User login
ACIP Recommends Pneumococcal Vaccine for Adults 50 Years or Older
The US Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP) now recommends a pneumococcal conjugate vaccine (PCV) for all PCV-naive adults aged 50 years or older. The new recommendation, which passed with an ACIP member vote of 14 for and one against, expands the current age-based recommendations, which include children younger than 5 years and adults older than 65 years, as well as adults aged 19-64 years with underlying conditions or risk factors who have not received a PCV and certain adults who have received PCV13 but not PCV20.
The decision was based in part on economic analyses of the use of PCV in adults aged 50-64 years in the United States. Miwako Kobayashi, MD, presented the summary of the Pneumococcal Vaccines Work Group’s interpretation of the evidence and the proposed recommendation in a meeting of the ACIP on October 23, 2024, when the ACIP voting occurred.
Data from the CDC show an increase in the relative burden of pneumococcal disease in adults aged 50-64 years based in part on the success of the pediatric PCV program, she said.
Health equity was another main factor in the Work Group’s decision to recommend vaccination for adults aged 50 years or older. “Disparities in pneumococcal vaccine coverage by race and ethnicity exist for both age-based and risk-based indications,” Kobayashi noted in her presentation. The Work Group acknowledged that the overall effect of a vaccine recommendation on health equity is complex, but the majority agreed that the update would improve health equity by increasing vaccine coverage for those with known or unknown risk factors and providing protection at an earlier age when some populations already experience elevated disease rates, she said.
As for safety, the Work Group concluded that the undesirable anticipated effects of PCVs are minimal, despite the potential signal for Guillain-Barré Syndrome, and the CDC and US Food and Drug Administration will continue to monitor post-licensure safety of PCVs.
Support Not Universal
A majority of the ACIP Pneumococcal Vaccines Work Group supported the approved option, but agreed that a future booster dose may be needed, Work Group Chair James Loehr, MD, said in his introductory presentation.
Overall, key uncertainties remain, including indirect effects of new pediatric pneumococcal vaccines on adults, data on the duration of protection with adult vaccinations, and the impact new higher-valency vaccines have on adults, several of which are in development, Loehr said.
A new 21-valent PCV, known as PCV 21, was approved by the FDA for adults aged 18 years or older in June 2024, said Loehr. “PCV21 is not PCV20 with one additional serotype” and provides additional protection, he emphasized. The Work Group examined models involving PCV21 and the existing PCV20. However, a majority of the Work Group agreed that having age-based recommendations based on vaccine product would be more challenging to implement and that insurance coverage may be a factor given the recent approval of PCV21. Therefore, the proposal submitted to the full ACIP was not for a specific PCV.
Notably, Loehr said that, although as Work Group Chair he was tasked with making the motion in favor of the recommendation, he voted against it as a voting member because of his strong opinion that only the PCV21 vaccine is needed for vaccine-naive adults aged 50 or older. “I think that PCV21 is a better vaccine that targets more serotypes,” he said during the discussion. Data presented at the February 2024 ACIP meeting showed more than 80% coverage vs less than 60% coverage for invasive pneumococcal disease with PCV21 vs PCV20 among adults aged 65 years or older and those aged 19-64 years with a risk-based indication, Loehr said.
A version of this article appeared on Medscape.com.
The US Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP) now recommends a pneumococcal conjugate vaccine (PCV) for all PCV-naive adults aged 50 years or older. The new recommendation, which passed with an ACIP member vote of 14 for and one against, expands the current age-based recommendations, which include children younger than 5 years and adults older than 65 years, as well as adults aged 19-64 years with underlying conditions or risk factors who have not received a PCV and certain adults who have received PCV13 but not PCV20.
The decision was based in part on economic analyses of the use of PCV in adults aged 50-64 years in the United States. Miwako Kobayashi, MD, presented the summary of the Pneumococcal Vaccines Work Group’s interpretation of the evidence and the proposed recommendation in a meeting of the ACIP on October 23, 2024, when the ACIP voting occurred.
Data from the CDC show an increase in the relative burden of pneumococcal disease in adults aged 50-64 years based in part on the success of the pediatric PCV program, she said.
Health equity was another main factor in the Work Group’s decision to recommend vaccination for adults aged 50 years or older. “Disparities in pneumococcal vaccine coverage by race and ethnicity exist for both age-based and risk-based indications,” Kobayashi noted in her presentation. The Work Group acknowledged that the overall effect of a vaccine recommendation on health equity is complex, but the majority agreed that the update would improve health equity by increasing vaccine coverage for those with known or unknown risk factors and providing protection at an earlier age when some populations already experience elevated disease rates, she said.
As for safety, the Work Group concluded that the undesirable anticipated effects of PCVs are minimal, despite the potential signal for Guillain-Barré Syndrome, and the CDC and US Food and Drug Administration will continue to monitor post-licensure safety of PCVs.
Support Not Universal
A majority of the ACIP Pneumococcal Vaccines Work Group supported the approved option, but agreed that a future booster dose may be needed, Work Group Chair James Loehr, MD, said in his introductory presentation.
Overall, key uncertainties remain, including indirect effects of new pediatric pneumococcal vaccines on adults, data on the duration of protection with adult vaccinations, and the impact new higher-valency vaccines have on adults, several of which are in development, Loehr said.
A new 21-valent PCV, known as PCV 21, was approved by the FDA for adults aged 18 years or older in June 2024, said Loehr. “PCV21 is not PCV20 with one additional serotype” and provides additional protection, he emphasized. The Work Group examined models involving PCV21 and the existing PCV20. However, a majority of the Work Group agreed that having age-based recommendations based on vaccine product would be more challenging to implement and that insurance coverage may be a factor given the recent approval of PCV21. Therefore, the proposal submitted to the full ACIP was not for a specific PCV.
Notably, Loehr said that, although as Work Group Chair he was tasked with making the motion in favor of the recommendation, he voted against it as a voting member because of his strong opinion that only the PCV21 vaccine is needed for vaccine-naive adults aged 50 or older. “I think that PCV21 is a better vaccine that targets more serotypes,” he said during the discussion. Data presented at the February 2024 ACIP meeting showed more than 80% coverage vs less than 60% coverage for invasive pneumococcal disease with PCV21 vs PCV20 among adults aged 65 years or older and those aged 19-64 years with a risk-based indication, Loehr said.
A version of this article appeared on Medscape.com.
The US Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices (ACIP) now recommends a pneumococcal conjugate vaccine (PCV) for all PCV-naive adults aged 50 years or older. The new recommendation, which passed with an ACIP member vote of 14 for and one against, expands the current age-based recommendations, which include children younger than 5 years and adults older than 65 years, as well as adults aged 19-64 years with underlying conditions or risk factors who have not received a PCV and certain adults who have received PCV13 but not PCV20.
The decision was based in part on economic analyses of the use of PCV in adults aged 50-64 years in the United States. Miwako Kobayashi, MD, presented the summary of the Pneumococcal Vaccines Work Group’s interpretation of the evidence and the proposed recommendation in a meeting of the ACIP on October 23, 2024, when the ACIP voting occurred.
Data from the CDC show an increase in the relative burden of pneumococcal disease in adults aged 50-64 years based in part on the success of the pediatric PCV program, she said.
Health equity was another main factor in the Work Group’s decision to recommend vaccination for adults aged 50 years or older. “Disparities in pneumococcal vaccine coverage by race and ethnicity exist for both age-based and risk-based indications,” Kobayashi noted in her presentation. The Work Group acknowledged that the overall effect of a vaccine recommendation on health equity is complex, but the majority agreed that the update would improve health equity by increasing vaccine coverage for those with known or unknown risk factors and providing protection at an earlier age when some populations already experience elevated disease rates, she said.
As for safety, the Work Group concluded that the undesirable anticipated effects of PCVs are minimal, despite the potential signal for Guillain-Barré Syndrome, and the CDC and US Food and Drug Administration will continue to monitor post-licensure safety of PCVs.
Support Not Universal
A majority of the ACIP Pneumococcal Vaccines Work Group supported the approved option, but agreed that a future booster dose may be needed, Work Group Chair James Loehr, MD, said in his introductory presentation.
Overall, key uncertainties remain, including indirect effects of new pediatric pneumococcal vaccines on adults, data on the duration of protection with adult vaccinations, and the impact new higher-valency vaccines have on adults, several of which are in development, Loehr said.
A new 21-valent PCV, known as PCV 21, was approved by the FDA for adults aged 18 years or older in June 2024, said Loehr. “PCV21 is not PCV20 with one additional serotype” and provides additional protection, he emphasized. The Work Group examined models involving PCV21 and the existing PCV20. However, a majority of the Work Group agreed that having age-based recommendations based on vaccine product would be more challenging to implement and that insurance coverage may be a factor given the recent approval of PCV21. Therefore, the proposal submitted to the full ACIP was not for a specific PCV.
Notably, Loehr said that, although as Work Group Chair he was tasked with making the motion in favor of the recommendation, he voted against it as a voting member because of his strong opinion that only the PCV21 vaccine is needed for vaccine-naive adults aged 50 or older. “I think that PCV21 is a better vaccine that targets more serotypes,” he said during the discussion. Data presented at the February 2024 ACIP meeting showed more than 80% coverage vs less than 60% coverage for invasive pneumococcal disease with PCV21 vs PCV20 among adults aged 65 years or older and those aged 19-64 years with a risk-based indication, Loehr said.
A version of this article appeared on Medscape.com.
Home HPV Testing: A New Frontier in Primary Care
Human papillomavirus (HPV) is one of the most common sexually transmitted infections and persistent infection with high-risk strains is the leading cause of cervical cancer. Fortunately, vaccines are available to prevent many HPV-related diseases, but they haven’t fully eliminated the risks. Cervical cancer screening remains essential for early detection and prevention.
The US Preventive Services Task Force (USPSTF) currently recommends regular cervical cancer screenings for women aged 21-65. These screenings can include a Pap test every 3 years, a combination of HPV testing and Pap smear every 5 years, or high-risk HPV testing alone every 5 years, depending on age and individual risk factors.
Although these guidelines are currently under review, routine screenings have been instrumental in reducing cervical cancer rates. However, many patients still face barriers that prevent them from accessing these services. Common challenges include discomfort with pelvic exams, lack of time, and limited access to healthcare services. In recent years, advancements in home-based diagnostic testing have opened new avenues for preventative care.
Home HPV testing is one such advancement, offering an alternative to traditional in-office screening methods. While the US Food and Drug Administration (FDA) has not yet approved home HPV testing, self-collection in clinical settings is available and gaining traction. Primary care physicians can integrate this self-collection method into their practices, helping to close the screening gap, especially for underserved populations.
If approved, home HPV testing could be a game-changer for patients who have difficulty attending in-person visits. Geographical barriers, transportation issues, and personal discomfort with in-office exams can prevent patients from receiving the care they need. Home testing eliminates many of these hurdles, enabling patients to perform the test in the comfort of their own homes at a time that works for them. This flexibility is particularly beneficial for rural and underserved populations, where access to healthcare is limited.
Similarly, in-office self-collection offers a comfortable alternative for those who find traditional pelvic exams uncomfortable or distressing. Self-administered HPV tests allow patients to take control of their cervical cancer screening, fostering empowerment and personal responsibility for their health. By reducing the discomfort and inconvenience of traditional screening, self-collection can improve adherence to screening guidelines, leading to earlier detection and prevention of cervical cancer.
Primary care physicians may soon offer both in-office and at-home testing options, tailoring the approach to each patient’s unique needs. Virtual appointments provide an excellent opportunity to educate patients about the importance of cervical cancer screening and offer guidance on using home HPV testing kits. This personalized care ensures patients feel supported even without in-person visits. If home testing becomes FDA approved, patients could receive test kits by mail, perform the test, and send it back to the lab for analysis. For those with positive results, primary care physicians can ensure timely follow-up, including Pap smears or colposcopies, to further evaluate cervical health.
Although home HPV testing offers many benefits, there are valid concerns about accuracy and follow-up care. Studies show that self-collected samples for HPV testing are highly accurate, with sensitivity and specificity comparable with clinician-collected samples, echoing the success of self-swabbing for other sexually transmitted infections.
It is crucial, however, that patients receive clear instructions on proper sample collection to maintain this accuracy. Follow-up care is another essential aspect of the screening process. While many HPV infections resolve on their own, high-risk strains require closer monitoring to prevent progression to cervical cancer. Primary care physicians must establish clear protocols for notifying patients of their results and ensuring appropriate follow-up appointments.
Additionally, there may be concerns about the cost and insurance coverage of home HPV tests. However, home testing could prove more cost-effective than multiple in-office visits, especially when factoring in travel expenses and missed work. Physicians should work to make home testing accessible to all patients, including those in low-income and rural communities.
Should these options become more widely available, it will be important to communicate that this does not fully eliminate the need for pelvic exams. As primary care physicians, we will still need to advise patients that they should bring up concerns of vaginal bleeding, vaginal discharge, and other symptoms. Pelvic exams will still be necessary for diagnosis when symptoms are present. Home HPV tests also will not replace in-office clinician collected exams for those who do not feel comfortable with self-collection.
Home and in-office self-collection for HPV testing are promising tools for improving cervical cancer screening rates and patient satisfaction. By offering a convenient, private, and accessible option, primary care physicians can help more patients stay on track with their preventive care and reduce their risk of cervical cancer. As this technology continues to evolve, embracing both in-office and home HPV testing will be essential to ensuring all patients benefit from these innovations.
Dr. Wheat is Vice Chair of Diversity, Equity, and Inclusion, Department of Family and Community Medicine and Associate Professor, Family and Community Medicine Feinberg School of Medicine, Northwestern University, Chicago. She serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
Daponte N et al. HPV-Based Self-Sampling in Cervical Cancer Screening: An Updated Review of the Current Evidence in the Literature. Cancers (Basel). 2023 Mar 8;15(6):1669.
Di Gennaro G et al. Does self-sampling for human papilloma virus testing have the potential to increase cervical cancer screening? An updated meta-analysis of observational studies and randomized clinical trials. Front Public Health. 2022 Dec 8;10:1003461.
US Preventive Services Task Force. Screening for Cervical Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320(7):674-686.
Human papillomavirus (HPV) is one of the most common sexually transmitted infections and persistent infection with high-risk strains is the leading cause of cervical cancer. Fortunately, vaccines are available to prevent many HPV-related diseases, but they haven’t fully eliminated the risks. Cervical cancer screening remains essential for early detection and prevention.
The US Preventive Services Task Force (USPSTF) currently recommends regular cervical cancer screenings for women aged 21-65. These screenings can include a Pap test every 3 years, a combination of HPV testing and Pap smear every 5 years, or high-risk HPV testing alone every 5 years, depending on age and individual risk factors.
Although these guidelines are currently under review, routine screenings have been instrumental in reducing cervical cancer rates. However, many patients still face barriers that prevent them from accessing these services. Common challenges include discomfort with pelvic exams, lack of time, and limited access to healthcare services. In recent years, advancements in home-based diagnostic testing have opened new avenues for preventative care.
Home HPV testing is one such advancement, offering an alternative to traditional in-office screening methods. While the US Food and Drug Administration (FDA) has not yet approved home HPV testing, self-collection in clinical settings is available and gaining traction. Primary care physicians can integrate this self-collection method into their practices, helping to close the screening gap, especially for underserved populations.
If approved, home HPV testing could be a game-changer for patients who have difficulty attending in-person visits. Geographical barriers, transportation issues, and personal discomfort with in-office exams can prevent patients from receiving the care they need. Home testing eliminates many of these hurdles, enabling patients to perform the test in the comfort of their own homes at a time that works for them. This flexibility is particularly beneficial for rural and underserved populations, where access to healthcare is limited.
Similarly, in-office self-collection offers a comfortable alternative for those who find traditional pelvic exams uncomfortable or distressing. Self-administered HPV tests allow patients to take control of their cervical cancer screening, fostering empowerment and personal responsibility for their health. By reducing the discomfort and inconvenience of traditional screening, self-collection can improve adherence to screening guidelines, leading to earlier detection and prevention of cervical cancer.
Primary care physicians may soon offer both in-office and at-home testing options, tailoring the approach to each patient’s unique needs. Virtual appointments provide an excellent opportunity to educate patients about the importance of cervical cancer screening and offer guidance on using home HPV testing kits. This personalized care ensures patients feel supported even without in-person visits. If home testing becomes FDA approved, patients could receive test kits by mail, perform the test, and send it back to the lab for analysis. For those with positive results, primary care physicians can ensure timely follow-up, including Pap smears or colposcopies, to further evaluate cervical health.
Although home HPV testing offers many benefits, there are valid concerns about accuracy and follow-up care. Studies show that self-collected samples for HPV testing are highly accurate, with sensitivity and specificity comparable with clinician-collected samples, echoing the success of self-swabbing for other sexually transmitted infections.
It is crucial, however, that patients receive clear instructions on proper sample collection to maintain this accuracy. Follow-up care is another essential aspect of the screening process. While many HPV infections resolve on their own, high-risk strains require closer monitoring to prevent progression to cervical cancer. Primary care physicians must establish clear protocols for notifying patients of their results and ensuring appropriate follow-up appointments.
Additionally, there may be concerns about the cost and insurance coverage of home HPV tests. However, home testing could prove more cost-effective than multiple in-office visits, especially when factoring in travel expenses and missed work. Physicians should work to make home testing accessible to all patients, including those in low-income and rural communities.
Should these options become more widely available, it will be important to communicate that this does not fully eliminate the need for pelvic exams. As primary care physicians, we will still need to advise patients that they should bring up concerns of vaginal bleeding, vaginal discharge, and other symptoms. Pelvic exams will still be necessary for diagnosis when symptoms are present. Home HPV tests also will not replace in-office clinician collected exams for those who do not feel comfortable with self-collection.
Home and in-office self-collection for HPV testing are promising tools for improving cervical cancer screening rates and patient satisfaction. By offering a convenient, private, and accessible option, primary care physicians can help more patients stay on track with their preventive care and reduce their risk of cervical cancer. As this technology continues to evolve, embracing both in-office and home HPV testing will be essential to ensuring all patients benefit from these innovations.
Dr. Wheat is Vice Chair of Diversity, Equity, and Inclusion, Department of Family and Community Medicine and Associate Professor, Family and Community Medicine Feinberg School of Medicine, Northwestern University, Chicago. She serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
Daponte N et al. HPV-Based Self-Sampling in Cervical Cancer Screening: An Updated Review of the Current Evidence in the Literature. Cancers (Basel). 2023 Mar 8;15(6):1669.
Di Gennaro G et al. Does self-sampling for human papilloma virus testing have the potential to increase cervical cancer screening? An updated meta-analysis of observational studies and randomized clinical trials. Front Public Health. 2022 Dec 8;10:1003461.
US Preventive Services Task Force. Screening for Cervical Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320(7):674-686.
Human papillomavirus (HPV) is one of the most common sexually transmitted infections and persistent infection with high-risk strains is the leading cause of cervical cancer. Fortunately, vaccines are available to prevent many HPV-related diseases, but they haven’t fully eliminated the risks. Cervical cancer screening remains essential for early detection and prevention.
The US Preventive Services Task Force (USPSTF) currently recommends regular cervical cancer screenings for women aged 21-65. These screenings can include a Pap test every 3 years, a combination of HPV testing and Pap smear every 5 years, or high-risk HPV testing alone every 5 years, depending on age and individual risk factors.
Although these guidelines are currently under review, routine screenings have been instrumental in reducing cervical cancer rates. However, many patients still face barriers that prevent them from accessing these services. Common challenges include discomfort with pelvic exams, lack of time, and limited access to healthcare services. In recent years, advancements in home-based diagnostic testing have opened new avenues for preventative care.
