hast.1065
8. Riley DS, Barber MS, Kienle GS, et al. CARE guidelines for case reports: explanation and elaboration document. J Clin Epidemiol. 2017;89:218-235. doi:10.1016/j.jclinepi.2017.04.026
9. International Committee of Medical Journal Editors. Recommendations for the conduct, reporting, editing, and publication of scholarly work in medical journals. Updated December 2021. Accessed January 31, 2022. http://www.icmje.org/news-and-editorials/new_journal_dec2021.html
Role and Experience of a Subintensive Care Unit in Caring for Patients With COVID-19 in Italy: The CO-RESP Study
From the Department of Emergency Medicine, Santa Croce e Carle Hospital, Cuneo, Italy (Drs. Abram, Tosello, Emanuele Bernardi, Allione, Cavalot, Dutto, Corsini, Martini, Sciolla, Sara Bernardi, and Lauria). From the School of Emergency Medicine, University of Turin, Turin, Italy (Drs. Paglietta and Giamello).
Objective: This retrospective and prospective cohort study was designed to describe the characteristics, treatments, and outcomes of patients with SARS-CoV-2 infection (COVID-19) admitted to subintensive care units (SICU) and to identify the variables associated with outcomes. SICUs have been extremely stressed during the pandemic, but most data regarding critically ill COVID-19 patients come from intensive care units (ICUs). Studies about COVID-19 patients in SICUs are lacking.
Setting and participants: The study included 88 COVID-19 patients admitted to our SICU in Cuneo, Italy, between March and May 2020.
Measurements: Clinical and ventilatory data were collected, and patients were divided by outcome. Multivariable logistic regression analysis examined the variables associated with negative outcomes (transfer to the ICU, palliation, or death in a SICU).
Results: A total of 60 patients (68%) had a positive outcome, and 28 patients (32%) had a negative outcome; 69 patients (78%) underwent continuous positive airway pressure (CPAP). Pronation (n = 37 [42%]) had been more frequently adopted in patients who had a positive outcome vs a negative outcome (n = 30 [50%] vs n = 7 [25%]; P = .048), and the median (interquartile range) Pa
Conclusion: SICUs have a fundamental role in the treatment of critically ill patients with COVID-19, who require long-term CPAP and pronation cycles. Diabetes, lymphopenia, and high D-dimer and LDH levels are associated with negative outcomes.
Keywords: emergency medicine, noninvasive ventilation, prone position, continuous positive airway pressure.
The COVID-19 pandemic has led to large increases in hospital admissions. Subintensive care units (SICUs) are among the wards most under pressure worldwide,1 dealing with the increased number of critically ill patients who need noninvasive ventilation, as well as serving as the best alternative to overfilled intensive care units (ICUs). In Italy, SICUs are playing a fundamental role in the management of COVID-19 patients, providing early treatment of respiratory failure by continuous noninvasive ventilation in order to reduce the need for intubation.2-5 Nevertheless, the great majority of available data about critically ill COVID-19 patients comes from ICUs. Full studies about outcomes of patients in SICUs are lacking and need to be conducted.
We sought to evaluate the characteristics and outcomes of patients admitted to our SICU for COVID-19 to describe the treatments they needed and their impact on prognosis, and to identify the variables associated with patient outcomes.
Methods
Study Design
This cohort study used data from patients who were admitted in the very first weeks of the pandemic. Data were collected retrospectively as well as prospectively, since the ethical committee approved our project. The quality and quantity of data in the 2 groups were comparable.
Data were collected from electronic and written medical records gathered during the patient’s entire stay in our SICU. Data were entered in a database with limited and controlled access. This study complied with the Declaration of Helsinki and was approved by the local ethics committees (ID: MEDURG10).
Study Population
Clinical Data
The past medical history and recent symptoms description were obtained by manually reviewing medical records. Epidemiological exposure was defined as contact with SARS-CoV-2–positive people or staying in an epidemic outbreak area. Initial vital parameters, venous blood tests, arterial blood gas analysis, chest x-ray, as well as the result of the nasopharyngeal swab were gathered from the emergency department (ED) examination. (Additional swabs could be requested when the first one was negative but clinical suspicion for COVID-19 was high.) Upon admission to the SICU, a standardized panel of blood tests was performed, which was repeated the next day and then every 48 hours. Arterial blood gas analysis was performed when clinically indicated, at least twice a day, or following a scheduled time in patients undergoing pronation. Charlson Comorbidity Index7 and MuLBSTA score8 were calculated based on the collected data.
Imaging
Chest ultrasonography was performed in the ED at the time of hospitalization and once a day in the SICU. Pulmonary high-resolution computed tomography (HRCT) was performed when clinically indicated or when the results of nasopharyngeal swabs and/or x-ray results were discordant with COVID-19 clinical suspicion. Contrast CT was performed when pulmonary embolism was suspected.
Medical Therapy
Hydroxychloroquine, antiviral agents, tocilizumab, and ruxolitinib were used in the early phase of the pandemic, then were dismissed after evidence of no efficacy.9-11 Steroids and low-molecular-weight heparin were used afterward. Enoxaparin was used at the standard prophylactic dosage, and 70% of the anticoagulant dosage was also adopted in patients with moderate-to-severe COVID-19 and D-dimer values >3 times the normal value.12-14 Antibiotics were given when a bacterial superinfection was suspected.
Oxygen and Ventilatory Therapy
Oxygen support or noninvasive ventilation were started based on patients’ respiratory efficacy, estimated by respiratory rate and the ratio of partial pressure of arterial oxygen and fraction of inspired oxygen (P/F ratio).15,16 Oxygen support was delivered through nasal cannula, Venturi mask, or reservoir mask. Noninvasive ventilation was performed by continuous positive airway pressure (CPAP) when the P/F ratio was <250 or the respiratory rate was >25 breaths per minute, using the helmet interface.5,17 Prone positioning during CPAP18-20 was adopted in patients meeting the acute respiratory distress syndrome (ARDS) criteria21 and having persistence of respiratory distress and P/F <300 after a 1-hour trial of CPAP.
The prone position was maintained based on patient tolerance. P/F ratio was measured before pronation (T0), after 1 hour of prone position (T1), before resupination (T2), and 6 hours after resupination (T3). With the same timing, the patient was asked to rate their comfort in each position, from 0 (lack of comfort) to 10 (optimal comfort). Delta P/F was defined as the difference between P/F at T3 and basal P/F at T0.
Outcomes
Statistical Analysis
Continuous data are reported as median and interquartile range (IQR); normal distribution of variables was tested using the Shapiro-Wilk test. Categorical variables were reported as absolute number and percentage. The Mann-Whitney test was used to compare continuous variables between groups, and chi-square test with continuity correction was used for categorical variables. The variables that were most significantly associated with a negative outcome on the univariate analysis were included in a stepwise logistic regression analysis, in order to identify independent predictors of patient outcome. Statistical analysis was performed using JASP (JASP Team) software.
Results
Study Population
Of the 88 patients included in the study, 70% were male; the median age was 66 years (IQR, 60-77). In most patients, the diagnosis of COVID-19 was derived from a positive SARS-CoV-2 nasopharyngeal swab. Six patients, however, maintained a negative swab at all determinations but had clinical and imaging features strongly suggesting COVID-19. No patients met the exclusion criteria. Most patients came from the ED (n = 58 [66%]) or general wards (n = 22 [25%]), while few were transferred from the ICU (n = 8 [9%]). The median length of stay in the SICU was 4 days (IQR, 2-7). An epidemiological link to affected persons or a known virus exposure was identifiable in 37 patients (42%).
Clinical, Laboratory, and Imaging Data
The clinical and anthropometric characteristics of patients are shown in Table 1. Hypertension and smoking habits were prevalent in our population, and the median Charlson Comorbidity Index was 3. Most patients experienced fever, dyspnea, and cough during the days before hospitalization.
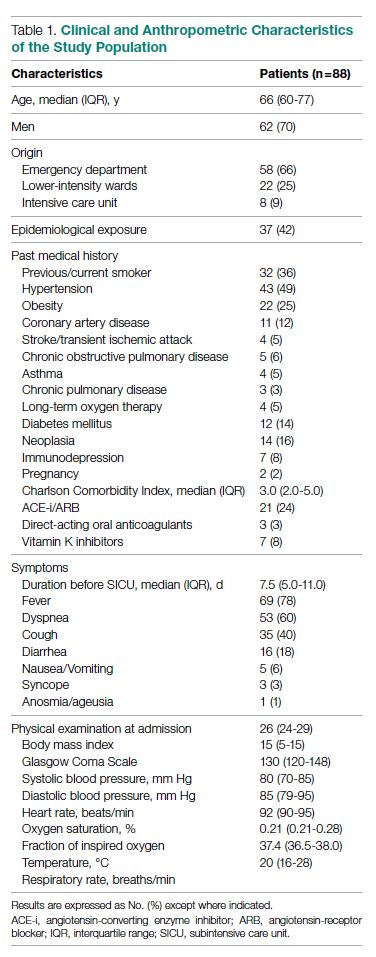
Laboratory data showed a marked inflammatory milieu in all studied patients, both at baseline and after 24 and 72 hours. Lymphopenia was observed, along with a significant increase of lactate dehydrogenase (LDH), C-reactive protein (CPR), and D-dimer, and a mild increase of procalcitonin. N-terminal pro-brain natriuretic peptide (NT-proBNP) values were also increased, with normal troponin I values (Table 2).
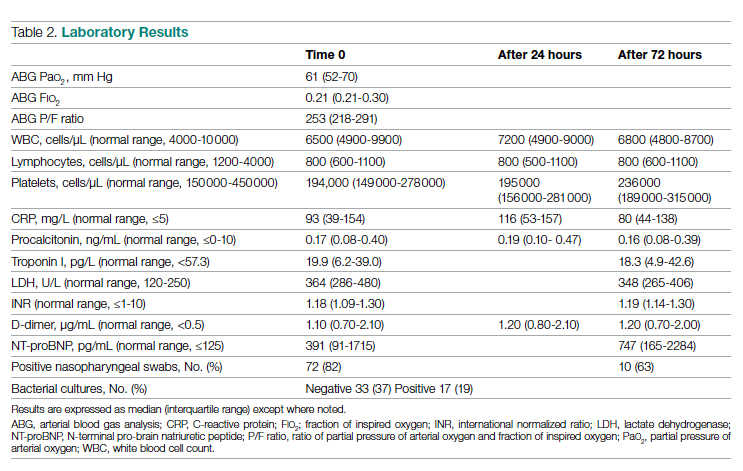
Chest x-rays were obtained in almost all patients, while HRCT was performed in nearly half of patients. Complete bedside pulmonary ultrasonography data were available for 64 patients. Heterogeneous pulmonary alterations were found, regardless of the radiological technique, and multilobe infiltrates were the prevalent radiological pattern (73%) (Table 3). Seven patients (8%) were diagnosed with associated pulmonary embolism.
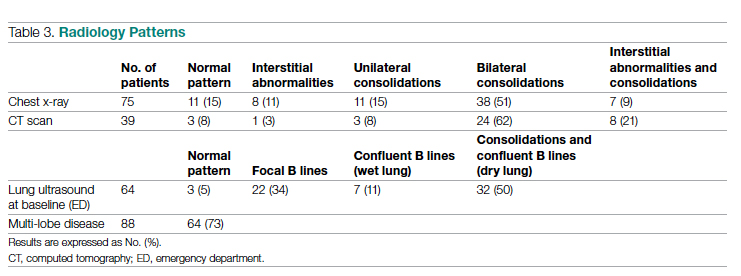
Medical Therapy
Most patients (89%) received hydroxychloroquine, whereas steroids were used in one-third of the population (36%). Immunomodulators (tocilizumab and ruxolitinib) were restricted to 12 patients (14%). Empirical antiviral therapy was introduced in the first 41 patients (47%). Enoxaparin was the default agent for thromboembolism prophylaxis, and 6 patients (7%) received 70% of the anticoagulating dose.
Oxygen and Ventilatory Therapy
Outcomes
A total of 28 patients (32%) had a negative outcome in the SICU: 8 patients (9%) died, having no clinical indication for higher-intensity care; 6 patients (7%) were transferred to general wards for palliation; and 14 patients (16%) needed an upgrade of cure intensity and were transferred to the ICU. Of these 14 patients, 9 died in the ICU. The total in-hospital mortality of COVID-19 patients, including patients transferred from the SICU to general wards in fair condition, was 27% (n = 24). Clinical, laboratory, and therapeutic characteristics between the 2 groups are shown in Table 4.
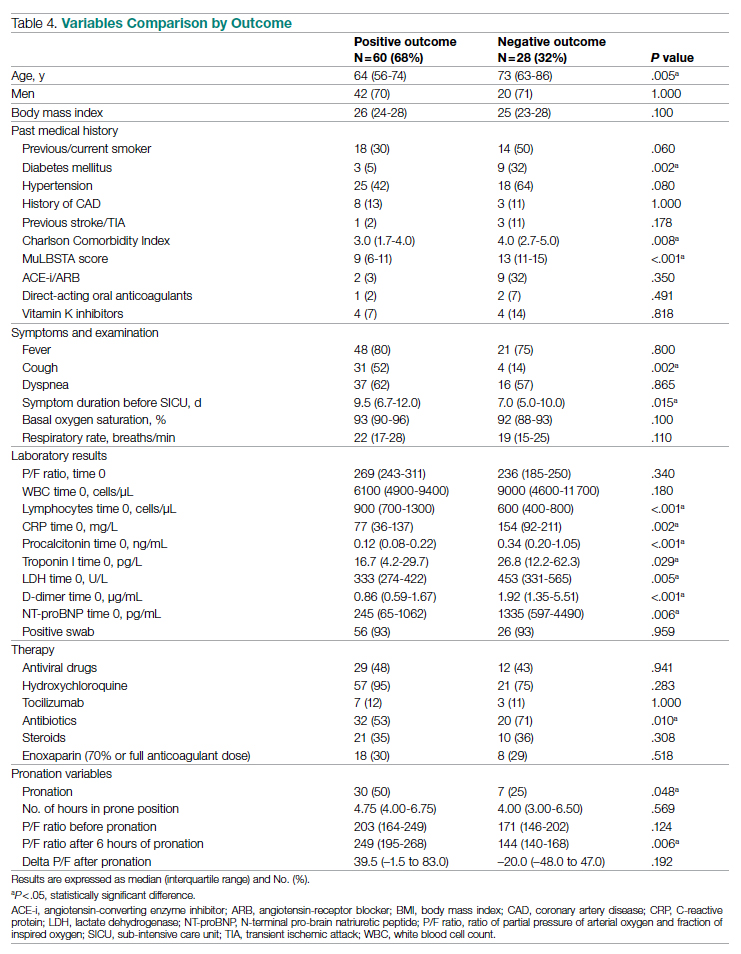
Patients who had a negative outcome were significantly older and had more comorbidities, as suggested by a significantly higher prevalence of diabetes and higher Charlson Comorbidity scores (reflecting the mortality risk based on age and comorbidities). The median MuLBSTA score, which estimates the 90-day mortality risk from viral pneumonia, was also higher in patients who had a negative outcome (9.33%). Symptom occurrence was not different in patients with a negative outcome (apart from cough, which was less frequent), but these patients underwent hospitalization earlier—since the appearance of their first COVID-19 symptoms—compared to patients who had a positive outcome. No difference was found in antihypertensive therapy with angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers among outcome groups.
More pronounced laboratory abnormalities were found in patients who had a negative outcome, compared to patients who had a positive outcome: lower lymphocytes and higher C-reactive protein (CRP), procalcitonin, D-dimer, LDH, and NT-proBNP. We found no differences in the radiological distribution of pulmonary involvement in patients who had negative or positive outcomes, nor in the adopted medical treatment.
Data showed no difference in CPAP implementation in the 2 groups. However, prone positioning had been more frequently adopted in the group of patients who had a positive outcome, compared with patients who had a negative outcome. No differences of basal P/F were found in patients who had a negative or positive outcome, but the median P/F after 6 hours of prone position was significantly lower in patients who had a negative outcome. The delta P/F ratio did not differ in the 2 groups of patients.
