User login
FDA working to improve U.S. baby formula supply
The Food and Drug Administration announced on May 10 that it is taking several steps to improve the supply of baby formula in the United States.
The nationwide formula shortage has grown worse in recent weeks due to supply chain issues and a recall of certain Abbott Nutrition products, including major labels such as Similac, Alimentum, and EleCare.
“We recognize that many consumers have been unable to access infant formula and critical medical foods they are accustomed to using and are frustrated by their inability to do so,” FDA Commissioner Robert Califf, MD, said in a statement.
“We are doing everything in our power to ensure there is adequate product available where and when they need it,” he said.
About three-quarters of babies are fed formula for the first 6 months of their lives as a substitute for human milk, Axios reported.
In mid-February, the FDA warned consumers not to use certain powdered infant formula products from Abbott’s facility in Sturgis, Mich. Since then, the FDA has been working with Abbott and other manufacturers to increase the supply in the U.S. market.
“In fact, other infant formula manufacturers are meeting or exceeding capacity levels to meet current demand,” the FDA said in the statement. “Notably, more infant formula was purchased in the month of April than in the month prior to the recall.”
The FDA released a list of steps the agency is taking to increase supply, such as meeting with major infant formula makers to increase output and prioritize product lines in high demand, particularly specialty formulas for infants with allergies or specific diet needs.
But other manufacturers have struggled to quickly increase production because their operations tend to focus on a steady level of supply, according to The New York Times.
“Some industries are very good at ramping up and ramping down,” Rudi Leuschner, PhD, an associate professor of supply chain management at Rutgers Business School, Newark, N.J., told the newspaper.
“You flip a switch and they can produce 10 times as much,” he said. “Baby formula is not that type of a product.”
The FDA is also keeping an eye on the infant formula shortage by using the agency’s 21 Forward food supply chain continuity system. The system was developed during the pandemic to provide a full understanding of how COVID-19 is impacting food supply chains, the FDA said.
The FDA is compiling data on trends for in-stock rates at national and regional levels to understand where infant formula is available and where it should go.
Products are also being brought in from other countries, the FDA said. The agency is trying to speed up the process to get more formula into the U.S. and move it more quickly around the country.
For babies on a special diet, the FDA has decided to release some Abbott products that have been on hold at the Sturgis facility to those who need an urgent supply of metabolic formulas, on a case-by-case basis.
“In these circumstances, the benefit of allowing caregivers, in consultation with their health care providers, to access these products may outweigh the potential risk of bacterial infection,” the FDA said in the statement.
The FDA continues to advise against making homemade infant formulas and recommends talking to the child’s health care provider for recommendations on changing feeding practices or switching to other formulas, if necessary.
A version of this article first appeared on WebMd.com.
The Food and Drug Administration announced on May 10 that it is taking several steps to improve the supply of baby formula in the United States.
The nationwide formula shortage has grown worse in recent weeks due to supply chain issues and a recall of certain Abbott Nutrition products, including major labels such as Similac, Alimentum, and EleCare.
“We recognize that many consumers have been unable to access infant formula and critical medical foods they are accustomed to using and are frustrated by their inability to do so,” FDA Commissioner Robert Califf, MD, said in a statement.
“We are doing everything in our power to ensure there is adequate product available where and when they need it,” he said.
About three-quarters of babies are fed formula for the first 6 months of their lives as a substitute for human milk, Axios reported.
In mid-February, the FDA warned consumers not to use certain powdered infant formula products from Abbott’s facility in Sturgis, Mich. Since then, the FDA has been working with Abbott and other manufacturers to increase the supply in the U.S. market.
“In fact, other infant formula manufacturers are meeting or exceeding capacity levels to meet current demand,” the FDA said in the statement. “Notably, more infant formula was purchased in the month of April than in the month prior to the recall.”
The FDA released a list of steps the agency is taking to increase supply, such as meeting with major infant formula makers to increase output and prioritize product lines in high demand, particularly specialty formulas for infants with allergies or specific diet needs.
But other manufacturers have struggled to quickly increase production because their operations tend to focus on a steady level of supply, according to The New York Times.
“Some industries are very good at ramping up and ramping down,” Rudi Leuschner, PhD, an associate professor of supply chain management at Rutgers Business School, Newark, N.J., told the newspaper.
“You flip a switch and they can produce 10 times as much,” he said. “Baby formula is not that type of a product.”
The FDA is also keeping an eye on the infant formula shortage by using the agency’s 21 Forward food supply chain continuity system. The system was developed during the pandemic to provide a full understanding of how COVID-19 is impacting food supply chains, the FDA said.
The FDA is compiling data on trends for in-stock rates at national and regional levels to understand where infant formula is available and where it should go.
Products are also being brought in from other countries, the FDA said. The agency is trying to speed up the process to get more formula into the U.S. and move it more quickly around the country.
For babies on a special diet, the FDA has decided to release some Abbott products that have been on hold at the Sturgis facility to those who need an urgent supply of metabolic formulas, on a case-by-case basis.
“In these circumstances, the benefit of allowing caregivers, in consultation with their health care providers, to access these products may outweigh the potential risk of bacterial infection,” the FDA said in the statement.
The FDA continues to advise against making homemade infant formulas and recommends talking to the child’s health care provider for recommendations on changing feeding practices or switching to other formulas, if necessary.
A version of this article first appeared on WebMd.com.
The Food and Drug Administration announced on May 10 that it is taking several steps to improve the supply of baby formula in the United States.
The nationwide formula shortage has grown worse in recent weeks due to supply chain issues and a recall of certain Abbott Nutrition products, including major labels such as Similac, Alimentum, and EleCare.
“We recognize that many consumers have been unable to access infant formula and critical medical foods they are accustomed to using and are frustrated by their inability to do so,” FDA Commissioner Robert Califf, MD, said in a statement.
“We are doing everything in our power to ensure there is adequate product available where and when they need it,” he said.
About three-quarters of babies are fed formula for the first 6 months of their lives as a substitute for human milk, Axios reported.
In mid-February, the FDA warned consumers not to use certain powdered infant formula products from Abbott’s facility in Sturgis, Mich. Since then, the FDA has been working with Abbott and other manufacturers to increase the supply in the U.S. market.
“In fact, other infant formula manufacturers are meeting or exceeding capacity levels to meet current demand,” the FDA said in the statement. “Notably, more infant formula was purchased in the month of April than in the month prior to the recall.”
The FDA released a list of steps the agency is taking to increase supply, such as meeting with major infant formula makers to increase output and prioritize product lines in high demand, particularly specialty formulas for infants with allergies or specific diet needs.
But other manufacturers have struggled to quickly increase production because their operations tend to focus on a steady level of supply, according to The New York Times.
“Some industries are very good at ramping up and ramping down,” Rudi Leuschner, PhD, an associate professor of supply chain management at Rutgers Business School, Newark, N.J., told the newspaper.
“You flip a switch and they can produce 10 times as much,” he said. “Baby formula is not that type of a product.”
The FDA is also keeping an eye on the infant formula shortage by using the agency’s 21 Forward food supply chain continuity system. The system was developed during the pandemic to provide a full understanding of how COVID-19 is impacting food supply chains, the FDA said.
The FDA is compiling data on trends for in-stock rates at national and regional levels to understand where infant formula is available and where it should go.
Products are also being brought in from other countries, the FDA said. The agency is trying to speed up the process to get more formula into the U.S. and move it more quickly around the country.
For babies on a special diet, the FDA has decided to release some Abbott products that have been on hold at the Sturgis facility to those who need an urgent supply of metabolic formulas, on a case-by-case basis.
“In these circumstances, the benefit of allowing caregivers, in consultation with their health care providers, to access these products may outweigh the potential risk of bacterial infection,” the FDA said in the statement.
The FDA continues to advise against making homemade infant formulas and recommends talking to the child’s health care provider for recommendations on changing feeding practices or switching to other formulas, if necessary.
A version of this article first appeared on WebMd.com.
Prior authorizations delay TNF inhibitors for children with JIA
Children with juvenile idiopathic arthritis (JIA) who need a tumor necrosis factor (TNF) inhibitor after failing conventional disease-modifying antirheumatic drug (DMARD) treatment often experience insurance delays before beginning the new drug because of prior authorization denials, according to research presented at the 2022 annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). The findings were also published as a research letter in JAMA Network Open.
“Prompt escalation to TNF inhibitors is recommended for children with JIA refractory to DMARDs,” author Jordan Roberts, MD, a clinical fellow of the Harvard Medical School Rheumatology Program, Boston, told CARRA attendees. TNF inhibitors are increasingly used as first-line treatment in JIA since growing evidence suggests better outcomes from early treatment with biologics. “Prior authorization requirements that delay TNF inhibitor initiation among children with JIA are common in clinical practice,” Dr. Roberts said, but little evidence exists to understand the extent of this problem and its causes.
The researchers therefore conducted a retrospective cohort study using a search of electronic health records from January 2018 to December 2019 to find all children at a single center with a new diagnosis of nonsystemic JIA. Then the authors pulled the timing of prior authorization requests, approvals, denials, and first TNF inhibitor dose from the medical notes. They also sought out any children who had been recommended a TNF inhibitor but never started one.
The total population included 54 children with an average age of 10 years, about two-thirds of whom had private insurance (63%). The group was predominantly White (63%), although 13% declined to provide race, and 7% were Hispanic. Most subtypes of disease were represented: oligoarticular persistent (28%), oligoarticular extended (2%), polyarticular rheumatoid factor-negative (15%), polyarticular rheumatoid factor-positive (15%), psoriatic arthritis (26%), enthesitis-related arthritis (12%), and undifferentiated arthritis (2%).
The 44 participants with private insurance had an average of two joints with active disease, while the 10 patients with public insurance had an average of four involved joints. Nearly all the patients (91%) had previously taken or were currently taking DMARDs when the prior authorization was submitted, and 61% had received NSAIDs.
All but one of the patients’ insurance plans required a prior authorization. The first prior authorization was denied for about one-third of the public insurance patients (30%) and a quarter of the private insurance patients. About 1 in 5 patients overall (22%) required a written appeal to override the denial, and 4% required peer-to-peer review. Meanwhile, 7% of patients began another medication because of the denial.
It took a median of 3 days for prior authorizations to be approved and a median of 24 days from the time the TNF inhibitor was recommended to the patient receiving the first dose. However, 22% of patients waited at least 2 weeks before the prior authorization was approved, and more than a quarter of the requests took over 30 days before the patient could begin the medication. In the public insurance group in particular, a quarter of children waited at least 19 days for approval and at least 44 days before starting the medication.
In fact, when the researchers looked at the difference in approval time between those who did and did not receive an initial denial, the difference was stark. Median approval time was 16 days when the prior authorization was denied, compared with a median of 5 days when the first prior authorization was approved. Similarly, time to initiation of the drug after recommendation was a median of 35 days for those whose prior authorization was first denied and 17 days for those with an initial approval.
The most common reason for an initial denial was the insurance company requiring a different TNF inhibitor than the one the rheumatologist wanted to prescribe. “These were all children whose rheumatologist has recommended either infliximab or etanercept that were required to use adalimumab instead,” Dr. Roberts said.
The other reasons for initial denial were similarly familiar ones:
- Required submission to another insurer
- Additional documentation required
- Lack of medical necessity
- Prescription was for an indication not approved by the Food and Drug Administration
- Age of patient
- Nonbiologic DMARD required
- NSAID required for step therapy
Only three children who were advised to begin a TNF inhibitor did not do so, including one who was lost to follow-up, one who had injection-related anxiety, and one who had safety concerns about the medication.
“Several children were required to use alternative TNF inhibitors than the one that was recommended due to restricted formularies, which may reduce shared decisionmaking between physicians and families and may not be the optimal clinical choice for an individual child,” Dr. Roberts said in her conclusion. Most children, however, were able to get approval for the TNF inhibitor originally requested, “suggesting that utilization management strategies present barriers to timely care despite appropriate specialty medication requests,” she said. “Therefore, it’s important for us to advocate for access to medications for children with JIA.”
Findings are not surprising
“I have these same experiences at my institution – often insurance will dictate clinical practice, and step therapy is the only option, causing a delay to initiation of TNFi even if we think, as the pediatric rheumatologist, that a child needs this medicine to be initiated on presentation to our clinic,” Nayimisha Balmuri, MD, assistant professor of pediatrics in the division of allergy, immunology, and rheumatology at the Johns Hopkins School of Medicine, Baltimore, told this news organization.
Dr. Balmuri, who was not involved in the study, noted that in her clinic at Johns Hopkins, it is hit or miss if an appeal to insurance companies or to the state (if it is Medicaid coverage) will be successful. “Unfortunately, [we are] mostly unsuccessful, and we have to try another DMARD for 8 to 12 weeks first before trying to get TNFi,” she said.
Dr. Balmuri called for bringing these issues to the attention of state and federal legislators. “It’s so important for us to continue to advocate for our patients at the state and national level! We are the advocates for our patients, and we are uniquely trained to know the best medications to initiate to help patients maximize their chance to reach remission of arthritis. Insurance companies need to hear our voices!”
Dr. Roberts reported grants from CARRA, the Lupus Foundation of America, and the National Institute of Allergy and Infectious Diseases during the conduct of the study.
A version of this article first appeared on Medscape.com.
Children with juvenile idiopathic arthritis (JIA) who need a tumor necrosis factor (TNF) inhibitor after failing conventional disease-modifying antirheumatic drug (DMARD) treatment often experience insurance delays before beginning the new drug because of prior authorization denials, according to research presented at the 2022 annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). The findings were also published as a research letter in JAMA Network Open.
“Prompt escalation to TNF inhibitors is recommended for children with JIA refractory to DMARDs,” author Jordan Roberts, MD, a clinical fellow of the Harvard Medical School Rheumatology Program, Boston, told CARRA attendees. TNF inhibitors are increasingly used as first-line treatment in JIA since growing evidence suggests better outcomes from early treatment with biologics. “Prior authorization requirements that delay TNF inhibitor initiation among children with JIA are common in clinical practice,” Dr. Roberts said, but little evidence exists to understand the extent of this problem and its causes.
The researchers therefore conducted a retrospective cohort study using a search of electronic health records from January 2018 to December 2019 to find all children at a single center with a new diagnosis of nonsystemic JIA. Then the authors pulled the timing of prior authorization requests, approvals, denials, and first TNF inhibitor dose from the medical notes. They also sought out any children who had been recommended a TNF inhibitor but never started one.
The total population included 54 children with an average age of 10 years, about two-thirds of whom had private insurance (63%). The group was predominantly White (63%), although 13% declined to provide race, and 7% were Hispanic. Most subtypes of disease were represented: oligoarticular persistent (28%), oligoarticular extended (2%), polyarticular rheumatoid factor-negative (15%), polyarticular rheumatoid factor-positive (15%), psoriatic arthritis (26%), enthesitis-related arthritis (12%), and undifferentiated arthritis (2%).
The 44 participants with private insurance had an average of two joints with active disease, while the 10 patients with public insurance had an average of four involved joints. Nearly all the patients (91%) had previously taken or were currently taking DMARDs when the prior authorization was submitted, and 61% had received NSAIDs.
All but one of the patients’ insurance plans required a prior authorization. The first prior authorization was denied for about one-third of the public insurance patients (30%) and a quarter of the private insurance patients. About 1 in 5 patients overall (22%) required a written appeal to override the denial, and 4% required peer-to-peer review. Meanwhile, 7% of patients began another medication because of the denial.
It took a median of 3 days for prior authorizations to be approved and a median of 24 days from the time the TNF inhibitor was recommended to the patient receiving the first dose. However, 22% of patients waited at least 2 weeks before the prior authorization was approved, and more than a quarter of the requests took over 30 days before the patient could begin the medication. In the public insurance group in particular, a quarter of children waited at least 19 days for approval and at least 44 days before starting the medication.
In fact, when the researchers looked at the difference in approval time between those who did and did not receive an initial denial, the difference was stark. Median approval time was 16 days when the prior authorization was denied, compared with a median of 5 days when the first prior authorization was approved. Similarly, time to initiation of the drug after recommendation was a median of 35 days for those whose prior authorization was first denied and 17 days for those with an initial approval.
The most common reason for an initial denial was the insurance company requiring a different TNF inhibitor than the one the rheumatologist wanted to prescribe. “These were all children whose rheumatologist has recommended either infliximab or etanercept that were required to use adalimumab instead,” Dr. Roberts said.
The other reasons for initial denial were similarly familiar ones:
- Required submission to another insurer
- Additional documentation required
- Lack of medical necessity
- Prescription was for an indication not approved by the Food and Drug Administration
- Age of patient
- Nonbiologic DMARD required
- NSAID required for step therapy
Only three children who were advised to begin a TNF inhibitor did not do so, including one who was lost to follow-up, one who had injection-related anxiety, and one who had safety concerns about the medication.
“Several children were required to use alternative TNF inhibitors than the one that was recommended due to restricted formularies, which may reduce shared decisionmaking between physicians and families and may not be the optimal clinical choice for an individual child,” Dr. Roberts said in her conclusion. Most children, however, were able to get approval for the TNF inhibitor originally requested, “suggesting that utilization management strategies present barriers to timely care despite appropriate specialty medication requests,” she said. “Therefore, it’s important for us to advocate for access to medications for children with JIA.”
Findings are not surprising
“I have these same experiences at my institution – often insurance will dictate clinical practice, and step therapy is the only option, causing a delay to initiation of TNFi even if we think, as the pediatric rheumatologist, that a child needs this medicine to be initiated on presentation to our clinic,” Nayimisha Balmuri, MD, assistant professor of pediatrics in the division of allergy, immunology, and rheumatology at the Johns Hopkins School of Medicine, Baltimore, told this news organization.
Dr. Balmuri, who was not involved in the study, noted that in her clinic at Johns Hopkins, it is hit or miss if an appeal to insurance companies or to the state (if it is Medicaid coverage) will be successful. “Unfortunately, [we are] mostly unsuccessful, and we have to try another DMARD for 8 to 12 weeks first before trying to get TNFi,” she said.
Dr. Balmuri called for bringing these issues to the attention of state and federal legislators. “It’s so important for us to continue to advocate for our patients at the state and national level! We are the advocates for our patients, and we are uniquely trained to know the best medications to initiate to help patients maximize their chance to reach remission of arthritis. Insurance companies need to hear our voices!”
Dr. Roberts reported grants from CARRA, the Lupus Foundation of America, and the National Institute of Allergy and Infectious Diseases during the conduct of the study.
A version of this article first appeared on Medscape.com.
Children with juvenile idiopathic arthritis (JIA) who need a tumor necrosis factor (TNF) inhibitor after failing conventional disease-modifying antirheumatic drug (DMARD) treatment often experience insurance delays before beginning the new drug because of prior authorization denials, according to research presented at the 2022 annual meeting of the Childhood Arthritis and Rheumatology Research Alliance (CARRA). The findings were also published as a research letter in JAMA Network Open.
“Prompt escalation to TNF inhibitors is recommended for children with JIA refractory to DMARDs,” author Jordan Roberts, MD, a clinical fellow of the Harvard Medical School Rheumatology Program, Boston, told CARRA attendees. TNF inhibitors are increasingly used as first-line treatment in JIA since growing evidence suggests better outcomes from early treatment with biologics. “Prior authorization requirements that delay TNF inhibitor initiation among children with JIA are common in clinical practice,” Dr. Roberts said, but little evidence exists to understand the extent of this problem and its causes.
The researchers therefore conducted a retrospective cohort study using a search of electronic health records from January 2018 to December 2019 to find all children at a single center with a new diagnosis of nonsystemic JIA. Then the authors pulled the timing of prior authorization requests, approvals, denials, and first TNF inhibitor dose from the medical notes. They also sought out any children who had been recommended a TNF inhibitor but never started one.
The total population included 54 children with an average age of 10 years, about two-thirds of whom had private insurance (63%). The group was predominantly White (63%), although 13% declined to provide race, and 7% were Hispanic. Most subtypes of disease were represented: oligoarticular persistent (28%), oligoarticular extended (2%), polyarticular rheumatoid factor-negative (15%), polyarticular rheumatoid factor-positive (15%), psoriatic arthritis (26%), enthesitis-related arthritis (12%), and undifferentiated arthritis (2%).
The 44 participants with private insurance had an average of two joints with active disease, while the 10 patients with public insurance had an average of four involved joints. Nearly all the patients (91%) had previously taken or were currently taking DMARDs when the prior authorization was submitted, and 61% had received NSAIDs.
All but one of the patients’ insurance plans required a prior authorization. The first prior authorization was denied for about one-third of the public insurance patients (30%) and a quarter of the private insurance patients. About 1 in 5 patients overall (22%) required a written appeal to override the denial, and 4% required peer-to-peer review. Meanwhile, 7% of patients began another medication because of the denial.
It took a median of 3 days for prior authorizations to be approved and a median of 24 days from the time the TNF inhibitor was recommended to the patient receiving the first dose. However, 22% of patients waited at least 2 weeks before the prior authorization was approved, and more than a quarter of the requests took over 30 days before the patient could begin the medication. In the public insurance group in particular, a quarter of children waited at least 19 days for approval and at least 44 days before starting the medication.
In fact, when the researchers looked at the difference in approval time between those who did and did not receive an initial denial, the difference was stark. Median approval time was 16 days when the prior authorization was denied, compared with a median of 5 days when the first prior authorization was approved. Similarly, time to initiation of the drug after recommendation was a median of 35 days for those whose prior authorization was first denied and 17 days for those with an initial approval.