Home HPV testing is one such advancement, offering an alternative to traditional in-office screening methods. While the US Food and Drug Administration (FDA) has not yet approved home HPV testing, self-collection in clinical settings is available and gaining traction. Primary care physicians can integrate this self-collection method into their practices, helping to close the screening gap, especially for underserved populations.
If approved, home HPV testing could be a game-changer for patients who have difficulty attending in-person visits. Geographical barriers, transportation issues, and personal discomfort with in-office exams can prevent patients from receiving the care they need. Home testing eliminates many of these hurdles, enabling patients to perform the test in the comfort of their own homes at a time that works for them. This flexibility is particularly beneficial for rural and underserved populations, where access to healthcare is limited.
Similarly, in-office self-collection offers a comfortable alternative for those who find traditional pelvic exams uncomfortable or distressing. Self-administered HPV tests allow patients to take control of their cervical cancer screening, fostering empowerment and personal responsibility for their health. By reducing the discomfort and inconvenience of traditional screening, self-collection can improve adherence to screening guidelines, leading to earlier detection and prevention of cervical cancer.
Primary care physicians may soon offer both in-office and at-home testing options, tailoring the approach to each patient’s unique needs. Virtual appointments provide an excellent opportunity to educate patients about the importance of cervical cancer screening and offer guidance on using home HPV testing kits. This personalized care ensures patients feel supported even without in-person visits. If home testing becomes FDA approved, patients could receive test kits by mail, perform the test, and send it back to the lab for analysis. For those with positive results, primary care physicians can ensure timely follow-up, including Pap smears or colposcopies, to further evaluate cervical health.
Although home HPV testing offers many benefits, there are valid concerns about accuracy and follow-up care. Studies show that self-collected samples for HPV testing are highly accurate, with sensitivity and specificity comparable with clinician-collected samples, echoing the success of self-swabbing for other sexually transmitted infections.
It is crucial, however, that patients receive clear instructions on proper sample collection to maintain this accuracy. Follow-up care is another essential aspect of the screening process. While many HPV infections resolve on their own, high-risk strains require closer monitoring to prevent progression to cervical cancer. Primary care physicians must establish clear protocols for notifying patients of their results and ensuring appropriate follow-up appointments.
Additionally, there may be concerns about the cost and insurance coverage of home HPV tests. However, home testing could prove more cost-effective than multiple in-office visits, especially when factoring in travel expenses and missed work. Physicians should work to make home testing accessible to all patients, including those in low-income and rural communities.
Should these options become more widely available, it will be important to communicate that this does not fully eliminate the need for pelvic exams. As primary care physicians, we will still need to advise patients that they should bring up concerns of vaginal bleeding, vaginal discharge, and other symptoms. Pelvic exams will still be necessary for diagnosis when symptoms are present. Home HPV tests also will not replace in-office clinician collected exams for those who do not feel comfortable with self-collection.
Home and in-office self-collection for HPV testing are promising tools for improving cervical cancer screening rates and patient satisfaction. By offering a convenient, private, and accessible option, primary care physicians can help more patients stay on track with their preventive care and reduce their risk of cervical cancer. As this technology continues to evolve, embracing both in-office and home HPV testing will be essential to ensuring all patients benefit from these innovations.
Dr. Wheat is Vice Chair of Diversity, Equity, and Inclusion, Department of Family and Community Medicine and Associate Professor, Family and Community Medicine Feinberg School of Medicine, Northwestern University, Chicago. She serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
Daponte N et al. HPV-Based Self-Sampling in Cervical Cancer Screening: An Updated Review of the Current Evidence in the Literature. Cancers (Basel). 2023 Mar 8;15(6):1669.
Di Gennaro G et al. Does self-sampling for human papilloma virus testing have the potential to increase cervical cancer screening? An updated meta-analysis of observational studies and randomized clinical trials. Front Public Health. 2022 Dec 8;10:1003461.
US Preventive Services Task Force. Screening for Cervical Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;320(7):674-686.
Groups With Highest Unmet Need for PrEP Highlighted in Analysis
LOS ANGELES — Use of preexposure prophylaxis (PrEP) to prevent HIV is increasing overall, but both the rate of increase for starting PrEP and the rate of unmet need differ widely by demographic group, according to new data from a large study.
An analysis by Li Tao, MD, MS, PhD, director of real-world evidence at Gilead Sciences, and colleagues looked at statistical trends from 2019 to 2023 and found that Black, Hispanic, and Medicaid-insured populations continue to lack equitable access to PrEP.
Among the findings were that most new PrEP users were men with HIV risk factors who are commercially insured and live in predominantly non-Hispanic White areas (53% in 2019 and 43% in 2023). For comparison, men living in predominantly Black or Hispanic neighborhoods, or who are insured by Medicaid, saw lower proportions of PrEP use (16% in 2019 and 17% in 2023) despite higher annual increases in PrEP use (11% per year) and higher unmet needs.
Half a Million Real-World Participants
Tao presented her team’s findings at the Infectious Disease Week (IDWeek) 2024 Annual Meeting. The study included “more than half a million real-world PrEP users over the past 5 years,” she said.
The group with the lowest growth in initiation of PrEP in the study period (an annual percentage increase of 2%) and the lowest unmet need included men with HIV risk factors, who were using commercial insurance and living in White-dominant neighborhoods.
HIV risk factors included diagnosis of any sexually transmitted disease, contact with and exposure to communicable diseases, high-risk sexual behavior, contact with a hypodermic needle, long-term prophylaxis, HIV prevention counseling, and HIV screening.
Other men with HIV risk factors (those who were commercially insured, living in Black/Hispanic neighborhoods, or those on Medicaid across all neighborhoods) had a moderate increase in PrEP initiation (an annual percentage increase of 11%-16%) and higher unmet needs.
Researchers gathered data on PrEP prescriptions and new HIV diagnoses (from 2019 to 2023) through the IQVIA pharmacy claims database. PrEP-to-need ratio (PNR) is the number of individuals using PrEP in a year divided by new HIV diagnoses in the previous year. It was calculated for subgroups defined by five PNR-associated factors: Sex, insurance, recorded HIV risk factors (identified by diagnosis or procedure codes), “Ending the HIV Epidemic” jurisdictions, and neighborhood race/ethnicity mix.
Disparities Persist
While PrEP use improved across all the groups studied in the 5 years, “disparities still persist and the need remains very significant,” Tao said. “It’s very crucial for guiding the future HIV prevention options.”
“Long-acting PrEP options may help to address some social determinants structural factors in HIV acquisition,” she added.
What Programs Are Helping?
Some guidelines and programs are helping increase uptake, Tao said.
The United States Preventive Services Task Force (USPSTF) guidelines “reinforce more accessible PrEP programs to individuals like zero-cost sharing or same-day dispensing,” Tao said in a press briefing. “Those kinds of policies are really effective. We can see that after the implementation of the USPSTF guidelines, the copay sharing is really decreasing and is coinciding with the HIV rates declining.”
The Medicaid coverage expansion in 40 states “has been really effective” in PrEP uptake, she added.
Colleen Kelley, MD, MPH, with the Division of Infectious Diseases at the Rollins School of Public Health, Emory University, in Atlanta, who was not part of the research, said there has been a slow but improving uptake of PrEP across the board in the United States, “but the issue is that the uptake has been inequitable.”
Large Study With Recent Data
“This is an extremely large study with very recent data,” Kelley said. “Additionally, they were able to couple (the uptake) with unmet need. People who are at higher risk of acquiring HIV or who live in high-risk areas for HIV should have greater access to PrEP. They have a greater need for PrEP. What we really need to do from an equity perspective is match the PrEP use with the PrEP need and we have not been successful in doing that.”
Kelley added that the finding that the group that had the highest unmet need for PrEP in the study also had no recorded HIV risk factors. “It’s an interesting time to start thinking about beyond risk factor coverage for PrEP,” she said.
Another issue, Kelley said, is that “people are using (PrEP) but they’re also stopping it. People will need to take PrEP many years for protection, but about half discontinue in the first 6-12 months.
“We need to look at how people will persist on PrEP over the long term. That’s the next frontier,” she said. “We hope the long-acting injectables will help overcome some of the PrEP fatigue. But some may just tire of taking medication repeatedly for an infection they don’t have,” she said.
The study was funded by Gilead Sciences. Tao is employed by and is a shareholder of Gilead Sciences. All relevant financial disclosures have been mitigated, according to the paper. Kelley has research grants to her institution from Gilead, Moderna, Novavax, ViiV, and Humanigen.
A version of this article first appeared on Medscape.com.
LOS ANGELES — Use of preexposure prophylaxis (PrEP) to prevent HIV is increasing overall, but both the rate of increase for starting PrEP and the rate of unmet need differ widely by demographic group, according to new data from a large study.
An analysis by Li Tao, MD, MS, PhD, director of real-world evidence at Gilead Sciences, and colleagues looked at statistical trends from 2019 to 2023 and found that Black, Hispanic, and Medicaid-insured populations continue to lack equitable access to PrEP.
Among the findings were that most new PrEP users were men with HIV risk factors who are commercially insured and live in predominantly non-Hispanic White areas (53% in 2019 and 43% in 2023). For comparison, men living in predominantly Black or Hispanic neighborhoods, or who are insured by Medicaid, saw lower proportions of PrEP use (16% in 2019 and 17% in 2023) despite higher annual increases in PrEP use (11% per year) and higher unmet needs.
Half a Million Real-World Participants
Tao presented her team’s findings at the Infectious Disease Week (IDWeek) 2024 Annual Meeting. The study included “more than half a million real-world PrEP users over the past 5 years,” she said.
The group with the lowest growth in initiation of PrEP in the study period (an annual percentage increase of 2%) and the lowest unmet need included men with HIV risk factors, who were using commercial insurance and living in White-dominant neighborhoods.
HIV risk factors included diagnosis of any sexually transmitted disease, contact with and exposure to communicable diseases, high-risk sexual behavior, contact with a hypodermic needle, long-term prophylaxis, HIV prevention counseling, and HIV screening.
Other men with HIV risk factors (those who were commercially insured, living in Black/Hispanic neighborhoods, or those on Medicaid across all neighborhoods) had a moderate increase in PrEP initiation (an annual percentage increase of 11%-16%) and higher unmet needs.
Researchers gathered data on PrEP prescriptions and new HIV diagnoses (from 2019 to 2023) through the IQVIA pharmacy claims database. PrEP-to-need ratio (PNR) is the number of individuals using PrEP in a year divided by new HIV diagnoses in the previous year. It was calculated for subgroups defined by five PNR-associated factors: Sex, insurance, recorded HIV risk factors (identified by diagnosis or procedure codes), “Ending the HIV Epidemic” jurisdictions, and neighborhood race/ethnicity mix.
Disparities Persist
While PrEP use improved across all the groups studied in the 5 years, “disparities still persist and the need remains very significant,” Tao said. “It’s very crucial for guiding the future HIV prevention options.”
“Long-acting PrEP options may help to address some social determinants structural factors in HIV acquisition,” she added.
What Programs Are Helping?
Some guidelines and programs are helping increase uptake, Tao said.
The United States Preventive Services Task Force (USPSTF) guidelines “reinforce more accessible PrEP programs to individuals like zero-cost sharing or same-day dispensing,” Tao said in a press briefing. “Those kinds of policies are really effective. We can see that after the implementation of the USPSTF guidelines, the copay sharing is really decreasing and is coinciding with the HIV rates declining.”
The Medicaid coverage expansion in 40 states “has been really effective” in PrEP uptake, she added.
Colleen Kelley, MD, MPH, with the Division of Infectious Diseases at the Rollins School of Public Health, Emory University, in Atlanta, who was not part of the research, said there has been a slow but improving uptake of PrEP across the board in the United States, “but the issue is that the uptake has been inequitable.”
Large Study With Recent Data
“This is an extremely large study with very recent data,” Kelley said. “Additionally, they were able to couple (the uptake) with unmet need. People who are at higher risk of acquiring HIV or who live in high-risk areas for HIV should have greater access to PrEP. They have a greater need for PrEP. What we really need to do from an equity perspective is match the PrEP use with the PrEP need and we have not been successful in doing that.”
Kelley added that the finding that the group that had the highest unmet need for PrEP in the study also had no recorded HIV risk factors. “It’s an interesting time to start thinking about beyond risk factor coverage for PrEP,” she said.
Another issue, Kelley said, is that “people are using (PrEP) but they’re also stopping it. People will need to take PrEP many years for protection, but about half discontinue in the first 6-12 months.
“We need to look at how people will persist on PrEP over the long term. That’s the next frontier,” she said. “We hope the long-acting injectables will help overcome some of the PrEP fatigue. But some may just tire of taking medication repeatedly for an infection they don’t have,” she said.
The study was funded by Gilead Sciences. Tao is employed by and is a shareholder of Gilead Sciences. All relevant financial disclosures have been mitigated, according to the paper. Kelley has research grants to her institution from Gilead, Moderna, Novavax, ViiV, and Humanigen.
A version of this article first appeared on Medscape.com.
LOS ANGELES — Use of preexposure prophylaxis (PrEP) to prevent HIV is increasing overall, but both the rate of increase for starting PrEP and the rate of unmet need differ widely by demographic group, according to new data from a large study.
An analysis by Li Tao, MD, MS, PhD, director of real-world evidence at Gilead Sciences, and colleagues looked at statistical trends from 2019 to 2023 and found that Black, Hispanic, and Medicaid-insured populations continue to lack equitable access to PrEP.
Among the findings were that most new PrEP users were men with HIV risk factors who are commercially insured and live in predominantly non-Hispanic White areas (53% in 2019 and 43% in 2023). For comparison, men living in predominantly Black or Hispanic neighborhoods, or who are insured by Medicaid, saw lower proportions of PrEP use (16% in 2019 and 17% in 2023) despite higher annual increases in PrEP use (11% per year) and higher unmet needs.
Half a Million Real-World Participants
Tao presented her team’s findings at the Infectious Disease Week (IDWeek) 2024 Annual Meeting. The study included “more than half a million real-world PrEP users over the past 5 years,” she said.
The group with the lowest growth in initiation of PrEP in the study period (an annual percentage increase of 2%) and the lowest unmet need included men with HIV risk factors, who were using commercial insurance and living in White-dominant neighborhoods.
HIV risk factors included diagnosis of any sexually transmitted disease, contact with and exposure to communicable diseases, high-risk sexual behavior, contact with a hypodermic needle, long-term prophylaxis, HIV prevention counseling, and HIV screening.
Other men with HIV risk factors (those who were commercially insured, living in Black/Hispanic neighborhoods, or those on Medicaid across all neighborhoods) had a moderate increase in PrEP initiation (an annual percentage increase of 11%-16%) and higher unmet needs.
Researchers gathered data on PrEP prescriptions and new HIV diagnoses (from 2019 to 2023) through the IQVIA pharmacy claims database. PrEP-to-need ratio (PNR) is the number of individuals using PrEP in a year divided by new HIV diagnoses in the previous year. It was calculated for subgroups defined by five PNR-associated factors: Sex, insurance, recorded HIV risk factors (identified by diagnosis or procedure codes), “Ending the HIV Epidemic” jurisdictions, and neighborhood race/ethnicity mix.
Disparities Persist
While PrEP use improved across all the groups studied in the 5 years, “disparities still persist and the need remains very significant,” Tao said. “It’s very crucial for guiding the future HIV prevention options.”
“Long-acting PrEP options may help to address some social determinants structural factors in HIV acquisition,” she added.
What Programs Are Helping?
Some guidelines and programs are helping increase uptake, Tao said.
The United States Preventive Services Task Force (USPSTF) guidelines “reinforce more accessible PrEP programs to individuals like zero-cost sharing or same-day dispensing,” Tao said in a press briefing. “Those kinds of policies are really effective. We can see that after the implementation of the USPSTF guidelines, the copay sharing is really decreasing and is coinciding with the HIV rates declining.”
The Medicaid coverage expansion in 40 states “has been really effective” in PrEP uptake, she added.
Colleen Kelley, MD, MPH, with the Division of Infectious Diseases at the Rollins School of Public Health, Emory University, in Atlanta, who was not part of the research, said there has been a slow but improving uptake of PrEP across the board in the United States, “but the issue is that the uptake has been inequitable.”
Large Study With Recent Data
“This is an extremely large study with very recent data,” Kelley said. “Additionally, they were able to couple (the uptake) with unmet need. People who are at higher risk of acquiring HIV or who live in high-risk areas for HIV should have greater access to PrEP. They have a greater need for PrEP. What we really need to do from an equity perspective is match the PrEP use with the PrEP need and we have not been successful in doing that.”
Kelley added that the finding that the group that had the highest unmet need for PrEP in the study also had no recorded HIV risk factors. “It’s an interesting time to start thinking about beyond risk factor coverage for PrEP,” she said.
Another issue, Kelley said, is that “people are using (PrEP) but they’re also stopping it. People will need to take PrEP many years for protection, but about half discontinue in the first 6-12 months.
“We need to look at how people will persist on PrEP over the long term. That’s the next frontier,” she said. “We hope the long-acting injectables will help overcome some of the PrEP fatigue. But some may just tire of taking medication repeatedly for an infection they don’t have,” she said.
The study was funded by Gilead Sciences. Tao is employed by and is a shareholder of Gilead Sciences. All relevant financial disclosures have been mitigated, according to the paper. Kelley has research grants to her institution from Gilead, Moderna, Novavax, ViiV, and Humanigen.
A version of this article first appeared on Medscape.com.
FROM IDWEEK 2024
How Effective Is the High-Dose Flu Vaccine in Older Adults?
How can the immunogenicity and effectiveness of flu vaccines be improved in older adults? Several strategies are available, one being the addition of an adjuvant. For example, the MF59-adjuvanted vaccine has shown superior immunogenicity. However, “we do not have data from controlled and randomized clinical trials showing superior clinical effectiveness versus the standard dose,” Professor Odile Launay, an infectious disease specialist at Cochin Hospital in Paris, France, noted during a press conference. Another option is to increase the antigen dose in the vaccine, creating a high-dose (HD) flu vaccine.
Why is there a need for an HD vaccine? “The elderly population bears the greatest burden from the flu,” explained Launay. “This is due to three factors: An aging immune system, a higher number of comorbidities, and increased frailty.” Standard-dose flu vaccines are seen as offering suboptimal protection for those older than 65 years, which led to the development of a quadrivalent vaccine with four times the antigen dose of standard flu vaccines. This HD vaccine was introduced in France during the 2021/2022 flu season. A real-world cohort study has since been conducted to evaluate its effectiveness in the target population — those aged 65 years or older. The results were recently published in Clinical Microbiology and Infection.
Cohort Study
The study included 405,385 noninstitutionalized people aged 65 years or older matched with 1,621,540 individuals in a 1:4 ratio. The first group received the HD vaccine, while the second group received the standard-dose vaccine. Both the groups had an average age of 77 years, with 56% women, and 51% vaccinated in pharmacies. The majority had been previously vaccinated against flu (91%), and 97% had completed a full COVID-19 vaccination schedule. More than half had at least one chronic illness.
Hospitalization rates for flu — the study’s primary outcome — were 69.5 vs 90.5 per 100,000 person-years in the HD vs standard-dose group. This represented a 23.3% reduction (95% CI, 8.4-35.8; P = .003).
Strengths and Limitations
Among the strengths of the study, Launay highlighted the large number of vaccinated participants older than 65 years — more than 7 million — and the widespread use of polymerase chain reaction flu tests in cases of hospitalization for respiratory infections, which improved flu coding in the database used. Additionally, the results were consistent with those of previous studies.
