User login
Trans teens less likely to commit acts of sexual violence, says new study
Transgender and nonbinary adolescents are twice as likely to experience sexual violence as their cisgendered peers but are less likely to attempt rape or commit sexual assault, researchers have found.
The study, which was published online in JAMA Network Open, is among the first on the sexual violence that trans, nonbinary, and other gender nonconforming adolescents experience. Previous studies have focused on adults.
“In the busy world of clinical care, it is essential that clinicians be aware of potential disparities their patients are navigating,” said Michele Ybarra, PhD, MPH, president and research director of the Center for Innovative Public Health Research, San Clemente, California, who led the study. “This includes sexual violence victimization for gender minority youth and the need to talk about consent and boundaries for youth of all genders.”
Dr. Ybarra said that while clinicians may be aware that transgender young people face stigma, discrimination, and bullying, they may not be aware that trans youth are also the targets of sexual violence.
Studies indicate that health care providers and communities have significant misconceptions about sexually explicit behavior among trans and nonbinary teens. Misconceptions can lead to discrimination, resulting in higher rates of drug abuse, dropping out of school, suicide, and homelessness.
Dr. Ybarra and her colleagues surveyed 911 trans, nonbinary, or questioning youth on Instagram and Facebook through a collaboration with Growing Up With Media, a national longitudinal survey designed to investigate sexual violence during adolescence.
They also surveyed 3,282 cisgender persons aged 14-16 years who were recruited to the study between June 2018 and March 2020. The term “cisgender” refers to youth who identify with their gender at birth.
The questionnaires asked teens about gender identity, race, economic status, and support systems at home. Factors associated with not experiencing sexual violence included having a strong network of friends, family, and educators; involvement in the community; and having people close who affirm their gender identity.
More than three-fourths (78%) of youth surveyed identified as cisgender, 13.9% identified as questioning, and 7.9% identified as transgender.
Roughly two-thirds (67%) of transgender adolescents said they had experienced serious sexual violence, 73% reported experiencing violence in their communities, and 63% said they had been exposed to aggressive behavior. In contrast, 6.7% of trans youth said they had ever committed sexual violence, while 7.4% of cisgender teens surveyed, or 243 students, said they had done so.
“The relative lack of visibility of gender minority youth in sexual violence research is unacceptable,” Dr. Ybarra told this news organization. “To be counted, one needs to be seen. We aimed to start addressing this exclusion with the current study.”
The findings provide a lens into the levels of sexual violence that LGBTQIA+ youth experience and an opportunity to provide more inclusive care, according to Elizabeth Miller, MD, PhD, FSAHM, Distinguished Professor of Pediatrics, director of the Division of Adolescent and Young Adult Medicine, and medical director of community and population health at UPMC Children’s Hospital of Pittsburgh, who was not involved in the study.
“There are unfortunately pervasive and harmful stereotypes in our society about the ‘sexual deviancy’ attributed to LGBTQIA+ individuals,” Dr. Miller told this news organization. “This study adds to the research literature that counters and challenges these harmful – and inaccurate – perceptions.”
Dr. Miller said clinicians can help this population by offering youth accurate information about relevant support and services, including how to help a friend.
Programs that providers could incorporate include gender transformative approaches, which guide youth to examine gender norms and inequities and that develop leadership skills.
Such programs are more common outside the United States and have been shown to decrease LGBTQIA+ youth exposure to sexual violence, she said.
Dr. Miller said more research is needed to understand the contexts in which gender minority youth experience sexual violence to guide prevention efforts: “We need to move beyond individual-focused interventions to considering community-level interventions to create safer and more inclusive spaces for all youth.”
Dr. Miller has received royalties for writing content for UptoDate Wolters Kluwer outside of the current study. Dr. Ybarra has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Transgender and nonbinary adolescents are twice as likely to experience sexual violence as their cisgendered peers but are less likely to attempt rape or commit sexual assault, researchers have found.
The study, which was published online in JAMA Network Open, is among the first on the sexual violence that trans, nonbinary, and other gender nonconforming adolescents experience. Previous studies have focused on adults.
“In the busy world of clinical care, it is essential that clinicians be aware of potential disparities their patients are navigating,” said Michele Ybarra, PhD, MPH, president and research director of the Center for Innovative Public Health Research, San Clemente, California, who led the study. “This includes sexual violence victimization for gender minority youth and the need to talk about consent and boundaries for youth of all genders.”
Dr. Ybarra said that while clinicians may be aware that transgender young people face stigma, discrimination, and bullying, they may not be aware that trans youth are also the targets of sexual violence.
Studies indicate that health care providers and communities have significant misconceptions about sexually explicit behavior among trans and nonbinary teens. Misconceptions can lead to discrimination, resulting in higher rates of drug abuse, dropping out of school, suicide, and homelessness.
Dr. Ybarra and her colleagues surveyed 911 trans, nonbinary, or questioning youth on Instagram and Facebook through a collaboration with Growing Up With Media, a national longitudinal survey designed to investigate sexual violence during adolescence.
They also surveyed 3,282 cisgender persons aged 14-16 years who were recruited to the study between June 2018 and March 2020. The term “cisgender” refers to youth who identify with their gender at birth.
The questionnaires asked teens about gender identity, race, economic status, and support systems at home. Factors associated with not experiencing sexual violence included having a strong network of friends, family, and educators; involvement in the community; and having people close who affirm their gender identity.
More than three-fourths (78%) of youth surveyed identified as cisgender, 13.9% identified as questioning, and 7.9% identified as transgender.
Roughly two-thirds (67%) of transgender adolescents said they had experienced serious sexual violence, 73% reported experiencing violence in their communities, and 63% said they had been exposed to aggressive behavior. In contrast, 6.7% of trans youth said they had ever committed sexual violence, while 7.4% of cisgender teens surveyed, or 243 students, said they had done so.
“The relative lack of visibility of gender minority youth in sexual violence research is unacceptable,” Dr. Ybarra told this news organization. “To be counted, one needs to be seen. We aimed to start addressing this exclusion with the current study.”
The findings provide a lens into the levels of sexual violence that LGBTQIA+ youth experience and an opportunity to provide more inclusive care, according to Elizabeth Miller, MD, PhD, FSAHM, Distinguished Professor of Pediatrics, director of the Division of Adolescent and Young Adult Medicine, and medical director of community and population health at UPMC Children’s Hospital of Pittsburgh, who was not involved in the study.
“There are unfortunately pervasive and harmful stereotypes in our society about the ‘sexual deviancy’ attributed to LGBTQIA+ individuals,” Dr. Miller told this news organization. “This study adds to the research literature that counters and challenges these harmful – and inaccurate – perceptions.”
Dr. Miller said clinicians can help this population by offering youth accurate information about relevant support and services, including how to help a friend.
Programs that providers could incorporate include gender transformative approaches, which guide youth to examine gender norms and inequities and that develop leadership skills.
Such programs are more common outside the United States and have been shown to decrease LGBTQIA+ youth exposure to sexual violence, she said.
Dr. Miller said more research is needed to understand the contexts in which gender minority youth experience sexual violence to guide prevention efforts: “We need to move beyond individual-focused interventions to considering community-level interventions to create safer and more inclusive spaces for all youth.”
Dr. Miller has received royalties for writing content for UptoDate Wolters Kluwer outside of the current study. Dr. Ybarra has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Transgender and nonbinary adolescents are twice as likely to experience sexual violence as their cisgendered peers but are less likely to attempt rape or commit sexual assault, researchers have found.
The study, which was published online in JAMA Network Open, is among the first on the sexual violence that trans, nonbinary, and other gender nonconforming adolescents experience. Previous studies have focused on adults.
“In the busy world of clinical care, it is essential that clinicians be aware of potential disparities their patients are navigating,” said Michele Ybarra, PhD, MPH, president and research director of the Center for Innovative Public Health Research, San Clemente, California, who led the study. “This includes sexual violence victimization for gender minority youth and the need to talk about consent and boundaries for youth of all genders.”
Dr. Ybarra said that while clinicians may be aware that transgender young people face stigma, discrimination, and bullying, they may not be aware that trans youth are also the targets of sexual violence.
Studies indicate that health care providers and communities have significant misconceptions about sexually explicit behavior among trans and nonbinary teens. Misconceptions can lead to discrimination, resulting in higher rates of drug abuse, dropping out of school, suicide, and homelessness.
Dr. Ybarra and her colleagues surveyed 911 trans, nonbinary, or questioning youth on Instagram and Facebook through a collaboration with Growing Up With Media, a national longitudinal survey designed to investigate sexual violence during adolescence.
They also surveyed 3,282 cisgender persons aged 14-16 years who were recruited to the study between June 2018 and March 2020. The term “cisgender” refers to youth who identify with their gender at birth.
The questionnaires asked teens about gender identity, race, economic status, and support systems at home. Factors associated with not experiencing sexual violence included having a strong network of friends, family, and educators; involvement in the community; and having people close who affirm their gender identity.
More than three-fourths (78%) of youth surveyed identified as cisgender, 13.9% identified as questioning, and 7.9% identified as transgender.
Roughly two-thirds (67%) of transgender adolescents said they had experienced serious sexual violence, 73% reported experiencing violence in their communities, and 63% said they had been exposed to aggressive behavior. In contrast, 6.7% of trans youth said they had ever committed sexual violence, while 7.4% of cisgender teens surveyed, or 243 students, said they had done so.
“The relative lack of visibility of gender minority youth in sexual violence research is unacceptable,” Dr. Ybarra told this news organization. “To be counted, one needs to be seen. We aimed to start addressing this exclusion with the current study.”
The findings provide a lens into the levels of sexual violence that LGBTQIA+ youth experience and an opportunity to provide more inclusive care, according to Elizabeth Miller, MD, PhD, FSAHM, Distinguished Professor of Pediatrics, director of the Division of Adolescent and Young Adult Medicine, and medical director of community and population health at UPMC Children’s Hospital of Pittsburgh, who was not involved in the study.
“There are unfortunately pervasive and harmful stereotypes in our society about the ‘sexual deviancy’ attributed to LGBTQIA+ individuals,” Dr. Miller told this news organization. “This study adds to the research literature that counters and challenges these harmful – and inaccurate – perceptions.”
Dr. Miller said clinicians can help this population by offering youth accurate information about relevant support and services, including how to help a friend.
Programs that providers could incorporate include gender transformative approaches, which guide youth to examine gender norms and inequities and that develop leadership skills.
Such programs are more common outside the United States and have been shown to decrease LGBTQIA+ youth exposure to sexual violence, she said.
Dr. Miller said more research is needed to understand the contexts in which gender minority youth experience sexual violence to guide prevention efforts: “We need to move beyond individual-focused interventions to considering community-level interventions to create safer and more inclusive spaces for all youth.”
Dr. Miller has received royalties for writing content for UptoDate Wolters Kluwer outside of the current study. Dr. Ybarra has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Encouraging’ results of baricitinib in juvenile idiopathic arthritis
COPENHAGEN – Baricitinib (Olumiant), a Janus kinase (JAK) inhibitor, significantly increases time to disease flare and decreases frequency of flares in patients with juvenile idiopathic arthritis (JIA), according to the results of a phase 3, placebo-controlled study.
The results support use of baricitinib when biologic or conventional synthetic disease-modifying antirheumatic drugs (DMARDs) fail.
The difference in the proportion of patients who flared between baricitinib and placebo was seen as soon as 4 weeks after half of the patients switched from active drug to placebo, at 3.7% versus 23.5% respectively, reported Athimalaipet Ramanan, MD, from the University of Bristol (England) who presented the findings of the withdrawal, efficacy, and safety study at the annual European Congress of Rheumatology.
“Our patients and parents have been waiting for alternative drugs for JIA, so JAK inhibitors have come at the right time,” he said. “These are really very encouraging findings for families, caregivers, and patients with JIA, to have an effective oral JAK inhibitor for managing these children.”
In reporting the key findings, Dr. Ramanan added that the majority of patients (76%) achieved a JIA-ACR (American College of Rheumatology) 30 score during the 12-week open-label phase and went on to enter the double-blind withdrawal phase of the trial.
Baricitinib 2-mg tablets are already Food and Drug Administration approved for the treatment of adults with moderately to severely active rheumatoid arthritis. This study, sponsored by the drug manufacturer Eli Lilly, aimed to investigate the efficacy and safety in pediatric patients with JIA who have shown an inadequate response to conventional synthetic or biologic DMARDs.
“For juvenile patients we need to make a dose adjustment [from the adult dosing], especially because we don’t have long-term safety data from JAK inhibitors in general,” said Osama Elfayad, MD, rheumatologist from Mouwasat Hospital, Dammam, Saudi Arabia who attended the presentation and commented on the findings.
He emphasized that safety was of primary concern in the pediatric population who have a long life expectancy. “For me it is essential to have good long-term safety data in juvenile patients. If we start with 4 mg and if the patient is controlled, we should shift to 2 mg which will be much better. I understand some clinicians are asking for 1 mg.”
Study details
The study population included patients aged from 2 to 17 years old with extended oligo- or polyarticular JIA, enthesitis-related juvenile idiopathic arthritis (ERA) and juvenile psoriatic arthritis.
The trial was divided into three periods: a 2-week safety assessment, a 12-week open-label lead-in phase, and an up-to 32-week double-blind withdrawal phase. After confirmation of dose and safety, children were enrolled in the open-label phase receiving age-based, oral, once daily doses of baricitinib.
“The primary endpoint is really concerned with the next phase of the study [double-blind withdrawal phase] looking at the proportion of patients who have shown a response at week 12 [achieved JIA-ACR30] but when switched from active drug to placebo have a flare,” explained Dr. Ramanan.
Patients were randomized 1:1 to continuing baricitinib or newly starting placebo until disease flare or up to week 32. The time to flare during the double-blind phase was the primary endpoint, while secondary endpoints included JIA-ACR30/50/70/90 response rates at week 12, and the proportion of patients with a flare during the double-blind phase.
“These secondary endpoints are more relevant to the clinic,” noted Dr. Ramanan.
A total of 219 patients entered the open-label phase, and of these, 163 achieved a JIA-ACR 30. These 163 children entered the double-blind stage and were randomized to baricitinib four times a day (56 completed), or placebo (32 completed).
Two-thirds of patients were female, which is typical of the disease, explained Dr. Ramanan, and over two-thirds were White. “Most patients had had disease for around 4 years, and about half had had prior biologic therapy. About half were on baseline methotrexate and almost one-third had used corticosteroids although at doses of under 0.2mg/kg.
“It’s gratifying to see that over 75% achieved a JIA-ACR 30 [76.3%]. More importantly, two-thirds of the patients have a JIA-ACR 50 [63.5%], and almost half of the patients have a JIA-ACR 70 [46.1%]. This is pretty significant at 12 weeks only,” he remarked.
The key finding, however, was in the withdrawal phase, said Dr. Ramanan. “We see that those patients who had a response at week 12 and were then switched to placebo, about half [50.6%] flared on placebo, compared to only 17% of those who continued with baricitinib. So not only do those who switch to placebo have a higher frequency of flares but they are more likely to flare quickly, as early as 4 weeks.”
With respect to safety, he said: “This shows short-term safety, but what we really need is medium and long-term safety data. It is no surprise that most of the events seen were as expected in children including nasopharyngitis, upper respiratory tract infections, and nausea.”