Discussion
Role of Subintensive Units and Mortality
The novelty of our report is its attempt to investigate the specific group of COVID-19 patients admitted to a SICU. In Italy, SICUs receive acutely ill, spontaneously breathing patients who need (invasive) hemodynamic monitoring, vasoactive medication, renal replacement therapy, chest- tube placement, thrombolysis, and respiratory noninvasive support. The nurse-to-patient ratio is higher than for general wards (usually 1 nurse to every 4 or 5 patients), though lower than for ICUs. In northern Italy, a great number of COVID-19 patients have required this kind of high-intensity care during the pandemic: Noninvasive ventilation support had to be maintained for several days, pronation maneuvers required a high number of people 2 or 3 times a day, and strict monitoring had to be assured. The SICU setting allows patients to buy time as a bridge to progressive reduction of pulmonary involvement, sometimes preventing the need for intubation.
The high prevalence of negative outcomes in the SICU underlines the complexity of COVID-19 patients in this setting. In fact, published data about mortality for patients with severe COVID-19 pneumonia are similar to ours.22,23
Clinical, Laboratory, and Imaging Data
Our analysis confirmed a high rate of comorbidities in COVID-19 patients24 and their prognostic role with age.25,26 A marked inflammatory milieu was a negative prognostic indicator, and associated concomitant bacterial superinfection could have led to a worse prognosis (procalcitonin was associated with negative outcomes).27 The cardiovascular system was nevertheless stressed, as suggested by higher values of NT-proBNP in patients with negative outcomes, which could reflect sepsis-related systemic involvement.28
It is known that the pulmonary damage caused by SARS-CoV-2 has a dynamic radiological and clinical course, with early areas of subsegmental consolidation, and bilateral ground-glass opacities predominating later in the course of the disease.29 This could explain why in our population we found no specific radiological pattern leading to a worse outcome.
Medical Therapy
No specific pharmacological therapy was found to be associated with a positive outcome in our study, just like antiviral and immunomodulator therapies failed to demonstrate effectiveness in subsequent pandemic surges. The low statistical power of our study did not allow us to give insight into the effectiveness of steroids and heparin at any dosage.
PEEP Support and Prone Positioning
Continuous positive airway pressure was initiated in the majority of patients and maintained for several days. This was an absolute novelty, because we rarely had to keep patients in helmets for long. This was feasible thanks to the SICU’s high nurse-to-patient ratio and the possibility of providing monitored sedation. Patients who could no longer tolerate CPAP helmets or did not improve with CPAP support were evaluated with anesthetists for programming further management. No initial data on respiratory rate, level of hypoxemia, or oxygen support need (level of PEEP and F
Prone positioning during CPAP was implemented in 42% of our study population: P/F ratio amelioration after prone positioning was highly variable, ranging from very good P/F ratio improvements to few responses or no response. No significantly greater delta P/F ratio was seen after the first prone positioning cycle in patients who had a positive outcome, probably due to the small size of our population, but we observed a clear positive trend. Interestingly, patients showing a negative outcome had a lower percentage of long-term responses to prone positioning: 6 hours after resupination, they lost the benefit of prone positioning in terms of P/F ratio amelioration. Similarly, a greater number of patients tolerating prone positioning had a positive outcome. These data give insight on the possible benefits of prone positioning in a noninvasively supported cohort of patients, which has been mentioned in previous studies.30,31
Outcomes and Variables Associated With Negative Outcomes
After correction for age and sex, we found in multiple regression analysis that higher D-dimer and LDH values, lymphopenia, and history of diabetes were independently associated with a worse outcome. Although our results had low statistical significance, we consider the trend of the obtained odds ratios important from a clinical point of view. These results could lead to greater attention being placed on COVID-19 patients who present with these characteristics upon their arrival to the ED because they have increased risk of death or intensive care need. Clinicians should consider SICU admission for these patients in order to guarantee closer monitoring and possibly more aggressive ventilatory treatments, earlier pronation, or earlier transfer to the ICU.
Limitations
The major limitation to our study is undoubtedly its statistical power, due to its relatively low patient population. Particularly, the small number of patients who underwent pronation did not allow speculation about the efficacy of this technique, although preliminary data seem promising. However, ours is among the first studies regarding patients with COVID-19 admitted to a SICU, and these preliminary data truthfully describe the Italian, and perhaps international, experience with the first surge of the pandemic.
Conclusions
Our data highlight the primary role of the SICU in COVID-19 in adequately treating critically ill patients who have high care needs different from intubation, and who require noninvasive ventilation for prolonged times as well as frequent pronation cycles. This setting of care may represent a valid, reliable, and effective option for critically ill respiratory patients. History of diabetes, lymphopenia, and high D-dimer and LDH values are independently associated with negative outcomes, and patients presenting with these characteristics should be strictly monitored.
Acknowledgments: The authors thank the Informatica System S.R.L., as well as Allessando Mendolia for the pro bono creation of the ISCovidCollect data collecting app.
Corresponding author: Sara Abram, MD, via Coppino, 12100 Cuneo, Italy; [email protected].
Disclosures: None.
1. Plate JDJ, Leenen LPH, Houwert M, Hietbrink F. Utilisation of intermediate care units: a systematic review. Crit Care Res Pract. 2017;2017:8038460. doi:10.1155/2017/8038460
2. Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med. 2007;35(1):18-25. doi:10.1097/01.CCM.0000251821.44259.F3
3. Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315(22):2435-2441. doi:10.1001/jama.2016.6338
4. Mas A, Masip J. Noninvasive ventilation in acute respiratory failure. Int J Chron Obstruct Pulmon Dis. 2014;9:837-852. doi:10.2147/COPD.S42664
5. Bellani G, Patroniti N, Greco M, Foti G, Pesenti A. The use of helmets to deliver non-invasive continuous positive airway pressure in hypoxemic acute respiratory failure. Minerva Anestesiol. 2008;74(11):651-656.
6. Lomoro P, Verde F, Zerboni F, et al. COVID-19 pneumonia manifestations at the admission on chest ultrasound, radiographs, and CT: single-center study and comprehensive radiologic literature review. Eur J Radiol Open. 2020;7:100231. doi:10.1016/j.ejro.2020.100231
7. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373-383. doi:10.1016/0021-9681(87)90171-8
8. Guo L, Wei D, Zhang X, et al. Clinical features predicting mortality risk in patients with viral pneumonia: the MuLBSTA score. Front Microbiol. 2019;10:2752. doi:10.3389/fmicb.2019.02752
9. Lombardy Section Italian Society Infectious and Tropical Disease. Vademecum for the treatment of people with COVID-19. Edition 2.0, 13 March 2020. Infez Med. 2020;28(2):143-152.
10. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30(3):269-271. doi:10.1038/s41422-020-0282-0
11. Cao B, Wang Y, Wen D, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382(19):1787-1799. doi:10.1056/NEJMoa2001282
12. Stone JH, Frigault MJ, Serling-Boyd NJ, et al; BACC Bay Tocilizumab Trial Investigators. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020;383(24):2333-2344. doi:10.1056/NEJMoa2028836
13. Shastri MD, Stewart N, Horne J, et al. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10(5):e0126763. doi:10.1371/journal.pone.0126763
14. Milewska A, Zarebski M, Nowak P, Stozek K, Potempa J, Pyrc K. Human coronavirus NL63 utilizes heparin sulfate proteoglycans for attachment to target cells. J Virol. 2014;88(22):13221-13230. doi:10.1128/JVI.02078-14
15. Marietta M, Vandelli P, Mighali P, Vicini R, Coluccio V, D’Amico R; COVID-19 HD Study Group. Randomised controlled trial comparing efficacy and safety of high versus low low-molecular weight heparin dosages in hospitalized patients with severe COVID-19 pneumonia and coagulopathy not requiring invasive mechanical ventilation (COVID-19 HD): a structured summary of a study protocol. Trials. 2020;21(1):574. doi:10.1186/s13063-020-04475-z
16. Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med. 1995;23(10):1638-1652. doi:10.1097/00003246-199510000-00007
17. Sinha P, Calfee CS. Phenotypes in acute respiratory distress syndrome: moving towards precision medicine. Curr Opin Crit Care. 2019;25(1):12-20. doi:10.1097/MCC.0000000000000571
18. Lucchini A, Giani M, Isgrò S, Rona R, Foti G. The “helmet bundle” in COVID-19 patients undergoing non-invasive ventilation. Intensive Crit Care Nurs. 2020;58:102859. doi:10.1016/j.iccn.2020.102859
19. Ding L, Wang L, Ma W, He H. Efficacy and safety of early prone positioning combined with HFNC or NIV in moderate to severe ARDS: a multi-center prospective cohort study. Crit Care. 2020;24(1):28. doi:10.1186/s13054-020-2738-5
20. Scaravilli V, Grasselli G, Castagna L, et al. Prone positioning improves oxygenation in spontaneously breathing nonintubated patients with hypoxemic acute respiratory failure: a retrospective study. J Crit Care. 2015;30(6):1390-1394. doi:10.1016/j.jcrc.2015.07.008
21. Caputo ND, Strayer RJ, Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med. 2020;27(5):375-378. doi:10.1111/acem.13994
22. ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307(23):2526-2533. doi:10.1001/jama.2012.5669
23. Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. doi:10.1136/bmj.m1966
24. Docherty AB, Harrison EM, Green CA, et al; ISARIC4C investigators. Features of 20 133 UK patients in hospital with Covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi:10.1136/bmj.m1985
25. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775
26. Muniyappa R, Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab. 2020;318(5):E736-E741. doi:10.1152/ajpendo.00124.2020
27. Guo W, Li M, Dong Y, et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020:e3319. doi:10.1002/dmrr.3319
28. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. doi:10.1016/S0140-6736(20)30211-7
29. Kooraki S, Hosseiny M, Myers L, Gholamrezanezhad A. Coronavirus (COVID-19) outbreak: what the Department of Radiology should know. J Am Coll Radiol. 2020;17(4):447-451. doi:10.1016/j.jacr.2020.02.008
30. Coppo A, Bellani G, Winterton D, et al. Feasibility and physiological effects of prone positioning in non-intubated patients with acute respiratory failure due to COVID-19 (PRON-COVID): a prospective cohort study. Lancet Respir Med. 2020;8(8):765-774. doi:10.1016/S2213-2600(20)30268-X
31. Weatherald J, Solverson K, Zuege DJ, Loroff N, Fiest KM, Parhar KKS. Awake prone positioning for COVID-19 hypoxemic respiratory failure: a rapid review. J Crit Care. 2021;61:63-70. doi:10.1016/j.jcrc.2020.08.018
From the Department of Emergency Medicine, Santa Croce e Carle Hospital, Cuneo, Italy (Drs. Abram, Tosello, Emanuele Bernardi, Allione, Cavalot, Dutto, Corsini, Martini, Sciolla, Sara Bernardi, and Lauria). From the School of Emergency Medicine, University of Turin, Turin, Italy (Drs. Paglietta and Giamello).
Objective: This retrospective and prospective cohort study was designed to describe the characteristics, treatments, and outcomes of patients with SARS-CoV-2 infection (COVID-19) admitted to subintensive care units (SICU) and to identify the variables associated with outcomes. SICUs have been extremely stressed during the pandemic, but most data regarding critically ill COVID-19 patients come from intensive care units (ICUs). Studies about COVID-19 patients in SICUs are lacking.
Setting and participants: The study included 88 COVID-19 patients admitted to our SICU in Cuneo, Italy, between March and May 2020.
Measurements: Clinical and ventilatory data were collected, and patients were divided by outcome. Multivariable logistic regression analysis examined the variables associated with negative outcomes (transfer to the ICU, palliation, or death in a SICU).
Results: A total of 60 patients (68%) had a positive outcome, and 28 patients (32%) had a negative outcome; 69 patients (78%) underwent continuous positive airway pressure (CPAP). Pronation (n = 37 [42%]) had been more frequently adopted in patients who had a positive outcome vs a negative outcome (n = 30 [50%] vs n = 7 [25%]; P = .048), and the median (interquartile range) Pa
Conclusion: SICUs have a fundamental role in the treatment of critically ill patients with COVID-19, who require long-term CPAP and pronation cycles. Diabetes, lymphopenia, and high D-dimer and LDH levels are associated with negative outcomes.
Keywords: emergency medicine, noninvasive ventilation, prone position, continuous positive airway pressure.
The COVID-19 pandemic has led to large increases in hospital admissions. Subintensive care units (SICUs) are among the wards most under pressure worldwide,1 dealing with the increased number of critically ill patients who need noninvasive ventilation, as well as serving as the best alternative to overfilled intensive care units (ICUs). In Italy, SICUs are playing a fundamental role in the management of COVID-19 patients, providing early treatment of respiratory failure by continuous noninvasive ventilation in order to reduce the need for intubation.2-5 Nevertheless, the great majority of available data about critically ill COVID-19 patients comes from ICUs. Full studies about outcomes of patients in SICUs are lacking and need to be conducted.
We sought to evaluate the characteristics and outcomes of patients admitted to our SICU for COVID-19 to describe the treatments they needed and their impact on prognosis, and to identify the variables associated with patient outcomes.
Methods
Study Design
This cohort study used data from patients who were admitted in the very first weeks of the pandemic. Data were collected retrospectively as well as prospectively, since the ethical committee approved our project. The quality and quantity of data in the 2 groups were comparable.
Data were collected from electronic and written medical records gathered during the patient’s entire stay in our SICU. Data were entered in a database with limited and controlled access. This study complied with the Declaration of Helsinki and was approved by the local ethics committees (ID: MEDURG10).
Study Population
Clinical Data
The past medical history and recent symptoms description were obtained by manually reviewing medical records. Epidemiological exposure was defined as contact with SARS-CoV-2–positive people or staying in an epidemic outbreak area. Initial vital parameters, venous blood tests, arterial blood gas analysis, chest x-ray, as well as the result of the nasopharyngeal swab were gathered from the emergency department (ED) examination. (Additional swabs could be requested when the first one was negative but clinical suspicion for COVID-19 was high.) Upon admission to the SICU, a standardized panel of blood tests was performed, which was repeated the next day and then every 48 hours. Arterial blood gas analysis was performed when clinically indicated, at least twice a day, or following a scheduled time in patients undergoing pronation. Charlson Comorbidity Index7 and MuLBSTA score8 were calculated based on the collected data.
Imaging
Chest ultrasonography was performed in the ED at the time of hospitalization and once a day in the SICU. Pulmonary high-resolution computed tomography (HRCT) was performed when clinically indicated or when the results of nasopharyngeal swabs and/or x-ray results were discordant with COVID-19 clinical suspicion. Contrast CT was performed when pulmonary embolism was suspected.
Medical Therapy
Hydroxychloroquine, antiviral agents, tocilizumab, and ruxolitinib were used in the early phase of the pandemic, then were dismissed after evidence of no efficacy.9-11 Steroids and low-molecular-weight heparin were used afterward. Enoxaparin was used at the standard prophylactic dosage, and 70% of the anticoagulant dosage was also adopted in patients with moderate-to-severe COVID-19 and D-dimer values >3 times the normal value.12-14 Antibiotics were given when a bacterial superinfection was suspected.
Oxygen and Ventilatory Therapy
Oxygen support or noninvasive ventilation were started based on patients’ respiratory efficacy, estimated by respiratory rate and the ratio of partial pressure of arterial oxygen and fraction of inspired oxygen (P/F ratio).15,16 Oxygen support was delivered through nasal cannula, Venturi mask, or reservoir mask. Noninvasive ventilation was performed by continuous positive airway pressure (CPAP) when the P/F ratio was <250 or the respiratory rate was >25 breaths per minute, using the helmet interface.5,17 Prone positioning during CPAP18-20 was adopted in patients meeting the acute respiratory distress syndrome (ARDS) criteria21 and having persistence of respiratory distress and P/F <300 after a 1-hour trial of CPAP.
The prone position was maintained based on patient tolerance. P/F ratio was measured before pronation (T0), after 1 hour of prone position (T1), before resupination (T2), and 6 hours after resupination (T3). With the same timing, the patient was asked to rate their comfort in each position, from 0 (lack of comfort) to 10 (optimal comfort). Delta P/F was defined as the difference between P/F at T3 and basal P/F at T0.
Outcomes
Statistical Analysis
Continuous data are reported as median and interquartile range (IQR); normal distribution of variables was tested using the Shapiro-Wilk test. Categorical variables were reported as absolute number and percentage. The Mann-Whitney test was used to compare continuous variables between groups, and chi-square test with continuity correction was used for categorical variables. The variables that were most significantly associated with a negative outcome on the univariate analysis were included in a stepwise logistic regression analysis, in order to identify independent predictors of patient outcome. Statistical analysis was performed using JASP (JASP Team) software.