However, limitations included the retrospective design, which did not randomize participants and introduced potential bias. For example, the HD vaccine may have been prioritized for the oldest people or those with multiple comorbidities. Additionally, the 2021/2022 flu season was atypical, with the simultaneous circulation of the flu virus and SARS-CoV-2, as noted by Launay.
Conclusion
In conclusion, this first evaluation of the HD flu vaccine’s effectiveness in France showed a 25% reduction in hospitalizations, consistent with existing data covering 12 flu seasons. The vaccine has been available for a longer period in the United States and Northern Europe.
“The latest unpublished data from the 2022/23 season show a 27% reduction in hospitalizations with the HD vaccine in people over 65,” added Launay.
Note: Due to a pricing disagreement with the French government, Sanofi’s HD flu vaccine Efluelda, intended for people older than 65 years, will not be available this year. (See: Withdrawal of the Efluelda Influenza Vaccine: The Academy of Medicine Reacts). However, the company has submitted a dossier for a trivalent form for a return in the 2025/2026 season and is working on developing mRNA vaccines. Additionally, a combined flu/COVID-19 vaccine is currently in development.
The study was funded by Sanofi. Several authors are Sanofi employees. Odile Launay reported conflicts of interest with Sanofi, MSD, Pfizer, GSK, and Moderna.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
How can the immunogenicity and effectiveness of flu vaccines be improved in older adults? Several strategies are available, one being the addition of an adjuvant. For example, the MF59-adjuvanted vaccine has shown superior immunogenicity. However, “we do not have data from controlled and randomized clinical trials showing superior clinical effectiveness versus the standard dose,” Professor Odile Launay, an infectious disease specialist at Cochin Hospital in Paris, France, noted during a press conference. Another option is to increase the antigen dose in the vaccine, creating a high-dose (HD) flu vaccine.
Why is there a need for an HD vaccine? “The elderly population bears the greatest burden from the flu,” explained Launay. “This is due to three factors: An aging immune system, a higher number of comorbidities, and increased frailty.” Standard-dose flu vaccines are seen as offering suboptimal protection for those older than 65 years, which led to the development of a quadrivalent vaccine with four times the antigen dose of standard flu vaccines. This HD vaccine was introduced in France during the 2021/2022 flu season. A real-world cohort study has since been conducted to evaluate its effectiveness in the target population — those aged 65 years or older. The results were recently published in Clinical Microbiology and Infection.
Cohort Study
The study included 405,385 noninstitutionalized people aged 65 years or older matched with 1,621,540 individuals in a 1:4 ratio. The first group received the HD vaccine, while the second group received the standard-dose vaccine. Both the groups had an average age of 77 years, with 56% women, and 51% vaccinated in pharmacies. The majority had been previously vaccinated against flu (91%), and 97% had completed a full COVID-19 vaccination schedule. More than half had at least one chronic illness.
Hospitalization rates for flu — the study’s primary outcome — were 69.5 vs 90.5 per 100,000 person-years in the HD vs standard-dose group. This represented a 23.3% reduction (95% CI, 8.4-35.8; P = .003).
Strengths and Limitations
Among the strengths of the study, Launay highlighted the large number of vaccinated participants older than 65 years — more than 7 million — and the widespread use of polymerase chain reaction flu tests in cases of hospitalization for respiratory infections, which improved flu coding in the database used. Additionally, the results were consistent with those of previous studies.
However, limitations included the retrospective design, which did not randomize participants and introduced potential bias. For example, the HD vaccine may have been prioritized for the oldest people or those with multiple comorbidities. Additionally, the 2021/2022 flu season was atypical, with the simultaneous circulation of the flu virus and SARS-CoV-2, as noted by Launay.
Conclusion
In conclusion, this first evaluation of the HD flu vaccine’s effectiveness in France showed a 25% reduction in hospitalizations, consistent with existing data covering 12 flu seasons. The vaccine has been available for a longer period in the United States and Northern Europe.
“The latest unpublished data from the 2022/23 season show a 27% reduction in hospitalizations with the HD vaccine in people over 65,” added Launay.
Note: Due to a pricing disagreement with the French government, Sanofi’s HD flu vaccine Efluelda, intended for people older than 65 years, will not be available this year. (See: Withdrawal of the Efluelda Influenza Vaccine: The Academy of Medicine Reacts). However, the company has submitted a dossier for a trivalent form for a return in the 2025/2026 season and is working on developing mRNA vaccines. Additionally, a combined flu/COVID-19 vaccine is currently in development.
The study was funded by Sanofi. Several authors are Sanofi employees. Odile Launay reported conflicts of interest with Sanofi, MSD, Pfizer, GSK, and Moderna.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
How can the immunogenicity and effectiveness of flu vaccines be improved in older adults? Several strategies are available, one being the addition of an adjuvant. For example, the MF59-adjuvanted vaccine has shown superior immunogenicity. However, “we do not have data from controlled and randomized clinical trials showing superior clinical effectiveness versus the standard dose,” Professor Odile Launay, an infectious disease specialist at Cochin Hospital in Paris, France, noted during a press conference. Another option is to increase the antigen dose in the vaccine, creating a high-dose (HD) flu vaccine.
Why is there a need for an HD vaccine? “The elderly population bears the greatest burden from the flu,” explained Launay. “This is due to three factors: An aging immune system, a higher number of comorbidities, and increased frailty.” Standard-dose flu vaccines are seen as offering suboptimal protection for those older than 65 years, which led to the development of a quadrivalent vaccine with four times the antigen dose of standard flu vaccines. This HD vaccine was introduced in France during the 2021/2022 flu season. A real-world cohort study has since been conducted to evaluate its effectiveness in the target population — those aged 65 years or older. The results were recently published in Clinical Microbiology and Infection.
Cohort Study
The study included 405,385 noninstitutionalized people aged 65 years or older matched with 1,621,540 individuals in a 1:4 ratio. The first group received the HD vaccine, while the second group received the standard-dose vaccine. Both the groups had an average age of 77 years, with 56% women, and 51% vaccinated in pharmacies. The majority had been previously vaccinated against flu (91%), and 97% had completed a full COVID-19 vaccination schedule. More than half had at least one chronic illness.
Hospitalization rates for flu — the study’s primary outcome — were 69.5 vs 90.5 per 100,000 person-years in the HD vs standard-dose group. This represented a 23.3% reduction (95% CI, 8.4-35.8; P = .003).
Strengths and Limitations
Among the strengths of the study, Launay highlighted the large number of vaccinated participants older than 65 years — more than 7 million — and the widespread use of polymerase chain reaction flu tests in cases of hospitalization for respiratory infections, which improved flu coding in the database used. Additionally, the results were consistent with those of previous studies.
However, limitations included the retrospective design, which did not randomize participants and introduced potential bias. For example, the HD vaccine may have been prioritized for the oldest people or those with multiple comorbidities. Additionally, the 2021/2022 flu season was atypical, with the simultaneous circulation of the flu virus and SARS-CoV-2, as noted by Launay.
Conclusion
In conclusion, this first evaluation of the HD flu vaccine’s effectiveness in France showed a 25% reduction in hospitalizations, consistent with existing data covering 12 flu seasons. The vaccine has been available for a longer period in the United States and Northern Europe.
“The latest unpublished data from the 2022/23 season show a 27% reduction in hospitalizations with the HD vaccine in people over 65,” added Launay.
Note: Due to a pricing disagreement with the French government, Sanofi’s HD flu vaccine Efluelda, intended for people older than 65 years, will not be available this year. (See: Withdrawal of the Efluelda Influenza Vaccine: The Academy of Medicine Reacts). However, the company has submitted a dossier for a trivalent form for a return in the 2025/2026 season and is working on developing mRNA vaccines. Additionally, a combined flu/COVID-19 vaccine is currently in development.
The study was funded by Sanofi. Several authors are Sanofi employees. Odile Launay reported conflicts of interest with Sanofi, MSD, Pfizer, GSK, and Moderna.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
One-Dose HPV Vaccine Program Would Be Efficient in Canada
In Canada, switching to a one-dose, gender-neutral vaccination program for human papillomavirus (HPV) could use vaccine doses more efficiently and prevent a similar number of cervical cancer cases, compared with a two-dose program, according to a new modeling analysis.
If vaccine protection remains high during the ages of peak sexual activity, all one-dose vaccination options are projected to be “substantially more efficient” than two-dose programs, even in the most pessimistic scenarios, the study authors wrote.
In addition, the scenarios projected the elimination of cervical cancer in Canada between 2032 and 2040. HPV can also lead to oral, throat, and penile cancers, and most are preventable through vaccination.
“The COVID-19 pandemic has impacted HPV vaccination in Canada, particularly among vulnerable population subgroups,” said study author Chantal Sauvageau, MD, a consultant in infectious diseases at the National Institute of Public Health of Quebec and associate professor of social and preventive medicine at the University of Laval, Quebec City, Canada.
Switching to one-dose vaccination would offer potential economic savings and programmatic flexibility, she added. The change also could enable investments aimed at increasing vaccination rates in regions where coverage is suboptimal, as well as in subgroups with a high HPV burden. Such initiatives could mitigate the pandemic’s impact on health programs and reduce inequalities.
The study was published online in CMAJ.
Vaccination Program Changes
Globally, countries have been investigating whether to shift from a two-dose to a one-dose HPV vaccine strategy since the World Health Organization’s Strategic Advisory Group of Experts on Immunization issued a single-dose recommendation in 2022.
In July, Canada’s National Advisory Committee on Immunization (NACI) updated its guidelines to recommend the single-dose approach for ages 9-20 years. The change aligns Canada with 35 other countries, including Australia and the United Kingdom. Canada›s vaccine advisory group still recommends two doses for ages 21-26 years and three doses for patients who are immunocompromised or have HIV.
To help inform new NACI policies, Sauvageau and colleagues modeled several one-dose and two-dose strategies using HPV-ADVISE, an individual-based transmission-dynamic model of HPV infections and diseases. They looked at vaccination programs in Quebec, which has a high HPV vaccine coverage rate of around 85%, and Ontario, which has lower coverage of around 65%.
For one-dose programs, the researchers analyzed noninferior (98% efficacy) and pessimistic (90% efficacy) scenarios and different average vaccine duration periods, including lifelong, 30-year, and 25-year coverage. They compared the scenarios with a two-dose program with 98% efficacy and lifelong duration, estimating the relative reduction in HPV-16 infection and cervical cancer incidence and the number of doses needed to prevent one cervical cancer case.
Overall, the model projected that gender-neutral HPV vaccine programs with either two doses or a noninferior one dose would nearly eliminate HPV-16 infection by 2040-2045 in Quebec and reduce infection by more than 90% in Ontario. Under a one-dose strategy with 90% vaccine efficacy, rebounds in HPV-16 infection would start more than 25-30 years after a switch to a lower-dose strategy, thus providing time for officials to detect any signs of waning efficacy and change policies, if needed, the authors wrote.
In addition, the model projected that a noninferior one-dose, gender-neutral HPV vaccination program would avert a similar number of cervical cancer cases, compared with a two-dose program. The reduction would be about 60% in Quebec and 55% in Ontario, compared with no vaccination. Under the most pessimistic scenario with 25-year vaccine duration, a one-dose program would be slightly less effective in averting cancer: about 3% lower than a two-dose program over 100 years.
All one-dose scenarios were projected to lead to the elimination of cervical cancer in 8-16 years — at fewer than four cervical cancer cases per 100,000 female-years.
One-dose programs would also lead to more efficient use of vaccine doses, with about 800-1000 doses needed to prevent one cervical cancer case in a one-dose program and more than 10,000 incremental doses needed to prevent one additional cervical cancer case in a two-dose program.
What Next?
In Canada, the HPV vaccine is authorized for patients aged 9-45 years. Current immunization coverage among adolescents and young adults varies across provinces and falls below the national target of 90%. In its July 2024 update, NACI estimated that 76% of 14-year-olds of both genders received at least one vaccine dose and that 67% received two doses in 2023. Vaccine uptake was slightly higher among girls than boys.
To boost the coverage rate, shifting to a one-dose schedule could appeal to young people, as well as maintain vaccination efficacy.
“When you look at the studies that have been published worldwide, the effectiveness of one dose of the HPV vaccine is actually quite high,” said Caroline Quach-Thanh, MD, professor of microbiology, infectious diseases, immunology, and pediatrics at the University of Montreal, Quebec, Canada.
Quach-Thanh, who wasn’t involved with this study, previously served as NACI chair and now serves as chair of the Quebec Immunization Committee.
“In terms of prevention of HPV infections that may lead to cancer, whether you give one dose or two doses basically gives you the same amount of protection,” she said.
However, not all physicians agree about the switch in vaccination approaches. In early October, the Federation of Medical Women of Canada released a report with 12 recommendations to increase HPV vaccination rates, including a call for healthcare providers to continue with multidose immunization schedules for now.
“Vaccination is the most powerful action we can take in preventing HPV-related cancers. Canada is falling behind, but we can get back on track if we act quickly,” said Vivien Brown, MD, chair of the group’s HPV Immunization Task Force, chair and cofounder of HPV Prevention Week in Canada, and a past president of the federation.
After the NACI update in July, the task force evaluated the risks and benefits of a single-dose vaccine regimen, she said. They concluded that a multidose schedule should continue at this time because of its proven effectiveness.
“Until more research on the efficacy of a single-dose schedule becomes available, healthcare providers and public health agencies should continue to offer patients a multidose schedule,” said Brown. “This is the only way to ensure individuals are protected against HPV infection and cancer over the long term.”
The study was supported by the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Bill & Melinda Gates Foundation, and Canadian Immunization Research Network. Sauvageau, Quach-Thanh, and Brown declared no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
In Canada, switching to a one-dose, gender-neutral vaccination program for human papillomavirus (HPV) could use vaccine doses more efficiently and prevent a similar number of cervical cancer cases, compared with a two-dose program, according to a new modeling analysis.
If vaccine protection remains high during the ages of peak sexual activity, all one-dose vaccination options are projected to be “substantially more efficient” than two-dose programs, even in the most pessimistic scenarios, the study authors wrote.
In addition, the scenarios projected the elimination of cervical cancer in Canada between 2032 and 2040. HPV can also lead to oral, throat, and penile cancers, and most are preventable through vaccination.
“The COVID-19 pandemic has impacted HPV vaccination in Canada, particularly among vulnerable population subgroups,” said study author Chantal Sauvageau, MD, a consultant in infectious diseases at the National Institute of Public Health of Quebec and associate professor of social and preventive medicine at the University of Laval, Quebec City, Canada.
Switching to one-dose vaccination would offer potential economic savings and programmatic flexibility, she added. The change also could enable investments aimed at increasing vaccination rates in regions where coverage is suboptimal, as well as in subgroups with a high HPV burden. Such initiatives could mitigate the pandemic’s impact on health programs and reduce inequalities.
The study was published online in CMAJ.
Vaccination Program Changes
Globally, countries have been investigating whether to shift from a two-dose to a one-dose HPV vaccine strategy since the World Health Organization’s Strategic Advisory Group of Experts on Immunization issued a single-dose recommendation in 2022.
In July, Canada’s National Advisory Committee on Immunization (NACI) updated its guidelines to recommend the single-dose approach for ages 9-20 years. The change aligns Canada with 35 other countries, including Australia and the United Kingdom. Canada›s vaccine advisory group still recommends two doses for ages 21-26 years and three doses for patients who are immunocompromised or have HIV.
To help inform new NACI policies, Sauvageau and colleagues modeled several one-dose and two-dose strategies using HPV-ADVISE, an individual-based transmission-dynamic model of HPV infections and diseases. They looked at vaccination programs in Quebec, which has a high HPV vaccine coverage rate of around 85%, and Ontario, which has lower coverage of around 65%.
For one-dose programs, the researchers analyzed noninferior (98% efficacy) and pessimistic (90% efficacy) scenarios and different average vaccine duration periods, including lifelong, 30-year, and 25-year coverage. They compared the scenarios with a two-dose program with 98% efficacy and lifelong duration, estimating the relative reduction in HPV-16 infection and cervical cancer incidence and the number of doses needed to prevent one cervical cancer case.
Overall, the model projected that gender-neutral HPV vaccine programs with either two doses or a noninferior one dose would nearly eliminate HPV-16 infection by 2040-2045 in Quebec and reduce infection by more than 90% in Ontario. Under a one-dose strategy with 90% vaccine efficacy, rebounds in HPV-16 infection would start more than 25-30 years after a switch to a lower-dose strategy, thus providing time for officials to detect any signs of waning efficacy and change policies, if needed, the authors wrote.
In addition, the model projected that a noninferior one-dose, gender-neutral HPV vaccination program would avert a similar number of cervical cancer cases, compared with a two-dose program. The reduction would be about 60% in Quebec and 55% in Ontario, compared with no vaccination. Under the most pessimistic scenario with 25-year vaccine duration, a one-dose program would be slightly less effective in averting cancer: about 3% lower than a two-dose program over 100 years.
All one-dose scenarios were projected to lead to the elimination of cervical cancer in 8-16 years — at fewer than four cervical cancer cases per 100,000 female-years.
One-dose programs would also lead to more efficient use of vaccine doses, with about 800-1000 doses needed to prevent one cervical cancer case in a one-dose program and more than 10,000 incremental doses needed to prevent one additional cervical cancer case in a two-dose program.
What Next?
In Canada, the HPV vaccine is authorized for patients aged 9-45 years. Current immunization coverage among adolescents and young adults varies across provinces and falls below the national target of 90%. In its July 2024 update, NACI estimated that 76% of 14-year-olds of both genders received at least one vaccine dose and that 67% received two doses in 2023. Vaccine uptake was slightly higher among girls than boys.
To boost the coverage rate, shifting to a one-dose schedule could appeal to young people, as well as maintain vaccination efficacy.
“When you look at the studies that have been published worldwide, the effectiveness of one dose of the HPV vaccine is actually quite high,” said Caroline Quach-Thanh, MD, professor of microbiology, infectious diseases, immunology, and pediatrics at the University of Montreal, Quebec, Canada.
Quach-Thanh, who wasn’t involved with this study, previously served as NACI chair and now serves as chair of the Quebec Immunization Committee.
“In terms of prevention of HPV infections that may lead to cancer, whether you give one dose or two doses basically gives you the same amount of protection,” she said.
However, not all physicians agree about the switch in vaccination approaches. In early October, the Federation of Medical Women of Canada released a report with 12 recommendations to increase HPV vaccination rates, including a call for healthcare providers to continue with multidose immunization schedules for now.
“Vaccination is the most powerful action we can take in preventing HPV-related cancers. Canada is falling behind, but we can get back on track if we act quickly,” said Vivien Brown, MD, chair of the group’s HPV Immunization Task Force, chair and cofounder of HPV Prevention Week in Canada, and a past president of the federation.
After the NACI update in July, the task force evaluated the risks and benefits of a single-dose vaccine regimen, she said. They concluded that a multidose schedule should continue at this time because of its proven effectiveness.
“Until more research on the efficacy of a single-dose schedule becomes available, healthcare providers and public health agencies should continue to offer patients a multidose schedule,” said Brown. “This is the only way to ensure individuals are protected against HPV infection and cancer over the long term.”
The study was supported by the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Bill & Melinda Gates Foundation, and Canadian Immunization Research Network. Sauvageau, Quach-Thanh, and Brown declared no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
In Canada, switching to a one-dose, gender-neutral vaccination program for human papillomavirus (HPV) could use vaccine doses more efficiently and prevent a similar number of cervical cancer cases, compared with a two-dose program, according to a new modeling analysis.
