User login
Cesarean deliveries drop in women at low risk
Although clinically indicated cesarean deliveries may improve outcomes for mothers and infants, “when not clinically indicated, cesarean delivery is a major surgical intervention that increases risk for adverse outcomes,” wrote Anna M. Frappaolo of Columbia University College of Physicians and Surgeons, New York, and colleagues.
The Healthy People 2030 campaign includes the reduction of cesarean deliveries, but trends in these procedures, especially with regard to diagnoses of labor arrest, have not been well studied, the researchers said.
In an analysis published in JAMA Network Open, the researchers reviewed delivery hospitalizations using data from the National Inpatient Sample from 2000 to 2019.
Births deemed low risk for cesarean delivery were identified by using criteria of the Society for Maternal-Fetal Medicine and additional criteria, and joinpoint regression analysis was used to estimate changes.
The researchers examined overall trends in cesarean deliveries as well as trends for three specific diagnoses: nonreassuring fetal status, labor arrest, and obstructed labor.
The final analysis included 40,517,867 deliveries; of these, 4,885,716 (12.1%) were cesarean deliveries.
Overall, cesarean deliveries in patients deemed at low risk increased from 9.7% in 2000 to 13.9% in 2009, then plateaued and decreased from 13.0% in 2012 to 11.1% in 2019. The average annual percentage change (AAPC) for cesarean delivery was 6.4% for the years from 2000 to 2005, 1.2% from 2005 to 2009, and −2.2% from 2009 to 2019.
Cesarean delivery for nonreassuring fetal status increased over the entire study period, from 3.4% in 2000 to 5.1% in 2019. By contrast, overall cesarean delivery for labor arrest increased from 3.6% in 2000 to a high of 4.8% in 2009, then decreased to 2.7% in 2019. Cesarean deliveries with a diagnosis of obstructed labor decreased from 0.9% in 2008 to 0.3% in 2019.
More specifically, cesarean deliveries for labor arrest in the active phase, latent phase, and second stage of labor increased from 1.5% to 2.1%, 1.1% to 1.5%, and 0.9% to 1.3%, respectively, from 2000 to 2009, and decreased from 2.1% to 1.7% for the active phase, from 1.5% to 1.2% for the latent phase, and from 1.2% to 0.9% for the second stage between 2010 and 2019.
Patients with increased odds of cesarean delivery were older (aged 35-39 years vs. 25-29 years, adjusted odds ratio 1.27), delivered in a hospital in the South vs. the Northeast of the United States (aOR 1.11), and were more likely to be non-Hispanic Black vs. non-Hispanic White (OR 1.23).
Notably, changes in nomenclature and interpretation of intrapartum electronic fetal heart monitoring occurred during the study period, with recommendations for the adoption of a three-tiered system for fetal heart rate patterns in 2008. “It is possible that current evidence and nomenclature related to intrapartum FHR interpretation may result in identification of a larger number of fetuses deemed at indeterminate risk for abnormal acid-base status,” the researchers wrote in their discussion.
The study findings were limited by several factors including the use of administrative discharge data rather than clinical records, the exclusion of patients with chronic conditions associated with cesarean delivery, changes in billing codes during the study period, and the inability to account for the effect of health factors, maternal age, and use of assisted reproductive technology, the researchers noted.
However, the results were strengthened by the large sample size and 20-year study period, as well as the stratification of labor arrest by stage, and suggest uptake of newer recommendations, they said. “Future reductions in cesarean deliveries among patients at low risk for cesarean delivery may be dependent on improved assessment of intrapartum fetal status,” they concluded.
Consider populations and outcomes in cesarean risk assessment
The decreasing rates of cesarean deliveries in the current study can be seen as positive, but more research is needed to examine maternal and neonatal outcomes, and to consider other conditions that affect risk for cesarean delivery, Paolo Ivo Cavoretto, MD, and Massimo Candiani, MD, of IRCCS San Raffaele Scientific Institute, and Antonio Farina, MD, of the University of Bologna, Italy, wrote in an accompanying editorial.
Notably, the study authors identified a population aged 15-39 years as low risk, and an increased risk for cesarean delivery within this range increased with age. “Maternal age remains a major risk factor associated with the risk of cesarean delivery, both from results of this study and those of previous analyses assessing its independence from other related risk factors,” the editorialists said.
The study findings also reflect the changes in standards for labor duration during the study period, they noted. The longer duration of labor may reduce cesarean delivery rates, but it is not without maternal and fetal-neonatal risks, they wrote.
“To be sure that the described trend of cesarean delivery rate reduction can be considered positive, there would be the theoretical need to analyze other maternal-fetal-neonatal outcomes (e.g., rates of operative deliveries, neonatal acidemia, intensive care unit use, maternal hemorrhage, pelvic floor trauma and dysfunction, and psychological distress),” the editorialists concluded.
More research needed to explore clinical decisions
“Reducing the cesarean delivery rate is a top priority, but evidence is lacking on an optimal rate that improves maternal and neonatal outcomes,” Iris Krishna, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Hospital quality and safety committees have been working to decrease cesarean deliveries amongst low-risk women, and identifying contemporary trends gives us insight on whether some of these efforts have translated to a lower cesarean delivery rate,” she said.
Dr. Krishna said she was not surprised by the higher cesarean section rate in the South. “The decision for cesarean delivery is multifaceted, and although this study was not able to assess clinical indications for cesarean delivery or maternal and fetal outcomes, we cannot ignore that social determinants of health contribute greatly to overall health outcomes,” she said. The trends in the current study further underscore the geographic disparities in access to health care present in the South, she added.
“This study notes that cesarean delivery for nonreassuring fetal status increased; however, nonreassuring fetal status as an indication for cesarean delivery can be subjective,” Dr. Krishna said. “Hospital quality and safety committees should consider reviewing the clinical scenarios that led to this decision to identify opportunities for improvement and further education,” she said.
“Defining contemporary trends in cesarean delivery for low-risk patients has merit, but the study findings should be interpreted with caution,” said Dr. Krishna, who is a member of the Ob.Gyn. News advisory board. More research is needed to define an optimal cesarean section rate that promotes positive maternal and fetal outcomes, and to determine whether identifying an optimal rate should be based on patient risk profiles, she said.
The study received no outside funding. Lead author Ms. Frappaolo had no financial conflicts to disclose; nor did the editorial authors or Dr. Krishna.
Although clinically indicated cesarean deliveries may improve outcomes for mothers and infants, “when not clinically indicated, cesarean delivery is a major surgical intervention that increases risk for adverse outcomes,” wrote Anna M. Frappaolo of Columbia University College of Physicians and Surgeons, New York, and colleagues.
The Healthy People 2030 campaign includes the reduction of cesarean deliveries, but trends in these procedures, especially with regard to diagnoses of labor arrest, have not been well studied, the researchers said.
In an analysis published in JAMA Network Open, the researchers reviewed delivery hospitalizations using data from the National Inpatient Sample from 2000 to 2019.
Births deemed low risk for cesarean delivery were identified by using criteria of the Society for Maternal-Fetal Medicine and additional criteria, and joinpoint regression analysis was used to estimate changes.
The researchers examined overall trends in cesarean deliveries as well as trends for three specific diagnoses: nonreassuring fetal status, labor arrest, and obstructed labor.
The final analysis included 40,517,867 deliveries; of these, 4,885,716 (12.1%) were cesarean deliveries.
Overall, cesarean deliveries in patients deemed at low risk increased from 9.7% in 2000 to 13.9% in 2009, then plateaued and decreased from 13.0% in 2012 to 11.1% in 2019. The average annual percentage change (AAPC) for cesarean delivery was 6.4% for the years from 2000 to 2005, 1.2% from 2005 to 2009, and −2.2% from 2009 to 2019.
Cesarean delivery for nonreassuring fetal status increased over the entire study period, from 3.4% in 2000 to 5.1% in 2019. By contrast, overall cesarean delivery for labor arrest increased from 3.6% in 2000 to a high of 4.8% in 2009, then decreased to 2.7% in 2019. Cesarean deliveries with a diagnosis of obstructed labor decreased from 0.9% in 2008 to 0.3% in 2019.
More specifically, cesarean deliveries for labor arrest in the active phase, latent phase, and second stage of labor increased from 1.5% to 2.1%, 1.1% to 1.5%, and 0.9% to 1.3%, respectively, from 2000 to 2009, and decreased from 2.1% to 1.7% for the active phase, from 1.5% to 1.2% for the latent phase, and from 1.2% to 0.9% for the second stage between 2010 and 2019.
Patients with increased odds of cesarean delivery were older (aged 35-39 years vs. 25-29 years, adjusted odds ratio 1.27), delivered in a hospital in the South vs. the Northeast of the United States (aOR 1.11), and were more likely to be non-Hispanic Black vs. non-Hispanic White (OR 1.23).
Notably, changes in nomenclature and interpretation of intrapartum electronic fetal heart monitoring occurred during the study period, with recommendations for the adoption of a three-tiered system for fetal heart rate patterns in 2008. “It is possible that current evidence and nomenclature related to intrapartum FHR interpretation may result in identification of a larger number of fetuses deemed at indeterminate risk for abnormal acid-base status,” the researchers wrote in their discussion.
The study findings were limited by several factors including the use of administrative discharge data rather than clinical records, the exclusion of patients with chronic conditions associated with cesarean delivery, changes in billing codes during the study period, and the inability to account for the effect of health factors, maternal age, and use of assisted reproductive technology, the researchers noted.
However, the results were strengthened by the large sample size and 20-year study period, as well as the stratification of labor arrest by stage, and suggest uptake of newer recommendations, they said. “Future reductions in cesarean deliveries among patients at low risk for cesarean delivery may be dependent on improved assessment of intrapartum fetal status,” they concluded.
Consider populations and outcomes in cesarean risk assessment
The decreasing rates of cesarean deliveries in the current study can be seen as positive, but more research is needed to examine maternal and neonatal outcomes, and to consider other conditions that affect risk for cesarean delivery, Paolo Ivo Cavoretto, MD, and Massimo Candiani, MD, of IRCCS San Raffaele Scientific Institute, and Antonio Farina, MD, of the University of Bologna, Italy, wrote in an accompanying editorial.
Notably, the study authors identified a population aged 15-39 years as low risk, and an increased risk for cesarean delivery within this range increased with age. “Maternal age remains a major risk factor associated with the risk of cesarean delivery, both from results of this study and those of previous analyses assessing its independence from other related risk factors,” the editorialists said.
The study findings also reflect the changes in standards for labor duration during the study period, they noted. The longer duration of labor may reduce cesarean delivery rates, but it is not without maternal and fetal-neonatal risks, they wrote.
“To be sure that the described trend of cesarean delivery rate reduction can be considered positive, there would be the theoretical need to analyze other maternal-fetal-neonatal outcomes (e.g., rates of operative deliveries, neonatal acidemia, intensive care unit use, maternal hemorrhage, pelvic floor trauma and dysfunction, and psychological distress),” the editorialists concluded.
More research needed to explore clinical decisions
“Reducing the cesarean delivery rate is a top priority, but evidence is lacking on an optimal rate that improves maternal and neonatal outcomes,” Iris Krishna, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Hospital quality and safety committees have been working to decrease cesarean deliveries amongst low-risk women, and identifying contemporary trends gives us insight on whether some of these efforts have translated to a lower cesarean delivery rate,” she said.
Dr. Krishna said she was not surprised by the higher cesarean section rate in the South. “The decision for cesarean delivery is multifaceted, and although this study was not able to assess clinical indications for cesarean delivery or maternal and fetal outcomes, we cannot ignore that social determinants of health contribute greatly to overall health outcomes,” she said. The trends in the current study further underscore the geographic disparities in access to health care present in the South, she added.
“This study notes that cesarean delivery for nonreassuring fetal status increased; however, nonreassuring fetal status as an indication for cesarean delivery can be subjective,” Dr. Krishna said. “Hospital quality and safety committees should consider reviewing the clinical scenarios that led to this decision to identify opportunities for improvement and further education,” she said.
“Defining contemporary trends in cesarean delivery for low-risk patients has merit, but the study findings should be interpreted with caution,” said Dr. Krishna, who is a member of the Ob.Gyn. News advisory board. More research is needed to define an optimal cesarean section rate that promotes positive maternal and fetal outcomes, and to determine whether identifying an optimal rate should be based on patient risk profiles, she said.
The study received no outside funding. Lead author Ms. Frappaolo had no financial conflicts to disclose; nor did the editorial authors or Dr. Krishna.
Although clinically indicated cesarean deliveries may improve outcomes for mothers and infants, “when not clinically indicated, cesarean delivery is a major surgical intervention that increases risk for adverse outcomes,” wrote Anna M. Frappaolo of Columbia University College of Physicians and Surgeons, New York, and colleagues.
The Healthy People 2030 campaign includes the reduction of cesarean deliveries, but trends in these procedures, especially with regard to diagnoses of labor arrest, have not been well studied, the researchers said.
In an analysis published in JAMA Network Open, the researchers reviewed delivery hospitalizations using data from the National Inpatient Sample from 2000 to 2019.
Births deemed low risk for cesarean delivery were identified by using criteria of the Society for Maternal-Fetal Medicine and additional criteria, and joinpoint regression analysis was used to estimate changes.
The researchers examined overall trends in cesarean deliveries as well as trends for three specific diagnoses: nonreassuring fetal status, labor arrest, and obstructed labor.
The final analysis included 40,517,867 deliveries; of these, 4,885,716 (12.1%) were cesarean deliveries.
Overall, cesarean deliveries in patients deemed at low risk increased from 9.7% in 2000 to 13.9% in 2009, then plateaued and decreased from 13.0% in 2012 to 11.1% in 2019. The average annual percentage change (AAPC) for cesarean delivery was 6.4% for the years from 2000 to 2005, 1.2% from 2005 to 2009, and −2.2% from 2009 to 2019.
Cesarean delivery for nonreassuring fetal status increased over the entire study period, from 3.4% in 2000 to 5.1% in 2019. By contrast, overall cesarean delivery for labor arrest increased from 3.6% in 2000 to a high of 4.8% in 2009, then decreased to 2.7% in 2019. Cesarean deliveries with a diagnosis of obstructed labor decreased from 0.9% in 2008 to 0.3% in 2019.
More specifically, cesarean deliveries for labor arrest in the active phase, latent phase, and second stage of labor increased from 1.5% to 2.1%, 1.1% to 1.5%, and 0.9% to 1.3%, respectively, from 2000 to 2009, and decreased from 2.1% to 1.7% for the active phase, from 1.5% to 1.2% for the latent phase, and from 1.2% to 0.9% for the second stage between 2010 and 2019.
Patients with increased odds of cesarean delivery were older (aged 35-39 years vs. 25-29 years, adjusted odds ratio 1.27), delivered in a hospital in the South vs. the Northeast of the United States (aOR 1.11), and were more likely to be non-Hispanic Black vs. non-Hispanic White (OR 1.23).
Notably, changes in nomenclature and interpretation of intrapartum electronic fetal heart monitoring occurred during the study period, with recommendations for the adoption of a three-tiered system for fetal heart rate patterns in 2008. “It is possible that current evidence and nomenclature related to intrapartum FHR interpretation may result in identification of a larger number of fetuses deemed at indeterminate risk for abnormal acid-base status,” the researchers wrote in their discussion.
The study findings were limited by several factors including the use of administrative discharge data rather than clinical records, the exclusion of patients with chronic conditions associated with cesarean delivery, changes in billing codes during the study period, and the inability to account for the effect of health factors, maternal age, and use of assisted reproductive technology, the researchers noted.
However, the results were strengthened by the large sample size and 20-year study period, as well as the stratification of labor arrest by stage, and suggest uptake of newer recommendations, they said. “Future reductions in cesarean deliveries among patients at low risk for cesarean delivery may be dependent on improved assessment of intrapartum fetal status,” they concluded.
Consider populations and outcomes in cesarean risk assessment
The decreasing rates of cesarean deliveries in the current study can be seen as positive, but more research is needed to examine maternal and neonatal outcomes, and to consider other conditions that affect risk for cesarean delivery, Paolo Ivo Cavoretto, MD, and Massimo Candiani, MD, of IRCCS San Raffaele Scientific Institute, and Antonio Farina, MD, of the University of Bologna, Italy, wrote in an accompanying editorial.
Notably, the study authors identified a population aged 15-39 years as low risk, and an increased risk for cesarean delivery within this range increased with age. “Maternal age remains a major risk factor associated with the risk of cesarean delivery, both from results of this study and those of previous analyses assessing its independence from other related risk factors,” the editorialists said.
The study findings also reflect the changes in standards for labor duration during the study period, they noted. The longer duration of labor may reduce cesarean delivery rates, but it is not without maternal and fetal-neonatal risks, they wrote.
“To be sure that the described trend of cesarean delivery rate reduction can be considered positive, there would be the theoretical need to analyze other maternal-fetal-neonatal outcomes (e.g., rates of operative deliveries, neonatal acidemia, intensive care unit use, maternal hemorrhage, pelvic floor trauma and dysfunction, and psychological distress),” the editorialists concluded.
More research needed to explore clinical decisions
“Reducing the cesarean delivery rate is a top priority, but evidence is lacking on an optimal rate that improves maternal and neonatal outcomes,” Iris Krishna, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
“Hospital quality and safety committees have been working to decrease cesarean deliveries amongst low-risk women, and identifying contemporary trends gives us insight on whether some of these efforts have translated to a lower cesarean delivery rate,” she said.
Dr. Krishna said she was not surprised by the higher cesarean section rate in the South. “The decision for cesarean delivery is multifaceted, and although this study was not able to assess clinical indications for cesarean delivery or maternal and fetal outcomes, we cannot ignore that social determinants of health contribute greatly to overall health outcomes,” she said. The trends in the current study further underscore the geographic disparities in access to health care present in the South, she added.
“This study notes that cesarean delivery for nonreassuring fetal status increased; however, nonreassuring fetal status as an indication for cesarean delivery can be subjective,” Dr. Krishna said. “Hospital quality and safety committees should consider reviewing the clinical scenarios that led to this decision to identify opportunities for improvement and further education,” she said.
“Defining contemporary trends in cesarean delivery for low-risk patients has merit, but the study findings should be interpreted with caution,” said Dr. Krishna, who is a member of the Ob.Gyn. News advisory board. More research is needed to define an optimal cesarean section rate that promotes positive maternal and fetal outcomes, and to determine whether identifying an optimal rate should be based on patient risk profiles, she said.
The study received no outside funding. Lead author Ms. Frappaolo had no financial conflicts to disclose; nor did the editorial authors or Dr. Krishna.
FROM JAMA NETWORK OPEN
Meet the JCOM Author with Dr. Barkoudah: A Multidisciplinary Team–Based Clinical Care Pathway for Infective Endocarditis
Luxe vacations, private jets: Medical device maker, surgeon to pay $46 million penalty in kickback scheme
according to experts familiar with the federal Anti-Kickback Statute.
Historically, enforcement actions have primarily focused on the person or organization offering the perks – and not necessarily the physicians accepting it, Steven W. Ortquist, founder and principal of Arete Compliance Solutions, LLC, in Phoenix, told this news organization.
But that’s changing.
“In recent years, we are seeing a trend toward holding physicians and others on the receiving end of the inducement accountable as well,” said Mr. Ortquist, who is a past board member and president of the Health Care Compliance Association. He noted that authorities usually pursue the inducing company first before moving on to individual clinicians or practices.
The Department of Justice followed a similar pattern in a recently announced kickback settlement that ensnared an intraocular lens distributor, an ophthalmology equipment supplier, two CEOs, and a surgeon. Precision Lens must pay more than $43 million for offering high-end vacations and other expensive perks to surgeons who used its cataract products.
The verdict marks the end of a 6-week civil jury trial, where evidence emerged that Paul Ehlen, owner of Precision Lens and its parent company, Cameron-Ehlen Group, maintained a secret “slush fund” for paying kickbacks to ophthalmic surgeons. The inducement scheme netted the Minnesota-based company millions in sales and led to the submission of 64,575 false Medicare claims from 2006 to 2015, a violation of the Anti-Kickback Statute and the False Claims Act.
According to court documents, physicians received luxury travel and entertainment packages, including skiing, fishing, and golfing excursions at exclusive destinations, often traveling via private jet to attend Broadway musicals and major sporting events. Mr. Ehlen and company representatives also sold frequent flyer miles to physicians at a steep discount, allowing them to take personal and business trips below fair market value.
Federal authorities initially announced an investigation into the business practices of Precision Lens in 2017 after receiving a whistleblower complaint from Kipp Fesenmaier, a former executive at Sightpath Medical, an ophthalmology supplier and “corporate partner” of Precision Lens. Mr. Fesenmaier alleged that both companies were involved in an inducement scheme.
Sightpath Medical and its CEO, James Tiffany, agreed to a $12 million settlement to resolve the kickback allegations.
The Department of Justice subsequently investigated Jitendra Swarup, MD, an ophthalmologist and cataract surgeon who allegedly received “unlawful remuneration from Sightpath, Precision, and Ehlen” and filed false insurance claims. In addition to accepting expensive hunting and fishing trips from the medical device companies, Dr. Swarup was paid more than $100,000 per year for consulting services he did not fully render.
Dr. Swarup agreed to a nearly $3 million settlement and participation in a 3-year corporate integrity agreement with the Office of Inspector General. In exchange for compliance with such contracts, the OIG permits physicians to continue participating in Medicare, Medicaid, and other federal health care programs.
In a statement from attorneys, Precision Lens and Mr. Ehlen pledged to appeal the verdict and “defend ... our wholly appropriate actions” while remaining focused on their commitment to health care clinicians and manufacturers.
‘Endless’ opportunities for inducement
Unfortunately, opportunities for inducement are “endless,” experts say. Extravagant trips, dinners, and gifts can trigger a violation, but so can nearly anything of value.
Just last year, Biotronik reached a $12.95 million settlement amid allegations that company representatives wined and dined physicians to induce their use of its pacemakers and defibrillators. To date, no physicians have been charged.
But after a record-breaking number of whistleblower judgments last fiscal year totaling more than $2 billion, physicians should take note, Radha Bhatnagar, Esq, director of compliance at The CM Group, told the news organization.
“When manufacturers offer physicians kickbacks with the added element of fraudulent Medicare or Medicaid reimbursements, that is typically when manufacturers and individuals face civil and criminal liability,” said Ms. Bhatnagar, something the Department of Justice alluded to when announcing a settlement involving 15 Texas physicians last year.
In another case, Kingsley R. Chin, an orthopedic surgeon and designer of a spinal implant, was indicted in 2021 for paying millions of dollars in sham consulting fees to physicians who used his products. At least six surgeons who accepted money from Dr. Chin were later named in a civil case and ordered to pay $3.3 million in penalties.
Jason Montone, DO, an orthopedic surgeon who accepted the illicit payments, agreed to a plea deal with a reduced prison sentence, 1 year of supervised release, and a fine of $379,000.
Although Dr. Chin’s sentencing hasn’t been announced, violating kickback laws can result in a sentence of up to 10 years.
A version of this article originally appeared on Medscape.com.
according to experts familiar with the federal Anti-Kickback Statute.
Historically, enforcement actions have primarily focused on the person or organization offering the perks – and not necessarily the physicians accepting it, Steven W. Ortquist, founder and principal of Arete Compliance Solutions, LLC, in Phoenix, told this news organization.
But that’s changing.
“In recent years, we are seeing a trend toward holding physicians and others on the receiving end of the inducement accountable as well,” said Mr. Ortquist, who is a past board member and president of the Health Care Compliance Association. He noted that authorities usually pursue the inducing company first before moving on to individual clinicians or practices.
The Department of Justice followed a similar pattern in a recently announced kickback settlement that ensnared an intraocular lens distributor, an ophthalmology equipment supplier, two CEOs, and a surgeon. Precision Lens must pay more than $43 million for offering high-end vacations and other expensive perks to surgeons who used its cataract products.
The verdict marks the end of a 6-week civil jury trial, where evidence emerged that Paul Ehlen, owner of Precision Lens and its parent company, Cameron-Ehlen Group, maintained a secret “slush fund” for paying kickbacks to ophthalmic surgeons. The inducement scheme netted the Minnesota-based company millions in sales and led to the submission of 64,575 false Medicare claims from 2006 to 2015, a violation of the Anti-Kickback Statute and the False Claims Act.
According to court documents, physicians received luxury travel and entertainment packages, including skiing, fishing, and golfing excursions at exclusive destinations, often traveling via private jet to attend Broadway musicals and major sporting events. Mr. Ehlen and company representatives also sold frequent flyer miles to physicians at a steep discount, allowing them to take personal and business trips below fair market value.
Federal authorities initially announced an investigation into the business practices of Precision Lens in 2017 after receiving a whistleblower complaint from Kipp Fesenmaier, a former executive at Sightpath Medical, an ophthalmology supplier and “corporate partner” of Precision Lens. Mr. Fesenmaier alleged that both companies were involved in an inducement scheme.
Sightpath Medical and its CEO, James Tiffany, agreed to a $12 million settlement to resolve the kickback allegations.
The Department of Justice subsequently investigated Jitendra Swarup, MD, an ophthalmologist and cataract surgeon who allegedly received “unlawful remuneration from Sightpath, Precision, and Ehlen” and filed false insurance claims. In addition to accepting expensive hunting and fishing trips from the medical device companies, Dr. Swarup was paid more than $100,000 per year for consulting services he did not fully render.
Dr. Swarup agreed to a nearly $3 million settlement and participation in a 3-year corporate integrity agreement with the Office of Inspector General. In exchange for compliance with such contracts, the OIG permits physicians to continue participating in Medicare, Medicaid, and other federal health care programs.
In a statement from attorneys, Precision Lens and Mr. Ehlen pledged to appeal the verdict and “defend ... our wholly appropriate actions” while remaining focused on their commitment to health care clinicians and manufacturers.
‘Endless’ opportunities for inducement
Unfortunately, opportunities for inducement are “endless,” experts say. Extravagant trips, dinners, and gifts can trigger a violation, but so can nearly anything of value.
Just last year, Biotronik reached a $12.95 million settlement amid allegations that company representatives wined and dined physicians to induce their use of its pacemakers and defibrillators. To date, no physicians have been charged.
But after a record-breaking number of whistleblower judgments last fiscal year totaling more than $2 billion, physicians should take note, Radha Bhatnagar, Esq, director of compliance at The CM Group, told the news organization.
“When manufacturers offer physicians kickbacks with the added element of fraudulent Medicare or Medicaid reimbursements, that is typically when manufacturers and individuals face civil and criminal liability,” said Ms. Bhatnagar, something the Department of Justice alluded to when announcing a settlement involving 15 Texas physicians last year.
In another case, Kingsley R. Chin, an orthopedic surgeon and designer of a spinal implant, was indicted in 2021 for paying millions of dollars in sham consulting fees to physicians who used his products. At least six surgeons who accepted money from Dr. Chin were later named in a civil case and ordered to pay $3.3 million in penalties.
Jason Montone, DO, an orthopedic surgeon who accepted the illicit payments, agreed to a plea deal with a reduced prison sentence, 1 year of supervised release, and a fine of $379,000.
Although Dr. Chin’s sentencing hasn’t been announced, violating kickback laws can result in a sentence of up to 10 years.
A version of this article originally appeared on Medscape.com.
according to experts familiar with the federal Anti-Kickback Statute.
Historically, enforcement actions have primarily focused on the person or organization offering the perks – and not necessarily the physicians accepting it, Steven W. Ortquist, founder and principal of Arete Compliance Solutions, LLC, in Phoenix, told this news organization.
But that’s changing.
“In recent years, we are seeing a trend toward holding physicians and others on the receiving end of the inducement accountable as well,” said Mr. Ortquist, who is a past board member and president of the Health Care Compliance Association. He noted that authorities usually pursue the inducing company first before moving on to individual clinicians or practices.
The Department of Justice followed a similar pattern in a recently announced kickback settlement that ensnared an intraocular lens distributor, an ophthalmology equipment supplier, two CEOs, and a surgeon. Precision Lens must pay more than $43 million for offering high-end vacations and other expensive perks to surgeons who used its cataract products.
The verdict marks the end of a 6-week civil jury trial, where evidence emerged that Paul Ehlen, owner of Precision Lens and its parent company, Cameron-Ehlen Group, maintained a secret “slush fund” for paying kickbacks to ophthalmic surgeons. The inducement scheme netted the Minnesota-based company millions in sales and led to the submission of 64,575 false Medicare claims from 2006 to 2015, a violation of the Anti-Kickback Statute and the False Claims Act.
According to court documents, physicians received luxury travel and entertainment packages, including skiing, fishing, and golfing excursions at exclusive destinations, often traveling via private jet to attend Broadway musicals and major sporting events. Mr. Ehlen and company representatives also sold frequent flyer miles to physicians at a steep discount, allowing them to take personal and business trips below fair market value.
Federal authorities initially announced an investigation into the business practices of Precision Lens in 2017 after receiving a whistleblower complaint from Kipp Fesenmaier, a former executive at Sightpath Medical, an ophthalmology supplier and “corporate partner” of Precision Lens. Mr. Fesenmaier alleged that both companies were involved in an inducement scheme.
Sightpath Medical and its CEO, James Tiffany, agreed to a $12 million settlement to resolve the kickback allegations.
The Department of Justice subsequently investigated Jitendra Swarup, MD, an ophthalmologist and cataract surgeon who allegedly received “unlawful remuneration from Sightpath, Precision, and Ehlen” and filed false insurance claims. In addition to accepting expensive hunting and fishing trips from the medical device companies, Dr. Swarup was paid more than $100,000 per year for consulting services he did not fully render.
Dr. Swarup agreed to a nearly $3 million settlement and participation in a 3-year corporate integrity agreement with the Office of Inspector General. In exchange for compliance with such contracts, the OIG permits physicians to continue participating in Medicare, Medicaid, and other federal health care programs.
In a statement from attorneys, Precision Lens and Mr. Ehlen pledged to appeal the verdict and “defend ... our wholly appropriate actions” while remaining focused on their commitment to health care clinicians and manufacturers.
‘Endless’ opportunities for inducement
Unfortunately, opportunities for inducement are “endless,” experts say. Extravagant trips, dinners, and gifts can trigger a violation, but so can nearly anything of value.
Just last year, Biotronik reached a $12.95 million settlement amid allegations that company representatives wined and dined physicians to induce their use of its pacemakers and defibrillators. To date, no physicians have been charged.