Results
Study Population
Of the 88 patients included in the study, 70% were male; the median age was 66 years (IQR, 60-77). In most patients, the diagnosis of COVID-19 was derived from a positive SARS-CoV-2 nasopharyngeal swab. Six patients, however, maintained a negative swab at all determinations but had clinical and imaging features strongly suggesting COVID-19. No patients met the exclusion criteria. Most patients came from the ED (n = 58 [66%]) or general wards (n = 22 [25%]), while few were transferred from the ICU (n = 8 [9%]). The median length of stay in the SICU was 4 days (IQR, 2-7). An epidemiological link to affected persons or a known virus exposure was identifiable in 37 patients (42%).
Clinical, Laboratory, and Imaging Data
The clinical and anthropometric characteristics of patients are shown in Table 1. Hypertension and smoking habits were prevalent in our population, and the median Charlson Comorbidity Index was 3. Most patients experienced fever, dyspnea, and cough during the days before hospitalization.

Laboratory data showed a marked inflammatory milieu in all studied patients, both at baseline and after 24 and 72 hours. Lymphopenia was observed, along with a significant increase of lactate dehydrogenase (LDH), C-reactive protein (CPR), and D-dimer, and a mild increase of procalcitonin. N-terminal pro-brain natriuretic peptide (NT-proBNP) values were also increased, with normal troponin I values (Table 2).

Chest x-rays were obtained in almost all patients, while HRCT was performed in nearly half of patients. Complete bedside pulmonary ultrasonography data were available for 64 patients. Heterogeneous pulmonary alterations were found, regardless of the radiological technique, and multilobe infiltrates were the prevalent radiological pattern (73%) (Table 3). Seven patients (8%) were diagnosed with associated pulmonary embolism.

Medical Therapy
Most patients (89%) received hydroxychloroquine, whereas steroids were used in one-third of the population (36%). Immunomodulators (tocilizumab and ruxolitinib) were restricted to 12 patients (14%). Empirical antiviral therapy was introduced in the first 41 patients (47%). Enoxaparin was the default agent for thromboembolism prophylaxis, and 6 patients (7%) received 70% of the anticoagulating dose.
Oxygen and Ventilatory Therapy
Outcomes
A total of 28 patients (32%) had a negative outcome in the SICU: 8 patients (9%) died, having no clinical indication for higher-intensity care; 6 patients (7%) were transferred to general wards for palliation; and 14 patients (16%) needed an upgrade of cure intensity and were transferred to the ICU. Of these 14 patients, 9 died in the ICU. The total in-hospital mortality of COVID-19 patients, including patients transferred from the SICU to general wards in fair condition, was 27% (n = 24). Clinical, laboratory, and therapeutic characteristics between the 2 groups are shown in Table 4.

Patients who had a negative outcome were significantly older and had more comorbidities, as suggested by a significantly higher prevalence of diabetes and higher Charlson Comorbidity scores (reflecting the mortality risk based on age and comorbidities). The median MuLBSTA score, which estimates the 90-day mortality risk from viral pneumonia, was also higher in patients who had a negative outcome (9.33%). Symptom occurrence was not different in patients with a negative outcome (apart from cough, which was less frequent), but these patients underwent hospitalization earlier—since the appearance of their first COVID-19 symptoms—compared to patients who had a positive outcome. No difference was found in antihypertensive therapy with angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers among outcome groups.
More pronounced laboratory abnormalities were found in patients who had a negative outcome, compared to patients who had a positive outcome: lower lymphocytes and higher C-reactive protein (CRP), procalcitonin, D-dimer, LDH, and NT-proBNP. We found no differences in the radiological distribution of pulmonary involvement in patients who had negative or positive outcomes, nor in the adopted medical treatment.
Data showed no difference in CPAP implementation in the 2 groups. However, prone positioning had been more frequently adopted in the group of patients who had a positive outcome, compared with patients who had a negative outcome. No differences of basal P/F were found in patients who had a negative or positive outcome, but the median P/F after 6 hours of prone position was significantly lower in patients who had a negative outcome. The delta P/F ratio did not differ in the 2 groups of patients.
Discussion
Role of Subintensive Units and Mortality
The novelty of our report is its attempt to investigate the specific group of COVID-19 patients admitted to a SICU. In Italy, SICUs receive acutely ill, spontaneously breathing patients who need (invasive) hemodynamic monitoring, vasoactive medication, renal replacement therapy, chest- tube placement, thrombolysis, and respiratory noninvasive support. The nurse-to-patient ratio is higher than for general wards (usually 1 nurse to every 4 or 5 patients), though lower than for ICUs. In northern Italy, a great number of COVID-19 patients have required this kind of high-intensity care during the pandemic: Noninvasive ventilation support had to be maintained for several days, pronation maneuvers required a high number of people 2 or 3 times a day, and strict monitoring had to be assured. The SICU setting allows patients to buy time as a bridge to progressive reduction of pulmonary involvement, sometimes preventing the need for intubation.
The high prevalence of negative outcomes in the SICU underlines the complexity of COVID-19 patients in this setting. In fact, published data about mortality for patients with severe COVID-19 pneumonia are similar to ours.22,23
Clinical, Laboratory, and Imaging Data
Our analysis confirmed a high rate of comorbidities in COVID-19 patients24 and their prognostic role with age.25,26 A marked inflammatory milieu was a negative prognostic indicator, and associated concomitant bacterial superinfection could have led to a worse prognosis (procalcitonin was associated with negative outcomes).27 The cardiovascular system was nevertheless stressed, as suggested by higher values of NT-proBNP in patients with negative outcomes, which could reflect sepsis-related systemic involvement.28
It is known that the pulmonary damage caused by SARS-CoV-2 has a dynamic radiological and clinical course, with early areas of subsegmental consolidation, and bilateral ground-glass opacities predominating later in the course of the disease.29 This could explain why in our population we found no specific radiological pattern leading to a worse outcome.
Medical Therapy
No specific pharmacological therapy was found to be associated with a positive outcome in our study, just like antiviral and immunomodulator therapies failed to demonstrate effectiveness in subsequent pandemic surges. The low statistical power of our study did not allow us to give insight into the effectiveness of steroids and heparin at any dosage.
PEEP Support and Prone Positioning
Continuous positive airway pressure was initiated in the majority of patients and maintained for several days. This was an absolute novelty, because we rarely had to keep patients in helmets for long. This was feasible thanks to the SICU’s high nurse-to-patient ratio and the possibility of providing monitored sedation. Patients who could no longer tolerate CPAP helmets or did not improve with CPAP support were evaluated with anesthetists for programming further management. No initial data on respiratory rate, level of hypoxemia, or oxygen support need (level of PEEP and F
Prone positioning during CPAP was implemented in 42% of our study population: P/F ratio amelioration after prone positioning was highly variable, ranging from very good P/F ratio improvements to few responses or no response. No significantly greater delta P/F ratio was seen after the first prone positioning cycle in patients who had a positive outcome, probably due to the small size of our population, but we observed a clear positive trend. Interestingly, patients showing a negative outcome had a lower percentage of long-term responses to prone positioning: 6 hours after resupination, they lost the benefit of prone positioning in terms of P/F ratio amelioration. Similarly, a greater number of patients tolerating prone positioning had a positive outcome. These data give insight on the possible benefits of prone positioning in a noninvasively supported cohort of patients, which has been mentioned in previous studies.30,31
Outcomes and Variables Associated With Negative Outcomes
After correction for age and sex, we found in multiple regression analysis that higher D-dimer and LDH values, lymphopenia, and history of diabetes were independently associated with a worse outcome. Although our results had low statistical significance, we consider the trend of the obtained odds ratios important from a clinical point of view. These results could lead to greater attention being placed on COVID-19 patients who present with these characteristics upon their arrival to the ED because they have increased risk of death or intensive care need. Clinicians should consider SICU admission for these patients in order to guarantee closer monitoring and possibly more aggressive ventilatory treatments, earlier pronation, or earlier transfer to the ICU.
Limitations
The major limitation to our study is undoubtedly its statistical power, due to its relatively low patient population. Particularly, the small number of patients who underwent pronation did not allow speculation about the efficacy of this technique, although preliminary data seem promising. However, ours is among the first studies regarding patients with COVID-19 admitted to a SICU, and these preliminary data truthfully describe the Italian, and perhaps international, experience with the first surge of the pandemic.
Conclusions
Our data highlight the primary role of the SICU in COVID-19 in adequately treating critically ill patients who have high care needs different from intubation, and who require noninvasive ventilation for prolonged times as well as frequent pronation cycles. This setting of care may represent a valid, reliable, and effective option for critically ill respiratory patients. History of diabetes, lymphopenia, and high D-dimer and LDH values are independently associated with negative outcomes, and patients presenting with these characteristics should be strictly monitored.
Acknowledgments: The authors thank the Informatica System S.R.L., as well as Allessando Mendolia for the pro bono creation of the ISCovidCollect data collecting app.
Corresponding author: Sara Abram, MD, via Coppino, 12100 Cuneo, Italy; [email protected].
Disclosures: None.
From the Department of Emergency Medicine, Santa Croce e Carle Hospital, Cuneo, Italy (Drs. Abram, Tosello, Emanuele Bernardi, Allione, Cavalot, Dutto, Corsini, Martini, Sciolla, Sara Bernardi, and Lauria). From the School of Emergency Medicine, University of Turin, Turin, Italy (Drs. Paglietta and Giamello).
Objective: This retrospective and prospective cohort study was designed to describe the characteristics, treatments, and outcomes of patients with SARS-CoV-2 infection (COVID-19) admitted to subintensive care units (SICU) and to identify the variables associated with outcomes. SICUs have been extremely stressed during the pandemic, but most data regarding critically ill COVID-19 patients come from intensive care units (ICUs). Studies about COVID-19 patients in SICUs are lacking.
Setting and participants: The study included 88 COVID-19 patients admitted to our SICU in Cuneo, Italy, between March and May 2020.
Measurements: Clinical and ventilatory data were collected, and patients were divided by outcome. Multivariable logistic regression analysis examined the variables associated with negative outcomes (transfer to the ICU, palliation, or death in a SICU).
Results: A total of 60 patients (68%) had a positive outcome, and 28 patients (32%) had a negative outcome; 69 patients (78%) underwent continuous positive airway pressure (CPAP). Pronation (n = 37 [42%]) had been more frequently adopted in patients who had a positive outcome vs a negative outcome (n = 30 [50%] vs n = 7 [25%]; P = .048), and the median (interquartile range) Pa
Conclusion: SICUs have a fundamental role in the treatment of critically ill patients with COVID-19, who require long-term CPAP and pronation cycles. Diabetes, lymphopenia, and high D-dimer and LDH levels are associated with negative outcomes.
Keywords: emergency medicine, noninvasive ventilation, prone position, continuous positive airway pressure.
The COVID-19 pandemic has led to large increases in hospital admissions. Subintensive care units (SICUs) are among the wards most under pressure worldwide,1 dealing with the increased number of critically ill patients who need noninvasive ventilation, as well as serving as the best alternative to overfilled intensive care units (ICUs). In Italy, SICUs are playing a fundamental role in the management of COVID-19 patients, providing early treatment of respiratory failure by continuous noninvasive ventilation in order to reduce the need for intubation.2-5 Nevertheless, the great majority of available data about critically ill COVID-19 patients comes from ICUs. Full studies about outcomes of patients in SICUs are lacking and need to be conducted.
We sought to evaluate the characteristics and outcomes of patients admitted to our SICU for COVID-19 to describe the treatments they needed and their impact on prognosis, and to identify the variables associated with patient outcomes.
Methods
Study Design
This cohort study used data from patients who were admitted in the very first weeks of the pandemic. Data were collected retrospectively as well as prospectively, since the ethical committee approved our project. The quality and quantity of data in the 2 groups were comparable.
Data were collected from electronic and written medical records gathered during the patient’s entire stay in our SICU. Data were entered in a database with limited and controlled access. This study complied with the Declaration of Helsinki and was approved by the local ethics committees (ID: MEDURG10).
Study Population
Clinical Data
The past medical history and recent symptoms description were obtained by manually reviewing medical records. Epidemiological exposure was defined as contact with SARS-CoV-2–positive people or staying in an epidemic outbreak area. Initial vital parameters, venous blood tests, arterial blood gas analysis, chest x-ray, as well as the result of the nasopharyngeal swab were gathered from the emergency department (ED) examination. (Additional swabs could be requested when the first one was negative but clinical suspicion for COVID-19 was high.) Upon admission to the SICU, a standardized panel of blood tests was performed, which was repeated the next day and then every 48 hours. Arterial blood gas analysis was performed when clinically indicated, at least twice a day, or following a scheduled time in patients undergoing pronation. Charlson Comorbidity Index7 and MuLBSTA score8 were calculated based on the collected data.
Imaging
Chest ultrasonography was performed in the ED at the time of hospitalization and once a day in the SICU. Pulmonary high-resolution computed tomography (HRCT) was performed when clinically indicated or when the results of nasopharyngeal swabs and/or x-ray results were discordant with COVID-19 clinical suspicion. Contrast CT was performed when pulmonary embolism was suspected.
Medical Therapy
Hydroxychloroquine, antiviral agents, tocilizumab, and ruxolitinib were used in the early phase of the pandemic, then were dismissed after evidence of no efficacy.9-11 Steroids and low-molecular-weight heparin were used afterward. Enoxaparin was used at the standard prophylactic dosage, and 70% of the anticoagulant dosage was also adopted in patients with moderate-to-severe COVID-19 and D-dimer values >3 times the normal value.12-14 Antibiotics were given when a bacterial superinfection was suspected.
Oxygen and Ventilatory Therapy
Oxygen support or noninvasive ventilation were started based on patients’ respiratory efficacy, estimated by respiratory rate and the ratio of partial pressure of arterial oxygen and fraction of inspired oxygen (P/F ratio).15,16 Oxygen support was delivered through nasal cannula, Venturi mask, or reservoir mask. Noninvasive ventilation was performed by continuous positive airway pressure (CPAP) when the P/F ratio was <250 or the respiratory rate was >25 breaths per minute, using the helmet interface.5,17 Prone positioning during CPAP18-20 was adopted in patients meeting the acute respiratory distress syndrome (ARDS) criteria21 and having persistence of respiratory distress and P/F <300 after a 1-hour trial of CPAP.
The prone position was maintained based on patient tolerance. P/F ratio was measured before pronation (T0), after 1 hour of prone position (T1), before resupination (T2), and 6 hours after resupination (T3). With the same timing, the patient was asked to rate their comfort in each position, from 0 (lack of comfort) to 10 (optimal comfort). Delta P/F was defined as the difference between P/F at T3 and basal P/F at T0.
Outcomes
Statistical Analysis
Continuous data are reported as median and interquartile range (IQR); normal distribution of variables was tested using the Shapiro-Wilk test. Categorical variables were reported as absolute number and percentage. The Mann-Whitney test was used to compare continuous variables between groups, and chi-square test with continuity correction was used for categorical variables. The variables that were most significantly associated with a negative outcome on the univariate analysis were included in a stepwise logistic regression analysis, in order to identify independent predictors of patient outcome. Statistical analysis was performed using JASP (JASP Team) software.
Results
Study Population
Of the 88 patients included in the study, 70% were male; the median age was 66 years (IQR, 60-77). In most patients, the diagnosis of COVID-19 was derived from a positive SARS-CoV-2 nasopharyngeal swab. Six patients, however, maintained a negative swab at all determinations but had clinical and imaging features strongly suggesting COVID-19. No patients met the exclusion criteria. Most patients came from the ED (n = 58 [66%]) or general wards (n = 22 [25%]), while few were transferred from the ICU (n = 8 [9%]). The median length of stay in the SICU was 4 days (IQR, 2-7). An epidemiological link to affected persons or a known virus exposure was identifiable in 37 patients (42%).
Clinical, Laboratory, and Imaging Data
The clinical and anthropometric characteristics of patients are shown in Table 1. Hypertension and smoking habits were prevalent in our population, and the median Charlson Comorbidity Index was 3. Most patients experienced fever, dyspnea, and cough during the days before hospitalization.