If vaccine protection remains high during the ages of peak sexual activity, all one-dose vaccination options are projected to be “substantially more efficient” than two-dose programs, even in the most pessimistic scenarios, the study authors wrote.
In addition, the scenarios projected the elimination of cervical cancer in Canada between 2032 and 2040. HPV can also lead to oral, throat, and penile cancers, and most are preventable through vaccination.
“The COVID-19 pandemic has impacted HPV vaccination in Canada, particularly among vulnerable population subgroups,” said study author Chantal Sauvageau, MD, a consultant in infectious diseases at the National Institute of Public Health of Quebec and associate professor of social and preventive medicine at the University of Laval, Quebec City, Canada.
Switching to one-dose vaccination would offer potential economic savings and programmatic flexibility, she added. The change also could enable investments aimed at increasing vaccination rates in regions where coverage is suboptimal, as well as in subgroups with a high HPV burden. Such initiatives could mitigate the pandemic’s impact on health programs and reduce inequalities.
The study was published online in CMAJ.
Vaccination Program Changes
Globally, countries have been investigating whether to shift from a two-dose to a one-dose HPV vaccine strategy since the World Health Organization’s Strategic Advisory Group of Experts on Immunization issued a single-dose recommendation in 2022.
In July, Canada’s National Advisory Committee on Immunization (NACI) updated its guidelines to recommend the single-dose approach for ages 9-20 years. The change aligns Canada with 35 other countries, including Australia and the United Kingdom. Canada›s vaccine advisory group still recommends two doses for ages 21-26 years and three doses for patients who are immunocompromised or have HIV.
To help inform new NACI policies, Sauvageau and colleagues modeled several one-dose and two-dose strategies using HPV-ADVISE, an individual-based transmission-dynamic model of HPV infections and diseases. They looked at vaccination programs in Quebec, which has a high HPV vaccine coverage rate of around 85%, and Ontario, which has lower coverage of around 65%.
For one-dose programs, the researchers analyzed noninferior (98% efficacy) and pessimistic (90% efficacy) scenarios and different average vaccine duration periods, including lifelong, 30-year, and 25-year coverage. They compared the scenarios with a two-dose program with 98% efficacy and lifelong duration, estimating the relative reduction in HPV-16 infection and cervical cancer incidence and the number of doses needed to prevent one cervical cancer case.
Overall, the model projected that gender-neutral HPV vaccine programs with either two doses or a noninferior one dose would nearly eliminate HPV-16 infection by 2040-2045 in Quebec and reduce infection by more than 90% in Ontario. Under a one-dose strategy with 90% vaccine efficacy, rebounds in HPV-16 infection would start more than 25-30 years after a switch to a lower-dose strategy, thus providing time for officials to detect any signs of waning efficacy and change policies, if needed, the authors wrote.
In addition, the model projected that a noninferior one-dose, gender-neutral HPV vaccination program would avert a similar number of cervical cancer cases, compared with a two-dose program. The reduction would be about 60% in Quebec and 55% in Ontario, compared with no vaccination. Under the most pessimistic scenario with 25-year vaccine duration, a one-dose program would be slightly less effective in averting cancer: about 3% lower than a two-dose program over 100 years.
All one-dose scenarios were projected to lead to the elimination of cervical cancer in 8-16 years — at fewer than four cervical cancer cases per 100,000 female-years.
One-dose programs would also lead to more efficient use of vaccine doses, with about 800-1000 doses needed to prevent one cervical cancer case in a one-dose program and more than 10,000 incremental doses needed to prevent one additional cervical cancer case in a two-dose program.
What Next?
In Canada, the HPV vaccine is authorized for patients aged 9-45 years. Current immunization coverage among adolescents and young adults varies across provinces and falls below the national target of 90%. In its July 2024 update, NACI estimated that 76% of 14-year-olds of both genders received at least one vaccine dose and that 67% received two doses in 2023. Vaccine uptake was slightly higher among girls than boys.
To boost the coverage rate, shifting to a one-dose schedule could appeal to young people, as well as maintain vaccination efficacy.
“When you look at the studies that have been published worldwide, the effectiveness of one dose of the HPV vaccine is actually quite high,” said Caroline Quach-Thanh, MD, professor of microbiology, infectious diseases, immunology, and pediatrics at the University of Montreal, Quebec, Canada.
Quach-Thanh, who wasn’t involved with this study, previously served as NACI chair and now serves as chair of the Quebec Immunization Committee.
“In terms of prevention of HPV infections that may lead to cancer, whether you give one dose or two doses basically gives you the same amount of protection,” she said.
However, not all physicians agree about the switch in vaccination approaches. In early October, the Federation of Medical Women of Canada released a report with 12 recommendations to increase HPV vaccination rates, including a call for healthcare providers to continue with multidose immunization schedules for now.
“Vaccination is the most powerful action we can take in preventing HPV-related cancers. Canada is falling behind, but we can get back on track if we act quickly,” said Vivien Brown, MD, chair of the group’s HPV Immunization Task Force, chair and cofounder of HPV Prevention Week in Canada, and a past president of the federation.
After the NACI update in July, the task force evaluated the risks and benefits of a single-dose vaccine regimen, she said. They concluded that a multidose schedule should continue at this time because of its proven effectiveness.
“Until more research on the efficacy of a single-dose schedule becomes available, healthcare providers and public health agencies should continue to offer patients a multidose schedule,” said Brown. “This is the only way to ensure individuals are protected against HPV infection and cancer over the long term.”
The study was supported by the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Bill & Melinda Gates Foundation, and Canadian Immunization Research Network. Sauvageau, Quach-Thanh, and Brown declared no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM CMAJ
Heart Attack, Stroke Survivors at High Risk for Long COVID
Primary care doctors and specialists should advise patients who have already experienced a heart attack or stroke that they are at a higher risk for long COVID and need to take steps to avoid contracting the virus, according to new research.
The study, led by researchers at Columbia University, New York City, suggests that anyone with cardiovascular disease (CVD) — defined as having experienced a heart attack or stroke — should consider getting the updated COVID vaccine boosters. They also suggest patients with CVD take other steps to avoid an acute infection, such as avoiding crowded indoor spaces.
There is no specific test or treatment for long COVID, which can become disabling and chronic. Long COVID is defined by the failure to recover from acute COVID-19 in 90 days.
The scientists used data from nearly 5000 people enrolled in 14 established, ongoing research programs, including the 76-year-old Framingham Heart Study. The results of the analysis of the “mega-cohort” were published in JAMA Network Open.
Most of the 14 studies already had 10-20 years of data on the cardiac health of thousands of enrollees, said Norrina B. Allen, one of the authors and a cardiac epidemiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
“This is a particularly strong study that looked at risk factors — or individual health — prior to developing COVID and their impact on the likely of recovering from COVID,” she said.
In addition to those with CVD, women and adults with preexisting chronic illnesses took longer to recover.
More than 20% of those in the large, racially and ethnically diverse US population–based study did not recover from COVID in 90 days. The researchers found that the median self-reported time to recovery from acute infection was 20 days.
While women and those with chronic illness had a higher risk for long COVID, vaccination and infection with the Omicron variant wave were associated with shorter recovery times.
These findings make sense, said Ziyad Al-Aly, MD, chief of research at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University in St. Louis, Missouri.
“We also see that COVID-19 can lead to new-onset cardiovascular disease,” said Al-Aly, who was not involved in the study. “There is clearly a (link) between COVID and cardiovascular disease. These two seem to be intimately intertwined. In my view, this emphasizes the importance of targeting these individuals for vaccination and potentially antivirals (when they get infected) to help reduce their risk of adverse events and ameliorate their chance of full and fast recovery.”
The study used data from the Collaborative Cohort of Cohorts for COVID-19 Research. The long list of researchers contributing to this study includes epidemiologists, biostatisticians, neurologists, pulmonologists, and cardiologists. The data come from a list of cohorts like the Framingham Heart Study, which identified key risk factors for CVD, including cholesterol levels. Other studies include the Atherosclerosis Risk in Communities study, which began in the mid-1980s. Researchers there recruited a cohort of 15,792 men and women in rural North Carolina and Mississippi and suburban Minneapolis. They enrolled a high number of African American participants, who have been underrepresented in past studies. Other cohorts focused on young adults with CVD and Hispanics, while another focused on people with chronic obstructive pulmonary disease.
Lead author Elizabeth C. Oelsner, MD, of Columbia University Irving Medical Center in New York City, said she was not surprised by the CVD-long COVID link.
“We were aware that individuals with CVD were at higher risk of a more severe acute infection,” she said. “We were also seeing evidence that long and severe infection led to persistent symptoms.”
Oelsner noted that many patients still take more than 3 months to recover, even during the Omicron wave.
“While that has improved over the course of the pandemic, many individuals are taking a very long time to recover, and that can have a huge burden on the patient,” she said.
She encourages healthcare providers to tell patients at higher risk to take steps to avoid the virus, including vaccination and boosters.
A version of this article first appeared on Medscape.com.
Primary care doctors and specialists should advise patients who have already experienced a heart attack or stroke that they are at a higher risk for long COVID and need to take steps to avoid contracting the virus, according to new research.
The study, led by researchers at Columbia University, New York City, suggests that anyone with cardiovascular disease (CVD) — defined as having experienced a heart attack or stroke — should consider getting the updated COVID vaccine boosters. They also suggest patients with CVD take other steps to avoid an acute infection, such as avoiding crowded indoor spaces.
There is no specific test or treatment for long COVID, which can become disabling and chronic. Long COVID is defined by the failure to recover from acute COVID-19 in 90 days.
The scientists used data from nearly 5000 people enrolled in 14 established, ongoing research programs, including the 76-year-old Framingham Heart Study. The results of the analysis of the “mega-cohort” were published in JAMA Network Open.
Most of the 14 studies already had 10-20 years of data on the cardiac health of thousands of enrollees, said Norrina B. Allen, one of the authors and a cardiac epidemiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
“This is a particularly strong study that looked at risk factors — or individual health — prior to developing COVID and their impact on the likely of recovering from COVID,” she said.
In addition to those with CVD, women and adults with preexisting chronic illnesses took longer to recover.
More than 20% of those in the large, racially and ethnically diverse US population–based study did not recover from COVID in 90 days. The researchers found that the median self-reported time to recovery from acute infection was 20 days.
While women and those with chronic illness had a higher risk for long COVID, vaccination and infection with the Omicron variant wave were associated with shorter recovery times.
These findings make sense, said Ziyad Al-Aly, MD, chief of research at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University in St. Louis, Missouri.
“We also see that COVID-19 can lead to new-onset cardiovascular disease,” said Al-Aly, who was not involved in the study. “There is clearly a (link) between COVID and cardiovascular disease. These two seem to be intimately intertwined. In my view, this emphasizes the importance of targeting these individuals for vaccination and potentially antivirals (when they get infected) to help reduce their risk of adverse events and ameliorate their chance of full and fast recovery.”
The study used data from the Collaborative Cohort of Cohorts for COVID-19 Research. The long list of researchers contributing to this study includes epidemiologists, biostatisticians, neurologists, pulmonologists, and cardiologists. The data come from a list of cohorts like the Framingham Heart Study, which identified key risk factors for CVD, including cholesterol levels. Other studies include the Atherosclerosis Risk in Communities study, which began in the mid-1980s. Researchers there recruited a cohort of 15,792 men and women in rural North Carolina and Mississippi and suburban Minneapolis. They enrolled a high number of African American participants, who have been underrepresented in past studies. Other cohorts focused on young adults with CVD and Hispanics, while another focused on people with chronic obstructive pulmonary disease.
Lead author Elizabeth C. Oelsner, MD, of Columbia University Irving Medical Center in New York City, said she was not surprised by the CVD-long COVID link.
“We were aware that individuals with CVD were at higher risk of a more severe acute infection,” she said. “We were also seeing evidence that long and severe infection led to persistent symptoms.”
Oelsner noted that many patients still take more than 3 months to recover, even during the Omicron wave.
“While that has improved over the course of the pandemic, many individuals are taking a very long time to recover, and that can have a huge burden on the patient,” she said.
She encourages healthcare providers to tell patients at higher risk to take steps to avoid the virus, including vaccination and boosters.
A version of this article first appeared on Medscape.com.
Primary care doctors and specialists should advise patients who have already experienced a heart attack or stroke that they are at a higher risk for long COVID and need to take steps to avoid contracting the virus, according to new research.
The study, led by researchers at Columbia University, New York City, suggests that anyone with cardiovascular disease (CVD) — defined as having experienced a heart attack or stroke — should consider getting the updated COVID vaccine boosters. They also suggest patients with CVD take other steps to avoid an acute infection, such as avoiding crowded indoor spaces.
There is no specific test or treatment for long COVID, which can become disabling and chronic. Long COVID is defined by the failure to recover from acute COVID-19 in 90 days.
The scientists used data from nearly 5000 people enrolled in 14 established, ongoing research programs, including the 76-year-old Framingham Heart Study. The results of the analysis of the “mega-cohort” were published in JAMA Network Open.
Most of the 14 studies already had 10-20 years of data on the cardiac health of thousands of enrollees, said Norrina B. Allen, one of the authors and a cardiac epidemiologist at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
“This is a particularly strong study that looked at risk factors — or individual health — prior to developing COVID and their impact on the likely of recovering from COVID,” she said.
In addition to those with CVD, women and adults with preexisting chronic illnesses took longer to recover.
More than 20% of those in the large, racially and ethnically diverse US population–based study did not recover from COVID in 90 days. The researchers found that the median self-reported time to recovery from acute infection was 20 days.
While women and those with chronic illness had a higher risk for long COVID, vaccination and infection with the Omicron variant wave were associated with shorter recovery times.
These findings make sense, said Ziyad Al-Aly, MD, chief of research at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University in St. Louis, Missouri.
“We also see that COVID-19 can lead to new-onset cardiovascular disease,” said Al-Aly, who was not involved in the study. “There is clearly a (link) between COVID and cardiovascular disease. These two seem to be intimately intertwined. In my view, this emphasizes the importance of targeting these individuals for vaccination and potentially antivirals (when they get infected) to help reduce their risk of adverse events and ameliorate their chance of full and fast recovery.”
The study used data from the Collaborative Cohort of Cohorts for COVID-19 Research. The long list of researchers contributing to this study includes epidemiologists, biostatisticians, neurologists, pulmonologists, and cardiologists. The data come from a list of cohorts like the Framingham Heart Study, which identified key risk factors for CVD, including cholesterol levels. Other studies include the Atherosclerosis Risk in Communities study, which began in the mid-1980s. Researchers there recruited a cohort of 15,792 men and women in rural North Carolina and Mississippi and suburban Minneapolis. They enrolled a high number of African American participants, who have been underrepresented in past studies. Other cohorts focused on young adults with CVD and Hispanics, while another focused on people with chronic obstructive pulmonary disease.
Lead author Elizabeth C. Oelsner, MD, of Columbia University Irving Medical Center in New York City, said she was not surprised by the CVD-long COVID link.
“We were aware that individuals with CVD were at higher risk of a more severe acute infection,” she said. “We were also seeing evidence that long and severe infection led to persistent symptoms.”
Oelsner noted that many patients still take more than 3 months to recover, even during the Omicron wave.
“While that has improved over the course of the pandemic, many individuals are taking a very long time to recover, and that can have a huge burden on the patient,” she said.
She encourages healthcare providers to tell patients at higher risk to take steps to avoid the virus, including vaccination and boosters.
A version of this article first appeared on Medscape.com.
Live Rotavirus Vaccine Safe for Newborns of Biologic-Treated Moms With IBD
No adverse events or impairment of the immune system emerged in babies at 7 days, 1 month, and 9 months post vaccination, in findings from a small Canadian study published in Clinical Gastroenterology and Hepatology.
The study found normal extended immune function testing in infants despite third-trimester maternal biologic therapy and regardless of circulating drug levels. The data provide reassurance about live rotavirus vaccination in this population and may also offer insights into the safety of other live vaccines in biologic-exposed individuals, wrote investigators led by gastroenterologist Cynthia H. Seow, MD, a professor in the Cumming School of Medicine at the University of Calgary in Alberta, Canada.
“Despite the well-established safety and effectiveness of non–live vaccination in individuals with IBD, including those on immunomodulators and biologic therapy, vaccine uptake in pregnant women with IBD and their infants remains suboptimal,” Seow said in an interview. This largely arises from maternal and physician concerns regarding transplacental transfer of IBD therapies and their impact on the safety of vaccination.
“These concerns were heightened after reports emerged of five fatal outcomes following the administration of the live Bacille Calmette-Guérin [BCG] vaccine in biologic-exposed infants. However, it had already been reported that inadvertent administration of the live oral rotavirus vaccine, a very different vaccine in terms of target and mechanism of action, in biologic-exposed individuals had not been associated with significant adverse effects,” she said.
They undertook their analysis with the hypothesis that vaccination would carry low risk, although the live oral vaccine is not currently recommended in biologic-exposed infants. “Yet rotavirus is a leading cause of severe, dehydrating diarrhea in children under the age of 5 years globally, and vaccination has led to significant reductions in hospitalizations and mortality,” Seow added.
Provision of the vaccine to anti–tumor necrosis factor (TNF)–exposed infants has been incorporated into the Canadian Public Health and Immunization guidelines, as the majority of the biologic-exposed infants were exposed to anti-TNF agents. “And with collection of further data, we expect that this will be extended to other biologic agent exposure. These data are important to pregnant women with IBD as they help to normalize their care. Pregnancy is difficult enough without having to remember exceptions to care,” Seow said.
“Before some of the studies came out, broad guidelines recommended that live vaccines should not be used in biologic-exposed infants, but this had been thought to be overly zealous and too conservative, and the risk was thought to be low,” said Elizabeth Spencer, MD, an assistant professor of pediatrics in the Division of Pediatric Gastroenterology at the Icahn School of Medicine at Mount Sinai in New York City, in an interview. Spencer was not involved in the Canadian study.
“At our center, we had some moms on biologics during pregnancy who forgot and had their babies vaccinated for rotavirus, and the babies were all fine,” she said.
The safety of this vaccine has been confirmed by several small studies and recently the PIANO Helmsley Global Consensus on Pregnancy and Inflammatory Bowel Disease, which was presented at Digestive Disease Week 2024. The consensus encompasses preconception counseling and the safety of IBD medications during pregnancy and lactation.
“Another concern, however, was that giving a live GI bug like rotavirus to babies might overstimulate their immune systems and provoke IBD,” Spencer added. “While a number of population-based studies in the US and Europe showed that was not the case, at least in the general population, there was a suggestion that, down the road, vaccination might be mildly protective against IBD in some cases.”
She added the caveat that these studies were not done in mothers and their babies with IBD, who might be inherently at greater risk for IBD. “So, a question for future research would be, ‘Is immune stimulation of the gut in IBD moms and their babies a good or a bad thing for their gut?’ ”
Spencer conceded that “the data present a bit of a blurry picture, but I think it’s always better just to vaccinate according to the regular schedule. The current data say there is no added risk, but it would be nice to look specifically at risk in moms with IBD and their children.”
The Study
The prospective cohort study is a substudy of a larger 2023 one that included biologic use in a range of maternal illnesses, not just IBD.
For the current study, Seow and colleagues identified 57 infants born to 52 mothers with IBD attending a pregnancy clinic at the University of Calgary in the period 2019-2023. Almost 81% of the mothers had Crohn’s disease, and the median duration of IBD was 10 years. The median gestational age at delivery was 39 weeks, and almost 60% of deliveries were vaginal. The infants had been exposed in utero to infliximab (n = 21), adalimumab (n = 19), vedolizumab (n = 10), and ustekinumab (n = 7) in the third trimester.