In the baricitinib versus placebo phase, 4.9% had serious adverse events in the baricitinib group compared to 3.7% in the placebo group. “There was nothing we didn’t expect to see which was mainly infection,” said Dr. Ramanan.
Dr. Elfayad has no disclosures. Professor Ramanan is a consultant for Eli Lilly, Abbvie, Roche, UCB, Novartis, Pfizer, and Sobi. He has received grant/research support from Eli Lilly.
COPENHAGEN – Baricitinib (Olumiant), a Janus kinase (JAK) inhibitor, significantly increases time to disease flare and decreases frequency of flares in patients with juvenile idiopathic arthritis (JIA), according to the results of a phase 3, placebo-controlled study.
The results support use of baricitinib when biologic or conventional synthetic disease-modifying antirheumatic drugs (DMARDs) fail.
The difference in the proportion of patients who flared between baricitinib and placebo was seen as soon as 4 weeks after half of the patients switched from active drug to placebo, at 3.7% versus 23.5% respectively, reported Athimalaipet Ramanan, MD, from the University of Bristol (England) who presented the findings of the withdrawal, efficacy, and safety study at the annual European Congress of Rheumatology.
“Our patients and parents have been waiting for alternative drugs for JIA, so JAK inhibitors have come at the right time,” he said. “These are really very encouraging findings for families, caregivers, and patients with JIA, to have an effective oral JAK inhibitor for managing these children.”
In reporting the key findings, Dr. Ramanan added that the majority of patients (76%) achieved a JIA-ACR (American College of Rheumatology) 30 score during the 12-week open-label phase and went on to enter the double-blind withdrawal phase of the trial.
Baricitinib 2-mg tablets are already Food and Drug Administration approved for the treatment of adults with moderately to severely active rheumatoid arthritis. This study, sponsored by the drug manufacturer Eli Lilly, aimed to investigate the efficacy and safety in pediatric patients with JIA who have shown an inadequate response to conventional synthetic or biologic DMARDs.
“For juvenile patients we need to make a dose adjustment [from the adult dosing], especially because we don’t have long-term safety data from JAK inhibitors in general,” said Osama Elfayad, MD, rheumatologist from Mouwasat Hospital, Dammam, Saudi Arabia who attended the presentation and commented on the findings.
He emphasized that safety was of primary concern in the pediatric population who have a long life expectancy. “For me it is essential to have good long-term safety data in juvenile patients. If we start with 4 mg and if the patient is controlled, we should shift to 2 mg which will be much better. I understand some clinicians are asking for 1 mg.”
Study details
The study population included patients aged from 2 to 17 years old with extended oligo- or polyarticular JIA, enthesitis-related juvenile idiopathic arthritis (ERA) and juvenile psoriatic arthritis.
The trial was divided into three periods: a 2-week safety assessment, a 12-week open-label lead-in phase, and an up-to 32-week double-blind withdrawal phase. After confirmation of dose and safety, children were enrolled in the open-label phase receiving age-based, oral, once daily doses of baricitinib.
“The primary endpoint is really concerned with the next phase of the study [double-blind withdrawal phase] looking at the proportion of patients who have shown a response at week 12 [achieved JIA-ACR30] but when switched from active drug to placebo have a flare,” explained Dr. Ramanan.
Patients were randomized 1:1 to continuing baricitinib or newly starting placebo until disease flare or up to week 32. The time to flare during the double-blind phase was the primary endpoint, while secondary endpoints included JIA-ACR30/50/70/90 response rates at week 12, and the proportion of patients with a flare during the double-blind phase.
“These secondary endpoints are more relevant to the clinic,” noted Dr. Ramanan.
A total of 219 patients entered the open-label phase, and of these, 163 achieved a JIA-ACR 30. These 163 children entered the double-blind stage and were randomized to baricitinib four times a day (56 completed), or placebo (32 completed).
Two-thirds of patients were female, which is typical of the disease, explained Dr. Ramanan, and over two-thirds were White. “Most patients had had disease for around 4 years, and about half had had prior biologic therapy. About half were on baseline methotrexate and almost one-third had used corticosteroids although at doses of under 0.2mg/kg.
“It’s gratifying to see that over 75% achieved a JIA-ACR 30 [76.3%]. More importantly, two-thirds of the patients have a JIA-ACR 50 [63.5%], and almost half of the patients have a JIA-ACR 70 [46.1%]. This is pretty significant at 12 weeks only,” he remarked.
The key finding, however, was in the withdrawal phase, said Dr. Ramanan. “We see that those patients who had a response at week 12 and were then switched to placebo, about half [50.6%] flared on placebo, compared to only 17% of those who continued with baricitinib. So not only do those who switch to placebo have a higher frequency of flares but they are more likely to flare quickly, as early as 4 weeks.”
With respect to safety, he said: “This shows short-term safety, but what we really need is medium and long-term safety data. It is no surprise that most of the events seen were as expected in children including nasopharyngitis, upper respiratory tract infections, and nausea.”
In the baricitinib versus placebo phase, 4.9% had serious adverse events in the baricitinib group compared to 3.7% in the placebo group. “There was nothing we didn’t expect to see which was mainly infection,” said Dr. Ramanan.
Dr. Elfayad has no disclosures. Professor Ramanan is a consultant for Eli Lilly, Abbvie, Roche, UCB, Novartis, Pfizer, and Sobi. He has received grant/research support from Eli Lilly.
COPENHAGEN – Baricitinib (Olumiant), a Janus kinase (JAK) inhibitor, significantly increases time to disease flare and decreases frequency of flares in patients with juvenile idiopathic arthritis (JIA), according to the results of a phase 3, placebo-controlled study.
The results support use of baricitinib when biologic or conventional synthetic disease-modifying antirheumatic drugs (DMARDs) fail.
The difference in the proportion of patients who flared between baricitinib and placebo was seen as soon as 4 weeks after half of the patients switched from active drug to placebo, at 3.7% versus 23.5% respectively, reported Athimalaipet Ramanan, MD, from the University of Bristol (England) who presented the findings of the withdrawal, efficacy, and safety study at the annual European Congress of Rheumatology.
“Our patients and parents have been waiting for alternative drugs for JIA, so JAK inhibitors have come at the right time,” he said. “These are really very encouraging findings for families, caregivers, and patients with JIA, to have an effective oral JAK inhibitor for managing these children.”
In reporting the key findings, Dr. Ramanan added that the majority of patients (76%) achieved a JIA-ACR (American College of Rheumatology) 30 score during the 12-week open-label phase and went on to enter the double-blind withdrawal phase of the trial.
Baricitinib 2-mg tablets are already Food and Drug Administration approved for the treatment of adults with moderately to severely active rheumatoid arthritis. This study, sponsored by the drug manufacturer Eli Lilly, aimed to investigate the efficacy and safety in pediatric patients with JIA who have shown an inadequate response to conventional synthetic or biologic DMARDs.
“For juvenile patients we need to make a dose adjustment [from the adult dosing], especially because we don’t have long-term safety data from JAK inhibitors in general,” said Osama Elfayad, MD, rheumatologist from Mouwasat Hospital, Dammam, Saudi Arabia who attended the presentation and commented on the findings.
He emphasized that safety was of primary concern in the pediatric population who have a long life expectancy. “For me it is essential to have good long-term safety data in juvenile patients. If we start with 4 mg and if the patient is controlled, we should shift to 2 mg which will be much better. I understand some clinicians are asking for 1 mg.”
Study details
The study population included patients aged from 2 to 17 years old with extended oligo- or polyarticular JIA, enthesitis-related juvenile idiopathic arthritis (ERA) and juvenile psoriatic arthritis.
The trial was divided into three periods: a 2-week safety assessment, a 12-week open-label lead-in phase, and an up-to 32-week double-blind withdrawal phase. After confirmation of dose and safety, children were enrolled in the open-label phase receiving age-based, oral, once daily doses of baricitinib.
“The primary endpoint is really concerned with the next phase of the study [double-blind withdrawal phase] looking at the proportion of patients who have shown a response at week 12 [achieved JIA-ACR30] but when switched from active drug to placebo have a flare,” explained Dr. Ramanan.
Patients were randomized 1:1 to continuing baricitinib or newly starting placebo until disease flare or up to week 32. The time to flare during the double-blind phase was the primary endpoint, while secondary endpoints included JIA-ACR30/50/70/90 response rates at week 12, and the proportion of patients with a flare during the double-blind phase.
“These secondary endpoints are more relevant to the clinic,” noted Dr. Ramanan.
A total of 219 patients entered the open-label phase, and of these, 163 achieved a JIA-ACR 30. These 163 children entered the double-blind stage and were randomized to baricitinib four times a day (56 completed), or placebo (32 completed).
Two-thirds of patients were female, which is typical of the disease, explained Dr. Ramanan, and over two-thirds were White. “Most patients had had disease for around 4 years, and about half had had prior biologic therapy. About half were on baseline methotrexate and almost one-third had used corticosteroids although at doses of under 0.2mg/kg.
“It’s gratifying to see that over 75% achieved a JIA-ACR 30 [76.3%]. More importantly, two-thirds of the patients have a JIA-ACR 50 [63.5%], and almost half of the patients have a JIA-ACR 70 [46.1%]. This is pretty significant at 12 weeks only,” he remarked.
The key finding, however, was in the withdrawal phase, said Dr. Ramanan. “We see that those patients who had a response at week 12 and were then switched to placebo, about half [50.6%] flared on placebo, compared to only 17% of those who continued with baricitinib. So not only do those who switch to placebo have a higher frequency of flares but they are more likely to flare quickly, as early as 4 weeks.”
With respect to safety, he said: “This shows short-term safety, but what we really need is medium and long-term safety data. It is no surprise that most of the events seen were as expected in children including nasopharyngitis, upper respiratory tract infections, and nausea.”
In the baricitinib versus placebo phase, 4.9% had serious adverse events in the baricitinib group compared to 3.7% in the placebo group. “There was nothing we didn’t expect to see which was mainly infection,” said Dr. Ramanan.
Dr. Elfayad has no disclosures. Professor Ramanan is a consultant for Eli Lilly, Abbvie, Roche, UCB, Novartis, Pfizer, and Sobi. He has received grant/research support from Eli Lilly.
AT THE EULAR 2022 CONGRESS
Pfizer asks FDA to authorize COVID vaccine for children younger than 5
The FDA has accepted Pfizer’s application for a COVID-19 vaccine for children under age 5, which clears the way for approval and distribution in June.
Pfizer announced June 1 that it completed the application for a three-dose vaccine for kids between 6 months and 5 years old, and the FDA said it received the emergency use application.
Children in this age group – the last to be eligible for COVID-19 vaccines – could begin getting shots as early as June 21, according to White House COVID-19 response coordinator Ashish Jha, MD.
Meanwhile, COVID-19 cases are still high – an average of 100,000 cases a day – but death numbers are about 90% lower than they were when President Joe Biden first took office, Dr. Jha said.
The FDA’s advisory group, the Vaccines and Related Biological Products Advisory Committee, is scheduled to meet June 14 and June 15 to discuss data submitted by both Pfizer and Moderna.
If the FDA gives them the green light, the CDC will then weigh in.
“We know that many, many parents are eager to vaccinate their youngest kids, and it’s important to do this right,” Dr. Jha said at a White House press briefing on June 2. “We expect that vaccinations will begin in earnest as early as June 21 and really roll on throughout that week.”
States can place their orders as early as June 3, Dr. Jha said, and there will initially be 10 million doses available. If the FDA gives emergency use authorization for the vaccines, the government will begin shipping doses to thousands of sites across the country.
“The good news is we have plenty of supply of Pfizer and Moderna vaccines,” Dr. Jha said. “We’ve asked states to distribute to their highest priority sites, serving the highest risk and hardest to reach areas.”
Pfizer’s clinical trials found that three doses of the vaccine for children 6 months to under 5 years were safe and effective and proved to be 80% effective against Omicron.
The FDA announced its meeting information with a conversation about the Moderna vaccine for ages 6-17 scheduled for June 14 and a conversation about the Pfizer and Moderna vaccines for young children scheduled for June 15.
Moderna applied for FDA authorization of its two-dose vaccine for children under age 6 on April 28. The company said the vaccine was 51% effective against infections with symptoms for children ages 6 months to 2 years and 37% effective for ages 2-5.
Pfizer’s 3-microgram dose is one-tenth of its adult dose. Moderna’s 25-microgram dose is one-quarter of its adult dose.
A version of this article first appeared on Medscape.com.
The FDA has accepted Pfizer’s application for a COVID-19 vaccine for children under age 5, which clears the way for approval and distribution in June.
Pfizer announced June 1 that it completed the application for a three-dose vaccine for kids between 6 months and 5 years old, and the FDA said it received the emergency use application.
Children in this age group – the last to be eligible for COVID-19 vaccines – could begin getting shots as early as June 21, according to White House COVID-19 response coordinator Ashish Jha, MD.
Meanwhile, COVID-19 cases are still high – an average of 100,000 cases a day – but death numbers are about 90% lower than they were when President Joe Biden first took office, Dr. Jha said.
The FDA’s advisory group, the Vaccines and Related Biological Products Advisory Committee, is scheduled to meet June 14 and June 15 to discuss data submitted by both Pfizer and Moderna.
If the FDA gives them the green light, the CDC will then weigh in.
“We know that many, many parents are eager to vaccinate their youngest kids, and it’s important to do this right,” Dr. Jha said at a White House press briefing on June 2. “We expect that vaccinations will begin in earnest as early as June 21 and really roll on throughout that week.”
States can place their orders as early as June 3, Dr. Jha said, and there will initially be 10 million doses available. If the FDA gives emergency use authorization for the vaccines, the government will begin shipping doses to thousands of sites across the country.
“The good news is we have plenty of supply of Pfizer and Moderna vaccines,” Dr. Jha said. “We’ve asked states to distribute to their highest priority sites, serving the highest risk and hardest to reach areas.”
Pfizer’s clinical trials found that three doses of the vaccine for children 6 months to under 5 years were safe and effective and proved to be 80% effective against Omicron.
The FDA announced its meeting information with a conversation about the Moderna vaccine for ages 6-17 scheduled for June 14 and a conversation about the Pfizer and Moderna vaccines for young children scheduled for June 15.
Moderna applied for FDA authorization of its two-dose vaccine for children under age 6 on April 28. The company said the vaccine was 51% effective against infections with symptoms for children ages 6 months to 2 years and 37% effective for ages 2-5.
Pfizer’s 3-microgram dose is one-tenth of its adult dose. Moderna’s 25-microgram dose is one-quarter of its adult dose.
A version of this article first appeared on Medscape.com.
The FDA has accepted Pfizer’s application for a COVID-19 vaccine for children under age 5, which clears the way for approval and distribution in June.
Pfizer announced June 1 that it completed the application for a three-dose vaccine for kids between 6 months and 5 years old, and the FDA said it received the emergency use application.
Children in this age group – the last to be eligible for COVID-19 vaccines – could begin getting shots as early as June 21, according to White House COVID-19 response coordinator Ashish Jha, MD.
Meanwhile, COVID-19 cases are still high – an average of 100,000 cases a day – but death numbers are about 90% lower than they were when President Joe Biden first took office, Dr. Jha said.
The FDA’s advisory group, the Vaccines and Related Biological Products Advisory Committee, is scheduled to meet June 14 and June 15 to discuss data submitted by both Pfizer and Moderna.
