User login
Can a Risk Score Predict Kidney Injury After Cisplatin?
Cisplatin is a preferred treatment for a wide range of cancers, including breast, head and neck, lung, ovary, and more. However, its side effects — particularly nephrotoxicity — can be severe. Kidney injury on cisplatin is associated with higher mortality and can jeopardize a patient’s eligibility for other therapies.
Now, in a large study using data from six US cancer centers, researchers have developed a risk algorithm to predict acute kidney injury (AKI) after cisplatin administration.
A risk prediction calculator based on the algorithm is available online for patients and providers to determine an individual patient›s risk for kidney injury from cisplatin using readily available clinical data.
Other risk scores and risk prediction models have been developed to help clinicians assess in advance whether a patient might develop AKI after receiving cisplatin, so that more careful monitoring, dose adjustments, or an alternative treatment, if available, might be considered.
However, previous models were limited by factors such as small sample sizes, lack of external validation, older data, and liberal definitions of AKI, said Shruti Gupta, MD, MPH, director of onco-nephrology at Brigham and Women’s Hospital (BWH) and Dana-Farber Cancer Institute, and David E. Leaf, MD, MMSc, director of clinical and translational research in AKI, Division of Renal Medicine, BWH, Boston.
Dr. Gupta and Dr. Leaf believe their risk score for predicting severe AKI after intravenous (IV) cisplatin, published online in The BMJ, is “more accurate and generalizable than prior models for several reasons,” they told this news organization in a joint email.
“First, we externally validated our findings across cancer centers other than the one where it was developed,” they said. “Second, we focused on moderate to severe kidney injury, the most clinically relevant form of kidney damage, whereas prior models examined more mild forms of kidney injury. Third, we collected data on nearly 25,000 patients receiving their first dose of IV cisplatin, which is larger than all previous studies combined.”
‘Herculean Effort’
“We conceived of this study back in 2018, contacted collaborators at each participating cancer center, and had numerous meetings to try to gather granular data on patients treated with their first dose of intravenous (IV) cisplatin,” Dr. Gupta and Dr. Leaf explained. They also incorporated patient feedback from focus groups and surveys.
“This was truly a Herculean effort that involved physicians, programmers, research coordinators, and patients,” they said.
The multicenter study included 24,717 patients — 11,766 in the derivation cohort and 12,951 in the validation cohort. Overall, the median age was about 60 years, about 58% were men, and about 78% were White.
The primary outcome was cisplatin-induced AKI (CP-AKI), defined as a twofold or greater increase in serum creatinine or kidney replacement therapy within 14 days of a first dose of IV cisplatin.
Their simple risk score consisting of nine covariates — age, hypertension, type 2 diabetes, hemoglobin level, white blood cell count, platelet count, serum albumin level, serum magnesium level, and cisplatin dose — predicted a higher risk for CP-AKI in both cohorts.
Notably, adding serum creatinine to the model did not change the area under the curve, and therefore, serum creatinine, though also an independent risk factor for CP-AKI, was not included in the score.
Patients in the highest risk category had 24-fold higher odds of CP-AKI in the derivation cohort and close to 18-fold higher odds in the validation cohort than those in the lowest risk category.
The primary model had a C statistic of 0.75 (95% CI, 0.73-0.76) and showed better discrimination for CP-AKI than previously published models, for which the C statistics ranged from 0.60 to 0.68. The first author of a paper on an earlier model, Shveta Motwani, MD, MMSc, of BWH and Dana-Farber Cancer Institute in Boston, is also a coauthor of the new study.
Greater severity of CP-AKI was associated with shorter 90-day survival (adjusted hazard ratio, 4.63; 95% CI, 3.56-6.02) for stage III CP-AKI vs no CP-AKI.
‘Definitive Work’
Joel M. Topf, MD, a nephrologist with expertise in chronic kidney disease in Detroit, who wasn’t involved in the development of the risk score, called the study “a definitive work on an important concept in oncology and nephrology.”
“While this is not the first attempt to devise a risk score, it is by far the biggest,” he told this news organization. Furthermore, the authors “used a diverse population, recruiting patients with a variety of cancers (previous attempts had often used a homogenous diagnosis, putting into question how generalizable the results were) from six different cancer centers.”
In addition, he said, “The authors did not restrict patients with chronic kidney disease or other significant comorbidities and used the geographic diversity to produce a cohort that has an age, gender, racial, and ethnic distribution, which is more representative of the US than previous, single-center attempts to risk score patients.”
An earlier model used the Kidney Disease: Improving Global Outcomes (KDIGO) consensus definition of AKI of an increase in serum creatinine of 0.3 mg/dL, he noted. “While a sensitive definition of AKI, it captures mild, hemodynamic increases in creatinine of questionable significance,” he said.
By contrast, the new score uses KDIGO stage II and above to define AKI. “This is a better choice, as we do not want to dissuade patients and doctors from choosing chemotherapy due to a fear of insignificant kidney damage,” he said.
All that said, Dr. Topf noted that neither the current score nor the earlier model included serum creatinine. “This is curious to me and may represent the small number of patients with representative elevated creatinine in the derivation cohort (only 1.3% with an estimated glomerular filtration rate [eGFR] < 45).”
“Since the cohort is made up of people who received cis-platinum, the low prevalence of eGFRs < 45 may be due to physicians steering away from cis-platinum in this group,” he suggested. “It would be unfortunate if this risk score gave an unintentional ‘green light’ to these patients, exposing them to predictable harm.”
‘Certainly Useful’
Anushree Shirali, MD, an associate professor in the Section of Nephrology and consulting physician, Yale Onco-Nephrology, Yale School of Medicine, in New Haven, Connecticut, said that having a prediction score for which patients are more likely to develop AKI after a single dose of cisplatin would be helpful for oncologists, as well as nephrologists.
As a nephrologist, Dr. Shirali mostly sees patients who already have AKI, she told this news organization. But there are circumstances in which the tool could still be helpful.
“Let’s say someone has abnormal kidney function at baseline — ie, creatinine is higher than the normal range — and they were on dialysis 5 years ago for something else, and now, they have cancer and may be given cisplatin. They worry about their chances of getting AKI and needing dialysis again,” she said. “That’s just one scenario in which I might be asked to answer that question and the tool would certainly be useful.”
Other scenarios could include someone who has just one kidney because they donated a kidney for transplant years ago, and now, they have a malignancy and wonder what their actual risk is of getting kidney issues on cisplatin.
Oncologists could use the tool to determine whether a patient should be treated with cisplatin, or if they’re at high risk, whether an alternative that’s not nephrotoxic might be used. By contrast, “if somebody’s low risk and an oncologist thinks cisplatin is the best agent they have, then they might want to go ahead and use it,” Dr. Shirali said.
Future research could take into consideration that CP-AKI is dose dependent, she suggested, because a prediction score that included the number of cisplatin doses could be even more helpful to determine risk. And, even though the derivation and validation cohorts for the new tool are representative of the US population, additional research should also include more racial/ethnic diversity, she said.
Dr. Gupta and Dr. Leaf hope their tool “will be utilized immediately by patients and providers to help predict an individual’s risk of cisplatin-associated kidney damage. It is easy to use, available for free online, and incorporates readily available clinical variables.”
If a patient is at high risk, the clinical team can consider preventive measures such as administering more IV fluids before receiving cisplatin or monitoring kidney function more closely afterward, they suggested.
Dr. Gupta reported research support from the National Institutes of Health (NIH) and the National Institute of Diabetes and Digestive and Kidney Diseases. She also reported research funding from BTG International, GE HealthCare, and AstraZeneca outside the submitted work. She is a member of GlaxoSmithKline’s Global Anemia Council, a consultant for Secretome and Proletariat Therapeutics, and founder and president emeritus of the American Society of Onconephrology (unpaid). Dr. Leaf is supported by NIH grants, reported research support from BioPorto, BTG International, and Metro International Biotech, and has served as a consultant. Dr. Topf reported an ownership stake in a few DaVita-run dialysis clinics. He also runs a vascular access center and has participated in advisory boards with Cara Therapeutics, Vifor, Astra Zeneca, Bayer, Renibus Therapeutics, Travere Therapeutics, and GlaxoSmithKline. He is president of NephJC, a nonprofit educational organization with no industry support. Dr. Shirali declared no competing interests.
A version of this article appeared on Medscape.com.
Cisplatin is a preferred treatment for a wide range of cancers, including breast, head and neck, lung, ovary, and more. However, its side effects — particularly nephrotoxicity — can be severe. Kidney injury on cisplatin is associated with higher mortality and can jeopardize a patient’s eligibility for other therapies.
Now, in a large study using data from six US cancer centers, researchers have developed a risk algorithm to predict acute kidney injury (AKI) after cisplatin administration.
A risk prediction calculator based on the algorithm is available online for patients and providers to determine an individual patient›s risk for kidney injury from cisplatin using readily available clinical data.
Other risk scores and risk prediction models have been developed to help clinicians assess in advance whether a patient might develop AKI after receiving cisplatin, so that more careful monitoring, dose adjustments, or an alternative treatment, if available, might be considered.
However, previous models were limited by factors such as small sample sizes, lack of external validation, older data, and liberal definitions of AKI, said Shruti Gupta, MD, MPH, director of onco-nephrology at Brigham and Women’s Hospital (BWH) and Dana-Farber Cancer Institute, and David E. Leaf, MD, MMSc, director of clinical and translational research in AKI, Division of Renal Medicine, BWH, Boston.
Dr. Gupta and Dr. Leaf believe their risk score for predicting severe AKI after intravenous (IV) cisplatin, published online in The BMJ, is “more accurate and generalizable than prior models for several reasons,” they told this news organization in a joint email.
“First, we externally validated our findings across cancer centers other than the one where it was developed,” they said. “Second, we focused on moderate to severe kidney injury, the most clinically relevant form of kidney damage, whereas prior models examined more mild forms of kidney injury. Third, we collected data on nearly 25,000 patients receiving their first dose of IV cisplatin, which is larger than all previous studies combined.”
‘Herculean Effort’
“We conceived of this study back in 2018, contacted collaborators at each participating cancer center, and had numerous meetings to try to gather granular data on patients treated with their first dose of intravenous (IV) cisplatin,” Dr. Gupta and Dr. Leaf explained. They also incorporated patient feedback from focus groups and surveys.
“This was truly a Herculean effort that involved physicians, programmers, research coordinators, and patients,” they said.
The multicenter study included 24,717 patients — 11,766 in the derivation cohort and 12,951 in the validation cohort. Overall, the median age was about 60 years, about 58% were men, and about 78% were White.
The primary outcome was cisplatin-induced AKI (CP-AKI), defined as a twofold or greater increase in serum creatinine or kidney replacement therapy within 14 days of a first dose of IV cisplatin.
Their simple risk score consisting of nine covariates — age, hypertension, type 2 diabetes, hemoglobin level, white blood cell count, platelet count, serum albumin level, serum magnesium level, and cisplatin dose — predicted a higher risk for CP-AKI in both cohorts.
Notably, adding serum creatinine to the model did not change the area under the curve, and therefore, serum creatinine, though also an independent risk factor for CP-AKI, was not included in the score.
Patients in the highest risk category had 24-fold higher odds of CP-AKI in the derivation cohort and close to 18-fold higher odds in the validation cohort than those in the lowest risk category.
The primary model had a C statistic of 0.75 (95% CI, 0.73-0.76) and showed better discrimination for CP-AKI than previously published models, for which the C statistics ranged from 0.60 to 0.68. The first author of a paper on an earlier model, Shveta Motwani, MD, MMSc, of BWH and Dana-Farber Cancer Institute in Boston, is also a coauthor of the new study.
Greater severity of CP-AKI was associated with shorter 90-day survival (adjusted hazard ratio, 4.63; 95% CI, 3.56-6.02) for stage III CP-AKI vs no CP-AKI.
‘Definitive Work’
Joel M. Topf, MD, a nephrologist with expertise in chronic kidney disease in Detroit, who wasn’t involved in the development of the risk score, called the study “a definitive work on an important concept in oncology and nephrology.”
“While this is not the first attempt to devise a risk score, it is by far the biggest,” he told this news organization. Furthermore, the authors “used a diverse population, recruiting patients with a variety of cancers (previous attempts had often used a homogenous diagnosis, putting into question how generalizable the results were) from six different cancer centers.”
In addition, he said, “The authors did not restrict patients with chronic kidney disease or other significant comorbidities and used the geographic diversity to produce a cohort that has an age, gender, racial, and ethnic distribution, which is more representative of the US than previous, single-center attempts to risk score patients.”
An earlier model used the Kidney Disease: Improving Global Outcomes (KDIGO) consensus definition of AKI of an increase in serum creatinine of 0.3 mg/dL, he noted. “While a sensitive definition of AKI, it captures mild, hemodynamic increases in creatinine of questionable significance,” he said.
By contrast, the new score uses KDIGO stage II and above to define AKI. “This is a better choice, as we do not want to dissuade patients and doctors from choosing chemotherapy due to a fear of insignificant kidney damage,” he said.
All that said, Dr. Topf noted that neither the current score nor the earlier model included serum creatinine. “This is curious to me and may represent the small number of patients with representative elevated creatinine in the derivation cohort (only 1.3% with an estimated glomerular filtration rate [eGFR] < 45).”
“Since the cohort is made up of people who received cis-platinum, the low prevalence of eGFRs < 45 may be due to physicians steering away from cis-platinum in this group,” he suggested. “It would be unfortunate if this risk score gave an unintentional ‘green light’ to these patients, exposing them to predictable harm.”
‘Certainly Useful’
Anushree Shirali, MD, an associate professor in the Section of Nephrology and consulting physician, Yale Onco-Nephrology, Yale School of Medicine, in New Haven, Connecticut, said that having a prediction score for which patients are more likely to develop AKI after a single dose of cisplatin would be helpful for oncologists, as well as nephrologists.
As a nephrologist, Dr. Shirali mostly sees patients who already have AKI, she told this news organization. But there are circumstances in which the tool could still be helpful.
“Let’s say someone has abnormal kidney function at baseline — ie, creatinine is higher than the normal range — and they were on dialysis 5 years ago for something else, and now, they have cancer and may be given cisplatin. They worry about their chances of getting AKI and needing dialysis again,” she said. “That’s just one scenario in which I might be asked to answer that question and the tool would certainly be useful.”
Other scenarios could include someone who has just one kidney because they donated a kidney for transplant years ago, and now, they have a malignancy and wonder what their actual risk is of getting kidney issues on cisplatin.
Oncologists could use the tool to determine whether a patient should be treated with cisplatin, or if they’re at high risk, whether an alternative that’s not nephrotoxic might be used. By contrast, “if somebody’s low risk and an oncologist thinks cisplatin is the best agent they have, then they might want to go ahead and use it,” Dr. Shirali said.
Future research could take into consideration that CP-AKI is dose dependent, she suggested, because a prediction score that included the number of cisplatin doses could be even more helpful to determine risk. And, even though the derivation and validation cohorts for the new tool are representative of the US population, additional research should also include more racial/ethnic diversity, she said.
Dr. Gupta and Dr. Leaf hope their tool “will be utilized immediately by patients and providers to help predict an individual’s risk of cisplatin-associated kidney damage. It is easy to use, available for free online, and incorporates readily available clinical variables.”
If a patient is at high risk, the clinical team can consider preventive measures such as administering more IV fluids before receiving cisplatin or monitoring kidney function more closely afterward, they suggested.
Dr. Gupta reported research support from the National Institutes of Health (NIH) and the National Institute of Diabetes and Digestive and Kidney Diseases. She also reported research funding from BTG International, GE HealthCare, and AstraZeneca outside the submitted work. She is a member of GlaxoSmithKline’s Global Anemia Council, a consultant for Secretome and Proletariat Therapeutics, and founder and president emeritus of the American Society of Onconephrology (unpaid). Dr. Leaf is supported by NIH grants, reported research support from BioPorto, BTG International, and Metro International Biotech, and has served as a consultant. Dr. Topf reported an ownership stake in a few DaVita-run dialysis clinics. He also runs a vascular access center and has participated in advisory boards with Cara Therapeutics, Vifor, Astra Zeneca, Bayer, Renibus Therapeutics, Travere Therapeutics, and GlaxoSmithKline. He is president of NephJC, a nonprofit educational organization with no industry support. Dr. Shirali declared no competing interests.
A version of this article appeared on Medscape.com.
Cisplatin is a preferred treatment for a wide range of cancers, including breast, head and neck, lung, ovary, and more. However, its side effects — particularly nephrotoxicity — can be severe. Kidney injury on cisplatin is associated with higher mortality and can jeopardize a patient’s eligibility for other therapies.
Now, in a large study using data from six US cancer centers, researchers have developed a risk algorithm to predict acute kidney injury (AKI) after cisplatin administration.
A risk prediction calculator based on the algorithm is available online for patients and providers to determine an individual patient›s risk for kidney injury from cisplatin using readily available clinical data.
Other risk scores and risk prediction models have been developed to help clinicians assess in advance whether a patient might develop AKI after receiving cisplatin, so that more careful monitoring, dose adjustments, or an alternative treatment, if available, might be considered.
However, previous models were limited by factors such as small sample sizes, lack of external validation, older data, and liberal definitions of AKI, said Shruti Gupta, MD, MPH, director of onco-nephrology at Brigham and Women’s Hospital (BWH) and Dana-Farber Cancer Institute, and David E. Leaf, MD, MMSc, director of clinical and translational research in AKI, Division of Renal Medicine, BWH, Boston.
Dr. Gupta and Dr. Leaf believe their risk score for predicting severe AKI after intravenous (IV) cisplatin, published online in The BMJ, is “more accurate and generalizable than prior models for several reasons,” they told this news organization in a joint email.
“First, we externally validated our findings across cancer centers other than the one where it was developed,” they said. “Second, we focused on moderate to severe kidney injury, the most clinically relevant form of kidney damage, whereas prior models examined more mild forms of kidney injury. Third, we collected data on nearly 25,000 patients receiving their first dose of IV cisplatin, which is larger than all previous studies combined.”
‘Herculean Effort’
“We conceived of this study back in 2018, contacted collaborators at each participating cancer center, and had numerous meetings to try to gather granular data on patients treated with their first dose of intravenous (IV) cisplatin,” Dr. Gupta and Dr. Leaf explained. They also incorporated patient feedback from focus groups and surveys.
“This was truly a Herculean effort that involved physicians, programmers, research coordinators, and patients,” they said.
The multicenter study included 24,717 patients — 11,766 in the derivation cohort and 12,951 in the validation cohort. Overall, the median age was about 60 years, about 58% were men, and about 78% were White.
The primary outcome was cisplatin-induced AKI (CP-AKI), defined as a twofold or greater increase in serum creatinine or kidney replacement therapy within 14 days of a first dose of IV cisplatin.
Their simple risk score consisting of nine covariates — age, hypertension, type 2 diabetes, hemoglobin level, white blood cell count, platelet count, serum albumin level, serum magnesium level, and cisplatin dose — predicted a higher risk for CP-AKI in both cohorts.
Notably, adding serum creatinine to the model did not change the area under the curve, and therefore, serum creatinine, though also an independent risk factor for CP-AKI, was not included in the score.
Patients in the highest risk category had 24-fold higher odds of CP-AKI in the derivation cohort and close to 18-fold higher odds in the validation cohort than those in the lowest risk category.
The primary model had a C statistic of 0.75 (95% CI, 0.73-0.76) and showed better discrimination for CP-AKI than previously published models, for which the C statistics ranged from 0.60 to 0.68. The first author of a paper on an earlier model, Shveta Motwani, MD, MMSc, of BWH and Dana-Farber Cancer Institute in Boston, is also a coauthor of the new study.
Greater severity of CP-AKI was associated with shorter 90-day survival (adjusted hazard ratio, 4.63; 95% CI, 3.56-6.02) for stage III CP-AKI vs no CP-AKI.
‘Definitive Work’
Joel M. Topf, MD, a nephrologist with expertise in chronic kidney disease in Detroit, who wasn’t involved in the development of the risk score, called the study “a definitive work on an important concept in oncology and nephrology.”
“While this is not the first attempt to devise a risk score, it is by far the biggest,” he told this news organization. Furthermore, the authors “used a diverse population, recruiting patients with a variety of cancers (previous attempts had often used a homogenous diagnosis, putting into question how generalizable the results were) from six different cancer centers.”
In addition, he said, “The authors did not restrict patients with chronic kidney disease or other significant comorbidities and used the geographic diversity to produce a cohort that has an age, gender, racial, and ethnic distribution, which is more representative of the US than previous, single-center attempts to risk score patients.”
An earlier model used the Kidney Disease: Improving Global Outcomes (KDIGO) consensus definition of AKI of an increase in serum creatinine of 0.3 mg/dL, he noted. “While a sensitive definition of AKI, it captures mild, hemodynamic increases in creatinine of questionable significance,” he said.
By contrast, the new score uses KDIGO stage II and above to define AKI. “This is a better choice, as we do not want to dissuade patients and doctors from choosing chemotherapy due to a fear of insignificant kidney damage,” he said.
All that said, Dr. Topf noted that neither the current score nor the earlier model included serum creatinine. “This is curious to me and may represent the small number of patients with representative elevated creatinine in the derivation cohort (only 1.3% with an estimated glomerular filtration rate [eGFR] < 45).”
“Since the cohort is made up of people who received cis-platinum, the low prevalence of eGFRs < 45 may be due to physicians steering away from cis-platinum in this group,” he suggested. “It would be unfortunate if this risk score gave an unintentional ‘green light’ to these patients, exposing them to predictable harm.”
‘Certainly Useful’
Anushree Shirali, MD, an associate professor in the Section of Nephrology and consulting physician, Yale Onco-Nephrology, Yale School of Medicine, in New Haven, Connecticut, said that having a prediction score for which patients are more likely to develop AKI after a single dose of cisplatin would be helpful for oncologists, as well as nephrologists.
As a nephrologist, Dr. Shirali mostly sees patients who already have AKI, she told this news organization. But there are circumstances in which the tool could still be helpful.
“Let’s say someone has abnormal kidney function at baseline — ie, creatinine is higher than the normal range — and they were on dialysis 5 years ago for something else, and now, they have cancer and may be given cisplatin. They worry about their chances of getting AKI and needing dialysis again,” she said. “That’s just one scenario in which I might be asked to answer that question and the tool would certainly be useful.”
Other scenarios could include someone who has just one kidney because they donated a kidney for transplant years ago, and now, they have a malignancy and wonder what their actual risk is of getting kidney issues on cisplatin.
Oncologists could use the tool to determine whether a patient should be treated with cisplatin, or if they’re at high risk, whether an alternative that’s not nephrotoxic might be used. By contrast, “if somebody’s low risk and an oncologist thinks cisplatin is the best agent they have, then they might want to go ahead and use it,” Dr. Shirali said.
Future research could take into consideration that CP-AKI is dose dependent, she suggested, because a prediction score that included the number of cisplatin doses could be even more helpful to determine risk. And, even though the derivation and validation cohorts for the new tool are representative of the US population, additional research should also include more racial/ethnic diversity, she said.
Dr. Gupta and Dr. Leaf hope their tool “will be utilized immediately by patients and providers to help predict an individual’s risk of cisplatin-associated kidney damage. It is easy to use, available for free online, and incorporates readily available clinical variables.”
If a patient is at high risk, the clinical team can consider preventive measures such as administering more IV fluids before receiving cisplatin or monitoring kidney function more closely afterward, they suggested.
Dr. Gupta reported research support from the National Institutes of Health (NIH) and the National Institute of Diabetes and Digestive and Kidney Diseases. She also reported research funding from BTG International, GE HealthCare, and AstraZeneca outside the submitted work. She is a member of GlaxoSmithKline’s Global Anemia Council, a consultant for Secretome and Proletariat Therapeutics, and founder and president emeritus of the American Society of Onconephrology (unpaid). Dr. Leaf is supported by NIH grants, reported research support from BioPorto, BTG International, and Metro International Biotech, and has served as a consultant. Dr. Topf reported an ownership stake in a few DaVita-run dialysis clinics. He also runs a vascular access center and has participated in advisory boards with Cara Therapeutics, Vifor, Astra Zeneca, Bayer, Renibus Therapeutics, Travere Therapeutics, and GlaxoSmithKline. He is president of NephJC, a nonprofit educational organization with no industry support. Dr. Shirali declared no competing interests.
A version of this article appeared on Medscape.com.
FROM THE BMJ
Purpuric Eruption in a Patient With Hairy Cell Leukemia
The Diagnosis: Purpuric Drug Eruption
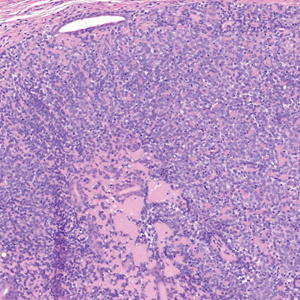
Histopathology revealed interface dermatitis, spongiosis, and a perivascular lymphocytic infiltrate with extravasated red blood cells consistent with a purpuric drug eruption. Our patient achieved remission of hairy cell leukemia after receiving only 2 of 5 expected doses of cladribine. The rash resolved completely in 3 weeks following a prednisone taper (Figure).
Hairy cell leukemia is a rare indolent lymphoproliferative disorder of B cells that accounts for approximately 2% of adult leukemias in the United States. Cladribine, a purine nucleoside analog that impairs DNA synthesis and repair, has become the mainstay of therapy, demonstrating a 95% complete response rate.1 Although few reports have addressed the cutaneous reactions seen with cladribine therapy, they can occur in more than 50% of patients.1,2 The most common skin manifestation associated with cladribine therapy is a morbilliform rash, but Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN) have been reported.1
Few cases of purpuric eruption secondary to cladribine treatment have been described, and nearly all reports involve concomitant medications such as allopurinol, which our patient was taking, and antibiotics including trimethoprim-sulfamethoxazole and penicillins.1,3,4 In a cohort of 35 patients receiving cladribine,1 only concomitant treatment with cladribine and allopurinol caused cutaneous reactions, further supporting the hypothesis of cladribine-induced drug sensitivity. Allopurinol often is prescribed during induction therapy for prophylaxis against tumor lysis syndrome; similarly, antibiotics frequently are given prophylactically and therapeutically for neutropenic fever. It is believed that T-cell imbalance and profound lymphopenia induced by cladribine increase susceptibility to drug hypersensitivity reactions.1,3
The typical purpuric eruption develops within 2 days of starting cladribine therapy. Diascopy will reveal petechiae, and biopsy should be performed to rule out other serious drug-induced reactions, such as erythema multiforme, Stevens-Johnson syndrome, and TEN. A cladribine-induced purpuric eruption typically is self-resolving and carries a favorable prognosis, though high-dose corticosteroids often are prescribed to hasten recovery. The rare reports of serious cutaneous reactions secondary to cladribine therapy have been with maculopapular, not purpuric eruptions.2 Based on limited available data, cladribine-induced purpura should not be a limitation to continued treatment in patients who need it.1 Careful consideration of concomitant drug use is necessary, as the current literature demonstrates resolution of rash with withdrawal of other therapies, namely allopurinol.2-4 Future studies are needed to examine the safety of withholding offending medications and to further elucidate the mechanisms contributing to drug hypersensitivity due to cladribine.
Widespread purpura and petechiae can pose a wide differential; the patient’s recent history of cladribine administration pointed to a classic purpuric eruption. Other diagnoses such as toxic erythema of chemotherapy (TEC) and TEN are not purpuric, though plaques can be violaceous. Lack of bullae, blisters, and facial or mucosal surface involvement suggest TEN.5 Thrombotic thrombocytopenic purpura and disseminated intravascular coagulation do manifest with petechiae and purpura, though such a robust eruption in the context of recent cladribine therapy is less likely. The classic retiform purpura and necrosis were not present to suggest purpura fulminans from disseminated intravascular coagulation.
Several of the proposed diagnoses as well as a purpuric drug eruption would demonstrate extravasated red blood cells on histopathology, but the presence of interface dermatitis narrows the differential to a purpuric drug eruption. Necrotic keratinocytes and full-thickness necrosis were not present on biopsy to support a diagnosis of TEN in our patient. Characteristic features of TEC—including eccrine squamous syringometaplasia, dermal edema, and keratinocyte atypia—were not present on biopsy.6 Finally, although TEN should resolve with steroid treatment, TEC is self-limited and thrombotic thrombocytopenic purpura and disseminated intravascular coagulation would not resolve with use of steroids alone.
- Ganzel C, Gatt ME, Maly A, et al. High incidence of skin rash in patients with hairy cell leukemia treated with cladribine. Leuk Lymphoma. 2012;53:1169-1173. doi:10.3109/10428194.2011.635864
- Chubar Y, Bennett M. Cutaneous reactions in hairy cell leukaemia treated with 2-chlorodeoxyadenosine and allopurinol. Br J Haematol. 2003;122:768-770. doi:10.1046/j.1365-2141.2003.04506.x
- Espinosa Lara P, Quirós Redondo V, Aguado Lobo M, et al. Purpuric exanthema in a patient with hairy cell leukemia treated with cladribine and allopurinol. Ann Hematol. 2017;96:1209-1210. doi:10.1007 /s00277-017-2992-z
- Hendrick A. Purpuric rash following treatment with 2-chlorodeoxyadenosine. Clin Lab Haematol. 2001;23:67-68. doi:10.1046 /j.1365-2257.2001.0346b.x
- Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw-Hill Education; 2019.
- Bolognia JL, Cooper DL, Glusac EJ. Toxic erythema of chemotherapy: a useful clinical term. J Am Acad Dermatol. 2008;59:524-529.
The Diagnosis: Purpuric Drug Eruption
Histopathology revealed interface dermatitis, spongiosis, and a perivascular lymphocytic infiltrate with extravasated red blood cells consistent with a purpuric drug eruption. Our patient achieved remission of hairy cell leukemia after receiving only 2 of 5 expected doses of cladribine. The rash resolved completely in 3 weeks following a prednisone taper (Figure).