Laboratory data showed a marked inflammatory milieu in all studied patients, both at baseline and after 24 and 72 hours. Lymphopenia was observed, along with a significant increase of lactate dehydrogenase (LDH), C-reactive protein (CPR), and D-dimer, and a mild increase of procalcitonin. N-terminal pro-brain natriuretic peptide (NT-proBNP) values were also increased, with normal troponin I values (Table 2).

Chest x-rays were obtained in almost all patients, while HRCT was performed in nearly half of patients. Complete bedside pulmonary ultrasonography data were available for 64 patients. Heterogeneous pulmonary alterations were found, regardless of the radiological technique, and multilobe infiltrates were the prevalent radiological pattern (73%) (Table 3). Seven patients (8%) were diagnosed with associated pulmonary embolism.

Medical Therapy
Most patients (89%) received hydroxychloroquine, whereas steroids were used in one-third of the population (36%). Immunomodulators (tocilizumab and ruxolitinib) were restricted to 12 patients (14%). Empirical antiviral therapy was introduced in the first 41 patients (47%). Enoxaparin was the default agent for thromboembolism prophylaxis, and 6 patients (7%) received 70% of the anticoagulating dose.
Oxygen and Ventilatory Therapy
Outcomes
A total of 28 patients (32%) had a negative outcome in the SICU: 8 patients (9%) died, having no clinical indication for higher-intensity care; 6 patients (7%) were transferred to general wards for palliation; and 14 patients (16%) needed an upgrade of cure intensity and were transferred to the ICU. Of these 14 patients, 9 died in the ICU. The total in-hospital mortality of COVID-19 patients, including patients transferred from the SICU to general wards in fair condition, was 27% (n = 24). Clinical, laboratory, and therapeutic characteristics between the 2 groups are shown in Table 4.

Patients who had a negative outcome were significantly older and had more comorbidities, as suggested by a significantly higher prevalence of diabetes and higher Charlson Comorbidity scores (reflecting the mortality risk based on age and comorbidities). The median MuLBSTA score, which estimates the 90-day mortality risk from viral pneumonia, was also higher in patients who had a negative outcome (9.33%). Symptom occurrence was not different in patients with a negative outcome (apart from cough, which was less frequent), but these patients underwent hospitalization earlier—since the appearance of their first COVID-19 symptoms—compared to patients who had a positive outcome. No difference was found in antihypertensive therapy with angiotensin-converting enzyme inhibitors or angiotensin-receptor blockers among outcome groups.
More pronounced laboratory abnormalities were found in patients who had a negative outcome, compared to patients who had a positive outcome: lower lymphocytes and higher C-reactive protein (CRP), procalcitonin, D-dimer, LDH, and NT-proBNP. We found no differences in the radiological distribution of pulmonary involvement in patients who had negative or positive outcomes, nor in the adopted medical treatment.
Data showed no difference in CPAP implementation in the 2 groups. However, prone positioning had been more frequently adopted in the group of patients who had a positive outcome, compared with patients who had a negative outcome. No differences of basal P/F were found in patients who had a negative or positive outcome, but the median P/F after 6 hours of prone position was significantly lower in patients who had a negative outcome. The delta P/F ratio did not differ in the 2 groups of patients.
Discussion
Role of Subintensive Units and Mortality
The novelty of our report is its attempt to investigate the specific group of COVID-19 patients admitted to a SICU. In Italy, SICUs receive acutely ill, spontaneously breathing patients who need (invasive) hemodynamic monitoring, vasoactive medication, renal replacement therapy, chest- tube placement, thrombolysis, and respiratory noninvasive support. The nurse-to-patient ratio is higher than for general wards (usually 1 nurse to every 4 or 5 patients), though lower than for ICUs. In northern Italy, a great number of COVID-19 patients have required this kind of high-intensity care during the pandemic: Noninvasive ventilation support had to be maintained for several days, pronation maneuvers required a high number of people 2 or 3 times a day, and strict monitoring had to be assured. The SICU setting allows patients to buy time as a bridge to progressive reduction of pulmonary involvement, sometimes preventing the need for intubation.
The high prevalence of negative outcomes in the SICU underlines the complexity of COVID-19 patients in this setting. In fact, published data about mortality for patients with severe COVID-19 pneumonia are similar to ours.22,23
Clinical, Laboratory, and Imaging Data
Our analysis confirmed a high rate of comorbidities in COVID-19 patients24 and their prognostic role with age.25,26 A marked inflammatory milieu was a negative prognostic indicator, and associated concomitant bacterial superinfection could have led to a worse prognosis (procalcitonin was associated with negative outcomes).27 The cardiovascular system was nevertheless stressed, as suggested by higher values of NT-proBNP in patients with negative outcomes, which could reflect sepsis-related systemic involvement.28
It is known that the pulmonary damage caused by SARS-CoV-2 has a dynamic radiological and clinical course, with early areas of subsegmental consolidation, and bilateral ground-glass opacities predominating later in the course of the disease.29 This could explain why in our population we found no specific radiological pattern leading to a worse outcome.
Medical Therapy
No specific pharmacological therapy was found to be associated with a positive outcome in our study, just like antiviral and immunomodulator therapies failed to demonstrate effectiveness in subsequent pandemic surges. The low statistical power of our study did not allow us to give insight into the effectiveness of steroids and heparin at any dosage.
PEEP Support and Prone Positioning
Continuous positive airway pressure was initiated in the majority of patients and maintained for several days. This was an absolute novelty, because we rarely had to keep patients in helmets for long. This was feasible thanks to the SICU’s high nurse-to-patient ratio and the possibility of providing monitored sedation. Patients who could no longer tolerate CPAP helmets or did not improve with CPAP support were evaluated with anesthetists for programming further management. No initial data on respiratory rate, level of hypoxemia, or oxygen support need (level of PEEP and F
Prone positioning during CPAP was implemented in 42% of our study population: P/F ratio amelioration after prone positioning was highly variable, ranging from very good P/F ratio improvements to few responses or no response. No significantly greater delta P/F ratio was seen after the first prone positioning cycle in patients who had a positive outcome, probably due to the small size of our population, but we observed a clear positive trend. Interestingly, patients showing a negative outcome had a lower percentage of long-term responses to prone positioning: 6 hours after resupination, they lost the benefit of prone positioning in terms of P/F ratio amelioration. Similarly, a greater number of patients tolerating prone positioning had a positive outcome. These data give insight on the possible benefits of prone positioning in a noninvasively supported cohort of patients, which has been mentioned in previous studies.30,31
Outcomes and Variables Associated With Negative Outcomes
After correction for age and sex, we found in multiple regression analysis that higher D-dimer and LDH values, lymphopenia, and history of diabetes were independently associated with a worse outcome. Although our results had low statistical significance, we consider the trend of the obtained odds ratios important from a clinical point of view. These results could lead to greater attention being placed on COVID-19 patients who present with these characteristics upon their arrival to the ED because they have increased risk of death or intensive care need. Clinicians should consider SICU admission for these patients in order to guarantee closer monitoring and possibly more aggressive ventilatory treatments, earlier pronation, or earlier transfer to the ICU.
Limitations
The major limitation to our study is undoubtedly its statistical power, due to its relatively low patient population. Particularly, the small number of patients who underwent pronation did not allow speculation about the efficacy of this technique, although preliminary data seem promising. However, ours is among the first studies regarding patients with COVID-19 admitted to a SICU, and these preliminary data truthfully describe the Italian, and perhaps international, experience with the first surge of the pandemic.
Conclusions
Our data highlight the primary role of the SICU in COVID-19 in adequately treating critically ill patients who have high care needs different from intubation, and who require noninvasive ventilation for prolonged times as well as frequent pronation cycles. This setting of care may represent a valid, reliable, and effective option for critically ill respiratory patients. History of diabetes, lymphopenia, and high D-dimer and LDH values are independently associated with negative outcomes, and patients presenting with these characteristics should be strictly monitored.
Acknowledgments: The authors thank the Informatica System S.R.L., as well as Allessando Mendolia for the pro bono creation of the ISCovidCollect data collecting app.
Corresponding author: Sara Abram, MD, via Coppino, 12100 Cuneo, Italy; [email protected].
Disclosures: None.
1. Plate JDJ, Leenen LPH, Houwert M, Hietbrink F. Utilisation of intermediate care units: a systematic review. Crit Care Res Pract. 2017;2017:8038460. doi:10.1155/2017/8038460
2. Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med. 2007;35(1):18-25. doi:10.1097/01.CCM.0000251821.44259.F3
3. Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315(22):2435-2441. doi:10.1001/jama.2016.6338
4. Mas A, Masip J. Noninvasive ventilation in acute respiratory failure. Int J Chron Obstruct Pulmon Dis. 2014;9:837-852. doi:10.2147/COPD.S42664
5. Bellani G, Patroniti N, Greco M, Foti G, Pesenti A. The use of helmets to deliver non-invasive continuous positive airway pressure in hypoxemic acute respiratory failure. Minerva Anestesiol. 2008;74(11):651-656.
6. Lomoro P, Verde F, Zerboni F, et al. COVID-19 pneumonia manifestations at the admission on chest ultrasound, radiographs, and CT: single-center study and comprehensive radiologic literature review. Eur J Radiol Open. 2020;7:100231. doi:10.1016/j.ejro.2020.100231
7. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373-383. doi:10.1016/0021-9681(87)90171-8
8. Guo L, Wei D, Zhang X, et al. Clinical features predicting mortality risk in patients with viral pneumonia: the MuLBSTA score. Front Microbiol. 2019;10:2752. doi:10.3389/fmicb.2019.02752
9. Lombardy Section Italian Society Infectious and Tropical Disease. Vademecum for the treatment of people with COVID-19. Edition 2.0, 13 March 2020. Infez Med. 2020;28(2):143-152.
10. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30(3):269-271. doi:10.1038/s41422-020-0282-0
11. Cao B, Wang Y, Wen D, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382(19):1787-1799. doi:10.1056/NEJMoa2001282
12. Stone JH, Frigault MJ, Serling-Boyd NJ, et al; BACC Bay Tocilizumab Trial Investigators. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020;383(24):2333-2344. doi:10.1056/NEJMoa2028836
13. Shastri MD, Stewart N, Horne J, et al. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10(5):e0126763. doi:10.1371/journal.pone.0126763
14. Milewska A, Zarebski M, Nowak P, Stozek K, Potempa J, Pyrc K. Human coronavirus NL63 utilizes heparin sulfate proteoglycans for attachment to target cells. J Virol. 2014;88(22):13221-13230. doi:10.1128/JVI.02078-14
15. Marietta M, Vandelli P, Mighali P, Vicini R, Coluccio V, D’Amico R; COVID-19 HD Study Group. Randomised controlled trial comparing efficacy and safety of high versus low low-molecular weight heparin dosages in hospitalized patients with severe COVID-19 pneumonia and coagulopathy not requiring invasive mechanical ventilation (COVID-19 HD): a structured summary of a study protocol. Trials. 2020;21(1):574. doi:10.1186/s13063-020-04475-z
16. Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med. 1995;23(10):1638-1652. doi:10.1097/00003246-199510000-00007
17. Sinha P, Calfee CS. Phenotypes in acute respiratory distress syndrome: moving towards precision medicine. Curr Opin Crit Care. 2019;25(1):12-20. doi:10.1097/MCC.0000000000000571
18. Lucchini A, Giani M, Isgrò S, Rona R, Foti G. The “helmet bundle” in COVID-19 patients undergoing non-invasive ventilation. Intensive Crit Care Nurs. 2020;58:102859. doi:10.1016/j.iccn.2020.102859
19. Ding L, Wang L, Ma W, He H. Efficacy and safety of early prone positioning combined with HFNC or NIV in moderate to severe ARDS: a multi-center prospective cohort study. Crit Care. 2020;24(1):28. doi:10.1186/s13054-020-2738-5
20. Scaravilli V, Grasselli G, Castagna L, et al. Prone positioning improves oxygenation in spontaneously breathing nonintubated patients with hypoxemic acute respiratory failure: a retrospective study. J Crit Care. 2015;30(6):1390-1394. doi:10.1016/j.jcrc.2015.07.008
21. Caputo ND, Strayer RJ, Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med. 2020;27(5):375-378. doi:10.1111/acem.13994
22. ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307(23):2526-2533. doi:10.1001/jama.2012.5669
23. Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. doi:10.1136/bmj.m1966
24. Docherty AB, Harrison EM, Green CA, et al; ISARIC4C investigators. Features of 20 133 UK patients in hospital with Covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi:10.1136/bmj.m1985
25. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775
26. Muniyappa R, Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab. 2020;318(5):E736-E741. doi:10.1152/ajpendo.00124.2020
27. Guo W, Li M, Dong Y, et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020:e3319. doi:10.1002/dmrr.3319
28. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. doi:10.1016/S0140-6736(20)30211-7
29. Kooraki S, Hosseiny M, Myers L, Gholamrezanezhad A. Coronavirus (COVID-19) outbreak: what the Department of Radiology should know. J Am Coll Radiol. 2020;17(4):447-451. doi:10.1016/j.jacr.2020.02.008
30. Coppo A, Bellani G, Winterton D, et al. Feasibility and physiological effects of prone positioning in non-intubated patients with acute respiratory failure due to COVID-19 (PRON-COVID): a prospective cohort study. Lancet Respir Med. 2020;8(8):765-774. doi:10.1016/S2213-2600(20)30268-X
31. Weatherald J, Solverson K, Zuege DJ, Loroff N, Fiest KM, Parhar KKS. Awake prone positioning for COVID-19 hypoxemic respiratory failure: a rapid review. J Crit Care. 2021;61:63-70. doi:10.1016/j.jcrc.2020.08.018
1. Plate JDJ, Leenen LPH, Houwert M, Hietbrink F. Utilisation of intermediate care units: a systematic review. Crit Care Res Pract. 2017;2017:8038460. doi:10.1155/2017/8038460
2. Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med. 2007;35(1):18-25. doi:10.1097/01.CCM.0000251821.44259.F3
3. Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315(22):2435-2441. doi:10.1001/jama.2016.6338
4. Mas A, Masip J. Noninvasive ventilation in acute respiratory failure. Int J Chron Obstruct Pulmon Dis. 2014;9:837-852. doi:10.2147/COPD.S42664
5. Bellani G, Patroniti N, Greco M, Foti G, Pesenti A. The use of helmets to deliver non-invasive continuous positive airway pressure in hypoxemic acute respiratory failure. Minerva Anestesiol. 2008;74(11):651-656.
6. Lomoro P, Verde F, Zerboni F, et al. COVID-19 pneumonia manifestations at the admission on chest ultrasound, radiographs, and CT: single-center study and comprehensive radiologic literature review. Eur J Radiol Open. 2020;7:100231. doi:10.1016/j.ejro.2020.100231
7. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373-383. doi:10.1016/0021-9681(87)90171-8
8. Guo L, Wei D, Zhang X, et al. Clinical features predicting mortality risk in patients with viral pneumonia: the MuLBSTA score. Front Microbiol. 2019;10:2752. doi:10.3389/fmicb.2019.02752
9. Lombardy Section Italian Society Infectious and Tropical Disease. Vademecum for the treatment of people with COVID-19. Edition 2.0, 13 March 2020. Infez Med. 2020;28(2):143-152.
10. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30(3):269-271. doi:10.1038/s41422-020-0282-0
11. Cao B, Wang Y, Wen D, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382(19):1787-1799. doi:10.1056/NEJMoa2001282
12. Stone JH, Frigault MJ, Serling-Boyd NJ, et al; BACC Bay Tocilizumab Trial Investigators. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020;383(24):2333-2344. doi:10.1056/NEJMoa2028836
13. Shastri MD, Stewart N, Horne J, et al. In-vitro suppression of IL-6 and IL-8 release from human pulmonary epithelial cells by non-anticoagulant fraction of enoxaparin. PLoS One. 2015;10(5):e0126763. doi:10.1371/journal.pone.0126763
14. Milewska A, Zarebski M, Nowak P, Stozek K, Potempa J, Pyrc K. Human coronavirus NL63 utilizes heparin sulfate proteoglycans for attachment to target cells. J Virol. 2014;88(22):13221-13230. doi:10.1128/JVI.02078-14
15. Marietta M, Vandelli P, Mighali P, Vicini R, Coluccio V, D’Amico R; COVID-19 HD Study Group. Randomised controlled trial comparing efficacy and safety of high versus low low-molecular weight heparin dosages in hospitalized patients with severe COVID-19 pneumonia and coagulopathy not requiring invasive mechanical ventilation (COVID-19 HD): a structured summary of a study protocol. Trials. 2020;21(1):574. doi:10.1186/s13063-020-04475-z
16. Marshall JC, Cook DJ, Christou NV, Bernard GR, Sprung CL, Sibbald WJ. Multiple organ dysfunction score: a reliable descriptor of a complex clinical outcome. Crit Care Med. 1995;23(10):1638-1652. doi:10.1097/00003246-199510000-00007
17. Sinha P, Calfee CS. Phenotypes in acute respiratory distress syndrome: moving towards precision medicine. Curr Opin Crit Care. 2019;25(1):12-20. doi:10.1097/MCC.0000000000000571
18. Lucchini A, Giani M, Isgrò S, Rona R, Foti G. The “helmet bundle” in COVID-19 patients undergoing non-invasive ventilation. Intensive Crit Care Nurs. 2020;58:102859. doi:10.1016/j.iccn.2020.102859
19. Ding L, Wang L, Ma W, He H. Efficacy and safety of early prone positioning combined with HFNC or NIV in moderate to severe ARDS: a multi-center prospective cohort study. Crit Care. 2020;24(1):28. doi:10.1186/s13054-020-2738-5
20. Scaravilli V, Grasselli G, Castagna L, et al. Prone positioning improves oxygenation in spontaneously breathing nonintubated patients with hypoxemic acute respiratory failure: a retrospective study. J Crit Care. 2015;30(6):1390-1394. doi:10.1016/j.jcrc.2015.07.008
21. Caputo ND, Strayer RJ, Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med. 2020;27(5):375-378. doi:10.1111/acem.13994
22. ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307(23):2526-2533. doi:10.1001/jama.2012.5669
23. Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. doi:10.1136/bmj.m1966
24. Docherty AB, Harrison EM, Green CA, et al; ISARIC4C investigators. Features of 20 133 UK patients in hospital with Covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369:m1985. doi:10.1136/bmj.m1985
25. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775
26. Muniyappa R, Gubbi S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab. 2020;318(5):E736-E741. doi:10.1152/ajpendo.00124.2020
27. Guo W, Li M, Dong Y, et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020:e3319. doi:10.1002/dmrr.3319
28. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. doi:10.1016/S0140-6736(20)30211-7
29. Kooraki S, Hosseiny M, Myers L, Gholamrezanezhad A. Coronavirus (COVID-19) outbreak: what the Department of Radiology should know. J Am Coll Radiol. 2020;17(4):447-451. doi:10.1016/j.jacr.2020.02.008
30. Coppo A, Bellani G, Winterton D, et al. Feasibility and physiological effects of prone positioning in non-intubated patients with acute respiratory failure due to COVID-19 (PRON-COVID): a prospective cohort study. Lancet Respir Med. 2020;8(8):765-774. doi:10.1016/S2213-2600(20)30268-X
31. Weatherald J, Solverson K, Zuege DJ, Loroff N, Fiest KM, Parhar KKS. Awake prone positioning for COVID-19 hypoxemic respiratory failure: a rapid review. J Crit Care. 2021;61:63-70. doi:10.1016/j.jcrc.2020.08.018
Structural Ableism: Defining Standards of Care Amid Crisis and Inequity
Equitable Standards for All Patients in a Crisis
Health care delivered during a pandemic instantiates medicine’s perspectives on the value of human life in clinical scenarios where resource allocation is limited. The COVID-19 pandemic has fostered dialogue and debate around the ethical principles that underly such resource allocation, which generally balance (1) utilitarian optimization of resources, (2) equality or equity in health access, (3) the instrumental value of individuals as agents in society, and (4) prioritizing the “worst off” in their natural history of disease.1,2 State legislatures and health systems have responded to the challeges posed by COVID-19 by considering both the scarcity of intensive care resources, such as mechanical ventilation and hemodialysis, and the clinical criteria to be used for determining which patients should receive said resources. These crisis guidelines have yielded several concerning themes vis-à-vis equitable distribution of health care resources, particularly when the disability status of patients is considered alongside life-expectancy or quality of life.3
Crisis standards of care (CSC) prioritize population-level health under a utilitarian paradigm, explicitly maximizing “life-years” within a population of patients rather than the life of any individual patient.4 Debated during initial COVID surges, these CSC guidelines have recently been enacted at the state level in several settings, including Alaska and Idaho.5 In a setting with scarce intensive care resources, balancing health equity in access to these resources against population-based survival metrics has been a challenge for commissions considering CSC.6,7 This need for balance has further promoted systemic views of “disability,” raising concern for structural “ableism” and highlighting the need for greater “ability awareness” in clinicians’ continued professional learning.
Structural Ableism: Defining Perspectives to Address Health Equity
Ableism has been defined as “a system that places value on people’s bodies and minds, based on societally constructed ideas of normalcy, intelligence, excellence, and productivity…[and] leads to people and society determining who is valuable and worthy based on their appearance and/or their ability to satisfactorily [re]produce, excel, and ‘behave.’”8 Regarding CSC, concerns about systemic bias in guideline design were raised early by disability advocacy groups during comment periods.9,10 More broadly, concerns about ableism sit alongside many deeply rooted societal perspectives of disabled individuals as pitiable or, conversely, heroic for having “overcome” their disability in some way. As a physician who sits in a manual wheelchair with paraplegia and mobility impairment, I have equally been subject to inappropriate bias and inappropriate praise for living in a wheelchair. I have also wondered, alongside my patients living with different levels of mobility or ability, why others often view us as “worse off.” Addressing directly whether disabled individuals are “worse off,” disability rights attorney and advocate Harriet McBryde Johnson has articulated a predominant sentiment among persons living with unique or different abilities:
Are we “worse off”? I don’t think so. Not in any meaningful way. There are too many variables. For those of us with congenital conditions, disability shapes all we are. Those disabled later in life adapt. We take constraints that no one would choose and build rich and satisfying lives within them. We enjoy pleasures other people enjoy and pleasures peculiarly our own. We have something the world needs.11
Many physician colleagues have common, invisible diseases such as diabetes and heart disease; fewer colleagues share conditions that are as visible as my spinal cord injury, as readily apparent to patients upon my entry to their hospital rooms. This simultaneous and inescapable identity as both patient and provider has afforded me wonderful doctor-patient interactions, particularly with those patients who appreciate how my patient experience impacts my ability to partially understand theirs. However, this simultaneous identity as doctor and patient also informed my personal and professional concerns regarding structural ableism as I considered scoring my own acutely ill hospital medicine patients with CSC triage scores in April 2020.
As a practicing hospital medicine physician, I have been emboldened by the efforts of my fellow clinicians amid COVID-19; their efforts have reaffirmed all the reasons I pursued a career in medicine. However, when I heard my clinical colleagues’ first explanation of the Massachusetts CSC guidelines in April 2020, I raised my hand to ask whether the “life-years” to which the guidelines referred were quality-adjusted. My concern regarding the implicit use of quality-adjusted life years (QALY) or disability-adjusted life years in clinical decision-making and implementation of these guidelines was validated when no clinical leaders could address this question directly. Sitting on the CSC committee for my hospital during this time was an honor. However, it was disconcerting to hear many clinicians’ unease when estimating mean survival for common chronic diseases, ranging from end-stage renal disease to advanced heart failure. If my expert colleagues, clinical specialists in kidney and heart disease, could not confidently apply mean survival estimates to multimorbid hospital patients, then idiosyncratic clinical judgment was sure to have a heavy hand in any calculation of “life-years.” Thus, my primary concern was that clinicians using triage heuristics would be subject to bias, regardless of their intention, and negatively adjust for the quality of a disabled life in their CSC triage scoring. My secondary concern was that the CSC guidelines themselves included systemic bias against disabled individuals.
According to CSC schema, triage scores index heavily on Sequential Organ Failure Assessment (SOFA) scores to define short-term survival; SOFA scores are partially driven by the Glasgow Coma Scale (GCS). Following professional and public comment periods, CSC guidelines in Massachusetts were revised to, among other critical points of revision, change prognostic estimation via “life years” in favor of generic estimation of short-term survival (Table). I wondered, if I presented to an emergency department with severe COVID-19 and was scored with the GCS for the purpose of making a CSC ventilator triage decision, how would my complete paraplegia and lower-extremity motor impairment be accounted for by a clinician assessing “best motor response” in the GCS? The purpose of these scores is to act algorithmically, to guide clinicians whose cognitive load and time limitations may not allow for adjustment of these algorithms based on the individual patient in front of them. Individualization of clinical decisions is part of medicine’s art, but is difficult in the best of times and no easier during a crisis in care delivery. As CSC triage scores were amended and addended throughout 2020, I returned to the COVID wards, time and again wondering, “What have we learned about systemic bias and health inequity in the CSC process and the pandemic broadly, with specific regard to disability?”
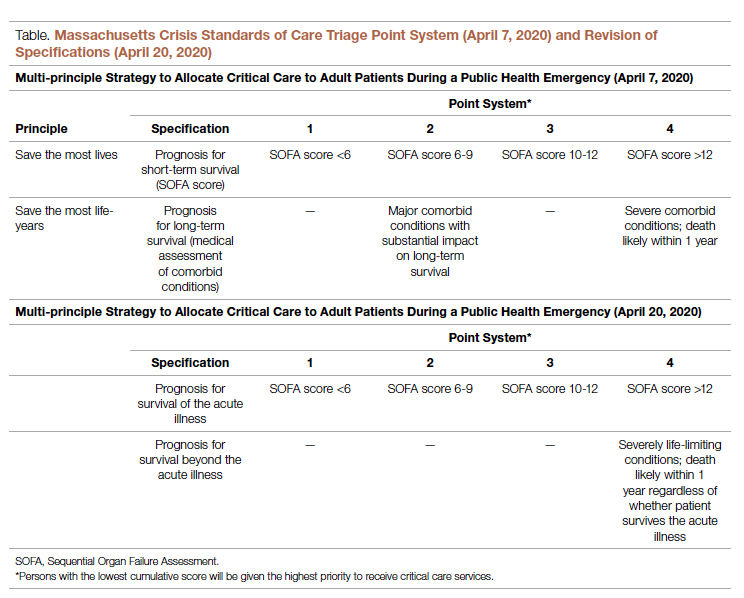
Ability Awareness: Room for Our Improvement
Unfortunately, there is reason to believe that clinical judgment is impaired by structural ableism. In seminal work on this topic, Gerhart et al12 demonstrated that clinicians considered spinal cord injury (SCI) survivors to have low self-perceptions of worthiness, overall negative attitudes, and low self-esteem as compared to able-bodied individuals. However, surveyed SCI survivors generally had similar self-perceptions of worth and positivity as compared to ”able-bodied” clinicians.12 For providers who care for persons with disabilities, the majority (82.4%) have rated their disabled patients’ quality of life as worse.13 It is no wonder that patients with disabilities are more likely to feel that their doctor-patient relationship is impacted by lack of understanding, negative sentiment, or simple lack of listening.14 Generally, this poor doctor-patient relationship with disabled patients is exacerbated by poor exposure of medical trainees to disability education; only 34.2% of internal medicine residents recall any form of disability education in medical school, while only 52% of medical school deans report having disability educational content in their curricula.15,16 There is a similar lack of disability representation in the population of medical trainees themselves. While approximately 20% of the American population lives with a disability, less than 2% of American medical students have a disability.17-19
While representation of disabled populations in medical practice remains poor, disabled patients are generally less likely to receive age-appropriate prevention, appropriate access to care, and equal access to treatment.20-22 “Diagnostic overshadowing” refers to clinicians’ attribution of nonspecific signs or symptoms to a patient’s chronic disability as opposed to acute illness.23 This phenomenon has led to higher rates of preventable malignancy in disabled patients and misattribution of common somatic symptoms to intellectual disability.24,25 With this disparity in place as status quo for health care delivery to disabled populations, it is no surprise that certain portions of the disabled population have accounted for disproportionate mortality due to COVID-19.26,27Disability advocates have called for “nothing about us without us,” a phrase associated with the United Nations Convention on the Rights of Persons with Disabilities. Understanding the profound neurodiversity among several forms of sensory and cognitive disabilities, as well as the functional difference between cognitive disabilities, mobility impairment, and inability to meet one’s instrumental activities of daily living independently, others have proposed a unique approach to certain disabled populations in COVID care.28 My own perspective is that definite progress may require a more general understanding of the prevalence of disability by clinicians, both via medical training and by directly addressing health equity for disabled populations in such calculations as the CSC. Systemic ableism is apparent in our most common clinical scoring systems, ranging from the GCS and Functional Assessment Staging Table to the Eastern Cooperative Oncology Group and Karnofsky Performance Status scales. I have reexamined these scoring systems in my own understanding given their general equation of ambulation with ability or normalcy. As a doctor in a manual wheelchair who values greatly my personal quality of life and professional contribution to patient care, I worry that these scoring systems inherently discount my own equitable access to care. Individualization of patients’ particular abilities in the context of these scales must occur alongside evidence-based, guideline-directed management via these scoring systems.
Conclusion: Future Orientation
Updated CSC guidelines have accounted for the unique considerations of disabled patients by effectively caveating their scoring algorithms, directing clinicians via disclaimers to uniquely consider their disabled patients in clinical judgement. This is a first step, but it is also one that erodes the value of algorithms, which generally obviate more deliberative thinking and individualization. For our patients who lack certain abilities, as CSC continue to be activated in several states, we have an opportunity to pursue more inherently equitable solutions before further suffering accrues.29 By way of example, adaptations to scoring systems that leverage QALYs for value-based drug pricing indices have been proposed by organizations like the Institute for Clinical and Economic Review, which proposed the Equal-Value-of Life-Years-Gained framework to inform QALY-based arbitration of drug pricing.30 This is not a perfect rubric but instead represents an attempt to balance consideration of drugs, as has been done with ventilators during the pandemic, as a scare and expensive resource while addressing the just concerns of advocacy groups in structural ableism.
Resource stewardship during a crisis should not discount those states of human life that are perceived to be less desirable, particularly if they are not experienced as less desirable but are experienced uniquely. Instead, we should consider equitably measuring our intervention to match a patient’s needs, as we would dose-adjust a medication for renal function or consider minimally invasive procedures for multimorbid patients. COVID-19 has reflected our profession’s ethical adaptation during crisis as resources have become scarce; there is no better time to define solutions for health equity. We should now be concerned equally by the influence our personal biases have on our clinical practice and by the way in which these crisis standards will influence patients’ perception of and trust in their care providers during periods of perceived plentiful resources in the future. Health care resources are always limited, allocated according to societal values; if we value health equity for people of all abilities, then we will consider these abilities equitably as we pursue new standards for health care delivery.
Corresponding author: Gregory D. Snyder, MD, MBA, 2014 Washington Street, Newton, MA 02462; [email protected].
Disclosures: None.