The 57 biologic-exposed infants underwent standardized clinical assessments, drug concentration, and immune function testing. The live oral rotavirus vaccine series was provided to 50 infants, with the first dose at a median of 13 weeks of age. Immunologic assessments validated for age were normal in all infants despite median infliximab concentrations of 6.1 μg/mL (range, 0.4-28.8 μg/mL), adalimumab concentrations of 1.7 μg/mL (range, 0.7-7.9 μg/mL), ustekinumab concentrations of 0.6 μg/mL (range, 0-1.1), and undetectable for vedolizumab at 10.7 weeks of age.
As anticipated, infant immune function was normal regardless of circulating drug levels.
The overall message, said Seow, is “healthy mum equals healthy baby. Be more concerned regarding active inflammation than active medications. In almost all circumstances, treat to target in pregnancy as you would in the nonpregnant state.” She added, however, that further studies are needed to determine the safety and optimal timing of other live vaccines, such as the BCG, in the presence of biologic therapy.
This study was funded by the Alberta Children’s Hospital Research Institute. Seow reported advisory/speaker’s fees for Janssen, AbbVie, Takeda, Pfizer, Fresenius Kabi, Bristol-Myers Squibb, Pharmascience, and Lilly, as well as funding from Alberta Children’s Hospital Research Institute, Crohn’s and Colitis Canada, the Canadian Institutes of Health Research, and Calgary Health Trust, and data safety monitoring from New South Wales Government Health, Australia. Multiple coauthors disclosed similar consulting or speaker relationships with private industry. Spencer had no competing interests with regard to her comments.
A version of this article first appeared on Medscape.com.
No adverse events or impairment of the immune system emerged in babies at 7 days, 1 month, and 9 months post vaccination, in findings from a small Canadian study published in Clinical Gastroenterology and Hepatology.
The study found normal extended immune function testing in infants despite third-trimester maternal biologic therapy and regardless of circulating drug levels. The data provide reassurance about live rotavirus vaccination in this population and may also offer insights into the safety of other live vaccines in biologic-exposed individuals, wrote investigators led by gastroenterologist Cynthia H. Seow, MD, a professor in the Cumming School of Medicine at the University of Calgary in Alberta, Canada.
“Despite the well-established safety and effectiveness of non–live vaccination in individuals with IBD, including those on immunomodulators and biologic therapy, vaccine uptake in pregnant women with IBD and their infants remains suboptimal,” Seow said in an interview. This largely arises from maternal and physician concerns regarding transplacental transfer of IBD therapies and their impact on the safety of vaccination.
“These concerns were heightened after reports emerged of five fatal outcomes following the administration of the live Bacille Calmette-Guérin [BCG] vaccine in biologic-exposed infants. However, it had already been reported that inadvertent administration of the live oral rotavirus vaccine, a very different vaccine in terms of target and mechanism of action, in biologic-exposed individuals had not been associated with significant adverse effects,” she said.
They undertook their analysis with the hypothesis that vaccination would carry low risk, although the live oral vaccine is not currently recommended in biologic-exposed infants. “Yet rotavirus is a leading cause of severe, dehydrating diarrhea in children under the age of 5 years globally, and vaccination has led to significant reductions in hospitalizations and mortality,” Seow added.
Provision of the vaccine to anti–tumor necrosis factor (TNF)–exposed infants has been incorporated into the Canadian Public Health and Immunization guidelines, as the majority of the biologic-exposed infants were exposed to anti-TNF agents. “And with collection of further data, we expect that this will be extended to other biologic agent exposure. These data are important to pregnant women with IBD as they help to normalize their care. Pregnancy is difficult enough without having to remember exceptions to care,” Seow said.
“Before some of the studies came out, broad guidelines recommended that live vaccines should not be used in biologic-exposed infants, but this had been thought to be overly zealous and too conservative, and the risk was thought to be low,” said Elizabeth Spencer, MD, an assistant professor of pediatrics in the Division of Pediatric Gastroenterology at the Icahn School of Medicine at Mount Sinai in New York City, in an interview. Spencer was not involved in the Canadian study.
“At our center, we had some moms on biologics during pregnancy who forgot and had their babies vaccinated for rotavirus, and the babies were all fine,” she said.
The safety of this vaccine has been confirmed by several small studies and recently the PIANO Helmsley Global Consensus on Pregnancy and Inflammatory Bowel Disease, which was presented at Digestive Disease Week 2024. The consensus encompasses preconception counseling and the safety of IBD medications during pregnancy and lactation.
“Another concern, however, was that giving a live GI bug like rotavirus to babies might overstimulate their immune systems and provoke IBD,” Spencer added. “While a number of population-based studies in the US and Europe showed that was not the case, at least in the general population, there was a suggestion that, down the road, vaccination might be mildly protective against IBD in some cases.”
She added the caveat that these studies were not done in mothers and their babies with IBD, who might be inherently at greater risk for IBD. “So, a question for future research would be, ‘Is immune stimulation of the gut in IBD moms and their babies a good or a bad thing for their gut?’ ”
Spencer conceded that “the data present a bit of a blurry picture, but I think it’s always better just to vaccinate according to the regular schedule. The current data say there is no added risk, but it would be nice to look specifically at risk in moms with IBD and their children.”
The Study
The prospective cohort study is a substudy of a larger 2023 one that included biologic use in a range of maternal illnesses, not just IBD.
For the current study, Seow and colleagues identified 57 infants born to 52 mothers with IBD attending a pregnancy clinic at the University of Calgary in the period 2019-2023. Almost 81% of the mothers had Crohn’s disease, and the median duration of IBD was 10 years. The median gestational age at delivery was 39 weeks, and almost 60% of deliveries were vaginal. The infants had been exposed in utero to infliximab (n = 21), adalimumab (n = 19), vedolizumab (n = 10), and ustekinumab (n = 7) in the third trimester.
The 57 biologic-exposed infants underwent standardized clinical assessments, drug concentration, and immune function testing. The live oral rotavirus vaccine series was provided to 50 infants, with the first dose at a median of 13 weeks of age. Immunologic assessments validated for age were normal in all infants despite median infliximab concentrations of 6.1 μg/mL (range, 0.4-28.8 μg/mL), adalimumab concentrations of 1.7 μg/mL (range, 0.7-7.9 μg/mL), ustekinumab concentrations of 0.6 μg/mL (range, 0-1.1), and undetectable for vedolizumab at 10.7 weeks of age.
As anticipated, infant immune function was normal regardless of circulating drug levels.
The overall message, said Seow, is “healthy mum equals healthy baby. Be more concerned regarding active inflammation than active medications. In almost all circumstances, treat to target in pregnancy as you would in the nonpregnant state.” She added, however, that further studies are needed to determine the safety and optimal timing of other live vaccines, such as the BCG, in the presence of biologic therapy.
This study was funded by the Alberta Children’s Hospital Research Institute. Seow reported advisory/speaker’s fees for Janssen, AbbVie, Takeda, Pfizer, Fresenius Kabi, Bristol-Myers Squibb, Pharmascience, and Lilly, as well as funding from Alberta Children’s Hospital Research Institute, Crohn’s and Colitis Canada, the Canadian Institutes of Health Research, and Calgary Health Trust, and data safety monitoring from New South Wales Government Health, Australia. Multiple coauthors disclosed similar consulting or speaker relationships with private industry. Spencer had no competing interests with regard to her comments.
A version of this article first appeared on Medscape.com.
No adverse events or impairment of the immune system emerged in babies at 7 days, 1 month, and 9 months post vaccination, in findings from a small Canadian study published in Clinical Gastroenterology and Hepatology.
The study found normal extended immune function testing in infants despite third-trimester maternal biologic therapy and regardless of circulating drug levels. The data provide reassurance about live rotavirus vaccination in this population and may also offer insights into the safety of other live vaccines in biologic-exposed individuals, wrote investigators led by gastroenterologist Cynthia H. Seow, MD, a professor in the Cumming School of Medicine at the University of Calgary in Alberta, Canada.
“Despite the well-established safety and effectiveness of non–live vaccination in individuals with IBD, including those on immunomodulators and biologic therapy, vaccine uptake in pregnant women with IBD and their infants remains suboptimal,” Seow said in an interview. This largely arises from maternal and physician concerns regarding transplacental transfer of IBD therapies and their impact on the safety of vaccination.
“These concerns were heightened after reports emerged of five fatal outcomes following the administration of the live Bacille Calmette-Guérin [BCG] vaccine in biologic-exposed infants. However, it had already been reported that inadvertent administration of the live oral rotavirus vaccine, a very different vaccine in terms of target and mechanism of action, in biologic-exposed individuals had not been associated with significant adverse effects,” she said.
They undertook their analysis with the hypothesis that vaccination would carry low risk, although the live oral vaccine is not currently recommended in biologic-exposed infants. “Yet rotavirus is a leading cause of severe, dehydrating diarrhea in children under the age of 5 years globally, and vaccination has led to significant reductions in hospitalizations and mortality,” Seow added.
Provision of the vaccine to anti–tumor necrosis factor (TNF)–exposed infants has been incorporated into the Canadian Public Health and Immunization guidelines, as the majority of the biologic-exposed infants were exposed to anti-TNF agents. “And with collection of further data, we expect that this will be extended to other biologic agent exposure. These data are important to pregnant women with IBD as they help to normalize their care. Pregnancy is difficult enough without having to remember exceptions to care,” Seow said.
“Before some of the studies came out, broad guidelines recommended that live vaccines should not be used in biologic-exposed infants, but this had been thought to be overly zealous and too conservative, and the risk was thought to be low,” said Elizabeth Spencer, MD, an assistant professor of pediatrics in the Division of Pediatric Gastroenterology at the Icahn School of Medicine at Mount Sinai in New York City, in an interview. Spencer was not involved in the Canadian study.
“At our center, we had some moms on biologics during pregnancy who forgot and had their babies vaccinated for rotavirus, and the babies were all fine,” she said.
The safety of this vaccine has been confirmed by several small studies and recently the PIANO Helmsley Global Consensus on Pregnancy and Inflammatory Bowel Disease, which was presented at Digestive Disease Week 2024. The consensus encompasses preconception counseling and the safety of IBD medications during pregnancy and lactation.
“Another concern, however, was that giving a live GI bug like rotavirus to babies might overstimulate their immune systems and provoke IBD,” Spencer added. “While a number of population-based studies in the US and Europe showed that was not the case, at least in the general population, there was a suggestion that, down the road, vaccination might be mildly protective against IBD in some cases.”
She added the caveat that these studies were not done in mothers and their babies with IBD, who might be inherently at greater risk for IBD. “So, a question for future research would be, ‘Is immune stimulation of the gut in IBD moms and their babies a good or a bad thing for their gut?’ ”
Spencer conceded that “the data present a bit of a blurry picture, but I think it’s always better just to vaccinate according to the regular schedule. The current data say there is no added risk, but it would be nice to look specifically at risk in moms with IBD and their children.”
The Study
The prospective cohort study is a substudy of a larger 2023 one that included biologic use in a range of maternal illnesses, not just IBD.
For the current study, Seow and colleagues identified 57 infants born to 52 mothers with IBD attending a pregnancy clinic at the University of Calgary in the period 2019-2023. Almost 81% of the mothers had Crohn’s disease, and the median duration of IBD was 10 years. The median gestational age at delivery was 39 weeks, and almost 60% of deliveries were vaginal. The infants had been exposed in utero to infliximab (n = 21), adalimumab (n = 19), vedolizumab (n = 10), and ustekinumab (n = 7) in the third trimester.
The 57 biologic-exposed infants underwent standardized clinical assessments, drug concentration, and immune function testing. The live oral rotavirus vaccine series was provided to 50 infants, with the first dose at a median of 13 weeks of age. Immunologic assessments validated for age were normal in all infants despite median infliximab concentrations of 6.1 μg/mL (range, 0.4-28.8 μg/mL), adalimumab concentrations of 1.7 μg/mL (range, 0.7-7.9 μg/mL), ustekinumab concentrations of 0.6 μg/mL (range, 0-1.1), and undetectable for vedolizumab at 10.7 weeks of age.
As anticipated, infant immune function was normal regardless of circulating drug levels.
The overall message, said Seow, is “healthy mum equals healthy baby. Be more concerned regarding active inflammation than active medications. In almost all circumstances, treat to target in pregnancy as you would in the nonpregnant state.” She added, however, that further studies are needed to determine the safety and optimal timing of other live vaccines, such as the BCG, in the presence of biologic therapy.
This study was funded by the Alberta Children’s Hospital Research Institute. Seow reported advisory/speaker’s fees for Janssen, AbbVie, Takeda, Pfizer, Fresenius Kabi, Bristol-Myers Squibb, Pharmascience, and Lilly, as well as funding from Alberta Children’s Hospital Research Institute, Crohn’s and Colitis Canada, the Canadian Institutes of Health Research, and Calgary Health Trust, and data safety monitoring from New South Wales Government Health, Australia. Multiple coauthors disclosed similar consulting or speaker relationships with private industry. Spencer had no competing interests with regard to her comments.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Too Few Immunocompromised Veterans Are Getting Zoster Vaccinations
TOPLINE:
the low rate of herpes zoster vaccination in this immunocompromised group, especially among younger individuals, is concerning.
METHODOLOGY:
- In 2021, the Food and Drug Administration authorized the use of RZV for adults aged 18 years or older on chronic immunosuppressive medications because of their high risk for herpes zoster and its related complications, followed by updated guidance from the Centers for Disease Control and Prevention and American College of Rheumatology in 2021 and 2022, respectively.
- This study aimed to assess the receipt of RZV among veterans receiving immunosuppressive medications within the Veterans Health Administration (VHA) healthcare system before and after the expanded indications in February 2022.
- It included 190,162 veterans who were prescribed one or more immunosuppressive medications for at least 90 days at 130 medical facilities between January 1, 2018, and June 30, 2023.
- A total of 23,295 veterans (12.3%) were younger than 50 years by the end of the study period.
- The outcome measured was the percentage of veterans with one or more doses of RZV documented during the study period.
TAKEAWAY:
- Among veterans aged 50 years or older, 36.2% and 49.8% received an RZV before the expanded indication and by mid-2023, respectively. Even though the rate of vaccination is higher than that observed in the 2021 National Health Interview Survey, significant room for improvement remains.
- Among veterans younger than 50 years, very few (2.8%) received an RZV before the expanded indication, and only 13.4% received it by mid-2023.
- Demographic factors associated with lower odds of vaccination included male sex, African American or unknown race, and nonurban residence (P ≤ .004 for all).
- Those who received targeted synthetic disease-modifying antirheumatic drugs (DMARDs) alone or in combination with other drugs or those who received other vaccines were more likely to receive RZV than those who received conventional synthetic DMARD monotherapy (P < .001 for both).
IN PRACTICE:
“Future work to improve RZV vaccination in patients at high risk should focus on creating informatics tools to identify individuals at high risk and standardizing vaccination guidelines across subspecialties,” the authors wrote.
SOURCE:
This study was led by Sharon Abada, MD, University of California, San Francisco. It was published online on October 11, 2024, in JAMA Network Open.
LIMITATIONS:
This study may not be generalizable to nonveteran populations or countries outside the United States. Limitations also included difficulty with capturing vaccinations not administered within the VHA system, which may have resulted in an underestimation of the percentage of patients vaccinated.
DISCLOSURES:
This work was funded by grants from the VA Quality Enhancement Research Initiative and the Agency for Healthcare Research and Quality. Some authors reported receiving grants from institutions and pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
the low rate of herpes zoster vaccination in this immunocompromised group, especially among younger individuals, is concerning.
METHODOLOGY:
- In 2021, the Food and Drug Administration authorized the use of RZV for adults aged 18 years or older on chronic immunosuppressive medications because of their high risk for herpes zoster and its related complications, followed by updated guidance from the Centers for Disease Control and Prevention and American College of Rheumatology in 2021 and 2022, respectively.
- This study aimed to assess the receipt of RZV among veterans receiving immunosuppressive medications within the Veterans Health Administration (VHA) healthcare system before and after the expanded indications in February 2022.
- It included 190,162 veterans who were prescribed one or more immunosuppressive medications for at least 90 days at 130 medical facilities between January 1, 2018, and June 30, 2023.
- A total of 23,295 veterans (12.3%) were younger than 50 years by the end of the study period.
- The outcome measured was the percentage of veterans with one or more doses of RZV documented during the study period.
TAKEAWAY:
- Among veterans aged 50 years or older, 36.2% and 49.8% received an RZV before the expanded indication and by mid-2023, respectively. Even though the rate of vaccination is higher than that observed in the 2021 National Health Interview Survey, significant room for improvement remains.
- Among veterans younger than 50 years, very few (2.8%) received an RZV before the expanded indication, and only 13.4% received it by mid-2023.
- Demographic factors associated with lower odds of vaccination included male sex, African American or unknown race, and nonurban residence (P ≤ .004 for all).
- Those who received targeted synthetic disease-modifying antirheumatic drugs (DMARDs) alone or in combination with other drugs or those who received other vaccines were more likely to receive RZV than those who received conventional synthetic DMARD monotherapy (P < .001 for both).
IN PRACTICE:
“Future work to improve RZV vaccination in patients at high risk should focus on creating informatics tools to identify individuals at high risk and standardizing vaccination guidelines across subspecialties,” the authors wrote.
SOURCE:
This study was led by Sharon Abada, MD, University of California, San Francisco. It was published online on October 11, 2024, in JAMA Network Open.
LIMITATIONS:
This study may not be generalizable to nonveteran populations or countries outside the United States. Limitations also included difficulty with capturing vaccinations not administered within the VHA system, which may have resulted in an underestimation of the percentage of patients vaccinated.
DISCLOSURES:
This work was funded by grants from the VA Quality Enhancement Research Initiative and the Agency for Healthcare Research and Quality. Some authors reported receiving grants from institutions and pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
the low rate of herpes zoster vaccination in this immunocompromised group, especially among younger individuals, is concerning.
METHODOLOGY:
- In 2021, the Food and Drug Administration authorized the use of RZV for adults aged 18 years or older on chronic immunosuppressive medications because of their high risk for herpes zoster and its related complications, followed by updated guidance from the Centers for Disease Control and Prevention and American College of Rheumatology in 2021 and 2022, respectively.
- This study aimed to assess the receipt of RZV among veterans receiving immunosuppressive medications within the Veterans Health Administration (VHA) healthcare system before and after the expanded indications in February 2022.
- It included 190,162 veterans who were prescribed one or more immunosuppressive medications for at least 90 days at 130 medical facilities between January 1, 2018, and June 30, 2023.
- A total of 23,295 veterans (12.3%) were younger than 50 years by the end of the study period.
- The outcome measured was the percentage of veterans with one or more doses of RZV documented during the study period.
TAKEAWAY:
- Among veterans aged 50 years or older, 36.2% and 49.8% received an RZV before the expanded indication and by mid-2023, respectively. Even though the rate of vaccination is higher than that observed in the 2021 National Health Interview Survey, significant room for improvement remains.
- Among veterans younger than 50 years, very few (2.8%) received an RZV before the expanded indication, and only 13.4% received it by mid-2023.
- Demographic factors associated with lower odds of vaccination included male sex, African American or unknown race, and nonurban residence (P ≤ .004 for all).
- Those who received targeted synthetic disease-modifying antirheumatic drugs (DMARDs) alone or in combination with other drugs or those who received other vaccines were more likely to receive RZV than those who received conventional synthetic DMARD monotherapy (P < .001 for both).