But after a record-breaking number of whistleblower judgments last fiscal year totaling more than $2 billion, physicians should take note, Radha Bhatnagar, Esq, director of compliance at The CM Group, told the news organization.
“When manufacturers offer physicians kickbacks with the added element of fraudulent Medicare or Medicaid reimbursements, that is typically when manufacturers and individuals face civil and criminal liability,” said Ms. Bhatnagar, something the Department of Justice alluded to when announcing a settlement involving 15 Texas physicians last year.
In another case, Kingsley R. Chin, an orthopedic surgeon and designer of a spinal implant, was indicted in 2021 for paying millions of dollars in sham consulting fees to physicians who used his products. At least six surgeons who accepted money from Dr. Chin were later named in a civil case and ordered to pay $3.3 million in penalties.
Jason Montone, DO, an orthopedic surgeon who accepted the illicit payments, agreed to a plea deal with a reduced prison sentence, 1 year of supervised release, and a fine of $379,000.
Although Dr. Chin’s sentencing hasn’t been announced, violating kickback laws can result in a sentence of up to 10 years.
A version of this article originally appeared on Medscape.com.
Does new heart transplant method challenge definition of death?
The relatively recent innovation of heart transplantation after circulatory death of the donor is increasing the number of donor hearts available and leading to many more lives on the heart transplant waiting list being saved. Experts agree it’s a major and very welcome advance in medicine.
However, some of the processes involved in one approach to donation after circulatory death has raised ethical concerns and questions about whether they violate the “dead donor rule” – a principle that requires patients be declared dead before removal of life-sustaining organs for transplant.
Experts in the fields of transplantation and medical ethics have yet to reach consensus, causing problems for the transplant community, who worry that this could cause a loss of confidence in the entire transplant process.
A new pathway for heart transplantation
The traditional approach to transplantation is to retrieve organs from a donor who has been declared brain dead, known as “donation after brain death (DBD).” These patients have usually suffered a catastrophic brain injury but survived to get to intensive care.
As the brain swells because of injury, it becomes evident that all brain function is lost, and the patient is declared brain dead. However, breathing is maintained by the ventilator and the heart is still beating. Because the organs are being oxygenated, there is no immediate rush to retrieve the organs and the heart can be evaluated for its suitability for transplant in a calm and methodical way before it is removed.
However, there is a massive shortage of organs, especially hearts, partially because of the limited number of donors who are declared brain dead in that setting.
In recent years, another pathway for organ transplantation has become available: “donation after circulatory death (DCD).” These patients also have suffered a catastrophic brain injury considered to be nonsurvivable, but unlike the DBD situation, the brain still has some function, so the patient does not meet the criteria for brain death.
Still, because the patient is considered to have no chance of a meaningful recovery, the family often recognizes the futility of treatment and agrees to the withdrawal of life support. When this happens, the heart normally stops beating after a period of time. There is then a “stand-off time” – normally 5 minutes – after which death is declared and the organs can be removed.
The difficulty with this approach, however, is that because the heart has been stopped, it has been deprived of oxygen, potentially causing injury. While DCD has been practiced for several years to retrieve organs such as the kidney, liver, lungs, and pancreas, the heart is more difficult as it is more susceptible to oxygen deprivation. And for the heart to be assessed for transplant suitability, it should ideally be beating, so it has to be reperfused and restarted quickly after death has been declared.
For many years it was thought the oxygen deprivation that occurs after circulatory death would be too much to provide a functional organ. But researchers in the United Kingdom and Australia developed techniques to overcome this problem, and early DCD heart transplants took place in 2014 in Australia, and in 2015 in the United Kingdom.
Heart transplantation after circulatory death has now become a routine part of the transplant program in many countries, including the United States, Spain, Belgium, the Netherlands, and Austria.
In the United States, 348 DCD heart transplants were performed in 2022, with numbers expected to reach 700 to 800 this year as more centers come online.
It is expected that most countries with heart transplant programs will follow suit and the number of donor hearts will increase by up to 30% worldwide because of DCD.
Currently, there are about 8,000 heart transplants worldwide each year and with DCD this could rise to about 10,000, potentially an extra 2,000 lives saved each year, experts estimate.
Two different approaches to DCD heart transplantation have been developed.
The direct procurement approach
The Australian group, based at St. Vincent’s Hospital in Sydney, developed a technique referred to as “direct procurement”: after the standoff period and declaration of circulatory death, the chest is opened, and the heart is removed. New technology, the Organ Care System (OCS) heart box (Transmedics), is then used to reperfuse and restart the heart outside the body so its suitability for transplant can be assessed.
The heart is kept perfused and beating in the OCS box while it is being transported to the recipient. This has enabled longer transit times than the traditional way of transporting the nonbeating heart on ice.
Peter MacDonald, MD, PhD, from the St Vincent’s group that developed this approach, said, “Most people thought a heart from a DCD donor would not survive transport – that the injury to the heart from the combination of life support withdrawal, stand-off time, and cold storage would be too much. But we modeled the process in the lab and were able to show that we were able to get the heart beating again after withdrawal of life support.”
Dr. McDonald noted that “the recipient of their first human DCD heart transplant using this machine in 2014 is still alive and well.” The Australian group has now done 85 of these DCD heart transplants, and they have increased the number of heart transplant procedures at St. Vincent’s Hospital by 25%.
Normothermic regional perfusion (NRP)
The U.K. group, based at the Royal Papworth Hospital in Cambridge, England, developed a different approach to DCD: After the standoff period and the declaration of circulatory death, the donor is connected to a heart/lung machine using extracorporeal membrane oxygenation (ECMO) so that the heart is perfused and starts beating again inside the body. This approach is known as normothermic regional perfusion (NRP).
Marius Berman, MD, surgical lead for Transplantation and Mechanical Circulatory Support at Papworth, explained that the NRP approach allows the heart to be perfused and restarted faster than direct procurement, resulting in a shorter ischemic time. The heart can be evaluated thoroughly for suitability for transplantation in situ before committing to transplantation, and because the heart is less damaged, it can be transported on ice without use of the OCS box.
“DCD is more complicated than DBD, because the heart has stopped and has to be restarted. Retrieval teams have to be very experienced,” Dr. Berman noted. “This is more of an issue for the direct procurement approach, where the chest has to be opened and the heart retrieved as fast as possible. It is a rush. The longer time without the heart being perfused correlates to an increased incidence of primary graft dysfunction. With NRP, we can get the heart started again more quickly, which is crucial.”
Stephen Large, MBBS, another cardiothoracic surgeon with the Papworth team, added that they have reduced ischemic time to about 15 minutes. “That’s considerably shorter than reperfusing the heart outside the body,” he said. “This results in a healthier organ for the recipient.”
The NRP approach is also less expensive than direct procurement as one OCS box costs about $75,000.
He pointed out that the NRP approach can also be used for heart transplants in children and even small babies, while currently the direct procurement technique is not typically suitable for children because the OCS box was not designed for small hearts.
DCD, using either technique, has increased the heart transplant rate by 40% at Papworth, and is being used at all seven transplant centers in the United Kingdom, “a world first,” noted Dr. Large.
The Papworth team recently published its 5-year experience with 25 NRP transplants and 85 direct procurement transplants. Survival in recipients was no different, although there was some suggestion that the NRP hearts may have been in slightly better condition, possibly being more resistant to immunological rejection.
Ethical concerns about NRP
Restarting the circulation during the NRP process has raised ethical concerns.
When the NRP technique was first used in the United States, these ethical questions were raised by several groups, including the American College of Physicians (ACP).
Harry Peled, MD, Providence St. Jude Medical Center, Fullerton, Calif., coauthor of a recent Viewpoint on the issue, is board-certified in both cardiology and critical care, and said he is a supporter of DCD using direct procurement, but he does not believe that NRP is ethical at present. He is not part of the ACP, but said his views align with those of the organization.
There are two ethical problems with NRP, he said. The first is whether by restarting the circulation, the NRP process violates the U.S. definition of death, and retrieval of organs would therefore violate the dead donor rule.
“American law states that death is the irreversible cessation of brain function or of circulatory function. But with NRP, the circulation is artificially restored, so the cessation of circulatory function is not irreversible,” Dr. Peled pointed out.
“I have no problem with DCD using direct procurement as we are not restarting the circulation. But NRP is restarting the circulation and that is a problem for me,” Dr. Peled said. “I would argue that by performing NRP, we are resuscitating the patient.”
The second ethical problem with NRP is concern about whether, during the process, there would be any circulation to the brain, and if so, would this be enough to restore some brain function? Before NRP is started, the main arch vessel arteries to the head are clamped to prevent flow to the brain, but there are worries that some blood flow may still be possible through small collateral vessels.
“We have established that these patients do not have enough brain function for a meaningful life, which is why a decision has been made to remove life support, but they have not been declared brain dead,” Dr. Peled said.
With direct procurement, the circulation is not restarted so there is no chance that any brain function will be restored, he said. “But with NRP, because the arch vessels have to be clamped to prevent brain circulation, that is admitting there is concern that brain function may be restored if circulation to the brain is reestablished, and brain function is compatible with life. As we do not know whether there is any meaningful circulation to the brain via the small collaterals, there is, in effect, a risk of bringing the patient back to life.”
The other major concern for some is whether even a very small amount of circulation to the brain would be enough to support consciousness, and “we don’t know that for certain,” Dr. Peled said.
The argument for NRP
Nader Moazami, MD, professor of cardiovascular surgery, NYU Langone Health, New York, is one of the more vocal proponents of NRP for DCD heart transplantation in the United States, and has coauthored responses to these ethical concerns.
“People are confusing many issues to produce an argument against NRP,” he said.
“Our position is that death has already been declared based on the lack of circulatory function for over 5 minutes and this has been with the full agreement of the family, knowing that the patient has no chance of a meaningful life. No one is thinking of trying to resuscitate the patient. It has already been established that any future efforts to resuscitate are futile. In this case, we are not resuscitating the patient by restarting the circulation. It is just regional perfusion of the organs.”
Dr. Moazami pointed out this concept was accepted for the practice of abdominal DCD when it first started in the United States in the 1990s where cold perfusion was used to preserve the abdominal organs before they were retrieved from the body.
“The new approach of using NRP is similar except that it involves circulating warm blood, which will preserve organs better and result in higher quality organs for the recipient.”
On the issue of concern about possible circulation to the brain, Dr. Moazami said: “The ethical critics of NRP are questioning whether the brain may not be dead. We are arguing that the patient has already been declared dead as they have had a circulatory death. You cannot die twice.”
He maintained that the clamping of the arch vessels to the head will ensure that when the circulation is restarted “the natural process of circulatory death leading to brain death will continue to progress.”
On the concerns about possible collateral flow to the brain, Dr. Moazami said there is no evidence that this occurs. “Prominent neurologists have said it is impossible for collaterals to provide any meaningful blood flow to the brain in this situation. And even if there is small amount of blood flow to the brain, this would be insufficient to maintain any meaningful brain function.”
But Dr. Peled argues that this has not been proved. “Even though we don’t think there is enough circulation to the brain for any function with NRP, we don’t know that with 100% certainty,” he said. “In my view, if there is a possibility of even the smallest amount of brain flow, we are going against the dead donor rule. We are rewriting the rules of death.”
Dr. Moazami countered: “Nothing in life is 100%, particularly in medicine. With that argument can you also prove with 100% certainty to me that there is absolutely no brain function with regular direct procurement DCD? We know that brain death has started, but the question is: Has it been completed? We don’t know the answer to this question with 100% certainty, but that is the case for regular direct procurement DCD as well, and that has been accepted by almost everyone.
“The whole issue revolves around when are we comfortable that death has occurred,” he said. “Those against NRP are concerned that organs are being taken before the patient is dead. But the key point is that the patient has already been declared dead.”
Since there is some concern over the ethics of NRP, why not just stick to DCD with direct procurement?
Dr. Moazami argued that NRP results in healthier organs. “NRP allows more successful heart transplants, liver transplants, lung transplants. It preserves all the organs better,” he said. “This will have a big impact on recipients – they would obviously much prefer a healthier organ. In addition, the process is easier and cheaper, so more centers will be able to do it, therefore more transplants will get done and more lives will be saved if NRP is used.”
He added: “I am a physician taking care of sick patients. I believe I have to respect the wishes of the donor and the donor family; make sure I’m not doing any harm to the donor; and ensure the best quality possible of the organ I am retrieving to best serve the recipient. I am happy I am doing this by using NRP for DCD heart transplantation.”
But Dr. Peled argued that while NRP may have some possible advantages over direct procurement, that does not justify allowing a process to go ahead that is unethical.
“The fact that NRP may result in some benefits doesn’t justify violating the dead donor rule or the possibility, however small, of causing pain to the donor. If it’s unethical, it’s unethical. Full stop,” he said.
“I feel that NRP is not respecting the rights of our patients and that the process does not have adequate transparency. We took it to our local ethics committee, and they decided not to approve NRP in our health care system. I agree with this decision,” Dr. Peled said.
“The trouble is different experts and different countries are not in agreement about this,” he added. “Reasonable, well-informed people are in disagreement. I do not believe we can have a standard of care where there is not consensus.”
Cautious nod
In a 2022 consensus statement, the International Society for Heart and Lung Transplantation (ISHLT) gave a cautious nod toward DCD and NRP, dependent on local recommendations.
The ISHLT conclusion reads: “With appropriate consideration of the ethical principles involved in organ donation, DCD can be undertaken in a morally permissible manner. In all cases, the introduction of DCD programs should be in accordance with local legal regulations. Countries lacking a DCD pathway should be encouraged to develop national ethical, professional, and legal frameworks to address both public and professional concerns.”
The author of a recent editorial on the subject, Ulrich P. Jorde, MD, head of the heart transplant program at Montefiore Medical Center, New York, said, “DCD is a great step forward. People regularly die on the heart transplant waiting list. DCD will increase the supply of donor hearts by 20% to 30%.”
However, he noted that while most societies have agreed on a protocol for organ donation based on brain death, the situation is more complicated with circulatory death.
“Different countries have different definitions of circulatory death. How long do we have to wait after the heart has stopped beating before the patient is declared dead? Most countries have agreed on 5 minutes, but other countries have imposed different periods and as such, different definitions of death.
“The ISHLT statement says that restarting the circulation is acceptable if death has been certified according to prevailing law and surgical interventions are undertaken to preclude any restoration of cerebral circulation. But our problem is that different regional societies have different definitions of circulatory, death which makes the situation confusing.”
Dr. Jorde added: “We also have to weigh the wishes of the donor and their family. If family, advocating what are presumed to be the donor’s wishes, have decided that DCD would be acceptable and they understand the concept and wish to donate the organs after circulatory death, this should be strongly considered under the concept of self-determination, a basic human right.”
Variations in practice around the world
This ethical debate has led to large variations in practice around the world, with some countries, such as Spain, allowing both methods of DCD, while Australia allows direct procurement but not NRP, and Germany currently does not allow DCD at all.
In the United States, things are even more complicated, with some states allowing NRP while others don’t. Even within states, some hospitals and transplant organizations allow NRP, and others don’t.
David A. D’Alessandro, MD, cardiac surgeon at Massachusetts General Hospital, Boston, uses only the direct procurement approach as his region does not allow NRP.
“The direct procurement approach is not controversial and to me that’s a big advantage. I believe we need to agree on the ethics first, and then get into a debate about which technique is better,” he told this news organization.
Dr. D’Alessandro and his group recently published the results of their study, with direct procurement DCD heart transplantation showing similar short-term clinical outcomes to DBD.
“We are only doing direct procurement and we are seeing good results that appear to be comparable to DBD. That is good enough for me,” he said.
Dr. D’Alessandro estimates that in the United States both types of DCD procedures are currently being done about equally.
“Anything we can do to increase the amount of hearts available for transplantation is a big deal,” he said. “At the moment, only the very sickest patients get a heart transplant, and many patients die on the transplant waiting list. Very sadly, many young people die every year from a circulatory death after having life support withdrawn. Before DCD, these beautiful functional organs were not able to be used. Now we have a way of saving lives with these organs.”
Dr. D’Alessandro noted that more and more centers in the United States are starting to perform DCD heart transplants.
“Not every transplant center may join in as the DCD procedures are very resource-intensive and time-consuming. For low-volume transplant centers, it may not be worth the expense and anguish to do DCD heart transplants. But bigger centers will need to engage in DCD to remain competitive. My guess is that 50%-70% of U.S. transplant centers will do DCD in future.”
He said he thinks it is a “medical shortcoming” that agreement cannot be reached on the ethics of NRP. “In an ideal world everyone would be on the same page. It makes me a bit uncomfortable that some people think it’s okay and some people don’t.”
Adam DeVore, MD, a cardiologist at Duke University Medical Center, Durham, N.C., the first U.S. center to perform an adult DCD heart transplant, reported that his institution uses both methods, with the choice sometimes depending on how far the heart must travel.
“If the recipient is near, NRP may be chosen as the heart is transported on ice, but if it needs to go further away we are more likely to choose direct procurement and use of the OCS box,” he said.
“I am really proud of what we’ve been able to do, helping to introduce DCD in the U.S.,” Dr. DeVore said. “This is having a massive benefit in increasing the number of hearts for donation with great outcomes.”
But he acknowledged that the whole concept of DCD is somewhat controversial.
“The idea of brain death really came about for the purpose of heart donation. The two things are very intricately tied. Trying to do heart donation without brain death having been declared is foreign to people. Also, in DCD there is the issue of [this]: When life support is removed, how long do we wait before death can be declared? That could be in conflict with how long the organ needs to remain viable. We are going through the process now of looking at these questions. There is a lot of variation in the U.S. about the withdrawal of care and the declaration of death, which is not completely standardized.
“But the concept of circulatory death itself is accepted after the withdrawal of life support. I think it’s the rush to take the organs out that makes it more difficult.”
Dr. DeVore said the field is moving forward now. “As the process has become more common, people have become more comfortable, probably because of the big difference it will make to saving lives. But we do need to try and standardize best practices.”
A recent Canadian review of the ethics of DCD concluded that the direct procurement approach would be in alignment with current medical guidelines, but that further work is required to evaluate the consistency of NRP with current Canadian death determination policy and to ensure the absence of brain perfusion during this process.
In the United Kingdom, the definition of death is brain-based, and brain death is defined on a neurological basis.
Dr. Stephen Large from Papworth explained that this recognizes the presence of brain-stem death through brain stem reflex testing after the withdrawal of life support, cardiorespiratory arrest and 5 further minutes of ischemia. As long as NRP does not restore intracranial (brainstem) perfusion after death has been confirmed, then it is consistent with laws for death determination and therefore both direct procurement and NRP are permissible.
However, the question over possible collateral flow to the brain has led the United Kingdom to pause the NRP technique as routine practice while this is investigated further. So, at the present time, the vast majority of DCD heart transplants are being conducted using the direct procurement approach.
But the United Kingdom is facing the bigger challenge: national funding that will soon end. “The DCD program in the U.K. has been extremely successful, increasing heart transplant rates by up to 28%,” Dr. Berman said. “Everybody wants it to continue. But at present the DCD program only has national funding in the U.K. until March 2023. We don’t know what will happen after that.”
The current model in the United Kingdom consists of three specialized DCD heart retrieval teams, a national protocol of direct organ procurement and delivery of DCD hearts to all seven transplant programs, both adult and pediatric.
If the national funding is not extended, “we will go back to individual hospitals trying to fund their own programs. That will be a serious threat to the program and could result in a large reduction in heart transplants,” said Dr. Berman.
Definition of death
The crux of the issue with regard to NRP seems to be variations in how death is defined and the interpretation of those definitions.
DCD donors will have had many tests indicating severe brain damage, a neurologist will have declared the prognosis is futile, and relatives will have agreed to withdraw life support, Dr. Jorde said. “The heart stops beating, and the stand-off time means that blood flow to the brain ceases completely for at least 5 minutes before circulatory death is declared. This is enough on its own to stop brain function.”
Dr. Large made the point that by the time the circulation is reestablished with NRP, more time has elapsed, and the brain will have been without perfusion for much longer than 5 minutes, so it would be “physiologically almost impossible” for there to be any blood flow to the brain.
“Because these brains are already very damaged before life support was removed, the intracranial pressure is high, which will further discourage blood flow to the brain,” he said. Then the donor goes through a period of anoxic heart arrest, up to 16 minutes at a minimum of no blood supply, enough on its own to stop meaningful brain function.
“It’s asking an awful lot to believe that there might be any brain function left,” he said. “And if, on reestablishing the circulation with NRP, there is any blood in the collaterals, the pressure of such flow is so low it won’t enter the brain.”
Dr. Large also pointed out that the fact that the United Kingdom requires a neurologic definition for brain-stem death makes the process easier.
In Australia, St. Vincent’s cardiologist Dr. MacDonald noted that death is defined as the irreversible cessation of circulation, so the NRP procedure is not allowed.
“With NRP, there is an ethical dilemma over whether the patient has legally died or not. Different countries have different ways of defining death. Perhaps society will have to review of the definition of death,” he suggested. Death is a process, “but for organ donation, we have to choose a moment in time of that process that satisfies everyone – when there is no prospect of recovery of the donor but the organs can still be utilized without harming the donor.”
Dr. MacDonald said the field is in transition. “I don’t want to argue that one technique is better than the other; I think it’s good to have access to both techniques. Anything that will increase the number of transplants we can do is a good thing.”
Collaborative decision
Everyone seems to agree that there should be an effort to try to define death in a uniform way worldwide, and that international, national and local regulations are aligned with each other.
Dr. Jorde said: “It is of critical importance that local guidelines are streamlined, firstly in any one given country and then globally, and these things must be discussed transparently within society with all stakeholders – doctors, patients, citizens.”
Dr. Peled, from Providence St. Jude in California, concurred: “There is the possibility that we could change the definition of death, but that cannot be a decision based solely on transplant organizations. It has to be a collaborative decision with a large input from groups who do not have an interest in the procurement of organs.”
He added: “The dialogue so far has been civil, and everybody is trying to do the right thing. My hope is that as a civilized society we will figure out a way forward. At present, there is significant controversy about NRP, and families need to know that. My main concern is that if there is any lack of transparency in getting informed consent, then this risks people losing trust in the donation system.”
Dr. Moazami, from NYU Langone, said the controversy has cast a cloud over the practice of NRP throughout the world. “We need to get it sorted out.”
He said he believes the way forward is to settle the question of whether there is any meaningful blood flow to the brain with the NRP technique.
“This is where the research has to focus. I believe this concern is hypothetical, but I am happy to do the studies to confirm that. Then, the issue should come to a rest. I think that is the right way forward – to do the studies rather than enforcing a moratorium on the practice because of a hypothetical concern.”
These studies on blood flow to the brain are now getting started in both the United Kingdom and the United States.
The U.K. study is being run by Antonio Rubino, MD, consultant in cardiothoracic anesthesia and intensive care at Papworth Hospital NHS Foundation and clinical lead, organ donation. Dr. Rubino explained that the study will assess cerebral blood flow using CT angiography of the brain. “We hypothesize that this will provide evidence to indicate that brain blood flow is not present during NRP and promote trust in the use of NRP in routine practice,” he said.
Dr. Large said: “Rather than having these tortured arguments, we will do the measurements. For the sake of society in this situation, I think it’s good to stop and take a breath. We must measure this, and we are doing just that.”
If there is any blood flow at all, Dr. Large said they will then have to seek expert guidance. “Say we find there is 50 mL of blood flow and normal blood flow is 1,500 mL/min. We will need expert guidance on whether it is remotely possible to be sentient on that. I would say it would be extraordinarily unlikely.”
Dr. Berman summarized the situation: “DCD is increasing the availability of hearts for transplant. This is saving lives, reducing the number of patients on the waiting list, and reducing hospital stays for patients unable to leave the hospital without a transplant. It is definitely here to stay. It is crucial that it gets funded properly, and it is also crucial that we resolve the NRP ethical issues as soon as possible.”
He is hopeful that some of these issues will be resolved this year.
Dr. MacDonald reported he has received “in-kind” support from Transmedics through provision of research modules for preclinical research studies. Dr. D’Alessandro reported he is on the speakers bureau for Abiomed, not relevant to this article. No other relevant disclosures were reported.
A version of this article first appeared on Medscape.com.
The relatively recent innovation of heart transplantation after circulatory death of the donor is increasing the number of donor hearts available and leading to many more lives on the heart transplant waiting list being saved. Experts agree it’s a major and very welcome advance in medicine.
However, some of the processes involved in one approach to donation after circulatory death has raised ethical concerns and questions about whether they violate the “dead donor rule” – a principle that requires patients be declared dead before removal of life-sustaining organs for transplant.
Experts in the fields of transplantation and medical ethics have yet to reach consensus, causing problems for the transplant community, who worry that this could cause a loss of confidence in the entire transplant process.
A new pathway for heart transplantation
The traditional approach to transplantation is to retrieve organs from a donor who has been declared brain dead, known as “donation after brain death (DBD).” These patients have usually suffered a catastrophic brain injury but survived to get to intensive care.
As the brain swells because of injury, it becomes evident that all brain function is lost, and the patient is declared brain dead. However, breathing is maintained by the ventilator and the heart is still beating. Because the organs are being oxygenated, there is no immediate rush to retrieve the organs and the heart can be evaluated for its suitability for transplant in a calm and methodical way before it is removed.
However, there is a massive shortage of organs, especially hearts, partially because of the limited number of donors who are declared brain dead in that setting.
In recent years, another pathway for organ transplantation has become available: “donation after circulatory death (DCD).” These patients also have suffered a catastrophic brain injury considered to be nonsurvivable, but unlike the DBD situation, the brain still has some function, so the patient does not meet the criteria for brain death.
Still, because the patient is considered to have no chance of a meaningful recovery, the family often recognizes the futility of treatment and agrees to the withdrawal of life support. When this happens, the heart normally stops beating after a period of time. There is then a “stand-off time” – normally 5 minutes – after which death is declared and the organs can be removed.
The difficulty with this approach, however, is that because the heart has been stopped, it has been deprived of oxygen, potentially causing injury. While DCD has been practiced for several years to retrieve organs such as the kidney, liver, lungs, and pancreas, the heart is more difficult as it is more susceptible to oxygen deprivation. And for the heart to be assessed for transplant suitability, it should ideally be beating, so it has to be reperfused and restarted quickly after death has been declared.
For many years it was thought the oxygen deprivation that occurs after circulatory death would be too much to provide a functional organ. But researchers in the United Kingdom and Australia developed techniques to overcome this problem, and early DCD heart transplants took place in 2014 in Australia, and in 2015 in the United Kingdom.
Heart transplantation after circulatory death has now become a routine part of the transplant program in many countries, including the United States, Spain, Belgium, the Netherlands, and Austria.
In the United States, 348 DCD heart transplants were performed in 2022, with numbers expected to reach 700 to 800 this year as more centers come online.
It is expected that most countries with heart transplant programs will follow suit and the number of donor hearts will increase by up to 30% worldwide because of DCD.
Currently, there are about 8,000 heart transplants worldwide each year and with DCD this could rise to about 10,000, potentially an extra 2,000 lives saved each year, experts estimate.
Two different approaches to DCD heart transplantation have been developed.
The direct procurement approach
The Australian group, based at St. Vincent’s Hospital in Sydney, developed a technique referred to as “direct procurement”: after the standoff period and declaration of circulatory death, the chest is opened, and the heart is removed. New technology, the Organ Care System (OCS) heart box (Transmedics), is then used to reperfuse and restart the heart outside the body so its suitability for transplant can be assessed.
The heart is kept perfused and beating in the OCS box while it is being transported to the recipient. This has enabled longer transit times than the traditional way of transporting the nonbeating heart on ice.
Peter MacDonald, MD, PhD, from the St Vincent’s group that developed this approach, said, “Most people thought a heart from a DCD donor would not survive transport – that the injury to the heart from the combination of life support withdrawal, stand-off time, and cold storage would be too much. But we modeled the process in the lab and were able to show that we were able to get the heart beating again after withdrawal of life support.”
Dr. McDonald noted that “the recipient of their first human DCD heart transplant using this machine in 2014 is still alive and well.” The Australian group has now done 85 of these DCD heart transplants, and they have increased the number of heart transplant procedures at St. Vincent’s Hospital by 25%.
Normothermic regional perfusion (NRP)
The U.K. group, based at the Royal Papworth Hospital in Cambridge, England, developed a different approach to DCD: After the standoff period and the declaration of circulatory death, the donor is connected to a heart/lung machine using extracorporeal membrane oxygenation (ECMO) so that the heart is perfused and starts beating again inside the body. This approach is known as normothermic regional perfusion (NRP).
Marius Berman, MD, surgical lead for Transplantation and Mechanical Circulatory Support at Papworth, explained that the NRP approach allows the heart to be perfused and restarted faster than direct procurement, resulting in a shorter ischemic time. The heart can be evaluated thoroughly for suitability for transplantation in situ before committing to transplantation, and because the heart is less damaged, it can be transported on ice without use of the OCS box.
“DCD is more complicated than DBD, because the heart has stopped and has to be restarted. Retrieval teams have to be very experienced,” Dr. Berman noted. “This is more of an issue for the direct procurement approach, where the chest has to be opened and the heart retrieved as fast as possible. It is a rush. The longer time without the heart being perfused correlates to an increased incidence of primary graft dysfunction. With NRP, we can get the heart started again more quickly, which is crucial.”
Stephen Large, MBBS, another cardiothoracic surgeon with the Papworth team, added that they have reduced ischemic time to about 15 minutes. “That’s considerably shorter than reperfusing the heart outside the body,” he said. “This results in a healthier organ for the recipient.”
The NRP approach is also less expensive than direct procurement as one OCS box costs about $75,000.
He pointed out that the NRP approach can also be used for heart transplants in children and even small babies, while currently the direct procurement technique is not typically suitable for children because the OCS box was not designed for small hearts.
DCD, using either technique, has increased the heart transplant rate by 40% at Papworth, and is being used at all seven transplant centers in the United Kingdom, “a world first,” noted Dr. Large.
The Papworth team recently published its 5-year experience with 25 NRP transplants and 85 direct procurement transplants. Survival in recipients was no different, although there was some suggestion that the NRP hearts may have been in slightly better condition, possibly being more resistant to immunological rejection.
Ethical concerns about NRP
Restarting the circulation during the NRP process has raised ethical concerns.
When the NRP technique was first used in the United States, these ethical questions were raised by several groups, including the American College of Physicians (ACP).