If the FDA gives them the green light, the CDC will then weigh in.
“We know that many, many parents are eager to vaccinate their youngest kids, and it’s important to do this right,” Dr. Jha said at a White House press briefing on June 2. “We expect that vaccinations will begin in earnest as early as June 21 and really roll on throughout that week.”
States can place their orders as early as June 3, Dr. Jha said, and there will initially be 10 million doses available. If the FDA gives emergency use authorization for the vaccines, the government will begin shipping doses to thousands of sites across the country.
“The good news is we have plenty of supply of Pfizer and Moderna vaccines,” Dr. Jha said. “We’ve asked states to distribute to their highest priority sites, serving the highest risk and hardest to reach areas.”
Pfizer’s clinical trials found that three doses of the vaccine for children 6 months to under 5 years were safe and effective and proved to be 80% effective against Omicron.
The FDA announced its meeting information with a conversation about the Moderna vaccine for ages 6-17 scheduled for June 14 and a conversation about the Pfizer and Moderna vaccines for young children scheduled for June 15.
Moderna applied for FDA authorization of its two-dose vaccine for children under age 6 on April 28. The company said the vaccine was 51% effective against infections with symptoms for children ages 6 months to 2 years and 37% effective for ages 2-5.
Pfizer’s 3-microgram dose is one-tenth of its adult dose. Moderna’s 25-microgram dose is one-quarter of its adult dose.
A version of this article first appeared on Medscape.com.
White children more likely to get imaging in EDs: Study
Non-Hispanic White children were more likely to receive diagnostic imaging at children’s hospitals’ emergency departments across the United States than were Hispanic children and non-Hispanic Black children, according to a large study published in JAMA Network Open.
Researchers found that, the more the percentage of children from minority groups cared for by a hospital increased, the wider the imaging gap between those children and non-Hispanic White children.
The cross-sectional study, led by Margaret E. Samuels-Kalow, MD, MPhil, MSHP, with the department of emergency medicine, Massachusetts General Hospital and Harvard Medical School in Boston, included 38 children’s hospitals and more than 12 million ED visits.
“These findings emphasize the urgent need for interventions at the hospital level to improve equity in imaging in pediatric emergency medicine,” the authors write.
Patients included in the study were younger than 18 and visited an ED from January 2016 through December 2019. Data were pulled from the Pediatric Health Information System.
Of the more than 12 million visits in this study, 3.5 million (28.7%) involved at least one diagnostic imaging test.
Diagnostic imaging was performed in 1.5 million visits (34.2%) for non-Hispanic White children; 790,961 (24.6%) for non-Hispanic Black children; and 907,222 (26.1%) for Hispanic children (P < .001).
Non-Hispanic Black children were consistently less likely to get diagnostic imaging than non-Hispanic White counterparts at every hospital in the study, no matter the imaging modality: radiography, ultrasonography, computed tomography, or magnetic resonance imaging.
Hispanic patients were generally less likely to get imaging than non-Hispanic White patients, though results were less consistent for ultrasound and MRI.
In a sensitivity analysis, when looking at imaging from patients’ first visit across the study cohort, non-Hispanic Black children were significantly less likely to get imaging than non-Hispanic White children (adjusted odds ratio, 0.77; 95% confidence interval, 0.74-0.79).
“This remained significant even after adjustment for a priori specified confounders including hospital propensity to image,” the authors write.
Authors acknowledge that it is possible that some of the differences may be attributable to the patient mix regarding severity of cases or indications for imaging by hospital, but they note that all models were adjusted for diagnosis-related group and other potential confounders.
This study did not assess whether one group is being overtested. Researchers also note that higher rates of imaging do not necessarily indicate higher quality of care.
However, the authors note, previous research has suggested overtesting of non-Hispanic White patients for head CT and chest pain, as well as patterns of overtreatment of non-Hispanic White patients who have bronchiolitis or viral upper respiratory tract infections.
Medell Briggs-Malonson, MD, MPH, chief of health equity, diversity and inclusion for the University of California, Los Angeles, Hospital and Clinic System, who was not part of the study, said in an interview “this all rings true.”
“This is not the first study we have had in either the pediatric or adult populations that shows disparate levels of care as well as health outcomes. Now we are starting to be able to measure it,” she said.
This study is further evidence of medical racism, she says, and highlights that it’s not the hospital choice or the insurance type affecting the numbers, she said.
“When you control for those factors, it looks to be it’s only due to race and that’s because of the very deep levels of implicit bias as well as explicit bias that we still have in our health systems and even in our providers,” said Dr. Briggs-Malonson, who is also an associate professor of emergency medicine at UCLA. “It’s incredibly important to identify and immediately address.”
What can be done?
Changing these patterns starts with knowing the numbers, the authors write.
“Hospitals should measure their own differences in imaging rates and increase awareness of existing areas of differential treatment as a starting point for improvement,” Dr. Samuels-Kalow and coauthors say.
Dr. Briggs-Malonson added that guidelines are very clear about when children should get imaging. Adhering to evidence-based guidelines can help avoid variations in care from external factors.
“If children are not receiving the absolute best comprehensive evaluation in the emergency department that they deserve, we can miss many different illnesses, which can lead to worse outcomes,” she noted.
As for what might motivate lack of imaging, Dr. Briggs-Malonson pointed to longstanding trends of providers thinking complaints raised by minority patients may not be as severe as they report. Conversely, in caring for White patients there may be a feeling that more tests and imaging may be better out of more fear of missing something, she said.
At UCLA, she says, dashboards have been developed to track statistics on care by age, race, ethnicity, language, insurance type, etc., though not specifically in pediatric imaging, to assess and address any care inequities.
Summer L. Kaplan, MD, MS, director of emergency radiology at Children’s Hospital of Philadelphia, who also was not part of the study, said the finding of racial disparities in pediatric ED imaging provides evidence that gaps still exist in providing the best care to all children and families seeking emergency care.
“However, it is important to recognize that more imaging does not equal better care,” she said. “More imaging may be associated with unnecessary, low-value tests that may add radiation and other risks but do not improve care.”
She said higher rates of imaging may occur when patients present early in the course of a disease, when the differential diagnosis remains broad.
If families have delayed seeking care because of time constraints, transportation problems, cost of care, or mistrust of the health system, children may present later in the course of a disease and require less imaging for a diagnosis, she explained.
“This paper offers a valuable look at the inequities that exist in pediatric emergency imaging use, and further research will be essential to understand and address the causes of these differences,” Dr. Kaplan said.
A coauthor reported compensation as a member of a Medical Review Committee for Highmark. Other coauthors reported grants from the U.S. Agency for Healthcare Research and Quality outside the submitted work. Dr. Briggs-Malonson and Dr. Kaplan reported no relevant financial relationships.
Non-Hispanic White children were more likely to receive diagnostic imaging at children’s hospitals’ emergency departments across the United States than were Hispanic children and non-Hispanic Black children, according to a large study published in JAMA Network Open.
Researchers found that, the more the percentage of children from minority groups cared for by a hospital increased, the wider the imaging gap between those children and non-Hispanic White children.
The cross-sectional study, led by Margaret E. Samuels-Kalow, MD, MPhil, MSHP, with the department of emergency medicine, Massachusetts General Hospital and Harvard Medical School in Boston, included 38 children’s hospitals and more than 12 million ED visits.
“These findings emphasize the urgent need for interventions at the hospital level to improve equity in imaging in pediatric emergency medicine,” the authors write.
Patients included in the study were younger than 18 and visited an ED from January 2016 through December 2019. Data were pulled from the Pediatric Health Information System.
Of the more than 12 million visits in this study, 3.5 million (28.7%) involved at least one diagnostic imaging test.
Diagnostic imaging was performed in 1.5 million visits (34.2%) for non-Hispanic White children; 790,961 (24.6%) for non-Hispanic Black children; and 907,222 (26.1%) for Hispanic children (P < .001).
Non-Hispanic Black children were consistently less likely to get diagnostic imaging than non-Hispanic White counterparts at every hospital in the study, no matter the imaging modality: radiography, ultrasonography, computed tomography, or magnetic resonance imaging.
Hispanic patients were generally less likely to get imaging than non-Hispanic White patients, though results were less consistent for ultrasound and MRI.
In a sensitivity analysis, when looking at imaging from patients’ first visit across the study cohort, non-Hispanic Black children were significantly less likely to get imaging than non-Hispanic White children (adjusted odds ratio, 0.77; 95% confidence interval, 0.74-0.79).
“This remained significant even after adjustment for a priori specified confounders including hospital propensity to image,” the authors write.
Authors acknowledge that it is possible that some of the differences may be attributable to the patient mix regarding severity of cases or indications for imaging by hospital, but they note that all models were adjusted for diagnosis-related group and other potential confounders.
This study did not assess whether one group is being overtested. Researchers also note that higher rates of imaging do not necessarily indicate higher quality of care.
However, the authors note, previous research has suggested overtesting of non-Hispanic White patients for head CT and chest pain, as well as patterns of overtreatment of non-Hispanic White patients who have bronchiolitis or viral upper respiratory tract infections.
Medell Briggs-Malonson, MD, MPH, chief of health equity, diversity and inclusion for the University of California, Los Angeles, Hospital and Clinic System, who was not part of the study, said in an interview “this all rings true.”
“This is not the first study we have had in either the pediatric or adult populations that shows disparate levels of care as well as health outcomes. Now we are starting to be able to measure it,” she said.
This study is further evidence of medical racism, she says, and highlights that it’s not the hospital choice or the insurance type affecting the numbers, she said.
“When you control for those factors, it looks to be it’s only due to race and that’s because of the very deep levels of implicit bias as well as explicit bias that we still have in our health systems and even in our providers,” said Dr. Briggs-Malonson, who is also an associate professor of emergency medicine at UCLA. “It’s incredibly important to identify and immediately address.”
What can be done?
Changing these patterns starts with knowing the numbers, the authors write.
“Hospitals should measure their own differences in imaging rates and increase awareness of existing areas of differential treatment as a starting point for improvement,” Dr. Samuels-Kalow and coauthors say.
Dr. Briggs-Malonson added that guidelines are very clear about when children should get imaging. Adhering to evidence-based guidelines can help avoid variations in care from external factors.
“If children are not receiving the absolute best comprehensive evaluation in the emergency department that they deserve, we can miss many different illnesses, which can lead to worse outcomes,” she noted.
As for what might motivate lack of imaging, Dr. Briggs-Malonson pointed to longstanding trends of providers thinking complaints raised by minority patients may not be as severe as they report. Conversely, in caring for White patients there may be a feeling that more tests and imaging may be better out of more fear of missing something, she said.
At UCLA, she says, dashboards have been developed to track statistics on care by age, race, ethnicity, language, insurance type, etc., though not specifically in pediatric imaging, to assess and address any care inequities.
Summer L. Kaplan, MD, MS, director of emergency radiology at Children’s Hospital of Philadelphia, who also was not part of the study, said the finding of racial disparities in pediatric ED imaging provides evidence that gaps still exist in providing the best care to all children and families seeking emergency care.
“However, it is important to recognize that more imaging does not equal better care,” she said. “More imaging may be associated with unnecessary, low-value tests that may add radiation and other risks but do not improve care.”
She said higher rates of imaging may occur when patients present early in the course of a disease, when the differential diagnosis remains broad.
If families have delayed seeking care because of time constraints, transportation problems, cost of care, or mistrust of the health system, children may present later in the course of a disease and require less imaging for a diagnosis, she explained.
“This paper offers a valuable look at the inequities that exist in pediatric emergency imaging use, and further research will be essential to understand and address the causes of these differences,” Dr. Kaplan said.
A coauthor reported compensation as a member of a Medical Review Committee for Highmark. Other coauthors reported grants from the U.S. Agency for Healthcare Research and Quality outside the submitted work. Dr. Briggs-Malonson and Dr. Kaplan reported no relevant financial relationships.
Non-Hispanic White children were more likely to receive diagnostic imaging at children’s hospitals’ emergency departments across the United States than were Hispanic children and non-Hispanic Black children, according to a large study published in JAMA Network Open.
Researchers found that, the more the percentage of children from minority groups cared for by a hospital increased, the wider the imaging gap between those children and non-Hispanic White children.
The cross-sectional study, led by Margaret E. Samuels-Kalow, MD, MPhil, MSHP, with the department of emergency medicine, Massachusetts General Hospital and Harvard Medical School in Boston, included 38 children’s hospitals and more than 12 million ED visits.
“These findings emphasize the urgent need for interventions at the hospital level to improve equity in imaging in pediatric emergency medicine,” the authors write.
Patients included in the study were younger than 18 and visited an ED from January 2016 through December 2019. Data were pulled from the Pediatric Health Information System.
Of the more than 12 million visits in this study, 3.5 million (28.7%) involved at least one diagnostic imaging test.
Diagnostic imaging was performed in 1.5 million visits (34.2%) for non-Hispanic White children; 790,961 (24.6%) for non-Hispanic Black children; and 907,222 (26.1%) for Hispanic children (P < .001).
Non-Hispanic Black children were consistently less likely to get diagnostic imaging than non-Hispanic White counterparts at every hospital in the study, no matter the imaging modality: radiography, ultrasonography, computed tomography, or magnetic resonance imaging.
Hispanic patients were generally less likely to get imaging than non-Hispanic White patients, though results were less consistent for ultrasound and MRI.
In a sensitivity analysis, when looking at imaging from patients’ first visit across the study cohort, non-Hispanic Black children were significantly less likely to get imaging than non-Hispanic White children (adjusted odds ratio, 0.77; 95% confidence interval, 0.74-0.79).
“This remained significant even after adjustment for a priori specified confounders including hospital propensity to image,” the authors write.
Authors acknowledge that it is possible that some of the differences may be attributable to the patient mix regarding severity of cases or indications for imaging by hospital, but they note that all models were adjusted for diagnosis-related group and other potential confounders.
This study did not assess whether one group is being overtested. Researchers also note that higher rates of imaging do not necessarily indicate higher quality of care.
However, the authors note, previous research has suggested overtesting of non-Hispanic White patients for head CT and chest pain, as well as patterns of overtreatment of non-Hispanic White patients who have bronchiolitis or viral upper respiratory tract infections.
Medell Briggs-Malonson, MD, MPH, chief of health equity, diversity and inclusion for the University of California, Los Angeles, Hospital and Clinic System, who was not part of the study, said in an interview “this all rings true.”
“This is not the first study we have had in either the pediatric or adult populations that shows disparate levels of care as well as health outcomes. Now we are starting to be able to measure it,” she said.
This study is further evidence of medical racism, she says, and highlights that it’s not the hospital choice or the insurance type affecting the numbers, she said.
“When you control for those factors, it looks to be it’s only due to race and that’s because of the very deep levels of implicit bias as well as explicit bias that we still have in our health systems and even in our providers,” said Dr. Briggs-Malonson, who is also an associate professor of emergency medicine at UCLA. “It’s incredibly important to identify and immediately address.”
What can be done?
Changing these patterns starts with knowing the numbers, the authors write.
“Hospitals should measure their own differences in imaging rates and increase awareness of existing areas of differential treatment as a starting point for improvement,” Dr. Samuels-Kalow and coauthors say.
Dr. Briggs-Malonson added that guidelines are very clear about when children should get imaging. Adhering to evidence-based guidelines can help avoid variations in care from external factors.