Hairy cell leukemia is a rare indolent lymphoproliferative disorder of B cells that accounts for approximately 2% of adult leukemias in the United States. Cladribine, a purine nucleoside analog that impairs DNA synthesis and repair, has become the mainstay of therapy, demonstrating a 95% complete response rate.1 Although few reports have addressed the cutaneous reactions seen with cladribine therapy, they can occur in more than 50% of patients.1,2 The most common skin manifestation associated with cladribine therapy is a morbilliform rash, but Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN) have been reported.1
Few cases of purpuric eruption secondary to cladribine treatment have been described, and nearly all reports involve concomitant medications such as allopurinol, which our patient was taking, and antibiotics including trimethoprim-sulfamethoxazole and penicillins.1,3,4 In a cohort of 35 patients receiving cladribine,1 only concomitant treatment with cladribine and allopurinol caused cutaneous reactions, further supporting the hypothesis of cladribine-induced drug sensitivity. Allopurinol often is prescribed during induction therapy for prophylaxis against tumor lysis syndrome; similarly, antibiotics frequently are given prophylactically and therapeutically for neutropenic fever. It is believed that T-cell imbalance and profound lymphopenia induced by cladribine increase susceptibility to drug hypersensitivity reactions.1,3
The typical purpuric eruption develops within 2 days of starting cladribine therapy. Diascopy will reveal petechiae, and biopsy should be performed to rule out other serious drug-induced reactions, such as erythema multiforme, Stevens-Johnson syndrome, and TEN. A cladribine-induced purpuric eruption typically is self-resolving and carries a favorable prognosis, though high-dose corticosteroids often are prescribed to hasten recovery. The rare reports of serious cutaneous reactions secondary to cladribine therapy have been with maculopapular, not purpuric eruptions.2 Based on limited available data, cladribine-induced purpura should not be a limitation to continued treatment in patients who need it.1 Careful consideration of concomitant drug use is necessary, as the current literature demonstrates resolution of rash with withdrawal of other therapies, namely allopurinol.2-4 Future studies are needed to examine the safety of withholding offending medications and to further elucidate the mechanisms contributing to drug hypersensitivity due to cladribine.
Widespread purpura and petechiae can pose a wide differential; the patient’s recent history of cladribine administration pointed to a classic purpuric eruption. Other diagnoses such as toxic erythema of chemotherapy (TEC) and TEN are not purpuric, though plaques can be violaceous. Lack of bullae, blisters, and facial or mucosal surface involvement suggest TEN.5 Thrombotic thrombocytopenic purpura and disseminated intravascular coagulation do manifest with petechiae and purpura, though such a robust eruption in the context of recent cladribine therapy is less likely. The classic retiform purpura and necrosis were not present to suggest purpura fulminans from disseminated intravascular coagulation.
Several of the proposed diagnoses as well as a purpuric drug eruption would demonstrate extravasated red blood cells on histopathology, but the presence of interface dermatitis narrows the differential to a purpuric drug eruption. Necrotic keratinocytes and full-thickness necrosis were not present on biopsy to support a diagnosis of TEN in our patient. Characteristic features of TEC—including eccrine squamous syringometaplasia, dermal edema, and keratinocyte atypia—were not present on biopsy.6 Finally, although TEN should resolve with steroid treatment, TEC is self-limited and thrombotic thrombocytopenic purpura and disseminated intravascular coagulation would not resolve with use of steroids alone.
The Diagnosis: Purpuric Drug Eruption
Histopathology revealed interface dermatitis, spongiosis, and a perivascular lymphocytic infiltrate with extravasated red blood cells consistent with a purpuric drug eruption. Our patient achieved remission of hairy cell leukemia after receiving only 2 of 5 expected doses of cladribine. The rash resolved completely in 3 weeks following a prednisone taper (Figure).