IN PRACTICE:
“Future work to improve RZV vaccination in patients at high risk should focus on creating informatics tools to identify individuals at high risk and standardizing vaccination guidelines across subspecialties,” the authors wrote.
SOURCE:
This study was led by Sharon Abada, MD, University of California, San Francisco. It was published online on October 11, 2024, in JAMA Network Open.
LIMITATIONS:
This study may not be generalizable to nonveteran populations or countries outside the United States. Limitations also included difficulty with capturing vaccinations not administered within the VHA system, which may have resulted in an underestimation of the percentage of patients vaccinated.
DISCLOSURES:
This work was funded by grants from the VA Quality Enhancement Research Initiative and the Agency for Healthcare Research and Quality. Some authors reported receiving grants from institutions and pharmaceutical companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Maternal Immunization to Prevent Serious Respiratory Illness
Editor’s Note: Sadly, this is the last column in the Master Class Obstetrics series. This award-winning column has been part of Ob.Gyn. News for 20 years. The deep discussion of cutting-edge topics in obstetrics by specialists and researchers will be missed as will the leadership and curation of topics by Dr. E. Albert Reece.
Introduction: The Need for Increased Vigilance About Maternal Immunization
Viruses are becoming increasingly prevalent in our world and the consequences of viral infections are implicated in a growing number of disease states. It is well established that certain cancers are caused by viruses and it is increasingly evident that viral infections can trigger the development of chronic illness. In pregnant women, viruses such as cytomegalovirus can cause infection in utero and lead to long-term impairments for the baby.
Likewise, it appears that the virulence of viruses is increasing, whether it be the respiratory syncytial virus (RSV) in children or the severe acute respiratory syndrome (SARS) coronaviruses in adults. Clearly, our environment is changing, with increases in population growth and urbanization, for instance, and an intensification of climate change and its effects. Viruses are part of this changing background.
Vaccines are our most powerful tool to protect people of all ages against viral threats, and fortunately, we benefit from increasing expertise in vaccinology. Since 1974, the University of Maryland School of Medicine has a Center for Vaccine Development and Global Health that has conducted research on vaccines to defend against the Zika virus, H1N1, Ebola, and SARS-CoV-2.
We’re not alone. Other vaccinology centers across the country — as well as the National Institutes of Health at the national level, through its National Institute of Allergy and Infectious Diseases — are doing research and developing vaccines to combat viral diseases.
In this column, we are focused on viral diseases in pregnancy and the role that vaccines can play in preventing serious respiratory illness in mothers and their newborns. I have invited Laura E. Riley, MD, the Given Foundation Professor and Chair of Obstetrics and Gynecology at Weill Cornell Medicine, to address the importance of maternal immunization and how we can best counsel our patients and improve immunization rates.
As Dr. Riley explains, we are in a new era, and it behooves us all to be more vigilant about recommending vaccines, combating misperceptions, addressing patients’ knowledge gaps, and administering vaccines whenever possible.
Dr. Reece is the former Dean of Medicine & University Executive VP, and The Distinguished University and Endowed Professor & Director of the Center for Advanced Research Training and Innovation (CARTI) at the University of Maryland School of Medicine, as well as senior scientist at the Center for Birth Defects Research.
The alarming decline in maternal immunization rates that occurred in the wake of the COVID-19 pandemic means that, now more than ever, we must fully embrace our responsibility to recommend immunizations in pregnancy and to communicate what is known about their efficacy and safety. Data show that vaccination rates drop when we do not offer vaccines in our offices, so whenever possible, we should administer them as well.
The ob.gyn. is the patient’s most trusted person in pregnancy. When patients decline or express hesitancy about vaccines, it is incumbent upon us to ask why. Oftentimes, we can identify areas in which patients lack knowledge or have misperceptions and we can successfully educate the patient or change their perspective or misunderstanding concerning the importance of vaccination for themselves and their babies. (See Table 1.) We can also successfully address concerns about safety.
The safety of COVID-19 vaccinations in pregnancy is now backed by several years of data from multiple studies showing no increase in birth defects, preterm delivery, miscarriage, or stillbirth.
Data also show that pregnant patients are more likely than patients who are not pregnant to need hospitalization and intensive care when infected with SARS-CoV-2 and are at risk of having complications that can affect pregnancy and the newborn, including preterm birth and stillbirth. Vaccination has been shown to reduce the risk of severe illness and the risk of such adverse obstetrical outcomes, in addition to providing protection for the infant early on.
Similarly, influenza has long been more likely to be severe in pregnant patients, with an increased risk of poor obstetrical outcomes. Vaccines similarly provide “two for one protection,” protecting both mother and baby, and are, of course, backed by many years of safety and efficacy data.
With the new maternal respiratory syncytial virus (RSV) vaccine, now in its second year of availability, the goal is to protect the baby from RSV-caused serious lower respiratory tract illness. The illness has contributed to tens of thousands of annual hospitalizations and up to several hundred deaths every year in children younger than 5 years — particularly in those under age 6 months.
The RSV monoclonal antibody nirsevimab is available for the newborn as an alternative to maternal immunization but the maternal vaccine is optimal in that it will provide immediate rather than delayed protection for the newborn. The maternal vaccine is recommended during weeks 32-36 of pregnancy in mothers who were not vaccinated during last year’s RSV season. With real-world experience from year one, the available safety data are reassuring.
Counseling About Influenza and COVID-19 Vaccination
The COVID-19 pandemic took a toll on vaccination interest/receptivity broadly in pregnant and nonpregnant people. Among pregnant individuals, influenza vaccination coverage declined from 71% in the 2019-2020 influenza season to 56% in the 2021-2022 season, according to data from the Centers for Disease Control and Prevention’s Vaccine Safety Datalink.4 Coverage for the 2022-2023 and 2023-2024 influenza seasons was even worse: well under 50%.5
Fewer pregnant women have received updated COVID-19 vaccines. Only 13% of pregnant persons overall received the updated 2023-2024 COVID-19 booster vaccine (through March 30, 2024), according to the CDC.6
Maternal immunization for influenza has been recommended in the United States since 2004 (part of the recommendation that everyone over the age of 6 months receive an annual flu vaccine), and flu vaccines have been given to millions of pregnant women, but the H1N1 pandemic of 2009 reinforced its value as a priority for prenatal care. Most of the women who became severely ill from the H1N1 virus were young and healthy, without co-existing conditions known to increase risk.7
It became clearer during the H1N1 pandemic that pregnancy itself — which is associated with physiologic changes such as decreased lung capacity, increased nasal congestion and changes in the immune system – is its own significant risk factor for severe illness from the influenza virus. This increased risk applies to COVID-19 as well.
As COVID-19 has become endemic, with hospitalizations and deaths not reaching the levels of previous surges — and with mask-wearing and other preventive measures having declined — patients understandably have become more complacent. Some patients are vaccine deniers, but in my practice, these patients are a much smaller group than those who believe COVID-19 “is no big deal,” especially if they have had infections recently.
This is why it’s important to actively listen to concerns and to ask patients who decline a vaccination why they are hesitant. Blanket messages about vaccine efficacy and safety are the first step, but individualized, more pointed conversations based on the patient’s personal experiences and beliefs have become increasingly important.
I routinely tell pregnant patients about the risks of COVID-19 and I explain that it has been difficult to predict who will develop severe illness. Sometimes more conversation is needed. For those who are still hesitant or who tell me they feel protected by a recent infection, for instance, I provide more detail on the unique risks of pregnancy — the fact that “pregnancy is different” — and that natural immunity wanes while the protection afforded by immunization is believed to last longer. Many women are also concerned about the safety of the COVID-19 vaccine, so having safety data at your fingertips is helpful. (See Table 2.)
The fact that influenza and COVID-19 vaccination protect the newborn as well as the mother is something that I find is underappreciated by many patients. Explaining that infants likely benefit from the passage of antibodies across the placenta should be part of patient counseling.
Counseling About RSV Vaccination
Importantly, for the 2024-2025 RSV season, the maternal RSV vaccine (Abrysvo, Pfizer) is recommended only for pregnant women who did not receive the vaccine during the 2023-2024 season. When more research is done and more data are obtained showing how long the immune response persists post vaccination, it may be that the US Food and Drug Administration (FDA) will approve the maternal RSV vaccine for use in every pregnancy.
The later timing of the vaccination recommendation — 32-36 weeks’ gestation — reflects a conservative approach taken by the FDA in response to data from one of the pivotal trials showing a numerical trend toward more preterm deliveries among vaccinated compared with unvaccinated patients. This imbalance in the original trial, which administered the vaccine during 24-36 weeks of gestation, was seen only in low-income countries with no temporal association, however.
In our experience at two Weill Cornell Medical College–associated hospitals we did not see this trend. Our cohort study of almost 3000 pregnant patients who delivered at 32 weeks’ gestation or later found no increased risk of preterm birth among the 35% of patients who received the RSV vaccine during the 2023-2024 RSV season. We also did not see any difference in preeclampsia, in contrast with original trial data that showed a signal for increased risk.11
When fewer than 2 weeks have elapsed between maternal vaccination and delivery, the monoclonal antibody nirsevimab is recommended for the newborn — ideally before the newborn leaves the hospital. Nirsevimab is also recommended for newborns of mothers who decline vaccination or were not candidates (e.g. vaccinated in a previous pregnancy), or when there is concern about the adequacy of the maternal immune response to the vaccine (e.g. in cases of immunosuppression).
While there was a limited supply of the monoclonal antibody last year, limitations are not expected this year, especially after October.
The ultimate goal is that patients choose the vaccine or the immunoglobulin, given the severity of RSV disease. Patient preferences should be considered. However, given that it takes 2 weeks after vaccination for protection to build up, I stress to patients that if they’ve vaccinated themselves, their newborn will leave the hospital with protection. If nirsevimab is relied upon, I explain, their newborn may not be protected for some period of time.
Take-home Messages
- When patients decline or are hesitant about vaccines, ask why. Listen actively, and work to correct misperceptions and knowledge gaps.
- Whenever possible, offer vaccines in your practice. Vaccination rates drop when this does not occur.
- COVID-vaccine safety is backed by many studies showing no increase in birth defects, preterm delivery, miscarriage, or stillbirth.
- Pregnant women are more likely to have severe illness from the influenza and SARS-CoV-2 viruses. Vaccines can prevent severe illness and can protect the newborn as well as the mother.
- Recommend/administer the maternal RSV vaccine at 32-36 weeks’ gestation in women who did not receive the vaccine in the 2023-2024 season. If mothers aren’t eligible their babies should be offered nirsevimab.
Dr. Riley is the Given Foundation Professor and Chair of Obstetrics and Gynecology at Weill Cornell Medicine and the obstetrician and gynecologist-in-chief at New York Presbyterian Hospital. She disclosed that she has provided one-time consultations to Pfizer (Abrysvo RSV vaccine) and GSK (cytomegalovirus vaccine), and is providing consultant education on CMV for Moderna. She is chair of ACOG’s task force on immunization and emerging infectious diseases, serves on the medical advisory board for MAVEN, and serves as an editor or editorial board member for several medical publications.
References
1. ACOG Committee Opinion No. 741: Maternal Immunization. Obstet Gynecol. 2018;131(6):e214-e217.
2. Centers for Disease Control and Prevention. COVID-19 Vaccination for People Who are Pregnant or Breastfeeding. https://www.cdc.gov/covid/vaccines/pregnant-or-breastfeeding.html.
3. ACOG Practice Advisory on Maternal Respiratory Syncytial Virus Vaccination, September 2023. (Updated August 2024).4. Irving S et al. Open Forum Infect Dis. 2023;10(Suppl 2):ofad500.1002.
5. Flu Vaccination Dashboard, CDC, National Center for Immunization and Respiratory Diseases.
6. Weekly COVID-19 Vaccination Dashboard, CDC. https://www.cdc.gov/covidvaxview/weekly-dashboard/index.html
7. Louie JK et al. N Engl J Med. 2010;362:27-35. 8. Ciapponi A et al. Vaccine. 2021;39(40):5891-908.
9. Prasad S et al. Nature Communications. 2022;13:2414. 10. Fleming-Dutra KE et al. Obstet Gynecol Clin North Am 2023;50(2):279-97. 11. Mouen S et al. JAMA Network Open 2024;7(7):e2419268.
Editor’s Note: Sadly, this is the last column in the Master Class Obstetrics series. This award-winning column has been part of Ob.Gyn. News for 20 years. The deep discussion of cutting-edge topics in obstetrics by specialists and researchers will be missed as will the leadership and curation of topics by Dr. E. Albert Reece.
Introduction: The Need for Increased Vigilance About Maternal Immunization
Viruses are becoming increasingly prevalent in our world and the consequences of viral infections are implicated in a growing number of disease states. It is well established that certain cancers are caused by viruses and it is increasingly evident that viral infections can trigger the development of chronic illness. In pregnant women, viruses such as cytomegalovirus can cause infection in utero and lead to long-term impairments for the baby.
Likewise, it appears that the virulence of viruses is increasing, whether it be the respiratory syncytial virus (RSV) in children or the severe acute respiratory syndrome (SARS) coronaviruses in adults. Clearly, our environment is changing, with increases in population growth and urbanization, for instance, and an intensification of climate change and its effects. Viruses are part of this changing background.
Vaccines are our most powerful tool to protect people of all ages against viral threats, and fortunately, we benefit from increasing expertise in vaccinology. Since 1974, the University of Maryland School of Medicine has a Center for Vaccine Development and Global Health that has conducted research on vaccines to defend against the Zika virus, H1N1, Ebola, and SARS-CoV-2.
We’re not alone. Other vaccinology centers across the country — as well as the National Institutes of Health at the national level, through its National Institute of Allergy and Infectious Diseases — are doing research and developing vaccines to combat viral diseases.
In this column, we are focused on viral diseases in pregnancy and the role that vaccines can play in preventing serious respiratory illness in mothers and their newborns. I have invited Laura E. Riley, MD, the Given Foundation Professor and Chair of Obstetrics and Gynecology at Weill Cornell Medicine, to address the importance of maternal immunization and how we can best counsel our patients and improve immunization rates.
As Dr. Riley explains, we are in a new era, and it behooves us all to be more vigilant about recommending vaccines, combating misperceptions, addressing patients’ knowledge gaps, and administering vaccines whenever possible.
Dr. Reece is the former Dean of Medicine & University Executive VP, and The Distinguished University and Endowed Professor & Director of the Center for Advanced Research Training and Innovation (CARTI) at the University of Maryland School of Medicine, as well as senior scientist at the Center for Birth Defects Research.
The alarming decline in maternal immunization rates that occurred in the wake of the COVID-19 pandemic means that, now more than ever, we must fully embrace our responsibility to recommend immunizations in pregnancy and to communicate what is known about their efficacy and safety. Data show that vaccination rates drop when we do not offer vaccines in our offices, so whenever possible, we should administer them as well.
The ob.gyn. is the patient’s most trusted person in pregnancy. When patients decline or express hesitancy about vaccines, it is incumbent upon us to ask why. Oftentimes, we can identify areas in which patients lack knowledge or have misperceptions and we can successfully educate the patient or change their perspective or misunderstanding concerning the importance of vaccination for themselves and their babies. (See Table 1.) We can also successfully address concerns about safety.
The safety of COVID-19 vaccinations in pregnancy is now backed by several years of data from multiple studies showing no increase in birth defects, preterm delivery, miscarriage, or stillbirth.
Data also show that pregnant patients are more likely than patients who are not pregnant to need hospitalization and intensive care when infected with SARS-CoV-2 and are at risk of having complications that can affect pregnancy and the newborn, including preterm birth and stillbirth. Vaccination has been shown to reduce the risk of severe illness and the risk of such adverse obstetrical outcomes, in addition to providing protection for the infant early on.
Similarly, influenza has long been more likely to be severe in pregnant patients, with an increased risk of poor obstetrical outcomes. Vaccines similarly provide “two for one protection,” protecting both mother and baby, and are, of course, backed by many years of safety and efficacy data.
With the new maternal respiratory syncytial virus (RSV) vaccine, now in its second year of availability, the goal is to protect the baby from RSV-caused serious lower respiratory tract illness. The illness has contributed to tens of thousands of annual hospitalizations and up to several hundred deaths every year in children younger than 5 years — particularly in those under age 6 months.
The RSV monoclonal antibody nirsevimab is available for the newborn as an alternative to maternal immunization but the maternal vaccine is optimal in that it will provide immediate rather than delayed protection for the newborn. The maternal vaccine is recommended during weeks 32-36 of pregnancy in mothers who were not vaccinated during last year’s RSV season. With real-world experience from year one, the available safety data are reassuring.
Counseling About Influenza and COVID-19 Vaccination
The COVID-19 pandemic took a toll on vaccination interest/receptivity broadly in pregnant and nonpregnant people. Among pregnant individuals, influenza vaccination coverage declined from 71% in the 2019-2020 influenza season to 56% in the 2021-2022 season, according to data from the Centers for Disease Control and Prevention’s Vaccine Safety Datalink.4 Coverage for the 2022-2023 and 2023-2024 influenza seasons was even worse: well under 50%.5
Fewer pregnant women have received updated COVID-19 vaccines. Only 13% of pregnant persons overall received the updated 2023-2024 COVID-19 booster vaccine (through March 30, 2024), according to the CDC.6
Maternal immunization for influenza has been recommended in the United States since 2004 (part of the recommendation that everyone over the age of 6 months receive an annual flu vaccine), and flu vaccines have been given to millions of pregnant women, but the H1N1 pandemic of 2009 reinforced its value as a priority for prenatal care. Most of the women who became severely ill from the H1N1 virus were young and healthy, without co-existing conditions known to increase risk.7
It became clearer during the H1N1 pandemic that pregnancy itself — which is associated with physiologic changes such as decreased lung capacity, increased nasal congestion and changes in the immune system – is its own significant risk factor for severe illness from the influenza virus. This increased risk applies to COVID-19 as well.
As COVID-19 has become endemic, with hospitalizations and deaths not reaching the levels of previous surges — and with mask-wearing and other preventive measures having declined — patients understandably have become more complacent. Some patients are vaccine deniers, but in my practice, these patients are a much smaller group than those who believe COVID-19 “is no big deal,” especially if they have had infections recently.
This is why it’s important to actively listen to concerns and to ask patients who decline a vaccination why they are hesitant. Blanket messages about vaccine efficacy and safety are the first step, but individualized, more pointed conversations based on the patient’s personal experiences and beliefs have become increasingly important.
I routinely tell pregnant patients about the risks of COVID-19 and I explain that it has been difficult to predict who will develop severe illness. Sometimes more conversation is needed. For those who are still hesitant or who tell me they feel protected by a recent infection, for instance, I provide more detail on the unique risks of pregnancy — the fact that “pregnancy is different” — and that natural immunity wanes while the protection afforded by immunization is believed to last longer. Many women are also concerned about the safety of the COVID-19 vaccine, so having safety data at your fingertips is helpful. (See Table 2.)
The fact that influenza and COVID-19 vaccination protect the newborn as well as the mother is something that I find is underappreciated by many patients. Explaining that infants likely benefit from the passage of antibodies across the placenta should be part of patient counseling.
Counseling About RSV Vaccination
Importantly, for the 2024-2025 RSV season, the maternal RSV vaccine (Abrysvo, Pfizer) is recommended only for pregnant women who did not receive the vaccine during the 2023-2024 season. When more research is done and more data are obtained showing how long the immune response persists post vaccination, it may be that the US Food and Drug Administration (FDA) will approve the maternal RSV vaccine for use in every pregnancy.