“If children are not receiving the absolute best comprehensive evaluation in the emergency department that they deserve, we can miss many different illnesses, which can lead to worse outcomes,” she noted.
As for what might motivate lack of imaging, Dr. Briggs-Malonson pointed to longstanding trends of providers thinking complaints raised by minority patients may not be as severe as they report. Conversely, in caring for White patients there may be a feeling that more tests and imaging may be better out of more fear of missing something, she said.
At UCLA, she says, dashboards have been developed to track statistics on care by age, race, ethnicity, language, insurance type, etc., though not specifically in pediatric imaging, to assess and address any care inequities.
Summer L. Kaplan, MD, MS, director of emergency radiology at Children’s Hospital of Philadelphia, who also was not part of the study, said the finding of racial disparities in pediatric ED imaging provides evidence that gaps still exist in providing the best care to all children and families seeking emergency care.
“However, it is important to recognize that more imaging does not equal better care,” she said. “More imaging may be associated with unnecessary, low-value tests that may add radiation and other risks but do not improve care.”
She said higher rates of imaging may occur when patients present early in the course of a disease, when the differential diagnosis remains broad.
If families have delayed seeking care because of time constraints, transportation problems, cost of care, or mistrust of the health system, children may present later in the course of a disease and require less imaging for a diagnosis, she explained.
“This paper offers a valuable look at the inequities that exist in pediatric emergency imaging use, and further research will be essential to understand and address the causes of these differences,” Dr. Kaplan said.
A coauthor reported compensation as a member of a Medical Review Committee for Highmark. Other coauthors reported grants from the U.S. Agency for Healthcare Research and Quality outside the submitted work. Dr. Briggs-Malonson and Dr. Kaplan reported no relevant financial relationships.
FROM JAMA NETWORK OPEN
Are teenagers tone deaf?
I suspect that you have heard or read about the recent study in the Journal of Neuroscience that claims to have discovered evidence that as children become teenagers, their brains begin to tune out their mother’s voices. The story appeared in at least 10 Internet news sources including the American Academy of Pediatrics’ daily briefing.
Based on functional MRI studies by a group at Stanford (Calif.) University, the researchers found that while in general, teenagers became more attentive to all voices as they reached puberty, novel voices were favored over the maternal voices that had flooded their environment as younger children. Of course none of this comes as a surprise to anyone who has parented a teenager or spent any time trying to communicate with adolescents. Although we all must be a bit careful not to put too much stock in functional MRI studies, these findings do suggest a physiologic basis for the peer pressure that becomes one of the hallmarks of adolescence. I wouldn’t be surprised if some clever entrepreneur has already begun using MRI to search for just the right tonal qualities that will make the perfect Internet influencer.
But, will these MRI studies help parents who have already thrown up their arms and admitted defeat mumbling, “He’s stopped listening to me?” The more observant parents already realized long ago that their words were often the least effective tools in their tool kit when it comes to modifying behavior.
Just listen in any neighborhood playground or grocery store to how often you hear a parent trying to get a toddler or young child to correct a misbehavior using threats or promises that you and everyone else within earshot knows will never be followed by any consequence. How often do you see a parent modeling behaviors that they expect their children to avoid?
Some more “enlightened” parents will avoid threats and instead attempt to engage in a dialogue with their misbehaving child hoping that a rational discussion with a sleep-deprived toddler in full tantrum mode can convince the youngster to self-correct.
I’m sure you learned and may have even used the playground retort “sticks and stones may break my bones but words will never hurt me.” Of course more untrue words were never spoken. Words can hurt and they can scar. But words and threats can also be hollow and will fall on ears deafened by months and years during which there were no consequences. It is certainly nice to know that there is some physiologic correlation to what we all suspected. The good news is that teenagers are still listening to us, although they are increasingly more interested in what their peers and the rest of the world has to say.
What the study fails to point out is that while teenagers may still be listening to us their behavior is molded not so much by what we say but how we as parents and adults behave. Have we parented in a way in which our words are followed up with appropriate consequences? And, more importantly, have we modeled behavior that matches our words? We need to help parents realize that words can be important but parenting by example is the gold standard.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
I suspect that you have heard or read about the recent study in the Journal of Neuroscience that claims to have discovered evidence that as children become teenagers, their brains begin to tune out their mother’s voices. The story appeared in at least 10 Internet news sources including the American Academy of Pediatrics’ daily briefing.
Based on functional MRI studies by a group at Stanford (Calif.) University, the researchers found that while in general, teenagers became more attentive to all voices as they reached puberty, novel voices were favored over the maternal voices that had flooded their environment as younger children. Of course none of this comes as a surprise to anyone who has parented a teenager or spent any time trying to communicate with adolescents. Although we all must be a bit careful not to put too much stock in functional MRI studies, these findings do suggest a physiologic basis for the peer pressure that becomes one of the hallmarks of adolescence. I wouldn’t be surprised if some clever entrepreneur has already begun using MRI to search for just the right tonal qualities that will make the perfect Internet influencer.
But, will these MRI studies help parents who have already thrown up their arms and admitted defeat mumbling, “He’s stopped listening to me?” The more observant parents already realized long ago that their words were often the least effective tools in their tool kit when it comes to modifying behavior.
Just listen in any neighborhood playground or grocery store to how often you hear a parent trying to get a toddler or young child to correct a misbehavior using threats or promises that you and everyone else within earshot knows will never be followed by any consequence. How often do you see a parent modeling behaviors that they expect their children to avoid?
Some more “enlightened” parents will avoid threats and instead attempt to engage in a dialogue with their misbehaving child hoping that a rational discussion with a sleep-deprived toddler in full tantrum mode can convince the youngster to self-correct.
I’m sure you learned and may have even used the playground retort “sticks and stones may break my bones but words will never hurt me.” Of course more untrue words were never spoken. Words can hurt and they can scar. But words and threats can also be hollow and will fall on ears deafened by months and years during which there were no consequences. It is certainly nice to know that there is some physiologic correlation to what we all suspected. The good news is that teenagers are still listening to us, although they are increasingly more interested in what their peers and the rest of the world has to say.
What the study fails to point out is that while teenagers may still be listening to us their behavior is molded not so much by what we say but how we as parents and adults behave. Have we parented in a way in which our words are followed up with appropriate consequences? And, more importantly, have we modeled behavior that matches our words? We need to help parents realize that words can be important but parenting by example is the gold standard.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
I suspect that you have heard or read about the recent study in the Journal of Neuroscience that claims to have discovered evidence that as children become teenagers, their brains begin to tune out their mother’s voices. The story appeared in at least 10 Internet news sources including the American Academy of Pediatrics’ daily briefing.
Based on functional MRI studies by a group at Stanford (Calif.) University, the researchers found that while in general, teenagers became more attentive to all voices as they reached puberty, novel voices were favored over the maternal voices that had flooded their environment as younger children. Of course none of this comes as a surprise to anyone who has parented a teenager or spent any time trying to communicate with adolescents. Although we all must be a bit careful not to put too much stock in functional MRI studies, these findings do suggest a physiologic basis for the peer pressure that becomes one of the hallmarks of adolescence. I wouldn’t be surprised if some clever entrepreneur has already begun using MRI to search for just the right tonal qualities that will make the perfect Internet influencer.
But, will these MRI studies help parents who have already thrown up their arms and admitted defeat mumbling, “He’s stopped listening to me?” The more observant parents already realized long ago that their words were often the least effective tools in their tool kit when it comes to modifying behavior.
Just listen in any neighborhood playground or grocery store to how often you hear a parent trying to get a toddler or young child to correct a misbehavior using threats or promises that you and everyone else within earshot knows will never be followed by any consequence. How often do you see a parent modeling behaviors that they expect their children to avoid?
Some more “enlightened” parents will avoid threats and instead attempt to engage in a dialogue with their misbehaving child hoping that a rational discussion with a sleep-deprived toddler in full tantrum mode can convince the youngster to self-correct.
I’m sure you learned and may have even used the playground retort “sticks and stones may break my bones but words will never hurt me.” Of course more untrue words were never spoken. Words can hurt and they can scar. But words and threats can also be hollow and will fall on ears deafened by months and years during which there were no consequences. It is certainly nice to know that there is some physiologic correlation to what we all suspected. The good news is that teenagers are still listening to us, although they are increasingly more interested in what their peers and the rest of the world has to say.
What the study fails to point out is that while teenagers may still be listening to us their behavior is molded not so much by what we say but how we as parents and adults behave. Have we parented in a way in which our words are followed up with appropriate consequences? And, more importantly, have we modeled behavior that matches our words? We need to help parents realize that words can be important but parenting by example is the gold standard.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Informal human-milk donation: How to counsel patients
I have become obsessed with the reality that the unprecedented national shortage of formula is keeping some families from adequately feeding their infants and young children. I am deeply concerned, both as a family medicine physician and a new mother, about the heartbreaking stories that I’ve heard from parents of all socioeconomic backgrounds. New mothers, unable to breastfeed for a multitude of reasons, find themselves standing in front of empty store shelves, in tears.
In recent months, many health care providers have had patients disclose that they are diluting ready-to-feed formula or mixing powdered formula with more water than instructed to make it go further. Some parents are giving cow’s milk to their children at too young an age because they can’t find formula. Others are foregoing milk altogether and feeding their children beverages such as juice or soda. All of these practices can threaten a child’s life, growth, and development.
When breastfeeding isn’t possible
We all know that human milk is the optimal, most nutritionally complete food source for newborn babies and infants. It can improve dental health and neurodevelopmental outcomes, as well as reduce the risk for asthma, eczema, diabetes, and obesity. An added benefit during the COVID-19 pandemic has been providing newborn infants with a boost of immunity before they are able to be vaccinated against SARS-CoV-2 infection.
But lactation and breastfeeding aren’t possible for everyone. Earlier this year, when my daughter was born more than a month prematurely, I worried that I would be unable to breastfeed her. The complications of prematurity can interfere with establishing lactation, and my daughter spent some time in the neonatal intensive care unit (NICU), requiring frequent feedings to treat hypoglycemia. She also lacked the muscle strength or coordination to latch on to the breast, so she was fed my colostrum and donor breast milk by bottle.
Not knowing when my mature milk would come in, my family scoured the retail stores for formula while I was still recovering from delivery. My daughter needed a specific type of high-calorie formula for premature infants. Eventually, my mother found one can of this powdered formula. The hospital also sent us home with 16 oz of ready-to-feed samples and enough donor breastmilk to last 24 hours at home. We considered ourselves lucky. The fear and anxiety about being able to feed my baby still stands out in my mind.
Pumping and sharing
Over the next few months, out of necessity, I became an “exclusively pumping” mother. My daughter, unable to latch, drank my pumped milk from a bottle. My body started to produce more milk than she needed in a day. In an effort to pay it forward and to put my extra milk to use, I became a human-milk donor. I underwent rigorous screening, including testing for infectious diseases such as HIV and hepatitis C. I was approved to donate to our local hospital’s milk bank, helping other families in the NICU feed their babies. Through informal connections on the internet, I also provide expressed milk to another mother in the community who is unable to lactate. To date, I’ve donated more than 1,500 oz of human milk (and counting).
The practice of human-milk donation dates back millennia with wet-nursing, when children were breastfed by someone other than their biological mothers: relatives, friends, or even strangers. The first milk bank in the United States opened in Boston in the early 20th century. In 1980, the World Health Organization and the United Nations Children’s Fund released a joint statement supporting the use of human-donor milk as the first alternative if the biological mother is unable to breastfeed. Donor milk is a safe option for families who cannot provide their own human milk to their children.
Human-milk banks
More than 30 nonprofit milk banks now operate in the United States. Because their mission is primarily to meet the needs of sick and hospitalized children rather than the general public, these milk banks are an impractical solution to the national formula shortage. Although families with healthy children can purchase donor milk with a prescription, supplies are scarce, and insurance doesn’t cover the cost.
Milk provided by formal human-milk banks is considered safe. Certain infections such as HIV and hepatitis can be transmitted through human milk. However, milk banks screen their donors and safely pasteurize and store donated breastmilk, following standard protocols. The risk of contracting an illness from banked donor milk is very low. The American Academy of Pediatrics recommends accepting donor milk only from a milk bank.
Informal human-milk donation
An increasingly popular alternative to formal human-milk banks is informal human-milk sharing. But many people, including health care professionals, hold misconceptions about how informal milk donation works. Today’s informal milk donation looks very different from age-old wet-nursing: Moms in support groups, often via social media, are requesting pumped milk from one another. (Note that this definition of “informal human-milk donation” does not include selling or purchasing human milk.)
Although the safety of sharing pumped human milk this way cannot be guaranteed, a harm-reduction approach is warranted, especially in view of the current formula scarcity.
I believe that medical professionals have a responsibility to raise awareness and dispel myths about donor breast milk. Many physicians acknowledge that informal milk sharing is common but rarely recommend it to patients. Whether they are donors or recipients, families who choose to participate need to be educated about how to go about the process as safely as possible.
Patients who are considering accepting informally donated human milk should ask key questions of the donor to gauge the risk of pathogens or other harmful substances being passed to their babies:
- What medications do you take?
- What supplements do you take?
- What recreational drugs do you take?
- Any recent travel?
- Any tattoos and if so, how recent?
- How much alcohol do you drink and how often?
- Have you been diagnosed with any infections?
- Any recent illness?
- How do you pump your breast milk?
- How do you store your breast milk?
- When was the available milk pumped?
We can help families by offering our medical expertise, allowing them to make an informed decision about whether to accept donated human milk. Clinicians can encourage patients and their families to use resources like the Infant Risk Center, which provides evidence-based information about medication safety and breast milk.
If your lactating patient is considering donating milk through informal channels to a family in need, encourage them to be open and honest about their medical history and lifestyle habits. If they cannot be transparent, they should not donate. A mutual level of respect and honesty can ensure the safety of those they hope to help. It is also important to counsel prospective milk donors to notify their milk recipients of any new illnesses, substance use, medications, travel, tattoos, or changes to their medical history.
Finally, encourage lactating patients who are able to do so to donate their extra milk to local nonprofit milk banks to increase the availability of screened, pasteurized breast milk in the community.
As a physician and mother, I hope that U.S. families will be less vulnerable to future formula shortages. Human milk is an ideal food source, but not everyone can lactate. Though not perfect, human milk donated outside of formal milk banks offers a safer alternative to diluting formula or feeding other unsuitable beverages to infants and children. As health care professionals, we need to counsel our patients about how to engage in this practice safely.
Dr. Mieses Malchuk is assistant professor in the department of family medicine at the University of North Carolina at Chapel Hill and a board-certified family physician and attending physician at UNC Health in Chapel Hill. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
I have become obsessed with the reality that the unprecedented national shortage of formula is keeping some families from adequately feeding their infants and young children. I am deeply concerned, both as a family medicine physician and a new mother, about the heartbreaking stories that I’ve heard from parents of all socioeconomic backgrounds. New mothers, unable to breastfeed for a multitude of reasons, find themselves standing in front of empty store shelves, in tears.
In recent months, many health care providers have had patients disclose that they are diluting ready-to-feed formula or mixing powdered formula with more water than instructed to make it go further. Some parents are giving cow’s milk to their children at too young an age because they can’t find formula. Others are foregoing milk altogether and feeding their children beverages such as juice or soda. All of these practices can threaten a child’s life, growth, and development.