Hairy cell leukemia is a rare indolent lymphoproliferative disorder of B cells that accounts for approximately 2% of adult leukemias in the United States. Cladribine, a purine nucleoside analog that impairs DNA synthesis and repair, has become the mainstay of therapy, demonstrating a 95% complete response rate.1 Although few reports have addressed the cutaneous reactions seen with cladribine therapy, they can occur in more than 50% of patients.1,2 The most common skin manifestation associated with cladribine therapy is a morbilliform rash, but Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN) have been reported.1
Few cases of purpuric eruption secondary to cladribine treatment have been described, and nearly all reports involve concomitant medications such as allopurinol, which our patient was taking, and antibiotics including trimethoprim-sulfamethoxazole and penicillins.1,3,4 In a cohort of 35 patients receiving cladribine,1 only concomitant treatment with cladribine and allopurinol caused cutaneous reactions, further supporting the hypothesis of cladribine-induced drug sensitivity. Allopurinol often is prescribed during induction therapy for prophylaxis against tumor lysis syndrome; similarly, antibiotics frequently are given prophylactically and therapeutically for neutropenic fever. It is believed that T-cell imbalance and profound lymphopenia induced by cladribine increase susceptibility to drug hypersensitivity reactions.1,3
The typical purpuric eruption develops within 2 days of starting cladribine therapy. Diascopy will reveal petechiae, and biopsy should be performed to rule out other serious drug-induced reactions, such as erythema multiforme, Stevens-Johnson syndrome, and TEN. A cladribine-induced purpuric eruption typically is self-resolving and carries a favorable prognosis, though high-dose corticosteroids often are prescribed to hasten recovery. The rare reports of serious cutaneous reactions secondary to cladribine therapy have been with maculopapular, not purpuric eruptions.2 Based on limited available data, cladribine-induced purpura should not be a limitation to continued treatment in patients who need it.1 Careful consideration of concomitant drug use is necessary, as the current literature demonstrates resolution of rash with withdrawal of other therapies, namely allopurinol.2-4 Future studies are needed to examine the safety of withholding offending medications and to further elucidate the mechanisms contributing to drug hypersensitivity due to cladribine.
Widespread purpura and petechiae can pose a wide differential; the patient’s recent history of cladribine administration pointed to a classic purpuric eruption. Other diagnoses such as toxic erythema of chemotherapy (TEC) and TEN are not purpuric, though plaques can be violaceous. Lack of bullae, blisters, and facial or mucosal surface involvement suggest TEN.5 Thrombotic thrombocytopenic purpura and disseminated intravascular coagulation do manifest with petechiae and purpura, though such a robust eruption in the context of recent cladribine therapy is less likely. The classic retiform purpura and necrosis were not present to suggest purpura fulminans from disseminated intravascular coagulation.
Several of the proposed diagnoses as well as a purpuric drug eruption would demonstrate extravasated red blood cells on histopathology, but the presence of interface dermatitis narrows the differential to a purpuric drug eruption. Necrotic keratinocytes and full-thickness necrosis were not present on biopsy to support a diagnosis of TEN in our patient. Characteristic features of TEC—including eccrine squamous syringometaplasia, dermal edema, and keratinocyte atypia—were not present on biopsy.6 Finally, although TEN should resolve with steroid treatment, TEC is self-limited and thrombotic thrombocytopenic purpura and disseminated intravascular coagulation would not resolve with use of steroids alone.
- Ganzel C, Gatt ME, Maly A, et al. High incidence of skin rash in patients with hairy cell leukemia treated with cladribine. Leuk Lymphoma. 2012;53:1169-1173. doi:10.3109/10428194.2011.635864
- Chubar Y, Bennett M. Cutaneous reactions in hairy cell leukaemia treated with 2-chlorodeoxyadenosine and allopurinol. Br J Haematol. 2003;122:768-770. doi:10.1046/j.1365-2141.2003.04506.x
- Espinosa Lara P, Quirós Redondo V, Aguado Lobo M, et al. Purpuric exanthema in a patient with hairy cell leukemia treated with cladribine and allopurinol. Ann Hematol. 2017;96:1209-1210. doi:10.1007 /s00277-017-2992-z
- Hendrick A. Purpuric rash following treatment with 2-chlorodeoxyadenosine. Clin Lab Haematol. 2001;23:67-68. doi:10.1046 /j.1365-2257.2001.0346b.x
- Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw-Hill Education; 2019.
- Bolognia JL, Cooper DL, Glusac EJ. Toxic erythema of chemotherapy: a useful clinical term. J Am Acad Dermatol. 2008;59:524-529.
- Ganzel C, Gatt ME, Maly A, et al. High incidence of skin rash in patients with hairy cell leukemia treated with cladribine. Leuk Lymphoma. 2012;53:1169-1173. doi:10.3109/10428194.2011.635864
- Chubar Y, Bennett M. Cutaneous reactions in hairy cell leukaemia treated with 2-chlorodeoxyadenosine and allopurinol. Br J Haematol. 2003;122:768-770. doi:10.1046/j.1365-2141.2003.04506.x
- Espinosa Lara P, Quirós Redondo V, Aguado Lobo M, et al. Purpuric exanthema in a patient with hairy cell leukemia treated with cladribine and allopurinol. Ann Hematol. 2017;96:1209-1210. doi:10.1007 /s00277-017-2992-z
- Hendrick A. Purpuric rash following treatment with 2-chlorodeoxyadenosine. Clin Lab Haematol. 2001;23:67-68. doi:10.1046 /j.1365-2257.2001.0346b.x
- Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw-Hill Education; 2019.
- Bolognia JL, Cooper DL, Glusac EJ. Toxic erythema of chemotherapy: a useful clinical term. J Am Acad Dermatol. 2008;59:524-529.
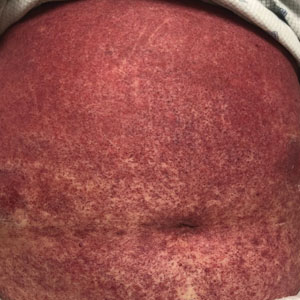
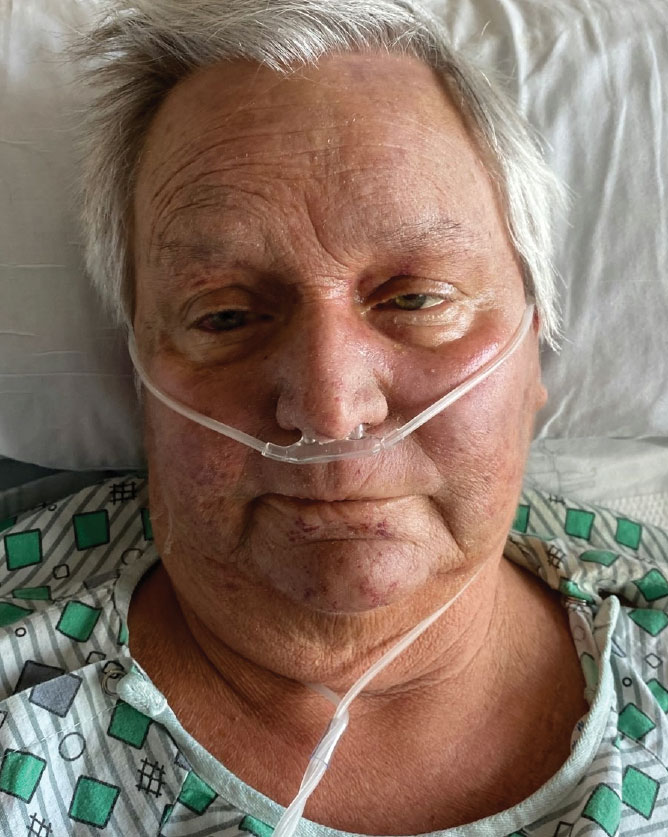
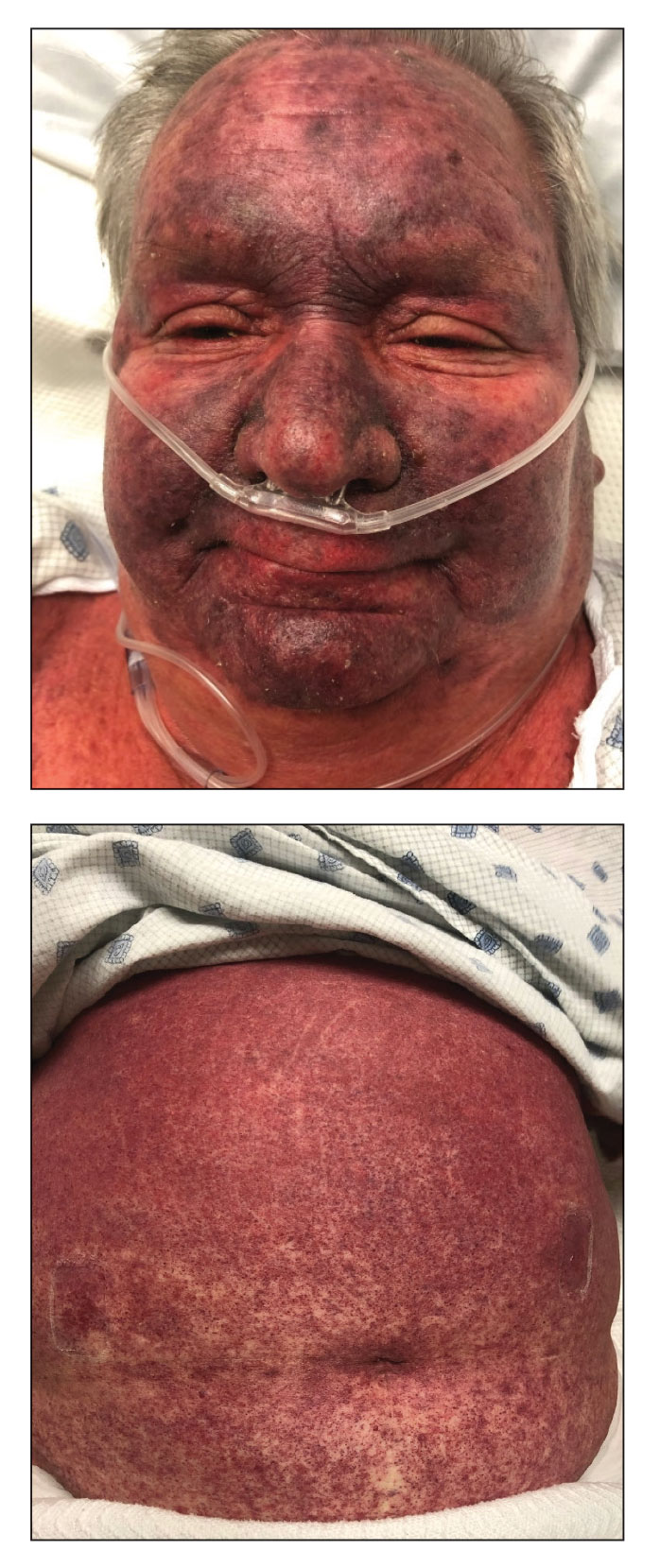
A 68-year-old woman presented to the emergency department with neutropenic fever and a rash over the body after receiving 2 doses of cladribine therapy for hairy cell leukemia. Physical examination demonstrated marked facial (top), lip, and tongue swelling, as well as a diffuse dusky nonpalpable purpuric rash on the abdomen (bottom) and back involving 90% of the body surface area. Bilateral ear edema was appreciated with accentuation of the earlobe crease. The patient exhibited subconjunctival hemorrhage, ectropion, and scleral injection. A punch biopsy of the thigh was performed.
Do Patients Benefit from Cancer Trial Participation?
TOPLINE:
METHODOLOGY:
- The view that patients with cancer benefit from access to investigational drugs in the clinical trial setting is widely held but does necessarily align with trial findings, which often show limited evidence of a clinical benefit. First, most investigational treatments assessed in clinical trials fail to gain regulatory approval, and the minority that are approved tend to offer minimal clinical benefit, experts explained.
- To estimate the survival benefit and toxicities associated with receiving experimental treatments, researchers conducted a meta-analysis of 128 trials comprising 141 comparisons of an investigational drug and a control treatment, which included immunotherapies and targeted therapies.
- The analysis included 42 trials in non–small cell lung cancer (NSCLC), 37 in breast cancer, 15 in hepatobiliary cancer, 13 in pancreatic cancer, 12 in colorectal cancer, and 10 in prostate cancer, involving a total of 47,050 patients.
- The primary outcome was PFS and secondary outcomes were overall survival and grades 3-5 serious adverse events.
TAKEAWAY:
- Overall, the experimental treatment was associated with a 20% improvement in PFS (pooled hazard ratio [HR], 0.80), corresponding to a median 1.25-month PFS advantage. The PFS benefit was seen across all cancer types, except pancreatic cancer.
- Overall survival improved by 8% with experimental agents (HR, 0.92), corresponding to 1.18 additional months. A significant overall survival benefit was seen across NSCLC, breast cancer, and hepatobiliary cancer trials but not pancreatic, prostate, colorectal cancer trials.
- Patients in the experimental intervention group, however, experienced much higher risk for grade 3-5 serious adverse events (risk ratio [RR], 1.27), corresponding to 7.40% increase in absolute risk. The greater risk for serious adverse events was significant for all indications except prostate cancer (RR, 1.13; 95% CI, 0.91-1.40).
IN PRACTICE:
“We believe our findings are best interpreted as suggesting that access to experimental interventions that have not yet received full FDA approval is associated with a marginal but nonzero clinical benefit,” the authors wrote.
“Although our findings seem to reflect poorly on trials as a vehicle for extending survival for participants, they have reassuring implications for clinical investigators, policymakers, and institutional review boards,” the researchers said, explaining that this “scenario allows clinical trials to continue to pursue promising new treatments — supporting incremental advances that sum to large gains over extended periods of research — without disadvantaging patients in comparator groups.”
SOURCE:
Renata Iskander, MSc, of McGill University, Montreal, Quebec, Canada, led this work, which was published online on April 29, 2024, in Annals of Internal Medicine.
LIMITATIONS:
There was high heterogeneity across studies due to variations in drugs tested, comparators used, and populations involved. The use of comparators below standard care could have inflated survival benefits. Additionally, data collected from ClinicalTrials.gov might be biased due to some trials not being reported.
DISCLOSURES:
Canadian Institutes of Health Research supported this work. The authors received grants for this work from McGill University, Rossy Cancer Network, and National Science Foundation. One author received consulting fees outside this work. The other authors declared no competing interests.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The view that patients with cancer benefit from access to investigational drugs in the clinical trial setting is widely held but does necessarily align with trial findings, which often show limited evidence of a clinical benefit. First, most investigational treatments assessed in clinical trials fail to gain regulatory approval, and the minority that are approved tend to offer minimal clinical benefit, experts explained.
- To estimate the survival benefit and toxicities associated with receiving experimental treatments, researchers conducted a meta-analysis of 128 trials comprising 141 comparisons of an investigational drug and a control treatment, which included immunotherapies and targeted therapies.
- The analysis included 42 trials in non–small cell lung cancer (NSCLC), 37 in breast cancer, 15 in hepatobiliary cancer, 13 in pancreatic cancer, 12 in colorectal cancer, and 10 in prostate cancer, involving a total of 47,050 patients.
- The primary outcome was PFS and secondary outcomes were overall survival and grades 3-5 serious adverse events.
TAKEAWAY:
- Overall, the experimental treatment was associated with a 20% improvement in PFS (pooled hazard ratio [HR], 0.80), corresponding to a median 1.25-month PFS advantage. The PFS benefit was seen across all cancer types, except pancreatic cancer.
- Overall survival improved by 8% with experimental agents (HR, 0.92), corresponding to 1.18 additional months. A significant overall survival benefit was seen across NSCLC, breast cancer, and hepatobiliary cancer trials but not pancreatic, prostate, colorectal cancer trials.
- Patients in the experimental intervention group, however, experienced much higher risk for grade 3-5 serious adverse events (risk ratio [RR], 1.27), corresponding to 7.40% increase in absolute risk. The greater risk for serious adverse events was significant for all indications except prostate cancer (RR, 1.13; 95% CI, 0.91-1.40).
IN PRACTICE:
“We believe our findings are best interpreted as suggesting that access to experimental interventions that have not yet received full FDA approval is associated with a marginal but nonzero clinical benefit,” the authors wrote.
“Although our findings seem to reflect poorly on trials as a vehicle for extending survival for participants, they have reassuring implications for clinical investigators, policymakers, and institutional review boards,” the researchers said, explaining that this “scenario allows clinical trials to continue to pursue promising new treatments — supporting incremental advances that sum to large gains over extended periods of research — without disadvantaging patients in comparator groups.”
SOURCE:
Renata Iskander, MSc, of McGill University, Montreal, Quebec, Canada, led this work, which was published online on April 29, 2024, in Annals of Internal Medicine.
LIMITATIONS:
There was high heterogeneity across studies due to variations in drugs tested, comparators used, and populations involved. The use of comparators below standard care could have inflated survival benefits. Additionally, data collected from ClinicalTrials.gov might be biased due to some trials not being reported.
DISCLOSURES:
Canadian Institutes of Health Research supported this work. The authors received grants for this work from McGill University, Rossy Cancer Network, and National Science Foundation. One author received consulting fees outside this work. The other authors declared no competing interests.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The view that patients with cancer benefit from access to investigational drugs in the clinical trial setting is widely held but does necessarily align with trial findings, which often show limited evidence of a clinical benefit. First, most investigational treatments assessed in clinical trials fail to gain regulatory approval, and the minority that are approved tend to offer minimal clinical benefit, experts explained.
- To estimate the survival benefit and toxicities associated with receiving experimental treatments, researchers conducted a meta-analysis of 128 trials comprising 141 comparisons of an investigational drug and a control treatment, which included immunotherapies and targeted therapies.
- The analysis included 42 trials in non–small cell lung cancer (NSCLC), 37 in breast cancer, 15 in hepatobiliary cancer, 13 in pancreatic cancer, 12 in colorectal cancer, and 10 in prostate cancer, involving a total of 47,050 patients.
- The primary outcome was PFS and secondary outcomes were overall survival and grades 3-5 serious adverse events.
TAKEAWAY:
- Overall, the experimental treatment was associated with a 20% improvement in PFS (pooled hazard ratio [HR], 0.80), corresponding to a median 1.25-month PFS advantage. The PFS benefit was seen across all cancer types, except pancreatic cancer.
- Overall survival improved by 8% with experimental agents (HR, 0.92), corresponding to 1.18 additional months. A significant overall survival benefit was seen across NSCLC, breast cancer, and hepatobiliary cancer trials but not pancreatic, prostate, colorectal cancer trials.
- Patients in the experimental intervention group, however, experienced much higher risk for grade 3-5 serious adverse events (risk ratio [RR], 1.27), corresponding to 7.40% increase in absolute risk. The greater risk for serious adverse events was significant for all indications except prostate cancer (RR, 1.13; 95% CI, 0.91-1.40).
IN PRACTICE:
“We believe our findings are best interpreted as suggesting that access to experimental interventions that have not yet received full FDA approval is associated with a marginal but nonzero clinical benefit,” the authors wrote.
“Although our findings seem to reflect poorly on trials as a vehicle for extending survival for participants, they have reassuring implications for clinical investigators, policymakers, and institutional review boards,” the researchers said, explaining that this “scenario allows clinical trials to continue to pursue promising new treatments — supporting incremental advances that sum to large gains over extended periods of research — without disadvantaging patients in comparator groups.”
SOURCE:
Renata Iskander, MSc, of McGill University, Montreal, Quebec, Canada, led this work, which was published online on April 29, 2024, in Annals of Internal Medicine.
LIMITATIONS:
There was high heterogeneity across studies due to variations in drugs tested, comparators used, and populations involved. The use of comparators below standard care could have inflated survival benefits. Additionally, data collected from ClinicalTrials.gov might be biased due to some trials not being reported.
DISCLOSURES:
Canadian Institutes of Health Research supported this work. The authors received grants for this work from McGill University, Rossy Cancer Network, and National Science Foundation. One author received consulting fees outside this work. The other authors declared no competing interests.
A version of this article appeared on Medscape.com.
Do Health-Related Social Needs Raise Mortality Risk in Cancer Survivors?
Little is known about the specific association between health-related social needs (HRSNs) and mortality risk even though HRSNs, defined as challenges in affording food, housing, and other necessities of daily living, are potential challenges for cancer survivors, wrote Zhiyuan Zheng, PhD, of the American Cancer Society, Atlanta, and colleagues.
A 2020 study by Dr. Zheng and colleagues published in the Journal of the National Comprehensive Cancer Network (NCCN) showed that food insecurity and financial worries had a negative impact on cancer survivorship. In the new study, published in Cancer, the researchers identified cancer survivors using the 2013-2018 National Health Interview Survey (NHIS) and the NHIS Mortality File through December 31, 2019. The researchers examined mortality using the data from the Centers for Disease Control and Prevention’s National Death Index (NDI) through December 31, 2019, which links to the National Health Interview Survey Data used in the study.
Individuals’ HRSNs were categorized into three groups: severe, moderate, and minor/none. HRSNs included food insecurity and nonmedical financial concerns, such as housing costs (rent, mortgage). Medical financial hardship included material, psychological, and behavioral domains and was divided into three groups: 2-3 domains, 1 domain, or 0 domains.
What Are the Potential Financial Implications of this Research?
The high costs of cancer care often cause medical financial hardships for cancer survivors, and expenses also may cause psychological distress and nonmedical financial hardship as survivors try to make ends meet while facing medical bills, wrote Dr. Zheng and colleagues.
Policy makers are increasingly interested in adding HRSNs to insurance coverage; recent guidance from the Centers for Medicare & Medicaid Services (CMS) allows individual states to apply to provide nutrition and housing supports through state Medicaid programs, according to authors of a 2023 article published in JAMA Health Forum.
The new study adds to the understanding of how HRSNs impact people with cancer by examining the association with mortality risk, Yelak Biru, MSc, president and chief executive officer of the International Myeloma Foundation, said in an interview.
“This is a key area of study for addressing the disparities in treatments and outcomes that result in inequities,” said Mr. Biru, a patient advocate and multiple myeloma survivor who was not involved in the study.
What Does the New Study Show?
The new study characterized HRSNs in 5,855 adult cancer survivors aged 18-64 years and 5,918 aged 65-79 years. In the 18- to 64-year-old group, 25.5% reported moderate levels of HRSNs, and 18.3% reported severe HRSNs. In patients aged 65-79 years, 15.6% and 6.6% reported moderate HRSNs and severe HRSNs, respectively.
Severe HRSN was significantly associated with higher mortality risk in an adjusted analysis in patients aged 18-64 years (hazard ratio 2.00, P < .001).
Among adults aged 65-79 years, severe HRSN was not associated with higher mortality risk; however, in this older age group, those with 2-3 domains of medical financial hardship had significantly increased mortality risk compared with adults aged 65-79 years with zero domains of medical financial hardship (HR 1.58, P = .007).
Although the findings that HRSNs were associated with increased mortality risk, especially in the younger group, were not surprising, they serve as a call to action to address how HRSNs are contributing to cancer mortality, Mr. Biru said in an interview. “HRSNs, like food or housing insecurity, can lead to patients being unable to undergo the best treatment approach for their cancer,” he said.
What Are the Limitations and Research Gaps?
The study findings were limited by several factors including the use of self-reports to measure medical financial hardship, food insecurity, and nonmedical financial concerns in the NHIS, the researchers wrote in their discussion. More research with longer follow-up time beyond 1-5 years is needed, wrote Dr. Zheng and colleagues.
Studies also are needed to illustrate how patient navigation can help prevent patients from falling through the cracks with regard to social needs and financial hardships, Mr. Biru told this news organization.
Other areas for research include how addressing social needs affects health outcomes and whether programs designed to address social needs are effective, he said.
“Finally, qualitative research is needed to capture the lived experiences of cancer survivors facing these challenges. This knowledge can inform the development of more patient-centered interventions and policies that effectively address the social determinants of health and improve overall outcomes for all cancer survivors,” Mr. Biru said.
What Is the Takeaway Message for Clinicians?
HRSNs and financial hardship are significantly associated with increased risk of mortality in adult cancer survivors, Dr. Zheng and colleagues concluded. Looking ahead, comprehensive assessment of HRSNs and financial hardship may help clinicians connect patients with relevant services to mitigate the social and financial impacts of cancer, they wrote.
“The takeaway message for oncologists in practice is that addressing [HRSNs] and financial hardship is crucial for providing comprehensive and equitable cancer care,” Mr. Biru said during his interview.
“The impact of social determinants of health on cancer outcomes cannot be ignored, and oncologists play a vital role in identifying and addressing these needs,” he said. Sensitive, discussion-based screenings are needed to identify core needs such as food and transportation, but clinicians also can consider broader social factors and work with a team to connect patients to appropriate resources, he added.
“By recognizing the importance of HRSN screening and taking proactive steps to address these needs, oncologists can contribute to improving health outcomes, reducing healthcare disparities, and providing more equitable cancer care for their patients,” he said.
What Other Guidance Is Available?
“High-quality cancer care requires treating the whole person; measuring and addressing anything in their life that could result in poorer health outcomes is a key component of comprehensive care,” Mr. Biru emphasized.
In September 2023, the National Comprehensive Cancer Network (NCCN) convened a working group cochaired by Mr. Biru that developed recommendations for how oncology practices should routinely measure HRSNs (NCCN.org/social-needs).
“The working group proposed that every cancer patient be assessed for food, transportation access, and financial and housing security at least once a year, and be reassessed at every care transition point as well,” Mr. Biru said. Such screenings should include follow-up to connect patients with services to address any HRSNs they are experiencing, he added.
Lead author Dr. Zheng is employed by the American Cancer Society, which as a nonprofit receives funds from the public through fundraising and contributions, as well as some support from corporations and industry to support its mission programs and services. Mr. Biru had no financial conflicts to disclose.
Little is known about the specific association between health-related social needs (HRSNs) and mortality risk even though HRSNs, defined as challenges in affording food, housing, and other necessities of daily living, are potential challenges for cancer survivors, wrote Zhiyuan Zheng, PhD, of the American Cancer Society, Atlanta, and colleagues.
A 2020 study by Dr. Zheng and colleagues published in the Journal of the National Comprehensive Cancer Network (NCCN) showed that food insecurity and financial worries had a negative impact on cancer survivorship. In the new study, published in Cancer, the researchers identified cancer survivors using the 2013-2018 National Health Interview Survey (NHIS) and the NHIS Mortality File through December 31, 2019. The researchers examined mortality using the data from the Centers for Disease Control and Prevention’s National Death Index (NDI) through December 31, 2019, which links to the National Health Interview Survey Data used in the study.
Individuals’ HRSNs were categorized into three groups: severe, moderate, and minor/none. HRSNs included food insecurity and nonmedical financial concerns, such as housing costs (rent, mortgage). Medical financial hardship included material, psychological, and behavioral domains and was divided into three groups: 2-3 domains, 1 domain, or 0 domains.
What Are the Potential Financial Implications of this Research?
The high costs of cancer care often cause medical financial hardships for cancer survivors, and expenses also may cause psychological distress and nonmedical financial hardship as survivors try to make ends meet while facing medical bills, wrote Dr. Zheng and colleagues.
Policy makers are increasingly interested in adding HRSNs to insurance coverage; recent guidance from the Centers for Medicare & Medicaid Services (CMS) allows individual states to apply to provide nutrition and housing supports through state Medicaid programs, according to authors of a 2023 article published in JAMA Health Forum.
The new study adds to the understanding of how HRSNs impact people with cancer by examining the association with mortality risk, Yelak Biru, MSc, president and chief executive officer of the International Myeloma Foundation, said in an interview.
“This is a key area of study for addressing the disparities in treatments and outcomes that result in inequities,” said Mr. Biru, a patient advocate and multiple myeloma survivor who was not involved in the study.
What Does the New Study Show?
The new study characterized HRSNs in 5,855 adult cancer survivors aged 18-64 years and 5,918 aged 65-79 years. In the 18- to 64-year-old group, 25.5% reported moderate levels of HRSNs, and 18.3% reported severe HRSNs. In patients aged 65-79 years, 15.6% and 6.6% reported moderate HRSNs and severe HRSNs, respectively.
Severe HRSN was significantly associated with higher mortality risk in an adjusted analysis in patients aged 18-64 years (hazard ratio 2.00, P < .001).
Among adults aged 65-79 years, severe HRSN was not associated with higher mortality risk; however, in this older age group, those with 2-3 domains of medical financial hardship had significantly increased mortality risk compared with adults aged 65-79 years with zero domains of medical financial hardship (HR 1.58, P = .007).
Although the findings that HRSNs were associated with increased mortality risk, especially in the younger group, were not surprising, they serve as a call to action to address how HRSNs are contributing to cancer mortality, Mr. Biru said in an interview. “HRSNs, like food or housing insecurity, can lead to patients being unable to undergo the best treatment approach for their cancer,” he said.
What Are the Limitations and Research Gaps?
The study findings were limited by several factors including the use of self-reports to measure medical financial hardship, food insecurity, and nonmedical financial concerns in the NHIS, the researchers wrote in their discussion. More research with longer follow-up time beyond 1-5 years is needed, wrote Dr. Zheng and colleagues.
Studies also are needed to illustrate how patient navigation can help prevent patients from falling through the cracks with regard to social needs and financial hardships, Mr. Biru told this news organization.
Other areas for research include how addressing social needs affects health outcomes and whether programs designed to address social needs are effective, he said.
“Finally, qualitative research is needed to capture the lived experiences of cancer survivors facing these challenges. This knowledge can inform the development of more patient-centered interventions and policies that effectively address the social determinants of health and improve overall outcomes for all cancer survivors,” Mr. Biru said.
What Is the Takeaway Message for Clinicians?
HRSNs and financial hardship are significantly associated with increased risk of mortality in adult cancer survivors, Dr. Zheng and colleagues concluded. Looking ahead, comprehensive assessment of HRSNs and financial hardship may help clinicians connect patients with relevant services to mitigate the social and financial impacts of cancer, they wrote.
“The takeaway message for oncologists in practice is that addressing [HRSNs] and financial hardship is crucial for providing comprehensive and equitable cancer care,” Mr. Biru said during his interview.
“The impact of social determinants of health on cancer outcomes cannot be ignored, and oncologists play a vital role in identifying and addressing these needs,” he said. Sensitive, discussion-based screenings are needed to identify core needs such as food and transportation, but clinicians also can consider broader social factors and work with a team to connect patients to appropriate resources, he added.
“By recognizing the importance of HRSN screening and taking proactive steps to address these needs, oncologists can contribute to improving health outcomes, reducing healthcare disparities, and providing more equitable cancer care for their patients,” he said.
What Other Guidance Is Available?
“High-quality cancer care requires treating the whole person; measuring and addressing anything in their life that could result in poorer health outcomes is a key component of comprehensive care,” Mr. Biru emphasized.
In September 2023, the National Comprehensive Cancer Network (NCCN) convened a working group cochaired by Mr. Biru that developed recommendations for how oncology practices should routinely measure HRSNs (NCCN.org/social-needs).
“The working group proposed that every cancer patient be assessed for food, transportation access, and financial and housing security at least once a year, and be reassessed at every care transition point as well,” Mr. Biru said. Such screenings should include follow-up to connect patients with services to address any HRSNs they are experiencing, he added.
Lead author Dr. Zheng is employed by the American Cancer Society, which as a nonprofit receives funds from the public through fundraising and contributions, as well as some support from corporations and industry to support its mission programs and services. Mr. Biru had no financial conflicts to disclose.
Little is known about the specific association between health-related social needs (HRSNs) and mortality risk even though HRSNs, defined as challenges in affording food, housing, and other necessities of daily living, are potential challenges for cancer survivors, wrote Zhiyuan Zheng, PhD, of the American Cancer Society, Atlanta, and colleagues.
A 2020 study by Dr. Zheng and colleagues published in the Journal of the National Comprehensive Cancer Network (NCCN) showed that food insecurity and financial worries had a negative impact on cancer survivorship. In the new study, published in Cancer, the researchers identified cancer survivors using the 2013-2018 National Health Interview Survey (NHIS) and the NHIS Mortality File through December 31, 2019. The researchers examined mortality using the data from the Centers for Disease Control and Prevention’s National Death Index (NDI) through December 31, 2019, which links to the National Health Interview Survey Data used in the study.
Individuals’ HRSNs were categorized into three groups: severe, moderate, and minor/none. HRSNs included food insecurity and nonmedical financial concerns, such as housing costs (rent, mortgage). Medical financial hardship included material, psychological, and behavioral domains and was divided into three groups: 2-3 domains, 1 domain, or 0 domains.
What Are the Potential Financial Implications of this Research?
The high costs of cancer care often cause medical financial hardships for cancer survivors, and expenses also may cause psychological distress and nonmedical financial hardship as survivors try to make ends meet while facing medical bills, wrote Dr. Zheng and colleagues.
Policy makers are increasingly interested in adding HRSNs to insurance coverage; recent guidance from the Centers for Medicare & Medicaid Services (CMS) allows individual states to apply to provide nutrition and housing supports through state Medicaid programs, according to authors of a 2023 article published in JAMA Health Forum.
The new study adds to the understanding of how HRSNs impact people with cancer by examining the association with mortality risk, Yelak Biru, MSc, president and chief executive officer of the International Myeloma Foundation, said in an interview.
“This is a key area of study for addressing the disparities in treatments and outcomes that result in inequities,” said Mr. Biru, a patient advocate and multiple myeloma survivor who was not involved in the study.
What Does the New Study Show?
The new study characterized HRSNs in 5,855 adult cancer survivors aged 18-64 years and 5,918 aged 65-79 years. In the 18- to 64-year-old group, 25.5% reported moderate levels of HRSNs, and 18.3% reported severe HRSNs. In patients aged 65-79 years, 15.6% and 6.6% reported moderate HRSNs and severe HRSNs, respectively.
Severe HRSN was significantly associated with higher mortality risk in an adjusted analysis in patients aged 18-64 years (hazard ratio 2.00, P < .001).
Among adults aged 65-79 years, severe HRSN was not associated with higher mortality risk; however, in this older age group, those with 2-3 domains of medical financial hardship had significantly increased mortality risk compared with adults aged 65-79 years with zero domains of medical financial hardship (HR 1.58, P = .007).
Although the findings that HRSNs were associated with increased mortality risk, especially in the younger group, were not surprising, they serve as a call to action to address how HRSNs are contributing to cancer mortality, Mr. Biru said in an interview. “HRSNs, like food or housing insecurity, can lead to patients being unable to undergo the best treatment approach for their cancer,” he said.
What Are the Limitations and Research Gaps?
The study findings were limited by several factors including the use of self-reports to measure medical financial hardship, food insecurity, and nonmedical financial concerns in the NHIS, the researchers wrote in their discussion. More research with longer follow-up time beyond 1-5 years is needed, wrote Dr. Zheng and colleagues.
Studies also are needed to illustrate how patient navigation can help prevent patients from falling through the cracks with regard to social needs and financial hardships, Mr. Biru told this news organization.
Other areas for research include how addressing social needs affects health outcomes and whether programs designed to address social needs are effective, he said.
“Finally, qualitative research is needed to capture the lived experiences of cancer survivors facing these challenges. This knowledge can inform the development of more patient-centered interventions and policies that effectively address the social determinants of health and improve overall outcomes for all cancer survivors,” Mr. Biru said.
What Is the Takeaway Message for Clinicians?
HRSNs and financial hardship are significantly associated with increased risk of mortality in adult cancer survivors, Dr. Zheng and colleagues concluded. Looking ahead, comprehensive assessment of HRSNs and financial hardship may help clinicians connect patients with relevant services to mitigate the social and financial impacts of cancer, they wrote.
“The takeaway message for oncologists in practice is that addressing [HRSNs] and financial hardship is crucial for providing comprehensive and equitable cancer care,” Mr. Biru said during his interview.
“The impact of social determinants of health on cancer outcomes cannot be ignored, and oncologists play a vital role in identifying and addressing these needs,” he said. Sensitive, discussion-based screenings are needed to identify core needs such as food and transportation, but clinicians also can consider broader social factors and work with a team to connect patients to appropriate resources, he added.
“By recognizing the importance of HRSN screening and taking proactive steps to address these needs, oncologists can contribute to improving health outcomes, reducing healthcare disparities, and providing more equitable cancer care for their patients,” he said.
What Other Guidance Is Available?
“High-quality cancer care requires treating the whole person; measuring and addressing anything in their life that could result in poorer health outcomes is a key component of comprehensive care,” Mr. Biru emphasized.
In September 2023, the National Comprehensive Cancer Network (NCCN) convened a working group cochaired by Mr. Biru that developed recommendations for how oncology practices should routinely measure HRSNs (NCCN.org/social-needs).
“The working group proposed that every cancer patient be assessed for food, transportation access, and financial and housing security at least once a year, and be reassessed at every care transition point as well,” Mr. Biru said. Such screenings should include follow-up to connect patients with services to address any HRSNs they are experiencing, he added.
Lead author Dr. Zheng is employed by the American Cancer Society, which as a nonprofit receives funds from the public through fundraising and contributions, as well as some support from corporations and industry to support its mission programs and services. Mr. Biru had no financial conflicts to disclose.
FROM CANCER
Terminal Cancer: What Matters to Patients and Caregivers
New research found that patients and caregivers both tend to prioritize symptom control over life extension but often preferring a balance. Patients and caregivers, however, are less aligned on decisions about cost containment, with patients more likely to prioritize cost containment.
“Our research has revealed that patients and caregivers generally share similar end-of-life goals,” with a “notable exception” when it comes to costs, first author Semra Ozdemir, PhD, with the Lien Centre for Palliative Care, Duke-NUS Medical School, Singapore, told this news organization.
However, when patients and caregivers have a better understanding of the patient’s prognosis, both may be more inclined to avoid costly life-extending treatments and prioritize symptom management.
In other words, the survey suggests that “knowing the prognosis helps patients and their families set realistic expectations for care and adequately prepare for end-of-life decisions,” said Dr. Ozdemir.
This study was published online in JAMA Network Open.
Patients with advanced cancer often face difficult decisions: Do they opt for treatments that may — or may not — extend life or do they focus more on symptom control?
Family caregivers, who also play an important role in this decision-making process, may have different care goals. Some research suggests that caregivers tend to prioritize treatments that could extend life, whereas patients prioritize symptom management, but it’s less clear how these priorities may change over time and how patients and caregivers may influence each other.
In the current study, the researchers examined goals of care among patients with stage IV solid tumors and caregivers during the last 2 years of life, focusing on life extension vs symptom management and cost containment, as well as how these goals changed over time.
The survey included 210 patient-caregiver pairs, recruited from outpatient clinics at two major cancer centers in Singapore. Patients had a mean age of 63 years, and about half were men. The caregivers had a mean age of 49 years, and almost two third (63%) were women.
Overall, 34% patients and 29% caregivers prioritized symptom management over life extension, whereas 24% patients and 19% caregivers prioritized life extension. Most patients and caregivers preferred balancing the two, with 34%-47% patients and 37%-69% caregivers supporting this approach.
When balancing cost and treatment decisions, however, patients were more likely to prioritize containing costs — 28% vs 17% for caregivers — over extending life — 26% of patients vs 35% of caregivers.
Cost containment tended to be more of a priority for older patients, those with a higher symptom burden, and those with less family caregiver support. For caregivers, cost containment was more of a priority for those who reported that caregiving had a big impact on their finances, those with worse self-esteem related to their caregiving abilities, as well as those caring for older patients.
To better align cost containment priorities between patients and caregivers, it’s essential for families to engage in open and thorough discussions about the allocation of resources, Dr. Ozdemir said.
Although “patients, families, and physicians often avoid discussions about prognosis,” such conversations are essential for setting realistic expectations for care and adequately preparing for end-of-life decisions, Dr. Ozdemir told this news organization.
“These conversations should aim to balance competing interests and create care plans that are mutually acceptable to both patients and caregivers,” she said, adding that “this approach will help in minimizing any potential conflicts and ensure that both parties feel respected and understood in their decision-making process.”
Managing Unrealistic Expectations
As patients approached the end of life, neither patients nor caregivers shifted their priorities from life extension to symptom management.
This finding raises concerns because it suggests that many patients hold unrealistic expectations regarding their care and “underscores the need for continuous dialogue and reassessment of care goals throughout the progression of illness,” Dr. Ozdemir said.
“This stability in preferences over time suggests that initial care decisions are deeply ingrained or that there may be a lack of ongoing communication about evolving care needs and possibilities as conditions change,” Ozdemir said.
Yet, it can be hard to define what unrealistic expectations mean, said Olivia Seecof, MD, who wasn’t involved in the study.
“I think people are hopeful that a devastating diagnosis won’t lead to the end of their life and that there will be a treatment or something that will change [their prognosis], and they’ll get better,” said Dr. Seecof, palliative care expert with the Supportive Oncology Program at NYU Langone Health’s Perlmutter Cancer Center in New York City.
Giving patients and caregivers a realistic understanding of the prognosis is important, but “there’s more to it than just telling the patient their diagnosis,” she said.
“We have to plan for end of life, what it can look like,” said Dr. Seecof, adding that “often we don’t do a very good job of talking about that early on in an illness course.”
Overall, though, Dr. Seecof stressed that no two patients or situations are the same, and it’s important to understand what’s important in each scenario. End-of-life care requires “an individual approach because every patient is different, even if they have the same diagnosis as someone else,” she said.
This work was supported by funding from the Singapore Millennium Foundation and the Lien Centre for Palliative Care. Dr. Ozdemir and Dr. Seecof had no relevant disclosures.
A version of this article appeared on Medscape.com.
New research found that patients and caregivers both tend to prioritize symptom control over life extension but often preferring a balance. Patients and caregivers, however, are less aligned on decisions about cost containment, with patients more likely to prioritize cost containment.
“Our research has revealed that patients and caregivers generally share similar end-of-life goals,” with a “notable exception” when it comes to costs, first author Semra Ozdemir, PhD, with the Lien Centre for Palliative Care, Duke-NUS Medical School, Singapore, told this news organization.
However, when patients and caregivers have a better understanding of the patient’s prognosis, both may be more inclined to avoid costly life-extending treatments and prioritize symptom management.
In other words, the survey suggests that “knowing the prognosis helps patients and their families set realistic expectations for care and adequately prepare for end-of-life decisions,” said Dr. Ozdemir.
This study was published online in JAMA Network Open.
Patients with advanced cancer often face difficult decisions: Do they opt for treatments that may — or may not — extend life or do they focus more on symptom control?
Family caregivers, who also play an important role in this decision-making process, may have different care goals. Some research suggests that caregivers tend to prioritize treatments that could extend life, whereas patients prioritize symptom management, but it’s less clear how these priorities may change over time and how patients and caregivers may influence each other.
In the current study, the researchers examined goals of care among patients with stage IV solid tumors and caregivers during the last 2 years of life, focusing on life extension vs symptom management and cost containment, as well as how these goals changed over time.
The survey included 210 patient-caregiver pairs, recruited from outpatient clinics at two major cancer centers in Singapore. Patients had a mean age of 63 years, and about half were men. The caregivers had a mean age of 49 years, and almost two third (63%) were women.
Overall, 34% patients and 29% caregivers prioritized symptom management over life extension, whereas 24% patients and 19% caregivers prioritized life extension. Most patients and caregivers preferred balancing the two, with 34%-47% patients and 37%-69% caregivers supporting this approach.
When balancing cost and treatment decisions, however, patients were more likely to prioritize containing costs — 28% vs 17% for caregivers — over extending life — 26% of patients vs 35% of caregivers.
Cost containment tended to be more of a priority for older patients, those with a higher symptom burden, and those with less family caregiver support. For caregivers, cost containment was more of a priority for those who reported that caregiving had a big impact on their finances, those with worse self-esteem related to their caregiving abilities, as well as those caring for older patients.
To better align cost containment priorities between patients and caregivers, it’s essential for families to engage in open and thorough discussions about the allocation of resources, Dr. Ozdemir said.
Although “patients, families, and physicians often avoid discussions about prognosis,” such conversations are essential for setting realistic expectations for care and adequately preparing for end-of-life decisions, Dr. Ozdemir told this news organization.
“These conversations should aim to balance competing interests and create care plans that are mutually acceptable to both patients and caregivers,” she said, adding that “this approach will help in minimizing any potential conflicts and ensure that both parties feel respected and understood in their decision-making process.”
Managing Unrealistic Expectations
As patients approached the end of life, neither patients nor caregivers shifted their priorities from life extension to symptom management.
This finding raises concerns because it suggests that many patients hold unrealistic expectations regarding their care and “underscores the need for continuous dialogue and reassessment of care goals throughout the progression of illness,” Dr. Ozdemir said.
“This stability in preferences over time suggests that initial care decisions are deeply ingrained or that there may be a lack of ongoing communication about evolving care needs and possibilities as conditions change,” Ozdemir said.
Yet, it can be hard to define what unrealistic expectations mean, said Olivia Seecof, MD, who wasn’t involved in the study.
“I think people are hopeful that a devastating diagnosis won’t lead to the end of their life and that there will be a treatment or something that will change [their prognosis], and they’ll get better,” said Dr. Seecof, palliative care expert with the Supportive Oncology Program at NYU Langone Health’s Perlmutter Cancer Center in New York City.
Giving patients and caregivers a realistic understanding of the prognosis is important, but “there’s more to it than just telling the patient their diagnosis,” she said.
“We have to plan for end of life, what it can look like,” said Dr. Seecof, adding that “often we don’t do a very good job of talking about that early on in an illness course.”
Overall, though, Dr. Seecof stressed that no two patients or situations are the same, and it’s important to understand what’s important in each scenario. End-of-life care requires “an individual approach because every patient is different, even if they have the same diagnosis as someone else,” she said.
This work was supported by funding from the Singapore Millennium Foundation and the Lien Centre for Palliative Care. Dr. Ozdemir and Dr. Seecof had no relevant disclosures.
A version of this article appeared on Medscape.com.
New research found that patients and caregivers both tend to prioritize symptom control over life extension but often preferring a balance. Patients and caregivers, however, are less aligned on decisions about cost containment, with patients more likely to prioritize cost containment.
“Our research has revealed that patients and caregivers generally share similar end-of-life goals,” with a “notable exception” when it comes to costs, first author Semra Ozdemir, PhD, with the Lien Centre for Palliative Care, Duke-NUS Medical School, Singapore, told this news organization.
However, when patients and caregivers have a better understanding of the patient’s prognosis, both may be more inclined to avoid costly life-extending treatments and prioritize symptom management.
In other words, the survey suggests that “knowing the prognosis helps patients and their families set realistic expectations for care and adequately prepare for end-of-life decisions,” said Dr. Ozdemir.
This study was published online in JAMA Network Open.
Patients with advanced cancer often face difficult decisions: Do they opt for treatments that may — or may not — extend life or do they focus more on symptom control?
Family caregivers, who also play an important role in this decision-making process, may have different care goals. Some research suggests that caregivers tend to prioritize treatments that could extend life, whereas patients prioritize symptom management, but it’s less clear how these priorities may change over time and how patients and caregivers may influence each other.
In the current study, the researchers examined goals of care among patients with stage IV solid tumors and caregivers during the last 2 years of life, focusing on life extension vs symptom management and cost containment, as well as how these goals changed over time.
The survey included 210 patient-caregiver pairs, recruited from outpatient clinics at two major cancer centers in Singapore. Patients had a mean age of 63 years, and about half were men. The caregivers had a mean age of 49 years, and almost two third (63%) were women.
Overall, 34% patients and 29% caregivers prioritized symptom management over life extension, whereas 24% patients and 19% caregivers prioritized life extension. Most patients and caregivers preferred balancing the two, with 34%-47% patients and 37%-69% caregivers supporting this approach.
When balancing cost and treatment decisions, however, patients were more likely to prioritize containing costs — 28% vs 17% for caregivers — over extending life — 26% of patients vs 35% of caregivers.
Cost containment tended to be more of a priority for older patients, those with a higher symptom burden, and those with less family caregiver support. For caregivers, cost containment was more of a priority for those who reported that caregiving had a big impact on their finances, those with worse self-esteem related to their caregiving abilities, as well as those caring for older patients.
To better align cost containment priorities between patients and caregivers, it’s essential for families to engage in open and thorough discussions about the allocation of resources, Dr. Ozdemir said.
Although “patients, families, and physicians often avoid discussions about prognosis,” such conversations are essential for setting realistic expectations for care and adequately preparing for end-of-life decisions, Dr. Ozdemir told this news organization.
“These conversations should aim to balance competing interests and create care plans that are mutually acceptable to both patients and caregivers,” she said, adding that “this approach will help in minimizing any potential conflicts and ensure that both parties feel respected and understood in their decision-making process.”
Managing Unrealistic Expectations
As patients approached the end of life, neither patients nor caregivers shifted their priorities from life extension to symptom management.
This finding raises concerns because it suggests that many patients hold unrealistic expectations regarding their care and “underscores the need for continuous dialogue and reassessment of care goals throughout the progression of illness,” Dr. Ozdemir said.
“This stability in preferences over time suggests that initial care decisions are deeply ingrained or that there may be a lack of ongoing communication about evolving care needs and possibilities as conditions change,” Ozdemir said.
Yet, it can be hard to define what unrealistic expectations mean, said Olivia Seecof, MD, who wasn’t involved in the study.
“I think people are hopeful that a devastating diagnosis won’t lead to the end of their life and that there will be a treatment or something that will change [their prognosis], and they’ll get better,” said Dr. Seecof, palliative care expert with the Supportive Oncology Program at NYU Langone Health’s Perlmutter Cancer Center in New York City.
Giving patients and caregivers a realistic understanding of the prognosis is important, but “there’s more to it than just telling the patient their diagnosis,” she said.
“We have to plan for end of life, what it can look like,” said Dr. Seecof, adding that “often we don’t do a very good job of talking about that early on in an illness course.”
Overall, though, Dr. Seecof stressed that no two patients or situations are the same, and it’s important to understand what’s important in each scenario. End-of-life care requires “an individual approach because every patient is different, even if they have the same diagnosis as someone else,” she said.
This work was supported by funding from the Singapore Millennium Foundation and the Lien Centre for Palliative Care. Dr. Ozdemir and Dr. Seecof had no relevant disclosures.
A version of this article appeared on Medscape.com.
Multiple Asymptomatic Dome-Shaped Papules on the Scalp
The Diagnosis: Spiradenocylindroma
Shave biopsies of our patient’s lesions showed wellcircumscribed dermal nodules resembling a spiradenoma with 3 cell populations: those with lighter nuclei, darker nuclei, and scattered lymphocytes. However, the conspicuous globules of basement membrane material were reminiscent of a cylindroma. These overlapping features and the patient’s history of cylindroma were suggestive of a diagnosis of spiradenocylindroma.
Spiradenocylindroma is an uncommon dermal tumor with features that overlap with spiradenoma and cylindroma.1 It may manifest as a solitary lesion or multiple lesions and can occur sporadically or in the context of a family history. Histologically, it must be distinguished from other intradermal basaloid neoplasms including conventional cylindroma and spiradenoma, dermal duct tumor, hidradenoma, and trichoblastoma.
When patients present with multiple cylindromas, spiradenomas, or spiradenocylindromas, physicians should consider genetic testing and review of the family history to assess for cylindromatosis gene mutations or Brooke-Spiegler syndrome. Biopsy and histologic examination are important because malignant tumors can evolve from pre-existing spiradenocylindromas, cylindromas, and spiradenomas,2 with an increased risk in patients with Brooke-Spiegler syndrome.1 Our patient declined further genetic workup but continues to follow up with dermatology for monitoring of lesions.
Dermal duct tumors are morphologic variants of poromas that are derived from sweat gland lineage and usually manifest as solitary dome-shaped papules, plaques, or nodules most often seen on acral surfaces as well as the head and neck.3 Clinically, they may be indistinguishable from spiradenocylindromas and require biopsy for histologic evaluation. They can be distinguished from spiradenocylindroma by the presence of small dermal nodules composed of cuboidal cells with ample pink cytoplasm and cuticle-lined ducts (Figure 1).
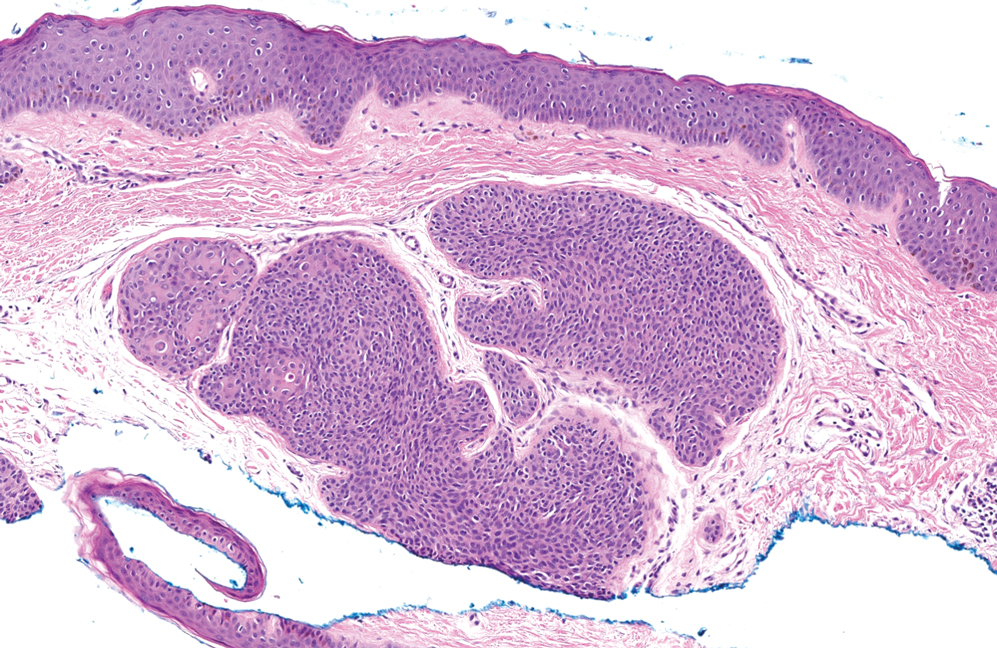
Trichoblastomas typically are deep-seated basaloid follicular neoplasms on the scalp with papillary mesenchyme resembling the normal fibrous sheath of the hair follicle, often replete with papillary mesenchymal bodies (Figure 2). There generally are no retraction spaces between its basaloid nests and the surrounding stroma, which is unlikely to contain mucin relative to basal cell carcinoma (BCC).4,5
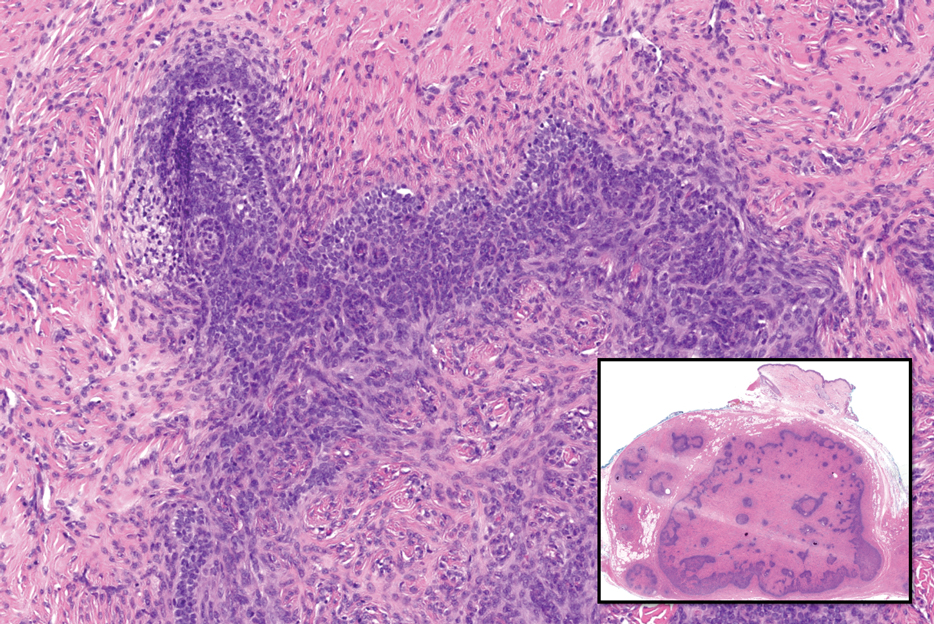
Adenoid cystic carcinoma is a rare salivary gland tumor that can metastasize to the skin and rarely arises as a primary skin adnexal tumor. It manifests as a slowgrowing mass that can be tender to palpation.6 Histologic examination shows dermal islands with cribriform blue and pink spaces. Compared to BCC, adenoid cystic carcinoma cells are enlarged and epithelioid with relatively scarce cytoplasm (Figure 3).6,7 Adenoid cystic carcinoma can show variable growth patterns including infiltrative nests and trabeculae. Perineural invasion is common, and there is a high risk for local recurrence.7 First-line therapy usually is surgical, and postoperative radiotherapy may be required.6,7
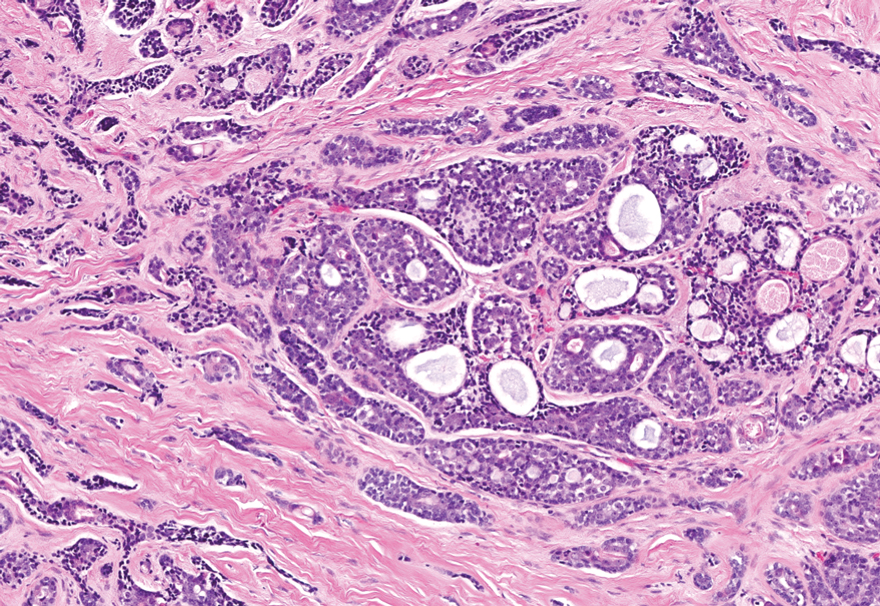
Nodular BCC commonly manifests as an enlarging nonhealing lesion on sun-exposed skin and has many subtypes, typically with arborizing telangiectases on dermoscopy. Histopathologic examination of nodular BCC reveals a nest of basaloid follicular germinative cells in the dermis with peripheral palisading and a fibromyxoid stroma (Figure 4).8 Patients with Brooke-Spiegler syndrome are at increased risk for nodular BCC, which may be clinically indistinguishable from spiradenoma, cylindroma, and spiradenocylindroma, necessitating histologic assessment.
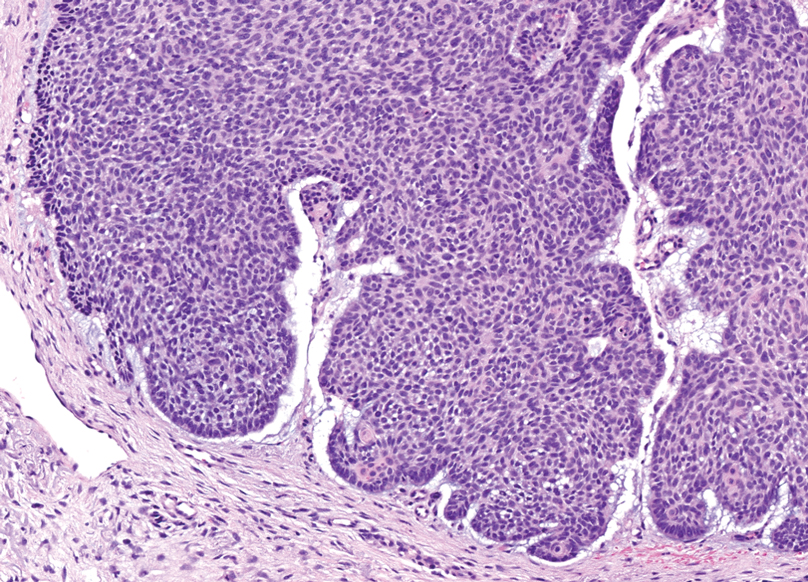
- Facchini V, Colangeli W, Bozza F, et al. A rare histopathological spiradenocylindroma: a case report. Clin Ter. 2022;173:292-294. doi:10.7417/ CT.2022.2433
- Kazakov DV. Brooke-Spiegler syndrome and phenotypic variants: an update [published online March 14, 2016]. Head Neck Pathol. 2016;10:125-30. doi:10.1007/s12105-016-0705-x
- Miller AC, Adjei S, Temiz LA, et al. Dermal duct tumor: a diagnostic dilemma. Dermatopathology (Basel). 2022;9:36-47. doi:10.3390/dermatopathology9010007
- Elston DM. Pilar and sebaceous neoplasms. In: Elston DM, Ferringer T, Ko C, et al. Dermatopathology. 3rd ed. Elsevier; 2018:71-85.
- McCalmont TH, Pincus LB. Adnexal neoplasms. In: Bolognia J, Schaffer J, Cerroni, L. Dermatology. 4th ed. Elsevier; 2017:1930-1953.
- Coca-Pelaz A, Rodrigo JP, Bradley PJ, et al. Adenoid cystic carcinoma of the head and neck—an update [published online May 2, 2015]. Oral Oncol. 2015;51:652-661. doi:10.1016/j.oraloncology.2015.04.005
- Tonev ID, Pirgova YS, Conev NV. Primary adenoid cystic carcinoma of the skin with multiple local recurrences. Case Rep Oncol. 2015;8:251- 255. doi:10.1159/000431082
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations [published online May 18, 2018]. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
The Diagnosis: Spiradenocylindroma
Shave biopsies of our patient’s lesions showed wellcircumscribed dermal nodules resembling a spiradenoma with 3 cell populations: those with lighter nuclei, darker nuclei, and scattered lymphocytes. However, the conspicuous globules of basement membrane material were reminiscent of a cylindroma. These overlapping features and the patient’s history of cylindroma were suggestive of a diagnosis of spiradenocylindroma.
Spiradenocylindroma is an uncommon dermal tumor with features that overlap with spiradenoma and cylindroma.1 It may manifest as a solitary lesion or multiple lesions and can occur sporadically or in the context of a family history. Histologically, it must be distinguished from other intradermal basaloid neoplasms including conventional cylindroma and spiradenoma, dermal duct tumor, hidradenoma, and trichoblastoma.
When patients present with multiple cylindromas, spiradenomas, or spiradenocylindromas, physicians should consider genetic testing and review of the family history to assess for cylindromatosis gene mutations or Brooke-Spiegler syndrome. Biopsy and histologic examination are important because malignant tumors can evolve from pre-existing spiradenocylindromas, cylindromas, and spiradenomas,2 with an increased risk in patients with Brooke-Spiegler syndrome.1 Our patient declined further genetic workup but continues to follow up with dermatology for monitoring of lesions.
Dermal duct tumors are morphologic variants of poromas that are derived from sweat gland lineage and usually manifest as solitary dome-shaped papules, plaques, or nodules most often seen on acral surfaces as well as the head and neck.3 Clinically, they may be indistinguishable from spiradenocylindromas and require biopsy for histologic evaluation. They can be distinguished from spiradenocylindroma by the presence of small dermal nodules composed of cuboidal cells with ample pink cytoplasm and cuticle-lined ducts (Figure 1).