1. Emanuel EJ, Persad G, Upshur R, et al. Fair Allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055. doi:10.1056/NEJMsb2005114
2. Savulescu J, Persson I, Wilkinson D. Utilitarianism and the pandemic. Bioethics. 2020;34(6):620-632. doi:10.1111/bioe.12771
3. Mello MM, Persad G, White DB. Respecting disability rights - toward improved crisis standards of care. N Engl J Med. 2020;383(5):e26. doi: 10.1056/NEJMp2011997
4. The Commonwealth of Massachusetts Executive Office of Health and Human Services Department of Public Health. Crisis Standards of Care Planning Guidance for the COVID-19 Pandemic. April 7, 2020. https://d279m997dpfwgl.cloudfront.net/wp/2020/04/CSC_April-7_2020.pdf
5. Knowles H. Hospitals overwhelmed by covid are turning to ‘crisis standards of care.’ What does that mean? The Washington Post. September 21, 2021. Accessed January 24, 2022. https://www.washingtonpost.com/health/2021/09/22/crisis-standards-of-care/
6. Hick JL, Hanfling D, Wynia MK, Toner E. Crisis standards of care and COVID-19: What did we learn? How do we ensure equity? What should we do? NAM Perspect. 2021;2021:10.31478/202108e. doi:10.31478/202108e
7. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
8. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
9. Kukla E. My life is more ‘disposable’ during this pandemic. The New York Times. March 19, 2020. Accessed January 24, 2022. https://www.nytimes.com/2020/03/19/opinion/coronavirus-disabled-health-care.html
10. CPR and Coalition Partners Secure Important Changes in Massachusetts’ Crisis Standards of Care. Center for Public Representation. December 1, 2020. Accessed January 24, 2022. https://www.centerforpublicrep.org/news/cpr-and-coalition-partners-secure-important-changes-in-massachusetts-crisis-standards-of-care/
11. Johnson HM. Unspeakable conversations. The New York Times. February 16, 2003. Accessed January 24, 2022. https://www.nytimes.com/2003/02/16/magazine/unspeakable-conversations.html
12. Gerhart KA, Koziol-McLain J, Lowenstein SR, Whiteneck GG. Quality of life following spinal cord injury: knowledge and attitudes of emergency care providers. Ann Emerg Med. 1994;23(4):807-812. doi:10.1016/s0196-0644(94)70318-3
13. Iezzoni LI, Rao SR, Ressalam J, et al. Physicians’ perceptions of people with disability and their health care. Health Aff (Millwood). 2021;40(2):297-306. doi:10.1377/hlthaff.2020.01452
14. Smith DL. Disparities in patient-physician communication for persons with a disability from the 2006 Medical Expenditure Panel Survey (MEPS). Disabil Health J. 2009;2(4):206-215. doi:10.1016/j.dhjo.2009.06.002
15. Stillman MD, Ankam N, Mallow M, Capron M, Williams S. A survey of internal and family medicine residents: Assessment of disability-specific education and knowledge. Disabil Health J. 2021;14(2):101011. doi:10.1016/j.dhjo.2020.101011
16. Seidel E, Crowe S. The state of disability awareness in American medical schools. Am J Phys Med Rehabil. 2017;96(9):673-676. doi:10.1097/PHM.0000000000000719
17. Okoro CA, Hollis ND, Cyrus AC, Griffin-Blake S. Prevalence of disabilities and health care access by disability status and type among adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(32):882-887. doi:10.15585/mmwr.mm6732a3
18. Peacock G, Iezzoni LI, Harkin TR. Health care for Americans with disabilities--25 years after the ADA. N Engl J Med. 2015;373(10):892-893. doi:10.1056/NEJMp1508854
19. DeLisa JA, Thomas P. Physicians with disabilities and the physician workforce: a need to reassess our policies. Am J Phys Med Rehabil. 2005;84(1):5-11. doi:10.1097/01.phm.0000153323.28396.de
20. Disability and Health. Healthy People 2020. Accessed January 24, 2022. https://www.healthypeople.gov/2020/topics-objectives/topic/disability-and-health
21. Lagu T, Hannon NS, Rothberg MB, et al. Access to subspecialty care for patients with mobility impairment: a survey. Ann Intern Med. 2013;158(6):441-446. doi: 10.7326/0003-4819-158-6-201303190-00003
22. McCarthy EP, Ngo LH, Roetzheim RG, et al. Disparities in breast cancer treatment and survival for women with disabilities. Ann Intern Med. 2006;145(9):637-645. doi: 10.7326/0003-4819-145-9-200611070-00005
23. Javaid A, Nakata V, Michael D. Diagnostic overshadowing in learning disability: think beyond the disability. Prog Neurol Psychiatry. 2019;23:8-10.
24. Iezzoni LI, Rao SR, Agaronnik ND, El-Jawahri A. Cross-sectional analysis of the associations between four common cancers and disability. J Natl Compr Canc Netw. 2020;18(8):1031-1044. doi:10.6004/jnccn.2020.7551
25. Sanders JS, Keller S, Aravamuthan BR. Caring for individuals with intellectual and developmental disabilities in the COVID-19 crisis. Neurol Clin Pract. 2021;11(2):e174-e178. doi:10.1212/CPJ.0000000000000886
26. Landes SD, Turk MA, Formica MK, McDonald KE, Stevens JD. COVID-19 outcomes among people with intellectual and developmental disability living in residential group homes in New York State. Disabil Health J. 2020;13(4):100969. doi:10.1016/j.dhjo.2020.100969
27. Gleason J, Ross W, Fossi A, Blonksy H, Tobias J, Stephens M. The devastating impact of Covid-19 on individuals with intellectual disabilities in the United States. NEJM Catalyst. 2021.doi.org/10.1056/CAT.21.0051
28. Nankervis K, Chan J. Applying the CRPD to people with intellectual and developmental disability with behaviors of concern during COVID-19. J Policy Pract Intellect Disabil. 2021:10.1111/jppi.12374. doi:10.1111/jppi.12374
29. Alaska Department of Health and Social Services, Division of Public Health, Rural and Community Health Systems. Patient care strategies for scarce resource situations. Version 1. August 2021. Accessed November 11, 2021, https://dhss.alaska.gov/dph/Epi/id/SiteAssets/Pages/HumanCoV/SOA_DHSS_CrisisStandardsOfCare.pdf
30. Cost-effectiveness, the QALY, and the evlyg. ICER. May 21, 2021. Accessed January 24, 2022. https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/
Equitable Standards for All Patients in a Crisis
Health care delivered during a pandemic instantiates medicine’s perspectives on the value of human life in clinical scenarios where resource allocation is limited. The COVID-19 pandemic has fostered dialogue and debate around the ethical principles that underly such resource allocation, which generally balance (1) utilitarian optimization of resources, (2) equality or equity in health access, (3) the instrumental value of individuals as agents in society, and (4) prioritizing the “worst off” in their natural history of disease.1,2 State legislatures and health systems have responded to the challeges posed by COVID-19 by considering both the scarcity of intensive care resources, such as mechanical ventilation and hemodialysis, and the clinical criteria to be used for determining which patients should receive said resources. These crisis guidelines have yielded several concerning themes vis-à-vis equitable distribution of health care resources, particularly when the disability status of patients is considered alongside life-expectancy or quality of life.3
Crisis standards of care (CSC) prioritize population-level health under a utilitarian paradigm, explicitly maximizing “life-years” within a population of patients rather than the life of any individual patient.4 Debated during initial COVID surges, these CSC guidelines have recently been enacted at the state level in several settings, including Alaska and Idaho.5 In a setting with scarce intensive care resources, balancing health equity in access to these resources against population-based survival metrics has been a challenge for commissions considering CSC.6,7 This need for balance has further promoted systemic views of “disability,” raising concern for structural “ableism” and highlighting the need for greater “ability awareness” in clinicians’ continued professional learning.
Structural Ableism: Defining Perspectives to Address Health Equity
Ableism has been defined as “a system that places value on people’s bodies and minds, based on societally constructed ideas of normalcy, intelligence, excellence, and productivity…[and] leads to people and society determining who is valuable and worthy based on their appearance and/or their ability to satisfactorily [re]produce, excel, and ‘behave.’”8 Regarding CSC, concerns about systemic bias in guideline design were raised early by disability advocacy groups during comment periods.9,10 More broadly, concerns about ableism sit alongside many deeply rooted societal perspectives of disabled individuals as pitiable or, conversely, heroic for having “overcome” their disability in some way. As a physician who sits in a manual wheelchair with paraplegia and mobility impairment, I have equally been subject to inappropriate bias and inappropriate praise for living in a wheelchair. I have also wondered, alongside my patients living with different levels of mobility or ability, why others often view us as “worse off.” Addressing directly whether disabled individuals are “worse off,” disability rights attorney and advocate Harriet McBryde Johnson has articulated a predominant sentiment among persons living with unique or different abilities:
Are we “worse off”? I don’t think so. Not in any meaningful way. There are too many variables. For those of us with congenital conditions, disability shapes all we are. Those disabled later in life adapt. We take constraints that no one would choose and build rich and satisfying lives within them. We enjoy pleasures other people enjoy and pleasures peculiarly our own. We have something the world needs.11
Many physician colleagues have common, invisible diseases such as diabetes and heart disease; fewer colleagues share conditions that are as visible as my spinal cord injury, as readily apparent to patients upon my entry to their hospital rooms. This simultaneous and inescapable identity as both patient and provider has afforded me wonderful doctor-patient interactions, particularly with those patients who appreciate how my patient experience impacts my ability to partially understand theirs. However, this simultaneous identity as doctor and patient also informed my personal and professional concerns regarding structural ableism as I considered scoring my own acutely ill hospital medicine patients with CSC triage scores in April 2020.
As a practicing hospital medicine physician, I have been emboldened by the efforts of my fellow clinicians amid COVID-19; their efforts have reaffirmed all the reasons I pursued a career in medicine. However, when I heard my clinical colleagues’ first explanation of the Massachusetts CSC guidelines in April 2020, I raised my hand to ask whether the “life-years” to which the guidelines referred were quality-adjusted. My concern regarding the implicit use of quality-adjusted life years (QALY) or disability-adjusted life years in clinical decision-making and implementation of these guidelines was validated when no clinical leaders could address this question directly. Sitting on the CSC committee for my hospital during this time was an honor. However, it was disconcerting to hear many clinicians’ unease when estimating mean survival for common chronic diseases, ranging from end-stage renal disease to advanced heart failure. If my expert colleagues, clinical specialists in kidney and heart disease, could not confidently apply mean survival estimates to multimorbid hospital patients, then idiosyncratic clinical judgment was sure to have a heavy hand in any calculation of “life-years.” Thus, my primary concern was that clinicians using triage heuristics would be subject to bias, regardless of their intention, and negatively adjust for the quality of a disabled life in their CSC triage scoring. My secondary concern was that the CSC guidelines themselves included systemic bias against disabled individuals.
According to CSC schema, triage scores index heavily on Sequential Organ Failure Assessment (SOFA) scores to define short-term survival; SOFA scores are partially driven by the Glasgow Coma Scale (GCS). Following professional and public comment periods, CSC guidelines in Massachusetts were revised to, among other critical points of revision, change prognostic estimation via “life years” in favor of generic estimation of short-term survival (Table). I wondered, if I presented to an emergency department with severe COVID-19 and was scored with the GCS for the purpose of making a CSC ventilator triage decision, how would my complete paraplegia and lower-extremity motor impairment be accounted for by a clinician assessing “best motor response” in the GCS? The purpose of these scores is to act algorithmically, to guide clinicians whose cognitive load and time limitations may not allow for adjustment of these algorithms based on the individual patient in front of them. Individualization of clinical decisions is part of medicine’s art, but is difficult in the best of times and no easier during a crisis in care delivery. As CSC triage scores were amended and addended throughout 2020, I returned to the COVID wards, time and again wondering, “What have we learned about systemic bias and health inequity in the CSC process and the pandemic broadly, with specific regard to disability?”

Ability Awareness: Room for Our Improvement
Unfortunately, there is reason to believe that clinical judgment is impaired by structural ableism. In seminal work on this topic, Gerhart et al12 demonstrated that clinicians considered spinal cord injury (SCI) survivors to have low self-perceptions of worthiness, overall negative attitudes, and low self-esteem as compared to able-bodied individuals. However, surveyed SCI survivors generally had similar self-perceptions of worth and positivity as compared to ”able-bodied” clinicians.12 For providers who care for persons with disabilities, the majority (82.4%) have rated their disabled patients’ quality of life as worse.13 It is no wonder that patients with disabilities are more likely to feel that their doctor-patient relationship is impacted by lack of understanding, negative sentiment, or simple lack of listening.14 Generally, this poor doctor-patient relationship with disabled patients is exacerbated by poor exposure of medical trainees to disability education; only 34.2% of internal medicine residents recall any form of disability education in medical school, while only 52% of medical school deans report having disability educational content in their curricula.15,16 There is a similar lack of disability representation in the population of medical trainees themselves. While approximately 20% of the American population lives with a disability, less than 2% of American medical students have a disability.17-19
While representation of disabled populations in medical practice remains poor, disabled patients are generally less likely to receive age-appropriate prevention, appropriate access to care, and equal access to treatment.20-22 “Diagnostic overshadowing” refers to clinicians’ attribution of nonspecific signs or symptoms to a patient’s chronic disability as opposed to acute illness.23 This phenomenon has led to higher rates of preventable malignancy in disabled patients and misattribution of common somatic symptoms to intellectual disability.24,25 With this disparity in place as status quo for health care delivery to disabled populations, it is no surprise that certain portions of the disabled population have accounted for disproportionate mortality due to COVID-19.26,27Disability advocates have called for “nothing about us without us,” a phrase associated with the United Nations Convention on the Rights of Persons with Disabilities. Understanding the profound neurodiversity among several forms of sensory and cognitive disabilities, as well as the functional difference between cognitive disabilities, mobility impairment, and inability to meet one’s instrumental activities of daily living independently, others have proposed a unique approach to certain disabled populations in COVID care.28 My own perspective is that definite progress may require a more general understanding of the prevalence of disability by clinicians, both via medical training and by directly addressing health equity for disabled populations in such calculations as the CSC. Systemic ableism is apparent in our most common clinical scoring systems, ranging from the GCS and Functional Assessment Staging Table to the Eastern Cooperative Oncology Group and Karnofsky Performance Status scales. I have reexamined these scoring systems in my own understanding given their general equation of ambulation with ability or normalcy. As a doctor in a manual wheelchair who values greatly my personal quality of life and professional contribution to patient care, I worry that these scoring systems inherently discount my own equitable access to care. Individualization of patients’ particular abilities in the context of these scales must occur alongside evidence-based, guideline-directed management via these scoring systems.
Conclusion: Future Orientation
Updated CSC guidelines have accounted for the unique considerations of disabled patients by effectively caveating their scoring algorithms, directing clinicians via disclaimers to uniquely consider their disabled patients in clinical judgement. This is a first step, but it is also one that erodes the value of algorithms, which generally obviate more deliberative thinking and individualization. For our patients who lack certain abilities, as CSC continue to be activated in several states, we have an opportunity to pursue more inherently equitable solutions before further suffering accrues.29 By way of example, adaptations to scoring systems that leverage QALYs for value-based drug pricing indices have been proposed by organizations like the Institute for Clinical and Economic Review, which proposed the Equal-Value-of Life-Years-Gained framework to inform QALY-based arbitration of drug pricing.30 This is not a perfect rubric but instead represents an attempt to balance consideration of drugs, as has been done with ventilators during the pandemic, as a scare and expensive resource while addressing the just concerns of advocacy groups in structural ableism.
Resource stewardship during a crisis should not discount those states of human life that are perceived to be less desirable, particularly if they are not experienced as less desirable but are experienced uniquely. Instead, we should consider equitably measuring our intervention to match a patient’s needs, as we would dose-adjust a medication for renal function or consider minimally invasive procedures for multimorbid patients. COVID-19 has reflected our profession’s ethical adaptation during crisis as resources have become scarce; there is no better time to define solutions for health equity. We should now be concerned equally by the influence our personal biases have on our clinical practice and by the way in which these crisis standards will influence patients’ perception of and trust in their care providers during periods of perceived plentiful resources in the future. Health care resources are always limited, allocated according to societal values; if we value health equity for people of all abilities, then we will consider these abilities equitably as we pursue new standards for health care delivery.
Corresponding author: Gregory D. Snyder, MD, MBA, 2014 Washington Street, Newton, MA 02462; [email protected].
Disclosures: None.
Equitable Standards for All Patients in a Crisis
Health care delivered during a pandemic instantiates medicine’s perspectives on the value of human life in clinical scenarios where resource allocation is limited. The COVID-19 pandemic has fostered dialogue and debate around the ethical principles that underly such resource allocation, which generally balance (1) utilitarian optimization of resources, (2) equality or equity in health access, (3) the instrumental value of individuals as agents in society, and (4) prioritizing the “worst off” in their natural history of disease.1,2 State legislatures and health systems have responded to the challeges posed by COVID-19 by considering both the scarcity of intensive care resources, such as mechanical ventilation and hemodialysis, and the clinical criteria to be used for determining which patients should receive said resources. These crisis guidelines have yielded several concerning themes vis-à-vis equitable distribution of health care resources, particularly when the disability status of patients is considered alongside life-expectancy or quality of life.3
Crisis standards of care (CSC) prioritize population-level health under a utilitarian paradigm, explicitly maximizing “life-years” within a population of patients rather than the life of any individual patient.4 Debated during initial COVID surges, these CSC guidelines have recently been enacted at the state level in several settings, including Alaska and Idaho.5 In a setting with scarce intensive care resources, balancing health equity in access to these resources against population-based survival metrics has been a challenge for commissions considering CSC.6,7 This need for balance has further promoted systemic views of “disability,” raising concern for structural “ableism” and highlighting the need for greater “ability awareness” in clinicians’ continued professional learning.