When breastfeeding isn’t possible
We all know that human milk is the optimal, most nutritionally complete food source for newborn babies and infants. It can improve dental health and neurodevelopmental outcomes, as well as reduce the risk for asthma, eczema, diabetes, and obesity. An added benefit during the COVID-19 pandemic has been providing newborn infants with a boost of immunity before they are able to be vaccinated against SARS-CoV-2 infection.
But lactation and breastfeeding aren’t possible for everyone. Earlier this year, when my daughter was born more than a month prematurely, I worried that I would be unable to breastfeed her. The complications of prematurity can interfere with establishing lactation, and my daughter spent some time in the neonatal intensive care unit (NICU), requiring frequent feedings to treat hypoglycemia. She also lacked the muscle strength or coordination to latch on to the breast, so she was fed my colostrum and donor breast milk by bottle.
Not knowing when my mature milk would come in, my family scoured the retail stores for formula while I was still recovering from delivery. My daughter needed a specific type of high-calorie formula for premature infants. Eventually, my mother found one can of this powdered formula. The hospital also sent us home with 16 oz of ready-to-feed samples and enough donor breastmilk to last 24 hours at home. We considered ourselves lucky. The fear and anxiety about being able to feed my baby still stands out in my mind.
Pumping and sharing
Over the next few months, out of necessity, I became an “exclusively pumping” mother. My daughter, unable to latch, drank my pumped milk from a bottle. My body started to produce more milk than she needed in a day. In an effort to pay it forward and to put my extra milk to use, I became a human-milk donor. I underwent rigorous screening, including testing for infectious diseases such as HIV and hepatitis C. I was approved to donate to our local hospital’s milk bank, helping other families in the NICU feed their babies. Through informal connections on the internet, I also provide expressed milk to another mother in the community who is unable to lactate. To date, I’ve donated more than 1,500 oz of human milk (and counting).
The practice of human-milk donation dates back millennia with wet-nursing, when children were breastfed by someone other than their biological mothers: relatives, friends, or even strangers. The first milk bank in the United States opened in Boston in the early 20th century. In 1980, the World Health Organization and the United Nations Children’s Fund released a joint statement supporting the use of human-donor milk as the first alternative if the biological mother is unable to breastfeed. Donor milk is a safe option for families who cannot provide their own human milk to their children.
Human-milk banks
More than 30 nonprofit milk banks now operate in the United States. Because their mission is primarily to meet the needs of sick and hospitalized children rather than the general public, these milk banks are an impractical solution to the national formula shortage. Although families with healthy children can purchase donor milk with a prescription, supplies are scarce, and insurance doesn’t cover the cost.
Milk provided by formal human-milk banks is considered safe. Certain infections such as HIV and hepatitis can be transmitted through human milk. However, milk banks screen their donors and safely pasteurize and store donated breastmilk, following standard protocols. The risk of contracting an illness from banked donor milk is very low. The American Academy of Pediatrics recommends accepting donor milk only from a milk bank.
Informal human-milk donation
An increasingly popular alternative to formal human-milk banks is informal human-milk sharing. But many people, including health care professionals, hold misconceptions about how informal milk donation works. Today’s informal milk donation looks very different from age-old wet-nursing: Moms in support groups, often via social media, are requesting pumped milk from one another. (Note that this definition of “informal human-milk donation” does not include selling or purchasing human milk.)
Although the safety of sharing pumped human milk this way cannot be guaranteed, a harm-reduction approach is warranted, especially in view of the current formula scarcity.
I believe that medical professionals have a responsibility to raise awareness and dispel myths about donor breast milk. Many physicians acknowledge that informal milk sharing is common but rarely recommend it to patients. Whether they are donors or recipients, families who choose to participate need to be educated about how to go about the process as safely as possible.
Patients who are considering accepting informally donated human milk should ask key questions of the donor to gauge the risk of pathogens or other harmful substances being passed to their babies:
- What medications do you take?
- What supplements do you take?
- What recreational drugs do you take?
- Any recent travel?
- Any tattoos and if so, how recent?
- How much alcohol do you drink and how often?
- Have you been diagnosed with any infections?
- Any recent illness?
- How do you pump your breast milk?
- How do you store your breast milk?
- When was the available milk pumped?
We can help families by offering our medical expertise, allowing them to make an informed decision about whether to accept donated human milk. Clinicians can encourage patients and their families to use resources like the Infant Risk Center, which provides evidence-based information about medication safety and breast milk.
If your lactating patient is considering donating milk through informal channels to a family in need, encourage them to be open and honest about their medical history and lifestyle habits. If they cannot be transparent, they should not donate. A mutual level of respect and honesty can ensure the safety of those they hope to help. It is also important to counsel prospective milk donors to notify their milk recipients of any new illnesses, substance use, medications, travel, tattoos, or changes to their medical history.
Finally, encourage lactating patients who are able to do so to donate their extra milk to local nonprofit milk banks to increase the availability of screened, pasteurized breast milk in the community.
As a physician and mother, I hope that U.S. families will be less vulnerable to future formula shortages. Human milk is an ideal food source, but not everyone can lactate. Though not perfect, human milk donated outside of formal milk banks offers a safer alternative to diluting formula or feeding other unsuitable beverages to infants and children. As health care professionals, we need to counsel our patients about how to engage in this practice safely.
Dr. Mieses Malchuk is assistant professor in the department of family medicine at the University of North Carolina at Chapel Hill and a board-certified family physician and attending physician at UNC Health in Chapel Hill. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
I have become obsessed with the reality that the unprecedented national shortage of formula is keeping some families from adequately feeding their infants and young children. I am deeply concerned, both as a family medicine physician and a new mother, about the heartbreaking stories that I’ve heard from parents of all socioeconomic backgrounds. New mothers, unable to breastfeed for a multitude of reasons, find themselves standing in front of empty store shelves, in tears.
In recent months, many health care providers have had patients disclose that they are diluting ready-to-feed formula or mixing powdered formula with more water than instructed to make it go further. Some parents are giving cow’s milk to their children at too young an age because they can’t find formula. Others are foregoing milk altogether and feeding their children beverages such as juice or soda. All of these practices can threaten a child’s life, growth, and development.
When breastfeeding isn’t possible
We all know that human milk is the optimal, most nutritionally complete food source for newborn babies and infants. It can improve dental health and neurodevelopmental outcomes, as well as reduce the risk for asthma, eczema, diabetes, and obesity. An added benefit during the COVID-19 pandemic has been providing newborn infants with a boost of immunity before they are able to be vaccinated against SARS-CoV-2 infection.
But lactation and breastfeeding aren’t possible for everyone. Earlier this year, when my daughter was born more than a month prematurely, I worried that I would be unable to breastfeed her. The complications of prematurity can interfere with establishing lactation, and my daughter spent some time in the neonatal intensive care unit (NICU), requiring frequent feedings to treat hypoglycemia. She also lacked the muscle strength or coordination to latch on to the breast, so she was fed my colostrum and donor breast milk by bottle.
Not knowing when my mature milk would come in, my family scoured the retail stores for formula while I was still recovering from delivery. My daughter needed a specific type of high-calorie formula for premature infants. Eventually, my mother found one can of this powdered formula. The hospital also sent us home with 16 oz of ready-to-feed samples and enough donor breastmilk to last 24 hours at home. We considered ourselves lucky. The fear and anxiety about being able to feed my baby still stands out in my mind.
Pumping and sharing
Over the next few months, out of necessity, I became an “exclusively pumping” mother. My daughter, unable to latch, drank my pumped milk from a bottle. My body started to produce more milk than she needed in a day. In an effort to pay it forward and to put my extra milk to use, I became a human-milk donor. I underwent rigorous screening, including testing for infectious diseases such as HIV and hepatitis C. I was approved to donate to our local hospital’s milk bank, helping other families in the NICU feed their babies. Through informal connections on the internet, I also provide expressed milk to another mother in the community who is unable to lactate. To date, I’ve donated more than 1,500 oz of human milk (and counting).
The practice of human-milk donation dates back millennia with wet-nursing, when children were breastfed by someone other than their biological mothers: relatives, friends, or even strangers. The first milk bank in the United States opened in Boston in the early 20th century. In 1980, the World Health Organization and the United Nations Children’s Fund released a joint statement supporting the use of human-donor milk as the first alternative if the biological mother is unable to breastfeed. Donor milk is a safe option for families who cannot provide their own human milk to their children.
Human-milk banks
More than 30 nonprofit milk banks now operate in the United States. Because their mission is primarily to meet the needs of sick and hospitalized children rather than the general public, these milk banks are an impractical solution to the national formula shortage. Although families with healthy children can purchase donor milk with a prescription, supplies are scarce, and insurance doesn’t cover the cost.
Milk provided by formal human-milk banks is considered safe. Certain infections such as HIV and hepatitis can be transmitted through human milk. However, milk banks screen their donors and safely pasteurize and store donated breastmilk, following standard protocols. The risk of contracting an illness from banked donor milk is very low. The American Academy of Pediatrics recommends accepting donor milk only from a milk bank.
Informal human-milk donation
An increasingly popular alternative to formal human-milk banks is informal human-milk sharing. But many people, including health care professionals, hold misconceptions about how informal milk donation works. Today’s informal milk donation looks very different from age-old wet-nursing: Moms in support groups, often via social media, are requesting pumped milk from one another. (Note that this definition of “informal human-milk donation” does not include selling or purchasing human milk.)
Although the safety of sharing pumped human milk this way cannot be guaranteed, a harm-reduction approach is warranted, especially in view of the current formula scarcity.
I believe that medical professionals have a responsibility to raise awareness and dispel myths about donor breast milk. Many physicians acknowledge that informal milk sharing is common but rarely recommend it to patients. Whether they are donors or recipients, families who choose to participate need to be educated about how to go about the process as safely as possible.
Patients who are considering accepting informally donated human milk should ask key questions of the donor to gauge the risk of pathogens or other harmful substances being passed to their babies:
- What medications do you take?
- What supplements do you take?
- What recreational drugs do you take?
- Any recent travel?
- Any tattoos and if so, how recent?
- How much alcohol do you drink and how often?
- Have you been diagnosed with any infections?
- Any recent illness?
- How do you pump your breast milk?
- How do you store your breast milk?
- When was the available milk pumped?
We can help families by offering our medical expertise, allowing them to make an informed decision about whether to accept donated human milk. Clinicians can encourage patients and their families to use resources like the Infant Risk Center, which provides evidence-based information about medication safety and breast milk.
If your lactating patient is considering donating milk through informal channels to a family in need, encourage them to be open and honest about their medical history and lifestyle habits. If they cannot be transparent, they should not donate. A mutual level of respect and honesty can ensure the safety of those they hope to help. It is also important to counsel prospective milk donors to notify their milk recipients of any new illnesses, substance use, medications, travel, tattoos, or changes to their medical history.
Finally, encourage lactating patients who are able to do so to donate their extra milk to local nonprofit milk banks to increase the availability of screened, pasteurized breast milk in the community.
As a physician and mother, I hope that U.S. families will be less vulnerable to future formula shortages. Human milk is an ideal food source, but not everyone can lactate. Though not perfect, human milk donated outside of formal milk banks offers a safer alternative to diluting formula or feeding other unsuitable beverages to infants and children. As health care professionals, we need to counsel our patients about how to engage in this practice safely.
Dr. Mieses Malchuk is assistant professor in the department of family medicine at the University of North Carolina at Chapel Hill and a board-certified family physician and attending physician at UNC Health in Chapel Hill. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The latest on COVID-19 and the heart in children
The 2022 Pediatric Academic Societies meeting included an excellent session on the acute and delayed effects of COVID-19 on children’s hearts. Data on the risk for cardiac injury during acute COVID-19, return-to-play guidelines after COVID-19–related heart injury, and post–vaccine-associated myocarditis were reviewed.
COVID-induced cardiac injury
The risk for COVID-induced cardiac injury is directly associated with age. Recent Centers for Disease Control and Prevention data revealed a “myocarditis or pericarditis” rate in the range of 12-17 cases per 100,000 SARS-CoV-2 infections among male children aged 5-11 years (lower rates for females); the rate jumps to 50-65 cases per 100,000 infections among male children aged 12-17 years. So cardiac injury caused by acute COVID-19 appears rare, but the risk is clearly associated with male sex and adolescent age.
Return to play after COVID-19
Clinicians may be pressed by patients and parents for advice on return to play after illness with COVID-19. In July 2020, the American College of Cardiology published an algorithm that has been adjusted over time, most recently in 2022 by the American Academy of Pediatrics. These algorithms stratify recommendations by degree of illness. One rule of thumb: Patients with severe COVID-19 (ICU care or multisystem inflammatory syndrome in children [MIS-C]) have only one box on the algorithm, and that is to rest for 3-6 months and only return to usual activity after cardiac clearance. Moderate disease (defined as ≥ 4 days of fever > 100.4 °F; ≥ 1 week of myalgia, chills, lethargy, or any non-ICU hospital stay; and no evidence of MIS-C) require undergoing an ECG to look for cardiac dysfunction, followed by at least 10 days of rest if the ECG is negative or referral for cardiac evaluation if either ECG or exam by a pediatric cardiologist is abnormal.
Clinicians can perhaps be more permissible with patients who are younger or who have had less severe disease. For example, if a patient aged younger than 12 years is asymptomatic with routine activity at the time of evaluation, an ECG is not indicated. For patients aged 12-15 years who are asymptomatic at the time of evaluation but participate in a high-intensity sport, clinicians might consider obtaining an ECG. As few as 3 days of rest might be enough for select patients who are asymptomatic at presentation. For other patients, clinicians should work with parents to introduce activity gradually and make it clear to parents that any activity intolerance requires quick reevaluation. On existing athlete registries, no deaths that are attributable to post–COVID-19 cardiac effects have been confirmed in children; however, all data presented during the session were from prior to the Omicron variant surge in early 2022, so more information may be forthcoming.
Considerations for MIS-C
Among children experiencing MIS-C, 35% had ECG changes, 40% exhibited left ventricular systolic or diastolic dysfunction, and 30% had mitral regurgitation, meaning that a large percentage of patients with MIS-C show some degree of cardiac dysfunction. Unfortunately, we are still in the data-gathering phase for long-term outcomes. Functional parameters tend to improve within a week, and most patients will return to normal cardiac function by 3-4 months.
Return to play after MIS-C is quite different from that for acute COVID-19. Patients with MIS-C should be treated much like other patients with myocarditis with an expected return to play in 3-6 months and only after cardiac follow-up. Another good-to-remember recommendation is to delay COVID-19 vaccination for at least 90 days after an episode of MIS-C.
Vaccine-related myocarditis
Once again, older age appears to be a risk factor because most patients with postvaccine myocarditis have been in their mid-teens to early 20s, with events more likely after the second vaccine dose and also more likely in male children (4:1 ratio to female children). No deaths have occurred from postvaccination myocarditis in patients younger than 30 years. Still, many individuals have exhibited residual MRI enhancement in the cardiac tissue for some time after experiencing postvaccination myocarditis; it’s currently unclear whether that has clinical implications. By comparison, CDC data demonstrates convincingly that the risk for cardiac effects is much greater after acute COVID-19 than after COVID-19 vaccination, with risk ratios often higher than 20, depending on age and condition (for example, myocarditis vs. pericarditis). Data are still insufficient to determine whether clinicians should recommend or avoid COVID-19 vaccination in children with congenital heart disease.