The most common reason for an initial denial was the insurance company requiring a different TNF inhibitor than the one the rheumatologist wanted to prescribe. “These were all children whose rheumatologist has recommended either infliximab or etanercept that were required to use adalimumab instead,” Dr. Roberts said.
The other reasons for initial denial were similarly familiar ones:
- Required submission to another insurer
- Additional documentation required
- Lack of medical necessity
- Prescription was for an indication not approved by the Food and Drug Administration
- Age of patient
- Nonbiologic DMARD required
- NSAID required for step therapy
Only three children who were advised to begin a TNF inhibitor did not do so, including one who was lost to follow-up, one who had injection-related anxiety, and one who had safety concerns about the medication.
“Several children were required to use alternative TNF inhibitors than the one that was recommended due to restricted formularies, which may reduce shared decisionmaking between physicians and families and may not be the optimal clinical choice for an individual child,” Dr. Roberts said in her conclusion. Most children, however, were able to get approval for the TNF inhibitor originally requested, “suggesting that utilization management strategies present barriers to timely care despite appropriate specialty medication requests,” she said. “Therefore, it’s important for us to advocate for access to medications for children with JIA.”
Findings are not surprising
“I have these same experiences at my institution – often insurance will dictate clinical practice, and step therapy is the only option, causing a delay to initiation of TNFi even if we think, as the pediatric rheumatologist, that a child needs this medicine to be initiated on presentation to our clinic,” Nayimisha Balmuri, MD, assistant professor of pediatrics in the division of allergy, immunology, and rheumatology at the Johns Hopkins School of Medicine, Baltimore, told this news organization.
Dr. Balmuri, who was not involved in the study, noted that in her clinic at Johns Hopkins, it is hit or miss if an appeal to insurance companies or to the state (if it is Medicaid coverage) will be successful. “Unfortunately, [we are] mostly unsuccessful, and we have to try another DMARD for 8 to 12 weeks first before trying to get TNFi,” she said.
Dr. Balmuri called for bringing these issues to the attention of state and federal legislators. “It’s so important for us to continue to advocate for our patients at the state and national level! We are the advocates for our patients, and we are uniquely trained to know the best medications to initiate to help patients maximize their chance to reach remission of arthritis. Insurance companies need to hear our voices!”
Dr. Roberts reported grants from CARRA, the Lupus Foundation of America, and the National Institute of Allergy and Infectious Diseases during the conduct of the study.
A version of this article first appeared on Medscape.com.
Ex–hospital porter a neglected giant of cancer research
We have a half-forgotten Indian immigrant to thank – a hospital night porter turned biochemist –for revolutionizing treatment of leukemia, the once deadly childhood scourge that is still the most common pediatric cancer.
Dr. Yellapragada SubbaRow has been called the “father of chemotherapy” for developing methotrexate, a powerful, inexpensive therapy for leukemia and other diseases, and he is celebrated for additional scientific achievements. Yet Dr. SubbaRow’s life was marked more by struggle than glory.
Born poor in southeastern India, he nearly succumbed to a tropical disease that killed two older brothers, and he didn’t focus on schoolwork until his father died. Later, prejudice dogged his years as an immigrant to the United States, and a blood clot took his life at the age of 53.
Scientifically, however, Dr. SubbaRow (pronounced sue-buh-rao) triumphed, despite mammoth challenges and a lack of recognition that persists to this day. National Cancer Research Month is a fitting time to look back on his extraordinary life and work and pay tribute to his accomplishments.
‘Yella,’ folic acid, and a paradigm shift
No one appreciates Dr. SubbaRow more than a cadre of Indian-born physicians who have kept his legacy alive in journal articles, presentations, and a Pulitzer Prize-winning book. Among them is author and oncologist Siddhartha Mukherjee, MD, who chronicled Dr. SubbaRow’s achievements in his New York Times No. 1 bestseller, “The Emperor of All Maladies: A Biography of Cancer.”
As Dr. Mukherjee wrote, Dr. SubbaRow was a “pioneer in many ways, a physician turned cellular physiologist, a chemist who had accidentally wandered into biology.” (Per Indian tradition, SubbaRow is the doctor’s first name, and Yellapragada is his surname, but medical literature uses SubbaRow as his cognomen, with some variations in spelling. Dr. Mukherjee wrote that his friends called him “Yella.”)
Dr. SubbaRow came to the United States in 1923, after enduring a difficult childhood and young adulthood. He’d survived bouts of religious fervor, childhood rebellion (including a bid to run away from home and become a banana trader), and a failed arranged marriage. His wife bore him a child who died in infancy. He left it all behind.
In Boston, medical officials rejected his degree. Broke, he worked for a time as a night porter at Brigham and Women’s Hospital in Boston, changing sheets and cleaning urinals. To a poor but proud high-caste Indian Brahmin, the culture shock of carrying out these tasks must have been especially jarring.
Dr. SubbaRow went on to earn a diploma from Harvard Medical School, also in Boston, and became a junior faculty member. As a foreigner, Dr. Mukherjee wrote, Dr. SubbaRow was a “reclusive, nocturnal, heavily accented vegetarian,” so different from his colleagues that advancement seemed impossible. Despite his pioneering biochemistry work, Harvard later declined to offer Dr. SubbaRow a tenured faculty position.
By the early 1940s, he took a job at an upstate New York pharmaceutical company called Lederle Labs (later purchased by Pfizer). At Lederle, Dr. SubbaRow strove to synthesize the vitamin known as folic acid. He ended up creating a kind of antivitamin, a lookalike that acted like folic acid but only succeeded in gumming up the works in receptors. But what good would it do to stop the body from absorbing folic acid? Plenty, it turned out.
Discoveries pile up, but credit and fame prove elusive
Dr. SubbaRow was no stranger to producing landmark biological work. He’d previously codiscovered phosphocreatine and ATP, which are crucial to muscular contractions. However, “in 1935, he had to disown the extent of his role in the discovery of the color test related to phosphorus, instead giving the credit to his co-author, who was being considered for promotion to a full professorship at Harvard,” wrote author Gerald Posner in his 2020 book, “Pharma: Greed, Lies and the Poisoning of America.”
Houston-area oncologist Kirtan Nautiyal, MD, who paid tribute to Dr. SubbaRow in a 2018 article, contended that “with his Indian instinct for self-effacement, he had irreparably sabotaged his own career.”
Dr. SubbaRow and his team also developed “the first effective treatment of filariasis, which causes elephantiasis of the lower limbs and genitals in millions of people, mainly in tropical countries,” Dr. Nautiyal wrote. “Later in the decade, his antibiotic program generated polymyxin, the first effective treatment against the class of bacteria called Gram negatives, and aureomycin, the first “broad-spectrum’ antibiotic.” (Aureomycin is also the first tetracycline antibiotic.)
Dr. SubbaRow’s discovery of a folic acid antagonist would again go largely unheralded. But first came the realization that folic acid made childhood leukemia worse, not better, and the prospect that this process could potentially be reversed.
Rise of methotrexate and fall of leukemia
In Boston, Sidney Farber, MD, a Boston pathologist, was desperate to help Robert Sandler, a 2-year-old leukemia patient. Dr. Farber contacted his ex-colleague Dr. SubbaRow to request a supply of aminopterin, an early version of methotrexate that Dr. SubbaRow and his team had developed. Dr. Farber injected Robert with the substance and within 3 days, the toddler’s white blood count started falling – fast. He stopped bleeding, resumed eating, and once again seemed almost identical to his twin brother, as Dr. Mukherjee wrote in his book.
Leukemia had never gone into remission before. Unfortunately, the treatment only worked temporarily. Robert, like other children treated with the drug, relapsed and died within months. But Dr. Farber “saw a door open” – a chemical, a kind of chemotherapy, that could turn back cancer. In the case of folic acid antagonists, they do so by stopping cancer cells from replicating.
Methotrexate, a related agent synthesized by Dr. SubbaRow, would become a mainstay of leukemia treatment and begin to produce long-term remission from acute lymphoblastic leukemia in 1970, when combination chemotherapy was developed.
Other cancers fell to methotrexate treatment. “Previous assumptions that cancer was nearly always fatal were revised, and the field of medical oncology (treatment of cancer with chemotherapy), which had not previously existed, was formally established in 1971,” according to the National Cancer Institute’s history of methotrexate. This account does not mention Dr. SubbaRow.
Death takes the doctor, but his legacy remains
In biographies, as well as his own words, Dr. SubbaRow comes across as a prickly, hard-driving workaholic who had little interest in intimate human connections. “It is not good to ask in every letter when I will be back,” he wrote to his wife back in India, before cutting off ties completely in the early 1930s. “I will come as early as possible. ... I do not want to write anything more.”
It seems, as his biographer S.P.K. Gupta noted, that “he was quite determined that the time allotted to him on Earth should be completely devoted to finding cures for ailments that plagued mankind.”
Still, Dr. SubbaRow’s research team was devoted to him, and he had plenty of reasons to be bitter, such as the prejudice and isolation he encountered in the United States and earlier, in British-run India. According to Mr. Posner’s book, even as a young medical student, Dr. SubbaRow heeded the call of Indian independence activist Mohandas Gandhi. He “refused the British surgical gown given him at school and instead donned a traditional and simple cotton Khadi. That act of defiance cost SubbaRow the college degree that was necessary for him to get into the State Medical College.”
During the last year of his life, Dr. SubbaRow faced yet another humiliation: In his landmark 1948 study about aminopterin as a treatment for leukemia, his colleague Dr. Farber failed to credit him, an “astonishing omission” as Yaddanapudi Ravindranath, MBBS, a pediatric hematologist/oncologist at Wayne State University, Detroit, put it. “From everything I know, Dr. Farber spent the rest of his career apologizing and trying to make amends for it,” Dr. Ravindranath said in an interview.
A career cut short, and a lasting legacy
In 1948, at the age of 53, Dr. SubbaRow suddenly died. “Many think Dr. SubbaRow would have won [the] Nobel Prize had he lived a few years longer,” said Dr. Ravindranath.
Like Dr. SubbaRow, Dr. Ravindranath was born in Andhra Pradesh state, near the city of Chennai formerly known as Madras. “Being a compatriot, in a way I continue his legacy, and I am obviously proud of him,” said Dr. Ravindranath, who has conducted his own landmark research regarding methotrexate and leukemia.
Nearly 75 years after Dr. SubbaRow’s death, Indian-born physicians like Dr. Ravindranath continue to honor him in print, trying to ensure that he’s not forgotten. Methotrexate remains a crucial treatment for leukemia, along with a long list of other ailments, including psoriasis.
Recognition for “Yella” may have come late and infrequently, but a Lederle Laboratories research library named after him offered Dr. SubbaRow a kind of immortality. A plaque there memorialized him in stone as a scientist, teacher, philosopher, and humanitarian, featuring the quote: “Science simply prolongs life. Religion deepens it.”
By all accounts, Dr. SubbaRow was a man of science and faith who had faith in science.
We have a half-forgotten Indian immigrant to thank – a hospital night porter turned biochemist –for revolutionizing treatment of leukemia, the once deadly childhood scourge that is still the most common pediatric cancer.
Dr. Yellapragada SubbaRow has been called the “father of chemotherapy” for developing methotrexate, a powerful, inexpensive therapy for leukemia and other diseases, and he is celebrated for additional scientific achievements. Yet Dr. SubbaRow’s life was marked more by struggle than glory.
Born poor in southeastern India, he nearly succumbed to a tropical disease that killed two older brothers, and he didn’t focus on schoolwork until his father died. Later, prejudice dogged his years as an immigrant to the United States, and a blood clot took his life at the age of 53.
Scientifically, however, Dr. SubbaRow (pronounced sue-buh-rao) triumphed, despite mammoth challenges and a lack of recognition that persists to this day. National Cancer Research Month is a fitting time to look back on his extraordinary life and work and pay tribute to his accomplishments.
‘Yella,’ folic acid, and a paradigm shift
No one appreciates Dr. SubbaRow more than a cadre of Indian-born physicians who have kept his legacy alive in journal articles, presentations, and a Pulitzer Prize-winning book. Among them is author and oncologist Siddhartha Mukherjee, MD, who chronicled Dr. SubbaRow’s achievements in his New York Times No. 1 bestseller, “The Emperor of All Maladies: A Biography of Cancer.”
As Dr. Mukherjee wrote, Dr. SubbaRow was a “pioneer in many ways, a physician turned cellular physiologist, a chemist who had accidentally wandered into biology.” (Per Indian tradition, SubbaRow is the doctor’s first name, and Yellapragada is his surname, but medical literature uses SubbaRow as his cognomen, with some variations in spelling. Dr. Mukherjee wrote that his friends called him “Yella.”)
Dr. SubbaRow came to the United States in 1923, after enduring a difficult childhood and young adulthood. He’d survived bouts of religious fervor, childhood rebellion (including a bid to run away from home and become a banana trader), and a failed arranged marriage. His wife bore him a child who died in infancy. He left it all behind.
In Boston, medical officials rejected his degree. Broke, he worked for a time as a night porter at Brigham and Women’s Hospital in Boston, changing sheets and cleaning urinals. To a poor but proud high-caste Indian Brahmin, the culture shock of carrying out these tasks must have been especially jarring.
Dr. SubbaRow went on to earn a diploma from Harvard Medical School, also in Boston, and became a junior faculty member. As a foreigner, Dr. Mukherjee wrote, Dr. SubbaRow was a “reclusive, nocturnal, heavily accented vegetarian,” so different from his colleagues that advancement seemed impossible. Despite his pioneering biochemistry work, Harvard later declined to offer Dr. SubbaRow a tenured faculty position.
By the early 1940s, he took a job at an upstate New York pharmaceutical company called Lederle Labs (later purchased by Pfizer). At Lederle, Dr. SubbaRow strove to synthesize the vitamin known as folic acid. He ended up creating a kind of antivitamin, a lookalike that acted like folic acid but only succeeded in gumming up the works in receptors. But what good would it do to stop the body from absorbing folic acid? Plenty, it turned out.
Discoveries pile up, but credit and fame prove elusive
Dr. SubbaRow was no stranger to producing landmark biological work. He’d previously codiscovered phosphocreatine and ATP, which are crucial to muscular contractions. However, “in 1935, he had to disown the extent of his role in the discovery of the color test related to phosphorus, instead giving the credit to his co-author, who was being considered for promotion to a full professorship at Harvard,” wrote author Gerald Posner in his 2020 book, “Pharma: Greed, Lies and the Poisoning of America.”
Houston-area oncologist Kirtan Nautiyal, MD, who paid tribute to Dr. SubbaRow in a 2018 article, contended that “with his Indian instinct for self-effacement, he had irreparably sabotaged his own career.”
Dr. SubbaRow and his team also developed “the first effective treatment of filariasis, which causes elephantiasis of the lower limbs and genitals in millions of people, mainly in tropical countries,” Dr. Nautiyal wrote. “Later in the decade, his antibiotic program generated polymyxin, the first effective treatment against the class of bacteria called Gram negatives, and aureomycin, the first “broad-spectrum’ antibiotic.” (Aureomycin is also the first tetracycline antibiotic.)
Dr. SubbaRow’s discovery of a folic acid antagonist would again go largely unheralded. But first came the realization that folic acid made childhood leukemia worse, not better, and the prospect that this process could potentially be reversed.
Rise of methotrexate and fall of leukemia
In Boston, Sidney Farber, MD, a Boston pathologist, was desperate to help Robert Sandler, a 2-year-old leukemia patient. Dr. Farber contacted his ex-colleague Dr. SubbaRow to request a supply of aminopterin, an early version of methotrexate that Dr. SubbaRow and his team had developed. Dr. Farber injected Robert with the substance and within 3 days, the toddler’s white blood count started falling – fast. He stopped bleeding, resumed eating, and once again seemed almost identical to his twin brother, as Dr. Mukherjee wrote in his book.
Leukemia had never gone into remission before. Unfortunately, the treatment only worked temporarily. Robert, like other children treated with the drug, relapsed and died within months. But Dr. Farber “saw a door open” – a chemical, a kind of chemotherapy, that could turn back cancer. In the case of folic acid antagonists, they do so by stopping cancer cells from replicating.
Methotrexate, a related agent synthesized by Dr. SubbaRow, would become a mainstay of leukemia treatment and begin to produce long-term remission from acute lymphoblastic leukemia in 1970, when combination chemotherapy was developed.
Other cancers fell to methotrexate treatment. “Previous assumptions that cancer was nearly always fatal were revised, and the field of medical oncology (treatment of cancer with chemotherapy), which had not previously existed, was formally established in 1971,” according to the National Cancer Institute’s history of methotrexate. This account does not mention Dr. SubbaRow.
Death takes the doctor, but his legacy remains
In biographies, as well as his own words, Dr. SubbaRow comes across as a prickly, hard-driving workaholic who had little interest in intimate human connections. “It is not good to ask in every letter when I will be back,” he wrote to his wife back in India, before cutting off ties completely in the early 1930s. “I will come as early as possible. ... I do not want to write anything more.”
It seems, as his biographer S.P.K. Gupta noted, that “he was quite determined that the time allotted to him on Earth should be completely devoted to finding cures for ailments that plagued mankind.”
Still, Dr. SubbaRow’s research team was devoted to him, and he had plenty of reasons to be bitter, such as the prejudice and isolation he encountered in the United States and earlier, in British-run India. According to Mr. Posner’s book, even as a young medical student, Dr. SubbaRow heeded the call of Indian independence activist Mohandas Gandhi. He “refused the British surgical gown given him at school and instead donned a traditional and simple cotton Khadi. That act of defiance cost SubbaRow the college degree that was necessary for him to get into the State Medical College.”
During the last year of his life, Dr. SubbaRow faced yet another humiliation: In his landmark 1948 study about aminopterin as a treatment for leukemia, his colleague Dr. Farber failed to credit him, an “astonishing omission” as Yaddanapudi Ravindranath, MBBS, a pediatric hematologist/oncologist at Wayne State University, Detroit, put it. “From everything I know, Dr. Farber spent the rest of his career apologizing and trying to make amends for it,” Dr. Ravindranath said in an interview.
A career cut short, and a lasting legacy
In 1948, at the age of 53, Dr. SubbaRow suddenly died. “Many think Dr. SubbaRow would have won [the] Nobel Prize had he lived a few years longer,” said Dr. Ravindranath.
Like Dr. SubbaRow, Dr. Ravindranath was born in Andhra Pradesh state, near the city of Chennai formerly known as Madras. “Being a compatriot, in a way I continue his legacy, and I am obviously proud of him,” said Dr. Ravindranath, who has conducted his own landmark research regarding methotrexate and leukemia.
Nearly 75 years after Dr. SubbaRow’s death, Indian-born physicians like Dr. Ravindranath continue to honor him in print, trying to ensure that he’s not forgotten. Methotrexate remains a crucial treatment for leukemia, along with a long list of other ailments, including psoriasis.
Recognition for “Yella” may have come late and infrequently, but a Lederle Laboratories research library named after him offered Dr. SubbaRow a kind of immortality. A plaque there memorialized him in stone as a scientist, teacher, philosopher, and humanitarian, featuring the quote: “Science simply prolongs life. Religion deepens it.”
By all accounts, Dr. SubbaRow was a man of science and faith who had faith in science.
We have a half-forgotten Indian immigrant to thank – a hospital night porter turned biochemist –for revolutionizing treatment of leukemia, the once deadly childhood scourge that is still the most common pediatric cancer.
Dr. Yellapragada SubbaRow has been called the “father of chemotherapy” for developing methotrexate, a powerful, inexpensive therapy for leukemia and other diseases, and he is celebrated for additional scientific achievements. Yet Dr. SubbaRow’s life was marked more by struggle than glory.
Born poor in southeastern India, he nearly succumbed to a tropical disease that killed two older brothers, and he didn’t focus on schoolwork until his father died. Later, prejudice dogged his years as an immigrant to the United States, and a blood clot took his life at the age of 53.
Scientifically, however, Dr. SubbaRow (pronounced sue-buh-rao) triumphed, despite mammoth challenges and a lack of recognition that persists to this day. National Cancer Research Month is a fitting time to look back on his extraordinary life and work and pay tribute to his accomplishments.
‘Yella,’ folic acid, and a paradigm shift
No one appreciates Dr. SubbaRow more than a cadre of Indian-born physicians who have kept his legacy alive in journal articles, presentations, and a Pulitzer Prize-winning book. Among them is author and oncologist Siddhartha Mukherjee, MD, who chronicled Dr. SubbaRow’s achievements in his New York Times No. 1 bestseller, “The Emperor of All Maladies: A Biography of Cancer.”
As Dr. Mukherjee wrote, Dr. SubbaRow was a “pioneer in many ways, a physician turned cellular physiologist, a chemist who had accidentally wandered into biology.” (Per Indian tradition, SubbaRow is the doctor’s first name, and Yellapragada is his surname, but medical literature uses SubbaRow as his cognomen, with some variations in spelling. Dr. Mukherjee wrote that his friends called him “Yella.”)
Dr. SubbaRow came to the United States in 1923, after enduring a difficult childhood and young adulthood. He’d survived bouts of religious fervor, childhood rebellion (including a bid to run away from home and become a banana trader), and a failed arranged marriage. His wife bore him a child who died in infancy. He left it all behind.
In Boston, medical officials rejected his degree. Broke, he worked for a time as a night porter at Brigham and Women’s Hospital in Boston, changing sheets and cleaning urinals. To a poor but proud high-caste Indian Brahmin, the culture shock of carrying out these tasks must have been especially jarring.