The later timing of the vaccination recommendation — 32-36 weeks’ gestation — reflects a conservative approach taken by the FDA in response to data from one of the pivotal trials showing a numerical trend toward more preterm deliveries among vaccinated compared with unvaccinated patients. This imbalance in the original trial, which administered the vaccine during 24-36 weeks of gestation, was seen only in low-income countries with no temporal association, however.
In our experience at two Weill Cornell Medical College–associated hospitals we did not see this trend. Our cohort study of almost 3000 pregnant patients who delivered at 32 weeks’ gestation or later found no increased risk of preterm birth among the 35% of patients who received the RSV vaccine during the 2023-2024 RSV season. We also did not see any difference in preeclampsia, in contrast with original trial data that showed a signal for increased risk.11
When fewer than 2 weeks have elapsed between maternal vaccination and delivery, the monoclonal antibody nirsevimab is recommended for the newborn — ideally before the newborn leaves the hospital. Nirsevimab is also recommended for newborns of mothers who decline vaccination or were not candidates (e.g. vaccinated in a previous pregnancy), or when there is concern about the adequacy of the maternal immune response to the vaccine (e.g. in cases of immunosuppression).
While there was a limited supply of the monoclonal antibody last year, limitations are not expected this year, especially after October.
The ultimate goal is that patients choose the vaccine or the immunoglobulin, given the severity of RSV disease. Patient preferences should be considered. However, given that it takes 2 weeks after vaccination for protection to build up, I stress to patients that if they’ve vaccinated themselves, their newborn will leave the hospital with protection. If nirsevimab is relied upon, I explain, their newborn may not be protected for some period of time.
Take-home Messages
- When patients decline or are hesitant about vaccines, ask why. Listen actively, and work to correct misperceptions and knowledge gaps.
- Whenever possible, offer vaccines in your practice. Vaccination rates drop when this does not occur.
- COVID-vaccine safety is backed by many studies showing no increase in birth defects, preterm delivery, miscarriage, or stillbirth.
- Pregnant women are more likely to have severe illness from the influenza and SARS-CoV-2 viruses. Vaccines can prevent severe illness and can protect the newborn as well as the mother.
- Recommend/administer the maternal RSV vaccine at 32-36 weeks’ gestation in women who did not receive the vaccine in the 2023-2024 season. If mothers aren’t eligible their babies should be offered nirsevimab.
Dr. Riley is the Given Foundation Professor and Chair of Obstetrics and Gynecology at Weill Cornell Medicine and the obstetrician and gynecologist-in-chief at New York Presbyterian Hospital. She disclosed that she has provided one-time consultations to Pfizer (Abrysvo RSV vaccine) and GSK (cytomegalovirus vaccine), and is providing consultant education on CMV for Moderna. She is chair of ACOG’s task force on immunization and emerging infectious diseases, serves on the medical advisory board for MAVEN, and serves as an editor or editorial board member for several medical publications.
References
1. ACOG Committee Opinion No. 741: Maternal Immunization. Obstet Gynecol. 2018;131(6):e214-e217.
2. Centers for Disease Control and Prevention. COVID-19 Vaccination for People Who are Pregnant or Breastfeeding. https://www.cdc.gov/covid/vaccines/pregnant-or-breastfeeding.html.
3. ACOG Practice Advisory on Maternal Respiratory Syncytial Virus Vaccination, September 2023. (Updated August 2024).4. Irving S et al. Open Forum Infect Dis. 2023;10(Suppl 2):ofad500.1002.
5. Flu Vaccination Dashboard, CDC, National Center for Immunization and Respiratory Diseases.
6. Weekly COVID-19 Vaccination Dashboard, CDC. https://www.cdc.gov/covidvaxview/weekly-dashboard/index.html
7. Louie JK et al. N Engl J Med. 2010;362:27-35. 8. Ciapponi A et al. Vaccine. 2021;39(40):5891-908.
9. Prasad S et al. Nature Communications. 2022;13:2414. 10. Fleming-Dutra KE et al. Obstet Gynecol Clin North Am 2023;50(2):279-97. 11. Mouen S et al. JAMA Network Open 2024;7(7):e2419268.
Editor’s Note: Sadly, this is the last column in the Master Class Obstetrics series. This award-winning column has been part of Ob.Gyn. News for 20 years. The deep discussion of cutting-edge topics in obstetrics by specialists and researchers will be missed as will the leadership and curation of topics by Dr. E. Albert Reece.
Introduction: The Need for Increased Vigilance About Maternal Immunization
Viruses are becoming increasingly prevalent in our world and the consequences of viral infections are implicated in a growing number of disease states. It is well established that certain cancers are caused by viruses and it is increasingly evident that viral infections can trigger the development of chronic illness. In pregnant women, viruses such as cytomegalovirus can cause infection in utero and lead to long-term impairments for the baby.
Likewise, it appears that the virulence of viruses is increasing, whether it be the respiratory syncytial virus (RSV) in children or the severe acute respiratory syndrome (SARS) coronaviruses in adults. Clearly, our environment is changing, with increases in population growth and urbanization, for instance, and an intensification of climate change and its effects. Viruses are part of this changing background.
Vaccines are our most powerful tool to protect people of all ages against viral threats, and fortunately, we benefit from increasing expertise in vaccinology. Since 1974, the University of Maryland School of Medicine has a Center for Vaccine Development and Global Health that has conducted research on vaccines to defend against the Zika virus, H1N1, Ebola, and SARS-CoV-2.
We’re not alone. Other vaccinology centers across the country — as well as the National Institutes of Health at the national level, through its National Institute of Allergy and Infectious Diseases — are doing research and developing vaccines to combat viral diseases.
In this column, we are focused on viral diseases in pregnancy and the role that vaccines can play in preventing serious respiratory illness in mothers and their newborns. I have invited Laura E. Riley, MD, the Given Foundation Professor and Chair of Obstetrics and Gynecology at Weill Cornell Medicine, to address the importance of maternal immunization and how we can best counsel our patients and improve immunization rates.
As Dr. Riley explains, we are in a new era, and it behooves us all to be more vigilant about recommending vaccines, combating misperceptions, addressing patients’ knowledge gaps, and administering vaccines whenever possible.
Dr. Reece is the former Dean of Medicine & University Executive VP, and The Distinguished University and Endowed Professor & Director of the Center for Advanced Research Training and Innovation (CARTI) at the University of Maryland School of Medicine, as well as senior scientist at the Center for Birth Defects Research.
The alarming decline in maternal immunization rates that occurred in the wake of the COVID-19 pandemic means that, now more than ever, we must fully embrace our responsibility to recommend immunizations in pregnancy and to communicate what is known about their efficacy and safety. Data show that vaccination rates drop when we do not offer vaccines in our offices, so whenever possible, we should administer them as well.
The ob.gyn. is the patient’s most trusted person in pregnancy. When patients decline or express hesitancy about vaccines, it is incumbent upon us to ask why. Oftentimes, we can identify areas in which patients lack knowledge or have misperceptions and we can successfully educate the patient or change their perspective or misunderstanding concerning the importance of vaccination for themselves and their babies. (See Table 1.) We can also successfully address concerns about safety.
The safety of COVID-19 vaccinations in pregnancy is now backed by several years of data from multiple studies showing no increase in birth defects, preterm delivery, miscarriage, or stillbirth.
Data also show that pregnant patients are more likely than patients who are not pregnant to need hospitalization and intensive care when infected with SARS-CoV-2 and are at risk of having complications that can affect pregnancy and the newborn, including preterm birth and stillbirth. Vaccination has been shown to reduce the risk of severe illness and the risk of such adverse obstetrical outcomes, in addition to providing protection for the infant early on.
Similarly, influenza has long been more likely to be severe in pregnant patients, with an increased risk of poor obstetrical outcomes. Vaccines similarly provide “two for one protection,” protecting both mother and baby, and are, of course, backed by many years of safety and efficacy data.
With the new maternal respiratory syncytial virus (RSV) vaccine, now in its second year of availability, the goal is to protect the baby from RSV-caused serious lower respiratory tract illness. The illness has contributed to tens of thousands of annual hospitalizations and up to several hundred deaths every year in children younger than 5 years — particularly in those under age 6 months.
The RSV monoclonal antibody nirsevimab is available for the newborn as an alternative to maternal immunization but the maternal vaccine is optimal in that it will provide immediate rather than delayed protection for the newborn. The maternal vaccine is recommended during weeks 32-36 of pregnancy in mothers who were not vaccinated during last year’s RSV season. With real-world experience from year one, the available safety data are reassuring.
Counseling About Influenza and COVID-19 Vaccination
The COVID-19 pandemic took a toll on vaccination interest/receptivity broadly in pregnant and nonpregnant people. Among pregnant individuals, influenza vaccination coverage declined from 71% in the 2019-2020 influenza season to 56% in the 2021-2022 season, according to data from the Centers for Disease Control and Prevention’s Vaccine Safety Datalink.4 Coverage for the 2022-2023 and 2023-2024 influenza seasons was even worse: well under 50%.5
Fewer pregnant women have received updated COVID-19 vaccines. Only 13% of pregnant persons overall received the updated 2023-2024 COVID-19 booster vaccine (through March 30, 2024), according to the CDC.6
Maternal immunization for influenza has been recommended in the United States since 2004 (part of the recommendation that everyone over the age of 6 months receive an annual flu vaccine), and flu vaccines have been given to millions of pregnant women, but the H1N1 pandemic of 2009 reinforced its value as a priority for prenatal care. Most of the women who became severely ill from the H1N1 virus were young and healthy, without co-existing conditions known to increase risk.7
It became clearer during the H1N1 pandemic that pregnancy itself — which is associated with physiologic changes such as decreased lung capacity, increased nasal congestion and changes in the immune system – is its own significant risk factor for severe illness from the influenza virus. This increased risk applies to COVID-19 as well.
As COVID-19 has become endemic, with hospitalizations and deaths not reaching the levels of previous surges — and with mask-wearing and other preventive measures having declined — patients understandably have become more complacent. Some patients are vaccine deniers, but in my practice, these patients are a much smaller group than those who believe COVID-19 “is no big deal,” especially if they have had infections recently.
This is why it’s important to actively listen to concerns and to ask patients who decline a vaccination why they are hesitant. Blanket messages about vaccine efficacy and safety are the first step, but individualized, more pointed conversations based on the patient’s personal experiences and beliefs have become increasingly important.
I routinely tell pregnant patients about the risks of COVID-19 and I explain that it has been difficult to predict who will develop severe illness. Sometimes more conversation is needed. For those who are still hesitant or who tell me they feel protected by a recent infection, for instance, I provide more detail on the unique risks of pregnancy — the fact that “pregnancy is different” — and that natural immunity wanes while the protection afforded by immunization is believed to last longer. Many women are also concerned about the safety of the COVID-19 vaccine, so having safety data at your fingertips is helpful. (See Table 2.)
The fact that influenza and COVID-19 vaccination protect the newborn as well as the mother is something that I find is underappreciated by many patients. Explaining that infants likely benefit from the passage of antibodies across the placenta should be part of patient counseling.
Counseling About RSV Vaccination
Importantly, for the 2024-2025 RSV season, the maternal RSV vaccine (Abrysvo, Pfizer) is recommended only for pregnant women who did not receive the vaccine during the 2023-2024 season. When more research is done and more data are obtained showing how long the immune response persists post vaccination, it may be that the US Food and Drug Administration (FDA) will approve the maternal RSV vaccine for use in every pregnancy.
The later timing of the vaccination recommendation — 32-36 weeks’ gestation — reflects a conservative approach taken by the FDA in response to data from one of the pivotal trials showing a numerical trend toward more preterm deliveries among vaccinated compared with unvaccinated patients. This imbalance in the original trial, which administered the vaccine during 24-36 weeks of gestation, was seen only in low-income countries with no temporal association, however.
In our experience at two Weill Cornell Medical College–associated hospitals we did not see this trend. Our cohort study of almost 3000 pregnant patients who delivered at 32 weeks’ gestation or later found no increased risk of preterm birth among the 35% of patients who received the RSV vaccine during the 2023-2024 RSV season. We also did not see any difference in preeclampsia, in contrast with original trial data that showed a signal for increased risk.11
When fewer than 2 weeks have elapsed between maternal vaccination and delivery, the monoclonal antibody nirsevimab is recommended for the newborn — ideally before the newborn leaves the hospital. Nirsevimab is also recommended for newborns of mothers who decline vaccination or were not candidates (e.g. vaccinated in a previous pregnancy), or when there is concern about the adequacy of the maternal immune response to the vaccine (e.g. in cases of immunosuppression).
While there was a limited supply of the monoclonal antibody last year, limitations are not expected this year, especially after October.
The ultimate goal is that patients choose the vaccine or the immunoglobulin, given the severity of RSV disease. Patient preferences should be considered. However, given that it takes 2 weeks after vaccination for protection to build up, I stress to patients that if they’ve vaccinated themselves, their newborn will leave the hospital with protection. If nirsevimab is relied upon, I explain, their newborn may not be protected for some period of time.
Take-home Messages
- When patients decline or are hesitant about vaccines, ask why. Listen actively, and work to correct misperceptions and knowledge gaps.
- Whenever possible, offer vaccines in your practice. Vaccination rates drop when this does not occur.
- COVID-vaccine safety is backed by many studies showing no increase in birth defects, preterm delivery, miscarriage, or stillbirth.
- Pregnant women are more likely to have severe illness from the influenza and SARS-CoV-2 viruses. Vaccines can prevent severe illness and can protect the newborn as well as the mother.
- Recommend/administer the maternal RSV vaccine at 32-36 weeks’ gestation in women who did not receive the vaccine in the 2023-2024 season. If mothers aren’t eligible their babies should be offered nirsevimab.
Dr. Riley is the Given Foundation Professor and Chair of Obstetrics and Gynecology at Weill Cornell Medicine and the obstetrician and gynecologist-in-chief at New York Presbyterian Hospital. She disclosed that she has provided one-time consultations to Pfizer (Abrysvo RSV vaccine) and GSK (cytomegalovirus vaccine), and is providing consultant education on CMV for Moderna. She is chair of ACOG’s task force on immunization and emerging infectious diseases, serves on the medical advisory board for MAVEN, and serves as an editor or editorial board member for several medical publications.
References
1. ACOG Committee Opinion No. 741: Maternal Immunization. Obstet Gynecol. 2018;131(6):e214-e217.
2. Centers for Disease Control and Prevention. COVID-19 Vaccination for People Who are Pregnant or Breastfeeding. https://www.cdc.gov/covid/vaccines/pregnant-or-breastfeeding.html.
3. ACOG Practice Advisory on Maternal Respiratory Syncytial Virus Vaccination, September 2023. (Updated August 2024).4. Irving S et al. Open Forum Infect Dis. 2023;10(Suppl 2):ofad500.1002.
5. Flu Vaccination Dashboard, CDC, National Center for Immunization and Respiratory Diseases.
6. Weekly COVID-19 Vaccination Dashboard, CDC. https://www.cdc.gov/covidvaxview/weekly-dashboard/index.html
7. Louie JK et al. N Engl J Med. 2010;362:27-35. 8. Ciapponi A et al. Vaccine. 2021;39(40):5891-908.
9. Prasad S et al. Nature Communications. 2022;13:2414. 10. Fleming-Dutra KE et al. Obstet Gynecol Clin North Am 2023;50(2):279-97. 11. Mouen S et al. JAMA Network Open 2024;7(7):e2419268.
Anticipated Effects of Pneumococcal Vaccines on Otitis
Acute otitis media (AOM) is caused by Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Since the introduction of pneumococcal conjugate vaccines (PCVs) shifts in the proportion of these three bacteria as causes of AOM and their antibiotic susceptibility profiles and strain diversity have occurred due to multiple factors including the PCVs and antibiotic selection pressure.
The 7-valent PCV (PCV7) was introduced in 2000 and was proven to be efficacious in preventing AOM, but no subsequent PCV has received an indication for prevention of AOM because the FDA required a tympanocentesis study to prove efficacy and that approval was not achieved for PCV13, PCV15, or PCV20. This is a little known fact. After introduction of PCV7, replacement pneumococcal strains expressing serotypes not in PCV7 emerged and antibiotic non-susceptible strains became predominant causes of AOM, especially antibiotic-resistant serotype 19A. To address the phenomena of pneumococcal serotype replacement, PCV13 was introduced in 2010. But serotype replacement continued to occur under PCV13 pressure, replacement serotypes increasingly caused AOM, and antibiotic-resistant serotype 35B emerged. Now we have two new higher valency PCVs: PCV15 (Merck) where serotypes 22F and 33F were added to the PCV13 serotypes and PCV20 (Pfizer) where 22F, 33F, 8, 10A, 11A, 12F, 15B were added to PCV13. Note that neither PCV15 nor PCV20 includes the most common serotype causing AOM – serotype 35B.1
While PCV15 and PCV20 should provide protection against more pneumococcal serotypes, increasing serotypes in both vaccines decreased immunogenicity of certain shared serotypes, more so with the addition of seven more in PCV20 than two more in PCV15, compared with PCV13. Whether lower antibody concentrations will make a difference clinically in terms of vaccine failure to prevent nasopharyngeal colonization, AOM, and/or invasive pneumococcal infections is currently unknown.
Our group from greater Rochester, New York, is the only one in the United States performing tympanocentesis to determine the etiology of AOM infections. Children between ages 6 and 36 months are studied. We recently reported our results for the time span September 2021 to September 2023, the immediate 2 years prior to recommendations for use of PCV15 and PCV20 in young children.2 Tympanocentesis was performed in 139 (78%) of 179 episodes of AOM, yielding 216 middle ear fluid samples (the higher number of middle ear fluids was due to bilateral tympanocentesis in some children). H. influenzae (40%) was the most common bacterial isolate, followed by S. pneumonia (19%) and M. catarrhalis (17%), with the remainder no growth. Polymerase chain reactions (PCR) was positive in many of those culture negative samples, suggesting prior use of antibiotics before tympanocentesis was performed. Among the pneumococcal isolates, 46% were oxacillin non-susceptible. Among the H. influenzae isolates, 27% were beta-lactamase producing and all M. catarrhalis were beta-lactamase-producing.
As we previously reported,1 we once again found that serotype 35B was the most frequent non-PCV15, non-PCV20, serotype. Other frequently detected non-PCV20 pneumococcal serotypes were 23A, 23B, 35D, 35F and 15C.2
Projected Pneumococcal Serotype Coverage by PCV15 and PCV20
PCV13 serotypes were identified in 9% of middle ear fluids, consistent with vaccine failure. Assuming 100% vaccine-type effectiveness, PCV15 will provide about 11% coverage of pneumococci causing AOM, the same PCV13 and PCV20 will provide 30% coverage, leaving 70% of pneumococci causing AOM in young children uncovered (Figure).