Trichoblastomas typically are deep-seated basaloid follicular neoplasms on the scalp with papillary mesenchyme resembling the normal fibrous sheath of the hair follicle, often replete with papillary mesenchymal bodies (Figure 2). There generally are no retraction spaces between its basaloid nests and the surrounding stroma, which is unlikely to contain mucin relative to basal cell carcinoma (BCC).4,5

Adenoid cystic carcinoma is a rare salivary gland tumor that can metastasize to the skin and rarely arises as a primary skin adnexal tumor. It manifests as a slowgrowing mass that can be tender to palpation.6 Histologic examination shows dermal islands with cribriform blue and pink spaces. Compared to BCC, adenoid cystic carcinoma cells are enlarged and epithelioid with relatively scarce cytoplasm (Figure 3).6,7 Adenoid cystic carcinoma can show variable growth patterns including infiltrative nests and trabeculae. Perineural invasion is common, and there is a high risk for local recurrence.7 First-line therapy usually is surgical, and postoperative radiotherapy may be required.6,7

Nodular BCC commonly manifests as an enlarging nonhealing lesion on sun-exposed skin and has many subtypes, typically with arborizing telangiectases on dermoscopy. Histopathologic examination of nodular BCC reveals a nest of basaloid follicular germinative cells in the dermis with peripheral palisading and a fibromyxoid stroma (Figure 4).8 Patients with Brooke-Spiegler syndrome are at increased risk for nodular BCC, which may be clinically indistinguishable from spiradenoma, cylindroma, and spiradenocylindroma, necessitating histologic assessment.

The Diagnosis: Spiradenocylindroma
Shave biopsies of our patient’s lesions showed wellcircumscribed dermal nodules resembling a spiradenoma with 3 cell populations: those with lighter nuclei, darker nuclei, and scattered lymphocytes. However, the conspicuous globules of basement membrane material were reminiscent of a cylindroma. These overlapping features and the patient’s history of cylindroma were suggestive of a diagnosis of spiradenocylindroma.
Spiradenocylindroma is an uncommon dermal tumor with features that overlap with spiradenoma and cylindroma.1 It may manifest as a solitary lesion or multiple lesions and can occur sporadically or in the context of a family history. Histologically, it must be distinguished from other intradermal basaloid neoplasms including conventional cylindroma and spiradenoma, dermal duct tumor, hidradenoma, and trichoblastoma.
When patients present with multiple cylindromas, spiradenomas, or spiradenocylindromas, physicians should consider genetic testing and review of the family history to assess for cylindromatosis gene mutations or Brooke-Spiegler syndrome. Biopsy and histologic examination are important because malignant tumors can evolve from pre-existing spiradenocylindromas, cylindromas, and spiradenomas,2 with an increased risk in patients with Brooke-Spiegler syndrome.1 Our patient declined further genetic workup but continues to follow up with dermatology for monitoring of lesions.
Dermal duct tumors are morphologic variants of poromas that are derived from sweat gland lineage and usually manifest as solitary dome-shaped papules, plaques, or nodules most often seen on acral surfaces as well as the head and neck.3 Clinically, they may be indistinguishable from spiradenocylindromas and require biopsy for histologic evaluation. They can be distinguished from spiradenocylindroma by the presence of small dermal nodules composed of cuboidal cells with ample pink cytoplasm and cuticle-lined ducts (Figure 1).

Trichoblastomas typically are deep-seated basaloid follicular neoplasms on the scalp with papillary mesenchyme resembling the normal fibrous sheath of the hair follicle, often replete with papillary mesenchymal bodies (Figure 2). There generally are no retraction spaces between its basaloid nests and the surrounding stroma, which is unlikely to contain mucin relative to basal cell carcinoma (BCC).4,5

Adenoid cystic carcinoma is a rare salivary gland tumor that can metastasize to the skin and rarely arises as a primary skin adnexal tumor. It manifests as a slowgrowing mass that can be tender to palpation.6 Histologic examination shows dermal islands with cribriform blue and pink spaces. Compared to BCC, adenoid cystic carcinoma cells are enlarged and epithelioid with relatively scarce cytoplasm (Figure 3).6,7 Adenoid cystic carcinoma can show variable growth patterns including infiltrative nests and trabeculae. Perineural invasion is common, and there is a high risk for local recurrence.7 First-line therapy usually is surgical, and postoperative radiotherapy may be required.6,7

Nodular BCC commonly manifests as an enlarging nonhealing lesion on sun-exposed skin and has many subtypes, typically with arborizing telangiectases on dermoscopy. Histopathologic examination of nodular BCC reveals a nest of basaloid follicular germinative cells in the dermis with peripheral palisading and a fibromyxoid stroma (Figure 4).8 Patients with Brooke-Spiegler syndrome are at increased risk for nodular BCC, which may be clinically indistinguishable from spiradenoma, cylindroma, and spiradenocylindroma, necessitating histologic assessment.

- Facchini V, Colangeli W, Bozza F, et al. A rare histopathological spiradenocylindroma: a case report. Clin Ter. 2022;173:292-294. doi:10.7417/ CT.2022.2433
- Kazakov DV. Brooke-Spiegler syndrome and phenotypic variants: an update [published online March 14, 2016]. Head Neck Pathol. 2016;10:125-30. doi:10.1007/s12105-016-0705-x
- Miller AC, Adjei S, Temiz LA, et al. Dermal duct tumor: a diagnostic dilemma. Dermatopathology (Basel). 2022;9:36-47. doi:10.3390/dermatopathology9010007
- Elston DM. Pilar and sebaceous neoplasms. In: Elston DM, Ferringer T, Ko C, et al. Dermatopathology. 3rd ed. Elsevier; 2018:71-85.
- McCalmont TH, Pincus LB. Adnexal neoplasms. In: Bolognia J, Schaffer J, Cerroni, L. Dermatology. 4th ed. Elsevier; 2017:1930-1953.
- Coca-Pelaz A, Rodrigo JP, Bradley PJ, et al. Adenoid cystic carcinoma of the head and neck—an update [published online May 2, 2015]. Oral Oncol. 2015;51:652-661. doi:10.1016/j.oraloncology.2015.04.005
- Tonev ID, Pirgova YS, Conev NV. Primary adenoid cystic carcinoma of the skin with multiple local recurrences. Case Rep Oncol. 2015;8:251- 255. doi:10.1159/000431082
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations [published online May 18, 2018]. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Facchini V, Colangeli W, Bozza F, et al. A rare histopathological spiradenocylindroma: a case report. Clin Ter. 2022;173:292-294. doi:10.7417/ CT.2022.2433
- Kazakov DV. Brooke-Spiegler syndrome and phenotypic variants: an update [published online March 14, 2016]. Head Neck Pathol. 2016;10:125-30. doi:10.1007/s12105-016-0705-x
- Miller AC, Adjei S, Temiz LA, et al. Dermal duct tumor: a diagnostic dilemma. Dermatopathology (Basel). 2022;9:36-47. doi:10.3390/dermatopathology9010007
- Elston DM. Pilar and sebaceous neoplasms. In: Elston DM, Ferringer T, Ko C, et al. Dermatopathology. 3rd ed. Elsevier; 2018:71-85.
- McCalmont TH, Pincus LB. Adnexal neoplasms. In: Bolognia J, Schaffer J, Cerroni, L. Dermatology. 4th ed. Elsevier; 2017:1930-1953.
- Coca-Pelaz A, Rodrigo JP, Bradley PJ, et al. Adenoid cystic carcinoma of the head and neck—an update [published online May 2, 2015]. Oral Oncol. 2015;51:652-661. doi:10.1016/j.oraloncology.2015.04.005
- Tonev ID, Pirgova YS, Conev NV. Primary adenoid cystic carcinoma of the skin with multiple local recurrences. Case Rep Oncol. 2015;8:251- 255. doi:10.1159/000431082
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations [published online May 18, 2018]. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
A 62-year-old man with a history of cylindromas presented to our clinic with multiple asymptomatic, 3- to 4-mm, nonmobile, dome-shaped, telangiectatic, pink papules over the parietal and vertex scalp that had been present for more than 10 years without change. Several family members had similar lesions that had not been evaluated by a physician, and there had been no genetic evaluation. Shave biopsies of several lesions were performed.
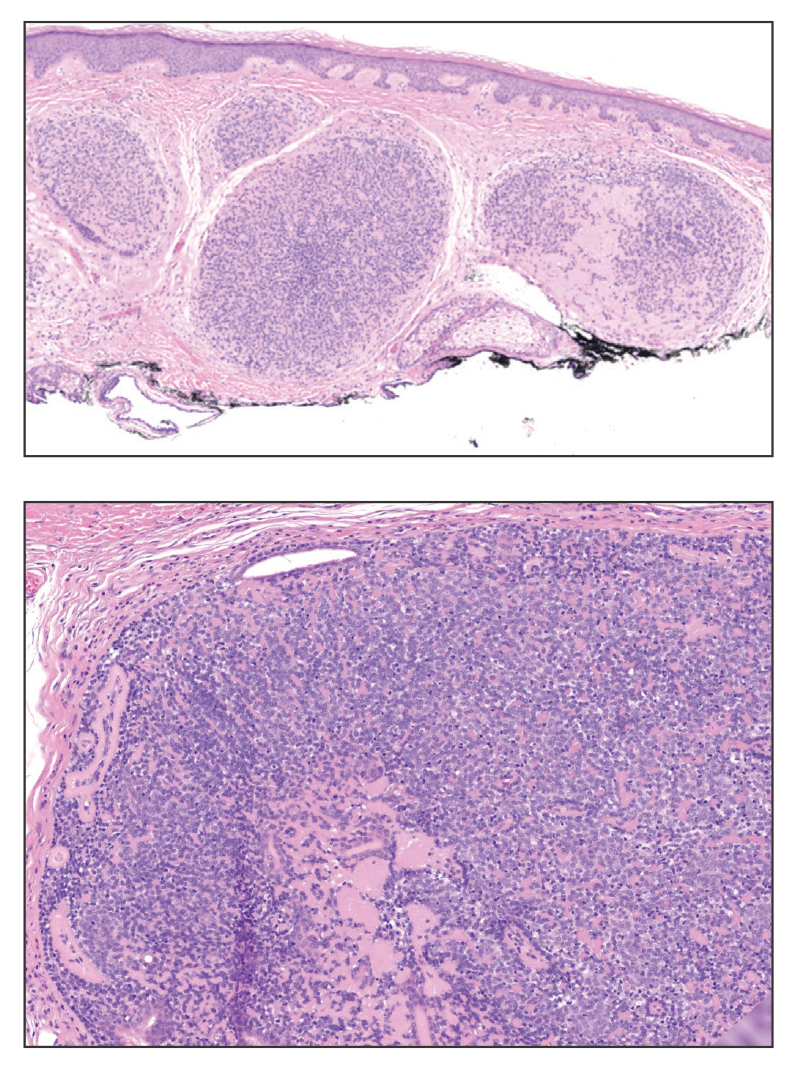
Impact of the COVID-19 Pandemic on Care for Patients With Skin Cancer
To the Editor:
The most common malignancy in the United States is skin cancer, with melanoma accounting for the majority of skin cancer deaths.1 Despite the lack of established guidelines for routine total-body skin examinations, many patients regularly visit their dermatologist for assessment of pigmented skin lesions.2 During the COVID-19 pandemic, many patients were unable to attend in-person dermatology visits, which resulted in many high-risk individuals not receiving care or alternatively seeking virtual care for cutaneous lesions.3 There has been a lack of research in the United States exploring the utilization of teledermatology during the pandemic and its overall impact on the care of patients with a history of skin cancer. We explored the impact of the COVID-19 pandemic on care for patients with skin cancer in a large US population.
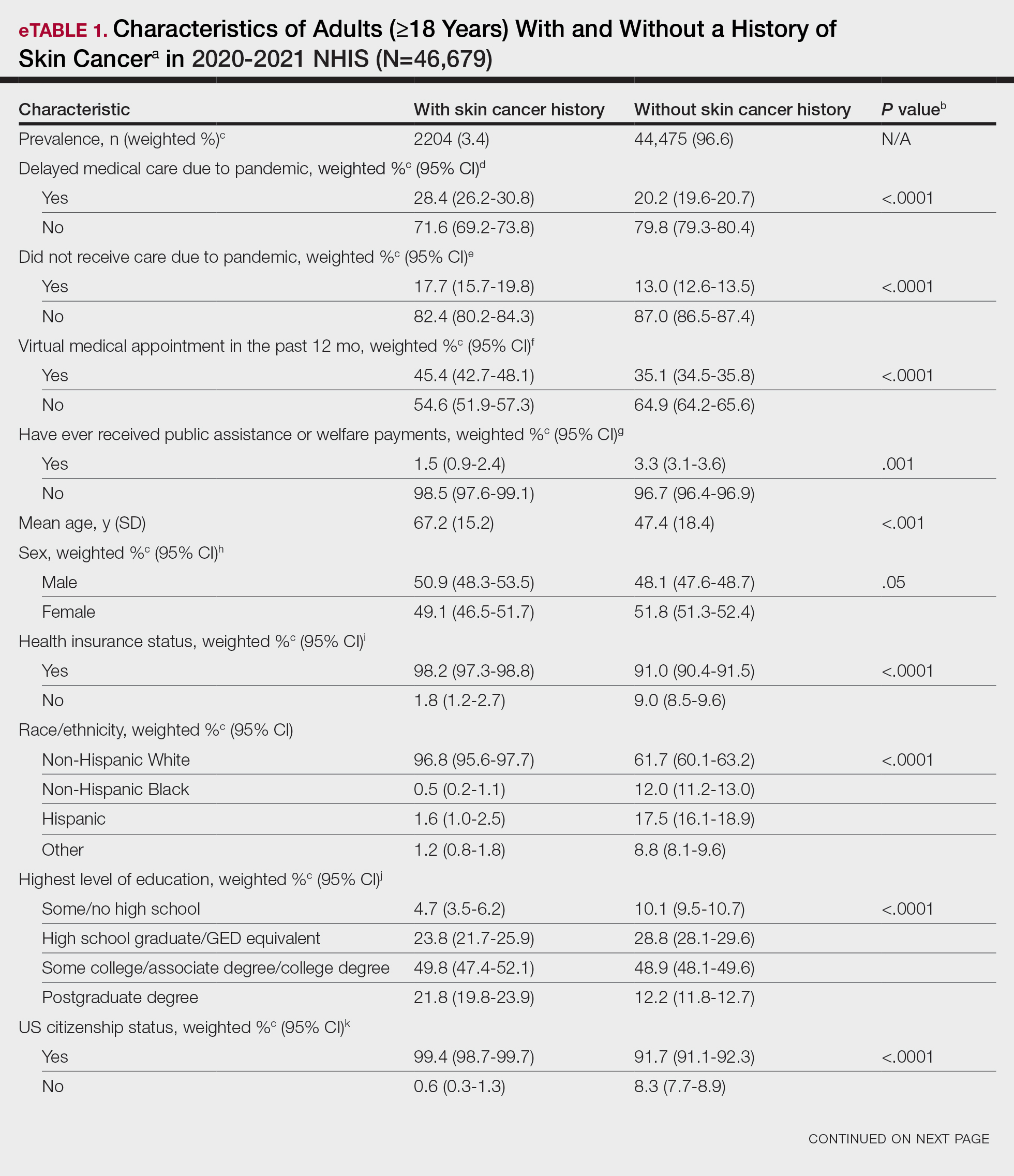

Using anonymous survey data from the 2020-2021 National Health Interview Survey,4 we conducted a population-based, cross-sectional study to evaluate access to care during the COVID-19 pandemic for patients with a self-reported history of skin cancer—melanoma, nonmelanoma skin cancer, or unknown skin cancer. The 3 outcome variables included having a virtual medical appointment in the past 12 months (yes/no), delaying medical care due to the COVID-19 pandemic (yes/no), and not receiving care due to the COVID-19 pandemic (yes/no). Multivariable logistic regression models evaluating the relationship between a history of skin cancer and access to care were constructed using Stata/MP 17.0 (StataCorp LLC). We controlled for patient age; education; race/ethnicity; received public assistance or welfare payments; sex; region; US citizenship status; health insurance status; comorbidities including history of hypertension, diabetes, and hypercholesterolemia; and birthplace in the United States in the logistic regression models.
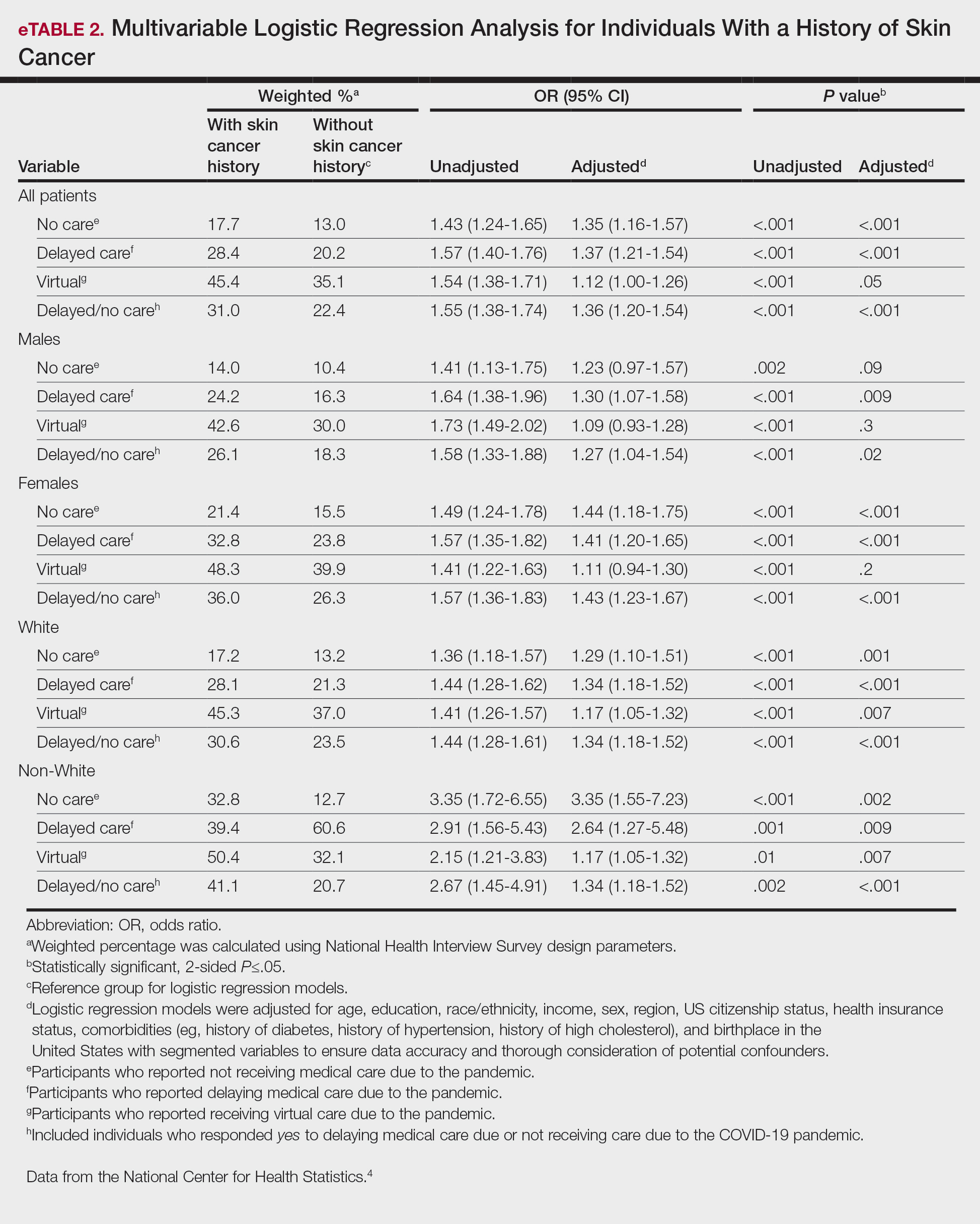
Our analysis included 46,679 patients aged 18 years or older, of whom 3.4% (weighted)(n=2204) reported a history of skin cancer (eTable 1). The weighted percentage was calculated using National Health Interview Survey design parameters (accounting for the multistage sampling design) to represent the general US population. Compared with those with no history of skin cancer, patients with a history of skin cancer were significantly more likely to delay medical care (adjusted odds ratio [AOR], 1.37; 95% CI, 1.21-1.54; P<.001) or not receive care (AOR, 1.35; 95% CI, 1.16-1.57; P<.001) due to the pandemic and were more likely to have had a virtual medical visit in the past 12 months (AOR, 1.12; 95% CI, 1.00-1.26; P=.05). Additionally, subgroup analysis revealed that females were more likely than males to forego medical care (eTable 2). β Coefficients for independent and dependent variables were further analyzed using logistic regression (eTable 3).
After adjusting for various potential confounders including comorbidities, our results revealed that patients with a history of skin cancer reported that they were less likely to receive in-person medical care due to the COVID-19 pandemic, as high-risk individuals with a history of skin cancer may have stopped receiving total-body skin examinations and dermatology care during the pandemic. Our findings showed that patients with a history of skin cancer were more likely than those without skin cancer to delay or forego care due to the pandemic, which may contribute to a higher incidence of advanced-stage melanomas postpandemic. Trepanowski et al5 reported an increased incidence of patients presenting with more advanced melanomas during the pandemic. Telemedicine was more commonly utilized by patients with a history of skin cancer during the pandemic.
In the future, virtual care may help limit advanced stages of skin cancer by serving as a viable alternative to in-person care.6 It has been reported that telemedicine can serve as a useful triage service reducing patient wait times.7 Teledermatology should not replace in-person care, as there is no evidence of the diagnostic accuracy of this service and many patients still will need to be seen in-person for confirmation of their diagnosis and potential biopsy. Further studies are needed to assess for missed skin cancer diagnoses due to the utilization of telemedicine.
Limitations of this study included a self-reported history of skin cancer, β coefficients that may suggest a high degree of collinearity, and lack of specific survey questions regarding dermatologic care during the COVID-19 pandemic. Further long-term studies exploring the clinical applicability and diagnostic accuracy of virtual medicine visits for cutaneous malignancies are vital, as teledermatology may play an essential role in curbing rising skin cancer rates even beyond the pandemic.
- Guy GP Jr, Thomas CC, Thompson T, et al. Vital signs: melanoma incidence and mortality trends and projections—United States, 1982-2030. MMWR Morb Mortal Wkly Rep. 2015;64:591-596.
- Whiteman DC, Olsen CM, MacGregor S, et al; QSkin Study. The effect of screening on melanoma incidence and biopsy rates. Br J Dermatol. 2022;187:515-522. doi:10.1111/bjd.21649
- Jobbágy A, Kiss N, Meznerics FA, et al. Emergency use and efficacy of an asynchronous teledermatology system as a novel tool for early diagnosis of skin cancer during the first wave of COVID-19 pandemic. Int J Environ Res Public Health. 2022;19:2699. doi:10.3390/ijerph19052699
- National Center for Health Statistics. NHIS Data, Questionnaires and Related Documentation. Centers for Disease Control and Prevention website. Accessed April 19, 2023. https://www.cdc.gov/nchs/nhis/data-questionnaires-documentation.htm
- Trepanowski N, Chang MS, Zhou G, et al. Delays in melanoma presentation during the COVID-19 pandemic: a nationwide multi-institutional cohort study. J Am Acad Dermatol. 2022;87:1217-1219. doi:10.1016/j.jaad.2022.06.031
- Chiru MR, Hindocha S, Burova E, et al. Management of the two-week wait pathway for skin cancer patients, before and during the pandemic: is virtual consultation an option? J Pers Med. 2022;12:1258. doi:10.3390/jpm12081258
- Finnane A Dallest K Janda M et al. Teledermatology for the diagnosis and management of skin cancer: a systematic review. JAMA Dermatol. 2017;153:319-327. doi:10.1001/jamadermatol.2016.4361
To the Editor:
The most common malignancy in the United States is skin cancer, with melanoma accounting for the majority of skin cancer deaths.1 Despite the lack of established guidelines for routine total-body skin examinations, many patients regularly visit their dermatologist for assessment of pigmented skin lesions.2 During the COVID-19 pandemic, many patients were unable to attend in-person dermatology visits, which resulted in many high-risk individuals not receiving care or alternatively seeking virtual care for cutaneous lesions.3 There has been a lack of research in the United States exploring the utilization of teledermatology during the pandemic and its overall impact on the care of patients with a history of skin cancer. We explored the impact of the COVID-19 pandemic on care for patients with skin cancer in a large US population.