Harry Peled, MD, Providence St. Jude Medical Center, Fullerton, Calif., coauthor of a recent Viewpoint on the issue, is board-certified in both cardiology and critical care, and said he is a supporter of DCD using direct procurement, but he does not believe that NRP is ethical at present. He is not part of the ACP, but said his views align with those of the organization.
There are two ethical problems with NRP, he said. The first is whether by restarting the circulation, the NRP process violates the U.S. definition of death, and retrieval of organs would therefore violate the dead donor rule.
“American law states that death is the irreversible cessation of brain function or of circulatory function. But with NRP, the circulation is artificially restored, so the cessation of circulatory function is not irreversible,” Dr. Peled pointed out.
“I have no problem with DCD using direct procurement as we are not restarting the circulation. But NRP is restarting the circulation and that is a problem for me,” Dr. Peled said. “I would argue that by performing NRP, we are resuscitating the patient.”
The second ethical problem with NRP is concern about whether, during the process, there would be any circulation to the brain, and if so, would this be enough to restore some brain function? Before NRP is started, the main arch vessel arteries to the head are clamped to prevent flow to the brain, but there are worries that some blood flow may still be possible through small collateral vessels.
“We have established that these patients do not have enough brain function for a meaningful life, which is why a decision has been made to remove life support, but they have not been declared brain dead,” Dr. Peled said.
With direct procurement, the circulation is not restarted so there is no chance that any brain function will be restored, he said. “But with NRP, because the arch vessels have to be clamped to prevent brain circulation, that is admitting there is concern that brain function may be restored if circulation to the brain is reestablished, and brain function is compatible with life. As we do not know whether there is any meaningful circulation to the brain via the small collaterals, there is, in effect, a risk of bringing the patient back to life.”
The other major concern for some is whether even a very small amount of circulation to the brain would be enough to support consciousness, and “we don’t know that for certain,” Dr. Peled said.
The argument for NRP
Nader Moazami, MD, professor of cardiovascular surgery, NYU Langone Health, New York, is one of the more vocal proponents of NRP for DCD heart transplantation in the United States, and has coauthored responses to these ethical concerns.
“People are confusing many issues to produce an argument against NRP,” he said.
“Our position is that death has already been declared based on the lack of circulatory function for over 5 minutes and this has been with the full agreement of the family, knowing that the patient has no chance of a meaningful life. No one is thinking of trying to resuscitate the patient. It has already been established that any future efforts to resuscitate are futile. In this case, we are not resuscitating the patient by restarting the circulation. It is just regional perfusion of the organs.”
Dr. Moazami pointed out this concept was accepted for the practice of abdominal DCD when it first started in the United States in the 1990s where cold perfusion was used to preserve the abdominal organs before they were retrieved from the body.
“The new approach of using NRP is similar except that it involves circulating warm blood, which will preserve organs better and result in higher quality organs for the recipient.”
On the issue of concern about possible circulation to the brain, Dr. Moazami said: “The ethical critics of NRP are questioning whether the brain may not be dead. We are arguing that the patient has already been declared dead as they have had a circulatory death. You cannot die twice.”
He maintained that the clamping of the arch vessels to the head will ensure that when the circulation is restarted “the natural process of circulatory death leading to brain death will continue to progress.”
On the concerns about possible collateral flow to the brain, Dr. Moazami said there is no evidence that this occurs. “Prominent neurologists have said it is impossible for collaterals to provide any meaningful blood flow to the brain in this situation. And even if there is small amount of blood flow to the brain, this would be insufficient to maintain any meaningful brain function.”
But Dr. Peled argues that this has not been proved. “Even though we don’t think there is enough circulation to the brain for any function with NRP, we don’t know that with 100% certainty,” he said. “In my view, if there is a possibility of even the smallest amount of brain flow, we are going against the dead donor rule. We are rewriting the rules of death.”
Dr. Moazami countered: “Nothing in life is 100%, particularly in medicine. With that argument can you also prove with 100% certainty to me that there is absolutely no brain function with regular direct procurement DCD? We know that brain death has started, but the question is: Has it been completed? We don’t know the answer to this question with 100% certainty, but that is the case for regular direct procurement DCD as well, and that has been accepted by almost everyone.
“The whole issue revolves around when are we comfortable that death has occurred,” he said. “Those against NRP are concerned that organs are being taken before the patient is dead. But the key point is that the patient has already been declared dead.”
Since there is some concern over the ethics of NRP, why not just stick to DCD with direct procurement?
Dr. Moazami argued that NRP results in healthier organs. “NRP allows more successful heart transplants, liver transplants, lung transplants. It preserves all the organs better,” he said. “This will have a big impact on recipients – they would obviously much prefer a healthier organ. In addition, the process is easier and cheaper, so more centers will be able to do it, therefore more transplants will get done and more lives will be saved if NRP is used.”
He added: “I am a physician taking care of sick patients. I believe I have to respect the wishes of the donor and the donor family; make sure I’m not doing any harm to the donor; and ensure the best quality possible of the organ I am retrieving to best serve the recipient. I am happy I am doing this by using NRP for DCD heart transplantation.”
But Dr. Peled argued that while NRP may have some possible advantages over direct procurement, that does not justify allowing a process to go ahead that is unethical.
“The fact that NRP may result in some benefits doesn’t justify violating the dead donor rule or the possibility, however small, of causing pain to the donor. If it’s unethical, it’s unethical. Full stop,” he said.
“I feel that NRP is not respecting the rights of our patients and that the process does not have adequate transparency. We took it to our local ethics committee, and they decided not to approve NRP in our health care system. I agree with this decision,” Dr. Peled said.
“The trouble is different experts and different countries are not in agreement about this,” he added. “Reasonable, well-informed people are in disagreement. I do not believe we can have a standard of care where there is not consensus.”
Cautious nod
In a 2022 consensus statement, the International Society for Heart and Lung Transplantation (ISHLT) gave a cautious nod toward DCD and NRP, dependent on local recommendations.
The ISHLT conclusion reads: “With appropriate consideration of the ethical principles involved in organ donation, DCD can be undertaken in a morally permissible manner. In all cases, the introduction of DCD programs should be in accordance with local legal regulations. Countries lacking a DCD pathway should be encouraged to develop national ethical, professional, and legal frameworks to address both public and professional concerns.”
The author of a recent editorial on the subject, Ulrich P. Jorde, MD, head of the heart transplant program at Montefiore Medical Center, New York, said, “DCD is a great step forward. People regularly die on the heart transplant waiting list. DCD will increase the supply of donor hearts by 20% to 30%.”
However, he noted that while most societies have agreed on a protocol for organ donation based on brain death, the situation is more complicated with circulatory death.
“Different countries have different definitions of circulatory death. How long do we have to wait after the heart has stopped beating before the patient is declared dead? Most countries have agreed on 5 minutes, but other countries have imposed different periods and as such, different definitions of death.
“The ISHLT statement says that restarting the circulation is acceptable if death has been certified according to prevailing law and surgical interventions are undertaken to preclude any restoration of cerebral circulation. But our problem is that different regional societies have different definitions of circulatory, death which makes the situation confusing.”
Dr. Jorde added: “We also have to weigh the wishes of the donor and their family. If family, advocating what are presumed to be the donor’s wishes, have decided that DCD would be acceptable and they understand the concept and wish to donate the organs after circulatory death, this should be strongly considered under the concept of self-determination, a basic human right.”
Variations in practice around the world
This ethical debate has led to large variations in practice around the world, with some countries, such as Spain, allowing both methods of DCD, while Australia allows direct procurement but not NRP, and Germany currently does not allow DCD at all.
In the United States, things are even more complicated, with some states allowing NRP while others don’t. Even within states, some hospitals and transplant organizations allow NRP, and others don’t.
David A. D’Alessandro, MD, cardiac surgeon at Massachusetts General Hospital, Boston, uses only the direct procurement approach as his region does not allow NRP.
“The direct procurement approach is not controversial and to me that’s a big advantage. I believe we need to agree on the ethics first, and then get into a debate about which technique is better,” he told this news organization.
Dr. D’Alessandro and his group recently published the results of their study, with direct procurement DCD heart transplantation showing similar short-term clinical outcomes to DBD.
“We are only doing direct procurement and we are seeing good results that appear to be comparable to DBD. That is good enough for me,” he said.
Dr. D’Alessandro estimates that in the United States both types of DCD procedures are currently being done about equally.
“Anything we can do to increase the amount of hearts available for transplantation is a big deal,” he said. “At the moment, only the very sickest patients get a heart transplant, and many patients die on the transplant waiting list. Very sadly, many young people die every year from a circulatory death after having life support withdrawn. Before DCD, these beautiful functional organs were not able to be used. Now we have a way of saving lives with these organs.”
Dr. D’Alessandro noted that more and more centers in the United States are starting to perform DCD heart transplants.
“Not every transplant center may join in as the DCD procedures are very resource-intensive and time-consuming. For low-volume transplant centers, it may not be worth the expense and anguish to do DCD heart transplants. But bigger centers will need to engage in DCD to remain competitive. My guess is that 50%-70% of U.S. transplant centers will do DCD in future.”
He said he thinks it is a “medical shortcoming” that agreement cannot be reached on the ethics of NRP. “In an ideal world everyone would be on the same page. It makes me a bit uncomfortable that some people think it’s okay and some people don’t.”
Adam DeVore, MD, a cardiologist at Duke University Medical Center, Durham, N.C., the first U.S. center to perform an adult DCD heart transplant, reported that his institution uses both methods, with the choice sometimes depending on how far the heart must travel.
“If the recipient is near, NRP may be chosen as the heart is transported on ice, but if it needs to go further away we are more likely to choose direct procurement and use of the OCS box,” he said.
“I am really proud of what we’ve been able to do, helping to introduce DCD in the U.S.,” Dr. DeVore said. “This is having a massive benefit in increasing the number of hearts for donation with great outcomes.”
But he acknowledged that the whole concept of DCD is somewhat controversial.
“The idea of brain death really came about for the purpose of heart donation. The two things are very intricately tied. Trying to do heart donation without brain death having been declared is foreign to people. Also, in DCD there is the issue of [this]: When life support is removed, how long do we wait before death can be declared? That could be in conflict with how long the organ needs to remain viable. We are going through the process now of looking at these questions. There is a lot of variation in the U.S. about the withdrawal of care and the declaration of death, which is not completely standardized.
“But the concept of circulatory death itself is accepted after the withdrawal of life support. I think it’s the rush to take the organs out that makes it more difficult.”
Dr. DeVore said the field is moving forward now. “As the process has become more common, people have become more comfortable, probably because of the big difference it will make to saving lives. But we do need to try and standardize best practices.”
A recent Canadian review of the ethics of DCD concluded that the direct procurement approach would be in alignment with current medical guidelines, but that further work is required to evaluate the consistency of NRP with current Canadian death determination policy and to ensure the absence of brain perfusion during this process.
In the United Kingdom, the definition of death is brain-based, and brain death is defined on a neurological basis.
Dr. Stephen Large from Papworth explained that this recognizes the presence of brain-stem death through brain stem reflex testing after the withdrawal of life support, cardiorespiratory arrest and 5 further minutes of ischemia. As long as NRP does not restore intracranial (brainstem) perfusion after death has been confirmed, then it is consistent with laws for death determination and therefore both direct procurement and NRP are permissible.
However, the question over possible collateral flow to the brain has led the United Kingdom to pause the NRP technique as routine practice while this is investigated further. So, at the present time, the vast majority of DCD heart transplants are being conducted using the direct procurement approach.
But the United Kingdom is facing the bigger challenge: national funding that will soon end. “The DCD program in the U.K. has been extremely successful, increasing heart transplant rates by up to 28%,” Dr. Berman said. “Everybody wants it to continue. But at present the DCD program only has national funding in the U.K. until March 2023. We don’t know what will happen after that.”
The current model in the United Kingdom consists of three specialized DCD heart retrieval teams, a national protocol of direct organ procurement and delivery of DCD hearts to all seven transplant programs, both adult and pediatric.
If the national funding is not extended, “we will go back to individual hospitals trying to fund their own programs. That will be a serious threat to the program and could result in a large reduction in heart transplants,” said Dr. Berman.
Definition of death
The crux of the issue with regard to NRP seems to be variations in how death is defined and the interpretation of those definitions.
DCD donors will have had many tests indicating severe brain damage, a neurologist will have declared the prognosis is futile, and relatives will have agreed to withdraw life support, Dr. Jorde said. “The heart stops beating, and the stand-off time means that blood flow to the brain ceases completely for at least 5 minutes before circulatory death is declared. This is enough on its own to stop brain function.”
Dr. Large made the point that by the time the circulation is reestablished with NRP, more time has elapsed, and the brain will have been without perfusion for much longer than 5 minutes, so it would be “physiologically almost impossible” for there to be any blood flow to the brain.
“Because these brains are already very damaged before life support was removed, the intracranial pressure is high, which will further discourage blood flow to the brain,” he said. Then the donor goes through a period of anoxic heart arrest, up to 16 minutes at a minimum of no blood supply, enough on its own to stop meaningful brain function.
“It’s asking an awful lot to believe that there might be any brain function left,” he said. “And if, on reestablishing the circulation with NRP, there is any blood in the collaterals, the pressure of such flow is so low it won’t enter the brain.”
Dr. Large also pointed out that the fact that the United Kingdom requires a neurologic definition for brain-stem death makes the process easier.
In Australia, St. Vincent’s cardiologist Dr. MacDonald noted that death is defined as the irreversible cessation of circulation, so the NRP procedure is not allowed.
“With NRP, there is an ethical dilemma over whether the patient has legally died or not. Different countries have different ways of defining death. Perhaps society will have to review of the definition of death,” he suggested. Death is a process, “but for organ donation, we have to choose a moment in time of that process that satisfies everyone – when there is no prospect of recovery of the donor but the organs can still be utilized without harming the donor.”
Dr. MacDonald said the field is in transition. “I don’t want to argue that one technique is better than the other; I think it’s good to have access to both techniques. Anything that will increase the number of transplants we can do is a good thing.”
Collaborative decision
Everyone seems to agree that there should be an effort to try to define death in a uniform way worldwide, and that international, national and local regulations are aligned with each other.
Dr. Jorde said: “It is of critical importance that local guidelines are streamlined, firstly in any one given country and then globally, and these things must be discussed transparently within society with all stakeholders – doctors, patients, citizens.”
Dr. Peled, from Providence St. Jude in California, concurred: “There is the possibility that we could change the definition of death, but that cannot be a decision based solely on transplant organizations. It has to be a collaborative decision with a large input from groups who do not have an interest in the procurement of organs.”
He added: “The dialogue so far has been civil, and everybody is trying to do the right thing. My hope is that as a civilized society we will figure out a way forward. At present, there is significant controversy about NRP, and families need to know that. My main concern is that if there is any lack of transparency in getting informed consent, then this risks people losing trust in the donation system.”
Dr. Moazami, from NYU Langone, said the controversy has cast a cloud over the practice of NRP throughout the world. “We need to get it sorted out.”
He said he believes the way forward is to settle the question of whether there is any meaningful blood flow to the brain with the NRP technique.
“This is where the research has to focus. I believe this concern is hypothetical, but I am happy to do the studies to confirm that. Then, the issue should come to a rest. I think that is the right way forward – to do the studies rather than enforcing a moratorium on the practice because of a hypothetical concern.”
These studies on blood flow to the brain are now getting started in both the United Kingdom and the United States.
The U.K. study is being run by Antonio Rubino, MD, consultant in cardiothoracic anesthesia and intensive care at Papworth Hospital NHS Foundation and clinical lead, organ donation. Dr. Rubino explained that the study will assess cerebral blood flow using CT angiography of the brain. “We hypothesize that this will provide evidence to indicate that brain blood flow is not present during NRP and promote trust in the use of NRP in routine practice,” he said.
Dr. Large said: “Rather than having these tortured arguments, we will do the measurements. For the sake of society in this situation, I think it’s good to stop and take a breath. We must measure this, and we are doing just that.”
If there is any blood flow at all, Dr. Large said they will then have to seek expert guidance. “Say we find there is 50 mL of blood flow and normal blood flow is 1,500 mL/min. We will need expert guidance on whether it is remotely possible to be sentient on that. I would say it would be extraordinarily unlikely.”
Dr. Berman summarized the situation: “DCD is increasing the availability of hearts for transplant. This is saving lives, reducing the number of patients on the waiting list, and reducing hospital stays for patients unable to leave the hospital without a transplant. It is definitely here to stay. It is crucial that it gets funded properly, and it is also crucial that we resolve the NRP ethical issues as soon as possible.”
He is hopeful that some of these issues will be resolved this year.
Dr. MacDonald reported he has received “in-kind” support from Transmedics through provision of research modules for preclinical research studies. Dr. D’Alessandro reported he is on the speakers bureau for Abiomed, not relevant to this article. No other relevant disclosures were reported.
A version of this article first appeared on Medscape.com.
The relatively recent innovation of heart transplantation after circulatory death of the donor is increasing the number of donor hearts available and leading to many more lives on the heart transplant waiting list being saved. Experts agree it’s a major and very welcome advance in medicine.
However, some of the processes involved in one approach to donation after circulatory death has raised ethical concerns and questions about whether they violate the “dead donor rule” – a principle that requires patients be declared dead before removal of life-sustaining organs for transplant.
Experts in the fields of transplantation and medical ethics have yet to reach consensus, causing problems for the transplant community, who worry that this could cause a loss of confidence in the entire transplant process.
A new pathway for heart transplantation
The traditional approach to transplantation is to retrieve organs from a donor who has been declared brain dead, known as “donation after brain death (DBD).” These patients have usually suffered a catastrophic brain injury but survived to get to intensive care.
As the brain swells because of injury, it becomes evident that all brain function is lost, and the patient is declared brain dead. However, breathing is maintained by the ventilator and the heart is still beating. Because the organs are being oxygenated, there is no immediate rush to retrieve the organs and the heart can be evaluated for its suitability for transplant in a calm and methodical way before it is removed.
However, there is a massive shortage of organs, especially hearts, partially because of the limited number of donors who are declared brain dead in that setting.
In recent years, another pathway for organ transplantation has become available: “donation after circulatory death (DCD).” These patients also have suffered a catastrophic brain injury considered to be nonsurvivable, but unlike the DBD situation, the brain still has some function, so the patient does not meet the criteria for brain death.
Still, because the patient is considered to have no chance of a meaningful recovery, the family often recognizes the futility of treatment and agrees to the withdrawal of life support. When this happens, the heart normally stops beating after a period of time. There is then a “stand-off time” – normally 5 minutes – after which death is declared and the organs can be removed.
The difficulty with this approach, however, is that because the heart has been stopped, it has been deprived of oxygen, potentially causing injury. While DCD has been practiced for several years to retrieve organs such as the kidney, liver, lungs, and pancreas, the heart is more difficult as it is more susceptible to oxygen deprivation. And for the heart to be assessed for transplant suitability, it should ideally be beating, so it has to be reperfused and restarted quickly after death has been declared.
For many years it was thought the oxygen deprivation that occurs after circulatory death would be too much to provide a functional organ. But researchers in the United Kingdom and Australia developed techniques to overcome this problem, and early DCD heart transplants took place in 2014 in Australia, and in 2015 in the United Kingdom.
Heart transplantation after circulatory death has now become a routine part of the transplant program in many countries, including the United States, Spain, Belgium, the Netherlands, and Austria.
In the United States, 348 DCD heart transplants were performed in 2022, with numbers expected to reach 700 to 800 this year as more centers come online.
It is expected that most countries with heart transplant programs will follow suit and the number of donor hearts will increase by up to 30% worldwide because of DCD.
Currently, there are about 8,000 heart transplants worldwide each year and with DCD this could rise to about 10,000, potentially an extra 2,000 lives saved each year, experts estimate.
Two different approaches to DCD heart transplantation have been developed.
The direct procurement approach
The Australian group, based at St. Vincent’s Hospital in Sydney, developed a technique referred to as “direct procurement”: after the standoff period and declaration of circulatory death, the chest is opened, and the heart is removed. New technology, the Organ Care System (OCS) heart box (Transmedics), is then used to reperfuse and restart the heart outside the body so its suitability for transplant can be assessed.
The heart is kept perfused and beating in the OCS box while it is being transported to the recipient. This has enabled longer transit times than the traditional way of transporting the nonbeating heart on ice.
Peter MacDonald, MD, PhD, from the St Vincent’s group that developed this approach, said, “Most people thought a heart from a DCD donor would not survive transport – that the injury to the heart from the combination of life support withdrawal, stand-off time, and cold storage would be too much. But we modeled the process in the lab and were able to show that we were able to get the heart beating again after withdrawal of life support.”
Dr. McDonald noted that “the recipient of their first human DCD heart transplant using this machine in 2014 is still alive and well.” The Australian group has now done 85 of these DCD heart transplants, and they have increased the number of heart transplant procedures at St. Vincent’s Hospital by 25%.
Normothermic regional perfusion (NRP)
The U.K. group, based at the Royal Papworth Hospital in Cambridge, England, developed a different approach to DCD: After the standoff period and the declaration of circulatory death, the donor is connected to a heart/lung machine using extracorporeal membrane oxygenation (ECMO) so that the heart is perfused and starts beating again inside the body. This approach is known as normothermic regional perfusion (NRP).
Marius Berman, MD, surgical lead for Transplantation and Mechanical Circulatory Support at Papworth, explained that the NRP approach allows the heart to be perfused and restarted faster than direct procurement, resulting in a shorter ischemic time. The heart can be evaluated thoroughly for suitability for transplantation in situ before committing to transplantation, and because the heart is less damaged, it can be transported on ice without use of the OCS box.
“DCD is more complicated than DBD, because the heart has stopped and has to be restarted. Retrieval teams have to be very experienced,” Dr. Berman noted. “This is more of an issue for the direct procurement approach, where the chest has to be opened and the heart retrieved as fast as possible. It is a rush. The longer time without the heart being perfused correlates to an increased incidence of primary graft dysfunction. With NRP, we can get the heart started again more quickly, which is crucial.”
Stephen Large, MBBS, another cardiothoracic surgeon with the Papworth team, added that they have reduced ischemic time to about 15 minutes. “That’s considerably shorter than reperfusing the heart outside the body,” he said. “This results in a healthier organ for the recipient.”
The NRP approach is also less expensive than direct procurement as one OCS box costs about $75,000.
He pointed out that the NRP approach can also be used for heart transplants in children and even small babies, while currently the direct procurement technique is not typically suitable for children because the OCS box was not designed for small hearts.
DCD, using either technique, has increased the heart transplant rate by 40% at Papworth, and is being used at all seven transplant centers in the United Kingdom, “a world first,” noted Dr. Large.
The Papworth team recently published its 5-year experience with 25 NRP transplants and 85 direct procurement transplants. Survival in recipients was no different, although there was some suggestion that the NRP hearts may have been in slightly better condition, possibly being more resistant to immunological rejection.
Ethical concerns about NRP
Restarting the circulation during the NRP process has raised ethical concerns.
When the NRP technique was first used in the United States, these ethical questions were raised by several groups, including the American College of Physicians (ACP).
Harry Peled, MD, Providence St. Jude Medical Center, Fullerton, Calif., coauthor of a recent Viewpoint on the issue, is board-certified in both cardiology and critical care, and said he is a supporter of DCD using direct procurement, but he does not believe that NRP is ethical at present. He is not part of the ACP, but said his views align with those of the organization.
There are two ethical problems with NRP, he said. The first is whether by restarting the circulation, the NRP process violates the U.S. definition of death, and retrieval of organs would therefore violate the dead donor rule.
“American law states that death is the irreversible cessation of brain function or of circulatory function. But with NRP, the circulation is artificially restored, so the cessation of circulatory function is not irreversible,” Dr. Peled pointed out.
“I have no problem with DCD using direct procurement as we are not restarting the circulation. But NRP is restarting the circulation and that is a problem for me,” Dr. Peled said. “I would argue that by performing NRP, we are resuscitating the patient.”
The second ethical problem with NRP is concern about whether, during the process, there would be any circulation to the brain, and if so, would this be enough to restore some brain function? Before NRP is started, the main arch vessel arteries to the head are clamped to prevent flow to the brain, but there are worries that some blood flow may still be possible through small collateral vessels.
“We have established that these patients do not have enough brain function for a meaningful life, which is why a decision has been made to remove life support, but they have not been declared brain dead,” Dr. Peled said.
With direct procurement, the circulation is not restarted so there is no chance that any brain function will be restored, he said. “But with NRP, because the arch vessels have to be clamped to prevent brain circulation, that is admitting there is concern that brain function may be restored if circulation to the brain is reestablished, and brain function is compatible with life. As we do not know whether there is any meaningful circulation to the brain via the small collaterals, there is, in effect, a risk of bringing the patient back to life.”
The other major concern for some is whether even a very small amount of circulation to the brain would be enough to support consciousness, and “we don’t know that for certain,” Dr. Peled said.
The argument for NRP
Nader Moazami, MD, professor of cardiovascular surgery, NYU Langone Health, New York, is one of the more vocal proponents of NRP for DCD heart transplantation in the United States, and has coauthored responses to these ethical concerns.
“People are confusing many issues to produce an argument against NRP,” he said.
“Our position is that death has already been declared based on the lack of circulatory function for over 5 minutes and this has been with the full agreement of the family, knowing that the patient has no chance of a meaningful life. No one is thinking of trying to resuscitate the patient. It has already been established that any future efforts to resuscitate are futile. In this case, we are not resuscitating the patient by restarting the circulation. It is just regional perfusion of the organs.”
Dr. Moazami pointed out this concept was accepted for the practice of abdominal DCD when it first started in the United States in the 1990s where cold perfusion was used to preserve the abdominal organs before they were retrieved from the body.
“The new approach of using NRP is similar except that it involves circulating warm blood, which will preserve organs better and result in higher quality organs for the recipient.”
On the issue of concern about possible circulation to the brain, Dr. Moazami said: “The ethical critics of NRP are questioning whether the brain may not be dead. We are arguing that the patient has already been declared dead as they have had a circulatory death. You cannot die twice.”
He maintained that the clamping of the arch vessels to the head will ensure that when the circulation is restarted “the natural process of circulatory death leading to brain death will continue to progress.”
On the concerns about possible collateral flow to the brain, Dr. Moazami said there is no evidence that this occurs. “Prominent neurologists have said it is impossible for collaterals to provide any meaningful blood flow to the brain in this situation. And even if there is small amount of blood flow to the brain, this would be insufficient to maintain any meaningful brain function.”
But Dr. Peled argues that this has not been proved. “Even though we don’t think there is enough circulation to the brain for any function with NRP, we don’t know that with 100% certainty,” he said. “In my view, if there is a possibility of even the smallest amount of brain flow, we are going against the dead donor rule. We are rewriting the rules of death.”
Dr. Moazami countered: “Nothing in life is 100%, particularly in medicine. With that argument can you also prove with 100% certainty to me that there is absolutely no brain function with regular direct procurement DCD? We know that brain death has started, but the question is: Has it been completed? We don’t know the answer to this question with 100% certainty, but that is the case for regular direct procurement DCD as well, and that has been accepted by almost everyone.
“The whole issue revolves around when are we comfortable that death has occurred,” he said. “Those against NRP are concerned that organs are being taken before the patient is dead. But the key point is that the patient has already been declared dead.”
Since there is some concern over the ethics of NRP, why not just stick to DCD with direct procurement?
Dr. Moazami argued that NRP results in healthier organs. “NRP allows more successful heart transplants, liver transplants, lung transplants. It preserves all the organs better,” he said. “This will have a big impact on recipients – they would obviously much prefer a healthier organ. In addition, the process is easier and cheaper, so more centers will be able to do it, therefore more transplants will get done and more lives will be saved if NRP is used.”
He added: “I am a physician taking care of sick patients. I believe I have to respect the wishes of the donor and the donor family; make sure I’m not doing any harm to the donor; and ensure the best quality possible of the organ I am retrieving to best serve the recipient. I am happy I am doing this by using NRP for DCD heart transplantation.”
But Dr. Peled argued that while NRP may have some possible advantages over direct procurement, that does not justify allowing a process to go ahead that is unethical.
“The fact that NRP may result in some benefits doesn’t justify violating the dead donor rule or the possibility, however small, of causing pain to the donor. If it’s unethical, it’s unethical. Full stop,” he said.
“I feel that NRP is not respecting the rights of our patients and that the process does not have adequate transparency. We took it to our local ethics committee, and they decided not to approve NRP in our health care system. I agree with this decision,” Dr. Peled said.
“The trouble is different experts and different countries are not in agreement about this,” he added. “Reasonable, well-informed people are in disagreement. I do not believe we can have a standard of care where there is not consensus.”
Cautious nod
In a 2022 consensus statement, the International Society for Heart and Lung Transplantation (ISHLT) gave a cautious nod toward DCD and NRP, dependent on local recommendations.
The ISHLT conclusion reads: “With appropriate consideration of the ethical principles involved in organ donation, DCD can be undertaken in a morally permissible manner. In all cases, the introduction of DCD programs should be in accordance with local legal regulations. Countries lacking a DCD pathway should be encouraged to develop national ethical, professional, and legal frameworks to address both public and professional concerns.”
The author of a recent editorial on the subject, Ulrich P. Jorde, MD, head of the heart transplant program at Montefiore Medical Center, New York, said, “DCD is a great step forward. People regularly die on the heart transplant waiting list. DCD will increase the supply of donor hearts by 20% to 30%.”
However, he noted that while most societies have agreed on a protocol for organ donation based on brain death, the situation is more complicated with circulatory death.
“Different countries have different definitions of circulatory death. How long do we have to wait after the heart has stopped beating before the patient is declared dead? Most countries have agreed on 5 minutes, but other countries have imposed different periods and as such, different definitions of death.
“The ISHLT statement says that restarting the circulation is acceptable if death has been certified according to prevailing law and surgical interventions are undertaken to preclude any restoration of cerebral circulation. But our problem is that different regional societies have different definitions of circulatory, death which makes the situation confusing.”
Dr. Jorde added: “We also have to weigh the wishes of the donor and their family. If family, advocating what are presumed to be the donor’s wishes, have decided that DCD would be acceptable and they understand the concept and wish to donate the organs after circulatory death, this should be strongly considered under the concept of self-determination, a basic human right.”
Variations in practice around the world
This ethical debate has led to large variations in practice around the world, with some countries, such as Spain, allowing both methods of DCD, while Australia allows direct procurement but not NRP, and Germany currently does not allow DCD at all.