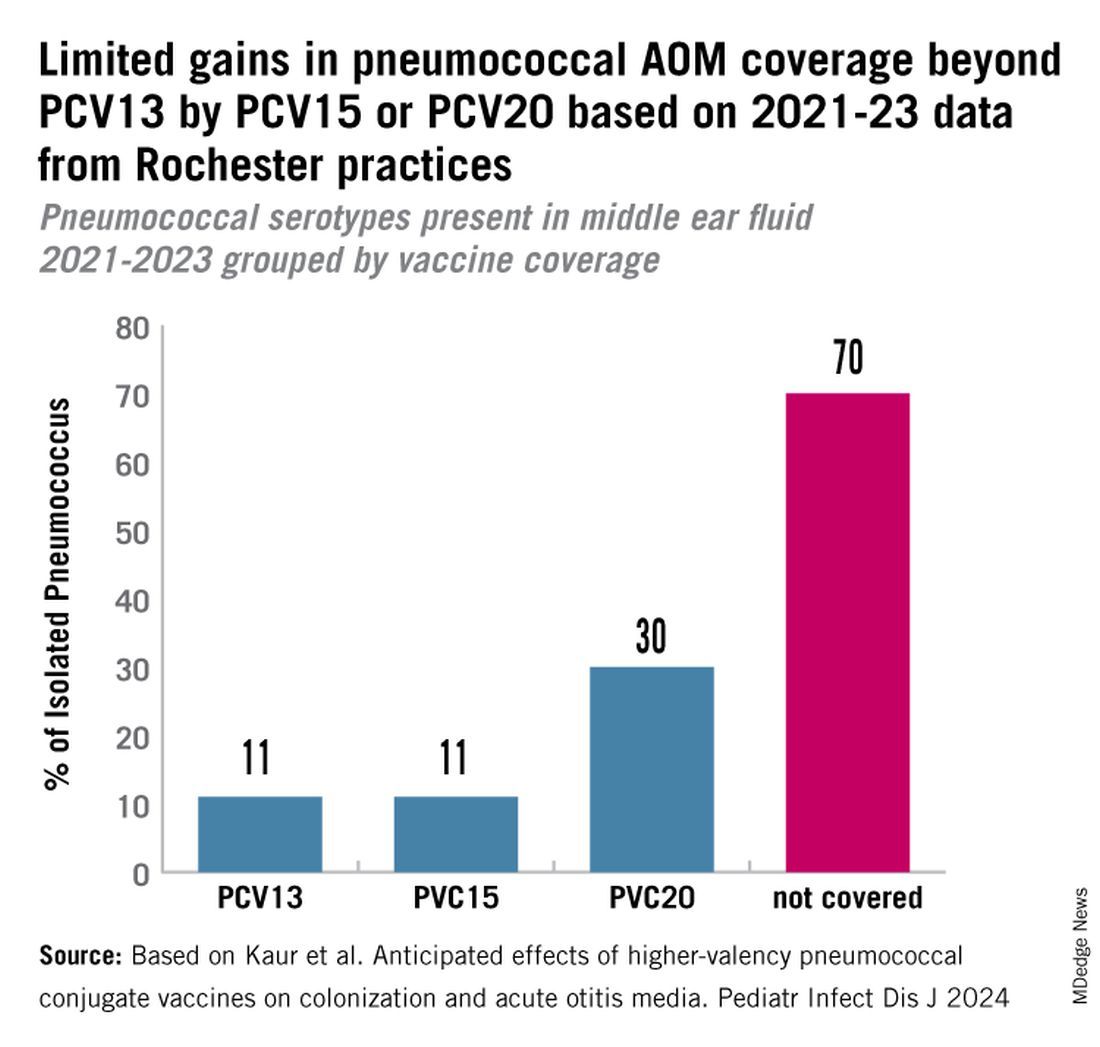
Thus, the high proportion of pneumococcal serotype 35B and other non-PCV15 or non-PCV20 serotypes will result in a relatively small incremental benefit over PCV13 in young children for AOM.
AOM is the most common cause of pediatric outpatient visits and antibiotic prescriptions in the United States that contributes to selection of antibiotic-resistant microbes.3 The economic burden of AOM is high, estimated at about $3 billion annually in the United States, when direct and indirect costs are calculated,4 thereby making AOM a major factor in calculations of cost effectiveness analyses of PCV immunizations in children.
While PCV15 and PCV20 include common serotypes associated with invasive pneumococcal diseases, their effectiveness in preventing AOM, acute sinusitis, and non-bacteremic community-acquired pneumonia is currently unknown because these vaccines were licensed based on safety and immunogenicity data, not proven efficacy.
The data on antibiotic susceptibility of pneumococci and H. influenza and M. catarrhalis isolated in the late post PCV13 era from young children in a pediatric primary-care setting raise a question about empiric antibiotic choice for AOM today. For penicillin non-susceptible pneumococcal strains, higher dosages of amoxicillin can improve eradication. However, higher dosages of amoxicillin cannot overcome beta-lactamase production by H. influenza and M. catarrhalis. Based on the mix of pathogens causing AOM and the antibiotic susceptibility of those bacteria, high-dose amoxicillin/clavulanate or alternative cephalosporin drugs active against pneumococci and beta-lactamase producing H. influenza and M. catarrhalis would be a better empiric choice over high-dose amoxicillin.
Limitations of our study include that it occurred in one center in New York, although we have previously shown results of tympanocentesis at our center are similar to those in Virginia and Pennsylvania5 and our study population was composed of children living in urban, suburban, and rural households of all economic levels. Because this study was conducted during a relatively short time frame (2021-2023), the numbers of subjects and samples were sometimes insufficient to identify statistically significant differences in some comparisons. Some children were lost to follow-up, and not every participant was consented for tympanocentesis. Some participants received antibiotics prior to middle ear fluid specimen collection.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Dynamic Changes in Otopathogens Colonizing the Nasopharynx and Causing Acute Otitis Media in Children After 13-Valent (PCV13) Pneumococcal Conjugate Vaccination During 2015-2019. Eur J Clin Microbiol Infect Dis. 2022 Jan;41(1):37-44. doi: 10.1007/s10096-021-04324-0.
2. Kaur R et al. Anticipated Effects of Higher-valency Pneumococcal Conjugate Vaccines on Colonization and Acute Otitis Media. Pediatr Infect Dis J. 2024 Oct 1;43(10):1004-1010. doi: 10.1097/INF.0000000000004413.
3. King LM et al. Pediatric Outpatient Visits and Antibiotic Use Attributable to Higher Valency Pneumococcal Conjugate Vaccine Serotypes. medRxiv [Preprint]. 2023 Aug 25:2023.08.24.23294570. doi: 10.1101/2023.08.24.23294570.
4. Ahmed S et al. Incremental Health Care Utilization and Costs for Acute Otitis Media in Children. Laryngoscope. 2014 Jan;124(1):301-5. doi: 10.1002/lary.24190.
5. Pichichero ME et al. Pathogens Causing Recurrent and Difficult-to-Treat Acute Otitis Media, 2003-2006. Clin Pediatr (Phila). 2008 Nov;47(9):901-6. doi: 10.1177/0009922808319966.
Acute otitis media (AOM) is caused by Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Since the introduction of pneumococcal conjugate vaccines (PCVs) shifts in the proportion of these three bacteria as causes of AOM and their antibiotic susceptibility profiles and strain diversity have occurred due to multiple factors including the PCVs and antibiotic selection pressure.
The 7-valent PCV (PCV7) was introduced in 2000 and was proven to be efficacious in preventing AOM, but no subsequent PCV has received an indication for prevention of AOM because the FDA required a tympanocentesis study to prove efficacy and that approval was not achieved for PCV13, PCV15, or PCV20. This is a little known fact. After introduction of PCV7, replacement pneumococcal strains expressing serotypes not in PCV7 emerged and antibiotic non-susceptible strains became predominant causes of AOM, especially antibiotic-resistant serotype 19A. To address the phenomena of pneumococcal serotype replacement, PCV13 was introduced in 2010. But serotype replacement continued to occur under PCV13 pressure, replacement serotypes increasingly caused AOM, and antibiotic-resistant serotype 35B emerged. Now we have two new higher valency PCVs: PCV15 (Merck) where serotypes 22F and 33F were added to the PCV13 serotypes and PCV20 (Pfizer) where 22F, 33F, 8, 10A, 11A, 12F, 15B were added to PCV13. Note that neither PCV15 nor PCV20 includes the most common serotype causing AOM – serotype 35B.1
While PCV15 and PCV20 should provide protection against more pneumococcal serotypes, increasing serotypes in both vaccines decreased immunogenicity of certain shared serotypes, more so with the addition of seven more in PCV20 than two more in PCV15, compared with PCV13. Whether lower antibody concentrations will make a difference clinically in terms of vaccine failure to prevent nasopharyngeal colonization, AOM, and/or invasive pneumococcal infections is currently unknown.
Our group from greater Rochester, New York, is the only one in the United States performing tympanocentesis to determine the etiology of AOM infections. Children between ages 6 and 36 months are studied. We recently reported our results for the time span September 2021 to September 2023, the immediate 2 years prior to recommendations for use of PCV15 and PCV20 in young children.2 Tympanocentesis was performed in 139 (78%) of 179 episodes of AOM, yielding 216 middle ear fluid samples (the higher number of middle ear fluids was due to bilateral tympanocentesis in some children). H. influenzae (40%) was the most common bacterial isolate, followed by S. pneumonia (19%) and M. catarrhalis (17%), with the remainder no growth. Polymerase chain reactions (PCR) was positive in many of those culture negative samples, suggesting prior use of antibiotics before tympanocentesis was performed. Among the pneumococcal isolates, 46% were oxacillin non-susceptible. Among the H. influenzae isolates, 27% were beta-lactamase producing and all M. catarrhalis were beta-lactamase-producing.
As we previously reported,1 we once again found that serotype 35B was the most frequent non-PCV15, non-PCV20, serotype. Other frequently detected non-PCV20 pneumococcal serotypes were 23A, 23B, 35D, 35F and 15C.2
Projected Pneumococcal Serotype Coverage by PCV15 and PCV20
PCV13 serotypes were identified in 9% of middle ear fluids, consistent with vaccine failure. Assuming 100% vaccine-type effectiveness, PCV15 will provide about 11% coverage of pneumococci causing AOM, the same PCV13 and PCV20 will provide 30% coverage, leaving 70% of pneumococci causing AOM in young children uncovered (Figure).

Thus, the high proportion of pneumococcal serotype 35B and other non-PCV15 or non-PCV20 serotypes will result in a relatively small incremental benefit over PCV13 in young children for AOM.
AOM is the most common cause of pediatric outpatient visits and antibiotic prescriptions in the United States that contributes to selection of antibiotic-resistant microbes.3 The economic burden of AOM is high, estimated at about $3 billion annually in the United States, when direct and indirect costs are calculated,4 thereby making AOM a major factor in calculations of cost effectiveness analyses of PCV immunizations in children.
While PCV15 and PCV20 include common serotypes associated with invasive pneumococcal diseases, their effectiveness in preventing AOM, acute sinusitis, and non-bacteremic community-acquired pneumonia is currently unknown because these vaccines were licensed based on safety and immunogenicity data, not proven efficacy.
The data on antibiotic susceptibility of pneumococci and H. influenza and M. catarrhalis isolated in the late post PCV13 era from young children in a pediatric primary-care setting raise a question about empiric antibiotic choice for AOM today. For penicillin non-susceptible pneumococcal strains, higher dosages of amoxicillin can improve eradication. However, higher dosages of amoxicillin cannot overcome beta-lactamase production by H. influenza and M. catarrhalis. Based on the mix of pathogens causing AOM and the antibiotic susceptibility of those bacteria, high-dose amoxicillin/clavulanate or alternative cephalosporin drugs active against pneumococci and beta-lactamase producing H. influenza and M. catarrhalis would be a better empiric choice over high-dose amoxicillin.
Limitations of our study include that it occurred in one center in New York, although we have previously shown results of tympanocentesis at our center are similar to those in Virginia and Pennsylvania5 and our study population was composed of children living in urban, suburban, and rural households of all economic levels. Because this study was conducted during a relatively short time frame (2021-2023), the numbers of subjects and samples were sometimes insufficient to identify statistically significant differences in some comparisons. Some children were lost to follow-up, and not every participant was consented for tympanocentesis. Some participants received antibiotics prior to middle ear fluid specimen collection.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Dynamic Changes in Otopathogens Colonizing the Nasopharynx and Causing Acute Otitis Media in Children After 13-Valent (PCV13) Pneumococcal Conjugate Vaccination During 2015-2019. Eur J Clin Microbiol Infect Dis. 2022 Jan;41(1):37-44. doi: 10.1007/s10096-021-04324-0.
2. Kaur R et al. Anticipated Effects of Higher-valency Pneumococcal Conjugate Vaccines on Colonization and Acute Otitis Media. Pediatr Infect Dis J. 2024 Oct 1;43(10):1004-1010. doi: 10.1097/INF.0000000000004413.
3. King LM et al. Pediatric Outpatient Visits and Antibiotic Use Attributable to Higher Valency Pneumococcal Conjugate Vaccine Serotypes. medRxiv [Preprint]. 2023 Aug 25:2023.08.24.23294570. doi: 10.1101/2023.08.24.23294570.
4. Ahmed S et al. Incremental Health Care Utilization and Costs for Acute Otitis Media in Children. Laryngoscope. 2014 Jan;124(1):301-5. doi: 10.1002/lary.24190.
5. Pichichero ME et al. Pathogens Causing Recurrent and Difficult-to-Treat Acute Otitis Media, 2003-2006. Clin Pediatr (Phila). 2008 Nov;47(9):901-6. doi: 10.1177/0009922808319966.
Acute otitis media (AOM) is caused by Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Since the introduction of pneumococcal conjugate vaccines (PCVs) shifts in the proportion of these three bacteria as causes of AOM and their antibiotic susceptibility profiles and strain diversity have occurred due to multiple factors including the PCVs and antibiotic selection pressure.
The 7-valent PCV (PCV7) was introduced in 2000 and was proven to be efficacious in preventing AOM, but no subsequent PCV has received an indication for prevention of AOM because the FDA required a tympanocentesis study to prove efficacy and that approval was not achieved for PCV13, PCV15, or PCV20. This is a little known fact. After introduction of PCV7, replacement pneumococcal strains expressing serotypes not in PCV7 emerged and antibiotic non-susceptible strains became predominant causes of AOM, especially antibiotic-resistant serotype 19A. To address the phenomena of pneumococcal serotype replacement, PCV13 was introduced in 2010. But serotype replacement continued to occur under PCV13 pressure, replacement serotypes increasingly caused AOM, and antibiotic-resistant serotype 35B emerged. Now we have two new higher valency PCVs: PCV15 (Merck) where serotypes 22F and 33F were added to the PCV13 serotypes and PCV20 (Pfizer) where 22F, 33F, 8, 10A, 11A, 12F, 15B were added to PCV13. Note that neither PCV15 nor PCV20 includes the most common serotype causing AOM – serotype 35B.1
While PCV15 and PCV20 should provide protection against more pneumococcal serotypes, increasing serotypes in both vaccines decreased immunogenicity of certain shared serotypes, more so with the addition of seven more in PCV20 than two more in PCV15, compared with PCV13. Whether lower antibody concentrations will make a difference clinically in terms of vaccine failure to prevent nasopharyngeal colonization, AOM, and/or invasive pneumococcal infections is currently unknown.
Our group from greater Rochester, New York, is the only one in the United States performing tympanocentesis to determine the etiology of AOM infections. Children between ages 6 and 36 months are studied. We recently reported our results for the time span September 2021 to September 2023, the immediate 2 years prior to recommendations for use of PCV15 and PCV20 in young children.2 Tympanocentesis was performed in 139 (78%) of 179 episodes of AOM, yielding 216 middle ear fluid samples (the higher number of middle ear fluids was due to bilateral tympanocentesis in some children). H. influenzae (40%) was the most common bacterial isolate, followed by S. pneumonia (19%) and M. catarrhalis (17%), with the remainder no growth. Polymerase chain reactions (PCR) was positive in many of those culture negative samples, suggesting prior use of antibiotics before tympanocentesis was performed. Among the pneumococcal isolates, 46% were oxacillin non-susceptible. Among the H. influenzae isolates, 27% were beta-lactamase producing and all M. catarrhalis were beta-lactamase-producing.
As we previously reported,1 we once again found that serotype 35B was the most frequent non-PCV15, non-PCV20, serotype. Other frequently detected non-PCV20 pneumococcal serotypes were 23A, 23B, 35D, 35F and 15C.2
Projected Pneumococcal Serotype Coverage by PCV15 and PCV20
PCV13 serotypes were identified in 9% of middle ear fluids, consistent with vaccine failure. Assuming 100% vaccine-type effectiveness, PCV15 will provide about 11% coverage of pneumococci causing AOM, the same PCV13 and PCV20 will provide 30% coverage, leaving 70% of pneumococci causing AOM in young children uncovered (Figure).

Thus, the high proportion of pneumococcal serotype 35B and other non-PCV15 or non-PCV20 serotypes will result in a relatively small incremental benefit over PCV13 in young children for AOM.
AOM is the most common cause of pediatric outpatient visits and antibiotic prescriptions in the United States that contributes to selection of antibiotic-resistant microbes.3 The economic burden of AOM is high, estimated at about $3 billion annually in the United States, when direct and indirect costs are calculated,4 thereby making AOM a major factor in calculations of cost effectiveness analyses of PCV immunizations in children.
While PCV15 and PCV20 include common serotypes associated with invasive pneumococcal diseases, their effectiveness in preventing AOM, acute sinusitis, and non-bacteremic community-acquired pneumonia is currently unknown because these vaccines were licensed based on safety and immunogenicity data, not proven efficacy.
The data on antibiotic susceptibility of pneumococci and H. influenza and M. catarrhalis isolated in the late post PCV13 era from young children in a pediatric primary-care setting raise a question about empiric antibiotic choice for AOM today. For penicillin non-susceptible pneumococcal strains, higher dosages of amoxicillin can improve eradication. However, higher dosages of amoxicillin cannot overcome beta-lactamase production by H. influenza and M. catarrhalis. Based on the mix of pathogens causing AOM and the antibiotic susceptibility of those bacteria, high-dose amoxicillin/clavulanate or alternative cephalosporin drugs active against pneumococci and beta-lactamase producing H. influenza and M. catarrhalis would be a better empiric choice over high-dose amoxicillin.
Limitations of our study include that it occurred in one center in New York, although we have previously shown results of tympanocentesis at our center are similar to those in Virginia and Pennsylvania5 and our study population was composed of children living in urban, suburban, and rural households of all economic levels. Because this study was conducted during a relatively short time frame (2021-2023), the numbers of subjects and samples were sometimes insufficient to identify statistically significant differences in some comparisons. Some children were lost to follow-up, and not every participant was consented for tympanocentesis. Some participants received antibiotics prior to middle ear fluid specimen collection.
Dr. Pichichero is a specialist in pediatric infectious diseases, Center for Infectious Diseases and Immunology, and director of the Research Institute, at Rochester (N.Y.) General Hospital. He has no conflicts of interest to declare.
References
1. Kaur R et al. Dynamic Changes in Otopathogens Colonizing the Nasopharynx and Causing Acute Otitis Media in Children After 13-Valent (PCV13) Pneumococcal Conjugate Vaccination During 2015-2019. Eur J Clin Microbiol Infect Dis. 2022 Jan;41(1):37-44. doi: 10.1007/s10096-021-04324-0.
2. Kaur R et al. Anticipated Effects of Higher-valency Pneumococcal Conjugate Vaccines on Colonization and Acute Otitis Media. Pediatr Infect Dis J. 2024 Oct 1;43(10):1004-1010. doi: 10.1097/INF.0000000000004413.
3. King LM et al. Pediatric Outpatient Visits and Antibiotic Use Attributable to Higher Valency Pneumococcal Conjugate Vaccine Serotypes. medRxiv [Preprint]. 2023 Aug 25:2023.08.24.23294570. doi: 10.1101/2023.08.24.23294570.
4. Ahmed S et al. Incremental Health Care Utilization and Costs for Acute Otitis Media in Children. Laryngoscope. 2014 Jan;124(1):301-5. doi: 10.1002/lary.24190.
5. Pichichero ME et al. Pathogens Causing Recurrent and Difficult-to-Treat Acute Otitis Media, 2003-2006. Clin Pediatr (Phila). 2008 Nov;47(9):901-6. doi: 10.1177/0009922808319966.