Using anonymous survey data from the 2020-2021 National Health Interview Survey,4 we conducted a population-based, cross-sectional study to evaluate access to care during the COVID-19 pandemic for patients with a self-reported history of skin cancer—melanoma, nonmelanoma skin cancer, or unknown skin cancer. The 3 outcome variables included having a virtual medical appointment in the past 12 months (yes/no), delaying medical care due to the COVID-19 pandemic (yes/no), and not receiving care due to the COVID-19 pandemic (yes/no). Multivariable logistic regression models evaluating the relationship between a history of skin cancer and access to care were constructed using Stata/MP 17.0 (StataCorp LLC). We controlled for patient age; education; race/ethnicity; received public assistance or welfare payments; sex; region; US citizenship status; health insurance status; comorbidities including history of hypertension, diabetes, and hypercholesterolemia; and birthplace in the United States in the logistic regression models.

Our analysis included 46,679 patients aged 18 years or older, of whom 3.4% (weighted)(n=2204) reported a history of skin cancer (eTable 1). The weighted percentage was calculated using National Health Interview Survey design parameters (accounting for the multistage sampling design) to represent the general US population. Compared with those with no history of skin cancer, patients with a history of skin cancer were significantly more likely to delay medical care (adjusted odds ratio [AOR], 1.37; 95% CI, 1.21-1.54; P<.001) or not receive care (AOR, 1.35; 95% CI, 1.16-1.57; P<.001) due to the pandemic and were more likely to have had a virtual medical visit in the past 12 months (AOR, 1.12; 95% CI, 1.00-1.26; P=.05). Additionally, subgroup analysis revealed that females were more likely than males to forego medical care (eTable 2). β Coefficients for independent and dependent variables were further analyzed using logistic regression (eTable 3).

After adjusting for various potential confounders including comorbidities, our results revealed that patients with a history of skin cancer reported that they were less likely to receive in-person medical care due to the COVID-19 pandemic, as high-risk individuals with a history of skin cancer may have stopped receiving total-body skin examinations and dermatology care during the pandemic. Our findings showed that patients with a history of skin cancer were more likely than those without skin cancer to delay or forego care due to the pandemic, which may contribute to a higher incidence of advanced-stage melanomas postpandemic. Trepanowski et al5 reported an increased incidence of patients presenting with more advanced melanomas during the pandemic. Telemedicine was more commonly utilized by patients with a history of skin cancer during the pandemic.
In the future, virtual care may help limit advanced stages of skin cancer by serving as a viable alternative to in-person care.6 It has been reported that telemedicine can serve as a useful triage service reducing patient wait times.7 Teledermatology should not replace in-person care, as there is no evidence of the diagnostic accuracy of this service and many patients still will need to be seen in-person for confirmation of their diagnosis and potential biopsy. Further studies are needed to assess for missed skin cancer diagnoses due to the utilization of telemedicine.
Limitations of this study included a self-reported history of skin cancer, β coefficients that may suggest a high degree of collinearity, and lack of specific survey questions regarding dermatologic care during the COVID-19 pandemic. Further long-term studies exploring the clinical applicability and diagnostic accuracy of virtual medicine visits for cutaneous malignancies are vital, as teledermatology may play an essential role in curbing rising skin cancer rates even beyond the pandemic.
To the Editor:
The most common malignancy in the United States is skin cancer, with melanoma accounting for the majority of skin cancer deaths.1 Despite the lack of established guidelines for routine total-body skin examinations, many patients regularly visit their dermatologist for assessment of pigmented skin lesions.2 During the COVID-19 pandemic, many patients were unable to attend in-person dermatology visits, which resulted in many high-risk individuals not receiving care or alternatively seeking virtual care for cutaneous lesions.3 There has been a lack of research in the United States exploring the utilization of teledermatology during the pandemic and its overall impact on the care of patients with a history of skin cancer. We explored the impact of the COVID-19 pandemic on care for patients with skin cancer in a large US population.


Using anonymous survey data from the 2020-2021 National Health Interview Survey,4 we conducted a population-based, cross-sectional study to evaluate access to care during the COVID-19 pandemic for patients with a self-reported history of skin cancer—melanoma, nonmelanoma skin cancer, or unknown skin cancer. The 3 outcome variables included having a virtual medical appointment in the past 12 months (yes/no), delaying medical care due to the COVID-19 pandemic (yes/no), and not receiving care due to the COVID-19 pandemic (yes/no). Multivariable logistic regression models evaluating the relationship between a history of skin cancer and access to care were constructed using Stata/MP 17.0 (StataCorp LLC). We controlled for patient age; education; race/ethnicity; received public assistance or welfare payments; sex; region; US citizenship status; health insurance status; comorbidities including history of hypertension, diabetes, and hypercholesterolemia; and birthplace in the United States in the logistic regression models.

Our analysis included 46,679 patients aged 18 years or older, of whom 3.4% (weighted)(n=2204) reported a history of skin cancer (eTable 1). The weighted percentage was calculated using National Health Interview Survey design parameters (accounting for the multistage sampling design) to represent the general US population. Compared with those with no history of skin cancer, patients with a history of skin cancer were significantly more likely to delay medical care (adjusted odds ratio [AOR], 1.37; 95% CI, 1.21-1.54; P<.001) or not receive care (AOR, 1.35; 95% CI, 1.16-1.57; P<.001) due to the pandemic and were more likely to have had a virtual medical visit in the past 12 months (AOR, 1.12; 95% CI, 1.00-1.26; P=.05). Additionally, subgroup analysis revealed that females were more likely than males to forego medical care (eTable 2). β Coefficients for independent and dependent variables were further analyzed using logistic regression (eTable 3).