Dr. SubbaRow went on to earn a diploma from Harvard Medical School, also in Boston, and became a junior faculty member. As a foreigner, Dr. Mukherjee wrote, Dr. SubbaRow was a “reclusive, nocturnal, heavily accented vegetarian,” so different from his colleagues that advancement seemed impossible. Despite his pioneering biochemistry work, Harvard later declined to offer Dr. SubbaRow a tenured faculty position.
By the early 1940s, he took a job at an upstate New York pharmaceutical company called Lederle Labs (later purchased by Pfizer). At Lederle, Dr. SubbaRow strove to synthesize the vitamin known as folic acid. He ended up creating a kind of antivitamin, a lookalike that acted like folic acid but only succeeded in gumming up the works in receptors. But what good would it do to stop the body from absorbing folic acid? Plenty, it turned out.
Discoveries pile up, but credit and fame prove elusive
Dr. SubbaRow was no stranger to producing landmark biological work. He’d previously codiscovered phosphocreatine and ATP, which are crucial to muscular contractions. However, “in 1935, he had to disown the extent of his role in the discovery of the color test related to phosphorus, instead giving the credit to his co-author, who was being considered for promotion to a full professorship at Harvard,” wrote author Gerald Posner in his 2020 book, “Pharma: Greed, Lies and the Poisoning of America.”
Houston-area oncologist Kirtan Nautiyal, MD, who paid tribute to Dr. SubbaRow in a 2018 article, contended that “with his Indian instinct for self-effacement, he had irreparably sabotaged his own career.”
Dr. SubbaRow and his team also developed “the first effective treatment of filariasis, which causes elephantiasis of the lower limbs and genitals in millions of people, mainly in tropical countries,” Dr. Nautiyal wrote. “Later in the decade, his antibiotic program generated polymyxin, the first effective treatment against the class of bacteria called Gram negatives, and aureomycin, the first “broad-spectrum’ antibiotic.” (Aureomycin is also the first tetracycline antibiotic.)
Dr. SubbaRow’s discovery of a folic acid antagonist would again go largely unheralded. But first came the realization that folic acid made childhood leukemia worse, not better, and the prospect that this process could potentially be reversed.
Rise of methotrexate and fall of leukemia
In Boston, Sidney Farber, MD, a Boston pathologist, was desperate to help Robert Sandler, a 2-year-old leukemia patient. Dr. Farber contacted his ex-colleague Dr. SubbaRow to request a supply of aminopterin, an early version of methotrexate that Dr. SubbaRow and his team had developed. Dr. Farber injected Robert with the substance and within 3 days, the toddler’s white blood count started falling – fast. He stopped bleeding, resumed eating, and once again seemed almost identical to his twin brother, as Dr. Mukherjee wrote in his book.
Leukemia had never gone into remission before. Unfortunately, the treatment only worked temporarily. Robert, like other children treated with the drug, relapsed and died within months. But Dr. Farber “saw a door open” – a chemical, a kind of chemotherapy, that could turn back cancer. In the case of folic acid antagonists, they do so by stopping cancer cells from replicating.
Methotrexate, a related agent synthesized by Dr. SubbaRow, would become a mainstay of leukemia treatment and begin to produce long-term remission from acute lymphoblastic leukemia in 1970, when combination chemotherapy was developed.
Other cancers fell to methotrexate treatment. “Previous assumptions that cancer was nearly always fatal were revised, and the field of medical oncology (treatment of cancer with chemotherapy), which had not previously existed, was formally established in 1971,” according to the National Cancer Institute’s history of methotrexate. This account does not mention Dr. SubbaRow.
Death takes the doctor, but his legacy remains
In biographies, as well as his own words, Dr. SubbaRow comes across as a prickly, hard-driving workaholic who had little interest in intimate human connections. “It is not good to ask in every letter when I will be back,” he wrote to his wife back in India, before cutting off ties completely in the early 1930s. “I will come as early as possible. ... I do not want to write anything more.”
It seems, as his biographer S.P.K. Gupta noted, that “he was quite determined that the time allotted to him on Earth should be completely devoted to finding cures for ailments that plagued mankind.”
Still, Dr. SubbaRow’s research team was devoted to him, and he had plenty of reasons to be bitter, such as the prejudice and isolation he encountered in the United States and earlier, in British-run India. According to Mr. Posner’s book, even as a young medical student, Dr. SubbaRow heeded the call of Indian independence activist Mohandas Gandhi. He “refused the British surgical gown given him at school and instead donned a traditional and simple cotton Khadi. That act of defiance cost SubbaRow the college degree that was necessary for him to get into the State Medical College.”
During the last year of his life, Dr. SubbaRow faced yet another humiliation: In his landmark 1948 study about aminopterin as a treatment for leukemia, his colleague Dr. Farber failed to credit him, an “astonishing omission” as Yaddanapudi Ravindranath, MBBS, a pediatric hematologist/oncologist at Wayne State University, Detroit, put it. “From everything I know, Dr. Farber spent the rest of his career apologizing and trying to make amends for it,” Dr. Ravindranath said in an interview.
A career cut short, and a lasting legacy
In 1948, at the age of 53, Dr. SubbaRow suddenly died. “Many think Dr. SubbaRow would have won [the] Nobel Prize had he lived a few years longer,” said Dr. Ravindranath.
Like Dr. SubbaRow, Dr. Ravindranath was born in Andhra Pradesh state, near the city of Chennai formerly known as Madras. “Being a compatriot, in a way I continue his legacy, and I am obviously proud of him,” said Dr. Ravindranath, who has conducted his own landmark research regarding methotrexate and leukemia.
Nearly 75 years after Dr. SubbaRow’s death, Indian-born physicians like Dr. Ravindranath continue to honor him in print, trying to ensure that he’s not forgotten. Methotrexate remains a crucial treatment for leukemia, along with a long list of other ailments, including psoriasis.
Recognition for “Yella” may have come late and infrequently, but a Lederle Laboratories research library named after him offered Dr. SubbaRow a kind of immortality. A plaque there memorialized him in stone as a scientist, teacher, philosopher, and humanitarian, featuring the quote: “Science simply prolongs life. Religion deepens it.”
By all accounts, Dr. SubbaRow was a man of science and faith who had faith in science.
Medical education programs tell how climate change affects health
Ms. Manivannan, copresident of Emory Medical Students for Climate Action, was in the first class of Emory’s medical students to experience the birth of a refined curriculum – lobbied for and partially created by students themselves. The new course of study addresses the myriad ways climate affects health: from air pollution and its effects on the lungs and cardiovascular system to heat-related kidney disease.
“We have known that climate has affected health for decades,” Ms. Manivannan said in a recent interview. “The narrative used to be that icebergs were melting and in 2050 polar bears would be extinct. The piece that’s different now is people are linking climate to increases in asthma and various diseases. We have a way to directly communicate that it’s not a far-off thing. It’s happening to your friends and family right now.”
Hospitals, medical schools, and public health programs are stepping up to educate the next generation of doctors as well as veteran medical workers on one of the most widespread, insidious health threats of our time – climate change – and specific ways it could affect their patients.
Although climate change may seem to many Americans like a distant threat, Marilyn Howarth, MD, a pediatrician in Philadelphia, is trying to make sure physicians are better prepared to treat a growing number of health problems associated with global warming.
“There isn’t a lot of education for pediatricians and internists on environmental health issues. It has not been a standard part of education in medical school or residency training,” Dr. Howarth, deputy director of the new Philadelphia Regional Center for Children’s Environmental Health, said. “With increasing attention on our climate, we really recognize there’s a real gap in physician knowledge, both in pediatric and adult care.”
Scientists have found that climate change can alter just about every system within the human body. Studies show that more extreme weather events, such as heat waves, thunderstorms, and floods, can worsen asthma and produce more pollen and mold, triggering debilitating respiratory problems.
According to the American Lung Association, ultrafine particles of air pollution can be inhaled and then travel throughout the bloodstream, wreaking havoc on organs and increasing risk of heart attack and stroke. Various types of air pollution also cause changes to the climate by trapping heat in the atmosphere, which leads to problems such as rising sea levels and extreme weather. Plus, in a new study published in Nature, scientists warn that warming climates are forcing animals to migrate to different areas, raising the risk that new infectious diseases will hop from animals – such as bats – to humans, a process called “zoonotic spillover” that many researchers believe is responsible for the COVID-19 pandemic.
The Philadelphia Regional Center for Children’s Environmental Health
One of the latest initiatives aimed at disseminating information about children’s health to health care providers is the Philadelphia Regional Center for Children’s Environmental Health, part of Children’s Hospital of Philadelphia and Penn Medicine. CHOP and Penn Medicine are jointly funding this center’s work, which will include educating health care providers on how to better screen for climate-caused health risks and treat related conditions, such as lead poisoning and asthma.
Outreach will focus on providers who treat patients with illnesses that researchers have linked to climate change, Dr. Howarth said. The center will offer clinicians access to seminars and webinars, along with online resources to help doctors treat environmental illnesses. For example, doctors at CHOP’s Poison Control Center are developing a toolkit for physicians to treat patients with elevated levels of lead in the blood. Scientists have linked extreme weather events related to climate change to flooding that pushes metals away from river banks where they were previously contained, allowing them to more easily contaminate homes, soils, and yards.
The initiative builds on CHOP’s Community Asthma Prevention Program (CAPP), which was launched in 1997 by Tyra Bryant-Stephens, MD, its current medical director. CAPP deploys community health workers into homes armed with supplies and tips for managing asthma. The new center will use similar tactics to provide education and resources to patients. The goal is to reach as many at-risk local children as possible.
Future generation of doctors fuel growth in climate change education
Lisa Doggett, MD, cofounder and president of the board of directors of Texas Physicians for Social Responsibility, announced in March that the University of Texas at Austin, Baylor College of Medicine, Houston, and the University of Texas Southwestern in Dallas have all decided to begin offering a course on environmental threats. Emory’s new curriculum has become more comprehensive every year since its start – thanks in part to the input of students like Ms. Manivannan. Faculty members tasked her with approving the new additions to the curriculum on how climate affects health, which in 2019 had consisted of a few slides about issues such as extreme heat exposure and air pollution and their effects on childbirth outcomes.
Material on climate change has now been woven into 13 courses. It is discussed at length in relation to pulmonology, cardiology, and gastropulmonology, for example, said Rebecca Philipsborn, MD, MPA, FAAP, faculty lead for the environmental and health curriculum at Emory.
The curriculum has only been incorporated into Emory’s program for the past 2 years. Dr. Philipsborn said the school plans to expand it to the clinical years to help trainees learn to treat conditions such as pediatric asthma.
“In the past few years, there has been so much momentum, and part of that is a testament to already seeing effects of climate change and how they affect delivery of health care,” she said.
At least one medical journal has recently ramped up its efforts to educate physicians on the links between health issues and climate change. Editors of Family Practice, from Oxford University Press, have announced that they plan to publish a special Climate Crisis and Primary Health Care issue in September.
Of course, not all climate initiatives in medicine are new. A select few have existed for decades.
But only now are physicians widely seeing the links between health and environment, according to Aaron Bernstein, MD, MPH, interim director of the Center for Climate, Health, and the Global Environment (C-CHANGE) at Harvard School of Public Health, Boston.
C-CHANGE, founded in 1996, was the first center in the world to focus on the health effects of environmental change.
“It’s taken 20 years, but what we’re seeing, I think, is the fruits of education,” Dr. Bernstein said. “There’s clearly a wave building here, and I think it really started with education and people younger than the people in charge calling them into account.”
Like the Philadelphia center, Harvard’s program conducts research on climate and health and educates people from high schoolers to health care veterans. Dr. Bernstein helps lead Climate MD, a program that aims to prepare health care workers for climate crises. The Climate MD team has published several articles in peer-reviewed journals on how to better treat patients struggling with environmental health problems. For example, an article on mapping patients in hurricane zones helped shed light on how systems can identify climate-vulnerable patients using public data.
They also developed a tool to help pediatricians provide “climate-informed primary care” – guidance on how to assess whether children are at risk of any harmful environmental exposures, a feature that is not part of standard pediatric visits.
Like the other programs, Climate MD uses community outreach to treat as many local patients as possible. Staff work with providers at more than 100 health clinics, particularly in areas where climate change disproportionately affects residents.
The next major step is to bring some of this into clinical practice, Dr. Bernstein said. In February 2020, C-CHANGE held its first symposium to address that issue.
“The key is to understand climate issues from a provider’s perspective,” he said. “Then those issues can really be brought to the bedside.”
A version of this article first appeared on Medscape.com.
Ms. Manivannan, copresident of Emory Medical Students for Climate Action, was in the first class of Emory’s medical students to experience the birth of a refined curriculum – lobbied for and partially created by students themselves. The new course of study addresses the myriad ways climate affects health: from air pollution and its effects on the lungs and cardiovascular system to heat-related kidney disease.
“We have known that climate has affected health for decades,” Ms. Manivannan said in a recent interview. “The narrative used to be that icebergs were melting and in 2050 polar bears would be extinct. The piece that’s different now is people are linking climate to increases in asthma and various diseases. We have a way to directly communicate that it’s not a far-off thing. It’s happening to your friends and family right now.”
Hospitals, medical schools, and public health programs are stepping up to educate the next generation of doctors as well as veteran medical workers on one of the most widespread, insidious health threats of our time – climate change – and specific ways it could affect their patients.
Although climate change may seem to many Americans like a distant threat, Marilyn Howarth, MD, a pediatrician in Philadelphia, is trying to make sure physicians are better prepared to treat a growing number of health problems associated with global warming.
“There isn’t a lot of education for pediatricians and internists on environmental health issues. It has not been a standard part of education in medical school or residency training,” Dr. Howarth, deputy director of the new Philadelphia Regional Center for Children’s Environmental Health, said. “With increasing attention on our climate, we really recognize there’s a real gap in physician knowledge, both in pediatric and adult care.”
Scientists have found that climate change can alter just about every system within the human body. Studies show that more extreme weather events, such as heat waves, thunderstorms, and floods, can worsen asthma and produce more pollen and mold, triggering debilitating respiratory problems.
According to the American Lung Association, ultrafine particles of air pollution can be inhaled and then travel throughout the bloodstream, wreaking havoc on organs and increasing risk of heart attack and stroke. Various types of air pollution also cause changes to the climate by trapping heat in the atmosphere, which leads to problems such as rising sea levels and extreme weather. Plus, in a new study published in Nature, scientists warn that warming climates are forcing animals to migrate to different areas, raising the risk that new infectious diseases will hop from animals – such as bats – to humans, a process called “zoonotic spillover” that many researchers believe is responsible for the COVID-19 pandemic.
The Philadelphia Regional Center for Children’s Environmental Health
One of the latest initiatives aimed at disseminating information about children’s health to health care providers is the Philadelphia Regional Center for Children’s Environmental Health, part of Children’s Hospital of Philadelphia and Penn Medicine. CHOP and Penn Medicine are jointly funding this center’s work, which will include educating health care providers on how to better screen for climate-caused health risks and treat related conditions, such as lead poisoning and asthma.
Outreach will focus on providers who treat patients with illnesses that researchers have linked to climate change, Dr. Howarth said. The center will offer clinicians access to seminars and webinars, along with online resources to help doctors treat environmental illnesses. For example, doctors at CHOP’s Poison Control Center are developing a toolkit for physicians to treat patients with elevated levels of lead in the blood. Scientists have linked extreme weather events related to climate change to flooding that pushes metals away from river banks where they were previously contained, allowing them to more easily contaminate homes, soils, and yards.
The initiative builds on CHOP’s Community Asthma Prevention Program (CAPP), which was launched in 1997 by Tyra Bryant-Stephens, MD, its current medical director. CAPP deploys community health workers into homes armed with supplies and tips for managing asthma. The new center will use similar tactics to provide education and resources to patients. The goal is to reach as many at-risk local children as possible.
Future generation of doctors fuel growth in climate change education
Lisa Doggett, MD, cofounder and president of the board of directors of Texas Physicians for Social Responsibility, announced in March that the University of Texas at Austin, Baylor College of Medicine, Houston, and the University of Texas Southwestern in Dallas have all decided to begin offering a course on environmental threats. Emory’s new curriculum has become more comprehensive every year since its start – thanks in part to the input of students like Ms. Manivannan. Faculty members tasked her with approving the new additions to the curriculum on how climate affects health, which in 2019 had consisted of a few slides about issues such as extreme heat exposure and air pollution and their effects on childbirth outcomes.
Material on climate change has now been woven into 13 courses. It is discussed at length in relation to pulmonology, cardiology, and gastropulmonology, for example, said Rebecca Philipsborn, MD, MPA, FAAP, faculty lead for the environmental and health curriculum at Emory.
The curriculum has only been incorporated into Emory’s program for the past 2 years. Dr. Philipsborn said the school plans to expand it to the clinical years to help trainees learn to treat conditions such as pediatric asthma.
“In the past few years, there has been so much momentum, and part of that is a testament to already seeing effects of climate change and how they affect delivery of health care,” she said.
At least one medical journal has recently ramped up its efforts to educate physicians on the links between health issues and climate change. Editors of Family Practice, from Oxford University Press, have announced that they plan to publish a special Climate Crisis and Primary Health Care issue in September.
Of course, not all climate initiatives in medicine are new. A select few have existed for decades.
But only now are physicians widely seeing the links between health and environment, according to Aaron Bernstein, MD, MPH, interim director of the Center for Climate, Health, and the Global Environment (C-CHANGE) at Harvard School of Public Health, Boston.
C-CHANGE, founded in 1996, was the first center in the world to focus on the health effects of environmental change.
“It’s taken 20 years, but what we’re seeing, I think, is the fruits of education,” Dr. Bernstein said. “There’s clearly a wave building here, and I think it really started with education and people younger than the people in charge calling them into account.”
Like the Philadelphia center, Harvard’s program conducts research on climate and health and educates people from high schoolers to health care veterans. Dr. Bernstein helps lead Climate MD, a program that aims to prepare health care workers for climate crises. The Climate MD team has published several articles in peer-reviewed journals on how to better treat patients struggling with environmental health problems. For example, an article on mapping patients in hurricane zones helped shed light on how systems can identify climate-vulnerable patients using public data.
They also developed a tool to help pediatricians provide “climate-informed primary care” – guidance on how to assess whether children are at risk of any harmful environmental exposures, a feature that is not part of standard pediatric visits.
Like the other programs, Climate MD uses community outreach to treat as many local patients as possible. Staff work with providers at more than 100 health clinics, particularly in areas where climate change disproportionately affects residents.
The next major step is to bring some of this into clinical practice, Dr. Bernstein said. In February 2020, C-CHANGE held its first symposium to address that issue.
“The key is to understand climate issues from a provider’s perspective,” he said. “Then those issues can really be brought to the bedside.”
A version of this article first appeared on Medscape.com.
Ms. Manivannan, copresident of Emory Medical Students for Climate Action, was in the first class of Emory’s medical students to experience the birth of a refined curriculum – lobbied for and partially created by students themselves. The new course of study addresses the myriad ways climate affects health: from air pollution and its effects on the lungs and cardiovascular system to heat-related kidney disease.
“We have known that climate has affected health for decades,” Ms. Manivannan said in a recent interview. “The narrative used to be that icebergs were melting and in 2050 polar bears would be extinct. The piece that’s different now is people are linking climate to increases in asthma and various diseases. We have a way to directly communicate that it’s not a far-off thing. It’s happening to your friends and family right now.”
Hospitals, medical schools, and public health programs are stepping up to educate the next generation of doctors as well as veteran medical workers on one of the most widespread, insidious health threats of our time – climate change – and specific ways it could affect their patients.
Although climate change may seem to many Americans like a distant threat, Marilyn Howarth, MD, a pediatrician in Philadelphia, is trying to make sure physicians are better prepared to treat a growing number of health problems associated with global warming.
“There isn’t a lot of education for pediatricians and internists on environmental health issues. It has not been a standard part of education in medical school or residency training,” Dr. Howarth, deputy director of the new Philadelphia Regional Center for Children’s Environmental Health, said. “With increasing attention on our climate, we really recognize there’s a real gap in physician knowledge, both in pediatric and adult care.”
Scientists have found that climate change can alter just about every system within the human body. Studies show that more extreme weather events, such as heat waves, thunderstorms, and floods, can worsen asthma and produce more pollen and mold, triggering debilitating respiratory problems.
According to the American Lung Association, ultrafine particles of air pollution can be inhaled and then travel throughout the bloodstream, wreaking havoc on organs and increasing risk of heart attack and stroke. Various types of air pollution also cause changes to the climate by trapping heat in the atmosphere, which leads to problems such as rising sea levels and extreme weather. Plus, in a new study published in Nature, scientists warn that warming climates are forcing animals to migrate to different areas, raising the risk that new infectious diseases will hop from animals – such as bats – to humans, a process called “zoonotic spillover” that many researchers believe is responsible for the COVID-19 pandemic.
The Philadelphia Regional Center for Children’s Environmental Health
One of the latest initiatives aimed at disseminating information about children’s health to health care providers is the Philadelphia Regional Center for Children’s Environmental Health, part of Children’s Hospital of Philadelphia and Penn Medicine. CHOP and Penn Medicine are jointly funding this center’s work, which will include educating health care providers on how to better screen for climate-caused health risks and treat related conditions, such as lead poisoning and asthma.