In summary, administering COVID-19 vaccines requires a great deal of shared decision-making with parents, and the clinician’s role is to educate parents about all potential risks related to both the vaccine and COVID-19 illness. Research has consistently shown that acute COVID-19 myocarditis and myocarditis associated with MIS-C are much more likely to occur in unvaccinated youth and more likely than postvaccination myocarditis, regardless of age.
William T. Basco, Jr., MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
The 2022 Pediatric Academic Societies meeting included an excellent session on the acute and delayed effects of COVID-19 on children’s hearts. Data on the risk for cardiac injury during acute COVID-19, return-to-play guidelines after COVID-19–related heart injury, and post–vaccine-associated myocarditis were reviewed.
COVID-induced cardiac injury
The risk for COVID-induced cardiac injury is directly associated with age. Recent Centers for Disease Control and Prevention data revealed a “myocarditis or pericarditis” rate in the range of 12-17 cases per 100,000 SARS-CoV-2 infections among male children aged 5-11 years (lower rates for females); the rate jumps to 50-65 cases per 100,000 infections among male children aged 12-17 years. So cardiac injury caused by acute COVID-19 appears rare, but the risk is clearly associated with male sex and adolescent age.
Return to play after COVID-19
Clinicians may be pressed by patients and parents for advice on return to play after illness with COVID-19. In July 2020, the American College of Cardiology published an algorithm that has been adjusted over time, most recently in 2022 by the American Academy of Pediatrics. These algorithms stratify recommendations by degree of illness. One rule of thumb: Patients with severe COVID-19 (ICU care or multisystem inflammatory syndrome in children [MIS-C]) have only one box on the algorithm, and that is to rest for 3-6 months and only return to usual activity after cardiac clearance. Moderate disease (defined as ≥ 4 days of fever > 100.4 °F; ≥ 1 week of myalgia, chills, lethargy, or any non-ICU hospital stay; and no evidence of MIS-C) require undergoing an ECG to look for cardiac dysfunction, followed by at least 10 days of rest if the ECG is negative or referral for cardiac evaluation if either ECG or exam by a pediatric cardiologist is abnormal.
Clinicians can perhaps be more permissible with patients who are younger or who have had less severe disease. For example, if a patient aged younger than 12 years is asymptomatic with routine activity at the time of evaluation, an ECG is not indicated. For patients aged 12-15 years who are asymptomatic at the time of evaluation but participate in a high-intensity sport, clinicians might consider obtaining an ECG. As few as 3 days of rest might be enough for select patients who are asymptomatic at presentation. For other patients, clinicians should work with parents to introduce activity gradually and make it clear to parents that any activity intolerance requires quick reevaluation. On existing athlete registries, no deaths that are attributable to post–COVID-19 cardiac effects have been confirmed in children; however, all data presented during the session were from prior to the Omicron variant surge in early 2022, so more information may be forthcoming.
Considerations for MIS-C
Among children experiencing MIS-C, 35% had ECG changes, 40% exhibited left ventricular systolic or diastolic dysfunction, and 30% had mitral regurgitation, meaning that a large percentage of patients with MIS-C show some degree of cardiac dysfunction. Unfortunately, we are still in the data-gathering phase for long-term outcomes. Functional parameters tend to improve within a week, and most patients will return to normal cardiac function by 3-4 months.
Return to play after MIS-C is quite different from that for acute COVID-19. Patients with MIS-C should be treated much like other patients with myocarditis with an expected return to play in 3-6 months and only after cardiac follow-up. Another good-to-remember recommendation is to delay COVID-19 vaccination for at least 90 days after an episode of MIS-C.
Vaccine-related myocarditis
Once again, older age appears to be a risk factor because most patients with postvaccine myocarditis have been in their mid-teens to early 20s, with events more likely after the second vaccine dose and also more likely in male children (4:1 ratio to female children). No deaths have occurred from postvaccination myocarditis in patients younger than 30 years. Still, many individuals have exhibited residual MRI enhancement in the cardiac tissue for some time after experiencing postvaccination myocarditis; it’s currently unclear whether that has clinical implications. By comparison, CDC data demonstrates convincingly that the risk for cardiac effects is much greater after acute COVID-19 than after COVID-19 vaccination, with risk ratios often higher than 20, depending on age and condition (for example, myocarditis vs. pericarditis). Data are still insufficient to determine whether clinicians should recommend or avoid COVID-19 vaccination in children with congenital heart disease.
In summary, administering COVID-19 vaccines requires a great deal of shared decision-making with parents, and the clinician’s role is to educate parents about all potential risks related to both the vaccine and COVID-19 illness. Research has consistently shown that acute COVID-19 myocarditis and myocarditis associated with MIS-C are much more likely to occur in unvaccinated youth and more likely than postvaccination myocarditis, regardless of age.
William T. Basco, Jr., MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
The 2022 Pediatric Academic Societies meeting included an excellent session on the acute and delayed effects of COVID-19 on children’s hearts. Data on the risk for cardiac injury during acute COVID-19, return-to-play guidelines after COVID-19–related heart injury, and post–vaccine-associated myocarditis were reviewed.
COVID-induced cardiac injury
The risk for COVID-induced cardiac injury is directly associated with age. Recent Centers for Disease Control and Prevention data revealed a “myocarditis or pericarditis” rate in the range of 12-17 cases per 100,000 SARS-CoV-2 infections among male children aged 5-11 years (lower rates for females); the rate jumps to 50-65 cases per 100,000 infections among male children aged 12-17 years. So cardiac injury caused by acute COVID-19 appears rare, but the risk is clearly associated with male sex and adolescent age.
Return to play after COVID-19
Clinicians may be pressed by patients and parents for advice on return to play after illness with COVID-19. In July 2020, the American College of Cardiology published an algorithm that has been adjusted over time, most recently in 2022 by the American Academy of Pediatrics. These algorithms stratify recommendations by degree of illness. One rule of thumb: Patients with severe COVID-19 (ICU care or multisystem inflammatory syndrome in children [MIS-C]) have only one box on the algorithm, and that is to rest for 3-6 months and only return to usual activity after cardiac clearance. Moderate disease (defined as ≥ 4 days of fever > 100.4 °F; ≥ 1 week of myalgia, chills, lethargy, or any non-ICU hospital stay; and no evidence of MIS-C) require undergoing an ECG to look for cardiac dysfunction, followed by at least 10 days of rest if the ECG is negative or referral for cardiac evaluation if either ECG or exam by a pediatric cardiologist is abnormal.
Clinicians can perhaps be more permissible with patients who are younger or who have had less severe disease. For example, if a patient aged younger than 12 years is asymptomatic with routine activity at the time of evaluation, an ECG is not indicated. For patients aged 12-15 years who are asymptomatic at the time of evaluation but participate in a high-intensity sport, clinicians might consider obtaining an ECG. As few as 3 days of rest might be enough for select patients who are asymptomatic at presentation. For other patients, clinicians should work with parents to introduce activity gradually and make it clear to parents that any activity intolerance requires quick reevaluation. On existing athlete registries, no deaths that are attributable to post–COVID-19 cardiac effects have been confirmed in children; however, all data presented during the session were from prior to the Omicron variant surge in early 2022, so more information may be forthcoming.
Considerations for MIS-C
Among children experiencing MIS-C, 35% had ECG changes, 40% exhibited left ventricular systolic or diastolic dysfunction, and 30% had mitral regurgitation, meaning that a large percentage of patients with MIS-C show some degree of cardiac dysfunction. Unfortunately, we are still in the data-gathering phase for long-term outcomes. Functional parameters tend to improve within a week, and most patients will return to normal cardiac function by 3-4 months.
Return to play after MIS-C is quite different from that for acute COVID-19. Patients with MIS-C should be treated much like other patients with myocarditis with an expected return to play in 3-6 months and only after cardiac follow-up. Another good-to-remember recommendation is to delay COVID-19 vaccination for at least 90 days after an episode of MIS-C.
Vaccine-related myocarditis
Once again, older age appears to be a risk factor because most patients with postvaccine myocarditis have been in their mid-teens to early 20s, with events more likely after the second vaccine dose and also more likely in male children (4:1 ratio to female children). No deaths have occurred from postvaccination myocarditis in patients younger than 30 years. Still, many individuals have exhibited residual MRI enhancement in the cardiac tissue for some time after experiencing postvaccination myocarditis; it’s currently unclear whether that has clinical implications. By comparison, CDC data demonstrates convincingly that the risk for cardiac effects is much greater after acute COVID-19 than after COVID-19 vaccination, with risk ratios often higher than 20, depending on age and condition (for example, myocarditis vs. pericarditis). Data are still insufficient to determine whether clinicians should recommend or avoid COVID-19 vaccination in children with congenital heart disease.
In summary, administering COVID-19 vaccines requires a great deal of shared decision-making with parents, and the clinician’s role is to educate parents about all potential risks related to both the vaccine and COVID-19 illness. Research has consistently shown that acute COVID-19 myocarditis and myocarditis associated with MIS-C are much more likely to occur in unvaccinated youth and more likely than postvaccination myocarditis, regardless of age.
William T. Basco, Jr., MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Children & COVID: Rise in new cases slows
New cases of COVID-19 in children climbed for the seventh consecutive week, but the latest increase was the smallest of the seven, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Since the weekly total bottomed out at just under 26,000 in early April, the new-case count has risen by 28.0%, 11.8%, 43.5%, 17.4%, 50%, 14.6%, and 5.0%, based on data from the AAP/CHA weekly COVID-19 report.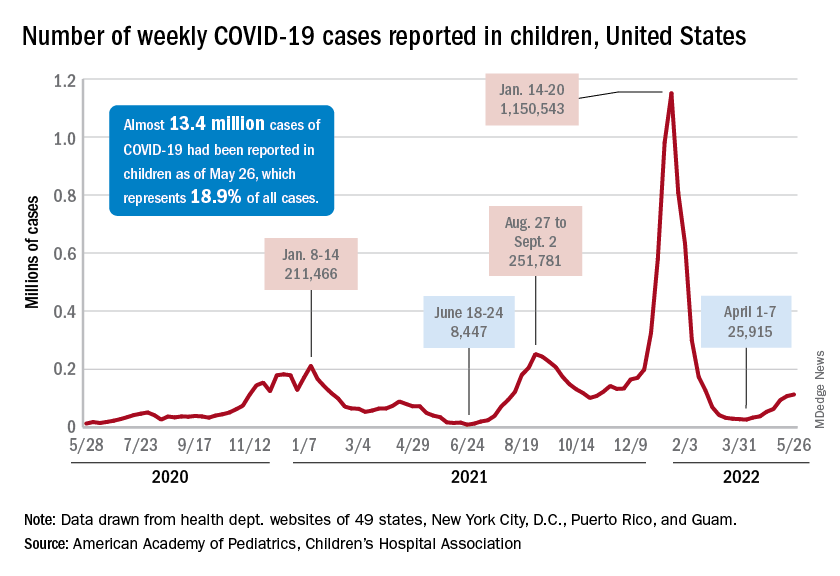
The cumulative number of pediatric cases is almost 13.4 million since the pandemic began, and those infected children represent 18.9% of all cases, the AAP and CHA said based on data from 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
That 18.9% is noteworthy because it marks the first decline in that particular measure since the AAP and CHA started keeping track in April of 2020. Children’s share of the overall COVID burden had been holding at 19.0% for 14 straight weeks, the AAP/CHA data show.
Regionally, new cases were up in the South and the West, where recent rising trends continued, and down in the Midwest and Northeast, where the recent rising trends were reversed for the first time. At the state/territory level, Puerto Rico had the largest percent increase over the last 2 weeks, followed by Maryland and Delaware, the organizations noted in their joint report.
Hospital admissions in children aged 0-17 have changed little in the last week, with the Centers for Disease Control and Prevention reporting rates of 0.25 per 100,000 population on May 23 and 0.25 per 100,000 on May 29, the latest date available. There was, however, a move up to 0.26 per 100,000 from May 24 to May 28, and the CDC acknowledges a possible reporting delay over the most recent 7-day period.
Emergency department visits have dipped slightly in recent days, with children aged 0-11 years at a 7-day average of 2.0% of ED visits with diagnosed COVID on May 28, down from a 5-day stretch at 2.2% from May 19 to May 23. Children aged 12-15 years were at 1.8% on May 28, compared with 2.0% on May 23-24, and 15- to 17-year-olds were at 2.0% on May 28, down from the 2.1% reached over the previous 2 days, the CDC reported on its COVID Data Tracker.
New cases of COVID-19 in children climbed for the seventh consecutive week, but the latest increase was the smallest of the seven, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Since the weekly total bottomed out at just under 26,000 in early April, the new-case count has risen by 28.0%, 11.8%, 43.5%, 17.4%, 50%, 14.6%, and 5.0%, based on data from the AAP/CHA weekly COVID-19 report.
The cumulative number of pediatric cases is almost 13.4 million since the pandemic began, and those infected children represent 18.9% of all cases, the AAP and CHA said based on data from 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
That 18.9% is noteworthy because it marks the first decline in that particular measure since the AAP and CHA started keeping track in April of 2020. Children’s share of the overall COVID burden had been holding at 19.0% for 14 straight weeks, the AAP/CHA data show.
Regionally, new cases were up in the South and the West, where recent rising trends continued, and down in the Midwest and Northeast, where the recent rising trends were reversed for the first time. At the state/territory level, Puerto Rico had the largest percent increase over the last 2 weeks, followed by Maryland and Delaware, the organizations noted in their joint report.
Hospital admissions in children aged 0-17 have changed little in the last week, with the Centers for Disease Control and Prevention reporting rates of 0.25 per 100,000 population on May 23 and 0.25 per 100,000 on May 29, the latest date available. There was, however, a move up to 0.26 per 100,000 from May 24 to May 28, and the CDC acknowledges a possible reporting delay over the most recent 7-day period.
Emergency department visits have dipped slightly in recent days, with children aged 0-11 years at a 7-day average of 2.0% of ED visits with diagnosed COVID on May 28, down from a 5-day stretch at 2.2% from May 19 to May 23. Children aged 12-15 years were at 1.8% on May 28, compared with 2.0% on May 23-24, and 15- to 17-year-olds were at 2.0% on May 28, down from the 2.1% reached over the previous 2 days, the CDC reported on its COVID Data Tracker.
New cases of COVID-19 in children climbed for the seventh consecutive week, but the latest increase was the smallest of the seven, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Since the weekly total bottomed out at just under 26,000 in early April, the new-case count has risen by 28.0%, 11.8%, 43.5%, 17.4%, 50%, 14.6%, and 5.0%, based on data from the AAP/CHA weekly COVID-19 report.
The cumulative number of pediatric cases is almost 13.4 million since the pandemic began, and those infected children represent 18.9% of all cases, the AAP and CHA said based on data from 49 states, New York City, the District of Columbia, Puerto Rico, and Guam.
That 18.9% is noteworthy because it marks the first decline in that particular measure since the AAP and CHA started keeping track in April of 2020. Children’s share of the overall COVID burden had been holding at 19.0% for 14 straight weeks, the AAP/CHA data show.