After adjusting for various potential confounders including comorbidities, our results revealed that patients with a history of skin cancer reported that they were less likely to receive in-person medical care due to the COVID-19 pandemic, as high-risk individuals with a history of skin cancer may have stopped receiving total-body skin examinations and dermatology care during the pandemic. Our findings showed that patients with a history of skin cancer were more likely than those without skin cancer to delay or forego care due to the pandemic, which may contribute to a higher incidence of advanced-stage melanomas postpandemic. Trepanowski et al5 reported an increased incidence of patients presenting with more advanced melanomas during the pandemic. Telemedicine was more commonly utilized by patients with a history of skin cancer during the pandemic.
In the future, virtual care may help limit advanced stages of skin cancer by serving as a viable alternative to in-person care.6 It has been reported that telemedicine can serve as a useful triage service reducing patient wait times.7 Teledermatology should not replace in-person care, as there is no evidence of the diagnostic accuracy of this service and many patients still will need to be seen in-person for confirmation of their diagnosis and potential biopsy. Further studies are needed to assess for missed skin cancer diagnoses due to the utilization of telemedicine.
Limitations of this study included a self-reported history of skin cancer, β coefficients that may suggest a high degree of collinearity, and lack of specific survey questions regarding dermatologic care during the COVID-19 pandemic. Further long-term studies exploring the clinical applicability and diagnostic accuracy of virtual medicine visits for cutaneous malignancies are vital, as teledermatology may play an essential role in curbing rising skin cancer rates even beyond the pandemic.
- Guy GP Jr, Thomas CC, Thompson T, et al. Vital signs: melanoma incidence and mortality trends and projections—United States, 1982-2030. MMWR Morb Mortal Wkly Rep. 2015;64:591-596.
- Whiteman DC, Olsen CM, MacGregor S, et al; QSkin Study. The effect of screening on melanoma incidence and biopsy rates. Br J Dermatol. 2022;187:515-522. doi:10.1111/bjd.21649
- Jobbágy A, Kiss N, Meznerics FA, et al. Emergency use and efficacy of an asynchronous teledermatology system as a novel tool for early diagnosis of skin cancer during the first wave of COVID-19 pandemic. Int J Environ Res Public Health. 2022;19:2699. doi:10.3390/ijerph19052699
- National Center for Health Statistics. NHIS Data, Questionnaires and Related Documentation. Centers for Disease Control and Prevention website. Accessed April 19, 2023. https://www.cdc.gov/nchs/nhis/data-questionnaires-documentation.htm
- Trepanowski N, Chang MS, Zhou G, et al. Delays in melanoma presentation during the COVID-19 pandemic: a nationwide multi-institutional cohort study. J Am Acad Dermatol. 2022;87:1217-1219. doi:10.1016/j.jaad.2022.06.031
- Chiru MR, Hindocha S, Burova E, et al. Management of the two-week wait pathway for skin cancer patients, before and during the pandemic: is virtual consultation an option? J Pers Med. 2022;12:1258. doi:10.3390/jpm12081258
- Finnane A Dallest K Janda M et al. Teledermatology for the diagnosis and management of skin cancer: a systematic review. JAMA Dermatol. 2017;153:319-327. doi:10.1001/jamadermatol.2016.4361
- Guy GP Jr, Thomas CC, Thompson T, et al. Vital signs: melanoma incidence and mortality trends and projections—United States, 1982-2030. MMWR Morb Mortal Wkly Rep. 2015;64:591-596.
- Whiteman DC, Olsen CM, MacGregor S, et al; QSkin Study. The effect of screening on melanoma incidence and biopsy rates. Br J Dermatol. 2022;187:515-522. doi:10.1111/bjd.21649
- Jobbágy A, Kiss N, Meznerics FA, et al. Emergency use and efficacy of an asynchronous teledermatology system as a novel tool for early diagnosis of skin cancer during the first wave of COVID-19 pandemic. Int J Environ Res Public Health. 2022;19:2699. doi:10.3390/ijerph19052699
- National Center for Health Statistics. NHIS Data, Questionnaires and Related Documentation. Centers for Disease Control and Prevention website. Accessed April 19, 2023. https://www.cdc.gov/nchs/nhis/data-questionnaires-documentation.htm
- Trepanowski N, Chang MS, Zhou G, et al. Delays in melanoma presentation during the COVID-19 pandemic: a nationwide multi-institutional cohort study. J Am Acad Dermatol. 2022;87:1217-1219. doi:10.1016/j.jaad.2022.06.031
- Chiru MR, Hindocha S, Burova E, et al. Management of the two-week wait pathway for skin cancer patients, before and during the pandemic: is virtual consultation an option? J Pers Med. 2022;12:1258. doi:10.3390/jpm12081258
- Finnane A Dallest K Janda M et al. Teledermatology for the diagnosis and management of skin cancer: a systematic review. JAMA Dermatol. 2017;153:319-327. doi:10.1001/jamadermatol.2016.4361
PRACTICE POINTS
- The COVID-19 pandemic has altered the landscape of medicine, as many individuals are now utilizing telemedicine to receive care.
- Many individuals will continue to receive telemedicine moving forward, making it crucial to understand access to care.
Comment on “Skin Cancer Screening: The Paradox of Melanoma and Improved All-Cause Mortality”
To the Editor:
I was unsurprised and gratified by the information presented in the Viewpoint on skin cancer screening by Ngo1 (Cutis. 2024;113:94-96). In my 30 years as a community dermatologist, I have observed that patients who opt to have periodic full-body skin examinations usually are more health literate, more likely to have a primary care physician (PCP) who has encouraged them to do so (ie, a conscientious practitioner directing their preventive care), more likely to have a strong will to live, and less likely to have multiple stressors that preclude self-care (eg, may be less likely to have a spouse for whom they are a caregiver) compared to those who do not get screened.
Findings on a full-body skin examination may impact patients in many ways, not only by the detection of skin cancers. I have discovered the following:
- evidence of diabetes/insulin resistance in the form of acanthosis nigricans, tinea corporis, erythrasma;
- evidence of rosacea associated with excessive alcohol intake;
- evidence of smoking-related issues such as psoriasis or hidradenitis suppurativa;
- cutaneous evidence of other systemic diseases (eg, autoimmune disease, cancer);
- elucidation of other chronic health problems (eg, psoriasis of the skin as a clue for undiagnosed psoriatic arthritis); and
- detection of parasites on the skin (eg, ticks) or signs of infection that may have notable ramifications (eg, interdigital maceration of a diabetic patient with tinea pedis).
I even saw a patient who had been sent for magnetic resonance imaging for back pain by her internist without any physical examination when she actually had an erosion over the sacrum from a rug burn!
When conducting full-body skin examinations, dermatologists should not underestimate these principles:
- The “magic” of using a relatively noninvasive and sensitive screening tool—comfort and stress reduction for the patient from a thorough visual, tactile, olfactory, and auditory examination.
- Human interaction—especially when the patient is seen annually or even more frequently over a period of years or decades, and especially when an excellent patient-physician rapport has been established.
- The impact of improving a patient’s appearance on their overall sense of well-being (eg, by controlling rosacea).
- The opportunity to introduce concepts (ie, educate patients) such as alcohol avoidance, smoking cessation, weight reduction, hygiene, diet, and exercise in a more tangential way than a PCP, as well as to consider with patients the idea that lifestyle modification may be an adjunct, if not a replacement, for prescription treatments.
- The stress reduction that ensues when a variety of self-identified health issues are addressed, for which the only treatment may be reassurance.
I would add to Dr. Ngo’s argument that stratifying patients into skin cancer risk categories may be a useful measure if the only goal of periodic dermatologic evaluation is skin cancer detection. One size rarely fits all when it comes to health recommendations.
In sum, I believe that periodic full-body skin examination is absolutely beneficial to patient care, and I am not at all surprised that all-cause mortality was lower in patients who have those examinations. Furthermore, when I offer my healthy, low-risk patients the option to return in 2 years rather than 1, the vast majority insist on 1 year. My mother used to say, “It’s better to be looked over than to be overlooked,” and I tell my patients that, too—but it seems they already know that instinctively.
- Ngo BT. Skin cancer screening: the paradox of melanoma and improved all-cause mortality. Cutis. 2024;113:94-96. doi:10.12788/cutis.0948
To the Editor:
I was unsurprised and gratified by the information presented in the Viewpoint on skin cancer screening by Ngo1 (Cutis. 2024;113:94-96). In my 30 years as a community dermatologist, I have observed that patients who opt to have periodic full-body skin examinations usually are more health literate, more likely to have a primary care physician (PCP) who has encouraged them to do so (ie, a conscientious practitioner directing their preventive care), more likely to have a strong will to live, and less likely to have multiple stressors that preclude self-care (eg, may be less likely to have a spouse for whom they are a caregiver) compared to those who do not get screened.
Findings on a full-body skin examination may impact patients in many ways, not only by the detection of skin cancers. I have discovered the following:
- evidence of diabetes/insulin resistance in the form of acanthosis nigricans, tinea corporis, erythrasma;
- evidence of rosacea associated with excessive alcohol intake;
- evidence of smoking-related issues such as psoriasis or hidradenitis suppurativa;
- cutaneous evidence of other systemic diseases (eg, autoimmune disease, cancer);
- elucidation of other chronic health problems (eg, psoriasis of the skin as a clue for undiagnosed psoriatic arthritis); and
- detection of parasites on the skin (eg, ticks) or signs of infection that may have notable ramifications (eg, interdigital maceration of a diabetic patient with tinea pedis).
I even saw a patient who had been sent for magnetic resonance imaging for back pain by her internist without any physical examination when she actually had an erosion over the sacrum from a rug burn!
When conducting full-body skin examinations, dermatologists should not underestimate these principles:
- The “magic” of using a relatively noninvasive and sensitive screening tool—comfort and stress reduction for the patient from a thorough visual, tactile, olfactory, and auditory examination.
- Human interaction—especially when the patient is seen annually or even more frequently over a period of years or decades, and especially when an excellent patient-physician rapport has been established.
- The impact of improving a patient’s appearance on their overall sense of well-being (eg, by controlling rosacea).
- The opportunity to introduce concepts (ie, educate patients) such as alcohol avoidance, smoking cessation, weight reduction, hygiene, diet, and exercise in a more tangential way than a PCP, as well as to consider with patients the idea that lifestyle modification may be an adjunct, if not a replacement, for prescription treatments.
- The stress reduction that ensues when a variety of self-identified health issues are addressed, for which the only treatment may be reassurance.
I would add to Dr. Ngo’s argument that stratifying patients into skin cancer risk categories may be a useful measure if the only goal of periodic dermatologic evaluation is skin cancer detection. One size rarely fits all when it comes to health recommendations.
In sum, I believe that periodic full-body skin examination is absolutely beneficial to patient care, and I am not at all surprised that all-cause mortality was lower in patients who have those examinations. Furthermore, when I offer my healthy, low-risk patients the option to return in 2 years rather than 1, the vast majority insist on 1 year. My mother used to say, “It’s better to be looked over than to be overlooked,” and I tell my patients that, too—but it seems they already know that instinctively.
To the Editor:
I was unsurprised and gratified by the information presented in the Viewpoint on skin cancer screening by Ngo1 (Cutis. 2024;113:94-96). In my 30 years as a community dermatologist, I have observed that patients who opt to have periodic full-body skin examinations usually are more health literate, more likely to have a primary care physician (PCP) who has encouraged them to do so (ie, a conscientious practitioner directing their preventive care), more likely to have a strong will to live, and less likely to have multiple stressors that preclude self-care (eg, may be less likely to have a spouse for whom they are a caregiver) compared to those who do not get screened.
Findings on a full-body skin examination may impact patients in many ways, not only by the detection of skin cancers. I have discovered the following:
- evidence of diabetes/insulin resistance in the form of acanthosis nigricans, tinea corporis, erythrasma;
- evidence of rosacea associated with excessive alcohol intake;
- evidence of smoking-related issues such as psoriasis or hidradenitis suppurativa;
- cutaneous evidence of other systemic diseases (eg, autoimmune disease, cancer);
- elucidation of other chronic health problems (eg, psoriasis of the skin as a clue for undiagnosed psoriatic arthritis); and
- detection of parasites on the skin (eg, ticks) or signs of infection that may have notable ramifications (eg, interdigital maceration of a diabetic patient with tinea pedis).
I even saw a patient who had been sent for magnetic resonance imaging for back pain by her internist without any physical examination when she actually had an erosion over the sacrum from a rug burn!
When conducting full-body skin examinations, dermatologists should not underestimate these principles:
- The “magic” of using a relatively noninvasive and sensitive screening tool—comfort and stress reduction for the patient from a thorough visual, tactile, olfactory, and auditory examination.
- Human interaction—especially when the patient is seen annually or even more frequently over a period of years or decades, and especially when an excellent patient-physician rapport has been established.
- The impact of improving a patient’s appearance on their overall sense of well-being (eg, by controlling rosacea).
- The opportunity to introduce concepts (ie, educate patients) such as alcohol avoidance, smoking cessation, weight reduction, hygiene, diet, and exercise in a more tangential way than a PCP, as well as to consider with patients the idea that lifestyle modification may be an adjunct, if not a replacement, for prescription treatments.
- The stress reduction that ensues when a variety of self-identified health issues are addressed, for which the only treatment may be reassurance.
I would add to Dr. Ngo’s argument that stratifying patients into skin cancer risk categories may be a useful measure if the only goal of periodic dermatologic evaluation is skin cancer detection. One size rarely fits all when it comes to health recommendations.
In sum, I believe that periodic full-body skin examination is absolutely beneficial to patient care, and I am not at all surprised that all-cause mortality was lower in patients who have those examinations. Furthermore, when I offer my healthy, low-risk patients the option to return in 2 years rather than 1, the vast majority insist on 1 year. My mother used to say, “It’s better to be looked over than to be overlooked,” and I tell my patients that, too—but it seems they already know that instinctively.
- Ngo BT. Skin cancer screening: the paradox of melanoma and improved all-cause mortality. Cutis. 2024;113:94-96. doi:10.12788/cutis.0948
- Ngo BT. Skin cancer screening: the paradox of melanoma and improved all-cause mortality. Cutis. 2024;113:94-96. doi:10.12788/cutis.0948
The DEA Plans to Reschedule Marijuana: What Happens Next?
The US Drug Enforcement Agency (DEA) is moving forward with plans to move marijuana from a Schedule I to a Schedule III controlled substance under the Controlled Substance Act (CSA), the US Department of Justice officials announced this week.
First reported by the Associated Press and since confirmed by this news organization through a US Department of Justice spokesperson, the news made international headlines. Despite the media splash, the final rule is still months away.
How did we get here? What happens next? What impact might rescheduling have on clinicians, patients, researchers, and the medical cannabis industry?
Why Reschedule? Why Now?
The DEA’s decision is based on a 2023 determination from the US Food and Drug Administration (FDA) that marijuana has a legitimate medical use and should be moved to Schedule III.
This class includes ketamine, acetaminophen with codeine, and buprenorphine.
Even though the manufacturing, distribution, sale, and use of marijuana has long violated federal law, 38 states and Washington, DC, have legalized medical cannabis, and 24 states and DC have legalized its recreational use.
Congress has allowed states leeway for the distribution and use of medical marijuana, and current and previous presidential administrations have chosen not to aggressively pursue prosecution of state-allowed marijuana use, the Congressional Research Service (CRS) reports.
Pressure to address the conflict between federal and state laws and an increasing interest in drug development of cannabis and cannabis-derived products probably contributed to the DEA’s decision, said Stephen Strakowski, MD, professor, and vice chair of psychiatry at Indiana University in Indianapolis, and professor and associate vice president at University of Texas in Austin.
“The trend toward legalization is everywhere and even though nationally the feds in this instance are lagging the states, the pressure to legalize has been intense for 50 years and it’s not surprising that the DEA is finally following that lead,” Dr. Strakowski told this news organization.
How Does Rescheduling Work? What’s the Timeline?
The DEA will submit a formal rule proposing that marijuana be moved from Schedule I to Schedule III to the White House Office of Management and Budget. The timing of the submission is unclear.
Once the proposed rule is posted to the Federal Register, there will be a public comment period, which usually lasts 30-60 days.
“This will likely generate a lot of public comment,” Robert Mikos, JD, LaRoche Family Chair in Law at Vanderbilt University Law School in Nashville, Tennessee, told this news organization. “Then the agency has to go back and wade through those comments and decide if they want to proceed with the rule as proposed or modify it.”
A final rule will probably be posted before the end of the current presidential term in January, Mr. Mikos said. While a lawsuit blocking its implementation is possible, there is a “low chance that a court would block this,” he added.
How Will Rescheduling Affect Medical Marijuana?
For medical marijuana, changing the drug to a Schedule III means that it can legally be prescribed but only in states that have legalized medical cannabis, Mr. Mikos said.
“If you’re a patient in a state with a medical marijuana law and your physician gives you a prescription for medical marijuana and you possess it, you will no longer be guilty of a federal crime,” he said.
Rescheduling could also benefit patients who receive care through the Veterans Administration (VA), Mr. Mikos said. For several years, the VA has had a policy that blocked clinicians from prescribing medical marijuana because as a Schedule I drug, it was determined to have no accepted medical use.
“It’s possible the VA may drop that policy once the drug gets rescheduled. If you’re in a medical marijuana state, if you’re a VA patient, and you don’t want to spend the extra money to go outside that system, this will have meaningful impact on their lives,” Mr. Mikos said.
But what about patients living in states that have not legalized medical cannabis?
“You still wouldn’t be committing a federal crime, but you could be violating state law,” Mr. Mikos said. “That’s a much more salient consideration because if you look at who goes after individuals who possess small amounts of drugs, the state handles 99% of those cases.”
The manufacture, distribution, and possession of recreational marijuana would remain illegal under federal law.
What Does It Mean for Medical Marijuana Dispensaries?
Though rescheduling makes it legal for clinicians to prescribe medical marijuana and for patients to use it, the actual sale of the drug will remain illegal under federal law because rescheduling only changes prescribing under the CSA, Mr. Mikos said.
“If you’re a dispensary and you sell it, even if it’s to somebody who’s got a prescription, you’re still probably violating the Food, Drug and Cosmetics Act. Rescheduling doesn’t change that,” he said.
“Even assuming the DEA follows through with this and it doesn’t come undone at some future date, the industry is still going struggle to comply with the Controlled Substances Act post rescheduling because that statute is going to continue to impose a number of regulations on the industry,” Mr. Mikos added.
However, rescheduling would change the tax status of the estimated 12,000-15,000 state-licensed cannabis dispensaries in the United States, allowing access to certain tax deductions that are unavailable to sales involving Schedule I controlled substances, James Daily, JD, MS, with Center for Empirical Research in the Law at Washington University School of Law in St. Louis, told this news organization.
“Many cannabis businesses do in fact pay federal taxes, but the inability to take any federal tax credits or deductions means that their effective tax rate is much higher than it would otherwise be,” Mr. Daily said.
Although new federal tax deductions would likely available to cannabis businesses if marijuana were rescheduled to Schedule III, “their business would still be in violation of federal law,” he said.
“This creates a further tension between state and federal law, which could be resolved by further legalization or it could be resolved by extending the prohibition on tax deductions to include cannabis and not just Schedule I and II drugs,” he added.
Will Rescheduling Make It Easier to Conduct Cannabis-Related Research?
Research on medical cannabis has been stymied by FDA and DEA regulations regarding the study of Schedule I controlled substances. Although rescheduling could lift that barrier, other challenges would remain.
“Schedule III drugs can be more easily researched, but it’s unclear if, for example, a clinical trial could lawfully obtain the cannabis from a dispensary or if they would still have to go through the one legal federal supplier of cannabis,” Daily said.
The FDA reports having received more than 800 investigational new drug applications for and pre-investigational new drug applications related to cannabis and cannabis-derived products since the 1970s, the agency reports. To date, the FDA has not approved any marketing drug applications for cannabis for the treatment of any disease or condition.
In January 2023, the agency published updated guidelines for researchers and sponsors interested in developing drugs containing cannabis or cannabis-derived compounds.
It’s unclear whether those guidelines would be updated if the rescheduling moves forward.
Does Rescheduling Marijuana Pose Any Risk?
In its report to the DEA that marijuana be rescheduled, the FDA was careful to note that the agency’s recommendation is “not meant to imply that safety and effectiveness have been established for marijuana that would support FDA approval of a marijuana drug product for a particular indication.”
That’s a notation that clinicians and patients should take to heart, Dr. Strakowski said.
“It’s important to remind people that Schedule III drugs, by definition, have addiction and other side effect risks,” he said. “The celebrity marketing that sits behind a lot of this is incompletely informed. It’s portrayed as fun and harmless in almost every movie and conversation you see, and we know that’s not true.”
Previous studies have linked cannabis to increased risk for mania, anxiety disorders, and schizophrenia.
“It is increasingly clear that marijuana use is linked to poor outcomes in people who struggle with mental illness,” Dr. Strakowski said. “We have no evidence that it can help you but there is evidence that it can harm you.”
Dr. Strakowski likens cannabis use to alcohol, which is a known depressant that is associated with worse outcomes in people with mental illness.
“I think with cannabis, we don’t know enough about it yet, but we do know that it does have some anxiety risks,” he said. “The risks in people with mental illness are simply different than in people who don’t have mental illness.”
Dr. Strakowski, Mr. Mikos, and Mr. Daily report no relevant disclosures.
A version of this article appeared on Medscape.com.
The US Drug Enforcement Agency (DEA) is moving forward with plans to move marijuana from a Schedule I to a Schedule III controlled substance under the Controlled Substance Act (CSA), the US Department of Justice officials announced this week.
First reported by the Associated Press and since confirmed by this news organization through a US Department of Justice spokesperson, the news made international headlines. Despite the media splash, the final rule is still months away.
How did we get here? What happens next? What impact might rescheduling have on clinicians, patients, researchers, and the medical cannabis industry?
Why Reschedule? Why Now?
The DEA’s decision is based on a 2023 determination from the US Food and Drug Administration (FDA) that marijuana has a legitimate medical use and should be moved to Schedule III.
This class includes ketamine, acetaminophen with codeine, and buprenorphine.
Even though the manufacturing, distribution, sale, and use of marijuana has long violated federal law, 38 states and Washington, DC, have legalized medical cannabis, and 24 states and DC have legalized its recreational use.
Congress has allowed states leeway for the distribution and use of medical marijuana, and current and previous presidential administrations have chosen not to aggressively pursue prosecution of state-allowed marijuana use, the Congressional Research Service (CRS) reports.
Pressure to address the conflict between federal and state laws and an increasing interest in drug development of cannabis and cannabis-derived products probably contributed to the DEA’s decision, said Stephen Strakowski, MD, professor, and vice chair of psychiatry at Indiana University in Indianapolis, and professor and associate vice president at University of Texas in Austin.
“The trend toward legalization is everywhere and even though nationally the feds in this instance are lagging the states, the pressure to legalize has been intense for 50 years and it’s not surprising that the DEA is finally following that lead,” Dr. Strakowski told this news organization.
How Does Rescheduling Work? What’s the Timeline?
The DEA will submit a formal rule proposing that marijuana be moved from Schedule I to Schedule III to the White House Office of Management and Budget. The timing of the submission is unclear.
Once the proposed rule is posted to the Federal Register, there will be a public comment period, which usually lasts 30-60 days.
“This will likely generate a lot of public comment,” Robert Mikos, JD, LaRoche Family Chair in Law at Vanderbilt University Law School in Nashville, Tennessee, told this news organization. “Then the agency has to go back and wade through those comments and decide if they want to proceed with the rule as proposed or modify it.”
A final rule will probably be posted before the end of the current presidential term in January, Mr. Mikos said. While a lawsuit blocking its implementation is possible, there is a “low chance that a court would block this,” he added.
How Will Rescheduling Affect Medical Marijuana?
For medical marijuana, changing the drug to a Schedule III means that it can legally be prescribed but only in states that have legalized medical cannabis, Mr. Mikos said.
“If you’re a patient in a state with a medical marijuana law and your physician gives you a prescription for medical marijuana and you possess it, you will no longer be guilty of a federal crime,” he said.
Rescheduling could also benefit patients who receive care through the Veterans Administration (VA), Mr. Mikos said. For several years, the VA has had a policy that blocked clinicians from prescribing medical marijuana because as a Schedule I drug, it was determined to have no accepted medical use.
“It’s possible the VA may drop that policy once the drug gets rescheduled. If you’re in a medical marijuana state, if you’re a VA patient, and you don’t want to spend the extra money to go outside that system, this will have meaningful impact on their lives,” Mr. Mikos said.
But what about patients living in states that have not legalized medical cannabis?
“You still wouldn’t be committing a federal crime, but you could be violating state law,” Mr. Mikos said. “That’s a much more salient consideration because if you look at who goes after individuals who possess small amounts of drugs, the state handles 99% of those cases.”
The manufacture, distribution, and possession of recreational marijuana would remain illegal under federal law.
What Does It Mean for Medical Marijuana Dispensaries?
Though rescheduling makes it legal for clinicians to prescribe medical marijuana and for patients to use it, the actual sale of the drug will remain illegal under federal law because rescheduling only changes prescribing under the CSA, Mr. Mikos said.
“If you’re a dispensary and you sell it, even if it’s to somebody who’s got a prescription, you’re still probably violating the Food, Drug and Cosmetics Act. Rescheduling doesn’t change that,” he said.
“Even assuming the DEA follows through with this and it doesn’t come undone at some future date, the industry is still going struggle to comply with the Controlled Substances Act post rescheduling because that statute is going to continue to impose a number of regulations on the industry,” Mr. Mikos added.
However, rescheduling would change the tax status of the estimated 12,000-15,000 state-licensed cannabis dispensaries in the United States, allowing access to certain tax deductions that are unavailable to sales involving Schedule I controlled substances, James Daily, JD, MS, with Center for Empirical Research in the Law at Washington University School of Law in St. Louis, told this news organization.
“Many cannabis businesses do in fact pay federal taxes, but the inability to take any federal tax credits or deductions means that their effective tax rate is much higher than it would otherwise be,” Mr. Daily said.
Although new federal tax deductions would likely available to cannabis businesses if marijuana were rescheduled to Schedule III, “their business would still be in violation of federal law,” he said.
“This creates a further tension between state and federal law, which could be resolved by further legalization or it could be resolved by extending the prohibition on tax deductions to include cannabis and not just Schedule I and II drugs,” he added.
Will Rescheduling Make It Easier to Conduct Cannabis-Related Research?
Research on medical cannabis has been stymied by FDA and DEA regulations regarding the study of Schedule I controlled substances. Although rescheduling could lift that barrier, other challenges would remain.
“Schedule III drugs can be more easily researched, but it’s unclear if, for example, a clinical trial could lawfully obtain the cannabis from a dispensary or if they would still have to go through the one legal federal supplier of cannabis,” Daily said.
The FDA reports having received more than 800 investigational new drug applications for and pre-investigational new drug applications related to cannabis and cannabis-derived products since the 1970s, the agency reports. To date, the FDA has not approved any marketing drug applications for cannabis for the treatment of any disease or condition.
In January 2023, the agency published updated guidelines for researchers and sponsors interested in developing drugs containing cannabis or cannabis-derived compounds.
It’s unclear whether those guidelines would be updated if the rescheduling moves forward.
Does Rescheduling Marijuana Pose Any Risk?
In its report to the DEA that marijuana be rescheduled, the FDA was careful to note that the agency’s recommendation is “not meant to imply that safety and effectiveness have been established for marijuana that would support FDA approval of a marijuana drug product for a particular indication.”
That’s a notation that clinicians and patients should take to heart, Dr. Strakowski said.
“It’s important to remind people that Schedule III drugs, by definition, have addiction and other side effect risks,” he said. “The celebrity marketing that sits behind a lot of this is incompletely informed. It’s portrayed as fun and harmless in almost every movie and conversation you see, and we know that’s not true.”
Previous studies have linked cannabis to increased risk for mania, anxiety disorders, and schizophrenia.
“It is increasingly clear that marijuana use is linked to poor outcomes in people who struggle with mental illness,” Dr. Strakowski said. “We have no evidence that it can help you but there is evidence that it can harm you.”
Dr. Strakowski likens cannabis use to alcohol, which is a known depressant that is associated with worse outcomes in people with mental illness.
“I think with cannabis, we don’t know enough about it yet, but we do know that it does have some anxiety risks,” he said. “The risks in people with mental illness are simply different than in people who don’t have mental illness.”
Dr. Strakowski, Mr. Mikos, and Mr. Daily report no relevant disclosures.
A version of this article appeared on Medscape.com.
The US Drug Enforcement Agency (DEA) is moving forward with plans to move marijuana from a Schedule I to a Schedule III controlled substance under the Controlled Substance Act (CSA), the US Department of Justice officials announced this week.
First reported by the Associated Press and since confirmed by this news organization through a US Department of Justice spokesperson, the news made international headlines. Despite the media splash, the final rule is still months away.
How did we get here? What happens next? What impact might rescheduling have on clinicians, patients, researchers, and the medical cannabis industry?
Why Reschedule? Why Now?
The DEA’s decision is based on a 2023 determination from the US Food and Drug Administration (FDA) that marijuana has a legitimate medical use and should be moved to Schedule III.
This class includes ketamine, acetaminophen with codeine, and buprenorphine.
Even though the manufacturing, distribution, sale, and use of marijuana has long violated federal law, 38 states and Washington, DC, have legalized medical cannabis, and 24 states and DC have legalized its recreational use.
Congress has allowed states leeway for the distribution and use of medical marijuana, and current and previous presidential administrations have chosen not to aggressively pursue prosecution of state-allowed marijuana use, the Congressional Research Service (CRS) reports.
Pressure to address the conflict between federal and state laws and an increasing interest in drug development of cannabis and cannabis-derived products probably contributed to the DEA’s decision, said Stephen Strakowski, MD, professor, and vice chair of psychiatry at Indiana University in Indianapolis, and professor and associate vice president at University of Texas in Austin.
“The trend toward legalization is everywhere and even though nationally the feds in this instance are lagging the states, the pressure to legalize has been intense for 50 years and it’s not surprising that the DEA is finally following that lead,” Dr. Strakowski told this news organization.
How Does Rescheduling Work? What’s the Timeline?
The DEA will submit a formal rule proposing that marijuana be moved from Schedule I to Schedule III to the White House Office of Management and Budget. The timing of the submission is unclear.
Once the proposed rule is posted to the Federal Register, there will be a public comment period, which usually lasts 30-60 days.
“This will likely generate a lot of public comment,” Robert Mikos, JD, LaRoche Family Chair in Law at Vanderbilt University Law School in Nashville, Tennessee, told this news organization. “Then the agency has to go back and wade through those comments and decide if they want to proceed with the rule as proposed or modify it.”
A final rule will probably be posted before the end of the current presidential term in January, Mr. Mikos said. While a lawsuit blocking its implementation is possible, there is a “low chance that a court would block this,” he added.
How Will Rescheduling Affect Medical Marijuana?
For medical marijuana, changing the drug to a Schedule III means that it can legally be prescribed but only in states that have legalized medical cannabis, Mr. Mikos said.
“If you’re a patient in a state with a medical marijuana law and your physician gives you a prescription for medical marijuana and you possess it, you will no longer be guilty of a federal crime,” he said.
Rescheduling could also benefit patients who receive care through the Veterans Administration (VA), Mr. Mikos said. For several years, the VA has had a policy that blocked clinicians from prescribing medical marijuana because as a Schedule I drug, it was determined to have no accepted medical use.
“It’s possible the VA may drop that policy once the drug gets rescheduled. If you’re in a medical marijuana state, if you’re a VA patient, and you don’t want to spend the extra money to go outside that system, this will have meaningful impact on their lives,” Mr. Mikos said.
But what about patients living in states that have not legalized medical cannabis?
“You still wouldn’t be committing a federal crime, but you could be violating state law,” Mr. Mikos said. “That’s a much more salient consideration because if you look at who goes after individuals who possess small amounts of drugs, the state handles 99% of those cases.”
The manufacture, distribution, and possession of recreational marijuana would remain illegal under federal law.
What Does It Mean for Medical Marijuana Dispensaries?
Though rescheduling makes it legal for clinicians to prescribe medical marijuana and for patients to use it, the actual sale of the drug will remain illegal under federal law because rescheduling only changes prescribing under the CSA, Mr. Mikos said.
“If you’re a dispensary and you sell it, even if it’s to somebody who’s got a prescription, you’re still probably violating the Food, Drug and Cosmetics Act. Rescheduling doesn’t change that,” he said.
“Even assuming the DEA follows through with this and it doesn’t come undone at some future date, the industry is still going struggle to comply with the Controlled Substances Act post rescheduling because that statute is going to continue to impose a number of regulations on the industry,” Mr. Mikos added.
However, rescheduling would change the tax status of the estimated 12,000-15,000 state-licensed cannabis dispensaries in the United States, allowing access to certain tax deductions that are unavailable to sales involving Schedule I controlled substances, James Daily, JD, MS, with Center for Empirical Research in the Law at Washington University School of Law in St. Louis, told this news organization.
“Many cannabis businesses do in fact pay federal taxes, but the inability to take any federal tax credits or deductions means that their effective tax rate is much higher than it would otherwise be,” Mr. Daily said.
Although new federal tax deductions would likely available to cannabis businesses if marijuana were rescheduled to Schedule III, “their business would still be in violation of federal law,” he said.
“This creates a further tension between state and federal law, which could be resolved by further legalization or it could be resolved by extending the prohibition on tax deductions to include cannabis and not just Schedule I and II drugs,” he added.
Will Rescheduling Make It Easier to Conduct Cannabis-Related Research?
Research on medical cannabis has been stymied by FDA and DEA regulations regarding the study of Schedule I controlled substances. Although rescheduling could lift that barrier, other challenges would remain.
“Schedule III drugs can be more easily researched, but it’s unclear if, for example, a clinical trial could lawfully obtain the cannabis from a dispensary or if they would still have to go through the one legal federal supplier of cannabis,” Daily said.
The FDA reports having received more than 800 investigational new drug applications for and pre-investigational new drug applications related to cannabis and cannabis-derived products since the 1970s, the agency reports. To date, the FDA has not approved any marketing drug applications for cannabis for the treatment of any disease or condition.
In January 2023, the agency published updated guidelines for researchers and sponsors interested in developing drugs containing cannabis or cannabis-derived compounds.
It’s unclear whether those guidelines would be updated if the rescheduling moves forward.
Does Rescheduling Marijuana Pose Any Risk?
In its report to the DEA that marijuana be rescheduled, the FDA was careful to note that the agency’s recommendation is “not meant to imply that safety and effectiveness have been established for marijuana that would support FDA approval of a marijuana drug product for a particular indication.”
That’s a notation that clinicians and patients should take to heart, Dr. Strakowski said.
“It’s important to remind people that Schedule III drugs, by definition, have addiction and other side effect risks,” he said. “The celebrity marketing that sits behind a lot of this is incompletely informed. It’s portrayed as fun and harmless in almost every movie and conversation you see, and we know that’s not true.”
Previous studies have linked cannabis to increased risk for mania, anxiety disorders, and schizophrenia.
“It is increasingly clear that marijuana use is linked to poor outcomes in people who struggle with mental illness,” Dr. Strakowski said. “We have no evidence that it can help you but there is evidence that it can harm you.”
Dr. Strakowski likens cannabis use to alcohol, which is a known depressant that is associated with worse outcomes in people with mental illness.
“I think with cannabis, we don’t know enough about it yet, but we do know that it does have some anxiety risks,” he said. “The risks in people with mental illness are simply different than in people who don’t have mental illness.”
Dr. Strakowski, Mr. Mikos, and Mr. Daily report no relevant disclosures.
A version of this article appeared on Medscape.com.
The Burden of Skin Cancer in the Military Health System, 2017-2022
This retrospective observational study investigates skin cancer prevalence and care patterns within the Military Health System (MHS) from 2017 to 2022. Utilizing the MHS Management Analysis and Reporting Tool (most commonly called M2), we analyzed more than 5 million patient encounters and documented skin cancer prevalence in the MHS beneficiary population utilizing available demographic data. Notable findings included an increased prevalence of skin cancer in the military population compared with the civilian population, a substantial decline in direct care (DC) visits at military treatment facilities compared with civilian purchased care (PC) visits, and a decreased total number of visits during COVID-19 restrictions.
The Military Health System (MHS) is a worldwide health care delivery system that serves 9.6 million beneficiaries, including military service members, retirees, and their families.1 Its mission is 2-fold: provide a medically ready force, and provide a medical benefit in keeping with the service and sacrifice of active-duty personnel, military retirees, and their families. For fiscal year (FY) 2022, active-duty service members and their families comprised 16.7% and 19.9% of beneficiaries, respectively, while retired service members and their families comprised 27% and 32% of beneficiaries, respectively.
The MHS operates under the authority of the Department of Defense (DoD) and is supported by an annual budget of approximately $50 billion.1 Health care provision within the MHS is managed by TRICARE regional networks.2 Within these networks, MHS beneficiaries may receive health care in 2 categories: direct care (DC) and purchased care (PC). Direct care is rendered in military treatment facilities by military or civilian providers contracted by the DoD, and PC is administered by civilian providers at civilian health care facilities within the TRICARE network, which is comprised of individual providers, clinics, and hospitals that have agreed to accept TRICARE beneficiaries.1 Purchased care is fee-for-service and paid for by the MHS. Of note, the MHS differs from the Veterans Affairs health care system in that the MHS through DC and PC sees only active-duty service members, active-duty dependents, retirees, and retirees’ dependents (primarily spouses), whereas Veterans Affairs sees only veterans (not necessarily retirees) discharged from military service with compensable medical conditions or disabilities.
Skin cancer presents a notable concern for the US Military, as the risk for skin cancer is thought to be higher than in the general population.3,4 This elevated risk is attributed to numerous factors inherent to active-duty service, including time spent in tropical environments, increased exposure to UV radiation, time spent at high altitudes, and decreased rates of sun-protective behaviors.3 Although numerous studies have explored the mechanisms that contribute to service members’ increased skin cancer risk, there are few (if any) that discuss the burden of skin cancer on the MHS and where its beneficiaries receive their skin cancer care. This study evaluated the burden of skin cancer within the MHS, as demonstrated by the period prevalence of skin cancer among its beneficiaries and the number and distribution of patient visits for skin cancer across both DC and PC from 2017 to 2022.
Methods
Data Collection—This retrospective observational study was designed to describe trends in outpatient visits with a skin cancer diagnosis and annual prevalence of skin cancer types in the MHS. Data are from all MHS beneficiaries who were eligible or enrolled in the analysis year. Our data source was the MHS Management Analysis and Reporting Tool (most commonly called M2), a query tool that contains the current and most recent 5 full FYs of Defense Health Agency corporate health care data including aggregated FY and calendar-year counts of MHS beneficiaries from 2017 to 2022 using encounter and claims data tables from both DC and PC. Data in M2 are coded using a pseudo-person identification number, and queries performed for this study were limited to de-identified visit and patient counts.
Skin cancer diagnoses were defined by relevant International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes recorded from outpatient visits in DC and PC. The M2 database was queried to find aggregate counts of visits and unique MHS beneficiaries with one or more diagnoses of a skin cancer type of interest (defined by relevant ICD-10-CM code) over the study period stratified by year and by patient demographic characteristics. Skin cancer types by ICD-10-CM code group included basal cell carcinoma (BCC), squamous cell carcinoma (SCC), malignant melanoma (MM), and other (including Merkel cell carcinoma and sebaceous carcinoma). Demographic strata included age, sex, military status (active duty, dependents of active duty, retired, or all others), sponsor military rank, and sponsor branch (army, air force, marine corps, or navy). Visit counts included diagnoses from any ICD position (for encounters that contained multiple ICD codes) to describe the total volume of care that addressed a diagnosed skin cancer. Counts of unique patients in prevalence analyses included relevant diagnoses in the primary ICD position only to increase the specificity of prevalence estimates.
Data Analysis—Descriptive analyses included the total number of outpatient visits with a skin cancer diagnosis in DC and PC over the study period, with percentages of total visits by year and by demographic strata. Separate analyses estimated annual prevalences of skin cancer types in the MHS by study year and within 2022 by demographic strata. Numerators in prevalence analyses were defined as the number of unique individuals with one or more relevant ICD codes in the analysis year. Denominators were defined as the total number of MHS beneficiaries in the analysis year and resulting period prevalences reported. Observed prevalences were qualitatively described, and trends were compared with prevalences in nonmilitary populations reported in the literature.
Ethics—This study was conducted as part of a study using secondary analyses of de-identified data from the M2 database. The study was reviewed and approved by the Walter Reed National Military Medical Center institutional review board.
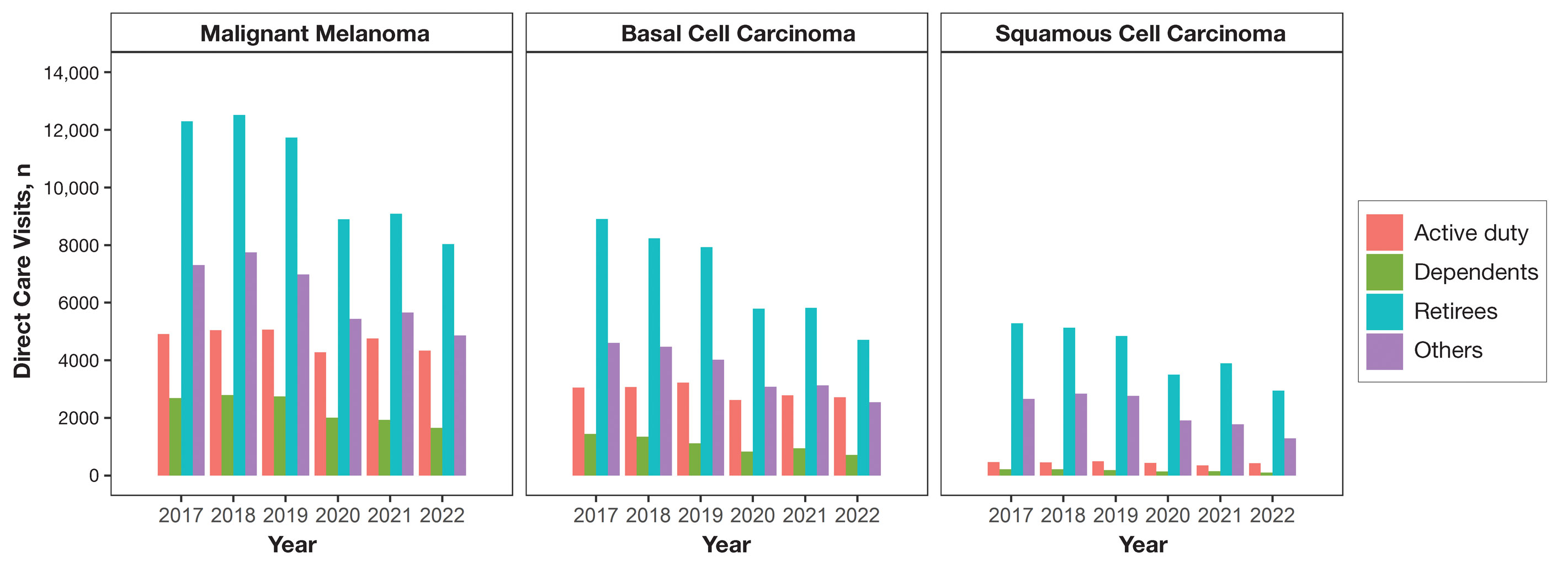
Results
Encounter data were analyzed from a total of 5,374,348 visits between DC and PC over the study period for each cancer type of interest. Figures 1 and 2 show temporal trends in DC visits compared with PC visits in each beneficiary category. The percentage of total DC visits subsequently declined each year throughout the study period, with percentage decreases from 2017 to 2022 of 1.45% or 8200 fewer visits for MM, 3.41% or 7280 fewer visits for BCC, and 2.26% or 3673 fewer visits for SCC.
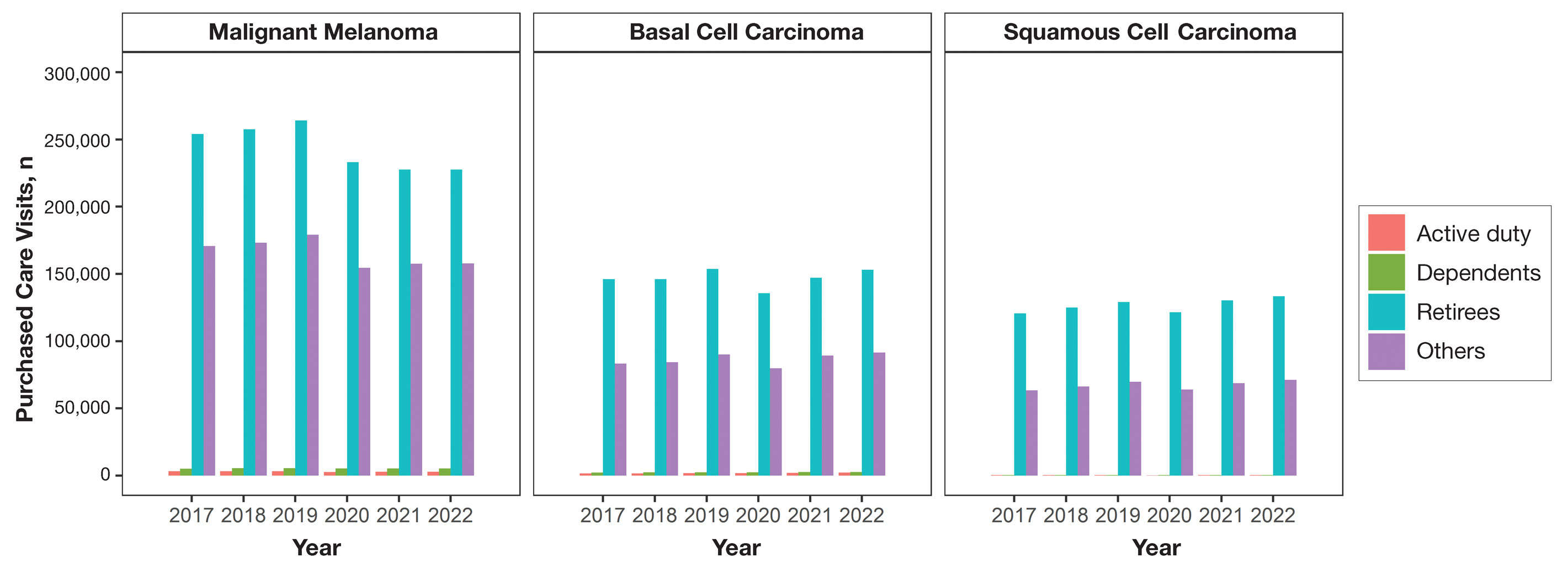
When stratified by beneficiary category, this trend remained consistent among dependents and retirees, with the most notable annual percentage decrease from 2019 to 2020. A higher proportion of younger adults and active-duty beneficiaries was seen in DC relative to PC, in which most visits were among retirees and others (primarily dependents of retirees, survivors, and Guard/Reserve on active duty, as well as inactive Guard/Reserve). No linear trends over time were apparent for active duty in DC and for dependents and retirees in PC. eTable 1 summarizes the demographic characteristics of MHS beneficiaries being seen in DC and PC over the study period for each cancer type of interest.
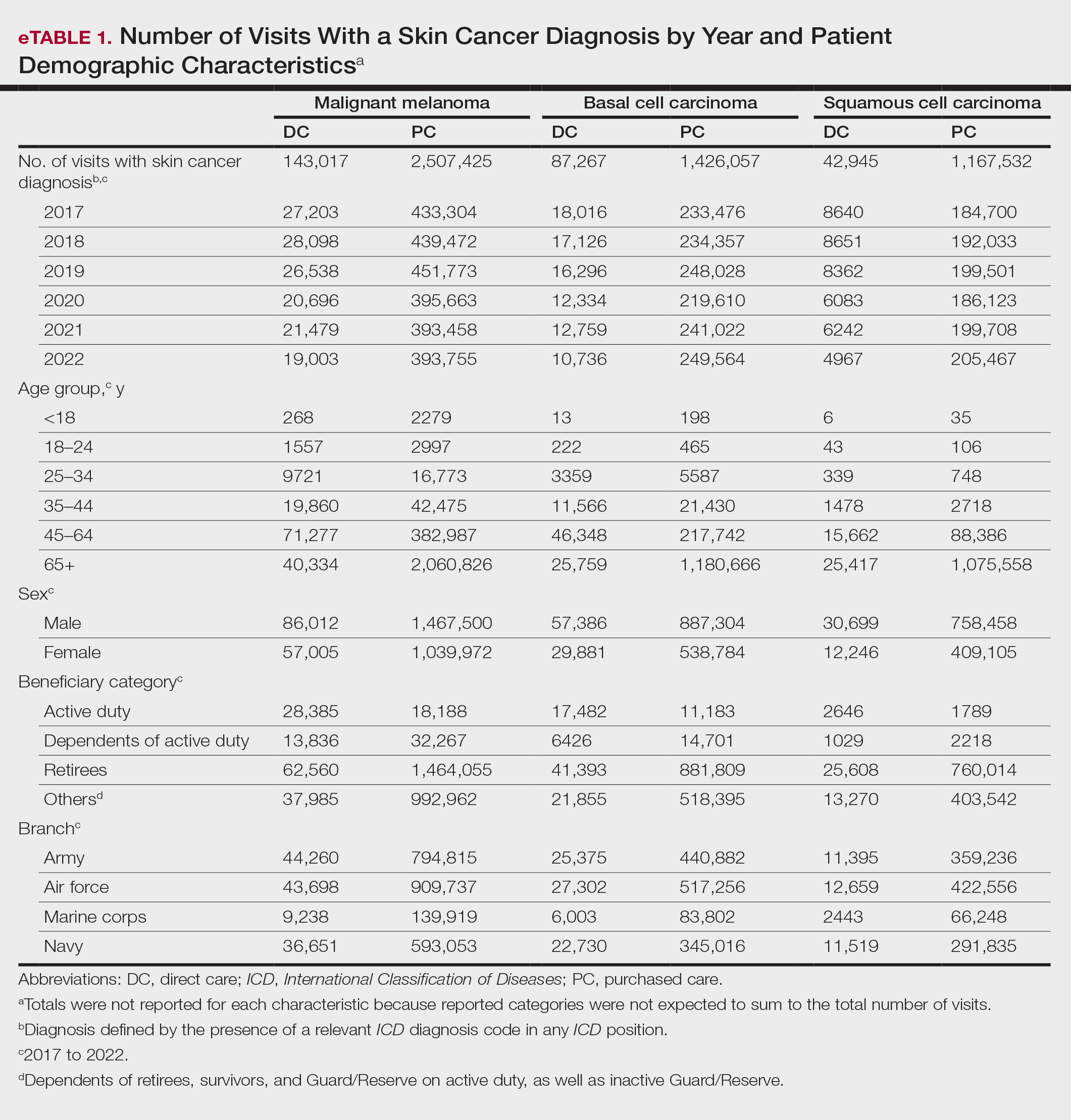
The Table shows the period prevalence of skin cancer diagnoses within the MHS beneficiary population from 2017 to 2022. These data were further analyzed by MM, BCC, and SCC (eTable 2) and demographics of interest for the year 2022. By beneficiary category, the period prevalence of MM was 0.08% in active duty, 0.06% in dependents, 0.48% in others, and 1.10% in retirees; the period prevalence of BCC was 0.12% in active duty, 0.07% in dependents, 0.91% in others, and 2.50% in retirees; and the period prevalence of SCC was 0.02% in active duty, 0.01% in dependents, 0.63% in others, and 1.87% in retirees. By sponsor branch, the period prevalence of MM was 0.35% in the army, 0.62% in the air force, 0.35% in the marine corps, and 0.65% in the navy; the period prevalence of BCC was 0.74% in the army, 1.30% in the air force, 0.74% in the marine corps, and 1.36% in the navy; and the period prevalence of SCC was 0.52% in the army, 0.92% in the air force, 0.51% in the marine corps, and 0.97% in the navy.