Structural Ableism: Defining Perspectives to Address Health Equity
Ableism has been defined as “a system that places value on people’s bodies and minds, based on societally constructed ideas of normalcy, intelligence, excellence, and productivity…[and] leads to people and society determining who is valuable and worthy based on their appearance and/or their ability to satisfactorily [re]produce, excel, and ‘behave.’”8 Regarding CSC, concerns about systemic bias in guideline design were raised early by disability advocacy groups during comment periods.9,10 More broadly, concerns about ableism sit alongside many deeply rooted societal perspectives of disabled individuals as pitiable or, conversely, heroic for having “overcome” their disability in some way. As a physician who sits in a manual wheelchair with paraplegia and mobility impairment, I have equally been subject to inappropriate bias and inappropriate praise for living in a wheelchair. I have also wondered, alongside my patients living with different levels of mobility or ability, why others often view us as “worse off.” Addressing directly whether disabled individuals are “worse off,” disability rights attorney and advocate Harriet McBryde Johnson has articulated a predominant sentiment among persons living with unique or different abilities:
Are we “worse off”? I don’t think so. Not in any meaningful way. There are too many variables. For those of us with congenital conditions, disability shapes all we are. Those disabled later in life adapt. We take constraints that no one would choose and build rich and satisfying lives within them. We enjoy pleasures other people enjoy and pleasures peculiarly our own. We have something the world needs.11
Many physician colleagues have common, invisible diseases such as diabetes and heart disease; fewer colleagues share conditions that are as visible as my spinal cord injury, as readily apparent to patients upon my entry to their hospital rooms. This simultaneous and inescapable identity as both patient and provider has afforded me wonderful doctor-patient interactions, particularly with those patients who appreciate how my patient experience impacts my ability to partially understand theirs. However, this simultaneous identity as doctor and patient also informed my personal and professional concerns regarding structural ableism as I considered scoring my own acutely ill hospital medicine patients with CSC triage scores in April 2020.
As a practicing hospital medicine physician, I have been emboldened by the efforts of my fellow clinicians amid COVID-19; their efforts have reaffirmed all the reasons I pursued a career in medicine. However, when I heard my clinical colleagues’ first explanation of the Massachusetts CSC guidelines in April 2020, I raised my hand to ask whether the “life-years” to which the guidelines referred were quality-adjusted. My concern regarding the implicit use of quality-adjusted life years (QALY) or disability-adjusted life years in clinical decision-making and implementation of these guidelines was validated when no clinical leaders could address this question directly. Sitting on the CSC committee for my hospital during this time was an honor. However, it was disconcerting to hear many clinicians’ unease when estimating mean survival for common chronic diseases, ranging from end-stage renal disease to advanced heart failure. If my expert colleagues, clinical specialists in kidney and heart disease, could not confidently apply mean survival estimates to multimorbid hospital patients, then idiosyncratic clinical judgment was sure to have a heavy hand in any calculation of “life-years.” Thus, my primary concern was that clinicians using triage heuristics would be subject to bias, regardless of their intention, and negatively adjust for the quality of a disabled life in their CSC triage scoring. My secondary concern was that the CSC guidelines themselves included systemic bias against disabled individuals.
According to CSC schema, triage scores index heavily on Sequential Organ Failure Assessment (SOFA) scores to define short-term survival; SOFA scores are partially driven by the Glasgow Coma Scale (GCS). Following professional and public comment periods, CSC guidelines in Massachusetts were revised to, among other critical points of revision, change prognostic estimation via “life years” in favor of generic estimation of short-term survival (Table). I wondered, if I presented to an emergency department with severe COVID-19 and was scored with the GCS for the purpose of making a CSC ventilator triage decision, how would my complete paraplegia and lower-extremity motor impairment be accounted for by a clinician assessing “best motor response” in the GCS? The purpose of these scores is to act algorithmically, to guide clinicians whose cognitive load and time limitations may not allow for adjustment of these algorithms based on the individual patient in front of them. Individualization of clinical decisions is part of medicine’s art, but is difficult in the best of times and no easier during a crisis in care delivery. As CSC triage scores were amended and addended throughout 2020, I returned to the COVID wards, time and again wondering, “What have we learned about systemic bias and health inequity in the CSC process and the pandemic broadly, with specific regard to disability?”

Ability Awareness: Room for Our Improvement
Unfortunately, there is reason to believe that clinical judgment is impaired by structural ableism. In seminal work on this topic, Gerhart et al12 demonstrated that clinicians considered spinal cord injury (SCI) survivors to have low self-perceptions of worthiness, overall negative attitudes, and low self-esteem as compared to able-bodied individuals. However, surveyed SCI survivors generally had similar self-perceptions of worth and positivity as compared to ”able-bodied” clinicians.12 For providers who care for persons with disabilities, the majority (82.4%) have rated their disabled patients’ quality of life as worse.13 It is no wonder that patients with disabilities are more likely to feel that their doctor-patient relationship is impacted by lack of understanding, negative sentiment, or simple lack of listening.14 Generally, this poor doctor-patient relationship with disabled patients is exacerbated by poor exposure of medical trainees to disability education; only 34.2% of internal medicine residents recall any form of disability education in medical school, while only 52% of medical school deans report having disability educational content in their curricula.15,16 There is a similar lack of disability representation in the population of medical trainees themselves. While approximately 20% of the American population lives with a disability, less than 2% of American medical students have a disability.17-19
While representation of disabled populations in medical practice remains poor, disabled patients are generally less likely to receive age-appropriate prevention, appropriate access to care, and equal access to treatment.20-22 “Diagnostic overshadowing” refers to clinicians’ attribution of nonspecific signs or symptoms to a patient’s chronic disability as opposed to acute illness.23 This phenomenon has led to higher rates of preventable malignancy in disabled patients and misattribution of common somatic symptoms to intellectual disability.24,25 With this disparity in place as status quo for health care delivery to disabled populations, it is no surprise that certain portions of the disabled population have accounted for disproportionate mortality due to COVID-19.26,27Disability advocates have called for “nothing about us without us,” a phrase associated with the United Nations Convention on the Rights of Persons with Disabilities. Understanding the profound neurodiversity among several forms of sensory and cognitive disabilities, as well as the functional difference between cognitive disabilities, mobility impairment, and inability to meet one’s instrumental activities of daily living independently, others have proposed a unique approach to certain disabled populations in COVID care.28 My own perspective is that definite progress may require a more general understanding of the prevalence of disability by clinicians, both via medical training and by directly addressing health equity for disabled populations in such calculations as the CSC. Systemic ableism is apparent in our most common clinical scoring systems, ranging from the GCS and Functional Assessment Staging Table to the Eastern Cooperative Oncology Group and Karnofsky Performance Status scales. I have reexamined these scoring systems in my own understanding given their general equation of ambulation with ability or normalcy. As a doctor in a manual wheelchair who values greatly my personal quality of life and professional contribution to patient care, I worry that these scoring systems inherently discount my own equitable access to care. Individualization of patients’ particular abilities in the context of these scales must occur alongside evidence-based, guideline-directed management via these scoring systems.
Conclusion: Future Orientation
Updated CSC guidelines have accounted for the unique considerations of disabled patients by effectively caveating their scoring algorithms, directing clinicians via disclaimers to uniquely consider their disabled patients in clinical judgement. This is a first step, but it is also one that erodes the value of algorithms, which generally obviate more deliberative thinking and individualization. For our patients who lack certain abilities, as CSC continue to be activated in several states, we have an opportunity to pursue more inherently equitable solutions before further suffering accrues.29 By way of example, adaptations to scoring systems that leverage QALYs for value-based drug pricing indices have been proposed by organizations like the Institute for Clinical and Economic Review, which proposed the Equal-Value-of Life-Years-Gained framework to inform QALY-based arbitration of drug pricing.30 This is not a perfect rubric but instead represents an attempt to balance consideration of drugs, as has been done with ventilators during the pandemic, as a scare and expensive resource while addressing the just concerns of advocacy groups in structural ableism.
Resource stewardship during a crisis should not discount those states of human life that are perceived to be less desirable, particularly if they are not experienced as less desirable but are experienced uniquely. Instead, we should consider equitably measuring our intervention to match a patient’s needs, as we would dose-adjust a medication for renal function or consider minimally invasive procedures for multimorbid patients. COVID-19 has reflected our profession’s ethical adaptation during crisis as resources have become scarce; there is no better time to define solutions for health equity. We should now be concerned equally by the influence our personal biases have on our clinical practice and by the way in which these crisis standards will influence patients’ perception of and trust in their care providers during periods of perceived plentiful resources in the future. Health care resources are always limited, allocated according to societal values; if we value health equity for people of all abilities, then we will consider these abilities equitably as we pursue new standards for health care delivery.
Corresponding author: Gregory D. Snyder, MD, MBA, 2014 Washington Street, Newton, MA 02462; [email protected].
Disclosures: None.
1. Emanuel EJ, Persad G, Upshur R, et al. Fair Allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055. doi:10.1056/NEJMsb2005114
2. Savulescu J, Persson I, Wilkinson D. Utilitarianism and the pandemic. Bioethics. 2020;34(6):620-632. doi:10.1111/bioe.12771
3. Mello MM, Persad G, White DB. Respecting disability rights - toward improved crisis standards of care. N Engl J Med. 2020;383(5):e26. doi: 10.1056/NEJMp2011997
4. The Commonwealth of Massachusetts Executive Office of Health and Human Services Department of Public Health. Crisis Standards of Care Planning Guidance for the COVID-19 Pandemic. April 7, 2020. https://d279m997dpfwgl.cloudfront.net/wp/2020/04/CSC_April-7_2020.pdf
5. Knowles H. Hospitals overwhelmed by covid are turning to ‘crisis standards of care.’ What does that mean? The Washington Post. September 21, 2021. Accessed January 24, 2022. https://www.washingtonpost.com/health/2021/09/22/crisis-standards-of-care/
6. Hick JL, Hanfling D, Wynia MK, Toner E. Crisis standards of care and COVID-19: What did we learn? How do we ensure equity? What should we do? NAM Perspect. 2021;2021:10.31478/202108e. doi:10.31478/202108e
7. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
8. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
9. Kukla E. My life is more ‘disposable’ during this pandemic. The New York Times. March 19, 2020. Accessed January 24, 2022. https://www.nytimes.com/2020/03/19/opinion/coronavirus-disabled-health-care.html
10. CPR and Coalition Partners Secure Important Changes in Massachusetts’ Crisis Standards of Care. Center for Public Representation. December 1, 2020. Accessed January 24, 2022. https://www.centerforpublicrep.org/news/cpr-and-coalition-partners-secure-important-changes-in-massachusetts-crisis-standards-of-care/
11. Johnson HM. Unspeakable conversations. The New York Times. February 16, 2003. Accessed January 24, 2022. https://www.nytimes.com/2003/02/16/magazine/unspeakable-conversations.html
12. Gerhart KA, Koziol-McLain J, Lowenstein SR, Whiteneck GG. Quality of life following spinal cord injury: knowledge and attitudes of emergency care providers. Ann Emerg Med. 1994;23(4):807-812. doi:10.1016/s0196-0644(94)70318-3
13. Iezzoni LI, Rao SR, Ressalam J, et al. Physicians’ perceptions of people with disability and their health care. Health Aff (Millwood). 2021;40(2):297-306. doi:10.1377/hlthaff.2020.01452
14. Smith DL. Disparities in patient-physician communication for persons with a disability from the 2006 Medical Expenditure Panel Survey (MEPS). Disabil Health J. 2009;2(4):206-215. doi:10.1016/j.dhjo.2009.06.002
15. Stillman MD, Ankam N, Mallow M, Capron M, Williams S. A survey of internal and family medicine residents: Assessment of disability-specific education and knowledge. Disabil Health J. 2021;14(2):101011. doi:10.1016/j.dhjo.2020.101011
16. Seidel E, Crowe S. The state of disability awareness in American medical schools. Am J Phys Med Rehabil. 2017;96(9):673-676. doi:10.1097/PHM.0000000000000719
17. Okoro CA, Hollis ND, Cyrus AC, Griffin-Blake S. Prevalence of disabilities and health care access by disability status and type among adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(32):882-887. doi:10.15585/mmwr.mm6732a3
18. Peacock G, Iezzoni LI, Harkin TR. Health care for Americans with disabilities--25 years after the ADA. N Engl J Med. 2015;373(10):892-893. doi:10.1056/NEJMp1508854
19. DeLisa JA, Thomas P. Physicians with disabilities and the physician workforce: a need to reassess our policies. Am J Phys Med Rehabil. 2005;84(1):5-11. doi:10.1097/01.phm.0000153323.28396.de
20. Disability and Health. Healthy People 2020. Accessed January 24, 2022. https://www.healthypeople.gov/2020/topics-objectives/topic/disability-and-health
21. Lagu T, Hannon NS, Rothberg MB, et al. Access to subspecialty care for patients with mobility impairment: a survey. Ann Intern Med. 2013;158(6):441-446. doi: 10.7326/0003-4819-158-6-201303190-00003
22. McCarthy EP, Ngo LH, Roetzheim RG, et al. Disparities in breast cancer treatment and survival for women with disabilities. Ann Intern Med. 2006;145(9):637-645. doi: 10.7326/0003-4819-145-9-200611070-00005
23. Javaid A, Nakata V, Michael D. Diagnostic overshadowing in learning disability: think beyond the disability. Prog Neurol Psychiatry. 2019;23:8-10.