In the United States, things are even more complicated, with some states allowing NRP while others don’t. Even within states, some hospitals and transplant organizations allow NRP, and others don’t.
David A. D’Alessandro, MD, cardiac surgeon at Massachusetts General Hospital, Boston, uses only the direct procurement approach as his region does not allow NRP.
“The direct procurement approach is not controversial and to me that’s a big advantage. I believe we need to agree on the ethics first, and then get into a debate about which technique is better,” he told this news organization.
Dr. D’Alessandro and his group recently published the results of their study, with direct procurement DCD heart transplantation showing similar short-term clinical outcomes to DBD.
“We are only doing direct procurement and we are seeing good results that appear to be comparable to DBD. That is good enough for me,” he said.
Dr. D’Alessandro estimates that in the United States both types of DCD procedures are currently being done about equally.
“Anything we can do to increase the amount of hearts available for transplantation is a big deal,” he said. “At the moment, only the very sickest patients get a heart transplant, and many patients die on the transplant waiting list. Very sadly, many young people die every year from a circulatory death after having life support withdrawn. Before DCD, these beautiful functional organs were not able to be used. Now we have a way of saving lives with these organs.”
Dr. D’Alessandro noted that more and more centers in the United States are starting to perform DCD heart transplants.
“Not every transplant center may join in as the DCD procedures are very resource-intensive and time-consuming. For low-volume transplant centers, it may not be worth the expense and anguish to do DCD heart transplants. But bigger centers will need to engage in DCD to remain competitive. My guess is that 50%-70% of U.S. transplant centers will do DCD in future.”
He said he thinks it is a “medical shortcoming” that agreement cannot be reached on the ethics of NRP. “In an ideal world everyone would be on the same page. It makes me a bit uncomfortable that some people think it’s okay and some people don’t.”
Adam DeVore, MD, a cardiologist at Duke University Medical Center, Durham, N.C., the first U.S. center to perform an adult DCD heart transplant, reported that his institution uses both methods, with the choice sometimes depending on how far the heart must travel.
“If the recipient is near, NRP may be chosen as the heart is transported on ice, but if it needs to go further away we are more likely to choose direct procurement and use of the OCS box,” he said.
“I am really proud of what we’ve been able to do, helping to introduce DCD in the U.S.,” Dr. DeVore said. “This is having a massive benefit in increasing the number of hearts for donation with great outcomes.”
But he acknowledged that the whole concept of DCD is somewhat controversial.
“The idea of brain death really came about for the purpose of heart donation. The two things are very intricately tied. Trying to do heart donation without brain death having been declared is foreign to people. Also, in DCD there is the issue of [this]: When life support is removed, how long do we wait before death can be declared? That could be in conflict with how long the organ needs to remain viable. We are going through the process now of looking at these questions. There is a lot of variation in the U.S. about the withdrawal of care and the declaration of death, which is not completely standardized.
“But the concept of circulatory death itself is accepted after the withdrawal of life support. I think it’s the rush to take the organs out that makes it more difficult.”
Dr. DeVore said the field is moving forward now. “As the process has become more common, people have become more comfortable, probably because of the big difference it will make to saving lives. But we do need to try and standardize best practices.”
A recent Canadian review of the ethics of DCD concluded that the direct procurement approach would be in alignment with current medical guidelines, but that further work is required to evaluate the consistency of NRP with current Canadian death determination policy and to ensure the absence of brain perfusion during this process.
In the United Kingdom, the definition of death is brain-based, and brain death is defined on a neurological basis.
Dr. Stephen Large from Papworth explained that this recognizes the presence of brain-stem death through brain stem reflex testing after the withdrawal of life support, cardiorespiratory arrest and 5 further minutes of ischemia. As long as NRP does not restore intracranial (brainstem) perfusion after death has been confirmed, then it is consistent with laws for death determination and therefore both direct procurement and NRP are permissible.
However, the question over possible collateral flow to the brain has led the United Kingdom to pause the NRP technique as routine practice while this is investigated further. So, at the present time, the vast majority of DCD heart transplants are being conducted using the direct procurement approach.
But the United Kingdom is facing the bigger challenge: national funding that will soon end. “The DCD program in the U.K. has been extremely successful, increasing heart transplant rates by up to 28%,” Dr. Berman said. “Everybody wants it to continue. But at present the DCD program only has national funding in the U.K. until March 2023. We don’t know what will happen after that.”
The current model in the United Kingdom consists of three specialized DCD heart retrieval teams, a national protocol of direct organ procurement and delivery of DCD hearts to all seven transplant programs, both adult and pediatric.
If the national funding is not extended, “we will go back to individual hospitals trying to fund their own programs. That will be a serious threat to the program and could result in a large reduction in heart transplants,” said Dr. Berman.
Definition of death
The crux of the issue with regard to NRP seems to be variations in how death is defined and the interpretation of those definitions.
DCD donors will have had many tests indicating severe brain damage, a neurologist will have declared the prognosis is futile, and relatives will have agreed to withdraw life support, Dr. Jorde said. “The heart stops beating, and the stand-off time means that blood flow to the brain ceases completely for at least 5 minutes before circulatory death is declared. This is enough on its own to stop brain function.”
Dr. Large made the point that by the time the circulation is reestablished with NRP, more time has elapsed, and the brain will have been without perfusion for much longer than 5 minutes, so it would be “physiologically almost impossible” for there to be any blood flow to the brain.
“Because these brains are already very damaged before life support was removed, the intracranial pressure is high, which will further discourage blood flow to the brain,” he said. Then the donor goes through a period of anoxic heart arrest, up to 16 minutes at a minimum of no blood supply, enough on its own to stop meaningful brain function.
“It’s asking an awful lot to believe that there might be any brain function left,” he said. “And if, on reestablishing the circulation with NRP, there is any blood in the collaterals, the pressure of such flow is so low it won’t enter the brain.”
Dr. Large also pointed out that the fact that the United Kingdom requires a neurologic definition for brain-stem death makes the process easier.
In Australia, St. Vincent’s cardiologist Dr. MacDonald noted that death is defined as the irreversible cessation of circulation, so the NRP procedure is not allowed.
“With NRP, there is an ethical dilemma over whether the patient has legally died or not. Different countries have different ways of defining death. Perhaps society will have to review of the definition of death,” he suggested. Death is a process, “but for organ donation, we have to choose a moment in time of that process that satisfies everyone – when there is no prospect of recovery of the donor but the organs can still be utilized without harming the donor.”
Dr. MacDonald said the field is in transition. “I don’t want to argue that one technique is better than the other; I think it’s good to have access to both techniques. Anything that will increase the number of transplants we can do is a good thing.”
Collaborative decision
Everyone seems to agree that there should be an effort to try to define death in a uniform way worldwide, and that international, national and local regulations are aligned with each other.
Dr. Jorde said: “It is of critical importance that local guidelines are streamlined, firstly in any one given country and then globally, and these things must be discussed transparently within society with all stakeholders – doctors, patients, citizens.”
Dr. Peled, from Providence St. Jude in California, concurred: “There is the possibility that we could change the definition of death, but that cannot be a decision based solely on transplant organizations. It has to be a collaborative decision with a large input from groups who do not have an interest in the procurement of organs.”
He added: “The dialogue so far has been civil, and everybody is trying to do the right thing. My hope is that as a civilized society we will figure out a way forward. At present, there is significant controversy about NRP, and families need to know that. My main concern is that if there is any lack of transparency in getting informed consent, then this risks people losing trust in the donation system.”
Dr. Moazami, from NYU Langone, said the controversy has cast a cloud over the practice of NRP throughout the world. “We need to get it sorted out.”
He said he believes the way forward is to settle the question of whether there is any meaningful blood flow to the brain with the NRP technique.
“This is where the research has to focus. I believe this concern is hypothetical, but I am happy to do the studies to confirm that. Then, the issue should come to a rest. I think that is the right way forward – to do the studies rather than enforcing a moratorium on the practice because of a hypothetical concern.”
These studies on blood flow to the brain are now getting started in both the United Kingdom and the United States.
The U.K. study is being run by Antonio Rubino, MD, consultant in cardiothoracic anesthesia and intensive care at Papworth Hospital NHS Foundation and clinical lead, organ donation. Dr. Rubino explained that the study will assess cerebral blood flow using CT angiography of the brain. “We hypothesize that this will provide evidence to indicate that brain blood flow is not present during NRP and promote trust in the use of NRP in routine practice,” he said.
Dr. Large said: “Rather than having these tortured arguments, we will do the measurements. For the sake of society in this situation, I think it’s good to stop and take a breath. We must measure this, and we are doing just that.”
If there is any blood flow at all, Dr. Large said they will then have to seek expert guidance. “Say we find there is 50 mL of blood flow and normal blood flow is 1,500 mL/min. We will need expert guidance on whether it is remotely possible to be sentient on that. I would say it would be extraordinarily unlikely.”
Dr. Berman summarized the situation: “DCD is increasing the availability of hearts for transplant. This is saving lives, reducing the number of patients on the waiting list, and reducing hospital stays for patients unable to leave the hospital without a transplant. It is definitely here to stay. It is crucial that it gets funded properly, and it is also crucial that we resolve the NRP ethical issues as soon as possible.”
He is hopeful that some of these issues will be resolved this year.
Dr. MacDonald reported he has received “in-kind” support from Transmedics through provision of research modules for preclinical research studies. Dr. D’Alessandro reported he is on the speakers bureau for Abiomed, not relevant to this article. No other relevant disclosures were reported.
A version of this article first appeared on Medscape.com.
Implementation of a Multidisciplinary Team–Based Clinical Care Pathway Is Associated With Increased Surgery Rates for Infective Endocarditis
From the University of Missouri School of Medicine, Columbia, MO (Haley Crosby); Department of Clinical Family and Community Medicine, University of Missouri, Columbia, MO (Dr. Pierce); and Department of Medicine, Divisions of Infectious Diseases and Pulmonary, Critical Care and Environmental Medicine, University of Missouri, Columbia, MO, and Divisions of Pulmonary and Critical Care Medicine and Infectious Diseases, University of Maryland Baltimore Washington Medical Center, Glen Burnie, MD (Dr. Regunath).
ABSTRACT
Objective: Multidisciplinary teams (MDTs) improve outcomes for patients with infective endocarditis (IE), but methods of implementation vary. In our academic medical center, we developed an MDT approach guided by a clinical care pathway and assessed outcomes of patients with IE.
Methods: We compared outcomes of patients with IE and indications for surgery between December 2018 and June 2020 with our prior published data for the period January to December 2016. MDT interventions involved recurring conferences with infectious diseases physicians in team meetings and promoting a clinical care pathway to guide providers on steps in management. Primary outcomes were surgery and in-hospital death.
Results: Prior to the intervention, 6 of 21 (28.6%) patients with indications for surgery underwent surgery or were transferred to higher centers for surgery, and 6 (28.6%) patients died. Post intervention, 17 of 31 (54.8%) patients underwent or were transferred for surgery, and 5 (16.1%) died. After adjusting for age and gender, the odds of surgery or transfer for surgery for patients in the postintervention period were 4.88 (95% CI, 1.20-19.79; P = .027) compared with the pre-intervention period. The odds ratio for death among patients in the postintervention period was 0.40 (95% CI, 0.09-1.69; P = .21).
Conclusion: An MDT team approach using a clinical pathway was associated with an increased number of surgeries performed for IE and may lower rates of in-hospital mortality.
Keywords: infective endocarditis, clinical pathway, quality improvement, multidisciplinary team, valve surgery.
Infective endocarditis (IE) is associated with significant morbidity and mortality.1 Rates of IE due to Staphylococcus aureus are increasing in the United States.2 Reported in-hospital mortality from IE ranges from 15% to 20%.3
Clinical pathways are defined as “structured, multidisciplinary plans of care used by health services to detail essential steps in the care of patients with a specific clinical problem.”12 In the modern era, these pathways are often developed and implemented via the electronic health record (EHR) system. Studies of clinical pathways generally demonstrate improvements in patient outcomes, quality of care, or resource utilization.13,14 Clinical pathways represent 1 possible approach to the implementation of a MDT in the care of patients with IE.15
In our earlier work, we used quality improvement principles in the design of an MDT approach to IE care at our institution.16 Despite having indications for surgery, 12 of 21 (57.1%) patients with IE did not undergo surgery, and we identified these missed opportunities for surgery as a leverage point for improvement of outcomes. With input from the various specialties and stakeholders, we developed a clinical pathway (algorithm) for the institutional management of IE that guides next steps in clinical care and their timelines, aiming to reduce by 50% (from 57.1% to 28.6%) the number of patients with IE who do not undergo surgery despite guideline indications for early surgical intervention. In this report, we describe the implementation of this clinical pathway as our MDT approach to the care of patients with IE at our institution.
Methods
The University of Missouri, Columbia, is a tertiary care academic health system with 5 hospitals and more than 60 clinic locations across central Missouri. In the spring of 2018, an MDT was developed, with support from administrative leaders, to improve the care of patients with IE at our institution. The work group prioritized one leverage point to improve IE outcomes, which was improving the number of surgeries performed on those IE patients who had guideline indications for surgery. A clinical pathway was developed around this leverage point (Figure 1). The pathway leveraged the 6 T’s (Table 1) to guide providers through the evaluation and management of IE.17 The pathway focused on improving adherence to standards of care and reduction in practice variation by defining indications for referrals and diagnostic interventions, helping to reduce delays in consultation and diagnosis. The pathway also clearly outlined the surgical indications and timing for patients with IE and provided the basis for decisions to proceed with surgery.
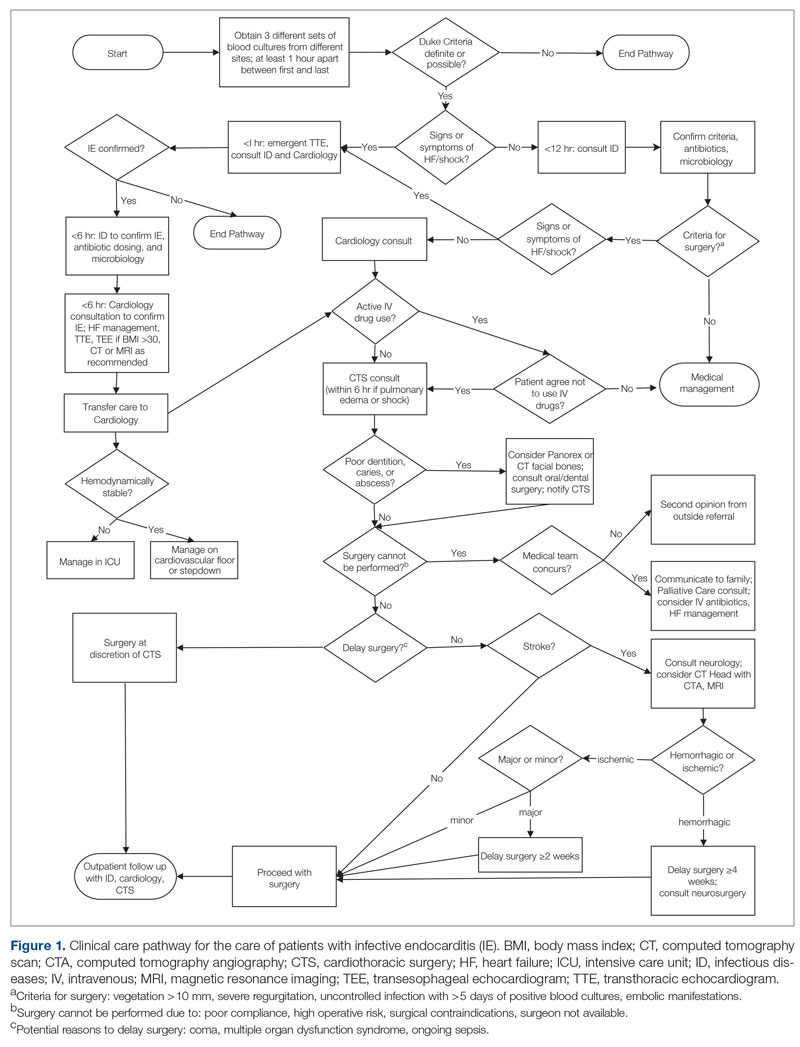
Starting in late 2018, in collaboration with cardiology and CTS teams, ID specialists socialized the clinical pathway to inpatient services that cared for patients with IE. Infectious diseases physicians also provided recurring conferences on the effectiveness of MDTs in IE management and participated in heart-valve team case discussions. Finally, in May 2019, an electronic version of the pathway was embedded in the EHR system using a Cerner PowerChart feature known as Care Pathways. The feature presents the user with algorithm questions in the EHR and provides recommendations, relevant orders, timelines, and other decision support in the clinical pathway. The feature is available to all providers in the health system.

To evaluate the effectiveness of our intervention, we recorded outcomes for patients with IE with surgical indications between December 2018 and June 2020 and compared them with our prior published data from January to December 2016. Cases of IE for the current study period were identified via retrospective chart review. Records from December 2018 to June 2020 were searched using International Statistical Classification of Diseases, Tenth Revision (ICD-10) discharge codes for IE (I33, I33.0, I33.9, I38, I39, M32.11). To select those patients with definitive IE and indications for surgery, the following criteria were applied: age ≥ 18 years; fulfilled modified Duke criteria for definite IE18; and met ≥ 1 American Heart Association (AHA)/Infection Diseases Society of America criteria for recommendation for surgery. Indications for surgery were ≥ 1 of the following: left-sided endocarditis caused by S aureus, fungal, or highly resistant organism; new heart block; annular or aortic abscess; persistent bacteremia or fever despite 5 days of appropriate antimicrobials; vegetation size ≥ 10 mm and evidence of embolic phenomena; recurrence of prosthetic valve infection; recurrent emboli and persistent vegetation despite antimicrobials; and increase in vegetation size despite antimicrobials.16
Age was treated as a categorical variable, using the age groups 18 to 44 years, 45 to 64 years, and 65 years and older. Gender was self-reported. Primary outcomes were surgery or transfer to a higher center for surgery and in-hospital death. Secondary outcomes included consults to teams involved in multidisciplinary care of patients with IE, including ID, cardiology, and CTS. Bivariate analyses were performed using Pearson χ2 tests. Odds ratios for surgery and death were calculated using a multivariate logistic regression model including age and gender covariates. Statistical significance was defined at α = 0.05, and statistical analysis was performed using Stata/IC v16.1 (StataCorp LLC). Our university institutional review board (IRB) reviewed the project (#2010858-QI) and determined that the project was quality-improvement activity, not human subject research, and therefore did not require additional IRB review.
Results
We identified 21 patients in the pre-intervention period and 31 patients in the postintervention period with definitive IE who had guideline indications for surgery. The postintervention cohort was older and had more male patients; this difference was not statistically significant. No differences were noted between the groups for race, gender, or intravenous (IV) drug use (Table 2). Chi-square tests of independence were performed to assess the relationship between age and our primary outcomes. There was a significant relationship between age and the likelihood of receiving or being transferred for surgery (59.3% vs 50% vs 7.7% for 18-44 y, 45-64 y, and ≥ 65 y, respectively; χ2 [2, N = 52] = 9.67; P = .008), but not between age and mortality (14.8% vs 25.0% vs 30.8% for 18-44 y, 45-64 y, and ≥ 65 y, respectively; χ2 = 1.48 [2, N = 52; P = .478]. The electronic version of the clinical pathway was activated and used in only 3 of the 31 patients in the postintervention period. Consultations to ID, cardiology, and CTS teams were compared between the study periods (Table 2). Although more consultations were seen in the postintervention period, differences were not statistically significant.
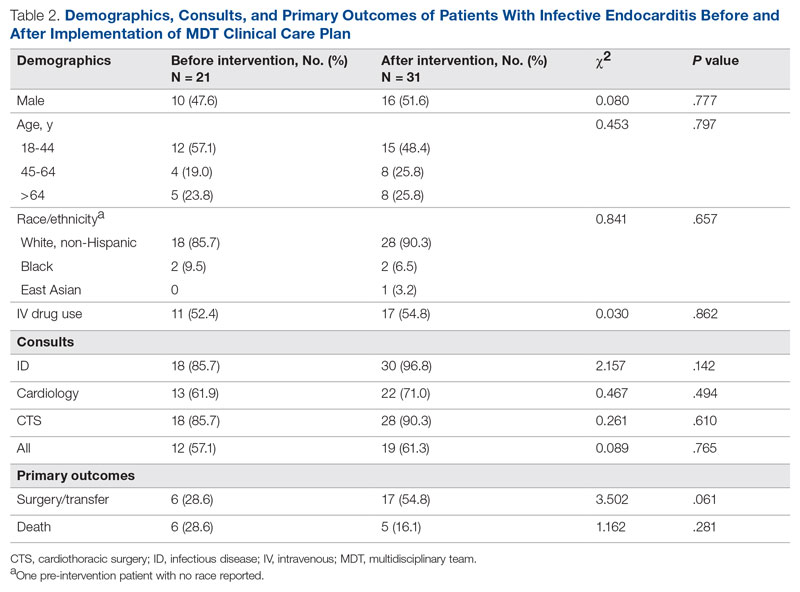
The unadjusted primary outcomes are shown in Table 2. More surgeries were performed per guideline indications, and fewer deaths were noted in the postintervention period than in the pre-intervention period, but the differences were not statistically significant (Table 2).
Because the postintervention period had more male patients and older patients, we evaluated the outcomes using a logistic regression model controlling for both age and gender. The odds of surgery or transfer for surgery for patients in the postintervention period were 4.88 (95% CI, 1.20-19.79; P = .027) as compared with the pre-intervention period, and the odds ratio for death among patients in the postintervention period compared with the pre-intervention period was 0.40 (95% CI, 0.09-1.69; P = .21) (Figure 2).
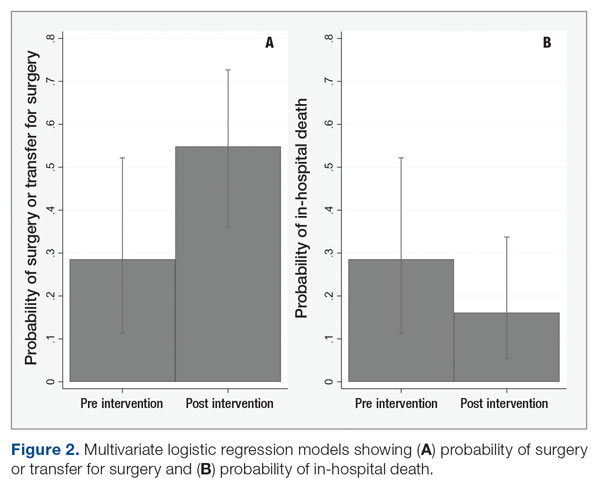
Discussion
In our study, patients with IE with guideline indications for surgery had significantly higher rates of surgery in the postintervention period than in the pre-intervention period. The implementation of an MDT, recurring educational sessions, and efforts to implement and familiarize team members with the clinical pathway approach are the most likely reasons for this change. The increased rates of surgery in the postintervention period were the likely proximate cause of the 60% reduction in in-hospital mortality. This improvement in mortality, while not statistically significant, is very likely to be clinically significant and helps reinforce the value of the MDT intervention used.
Our findings are consistent with existing and mounting literature on the use of MDTs to improve outcomes for patients with IE, including 2 studies that noted an increased rate of surgery for patients with indications.8,19 Several other studies in both Europe and North America have found significant decreases in mortality,6-11,20,21 rates of complications,9 time to diagnosis and treatment,11 and length of stay9,20 for patients with IE managed with an MDT strategy. Although current AHA guidelines for care of patients with IE do suggest an MDT approach, the strategy for this approach is not well established.22 Only 1 study that has implemented a new MDT protocol for care of IE has been conducted in the United States.8
While effective MDTs certainly improve outcomes in patients with IE, there are reported differences in implementation of such an approach. With the MDT model as the core, various implementations included regular case conferences,10,11,19,21,23 formation of a consulting team,6,8 or establishment of a new protocol or algorithm for care.8,9,20 Our approach used a clinical pathway as a basis for improved communication among consulting services, education of learning providers via regular case conferences, and implementation of an electronic clinical care pathway to guide them step by step. Our pathway followed the institutionally standardized algorithm (Figure 1), using what we called the 6 T’s approach (Table 1), that guides providers to evaluate critical cases in a fast track.17
To the best of our knowledge, ours is the first report of an MDT that used an electronic clinical care pathway embedded within the EHR. The electronic version of our clinical pathway went live for only the second half of the postintervention study period, which is the most likely reason for its limited utilization. It is also possible that educational efforts in the first half of the intervention period were sufficient to familiarize providers with the care pathway such that the electronic version was seldom needed. We are exploring other possible ways of improving electronic pathway utilization, such as improving the feature usability and further systemwide educational efforts.
Our study has other limitations. Quasi-experimental before-and-after comparisons are subject to confounding from concurrent interventions. We had a substantial change in cardiothoracic faculty soon after the commencement of our efforts to form the MDT, and thus cannot rule out differences related to their comfort level in considering or offering surgery. We also cannot rule out a Hawthorne effect, where knowledge of the ongoing quality-improvement project changed provider behavior, making surgery more likely. We did not evaluate rates of right- versus left-sided endocarditis, which have been linked to mortality.24 Our study also was performed across a single academic institution, which may limit its generalizability. Finally, our study may not have been adequately powered to detect differences in mortality due to implementation of the MDT approach.
Conclusion
Our work suggests that an MDT for IE can be successfully designed and implemented with a clinical pathway using quality-improvement tools in centers where subspecialty services are available. Our approach was associated with a higher rate of surgery among patients with guideline indications for surgery and may reduce in-hospital mortality. An electronic clinical care pathway embedded in the EHR is feasible and may have a role in MDT implementation.
These data were also accepted as a poster at IDWeek 2022, Washington, DC. The poster abstract is published in an online supplement of Open Forum Infectious Diseases as an abstract publication.
Corresponding author: Haley Crosby; [email protected]
Disclosures: None reported.
1. Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015;132(15):1435-1486. doi:10.1161/cir.0000000000000296
2. Federspiel JJ, Stearns SC, Peppercorn AF, et al. Increasing US rates of endocarditis with Staphylococcus aureus: 1999-2008. Arch Intern Med. 2012;172(4):363-365. doi:10.1001/archinternmed.2011.1027
3. Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(23):e521-e643. doi:10.1161/cir.0000000000000031
4. Chambers J, Sandoe J, Ray S, et al. The infective endocarditis team: recommendations from an international working group. Heart. 2014;100(7):524-527. doi:10.1136/heartjnl-2013-304354
5. Habib G, Lancellotti P, Antunes MJ, et al. 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36(44):3075-3128. doi:10.1093/eurheartj/ehv319
6. Chirillo F, Scotton P, Rocco F, et al. Impact of a multidisciplinary management strategy on the outcome of patients with native valve infective endocarditis. Am J Cardiol. 2013;112(8):1171-1176. doi:10.1016/j.amjcard.2013.05.060
7. Botelho-Nevers E, Thuny F, Casalta JP, et al. Dramatic reduction in infective endocarditis-related mortality with a management-based approach. Arch Intern Med. 2009;169(14):1290-1298. doi:10.1001/archinternmed.2009.192
8. El-Dalati S, Cronin D, Riddell IV J, et al. The clinical impact of implementation of a multidisciplinary endocarditis team. Ann Thorac Surg. 2022;113(1):118-124.
9. Carrasco-Chinchilla F, Sánchez-Espín G, Ruiz-Morales J, et al. Influence of a multidisciplinary alert strategy on mortality due to left-sided infective endocarditis. Rev Esp Cardiol (Engl Ed). 2014;67(5):380-386. doi:10.1016/j.rec.2013.09.010
10. Issa N, Dijos M, Greib C, et al. Impact of an endocarditis team in the management of 357 infective endocarditis [abstract]. Open Forum Infect Dis. 2016;3(suppl 1):S201. doi:10.1093/ofid/ofw172.825
11. Kaura A, Byrne J, Fife A, et al. Inception of the ‘endocarditis team’ is associated with improved survival in patients with infective endocarditis who are managed medically: findings from a before-and-after study. Open Heart. 2017;4(2):e000699. doi:10.1136/openhrt-2017-000699
12. Rotter T, Kinsman L, James E, et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev. 2010;(3):Cd006632. doi:10.1002/14651858.CD006632.pub2
13. Neame MT, Chacko J, Surace AE, et al. A systematic review of the effects of implementing clinical pathways supported by health information technologies. J Am Med Inform Assoc. 2019;26(4):356-363. doi:10.1093/jamia/ocy176
14. Trimarchi L, Caruso R, Magon G, et al. Clinical pathways and patient-related outcomes in hospital-based settings: a systematic review and meta-analysis of randomized controlled trials. Acta Biomed. 2021;92(1):e2021093. doi:10.23750/abm.v92i1.10639
15. Gibbons EF, Huang G, Aldea G, et al. A multidisciplinary pathway for the diagnosis and treatment of infectious endocarditis. Crit Pathw Cardiol. 2020;19(4):187-194. doi:10.1097/hpc.0000000000000224
16. Regunath H, Vasudevan A, Vyas K, et al. A quality improvement initiative: developing a multi-disciplinary team for infective endocarditis. Mo Med. 2019;116(4):291-296.
17. Regunath H, Whitt SP. Multidisciplinary service delivery for the endocarditis patient. In: Infective Endocarditis: A Multidisciplinary Approach. 1st ed. Kilic A, ed. Academic Press; 2022.
18. Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med. 1994;96(3):200-209. doi:10.1016/0002-9343(94)90143-0
19. Tan C, Hansen MS, Cohen G, et al. Case conferences for infective endocarditis: a quality improvement initiative. PLoS One. 2018;13(10):e0205528. doi:10.1371/journal.pone.0205528
20. Ruch Y, Mazzucotelli JP, Lefebvre F, et al. Impact of setting up an “endocarditis team” on the management of infective endocarditis. Open Forum Infect Dis. 2019;6(9):ofz308. doi:10.1093/ofid/ofz308
21. Camou F, Dijos M, Barandon L, et al. Management of infective endocarditis and multidisciplinary approach. Med Mal Infect. 2019;49(1):17-22. doi:10.1016/j.medmal.2018.06.007
22. Pettersson GB, Hussain ST. Current AATS guidelines on surgical treatment of infective endocarditis. Ann Cardiothorac Surg. 2019;8(6):630-644. doi:10.21037/acs.2019.10.05
23. Mestres CA, Paré JC, Miró JM. Organization and functioning of a multidisciplinary team for the diagnosis and treatment of infective endocarditis: a 30-year perspective (1985-2014). Rev Esp Cardiol (Engl Ed). 2015;68(5):363-368. doi:10.1016/j.rec.2014.10.006
24. Stavi V, Brandstaetter E, Sagy I, et al. Comparison of clinical characteristics and prognosis in patients with right- and left-sided infective endocarditis. Rambam Maimonides Med J. 2019;10(1):e00003. doi:10.5041/rmmj.10338
From the University of Missouri School of Medicine, Columbia, MO (Haley Crosby); Department of Clinical Family and Community Medicine, University of Missouri, Columbia, MO (Dr. Pierce); and Department of Medicine, Divisions of Infectious Diseases and Pulmonary, Critical Care and Environmental Medicine, University of Missouri, Columbia, MO, and Divisions of Pulmonary and Critical Care Medicine and Infectious Diseases, University of Maryland Baltimore Washington Medical Center, Glen Burnie, MD (Dr. Regunath).