Regionally, new cases were up in the South and the West, where recent rising trends continued, and down in the Midwest and Northeast, where the recent rising trends were reversed for the first time. At the state/territory level, Puerto Rico had the largest percent increase over the last 2 weeks, followed by Maryland and Delaware, the organizations noted in their joint report.
Hospital admissions in children aged 0-17 have changed little in the last week, with the Centers for Disease Control and Prevention reporting rates of 0.25 per 100,000 population on May 23 and 0.25 per 100,000 on May 29, the latest date available. There was, however, a move up to 0.26 per 100,000 from May 24 to May 28, and the CDC acknowledges a possible reporting delay over the most recent 7-day period.
Emergency department visits have dipped slightly in recent days, with children aged 0-11 years at a 7-day average of 2.0% of ED visits with diagnosed COVID on May 28, down from a 5-day stretch at 2.2% from May 19 to May 23. Children aged 12-15 years were at 1.8% on May 28, compared with 2.0% on May 23-24, and 15- to 17-year-olds were at 2.0% on May 28, down from the 2.1% reached over the previous 2 days, the CDC reported on its COVID Data Tracker.
FDA expands indication for spinal muscular atrophy drug
As previously reported, the FDA first approved oral risdiplam for SMA in children older than age 2 years in 2020.
The FDA expanded the indication for risdiplam to include babies younger than 2 months old because of interim safety and efficacy data from the ongoing RAINBOWFISH study. It includes 25 babies from birth to 6 weeks of age at first dose, all of whom have genetically diagnosed SMA but are not yet presenting with symptoms.
After 12 months of risdiplam treatment, the majority of presymptomatic infants with SMA reached key motor milestones, Genentech said in a news release.
Of the six babies with two or three copies of the SMN2 gene, all were able to sit after 1 year of active treatment, roughly two-thirds could stand, and half could walk independently.
All babies were alive at 12 months without permanent ventilation.
“The approval of Evrysdi for presymptomatic babies is particularly important, as early treatment of SMA, before symptoms start to arise, can help babies to achieve motor milestones,” Richard Finkel, MD, principal investigator of the trial, said in the release.
“With the inclusion of SMA in newborn screening programs, this approval provides the opportunity to start treating at home with Evrysdi soon after the diagnosis is confirmed,” added Dr. Finkel, who is director of the experimental neuroscience program, St. Jude Children’s Research Hospital, Memphis.
From newborns to older adults?
SMA is a rare and often fatal genetic disease that causes muscle weakness and progressive loss of movement.
SMA, which affects about 1 in 10,000 babies, is caused by a mutation in the survival motor neuron 1 (SMN1) gene. The gene encodes the SMN protein, which is critical for the maintenance and function of motor neurons.
Risdiplam is an orally administered, centrally and peripherally distributed small molecule that modulates survival motor neuron 2 (SMN2) premessenger RNA splicing to increase SMN protein levels.
As part of the label extension, the prescribing information for risdiplam has also been updated to include 2-year pooled data from parts 1 and 2 of the FIREFISH study, which demonstrated long-term efficacy and safety in symptomatic infants with Type 1 SMA, the company noted.
“Because of its efficacy in multiple settings, Evrysdi is now available for people with SMA, from presymptomatic newborns to older adults,” Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, said in the release.
“We are proud of this achievement, which has the potential to make a real difference to those living with SMA and their caregivers,” Dr. Garraway added.
A version of this article first appeared on Medscape.com.
As previously reported, the FDA first approved oral risdiplam for SMA in children older than age 2 years in 2020.
The FDA expanded the indication for risdiplam to include babies younger than 2 months old because of interim safety and efficacy data from the ongoing RAINBOWFISH study. It includes 25 babies from birth to 6 weeks of age at first dose, all of whom have genetically diagnosed SMA but are not yet presenting with symptoms.
After 12 months of risdiplam treatment, the majority of presymptomatic infants with SMA reached key motor milestones, Genentech said in a news release.
Of the six babies with two or three copies of the SMN2 gene, all were able to sit after 1 year of active treatment, roughly two-thirds could stand, and half could walk independently.
All babies were alive at 12 months without permanent ventilation.
“The approval of Evrysdi for presymptomatic babies is particularly important, as early treatment of SMA, before symptoms start to arise, can help babies to achieve motor milestones,” Richard Finkel, MD, principal investigator of the trial, said in the release.
“With the inclusion of SMA in newborn screening programs, this approval provides the opportunity to start treating at home with Evrysdi soon after the diagnosis is confirmed,” added Dr. Finkel, who is director of the experimental neuroscience program, St. Jude Children’s Research Hospital, Memphis.
From newborns to older adults?
SMA is a rare and often fatal genetic disease that causes muscle weakness and progressive loss of movement.
SMA, which affects about 1 in 10,000 babies, is caused by a mutation in the survival motor neuron 1 (SMN1) gene. The gene encodes the SMN protein, which is critical for the maintenance and function of motor neurons.
Risdiplam is an orally administered, centrally and peripherally distributed small molecule that modulates survival motor neuron 2 (SMN2) premessenger RNA splicing to increase SMN protein levels.
As part of the label extension, the prescribing information for risdiplam has also been updated to include 2-year pooled data from parts 1 and 2 of the FIREFISH study, which demonstrated long-term efficacy and safety in symptomatic infants with Type 1 SMA, the company noted.
“Because of its efficacy in multiple settings, Evrysdi is now available for people with SMA, from presymptomatic newborns to older adults,” Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, said in the release.
“We are proud of this achievement, which has the potential to make a real difference to those living with SMA and their caregivers,” Dr. Garraway added.
A version of this article first appeared on Medscape.com.
As previously reported, the FDA first approved oral risdiplam for SMA in children older than age 2 years in 2020.
The FDA expanded the indication for risdiplam to include babies younger than 2 months old because of interim safety and efficacy data from the ongoing RAINBOWFISH study. It includes 25 babies from birth to 6 weeks of age at first dose, all of whom have genetically diagnosed SMA but are not yet presenting with symptoms.
After 12 months of risdiplam treatment, the majority of presymptomatic infants with SMA reached key motor milestones, Genentech said in a news release.
Of the six babies with two or three copies of the SMN2 gene, all were able to sit after 1 year of active treatment, roughly two-thirds could stand, and half could walk independently.
All babies were alive at 12 months without permanent ventilation.
“The approval of Evrysdi for presymptomatic babies is particularly important, as early treatment of SMA, before symptoms start to arise, can help babies to achieve motor milestones,” Richard Finkel, MD, principal investigator of the trial, said in the release.
“With the inclusion of SMA in newborn screening programs, this approval provides the opportunity to start treating at home with Evrysdi soon after the diagnosis is confirmed,” added Dr. Finkel, who is director of the experimental neuroscience program, St. Jude Children’s Research Hospital, Memphis.
From newborns to older adults?
SMA is a rare and often fatal genetic disease that causes muscle weakness and progressive loss of movement.
SMA, which affects about 1 in 10,000 babies, is caused by a mutation in the survival motor neuron 1 (SMN1) gene. The gene encodes the SMN protein, which is critical for the maintenance and function of motor neurons.
Risdiplam is an orally administered, centrally and peripherally distributed small molecule that modulates survival motor neuron 2 (SMN2) premessenger RNA splicing to increase SMN protein levels.
As part of the label extension, the prescribing information for risdiplam has also been updated to include 2-year pooled data from parts 1 and 2 of the FIREFISH study, which demonstrated long-term efficacy and safety in symptomatic infants with Type 1 SMA, the company noted.
“Because of its efficacy in multiple settings, Evrysdi is now available for people with SMA, from presymptomatic newborns to older adults,” Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, said in the release.
“We are proud of this achievement, which has the potential to make a real difference to those living with SMA and their caregivers,” Dr. Garraway added.
A version of this article first appeared on Medscape.com.
Mother’s distress disrupts fetal brain development
Babies of mothers who experience significant psychological distress during pregnancy showed evidence of altered brain development in utero and reduced cognitive outcomes at 18 months, based on data from a pair of studies including approximately 300 women.
In a longitudinal study published in JAMA Network Open, Yao Wu, PhD, of Children’s National Hospital, Washington, and colleagues recruited 97 healthy mother-infant dyads between January 2016 and October 2020 at a single center. Of these, 87 underwent two fetal brain imaging studies each, and 10 completed the first MRI visit, for a total of 184 fetal MRIs.
Neurodevelopment and social-emotional development for infants at 18 months of age was measured using the Bayley Scales of Infant and Toddler Development and Infant-Toddler Social and Emotional Assessment. The mean age of the mothers was 35 years; maternal distress was assessed between 24 and 40 weeks’ gestation using validated self-report questionnaires. Parenting stress was assessed at the 18-month infant testing using the Parenting Stress Index-Short Form.
Overall, prenatal maternal stress was negatively associated with infant cognitive performance (P = .01) at 18 months, mediated by fetal left hippocampal volume.
In addition, increased fetal cortical local gyrification index and sulcal depth measured during reported times of prenatal maternal distress were associated with significantly poorer social-emotional scores and competence scores at age 18 months. The beta coefficients for local gyrification index and sulcal depth were –54.62 and –14.22, respectively, for social-emotional and competence scores, –24.01 and –7.53, respectively; P values were P < .001, P < .002, P = .003, P < .001, respectively.
“Increased cortical gyrification has been suggested in children with dyslexia and autism, and sulcal depth has been associated with the severity of impaired performance on working memory and executive function in adults with schizophrenia,” the researchers wrote in their discussion of the findings.
The current study “extends our previous findings and suggests a critical role for disturbances in emerging fetal cerebral cortical folding development in mediating the association between prenatal maternal distress and neurodevelopmental problems that later manifest in infancy,” they explained.
The researchers also found that prenatal maternal anxiety, stress, and depression were positively associated with all measures of parenting stress at the 18-month testing visit.
The study findings were limited by several factors including the use of self-reports for both maternal distress and infant social-emotional assessment, despite the use of validated questionnaires, and the fact that assessment of maternal distress at specific times may not reflect the entire pregnancy, the researchers noted. Other potential limitations included the inability to use some MRI data because of fetal movement and the homogenous population of relatively highly educated women with access to health care that may not reflect other areas, they said.
“Identifying early brain developmental biomarkers may help improve the identification of infants at risk for later neurodevelopmental impairment who might benefit from early targeted interventions,” the researchers concluded.
Technology enhances health and disease models
The effect of the prenatal period on future well-being is recognized, but the current study makes “substantial contributions to prenatal programming science, with implications for ways to transform the prenatal care ecosystem for two-generation impact,” Catherine Monk, PhD, and Cristina R. Fernández, MD, both of Columbia University, New York, wrote in an accompanying editorial.
The developmental origins of health and disease (DOHaD) conceptual model introduced by Dr. David Barker in 1995 were later applied to show that maternal stress, depression, and anxiety affected child prenatal and future development, they said. However, the current study uses cutting-edge neuroscience to directly assess developing fetal brains. The finding of reduced cognitive functioning at 18 months associated with maternal stress is consistent with other findings, they noted.
“Finding an association between maternal prenatal stress and infant cognitive outcomes in the setting of what may be modest stress relative to that of a low-resourced or historically marginalized sample underscores the importance of this research; presumably, with higher stress, and greater social determinants of health burden, the effect sizes would be even greater and of greater concern,” they said.
However, studies such as the current one “have the potential to transform the prenatal and postpartum care ecosystems,” by encouraging a whole-person approach to the care of pregnant women, including attention to mental well-being and quality of life, they emphasized.
COVID-19 stress considerations
In a separate study published in Communications Medicine, Yuan-Chiao Lu, MD, also of Children’s National Hospital in Washington, and colleagues found a similar effect of maternal stress on fetal brain development.
The researchers imaged the brains of fetuses before and during the COVID-19 pandemic and interviewed mothers about any distress they experienced during pregnancy.
The study population included 65 women with known COVID-19 exposures who underwent 92 fetal MRIs and 137 prepandemic controls who underwent 182 fetal MRIs. Maternal distress was measured via the Spielberger State Anxiety Inventory, Spielberger Trait Anxiety Inventory, Perceived Stress Scale, and Edinburgh Postnatal Depression Scale.
Overall, scores on measures of stress and depression were significantly higher for women in the pandemic group compared with controls. Of the 173 women for whom maternal distress measures were available, 28% of the prepandemic group and 52% of the pandemic group met criteria for elevated maternal psychological distress, defined as above the threshold for distress on any one of the four measures.
After the researchers controlled for maternal distress, MRI data showed decreases in fetal white matter and in hippocampal and cerebellar volumes in fetuses in the pandemic group compared with controls.
Other signs of impaired brain development were similar to those seen in the JAMA Network Open study, including decreased cortical surface area and local gyrification index, as well as reduced sulcal depth in multiple brain lobes, indicating delayed cerebral cortical gyrification.
The second study was limited by a lack of data on other lifestyle changes during the pandemic that might influence maternal health and fetal development, the researchers noted. Other limitations were the possible lack of generalizability to a range of racial and ethnic populations and geographic areas outside of Washington, and the inability to control for unknown COVID-19 exposures or subclinical infections in controls, they said.
However, the results support findings from previous studies, and provide a unique opportunity to study the effect of prenatal stress on early development, as well as a chance to implement “novel and timely interventions,” the researchers wrote.
“Monitoring the COVID generation of infants for long-term cognitive and health outcomes after birth is warranted and currently underway,” and continued research may inform preventive strategies for pregnant women experiencing multiple stressors beyond the pandemic, they concluded.
Interpret pandemic effect with caution
“Research studies, as well as our own daily experiences, have made it abundantly clear that stress is on the rise as a consequence of the COVID-19 pandemic,” said editorial author Dr. Monk, who commented on the second study in an interview. “This is an important public health question: Early identification of pandemic effects on child development can help garner the necessary resources to intervene early, dramatically increasing the likelihood of improving that child’s developmental trajectory,” she said.
“The pandemic is an unprecedented experience that has widespread impact on people’s lives, how could it not also alter gestational biology and the developing brain? That being said, we need to be cautious in that we do not yet know the functional implications of these brain changes for longer-term development,” Dr. Monk said. “Also, we do not know what aspects of women’s pandemic-affected lives had an influence on fetal brain development. The authors found higher stress in pandemic versus nonpandemic women, but not evidence that distress was the mediating variable relating pregnancy during the pandemic to altered brain development,” she explained.
The take-home message for clinicians is to “provide your patients with realistic avenues for neurodevelopmental assessments of their children if they, or you, have concerns,” Dr. Monk said. “However, do not prejudge ‘pandemic babies,’ as not all children will be affected by these potential pandemic effects,” she emphasized. “It is possible to misjudge normal variation in children’s development and unnecessarily raise parents’ anxiety levels. Importantly, this period of brain plasticity means any needed intervention likely can have a big, ameliorating impact,” she added.
“We need follow-up studies looking at pandemic effects on prenatal and postnatal development and what factors protect the fetus and birthing person from the negative influences,” she said.
The JAMA study was supported by the National Institutes of Health and the A. James & Alice B. Clark Foundation. The study in Communications Medicine was supported by the National Institutes of Health, the Intellectual and Developmental Disabilities Research Center, and the A. James & Alice B. Clark Foundation. None of the researchers in either study disclosed conflicts of interest. Dr. Monk disclosed grants from the National Institutes of Health, the Bezos Family Foundation, and the Robin Hood Foundation outside the submitted work.