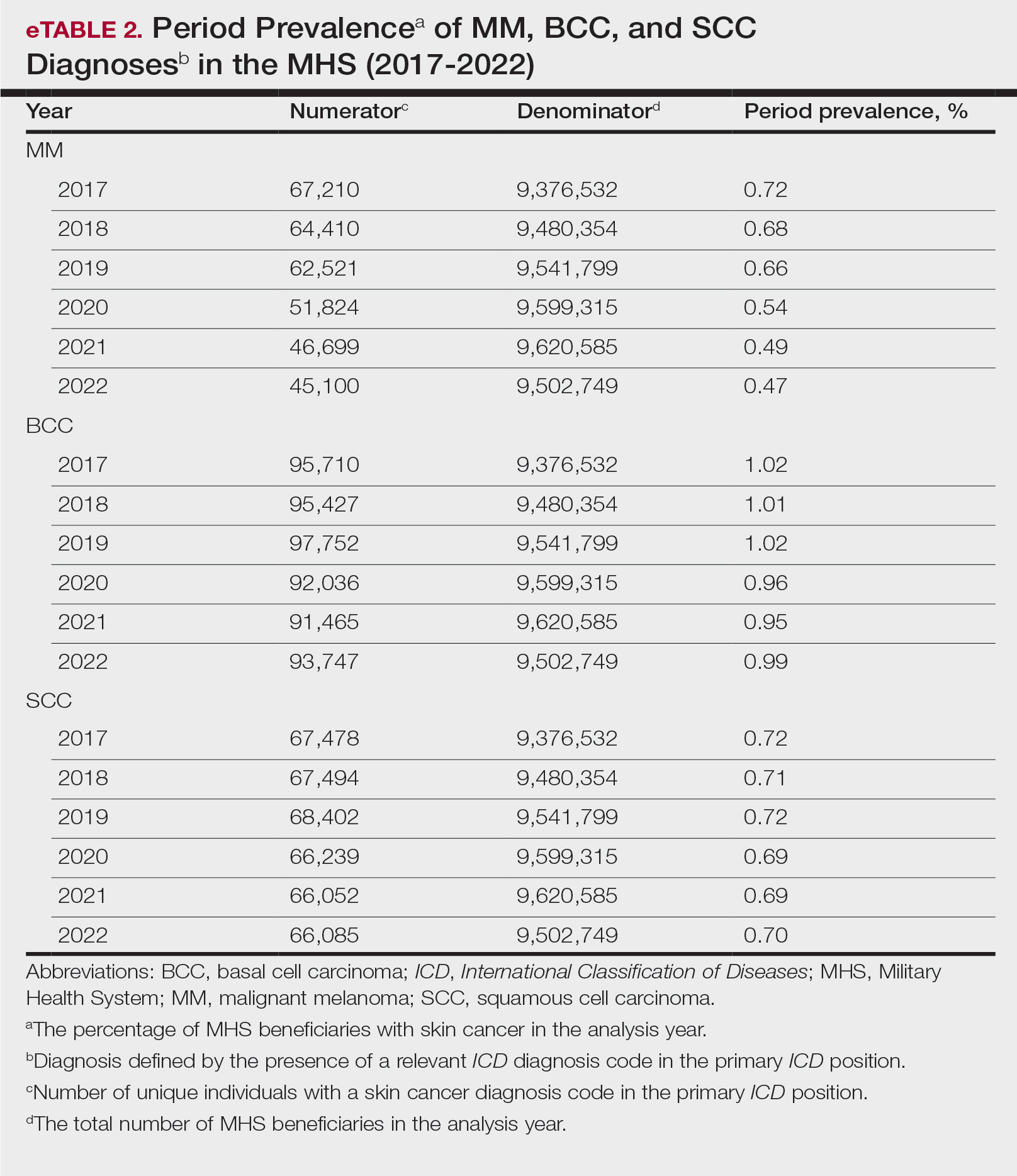
Comment
This study aimed to provide insight into the burden of skin cancer within the MHS beneficiary population and to identify temporal trends in where these beneficiaries receive their care. We examined patient encounter data from more than 9.6 million MHS beneficiaries.
The utilization of ICD codes from patient encounters to estimate the prevalence of nonmelanoma skin cancer (NMSC) has demonstrated a high positive predictive value. In one study, NMSC cases were confirmed in 96.5% of ICD code–identified patients.5 We presented an extensive collection of epidemiologic data on BCC and SCC, which posed unique challenges for tracking, as they are not reported to or monitored by cancer registries such as the Surveillance, Epidemiology, and End Results (SEER) Program.6
MHS Compared to the US Population—A study using the Global Burden of Disease 2019 database revealed an increasing trend in the incidence and prevalence of NMSC and melanoma since 1990. The same study found the period prevalence in 2019 of MM, SCC, and BCC in the general US population to be 0.13%, 0.31%, and 0.05%, respectively.7 In contrast, among MHS beneficiaries, we observed a higher prevalence in the same year, with figures of 0.66% for MM, 0.72% for SCC, and 1.02% for BCC. According to the SEER database, the period prevalence of MM within the general US population in 2020 was 0.4%.8 That same year, we identified a higher period prevalence of MM—0.54%—within the MHS beneficiary population. Specifically, within the MHS retiree population, the prevalence in 2022 was double that of the general MHS population, with a rate of 1.10%, underscoring the importance of skin cancer screening in older, at-risk adult populations. Prior studies similarly found increased rates of skin cancer within the military beneficiary population. Further studies are needed to compare age-adjusted rates in the MHS vs US population.9-11
COVID-19 Trends—Our data showed an overall decreasing prevalence of skin cancer in the MHS from 2019 to 2021. We suspect that the apparent decrease in skin cancer prevalence may be attributed to underdiagnosis from COVID-19 pandemic restrictions. During that time, many dermatology clinics at military treatment facilities underwent temporary closures, and some dermatologists were sent on nondermatologic utilization tours. Likewise, a US multi-institutional study described declining rates of new melanomas from 2020 to 2021, with an increased proportion of patient presentations with advanced melanoma, suggesting an underdiagnosis of melanoma cases during pandemic restrictions. That study also noted an increased rate of patient-identified melanomas and a decreased rate of provider-identified melanomas during that time.12 Contributing factors may include excess hospital demand, increased patient complexity and acute care needs, and long outpatient clinic backlogs during this time.13Financial Burden—Over our 5-year study period, there were 5,374,348 patient encounters addressing skin cancer, both in DC and PC (Figures 1 and 2; eTable 1). In 2016 to 2018, the average annual cost of treating skin cancer in the US civilian, noninstitutionalized population was $1243 for NMSC (BCC and SCC) and $2430 for melanoma.6 Using this metric, the estimated total cost of care rendered in the MHS in 2018 for NMSC and melanoma was $202,510,803 and $156,516,300, respectively.
Trends in DC vs PC—In the years examined, we found a notable decrease in the number of beneficiaries receiving treatment for MM, BCC, and SCC in DC. Simultaneously, there has been an increase in the number of beneficiaries receiving PC for BCC and SCC, though this trend was not apparent for MM.
Our data provided interesting insights into the percentage of PC compared with DC offered within the MHS. Importantly, our findings suggested that the majority of skin cancer in active-duty service members is managed with DC within the military treatment facility setting (61% DC management over the period analyzed). This finding was true across all years of data analyzed, suggesting that the COVID-19 pandemic did not result in a quantifiable shift in care of skin cancer within the active-duty component to outside providers. One of the critical roles of dermatologists in the MHS is to diagnose and treat skin cancer, and our study suggested that the current global manning and staffing for MHS dermatologists may not be sufficient to meet the burden of skin cancers encountered within our active-duty troops, as only 61% are managed with DC. In particular, service members in more austere and/or overseas locations may not have ready access to a dermatologist.
The burden of skin cancer shifts dramatically when analyzing care of all other populations included in these data, including dependents of active-duty service members, retirees, and the category of “other” (ie, principally dependents of retirees). Within these populations, the rate of DC falls to 30%, with 70% of active-duty dependent care being deferred to network. The findings are even more noticeable for retirees and others within these 2 cohorts in all types of skin cancer analyzed, where DC only accounted for 5.2% of those skin cancers encountered and managed across TRICARE-eligible beneficiaries. For MM, BCC, and SCC, percentages of DC were 5.4%, 5.8%, and 3.5%, respectively. Although it is interesting to note the lower percentage of SCC managed via DC, our data did not allow for extrapolation as to why more SCC cases may be deferred to network. The shift to PC may align with DoD initiatives to increase the private sector’s involvement in military medicine and transition to civilianizing the MHS.14 In the end, the findings are remarkable, with approximately 95% of skin cancer care and management provided overall via PC.
These findings differ from previously published data regarding DC and PC from other specialty areas. Results from an analysis of DC vs PC for plastic surgery for the entire MHS from 2016 to 2019 found 83.2% of cases were deferred to network.15 A similar publication in the orthopedics literature examined TRICARE claims for patients who underwent total hip or knee arthroplasties between 2006 and 2019 and found 84.6% of cases were referred for PC. Notably, the authors utilized generalized linear models for cost analysis and found that DC was more expensive than PC, though this likely was a result of higher rates of hospital readmission within DC cases.16 Lastly, an article on the DC vs PC disposition of MHS patients with breast cancer from 2003 to 2008 found 46% of cases managed with DC vs 26.% with PC and 27.8% receiving a combination. In this case, the authors found a reduced cost associated with DC vs PC.17
Little additional literature exists regarding the costs of DC vs PC. An article published in 2016 designed to assess costs of DC vs PC showed that almost all military treatment facilities have higher costs than their private sector counterparts, with a few exceptions.18 This does not assess the costs of specific procedures, however, and only the overall cost to maintain a treatment facility. Importantly, this study was based on data from FY 2014 and has not been updated to reflect complex changes within the MHS system and the private health care system. Indeed, a US Government Accountability Office FY 2023 study highlighted staffing and efficiency issues within this transition to civilian medicine; subsequently, the 2024 President’s Budget suspended all planned clinical medical military end strength divestitures, underscoring the potential ineffectiveness of a civilianized MHS at meeting the health care needs of its beneficiaries.19,20 Future research on a national scale will be necessary to see if there is a reversal of this trend to PC and if doing so has any impact on access to DC for active-duty troops or active-duty dependents.
In addition to PC vs DC trends, we also can get a sense of the impact of the COVID pandemic restrictions on access to DC vs PC by assessing the change in rates seen in the data from the pre-COVID years (2017-2019) to the “post-COVID” years (2020-2022) included. Overall, rates of DC decreased uniformly from their already low percentages. In our study, rates of DC decreased from 5.8% in 2019 to 4.8% in 2022 for MM, from 6.6% to 4.3% for BCC, and from 4.2% to 2.9% for SCC. Although these changes seem small at first, they represent a 30.6% overall decrease in DC for BCC and an overall decrease of 55.4% in DC for SCC. Although our data do not allow us to extrapolate the real cost of this reduction across a nationwide health care system and more than 5 million care encounters, the financial and personal (ie, lost man-hours) costs of this decrease in DC likely are substantial.
In addition to costs, qualitative aspects that contribute to the burden of skin cancer include treatment-related morbidity, such as scarring, pain, and time spent away from family, work, and hobbies, as well as overall patient satisfaction with the quality of care they receive.21 Future work is critical to assess the real cost of this immense burden of PC for the treatment and management of skin cancers within the DoD beneficiary population.
Limitations—This study is limited by its observational nature. Given the mechanism of our data collection, we may have underestimated disease prevalence, as not all patients are seen for their diagnosis annually. Furthermore, reported demographic strata (eg, age, sex) were limited to those available and valid in the M2 reporting system. Finally, our study only collected data from those service members or former service members seen within the MHS and does not reflect any care rendered to those who are no longer active duty but did not officially retire from the military (ie, nonretired service members receiving care in the Veterans Affairs system for skin cancer).
Conclusion
We describe the annual burden of care for skin cancer in the MHS beneficiary population. Noteworthy findings observed were an overall decrease in beneficiaries being treated for skin cancer through DC; a decreasing annual prevalence of skin cancer diagnosis between 2019 and 2021, which may represent underdiagnosis or decreased follow-up in the setting of the COVID-19 pandemic; and a higher rate of skin cancer in the military beneficiary population compared to the civilian population.
- US Department of Defense. Military health. Accessed October 5, 2023. https://www.defense.gov/
- Wooten NR, Brittingham JA, Pitner RO, et al. Purchased behavioral health care received by Military Health System beneficiaries in civilian medical facilities, 2000-2014. Mil Med. 2018;183:E278-E290. doi:10.1093/milmed/usx101
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- American Academy of Dermatology. Skin cancer. Updated April 22, 2022. Accessed April 17, 2024. https://www.aad.org/media/stats-skin-cancer
- Eide MJ, Krajenta R, Johnson D, et al. Identification of patients with nonmelanoma skin cancer using health maintenance organization claims data. Am J Epidemiol. 2010;171:123-128. doi:10.1093/aje/kwp352
- Kao SYZ, Ekwueme DU, Holman DM, et al. Economic burden of skin cancer treatment in the USA: an analysis of the Medical Expenditure Panel Survey Data, 2012-2018. Cancer Causes Control. 2023;34:205-212. doi:10.1007/s10552-022-01644-0
- Aggarwal P, Knabel P, Fleischer AB. United States burden of melanoma and non-melanoma skin cancer from 1990 to 2019. J Am Acad Dermatol. 2021;85:388-395. doi:10.1016/j.jaad.2021.03.109
- SEER*Explorer. SEER Incidence Data, November 2023 Submission (1975-2021). National Cancer Institute; 2024. Accessed April 17, 2024. https://seer.cancer.gov/statistics-network/explorer/application.html?site=53&data_type=1&graph_type=1&compareBy=sex&chk_sex_1=1&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=1&age_range=1&advopt_precision=1&advopt_show_ci=on&hdn_view=1&advopt_show_apc=on&advopt_display=1
- Brown J, Kopf AW, Rigel DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663. doi:10.1111/j.1365-4362.1984.tb01228.x
- Page WF, Whiteman D, Murphy M. A comparison of melanoma mortality among WWII veterans of the Pacific and European theaters. Ann Epidemiol. 2000;10:192-195. doi:10.1016/s1047-2797(99)00050-2
- Ramani ML, Bennett RG. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J Am Acad Dermatol. 1993;28:733-737. doi:10.1016/0190-9622(93)70102-Y
- Trepanowski N, Chang MS, Zhou G, et al. Delays in melanoma presentation during the COVID-19 pandemic: a nationwide multi-institutional cohort study. J Am Acad Dermatol. 2022;87:1217-1219. doi:10.1016/j.jaad.2022.06.031
- Gibbs A. COVID-19 shutdowns caused delays in melanoma diagnoses, study finds. OHSU News. August 4, 2022. Accessed April 17, 2024. https://news.ohsu.edu/2022/08/04/covid-19-shutdowns-caused-delays-in-melanoma-diagnoses-study-finds
- Kime P. Pentagon budget calls for ‘civilianizing’ military hospitals. Military Times. Published February 10, 2020. Accessed April 17, 2024. https://www.militarytimes.com/news/your-military/2020/02/10/pentagon-budget-calls-for-civilianizing-military-hospitals/
- O’Reilly EB, Norris E, Ortiz-Pomales YT, et al. A comparison of direct care at military medical treatment facilities with purchased care in plastic surgery operative volume. Plast Reconstr Surg Glob Open. 2022;10(10 suppl):124-125. doi:10.1097/01.GOX.0000898976.03344.62
- Haag A, Hosein S, Lyon S, et al. Outcomes for arthroplasties in military health: a retrospective analysis of direct versus purchased care. Mil Med. 2023;188(suppl 6):45-51. doi:10.1093/milmed/usac441
- Eaglehouse YL, Georg MW, Richard P, et al. Cost-efficiency of breast cancer care in the US Military Health System: an economic evaluation in direct and purchased care. Mil Med. 2019;184:e494-e501. doi:10.1093/milmed/usz025
- Lurie PM. Comparing the cost of military treatment facilities with private sector care. Institute for Defense Analyses; February 2016. Accessed April 17, 2024. https://www.ida.org/research-and-publications/publications/all/c/co/comparing-the-costs-of-military-treatment-facilities-with-private-sector-care
- Defense Health Program. Fiscal Year (FY) 2024 President’s Budget: Operation and Maintenance Procurement Research, Development, Test and Evaluation. Department of Defense; March 2023. Accessed April 17, 2024. https://comptroller.defense.gov/Portals/45/Documents/defbudget/fy2024/budget_justification/pdfs/09_Defense_Health_Program/00-DHP_Vols_I_II_and_III_PB24.pdf
- US Government Accountability Office. Defense Health Care. DOD should reevaluate market structure for military medical treatment facility management. Published August 21, 2023. Accessed April 17, 2024. https://www.gao.gov/products/gao-23-105441
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
This retrospective observational study investigates skin cancer prevalence and care patterns within the Military Health System (MHS) from 2017 to 2022. Utilizing the MHS Management Analysis and Reporting Tool (most commonly called M2), we analyzed more than 5 million patient encounters and documented skin cancer prevalence in the MHS beneficiary population utilizing available demographic data. Notable findings included an increased prevalence of skin cancer in the military population compared with the civilian population, a substantial decline in direct care (DC) visits at military treatment facilities compared with civilian purchased care (PC) visits, and a decreased total number of visits during COVID-19 restrictions.
The Military Health System (MHS) is a worldwide health care delivery system that serves 9.6 million beneficiaries, including military service members, retirees, and their families.1 Its mission is 2-fold: provide a medically ready force, and provide a medical benefit in keeping with the service and sacrifice of active-duty personnel, military retirees, and their families. For fiscal year (FY) 2022, active-duty service members and their families comprised 16.7% and 19.9% of beneficiaries, respectively, while retired service members and their families comprised 27% and 32% of beneficiaries, respectively.
The MHS operates under the authority of the Department of Defense (DoD) and is supported by an annual budget of approximately $50 billion.1 Health care provision within the MHS is managed by TRICARE regional networks.2 Within these networks, MHS beneficiaries may receive health care in 2 categories: direct care (DC) and purchased care (PC). Direct care is rendered in military treatment facilities by military or civilian providers contracted by the DoD, and PC is administered by civilian providers at civilian health care facilities within the TRICARE network, which is comprised of individual providers, clinics, and hospitals that have agreed to accept TRICARE beneficiaries.1 Purchased care is fee-for-service and paid for by the MHS. Of note, the MHS differs from the Veterans Affairs health care system in that the MHS through DC and PC sees only active-duty service members, active-duty dependents, retirees, and retirees’ dependents (primarily spouses), whereas Veterans Affairs sees only veterans (not necessarily retirees) discharged from military service with compensable medical conditions or disabilities.
Skin cancer presents a notable concern for the US Military, as the risk for skin cancer is thought to be higher than in the general population.3,4 This elevated risk is attributed to numerous factors inherent to active-duty service, including time spent in tropical environments, increased exposure to UV radiation, time spent at high altitudes, and decreased rates of sun-protective behaviors.3 Although numerous studies have explored the mechanisms that contribute to service members’ increased skin cancer risk, there are few (if any) that discuss the burden of skin cancer on the MHS and where its beneficiaries receive their skin cancer care. This study evaluated the burden of skin cancer within the MHS, as demonstrated by the period prevalence of skin cancer among its beneficiaries and the number and distribution of patient visits for skin cancer across both DC and PC from 2017 to 2022.
Methods
Data Collection—This retrospective observational study was designed to describe trends in outpatient visits with a skin cancer diagnosis and annual prevalence of skin cancer types in the MHS. Data are from all MHS beneficiaries who were eligible or enrolled in the analysis year. Our data source was the MHS Management Analysis and Reporting Tool (most commonly called M2), a query tool that contains the current and most recent 5 full FYs of Defense Health Agency corporate health care data including aggregated FY and calendar-year counts of MHS beneficiaries from 2017 to 2022 using encounter and claims data tables from both DC and PC. Data in M2 are coded using a pseudo-person identification number, and queries performed for this study were limited to de-identified visit and patient counts.
Skin cancer diagnoses were defined by relevant International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes recorded from outpatient visits in DC and PC. The M2 database was queried to find aggregate counts of visits and unique MHS beneficiaries with one or more diagnoses of a skin cancer type of interest (defined by relevant ICD-10-CM code) over the study period stratified by year and by patient demographic characteristics. Skin cancer types by ICD-10-CM code group included basal cell carcinoma (BCC), squamous cell carcinoma (SCC), malignant melanoma (MM), and other (including Merkel cell carcinoma and sebaceous carcinoma). Demographic strata included age, sex, military status (active duty, dependents of active duty, retired, or all others), sponsor military rank, and sponsor branch (army, air force, marine corps, or navy). Visit counts included diagnoses from any ICD position (for encounters that contained multiple ICD codes) to describe the total volume of care that addressed a diagnosed skin cancer. Counts of unique patients in prevalence analyses included relevant diagnoses in the primary ICD position only to increase the specificity of prevalence estimates.
Data Analysis—Descriptive analyses included the total number of outpatient visits with a skin cancer diagnosis in DC and PC over the study period, with percentages of total visits by year and by demographic strata. Separate analyses estimated annual prevalences of skin cancer types in the MHS by study year and within 2022 by demographic strata. Numerators in prevalence analyses were defined as the number of unique individuals with one or more relevant ICD codes in the analysis year. Denominators were defined as the total number of MHS beneficiaries in the analysis year and resulting period prevalences reported. Observed prevalences were qualitatively described, and trends were compared with prevalences in nonmilitary populations reported in the literature.
Ethics—This study was conducted as part of a study using secondary analyses of de-identified data from the M2 database. The study was reviewed and approved by the Walter Reed National Military Medical Center institutional review board.

Results
Encounter data were analyzed from a total of 5,374,348 visits between DC and PC over the study period for each cancer type of interest. Figures 1 and 2 show temporal trends in DC visits compared with PC visits in each beneficiary category. The percentage of total DC visits subsequently declined each year throughout the study period, with percentage decreases from 2017 to 2022 of 1.45% or 8200 fewer visits for MM, 3.41% or 7280 fewer visits for BCC, and 2.26% or 3673 fewer visits for SCC.

When stratified by beneficiary category, this trend remained consistent among dependents and retirees, with the most notable annual percentage decrease from 2019 to 2020. A higher proportion of younger adults and active-duty beneficiaries was seen in DC relative to PC, in which most visits were among retirees and others (primarily dependents of retirees, survivors, and Guard/Reserve on active duty, as well as inactive Guard/Reserve). No linear trends over time were apparent for active duty in DC and for dependents and retirees in PC. eTable 1 summarizes the demographic characteristics of MHS beneficiaries being seen in DC and PC over the study period for each cancer type of interest.

The Table shows the period prevalence of skin cancer diagnoses within the MHS beneficiary population from 2017 to 2022. These data were further analyzed by MM, BCC, and SCC (eTable 2) and demographics of interest for the year 2022. By beneficiary category, the period prevalence of MM was 0.08% in active duty, 0.06% in dependents, 0.48% in others, and 1.10% in retirees; the period prevalence of BCC was 0.12% in active duty, 0.07% in dependents, 0.91% in others, and 2.50% in retirees; and the period prevalence of SCC was 0.02% in active duty, 0.01% in dependents, 0.63% in others, and 1.87% in retirees. By sponsor branch, the period prevalence of MM was 0.35% in the army, 0.62% in the air force, 0.35% in the marine corps, and 0.65% in the navy; the period prevalence of BCC was 0.74% in the army, 1.30% in the air force, 0.74% in the marine corps, and 1.36% in the navy; and the period prevalence of SCC was 0.52% in the army, 0.92% in the air force, 0.51% in the marine corps, and 0.97% in the navy.