ABSTRACT
Objective: Multidisciplinary teams (MDTs) improve outcomes for patients with infective endocarditis (IE), but methods of implementation vary. In our academic medical center, we developed an MDT approach guided by a clinical care pathway and assessed outcomes of patients with IE.
Methods: We compared outcomes of patients with IE and indications for surgery between December 2018 and June 2020 with our prior published data for the period January to December 2016. MDT interventions involved recurring conferences with infectious diseases physicians in team meetings and promoting a clinical care pathway to guide providers on steps in management. Primary outcomes were surgery and in-hospital death.
Results: Prior to the intervention, 6 of 21 (28.6%) patients with indications for surgery underwent surgery or were transferred to higher centers for surgery, and 6 (28.6%) patients died. Post intervention, 17 of 31 (54.8%) patients underwent or were transferred for surgery, and 5 (16.1%) died. After adjusting for age and gender, the odds of surgery or transfer for surgery for patients in the postintervention period were 4.88 (95% CI, 1.20-19.79; P = .027) compared with the pre-intervention period. The odds ratio for death among patients in the postintervention period was 0.40 (95% CI, 0.09-1.69; P = .21).
Conclusion: An MDT team approach using a clinical pathway was associated with an increased number of surgeries performed for IE and may lower rates of in-hospital mortality.
Keywords: infective endocarditis, clinical pathway, quality improvement, multidisciplinary team, valve surgery.
Infective endocarditis (IE) is associated with significant morbidity and mortality.1 Rates of IE due to Staphylococcus aureus are increasing in the United States.2 Reported in-hospital mortality from IE ranges from 15% to 20%.3
Clinical pathways are defined as “structured, multidisciplinary plans of care used by health services to detail essential steps in the care of patients with a specific clinical problem.”12 In the modern era, these pathways are often developed and implemented via the electronic health record (EHR) system. Studies of clinical pathways generally demonstrate improvements in patient outcomes, quality of care, or resource utilization.13,14 Clinical pathways represent 1 possible approach to the implementation of a MDT in the care of patients with IE.15
In our earlier work, we used quality improvement principles in the design of an MDT approach to IE care at our institution.16 Despite having indications for surgery, 12 of 21 (57.1%) patients with IE did not undergo surgery, and we identified these missed opportunities for surgery as a leverage point for improvement of outcomes. With input from the various specialties and stakeholders, we developed a clinical pathway (algorithm) for the institutional management of IE that guides next steps in clinical care and their timelines, aiming to reduce by 50% (from 57.1% to 28.6%) the number of patients with IE who do not undergo surgery despite guideline indications for early surgical intervention. In this report, we describe the implementation of this clinical pathway as our MDT approach to the care of patients with IE at our institution.
Methods
The University of Missouri, Columbia, is a tertiary care academic health system with 5 hospitals and more than 60 clinic locations across central Missouri. In the spring of 2018, an MDT was developed, with support from administrative leaders, to improve the care of patients with IE at our institution. The work group prioritized one leverage point to improve IE outcomes, which was improving the number of surgeries performed on those IE patients who had guideline indications for surgery. A clinical pathway was developed around this leverage point (Figure 1). The pathway leveraged the 6 T’s (Table 1) to guide providers through the evaluation and management of IE.17 The pathway focused on improving adherence to standards of care and reduction in practice variation by defining indications for referrals and diagnostic interventions, helping to reduce delays in consultation and diagnosis. The pathway also clearly outlined the surgical indications and timing for patients with IE and provided the basis for decisions to proceed with surgery.

Starting in late 2018, in collaboration with cardiology and CTS teams, ID specialists socialized the clinical pathway to inpatient services that cared for patients with IE. Infectious diseases physicians also provided recurring conferences on the effectiveness of MDTs in IE management and participated in heart-valve team case discussions. Finally, in May 2019, an electronic version of the pathway was embedded in the EHR system using a Cerner PowerChart feature known as Care Pathways. The feature presents the user with algorithm questions in the EHR and provides recommendations, relevant orders, timelines, and other decision support in the clinical pathway. The feature is available to all providers in the health system.

To evaluate the effectiveness of our intervention, we recorded outcomes for patients with IE with surgical indications between December 2018 and June 2020 and compared them with our prior published data from January to December 2016. Cases of IE for the current study period were identified via retrospective chart review. Records from December 2018 to June 2020 were searched using International Statistical Classification of Diseases, Tenth Revision (ICD-10) discharge codes for IE (I33, I33.0, I33.9, I38, I39, M32.11). To select those patients with definitive IE and indications for surgery, the following criteria were applied: age ≥ 18 years; fulfilled modified Duke criteria for definite IE18; and met ≥ 1 American Heart Association (AHA)/Infection Diseases Society of America criteria for recommendation for surgery. Indications for surgery were ≥ 1 of the following: left-sided endocarditis caused by S aureus, fungal, or highly resistant organism; new heart block; annular or aortic abscess; persistent bacteremia or fever despite 5 days of appropriate antimicrobials; vegetation size ≥ 10 mm and evidence of embolic phenomena; recurrence of prosthetic valve infection; recurrent emboli and persistent vegetation despite antimicrobials; and increase in vegetation size despite antimicrobials.16
Age was treated as a categorical variable, using the age groups 18 to 44 years, 45 to 64 years, and 65 years and older. Gender was self-reported. Primary outcomes were surgery or transfer to a higher center for surgery and in-hospital death. Secondary outcomes included consults to teams involved in multidisciplinary care of patients with IE, including ID, cardiology, and CTS. Bivariate analyses were performed using Pearson χ2 tests. Odds ratios for surgery and death were calculated using a multivariate logistic regression model including age and gender covariates. Statistical significance was defined at α = 0.05, and statistical analysis was performed using Stata/IC v16.1 (StataCorp LLC). Our university institutional review board (IRB) reviewed the project (#2010858-QI) and determined that the project was quality-improvement activity, not human subject research, and therefore did not require additional IRB review.
Results
We identified 21 patients in the pre-intervention period and 31 patients in the postintervention period with definitive IE who had guideline indications for surgery. The postintervention cohort was older and had more male patients; this difference was not statistically significant. No differences were noted between the groups for race, gender, or intravenous (IV) drug use (Table 2). Chi-square tests of independence were performed to assess the relationship between age and our primary outcomes. There was a significant relationship between age and the likelihood of receiving or being transferred for surgery (59.3% vs 50% vs 7.7% for 18-44 y, 45-64 y, and ≥ 65 y, respectively; χ2 [2, N = 52] = 9.67; P = .008), but not between age and mortality (14.8% vs 25.0% vs 30.8% for 18-44 y, 45-64 y, and ≥ 65 y, respectively; χ2 = 1.48 [2, N = 52; P = .478]. The electronic version of the clinical pathway was activated and used in only 3 of the 31 patients in the postintervention period. Consultations to ID, cardiology, and CTS teams were compared between the study periods (Table 2). Although more consultations were seen in the postintervention period, differences were not statistically significant.

The unadjusted primary outcomes are shown in Table 2. More surgeries were performed per guideline indications, and fewer deaths were noted in the postintervention period than in the pre-intervention period, but the differences were not statistically significant (Table 2).
Because the postintervention period had more male patients and older patients, we evaluated the outcomes using a logistic regression model controlling for both age and gender. The odds of surgery or transfer for surgery for patients in the postintervention period were 4.88 (95% CI, 1.20-19.79; P = .027) as compared with the pre-intervention period, and the odds ratio for death among patients in the postintervention period compared with the pre-intervention period was 0.40 (95% CI, 0.09-1.69; P = .21) (Figure 2).

Discussion
In our study, patients with IE with guideline indications for surgery had significantly higher rates of surgery in the postintervention period than in the pre-intervention period. The implementation of an MDT, recurring educational sessions, and efforts to implement and familiarize team members with the clinical pathway approach are the most likely reasons for this change. The increased rates of surgery in the postintervention period were the likely proximate cause of the 60% reduction in in-hospital mortality. This improvement in mortality, while not statistically significant, is very likely to be clinically significant and helps reinforce the value of the MDT intervention used.
Our findings are consistent with existing and mounting literature on the use of MDTs to improve outcomes for patients with IE, including 2 studies that noted an increased rate of surgery for patients with indications.8,19 Several other studies in both Europe and North America have found significant decreases in mortality,6-11,20,21 rates of complications,9 time to diagnosis and treatment,11 and length of stay9,20 for patients with IE managed with an MDT strategy. Although current AHA guidelines for care of patients with IE do suggest an MDT approach, the strategy for this approach is not well established.22 Only 1 study that has implemented a new MDT protocol for care of IE has been conducted in the United States.8
While effective MDTs certainly improve outcomes in patients with IE, there are reported differences in implementation of such an approach. With the MDT model as the core, various implementations included regular case conferences,10,11,19,21,23 formation of a consulting team,6,8 or establishment of a new protocol or algorithm for care.8,9,20 Our approach used a clinical pathway as a basis for improved communication among consulting services, education of learning providers via regular case conferences, and implementation of an electronic clinical care pathway to guide them step by step. Our pathway followed the institutionally standardized algorithm (Figure 1), using what we called the 6 T’s approach (Table 1), that guides providers to evaluate critical cases in a fast track.17
To the best of our knowledge, ours is the first report of an MDT that used an electronic clinical care pathway embedded within the EHR. The electronic version of our clinical pathway went live for only the second half of the postintervention study period, which is the most likely reason for its limited utilization. It is also possible that educational efforts in the first half of the intervention period were sufficient to familiarize providers with the care pathway such that the electronic version was seldom needed. We are exploring other possible ways of improving electronic pathway utilization, such as improving the feature usability and further systemwide educational efforts.
Our study has other limitations. Quasi-experimental before-and-after comparisons are subject to confounding from concurrent interventions. We had a substantial change in cardiothoracic faculty soon after the commencement of our efforts to form the MDT, and thus cannot rule out differences related to their comfort level in considering or offering surgery. We also cannot rule out a Hawthorne effect, where knowledge of the ongoing quality-improvement project changed provider behavior, making surgery more likely. We did not evaluate rates of right- versus left-sided endocarditis, which have been linked to mortality.24 Our study also was performed across a single academic institution, which may limit its generalizability. Finally, our study may not have been adequately powered to detect differences in mortality due to implementation of the MDT approach.
Conclusion
Our work suggests that an MDT for IE can be successfully designed and implemented with a clinical pathway using quality-improvement tools in centers where subspecialty services are available. Our approach was associated with a higher rate of surgery among patients with guideline indications for surgery and may reduce in-hospital mortality. An electronic clinical care pathway embedded in the EHR is feasible and may have a role in MDT implementation.
These data were also accepted as a poster at IDWeek 2022, Washington, DC. The poster abstract is published in an online supplement of Open Forum Infectious Diseases as an abstract publication.
Corresponding author: Haley Crosby; [email protected]
Disclosures: None reported.
From the University of Missouri School of Medicine, Columbia, MO (Haley Crosby); Department of Clinical Family and Community Medicine, University of Missouri, Columbia, MO (Dr. Pierce); and Department of Medicine, Divisions of Infectious Diseases and Pulmonary, Critical Care and Environmental Medicine, University of Missouri, Columbia, MO, and Divisions of Pulmonary and Critical Care Medicine and Infectious Diseases, University of Maryland Baltimore Washington Medical Center, Glen Burnie, MD (Dr. Regunath).
ABSTRACT
Objective: Multidisciplinary teams (MDTs) improve outcomes for patients with infective endocarditis (IE), but methods of implementation vary. In our academic medical center, we developed an MDT approach guided by a clinical care pathway and assessed outcomes of patients with IE.
Methods: We compared outcomes of patients with IE and indications for surgery between December 2018 and June 2020 with our prior published data for the period January to December 2016. MDT interventions involved recurring conferences with infectious diseases physicians in team meetings and promoting a clinical care pathway to guide providers on steps in management. Primary outcomes were surgery and in-hospital death.
Results: Prior to the intervention, 6 of 21 (28.6%) patients with indications for surgery underwent surgery or were transferred to higher centers for surgery, and 6 (28.6%) patients died. Post intervention, 17 of 31 (54.8%) patients underwent or were transferred for surgery, and 5 (16.1%) died. After adjusting for age and gender, the odds of surgery or transfer for surgery for patients in the postintervention period were 4.88 (95% CI, 1.20-19.79; P = .027) compared with the pre-intervention period. The odds ratio for death among patients in the postintervention period was 0.40 (95% CI, 0.09-1.69; P = .21).
Conclusion: An MDT team approach using a clinical pathway was associated with an increased number of surgeries performed for IE and may lower rates of in-hospital mortality.
Keywords: infective endocarditis, clinical pathway, quality improvement, multidisciplinary team, valve surgery.
Infective endocarditis (IE) is associated with significant morbidity and mortality.1 Rates of IE due to Staphylococcus aureus are increasing in the United States.2 Reported in-hospital mortality from IE ranges from 15% to 20%.3
Clinical pathways are defined as “structured, multidisciplinary plans of care used by health services to detail essential steps in the care of patients with a specific clinical problem.”12 In the modern era, these pathways are often developed and implemented via the electronic health record (EHR) system. Studies of clinical pathways generally demonstrate improvements in patient outcomes, quality of care, or resource utilization.13,14 Clinical pathways represent 1 possible approach to the implementation of a MDT in the care of patients with IE.15
In our earlier work, we used quality improvement principles in the design of an MDT approach to IE care at our institution.16 Despite having indications for surgery, 12 of 21 (57.1%) patients with IE did not undergo surgery, and we identified these missed opportunities for surgery as a leverage point for improvement of outcomes. With input from the various specialties and stakeholders, we developed a clinical pathway (algorithm) for the institutional management of IE that guides next steps in clinical care and their timelines, aiming to reduce by 50% (from 57.1% to 28.6%) the number of patients with IE who do not undergo surgery despite guideline indications for early surgical intervention. In this report, we describe the implementation of this clinical pathway as our MDT approach to the care of patients with IE at our institution.
Methods
The University of Missouri, Columbia, is a tertiary care academic health system with 5 hospitals and more than 60 clinic locations across central Missouri. In the spring of 2018, an MDT was developed, with support from administrative leaders, to improve the care of patients with IE at our institution. The work group prioritized one leverage point to improve IE outcomes, which was improving the number of surgeries performed on those IE patients who had guideline indications for surgery. A clinical pathway was developed around this leverage point (Figure 1). The pathway leveraged the 6 T’s (Table 1) to guide providers through the evaluation and management of IE.17 The pathway focused on improving adherence to standards of care and reduction in practice variation by defining indications for referrals and diagnostic interventions, helping to reduce delays in consultation and diagnosis. The pathway also clearly outlined the surgical indications and timing for patients with IE and provided the basis for decisions to proceed with surgery.

Starting in late 2018, in collaboration with cardiology and CTS teams, ID specialists socialized the clinical pathway to inpatient services that cared for patients with IE. Infectious diseases physicians also provided recurring conferences on the effectiveness of MDTs in IE management and participated in heart-valve team case discussions. Finally, in May 2019, an electronic version of the pathway was embedded in the EHR system using a Cerner PowerChart feature known as Care Pathways. The feature presents the user with algorithm questions in the EHR and provides recommendations, relevant orders, timelines, and other decision support in the clinical pathway. The feature is available to all providers in the health system.

To evaluate the effectiveness of our intervention, we recorded outcomes for patients with IE with surgical indications between December 2018 and June 2020 and compared them with our prior published data from January to December 2016. Cases of IE for the current study period were identified via retrospective chart review. Records from December 2018 to June 2020 were searched using International Statistical Classification of Diseases, Tenth Revision (ICD-10) discharge codes for IE (I33, I33.0, I33.9, I38, I39, M32.11). To select those patients with definitive IE and indications for surgery, the following criteria were applied: age ≥ 18 years; fulfilled modified Duke criteria for definite IE18; and met ≥ 1 American Heart Association (AHA)/Infection Diseases Society of America criteria for recommendation for surgery. Indications for surgery were ≥ 1 of the following: left-sided endocarditis caused by S aureus, fungal, or highly resistant organism; new heart block; annular or aortic abscess; persistent bacteremia or fever despite 5 days of appropriate antimicrobials; vegetation size ≥ 10 mm and evidence of embolic phenomena; recurrence of prosthetic valve infection; recurrent emboli and persistent vegetation despite antimicrobials; and increase in vegetation size despite antimicrobials.16
Age was treated as a categorical variable, using the age groups 18 to 44 years, 45 to 64 years, and 65 years and older. Gender was self-reported. Primary outcomes were surgery or transfer to a higher center for surgery and in-hospital death. Secondary outcomes included consults to teams involved in multidisciplinary care of patients with IE, including ID, cardiology, and CTS. Bivariate analyses were performed using Pearson χ2 tests. Odds ratios for surgery and death were calculated using a multivariate logistic regression model including age and gender covariates. Statistical significance was defined at α = 0.05, and statistical analysis was performed using Stata/IC v16.1 (StataCorp LLC). Our university institutional review board (IRB) reviewed the project (#2010858-QI) and determined that the project was quality-improvement activity, not human subject research, and therefore did not require additional IRB review.
Results
We identified 21 patients in the pre-intervention period and 31 patients in the postintervention period with definitive IE who had guideline indications for surgery. The postintervention cohort was older and had more male patients; this difference was not statistically significant. No differences were noted between the groups for race, gender, or intravenous (IV) drug use (Table 2). Chi-square tests of independence were performed to assess the relationship between age and our primary outcomes. There was a significant relationship between age and the likelihood of receiving or being transferred for surgery (59.3% vs 50% vs 7.7% for 18-44 y, 45-64 y, and ≥ 65 y, respectively; χ2 [2, N = 52] = 9.67; P = .008), but not between age and mortality (14.8% vs 25.0% vs 30.8% for 18-44 y, 45-64 y, and ≥ 65 y, respectively; χ2 = 1.48 [2, N = 52; P = .478]. The electronic version of the clinical pathway was activated and used in only 3 of the 31 patients in the postintervention period. Consultations to ID, cardiology, and CTS teams were compared between the study periods (Table 2). Although more consultations were seen in the postintervention period, differences were not statistically significant.

The unadjusted primary outcomes are shown in Table 2. More surgeries were performed per guideline indications, and fewer deaths were noted in the postintervention period than in the pre-intervention period, but the differences were not statistically significant (Table 2).
Because the postintervention period had more male patients and older patients, we evaluated the outcomes using a logistic regression model controlling for both age and gender. The odds of surgery or transfer for surgery for patients in the postintervention period were 4.88 (95% CI, 1.20-19.79; P = .027) as compared with the pre-intervention period, and the odds ratio for death among patients in the postintervention period compared with the pre-intervention period was 0.40 (95% CI, 0.09-1.69; P = .21) (Figure 2).

Discussion
In our study, patients with IE with guideline indications for surgery had significantly higher rates of surgery in the postintervention period than in the pre-intervention period. The implementation of an MDT, recurring educational sessions, and efforts to implement and familiarize team members with the clinical pathway approach are the most likely reasons for this change. The increased rates of surgery in the postintervention period were the likely proximate cause of the 60% reduction in in-hospital mortality. This improvement in mortality, while not statistically significant, is very likely to be clinically significant and helps reinforce the value of the MDT intervention used.
Our findings are consistent with existing and mounting literature on the use of MDTs to improve outcomes for patients with IE, including 2 studies that noted an increased rate of surgery for patients with indications.8,19 Several other studies in both Europe and North America have found significant decreases in mortality,6-11,20,21 rates of complications,9 time to diagnosis and treatment,11 and length of stay9,20 for patients with IE managed with an MDT strategy. Although current AHA guidelines for care of patients with IE do suggest an MDT approach, the strategy for this approach is not well established.22 Only 1 study that has implemented a new MDT protocol for care of IE has been conducted in the United States.8
While effective MDTs certainly improve outcomes in patients with IE, there are reported differences in implementation of such an approach. With the MDT model as the core, various implementations included regular case conferences,10,11,19,21,23 formation of a consulting team,6,8 or establishment of a new protocol or algorithm for care.8,9,20 Our approach used a clinical pathway as a basis for improved communication among consulting services, education of learning providers via regular case conferences, and implementation of an electronic clinical care pathway to guide them step by step. Our pathway followed the institutionally standardized algorithm (Figure 1), using what we called the 6 T’s approach (Table 1), that guides providers to evaluate critical cases in a fast track.17
To the best of our knowledge, ours is the first report of an MDT that used an electronic clinical care pathway embedded within the EHR. The electronic version of our clinical pathway went live for only the second half of the postintervention study period, which is the most likely reason for its limited utilization. It is also possible that educational efforts in the first half of the intervention period were sufficient to familiarize providers with the care pathway such that the electronic version was seldom needed. We are exploring other possible ways of improving electronic pathway utilization, such as improving the feature usability and further systemwide educational efforts.
Our study has other limitations. Quasi-experimental before-and-after comparisons are subject to confounding from concurrent interventions. We had a substantial change in cardiothoracic faculty soon after the commencement of our efforts to form the MDT, and thus cannot rule out differences related to their comfort level in considering or offering surgery. We also cannot rule out a Hawthorne effect, where knowledge of the ongoing quality-improvement project changed provider behavior, making surgery more likely. We did not evaluate rates of right- versus left-sided endocarditis, which have been linked to mortality.24 Our study also was performed across a single academic institution, which may limit its generalizability. Finally, our study may not have been adequately powered to detect differences in mortality due to implementation of the MDT approach.
Conclusion
Our work suggests that an MDT for IE can be successfully designed and implemented with a clinical pathway using quality-improvement tools in centers where subspecialty services are available. Our approach was associated with a higher rate of surgery among patients with guideline indications for surgery and may reduce in-hospital mortality. An electronic clinical care pathway embedded in the EHR is feasible and may have a role in MDT implementation.
These data were also accepted as a poster at IDWeek 2022, Washington, DC. The poster abstract is published in an online supplement of Open Forum Infectious Diseases as an abstract publication.
Corresponding author: Haley Crosby; [email protected]
Disclosures: None reported.
1. Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015;132(15):1435-1486. doi:10.1161/cir.0000000000000296
2. Federspiel JJ, Stearns SC, Peppercorn AF, et al. Increasing US rates of endocarditis with Staphylococcus aureus: 1999-2008. Arch Intern Med. 2012;172(4):363-365. doi:10.1001/archinternmed.2011.1027
3. Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(23):e521-e643. doi:10.1161/cir.0000000000000031
4. Chambers J, Sandoe J, Ray S, et al. The infective endocarditis team: recommendations from an international working group. Heart. 2014;100(7):524-527. doi:10.1136/heartjnl-2013-304354
5. Habib G, Lancellotti P, Antunes MJ, et al. 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36(44):3075-3128. doi:10.1093/eurheartj/ehv319
6. Chirillo F, Scotton P, Rocco F, et al. Impact of a multidisciplinary management strategy on the outcome of patients with native valve infective endocarditis. Am J Cardiol. 2013;112(8):1171-1176. doi:10.1016/j.amjcard.2013.05.060
7. Botelho-Nevers E, Thuny F, Casalta JP, et al. Dramatic reduction in infective endocarditis-related mortality with a management-based approach. Arch Intern Med. 2009;169(14):1290-1298. doi:10.1001/archinternmed.2009.192
8. El-Dalati S, Cronin D, Riddell IV J, et al. The clinical impact of implementation of a multidisciplinary endocarditis team. Ann Thorac Surg. 2022;113(1):118-124.
9. Carrasco-Chinchilla F, Sánchez-Espín G, Ruiz-Morales J, et al. Influence of a multidisciplinary alert strategy on mortality due to left-sided infective endocarditis. Rev Esp Cardiol (Engl Ed). 2014;67(5):380-386. doi:10.1016/j.rec.2013.09.010
10. Issa N, Dijos M, Greib C, et al. Impact of an endocarditis team in the management of 357 infective endocarditis [abstract]. Open Forum Infect Dis. 2016;3(suppl 1):S201. doi:10.1093/ofid/ofw172.825
11. Kaura A, Byrne J, Fife A, et al. Inception of the ‘endocarditis team’ is associated with improved survival in patients with infective endocarditis who are managed medically: findings from a before-and-after study. Open Heart. 2017;4(2):e000699. doi:10.1136/openhrt-2017-000699
12. Rotter T, Kinsman L, James E, et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev. 2010;(3):Cd006632. doi:10.1002/14651858.CD006632.pub2
13. Neame MT, Chacko J, Surace AE, et al. A systematic review of the effects of implementing clinical pathways supported by health information technologies. J Am Med Inform Assoc. 2019;26(4):356-363. doi:10.1093/jamia/ocy176
14. Trimarchi L, Caruso R, Magon G, et al. Clinical pathways and patient-related outcomes in hospital-based settings: a systematic review and meta-analysis of randomized controlled trials. Acta Biomed. 2021;92(1):e2021093. doi:10.23750/abm.v92i1.10639
15. Gibbons EF, Huang G, Aldea G, et al. A multidisciplinary pathway for the diagnosis and treatment of infectious endocarditis. Crit Pathw Cardiol. 2020;19(4):187-194. doi:10.1097/hpc.0000000000000224
16. Regunath H, Vasudevan A, Vyas K, et al. A quality improvement initiative: developing a multi-disciplinary team for infective endocarditis. Mo Med. 2019;116(4):291-296.
17. Regunath H, Whitt SP. Multidisciplinary service delivery for the endocarditis patient. In: Infective Endocarditis: A Multidisciplinary Approach. 1st ed. Kilic A, ed. Academic Press; 2022.
18. Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med. 1994;96(3):200-209. doi:10.1016/0002-9343(94)90143-0
19. Tan C, Hansen MS, Cohen G, et al. Case conferences for infective endocarditis: a quality improvement initiative. PLoS One. 2018;13(10):e0205528. doi:10.1371/journal.pone.0205528
20. Ruch Y, Mazzucotelli JP, Lefebvre F, et al. Impact of setting up an “endocarditis team” on the management of infective endocarditis. Open Forum Infect Dis. 2019;6(9):ofz308. doi:10.1093/ofid/ofz308
21. Camou F, Dijos M, Barandon L, et al. Management of infective endocarditis and multidisciplinary approach. Med Mal Infect. 2019;49(1):17-22. doi:10.1016/j.medmal.2018.06.007
22. Pettersson GB, Hussain ST. Current AATS guidelines on surgical treatment of infective endocarditis. Ann Cardiothorac Surg. 2019;8(6):630-644. doi:10.21037/acs.2019.10.05
23. Mestres CA, Paré JC, Miró JM. Organization and functioning of a multidisciplinary team for the diagnosis and treatment of infective endocarditis: a 30-year perspective (1985-2014). Rev Esp Cardiol (Engl Ed). 2015;68(5):363-368. doi:10.1016/j.rec.2014.10.006
24. Stavi V, Brandstaetter E, Sagy I, et al. Comparison of clinical characteristics and prognosis in patients with right- and left-sided infective endocarditis. Rambam Maimonides Med J. 2019;10(1):e00003. doi:10.5041/rmmj.10338
1. Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015;132(15):1435-1486. doi:10.1161/cir.0000000000000296
2. Federspiel JJ, Stearns SC, Peppercorn AF, et al. Increasing US rates of endocarditis with Staphylococcus aureus: 1999-2008. Arch Intern Med. 2012;172(4):363-365. doi:10.1001/archinternmed.2011.1027
3. Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(23):e521-e643. doi:10.1161/cir.0000000000000031
4. Chambers J, Sandoe J, Ray S, et al. The infective endocarditis team: recommendations from an international working group. Heart. 2014;100(7):524-527. doi:10.1136/heartjnl-2013-304354
5. Habib G, Lancellotti P, Antunes MJ, et al. 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36(44):3075-3128. doi:10.1093/eurheartj/ehv319
6. Chirillo F, Scotton P, Rocco F, et al. Impact of a multidisciplinary management strategy on the outcome of patients with native valve infective endocarditis. Am J Cardiol. 2013;112(8):1171-1176. doi:10.1016/j.amjcard.2013.05.060
7. Botelho-Nevers E, Thuny F, Casalta JP, et al. Dramatic reduction in infective endocarditis-related mortality with a management-based approach. Arch Intern Med. 2009;169(14):1290-1298. doi:10.1001/archinternmed.2009.192
8. El-Dalati S, Cronin D, Riddell IV J, et al. The clinical impact of implementation of a multidisciplinary endocarditis team. Ann Thorac Surg. 2022;113(1):118-124.
9. Carrasco-Chinchilla F, Sánchez-Espín G, Ruiz-Morales J, et al. Influence of a multidisciplinary alert strategy on mortality due to left-sided infective endocarditis. Rev Esp Cardiol (Engl Ed). 2014;67(5):380-386. doi:10.1016/j.rec.2013.09.010
10. Issa N, Dijos M, Greib C, et al. Impact of an endocarditis team in the management of 357 infective endocarditis [abstract]. Open Forum Infect Dis. 2016;3(suppl 1):S201. doi:10.1093/ofid/ofw172.825
11. Kaura A, Byrne J, Fife A, et al. Inception of the ‘endocarditis team’ is associated with improved survival in patients with infective endocarditis who are managed medically: findings from a before-and-after study. Open Heart. 2017;4(2):e000699. doi:10.1136/openhrt-2017-000699
12. Rotter T, Kinsman L, James E, et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev. 2010;(3):Cd006632. doi:10.1002/14651858.CD006632.pub2
13. Neame MT, Chacko J, Surace AE, et al. A systematic review of the effects of implementing clinical pathways supported by health information technologies. J Am Med Inform Assoc. 2019;26(4):356-363. doi:10.1093/jamia/ocy176
14. Trimarchi L, Caruso R, Magon G, et al. Clinical pathways and patient-related outcomes in hospital-based settings: a systematic review and meta-analysis of randomized controlled trials. Acta Biomed. 2021;92(1):e2021093. doi:10.23750/abm.v92i1.10639
15. Gibbons EF, Huang G, Aldea G, et al. A multidisciplinary pathway for the diagnosis and treatment of infectious endocarditis. Crit Pathw Cardiol. 2020;19(4):187-194. doi:10.1097/hpc.0000000000000224
16. Regunath H, Vasudevan A, Vyas K, et al. A quality improvement initiative: developing a multi-disciplinary team for infective endocarditis. Mo Med. 2019;116(4):291-296.