Outreach will focus on providers who treat patients with illnesses that researchers have linked to climate change, Dr. Howarth said. The center will offer clinicians access to seminars and webinars, along with online resources to help doctors treat environmental illnesses. For example, doctors at CHOP’s Poison Control Center are developing a toolkit for physicians to treat patients with elevated levels of lead in the blood. Scientists have linked extreme weather events related to climate change to flooding that pushes metals away from river banks where they were previously contained, allowing them to more easily contaminate homes, soils, and yards.
The initiative builds on CHOP’s Community Asthma Prevention Program (CAPP), which was launched in 1997 by Tyra Bryant-Stephens, MD, its current medical director. CAPP deploys community health workers into homes armed with supplies and tips for managing asthma. The new center will use similar tactics to provide education and resources to patients. The goal is to reach as many at-risk local children as possible.
Future generation of doctors fuel growth in climate change education
Lisa Doggett, MD, cofounder and president of the board of directors of Texas Physicians for Social Responsibility, announced in March that the University of Texas at Austin, Baylor College of Medicine, Houston, and the University of Texas Southwestern in Dallas have all decided to begin offering a course on environmental threats. Emory’s new curriculum has become more comprehensive every year since its start – thanks in part to the input of students like Ms. Manivannan. Faculty members tasked her with approving the new additions to the curriculum on how climate affects health, which in 2019 had consisted of a few slides about issues such as extreme heat exposure and air pollution and their effects on childbirth outcomes.
Material on climate change has now been woven into 13 courses. It is discussed at length in relation to pulmonology, cardiology, and gastropulmonology, for example, said Rebecca Philipsborn, MD, MPA, FAAP, faculty lead for the environmental and health curriculum at Emory.
The curriculum has only been incorporated into Emory’s program for the past 2 years. Dr. Philipsborn said the school plans to expand it to the clinical years to help trainees learn to treat conditions such as pediatric asthma.
“In the past few years, there has been so much momentum, and part of that is a testament to already seeing effects of climate change and how they affect delivery of health care,” she said.
At least one medical journal has recently ramped up its efforts to educate physicians on the links between health issues and climate change. Editors of Family Practice, from Oxford University Press, have announced that they plan to publish a special Climate Crisis and Primary Health Care issue in September.
Of course, not all climate initiatives in medicine are new. A select few have existed for decades.
But only now are physicians widely seeing the links between health and environment, according to Aaron Bernstein, MD, MPH, interim director of the Center for Climate, Health, and the Global Environment (C-CHANGE) at Harvard School of Public Health, Boston.
C-CHANGE, founded in 1996, was the first center in the world to focus on the health effects of environmental change.
“It’s taken 20 years, but what we’re seeing, I think, is the fruits of education,” Dr. Bernstein said. “There’s clearly a wave building here, and I think it really started with education and people younger than the people in charge calling them into account.”
Like the Philadelphia center, Harvard’s program conducts research on climate and health and educates people from high schoolers to health care veterans. Dr. Bernstein helps lead Climate MD, a program that aims to prepare health care workers for climate crises. The Climate MD team has published several articles in peer-reviewed journals on how to better treat patients struggling with environmental health problems. For example, an article on mapping patients in hurricane zones helped shed light on how systems can identify climate-vulnerable patients using public data.
They also developed a tool to help pediatricians provide “climate-informed primary care” – guidance on how to assess whether children are at risk of any harmful environmental exposures, a feature that is not part of standard pediatric visits.
Like the other programs, Climate MD uses community outreach to treat as many local patients as possible. Staff work with providers at more than 100 health clinics, particularly in areas where climate change disproportionately affects residents.
The next major step is to bring some of this into clinical practice, Dr. Bernstein said. In February 2020, C-CHANGE held its first symposium to address that issue.
“The key is to understand climate issues from a provider’s perspective,” he said. “Then those issues can really be brought to the bedside.”
A version of this article first appeared on Medscape.com.
Children and COVID: New cases climb slowly but steadily
The current sustained increase in COVID-19 has brought the total number of cases in children to over 13 million since the start of the pandemic, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, when cases dropped to their lowest point since last summer. The cumulative number of cases in children is 13,052,988, which accounts for 19.0% of all cases reported in the United States, the AAP and CHA said in their weekly COVID-19 report.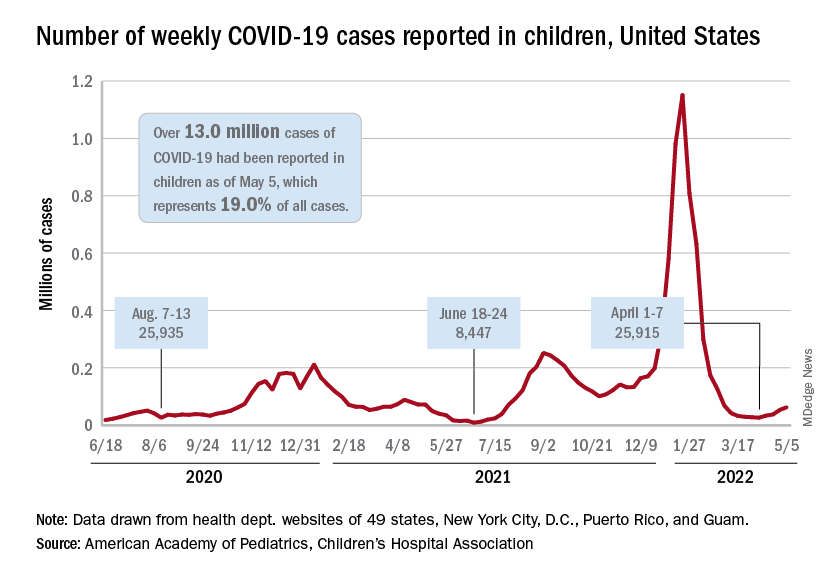
Other measures of incidence show the same steady rise. The rate of new admissions of children aged 0-17 with confirmed COVID-19, which had dipped as low as 0.13 per 100,000 population on April 11, was up to 0.19 per 100,000 on May 6, and the 7-day average for total admissions was 136 per day for May 1-7, compared with 118 for the last week of April, according to the Centers for Disease Control and Prevention.
At the state level, new admission rates for May 6 show wide variation, even regionally. Rhode Island came in with a 0.00 per 100,000 on that day, while Vermont recorded 0.88 admissions per 100,000, the highest of any state and lower only than the District of Columbia’s 1.23 per 100,000. Connecticut (0.45) and Massachusetts (0.33) also were in the highest group (see map), while Maine was in the lowest, CDC data show.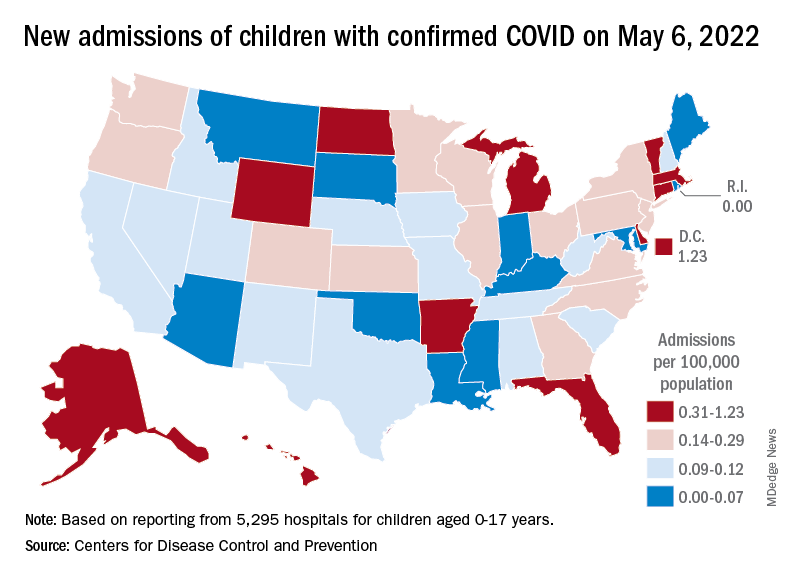
Nationally, emergency department visits also have been rising over the last month or so. Children aged 0-11 years, who were down to a 7-day average of 0.5% of ED visits with diagnosed COVID-19 in early April, saw that number rise to 1.4% on May 5. Children aged 12-15 years went from a rate of 0.3% in late March to the current 1.2%, as did 16- to 17-year-olds, the CDC said on its COVID Data Tracker.
The vaccination effort, meanwhile, continues to lose steam, at least among children who are currently eligible. Initial vaccinations in those aged 5-11 slipped to their lowest-ever 1-week total, 47,000 for April 28 to May 4, while children aged 16-17 continued a long-term slide that has the weekly count down to just 29,000, the AAP said in its weekly vaccination report.
Here’s how those latest recipients changed the populations of vaccinated children in the last week: 35.4% of all 5- to 11-year-olds had received at least one dose as of May 4, compared with 35.3% on April 27, with increases from 67.4% to 67.5% for 12- to 15-year-olds and 72.7% to 72.8% among those aged 16-17, the CDC reported.
The current sustained increase in COVID-19 has brought the total number of cases in children to over 13 million since the start of the pandemic, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, when cases dropped to their lowest point since last summer. The cumulative number of cases in children is 13,052,988, which accounts for 19.0% of all cases reported in the United States, the AAP and CHA said in their weekly COVID-19 report.
Other measures of incidence show the same steady rise. The rate of new admissions of children aged 0-17 with confirmed COVID-19, which had dipped as low as 0.13 per 100,000 population on April 11, was up to 0.19 per 100,000 on May 6, and the 7-day average for total admissions was 136 per day for May 1-7, compared with 118 for the last week of April, according to the Centers for Disease Control and Prevention.
At the state level, new admission rates for May 6 show wide variation, even regionally. Rhode Island came in with a 0.00 per 100,000 on that day, while Vermont recorded 0.88 admissions per 100,000, the highest of any state and lower only than the District of Columbia’s 1.23 per 100,000. Connecticut (0.45) and Massachusetts (0.33) also were in the highest group (see map), while Maine was in the lowest, CDC data show.
Nationally, emergency department visits also have been rising over the last month or so. Children aged 0-11 years, who were down to a 7-day average of 0.5% of ED visits with diagnosed COVID-19 in early April, saw that number rise to 1.4% on May 5. Children aged 12-15 years went from a rate of 0.3% in late March to the current 1.2%, as did 16- to 17-year-olds, the CDC said on its COVID Data Tracker.
The vaccination effort, meanwhile, continues to lose steam, at least among children who are currently eligible. Initial vaccinations in those aged 5-11 slipped to their lowest-ever 1-week total, 47,000 for April 28 to May 4, while children aged 16-17 continued a long-term slide that has the weekly count down to just 29,000, the AAP said in its weekly vaccination report.
Here’s how those latest recipients changed the populations of vaccinated children in the last week: 35.4% of all 5- to 11-year-olds had received at least one dose as of May 4, compared with 35.3% on April 27, with increases from 67.4% to 67.5% for 12- to 15-year-olds and 72.7% to 72.8% among those aged 16-17, the CDC reported.
The current sustained increase in COVID-19 has brought the total number of cases in children to over 13 million since the start of the pandemic, according to the American Academy of Pediatrics and the Children’s Hospital Association.
, when cases dropped to their lowest point since last summer. The cumulative number of cases in children is 13,052,988, which accounts for 19.0% of all cases reported in the United States, the AAP and CHA said in their weekly COVID-19 report.
Other measures of incidence show the same steady rise. The rate of new admissions of children aged 0-17 with confirmed COVID-19, which had dipped as low as 0.13 per 100,000 population on April 11, was up to 0.19 per 100,000 on May 6, and the 7-day average for total admissions was 136 per day for May 1-7, compared with 118 for the last week of April, according to the Centers for Disease Control and Prevention.
At the state level, new admission rates for May 6 show wide variation, even regionally. Rhode Island came in with a 0.00 per 100,000 on that day, while Vermont recorded 0.88 admissions per 100,000, the highest of any state and lower only than the District of Columbia’s 1.23 per 100,000. Connecticut (0.45) and Massachusetts (0.33) also were in the highest group (see map), while Maine was in the lowest, CDC data show.
Nationally, emergency department visits also have been rising over the last month or so. Children aged 0-11 years, who were down to a 7-day average of 0.5% of ED visits with diagnosed COVID-19 in early April, saw that number rise to 1.4% on May 5. Children aged 12-15 years went from a rate of 0.3% in late March to the current 1.2%, as did 16- to 17-year-olds, the CDC said on its COVID Data Tracker.
The vaccination effort, meanwhile, continues to lose steam, at least among children who are currently eligible. Initial vaccinations in those aged 5-11 slipped to their lowest-ever 1-week total, 47,000 for April 28 to May 4, while children aged 16-17 continued a long-term slide that has the weekly count down to just 29,000, the AAP said in its weekly vaccination report.
Here’s how those latest recipients changed the populations of vaccinated children in the last week: 35.4% of all 5- to 11-year-olds had received at least one dose as of May 4, compared with 35.3% on April 27, with increases from 67.4% to 67.5% for 12- to 15-year-olds and 72.7% to 72.8% among those aged 16-17, the CDC reported.
Anorexia nervosa in adolescent patients: What pediatricians need to know
Eating disorders are among the most prevalent, disabling, and potentially fatal psychiatric illnesses, and the COVID-19 pandemic has exacerbated their burden, with a 15.3% increase in incidence in 2020 compared with previous years.1 This increase was almost solely among adolescent girls with anorexia nervosa (AN), which is often insidious in onset and more difficult to treat as it advances. Adolescents with AN are most likely to present to their pediatricians, so awareness and early recognition of the symptoms is critical. Pediatricians are also an integral part of the treatment team in AN and can offer monitoring for serious complications, alongside valuable guidance to parents, who are central to treatment and the reestablishment of healthy eating habits in their children. Here we will review the epidemiology, diagnosis, and treatment of anorexia, with an emphasis on what pediatricians need to know to screen and to facilitate treatment.
Epidemiology
AN is marked by a fear of gaining weight or behaviors that interfere with weight gain and a self-evaluation unduly influenced by weight and body shape. Youth with AN often deny the seriousness of their malnutrition, although that is not required for diagnosis. AN can be of a restrictive or binge-purge subtype, and amenorrhea is no longer a requirement for diagnosis. There is not a specific weight or body mass index cutoff for the diagnosis, but the severity of AN is determined by the BMI percentile normed to age and sex. The average age of onset is 18, and the prepandemic prevalence of AN was about 1% of the population. It affects about 10 times as many females as males. It is quite rare prior to puberty, affecting about 0.01% of that age group. There is a heritable component, with a fivefold relative risk in youth with a parent with AN, and twin studies suggest heritability rates as high as 75%. Youth with rigid cognitive styles appear more vulnerable, as do those who participate in activities such as ballet, gymnastics, modeling, and wrestling because of the role of appearance and weight in performance. More than half of patients with AN will have another psychiatric illness, most commonly anxiety disorders, depression, or obsessive-compulsive disorder. AN becomes chronic in up to 15% of sufferers and the mortality rate is close to 10%, with approximately half dying from medical complications and half dying by suicide.
Screening
Parents and pediatricians are usually the first to notice that a child has started to lose weight or is falling off the growth curve. But weight changes usually emerge after feelings of preoccupation with weight, body shape, and body satisfaction. If parents report escalating pickiness around food, increased or compulsive exercise, persistent self-consciousness and self-criticism around weight and body shape, it is worth starting with screening questions.
If you notice preoccupation or anxiety around being weighed, even if the weight or growth curve are still normal, it is worthwhile to screen. Screening questions, such as the SCOFF questionnaire with five simple questions, can be very sensitive for both AN and bulimia nervosa.2 There are also many validated screening instruments, such as the Eating Disorder Inventory or Eating Attitudes Test (for adolescents) and the Kids Eating Disorder Survey and the Child Eating Attitudes Test (for younger children), that are short self-reports that you can have your patients fill out when you have a higher index of suspicion. Weight loss or growth failure without a preoccupation around weight or appearance needs a thorough a medical workup, and could be a function of other psychiatric problems, such as depression.
If a child screens positive for an eating disorder, your full physical examination, growth curves, and longitudinal growth charts are critical for diagnosis. Percentile BMIs must be used, given the inaccuracy of standard BMI calculations in this age group. (Centers for Disease Control and Prevention age and sex growth charts include methods for this calculation). Laboratory assessment, including metabolic, kidney, pancreatic, and thyroid function, and an EKG can illuminate if there are consequences of restricting or purging. Of course, you want to evaluate for significant medical symptoms, including bradycardia, orthostasis, and hypokalemia. These medical symptoms are not limited to the severely underweight and merit referral to an emergency department and possible medical admission.
Then, a referral to a clinician who is expert in the assessment and treatment of eating disorders is needed. This may be a child psychiatrist, psychologist, or a colleague pediatrician with this specialization. It is also very important to begin the conversation with the family to introduce your concerns, describe what you have noticed, and discuss the need for further assessment and possibly treatment.
Be mindful that discussing this in front of your patient may heighten the patient’s anxiety or distress. Be prepared to offer support and understanding for your patient’s anxiety, while steadfastly providing absolute clarity for the parents about the necessity of further evaluation and treatment. Many parents will be concerned and ready to do whatever is needed to get their child’s eating and growth back on track. But some parents may have more difficulty. They may have their own history with an eating disorder. They may be avoiding a sense of shame or alarm. They may be eager to avoid adding to their child’s stress. They may be tired of engaging in power struggles with the child. They may be proud of their ambitious, accomplished young athlete. Their trust in you makes you uniquely positioned to complicate their thinking. And treatment will hinge on them, so this is a critical bridge to care.
Beyond telling parents that they will need to bring more structure and supervision to mealtimes to begin addressing their child’s nutrition, you might offer guidance on other strategies. Empower parents to limit their child’s use of social media sites such as Instagram, YouTube, and TikTok, where they may be immersed in comparing themselves to idealized (and airbrushed) influencers. Empower them to make their child’s participation in beloved sports contingent on eating meals together and completely or on a stabilized weight (as will be common in treatment). Remind them that there are no bad foods, that the goal is health, and that they are not in a power struggle with their child, but instead allied with their child to treat AN. Remind them to also look for chances to have fun with their child, to help everyone remember what matters.
Treatment
Family-based therapy (FBT) is the first-line treatment of shorter-duration AN in children and adolescents. It focuses on the parents, helping them to calmly and effectively manage their child’s eating behaviors until their weight and behaviors have normalized. As a patient’s nutritional status improves, so does cognitive function, emotional flexibility, and mood. Individual therapy and psychopharmacologic treatment can be very effective for comorbid anxiety, mood, attentional, and thought disorders. Family-based work does include the child and is often done in group-based settings with clinicians from multiple disciplines. Dietitians provide education and guidance about healthy nutrition to the child and parents. Therapists may work with the child, parents, or full family to focus on behavior modification and managing distress. Most academic medical centers provide access to FBT, but there are many regions with no providers of this evidence-based treatment. One of the silver linings of the COVID-19 pandemic is that several online services have emerged offering FBT, working with families to manage mealtimes and treatment entirely at home.3 Pediatricians provide regular medical checks to measure progress and help with decisions about when it is safe to permit exercise or advance privileges and independence around eating. Some pediatricians have discovered a deep interest in this area of pediatrics and built their practices on it. Given the surge in prevalence of AN and the needs for adolescent mental health services, we hope more will do so.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected].
References
1. Taquet M et al. Br J Psychiatry. 2022;220:262-4.
2. Morgan JF et al. West J Med. 2000 Mar;172(3):164-5.
3. Matheson BE et al. Int J Eat Disord. 2020 Jul;53(7):1142-54.
Eating disorders are among the most prevalent, disabling, and potentially fatal psychiatric illnesses, and the COVID-19 pandemic has exacerbated their burden, with a 15.3% increase in incidence in 2020 compared with previous years.1 This increase was almost solely among adolescent girls with anorexia nervosa (AN), which is often insidious in onset and more difficult to treat as it advances. Adolescents with AN are most likely to present to their pediatricians, so awareness and early recognition of the symptoms is critical. Pediatricians are also an integral part of the treatment team in AN and can offer monitoring for serious complications, alongside valuable guidance to parents, who are central to treatment and the reestablishment of healthy eating habits in their children. Here we will review the epidemiology, diagnosis, and treatment of anorexia, with an emphasis on what pediatricians need to know to screen and to facilitate treatment.
Epidemiology
AN is marked by a fear of gaining weight or behaviors that interfere with weight gain and a self-evaluation unduly influenced by weight and body shape. Youth with AN often deny the seriousness of their malnutrition, although that is not required for diagnosis. AN can be of a restrictive or binge-purge subtype, and amenorrhea is no longer a requirement for diagnosis. There is not a specific weight or body mass index cutoff for the diagnosis, but the severity of AN is determined by the BMI percentile normed to age and sex. The average age of onset is 18, and the prepandemic prevalence of AN was about 1% of the population. It affects about 10 times as many females as males. It is quite rare prior to puberty, affecting about 0.01% of that age group. There is a heritable component, with a fivefold relative risk in youth with a parent with AN, and twin studies suggest heritability rates as high as 75%. Youth with rigid cognitive styles appear more vulnerable, as do those who participate in activities such as ballet, gymnastics, modeling, and wrestling because of the role of appearance and weight in performance. More than half of patients with AN will have another psychiatric illness, most commonly anxiety disorders, depression, or obsessive-compulsive disorder. AN becomes chronic in up to 15% of sufferers and the mortality rate is close to 10%, with approximately half dying from medical complications and half dying by suicide.