Babies of mothers who experience significant psychological distress during pregnancy showed evidence of altered brain development in utero and reduced cognitive outcomes at 18 months, based on data from a pair of studies including approximately 300 women.
In a longitudinal study published in JAMA Network Open, Yao Wu, PhD, of Children’s National Hospital, Washington, and colleagues recruited 97 healthy mother-infant dyads between January 2016 and October 2020 at a single center. Of these, 87 underwent two fetal brain imaging studies each, and 10 completed the first MRI visit, for a total of 184 fetal MRIs.
Neurodevelopment and social-emotional development for infants at 18 months of age was measured using the Bayley Scales of Infant and Toddler Development and Infant-Toddler Social and Emotional Assessment. The mean age of the mothers was 35 years; maternal distress was assessed between 24 and 40 weeks’ gestation using validated self-report questionnaires. Parenting stress was assessed at the 18-month infant testing using the Parenting Stress Index-Short Form.
Overall, prenatal maternal stress was negatively associated with infant cognitive performance (P = .01) at 18 months, mediated by fetal left hippocampal volume.
In addition, increased fetal cortical local gyrification index and sulcal depth measured during reported times of prenatal maternal distress were associated with significantly poorer social-emotional scores and competence scores at age 18 months. The beta coefficients for local gyrification index and sulcal depth were –54.62 and –14.22, respectively, for social-emotional and competence scores, –24.01 and –7.53, respectively; P values were P < .001, P < .002, P = .003, P < .001, respectively.
“Increased cortical gyrification has been suggested in children with dyslexia and autism, and sulcal depth has been associated with the severity of impaired performance on working memory and executive function in adults with schizophrenia,” the researchers wrote in their discussion of the findings.
The current study “extends our previous findings and suggests a critical role for disturbances in emerging fetal cerebral cortical folding development in mediating the association between prenatal maternal distress and neurodevelopmental problems that later manifest in infancy,” they explained.
The researchers also found that prenatal maternal anxiety, stress, and depression were positively associated with all measures of parenting stress at the 18-month testing visit.
The study findings were limited by several factors including the use of self-reports for both maternal distress and infant social-emotional assessment, despite the use of validated questionnaires, and the fact that assessment of maternal distress at specific times may not reflect the entire pregnancy, the researchers noted. Other potential limitations included the inability to use some MRI data because of fetal movement and the homogenous population of relatively highly educated women with access to health care that may not reflect other areas, they said.
“Identifying early brain developmental biomarkers may help improve the identification of infants at risk for later neurodevelopmental impairment who might benefit from early targeted interventions,” the researchers concluded.
Technology enhances health and disease models
The effect of the prenatal period on future well-being is recognized, but the current study makes “substantial contributions to prenatal programming science, with implications for ways to transform the prenatal care ecosystem for two-generation impact,” Catherine Monk, PhD, and Cristina R. Fernández, MD, both of Columbia University, New York, wrote in an accompanying editorial.
The developmental origins of health and disease (DOHaD) conceptual model introduced by Dr. David Barker in 1995 were later applied to show that maternal stress, depression, and anxiety affected child prenatal and future development, they said. However, the current study uses cutting-edge neuroscience to directly assess developing fetal brains. The finding of reduced cognitive functioning at 18 months associated with maternal stress is consistent with other findings, they noted.
“Finding an association between maternal prenatal stress and infant cognitive outcomes in the setting of what may be modest stress relative to that of a low-resourced or historically marginalized sample underscores the importance of this research; presumably, with higher stress, and greater social determinants of health burden, the effect sizes would be even greater and of greater concern,” they said.
However, studies such as the current one “have the potential to transform the prenatal and postpartum care ecosystems,” by encouraging a whole-person approach to the care of pregnant women, including attention to mental well-being and quality of life, they emphasized.
COVID-19 stress considerations
In a separate study published in Communications Medicine, Yuan-Chiao Lu, MD, also of Children’s National Hospital in Washington, and colleagues found a similar effect of maternal stress on fetal brain development.
The researchers imaged the brains of fetuses before and during the COVID-19 pandemic and interviewed mothers about any distress they experienced during pregnancy.
The study population included 65 women with known COVID-19 exposures who underwent 92 fetal MRIs and 137 prepandemic controls who underwent 182 fetal MRIs. Maternal distress was measured via the Spielberger State Anxiety Inventory, Spielberger Trait Anxiety Inventory, Perceived Stress Scale, and Edinburgh Postnatal Depression Scale.
Overall, scores on measures of stress and depression were significantly higher for women in the pandemic group compared with controls. Of the 173 women for whom maternal distress measures were available, 28% of the prepandemic group and 52% of the pandemic group met criteria for elevated maternal psychological distress, defined as above the threshold for distress on any one of the four measures.
After the researchers controlled for maternal distress, MRI data showed decreases in fetal white matter and in hippocampal and cerebellar volumes in fetuses in the pandemic group compared with controls.
Other signs of impaired brain development were similar to those seen in the JAMA Network Open study, including decreased cortical surface area and local gyrification index, as well as reduced sulcal depth in multiple brain lobes, indicating delayed cerebral cortical gyrification.
The second study was limited by a lack of data on other lifestyle changes during the pandemic that might influence maternal health and fetal development, the researchers noted. Other limitations were the possible lack of generalizability to a range of racial and ethnic populations and geographic areas outside of Washington, and the inability to control for unknown COVID-19 exposures or subclinical infections in controls, they said.
However, the results support findings from previous studies, and provide a unique opportunity to study the effect of prenatal stress on early development, as well as a chance to implement “novel and timely interventions,” the researchers wrote.
“Monitoring the COVID generation of infants for long-term cognitive and health outcomes after birth is warranted and currently underway,” and continued research may inform preventive strategies for pregnant women experiencing multiple stressors beyond the pandemic, they concluded.
Interpret pandemic effect with caution
“Research studies, as well as our own daily experiences, have made it abundantly clear that stress is on the rise as a consequence of the COVID-19 pandemic,” said editorial author Dr. Monk, who commented on the second study in an interview. “This is an important public health question: Early identification of pandemic effects on child development can help garner the necessary resources to intervene early, dramatically increasing the likelihood of improving that child’s developmental trajectory,” she said.
“The pandemic is an unprecedented experience that has widespread impact on people’s lives, how could it not also alter gestational biology and the developing brain? That being said, we need to be cautious in that we do not yet know the functional implications of these brain changes for longer-term development,” Dr. Monk said. “Also, we do not know what aspects of women’s pandemic-affected lives had an influence on fetal brain development. The authors found higher stress in pandemic versus nonpandemic women, but not evidence that distress was the mediating variable relating pregnancy during the pandemic to altered brain development,” she explained.
The take-home message for clinicians is to “provide your patients with realistic avenues for neurodevelopmental assessments of their children if they, or you, have concerns,” Dr. Monk said. “However, do not prejudge ‘pandemic babies,’ as not all children will be affected by these potential pandemic effects,” she emphasized. “It is possible to misjudge normal variation in children’s development and unnecessarily raise parents’ anxiety levels. Importantly, this period of brain plasticity means any needed intervention likely can have a big, ameliorating impact,” she added.
“We need follow-up studies looking at pandemic effects on prenatal and postnatal development and what factors protect the fetus and birthing person from the negative influences,” she said.
The JAMA study was supported by the National Institutes of Health and the A. James & Alice B. Clark Foundation. The study in Communications Medicine was supported by the National Institutes of Health, the Intellectual and Developmental Disabilities Research Center, and the A. James & Alice B. Clark Foundation. None of the researchers in either study disclosed conflicts of interest. Dr. Monk disclosed grants from the National Institutes of Health, the Bezos Family Foundation, and the Robin Hood Foundation outside the submitted work.
Babies of mothers who experience significant psychological distress during pregnancy showed evidence of altered brain development in utero and reduced cognitive outcomes at 18 months, based on data from a pair of studies including approximately 300 women.
In a longitudinal study published in JAMA Network Open, Yao Wu, PhD, of Children’s National Hospital, Washington, and colleagues recruited 97 healthy mother-infant dyads between January 2016 and October 2020 at a single center. Of these, 87 underwent two fetal brain imaging studies each, and 10 completed the first MRI visit, for a total of 184 fetal MRIs.
Neurodevelopment and social-emotional development for infants at 18 months of age was measured using the Bayley Scales of Infant and Toddler Development and Infant-Toddler Social and Emotional Assessment. The mean age of the mothers was 35 years; maternal distress was assessed between 24 and 40 weeks’ gestation using validated self-report questionnaires. Parenting stress was assessed at the 18-month infant testing using the Parenting Stress Index-Short Form.
Overall, prenatal maternal stress was negatively associated with infant cognitive performance (P = .01) at 18 months, mediated by fetal left hippocampal volume.
In addition, increased fetal cortical local gyrification index and sulcal depth measured during reported times of prenatal maternal distress were associated with significantly poorer social-emotional scores and competence scores at age 18 months. The beta coefficients for local gyrification index and sulcal depth were –54.62 and –14.22, respectively, for social-emotional and competence scores, –24.01 and –7.53, respectively; P values were P < .001, P < .002, P = .003, P < .001, respectively.
“Increased cortical gyrification has been suggested in children with dyslexia and autism, and sulcal depth has been associated with the severity of impaired performance on working memory and executive function in adults with schizophrenia,” the researchers wrote in their discussion of the findings.
The current study “extends our previous findings and suggests a critical role for disturbances in emerging fetal cerebral cortical folding development in mediating the association between prenatal maternal distress and neurodevelopmental problems that later manifest in infancy,” they explained.
The researchers also found that prenatal maternal anxiety, stress, and depression were positively associated with all measures of parenting stress at the 18-month testing visit.
The study findings were limited by several factors including the use of self-reports for both maternal distress and infant social-emotional assessment, despite the use of validated questionnaires, and the fact that assessment of maternal distress at specific times may not reflect the entire pregnancy, the researchers noted. Other potential limitations included the inability to use some MRI data because of fetal movement and the homogenous population of relatively highly educated women with access to health care that may not reflect other areas, they said.
“Identifying early brain developmental biomarkers may help improve the identification of infants at risk for later neurodevelopmental impairment who might benefit from early targeted interventions,” the researchers concluded.
Technology enhances health and disease models
The effect of the prenatal period on future well-being is recognized, but the current study makes “substantial contributions to prenatal programming science, with implications for ways to transform the prenatal care ecosystem for two-generation impact,” Catherine Monk, PhD, and Cristina R. Fernández, MD, both of Columbia University, New York, wrote in an accompanying editorial.
The developmental origins of health and disease (DOHaD) conceptual model introduced by Dr. David Barker in 1995 were later applied to show that maternal stress, depression, and anxiety affected child prenatal and future development, they said. However, the current study uses cutting-edge neuroscience to directly assess developing fetal brains. The finding of reduced cognitive functioning at 18 months associated with maternal stress is consistent with other findings, they noted.
“Finding an association between maternal prenatal stress and infant cognitive outcomes in the setting of what may be modest stress relative to that of a low-resourced or historically marginalized sample underscores the importance of this research; presumably, with higher stress, and greater social determinants of health burden, the effect sizes would be even greater and of greater concern,” they said.
However, studies such as the current one “have the potential to transform the prenatal and postpartum care ecosystems,” by encouraging a whole-person approach to the care of pregnant women, including attention to mental well-being and quality of life, they emphasized.
COVID-19 stress considerations
In a separate study published in Communications Medicine, Yuan-Chiao Lu, MD, also of Children’s National Hospital in Washington, and colleagues found a similar effect of maternal stress on fetal brain development.
The researchers imaged the brains of fetuses before and during the COVID-19 pandemic and interviewed mothers about any distress they experienced during pregnancy.
The study population included 65 women with known COVID-19 exposures who underwent 92 fetal MRIs and 137 prepandemic controls who underwent 182 fetal MRIs. Maternal distress was measured via the Spielberger State Anxiety Inventory, Spielberger Trait Anxiety Inventory, Perceived Stress Scale, and Edinburgh Postnatal Depression Scale.
Overall, scores on measures of stress and depression were significantly higher for women in the pandemic group compared with controls. Of the 173 women for whom maternal distress measures were available, 28% of the prepandemic group and 52% of the pandemic group met criteria for elevated maternal psychological distress, defined as above the threshold for distress on any one of the four measures.
After the researchers controlled for maternal distress, MRI data showed decreases in fetal white matter and in hippocampal and cerebellar volumes in fetuses in the pandemic group compared with controls.
Other signs of impaired brain development were similar to those seen in the JAMA Network Open study, including decreased cortical surface area and local gyrification index, as well as reduced sulcal depth in multiple brain lobes, indicating delayed cerebral cortical gyrification.
The second study was limited by a lack of data on other lifestyle changes during the pandemic that might influence maternal health and fetal development, the researchers noted. Other limitations were the possible lack of generalizability to a range of racial and ethnic populations and geographic areas outside of Washington, and the inability to control for unknown COVID-19 exposures or subclinical infections in controls, they said.
However, the results support findings from previous studies, and provide a unique opportunity to study the effect of prenatal stress on early development, as well as a chance to implement “novel and timely interventions,” the researchers wrote.
“Monitoring the COVID generation of infants for long-term cognitive and health outcomes after birth is warranted and currently underway,” and continued research may inform preventive strategies for pregnant women experiencing multiple stressors beyond the pandemic, they concluded.
Interpret pandemic effect with caution
“Research studies, as well as our own daily experiences, have made it abundantly clear that stress is on the rise as a consequence of the COVID-19 pandemic,” said editorial author Dr. Monk, who commented on the second study in an interview. “This is an important public health question: Early identification of pandemic effects on child development can help garner the necessary resources to intervene early, dramatically increasing the likelihood of improving that child’s developmental trajectory,” she said.
“The pandemic is an unprecedented experience that has widespread impact on people’s lives, how could it not also alter gestational biology and the developing brain? That being said, we need to be cautious in that we do not yet know the functional implications of these brain changes for longer-term development,” Dr. Monk said. “Also, we do not know what aspects of women’s pandemic-affected lives had an influence on fetal brain development. The authors found higher stress in pandemic versus nonpandemic women, but not evidence that distress was the mediating variable relating pregnancy during the pandemic to altered brain development,” she explained.
The take-home message for clinicians is to “provide your patients with realistic avenues for neurodevelopmental assessments of their children if they, or you, have concerns,” Dr. Monk said. “However, do not prejudge ‘pandemic babies,’ as not all children will be affected by these potential pandemic effects,” she emphasized. “It is possible to misjudge normal variation in children’s development and unnecessarily raise parents’ anxiety levels. Importantly, this period of brain plasticity means any needed intervention likely can have a big, ameliorating impact,” she added.
“We need follow-up studies looking at pandemic effects on prenatal and postnatal development and what factors protect the fetus and birthing person from the negative influences,” she said.
The JAMA study was supported by the National Institutes of Health and the A. James & Alice B. Clark Foundation. The study in Communications Medicine was supported by the National Institutes of Health, the Intellectual and Developmental Disabilities Research Center, and the A. James & Alice B. Clark Foundation. None of the researchers in either study disclosed conflicts of interest. Dr. Monk disclosed grants from the National Institutes of Health, the Bezos Family Foundation, and the Robin Hood Foundation outside the submitted work.
FROM JAMA NETWORK OPEN AND COMMUNICATIONS MEDICINE