17. Regunath H, Whitt SP. Multidisciplinary service delivery for the endocarditis patient. In: Infective Endocarditis: A Multidisciplinary Approach. 1st ed. Kilic A, ed. Academic Press; 2022.
18. Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med. 1994;96(3):200-209. doi:10.1016/0002-9343(94)90143-0
19. Tan C, Hansen MS, Cohen G, et al. Case conferences for infective endocarditis: a quality improvement initiative. PLoS One. 2018;13(10):e0205528. doi:10.1371/journal.pone.0205528
20. Ruch Y, Mazzucotelli JP, Lefebvre F, et al. Impact of setting up an “endocarditis team” on the management of infective endocarditis. Open Forum Infect Dis. 2019;6(9):ofz308. doi:10.1093/ofid/ofz308
21. Camou F, Dijos M, Barandon L, et al. Management of infective endocarditis and multidisciplinary approach. Med Mal Infect. 2019;49(1):17-22. doi:10.1016/j.medmal.2018.06.007
22. Pettersson GB, Hussain ST. Current AATS guidelines on surgical treatment of infective endocarditis. Ann Cardiothorac Surg. 2019;8(6):630-644. doi:10.21037/acs.2019.10.05
23. Mestres CA, Paré JC, Miró JM. Organization and functioning of a multidisciplinary team for the diagnosis and treatment of infective endocarditis: a 30-year perspective (1985-2014). Rev Esp Cardiol (Engl Ed). 2015;68(5):363-368. doi:10.1016/j.rec.2014.10.006
24. Stavi V, Brandstaetter E, Sagy I, et al. Comparison of clinical characteristics and prognosis in patients with right- and left-sided infective endocarditis. Rambam Maimonides Med J. 2019;10(1):e00003. doi:10.5041/rmmj.10338
High school athletes sustaining worse injuries
High school students are injuring themselves more severely even as overall injury rates have declined, according to a new study presented at the annual meeting of the American Academy of Orthopaedic Surgeons.
The study compared injuries from a 4-year period ending in 2019 to data from 2005 and 2006. The overall rate of injuries dropped 9%, from 2.51 injuries per 1,000 athletic games or practices to 2.29 per 1,000; injuries requiring less than 1 week of recovery time fell by 13%. But, the number of head and neck injuries increased by 10%, injuries requiring surgery increased by 1%, and injuries leading to medical disqualification jumped by 11%.
“It’s wonderful that the injury rate is declining,” said Jordan Neoma Pizzarro, a medical student at George Washington University, Washington, who led the study. “But the data does suggest that the injuries that are happening are worse.”
The increases may also reflect increased education and awareness of how to detect concussions and other injuries that need medical attention, said Micah Lissy, MD, MS, an orthopedic surgeon specializing in sports medicine at Michigan State University, East Lansing. Dr. Lissy cautioned against physicians and others taking the data at face value.
“We need to be implementing preventive measures wherever possible, but I think we can also consider that there may be some confounding factors in the data,” Dr. Lissy told this news organization.
Ms. Pizzarro and her team analyzed data collected from athletic trainers at 100 high schools across the country for the ongoing National Health School Sports-Related Injury Surveillance Study.
Athletes participating in sports such as football, soccer, basketball, volleyball, and softball were included in the analysis. Trainers report the number of injuries for every competition and practice, also known as “athletic exposures.”
Boys’ football carried the highest injury rate, with 3.96 injuries per 1,000 AEs, amounting to 44% of all injuries reported. Girls’ soccer and boys’ wrestling followed, with injury rates of 2.65 and 1.56, respectively.
Sprains and strains accounted for 37% of injuries, followed by concussions (21.6%). The head and/or face was the most injured body site, followed by the ankles and/or knees. Most injuries took place during competitions rather than in practices (relative risk, 3.39; 95% confidence interval, 3.28-3.49; P < .05).
Ms. Pizzarro said that an overall increase in intensity, physical contact, and collisions may account for the spike in more severe injuries.
“Kids are encouraged to specialize in one sport early on and stick with it year-round,” she said. “They’re probably becoming more agile and better athletes, but they’re probably also getting more competitive.”
Dr. Lissy, who has worked with high school athletes as a surgeon, physical therapist, athletic trainer, and coach, said that some of the increases in severity of injuries may reflect trends in sports over the past two decades: Student athletes have become stronger and faster and have put on more muscle mass.
“When you have something that’s much larger, moving much faster and with more force, you’re going to have more force when you bump into things,” he said. “This can lead to more significant injuries.”
The study was independently supported. Study authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
High school students are injuring themselves more severely even as overall injury rates have declined, according to a new study presented at the annual meeting of the American Academy of Orthopaedic Surgeons.
The study compared injuries from a 4-year period ending in 2019 to data from 2005 and 2006. The overall rate of injuries dropped 9%, from 2.51 injuries per 1,000 athletic games or practices to 2.29 per 1,000; injuries requiring less than 1 week of recovery time fell by 13%. But, the number of head and neck injuries increased by 10%, injuries requiring surgery increased by 1%, and injuries leading to medical disqualification jumped by 11%.
“It’s wonderful that the injury rate is declining,” said Jordan Neoma Pizzarro, a medical student at George Washington University, Washington, who led the study. “But the data does suggest that the injuries that are happening are worse.”
The increases may also reflect increased education and awareness of how to detect concussions and other injuries that need medical attention, said Micah Lissy, MD, MS, an orthopedic surgeon specializing in sports medicine at Michigan State University, East Lansing. Dr. Lissy cautioned against physicians and others taking the data at face value.
“We need to be implementing preventive measures wherever possible, but I think we can also consider that there may be some confounding factors in the data,” Dr. Lissy told this news organization.
Ms. Pizzarro and her team analyzed data collected from athletic trainers at 100 high schools across the country for the ongoing National Health School Sports-Related Injury Surveillance Study.
Athletes participating in sports such as football, soccer, basketball, volleyball, and softball were included in the analysis. Trainers report the number of injuries for every competition and practice, also known as “athletic exposures.”
Boys’ football carried the highest injury rate, with 3.96 injuries per 1,000 AEs, amounting to 44% of all injuries reported. Girls’ soccer and boys’ wrestling followed, with injury rates of 2.65 and 1.56, respectively.
Sprains and strains accounted for 37% of injuries, followed by concussions (21.6%). The head and/or face was the most injured body site, followed by the ankles and/or knees. Most injuries took place during competitions rather than in practices (relative risk, 3.39; 95% confidence interval, 3.28-3.49; P < .05).
Ms. Pizzarro said that an overall increase in intensity, physical contact, and collisions may account for the spike in more severe injuries.
“Kids are encouraged to specialize in one sport early on and stick with it year-round,” she said. “They’re probably becoming more agile and better athletes, but they’re probably also getting more competitive.”
Dr. Lissy, who has worked with high school athletes as a surgeon, physical therapist, athletic trainer, and coach, said that some of the increases in severity of injuries may reflect trends in sports over the past two decades: Student athletes have become stronger and faster and have put on more muscle mass.
“When you have something that’s much larger, moving much faster and with more force, you’re going to have more force when you bump into things,” he said. “This can lead to more significant injuries.”
The study was independently supported. Study authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
High school students are injuring themselves more severely even as overall injury rates have declined, according to a new study presented at the annual meeting of the American Academy of Orthopaedic Surgeons.
The study compared injuries from a 4-year period ending in 2019 to data from 2005 and 2006. The overall rate of injuries dropped 9%, from 2.51 injuries per 1,000 athletic games or practices to 2.29 per 1,000; injuries requiring less than 1 week of recovery time fell by 13%. But, the number of head and neck injuries increased by 10%, injuries requiring surgery increased by 1%, and injuries leading to medical disqualification jumped by 11%.
“It’s wonderful that the injury rate is declining,” said Jordan Neoma Pizzarro, a medical student at George Washington University, Washington, who led the study. “But the data does suggest that the injuries that are happening are worse.”
The increases may also reflect increased education and awareness of how to detect concussions and other injuries that need medical attention, said Micah Lissy, MD, MS, an orthopedic surgeon specializing in sports medicine at Michigan State University, East Lansing. Dr. Lissy cautioned against physicians and others taking the data at face value.
“We need to be implementing preventive measures wherever possible, but I think we can also consider that there may be some confounding factors in the data,” Dr. Lissy told this news organization.
Ms. Pizzarro and her team analyzed data collected from athletic trainers at 100 high schools across the country for the ongoing National Health School Sports-Related Injury Surveillance Study.
Athletes participating in sports such as football, soccer, basketball, volleyball, and softball were included in the analysis. Trainers report the number of injuries for every competition and practice, also known as “athletic exposures.”
Boys’ football carried the highest injury rate, with 3.96 injuries per 1,000 AEs, amounting to 44% of all injuries reported. Girls’ soccer and boys’ wrestling followed, with injury rates of 2.65 and 1.56, respectively.
Sprains and strains accounted for 37% of injuries, followed by concussions (21.6%). The head and/or face was the most injured body site, followed by the ankles and/or knees. Most injuries took place during competitions rather than in practices (relative risk, 3.39; 95% confidence interval, 3.28-3.49; P < .05).
Ms. Pizzarro said that an overall increase in intensity, physical contact, and collisions may account for the spike in more severe injuries.
“Kids are encouraged to specialize in one sport early on and stick with it year-round,” she said. “They’re probably becoming more agile and better athletes, but they’re probably also getting more competitive.”
Dr. Lissy, who has worked with high school athletes as a surgeon, physical therapist, athletic trainer, and coach, said that some of the increases in severity of injuries may reflect trends in sports over the past two decades: Student athletes have become stronger and faster and have put on more muscle mass.
“When you have something that’s much larger, moving much faster and with more force, you’re going to have more force when you bump into things,” he said. “This can lead to more significant injuries.”
The study was independently supported. Study authors report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Robotic peritoneal vaginoplasty
When the Food and Drug Administration first approved the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, Calif.) for adult use in 2000, it altered the face of minimally invasive surgery across a multitude of specialties. Improved three-dimensional visualization and enhanced instrument articulation facilitates complex dissections and intracorporeal suturing. While the standard of care for gender-affirming vaginoplasty remains the single-stage penile inversion vaginoplasty, robotic procedures are quickly emerging as alternative options for both primary and revisional surgeries.
The single-stage penile inversion vaginoplasty requires an adequate amount of penoscrotal tissue not only to line a neovaginal canal that measures 12-15 cm, but also to create external vulvar structures. While this is often sufficient in most candidates, there is an increasing number of patients who are receiving puberty blockers, resulting in penoscrotal hypoplasia.
Alternatively, there are patients who experience loss of vaginal depth and vaginal stenosis who seek revisional surgeries. Additional donor sites for skin grafting are available and include the lower abdomen and thighs, although patients may not want these donor site scars. With these donor sites, there is also concern about graft contracture, which could lead to recurrent vaginal stenosis.1 Robotic peritoneal vaginoplasty and robotic enteric vaginoplasty can serve as additional options for patients seeking revisional surgery or who have insufficient genital skin. One benefit of using peritoneal flaps is that they are hairless and are well vascularized with minimal donor site morbidity.1 Currently, there are two predominant techniques that utilize peritoneal flaps: the modified Davydov procedure and the tubularized urachus-peritoneal hinge flap.
The modified Davydov technique, which originated in the treatment of congenital vaginal agenesis in cisgender women, involves the creation of anterior and posterior peritoneal flaps. This type of peritoneal vaginoplasty is more commonly utilized for primary cases.
Ideally, there is a robotic surgeon (typically a urologist) working in tandem with the perineal surgeon. The robotic surgeon makes a horizontal incision along the peritoneal ridge at the rectovesical junction and continues the dissection within Denonvilliers fascia, between the prostate and rectum, to the pelvic floor. This dissection is like that performed in a robot-assisted laparoscopic prostatectomy.
Simultaneously, the perineal surgeon will break through the pelvic floor with assistance of the robotic view. Peritoneal flaps are raised from the anterior rectum and posterior bladder.2,3 In primary cases, the penoscrotal flap is introduced into the abdomen from the perineum and sutured to the anterior and posterior peritoneum to create a circumferential canal. At the apex of the neovagina, these anterior and posterior flaps are then sutured together.2,3
The tubularized urachus-peritoneal hinge flap technique is predominantly used for revision cases in patients who experienced neovaginal shortening and desire increased neovaginal depth. As peritoneal reach is limited, candidates for this procedure must have both adequate width and neovaginal canal depth.4 Once intra-abdominal access is achieved, an anterior peritoneal flap is mobilized to the level of the bladder and rotated 180 degrees inferiorly.4 The superior aspect of the flap is flipped is mobilized and is sutured to the peritoneum at the apex of the neovaginal canal.
The main benefit of these procedures, compared with traditional techniques, is increased neovaginal depth. The average vaginal length in patients undergoing peritoneal vaginoplasties is 14.2 cm, compared with 11.6 cm achieved in those using skin grafts.1,3 However, many surgeons report achieving 14-15 cm of depth with the traditional vaginoplasty. There are insufficient short- and long-term data for the peritoneal technique to recommend this as a first-line procedure.
Complications for peritoneal vaginoplasty procedures are similar to those of single-stage penile inversion vaginoplasty cases but with additional operative risks associated with laparoscopic/robotic surgery. These risks include injury to viscera and major vessels during initial intra-abdominal access, intra-abdominal adhesions, port site hernias, need to convert to an open procedure, and equipment malfunction.2 Additional postoperative risks include pelvic abscess formation, dehiscence of the peritoneal-vaginal incision, and peritoneal perforation during dilation.2,3 Surgeons and institutions must also weigh the cost of using the robot versus the cost of additional revisional surgical procedures. While initial studies evaluating robotic peritoneal vaginoplasty procedures have yielded promising preliminary results, additional studies are warranted.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Salibian AA et al. Plast Reconstr Surg. 2021;147(4):634e-43e.
2. Dy GW et al. In: Nikolavsky D and Blakely SA, eds. Urological care for the transgender patient: A comprehensive guide. Switzerland: Springer, 2021:237-48.
3. Jacoby A et al. J Urol. 2019;201(6):1171-5.
4. Smith SM et al. J Sex Med. 2022;10(6):100572.
When the Food and Drug Administration first approved the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, Calif.) for adult use in 2000, it altered the face of minimally invasive surgery across a multitude of specialties. Improved three-dimensional visualization and enhanced instrument articulation facilitates complex dissections and intracorporeal suturing. While the standard of care for gender-affirming vaginoplasty remains the single-stage penile inversion vaginoplasty, robotic procedures are quickly emerging as alternative options for both primary and revisional surgeries.
The single-stage penile inversion vaginoplasty requires an adequate amount of penoscrotal tissue not only to line a neovaginal canal that measures 12-15 cm, but also to create external vulvar structures. While this is often sufficient in most candidates, there is an increasing number of patients who are receiving puberty blockers, resulting in penoscrotal hypoplasia.
Alternatively, there are patients who experience loss of vaginal depth and vaginal stenosis who seek revisional surgeries. Additional donor sites for skin grafting are available and include the lower abdomen and thighs, although patients may not want these donor site scars. With these donor sites, there is also concern about graft contracture, which could lead to recurrent vaginal stenosis.1 Robotic peritoneal vaginoplasty and robotic enteric vaginoplasty can serve as additional options for patients seeking revisional surgery or who have insufficient genital skin. One benefit of using peritoneal flaps is that they are hairless and are well vascularized with minimal donor site morbidity.1 Currently, there are two predominant techniques that utilize peritoneal flaps: the modified Davydov procedure and the tubularized urachus-peritoneal hinge flap.
The modified Davydov technique, which originated in the treatment of congenital vaginal agenesis in cisgender women, involves the creation of anterior and posterior peritoneal flaps. This type of peritoneal vaginoplasty is more commonly utilized for primary cases.
Ideally, there is a robotic surgeon (typically a urologist) working in tandem with the perineal surgeon. The robotic surgeon makes a horizontal incision along the peritoneal ridge at the rectovesical junction and continues the dissection within Denonvilliers fascia, between the prostate and rectum, to the pelvic floor. This dissection is like that performed in a robot-assisted laparoscopic prostatectomy.
Simultaneously, the perineal surgeon will break through the pelvic floor with assistance of the robotic view. Peritoneal flaps are raised from the anterior rectum and posterior bladder.2,3 In primary cases, the penoscrotal flap is introduced into the abdomen from the perineum and sutured to the anterior and posterior peritoneum to create a circumferential canal. At the apex of the neovagina, these anterior and posterior flaps are then sutured together.2,3
The tubularized urachus-peritoneal hinge flap technique is predominantly used for revision cases in patients who experienced neovaginal shortening and desire increased neovaginal depth. As peritoneal reach is limited, candidates for this procedure must have both adequate width and neovaginal canal depth.4 Once intra-abdominal access is achieved, an anterior peritoneal flap is mobilized to the level of the bladder and rotated 180 degrees inferiorly.4 The superior aspect of the flap is flipped is mobilized and is sutured to the peritoneum at the apex of the neovaginal canal.
The main benefit of these procedures, compared with traditional techniques, is increased neovaginal depth. The average vaginal length in patients undergoing peritoneal vaginoplasties is 14.2 cm, compared with 11.6 cm achieved in those using skin grafts.1,3 However, many surgeons report achieving 14-15 cm of depth with the traditional vaginoplasty. There are insufficient short- and long-term data for the peritoneal technique to recommend this as a first-line procedure.
Complications for peritoneal vaginoplasty procedures are similar to those of single-stage penile inversion vaginoplasty cases but with additional operative risks associated with laparoscopic/robotic surgery. These risks include injury to viscera and major vessels during initial intra-abdominal access, intra-abdominal adhesions, port site hernias, need to convert to an open procedure, and equipment malfunction.2 Additional postoperative risks include pelvic abscess formation, dehiscence of the peritoneal-vaginal incision, and peritoneal perforation during dilation.2,3 Surgeons and institutions must also weigh the cost of using the robot versus the cost of additional revisional surgical procedures. While initial studies evaluating robotic peritoneal vaginoplasty procedures have yielded promising preliminary results, additional studies are warranted.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Salibian AA et al. Plast Reconstr Surg. 2021;147(4):634e-43e.
2. Dy GW et al. In: Nikolavsky D and Blakely SA, eds. Urological care for the transgender patient: A comprehensive guide. Switzerland: Springer, 2021:237-48.
3. Jacoby A et al. J Urol. 2019;201(6):1171-5.
4. Smith SM et al. J Sex Med. 2022;10(6):100572.
When the Food and Drug Administration first approved the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, Calif.) for adult use in 2000, it altered the face of minimally invasive surgery across a multitude of specialties. Improved three-dimensional visualization and enhanced instrument articulation facilitates complex dissections and intracorporeal suturing. While the standard of care for gender-affirming vaginoplasty remains the single-stage penile inversion vaginoplasty, robotic procedures are quickly emerging as alternative options for both primary and revisional surgeries.
The single-stage penile inversion vaginoplasty requires an adequate amount of penoscrotal tissue not only to line a neovaginal canal that measures 12-15 cm, but also to create external vulvar structures. While this is often sufficient in most candidates, there is an increasing number of patients who are receiving puberty blockers, resulting in penoscrotal hypoplasia.
Alternatively, there are patients who experience loss of vaginal depth and vaginal stenosis who seek revisional surgeries. Additional donor sites for skin grafting are available and include the lower abdomen and thighs, although patients may not want these donor site scars. With these donor sites, there is also concern about graft contracture, which could lead to recurrent vaginal stenosis.1 Robotic peritoneal vaginoplasty and robotic enteric vaginoplasty can serve as additional options for patients seeking revisional surgery or who have insufficient genital skin. One benefit of using peritoneal flaps is that they are hairless and are well vascularized with minimal donor site morbidity.1 Currently, there are two predominant techniques that utilize peritoneal flaps: the modified Davydov procedure and the tubularized urachus-peritoneal hinge flap.
The modified Davydov technique, which originated in the treatment of congenital vaginal agenesis in cisgender women, involves the creation of anterior and posterior peritoneal flaps. This type of peritoneal vaginoplasty is more commonly utilized for primary cases.
Ideally, there is a robotic surgeon (typically a urologist) working in tandem with the perineal surgeon. The robotic surgeon makes a horizontal incision along the peritoneal ridge at the rectovesical junction and continues the dissection within Denonvilliers fascia, between the prostate and rectum, to the pelvic floor. This dissection is like that performed in a robot-assisted laparoscopic prostatectomy.
Simultaneously, the perineal surgeon will break through the pelvic floor with assistance of the robotic view. Peritoneal flaps are raised from the anterior rectum and posterior bladder.2,3 In primary cases, the penoscrotal flap is introduced into the abdomen from the perineum and sutured to the anterior and posterior peritoneum to create a circumferential canal. At the apex of the neovagina, these anterior and posterior flaps are then sutured together.2,3
The tubularized urachus-peritoneal hinge flap technique is predominantly used for revision cases in patients who experienced neovaginal shortening and desire increased neovaginal depth. As peritoneal reach is limited, candidates for this procedure must have both adequate width and neovaginal canal depth.4 Once intra-abdominal access is achieved, an anterior peritoneal flap is mobilized to the level of the bladder and rotated 180 degrees inferiorly.4 The superior aspect of the flap is flipped is mobilized and is sutured to the peritoneum at the apex of the neovaginal canal.
The main benefit of these procedures, compared with traditional techniques, is increased neovaginal depth. The average vaginal length in patients undergoing peritoneal vaginoplasties is 14.2 cm, compared with 11.6 cm achieved in those using skin grafts.1,3 However, many surgeons report achieving 14-15 cm of depth with the traditional vaginoplasty. There are insufficient short- and long-term data for the peritoneal technique to recommend this as a first-line procedure.
Complications for peritoneal vaginoplasty procedures are similar to those of single-stage penile inversion vaginoplasty cases but with additional operative risks associated with laparoscopic/robotic surgery. These risks include injury to viscera and major vessels during initial intra-abdominal access, intra-abdominal adhesions, port site hernias, need to convert to an open procedure, and equipment malfunction.2 Additional postoperative risks include pelvic abscess formation, dehiscence of the peritoneal-vaginal incision, and peritoneal perforation during dilation.2,3 Surgeons and institutions must also weigh the cost of using the robot versus the cost of additional revisional surgical procedures. While initial studies evaluating robotic peritoneal vaginoplasty procedures have yielded promising preliminary results, additional studies are warranted.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Salibian AA et al. Plast Reconstr Surg. 2021;147(4):634e-43e.
2. Dy GW et al. In: Nikolavsky D and Blakely SA, eds. Urological care for the transgender patient: A comprehensive guide. Switzerland: Springer, 2021:237-48.
3. Jacoby A et al. J Urol. 2019;201(6):1171-5.
4. Smith SM et al. J Sex Med. 2022;10(6):100572.
Prepare for endometriosis excision surgery with a multidisciplinary approach
Introduction: The preoperative evaluation for endometriosis – more than meets the eye
It is well known that it often takes 6-10 years for endometriosis to be diagnosed in patients who have the disease, depending on where the patient lives. I certainly am not surprised. During my residency at Parkland Memorial Hospital, if a patient had chronic pelvic pain and no fibroids, her diagnosis was usually pelvic inflammatory disease. Later, during my fellowship in reproductive endocrinology at the University of Pennsylvania, the diagnosis became endometriosis.
As I gained more interest and expertise in the treatment of endometriosis, I became aware of several articles concluding that if a woman sought treatment for chronic pelvic pain with an internist, the diagnosis would be irritable bowel syndrome (IBS); with a urologist, it would be interstitial cystitis; and with a gynecologist, endometriosis. Moreover, there is an increased propensity for IBS and IC in patients with endometriosis. There also is an increased risk of small intestine bacterial overgrowth (SIBO), as noted by our guest author for this latest installment of the Master Class in Gynecologic Surgery, Iris Orbuch, MD.
Like our guest author, I have also noted increased risk of pelvic floor myalgia. Dr. Orbuch clearly outlines why this occurs. In fact, we can now understand why many patients have multiple pelvic pain–inducing issues compounding their pain secondary to endometriosis and leading to remodeling of the central nervous system. Therefore, it certainly makes sense to follow Dr. Orbuch’s recommendation for a multidisciplinary pre- and postsurgical approach “to downregulate the pain generators.”
Dr. Orbuch is a minimally invasive gynecologic surgeon in Los Angeles who specializes in the treatment of patients diagnosed with endometriosis. Dr. Orbuch serves on the Board of Directors of the Foundation of the American Association of Gynecologic Laparoscopists and has served as the chair of the AAGL’s Special Interest Group on Endometriosis and Reproductive Surgery. She is the coauthor of the book “Beating Endo – How to Reclaim Your Life From Endometriosis” (New York: HarperCollins; 2019). The book is written for patients but addresses many issues discussed in this installment of the Master Class in Gynecologic Surgery.
Dr. Miller, MD, FACOG, is professor of obstetrics and gynecology, department of clinical sciences, Rosalind Franklin University of Medicine and Science, North Chicago. He has no conflicts of interest to report.
Patients with endometriosis and the all-too-often decade-long diagnostic delay have a variety of coexisting conditions that are pain generators – from painful bladder syndrome and pelvic floor dysfunction to a small intestine bacterial system that is significantly upregulated and sensitized.
For optimal surgical outcomes, and to help our patients recover from years of this inflammatory, systemic disease, we must treat our patients holistically and work to downregulate their pain as much as possible before excision surgery. I work with patients a few months prior to surgery, often for 4-5 months, during which time they not only see me for informative follow-ups, but also pelvic floor physical therapists, gastroenterologists, mental health professionals, integrative nutritionists, and physiatrists or pain specialists, depending on their needs.1
By identifying coexisting conditions in an initial consult and employing a presurgical multidisciplinary approach to downregulate the pain generators, my patients recover well from excision surgery, with greater and faster relief from pain, compared with those using standard approaches, and with little to no use of opioids.
At a minimum, given the unfortunate time constraints and productivity demands of working within health systems – and considering that surgeries are often scheduled a couple of months out – the surgeon could ensure that patients are engaged in at least 6-8 weeks of pelvic floor physical therapy before surgery to sufficiently lengthen the pelvic muscles and loosen surrounding fascia.
Short, tight pelvic floor muscles are almost universal in patients with delayed diagnosis of endometriosis and are significant generators of pain.
Appreciating sequelae of diagnostic delay
After my fellowship in advanced laparoscopic and pelvic surgery with Harry Reich, MD, and C. Y. Liu, MD, pioneers of endometriosis excision surgery, and as I did my residency in the early 2000s, I noticed puzzlement in the literature about why some patients still had lasting pain after thorough excision.
I didn’t doubt the efficacy of excision. It is the cornerstone of treatment, and at least one randomized double-blind trial2 and a systematic review and meta-analysis3 have demonstrated its superior efficacy over ablation in symptom reduction. What I did doubt was any presumption that surgery alone was enough. I knew there was more to healing when a disease process wreaks havoc on the body for more than a decade and that there were other generators of pain in addition to the endometriosis implants themselves.
As I began to focus on endometriosis in my own surgical practice, I strove to detect and treat endometriosis in teens. But in those patients with longstanding disease, I recognized patterns and began to more fully appreciate the systemic sequelae of endometriosis.
To cope with dysmenorrhea, patients curl up and assume a fetal position, tensing the abdominal muscles, inner thigh muscles, and pelvic floor muscles. Over time, these muscles come to maintain a short, tight, and painful state. (Hence the need for physical therapy to undo this decade-long pattern.)
Endometriosis implants on or near the gastrointestinal tract tug on fascia and muscles and commonly cause constipation, leading women to further overwork the pelvic floor muscles. In the case of diarrhea-predominant dysfunction, our patients squeeze pelvic floor muscles to prevent leakage. And in the case of urinary urgency, they squeeze muscles to release urine that isn’t really there.
As the chronic inflammation of the disease grows, and as pain worsens, the patient is increasingly in sympathetic overdrive (also known as ”fight or flight”), as opposed to a parasympathetic state (also known as “rest and digest”). The bowel’s motility slows, allowing the bacteria of the small intestine to grow beyond what is normal, leading to SIBO, a condition increasingly recognized by gastroenterologists and others that can impede nutrient absorption and cause bloat and pain and exacerbate constipation and diarrhea.
Key to my conceptualization of pain was a review published in 2011 by Pam Stratton, MD, of the National Institutes of Health, and Karen J. Berkley, PhD, then of Florida State University, on chronic pain and endometriosis.4 They detailed how endometriotic lesions can develop their own nerve supply that interacts directly and in a two-way fashion with the CNS – and how the lesions can engage the nervous system in ways that create comorbid conditions and pain that becomes “independent of the disease itself.”
Sensitized peripheral nerve fibers innervating a deeply infiltrating lesion on the left uterosacral ligament, for instance, can sensitize neurons in the spinal sacral segment. Branches of these nerve fibers can extend to other segments of the spinal cord, and, once sensitized themselves, turn on neurons in these other segments. There is a resultant remodeling of the central nervous system, in essence, and what is called “remote central sensitization.” The CNS becomes independent from peripheral neural processes.
I now explain to both patients and physicians that those who have had endometriosis for years have had an enduring “hand on the stove,” with a persistent signal to the CNS. Tight muscles are a hand on the stove, painful bladder syndrome is another hand on the stove, and SIBO is yet another. So are anxiety and depression.
The CNS becomes so upregulated and overloaded that messages branch out through the spinal cord to other available pathways and to other organs, muscles, and nerves. The CNS also starts firing on its own – and once it becomes its own pain generator, taking one hand off the stove (for instance, excising implants) while leaving multiple other hands on the hot stove won’t remove all pain. We must downregulate the CNS more broadly.
As I began addressing pain generators and instigators of CNS sensitization – and waiting for excision surgery until the CNS had sufficiently cooled – I saw that my patients had a better chance of more significant and lasting pain relief.