Screening
Parents and pediatricians are usually the first to notice that a child has started to lose weight or is falling off the growth curve. But weight changes usually emerge after feelings of preoccupation with weight, body shape, and body satisfaction. If parents report escalating pickiness around food, increased or compulsive exercise, persistent self-consciousness and self-criticism around weight and body shape, it is worth starting with screening questions.
If you notice preoccupation or anxiety around being weighed, even if the weight or growth curve are still normal, it is worthwhile to screen. Screening questions, such as the SCOFF questionnaire with five simple questions, can be very sensitive for both AN and bulimia nervosa.2 There are also many validated screening instruments, such as the Eating Disorder Inventory or Eating Attitudes Test (for adolescents) and the Kids Eating Disorder Survey and the Child Eating Attitudes Test (for younger children), that are short self-reports that you can have your patients fill out when you have a higher index of suspicion. Weight loss or growth failure without a preoccupation around weight or appearance needs a thorough a medical workup, and could be a function of other psychiatric problems, such as depression.
If a child screens positive for an eating disorder, your full physical examination, growth curves, and longitudinal growth charts are critical for diagnosis. Percentile BMIs must be used, given the inaccuracy of standard BMI calculations in this age group. (Centers for Disease Control and Prevention age and sex growth charts include methods for this calculation). Laboratory assessment, including metabolic, kidney, pancreatic, and thyroid function, and an EKG can illuminate if there are consequences of restricting or purging. Of course, you want to evaluate for significant medical symptoms, including bradycardia, orthostasis, and hypokalemia. These medical symptoms are not limited to the severely underweight and merit referral to an emergency department and possible medical admission.
Then, a referral to a clinician who is expert in the assessment and treatment of eating disorders is needed. This may be a child psychiatrist, psychologist, or a colleague pediatrician with this specialization. It is also very important to begin the conversation with the family to introduce your concerns, describe what you have noticed, and discuss the need for further assessment and possibly treatment.
Be mindful that discussing this in front of your patient may heighten the patient’s anxiety or distress. Be prepared to offer support and understanding for your patient’s anxiety, while steadfastly providing absolute clarity for the parents about the necessity of further evaluation and treatment. Many parents will be concerned and ready to do whatever is needed to get their child’s eating and growth back on track. But some parents may have more difficulty. They may have their own history with an eating disorder. They may be avoiding a sense of shame or alarm. They may be eager to avoid adding to their child’s stress. They may be tired of engaging in power struggles with the child. They may be proud of their ambitious, accomplished young athlete. Their trust in you makes you uniquely positioned to complicate their thinking. And treatment will hinge on them, so this is a critical bridge to care.
Beyond telling parents that they will need to bring more structure and supervision to mealtimes to begin addressing their child’s nutrition, you might offer guidance on other strategies. Empower parents to limit their child’s use of social media sites such as Instagram, YouTube, and TikTok, where they may be immersed in comparing themselves to idealized (and airbrushed) influencers. Empower them to make their child’s participation in beloved sports contingent on eating meals together and completely or on a stabilized weight (as will be common in treatment). Remind them that there are no bad foods, that the goal is health, and that they are not in a power struggle with their child, but instead allied with their child to treat AN. Remind them to also look for chances to have fun with their child, to help everyone remember what matters.
Treatment
Family-based therapy (FBT) is the first-line treatment of shorter-duration AN in children and adolescents. It focuses on the parents, helping them to calmly and effectively manage their child’s eating behaviors until their weight and behaviors have normalized. As a patient’s nutritional status improves, so does cognitive function, emotional flexibility, and mood. Individual therapy and psychopharmacologic treatment can be very effective for comorbid anxiety, mood, attentional, and thought disorders. Family-based work does include the child and is often done in group-based settings with clinicians from multiple disciplines. Dietitians provide education and guidance about healthy nutrition to the child and parents. Therapists may work with the child, parents, or full family to focus on behavior modification and managing distress. Most academic medical centers provide access to FBT, but there are many regions with no providers of this evidence-based treatment. One of the silver linings of the COVID-19 pandemic is that several online services have emerged offering FBT, working with families to manage mealtimes and treatment entirely at home.3 Pediatricians provide regular medical checks to measure progress and help with decisions about when it is safe to permit exercise or advance privileges and independence around eating. Some pediatricians have discovered a deep interest in this area of pediatrics and built their practices on it. Given the surge in prevalence of AN and the needs for adolescent mental health services, we hope more will do so.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected].
References
1. Taquet M et al. Br J Psychiatry. 2022;220:262-4.
2. Morgan JF et al. West J Med. 2000 Mar;172(3):164-5.
3. Matheson BE et al. Int J Eat Disord. 2020 Jul;53(7):1142-54.
Eating disorders are among the most prevalent, disabling, and potentially fatal psychiatric illnesses, and the COVID-19 pandemic has exacerbated their burden, with a 15.3% increase in incidence in 2020 compared with previous years.1 This increase was almost solely among adolescent girls with anorexia nervosa (AN), which is often insidious in onset and more difficult to treat as it advances. Adolescents with AN are most likely to present to their pediatricians, so awareness and early recognition of the symptoms is critical. Pediatricians are also an integral part of the treatment team in AN and can offer monitoring for serious complications, alongside valuable guidance to parents, who are central to treatment and the reestablishment of healthy eating habits in their children. Here we will review the epidemiology, diagnosis, and treatment of anorexia, with an emphasis on what pediatricians need to know to screen and to facilitate treatment.
Epidemiology
AN is marked by a fear of gaining weight or behaviors that interfere with weight gain and a self-evaluation unduly influenced by weight and body shape. Youth with AN often deny the seriousness of their malnutrition, although that is not required for diagnosis. AN can be of a restrictive or binge-purge subtype, and amenorrhea is no longer a requirement for diagnosis. There is not a specific weight or body mass index cutoff for the diagnosis, but the severity of AN is determined by the BMI percentile normed to age and sex. The average age of onset is 18, and the prepandemic prevalence of AN was about 1% of the population. It affects about 10 times as many females as males. It is quite rare prior to puberty, affecting about 0.01% of that age group. There is a heritable component, with a fivefold relative risk in youth with a parent with AN, and twin studies suggest heritability rates as high as 75%. Youth with rigid cognitive styles appear more vulnerable, as do those who participate in activities such as ballet, gymnastics, modeling, and wrestling because of the role of appearance and weight in performance. More than half of patients with AN will have another psychiatric illness, most commonly anxiety disorders, depression, or obsessive-compulsive disorder. AN becomes chronic in up to 15% of sufferers and the mortality rate is close to 10%, with approximately half dying from medical complications and half dying by suicide.
Screening
Parents and pediatricians are usually the first to notice that a child has started to lose weight or is falling off the growth curve. But weight changes usually emerge after feelings of preoccupation with weight, body shape, and body satisfaction. If parents report escalating pickiness around food, increased or compulsive exercise, persistent self-consciousness and self-criticism around weight and body shape, it is worth starting with screening questions.
If you notice preoccupation or anxiety around being weighed, even if the weight or growth curve are still normal, it is worthwhile to screen. Screening questions, such as the SCOFF questionnaire with five simple questions, can be very sensitive for both AN and bulimia nervosa.2 There are also many validated screening instruments, such as the Eating Disorder Inventory or Eating Attitudes Test (for adolescents) and the Kids Eating Disorder Survey and the Child Eating Attitudes Test (for younger children), that are short self-reports that you can have your patients fill out when you have a higher index of suspicion. Weight loss or growth failure without a preoccupation around weight or appearance needs a thorough a medical workup, and could be a function of other psychiatric problems, such as depression.
If a child screens positive for an eating disorder, your full physical examination, growth curves, and longitudinal growth charts are critical for diagnosis. Percentile BMIs must be used, given the inaccuracy of standard BMI calculations in this age group. (Centers for Disease Control and Prevention age and sex growth charts include methods for this calculation). Laboratory assessment, including metabolic, kidney, pancreatic, and thyroid function, and an EKG can illuminate if there are consequences of restricting or purging. Of course, you want to evaluate for significant medical symptoms, including bradycardia, orthostasis, and hypokalemia. These medical symptoms are not limited to the severely underweight and merit referral to an emergency department and possible medical admission.
Then, a referral to a clinician who is expert in the assessment and treatment of eating disorders is needed. This may be a child psychiatrist, psychologist, or a colleague pediatrician with this specialization. It is also very important to begin the conversation with the family to introduce your concerns, describe what you have noticed, and discuss the need for further assessment and possibly treatment.
Be mindful that discussing this in front of your patient may heighten the patient’s anxiety or distress. Be prepared to offer support and understanding for your patient’s anxiety, while steadfastly providing absolute clarity for the parents about the necessity of further evaluation and treatment. Many parents will be concerned and ready to do whatever is needed to get their child’s eating and growth back on track. But some parents may have more difficulty. They may have their own history with an eating disorder. They may be avoiding a sense of shame or alarm. They may be eager to avoid adding to their child’s stress. They may be tired of engaging in power struggles with the child. They may be proud of their ambitious, accomplished young athlete. Their trust in you makes you uniquely positioned to complicate their thinking. And treatment will hinge on them, so this is a critical bridge to care.
Beyond telling parents that they will need to bring more structure and supervision to mealtimes to begin addressing their child’s nutrition, you might offer guidance on other strategies. Empower parents to limit their child’s use of social media sites such as Instagram, YouTube, and TikTok, where they may be immersed in comparing themselves to idealized (and airbrushed) influencers. Empower them to make their child’s participation in beloved sports contingent on eating meals together and completely or on a stabilized weight (as will be common in treatment). Remind them that there are no bad foods, that the goal is health, and that they are not in a power struggle with their child, but instead allied with their child to treat AN. Remind them to also look for chances to have fun with their child, to help everyone remember what matters.
Treatment
Family-based therapy (FBT) is the first-line treatment of shorter-duration AN in children and adolescents. It focuses on the parents, helping them to calmly and effectively manage their child’s eating behaviors until their weight and behaviors have normalized. As a patient’s nutritional status improves, so does cognitive function, emotional flexibility, and mood. Individual therapy and psychopharmacologic treatment can be very effective for comorbid anxiety, mood, attentional, and thought disorders. Family-based work does include the child and is often done in group-based settings with clinicians from multiple disciplines. Dietitians provide education and guidance about healthy nutrition to the child and parents. Therapists may work with the child, parents, or full family to focus on behavior modification and managing distress. Most academic medical centers provide access to FBT, but there are many regions with no providers of this evidence-based treatment. One of the silver linings of the COVID-19 pandemic is that several online services have emerged offering FBT, working with families to manage mealtimes and treatment entirely at home.3 Pediatricians provide regular medical checks to measure progress and help with decisions about when it is safe to permit exercise or advance privileges and independence around eating. Some pediatricians have discovered a deep interest in this area of pediatrics and built their practices on it. Given the surge in prevalence of AN and the needs for adolescent mental health services, we hope more will do so.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected].
References
1. Taquet M et al. Br J Psychiatry. 2022;220:262-4.
2. Morgan JF et al. West J Med. 2000 Mar;172(3):164-5.
3. Matheson BE et al. Int J Eat Disord. 2020 Jul;53(7):1142-54.
When coping skills and parenting behavioral interventions ‘don’t work’
You have an appointment with a 14-year-old youth you last saw for an annual camp physical. He had screened positive for depression, and you had referred him to a local therapist. He did not have an appointment until after camp, and you have only met a few times, but since you had spoken with him about his depression, he set up an appointment with you to ask about medications. When you meet him you ask about what he had been doing in therapy and he says, “I’m learning ‘coping skills,’ but they don’t work.”
From breathing exercises and sticker charts to mindfulness and grounding exercise, coping skills can be crucial for learning how to manage distress, regulate emotions, become more effective interpersonally, and function better. Similarly, parenting interventions, which change the way parents and youth interact, are a central family intervention for behavioral problems in youth.
It is very common, however, to hear that they “don’t work” or have a parent say, “We tried that, it doesn’t work.”
When kids and parents reject coping skills and behavioral interventions by saying they do not work, the consequences can be substantial. It can mean the rejection of coping skills and strategies that actually would have helped, given time and support; that kids and families bounce between services with increasing frustration; that they search for a magic bullet (which also won’t work); and, particularly concerning for physicians, a belief that the youth have not received the right medication, resulting in potentially unhelpful concoctions of medication.
One of the biggest challenges in helping youth and parents overcome their difficulties – whether these difficulties are depression and anxiety or being better parents to struggling kids – is helping them understand that despite the fact that coping skills and behavioral interventions do not seem to work, they work.
We just have to do a better job explaining what that “work” is.
There are five points you can make.
- First, the coping skill or behavioral intervention is not supposed to work if that means solving the underlying problem. Coping skills and behavioral interventions do not immediately cure anxiety, mend broken hearts, correct disruptive behaviors, disentangle power struggles, or alleviate depression. That is not what their job is. Coping skills and behavioral interventions are there to help us get better at handling complex situations and feelings. In particular, they are good at helping us manage our thoughts (“I can’t do it,” “He should behave better”) and our affect (anger, frustration, rage, anxiety, sadness), so that over time we get better at solving the problems, and break out of the patterns that perpetuate these problems.
- Second, kids and parents do not give skills credit for when they do work. That time you were spiraling out of control and told your mom you needed a break and watched some YouTube videos and then joined the family for dinner? Your coping skills worked, but nobody noticed because they worked. We need to help our young patients and families identify those times that coping skills and behavioral interventions worked.
- Third, let’s face it: Nothing works all the time. It is no wonder kids and families are disappointed by coping skills and behavioral interventions if they think they magically work once and forever. We need to manage expectations.
- Fourth, we know they are supposed to fail, and we should discuss this openly up front. This may sound surprising, but challenging behaviors often get worse when we begin to work on them. “Extinction bursts” is probably the easiest explanation, but for psychodynamically oriented youth and families we could talk about “resistance.” No matter what, things tend to get worse before they get better. We should let people know this ahead of time.
- Fifth, and this is the one that forces youth and parents to ask how hard they actually tried, these skills need to be practiced. You can’t be in the middle of a panic attack and for the first time start trying to pace your breathing with a technique a therapist told you about 3 weeks ago. This makes about as much sense as not training for a marathon. You need to practice and build up the skills, recognizing that as you become more familiar with them, they will help you manage during stressful situations. Every skill should be practiced, preferably several times or more in sessions, maybe every session, and definitely outside of sessions when not in distress.
We cannot blame children and parents for thinking that coping skills and behavioral interventions do not work. They are struggling, suffering, fighting, frightened, angry, anxious, frustrated, and often desperate for something to make everything better. Helping them recognize this desire for things to be better while managing expectations is an essential complement to supporting the use of coping skills and behavioral interventions, and a fairly easy conversation to have with youth.
So when you are talking about coping skills and parental behavioral interventions, it is important to be prepared for the “it didn’t work” conversation, and even to address these issues up front. After all, these strategies may not solve all the problems in the world, but can be lifelong ways of coping with life’s challenges.
Dr. Henderson is associate professor of clinical psychiatry at New York University and deputy director of child and adolescent psychiatry at Bellevue Hospital, New York.
You have an appointment with a 14-year-old youth you last saw for an annual camp physical. He had screened positive for depression, and you had referred him to a local therapist. He did not have an appointment until after camp, and you have only met a few times, but since you had spoken with him about his depression, he set up an appointment with you to ask about medications. When you meet him you ask about what he had been doing in therapy and he says, “I’m learning ‘coping skills,’ but they don’t work.”
From breathing exercises and sticker charts to mindfulness and grounding exercise, coping skills can be crucial for learning how to manage distress, regulate emotions, become more effective interpersonally, and function better. Similarly, parenting interventions, which change the way parents and youth interact, are a central family intervention for behavioral problems in youth.
It is very common, however, to hear that they “don’t work” or have a parent say, “We tried that, it doesn’t work.”
When kids and parents reject coping skills and behavioral interventions by saying they do not work, the consequences can be substantial. It can mean the rejection of coping skills and strategies that actually would have helped, given time and support; that kids and families bounce between services with increasing frustration; that they search for a magic bullet (which also won’t work); and, particularly concerning for physicians, a belief that the youth have not received the right medication, resulting in potentially unhelpful concoctions of medication.
One of the biggest challenges in helping youth and parents overcome their difficulties – whether these difficulties are depression and anxiety or being better parents to struggling kids – is helping them understand that despite the fact that coping skills and behavioral interventions do not seem to work, they work.
We just have to do a better job explaining what that “work” is.
There are five points you can make.
- First, the coping skill or behavioral intervention is not supposed to work if that means solving the underlying problem. Coping skills and behavioral interventions do not immediately cure anxiety, mend broken hearts, correct disruptive behaviors, disentangle power struggles, or alleviate depression. That is not what their job is. Coping skills and behavioral interventions are there to help us get better at handling complex situations and feelings. In particular, they are good at helping us manage our thoughts (“I can’t do it,” “He should behave better”) and our affect (anger, frustration, rage, anxiety, sadness), so that over time we get better at solving the problems, and break out of the patterns that perpetuate these problems.
- Second, kids and parents do not give skills credit for when they do work. That time you were spiraling out of control and told your mom you needed a break and watched some YouTube videos and then joined the family for dinner? Your coping skills worked, but nobody noticed because they worked. We need to help our young patients and families identify those times that coping skills and behavioral interventions worked.
- Third, let’s face it: Nothing works all the time. It is no wonder kids and families are disappointed by coping skills and behavioral interventions if they think they magically work once and forever. We need to manage expectations.
- Fourth, we know they are supposed to fail, and we should discuss this openly up front. This may sound surprising, but challenging behaviors often get worse when we begin to work on them. “Extinction bursts” is probably the easiest explanation, but for psychodynamically oriented youth and families we could talk about “resistance.” No matter what, things tend to get worse before they get better. We should let people know this ahead of time.
- Fifth, and this is the one that forces youth and parents to ask how hard they actually tried, these skills need to be practiced. You can’t be in the middle of a panic attack and for the first time start trying to pace your breathing with a technique a therapist told you about 3 weeks ago. This makes about as much sense as not training for a marathon. You need to practice and build up the skills, recognizing that as you become more familiar with them, they will help you manage during stressful situations. Every skill should be practiced, preferably several times or more in sessions, maybe every session, and definitely outside of sessions when not in distress.
We cannot blame children and parents for thinking that coping skills and behavioral interventions do not work. They are struggling, suffering, fighting, frightened, angry, anxious, frustrated, and often desperate for something to make everything better. Helping them recognize this desire for things to be better while managing expectations is an essential complement to supporting the use of coping skills and behavioral interventions, and a fairly easy conversation to have with youth.
So when you are talking about coping skills and parental behavioral interventions, it is important to be prepared for the “it didn’t work” conversation, and even to address these issues up front. After all, these strategies may not solve all the problems in the world, but can be lifelong ways of coping with life’s challenges.
Dr. Henderson is associate professor of clinical psychiatry at New York University and deputy director of child and adolescent psychiatry at Bellevue Hospital, New York.
You have an appointment with a 14-year-old youth you last saw for an annual camp physical. He had screened positive for depression, and you had referred him to a local therapist. He did not have an appointment until after camp, and you have only met a few times, but since you had spoken with him about his depression, he set up an appointment with you to ask about medications. When you meet him you ask about what he had been doing in therapy and he says, “I’m learning ‘coping skills,’ but they don’t work.”
From breathing exercises and sticker charts to mindfulness and grounding exercise, coping skills can be crucial for learning how to manage distress, regulate emotions, become more effective interpersonally, and function better. Similarly, parenting interventions, which change the way parents and youth interact, are a central family intervention for behavioral problems in youth.
It is very common, however, to hear that they “don’t work” or have a parent say, “We tried that, it doesn’t work.”
When kids and parents reject coping skills and behavioral interventions by saying they do not work, the consequences can be substantial. It can mean the rejection of coping skills and strategies that actually would have helped, given time and support; that kids and families bounce between services with increasing frustration; that they search for a magic bullet (which also won’t work); and, particularly concerning for physicians, a belief that the youth have not received the right medication, resulting in potentially unhelpful concoctions of medication.
One of the biggest challenges in helping youth and parents overcome their difficulties – whether these difficulties are depression and anxiety or being better parents to struggling kids – is helping them understand that despite the fact that coping skills and behavioral interventions do not seem to work, they work.
We just have to do a better job explaining what that “work” is.
There are five points you can make.
- First, the coping skill or behavioral intervention is not supposed to work if that means solving the underlying problem. Coping skills and behavioral interventions do not immediately cure anxiety, mend broken hearts, correct disruptive behaviors, disentangle power struggles, or alleviate depression. That is not what their job is. Coping skills and behavioral interventions are there to help us get better at handling complex situations and feelings. In particular, they are good at helping us manage our thoughts (“I can’t do it,” “He should behave better”) and our affect (anger, frustration, rage, anxiety, sadness), so that over time we get better at solving the problems, and break out of the patterns that perpetuate these problems.
- Second, kids and parents do not give skills credit for when they do work. That time you were spiraling out of control and told your mom you needed a break and watched some YouTube videos and then joined the family for dinner? Your coping skills worked, but nobody noticed because they worked. We need to help our young patients and families identify those times that coping skills and behavioral interventions worked.
- Third, let’s face it: Nothing works all the time. It is no wonder kids and families are disappointed by coping skills and behavioral interventions if they think they magically work once and forever. We need to manage expectations.