Comment
This study aimed to provide insight into the burden of skin cancer within the MHS beneficiary population and to identify temporal trends in where these beneficiaries receive their care. We examined patient encounter data from more than 9.6 million MHS beneficiaries.
The utilization of ICD codes from patient encounters to estimate the prevalence of nonmelanoma skin cancer (NMSC) has demonstrated a high positive predictive value. In one study, NMSC cases were confirmed in 96.5% of ICD code–identified patients.5 We presented an extensive collection of epidemiologic data on BCC and SCC, which posed unique challenges for tracking, as they are not reported to or monitored by cancer registries such as the Surveillance, Epidemiology, and End Results (SEER) Program.6
MHS Compared to the US Population—A study using the Global Burden of Disease 2019 database revealed an increasing trend in the incidence and prevalence of NMSC and melanoma since 1990. The same study found the period prevalence in 2019 of MM, SCC, and BCC in the general US population to be 0.13%, 0.31%, and 0.05%, respectively.7 In contrast, among MHS beneficiaries, we observed a higher prevalence in the same year, with figures of 0.66% for MM, 0.72% for SCC, and 1.02% for BCC. According to the SEER database, the period prevalence of MM within the general US population in 2020 was 0.4%.8 That same year, we identified a higher period prevalence of MM—0.54%—within the MHS beneficiary population. Specifically, within the MHS retiree population, the prevalence in 2022 was double that of the general MHS population, with a rate of 1.10%, underscoring the importance of skin cancer screening in older, at-risk adult populations. Prior studies similarly found increased rates of skin cancer within the military beneficiary population. Further studies are needed to compare age-adjusted rates in the MHS vs US population.9-11
COVID-19 Trends—Our data showed an overall decreasing prevalence of skin cancer in the MHS from 2019 to 2021. We suspect that the apparent decrease in skin cancer prevalence may be attributed to underdiagnosis from COVID-19 pandemic restrictions. During that time, many dermatology clinics at military treatment facilities underwent temporary closures, and some dermatologists were sent on nondermatologic utilization tours. Likewise, a US multi-institutional study described declining rates of new melanomas from 2020 to 2021, with an increased proportion of patient presentations with advanced melanoma, suggesting an underdiagnosis of melanoma cases during pandemic restrictions. That study also noted an increased rate of patient-identified melanomas and a decreased rate of provider-identified melanomas during that time.12 Contributing factors may include excess hospital demand, increased patient complexity and acute care needs, and long outpatient clinic backlogs during this time.13Financial Burden—Over our 5-year study period, there were 5,374,348 patient encounters addressing skin cancer, both in DC and PC (Figures 1 and 2; eTable 1). In 2016 to 2018, the average annual cost of treating skin cancer in the US civilian, noninstitutionalized population was $1243 for NMSC (BCC and SCC) and $2430 for melanoma.6 Using this metric, the estimated total cost of care rendered in the MHS in 2018 for NMSC and melanoma was $202,510,803 and $156,516,300, respectively.
Trends in DC vs PC—In the years examined, we found a notable decrease in the number of beneficiaries receiving treatment for MM, BCC, and SCC in DC. Simultaneously, there has been an increase in the number of beneficiaries receiving PC for BCC and SCC, though this trend was not apparent for MM.
Our data provided interesting insights into the percentage of PC compared with DC offered within the MHS. Importantly, our findings suggested that the majority of skin cancer in active-duty service members is managed with DC within the military treatment facility setting (61% DC management over the period analyzed). This finding was true across all years of data analyzed, suggesting that the COVID-19 pandemic did not result in a quantifiable shift in care of skin cancer within the active-duty component to outside providers. One of the critical roles of dermatologists in the MHS is to diagnose and treat skin cancer, and our study suggested that the current global manning and staffing for MHS dermatologists may not be sufficient to meet the burden of skin cancers encountered within our active-duty troops, as only 61% are managed with DC. In particular, service members in more austere and/or overseas locations may not have ready access to a dermatologist.
The burden of skin cancer shifts dramatically when analyzing care of all other populations included in these data, including dependents of active-duty service members, retirees, and the category of “other” (ie, principally dependents of retirees). Within these populations, the rate of DC falls to 30%, with 70% of active-duty dependent care being deferred to network. The findings are even more noticeable for retirees and others within these 2 cohorts in all types of skin cancer analyzed, where DC only accounted for 5.2% of those skin cancers encountered and managed across TRICARE-eligible beneficiaries. For MM, BCC, and SCC, percentages of DC were 5.4%, 5.8%, and 3.5%, respectively. Although it is interesting to note the lower percentage of SCC managed via DC, our data did not allow for extrapolation as to why more SCC cases may be deferred to network. The shift to PC may align with DoD initiatives to increase the private sector’s involvement in military medicine and transition to civilianizing the MHS.14 In the end, the findings are remarkable, with approximately 95% of skin cancer care and management provided overall via PC.
These findings differ from previously published data regarding DC and PC from other specialty areas. Results from an analysis of DC vs PC for plastic surgery for the entire MHS from 2016 to 2019 found 83.2% of cases were deferred to network.15 A similar publication in the orthopedics literature examined TRICARE claims for patients who underwent total hip or knee arthroplasties between 2006 and 2019 and found 84.6% of cases were referred for PC. Notably, the authors utilized generalized linear models for cost analysis and found that DC was more expensive than PC, though this likely was a result of higher rates of hospital readmission within DC cases.16 Lastly, an article on the DC vs PC disposition of MHS patients with breast cancer from 2003 to 2008 found 46% of cases managed with DC vs 26.% with PC and 27.8% receiving a combination. In this case, the authors found a reduced cost associated with DC vs PC.17
Little additional literature exists regarding the costs of DC vs PC. An article published in 2016 designed to assess costs of DC vs PC showed that almost all military treatment facilities have higher costs than their private sector counterparts, with a few exceptions.18 This does not assess the costs of specific procedures, however, and only the overall cost to maintain a treatment facility. Importantly, this study was based on data from FY 2014 and has not been updated to reflect complex changes within the MHS system and the private health care system. Indeed, a US Government Accountability Office FY 2023 study highlighted staffing and efficiency issues within this transition to civilian medicine; subsequently, the 2024 President’s Budget suspended all planned clinical medical military end strength divestitures, underscoring the potential ineffectiveness of a civilianized MHS at meeting the health care needs of its beneficiaries.19,20 Future research on a national scale will be necessary to see if there is a reversal of this trend to PC and if doing so has any impact on access to DC for active-duty troops or active-duty dependents.
In addition to PC vs DC trends, we also can get a sense of the impact of the COVID pandemic restrictions on access to DC vs PC by assessing the change in rates seen in the data from the pre-COVID years (2017-2019) to the “post-COVID” years (2020-2022) included. Overall, rates of DC decreased uniformly from their already low percentages. In our study, rates of DC decreased from 5.8% in 2019 to 4.8% in 2022 for MM, from 6.6% to 4.3% for BCC, and from 4.2% to 2.9% for SCC. Although these changes seem small at first, they represent a 30.6% overall decrease in DC for BCC and an overall decrease of 55.4% in DC for SCC. Although our data do not allow us to extrapolate the real cost of this reduction across a nationwide health care system and more than 5 million care encounters, the financial and personal (ie, lost man-hours) costs of this decrease in DC likely are substantial.
In addition to costs, qualitative aspects that contribute to the burden of skin cancer include treatment-related morbidity, such as scarring, pain, and time spent away from family, work, and hobbies, as well as overall patient satisfaction with the quality of care they receive.21 Future work is critical to assess the real cost of this immense burden of PC for the treatment and management of skin cancers within the DoD beneficiary population.
Limitations—This study is limited by its observational nature. Given the mechanism of our data collection, we may have underestimated disease prevalence, as not all patients are seen for their diagnosis annually. Furthermore, reported demographic strata (eg, age, sex) were limited to those available and valid in the M2 reporting system. Finally, our study only collected data from those service members or former service members seen within the MHS and does not reflect any care rendered to those who are no longer active duty but did not officially retire from the military (ie, nonretired service members receiving care in the Veterans Affairs system for skin cancer).
Conclusion
We describe the annual burden of care for skin cancer in the MHS beneficiary population. Noteworthy findings observed were an overall decrease in beneficiaries being treated for skin cancer through DC; a decreasing annual prevalence of skin cancer diagnosis between 2019 and 2021, which may represent underdiagnosis or decreased follow-up in the setting of the COVID-19 pandemic; and a higher rate of skin cancer in the military beneficiary population compared to the civilian population.
This retrospective observational study investigates skin cancer prevalence and care patterns within the Military Health System (MHS) from 2017 to 2022. Utilizing the MHS Management Analysis and Reporting Tool (most commonly called M2), we analyzed more than 5 million patient encounters and documented skin cancer prevalence in the MHS beneficiary population utilizing available demographic data. Notable findings included an increased prevalence of skin cancer in the military population compared with the civilian population, a substantial decline in direct care (DC) visits at military treatment facilities compared with civilian purchased care (PC) visits, and a decreased total number of visits during COVID-19 restrictions.
The Military Health System (MHS) is a worldwide health care delivery system that serves 9.6 million beneficiaries, including military service members, retirees, and their families.1 Its mission is 2-fold: provide a medically ready force, and provide a medical benefit in keeping with the service and sacrifice of active-duty personnel, military retirees, and their families. For fiscal year (FY) 2022, active-duty service members and their families comprised 16.7% and 19.9% of beneficiaries, respectively, while retired service members and their families comprised 27% and 32% of beneficiaries, respectively.
The MHS operates under the authority of the Department of Defense (DoD) and is supported by an annual budget of approximately $50 billion.1 Health care provision within the MHS is managed by TRICARE regional networks.2 Within these networks, MHS beneficiaries may receive health care in 2 categories: direct care (DC) and purchased care (PC). Direct care is rendered in military treatment facilities by military or civilian providers contracted by the DoD, and PC is administered by civilian providers at civilian health care facilities within the TRICARE network, which is comprised of individual providers, clinics, and hospitals that have agreed to accept TRICARE beneficiaries.1 Purchased care is fee-for-service and paid for by the MHS. Of note, the MHS differs from the Veterans Affairs health care system in that the MHS through DC and PC sees only active-duty service members, active-duty dependents, retirees, and retirees’ dependents (primarily spouses), whereas Veterans Affairs sees only veterans (not necessarily retirees) discharged from military service with compensable medical conditions or disabilities.
Skin cancer presents a notable concern for the US Military, as the risk for skin cancer is thought to be higher than in the general population.3,4 This elevated risk is attributed to numerous factors inherent to active-duty service, including time spent in tropical environments, increased exposure to UV radiation, time spent at high altitudes, and decreased rates of sun-protective behaviors.3 Although numerous studies have explored the mechanisms that contribute to service members’ increased skin cancer risk, there are few (if any) that discuss the burden of skin cancer on the MHS and where its beneficiaries receive their skin cancer care. This study evaluated the burden of skin cancer within the MHS, as demonstrated by the period prevalence of skin cancer among its beneficiaries and the number and distribution of patient visits for skin cancer across both DC and PC from 2017 to 2022.
Methods
Data Collection—This retrospective observational study was designed to describe trends in outpatient visits with a skin cancer diagnosis and annual prevalence of skin cancer types in the MHS. Data are from all MHS beneficiaries who were eligible or enrolled in the analysis year. Our data source was the MHS Management Analysis and Reporting Tool (most commonly called M2), a query tool that contains the current and most recent 5 full FYs of Defense Health Agency corporate health care data including aggregated FY and calendar-year counts of MHS beneficiaries from 2017 to 2022 using encounter and claims data tables from both DC and PC. Data in M2 are coded using a pseudo-person identification number, and queries performed for this study were limited to de-identified visit and patient counts.
Skin cancer diagnoses were defined by relevant International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes recorded from outpatient visits in DC and PC. The M2 database was queried to find aggregate counts of visits and unique MHS beneficiaries with one or more diagnoses of a skin cancer type of interest (defined by relevant ICD-10-CM code) over the study period stratified by year and by patient demographic characteristics. Skin cancer types by ICD-10-CM code group included basal cell carcinoma (BCC), squamous cell carcinoma (SCC), malignant melanoma (MM), and other (including Merkel cell carcinoma and sebaceous carcinoma). Demographic strata included age, sex, military status (active duty, dependents of active duty, retired, or all others), sponsor military rank, and sponsor branch (army, air force, marine corps, or navy). Visit counts included diagnoses from any ICD position (for encounters that contained multiple ICD codes) to describe the total volume of care that addressed a diagnosed skin cancer. Counts of unique patients in prevalence analyses included relevant diagnoses in the primary ICD position only to increase the specificity of prevalence estimates.
Data Analysis—Descriptive analyses included the total number of outpatient visits with a skin cancer diagnosis in DC and PC over the study period, with percentages of total visits by year and by demographic strata. Separate analyses estimated annual prevalences of skin cancer types in the MHS by study year and within 2022 by demographic strata. Numerators in prevalence analyses were defined as the number of unique individuals with one or more relevant ICD codes in the analysis year. Denominators were defined as the total number of MHS beneficiaries in the analysis year and resulting period prevalences reported. Observed prevalences were qualitatively described, and trends were compared with prevalences in nonmilitary populations reported in the literature.
Ethics—This study was conducted as part of a study using secondary analyses of de-identified data from the M2 database. The study was reviewed and approved by the Walter Reed National Military Medical Center institutional review board.

Results
Encounter data were analyzed from a total of 5,374,348 visits between DC and PC over the study period for each cancer type of interest. Figures 1 and 2 show temporal trends in DC visits compared with PC visits in each beneficiary category. The percentage of total DC visits subsequently declined each year throughout the study period, with percentage decreases from 2017 to 2022 of 1.45% or 8200 fewer visits for MM, 3.41% or 7280 fewer visits for BCC, and 2.26% or 3673 fewer visits for SCC.

When stratified by beneficiary category, this trend remained consistent among dependents and retirees, with the most notable annual percentage decrease from 2019 to 2020. A higher proportion of younger adults and active-duty beneficiaries was seen in DC relative to PC, in which most visits were among retirees and others (primarily dependents of retirees, survivors, and Guard/Reserve on active duty, as well as inactive Guard/Reserve). No linear trends over time were apparent for active duty in DC and for dependents and retirees in PC. eTable 1 summarizes the demographic characteristics of MHS beneficiaries being seen in DC and PC over the study period for each cancer type of interest.

The Table shows the period prevalence of skin cancer diagnoses within the MHS beneficiary population from 2017 to 2022. These data were further analyzed by MM, BCC, and SCC (eTable 2) and demographics of interest for the year 2022. By beneficiary category, the period prevalence of MM was 0.08% in active duty, 0.06% in dependents, 0.48% in others, and 1.10% in retirees; the period prevalence of BCC was 0.12% in active duty, 0.07% in dependents, 0.91% in others, and 2.50% in retirees; and the period prevalence of SCC was 0.02% in active duty, 0.01% in dependents, 0.63% in others, and 1.87% in retirees. By sponsor branch, the period prevalence of MM was 0.35% in the army, 0.62% in the air force, 0.35% in the marine corps, and 0.65% in the navy; the period prevalence of BCC was 0.74% in the army, 1.30% in the air force, 0.74% in the marine corps, and 1.36% in the navy; and the period prevalence of SCC was 0.52% in the army, 0.92% in the air force, 0.51% in the marine corps, and 0.97% in the navy.


Comment
This study aimed to provide insight into the burden of skin cancer within the MHS beneficiary population and to identify temporal trends in where these beneficiaries receive their care. We examined patient encounter data from more than 9.6 million MHS beneficiaries.
The utilization of ICD codes from patient encounters to estimate the prevalence of nonmelanoma skin cancer (NMSC) has demonstrated a high positive predictive value. In one study, NMSC cases were confirmed in 96.5% of ICD code–identified patients.5 We presented an extensive collection of epidemiologic data on BCC and SCC, which posed unique challenges for tracking, as they are not reported to or monitored by cancer registries such as the Surveillance, Epidemiology, and End Results (SEER) Program.6
MHS Compared to the US Population—A study using the Global Burden of Disease 2019 database revealed an increasing trend in the incidence and prevalence of NMSC and melanoma since 1990. The same study found the period prevalence in 2019 of MM, SCC, and BCC in the general US population to be 0.13%, 0.31%, and 0.05%, respectively.7 In contrast, among MHS beneficiaries, we observed a higher prevalence in the same year, with figures of 0.66% for MM, 0.72% for SCC, and 1.02% for BCC. According to the SEER database, the period prevalence of MM within the general US population in 2020 was 0.4%.8 That same year, we identified a higher period prevalence of MM—0.54%—within the MHS beneficiary population. Specifically, within the MHS retiree population, the prevalence in 2022 was double that of the general MHS population, with a rate of 1.10%, underscoring the importance of skin cancer screening in older, at-risk adult populations. Prior studies similarly found increased rates of skin cancer within the military beneficiary population. Further studies are needed to compare age-adjusted rates in the MHS vs US population.9-11
COVID-19 Trends—Our data showed an overall decreasing prevalence of skin cancer in the MHS from 2019 to 2021. We suspect that the apparent decrease in skin cancer prevalence may be attributed to underdiagnosis from COVID-19 pandemic restrictions. During that time, many dermatology clinics at military treatment facilities underwent temporary closures, and some dermatologists were sent on nondermatologic utilization tours. Likewise, a US multi-institutional study described declining rates of new melanomas from 2020 to 2021, with an increased proportion of patient presentations with advanced melanoma, suggesting an underdiagnosis of melanoma cases during pandemic restrictions. That study also noted an increased rate of patient-identified melanomas and a decreased rate of provider-identified melanomas during that time.12 Contributing factors may include excess hospital demand, increased patient complexity and acute care needs, and long outpatient clinic backlogs during this time.13Financial Burden—Over our 5-year study period, there were 5,374,348 patient encounters addressing skin cancer, both in DC and PC (Figures 1 and 2; eTable 1). In 2016 to 2018, the average annual cost of treating skin cancer in the US civilian, noninstitutionalized population was $1243 for NMSC (BCC and SCC) and $2430 for melanoma.6 Using this metric, the estimated total cost of care rendered in the MHS in 2018 for NMSC and melanoma was $202,510,803 and $156,516,300, respectively.
Trends in DC vs PC—In the years examined, we found a notable decrease in the number of beneficiaries receiving treatment for MM, BCC, and SCC in DC. Simultaneously, there has been an increase in the number of beneficiaries receiving PC for BCC and SCC, though this trend was not apparent for MM.
Our data provided interesting insights into the percentage of PC compared with DC offered within the MHS. Importantly, our findings suggested that the majority of skin cancer in active-duty service members is managed with DC within the military treatment facility setting (61% DC management over the period analyzed). This finding was true across all years of data analyzed, suggesting that the COVID-19 pandemic did not result in a quantifiable shift in care of skin cancer within the active-duty component to outside providers. One of the critical roles of dermatologists in the MHS is to diagnose and treat skin cancer, and our study suggested that the current global manning and staffing for MHS dermatologists may not be sufficient to meet the burden of skin cancers encountered within our active-duty troops, as only 61% are managed with DC. In particular, service members in more austere and/or overseas locations may not have ready access to a dermatologist.
The burden of skin cancer shifts dramatically when analyzing care of all other populations included in these data, including dependents of active-duty service members, retirees, and the category of “other” (ie, principally dependents of retirees). Within these populations, the rate of DC falls to 30%, with 70% of active-duty dependent care being deferred to network. The findings are even more noticeable for retirees and others within these 2 cohorts in all types of skin cancer analyzed, where DC only accounted for 5.2% of those skin cancers encountered and managed across TRICARE-eligible beneficiaries. For MM, BCC, and SCC, percentages of DC were 5.4%, 5.8%, and 3.5%, respectively. Although it is interesting to note the lower percentage of SCC managed via DC, our data did not allow for extrapolation as to why more SCC cases may be deferred to network. The shift to PC may align with DoD initiatives to increase the private sector’s involvement in military medicine and transition to civilianizing the MHS.14 In the end, the findings are remarkable, with approximately 95% of skin cancer care and management provided overall via PC.
These findings differ from previously published data regarding DC and PC from other specialty areas. Results from an analysis of DC vs PC for plastic surgery for the entire MHS from 2016 to 2019 found 83.2% of cases were deferred to network.15 A similar publication in the orthopedics literature examined TRICARE claims for patients who underwent total hip or knee arthroplasties between 2006 and 2019 and found 84.6% of cases were referred for PC. Notably, the authors utilized generalized linear models for cost analysis and found that DC was more expensive than PC, though this likely was a result of higher rates of hospital readmission within DC cases.16 Lastly, an article on the DC vs PC disposition of MHS patients with breast cancer from 2003 to 2008 found 46% of cases managed with DC vs 26.% with PC and 27.8% receiving a combination. In this case, the authors found a reduced cost associated with DC vs PC.17
Little additional literature exists regarding the costs of DC vs PC. An article published in 2016 designed to assess costs of DC vs PC showed that almost all military treatment facilities have higher costs than their private sector counterparts, with a few exceptions.18 This does not assess the costs of specific procedures, however, and only the overall cost to maintain a treatment facility. Importantly, this study was based on data from FY 2014 and has not been updated to reflect complex changes within the MHS system and the private health care system. Indeed, a US Government Accountability Office FY 2023 study highlighted staffing and efficiency issues within this transition to civilian medicine; subsequently, the 2024 President’s Budget suspended all planned clinical medical military end strength divestitures, underscoring the potential ineffectiveness of a civilianized MHS at meeting the health care needs of its beneficiaries.19,20 Future research on a national scale will be necessary to see if there is a reversal of this trend to PC and if doing so has any impact on access to DC for active-duty troops or active-duty dependents.
In addition to PC vs DC trends, we also can get a sense of the impact of the COVID pandemic restrictions on access to DC vs PC by assessing the change in rates seen in the data from the pre-COVID years (2017-2019) to the “post-COVID” years (2020-2022) included. Overall, rates of DC decreased uniformly from their already low percentages. In our study, rates of DC decreased from 5.8% in 2019 to 4.8% in 2022 for MM, from 6.6% to 4.3% for BCC, and from 4.2% to 2.9% for SCC. Although these changes seem small at first, they represent a 30.6% overall decrease in DC for BCC and an overall decrease of 55.4% in DC for SCC. Although our data do not allow us to extrapolate the real cost of this reduction across a nationwide health care system and more than 5 million care encounters, the financial and personal (ie, lost man-hours) costs of this decrease in DC likely are substantial.
In addition to costs, qualitative aspects that contribute to the burden of skin cancer include treatment-related morbidity, such as scarring, pain, and time spent away from family, work, and hobbies, as well as overall patient satisfaction with the quality of care they receive.21 Future work is critical to assess the real cost of this immense burden of PC for the treatment and management of skin cancers within the DoD beneficiary population.
Limitations—This study is limited by its observational nature. Given the mechanism of our data collection, we may have underestimated disease prevalence, as not all patients are seen for their diagnosis annually. Furthermore, reported demographic strata (eg, age, sex) were limited to those available and valid in the M2 reporting system. Finally, our study only collected data from those service members or former service members seen within the MHS and does not reflect any care rendered to those who are no longer active duty but did not officially retire from the military (ie, nonretired service members receiving care in the Veterans Affairs system for skin cancer).
Conclusion
We describe the annual burden of care for skin cancer in the MHS beneficiary population. Noteworthy findings observed were an overall decrease in beneficiaries being treated for skin cancer through DC; a decreasing annual prevalence of skin cancer diagnosis between 2019 and 2021, which may represent underdiagnosis or decreased follow-up in the setting of the COVID-19 pandemic; and a higher rate of skin cancer in the military beneficiary population compared to the civilian population.
- US Department of Defense. Military health. Accessed October 5, 2023. https://www.defense.gov/
- Wooten NR, Brittingham JA, Pitner RO, et al. Purchased behavioral health care received by Military Health System beneficiaries in civilian medical facilities, 2000-2014. Mil Med. 2018;183:E278-E290. doi:10.1093/milmed/usx101
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- American Academy of Dermatology. Skin cancer. Updated April 22, 2022. Accessed April 17, 2024. https://www.aad.org/media/stats-skin-cancer
- Eide MJ, Krajenta R, Johnson D, et al. Identification of patients with nonmelanoma skin cancer using health maintenance organization claims data. Am J Epidemiol. 2010;171:123-128. doi:10.1093/aje/kwp352
- Kao SYZ, Ekwueme DU, Holman DM, et al. Economic burden of skin cancer treatment in the USA: an analysis of the Medical Expenditure Panel Survey Data, 2012-2018. Cancer Causes Control. 2023;34:205-212. doi:10.1007/s10552-022-01644-0
- Aggarwal P, Knabel P, Fleischer AB. United States burden of melanoma and non-melanoma skin cancer from 1990 to 2019. J Am Acad Dermatol. 2021;85:388-395. doi:10.1016/j.jaad.2021.03.109
- SEER*Explorer. SEER Incidence Data, November 2023 Submission (1975-2021). National Cancer Institute; 2024. Accessed April 17, 2024. https://seer.cancer.gov/statistics-network/explorer/application.html?site=53&data_type=1&graph_type=1&compareBy=sex&chk_sex_1=1&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=1&age_range=1&advopt_precision=1&advopt_show_ci=on&hdn_view=1&advopt_show_apc=on&advopt_display=1
- Brown J, Kopf AW, Rigel DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663. doi:10.1111/j.1365-4362.1984.tb01228.x
- Page WF, Whiteman D, Murphy M. A comparison of melanoma mortality among WWII veterans of the Pacific and European theaters. Ann Epidemiol. 2000;10:192-195. doi:10.1016/s1047-2797(99)00050-2
- Ramani ML, Bennett RG. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J Am Acad Dermatol. 1993;28:733-737. doi:10.1016/0190-9622(93)70102-Y
- Trepanowski N, Chang MS, Zhou G, et al. Delays in melanoma presentation during the COVID-19 pandemic: a nationwide multi-institutional cohort study. J Am Acad Dermatol. 2022;87:1217-1219. doi:10.1016/j.jaad.2022.06.031
- Gibbs A. COVID-19 shutdowns caused delays in melanoma diagnoses, study finds. OHSU News. August 4, 2022. Accessed April 17, 2024. https://news.ohsu.edu/2022/08/04/covid-19-shutdowns-caused-delays-in-melanoma-diagnoses-study-finds
- Kime P. Pentagon budget calls for ‘civilianizing’ military hospitals. Military Times. Published February 10, 2020. Accessed April 17, 2024. https://www.militarytimes.com/news/your-military/2020/02/10/pentagon-budget-calls-for-civilianizing-military-hospitals/
- O’Reilly EB, Norris E, Ortiz-Pomales YT, et al. A comparison of direct care at military medical treatment facilities with purchased care in plastic surgery operative volume. Plast Reconstr Surg Glob Open. 2022;10(10 suppl):124-125. doi:10.1097/01.GOX.0000898976.03344.62
- Haag A, Hosein S, Lyon S, et al. Outcomes for arthroplasties in military health: a retrospective analysis of direct versus purchased care. Mil Med. 2023;188(suppl 6):45-51. doi:10.1093/milmed/usac441
- Eaglehouse YL, Georg MW, Richard P, et al. Cost-efficiency of breast cancer care in the US Military Health System: an economic evaluation in direct and purchased care. Mil Med. 2019;184:e494-e501. doi:10.1093/milmed/usz025
- Lurie PM. Comparing the cost of military treatment facilities with private sector care. Institute for Defense Analyses; February 2016. Accessed April 17, 2024. https://www.ida.org/research-and-publications/publications/all/c/co/comparing-the-costs-of-military-treatment-facilities-with-private-sector-care
- Defense Health Program. Fiscal Year (FY) 2024 President’s Budget: Operation and Maintenance Procurement Research, Development, Test and Evaluation. Department of Defense; March 2023. Accessed April 17, 2024. https://comptroller.defense.gov/Portals/45/Documents/defbudget/fy2024/budget_justification/pdfs/09_Defense_Health_Program/00-DHP_Vols_I_II_and_III_PB24.pdf
- US Government Accountability Office. Defense Health Care. DOD should reevaluate market structure for military medical treatment facility management. Published August 21, 2023. Accessed April 17, 2024. https://www.gao.gov/products/gao-23-105441
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- US Department of Defense. Military health. Accessed October 5, 2023. https://www.defense.gov/
- Wooten NR, Brittingham JA, Pitner RO, et al. Purchased behavioral health care received by Military Health System beneficiaries in civilian medical facilities, 2000-2014. Mil Med. 2018;183:E278-E290. doi:10.1093/milmed/usx101
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- American Academy of Dermatology. Skin cancer. Updated April 22, 2022. Accessed April 17, 2024. https://www.aad.org/media/stats-skin-cancer
- Eide MJ, Krajenta R, Johnson D, et al. Identification of patients with nonmelanoma skin cancer using health maintenance organization claims data. Am J Epidemiol. 2010;171:123-128. doi:10.1093/aje/kwp352
- Kao SYZ, Ekwueme DU, Holman DM, et al. Economic burden of skin cancer treatment in the USA: an analysis of the Medical Expenditure Panel Survey Data, 2012-2018. Cancer Causes Control. 2023;34:205-212. doi:10.1007/s10552-022-01644-0
- Aggarwal P, Knabel P, Fleischer AB. United States burden of melanoma and non-melanoma skin cancer from 1990 to 2019. J Am Acad Dermatol. 2021;85:388-395. doi:10.1016/j.jaad.2021.03.109
- SEER*Explorer. SEER Incidence Data, November 2023 Submission (1975-2021). National Cancer Institute; 2024. Accessed April 17, 2024. https://seer.cancer.gov/statistics-network/explorer/application.html?site=53&data_type=1&graph_type=1&compareBy=sex&chk_sex_1=1&chk_sex_3=3&chk_sex_2=2&rate_type=2&race=1&age_range=1&advopt_precision=1&advopt_show_ci=on&hdn_view=1&advopt_show_apc=on&advopt_display=1
- Brown J, Kopf AW, Rigel DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663. doi:10.1111/j.1365-4362.1984.tb01228.x
- Page WF, Whiteman D, Murphy M. A comparison of melanoma mortality among WWII veterans of the Pacific and European theaters. Ann Epidemiol. 2000;10:192-195. doi:10.1016/s1047-2797(99)00050-2
- Ramani ML, Bennett RG. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J Am Acad Dermatol. 1993;28:733-737. doi:10.1016/0190-9622(93)70102-Y
- Trepanowski N, Chang MS, Zhou G, et al. Delays in melanoma presentation during the COVID-19 pandemic: a nationwide multi-institutional cohort study. J Am Acad Dermatol. 2022;87:1217-1219. doi:10.1016/j.jaad.2022.06.031
- Gibbs A. COVID-19 shutdowns caused delays in melanoma diagnoses, study finds. OHSU News. August 4, 2022. Accessed April 17, 2024. https://news.ohsu.edu/2022/08/04/covid-19-shutdowns-caused-delays-in-melanoma-diagnoses-study-finds
- Kime P. Pentagon budget calls for ‘civilianizing’ military hospitals. Military Times. Published February 10, 2020. Accessed April 17, 2024. https://www.militarytimes.com/news/your-military/2020/02/10/pentagon-budget-calls-for-civilianizing-military-hospitals/
- O’Reilly EB, Norris E, Ortiz-Pomales YT, et al. A comparison of direct care at military medical treatment facilities with purchased care in plastic surgery operative volume. Plast Reconstr Surg Glob Open. 2022;10(10 suppl):124-125. doi:10.1097/01.GOX.0000898976.03344.62
- Haag A, Hosein S, Lyon S, et al. Outcomes for arthroplasties in military health: a retrospective analysis of direct versus purchased care. Mil Med. 2023;188(suppl 6):45-51. doi:10.1093/milmed/usac441
- Eaglehouse YL, Georg MW, Richard P, et al. Cost-efficiency of breast cancer care in the US Military Health System: an economic evaluation in direct and purchased care. Mil Med. 2019;184:e494-e501. doi:10.1093/milmed/usz025
- Lurie PM. Comparing the cost of military treatment facilities with private sector care. Institute for Defense Analyses; February 2016. Accessed April 17, 2024. https://www.ida.org/research-and-publications/publications/all/c/co/comparing-the-costs-of-military-treatment-facilities-with-private-sector-care
- Defense Health Program. Fiscal Year (FY) 2024 President’s Budget: Operation and Maintenance Procurement Research, Development, Test and Evaluation. Department of Defense; March 2023. Accessed April 17, 2024. https://comptroller.defense.gov/Portals/45/Documents/defbudget/fy2024/budget_justification/pdfs/09_Defense_Health_Program/00-DHP_Vols_I_II_and_III_PB24.pdf
- US Government Accountability Office. Defense Health Care. DOD should reevaluate market structure for military medical treatment facility management. Published August 21, 2023. Accessed April 17, 2024. https://www.gao.gov/products/gao-23-105441
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
PRACTICE POINTS
- Study data showed an overall decreasing prevalence of skin cancer in the Military Health System (MHS) from 2019 to 2021, possibly attributable to underdiagnosis resulting from the COVID-19 pandemic. Providers should be mindful of this trend when screening patients who have experienced interruptions in care.
- An overall increased prevalence of skin cancer was noted in the military beneficiary population compared with publicly available civilian data—and thus this diagnosis should be given special consideration within this population.