Pearls for a multimodal approach
My initial physical exam includes an assessment of the pelvic floor for overly tight musculature. An abdominal exam will usually reveal whether there is asymmetry of the abdominal wall muscles, which typically informs me of the likelihood of tightness and pulling on either side of the pelvic anatomy. On the internal exam, then, the pelvic floor muscles can be palpated and assessed. These findings will guide my referrals and my discussions with patients about the value of pelvic floor physical therapy. The cervix should be in the midline of the vagina – equidistant from the left and right vaginal fornices. If the cervix is pulled away from this midline, and a palpation of a thickened uterosacral ligament reproduces pain, endometriosis is 90% likely.
Patients who report significant “burning” pain that’s suggestive of neuropathic pain should be referred to a physical medicine rehabilitation physician or a pain specialist who can help downregulate their CNS. And patients who have symptoms of depression, anxiety disorders (including obsessive-compulsive disorder), or posttraumatic stress disorder should be referred to pain therapists, psychologists, or other mental health professionals, preferably well before surgery. I will also often discuss mindfulness practices and give my patients “meditation challenges” to achieve during the presurgical phase.
Additional points of emphasis about a multidisciplinary, multimodal approach include:
Advanced pelvic floor therapy: Therapists with specialized training in pelvic health and manual therapy utilize a range of techniques and modalities to release tension in affected muscles, fascia, nerves, and bone, and in doing so, they help to downregulate the CNS. Myofascial release, myofascial trigger point release, neural mobilization, and visceral mobilization are among these techniques. In addition to using manual therapy, many of these therapists may also employ neuromuscular reeducation and other techniques that will be helpful for the longer term.
It is important to identify physical therapists who have training in this approach; women with endometriosis often have a history of treatment by physical therapists whose focus is on incontinence and muscle strengthening (that is, Kegel exercises), which is the opposite of what endometriosis patients need.
Treating SIBO: Symptoms commonly associated with SIBO often overlap with symptoms of irritable bowel syndrome (IBS) – namely constipation, diarrhea (or both), and bloating. Indeed, many patients with undiagnosed endometriosis have been diagnosed with IBS. I send every patient who has one of these symptoms for SIBO breath testing, which utilizes carbohydrate substrates (glucose or lactulose) and measures hydrogen and/or methane in the breath.
SIBO is typically treated with rifampin, which stays in the small bowel and will not negatively affect beneficial bacteria, with or without neomycin. Gastroenterologists with more integrative practices also consider the use of herbals in addition to – or instead of – antibiotics. It can sometimes take months or a couple of years to correct SIBO, depending on how long the patient has been affected, but with presurgical diagnosis and a start on treatment, we can remove or at least tone down another instigator of CNS sensitization.
I estimate that 80% of my patients have tested positive for SIBO. Notably, in a testament to the systemic nature of endometriosis, a study published in 2009 of 355 women undergoing operative laparoscopy for suspected endometriosis found that 90% had gastrointestinal symptoms, but only 7.6% of the vast majority whose endometriosis was confirmed were found to have endometrial implants on the bowel itself.5
Addressing bladder issues: I routinely administer the PUF (Pain, Urgency, Frequency) questionnaire as part of my intake package and follow it up with conversation. For just about every patient with painful bladder syndrome, pelvic floor physical therapy in combination with a low-acid, low-potassium diet will work effectively together to reduce symptoms and pain. The IC Network offers a helpful food list, and patients can be counseled to choose foods that are also anti-inflammatory. When referrals to a urologist for bladder instillations are possible, these can be helpful as well.
Our communication with patients
Our patients need to have their symptoms and pain validated and to understand why we’re recommending these measures before surgery. Some education is necessary. Few patients will go to an integrative nutritionist, for example, if we just write a referral without explaining how years of inflammation and disruption in the gut can affect the whole body – including mental health – and that it can be corrected over time.
Also necessary is an appreciation of the fact that patients with delayed diagnoses have lived with gastrointestinal and other symptoms and patterns for so long – and often have mothers whose endometriosis caused similar symptoms – that some of their own experiences can seem almost “normal.” A patient whose mother had bowel movements every 7 days may think that 4-5 day intervals are acceptable, for instance. This means we have to carefully consider how we ask our questions.
I always ask my patients as we’re going into surgery, what percentage better are you? I’ve long aimed for at least 30% improvement, but most of the time, with pelvic floor therapy and as many other pain-generator–focused measures as possible, we’re getting them 70% better.
Excision surgery will remove the inflammation that has helped fuel the SIBO and other coconditions. Then, everything done to prepare the body must continue for some time. Certain practices, such as eating an anti-inflammatory diet, should be lifelong.
One day, it is hoped, a pediatrician or other physician will suspect endometriosis early on. The patient will see the surgeon within several months of the onset of pain, and we won’t need to unravel layers of pain generation and CNS upregulation before operating. But until this happens and we shorten the diagnostic delay, we must consider the benefits of presurgical preparation.
References
1. Orbuch I, Stein A. Beating Endo: How to Reclaim Your Life From Endometriosis. (New York: HarperCollins, 2019).
2. Healey M et al. J Minim Invasive Gynecol. 2014;21(6):999-1004.
3. Pundir J et al. J Minim Invasive Gynecol. 2017;24(5):747-56.
4. Stratton P, Berkley KJ. Hum Repro Update. 2011;17(3):327-46.
5. Maroun P et al. Aust N Z J Obstet Gynaecol. 2009;49(4):411-4.
Dr. Orbuch is a minimally invasive gynecologic surgeon in Los Angeles who specializes in endometriosis. She has no conflicts of interest to report.
Introduction: The preoperative evaluation for endometriosis – more than meets the eye
It is well known that it often takes 6-10 years for endometriosis to be diagnosed in patients who have the disease, depending on where the patient lives. I certainly am not surprised. During my residency at Parkland Memorial Hospital, if a patient had chronic pelvic pain and no fibroids, her diagnosis was usually pelvic inflammatory disease. Later, during my fellowship in reproductive endocrinology at the University of Pennsylvania, the diagnosis became endometriosis.
As I gained more interest and expertise in the treatment of endometriosis, I became aware of several articles concluding that if a woman sought treatment for chronic pelvic pain with an internist, the diagnosis would be irritable bowel syndrome (IBS); with a urologist, it would be interstitial cystitis; and with a gynecologist, endometriosis. Moreover, there is an increased propensity for IBS and IC in patients with endometriosis. There also is an increased risk of small intestine bacterial overgrowth (SIBO), as noted by our guest author for this latest installment of the Master Class in Gynecologic Surgery, Iris Orbuch, MD.
Like our guest author, I have also noted increased risk of pelvic floor myalgia. Dr. Orbuch clearly outlines why this occurs. In fact, we can now understand why many patients have multiple pelvic pain–inducing issues compounding their pain secondary to endometriosis and leading to remodeling of the central nervous system. Therefore, it certainly makes sense to follow Dr. Orbuch’s recommendation for a multidisciplinary pre- and postsurgical approach “to downregulate the pain generators.”
Dr. Orbuch is a minimally invasive gynecologic surgeon in Los Angeles who specializes in the treatment of patients diagnosed with endometriosis. Dr. Orbuch serves on the Board of Directors of the Foundation of the American Association of Gynecologic Laparoscopists and has served as the chair of the AAGL’s Special Interest Group on Endometriosis and Reproductive Surgery. She is the coauthor of the book “Beating Endo – How to Reclaim Your Life From Endometriosis” (New York: HarperCollins; 2019). The book is written for patients but addresses many issues discussed in this installment of the Master Class in Gynecologic Surgery.
Dr. Miller, MD, FACOG, is professor of obstetrics and gynecology, department of clinical sciences, Rosalind Franklin University of Medicine and Science, North Chicago. He has no conflicts of interest to report.
Patients with endometriosis and the all-too-often decade-long diagnostic delay have a variety of coexisting conditions that are pain generators – from painful bladder syndrome and pelvic floor dysfunction to a small intestine bacterial system that is significantly upregulated and sensitized.
For optimal surgical outcomes, and to help our patients recover from years of this inflammatory, systemic disease, we must treat our patients holistically and work to downregulate their pain as much as possible before excision surgery. I work with patients a few months prior to surgery, often for 4-5 months, during which time they not only see me for informative follow-ups, but also pelvic floor physical therapists, gastroenterologists, mental health professionals, integrative nutritionists, and physiatrists or pain specialists, depending on their needs.1
By identifying coexisting conditions in an initial consult and employing a presurgical multidisciplinary approach to downregulate the pain generators, my patients recover well from excision surgery, with greater and faster relief from pain, compared with those using standard approaches, and with little to no use of opioids.
At a minimum, given the unfortunate time constraints and productivity demands of working within health systems – and considering that surgeries are often scheduled a couple of months out – the surgeon could ensure that patients are engaged in at least 6-8 weeks of pelvic floor physical therapy before surgery to sufficiently lengthen the pelvic muscles and loosen surrounding fascia.
Short, tight pelvic floor muscles are almost universal in patients with delayed diagnosis of endometriosis and are significant generators of pain.
Appreciating sequelae of diagnostic delay
After my fellowship in advanced laparoscopic and pelvic surgery with Harry Reich, MD, and C. Y. Liu, MD, pioneers of endometriosis excision surgery, and as I did my residency in the early 2000s, I noticed puzzlement in the literature about why some patients still had lasting pain after thorough excision.
I didn’t doubt the efficacy of excision. It is the cornerstone of treatment, and at least one randomized double-blind trial2 and a systematic review and meta-analysis3 have demonstrated its superior efficacy over ablation in symptom reduction. What I did doubt was any presumption that surgery alone was enough. I knew there was more to healing when a disease process wreaks havoc on the body for more than a decade and that there were other generators of pain in addition to the endometriosis implants themselves.
As I began to focus on endometriosis in my own surgical practice, I strove to detect and treat endometriosis in teens. But in those patients with longstanding disease, I recognized patterns and began to more fully appreciate the systemic sequelae of endometriosis.
To cope with dysmenorrhea, patients curl up and assume a fetal position, tensing the abdominal muscles, inner thigh muscles, and pelvic floor muscles. Over time, these muscles come to maintain a short, tight, and painful state. (Hence the need for physical therapy to undo this decade-long pattern.)
Endometriosis implants on or near the gastrointestinal tract tug on fascia and muscles and commonly cause constipation, leading women to further overwork the pelvic floor muscles. In the case of diarrhea-predominant dysfunction, our patients squeeze pelvic floor muscles to prevent leakage. And in the case of urinary urgency, they squeeze muscles to release urine that isn’t really there.
As the chronic inflammation of the disease grows, and as pain worsens, the patient is increasingly in sympathetic overdrive (also known as ”fight or flight”), as opposed to a parasympathetic state (also known as “rest and digest”). The bowel’s motility slows, allowing the bacteria of the small intestine to grow beyond what is normal, leading to SIBO, a condition increasingly recognized by gastroenterologists and others that can impede nutrient absorption and cause bloat and pain and exacerbate constipation and diarrhea.
Key to my conceptualization of pain was a review published in 2011 by Pam Stratton, MD, of the National Institutes of Health, and Karen J. Berkley, PhD, then of Florida State University, on chronic pain and endometriosis.4 They detailed how endometriotic lesions can develop their own nerve supply that interacts directly and in a two-way fashion with the CNS – and how the lesions can engage the nervous system in ways that create comorbid conditions and pain that becomes “independent of the disease itself.”
Sensitized peripheral nerve fibers innervating a deeply infiltrating lesion on the left uterosacral ligament, for instance, can sensitize neurons in the spinal sacral segment. Branches of these nerve fibers can extend to other segments of the spinal cord, and, once sensitized themselves, turn on neurons in these other segments. There is a resultant remodeling of the central nervous system, in essence, and what is called “remote central sensitization.” The CNS becomes independent from peripheral neural processes.
I now explain to both patients and physicians that those who have had endometriosis for years have had an enduring “hand on the stove,” with a persistent signal to the CNS. Tight muscles are a hand on the stove, painful bladder syndrome is another hand on the stove, and SIBO is yet another. So are anxiety and depression.
The CNS becomes so upregulated and overloaded that messages branch out through the spinal cord to other available pathways and to other organs, muscles, and nerves. The CNS also starts firing on its own – and once it becomes its own pain generator, taking one hand off the stove (for instance, excising implants) while leaving multiple other hands on the hot stove won’t remove all pain. We must downregulate the CNS more broadly.
As I began addressing pain generators and instigators of CNS sensitization – and waiting for excision surgery until the CNS had sufficiently cooled – I saw that my patients had a better chance of more significant and lasting pain relief.
Pearls for a multimodal approach
My initial physical exam includes an assessment of the pelvic floor for overly tight musculature. An abdominal exam will usually reveal whether there is asymmetry of the abdominal wall muscles, which typically informs me of the likelihood of tightness and pulling on either side of the pelvic anatomy. On the internal exam, then, the pelvic floor muscles can be palpated and assessed. These findings will guide my referrals and my discussions with patients about the value of pelvic floor physical therapy. The cervix should be in the midline of the vagina – equidistant from the left and right vaginal fornices. If the cervix is pulled away from this midline, and a palpation of a thickened uterosacral ligament reproduces pain, endometriosis is 90% likely.
Patients who report significant “burning” pain that’s suggestive of neuropathic pain should be referred to a physical medicine rehabilitation physician or a pain specialist who can help downregulate their CNS. And patients who have symptoms of depression, anxiety disorders (including obsessive-compulsive disorder), or posttraumatic stress disorder should be referred to pain therapists, psychologists, or other mental health professionals, preferably well before surgery. I will also often discuss mindfulness practices and give my patients “meditation challenges” to achieve during the presurgical phase.
Additional points of emphasis about a multidisciplinary, multimodal approach include:
Advanced pelvic floor therapy: Therapists with specialized training in pelvic health and manual therapy utilize a range of techniques and modalities to release tension in affected muscles, fascia, nerves, and bone, and in doing so, they help to downregulate the CNS. Myofascial release, myofascial trigger point release, neural mobilization, and visceral mobilization are among these techniques. In addition to using manual therapy, many of these therapists may also employ neuromuscular reeducation and other techniques that will be helpful for the longer term.
It is important to identify physical therapists who have training in this approach; women with endometriosis often have a history of treatment by physical therapists whose focus is on incontinence and muscle strengthening (that is, Kegel exercises), which is the opposite of what endometriosis patients need.
Treating SIBO: Symptoms commonly associated with SIBO often overlap with symptoms of irritable bowel syndrome (IBS) – namely constipation, diarrhea (or both), and bloating. Indeed, many patients with undiagnosed endometriosis have been diagnosed with IBS. I send every patient who has one of these symptoms for SIBO breath testing, which utilizes carbohydrate substrates (glucose or lactulose) and measures hydrogen and/or methane in the breath.
SIBO is typically treated with rifampin, which stays in the small bowel and will not negatively affect beneficial bacteria, with or without neomycin. Gastroenterologists with more integrative practices also consider the use of herbals in addition to – or instead of – antibiotics. It can sometimes take months or a couple of years to correct SIBO, depending on how long the patient has been affected, but with presurgical diagnosis and a start on treatment, we can remove or at least tone down another instigator of CNS sensitization.
I estimate that 80% of my patients have tested positive for SIBO. Notably, in a testament to the systemic nature of endometriosis, a study published in 2009 of 355 women undergoing operative laparoscopy for suspected endometriosis found that 90% had gastrointestinal symptoms, but only 7.6% of the vast majority whose endometriosis was confirmed were found to have endometrial implants on the bowel itself.5
Addressing bladder issues: I routinely administer the PUF (Pain, Urgency, Frequency) questionnaire as part of my intake package and follow it up with conversation. For just about every patient with painful bladder syndrome, pelvic floor physical therapy in combination with a low-acid, low-potassium diet will work effectively together to reduce symptoms and pain. The IC Network offers a helpful food list, and patients can be counseled to choose foods that are also anti-inflammatory. When referrals to a urologist for bladder instillations are possible, these can be helpful as well.
Our communication with patients
Our patients need to have their symptoms and pain validated and to understand why we’re recommending these measures before surgery. Some education is necessary. Few patients will go to an integrative nutritionist, for example, if we just write a referral without explaining how years of inflammation and disruption in the gut can affect the whole body – including mental health – and that it can be corrected over time.
Also necessary is an appreciation of the fact that patients with delayed diagnoses have lived with gastrointestinal and other symptoms and patterns for so long – and often have mothers whose endometriosis caused similar symptoms – that some of their own experiences can seem almost “normal.” A patient whose mother had bowel movements every 7 days may think that 4-5 day intervals are acceptable, for instance. This means we have to carefully consider how we ask our questions.
I always ask my patients as we’re going into surgery, what percentage better are you? I’ve long aimed for at least 30% improvement, but most of the time, with pelvic floor therapy and as many other pain-generator–focused measures as possible, we’re getting them 70% better.
Excision surgery will remove the inflammation that has helped fuel the SIBO and other coconditions. Then, everything done to prepare the body must continue for some time. Certain practices, such as eating an anti-inflammatory diet, should be lifelong.
One day, it is hoped, a pediatrician or other physician will suspect endometriosis early on. The patient will see the surgeon within several months of the onset of pain, and we won’t need to unravel layers of pain generation and CNS upregulation before operating. But until this happens and we shorten the diagnostic delay, we must consider the benefits of presurgical preparation.
References
1. Orbuch I, Stein A. Beating Endo: How to Reclaim Your Life From Endometriosis. (New York: HarperCollins, 2019).
2. Healey M et al. J Minim Invasive Gynecol. 2014;21(6):999-1004.
3. Pundir J et al. J Minim Invasive Gynecol. 2017;24(5):747-56.
4. Stratton P, Berkley KJ. Hum Repro Update. 2011;17(3):327-46.
5. Maroun P et al. Aust N Z J Obstet Gynaecol. 2009;49(4):411-4.
Dr. Orbuch is a minimally invasive gynecologic surgeon in Los Angeles who specializes in endometriosis. She has no conflicts of interest to report.
Introduction: The preoperative evaluation for endometriosis – more than meets the eye
It is well known that it often takes 6-10 years for endometriosis to be diagnosed in patients who have the disease, depending on where the patient lives. I certainly am not surprised. During my residency at Parkland Memorial Hospital, if a patient had chronic pelvic pain and no fibroids, her diagnosis was usually pelvic inflammatory disease. Later, during my fellowship in reproductive endocrinology at the University of Pennsylvania, the diagnosis became endometriosis.
As I gained more interest and expertise in the treatment of endometriosis, I became aware of several articles concluding that if a woman sought treatment for chronic pelvic pain with an internist, the diagnosis would be irritable bowel syndrome (IBS); with a urologist, it would be interstitial cystitis; and with a gynecologist, endometriosis. Moreover, there is an increased propensity for IBS and IC in patients with endometriosis. There also is an increased risk of small intestine bacterial overgrowth (SIBO), as noted by our guest author for this latest installment of the Master Class in Gynecologic Surgery, Iris Orbuch, MD.
Like our guest author, I have also noted increased risk of pelvic floor myalgia. Dr. Orbuch clearly outlines why this occurs. In fact, we can now understand why many patients have multiple pelvic pain–inducing issues compounding their pain secondary to endometriosis and leading to remodeling of the central nervous system. Therefore, it certainly makes sense to follow Dr. Orbuch’s recommendation for a multidisciplinary pre- and postsurgical approach “to downregulate the pain generators.”
Dr. Orbuch is a minimally invasive gynecologic surgeon in Los Angeles who specializes in the treatment of patients diagnosed with endometriosis. Dr. Orbuch serves on the Board of Directors of the Foundation of the American Association of Gynecologic Laparoscopists and has served as the chair of the AAGL’s Special Interest Group on Endometriosis and Reproductive Surgery. She is the coauthor of the book “Beating Endo – How to Reclaim Your Life From Endometriosis” (New York: HarperCollins; 2019). The book is written for patients but addresses many issues discussed in this installment of the Master Class in Gynecologic Surgery.
Dr. Miller, MD, FACOG, is professor of obstetrics and gynecology, department of clinical sciences, Rosalind Franklin University of Medicine and Science, North Chicago. He has no conflicts of interest to report.
Patients with endometriosis and the all-too-often decade-long diagnostic delay have a variety of coexisting conditions that are pain generators – from painful bladder syndrome and pelvic floor dysfunction to a small intestine bacterial system that is significantly upregulated and sensitized.
For optimal surgical outcomes, and to help our patients recover from years of this inflammatory, systemic disease, we must treat our patients holistically and work to downregulate their pain as much as possible before excision surgery. I work with patients a few months prior to surgery, often for 4-5 months, during which time they not only see me for informative follow-ups, but also pelvic floor physical therapists, gastroenterologists, mental health professionals, integrative nutritionists, and physiatrists or pain specialists, depending on their needs.1
By identifying coexisting conditions in an initial consult and employing a presurgical multidisciplinary approach to downregulate the pain generators, my patients recover well from excision surgery, with greater and faster relief from pain, compared with those using standard approaches, and with little to no use of opioids.
At a minimum, given the unfortunate time constraints and productivity demands of working within health systems – and considering that surgeries are often scheduled a couple of months out – the surgeon could ensure that patients are engaged in at least 6-8 weeks of pelvic floor physical therapy before surgery to sufficiently lengthen the pelvic muscles and loosen surrounding fascia.
Short, tight pelvic floor muscles are almost universal in patients with delayed diagnosis of endometriosis and are significant generators of pain.
Appreciating sequelae of diagnostic delay
After my fellowship in advanced laparoscopic and pelvic surgery with Harry Reich, MD, and C. Y. Liu, MD, pioneers of endometriosis excision surgery, and as I did my residency in the early 2000s, I noticed puzzlement in the literature about why some patients still had lasting pain after thorough excision.
I didn’t doubt the efficacy of excision. It is the cornerstone of treatment, and at least one randomized double-blind trial2 and a systematic review and meta-analysis3 have demonstrated its superior efficacy over ablation in symptom reduction. What I did doubt was any presumption that surgery alone was enough. I knew there was more to healing when a disease process wreaks havoc on the body for more than a decade and that there were other generators of pain in addition to the endometriosis implants themselves.
As I began to focus on endometriosis in my own surgical practice, I strove to detect and treat endometriosis in teens. But in those patients with longstanding disease, I recognized patterns and began to more fully appreciate the systemic sequelae of endometriosis.
To cope with dysmenorrhea, patients curl up and assume a fetal position, tensing the abdominal muscles, inner thigh muscles, and pelvic floor muscles. Over time, these muscles come to maintain a short, tight, and painful state. (Hence the need for physical therapy to undo this decade-long pattern.)
Endometriosis implants on or near the gastrointestinal tract tug on fascia and muscles and commonly cause constipation, leading women to further overwork the pelvic floor muscles. In the case of diarrhea-predominant dysfunction, our patients squeeze pelvic floor muscles to prevent leakage. And in the case of urinary urgency, they squeeze muscles to release urine that isn’t really there.
As the chronic inflammation of the disease grows, and as pain worsens, the patient is increasingly in sympathetic overdrive (also known as ”fight or flight”), as opposed to a parasympathetic state (also known as “rest and digest”). The bowel’s motility slows, allowing the bacteria of the small intestine to grow beyond what is normal, leading to SIBO, a condition increasingly recognized by gastroenterologists and others that can impede nutrient absorption and cause bloat and pain and exacerbate constipation and diarrhea.
Key to my conceptualization of pain was a review published in 2011 by Pam Stratton, MD, of the National Institutes of Health, and Karen J. Berkley, PhD, then of Florida State University, on chronic pain and endometriosis.4 They detailed how endometriotic lesions can develop their own nerve supply that interacts directly and in a two-way fashion with the CNS – and how the lesions can engage the nervous system in ways that create comorbid conditions and pain that becomes “independent of the disease itself.”
Sensitized peripheral nerve fibers innervating a deeply infiltrating lesion on the left uterosacral ligament, for instance, can sensitize neurons in the spinal sacral segment. Branches of these nerve fibers can extend to other segments of the spinal cord, and, once sensitized themselves, turn on neurons in these other segments. There is a resultant remodeling of the central nervous system, in essence, and what is called “remote central sensitization.” The CNS becomes independent from peripheral neural processes.
I now explain to both patients and physicians that those who have had endometriosis for years have had an enduring “hand on the stove,” with a persistent signal to the CNS. Tight muscles are a hand on the stove, painful bladder syndrome is another hand on the stove, and SIBO is yet another. So are anxiety and depression.
The CNS becomes so upregulated and overloaded that messages branch out through the spinal cord to other available pathways and to other organs, muscles, and nerves. The CNS also starts firing on its own – and once it becomes its own pain generator, taking one hand off the stove (for instance, excising implants) while leaving multiple other hands on the hot stove won’t remove all pain. We must downregulate the CNS more broadly.
As I began addressing pain generators and instigators of CNS sensitization – and waiting for excision surgery until the CNS had sufficiently cooled – I saw that my patients had a better chance of more significant and lasting pain relief.
Pearls for a multimodal approach
My initial physical exam includes an assessment of the pelvic floor for overly tight musculature. An abdominal exam will usually reveal whether there is asymmetry of the abdominal wall muscles, which typically informs me of the likelihood of tightness and pulling on either side of the pelvic anatomy. On the internal exam, then, the pelvic floor muscles can be palpated and assessed. These findings will guide my referrals and my discussions with patients about the value of pelvic floor physical therapy. The cervix should be in the midline of the vagina – equidistant from the left and right vaginal fornices. If the cervix is pulled away from this midline, and a palpation of a thickened uterosacral ligament reproduces pain, endometriosis is 90% likely.
Patients who report significant “burning” pain that’s suggestive of neuropathic pain should be referred to a physical medicine rehabilitation physician or a pain specialist who can help downregulate their CNS. And patients who have symptoms of depression, anxiety disorders (including obsessive-compulsive disorder), or posttraumatic stress disorder should be referred to pain therapists, psychologists, or other mental health professionals, preferably well before surgery. I will also often discuss mindfulness practices and give my patients “meditation challenges” to achieve during the presurgical phase.
Additional points of emphasis about a multidisciplinary, multimodal approach include:
Advanced pelvic floor therapy: Therapists with specialized training in pelvic health and manual therapy utilize a range of techniques and modalities to release tension in affected muscles, fascia, nerves, and bone, and in doing so, they help to downregulate the CNS. Myofascial release, myofascial trigger point release, neural mobilization, and visceral mobilization are among these techniques. In addition to using manual therapy, many of these therapists may also employ neuromuscular reeducation and other techniques that will be helpful for the longer term.
It is important to identify physical therapists who have training in this approach; women with endometriosis often have a history of treatment by physical therapists whose focus is on incontinence and muscle strengthening (that is, Kegel exercises), which is the opposite of what endometriosis patients need.
Treating SIBO: Symptoms commonly associated with SIBO often overlap with symptoms of irritable bowel syndrome (IBS) – namely constipation, diarrhea (or both), and bloating. Indeed, many patients with undiagnosed endometriosis have been diagnosed with IBS. I send every patient who has one of these symptoms for SIBO breath testing, which utilizes carbohydrate substrates (glucose or lactulose) and measures hydrogen and/or methane in the breath.
SIBO is typically treated with rifampin, which stays in the small bowel and will not negatively affect beneficial bacteria, with or without neomycin. Gastroenterologists with more integrative practices also consider the use of herbals in addition to – or instead of – antibiotics. It can sometimes take months or a couple of years to correct SIBO, depending on how long the patient has been affected, but with presurgical diagnosis and a start on treatment, we can remove or at least tone down another instigator of CNS sensitization.
I estimate that 80% of my patients have tested positive for SIBO. Notably, in a testament to the systemic nature of endometriosis, a study published in 2009 of 355 women undergoing operative laparoscopy for suspected endometriosis found that 90% had gastrointestinal symptoms, but only 7.6% of the vast majority whose endometriosis was confirmed were found to have endometrial implants on the bowel itself.5
Addressing bladder issues: I routinely administer the PUF (Pain, Urgency, Frequency) questionnaire as part of my intake package and follow it up with conversation. For just about every patient with painful bladder syndrome, pelvic floor physical therapy in combination with a low-acid, low-potassium diet will work effectively together to reduce symptoms and pain. The IC Network offers a helpful food list, and patients can be counseled to choose foods that are also anti-inflammatory. When referrals to a urologist for bladder instillations are possible, these can be helpful as well.
Our communication with patients
Our patients need to have their symptoms and pain validated and to understand why we’re recommending these measures before surgery. Some education is necessary. Few patients will go to an integrative nutritionist, for example, if we just write a referral without explaining how years of inflammation and disruption in the gut can affect the whole body – including mental health – and that it can be corrected over time.
Also necessary is an appreciation of the fact that patients with delayed diagnoses have lived with gastrointestinal and other symptoms and patterns for so long – and often have mothers whose endometriosis caused similar symptoms – that some of their own experiences can seem almost “normal.” A patient whose mother had bowel movements every 7 days may think that 4-5 day intervals are acceptable, for instance. This means we have to carefully consider how we ask our questions.
I always ask my patients as we’re going into surgery, what percentage better are you? I’ve long aimed for at least 30% improvement, but most of the time, with pelvic floor therapy and as many other pain-generator–focused measures as possible, we’re getting them 70% better.
Excision surgery will remove the inflammation that has helped fuel the SIBO and other coconditions. Then, everything done to prepare the body must continue for some time. Certain practices, such as eating an anti-inflammatory diet, should be lifelong.
One day, it is hoped, a pediatrician or other physician will suspect endometriosis early on. The patient will see the surgeon within several months of the onset of pain, and we won’t need to unravel layers of pain generation and CNS upregulation before operating. But until this happens and we shorten the diagnostic delay, we must consider the benefits of presurgical preparation.
References
1. Orbuch I, Stein A. Beating Endo: How to Reclaim Your Life From Endometriosis. (New York: HarperCollins, 2019).
2. Healey M et al. J Minim Invasive Gynecol. 2014;21(6):999-1004.
3. Pundir J et al. J Minim Invasive Gynecol. 2017;24(5):747-56.
4. Stratton P, Berkley KJ. Hum Repro Update. 2011;17(3):327-46.
5. Maroun P et al. Aust N Z J Obstet Gynaecol. 2009;49(4):411-4.
Dr. Orbuch is a minimally invasive gynecologic surgeon in Los Angeles who specializes in endometriosis. She has no conflicts of interest to report.
Is there still a role for tubal surgery in the modern world of IVF?