- Fourth, we know they are supposed to fail, and we should discuss this openly up front. This may sound surprising, but challenging behaviors often get worse when we begin to work on them. “Extinction bursts” is probably the easiest explanation, but for psychodynamically oriented youth and families we could talk about “resistance.” No matter what, things tend to get worse before they get better. We should let people know this ahead of time.
- Fifth, and this is the one that forces youth and parents to ask how hard they actually tried, these skills need to be practiced. You can’t be in the middle of a panic attack and for the first time start trying to pace your breathing with a technique a therapist told you about 3 weeks ago. This makes about as much sense as not training for a marathon. You need to practice and build up the skills, recognizing that as you become more familiar with them, they will help you manage during stressful situations. Every skill should be practiced, preferably several times or more in sessions, maybe every session, and definitely outside of sessions when not in distress.
We cannot blame children and parents for thinking that coping skills and behavioral interventions do not work. They are struggling, suffering, fighting, frightened, angry, anxious, frustrated, and often desperate for something to make everything better. Helping them recognize this desire for things to be better while managing expectations is an essential complement to supporting the use of coping skills and behavioral interventions, and a fairly easy conversation to have with youth.
So when you are talking about coping skills and parental behavioral interventions, it is important to be prepared for the “it didn’t work” conversation, and even to address these issues up front. After all, these strategies may not solve all the problems in the world, but can be lifelong ways of coping with life’s challenges.
Dr. Henderson is associate professor of clinical psychiatry at New York University and deputy director of child and adolescent psychiatry at Bellevue Hospital, New York.
Tactile stimulation for inadequate neonatal respiration at birth
Recently, I encountered a study in Pediatrics that hoped to answer the question of whether there was any benefit to tactile stimulation in those nerve-rattling moments when a newborn didn’t seem to take much interest in breathing: “Tactile stimulation in newborn infants with inadequate respiration at birth: A systematic review.” Now there is a title that grabs the attention of every frontline pediatrician who has sweated through those minutes that seemed like hours in the delivery room when some little rascal has decided that breathing isn’t a priority.
Of course, your great grandmother and everyone else knew what needed to be done – the obstetrician hung the baby by his or her ankles and slapped it on the bottom a couple of times. But you went to medical school and learned that was barbaric. Instead, you modeled the behavior of the residents and delivery room nurses who had more refined techniques such as heel flicking and vigorous spine rubbing. You never thought to ask if there was any science behind those activities because everyone did them.
Well, the authors of the article in Pediatrics, writing on behalf of the International Liaison Committee on Resuscitation and Neonatal Life Support Task Force, thought the time had come to turn over a few stones and see if tactile stimulation was a benefit in resuscitation. Beginning with 2,455 possibly relevant articles, they quickly (I suspect they would quibble with the “quickly” part) winnowed these down to two observational studies, one of which was rejected because of “critical risk of bias.” The surviving study showed a reduction in tracheal intubation in infants who had received tactile stimulation. However, the authors felt that the “certainty of evidence was very low.”
So, there you have it. Aren’t you glad you didn’t invest 15 or 20 minutes discovering what you probably had guessed already? You can thank me later.
You already suspected that it may not help. However, like any good physician, what you really wanted to know is whether were you doing any harm by heel flicking and spine rubbing. And I bet you already had an opinion about the answer to that question. During your training, you may have seen delivery room personnel who were clearly too vigorous in their tactile stimulation and/or too persistent in their heel flicking and spine rubbing when the next steps in resuscitation needed to be taken. That’s the next study that needs to be done. I hope that study finds that tactile stimulation may not help but as long as it is done using specific techniques and within certain temporal parameters it does no harm.
I was never much for heel flicking. My favorite tactile stimulation was encircling the pokey infant’s chest in my hand, gently compressing and then quickly releasing a couple of times. My hope was that by mimicking the birth process the sensors in the infant’s chest wall would remind him it was time to breathe. That, and a silent plea to Mother Nature, worked most of the time.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Recently, I encountered a study in Pediatrics that hoped to answer the question of whether there was any benefit to tactile stimulation in those nerve-rattling moments when a newborn didn’t seem to take much interest in breathing: “Tactile stimulation in newborn infants with inadequate respiration at birth: A systematic review.” Now there is a title that grabs the attention of every frontline pediatrician who has sweated through those minutes that seemed like hours in the delivery room when some little rascal has decided that breathing isn’t a priority.
Of course, your great grandmother and everyone else knew what needed to be done – the obstetrician hung the baby by his or her ankles and slapped it on the bottom a couple of times. But you went to medical school and learned that was barbaric. Instead, you modeled the behavior of the residents and delivery room nurses who had more refined techniques such as heel flicking and vigorous spine rubbing. You never thought to ask if there was any science behind those activities because everyone did them.
Well, the authors of the article in Pediatrics, writing on behalf of the International Liaison Committee on Resuscitation and Neonatal Life Support Task Force, thought the time had come to turn over a few stones and see if tactile stimulation was a benefit in resuscitation. Beginning with 2,455 possibly relevant articles, they quickly (I suspect they would quibble with the “quickly” part) winnowed these down to two observational studies, one of which was rejected because of “critical risk of bias.” The surviving study showed a reduction in tracheal intubation in infants who had received tactile stimulation. However, the authors felt that the “certainty of evidence was very low.”
So, there you have it. Aren’t you glad you didn’t invest 15 or 20 minutes discovering what you probably had guessed already? You can thank me later.
You already suspected that it may not help. However, like any good physician, what you really wanted to know is whether were you doing any harm by heel flicking and spine rubbing. And I bet you already had an opinion about the answer to that question. During your training, you may have seen delivery room personnel who were clearly too vigorous in their tactile stimulation and/or too persistent in their heel flicking and spine rubbing when the next steps in resuscitation needed to be taken. That’s the next study that needs to be done. I hope that study finds that tactile stimulation may not help but as long as it is done using specific techniques and within certain temporal parameters it does no harm.
I was never much for heel flicking. My favorite tactile stimulation was encircling the pokey infant’s chest in my hand, gently compressing and then quickly releasing a couple of times. My hope was that by mimicking the birth process the sensors in the infant’s chest wall would remind him it was time to breathe. That, and a silent plea to Mother Nature, worked most of the time.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Recently, I encountered a study in Pediatrics that hoped to answer the question of whether there was any benefit to tactile stimulation in those nerve-rattling moments when a newborn didn’t seem to take much interest in breathing: “Tactile stimulation in newborn infants with inadequate respiration at birth: A systematic review.” Now there is a title that grabs the attention of every frontline pediatrician who has sweated through those minutes that seemed like hours in the delivery room when some little rascal has decided that breathing isn’t a priority.
Of course, your great grandmother and everyone else knew what needed to be done – the obstetrician hung the baby by his or her ankles and slapped it on the bottom a couple of times. But you went to medical school and learned that was barbaric. Instead, you modeled the behavior of the residents and delivery room nurses who had more refined techniques such as heel flicking and vigorous spine rubbing. You never thought to ask if there was any science behind those activities because everyone did them.
Well, the authors of the article in Pediatrics, writing on behalf of the International Liaison Committee on Resuscitation and Neonatal Life Support Task Force, thought the time had come to turn over a few stones and see if tactile stimulation was a benefit in resuscitation. Beginning with 2,455 possibly relevant articles, they quickly (I suspect they would quibble with the “quickly” part) winnowed these down to two observational studies, one of which was rejected because of “critical risk of bias.” The surviving study showed a reduction in tracheal intubation in infants who had received tactile stimulation. However, the authors felt that the “certainty of evidence was very low.”
So, there you have it. Aren’t you glad you didn’t invest 15 or 20 minutes discovering what you probably had guessed already? You can thank me later.
You already suspected that it may not help. However, like any good physician, what you really wanted to know is whether were you doing any harm by heel flicking and spine rubbing. And I bet you already had an opinion about the answer to that question. During your training, you may have seen delivery room personnel who were clearly too vigorous in their tactile stimulation and/or too persistent in their heel flicking and spine rubbing when the next steps in resuscitation needed to be taken. That’s the next study that needs to be done. I hope that study finds that tactile stimulation may not help but as long as it is done using specific techniques and within certain temporal parameters it does no harm.
I was never much for heel flicking. My favorite tactile stimulation was encircling the pokey infant’s chest in my hand, gently compressing and then quickly releasing a couple of times. My hope was that by mimicking the birth process the sensors in the infant’s chest wall would remind him it was time to breathe. That, and a silent plea to Mother Nature, worked most of the time.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
USPSTF recommendation roundup
In 2021, the US Preventive Services Task Force (USPSTF) considered 13 topics and made a total of 23 recommendations. They reviewed only 1 new topic. The other 12 were updates of topics previously addressed; no changes were made in 9 of them. In 3, the recommended age of screening or the criteria for screening were expanded. This Practice Alert will review the recommendations made and highlight new recommendations and any changes to previous ones. All complete recommendation statements, rationales, clinical considerations, and evidence reports can be found on the USPSTF website at https://uspreventiveservicestaskforce.org/uspstf/home.1
Dental caries in children
Dental caries affect about 23% of children between the ages of 2 and 5 years and are associated with multiple adverse social outcomes and medical conditions.2 The best way to prevent tooth decay, other than regular brushing with fluoride toothpaste, is to drink water with recommended amounts of fluoride (≥ 0.6 parts fluoride per million parts water).2 The USPSTF reaffirmed its recommendation from 2014 that stated when a local water supply lacks sufficient fluoride, primary care clinicians should prescribe oral supplementation for infants and children in the form of fluoride drops starting at age 6 months. The dosage of fluoride depends on patient age and fluoride concentration in the local water (TABLE 13). The USPSTF also recommends applying topical fluoride as 5% sodium fluoride varnish, every 6 months, starting when the primary teeth erupt.2
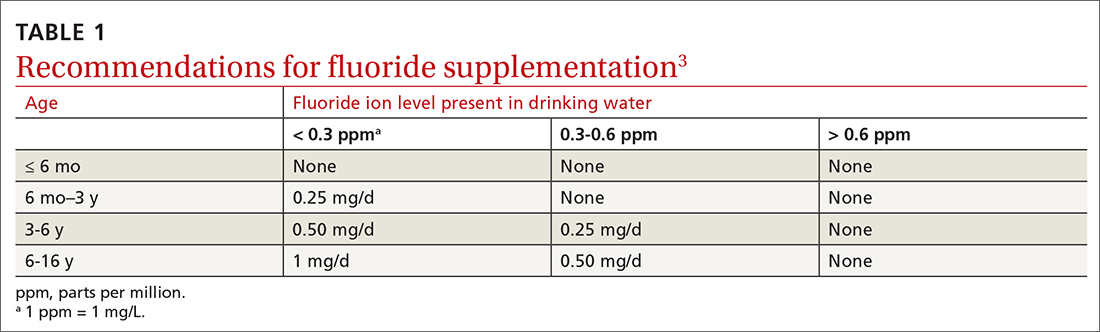
In addition to fluoride supplements and topical varnish, should clinicians perform screening examinations looking for dental caries? The USPSTF feels there is not enough evidence to assess this practice and gives it an “I” rating (insufficient evidence).
Preventive interventions in pregnancy
In 2021, the USPSTF assessed 3 topics related to pregnancy and prenatal care.
Screening for gestational diabetes. The USPSTF gave a “B” recommendation for screening at 24 weeks of pregnancy or after, but an “I” statement for screening prior to 24 weeks.4 Screening can involve a 1-step or 2-step protocol.
The 2-step protocol is most commonly used in the United States. It involves first measuring serum glucose after a nonfasting 50-g oral glucose challenge; if the resulting level is high, the second step is a 75- or 100-g oral glucose tolerance test lasting 3 hours. The 1-step protocol involves measuring a fasting glucose level, followed by a 75-g oral glucose challenge with glucose levels measured at 1 and 2 hours.
Healthy weight gain in pregnancy. This was the only new topic the USPSTF assessed last year. The resulting recommendation is to offer pregnant women behavioral counseling to promote healthy weight gain and to prevent excessive weight gain in pregnancy. The recommended weight gain depends on the mother’s prepregnancy weight status: 28 to 40 lbs if the mother is underweight; 25 to 35 lbs if she is not under- or overweight; 15 to 25 lbs if she is overweight; and 11 to 20 lbs if she is obese.5 Healthy weight gain contributes to preventing gestational diabetes, emergency cesarean sections, and infant macrosomia.
Continue to: Low-dose aspirin
Low-dose aspirin. Reaffirming a recommendation from 2014, the USPSTF advises low-dose aspirin (81 mg/d) starting after 12 weeks’ gestation for all pregnant women who are at high risk for preeclampsia. TABLE 26 lists high- and moderate-risk conditions for preeclampsia and the recommendation for the use of low-dose aspirin.
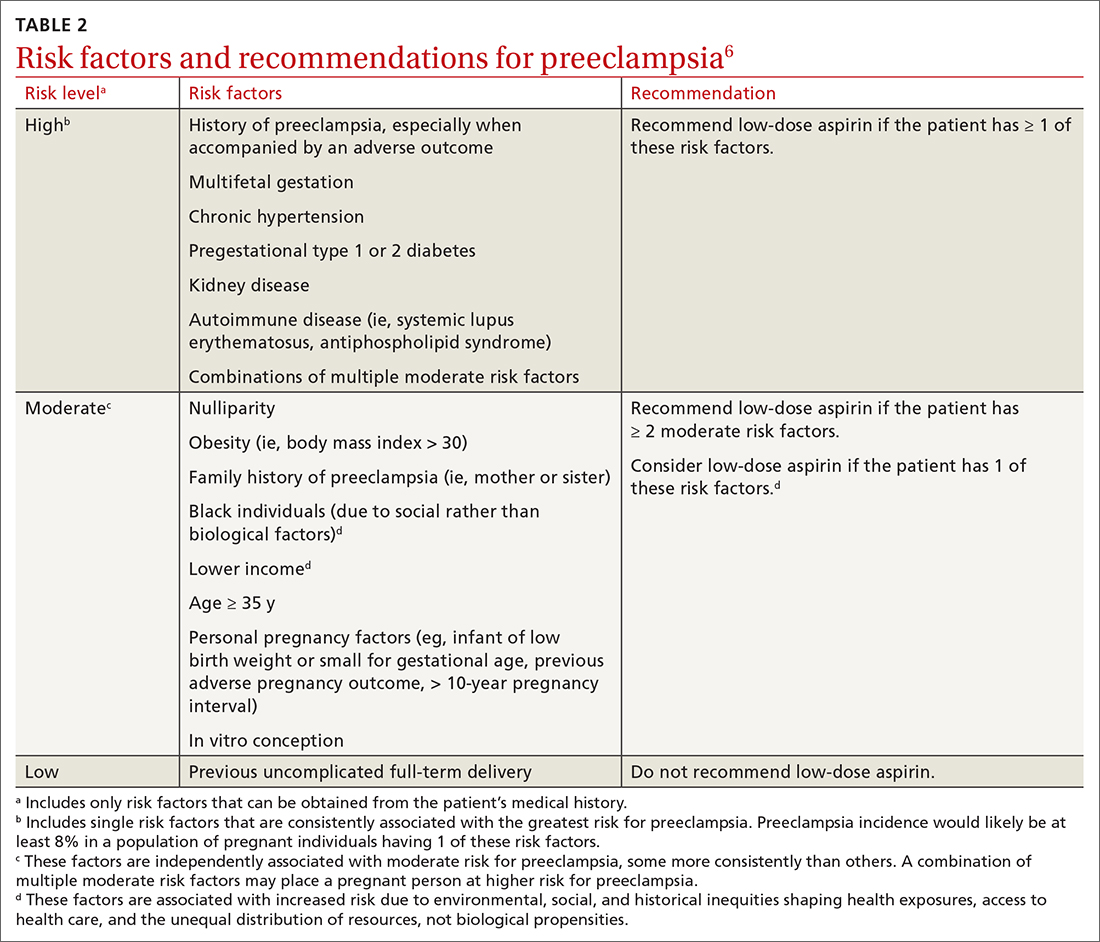
Sexually transmitted infections
Screening for both chlamydia and gonorrhea in sexually active females through age 24 years was given a “B” recommendation, reaffirming the 2014 recommendation.7 Screening for these 2 sexually transmitted infections (STIs) is also recommended for women 25 years and older who are at increased risk of STIs. Risk is defined as having a new sex partner, more than 1 sex partner, a sex partner who has other sex partners, or a sex partner who has an STI; not using condoms consistently; having a previous STI; exchanging sex for money or drugs; or having a history of incarceration.
Screen for both infections simultaneously using a nucleic acid amplification test, testing all sites of sexual exposure. Urine testing can replace cervical, vaginal, and urethral testing. Those found to be positive for either STI should be treated according to the most recent treatment guidelines from the Centers for Disease Control and Prevention (CDC). And sexual partners should be advised to undergo testing.8,9
The USPSTF could not find evidence for the benefits and harms of screening for STIs in men. Remember that screening applies to those who are asymptomatic. Male sex partners of those found to be infected should be tested, as should those who show any signs or symptoms of an STI. A recent Practice Alert described the most current CDC guidance for diagnosing and treating STIs.9
Type 2 diabetes and prediabetes
Screening for type 2 diabetes (T2D) and prediabetes is now recommended for adults ages 35 to 70 years who are overweight or obese.10 The age to start screening has been lowered to 35 years from the previous recommendation in 2015, which recommended starting at age 40. In addition, the recommendation states that patients with prediabetes should be referred for preventive interventions. It is important that referral is included in the statement because the Affordable Care Act mandates that USPSTF “A” and “B” recommendations must be covered by commercial health insurance with no copay or deductible.
Continue to: Screening can be conducted...
Screening can be conducted using a fasting plasma glucose or A1C level, or with an oral glucose tolerance test. Interventions that can prevent or delay the onset of T2D in those with prediabetes include lifestyle interventions that focus on diet and physical activity, and the use of metformin (although metformin has not been approved for this by the US Food and Drug Administration).
Changes to cancer screening recommendations
In 2021, the USPSTF reviewed and modified its recommendations on screening for 2 types of cancer: colorectal and lung.
For colorectal cancer, the age at which to start screening was lowered from 50 years to 45 years.11 Screening at this earlier age is a “B” recommendation, because, while there is benefit from screening, it is less than for older age groups. Screening individuals ages 50 to 75 years remains an “A” recommendation, and for those ages 76 to 85 years it remains a “C” recommendation. A “C” recommendation means that the overall benefits are small but some individuals might benefit based on their overall health and prior screening results. In its clinical considerations, the USPSTF recommends against screening in those ages 85 and older but, curiously, does not list it as a “D” recommendation. The screening methods and recommended screening intervals for each appear in TABLE 3.11
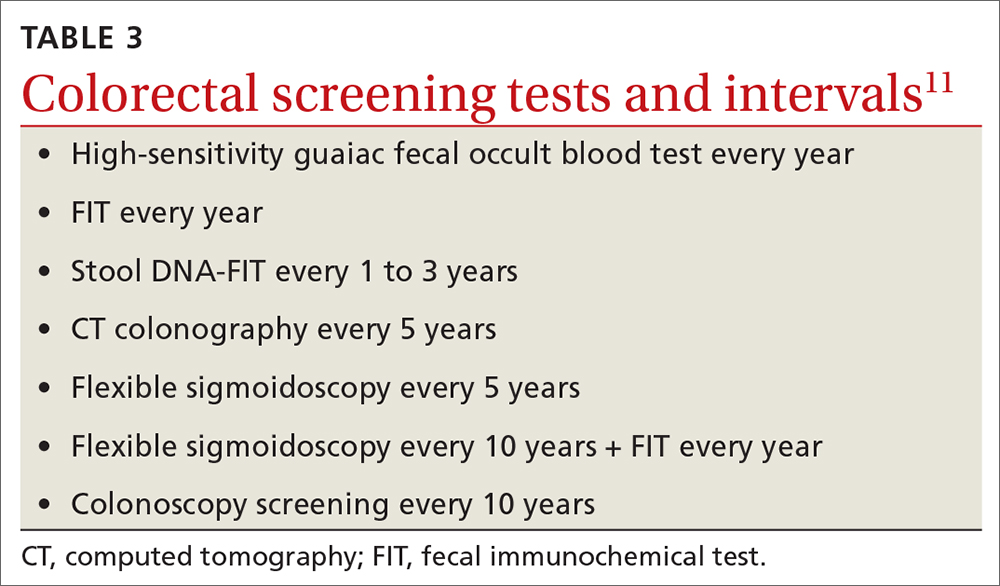
For lung cancer, annual screening using low-dose computed tomography (CT) was first recommended by the USPSTF in 2013 for adults ages 55 to 80 years with a 30-pack-year smoking history. Screening could stop once 15 years had passed since smoking cessation. In 2021, the USPSTF lowered the age to initiate screening to 50 years, and the smoking history threshold to 20 pack-years.12 If these recommendations are followed, a current smoker who does not quit smoking could possibly receive 30 annual CT scans. The recommendation does state that screening should stop once a person develops a health condition that significantly affects life expectancy or ability to have lung surgery.
For primary prevention of lung cancer and other chronic diseases through smoking cessation, the USPSTF also reassessed its 2015 recommendations. It reaffirmed the “A” recommendation to ask adults about tobacco use and, for tobacco users, to recommend cessation and provide behavioral therapy and approved pharmacotherapy.13 The recommendation differed for pregnant adults in that the USPSTF is unsure about the potential harms of pharmacotherapy in pregnancy and gives that an “I” statement.13 An additional “I” statement was made about the use of electronic cigarettes for smoking cessation; the USPSTF recommends using behavioral and pharmacotherapy interventions with proven effectiveness and safety instead.