24. Iezzoni LI, Rao SR, Agaronnik ND, El-Jawahri A. Cross-sectional analysis of the associations between four common cancers and disability. J Natl Compr Canc Netw. 2020;18(8):1031-1044. doi:10.6004/jnccn.2020.7551
25. Sanders JS, Keller S, Aravamuthan BR. Caring for individuals with intellectual and developmental disabilities in the COVID-19 crisis. Neurol Clin Pract. 2021;11(2):e174-e178. doi:10.1212/CPJ.0000000000000886
26. Landes SD, Turk MA, Formica MK, McDonald KE, Stevens JD. COVID-19 outcomes among people with intellectual and developmental disability living in residential group homes in New York State. Disabil Health J. 2020;13(4):100969. doi:10.1016/j.dhjo.2020.100969
27. Gleason J, Ross W, Fossi A, Blonksy H, Tobias J, Stephens M. The devastating impact of Covid-19 on individuals with intellectual disabilities in the United States. NEJM Catalyst. 2021.doi.org/10.1056/CAT.21.0051
28. Nankervis K, Chan J. Applying the CRPD to people with intellectual and developmental disability with behaviors of concern during COVID-19. J Policy Pract Intellect Disabil. 2021:10.1111/jppi.12374. doi:10.1111/jppi.12374
29. Alaska Department of Health and Social Services, Division of Public Health, Rural and Community Health Systems. Patient care strategies for scarce resource situations. Version 1. August 2021. Accessed November 11, 2021, https://dhss.alaska.gov/dph/Epi/id/SiteAssets/Pages/HumanCoV/SOA_DHSS_CrisisStandardsOfCare.pdf
30. Cost-effectiveness, the QALY, and the evlyg. ICER. May 21, 2021. Accessed January 24, 2022. https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/
1. Emanuel EJ, Persad G, Upshur R, et al. Fair Allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055. doi:10.1056/NEJMsb2005114
2. Savulescu J, Persson I, Wilkinson D. Utilitarianism and the pandemic. Bioethics. 2020;34(6):620-632. doi:10.1111/bioe.12771
3. Mello MM, Persad G, White DB. Respecting disability rights - toward improved crisis standards of care. N Engl J Med. 2020;383(5):e26. doi: 10.1056/NEJMp2011997
4. The Commonwealth of Massachusetts Executive Office of Health and Human Services Department of Public Health. Crisis Standards of Care Planning Guidance for the COVID-19 Pandemic. April 7, 2020. https://d279m997dpfwgl.cloudfront.net/wp/2020/04/CSC_April-7_2020.pdf
5. Knowles H. Hospitals overwhelmed by covid are turning to ‘crisis standards of care.’ What does that mean? The Washington Post. September 21, 2021. Accessed January 24, 2022. https://www.washingtonpost.com/health/2021/09/22/crisis-standards-of-care/
6. Hick JL, Hanfling D, Wynia MK, Toner E. Crisis standards of care and COVID-19: What did we learn? How do we ensure equity? What should we do? NAM Perspect. 2021;2021:10.31478/202108e. doi:10.31478/202108e
7. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
8. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
9. Kukla E. My life is more ‘disposable’ during this pandemic. The New York Times. March 19, 2020. Accessed January 24, 2022. https://www.nytimes.com/2020/03/19/opinion/coronavirus-disabled-health-care.html
10. CPR and Coalition Partners Secure Important Changes in Massachusetts’ Crisis Standards of Care. Center for Public Representation. December 1, 2020. Accessed January 24, 2022. https://www.centerforpublicrep.org/news/cpr-and-coalition-partners-secure-important-changes-in-massachusetts-crisis-standards-of-care/
11. Johnson HM. Unspeakable conversations. The New York Times. February 16, 2003. Accessed January 24, 2022. https://www.nytimes.com/2003/02/16/magazine/unspeakable-conversations.html
12. Gerhart KA, Koziol-McLain J, Lowenstein SR, Whiteneck GG. Quality of life following spinal cord injury: knowledge and attitudes of emergency care providers. Ann Emerg Med. 1994;23(4):807-812. doi:10.1016/s0196-0644(94)70318-3
13. Iezzoni LI, Rao SR, Ressalam J, et al. Physicians’ perceptions of people with disability and their health care. Health Aff (Millwood). 2021;40(2):297-306. doi:10.1377/hlthaff.2020.01452
14. Smith DL. Disparities in patient-physician communication for persons with a disability from the 2006 Medical Expenditure Panel Survey (MEPS). Disabil Health J. 2009;2(4):206-215. doi:10.1016/j.dhjo.2009.06.002
15. Stillman MD, Ankam N, Mallow M, Capron M, Williams S. A survey of internal and family medicine residents: Assessment of disability-specific education and knowledge. Disabil Health J. 2021;14(2):101011. doi:10.1016/j.dhjo.2020.101011
16. Seidel E, Crowe S. The state of disability awareness in American medical schools. Am J Phys Med Rehabil. 2017;96(9):673-676. doi:10.1097/PHM.0000000000000719
17. Okoro CA, Hollis ND, Cyrus AC, Griffin-Blake S. Prevalence of disabilities and health care access by disability status and type among adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(32):882-887. doi:10.15585/mmwr.mm6732a3
18. Peacock G, Iezzoni LI, Harkin TR. Health care for Americans with disabilities--25 years after the ADA. N Engl J Med. 2015;373(10):892-893. doi:10.1056/NEJMp1508854
19. DeLisa JA, Thomas P. Physicians with disabilities and the physician workforce: a need to reassess our policies. Am J Phys Med Rehabil. 2005;84(1):5-11. doi:10.1097/01.phm.0000153323.28396.de
20. Disability and Health. Healthy People 2020. Accessed January 24, 2022. https://www.healthypeople.gov/2020/topics-objectives/topic/disability-and-health
21. Lagu T, Hannon NS, Rothberg MB, et al. Access to subspecialty care for patients with mobility impairment: a survey. Ann Intern Med. 2013;158(6):441-446. doi: 10.7326/0003-4819-158-6-201303190-00003
22. McCarthy EP, Ngo LH, Roetzheim RG, et al. Disparities in breast cancer treatment and survival for women with disabilities. Ann Intern Med. 2006;145(9):637-645. doi: 10.7326/0003-4819-145-9-200611070-00005
23. Javaid A, Nakata V, Michael D. Diagnostic overshadowing in learning disability: think beyond the disability. Prog Neurol Psychiatry. 2019;23:8-10.
24. Iezzoni LI, Rao SR, Agaronnik ND, El-Jawahri A. Cross-sectional analysis of the associations between four common cancers and disability. J Natl Compr Canc Netw. 2020;18(8):1031-1044. doi:10.6004/jnccn.2020.7551
25. Sanders JS, Keller S, Aravamuthan BR. Caring for individuals with intellectual and developmental disabilities in the COVID-19 crisis. Neurol Clin Pract. 2021;11(2):e174-e178. doi:10.1212/CPJ.0000000000000886
26. Landes SD, Turk MA, Formica MK, McDonald KE, Stevens JD. COVID-19 outcomes among people with intellectual and developmental disability living in residential group homes in New York State. Disabil Health J. 2020;13(4):100969. doi:10.1016/j.dhjo.2020.100969
27. Gleason J, Ross W, Fossi A, Blonksy H, Tobias J, Stephens M. The devastating impact of Covid-19 on individuals with intellectual disabilities in the United States. NEJM Catalyst. 2021.doi.org/10.1056/CAT.21.0051
28. Nankervis K, Chan J. Applying the CRPD to people with intellectual and developmental disability with behaviors of concern during COVID-19. J Policy Pract Intellect Disabil. 2021:10.1111/jppi.12374. doi:10.1111/jppi.12374
29. Alaska Department of Health and Social Services, Division of Public Health, Rural and Community Health Systems. Patient care strategies for scarce resource situations. Version 1. August 2021. Accessed November 11, 2021, https://dhss.alaska.gov/dph/Epi/id/SiteAssets/Pages/HumanCoV/SOA_DHSS_CrisisStandardsOfCare.pdf
30. Cost-effectiveness, the QALY, and the evlyg. ICER. May 21, 2021. Accessed January 24, 2022. https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/
Keeping an open mind about functional medicine
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
Considering the controversy surrounding functional medicine, you may be wondering why JFP published an article about it last month.1 David Gorski, MD, PhD, FACS, a vocal critic of functional medicine, commented: “Functional medicine. It sounds so … scientific and reasonable. It’s anything but. In fact, functional medicine combines the worst features of conventional medicine with a heapin’ helpin’ of quackery.”2 On its website, however, The Institute for Functional Medicine claims that “functional medicine determines how and why illness occurs and restores health by addressing the root causes of disease for each individual.”3
I suspect the truth lies somewhere in between.
Because functional medicine has gained a certain degree of popularity, I felt it was important for family physicians and other primary care clinicians to know enough about this alternative healing method to discuss it with patients who express interest.
In their review article in JFP, Orlando and colleagues tell us there are 7 defining characteristics of functional medicine.1 It is patient centered rather than disease centered, uses a “systems biology” approach, considers the dynamic balance of gene-environment interactions, is personalized based on biochemical individuality, promotes organ reserve and sustained health span, sees health as a positive vitality (not merely the absence of disease), and focuses on function rather than pathology.
Most of these statements about functional medicine apply to traditional family medicine. The clinical approach stressing lifestyle changes is mainstream, not unique. The focus on digestion and the microbiome as an important determinant of health is based on interesting basic science studies and associations noted between certain microbiome profiles and diseases.
But association is not causation. So far there is scant evidence that changing the microbiome results in better health, although some preliminary case series have generated intriguing hypotheses. And there is evidence that probiotics improve some symptoms. Ongoing research into the microbiome and health will, no doubt, be illuminating. We have much to learn.
What does seem unique, but suspect, about functional medicine is its focus on biochemical testing of unproven value and the prescribing of diets and supplements based on the test results. There are no sound scientific studies showing the benefit of this approach.
I suggest you read Orlando et al’s article. Functional medicine is an interesting, mostly unproven, approach to patient care. But I will keep an open mind until we see better research that either does—or doesn’t—support the validity of its practices.
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
1. Orlando FA, Chang KL, Estores IM. Functional medicine: focusing on imbalances in core metabolic processes. J Fam Pract. 2021;70:482-488,498.
2. Gorski D. Functional medicine: the ultimate misnomer in the world of integrative medicine. Science-Based Medicine. April 11, 2016. Accessed January 4, 2022. https://sciencebasedmedicine.org/functional-medicine-the-ultimate-misnomer-in-the-world-of-integrative-medicine/
3. The Institute for Functional Medicine. Accessed January 4, 2022. www.ifm.org
Feds’ website for free at-home COVID tests launches day early
The Biden administration’s new no-cost, at-home testing program launched Jan. 18, a day ahead of schedule.
The administration said 500 million tests are available to be delivered to homes across the country. This accounts for half of the president’s recent pledge to purchase 1 billion free at-home COVID-19 tests to distribute to the American public.
On a Jan. 14 call with reporters, senior White House officials offered some details about the new program.
Here’s what we know so far.
How do I order my free tests?
Americans can visit COVIDtests.gov to order their rapid at-home tests. You can also order directly from the U.S. Postal Service website. After you order, you’ll receive a confirmation email that promises to send tracking information once your order ships.
What information do I need to order the tests?
You only need your name and home mailing address.
There is also an option to provide your email address to get updates on the status of your order.
What if someone needs help ordering the tests?
There will be a free call-in line for people needing more help, including those having trouble accessing the internet, according to White House officials.
What tests will be available?
There are nine at-home tests available through FDA emergency use authorization. According to the Frequently Asked Questions section of COVIDtests.gov, "You will not be able to choose the brand you order as part of this program.”
How long will it take to get the tests once I order them?
Tests are expected to ship 7 to 12 days after you order them.
But White House officials say that the time frame will likely shorten as the program gains steam.
How many can I order?
There’s a limit of four tests per residential mailing address.
For larger families, White House officials suggest trying other free testing options, like visiting COVID-19 testing sites or your local health center.
Is this a one-time opportunity?
The White House doesn’t say, but officials did mention that if you run out of your four free tests, there are many other ways to access free at-home tests, such as COVID-19 testing sites, pharmacies, and community health centers.
The free tests available through COVIDtests.gov are in addition to an estimated 375 million at-home rapid tests on the market in the U.S. this month.
When should people use a rapid at-home test?
The CDC and experts with other public health groups agree that Americans should consider using at-home rapid tests in the following situations:
- If they begin to have symptoms consistent with COVID-19;
- At least 5 days after close contact with someone who has COVID;
- If someone is indoors with a group of people who are at risk of severe disease or are unvaccinated.
Are at-home rapid tests accurate?
The U.S. Department of Health and Human Services and other federal officials confirmed through studies that all tests distributed through this program can detect the Omicron variant. These agencies also confirmed that their performance is consistent with the FDA’s emergency use authorization.
Is the website designed to handle high demand?
After the original website to sign up for health insurance under the Affordable Care Act crashed repeatedly at launch, the government says it has prepared for high demand for ordering at-home rapid tests.
The U.S. Digital Service (USDS), an organization founded after Healthcare.gov, has partnered with the Postal Service to plan for the launch.
The Postal Service has expanded its staffing, similar to what’s done during the holidays.
All orders in the continental United States will be shipped through first-class mail, with shipments to Alaska, Hawaii, U.S. territories, and military and overseas addresses sent through priority mail.
A version of this article first appeared on WebMD.com.
The Biden administration’s new no-cost, at-home testing program launched Jan. 18, a day ahead of schedule.
The administration said 500 million tests are available to be delivered to homes across the country. This accounts for half of the president’s recent pledge to purchase 1 billion free at-home COVID-19 tests to distribute to the American public.
On a Jan. 14 call with reporters, senior White House officials offered some details about the new program.
Here’s what we know so far.
How do I order my free tests?
Americans can visit COVIDtests.gov to order their rapid at-home tests. You can also order directly from the U.S. Postal Service website. After you order, you’ll receive a confirmation email that promises to send tracking information once your order ships.
What information do I need to order the tests?
You only need your name and home mailing address.
There is also an option to provide your email address to get updates on the status of your order.
What if someone needs help ordering the tests?
There will be a free call-in line for people needing more help, including those having trouble accessing the internet, according to White House officials.
What tests will be available?
There are nine at-home tests available through FDA emergency use authorization. According to the Frequently Asked Questions section of COVIDtests.gov, "You will not be able to choose the brand you order as part of this program.”
How long will it take to get the tests once I order them?
Tests are expected to ship 7 to 12 days after you order them.
But White House officials say that the time frame will likely shorten as the program gains steam.
How many can I order?
There’s a limit of four tests per residential mailing address.
For larger families, White House officials suggest trying other free testing options, like visiting COVID-19 testing sites or your local health center.
Is this a one-time opportunity?
The White House doesn’t say, but officials did mention that if you run out of your four free tests, there are many other ways to access free at-home tests, such as COVID-19 testing sites, pharmacies, and community health centers.
The free tests available through COVIDtests.gov are in addition to an estimated 375 million at-home rapid tests on the market in the U.S. this month.
When should people use a rapid at-home test?
The CDC and experts with other public health groups agree that Americans should consider using at-home rapid tests in the following situations:
- If they begin to have symptoms consistent with COVID-19;
- At least 5 days after close contact with someone who has COVID;
- If someone is indoors with a group of people who are at risk of severe disease or are unvaccinated.
Are at-home rapid tests accurate?
The U.S. Department of Health and Human Services and other federal officials confirmed through studies that all tests distributed through this program can detect the Omicron variant. These agencies also confirmed that their performance is consistent with the FDA’s emergency use authorization.
Is the website designed to handle high demand?
After the original website to sign up for health insurance under the Affordable Care Act crashed repeatedly at launch, the government says it has prepared for high demand for ordering at-home rapid tests.
The U.S. Digital Service (USDS), an organization founded after Healthcare.gov, has partnered with the Postal Service to plan for the launch.
The Postal Service has expanded its staffing, similar to what’s done during the holidays.
All orders in the continental United States will be shipped through first-class mail, with shipments to Alaska, Hawaii, U.S. territories, and military and overseas addresses sent through priority mail.
A version of this article first appeared on WebMD.com.
The Biden administration’s new no-cost, at-home testing program launched Jan. 18, a day ahead of schedule.
The administration said 500 million tests are available to be delivered to homes across the country. This accounts for half of the president’s recent pledge to purchase 1 billion free at-home COVID-19 tests to distribute to the American public.
On a Jan. 14 call with reporters, senior White House officials offered some details about the new program.
Here’s what we know so far.
How do I order my free tests?
Americans can visit COVIDtests.gov to order their rapid at-home tests. You can also order directly from the U.S. Postal Service website. After you order, you’ll receive a confirmation email that promises to send tracking information once your order ships.
What information do I need to order the tests?
You only need your name and home mailing address.
There is also an option to provide your email address to get updates on the status of your order.
What if someone needs help ordering the tests?
There will be a free call-in line for people needing more help, including those having trouble accessing the internet, according to White House officials.
What tests will be available?
There are nine at-home tests available through FDA emergency use authorization. According to the Frequently Asked Questions section of COVIDtests.gov, "You will not be able to choose the brand you order as part of this program.”
How long will it take to get the tests once I order them?
Tests are expected to ship 7 to 12 days after you order them.
But White House officials say that the time frame will likely shorten as the program gains steam.
How many can I order?
There’s a limit of four tests per residential mailing address.
For larger families, White House officials suggest trying other free testing options, like visiting COVID-19 testing sites or your local health center.
Is this a one-time opportunity?
The White House doesn’t say, but officials did mention that if you run out of your four free tests, there are many other ways to access free at-home tests, such as COVID-19 testing sites, pharmacies, and community health centers.
The free tests available through COVIDtests.gov are in addition to an estimated 375 million at-home rapid tests on the market in the U.S. this month.
When should people use a rapid at-home test?
The CDC and experts with other public health groups agree that Americans should consider using at-home rapid tests in the following situations:
- If they begin to have symptoms consistent with COVID-19;
- At least 5 days after close contact with someone who has COVID;
- If someone is indoors with a group of people who are at risk of severe disease or are unvaccinated.
Are at-home rapid tests accurate?
The U.S. Department of Health and Human Services and other federal officials confirmed through studies that all tests distributed through this program can detect the Omicron variant. These agencies also confirmed that their performance is consistent with the FDA’s emergency use authorization.
Is the website designed to handle high demand?
After the original website to sign up for health insurance under the Affordable Care Act crashed repeatedly at launch, the government says it has prepared for high demand for ordering at-home rapid tests.
The U.S. Digital Service (USDS), an organization founded after Healthcare.gov, has partnered with the Postal Service to plan for the launch.
The Postal Service has expanded its staffing, similar to what’s done during the holidays.
All orders in the continental United States will be shipped through first-class mail, with shipments to Alaska, Hawaii, U.S. territories, and military and overseas addresses sent through priority mail.
A version of this article first appeared on WebMD.com.