According to the Centers for Disease Control and Preventions, in 2019 2.1% of all infants born in the United States were conceived by assisted reproductive technology (ART). Now 45 years old, ART, namely in vitro fertilization (IVF), is offered in nearly 500 clinics in the United States, contributing to over 300,000 treatment cycles per year.
A tubal factor is responsible for 30% of female infertility and may involve proximal and/or distal tubal occlusion, irrespective of pelvic adhesions.1 Before the advent of IVF, the sole approach to the treatment of a tubal factor had been surgery. Given its success and minimal invasiveness, IVF is increasingly being offered to circumvent a tubal factor for infertility. This month we examine the utility of surgical treatment of tubal factor infertility. The options for fertility with a history of bilateral tubal ligation was covered in a prior Reproductive Rounds column.
Tubal disease and pelvic adhesions prevent the normal transport of the oocyte and sperm through the fallopian tube. The primary etiology of tubal factor infertility is pelvic inflammatory disease, mainly caused by chlamydia or gonorrhea. Other conditions that may interfere with tubal transport include severe endometriosis, adhesions from previous surgery, or nontubal infection (for example, appendicitis, inflammatory bowel disease), pelvic tuberculosis, and salpingitis isthmica nodosa (that is, diverticulosis of the fallopian tube).
Proximal tubal occlusion
During a hysterosalpingogram (HSG), transient uterine cornual spasm can result if a woman experiences significant uterine cramping, thereby resulting in a false-positive diagnosis of proximal tubal occlusion. When a repeat HSG is gently performed with slow instillation of contrast, uterine cramping is less likely, and the tubal patency rate is 60%. PTO may also result from plugs of mucus and amorphous debris, but this is not true occlusion.2 In cases with unilateral PTO, controlled ovarian hyperstimulation with intrauterine insemination has resulted in pregnancy rates similar to those in patients with unexplained infertility.3
Reconstructive surgery for bilateral PTO has limited effectiveness and the risk of subsequent ectopic pregnancy is as high as 20%.4 A more successful option is fluoroscopic tubal catheterization (FTC), an outpatient procedure performed in a radiology or infertility center. FTC uses a coaxial catheter system where the outer catheter is guided through the tubal ostium and an inner catheter is atraumatically advanced to overcome the blockage. This procedure is 85% successful for tubal patency with 50% of patients conceiving in the first 12 months; one-third of time the tubes reocclude. After the reestablishment of patency with FTC, the chance of achieving a live birth is 22% and the risk of ectopic pregnancy is 4%.5
Treatment of distal tubal occlusion – the hydrosalpinx
Surgery for treating tubal factor infertility is most successful in women with distal tubal obstruction (DTO), often caused by a hydrosalpinx. Fimbrioplasty is the lysis of fimbrial adhesions or dilatation of fimbrial strictures; the tube is patent, but there are adhesive bands that surround the terminal end with preserved tubal rugae. Gentle introduction of an alligator laparoscopic forceps into the tubal ostium followed by opening and withdrawal of the forceps helps to stretch the tube and release minor degrees of fimbrial agglutination.6
A hydrosalpinx is diagnosed by DTO with dilation and intraluminal fluid accumulation along with the reduction/loss of endothelial cilia. Left untreated, a hydrosalpinx can lead to a 50% reduction in IVF pregnancy rates.7 Tube-sparing treatment involves neosalpingostomy to create a new tubal opening. A nonsurgical approach, ultrasound-guided aspiration of hydrosalpinges, has not been shown to significantly increase the rate of clinical pregnancy. Efficacy for improving fertility is generally poor, but depends upon tubal wall thickness, ampullary dilation, presence of mucosal folds, percentage of ciliated cells in the fimbrial end, and peritubal adhesions.8
Evidence supports that laparoscopic salpingectomy in women with hydrosalpinges improves the outcomes of IVF treatment, compared with no surgical intervention.9 The improvement in pregnancy and live birth rates likely stems from the elimination of the retrograde flow of embryotoxic fluid that disrupts implantation. Endometrial receptivity markers (endometrial cell adhesion molecules, integrins, and HOXA10) have been shown to be reduced in the presence of hydrosalpinx.10 A small, randomized trial demonstrated that bipolar diathermy prior to IVF improved pregnancy outcomes.11 PTO was not more effective than salpingectomy. Conceptions, without IVF, have been reported following salpingectomy for unilateral hydrosalpinx.12
In a series including 434 patients with DTO who underwent laparoscopic fimbrioplasty (enlargement of the ostium) or neosalpingostomy (creation of a new ostium) by a single surgeon, 5-year actuarial delivery rates decreased as the severity of tubal occlusion increased; the ectopic rate was stable at approximately 15%.13 A prospective study reported that the relative increase in the pregnancy rate after salpingectomy was greatest in women with a large hydrosalpinx visible on ultrasound.14
Because of the possible risks of decreased ovarian reserve secondary to interruption of ovarian blood supply, salpingectomy should be done with minimal thermal injury and very close to the fallopian tube.
Summary
Surgery may be considered for young women with mild distal tubal disease as one surgical procedure can lead to several pregnancies whereas IVF must be performed each time pregnancy is desired. IVF is more likely than surgery to be successful in women with bilateral hydrosalpinx, in those with pelvic adhesions, in older reproductive aged women, and for both proximal and distal tubal occlusion.15 An online prediction calculator from the Society for Assisted Reproductive Technology (SART) can be helpful in counseling patients on personalized expectations for IVF pregnancy outcomes.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Ambildhuke K et al. Cureus. 2022;1:14(11):e30990.
2. Fatemeh Z et al. Br J Radiol. 2021 Jun 1;94(1122):20201386.
3. Farhi J et al. Fertil Steril. 2007 Aug;88(2):396.
4. Honoré GM et al. Fertil Steril. 1999;71(5):785.
5. De Silva PM et al. Hum Reprod. 2017;32(4):836.
6. Namnoum A and Murphy A. “Diagnostic and Operative Laparoscopy,” in Te Linde’s Operative Gynecology, 8th ed. Philadelphia: Lippincott-Raven, 1997, pp. 389.
7. Camus E et al.Hum Reprod. 1999;14(5):1243.
8. Marana R et al. Hum Reprod. 1999;14(12):2991-5.
9. Johnson N et al. Cochrane Database Syst Rev. 2010 Jan 20;2010(1):CD002125.
10. Savaris RF et al. Fertil Steril. 2006 Jan;85(1):188.
11. Kontoravdis A et al. Fertil Steril. 2006;86(6):1642.
12. Sagoskin AW et al. Hum Reprod. 2003;18(12):2634.
13. Audebert A et al. Fertil Steril. 2014;102(4):1203.
14. Bildirici I et al. Hum Reprod. 2001;16(11):2422.
15. Practice Committee of the American Society for Reproductive Medicine. Fertil Steril. 2012;97(3):539.
According to the Centers for Disease Control and Preventions, in 2019 2.1% of all infants born in the United States were conceived by assisted reproductive technology (ART). Now 45 years old, ART, namely in vitro fertilization (IVF), is offered in nearly 500 clinics in the United States, contributing to over 300,000 treatment cycles per year.
A tubal factor is responsible for 30% of female infertility and may involve proximal and/or distal tubal occlusion, irrespective of pelvic adhesions.1 Before the advent of IVF, the sole approach to the treatment of a tubal factor had been surgery. Given its success and minimal invasiveness, IVF is increasingly being offered to circumvent a tubal factor for infertility. This month we examine the utility of surgical treatment of tubal factor infertility. The options for fertility with a history of bilateral tubal ligation was covered in a prior Reproductive Rounds column.
Tubal disease and pelvic adhesions prevent the normal transport of the oocyte and sperm through the fallopian tube. The primary etiology of tubal factor infertility is pelvic inflammatory disease, mainly caused by chlamydia or gonorrhea. Other conditions that may interfere with tubal transport include severe endometriosis, adhesions from previous surgery, or nontubal infection (for example, appendicitis, inflammatory bowel disease), pelvic tuberculosis, and salpingitis isthmica nodosa (that is, diverticulosis of the fallopian tube).
Proximal tubal occlusion
During a hysterosalpingogram (HSG), transient uterine cornual spasm can result if a woman experiences significant uterine cramping, thereby resulting in a false-positive diagnosis of proximal tubal occlusion. When a repeat HSG is gently performed with slow instillation of contrast, uterine cramping is less likely, and the tubal patency rate is 60%. PTO may also result from plugs of mucus and amorphous debris, but this is not true occlusion.2 In cases with unilateral PTO, controlled ovarian hyperstimulation with intrauterine insemination has resulted in pregnancy rates similar to those in patients with unexplained infertility.3
Reconstructive surgery for bilateral PTO has limited effectiveness and the risk of subsequent ectopic pregnancy is as high as 20%.4 A more successful option is fluoroscopic tubal catheterization (FTC), an outpatient procedure performed in a radiology or infertility center. FTC uses a coaxial catheter system where the outer catheter is guided through the tubal ostium and an inner catheter is atraumatically advanced to overcome the blockage. This procedure is 85% successful for tubal patency with 50% of patients conceiving in the first 12 months; one-third of time the tubes reocclude. After the reestablishment of patency with FTC, the chance of achieving a live birth is 22% and the risk of ectopic pregnancy is 4%.5
Treatment of distal tubal occlusion – the hydrosalpinx
Surgery for treating tubal factor infertility is most successful in women with distal tubal obstruction (DTO), often caused by a hydrosalpinx. Fimbrioplasty is the lysis of fimbrial adhesions or dilatation of fimbrial strictures; the tube is patent, but there are adhesive bands that surround the terminal end with preserved tubal rugae. Gentle introduction of an alligator laparoscopic forceps into the tubal ostium followed by opening and withdrawal of the forceps helps to stretch the tube and release minor degrees of fimbrial agglutination.6
A hydrosalpinx is diagnosed by DTO with dilation and intraluminal fluid accumulation along with the reduction/loss of endothelial cilia. Left untreated, a hydrosalpinx can lead to a 50% reduction in IVF pregnancy rates.7 Tube-sparing treatment involves neosalpingostomy to create a new tubal opening. A nonsurgical approach, ultrasound-guided aspiration of hydrosalpinges, has not been shown to significantly increase the rate of clinical pregnancy. Efficacy for improving fertility is generally poor, but depends upon tubal wall thickness, ampullary dilation, presence of mucosal folds, percentage of ciliated cells in the fimbrial end, and peritubal adhesions.8
Evidence supports that laparoscopic salpingectomy in women with hydrosalpinges improves the outcomes of IVF treatment, compared with no surgical intervention.9 The improvement in pregnancy and live birth rates likely stems from the elimination of the retrograde flow of embryotoxic fluid that disrupts implantation. Endometrial receptivity markers (endometrial cell adhesion molecules, integrins, and HOXA10) have been shown to be reduced in the presence of hydrosalpinx.10 A small, randomized trial demonstrated that bipolar diathermy prior to IVF improved pregnancy outcomes.11 PTO was not more effective than salpingectomy. Conceptions, without IVF, have been reported following salpingectomy for unilateral hydrosalpinx.12
In a series including 434 patients with DTO who underwent laparoscopic fimbrioplasty (enlargement of the ostium) or neosalpingostomy (creation of a new ostium) by a single surgeon, 5-year actuarial delivery rates decreased as the severity of tubal occlusion increased; the ectopic rate was stable at approximately 15%.13 A prospective study reported that the relative increase in the pregnancy rate after salpingectomy was greatest in women with a large hydrosalpinx visible on ultrasound.14
Because of the possible risks of decreased ovarian reserve secondary to interruption of ovarian blood supply, salpingectomy should be done with minimal thermal injury and very close to the fallopian tube.
Summary
Surgery may be considered for young women with mild distal tubal disease as one surgical procedure can lead to several pregnancies whereas IVF must be performed each time pregnancy is desired. IVF is more likely than surgery to be successful in women with bilateral hydrosalpinx, in those with pelvic adhesions, in older reproductive aged women, and for both proximal and distal tubal occlusion.15 An online prediction calculator from the Society for Assisted Reproductive Technology (SART) can be helpful in counseling patients on personalized expectations for IVF pregnancy outcomes.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Ambildhuke K et al. Cureus. 2022;1:14(11):e30990.
2. Fatemeh Z et al. Br J Radiol. 2021 Jun 1;94(1122):20201386.
3. Farhi J et al. Fertil Steril. 2007 Aug;88(2):396.
4. Honoré GM et al. Fertil Steril. 1999;71(5):785.
5. De Silva PM et al. Hum Reprod. 2017;32(4):836.
6. Namnoum A and Murphy A. “Diagnostic and Operative Laparoscopy,” in Te Linde’s Operative Gynecology, 8th ed. Philadelphia: Lippincott-Raven, 1997, pp. 389.
7. Camus E et al.Hum Reprod. 1999;14(5):1243.
8. Marana R et al. Hum Reprod. 1999;14(12):2991-5.
9. Johnson N et al. Cochrane Database Syst Rev. 2010 Jan 20;2010(1):CD002125.
10. Savaris RF et al. Fertil Steril. 2006 Jan;85(1):188.
11. Kontoravdis A et al. Fertil Steril. 2006;86(6):1642.
12. Sagoskin AW et al. Hum Reprod. 2003;18(12):2634.
13. Audebert A et al. Fertil Steril. 2014;102(4):1203.
14. Bildirici I et al. Hum Reprod. 2001;16(11):2422.
15. Practice Committee of the American Society for Reproductive Medicine. Fertil Steril. 2012;97(3):539.
According to the Centers for Disease Control and Preventions, in 2019 2.1% of all infants born in the United States were conceived by assisted reproductive technology (ART). Now 45 years old, ART, namely in vitro fertilization (IVF), is offered in nearly 500 clinics in the United States, contributing to over 300,000 treatment cycles per year.
A tubal factor is responsible for 30% of female infertility and may involve proximal and/or distal tubal occlusion, irrespective of pelvic adhesions.1 Before the advent of IVF, the sole approach to the treatment of a tubal factor had been surgery. Given its success and minimal invasiveness, IVF is increasingly being offered to circumvent a tubal factor for infertility. This month we examine the utility of surgical treatment of tubal factor infertility. The options for fertility with a history of bilateral tubal ligation was covered in a prior Reproductive Rounds column.
Tubal disease and pelvic adhesions prevent the normal transport of the oocyte and sperm through the fallopian tube. The primary etiology of tubal factor infertility is pelvic inflammatory disease, mainly caused by chlamydia or gonorrhea. Other conditions that may interfere with tubal transport include severe endometriosis, adhesions from previous surgery, or nontubal infection (for example, appendicitis, inflammatory bowel disease), pelvic tuberculosis, and salpingitis isthmica nodosa (that is, diverticulosis of the fallopian tube).
Proximal tubal occlusion
During a hysterosalpingogram (HSG), transient uterine cornual spasm can result if a woman experiences significant uterine cramping, thereby resulting in a false-positive diagnosis of proximal tubal occlusion. When a repeat HSG is gently performed with slow instillation of contrast, uterine cramping is less likely, and the tubal patency rate is 60%. PTO may also result from plugs of mucus and amorphous debris, but this is not true occlusion.2 In cases with unilateral PTO, controlled ovarian hyperstimulation with intrauterine insemination has resulted in pregnancy rates similar to those in patients with unexplained infertility.3
Reconstructive surgery for bilateral PTO has limited effectiveness and the risk of subsequent ectopic pregnancy is as high as 20%.4 A more successful option is fluoroscopic tubal catheterization (FTC), an outpatient procedure performed in a radiology or infertility center. FTC uses a coaxial catheter system where the outer catheter is guided through the tubal ostium and an inner catheter is atraumatically advanced to overcome the blockage. This procedure is 85% successful for tubal patency with 50% of patients conceiving in the first 12 months; one-third of time the tubes reocclude. After the reestablishment of patency with FTC, the chance of achieving a live birth is 22% and the risk of ectopic pregnancy is 4%.5
Treatment of distal tubal occlusion – the hydrosalpinx
Surgery for treating tubal factor infertility is most successful in women with distal tubal obstruction (DTO), often caused by a hydrosalpinx. Fimbrioplasty is the lysis of fimbrial adhesions or dilatation of fimbrial strictures; the tube is patent, but there are adhesive bands that surround the terminal end with preserved tubal rugae. Gentle introduction of an alligator laparoscopic forceps into the tubal ostium followed by opening and withdrawal of the forceps helps to stretch the tube and release minor degrees of fimbrial agglutination.6
A hydrosalpinx is diagnosed by DTO with dilation and intraluminal fluid accumulation along with the reduction/loss of endothelial cilia. Left untreated, a hydrosalpinx can lead to a 50% reduction in IVF pregnancy rates.7 Tube-sparing treatment involves neosalpingostomy to create a new tubal opening. A nonsurgical approach, ultrasound-guided aspiration of hydrosalpinges, has not been shown to significantly increase the rate of clinical pregnancy. Efficacy for improving fertility is generally poor, but depends upon tubal wall thickness, ampullary dilation, presence of mucosal folds, percentage of ciliated cells in the fimbrial end, and peritubal adhesions.8
Evidence supports that laparoscopic salpingectomy in women with hydrosalpinges improves the outcomes of IVF treatment, compared with no surgical intervention.9 The improvement in pregnancy and live birth rates likely stems from the elimination of the retrograde flow of embryotoxic fluid that disrupts implantation. Endometrial receptivity markers (endometrial cell adhesion molecules, integrins, and HOXA10) have been shown to be reduced in the presence of hydrosalpinx.10 A small, randomized trial demonstrated that bipolar diathermy prior to IVF improved pregnancy outcomes.11 PTO was not more effective than salpingectomy. Conceptions, without IVF, have been reported following salpingectomy for unilateral hydrosalpinx.12
In a series including 434 patients with DTO who underwent laparoscopic fimbrioplasty (enlargement of the ostium) or neosalpingostomy (creation of a new ostium) by a single surgeon, 5-year actuarial delivery rates decreased as the severity of tubal occlusion increased; the ectopic rate was stable at approximately 15%.13 A prospective study reported that the relative increase in the pregnancy rate after salpingectomy was greatest in women with a large hydrosalpinx visible on ultrasound.14
Because of the possible risks of decreased ovarian reserve secondary to interruption of ovarian blood supply, salpingectomy should be done with minimal thermal injury and very close to the fallopian tube.
Summary
Surgery may be considered for young women with mild distal tubal disease as one surgical procedure can lead to several pregnancies whereas IVF must be performed each time pregnancy is desired. IVF is more likely than surgery to be successful in women with bilateral hydrosalpinx, in those with pelvic adhesions, in older reproductive aged women, and for both proximal and distal tubal occlusion.15 An online prediction calculator from the Society for Assisted Reproductive Technology (SART) can be helpful in counseling patients on personalized expectations for IVF pregnancy outcomes.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
References
1. Ambildhuke K et al. Cureus. 2022;1:14(11):e30990.
2. Fatemeh Z et al. Br J Radiol. 2021 Jun 1;94(1122):20201386.
3. Farhi J et al. Fertil Steril. 2007 Aug;88(2):396.
4. Honoré GM et al. Fertil Steril. 1999;71(5):785.
5. De Silva PM et al. Hum Reprod. 2017;32(4):836.
6. Namnoum A and Murphy A. “Diagnostic and Operative Laparoscopy,” in Te Linde’s Operative Gynecology, 8th ed. Philadelphia: Lippincott-Raven, 1997, pp. 389.
7. Camus E et al.Hum Reprod. 1999;14(5):1243.
8. Marana R et al. Hum Reprod. 1999;14(12):2991-5.
9. Johnson N et al. Cochrane Database Syst Rev. 2010 Jan 20;2010(1):CD002125.
10. Savaris RF et al. Fertil Steril. 2006 Jan;85(1):188.
11. Kontoravdis A et al. Fertil Steril. 2006;86(6):1642.
12. Sagoskin AW et al. Hum Reprod. 2003;18(12):2634.
13. Audebert A et al. Fertil Steril. 2014;102(4):1203.
14. Bildirici I et al. Hum Reprod. 2001;16(11):2422.
15. Practice Committee of the American Society for Reproductive Medicine. Fertil Steril. 2012;97(3):539.
Vacuum device for postpartum hemorrhage works well in real world
Postpartum hemorrhage is the leading cause of maternal mortality worldwide, accounting for 25% of deaths from obstetric causes. Although balloon tamponade has been widely used to manage uncontrolled postpartum bleeding, a recent evaluation of an intrauterine vacuum-induced hemorrhage control device demonstrated impressive safety and effectiveness, researchers reported at the meeting sponsored by the Society for Maternal-Fetal Medicine.
“It’s exciting to see new technology and new potential treatment modalities. We just don’t have that many tools in our toolkit right now,” said Dena Goffman, MD, professor of women’s health and obstetrics and gynecology and vice chair of quality and patient safety at Columbia University’s Irving Medical Center, in New York, who presented the findings.
Dr. Goffman led an earlier multicenter prospective single-arm treatment study of the Jada System, a vacuum device marketed by Organon. The U.S. Food and Drug Administration approved use of the Jada System in October 2020.
Dr. Goffman said she and her colleagues felt “a next logical step would be to see what happens with real-world use.” In the new study, researchers at 16 U.S. medical centers reviewed medical charts of 800 women who underwent treatment with the Jada System between October 2020 and April 2022.
Treatment was successful in 92.5% of the vaginal births (n = 530) and 83.7% of the cesarean births (n = 270), similar to the results of the initial treatment trial that led to FDA approval, according to the researchers. For both types of delivery, bleeding was controlled in less than 5 minutes for most patients. Three serious adverse events were identified that could have been related to use of the device (two in vaginal births, one in cesarean birth), they reported.
Although the study was not designed to directly compare the Jada System with balloon tamponade, in a recent meta-analysis, it was estimated that tamponade controls postpartum hemorrhage in roughly 87% of cases, with complication rates in as many as 6.5% among women who undergo the procedure.
Dr. Goffman pointed out additional benefits. The vacuum device typically must stay in place for less time (3.1 hours for vaginal birth and 4.6 hours for cesarean birth) than balloon tamponade, allowing women to recover more quickly. In the initial trial, which Dr. Goffman helped conduct, 98% of clinicians reported that the device was easy to use, which increases its attractiveness in lower-income countries. Dr. Goffman felt that the device “has potential for huge impact” in those countries, given the high rates of maternal morbidity and mortality in these areas.
Amber Samuel, MD, medical director of OBSETRIX Maternal Fetal Medicine Specialists of Houston, said the device recently became available in the hospitals in which she works, and she has used the Jada System several times. Like Dr. Goffman, she was excited to have a new tool for treating a life-threatening condition.
Although the device has been on the market for more than 2 years, Dr. Samuel felt clinicians who were reluctant to adopt a new technology would be reassured by the findings.
“We should make sure that it’s effective, and we should know what the safety profile is,” said Dr. Samuel, adding that “the more data we have, the more we’re able to counsel patients and work this into our protocols for what is a really common obstetric problem.”
Both Dr. Goffman and Dr. Samuel agreed that more data, ideally from randomized clinical trials, are needed to convince professional groups such as the American College of Obstetricians and Gynecologists to state a clear preference for use of vacuum-induced hemorrhage control devices over balloon tamponade.
“We should be supporting further investigation,” Dr. Goffman said, “but for people who have this tool available to them now, I think they can feel confident in using it.”
The study was funded by Alydia Health, the manufacturer of the Jada System. Alydia Health was acquired by Organon in 2021. Study sites received research-related financial support, but none of the authors received direct payments from Alydia Health/Organon. Dr. Goffman serves on the scientific advisory board of Alydia Health/Organon. Dr. Samuel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Postpartum hemorrhage is the leading cause of maternal mortality worldwide, accounting for 25% of deaths from obstetric causes. Although balloon tamponade has been widely used to manage uncontrolled postpartum bleeding, a recent evaluation of an intrauterine vacuum-induced hemorrhage control device demonstrated impressive safety and effectiveness, researchers reported at the meeting sponsored by the Society for Maternal-Fetal Medicine.
“It’s exciting to see new technology and new potential treatment modalities. We just don’t have that many tools in our toolkit right now,” said Dena Goffman, MD, professor of women’s health and obstetrics and gynecology and vice chair of quality and patient safety at Columbia University’s Irving Medical Center, in New York, who presented the findings.
Dr. Goffman led an earlier multicenter prospective single-arm treatment study of the Jada System, a vacuum device marketed by Organon. The U.S. Food and Drug Administration approved use of the Jada System in October 2020.
Dr. Goffman said she and her colleagues felt “a next logical step would be to see what happens with real-world use.” In the new study, researchers at 16 U.S. medical centers reviewed medical charts of 800 women who underwent treatment with the Jada System between October 2020 and April 2022.
Treatment was successful in 92.5% of the vaginal births (n = 530) and 83.7% of the cesarean births (n = 270), similar to the results of the initial treatment trial that led to FDA approval, according to the researchers. For both types of delivery, bleeding was controlled in less than 5 minutes for most patients. Three serious adverse events were identified that could have been related to use of the device (two in vaginal births, one in cesarean birth), they reported.
Although the study was not designed to directly compare the Jada System with balloon tamponade, in a recent meta-analysis, it was estimated that tamponade controls postpartum hemorrhage in roughly 87% of cases, with complication rates in as many as 6.5% among women who undergo the procedure.
Dr. Goffman pointed out additional benefits. The vacuum device typically must stay in place for less time (3.1 hours for vaginal birth and 4.6 hours for cesarean birth) than balloon tamponade, allowing women to recover more quickly. In the initial trial, which Dr. Goffman helped conduct, 98% of clinicians reported that the device was easy to use, which increases its attractiveness in lower-income countries. Dr. Goffman felt that the device “has potential for huge impact” in those countries, given the high rates of maternal morbidity and mortality in these areas.
Amber Samuel, MD, medical director of OBSETRIX Maternal Fetal Medicine Specialists of Houston, said the device recently became available in the hospitals in which she works, and she has used the Jada System several times. Like Dr. Goffman, she was excited to have a new tool for treating a life-threatening condition.
Although the device has been on the market for more than 2 years, Dr. Samuel felt clinicians who were reluctant to adopt a new technology would be reassured by the findings.
“We should make sure that it’s effective, and we should know what the safety profile is,” said Dr. Samuel, adding that “the more data we have, the more we’re able to counsel patients and work this into our protocols for what is a really common obstetric problem.”
Both Dr. Goffman and Dr. Samuel agreed that more data, ideally from randomized clinical trials, are needed to convince professional groups such as the American College of Obstetricians and Gynecologists to state a clear preference for use of vacuum-induced hemorrhage control devices over balloon tamponade.
“We should be supporting further investigation,” Dr. Goffman said, “but for people who have this tool available to them now, I think they can feel confident in using it.”
The study was funded by Alydia Health, the manufacturer of the Jada System. Alydia Health was acquired by Organon in 2021. Study sites received research-related financial support, but none of the authors received direct payments from Alydia Health/Organon. Dr. Goffman serves on the scientific advisory board of Alydia Health/Organon. Dr. Samuel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Postpartum hemorrhage is the leading cause of maternal mortality worldwide, accounting for 25% of deaths from obstetric causes. Although balloon tamponade has been widely used to manage uncontrolled postpartum bleeding, a recent evaluation of an intrauterine vacuum-induced hemorrhage control device demonstrated impressive safety and effectiveness, researchers reported at the meeting sponsored by the Society for Maternal-Fetal Medicine.
“It’s exciting to see new technology and new potential treatment modalities. We just don’t have that many tools in our toolkit right now,” said Dena Goffman, MD, professor of women’s health and obstetrics and gynecology and vice chair of quality and patient safety at Columbia University’s Irving Medical Center, in New York, who presented the findings.
Dr. Goffman led an earlier multicenter prospective single-arm treatment study of the Jada System, a vacuum device marketed by Organon. The U.S. Food and Drug Administration approved use of the Jada System in October 2020.
Dr. Goffman said she and her colleagues felt “a next logical step would be to see what happens with real-world use.” In the new study, researchers at 16 U.S. medical centers reviewed medical charts of 800 women who underwent treatment with the Jada System between October 2020 and April 2022.
Treatment was successful in 92.5% of the vaginal births (n = 530) and 83.7% of the cesarean births (n = 270), similar to the results of the initial treatment trial that led to FDA approval, according to the researchers. For both types of delivery, bleeding was controlled in less than 5 minutes for most patients. Three serious adverse events were identified that could have been related to use of the device (two in vaginal births, one in cesarean birth), they reported.
Although the study was not designed to directly compare the Jada System with balloon tamponade, in a recent meta-analysis, it was estimated that tamponade controls postpartum hemorrhage in roughly 87% of cases, with complication rates in as many as 6.5% among women who undergo the procedure.
Dr. Goffman pointed out additional benefits. The vacuum device typically must stay in place for less time (3.1 hours for vaginal birth and 4.6 hours for cesarean birth) than balloon tamponade, allowing women to recover more quickly. In the initial trial, which Dr. Goffman helped conduct, 98% of clinicians reported that the device was easy to use, which increases its attractiveness in lower-income countries. Dr. Goffman felt that the device “has potential for huge impact” in those countries, given the high rates of maternal morbidity and mortality in these areas.
Amber Samuel, MD, medical director of OBSETRIX Maternal Fetal Medicine Specialists of Houston, said the device recently became available in the hospitals in which she works, and she has used the Jada System several times. Like Dr. Goffman, she was excited to have a new tool for treating a life-threatening condition.
Although the device has been on the market for more than 2 years, Dr. Samuel felt clinicians who were reluctant to adopt a new technology would be reassured by the findings.
“We should make sure that it’s effective, and we should know what the safety profile is,” said Dr. Samuel, adding that “the more data we have, the more we’re able to counsel patients and work this into our protocols for what is a really common obstetric problem.”
Both Dr. Goffman and Dr. Samuel agreed that more data, ideally from randomized clinical trials, are needed to convince professional groups such as the American College of Obstetricians and Gynecologists to state a clear preference for use of vacuum-induced hemorrhage control devices over balloon tamponade.
“We should be supporting further investigation,” Dr. Goffman said, “but for people who have this tool available to them now, I think they can feel confident in using it.”
The study was funded by Alydia Health, the manufacturer of the Jada System. Alydia Health was acquired by Organon in 2021. Study sites received research-related financial support, but none of the authors received direct payments from Alydia Health/Organon. Dr. Goffman serves on the scientific advisory board of Alydia Health/Organon. Dr. Samuel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE PREGNANCY MEETING