Continue to: 4 additional recommendation updates with no changes
4 additional recommendation updates with no changes
Screening for high blood pressure in adults ages 18 years and older continues to receive an “A” recommendation.14 Importantly, the recommendation states that confirmation of high blood pressure should be made in an out-of-office setting before initiating treatment. Screening for vitamin D deficiency in adults and hearing loss in older adults both continue with “I” statements,15,16 and screening for asymptomatic carotid artery stenosis continues to receive a “D” recommendation.17 The implications of the vitamin D “I” statement were discussed in a previous Practice Alert.18
Continuing value of the USPSTF
The USPSTF continues to set the gold standard for assessment of preventive interventions, and its decisions affect first-dollar coverage by commercial health insurance. The reaffirmation of past recommendations demonstrates the value of adhering to rigorous evidence-based methods (if they are done correctly, they rarely must be markedly changed). And the updating of screening criteria shows the need to constantly review the evolving evidence for current recommendations. Once again, however, funding and staffing limitations allowed the USPSTF to assess only 1 new topic. A listing of all the 2021 recommendations is in TABLE 4.1


1. USPSTF. Recommendation topics. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation-topics
2. USPSTF. Prevention of dental caries in children younger than 5 years: screening and interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1#bootstrap-panel—4
3. ADA. Dietary fluoride supplements: evidence-based clinical recommendations. Accessed April 14, 2022. www.ada.org/-/media/project/ada-organization/ada/ada-org/files/resources/research/ada_evidence-based_fluoride_supplement_chairside_guide.pdf?rev=60850dca0dcc41038efda83d42b1c2e0&hash=FEC2BBEA0C892FB12C098E33344E48B4
4. USPSTF. Gestational diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening
5. USPSTF. Healthy weight and weight gain in pregnancy: behavioral counseling interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/healthy-weight-and-weight-gain-during-pregnancy-behavioral-counseling-interventions
6. USPSTF. Aspirin use to prevent preeclampsia and related morbidity and mortality: preventive medication. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/low-dose-aspirin-use-for-the-prevention-of-morbidity-and-mortality-from-preeclampsia-preventive-medication
7. USPSTF. Chlamydia and gonorrhea: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/chlamydia-and-gonorrhea-screening
8. Workowski KA, Bauchman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70:1-187.
9. Campos-Outcalt D. CDC guidelines on sexually transmitted infections. J Fam Pract. 2021;70:506-509.
10. USPSTF. Prediabetes and type 2 diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/screening-for-prediabetes-and-type-2-diabetes
11. USPSTF. Colorectal cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
12. USPSTF. Lung cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
13. USPSTF. Tobacco smoking cessation in adults, including pregnant persons: interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
14. USPSTF. Hypertension in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hypertension-in-adults-screening
15. USPSTF. Vitamin D deficiency in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/vitamin-d-deficiency-screening
16. USPSTF. Hearing loss in older adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hearing-loss-in-older-adults-screening
17. USPSTF. Asymptomatic carotid artery stenosis: screening. Access April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
18. Campos-Outcalt D. How to proceed when it comes to vitamin D. J Fam Pract. 2021;70:289-292.
In 2021, the US Preventive Services Task Force (USPSTF) considered 13 topics and made a total of 23 recommendations. They reviewed only 1 new topic. The other 12 were updates of topics previously addressed; no changes were made in 9 of them. In 3, the recommended age of screening or the criteria for screening were expanded. This Practice Alert will review the recommendations made and highlight new recommendations and any changes to previous ones. All complete recommendation statements, rationales, clinical considerations, and evidence reports can be found on the USPSTF website at https://uspreventiveservicestaskforce.org/uspstf/home.1
Dental caries in children
Dental caries affect about 23% of children between the ages of 2 and 5 years and are associated with multiple adverse social outcomes and medical conditions.2 The best way to prevent tooth decay, other than regular brushing with fluoride toothpaste, is to drink water with recommended amounts of fluoride (≥ 0.6 parts fluoride per million parts water).2 The USPSTF reaffirmed its recommendation from 2014 that stated when a local water supply lacks sufficient fluoride, primary care clinicians should prescribe oral supplementation for infants and children in the form of fluoride drops starting at age 6 months. The dosage of fluoride depends on patient age and fluoride concentration in the local water (TABLE 13). The USPSTF also recommends applying topical fluoride as 5% sodium fluoride varnish, every 6 months, starting when the primary teeth erupt.2

In addition to fluoride supplements and topical varnish, should clinicians perform screening examinations looking for dental caries? The USPSTF feels there is not enough evidence to assess this practice and gives it an “I” rating (insufficient evidence).
Preventive interventions in pregnancy
In 2021, the USPSTF assessed 3 topics related to pregnancy and prenatal care.
Screening for gestational diabetes. The USPSTF gave a “B” recommendation for screening at 24 weeks of pregnancy or after, but an “I” statement for screening prior to 24 weeks.4 Screening can involve a 1-step or 2-step protocol.
The 2-step protocol is most commonly used in the United States. It involves first measuring serum glucose after a nonfasting 50-g oral glucose challenge; if the resulting level is high, the second step is a 75- or 100-g oral glucose tolerance test lasting 3 hours. The 1-step protocol involves measuring a fasting glucose level, followed by a 75-g oral glucose challenge with glucose levels measured at 1 and 2 hours.
Healthy weight gain in pregnancy. This was the only new topic the USPSTF assessed last year. The resulting recommendation is to offer pregnant women behavioral counseling to promote healthy weight gain and to prevent excessive weight gain in pregnancy. The recommended weight gain depends on the mother’s prepregnancy weight status: 28 to 40 lbs if the mother is underweight; 25 to 35 lbs if she is not under- or overweight; 15 to 25 lbs if she is overweight; and 11 to 20 lbs if she is obese.5 Healthy weight gain contributes to preventing gestational diabetes, emergency cesarean sections, and infant macrosomia.
Continue to: Low-dose aspirin
Low-dose aspirin. Reaffirming a recommendation from 2014, the USPSTF advises low-dose aspirin (81 mg/d) starting after 12 weeks’ gestation for all pregnant women who are at high risk for preeclampsia. TABLE 26 lists high- and moderate-risk conditions for preeclampsia and the recommendation for the use of low-dose aspirin.

Sexually transmitted infections
Screening for both chlamydia and gonorrhea in sexually active females through age 24 years was given a “B” recommendation, reaffirming the 2014 recommendation.7 Screening for these 2 sexually transmitted infections (STIs) is also recommended for women 25 years and older who are at increased risk of STIs. Risk is defined as having a new sex partner, more than 1 sex partner, a sex partner who has other sex partners, or a sex partner who has an STI; not using condoms consistently; having a previous STI; exchanging sex for money or drugs; or having a history of incarceration.
Screen for both infections simultaneously using a nucleic acid amplification test, testing all sites of sexual exposure. Urine testing can replace cervical, vaginal, and urethral testing. Those found to be positive for either STI should be treated according to the most recent treatment guidelines from the Centers for Disease Control and Prevention (CDC). And sexual partners should be advised to undergo testing.8,9
The USPSTF could not find evidence for the benefits and harms of screening for STIs in men. Remember that screening applies to those who are asymptomatic. Male sex partners of those found to be infected should be tested, as should those who show any signs or symptoms of an STI. A recent Practice Alert described the most current CDC guidance for diagnosing and treating STIs.9
Type 2 diabetes and prediabetes
Screening for type 2 diabetes (T2D) and prediabetes is now recommended for adults ages 35 to 70 years who are overweight or obese.10 The age to start screening has been lowered to 35 years from the previous recommendation in 2015, which recommended starting at age 40. In addition, the recommendation states that patients with prediabetes should be referred for preventive interventions. It is important that referral is included in the statement because the Affordable Care Act mandates that USPSTF “A” and “B” recommendations must be covered by commercial health insurance with no copay or deductible.
Continue to: Screening can be conducted...
Screening can be conducted using a fasting plasma glucose or A1C level, or with an oral glucose tolerance test. Interventions that can prevent or delay the onset of T2D in those with prediabetes include lifestyle interventions that focus on diet and physical activity, and the use of metformin (although metformin has not been approved for this by the US Food and Drug Administration).
Changes to cancer screening recommendations
In 2021, the USPSTF reviewed and modified its recommendations on screening for 2 types of cancer: colorectal and lung.
For colorectal cancer, the age at which to start screening was lowered from 50 years to 45 years.11 Screening at this earlier age is a “B” recommendation, because, while there is benefit from screening, it is less than for older age groups. Screening individuals ages 50 to 75 years remains an “A” recommendation, and for those ages 76 to 85 years it remains a “C” recommendation. A “C” recommendation means that the overall benefits are small but some individuals might benefit based on their overall health and prior screening results. In its clinical considerations, the USPSTF recommends against screening in those ages 85 and older but, curiously, does not list it as a “D” recommendation. The screening methods and recommended screening intervals for each appear in TABLE 3.11

For lung cancer, annual screening using low-dose computed tomography (CT) was first recommended by the USPSTF in 2013 for adults ages 55 to 80 years with a 30-pack-year smoking history. Screening could stop once 15 years had passed since smoking cessation. In 2021, the USPSTF lowered the age to initiate screening to 50 years, and the smoking history threshold to 20 pack-years.12 If these recommendations are followed, a current smoker who does not quit smoking could possibly receive 30 annual CT scans. The recommendation does state that screening should stop once a person develops a health condition that significantly affects life expectancy or ability to have lung surgery.
For primary prevention of lung cancer and other chronic diseases through smoking cessation, the USPSTF also reassessed its 2015 recommendations. It reaffirmed the “A” recommendation to ask adults about tobacco use and, for tobacco users, to recommend cessation and provide behavioral therapy and approved pharmacotherapy.13 The recommendation differed for pregnant adults in that the USPSTF is unsure about the potential harms of pharmacotherapy in pregnancy and gives that an “I” statement.13 An additional “I” statement was made about the use of electronic cigarettes for smoking cessation; the USPSTF recommends using behavioral and pharmacotherapy interventions with proven effectiveness and safety instead.
Continue to: 4 additional recommendation updates with no changes
4 additional recommendation updates with no changes
Screening for high blood pressure in adults ages 18 years and older continues to receive an “A” recommendation.14 Importantly, the recommendation states that confirmation of high blood pressure should be made in an out-of-office setting before initiating treatment. Screening for vitamin D deficiency in adults and hearing loss in older adults both continue with “I” statements,15,16 and screening for asymptomatic carotid artery stenosis continues to receive a “D” recommendation.17 The implications of the vitamin D “I” statement were discussed in a previous Practice Alert.18
Continuing value of the USPSTF
The USPSTF continues to set the gold standard for assessment of preventive interventions, and its decisions affect first-dollar coverage by commercial health insurance. The reaffirmation of past recommendations demonstrates the value of adhering to rigorous evidence-based methods (if they are done correctly, they rarely must be markedly changed). And the updating of screening criteria shows the need to constantly review the evolving evidence for current recommendations. Once again, however, funding and staffing limitations allowed the USPSTF to assess only 1 new topic. A listing of all the 2021 recommendations is in TABLE 4.1


In 2021, the US Preventive Services Task Force (USPSTF) considered 13 topics and made a total of 23 recommendations. They reviewed only 1 new topic. The other 12 were updates of topics previously addressed; no changes were made in 9 of them. In 3, the recommended age of screening or the criteria for screening were expanded. This Practice Alert will review the recommendations made and highlight new recommendations and any changes to previous ones. All complete recommendation statements, rationales, clinical considerations, and evidence reports can be found on the USPSTF website at https://uspreventiveservicestaskforce.org/uspstf/home.1
Dental caries in children
Dental caries affect about 23% of children between the ages of 2 and 5 years and are associated with multiple adverse social outcomes and medical conditions.2 The best way to prevent tooth decay, other than regular brushing with fluoride toothpaste, is to drink water with recommended amounts of fluoride (≥ 0.6 parts fluoride per million parts water).2 The USPSTF reaffirmed its recommendation from 2014 that stated when a local water supply lacks sufficient fluoride, primary care clinicians should prescribe oral supplementation for infants and children in the form of fluoride drops starting at age 6 months. The dosage of fluoride depends on patient age and fluoride concentration in the local water (TABLE 13). The USPSTF also recommends applying topical fluoride as 5% sodium fluoride varnish, every 6 months, starting when the primary teeth erupt.2

In addition to fluoride supplements and topical varnish, should clinicians perform screening examinations looking for dental caries? The USPSTF feels there is not enough evidence to assess this practice and gives it an “I” rating (insufficient evidence).
Preventive interventions in pregnancy
In 2021, the USPSTF assessed 3 topics related to pregnancy and prenatal care.
Screening for gestational diabetes. The USPSTF gave a “B” recommendation for screening at 24 weeks of pregnancy or after, but an “I” statement for screening prior to 24 weeks.4 Screening can involve a 1-step or 2-step protocol.
The 2-step protocol is most commonly used in the United States. It involves first measuring serum glucose after a nonfasting 50-g oral glucose challenge; if the resulting level is high, the second step is a 75- or 100-g oral glucose tolerance test lasting 3 hours. The 1-step protocol involves measuring a fasting glucose level, followed by a 75-g oral glucose challenge with glucose levels measured at 1 and 2 hours.
Healthy weight gain in pregnancy. This was the only new topic the USPSTF assessed last year. The resulting recommendation is to offer pregnant women behavioral counseling to promote healthy weight gain and to prevent excessive weight gain in pregnancy. The recommended weight gain depends on the mother’s prepregnancy weight status: 28 to 40 lbs if the mother is underweight; 25 to 35 lbs if she is not under- or overweight; 15 to 25 lbs if she is overweight; and 11 to 20 lbs if she is obese.5 Healthy weight gain contributes to preventing gestational diabetes, emergency cesarean sections, and infant macrosomia.
Continue to: Low-dose aspirin
Low-dose aspirin. Reaffirming a recommendation from 2014, the USPSTF advises low-dose aspirin (81 mg/d) starting after 12 weeks’ gestation for all pregnant women who are at high risk for preeclampsia. TABLE 26 lists high- and moderate-risk conditions for preeclampsia and the recommendation for the use of low-dose aspirin.

Sexually transmitted infections
Screening for both chlamydia and gonorrhea in sexually active females through age 24 years was given a “B” recommendation, reaffirming the 2014 recommendation.7 Screening for these 2 sexually transmitted infections (STIs) is also recommended for women 25 years and older who are at increased risk of STIs. Risk is defined as having a new sex partner, more than 1 sex partner, a sex partner who has other sex partners, or a sex partner who has an STI; not using condoms consistently; having a previous STI; exchanging sex for money or drugs; or having a history of incarceration.
Screen for both infections simultaneously using a nucleic acid amplification test, testing all sites of sexual exposure. Urine testing can replace cervical, vaginal, and urethral testing. Those found to be positive for either STI should be treated according to the most recent treatment guidelines from the Centers for Disease Control and Prevention (CDC). And sexual partners should be advised to undergo testing.8,9
The USPSTF could not find evidence for the benefits and harms of screening for STIs in men. Remember that screening applies to those who are asymptomatic. Male sex partners of those found to be infected should be tested, as should those who show any signs or symptoms of an STI. A recent Practice Alert described the most current CDC guidance for diagnosing and treating STIs.9
Type 2 diabetes and prediabetes
Screening for type 2 diabetes (T2D) and prediabetes is now recommended for adults ages 35 to 70 years who are overweight or obese.10 The age to start screening has been lowered to 35 years from the previous recommendation in 2015, which recommended starting at age 40. In addition, the recommendation states that patients with prediabetes should be referred for preventive interventions. It is important that referral is included in the statement because the Affordable Care Act mandates that USPSTF “A” and “B” recommendations must be covered by commercial health insurance with no copay or deductible.
Continue to: Screening can be conducted...
Screening can be conducted using a fasting plasma glucose or A1C level, or with an oral glucose tolerance test. Interventions that can prevent or delay the onset of T2D in those with prediabetes include lifestyle interventions that focus on diet and physical activity, and the use of metformin (although metformin has not been approved for this by the US Food and Drug Administration).
Changes to cancer screening recommendations
In 2021, the USPSTF reviewed and modified its recommendations on screening for 2 types of cancer: colorectal and lung.
For colorectal cancer, the age at which to start screening was lowered from 50 years to 45 years.11 Screening at this earlier age is a “B” recommendation, because, while there is benefit from screening, it is less than for older age groups. Screening individuals ages 50 to 75 years remains an “A” recommendation, and for those ages 76 to 85 years it remains a “C” recommendation. A “C” recommendation means that the overall benefits are small but some individuals might benefit based on their overall health and prior screening results. In its clinical considerations, the USPSTF recommends against screening in those ages 85 and older but, curiously, does not list it as a “D” recommendation. The screening methods and recommended screening intervals for each appear in TABLE 3.11

For lung cancer, annual screening using low-dose computed tomography (CT) was first recommended by the USPSTF in 2013 for adults ages 55 to 80 years with a 30-pack-year smoking history. Screening could stop once 15 years had passed since smoking cessation. In 2021, the USPSTF lowered the age to initiate screening to 50 years, and the smoking history threshold to 20 pack-years.12 If these recommendations are followed, a current smoker who does not quit smoking could possibly receive 30 annual CT scans. The recommendation does state that screening should stop once a person develops a health condition that significantly affects life expectancy or ability to have lung surgery.
For primary prevention of lung cancer and other chronic diseases through smoking cessation, the USPSTF also reassessed its 2015 recommendations. It reaffirmed the “A” recommendation to ask adults about tobacco use and, for tobacco users, to recommend cessation and provide behavioral therapy and approved pharmacotherapy.13 The recommendation differed for pregnant adults in that the USPSTF is unsure about the potential harms of pharmacotherapy in pregnancy and gives that an “I” statement.13 An additional “I” statement was made about the use of electronic cigarettes for smoking cessation; the USPSTF recommends using behavioral and pharmacotherapy interventions with proven effectiveness and safety instead.
Continue to: 4 additional recommendation updates with no changes
4 additional recommendation updates with no changes
Screening for high blood pressure in adults ages 18 years and older continues to receive an “A” recommendation.14 Importantly, the recommendation states that confirmation of high blood pressure should be made in an out-of-office setting before initiating treatment. Screening for vitamin D deficiency in adults and hearing loss in older adults both continue with “I” statements,15,16 and screening for asymptomatic carotid artery stenosis continues to receive a “D” recommendation.17 The implications of the vitamin D “I” statement were discussed in a previous Practice Alert.18
Continuing value of the USPSTF
The USPSTF continues to set the gold standard for assessment of preventive interventions, and its decisions affect first-dollar coverage by commercial health insurance. The reaffirmation of past recommendations demonstrates the value of adhering to rigorous evidence-based methods (if they are done correctly, they rarely must be markedly changed). And the updating of screening criteria shows the need to constantly review the evolving evidence for current recommendations. Once again, however, funding and staffing limitations allowed the USPSTF to assess only 1 new topic. A listing of all the 2021 recommendations is in TABLE 4.1


1. USPSTF. Recommendation topics. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation-topics
2. USPSTF. Prevention of dental caries in children younger than 5 years: screening and interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1#bootstrap-panel—4
3. ADA. Dietary fluoride supplements: evidence-based clinical recommendations. Accessed April 14, 2022. www.ada.org/-/media/project/ada-organization/ada/ada-org/files/resources/research/ada_evidence-based_fluoride_supplement_chairside_guide.pdf?rev=60850dca0dcc41038efda83d42b1c2e0&hash=FEC2BBEA0C892FB12C098E33344E48B4
4. USPSTF. Gestational diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening
5. USPSTF. Healthy weight and weight gain in pregnancy: behavioral counseling interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/healthy-weight-and-weight-gain-during-pregnancy-behavioral-counseling-interventions
6. USPSTF. Aspirin use to prevent preeclampsia and related morbidity and mortality: preventive medication. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/low-dose-aspirin-use-for-the-prevention-of-morbidity-and-mortality-from-preeclampsia-preventive-medication
7. USPSTF. Chlamydia and gonorrhea: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/chlamydia-and-gonorrhea-screening
8. Workowski KA, Bauchman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70:1-187.
9. Campos-Outcalt D. CDC guidelines on sexually transmitted infections. J Fam Pract. 2021;70:506-509.
10. USPSTF. Prediabetes and type 2 diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/screening-for-prediabetes-and-type-2-diabetes
11. USPSTF. Colorectal cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
12. USPSTF. Lung cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
13. USPSTF. Tobacco smoking cessation in adults, including pregnant persons: interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
14. USPSTF. Hypertension in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hypertension-in-adults-screening
15. USPSTF. Vitamin D deficiency in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/vitamin-d-deficiency-screening
16. USPSTF. Hearing loss in older adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hearing-loss-in-older-adults-screening
17. USPSTF. Asymptomatic carotid artery stenosis: screening. Access April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
18. Campos-Outcalt D. How to proceed when it comes to vitamin D. J Fam Pract. 2021;70:289-292.
1. USPSTF. Recommendation topics. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation-topics
2. USPSTF. Prevention of dental caries in children younger than 5 years: screening and interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-dental-caries-in-children-younger-than-age-5-years-screening-and-interventions1#bootstrap-panel—4
3. ADA. Dietary fluoride supplements: evidence-based clinical recommendations. Accessed April 14, 2022. www.ada.org/-/media/project/ada-organization/ada/ada-org/files/resources/research/ada_evidence-based_fluoride_supplement_chairside_guide.pdf?rev=60850dca0dcc41038efda83d42b1c2e0&hash=FEC2BBEA0C892FB12C098E33344E48B4
4. USPSTF. Gestational diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/gestational-diabetes-screening
5. USPSTF. Healthy weight and weight gain in pregnancy: behavioral counseling interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/healthy-weight-and-weight-gain-during-pregnancy-behavioral-counseling-interventions
6. USPSTF. Aspirin use to prevent preeclampsia and related morbidity and mortality: preventive medication. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/low-dose-aspirin-use-for-the-prevention-of-morbidity-and-mortality-from-preeclampsia-preventive-medication
7. USPSTF. Chlamydia and gonorrhea: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/chlamydia-and-gonorrhea-screening
8. Workowski KA, Bauchman LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70:1-187.
9. Campos-Outcalt D. CDC guidelines on sexually transmitted infections. J Fam Pract. 2021;70:506-509.
10. USPSTF. Prediabetes and type 2 diabetes: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/screening-for-prediabetes-and-type-2-diabetes
11. USPSTF. Colorectal cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening
12. USPSTF. Lung cancer: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
13. USPSTF. Tobacco smoking cessation in adults, including pregnant persons: interventions. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/tobacco-use-in-adults-and-pregnant-women-counseling-and-interventions
14. USPSTF. Hypertension in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hypertension-in-adults-screening
15. USPSTF. Vitamin D deficiency in adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/vitamin-d-deficiency-screening
16. USPSTF. Hearing loss in older adults: screening. Accessed April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/hearing-loss-in-older-adults-screening
17. USPSTF. Asymptomatic carotid artery stenosis: screening. Access April 14, 2022. https://uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening
18. Campos-Outcalt D. How to proceed when it comes to vitamin D. J Fam Pract. 2021;70:289-292.
Alarming global rise in pediatric hepatitis: Expert Q&A
This spring, global health advisories have been issued regarding an alarming – and as-yet unexplained – uptick of hepatitis in children. Currently, over 200 cases have been reported worldwide, a relatively small amount that nonetheless belies a considerable toll, including several deaths and the need for liver transplantation in a number of patients. The long-term implications are not yet known. Global health officials are working hard to determine a cause, with many focusing on the underlying cases of adenovirus that several patients have presented with.
To understand more, this news organization reached out to frequent contributor William F. Balistreri, MD, a specialist in pediatric gastroenterology and hepatology at Cincinnati Children’s Hospital Medical Center, where to date they have treated at least six cases of hepatitis in otherwise healthy young children, with one requiring a liver transplant. Dr. Balistreri discussed how the outbreak has developed to date, his advice to hepatologists and pediatricians, and where we stand now in this fast-evolving crisis.
Tracing the outbreak in the United States
How has this outbreak played out thus far in the United States, and what have we learned from that?
Sporadic reports of cases in multiple states are appearing. On April 21, 2022, a health alert was issued by the Centers for Disease Control and Prevention, recommending testing for adenovirus in children with acute hepatitis of an unknown etiology.
Baker and colleagues recently described five children with severe hepatitis and adenovirus viremia who were admitted to a children’s hospital in Birmingham, Ala., between October and November 2021. In collaboration with local and state officials, the CDC reviewed clinical records in order to identify patients with hepatitis and concomitant adenovirus infection, confirmed by polymerase chain reaction (PCR).
By February 2022, a total of nine children were identified. There was no epidemiologic linkage among these nine patients; all were well and immunocompetent. The prodromal features were somewhat similar: upper respiratory infection, vomiting, diarrhea, and jaundice. All children had markedly elevated aminotransferase levels and variably elevated total bilirubin levels. Extensive workup for other causes of acute liver injury (for example, other viruses, toxins/drugs, metabolic and autoimmune diseases) was unrevealing.
Specifically, none had documented SARS-CoV-2 infection. However, in all nine children, adenovirus was detected in whole blood samples. In the six children who underwent liver biopsy, there was nonspecific hepatitis, without inclusions or immunohistochemical detection of viral agents, including adenovirus. In three patients, the liver injury progressed, and despite the administration of antiviral agents, two underwent liver transplantation.
Baker and colleagues also suggested that measurement of adenovirus titers in whole blood (rather than plasma) may be more sensitive.
The CDC has recommended monitoring and surveillance in order to more fully understand the nature of the illness.
European and global cases
What has been the experience with this in Europe and elsewhere globally?
In mid-to-late 2021, several cases of acute hepatitis of unknown nature in children were identified in Europe. Public health officials in the United Kingdom investigated the high number of cases seen in children from England, Scotland, and Wales. They noted approximately 60 cases in England, mostly in children aged 2-5 years.
Marsh and colleagues reported a cluster of cases of severe hepatitis of unknown origin in Scotland affecting children aged 3-5 years. In Scotland, admitted cases were routinely tested for SARS-CoV-2. Of the 13 cases, five had a recent positive test. They discussed the possibility of increased severity of disease following infection with Omicron BA.2 (the dominant SARS-CoV-2 virus circulating in Scotland at that time) or infection by an uncharacterized SARS-CoV-2 variant. None of the children had been vaccinated for SARS-CoV-2.
On April 15, 2022, the World Health Organization Disease Outbreak News published a report of acute hepatitis of unknown etiology occurring in Great Britain and Northern Ireland. By April 21, 2022, 169 cases of acute hepatitis of unknown origin in children younger than 16 years had been reported from 11 countries in the WHO European region and 1 country in the WHO region of the Americas. Approximately 10% required a liver transplantation and at least one death was reported.
What has been established about the possible connection to the SARS-CoV-2 virus, particularly as it relates to coinfection with adenovirus?
In that WHO report of 169 cases, adenovirus was detected in 74 and SARS-CoV-2 in 20. Of note, 19 cases had a SARS-CoV-2 and adenovirus coinfection.
The report’s authors emphasized that, “while adenovirus is a possible hypothesis, investigations are ongoing for the causative agent.” The authors questioned whether this represents a continuing increase in cases of hepatitis or reflects an increased awareness.
The stated priority of the WHO is to determine the cause and to further refine control and prevention actions.
Given the worldwide nature of this outbreak, have connections between any of the cases been made yet?
Not to my knowledge.
What clinicians need to know
What makes this outbreak of hepatitis cases particularly concerning to the health care community, in comparison to other childhood diseases that occur globally? Is it because the cause is unknown or is it for other reasons?
It may be a collective heightened concern following the emergence of COVID.
Whether it represents a new form of acute hepatitis, a continuing increase in cases of hepatitis, or an increased awareness because of the well-publicized alerts remains to be determined. We certainly saw “viral-induced hepatitis” in the past.
Young patients may first be brought to pediatricians. What, if anything, should pediatricians be on the lookout for? Do they need a heightened index of suspicion or are the cases too rare at this point?
An awareness of the “outbreak” may allow the clinician to extend the typical workup of a child presenting with an undefined, presumably viral illness.
In the cases reported, the prodromal and/or presenting symptoms were respiratory and gastrointestinal in nature. They include nausea, vomiting, diarrhea, and abdominal pain.
Specifically, if jaundice and/or scleral icterus is noted, then hepatitis should be suspected.
Should pediatricians consider early referral to a pediatric gastroenterologist or hepatologist?
Yes, because there is the potential for finding a treatable cause (for example, autoimmune hepatitis or a specific metabolic disease) in a patient presenting in this fashion.
In addition, the potential for progression to acute liver failure (with coagulopathy and encephalopathy), albeit rare, exists.
What do hepatologists need to be doing when presented with suspected cases?
The typical clinical picture holds and the workup is standard. The one new key, given the recent data, is to test for adenovirus, using whole blood versus plasma, as the former may be more sensitive.
In addition, it is prudent to check for SARS-CoV-2 by PCR.
What are the major questions that remain and that you’d like to see elucidated going forward?
There are many. Is this a new disease? A new variant of adenovirus? A synergy or susceptibility related to SARS-CoV-2? Is it related to a variant of SARS-CoV-2? Is it triggering an adverse immune response? Are there other epigenetic factors involved? And finally, is this an increase, or is it related to a collective heightened concern following the pandemic?
Dr. Balistreri is the Dorothy M.M. Kersten Professor of Pediatrics, director emeritus of the Pediatric Liver Care Center, medical director emeritus of liver transplantation, and professor at the University of Cincinnati; he is also with the department of pediatrics at Cincinnati Children’s Hospital Medical Center.
A version of this article first appeared on Medscape.com.
This spring, global health advisories have been issued regarding an alarming – and as-yet unexplained – uptick of hepatitis in children. Currently, over 200 cases have been reported worldwide, a relatively small amount that nonetheless belies a considerable toll, including several deaths and the need for liver transplantation in a number of patients. The long-term implications are not yet known. Global health officials are working hard to determine a cause, with many focusing on the underlying cases of adenovirus that several patients have presented with.
To understand more, this news organization reached out to frequent contributor William F. Balistreri, MD, a specialist in pediatric gastroenterology and hepatology at Cincinnati Children’s Hospital Medical Center, where to date they have treated at least six cases of hepatitis in otherwise healthy young children, with one requiring a liver transplant. Dr. Balistreri discussed how the outbreak has developed to date, his advice to hepatologists and pediatricians, and where we stand now in this fast-evolving crisis.
Tracing the outbreak in the United States
How has this outbreak played out thus far in the United States, and what have we learned from that?
Sporadic reports of cases in multiple states are appearing. On April 21, 2022, a health alert was issued by the Centers for Disease Control and Prevention, recommending testing for adenovirus in children with acute hepatitis of an unknown etiology.
Baker and colleagues recently described five children with severe hepatitis and adenovirus viremia who were admitted to a children’s hospital in Birmingham, Ala., between October and November 2021. In collaboration with local and state officials, the CDC reviewed clinical records in order to identify patients with hepatitis and concomitant adenovirus infection, confirmed by polymerase chain reaction (PCR).
By February 2022, a total of nine children were identified. There was no epidemiologic linkage among these nine patients; all were well and immunocompetent. The prodromal features were somewhat similar: upper respiratory infection, vomiting, diarrhea, and jaundice. All children had markedly elevated aminotransferase levels and variably elevated total bilirubin levels. Extensive workup for other causes of acute liver injury (for example, other viruses, toxins/drugs, metabolic and autoimmune diseases) was unrevealing.
Specifically, none had documented SARS-CoV-2 infection. However, in all nine children, adenovirus was detected in whole blood samples. In the six children who underwent liver biopsy, there was nonspecific hepatitis, without inclusions or immunohistochemical detection of viral agents, including adenovirus. In three patients, the liver injury progressed, and despite the administration of antiviral agents, two underwent liver transplantation.
Baker and colleagues also suggested that measurement of adenovirus titers in whole blood (rather than plasma) may be more sensitive.
The CDC has recommended monitoring and surveillance in order to more fully understand the nature of the illness.
European and global cases
What has been the experience with this in Europe and elsewhere globally?
In mid-to-late 2021, several cases of acute hepatitis of unknown nature in children were identified in Europe. Public health officials in the United Kingdom investigated the high number of cases seen in children from England, Scotland, and Wales. They noted approximately 60 cases in England, mostly in children aged 2-5 years.
Marsh and colleagues reported a cluster of cases of severe hepatitis of unknown origin in Scotland affecting children aged 3-5 years. In Scotland, admitted cases were routinely tested for SARS-CoV-2. Of the 13 cases, five had a recent positive test. They discussed the possibility of increased severity of disease following infection with Omicron BA.2 (the dominant SARS-CoV-2 virus circulating in Scotland at that time) or infection by an uncharacterized SARS-CoV-2 variant. None of the children had been vaccinated for SARS-CoV-2.
On April 15, 2022, the World Health Organization Disease Outbreak News published a report of acute hepatitis of unknown etiology occurring in Great Britain and Northern Ireland. By April 21, 2022, 169 cases of acute hepatitis of unknown origin in children younger than 16 years had been reported from 11 countries in the WHO European region and 1 country in the WHO region of the Americas. Approximately 10% required a liver transplantation and at least one death was reported.
What has been established about the possible connection to the SARS-CoV-2 virus, particularly as it relates to coinfection with adenovirus?
In that WHO report of 169 cases, adenovirus was detected in 74 and SARS-CoV-2 in 20. Of note, 19 cases had a SARS-CoV-2 and adenovirus coinfection.
The report’s authors emphasized that, “while adenovirus is a possible hypothesis, investigations are ongoing for the causative agent.” The authors questioned whether this represents a continuing increase in cases of hepatitis or reflects an increased awareness.
The stated priority of the WHO is to determine the cause and to further refine control and prevention actions.
Given the worldwide nature of this outbreak, have connections between any of the cases been made yet?
Not to my knowledge.
What clinicians need to know
What makes this outbreak of hepatitis cases particularly concerning to the health care community, in comparison to other childhood diseases that occur globally? Is it because the cause is unknown or is it for other reasons?
It may be a collective heightened concern following the emergence of COVID.
Whether it represents a new form of acute hepatitis, a continuing increase in cases of hepatitis, or an increased awareness because of the well-publicized alerts remains to be determined. We certainly saw “viral-induced hepatitis” in the past.
Young patients may first be brought to pediatricians. What, if anything, should pediatricians be on the lookout for? Do they need a heightened index of suspicion or are the cases too rare at this point?
An awareness of the “outbreak” may allow the clinician to extend the typical workup of a child presenting with an undefined, presumably viral illness.
In the cases reported, the prodromal and/or presenting symptoms were respiratory and gastrointestinal in nature. They include nausea, vomiting, diarrhea, and abdominal pain.
Specifically, if jaundice and/or scleral icterus is noted, then hepatitis should be suspected.
Should pediatricians consider early referral to a pediatric gastroenterologist or hepatologist?
Yes, because there is the potential for finding a treatable cause (for example, autoimmune hepatitis or a specific metabolic disease) in a patient presenting in this fashion.
In addition, the potential for progression to acute liver failure (with coagulopathy and encephalopathy), albeit rare, exists.
What do hepatologists need to be doing when presented with suspected cases?
The typical clinical picture holds and the workup is standard. The one new key, given the recent data, is to test for adenovirus, using whole blood versus plasma, as the former may be more sensitive.
In addition, it is prudent to check for SARS-CoV-2 by PCR.
What are the major questions that remain and that you’d like to see elucidated going forward?
There are many. Is this a new disease? A new variant of adenovirus? A synergy or susceptibility related to SARS-CoV-2? Is it related to a variant of SARS-CoV-2? Is it triggering an adverse immune response? Are there other epigenetic factors involved? And finally, is this an increase, or is it related to a collective heightened concern following the pandemic?
Dr. Balistreri is the Dorothy M.M. Kersten Professor of Pediatrics, director emeritus of the Pediatric Liver Care Center, medical director emeritus of liver transplantation, and professor at the University of Cincinnati; he is also with the department of pediatrics at Cincinnati Children’s Hospital Medical Center.
A version of this article first appeared on Medscape.com.
This spring, global health advisories have been issued regarding an alarming – and as-yet unexplained – uptick of hepatitis in children. Currently, over 200 cases have been reported worldwide, a relatively small amount that nonetheless belies a considerable toll, including several deaths and the need for liver transplantation in a number of patients. The long-term implications are not yet known. Global health officials are working hard to determine a cause, with many focusing on the underlying cases of adenovirus that several patients have presented with.
To understand more, this news organization reached out to frequent contributor William F. Balistreri, MD, a specialist in pediatric gastroenterology and hepatology at Cincinnati Children’s Hospital Medical Center, where to date they have treated at least six cases of hepatitis in otherwise healthy young children, with one requiring a liver transplant. Dr. Balistreri discussed how the outbreak has developed to date, his advice to hepatologists and pediatricians, and where we stand now in this fast-evolving crisis.
Tracing the outbreak in the United States
How has this outbreak played out thus far in the United States, and what have we learned from that?
Sporadic reports of cases in multiple states are appearing. On April 21, 2022, a health alert was issued by the Centers for Disease Control and Prevention, recommending testing for adenovirus in children with acute hepatitis of an unknown etiology.
Baker and colleagues recently described five children with severe hepatitis and adenovirus viremia who were admitted to a children’s hospital in Birmingham, Ala., between October and November 2021. In collaboration with local and state officials, the CDC reviewed clinical records in order to identify patients with hepatitis and concomitant adenovirus infection, confirmed by polymerase chain reaction (PCR).
By February 2022, a total of nine children were identified. There was no epidemiologic linkage among these nine patients; all were well and immunocompetent. The prodromal features were somewhat similar: upper respiratory infection, vomiting, diarrhea, and jaundice. All children had markedly elevated aminotransferase levels and variably elevated total bilirubin levels. Extensive workup for other causes of acute liver injury (for example, other viruses, toxins/drugs, metabolic and autoimmune diseases) was unrevealing.
Specifically, none had documented SARS-CoV-2 infection. However, in all nine children, adenovirus was detected in whole blood samples. In the six children who underwent liver biopsy, there was nonspecific hepatitis, without inclusions or immunohistochemical detection of viral agents, including adenovirus. In three patients, the liver injury progressed, and despite the administration of antiviral agents, two underwent liver transplantation.
Baker and colleagues also suggested that measurement of adenovirus titers in whole blood (rather than plasma) may be more sensitive.
The CDC has recommended monitoring and surveillance in order to more fully understand the nature of the illness.
European and global cases
What has been the experience with this in Europe and elsewhere globally?
In mid-to-late 2021, several cases of acute hepatitis of unknown nature in children were identified in Europe. Public health officials in the United Kingdom investigated the high number of cases seen in children from England, Scotland, and Wales. They noted approximately 60 cases in England, mostly in children aged 2-5 years.
Marsh and colleagues reported a cluster of cases of severe hepatitis of unknown origin in Scotland affecting children aged 3-5 years. In Scotland, admitted cases were routinely tested for SARS-CoV-2. Of the 13 cases, five had a recent positive test. They discussed the possibility of increased severity of disease following infection with Omicron BA.2 (the dominant SARS-CoV-2 virus circulating in Scotland at that time) or infection by an uncharacterized SARS-CoV-2 variant. None of the children had been vaccinated for SARS-CoV-2.
On April 15, 2022, the World Health Organization Disease Outbreak News published a report of acute hepatitis of unknown etiology occurring in Great Britain and Northern Ireland. By April 21, 2022, 169 cases of acute hepatitis of unknown origin in children younger than 16 years had been reported from 11 countries in the WHO European region and 1 country in the WHO region of the Americas. Approximately 10% required a liver transplantation and at least one death was reported.
What has been established about the possible connection to the SARS-CoV-2 virus, particularly as it relates to coinfection with adenovirus?
In that WHO report of 169 cases, adenovirus was detected in 74 and SARS-CoV-2 in 20. Of note, 19 cases had a SARS-CoV-2 and adenovirus coinfection.
The report’s authors emphasized that, “while adenovirus is a possible hypothesis, investigations are ongoing for the causative agent.” The authors questioned whether this represents a continuing increase in cases of hepatitis or reflects an increased awareness.
The stated priority of the WHO is to determine the cause and to further refine control and prevention actions.
Given the worldwide nature of this outbreak, have connections between any of the cases been made yet?
Not to my knowledge.
What clinicians need to know
What makes this outbreak of hepatitis cases particularly concerning to the health care community, in comparison to other childhood diseases that occur globally? Is it because the cause is unknown or is it for other reasons?
It may be a collective heightened concern following the emergence of COVID.
Whether it represents a new form of acute hepatitis, a continuing increase in cases of hepatitis, or an increased awareness because of the well-publicized alerts remains to be determined. We certainly saw “viral-induced hepatitis” in the past.
Young patients may first be brought to pediatricians. What, if anything, should pediatricians be on the lookout for? Do they need a heightened index of suspicion or are the cases too rare at this point?
An awareness of the “outbreak” may allow the clinician to extend the typical workup of a child presenting with an undefined, presumably viral illness.
In the cases reported, the prodromal and/or presenting symptoms were respiratory and gastrointestinal in nature. They include nausea, vomiting, diarrhea, and abdominal pain.
Specifically, if jaundice and/or scleral icterus is noted, then hepatitis should be suspected.
Should pediatricians consider early referral to a pediatric gastroenterologist or hepatologist?
Yes, because there is the potential for finding a treatable cause (for example, autoimmune hepatitis or a specific metabolic disease) in a patient presenting in this fashion.
In addition, the potential for progression to acute liver failure (with coagulopathy and encephalopathy), albeit rare, exists.
What do hepatologists need to be doing when presented with suspected cases?
The typical clinical picture holds and the workup is standard. The one new key, given the recent data, is to test for adenovirus, using whole blood versus plasma, as the former may be more sensitive.
In addition, it is prudent to check for SARS-CoV-2 by PCR.
What are the major questions that remain and that you’d like to see elucidated going forward?
There are many. Is this a new disease? A new variant of adenovirus? A synergy or susceptibility related to SARS-CoV-2? Is it related to a variant of SARS-CoV-2? Is it triggering an adverse immune response? Are there other epigenetic factors involved? And finally, is this an increase, or is it related to a collective heightened concern following the pandemic?
Dr. Balistreri is the Dorothy M.M. Kersten Professor of Pediatrics, director emeritus of the Pediatric Liver Care Center, medical director emeritus of liver transplantation, and professor at the University of Cincinnati; he is also with the department of pediatrics at Cincinnati Children’s Hospital Medical Center.
A version of this article first appeared on Medscape.com.